User login
Implanted phrenic-nerve stimulator improves central sleep apnea
FLORENCE, ITALY – In patients with moderate to severe central sleep apnea, an implanted device that stimulates the phrenic nerve to optimize diaphragm-driven breathing met its efficacy and safety goals, based on results from a multicenter, controlled trial with 151 patients.
Among the 68 patients randomized to active treatment with the device and available for follow-up after 6 months on treatment, 35 patients (51%) had a 50% or better reduction in their apnea-hypopnea index compared with 8 of 73 patients (11%) who had this level of response following device implantation but without its active use.
This statistically significant difference in response to the study’s primary endpoint should pave the way for the device’s approval, Dr. Maria Rosa Costanzo said at a meeting held by the Heart Failure Association of the European Society of Cardiology.
Although the trial enrolled patients with a mix of disorders that caused their central sleep apnea, the majority, 80 patients, had heart failure. Other enrollees had their breathing disorder secondary to atrial fibrillation, hypertension, and other diseases, suggesting that the implanted device, called the remede System, is suitable for patients with moderate to severe central sleep apnea regardless of the etiology, said Dr. Costanzo, medical director for heart failure at the Advocate Medical Group in Naperville, Ill. Among the 80 heart failure patients in the trial, the percentage of patients on active treatment who had a 50% or better reduction in their apnea-hypopnea index closely matched the rate in the entire study group.
The results also demonstrated the treatment’s safety, with a 9% rate of serious adverse events secondary to either the device’s implantation or function during the 12 months following placement in all 151 patients enrolled. Patients in the control arm had a device implanted but not turned on during the first 6 months of the study. The device was turned on and they received active treatment during the next 6 months. The trial’s prespecified safety goal, developed in conjunction with the Food and Drug Administration, was a 1-year rate of freedom from a serious adverse event of at least 80%; the actual rate achieved was 91%.
Successful implantation of the device by electrophysiology cardiologists occurred in 97% of enrolled patients, a procedure that took an average of nearly 3 hours. Need for a lead revision, one of the serious adverse events tallied during follow-up, occurred in 3% of patients. No patients in the study died during 1-year follow-up. Most other serious adverse events involved lead reposition (but not revision) to better optimize the phrenic nerve stimulation. Dr. Costanzo likened the complexity of implanting and operating the device to placement and use of a cardiac resynchronization device.
The efficacy and safety of the device shown in this pivotal trial “should be plenty” for obtaining FDA approval, predicted Dr. Costanzo, the study’s lead investigator, which would make it the first approved intervention for central sleep apnea. “I think this is a game changer,” she said in an interview.
But coming less than a year after a report of an unexpected excess-mortality rate in heart failure patients treated for central sleep apnea with an adaptive servo-ventilation device (N Engl J Med. 2015 Sept 17;373[12]:1095-1105), heart-failure specialists are now more demanding about the data needed to prove safety and clinical benefit from an intervention that targets central sleep apnea and sleep-disordered breathing.
“I think we need an endpoint that involves hospitalizations and death” to more clearly demonstrate meaningful clinical benefit and safety, said Dr. Mariell Jessup, a professor of medicine and heart failure specialist at the University of Pennsylvania in Philadelphia. Following the experience with increased mortality from servo-ventilation “we now need to demand” more comprehensive safety data in sleep trials. Also, the approach tested in this study involves “putting a device into patients, so it’s not completely benign,” she said in an interview. “A lot of things that we thought made a lot of sense, like treating a heart-failure patient’s sleep apnea, turned out to cause things we didn’t expect. We need to be cautious.”
Dr. Costanzo agreed that there is need for additional studies of the phrenic-nerve stimulating device in larger number of heart failure patients that involve heart-failure-specific endpoints. But she also stressed how life changing this intervention was for some of the patients in the study. “The transformation of their lives was unbelievable. They said things like ‘I feel I have my life back.’ ”
She also stressed that the mechanism of action of phrenic nerve stimulation is dramatically different from more traditional sleep-apnea treatments that have relied on positive air pressure devices.
Phrenic nerve stimulation causes contraction of a patient’s diaphragm that creates negative pressure within the chest cavity in a manner similar to that of natural breathing. The stimulation is adjusted to make it imperceptible to patients, and stimulation does not occur when a patient is standing or sitting, only when lying down. “With positive airway pressure in patients with advanced heart failure you reduce venous return, and when a patient’s heart is sick and depends on preload this can hurt the patient. Phrenic nerve stimulation does the opposite. It contracts the diaphragm and creates negative pressure, so it anything it facilitates venous return,” she explained.
The trial, run at 31 centers, mostly in the United States with the others in Europe, enrolled patients with moderate to severe central sleep apnea with an average apnea-hypopnea index of 45 episodes per hour while sleeping. The average age was 65 years, about 90% of the patients were men, and average body mass index was 31 kg/m2.
In addition to the primary efficacy endpoint of reduced apnea-hypopnea index, the patients on active treatment also showed statistically significant reductions compared with baseline in central apnea episodes and in daytime sleepiness measured on the Epworth Sleepiness Scale and an improvement in the patients’ global assessment of their condition. The changes did not occur in the control patients. In the treated patients central apnea episodes fell from an average of 32 episodes per hour at baseline to an average of 6 central episodes an hour after 6 months on treatment.
Dr. Costanzo is a consultant to and has received research support from Respicardia, the company developing the tested phrenic-nerve stimulation device. Dr. Jessup had no disclosures.
On Twitter @mitchelzoler
This is a really interesting and provocative study that tests a concept that flies in the face of conventional wisdom. In the past, we tried our best to avoid phrenic nerve stimulation when implanting pacemakers or other devices; it was considered a side effect. With the approach studied in this new trial, phrenic-nerve stimulation is the goal.

|
Dr. Frank Ruschitzka |
I see inherent drawbacks with a treatment that requires implantation of a device. Implanted devices never deliver an unmitigated good; there is always a downside. I am very interested in seeing more complete safety data to better judge the potential risks from this treatment. The report from the SERVE-HF trial last year (N Engl J Med. 2015 Sept 17;373[12]:1095-1105), which showed excess mortality from a different treatment for central sleep apnea, further highlighted the importance of fully evaluating safety when treating central sleep apnea.
Another important issue is does the reduction in apnea-hypopnea index seen in this trial translate into a clinically-meaningful benefit? Ultimately that is what is important, but so far it hasn’t been shown. The treatment did, on average, decrease the apnea-hypopnea index, but does it decrease outcomes like hospitalizations or deaths? We need to see is more data specifically in heart failure patients. When judging the value of a device what we want to see are no safety concerns and fewer clinical events.
Dr. Frank Ruschitzka is professor of cardiology and head of the heart failure unit at University Hospital in Zurich. He made these comments in an interview. He had no disclosures.
This is a really interesting and provocative study that tests a concept that flies in the face of conventional wisdom. In the past, we tried our best to avoid phrenic nerve stimulation when implanting pacemakers or other devices; it was considered a side effect. With the approach studied in this new trial, phrenic-nerve stimulation is the goal.

|
Dr. Frank Ruschitzka |
I see inherent drawbacks with a treatment that requires implantation of a device. Implanted devices never deliver an unmitigated good; there is always a downside. I am very interested in seeing more complete safety data to better judge the potential risks from this treatment. The report from the SERVE-HF trial last year (N Engl J Med. 2015 Sept 17;373[12]:1095-1105), which showed excess mortality from a different treatment for central sleep apnea, further highlighted the importance of fully evaluating safety when treating central sleep apnea.
Another important issue is does the reduction in apnea-hypopnea index seen in this trial translate into a clinically-meaningful benefit? Ultimately that is what is important, but so far it hasn’t been shown. The treatment did, on average, decrease the apnea-hypopnea index, but does it decrease outcomes like hospitalizations or deaths? We need to see is more data specifically in heart failure patients. When judging the value of a device what we want to see are no safety concerns and fewer clinical events.
Dr. Frank Ruschitzka is professor of cardiology and head of the heart failure unit at University Hospital in Zurich. He made these comments in an interview. He had no disclosures.
This is a really interesting and provocative study that tests a concept that flies in the face of conventional wisdom. In the past, we tried our best to avoid phrenic nerve stimulation when implanting pacemakers or other devices; it was considered a side effect. With the approach studied in this new trial, phrenic-nerve stimulation is the goal.

|
Dr. Frank Ruschitzka |
I see inherent drawbacks with a treatment that requires implantation of a device. Implanted devices never deliver an unmitigated good; there is always a downside. I am very interested in seeing more complete safety data to better judge the potential risks from this treatment. The report from the SERVE-HF trial last year (N Engl J Med. 2015 Sept 17;373[12]:1095-1105), which showed excess mortality from a different treatment for central sleep apnea, further highlighted the importance of fully evaluating safety when treating central sleep apnea.
Another important issue is does the reduction in apnea-hypopnea index seen in this trial translate into a clinically-meaningful benefit? Ultimately that is what is important, but so far it hasn’t been shown. The treatment did, on average, decrease the apnea-hypopnea index, but does it decrease outcomes like hospitalizations or deaths? We need to see is more data specifically in heart failure patients. When judging the value of a device what we want to see are no safety concerns and fewer clinical events.
Dr. Frank Ruschitzka is professor of cardiology and head of the heart failure unit at University Hospital in Zurich. He made these comments in an interview. He had no disclosures.
FLORENCE, ITALY – In patients with moderate to severe central sleep apnea, an implanted device that stimulates the phrenic nerve to optimize diaphragm-driven breathing met its efficacy and safety goals, based on results from a multicenter, controlled trial with 151 patients.
Among the 68 patients randomized to active treatment with the device and available for follow-up after 6 months on treatment, 35 patients (51%) had a 50% or better reduction in their apnea-hypopnea index compared with 8 of 73 patients (11%) who had this level of response following device implantation but without its active use.
This statistically significant difference in response to the study’s primary endpoint should pave the way for the device’s approval, Dr. Maria Rosa Costanzo said at a meeting held by the Heart Failure Association of the European Society of Cardiology.
Although the trial enrolled patients with a mix of disorders that caused their central sleep apnea, the majority, 80 patients, had heart failure. Other enrollees had their breathing disorder secondary to atrial fibrillation, hypertension, and other diseases, suggesting that the implanted device, called the remede System, is suitable for patients with moderate to severe central sleep apnea regardless of the etiology, said Dr. Costanzo, medical director for heart failure at the Advocate Medical Group in Naperville, Ill. Among the 80 heart failure patients in the trial, the percentage of patients on active treatment who had a 50% or better reduction in their apnea-hypopnea index closely matched the rate in the entire study group.
The results also demonstrated the treatment’s safety, with a 9% rate of serious adverse events secondary to either the device’s implantation or function during the 12 months following placement in all 151 patients enrolled. Patients in the control arm had a device implanted but not turned on during the first 6 months of the study. The device was turned on and they received active treatment during the next 6 months. The trial’s prespecified safety goal, developed in conjunction with the Food and Drug Administration, was a 1-year rate of freedom from a serious adverse event of at least 80%; the actual rate achieved was 91%.
Successful implantation of the device by electrophysiology cardiologists occurred in 97% of enrolled patients, a procedure that took an average of nearly 3 hours. Need for a lead revision, one of the serious adverse events tallied during follow-up, occurred in 3% of patients. No patients in the study died during 1-year follow-up. Most other serious adverse events involved lead reposition (but not revision) to better optimize the phrenic nerve stimulation. Dr. Costanzo likened the complexity of implanting and operating the device to placement and use of a cardiac resynchronization device.
The efficacy and safety of the device shown in this pivotal trial “should be plenty” for obtaining FDA approval, predicted Dr. Costanzo, the study’s lead investigator, which would make it the first approved intervention for central sleep apnea. “I think this is a game changer,” she said in an interview.
But coming less than a year after a report of an unexpected excess-mortality rate in heart failure patients treated for central sleep apnea with an adaptive servo-ventilation device (N Engl J Med. 2015 Sept 17;373[12]:1095-1105), heart-failure specialists are now more demanding about the data needed to prove safety and clinical benefit from an intervention that targets central sleep apnea and sleep-disordered breathing.
“I think we need an endpoint that involves hospitalizations and death” to more clearly demonstrate meaningful clinical benefit and safety, said Dr. Mariell Jessup, a professor of medicine and heart failure specialist at the University of Pennsylvania in Philadelphia. Following the experience with increased mortality from servo-ventilation “we now need to demand” more comprehensive safety data in sleep trials. Also, the approach tested in this study involves “putting a device into patients, so it’s not completely benign,” she said in an interview. “A lot of things that we thought made a lot of sense, like treating a heart-failure patient’s sleep apnea, turned out to cause things we didn’t expect. We need to be cautious.”
Dr. Costanzo agreed that there is need for additional studies of the phrenic-nerve stimulating device in larger number of heart failure patients that involve heart-failure-specific endpoints. But she also stressed how life changing this intervention was for some of the patients in the study. “The transformation of their lives was unbelievable. They said things like ‘I feel I have my life back.’ ”
She also stressed that the mechanism of action of phrenic nerve stimulation is dramatically different from more traditional sleep-apnea treatments that have relied on positive air pressure devices.
Phrenic nerve stimulation causes contraction of a patient’s diaphragm that creates negative pressure within the chest cavity in a manner similar to that of natural breathing. The stimulation is adjusted to make it imperceptible to patients, and stimulation does not occur when a patient is standing or sitting, only when lying down. “With positive airway pressure in patients with advanced heart failure you reduce venous return, and when a patient’s heart is sick and depends on preload this can hurt the patient. Phrenic nerve stimulation does the opposite. It contracts the diaphragm and creates negative pressure, so it anything it facilitates venous return,” she explained.
The trial, run at 31 centers, mostly in the United States with the others in Europe, enrolled patients with moderate to severe central sleep apnea with an average apnea-hypopnea index of 45 episodes per hour while sleeping. The average age was 65 years, about 90% of the patients were men, and average body mass index was 31 kg/m2.
