User login
Comprehensive wound malodor management: Win the RACE
Wounds that fail to heal become more than mere skin lesions. Pain, malodor, and the accompanying psychological distress often complicate nonhealing wounds and impair quality of life.1 Management of malodor requires perseverance, sensitivity, and familiarity with tools and procedures that range from surgical debridement to medical-grade honey.
Chronic, nonhealing wounds are defined as persisting for more than 6 months.2 These lesions are incapable of undergoing anatomic and functional repair on their own. Commonly encountered nonhealing wounds include pressure ulcers, venous stasis ulcers, arterial insufficiency ulcers, and malignant cutaneous wounds.
Typically, the patient with a nonhealing wound is frail, debilitated, medically complex, and often faced with one or more life-limiting illnesses. Complete wound healing may therefore be unrealistic, and optimal wound management becomes the goal of care.3,4
Healthcare providers encounter nonhealing wounds in varied settings—acute inpatient, outpatient, long-term, and home care. For instance, in the home care setting, a study of 383 patients enrolled in hospice found that 35% had skin ulcers and wounds.3 Half of those affected had pressure ulcers, 20% had ischemic ulcers, and 30% had other skin disorders such as stasis ulcers, burns, skin tears, and tumors. A larger study, also in hospice patients, found that 26% had pressure ulcers and 10% more developed them within 6 months.5
While pressure ulcers are the most common nonhealing wounds, malignant or fungating wounds are found in 5% to 10% of patients with metastatic disease, usually with cancers of the breast, head, and neck.6
Maximizing wound care provides comfort, relieves suffering, and promotes quality of life.3,7 To achieve these goals, clinicians must be familiar with strategies to manage complications associated with nonhealing wounds such as pain, malodor, and psychosocial adverse effects. Of these complications, malodor has been pointed out by both patients and caregivers as the most distressing.8
This article focuses on wound malodor, discusses the processes that cause wounds to emit an offensive smell, and outlines a comprehensive management approach.
MRS. A., AGE 61, WITH STAGE IV BREAST CANCER
Mrs. A., 61 years old, had a fungating mass in her left breast, which began as a small nodule and progressively enlarged to deform her breast over several months. Her oncologist subsequently staged the extent of her cancer as stage IV after workup revealed lung metastasis. Mrs. A. and her family decided to forgo cancer treatment, including radiotherapy, and to transition to hospice care after discussions with the oncologist.
Mrs. A. lived at home with her husband. Her daughter and three grandchildren all lived nearby.
When her hospice physician arrived at her home to meet her, a strong, pungent, and nauseating smell greeted him as he entered her bedroom. The patient said that for the past few months she had been increasingly distressed by the revolting odor. She rarely left home and had been ashamed to have people visit her, including her family.
On examination, the physician noticed a large fungating mass with yellowish discharge and necrotic tissue in her left breast. In addition to mild pain, she was immensely bothered by the strong odor coming from her breast.
THE IMPACT OF MALODOR
As seen in the case of Mrs. A., malodor has grave effects, both physical and psychological. Patients experience impaired or socially unacceptable body image, social rejection, personal shame, and embarrassment.9,10 Feelings of fear, anxiety, and depression are common. If left uncontrolled, malodor results in social isolation, reluctance to engage in social activities, diminished appetite, and nausea. In addition, malodor is a constant reminder of patients’ pain and cancer, and it results in further suffering.11
Reactions of family members and caregivers can worsen the situation.9,12 Expressions of revulsion limit contact and inhibit intimacy, especially near the end of life. Caregivers are often frustrated and distressed over their inability to control the malodor. The environment becomes uninhabitable, and the malodor can permeate clothing, furniture, and living quarters.
Managing malodor can be emotionally draining, physically daunting, and frustrating for healthcare professionals, as several methods are usually employed, often in a trial-and-error approach, to achieve an acceptable degree of odor control. In addition, clinicians must face the challenge of treating malodorous wounds at very close distance without reacting in a way that offends or alarms patients and family members.13
MALODOR PRODUCTION: WHERE IS THAT SMELL COMING FROM?
All wounds can produce an odor.14 Wounds that are expected to heal typically emit a faint but not unpleasant odor, akin to fresh blood. Wounds colonized by Pseudomonas aeruginosa produce a fruity or grapelike odor that is tolerable. Malodor occurs with wounds infected by other gram-negative organisms or anaerobic bacteria.15 Similarly, wounds covered by necrotic tissue smell like decaying flesh.
Three major causes
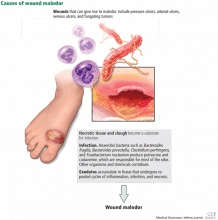
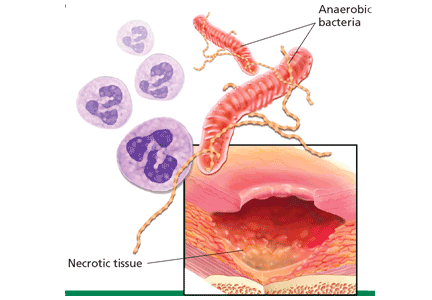
The three major causes of wound malodor are slough, infection, and exudate (Figure 1).
Slough is dead or necrotic tissue, usually resulting from vascular compromise. Arterial ulcers, pressure ulcers, and malignant wounds all form slough from capillary occlusion, subsequent ischemia, and tissue necrosis.
Infection. Devitalized tissue, an ideal medium in which bacteria thrive, becomes the source of infection. Anaerobic bacteria are usually implicated in malodor. These include Bacteroides fragilis, Bacteroides prevotella, Clostridium perfringens, and Fusobacterium nucleatum.16,17 Anaerobic organisms produce putrescine and cadaverine, which are largely responsible for the offensive odor.16,18 Volatile fatty acids such as propionic, butyric, isovaleric, and valeric acid are formed from lipid catabolism by anaerobes and add to malodor.17 Aerobic bacteria such as Proteus, Klebsiella, and Pseudomonas species supercolonize necrotic tissue as well and contribute to malodor.17,18
Exudate. Since nonhealing wounds undergo repeated cycles of inflammation, infection, and necrosis, accumulation of exudate becomes inevitable. Exudate typically is a pus-like fluid containing serum, fibrin, and white blood cells, which leak from blood vessels. In addition, bacteria that colonize chronic wounds filled with necrotic tissue activate proteases that degrade and liquefy dead tissue, thereby forming extensive amounts of exudate.19
Apart from slough, infection, and exudate, poor general hygiene and dressings left on for too long may contribute to malodor.16 Moisture-retentive dressings such as hydrocolloids leave an odor after removal. Dressings that liquefy upon contact with the wound surface leave a pus-like, potentially malodorous material.
MALODOR ASSESSMENT: DO YOU SMELL SOMETHING?
Various ways to document wound malodor can prove useful in guiding assessment and treatment. Descriptions such as “foul,” “putrid,” “fishy,” or “filled the room” vividly portray the initial presentation. A 10-point numerical scale similar to a numerical pain scale or a visual analogue scale can be used as a subjective measure.
Other grading methods, which to the authors’ knowledge are not validated, may be helpful. In a study that focused on patients suffering from malodorous gynecologic malignancies, von Gruenigen et al20 used a 0-to-3 scale:
- 0 Absent
- 1 Not offensive
- 2 Offensive but tolerable
- 3 Offensive and intolerable.
A scale often adapted by other authors was devised by Baker and Haig,21 which clearly defines four classes:
- 1 Strong—odor is evident upon entering the room (6 to 10 feet from the patient) with the dressing intact
- 2 Moderate—odor is evident upon entering the room with dressing removed
- 3 Slight—odor is evident at close proximity to the patient when the dressing is removed
- 4 No odor—no odor is evident, even at the patient’s bedside with the dressing removed.
COMPREHENSIVE MANAGEMENT: HOW DO WE WIN THE ‘RACE’?
The acronym RACE outlines an approach to dealing with malodor. It stands for removal of necrotic tissue; antibacterials; odor concealers; and education and support (Table 1).
Remove necrotic tissue
An important step in eliminating malodor is to remove necrotic tissue. This starts with debridement, which decreases the incidence of infection and hastens wound closure.22,23 Table 2 compares the different types of debridement.
Sharp or surgical debridement involves the use of a scalpel or scissors. This type of debridement may increase the risk of bleeding, pain, and malignant cell seeding in fungating wounds.4,24
Enzymatic debridement employs chemicals with proteolytic action (eg, collagenase) to digest extracellular proteins in wounds.18,25
Mechanical debridement involves aggressive therapies such as forceful irrigation and hydrotherapy, which may fail to discriminate between necrotic and viable tissues.18,26
Biological debridement using maggots, which ingest bacteria and devitalized tissue, may cause increased wound bleeding and may be unacceptable for patients and families.24,27
Autolytic debridement is often recommended, particularly if complete healing is not the primary goal.17,24,28,29 Autolysis uses proteolytic enzymes and phagocytic cells present in the wound bed and wound fluid to clear devitalized tissue. It is easy, inexpensive, noninvasive, and painless,4 and it requires less frequent dressing changes relative to standard dressing or wet-to-dry dressing.
Autolytic debridement is commonly accomplished using hydrocolloid and hydrogel dressings.15,29 Hydrocolloids are adhesive, occlusive, and conformable dressings that are suitable for wounds with low to moderate amounts of exudate. Upon contact with the wound surface, the dressing absorbs the exudate, forms a gel layer, and maintains a moist environment. Hydrocolloids are not recommended for infected wounds or for those with copious exudate as they may lead to maceration around the wound. A disadvantage of hydrocolloid dressings is their tendency to generate brown, often malodorous exudate when removed.
On the other hand, hydrogels in amorphous gel, dressing, sheet, or impregnated gauze form are water-based products that create a moist environment similar to hydrocolloids. Aside from causing minimal trauma to the wound bed when removed, the dressing’s cooling effect may bring some pain relief. Hydrogels are appropriate for dry wounds and for those with minimal exudate.
After debridement, the wound is cleansed and irrigated. A number of cleansers and solutions are available, but normal saline is a cheap alternative. To irrigate, experts recommend an 18- or 20-gauge intravenous catheter attached to a 30- or 60-mL syringe.15 This technique provides 8 to 15 psi of pressure, enough to cleanse the wound without causing tissue trauma.
Antibacterials and absorption
Antibacterials. Topical antibiotics have several advantages over systemic antibiotics in treating chronic wounds.30,31 These include a high and sustained concentration of the antimicrobial at the site of infection, limited potential for systemic absorption and toxicity, reduced potential for antibiotic resistance, and drawing of the patient’s and caregiver’s attention to the wound.
Metronidazole is the most widely used topical antibacterial for malodor management. Its efficacy is likely due to the predominant involvement of anaerobic bacteria in foul-smelling wounds. Topical metronidazole is available as a gel and as a cream. A systematic review showed that on average, topical metronidazole was used once daily for 14 consecutive days.19 The layer of topical metronidazole is typically covered with a nonadherent primary dressing followed by an absorbent secondary dressing.
The best clinical evidence for topical metronidazole consists of case reports and series.32–35 The largest of these studies was done by Finlay et al, who treated 47 patients with malodorous benign and malignant cutaneous wounds with 0.75% metronidazole gel daily.32 Forty-five (96%) of the patients reported significantly decreased odor by 14 days, as well as decreased pain, discharge, and surrounding cellulitis.
A randomized, placebo-controlled trial conducted by Bale et al had equivocal findings.9 All 41 patients who received metronidazole gel reported a decrease in malodor within 3 days of starting it. However, 76% of patients who received placebo also reported malodor control; in the final analysis, no significant difference was noted in the success rate between the two groups.
Metronidazole tablets can be crushed and sprinkled over the wound. As with metronidazole gel or cream, the crushed tablets are applied daily and covered by a primary nonadherent dressing and an absorbent secondary dressing. This off-label use of metronidazole serves as a cheaper alternative to commercially available topical preparations. To our knowledge, there has been no head-to-head trial comparing the two topical strategies.
Systemic metronidazole, often given orally, has been recommended if evidence of deep tissue or systemic infection is noted15 and in cases of fungating wounds with fistulas invading either the gastrointestinal or genitourinary tracts.18 Side effects such as nausea, neuropathy, and alcohol intolerance (ie, disulfiram reaction) may occur, which are not seen with topical metronidazole.
Both topical and systemic metronidazole can be used together on a time-limited basis for extensive malodorous wounds, such as fungating malignant wounds or stage IV sacral pressure ulcers.
Other antimicrobial agents used to treat malodor include silver-containing products, iodine-containing topical agents, mupirocin, bacitracin, neomycin, and polymyxin B.
Honey was used for wound care by the ancient Egyptians, and it is still used.36 Its beneficial effects include antimicrobial, debriding, deodorizing, anti-inflammatory, and granulation tissue-stimulating. Honey has even been shown to significantly decrease skin colonization with various kinds of bacteria, including methicillin-resistant Staphylococcus aureus.37 Medical-grade honey is preferred over table honey, as the latter is nonsterile and can contain Clostridium spores, which contaminate the wound.38
Yogurt and buttermilk lower the pH of the wound and control bacterial proliferation to control malodor.39,40 Either is applied for 10 to 15 minutes after the wound is cleansed and is then washed off thoroughly.
Absorbent dressings are used either over a layer of topical metronidazole and a nonadherent primary dressing or as a primary dressing itself. An absorbent dressing containing activated charcoal is used for rapid improvement, although cost may be prohibitive, especially in developing countries.13,19 Another type of absorbent dressing, composed of polyester impregnated with sodium chloride, has been found to be useful in malodor control.41 An important pointer is to maintain a tight seal around the absorbent dressing to prevent leakage of exudate.
Concealers
Aromatics used to conceal malodor include scented candles, incense, fragrant flowers and plants, and air-freshener sprays. When circumstances allow, candles are good options since they conceal malodor by emitting fragrance, and the flame burns off foul-smelling chemicals. Aromatics such as coffee beans, vanilla beans, and cider vinegar can be placed in a pan and left under the patient’s bed or close to it. Drops of peppermint oil or oil of wintergreen can be placed on wound dressings.
Other odor concealers are adsorbent materials that attract and cause ions and molecules to adhere to their surface. Examples are charcoal, baking soda, and cat litter. As with other aromatics, these materials are placed in pans and left under the bed or near the patient.
Aromatics can have disadvantages, as certain scents, especially strong ones, can be nauseating for patients. Some fragrances trigger asthma or skin irritation. Patients and caregivers can be left with an unpleasant association of certain fragrances with malodor by conditioning.15,17,18
Education and support
Concerns of the patient and family members need to be heard, addressed promptly, and reassessed with each visit, since uncontrolled malodor can be a chief source of caregiver fatigue.
Foremost in formulating a patient- and family-centered malodor management strategy is to commit to controlling malodor as much as possible. Regular follow-up appointments should be made, whether in the office or at home, to check on the patient’s progress and address new and ongoing concerns. Symptoms accompanying malodor, such as pain, bleeding, and sleep disturbance, need to be addressed, as they all affect quality of life.1 Audience-appropriate educational materials should be made available.26 Online resources that patients and families can explore include the websites of the Wound Ostomy and Continence Nurses Society (www.wocn.org) and the Association for the Advancement of Wound Care (aawconline.org).
Healthcare professionals need to be prepared to deal with problems and complications involving patients and family members that may arise in the course of treatment.12 Problems include the cost and local unavailability of dressing supplies, insurance coverage for dressings and topical agents, lack of assistance at home, and fear of changing dressings. A cardinal rule for healthcare providers is to avoid expressing distress at odors in front of or within hearing of patients and families.
OTHER STRATEGIES: WHAT ELSE CAN WE DO?
Curcumin, the main biologically active compound in the herb turmeric, applied directly to wounds three times daily as an ointment, has been shown to have odor-controlling properties.42
Sugar paste has been reported to control malodor by drawing out exudative and tissue fluid osmotically, and inhibiting bacterial growth.16,17 Water is mixed with sugar (ie, granulated, caster, or powdered) to form a paste, with additives like glycerin and polyethylene glycol used to alter the consistency. Thick clay-like paste is good for wounds with large cavities, while thin paste is useful for wounds with small or superficial openings. The paste is applied twice daily and is covered by an absorbent dressing.
Pressure relief is vital in managing pressure ulcers.18,43 Repositioning every 2 hours and using special devices, such as mattress overlays, alternating pressure mattresses, and low air loss mattresses, are frequently employed techniques.
If circumstances permit and when congruent with the patient’s goals of care, intra-arterial chemotherapy and radiotherapy can be contemplated for malignant fungating wounds.44,45
Other strategies include opening the windows during dressing changes, increasing the frequency of dressing changes, promptly removing used dressings from the house, and ensuring good general hygiene.
CASE RESOLUTION
After telling her that he was committed to control the malodor or, if possible, eliminate it, Mrs. A.’s doctor prepared two lists of materials—one for himself and one for Mrs. A.’s husband. He returned the next day, brought out his supplies, asked Mrs. A. to lie in bed, and invited her husband to assist him.
He cleansed and irrigated the breast lesion with normal saline, making sure to remove as much dead tissue as he could. He applied a layer of metronidazole cream to the wound cavity, then covered it with a nonadherent dressing. He then covered the wound with gauze, sealed the edges with medical adhesive tape, and applied a few drops of oil of wintergreen to the surface. A pan of charcoal briquettes was put under the bed, and a candle with Mrs. A.’s favorite scent was lit by the bedside. The physician then instructed Mrs. A.’s husband to repeat the procedure once daily for 1 week.
After 2 weeks, Mrs. A. and her husband said the foul odor had greatly decreased. She appeared more cheerful and energetic, especially after her grandchildren visited a few days earlier. The physician then instructed the husband to stop using metronidazole cream and to apply a hydrocolloid dressing every 3 days instead. He advised them to continue the rest of the process of applying a few drops of oil of wintergreen on the dressing surface, placing a pan of charcoal briquettes under the bed, and lighting a scented candle by the bedside.
FINISH THE RACE!
Complex nonhealing wounds are encountered across various healthcare settings. Wound malodor is an important component of nonhealing wounds, which adversely affects patients, families, and healthcare providers. Infection, slough, and exudate are the major causes of wound malodor. The essential steps to reduce malodor are to remove necrotic tissue, use antibacterial and odor-absorbing agents, apply appropriate odor “concealers,” educate families, and formulate a patient- and family-centered strategy (Table 1).
Acknowledgment: The authors would like to thank Sue Reif, CNP, for her assistance in completing the manuscript.
- Lo SF, Hayter M, Hu WY, Tai CY, Hsu MY, Li YF. Symptom burden and quality of life in patients with malignant fungating wounds. J Adv Nurs 2012; 68:1312–1321.
- Lazarus GS, Cooper DM, Knighton DR, et al. Definitions and guidelines for assessment of wounds and evaluation of healing. Arch Dermatol 1994; 130:489–493.
- Tippett AW. Wounds at the end of life. Wounds 2005; 17:91–98.
- Burt T. Palliative care of pressure ulcers in long-term care. Ann Long-Term Care 2013; 21:20–28.
- Reifsnyder J, Magee HS. Development of pressure ulcers in patients receiving home hospice care. Wounds 2005; 17:74–79.
- Haisfield-Wolfe ME, Rund C. Malignant cutaneous wounds: a management protocol. Ostomy Wound Manage 1997; 43:56–66.
- O’Brien C. Malignant wounds: managing odour. Can Fam Physician 2012; 58:272–274.
- Gethin G, Grocott P, Probst S, Clarke E. Current practice in the management of wound odour: an international survey. Int J Nurs Stud 2014; 51:865–874.
- Bale S, Tebble N, Price P. A topical metronidazole gel used to treat malodorous wounds. Br J Nurs 2004; 13:S4–S11.
- Hack A. Malodorous wounds—taking the patient’s perspective into account. J Wound Care 2003; 12:319–321.
- Price E. Wound care. The stigma of smell. Nurs Times 1996; 92:71–72.
- Paul JC, Pieper BA. Topical metronidazole for the treatment of wound odor: a review of the literature. Ostomy Wound Manage 2008; 54:18–27.
- Lee G, Anand SC, Rajendran S, Walker I. Overview of current practice and future trends in the evaluation of dressings for malodorous wounds. J Wound Care 2006; 15:344–346.
- Cutting K, Harding K. Criteria for identifying wound infection. J Wound Care 1994; 3:198–201.
- McDonald A, Lesage P. Palliative management of pressure ulcers and malignant wounds in patients with advanced illness. J Palliat Med 2006; 9:285–295.
- Holloway S. Recognising and treating the causes of chronic malodorous wounds. Prof Nurse 2004; 19:380–384.
- Haughton W, Young T. Common problems in wound care: malodorous wounds. Br J Nurs 1995; 4:959–963.
- Alvarez OM, Kalinski C, Nusbaum J, et al. Incorporating wound healing strategies to improve palliation (symptom management) in patients with chronic wounds. J Palliat Med 2007; 10:1161–1189.
- da Costa Santos CM, de Mattos Pimenta CA, Nobre MR. A systematic review of topical treatments to control the odor of malignant fungating wounds. J Pain Symptom Manage 2010; 39:1065–1076.
- Von Gruenigen VE, Coleman RL, et al. Bacteriology and treatment of malodorous lower reproductive tract in gynecologic cancer patients. Obstet Gynecol 2000; 96:23–27.
- Baker PG, Haig G. Metronidazole in the treatment of chronic pressure sores and ulcers: a comparison with standard treatment in general practice. Practitioner 1981; 225:569–573.
- Whitney J, Phillips L, Aslam R, et al. Guidelines for the treatment of pressure ulcers. Wound Repair Regen 2006; 14:663–679.
- Williams D, Enoch S, Miller D, Harris K, Price P, Harding KG. Effect of sharp debridement using curette on recalcitrant nonhealing venous ulcers: a concurrently controlled, prospective cohort study. Wound Repair Regen 2005; 13:131–137.
- Bergstrom KJ. Assessment and management of fungating wounds. J Wound Ostomy Continence Nurs 2011: 38:31–37.
- Sinclair RD, Ryan TJ. Proteolytic enzymes in wound healing: the role of enzymatic debridement. Australas J Dermatol 1994; 35:35–41.
- Enoch S, Harding KG. Wound bed preparation: the science behind the removal of barriers to healing. Wounds 2003;15:213–229.
- Mumcuoglu KY. Clinical applications for maggots in wound care. Am J Clin Dermatol 2001; 2:219–227.
- Langemo DK, Black J; National Pressure Ulcer Advisory Panel. Pressure ulcers in individuals receiving palliative care: a National Pressure Ulcer Advisory Panel white paper. Adv Skin Wound Care 2010; 23:59–72.
- Fonder MA, Lazarus GS, Cowan DA, Aronson-Cook B, Kohli AR, Mamelak AJ. Treating the chronic wound: a practical approach to the care of nonhealing wounds and wound care dressings. J Am Acad Dermatol 2008; 58:185–206.
- Lio PA, Kaye ET. Topical antibacterial agents. Infect Dis Clin North Am 2004; 18:717–733.
- Gelmetti C. Local antibiotics in dermatology. Dermatol Ther 2008; 21:187–195.
- Finlay IG, Bowszyc J, Ramlau C, Gwiezdzinski Z. The effect of topical 0.75% metronidazole gel on malodorous cutaneous ulcers. J Pain Symptom Manage 1996; 11:158–162.
- Bower M, Stein R, Evans TR, Hedley A, Pert P, Coombes RC. A double-blind study of the efficacy of metronidazole gel in the treatment of malodorous fungating tumours. Eur J Cancer 1992; 28A:888–889.
- Kalinski C, Schnepf M, Laboy D, et al. Effectiveness of a topical formulation containing metronidazole for wound odor and exudate control. Wounds 2005; 17:84–90.
- Kuge S, Tokuda Y, Ohta M, et al. Use of metronidazole gel to control malodor in advanced and recurrent breast cancer. Jpn J Clin Oncol 1996; 26:207–210.
- Belcher J. A review of medical-grade honey in wound care. Br J Nurs 2012: 21:S4–S9.
- Kwakman PH, Van den Akker JP, Güçlü A, et al. Medical-grade honey kills antibiotic-resistant bacteria in vitro and eradicates skin colonization. Clin Infect Dis 2008; 46:1677–1682.
- Cooper RA, Jenkins L. A comparison between medical grade honey and table honeys in relation to antimicrobial efficacy. Wounds 2009; 21:29–36.
- Patel B, Cox-Hayley D. Managing wound odor #218. J Palliat Med 2010; 13:1286–1287.
- Schulte MJ. Yogurt helps to control wound odor. Oncol Nurs Forum 1993; 20:1262.
- Upright CA, Salton C, Roberts F, Murphy J. Evaluation of Mesalt dressings and continuous wet saline dressings in ulcerating metastatic skin lesions. Cancer Nurs 1994; 17:149–155.
- Kuttan R, Sudheeran PC, Josph CD. Turmeric and curcumin as topical agents in cancer therapy. Tumori 1987; 73:29–31.
- Bass MJ, Phillips LG. Pressure sores. Curr Probl Surg 2007; 44:101–143.
- Bufill JA, Grace WR, Neff R. Intra-arterial chemotherapy for palliation of fungating breast cancer: a case report and review of the literature. Am J Clin Oncol 1994; 17:118–124.
- Murakami M, Kuroda Y, Sano A, et al. Validity of local treatment including intraarterial infusion chemotherapy and radiotherapy for fungating adenocarcinoma of the breast: case report of more than 8-year survival. Am J Clin Oncol 2001; 24:388–391.
Wounds that fail to heal become more than mere skin lesions. Pain, malodor, and the accompanying psychological distress often complicate nonhealing wounds and impair quality of life.1 Management of malodor requires perseverance, sensitivity, and familiarity with tools and procedures that range from surgical debridement to medical-grade honey.
Chronic, nonhealing wounds are defined as persisting for more than 6 months.2 These lesions are incapable of undergoing anatomic and functional repair on their own. Commonly encountered nonhealing wounds include pressure ulcers, venous stasis ulcers, arterial insufficiency ulcers, and malignant cutaneous wounds.
