User login
Novel agent fails to reduce infarct size, but may cut heart failure, protect kidneys
SAN DIEGO– Bendavia, a novel agent designed to prevent reperfusion injury by improving myocardial energetics, failed to reduce infarct size in ST-elevation MI patients in a phase II randomized trial.
Despite this disappointing result on the primary study endpoint, there was a silver lining. The study, known as the EMBRACE STEMI trial, generated two provocative findings in exploratory, hypothesis-generating secondary analyses: Bendavia appeared to reduce new-onset symptomatic heart failure early on after percutaneous coronary intervention, and the drug also had an apparent renal protective effect, Dr. C. Michael Gibson reported at the annual meeting of the American College of Cardiology.
These two potentially important findings are being studied more fully in three ongoing prospective, randomized, double-blind, placebo-controlled trials, added Dr. Gibson, professor of medicine at Harvard Medical School, Boston, and chairman of EMBRACE STEMI (Evaluation of Myocardial Effects of Bendavia for Reducing Reperfusion Injury in Patients With Acute Coronary Events).
As for the negative result in terms of the primary study hypothesis that Bendavia would reduce infarct size by preventing reperfusion injury in patients undergoing PCI for large STEMIs, as it had done successfully in animal models, the cardiologist said all indications are that the investigators targeted the right patients at the right time with the right dose of the medication.
“I think we conducted the experiment in such a way that if there was a ‘there’ there, we were going to see it,” Dr. Gibson said.
EMBRACE STEMI included 118 patients with large, first-time, anterior MIs featuring proximal or mid-left anterior descending coronary artery lesions. To be eligible for the study, patients had to arrive at the catheterization lab with a closed artery within 4 hours after symptom onset. They were then randomized in double-blind fashion to intravenous Bendavia at 0.05 mg/kg per hour for 1 hour beginning 15 minutes before PCI or to an equal volume of placebo.
The study, conducted by the PERFUSE study group at 24 sites in the United States and three European countries, was planned to be considerably larger, but to the surprise of investigators, 40% of patients arrived at the cath lab with an open artery.
“This is higher than the historical figure of 20%,” Dr. Gibson noted. “I think this is one of the notable findings in this study: Whether due to earlier presentation or better pharmacotherapy, it’s tougher to locate people with closed arteries for these kinds of studies in 2015.”
Bendavia did not significantly reduce infarct size as measured by the areas under the curve for creatine kinase MB (CK-MB) or troponin I over the initial 72 hours after PCI. Moreover, cardiac MRIs obtained at 4 and 30 days post PCI showed no significant difference between the Bendavia and placebo groups in infarct volume, left ventricular mass, edema volume, or left ventricular ejection fraction. Rates of ST-segment resolution immediately and 24 hours post PCI did not differ between the two groups, either. Nor did TIMI flow grade, frame count, or myocardial perfusion grade. Also, 30-day and 6-month rates of the composite clinical endpoint of death, new-onset heart failure beginning more than 24 hours post PCI, and heart failure rehospitalization did not differ in the Bendavia and control groups.
However, the incidence of new-onset heart failure within the first 8 hours post PCI was 8.6% in the Bendavia group compared with 18.3% in controls. This finding has prompted the two ongoing randomized trials where Bendavia is being tested in higher doses in patients with stable heart failure with reduced ejection fraction.
Bendavia is a peptide targeting the mitochondria, where it binds with cardiolipin and preserves the integrity of the electron transport system. In animal models of heart failure, it improves left ventricular function.
“It’s not an inotrope. It doesn’t change heart rate or blood pressure. Rather than increasing demands on the heart, it offers a supply side solution in improving myocardial energetics,” Dr. Gibson explained.
Bendavia was associated with renal preservation as reflected in a significantly smaller increase in creatinine level in the first 12 hours post PCI: 1.0 mcmol/L compared with 3.7 mcmol/L in controls.
This finding piqued the interest of discussant Dr. James B. McClurken, professor and vice chair of surgery at Temple University in Philadelphia.
“The renal protective findings have widespread potential application, including for coronary artery bypass surgery patients. What are your thoughts about the mechanism?” he asked.
Dr. Gibson replied that a small study done at the Mayo Clinic suggests a biologically plausible mechanism: Bendavia dramatically improved kidney hypoxia secondary to ischemia and the dye load entailed in longer PCI procedures.
Dr. Gibson reported receiving a research grant from Stealth BioTherapeutics to conduct the EMBRACE STEMI trial. He serves as a consultant to numerous pharmaceutical and medical device companies.
SAN DIEGO– Bendavia, a novel agent designed to prevent reperfusion injury by improving myocardial energetics, failed to reduce infarct size in ST-elevation MI patients in a phase II randomized trial.
Despite this disappointing result on the primary study endpoint, there was a silver lining. The study, known as the EMBRACE STEMI trial, generated two provocative findings in exploratory, hypothesis-generating secondary analyses: Bendavia appeared to reduce new-onset symptomatic heart failure early on after percutaneous coronary intervention, and the drug also had an apparent renal protective effect, Dr. C. Michael Gibson reported at the annual meeting of the American College of Cardiology.
These two potentially important findings are being studied more fully in three ongoing prospective, randomized, double-blind, placebo-controlled trials, added Dr. Gibson, professor of medicine at Harvard Medical School, Boston, and chairman of EMBRACE STEMI (Evaluation of Myocardial Effects of Bendavia for Reducing Reperfusion Injury in Patients With Acute Coronary Events).
As for the negative result in terms of the primary study hypothesis that Bendavia would reduce infarct size by preventing reperfusion injury in patients undergoing PCI for large STEMIs, as it had done successfully in animal models, the cardiologist said all indications are that the investigators targeted the right patients at the right time with the right dose of the medication.
“I think we conducted the experiment in such a way that if there was a ‘there’ there, we were going to see it,” Dr. Gibson said.
EMBRACE STEMI included 118 patients with large, first-time, anterior MIs featuring proximal or mid-left anterior descending coronary artery lesions. To be eligible for the study, patients had to arrive at the catheterization lab with a closed artery within 4 hours after symptom onset. They were then randomized in double-blind fashion to intravenous Bendavia at 0.05 mg/kg per hour for 1 hour beginning 15 minutes before PCI or to an equal volume of placebo.
The study, conducted by the PERFUSE study group at 24 sites in the United States and three European countries, was planned to be considerably larger, but to the surprise of investigators, 40% of patients arrived at the cath lab with an open artery.
“This is higher than the historical figure of 20%,” Dr. Gibson noted. “I think this is one of the notable findings in this study: Whether due to earlier presentation or better pharmacotherapy, it’s tougher to locate people with closed arteries for these kinds of studies in 2015.”
Bendavia did not significantly reduce infarct size as measured by the areas under the curve for creatine kinase MB (CK-MB) or troponin I over the initial 72 hours after PCI. Moreover, cardiac MRIs obtained at 4 and 30 days post PCI showed no significant difference between the Bendavia and placebo groups in infarct volume, left ventricular mass, edema volume, or left ventricular ejection fraction. Rates of ST-segment resolution immediately and 24 hours post PCI did not differ between the two groups, either. Nor did TIMI flow grade, frame count, or myocardial perfusion grade. Also, 30-day and 6-month rates of the composite clinical endpoint of death, new-onset heart failure beginning more than 24 hours post PCI, and heart failure rehospitalization did not differ in the Bendavia and control groups.
However, the incidence of new-onset heart failure within the first 8 hours post PCI was 8.6% in the Bendavia group compared with 18.3% in controls. This finding has prompted the two ongoing randomized trials where Bendavia is being tested in higher doses in patients with stable heart failure with reduced ejection fraction.
Bendavia is a peptide targeting the mitochondria, where it binds with cardiolipin and preserves the integrity of the electron transport system. In animal models of heart failure, it improves left ventricular function.
“It’s not an inotrope. It doesn’t change heart rate or blood pressure. Rather than increasing demands on the heart, it offers a supply side solution in improving myocardial energetics,” Dr. Gibson explained.
Bendavia was associated with renal preservation as reflected in a significantly smaller increase in creatinine level in the first 12 hours post PCI: 1.0 mcmol/L compared with 3.7 mcmol/L in controls.
This finding piqued the interest of discussant Dr. James B. McClurken, professor and vice chair of surgery at Temple University in Philadelphia.
“The renal protective findings have widespread potential application, including for coronary artery bypass surgery patients. What are your thoughts about the mechanism?” he asked.
Dr. Gibson replied that a small study done at the Mayo Clinic suggests a biologically plausible mechanism: Bendavia dramatically improved kidney hypoxia secondary to ischemia and the dye load entailed in longer PCI procedures.
Dr. Gibson reported receiving a research grant from Stealth BioTherapeutics to conduct the EMBRACE STEMI trial. He serves as a consultant to numerous pharmaceutical and medical device companies.
SAN DIEGO– Bendavia, a novel agent designed to prevent reperfusion injury by improving myocardial energetics, failed to reduce infarct size in ST-elevation MI patients in a phase II randomized trial.
Despite this disappointing result on the primary study endpoint, there was a silver lining. The study, known as the EMBRACE STEMI trial, generated two provocative findings in exploratory, hypothesis-generating secondary analyses: Bendavia appeared to reduce new-onset symptomatic heart failure early on after percutaneous coronary intervention, and the drug also had an apparent renal protective effect, Dr. C. Michael Gibson reported at the annual meeting of the American College of Cardiology.
These two potentially important findings are being studied more fully in three ongoing prospective, randomized, double-blind, placebo-controlled trials, added Dr. Gibson, professor of medicine at Harvard Medical School, Boston, and chairman of EMBRACE STEMI (Evaluation of Myocardial Effects of Bendavia for Reducing Reperfusion Injury in Patients With Acute Coronary Events).
As for the negative result in terms of the primary study hypothesis that Bendavia would reduce infarct size by preventing reperfusion injury in patients undergoing PCI for large STEMIs, as it had done successfully in animal models, the cardiologist said all indications are that the investigators targeted the right patients at the right time with the right dose of the medication.
“I think we conducted the experiment in such a way that if there was a ‘there’ there, we were going to see it,” Dr. Gibson said.
EMBRACE STEMI included 118 patients with large, first-time, anterior MIs featuring proximal or mid-left anterior descending coronary artery lesions. To be eligible for the study, patients had to arrive at the catheterization lab with a closed artery within 4 hours after symptom onset. They were then randomized in double-blind fashion to intravenous Bendavia at 0.05 mg/kg per hour for 1 hour beginning 15 minutes before PCI or to an equal volume of placebo.
The study, conducted by the PERFUSE study group at 24 sites in the United States and three European countries, was planned to be considerably larger, but to the surprise of investigators, 40% of patients arrived at the cath lab with an open artery.
“This is higher than the historical figure of 20%,” Dr. Gibson noted. “I think this is one of the notable findings in this study: Whether due to earlier presentation or better pharmacotherapy, it’s tougher to locate people with closed arteries for these kinds of studies in 2015.”
Bendavia did not significantly reduce infarct size as measured by the areas under the curve for creatine kinase MB (CK-MB) or troponin I over the initial 72 hours after PCI. Moreover, cardiac MRIs obtained at 4 and 30 days post PCI showed no significant difference between the Bendavia and placebo groups in infarct volume, left ventricular mass, edema volume, or left ventricular ejection fraction. Rates of ST-segment resolution immediately and 24 hours post PCI did not differ between the two groups, either. Nor did TIMI flow grade, frame count, or myocardial perfusion grade. Also, 30-day and 6-month rates of the composite clinical endpoint of death, new-onset heart failure beginning more than 24 hours post PCI, and heart failure rehospitalization did not differ in the Bendavia and control groups.
However, the incidence of new-onset heart failure within the first 8 hours post PCI was 8.6% in the Bendavia group compared with 18.3% in controls. This finding has prompted the two ongoing randomized trials where Bendavia is being tested in higher doses in patients with stable heart failure with reduced ejection fraction.
Bendavia is a peptide targeting the mitochondria, where it binds with cardiolipin and preserves the integrity of the electron transport system. In animal models of heart failure, it improves left ventricular function.
“It’s not an inotrope. It doesn’t change heart rate or blood pressure. Rather than increasing demands on the heart, it offers a supply side solution in improving myocardial energetics,” Dr. Gibson explained.
Bendavia was associated with renal preservation as reflected in a significantly smaller increase in creatinine level in the first 12 hours post PCI: 1.0 mcmol/L compared with 3.7 mcmol/L in controls.
This finding piqued the interest of discussant Dr. James B. McClurken, professor and vice chair of surgery at Temple University in Philadelphia.
“The renal protective findings have widespread potential application, including for coronary artery bypass surgery patients. What are your thoughts about the mechanism?” he asked.
Dr. Gibson replied that a small study done at the Mayo Clinic suggests a biologically plausible mechanism: Bendavia dramatically improved kidney hypoxia secondary to ischemia and the dye load entailed in longer PCI procedures.
Dr. Gibson reported receiving a research grant from Stealth BioTherapeutics to conduct the EMBRACE STEMI trial. He serves as a consultant to numerous pharmaceutical and medical device companies.
AT ACC 15
Key clinical point: A novel agent that boosts mitochondrial energy did not prevent irreversible reperfusion injury in MI patients undergoing percutaneous coronary intervention.
Major finding: The geometric mean of the area under the curve for serum CK-MB over the first 72 hours post-PCI was 5,570 ng/mL with Bendavia and similar at 5,785 ng/mL with placebo.
Data source: EMBRACE STEMI, a four-country, 24-site, randomized, double-blind, placebo-controlled phase II study conducted in 118 patients who presented with large ST-elevation MIs.
Disclosures: EMBRACE STEMI was funded by Stealth BioTherapeutics. The presenter received a research grant as study chairman.
Ablation cuts AF recurrence 2.5-fold vs. amiodarone in heart failure
SAN DIEGO – Catheter ablation proved superior to amiodarone for treatment of persistent atrial fibrillation in patients with systolic heart failure in the randomized AATAC-AF trial.
The rate of the primary study endpoint – freedom from recurrent AF through 26 months of prospective follow-up– was 70% in the catheter ablation group, twice the 34% rate with amiodarone, Dr. Luigi Di Biase reported at the annual meeting of the American College of Cardiology. After covariate adjustment, the investigators found that recurrence was 2.5 times more likely in the patients treated with amiodarone.
But he added a major caveat: pulmonary vein antrum isolation (PVI) alone was no better than the antiarrhythmic drug. The high overall treatment success rate seen with catheter ablation in the trial was achieved by operators who performed PVI plus some additional form of ablation of their own choosing, such as elimination of non–pulmonary vein triggers, ablation of complex fractionated electrograms, and/or additional linear ablation lesions, according to Dr. Di Biase, head of electrophysiology at the Albert Einstein College of Medicine, New York.
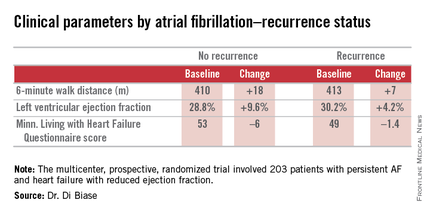
AATAC-AF (Ablation versus Amiodarone for Treatment of Atrial Fibrillation in Patients with Congestive Heart Failure and an Implanted ICD/CRTD) was a multicenter, prospective, randomized trial involving 203 patients with persistent AF and heart failure with reduced ejection fraction. Patients randomized to ablation had to receive PVI at a minimum; operators could perform additional ablation according to their preference. Twenty percent of patients randomized to ablation received PVI alone; 80% underwent additional posterior wall and non–pulmonary vein trigger ablation. The 26-month rate of freedom from recurrence of AF was 36% in patients who received PVI alone and 79% in those who underwent more extensive ablations. A particular strength of the AATAC study was that all participants had an implantable cardioverter-defibrillator and/or cardiac resynchronization therapy device, permitting detection of AF with a much higher degree of accuracy than possible in most AF ablation trials.
Any recurrent AF episodes during the first 3 months of follow-up were excluded from the analysis, regardless of whether patients were in the ablation or amiodarone arms, in accord with the 3-month blanking period that’s standard among electrophysiologists. Patients averaged 1.4 ablation sessions during the first 3 months of the trial.
Regardless of treatment, patients in whom AF did not recur showed significantly greater improvement in left ventricular ejection fraction, exercise capacity, and heart failure–related quality of life.
