User login
AHA: Three measures risk stratify acute heart failure
ORLANDO – Three simple, routinely collected measurements together provide a lot of insight into the risk faced by community-dwelling patients hospitalized for acute decompensated heart failure, according to data collected from 3,628 patients at one U.S. center.
The three measures are blood urea nitrogen (BUN), systolic blood pressure, and serum creatinine. Using dichotomous cutoffs first calculated a decade ago, these three parameters distinguish up to an eightfold range of postdischarge mortality during the 30 or 90 days following an index hospitalization, and up to a fourfold range of risk for rehospitalization for heart failure during the ensuing 30 or 90 days, Dr. Sithu Win said at the American Heart Association scientific sessions.
Applying this three-measure assessment to patients hospitalized with acute decompensated heart failure “may guide care-transition planning and promote efficient allocation of limited resources,” said Dr. Win, a cardiologist at the Mayo Clinic in Rochester, Minn. The next step is to try to figure out the best way to use this risk prognostication in routine practice, he added.
Researchers published the original analysis that identified BUN, systolic BP, and serum creatinine as key prognostic measures in 2005 using data taken from more than 65,000 U.S. heart failure patients enrolled in ADHERE (Acute Decompensated Heart Failure National Registry) (JAMA. 2005 Feb 2;293[5]:572-80). Using a classification and regression tree analysis, the 2005 study verified dichotomous cutoffs for these three parameters that identified patients at highest risk for in-hospital mortality.
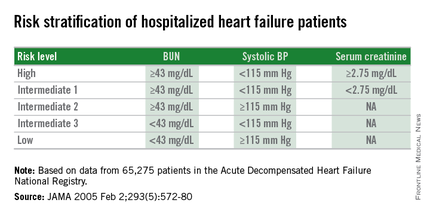
The 2005 study prioritized the application of these cutoffs to define in-hospital mortality risk: first BUN, then the systolic BP criterion, and lastly the serum creatinine criterion. This resulted in five risk levels: Highest-risk patients had a BUN of at least 43 mg/dL, a systolic BP of less than 115 mm Hg, and a serum creatinine of at least 2.75 mg/dL. Lowest-risk patients had a BUN of less than 43 mg/dL and a systolic BP of more than 115 mm Hg. (In lower-risk patients, serum-creatinine level dropped out as a risk determinant.) The analysis also created three categories of patients with intermediate risk based on various combinations of the three measures.
The new study run by Dr. Win and his associates evaluated how this risk-assessment tool developed to predict in-hospital mortality performed for predicting event rates among community-based heart failure patients who had a total of 5,918 hospitalizations for acute decompensated heart failure at the Mayo Clinic during 2000-2013. They averaged 78 years old, half were women, and 48% had heart failure with preserved ejection fraction.
The risk-level distribution of the 3,628 Mayo patients closely matched the pattern seen in the original ADHERE registry: 63% were low risk, 17% were at intermediate level 3 (the lowest risk level in the intermediate range), 13% were at intermediate level 2, 5% at intermediate level 1, and 2% were categorized as high risk.
For 30-day mortality post hospitalization, patients at the highest risk level had a mortality rate eightfold higher than did the lowest-risk patients, those rated intermediate level 1 had a fivefold higher mortality rate, intermediate level 2 patients had a threefold higher rate, and those at intermediate 3 had a 50% higher rate, Dr. Win reported. During the 90 days after discharge, mortality rates relative to the lowest risk level ranged from a sixfold higher rate among the highest-risk patients to a 50% higher rate among patients with an intermediate 3 designation.
Analysis of rehospitalizations for heart failure showed that, by 30 days after hospitalization, the readmission rate ran threefold higher in the highest-risk patients, compared with those at the lowest risk and fourfold higher among those at intermediate risk level 1. Heart failure readmissions by 90 days following the index hospitalization ran threefold higher for both the highest-risk patients as well as those at intermediate level 1, compared with the patients at lowest risk.
The new analyses also showed that roughly similar risk patterns occurred regardless of whether patients had heart failure with reduced or preserved ejection fraction during their index hospitalization, although the relatively increased rate of 30-day mortality with a worse risk profile was most dramatic among patients with reduced ejection fraction. Age, sex, and comorbidity severity did not have a marked effect on the relationships between event rates and risk levels, Dr. Win said.
Dr. Win had no disclosures.
On Twitter @mitchelzoler
ORLANDO – Three simple, routinely collected measurements together provide a lot of insight into the risk faced by community-dwelling patients hospitalized for acute decompensated heart failure, according to data collected from 3,628 patients at one U.S. center.
The three measures are blood urea nitrogen (BUN), systolic blood pressure, and serum creatinine. Using dichotomous cutoffs first calculated a decade ago, these three parameters distinguish up to an eightfold range of postdischarge mortality during the 30 or 90 days following an index hospitalization, and up to a fourfold range of risk for rehospitalization for heart failure during the ensuing 30 or 90 days, Dr. Sithu Win said at the American Heart Association scientific sessions.
Applying this three-measure assessment to patients hospitalized with acute decompensated heart failure “may guide care-transition planning and promote efficient allocation of limited resources,” said Dr. Win, a cardiologist at the Mayo Clinic in Rochester, Minn. The next step is to try to figure out the best way to use this risk prognostication in routine practice, he added.
Researchers published the original analysis that identified BUN, systolic BP, and serum creatinine as key prognostic measures in 2005 using data taken from more than 65,000 U.S. heart failure patients enrolled in ADHERE (Acute Decompensated Heart Failure National Registry) (JAMA. 2005 Feb 2;293[5]:572-80). Using a classification and regression tree analysis, the 2005 study verified dichotomous cutoffs for these three parameters that identified patients at highest risk for in-hospital mortality.
The 2005 study prioritized the application of these cutoffs to define in-hospital mortality risk: first BUN, then the systolic BP criterion, and lastly the serum creatinine criterion. This resulted in five risk levels: Highest-risk patients had a BUN of at least 43 mg/dL, a systolic BP of less than 115 mm Hg, and a serum creatinine of at least 2.75 mg/dL. Lowest-risk patients had a BUN of less than 43 mg/dL and a systolic BP of more than 115 mm Hg. (In lower-risk patients, serum-creatinine level dropped out as a risk determinant.) The analysis also created three categories of patients with intermediate risk based on various combinations of the three measures.

The new study run by Dr. Win and his associates evaluated how this risk-assessment tool developed to predict in-hospital mortality performed for predicting event rates among community-based heart failure patients who had a total of 5,918 hospitalizations for acute decompensated heart failure at the Mayo Clinic during 2000-2013. They averaged 78 years old, half were women, and 48% had heart failure with preserved ejection fraction.
The risk-level distribution of the 3,628 Mayo patients closely matched the pattern seen in the original ADHERE registry: 63% were low risk, 17% were at intermediate level 3 (the lowest risk level in the intermediate range), 13% were at intermediate level 2, 5% at intermediate level 1, and 2% were categorized as high risk.
For 30-day mortality post hospitalization, patients at the highest risk level had a mortality rate eightfold higher than did the lowest-risk patients, those rated intermediate level 1 had a fivefold higher mortality rate, intermediate level 2 patients had a threefold higher rate, and those at intermediate 3 had a 50% higher rate, Dr. Win reported. During the 90 days after discharge, mortality rates relative to the lowest risk level ranged from a sixfold higher rate among the highest-risk patients to a 50% higher rate among patients with an intermediate 3 designation.
Analysis of rehospitalizations for heart failure showed that, by 30 days after hospitalization, the readmission rate ran threefold higher in the highest-risk patients, compared with those at the lowest risk and fourfold higher among those at intermediate risk level 1. Heart failure readmissions by 90 days following the index hospitalization ran threefold higher for both the highest-risk patients as well as those at intermediate level 1, compared with the patients at lowest risk.
The new analyses also showed that roughly similar risk patterns occurred regardless of whether patients had heart failure with reduced or preserved ejection fraction during their index hospitalization, although the relatively increased rate of 30-day mortality with a worse risk profile was most dramatic among patients with reduced ejection fraction. Age, sex, and comorbidity severity did not have a marked effect on the relationships between event rates and risk levels, Dr. Win said.
Dr. Win had no disclosures.
On Twitter @mitchelzoler
ORLANDO – Three simple, routinely collected measurements together provide a lot of insight into the risk faced by community-dwelling patients hospitalized for acute decompensated heart failure, according to data collected from 3,628 patients at one U.S. center.
The three measures are blood urea nitrogen (BUN), systolic blood pressure, and serum creatinine. Using dichotomous cutoffs first calculated a decade ago, these three parameters distinguish up to an eightfold range of postdischarge mortality during the 30 or 90 days following an index hospitalization, and up to a fourfold range of risk for rehospitalization for heart failure during the ensuing 30 or 90 days, Dr. Sithu Win said at the American Heart Association scientific sessions.
Applying this three-measure assessment to patients hospitalized with acute decompensated heart failure “may guide care-transition planning and promote efficient allocation of limited resources,” said Dr. Win, a cardiologist at the Mayo Clinic in Rochester, Minn. The next step is to try to figure out the best way to use this risk prognostication in routine practice, he added.
Researchers published the original analysis that identified BUN, systolic BP, and serum creatinine as key prognostic measures in 2005 using data taken from more than 65,000 U.S. heart failure patients enrolled in ADHERE (Acute Decompensated Heart Failure National Registry) (JAMA. 2005 Feb 2;293[5]:572-80). Using a classification and regression tree analysis, the 2005 study verified dichotomous cutoffs for these three parameters that identified patients at highest risk for in-hospital mortality.
The 2005 study prioritized the application of these cutoffs to define in-hospital mortality risk: first BUN, then the systolic BP criterion, and lastly the serum creatinine criterion. This resulted in five risk levels: Highest-risk patients had a BUN of at least 43 mg/dL, a systolic BP of less than 115 mm Hg, and a serum creatinine of at least 2.75 mg/dL. Lowest-risk patients had a BUN of less than 43 mg/dL and a systolic BP of more than 115 mm Hg. (In lower-risk patients, serum-creatinine level dropped out as a risk determinant.) The analysis also created three categories of patients with intermediate risk based on various combinations of the three measures.

The new study run by Dr. Win and his associates evaluated how this risk-assessment tool developed to predict in-hospital mortality performed for predicting event rates among community-based heart failure patients who had a total of 5,918 hospitalizations for acute decompensated heart failure at the Mayo Clinic during 2000-2013. They averaged 78 years old, half were women, and 48% had heart failure with preserved ejection fraction.
The risk-level distribution of the 3,628 Mayo patients closely matched the pattern seen in the original ADHERE registry: 63% were low risk, 17% were at intermediate level 3 (the lowest risk level in the intermediate range), 13% were at intermediate level 2, 5% at intermediate level 1, and 2% were categorized as high risk.
For 30-day mortality post hospitalization, patients at the highest risk level had a mortality rate eightfold higher than did the lowest-risk patients, those rated intermediate level 1 had a fivefold higher mortality rate, intermediate level 2 patients had a threefold higher rate, and those at intermediate 3 had a 50% higher rate, Dr. Win reported. During the 90 days after discharge, mortality rates relative to the lowest risk level ranged from a sixfold higher rate among the highest-risk patients to a 50% higher rate among patients with an intermediate 3 designation.
Analysis of rehospitalizations for heart failure showed that, by 30 days after hospitalization, the readmission rate ran threefold higher in the highest-risk patients, compared with those at the lowest risk and fourfold higher among those at intermediate risk level 1. Heart failure readmissions by 90 days following the index hospitalization ran threefold higher for both the highest-risk patients as well as those at intermediate level 1, compared with the patients at lowest risk.
The new analyses also showed that roughly similar risk patterns occurred regardless of whether patients had heart failure with reduced or preserved ejection fraction during their index hospitalization, although the relatively increased rate of 30-day mortality with a worse risk profile was most dramatic among patients with reduced ejection fraction. Age, sex, and comorbidity severity did not have a marked effect on the relationships between event rates and risk levels, Dr. Win said.
Dr. Win had no disclosures.
On Twitter @mitchelzoler
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point: Dichotomous cutoffs for BUN, systolic BP, and serum creatinine together robustly risk stratified patients hospitalized for acute decompensated heart failure.
Major finding: Three baseline measures together stratified patients over an eightfold range for 30-day mortality following hospital discharge.
Data source: Application of the risk-stratification formula to 3,628 heart failure patients hospitalized at one U.S. center during 2000-2013.
Disclosures: Dr. Win had no disclosures.
AHA: Spirometry identifies mortality risk in asymptomatic adults
ORLANDO – Unselected people from the general population without clinically apparent lung disease but with low lung function had significantly increased mortality during follow-up that was independent of cardiac function, in results from more than 13,000 middle-aged Germans.
“Subtle, subclinical pulmonary impairment is a risk indicator for increased mortality independent of cardiac performance,” Dr. Christina Baum said at the American Heart Association scientific sessions.
The researchers used spirometry to measure each subject’s forced expiratory volume in 1 second (FEV1) and forced vital capacity (FVC). The results showed that “spirometry is a good screening tool that is not very expensive,” making spirometry an effective risk assessment tool for use in the general adult population, said Dr. Baum of the department of general and interventional cardiology at the University Heart Center in Hamburg, Germany.
She and her associates used data collected in the Gutenberg Health Study, which enrolled more than 15,000 German women and men aged 35-74 years during 2007-2012. The investigators excluded people with a history of pulmonary disease, resulting in a study cohort of 13,191, who averaged 55 years old, with 51% men.
At enrollment into the study, all people underwent screening spirometry and echocardiography. Their average baseline FEV1 was 2.9 L and their average FVC was 3.7 L, and 4% had heart failure based on assessments of left ventricular size and function by echocardiography. The first 5,000 enrollees also had measurements taken of their serum levels of N-terminal probrain natriuretic peptide and cardiac troponin I through use of a high-sensitivity assay. The researchers used data from patients followed for a median of 5.5 years.
During follow-up, people in the lowest tertile for FEV1 and those in the lowest tertile for FVC had higher rates of all-cause mortality, compared with those in the highest tertile for each of these two parameters.
In a multivariate analysis that adjusted for age, sex, body mass index, smoking status, hypertension, dyslipidemia, heart failure status, serum levels of N-terminal probrain natriuretic peptide and cardiac troponin I, and other parameters, people with lower FEV1 and FVC readings had significantly worse survival, Dr. Baum said. Every 1–standard deviation increase in FEV1 was linked with a statistically significant, 38% reduced mortality rate; furthermore, a similar significant inverse association existed between FVC and mortality, she reported.
On Twitter@mitchelzoler
ORLANDO – Unselected people from the general population without clinically apparent lung disease but with low lung function had significantly increased mortality during follow-up that was independent of cardiac function, in results from more than 13,000 middle-aged Germans.
“Subtle, subclinical pulmonary impairment is a risk indicator for increased mortality independent of cardiac performance,” Dr. Christina Baum said at the American Heart Association scientific sessions.
The researchers used spirometry to measure each subject’s forced expiratory volume in 1 second (FEV1) and forced vital capacity (FVC). The results showed that “spirometry is a good screening tool that is not very expensive,” making spirometry an effective risk assessment tool for use in the general adult population, said Dr. Baum of the department of general and interventional cardiology at the University Heart Center in Hamburg, Germany.