Typically, the patient with a nonhealing wound is frail, debilitated, medically complex, and often faced with one or more life-limiting illnesses. Complete wound healing may therefore be unrealistic, and optimal wound management becomes the goal of care.3,4
Healthcare providers encounter nonhealing wounds in varied settings—acute inpatient, outpatient, long-term, and home care. For instance, in the home care setting, a study of 383 patients enrolled in hospice found that 35% had skin ulcers and wounds.3 Half of those affected had pressure ulcers, 20% had ischemic ulcers, and 30% had other skin disorders such as stasis ulcers, burns, skin tears, and tumors. A larger study, also in hospice patients, found that 26% had pressure ulcers and 10% more developed them within 6 months.5
While pressure ulcers are the most common nonhealing wounds, malignant or fungating wounds are found in 5% to 10% of patients with metastatic disease, usually with cancers of the breast, head, and neck.6
Maximizing wound care provides comfort, relieves suffering, and promotes quality of life.3,7 To achieve these goals, clinicians must be familiar with strategies to manage complications associated with nonhealing wounds such as pain, malodor, and psychosocial adverse effects. Of these complications, malodor has been pointed out by both patients and caregivers as the most distressing.8
This article focuses on wound malodor, discusses the processes that cause wounds to emit an offensive smell, and outlines a comprehensive management approach.
MRS. A., AGE 61, WITH STAGE IV BREAST CANCER
Mrs. A., 61 years old, had a fungating mass in her left breast, which began as a small nodule and progressively enlarged to deform her breast over several months. Her oncologist subsequently staged the extent of her cancer as stage IV after workup revealed lung metastasis. Mrs. A. and her family decided to forgo cancer treatment, including radiotherapy, and to transition to hospice care after discussions with the oncologist.
Mrs. A. lived at home with her husband. Her daughter and three grandchildren all lived nearby.
When her hospice physician arrived at her home to meet her, a strong, pungent, and nauseating smell greeted him as he entered her bedroom. The patient said that for the past few months she had been increasingly distressed by the revolting odor. She rarely left home and had been ashamed to have people visit her, including her family.
On examination, the physician noticed a large fungating mass with yellowish discharge and necrotic tissue in her left breast. In addition to mild pain, she was immensely bothered by the strong odor coming from her breast.
THE IMPACT OF MALODOR
As seen in the case of Mrs. A., malodor has grave effects, both physical and psychological. Patients experience impaired or socially unacceptable body image, social rejection, personal shame, and embarrassment.9,10 Feelings of fear, anxiety, and depression are common. If left uncontrolled, malodor results in social isolation, reluctance to engage in social activities, diminished appetite, and nausea. In addition, malodor is a constant reminder of patients’ pain and cancer, and it results in further suffering.11
Reactions of family members and caregivers can worsen the situation.9,12 Expressions of revulsion limit contact and inhibit intimacy, especially near the end of life. Caregivers are often frustrated and distressed over their inability to control the malodor. The environment becomes uninhabitable, and the malodor can permeate clothing, furniture, and living quarters.
Managing malodor can be emotionally draining, physically daunting, and frustrating for healthcare professionals, as several methods are usually employed, often in a trial-and-error approach, to achieve an acceptable degree of odor control. In addition, clinicians must face the challenge of treating malodorous wounds at very close distance without reacting in a way that offends or alarms patients and family members.13
MALODOR PRODUCTION: WHERE IS THAT SMELL COMING FROM?
All wounds can produce an odor.14 Wounds that are expected to heal typically emit a faint but not unpleasant odor, akin to fresh blood. Wounds colonized by Pseudomonas aeruginosa produce a fruity or grapelike odor that is tolerable. Malodor occurs with wounds infected by other gram-negative organisms or anaerobic bacteria.15 Similarly, wounds covered by necrotic tissue smell like decaying flesh.
Three major causes
The three major causes of wound malodor are slough, infection, and exudate (Figure 1).
Slough is dead or necrotic tissue, usually resulting from vascular compromise. Arterial ulcers, pressure ulcers, and malignant wounds all form slough from capillary occlusion, subsequent ischemia, and tissue necrosis.
Infection. Devitalized tissue, an ideal medium in which bacteria thrive, becomes the source of infection. Anaerobic bacteria are usually implicated in malodor. These include Bacteroides fragilis, Bacteroides prevotella, Clostridium perfringens, and Fusobacterium nucleatum.16,17 Anaerobic organisms produce putrescine and cadaverine, which are largely responsible for the offensive odor.16,18 Volatile fatty acids such as propionic, butyric, isovaleric, and valeric acid are formed from lipid catabolism by anaerobes and add to malodor.17 Aerobic bacteria such as Proteus, Klebsiella, and Pseudomonas species supercolonize necrotic tissue as well and contribute to malodor.17,18
Exudate. Since nonhealing wounds undergo repeated cycles of inflammation, infection, and necrosis, accumulation of exudate becomes inevitable. Exudate typically is a pus-like fluid containing serum, fibrin, and white blood cells, which leak from blood vessels. In addition, bacteria that colonize chronic wounds filled with necrotic tissue activate proteases that degrade and liquefy dead tissue, thereby forming extensive amounts of exudate.19
Apart from slough, infection, and exudate, poor general hygiene and dressings left on for too long may contribute to malodor.16 Moisture-retentive dressings such as hydrocolloids leave an odor after removal. Dressings that liquefy upon contact with the wound surface leave a pus-like, potentially malodorous material.
MALODOR ASSESSMENT: DO YOU SMELL SOMETHING?
Various ways to document wound malodor can prove useful in guiding assessment and treatment. Descriptions such as “foul,” “putrid,” “fishy,” or “filled the room” vividly portray the initial presentation. A 10-point numerical scale similar to a numerical pain scale or a visual analogue scale can be used as a subjective measure.
Other grading methods, which to the authors’ knowledge are not validated, may be helpful. In a study that focused on patients suffering from malodorous gynecologic malignancies, von Gruenigen et al20 used a 0-to-3 scale:
- 0 Absent
- 1 Not offensive
- 2 Offensive but tolerable
- 3 Offensive and intolerable.
A scale often adapted by other authors was devised by Baker and Haig,21 which clearly defines four classes:
- 1 Strong—odor is evident upon entering the room (6 to 10 feet from the patient) with the dressing intact
- 2 Moderate—odor is evident upon entering the room with dressing removed
- 3 Slight—odor is evident at close proximity to the patient when the dressing is removed
- 4 No odor—no odor is evident, even at the patient’s bedside with the dressing removed.
COMPREHENSIVE MANAGEMENT: HOW DO WE WIN THE ‘RACE’?
The acronym RACE outlines an approach to dealing with malodor. It stands for removal of necrotic tissue; antibacterials; odor concealers; and education and support (Table 1).
Remove necrotic tissue
An important step in eliminating malodor is to remove necrotic tissue. This starts with debridement, which decreases the incidence of infection and hastens wound closure.22,23 Table 2 compares the different types of debridement.
Sharp or surgical debridement involves the use of a scalpel or scissors. This type of debridement may increase the risk of bleeding, pain, and malignant cell seeding in fungating wounds.4,24
Enzymatic debridement employs chemicals with proteolytic action (eg, collagenase) to digest extracellular proteins in wounds.18,25
Mechanical debridement involves aggressive therapies such as forceful irrigation and hydrotherapy, which may fail to discriminate between necrotic and viable tissues.18,26
Biological debridement using maggots, which ingest bacteria and devitalized tissue, may cause increased wound bleeding and may be unacceptable for patients and families.24,27
Autolytic debridement is often recommended, particularly if complete healing is not the primary goal.17,24,28,29 Autolysis uses proteolytic enzymes and phagocytic cells present in the wound bed and wound fluid to clear devitalized tissue. It is easy, inexpensive, noninvasive, and painless,4 and it requires less frequent dressing changes relative to standard dressing or wet-to-dry dressing.
Autolytic debridement is commonly accomplished using hydrocolloid and hydrogel dressings.15,29 Hydrocolloids are adhesive, occlusive, and conformable dressings that are suitable for wounds with low to moderate amounts of exudate. Upon contact with the wound surface, the dressing absorbs the exudate, forms a gel layer, and maintains a moist environment. Hydrocolloids are not recommended for infected wounds or for those with copious exudate as they may lead to maceration around the wound. A disadvantage of hydrocolloid dressings is their tendency to generate brown, often malodorous exudate when removed.
On the other hand, hydrogels in amorphous gel, dressing, sheet, or impregnated gauze form are water-based products that create a moist environment similar to hydrocolloids. Aside from causing minimal trauma to the wound bed when removed, the dressing’s cooling effect may bring some pain relief. Hydrogels are appropriate for dry wounds and for those with minimal exudate.
After debridement, the wound is cleansed and irrigated. A number of cleansers and solutions are available, but normal saline is a cheap alternative. To irrigate, experts recommend an 18- or 20-gauge intravenous catheter attached to a 30- or 60-mL syringe.15 This technique provides 8 to 15 psi of pressure, enough to cleanse the wound without causing tissue trauma.
Antibacterials and absorption
Antibacterials. Topical antibiotics have several advantages over systemic antibiotics in treating chronic wounds.30,31 These include a high and sustained concentration of the antimicrobial at the site of infection, limited potential for systemic absorption and toxicity, reduced potential for antibiotic resistance, and drawing of the patient’s and caregiver’s attention to the wound.
Metronidazole is the most widely used topical antibacterial for malodor management. Its efficacy is likely due to the predominant involvement of anaerobic bacteria in foul-smelling wounds. Topical metronidazole is available as a gel and as a cream. A systematic review showed that on average, topical metronidazole was used once daily for 14 consecutive days.19 The layer of topical metronidazole is typically covered with a nonadherent primary dressing followed by an absorbent secondary dressing.
The best clinical evidence for topical metronidazole consists of case reports and series.32–35 The largest of these studies was done by Finlay et al, who treated 47 patients with malodorous benign and malignant cutaneous wounds with 0.75% metronidazole gel daily.32 Forty-five (96%) of the patients reported significantly decreased odor by 14 days, as well as decreased pain, discharge, and surrounding cellulitis.
A randomized, placebo-controlled trial conducted by Bale et al had equivocal findings.9 All 41 patients who received metronidazole gel reported a decrease in malodor within 3 days of starting it. However, 76% of patients who received placebo also reported malodor control; in the final analysis, no significant difference was noted in the success rate between the two groups.
Metronidazole tablets can be crushed and sprinkled over the wound. As with metronidazole gel or cream, the crushed tablets are applied daily and covered by a primary nonadherent dressing and an absorbent secondary dressing. This off-label use of metronidazole serves as a cheaper alternative to commercially available topical preparations. To our knowledge, there has been no head-to-head trial comparing the two topical strategies.
Systemic metronidazole, often given orally, has been recommended if evidence of deep tissue or systemic infection is noted15 and in cases of fungating wounds with fistulas invading either the gastrointestinal or genitourinary tracts.18 Side effects such as nausea, neuropathy, and alcohol intolerance (ie, disulfiram reaction) may occur, which are not seen with topical metronidazole.
Both topical and systemic metronidazole can be used together on a time-limited basis for extensive malodorous wounds, such as fungating malignant wounds or stage IV sacral pressure ulcers.
Other antimicrobial agents used to treat malodor include silver-containing products, iodine-containing topical agents, mupirocin, bacitracin, neomycin, and polymyxin B.
Honey was used for wound care by the ancient Egyptians, and it is still used.36 Its beneficial effects include antimicrobial, debriding, deodorizing, anti-inflammatory, and granulation tissue-stimulating. Honey has even been shown to significantly decrease skin colonization with various kinds of bacteria, including methicillin-resistant Staphylococcus aureus.37 Medical-grade honey is preferred over table honey, as the latter is nonsterile and can contain Clostridium spores, which contaminate the wound.38
Yogurt and buttermilk lower the pH of the wound and control bacterial proliferation to control malodor.39,40 Either is applied for 10 to 15 minutes after the wound is cleansed and is then washed off thoroughly.
Absorbent dressings are used either over a layer of topical metronidazole and a nonadherent primary dressing or as a primary dressing itself. An absorbent dressing containing activated charcoal is used for rapid improvement, although cost may be prohibitive, especially in developing countries.13,19 Another type of absorbent dressing, composed of polyester impregnated with sodium chloride, has been found to be useful in malodor control.41 An important pointer is to maintain a tight seal around the absorbent dressing to prevent leakage of exudate.
Concealers
Aromatics used to conceal malodor include scented candles, incense, fragrant flowers and plants, and air-freshener sprays. When circumstances allow, candles are good options since they conceal malodor by emitting fragrance, and the flame burns off foul-smelling chemicals. Aromatics such as coffee beans, vanilla beans, and cider vinegar can be placed in a pan and left under the patient’s bed or close to it. Drops of peppermint oil or oil of wintergreen can be placed on wound dressings.
Other odor concealers are adsorbent materials that attract and cause ions and molecules to adhere to their surface. Examples are charcoal, baking soda, and cat litter. As with other aromatics, these materials are placed in pans and left under the bed or near the patient.
Aromatics can have disadvantages, as certain scents, especially strong ones, can be nauseating for patients. Some fragrances trigger asthma or skin irritation. Patients and caregivers can be left with an unpleasant association of certain fragrances with malodor by conditioning.15,17,18
Education and support
Concerns of the patient and family members need to be heard, addressed promptly, and reassessed with each visit, since uncontrolled malodor can be a chief source of caregiver fatigue.
Foremost in formulating a patient- and family-centered malodor management strategy is to commit to controlling malodor as much as possible. Regular follow-up appointments should be made, whether in the office or at home, to check on the patient’s progress and address new and ongoing concerns. Symptoms accompanying malodor, such as pain, bleeding, and sleep disturbance, need to be addressed, as they all affect quality of life.1 Audience-appropriate educational materials should be made available.26 Online resources that patients and families can explore include the websites of the Wound Ostomy and Continence Nurses Society (www.wocn.org) and the Association for the Advancement of Wound Care (aawconline.org).
Healthcare professionals need to be prepared to deal with problems and complications involving patients and family members that may arise in the course of treatment.12 Problems include the cost and local unavailability of dressing supplies, insurance coverage for dressings and topical agents, lack of assistance at home, and fear of changing dressings. A cardinal rule for healthcare providers is to avoid expressing distress at odors in front of or within hearing of patients and families.
OTHER STRATEGIES: WHAT ELSE CAN WE DO?
Curcumin, the main biologically active compound in the herb turmeric, applied directly to wounds three times daily as an ointment, has been shown to have odor-controlling properties.42
Sugar paste has been reported to control malodor by drawing out exudative and tissue fluid osmotically, and inhibiting bacterial growth.16,17 Water is mixed with sugar (ie, granulated, caster, or powdered) to form a paste, with additives like glycerin and polyethylene glycol used to alter the consistency. Thick clay-like paste is good for wounds with large cavities, while thin paste is useful for wounds with small or superficial openings. The paste is applied twice daily and is covered by an absorbent dressing.
Pressure relief is vital in managing pressure ulcers.18,43 Repositioning every 2 hours and using special devices, such as mattress overlays, alternating pressure mattresses, and low air loss mattresses, are frequently employed techniques.
If circumstances permit and when congruent with the patient’s goals of care, intra-arterial chemotherapy and radiotherapy can be contemplated for malignant fungating wounds.44,45
Other strategies include opening the windows during dressing changes, increasing the frequency of dressing changes, promptly removing used dressings from the house, and ensuring good general hygiene.
CASE RESOLUTION
After telling her that he was committed to control the malodor or, if possible, eliminate it, Mrs. A.’s doctor prepared two lists of materials—one for himself and one for Mrs. A.’s husband. He returned the next day, brought out his supplies, asked Mrs. A. to lie in bed, and invited her husband to assist him.
He cleansed and irrigated the breast lesion with normal saline, making sure to remove as much dead tissue as he could. He applied a layer of metronidazole cream to the wound cavity, then covered it with a nonadherent dressing. He then covered the wound with gauze, sealed the edges with medical adhesive tape, and applied a few drops of oil of wintergreen to the surface. A pan of charcoal briquettes was put under the bed, and a candle with Mrs. A.’s favorite scent was lit by the bedside. The physician then instructed Mrs. A.’s husband to repeat the procedure once daily for 1 week.
After 2 weeks, Mrs. A. and her husband said the foul odor had greatly decreased. She appeared more cheerful and energetic, especially after her grandchildren visited a few days earlier. The physician then instructed the husband to stop using metronidazole cream and to apply a hydrocolloid dressing every 3 days instead. He advised them to continue the rest of the process of applying a few drops of oil of wintergreen on the dressing surface, placing a pan of charcoal briquettes under the bed, and lighting a scented candle by the bedside.
FINISH THE RACE!
Complex nonhealing wounds are encountered across various healthcare settings. Wound malodor is an important component of nonhealing wounds, which adversely affects patients, families, and healthcare providers. Infection, slough, and exudate are the major causes of wound malodor. The essential steps to reduce malodor are to remove necrotic tissue, use antibacterial and odor-absorbing agents, apply appropriate odor “concealers,” educate families, and formulate a patient- and family-centered strategy (Table 1).
Acknowledgment: The authors would like to thank Sue Reif, CNP, for her assistance in completing the manuscript.
Wounds that fail to heal become more than mere skin lesions. Pain, malodor, and the accompanying psychological distress often complicate nonhealing wounds and impair quality of life.1 Management of malodor requires perseverance, sensitivity, and familiarity with tools and procedures that range from surgical debridement to medical-grade honey.
Chronic, nonhealing wounds are defined as persisting for more than 6 months.2 These lesions are incapable of undergoing anatomic and functional repair on their own. Commonly encountered nonhealing wounds include pressure ulcers, venous stasis ulcers, arterial insufficiency ulcers, and malignant cutaneous wounds.
Typically, the patient with a nonhealing wound is frail, debilitated, medically complex, and often faced with one or more life-limiting illnesses. Complete wound healing may therefore be unrealistic, and optimal wound management becomes the goal of care.3,4
Healthcare providers encounter nonhealing wounds in varied settings—acute inpatient, outpatient, long-term, and home care. For instance, in the home care setting, a study of 383 patients enrolled in hospice found that 35% had skin ulcers and wounds.3 Half of those affected had pressure ulcers, 20% had ischemic ulcers, and 30% had other skin disorders such as stasis ulcers, burns, skin tears, and tumors. A larger study, also in hospice patients, found that 26% had pressure ulcers and 10% more developed them within 6 months.5
While pressure ulcers are the most common nonhealing wounds, malignant or fungating wounds are found in 5% to 10% of patients with metastatic disease, usually with cancers of the breast, head, and neck.6
Maximizing wound care provides comfort, relieves suffering, and promotes quality of life.3,7 To achieve these goals, clinicians must be familiar with strategies to manage complications associated with nonhealing wounds such as pain, malodor, and psychosocial adverse effects. Of these complications, malodor has been pointed out by both patients and caregivers as the most distressing.8
This article focuses on wound malodor, discusses the processes that cause wounds to emit an offensive smell, and outlines a comprehensive management approach.
MRS. A., AGE 61, WITH STAGE IV BREAST CANCER
Mrs. A., 61 years old, had a fungating mass in her left breast, which began as a small nodule and progressively enlarged to deform her breast over several months. Her oncologist subsequently staged the extent of her cancer as stage IV after workup revealed lung metastasis. Mrs. A. and her family decided to forgo cancer treatment, including radiotherapy, and to transition to hospice care after discussions with the oncologist.
Mrs. A. lived at home with her husband. Her daughter and three grandchildren all lived nearby.
When her hospice physician arrived at her home to meet her, a strong, pungent, and nauseating smell greeted him as he entered her bedroom. The patient said that for the past few months she had been increasingly distressed by the revolting odor. She rarely left home and had been ashamed to have people visit her, including her family.
On examination, the physician noticed a large fungating mass with yellowish discharge and necrotic tissue in her left breast. In addition to mild pain, she was immensely bothered by the strong odor coming from her breast.
THE IMPACT OF MALODOR
As seen in the case of Mrs. A., malodor has grave effects, both physical and psychological. Patients experience impaired or socially unacceptable body image, social rejection, personal shame, and embarrassment.9,10 Feelings of fear, anxiety, and depression are common. If left uncontrolled, malodor results in social isolation, reluctance to engage in social activities, diminished appetite, and nausea. In addition, malodor is a constant reminder of patients’ pain and cancer, and it results in further suffering.11
Reactions of family members and caregivers can worsen the situation.9,12 Expressions of revulsion limit contact and inhibit intimacy, especially near the end of life. Caregivers are often frustrated and distressed over their inability to control the malodor. The environment becomes uninhabitable, and the malodor can permeate clothing, furniture, and living quarters.
Managing malodor can be emotionally draining, physically daunting, and frustrating for healthcare professionals, as several methods are usually employed, often in a trial-and-error approach, to achieve an acceptable degree of odor control. In addition, clinicians must face the challenge of treating malodorous wounds at very close distance without reacting in a way that offends or alarms patients and family members.13
MALODOR PRODUCTION: WHERE IS THAT SMELL COMING FROM?
All wounds can produce an odor.14 Wounds that are expected to heal typically emit a faint but not unpleasant odor, akin to fresh blood. Wounds colonized by Pseudomonas aeruginosa produce a fruity or grapelike odor that is tolerable. Malodor occurs with wounds infected by other gram-negative organisms or anaerobic bacteria.15 Similarly, wounds covered by necrotic tissue smell like decaying flesh.
Three major causes
The three major causes of wound malodor are slough, infection, and exudate (Figure 1).
Slough is dead or necrotic tissue, usually resulting from vascular compromise. Arterial ulcers, pressure ulcers, and malignant wounds all form slough from capillary occlusion, subsequent ischemia, and tissue necrosis.
Infection. Devitalized tissue, an ideal medium in which bacteria thrive, becomes the source of infection. Anaerobic bacteria are usually implicated in malodor. These include Bacteroides fragilis, Bacteroides prevotella, Clostridium perfringens, and Fusobacterium nucleatum.16,17 Anaerobic organisms produce putrescine and cadaverine, which are largely responsible for the offensive odor.16,18 Volatile fatty acids such as propionic, butyric, isovaleric, and valeric acid are formed from lipid catabolism by anaerobes and add to malodor.17 Aerobic bacteria such as Proteus, Klebsiella, and Pseudomonas species supercolonize necrotic tissue as well and contribute to malodor.17,18
Exudate. Since nonhealing wounds undergo repeated cycles of inflammation, infection, and necrosis, accumulation of exudate becomes inevitable. Exudate typically is a pus-like fluid containing serum, fibrin, and white blood cells, which leak from blood vessels. In addition, bacteria that colonize chronic wounds filled with necrotic tissue activate proteases that degrade and liquefy dead tissue, thereby forming extensive amounts of exudate.19
Apart from slough, infection, and exudate, poor general hygiene and dressings left on for too long may contribute to malodor.16 Moisture-retentive dressings such as hydrocolloids leave an odor after removal. Dressings that liquefy upon contact with the wound surface leave a pus-like, potentially malodorous material.
MALODOR ASSESSMENT: DO YOU SMELL SOMETHING?
Various ways to document wound malodor can prove useful in guiding assessment and treatment. Descriptions such as “foul,” “putrid,” “fishy,” or “filled the room” vividly portray the initial presentation. A 10-point numerical scale similar to a numerical pain scale or a visual analogue scale can be used as a subjective measure.
Other grading methods, which to the authors’ knowledge are not validated, may be helpful. In a study that focused on patients suffering from malodorous gynecologic malignancies, von Gruenigen et al20 used a 0-to-3 scale:
- 0 Absent
- 1 Not offensive
- 2 Offensive but tolerable
- 3 Offensive and intolerable.
A scale often adapted by other authors was devised by Baker and Haig,21 which clearly defines four classes:
- 1 Strong—odor is evident upon entering the room (6 to 10 feet from the patient) with the dressing intact
- 2 Moderate—odor is evident upon entering the room with dressing removed
- 3 Slight—odor is evident at close proximity to the patient when the dressing is removed
- 4 No odor—no odor is evident, even at the patient’s bedside with the dressing removed.
COMPREHENSIVE MANAGEMENT: HOW DO WE WIN THE ‘RACE’?
The acronym RACE outlines an approach to dealing with malodor. It stands for removal of necrotic tissue; antibacterials; odor concealers; and education and support (Table 1).
Remove necrotic tissue
An important step in eliminating malodor is to remove necrotic tissue. This starts with debridement, which decreases the incidence of infection and hastens wound closure.22,23 Table 2 compares the different types of debridement.
Sharp or surgical debridement involves the use of a scalpel or scissors. This type of debridement may increase the risk of bleeding, pain, and malignant cell seeding in fungating wounds.4,24
Enzymatic debridement employs chemicals with proteolytic action (eg, collagenase) to digest extracellular proteins in wounds.18,25
Mechanical debridement involves aggressive therapies such as forceful irrigation and hydrotherapy, which may fail to discriminate between necrotic and viable tissues.18,26
Biological debridement using maggots, which ingest bacteria and devitalized tissue, may cause increased wound bleeding and may be unacceptable for patients and families.24,27
Autolytic debridement is often recommended, particularly if complete healing is not the primary goal.17,24,28,29 Autolysis uses proteolytic enzymes and phagocytic cells present in the wound bed and wound fluid to clear devitalized tissue. It is easy, inexpensive, noninvasive, and painless,4 and it requires less frequent dressing changes relative to standard dressing or wet-to-dry dressing.
Autolytic debridement is commonly accomplished using hydrocolloid and hydrogel dressings.15,29 Hydrocolloids are adhesive, occlusive, and conformable dressings that are suitable for wounds with low to moderate amounts of exudate. Upon contact with the wound surface, the dressing absorbs the exudate, forms a gel layer, and maintains a moist environment. Hydrocolloids are not recommended for infected wounds or for those with copious exudate as they may lead to maceration around the wound. A disadvantage of hydrocolloid dressings is their tendency to generate brown, often malodorous exudate when removed.
On the other hand, hydrogels in amorphous gel, dressing, sheet, or impregnated gauze form are water-based products that create a moist environment similar to hydrocolloids. Aside from causing minimal trauma to the wound bed when removed, the dressing’s cooling effect may bring some pain relief. Hydrogels are appropriate for dry wounds and for those with minimal exudate.
After debridement, the wound is cleansed and irrigated. A number of cleansers and solutions are available, but normal saline is a cheap alternative. To irrigate, experts recommend an 18- or 20-gauge intravenous catheter attached to a 30- or 60-mL syringe.15 This technique provides 8 to 15 psi of pressure, enough to cleanse the wound without causing tissue trauma.
Antibacterials and absorption
Antibacterials. Topical antibiotics have several advantages over systemic antibiotics in treating chronic wounds.30,31 These include a high and sustained concentration of the antimicrobial at the site of infection, limited potential for systemic absorption and toxicity, reduced potential for antibiotic resistance, and drawing of the patient’s and caregiver’s attention to the wound.