In addition, all-cause mortality during follow-up was significantly lower in the ablation group: 8%, compared with 18% in patients assigned to amiodarone. Moreover, the rate of hospitalization for arrhythmia or worsening heart failure was 31% in the ablation group versus 57% in patients on amiodarone. The economic implications of this sharp reduction in hospitalizations will be the subject of further study, according to Dr. Di Biase.
Also noteworthy was the finding that seven patients had to discontinue amiodarone due to serious side effects: four because of thyroid toxicity, two for pulmonary toxicity, and one owing to hepatic dysfunction, he continued.
Discussant Dr. Richard I. Fogel, current president of the Heart Rhythm Society, commented that “the 70% arrhythmia-free follow-up was a little surprising to me.”
“That seems a little bit high, particularly in a group with persistent atrial fibrillation,” observed Dr. Fogel, who is chief executive officer at St. Vincent Medical Group, Indianapolis.
Dr. Di Biase attributed the high success rate to two factors: One, only highly experienced operators participated in AATAC, and two, most of them weren’t content to stick to PVI alone.
“If you try to do a more extensive procedure addressing non–pulmonary vein triggers in other areas in the left atrium, the success rate is increased by far,” the electrophysiologist said.
As for a possible mechanism for the mortality benefit seen with ablation, “several studies have shown that in a population with heart failure with reduced ejection fraction, atrial fibrillation is an independent predictor of mortality,” Dr. Di Biase said. “So I believe that staying in sinus rhythm may have affected the long-term mortality. If you have a treatment that reduces the amount of time in atrial fibrillation, you may reduce mortality.”
While catheter ablation is an increasingly popular treatment strategy in patients with drug-refractory paroxysmal AF, it has been understudied in the setting of AF and comorbid heart failure. These two conditions are commonly coexistent, and they feed on each other in a destructive way: AF worsens heart failure, and heart failure tends to make AF worse.
AATAC was funded by the participating investigators and institutions without external financial support. Dr. Di Biase reported serving as a consultant to Biosense Webster and St. Jude Medical and serving as a paid speaker for Atricure, Biotronik, Medtronic, Boston Scientific, and Epi EP.
SAN DIEGO – Catheter ablation proved superior to amiodarone for treatment of persistent atrial fibrillation in patients with systolic heart failure in the randomized AATAC-AF trial.
The rate of the primary study endpoint – freedom from recurrent AF through 26 months of prospective follow-up– was 70% in the catheter ablation group, twice the 34% rate with amiodarone, Dr. Luigi Di Biase reported at the annual meeting of the American College of Cardiology. After covariate adjustment, the investigators found that recurrence was 2.5 times more likely in the patients treated with amiodarone.

But he added a major caveat: pulmonary vein antrum isolation (PVI) alone was no better than the antiarrhythmic drug. The high overall treatment success rate seen with catheter ablation in the trial was achieved by operators who performed PVI plus some additional form of ablation of their own choosing, such as elimination of non–pulmonary vein triggers, ablation of complex fractionated electrograms, and/or additional linear ablation lesions, according to Dr. Di Biase, head of electrophysiology at the Albert Einstein College of Medicine, New York.
AATAC-AF (Ablation versus Amiodarone for Treatment of Atrial Fibrillation in Patients with Congestive Heart Failure and an Implanted ICD/CRTD) was a multicenter, prospective, randomized trial involving 203 patients with persistent AF and heart failure with reduced ejection fraction. Patients randomized to ablation had to receive PVI at a minimum; operators could perform additional ablation according to their preference. Twenty percent of patients randomized to ablation received PVI alone; 80% underwent additional posterior wall and non–pulmonary vein trigger ablation. The 26-month rate of freedom from recurrence of AF was 36% in patients who received PVI alone and 79% in those who underwent more extensive ablations. A particular strength of the AATAC study was that all participants had an implantable cardioverter-defibrillator and/or cardiac resynchronization therapy device, permitting detection of AF with a much higher degree of accuracy than possible in most AF ablation trials.
Any recurrent AF episodes during the first 3 months of follow-up were excluded from the analysis, regardless of whether patients were in the ablation or amiodarone arms, in accord with the 3-month blanking period that’s standard among electrophysiologists. Patients averaged 1.4 ablation sessions during the first 3 months of the trial.
Regardless of treatment, patients in whom AF did not recur showed significantly greater improvement in left ventricular ejection fraction, exercise capacity, and heart failure–related quality of life.
In addition, all-cause mortality during follow-up was significantly lower in the ablation group: 8%, compared with 18% in patients assigned to amiodarone. Moreover, the rate of hospitalization for arrhythmia or worsening heart failure was 31% in the ablation group versus 57% in patients on amiodarone. The economic implications of this sharp reduction in hospitalizations will be the subject of further study, according to Dr. Di Biase.
Also noteworthy was the finding that seven patients had to discontinue amiodarone due to serious side effects: four because of thyroid toxicity, two for pulmonary toxicity, and one owing to hepatic dysfunction, he continued.
Discussant Dr. Richard I. Fogel, current president of the Heart Rhythm Society, commented that “the 70% arrhythmia-free follow-up was a little surprising to me.”
“That seems a little bit high, particularly in a group with persistent atrial fibrillation,” observed Dr. Fogel, who is chief executive officer at St. Vincent Medical Group, Indianapolis.
Dr. Di Biase attributed the high success rate to two factors: One, only highly experienced operators participated in AATAC, and two, most of them weren’t content to stick to PVI alone.
“If you try to do a more extensive procedure addressing non–pulmonary vein triggers in other areas in the left atrium, the success rate is increased by far,” the electrophysiologist said.
As for a possible mechanism for the mortality benefit seen with ablation, “several studies have shown that in a population with heart failure with reduced ejection fraction, atrial fibrillation is an independent predictor of mortality,” Dr. Di Biase said. “So I believe that staying in sinus rhythm may have affected the long-term mortality. If you have a treatment that reduces the amount of time in atrial fibrillation, you may reduce mortality.”
While catheter ablation is an increasingly popular treatment strategy in patients with drug-refractory paroxysmal AF, it has been understudied in the setting of AF and comorbid heart failure. These two conditions are commonly coexistent, and they feed on each other in a destructive way: AF worsens heart failure, and heart failure tends to make AF worse.
AATAC was funded by the participating investigators and institutions without external financial support. Dr. Di Biase reported serving as a consultant to Biosense Webster and St. Jude Medical and serving as a paid speaker for Atricure, Biotronik, Medtronic, Boston Scientific, and Epi EP.
SAN DIEGO – Catheter ablation proved superior to amiodarone for treatment of persistent atrial fibrillation in patients with systolic heart failure in the randomized AATAC-AF trial.
The rate of the primary study endpoint – freedom from recurrent AF through 26 months of prospective follow-up– was 70% in the catheter ablation group, twice the 34% rate with amiodarone, Dr. Luigi Di Biase reported at the annual meeting of the American College of Cardiology. After covariate adjustment, the investigators found that recurrence was 2.5 times more likely in the patients treated with amiodarone.

But he added a major caveat: pulmonary vein antrum isolation (PVI) alone was no better than the antiarrhythmic drug. The high overall treatment success rate seen with catheter ablation in the trial was achieved by operators who performed PVI plus some additional form of ablation of their own choosing, such as elimination of non–pulmonary vein triggers, ablation of complex fractionated electrograms, and/or additional linear ablation lesions, according to Dr. Di Biase, head of electrophysiology at the Albert Einstein College of Medicine, New York.
AATAC-AF (Ablation versus Amiodarone for Treatment of Atrial Fibrillation in Patients with Congestive Heart Failure and an Implanted ICD/CRTD) was a multicenter, prospective, randomized trial involving 203 patients with persistent AF and heart failure with reduced ejection fraction. Patients randomized to ablation had to receive PVI at a minimum; operators could perform additional ablation according to their preference. Twenty percent of patients randomized to ablation received PVI alone; 80% underwent additional posterior wall and non–pulmonary vein trigger ablation. The 26-month rate of freedom from recurrence of AF was 36% in patients who received PVI alone and 79% in those who underwent more extensive ablations. A particular strength of the AATAC study was that all participants had an implantable cardioverter-defibrillator and/or cardiac resynchronization therapy device, permitting detection of AF with a much higher degree of accuracy than possible in most AF ablation trials.
Any recurrent AF episodes during the first 3 months of follow-up were excluded from the analysis, regardless of whether patients were in the ablation or amiodarone arms, in accord with the 3-month blanking period that’s standard among electrophysiologists. Patients averaged 1.4 ablation sessions during the first 3 months of the trial.
Regardless of treatment, patients in whom AF did not recur showed significantly greater improvement in left ventricular ejection fraction, exercise capacity, and heart failure–related quality of life.
In addition, all-cause mortality during follow-up was significantly lower in the ablation group: 8%, compared with 18% in patients assigned to amiodarone. Moreover, the rate of hospitalization for arrhythmia or worsening heart failure was 31% in the ablation group versus 57% in patients on amiodarone. The economic implications of this sharp reduction in hospitalizations will be the subject of further study, according to Dr. Di Biase.
Also noteworthy was the finding that seven patients had to discontinue amiodarone due to serious side effects: four because of thyroid toxicity, two for pulmonary toxicity, and one owing to hepatic dysfunction, he continued.
Discussant Dr. Richard I. Fogel, current president of the Heart Rhythm Society, commented that “the 70% arrhythmia-free follow-up was a little surprising to me.”
“That seems a little bit high, particularly in a group with persistent atrial fibrillation,” observed Dr. Fogel, who is chief executive officer at St. Vincent Medical Group, Indianapolis.
Dr. Di Biase attributed the high success rate to two factors: One, only highly experienced operators participated in AATAC, and two, most of them weren’t content to stick to PVI alone.
“If you try to do a more extensive procedure addressing non–pulmonary vein triggers in other areas in the left atrium, the success rate is increased by far,” the electrophysiologist said.
As for a possible mechanism for the mortality benefit seen with ablation, “several studies have shown that in a population with heart failure with reduced ejection fraction, atrial fibrillation is an independent predictor of mortality,” Dr. Di Biase said. “So I believe that staying in sinus rhythm may have affected the long-term mortality. If you have a treatment that reduces the amount of time in atrial fibrillation, you may reduce mortality.”
While catheter ablation is an increasingly popular treatment strategy in patients with drug-refractory paroxysmal AF, it has been understudied in the setting of AF and comorbid heart failure. These two conditions are commonly coexistent, and they feed on each other in a destructive way: AF worsens heart failure, and heart failure tends to make AF worse.
AATAC was funded by the participating investigators and institutions without external financial support. Dr. Di Biase reported serving as a consultant to Biosense Webster and St. Jude Medical and serving as a paid speaker for Atricure, Biotronik, Medtronic, Boston Scientific, and Epi EP.
AT ACC 15
Key clinical point: Catheter ablation is hands down more effective than amiodarone for the treatment of persistent atrial fibrillation in patients with systolic heart failure.
Major finding: The rate of freedom from recurrent atrial fibrillation during 26 months of follow-up was 70% in patients randomized to catheter ablation, compared with 34% in those assigned to amiodarone.
Data source: The AATAC-AF study was a multicenter, randomized, prospective clinical trial inc 203 patients.
Disclosures: The trial was funded by the participating investigators and institutions without commercial support. Dr. Di Biase reported serving as a consultant to Biosense Webster and St. Jude Medical and serving as a paid speaker for Atricure, Biotronik, Medtronic, Boston Scientific, and Epi EP.
Baroreflex activation therapy improves heart failure symptoms
SAN DIEGO– Baroreflex activation therapy showed promise as an important new device therapy for patients with New York Heart Association Class III heart failure, according to researchers.
In a preliminary randomized trial, the investigational therapy demonstrated safety comparable to that of established device therapies for heart failure. And it significantly improved functional status, exercise capacity, and quality of life while reducing levels of the biomarker N-terminal pro-brain natriuretic peptide (NT-proBNP) and days-in-hospital for worsening heart failure, Dr. William T. Abraham said at the annual meeting of the American College of Cardiology.
“The trial showed important results,” he said. “If these observations are confirmed in larger studies, baroreflex activation therapy may offer a new addition for the treatment of advanced heart failure patients with a reduced left ventricular ejection fraction.”
Novel therapies for patients with NYHA class III heart failure with a reduced ejection fraction (HFrEF) are sorely needed, he added, as 25%-35% of patients with HFrEF remain in NYHA class III despite current drug and device therapies. These patients are moderately symptomatic, meaning they are sick enough that their quality of life is sharply diminished, but not sufficiently ill to qualify for advanced heart therapies, such as cardiac transplantation or a left ventricular assist device.
Dr. Abraham presented the findings of a multinational, prospective, randomized, 6-month, controlled trial involving 140 NYHA class III HFrEF patients in the United States, Canada, Germany, and France. They were randomized to optimal guideline-directed medical therapy alone or in conjunction with baroreflex activation therapy (BAT), a form of neuromodulatory therapy involving electrical stimulation of the carotid baroreflex baroreceptor delivered by an implanted device similar to a pacemaker.
Progressive heart failure is characterized by increased sympathetic and reduced parasympathetic nerve activity. BAT addresses both abnormalities.
“This form of neuromodulation differs from other forms of neuromodulation in that it does not target a peripheral efferent nerve, but rather it targets the carotid baroreceptor. It targets afferent signals to the brain, which then produce an integrated autonomic nervous system response resulting in inhibition of sympathetic activity and enhancement of parasympathetic activity. So this is a physiologic form of autonomic rebalancing that is mediated via the CNS,” explained Dr. Abraham, professor of medicine and director of the division of cardiovascular medicine at Ohio State University, Columbus.
The primary safety endpoint was freedom from system- and procedure-related major adverse neurologic and cardiovascular events at 6 months. The rate was 97.2%. There were no deaths, and complications – all of which occurred within the first 7 days – were few and short lived, with rates similar to those seen with implantable cardioverter-defibrillators (ICDs) and cardiac resynchronization therapy (CRT). The BAT device, known as the CVRx Barostim neo, did not interact with the ICDs and CRT devices present in a large number of participants. No hypotension occurred in this normotensive HFrEF population.
To place the efficacy outcomes in perspective, Dr. Abraham continued, it’s worth noting that the average 19.5-point between-group difference favoring BAT in scores on the Minnesota Living with Heart Failure Quality of Life Questionnaire (MLHFQ) at 6 months compared to a baseline of 45 points dwarfs the benefits obtainable with standard therapies.
“Please remember that our best drug therapies for HFrEF – ACE inhibitors and beta blockers– improve the MLHFQ score by 4 or 5 points, and cardiac resynchronization therapy improves that score by an average of 9 or 10 points,” he said. From a baseline of 300 meters, the 6-minute hall walk distance improved by 58 meters more in the BAT group than in controls. By comparison, CRT improves the distance walked in 6 minutes by about 30 meters.
“The magnitude of benefit of BAT exceeds that seen with standard therapies, it was seen on top of those standard therapies, and it certainly falls into a range that would be considered clinically meaningful,” the cardiologist asserted.
Left ventricular ejection fraction improved in the BAT group by an average of 2.4% from a baseline of 24% while decreasing by 0.1% in controls. The between-group difference in NT-proBNP at 6 months was 342 pg/mL in favor of the BAT group, starting from a baseline level of roughly 1,300 pg/mL.
The BAT group averaged 6.95 hospital days per year for worsening heart failure during the 6 months prior to enrollment and 0.67 days per year in the 6 months following device activation, for an adjusted 82% relative risk reduction. In contrast, hospital days for heart failure remained steady in controls. The possibility of a new treatment that reduces heart failure hospitalizations is of particular interest in light of the enormous financial burden such hospitalizations currently place upon the Medicare system.
Simultaneously with Dr. Abraham’s presentation at ACC 15, the study manuscript was published online (JACC: Heart Failure 2015 [doi: 10.1016/j.jchf.2015.02.006]).
Dr. Abraham is a consultant to CVRx (which funded the study), as well as Novartis, St. Jude Medical, CardioMEMS, and Abbott Vascular.
SAN DIEGO– Baroreflex activation therapy showed promise as an important new device therapy for patients with New York Heart Association Class III heart failure, according to researchers.
In a preliminary randomized trial, the investigational therapy demonstrated safety comparable to that of established device therapies for heart failure. And it significantly improved functional status, exercise capacity, and quality of life while reducing levels of the biomarker N-terminal pro-brain natriuretic peptide (NT-proBNP) and days-in-hospital for worsening heart failure, Dr. William T. Abraham said at the annual meeting of the American College of Cardiology.
“The trial showed important results,” he said. “If these observations are confirmed in larger studies, baroreflex activation therapy may offer a new addition for the treatment of advanced heart failure patients with a reduced left ventricular ejection fraction.”