In addition to the primary efficacy endpoint of reduced apnea-hypopnea index, the patients on active treatment also showed statistically significant reductions compared with baseline in central apnea episodes and in daytime sleepiness measured on the Epworth Sleepiness Scale and an improvement in the patients’ global assessment of their condition. The changes did not occur in the control patients. In the treated patients central apnea episodes fell from an average of 32 episodes per hour at baseline to an average of 6 central episodes an hour after 6 months on treatment.
Dr. Costanzo is a consultant to and has received research support from Respicardia, the company developing the tested phrenic-nerve stimulation device. Dr. Jessup had no disclosures.
On Twitter @mitchelzoler
FLORENCE, ITALY – In patients with moderate to severe central sleep apnea, an implanted device that stimulates the phrenic nerve to optimize diaphragm-driven breathing met its efficacy and safety goals, based on results from a multicenter, controlled trial with 151 patients.
Among the 68 patients randomized to active treatment with the device and available for follow-up after 6 months on treatment, 35 patients (51%) had a 50% or better reduction in their apnea-hypopnea index compared with 8 of 73 patients (11%) who had this level of response following device implantation but without its active use.
This statistically significant difference in response to the study’s primary endpoint should pave the way for the device’s approval, Dr. Maria Rosa Costanzo said at a meeting held by the Heart Failure Association of the European Society of Cardiology.
Although the trial enrolled patients with a mix of disorders that caused their central sleep apnea, the majority, 80 patients, had heart failure. Other enrollees had their breathing disorder secondary to atrial fibrillation, hypertension, and other diseases, suggesting that the implanted device, called the remede System, is suitable for patients with moderate to severe central sleep apnea regardless of the etiology, said Dr. Costanzo, medical director for heart failure at the Advocate Medical Group in Naperville, Ill. Among the 80 heart failure patients in the trial, the percentage of patients on active treatment who had a 50% or better reduction in their apnea-hypopnea index closely matched the rate in the entire study group.
The results also demonstrated the treatment’s safety, with a 9% rate of serious adverse events secondary to either the device’s implantation or function during the 12 months following placement in all 151 patients enrolled. Patients in the control arm had a device implanted but not turned on during the first 6 months of the study. The device was turned on and they received active treatment during the next 6 months. The trial’s prespecified safety goal, developed in conjunction with the Food and Drug Administration, was a 1-year rate of freedom from a serious adverse event of at least 80%; the actual rate achieved was 91%.
Successful implantation of the device by electrophysiology cardiologists occurred in 97% of enrolled patients, a procedure that took an average of nearly 3 hours. Need for a lead revision, one of the serious adverse events tallied during follow-up, occurred in 3% of patients. No patients in the study died during 1-year follow-up. Most other serious adverse events involved lead reposition (but not revision) to better optimize the phrenic nerve stimulation. Dr. Costanzo likened the complexity of implanting and operating the device to placement and use of a cardiac resynchronization device.
The efficacy and safety of the device shown in this pivotal trial “should be plenty” for obtaining FDA approval, predicted Dr. Costanzo, the study’s lead investigator, which would make it the first approved intervention for central sleep apnea. “I think this is a game changer,” she said in an interview.
But coming less than a year after a report of an unexpected excess-mortality rate in heart failure patients treated for central sleep apnea with an adaptive servo-ventilation device (N Engl J Med. 2015 Sept 17;373[12]:1095-1105), heart-failure specialists are now more demanding about the data needed to prove safety and clinical benefit from an intervention that targets central sleep apnea and sleep-disordered breathing.
“I think we need an endpoint that involves hospitalizations and death” to more clearly demonstrate meaningful clinical benefit and safety, said Dr. Mariell Jessup, a professor of medicine and heart failure specialist at the University of Pennsylvania in Philadelphia. Following the experience with increased mortality from servo-ventilation “we now need to demand” more comprehensive safety data in sleep trials. Also, the approach tested in this study involves “putting a device into patients, so it’s not completely benign,” she said in an interview. “A lot of things that we thought made a lot of sense, like treating a heart-failure patient’s sleep apnea, turned out to cause things we didn’t expect. We need to be cautious.”
Dr. Costanzo agreed that there is need for additional studies of the phrenic-nerve stimulating device in larger number of heart failure patients that involve heart-failure-specific endpoints. But she also stressed how life changing this intervention was for some of the patients in the study. “The transformation of their lives was unbelievable. They said things like ‘I feel I have my life back.’ ”
She also stressed that the mechanism of action of phrenic nerve stimulation is dramatically different from more traditional sleep-apnea treatments that have relied on positive air pressure devices.
Phrenic nerve stimulation causes contraction of a patient’s diaphragm that creates negative pressure within the chest cavity in a manner similar to that of natural breathing. The stimulation is adjusted to make it imperceptible to patients, and stimulation does not occur when a patient is standing or sitting, only when lying down. “With positive airway pressure in patients with advanced heart failure you reduce venous return, and when a patient’s heart is sick and depends on preload this can hurt the patient. Phrenic nerve stimulation does the opposite. It contracts the diaphragm and creates negative pressure, so it anything it facilitates venous return,” she explained.
The trial, run at 31 centers, mostly in the United States with the others in Europe, enrolled patients with moderate to severe central sleep apnea with an average apnea-hypopnea index of 45 episodes per hour while sleeping. The average age was 65 years, about 90% of the patients were men, and average body mass index was 31 kg/m2.
In addition to the primary efficacy endpoint of reduced apnea-hypopnea index, the patients on active treatment also showed statistically significant reductions compared with baseline in central apnea episodes and in daytime sleepiness measured on the Epworth Sleepiness Scale and an improvement in the patients’ global assessment of their condition. The changes did not occur in the control patients. In the treated patients central apnea episodes fell from an average of 32 episodes per hour at baseline to an average of 6 central episodes an hour after 6 months on treatment.
Dr. Costanzo is a consultant to and has received research support from Respicardia, the company developing the tested phrenic-nerve stimulation device. Dr. Jessup had no disclosures.
On Twitter @mitchelzoler
AT HEART FAILURE 2016
Key clinical point: An implanted device that cuts patients’ apnea-hypopnea index was safe and effective, based on results from a pivotal trial.
Major finding: A 50% or greater reduction in apnea-hypopnea index was seen in 51% of patients who received a phrenic-nerve stimulation device and 11% of control patients.
Data source: Randomized, multicenter trial with 151 patients with central sleep apnea of various etiologies.
Disclosures: Dr. Costanzo is a consultant to and has received research support from Respicardia, the company developing the tested phrenic-nerve stimulation device. Dr. Jessup had no disclosures.
Nitroxl prodrug shows promise in acute heart failure
FLORENCE, ITALY – A novel intravenous prodrug that results in formation of nitroxyl once inside the body showed several potentially beneficial hemodynamic effects during a single, 6-hour infusion in a controlled proof-of-concept study with 46 patients hospitalized with advanced heart failure with reduced ejection fraction.
While receiving the drug, patients showed “statistically significant and clinically meaningful” reductions in pulmonary capillary wedge pressure and in pulmonary artery diastolic pressure, two of the studies three primary endpoints, Dr. Veselin Mitrovic said at a meeting held by the Heart Failure Association of the ESC.
For the study’s third primary endpoint, a change in cardiac index, treatment with the drug led to increased cardiac output using noninvasive measures, especially at the highest tested dose, as well as in all of the subset of treated patients in whom cardiac index was measured by thermodilution.
On the safety side, the drug appeared safe and well tolerated at all four tested doses, while causing no episodes of symptomatic hypotension and no increase in heart rate. Transient, asymptomatic reductions in blood pressure were similar in the treated and control patients.
“This is a very interesting and exciting drug that went in the right direction,” summed up Dr. Mitrovic, professor of cardiology at Goethe University in Frankfurt, Germany, and head of the department of cardiovascular research at the Kerckhoff Clinic in Bad Nauheim, Germany.
“This is the first demonstration of safety and preliminary efficacy in patients with advanced heart failure,” he said in an interview. “We had a very good clinical signal in a relatively small study. We now need a larger study.”
The drug “improved myocardial function in several ways: inotropy, lusitropy, and unloading. It also causes arterial vasodilation and increased stroke volume,” Dr. Mitrovic said. “Other drugs with a positive inotropic effect increase myocardial oxygen consumption; but with this drug, we see a neutral effect on mixed venous oxygen saturation. It balances myocardial oxygen consumption by its effect on unloading and reduced vascular resistance. This is a big advantage.”
Each molecule of nitroxyl is made from single atoms of hydrogen, nitrogen, and oxygen, and its physiologic action is distinct from nitric monoxide. Nitroxyl improves calcium efficiency and recycling without producing intracellular calcium overload, and its effects are not mediated by cyclic AMP or cyclic GMP.
The dose-ranging study enrolled 34 patients with New York Heart Association class III heart failure and 11 with class IV. All patients had to have a left ventricular ejection fraction of 40% or less, and their actual ejection fractions averaged about 25%.
When measured after 2, 4, and 6 hours of infusion, pulmonary capillary wedge pressure (PCWP) fell by an average of about 5 mm Hg, compared with baseline, among patients who received the three highest-dose infusions of the nitroxyl prodrug, known as CXL-1427, and by an average of about 3 mm Hg in those who received the lowest dose.
That effect had disappeared when PCWP was remeasured 2 hours after the end of the 6-hour infusion, and the 11 patients randomized to placebo showed no change at any time point in PCWP. The average declines in PCWP in each of the four dose groups were statistically significant changes, compared with the control patients.
All the drug-treated patients showed an average drop in pulmonary artery systolic pressure of about 5 mm Hg, compared with baseline, and about 3-4 mm Hg in pulmonary artery diastolic pressure. The patients who received the highest dose of CXL-1427 had an average drop in their right artery pressure of about 4 mm Hg. All of those decreases were statistically significant, compared with the lack of any measurable changes in the control patients.
Total peripheral resistance also showed statistically significant declines relative to baseline in all the treated patients when these decreases were compared with the controls.
CXL-1427 was initially developed by Cardioxyl Pharmaceuticals. Bristol-Myers Squibb acquired the company in late 2015. The study was sponsored by Cardioxyl. Dr. Mitrovic has been a consultant to Bayer, Cardiorentis, and Novartis.
On Twitter @mitchelzoler
Nitroxyl is a very promising and interesting drug. It had an effect on both contractility and inotropy, and also affected diastolic function and reduced afterload. The study’s inclusion criteria enrolled patients who are typical for acute heart failure. It is very important to conduct these sorts of hemodynamic studies of a drug’s effect in this setting.
This drug is obviously very powerful in reducing pulmonary capillary wedge pressure; only about 20% of patients did not respond. It also reduced both systolic and diastolic pulmonary artery pressure and right artery pressure, suggesting that it has a powerful effect on contractility in a way that not only affects the periphery by reducing systolic and diastolic pressures, but also produced little change in heart rate. What is important is that this drug acts at multiple points in the cardiovascular system.
The drug’s safety and tolerability looked very good, but it needs to undergo further study.
Dr. Petar M. Seferovic is a professor of cardiology at Belgrade University, Serbia. He made these comments as designated discussant for the report. He has been a speaker for and consultant to Berlin-Chemie, Boehringer Ingelheim, Pfizer, and Gedeon Richter.
Nitroxyl is a very promising and interesting drug. It had an effect on both contractility and inotropy, and also affected diastolic function and reduced afterload. The study’s inclusion criteria enrolled patients who are typical for acute heart failure. It is very important to conduct these sorts of hemodynamic studies of a drug’s effect in this setting.
This drug is obviously very powerful in reducing pulmonary capillary wedge pressure; only about 20% of patients did not respond. It also reduced both systolic and diastolic pulmonary artery pressure and right artery pressure, suggesting that it has a powerful effect on contractility in a way that not only affects the periphery by reducing systolic and diastolic pressures, but also produced little change in heart rate. What is important is that this drug acts at multiple points in the cardiovascular system.
The drug’s safety and tolerability looked very good, but it needs to undergo further study.
Dr. Petar M. Seferovic is a professor of cardiology at Belgrade University, Serbia. He made these comments as designated discussant for the report. He has been a speaker for and consultant to Berlin-Chemie, Boehringer Ingelheim, Pfizer, and Gedeon Richter.
Nitroxyl is a very promising and interesting drug. It had an effect on both contractility and inotropy, and also affected diastolic function and reduced afterload. The study’s inclusion criteria enrolled patients who are typical for acute heart failure. It is very important to conduct these sorts of hemodynamic studies of a drug’s effect in this setting.
This drug is obviously very powerful in reducing pulmonary capillary wedge pressure; only about 20% of patients did not respond. It also reduced both systolic and diastolic pulmonary artery pressure and right artery pressure, suggesting that it has a powerful effect on contractility in a way that not only affects the periphery by reducing systolic and diastolic pressures, but also produced little change in heart rate. What is important is that this drug acts at multiple points in the cardiovascular system.
The drug’s safety and tolerability looked very good, but it needs to undergo further study.
Dr. Petar M. Seferovic is a professor of cardiology at Belgrade University, Serbia. He made these comments as designated discussant for the report. He has been a speaker for and consultant to Berlin-Chemie, Boehringer Ingelheim, Pfizer, and Gedeon Richter.
FLORENCE, ITALY – A novel intravenous prodrug that results in formation of nitroxyl once inside the body showed several potentially beneficial hemodynamic effects during a single, 6-hour infusion in a controlled proof-of-concept study with 46 patients hospitalized with advanced heart failure with reduced ejection fraction.
While receiving the drug, patients showed “statistically significant and clinically meaningful” reductions in pulmonary capillary wedge pressure and in pulmonary artery diastolic pressure, two of the studies three primary endpoints, Dr. Veselin Mitrovic said at a meeting held by the Heart Failure Association of the ESC.
For the study’s third primary endpoint, a change in cardiac index, treatment with the drug led to increased cardiac output using noninvasive measures, especially at the highest tested dose, as well as in all of the subset of treated patients in whom cardiac index was measured by thermodilution.
