User login
COMP recommends orphan designations for KTE-C19

The European Medicines Agency’s Committee for Orphan Medicinal Products (COMP) has adopted positive opinions recommending orphan designation for KTE-C19 to treat acute lymphoblastic leukemia, chronic lymphocytic leukemia/small lymphocytic lymphoma, and follicular lymphoma.
KTE-C19 is an investigational chimeric antigen receptor (CAR) T-cell therapy designed to target CD19, a protein expressed on the surface of B cells.
The CAR T-cell therapy already has orphan designation for the treatment of diffuse large B-cell lymphoma in the US and the European Union (EU).
KTE-C19 also has COMP positive opinions for orphan designation in the EU for primary mediastinal B-cell lymphoma and mantle cell lymphoma.
About orphan designation
The COMP adopts an opinion on the granting of orphan designation, and that opinion is submitted to the European Commission for endorsement.
In the EU, orphan designation is granted to therapies intended to treat a life-threatening or chronically debilitating condition that affects no more than 5 in 10,000 persons and where no satisfactory treatment is available.
Companies that obtain orphan designation for a drug benefit from a number of incentives, including protocol assistance, a type of scientific advice specific for designated orphan medicines, and 10 years of market exclusivity once the medicine is approved. Fee reductions are also available, depending on the status of the sponsor and the type of service required.
KTE-C19 research
Last year, researchers reported results with KTE-C19 in the Journal of Clinical Oncology. The study included 15 patients with advanced B-cell malignancies.
The patients received a conditioning regimen of cyclophosphamide and fludarabine, followed 1 day later by a single infusion of the CAR T-cell therapy. The researchers noted that the conditioning regimen is known to be active against B-cell malignancies and could have made a direct contribution to patient responses.
Thirteen patients were evaluable for response. Eight patients achieved a complete response (CR), and 4 had a partial response (PR).
Of the 7 patients with chemotherapy-refractory diffuse large B-cell lymphoma, 4 achieved a CR, 2 achieved a PR, and 1 had stable disease. Of the 4 patients with chronic lymphocytic leukemia, 3 had a CR, and 1 had a PR. Among the 2 patients with indolent lymphomas, 1 achieved a CR, and 1 had a PR.
KTE-C19 was associated with fever, low blood pressure, focal neurological deficits, and delirium. Toxicities largely occurred in the first 2 weeks after infusion.
All but 2 patients experienced grade 3/4 adverse events. Four patients had grade 3/4 hypotension.
All patients had elevations in serum interferon gamma and/or interleukin 6 around the time of peak toxicity, but most did not develop elevations in serum tumor necrosis factor.
Neurologic toxicities included confusion and obtundation, which have been reported in previous studies. However, 3 patients developed unexpected neurologic abnormalities.
KTE-C19 is currently under investigation in a phase 1/2 trial (ZUMA-1) of patients with refractory, aggressive non-Hodgkin lymphomas. Kite Pharma, Inc., the company developing KTE-C19, plans to present top-line phase 1 data at the 2015 ASH Annual Meeting. ![]()

The European Medicines Agency’s Committee for Orphan Medicinal Products (COMP) has adopted positive opinions recommending orphan designation for KTE-C19 to treat acute lymphoblastic leukemia, chronic lymphocytic leukemia/small lymphocytic lymphoma, and follicular lymphoma.
KTE-C19 is an investigational chimeric antigen receptor (CAR) T-cell therapy designed to target CD19, a protein expressed on the surface of B cells.
The CAR T-cell therapy already has orphan designation for the treatment of diffuse large B-cell lymphoma in the US and the European Union (EU).
KTE-C19 also has COMP positive opinions for orphan designation in the EU for primary mediastinal B-cell lymphoma and mantle cell lymphoma.
About orphan designation
The COMP adopts an opinion on the granting of orphan designation, and that opinion is submitted to the European Commission for endorsement.
In the EU, orphan designation is granted to therapies intended to treat a life-threatening or chronically debilitating condition that affects no more than 5 in 10,000 persons and where no satisfactory treatment is available.
Companies that obtain orphan designation for a drug benefit from a number of incentives, including protocol assistance, a type of scientific advice specific for designated orphan medicines, and 10 years of market exclusivity once the medicine is approved. Fee reductions are also available, depending on the status of the sponsor and the type of service required.
KTE-C19 research
Last year, researchers reported results with KTE-C19 in the Journal of Clinical Oncology. The study included 15 patients with advanced B-cell malignancies.
The patients received a conditioning regimen of cyclophosphamide and fludarabine, followed 1 day later by a single infusion of the CAR T-cell therapy. The researchers noted that the conditioning regimen is known to be active against B-cell malignancies and could have made a direct contribution to patient responses.
Thirteen patients were evaluable for response. Eight patients achieved a complete response (CR), and 4 had a partial response (PR).
Of the 7 patients with chemotherapy-refractory diffuse large B-cell lymphoma, 4 achieved a CR, 2 achieved a PR, and 1 had stable disease. Of the 4 patients with chronic lymphocytic leukemia, 3 had a CR, and 1 had a PR. Among the 2 patients with indolent lymphomas, 1 achieved a CR, and 1 had a PR.
KTE-C19 was associated with fever, low blood pressure, focal neurological deficits, and delirium. Toxicities largely occurred in the first 2 weeks after infusion.
All but 2 patients experienced grade 3/4 adverse events. Four patients had grade 3/4 hypotension.
All patients had elevations in serum interferon gamma and/or interleukin 6 around the time of peak toxicity, but most did not develop elevations in serum tumor necrosis factor.
Neurologic toxicities included confusion and obtundation, which have been reported in previous studies. However, 3 patients developed unexpected neurologic abnormalities.
KTE-C19 is currently under investigation in a phase 1/2 trial (ZUMA-1) of patients with refractory, aggressive non-Hodgkin lymphomas. Kite Pharma, Inc., the company developing KTE-C19, plans to present top-line phase 1 data at the 2015 ASH Annual Meeting. ![]()

The European Medicines Agency’s Committee for Orphan Medicinal Products (COMP) has adopted positive opinions recommending orphan designation for KTE-C19 to treat acute lymphoblastic leukemia, chronic lymphocytic leukemia/small lymphocytic lymphoma, and follicular lymphoma.
KTE-C19 is an investigational chimeric antigen receptor (CAR) T-cell therapy designed to target CD19, a protein expressed on the surface of B cells.
The CAR T-cell therapy already has orphan designation for the treatment of diffuse large B-cell lymphoma in the US and the European Union (EU).
KTE-C19 also has COMP positive opinions for orphan designation in the EU for primary mediastinal B-cell lymphoma and mantle cell lymphoma.
About orphan designation
The COMP adopts an opinion on the granting of orphan designation, and that opinion is submitted to the European Commission for endorsement.
In the EU, orphan designation is granted to therapies intended to treat a life-threatening or chronically debilitating condition that affects no more than 5 in 10,000 persons and where no satisfactory treatment is available.
Companies that obtain orphan designation for a drug benefit from a number of incentives, including protocol assistance, a type of scientific advice specific for designated orphan medicines, and 10 years of market exclusivity once the medicine is approved. Fee reductions are also available, depending on the status of the sponsor and the type of service required.
KTE-C19 research
Last year, researchers reported results with KTE-C19 in the Journal of Clinical Oncology. The study included 15 patients with advanced B-cell malignancies.
The patients received a conditioning regimen of cyclophosphamide and fludarabine, followed 1 day later by a single infusion of the CAR T-cell therapy. The researchers noted that the conditioning regimen is known to be active against B-cell malignancies and could have made a direct contribution to patient responses.
Thirteen patients were evaluable for response. Eight patients achieved a complete response (CR), and 4 had a partial response (PR).
Of the 7 patients with chemotherapy-refractory diffuse large B-cell lymphoma, 4 achieved a CR, 2 achieved a PR, and 1 had stable disease. Of the 4 patients with chronic lymphocytic leukemia, 3 had a CR, and 1 had a PR. Among the 2 patients with indolent lymphomas, 1 achieved a CR, and 1 had a PR.
KTE-C19 was associated with fever, low blood pressure, focal neurological deficits, and delirium. Toxicities largely occurred in the first 2 weeks after infusion.
All but 2 patients experienced grade 3/4 adverse events. Four patients had grade 3/4 hypotension.
All patients had elevations in serum interferon gamma and/or interleukin 6 around the time of peak toxicity, but most did not develop elevations in serum tumor necrosis factor.
Neurologic toxicities included confusion and obtundation, which have been reported in previous studies. However, 3 patients developed unexpected neurologic abnormalities.
KTE-C19 is currently under investigation in a phase 1/2 trial (ZUMA-1) of patients with refractory, aggressive non-Hodgkin lymphomas. Kite Pharma, Inc., the company developing KTE-C19, plans to present top-line phase 1 data at the 2015 ASH Annual Meeting. ![]()
Computerized cognitive training benefits childhood cancer survivors

