User login
Monkeypox: What FPs need to know, now
The Centers for Disease Control and Prevention (CDC) and the World Health Organization are investigating an outbreak of monkeypox cases that have occurred around the world in countries that do not have endemic monkeypox virus.1,2 As of July 5, there have been 6924 cases documented in 52 countries, including 560 cases that have occurred in the United States.2 In the United States, as well as globally, a large proportion of cases have been in men who have sex with men.
First, what is monkeypox? Monkeypox is an orthopox virus that is closely related to variola (smallpox) and vaccinia (the virus used in the smallpox vaccine). It is endemic in western and central Africa and is contracted by contact with an infected mammal (including humans). Transmission can occur through direct contact with infected body fluids or lesions, via infectious fomites, or through respiratory secretions (although this usually requires prolonged exposure).
What is the disease course? The incubation period is 4 to 17 days. The initial symptoms include fever, malaise, headache, sore throat, and lymphadenopathy. A rash erupts 1 to 4 days after the prodrome and progresses synchronously from macules to papules to vesicles and then to pustules, which eventually scab over and fall off. In some cases reported in the United States, the rash started in the groin and genital area.
Don’t be fooled by other exanthems. Monkeypox can be confused with chickenpox and molluscum contagiosum (MC). However, the lesions in chickenpox appear asynchronously (all 4 stages present at the same time) and the papules of MC contain a central pit.
Can monkeypox be prevented? There are currently 2 vaccines against orthopox viruses: ACAM2000 and Jynneos. Currently, these vaccines are routinely recommended only for those at occupational risk of orthopox exposure.3
What you should know—and do. Be alert for any patient who presents with a suspicious rash; if there is a possibility of monkeypox, the local public health department should be contacted. They will investigate and collect samples for laboratory testing and will elicit contact names and locations. If monkeypox is confirmed, they may offer close contacts 1 of the 2 vaccines, which if administered within 4 days of exposure can prevent infection.
Advise all patients confirmed to have monkeypox to self-isolate until all skin lesions have healed. Good infection control practices in the clinical setting will prevent spread to staff and other patients.
More information about monkeypox, including images of typical lesions—as well as an update on the current investigation in the United States and worldwide—can be found on the CDC website.4
1. Minhaj FS, Ogale YP, Whitehill F, et al. Monkeypox outbreak—nine states, May 2022. MMWR Morbid Mortal Wkly Rep. 2022;71:764-769. doi: http://dx.doi.org/10.15585/mmwr.mm7123e1
2. CDC. US monkeypox outbreak 2022: situation summary. Updated June 29, 2022. Accessed July 5, 2022.
3. Rao AK, Petersen BW, Whitehill F, et al. Use of JYNNEOS (smallpox and monkeypox vaccine, live, nonreplicating) for preexposure vaccination of persons at risk for occupational exposure to orthopoxviruses: recommendations of the Advisory Committee on Immunization Practices—United States, 2022. MMWR Morbid Mortal Wkly Rep. 2022;71:734-742. doi: http://dx.doi.org/10.15585/mmwr.mm7122e1
4. CDC. 2022 monkeypox: information for healthcare professionals. Updated June 23, 2022. Accessed July 5, 2022.
The Centers for Disease Control and Prevention (CDC) and the World Health Organization are investigating an outbreak of monkeypox cases that have occurred around the world in countries that do not have endemic monkeypox virus.1,2 As of July 5, there have been 6924 cases documented in 52 countries, including 560 cases that have occurred in the United States.2 In the United States, as well as globally, a large proportion of cases have been in men who have sex with men.
First, what is monkeypox? Monkeypox is an orthopox virus that is closely related to variola (smallpox) and vaccinia (the virus used in the smallpox vaccine). It is endemic in western and central Africa and is contracted by contact with an infected mammal (including humans). Transmission can occur through direct contact with infected body fluids or lesions, via infectious fomites, or through respiratory secretions (although this usually requires prolonged exposure).
What is the disease course? The incubation period is 4 to 17 days. The initial symptoms include fever, malaise, headache, sore throat, and lymphadenopathy. A rash erupts 1 to 4 days after the prodrome and progresses synchronously from macules to papules to vesicles and then to pustules, which eventually scab over and fall off. In some cases reported in the United States, the rash started in the groin and genital area.
Don’t be fooled by other exanthems. Monkeypox can be confused with chickenpox and molluscum contagiosum (MC). However, the lesions in chickenpox appear asynchronously (all 4 stages present at the same time) and the papules of MC contain a central pit.
Can monkeypox be prevented? There are currently 2 vaccines against orthopox viruses: ACAM2000 and Jynneos. Currently, these vaccines are routinely recommended only for those at occupational risk of orthopox exposure.3
What you should know—and do. Be alert for any patient who presents with a suspicious rash; if there is a possibility of monkeypox, the local public health department should be contacted. They will investigate and collect samples for laboratory testing and will elicit contact names and locations. If monkeypox is confirmed, they may offer close contacts 1 of the 2 vaccines, which if administered within 4 days of exposure can prevent infection.
Advise all patients confirmed to have monkeypox to self-isolate until all skin lesions have healed. Good infection control practices in the clinical setting will prevent spread to staff and other patients.
More information about monkeypox, including images of typical lesions—as well as an update on the current investigation in the United States and worldwide—can be found on the CDC website.4
The Centers for Disease Control and Prevention (CDC) and the World Health Organization are investigating an outbreak of monkeypox cases that have occurred around the world in countries that do not have endemic monkeypox virus.1,2 As of July 5, there have been 6924 cases documented in 52 countries, including 560 cases that have occurred in the United States.2 In the United States, as well as globally, a large proportion of cases have been in men who have sex with men.
First, what is monkeypox? Monkeypox is an orthopox virus that is closely related to variola (smallpox) and vaccinia (the virus used in the smallpox vaccine). It is endemic in western and central Africa and is contracted by contact with an infected mammal (including humans). Transmission can occur through direct contact with infected body fluids or lesions, via infectious fomites, or through respiratory secretions (although this usually requires prolonged exposure).
What is the disease course? The incubation period is 4 to 17 days. The initial symptoms include fever, malaise, headache, sore throat, and lymphadenopathy. A rash erupts 1 to 4 days after the prodrome and progresses synchronously from macules to papules to vesicles and then to pustules, which eventually scab over and fall off. In some cases reported in the United States, the rash started in the groin and genital area.
Don’t be fooled by other exanthems. Monkeypox can be confused with chickenpox and molluscum contagiosum (MC). However, the lesions in chickenpox appear asynchronously (all 4 stages present at the same time) and the papules of MC contain a central pit.
Can monkeypox be prevented? There are currently 2 vaccines against orthopox viruses: ACAM2000 and Jynneos. Currently, these vaccines are routinely recommended only for those at occupational risk of orthopox exposure.3
What you should know—and do. Be alert for any patient who presents with a suspicious rash; if there is a possibility of monkeypox, the local public health department should be contacted. They will investigate and collect samples for laboratory testing and will elicit contact names and locations. If monkeypox is confirmed, they may offer close contacts 1 of the 2 vaccines, which if administered within 4 days of exposure can prevent infection.
Advise all patients confirmed to have monkeypox to self-isolate until all skin lesions have healed. Good infection control practices in the clinical setting will prevent spread to staff and other patients.
More information about monkeypox, including images of typical lesions—as well as an update on the current investigation in the United States and worldwide—can be found on the CDC website.4
1. Minhaj FS, Ogale YP, Whitehill F, et al. Monkeypox outbreak—nine states, May 2022. MMWR Morbid Mortal Wkly Rep. 2022;71:764-769. doi: http://dx.doi.org/10.15585/mmwr.mm7123e1
2. CDC. US monkeypox outbreak 2022: situation summary. Updated June 29, 2022. Accessed July 5, 2022.
3. Rao AK, Petersen BW, Whitehill F, et al. Use of JYNNEOS (smallpox and monkeypox vaccine, live, nonreplicating) for preexposure vaccination of persons at risk for occupational exposure to orthopoxviruses: recommendations of the Advisory Committee on Immunization Practices—United States, 2022. MMWR Morbid Mortal Wkly Rep. 2022;71:734-742. doi: http://dx.doi.org/10.15585/mmwr.mm7122e1
4. CDC. 2022 monkeypox: information for healthcare professionals. Updated June 23, 2022. Accessed July 5, 2022.
1. Minhaj FS, Ogale YP, Whitehill F, et al. Monkeypox outbreak—nine states, May 2022. MMWR Morbid Mortal Wkly Rep. 2022;71:764-769. doi: http://dx.doi.org/10.15585/mmwr.mm7123e1
2. CDC. US monkeypox outbreak 2022: situation summary. Updated June 29, 2022. Accessed July 5, 2022.
3. Rao AK, Petersen BW, Whitehill F, et al. Use of JYNNEOS (smallpox and monkeypox vaccine, live, nonreplicating) for preexposure vaccination of persons at risk for occupational exposure to orthopoxviruses: recommendations of the Advisory Committee on Immunization Practices—United States, 2022. MMWR Morbid Mortal Wkly Rep. 2022;71:734-742. doi: http://dx.doi.org/10.15585/mmwr.mm7122e1
4. CDC. 2022 monkeypox: information for healthcare professionals. Updated June 23, 2022. Accessed July 5, 2022.
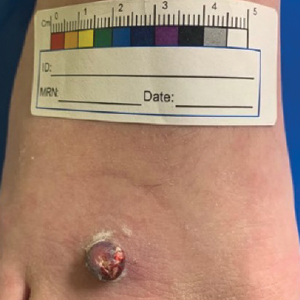
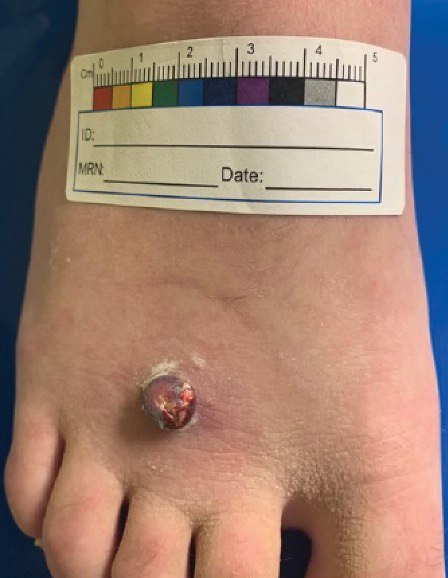
Erythematous Pedunculated Plaque on the Dorsal Aspect of the Foot
The Diagnosis: Molluscum Contagiosum
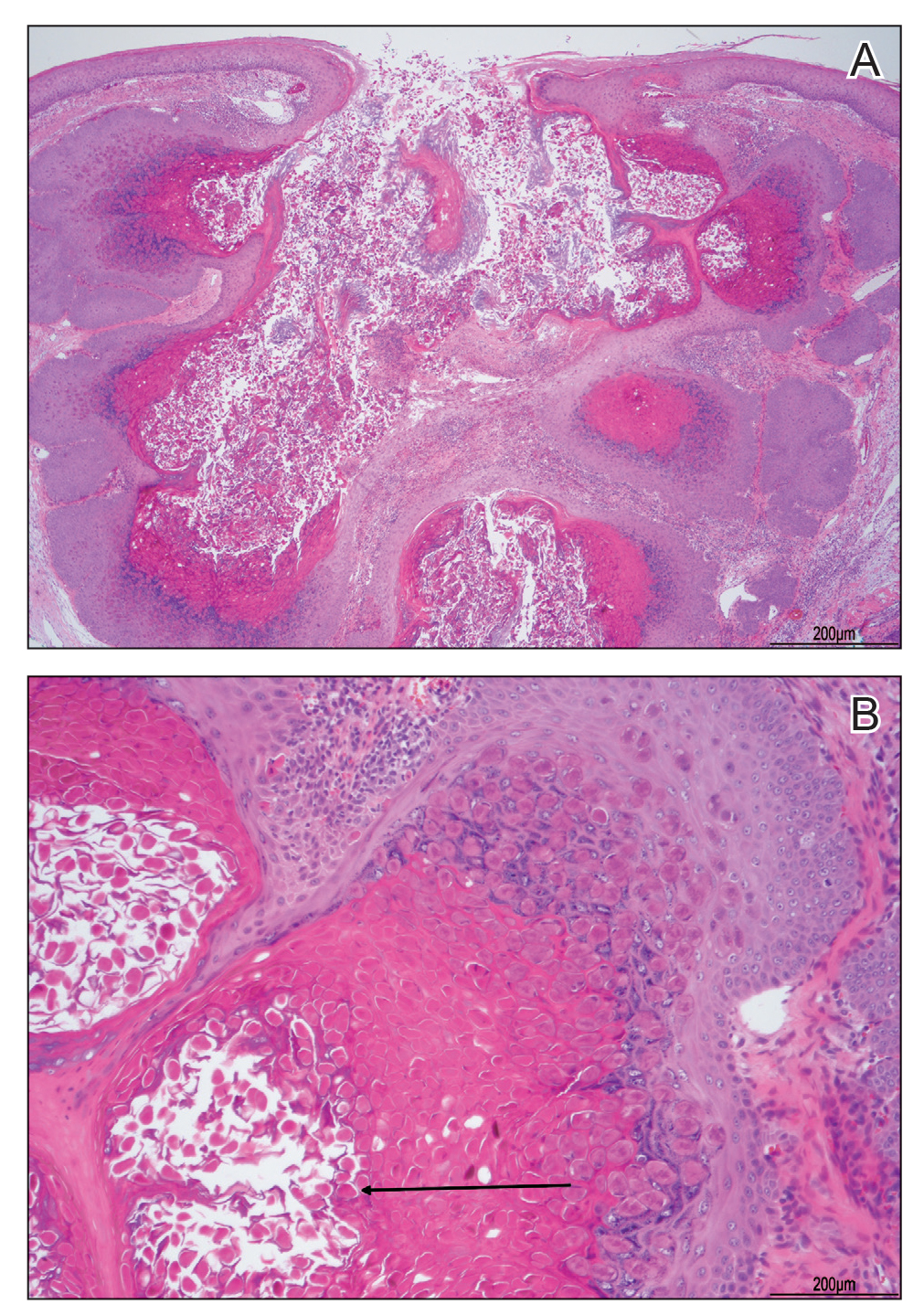
A tangential shave removal with electrocautery was performed. Histopathology demonstrated numerous eosinophilic intracytoplasmic inclusion bodies (Figure), confirming a diagnosis of molluscum contagiosum (MC).