She and her associates used data collected in the Gutenberg Health Study, which enrolled more than 15,000 German women and men aged 35-74 years during 2007-2012. The investigators excluded people with a history of pulmonary disease, resulting in a study cohort of 13,191, who averaged 55 years old, with 51% men.
At enrollment into the study, all people underwent screening spirometry and echocardiography. Their average baseline FEV1 was 2.9 L and their average FVC was 3.7 L, and 4% had heart failure based on assessments of left ventricular size and function by echocardiography. The first 5,000 enrollees also had measurements taken of their serum levels of N-terminal probrain natriuretic peptide and cardiac troponin I through use of a high-sensitivity assay. The researchers used data from patients followed for a median of 5.5 years.
During follow-up, people in the lowest tertile for FEV1 and those in the lowest tertile for FVC had higher rates of all-cause mortality, compared with those in the highest tertile for each of these two parameters.
In a multivariate analysis that adjusted for age, sex, body mass index, smoking status, hypertension, dyslipidemia, heart failure status, serum levels of N-terminal probrain natriuretic peptide and cardiac troponin I, and other parameters, people with lower FEV1 and FVC readings had significantly worse survival, Dr. Baum said. Every 1–standard deviation increase in FEV1 was linked with a statistically significant, 38% reduced mortality rate; furthermore, a similar significant inverse association existed between FVC and mortality, she reported.
On Twitter@mitchelzoler
ORLANDO – Unselected people from the general population without clinically apparent lung disease but with low lung function had significantly increased mortality during follow-up that was independent of cardiac function, in results from more than 13,000 middle-aged Germans.
“Subtle, subclinical pulmonary impairment is a risk indicator for increased mortality independent of cardiac performance,” Dr. Christina Baum said at the American Heart Association scientific sessions.
The researchers used spirometry to measure each subject’s forced expiratory volume in 1 second (FEV1) and forced vital capacity (FVC). The results showed that “spirometry is a good screening tool that is not very expensive,” making spirometry an effective risk assessment tool for use in the general adult population, said Dr. Baum of the department of general and interventional cardiology at the University Heart Center in Hamburg, Germany.
She and her associates used data collected in the Gutenberg Health Study, which enrolled more than 15,000 German women and men aged 35-74 years during 2007-2012. The investigators excluded people with a history of pulmonary disease, resulting in a study cohort of 13,191, who averaged 55 years old, with 51% men.
At enrollment into the study, all people underwent screening spirometry and echocardiography. Their average baseline FEV1 was 2.9 L and their average FVC was 3.7 L, and 4% had heart failure based on assessments of left ventricular size and function by echocardiography. The first 5,000 enrollees also had measurements taken of their serum levels of N-terminal probrain natriuretic peptide and cardiac troponin I through use of a high-sensitivity assay. The researchers used data from patients followed for a median of 5.5 years.
During follow-up, people in the lowest tertile for FEV1 and those in the lowest tertile for FVC had higher rates of all-cause mortality, compared with those in the highest tertile for each of these two parameters.
In a multivariate analysis that adjusted for age, sex, body mass index, smoking status, hypertension, dyslipidemia, heart failure status, serum levels of N-terminal probrain natriuretic peptide and cardiac troponin I, and other parameters, people with lower FEV1 and FVC readings had significantly worse survival, Dr. Baum said. Every 1–standard deviation increase in FEV1 was linked with a statistically significant, 38% reduced mortality rate; furthermore, a similar significant inverse association existed between FVC and mortality, she reported.
On Twitter@mitchelzoler
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point: Measurement of low FEV1 or low FVC by spirometry identified people at increased mortality risk independent of their cardiac function.
Major finding: For each standard deviation rise in FEV1, mortality fell by 38%.
Data source: The Gutenberg Heart Study, which enrolled 15,010 German residents aged 35-74 years old, including 13,191 without prevalent pulmonary disease.
Disclosures: Dr. Baum had no relevant financial disclosures.
AHA: Empagliflozin for T2D reduces heart failure endpoints
ORLANDO – The SGLT2 inhibitor empagliflozin reduced the risk of the composite endpoint of hospitalization for heart failure or death due to cardiovascular disease by 34% in patients with type 2 diabetes and prior cardiovascular events in the landmark EMPA-REG OUTCOME trial, Dr. Silvio E. Inzucchi reported at the American Heart Association scientific sessions.
The benefit was consistent regardless of whether or not patients had heart failure at enrollment, added Dr. Inzucchi, professor of medicine and clinical director of the endocrinology section at Yale University in New Haven, Conn.
He presented a prespecified secondary analysis from EMPA-REG OUTCOME that focused on heart failure–related endpoints. He had previously presented the primary outcome in Stockholm at the annual meeting of the European Association for the Study of Diabetes: a 14% relative risk reduction for empagliflozin in the composite of cardiovascular death, MI, or stroke, compared with placebo, driven largely by a 38% relative risk reduction in cardiovascular death.
The phase III, randomized, double-blind study has taken the worlds of cardiology and endocrinology by storm because it has convincingly demonstrated that empagliflozin (Jardiance) is the first glucose-lowering agent that also prevents cardiovascular complications.
“There are 12 different types of medications for lowering blood sugar levels in patients with type 2 diabetes – more than there are for lowering high blood pressure,” he noted. “We have been searching for decades for a diabetes medicine that will not only lower blood sugar but also reduce cardiovascular complications.”
The EMPA-REG OUTCOME trial included 7,020 patients with type 2 diabetes and a history of cardiovascular disease who were followed for a median of 3.1 years. Participants had a high rate of background optimal medical therapy for secondary cardiovascular prevention. They were randomized to empagliflozin at 10 or 25 mg/day or placebo. Results with the two doses of empagliflozin were pooled because the outcomes were so similar.
In addition to the 34% relative risk reduction in the combined endpoint of heart failure hospitalization or heart failure death, the empagliflozin group also experienced a 39% reduction in the composite of heart failure hospitalization or death due to heart failure.
These advantages for empagliflozin held true for all prespecified patient subgroups, including those based upon age, renal function, use of insulin, and background cardioprotective medications, Dr. Inzucchi noted.
Of study participants, 10% already had heart failure at baseline. Their rate of heart failure hospitalization or cardiovascular death was 20.1% on placebo and 16.2% with empagliflozin, a 28% relative risk reduction. In patients without heart failure at enrollment, the rates of this composite endpoint were 7.1% with placebo and 4.5% with the SGLT2 (sodium-glucose transporter 2) inhibitor, for a 37% relative risk reduction.
The only adverse event that occurred more frequently in empagliflozin-treated patients with or without heart failure than in controls was a threefold increase in genital infections. These easily treatable infections are a consequence of empagliflozin’s mechanism of action in reducing blood glucose, which entails increasing urinary excretion, the endocrinologist explained.
Discussant Dr. Allison B. Goldfine called Dr. Inzucchi’s update “really exciting.”
“Up to this time there has been little within the diabetes therapeutic pharmacologic options that has established clear safety, specifically with regard to heart failure. While we may not fully understand the mechanism behind the observed cardiovascular risk reduction, the uniformity of the findings is really remarkably consistent,” said Dr. Goldfine, head of the section of endocrinology at the Joslin Diabetes Center and an endocrinologist at Harvard Medical School, Boston.
“This is very promising,” Dr. David Goff, dean of the Colorado School of Public Health, declared in an interview. “We’ve had a lot of disappointments in the field of cardiovascular disease prevention for patients with diabetes mellitus. I think this medication should change the way we take care of people with diabetes.”
Metformin is widely accepted within endocrinology as the first-line agent for the treatment of type 2 diabetes, Dr. Inzucchi said. But he added that cardiovascular disease is a major problem in patients with diabetes. Heart failure, for example, is present in more than one in five type 2 diabetic patients over age 65.
Asked if he believes the cardiovascular benefits seen with empagliflozin in EMPA-REG OUTCOME are likely to be due to a class effect for SGLT2 inhibitors, Dr. Inzucchi replied that it’s impossible to say, since empagliflozin’s mechanism of cardiovascular benefit is unknown.
“Large randomized trials with hard cardiovascular endpoints are ongoing for the other two SGLT2 inhibitors, canagliflozin and dapagliflozin. Results should be available in 2-3 years. Then we’ll know,” he said.
Dr. Inzucchi disclosed ties with Boehringer Ingelheim, Merck, Janssen, Novo Nordisk, Sanofi/Regeron, Intarcia, Lexicon, Paxel, Takeda, and Eli Lilly. He acknowledged CME funding to Yale University from Boehringer Ingelheim, Eli Lilly, Novo Nordisk, Abbot, Merck Sharp & Dohme, and Sanofi.
ORLANDO – The SGLT2 inhibitor empagliflozin reduced the risk of the composite endpoint of hospitalization for heart failure or death due to cardiovascular disease by 34% in patients with type 2 diabetes and prior cardiovascular events in the landmark EMPA-REG OUTCOME trial, Dr. Silvio E. Inzucchi reported at the American Heart Association scientific sessions.
The benefit was consistent regardless of whether or not patients had heart failure at enrollment, added Dr. Inzucchi, professor of medicine and clinical director of the endocrinology section at Yale University in New Haven, Conn.
He presented a prespecified secondary analysis from EMPA-REG OUTCOME that focused on heart failure–related endpoints. He had previously presented the primary outcome in Stockholm at the annual meeting of the European Association for the Study of Diabetes: a 14% relative risk reduction for empagliflozin in the composite of cardiovascular death, MI, or stroke, compared with placebo, driven largely by a 38% relative risk reduction in cardiovascular death.
The phase III, randomized, double-blind study has taken the worlds of cardiology and endocrinology by storm because it has convincingly demonstrated that empagliflozin (Jardiance) is the first glucose-lowering agent that also prevents cardiovascular complications.
“There are 12 different types of medications for lowering blood sugar levels in patients with type 2 diabetes – more than there are for lowering high blood pressure,” he noted. “We have been searching for decades for a diabetes medicine that will not only lower blood sugar but also reduce cardiovascular complications.”
The EMPA-REG OUTCOME trial included 7,020 patients with type 2 diabetes and a history of cardiovascular disease who were followed for a median of 3.1 years. Participants had a high rate of background optimal medical therapy for secondary cardiovascular prevention. They were randomized to empagliflozin at 10 or 25 mg/day or placebo. Results with the two doses of empagliflozin were pooled because the outcomes were so similar.
In addition to the 34% relative risk reduction in the combined endpoint of heart failure hospitalization or heart failure death, the empagliflozin group also experienced a 39% reduction in the composite of heart failure hospitalization or death due to heart failure.
These advantages for empagliflozin held true for all prespecified patient subgroups, including those based upon age, renal function, use of insulin, and background cardioprotective medications, Dr. Inzucchi noted.
Of study participants, 10% already had heart failure at baseline. Their rate of heart failure hospitalization or cardiovascular death was 20.1% on placebo and 16.2% with empagliflozin, a 28% relative risk reduction. In patients without heart failure at enrollment, the rates of this composite endpoint were 7.1% with placebo and 4.5% with the SGLT2 (sodium-glucose transporter 2) inhibitor, for a 37% relative risk reduction.
The only adverse event that occurred more frequently in empagliflozin-treated patients with or without heart failure than in controls was a threefold increase in genital infections. These easily treatable infections are a consequence of empagliflozin’s mechanism of action in reducing blood glucose, which entails increasing urinary excretion, the endocrinologist explained.
Discussant Dr. Allison B. Goldfine called Dr. Inzucchi’s update “really exciting.”
“Up to this time there has been little within the diabetes therapeutic pharmacologic options that has established clear safety, specifically with regard to heart failure. While we may not fully understand the mechanism behind the observed cardiovascular risk reduction, the uniformity of the findings is really remarkably consistent,” said Dr. Goldfine, head of the section of endocrinology at the Joslin Diabetes Center and an endocrinologist at Harvard Medical School, Boston.
“This is very promising,” Dr. David Goff, dean of the Colorado School of Public Health, declared in an interview. “We’ve had a lot of disappointments in the field of cardiovascular disease prevention for patients with diabetes mellitus. I think this medication should change the way we take care of people with diabetes.”
Metformin is widely accepted within endocrinology as the first-line agent for the treatment of type 2 diabetes, Dr. Inzucchi said. But he added that cardiovascular disease is a major problem in patients with diabetes. Heart failure, for example, is present in more than one in five type 2 diabetic patients over age 65.
Asked if he believes the cardiovascular benefits seen with empagliflozin in EMPA-REG OUTCOME are likely to be due to a class effect for SGLT2 inhibitors, Dr. Inzucchi replied that it’s impossible to say, since empagliflozin’s mechanism of cardiovascular benefit is unknown.
“Large randomized trials with hard cardiovascular endpoints are ongoing for the other two SGLT2 inhibitors, canagliflozin and dapagliflozin. Results should be available in 2-3 years. Then we’ll know,” he said.
Dr. Inzucchi disclosed ties with Boehringer Ingelheim, Merck, Janssen, Novo Nordisk, Sanofi/Regeron, Intarcia, Lexicon, Paxel, Takeda, and Eli Lilly. He acknowledged CME funding to Yale University from Boehringer Ingelheim, Eli Lilly, Novo Nordisk, Abbot, Merck Sharp & Dohme, and Sanofi.
ORLANDO – The SGLT2 inhibitor empagliflozin reduced the risk of the composite endpoint of hospitalization for heart failure or death due to cardiovascular disease by 34% in patients with type 2 diabetes and prior cardiovascular events in the landmark EMPA-REG OUTCOME trial, Dr. Silvio E. Inzucchi reported at the American Heart Association scientific sessions.
The benefit was consistent regardless of whether or not patients had heart failure at enrollment, added Dr. Inzucchi, professor of medicine and clinical director of the endocrinology section at Yale University in New Haven, Conn.
He presented a prespecified secondary analysis from EMPA-REG OUTCOME that focused on heart failure–related endpoints. He had previously presented the primary outcome in Stockholm at the annual meeting of the European Association for the Study of Diabetes: a 14% relative risk reduction for empagliflozin in the composite of cardiovascular death, MI, or stroke, compared with placebo, driven largely by a 38% relative risk reduction in cardiovascular death.
The phase III, randomized, double-blind study has taken the worlds of cardiology and endocrinology by storm because it has convincingly demonstrated that empagliflozin (Jardiance) is the first glucose-lowering agent that also prevents cardiovascular complications.
“There are 12 different types of medications for lowering blood sugar levels in patients with type 2 diabetes – more than there are for lowering high blood pressure,” he noted. “We have been searching for decades for a diabetes medicine that will not only lower blood sugar but also reduce cardiovascular complications.”
The EMPA-REG OUTCOME trial included 7,020 patients with type 2 diabetes and a history of cardiovascular disease who were followed for a median of 3.1 years. Participants had a high rate of background optimal medical therapy for secondary cardiovascular prevention. They were randomized to empagliflozin at 10 or 25 mg/day or placebo. Results with the two doses of empagliflozin were pooled because the outcomes were so similar.
In addition to the 34% relative risk reduction in the combined endpoint of heart failure hospitalization or heart failure death, the empagliflozin group also experienced a 39% reduction in the composite of heart failure hospitalization or death due to heart failure.
These advantages for empagliflozin held true for all prespecified patient subgroups, including those based upon age, renal function, use of insulin, and background cardioprotective medications, Dr. Inzucchi noted.
Of study participants, 10% already had heart failure at baseline. Their rate of heart failure hospitalization or cardiovascular death was 20.1% on placebo and 16.2% with empagliflozin, a 28% relative risk reduction. In patients without heart failure at enrollment, the rates of this composite endpoint were 7.1% with placebo and 4.5% with the SGLT2 (sodium-glucose transporter 2) inhibitor, for a 37% relative risk reduction.