Conklin and Jason Ashford
Photo courtesy of St. Jude
Children’s Research Hospital
and Peter Barta
Cognitive training presented as a video game can help improve cognitive skills in childhood cancer survivors (CCSs), new research suggests.
CCSs who completed 20 to 30 training sessions with this game experienced significant improvements in working memory, attention, and the speed at which their brains process information.
However, these improvements did not translate to improved math or reading performance.
Heather Conklin, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and her colleagues conducted this research and described the results in the Journal of Clinical Oncology.
Study design
This study included 68 CCSs who had received cranial irradiation, intrathecal chemotherapy, or both for the treatment of acute lymphoblastic leukemia or brain tumors.
Participants were 8 to 16 years old, had completed treatment, and had been disease-free for at least a year. Prior to joining the study, all scored below expectations on measures of working memory.
The computerized intervention the CCSs used is called Cogmed (http://www.cogmed.com). It’s a working memory intervention that has previously demonstrated efficacy for individuals with developmental and acquired attention disorders as well as for healthy adults.
For this study, half the CCSs were randomized to begin using Cogmed immediately. The remaining survivors, who served as the control group, were given the opportunity to use Cogmed about 6 months later.
The CCSs assigned to Cogmed first were asked to complete 25 training sessions at home, along with weekly, telephone-based coaching. The training sessions lasted 30 to 45 minutes and included verbal and visual-spatial exercises that were presented as games and are designed to improve working memory.
CCSs who began training immediately underwent functional brain MRI before and soon after completing the intervention. The imaging tracked brain activity as the survivors completed a working memory exercise.
Results
CCSs who completed the intervention (n=30) showed greater improvements than controls on measures of working memory (P=0.002), attention (P=0.01), and processing speed (P=0.02).
The researchers said the benefits to working memory and attention were comparable to gains reported in previous studies of stimulant medications. And the gains from cognitive training moved the CCSs’ performance into the normal range.
Caregivers also reported significant improvement in the attention and executive functioning of CCSs who completed the training. (Executive functioning includes skills like planning and focus needed to juggle multiple tasks and get things done.)
“These results suggest that computerized cognitive training may help fill a void in management of cognitive late effects that impact quality of life for childhood cancer survivors, such as the likelihood they will complete school and live independently,” Dr Conklin said.
In addition, post-intervention brain imaging showed decreased activation of left lateral prefrontal and bilateral medial frontal areas.
“That suggests the intervention exercised and strengthened the well-established working memory network,” Dr Conklin said. “The implication is that the brain may operate more efficiently and have less need for compensatory strategies. Such training-induced neuroplasticity suggests the benefits might be sustained going forward.”
The researchers are now studying the possible benefits of starting brain training during treatment or combining it with other interventions. The team is also tracking whether the cognitive benefits are sustained and might eventually translate to the improved academic performance reported for other populations. ![]()

Conklin and Jason Ashford
Photo courtesy of St. Jude
Children’s Research Hospital
and Peter Barta
Cognitive training presented as a video game can help improve cognitive skills in childhood cancer survivors (CCSs), new research suggests.
CCSs who completed 20 to 30 training sessions with this game experienced significant improvements in working memory, attention, and the speed at which their brains process information.
However, these improvements did not translate to improved math or reading performance.
Heather Conklin, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and her colleagues conducted this research and described the results in the Journal of Clinical Oncology.
Study design
This study included 68 CCSs who had received cranial irradiation, intrathecal chemotherapy, or both for the treatment of acute lymphoblastic leukemia or brain tumors.
Participants were 8 to 16 years old, had completed treatment, and had been disease-free for at least a year. Prior to joining the study, all scored below expectations on measures of working memory.
The computerized intervention the CCSs used is called Cogmed (http://www.cogmed.com). It’s a working memory intervention that has previously demonstrated efficacy for individuals with developmental and acquired attention disorders as well as for healthy adults.
For this study, half the CCSs were randomized to begin using Cogmed immediately. The remaining survivors, who served as the control group, were given the opportunity to use Cogmed about 6 months later.
The CCSs assigned to Cogmed first were asked to complete 25 training sessions at home, along with weekly, telephone-based coaching. The training sessions lasted 30 to 45 minutes and included verbal and visual-spatial exercises that were presented as games and are designed to improve working memory.
CCSs who began training immediately underwent functional brain MRI before and soon after completing the intervention. The imaging tracked brain activity as the survivors completed a working memory exercise.
Results
CCSs who completed the intervention (n=30) showed greater improvements than controls on measures of working memory (P=0.002), attention (P=0.01), and processing speed (P=0.02).
The researchers said the benefits to working memory and attention were comparable to gains reported in previous studies of stimulant medications. And the gains from cognitive training moved the CCSs’ performance into the normal range.
Caregivers also reported significant improvement in the attention and executive functioning of CCSs who completed the training. (Executive functioning includes skills like planning and focus needed to juggle multiple tasks and get things done.)
“These results suggest that computerized cognitive training may help fill a void in management of cognitive late effects that impact quality of life for childhood cancer survivors, such as the likelihood they will complete school and live independently,” Dr Conklin said.
In addition, post-intervention brain imaging showed decreased activation of left lateral prefrontal and bilateral medial frontal areas.
“That suggests the intervention exercised and strengthened the well-established working memory network,” Dr Conklin said. “The implication is that the brain may operate more efficiently and have less need for compensatory strategies. Such training-induced neuroplasticity suggests the benefits might be sustained going forward.”
The researchers are now studying the possible benefits of starting brain training during treatment or combining it with other interventions. The team is also tracking whether the cognitive benefits are sustained and might eventually translate to the improved academic performance reported for other populations. ![]()