On the safety side, the drug appeared safe and well tolerated at all four tested doses, while causing no episodes of symptomatic hypotension and no increase in heart rate. Transient, asymptomatic reductions in blood pressure were similar in the treated and control patients.
“This is a very interesting and exciting drug that went in the right direction,” summed up Dr. Mitrovic, professor of cardiology at Goethe University in Frankfurt, Germany, and head of the department of cardiovascular research at the Kerckhoff Clinic in Bad Nauheim, Germany.
“This is the first demonstration of safety and preliminary efficacy in patients with advanced heart failure,” he said in an interview. “We had a very good clinical signal in a relatively small study. We now need a larger study.”
The drug “improved myocardial function in several ways: inotropy, lusitropy, and unloading. It also causes arterial vasodilation and increased stroke volume,” Dr. Mitrovic said. “Other drugs with a positive inotropic effect increase myocardial oxygen consumption; but with this drug, we see a neutral effect on mixed venous oxygen saturation. It balances myocardial oxygen consumption by its effect on unloading and reduced vascular resistance. This is a big advantage.”
Each molecule of nitroxyl is made from single atoms of hydrogen, nitrogen, and oxygen, and its physiologic action is distinct from nitric monoxide. Nitroxyl improves calcium efficiency and recycling without producing intracellular calcium overload, and its effects are not mediated by cyclic AMP or cyclic GMP.
The dose-ranging study enrolled 34 patients with New York Heart Association class III heart failure and 11 with class IV. All patients had to have a left ventricular ejection fraction of 40% or less, and their actual ejection fractions averaged about 25%.
When measured after 2, 4, and 6 hours of infusion, pulmonary capillary wedge pressure (PCWP) fell by an average of about 5 mm Hg, compared with baseline, among patients who received the three highest-dose infusions of the nitroxyl prodrug, known as CXL-1427, and by an average of about 3 mm Hg in those who received the lowest dose.
That effect had disappeared when PCWP was remeasured 2 hours after the end of the 6-hour infusion, and the 11 patients randomized to placebo showed no change at any time point in PCWP. The average declines in PCWP in each of the four dose groups were statistically significant changes, compared with the control patients.
All the drug-treated patients showed an average drop in pulmonary artery systolic pressure of about 5 mm Hg, compared with baseline, and about 3-4 mm Hg in pulmonary artery diastolic pressure. The patients who received the highest dose of CXL-1427 had an average drop in their right artery pressure of about 4 mm Hg. All of those decreases were statistically significant, compared with the lack of any measurable changes in the control patients.
Total peripheral resistance also showed statistically significant declines relative to baseline in all the treated patients when these decreases were compared with the controls.
CXL-1427 was initially developed by Cardioxyl Pharmaceuticals. Bristol-Myers Squibb acquired the company in late 2015. The study was sponsored by Cardioxyl. Dr. Mitrovic has been a consultant to Bayer, Cardiorentis, and Novartis.
On Twitter @mitchelzoler
FLORENCE, ITALY – A novel intravenous prodrug that results in formation of nitroxyl once inside the body showed several potentially beneficial hemodynamic effects during a single, 6-hour infusion in a controlled proof-of-concept study with 46 patients hospitalized with advanced heart failure with reduced ejection fraction.
While receiving the drug, patients showed “statistically significant and clinically meaningful” reductions in pulmonary capillary wedge pressure and in pulmonary artery diastolic pressure, two of the studies three primary endpoints, Dr. Veselin Mitrovic said at a meeting held by the Heart Failure Association of the ESC.
For the study’s third primary endpoint, a change in cardiac index, treatment with the drug led to increased cardiac output using noninvasive measures, especially at the highest tested dose, as well as in all of the subset of treated patients in whom cardiac index was measured by thermodilution.
On the safety side, the drug appeared safe and well tolerated at all four tested doses, while causing no episodes of symptomatic hypotension and no increase in heart rate. Transient, asymptomatic reductions in blood pressure were similar in the treated and control patients.
“This is a very interesting and exciting drug that went in the right direction,” summed up Dr. Mitrovic, professor of cardiology at Goethe University in Frankfurt, Germany, and head of the department of cardiovascular research at the Kerckhoff Clinic in Bad Nauheim, Germany.
“This is the first demonstration of safety and preliminary efficacy in patients with advanced heart failure,” he said in an interview. “We had a very good clinical signal in a relatively small study. We now need a larger study.”
The drug “improved myocardial function in several ways: inotropy, lusitropy, and unloading. It also causes arterial vasodilation and increased stroke volume,” Dr. Mitrovic said. “Other drugs with a positive inotropic effect increase myocardial oxygen consumption; but with this drug, we see a neutral effect on mixed venous oxygen saturation. It balances myocardial oxygen consumption by its effect on unloading and reduced vascular resistance. This is a big advantage.”
Each molecule of nitroxyl is made from single atoms of hydrogen, nitrogen, and oxygen, and its physiologic action is distinct from nitric monoxide. Nitroxyl improves calcium efficiency and recycling without producing intracellular calcium overload, and its effects are not mediated by cyclic AMP or cyclic GMP.
The dose-ranging study enrolled 34 patients with New York Heart Association class III heart failure and 11 with class IV. All patients had to have a left ventricular ejection fraction of 40% or less, and their actual ejection fractions averaged about 25%.
When measured after 2, 4, and 6 hours of infusion, pulmonary capillary wedge pressure (PCWP) fell by an average of about 5 mm Hg, compared with baseline, among patients who received the three highest-dose infusions of the nitroxyl prodrug, known as CXL-1427, and by an average of about 3 mm Hg in those who received the lowest dose.
That effect had disappeared when PCWP was remeasured 2 hours after the end of the 6-hour infusion, and the 11 patients randomized to placebo showed no change at any time point in PCWP. The average declines in PCWP in each of the four dose groups were statistically significant changes, compared with the control patients.
All the drug-treated patients showed an average drop in pulmonary artery systolic pressure of about 5 mm Hg, compared with baseline, and about 3-4 mm Hg in pulmonary artery diastolic pressure. The patients who received the highest dose of CXL-1427 had an average drop in their right artery pressure of about 4 mm Hg. All of those decreases were statistically significant, compared with the lack of any measurable changes in the control patients.
Total peripheral resistance also showed statistically significant declines relative to baseline in all the treated patients when these decreases were compared with the controls.
CXL-1427 was initially developed by Cardioxyl Pharmaceuticals. Bristol-Myers Squibb acquired the company in late 2015. The study was sponsored by Cardioxyl. Dr. Mitrovic has been a consultant to Bayer, Cardiorentis, and Novartis.
On Twitter @mitchelzoler
AT HEART FAILURE 2016
Key clinical point: Intravenous infusion of a nitroxyl prodrug for 6 hours showed safety and several promising hemodynamic effects in a pilot, controlled study with 46 patients with advanced heart failure.
Major finding: Pulmonary capillary wedge pressure fell by about 5 mm Hg throughout a 6-hour infusion at the three highest tested doses of a nitroxyl prodrug.
Data source: A single-center, single-dose study with 46 patients hospitalized with advanced heart failure.
Disclosures: The study was sponsored by Cardioxyl, the drug’s developer, which was recently acquired by Bristol-Myers Squibb. Dr. Mitrovic has been a consultant to Bayer, Cardiorentis, and Novartis.
Guidelines add two new heart failure treatments
Optimal use of two recently approved medications for heart failure has been detailed by the major heart societies in a guideline update.
The American College of Cardiology, the American Heart Association, and the Heart Failure Society of America issued joint recommendations May 20 on the two new medicines for stage C heart failure patients with a reduced ejection fraction.
Valsartan/sacubitril (Entresto, Novartis), is a combination angiotensin receptor–neprilysin inhibitor, the first in a novel class of drugs slugged ARNIs. Ivabradine (Corlanor, Amgen), is a sinoatrial node modulator. Both medicines were approved by the Food and Drug Administration in 2015, though ivabradine has been licensed for a decade in Europe.
Although a comprehensive update to ACC/AHA/HSFA heart failure guidelines is still being developed, the focused update is intended to coincide with the release of new European Society of Cardiology heart failure guidelines, “in order to minimize confusion and improve the care of patients with heart failure,” the societies said in a statement May 20. The recommendations were published online simultaneously in Circulation and the Journal of Cardiac Failure.
The guideline authors, led by Dr. Clyde W. Yancy of Northwestern University in Chicago, recommend that the ARNI replace an ACE inhibitor or an angiotensin II receptor blocker (ARB) for patients who have been tolerating these therapies alongside standard care with a beta-blocker and, for some patients, an aldosterone antagonist as well. The guidelines caution against combining an ARNI with an ACE inhibitor, and against using ARNIs in patients with a history of angioedema.
For patients not suited to treatment with an ARNI, continued use of an ACE inhibitor is recommended. In patients for whom an ACE inhibitor or an ARNI is inappropriate, use of an ARB remains advised. The authors noted that head-to-head comparisons of an ARB versus an ARNI for heart failure do not exist; however, in a randomized, controlled trial in heart failure patients, treatment with valsartan/sacubitril plus standard care reduced cardiovascular death or heart failure hospitalization by 20%, compared with treatment with an ACE inhibitor plus standard care.
Ivabradine, meanwhile, has shown benefit in reducing heart failure hospitalizations in patients with symptomatic, stable, chronic heart failure with reduced ejection fraction who are receiving standard treatment including a beta-blocker, and who are in sinus rhythm with a heart rate of 70 beats per minute or greater at rest.
The new therapies, “when applied judiciously, complement established pharmacological and device-based therapies, representing milestones in the evolution of care for patients with heart failure,” wrote Dr. Elliott M. Antman of Brigham and Women’s Hospital and Harvard Medical School in Boston, Mass., in an editorial accompanying the guidelines.
About half the guideline writing committee members and guideline reviewers disclosed financial relationships with pharmaceutical companies or device manufacturers. Dr. Yancy disclosed no conflicts of interest.
Optimal use of two recently approved medications for heart failure has been detailed by the major heart societies in a guideline update.
The American College of Cardiology, the American Heart Association, and the Heart Failure Society of America issued joint recommendations May 20 on the two new medicines for stage C heart failure patients with a reduced ejection fraction.
Valsartan/sacubitril (Entresto, Novartis), is a combination angiotensin receptor–neprilysin inhibitor, the first in a novel class of drugs slugged ARNIs. Ivabradine (Corlanor, Amgen), is a sinoatrial node modulator. Both medicines were approved by the Food and Drug Administration in 2015, though ivabradine has been licensed for a decade in Europe.
Although a comprehensive update to ACC/AHA/HSFA heart failure guidelines is still being developed, the focused update is intended to coincide with the release of new European Society of Cardiology heart failure guidelines, “in order to minimize confusion and improve the care of patients with heart failure,” the societies said in a statement May 20. The recommendations were published online simultaneously in Circulation and the Journal of Cardiac Failure.
The guideline authors, led by Dr. Clyde W. Yancy of Northwestern University in Chicago, recommend that the ARNI replace an ACE inhibitor or an angiotensin II receptor blocker (ARB) for patients who have been tolerating these therapies alongside standard care with a beta-blocker and, for some patients, an aldosterone antagonist as well. The guidelines caution against combining an ARNI with an ACE inhibitor, and against using ARNIs in patients with a history of angioedema.
For patients not suited to treatment with an ARNI, continued use of an ACE inhibitor is recommended. In patients for whom an ACE inhibitor or an ARNI is inappropriate, use of an ARB remains advised. The authors noted that head-to-head comparisons of an ARB versus an ARNI for heart failure do not exist; however, in a randomized, controlled trial in heart failure patients, treatment with valsartan/sacubitril plus standard care reduced cardiovascular death or heart failure hospitalization by 20%, compared with treatment with an ACE inhibitor plus standard care.
Ivabradine, meanwhile, has shown benefit in reducing heart failure hospitalizations in patients with symptomatic, stable, chronic heart failure with reduced ejection fraction who are receiving standard treatment including a beta-blocker, and who are in sinus rhythm with a heart rate of 70 beats per minute or greater at rest.
The new therapies, “when applied judiciously, complement established pharmacological and device-based therapies, representing milestones in the evolution of care for patients with heart failure,” wrote Dr. Elliott M. Antman of Brigham and Women’s Hospital and Harvard Medical School in Boston, Mass., in an editorial accompanying the guidelines.
About half the guideline writing committee members and guideline reviewers disclosed financial relationships with pharmaceutical companies or device manufacturers. Dr. Yancy disclosed no conflicts of interest.
Optimal use of two recently approved medications for heart failure has been detailed by the major heart societies in a guideline update.
The American College of Cardiology, the American Heart Association, and the Heart Failure Society of America issued joint recommendations May 20 on the two new medicines for stage C heart failure patients with a reduced ejection fraction.
Valsartan/sacubitril (Entresto, Novartis), is a combination angiotensin receptor–neprilysin inhibitor, the first in a novel class of drugs slugged ARNIs. Ivabradine (Corlanor, Amgen), is a sinoatrial node modulator. Both medicines were approved by the Food and Drug Administration in 2015, though ivabradine has been licensed for a decade in Europe.
Although a comprehensive update to ACC/AHA/HSFA heart failure guidelines is still being developed, the focused update is intended to coincide with the release of new European Society of Cardiology heart failure guidelines, “in order to minimize confusion and improve the care of patients with heart failure,” the societies said in a statement May 20. The recommendations were published online simultaneously in Circulation and the Journal of Cardiac Failure.
The guideline authors, led by Dr. Clyde W. Yancy of Northwestern University in Chicago, recommend that the ARNI replace an ACE inhibitor or an angiotensin II receptor blocker (ARB) for patients who have been tolerating these therapies alongside standard care with a beta-blocker and, for some patients, an aldosterone antagonist as well. The guidelines caution against combining an ARNI with an ACE inhibitor, and against using ARNIs in patients with a history of angioedema.