Molluscum contagiosum is a common poxvirus infection that is transmitted through fomites, contact, or self-inoculation.1 This infection most frequently occurs in school-aged children younger than 8 years1-3; peak incidence is 6 years of age.2,3 The worldwide estimated prevalence in children is 5.1% to 11.5%.1,3 In children cohabitating with others infected by MC, approximately 40% of households experienced a spread of infection; the risk of transmission is not associated with greater number of lesions.4 In adults, infection most commonly occurs in the setting of immunodeficiency or as a sexually transmitted infection in immunocompetent patients.3 Molluscum contagiosum infection classically presents as 1- to 3-mm, flesh- or white-colored, dome-shaped, smooth papules with central umbilication.1 Lesions often occur in clusters or lines, indicating local spread. The trunk, extremities, and face are areas that frequently are involved.2,3
Atypical presentations of MC infection can occur, as demonstrated by our case. Involvement of hair follicles by the infection can result in follicular induction.1,5 Secondary infection can mimic abscess formation.1 Inflamed MC lesions demonstrating the “beginning of the end” sign often are mistaken for primary infection, which is thought to be an inflammatory immune response to the virus.6 Lesions located on the eye or eyelid can present as unilateral conjunctivitis, conjunctival or corneal nodules, eyelid abscesses, or chalazions.1 Giant MC is a nodular variant of this infection measuring larger than 1 cm in size that can present similar to epidermoid cysts, condyloma acuminatum, or verruca vulgaris.1,7 Other reported mimicked conditions include basal cell carcinoma, trichoepithelioma, appendageal tumors, keratoacanthoma, foreign body granulomas, nevus sebaceous, or ecthyma.1,3 Molluscum contagiosum also has been reported to present as large ulcerative growths.8 In immunocompromised patients, deep fungal infection is another mimicker.1 Lesions on the plantar surfaces of the feet often are misdiagnosed as plantar verruca and present with pain during ambulation.9
The diagnosis of MC is clinical, with additional diagnostic tools reserved for more challenging situations.1 In cases with atypical presentations, dermoscopy may aid diagnosis through visualization of orifices and vascular patterns including crown, radial, and punctiform vessels.10 Biopsy or fine-needle aspiration also can be utilized as a diagnostic tool. Histopathology often reveals pathognomonic intracytoplasmic inclusions or Henderson-Paterson bodies.8,10 The appearance of MC can mimic other conditions that should be included in the differential diagnosis. Pyogenic granuloma often presents as a benign red papule that may grow rapidly and become pedunculated, sometimes with bleeding and crusting, though histology reveals groups of proliferating capillaries.11 More than half of amelanotic melanomas present in the papulonodular form as vascular or ulcerated nodules, and others may appear as erythematous macules. Diagnosis of amelanotic melanoma is made through histologic examination, which reveals atypical melanocytes in nests or cords, in conjunction with immunohistochemical stains such as S-100.12 Spitz nevi often appear as round, dome-shaped papules that most commonly are red, pink, or fleshcolored. They appear histologically similar to melanoma with nests of atypical melanocytes and nuclear atypia.13
A variety of treatment modalities can be used for MC including cantharidin, curettage, and cryotherapy.14 Imiquimod no longer is recommended due to a lack of demonstrated superiority over placebo in recent studies as well as its adverse effects.3 Topical retinoids have been recommended; however, their use frequently is limited by local irritation.3,14 Cantharidin is the most frequently utilized treatment by pediatric dermatologists. Most health care providers report subjective satisfaction with its results and efficacy, though some side effects may occur including discomfort and temporary changes in pigmentation. Treatment for MC is not required, as the condition is self-limiting.14 Therapy often is reserved for those with extensive disease, complications from lesions, cosmetic or psychological concerns, or genital involvement given the potential for sexual transmission.3 Time to resolution without treatment varies and is more prolonged in immunocompromised patients. Mean time to resolution in immunocompetent hosts has been reported as 13.3 months, but most infections are noted to clear within 2 to 4 years.1,4 Although resolution without treatment occurs, transmission to others and negative impact on quality of life (QOL) can occur and support the need for treatment. Greater impact on QOL was observed in females, those with more lesions, and patients with a longer duration of symptoms. Moderate impact on QOL was reported in 28% of patients (n=301), and severe effects were reported in 11%.4
In conclusion, MC is a common, benign, treatable cutaneous viral infection that often presents as small, flesh-colored papules in children. Its appearance can mimic a variety of other conditions. In cases with abnormal presentations, definitive diagnosis with pathology can be important to differentiate MC from more dangerous etiologies that may require further treatment.
- Brown J, Janniger CK, Schwartz RA, et al. Childhood molluscum contagiosum. Int J Dermatol. 2006;45:93-99. doi:10.1111 /j.1365-4632.2006.02737.x
- Dohil MA, Lin P, Lee J, et al. The epidemiology of molluscum contagiosum in children. J Am Acad Dermatol. 2006;54:47-54. doi:10.1016/j.jaad.2005.08.035
- Robinson G, Townsend S, Jahnke MN. Molluscum contagiosum: review and update on clinical presentation, diagnosis, risk, prevention, and treatment. Curr Derm Rep. 2020;9:83-92.
- Olsen JR, Gallacher J, Finlay AY, et al. Time to resolution and effect on quality of life of molluscum contagiosum in children in the UK: a prospective community cohort study. Lancet Infect Dis. 2015;15:190-195. doi:10.1016/S1473-3099(14)71053-9
- Davey J, Biswas A. Follicular induction in a case of molluscum contagiosum: possible link with secondary anetoderma-like changes? Am J Dermatopathol. 2014;36:E19-E21. doi:10.1097/DAD.0b013e31828bc7c7
- Butala N, Siegfried E, Weissler A. Molluscum BOTE sign: a predictor of imminent resolution. Pediatrics. 2013;131:E1650-E1653. doi:10.1542/peds.2012-2933
- Uzuncakmak TK, Kuru BC, Zemheri EI, et al. Isolated giant molluscum contagiosum mimicking epidermoid cyst. Dermatol Pract Concept. 2016;6:71-73. doi:10.5826/dpc.0603a15
- Singh S, Swain M, Shukla S, et al. An unusual presentation of giant molluscum contagiosum diagnosed on cytology. Diagn Cytopathol. 2018;46:794-796. doi:10.1002/dc.23964
- Cohen PR, Tschen JA. Plantar molluscum contagiosum: a case report of molluscum contagiosum occurring on the sole of the foot and a review of the world literature. Cutis. 2012;90:35-41.
- Megalla M, Bronsnick T, Noor O, et al. Dermoscopic, confocal microscopic, and histologic characteristics of an atypical presentation of molluscum contagiosum. Ann Clin Pathol. 2014;2:1038.
- Patrice SJ, Wiss K, Mulliken JB. Pyogenic granuloma (lobular capillary hemangioma): a clinicopathologic study of 178 cases. Pediatr Dermatol. 1991;8:267-276. doi:10.1111/j.1525-1470.1991.tb00931.x
- Gong H-Z, Zheng H-Y, Li J. Amelanotic melanoma. Melanoma Res. 2019;29:221-230. doi:10.1097/CMR.0000000000000571
- Casso EM, Grin-Jorgensen CM, Grant-Kels JM. Spitz nevi. J Am Acad Dermatol. 1992;27(6 pt 1):901-913. doi:10.1016/0190-9622(92)70286-o
- Coloe J, Morrell DS. Cantharidin use among pediatric dermatologists in the treatment of molluscum contagiosum. Pediatr Dermatol. 2009;26:405-408.
The Diagnosis: Molluscum Contagiosum
A tangential shave removal with electrocautery was performed. Histopathology demonstrated numerous eosinophilic intracytoplasmic inclusion bodies (Figure), confirming a diagnosis of molluscum contagiosum (MC).

Molluscum contagiosum is a common poxvirus infection that is transmitted through fomites, contact, or self-inoculation.1 This infection most frequently occurs in school-aged children younger than 8 years1-3; peak incidence is 6 years of age.2,3 The worldwide estimated prevalence in children is 5.1% to 11.5%.1,3 In children cohabitating with others infected by MC, approximately 40% of households experienced a spread of infection; the risk of transmission is not associated with greater number of lesions.4 In adults, infection most commonly occurs in the setting of immunodeficiency or as a sexually transmitted infection in immunocompetent patients.3 Molluscum contagiosum infection classically presents as 1- to 3-mm, flesh- or white-colored, dome-shaped, smooth papules with central umbilication.1 Lesions often occur in clusters or lines, indicating local spread. The trunk, extremities, and face are areas that frequently are involved.2,3
Atypical presentations of MC infection can occur, as demonstrated by our case. Involvement of hair follicles by the infection can result in follicular induction.1,5 Secondary infection can mimic abscess formation.1 Inflamed MC lesions demonstrating the “beginning of the end” sign often are mistaken for primary infection, which is thought to be an inflammatory immune response to the virus.6 Lesions located on the eye or eyelid can present as unilateral conjunctivitis, conjunctival or corneal nodules, eyelid abscesses, or chalazions.1 Giant MC is a nodular variant of this infection measuring larger than 1 cm in size that can present similar to epidermoid cysts, condyloma acuminatum, or verruca vulgaris.1,7 Other reported mimicked conditions include basal cell carcinoma, trichoepithelioma, appendageal tumors, keratoacanthoma, foreign body granulomas, nevus sebaceous, or ecthyma.1,3 Molluscum contagiosum also has been reported to present as large ulcerative growths.8 In immunocompromised patients, deep fungal infection is another mimicker.1 Lesions on the plantar surfaces of the feet often are misdiagnosed as plantar verruca and present with pain during ambulation.9
The diagnosis of MC is clinical, with additional diagnostic tools reserved for more challenging situations.1 In cases with atypical presentations, dermoscopy may aid diagnosis through visualization of orifices and vascular patterns including crown, radial, and punctiform vessels.10 Biopsy or fine-needle aspiration also can be utilized as a diagnostic tool. Histopathology often reveals pathognomonic intracytoplasmic inclusions or Henderson-Paterson bodies.8,10 The appearance of MC can mimic other conditions that should be included in the differential diagnosis. Pyogenic granuloma often presents as a benign red papule that may grow rapidly and become pedunculated, sometimes with bleeding and crusting, though histology reveals groups of proliferating capillaries.11 More than half of amelanotic melanomas present in the papulonodular form as vascular or ulcerated nodules, and others may appear as erythematous macules. Diagnosis of amelanotic melanoma is made through histologic examination, which reveals atypical melanocytes in nests or cords, in conjunction with immunohistochemical stains such as S-100.12 Spitz nevi often appear as round, dome-shaped papules that most commonly are red, pink, or fleshcolored. They appear histologically similar to melanoma with nests of atypical melanocytes and nuclear atypia.13
A variety of treatment modalities can be used for MC including cantharidin, curettage, and cryotherapy.14 Imiquimod no longer is recommended due to a lack of demonstrated superiority over placebo in recent studies as well as its adverse effects.3 Topical retinoids have been recommended; however, their use frequently is limited by local irritation.3,14 Cantharidin is the most frequently utilized treatment by pediatric dermatologists. Most health care providers report subjective satisfaction with its results and efficacy, though some side effects may occur including discomfort and temporary changes in pigmentation. Treatment for MC is not required, as the condition is self-limiting.14 Therapy often is reserved for those with extensive disease, complications from lesions, cosmetic or psychological concerns, or genital involvement given the potential for sexual transmission.3 Time to resolution without treatment varies and is more prolonged in immunocompromised patients. Mean time to resolution in immunocompetent hosts has been reported as 13.3 months, but most infections are noted to clear within 2 to 4 years.1,4 Although resolution without treatment occurs, transmission to others and negative impact on quality of life (QOL) can occur and support the need for treatment. Greater impact on QOL was observed in females, those with more lesions, and patients with a longer duration of symptoms. Moderate impact on QOL was reported in 28% of patients (n=301), and severe effects were reported in 11%.4
In conclusion, MC is a common, benign, treatable cutaneous viral infection that often presents as small, flesh-colored papules in children. Its appearance can mimic a variety of other conditions. In cases with abnormal presentations, definitive diagnosis with pathology can be important to differentiate MC from more dangerous etiologies that may require further treatment.
The Diagnosis: Molluscum Contagiosum
A tangential shave removal with electrocautery was performed. Histopathology demonstrated numerous eosinophilic intracytoplasmic inclusion bodies (Figure), confirming a diagnosis of molluscum contagiosum (MC).

Molluscum contagiosum is a common poxvirus infection that is transmitted through fomites, contact, or self-inoculation.1 This infection most frequently occurs in school-aged children younger than 8 years1-3; peak incidence is 6 years of age.2,3 The worldwide estimated prevalence in children is 5.1% to 11.5%.1,3 In children cohabitating with others infected by MC, approximately 40% of households experienced a spread of infection; the risk of transmission is not associated with greater number of lesions.4 In adults, infection most commonly occurs in the setting of immunodeficiency or as a sexually transmitted infection in immunocompetent patients.3 Molluscum contagiosum infection classically presents as 1- to 3-mm, flesh- or white-colored, dome-shaped, smooth papules with central umbilication.1 Lesions often occur in clusters or lines, indicating local spread. The trunk, extremities, and face are areas that frequently are involved.2,3
Atypical presentations of MC infection can occur, as demonstrated by our case. Involvement of hair follicles by the infection can result in follicular induction.1,5 Secondary infection can mimic abscess formation.1 Inflamed MC lesions demonstrating the “beginning of the end” sign often are mistaken for primary infection, which is thought to be an inflammatory immune response to the virus.6 Lesions located on the eye or eyelid can present as unilateral conjunctivitis, conjunctival or corneal nodules, eyelid abscesses, or chalazions.1 Giant MC is a nodular variant of this infection measuring larger than 1 cm in size that can present similar to epidermoid cysts, condyloma acuminatum, or verruca vulgaris.1,7 Other reported mimicked conditions include basal cell carcinoma, trichoepithelioma, appendageal tumors, keratoacanthoma, foreign body granulomas, nevus sebaceous, or ecthyma.1,3 Molluscum contagiosum also has been reported to present as large ulcerative growths.8 In immunocompromised patients, deep fungal infection is another mimicker.1 Lesions on the plantar surfaces of the feet often are misdiagnosed as plantar verruca and present with pain during ambulation.9
The diagnosis of MC is clinical, with additional diagnostic tools reserved for more challenging situations.1 In cases with atypical presentations, dermoscopy may aid diagnosis through visualization of orifices and vascular patterns including crown, radial, and punctiform vessels.10 Biopsy or fine-needle aspiration also can be utilized as a diagnostic tool. Histopathology often reveals pathognomonic intracytoplasmic inclusions or Henderson-Paterson bodies.8,10 The appearance of MC can mimic other conditions that should be included in the differential diagnosis. Pyogenic granuloma often presents as a benign red papule that may grow rapidly and become pedunculated, sometimes with bleeding and crusting, though histology reveals groups of proliferating capillaries.11 More than half of amelanotic melanomas present in the papulonodular form as vascular or ulcerated nodules, and others may appear as erythematous macules. Diagnosis of amelanotic melanoma is made through histologic examination, which reveals atypical melanocytes in nests or cords, in conjunction with immunohistochemical stains such as S-100.12 Spitz nevi often appear as round, dome-shaped papules that most commonly are red, pink, or fleshcolored. They appear histologically similar to melanoma with nests of atypical melanocytes and nuclear atypia.13
A variety of treatment modalities can be used for MC including cantharidin, curettage, and cryotherapy.14 Imiquimod no longer is recommended due to a lack of demonstrated superiority over placebo in recent studies as well as its adverse effects.3 Topical retinoids have been recommended; however, their use frequently is limited by local irritation.3,14 Cantharidin is the most frequently utilized treatment by pediatric dermatologists. Most health care providers report subjective satisfaction with its results and efficacy, though some side effects may occur including discomfort and temporary changes in pigmentation. Treatment for MC is not required, as the condition is self-limiting.14 Therapy often is reserved for those with extensive disease, complications from lesions, cosmetic or psychological concerns, or genital involvement given the potential for sexual transmission.3 Time to resolution without treatment varies and is more prolonged in immunocompromised patients. Mean time to resolution in immunocompetent hosts has been reported as 13.3 months, but most infections are noted to clear within 2 to 4 years.1,4 Although resolution without treatment occurs, transmission to others and negative impact on quality of life (QOL) can occur and support the need for treatment. Greater impact on QOL was observed in females, those with more lesions, and patients with a longer duration of symptoms. Moderate impact on QOL was reported in 28% of patients (n=301), and severe effects were reported in 11%.4
In conclusion, MC is a common, benign, treatable cutaneous viral infection that often presents as small, flesh-colored papules in children. Its appearance can mimic a variety of other conditions. In cases with abnormal presentations, definitive diagnosis with pathology can be important to differentiate MC from more dangerous etiologies that may require further treatment.