Conklin and Jason Ashford
Photo courtesy of St. Jude
Children’s Research Hospital
and Peter Barta
Cognitive training presented as a video game can help improve cognitive skills in childhood cancer survivors (CCSs), new research suggests.
CCSs who completed 20 to 30 training sessions with this game experienced significant improvements in working memory, attention, and the speed at which their brains process information.
However, these improvements did not translate to improved math or reading performance.
Heather Conklin, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and her colleagues conducted this research and described the results in the Journal of Clinical Oncology.
Study design
This study included 68 CCSs who had received cranial irradiation, intrathecal chemotherapy, or both for the treatment of acute lymphoblastic leukemia or brain tumors.
Participants were 8 to 16 years old, had completed treatment, and had been disease-free for at least a year. Prior to joining the study, all scored below expectations on measures of working memory.
The computerized intervention the CCSs used is called Cogmed (http://www.cogmed.com). It’s a working memory intervention that has previously demonstrated efficacy for individuals with developmental and acquired attention disorders as well as for healthy adults.
For this study, half the CCSs were randomized to begin using Cogmed immediately. The remaining survivors, who served as the control group, were given the opportunity to use Cogmed about 6 months later.
The CCSs assigned to Cogmed first were asked to complete 25 training sessions at home, along with weekly, telephone-based coaching. The training sessions lasted 30 to 45 minutes and included verbal and visual-spatial exercises that were presented as games and are designed to improve working memory.
CCSs who began training immediately underwent functional brain MRI before and soon after completing the intervention. The imaging tracked brain activity as the survivors completed a working memory exercise.
Results
CCSs who completed the intervention (n=30) showed greater improvements than controls on measures of working memory (P=0.002), attention (P=0.01), and processing speed (P=0.02).
The researchers said the benefits to working memory and attention were comparable to gains reported in previous studies of stimulant medications. And the gains from cognitive training moved the CCSs’ performance into the normal range.
Caregivers also reported significant improvement in the attention and executive functioning of CCSs who completed the training. (Executive functioning includes skills like planning and focus needed to juggle multiple tasks and get things done.)
“These results suggest that computerized cognitive training may help fill a void in management of cognitive late effects that impact quality of life for childhood cancer survivors, such as the likelihood they will complete school and live independently,” Dr Conklin said.
In addition, post-intervention brain imaging showed decreased activation of left lateral prefrontal and bilateral medial frontal areas.
“That suggests the intervention exercised and strengthened the well-established working memory network,” Dr Conklin said. “The implication is that the brain may operate more efficiently and have less need for compensatory strategies. Such training-induced neuroplasticity suggests the benefits might be sustained going forward.”
The researchers are now studying the possible benefits of starting brain training during treatment or combining it with other interventions. The team is also tracking whether the cognitive benefits are sustained and might eventually translate to the improved academic performance reported for other populations. ![]()
Case suggests GSIs could treat Notch-mutated ALL
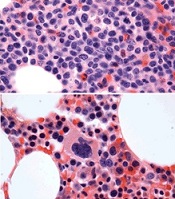
before (top) and after 7 weeks
of treatment (bottom)
© Knoechel et al.
Results of a case study suggest a gamma-secretase inhibitor (GSI) can be effective against Notch-mutated acute lymphoblastic leukemia (ALL).
The patient, who had early T-cell precursor ALL (ETP-ALL), achieved a complete hematologic response to treatment with BMS-906024, a GSI with anti-Notch
activity.
The patient was then able to proceed to hematopoietic stem cell transplant and was leukemia-free at last follow-up.
The researchers said this suggests that GSIs might hold promise for treating ALL and other cancers characterized by Notch mutations.
Birgit Knoechel, MD, PhD, of the Dana-Farber Cancer Institute in Boston, Massachusetts, and her colleagues described this case study in Cold Spring Harbor Molecular Case Studies.
The patient was a 53-year-old male with ETP-ALL who had failed previous rounds of chemotherapy and was then enrolled in a clinical trial of BMS-906024.
The patient began to show immediate improvement after starting treatment with the GSI. After 3 cycles, he went on to transplant and has since been leukemia-free—for 19 months so far.
To determine the genetic basis for the patient’s response to BMS-906024, researchers performed targeted and whole-exome sequencing on his leukemic cells.
They identified 4 potential mutations driving disease progression, including a novel mutation in the NOTCH1 gene that resulted in hyperactive signaling. This mutated gene copy was also duplicated in the cancer genome, resulting in elevated expression.
However, the NOTCH1 mutation, along with 2 of the other mutations, were absent in the remission bone marrow.
The researchers also cultured the patient’s leukemic cells to determine the molecular response to treatment.
Cells treated with BMS-906024 had greatly reduced levels of mutated NOTCH1 protein. RNA sequencing demonstrated that Notch target genes were sensitive to the treatment.
The MYC oncogene, on the other hand, was not sensitive to BMS-906024.
Epigenetic analysis revealed that the enhancer driving MYC expression in the leukemic cells was not Notch-dependent, but rather BRD4-dependent, suggesting another possible therapeutic option for MYC-expressing tumors. ![]()

before (top) and after 7 weeks
of treatment (bottom)
© Knoechel et al.
Results of a case study suggest a gamma-secretase inhibitor (GSI) can be effective against Notch-mutated acute lymphoblastic leukemia (ALL).
The patient, who had early T-cell precursor ALL (ETP-ALL), achieved a complete hematologic response to treatment with BMS-906024, a GSI with anti-Notch
activity.
The patient was then able to proceed to hematopoietic stem cell transplant and was leukemia-free at last follow-up.
The researchers said this suggests that GSIs might hold promise for treating ALL and other cancers characterized by Notch mutations.
Birgit Knoechel, MD, PhD, of the Dana-Farber Cancer Institute in Boston, Massachusetts, and her colleagues described this case study in Cold Spring Harbor Molecular Case Studies.
The patient was a 53-year-old male with ETP-ALL who had failed previous rounds of chemotherapy and was then enrolled in a clinical trial of BMS-906024.
The patient began to show immediate improvement after starting treatment with the GSI. After 3 cycles, he went on to transplant and has since been leukemia-free—for 19 months so far.
To determine the genetic basis for the patient’s response to BMS-906024, researchers performed targeted and whole-exome sequencing on his leukemic cells.
They identified 4 potential mutations driving disease progression, including a novel mutation in the NOTCH1 gene that resulted in hyperactive signaling. This mutated gene copy was also duplicated in the cancer genome, resulting in elevated expression.
However, the NOTCH1 mutation, along with 2 of the other mutations, were absent in the remission bone marrow.
The researchers also cultured the patient’s leukemic cells to determine the molecular response to treatment.
Cells treated with BMS-906024 had greatly reduced levels of mutated NOTCH1 protein. RNA sequencing demonstrated that Notch target genes were sensitive to the treatment.
The MYC oncogene, on the other hand, was not sensitive to BMS-906024.
Epigenetic analysis revealed that the enhancer driving MYC expression in the leukemic cells was not Notch-dependent, but rather BRD4-dependent, suggesting another possible therapeutic option for MYC-expressing tumors. ![]()

before (top) and after 7 weeks
of treatment (bottom)
© Knoechel et al.
Results of a case study suggest a gamma-secretase inhibitor (GSI) can be effective against Notch-mutated acute lymphoblastic leukemia (ALL).
The patient, who had early T-cell precursor ALL (ETP-ALL), achieved a complete hematologic response to treatment with BMS-906024, a GSI with anti-Notch
activity.
The patient was then able to proceed to hematopoietic stem cell transplant and was leukemia-free at last follow-up.
The researchers said this suggests that GSIs might hold promise for treating ALL and other cancers characterized by Notch mutations.
Birgit Knoechel, MD, PhD, of the Dana-Farber Cancer Institute in Boston, Massachusetts, and her colleagues described this case study in Cold Spring Harbor Molecular Case Studies.
The patient was a 53-year-old male with ETP-ALL who had failed previous rounds of chemotherapy and was then enrolled in a clinical trial of BMS-906024.
The patient began to show immediate improvement after starting treatment with the GSI. After 3 cycles, he went on to transplant and has since been leukemia-free—for 19 months so far.
To determine the genetic basis for the patient’s response to BMS-906024, researchers performed targeted and whole-exome sequencing on his leukemic cells.
They identified 4 potential mutations driving disease progression, including a novel mutation in the NOTCH1 gene that resulted in hyperactive signaling. This mutated gene copy was also duplicated in the cancer genome, resulting in elevated expression.
However, the NOTCH1 mutation, along with 2 of the other mutations, were absent in the remission bone marrow.
The researchers also cultured the patient’s leukemic cells to determine the molecular response to treatment.
Cells treated with BMS-906024 had greatly reduced levels of mutated NOTCH1 protein. RNA sequencing demonstrated that Notch target genes were sensitive to the treatment.
The MYC oncogene, on the other hand, was not sensitive to BMS-906024.
Epigenetic analysis revealed that the enhancer driving MYC expression in the leukemic cells was not Notch-dependent, but rather BRD4-dependent, suggesting another possible therapeutic option for MYC-expressing tumors. ![]()
New chimeric CD19 antibody may reduce MRD in ALL