Metronidazole is the most widely used topical antibacterial for malodor management. Its efficacy is likely due to the predominant involvement of anaerobic bacteria in foul-smelling wounds. Topical metronidazole is available as a gel and as a cream. A systematic review showed that on average, topical metronidazole was used once daily for 14 consecutive days.19 The layer of topical metronidazole is typically covered with a nonadherent primary dressing followed by an absorbent secondary dressing.
The best clinical evidence for topical metronidazole consists of case reports and series.32–35 The largest of these studies was done by Finlay et al, who treated 47 patients with malodorous benign and malignant cutaneous wounds with 0.75% metronidazole gel daily.32 Forty-five (96%) of the patients reported significantly decreased odor by 14 days, as well as decreased pain, discharge, and surrounding cellulitis.
A randomized, placebo-controlled trial conducted by Bale et al had equivocal findings.9 All 41 patients who received metronidazole gel reported a decrease in malodor within 3 days of starting it. However, 76% of patients who received placebo also reported malodor control; in the final analysis, no significant difference was noted in the success rate between the two groups.
Metronidazole tablets can be crushed and sprinkled over the wound. As with metronidazole gel or cream, the crushed tablets are applied daily and covered by a primary nonadherent dressing and an absorbent secondary dressing. This off-label use of metronidazole serves as a cheaper alternative to commercially available topical preparations. To our knowledge, there has been no head-to-head trial comparing the two topical strategies.
Systemic metronidazole, often given orally, has been recommended if evidence of deep tissue or systemic infection is noted15 and in cases of fungating wounds with fistulas invading either the gastrointestinal or genitourinary tracts.18 Side effects such as nausea, neuropathy, and alcohol intolerance (ie, disulfiram reaction) may occur, which are not seen with topical metronidazole.
Both topical and systemic metronidazole can be used together on a time-limited basis for extensive malodorous wounds, such as fungating malignant wounds or stage IV sacral pressure ulcers.
Other antimicrobial agents used to treat malodor include silver-containing products, iodine-containing topical agents, mupirocin, bacitracin, neomycin, and polymyxin B.
Honey was used for wound care by the ancient Egyptians, and it is still used.36 Its beneficial effects include antimicrobial, debriding, deodorizing, anti-inflammatory, and granulation tissue-stimulating. Honey has even been shown to significantly decrease skin colonization with various kinds of bacteria, including methicillin-resistant Staphylococcus aureus.37 Medical-grade honey is preferred over table honey, as the latter is nonsterile and can contain Clostridium spores, which contaminate the wound.38
Yogurt and buttermilk lower the pH of the wound and control bacterial proliferation to control malodor.39,40 Either is applied for 10 to 15 minutes after the wound is cleansed and is then washed off thoroughly.
Absorbent dressings are used either over a layer of topical metronidazole and a nonadherent primary dressing or as a primary dressing itself. An absorbent dressing containing activated charcoal is used for rapid improvement, although cost may be prohibitive, especially in developing countries.13,19 Another type of absorbent dressing, composed of polyester impregnated with sodium chloride, has been found to be useful in malodor control.41 An important pointer is to maintain a tight seal around the absorbent dressing to prevent leakage of exudate.
Concealers
Aromatics used to conceal malodor include scented candles, incense, fragrant flowers and plants, and air-freshener sprays. When circumstances allow, candles are good options since they conceal malodor by emitting fragrance, and the flame burns off foul-smelling chemicals. Aromatics such as coffee beans, vanilla beans, and cider vinegar can be placed in a pan and left under the patient’s bed or close to it. Drops of peppermint oil or oil of wintergreen can be placed on wound dressings.
Other odor concealers are adsorbent materials that attract and cause ions and molecules to adhere to their surface. Examples are charcoal, baking soda, and cat litter. As with other aromatics, these materials are placed in pans and left under the bed or near the patient.
Aromatics can have disadvantages, as certain scents, especially strong ones, can be nauseating for patients. Some fragrances trigger asthma or skin irritation. Patients and caregivers can be left with an unpleasant association of certain fragrances with malodor by conditioning.15,17,18
Education and support
Concerns of the patient and family members need to be heard, addressed promptly, and reassessed with each visit, since uncontrolled malodor can be a chief source of caregiver fatigue.
Foremost in formulating a patient- and family-centered malodor management strategy is to commit to controlling malodor as much as possible. Regular follow-up appointments should be made, whether in the office or at home, to check on the patient’s progress and address new and ongoing concerns. Symptoms accompanying malodor, such as pain, bleeding, and sleep disturbance, need to be addressed, as they all affect quality of life.1 Audience-appropriate educational materials should be made available.26 Online resources that patients and families can explore include the websites of the Wound Ostomy and Continence Nurses Society (www.wocn.org) and the Association for the Advancement of Wound Care (aawconline.org).
Healthcare professionals need to be prepared to deal with problems and complications involving patients and family members that may arise in the course of treatment.12 Problems include the cost and local unavailability of dressing supplies, insurance coverage for dressings and topical agents, lack of assistance at home, and fear of changing dressings. A cardinal rule for healthcare providers is to avoid expressing distress at odors in front of or within hearing of patients and families.
OTHER STRATEGIES: WHAT ELSE CAN WE DO?
Curcumin, the main biologically active compound in the herb turmeric, applied directly to wounds three times daily as an ointment, has been shown to have odor-controlling properties.42
Sugar paste has been reported to control malodor by drawing out exudative and tissue fluid osmotically, and inhibiting bacterial growth.16,17 Water is mixed with sugar (ie, granulated, caster, or powdered) to form a paste, with additives like glycerin and polyethylene glycol used to alter the consistency. Thick clay-like paste is good for wounds with large cavities, while thin paste is useful for wounds with small or superficial openings. The paste is applied twice daily and is covered by an absorbent dressing.
Pressure relief is vital in managing pressure ulcers.18,43 Repositioning every 2 hours and using special devices, such as mattress overlays, alternating pressure mattresses, and low air loss mattresses, are frequently employed techniques.
If circumstances permit and when congruent with the patient’s goals of care, intra-arterial chemotherapy and radiotherapy can be contemplated for malignant fungating wounds.44,45
Other strategies include opening the windows during dressing changes, increasing the frequency of dressing changes, promptly removing used dressings from the house, and ensuring good general hygiene.
CASE RESOLUTION
After telling her that he was committed to control the malodor or, if possible, eliminate it, Mrs. A.’s doctor prepared two lists of materials—one for himself and one for Mrs. A.’s husband. He returned the next day, brought out his supplies, asked Mrs. A. to lie in bed, and invited her husband to assist him.
He cleansed and irrigated the breast lesion with normal saline, making sure to remove as much dead tissue as he could. He applied a layer of metronidazole cream to the wound cavity, then covered it with a nonadherent dressing. He then covered the wound with gauze, sealed the edges with medical adhesive tape, and applied a few drops of oil of wintergreen to the surface. A pan of charcoal briquettes was put under the bed, and a candle with Mrs. A.’s favorite scent was lit by the bedside. The physician then instructed Mrs. A.’s husband to repeat the procedure once daily for 1 week.
After 2 weeks, Mrs. A. and her husband said the foul odor had greatly decreased. She appeared more cheerful and energetic, especially after her grandchildren visited a few days earlier. The physician then instructed the husband to stop using metronidazole cream and to apply a hydrocolloid dressing every 3 days instead. He advised them to continue the rest of the process of applying a few drops of oil of wintergreen on the dressing surface, placing a pan of charcoal briquettes under the bed, and lighting a scented candle by the bedside.
FINISH THE RACE!
Complex nonhealing wounds are encountered across various healthcare settings. Wound malodor is an important component of nonhealing wounds, which adversely affects patients, families, and healthcare providers. Infection, slough, and exudate are the major causes of wound malodor. The essential steps to reduce malodor are to remove necrotic tissue, use antibacterial and odor-absorbing agents, apply appropriate odor “concealers,” educate families, and formulate a patient- and family-centered strategy (Table 1).
Acknowledgment: The authors would like to thank Sue Reif, CNP, for her assistance in completing the manuscript.
- Lo SF, Hayter M, Hu WY, Tai CY, Hsu MY, Li YF. Symptom burden and quality of life in patients with malignant fungating wounds. J Adv Nurs 2012; 68:1312–1321.
- Lazarus GS, Cooper DM, Knighton DR, et al. Definitions and guidelines for assessment of wounds and evaluation of healing. Arch Dermatol 1994; 130:489–493.
- Tippett AW. Wounds at the end of life. Wounds 2005; 17:91–98.
- Burt T. Palliative care of pressure ulcers in long-term care. Ann Long-Term Care 2013; 21:20–28.
- Reifsnyder J, Magee HS. Development of pressure ulcers in patients receiving home hospice care. Wounds 2005; 17:74–79.
- Haisfield-Wolfe ME, Rund C. Malignant cutaneous wounds: a management protocol. Ostomy Wound Manage 1997; 43:56–66.
- O’Brien C. Malignant wounds: managing odour. Can Fam Physician 2012; 58:272–274.
- Gethin G, Grocott P, Probst S, Clarke E. Current practice in the management of wound odour: an international survey. Int J Nurs Stud 2014; 51:865–874.
- Bale S, Tebble N, Price P. A topical metronidazole gel used to treat malodorous wounds. Br J Nurs 2004; 13:S4–S11.
- Hack A. Malodorous wounds—taking the patient’s perspective into account. J Wound Care 2003; 12:319–321.
- Price E. Wound care. The stigma of smell. Nurs Times 1996; 92:71–72.
- Paul JC, Pieper BA. Topical metronidazole for the treatment of wound odor: a review of the literature. Ostomy Wound Manage 2008; 54:18–27.
- Lee G, Anand SC, Rajendran S, Walker I. Overview of current practice and future trends in the evaluation of dressings for malodorous wounds. J Wound Care 2006; 15:344–346.
- Cutting K, Harding K. Criteria for identifying wound infection. J Wound Care 1994; 3:198–201.
- McDonald A, Lesage P. Palliative management of pressure ulcers and malignant wounds in patients with advanced illness. J Palliat Med 2006; 9:285–295.
- Holloway S. Recognising and treating the causes of chronic malodorous wounds. Prof Nurse 2004; 19:380–384.
- Haughton W, Young T. Common problems in wound care: malodorous wounds. Br J Nurs 1995; 4:959–963.
- Alvarez OM, Kalinski C, Nusbaum J, et al. Incorporating wound healing strategies to improve palliation (symptom management) in patients with chronic wounds. J Palliat Med 2007; 10:1161–1189.
- da Costa Santos CM, de Mattos Pimenta CA, Nobre MR. A systematic review of topical treatments to control the odor of malignant fungating wounds. J Pain Symptom Manage 2010; 39:1065–1076.
- Von Gruenigen VE, Coleman RL, et al. Bacteriology and treatment of malodorous lower reproductive tract in gynecologic cancer patients. Obstet Gynecol 2000; 96:23–27.
- Baker PG, Haig G. Metronidazole in the treatment of chronic pressure sores and ulcers: a comparison with standard treatment in general practice. Practitioner 1981; 225:569–573.
- Whitney J, Phillips L, Aslam R, et al. Guidelines for the treatment of pressure ulcers. Wound Repair Regen 2006; 14:663–679.
- Williams D, Enoch S, Miller D, Harris K, Price P, Harding KG. Effect of sharp debridement using curette on recalcitrant nonhealing venous ulcers: a concurrently controlled, prospective cohort study. Wound Repair Regen 2005; 13:131–137.
- Bergstrom KJ. Assessment and management of fungating wounds. J Wound Ostomy Continence Nurs 2011: 38:31–37.
- Sinclair RD, Ryan TJ. Proteolytic enzymes in wound healing: the role of enzymatic debridement. Australas J Dermatol 1994; 35:35–41.
- Enoch S, Harding KG. Wound bed preparation: the science behind the removal of barriers to healing. Wounds 2003;15:213–229.
- Mumcuoglu KY. Clinical applications for maggots in wound care. Am J Clin Dermatol 2001; 2:219–227.
- Langemo DK, Black J; National Pressure Ulcer Advisory Panel. Pressure ulcers in individuals receiving palliative care: a National Pressure Ulcer Advisory Panel white paper. Adv Skin Wound Care 2010; 23:59–72.
- Fonder MA, Lazarus GS, Cowan DA, Aronson-Cook B, Kohli AR, Mamelak AJ. Treating the chronic wound: a practical approach to the care of nonhealing wounds and wound care dressings. J Am Acad Dermatol 2008; 58:185–206.
- Lio PA, Kaye ET. Topical antibacterial agents. Infect Dis Clin North Am 2004; 18:717–733.
- Gelmetti C. Local antibiotics in dermatology. Dermatol Ther 2008; 21:187–195.
- Finlay IG, Bowszyc J, Ramlau C, Gwiezdzinski Z. The effect of topical 0.75% metronidazole gel on malodorous cutaneous ulcers. J Pain Symptom Manage 1996; 11:158–162.
- Bower M, Stein R, Evans TR, Hedley A, Pert P, Coombes RC. A double-blind study of the efficacy of metronidazole gel in the treatment of malodorous fungating tumours. Eur J Cancer 1992; 28A:888–889.
- Kalinski C, Schnepf M, Laboy D, et al. Effectiveness of a topical formulation containing metronidazole for wound odor and exudate control. Wounds 2005; 17:84–90.
- Kuge S, Tokuda Y, Ohta M, et al. Use of metronidazole gel to control malodor in advanced and recurrent breast cancer. Jpn J Clin Oncol 1996; 26:207–210.
- Belcher J. A review of medical-grade honey in wound care. Br J Nurs 2012: 21:S4–S9.
- Kwakman PH, Van den Akker JP, Güçlü A, et al. Medical-grade honey kills antibiotic-resistant bacteria in vitro and eradicates skin colonization. Clin Infect Dis 2008; 46:1677–1682.
- Cooper RA, Jenkins L. A comparison between medical grade honey and table honeys in relation to antimicrobial efficacy. Wounds 2009; 21:29–36.
- Patel B, Cox-Hayley D. Managing wound odor #218. J Palliat Med 2010; 13:1286–1287.
- Schulte MJ. Yogurt helps to control wound odor. Oncol Nurs Forum 1993; 20:1262.
- Upright CA, Salton C, Roberts F, Murphy J. Evaluation of Mesalt dressings and continuous wet saline dressings in ulcerating metastatic skin lesions. Cancer Nurs 1994; 17:149–155.
- Kuttan R, Sudheeran PC, Josph CD. Turmeric and curcumin as topical agents in cancer therapy. Tumori 1987; 73:29–31.
- Bass MJ, Phillips LG. Pressure sores. Curr Probl Surg 2007; 44:101–143.
- Bufill JA, Grace WR, Neff R. Intra-arterial chemotherapy for palliation of fungating breast cancer: a case report and review of the literature. Am J Clin Oncol 1994; 17:118–124.
- Murakami M, Kuroda Y, Sano A, et al. Validity of local treatment including intraarterial infusion chemotherapy and radiotherapy for fungating adenocarcinoma of the breast: case report of more than 8-year survival. Am J Clin Oncol 2001; 24:388–391.
- Lo SF, Hayter M, Hu WY, Tai CY, Hsu MY, Li YF. Symptom burden and quality of life in patients with malignant fungating wounds. J Adv Nurs 2012; 68:1312–1321.
- Lazarus GS, Cooper DM, Knighton DR, et al. Definitions and guidelines for assessment of wounds and evaluation of healing. Arch Dermatol 1994; 130:489–493.
- Tippett AW. Wounds at the end of life. Wounds 2005; 17:91–98.
- Burt T. Palliative care of pressure ulcers in long-term care. Ann Long-Term Care 2013; 21:20–28.
- Reifsnyder J, Magee HS. Development of pressure ulcers in patients receiving home hospice care. Wounds 2005; 17:74–79.
- Haisfield-Wolfe ME, Rund C. Malignant cutaneous wounds: a management protocol. Ostomy Wound Manage 1997; 43:56–66.
- O’Brien C. Malignant wounds: managing odour. Can Fam Physician 2012; 58:272–274.
- Gethin G, Grocott P, Probst S, Clarke E. Current practice in the management of wound odour: an international survey. Int J Nurs Stud 2014; 51:865–874.
- Bale S, Tebble N, Price P. A topical metronidazole gel used to treat malodorous wounds. Br J Nurs 2004; 13:S4–S11.
- Hack A. Malodorous wounds—taking the patient’s perspective into account. J Wound Care 2003; 12:319–321.
- Price E. Wound care. The stigma of smell. Nurs Times 1996; 92:71–72.
- Paul JC, Pieper BA. Topical metronidazole for the treatment of wound odor: a review of the literature. Ostomy Wound Manage 2008; 54:18–27.
- Lee G, Anand SC, Rajendran S, Walker I. Overview of current practice and future trends in the evaluation of dressings for malodorous wounds. J Wound Care 2006; 15:344–346.
- Cutting K, Harding K. Criteria for identifying wound infection. J Wound Care 1994; 3:198–201.
- McDonald A, Lesage P. Palliative management of pressure ulcers and malignant wounds in patients with advanced illness. J Palliat Med 2006; 9:285–295.
- Holloway S. Recognising and treating the causes of chronic malodorous wounds. Prof Nurse 2004; 19:380–384.
- Haughton W, Young T. Common problems in wound care: malodorous wounds. Br J Nurs 1995; 4:959–963.
- Alvarez OM, Kalinski C, Nusbaum J, et al. Incorporating wound healing strategies to improve palliation (symptom management) in patients with chronic wounds. J Palliat Med 2007; 10:1161–1189.
- da Costa Santos CM, de Mattos Pimenta CA, Nobre MR. A systematic review of topical treatments to control the odor of malignant fungating wounds. J Pain Symptom Manage 2010; 39:1065–1076.
- Von Gruenigen VE, Coleman RL, et al. Bacteriology and treatment of malodorous lower reproductive tract in gynecologic cancer patients. Obstet Gynecol 2000; 96:23–27.
- Baker PG, Haig G. Metronidazole in the treatment of chronic pressure sores and ulcers: a comparison with standard treatment in general practice. Practitioner 1981; 225:569–573.
- Whitney J, Phillips L, Aslam R, et al. Guidelines for the treatment of pressure ulcers. Wound Repair Regen 2006; 14:663–679.
- Williams D, Enoch S, Miller D, Harris K, Price P, Harding KG. Effect of sharp debridement using curette on recalcitrant nonhealing venous ulcers: a concurrently controlled, prospective cohort study. Wound Repair Regen 2005; 13:131–137.
- Bergstrom KJ. Assessment and management of fungating wounds. J Wound Ostomy Continence Nurs 2011: 38:31–37.
- Sinclair RD, Ryan TJ. Proteolytic enzymes in wound healing: the role of enzymatic debridement. Australas J Dermatol 1994; 35:35–41.
- Enoch S, Harding KG. Wound bed preparation: the science behind the removal of barriers to healing. Wounds 2003;15:213–229.
- Mumcuoglu KY. Clinical applications for maggots in wound care. Am J Clin Dermatol 2001; 2:219–227.
- Langemo DK, Black J; National Pressure Ulcer Advisory Panel. Pressure ulcers in individuals receiving palliative care: a National Pressure Ulcer Advisory Panel white paper. Adv Skin Wound Care 2010; 23:59–72.
- Fonder MA, Lazarus GS, Cowan DA, Aronson-Cook B, Kohli AR, Mamelak AJ. Treating the chronic wound: a practical approach to the care of nonhealing wounds and wound care dressings. J Am Acad Dermatol 2008; 58:185–206.
- Lio PA, Kaye ET. Topical antibacterial agents. Infect Dis Clin North Am 2004; 18:717–733.
- Gelmetti C. Local antibiotics in dermatology. Dermatol Ther 2008; 21:187–195.
- Finlay IG, Bowszyc J, Ramlau C, Gwiezdzinski Z. The effect of topical 0.75% metronidazole gel on malodorous cutaneous ulcers. J Pain Symptom Manage 1996; 11:158–162.
- Bower M, Stein R, Evans TR, Hedley A, Pert P, Coombes RC. A double-blind study of the efficacy of metronidazole gel in the treatment of malodorous fungating tumours. Eur J Cancer 1992; 28A:888–889.
- Kalinski C, Schnepf M, Laboy D, et al. Effectiveness of a topical formulation containing metronidazole for wound odor and exudate control. Wounds 2005; 17:84–90.
- Kuge S, Tokuda Y, Ohta M, et al. Use of metronidazole gel to control malodor in advanced and recurrent breast cancer. Jpn J Clin Oncol 1996; 26:207–210.
- Belcher J. A review of medical-grade honey in wound care. Br J Nurs 2012: 21:S4–S9.
- Kwakman PH, Van den Akker JP, Güçlü A, et al. Medical-grade honey kills antibiotic-resistant bacteria in vitro and eradicates skin colonization. Clin Infect Dis 2008; 46:1677–1682.
- Cooper RA, Jenkins L. A comparison between medical grade honey and table honeys in relation to antimicrobial efficacy. Wounds 2009; 21:29–36.
- Patel B, Cox-Hayley D. Managing wound odor #218. J Palliat Med 2010; 13:1286–1287.
- Schulte MJ. Yogurt helps to control wound odor. Oncol Nurs Forum 1993; 20:1262.
- Upright CA, Salton C, Roberts F, Murphy J. Evaluation of Mesalt dressings and continuous wet saline dressings in ulcerating metastatic skin lesions. Cancer Nurs 1994; 17:149–155.
- Kuttan R, Sudheeran PC, Josph CD. Turmeric and curcumin as topical agents in cancer therapy. Tumori 1987; 73:29–31.
- Bass MJ, Phillips LG. Pressure sores. Curr Probl Surg 2007; 44:101–143.
- Bufill JA, Grace WR, Neff R. Intra-arterial chemotherapy for palliation of fungating breast cancer: a case report and review of the literature. Am J Clin Oncol 1994; 17:118–124.
- Murakami M, Kuroda Y, Sano A, et al. Validity of local treatment including intraarterial infusion chemotherapy and radiotherapy for fungating adenocarcinoma of the breast: case report of more than 8-year survival. Am J Clin Oncol 2001; 24:388–391.
KEY POINTS
- Necrotic tissue is a substrate for bacterial growth and should be debrided. A variety of methods can be used.
- Malodor is most often from infection with anaerobic organisms, which topical metronidazole and other agents can help control.
- An absorbent dressing should be used either as a primary dressing, or over a layer of topical metronidazole and a nonadherent primary dressing.
- Foremost in formulating a patient- and family-centered malodor management strategy is to commit to controlling it as much as possible.
Most Important Elements of End-of-Life Care
An Australian team conducted a literature review of expected deaths in the hospital—where the majority of deaths in the developed world occur—and identified elements of end-of-life care that are important to patients and families.1 Published in the British journal Palliative Medicine, the review of nine electronic data bases and 1859 articles released between 1990 and 2014 identified eight quantitative studies that met inclusion criteria.
The authors, led by Claudia Virdun, RN, of the faculty of health at the University of Technology in Sydney, found four end-of-life domains that were most important to both patients and families:
- Effective communication and shared decision-making;
- Expert care;
- Respectful and compassionate care; and
- Trust and confidence in clinicians.
Not all patients dying in hospitals receive best evidence-based palliative care, the authors note, adding that the “challenge for healthcare services is to act on this evidence, reconfigure care systems accordingly and ensure universal access to optimal end-of-life care within hospitals.”
Reference
- Virdun C, Luckett T, Davidson PM, Phillips J. Dying in the hospital setting: A systematic review of quantitative studies identifying the elements of end-of-life care that patients and their families rank as being most important [published online ahead of print April 28, 2015]. Palliat Med.
An Australian team conducted a literature review of expected deaths in the hospital—where the majority of deaths in the developed world occur—and identified elements of end-of-life care that are important to patients and families.1 Published in the British journal Palliative Medicine, the review of nine electronic data bases and 1859 articles released between 1990 and 2014 identified eight quantitative studies that met inclusion criteria.
The authors, led by Claudia Virdun, RN, of the faculty of health at the University of Technology in Sydney, found four end-of-life domains that were most important to both patients and families:
- Effective communication and shared decision-making;
- Expert care;
- Respectful and compassionate care; and
- Trust and confidence in clinicians.
Not all patients dying in hospitals receive best evidence-based palliative care, the authors note, adding that the “challenge for healthcare services is to act on this evidence, reconfigure care systems accordingly and ensure universal access to optimal end-of-life care within hospitals.”
Reference
- Virdun C, Luckett T, Davidson PM, Phillips J. Dying in the hospital setting: A systematic review of quantitative studies identifying the elements of end-of-life care that patients and their families rank as being most important [published online ahead of print April 28, 2015]. Palliat Med.
An Australian team conducted a literature review of expected deaths in the hospital—where the majority of deaths in the developed world occur—and identified elements of end-of-life care that are important to patients and families.1 Published in the British journal Palliative Medicine, the review of nine electronic data bases and 1859 articles released between 1990 and 2014 identified eight quantitative studies that met inclusion criteria.
The authors, led by Claudia Virdun, RN, of the faculty of health at the University of Technology in Sydney, found four end-of-life domains that were most important to both patients and families:
- Effective communication and shared decision-making;
- Expert care;
- Respectful and compassionate care; and
- Trust and confidence in clinicians.
Not all patients dying in hospitals receive best evidence-based palliative care, the authors note, adding that the “challenge for healthcare services is to act on this evidence, reconfigure care systems accordingly and ensure universal access to optimal end-of-life care within hospitals.”
Reference
- Virdun C, Luckett T, Davidson PM, Phillips J. Dying in the hospital setting: A systematic review of quantitative studies identifying the elements of end-of-life care that patients and their families rank as being most important [published online ahead of print April 28, 2015]. Palliat Med.
Fellowship Training in Hospice and Palliative Care: New Pathways for Surgeons
Hospice and Palliative Medicine (HPM) has been a medical subspecialty recognized by the American Board of Medical Specialties since 2006. The American Board of Surgery (ABS) is one of 10 medical boards that offers board certification in HPM. Cosponsorship of HPM board certification by the ABS is significant because it signals an understanding and appreciation that patients in need of hospice and palliative care frequently require surgical services and also recognizes that many surgical patients benefit from palliative care.
Two specific domains of palliative care have been identified as core competencies for all surgeons: pain management and communication skills (Otolaryngology Clinics of North America 2009;42:1-13). Incorporation of these basic domains of care is considered primary palliative care for surgeons and inherent to good surgical care. For surgeons interested in obtaining subspecialty training in HPM, fellowship training is now required (the experiential pathway is no longer available).