For patients not suited to treatment with an ARNI, continued use of an ACE inhibitor is recommended. In patients for whom an ACE inhibitor or an ARNI is inappropriate, use of an ARB remains advised. The authors noted that head-to-head comparisons of an ARB versus an ARNI for heart failure do not exist; however, in a randomized, controlled trial in heart failure patients, treatment with valsartan/sacubitril plus standard care reduced cardiovascular death or heart failure hospitalization by 20%, compared with treatment with an ACE inhibitor plus standard care.
Ivabradine, meanwhile, has shown benefit in reducing heart failure hospitalizations in patients with symptomatic, stable, chronic heart failure with reduced ejection fraction who are receiving standard treatment including a beta-blocker, and who are in sinus rhythm with a heart rate of 70 beats per minute or greater at rest.
The new therapies, “when applied judiciously, complement established pharmacological and device-based therapies, representing milestones in the evolution of care for patients with heart failure,” wrote Dr. Elliott M. Antman of Brigham and Women’s Hospital and Harvard Medical School in Boston, Mass., in an editorial accompanying the guidelines.
About half the guideline writing committee members and guideline reviewers disclosed financial relationships with pharmaceutical companies or device manufacturers. Dr. Yancy disclosed no conflicts of interest.
FROM CIRCULATION
Rhythm control may be best for atrial fib in HFpEF
CHICAGO – Atrial fibrillation with good heart rate control in patients who have heart failure with preserved ejection fraction is independently associated with exercise intolerance, impaired contractile reserve, and a sharply higher mortality rate than in matched HFpEF patients without the arrhythmia, a retrospective analysis showed.
“Our study, the largest of its kind, provides mechanistic evidence from cardiopulmonary testing that a rhythm control strategy may potentially improve peak exercise capacity and survival in this patient population, a finding that of course requires future prospective appraisal in randomized trials comparing rate and rhythm control of atrial fibrillation in HFpEF,” Dr. Mohamed Badreldin Elshazly reported at the annual meeting of the American College of Cardiology.
In the meantime, his study also shows the useful role cardiopulmonary stress testing can play in the setting of atrial fibrillation (AF) in HFpEF, he added.
“Cardiopulmonary stress tests are cheap and easy to do. They’re a big asset for personalized medicine. Using an objective measure like cardiopulmonary stress testing to define the physiologic and hemodynamic consequences of atrial fibrillation in individual patients may help identify those in whom rhythm control may improve exercise tolerance and quality of life, and those who may be okay with rate control,” according to Dr. Elshazly of the Cleveland Clinic.
He noted that while it’s well established that atrial fibrillation is associated with exercise intolerance in patients with heart failure with reduced ejection fraction (HFrEF) and that restoration of sinus rhythm in such patients has a positive impact on exercise hemodynamics, symptom severity, and quality of life, the situation is murkier regarding AF in patients with HFpEF. Prior studies were generally small and unable to establish whether AF was independently associated with exercise intolerance or if HFpEF patients who developed AF were sicker and higher risk.
He presented a retrospective, case-control study in a cohort of 1,825 patients with HFpEF referred for maximal, symptom-limited cardiopulmonary stress testing at the Cleveland Clinic. Among these were 242 patients with AF. They were extensively propensity matched – including on the basis of heart failure etiology – to 484 HFpEF patients without AF.
“That’s what makes our study strong. We were the first to be able to do propensity matching and therefore account for other risk factors in our analysis,” Dr. Elshazly explained.
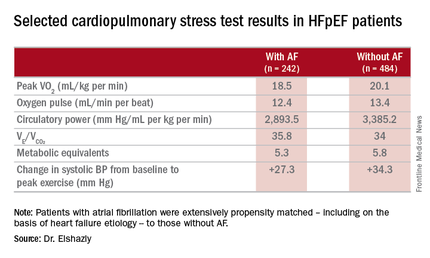
The investigators measured peak oxygen uptake (VO2), the minute ventilation–carbon dioxide production relationship (VE/VCO2) as an indicator of ventilatory efficiency, metabolic equivalents (METS), ventilatory anaerobic threshold, circulatory power as a proxy for cardiac power, peak oxygen pulse as a surrogate for stroke volume, and resting and peak heart rate and systolic blood pressure. The patients with AF were in fibrillation at the time of their cardiopulmonary stress testing.
The HFpEF patients with AF had a mean resting heart rate of 70 beats per minute and a peak rate of 130 bpm. This group showed evidence of impaired peak exercise tolerance as reflected in lower peak VO2, oxygen pulse, and circulatory power at peak exercise. Their VE/VCO2 was higher, indicating impaired ventilatory efficiency. Notably, however, their submaximal exercise capacity was similar to the non-AF controls.
“Atrial fibrillation in these patients is really more of a disease that shows itself in patients when you take them to their peak exercise capacity,” he observed.
All-cause mortality was significantly higher in the AF as compared with no-AF patients with HFpEF. The mortality curves separated early and the divergence grew larger over the course of 8 years of follow-up.
One audience member pointed out that the large mortality difference between the two groups seems disproportionate to the rather modest differences in exercise capacity.
“It brings up an interesting point,” Dr. Elshazly replied. “Maybe the increase in total mortality that we see is being driven by other things besides cardiovascular mortality. Our data doesn’t capture the specific cause of death, be it cancer, for example, but it does raise the idea that this mortality difference is not all driven by cardiovascular mortality, but by atrial fibrillation.”
Dr. Elshazly reported having no financial conflicts of interest regarding his institutionally supported study.
CHICAGO – Atrial fibrillation with good heart rate control in patients who have heart failure with preserved ejection fraction is independently associated with exercise intolerance, impaired contractile reserve, and a sharply higher mortality rate than in matched HFpEF patients without the arrhythmia, a retrospective analysis showed.
“Our study, the largest of its kind, provides mechanistic evidence from cardiopulmonary testing that a rhythm control strategy may potentially improve peak exercise capacity and survival in this patient population, a finding that of course requires future prospective appraisal in randomized trials comparing rate and rhythm control of atrial fibrillation in HFpEF,” Dr. Mohamed Badreldin Elshazly reported at the annual meeting of the American College of Cardiology.
In the meantime, his study also shows the useful role cardiopulmonary stress testing can play in the setting of atrial fibrillation (AF) in HFpEF, he added.
“Cardiopulmonary stress tests are cheap and easy to do. They’re a big asset for personalized medicine. Using an objective measure like cardiopulmonary stress testing to define the physiologic and hemodynamic consequences of atrial fibrillation in individual patients may help identify those in whom rhythm control may improve exercise tolerance and quality of life, and those who may be okay with rate control,” according to Dr. Elshazly of the Cleveland Clinic.
He noted that while it’s well established that atrial fibrillation is associated with exercise intolerance in patients with heart failure with reduced ejection fraction (HFrEF) and that restoration of sinus rhythm in such patients has a positive impact on exercise hemodynamics, symptom severity, and quality of life, the situation is murkier regarding AF in patients with HFpEF. Prior studies were generally small and unable to establish whether AF was independently associated with exercise intolerance or if HFpEF patients who developed AF were sicker and higher risk.
He presented a retrospective, case-control study in a cohort of 1,825 patients with HFpEF referred for maximal, symptom-limited cardiopulmonary stress testing at the Cleveland Clinic. Among these were 242 patients with AF. They were extensively propensity matched – including on the basis of heart failure etiology – to 484 HFpEF patients without AF.
“That’s what makes our study strong. We were the first to be able to do propensity matching and therefore account for other risk factors in our analysis,” Dr. Elshazly explained.
The investigators measured peak oxygen uptake (VO2), the minute ventilation–carbon dioxide production relationship (VE/VCO2) as an indicator of ventilatory efficiency, metabolic equivalents (METS), ventilatory anaerobic threshold, circulatory power as a proxy for cardiac power, peak oxygen pulse as a surrogate for stroke volume, and resting and peak heart rate and systolic blood pressure. The patients with AF were in fibrillation at the time of their cardiopulmonary stress testing.
The HFpEF patients with AF had a mean resting heart rate of 70 beats per minute and a peak rate of 130 bpm. This group showed evidence of impaired peak exercise tolerance as reflected in lower peak VO2, oxygen pulse, and circulatory power at peak exercise. Their VE/VCO2 was higher, indicating impaired ventilatory efficiency. Notably, however, their submaximal exercise capacity was similar to the non-AF controls.

“Atrial fibrillation in these patients is really more of a disease that shows itself in patients when you take them to their peak exercise capacity,” he observed.
All-cause mortality was significantly higher in the AF as compared with no-AF patients with HFpEF. The mortality curves separated early and the divergence grew larger over the course of 8 years of follow-up.
One audience member pointed out that the large mortality difference between the two groups seems disproportionate to the rather modest differences in exercise capacity.
“It brings up an interesting point,” Dr. Elshazly replied. “Maybe the increase in total mortality that we see is being driven by other things besides cardiovascular mortality. Our data doesn’t capture the specific cause of death, be it cancer, for example, but it does raise the idea that this mortality difference is not all driven by cardiovascular mortality, but by atrial fibrillation.”
Dr. Elshazly reported having no financial conflicts of interest regarding his institutionally supported study.
CHICAGO – Atrial fibrillation with good heart rate control in patients who have heart failure with preserved ejection fraction is independently associated with exercise intolerance, impaired contractile reserve, and a sharply higher mortality rate than in matched HFpEF patients without the arrhythmia, a retrospective analysis showed.
“Our study, the largest of its kind, provides mechanistic evidence from cardiopulmonary testing that a rhythm control strategy may potentially improve peak exercise capacity and survival in this patient population, a finding that of course requires future prospective appraisal in randomized trials comparing rate and rhythm control of atrial fibrillation in HFpEF,” Dr. Mohamed Badreldin Elshazly reported at the annual meeting of the American College of Cardiology.
In the meantime, his study also shows the useful role cardiopulmonary stress testing can play in the setting of atrial fibrillation (AF) in HFpEF, he added.
“Cardiopulmonary stress tests are cheap and easy to do. They’re a big asset for personalized medicine. Using an objective measure like cardiopulmonary stress testing to define the physiologic and hemodynamic consequences of atrial fibrillation in individual patients may help identify those in whom rhythm control may improve exercise tolerance and quality of life, and those who may be okay with rate control,” according to Dr. Elshazly of the Cleveland Clinic.
He noted that while it’s well established that atrial fibrillation is associated with exercise intolerance in patients with heart failure with reduced ejection fraction (HFrEF) and that restoration of sinus rhythm in such patients has a positive impact on exercise hemodynamics, symptom severity, and quality of life, the situation is murkier regarding AF in patients with HFpEF. Prior studies were generally small and unable to establish whether AF was independently associated with exercise intolerance or if HFpEF patients who developed AF were sicker and higher risk.
He presented a retrospective, case-control study in a cohort of 1,825 patients with HFpEF referred for maximal, symptom-limited cardiopulmonary stress testing at the Cleveland Clinic. Among these were 242 patients with AF. They were extensively propensity matched – including on the basis of heart failure etiology – to 484 HFpEF patients without AF.
“That’s what makes our study strong. We were the first to be able to do propensity matching and therefore account for other risk factors in our analysis,” Dr. Elshazly explained.
The investigators measured peak oxygen uptake (VO2), the minute ventilation–carbon dioxide production relationship (VE/VCO2) as an indicator of ventilatory efficiency, metabolic equivalents (METS), ventilatory anaerobic threshold, circulatory power as a proxy for cardiac power, peak oxygen pulse as a surrogate for stroke volume, and resting and peak heart rate and systolic blood pressure. The patients with AF were in fibrillation at the time of their cardiopulmonary stress testing.
The HFpEF patients with AF had a mean resting heart rate of 70 beats per minute and a peak rate of 130 bpm. This group showed evidence of impaired peak exercise tolerance as reflected in lower peak VO2, oxygen pulse, and circulatory power at peak exercise. Their VE/VCO2 was higher, indicating impaired ventilatory efficiency. Notably, however, their submaximal exercise capacity was similar to the non-AF controls.

“Atrial fibrillation in these patients is really more of a disease that shows itself in patients when you take them to their peak exercise capacity,” he observed.
All-cause mortality was significantly higher in the AF as compared with no-AF patients with HFpEF. The mortality curves separated early and the divergence grew larger over the course of 8 years of follow-up.
One audience member pointed out that the large mortality difference between the two groups seems disproportionate to the rather modest differences in exercise capacity.
“It brings up an interesting point,” Dr. Elshazly replied. “Maybe the increase in total mortality that we see is being driven by other things besides cardiovascular mortality. Our data doesn’t capture the specific cause of death, be it cancer, for example, but it does raise the idea that this mortality difference is not all driven by cardiovascular mortality, but by atrial fibrillation.”
Dr. Elshazly reported having no financial conflicts of interest regarding his institutionally supported study.
AT ACC 16
Key clinical point: Atrial fibrillation in patients with heart failure with preserved ejection fraction is associated with exercise intolerance and increased mortality.
Major finding: Mean peak VO2 was 18.5 mL/kg per minute in patients with HFpEF and atrial fibrillation, significantly less than the 20.1 mL/kg per minute in controls.
Data source: A retrospective, single-institution study of cardiopulmonary stress test findings and 8-year mortality in 242 patients with HFpEF and atrial fibrillation and 484 propensity-matched controls with HFpEF and no arrhythmia.
Disclosures: The presenter reported having no financial conflicts of interest regarding his institutionally supported study.
Prompt antidepressant treatment swiftly chops cardiovascular risk
CHICAGO – Prompt, effective treatment for depression in the primary care setting appears to swiftly reduce the elevated cardiovascular risk known to be tied to the mood disorder, Heidi Thomas May, Ph.D., reported at the annual meeting of the American College of Cardiology.
“We know that depression is a risk factor for long-term adverse cardiovascular outcomes. Our study shows that it can also have immediate effects on someone’s cardiovascular health. I think our study highlights the importance of screening for depression in the primary care setting – and if someone’s depressed, they need to be treated,” said Dr. May, a cardiovascular and genetic epidemiologist at Intermountain Medical Center in Murray, Utah.