Novel therapies for patients with NYHA class III heart failure with a reduced ejection fraction (HFrEF) are sorely needed, he added, as 25%-35% of patients with HFrEF remain in NYHA class III despite current drug and device therapies. These patients are moderately symptomatic, meaning they are sick enough that their quality of life is sharply diminished, but not sufficiently ill to qualify for advanced heart therapies, such as cardiac transplantation or a left ventricular assist device.
Dr. Abraham presented the findings of a multinational, prospective, randomized, 6-month, controlled trial involving 140 NYHA class III HFrEF patients in the United States, Canada, Germany, and France. They were randomized to optimal guideline-directed medical therapy alone or in conjunction with baroreflex activation therapy (BAT), a form of neuromodulatory therapy involving electrical stimulation of the carotid baroreflex baroreceptor delivered by an implanted device similar to a pacemaker.
Progressive heart failure is characterized by increased sympathetic and reduced parasympathetic nerve activity. BAT addresses both abnormalities.
“This form of neuromodulation differs from other forms of neuromodulation in that it does not target a peripheral efferent nerve, but rather it targets the carotid baroreceptor. It targets afferent signals to the brain, which then produce an integrated autonomic nervous system response resulting in inhibition of sympathetic activity and enhancement of parasympathetic activity. So this is a physiologic form of autonomic rebalancing that is mediated via the CNS,” explained Dr. Abraham, professor of medicine and director of the division of cardiovascular medicine at Ohio State University, Columbus.
The primary safety endpoint was freedom from system- and procedure-related major adverse neurologic and cardiovascular events at 6 months. The rate was 97.2%. There were no deaths, and complications – all of which occurred within the first 7 days – were few and short lived, with rates similar to those seen with implantable cardioverter-defibrillators (ICDs) and cardiac resynchronization therapy (CRT). The BAT device, known as the CVRx Barostim neo, did not interact with the ICDs and CRT devices present in a large number of participants. No hypotension occurred in this normotensive HFrEF population.
To place the efficacy outcomes in perspective, Dr. Abraham continued, it’s worth noting that the average 19.5-point between-group difference favoring BAT in scores on the Minnesota Living with Heart Failure Quality of Life Questionnaire (MLHFQ) at 6 months compared to a baseline of 45 points dwarfs the benefits obtainable with standard therapies.
“Please remember that our best drug therapies for HFrEF – ACE inhibitors and beta blockers– improve the MLHFQ score by 4 or 5 points, and cardiac resynchronization therapy improves that score by an average of 9 or 10 points,” he said. From a baseline of 300 meters, the 6-minute hall walk distance improved by 58 meters more in the BAT group than in controls. By comparison, CRT improves the distance walked in 6 minutes by about 30 meters.
“The magnitude of benefit of BAT exceeds that seen with standard therapies, it was seen on top of those standard therapies, and it certainly falls into a range that would be considered clinically meaningful,” the cardiologist asserted.
Left ventricular ejection fraction improved in the BAT group by an average of 2.4% from a baseline of 24% while decreasing by 0.1% in controls. The between-group difference in NT-proBNP at 6 months was 342 pg/mL in favor of the BAT group, starting from a baseline level of roughly 1,300 pg/mL.
The BAT group averaged 6.95 hospital days per year for worsening heart failure during the 6 months prior to enrollment and 0.67 days per year in the 6 months following device activation, for an adjusted 82% relative risk reduction. In contrast, hospital days for heart failure remained steady in controls. The possibility of a new treatment that reduces heart failure hospitalizations is of particular interest in light of the enormous financial burden such hospitalizations currently place upon the Medicare system.
Simultaneously with Dr. Abraham’s presentation at ACC 15, the study manuscript was published online (JACC: Heart Failure 2015 [doi: 10.1016/j.jchf.2015.02.006]).
Dr. Abraham is a consultant to CVRx (which funded the study), as well as Novartis, St. Jude Medical, CardioMEMS, and Abbott Vascular.
SAN DIEGO– Baroreflex activation therapy showed promise as an important new device therapy for patients with New York Heart Association Class III heart failure, according to researchers.
In a preliminary randomized trial, the investigational therapy demonstrated safety comparable to that of established device therapies for heart failure. And it significantly improved functional status, exercise capacity, and quality of life while reducing levels of the biomarker N-terminal pro-brain natriuretic peptide (NT-proBNP) and days-in-hospital for worsening heart failure, Dr. William T. Abraham said at the annual meeting of the American College of Cardiology.
“The trial showed important results,” he said. “If these observations are confirmed in larger studies, baroreflex activation therapy may offer a new addition for the treatment of advanced heart failure patients with a reduced left ventricular ejection fraction.”
Novel therapies for patients with NYHA class III heart failure with a reduced ejection fraction (HFrEF) are sorely needed, he added, as 25%-35% of patients with HFrEF remain in NYHA class III despite current drug and device therapies. These patients are moderately symptomatic, meaning they are sick enough that their quality of life is sharply diminished, but not sufficiently ill to qualify for advanced heart therapies, such as cardiac transplantation or a left ventricular assist device.
Dr. Abraham presented the findings of a multinational, prospective, randomized, 6-month, controlled trial involving 140 NYHA class III HFrEF patients in the United States, Canada, Germany, and France. They were randomized to optimal guideline-directed medical therapy alone or in conjunction with baroreflex activation therapy (BAT), a form of neuromodulatory therapy involving electrical stimulation of the carotid baroreflex baroreceptor delivered by an implanted device similar to a pacemaker.
Progressive heart failure is characterized by increased sympathetic and reduced parasympathetic nerve activity. BAT addresses both abnormalities.
“This form of neuromodulation differs from other forms of neuromodulation in that it does not target a peripheral efferent nerve, but rather it targets the carotid baroreceptor. It targets afferent signals to the brain, which then produce an integrated autonomic nervous system response resulting in inhibition of sympathetic activity and enhancement of parasympathetic activity. So this is a physiologic form of autonomic rebalancing that is mediated via the CNS,” explained Dr. Abraham, professor of medicine and director of the division of cardiovascular medicine at Ohio State University, Columbus.
The primary safety endpoint was freedom from system- and procedure-related major adverse neurologic and cardiovascular events at 6 months. The rate was 97.2%. There were no deaths, and complications – all of which occurred within the first 7 days – were few and short lived, with rates similar to those seen with implantable cardioverter-defibrillators (ICDs) and cardiac resynchronization therapy (CRT). The BAT device, known as the CVRx Barostim neo, did not interact with the ICDs and CRT devices present in a large number of participants. No hypotension occurred in this normotensive HFrEF population.
To place the efficacy outcomes in perspective, Dr. Abraham continued, it’s worth noting that the average 19.5-point between-group difference favoring BAT in scores on the Minnesota Living with Heart Failure Quality of Life Questionnaire (MLHFQ) at 6 months compared to a baseline of 45 points dwarfs the benefits obtainable with standard therapies.
“Please remember that our best drug therapies for HFrEF – ACE inhibitors and beta blockers– improve the MLHFQ score by 4 or 5 points, and cardiac resynchronization therapy improves that score by an average of 9 or 10 points,” he said. From a baseline of 300 meters, the 6-minute hall walk distance improved by 58 meters more in the BAT group than in controls. By comparison, CRT improves the distance walked in 6 minutes by about 30 meters.
“The magnitude of benefit of BAT exceeds that seen with standard therapies, it was seen on top of those standard therapies, and it certainly falls into a range that would be considered clinically meaningful,” the cardiologist asserted.
Left ventricular ejection fraction improved in the BAT group by an average of 2.4% from a baseline of 24% while decreasing by 0.1% in controls. The between-group difference in NT-proBNP at 6 months was 342 pg/mL in favor of the BAT group, starting from a baseline level of roughly 1,300 pg/mL.
The BAT group averaged 6.95 hospital days per year for worsening heart failure during the 6 months prior to enrollment and 0.67 days per year in the 6 months following device activation, for an adjusted 82% relative risk reduction. In contrast, hospital days for heart failure remained steady in controls. The possibility of a new treatment that reduces heart failure hospitalizations is of particular interest in light of the enormous financial burden such hospitalizations currently place upon the Medicare system.
Simultaneously with Dr. Abraham’s presentation at ACC 15, the study manuscript was published online (JACC: Heart Failure 2015 [doi: 10.1016/j.jchf.2015.02.006]).
Dr. Abraham is a consultant to CVRx (which funded the study), as well as Novartis, St. Jude Medical, CardioMEMS, and Abbott Vascular.
AT ACC 15
Key clinical point: Baroreflex activation therapy is safe and improves multiple aspects of NYHA class III heart failure with reduced ejection fraction.
Major finding: Fifty-five percent of patients assigned to baroreflex activation therapy plus optimal guideline-directed medical therapy showed at least a one-class-rank improvement at 6 months of follow-up, compared with 24% of controls on optimal medical therapy alone.
Data source: A multinational, prospective, randomized, 6-month, controlled clinical trial of 140 patients with NYHA class III HFrEF.
Disclosures: Dr. Abraham is a consultant to CVRx (which funded the study), as well as Novartis, St. Jude Medical, CardioMEMS, and Abbott Vascular.
VIDEO: Long-term PARTNER 1 data tip scales toward TAVR
SAN DIEGO – The 5-year results of the PARTNER 1 trial in presented at the annual meeting of the American College of Cardiology were reassuring for clinicians treating patients with severe aortic stenosis at high risk for surgery, said to Dr. Jeffrey J. Popma of Beth Israel DeaconessMedical Center, Boston, in a video interview. The data showed comparable mortality between transcatheter aortic valve replacement (TAVR) and surgical aortic valve replacement (SAVR), as well as long-term durability of the SAPIEN transcatheter valve, he said.
With these similar outcome results, Dr. Popma asks, can the less-invasive TAVR procedure be considered the preferred treatment over SAVR in these very-sick patients?
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN DIEGO – The 5-year results of the PARTNER 1 trial in presented at the annual meeting of the American College of Cardiology were reassuring for clinicians treating patients with severe aortic stenosis at high risk for surgery, said to Dr. Jeffrey J. Popma of Beth Israel DeaconessMedical Center, Boston, in a video interview. The data showed comparable mortality between transcatheter aortic valve replacement (TAVR) and surgical aortic valve replacement (SAVR), as well as long-term durability of the SAPIEN transcatheter valve, he said.
With these similar outcome results, Dr. Popma asks, can the less-invasive TAVR procedure be considered the preferred treatment over SAVR in these very-sick patients?
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN DIEGO – The 5-year results of the PARTNER 1 trial in presented at the annual meeting of the American College of Cardiology were reassuring for clinicians treating patients with severe aortic stenosis at high risk for surgery, said to Dr. Jeffrey J. Popma of Beth Israel DeaconessMedical Center, Boston, in a video interview. The data showed comparable mortality between transcatheter aortic valve replacement (TAVR) and surgical aortic valve replacement (SAVR), as well as long-term durability of the SAPIEN transcatheter valve, he said.
With these similar outcome results, Dr. Popma asks, can the less-invasive TAVR procedure be considered the preferred treatment over SAVR in these very-sick patients?
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT ACC 15
Five-year PARTNER 1 results held up
SAN DIEGO – Five years out, transcatheter aortic valve replacement beat standard therapy in patients with severe, inoperable aortic stenosis, and measured up to surgery in high-risk patients.
The final data from the PARTNER 1 data showed TAVR as an alternative to surgery for some high-risk surgical patients, Dr. Michael Mack reported at the annual scientific sessions of the American College of Cardiology. High-risk surgical patients had similar all-cause mortality, cardiovascular mortality, stroke, and hospital readmission rates, regardless of whether they underwent TAVR or surgical valve replacement, Dr. Mack of Baylor Scott & White Health in Plano, Texas, and his associates wrote in an article published online simultaneously with the presentation (Lancet 2015 Mar. 15 [doi: 10.1016/ S0140-6736(15)60308-7]). “Functional outcomes were also similar and preservation of valve hemodynamics was equivalent in both groups,” they wrote.
The trial also showed “a sustained benefit of TAVR” for inoperable aortic stenosis, “as measured by all-cause mortality, cardiovascular mortality, repeat hospital admission, and functional status. Valves were durable, with no increase in transvalvular gradient, attrition of valve area, or worsening of aortic regurgitation,” Dr. Samir Kapadia at the Cleveland Clinic in Ohio and his associates reported in an related article that was not presented at the ACC meeting but was published at the same time as Dr. Mack’s presentation (Lancet 2015 Mar. 15 [doi: 10.1016/ S0140-6736(15)60290-2]). Based on the findings, “TAVR should be strongly considered for patients who are not surgical candidates for aortic valve replacement,” the researchers added. “Appropriate selection of patients will help to maximize the benefit of TAVR and reduce mortality from coexisting severe comorbidities.”
The Placement of Aortic Transcatheter Valves (PARTNER 1) trial compared TAVR, standard nonsurgical treatment, and surgery in patients with severe, symptomatic aortic stenosis. The inoperable cohort included 358 patients who averaged 83 years of age. The high-risk cohort enrolled 699 patients whose overall average Society of Thoracic Surgeons (STS) Predicted Risk of Mortality Score was more than 11%. Inoperable patients were randomized to TAVR or standard treatment (usually including balloon aortic valvuloplasty), while high-risk patients were randomized to TAVR or surgical valve replacement.
Five years after treatment, almost 72% of TAVR patients in the inoperable cohort had died, compared with 94% of patients who received standard treatment (hazard ratio, 0.50; 95% confidence interval, 0.39-0.65; P < .0001), Dr. Kapadia and his associates reported. Notably, 86% (or 42 of 49) of surviving TAVR patients had New York Heart Association class 1 or 2 symptoms, compared with only 60% of patients who received standard treatment. Echocardiography did not reveal valve deterioration, the investigators said.
Patients in the high-risk group also faced substantial mortality – only about a third were alive 5 years after TAVR or surgery, Dr. Mack reported. Also, 14% of TAVR patients developed moderate to severe valvular regurgitation, compared with only 1% of the surgery group; P < .0001), and this complication was tied to lower survival, they wrote. “The clinical outcomes and valve performance in this trial might not reflect that of subsequent generations of balloon-expandable transcatheter valves, present operator expertise and experience, and more rigorous patient selection for TAVR,” he cautioned. “The patients selected for treatment in this trial, which started in 2007, are also representative of clinical practice at that time; clinicians have since refined patient selection, at least partly on the basis of early outcomes from this trial.”
Edwards Lifesciences funded the study. Several authors reported receiving travel reimbursements from Edwards Lifesciences and financial or consulting relationships with Abbott Vascular, Edwards Lifesciences, Medtronic, Thubrikar Aortic Valve, St Jude Medical, Philips Healthcare, Sorin Medical, DirectFlow, Boston Scientific, Cardiosolutions, ValvXchange, and Posthorax.
The PARTNER trial was perhaps unique in showing that the first generation of a medical device resulted in a substantial mortality benefit compared with standard treatment. However, some uncertainties remain. The conclusions apply only to appropriately selected patients because many more patients were screened than were enrolled. Other patient populations that might ultimately benefit most from treatment with these new technologies should become better defined over the coming years.
In patients with aortic stenosis who were unsuitable for surgery, transcatheter aortic valve replacement provided a survival benefit of almost 22% [compared with standard treatment]... and a 28% lower cardiovascular mortality. Even more important for elderly patients is quality of life, and 86% of the 49 survivors who received TAVR had New York Heart Association (NYHA) functional class 1 or 2. A benefit of this size is remarkable for inoperable old patients treated with a first-generation medical device. However, a concern is that 48% of the patients undergoing TAVR were readmitted to hospital … and 34% of deaths were noncardiovascular. To treat one disease process, only for another to take its place, is not the objective of an invasive and expensive treatment with complications.
For high-risk patients, the clinical results of TAVR equaled those of SAVR, and the valve showed itself to be durable. The findings challenge whether surgery can still be considered the gold standard for patients at high surgical risk. In 2008, Dr. Mack predicted that the benefits of new, less invasive procedures for percutaneous heart valve treatment would equal or surpass those of their open-surgery predecessors, and concluded that patients will choose a less invasive approach over a more invasive one even if there is uncertainty. With more than 150,000 implantations worldwide and the indication shifting towards intermediate-risk patients, this prediction has been met.
Arie P. Kappetein, M.D., Ph.D., is a cardiothoracic surgeon at Erasmus University in Rotterdam, the Netherlands. These comments were excerpted from his accompanying editorial (Lancet 2015 Mar. 15 [doi: 10.1016/ S0140-6736(15)60448-2]).