Surgeons most likely to benefit from this additional training are those involved in the interdisciplinary care of chronically disease, critically ill, or terminally ill patients. Apparent in this description is the need for surgical palliative care providers across the full range of surgical subspecialties: pediatric to adult providers, trauma/critical care to oncology, cardiovascular surgery to transplant surgery. Currently, there are fewer than 100 surgeons with subspecialty board certification in HPM, constituting <1% of all physicians board certified in HPM.
The nascent field of HPM and needs surgeons as do the growing number of patients who require hospice and palliative medicine services. There currently exists a critical shortage of HPM physicians. A 2010 report by a task force appointed by the American Academy of Hospice and Palliative Medicine to study HPM physician workforce found that an estimated 6,000-18,000 additional physicians were needed to staff the then existing hospice and hospital-based palliative care programs (J. Pain Symptom Manage. 2010;40:899-91). The authors concluded that the capacity of fellowship programs at that time was insufficient to fill the shortage and changes in graduate medical education funding and structures were needed to increase the capacity to train sufficient numbers of HPM physicians. There are currently 108 Accreditation Council for Graduate Medical Education–accredited fellowships, up from 63 in 2009.
Surgeons interested in pursuing subspecialty training in HPM must complete a 1-year ACGME-accredited fellowship. Surgeons currently board-certified in surgery are eligible to apply. Many fellowship training programs have trained, or are willing to consider applications from, mid-career physicians, including surgeons (personal communication via HPM fellowship program directors listserv). Beginning July 1, 2015, an important change in eligibility for HPM fellowships goes into effect: Surgical residents with 3 years of training are now eligible to apply for ACGME-accredited HPM fellowships. This change in eligibility opens up an important pathway to HPM board certification, similar to that currently available for Surgical Critical Care. Trainees who complete HPM fellowship training through this pathway will not be eligible to obtain board certification in HPM until they have successfully achieved their primary board certification through the ABS.
Surgeons currently board certified in HPM incorporate their HPM training in a variety of ways: Some practice HPM full-time as members of a multidisciplinary in-patient palliative medicine consultation service while others integrate their training into their daily surgical practice, often serving as a resource on issues of surgical palliative care for their surgical colleagues. In my practice, I spend 1 day a week as a consultant on our in-patient palliative medicine consultation service in addition to providing faculty supervision to our palliative medicine fellows in our weekly outpatient clinic. For the remainder of the week, I am a practicing surgical oncologist and have found my training in palliative medicine invaluable in my daily care of patients with a variety of malignancies.
Like many of my surgical colleagues with HPM board certification, I am also actively engaged in teaching medical students, residents, and fellows about a variety of topics in palliative medicine, from evidence-based management of malignant bowel obstruction to communication skills for breaking bad news. Incorporating palliative medicine into my surgical practice has been incredibly rewarding, both personally and professionally.
In summary, HPM is in critical need of specialty trained physicians, including surgeons. Fellowship training is currently available to mid-career surgeons and, beginning July 1, 2015, surgical residents with 3 years of clinical training. Surgeons with subspecialty training in HPM are certain to find a wealth of clinical and academic opportunities as well as a path to a personally and professionally rewarding career.
For surgeons interested in obtaining more information about HPM fellowship training, go to the for a full list of programs and other training information.
Dr. Fahy is an associate professor of surgery at the University of New Mexico, Albuquerque. She is a surgical oncologist who is also board certified in hospice and palliative medicine. Dr. Fahy does not have any relevant conflicts of interest to disclose.
Hospice and Palliative Medicine (HPM) has been a medical subspecialty recognized by the American Board of Medical Specialties since 2006. The American Board of Surgery (ABS) is one of 10 medical boards that offers board certification in HPM. Cosponsorship of HPM board certification by the ABS is significant because it signals an understanding and appreciation that patients in need of hospice and palliative care frequently require surgical services and also recognizes that many surgical patients benefit from palliative care.
Two specific domains of palliative care have been identified as core competencies for all surgeons: pain management and communication skills (Otolaryngology Clinics of North America 2009;42:1-13). Incorporation of these basic domains of care is considered primary palliative care for surgeons and inherent to good surgical care. For surgeons interested in obtaining subspecialty training in HPM, fellowship training is now required (the experiential pathway is no longer available).
Surgeons most likely to benefit from this additional training are those involved in the interdisciplinary care of chronically disease, critically ill, or terminally ill patients. Apparent in this description is the need for surgical palliative care providers across the full range of surgical subspecialties: pediatric to adult providers, trauma/critical care to oncology, cardiovascular surgery to transplant surgery. Currently, there are fewer than 100 surgeons with subspecialty board certification in HPM, constituting <1% of all physicians board certified in HPM.
The nascent field of HPM and needs surgeons as do the growing number of patients who require hospice and palliative medicine services. There currently exists a critical shortage of HPM physicians. A 2010 report by a task force appointed by the American Academy of Hospice and Palliative Medicine to study HPM physician workforce found that an estimated 6,000-18,000 additional physicians were needed to staff the then existing hospice and hospital-based palliative care programs (J. Pain Symptom Manage. 2010;40:899-91). The authors concluded that the capacity of fellowship programs at that time was insufficient to fill the shortage and changes in graduate medical education funding and structures were needed to increase the capacity to train sufficient numbers of HPM physicians. There are currently 108 Accreditation Council for Graduate Medical Education–accredited fellowships, up from 63 in 2009.
Surgeons interested in pursuing subspecialty training in HPM must complete a 1-year ACGME-accredited fellowship. Surgeons currently board-certified in surgery are eligible to apply. Many fellowship training programs have trained, or are willing to consider applications from, mid-career physicians, including surgeons (personal communication via HPM fellowship program directors listserv). Beginning July 1, 2015, an important change in eligibility for HPM fellowships goes into effect: Surgical residents with 3 years of training are now eligible to apply for ACGME-accredited HPM fellowships. This change in eligibility opens up an important pathway to HPM board certification, similar to that currently available for Surgical Critical Care. Trainees who complete HPM fellowship training through this pathway will not be eligible to obtain board certification in HPM until they have successfully achieved their primary board certification through the ABS.
Surgeons currently board certified in HPM incorporate their HPM training in a variety of ways: Some practice HPM full-time as members of a multidisciplinary in-patient palliative medicine consultation service while others integrate their training into their daily surgical practice, often serving as a resource on issues of surgical palliative care for their surgical colleagues. In my practice, I spend 1 day a week as a consultant on our in-patient palliative medicine consultation service in addition to providing faculty supervision to our palliative medicine fellows in our weekly outpatient clinic. For the remainder of the week, I am a practicing surgical oncologist and have found my training in palliative medicine invaluable in my daily care of patients with a variety of malignancies.
Like many of my surgical colleagues with HPM board certification, I am also actively engaged in teaching medical students, residents, and fellows about a variety of topics in palliative medicine, from evidence-based management of malignant bowel obstruction to communication skills for breaking bad news. Incorporating palliative medicine into my surgical practice has been incredibly rewarding, both personally and professionally.
In summary, HPM is in critical need of specialty trained physicians, including surgeons. Fellowship training is currently available to mid-career surgeons and, beginning July 1, 2015, surgical residents with 3 years of clinical training. Surgeons with subspecialty training in HPM are certain to find a wealth of clinical and academic opportunities as well as a path to a personally and professionally rewarding career.
For surgeons interested in obtaining more information about HPM fellowship training, go to the for a full list of programs and other training information.
Dr. Fahy is an associate professor of surgery at the University of New Mexico, Albuquerque. She is a surgical oncologist who is also board certified in hospice and palliative medicine. Dr. Fahy does not have any relevant conflicts of interest to disclose.
Hospice and Palliative Medicine (HPM) has been a medical subspecialty recognized by the American Board of Medical Specialties since 2006. The American Board of Surgery (ABS) is one of 10 medical boards that offers board certification in HPM. Cosponsorship of HPM board certification by the ABS is significant because it signals an understanding and appreciation that patients in need of hospice and palliative care frequently require surgical services and also recognizes that many surgical patients benefit from palliative care.
Two specific domains of palliative care have been identified as core competencies for all surgeons: pain management and communication skills (Otolaryngology Clinics of North America 2009;42:1-13). Incorporation of these basic domains of care is considered primary palliative care for surgeons and inherent to good surgical care. For surgeons interested in obtaining subspecialty training in HPM, fellowship training is now required (the experiential pathway is no longer available).
Surgeons most likely to benefit from this additional training are those involved in the interdisciplinary care of chronically disease, critically ill, or terminally ill patients. Apparent in this description is the need for surgical palliative care providers across the full range of surgical subspecialties: pediatric to adult providers, trauma/critical care to oncology, cardiovascular surgery to transplant surgery. Currently, there are fewer than 100 surgeons with subspecialty board certification in HPM, constituting <1% of all physicians board certified in HPM.
The nascent field of HPM and needs surgeons as do the growing number of patients who require hospice and palliative medicine services. There currently exists a critical shortage of HPM physicians. A 2010 report by a task force appointed by the American Academy of Hospice and Palliative Medicine to study HPM physician workforce found that an estimated 6,000-18,000 additional physicians were needed to staff the then existing hospice and hospital-based palliative care programs (J. Pain Symptom Manage. 2010;40:899-91). The authors concluded that the capacity of fellowship programs at that time was insufficient to fill the shortage and changes in graduate medical education funding and structures were needed to increase the capacity to train sufficient numbers of HPM physicians. There are currently 108 Accreditation Council for Graduate Medical Education–accredited fellowships, up from 63 in 2009.
Surgeons interested in pursuing subspecialty training in HPM must complete a 1-year ACGME-accredited fellowship. Surgeons currently board-certified in surgery are eligible to apply. Many fellowship training programs have trained, or are willing to consider applications from, mid-career physicians, including surgeons (personal communication via HPM fellowship program directors listserv). Beginning July 1, 2015, an important change in eligibility for HPM fellowships goes into effect: Surgical residents with 3 years of training are now eligible to apply for ACGME-accredited HPM fellowships. This change in eligibility opens up an important pathway to HPM board certification, similar to that currently available for Surgical Critical Care. Trainees who complete HPM fellowship training through this pathway will not be eligible to obtain board certification in HPM until they have successfully achieved their primary board certification through the ABS.
Surgeons currently board certified in HPM incorporate their HPM training in a variety of ways: Some practice HPM full-time as members of a multidisciplinary in-patient palliative medicine consultation service while others integrate their training into their daily surgical practice, often serving as a resource on issues of surgical palliative care for their surgical colleagues. In my practice, I spend 1 day a week as a consultant on our in-patient palliative medicine consultation service in addition to providing faculty supervision to our palliative medicine fellows in our weekly outpatient clinic. For the remainder of the week, I am a practicing surgical oncologist and have found my training in palliative medicine invaluable in my daily care of patients with a variety of malignancies.
Like many of my surgical colleagues with HPM board certification, I am also actively engaged in teaching medical students, residents, and fellows about a variety of topics in palliative medicine, from evidence-based management of malignant bowel obstruction to communication skills for breaking bad news. Incorporating palliative medicine into my surgical practice has been incredibly rewarding, both personally and professionally.
In summary, HPM is in critical need of specialty trained physicians, including surgeons. Fellowship training is currently available to mid-career surgeons and, beginning July 1, 2015, surgical residents with 3 years of clinical training. Surgeons with subspecialty training in HPM are certain to find a wealth of clinical and academic opportunities as well as a path to a personally and professionally rewarding career.
For surgeons interested in obtaining more information about HPM fellowship training, go to the for a full list of programs and other training information.
Dr. Fahy is an associate professor of surgery at the University of New Mexico, Albuquerque. She is a surgical oncologist who is also board certified in hospice and palliative medicine. Dr. Fahy does not have any relevant conflicts of interest to disclose.
ICOO: Approach to opioids for cancer pain evolves
BOSTON – Opioid abuse might be as much of a problem in patients with cancer pain as in those who need analgesia for another reason, according to palliative care physicians at the Dana Farber Cancer Institute who outlined their safeguards at the International Conference of Opioids.
“Let’s not lose sight of the fact that the very access to these medications, which can do so much good, is in jeopardy,” said Dr. Douglas E. Brandoff, a palliative-care attending physician at the cancer institute. In the current era of “unprecedented regulation and scrutiny,” Dr. Brandoff said, pain practices in cancer care must evolve “to keep up with the times.”
Evidence that opioid abuse among cancer patients rivals that of other patients prescribed those agents is limited but reasonably consistent, according to Dr. Brandoff. He cited several published studies, including a survey of hospices in which substance abuse and diversion were considered a problem in 38% (J. Palliat. Med. 2013;16:237-42) of patients.
“It’s a little disconcerting. This is hospice, right? This doesn’t happen in hospice, but unfortunately, it does,” Dr. Brandoff reported.
At the cancer institute, a multidisciplinary task force convened in 2013 has now produced numerous specific policies designed to protect patients and institutions from abuse of controlled pain medications. Those steps are not much different from those being increasingly employed in clinics for nonmalignant chronic pain, but they are applied uniformly in essentially every patient – not just those singled out for high risk.
One required step is the implementation of a prescription-monitoring program for every patient started on a narcotic drug in controlled substances schedule II or III, a benzodiazepine, or a department of public health scheduled IV or V controlled substance. Another is the use of a medication management agreement designed to educate patients about the benefits and risks of controlled substances and outline expectations. All patients and their clinicians are required to sign the agreement.
“If we have someone who is imminently dying within hours or days, then, no, I would not impose a management agreement expectation on them or myself,” said Dr. Brandoff, but he said that there are essentially no other exceptions.
The agreement, crafted with nonjudgmental language aimed at clarifying the goals of chronic pain relief, is entered into the medical record. It generally has been well accepted, according to Dr. Lida Nabati, also a palliative care attending physician at the cancer institute, Dr. Nabati, who participated with Dr. Brandoff in presenting the cancer institute’s safeguards, noted that patient resistance to the agreement often is a red flag for potential problems with abuse.
The movement to control opioid abuse in cancer patients is relatively new. At the time that the task force began, Dr. Nabati noted that few other institutions had formal policies in place even though others also were beginning to review their approach. As recently as 2014, a directive from the Department of Veterans Affairs for opioid therapy in chronic pain patients specifically excluded those with cancer, Dr. Nabati reported.
Yet, cancer “does not afford some magical protective effect” from the very same risk factors associated with opioid use in noncancer patients, such as anxiety, depression, or history of substance use, according to Dr. Brandoff. Rather, he suggested that the added stress of a cancer diagnosis could exacerbate those factors.
The implementation of strategies aimed at reducing the risk of opioid abuse in patients with chronic cancer pain is needed and timely, according to Dr. Mellar P. Davis, the co-chair of the 2015 ICOO meeting and director of the palliative medicine fellowship program, Taussig Cancer Institute, Cleveland Clinic.
In an interview, Dr. Davis applauded the types of strategies implemented at the cancer institute, which he believes protect the patient, the physician, and the institution. He believes that the patients might be the greatest beneficiaries when appropriate opioid use permits a gain in quality of life through effective but nondebilitating pain control.
Dr. Brandoff and Dr. Nabati reported having no financial disclosures.
BOSTON – Opioid abuse might be as much of a problem in patients with cancer pain as in those who need analgesia for another reason, according to palliative care physicians at the Dana Farber Cancer Institute who outlined their safeguards at the International Conference of Opioids.
“Let’s not lose sight of the fact that the very access to these medications, which can do so much good, is in jeopardy,” said Dr. Douglas E. Brandoff, a palliative-care attending physician at the cancer institute. In the current era of “unprecedented regulation and scrutiny,” Dr. Brandoff said, pain practices in cancer care must evolve “to keep up with the times.”
Evidence that opioid abuse among cancer patients rivals that of other patients prescribed those agents is limited but reasonably consistent, according to Dr. Brandoff. He cited several published studies, including a survey of hospices in which substance abuse and diversion were considered a problem in 38% (J. Palliat. Med. 2013;16:237-42) of patients.
“It’s a little disconcerting. This is hospice, right? This doesn’t happen in hospice, but unfortunately, it does,” Dr. Brandoff reported.
At the cancer institute, a multidisciplinary task force convened in 2013 has now produced numerous specific policies designed to protect patients and institutions from abuse of controlled pain medications. Those steps are not much different from those being increasingly employed in clinics for nonmalignant chronic pain, but they are applied uniformly in essentially every patient – not just those singled out for high risk.
One required step is the implementation of a prescription-monitoring program for every patient started on a narcotic drug in controlled substances schedule II or III, a benzodiazepine, or a department of public health scheduled IV or V controlled substance. Another is the use of a medication management agreement designed to educate patients about the benefits and risks of controlled substances and outline expectations. All patients and their clinicians are required to sign the agreement.
“If we have someone who is imminently dying within hours or days, then, no, I would not impose a management agreement expectation on them or myself,” said Dr. Brandoff, but he said that there are essentially no other exceptions.
The agreement, crafted with nonjudgmental language aimed at clarifying the goals of chronic pain relief, is entered into the medical record. It generally has been well accepted, according to Dr. Lida Nabati, also a palliative care attending physician at the cancer institute, Dr. Nabati, who participated with Dr. Brandoff in presenting the cancer institute’s safeguards, noted that patient resistance to the agreement often is a red flag for potential problems with abuse.
The movement to control opioid abuse in cancer patients is relatively new. At the time that the task force began, Dr. Nabati noted that few other institutions had formal policies in place even though others also were beginning to review their approach. As recently as 2014, a directive from the Department of Veterans Affairs for opioid therapy in chronic pain patients specifically excluded those with cancer, Dr. Nabati reported.
Yet, cancer “does not afford some magical protective effect” from the very same risk factors associated with opioid use in noncancer patients, such as anxiety, depression, or history of substance use, according to Dr. Brandoff. Rather, he suggested that the added stress of a cancer diagnosis could exacerbate those factors.
The implementation of strategies aimed at reducing the risk of opioid abuse in patients with chronic cancer pain is needed and timely, according to Dr. Mellar P. Davis, the co-chair of the 2015 ICOO meeting and director of the palliative medicine fellowship program, Taussig Cancer Institute, Cleveland Clinic.
In an interview, Dr. Davis applauded the types of strategies implemented at the cancer institute, which he believes protect the patient, the physician, and the institution. He believes that the patients might be the greatest beneficiaries when appropriate opioid use permits a gain in quality of life through effective but nondebilitating pain control.
Dr. Brandoff and Dr. Nabati reported having no financial disclosures.
BOSTON – Opioid abuse might be as much of a problem in patients with cancer pain as in those who need analgesia for another reason, according to palliative care physicians at the Dana Farber Cancer Institute who outlined their safeguards at the International Conference of Opioids.
“Let’s not lose sight of the fact that the very access to these medications, which can do so much good, is in jeopardy,” said Dr. Douglas E. Brandoff, a palliative-care attending physician at the cancer institute. In the current era of “unprecedented regulation and scrutiny,” Dr. Brandoff said, pain practices in cancer care must evolve “to keep up with the times.”
Evidence that opioid abuse among cancer patients rivals that of other patients prescribed those agents is limited but reasonably consistent, according to Dr. Brandoff. He cited several published studies, including a survey of hospices in which substance abuse and diversion were considered a problem in 38% (J. Palliat. Med. 2013;16:237-42) of patients.
“It’s a little disconcerting. This is hospice, right? This doesn’t happen in hospice, but unfortunately, it does,” Dr. Brandoff reported.
At the cancer institute, a multidisciplinary task force convened in 2013 has now produced numerous specific policies designed to protect patients and institutions from abuse of controlled pain medications. Those steps are not much different from those being increasingly employed in clinics for nonmalignant chronic pain, but they are applied uniformly in essentially every patient – not just those singled out for high risk.
One required step is the implementation of a prescription-monitoring program for every patient started on a narcotic drug in controlled substances schedule II or III, a benzodiazepine, or a department of public health scheduled IV or V controlled substance. Another is the use of a medication management agreement designed to educate patients about the benefits and risks of controlled substances and outline expectations. All patients and their clinicians are required to sign the agreement.
“If we have someone who is imminently dying within hours or days, then, no, I would not impose a management agreement expectation on them or myself,” said Dr. Brandoff, but he said that there are essentially no other exceptions.
The agreement, crafted with nonjudgmental language aimed at clarifying the goals of chronic pain relief, is entered into the medical record. It generally has been well accepted, according to Dr. Lida Nabati, also a palliative care attending physician at the cancer institute, Dr. Nabati, who participated with Dr. Brandoff in presenting the cancer institute’s safeguards, noted that patient resistance to the agreement often is a red flag for potential problems with abuse.
The movement to control opioid abuse in cancer patients is relatively new. At the time that the task force began, Dr. Nabati noted that few other institutions had formal policies in place even though others also were beginning to review their approach. As recently as 2014, a directive from the Department of Veterans Affairs for opioid therapy in chronic pain patients specifically excluded those with cancer, Dr. Nabati reported.
Yet, cancer “does not afford some magical protective effect” from the very same risk factors associated with opioid use in noncancer patients, such as anxiety, depression, or history of substance use, according to Dr. Brandoff. Rather, he suggested that the added stress of a cancer diagnosis could exacerbate those factors.
The implementation of strategies aimed at reducing the risk of opioid abuse in patients with chronic cancer pain is needed and timely, according to Dr. Mellar P. Davis, the co-chair of the 2015 ICOO meeting and director of the palliative medicine fellowship program, Taussig Cancer Institute, Cleveland Clinic.
In an interview, Dr. Davis applauded the types of strategies implemented at the cancer institute, which he believes protect the patient, the physician, and the institution. He believes that the patients might be the greatest beneficiaries when appropriate opioid use permits a gain in quality of life through effective but nondebilitating pain control.
Dr. Brandoff and Dr. Nabati reported having no financial disclosures.
EXPERT ANALYSIS AT THE INTERNATIONAL CONFERENCE ON OPIOIDS
ICOO: Opioid self-dosing falls short of pain control
BOSTON – Many cancer patients do not pursue or at least do not achieve complete freedom from pain when permitted control over their opioid dose, according to a comprehensive analysis of published studies that evaluated patient-controlled analgesia.
“We do not know why. Patients were encouraged in these studies to titrate opioids until they were pain free or until they had side effects. Although this could be an issue of side effects, another interpretation is that complete pain control is not the goal for many individuals,” reported Dr. Brian H. Wetherington of the University of Kentucky, Lexington.
The data from this analysis were presented at the International Conference on Opioids from a comprehensive literature search that included 905 potentially relevant articles. Of these, 62 met inclusion criteria, particularly an assessment of patient-controlled opioids in patients with cancer pain. The studies also had to assess pain control with a visual analog scale (VAS) or the Neuropathy Pain Scale (NPS) using a 10-point system with 10 being the greatest level of pain imaginable.
“We were interested in evaluating whether patients, when given complete control over their opioids, would take sufficient doses to provide complete pain relief, which is often stated as the goal in pain management,” explained Dr. Wetherington, who was coauthor of a study led by his colleague at University of Kentucky, Dr. Michael Harned.
The answer was no. When the data from the 62 studies, which included 5,251 patients with cancer pain were collated, the average pain score at baseline was 5.4. At the time of assessment of pain control, the mean pain score was 2.7.
“The mean pain score for patients managing their own cancer pain on opioids was reduced from study entry but remained at the moderate to severe pain level or higher than what many health care providers would recommend,” Dr. Wetherington reported.
This review of published studies does not explain why lower pain scores are not reached, but the Dr. Wetherington and his coauthors hypothesized that patients are demonstrating their own benefit-to-risk ratio assessment.
This is thought to be the first systematic review to find that patients do not seek complete control of pain when given access to unrestricted analgesia, but several individual studies have made the same point. In one study cited by the authors, patients on a fentanyl patch only reduced their pain scores to 3.0 on average when given unlimited access to oral morphine for breakthroughs (J. Pain Symptom Manage. 1998;16:102-11).
“We think this deserves further study, because there may be lessons regarding how we think of optimal pain control. While the therapeutic target is often described as complete pain relief, these data suggest that this may not be the goal for patients when they are left to select their own level of pain control,” Dr. Wetherington explained.
The same observation regarding the failure of patients to eliminate all pain on patient-controlled analgesia has been made anecdotally by Dr. William G. Brose of Stanford (Calif.) University. However, he suggested in an interview that patients might be reluctant to rate themselves completely pain free on a subjective scale. He also believes that level of analgesia may not be the most relevant endpoint.
“We are increasingly evaluating change in patient function, which may be a more useful tool for evaluating the efficacy of pain control,” Dr. Brose said.
BOSTON – Many cancer patients do not pursue or at least do not achieve complete freedom from pain when permitted control over their opioid dose, according to a comprehensive analysis of published studies that evaluated patient-controlled analgesia.
“We do not know why. Patients were encouraged in these studies to titrate opioids until they were pain free or until they had side effects. Although this could be an issue of side effects, another interpretation is that complete pain control is not the goal for many individuals,” reported Dr. Brian H. Wetherington of the University of Kentucky, Lexington.
The data from this analysis were presented at the International Conference on Opioids from a comprehensive literature search that included 905 potentially relevant articles. Of these, 62 met inclusion criteria, particularly an assessment of patient-controlled opioids in patients with cancer pain. The studies also had to assess pain control with a visual analog scale (VAS) or the Neuropathy Pain Scale (NPS) using a 10-point system with 10 being the greatest level of pain imaginable.
“We were interested in evaluating whether patients, when given complete control over their opioids, would take sufficient doses to provide complete pain relief, which is often stated as the goal in pain management,” explained Dr. Wetherington, who was coauthor of a study led by his colleague at University of Kentucky, Dr. Michael Harned.
The answer was no. When the data from the 62 studies, which included 5,251 patients with cancer pain were collated, the average pain score at baseline was 5.4. At the time of assessment of pain control, the mean pain score was 2.7.
“The mean pain score for patients managing their own cancer pain on opioids was reduced from study entry but remained at the moderate to severe pain level or higher than what many health care providers would recommend,” Dr. Wetherington reported.
This review of published studies does not explain why lower pain scores are not reached, but the Dr. Wetherington and his coauthors hypothesized that patients are demonstrating their own benefit-to-risk ratio assessment.
This is thought to be the first systematic review to find that patients do not seek complete control of pain when given access to unrestricted analgesia, but several individual studies have made the same point. In one study cited by the authors, patients on a fentanyl patch only reduced their pain scores to 3.0 on average when given unlimited access to oral morphine for breakthroughs (J. Pain Symptom Manage. 1998;16:102-11).
“We think this deserves further study, because there may be lessons regarding how we think of optimal pain control. While the therapeutic target is often described as complete pain relief, these data suggest that this may not be the goal for patients when they are left to select their own level of pain control,” Dr. Wetherington explained.