She presented an observational study of the electronic medical records of 7,559 Intermountain Healthcare patients over age 40 years who completed the Patient Health Questionnaire-9 (PHQ-9) depression screening tool during a visit to an Intermountain primary care clinic for any reason. They completed another PHQ-9 a median of 2.7 years later. Under the Intermountain system, a PHQ-9 score of 10 or more triggers implementation of a depression treatment pathway, the specifics of which vary depending upon the severity of symptoms.
On the basis of their two PHQ-9 scores, all patients were classified into one of four groups: The “nondepressed” group of 3,286 patients had a score of 9 or less on both occasions; the “remained depressed” cohort of 1,987 patients scored 10 or more on both PHQ-9s; the “no longer depressed” group of 1,542 patients scored at least 10 but subsequently improved by at least 5 points to a score of 9 or less; and the 735 patients in the “became depressed” group first scored 9 or less on the PHQ-9 but subsequently had at least a 5-point increase to a score of 10 or more.
The subjects were then followed for major adverse cardiovascular events, or MACE – defined as a composite of death, diagnosis of coronary artery disease, acute MI, stroke, and heart failure hospitalization – for a median of 208 days after completing their second PHQ-9.
The MACE rate was 4.8% in the nondepressed group and similar at 4.6% in the “no longer depressed” group, Dr. May reported. Both groups fared significantly better than the “remained depressed” and “became depressed” groups, which had MACE rates of 6% and 6.4%, respectively.
In a multivariate regression analysis adjusted for demographics, cardiovascular risk factors, prior disease diagnoses, medications, and other potential confounders, the “remained depressed” group was 33% more likely to experience a cardiovascular event than was the nondepressed group, she said. The “became depressed” group had a 44% increase in risk, compared with the nondepressed individuals. In contrast, the MACE risk in patients in the “no longer depressed” group was not significantly different from that of patients who weren’t depressed at either time point. And the MACE risk of patients who became depressed during the course of the study was no different from that of patients who remained depressed at both time points.
This is the first study of its kind, Dr. May said. Hence, the results require confirmation, ideally in a randomized clinical trial.
She reported having no financial conflicts regarding the study, which was supported by Intermountain Healthcare.
CHICAGO – Prompt, effective treatment for depression in the primary care setting appears to swiftly reduce the elevated cardiovascular risk known to be tied to the mood disorder, Heidi Thomas May, Ph.D., reported at the annual meeting of the American College of Cardiology.
“We know that depression is a risk factor for long-term adverse cardiovascular outcomes. Our study shows that it can also have immediate effects on someone’s cardiovascular health. I think our study highlights the importance of screening for depression in the primary care setting – and if someone’s depressed, they need to be treated,” said Dr. May, a cardiovascular and genetic epidemiologist at Intermountain Medical Center in Murray, Utah.
She presented an observational study of the electronic medical records of 7,559 Intermountain Healthcare patients over age 40 years who completed the Patient Health Questionnaire-9 (PHQ-9) depression screening tool during a visit to an Intermountain primary care clinic for any reason. They completed another PHQ-9 a median of 2.7 years later. Under the Intermountain system, a PHQ-9 score of 10 or more triggers implementation of a depression treatment pathway, the specifics of which vary depending upon the severity of symptoms.
On the basis of their two PHQ-9 scores, all patients were classified into one of four groups: The “nondepressed” group of 3,286 patients had a score of 9 or less on both occasions; the “remained depressed” cohort of 1,987 patients scored 10 or more on both PHQ-9s; the “no longer depressed” group of 1,542 patients scored at least 10 but subsequently improved by at least 5 points to a score of 9 or less; and the 735 patients in the “became depressed” group first scored 9 or less on the PHQ-9 but subsequently had at least a 5-point increase to a score of 10 or more.
The subjects were then followed for major adverse cardiovascular events, or MACE – defined as a composite of death, diagnosis of coronary artery disease, acute MI, stroke, and heart failure hospitalization – for a median of 208 days after completing their second PHQ-9.
The MACE rate was 4.8% in the nondepressed group and similar at 4.6% in the “no longer depressed” group, Dr. May reported. Both groups fared significantly better than the “remained depressed” and “became depressed” groups, which had MACE rates of 6% and 6.4%, respectively.
In a multivariate regression analysis adjusted for demographics, cardiovascular risk factors, prior disease diagnoses, medications, and other potential confounders, the “remained depressed” group was 33% more likely to experience a cardiovascular event than was the nondepressed group, she said. The “became depressed” group had a 44% increase in risk, compared with the nondepressed individuals. In contrast, the MACE risk in patients in the “no longer depressed” group was not significantly different from that of patients who weren’t depressed at either time point. And the MACE risk of patients who became depressed during the course of the study was no different from that of patients who remained depressed at both time points.
This is the first study of its kind, Dr. May said. Hence, the results require confirmation, ideally in a randomized clinical trial.
She reported having no financial conflicts regarding the study, which was supported by Intermountain Healthcare.
CHICAGO – Prompt, effective treatment for depression in the primary care setting appears to swiftly reduce the elevated cardiovascular risk known to be tied to the mood disorder, Heidi Thomas May, Ph.D., reported at the annual meeting of the American College of Cardiology.
“We know that depression is a risk factor for long-term adverse cardiovascular outcomes. Our study shows that it can also have immediate effects on someone’s cardiovascular health. I think our study highlights the importance of screening for depression in the primary care setting – and if someone’s depressed, they need to be treated,” said Dr. May, a cardiovascular and genetic epidemiologist at Intermountain Medical Center in Murray, Utah.
She presented an observational study of the electronic medical records of 7,559 Intermountain Healthcare patients over age 40 years who completed the Patient Health Questionnaire-9 (PHQ-9) depression screening tool during a visit to an Intermountain primary care clinic for any reason. They completed another PHQ-9 a median of 2.7 years later. Under the Intermountain system, a PHQ-9 score of 10 or more triggers implementation of a depression treatment pathway, the specifics of which vary depending upon the severity of symptoms.
On the basis of their two PHQ-9 scores, all patients were classified into one of four groups: The “nondepressed” group of 3,286 patients had a score of 9 or less on both occasions; the “remained depressed” cohort of 1,987 patients scored 10 or more on both PHQ-9s; the “no longer depressed” group of 1,542 patients scored at least 10 but subsequently improved by at least 5 points to a score of 9 or less; and the 735 patients in the “became depressed” group first scored 9 or less on the PHQ-9 but subsequently had at least a 5-point increase to a score of 10 or more.
The subjects were then followed for major adverse cardiovascular events, or MACE – defined as a composite of death, diagnosis of coronary artery disease, acute MI, stroke, and heart failure hospitalization – for a median of 208 days after completing their second PHQ-9.
The MACE rate was 4.8% in the nondepressed group and similar at 4.6% in the “no longer depressed” group, Dr. May reported. Both groups fared significantly better than the “remained depressed” and “became depressed” groups, which had MACE rates of 6% and 6.4%, respectively.
In a multivariate regression analysis adjusted for demographics, cardiovascular risk factors, prior disease diagnoses, medications, and other potential confounders, the “remained depressed” group was 33% more likely to experience a cardiovascular event than was the nondepressed group, she said. The “became depressed” group had a 44% increase in risk, compared with the nondepressed individuals. In contrast, the MACE risk in patients in the “no longer depressed” group was not significantly different from that of patients who weren’t depressed at either time point. And the MACE risk of patients who became depressed during the course of the study was no different from that of patients who remained depressed at both time points.
This is the first study of its kind, Dr. May said. Hence, the results require confirmation, ideally in a randomized clinical trial.
She reported having no financial conflicts regarding the study, which was supported by Intermountain Healthcare.
AT ACC 16
Key clinical point: Event rate was no different in “no longer depressed” group than in “never depressed.”
Major finding: Major adverse cardiovascular events were 44% more likely in primary care patients who became depressed during a median 2.7-year period, compared with those who weren’t depressed at either time point.
Data source: An observational study of 7,550 patients screened for depression in primary care clinics.
Disclosures: The study was supported by Intermountain Healthcare. Dr. May reported having no financial conflicts of interest.
Exercise is protective but underutilized in atrial fib patients
CHICAGO – Efforts to encourage even modest amounts of physical activity in sedentary patients with atrial fibrillation are likely to pay off in reduced risks of cardiovascular and all-cause mortality, according to a report from the EurObservational Research Program Pilot Survey on Atrial Fibrillation General Registry.
“Clearly we would recommend regular physical activity for patients with atrial fibrillation on the basis of the mortality benefit we see in the registry. If we give patients with atrial fibrillation oral anticoagulation, they are protected against stroke risk, but clearly they are still dying a lot,” Dr. Marco Proietti said at the annual meeting of the American College of Cardiology.
He presented 1-year follow-up data on 2,442 “real world” patients enrolled in the nine-country, observational, prospective registry, known as EORP-AF, shortly after being diagnosed with AF. One of the goals of EORP-AF is to learn whether physical exercise protects against cardiovascular events and all-cause mortality in AF patients, as has been well established in the general population and in patients at high cardiovascular risk.
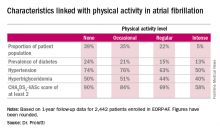
One striking finding was that nearly 40% of patients in EORP-AF reported engaging in no physical activity, defined for study purposes as zero to less than 3 hours of physical activity per week for less than 2 years.
The other three categories employed by investigators were “occasional,” meaning less than 3 hours per week but for 2 years or more; “regular,” defined as at least 3 hours weekly for at least 2 years; and “intense,” which required more than 7 hours of physical activity per week for at least 2 years. Levels of cardiovascular and stroke risk factors decreased progressively with increasing levels of physical activity. Only 5% of the AF patients met the ‘intense’ standard, noted Dr. Proietti of the University of Birmingham (England).
The 1-year cardiovascular mortality rate approached 6% in the no physical activity group and hovered around 1% in the other three groups. The 1-year all-cause mortality rate exceeded 12% in the no-exercise group, was 4%% in the occasional exercisers, and 1%-2% in the groups reporting regular or intense physical activity.
The 1-year composite endpoint of cardiovascular death, any thromboembolism, or a bleeding event occurred in 12% of the sedentary patients, a rate two-to-three times higher than in the others.
Updated outcomes are to be reported from the EORP-AF pilot registry after 2 and 3 years of follow-up. Meanwhile, on the basis of the success of the pilot registry, more than 10,000 patients with AF have been enrolled in the EORP-AF main registry, according to Dr. Proietti.
A study limitation, he conceded, is that the registry includes no objective measure of physical capacity, such as METS.
Session co-chair Dr. Brian Olshansky, emeritus professor of internal medicine at the University of Iowa, Iowa City, observed that the registry data raise a classic chicken-versus-egg issue: Do the sedentary patients do worse because they’re inactive, or are they inactive because they are sicker and hence have worse outcomes?
Dr. Proietti said the registry data provide some support for the latter idea, since the no-physical-activity group had higher prevalences of coronary artery disease and heart failure.
Dr. Olshansky raised another point: “It’s interesting to me that there’s a whole bunch of literature showing that elite endurance athletes – bike racers, cross country skiers – have a very high incidence of atrial fibrillation. It seems to be either an inflammatory or an autonomic issue.”
Dr. Proietti replied that he’s familiar with that extensive literature, but the EORP-AF data through 1 year don’t provide validation. While the intense physical activity group tended to have more symptomatic AF than the other groups, they were no more likely to show progression from paroxysmal to permanent AF. The much larger main registry now underway may be able to better clarify the relationship between physical activity and incidence and progression of AF, including the possibility of a U-shaped dose-response curve.
The EORP-AF registry is supported by the European Society of Cardiology. Dr. Proietti reported having no financial conflicts of interest.
CHICAGO – Efforts to encourage even modest amounts of physical activity in sedentary patients with atrial fibrillation are likely to pay off in reduced risks of cardiovascular and all-cause mortality, according to a report from the EurObservational Research Program Pilot Survey on Atrial Fibrillation General Registry.
“Clearly we would recommend regular physical activity for patients with atrial fibrillation on the basis of the mortality benefit we see in the registry. If we give patients with atrial fibrillation oral anticoagulation, they are protected against stroke risk, but clearly they are still dying a lot,” Dr. Marco Proietti said at the annual meeting of the American College of Cardiology.
He presented 1-year follow-up data on 2,442 “real world” patients enrolled in the nine-country, observational, prospective registry, known as EORP-AF, shortly after being diagnosed with AF. One of the goals of EORP-AF is to learn whether physical exercise protects against cardiovascular events and all-cause mortality in AF patients, as has been well established in the general population and in patients at high cardiovascular risk.
One striking finding was that nearly 40% of patients in EORP-AF reported engaging in no physical activity, defined for study purposes as zero to less than 3 hours of physical activity per week for less than 2 years.
The other three categories employed by investigators were “occasional,” meaning less than 3 hours per week but for 2 years or more; “regular,” defined as at least 3 hours weekly for at least 2 years; and “intense,” which required more than 7 hours of physical activity per week for at least 2 years. Levels of cardiovascular and stroke risk factors decreased progressively with increasing levels of physical activity. Only 5% of the AF patients met the ‘intense’ standard, noted Dr. Proietti of the University of Birmingham (England).
The 1-year cardiovascular mortality rate approached 6% in the no physical activity group and hovered around 1% in the other three groups. The 1-year all-cause mortality rate exceeded 12% in the no-exercise group, was 4%% in the occasional exercisers, and 1%-2% in the groups reporting regular or intense physical activity.
The 1-year composite endpoint of cardiovascular death, any thromboembolism, or a bleeding event occurred in 12% of the sedentary patients, a rate two-to-three times higher than in the others.
Updated outcomes are to be reported from the EORP-AF pilot registry after 2 and 3 years of follow-up. Meanwhile, on the basis of the success of the pilot registry, more than 10,000 patients with AF have been enrolled in the EORP-AF main registry, according to Dr. Proietti.
A study limitation, he conceded, is that the registry includes no objective measure of physical capacity, such as METS.