- Brown J, Janniger CK, Schwartz RA, et al. Childhood molluscum contagiosum. Int J Dermatol. 2006;45:93-99. doi:10.1111 /j.1365-4632.2006.02737.x
- Dohil MA, Lin P, Lee J, et al. The epidemiology of molluscum contagiosum in children. J Am Acad Dermatol. 2006;54:47-54. doi:10.1016/j.jaad.2005.08.035
- Robinson G, Townsend S, Jahnke MN. Molluscum contagiosum: review and update on clinical presentation, diagnosis, risk, prevention, and treatment. Curr Derm Rep. 2020;9:83-92.
- Olsen JR, Gallacher J, Finlay AY, et al. Time to resolution and effect on quality of life of molluscum contagiosum in children in the UK: a prospective community cohort study. Lancet Infect Dis. 2015;15:190-195. doi:10.1016/S1473-3099(14)71053-9
- Davey J, Biswas A. Follicular induction in a case of molluscum contagiosum: possible link with secondary anetoderma-like changes? Am J Dermatopathol. 2014;36:E19-E21. doi:10.1097/DAD.0b013e31828bc7c7
- Butala N, Siegfried E, Weissler A. Molluscum BOTE sign: a predictor of imminent resolution. Pediatrics. 2013;131:E1650-E1653. doi:10.1542/peds.2012-2933
- Uzuncakmak TK, Kuru BC, Zemheri EI, et al. Isolated giant molluscum contagiosum mimicking epidermoid cyst. Dermatol Pract Concept. 2016;6:71-73. doi:10.5826/dpc.0603a15
- Singh S, Swain M, Shukla S, et al. An unusual presentation of giant molluscum contagiosum diagnosed on cytology. Diagn Cytopathol. 2018;46:794-796. doi:10.1002/dc.23964
- Cohen PR, Tschen JA. Plantar molluscum contagiosum: a case report of molluscum contagiosum occurring on the sole of the foot and a review of the world literature. Cutis. 2012;90:35-41.
- Megalla M, Bronsnick T, Noor O, et al. Dermoscopic, confocal microscopic, and histologic characteristics of an atypical presentation of molluscum contagiosum. Ann Clin Pathol. 2014;2:1038.
- Patrice SJ, Wiss K, Mulliken JB. Pyogenic granuloma (lobular capillary hemangioma): a clinicopathologic study of 178 cases. Pediatr Dermatol. 1991;8:267-276. doi:10.1111/j.1525-1470.1991.tb00931.x
- Gong H-Z, Zheng H-Y, Li J. Amelanotic melanoma. Melanoma Res. 2019;29:221-230. doi:10.1097/CMR.0000000000000571
- Casso EM, Grin-Jorgensen CM, Grant-Kels JM. Spitz nevi. J Am Acad Dermatol. 1992;27(6 pt 1):901-913. doi:10.1016/0190-9622(92)70286-o
- Coloe J, Morrell DS. Cantharidin use among pediatric dermatologists in the treatment of molluscum contagiosum. Pediatr Dermatol. 2009;26:405-408.
- Brown J, Janniger CK, Schwartz RA, et al. Childhood molluscum contagiosum. Int J Dermatol. 2006;45:93-99. doi:10.1111 /j.1365-4632.2006.02737.x
- Dohil MA, Lin P, Lee J, et al. The epidemiology of molluscum contagiosum in children. J Am Acad Dermatol. 2006;54:47-54. doi:10.1016/j.jaad.2005.08.035
- Robinson G, Townsend S, Jahnke MN. Molluscum contagiosum: review and update on clinical presentation, diagnosis, risk, prevention, and treatment. Curr Derm Rep. 2020;9:83-92.
- Olsen JR, Gallacher J, Finlay AY, et al. Time to resolution and effect on quality of life of molluscum contagiosum in children in the UK: a prospective community cohort study. Lancet Infect Dis. 2015;15:190-195. doi:10.1016/S1473-3099(14)71053-9
- Davey J, Biswas A. Follicular induction in a case of molluscum contagiosum: possible link with secondary anetoderma-like changes? Am J Dermatopathol. 2014;36:E19-E21. doi:10.1097/DAD.0b013e31828bc7c7
- Butala N, Siegfried E, Weissler A. Molluscum BOTE sign: a predictor of imminent resolution. Pediatrics. 2013;131:E1650-E1653. doi:10.1542/peds.2012-2933
- Uzuncakmak TK, Kuru BC, Zemheri EI, et al. Isolated giant molluscum contagiosum mimicking epidermoid cyst. Dermatol Pract Concept. 2016;6:71-73. doi:10.5826/dpc.0603a15
- Singh S, Swain M, Shukla S, et al. An unusual presentation of giant molluscum contagiosum diagnosed on cytology. Diagn Cytopathol. 2018;46:794-796. doi:10.1002/dc.23964
- Cohen PR, Tschen JA. Plantar molluscum contagiosum: a case report of molluscum contagiosum occurring on the sole of the foot and a review of the world literature. Cutis. 2012;90:35-41.
- Megalla M, Bronsnick T, Noor O, et al. Dermoscopic, confocal microscopic, and histologic characteristics of an atypical presentation of molluscum contagiosum. Ann Clin Pathol. 2014;2:1038.
- Patrice SJ, Wiss K, Mulliken JB. Pyogenic granuloma (lobular capillary hemangioma): a clinicopathologic study of 178 cases. Pediatr Dermatol. 1991;8:267-276. doi:10.1111/j.1525-1470.1991.tb00931.x
- Gong H-Z, Zheng H-Y, Li J. Amelanotic melanoma. Melanoma Res. 2019;29:221-230. doi:10.1097/CMR.0000000000000571
- Casso EM, Grin-Jorgensen CM, Grant-Kels JM. Spitz nevi. J Am Acad Dermatol. 1992;27(6 pt 1):901-913. doi:10.1016/0190-9622(92)70286-o
- Coloe J, Morrell DS. Cantharidin use among pediatric dermatologists in the treatment of molluscum contagiosum. Pediatr Dermatol. 2009;26:405-408.
A 13-year-old adolescent girl presented for evaluation of a lesion on the dorsal aspect of the right foot of 1 week’s duration. She had a history of acne vulgaris and seasonal allergic rhinitis. She previously had noticed a persistent, small, flesh-colored bump of unknown chronicity in the same location, which had been diagnosed as a skin tag at an outside clinic. She denied any prior treatment in this area. Approximately a week prior to presentation, the lesion became painful, larger, and darkened in color before draining yellowish fluid. Due to concern for superinfection, the patient was prescribed cephalexin by her pediatrician. Dermatologic examination revealed a 1-cm, violaceous, pedunculated plaque with hemorrhagic crust on the dorsal aspect of the right foot with surrounding erythema and tenderness.
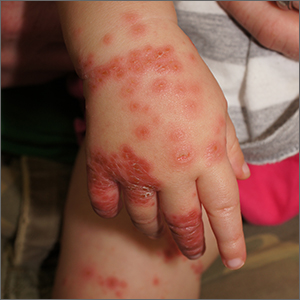
Widespread rash in toddler
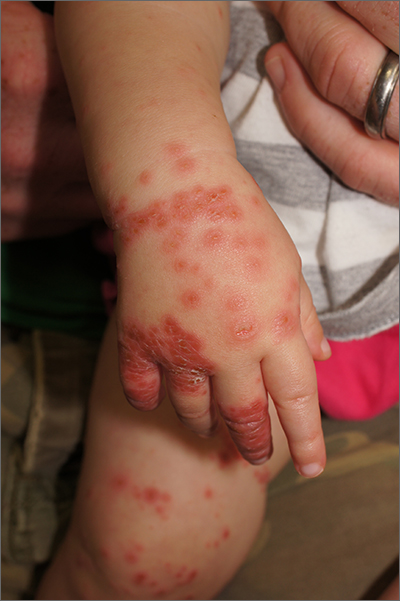
This patient was given a diagnosis of Gianotti Crosti syndrome (GCS; also called infantile acrodermatitis of childhood), which is a self-resolving (often dramatic) dermatosis triggered by a viral infection or immunization. Patients with this syndrome develop papules, vesicles, and plaques on their face, hands, feet, and extremities a week (or more) after having a viral illness or receiving an immunization. In patients with darker skin types, lesions may appear purple to brown rather than bright red to red/orange. The syndrome typically occurs in children between the ages of 1 to 4 years, but almost all patients are under the age of 15.1 Scratching and sleep disturbance are common. The condition typically resolves on its own after 3 or 4 weeks.
Globally, the hepatitis B virus (HBV) is the most common cause of GCS.1 Other reported triggering viruses include hepatitis A and C, cytomegalovirus, Epstein-Barr virus, enteroviruses, HIV, parainfluenza viruses, parvoviruses, rubella, and COVID-19.2
Since the cause of this patient’s case of GCS was likely linked to a viral infection that produced the loose stools in a population with low-HBV risk, no further serologic testing was performed. Serologic testing may have been necessary if other infections, disease risks, or symptoms were identified. To relieve itching, topical triamcinolone 0.1% cream was prescribed for use once to twice daily on the extremities and hydrocortisone 1% cream was prescribed once to twice daily for use on the child’s face. At the 6-week follow-up visit, the lesions had resolved; light pink discoloration remained but was expected to further fade. In patients with darker skin, post-inflammatory hyperpigmentation may take several months to resolve.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Brandt O, Abeck D, Gianotti R, et al. Gianotti-Crosti syndrome. J Am Acad Dermatol. 2006;54:136-45. doi: 10.1016/j.jaad.2005.09.033
2. Berná-Rico ED, Álvarez-Pinheiro C, Burgos-Blasco P, et al. A Gianotti-Crosti-like eruption in the setting of SARS-CoV-2 infection. Dermatol Ther. 2021;34:e15071. Doi:10.1111/dth.15071

This patient was given a diagnosis of Gianotti Crosti syndrome (GCS; also called infantile acrodermatitis of childhood), which is a self-resolving (often dramatic) dermatosis triggered by a viral infection or immunization. Patients with this syndrome develop papules, vesicles, and plaques on their face, hands, feet, and extremities a week (or more) after having a viral illness or receiving an immunization. In patients with darker skin types, lesions may appear purple to brown rather than bright red to red/orange. The syndrome typically occurs in children between the ages of 1 to 4 years, but almost all patients are under the age of 15.1 Scratching and sleep disturbance are common. The condition typically resolves on its own after 3 or 4 weeks.
Globally, the hepatitis B virus (HBV) is the most common cause of GCS.1 Other reported triggering viruses include hepatitis A and C, cytomegalovirus, Epstein-Barr virus, enteroviruses, HIV, parainfluenza viruses, parvoviruses, rubella, and COVID-19.2
Since the cause of this patient’s case of GCS was likely linked to a viral infection that produced the loose stools in a population with low-HBV risk, no further serologic testing was performed. Serologic testing may have been necessary if other infections, disease risks, or symptoms were identified. To relieve itching, topical triamcinolone 0.1% cream was prescribed for use once to twice daily on the extremities and hydrocortisone 1% cream was prescribed once to twice daily for use on the child’s face. At the 6-week follow-up visit, the lesions had resolved; light pink discoloration remained but was expected to further fade. In patients with darker skin, post-inflammatory hyperpigmentation may take several months to resolve.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).

This patient was given a diagnosis of Gianotti Crosti syndrome (GCS; also called infantile acrodermatitis of childhood), which is a self-resolving (often dramatic) dermatosis triggered by a viral infection or immunization. Patients with this syndrome develop papules, vesicles, and plaques on their face, hands, feet, and extremities a week (or more) after having a viral illness or receiving an immunization. In patients with darker skin types, lesions may appear purple to brown rather than bright red to red/orange. The syndrome typically occurs in children between the ages of 1 to 4 years, but almost all patients are under the age of 15.1 Scratching and sleep disturbance are common. The condition typically resolves on its own after 3 or 4 weeks.
Globally, the hepatitis B virus (HBV) is the most common cause of GCS.1 Other reported triggering viruses include hepatitis A and C, cytomegalovirus, Epstein-Barr virus, enteroviruses, HIV, parainfluenza viruses, parvoviruses, rubella, and COVID-19.2
Since the cause of this patient’s case of GCS was likely linked to a viral infection that produced the loose stools in a population with low-HBV risk, no further serologic testing was performed. Serologic testing may have been necessary if other infections, disease risks, or symptoms were identified. To relieve itching, topical triamcinolone 0.1% cream was prescribed for use once to twice daily on the extremities and hydrocortisone 1% cream was prescribed once to twice daily for use on the child’s face. At the 6-week follow-up visit, the lesions had resolved; light pink discoloration remained but was expected to further fade. In patients with darker skin, post-inflammatory hyperpigmentation may take several months to resolve.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Brandt O, Abeck D, Gianotti R, et al. Gianotti-Crosti syndrome. J Am Acad Dermatol. 2006;54:136-45. doi: 10.1016/j.jaad.2005.09.033
2. Berná-Rico ED, Álvarez-Pinheiro C, Burgos-Blasco P, et al. A Gianotti-Crosti-like eruption in the setting of SARS-CoV-2 infection. Dermatol Ther. 2021;34:e15071. Doi:10.1111/dth.15071
1. Brandt O, Abeck D, Gianotti R, et al. Gianotti-Crosti syndrome. J Am Acad Dermatol. 2006;54:136-45. doi: 10.1016/j.jaad.2005.09.033
2. Berná-Rico ED, Álvarez-Pinheiro C, Burgos-Blasco P, et al. A Gianotti-Crosti-like eruption in the setting of SARS-CoV-2 infection. Dermatol Ther. 2021;34:e15071. Doi:10.1111/dth.15071
White House expands access to monkeypox vaccines
The White House is scaling up its response to the monkeypox outbreak, expanding access to vaccines to more at-risk individuals, officials said in a press call. More than 56,000 doses of the monkeypox vaccine JYNNEOS will be made available immediately, and more than 240,000 doses will be allocated in the coming weeks.
“The administration’s current strategy is focused on containing the outbreak by providing vaccines to those most in need to prevent further spread of monkeypox in the communities most impacted,” CDC Director Rochelle Walensky, MD, MPH, said on a June 28 press call. “As additional supply becomes available, we will further expand our efforts making vaccines available to a wider population.”
As of June 28, there were 4,700 detected cases of monkeypox globally in 49 countries. Since the first U.S. case of monkeypox was identified on May 17, there have been 306 confirmed cases across 28 jurisdictions.
Prior to this announcement, vaccination against monkeypox was recommended only for people with known exposures to the virus. Now, the vaccine is available to people who are likely to be exposed to the virus, including:
- People who have had close physical contact with someone diagnosed with monkeypox.