The only adverse event that occurred more frequently in empagliflozin-treated patients with or without heart failure than in controls was a threefold increase in genital infections. These easily treatable infections are a consequence of empagliflozin’s mechanism of action in reducing blood glucose, which entails increasing urinary excretion, the endocrinologist explained.
Discussant Dr. Allison B. Goldfine called Dr. Inzucchi’s update “really exciting.”
“Up to this time there has been little within the diabetes therapeutic pharmacologic options that has established clear safety, specifically with regard to heart failure. While we may not fully understand the mechanism behind the observed cardiovascular risk reduction, the uniformity of the findings is really remarkably consistent,” said Dr. Goldfine, head of the section of endocrinology at the Joslin Diabetes Center and an endocrinologist at Harvard Medical School, Boston.
“This is very promising,” Dr. David Goff, dean of the Colorado School of Public Health, declared in an interview. “We’ve had a lot of disappointments in the field of cardiovascular disease prevention for patients with diabetes mellitus. I think this medication should change the way we take care of people with diabetes.”
Metformin is widely accepted within endocrinology as the first-line agent for the treatment of type 2 diabetes, Dr. Inzucchi said. But he added that cardiovascular disease is a major problem in patients with diabetes. Heart failure, for example, is present in more than one in five type 2 diabetic patients over age 65.
Asked if he believes the cardiovascular benefits seen with empagliflozin in EMPA-REG OUTCOME are likely to be due to a class effect for SGLT2 inhibitors, Dr. Inzucchi replied that it’s impossible to say, since empagliflozin’s mechanism of cardiovascular benefit is unknown.
“Large randomized trials with hard cardiovascular endpoints are ongoing for the other two SGLT2 inhibitors, canagliflozin and dapagliflozin. Results should be available in 2-3 years. Then we’ll know,” he said.
Dr. Inzucchi disclosed ties with Boehringer Ingelheim, Merck, Janssen, Novo Nordisk, Sanofi/Regeron, Intarcia, Lexicon, Paxel, Takeda, and Eli Lilly. He acknowledged CME funding to Yale University from Boehringer Ingelheim, Eli Lilly, Novo Nordisk, Abbot, Merck Sharp & Dohme, and Sanofi.
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point: Empagliflozin is the first glucose-lowering medication ever shown to reduce heart failure hospitalizations or cardiovascular death in patients with type 2 diabetes.
Major finding: The composite rate of heart failure hospitalization or death due to heart failure was reduced by 39% in type 2 diabetic patients on empagliflozin compared with placebo.
Data source: This was a secondary analysis of the EMPA-REG OUTCOME trial, a phase-III, double-blind, randomized trial of 7,020 patients with type 2 diabetes and prior cardiovascular disease.
Disclosures: The presenter disclosed ties with Boehringer Ingelheim, Merck, Janssen, Novo Nordisk, Sanofi/Regeron, Intarcia, Lexicon, Paxel, Takeda, and Eli Lilly. He acknowledged CME-funding to Yale University from Boehringer Ingelheim, Eli Lilly, Novo Nordisk, Abbot, Merck Sharp & Dohme, and Sanofi.
AHA: HFpEF, HFrEF cause similar acute hospitalization rates
ORLANDO – The number of Americans hospitalized for acute decompensated heart failure (ADHF) with preserved ejection fraction during 2003-2012 nearly equaled the number hospitalized with ADHF with reduced ejection fraction, in an analysis of more than 5 million hospitalized heart failure patients tracked in a national-sample database.
But the profile of patients hospitalized with ADHF with preserved ejection fraction (HFpEF) differed from patients hospitalized with acute heart failure and reduced ejection fraction (HFrEF), with a substantially higher percentage of women and patients aged 75 years or older, Dr. Parag Goyal said at the American Heart Association scientific sessions.
The analysis also showed the strongest correlate for in-hospital mortality among HFpEF patients hospitalized with acute decompensation was a pulmonary circulation disorder, such as pulmonary hypertension, which nearly doubled the rate of in-hospital death among HFpEF patients. Other strong correlates of mortality during hospitalization were liver disease, which was linked with about a 50% boost in hospitalized mortality; and chronic renal failure, which was tied to a roughly one-third higher mortality, said Dr. Goyal, a cardiologist at New York–Presbyterian Hospital.
His study used data collected by the Nationwide Inpatient Sample, which included data on more than 388 million hospitalized U.S. patients during 2003-2012, including 5,046,879 hospitalized with acute heart failure. This total included 2,329,391 patients (46%) diagnosed with HFpEF and 2,717,488 patients (54%) diagnosed with HFrEF.
The HFpEF patients’ average age was 76 years, with 60% at least 75 years old, while the HFrEF patients’ average age was 72 years, with 49% age 75 years or older. Nearly two-thirds of the HFpEF patients were women, compared with 42% in the HFrEF group. The HFrEF patients also had a substantially higher prevalence of coronary artery disease, 59%, compared with 41% in the HFpEF group. The prevalence of several comorbidities – including diabetes, hypertension, and chronic renal failure – were each roughly similar in both subgroups, but the obesity rate of 19% in the HFpEF patients substantially exceeded the 12% rate in HFrEF patients.
In-hospital mortality ran 4.3% in the HFpEF patients and 5.1% in the HFrEF patients, a 13% relative-risk reduction that was statistically significant. But average length of stay was similar between the two groups, about 7 days with either type of heart failure.
Dr. Goyal and his associates also examined time trends during 2003-2012. During this period, the percentage of patients with HFpEF aged 75 years or older rose from 57% to 60%. Even more notably, the percentage of men with HFpEF rose from 31% in 2003 to 37% in 2012. Furthermore, the reduced in-hospital mortality during the period was largely driven by mortality reductions among HFpEF patients aged 65 years or older. A multivariate analysis for significant correlates of in-hospital mortality identified age 75 years or older, male sex, and white race in both the HFpEF subgroup and in those with HFrEF. Older age had the highest impact, linked with about a 60% relatively higher mortality rate in patients with either type of heart failure.
The multivariate analysis also identified three comorbidities linked with in-hospital mortality. A pulmonary circulation disorder was associated with a 90% higher mortality rate among HFpEF patients and a 79% higher rate among those with HFrEF. Liver disease and chronic renal disease linked with smaller mortality increases for both heart failure types. The presence of treatable comorbidities, including hypertension, diabetes, and coronary artery disease, linked with significantly lower in-hospital mortality rates. Dr. Goyal speculated that the reduced mortality resulted from successful treatment of these conditions.
On Twitter @mitchelzoler
ORLANDO – The number of Americans hospitalized for acute decompensated heart failure (ADHF) with preserved ejection fraction during 2003-2012 nearly equaled the number hospitalized with ADHF with reduced ejection fraction, in an analysis of more than 5 million hospitalized heart failure patients tracked in a national-sample database.
But the profile of patients hospitalized with ADHF with preserved ejection fraction (HFpEF) differed from patients hospitalized with acute heart failure and reduced ejection fraction (HFrEF), with a substantially higher percentage of women and patients aged 75 years or older, Dr. Parag Goyal said at the American Heart Association scientific sessions.
The analysis also showed the strongest correlate for in-hospital mortality among HFpEF patients hospitalized with acute decompensation was a pulmonary circulation disorder, such as pulmonary hypertension, which nearly doubled the rate of in-hospital death among HFpEF patients. Other strong correlates of mortality during hospitalization were liver disease, which was linked with about a 50% boost in hospitalized mortality; and chronic renal failure, which was tied to a roughly one-third higher mortality, said Dr. Goyal, a cardiologist at New York–Presbyterian Hospital.
His study used data collected by the Nationwide Inpatient Sample, which included data on more than 388 million hospitalized U.S. patients during 2003-2012, including 5,046,879 hospitalized with acute heart failure. This total included 2,329,391 patients (46%) diagnosed with HFpEF and 2,717,488 patients (54%) diagnosed with HFrEF.
The HFpEF patients’ average age was 76 years, with 60% at least 75 years old, while the HFrEF patients’ average age was 72 years, with 49% age 75 years or older. Nearly two-thirds of the HFpEF patients were women, compared with 42% in the HFrEF group. The HFrEF patients also had a substantially higher prevalence of coronary artery disease, 59%, compared with 41% in the HFpEF group. The prevalence of several comorbidities – including diabetes, hypertension, and chronic renal failure – were each roughly similar in both subgroups, but the obesity rate of 19% in the HFpEF patients substantially exceeded the 12% rate in HFrEF patients.
In-hospital mortality ran 4.3% in the HFpEF patients and 5.1% in the HFrEF patients, a 13% relative-risk reduction that was statistically significant. But average length of stay was similar between the two groups, about 7 days with either type of heart failure.
Dr. Goyal and his associates also examined time trends during 2003-2012. During this period, the percentage of patients with HFpEF aged 75 years or older rose from 57% to 60%. Even more notably, the percentage of men with HFpEF rose from 31% in 2003 to 37% in 2012. Furthermore, the reduced in-hospital mortality during the period was largely driven by mortality reductions among HFpEF patients aged 65 years or older. A multivariate analysis for significant correlates of in-hospital mortality identified age 75 years or older, male sex, and white race in both the HFpEF subgroup and in those with HFrEF. Older age had the highest impact, linked with about a 60% relatively higher mortality rate in patients with either type of heart failure.
The multivariate analysis also identified three comorbidities linked with in-hospital mortality. A pulmonary circulation disorder was associated with a 90% higher mortality rate among HFpEF patients and a 79% higher rate among those with HFrEF. Liver disease and chronic renal disease linked with smaller mortality increases for both heart failure types. The presence of treatable comorbidities, including hypertension, diabetes, and coronary artery disease, linked with significantly lower in-hospital mortality rates. Dr. Goyal speculated that the reduced mortality resulted from successful treatment of these conditions.
On Twitter @mitchelzoler
ORLANDO – The number of Americans hospitalized for acute decompensated heart failure (ADHF) with preserved ejection fraction during 2003-2012 nearly equaled the number hospitalized with ADHF with reduced ejection fraction, in an analysis of more than 5 million hospitalized heart failure patients tracked in a national-sample database.
But the profile of patients hospitalized with ADHF with preserved ejection fraction (HFpEF) differed from patients hospitalized with acute heart failure and reduced ejection fraction (HFrEF), with a substantially higher percentage of women and patients aged 75 years or older, Dr. Parag Goyal said at the American Heart Association scientific sessions.
The analysis also showed the strongest correlate for in-hospital mortality among HFpEF patients hospitalized with acute decompensation was a pulmonary circulation disorder, such as pulmonary hypertension, which nearly doubled the rate of in-hospital death among HFpEF patients. Other strong correlates of mortality during hospitalization were liver disease, which was linked with about a 50% boost in hospitalized mortality; and chronic renal failure, which was tied to a roughly one-third higher mortality, said Dr. Goyal, a cardiologist at New York–Presbyterian Hospital.
His study used data collected by the Nationwide Inpatient Sample, which included data on more than 388 million hospitalized U.S. patients during 2003-2012, including 5,046,879 hospitalized with acute heart failure. This total included 2,329,391 patients (46%) diagnosed with HFpEF and 2,717,488 patients (54%) diagnosed with HFrEF.
The HFpEF patients’ average age was 76 years, with 60% at least 75 years old, while the HFrEF patients’ average age was 72 years, with 49% age 75 years or older. Nearly two-thirds of the HFpEF patients were women, compared with 42% in the HFrEF group. The HFrEF patients also had a substantially higher prevalence of coronary artery disease, 59%, compared with 41% in the HFpEF group. The prevalence of several comorbidities – including diabetes, hypertension, and chronic renal failure – were each roughly similar in both subgroups, but the obesity rate of 19% in the HFpEF patients substantially exceeded the 12% rate in HFrEF patients.
In-hospital mortality ran 4.3% in the HFpEF patients and 5.1% in the HFrEF patients, a 13% relative-risk reduction that was statistically significant. But average length of stay was similar between the two groups, about 7 days with either type of heart failure.
Dr. Goyal and his associates also examined time trends during 2003-2012. During this period, the percentage of patients with HFpEF aged 75 years or older rose from 57% to 60%. Even more notably, the percentage of men with HFpEF rose from 31% in 2003 to 37% in 2012. Furthermore, the reduced in-hospital mortality during the period was largely driven by mortality reductions among HFpEF patients aged 65 years or older. A multivariate analysis for significant correlates of in-hospital mortality identified age 75 years or older, male sex, and white race in both the HFpEF subgroup and in those with HFrEF. Older age had the highest impact, linked with about a 60% relatively higher mortality rate in patients with either type of heart failure.
The multivariate analysis also identified three comorbidities linked with in-hospital mortality. A pulmonary circulation disorder was associated with a 90% higher mortality rate among HFpEF patients and a 79% higher rate among those with HFrEF. Liver disease and chronic renal disease linked with smaller mortality increases for both heart failure types. The presence of treatable comorbidities, including hypertension, diabetes, and coronary artery disease, linked with significantly lower in-hospital mortality rates. Dr. Goyal speculated that the reduced mortality resulted from successful treatment of these conditions.
On Twitter @mitchelzoler
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point: Heart failure with preserved ejection fraction closely tracked to heart failure with reduced ejection fraction for causing U.S. heart failure hospitalizations.
Major finding: Among U.S. heart failure patients hospitalized during 2003-2012, 46% had preserved ejection fraction and 54% had reduced ejection fraction.
Data source: Retrospective analysis of 5 million U.S. patients hospitalized for heart failure during 2003-2012 and included in the Nationwide Inpatient Sample.
Disclosures: Dr. Goyal had no disclosures.
VIDEO: Monitoring helps only adherent heart failure patients
ORLANDO – A multipronged approach to following and managing heart failure patients closely after they are hospitalized for acute decompensation led to significant reductions in subsequent rehospitalization or death in a randomized trial, but only in the subgroup of patients who actually adhered to the program.
The main message from the study was “this type of telemonitoring should not get used on everyone,” said Dr. Michael K. Ong in an interview at the American Heart Association scientific sessions. “A key issue is who are the people who would benefit” from an intensified at-home monitoring program following hospitalization for an acute heart failure episode.
Another issue is that new monitoring technologies introduced after launch of the BEAT-HF (Better Effectiveness After Transition–Heart Failure) trial more than 4 years ago have produced unobtrusive and implantable monitoring devices that could help boost monitoring compliance, said Dr. Ong, an internist at the University of California, Los Angeles.
“We all monitor our patients remotely on a variety of ways,” commented Dr. Mariell Jessup, professor of medicine and heart failure specialist and at the University of Pennsylvania in Philadelphia. “Depending on our resources and technology, we might use implantable monitors or have nurses call patients, and have patients send us emails. There is a wide range of telemonitoring available. But we need to find out what works. An enormous effort has been made to enhance patients’ ability to monitor themselves, so they can take charge of their disease,” Dr. Jessup said.
BEAT-HF randomized 1,437 patients with confirmed heart failure and an index hospitalization to an intensive monitoring and education program or usual care during 2011-2013 at six academic health centers in California. Patients averaged 73 years old, and most patients had class III New York Heart Association heart failure, with three quarters having either class III or IV.
The intensive program included three elements:
• An in-hospital education program.