Session co-chair Dr. Brian Olshansky, emeritus professor of internal medicine at the University of Iowa, Iowa City, observed that the registry data raise a classic chicken-versus-egg issue: Do the sedentary patients do worse because they’re inactive, or are they inactive because they are sicker and hence have worse outcomes?
Dr. Proietti said the registry data provide some support for the latter idea, since the no-physical-activity group had higher prevalences of coronary artery disease and heart failure.
Dr. Olshansky raised another point: “It’s interesting to me that there’s a whole bunch of literature showing that elite endurance athletes – bike racers, cross country skiers – have a very high incidence of atrial fibrillation. It seems to be either an inflammatory or an autonomic issue.”
Dr. Proietti replied that he’s familiar with that extensive literature, but the EORP-AF data through 1 year don’t provide validation. While the intense physical activity group tended to have more symptomatic AF than the other groups, they were no more likely to show progression from paroxysmal to permanent AF. The much larger main registry now underway may be able to better clarify the relationship between physical activity and incidence and progression of AF, including the possibility of a U-shaped dose-response curve.
The EORP-AF registry is supported by the European Society of Cardiology. Dr. Proietti reported having no financial conflicts of interest.
CHICAGO – Efforts to encourage even modest amounts of physical activity in sedentary patients with atrial fibrillation are likely to pay off in reduced risks of cardiovascular and all-cause mortality, according to a report from the EurObservational Research Program Pilot Survey on Atrial Fibrillation General Registry.
“Clearly we would recommend regular physical activity for patients with atrial fibrillation on the basis of the mortality benefit we see in the registry. If we give patients with atrial fibrillation oral anticoagulation, they are protected against stroke risk, but clearly they are still dying a lot,” Dr. Marco Proietti said at the annual meeting of the American College of Cardiology.
He presented 1-year follow-up data on 2,442 “real world” patients enrolled in the nine-country, observational, prospective registry, known as EORP-AF, shortly after being diagnosed with AF. One of the goals of EORP-AF is to learn whether physical exercise protects against cardiovascular events and all-cause mortality in AF patients, as has been well established in the general population and in patients at high cardiovascular risk.
One striking finding was that nearly 40% of patients in EORP-AF reported engaging in no physical activity, defined for study purposes as zero to less than 3 hours of physical activity per week for less than 2 years.
The other three categories employed by investigators were “occasional,” meaning less than 3 hours per week but for 2 years or more; “regular,” defined as at least 3 hours weekly for at least 2 years; and “intense,” which required more than 7 hours of physical activity per week for at least 2 years. Levels of cardiovascular and stroke risk factors decreased progressively with increasing levels of physical activity. Only 5% of the AF patients met the ‘intense’ standard, noted Dr. Proietti of the University of Birmingham (England).
The 1-year cardiovascular mortality rate approached 6% in the no physical activity group and hovered around 1% in the other three groups. The 1-year all-cause mortality rate exceeded 12% in the no-exercise group, was 4%% in the occasional exercisers, and 1%-2% in the groups reporting regular or intense physical activity.
The 1-year composite endpoint of cardiovascular death, any thromboembolism, or a bleeding event occurred in 12% of the sedentary patients, a rate two-to-three times higher than in the others.
Updated outcomes are to be reported from the EORP-AF pilot registry after 2 and 3 years of follow-up. Meanwhile, on the basis of the success of the pilot registry, more than 10,000 patients with AF have been enrolled in the EORP-AF main registry, according to Dr. Proietti.
A study limitation, he conceded, is that the registry includes no objective measure of physical capacity, such as METS.
Session co-chair Dr. Brian Olshansky, emeritus professor of internal medicine at the University of Iowa, Iowa City, observed that the registry data raise a classic chicken-versus-egg issue: Do the sedentary patients do worse because they’re inactive, or are they inactive because they are sicker and hence have worse outcomes?
Dr. Proietti said the registry data provide some support for the latter idea, since the no-physical-activity group had higher prevalences of coronary artery disease and heart failure.
Dr. Olshansky raised another point: “It’s interesting to me that there’s a whole bunch of literature showing that elite endurance athletes – bike racers, cross country skiers – have a very high incidence of atrial fibrillation. It seems to be either an inflammatory or an autonomic issue.”
Dr. Proietti replied that he’s familiar with that extensive literature, but the EORP-AF data through 1 year don’t provide validation. While the intense physical activity group tended to have more symptomatic AF than the other groups, they were no more likely to show progression from paroxysmal to permanent AF. The much larger main registry now underway may be able to better clarify the relationship between physical activity and incidence and progression of AF, including the possibility of a U-shaped dose-response curve.
The EORP-AF registry is supported by the European Society of Cardiology. Dr. Proietti reported having no financial conflicts of interest.
AT ACC 16
Key clinical point: Atrial fibrillation patients who report engaging in even occasional physical activity have a markedly lower risk of all-cause mortality than those who are sedentary.
Major finding: The 1-year composite outcome of cardiovascular death, any thromboembolism, or a bleeding event occurred in 12% in patients with atrial fibrillation who were sedentary, a rate two to three times greater than in those who engaged in various amounts of physical activity.
Data source: An analysis of 1-year outcomes in 2,442 patients with AF enrolled in the prospective, observational EORP-AF pilot registry.
Disclosures: The EORP-AF registry is supported by the European Society of Cardiology. The presenter reported having no financial conflicts of interest.
Drilling down on end-of-life health care costs in heart failure
CHICAGO – Health care costs for heart failure patients spike dramatically in the last 6 months of life, with lack of adherence to guideline-directed outpatient medical therapy being the major modifiable factor driving the costs, Jason P. Swindle, Ph.D., reported at the annual meeting of the American College of Cardiology.
He presented a retrospective study of heart failure–related and total health care costs during the final 24 months of life for 48,026 Medicare Advantage managed care plan members with heart failure.
The researchers were interested in exploring possible racial/ethnic differences in costs, particularly in light of evidence that African Americans have a higher risk of heart failure and higher all-cause mortality. And while a first look at the data indicated racial differences in the size of end-of-life cost spikes, those differences lost their significance in multivariate analysis.
“Lack of guideline-directed outpatient heart failure therapy was a key. Also older age and the presence of coronary heart disease – those were really the big three items that were driving the spike in costs,” said Dr. Swindle of the Chicago office of Optum, a health care consulting group.
He was quick to add that, since the study was based upon administrative data, the lack of adherence to guideline-directed therapy may be unrelated to physician prescribing.
“We see the prescriptions that patients are actually filling. Patients may very well be seeing their cardiologist and being prescribed a medication, but they simply don’t fill the prescription,” he explained in an interview.
Over patients’ final 2 years of life, semiannual all-cause health care costs climbed from a baseline of roughly $10,000 during months 24-19 before death by about 4-fold during months 6-1 before death. Heart failure–related medical costs jumped 10-fold in Asians, 7.8-fold in Hispanics, 6.6-fold in African Americans, and 6.7-fold in whites. Most of the increases occurred in the final 6 months.
Zeroing in on the final 6 months of life, the mean cumulative total medical costs were $44,599, with heart failure–related medical costs accounting for $24,818 of that figure. Total medical costs averaged just under $5,000 during month 6 prior to death and rose roughly 3.5-fold over the remaining months.
This study was supported by Novartis Pharmaceuticals.
CHICAGO – Health care costs for heart failure patients spike dramatically in the last 6 months of life, with lack of adherence to guideline-directed outpatient medical therapy being the major modifiable factor driving the costs, Jason P. Swindle, Ph.D., reported at the annual meeting of the American College of Cardiology.
He presented a retrospective study of heart failure–related and total health care costs during the final 24 months of life for 48,026 Medicare Advantage managed care plan members with heart failure.
The researchers were interested in exploring possible racial/ethnic differences in costs, particularly in light of evidence that African Americans have a higher risk of heart failure and higher all-cause mortality. And while a first look at the data indicated racial differences in the size of end-of-life cost spikes, those differences lost their significance in multivariate analysis.
“Lack of guideline-directed outpatient heart failure therapy was a key. Also older age and the presence of coronary heart disease – those were really the big three items that were driving the spike in costs,” said Dr. Swindle of the Chicago office of Optum, a health care consulting group.
He was quick to add that, since the study was based upon administrative data, the lack of adherence to guideline-directed therapy may be unrelated to physician prescribing.
“We see the prescriptions that patients are actually filling. Patients may very well be seeing their cardiologist and being prescribed a medication, but they simply don’t fill the prescription,” he explained in an interview.
Over patients’ final 2 years of life, semiannual all-cause health care costs climbed from a baseline of roughly $10,000 during months 24-19 before death by about 4-fold during months 6-1 before death. Heart failure–related medical costs jumped 10-fold in Asians, 7.8-fold in Hispanics, 6.6-fold in African Americans, and 6.7-fold in whites. Most of the increases occurred in the final 6 months.
Zeroing in on the final 6 months of life, the mean cumulative total medical costs were $44,599, with heart failure–related medical costs accounting for $24,818 of that figure. Total medical costs averaged just under $5,000 during month 6 prior to death and rose roughly 3.5-fold over the remaining months.
This study was supported by Novartis Pharmaceuticals.
CHICAGO – Health care costs for heart failure patients spike dramatically in the last 6 months of life, with lack of adherence to guideline-directed outpatient medical therapy being the major modifiable factor driving the costs, Jason P. Swindle, Ph.D., reported at the annual meeting of the American College of Cardiology.
He presented a retrospective study of heart failure–related and total health care costs during the final 24 months of life for 48,026 Medicare Advantage managed care plan members with heart failure.
The researchers were interested in exploring possible racial/ethnic differences in costs, particularly in light of evidence that African Americans have a higher risk of heart failure and higher all-cause mortality. And while a first look at the data indicated racial differences in the size of end-of-life cost spikes, those differences lost their significance in multivariate analysis.
“Lack of guideline-directed outpatient heart failure therapy was a key. Also older age and the presence of coronary heart disease – those were really the big three items that were driving the spike in costs,” said Dr. Swindle of the Chicago office of Optum, a health care consulting group.
He was quick to add that, since the study was based upon administrative data, the lack of adherence to guideline-directed therapy may be unrelated to physician prescribing.
“We see the prescriptions that patients are actually filling. Patients may very well be seeing their cardiologist and being prescribed a medication, but they simply don’t fill the prescription,” he explained in an interview.
Over patients’ final 2 years of life, semiannual all-cause health care costs climbed from a baseline of roughly $10,000 during months 24-19 before death by about 4-fold during months 6-1 before death. Heart failure–related medical costs jumped 10-fold in Asians, 7.8-fold in Hispanics, 6.6-fold in African Americans, and 6.7-fold in whites. Most of the increases occurred in the final 6 months.
Zeroing in on the final 6 months of life, the mean cumulative total medical costs were $44,599, with heart failure–related medical costs accounting for $24,818 of that figure. Total medical costs averaged just under $5,000 during month 6 prior to death and rose roughly 3.5-fold over the remaining months.
This study was supported by Novartis Pharmaceuticals.
AT ACC 16
Key clinical point: Addressing lack of adherence to guideline-directed medical therapy could curb end-of-life health care costs in heart failure.
Major finding: Total monthly medical costs in heart failure patients during their final 6 months of life climbed roughly 3.5-fold.
Data source: This was a retrospective study of total and heart failure–related health care costs during the final 24 months of life for more than 48,000 patients with heart failure.
Disclosures: This study was supported by Novartis Pharmaceuticals. Dr. Swindle is an employee of Optum, which conducted the research.
Acute heart failure mortality climbs with severity of peripheral edema
CHICAGO – Breathlessness typically results in hospital admission for patients with heart failure, but peripheral edema is what prolongs their stay, according to Dr. John G.F. Cleland.
Moreover, it’s not only hospital length of stay that climbs with increasing severity of peripheral edema on admission. So does mortality, both during the index admission and long term, Dr. Cleland reported at the annual meeting of the American College of Cardiology.
He presented a retrospective analysis of 121,229 patients admitted with a primary diagnosis of heart failure to more than 90% of the hospitals in England and Wales during 2007-2013.
“It turns out that the majority of patients we’re seeing admitted with heart failure at U.K. hospitals are not admitted because of severe breathlessness at rest; they’re admitted for increasing fluid retention. They have lots of peripheral edema, which is actually associated with bad outcome. The patients who are very breathless tend to have the better outcome because they have left heart failure. The ones who are full of peripheral edema have more renal dysfunction and anemia, and they’re more likely to have right heart failure,” said Dr. Cleland, professor of cardiology at Imperial College London.
“I’m not saying the breathless group isn’t a target, but this group with peripheral edema is a bigger target – and we’re not designing trials to address their problems,” he added.
Compared with patients with no peripheral edema on hospital admission, the risk of mortality during that hospitalization and up to 3 years of subsequent follow-up was increased 1.2-fold in those with mild peripheral edema, 1.48-fold with moderate peripheral edema, and 1.89-fold in those with severe peripheral edema.
“We’re designing all the big clinical trials in acute heart failure to capture what I regard as neither fish nor fowl. They’re recruiting patients 6-12 hours after admission for acute heart failure. But by that point they’ve pretty well responded to their intravenous diuretics, and we’re just catching the tail end of their breathlessness. They’re not an emergency. The problem is really their peripheral edema, and that’s a day 2/day 3 problem. The first 6 hours of care really isn’t relevant to this group,” according to the internationally renowned heart failure researcher.
Indeed, he said he’d like to see the term “acute heart failure” laid to rest.
“If you think of acute decompensated heart failure, you think of patients wearing an oxygen mask coming in by an ambulance with blue lights flashing, and it’s an emergency. We need to move the mind-set. We shouldn’t call it acute heart failure at all, we should start talking about hospitalized heart failure, of which some is acute but much of it is subacute in people who have been deteriorating over several weeks and have just gotten to the point where they’re not coping at home anymore. They call a taxi or a friend who takes them to the hospital. Then they take a wheelchair from the taxi to the ER, but they don’t really need an ER at all,” Dr. Cleland said.