The same observation regarding the failure of patients to eliminate all pain on patient-controlled analgesia has been made anecdotally by Dr. William G. Brose of Stanford (Calif.) University. However, he suggested in an interview that patients might be reluctant to rate themselves completely pain free on a subjective scale. He also believes that level of analgesia may not be the most relevant endpoint.
“We are increasingly evaluating change in patient function, which may be a more useful tool for evaluating the efficacy of pain control,” Dr. Brose said.
BOSTON – Many cancer patients do not pursue or at least do not achieve complete freedom from pain when permitted control over their opioid dose, according to a comprehensive analysis of published studies that evaluated patient-controlled analgesia.
“We do not know why. Patients were encouraged in these studies to titrate opioids until they were pain free or until they had side effects. Although this could be an issue of side effects, another interpretation is that complete pain control is not the goal for many individuals,” reported Dr. Brian H. Wetherington of the University of Kentucky, Lexington.
The data from this analysis were presented at the International Conference on Opioids from a comprehensive literature search that included 905 potentially relevant articles. Of these, 62 met inclusion criteria, particularly an assessment of patient-controlled opioids in patients with cancer pain. The studies also had to assess pain control with a visual analog scale (VAS) or the Neuropathy Pain Scale (NPS) using a 10-point system with 10 being the greatest level of pain imaginable.
“We were interested in evaluating whether patients, when given complete control over their opioids, would take sufficient doses to provide complete pain relief, which is often stated as the goal in pain management,” explained Dr. Wetherington, who was coauthor of a study led by his colleague at University of Kentucky, Dr. Michael Harned.
The answer was no. When the data from the 62 studies, which included 5,251 patients with cancer pain were collated, the average pain score at baseline was 5.4. At the time of assessment of pain control, the mean pain score was 2.7.
“The mean pain score for patients managing their own cancer pain on opioids was reduced from study entry but remained at the moderate to severe pain level or higher than what many health care providers would recommend,” Dr. Wetherington reported.
This review of published studies does not explain why lower pain scores are not reached, but the Dr. Wetherington and his coauthors hypothesized that patients are demonstrating their own benefit-to-risk ratio assessment.
This is thought to be the first systematic review to find that patients do not seek complete control of pain when given access to unrestricted analgesia, but several individual studies have made the same point. In one study cited by the authors, patients on a fentanyl patch only reduced their pain scores to 3.0 on average when given unlimited access to oral morphine for breakthroughs (J. Pain Symptom Manage. 1998;16:102-11).
“We think this deserves further study, because there may be lessons regarding how we think of optimal pain control. While the therapeutic target is often described as complete pain relief, these data suggest that this may not be the goal for patients when they are left to select their own level of pain control,” Dr. Wetherington explained.
The same observation regarding the failure of patients to eliminate all pain on patient-controlled analgesia has been made anecdotally by Dr. William G. Brose of Stanford (Calif.) University. However, he suggested in an interview that patients might be reluctant to rate themselves completely pain free on a subjective scale. He also believes that level of analgesia may not be the most relevant endpoint.
“We are increasingly evaluating change in patient function, which may be a more useful tool for evaluating the efficacy of pain control,” Dr. Brose said.
AT ICOO 2015
Key clinical point: Given the opportunity, patients do not titrate opioid therapy to a point of complete pain control, according to a comprehensive survey of published studies.
Major finding: In a survey of 62 published studies of cancer patients who were provided unlimited access to opioids for pain control, the average pain control was 2.7 on a scale of 10, indicating that most patients do not seek or are unable to achieve complete control with an acceptable benefit-to-risk ratio.
Data source: Retrospective data review.
Disclosures: The study was investigator initiated. Dr. Wetherington reported having no financial disclosures.
Palliative Radiotherapy for the Management of Metastatic Cancer
In recent years, there has been increasing interest in palliative care for patients with cancer at the end of life. Up to 23% of patients have metastatic disease at presentation, and symptoms from metastatic lesions can cause significant anxiety and impair patients’ quality of life (QOL).1
Palliative radiotherapy (RT) plays a valuable role in the management of metastatic disease to relieve tumor-related symptoms. Although palliative RT does not provide a chance for a cure, it improves QOL and may prolong survival time.2-4 An estimated 20% to 50% of radiation courses are prescribed with palliative intent, because RT is highly effective in providing symptom relief, and the toxicity associated with palliative doses is typically mild.5,6 Palliative RT can be used to manage bone and brain metastases, prevent or treat spinal cord compression, and manage numerous tumor-related symptoms, such as pain and bleeding in patients with terminal cancer.
Palliative RT for bone and brain metastases is supported by high-quality evidence and is considered one of the most effective and cost-effective options available.7,8 This article aims to review the role of RT in treating 3 conditions commonly encountered in patients with metastatic disease—bone metastases, spinal cord compression, and brain metastases—and to emphasize the importance of timely integration of RT for optimal results.
Bone Metastases
About 80% of patients with metastatic solid tumors develop bone metastases, and about 350,000 deaths are linked to bone metastases in the U.S. each year.9 Osseous
metastases can lead to pain, fracture, hypercalcemia, and spinal cord compression. The primary modality for treatment of pain and prevention of morbidity from bone metastases is external beam RT.10
The likelihood of bone pain relief with palliative RT is 60% to 80%, and 30% to 40% of patients achieving complete pain relief. Randomized studies have shown multiple-dose and fractionation regimens provided effective symptom relief for bone metastases. Most commonly used regimens include a single fraction of 8 gray (Gy) delivered in 1 treatment, 20 Gy in 5 fractions delivered daily over 1 week, and 30 Gy in 10 fractions delivered over 2 weeks. Treatment with a single fraction improves access to treatment and patient convenience, whereas more prolonged courses have been associated with lower rates of retreatment.11,12 Regarding the higher rate of retreatment with single-fraction RT, no clear evidence exists that this is due to a less durable pain response or lower level of pain relief.13
There has been recent interest in using predictive models to estimate life expectancy to avoid long courses of RT at the end of life.14,15 Shorter treatment courses of 8 Gyonce or 20 Gy in 5 fractions are particularly valuable for patients with a life expectancy < 3 months to avoid long courses of treatment, and thereby improve QOL as patients transition into hospice. A recent survey demonstrated that 93% of radiation oncologists within the VHA are willing to prescribe short courses of RT consisting of ≤ 6 fractions, and 76% have experience with single-fraction RT.16 These findings are in contradiction to the findings in the non-VA radiation oncology community, in which < 10% of patients with uncomplicated bone metastases are treated with a single fraction.17,18
In addition to providing pain relief, RT is used in the treatment of impending fractures either, adjuvant after surgical stabilization or alone for lower risk lesions.19 Factors that impact fracture risk include location of the metastasis (weight-bearing bones, such as femurs, which are at particularly high risk), length of bone involved, and extent of cortical involvement. Mirels’ scoring system was developed to predict fracture risk in patients with bone metastasis, based on 4 criteria: the
extent of cortical involvement, the location of the metastasis, the osteolytic vs osteoblastic appearance of the lesion, and the degree of pain.20 Surgical fixation can be considered, based on the total score and corresponding fracture risk. When appropriate, surgical stabilization should be considered by an orthopedic surgeon prior to initiating RT.
Postoperative RT after surgical stabilization has been associated with a reduced rate of secondary surgical procedures as well as with improved functional status.21 Radiotherapy promotes remineralization and bone healing and prevents the loss of surgical fixation by treating any residual tumor. A retrospective review of 60 patients with metastatic disease in weight-bearing bones with pathologic fracture or impending pathologic fracture demonstrated that surgery followed by RT was associated with improved functional status as well as with improved overall survival (OS).22,23 For patients in whom surgery is not indicated, the consulting radiation oncologist should consider factors such as the location of the metastasis in weight-bearing vs nonweight bearing bones, the size and extent of the metastasis, and associated symptoms when making a treatment recommendation. In patients at fracture risk from bone metastases, bisphosphonates should also be considered as part of the treatment regimen.24
Spinal Cord Compression
About 5% to 10% of patients diagnosed with cancer will develop spinal cord compression during the course of their disease.25 Spinal cord compression is considered a medical emergency that can result in significant pain and neurologic symptoms, including weakness, paralysis, parasthesias, and incontinence. Early treatment of spinal cord compression can prevent onset or progression of these symptoms; furthermore, early treatment prior to loss of ambulation is associated with improved long-term ambulatory function.26,27
Treatment decisions for spinal metastases with an associated concern for cord compression should be made after a consultation with both a neurosurgeon and a radiation oncologist. Early initiation of steroids is recommended to aid in tumor shrinkage for potential symptom relief.28 A standard way to administer dexamethasone is with a 10-mg loading dose followed by 16 mg per day, divided into 4 doses of 4 mg. Higher steroid doses showed no benefit in a prospective randomized trial comparing 96 mg with 16 mg of dexamethasone daily.29
Surgical decompression should be considered initial management of spinal cord compression. For patients treated surgically, local RT is indicated postoperatively as well. Randomized data show that surgery followed by RT provides better ambulatory function than does RT alone in patients with paralysis of < 2 days’ duration.30 Some patients with metastatic disease are not good candidates for surgery due to comorbidities, poor performance status, life expectancy < 3 months, or multilevel spinal involvement.
In patients who are not operative candidates, radiation alone is an appropriate alternative. However, several factors need consideration in deciding whether to manage cord compression with surgery followed by RT vs RT alone. These factors include life expectancy, tumor type (myeloma and lymphoma are more radiosensitive), interval since tumor diagnosis, and the presence of visceral metastases.31 Factors favoring surgical decompression plus postoperative RT over RT alone include spinal instability, KPS (Karnofsky Performance Status) > 70, radio-resistant tumor histology, minimal metastatic disease, and projected survival > 3 months.10
For patients managed with RT alone, early diagnosis and treatment is associated with improved outcomes. A prospective study of patients treated with RT without surgery for spinal cord compression demonstrated that 82% of patients experienced back pain relief, 76% achieved improvement in or preservation of ambulation, and 44% of patients with sphincter dysfunction experienced improvement with treatment.32 Patients with certain tumor histologies, such as myeloma, breast cancer, and prostate cancer, had better responses to RT.32
In the setting of spinal cord compression, longer courses of RT may provide better local control than do shorter courses.33 Therefore, longer courses of RT, such as 30 Gy in 10 fractions delivered over 2 weeks, are often preferred in cases of spinal cord compression treated with definitive RT as well as after surgical decompression. However, overall life expectancy is an important factor considered by the treating radiation oncologist when selecting a short course vs a longer course of RT.
In the instance of painful vertebral body metastases without spinal cord compression, a new subset analysis of the Radiation Therapy Oncology Group (RTOG) 9714 randomized trial indicated that single fraction RT (8 Gy) is just as effective as multiple fractions (30 Gy in 10 fractions), with this study demonstrating comparable rates of pain relief and narcotic use in both groups 3 months after RT.34 Advantages to the single-fraction plan compared with those of multiple fractions include mitigation of logistic concerns for patients and family at the end of life and less acute adverse effects.
Brain Metastases
An estimated 20% to 40% of patients with cancer develop brain metastases.35 The incidence of brain metastases has been rising most likely due to improved detection rates with magnetic resonance imaging (MRI) and improved cancer survival, because treatment regimens have improved with targeted chemotherapy and radiation techniques. Currently, the annual incidence of brain metastases is 170,000 to 200,000 in the U.S.36 Prognosis for these patients is poor, with median survival of 1 month without treatment and about 4 months with whole brain RT (WBRT) (Table).25,37-39
The goal of management for patients with brain metastases is to prevent or treat neurologic symptoms and to prolong survival. Treatment options include corticosteroids, WBRT, surgery, and stereotactic radiosurgery (SRS). Recommendations for treatment should involve both a radiation oncologist and neurosurgeon to determine the best treatment for an individual based on patient age, performance status, extent of systemic disease, and number of brain metastases. These prognostic factors that may predict life expectancy and impact treatment recommendations.40
Factors that have been correlated with improved survival include younger age, better performance status, fewer brain metastases, and lower burden of systemic disease.41,42 Prognostic assessment tools such as the Graded Prognostic Assessment and RTOG-Recursive Partitioning Analysis can be used to predict life expectancy in patients with brain metastases.41,43 However, routine use of these tools is lagging, as evidenced by a recent survey of VHA radiation oncologists. Use of these tools in the clinic will enhance the quality of end of life care and decision making.
Corticosteroids have classically been used in the treatment of brain metastases either alone for supportive care or in combination with RT. Steroids are recommended to provide symptom relief in patients with symptoms related to cerebral edema or mass effect.44 Steroids have been shown to mitigate edema and improve neurologic deficits in about two-thirds of patients with brain metastases.36,45 The effect of corticosteroids is thought to be mediated through inhibition of prostaglandin synthesis, reduction in vascular permeability, and anti-inflammatory properties.46 A common corticosteroid regimen is a 10-mg loading dose of dexamethasone, followed by 16 mg daily in divided doses. For patients without neurologic deficits or cerebral edema, it is reasonable to defer corticosteroid use only when patients are symptomatic.
In general, WBRT is considered an appropriate treatment option for patients with multiple brain metastases based on data suggesting an improvement in OS compared with the use of corticosteroids alone.47 Whole brain radiation has been shown to result in the improvement of baseline neurologic deficits or the prevention of further symptom progression.48 The partial or complete metastasis response rates are on the order of 60%.38 Tumor regression after WBRT has been associated with preservation of neurocognitive function as well as prolonged survival.49
For good prognosis patients with a single brain metastasis and good performance status, the use of surgery or radiosurgery added to WBRT has been associated with improved OS (Table). The RTOG 9508 randomized trial of WBRT with or without SRS demonstrated a survival advantage with SRS, with median survival times of 6.5 months with WBRT + SRS vs 4.9 months with WBRT alone.50 Similarly, a randomized trial evaluating WBRT alone compared with surgery followed by WBRT in patients
with good prognosis demonstrated significantly improved OS in the surgery group (median 40 weeks vs 15 weeks).51 In general, WBRT or postoperative RT to the tumor bed is still indicated after surgical resection, based on randomized data showing a reduction in tumor bed recurrence with postoperative RT.52
For patients with only 1 to 3 brain metastases and a favorable prognosis, surgery and SRS can be considered treatment options, oftentimes with WBRT. The EORTC randomized trial of patients with 1 to 3 brain metastases was designed to determine the benefit of WBRT after treatment with surgery or SRS. In this study, 119 patients underwent SRS and 160 patients underwent surgical resection.53 Both groups of patients were randomized to observation vs adjuvant WBRT. This study demonstrated reduced rates of intracranial relapse with WBRT, however, without any change in OS. Although there is concern that WBRT may impair cognitive function with no clear survival benefit after surgery or SRS, WBRT does reduce recurrence rates in the brain and the need for further treatment.54 Therefore, decisions regarding WBRT in such a setting should be made only after a detailed discussion with a radiation oncologist regarding risks vs benefits of treatment as part of the informed decision-making process.
Conclusions
Palliative RT plays an important role in the management of metastatic cancer to provide symptom relief and is a cost-effective treatment option for bone and brain metastases. Life expectancy and tumor characteristics should be considered when making treatment recommendations to ensure selection of regimens that complement patients’ unique situations. Timely referrals for treatment are important to optimize treatment results.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Click here to read the digital edition.
1. Porter A and David M. Palliative care for bone, spinal cord, brain, and liver metastases. In: Gunderson LL, Tepper JE, eds. Clinical Radiation Oncology. 2nd ed. Philadelphia, PA: Elsevier Churchill Livingstone; 2007:437-451.
2. Yamaguchi S, Ohguri T, Matsuki Y, et al. Palliative radiotherapy in patients with a poor performance status: the palliative effect is correlated with prolongation of survival time. Radiat Oncol. 2013;8:166.
3. Mac Manus MP, Matthews JP, Wada M, Wirth A, Worotniuk V, Ball DL. Unexpected long-term survival after low-dose palliative radiotherapy for non-small cell lung cancer. Cancer. 2006;106(5):1110-1116.
4. Rastogi M, Revannasiddaiah S, Gupta MK, Seam RK, Thakur P, Gupta M. When palliative treatment achieves more than palliation: instances of long-term survival after palliative radiotherapy. Indian J Palliat Care. 2012;18(2):117-121.
5. Nieder C, Pawinski A, Haukland E, Dokmo R, Phillipi I, Dalhaug A. Estimating need for palliative external beam radiotherapy in adult cancer patients. Int J Radiat Oncol Biol Phys. 2010;76(1):207-211.
6. Hoegler D. Radiotherapy for palliation of symptoms in incurable cancer. Curr Probl Cancer. 1997;21(3):129-183.
7. Expósito J, Jaén J, Alonso E, Tovar I. Use of palliative radiotherapy in brain and bone metastases (VARA II study). Radiat Oncol. 2012;7:131.
8. Konski A. Radiotherapy is a cost-effective palliative treatment for patients with bone metastasis from prostate cancer. Int J Radiat Oncol Biol Phys. 2004;60(5):1373-1378.
9. Popovic M, den Hartogh M, Zhang L, et al. Review of international patterns of practice for the treatment of painful bone metastases with palliative radiotherapy from 1993 to 2013. Radiother Oncol. 2014;111(1):11-17.
10. Lutz S, Berk L, Chang E, et al; American Society for Radiation Oncology (ASTRO). Palliative radiotherapy for bone metastases: an ASTRO evidence-based guideline. Int J Radiat Oncol Biol Phys. 2011;79(4):965-976.
11. Chow E, Harris K, Fan G, Tsao M, Sze WM. Palliative radiotherapy trials for bone metastases: a systemic review. J Clin Oncol. 2007;25(11):1423-1436.
12. Sze WM, Shelley M, Held I, Mason M. Palliation of metastatic bone pain: single fraction versus multifraction radiotherapy—a systemic review of the randomized trials. Cochrane Database Syst Rev. 2004;(2):CD004721.
13. Steenland E, Leer JW, van Houwelingen H, et al. The effect of a single fraction compared to multiple fractions on painful bone metastases: a global analysis of the Dutch Bone Metastasis Study. Radiother Oncol. 1999;52(2):101-109.
14. Krishnan MS, Epstein-Peterson Z, Chen YH, et al. Predicting life expectance in patients with metastatic cancer receiving palliative radiotherapy: the TEACHH model. Cancer. 2014;120(1):134-141.
15. Guadagnolo BA, Liao KP, Elting L, Giordano S, Buccholz TA, Shih YC. Use of radiation therapy in the last 30 days of life among a large population-based cohort of elderly patients in the United States. J Clin Oncol. 2013;31(1):80-87.
16. Moghanaki D, Cheuk AV, Fosmire H, et al; U.S. Veterans Healthcare Administration National Palliative Radiotherapy Taskforce. Availability of single fraction palliative radiotherapy for cancer patients receiving end-of-life care within the Veterans Healthcare Administration. J Palliat Med. 2014;17(11):1221-1225.
17. Ellsworth SG, Alcorn SR, Hales RK, McNutt TR, DeWeese TL, Smith TJ. Patterns of care among patients receiving radiation therapy for bone metastases at a large academic institution. Int J Radiat Oncol Biol Phys. 2014;89(5):1100-1105.
18. Bradley NM, Husted J, Sey MS, et al. Review of patterns of practice and patients’ preferences in the treatment of bone metastases with palliative radiotherapy. Support Care Cancer. 2007;15(4):373-385.
19. Haidukewych GJ. Metastatic disease around the hip: maintaining quality of life. J Bone Joint Surg Br. 2012;94(11 suppl A):22-25.
20. Mirels H. Metastatic disease in long bones. A proposed scoring system for diagnosing impending pathologic fractures. Clin Orthop Relat Res. 1989;(249):256-264.
21. Jacofsky DJ, Haidukewych GJ. Management of pathologic fractures of the proximal femur: state of the art. J Orthop Trauma. 2004;18(7):459-469.
22. Townsend PW, Rosenthal HG, Smalley SR, Cozad SC, Hassanein RE. Impact of postoperative radiation therapy and other perioperative factors on outcome after orthopedic stabilization of impending or pathologic fractures due to metastatic disease. J Clin Oncol. 1994;12(11):2345-2350.
23. Townsend PW, Smalley SR, Cozad SC, Rosenthal HG, Hassanein RE. Role of postoperative radiation therapy after stabilization of fractures caused by metastatic disease. Int J Radiat Oncol Biol Phys. 1995;31(1):43-49.
24. Farooki A. NCCN bone health task force: key recommendations. J Natl Compr Canc Netw. 2014;12(5 suppl):813-816.
25. Sejpal SV, Bhate A, Small W. Palliative radiation therapy in the management of brain metastases, spinal cord compression, and bone metastases. Semin Intervent Radiol. 2007;24(4):362-374.
26. Abrahm JL, Banffy MB, Harris MB. Spinal cord compression in patients with advanced metastatic cancer: “all I care about is walking and living my life.” JAMA. 2008;299(8):937-946.
27. Kim RY, Spencer SA, Meredith RF, et al. Extradural spinal cord compression: analysis of factors determining functional prognosis—prospective study. Radiology. 1990;176(1):279-282.
28. Kaloostian PE, Yurter A, Etame AB, Vrionis FD, Sciubba DM, Gokaslan ZL. Palliative strategies for the management of primary and metastatic spinal tumors. Cancer Control. 2014;21(2):140-143.
29. Graham PH, Capp A, Delaney G, et al. A pilot randomized comparison of dexamethasone 96 mg vs 16 mg per day for malignant spinal-cord compression treated by radiotherapy: TROG 01.05 Superdex study. Clin Oncol (R Coll Radiol). 2006;18(1):70-76.
30. Patchell RA, Tibbs PA, Regine WF, et al. Direct decompressive surgical resection in the treatment of spinal cord compression caused by metastatic cancer: a randomised trial. Lancet. 2005;366(9486):643-648.
31. Rades D, Huttenlocher S, Bajrovic A, et al. Surgery followed by radiotherapy versus radiotherapy alone for metastatic spinal cord compression from unfavorable tumors. Int J Radiat Oncol Biol Phys. 2011;81(5):e861-e868.
32. Maranzano E, Latini P. Effectiveness of radiation therapy without surgery in metastatic spinal cord compression: final results from a prospective trial. Int J Radiat Oncol Biol Phys. 1995;32(4):959-967.
33. Rades D, Fehlauer F, Schulte R, et al. Prognostic factors for local control and survival after radiotherapy of metastatic spinal cord compression. J Clin Oncol. 2006;24(21):3388-3393.
34. Howell DD, James JL, Hartsell WF, et al. Single-fraction radiotherapy versus multifraction radiotherapy for palliation of bone metastases-equivalent efficacy, less toxicity, more convenient: a subset analysis of Radiation Therapy Oncology Group trial 97-14. Cancer. 2013;119(4):888-896.
35. Wong J, Hird A, Kirou-Mauro, Napolskikh J, Chow E. Quality of life in brain metastases radiation trials: a literature review. Curr Oncol. 2008;15(5):25-45.
36. Nichols EM, Patchell RA, Regine WF, Kwok Y. Palliation of brain and spinal cord metastases. In: Halperin EC, Brady LW, Perez CA, Wazer DE, eds. Perez and Brady’s Principles and Practice of Radiation Oncology. 6th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2013:1974.
37. Zimm S, Wampler GL, Stablein D, Hazra T, Young HF. Intracerebral metastases in solid-tumor patients: natural history and results of treatment. Cancer. 1981;48(2):384-394.
38. Khuntia D, Brown P, Li J, Mehta MP. Whole-brain radiotherapy in the management of brain metastasis. J Clin Oncol. 2006;24(8):1295-1304.
39. Sundström JT, Minn H, Lertola KK, Nordman E. Prognosis of patients treated for intracranial metastases with whole-brain irradiation. Ann Med. 1998;30(3):296-299.
40. Tsao MN, Rades D, Wirth A, et al. Radiotherapeutic and surgical management for newly diagnosed brain metastasis(es): an American Society for Radiation Oncology evidence-based guideline. Pract Radiat Oncol. 2012;2(3):210-225.
41. Gaspar L, Scott C, Rotman M, et al. Recursive partitioning analysis (RPA) of prognostic factors in three Radiation Therapy Oncology Group (RTOG) brain metastases trials. Int J Radiat Oncol Biol Phys. 1997;37(4):745-751.
42. Sperduto PW, Berkey B, Gaspar LE, Mehta M, Curran W. A new prognostic index and comparison to three other indices for patients with brain metastases: an analysis of 1,960 patients in the RTOG databases. Int J Radiat Oncol Biol Phys. 2008;70(2):510-514.
43. Sperduto PW, Kased N, Roberge D, et al. Summary report on the graded prognostic assessment: an accurate and facile diagnosis-specific tool to estimate survival for patients with brain metastases. J Clin Oncol. 2012;30(4):419-425.
44. Ryken TC, McDermott M, Robinson PD, et al. The role of steroids in the management of brain metastases: a systematic review and evidence-based clinical practice guideline. J Neurooncol. 2010;96(1):103-114.
45. Ruderman NB, Hall TC. Use of glucocorticoids in the palliative treatment of metastatic brain tumors. Cancer. 1965;18:298-306.
46. Kaloostian PE, Yurter A, Etame AB, Vrionis FD, Sciubba DM, Gokaslan ZL. Palliative strategies for the management of primary and metastatic spinal tumors. Cancer Control. 2014;21(2):140-143.
47. Horton J, Baxter DH, Olson KB. The management of metastases to the brain by irradiation and corticosteroids. Am J Roentgenol Radium Ther Nucl Med. 1971;111(2)334-336.
48. Wong J, Hird A, Zhang L, et al. Symptoms and quality of life in cancer patients with brain metastases following palliative radiotherapy. Int J Radiat Oncol Biol Phys. 2009;75(4):1125-1131.
49. Li J, Bentzen SM, Renschler M, Mehta MP. Regression after whole-brain radiation therapy for brain metastases correlates with survival and improved neurocognitive function. J Clin Oncol. 2007;25(10):1260-1266.
50. Andrews DW, Scott CB, Sperduto PW, et al. Whole brain radiation therapy with or without stereotactic radiosurgery boost for patients with one to three brain metastases: phase III results of the RTOG 9508 randomised trial. Lancet. 2004;363(9422):1665-1672.
51. Patchell RA, Tibbs PA, Walsh JW, et al. A randomized trial of surgery in the treatment of single metastases to the brain. N Engl J Med. 1990;322(8):494-500.
52. Patchell RA, Tibbs PA, Regine WF, et al. Postoperative radiotherapy in the treatment of single metastases to the brain: a randomized trial. JAMA. 1998;280(17):1485-1489.