• A schedule of nine follow-up telephone calls by a registered nurse starting 2-3 days post discharge and continuing out to 6 months. Patients in the intervention arm completed a median of six of these calls.
• Telemonitoring of daily measurement of weight, blood pressure, and heart rate using electronically linked monitoring devices supplied to each patient. The monitoring equipment actually was used by 83% of the 715 patients randomized to this arm, and at 180 days, 52% of the patients in this arm had transmitted more than half of their daily measurement updates.
The study showed no significant benefit from the intensive monitoring arm compared with usual care for the primary endpoint of all-cause hospitalizations after 180 days, Dr. Ong reported. However, in a post hoc analysis that divided the intervention arm patients into those with more than 50% days with monitoring information sent and those with 50% or less, the rehospitalization rate was 61% among the patients who complied 50% or less of the time with daily home monitoring, and 41% in patients with greater than 50% compliance, a one-third relative drop. The more-compliant patients also substantially and significantly reduced their mortality rates at both 30 and 180 days, compared with the less-adherent patients in the intervention arm.
Additional studies must now examine how to optimize adherence and better match patients with various monitoring techniques. “If patients won’t use a treatment, they won’t benefit,” said Dr. Ong. Finding out what makes people adherent and encourage them to participate is the next research issue, he added.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @mitchelzoler
We are now in the second decade of research aimed at finding the most effective ways to monitor patients following a hospitalization for acute heart failure decompensation. Interest runs high to find the best ways to monitor these patients and treat them with interventions intended to cut the rate of future decompensation events and hospitalizations and mortality.
Despite all this, research and interest studies have not yet clearly identified monitoring and intervention strategies that are consistently effective. In fact, sometimes so many monitoring strategies are begun by both health systems and by payers that it can become can become confusing.

|
Dr. Mary Norine Walsh |
A key issue is, who receives the monitoring data and what do they do with it? The way that physicians and nurses act on monitoring data really matters, and ideally, patients should also know their monitoring data and be an active part of maintaining their stability.
At the center where I work, we routinely educate patients during their hospitalization on the importance of maintaining a low-sodium diet and daily weight monitoring. Daily weights as a way to track the fluid-balance status of patients has been unfairly criticized, as new technology has made implantable monitors routinely available. Although they are routinely available, implanted technologies are not yet for the masses. I am a firm believer in the value of daily weights.
At my center, we put a paper weight chart in each patient’s room, recorded in pounds, so that patients can track their weight fluctuations themselves. We try to educate and indoctrinate our heart failure patients to the importance of tracking their weight, and tell them to bring the charts they maintain at home to their clinic visits. We even instruct selected patients who have taken good, personal control of their heart failure to adjust their daily furosemide dosage themselves – within specified limits and while keeping us informed – when they see their weight tracking up or down.
The better patients with heart failure understand the tight relationship between their lifestyle choices and their status, the better it is for their long-term success.
Dr. Mary Norine Walsh is medical director of the Heart Failure and Cardiac Transplantation program at the St. Vincent Heart Center in Indianapolis. She had no disclosures. She made these comments in an interview.
We are now in the second decade of research aimed at finding the most effective ways to monitor patients following a hospitalization for acute heart failure decompensation. Interest runs high to find the best ways to monitor these patients and treat them with interventions intended to cut the rate of future decompensation events and hospitalizations and mortality.
Despite all this, research and interest studies have not yet clearly identified monitoring and intervention strategies that are consistently effective. In fact, sometimes so many monitoring strategies are begun by both health systems and by payers that it can become can become confusing.

|
Dr. Mary Norine Walsh |
A key issue is, who receives the monitoring data and what do they do with it? The way that physicians and nurses act on monitoring data really matters, and ideally, patients should also know their monitoring data and be an active part of maintaining their stability.
At the center where I work, we routinely educate patients during their hospitalization on the importance of maintaining a low-sodium diet and daily weight monitoring. Daily weights as a way to track the fluid-balance status of patients has been unfairly criticized, as new technology has made implantable monitors routinely available. Although they are routinely available, implanted technologies are not yet for the masses. I am a firm believer in the value of daily weights.
At my center, we put a paper weight chart in each patient’s room, recorded in pounds, so that patients can track their weight fluctuations themselves. We try to educate and indoctrinate our heart failure patients to the importance of tracking their weight, and tell them to bring the charts they maintain at home to their clinic visits. We even instruct selected patients who have taken good, personal control of their heart failure to adjust their daily furosemide dosage themselves – within specified limits and while keeping us informed – when they see their weight tracking up or down.
The better patients with heart failure understand the tight relationship between their lifestyle choices and their status, the better it is for their long-term success.
Dr. Mary Norine Walsh is medical director of the Heart Failure and Cardiac Transplantation program at the St. Vincent Heart Center in Indianapolis. She had no disclosures. She made these comments in an interview.
We are now in the second decade of research aimed at finding the most effective ways to monitor patients following a hospitalization for acute heart failure decompensation. Interest runs high to find the best ways to monitor these patients and treat them with interventions intended to cut the rate of future decompensation events and hospitalizations and mortality.
Despite all this, research and interest studies have not yet clearly identified monitoring and intervention strategies that are consistently effective. In fact, sometimes so many monitoring strategies are begun by both health systems and by payers that it can become can become confusing.

|
Dr. Mary Norine Walsh |
A key issue is, who receives the monitoring data and what do they do with it? The way that physicians and nurses act on monitoring data really matters, and ideally, patients should also know their monitoring data and be an active part of maintaining their stability.
At the center where I work, we routinely educate patients during their hospitalization on the importance of maintaining a low-sodium diet and daily weight monitoring. Daily weights as a way to track the fluid-balance status of patients has been unfairly criticized, as new technology has made implantable monitors routinely available. Although they are routinely available, implanted technologies are not yet for the masses. I am a firm believer in the value of daily weights.
At my center, we put a paper weight chart in each patient’s room, recorded in pounds, so that patients can track their weight fluctuations themselves. We try to educate and indoctrinate our heart failure patients to the importance of tracking their weight, and tell them to bring the charts they maintain at home to their clinic visits. We even instruct selected patients who have taken good, personal control of their heart failure to adjust their daily furosemide dosage themselves – within specified limits and while keeping us informed – when they see their weight tracking up or down.
The better patients with heart failure understand the tight relationship between their lifestyle choices and their status, the better it is for their long-term success.
Dr. Mary Norine Walsh is medical director of the Heart Failure and Cardiac Transplantation program at the St. Vincent Heart Center in Indianapolis. She had no disclosures. She made these comments in an interview.
ORLANDO – A multipronged approach to following and managing heart failure patients closely after they are hospitalized for acute decompensation led to significant reductions in subsequent rehospitalization or death in a randomized trial, but only in the subgroup of patients who actually adhered to the program.
The main message from the study was “this type of telemonitoring should not get used on everyone,” said Dr. Michael K. Ong in an interview at the American Heart Association scientific sessions. “A key issue is who are the people who would benefit” from an intensified at-home monitoring program following hospitalization for an acute heart failure episode.
Another issue is that new monitoring technologies introduced after launch of the BEAT-HF (Better Effectiveness After Transition–Heart Failure) trial more than 4 years ago have produced unobtrusive and implantable monitoring devices that could help boost monitoring compliance, said Dr. Ong, an internist at the University of California, Los Angeles.
“We all monitor our patients remotely on a variety of ways,” commented Dr. Mariell Jessup, professor of medicine and heart failure specialist and at the University of Pennsylvania in Philadelphia. “Depending on our resources and technology, we might use implantable monitors or have nurses call patients, and have patients send us emails. There is a wide range of telemonitoring available. But we need to find out what works. An enormous effort has been made to enhance patients’ ability to monitor themselves, so they can take charge of their disease,” Dr. Jessup said.
BEAT-HF randomized 1,437 patients with confirmed heart failure and an index hospitalization to an intensive monitoring and education program or usual care during 2011-2013 at six academic health centers in California. Patients averaged 73 years old, and most patients had class III New York Heart Association heart failure, with three quarters having either class III or IV.
The intensive program included three elements:
• An in-hospital education program.
• A schedule of nine follow-up telephone calls by a registered nurse starting 2-3 days post discharge and continuing out to 6 months. Patients in the intervention arm completed a median of six of these calls.
• Telemonitoring of daily measurement of weight, blood pressure, and heart rate using electronically linked monitoring devices supplied to each patient. The monitoring equipment actually was used by 83% of the 715 patients randomized to this arm, and at 180 days, 52% of the patients in this arm had transmitted more than half of their daily measurement updates.
The study showed no significant benefit from the intensive monitoring arm compared with usual care for the primary endpoint of all-cause hospitalizations after 180 days, Dr. Ong reported. However, in a post hoc analysis that divided the intervention arm patients into those with more than 50% days with monitoring information sent and those with 50% or less, the rehospitalization rate was 61% among the patients who complied 50% or less of the time with daily home monitoring, and 41% in patients with greater than 50% compliance, a one-third relative drop. The more-compliant patients also substantially and significantly reduced their mortality rates at both 30 and 180 days, compared with the less-adherent patients in the intervention arm.
Additional studies must now examine how to optimize adherence and better match patients with various monitoring techniques. “If patients won’t use a treatment, they won’t benefit,” said Dr. Ong. Finding out what makes people adherent and encourage them to participate is the next research issue, he added.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @mitchelzoler
ORLANDO – A multipronged approach to following and managing heart failure patients closely after they are hospitalized for acute decompensation led to significant reductions in subsequent rehospitalization or death in a randomized trial, but only in the subgroup of patients who actually adhered to the program.
The main message from the study was “this type of telemonitoring should not get used on everyone,” said Dr. Michael K. Ong in an interview at the American Heart Association scientific sessions. “A key issue is who are the people who would benefit” from an intensified at-home monitoring program following hospitalization for an acute heart failure episode.
Another issue is that new monitoring technologies introduced after launch of the BEAT-HF (Better Effectiveness After Transition–Heart Failure) trial more than 4 years ago have produced unobtrusive and implantable monitoring devices that could help boost monitoring compliance, said Dr. Ong, an internist at the University of California, Los Angeles.
“We all monitor our patients remotely on a variety of ways,” commented Dr. Mariell Jessup, professor of medicine and heart failure specialist and at the University of Pennsylvania in Philadelphia. “Depending on our resources and technology, we might use implantable monitors or have nurses call patients, and have patients send us emails. There is a wide range of telemonitoring available. But we need to find out what works. An enormous effort has been made to enhance patients’ ability to monitor themselves, so they can take charge of their disease,” Dr. Jessup said.
BEAT-HF randomized 1,437 patients with confirmed heart failure and an index hospitalization to an intensive monitoring and education program or usual care during 2011-2013 at six academic health centers in California. Patients averaged 73 years old, and most patients had class III New York Heart Association heart failure, with three quarters having either class III or IV.
The intensive program included three elements:
• An in-hospital education program.
• A schedule of nine follow-up telephone calls by a registered nurse starting 2-3 days post discharge and continuing out to 6 months. Patients in the intervention arm completed a median of six of these calls.
• Telemonitoring of daily measurement of weight, blood pressure, and heart rate using electronically linked monitoring devices supplied to each patient. The monitoring equipment actually was used by 83% of the 715 patients randomized to this arm, and at 180 days, 52% of the patients in this arm had transmitted more than half of their daily measurement updates.
The study showed no significant benefit from the intensive monitoring arm compared with usual care for the primary endpoint of all-cause hospitalizations after 180 days, Dr. Ong reported. However, in a post hoc analysis that divided the intervention arm patients into those with more than 50% days with monitoring information sent and those with 50% or less, the rehospitalization rate was 61% among the patients who complied 50% or less of the time with daily home monitoring, and 41% in patients with greater than 50% compliance, a one-third relative drop. The more-compliant patients also substantially and significantly reduced their mortality rates at both 30 and 180 days, compared with the less-adherent patients in the intervention arm.
Additional studies must now examine how to optimize adherence and better match patients with various monitoring techniques. “If patients won’t use a treatment, they won’t benefit,” said Dr. Ong. Finding out what makes people adherent and encourage them to participate is the next research issue, he added.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @mitchelzoler
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point: Remote monitoring failed to reduce mortality or hospitalizations for all patients during the 180 days following heart failure hospitalization, but was effective for patients who adhered to the program.
Major finding: Telemonitoring more than half the time cut 180-day readmissions by a third relative to usual care.
Data source: The BEAT-HF study, which enrolled 1,437 patients hospitalized for acute heart failure at six California centers.
Disclosures: BEAT-HF had no commercial sponsors. Dr. Ong had no disclosures. Dr. Jessup had no disclosures.
AHA: Sacubitril/valsartan cuts heart failure hospital readmissions
ORLANDO – The combined formulation of sacubitril and valsartan substantially cut the rate of 30-day heart failure rehospitalizations, trimming the control rate by 38% in an analysis of data from the PARADIGM-HF trial, Dr. Scott D. Solomon reported at the American Heart Association scientific sessions.
This is an especially meaningful additional benefit for heart failure patients who take sacubitril/valsartan (Entresto) in place of enalapril or similar drugs because heart failure rehospitalizations have become a closely tracked metric for U.S. hospitals.
The sacubitril/valsartan combination received Food and Drug Administration approval last summer for treating chronic heart failure with reduced ejection fraction on the strength of results from PARADIGM-HF, which showed the two-drug combination substantially cut the rate of cardiovascular death and heart failure hospitalizations, compared with enalapril (N Engl J Med. 2014 Sep 11;371:993-1004).
“The data suggest that chronic heart failure patients treated with sacubitril/valsartan relative to enalapril are less likely to be initially hospitalized, and subsequent to discharge are less likely to return to the hospital within 30 days, thereby reducing the risk to patients and the potential financial burden to the health care system,” said Dr. Solomon, professor of medicine at Harvard Medical School and director of noninvasive cardiology at Brigham and Women’s Hospital in Boston.
This finding may help spur faster adoption of sacubitril/valsartan as the top drug for treating the renin-angiotensin-aldosterone system in heart failure patients, commented Dr. Adrian F. Hernandez, professor and heart failure specialist at Duke University in Durham, N.C. “The fact that you can derive an early clinical benefit” that becomes an early financial benefit should help counter the higher cost for sacubitril/valsartan, compared with generic ACE inhibitors and angiotensin-receptor blockers, he said in an interview. Health system administrators “face an issue when they can only look at long-term horizons. But data like these, with the early benefit of reduced readmissions” make it easier to justify paying a higher drug cost. Health care systems increasingly focus on treatments that can produce rapid benefits, both clinically and financially, said Dr. Hernandez, director of health services and outcomes research at Duke.
In fact, a cost-effectiveness analysis of sacubitril/valsartan treatment in PARADIGM-HF that included the hospital readmissions data showed that the combined formulation was “highly cost effective,” compared with enalapril, said Dr. Solomon, who added that he and his associates will have a full report on this in 2016.
“Not only does sacubitril/valsartan reduce mortality and hospital admissions, but it also reduced readmissions. That is very exciting. This is one of the few treatments to have this effect”, commented Dr. Jennifer Thibodeau, medical director of the heart failure disease management program at the University of Texas Southwestern Medical Center in Dallas. The 38% reduction in total heart failure readmissions, compared with enalapril, and the 44% reduction in number of patients with a 30-day readmission was “pretty good,” she said in an interview. “Anything that could reduce readmissions that much is pretty good.” Plus, clinicians have already been quite excited about sacubitril/valsartan based on the primary-endpoint benefits it showed in PARADIGM-HF, “although there is always caution when a drug is brand new,” she added.