The PARTNER trial was perhaps unique in showing that the first generation of a medical device resulted in a substantial mortality benefit compared with standard treatment. However, some uncertainties remain. The conclusions apply only to appropriately selected patients because many more patients were screened than were enrolled. Other patient populations that might ultimately benefit most from treatment with these new technologies should become better defined over the coming years.
In patients with aortic stenosis who were unsuitable for surgery, transcatheter aortic valve replacement provided a survival benefit of almost 22% [compared with standard treatment]... and a 28% lower cardiovascular mortality. Even more important for elderly patients is quality of life, and 86% of the 49 survivors who received TAVR had New York Heart Association (NYHA) functional class 1 or 2. A benefit of this size is remarkable for inoperable old patients treated with a first-generation medical device. However, a concern is that 48% of the patients undergoing TAVR were readmitted to hospital … and 34% of deaths were noncardiovascular. To treat one disease process, only for another to take its place, is not the objective of an invasive and expensive treatment with complications.
For high-risk patients, the clinical results of TAVR equaled those of SAVR, and the valve showed itself to be durable. The findings challenge whether surgery can still be considered the gold standard for patients at high surgical risk. In 2008, Dr. Mack predicted that the benefits of new, less invasive procedures for percutaneous heart valve treatment would equal or surpass those of their open-surgery predecessors, and concluded that patients will choose a less invasive approach over a more invasive one even if there is uncertainty. With more than 150,000 implantations worldwide and the indication shifting towards intermediate-risk patients, this prediction has been met.
Arie P. Kappetein, M.D., Ph.D., is a cardiothoracic surgeon at Erasmus University in Rotterdam, the Netherlands. These comments were excerpted from his accompanying editorial (Lancet 2015 Mar. 15 [doi: 10.1016/ S0140-6736(15)60448-2]).
The PARTNER trial was perhaps unique in showing that the first generation of a medical device resulted in a substantial mortality benefit compared with standard treatment. However, some uncertainties remain. The conclusions apply only to appropriately selected patients because many more patients were screened than were enrolled. Other patient populations that might ultimately benefit most from treatment with these new technologies should become better defined over the coming years.
In patients with aortic stenosis who were unsuitable for surgery, transcatheter aortic valve replacement provided a survival benefit of almost 22% [compared with standard treatment]... and a 28% lower cardiovascular mortality. Even more important for elderly patients is quality of life, and 86% of the 49 survivors who received TAVR had New York Heart Association (NYHA) functional class 1 or 2. A benefit of this size is remarkable for inoperable old patients treated with a first-generation medical device. However, a concern is that 48% of the patients undergoing TAVR were readmitted to hospital … and 34% of deaths were noncardiovascular. To treat one disease process, only for another to take its place, is not the objective of an invasive and expensive treatment with complications.
For high-risk patients, the clinical results of TAVR equaled those of SAVR, and the valve showed itself to be durable. The findings challenge whether surgery can still be considered the gold standard for patients at high surgical risk. In 2008, Dr. Mack predicted that the benefits of new, less invasive procedures for percutaneous heart valve treatment would equal or surpass those of their open-surgery predecessors, and concluded that patients will choose a less invasive approach over a more invasive one even if there is uncertainty. With more than 150,000 implantations worldwide and the indication shifting towards intermediate-risk patients, this prediction has been met.
Arie P. Kappetein, M.D., Ph.D., is a cardiothoracic surgeon at Erasmus University in Rotterdam, the Netherlands. These comments were excerpted from his accompanying editorial (Lancet 2015 Mar. 15 [doi: 10.1016/ S0140-6736(15)60448-2]).
SAN DIEGO – Five years out, transcatheter aortic valve replacement beat standard therapy in patients with severe, inoperable aortic stenosis, and measured up to surgery in high-risk patients.
The final data from the PARTNER 1 data showed TAVR as an alternative to surgery for some high-risk surgical patients, Dr. Michael Mack reported at the annual scientific sessions of the American College of Cardiology. High-risk surgical patients had similar all-cause mortality, cardiovascular mortality, stroke, and hospital readmission rates, regardless of whether they underwent TAVR or surgical valve replacement, Dr. Mack of Baylor Scott & White Health in Plano, Texas, and his associates wrote in an article published online simultaneously with the presentation (Lancet 2015 Mar. 15 [doi: 10.1016/ S0140-6736(15)60308-7]). “Functional outcomes were also similar and preservation of valve hemodynamics was equivalent in both groups,” they wrote.
The trial also showed “a sustained benefit of TAVR” for inoperable aortic stenosis, “as measured by all-cause mortality, cardiovascular mortality, repeat hospital admission, and functional status. Valves were durable, with no increase in transvalvular gradient, attrition of valve area, or worsening of aortic regurgitation,” Dr. Samir Kapadia at the Cleveland Clinic in Ohio and his associates reported in an related article that was not presented at the ACC meeting but was published at the same time as Dr. Mack’s presentation (Lancet 2015 Mar. 15 [doi: 10.1016/ S0140-6736(15)60290-2]). Based on the findings, “TAVR should be strongly considered for patients who are not surgical candidates for aortic valve replacement,” the researchers added. “Appropriate selection of patients will help to maximize the benefit of TAVR and reduce mortality from coexisting severe comorbidities.”
The Placement of Aortic Transcatheter Valves (PARTNER 1) trial compared TAVR, standard nonsurgical treatment, and surgery in patients with severe, symptomatic aortic stenosis. The inoperable cohort included 358 patients who averaged 83 years of age. The high-risk cohort enrolled 699 patients whose overall average Society of Thoracic Surgeons (STS) Predicted Risk of Mortality Score was more than 11%. Inoperable patients were randomized to TAVR or standard treatment (usually including balloon aortic valvuloplasty), while high-risk patients were randomized to TAVR or surgical valve replacement.
Five years after treatment, almost 72% of TAVR patients in the inoperable cohort had died, compared with 94% of patients who received standard treatment (hazard ratio, 0.50; 95% confidence interval, 0.39-0.65; P < .0001), Dr. Kapadia and his associates reported. Notably, 86% (or 42 of 49) of surviving TAVR patients had New York Heart Association class 1 or 2 symptoms, compared with only 60% of patients who received standard treatment. Echocardiography did not reveal valve deterioration, the investigators said.
Patients in the high-risk group also faced substantial mortality – only about a third were alive 5 years after TAVR or surgery, Dr. Mack reported. Also, 14% of TAVR patients developed moderate to severe valvular regurgitation, compared with only 1% of the surgery group; P < .0001), and this complication was tied to lower survival, they wrote. “The clinical outcomes and valve performance in this trial might not reflect that of subsequent generations of balloon-expandable transcatheter valves, present operator expertise and experience, and more rigorous patient selection for TAVR,” he cautioned. “The patients selected for treatment in this trial, which started in 2007, are also representative of clinical practice at that time; clinicians have since refined patient selection, at least partly on the basis of early outcomes from this trial.”
Edwards Lifesciences funded the study. Several authors reported receiving travel reimbursements from Edwards Lifesciences and financial or consulting relationships with Abbott Vascular, Edwards Lifesciences, Medtronic, Thubrikar Aortic Valve, St Jude Medical, Philips Healthcare, Sorin Medical, DirectFlow, Boston Scientific, Cardiosolutions, ValvXchange, and Posthorax.
SAN DIEGO – Five years out, transcatheter aortic valve replacement beat standard therapy in patients with severe, inoperable aortic stenosis, and measured up to surgery in high-risk patients.
The final data from the PARTNER 1 data showed TAVR as an alternative to surgery for some high-risk surgical patients, Dr. Michael Mack reported at the annual scientific sessions of the American College of Cardiology. High-risk surgical patients had similar all-cause mortality, cardiovascular mortality, stroke, and hospital readmission rates, regardless of whether they underwent TAVR or surgical valve replacement, Dr. Mack of Baylor Scott & White Health in Plano, Texas, and his associates wrote in an article published online simultaneously with the presentation (Lancet 2015 Mar. 15 [doi: 10.1016/ S0140-6736(15)60308-7]). “Functional outcomes were also similar and preservation of valve hemodynamics was equivalent in both groups,” they wrote.
The trial also showed “a sustained benefit of TAVR” for inoperable aortic stenosis, “as measured by all-cause mortality, cardiovascular mortality, repeat hospital admission, and functional status. Valves were durable, with no increase in transvalvular gradient, attrition of valve area, or worsening of aortic regurgitation,” Dr. Samir Kapadia at the Cleveland Clinic in Ohio and his associates reported in an related article that was not presented at the ACC meeting but was published at the same time as Dr. Mack’s presentation (Lancet 2015 Mar. 15 [doi: 10.1016/ S0140-6736(15)60290-2]). Based on the findings, “TAVR should be strongly considered for patients who are not surgical candidates for aortic valve replacement,” the researchers added. “Appropriate selection of patients will help to maximize the benefit of TAVR and reduce mortality from coexisting severe comorbidities.”
The Placement of Aortic Transcatheter Valves (PARTNER 1) trial compared TAVR, standard nonsurgical treatment, and surgery in patients with severe, symptomatic aortic stenosis. The inoperable cohort included 358 patients who averaged 83 years of age. The high-risk cohort enrolled 699 patients whose overall average Society of Thoracic Surgeons (STS) Predicted Risk of Mortality Score was more than 11%. Inoperable patients were randomized to TAVR or standard treatment (usually including balloon aortic valvuloplasty), while high-risk patients were randomized to TAVR or surgical valve replacement.
Five years after treatment, almost 72% of TAVR patients in the inoperable cohort had died, compared with 94% of patients who received standard treatment (hazard ratio, 0.50; 95% confidence interval, 0.39-0.65; P < .0001), Dr. Kapadia and his associates reported. Notably, 86% (or 42 of 49) of surviving TAVR patients had New York Heart Association class 1 or 2 symptoms, compared with only 60% of patients who received standard treatment. Echocardiography did not reveal valve deterioration, the investigators said.
Patients in the high-risk group also faced substantial mortality – only about a third were alive 5 years after TAVR or surgery, Dr. Mack reported. Also, 14% of TAVR patients developed moderate to severe valvular regurgitation, compared with only 1% of the surgery group; P < .0001), and this complication was tied to lower survival, they wrote. “The clinical outcomes and valve performance in this trial might not reflect that of subsequent generations of balloon-expandable transcatheter valves, present operator expertise and experience, and more rigorous patient selection for TAVR,” he cautioned. “The patients selected for treatment in this trial, which started in 2007, are also representative of clinical practice at that time; clinicians have since refined patient selection, at least partly on the basis of early outcomes from this trial.”
Edwards Lifesciences funded the study. Several authors reported receiving travel reimbursements from Edwards Lifesciences and financial or consulting relationships with Abbott Vascular, Edwards Lifesciences, Medtronic, Thubrikar Aortic Valve, St Jude Medical, Philips Healthcare, Sorin Medical, DirectFlow, Boston Scientific, Cardiosolutions, ValvXchange, and Posthorax.
Key clinical point:Transcatheter aortic valve replacement is an acceptable alternative to standard treatment in some patients with aortic stenosis.
Major finding:For inoperable patients, TAVR had lower five-year mortality than did standard therapy (P < .0001). For high-risk patients, TAVR and surgical mortality rates were similar.
Data source: Five-year data from the randomized Placement of Aortic Transcatheter Valves (PARTNER 1) trial.
Disclosures: Edwards Lifesciences funded the study. Several authors reported receiving travel reimbursements from Edwards Lifesciences and financial or consulting relationships with Abbott Vascular, Edwards Lifesciences, Medtronic, Thubrikar Aortic Valve, St Jude Medical, Philips Healthcare, Sorin Medical, DirectFlow, Boston Scientific, Cardiosolutions, ValvXchange, and Posthorax.
Digoxin linked to higher mortality in AF
SAN DIEGO – Digoxin increases the risk of death by 27% in patients with atrial fibrillation, a meta-analysis of 19 studies showed.
Patients with AF and kidney failure faced a 60% to 70 % increase in mortality compared to their counterparts not taking digoxin, according to a press release on the study.
A weaker association between digoxin and death was observed in AF patients who also had heart failure, a finding the authors suggest warrants further investigation.
“Until further research can be done, I would suggest physicians use caution when prescribing digoxin for patients with atrial fibrillation, especially given that there are alternative drugs available that might be safer,” lead author Dr. Waqas Qureshi said in a statement.
The results were released in advance of their March 15 presentation at the annual meeting of the American College of Cardiology in San Diego.
About 5.6 million Americans have atrial fibrillation (AF) and roughly 1 in 5 are prescribed digoxin for heart rate control.
Current guidelines recommend digoxin as first-line therapy in patients who aren’t physically active and as a second-line drug for more active patients.
“Based on consistent results coming out of many studies, our results suggest digoxin should be downgraded from its position as a front-line agent for certain patients with atrial fibrillation,” Dr. Qureshi, a clinical and research cardiology fellow at Wake Forest School of Medicine in Winston-Salem, N.C., recommended.
The authors reviewed 19 studies including five cohort and randomized controlled trials involving 501,681 patients. Of these, 458,311 patients had AF and 111,978 were prescribed digoxin.
In a random effects model, digoxin was associated with an increased risk of mortality, with a pooled hazard ratio of 1.27 (95% confidence interval 1.19-1.36; P value < .001).
Several studies in the meta-analysis suggest that higher blood levels of digoxin increase the risk of death. The mechanism behind the increased mortality is not known, although previous studies have suggested digoxin increases the risk of thromboembolism.
The meta-analysis accounted for risk factors and co-morbidities reported in the various studies, but it’s possible that some confounding factors may not have been accounted for, the authors acknowledge.
“The study points to the need for a well-structured, targeted trial to investigate digoxin’s safety,” Dr. Qureshi stated.
Digoxin remains a commonly used agent for control of ventricular rate in atrial fibrillation (AF) and is accepted as a valid therapy. Despite endorsement of digoxin in clinical practice guidelines for rate control in atrial fibrillation, there are only limited, conflicting, and mostly older observational data on the safety of digoxin in AF. There have been no appropriately designed clinical trials to assess the safety of digoxin in any patient population. In heart failure cohorts, the effectiveness and safety of digoxin has been shown to vary by serum digoxin concentrations, indicating possible moderation by kidney function.
The meta-analysis of five cohort studies and randomized controlled trials by Dr. Qureshi and colleagues concludes that digoxin is associated with a 27% increased risk of mortality in patients with AF. These results confirm another recently published analysis with similar conclusions, TREAT-AF (J. Am. Coll. Cardiol. 2014;64:660-8).
The TREAT-AF study was a retrospective analysis of patients with newly diagnosed AF. In this study, treatment with digoxin was independently associated with mortality, regardless of age, sex, kidney function, heart failure status, concomitant therapies, or drug adherence. Sensitivity analyses to assess the possible impact of unmeasured confounders make it highly unlikely that any influenced the result of the TREAT-AF Study.
Prospective studies are needed to confirm the findings of these observational reports and to explore the mechanisms responsible for the increased risk of mortality in patients with AF treated with digoxin. In the meantime, physicians should consider alternatives to digoxin in managing patients with AF.
N.A. Mark Estes III, MD, is professor of medicine at Tufts University, Boston. He has no relevant disclosures.
Digoxin remains a commonly used agent for control of ventricular rate in atrial fibrillation (AF) and is accepted as a valid therapy. Despite endorsement of digoxin in clinical practice guidelines for rate control in atrial fibrillation, there are only limited, conflicting, and mostly older observational data on the safety of digoxin in AF. There have been no appropriately designed clinical trials to assess the safety of digoxin in any patient population. In heart failure cohorts, the effectiveness and safety of digoxin has been shown to vary by serum digoxin concentrations, indicating possible moderation by kidney function.
The meta-analysis of five cohort studies and randomized controlled trials by Dr. Qureshi and colleagues concludes that digoxin is associated with a 27% increased risk of mortality in patients with AF. These results confirm another recently published analysis with similar conclusions, TREAT-AF (J. Am. Coll. Cardiol. 2014;64:660-8).
The TREAT-AF study was a retrospective analysis of patients with newly diagnosed AF. In this study, treatment with digoxin was independently associated with mortality, regardless of age, sex, kidney function, heart failure status, concomitant therapies, or drug adherence. Sensitivity analyses to assess the possible impact of unmeasured confounders make it highly unlikely that any influenced the result of the TREAT-AF Study.