- People with a sexual partner diagnosed with monkeypox.
- Men who have sex with men who have had multiple sex partners in a venue where monkeypox was identified.
The JYNNEOS vaccine is administered in two doses, delivered 28 days apart. People will have maximum immunity 2 weeks after the second dose. People should be vaccinated within 2 weeks of a possible monkeypox exposure, Dr. Walensky said, adding, “The sooner you can get vaccinated after exposure, the better.”
The U.S. Department of Health and Human Services will immediately allocate the 56,000 JYNNEOS doses across the country, prioritizing jurisdictions to areas of high transmission. A second vaccine, ACAM2000, can also be requested, but it has a greater risk for serious side effects and is not appropriate for immunocompromised individuals or people with heart disease. In the coming weeks, 240,000 JYNNEOS doses will be made available for second doses as well as first doses “as the vaccine strategy broadens,” said David Boucher, director of infectious disease preparedness and response for HHS. There are currently 800,000 JYNNEOS doses that have been manufactured and approved for release, he said, and awaiting inspection by the Food and Drug Administration, which should be completed in the beginning of July.
At the same time, the administration is focusing on increasing access to testing. Monkeypox testing is now available in 78 state public health labs in 48 states that can collectively conduct 10,000 tests per week. In addition, the administration announced on June 23 that HHS began shipping monkeypox tests to five commercial lab companies to expand testing capacity as well as make testing more accessible.
“We continue to work very closely with the community and with public health partners and clinicians to increase awareness of the monkey pox outbreak and to facilitate adequate capacity and equitable access to testing,” Dr. Walensky said. “I strongly encourage all health care providers to have a high clinical suspicion for monkeypox among their patients. Patients presenting with a suspicious rash should be tested.”
A version of this article first appeared on Medscape.com.
The White House is scaling up its response to the monkeypox outbreak, expanding access to vaccines to more at-risk individuals, officials said in a press call. More than 56,000 doses of the monkeypox vaccine JYNNEOS will be made available immediately, and more than 240,000 doses will be allocated in the coming weeks.
“The administration’s current strategy is focused on containing the outbreak by providing vaccines to those most in need to prevent further spread of monkeypox in the communities most impacted,” CDC Director Rochelle Walensky, MD, MPH, said on a June 28 press call. “As additional supply becomes available, we will further expand our efforts making vaccines available to a wider population.”
As of June 28, there were 4,700 detected cases of monkeypox globally in 49 countries. Since the first U.S. case of monkeypox was identified on May 17, there have been 306 confirmed cases across 28 jurisdictions.
Prior to this announcement, vaccination against monkeypox was recommended only for people with known exposures to the virus. Now, the vaccine is available to people who are likely to be exposed to the virus, including:
- People who have had close physical contact with someone diagnosed with monkeypox.
- People with a sexual partner diagnosed with monkeypox.
- Men who have sex with men who have had multiple sex partners in a venue where monkeypox was identified.
The JYNNEOS vaccine is administered in two doses, delivered 28 days apart. People will have maximum immunity 2 weeks after the second dose. People should be vaccinated within 2 weeks of a possible monkeypox exposure, Dr. Walensky said, adding, “The sooner you can get vaccinated after exposure, the better.”
The U.S. Department of Health and Human Services will immediately allocate the 56,000 JYNNEOS doses across the country, prioritizing jurisdictions to areas of high transmission. A second vaccine, ACAM2000, can also be requested, but it has a greater risk for serious side effects and is not appropriate for immunocompromised individuals or people with heart disease. In the coming weeks, 240,000 JYNNEOS doses will be made available for second doses as well as first doses “as the vaccine strategy broadens,” said David Boucher, director of infectious disease preparedness and response for HHS. There are currently 800,000 JYNNEOS doses that have been manufactured and approved for release, he said, and awaiting inspection by the Food and Drug Administration, which should be completed in the beginning of July.
At the same time, the administration is focusing on increasing access to testing. Monkeypox testing is now available in 78 state public health labs in 48 states that can collectively conduct 10,000 tests per week. In addition, the administration announced on June 23 that HHS began shipping monkeypox tests to five commercial lab companies to expand testing capacity as well as make testing more accessible.
“We continue to work very closely with the community and with public health partners and clinicians to increase awareness of the monkey pox outbreak and to facilitate adequate capacity and equitable access to testing,” Dr. Walensky said. “I strongly encourage all health care providers to have a high clinical suspicion for monkeypox among their patients. Patients presenting with a suspicious rash should be tested.”
A version of this article first appeared on Medscape.com.
The White House is scaling up its response to the monkeypox outbreak, expanding access to vaccines to more at-risk individuals, officials said in a press call. More than 56,000 doses of the monkeypox vaccine JYNNEOS will be made available immediately, and more than 240,000 doses will be allocated in the coming weeks.
“The administration’s current strategy is focused on containing the outbreak by providing vaccines to those most in need to prevent further spread of monkeypox in the communities most impacted,” CDC Director Rochelle Walensky, MD, MPH, said on a June 28 press call. “As additional supply becomes available, we will further expand our efforts making vaccines available to a wider population.”
As of June 28, there were 4,700 detected cases of monkeypox globally in 49 countries. Since the first U.S. case of monkeypox was identified on May 17, there have been 306 confirmed cases across 28 jurisdictions.
Prior to this announcement, vaccination against monkeypox was recommended only for people with known exposures to the virus. Now, the vaccine is available to people who are likely to be exposed to the virus, including:
- People who have had close physical contact with someone diagnosed with monkeypox.
- People with a sexual partner diagnosed with monkeypox.
- Men who have sex with men who have had multiple sex partners in a venue where monkeypox was identified.
The JYNNEOS vaccine is administered in two doses, delivered 28 days apart. People will have maximum immunity 2 weeks after the second dose. People should be vaccinated within 2 weeks of a possible monkeypox exposure, Dr. Walensky said, adding, “The sooner you can get vaccinated after exposure, the better.”
The U.S. Department of Health and Human Services will immediately allocate the 56,000 JYNNEOS doses across the country, prioritizing jurisdictions to areas of high transmission. A second vaccine, ACAM2000, can also be requested, but it has a greater risk for serious side effects and is not appropriate for immunocompromised individuals or people with heart disease. In the coming weeks, 240,000 JYNNEOS doses will be made available for second doses as well as first doses “as the vaccine strategy broadens,” said David Boucher, director of infectious disease preparedness and response for HHS. There are currently 800,000 JYNNEOS doses that have been manufactured and approved for release, he said, and awaiting inspection by the Food and Drug Administration, which should be completed in the beginning of July.
At the same time, the administration is focusing on increasing access to testing. Monkeypox testing is now available in 78 state public health labs in 48 states that can collectively conduct 10,000 tests per week. In addition, the administration announced on June 23 that HHS began shipping monkeypox tests to five commercial lab companies to expand testing capacity as well as make testing more accessible.
“We continue to work very closely with the community and with public health partners and clinicians to increase awareness of the monkey pox outbreak and to facilitate adequate capacity and equitable access to testing,” Dr. Walensky said. “I strongly encourage all health care providers to have a high clinical suspicion for monkeypox among their patients. Patients presenting with a suspicious rash should be tested.”
A version of this article first appeared on Medscape.com.
Children and COVID: Vaccination off to slow start for the newly eligible
New cases of COVID-19 continue to drop among children, but the vaccination effort in those under age 5 years began with something less than a bang.
In the first 2 days after their respective approvals, almost 99,000 children aged 5-11 years and over 675,000 children aged 12-15 were vaccinated, according to data from the Centers for Disease Control and Prevention. Children aged 0-4 years represent almost 6% of the overall population, compared with 8.7% for the 5- to 11-year-olds and 5.1% for those aged 12-15.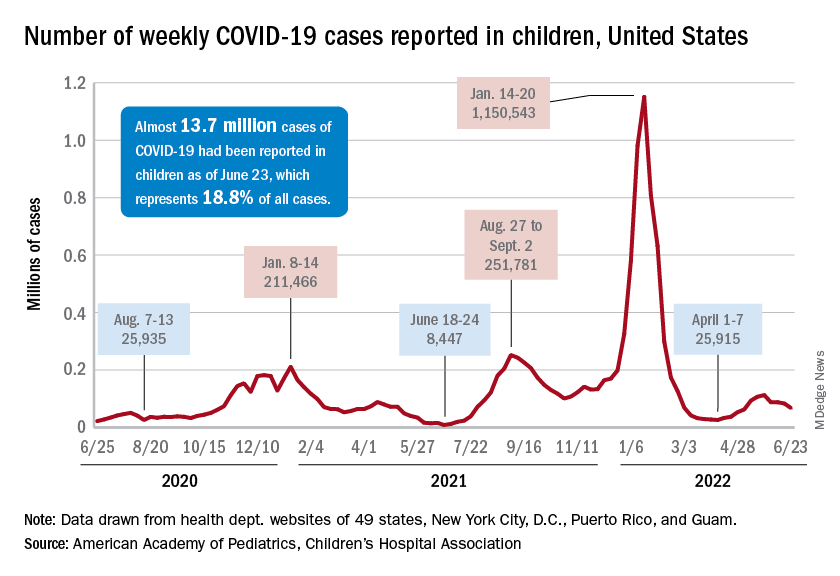
The recent decline in new cases over the past 4 weeks and the substantial decline since the Omicron surge could be a factor in the lack of response, but it is worth noting that the almost 68,000 new child cases reported in the past week, June 17-23, are “far higher than 1 year ago, June 24, 2021, when 8,400 child cases were reported,” the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID report.
That total for June 17-23 was 19% lower than the previous week and down by 40% since new cases hit a spring peak of 112,000 in late May. Regionally, new cases were down in the Midwest, the South, and the West, the AAP/CHA report showed, but the Northeast saw a small increase, which could be a signal of things to come for the summer.
The decline in new cases, however, has not been accompanied by decreases in hospitalizations or emergency department visits. New admissions of children aged 0-17 with confirmed COVID were at 0.31 per 100,000 population on June 24 after reaching that level on June 15, so no drop-off has occurred yet but there are signs of leveling off, based on CDC data.
The ED visit rates have been fairly steady through June, although COVID-related visits were up to 3.4% of all ED visits on June 22 for children aged 0-11 years, after being below 3% for the first 2 weeks of the month. The rate for children aged 12-15 has been between 1.6% and 1.9% for the past 3 weeks and the rate for 16- and 17-year-olds has been hovering between 1.7% and 2.2% for most of June, after going as high as 2.7% in late May, the CDC said on its COVID Data Tracker.
New cases of COVID-19 continue to drop among children, but the vaccination effort in those under age 5 years began with something less than a bang.
In the first 2 days after their respective approvals, almost 99,000 children aged 5-11 years and over 675,000 children aged 12-15 were vaccinated, according to data from the Centers for Disease Control and Prevention. Children aged 0-4 years represent almost 6% of the overall population, compared with 8.7% for the 5- to 11-year-olds and 5.1% for those aged 12-15.
The recent decline in new cases over the past 4 weeks and the substantial decline since the Omicron surge could be a factor in the lack of response, but it is worth noting that the almost 68,000 new child cases reported in the past week, June 17-23, are “far higher than 1 year ago, June 24, 2021, when 8,400 child cases were reported,” the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID report.
That total for June 17-23 was 19% lower than the previous week and down by 40% since new cases hit a spring peak of 112,000 in late May. Regionally, new cases were down in the Midwest, the South, and the West, the AAP/CHA report showed, but the Northeast saw a small increase, which could be a signal of things to come for the summer.
The decline in new cases, however, has not been accompanied by decreases in hospitalizations or emergency department visits. New admissions of children aged 0-17 with confirmed COVID were at 0.31 per 100,000 population on June 24 after reaching that level on June 15, so no drop-off has occurred yet but there are signs of leveling off, based on CDC data.
The ED visit rates have been fairly steady through June, although COVID-related visits were up to 3.4% of all ED visits on June 22 for children aged 0-11 years, after being below 3% for the first 2 weeks of the month. The rate for children aged 12-15 has been between 1.6% and 1.9% for the past 3 weeks and the rate for 16- and 17-year-olds has been hovering between 1.7% and 2.2% for most of June, after going as high as 2.7% in late May, the CDC said on its COVID Data Tracker.
New cases of COVID-19 continue to drop among children, but the vaccination effort in those under age 5 years began with something less than a bang.
In the first 2 days after their respective approvals, almost 99,000 children aged 5-11 years and over 675,000 children aged 12-15 were vaccinated, according to data from the Centers for Disease Control and Prevention. Children aged 0-4 years represent almost 6% of the overall population, compared with 8.7% for the 5- to 11-year-olds and 5.1% for those aged 12-15.
The recent decline in new cases over the past 4 weeks and the substantial decline since the Omicron surge could be a factor in the lack of response, but it is worth noting that the almost 68,000 new child cases reported in the past week, June 17-23, are “far higher than 1 year ago, June 24, 2021, when 8,400 child cases were reported,” the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID report.
That total for June 17-23 was 19% lower than the previous week and down by 40% since new cases hit a spring peak of 112,000 in late May. Regionally, new cases were down in the Midwest, the South, and the West, the AAP/CHA report showed, but the Northeast saw a small increase, which could be a signal of things to come for the summer.
The decline in new cases, however, has not been accompanied by decreases in hospitalizations or emergency department visits. New admissions of children aged 0-17 with confirmed COVID were at 0.31 per 100,000 population on June 24 after reaching that level on June 15, so no drop-off has occurred yet but there are signs of leveling off, based on CDC data.
The ED visit rates have been fairly steady through June, although COVID-related visits were up to 3.4% of all ED visits on June 22 for children aged 0-11 years, after being below 3% for the first 2 weeks of the month. The rate for children aged 12-15 has been between 1.6% and 1.9% for the past 3 weeks and the rate for 16- and 17-year-olds has been hovering between 1.7% and 2.2% for most of June, after going as high as 2.7% in late May, the CDC said on its COVID Data Tracker.
Racial/ethnic disparities exacerbated maternal death rise during 2020 pandemic.
U.S. maternal deaths – those during pregnancy or within 42 days of pregnancy – increased substantially by 33.3% after March 2020 corresponding to the COVID-19 pandemic onset, according to new research published in JAMA Network Open.
Data from the National Center for Health Statistics (NCHS) revealed this rise in maternal deaths was higher than the 22% overall excess death estimate associated with the pandemic in 2020.
Increases were highest for Hispanic and non-Hispanic Black women, exacerbating already high rates of disparity in comparison with White women, wrote Marie E. Thoma, PhD, an associate professor at the University of Maryland, College Park, and Eugene R. Declercq, PhD, a professor at Boston University.
The authors noted that this spike in maternal deaths might be caused either by conditions directly related to COVID-19, such as respiratory or viral infections, or by conditions worsened by pandemic-associated health care disruptions including those for diabetes or cardiovascular disease.
The precise causes, however, could not be discerned from the data, the authors noted.
The NCHS reported an 18.4% increase in U.S. maternal mortality from 2019 to 2020. The relative increase was 44.4% among Hispanic, 25.7% among non-Hispanic Black, and 6.1% among non-Hispanic White women.
“The rise in maternal mortality among Hispanic women was unprecedented,” Dr. Thoma said in an interview. Given a 16.8% increase in overall U.S. mortality in 2020, largely attributed to the COVID-19 pandemic, the authors examined the pandemic’s role in [the higher] maternal death rates for 2020.