NEW YORK—Researchers have developed a pharmaceutical-grade, third-generation, CD19-specific antibody that reduced minimal residual disease (MRD) in pediatric patients with B-cell precursor acute lymphoblastic leukemia (BCP-ALL).
This chimerized, Fc-optimized antibody—4G7SDIE—was used on a compassionate-need basis in 14 patients with relapsed or refractory BCP-ALL. Nine of the patients had prior stem cell transplants.
Ursula JE Seidel, a PhD candidate at University Children’s Hospital Tubingen in Germany, discussed early results with the new antibody (poster B144) during the inaugural CRI-CIMT-EATI-AACR International Cancer Immunotherapy Conference.
Patients received 4G7SDIE infusions ranging from 5 mg/m2 to 50 mg/m2 twice a week for a year or longer.
They rarely experienced fever, nausea, or headache, according to the investigators, and all had B-cell depletion.
“The good thing about this antibody is it has a very low toxicity profile,” Seidel noted.
Upon discontinuation of therapy, B-cell counts recovered rapidly to normal levels.
The researchers followed the patients for a median of 543 days after transplant (range, 208–1137) and a median of 720 days after administration of 4G7SDIE (range, 264–1115).
Nine of the 14 patients had a reduction in MRD by 1 log or more, 2 of whom were receiving additional therapy with tyrosine kinase inhibitors.
Five patients had a reduction in MRD below the quantifiable level, and 2 patients became MRD-negative.
Six patients relapsed, and 5 of them died from relapsed disease. Two patients died of sepsis or chemotoxicity while in complete molecular remission. And 6 patients remain in complete molecular remission.
Functional characterization of 4G7SDIE
Through analysis of cells from healthy volunteers and BCP-ALL blasts of untreated and treated patients, the researchers determined that 4G7SDIE mediates enhanced antibody‑dependent cellular cytotoxicity through its improved capability to recruit FcγRIIIa-bearing effector cells.
They identified natural killer cells and γδ T cells as the main effector cells. And they determined that the FcγRIIIa-V158F polymorphism did not influence the effect of 4G7SDIE-mediated antibody‑dependent cellular cytotoxicity.
The researchers believe that the promising anti-leukemic effects of 4G7SDIE both in vitro and in vivo call for additional exploration. They are currently planning a phase 1/2 study to further assess the therapeutic activity of 4G7SDIE. ![]()

NEW YORK—Researchers have developed a pharmaceutical-grade, third-generation, CD19-specific antibody that reduced minimal residual disease (MRD) in pediatric patients with B-cell precursor acute lymphoblastic leukemia (BCP-ALL).
This chimerized, Fc-optimized antibody—4G7SDIE—was used on a compassionate-need basis in 14 patients with relapsed or refractory BCP-ALL. Nine of the patients had prior stem cell transplants.
Ursula JE Seidel, a PhD candidate at University Children’s Hospital Tubingen in Germany, discussed early results with the new antibody (poster B144) during the inaugural CRI-CIMT-EATI-AACR International Cancer Immunotherapy Conference.
Patients received 4G7SDIE infusions ranging from 5 mg/m2 to 50 mg/m2 twice a week for a year or longer.
They rarely experienced fever, nausea, or headache, according to the investigators, and all had B-cell depletion.
“The good thing about this antibody is it has a very low toxicity profile,” Seidel noted.
Upon discontinuation of therapy, B-cell counts recovered rapidly to normal levels.
The researchers followed the patients for a median of 543 days after transplant (range, 208–1137) and a median of 720 days after administration of 4G7SDIE (range, 264–1115).
Nine of the 14 patients had a reduction in MRD by 1 log or more, 2 of whom were receiving additional therapy with tyrosine kinase inhibitors.
Five patients had a reduction in MRD below the quantifiable level, and 2 patients became MRD-negative.
Six patients relapsed, and 5 of them died from relapsed disease. Two patients died of sepsis or chemotoxicity while in complete molecular remission. And 6 patients remain in complete molecular remission.
Functional characterization of 4G7SDIE
Through analysis of cells from healthy volunteers and BCP-ALL blasts of untreated and treated patients, the researchers determined that 4G7SDIE mediates enhanced antibody‑dependent cellular cytotoxicity through its improved capability to recruit FcγRIIIa-bearing effector cells.
They identified natural killer cells and γδ T cells as the main effector cells. And they determined that the FcγRIIIa-V158F polymorphism did not influence the effect of 4G7SDIE-mediated antibody‑dependent cellular cytotoxicity.
The researchers believe that the promising anti-leukemic effects of 4G7SDIE both in vitro and in vivo call for additional exploration. They are currently planning a phase 1/2 study to further assess the therapeutic activity of 4G7SDIE. ![]()

NEW YORK—Researchers have developed a pharmaceutical-grade, third-generation, CD19-specific antibody that reduced minimal residual disease (MRD) in pediatric patients with B-cell precursor acute lymphoblastic leukemia (BCP-ALL).
This chimerized, Fc-optimized antibody—4G7SDIE—was used on a compassionate-need basis in 14 patients with relapsed or refractory BCP-ALL. Nine of the patients had prior stem cell transplants.
Ursula JE Seidel, a PhD candidate at University Children’s Hospital Tubingen in Germany, discussed early results with the new antibody (poster B144) during the inaugural CRI-CIMT-EATI-AACR International Cancer Immunotherapy Conference.
Patients received 4G7SDIE infusions ranging from 5 mg/m2 to 50 mg/m2 twice a week for a year or longer.
They rarely experienced fever, nausea, or headache, according to the investigators, and all had B-cell depletion.
“The good thing about this antibody is it has a very low toxicity profile,” Seidel noted.
Upon discontinuation of therapy, B-cell counts recovered rapidly to normal levels.
The researchers followed the patients for a median of 543 days after transplant (range, 208–1137) and a median of 720 days after administration of 4G7SDIE (range, 264–1115).
Nine of the 14 patients had a reduction in MRD by 1 log or more, 2 of whom were receiving additional therapy with tyrosine kinase inhibitors.
Five patients had a reduction in MRD below the quantifiable level, and 2 patients became MRD-negative.
Six patients relapsed, and 5 of them died from relapsed disease. Two patients died of sepsis or chemotoxicity while in complete molecular remission. And 6 patients remain in complete molecular remission.
Functional characterization of 4G7SDIE
Through analysis of cells from healthy volunteers and BCP-ALL blasts of untreated and treated patients, the researchers determined that 4G7SDIE mediates enhanced antibody‑dependent cellular cytotoxicity through its improved capability to recruit FcγRIIIa-bearing effector cells.
They identified natural killer cells and γδ T cells as the main effector cells. And they determined that the FcγRIIIa-V158F polymorphism did not influence the effect of 4G7SDIE-mediated antibody‑dependent cellular cytotoxicity.
The researchers believe that the promising anti-leukemic effects of 4G7SDIE both in vitro and in vivo call for additional exploration. They are currently planning a phase 1/2 study to further assess the therapeutic activity of 4G7SDIE. ![]()
Nonviral gene transfer of CARs tested in humans