Since U.S. marketing for sacubitril/valsartan began last summer, “there has not been a big rush to adopt it,” primarily out of the usual concerns about new agents. “As we continue to see findings like these [reduced readmissions], there will be [substantial] adoption of this drug. The new findings definitely add to its attraction.” Dr. Thibodeau said.
The two subgroups of patients who had heart failure hospitalizations in PARADIGM-HF, the 675 patients in the sacubitril/valsartan arm and the 775 in the enalapril arm, closely matched each other for virtually all demographic and clinical parameters aside from history of atrial fibrillation, which was significantly more common in the enalapril patients. Even though these two subgroups had not been randomized, the near uniform consistency of their profiles made this “a valid analysis,” Dr. Solomon said. Overall, 20% of the PARADIGM-HF patients who had a heart failure hospitalization had a rehospitalization for any cause within 30 days.
The 30-day heart failure readmission rate was 10% among patients on sacubitril/valsartan and 13% among those on enalapril, a 38% relative risk reduction that was statistically significant. The number of patients with a heart failure readmission was 44% lower in the group on the combined formulation. After 60 days, readmissions for any cause were 23% lower in the sacubitril/valsartan arm, compared with enalapril, and the combined formulation dropped the number with any 60-day readmission by 30%, he reported.
Sacubitril/valsartan patients also had significantly fewer 30-day rehospitalizations of any kind, compared with the enalapril patients, whether based on investigator-reported rehospitalizations, first rehospitalizations only, or rehospitalizations confirmed by a clinical evaluation committee. Adjustments for baseline characteristics also did not affect the findings.
PARADIGM-HF was sponsored by Novartis, the company marketing sacubitril/valsartan (Entresto). Dr. Solomon has been a consultant to and has received research support from Novartis. Dr. Hernandez has received honoraria and research support from Novartis and from several other companies. Dr. Thibodeau had no financial disclosures.
On Twitter @mitchelzoler
ORLANDO – The combined formulation of sacubitril and valsartan substantially cut the rate of 30-day heart failure rehospitalizations, trimming the control rate by 38% in an analysis of data from the PARADIGM-HF trial, Dr. Scott D. Solomon reported at the American Heart Association scientific sessions.
This is an especially meaningful additional benefit for heart failure patients who take sacubitril/valsartan (Entresto) in place of enalapril or similar drugs because heart failure rehospitalizations have become a closely tracked metric for U.S. hospitals.
The sacubitril/valsartan combination received Food and Drug Administration approval last summer for treating chronic heart failure with reduced ejection fraction on the strength of results from PARADIGM-HF, which showed the two-drug combination substantially cut the rate of cardiovascular death and heart failure hospitalizations, compared with enalapril (N Engl J Med. 2014 Sep 11;371:993-1004).
“The data suggest that chronic heart failure patients treated with sacubitril/valsartan relative to enalapril are less likely to be initially hospitalized, and subsequent to discharge are less likely to return to the hospital within 30 days, thereby reducing the risk to patients and the potential financial burden to the health care system,” said Dr. Solomon, professor of medicine at Harvard Medical School and director of noninvasive cardiology at Brigham and Women’s Hospital in Boston.
This finding may help spur faster adoption of sacubitril/valsartan as the top drug for treating the renin-angiotensin-aldosterone system in heart failure patients, commented Dr. Adrian F. Hernandez, professor and heart failure specialist at Duke University in Durham, N.C. “The fact that you can derive an early clinical benefit” that becomes an early financial benefit should help counter the higher cost for sacubitril/valsartan, compared with generic ACE inhibitors and angiotensin-receptor blockers, he said in an interview. Health system administrators “face an issue when they can only look at long-term horizons. But data like these, with the early benefit of reduced readmissions” make it easier to justify paying a higher drug cost. Health care systems increasingly focus on treatments that can produce rapid benefits, both clinically and financially, said Dr. Hernandez, director of health services and outcomes research at Duke.
In fact, a cost-effectiveness analysis of sacubitril/valsartan treatment in PARADIGM-HF that included the hospital readmissions data showed that the combined formulation was “highly cost effective,” compared with enalapril, said Dr. Solomon, who added that he and his associates will have a full report on this in 2016.
“Not only does sacubitril/valsartan reduce mortality and hospital admissions, but it also reduced readmissions. That is very exciting. This is one of the few treatments to have this effect”, commented Dr. Jennifer Thibodeau, medical director of the heart failure disease management program at the University of Texas Southwestern Medical Center in Dallas. The 38% reduction in total heart failure readmissions, compared with enalapril, and the 44% reduction in number of patients with a 30-day readmission was “pretty good,” she said in an interview. “Anything that could reduce readmissions that much is pretty good.” Plus, clinicians have already been quite excited about sacubitril/valsartan based on the primary-endpoint benefits it showed in PARADIGM-HF, “although there is always caution when a drug is brand new,” she added.
Since U.S. marketing for sacubitril/valsartan began last summer, “there has not been a big rush to adopt it,” primarily out of the usual concerns about new agents. “As we continue to see findings like these [reduced readmissions], there will be [substantial] adoption of this drug. The new findings definitely add to its attraction.” Dr. Thibodeau said.
The two subgroups of patients who had heart failure hospitalizations in PARADIGM-HF, the 675 patients in the sacubitril/valsartan arm and the 775 in the enalapril arm, closely matched each other for virtually all demographic and clinical parameters aside from history of atrial fibrillation, which was significantly more common in the enalapril patients. Even though these two subgroups had not been randomized, the near uniform consistency of their profiles made this “a valid analysis,” Dr. Solomon said. Overall, 20% of the PARADIGM-HF patients who had a heart failure hospitalization had a rehospitalization for any cause within 30 days.
The 30-day heart failure readmission rate was 10% among patients on sacubitril/valsartan and 13% among those on enalapril, a 38% relative risk reduction that was statistically significant. The number of patients with a heart failure readmission was 44% lower in the group on the combined formulation. After 60 days, readmissions for any cause were 23% lower in the sacubitril/valsartan arm, compared with enalapril, and the combined formulation dropped the number with any 60-day readmission by 30%, he reported.
Sacubitril/valsartan patients also had significantly fewer 30-day rehospitalizations of any kind, compared with the enalapril patients, whether based on investigator-reported rehospitalizations, first rehospitalizations only, or rehospitalizations confirmed by a clinical evaluation committee. Adjustments for baseline characteristics also did not affect the findings.
PARADIGM-HF was sponsored by Novartis, the company marketing sacubitril/valsartan (Entresto). Dr. Solomon has been a consultant to and has received research support from Novartis. Dr. Hernandez has received honoraria and research support from Novartis and from several other companies. Dr. Thibodeau had no financial disclosures.
On Twitter @mitchelzoler
ORLANDO – The combined formulation of sacubitril and valsartan substantially cut the rate of 30-day heart failure rehospitalizations, trimming the control rate by 38% in an analysis of data from the PARADIGM-HF trial, Dr. Scott D. Solomon reported at the American Heart Association scientific sessions.
This is an especially meaningful additional benefit for heart failure patients who take sacubitril/valsartan (Entresto) in place of enalapril or similar drugs because heart failure rehospitalizations have become a closely tracked metric for U.S. hospitals.
The sacubitril/valsartan combination received Food and Drug Administration approval last summer for treating chronic heart failure with reduced ejection fraction on the strength of results from PARADIGM-HF, which showed the two-drug combination substantially cut the rate of cardiovascular death and heart failure hospitalizations, compared with enalapril (N Engl J Med. 2014 Sep 11;371:993-1004).
“The data suggest that chronic heart failure patients treated with sacubitril/valsartan relative to enalapril are less likely to be initially hospitalized, and subsequent to discharge are less likely to return to the hospital within 30 days, thereby reducing the risk to patients and the potential financial burden to the health care system,” said Dr. Solomon, professor of medicine at Harvard Medical School and director of noninvasive cardiology at Brigham and Women’s Hospital in Boston.
This finding may help spur faster adoption of sacubitril/valsartan as the top drug for treating the renin-angiotensin-aldosterone system in heart failure patients, commented Dr. Adrian F. Hernandez, professor and heart failure specialist at Duke University in Durham, N.C. “The fact that you can derive an early clinical benefit” that becomes an early financial benefit should help counter the higher cost for sacubitril/valsartan, compared with generic ACE inhibitors and angiotensin-receptor blockers, he said in an interview. Health system administrators “face an issue when they can only look at long-term horizons. But data like these, with the early benefit of reduced readmissions” make it easier to justify paying a higher drug cost. Health care systems increasingly focus on treatments that can produce rapid benefits, both clinically and financially, said Dr. Hernandez, director of health services and outcomes research at Duke.
In fact, a cost-effectiveness analysis of sacubitril/valsartan treatment in PARADIGM-HF that included the hospital readmissions data showed that the combined formulation was “highly cost effective,” compared with enalapril, said Dr. Solomon, who added that he and his associates will have a full report on this in 2016.
“Not only does sacubitril/valsartan reduce mortality and hospital admissions, but it also reduced readmissions. That is very exciting. This is one of the few treatments to have this effect”, commented Dr. Jennifer Thibodeau, medical director of the heart failure disease management program at the University of Texas Southwestern Medical Center in Dallas. The 38% reduction in total heart failure readmissions, compared with enalapril, and the 44% reduction in number of patients with a 30-day readmission was “pretty good,” she said in an interview. “Anything that could reduce readmissions that much is pretty good.” Plus, clinicians have already been quite excited about sacubitril/valsartan based on the primary-endpoint benefits it showed in PARADIGM-HF, “although there is always caution when a drug is brand new,” she added.
Since U.S. marketing for sacubitril/valsartan began last summer, “there has not been a big rush to adopt it,” primarily out of the usual concerns about new agents. “As we continue to see findings like these [reduced readmissions], there will be [substantial] adoption of this drug. The new findings definitely add to its attraction.” Dr. Thibodeau said.
The two subgroups of patients who had heart failure hospitalizations in PARADIGM-HF, the 675 patients in the sacubitril/valsartan arm and the 775 in the enalapril arm, closely matched each other for virtually all demographic and clinical parameters aside from history of atrial fibrillation, which was significantly more common in the enalapril patients. Even though these two subgroups had not been randomized, the near uniform consistency of their profiles made this “a valid analysis,” Dr. Solomon said. Overall, 20% of the PARADIGM-HF patients who had a heart failure hospitalization had a rehospitalization for any cause within 30 days.
The 30-day heart failure readmission rate was 10% among patients on sacubitril/valsartan and 13% among those on enalapril, a 38% relative risk reduction that was statistically significant. The number of patients with a heart failure readmission was 44% lower in the group on the combined formulation. After 60 days, readmissions for any cause were 23% lower in the sacubitril/valsartan arm, compared with enalapril, and the combined formulation dropped the number with any 60-day readmission by 30%, he reported.
Sacubitril/valsartan patients also had significantly fewer 30-day rehospitalizations of any kind, compared with the enalapril patients, whether based on investigator-reported rehospitalizations, first rehospitalizations only, or rehospitalizations confirmed by a clinical evaluation committee. Adjustments for baseline characteristics also did not affect the findings.
PARADIGM-HF was sponsored by Novartis, the company marketing sacubitril/valsartan (Entresto). Dr. Solomon has been a consultant to and has received research support from Novartis. Dr. Hernandez has received honoraria and research support from Novartis and from several other companies. Dr. Thibodeau had no financial disclosures.
On Twitter @mitchelzoler
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point: In the PARADIGM-HF pivotal trial, treating chronic heart failure with reduced ejection fraction with sacubitril/valsartan cut 30-day hospital readmissions, compared with enalapril treatment.
Major finding: Thirty-day heart-failure rehospitalizations occurred in 10% of sacubitril/valsartan patients and 13% of those on enalapril.
Data source: PARADIGM-HF, a multicenter, international randomized, pivotal trial with 4,187 patients.
Disclosures: PARADIGM-HF was sponsored by Novartis, the company marketing sacubitril/valsartan (Entresto). Dr. Solomon has been a consultant to and has received research support from Novartis. Dr. Hernandez has received honoraria and research support from Novartis and from several other companies. Dr. Thibodeau had no financial disclosures.
AHA: Candesartan protects against cardiotoxicity in breast cancer patients in PRADA
ORLANDO – Concomitant treatment with candesartan protects against the early decline in left ventricular ejection fraction associated with adjunct therapy for early breast cancer.
That was the key finding in the PRADA trial (PRevention of cArdiac Dysfunction during Adjuvant breast cancer therapy), the largest study to date looking at prevention of cardiac dysfunction in a breast cancer population.
Another important finding in PRADA was that unlike the angiotensin receptor blocker candesartan (Atacand), metoprolol, a beta blocker, didn’t prevent the early drop in LVEF commonly seen in breast cancer patients treated with anthracyclines and trastuzumab (Herceptin), even though both classes of heart medications are cornerstones of the treatment of ischemic and hypertensive cardiomyopathy, Dr. Geeta Gulati reported at the American Heart Association scientific sessions.
Although the cardiotoxicity of certain breast cancer treatments is widely recognized and has spawned the emerging field of cardio-oncology, the literature in this area is weak. Indeed, a recent meta-analysis identified only four published randomized studies evaluating the possible cardioprotective role for beta blockers and angiotensin antagonists in patients undergoing anthracycline-based chemotherapy (Postgrad Med J doi:10.1136/ postgradmedj-2015-133535). None of the studies was double-blind, all relied upon echocardiographic assessment of changes in LVEF rather than gold-standard cardiac MRI, and the study sizes were small -- just 18-45 breast cancer patients.
Most problematic of all, the studies employed a variety of different definitions of cardiotoxicity, noted Dr. Gulati of Akershus University Hospital in Lorenskog, Norway.
In contrast, PRADA was a double-blind, placebo-controlled, 2 by 2 factorial design, single-center trial, which included 120 patients with early breast cancer. Participants were randomized to candesartan at a starting dose of 8 mg and target dose of 32 mg/day, metoprolol starting at 25 mg with a target of 100 mg/day, or placebo after breast cancer surgery but before the start of anthracycline-containing chemotherapy.
The primary endpoint was change in LVEF from baseline to completion of adjuvant therapy, a period as short as 10 weeks and as long as 64 weeks depending upon whether a woman also underwent courses of trastuzumab, taxanes, and/or radiation therapy.
The overall decline in LVEF was 2.6% in the placebo group and 0.6% in the candesartan group, a significant difference. Metoprolol didn’t put a dent in the LVEF decline.
“Observational studies show early reduction in LVEF is associated with increased risk of developing heart failure later. So if a sustained, long-term effect of angiotensin inhibition can be confirmed in larger multicenter trials, preventive therapy may be indicated as standard care for breast cancer patients,” Dr. Gulati said.
Discussant Dr. Bonnie Ky of the University of Pennsylvania, Philadelphia, called PRADA an important study that moves the field of cardio-oncology forward, yet it’s also a trial that raises more questions than it answers.
PRADA certainly addresses a major problem: “The incidence of heart failure and cardiomyopathy increases over time in breast cancer patients exposed to anthracyclines and trastuzumab. Because patients are living longer because of cancer chemotherapy, their risk of dying of cardiovascular disease actually exceeds that of recurrent cancer in the long term,” she observed.