“Prior to this report, the NCHS released an e-report that there had been a rise in maternal mortality in 2020, but questions remained about the role of the pandemic in this rise that their report hadn’t addressed,” Dr. Thoma said in an interview “So we decided to look at the data further to assess whether the rise coincided with the pandemic and how this differed by race/ethnicity, whether there were changes in the causes of maternal death, and how often COVID-19 was listed as a contributory factor in those deaths.”
A total of 1,588 maternal deaths (18.8 per 100,000 live births) occurred before the pandemic versus 684 deaths (25.1 per 100,000 live births) during the 2020 phase of the pandemic, for a relative increase of 33.3%.
Direct obstetrical causes of death included diabetes, hypertensive and liver disorders, pregnancy-related infections, and obstetrical hemorrhage and embolism. Indirect causes comprised, among others, nonobstetrical infections and diseases of the circulatory and respiratory systems as well as mental and nervous disorders.
Relative increases in direct causes (27.7%) were mostly associated with diabetes (95.9%), hypertensive disorders (39.0%), and other specified pregnancy-related conditions (48.0%).
COVID-19 was commonly listed as a lethal condition along with other viral diseases (16 of 16 deaths and diseases of the respiratory system (11 of 19 deaths).
Late maternal mortality – defined as more than 42 days but less than 1 year after pregnancy – increased by 41%. “This was surprising as we might anticipate risk being higher during pregnancy given that pregnant women may be more susceptible, but we see that this rise was also found among people in the later postpartum period,” Dr. Thoma said.
Absolute and relative changes were highest for Hispanic women (8.9 per 100,000 live births and 74.2%, respectively) and non-Hispanic Black women (16.8 per 100,000 live births and 40.2%). In contrast, non-Hispanic White women saw increases of just 2.9 per 100,000 live births and 17.2%.
“Overall, we found the rise in maternal mortality in 2020 was concentrated after the start of pandemic, particularly for non-Hispanic Black and Hispanic women, and we saw a dramatic rise in respiratory-related conditions,” Dr. Thoma said.
In a comment, Steven Woolf, MD, MPH, director emeritus of the Center on Society and Health at Virginia Commonwealth University, Richmond, said the findings are very consistent with his and others research showing dramatic increases in overall death rates from many causes during the pandemic, with these ranging from COVID-19 leading conditions such as diabetes, cardiovascular and Alzheimer’s disease to less-studied causes such as drug overdoses and alcoholism caused by the stresses of the pandemic. Again, deaths were likely caused by both COVID-19 infections and disruptions in diagnosis and care.
“So a rise in maternal mortality would unfortunately also be expected, and these researchers have shown that,” he said in an interview. In addition, they have confirmed “the pattern of stark health disparities in the Hispanic and Black populations relative to the White. Our group has shown marked decreases in the life expectancies of the Black and Hispanic populations relative to the White population.”
While he might take issue with the study’s research methodology, Dr. Woolf said, “The work is useful partly because we need to work out the best research methods to do this kind of analysis because we really need to understand the effects on maternal mortality.”
He said sorting out the best way to do this type of research will be important for looking at excess deaths and maternal mortality following other events, for example, in the wake of the Supreme Court’s recent decision to reverse Roe v. Wade.
The authors acknowledged certain study limitations, including the large percentage of COVID-19 cases with a nonspecific underlying cause. According to Dr. Thoma and Dr. Declercq, that reflects a maternal death coding problem that needs to be addressed, as well as a partitioning of data. The latter resulted in small numbers for some categories, with rates suppressed for fewer than 16 deaths because of reduced reliability.
“We found that more specific information is often available on death certificates but is lost in the process of coding,” said Dr. Thoma. “We were able to reclassify many of these causes to a more specific cause that we attributed to be the primary cause of death.”
The authors said future studies of maternal death should examine the contribution of the pandemic to racial and ethnic disparities and should identify specific causes of maternal deaths overall and associated with COVID-19.
In earlier research, the authors previously warned of possible misclassifications of maternal deaths.
They found evidence of both underreporting and overreporting of deaths, with possible overreporting predominant, whereas accurate data are essential for measuring the effectiveness of maternal mortality reduction programs.
Dr. Thoma’s group will continue to monitor mortality trends with the release of 2021 data. “We hope we will see improvements in 2021 given greater access to vaccines, treatments, and fewer health care disruptions,” Dr. Thoma said. “It will be important to continue to stress the importance of COVID-19 vaccines for pregnant and postpartum people.”
This study had no external funding. The authors disclosed no competing interests. Dr. Woolf declared no conflicts of interest.
U.S. maternal deaths – those during pregnancy or within 42 days of pregnancy – increased substantially by 33.3% after March 2020 corresponding to the COVID-19 pandemic onset, according to new research published in JAMA Network Open.
Data from the National Center for Health Statistics (NCHS) revealed this rise in maternal deaths was higher than the 22% overall excess death estimate associated with the pandemic in 2020.
Increases were highest for Hispanic and non-Hispanic Black women, exacerbating already high rates of disparity in comparison with White women, wrote Marie E. Thoma, PhD, an associate professor at the University of Maryland, College Park, and Eugene R. Declercq, PhD, a professor at Boston University.
The authors noted that this spike in maternal deaths might be caused either by conditions directly related to COVID-19, such as respiratory or viral infections, or by conditions worsened by pandemic-associated health care disruptions including those for diabetes or cardiovascular disease.
The precise causes, however, could not be discerned from the data, the authors noted.
The NCHS reported an 18.4% increase in U.S. maternal mortality from 2019 to 2020. The relative increase was 44.4% among Hispanic, 25.7% among non-Hispanic Black, and 6.1% among non-Hispanic White women.
“The rise in maternal mortality among Hispanic women was unprecedented,” Dr. Thoma said in an interview. Given a 16.8% increase in overall U.S. mortality in 2020, largely attributed to the COVID-19 pandemic, the authors examined the pandemic’s role in [the higher] maternal death rates for 2020.
“Prior to this report, the NCHS released an e-report that there had been a rise in maternal mortality in 2020, but questions remained about the role of the pandemic in this rise that their report hadn’t addressed,” Dr. Thoma said in an interview “So we decided to look at the data further to assess whether the rise coincided with the pandemic and how this differed by race/ethnicity, whether there were changes in the causes of maternal death, and how often COVID-19 was listed as a contributory factor in those deaths.”
A total of 1,588 maternal deaths (18.8 per 100,000 live births) occurred before the pandemic versus 684 deaths (25.1 per 100,000 live births) during the 2020 phase of the pandemic, for a relative increase of 33.3%.
Direct obstetrical causes of death included diabetes, hypertensive and liver disorders, pregnancy-related infections, and obstetrical hemorrhage and embolism. Indirect causes comprised, among others, nonobstetrical infections and diseases of the circulatory and respiratory systems as well as mental and nervous disorders.
Relative increases in direct causes (27.7%) were mostly associated with diabetes (95.9%), hypertensive disorders (39.0%), and other specified pregnancy-related conditions (48.0%).
COVID-19 was commonly listed as a lethal condition along with other viral diseases (16 of 16 deaths and diseases of the respiratory system (11 of 19 deaths).
Late maternal mortality – defined as more than 42 days but less than 1 year after pregnancy – increased by 41%. “This was surprising as we might anticipate risk being higher during pregnancy given that pregnant women may be more susceptible, but we see that this rise was also found among people in the later postpartum period,” Dr. Thoma said.
Absolute and relative changes were highest for Hispanic women (8.9 per 100,000 live births and 74.2%, respectively) and non-Hispanic Black women (16.8 per 100,000 live births and 40.2%). In contrast, non-Hispanic White women saw increases of just 2.9 per 100,000 live births and 17.2%.
“Overall, we found the rise in maternal mortality in 2020 was concentrated after the start of pandemic, particularly for non-Hispanic Black and Hispanic women, and we saw a dramatic rise in respiratory-related conditions,” Dr. Thoma said.
In a comment, Steven Woolf, MD, MPH, director emeritus of the Center on Society and Health at Virginia Commonwealth University, Richmond, said the findings are very consistent with his and others research showing dramatic increases in overall death rates from many causes during the pandemic, with these ranging from COVID-19 leading conditions such as diabetes, cardiovascular and Alzheimer’s disease to less-studied causes such as drug overdoses and alcoholism caused by the stresses of the pandemic. Again, deaths were likely caused by both COVID-19 infections and disruptions in diagnosis and care.
“So a rise in maternal mortality would unfortunately also be expected, and these researchers have shown that,” he said in an interview. In addition, they have confirmed “the pattern of stark health disparities in the Hispanic and Black populations relative to the White. Our group has shown marked decreases in the life expectancies of the Black and Hispanic populations relative to the White population.”
While he might take issue with the study’s research methodology, Dr. Woolf said, “The work is useful partly because we need to work out the best research methods to do this kind of analysis because we really need to understand the effects on maternal mortality.”
He said sorting out the best way to do this type of research will be important for looking at excess deaths and maternal mortality following other events, for example, in the wake of the Supreme Court’s recent decision to reverse Roe v. Wade.
The authors acknowledged certain study limitations, including the large percentage of COVID-19 cases with a nonspecific underlying cause. According to Dr. Thoma and Dr. Declercq, that reflects a maternal death coding problem that needs to be addressed, as well as a partitioning of data. The latter resulted in small numbers for some categories, with rates suppressed for fewer than 16 deaths because of reduced reliability.
“We found that more specific information is often available on death certificates but is lost in the process of coding,” said Dr. Thoma. “We were able to reclassify many of these causes to a more specific cause that we attributed to be the primary cause of death.”
The authors said future studies of maternal death should examine the contribution of the pandemic to racial and ethnic disparities and should identify specific causes of maternal deaths overall and associated with COVID-19.
In earlier research, the authors previously warned of possible misclassifications of maternal deaths.
They found evidence of both underreporting and overreporting of deaths, with possible overreporting predominant, whereas accurate data are essential for measuring the effectiveness of maternal mortality reduction programs.
Dr. Thoma’s group will continue to monitor mortality trends with the release of 2021 data. “We hope we will see improvements in 2021 given greater access to vaccines, treatments, and fewer health care disruptions,” Dr. Thoma said. “It will be important to continue to stress the importance of COVID-19 vaccines for pregnant and postpartum people.”
This study had no external funding. The authors disclosed no competing interests. Dr. Woolf declared no conflicts of interest.
U.S. maternal deaths – those during pregnancy or within 42 days of pregnancy – increased substantially by 33.3% after March 2020 corresponding to the COVID-19 pandemic onset, according to new research published in JAMA Network Open.
Data from the National Center for Health Statistics (NCHS) revealed this rise in maternal deaths was higher than the 22% overall excess death estimate associated with the pandemic in 2020.
Increases were highest for Hispanic and non-Hispanic Black women, exacerbating already high rates of disparity in comparison with White women, wrote Marie E. Thoma, PhD, an associate professor at the University of Maryland, College Park, and Eugene R. Declercq, PhD, a professor at Boston University.
The authors noted that this spike in maternal deaths might be caused either by conditions directly related to COVID-19, such as respiratory or viral infections, or by conditions worsened by pandemic-associated health care disruptions including those for diabetes or cardiovascular disease.
The precise causes, however, could not be discerned from the data, the authors noted.
The NCHS reported an 18.4% increase in U.S. maternal mortality from 2019 to 2020. The relative increase was 44.4% among Hispanic, 25.7% among non-Hispanic Black, and 6.1% among non-Hispanic White women.
“The rise in maternal mortality among Hispanic women was unprecedented,” Dr. Thoma said in an interview. Given a 16.8% increase in overall U.S. mortality in 2020, largely attributed to the COVID-19 pandemic, the authors examined the pandemic’s role in [the higher] maternal death rates for 2020.
“Prior to this report, the NCHS released an e-report that there had been a rise in maternal mortality in 2020, but questions remained about the role of the pandemic in this rise that their report hadn’t addressed,” Dr. Thoma said in an interview “So we decided to look at the data further to assess whether the rise coincided with the pandemic and how this differed by race/ethnicity, whether there were changes in the causes of maternal death, and how often COVID-19 was listed as a contributory factor in those deaths.”
A total of 1,588 maternal deaths (18.8 per 100,000 live births) occurred before the pandemic versus 684 deaths (25.1 per 100,000 live births) during the 2020 phase of the pandemic, for a relative increase of 33.3%.
Direct obstetrical causes of death included diabetes, hypertensive and liver disorders, pregnancy-related infections, and obstetrical hemorrhage and embolism. Indirect causes comprised, among others, nonobstetrical infections and diseases of the circulatory and respiratory systems as well as mental and nervous disorders.
Relative increases in direct causes (27.7%) were mostly associated with diabetes (95.9%), hypertensive disorders (39.0%), and other specified pregnancy-related conditions (48.0%).
COVID-19 was commonly listed as a lethal condition along with other viral diseases (16 of 16 deaths and diseases of the respiratory system (11 of 19 deaths).
Late maternal mortality – defined as more than 42 days but less than 1 year after pregnancy – increased by 41%. “This was surprising as we might anticipate risk being higher during pregnancy given that pregnant women may be more susceptible, but we see that this rise was also found among people in the later postpartum period,” Dr. Thoma said.
Absolute and relative changes were highest for Hispanic women (8.9 per 100,000 live births and 74.2%, respectively) and non-Hispanic Black women (16.8 per 100,000 live births and 40.2%). In contrast, non-Hispanic White women saw increases of just 2.9 per 100,000 live births and 17.2%.
“Overall, we found the rise in maternal mortality in 2020 was concentrated after the start of pandemic, particularly for non-Hispanic Black and Hispanic women, and we saw a dramatic rise in respiratory-related conditions,” Dr. Thoma said.
In a comment, Steven Woolf, MD, MPH, director emeritus of the Center on Society and Health at Virginia Commonwealth University, Richmond, said the findings are very consistent with his and others research showing dramatic increases in overall death rates from many causes during the pandemic, with these ranging from COVID-19 leading conditions such as diabetes, cardiovascular and Alzheimer’s disease to less-studied causes such as drug overdoses and alcoholism caused by the stresses of the pandemic. Again, deaths were likely caused by both COVID-19 infections and disruptions in diagnosis and care.
“So a rise in maternal mortality would unfortunately also be expected, and these researchers have shown that,” he said in an interview. In addition, they have confirmed “the pattern of stark health disparities in the Hispanic and Black populations relative to the White. Our group has shown marked decreases in the life expectancies of the Black and Hispanic populations relative to the White population.”
While he might take issue with the study’s research methodology, Dr. Woolf said, “The work is useful partly because we need to work out the best research methods to do this kind of analysis because we really need to understand the effects on maternal mortality.”
He said sorting out the best way to do this type of research will be important for looking at excess deaths and maternal mortality following other events, for example, in the wake of the Supreme Court’s recent decision to reverse Roe v. Wade.
The authors acknowledged certain study limitations, including the large percentage of COVID-19 cases with a nonspecific underlying cause. According to Dr. Thoma and Dr. Declercq, that reflects a maternal death coding problem that needs to be addressed, as well as a partitioning of data. The latter resulted in small numbers for some categories, with rates suppressed for fewer than 16 deaths because of reduced reliability.
“We found that more specific information is often available on death certificates but is lost in the process of coding,” said Dr. Thoma. “We were able to reclassify many of these causes to a more specific cause that we attributed to be the primary cause of death.”