53. Kocher M, Soffietti R, Abacioglu U, et al. Adjuvant whole-brain radiotherapy versus observation after radiosurgery or surgical resection of one to three cerebral metastases: results of the EORTC 22952-26001 study. J Clin Oncol. 2011;29(2):134-141.
54. Chang EL, Wefel JS, Hess KR, et al. Neurocognition in patients with brain metastases treated with radiosurgery or radiosurgery plus whole-brain irradiation: a randomised controlled trial. Lancet Oncol. 2009;10(11):1037-1044.
In recent years, there has been increasing interest in palliative care for patients with cancer at the end of life. Up to 23% of patients have metastatic disease at presentation, and symptoms from metastatic lesions can cause significant anxiety and impair patients’ quality of life (QOL).1
Palliative radiotherapy (RT) plays a valuable role in the management of metastatic disease to relieve tumor-related symptoms. Although palliative RT does not provide a chance for a cure, it improves QOL and may prolong survival time.2-4 An estimated 20% to 50% of radiation courses are prescribed with palliative intent, because RT is highly effective in providing symptom relief, and the toxicity associated with palliative doses is typically mild.5,6 Palliative RT can be used to manage bone and brain metastases, prevent or treat spinal cord compression, and manage numerous tumor-related symptoms, such as pain and bleeding in patients with terminal cancer.
Palliative RT for bone and brain metastases is supported by high-quality evidence and is considered one of the most effective and cost-effective options available.7,8 This article aims to review the role of RT in treating 3 conditions commonly encountered in patients with metastatic disease—bone metastases, spinal cord compression, and brain metastases—and to emphasize the importance of timely integration of RT for optimal results.
Bone Metastases
About 80% of patients with metastatic solid tumors develop bone metastases, and about 350,000 deaths are linked to bone metastases in the U.S. each year.9 Osseous
metastases can lead to pain, fracture, hypercalcemia, and spinal cord compression. The primary modality for treatment of pain and prevention of morbidity from bone metastases is external beam RT.10
The likelihood of bone pain relief with palliative RT is 60% to 80%, and 30% to 40% of patients achieving complete pain relief. Randomized studies have shown multiple-dose and fractionation regimens provided effective symptom relief for bone metastases. Most commonly used regimens include a single fraction of 8 gray (Gy) delivered in 1 treatment, 20 Gy in 5 fractions delivered daily over 1 week, and 30 Gy in 10 fractions delivered over 2 weeks. Treatment with a single fraction improves access to treatment and patient convenience, whereas more prolonged courses have been associated with lower rates of retreatment.11,12 Regarding the higher rate of retreatment with single-fraction RT, no clear evidence exists that this is due to a less durable pain response or lower level of pain relief.13
There has been recent interest in using predictive models to estimate life expectancy to avoid long courses of RT at the end of life.14,15 Shorter treatment courses of 8 Gyonce or 20 Gy in 5 fractions are particularly valuable for patients with a life expectancy < 3 months to avoid long courses of treatment, and thereby improve QOL as patients transition into hospice. A recent survey demonstrated that 93% of radiation oncologists within the VHA are willing to prescribe short courses of RT consisting of ≤ 6 fractions, and 76% have experience with single-fraction RT.16 These findings are in contradiction to the findings in the non-VA radiation oncology community, in which < 10% of patients with uncomplicated bone metastases are treated with a single fraction.17,18
In addition to providing pain relief, RT is used in the treatment of impending fractures either, adjuvant after surgical stabilization or alone for lower risk lesions.19 Factors that impact fracture risk include location of the metastasis (weight-bearing bones, such as femurs, which are at particularly high risk), length of bone involved, and extent of cortical involvement. Mirels’ scoring system was developed to predict fracture risk in patients with bone metastasis, based on 4 criteria: the
extent of cortical involvement, the location of the metastasis, the osteolytic vs osteoblastic appearance of the lesion, and the degree of pain.20 Surgical fixation can be considered, based on the total score and corresponding fracture risk. When appropriate, surgical stabilization should be considered by an orthopedic surgeon prior to initiating RT.
Postoperative RT after surgical stabilization has been associated with a reduced rate of secondary surgical procedures as well as with improved functional status.21 Radiotherapy promotes remineralization and bone healing and prevents the loss of surgical fixation by treating any residual tumor. A retrospective review of 60 patients with metastatic disease in weight-bearing bones with pathologic fracture or impending pathologic fracture demonstrated that surgery followed by RT was associated with improved functional status as well as with improved overall survival (OS).22,23 For patients in whom surgery is not indicated, the consulting radiation oncologist should consider factors such as the location of the metastasis in weight-bearing vs nonweight bearing bones, the size and extent of the metastasis, and associated symptoms when making a treatment recommendation. In patients at fracture risk from bone metastases, bisphosphonates should also be considered as part of the treatment regimen.24
Spinal Cord Compression
About 5% to 10% of patients diagnosed with cancer will develop spinal cord compression during the course of their disease.25 Spinal cord compression is considered a medical emergency that can result in significant pain and neurologic symptoms, including weakness, paralysis, parasthesias, and incontinence. Early treatment of spinal cord compression can prevent onset or progression of these symptoms; furthermore, early treatment prior to loss of ambulation is associated with improved long-term ambulatory function.26,27
Treatment decisions for spinal metastases with an associated concern for cord compression should be made after a consultation with both a neurosurgeon and a radiation oncologist. Early initiation of steroids is recommended to aid in tumor shrinkage for potential symptom relief.28 A standard way to administer dexamethasone is with a 10-mg loading dose followed by 16 mg per day, divided into 4 doses of 4 mg. Higher steroid doses showed no benefit in a prospective randomized trial comparing 96 mg with 16 mg of dexamethasone daily.29
Surgical decompression should be considered initial management of spinal cord compression. For patients treated surgically, local RT is indicated postoperatively as well. Randomized data show that surgery followed by RT provides better ambulatory function than does RT alone in patients with paralysis of < 2 days’ duration.30 Some patients with metastatic disease are not good candidates for surgery due to comorbidities, poor performance status, life expectancy < 3 months, or multilevel spinal involvement.
In patients who are not operative candidates, radiation alone is an appropriate alternative. However, several factors need consideration in deciding whether to manage cord compression with surgery followed by RT vs RT alone. These factors include life expectancy, tumor type (myeloma and lymphoma are more radiosensitive), interval since tumor diagnosis, and the presence of visceral metastases.31 Factors favoring surgical decompression plus postoperative RT over RT alone include spinal instability, KPS (Karnofsky Performance Status) > 70, radio-resistant tumor histology, minimal metastatic disease, and projected survival > 3 months.10
For patients managed with RT alone, early diagnosis and treatment is associated with improved outcomes. A prospective study of patients treated with RT without surgery for spinal cord compression demonstrated that 82% of patients experienced back pain relief, 76% achieved improvement in or preservation of ambulation, and 44% of patients with sphincter dysfunction experienced improvement with treatment.32 Patients with certain tumor histologies, such as myeloma, breast cancer, and prostate cancer, had better responses to RT.32
In the setting of spinal cord compression, longer courses of RT may provide better local control than do shorter courses.33 Therefore, longer courses of RT, such as 30 Gy in 10 fractions delivered over 2 weeks, are often preferred in cases of spinal cord compression treated with definitive RT as well as after surgical decompression. However, overall life expectancy is an important factor considered by the treating radiation oncologist when selecting a short course vs a longer course of RT.
In the instance of painful vertebral body metastases without spinal cord compression, a new subset analysis of the Radiation Therapy Oncology Group (RTOG) 9714 randomized trial indicated that single fraction RT (8 Gy) is just as effective as multiple fractions (30 Gy in 10 fractions), with this study demonstrating comparable rates of pain relief and narcotic use in both groups 3 months after RT.34 Advantages to the single-fraction plan compared with those of multiple fractions include mitigation of logistic concerns for patients and family at the end of life and less acute adverse effects.
Brain Metastases
An estimated 20% to 40% of patients with cancer develop brain metastases.35 The incidence of brain metastases has been rising most likely due to improved detection rates with magnetic resonance imaging (MRI) and improved cancer survival, because treatment regimens have improved with targeted chemotherapy and radiation techniques. Currently, the annual incidence of brain metastases is 170,000 to 200,000 in the U.S.36 Prognosis for these patients is poor, with median survival of 1 month without treatment and about 4 months with whole brain RT (WBRT) (Table).25,37-39
The goal of management for patients with brain metastases is to prevent or treat neurologic symptoms and to prolong survival. Treatment options include corticosteroids, WBRT, surgery, and stereotactic radiosurgery (SRS). Recommendations for treatment should involve both a radiation oncologist and neurosurgeon to determine the best treatment for an individual based on patient age, performance status, extent of systemic disease, and number of brain metastases. These prognostic factors that may predict life expectancy and impact treatment recommendations.40
Factors that have been correlated with improved survival include younger age, better performance status, fewer brain metastases, and lower burden of systemic disease.41,42 Prognostic assessment tools such as the Graded Prognostic Assessment and RTOG-Recursive Partitioning Analysis can be used to predict life expectancy in patients with brain metastases.41,43 However, routine use of these tools is lagging, as evidenced by a recent survey of VHA radiation oncologists. Use of these tools in the clinic will enhance the quality of end of life care and decision making.
Corticosteroids have classically been used in the treatment of brain metastases either alone for supportive care or in combination with RT. Steroids are recommended to provide symptom relief in patients with symptoms related to cerebral edema or mass effect.44 Steroids have been shown to mitigate edema and improve neurologic deficits in about two-thirds of patients with brain metastases.36,45 The effect of corticosteroids is thought to be mediated through inhibition of prostaglandin synthesis, reduction in vascular permeability, and anti-inflammatory properties.46 A common corticosteroid regimen is a 10-mg loading dose of dexamethasone, followed by 16 mg daily in divided doses. For patients without neurologic deficits or cerebral edema, it is reasonable to defer corticosteroid use only when patients are symptomatic.
In general, WBRT is considered an appropriate treatment option for patients with multiple brain metastases based on data suggesting an improvement in OS compared with the use of corticosteroids alone.47 Whole brain radiation has been shown to result in the improvement of baseline neurologic deficits or the prevention of further symptom progression.48 The partial or complete metastasis response rates are on the order of 60%.38 Tumor regression after WBRT has been associated with preservation of neurocognitive function as well as prolonged survival.49
For good prognosis patients with a single brain metastasis and good performance status, the use of surgery or radiosurgery added to WBRT has been associated with improved OS (Table). The RTOG 9508 randomized trial of WBRT with or without SRS demonstrated a survival advantage with SRS, with median survival times of 6.5 months with WBRT + SRS vs 4.9 months with WBRT alone.50 Similarly, a randomized trial evaluating WBRT alone compared with surgery followed by WBRT in patients
with good prognosis demonstrated significantly improved OS in the surgery group (median 40 weeks vs 15 weeks).51 In general, WBRT or postoperative RT to the tumor bed is still indicated after surgical resection, based on randomized data showing a reduction in tumor bed recurrence with postoperative RT.52
For patients with only 1 to 3 brain metastases and a favorable prognosis, surgery and SRS can be considered treatment options, oftentimes with WBRT. The EORTC randomized trial of patients with 1 to 3 brain metastases was designed to determine the benefit of WBRT after treatment with surgery or SRS. In this study, 119 patients underwent SRS and 160 patients underwent surgical resection.53 Both groups of patients were randomized to observation vs adjuvant WBRT. This study demonstrated reduced rates of intracranial relapse with WBRT, however, without any change in OS. Although there is concern that WBRT may impair cognitive function with no clear survival benefit after surgery or SRS, WBRT does reduce recurrence rates in the brain and the need for further treatment.54 Therefore, decisions regarding WBRT in such a setting should be made only after a detailed discussion with a radiation oncologist regarding risks vs benefits of treatment as part of the informed decision-making process.
Conclusions
Palliative RT plays an important role in the management of metastatic cancer to provide symptom relief and is a cost-effective treatment option for bone and brain metastases. Life expectancy and tumor characteristics should be considered when making treatment recommendations to ensure selection of regimens that complement patients’ unique situations. Timely referrals for treatment are important to optimize treatment results.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Click here to read the digital edition.
In recent years, there has been increasing interest in palliative care for patients with cancer at the end of life. Up to 23% of patients have metastatic disease at presentation, and symptoms from metastatic lesions can cause significant anxiety and impair patients’ quality of life (QOL).1
Palliative radiotherapy (RT) plays a valuable role in the management of metastatic disease to relieve tumor-related symptoms. Although palliative RT does not provide a chance for a cure, it improves QOL and may prolong survival time.2-4 An estimated 20% to 50% of radiation courses are prescribed with palliative intent, because RT is highly effective in providing symptom relief, and the toxicity associated with palliative doses is typically mild.5,6 Palliative RT can be used to manage bone and brain metastases, prevent or treat spinal cord compression, and manage numerous tumor-related symptoms, such as pain and bleeding in patients with terminal cancer.
Palliative RT for bone and brain metastases is supported by high-quality evidence and is considered one of the most effective and cost-effective options available.7,8 This article aims to review the role of RT in treating 3 conditions commonly encountered in patients with metastatic disease—bone metastases, spinal cord compression, and brain metastases—and to emphasize the importance of timely integration of RT for optimal results.
Bone Metastases
About 80% of patients with metastatic solid tumors develop bone metastases, and about 350,000 deaths are linked to bone metastases in the U.S. each year.9 Osseous
metastases can lead to pain, fracture, hypercalcemia, and spinal cord compression. The primary modality for treatment of pain and prevention of morbidity from bone metastases is external beam RT.10
The likelihood of bone pain relief with palliative RT is 60% to 80%, and 30% to 40% of patients achieving complete pain relief. Randomized studies have shown multiple-dose and fractionation regimens provided effective symptom relief for bone metastases. Most commonly used regimens include a single fraction of 8 gray (Gy) delivered in 1 treatment, 20 Gy in 5 fractions delivered daily over 1 week, and 30 Gy in 10 fractions delivered over 2 weeks. Treatment with a single fraction improves access to treatment and patient convenience, whereas more prolonged courses have been associated with lower rates of retreatment.11,12 Regarding the higher rate of retreatment with single-fraction RT, no clear evidence exists that this is due to a less durable pain response or lower level of pain relief.13
There has been recent interest in using predictive models to estimate life expectancy to avoid long courses of RT at the end of life.14,15 Shorter treatment courses of 8 Gyonce or 20 Gy in 5 fractions are particularly valuable for patients with a life expectancy < 3 months to avoid long courses of treatment, and thereby improve QOL as patients transition into hospice. A recent survey demonstrated that 93% of radiation oncologists within the VHA are willing to prescribe short courses of RT consisting of ≤ 6 fractions, and 76% have experience with single-fraction RT.16 These findings are in contradiction to the findings in the non-VA radiation oncology community, in which < 10% of patients with uncomplicated bone metastases are treated with a single fraction.17,18
In addition to providing pain relief, RT is used in the treatment of impending fractures either, adjuvant after surgical stabilization or alone for lower risk lesions.19 Factors that impact fracture risk include location of the metastasis (weight-bearing bones, such as femurs, which are at particularly high risk), length of bone involved, and extent of cortical involvement. Mirels’ scoring system was developed to predict fracture risk in patients with bone metastasis, based on 4 criteria: the
extent of cortical involvement, the location of the metastasis, the osteolytic vs osteoblastic appearance of the lesion, and the degree of pain.20 Surgical fixation can be considered, based on the total score and corresponding fracture risk. When appropriate, surgical stabilization should be considered by an orthopedic surgeon prior to initiating RT.
Postoperative RT after surgical stabilization has been associated with a reduced rate of secondary surgical procedures as well as with improved functional status.21 Radiotherapy promotes remineralization and bone healing and prevents the loss of surgical fixation by treating any residual tumor. A retrospective review of 60 patients with metastatic disease in weight-bearing bones with pathologic fracture or impending pathologic fracture demonstrated that surgery followed by RT was associated with improved functional status as well as with improved overall survival (OS).22,23 For patients in whom surgery is not indicated, the consulting radiation oncologist should consider factors such as the location of the metastasis in weight-bearing vs nonweight bearing bones, the size and extent of the metastasis, and associated symptoms when making a treatment recommendation. In patients at fracture risk from bone metastases, bisphosphonates should also be considered as part of the treatment regimen.24
Spinal Cord Compression
About 5% to 10% of patients diagnosed with cancer will develop spinal cord compression during the course of their disease.25 Spinal cord compression is considered a medical emergency that can result in significant pain and neurologic symptoms, including weakness, paralysis, parasthesias, and incontinence. Early treatment of spinal cord compression can prevent onset or progression of these symptoms; furthermore, early treatment prior to loss of ambulation is associated with improved long-term ambulatory function.26,27
Treatment decisions for spinal metastases with an associated concern for cord compression should be made after a consultation with both a neurosurgeon and a radiation oncologist. Early initiation of steroids is recommended to aid in tumor shrinkage for potential symptom relief.28 A standard way to administer dexamethasone is with a 10-mg loading dose followed by 16 mg per day, divided into 4 doses of 4 mg. Higher steroid doses showed no benefit in a prospective randomized trial comparing 96 mg with 16 mg of dexamethasone daily.29
Surgical decompression should be considered initial management of spinal cord compression. For patients treated surgically, local RT is indicated postoperatively as well. Randomized data show that surgery followed by RT provides better ambulatory function than does RT alone in patients with paralysis of < 2 days’ duration.30 Some patients with metastatic disease are not good candidates for surgery due to comorbidities, poor performance status, life expectancy < 3 months, or multilevel spinal involvement.
In patients who are not operative candidates, radiation alone is an appropriate alternative. However, several factors need consideration in deciding whether to manage cord compression with surgery followed by RT vs RT alone. These factors include life expectancy, tumor type (myeloma and lymphoma are more radiosensitive), interval since tumor diagnosis, and the presence of visceral metastases.31 Factors favoring surgical decompression plus postoperative RT over RT alone include spinal instability, KPS (Karnofsky Performance Status) > 70, radio-resistant tumor histology, minimal metastatic disease, and projected survival > 3 months.10
For patients managed with RT alone, early diagnosis and treatment is associated with improved outcomes. A prospective study of patients treated with RT without surgery for spinal cord compression demonstrated that 82% of patients experienced back pain relief, 76% achieved improvement in or preservation of ambulation, and 44% of patients with sphincter dysfunction experienced improvement with treatment.32 Patients with certain tumor histologies, such as myeloma, breast cancer, and prostate cancer, had better responses to RT.32
In the setting of spinal cord compression, longer courses of RT may provide better local control than do shorter courses.33 Therefore, longer courses of RT, such as 30 Gy in 10 fractions delivered over 2 weeks, are often preferred in cases of spinal cord compression treated with definitive RT as well as after surgical decompression. However, overall life expectancy is an important factor considered by the treating radiation oncologist when selecting a short course vs a longer course of RT.
In the instance of painful vertebral body metastases without spinal cord compression, a new subset analysis of the Radiation Therapy Oncology Group (RTOG) 9714 randomized trial indicated that single fraction RT (8 Gy) is just as effective as multiple fractions (30 Gy in 10 fractions), with this study demonstrating comparable rates of pain relief and narcotic use in both groups 3 months after RT.34 Advantages to the single-fraction plan compared with those of multiple fractions include mitigation of logistic concerns for patients and family at the end of life and less acute adverse effects.
Brain Metastases
An estimated 20% to 40% of patients with cancer develop brain metastases.35 The incidence of brain metastases has been rising most likely due to improved detection rates with magnetic resonance imaging (MRI) and improved cancer survival, because treatment regimens have improved with targeted chemotherapy and radiation techniques. Currently, the annual incidence of brain metastases is 170,000 to 200,000 in the U.S.36 Prognosis for these patients is poor, with median survival of 1 month without treatment and about 4 months with whole brain RT (WBRT) (Table).25,37-39
The goal of management for patients with brain metastases is to prevent or treat neurologic symptoms and to prolong survival. Treatment options include corticosteroids, WBRT, surgery, and stereotactic radiosurgery (SRS). Recommendations for treatment should involve both a radiation oncologist and neurosurgeon to determine the best treatment for an individual based on patient age, performance status, extent of systemic disease, and number of brain metastases. These prognostic factors that may predict life expectancy and impact treatment recommendations.40
Factors that have been correlated with improved survival include younger age, better performance status, fewer brain metastases, and lower burden of systemic disease.41,42 Prognostic assessment tools such as the Graded Prognostic Assessment and RTOG-Recursive Partitioning Analysis can be used to predict life expectancy in patients with brain metastases.41,43 However, routine use of these tools is lagging, as evidenced by a recent survey of VHA radiation oncologists. Use of these tools in the clinic will enhance the quality of end of life care and decision making.
Corticosteroids have classically been used in the treatment of brain metastases either alone for supportive care or in combination with RT. Steroids are recommended to provide symptom relief in patients with symptoms related to cerebral edema or mass effect.44 Steroids have been shown to mitigate edema and improve neurologic deficits in about two-thirds of patients with brain metastases.36,45 The effect of corticosteroids is thought to be mediated through inhibition of prostaglandin synthesis, reduction in vascular permeability, and anti-inflammatory properties.46 A common corticosteroid regimen is a 10-mg loading dose of dexamethasone, followed by 16 mg daily in divided doses. For patients without neurologic deficits or cerebral edema, it is reasonable to defer corticosteroid use only when patients are symptomatic.
In general, WBRT is considered an appropriate treatment option for patients with multiple brain metastases based on data suggesting an improvement in OS compared with the use of corticosteroids alone.47 Whole brain radiation has been shown to result in the improvement of baseline neurologic deficits or the prevention of further symptom progression.48 The partial or complete metastasis response rates are on the order of 60%.38 Tumor regression after WBRT has been associated with preservation of neurocognitive function as well as prolonged survival.49
For good prognosis patients with a single brain metastasis and good performance status, the use of surgery or radiosurgery added to WBRT has been associated with improved OS (Table). The RTOG 9508 randomized trial of WBRT with or without SRS demonstrated a survival advantage with SRS, with median survival times of 6.5 months with WBRT + SRS vs 4.9 months with WBRT alone.50 Similarly, a randomized trial evaluating WBRT alone compared with surgery followed by WBRT in patients
with good prognosis demonstrated significantly improved OS in the surgery group (median 40 weeks vs 15 weeks).51 In general, WBRT or postoperative RT to the tumor bed is still indicated after surgical resection, based on randomized data showing a reduction in tumor bed recurrence with postoperative RT.52
For patients with only 1 to 3 brain metastases and a favorable prognosis, surgery and SRS can be considered treatment options, oftentimes with WBRT. The EORTC randomized trial of patients with 1 to 3 brain metastases was designed to determine the benefit of WBRT after treatment with surgery or SRS. In this study, 119 patients underwent SRS and 160 patients underwent surgical resection.53 Both groups of patients were randomized to observation vs adjuvant WBRT. This study demonstrated reduced rates of intracranial relapse with WBRT, however, without any change in OS. Although there is concern that WBRT may impair cognitive function with no clear survival benefit after surgery or SRS, WBRT does reduce recurrence rates in the brain and the need for further treatment.54 Therefore, decisions regarding WBRT in such a setting should be made only after a detailed discussion with a radiation oncologist regarding risks vs benefits of treatment as part of the informed decision-making process.
Conclusions
Palliative RT plays an important role in the management of metastatic cancer to provide symptom relief and is a cost-effective treatment option for bone and brain metastases. Life expectancy and tumor characteristics should be considered when making treatment recommendations to ensure selection of regimens that complement patients’ unique situations. Timely referrals for treatment are important to optimize treatment results.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Click here to read the digital edition.
1. Porter A and David M. Palliative care for bone, spinal cord, brain, and liver metastases. In: Gunderson LL, Tepper JE, eds. Clinical Radiation Oncology. 2nd ed. Philadelphia, PA: Elsevier Churchill Livingstone; 2007:437-451.
2. Yamaguchi S, Ohguri T, Matsuki Y, et al. Palliative radiotherapy in patients with a poor performance status: the palliative effect is correlated with prolongation of survival time. Radiat Oncol. 2013;8:166.
3. Mac Manus MP, Matthews JP, Wada M, Wirth A, Worotniuk V, Ball DL. Unexpected long-term survival after low-dose palliative radiotherapy for non-small cell lung cancer. Cancer. 2006;106(5):1110-1116.
4. Rastogi M, Revannasiddaiah S, Gupta MK, Seam RK, Thakur P, Gupta M. When palliative treatment achieves more than palliation: instances of long-term survival after palliative radiotherapy. Indian J Palliat Care. 2012;18(2):117-121.
5. Nieder C, Pawinski A, Haukland E, Dokmo R, Phillipi I, Dalhaug A. Estimating need for palliative external beam radiotherapy in adult cancer patients. Int J Radiat Oncol Biol Phys. 2010;76(1):207-211.
6. Hoegler D. Radiotherapy for palliation of symptoms in incurable cancer. Curr Probl Cancer. 1997;21(3):129-183.
7. Expósito J, Jaén J, Alonso E, Tovar I. Use of palliative radiotherapy in brain and bone metastases (VARA II study). Radiat Oncol. 2012;7:131.
8. Konski A. Radiotherapy is a cost-effective palliative treatment for patients with bone metastasis from prostate cancer. Int J Radiat Oncol Biol Phys. 2004;60(5):1373-1378.
9. Popovic M, den Hartogh M, Zhang L, et al. Review of international patterns of practice for the treatment of painful bone metastases with palliative radiotherapy from 1993 to 2013. Radiother Oncol. 2014;111(1):11-17.
10. Lutz S, Berk L, Chang E, et al; American Society for Radiation Oncology (ASTRO). Palliative radiotherapy for bone metastases: an ASTRO evidence-based guideline. Int J Radiat Oncol Biol Phys. 2011;79(4):965-976.
11. Chow E, Harris K, Fan G, Tsao M, Sze WM. Palliative radiotherapy trials for bone metastases: a systemic review. J Clin Oncol. 2007;25(11):1423-1436.
12. Sze WM, Shelley M, Held I, Mason M. Palliation of metastatic bone pain: single fraction versus multifraction radiotherapy—a systemic review of the randomized trials. Cochrane Database Syst Rev. 2004;(2):CD004721.
13. Steenland E, Leer JW, van Houwelingen H, et al. The effect of a single fraction compared to multiple fractions on painful bone metastases: a global analysis of the Dutch Bone Metastasis Study. Radiother Oncol. 1999;52(2):101-109.
14. Krishnan MS, Epstein-Peterson Z, Chen YH, et al. Predicting life expectance in patients with metastatic cancer receiving palliative radiotherapy: the TEACHH model. Cancer. 2014;120(1):134-141.