Photo courtesy of MDACC
NEW YORK—Researchers have used a nonviral approach to create chimeric antigen receptor (CAR) T cells and tested these cells in safety trials.
Patients with advanced lymphoma or leukemia were infused with the nonvirally modified CD19-directed CAR T cells after autologous or allogeneic hematopoietic stem cell transplant (HSCT).
Eighty-six percent of autologous HSCT recipients were alive 24 months after infusion, and 53% of allogeneic HSCT recipients were alive with a median follow-up of 6.5 months.
“Gratifyingly, the patients have not demonstrated any acute or late toxicity to these CAR T-cell infusions,” said Laurence Cooper, MD, PhD, formerly of MD Anderson Cancer Center (MDACC) in Houston, Texas, and now with Ziopharm Oncology.
Dr Cooper presented these results at the inaugural CRI-CIMT-EATI-AACR International Cancer Immunotherapy Conference.
Some of the technology he described was conducted at MDACC. Dr Cooper is currently a visiting scientist there and will continue to supervise the development of this technology.
Dr Cooper said the appeal of this nonviral approach, which is a modified Sleeping Beauty approach, “is it essentially avoids the complexity of making a virus, a lentivirus or a retrovirus, it can be done at quite low cost, and really allows for a nimbleness to this system.”
Using a simple blood draw of 200 cc of peripheral blood—the process does not require apheresis—the T cells can be expanded on a feeder cell layer and genetically reprogrammed.
Sleeping Beauty system
The researchers reprogrammed the T cells using a 2-plasmid Sleeping Beauty system, which is a transposon/transposase system.
The transposon DNA plasmid codes for the cargo load, which, in this case, is the CAR. At the same time, the transposase DNA plasmid is electroporated, “which is really the secret sauce of the transposition event,” Dr Cooper explained.
After electroporation, the transposon/transposase are co-cultured with K562-derived artificial antigen-presenting cells (aAPC) and expanded with the integrated transposon of K562-aAPC. In this case, CD19 is on the aAPC.
CD19 is co-expressed with other co-stimulatory molecules, CD86 and 4-1BB ligand.
In addition, the researchers added a molecule of interleukin 15 that’s sewn in frame to the Fc region of an immunoglobulin that then activates the T cell in the context of these co-stimulatory molecules.
The T cells that have stable integrants of the CAR grow out over time. And those that have transient expression of the CAR die by neglect.
“By day 14, most of the T cells have the CAR sewn into the genome and are stably expressed,” Dr Cooper said.
The CAR used for these safety trials at MDACC targets CD19 and uses mouse scFv held in frame with an immunoglobulin 4 Fc (IgG4Fc) stalk.
It’s tunneled through the T-cell membrane and has 2 costimulatory molecules, signal 1 delivered by phosphorylation of the immunoreceptor tyrosine-based activation motif in CD3ζ and signal 2 by the costimulatory domain CD28.
The researchers tested the CD19 CARs in 2 clinical settings—one with T cells that were patient-derived and infused after autologous HSCT, and the second with T cells that were derived from a third party and infused after allogeneic HSCT.
Infusion after autologous HSCT
The researchers first tried the CARs in 7 non-Hodgkin lymphoma patients who had an autologous HSCT. Their median age was 52 (range, 36-61).
Five patients received a starting CAR T-cell dose of 5x108 cells/m2, and 2 received 5x109 cells/m2.
Six patients (86%) remain alive and are in complete remission (CR) at a median follow-up of 24 months.
Infusion after allogeneic HSCT
The researchers expanded the investigation to a wider cohort of 19 patients who had undergone allogeneic HSCT.
Seventeen patients had advanced CD19-positive acute lymphoblastic leukemia, and 2 had non-Hodgkin lymphoma. Their median age was 35 (range, 21-56).
All patients were on graft-versus-host disease (GVHD) prophylaxis with tacrolimus at the time of CAR infusion. A subset of these allogeneic transplant patients had haploidentical donors rather than matched sibling donors.
Five patients received a CAR T-cell dose of 106, 6 patients received 107, 5 received 5x107, and 3 received 5x108 cells/m2 based on recipient body surface area.
Fifty-eight percent of patients (11/19) achieved a CR, and 10 remain alive a median of 6.5 months after CAR T-cell infusion.
Three patients developed GVHD, 1 with steroid-refractory acute liver disease, 1 with grade 2 acute skin disease, and 1 with chronic limited skin disease. The incidence of GVHD was lower than historical controls at MDACC, Dr Cooper said.
“[G]ratifyingly, in this clinical setting of minimal disease, patients did not have any acute or late toxicity from these infusions,” he added.
And the rate of cytomegalovirus reactivation after CAR T-cell infusion was 24%, compared with 41% for patients after transplant at MDACC without CAR T-cell infusion.
Eight patients received haploidentical HSCT followed by CAR T-cell infusion, and 75% (6/8) remain in CR.
Persistence of infused T cells
The researchers used 2 forms of PCR—qPCR and droplet PCR—to map the fate of the CARs.
“Roughly speaking, for these patients, and this is in line with the literature, in terms of those T cells that are activated through CD28 in contrast to 4-1BB, these T cells are, on average, living about 28 or so days post-infusion,” Dr Cooper noted.
He said this is similar to results observed with CARs being tested at the National Cancer Institute and Memorial Sloan-Kettering Cancer Center. ![]()

Photo courtesy of MDACC
NEW YORK—Researchers have used a nonviral approach to create chimeric antigen receptor (CAR) T cells and tested these cells in safety trials.
Patients with advanced lymphoma or leukemia were infused with the nonvirally modified CD19-directed CAR T cells after autologous or allogeneic hematopoietic stem cell transplant (HSCT).
Eighty-six percent of autologous HSCT recipients were alive 24 months after infusion, and 53% of allogeneic HSCT recipients were alive with a median follow-up of 6.5 months.
“Gratifyingly, the patients have not demonstrated any acute or late toxicity to these CAR T-cell infusions,” said Laurence Cooper, MD, PhD, formerly of MD Anderson Cancer Center (MDACC) in Houston, Texas, and now with Ziopharm Oncology.
Dr Cooper presented these results at the inaugural CRI-CIMT-EATI-AACR International Cancer Immunotherapy Conference.
Some of the technology he described was conducted at MDACC. Dr Cooper is currently a visiting scientist there and will continue to supervise the development of this technology.
Dr Cooper said the appeal of this nonviral approach, which is a modified Sleeping Beauty approach, “is it essentially avoids the complexity of making a virus, a lentivirus or a retrovirus, it can be done at quite low cost, and really allows for a nimbleness to this system.”
Using a simple blood draw of 200 cc of peripheral blood—the process does not require apheresis—the T cells can be expanded on a feeder cell layer and genetically reprogrammed.
Sleeping Beauty system
The researchers reprogrammed the T cells using a 2-plasmid Sleeping Beauty system, which is a transposon/transposase system.
The transposon DNA plasmid codes for the cargo load, which, in this case, is the CAR. At the same time, the transposase DNA plasmid is electroporated, “which is really the secret sauce of the transposition event,” Dr Cooper explained.
After electroporation, the transposon/transposase are co-cultured with K562-derived artificial antigen-presenting cells (aAPC) and expanded with the integrated transposon of K562-aAPC. In this case, CD19 is on the aAPC.
CD19 is co-expressed with other co-stimulatory molecules, CD86 and 4-1BB ligand.
In addition, the researchers added a molecule of interleukin 15 that’s sewn in frame to the Fc region of an immunoglobulin that then activates the T cell in the context of these co-stimulatory molecules.
The T cells that have stable integrants of the CAR grow out over time. And those that have transient expression of the CAR die by neglect.
“By day 14, most of the T cells have the CAR sewn into the genome and are stably expressed,” Dr Cooper said.
The CAR used for these safety trials at MDACC targets CD19 and uses mouse scFv held in frame with an immunoglobulin 4 Fc (IgG4Fc) stalk.
It’s tunneled through the T-cell membrane and has 2 costimulatory molecules, signal 1 delivered by phosphorylation of the immunoreceptor tyrosine-based activation motif in CD3ζ and signal 2 by the costimulatory domain CD28.
The researchers tested the CD19 CARs in 2 clinical settings—one with T cells that were patient-derived and infused after autologous HSCT, and the second with T cells that were derived from a third party and infused after allogeneic HSCT.
Infusion after autologous HSCT
The researchers first tried the CARs in 7 non-Hodgkin lymphoma patients who had an autologous HSCT. Their median age was 52 (range, 36-61).
Five patients received a starting CAR T-cell dose of 5x108 cells/m2, and 2 received 5x109 cells/m2.
Six patients (86%) remain alive and are in complete remission (CR) at a median follow-up of 24 months.
Infusion after allogeneic HSCT
The researchers expanded the investigation to a wider cohort of 19 patients who had undergone allogeneic HSCT.
Seventeen patients had advanced CD19-positive acute lymphoblastic leukemia, and 2 had non-Hodgkin lymphoma. Their median age was 35 (range, 21-56).
All patients were on graft-versus-host disease (GVHD) prophylaxis with tacrolimus at the time of CAR infusion. A subset of these allogeneic transplant patients had haploidentical donors rather than matched sibling donors.
Five patients received a CAR T-cell dose of 106, 6 patients received 107, 5 received 5x107, and 3 received 5x108 cells/m2 based on recipient body surface area.
Fifty-eight percent of patients (11/19) achieved a CR, and 10 remain alive a median of 6.5 months after CAR T-cell infusion.
Three patients developed GVHD, 1 with steroid-refractory acute liver disease, 1 with grade 2 acute skin disease, and 1 with chronic limited skin disease. The incidence of GVHD was lower than historical controls at MDACC, Dr Cooper said.
“[G]ratifyingly, in this clinical setting of minimal disease, patients did not have any acute or late toxicity from these infusions,” he added.
And the rate of cytomegalovirus reactivation after CAR T-cell infusion was 24%, compared with 41% for patients after transplant at MDACC without CAR T-cell infusion.
Eight patients received haploidentical HSCT followed by CAR T-cell infusion, and 75% (6/8) remain in CR.
Persistence of infused T cells
The researchers used 2 forms of PCR—qPCR and droplet PCR—to map the fate of the CARs.
“Roughly speaking, for these patients, and this is in line with the literature, in terms of those T cells that are activated through CD28 in contrast to 4-1BB, these T cells are, on average, living about 28 or so days post-infusion,” Dr Cooper noted.
He said this is similar to results observed with CARs being tested at the National Cancer Institute and Memorial Sloan-Kettering Cancer Center. ![]()