The authors said future studies of maternal death should examine the contribution of the pandemic to racial and ethnic disparities and should identify specific causes of maternal deaths overall and associated with COVID-19.
In earlier research, the authors previously warned of possible misclassifications of maternal deaths.
They found evidence of both underreporting and overreporting of deaths, with possible overreporting predominant, whereas accurate data are essential for measuring the effectiveness of maternal mortality reduction programs.
Dr. Thoma’s group will continue to monitor mortality trends with the release of 2021 data. “We hope we will see improvements in 2021 given greater access to vaccines, treatments, and fewer health care disruptions,” Dr. Thoma said. “It will be important to continue to stress the importance of COVID-19 vaccines for pregnant and postpartum people.”
This study had no external funding. The authors disclosed no competing interests. Dr. Woolf declared no conflicts of interest.
FROM JAMA NETWORK OPEN
CDC releases new details on mysterious hepatitis in children
A new analysis from the Centers for Disease Control and Prevention provides further details on mysterious cases of pediatric hepatitis identified across the United States. While 45% of patients have tested positive for adenovirus infection, it is likely that these children “represent a heterogenous group of hepatitis etiologies,” the CDC authors wrote.
Of the 296 children diagnosed between Oct. 1, 2021, and June 15, 2022, in the United States, 18 have required liver transplants and 11 have died.
On April 21, 2022, the CDC issued an alert to providers to report pediatric hepatitis cases of unknown etiology in children under 10 after similar cases had been identified in Europe and the United States. While the United Kingdom has found an uptick in cases over the past year, researchers from the CDC published data on June 14 that suggested pediatric hepatitis cases had not increased from 2017 to 2021.
This newest analysis, published Morbidity and Mortality Weekly Report, provides additional demographic data on affected patients and explores possible causes, including previous infection with COVID-19. Investigators had earlier ruled out COVID-19 vaccination as a potential factor in these cases, as most children were unvaccinated or not yet eligible to receive the vaccine. According to the analysis, only five patients had received at least one dose of a COVID-19 vaccine.
The 296 cases included in the analysis occurred in 42 U.S. states and territories, and the median age for patients was 2 years and 2 months. Nearly 60% of patients were male (58.1%) and 40.9% were female. The largest percentage of cases occurred in Hispanic or Latino children (37.8%), followed by non-Hispanic White (32.4%) children. Black patients made up 9.8% of all cases, and 3.7% of affected children were of Asian descent. Vomiting, fatigue, and jaundice were all common symptoms, and about 90% (89.9%) of children required hospitalization..
Of 224 children tested for adenovirus, 44.6% were positive. The analysis also included information on 123 of these hepatitis patients tested for other various pathogens. Nearly 80% (98/123) received a COVID-19 test and just 10.2% were positive. About 26% of patients had previously had COVID-19, and hepatitis onset occurred, on average, 133 days after the reported SARS-CoV-2 infection.
Other viruses detected included rhinovirus/enterovirus (24.5%), rotavirus (14.0%), and acute Epstein-Barr virus (11.4%)
Simultaneous infection with SARS-CoV-2 and adenovirus occurred in three patients.
There was no evidence of viral inclusions in the 36 patients who had pathological evaluation liver biopsies, explants, or autopsied tissue.
The findings suggest that there may be many different causes behind these severe hepatitis cases, and it is estimated that about one-third of hepatitis cases in children do not have a known cause. However, the identification of adenovirus infection in many cases “raises the question whether a new pattern of disease is emerging in this population or if adenovirus might be an underrecognized cause or cofactor in previously indeterminate cases of pediatric hepatitis,” the authors wrote. As the investigation continues, “further clinical data are needed to understand the cause of these cases and to assess the potential association with adenovirus.”
A version of this article first appeared on Medscape.com.
A new analysis from the Centers for Disease Control and Prevention provides further details on mysterious cases of pediatric hepatitis identified across the United States. While 45% of patients have tested positive for adenovirus infection, it is likely that these children “represent a heterogenous group of hepatitis etiologies,” the CDC authors wrote.
Of the 296 children diagnosed between Oct. 1, 2021, and June 15, 2022, in the United States, 18 have required liver transplants and 11 have died.
On April 21, 2022, the CDC issued an alert to providers to report pediatric hepatitis cases of unknown etiology in children under 10 after similar cases had been identified in Europe and the United States. While the United Kingdom has found an uptick in cases over the past year, researchers from the CDC published data on June 14 that suggested pediatric hepatitis cases had not increased from 2017 to 2021.
This newest analysis, published Morbidity and Mortality Weekly Report, provides additional demographic data on affected patients and explores possible causes, including previous infection with COVID-19. Investigators had earlier ruled out COVID-19 vaccination as a potential factor in these cases, as most children were unvaccinated or not yet eligible to receive the vaccine. According to the analysis, only five patients had received at least one dose of a COVID-19 vaccine.
The 296 cases included in the analysis occurred in 42 U.S. states and territories, and the median age for patients was 2 years and 2 months. Nearly 60% of patients were male (58.1%) and 40.9% were female. The largest percentage of cases occurred in Hispanic or Latino children (37.8%), followed by non-Hispanic White (32.4%) children. Black patients made up 9.8% of all cases, and 3.7% of affected children were of Asian descent. Vomiting, fatigue, and jaundice were all common symptoms, and about 90% (89.9%) of children required hospitalization..
Of 224 children tested for adenovirus, 44.6% were positive. The analysis also included information on 123 of these hepatitis patients tested for other various pathogens. Nearly 80% (98/123) received a COVID-19 test and just 10.2% were positive. About 26% of patients had previously had COVID-19, and hepatitis onset occurred, on average, 133 days after the reported SARS-CoV-2 infection.
Other viruses detected included rhinovirus/enterovirus (24.5%), rotavirus (14.0%), and acute Epstein-Barr virus (11.4%)
Simultaneous infection with SARS-CoV-2 and adenovirus occurred in three patients.
There was no evidence of viral inclusions in the 36 patients who had pathological evaluation liver biopsies, explants, or autopsied tissue.
The findings suggest that there may be many different causes behind these severe hepatitis cases, and it is estimated that about one-third of hepatitis cases in children do not have a known cause. However, the identification of adenovirus infection in many cases “raises the question whether a new pattern of disease is emerging in this population or if adenovirus might be an underrecognized cause or cofactor in previously indeterminate cases of pediatric hepatitis,” the authors wrote. As the investigation continues, “further clinical data are needed to understand the cause of these cases and to assess the potential association with adenovirus.”
A version of this article first appeared on Medscape.com.
A new analysis from the Centers for Disease Control and Prevention provides further details on mysterious cases of pediatric hepatitis identified across the United States. While 45% of patients have tested positive for adenovirus infection, it is likely that these children “represent a heterogenous group of hepatitis etiologies,” the CDC authors wrote.
Of the 296 children diagnosed between Oct. 1, 2021, and June 15, 2022, in the United States, 18 have required liver transplants and 11 have died.
On April 21, 2022, the CDC issued an alert to providers to report pediatric hepatitis cases of unknown etiology in children under 10 after similar cases had been identified in Europe and the United States. While the United Kingdom has found an uptick in cases over the past year, researchers from the CDC published data on June 14 that suggested pediatric hepatitis cases had not increased from 2017 to 2021.
This newest analysis, published Morbidity and Mortality Weekly Report, provides additional demographic data on affected patients and explores possible causes, including previous infection with COVID-19. Investigators had earlier ruled out COVID-19 vaccination as a potential factor in these cases, as most children were unvaccinated or not yet eligible to receive the vaccine. According to the analysis, only five patients had received at least one dose of a COVID-19 vaccine.
The 296 cases included in the analysis occurred in 42 U.S. states and territories, and the median age for patients was 2 years and 2 months. Nearly 60% of patients were male (58.1%) and 40.9% were female. The largest percentage of cases occurred in Hispanic or Latino children (37.8%), followed by non-Hispanic White (32.4%) children. Black patients made up 9.8% of all cases, and 3.7% of affected children were of Asian descent. Vomiting, fatigue, and jaundice were all common symptoms, and about 90% (89.9%) of children required hospitalization..
Of 224 children tested for adenovirus, 44.6% were positive. The analysis also included information on 123 of these hepatitis patients tested for other various pathogens. Nearly 80% (98/123) received a COVID-19 test and just 10.2% were positive. About 26% of patients had previously had COVID-19, and hepatitis onset occurred, on average, 133 days after the reported SARS-CoV-2 infection.
Other viruses detected included rhinovirus/enterovirus (24.5%), rotavirus (14.0%), and acute Epstein-Barr virus (11.4%)
Simultaneous infection with SARS-CoV-2 and adenovirus occurred in three patients.
There was no evidence of viral inclusions in the 36 patients who had pathological evaluation liver biopsies, explants, or autopsied tissue.
The findings suggest that there may be many different causes behind these severe hepatitis cases, and it is estimated that about one-third of hepatitis cases in children do not have a known cause. However, the identification of adenovirus infection in many cases “raises the question whether a new pattern of disease is emerging in this population or if adenovirus might be an underrecognized cause or cofactor in previously indeterminate cases of pediatric hepatitis,” the authors wrote. As the investigation continues, “further clinical data are needed to understand the cause of these cases and to assess the potential association with adenovirus.”
A version of this article first appeared on Medscape.com.
FROM THE MMWR
Typhoid fever bacteria becoming more resistant to antibiotics
Bacteria that cause typhoid fever are becoming increasingly resistant to common antibiotics worldwide, a new analysis indicates.
Resistant strains of Salmonella enterica serovar typhi – almost all originating in South Asia – have spread across borders nearly 200 times since 1990.
Until now, analysis has been limited by small samples. This genome analysis is the largest to date and included 3,489 newly sequenced isolates (collected between 2014 and 2019) from prospective surveillance studies in four of the countries with the highest typhoid burden: Bangladesh, Nepal, Pakistan, and India.
Findings of the study, led by Kesia Esther da Silva, PhD, with the division of infectious diseases and geographic medicine at Stanford (Calif.) University, were published online in The Lancet Microbe.
Global deaths: 100,000 annually
Typhoid fever remains a global public health threat, causing 11 million infections and more than 100,000 deaths each year. Most cases (70%) are in South Asia, but typhoid also has significant presence in sub-Saharan Africa, Southeast Asia, and Oceania.
The findings are further evidence of the need for a global response, the authors write.
Jason Andrews, MD, a coauthor and associate professor in the division of infectious diseases and geographic medicine at Stanford University, said in an interview that the research helps pinpoint where the highest burden is and where the biggest need is for the two highly effective typhoid vaccines.
“We’re seeing higher levels of resistance than we’ve ever seen before against our latest and greatest antibiotics,” he said.
He said so far, strategies for tackling typhoid have involved country-level decisions and local funding and that needs to be shifted to a global priority. “Given contemporary travel migration patterns, what we see is that when antimicrobial resistance develops in one country, it quickly spreads to other countries.”
Dr. Andrews said the United States sees about 300-500 typhoid cases a year. “About 80% of those cases involve people traveling from South Asia,” he said.
Infections also come from people from the United States visiting high-burden countries, especially to see family. Often they don’t perceive the risk and skip vaccination, he said. U.S. clinicians can help with educating patients traveling to typhoid-endemic regions on pretravel vaccination.
Physician awareness is also important when patients have recently returned from such regions. Data from this study show a need to carefully consider which antibiotics will be effective with the growing resistance.
Only one oral option left in Pakistan
“We are running low on treatment options for typhoid,” Dr. Andrews said. The resistance pattern in Pakistan, for example, has left only one oral option, azithromycin, and resistance is building to that.
Without that option, “we’ll have to hospitalize patients and give intravenous antibiotics,” he said. “That’s concerning.”
Moreover, some resistant strains from Pakistan have been turning up in the United States.
“There are actually some cases that have not been tracked at all to travelers going to Pakistan and are thought to be from local transmission in the United States,” he said.
Valida Bajrovic, MD, assistant professor of medicine in infectious diseases at the Icahn School of Medicine at Mount Sinai, New York, said in an interview that, in addition to vaccinating travelers before they head to typhoid-endemic areas, physicians should educate patients on avoiding fecal transmission of typhoid with vigilant hand washing, drinking bottled water, and avoiding foods that may have been prepared in unsanitary conditions.
Dr. Bajrovic, who directs the antimicrobial stewardship efforts at the Mount Sinai Morningside and Mount Sinai West Hospitals, said stricter antimicrobial stewardship efforts are needed, particularly in Europe and South Asia, but also in the United States.
“Restriction of antibiotic use is the way to prevent antibiotic resistance,” she said, adding that such restrictions need to be part of a global effort.
Strains in the study were classified as multidrug resistant (MDR) if they contained genes resistant to ampicillin, chloramphenicol, and trimethoprim/sulfamethoxazole. The authors also traced the presence of genes demonstrating resistance to macrolides and quinolones.
At first, fluoroquinolones were effective against MDR S. typhi and in the 1990s became the primary therapy. By the 2010s, however, the majority of S. typhi in south Asia contained mutations in the quinolone resistance-determining regions.
The authors wrote: “We found evidence of frequent international (n = 138) and intercontinental transfers (n = 59) of antimicrobial-resistant S. typhi.”
According to the analysis, since 2000, MDR S. typhi has declined steadily in Bangladesh and India and remained at less than 5% of typhoid strains in Nepal, though it has increased slightly in Pakistan.
However, these are being replaced “with strains containing ceftriaxone resistance (extensively drug resistant), high-level fluoroquinolone resistance, or azithromycin resistance, which are reversing declines in the effective population size of S. typhi,” the authors wrote.
The analysis supports urgency for prevention measures, including use of typhoid conjugate vaccines in typhoid-endemic countries, the authors said.
But given the rise in international spread of increasingly resistant strains, they said, preventive measures should not be limited to those countries.
The study was funded by the Bill & Melinda Gates Foundation. Dr. Da Silva, Dr. Andrews, and Dr. Bajrovic have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Bacteria that cause typhoid fever are becoming increasingly resistant to common antibiotics worldwide, a new analysis indicates.
Resistant strains of Salmonella enterica serovar typhi – almost all originating in South Asia – have spread across borders nearly 200 times since 1990.
Until now, analysis has been limited by small samples. This genome analysis is the largest to date and included 3,489 newly sequenced isolates (collected between 2014 and 2019) from prospective surveillance studies in four of the countries with the highest typhoid burden: Bangladesh, Nepal, Pakistan, and India.
Findings of the study, led by Kesia Esther da Silva, PhD, with the division of infectious diseases and geographic medicine at Stanford (Calif.) University, were published online in The Lancet Microbe.
Global deaths: 100,000 annually
Typhoid fever remains a global public health threat, causing 11 million infections and more than 100,000 deaths each year. Most cases (70%) are in South Asia, but typhoid also has significant presence in sub-Saharan Africa, Southeast Asia, and Oceania.
The findings are further evidence of the need for a global response, the authors write.
Jason Andrews, MD, a coauthor and associate professor in the division of infectious diseases and geographic medicine at Stanford University, said in an interview that the research helps pinpoint where the highest burden is and where the biggest need is for the two highly effective typhoid vaccines.
“We’re seeing higher levels of resistance than we’ve ever seen before against our latest and greatest antibiotics,” he said.