Prospective studies are needed to confirm the findings of these observational reports and to explore the mechanisms responsible for the increased risk of mortality in patients with AF treated with digoxin. In the meantime, physicians should consider alternatives to digoxin in managing patients with AF.
N.A. Mark Estes III, MD, is professor of medicine at Tufts University, Boston. He has no relevant disclosures.
Digoxin remains a commonly used agent for control of ventricular rate in atrial fibrillation (AF) and is accepted as a valid therapy. Despite endorsement of digoxin in clinical practice guidelines for rate control in atrial fibrillation, there are only limited, conflicting, and mostly older observational data on the safety of digoxin in AF. There have been no appropriately designed clinical trials to assess the safety of digoxin in any patient population. In heart failure cohorts, the effectiveness and safety of digoxin has been shown to vary by serum digoxin concentrations, indicating possible moderation by kidney function.
The meta-analysis of five cohort studies and randomized controlled trials by Dr. Qureshi and colleagues concludes that digoxin is associated with a 27% increased risk of mortality in patients with AF. These results confirm another recently published analysis with similar conclusions, TREAT-AF (J. Am. Coll. Cardiol. 2014;64:660-8).
The TREAT-AF study was a retrospective analysis of patients with newly diagnosed AF. In this study, treatment with digoxin was independently associated with mortality, regardless of age, sex, kidney function, heart failure status, concomitant therapies, or drug adherence. Sensitivity analyses to assess the possible impact of unmeasured confounders make it highly unlikely that any influenced the result of the TREAT-AF Study.
Prospective studies are needed to confirm the findings of these observational reports and to explore the mechanisms responsible for the increased risk of mortality in patients with AF treated with digoxin. In the meantime, physicians should consider alternatives to digoxin in managing patients with AF.
N.A. Mark Estes III, MD, is professor of medicine at Tufts University, Boston. He has no relevant disclosures.
SAN DIEGO – Digoxin increases the risk of death by 27% in patients with atrial fibrillation, a meta-analysis of 19 studies showed.
Patients with AF and kidney failure faced a 60% to 70 % increase in mortality compared to their counterparts not taking digoxin, according to a press release on the study.
A weaker association between digoxin and death was observed in AF patients who also had heart failure, a finding the authors suggest warrants further investigation.
“Until further research can be done, I would suggest physicians use caution when prescribing digoxin for patients with atrial fibrillation, especially given that there are alternative drugs available that might be safer,” lead author Dr. Waqas Qureshi said in a statement.
The results were released in advance of their March 15 presentation at the annual meeting of the American College of Cardiology in San Diego.
About 5.6 million Americans have atrial fibrillation (AF) and roughly 1 in 5 are prescribed digoxin for heart rate control.
Current guidelines recommend digoxin as first-line therapy in patients who aren’t physically active and as a second-line drug for more active patients.
“Based on consistent results coming out of many studies, our results suggest digoxin should be downgraded from its position as a front-line agent for certain patients with atrial fibrillation,” Dr. Qureshi, a clinical and research cardiology fellow at Wake Forest School of Medicine in Winston-Salem, N.C., recommended.
The authors reviewed 19 studies including five cohort and randomized controlled trials involving 501,681 patients. Of these, 458,311 patients had AF and 111,978 were prescribed digoxin.
In a random effects model, digoxin was associated with an increased risk of mortality, with a pooled hazard ratio of 1.27 (95% confidence interval 1.19-1.36; P value < .001).
Several studies in the meta-analysis suggest that higher blood levels of digoxin increase the risk of death. The mechanism behind the increased mortality is not known, although previous studies have suggested digoxin increases the risk of thromboembolism.
The meta-analysis accounted for risk factors and co-morbidities reported in the various studies, but it’s possible that some confounding factors may not have been accounted for, the authors acknowledge.
“The study points to the need for a well-structured, targeted trial to investigate digoxin’s safety,” Dr. Qureshi stated.
SAN DIEGO – Digoxin increases the risk of death by 27% in patients with atrial fibrillation, a meta-analysis of 19 studies showed.
Patients with AF and kidney failure faced a 60% to 70 % increase in mortality compared to their counterparts not taking digoxin, according to a press release on the study.
A weaker association between digoxin and death was observed in AF patients who also had heart failure, a finding the authors suggest warrants further investigation.
“Until further research can be done, I would suggest physicians use caution when prescribing digoxin for patients with atrial fibrillation, especially given that there are alternative drugs available that might be safer,” lead author Dr. Waqas Qureshi said in a statement.
The results were released in advance of their March 15 presentation at the annual meeting of the American College of Cardiology in San Diego.
About 5.6 million Americans have atrial fibrillation (AF) and roughly 1 in 5 are prescribed digoxin for heart rate control.
Current guidelines recommend digoxin as first-line therapy in patients who aren’t physically active and as a second-line drug for more active patients.
“Based on consistent results coming out of many studies, our results suggest digoxin should be downgraded from its position as a front-line agent for certain patients with atrial fibrillation,” Dr. Qureshi, a clinical and research cardiology fellow at Wake Forest School of Medicine in Winston-Salem, N.C., recommended.
The authors reviewed 19 studies including five cohort and randomized controlled trials involving 501,681 patients. Of these, 458,311 patients had AF and 111,978 were prescribed digoxin.
In a random effects model, digoxin was associated with an increased risk of mortality, with a pooled hazard ratio of 1.27 (95% confidence interval 1.19-1.36; P value < .001).
Several studies in the meta-analysis suggest that higher blood levels of digoxin increase the risk of death. The mechanism behind the increased mortality is not known, although previous studies have suggested digoxin increases the risk of thromboembolism.
The meta-analysis accounted for risk factors and co-morbidities reported in the various studies, but it’s possible that some confounding factors may not have been accounted for, the authors acknowledge.
“The study points to the need for a well-structured, targeted trial to investigate digoxin’s safety,” Dr. Qureshi stated.
FROM ACC 2015
Key clinical point: Alternatives to digoxin should be considered when prescribing for patients with atrial fibrillation.
Major finding: Digoxin was associated with an increased risk of mortality in patients with AF (Hazard ratio, 1.27; P < 001).
Data source: Pooled analysis of 19 studies involving 501,681 patients, 458,311 with atrial fibrillation.
Disclosures: Dr. Qureshi and his co-authors reported having no financial disclosures.
Emergency department holds key to early readmissions for heart failure
SNOWMASS, COLO. – Under intense fiscal pressure to curb early hospital readmissions for heart failure, cardiologists and hospital administrators are taking a hard look at the traditional role of the emergency department as the point of triage for patients with decompensated heart failure.
“Alternatives to the emergency department for ambulatory triage and intervention are essential,” Dr. Akshay Desai said at the Annual Cardiovascular Conference at Snowmass.
“Traditionally, when our patients become decompensated, we send them from our office or clinic to the ED. And 80%-90% of those who present to the ED with the diagnosis of heart failure are admitted to the hospital. So this means that the ED is a pretty ineffective triage point for heart failure patients. Most ED staff are concerned about ambulatory follow-up and feel it’s safer to follow patients in the hospital. The message here is we need a more robust ambulatory framework to manage patients with milder decompensation so they don’t all need to come into the hospital,” said Dr. Desai of Brigham and Women’s Hospital and Harvard Medical School, Boston.
Hospital admission is of questionable value for a large fraction of decompensating heart failure patients. After all, not that much happens to them in the hospital that couldn’t take place in a less costly setting.
“For the most part, our patients in the hospital for decompensated heart failure get IV diuretics, with an average weight loss of about 4 kg. Surveillance is typically once- or twice-daily laboratory tests and a bedside clinical visit by a physician at 7:30 in the morning. Few patients get much else. There’s little diagnostic testing, few other therapies that require intensive monitoring, and most patients feel a little better by their first day in the hospital,” he said.
This suggests the need for what he called “an evolved model of heart failure care” in which the ED is replaced as the point of service by some form of ambulatory center that can serve as a buffer limiting the number of patients who need to come into the hospital.
“It could be a home-based strategy of IV diuretic administration, a clinic-based strategy of outpatient diuretic administration, or an observation unit based in the ED. All of these are now being tested in various models across the country as alternatives to help manage the readmission problem, and also to make life better for our patients, who’d prefer not to be in the hospital if they could be managed in other ways,” Dr. Desai continued.
Reducing 30-day readmission rates after a hospitalization for heart failure is seen by health policy makers as an opportunity to simultaneously improve care and reduce costs. But studies show only about half of readmissions in patients with heart failure are cardiovascular related, just half of those cardiovascular readmissions are heart failure related, and only about 30% of heart failure readmissions are truly preventable. However, wide variation exists across the country in risk-adjusted 30-day readmission rates, suggesting there is an opportunity for improvement in outlier hospitals.
Numerous factors have been linked to high heart failure readmission rates, including patient sociodemographic characteristics, comorbid conditions, and serum markers of heart failure severity. One underappreciated factor, in Dr. Desai’s view, is that readmission rates are significantly higher in hospitals that are financially and clinically resource poor, as shown in a Harvard School of Public Health analysis of Medicare claims data for more than 900,000 heart failure discharges. These resource-poor hospitals provide care for underserved populations, and they are experiencing a disproportionate burden of the financial penalties imposed for early readmission.
“As we seek to improve care for patients with heart failure, we should ensure that penalties for poor performance do not worsen disparities in quality of care,” according to the Harvard investigators (Circ. Cardiovasc. Qual. Outcomes 2011;4:53-9).
Dr. Desai reported serving as a consultant to 5AM Ventures, AtCor Medical, Novartis, and St. Jude Medical.
SNOWMASS, COLO. – Under intense fiscal pressure to curb early hospital readmissions for heart failure, cardiologists and hospital administrators are taking a hard look at the traditional role of the emergency department as the point of triage for patients with decompensated heart failure.
“Alternatives to the emergency department for ambulatory triage and intervention are essential,” Dr. Akshay Desai said at the Annual Cardiovascular Conference at Snowmass.
“Traditionally, when our patients become decompensated, we send them from our office or clinic to the ED. And 80%-90% of those who present to the ED with the diagnosis of heart failure are admitted to the hospital. So this means that the ED is a pretty ineffective triage point for heart failure patients. Most ED staff are concerned about ambulatory follow-up and feel it’s safer to follow patients in the hospital. The message here is we need a more robust ambulatory framework to manage patients with milder decompensation so they don’t all need to come into the hospital,” said Dr. Desai of Brigham and Women’s Hospital and Harvard Medical School, Boston.
Hospital admission is of questionable value for a large fraction of decompensating heart failure patients. After all, not that much happens to them in the hospital that couldn’t take place in a less costly setting.
“For the most part, our patients in the hospital for decompensated heart failure get IV diuretics, with an average weight loss of about 4 kg. Surveillance is typically once- or twice-daily laboratory tests and a bedside clinical visit by a physician at 7:30 in the morning. Few patients get much else. There’s little diagnostic testing, few other therapies that require intensive monitoring, and most patients feel a little better by their first day in the hospital,” he said.
This suggests the need for what he called “an evolved model of heart failure care” in which the ED is replaced as the point of service by some form of ambulatory center that can serve as a buffer limiting the number of patients who need to come into the hospital.
“It could be a home-based strategy of IV diuretic administration, a clinic-based strategy of outpatient diuretic administration, or an observation unit based in the ED. All of these are now being tested in various models across the country as alternatives to help manage the readmission problem, and also to make life better for our patients, who’d prefer not to be in the hospital if they could be managed in other ways,” Dr. Desai continued.
Reducing 30-day readmission rates after a hospitalization for heart failure is seen by health policy makers as an opportunity to simultaneously improve care and reduce costs. But studies show only about half of readmissions in patients with heart failure are cardiovascular related, just half of those cardiovascular readmissions are heart failure related, and only about 30% of heart failure readmissions are truly preventable. However, wide variation exists across the country in risk-adjusted 30-day readmission rates, suggesting there is an opportunity for improvement in outlier hospitals.
Numerous factors have been linked to high heart failure readmission rates, including patient sociodemographic characteristics, comorbid conditions, and serum markers of heart failure severity. One underappreciated factor, in Dr. Desai’s view, is that readmission rates are significantly higher in hospitals that are financially and clinically resource poor, as shown in a Harvard School of Public Health analysis of Medicare claims data for more than 900,000 heart failure discharges. These resource-poor hospitals provide care for underserved populations, and they are experiencing a disproportionate burden of the financial penalties imposed for early readmission.
“As we seek to improve care for patients with heart failure, we should ensure that penalties for poor performance do not worsen disparities in quality of care,” according to the Harvard investigators (Circ. Cardiovasc. Qual. Outcomes 2011;4:53-9).
Dr. Desai reported serving as a consultant to 5AM Ventures, AtCor Medical, Novartis, and St. Jude Medical.
SNOWMASS, COLO. – Under intense fiscal pressure to curb early hospital readmissions for heart failure, cardiologists and hospital administrators are taking a hard look at the traditional role of the emergency department as the point of triage for patients with decompensated heart failure.
“Alternatives to the emergency department for ambulatory triage and intervention are essential,” Dr. Akshay Desai said at the Annual Cardiovascular Conference at Snowmass.
“Traditionally, when our patients become decompensated, we send them from our office or clinic to the ED. And 80%-90% of those who present to the ED with the diagnosis of heart failure are admitted to the hospital. So this means that the ED is a pretty ineffective triage point for heart failure patients. Most ED staff are concerned about ambulatory follow-up and feel it’s safer to follow patients in the hospital. The message here is we need a more robust ambulatory framework to manage patients with milder decompensation so they don’t all need to come into the hospital,” said Dr. Desai of Brigham and Women’s Hospital and Harvard Medical School, Boston.
Hospital admission is of questionable value for a large fraction of decompensating heart failure patients. After all, not that much happens to them in the hospital that couldn’t take place in a less costly setting.
“For the most part, our patients in the hospital for decompensated heart failure get IV diuretics, with an average weight loss of about 4 kg. Surveillance is typically once- or twice-daily laboratory tests and a bedside clinical visit by a physician at 7:30 in the morning. Few patients get much else. There’s little diagnostic testing, few other therapies that require intensive monitoring, and most patients feel a little better by their first day in the hospital,” he said.
This suggests the need for what he called “an evolved model of heart failure care” in which the ED is replaced as the point of service by some form of ambulatory center that can serve as a buffer limiting the number of patients who need to come into the hospital.
“It could be a home-based strategy of IV diuretic administration, a clinic-based strategy of outpatient diuretic administration, or an observation unit based in the ED. All of these are now being tested in various models across the country as alternatives to help manage the readmission problem, and also to make life better for our patients, who’d prefer not to be in the hospital if they could be managed in other ways,” Dr. Desai continued.
Reducing 30-day readmission rates after a hospitalization for heart failure is seen by health policy makers as an opportunity to simultaneously improve care and reduce costs. But studies show only about half of readmissions in patients with heart failure are cardiovascular related, just half of those cardiovascular readmissions are heart failure related, and only about 30% of heart failure readmissions are truly preventable. However, wide variation exists across the country in risk-adjusted 30-day readmission rates, suggesting there is an opportunity for improvement in outlier hospitals.
Numerous factors have been linked to high heart failure readmission rates, including patient sociodemographic characteristics, comorbid conditions, and serum markers of heart failure severity. One underappreciated factor, in Dr. Desai’s view, is that readmission rates are significantly higher in hospitals that are financially and clinically resource poor, as shown in a Harvard School of Public Health analysis of Medicare claims data for more than 900,000 heart failure discharges. These resource-poor hospitals provide care for underserved populations, and they are experiencing a disproportionate burden of the financial penalties imposed for early readmission.
“As we seek to improve care for patients with heart failure, we should ensure that penalties for poor performance do not worsen disparities in quality of care,” according to the Harvard investigators (Circ. Cardiovasc. Qual. Outcomes 2011;4:53-9).
Dr. Desai reported serving as a consultant to 5AM Ventures, AtCor Medical, Novartis, and St. Jude Medical.
EXPERT ANALYSIS FROM THE CARDIOVASCULAR CONFERENCE AT SNOWMASS
Paroxetine improves cardiac function in mice after myocardial infarction
After noticing that paroxetine, a selective serotonin reuptake inhibitor normally used to treat depression, stopped the progression of heart failure in mice, researchers have concluded that the drug could be reclassified for use in humans, investigators wrote in Science Translational Medicine.
According to researchers from Temple University School of Medicine, genetically modified mice with heart failure (HF) were given paroxetine for 4 weeks. The drug blocked GRK2 (a protein normally elevated in people with heart failure), improved the left ventricle’s capacity to pump blood, and protected the heart muscle from scar formation and fibrosis within the first 2 weeks of treatment.