The study has three major limitations that prevent its findings being implemented in routine clinical practice at this time, Dr. Ky said. One is its relatively small size, even though it’s far bigger than any previous study. Another limitation is that this was an extremely low-cardiovascular-risk patient cohort: the baseline prevalence of diabetes was only 1.5%, fewer than 7% of patients had hypertension, and the baseline LVEF was 63%. That may be why no one developed a substantial decrement in LVEF or actual heart failure.
And since the incidence of cardiomyopathy following breast cancer therapy is known to climb over time, reaching a cumulative 12% at 6 years followup in trastuzumab-treated patients and 20% in those who receive both anthracyclines and trastuzumab (J Natl Cancer Inst. 2012 Sep 5;104(17):1293-305), the lack of extended followup time in PRADA is a significant shortcoming, she added.
The important questions raised by PRADA, Dr. Ky continued, include whether carvedilol or another beta blocker would have generated a positive result where metoprolol failed. Also, should the target population for prevention of cardiotoxicity be more narrowly focused on those at higher baseline cardiovascular risk? And bearing in mind that change in LVEF is a surrogate endpoint, what might be a more clinically meaningful and valid outcome measure? What’s the effect of carvedilol and other cardioprotective medications on cardiac biomarkers in breast cancer patients? And the most important questions of all, she said: What would be the effects of longer followup time and extended therapy?
“This study highlights for us in the field of cardio-oncology the critical need to develop a robust consensus definition of cardiotoxicity and a methodology to identify high cardiovascular risk patients,” she concluded.
PRADA was funded primarily by the University of Oslo and the Norwegian Cancer Society. Dr. Gulati reported having no financial conflicts of interest. Dr. Ky reported receiving a research grant from Pfizer and serving as a consultant to Bristol Myers Squibb.
ORLANDO – Concomitant treatment with candesartan protects against the early decline in left ventricular ejection fraction associated with adjunct therapy for early breast cancer.
That was the key finding in the PRADA trial (PRevention of cArdiac Dysfunction during Adjuvant breast cancer therapy), the largest study to date looking at prevention of cardiac dysfunction in a breast cancer population.
Another important finding in PRADA was that unlike the angiotensin receptor blocker candesartan (Atacand), metoprolol, a beta blocker, didn’t prevent the early drop in LVEF commonly seen in breast cancer patients treated with anthracyclines and trastuzumab (Herceptin), even though both classes of heart medications are cornerstones of the treatment of ischemic and hypertensive cardiomyopathy, Dr. Geeta Gulati reported at the American Heart Association scientific sessions.
Although the cardiotoxicity of certain breast cancer treatments is widely recognized and has spawned the emerging field of cardio-oncology, the literature in this area is weak. Indeed, a recent meta-analysis identified only four published randomized studies evaluating the possible cardioprotective role for beta blockers and angiotensin antagonists in patients undergoing anthracycline-based chemotherapy (Postgrad Med J doi:10.1136/ postgradmedj-2015-133535). None of the studies was double-blind, all relied upon echocardiographic assessment of changes in LVEF rather than gold-standard cardiac MRI, and the study sizes were small -- just 18-45 breast cancer patients.
Most problematic of all, the studies employed a variety of different definitions of cardiotoxicity, noted Dr. Gulati of Akershus University Hospital in Lorenskog, Norway.
In contrast, PRADA was a double-blind, placebo-controlled, 2 by 2 factorial design, single-center trial, which included 120 patients with early breast cancer. Participants were randomized to candesartan at a starting dose of 8 mg and target dose of 32 mg/day, metoprolol starting at 25 mg with a target of 100 mg/day, or placebo after breast cancer surgery but before the start of anthracycline-containing chemotherapy.
The primary endpoint was change in LVEF from baseline to completion of adjuvant therapy, a period as short as 10 weeks and as long as 64 weeks depending upon whether a woman also underwent courses of trastuzumab, taxanes, and/or radiation therapy.
The overall decline in LVEF was 2.6% in the placebo group and 0.6% in the candesartan group, a significant difference. Metoprolol didn’t put a dent in the LVEF decline.
“Observational studies show early reduction in LVEF is associated with increased risk of developing heart failure later. So if a sustained, long-term effect of angiotensin inhibition can be confirmed in larger multicenter trials, preventive therapy may be indicated as standard care for breast cancer patients,” Dr. Gulati said.
Discussant Dr. Bonnie Ky of the University of Pennsylvania, Philadelphia, called PRADA an important study that moves the field of cardio-oncology forward, yet it’s also a trial that raises more questions than it answers.
PRADA certainly addresses a major problem: “The incidence of heart failure and cardiomyopathy increases over time in breast cancer patients exposed to anthracyclines and trastuzumab. Because patients are living longer because of cancer chemotherapy, their risk of dying of cardiovascular disease actually exceeds that of recurrent cancer in the long term,” she observed.
The study has three major limitations that prevent its findings being implemented in routine clinical practice at this time, Dr. Ky said. One is its relatively small size, even though it’s far bigger than any previous study. Another limitation is that this was an extremely low-cardiovascular-risk patient cohort: the baseline prevalence of diabetes was only 1.5%, fewer than 7% of patients had hypertension, and the baseline LVEF was 63%. That may be why no one developed a substantial decrement in LVEF or actual heart failure.
And since the incidence of cardiomyopathy following breast cancer therapy is known to climb over time, reaching a cumulative 12% at 6 years followup in trastuzumab-treated patients and 20% in those who receive both anthracyclines and trastuzumab (J Natl Cancer Inst. 2012 Sep 5;104(17):1293-305), the lack of extended followup time in PRADA is a significant shortcoming, she added.
The important questions raised by PRADA, Dr. Ky continued, include whether carvedilol or another beta blocker would have generated a positive result where metoprolol failed. Also, should the target population for prevention of cardiotoxicity be more narrowly focused on those at higher baseline cardiovascular risk? And bearing in mind that change in LVEF is a surrogate endpoint, what might be a more clinically meaningful and valid outcome measure? What’s the effect of carvedilol and other cardioprotective medications on cardiac biomarkers in breast cancer patients? And the most important questions of all, she said: What would be the effects of longer followup time and extended therapy?
“This study highlights for us in the field of cardio-oncology the critical need to develop a robust consensus definition of cardiotoxicity and a methodology to identify high cardiovascular risk patients,” she concluded.
PRADA was funded primarily by the University of Oslo and the Norwegian Cancer Society. Dr. Gulati reported having no financial conflicts of interest. Dr. Ky reported receiving a research grant from Pfizer and serving as a consultant to Bristol Myers Squibb.
ORLANDO – Concomitant treatment with candesartan protects against the early decline in left ventricular ejection fraction associated with adjunct therapy for early breast cancer.
That was the key finding in the PRADA trial (PRevention of cArdiac Dysfunction during Adjuvant breast cancer therapy), the largest study to date looking at prevention of cardiac dysfunction in a breast cancer population.
Another important finding in PRADA was that unlike the angiotensin receptor blocker candesartan (Atacand), metoprolol, a beta blocker, didn’t prevent the early drop in LVEF commonly seen in breast cancer patients treated with anthracyclines and trastuzumab (Herceptin), even though both classes of heart medications are cornerstones of the treatment of ischemic and hypertensive cardiomyopathy, Dr. Geeta Gulati reported at the American Heart Association scientific sessions.
Although the cardiotoxicity of certain breast cancer treatments is widely recognized and has spawned the emerging field of cardio-oncology, the literature in this area is weak. Indeed, a recent meta-analysis identified only four published randomized studies evaluating the possible cardioprotective role for beta blockers and angiotensin antagonists in patients undergoing anthracycline-based chemotherapy (Postgrad Med J doi:10.1136/ postgradmedj-2015-133535). None of the studies was double-blind, all relied upon echocardiographic assessment of changes in LVEF rather than gold-standard cardiac MRI, and the study sizes were small -- just 18-45 breast cancer patients.
Most problematic of all, the studies employed a variety of different definitions of cardiotoxicity, noted Dr. Gulati of Akershus University Hospital in Lorenskog, Norway.
In contrast, PRADA was a double-blind, placebo-controlled, 2 by 2 factorial design, single-center trial, which included 120 patients with early breast cancer. Participants were randomized to candesartan at a starting dose of 8 mg and target dose of 32 mg/day, metoprolol starting at 25 mg with a target of 100 mg/day, or placebo after breast cancer surgery but before the start of anthracycline-containing chemotherapy.
The primary endpoint was change in LVEF from baseline to completion of adjuvant therapy, a period as short as 10 weeks and as long as 64 weeks depending upon whether a woman also underwent courses of trastuzumab, taxanes, and/or radiation therapy.
The overall decline in LVEF was 2.6% in the placebo group and 0.6% in the candesartan group, a significant difference. Metoprolol didn’t put a dent in the LVEF decline.
“Observational studies show early reduction in LVEF is associated with increased risk of developing heart failure later. So if a sustained, long-term effect of angiotensin inhibition can be confirmed in larger multicenter trials, preventive therapy may be indicated as standard care for breast cancer patients,” Dr. Gulati said.
Discussant Dr. Bonnie Ky of the University of Pennsylvania, Philadelphia, called PRADA an important study that moves the field of cardio-oncology forward, yet it’s also a trial that raises more questions than it answers.
PRADA certainly addresses a major problem: “The incidence of heart failure and cardiomyopathy increases over time in breast cancer patients exposed to anthracyclines and trastuzumab. Because patients are living longer because of cancer chemotherapy, their risk of dying of cardiovascular disease actually exceeds that of recurrent cancer in the long term,” she observed.
The study has three major limitations that prevent its findings being implemented in routine clinical practice at this time, Dr. Ky said. One is its relatively small size, even though it’s far bigger than any previous study. Another limitation is that this was an extremely low-cardiovascular-risk patient cohort: the baseline prevalence of diabetes was only 1.5%, fewer than 7% of patients had hypertension, and the baseline LVEF was 63%. That may be why no one developed a substantial decrement in LVEF or actual heart failure.
And since the incidence of cardiomyopathy following breast cancer therapy is known to climb over time, reaching a cumulative 12% at 6 years followup in trastuzumab-treated patients and 20% in those who receive both anthracyclines and trastuzumab (J Natl Cancer Inst. 2012 Sep 5;104(17):1293-305), the lack of extended followup time in PRADA is a significant shortcoming, she added.
The important questions raised by PRADA, Dr. Ky continued, include whether carvedilol or another beta blocker would have generated a positive result where metoprolol failed. Also, should the target population for prevention of cardiotoxicity be more narrowly focused on those at higher baseline cardiovascular risk? And bearing in mind that change in LVEF is a surrogate endpoint, what might be a more clinically meaningful and valid outcome measure? What’s the effect of carvedilol and other cardioprotective medications on cardiac biomarkers in breast cancer patients? And the most important questions of all, she said: What would be the effects of longer followup time and extended therapy?
“This study highlights for us in the field of cardio-oncology the critical need to develop a robust consensus definition of cardiotoxicity and a methodology to identify high cardiovascular risk patients,” she concluded.
PRADA was funded primarily by the University of Oslo and the Norwegian Cancer Society. Dr. Gulati reported having no financial conflicts of interest. Dr. Ky reported receiving a research grant from Pfizer and serving as a consultant to Bristol Myers Squibb.
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point: Inroads are being made in the growing problem of breast cancer treatment-associated cardiomyopathy.
Major finding: The average 2.6% decline from baseline in breast cancer patients during adjuvant therapy with anthracyclines with or without trastuzumab was negated by concomitant candesartan but not by metoprolol.
Data source: The PRADA trial was a randomized, double-blind, placebo-controlled, 2 by 2 factorial design study involving 120 patients undergoing adjuvant therapy for early breast cancer.
Disclosures: The primary sponsors of the study were the University of Oslo and the Norwegian Cancer Society. Additional support was provided by Abbott Diagnostic and AstraZeneca. The presenter reported no financial conflicts of interest.
AHA: SPRINT’s results upend hypertension targets
ORLANDO – Results from the SPRINT hypertension trial had been highly anticipated ever since the study stopped early in August and the sponsoring National Heart, Lung, and Blood Institute released the top-line positive result in September that treating systolic blood pressure to a target of less than 120 mm Hg led to statistically significant drops in a composite of cardiovascular endpoints as well as in all-cause death, compared with the standard target of less than 140 mm Hg.
When the much fuller report on the results finally came out in a special session at the American Heart Association scientific sessions as well as in a simultaneous publication (N Engl J Med. 2015 Nov 9. doi: 10.1056/NEJMoa1511939), the data left attendees buzzing and debating what the results will mean for revised hypertension guidelines and for clinical practice.
The most prominent reactions were accolades for the trial, starting with the independent discussants that the AHA invited to comment at the session, an outpouring of praise reminiscent of that showered on a hit movie:
“A major coup. Thank you, NHLBI,” declared Dr. Marc A. Pfeffer, professor of medicine at Harvard and a cardiologist at Brigham and Women’s Hospital in Boston.
“Thank you for this groundbreaking study,” said Dr. Clive Rosendorff, professor and cardiologist at Mount Sinai Hospital in New York.
“A remarkable trial. The most important blood pressure study in the last 40 years,” gushed Dr. Daniel W. Jones, professor of medicine at the University of Mississippi, Oxford, and director of clinical and population sciences at the Mississippi Center for Obesity Research, Jackson.
Following the huzzahs came a more substantive discussion among meeting attendees of what results from the 9,361-patient Systolic Blood Pressure Intervention Trial will mean for revised blood pressure goals in U.S. guidelines, what it might mean for defining who has hypertension, and how it might influence practice. Perhaps the most pressing issue for the AHA and American College of Cardiology panel that began work on a new revision of hypertension treatment guidelines earlier this year is how to reconcile the SPRINT results with finding from prior studies, especially the 2010 report of results from the ACCORD (Action to Control Cardiovascular Risk in Diabetes) trial (N Engl J Med. 2010;362[17]:1575-85.).
ACCORD, at half the size of SPRINT with 4,733 patients, had a very similar design as SPRINT but included only patients with diabetes while SPRINT excluded patients with diabetes. ACCORD failed to show a significant difference in its primary composite outcome after an average of 4.7 years between patients randomized to a hypertension treatment target of less than 140 mm Hg or less than 120 mm Hg, the same goals as in SPRINT. ACCORD did show a statistically significant 41% relative risk reduction for stroke, also in contrast to SPRINT, which showed a much less robust and nonsignificant 11% relative risk reduction in stroke.
In his commentary on SPRINT, Dr. Jones offered several possible explanations for the divergent results, including a possible inherent difference in vascular physiology between patients with diabetes and those with normal glycemic control; the younger patients enrolled in ACCORD (patients averaged 62 years old in ACCORD and 68 years old in SPRINT, and 28% of patients in SPRINT were at least 75 years old); the use of hydrochlorothiazide as the predominant diuretic in ACCORD versus predominant use of chlorthalidone in SPRINT; and the multiple interventions simultaneously tested in ACCORD, which also randomized patients into two arms with respect to glycemic control and into two arms of different lipid-controlling treatment.
SPRINT’s results “need to be assessed in the context of ACCORD,” commented Dr. Salim Yusuf in an interview. “I think the real result is somewhere in between the results of SPRINT and ACCORD” in terms of the appropriate systolic blood pressure target. What we need is a balanced perspective that takes all the trials. SPRINT was a very good trial, but like all studies it should be interpreted in the context of all the other related studies, not in isolation,” said Dr. Yusuf, professor and director of the Population Health Research Institute of McMaster University in Hamilton, Ont.