He said so far, strategies for tackling typhoid have involved country-level decisions and local funding and that needs to be shifted to a global priority. “Given contemporary travel migration patterns, what we see is that when antimicrobial resistance develops in one country, it quickly spreads to other countries.”
Dr. Andrews said the United States sees about 300-500 typhoid cases a year. “About 80% of those cases involve people traveling from South Asia,” he said.
Infections also come from people from the United States visiting high-burden countries, especially to see family. Often they don’t perceive the risk and skip vaccination, he said. U.S. clinicians can help with educating patients traveling to typhoid-endemic regions on pretravel vaccination.
Physician awareness is also important when patients have recently returned from such regions. Data from this study show a need to carefully consider which antibiotics will be effective with the growing resistance.
Only one oral option left in Pakistan
“We are running low on treatment options for typhoid,” Dr. Andrews said. The resistance pattern in Pakistan, for example, has left only one oral option, azithromycin, and resistance is building to that.
Without that option, “we’ll have to hospitalize patients and give intravenous antibiotics,” he said. “That’s concerning.”
Moreover, some resistant strains from Pakistan have been turning up in the United States.
“There are actually some cases that have not been tracked at all to travelers going to Pakistan and are thought to be from local transmission in the United States,” he said.
Valida Bajrovic, MD, assistant professor of medicine in infectious diseases at the Icahn School of Medicine at Mount Sinai, New York, said in an interview that, in addition to vaccinating travelers before they head to typhoid-endemic areas, physicians should educate patients on avoiding fecal transmission of typhoid with vigilant hand washing, drinking bottled water, and avoiding foods that may have been prepared in unsanitary conditions.
Dr. Bajrovic, who directs the antimicrobial stewardship efforts at the Mount Sinai Morningside and Mount Sinai West Hospitals, said stricter antimicrobial stewardship efforts are needed, particularly in Europe and South Asia, but also in the United States.
“Restriction of antibiotic use is the way to prevent antibiotic resistance,” she said, adding that such restrictions need to be part of a global effort.
Strains in the study were classified as multidrug resistant (MDR) if they contained genes resistant to ampicillin, chloramphenicol, and trimethoprim/sulfamethoxazole. The authors also traced the presence of genes demonstrating resistance to macrolides and quinolones.
At first, fluoroquinolones were effective against MDR S. typhi and in the 1990s became the primary therapy. By the 2010s, however, the majority of S. typhi in south Asia contained mutations in the quinolone resistance-determining regions.
The authors wrote: “We found evidence of frequent international (n = 138) and intercontinental transfers (n = 59) of antimicrobial-resistant S. typhi.”
According to the analysis, since 2000, MDR S. typhi has declined steadily in Bangladesh and India and remained at less than 5% of typhoid strains in Nepal, though it has increased slightly in Pakistan.
However, these are being replaced “with strains containing ceftriaxone resistance (extensively drug resistant), high-level fluoroquinolone resistance, or azithromycin resistance, which are reversing declines in the effective population size of S. typhi,” the authors wrote.
The analysis supports urgency for prevention measures, including use of typhoid conjugate vaccines in typhoid-endemic countries, the authors said.
But given the rise in international spread of increasingly resistant strains, they said, preventive measures should not be limited to those countries.
The study was funded by the Bill & Melinda Gates Foundation. Dr. Da Silva, Dr. Andrews, and Dr. Bajrovic have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Bacteria that cause typhoid fever are becoming increasingly resistant to common antibiotics worldwide, a new analysis indicates.
Resistant strains of Salmonella enterica serovar typhi – almost all originating in South Asia – have spread across borders nearly 200 times since 1990.
Until now, analysis has been limited by small samples. This genome analysis is the largest to date and included 3,489 newly sequenced isolates (collected between 2014 and 2019) from prospective surveillance studies in four of the countries with the highest typhoid burden: Bangladesh, Nepal, Pakistan, and India.
Findings of the study, led by Kesia Esther da Silva, PhD, with the division of infectious diseases and geographic medicine at Stanford (Calif.) University, were published online in The Lancet Microbe.
Global deaths: 100,000 annually
Typhoid fever remains a global public health threat, causing 11 million infections and more than 100,000 deaths each year. Most cases (70%) are in South Asia, but typhoid also has significant presence in sub-Saharan Africa, Southeast Asia, and Oceania.
The findings are further evidence of the need for a global response, the authors write.
Jason Andrews, MD, a coauthor and associate professor in the division of infectious diseases and geographic medicine at Stanford University, said in an interview that the research helps pinpoint where the highest burden is and where the biggest need is for the two highly effective typhoid vaccines.
“We’re seeing higher levels of resistance than we’ve ever seen before against our latest and greatest antibiotics,” he said.
He said so far, strategies for tackling typhoid have involved country-level decisions and local funding and that needs to be shifted to a global priority. “Given contemporary travel migration patterns, what we see is that when antimicrobial resistance develops in one country, it quickly spreads to other countries.”
Dr. Andrews said the United States sees about 300-500 typhoid cases a year. “About 80% of those cases involve people traveling from South Asia,” he said.
Infections also come from people from the United States visiting high-burden countries, especially to see family. Often they don’t perceive the risk and skip vaccination, he said. U.S. clinicians can help with educating patients traveling to typhoid-endemic regions on pretravel vaccination.
Physician awareness is also important when patients have recently returned from such regions. Data from this study show a need to carefully consider which antibiotics will be effective with the growing resistance.
Only one oral option left in Pakistan
“We are running low on treatment options for typhoid,” Dr. Andrews said. The resistance pattern in Pakistan, for example, has left only one oral option, azithromycin, and resistance is building to that.
Without that option, “we’ll have to hospitalize patients and give intravenous antibiotics,” he said. “That’s concerning.”
Moreover, some resistant strains from Pakistan have been turning up in the United States.
“There are actually some cases that have not been tracked at all to travelers going to Pakistan and are thought to be from local transmission in the United States,” he said.
Valida Bajrovic, MD, assistant professor of medicine in infectious diseases at the Icahn School of Medicine at Mount Sinai, New York, said in an interview that, in addition to vaccinating travelers before they head to typhoid-endemic areas, physicians should educate patients on avoiding fecal transmission of typhoid with vigilant hand washing, drinking bottled water, and avoiding foods that may have been prepared in unsanitary conditions.
Dr. Bajrovic, who directs the antimicrobial stewardship efforts at the Mount Sinai Morningside and Mount Sinai West Hospitals, said stricter antimicrobial stewardship efforts are needed, particularly in Europe and South Asia, but also in the United States.
“Restriction of antibiotic use is the way to prevent antibiotic resistance,” she said, adding that such restrictions need to be part of a global effort.
Strains in the study were classified as multidrug resistant (MDR) if they contained genes resistant to ampicillin, chloramphenicol, and trimethoprim/sulfamethoxazole. The authors also traced the presence of genes demonstrating resistance to macrolides and quinolones.
At first, fluoroquinolones were effective against MDR S. typhi and in the 1990s became the primary therapy. By the 2010s, however, the majority of S. typhi in south Asia contained mutations in the quinolone resistance-determining regions.
The authors wrote: “We found evidence of frequent international (n = 138) and intercontinental transfers (n = 59) of antimicrobial-resistant S. typhi.”
According to the analysis, since 2000, MDR S. typhi has declined steadily in Bangladesh and India and remained at less than 5% of typhoid strains in Nepal, though it has increased slightly in Pakistan.
However, these are being replaced “with strains containing ceftriaxone resistance (extensively drug resistant), high-level fluoroquinolone resistance, or azithromycin resistance, which are reversing declines in the effective population size of S. typhi,” the authors wrote.
The analysis supports urgency for prevention measures, including use of typhoid conjugate vaccines in typhoid-endemic countries, the authors said.
But given the rise in international spread of increasingly resistant strains, they said, preventive measures should not be limited to those countries.
The study was funded by the Bill & Melinda Gates Foundation. Dr. Da Silva, Dr. Andrews, and Dr. Bajrovic have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
How can doctors protect their practices against monkeypox?
Globally, as of June 22, the number of patients with monkeypox has risen to 3,308, according to the U.S. Centers for Disease Control and Prevention. In Germany, 521 people have been infected to date. “There does not seem to be a monkeypox pandemic,” wrote Germany’s Federal Minister of Health Karl Lauterbach, MD. At the moment, the probability that doctors will see a patient infected with the monkeypox virus is quite small. Nevertheless, health care professionals should be prepared. The Robert Koch Institute (RKI), a German federal government agency, has compiled suggestions for inpatient and outpatient sectors.
Characteristics of the virus
All hygiene measures are oriented around the currently known characteristics of the monkeypox virus. According to the RKI, skin or mucosal contact with infectious material from the skin lesions of an infected person plays a key role in human-to-human transmission.
The virus remains biologically active for a certain amount of time, even in dried flakes of skin or dried secretion. Therefore, in general, “careful and thorough cleaning and disinfection of the patient environment or surfaces is necessary,” wrote the RKI. Droplet infections or contaminated surfaces are less often of importance.
Basic hygiene measures
“Fundamentally, all basic hygiene measures should of course be followed when dealing with the infected,” said the RKI. Doctors and other health care professionals should use hand sanitizer with proven, at least viricidal, efficacy.
Manufacturers provide such details on the packaging. Both the RKI and the Association for Applied Hygiene (VAH) have published compilations.
Measures in medical practices
In the outpatient sector, there is the (currently still quite low) danger that patients with monkeypox will infect other patients or practice employees. To prevent this, the RKI advised organizational measures.
If employees suspect that patients have monkeypox when they first arrive at the practice, or when they first speak to them over the phone, they must be separated. Waiting and treatment rooms with surfaces that can be wipe disinfected are well suited for this. Even if only suspected, all employees should wear disposable gloves and mouth-and-nose protection, which has become standard during COVID.
Measures in the clinical sector
In terms of accommodation, the RKI recommends isolation rooms with a washroom and, if possible, an antechamber that doctors and nurses can use to put on and take off their personal protective equipment (PPE). PPE includes disposable gloves, mouth-and-nose protection (for direct treatment, at least an FFP2 mask), and protective eyeglasses.
Special attention should be paid to the disinfection of surfaces. In addition to the selection of suitable preparations, the RKI advised that the high stability of the virus, especially in skin particles, be taken into account. When cleaning, particular care should be taken not to disturb any particles, according to the recommendations. In addition, the manufacturer’s application time must be strictly observed.
In the inpatient sector, such measures are important for all surfaces close to patients, such as bedside tables, wet zones, or door handles.
Medical devices such as stethoscopes or electrodes should be disinfected immediately after use. If possible, thermal treatment is preferred, such as for surgical apparatus, as long as they are not disposable products. The RKI has compiled separate recommendations for medical devices.
For laundry such as towels or bed linen, there is the danger that infectious particles will be stirred up. They should be collected and transported for treatment in sealable bags. Details on the selection of preparations can be found in the RKI or VAH list.
Contaminated waste is classified under waste code ASN 18 01 03 (“Guidelines for disposal of waste from healthcare institutions”) and may only be destroyed thermally in suitable facilities.
This article was translated from the Medscape German edition. A version of this article first appeared on Medscape.com.
Globally, as of June 22, the number of patients with monkeypox has risen to 3,308, according to the U.S. Centers for Disease Control and Prevention. In Germany, 521 people have been infected to date. “There does not seem to be a monkeypox pandemic,” wrote Germany’s Federal Minister of Health Karl Lauterbach, MD. At the moment, the probability that doctors will see a patient infected with the monkeypox virus is quite small. Nevertheless, health care professionals should be prepared. The Robert Koch Institute (RKI), a German federal government agency, has compiled suggestions for inpatient and outpatient sectors.
Characteristics of the virus
All hygiene measures are oriented around the currently known characteristics of the monkeypox virus. According to the RKI, skin or mucosal contact with infectious material from the skin lesions of an infected person plays a key role in human-to-human transmission.
The virus remains biologically active for a certain amount of time, even in dried flakes of skin or dried secretion. Therefore, in general, “careful and thorough cleaning and disinfection of the patient environment or surfaces is necessary,” wrote the RKI. Droplet infections or contaminated surfaces are less often of importance.
Basic hygiene measures
“Fundamentally, all basic hygiene measures should of course be followed when dealing with the infected,” said the RKI. Doctors and other health care professionals should use hand sanitizer with proven, at least viricidal, efficacy.
Manufacturers provide such details on the packaging. Both the RKI and the Association for Applied Hygiene (VAH) have published compilations.
Measures in medical practices
In the outpatient sector, there is the (currently still quite low) danger that patients with monkeypox will infect other patients or practice employees. To prevent this, the RKI advised organizational measures.
If employees suspect that patients have monkeypox when they first arrive at the practice, or when they first speak to them over the phone, they must be separated. Waiting and treatment rooms with surfaces that can be wipe disinfected are well suited for this. Even if only suspected, all employees should wear disposable gloves and mouth-and-nose protection, which has become standard during COVID.
Measures in the clinical sector
In terms of accommodation, the RKI recommends isolation rooms with a washroom and, if possible, an antechamber that doctors and nurses can use to put on and take off their personal protective equipment (PPE). PPE includes disposable gloves, mouth-and-nose protection (for direct treatment, at least an FFP2 mask), and protective eyeglasses.
Special attention should be paid to the disinfection of surfaces. In addition to the selection of suitable preparations, the RKI advised that the high stability of the virus, especially in skin particles, be taken into account. When cleaning, particular care should be taken not to disturb any particles, according to the recommendations. In addition, the manufacturer’s application time must be strictly observed.
In the inpatient sector, such measures are important for all surfaces close to patients, such as bedside tables, wet zones, or door handles.
Medical devices such as stethoscopes or electrodes should be disinfected immediately after use. If possible, thermal treatment is preferred, such as for surgical apparatus, as long as they are not disposable products. The RKI has compiled separate recommendations for medical devices.
For laundry such as towels or bed linen, there is the danger that infectious particles will be stirred up. They should be collected and transported for treatment in sealable bags. Details on the selection of preparations can be found in the RKI or VAH list.
Contaminated waste is classified under waste code ASN 18 01 03 (“Guidelines for disposal of waste from healthcare institutions”) and may only be destroyed thermally in suitable facilities.
This article was translated from the Medscape German edition. A version of this article first appeared on Medscape.com.
Globally, as of June 22, the number of patients with monkeypox has risen to 3,308, according to the U.S. Centers for Disease Control and Prevention. In Germany, 521 people have been infected to date. “There does not seem to be a monkeypox pandemic,” wrote Germany’s Federal Minister of Health Karl Lauterbach, MD. At the moment, the probability that doctors will see a patient infected with the monkeypox virus is quite small. Nevertheless, health care professionals should be prepared. The Robert Koch Institute (RKI), a German federal government agency, has compiled suggestions for inpatient and outpatient sectors.
Characteristics of the virus
All hygiene measures are oriented around the currently known characteristics of the monkeypox virus. According to the RKI, skin or mucosal contact with infectious material from the skin lesions of an infected person plays a key role in human-to-human transmission.
The virus remains biologically active for a certain amount of time, even in dried flakes of skin or dried secretion. Therefore, in general, “careful and thorough cleaning and disinfection of the patient environment or surfaces is necessary,” wrote the RKI. Droplet infections or contaminated surfaces are less often of importance.