Photo courtesy of MDACC
NEW YORK—Researchers have used a nonviral approach to create chimeric antigen receptor (CAR) T cells and tested these cells in safety trials.
Patients with advanced lymphoma or leukemia were infused with the nonvirally modified CD19-directed CAR T cells after autologous or allogeneic hematopoietic stem cell transplant (HSCT).
Eighty-six percent of autologous HSCT recipients were alive 24 months after infusion, and 53% of allogeneic HSCT recipients were alive with a median follow-up of 6.5 months.
“Gratifyingly, the patients have not demonstrated any acute or late toxicity to these CAR T-cell infusions,” said Laurence Cooper, MD, PhD, formerly of MD Anderson Cancer Center (MDACC) in Houston, Texas, and now with Ziopharm Oncology.
Dr Cooper presented these results at the inaugural CRI-CIMT-EATI-AACR International Cancer Immunotherapy Conference.
Some of the technology he described was conducted at MDACC. Dr Cooper is currently a visiting scientist there and will continue to supervise the development of this technology.
Dr Cooper said the appeal of this nonviral approach, which is a modified Sleeping Beauty approach, “is it essentially avoids the complexity of making a virus, a lentivirus or a retrovirus, it can be done at quite low cost, and really allows for a nimbleness to this system.”
Using a simple blood draw of 200 cc of peripheral blood—the process does not require apheresis—the T cells can be expanded on a feeder cell layer and genetically reprogrammed.
Sleeping Beauty system
The researchers reprogrammed the T cells using a 2-plasmid Sleeping Beauty system, which is a transposon/transposase system.
The transposon DNA plasmid codes for the cargo load, which, in this case, is the CAR. At the same time, the transposase DNA plasmid is electroporated, “which is really the secret sauce of the transposition event,” Dr Cooper explained.
After electroporation, the transposon/transposase are co-cultured with K562-derived artificial antigen-presenting cells (aAPC) and expanded with the integrated transposon of K562-aAPC. In this case, CD19 is on the aAPC.
CD19 is co-expressed with other co-stimulatory molecules, CD86 and 4-1BB ligand.
In addition, the researchers added a molecule of interleukin 15 that’s sewn in frame to the Fc region of an immunoglobulin that then activates the T cell in the context of these co-stimulatory molecules.
The T cells that have stable integrants of the CAR grow out over time. And those that have transient expression of the CAR die by neglect.
“By day 14, most of the T cells have the CAR sewn into the genome and are stably expressed,” Dr Cooper said.
The CAR used for these safety trials at MDACC targets CD19 and uses mouse scFv held in frame with an immunoglobulin 4 Fc (IgG4Fc) stalk.
It’s tunneled through the T-cell membrane and has 2 costimulatory molecules, signal 1 delivered by phosphorylation of the immunoreceptor tyrosine-based activation motif in CD3ζ and signal 2 by the costimulatory domain CD28.
The researchers tested the CD19 CARs in 2 clinical settings—one with T cells that were patient-derived and infused after autologous HSCT, and the second with T cells that were derived from a third party and infused after allogeneic HSCT.
Infusion after autologous HSCT
The researchers first tried the CARs in 7 non-Hodgkin lymphoma patients who had an autologous HSCT. Their median age was 52 (range, 36-61).
Five patients received a starting CAR T-cell dose of 5x108 cells/m2, and 2 received 5x109 cells/m2.
Six patients (86%) remain alive and are in complete remission (CR) at a median follow-up of 24 months.
Infusion after allogeneic HSCT
The researchers expanded the investigation to a wider cohort of 19 patients who had undergone allogeneic HSCT.
Seventeen patients had advanced CD19-positive acute lymphoblastic leukemia, and 2 had non-Hodgkin lymphoma. Their median age was 35 (range, 21-56).
All patients were on graft-versus-host disease (GVHD) prophylaxis with tacrolimus at the time of CAR infusion. A subset of these allogeneic transplant patients had haploidentical donors rather than matched sibling donors.
Five patients received a CAR T-cell dose of 106, 6 patients received 107, 5 received 5x107, and 3 received 5x108 cells/m2 based on recipient body surface area.
Fifty-eight percent of patients (11/19) achieved a CR, and 10 remain alive a median of 6.5 months after CAR T-cell infusion.
Three patients developed GVHD, 1 with steroid-refractory acute liver disease, 1 with grade 2 acute skin disease, and 1 with chronic limited skin disease. The incidence of GVHD was lower than historical controls at MDACC, Dr Cooper said.
“[G]ratifyingly, in this clinical setting of minimal disease, patients did not have any acute or late toxicity from these infusions,” he added.
And the rate of cytomegalovirus reactivation after CAR T-cell infusion was 24%, compared with 41% for patients after transplant at MDACC without CAR T-cell infusion.
Eight patients received haploidentical HSCT followed by CAR T-cell infusion, and 75% (6/8) remain in CR.
Persistence of infused T cells
The researchers used 2 forms of PCR—qPCR and droplet PCR—to map the fate of the CARs.
“Roughly speaking, for these patients, and this is in line with the literature, in terms of those T cells that are activated through CD28 in contrast to 4-1BB, these T cells are, on average, living about 28 or so days post-infusion,” Dr Cooper noted.
He said this is similar to results observed with CARs being tested at the National Cancer Institute and Memorial Sloan-Kettering Cancer Center. ![]()
CHMP recommends blinatumomab for ALL
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended conditional marketing authorization for blinatumomab (Blincyto) to treat adults with relapsed or refractory Philadelphia chromosome-negative (Ph-) B-precursor acute lymphoblastic leukemia (ALL).
Blinatumomab is a bispecific T-cell engager (BiTE®) antibody construct that binds to CD19 on the surface of B cells and CD3 on the surface of T cells.
The product already has conditional approval in the US to treat patients with relapsed or refractory Ph- B-precursor ALL.
The CHMP’s positive opinion of blinatumomab will be reviewed by the European Commission (EC).
The EC usually follows the CHMP’s recommendations and is expected to deliver its final decision within 3 months. The EC’s decision will apply to the 28 member countries of the European Union, as well as Iceland, Lichtenstein, and Norway.
Conditional marketing authorizations are valid for 1 year, on a renewable basis. The holder is required to complete ongoing studies or conduct new studies with the goal of confirming that a drug’s benefit-risk balance is positive.
Conditional marketing authorization is converted to a full authorization once these commitments have been fulfilled.
The conditional marketing authorization application for blinatumomab is based on a pair of phase 2 trials—Study ‘211 and Study ‘206.
Study ‘211
Results of Study ‘211 were presented at EHA 2014. The trial included 189 patients with Ph- relapsed or refractory B-precursor ALL.
The primary endpoint was complete remission or complete remission with partial hematologic recovery (CR/CRh). About 43% of patients achieved this endpoint within 2 cycles of therapy.
According to researchers, the most serious adverse events in this study were infection (31.7%), neurologic events (16.4%), neutropenia/febrile neutropenia (15.3%), cytokine release syndrome (CRS, 0.5%), and tumor lysis syndrome (0.5%).
Study ‘206
Results of Study ‘206 were presented at ASCO 2012. The trial included 36 patients with relapsed or refractory B-precursor ALL.
In this trial, the CR/CRh rate was 69.4% (25/36), with 15 patients achieving a CR (41.7%), and 10 patients achieving CRh (27.8%).
The “medically important” adverse events in this study, according to researchers, were CRS (n=3), central nervous system (CNS) events (3 seizures and 3 cases of encephalopathy), and fungal infection resulting in death (n=1).
However, the researchers found they could prevent CRS with dexamethasone. In addition, the CNS events were reversible, and blinatumomab could be reintroduced in 4 of the 6 patients with CNS events.
Blinatumomab is under development by Amgen. For more details on the drug, visit www.blincyto.com. ![]()