The researchers concluded, “These data present compelling evidence of a causal role for GRK2 in the maladaptive progression of cardiac remodeling and dysfunction leading to HF, especially after ischemic injury. Therefore, the development of small-molecule inhibitors of GRK2 appears warranted for pharmacologic treatment of HF.”
Read more in Science Translational Medicine.
After noticing that paroxetine, a selective serotonin reuptake inhibitor normally used to treat depression, stopped the progression of heart failure in mice, researchers have concluded that the drug could be reclassified for use in humans, investigators wrote in Science Translational Medicine.
According to researchers from Temple University School of Medicine, genetically modified mice with heart failure (HF) were given paroxetine for 4 weeks. The drug blocked GRK2 (a protein normally elevated in people with heart failure), improved the left ventricle’s capacity to pump blood, and protected the heart muscle from scar formation and fibrosis within the first 2 weeks of treatment.
The researchers concluded, “These data present compelling evidence of a causal role for GRK2 in the maladaptive progression of cardiac remodeling and dysfunction leading to HF, especially after ischemic injury. Therefore, the development of small-molecule inhibitors of GRK2 appears warranted for pharmacologic treatment of HF.”
Read more in Science Translational Medicine.
After noticing that paroxetine, a selective serotonin reuptake inhibitor normally used to treat depression, stopped the progression of heart failure in mice, researchers have concluded that the drug could be reclassified for use in humans, investigators wrote in Science Translational Medicine.
According to researchers from Temple University School of Medicine, genetically modified mice with heart failure (HF) were given paroxetine for 4 weeks. The drug blocked GRK2 (a protein normally elevated in people with heart failure), improved the left ventricle’s capacity to pump blood, and protected the heart muscle from scar formation and fibrosis within the first 2 weeks of treatment.
The researchers concluded, “These data present compelling evidence of a causal role for GRK2 in the maladaptive progression of cardiac remodeling and dysfunction leading to HF, especially after ischemic injury. Therefore, the development of small-molecule inhibitors of GRK2 appears warranted for pharmacologic treatment of HF.”
Read more in Science Translational Medicine.
Genetic testing enters cardiovascular-disease mainstream
Genetic testing is now part of routine practice in at least some U.S. centers for managing a short list of cardiovascular disease scenarios.
Although it remains well shy of ubiquitous for cardiovascular practice, for at least four important settings genetic testing has begun to play a key role: diagnosis of inherited cardiac conditions such as cardiomyopathies and channelopathies, diagnosis of homozygous and heterozygous familial hypercholesterolemia, treatment of stented coronary-disease patients with clopidogrel, and use when starting patients on warfarin.
Cardiomyopathies and channelopathies
Genetic testing of patients with any of the several inherited diseases that fall into these categories, including hypertrophic cardiomyopathy, dilated cardiomyopathy, long QT syndrome, and Brugada, received official recommendation from the Heart Rhythm Society and European Heart Rhythm Society in 2011 (Europace 2011;813:1077-109), and genetic testing for patients with hypertrophic cardiomyopathy and their first-degree relatives received a class I level B rating in guidelines for this specific cardiomyopathy released last year from the European Society of Cardiology (Eur. Heart J. 2014;35:2733-79).
Those endorsements put these cardiac diseases on the firmest ground for categorization as routine practice, a status that grew firmer still when researchers announced in January 2015 new evidence that better clarified the genetic underpinnings of dilated cardiomyopathy, the most common inherited cardiac condition.
“Genetic testing clearly contributes to risk assessment” of cardiomyopathies and channelopathies, Dr. Ray E. Hershberger said during a talk last November at the American Heart Association scientific sessions in Chicago. “Finding a plausible genetic cause for a condition lays the foundation for more durable intervention,” said Dr. Hershberger, professor and director of human genetics at Ohio State University in Columbus.
“Genetic testing for dilated cardiomyopathy is becoming increasingly important in clinical practice. Genetic testing is available, affordable, and efficient,” Dr. Luisa Mestroni, director of the Adult Medical Genetics Program of the University of Colorado, Denver, said during a separate talk at the AHA meeting. She cited three commercially available genetic tests for dilated cardiomyopathy that use next-generation sequencing and screen dozens of implicated genes. The genetic findings are available within about 10 weeks and cost in the range of several thousand dollars. Similar panels also exist for the other cardiomyopathies and channelopathies. Perhaps the most significant limitation on genetic testing for these patients is that it be done within the context of genetic counseling, said both Dr. Mestroni and Dr. Hershberger.
Genetic testing for dilated cardiomyopathy has been hindered by the disorder’s genetic heterogeneity, but understanding of the genetic picture grew much clearer in January with publication of genetic findings from 5,267 people representing the full spectrum of cardiac status, including fully-assessed healthy controls; 3,603 unselected community dwellers taken from the Framingham and Jackson Heart Studies; 374 unselected, ambulatory patients with dilated cardiomyopathy; and 155 patients with severe, end-stage dilated cardiomyopathy. The large, multicenter team of mostly British and U.S. researchers validated the findings in 163 additional patients with familial dilated cardiomyopathy (Sci. Transl. Med. 2015;7:[doi: 10.1126/scitranslmed.3010134].
The findings showed that mutations that produce truncated forms of titin, the body’s largest protein and a critical part of cardiac muscle, appear in about 2% of the general population, about 13% of ambulatory patients with dilated cardiomyopathy, and about 20% of patients with end-stage DCM. In addition, the truncations that occur in people who lack clinical disease tended to affect minor titin isoforms and produce mild functional changes, while the titin truncation mutations in people with severe disease were in the most commonly used isoforms and at sites that severely impaired function.
Until now, in the United Kingdom, “it would not have been standard of care to do gene testing [for patients with suspected dilated cardiomyopathy] unless there was a clear family history or some phenotypic indicator, but going forward we believe the yield from titin truncations [as well as from other, less common known mutations] is sufficient to include genetic testing in a patient’s work-up,” Dr. James S. Ware, a coauthor of the new report and a cardiologist at Imperial College, London, said during a press conference in January.
“The American College of Cardiology and American Heart Association guidelines for managing cardiomyopathy say that it is critical to perform longitudinal clinical follow-up on all first-degree relatives of patients with cardiomyopathy. The cost for longitudinal clinical screening is huge and also poses inconvenience for the family members screened. Genetic testing can now identify which family members really need clinical follow-up and which ones don’t need it,” said Dr. Cristine E. Seidman, a coauthor of the new titin-genetics report, professor of genetics and medicine at Harvard Medical School, and director of the Cardiovascular Genetics Center at Brigham and Women’s Hospital, Boston.
Familial hypercholesterolemia
Routine screening for familial hypercholesterolemia (FH) is not as widely endorsed, but it does have backers, and evidence shows that the approach is cost effective. In part, that’s because both heterozygous and homozygous FH are substantially more common than had been believed until recently. Recent study results documented prevalence rates roughly threefold more common than previously calculated.
For example, a study published last year of more than 104,000 residents of the Netherlands documented a homozygous FH prevalence of 1 in 300,000 (Eur. Heart J. 2014 [doi.org/10.1093/eurheartj/ehu058]). Results from a 2012 study of more than 69,000 Danish residents demonstrated a prevalence of 1 in 137 for people with heterozygous FH (J. Clin. Endocrinol. Metab. 2012;97:3956-64).
“Genetic testing improves the care of individual patients with FH because with genetic testing you can catch patients with FH presymptomatically and you can then treat them and change their long-term prognosis,” Dr. Anne Tybjaerg-Hansen, professor of clinical biochemistry and chief physician at Copenhagen University Hospital, said in a talk at the AHA meeting last November. She cited a report last year from Australia on a model demonstrating the cost effectiveness of testing first-degree relatives of index patients diagnosed with FH, an approach known as cascade screening (J. Clin. Lipidology 2014;8:390-400).
Last year, a consensus panel of the European Atherosclerosis Society recommended that genetic testing “should be considered” for both the diagnostic work-up of index cases with suspected FH and for cascade screening of first-degree relatives of confirmed cases (Eur. Heart J. 2014;35:2146-57). The European panel noted that genetic testing was potentially useful not only to confirm a clinical diagnosis but also to better distinguish patients with heterozygous and homozygous FH. The panel’s report last year endorsed the updated understanding that heterozygous FH occurs in roughly 1 in every 200 people, while homozygous FH has a prevalence of 1 case in every 160,000-300,000 people.
Despite these relatively high prevalence rates, FH is woefully underdiagnosed. A 2013 report from the European Atherosclerosis Society cited recent data documenting that less than 1% of U.S. residents with FH are currently identified (Eur. Heart J. 2013;34:3478-90). The same report noted that less than half of all FH patients have been identified in every country worldwide with reported statistics – aside from the Netherlands, which topped the world with an estimated identification rate of 71% of patients with either form of FH.
Clopidogrel responsiveness
It’s now been several years since researchers determined that roughly 30% of people fail to adequately metabolize the antiplatelet drug clopidogrel into its active form, a finding that in 2010 led the Food and Drug Administration to mandate a boxed warning on clopidogrel’s labeling. The cytochrome P450 (CYP) 2C19 gene codes for the enzyme that activates clopidogrel, and the warning states that people in this significant minority carry alleles of the CYP2C19 gene that make them poor clopidogrel metabolizers and hence clinicians should consider using “alternative treatment strategies.” Usually this means treatment with prasugrel (Effient) or ticagrelor (Brilinta), two thienopyridines that match clopidogrel’s efficacy but do not require metabolic conversion and so are effective even in poor-metabolizing patients.
But years after this well documented and well publicized problem with blind clopidogrel dosing first became apparent, many clinicians remain uncertain how to deal with the issue and how to use genetic testing to clarify the risk faced by individual patients (Circulation 2012;122:445-8).
“The evidence for CYP2C19 is really strong, but because prospective, randomized clinical trials of genotype-directed antiplatelet testing have not been performed, there is a lot of resistance to adoption of genotype-directed treatment,” Dr. Alan R. Shuldiner said in a talk at the November AHA meeting. An editorial published in late 2011 called the boxed warning on clopidogrel “irrational exuberance” (JAMA 2011;306:2727-8). Dr. Shuldiner and others contend that overall evidence today supports a need to screen the CYP2C19 gene before starting clopidogrel, although he conceded that the optimal clinical algorithm for using testing has not yet been devised and that paying for screening remains problematic.
Despite these limitations, Dr. Shuldiner and several colleagues from around the United States issued recommendations in 2013 for the implementation of CYP2C19 genetic testing in patients (Clin. Pharmacol. Ther. 2013;94:317-23) on behalf of the Clinical Pharmacogenetics Implementation Consortium. And in February 2013, clinicians at the University of Maryland in Baltimore began to implement those recommendations as one of eight U.S. sites participating in the PGRN Translational Pharmacogenetics Program.
“It took us 18 months to figure out how to implement pharmacogenetics in the cath lab,” said Dr. Shuldiner, former director of the program for personalized and genomic medicine at the University of Maryland, and now vice president of translation genomics at Regeneron in Tarrytown, N.Y.
From February 2013 through August 2014, the University of Maryland program screened on a routine basis 557 patients undergoing percutaneous coronary intervention (PCI) – with a 5-hour turnaround time on the results – enrolled 446 into the program, found 67 patients with an “actionable” genotype, and actually prescribed an alternative therapy because of the genotype in 37 patients. Patients usually did not receive the genomic-guided treatment because of a contraindication, Dr. Shuldiner said. He concluded that the Maryland experience shows routine screening is doable, and with its strong evidence base warrants use in all catheterization labs.
PCI patients at the University of Florida in Gainesville also now routinely undergo genomic assessment,said during a separate session at the AHA meeting. “Physicians recently began starting their acute coronary syndrome (ACS) patients on ticagrelor, and then when they get the genotype if the patient has an allele associated with good metabolism and can’t afford ticagrelor and needs to change to clopidogrel we know it will be okay. That’s the way we deal with the delay” in getting the genomic results. “At least you know that the patient is protected [with ticagrelor] during the really hot period,” said Dr. Cavallari, director of the center for pharmacogenomics of the University of Florida.
“For ACS patients if you treat with prasugrel or ticagrelor then you’ve pretty much overcome the roadblock, but for patients on clopidogrel I think it is very reasonable to use genetic testing or platelet-reactivity testing, especially for patients with a stented left main coronary,” Dr. Jessica L. Mega said during a panel discussion with Dr. Cavallari at the meeting. “We have pretty good data to suggest that at least in the peri-PCI patients who are homozygous for the *2 allele [the worst clopidogrel metabolizers] should not be on a standard clopidogrel dosage.”
Dr. Mega agreed with other genetic-testing proponents who see a double standard surrounding these tests. “I think there is a little genetic exceptionalism going on, and people feel differently about using genomics compared with other biomarkers. We have good data, and we just need to act on it,” said Dr. Mega, a cardiologist at Brigham and Women’s Hospital in Boston. “If you had an ACS event and received a stent in your left anterior descending coronary and were known to have a *2 allele or had high on-treatment platelet activity would you want to go home on 75 mg a day of clopidogrel?” Dr. Mega asked during a talk at the meeting.
Warfarin dosing
Evidence has implicated polymorphisms in two genes, CYP2C9 and VKORC1, as playing key roles in warfarin metabolism and suggested that determination of a patient’s allele profile for these two genes could assist in more quickly finding the best warfarin dose for a patients requiring oral antithrombotic therapy to reach their target International Normalized Ratio (INR).
In late 2013, researchers reported results from two major prospective trials designed to test the efficacy of genetic analysis of these two genes. The results were split. The European Pharmacogenetics of Anticoagulant Therapy (EU-PACT) trial with 455 patients in the United Kingdom and Sweden showed that genotype-based dosing at the start of warfarin therapy increased the time in the therapeutic range, the primary outcome, by 7 percentage points and reduced the incidence of excessive anticoagulation, the time required to reach a therapeutic INR, the time required to reach a stable dose, and the number of adjustments in the dose of warfarin (N. Engl. J. Med. 2013;369:2294-303). But results from the Clarification of Optimal Anticoagulation through Genetics (COAG) trial, which included 1,015 patients at 18 U.S. centers, showed that after 4 weeks genotyping-based warfarin dosing produced no significant between-group difference in the mean percentage of time in the therapeutic range (N. Engl. J. Med. 2013;369:2283-93).These inconsistent results, and especially the COAG study’s inability to show a benefit from genetic analysis in improving warfarin-dose selection, “called into question the clinical utility” of genetic testing, Dr. Cavallari wrote in a comment she coauthored last July (Clin. Pharmacol. Ther. 2014;96:22-4).
But recent data from Dr. Cavallari’s research group suggests that the value of genetic testing in patients starting warfarin may have been masked by the high proportion of African Americans in the COAG study, 27% of enrolled patients, and the finding that these patients did poorly because the alleles they carry most frequently were not covered by the genetic-test panel used in the study.
Before she moved to Gainesville, Dr. Cavallari worked at the University of Illinois in Chicago and she and her associates there began in August 2012 to routinely genotype patients prior to the start of warfarin treatment, using the information to guide initial dosing levels. The analyses they performed included several alleles commonly seen in African Americans – roughly two-thirds of the 389 patients they screened under this program were African American.
Expanding the testing panel had a major impact on efficacy. Results reported by her group at the AHA meeting last November showed that immediately following the start of warfarin treatment the percentage of INR levels that fell out of the therapeutic range was 42%, a statistically significant improvement over the 65% rate seen in 308 historic controls drawn from the year immediately preceding the start of routine genotyping (Circulation 2014;130A:16119). The results showed that the average number of days for patients to reach their first INR measurement in the therapeutic range fell from 11 days prior to genotyping to 4 days, and the average number of days needed on treatment with low-molecular-weight heparin, a bridging strategy until a therapeutic INR is achieved, fell from 10 days among the historic controls to an average of 2 days. The between-group differences for all three of these metrics reached statistical significance in analyses that adjusted for multiple variables, and that used propensity scoring, Dr. Cavallari said.But although genotyping prior to starting warfarin “is available and reasonable to use, until Medicare starts paying for it, doing this will be hard to implement,” Dr. Cavallari conceded during a November talk. Although warfarin-oriented genetic testing may be performed routinely at the University of Illinois, a lot of clinicians elsewhere are holding back, in part because of reimbursement issues and in part because “they want to see proof of better patient outcomes,” said Pharm.D., a cardiovascular pharmacology researcher at the University of Connecticut in Storrs. Like many who look at the evidence for routine genetic testing in cardiovcascular medicine, they “want to see differences in hard clinical outcomes before incorporating it into their practices,” Dr. Baker said.