15. Guadagnolo BA, Liao KP, Elting L, Giordano S, Buccholz TA, Shih YC. Use of radiation therapy in the last 30 days of life among a large population-based cohort of elderly patients in the United States. J Clin Oncol. 2013;31(1):80-87.
16. Moghanaki D, Cheuk AV, Fosmire H, et al; U.S. Veterans Healthcare Administration National Palliative Radiotherapy Taskforce. Availability of single fraction palliative radiotherapy for cancer patients receiving end-of-life care within the Veterans Healthcare Administration. J Palliat Med. 2014;17(11):1221-1225.
17. Ellsworth SG, Alcorn SR, Hales RK, McNutt TR, DeWeese TL, Smith TJ. Patterns of care among patients receiving radiation therapy for bone metastases at a large academic institution. Int J Radiat Oncol Biol Phys. 2014;89(5):1100-1105.
18. Bradley NM, Husted J, Sey MS, et al. Review of patterns of practice and patients’ preferences in the treatment of bone metastases with palliative radiotherapy. Support Care Cancer. 2007;15(4):373-385.
19. Haidukewych GJ. Metastatic disease around the hip: maintaining quality of life. J Bone Joint Surg Br. 2012;94(11 suppl A):22-25.
20. Mirels H. Metastatic disease in long bones. A proposed scoring system for diagnosing impending pathologic fractures. Clin Orthop Relat Res. 1989;(249):256-264.
21. Jacofsky DJ, Haidukewych GJ. Management of pathologic fractures of the proximal femur: state of the art. J Orthop Trauma. 2004;18(7):459-469.
22. Townsend PW, Rosenthal HG, Smalley SR, Cozad SC, Hassanein RE. Impact of postoperative radiation therapy and other perioperative factors on outcome after orthopedic stabilization of impending or pathologic fractures due to metastatic disease. J Clin Oncol. 1994;12(11):2345-2350.
23. Townsend PW, Smalley SR, Cozad SC, Rosenthal HG, Hassanein RE. Role of postoperative radiation therapy after stabilization of fractures caused by metastatic disease. Int J Radiat Oncol Biol Phys. 1995;31(1):43-49.
24. Farooki A. NCCN bone health task force: key recommendations. J Natl Compr Canc Netw. 2014;12(5 suppl):813-816.
25. Sejpal SV, Bhate A, Small W. Palliative radiation therapy in the management of brain metastases, spinal cord compression, and bone metastases. Semin Intervent Radiol. 2007;24(4):362-374.
26. Abrahm JL, Banffy MB, Harris MB. Spinal cord compression in patients with advanced metastatic cancer: “all I care about is walking and living my life.” JAMA. 2008;299(8):937-946.
27. Kim RY, Spencer SA, Meredith RF, et al. Extradural spinal cord compression: analysis of factors determining functional prognosis—prospective study. Radiology. 1990;176(1):279-282.
28. Kaloostian PE, Yurter A, Etame AB, Vrionis FD, Sciubba DM, Gokaslan ZL. Palliative strategies for the management of primary and metastatic spinal tumors. Cancer Control. 2014;21(2):140-143.
29. Graham PH, Capp A, Delaney G, et al. A pilot randomized comparison of dexamethasone 96 mg vs 16 mg per day for malignant spinal-cord compression treated by radiotherapy: TROG 01.05 Superdex study. Clin Oncol (R Coll Radiol). 2006;18(1):70-76.
30. Patchell RA, Tibbs PA, Regine WF, et al. Direct decompressive surgical resection in the treatment of spinal cord compression caused by metastatic cancer: a randomised trial. Lancet. 2005;366(9486):643-648.
31. Rades D, Huttenlocher S, Bajrovic A, et al. Surgery followed by radiotherapy versus radiotherapy alone for metastatic spinal cord compression from unfavorable tumors. Int J Radiat Oncol Biol Phys. 2011;81(5):e861-e868.
32. Maranzano E, Latini P. Effectiveness of radiation therapy without surgery in metastatic spinal cord compression: final results from a prospective trial. Int J Radiat Oncol Biol Phys. 1995;32(4):959-967.
33. Rades D, Fehlauer F, Schulte R, et al. Prognostic factors for local control and survival after radiotherapy of metastatic spinal cord compression. J Clin Oncol. 2006;24(21):3388-3393.
34. Howell DD, James JL, Hartsell WF, et al. Single-fraction radiotherapy versus multifraction radiotherapy for palliation of bone metastases-equivalent efficacy, less toxicity, more convenient: a subset analysis of Radiation Therapy Oncology Group trial 97-14. Cancer. 2013;119(4):888-896.
35. Wong J, Hird A, Kirou-Mauro, Napolskikh J, Chow E. Quality of life in brain metastases radiation trials: a literature review. Curr Oncol. 2008;15(5):25-45.
36. Nichols EM, Patchell RA, Regine WF, Kwok Y. Palliation of brain and spinal cord metastases. In: Halperin EC, Brady LW, Perez CA, Wazer DE, eds. Perez and Brady’s Principles and Practice of Radiation Oncology. 6th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2013:1974.
37. Zimm S, Wampler GL, Stablein D, Hazra T, Young HF. Intracerebral metastases in solid-tumor patients: natural history and results of treatment. Cancer. 1981;48(2):384-394.
38. Khuntia D, Brown P, Li J, Mehta MP. Whole-brain radiotherapy in the management of brain metastasis. J Clin Oncol. 2006;24(8):1295-1304.
39. Sundström JT, Minn H, Lertola KK, Nordman E. Prognosis of patients treated for intracranial metastases with whole-brain irradiation. Ann Med. 1998;30(3):296-299.
40. Tsao MN, Rades D, Wirth A, et al. Radiotherapeutic and surgical management for newly diagnosed brain metastasis(es): an American Society for Radiation Oncology evidence-based guideline. Pract Radiat Oncol. 2012;2(3):210-225.
41. Gaspar L, Scott C, Rotman M, et al. Recursive partitioning analysis (RPA) of prognostic factors in three Radiation Therapy Oncology Group (RTOG) brain metastases trials. Int J Radiat Oncol Biol Phys. 1997;37(4):745-751.
42. Sperduto PW, Berkey B, Gaspar LE, Mehta M, Curran W. A new prognostic index and comparison to three other indices for patients with brain metastases: an analysis of 1,960 patients in the RTOG databases. Int J Radiat Oncol Biol Phys. 2008;70(2):510-514.
43. Sperduto PW, Kased N, Roberge D, et al. Summary report on the graded prognostic assessment: an accurate and facile diagnosis-specific tool to estimate survival for patients with brain metastases. J Clin Oncol. 2012;30(4):419-425.
44. Ryken TC, McDermott M, Robinson PD, et al. The role of steroids in the management of brain metastases: a systematic review and evidence-based clinical practice guideline. J Neurooncol. 2010;96(1):103-114.
45. Ruderman NB, Hall TC. Use of glucocorticoids in the palliative treatment of metastatic brain tumors. Cancer. 1965;18:298-306.
46. Kaloostian PE, Yurter A, Etame AB, Vrionis FD, Sciubba DM, Gokaslan ZL. Palliative strategies for the management of primary and metastatic spinal tumors. Cancer Control. 2014;21(2):140-143.
47. Horton J, Baxter DH, Olson KB. The management of metastases to the brain by irradiation and corticosteroids. Am J Roentgenol Radium Ther Nucl Med. 1971;111(2)334-336.
48. Wong J, Hird A, Zhang L, et al. Symptoms and quality of life in cancer patients with brain metastases following palliative radiotherapy. Int J Radiat Oncol Biol Phys. 2009;75(4):1125-1131.
49. Li J, Bentzen SM, Renschler M, Mehta MP. Regression after whole-brain radiation therapy for brain metastases correlates with survival and improved neurocognitive function. J Clin Oncol. 2007;25(10):1260-1266.
50. Andrews DW, Scott CB, Sperduto PW, et al. Whole brain radiation therapy with or without stereotactic radiosurgery boost for patients with one to three brain metastases: phase III results of the RTOG 9508 randomised trial. Lancet. 2004;363(9422):1665-1672.
51. Patchell RA, Tibbs PA, Walsh JW, et al. A randomized trial of surgery in the treatment of single metastases to the brain. N Engl J Med. 1990;322(8):494-500.
52. Patchell RA, Tibbs PA, Regine WF, et al. Postoperative radiotherapy in the treatment of single metastases to the brain: a randomized trial. JAMA. 1998;280(17):1485-1489.
53. Kocher M, Soffietti R, Abacioglu U, et al. Adjuvant whole-brain radiotherapy versus observation after radiosurgery or surgical resection of one to three cerebral metastases: results of the EORTC 22952-26001 study. J Clin Oncol. 2011;29(2):134-141.
54. Chang EL, Wefel JS, Hess KR, et al. Neurocognition in patients with brain metastases treated with radiosurgery or radiosurgery plus whole-brain irradiation: a randomised controlled trial. Lancet Oncol. 2009;10(11):1037-1044.
1. Porter A and David M. Palliative care for bone, spinal cord, brain, and liver metastases. In: Gunderson LL, Tepper JE, eds. Clinical Radiation Oncology. 2nd ed. Philadelphia, PA: Elsevier Churchill Livingstone; 2007:437-451.
2. Yamaguchi S, Ohguri T, Matsuki Y, et al. Palliative radiotherapy in patients with a poor performance status: the palliative effect is correlated with prolongation of survival time. Radiat Oncol. 2013;8:166.
3. Mac Manus MP, Matthews JP, Wada M, Wirth A, Worotniuk V, Ball DL. Unexpected long-term survival after low-dose palliative radiotherapy for non-small cell lung cancer. Cancer. 2006;106(5):1110-1116.
4. Rastogi M, Revannasiddaiah S, Gupta MK, Seam RK, Thakur P, Gupta M. When palliative treatment achieves more than palliation: instances of long-term survival after palliative radiotherapy. Indian J Palliat Care. 2012;18(2):117-121.
5. Nieder C, Pawinski A, Haukland E, Dokmo R, Phillipi I, Dalhaug A. Estimating need for palliative external beam radiotherapy in adult cancer patients. Int J Radiat Oncol Biol Phys. 2010;76(1):207-211.
6. Hoegler D. Radiotherapy for palliation of symptoms in incurable cancer. Curr Probl Cancer. 1997;21(3):129-183.
7. Expósito J, Jaén J, Alonso E, Tovar I. Use of palliative radiotherapy in brain and bone metastases (VARA II study). Radiat Oncol. 2012;7:131.
8. Konski A. Radiotherapy is a cost-effective palliative treatment for patients with bone metastasis from prostate cancer. Int J Radiat Oncol Biol Phys. 2004;60(5):1373-1378.
9. Popovic M, den Hartogh M, Zhang L, et al. Review of international patterns of practice for the treatment of painful bone metastases with palliative radiotherapy from 1993 to 2013. Radiother Oncol. 2014;111(1):11-17.
10. Lutz S, Berk L, Chang E, et al; American Society for Radiation Oncology (ASTRO). Palliative radiotherapy for bone metastases: an ASTRO evidence-based guideline. Int J Radiat Oncol Biol Phys. 2011;79(4):965-976.
11. Chow E, Harris K, Fan G, Tsao M, Sze WM. Palliative radiotherapy trials for bone metastases: a systemic review. J Clin Oncol. 2007;25(11):1423-1436.
12. Sze WM, Shelley M, Held I, Mason M. Palliation of metastatic bone pain: single fraction versus multifraction radiotherapy—a systemic review of the randomized trials. Cochrane Database Syst Rev. 2004;(2):CD004721.
13. Steenland E, Leer JW, van Houwelingen H, et al. The effect of a single fraction compared to multiple fractions on painful bone metastases: a global analysis of the Dutch Bone Metastasis Study. Radiother Oncol. 1999;52(2):101-109.
14. Krishnan MS, Epstein-Peterson Z, Chen YH, et al. Predicting life expectance in patients with metastatic cancer receiving palliative radiotherapy: the TEACHH model. Cancer. 2014;120(1):134-141.
15. Guadagnolo BA, Liao KP, Elting L, Giordano S, Buccholz TA, Shih YC. Use of radiation therapy in the last 30 days of life among a large population-based cohort of elderly patients in the United States. J Clin Oncol. 2013;31(1):80-87.
16. Moghanaki D, Cheuk AV, Fosmire H, et al; U.S. Veterans Healthcare Administration National Palliative Radiotherapy Taskforce. Availability of single fraction palliative radiotherapy for cancer patients receiving end-of-life care within the Veterans Healthcare Administration. J Palliat Med. 2014;17(11):1221-1225.
17. Ellsworth SG, Alcorn SR, Hales RK, McNutt TR, DeWeese TL, Smith TJ. Patterns of care among patients receiving radiation therapy for bone metastases at a large academic institution. Int J Radiat Oncol Biol Phys. 2014;89(5):1100-1105.
18. Bradley NM, Husted J, Sey MS, et al. Review of patterns of practice and patients’ preferences in the treatment of bone metastases with palliative radiotherapy. Support Care Cancer. 2007;15(4):373-385.
19. Haidukewych GJ. Metastatic disease around the hip: maintaining quality of life. J Bone Joint Surg Br. 2012;94(11 suppl A):22-25.
20. Mirels H. Metastatic disease in long bones. A proposed scoring system for diagnosing impending pathologic fractures. Clin Orthop Relat Res. 1989;(249):256-264.
21. Jacofsky DJ, Haidukewych GJ. Management of pathologic fractures of the proximal femur: state of the art. J Orthop Trauma. 2004;18(7):459-469.
22. Townsend PW, Rosenthal HG, Smalley SR, Cozad SC, Hassanein RE. Impact of postoperative radiation therapy and other perioperative factors on outcome after orthopedic stabilization of impending or pathologic fractures due to metastatic disease. J Clin Oncol. 1994;12(11):2345-2350.
23. Townsend PW, Smalley SR, Cozad SC, Rosenthal HG, Hassanein RE. Role of postoperative radiation therapy after stabilization of fractures caused by metastatic disease. Int J Radiat Oncol Biol Phys. 1995;31(1):43-49.
24. Farooki A. NCCN bone health task force: key recommendations. J Natl Compr Canc Netw. 2014;12(5 suppl):813-816.
25. Sejpal SV, Bhate A, Small W. Palliative radiation therapy in the management of brain metastases, spinal cord compression, and bone metastases. Semin Intervent Radiol. 2007;24(4):362-374.
26. Abrahm JL, Banffy MB, Harris MB. Spinal cord compression in patients with advanced metastatic cancer: “all I care about is walking and living my life.” JAMA. 2008;299(8):937-946.
27. Kim RY, Spencer SA, Meredith RF, et al. Extradural spinal cord compression: analysis of factors determining functional prognosis—prospective study. Radiology. 1990;176(1):279-282.
28. Kaloostian PE, Yurter A, Etame AB, Vrionis FD, Sciubba DM, Gokaslan ZL. Palliative strategies for the management of primary and metastatic spinal tumors. Cancer Control. 2014;21(2):140-143.
29. Graham PH, Capp A, Delaney G, et al. A pilot randomized comparison of dexamethasone 96 mg vs 16 mg per day for malignant spinal-cord compression treated by radiotherapy: TROG 01.05 Superdex study. Clin Oncol (R Coll Radiol). 2006;18(1):70-76.
30. Patchell RA, Tibbs PA, Regine WF, et al. Direct decompressive surgical resection in the treatment of spinal cord compression caused by metastatic cancer: a randomised trial. Lancet. 2005;366(9486):643-648.
31. Rades D, Huttenlocher S, Bajrovic A, et al. Surgery followed by radiotherapy versus radiotherapy alone for metastatic spinal cord compression from unfavorable tumors. Int J Radiat Oncol Biol Phys. 2011;81(5):e861-e868.
32. Maranzano E, Latini P. Effectiveness of radiation therapy without surgery in metastatic spinal cord compression: final results from a prospective trial. Int J Radiat Oncol Biol Phys. 1995;32(4):959-967.
33. Rades D, Fehlauer F, Schulte R, et al. Prognostic factors for local control and survival after radiotherapy of metastatic spinal cord compression. J Clin Oncol. 2006;24(21):3388-3393.
34. Howell DD, James JL, Hartsell WF, et al. Single-fraction radiotherapy versus multifraction radiotherapy for palliation of bone metastases-equivalent efficacy, less toxicity, more convenient: a subset analysis of Radiation Therapy Oncology Group trial 97-14. Cancer. 2013;119(4):888-896.
35. Wong J, Hird A, Kirou-Mauro, Napolskikh J, Chow E. Quality of life in brain metastases radiation trials: a literature review. Curr Oncol. 2008;15(5):25-45.
36. Nichols EM, Patchell RA, Regine WF, Kwok Y. Palliation of brain and spinal cord metastases. In: Halperin EC, Brady LW, Perez CA, Wazer DE, eds. Perez and Brady’s Principles and Practice of Radiation Oncology. 6th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2013:1974.
37. Zimm S, Wampler GL, Stablein D, Hazra T, Young HF. Intracerebral metastases in solid-tumor patients: natural history and results of treatment. Cancer. 1981;48(2):384-394.
38. Khuntia D, Brown P, Li J, Mehta MP. Whole-brain radiotherapy in the management of brain metastasis. J Clin Oncol. 2006;24(8):1295-1304.
39. Sundström JT, Minn H, Lertola KK, Nordman E. Prognosis of patients treated for intracranial metastases with whole-brain irradiation. Ann Med. 1998;30(3):296-299.
40. Tsao MN, Rades D, Wirth A, et al. Radiotherapeutic and surgical management for newly diagnosed brain metastasis(es): an American Society for Radiation Oncology evidence-based guideline. Pract Radiat Oncol. 2012;2(3):210-225.
41. Gaspar L, Scott C, Rotman M, et al. Recursive partitioning analysis (RPA) of prognostic factors in three Radiation Therapy Oncology Group (RTOG) brain metastases trials. Int J Radiat Oncol Biol Phys. 1997;37(4):745-751.
42. Sperduto PW, Berkey B, Gaspar LE, Mehta M, Curran W. A new prognostic index and comparison to three other indices for patients with brain metastases: an analysis of 1,960 patients in the RTOG databases. Int J Radiat Oncol Biol Phys. 2008;70(2):510-514.
43. Sperduto PW, Kased N, Roberge D, et al. Summary report on the graded prognostic assessment: an accurate and facile diagnosis-specific tool to estimate survival for patients with brain metastases. J Clin Oncol. 2012;30(4):419-425.
44. Ryken TC, McDermott M, Robinson PD, et al. The role of steroids in the management of brain metastases: a systematic review and evidence-based clinical practice guideline. J Neurooncol. 2010;96(1):103-114.
45. Ruderman NB, Hall TC. Use of glucocorticoids in the palliative treatment of metastatic brain tumors. Cancer. 1965;18:298-306.
46. Kaloostian PE, Yurter A, Etame AB, Vrionis FD, Sciubba DM, Gokaslan ZL. Palliative strategies for the management of primary and metastatic spinal tumors. Cancer Control. 2014;21(2):140-143.
47. Horton J, Baxter DH, Olson KB. The management of metastases to the brain by irradiation and corticosteroids. Am J Roentgenol Radium Ther Nucl Med. 1971;111(2)334-336.
48. Wong J, Hird A, Zhang L, et al. Symptoms and quality of life in cancer patients with brain metastases following palliative radiotherapy. Int J Radiat Oncol Biol Phys. 2009;75(4):1125-1131.
49. Li J, Bentzen SM, Renschler M, Mehta MP. Regression after whole-brain radiation therapy for brain metastases correlates with survival and improved neurocognitive function. J Clin Oncol. 2007;25(10):1260-1266.
50. Andrews DW, Scott CB, Sperduto PW, et al. Whole brain radiation therapy with or without stereotactic radiosurgery boost for patients with one to three brain metastases: phase III results of the RTOG 9508 randomised trial. Lancet. 2004;363(9422):1665-1672.
51. Patchell RA, Tibbs PA, Walsh JW, et al. A randomized trial of surgery in the treatment of single metastases to the brain. N Engl J Med. 1990;322(8):494-500.
52. Patchell RA, Tibbs PA, Regine WF, et al. Postoperative radiotherapy in the treatment of single metastases to the brain: a randomized trial. JAMA. 1998;280(17):1485-1489.
53. Kocher M, Soffietti R, Abacioglu U, et al. Adjuvant whole-brain radiotherapy versus observation after radiosurgery or surgical resection of one to three cerebral metastases: results of the EORTC 22952-26001 study. J Clin Oncol. 2011;29(2):134-141.
54. Chang EL, Wefel JS, Hess KR, et al. Neurocognition in patients with brain metastases treated with radiosurgery or radiosurgery plus whole-brain irradiation: a randomised controlled trial. Lancet Oncol. 2009;10(11):1037-1044.
Lessons from our dying patients
Dying patients teach us to think more carefully about whether or not our surgical interventions will be beneficial.
I work in palliative care, and my surgical colleagues, especially the residents, are often surprised when I call them and ask them to consult on my patients who are very ill and have a “Do Not Resuscitate” order in their charts. I’m also an anesthesiologist working in interventional pain management, and I regularly do procedures on patients who have prognoses that are extremely limited. For other patients, I recommend against any interventions at all.
How do we know when to intervene on patients who are dying? Perhaps more importantly, how do we know when NOT to intervene? Two recent cases of almost identical fractures illustrated for me the need to think beyond the anatomic problem when evaluating options for care.
Last year, I admitted a woman, “Donna,” with widely metastatic breast cancer to our inpatient palliative care service. She had fallen at home and hurt her arm about 2 months prior to admission. She had been confined to her bed for about 6 weeks. She was brought to the hospital because she was becoming delirious. She had many sources of pain that were relatively well controlled when she was lying down, but her worst pain was in her left arm. We found a fracture of her humerus. When her family learned that the fracture would not heal on its own because of the large metastasis there, they demanded surgery to fix it. Shortly thereafter, I re-admitted a patient, “Cindy,” with a very similar story. She also had widely metastatic breast cancer, and her pain had been very difficult to control. We had found a pain regimen that worked well for her on her previous admission, but she had fallen over her walker and broke her humerus after we had discharged her to a rehab facility. When I saw her back in the hospital, I told her that I thought she would need surgery to fix her arm. She was depressed by this setback, she was in pain again, and she told me that she would prefer not to have any intervention because she feared the additional pain that it would cause.
With Donna, we sat down with her and her family to hear what their hopes were for her care. They understood that she did not have further chemotherapy or radiation options for her cancer, but they thought if she got the surgery that at least she would be able to get out of bed and walk again. My colleague carefully explained that yes, he could fix the fracture and that this could mean that the pain in her left arm would improve. He went on to say, however, that he did not think that the surgery would allow her to walk again as she had not been able to walk for a few weeks after the injury. When the family heard that the surgery probably wouldn’t restore her mobility, they decided against the procedure. With Cindy, we had a very different conversation. She was not inclined to have the procedure, but I expressed my concern that she wouldn’t be able to walk again unless she had the procedure because she needed her arms to use her walker. Although she did not have any further chemotherapy or radiation options, her oncologist had told us that her prognosis could be several months. In this case, my surgical colleague explained that he could perform surgery for the fracture and that he thought that it would both help her pain and allow her to use her walker again. We recommended that she have the surgery given her hope to continue to live independently, as she had been, for as long as possible. She ultimately agreed to do so and was able to return home.
These two patients reminded me again of how important it is for us to understand what our patients’ hopes and expectations are for a procedure. It is very distressing for clinicians when desperate families want treatments that likely have little benefit. When patients have limited prognoses, aligning patient goals and procedure goals is especially important as the outcome of the procedure can define the patient’s remaining days.
Donna’s family demanded a surgery expecting a result that was very unlikely, and Cindy initially declined the same surgery that ultimately benefitted her greatly. Our job is to make and execute the medical recommendations that best fit with our patients’ goals and understanding. Sometimes this will mean performing procedures on patients who are extremely ill and have “Do Not Resuscitate” orders, and at other times, it will mean not doing procedures, even if a patient and family want them to be done.
Dr. Rickerson is an anesthesiologist at the Dana-Farber Cancer Institute and Brigham and Women’s Hospital, Boston.
Dying patients teach us to think more carefully about whether or not our surgical interventions will be beneficial.
I work in palliative care, and my surgical colleagues, especially the residents, are often surprised when I call them and ask them to consult on my patients who are very ill and have a “Do Not Resuscitate” order in their charts. I’m also an anesthesiologist working in interventional pain management, and I regularly do procedures on patients who have prognoses that are extremely limited. For other patients, I recommend against any interventions at all.
How do we know when to intervene on patients who are dying? Perhaps more importantly, how do we know when NOT to intervene? Two recent cases of almost identical fractures illustrated for me the need to think beyond the anatomic problem when evaluating options for care.
Last year, I admitted a woman, “Donna,” with widely metastatic breast cancer to our inpatient palliative care service. She had fallen at home and hurt her arm about 2 months prior to admission. She had been confined to her bed for about 6 weeks. She was brought to the hospital because she was becoming delirious. She had many sources of pain that were relatively well controlled when she was lying down, but her worst pain was in her left arm. We found a fracture of her humerus. When her family learned that the fracture would not heal on its own because of the large metastasis there, they demanded surgery to fix it. Shortly thereafter, I re-admitted a patient, “Cindy,” with a very similar story. She also had widely metastatic breast cancer, and her pain had been very difficult to control. We had found a pain regimen that worked well for her on her previous admission, but she had fallen over her walker and broke her humerus after we had discharged her to a rehab facility. When I saw her back in the hospital, I told her that I thought she would need surgery to fix her arm. She was depressed by this setback, she was in pain again, and she told me that she would prefer not to have any intervention because she feared the additional pain that it would cause.
With Donna, we sat down with her and her family to hear what their hopes were for her care. They understood that she did not have further chemotherapy or radiation options for her cancer, but they thought if she got the surgery that at least she would be able to get out of bed and walk again. My colleague carefully explained that yes, he could fix the fracture and that this could mean that the pain in her left arm would improve. He went on to say, however, that he did not think that the surgery would allow her to walk again as she had not been able to walk for a few weeks after the injury. When the family heard that the surgery probably wouldn’t restore her mobility, they decided against the procedure. With Cindy, we had a very different conversation. She was not inclined to have the procedure, but I expressed my concern that she wouldn’t be able to walk again unless she had the procedure because she needed her arms to use her walker. Although she did not have any further chemotherapy or radiation options, her oncologist had told us that her prognosis could be several months. In this case, my surgical colleague explained that he could perform surgery for the fracture and that he thought that it would both help her pain and allow her to use her walker again. We recommended that she have the surgery given her hope to continue to live independently, as she had been, for as long as possible. She ultimately agreed to do so and was able to return home.