The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended conditional marketing authorization for blinatumomab (Blincyto) to treat adults with relapsed or refractory Philadelphia chromosome-negative (Ph-) B-precursor acute lymphoblastic leukemia (ALL).
Blinatumomab is a bispecific T-cell engager (BiTE®) antibody construct that binds to CD19 on the surface of B cells and CD3 on the surface of T cells.
The product already has conditional approval in the US to treat patients with relapsed or refractory Ph- B-precursor ALL.
The CHMP’s positive opinion of blinatumomab will be reviewed by the European Commission (EC).
The EC usually follows the CHMP’s recommendations and is expected to deliver its final decision within 3 months. The EC’s decision will apply to the 28 member countries of the European Union, as well as Iceland, Lichtenstein, and Norway.
Conditional marketing authorizations are valid for 1 year, on a renewable basis. The holder is required to complete ongoing studies or conduct new studies with the goal of confirming that a drug’s benefit-risk balance is positive.
Conditional marketing authorization is converted to a full authorization once these commitments have been fulfilled.
The conditional marketing authorization application for blinatumomab is based on a pair of phase 2 trials—Study ‘211 and Study ‘206.
Study ‘211
Results of Study ‘211 were presented at EHA 2014. The trial included 189 patients with Ph- relapsed or refractory B-precursor ALL.
The primary endpoint was complete remission or complete remission with partial hematologic recovery (CR/CRh). About 43% of patients achieved this endpoint within 2 cycles of therapy.
According to researchers, the most serious adverse events in this study were infection (31.7%), neurologic events (16.4%), neutropenia/febrile neutropenia (15.3%), cytokine release syndrome (CRS, 0.5%), and tumor lysis syndrome (0.5%).
Study ‘206
Results of Study ‘206 were presented at ASCO 2012. The trial included 36 patients with relapsed or refractory B-precursor ALL.
In this trial, the CR/CRh rate was 69.4% (25/36), with 15 patients achieving a CR (41.7%), and 10 patients achieving CRh (27.8%).
The “medically important” adverse events in this study, according to researchers, were CRS (n=3), central nervous system (CNS) events (3 seizures and 3 cases of encephalopathy), and fungal infection resulting in death (n=1).
However, the researchers found they could prevent CRS with dexamethasone. In addition, the CNS events were reversible, and blinatumomab could be reintroduced in 4 of the 6 patients with CNS events.
Blinatumomab is under development by Amgen. For more details on the drug, visit www.blincyto.com. ![]()

The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended conditional marketing authorization for blinatumomab (Blincyto) to treat adults with relapsed or refractory Philadelphia chromosome-negative (Ph-) B-precursor acute lymphoblastic leukemia (ALL).
Blinatumomab is a bispecific T-cell engager (BiTE®) antibody construct that binds to CD19 on the surface of B cells and CD3 on the surface of T cells.
The product already has conditional approval in the US to treat patients with relapsed or refractory Ph- B-precursor ALL.
The CHMP’s positive opinion of blinatumomab will be reviewed by the European Commission (EC).
The EC usually follows the CHMP’s recommendations and is expected to deliver its final decision within 3 months. The EC’s decision will apply to the 28 member countries of the European Union, as well as Iceland, Lichtenstein, and Norway.
Conditional marketing authorizations are valid for 1 year, on a renewable basis. The holder is required to complete ongoing studies or conduct new studies with the goal of confirming that a drug’s benefit-risk balance is positive.
Conditional marketing authorization is converted to a full authorization once these commitments have been fulfilled.
The conditional marketing authorization application for blinatumomab is based on a pair of phase 2 trials—Study ‘211 and Study ‘206.
Study ‘211
Results of Study ‘211 were presented at EHA 2014. The trial included 189 patients with Ph- relapsed or refractory B-precursor ALL.
The primary endpoint was complete remission or complete remission with partial hematologic recovery (CR/CRh). About 43% of patients achieved this endpoint within 2 cycles of therapy.
According to researchers, the most serious adverse events in this study were infection (31.7%), neurologic events (16.4%), neutropenia/febrile neutropenia (15.3%), cytokine release syndrome (CRS, 0.5%), and tumor lysis syndrome (0.5%).
Study ‘206
Results of Study ‘206 were presented at ASCO 2012. The trial included 36 patients with relapsed or refractory B-precursor ALL.
In this trial, the CR/CRh rate was 69.4% (25/36), with 15 patients achieving a CR (41.7%), and 10 patients achieving CRh (27.8%).
The “medically important” adverse events in this study, according to researchers, were CRS (n=3), central nervous system (CNS) events (3 seizures and 3 cases of encephalopathy), and fungal infection resulting in death (n=1).
However, the researchers found they could prevent CRS with dexamethasone. In addition, the CNS events were reversible, and blinatumomab could be reintroduced in 4 of the 6 patients with CNS events.
Blinatumomab is under development by Amgen. For more details on the drug, visit www.blincyto.com. ![]()
Traffic-related pollution linked to AML, not ALL, in kids

A French study has revealed an increased incidence of acute myeloid leukemia (AML) among children living close to heavily used roads.
The incidence of AML was 30% higher among children who lived within 150 m of heavily used roads and where the combined length of road sections
within this radius exceeded 260 m.
The researchers believe the association between AML and road proximity may be driven by traffic-related benzene exposure.
Previous research has shown an increased risk of leukemia among adults with a history of occupational exposure to benzene.
The current study did not suggest an increased risk of acute lymphoblastic leukemia (ALL) among children living closed to heavily used roads.
Jacqueline Clavel, MD, PhD, of INSERM in Paris, France, and her colleagues reported these findings in the American Journal of Epidemiology.
The team analyzed 2760 cases of leukemia diagnosed in children younger than 15 years of age in metropolitan France between 2002 and 2007, including 418 cases of AML and 2275 cases of ALL.
The researchers compared these cases to a contemporary sample of 30,000 control children representative of the metropolitan population.
The data showed that neither distance from the nearest major road(s) nor the length of major roads within 150 m of a child’s residence was associated with ALL.
However, there was an association for AML. For children whose home was less than 150 m from the nearest major road(s), the odds ratio (OR) was 1.2.
When the total length of major road(s) within 150 m from the child’s residence was 257-308 m (second tertile) or 309 m or greater (third tertile), the OR was 1.3. When the total length of major roads was 1-256 m (first tertile), the OR was 0.90.
The researchers noted that traffic-related nitrogen dioxide concentration was not associated with ALL or AML. But their data indicated that benzene concentration was associated with AML.
To assess this potential association, the team studied the Île-de-France region of Paris, the most urbanized region, for which the mean annual concentration of benzene, mainly from road traffic, was estimated in the vicinity of each residence.
The median estimated benzene concentration for controls living in the Île-de-France region was 1.3 μg/m3 (range, 0.3 to 8.5 μg/m3). And the length of major roads within 150 m of a child’s residence was positively and significantly correlated with log benzene concentration (r=0.3, P<0.001).
So it followed that exposure to an estimated benzene concentration greater than the median was associated with AML (OR=1.6).
The researchers also used a composite variable based on the estimated benzene concentration and the length of major roads around a child’s residence.
The association with AML was largest among children with at least 309 m of major roads within 150 m of their residence and estimated benzene concentrations of 1.3 μg/m3 or greater (OR=2.2).
The researchers said these results support a role for traffic-related benzene exposure in the etiology of childhood AML. ![]()