Dr. Hershberger, Dr. Mestroni, Dr. Ware, Dr. Seidman, Dr. Tybjaerg-Hansen, and Dr. Cavallari had no disclosures. Dr. Shuldiner is an employee of Regeneron. Dr. Mega has been a consultant to Janssen, Boehinger Ingelheim, American Genomics, Bayer, and Portola, and has received research grants from eight companies. Dr. Baker received a research grant from Gilead.
On Twitter @mitchelzoler
Genetic testing is now part of routine practice in at least some U.S. centers for managing a short list of cardiovascular disease scenarios.
Although it remains well shy of ubiquitous for cardiovascular practice, for at least four important settings genetic testing has begun to play a key role: diagnosis of inherited cardiac conditions such as cardiomyopathies and channelopathies, diagnosis of homozygous and heterozygous familial hypercholesterolemia, treatment of stented coronary-disease patients with clopidogrel, and use when starting patients on warfarin.
Cardiomyopathies and channelopathies
Genetic testing of patients with any of the several inherited diseases that fall into these categories, including hypertrophic cardiomyopathy, dilated cardiomyopathy, long QT syndrome, and Brugada, received official recommendation from the Heart Rhythm Society and European Heart Rhythm Society in 2011 (Europace 2011;813:1077-109), and genetic testing for patients with hypertrophic cardiomyopathy and their first-degree relatives received a class I level B rating in guidelines for this specific cardiomyopathy released last year from the European Society of Cardiology (Eur. Heart J. 2014;35:2733-79).
Those endorsements put these cardiac diseases on the firmest ground for categorization as routine practice, a status that grew firmer still when researchers announced in January 2015 new evidence that better clarified the genetic underpinnings of dilated cardiomyopathy, the most common inherited cardiac condition.
“Genetic testing clearly contributes to risk assessment” of cardiomyopathies and channelopathies, Dr. Ray E. Hershberger said during a talk last November at the American Heart Association scientific sessions in Chicago. “Finding a plausible genetic cause for a condition lays the foundation for more durable intervention,” said Dr. Hershberger, professor and director of human genetics at Ohio State University in Columbus.
“Genetic testing for dilated cardiomyopathy is becoming increasingly important in clinical practice. Genetic testing is available, affordable, and efficient,” Dr. Luisa Mestroni, director of the Adult Medical Genetics Program of the University of Colorado, Denver, said during a separate talk at the AHA meeting. She cited three commercially available genetic tests for dilated cardiomyopathy that use next-generation sequencing and screen dozens of implicated genes. The genetic findings are available within about 10 weeks and cost in the range of several thousand dollars. Similar panels also exist for the other cardiomyopathies and channelopathies. Perhaps the most significant limitation on genetic testing for these patients is that it be done within the context of genetic counseling, said both Dr. Mestroni and Dr. Hershberger.
Genetic testing for dilated cardiomyopathy has been hindered by the disorder’s genetic heterogeneity, but understanding of the genetic picture grew much clearer in January with publication of genetic findings from 5,267 people representing the full spectrum of cardiac status, including fully-assessed healthy controls; 3,603 unselected community dwellers taken from the Framingham and Jackson Heart Studies; 374 unselected, ambulatory patients with dilated cardiomyopathy; and 155 patients with severe, end-stage dilated cardiomyopathy. The large, multicenter team of mostly British and U.S. researchers validated the findings in 163 additional patients with familial dilated cardiomyopathy (Sci. Transl. Med. 2015;7:[doi: 10.1126/scitranslmed.3010134].
The findings showed that mutations that produce truncated forms of titin, the body’s largest protein and a critical part of cardiac muscle, appear in about 2% of the general population, about 13% of ambulatory patients with dilated cardiomyopathy, and about 20% of patients with end-stage DCM. In addition, the truncations that occur in people who lack clinical disease tended to affect minor titin isoforms and produce mild functional changes, while the titin truncation mutations in people with severe disease were in the most commonly used isoforms and at sites that severely impaired function.
Until now, in the United Kingdom, “it would not have been standard of care to do gene testing [for patients with suspected dilated cardiomyopathy] unless there was a clear family history or some phenotypic indicator, but going forward we believe the yield from titin truncations [as well as from other, less common known mutations] is sufficient to include genetic testing in a patient’s work-up,” Dr. James S. Ware, a coauthor of the new report and a cardiologist at Imperial College, London, said during a press conference in January.
“The American College of Cardiology and American Heart Association guidelines for managing cardiomyopathy say that it is critical to perform longitudinal clinical follow-up on all first-degree relatives of patients with cardiomyopathy. The cost for longitudinal clinical screening is huge and also poses inconvenience for the family members screened. Genetic testing can now identify which family members really need clinical follow-up and which ones don’t need it,” said Dr. Cristine E. Seidman, a coauthor of the new titin-genetics report, professor of genetics and medicine at Harvard Medical School, and director of the Cardiovascular Genetics Center at Brigham and Women’s Hospital, Boston.
Familial hypercholesterolemia
Routine screening for familial hypercholesterolemia (FH) is not as widely endorsed, but it does have backers, and evidence shows that the approach is cost effective. In part, that’s because both heterozygous and homozygous FH are substantially more common than had been believed until recently. Recent study results documented prevalence rates roughly threefold more common than previously calculated.
For example, a study published last year of more than 104,000 residents of the Netherlands documented a homozygous FH prevalence of 1 in 300,000 (Eur. Heart J. 2014 [doi.org/10.1093/eurheartj/ehu058]). Results from a 2012 study of more than 69,000 Danish residents demonstrated a prevalence of 1 in 137 for people with heterozygous FH (J. Clin. Endocrinol. Metab. 2012;97:3956-64).
“Genetic testing improves the care of individual patients with FH because with genetic testing you can catch patients with FH presymptomatically and you can then treat them and change their long-term prognosis,” Dr. Anne Tybjaerg-Hansen, professor of clinical biochemistry and chief physician at Copenhagen University Hospital, said in a talk at the AHA meeting last November. She cited a report last year from Australia on a model demonstrating the cost effectiveness of testing first-degree relatives of index patients diagnosed with FH, an approach known as cascade screening (J. Clin. Lipidology 2014;8:390-400).
Last year, a consensus panel of the European Atherosclerosis Society recommended that genetic testing “should be considered” for both the diagnostic work-up of index cases with suspected FH and for cascade screening of first-degree relatives of confirmed cases (Eur. Heart J. 2014;35:2146-57). The European panel noted that genetic testing was potentially useful not only to confirm a clinical diagnosis but also to better distinguish patients with heterozygous and homozygous FH. The panel’s report last year endorsed the updated understanding that heterozygous FH occurs in roughly 1 in every 200 people, while homozygous FH has a prevalence of 1 case in every 160,000-300,000 people.
Despite these relatively high prevalence rates, FH is woefully underdiagnosed. A 2013 report from the European Atherosclerosis Society cited recent data documenting that less than 1% of U.S. residents with FH are currently identified (Eur. Heart J. 2013;34:3478-90). The same report noted that less than half of all FH patients have been identified in every country worldwide with reported statistics – aside from the Netherlands, which topped the world with an estimated identification rate of 71% of patients with either form of FH.
Clopidogrel responsiveness
It’s now been several years since researchers determined that roughly 30% of people fail to adequately metabolize the antiplatelet drug clopidogrel into its active form, a finding that in 2010 led the Food and Drug Administration to mandate a boxed warning on clopidogrel’s labeling. The cytochrome P450 (CYP) 2C19 gene codes for the enzyme that activates clopidogrel, and the warning states that people in this significant minority carry alleles of the CYP2C19 gene that make them poor clopidogrel metabolizers and hence clinicians should consider using “alternative treatment strategies.” Usually this means treatment with prasugrel (Effient) or ticagrelor (Brilinta), two thienopyridines that match clopidogrel’s efficacy but do not require metabolic conversion and so are effective even in poor-metabolizing patients.
But years after this well documented and well publicized problem with blind clopidogrel dosing first became apparent, many clinicians remain uncertain how to deal with the issue and how to use genetic testing to clarify the risk faced by individual patients (Circulation 2012;122:445-8).
“The evidence for CYP2C19 is really strong, but because prospective, randomized clinical trials of genotype-directed antiplatelet testing have not been performed, there is a lot of resistance to adoption of genotype-directed treatment,” Dr. Alan R. Shuldiner said in a talk at the November AHA meeting. An editorial published in late 2011 called the boxed warning on clopidogrel “irrational exuberance” (JAMA 2011;306:2727-8). Dr. Shuldiner and others contend that overall evidence today supports a need to screen the CYP2C19 gene before starting clopidogrel, although he conceded that the optimal clinical algorithm for using testing has not yet been devised and that paying for screening remains problematic.
Despite these limitations, Dr. Shuldiner and several colleagues from around the United States issued recommendations in 2013 for the implementation of CYP2C19 genetic testing in patients (Clin. Pharmacol. Ther. 2013;94:317-23) on behalf of the Clinical Pharmacogenetics Implementation Consortium. And in February 2013, clinicians at the University of Maryland in Baltimore began to implement those recommendations as one of eight U.S. sites participating in the PGRN Translational Pharmacogenetics Program.
“It took us 18 months to figure out how to implement pharmacogenetics in the cath lab,” said Dr. Shuldiner, former director of the program for personalized and genomic medicine at the University of Maryland, and now vice president of translation genomics at Regeneron in Tarrytown, N.Y.
From February 2013 through August 2014, the University of Maryland program screened on a routine basis 557 patients undergoing percutaneous coronary intervention (PCI) – with a 5-hour turnaround time on the results – enrolled 446 into the program, found 67 patients with an “actionable” genotype, and actually prescribed an alternative therapy because of the genotype in 37 patients. Patients usually did not receive the genomic-guided treatment because of a contraindication, Dr. Shuldiner said. He concluded that the Maryland experience shows routine screening is doable, and with its strong evidence base warrants use in all catheterization labs.
PCI patients at the University of Florida in Gainesville also now routinely undergo genomic assessment,said during a separate session at the AHA meeting. “Physicians recently began starting their acute coronary syndrome (ACS) patients on ticagrelor, and then when they get the genotype if the patient has an allele associated with good metabolism and can’t afford ticagrelor and needs to change to clopidogrel we know it will be okay. That’s the way we deal with the delay” in getting the genomic results. “At least you know that the patient is protected [with ticagrelor] during the really hot period,” said Dr. Cavallari, director of the center for pharmacogenomics of the University of Florida.
“For ACS patients if you treat with prasugrel or ticagrelor then you’ve pretty much overcome the roadblock, but for patients on clopidogrel I think it is very reasonable to use genetic testing or platelet-reactivity testing, especially for patients with a stented left main coronary,” Dr. Jessica L. Mega said during a panel discussion with Dr. Cavallari at the meeting. “We have pretty good data to suggest that at least in the peri-PCI patients who are homozygous for the *2 allele [the worst clopidogrel metabolizers] should not be on a standard clopidogrel dosage.”
Dr. Mega agreed with other genetic-testing proponents who see a double standard surrounding these tests. “I think there is a little genetic exceptionalism going on, and people feel differently about using genomics compared with other biomarkers. We have good data, and we just need to act on it,” said Dr. Mega, a cardiologist at Brigham and Women’s Hospital in Boston. “If you had an ACS event and received a stent in your left anterior descending coronary and were known to have a *2 allele or had high on-treatment platelet activity would you want to go home on 75 mg a day of clopidogrel?” Dr. Mega asked during a talk at the meeting.
Warfarin dosing
Evidence has implicated polymorphisms in two genes, CYP2C9 and VKORC1, as playing key roles in warfarin metabolism and suggested that determination of a patient’s allele profile for these two genes could assist in more quickly finding the best warfarin dose for a patients requiring oral antithrombotic therapy to reach their target International Normalized Ratio (INR).
In late 2013, researchers reported results from two major prospective trials designed to test the efficacy of genetic analysis of these two genes. The results were split. The European Pharmacogenetics of Anticoagulant Therapy (EU-PACT) trial with 455 patients in the United Kingdom and Sweden showed that genotype-based dosing at the start of warfarin therapy increased the time in the therapeutic range, the primary outcome, by 7 percentage points and reduced the incidence of excessive anticoagulation, the time required to reach a therapeutic INR, the time required to reach a stable dose, and the number of adjustments in the dose of warfarin (N. Engl. J. Med. 2013;369:2294-303). But results from the Clarification of Optimal Anticoagulation through Genetics (COAG) trial, which included 1,015 patients at 18 U.S. centers, showed that after 4 weeks genotyping-based warfarin dosing produced no significant between-group difference in the mean percentage of time in the therapeutic range (N. Engl. J. Med. 2013;369:2283-93).These inconsistent results, and especially the COAG study’s inability to show a benefit from genetic analysis in improving warfarin-dose selection, “called into question the clinical utility” of genetic testing, Dr. Cavallari wrote in a comment she coauthored last July (Clin. Pharmacol. Ther. 2014;96:22-4).
But recent data from Dr. Cavallari’s research group suggests that the value of genetic testing in patients starting warfarin may have been masked by the high proportion of African Americans in the COAG study, 27% of enrolled patients, and the finding that these patients did poorly because the alleles they carry most frequently were not covered by the genetic-test panel used in the study.
Before she moved to Gainesville, Dr. Cavallari worked at the University of Illinois in Chicago and she and her associates there began in August 2012 to routinely genotype patients prior to the start of warfarin treatment, using the information to guide initial dosing levels. The analyses they performed included several alleles commonly seen in African Americans – roughly two-thirds of the 389 patients they screened under this program were African American.
Expanding the testing panel had a major impact on efficacy. Results reported by her group at the AHA meeting last November showed that immediately following the start of warfarin treatment the percentage of INR levels that fell out of the therapeutic range was 42%, a statistically significant improvement over the 65% rate seen in 308 historic controls drawn from the year immediately preceding the start of routine genotyping (Circulation 2014;130A:16119). The results showed that the average number of days for patients to reach their first INR measurement in the therapeutic range fell from 11 days prior to genotyping to 4 days, and the average number of days needed on treatment with low-molecular-weight heparin, a bridging strategy until a therapeutic INR is achieved, fell from 10 days among the historic controls to an average of 2 days. The between-group differences for all three of these metrics reached statistical significance in analyses that adjusted for multiple variables, and that used propensity scoring, Dr. Cavallari said.But although genotyping prior to starting warfarin “is available and reasonable to use, until Medicare starts paying for it, doing this will be hard to implement,” Dr. Cavallari conceded during a November talk. Although warfarin-oriented genetic testing may be performed routinely at the University of Illinois, a lot of clinicians elsewhere are holding back, in part because of reimbursement issues and in part because “they want to see proof of better patient outcomes,” said Pharm.D., a cardiovascular pharmacology researcher at the University of Connecticut in Storrs. Like many who look at the evidence for routine genetic testing in cardiovcascular medicine, they “want to see differences in hard clinical outcomes before incorporating it into their practices,” Dr. Baker said.
Dr. Hershberger, Dr. Mestroni, Dr. Ware, Dr. Seidman, Dr. Tybjaerg-Hansen, and Dr. Cavallari had no disclosures. Dr. Shuldiner is an employee of Regeneron. Dr. Mega has been a consultant to Janssen, Boehinger Ingelheim, American Genomics, Bayer, and Portola, and has received research grants from eight companies. Dr. Baker received a research grant from Gilead.
On Twitter @mitchelzoler
Genetic testing is now part of routine practice in at least some U.S. centers for managing a short list of cardiovascular disease scenarios.
Although it remains well shy of ubiquitous for cardiovascular practice, for at least four important settings genetic testing has begun to play a key role: diagnosis of inherited cardiac conditions such as cardiomyopathies and channelopathies, diagnosis of homozygous and heterozygous familial hypercholesterolemia, treatment of stented coronary-disease patients with clopidogrel, and use when starting patients on warfarin.
Cardiomyopathies and channelopathies
Genetic testing of patients with any of the several inherited diseases that fall into these categories, including hypertrophic cardiomyopathy, dilated cardiomyopathy, long QT syndrome, and Brugada, received official recommendation from the Heart Rhythm Society and European Heart Rhythm Society in 2011 (Europace 2011;813:1077-109), and genetic testing for patients with hypertrophic cardiomyopathy and their first-degree relatives received a class I level B rating in guidelines for this specific cardiomyopathy released last year from the European Society of Cardiology (Eur. Heart J. 2014;35:2733-79).