“Understandably, when something like SPRINT comes out there is a lot of enthusiasm. The first reaction is always ‘Wow!’ For patients who meet SPRINT’s enrollment criteria I think we will treat to a target of less than 120 mm Hg. But the guideline writers need to discuss SPRINT and balance it,” he said.
Despite his regard for SPRINT, Dr. Yusuf cited several additional concerns he has about the trial:
• Its early stoppage (SPRINT had originally been designed to run 5-6 years, but it was halted after an average treatment duration of just over 3 years). “When you stop a trial early there is always an upward bias. The apparent treatment effect gets inflated,” he said.
• The increased rate of acute kidney injury among patients randomized to the more aggressive treatment arm, a 4.1% rate, compared with a 2.5% rate in the control patients randomized to treatment to a goal of systolic pressure less than 140 mm Hg, a statistically significant difference.
• The “highly selected, high-risk” patients enrolled into SPRINT. “You can’t extrapolate the results to the average patient,” Dr. Yusuf said.
Some of these concerns and cautions were shared by Dr. Prakash Deedwania, professor of medicine at the University of California, San Francisco, although overall he called the SPRINT results “very exciting.”
“Superficially, SPRINT seems to say treat everyone to a blood pressure of less than 120 mm Hg, but that’s not the case. The patients in SPRINT were primarily very well established patients with hypertension. I’d be concerned about an elderly patient with cardiovascular disease and a blood pressure of 130 mm Hg. If you reduce that to less than 120 mm Hg the diastolic pressure may also fall and that’s important for coronary perfusion.” He also cited the absence so far of a subanalysis of what happened to patients with preexisting renal disease and the lack of data on the outcomes of patients whose systolic pressure fell to levels well below 120 mm Hg.
For others, however, the overall, statistically significant 27% reduction in overall mortality was a reassuring indicator of the safety of the aggressive treatment regimen used in SPRINT. “If there was a meaningful worsening of renal function that harmed patients, you would not see a reduction in all-cause mortality,” commented Dr. Gregg C. Fonarow, professor and associate chief of cardiology at the University of California, Los Angeles.
“We have had so many trials that couldn’t dream of producing a reduction in all-cause mortality. Here we have a trial with a robust, clinically meaningful reduction in all-cause mortality that ultimately demonstrates the benefits outweigh the risks,” he said in an interview.
SPRINT “is a phenomenal breakthrough. It’s data we’ve been awaiting for 20-plus years, to now know that a lower blood pressure target is safe and absolutely essential, and where the benefits outweigh the risks,” Dr. Fonarow said. “Now implementation becomes critical. The SPRINT results are truly practice changing.”
SPRINT received no commercial support. The study received antihypertensive drugs from Arbor and Takeda at no charge for a small percentage of enrolled patients. Dr. Pfeffer has been a consultant to more than 20 companies. Dr. Rosendorff has been a consultant to McNeil and received research funding from Eisai. Dr. Yusuf has received honoraria and research grants from Sanofi-Aventis, Bristol-Myers Squibb, Pfizer, Boehringer-Ingelheim, Bayer, and Astra Zeneca. Dr. Jones, Dr. Deedwania, and Dr. Fonarow had no disclosures.
On Twitter @mitchelzoler
ORLANDO – Results from the SPRINT hypertension trial had been highly anticipated ever since the study stopped early in August and the sponsoring National Heart, Lung, and Blood Institute released the top-line positive result in September that treating systolic blood pressure to a target of less than 120 mm Hg led to statistically significant drops in a composite of cardiovascular endpoints as well as in all-cause death, compared with the standard target of less than 140 mm Hg.
When the much fuller report on the results finally came out in a special session at the American Heart Association scientific sessions as well as in a simultaneous publication (N Engl J Med. 2015 Nov 9. doi: 10.1056/NEJMoa1511939), the data left attendees buzzing and debating what the results will mean for revised hypertension guidelines and for clinical practice.
The most prominent reactions were accolades for the trial, starting with the independent discussants that the AHA invited to comment at the session, an outpouring of praise reminiscent of that showered on a hit movie:
“A major coup. Thank you, NHLBI,” declared Dr. Marc A. Pfeffer, professor of medicine at Harvard and a cardiologist at Brigham and Women’s Hospital in Boston.
“Thank you for this groundbreaking study,” said Dr. Clive Rosendorff, professor and cardiologist at Mount Sinai Hospital in New York.
“A remarkable trial. The most important blood pressure study in the last 40 years,” gushed Dr. Daniel W. Jones, professor of medicine at the University of Mississippi, Oxford, and director of clinical and population sciences at the Mississippi Center for Obesity Research, Jackson.
Following the huzzahs came a more substantive discussion among meeting attendees of what results from the 9,361-patient Systolic Blood Pressure Intervention Trial will mean for revised blood pressure goals in U.S. guidelines, what it might mean for defining who has hypertension, and how it might influence practice. Perhaps the most pressing issue for the AHA and American College of Cardiology panel that began work on a new revision of hypertension treatment guidelines earlier this year is how to reconcile the SPRINT results with finding from prior studies, especially the 2010 report of results from the ACCORD (Action to Control Cardiovascular Risk in Diabetes) trial (N Engl J Med. 2010;362[17]:1575-85.).
ACCORD, at half the size of SPRINT with 4,733 patients, had a very similar design as SPRINT but included only patients with diabetes while SPRINT excluded patients with diabetes. ACCORD failed to show a significant difference in its primary composite outcome after an average of 4.7 years between patients randomized to a hypertension treatment target of less than 140 mm Hg or less than 120 mm Hg, the same goals as in SPRINT. ACCORD did show a statistically significant 41% relative risk reduction for stroke, also in contrast to SPRINT, which showed a much less robust and nonsignificant 11% relative risk reduction in stroke.
In his commentary on SPRINT, Dr. Jones offered several possible explanations for the divergent results, including a possible inherent difference in vascular physiology between patients with diabetes and those with normal glycemic control; the younger patients enrolled in ACCORD (patients averaged 62 years old in ACCORD and 68 years old in SPRINT, and 28% of patients in SPRINT were at least 75 years old); the use of hydrochlorothiazide as the predominant diuretic in ACCORD versus predominant use of chlorthalidone in SPRINT; and the multiple interventions simultaneously tested in ACCORD, which also randomized patients into two arms with respect to glycemic control and into two arms of different lipid-controlling treatment.
SPRINT’s results “need to be assessed in the context of ACCORD,” commented Dr. Salim Yusuf in an interview. “I think the real result is somewhere in between the results of SPRINT and ACCORD” in terms of the appropriate systolic blood pressure target. What we need is a balanced perspective that takes all the trials. SPRINT was a very good trial, but like all studies it should be interpreted in the context of all the other related studies, not in isolation,” said Dr. Yusuf, professor and director of the Population Health Research Institute of McMaster University in Hamilton, Ont.
“Understandably, when something like SPRINT comes out there is a lot of enthusiasm. The first reaction is always ‘Wow!’ For patients who meet SPRINT’s enrollment criteria I think we will treat to a target of less than 120 mm Hg. But the guideline writers need to discuss SPRINT and balance it,” he said.
Despite his regard for SPRINT, Dr. Yusuf cited several additional concerns he has about the trial:
• Its early stoppage (SPRINT had originally been designed to run 5-6 years, but it was halted after an average treatment duration of just over 3 years). “When you stop a trial early there is always an upward bias. The apparent treatment effect gets inflated,” he said.
• The increased rate of acute kidney injury among patients randomized to the more aggressive treatment arm, a 4.1% rate, compared with a 2.5% rate in the control patients randomized to treatment to a goal of systolic pressure less than 140 mm Hg, a statistically significant difference.
• The “highly selected, high-risk” patients enrolled into SPRINT. “You can’t extrapolate the results to the average patient,” Dr. Yusuf said.
Some of these concerns and cautions were shared by Dr. Prakash Deedwania, professor of medicine at the University of California, San Francisco, although overall he called the SPRINT results “very exciting.”
“Superficially, SPRINT seems to say treat everyone to a blood pressure of less than 120 mm Hg, but that’s not the case. The patients in SPRINT were primarily very well established patients with hypertension. I’d be concerned about an elderly patient with cardiovascular disease and a blood pressure of 130 mm Hg. If you reduce that to less than 120 mm Hg the diastolic pressure may also fall and that’s important for coronary perfusion.” He also cited the absence so far of a subanalysis of what happened to patients with preexisting renal disease and the lack of data on the outcomes of patients whose systolic pressure fell to levels well below 120 mm Hg.
For others, however, the overall, statistically significant 27% reduction in overall mortality was a reassuring indicator of the safety of the aggressive treatment regimen used in SPRINT. “If there was a meaningful worsening of renal function that harmed patients, you would not see a reduction in all-cause mortality,” commented Dr. Gregg C. Fonarow, professor and associate chief of cardiology at the University of California, Los Angeles.
“We have had so many trials that couldn’t dream of producing a reduction in all-cause mortality. Here we have a trial with a robust, clinically meaningful reduction in all-cause mortality that ultimately demonstrates the benefits outweigh the risks,” he said in an interview.
SPRINT “is a phenomenal breakthrough. It’s data we’ve been awaiting for 20-plus years, to now know that a lower blood pressure target is safe and absolutely essential, and where the benefits outweigh the risks,” Dr. Fonarow said. “Now implementation becomes critical. The SPRINT results are truly practice changing.”
SPRINT received no commercial support. The study received antihypertensive drugs from Arbor and Takeda at no charge for a small percentage of enrolled patients. Dr. Pfeffer has been a consultant to more than 20 companies. Dr. Rosendorff has been a consultant to McNeil and received research funding from Eisai. Dr. Yusuf has received honoraria and research grants from Sanofi-Aventis, Bristol-Myers Squibb, Pfizer, Boehringer-Ingelheim, Bayer, and Astra Zeneca. Dr. Jones, Dr. Deedwania, and Dr. Fonarow had no disclosures.
On Twitter @mitchelzoler
ORLANDO – Results from the SPRINT hypertension trial had been highly anticipated ever since the study stopped early in August and the sponsoring National Heart, Lung, and Blood Institute released the top-line positive result in September that treating systolic blood pressure to a target of less than 120 mm Hg led to statistically significant drops in a composite of cardiovascular endpoints as well as in all-cause death, compared with the standard target of less than 140 mm Hg.
When the much fuller report on the results finally came out in a special session at the American Heart Association scientific sessions as well as in a simultaneous publication (N Engl J Med. 2015 Nov 9. doi: 10.1056/NEJMoa1511939), the data left attendees buzzing and debating what the results will mean for revised hypertension guidelines and for clinical practice.
The most prominent reactions were accolades for the trial, starting with the independent discussants that the AHA invited to comment at the session, an outpouring of praise reminiscent of that showered on a hit movie:
“A major coup. Thank you, NHLBI,” declared Dr. Marc A. Pfeffer, professor of medicine at Harvard and a cardiologist at Brigham and Women’s Hospital in Boston.
“Thank you for this groundbreaking study,” said Dr. Clive Rosendorff, professor and cardiologist at Mount Sinai Hospital in New York.
“A remarkable trial. The most important blood pressure study in the last 40 years,” gushed Dr. Daniel W. Jones, professor of medicine at the University of Mississippi, Oxford, and director of clinical and population sciences at the Mississippi Center for Obesity Research, Jackson.
Following the huzzahs came a more substantive discussion among meeting attendees of what results from the 9,361-patient Systolic Blood Pressure Intervention Trial will mean for revised blood pressure goals in U.S. guidelines, what it might mean for defining who has hypertension, and how it might influence practice. Perhaps the most pressing issue for the AHA and American College of Cardiology panel that began work on a new revision of hypertension treatment guidelines earlier this year is how to reconcile the SPRINT results with finding from prior studies, especially the 2010 report of results from the ACCORD (Action to Control Cardiovascular Risk in Diabetes) trial (N Engl J Med. 2010;362[17]:1575-85.).
ACCORD, at half the size of SPRINT with 4,733 patients, had a very similar design as SPRINT but included only patients with diabetes while SPRINT excluded patients with diabetes. ACCORD failed to show a significant difference in its primary composite outcome after an average of 4.7 years between patients randomized to a hypertension treatment target of less than 140 mm Hg or less than 120 mm Hg, the same goals as in SPRINT. ACCORD did show a statistically significant 41% relative risk reduction for stroke, also in contrast to SPRINT, which showed a much less robust and nonsignificant 11% relative risk reduction in stroke.
In his commentary on SPRINT, Dr. Jones offered several possible explanations for the divergent results, including a possible inherent difference in vascular physiology between patients with diabetes and those with normal glycemic control; the younger patients enrolled in ACCORD (patients averaged 62 years old in ACCORD and 68 years old in SPRINT, and 28% of patients in SPRINT were at least 75 years old); the use of hydrochlorothiazide as the predominant diuretic in ACCORD versus predominant use of chlorthalidone in SPRINT; and the multiple interventions simultaneously tested in ACCORD, which also randomized patients into two arms with respect to glycemic control and into two arms of different lipid-controlling treatment.
SPRINT’s results “need to be assessed in the context of ACCORD,” commented Dr. Salim Yusuf in an interview. “I think the real result is somewhere in between the results of SPRINT and ACCORD” in terms of the appropriate systolic blood pressure target. What we need is a balanced perspective that takes all the trials. SPRINT was a very good trial, but like all studies it should be interpreted in the context of all the other related studies, not in isolation,” said Dr. Yusuf, professor and director of the Population Health Research Institute of McMaster University in Hamilton, Ont.
“Understandably, when something like SPRINT comes out there is a lot of enthusiasm. The first reaction is always ‘Wow!’ For patients who meet SPRINT’s enrollment criteria I think we will treat to a target of less than 120 mm Hg. But the guideline writers need to discuss SPRINT and balance it,” he said.
Despite his regard for SPRINT, Dr. Yusuf cited several additional concerns he has about the trial:
• Its early stoppage (SPRINT had originally been designed to run 5-6 years, but it was halted after an average treatment duration of just over 3 years). “When you stop a trial early there is always an upward bias. The apparent treatment effect gets inflated,” he said.
• The increased rate of acute kidney injury among patients randomized to the more aggressive treatment arm, a 4.1% rate, compared with a 2.5% rate in the control patients randomized to treatment to a goal of systolic pressure less than 140 mm Hg, a statistically significant difference.
• The “highly selected, high-risk” patients enrolled into SPRINT. “You can’t extrapolate the results to the average patient,” Dr. Yusuf said.
Some of these concerns and cautions were shared by Dr. Prakash Deedwania, professor of medicine at the University of California, San Francisco, although overall he called the SPRINT results “very exciting.”
“Superficially, SPRINT seems to say treat everyone to a blood pressure of less than 120 mm Hg, but that’s not the case. The patients in SPRINT were primarily very well established patients with hypertension. I’d be concerned about an elderly patient with cardiovascular disease and a blood pressure of 130 mm Hg. If you reduce that to less than 120 mm Hg the diastolic pressure may also fall and that’s important for coronary perfusion.” He also cited the absence so far of a subanalysis of what happened to patients with preexisting renal disease and the lack of data on the outcomes of patients whose systolic pressure fell to levels well below 120 mm Hg.
For others, however, the overall, statistically significant 27% reduction in overall mortality was a reassuring indicator of the safety of the aggressive treatment regimen used in SPRINT. “If there was a meaningful worsening of renal function that harmed patients, you would not see a reduction in all-cause mortality,” commented Dr. Gregg C. Fonarow, professor and associate chief of cardiology at the University of California, Los Angeles.