Basic hygiene measures
“Fundamentally, all basic hygiene measures should of course be followed when dealing with the infected,” said the RKI. Doctors and other health care professionals should use hand sanitizer with proven, at least viricidal, efficacy.
Manufacturers provide such details on the packaging. Both the RKI and the Association for Applied Hygiene (VAH) have published compilations.
Measures in medical practices
In the outpatient sector, there is the (currently still quite low) danger that patients with monkeypox will infect other patients or practice employees. To prevent this, the RKI advised organizational measures.
If employees suspect that patients have monkeypox when they first arrive at the practice, or when they first speak to them over the phone, they must be separated. Waiting and treatment rooms with surfaces that can be wipe disinfected are well suited for this. Even if only suspected, all employees should wear disposable gloves and mouth-and-nose protection, which has become standard during COVID.
Measures in the clinical sector
In terms of accommodation, the RKI recommends isolation rooms with a washroom and, if possible, an antechamber that doctors and nurses can use to put on and take off their personal protective equipment (PPE). PPE includes disposable gloves, mouth-and-nose protection (for direct treatment, at least an FFP2 mask), and protective eyeglasses.
Special attention should be paid to the disinfection of surfaces. In addition to the selection of suitable preparations, the RKI advised that the high stability of the virus, especially in skin particles, be taken into account. When cleaning, particular care should be taken not to disturb any particles, according to the recommendations. In addition, the manufacturer’s application time must be strictly observed.
In the inpatient sector, such measures are important for all surfaces close to patients, such as bedside tables, wet zones, or door handles.
Medical devices such as stethoscopes or electrodes should be disinfected immediately after use. If possible, thermal treatment is preferred, such as for surgical apparatus, as long as they are not disposable products. The RKI has compiled separate recommendations for medical devices.
For laundry such as towels or bed linen, there is the danger that infectious particles will be stirred up. They should be collected and transported for treatment in sealable bags. Details on the selection of preparations can be found in the RKI or VAH list.
Contaminated waste is classified under waste code ASN 18 01 03 (“Guidelines for disposal of waste from healthcare institutions”) and may only be destroyed thermally in suitable facilities.
This article was translated from the Medscape German edition. A version of this article first appeared on Medscape.com.
Aging HIV patients face comorbidities and hospitalizations
Thanks to effective treatment, people with HIV are living longer. But as they age, they face higher rates of age-related comorbidities and hospitalizations, according to a recent study of hospitalized patients.
Decision-makers will need to allocate resources, train providers, and plan ways to manage chronic diseases, such as diabetes and cancer, among geriatric HIV inpatients, according to the authors.
“There will be more [HIV] patients with age-related chronic conditions at an earlier age and who will utilize or will have a unique need for [health care for] these geriatric conditions,” first author Khairul A. Siddiqi, PhD, University of Florida, Gainesville, said in an interview. “Eventually, that may increase inpatient resource utilization and costs.”
The study was published online in HIV Medicine.
Aging with HIV
Analyzing the National Inpatient Sample (NIS) of the Healthcare Cost and Utilization Project, the authors compared characteristics and comorbidities linked to hospital stays among people with HIV (HSWH) to those linked to hospital stays among people without HIV (HSWOH).
The NIS is a database of hospital records that captures 20% of discharges in the United States and covers all payers. Data in this analysis covered the years 2003-2015.
Among HSWH, patients aged 50 or older accounted for an increasing proportion over time, from fewer than 25% in 2003 to over 50% by 2015, the authors found. The subgroup aged 65-80 had risen from 2.39% to 8.63% by 2015.
The authors also studied rates of eight comorbidities, termed HIV-associated non-AIDS (HANA) conditions: cardiovascular, lung, liver, neurologic, and kidney diseases; diabetes; cancer; and bone loss.
The average number of these conditions among both HSWH and HSWOH rose over time. But this change was disproportionately high among HSWH aged 50-64 and those aged 65 and older.
Over the study period, among patients aged 65 or older, six of the eight age-related conditions the researchers studied rose disproportionately among HSWH in comparison with HSWOH; among those aged 50-64, five conditions did so.
The researchers are now building on the current study of HSWH by examining rates of resource utilization, such as MRIs and procedures, Dr. Siddiqi said.
Study limitations included a lack of data from long-term facilities, potential skewing by patients hospitalized multiple times, and the inherent limitations of administrative data.
A unique group of older people
Among people with HIV (PWH) in the United States, nearly half are aged 50 or older. By 2030, this group is expected to account for some 70% of PWH.
“We need to pay attention to what we know about aging generally. It is also important to study aging in this special population, because we don’t necessarily know a lot about that,” Amy Justice, MD, PhD, professor of medicine and of public health at Yale University, New Haven, Conn., said in an interview. Dr. Justice was not involved in the study.
The HIV epidemic has disproportionately affected people of color, men who have sex with men, and people with a history of injection drug use, Dr. Justice said.
“We don’t know about aging with [a] past history of injection drug use. We don’t even know much about aging with hepatitis C, necessarily,” she said. “So there are lots of reasons to pay some attention to this population to try to optimize their care.”
In addition, compared with their non–HIV-affected counterparts, these individuals are more susceptible to HANA comorbidities. They may experience these conditions at a younger age or more severely. Chronic inflammation and polypharmacy may be to blame, said Dr. Justice.
Given the burden of comorbidities and polypharmacy in this patient population, Dr. Siddiqi said, policy makers will need to focus on developing chronic disease management interventions for them.
However, Dr. Justice added, the risk for multimorbidity is higher among people with HIV throughout the age cycle: “It’s not like I turn 50 with HIV and all of a sudden all the wheels come off. There are ways to successfully age with HIV.”
Geriatric HIV expertise needed
Dr. Justice called the study’s analysis a useful addition to the literature and noted its implications for training.
“One of the biggest challenges with this large bolus of folks who are aging with HIV,” she said, “is to what extent should they be cared for by the people who have been caring for them – largely infectious disease docs – and to what extent should we really be transitioning their care to people with more experience with aging.”
Another key question, Dr. Justice said, relates to nursing homes and assisted-living facilities, whose staff may lack experience caring for HIV patients. Training them and hospital-based providers is crucial, in part to avoid key errors, such as missed antiretroviral doses, she said: “We need to really think about how to get non-HIV providers up to speed.”
That may begin by simply making it clear that this population is here.
“A decade ago, HIV patients used to have a lower life expectancy, so all HIV studies used to use 50 years as the cutoff point for [the] older population,” Dr. Siddiqi said. “Now we know they’re living longer.”
Added Dr. Justice: “Previously, people thought aging and HIV were not coincident findings.”
The study was funded by the Office of the Vice President for Research at the University of South Carolina. The authors and Dr. Justice disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Thanks to effective treatment, people with HIV are living longer. But as they age, they face higher rates of age-related comorbidities and hospitalizations, according to a recent study of hospitalized patients.
Decision-makers will need to allocate resources, train providers, and plan ways to manage chronic diseases, such as diabetes and cancer, among geriatric HIV inpatients, according to the authors.
“There will be more [HIV] patients with age-related chronic conditions at an earlier age and who will utilize or will have a unique need for [health care for] these geriatric conditions,” first author Khairul A. Siddiqi, PhD, University of Florida, Gainesville, said in an interview. “Eventually, that may increase inpatient resource utilization and costs.”
The study was published online in HIV Medicine.
Aging with HIV
Analyzing the National Inpatient Sample (NIS) of the Healthcare Cost and Utilization Project, the authors compared characteristics and comorbidities linked to hospital stays among people with HIV (HSWH) to those linked to hospital stays among people without HIV (HSWOH).
The NIS is a database of hospital records that captures 20% of discharges in the United States and covers all payers. Data in this analysis covered the years 2003-2015.
Among HSWH, patients aged 50 or older accounted for an increasing proportion over time, from fewer than 25% in 2003 to over 50% by 2015, the authors found. The subgroup aged 65-80 had risen from 2.39% to 8.63% by 2015.
The authors also studied rates of eight comorbidities, termed HIV-associated non-AIDS (HANA) conditions: cardiovascular, lung, liver, neurologic, and kidney diseases; diabetes; cancer; and bone loss.
The average number of these conditions among both HSWH and HSWOH rose over time. But this change was disproportionately high among HSWH aged 50-64 and those aged 65 and older.
Over the study period, among patients aged 65 or older, six of the eight age-related conditions the researchers studied rose disproportionately among HSWH in comparison with HSWOH; among those aged 50-64, five conditions did so.
The researchers are now building on the current study of HSWH by examining rates of resource utilization, such as MRIs and procedures, Dr. Siddiqi said.
Study limitations included a lack of data from long-term facilities, potential skewing by patients hospitalized multiple times, and the inherent limitations of administrative data.
A unique group of older people
Among people with HIV (PWH) in the United States, nearly half are aged 50 or older. By 2030, this group is expected to account for some 70% of PWH.
“We need to pay attention to what we know about aging generally. It is also important to study aging in this special population, because we don’t necessarily know a lot about that,” Amy Justice, MD, PhD, professor of medicine and of public health at Yale University, New Haven, Conn., said in an interview. Dr. Justice was not involved in the study.
The HIV epidemic has disproportionately affected people of color, men who have sex with men, and people with a history of injection drug use, Dr. Justice said.
“We don’t know about aging with [a] past history of injection drug use. We don’t even know much about aging with hepatitis C, necessarily,” she said. “So there are lots of reasons to pay some attention to this population to try to optimize their care.”
In addition, compared with their non–HIV-affected counterparts, these individuals are more susceptible to HANA comorbidities. They may experience these conditions at a younger age or more severely. Chronic inflammation and polypharmacy may be to blame, said Dr. Justice.
Given the burden of comorbidities and polypharmacy in this patient population, Dr. Siddiqi said, policy makers will need to focus on developing chronic disease management interventions for them.
However, Dr. Justice added, the risk for multimorbidity is higher among people with HIV throughout the age cycle: “It’s not like I turn 50 with HIV and all of a sudden all the wheels come off. There are ways to successfully age with HIV.”
Geriatric HIV expertise needed
Dr. Justice called the study’s analysis a useful addition to the literature and noted its implications for training.
“One of the biggest challenges with this large bolus of folks who are aging with HIV,” she said, “is to what extent should they be cared for by the people who have been caring for them – largely infectious disease docs – and to what extent should we really be transitioning their care to people with more experience with aging.”
Another key question, Dr. Justice said, relates to nursing homes and assisted-living facilities, whose staff may lack experience caring for HIV patients. Training them and hospital-based providers is crucial, in part to avoid key errors, such as missed antiretroviral doses, she said: “We need to really think about how to get non-HIV providers up to speed.”
That may begin by simply making it clear that this population is here.
“A decade ago, HIV patients used to have a lower life expectancy, so all HIV studies used to use 50 years as the cutoff point for [the] older population,” Dr. Siddiqi said. “Now we know they’re living longer.”
Added Dr. Justice: “Previously, people thought aging and HIV were not coincident findings.”
The study was funded by the Office of the Vice President for Research at the University of South Carolina. The authors and Dr. Justice disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Thanks to effective treatment, people with HIV are living longer. But as they age, they face higher rates of age-related comorbidities and hospitalizations, according to a recent study of hospitalized patients.
Decision-makers will need to allocate resources, train providers, and plan ways to manage chronic diseases, such as diabetes and cancer, among geriatric HIV inpatients, according to the authors.
“There will be more [HIV] patients with age-related chronic conditions at an earlier age and who will utilize or will have a unique need for [health care for] these geriatric conditions,” first author Khairul A. Siddiqi, PhD, University of Florida, Gainesville, said in an interview. “Eventually, that may increase inpatient resource utilization and costs.”
The study was published online in HIV Medicine.
Aging with HIV
Analyzing the National Inpatient Sample (NIS) of the Healthcare Cost and Utilization Project, the authors compared characteristics and comorbidities linked to hospital stays among people with HIV (HSWH) to those linked to hospital stays among people without HIV (HSWOH).
The NIS is a database of hospital records that captures 20% of discharges in the United States and covers all payers. Data in this analysis covered the years 2003-2015.
Among HSWH, patients aged 50 or older accounted for an increasing proportion over time, from fewer than 25% in 2003 to over 50% by 2015, the authors found. The subgroup aged 65-80 had risen from 2.39% to 8.63% by 2015.
The authors also studied rates of eight comorbidities, termed HIV-associated non-AIDS (HANA) conditions: cardiovascular, lung, liver, neurologic, and kidney diseases; diabetes; cancer; and bone loss.
The average number of these conditions among both HSWH and HSWOH rose over time. But this change was disproportionately high among HSWH aged 50-64 and those aged 65 and older.
Over the study period, among patients aged 65 or older, six of the eight age-related conditions the researchers studied rose disproportionately among HSWH in comparison with HSWOH; among those aged 50-64, five conditions did so.
The researchers are now building on the current study of HSWH by examining rates of resource utilization, such as MRIs and procedures, Dr. Siddiqi said.
Study limitations included a lack of data from long-term facilities, potential skewing by patients hospitalized multiple times, and the inherent limitations of administrative data.
A unique group of older people
Among people with HIV (PWH) in the United States, nearly half are aged 50 or older. By 2030, this group is expected to account for some 70% of PWH.
“We need to pay attention to what we know about aging generally. It is also important to study aging in this special population, because we don’t necessarily know a lot about that,” Amy Justice, MD, PhD, professor of medicine and of public health at Yale University, New Haven, Conn., said in an interview. Dr. Justice was not involved in the study.
The HIV epidemic has disproportionately affected people of color, men who have sex with men, and people with a history of injection drug use, Dr. Justice said.
“We don’t know about aging with [a] past history of injection drug use. We don’t even know much about aging with hepatitis C, necessarily,” she said. “So there are lots of reasons to pay some attention to this population to try to optimize their care.”
In addition, compared with their non–HIV-affected counterparts, these individuals are more susceptible to HANA comorbidities. They may experience these conditions at a younger age or more severely. Chronic inflammation and polypharmacy may be to blame, said Dr. Justice.
Given the burden of comorbidities and polypharmacy in this patient population, Dr. Siddiqi said, policy makers will need to focus on developing chronic disease management interventions for them.
However, Dr. Justice added, the risk for multimorbidity is higher among people with HIV throughout the age cycle: “It’s not like I turn 50 with HIV and all of a sudden all the wheels come off. There are ways to successfully age with HIV.”
Geriatric HIV expertise needed
Dr. Justice called the study’s analysis a useful addition to the literature and noted its implications for training.
“One of the biggest challenges with this large bolus of folks who are aging with HIV,” she said, “is to what extent should they be cared for by the people who have been caring for them – largely infectious disease docs – and to what extent should we really be transitioning their care to people with more experience with aging.”
Another key question, Dr. Justice said, relates to nursing homes and assisted-living facilities, whose staff may lack experience caring for HIV patients. Training them and hospital-based providers is crucial, in part to avoid key errors, such as missed antiretroviral doses, she said: “We need to really think about how to get non-HIV providers up to speed.”
That may begin by simply making it clear that this population is here.
“A decade ago, HIV patients used to have a lower life expectancy, so all HIV studies used to use 50 years as the cutoff point for [the] older population,” Dr. Siddiqi said. “Now we know they’re living longer.”
Added Dr. Justice: “Previously, people thought aging and HIV were not coincident findings.”
The study was funded by the Office of the Vice President for Research at the University of South Carolina. The authors and Dr. Justice disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM HIV MEDICINE