Those endorsements put these cardiac diseases on the firmest ground for categorization as routine practice, a status that grew firmer still when researchers announced in January 2015 new evidence that better clarified the genetic underpinnings of dilated cardiomyopathy, the most common inherited cardiac condition.
“Genetic testing clearly contributes to risk assessment” of cardiomyopathies and channelopathies, Dr. Ray E. Hershberger said during a talk last November at the American Heart Association scientific sessions in Chicago. “Finding a plausible genetic cause for a condition lays the foundation for more durable intervention,” said Dr. Hershberger, professor and director of human genetics at Ohio State University in Columbus.
“Genetic testing for dilated cardiomyopathy is becoming increasingly important in clinical practice. Genetic testing is available, affordable, and efficient,” Dr. Luisa Mestroni, director of the Adult Medical Genetics Program of the University of Colorado, Denver, said during a separate talk at the AHA meeting. She cited three commercially available genetic tests for dilated cardiomyopathy that use next-generation sequencing and screen dozens of implicated genes. The genetic findings are available within about 10 weeks and cost in the range of several thousand dollars. Similar panels also exist for the other cardiomyopathies and channelopathies. Perhaps the most significant limitation on genetic testing for these patients is that it be done within the context of genetic counseling, said both Dr. Mestroni and Dr. Hershberger.
Genetic testing for dilated cardiomyopathy has been hindered by the disorder’s genetic heterogeneity, but understanding of the genetic picture grew much clearer in January with publication of genetic findings from 5,267 people representing the full spectrum of cardiac status, including fully-assessed healthy controls; 3,603 unselected community dwellers taken from the Framingham and Jackson Heart Studies; 374 unselected, ambulatory patients with dilated cardiomyopathy; and 155 patients with severe, end-stage dilated cardiomyopathy. The large, multicenter team of mostly British and U.S. researchers validated the findings in 163 additional patients with familial dilated cardiomyopathy (Sci. Transl. Med. 2015;7:[doi: 10.1126/scitranslmed.3010134].
The findings showed that mutations that produce truncated forms of titin, the body’s largest protein and a critical part of cardiac muscle, appear in about 2% of the general population, about 13% of ambulatory patients with dilated cardiomyopathy, and about 20% of patients with end-stage DCM. In addition, the truncations that occur in people who lack clinical disease tended to affect minor titin isoforms and produce mild functional changes, while the titin truncation mutations in people with severe disease were in the most commonly used isoforms and at sites that severely impaired function.
Until now, in the United Kingdom, “it would not have been standard of care to do gene testing [for patients with suspected dilated cardiomyopathy] unless there was a clear family history or some phenotypic indicator, but going forward we believe the yield from titin truncations [as well as from other, less common known mutations] is sufficient to include genetic testing in a patient’s work-up,” Dr. James S. Ware, a coauthor of the new report and a cardiologist at Imperial College, London, said during a press conference in January.
“The American College of Cardiology and American Heart Association guidelines for managing cardiomyopathy say that it is critical to perform longitudinal clinical follow-up on all first-degree relatives of patients with cardiomyopathy. The cost for longitudinal clinical screening is huge and also poses inconvenience for the family members screened. Genetic testing can now identify which family members really need clinical follow-up and which ones don’t need it,” said Dr. Cristine E. Seidman, a coauthor of the new titin-genetics report, professor of genetics and medicine at Harvard Medical School, and director of the Cardiovascular Genetics Center at Brigham and Women’s Hospital, Boston.
Familial hypercholesterolemia
Routine screening for familial hypercholesterolemia (FH) is not as widely endorsed, but it does have backers, and evidence shows that the approach is cost effective. In part, that’s because both heterozygous and homozygous FH are substantially more common than had been believed until recently. Recent study results documented prevalence rates roughly threefold more common than previously calculated.
For example, a study published last year of more than 104,000 residents of the Netherlands documented a homozygous FH prevalence of 1 in 300,000 (Eur. Heart J. 2014 [doi.org/10.1093/eurheartj/ehu058]). Results from a 2012 study of more than 69,000 Danish residents demonstrated a prevalence of 1 in 137 for people with heterozygous FH (J. Clin. Endocrinol. Metab. 2012;97:3956-64).
“Genetic testing improves the care of individual patients with FH because with genetic testing you can catch patients with FH presymptomatically and you can then treat them and change their long-term prognosis,” Dr. Anne Tybjaerg-Hansen, professor of clinical biochemistry and chief physician at Copenhagen University Hospital, said in a talk at the AHA meeting last November. She cited a report last year from Australia on a model demonstrating the cost effectiveness of testing first-degree relatives of index patients diagnosed with FH, an approach known as cascade screening (J. Clin. Lipidology 2014;8:390-400).
Last year, a consensus panel of the European Atherosclerosis Society recommended that genetic testing “should be considered” for both the diagnostic work-up of index cases with suspected FH and for cascade screening of first-degree relatives of confirmed cases (Eur. Heart J. 2014;35:2146-57). The European panel noted that genetic testing was potentially useful not only to confirm a clinical diagnosis but also to better distinguish patients with heterozygous and homozygous FH. The panel’s report last year endorsed the updated understanding that heterozygous FH occurs in roughly 1 in every 200 people, while homozygous FH has a prevalence of 1 case in every 160,000-300,000 people.
Despite these relatively high prevalence rates, FH is woefully underdiagnosed. A 2013 report from the European Atherosclerosis Society cited recent data documenting that less than 1% of U.S. residents with FH are currently identified (Eur. Heart J. 2013;34:3478-90). The same report noted that less than half of all FH patients have been identified in every country worldwide with reported statistics – aside from the Netherlands, which topped the world with an estimated identification rate of 71% of patients with either form of FH.
Clopidogrel responsiveness
It’s now been several years since researchers determined that roughly 30% of people fail to adequately metabolize the antiplatelet drug clopidogrel into its active form, a finding that in 2010 led the Food and Drug Administration to mandate a boxed warning on clopidogrel’s labeling. The cytochrome P450 (CYP) 2C19 gene codes for the enzyme that activates clopidogrel, and the warning states that people in this significant minority carry alleles of the CYP2C19 gene that make them poor clopidogrel metabolizers and hence clinicians should consider using “alternative treatment strategies.” Usually this means treatment with prasugrel (Effient) or ticagrelor (Brilinta), two thienopyridines that match clopidogrel’s efficacy but do not require metabolic conversion and so are effective even in poor-metabolizing patients.
But years after this well documented and well publicized problem with blind clopidogrel dosing first became apparent, many clinicians remain uncertain how to deal with the issue and how to use genetic testing to clarify the risk faced by individual patients (Circulation 2012;122:445-8).
“The evidence for CYP2C19 is really strong, but because prospective, randomized clinical trials of genotype-directed antiplatelet testing have not been performed, there is a lot of resistance to adoption of genotype-directed treatment,” Dr. Alan R. Shuldiner said in a talk at the November AHA meeting. An editorial published in late 2011 called the boxed warning on clopidogrel “irrational exuberance” (JAMA 2011;306:2727-8). Dr. Shuldiner and others contend that overall evidence today supports a need to screen the CYP2C19 gene before starting clopidogrel, although he conceded that the optimal clinical algorithm for using testing has not yet been devised and that paying for screening remains problematic.
Despite these limitations, Dr. Shuldiner and several colleagues from around the United States issued recommendations in 2013 for the implementation of CYP2C19 genetic testing in patients (Clin. Pharmacol. Ther. 2013;94:317-23) on behalf of the Clinical Pharmacogenetics Implementation Consortium. And in February 2013, clinicians at the University of Maryland in Baltimore began to implement those recommendations as one of eight U.S. sites participating in the PGRN Translational Pharmacogenetics Program.
“It took us 18 months to figure out how to implement pharmacogenetics in the cath lab,” said Dr. Shuldiner, former director of the program for personalized and genomic medicine at the University of Maryland, and now vice president of translation genomics at Regeneron in Tarrytown, N.Y.
From February 2013 through August 2014, the University of Maryland program screened on a routine basis 557 patients undergoing percutaneous coronary intervention (PCI) – with a 5-hour turnaround time on the results – enrolled 446 into the program, found 67 patients with an “actionable” genotype, and actually prescribed an alternative therapy because of the genotype in 37 patients. Patients usually did not receive the genomic-guided treatment because of a contraindication, Dr. Shuldiner said. He concluded that the Maryland experience shows routine screening is doable, and with its strong evidence base warrants use in all catheterization labs.
PCI patients at the University of Florida in Gainesville also now routinely undergo genomic assessment,said during a separate session at the AHA meeting. “Physicians recently began starting their acute coronary syndrome (ACS) patients on ticagrelor, and then when they get the genotype if the patient has an allele associated with good metabolism and can’t afford ticagrelor and needs to change to clopidogrel we know it will be okay. That’s the way we deal with the delay” in getting the genomic results. “At least you know that the patient is protected [with ticagrelor] during the really hot period,” said Dr. Cavallari, director of the center for pharmacogenomics of the University of Florida.
“For ACS patients if you treat with prasugrel or ticagrelor then you’ve pretty much overcome the roadblock, but for patients on clopidogrel I think it is very reasonable to use genetic testing or platelet-reactivity testing, especially for patients with a stented left main coronary,” Dr. Jessica L. Mega said during a panel discussion with Dr. Cavallari at the meeting. “We have pretty good data to suggest that at least in the peri-PCI patients who are homozygous for the *2 allele [the worst clopidogrel metabolizers] should not be on a standard clopidogrel dosage.”
Dr. Mega agreed with other genetic-testing proponents who see a double standard surrounding these tests. “I think there is a little genetic exceptionalism going on, and people feel differently about using genomics compared with other biomarkers. We have good data, and we just need to act on it,” said Dr. Mega, a cardiologist at Brigham and Women’s Hospital in Boston. “If you had an ACS event and received a stent in your left anterior descending coronary and were known to have a *2 allele or had high on-treatment platelet activity would you want to go home on 75 mg a day of clopidogrel?” Dr. Mega asked during a talk at the meeting.
Warfarin dosing
Evidence has implicated polymorphisms in two genes, CYP2C9 and VKORC1, as playing key roles in warfarin metabolism and suggested that determination of a patient’s allele profile for these two genes could assist in more quickly finding the best warfarin dose for a patients requiring oral antithrombotic therapy to reach their target International Normalized Ratio (INR).
In late 2013, researchers reported results from two major prospective trials designed to test the efficacy of genetic analysis of these two genes. The results were split. The European Pharmacogenetics of Anticoagulant Therapy (EU-PACT) trial with 455 patients in the United Kingdom and Sweden showed that genotype-based dosing at the start of warfarin therapy increased the time in the therapeutic range, the primary outcome, by 7 percentage points and reduced the incidence of excessive anticoagulation, the time required to reach a therapeutic INR, the time required to reach a stable dose, and the number of adjustments in the dose of warfarin (N. Engl. J. Med. 2013;369:2294-303). But results from the Clarification of Optimal Anticoagulation through Genetics (COAG) trial, which included 1,015 patients at 18 U.S. centers, showed that after 4 weeks genotyping-based warfarin dosing produced no significant between-group difference in the mean percentage of time in the therapeutic range (N. Engl. J. Med. 2013;369:2283-93).These inconsistent results, and especially the COAG study’s inability to show a benefit from genetic analysis in improving warfarin-dose selection, “called into question the clinical utility” of genetic testing, Dr. Cavallari wrote in a comment she coauthored last July (Clin. Pharmacol. Ther. 2014;96:22-4).
But recent data from Dr. Cavallari’s research group suggests that the value of genetic testing in patients starting warfarin may have been masked by the high proportion of African Americans in the COAG study, 27% of enrolled patients, and the finding that these patients did poorly because the alleles they carry most frequently were not covered by the genetic-test panel used in the study.
Before she moved to Gainesville, Dr. Cavallari worked at the University of Illinois in Chicago and she and her associates there began in August 2012 to routinely genotype patients prior to the start of warfarin treatment, using the information to guide initial dosing levels. The analyses they performed included several alleles commonly seen in African Americans – roughly two-thirds of the 389 patients they screened under this program were African American.
Expanding the testing panel had a major impact on efficacy. Results reported by her group at the AHA meeting last November showed that immediately following the start of warfarin treatment the percentage of INR levels that fell out of the therapeutic range was 42%, a statistically significant improvement over the 65% rate seen in 308 historic controls drawn from the year immediately preceding the start of routine genotyping (Circulation 2014;130A:16119). The results showed that the average number of days for patients to reach their first INR measurement in the therapeutic range fell from 11 days prior to genotyping to 4 days, and the average number of days needed on treatment with low-molecular-weight heparin, a bridging strategy until a therapeutic INR is achieved, fell from 10 days among the historic controls to an average of 2 days. The between-group differences for all three of these metrics reached statistical significance in analyses that adjusted for multiple variables, and that used propensity scoring, Dr. Cavallari said.But although genotyping prior to starting warfarin “is available and reasonable to use, until Medicare starts paying for it, doing this will be hard to implement,” Dr. Cavallari conceded during a November talk. Although warfarin-oriented genetic testing may be performed routinely at the University of Illinois, a lot of clinicians elsewhere are holding back, in part because of reimbursement issues and in part because “they want to see proof of better patient outcomes,” said Pharm.D., a cardiovascular pharmacology researcher at the University of Connecticut in Storrs. Like many who look at the evidence for routine genetic testing in cardiovcascular medicine, they “want to see differences in hard clinical outcomes before incorporating it into their practices,” Dr. Baker said.
Dr. Hershberger, Dr. Mestroni, Dr. Ware, Dr. Seidman, Dr. Tybjaerg-Hansen, and Dr. Cavallari had no disclosures. Dr. Shuldiner is an employee of Regeneron. Dr. Mega has been a consultant to Janssen, Boehinger Ingelheim, American Genomics, Bayer, and Portola, and has received research grants from eight companies. Dr. Baker received a research grant from Gilead.
On Twitter @mitchelzoler
Functional disability tied to hospitalization, mortality in heart failure
Heart failure patients who reported difficulty performing everyday tasks were more likely to be hospitalized and to die early, reported Dr. Shannon M. Dunlay and associates from the Mayo Clinic.
In a cohort study of 1,128 Minnesota adults aged 20 years or older, nearly 60% reported having difficulty with at least one daily activity such as eating, dressing, using the toilet, housekeeping, climbing stairs, bathing, walking, using transportation, and managing medications. Patients with severe or worsening difficulty at follow-up were at significantly increased risk for readmission (hazard ratio, 1.51) and death (HR, 2.10), the investigators reported.
These results “provide new insight into the burden of functional disability in patients with” heart failure, Dr. Dunlay and her associates wrote.
Read the full article at Circulation Heart Failure (2015 Feb. 25 [doi:10.1161/CIRCHEARTFAILURE.114.001542]).
Heart failure patients who reported difficulty performing everyday tasks were more likely to be hospitalized and to die early, reported Dr. Shannon M. Dunlay and associates from the Mayo Clinic.
In a cohort study of 1,128 Minnesota adults aged 20 years or older, nearly 60% reported having difficulty with at least one daily activity such as eating, dressing, using the toilet, housekeeping, climbing stairs, bathing, walking, using transportation, and managing medications. Patients with severe or worsening difficulty at follow-up were at significantly increased risk for readmission (hazard ratio, 1.51) and death (HR, 2.10), the investigators reported.
These results “provide new insight into the burden of functional disability in patients with” heart failure, Dr. Dunlay and her associates wrote.
Read the full article at Circulation Heart Failure (2015 Feb. 25 [doi:10.1161/CIRCHEARTFAILURE.114.001542]).
Heart failure patients who reported difficulty performing everyday tasks were more likely to be hospitalized and to die early, reported Dr. Shannon M. Dunlay and associates from the Mayo Clinic.
In a cohort study of 1,128 Minnesota adults aged 20 years or older, nearly 60% reported having difficulty with at least one daily activity such as eating, dressing, using the toilet, housekeeping, climbing stairs, bathing, walking, using transportation, and managing medications. Patients with severe or worsening difficulty at follow-up were at significantly increased risk for readmission (hazard ratio, 1.51) and death (HR, 2.10), the investigators reported.
These results “provide new insight into the burden of functional disability in patients with” heart failure, Dr. Dunlay and her associates wrote.
Read the full article at Circulation Heart Failure (2015 Feb. 25 [doi:10.1161/CIRCHEARTFAILURE.114.001542]).