These two patients reminded me again of how important it is for us to understand what our patients’ hopes and expectations are for a procedure. It is very distressing for clinicians when desperate families want treatments that likely have little benefit. When patients have limited prognoses, aligning patient goals and procedure goals is especially important as the outcome of the procedure can define the patient’s remaining days.
Donna’s family demanded a surgery expecting a result that was very unlikely, and Cindy initially declined the same surgery that ultimately benefitted her greatly. Our job is to make and execute the medical recommendations that best fit with our patients’ goals and understanding. Sometimes this will mean performing procedures on patients who are extremely ill and have “Do Not Resuscitate” orders, and at other times, it will mean not doing procedures, even if a patient and family want them to be done.
Dr. Rickerson is an anesthesiologist at the Dana-Farber Cancer Institute and Brigham and Women’s Hospital, Boston.
Dying patients teach us to think more carefully about whether or not our surgical interventions will be beneficial.
I work in palliative care, and my surgical colleagues, especially the residents, are often surprised when I call them and ask them to consult on my patients who are very ill and have a “Do Not Resuscitate” order in their charts. I’m also an anesthesiologist working in interventional pain management, and I regularly do procedures on patients who have prognoses that are extremely limited. For other patients, I recommend against any interventions at all.
How do we know when to intervene on patients who are dying? Perhaps more importantly, how do we know when NOT to intervene? Two recent cases of almost identical fractures illustrated for me the need to think beyond the anatomic problem when evaluating options for care.
Last year, I admitted a woman, “Donna,” with widely metastatic breast cancer to our inpatient palliative care service. She had fallen at home and hurt her arm about 2 months prior to admission. She had been confined to her bed for about 6 weeks. She was brought to the hospital because she was becoming delirious. She had many sources of pain that were relatively well controlled when she was lying down, but her worst pain was in her left arm. We found a fracture of her humerus. When her family learned that the fracture would not heal on its own because of the large metastasis there, they demanded surgery to fix it. Shortly thereafter, I re-admitted a patient, “Cindy,” with a very similar story. She also had widely metastatic breast cancer, and her pain had been very difficult to control. We had found a pain regimen that worked well for her on her previous admission, but she had fallen over her walker and broke her humerus after we had discharged her to a rehab facility. When I saw her back in the hospital, I told her that I thought she would need surgery to fix her arm. She was depressed by this setback, she was in pain again, and she told me that she would prefer not to have any intervention because she feared the additional pain that it would cause.
With Donna, we sat down with her and her family to hear what their hopes were for her care. They understood that she did not have further chemotherapy or radiation options for her cancer, but they thought if she got the surgery that at least she would be able to get out of bed and walk again. My colleague carefully explained that yes, he could fix the fracture and that this could mean that the pain in her left arm would improve. He went on to say, however, that he did not think that the surgery would allow her to walk again as she had not been able to walk for a few weeks after the injury. When the family heard that the surgery probably wouldn’t restore her mobility, they decided against the procedure. With Cindy, we had a very different conversation. She was not inclined to have the procedure, but I expressed my concern that she wouldn’t be able to walk again unless she had the procedure because she needed her arms to use her walker. Although she did not have any further chemotherapy or radiation options, her oncologist had told us that her prognosis could be several months. In this case, my surgical colleague explained that he could perform surgery for the fracture and that he thought that it would both help her pain and allow her to use her walker again. We recommended that she have the surgery given her hope to continue to live independently, as she had been, for as long as possible. She ultimately agreed to do so and was able to return home.
These two patients reminded me again of how important it is for us to understand what our patients’ hopes and expectations are for a procedure. It is very distressing for clinicians when desperate families want treatments that likely have little benefit. When patients have limited prognoses, aligning patient goals and procedure goals is especially important as the outcome of the procedure can define the patient’s remaining days.
Donna’s family demanded a surgery expecting a result that was very unlikely, and Cindy initially declined the same surgery that ultimately benefitted her greatly. Our job is to make and execute the medical recommendations that best fit with our patients’ goals and understanding. Sometimes this will mean performing procedures on patients who are extremely ill and have “Do Not Resuscitate” orders, and at other times, it will mean not doing procedures, even if a patient and family want them to be done.
Dr. Rickerson is an anesthesiologist at the Dana-Farber Cancer Institute and Brigham and Women’s Hospital, Boston.
Hospice, Palliative Care Groups Release Quality Care Measures
The American Academy of Hospice and Palliative Medicine (AAHPM) and the Hospice & Palliative Nurses Association (HPNA) recently published a list of performance measures to assess the quality of palliative and hospice patient care.
Refined over two years, the groups' Measuring What Matters recommendations [PDF] outline 10 clinically relevant measures to drive quality care. The list includes:
- Documenting patients’ preferences for life-sustaining treatments and their surrogate decision makers’ names;
- Screening patients for physical symptoms;
- Treating pain;
- Screening and managing dyspnea; and,
- Discussing patients' emotional and psychological needs.
"I'd say these things are relevant for hospitalists' patients, and for all seriously ill patients, whether or not a palliative care need has been identified," says Joe Rotella, MD, MBA, AAHPM's CMO and co-chair of the Measuring What Matters clinical user panel. The measures should make it possible to raise awareness about what constitutes quality of care for seriously ill patients and to compare quality between settings and between patients who receive palliative care and equally ill patients who do not, he notes.
The quality indicators, which have been reviewed by the National Quality Forum, focus on processes of providing palliative and hospice care and seek to achieve consistency in care quality among providers. For instance, do patients who screen positive for at least moderate pain receive treatments within 24 hours? Likewise, patients receiving hospice care should have a documented discussion of their spiritual concerns or of their preference not to have such a discussion, the recommendations state.
"It's worth looking at what really matters to these patients and maybe adapting a few measures for your hospital's quality improvement program," Dr. Rotella says.
Listen to our recent podcast on hospitalists and palliative care.
The American Academy of Hospice and Palliative Medicine (AAHPM) and the Hospice & Palliative Nurses Association (HPNA) recently published a list of performance measures to assess the quality of palliative and hospice patient care.
Refined over two years, the groups' Measuring What Matters recommendations [PDF] outline 10 clinically relevant measures to drive quality care. The list includes:
- Documenting patients’ preferences for life-sustaining treatments and their surrogate decision makers’ names;
- Screening patients for physical symptoms;
- Treating pain;
- Screening and managing dyspnea; and,
- Discussing patients' emotional and psychological needs.
"I'd say these things are relevant for hospitalists' patients, and for all seriously ill patients, whether or not a palliative care need has been identified," says Joe Rotella, MD, MBA, AAHPM's CMO and co-chair of the Measuring What Matters clinical user panel. The measures should make it possible to raise awareness about what constitutes quality of care for seriously ill patients and to compare quality between settings and between patients who receive palliative care and equally ill patients who do not, he notes.
The quality indicators, which have been reviewed by the National Quality Forum, focus on processes of providing palliative and hospice care and seek to achieve consistency in care quality among providers. For instance, do patients who screen positive for at least moderate pain receive treatments within 24 hours? Likewise, patients receiving hospice care should have a documented discussion of their spiritual concerns or of their preference not to have such a discussion, the recommendations state.
"It's worth looking at what really matters to these patients and maybe adapting a few measures for your hospital's quality improvement program," Dr. Rotella says.
Listen to our recent podcast on hospitalists and palliative care.
The American Academy of Hospice and Palliative Medicine (AAHPM) and the Hospice & Palliative Nurses Association (HPNA) recently published a list of performance measures to assess the quality of palliative and hospice patient care.
Refined over two years, the groups' Measuring What Matters recommendations [PDF] outline 10 clinically relevant measures to drive quality care. The list includes:
- Documenting patients’ preferences for life-sustaining treatments and their surrogate decision makers’ names;
- Screening patients for physical symptoms;
- Treating pain;
- Screening and managing dyspnea; and,
- Discussing patients' emotional and psychological needs.
"I'd say these things are relevant for hospitalists' patients, and for all seriously ill patients, whether or not a palliative care need has been identified," says Joe Rotella, MD, MBA, AAHPM's CMO and co-chair of the Measuring What Matters clinical user panel. The measures should make it possible to raise awareness about what constitutes quality of care for seriously ill patients and to compare quality between settings and between patients who receive palliative care and equally ill patients who do not, he notes.
The quality indicators, which have been reviewed by the National Quality Forum, focus on processes of providing palliative and hospice care and seek to achieve consistency in care quality among providers. For instance, do patients who screen positive for at least moderate pain receive treatments within 24 hours? Likewise, patients receiving hospice care should have a documented discussion of their spiritual concerns or of their preference not to have such a discussion, the recommendations state.
"It's worth looking at what really matters to these patients and maybe adapting a few measures for your hospital's quality improvement program," Dr. Rotella says.
Listen to our recent podcast on hospitalists and palliative care.
Using Genetics to Fight Disease
In January, President Obama launched the Precision Medicine Initiative, a far-reaching national program aimed at incorporating individual variability in genes, environment, and lifestyle into the treatment of diseases. The initiative incorporates genomic medicine, a growing medical field that applies individual genetic information and variation to better tailor clinical care for each patient. The goal: to make a difference for the millions of Americans facing illness, improve treatment options, and revolutionize everyday clinical practice.
Federal Practitioner talked with Eric Green, MD, PhD, the director of NIH’s National Human Genome Research Institute about how the NIH is using genomics to meet the goals of the Precision Medicine Initiative and its impact on the future of medicine.
Click below to hear the full discussion.
Federal Practitioner: What is precision and personalized medicine?
Dr. Eric Green: Genomic medicine is something that reflects the use of genomic information about an individual patient to tailor their clinical care. That is very much focused on genomic information, but of course, there are other components of disease beyond genomics and genetics. It is not all in our DNA. There are also aspects of everyday life that influence our health and our well-being and our risk for diseases such as our diet, our lifestyle, what things we are exposed to in the environment, and so forth.
A larger framing of the notion of individualizing medical care around each individual’s makeup in terms of their blueprint and what they are exposed to brings us to phrases like personalized medicine or individualized medicine. More recently, the phrase that has been used is precision medicine. Precision medicine, essentially, takes a broader view of what are the things that contribute to health and disease. It is genomics, it is lifestyle, it is diet, it is environmental exposures, and then thinking about how to tailor medical treatment based on taking those individual aspects of a person into account.
FP: How can genomics be applied to hematology and oncology?
EG: Some of the earliest applications of using genomic information to tailor medical care come in the area of cancer; in particular, some of the leukemias that are studied and treated every day in hospitals around the world where we have learned by taking samples from individuals who have certain types of leukemia and opening up their genomes. In those tumor samples, in those leukemia samples, there turns out to be some very characteristic genomic changes that have taken place that are basically driving those cancers and making them behave the way they do.
It is now standard practice for some hematologic disorders, in particular types of leukemia, to consider getting genomic information on that specific patient—a blood sample—in order to help guide the best way to treat the patient and to get information about prognosis. It is only going to get more exciting and more advanced over the next 5 and 10 years.
FP: Why do you think this topic warrants discussion?
EG: I think this warrants discussing in almost any clinical group, because I completely believe that over the next 5 to 10 years, many aspects of medicine are going to be changed because of genomics. It is not going to just be in certain areas of medicine; I think it is going to be in almost all areas of medicine. This makes it very relevant to talk to clinical groups about this fast-moving area and how it will find its way into clinical practice in the next few years.
FP: You will be presenting the keynote presentation at the 2015 AVAHO Meeting in Washington, DC, in October. Are you excited to bring the discussion of genomics to AVAHO?
EG: I talk to clinical groups all the time, because I firmly believe this is going to be really important for them to know. I also fully appreciate that the field has moved so quickly that a lot of people who are out there in practice—whether they are physicians, pharmacists, nurses, physician assistants, or genetic counselors—when they were trained even 5, or 10, or 20 years ago, the things that we can do know using genomic information simply weren’t known about then. It is very important for us to use professional meetings as venues for furthering the education of professionals, especially in fast-moving fields where this is a great opportunity for them to catch up on what the latest is.
FP: Is there anything else you would like to say?
EG: The Precision Medicine Initiative aims to accelerate progress in precision medicine so that people in the United States can be the beneficiaries of these exciting developments. I think it is going to be an important partnership with scientists and practicing physicians to make this a reality, so it is really important for us to be talking about these things.
Don't have time to listen to the entire discussion? We understand. Use this guide to skip ahead to the topic that most interests you.
1:04 What is genomic medicine?
2:36 What is precision medicine?
3:48 How can genomics be used in hematology and oncology?
6:25 How is genomics changing the future of medicine?
7:22 Why should clinical groups like AVAHO learn about genomics?
8:20 How is the Precision Medicine Initiative using genomics?
In January, President Obama launched the Precision Medicine Initiative, a far-reaching national program aimed at incorporating individual variability in genes, environment, and lifestyle into the treatment of diseases. The initiative incorporates genomic medicine, a growing medical field that applies individual genetic information and variation to better tailor clinical care for each patient. The goal: to make a difference for the millions of Americans facing illness, improve treatment options, and revolutionize everyday clinical practice.
Federal Practitioner talked with Eric Green, MD, PhD, the director of NIH’s National Human Genome Research Institute about how the NIH is using genomics to meet the goals of the Precision Medicine Initiative and its impact on the future of medicine.
Click below to hear the full discussion.
Federal Practitioner: What is precision and personalized medicine?
Dr. Eric Green: Genomic medicine is something that reflects the use of genomic information about an individual patient to tailor their clinical care. That is very much focused on genomic information, but of course, there are other components of disease beyond genomics and genetics. It is not all in our DNA. There are also aspects of everyday life that influence our health and our well-being and our risk for diseases such as our diet, our lifestyle, what things we are exposed to in the environment, and so forth.
A larger framing of the notion of individualizing medical care around each individual’s makeup in terms of their blueprint and what they are exposed to brings us to phrases like personalized medicine or individualized medicine. More recently, the phrase that has been used is precision medicine. Precision medicine, essentially, takes a broader view of what are the things that contribute to health and disease. It is genomics, it is lifestyle, it is diet, it is environmental exposures, and then thinking about how to tailor medical treatment based on taking those individual aspects of a person into account.
FP: How can genomics be applied to hematology and oncology?
EG: Some of the earliest applications of using genomic information to tailor medical care come in the area of cancer; in particular, some of the leukemias that are studied and treated every day in hospitals around the world where we have learned by taking samples from individuals who have certain types of leukemia and opening up their genomes. In those tumor samples, in those leukemia samples, there turns out to be some very characteristic genomic changes that have taken place that are basically driving those cancers and making them behave the way they do.
It is now standard practice for some hematologic disorders, in particular types of leukemia, to consider getting genomic information on that specific patient—a blood sample—in order to help guide the best way to treat the patient and to get information about prognosis. It is only going to get more exciting and more advanced over the next 5 and 10 years.
FP: Why do you think this topic warrants discussion?
EG: I think this warrants discussing in almost any clinical group, because I completely believe that over the next 5 to 10 years, many aspects of medicine are going to be changed because of genomics. It is not going to just be in certain areas of medicine; I think it is going to be in almost all areas of medicine. This makes it very relevant to talk to clinical groups about this fast-moving area and how it will find its way into clinical practice in the next few years.
FP: You will be presenting the keynote presentation at the 2015 AVAHO Meeting in Washington, DC, in October. Are you excited to bring the discussion of genomics to AVAHO?
EG: I talk to clinical groups all the time, because I firmly believe this is going to be really important for them to know. I also fully appreciate that the field has moved so quickly that a lot of people who are out there in practice—whether they are physicians, pharmacists, nurses, physician assistants, or genetic counselors—when they were trained even 5, or 10, or 20 years ago, the things that we can do know using genomic information simply weren’t known about then. It is very important for us to use professional meetings as venues for furthering the education of professionals, especially in fast-moving fields where this is a great opportunity for them to catch up on what the latest is.
FP: Is there anything else you would like to say?
EG: The Precision Medicine Initiative aims to accelerate progress in precision medicine so that people in the United States can be the beneficiaries of these exciting developments. I think it is going to be an important partnership with scientists and practicing physicians to make this a reality, so it is really important for us to be talking about these things.
Don't have time to listen to the entire discussion? We understand. Use this guide to skip ahead to the topic that most interests you.
1:04 What is genomic medicine?
2:36 What is precision medicine?
3:48 How can genomics be used in hematology and oncology?
6:25 How is genomics changing the future of medicine?
7:22 Why should clinical groups like AVAHO learn about genomics?
8:20 How is the Precision Medicine Initiative using genomics?
In January, President Obama launched the Precision Medicine Initiative, a far-reaching national program aimed at incorporating individual variability in genes, environment, and lifestyle into the treatment of diseases. The initiative incorporates genomic medicine, a growing medical field that applies individual genetic information and variation to better tailor clinical care for each patient. The goal: to make a difference for the millions of Americans facing illness, improve treatment options, and revolutionize everyday clinical practice.
Federal Practitioner talked with Eric Green, MD, PhD, the director of NIH’s National Human Genome Research Institute about how the NIH is using genomics to meet the goals of the Precision Medicine Initiative and its impact on the future of medicine.
Click below to hear the full discussion.
Federal Practitioner: What is precision and personalized medicine?
Dr. Eric Green: Genomic medicine is something that reflects the use of genomic information about an individual patient to tailor their clinical care. That is very much focused on genomic information, but of course, there are other components of disease beyond genomics and genetics. It is not all in our DNA. There are also aspects of everyday life that influence our health and our well-being and our risk for diseases such as our diet, our lifestyle, what things we are exposed to in the environment, and so forth.
A larger framing of the notion of individualizing medical care around each individual’s makeup in terms of their blueprint and what they are exposed to brings us to phrases like personalized medicine or individualized medicine. More recently, the phrase that has been used is precision medicine. Precision medicine, essentially, takes a broader view of what are the things that contribute to health and disease. It is genomics, it is lifestyle, it is diet, it is environmental exposures, and then thinking about how to tailor medical treatment based on taking those individual aspects of a person into account.
FP: How can genomics be applied to hematology and oncology?
EG: Some of the earliest applications of using genomic information to tailor medical care come in the area of cancer; in particular, some of the leukemias that are studied and treated every day in hospitals around the world where we have learned by taking samples from individuals who have certain types of leukemia and opening up their genomes. In those tumor samples, in those leukemia samples, there turns out to be some very characteristic genomic changes that have taken place that are basically driving those cancers and making them behave the way they do.
It is now standard practice for some hematologic disorders, in particular types of leukemia, to consider getting genomic information on that specific patient—a blood sample—in order to help guide the best way to treat the patient and to get information about prognosis. It is only going to get more exciting and more advanced over the next 5 and 10 years.
FP: Why do you think this topic warrants discussion?
EG: I think this warrants discussing in almost any clinical group, because I completely believe that over the next 5 to 10 years, many aspects of medicine are going to be changed because of genomics. It is not going to just be in certain areas of medicine; I think it is going to be in almost all areas of medicine. This makes it very relevant to talk to clinical groups about this fast-moving area and how it will find its way into clinical practice in the next few years.
FP: You will be presenting the keynote presentation at the 2015 AVAHO Meeting in Washington, DC, in October. Are you excited to bring the discussion of genomics to AVAHO?
EG: I talk to clinical groups all the time, because I firmly believe this is going to be really important for them to know. I also fully appreciate that the field has moved so quickly that a lot of people who are out there in practice—whether they are physicians, pharmacists, nurses, physician assistants, or genetic counselors—when they were trained even 5, or 10, or 20 years ago, the things that we can do know using genomic information simply weren’t known about then. It is very important for us to use professional meetings as venues for furthering the education of professionals, especially in fast-moving fields where this is a great opportunity for them to catch up on what the latest is.
FP: Is there anything else you would like to say?
EG: The Precision Medicine Initiative aims to accelerate progress in precision medicine so that people in the United States can be the beneficiaries of these exciting developments. I think it is going to be an important partnership with scientists and practicing physicians to make this a reality, so it is really important for us to be talking about these things.
Don't have time to listen to the entire discussion? We understand. Use this guide to skip ahead to the topic that most interests you.
1:04 What is genomic medicine?
2:36 What is precision medicine?
3:48 How can genomics be used in hematology and oncology?
6:25 How is genomics changing the future of medicine?
7:22 Why should clinical groups like AVAHO learn about genomics?
8:20 How is the Precision Medicine Initiative using genomics?
Palliative Care and Last-Minute Heroics
4/8/15
HM15 Presenter: Tammie Quest, MD
Summation: Heroics- a set of medical actions that attempt to prolong life with a low likelihood of success.
Palliative care- an approach of care provided to patients and families suffering from serious and/or life limiting illness; focus on physical, spiritual, psychological and social aspects of distress.
Hospice care- intense palliative care provided when the patient has terminal illness with a prognosis of 6 months or less if the disease runs its usual course.
We underutilize Palliative and Hospice care in the US. Here in the US fewer than 50% of all persons receive hospice care at EOL, of those who receive hospice care more than half receive care for less than 20 days, and 1 in 5 patients die in an ICU. Palliative Care can/should co-exist with life prolonging care following the diagnosis of serious illness.
Common therapies/interventions to be contemplated and discussed with patient at end of life: cpr, mechanical ventilation, central venous/arterial access, renal replacement therapy, surgical procedures, valve therapies, ventricular assist devices, continuous infusions, IV fluids, supplemental oxygen, artificial nutrition, antimicrobials, blood products, cancer directed therapy, antithrombotics, anticoagulation.
Practical Elements of Palliative Care: pain and symptom management, advance care planning, communication/goals of care, truth-telling, social support, spiritual support, psychological support, risk/burden assessment of treatments.
Key Points/HM Takeaways:
1-Palliative Care Bedside Talking Points-
- Cardiac arrest is the moment of death, very few people survive an attempt at reversing death
- If you are one of the few who survive to discharge, you may do well but few will survive to discharge
- Antibiotics DO improve survival, antibiotics DO NOT improve comfort
- No evidence to show that dying from pneumonia, or other infection, is painful
- Allowing natural death includes permitting the body to shut itself down through natural mechanisms, including infection
- Dialysis may extend life, but there will be progressive functional decline
2-Goals of Care define what therapies are indicated. Balance prolongation of life with illness experience.
Julianna Lindsey is a hospitalist and physician leader based in the Dallas-Fort Worth Metroplex. Her focus is patient safety/quality and physician leadership. She is a member of TeamHospitalist.
4/8/15
HM15 Presenter: Tammie Quest, MD
Summation: Heroics- a set of medical actions that attempt to prolong life with a low likelihood of success.
Palliative care- an approach of care provided to patients and families suffering from serious and/or life limiting illness; focus on physical, spiritual, psychological and social aspects of distress.
Hospice care- intense palliative care provided when the patient has terminal illness with a prognosis of 6 months or less if the disease runs its usual course.
We underutilize Palliative and Hospice care in the US. Here in the US fewer than 50% of all persons receive hospice care at EOL, of those who receive hospice care more than half receive care for less than 20 days, and 1 in 5 patients die in an ICU. Palliative Care can/should co-exist with life prolonging care following the diagnosis of serious illness.
Common therapies/interventions to be contemplated and discussed with patient at end of life: cpr, mechanical ventilation, central venous/arterial access, renal replacement therapy, surgical procedures, valve therapies, ventricular assist devices, continuous infusions, IV fluids, supplemental oxygen, artificial nutrition, antimicrobials, blood products, cancer directed therapy, antithrombotics, anticoagulation.
Practical Elements of Palliative Care: pain and symptom management, advance care planning, communication/goals of care, truth-telling, social support, spiritual support, psychological support, risk/burden assessment of treatments.
Key Points/HM Takeaways:
1-Palliative Care Bedside Talking Points-
- Cardiac arrest is the moment of death, very few people survive an attempt at reversing death
- If you are one of the few who survive to discharge, you may do well but few will survive to discharge
- Antibiotics DO improve survival, antibiotics DO NOT improve comfort
- No evidence to show that dying from pneumonia, or other infection, is painful
- Allowing natural death includes permitting the body to shut itself down through natural mechanisms, including infection
- Dialysis may extend life, but there will be progressive functional decline
2-Goals of Care define what therapies are indicated. Balance prolongation of life with illness experience.
Julianna Lindsey is a hospitalist and physician leader based in the Dallas-Fort Worth Metroplex. Her focus is patient safety/quality and physician leadership. She is a member of TeamHospitalist.
4/8/15
HM15 Presenter: Tammie Quest, MD
Summation: Heroics- a set of medical actions that attempt to prolong life with a low likelihood of success.
Palliative care- an approach of care provided to patients and families suffering from serious and/or life limiting illness; focus on physical, spiritual, psychological and social aspects of distress.
Hospice care- intense palliative care provided when the patient has terminal illness with a prognosis of 6 months or less if the disease runs its usual course.
We underutilize Palliative and Hospice care in the US. Here in the US fewer than 50% of all persons receive hospice care at EOL, of those who receive hospice care more than half receive care for less than 20 days, and 1 in 5 patients die in an ICU. Palliative Care can/should co-exist with life prolonging care following the diagnosis of serious illness.
Common therapies/interventions to be contemplated and discussed with patient at end of life: cpr, mechanical ventilation, central venous/arterial access, renal replacement therapy, surgical procedures, valve therapies, ventricular assist devices, continuous infusions, IV fluids, supplemental oxygen, artificial nutrition, antimicrobials, blood products, cancer directed therapy, antithrombotics, anticoagulation.
Practical Elements of Palliative Care: pain and symptom management, advance care planning, communication/goals of care, truth-telling, social support, spiritual support, psychological support, risk/burden assessment of treatments.
Key Points/HM Takeaways:
1-Palliative Care Bedside Talking Points-
- Cardiac arrest is the moment of death, very few people survive an attempt at reversing death
- If you are one of the few who survive to discharge, you may do well but few will survive to discharge
- Antibiotics DO improve survival, antibiotics DO NOT improve comfort
- No evidence to show that dying from pneumonia, or other infection, is painful
- Allowing natural death includes permitting the body to shut itself down through natural mechanisms, including infection
- Dialysis may extend life, but there will be progressive functional decline
2-Goals of Care define what therapies are indicated. Balance prolongation of life with illness experience.
Julianna Lindsey is a hospitalist and physician leader based in the Dallas-Fort Worth Metroplex. Her focus is patient safety/quality and physician leadership. She is a member of TeamHospitalist.