Many of these patients could be redirected to a different sort of facility at great cost savings, he added.
“In the United Kingdom and I think in the States, we’re now talking about furosemide lounges where, rather than admit the patient, you can bring them up as a day case, give them intravenous therapy, then [have them] go home at night, perhaps coming back for 3 or 4 days if needed. People are also now looking at home infusion services, and there’s a nice device for giving subcutaneous doses of furosemide as well,” the cardiologist said.
Apart from diuretics, there really aren’t any effective medications at present for peripheral edema in heart failure. But there are novel investigational agents worthy of evaluation, according to Dr. Cleland, including drugs aimed at improving mitochondrial function, agents that inhibit channels that allow edema to gather, and iron therapy.
The study was supported by the British Society for Heart Failure and the National Institute for Cardiovascular Outcomes Research. Dr. Cleland reported having no relevant financial conflicts.
CHICAGO – Breathlessness typically results in hospital admission for patients with heart failure, but peripheral edema is what prolongs their stay, according to Dr. John G.F. Cleland.
Moreover, it’s not only hospital length of stay that climbs with increasing severity of peripheral edema on admission. So does mortality, both during the index admission and long term, Dr. Cleland reported at the annual meeting of the American College of Cardiology.
He presented a retrospective analysis of 121,229 patients admitted with a primary diagnosis of heart failure to more than 90% of the hospitals in England and Wales during 2007-2013.
“It turns out that the majority of patients we’re seeing admitted with heart failure at U.K. hospitals are not admitted because of severe breathlessness at rest; they’re admitted for increasing fluid retention. They have lots of peripheral edema, which is actually associated with bad outcome. The patients who are very breathless tend to have the better outcome because they have left heart failure. The ones who are full of peripheral edema have more renal dysfunction and anemia, and they’re more likely to have right heart failure,” said Dr. Cleland, professor of cardiology at Imperial College London.
“I’m not saying the breathless group isn’t a target, but this group with peripheral edema is a bigger target – and we’re not designing trials to address their problems,” he added.
Compared with patients with no peripheral edema on hospital admission, the risk of mortality during that hospitalization and up to 3 years of subsequent follow-up was increased 1.2-fold in those with mild peripheral edema, 1.48-fold with moderate peripheral edema, and 1.89-fold in those with severe peripheral edema.

“We’re designing all the big clinical trials in acute heart failure to capture what I regard as neither fish nor fowl. They’re recruiting patients 6-12 hours after admission for acute heart failure. But by that point they’ve pretty well responded to their intravenous diuretics, and we’re just catching the tail end of their breathlessness. They’re not an emergency. The problem is really their peripheral edema, and that’s a day 2/day 3 problem. The first 6 hours of care really isn’t relevant to this group,” according to the internationally renowned heart failure researcher.
Indeed, he said he’d like to see the term “acute heart failure” laid to rest.
“If you think of acute decompensated heart failure, you think of patients wearing an oxygen mask coming in by an ambulance with blue lights flashing, and it’s an emergency. We need to move the mind-set. We shouldn’t call it acute heart failure at all, we should start talking about hospitalized heart failure, of which some is acute but much of it is subacute in people who have been deteriorating over several weeks and have just gotten to the point where they’re not coping at home anymore. They call a taxi or a friend who takes them to the hospital. Then they take a wheelchair from the taxi to the ER, but they don’t really need an ER at all,” Dr. Cleland said.
Many of these patients could be redirected to a different sort of facility at great cost savings, he added.
“In the United Kingdom and I think in the States, we’re now talking about furosemide lounges where, rather than admit the patient, you can bring them up as a day case, give them intravenous therapy, then [have them] go home at night, perhaps coming back for 3 or 4 days if needed. People are also now looking at home infusion services, and there’s a nice device for giving subcutaneous doses of furosemide as well,” the cardiologist said.
Apart from diuretics, there really aren’t any effective medications at present for peripheral edema in heart failure. But there are novel investigational agents worthy of evaluation, according to Dr. Cleland, including drugs aimed at improving mitochondrial function, agents that inhibit channels that allow edema to gather, and iron therapy.
The study was supported by the British Society for Heart Failure and the National Institute for Cardiovascular Outcomes Research. Dr. Cleland reported having no relevant financial conflicts.
CHICAGO – Breathlessness typically results in hospital admission for patients with heart failure, but peripheral edema is what prolongs their stay, according to Dr. John G.F. Cleland.
Moreover, it’s not only hospital length of stay that climbs with increasing severity of peripheral edema on admission. So does mortality, both during the index admission and long term, Dr. Cleland reported at the annual meeting of the American College of Cardiology.
He presented a retrospective analysis of 121,229 patients admitted with a primary diagnosis of heart failure to more than 90% of the hospitals in England and Wales during 2007-2013.
“It turns out that the majority of patients we’re seeing admitted with heart failure at U.K. hospitals are not admitted because of severe breathlessness at rest; they’re admitted for increasing fluid retention. They have lots of peripheral edema, which is actually associated with bad outcome. The patients who are very breathless tend to have the better outcome because they have left heart failure. The ones who are full of peripheral edema have more renal dysfunction and anemia, and they’re more likely to have right heart failure,” said Dr. Cleland, professor of cardiology at Imperial College London.
“I’m not saying the breathless group isn’t a target, but this group with peripheral edema is a bigger target – and we’re not designing trials to address their problems,” he added.
Compared with patients with no peripheral edema on hospital admission, the risk of mortality during that hospitalization and up to 3 years of subsequent follow-up was increased 1.2-fold in those with mild peripheral edema, 1.48-fold with moderate peripheral edema, and 1.89-fold in those with severe peripheral edema.

“We’re designing all the big clinical trials in acute heart failure to capture what I regard as neither fish nor fowl. They’re recruiting patients 6-12 hours after admission for acute heart failure. But by that point they’ve pretty well responded to their intravenous diuretics, and we’re just catching the tail end of their breathlessness. They’re not an emergency. The problem is really their peripheral edema, and that’s a day 2/day 3 problem. The first 6 hours of care really isn’t relevant to this group,” according to the internationally renowned heart failure researcher.
Indeed, he said he’d like to see the term “acute heart failure” laid to rest.
“If you think of acute decompensated heart failure, you think of patients wearing an oxygen mask coming in by an ambulance with blue lights flashing, and it’s an emergency. We need to move the mind-set. We shouldn’t call it acute heart failure at all, we should start talking about hospitalized heart failure, of which some is acute but much of it is subacute in people who have been deteriorating over several weeks and have just gotten to the point where they’re not coping at home anymore. They call a taxi or a friend who takes them to the hospital. Then they take a wheelchair from the taxi to the ER, but they don’t really need an ER at all,” Dr. Cleland said.
Many of these patients could be redirected to a different sort of facility at great cost savings, he added.
“In the United Kingdom and I think in the States, we’re now talking about furosemide lounges where, rather than admit the patient, you can bring them up as a day case, give them intravenous therapy, then [have them] go home at night, perhaps coming back for 3 or 4 days if needed. People are also now looking at home infusion services, and there’s a nice device for giving subcutaneous doses of furosemide as well,” the cardiologist said.
Apart from diuretics, there really aren’t any effective medications at present for peripheral edema in heart failure. But there are novel investigational agents worthy of evaluation, according to Dr. Cleland, including drugs aimed at improving mitochondrial function, agents that inhibit channels that allow edema to gather, and iron therapy.
The study was supported by the British Society for Heart Failure and the National Institute for Cardiovascular Outcomes Research. Dr. Cleland reported having no relevant financial conflicts.
AT ACC 16
Key clinical point: Leg swelling warrants greater attention in patients hospitalized for acute heart failure.
Major finding: In-hospital mortality was more than twice as great in patients admitted for acute heart failure with severe peripheral edema, compared with no leg swelling.
Data source: A retrospective study of more than 121,000 patients hospitalized for acute heart failure in England and Wales.
Disclosures: The study was supported by the British Society for Heart Failure and the National Institute for Cardiovascular Outcomes Research. The presenter reported having no relevant financial conflicts.
Valve hemodynamic deterioration 2.5% at 1 year
CHICAGO – The incidence of valve hemodynamic deterioration in the first year after transcatheter aortic valve replacement is about 2.5%, but this event wasn’t clearly associated with adverse clinical outcomes out to 18 months of follow-up in an analysis of the large U.S. registry collaboratively maintained by the Society of Thoracic Surgeons and the American College of Cardiology.
“These findings, especially the patient and procedural predictors of valve hemodynamic deterioration we identified, may help to inform TAVR care, including patient selection, surveillance, and preventive strategies,” Dr. Sreekanth Vemulapalli reported at the annual meeting of the American College of Cardiology.
Recent reports have linked TAVR to subsequent development of leaflet abnormalities and valve thrombosis, with widely ranging estimates of incidence. Definitive answers as to the true rate of these adverse events and the underlying mechanisms will come from ongoing prospective studies using advanced imaging via four-dimensional CT or transesophageal echocardiography, but those studies will take years to complete, noted Dr. Vemulapalli of the Duke Clinical Research Institute in Durham, N.C.
In the meantime, he continued, the STS/ACC Transcatheter Valve Therapy Registry provides a unique opportunity to shed light on the incidence and consequences of valve hemodynamic deterioration (VHD) in real-world clinical practice. The registry includes all commercial TAVR procedures performed in the United States, with transthoracic echocardiograms obtained pre- and post-TAVR, at 30 days, and at 1 year after the procedure.
To examine the short- and longer-term rates of VHD, which Dr. Vemulapalli and his coinvestigators defined as an increase in the mean aortic valve gradient of 10 mm or more, the researchers assembled two separate patient cohorts. They comprised a short-term–risk group of 10,095 patients who underwent TAVR at 334 sites, with an incidence of VHD of 2.1% during the first 30 days after the procedure, and 3,175 patients at 254 sites, whose incidence of VHD from day 30 through 1 year post TAVR was 2.5%.
The combined rate of VHD and all-cause mortality during the first 30 days was 7.1%. For the long-term cohort, the combined endpoint rate from day 30 to 1 year was 23.5%.
Importantly, the occurrence of VHD was not associated with an excess of the composite endpoint of mortality, stroke, heart failure hospitalization, or aortic valve reintervention at 1 year in either the short- or long-term cohort. The same held true in an analysis covering the period of 12-18 months post TAVR, according to Dr. Vemulapalli.
In a multivariate analysis, the significant predictors of VHD in the short-term cohort were male sex; increased body mass index, with the risk rising stepwise with every additional 5 kg/m above normal weight; baseline severe chronic lung disease; a valve-in-valve procedure; a larger baseline aortic valve gradient; a TAVR valve size of 23 mm or less; and severe patient/prosthesis mismatch.
In the long-term cohort, the risk factors for VHD were hospital discharge on a factor Xa inhibitor and a larger predischarge aortic valve gradient.
Change in left ventricular ejection fraction over the course of the study bore no relation to VHD risk. Neither did which of the two commercially available TAVR valves a patient received.
This study was funded by the American College of Cardiology’s National Cardiovascular Data Registry. Dr. Vemulapalli reported serving as a consultant to Novella and Premiere and receiving research grants from the Agency for Healthcare Research and Quality, Boston Scientific, Abbott Vascular, and the ACC.
CHICAGO – The incidence of valve hemodynamic deterioration in the first year after transcatheter aortic valve replacement is about 2.5%, but this event wasn’t clearly associated with adverse clinical outcomes out to 18 months of follow-up in an analysis of the large U.S. registry collaboratively maintained by the Society of Thoracic Surgeons and the American College of Cardiology.
“These findings, especially the patient and procedural predictors of valve hemodynamic deterioration we identified, may help to inform TAVR care, including patient selection, surveillance, and preventive strategies,” Dr. Sreekanth Vemulapalli reported at the annual meeting of the American College of Cardiology.
Recent reports have linked TAVR to subsequent development of leaflet abnormalities and valve thrombosis, with widely ranging estimates of incidence. Definitive answers as to the true rate of these adverse events and the underlying mechanisms will come from ongoing prospective studies using advanced imaging via four-dimensional CT or transesophageal echocardiography, but those studies will take years to complete, noted Dr. Vemulapalli of the Duke Clinical Research Institute in Durham, N.C.
In the meantime, he continued, the STS/ACC Transcatheter Valve Therapy Registry provides a unique opportunity to shed light on the incidence and consequences of valve hemodynamic deterioration (VHD) in real-world clinical practice. The registry includes all commercial TAVR procedures performed in the United States, with transthoracic echocardiograms obtained pre- and post-TAVR, at 30 days, and at 1 year after the procedure.
To examine the short- and longer-term rates of VHD, which Dr. Vemulapalli and his coinvestigators defined as an increase in the mean aortic valve gradient of 10 mm or more, the researchers assembled two separate patient cohorts. They comprised a short-term–risk group of 10,095 patients who underwent TAVR at 334 sites, with an incidence of VHD of 2.1% during the first 30 days after the procedure, and 3,175 patients at 254 sites, whose incidence of VHD from day 30 through 1 year post TAVR was 2.5%.
The combined rate of VHD and all-cause mortality during the first 30 days was 7.1%. For the long-term cohort, the combined endpoint rate from day 30 to 1 year was 23.5%.
Importantly, the occurrence of VHD was not associated with an excess of the composite endpoint of mortality, stroke, heart failure hospitalization, or aortic valve reintervention at 1 year in either the short- or long-term cohort. The same held true in an analysis covering the period of 12-18 months post TAVR, according to Dr. Vemulapalli.
In a multivariate analysis, the significant predictors of VHD in the short-term cohort were male sex; increased body mass index, with the risk rising stepwise with every additional 5 kg/m above normal weight; baseline severe chronic lung disease; a valve-in-valve procedure; a larger baseline aortic valve gradient; a TAVR valve size of 23 mm or less; and severe patient/prosthesis mismatch.
In the long-term cohort, the risk factors for VHD were hospital discharge on a factor Xa inhibitor and a larger predischarge aortic valve gradient.