“We have had so many trials that couldn’t dream of producing a reduction in all-cause mortality. Here we have a trial with a robust, clinically meaningful reduction in all-cause mortality that ultimately demonstrates the benefits outweigh the risks,” he said in an interview.
SPRINT “is a phenomenal breakthrough. It’s data we’ve been awaiting for 20-plus years, to now know that a lower blood pressure target is safe and absolutely essential, and where the benefits outweigh the risks,” Dr. Fonarow said. “Now implementation becomes critical. The SPRINT results are truly practice changing.”
SPRINT received no commercial support. The study received antihypertensive drugs from Arbor and Takeda at no charge for a small percentage of enrolled patients. Dr. Pfeffer has been a consultant to more than 20 companies. Dr. Rosendorff has been a consultant to McNeil and received research funding from Eisai. Dr. Yusuf has received honoraria and research grants from Sanofi-Aventis, Bristol-Myers Squibb, Pfizer, Boehringer-Ingelheim, Bayer, and Astra Zeneca. Dr. Jones, Dr. Deedwania, and Dr. Fonarow had no disclosures.
On Twitter @mitchelzoler
EXPERT ANALYSIS FROM THE AHA SCIENTIFIC SESSIONS
Key clinical point: The first full report of results from the SPRINT trial of hypertension treatment targets generated lots of opinions on their implications.
Major finding: Combined cardiovascular events occurred in 5.2% of patients treated to a target systolic blood pressure of less than 120 mm Hg and 6.8% of patients treated to a target of less than 140 mm Hg.
Data source: The multicenter, randomized trial involved 9,361 patients.
Disclosures: SPRINT received no commercial support. The study received antihypertensive drugs from Arbor and Takeda at no charge for a small percentage of enrolled patients. Dr. Pfeffer has been a consultant to more than 20 companies. Dr. Rosendorff has been a consultant to McNeil and received research funding from Eisai. Dr. Yusuf has received honoraria and research grants from Sanofi-Aventis, Bristol-Myers Squibb, Pfizer, Boehringer-Ingelheim, Bayer, and Astra Zeneca. Dr. Jones, Dr. Deedwania, and Dr. Fonarow had no disclosures.
AHA: Mixed results for mitral valve replacement vs. repair
Patients undergoing mitral valve replacement had a lower risk of regurgitation and heart failure–related adverse events at 2 years than those undergoing valve repair, according to the results of a trial presented at the American Heart Association scientific sessions and published simultaneously in the New England Journal of Medicine.
The results of the trial appear to associate mitral valve replacement with clinical advantages over mitral valve repair after 2 years of follow-up. However, replacement held no significant advantages over repair in the primary endpoint of left ventricular end-systolic volume index (LVESVI) or in overall survival, said Dr. Daniel Goldstein of the department of cardiothoracic surgery at Montefiore Medical Center, New York.
In the trial conducted by the Cardiothoracic Surgical Trials Network (CTSN), 251 patients with chronic severe ischemic mitral regurgitation were randomly assigned to undergo surgical repair of the mitral valve or to receive a mitral valve replacement with a prosthetic and procedure selected at the discretion of the surgeon.
In addition to the primary endpoint of LVESVI, the two approaches were also compared for survival, regurgitation recurrence, and heart failure events.
At 2 years, the mean change from baseline in LVESVI, a measure of remodeling, did not differ significantly between the repair and replacement arms (–9.0 vs. –6.5 mL/m2, respectively). In addition, although the 2-year mortality rate was numerically lower in the repair arm relative to the replacement arm (19% vs. 23.2%, respectively), it was also not statistically different (P = .39).
However, the rate of recurrence of moderate or severe mitral regurgitation favored replacement over repair and was significant (3.8% vs. 58.8%, respectively; P less than .001). In addition, the rate of cardiovascular readmissions was significantly lower in the replacement group (P = .01).
For those in the repair group, there were significant trends for more serious adverse events related to heart failure (P = .05) and for a lower quality of life improvement (P = .07) on the Minnesota Living With Heart Failure questionnaire. There were no significant differences in rates of all serious adverse events or overall readmissions.
All of the differences between groups observed at 2 years amplify differences previously reported after 12 months (N Engl J Med. 2014 Jan 2;370[1]:23-32). For example, the difference in the rate of moderate to severe regurgitation favoring replacement over repair was already significant at that time (2.3% vs. 32.6%, respectively; P less than .001), even though the mortality rates were then, as now, numerically lower in the repair group versus the replacement group (14.3% vs. 17.6%, respectively; P = .45).
Dr. Goldstein reported no relevant financial relationships.
Patients undergoing mitral valve replacement had a lower risk of regurgitation and heart failure–related adverse events at 2 years than those undergoing valve repair, according to the results of a trial presented at the American Heart Association scientific sessions and published simultaneously in the New England Journal of Medicine.
The results of the trial appear to associate mitral valve replacement with clinical advantages over mitral valve repair after 2 years of follow-up. However, replacement held no significant advantages over repair in the primary endpoint of left ventricular end-systolic volume index (LVESVI) or in overall survival, said Dr. Daniel Goldstein of the department of cardiothoracic surgery at Montefiore Medical Center, New York.
In the trial conducted by the Cardiothoracic Surgical Trials Network (CTSN), 251 patients with chronic severe ischemic mitral regurgitation were randomly assigned to undergo surgical repair of the mitral valve or to receive a mitral valve replacement with a prosthetic and procedure selected at the discretion of the surgeon.
In addition to the primary endpoint of LVESVI, the two approaches were also compared for survival, regurgitation recurrence, and heart failure events.
At 2 years, the mean change from baseline in LVESVI, a measure of remodeling, did not differ significantly between the repair and replacement arms (–9.0 vs. –6.5 mL/m2, respectively). In addition, although the 2-year mortality rate was numerically lower in the repair arm relative to the replacement arm (19% vs. 23.2%, respectively), it was also not statistically different (P = .39).
However, the rate of recurrence of moderate or severe mitral regurgitation favored replacement over repair and was significant (3.8% vs. 58.8%, respectively; P less than .001). In addition, the rate of cardiovascular readmissions was significantly lower in the replacement group (P = .01).
For those in the repair group, there were significant trends for more serious adverse events related to heart failure (P = .05) and for a lower quality of life improvement (P = .07) on the Minnesota Living With Heart Failure questionnaire. There were no significant differences in rates of all serious adverse events or overall readmissions.
All of the differences between groups observed at 2 years amplify differences previously reported after 12 months (N Engl J Med. 2014 Jan 2;370[1]:23-32). For example, the difference in the rate of moderate to severe regurgitation favoring replacement over repair was already significant at that time (2.3% vs. 32.6%, respectively; P less than .001), even though the mortality rates were then, as now, numerically lower in the repair group versus the replacement group (14.3% vs. 17.6%, respectively; P = .45).
Dr. Goldstein reported no relevant financial relationships.
Patients undergoing mitral valve replacement had a lower risk of regurgitation and heart failure–related adverse events at 2 years than those undergoing valve repair, according to the results of a trial presented at the American Heart Association scientific sessions and published simultaneously in the New England Journal of Medicine.
The results of the trial appear to associate mitral valve replacement with clinical advantages over mitral valve repair after 2 years of follow-up. However, replacement held no significant advantages over repair in the primary endpoint of left ventricular end-systolic volume index (LVESVI) or in overall survival, said Dr. Daniel Goldstein of the department of cardiothoracic surgery at Montefiore Medical Center, New York.
In the trial conducted by the Cardiothoracic Surgical Trials Network (CTSN), 251 patients with chronic severe ischemic mitral regurgitation were randomly assigned to undergo surgical repair of the mitral valve or to receive a mitral valve replacement with a prosthetic and procedure selected at the discretion of the surgeon.
In addition to the primary endpoint of LVESVI, the two approaches were also compared for survival, regurgitation recurrence, and heart failure events.
At 2 years, the mean change from baseline in LVESVI, a measure of remodeling, did not differ significantly between the repair and replacement arms (–9.0 vs. –6.5 mL/m2, respectively). In addition, although the 2-year mortality rate was numerically lower in the repair arm relative to the replacement arm (19% vs. 23.2%, respectively), it was also not statistically different (P = .39).
However, the rate of recurrence of moderate or severe mitral regurgitation favored replacement over repair and was significant (3.8% vs. 58.8%, respectively; P less than .001). In addition, the rate of cardiovascular readmissions was significantly lower in the replacement group (P = .01).
For those in the repair group, there were significant trends for more serious adverse events related to heart failure (P = .05) and for a lower quality of life improvement (P = .07) on the Minnesota Living With Heart Failure questionnaire. There were no significant differences in rates of all serious adverse events or overall readmissions.
All of the differences between groups observed at 2 years amplify differences previously reported after 12 months (N Engl J Med. 2014 Jan 2;370[1]:23-32). For example, the difference in the rate of moderate to severe regurgitation favoring replacement over repair was already significant at that time (2.3% vs. 32.6%, respectively; P less than .001), even though the mortality rates were then, as now, numerically lower in the repair group versus the replacement group (14.3% vs. 17.6%, respectively; P = .45).
Dr. Goldstein reported no relevant financial relationships.
FROM THE AHA SCIENTIFIC SESSIONS
Key clinical point: Mitral valve replacement reduced regurgitation better than valve repair, but it didn’t significantly improve left ventricular function or survival.
Major finding: In patients with severe ischemic mitral regurgitation, regurgitation occurred more frequently after mitral valve repair than after valve replacement (58.8% vs. 3.8%; P less than .001), but left ventricular end-systolic volume indexes and survival rates were not significantly different.
Data source: A randomized, multicenter trial with 251 patients.
Disclosures: Dr. Goldstein reported no relevant financial relationships.
Nitrate therapy linked to less exercise in heart failure
Isosorbide mononitrate therapy for patients with heart failure and preserved ejection fraction may actually be associated with lower levels of daily activity and does not appear to achieve significant improvements in quality of life or exercise capacity, compared with placebo.
In a double-blind crossover trial in 110 patients who were prescribed escalating doses of isosorbide mononitrate over a period of 6 weeks and then switched, those patients treated with the highest dose of 120 mg once daily showed a nonsignificant trend (P = .06) toward lower daily activity and hours of activity per day, compared with those given placebo.
According to data presented at the American Heart Association scientific sessions and published simultaneously Nov. 8 in the New England Journal of Medicine, researchers observed a decrease in average daily accelerometer units with increasing doses of isosorbide mononitrate, but not with placebo (N Engl J Med. 2015 Nov 8 [doi: 10.1056/NEJMoa1510774]).
They also noted a nonsignificant decrease in quality of life scores, N-terminal probrain natriuretic peptide, and blood pressure with isosorbide mononitrate, as well as a greater number of discontinuations and adverse events.
Although nitrates are commonly prescribed for symptom relief in heart failure, the authors said, the pathophysiology of heart failure with preserved ejection fraction may be responsible for the observed lack of improvement in exercise tolerance and daily activity.
“Increased ventricular systolic and vascular stiffness, autonomic dysfunction, chronotropic incompetence, and altered baroreflex sensitivity are common and may limit the hemodynamic benefit of nitrates,” wrote Dr. Margaret M. Redfield of the Mayo Clinic, Rochester, Minn., and her coauthors.
The National Heart, Lung, and Blood Institute supported the study. Several authors reported grants, fees, royalties and consultancies from private industry, including a patent relating to natriuretic peptide.
Isosorbide mononitrate therapy for patients with heart failure and preserved ejection fraction may actually be associated with lower levels of daily activity and does not appear to achieve significant improvements in quality of life or exercise capacity, compared with placebo.
In a double-blind crossover trial in 110 patients who were prescribed escalating doses of isosorbide mononitrate over a period of 6 weeks and then switched, those patients treated with the highest dose of 120 mg once daily showed a nonsignificant trend (P = .06) toward lower daily activity and hours of activity per day, compared with those given placebo.
According to data presented at the American Heart Association scientific sessions and published simultaneously Nov. 8 in the New England Journal of Medicine, researchers observed a decrease in average daily accelerometer units with increasing doses of isosorbide mononitrate, but not with placebo (N Engl J Med. 2015 Nov 8 [doi: 10.1056/NEJMoa1510774]).
They also noted a nonsignificant decrease in quality of life scores, N-terminal probrain natriuretic peptide, and blood pressure with isosorbide mononitrate, as well as a greater number of discontinuations and adverse events.
Although nitrates are commonly prescribed for symptom relief in heart failure, the authors said, the pathophysiology of heart failure with preserved ejection fraction may be responsible for the observed lack of improvement in exercise tolerance and daily activity.
“Increased ventricular systolic and vascular stiffness, autonomic dysfunction, chronotropic incompetence, and altered baroreflex sensitivity are common and may limit the hemodynamic benefit of nitrates,” wrote Dr. Margaret M. Redfield of the Mayo Clinic, Rochester, Minn., and her coauthors.
The National Heart, Lung, and Blood Institute supported the study. Several authors reported grants, fees, royalties and consultancies from private industry, including a patent relating to natriuretic peptide.
Isosorbide mononitrate therapy for patients with heart failure and preserved ejection fraction may actually be associated with lower levels of daily activity and does not appear to achieve significant improvements in quality of life or exercise capacity, compared with placebo.
In a double-blind crossover trial in 110 patients who were prescribed escalating doses of isosorbide mononitrate over a period of 6 weeks and then switched, those patients treated with the highest dose of 120 mg once daily showed a nonsignificant trend (P = .06) toward lower daily activity and hours of activity per day, compared with those given placebo.
According to data presented at the American Heart Association scientific sessions and published simultaneously Nov. 8 in the New England Journal of Medicine, researchers observed a decrease in average daily accelerometer units with increasing doses of isosorbide mononitrate, but not with placebo (N Engl J Med. 2015 Nov 8 [doi: 10.1056/NEJMoa1510774]).
They also noted a nonsignificant decrease in quality of life scores, N-terminal probrain natriuretic peptide, and blood pressure with isosorbide mononitrate, as well as a greater number of discontinuations and adverse events.
Although nitrates are commonly prescribed for symptom relief in heart failure, the authors said, the pathophysiology of heart failure with preserved ejection fraction may be responsible for the observed lack of improvement in exercise tolerance and daily activity.
“Increased ventricular systolic and vascular stiffness, autonomic dysfunction, chronotropic incompetence, and altered baroreflex sensitivity are common and may limit the hemodynamic benefit of nitrates,” wrote Dr. Margaret M. Redfield of the Mayo Clinic, Rochester, Minn., and her coauthors.
The National Heart, Lung, and Blood Institute supported the study. Several authors reported grants, fees, royalties and consultancies from private industry, including a patent relating to natriuretic peptide.
FROM THE AHA SCIENTIFIC SESSIONS
Key clinical point: Isosorbide mononitrate therapy may be associated with lower levels of daily activity in patients with heart failure and preserved ejection fraction.
Major finding: Patients treated with the highest dose of isosorbide mononitrate showed a nonsignificant trend toward lower daily activity and hours of activity per day, compared with patients on placebo.
Data source: A double-blind crossover trial in 110 patients with heart failure and preserved ejection fraction.
Disclosures: The National Heart, Lung, and Blood Institute supported the study. Several authors reported grants, fees, royalties, and consultancies from private industry, including a patent relating to natriuretic peptide.