Change in left ventricular ejection fraction over the course of the study bore no relation to VHD risk. Neither did which of the two commercially available TAVR valves a patient received.
This study was funded by the American College of Cardiology’s National Cardiovascular Data Registry. Dr. Vemulapalli reported serving as a consultant to Novella and Premiere and receiving research grants from the Agency for Healthcare Research and Quality, Boston Scientific, Abbott Vascular, and the ACC.
CHICAGO – The incidence of valve hemodynamic deterioration in the first year after transcatheter aortic valve replacement is about 2.5%, but this event wasn’t clearly associated with adverse clinical outcomes out to 18 months of follow-up in an analysis of the large U.S. registry collaboratively maintained by the Society of Thoracic Surgeons and the American College of Cardiology.
“These findings, especially the patient and procedural predictors of valve hemodynamic deterioration we identified, may help to inform TAVR care, including patient selection, surveillance, and preventive strategies,” Dr. Sreekanth Vemulapalli reported at the annual meeting of the American College of Cardiology.
Recent reports have linked TAVR to subsequent development of leaflet abnormalities and valve thrombosis, with widely ranging estimates of incidence. Definitive answers as to the true rate of these adverse events and the underlying mechanisms will come from ongoing prospective studies using advanced imaging via four-dimensional CT or transesophageal echocardiography, but those studies will take years to complete, noted Dr. Vemulapalli of the Duke Clinical Research Institute in Durham, N.C.
In the meantime, he continued, the STS/ACC Transcatheter Valve Therapy Registry provides a unique opportunity to shed light on the incidence and consequences of valve hemodynamic deterioration (VHD) in real-world clinical practice. The registry includes all commercial TAVR procedures performed in the United States, with transthoracic echocardiograms obtained pre- and post-TAVR, at 30 days, and at 1 year after the procedure.
To examine the short- and longer-term rates of VHD, which Dr. Vemulapalli and his coinvestigators defined as an increase in the mean aortic valve gradient of 10 mm or more, the researchers assembled two separate patient cohorts. They comprised a short-term–risk group of 10,095 patients who underwent TAVR at 334 sites, with an incidence of VHD of 2.1% during the first 30 days after the procedure, and 3,175 patients at 254 sites, whose incidence of VHD from day 30 through 1 year post TAVR was 2.5%.
The combined rate of VHD and all-cause mortality during the first 30 days was 7.1%. For the long-term cohort, the combined endpoint rate from day 30 to 1 year was 23.5%.
Importantly, the occurrence of VHD was not associated with an excess of the composite endpoint of mortality, stroke, heart failure hospitalization, or aortic valve reintervention at 1 year in either the short- or long-term cohort. The same held true in an analysis covering the period of 12-18 months post TAVR, according to Dr. Vemulapalli.
In a multivariate analysis, the significant predictors of VHD in the short-term cohort were male sex; increased body mass index, with the risk rising stepwise with every additional 5 kg/m above normal weight; baseline severe chronic lung disease; a valve-in-valve procedure; a larger baseline aortic valve gradient; a TAVR valve size of 23 mm or less; and severe patient/prosthesis mismatch.
In the long-term cohort, the risk factors for VHD were hospital discharge on a factor Xa inhibitor and a larger predischarge aortic valve gradient.
Change in left ventricular ejection fraction over the course of the study bore no relation to VHD risk. Neither did which of the two commercially available TAVR valves a patient received.
This study was funded by the American College of Cardiology’s National Cardiovascular Data Registry. Dr. Vemulapalli reported serving as a consultant to Novella and Premiere and receiving research grants from the Agency for Healthcare Research and Quality, Boston Scientific, Abbott Vascular, and the ACC.
AT ACC 16
Key clinical point: The incidence of valve hemodynamic deterioration after transaortic valve replacement is 2.5% from day 30 through 12 months post procedure.
Major finding: Patients who experienced valve hemodynamic deterioration had a rate of adverse clinical outcomes similar to those without valve deterioration.
Data source: This was a retrospective study of 18-month outcomes in the Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy Registry, which covers all commercial transcatheter valve replacements done in the United States.
Disclosures: This study was funded by the American College of Cardiology’s National Cardiovascular Data Registry. The presenter reported serving as a consultant to Novella and Premiere and receiving research grants from the Agency for Healthcare Research and Quality, Boston Scientific, Abbott Vascular, and the ACC.
No rise in serious HF seen in patients taking saxagliptin or sitagliptin
Neither saxagliptin nor sitagliptin, the two oral DPP-4 inhibitors most commonly used as antihyperglycemic medications, raised the risk of hospitalization for heart failure in a large population-based cohort study that analyzed data from a Food and Drug Administration surveillance program.
The report was published online April 25 in Annals of Internal Medicine.
The cardiovascular safety of DPP-4 inhibitors is controversial: Several postmarketing studies have produced conflicting results, particularly with regard to HF risk. “Patients with diabetes have a higher HF risk than those without, so any antihyperglycemic agent that modifies the risk warrants further examination,” said Sengwee Toh, Sc.D., a pharmacoepidemiologist in the department of population medicine, Harvard Medical School and Harvard Pilgrim Health Institute, Boston, and his associates.
They compared rates of HF among demographically and geographically diverse patients who initiated antidiabetic medications during a 7-year period in routine clinical settings. The study population included 78,553 adults who initiated saxagliptin and 298,124 who initiated sitagliptin, who were compared with patients who initiated pioglitazone, second-generation sulfonylureas, or long-acting insulins. Mean follow-up was 7-9 months.
There was no evidence of an increased risk of hospitalization for HF among new users of saxagliptin or sitagliptin. The hazard ratios for developing HF were 0.83 for saxagliptin vs. sitagliptin, 0.63 for saxagliptin vs. pioglitazone, 0.69 for saxagliptin vs. sulfonylureas, and 0.61 for saxagliptin vs. insulin. Similarly, the hazard ratios for developing HF were 0.74 for sitagliptin vs. pioglitazone, 0.86 for sitagliptin vs. sulfonylureas, and 0.71 for sitagliptin vs. insulin.
These results were consistent across sensitivity analyses and subgroup analyses that categorized patients by whether or not they had preexisting cardiovascular disease and whether or not they had a history of prior HF, the investigators said (Ann Intern Med. 2016 April 25. doi:10.7326/M15-2568).
However, this was an observational study with a relatively short follow-up. “Well-designed randomized trials with hospitalization for HF as the main endpoint or observational studies that address the limitations of our study will help provide more definitive evidence on the topic,” Dr. Toh and his associates said.
This study was supported by the FDA. Dr. Toh reported having no relevant financial disclosures; one of his associates reported receiving personal fees from Novartis unrelated to this work.
The findings of Toh et al. allay concerns about a saxagliptin- or sitagliptin-associated risk for heart failure. This risk was similar between the two agents and either comparable to or lower than that in all other comparator groups.
Beyond reassuring clinicians, this study illustrates the value of large, longitudinal databases built from clinical and administrative data, to complement the findings of clinical trials. These investigators were able to draw their conclusions from rich demographic, diagnostic, prescription, and utilization data based in routine real-world practice.
Joseph V. Selby, M.D., is at the Patient-Centered Outcomes Research Institute, Washington. He reported having no relevant financial disclosures. Dr. Selby made these remarks in an editorial accompanying Dr. Toh’s report (Ann. Intern. Med. 2016 April 25. doi:10.7326/M16-0869).
The findings of Toh et al. allay concerns about a saxagliptin- or sitagliptin-associated risk for heart failure. This risk was similar between the two agents and either comparable to or lower than that in all other comparator groups.
Beyond reassuring clinicians, this study illustrates the value of large, longitudinal databases built from clinical and administrative data, to complement the findings of clinical trials. These investigators were able to draw their conclusions from rich demographic, diagnostic, prescription, and utilization data based in routine real-world practice.
Joseph V. Selby, M.D., is at the Patient-Centered Outcomes Research Institute, Washington. He reported having no relevant financial disclosures. Dr. Selby made these remarks in an editorial accompanying Dr. Toh’s report (Ann. Intern. Med. 2016 April 25. doi:10.7326/M16-0869).
The findings of Toh et al. allay concerns about a saxagliptin- or sitagliptin-associated risk for heart failure. This risk was similar between the two agents and either comparable to or lower than that in all other comparator groups.
Beyond reassuring clinicians, this study illustrates the value of large, longitudinal databases built from clinical and administrative data, to complement the findings of clinical trials. These investigators were able to draw their conclusions from rich demographic, diagnostic, prescription, and utilization data based in routine real-world practice.
Joseph V. Selby, M.D., is at the Patient-Centered Outcomes Research Institute, Washington. He reported having no relevant financial disclosures. Dr. Selby made these remarks in an editorial accompanying Dr. Toh’s report (Ann. Intern. Med. 2016 April 25. doi:10.7326/M16-0869).
Neither saxagliptin nor sitagliptin, the two oral DPP-4 inhibitors most commonly used as antihyperglycemic medications, raised the risk of hospitalization for heart failure in a large population-based cohort study that analyzed data from a Food and Drug Administration surveillance program.
The report was published online April 25 in Annals of Internal Medicine.
The cardiovascular safety of DPP-4 inhibitors is controversial: Several postmarketing studies have produced conflicting results, particularly with regard to HF risk. “Patients with diabetes have a higher HF risk than those without, so any antihyperglycemic agent that modifies the risk warrants further examination,” said Sengwee Toh, Sc.D., a pharmacoepidemiologist in the department of population medicine, Harvard Medical School and Harvard Pilgrim Health Institute, Boston, and his associates.
They compared rates of HF among demographically and geographically diverse patients who initiated antidiabetic medications during a 7-year period in routine clinical settings. The study population included 78,553 adults who initiated saxagliptin and 298,124 who initiated sitagliptin, who were compared with patients who initiated pioglitazone, second-generation sulfonylureas, or long-acting insulins. Mean follow-up was 7-9 months.
There was no evidence of an increased risk of hospitalization for HF among new users of saxagliptin or sitagliptin. The hazard ratios for developing HF were 0.83 for saxagliptin vs. sitagliptin, 0.63 for saxagliptin vs. pioglitazone, 0.69 for saxagliptin vs. sulfonylureas, and 0.61 for saxagliptin vs. insulin. Similarly, the hazard ratios for developing HF were 0.74 for sitagliptin vs. pioglitazone, 0.86 for sitagliptin vs. sulfonylureas, and 0.71 for sitagliptin vs. insulin.
These results were consistent across sensitivity analyses and subgroup analyses that categorized patients by whether or not they had preexisting cardiovascular disease and whether or not they had a history of prior HF, the investigators said (Ann Intern Med. 2016 April 25. doi:10.7326/M15-2568).
However, this was an observational study with a relatively short follow-up. “Well-designed randomized trials with hospitalization for HF as the main endpoint or observational studies that address the limitations of our study will help provide more definitive evidence on the topic,” Dr. Toh and his associates said.
This study was supported by the FDA. Dr. Toh reported having no relevant financial disclosures; one of his associates reported receiving personal fees from Novartis unrelated to this work.
Neither saxagliptin nor sitagliptin, the two oral DPP-4 inhibitors most commonly used as antihyperglycemic medications, raised the risk of hospitalization for heart failure in a large population-based cohort study that analyzed data from a Food and Drug Administration surveillance program.
The report was published online April 25 in Annals of Internal Medicine.
The cardiovascular safety of DPP-4 inhibitors is controversial: Several postmarketing studies have produced conflicting results, particularly with regard to HF risk. “Patients with diabetes have a higher HF risk than those without, so any antihyperglycemic agent that modifies the risk warrants further examination,” said Sengwee Toh, Sc.D., a pharmacoepidemiologist in the department of population medicine, Harvard Medical School and Harvard Pilgrim Health Institute, Boston, and his associates.
They compared rates of HF among demographically and geographically diverse patients who initiated antidiabetic medications during a 7-year period in routine clinical settings. The study population included 78,553 adults who initiated saxagliptin and 298,124 who initiated sitagliptin, who were compared with patients who initiated pioglitazone, second-generation sulfonylureas, or long-acting insulins. Mean follow-up was 7-9 months.
There was no evidence of an increased risk of hospitalization for HF among new users of saxagliptin or sitagliptin. The hazard ratios for developing HF were 0.83 for saxagliptin vs. sitagliptin, 0.63 for saxagliptin vs. pioglitazone, 0.69 for saxagliptin vs. sulfonylureas, and 0.61 for saxagliptin vs. insulin. Similarly, the hazard ratios for developing HF were 0.74 for sitagliptin vs. pioglitazone, 0.86 for sitagliptin vs. sulfonylureas, and 0.71 for sitagliptin vs. insulin.
These results were consistent across sensitivity analyses and subgroup analyses that categorized patients by whether or not they had preexisting cardiovascular disease and whether or not they had a history of prior HF, the investigators said (Ann Intern Med. 2016 April 25. doi:10.7326/M15-2568).
However, this was an observational study with a relatively short follow-up. “Well-designed randomized trials with hospitalization for HF as the main endpoint or observational studies that address the limitations of our study will help provide more definitive evidence on the topic,” Dr. Toh and his associates said.
This study was supported by the FDA. Dr. Toh reported having no relevant financial disclosures; one of his associates reported receiving personal fees from Novartis unrelated to this work.
FROM ANNALS OF INTERNAL MEDICINE
Key clinical point: No increase in the risk of hospitalization for heart failure was found in a large cohort study that used an FDA surveillance program.
Major finding: The hazard ratios for developing HF were 0.83 for saxagliptin vs. sitagliptin, 0.63 for saxagliptin vs. pioglitazone, 0.69 for saxagliptin vs. sulfonylureas, and 0.61 for saxagliptin vs. insulin.
Data source: A population-based retrospective cohort study involving 78,553 new users of saxagliptin and 298,124 of sitagliptin during a 7-year period.
Disclosures: This study was supported by the FDA. Dr. Toh reported having no relevant financial disclosures; one of his associates reported receiving personal fees from Novartis unrelated to this work.