A French study has revealed an increased incidence of acute myeloid leukemia (AML) among children living close to heavily used roads.
The incidence of AML was 30% higher among children who lived within 150 m of heavily used roads and where the combined length of road sections
within this radius exceeded 260 m.
The researchers believe the association between AML and road proximity may be driven by traffic-related benzene exposure.
Previous research has shown an increased risk of leukemia among adults with a history of occupational exposure to benzene.
The current study did not suggest an increased risk of acute lymphoblastic leukemia (ALL) among children living closed to heavily used roads.
Jacqueline Clavel, MD, PhD, of INSERM in Paris, France, and her colleagues reported these findings in the American Journal of Epidemiology.
The team analyzed 2760 cases of leukemia diagnosed in children younger than 15 years of age in metropolitan France between 2002 and 2007, including 418 cases of AML and 2275 cases of ALL.
The researchers compared these cases to a contemporary sample of 30,000 control children representative of the metropolitan population.
The data showed that neither distance from the nearest major road(s) nor the length of major roads within 150 m of a child’s residence was associated with ALL.
However, there was an association for AML. For children whose home was less than 150 m from the nearest major road(s), the odds ratio (OR) was 1.2.
When the total length of major road(s) within 150 m from the child’s residence was 257-308 m (second tertile) or 309 m or greater (third tertile), the OR was 1.3. When the total length of major roads was 1-256 m (first tertile), the OR was 0.90.
The researchers noted that traffic-related nitrogen dioxide concentration was not associated with ALL or AML. But their data indicated that benzene concentration was associated with AML.
To assess this potential association, the team studied the Île-de-France region of Paris, the most urbanized region, for which the mean annual concentration of benzene, mainly from road traffic, was estimated in the vicinity of each residence.
The median estimated benzene concentration for controls living in the Île-de-France region was 1.3 μg/m3 (range, 0.3 to 8.5 μg/m3). And the length of major roads within 150 m of a child’s residence was positively and significantly correlated with log benzene concentration (r=0.3, P<0.001).
So it followed that exposure to an estimated benzene concentration greater than the median was associated with AML (OR=1.6).
The researchers also used a composite variable based on the estimated benzene concentration and the length of major roads around a child’s residence.
The association with AML was largest among children with at least 309 m of major roads within 150 m of their residence and estimated benzene concentrations of 1.3 μg/m3 or greater (OR=2.2).
The researchers said these results support a role for traffic-related benzene exposure in the etiology of childhood AML. ![]()

A French study has revealed an increased incidence of acute myeloid leukemia (AML) among children living close to heavily used roads.
The incidence of AML was 30% higher among children who lived within 150 m of heavily used roads and where the combined length of road sections
within this radius exceeded 260 m.
The researchers believe the association between AML and road proximity may be driven by traffic-related benzene exposure.
Previous research has shown an increased risk of leukemia among adults with a history of occupational exposure to benzene.
The current study did not suggest an increased risk of acute lymphoblastic leukemia (ALL) among children living closed to heavily used roads.
Jacqueline Clavel, MD, PhD, of INSERM in Paris, France, and her colleagues reported these findings in the American Journal of Epidemiology.
The team analyzed 2760 cases of leukemia diagnosed in children younger than 15 years of age in metropolitan France between 2002 and 2007, including 418 cases of AML and 2275 cases of ALL.
The researchers compared these cases to a contemporary sample of 30,000 control children representative of the metropolitan population.
The data showed that neither distance from the nearest major road(s) nor the length of major roads within 150 m of a child’s residence was associated with ALL.
However, there was an association for AML. For children whose home was less than 150 m from the nearest major road(s), the odds ratio (OR) was 1.2.
When the total length of major road(s) within 150 m from the child’s residence was 257-308 m (second tertile) or 309 m or greater (third tertile), the OR was 1.3. When the total length of major roads was 1-256 m (first tertile), the OR was 0.90.
The researchers noted that traffic-related nitrogen dioxide concentration was not associated with ALL or AML. But their data indicated that benzene concentration was associated with AML.
To assess this potential association, the team studied the Île-de-France region of Paris, the most urbanized region, for which the mean annual concentration of benzene, mainly from road traffic, was estimated in the vicinity of each residence.
The median estimated benzene concentration for controls living in the Île-de-France region was 1.3 μg/m3 (range, 0.3 to 8.5 μg/m3). And the length of major roads within 150 m of a child’s residence was positively and significantly correlated with log benzene concentration (r=0.3, P<0.001).
So it followed that exposure to an estimated benzene concentration greater than the median was associated with AML (OR=1.6).
The researchers also used a composite variable based on the estimated benzene concentration and the length of major roads around a child’s residence.
The association with AML was largest among children with at least 309 m of major roads within 150 m of their residence and estimated benzene concentrations of 1.3 μg/m3 or greater (OR=2.2).
The researchers said these results support a role for traffic-related benzene exposure in the etiology of childhood AML.
CAR T-cell therapy tested in Sweden

NEW YORK—For the first time, according to researchers, chimeric antigen receptor (CAR) T-cell therapy has been tested in a clinical trial in Sweden.
Early results have shown the treatment can produce complete responses (CRs) in leukemia and lymphoma, although most patients ultimately progressed.
Hannah Karlsson, PhD, of Uppsala University in Sweden, presented data from the phase 1/2a trial of the third-generation CD19 CAR T-cell therapy (abstract A041*) at the inaugural CRI-CIMT-EATI-AACR International Cancer Immunotherapy Conference.
The trial is a collaboration between Uppsala University and Baylor College of Medicine and was funded by AFA Insurances AB and the Swedish Cancer Society.
“Third-generation CAR T cells are being tested in clinical trials for leukemia patients in the United States with success,” said senior study author Angelica Loskog, PhD, also of Uppsala University.
“[T]he main purpose of our clinical trial was to evaluate whether we could reproduce the successful results in leukemia patients in Sweden and to also test if patients with lymphoma will also respond to this treatment.”
So the investigators enrolled 13 patients, 11 of whom were evaluable for efficacy at 3 months after CAR T-cell infusion. All patients had relapsed or refractory, CD19-positive, B-cell disease.
Two patients had acute lymphoblastic leukemia (ALL), 2 had chronic lymphocytic leukemia (CLL), and 7 had lymphoma—3 with diffuse large B-cell lymphoma (DLBCL), 2 with mantle cell lymphoma (MCL), 1 with follicular lymphoma (FL)/DLBCL, and 1 with Burkitt lymphoma.
All of the lymphoma patients received chemotherapy before CAR T-cell infusion to shrink their tumors. Seven patients—3 with leukemia and 4 with lymphoma—received pre-conditioning with cyclophosphamide plus fludarabine to reduce their immunosuppressive cell counts.
The investigators used CAR T cells containing signaling domains from both CD28 and 4-1BB and manufactured using a gamma retrovirus.
Patients received a single infusion of the CAR T cells, 2 patients at a dose of 2 x 107 cells/m2, 4 at a dose of 1 x 108 cells/m2, and 5 at 2 x 108 cells/m2.
Response and toxicity
Six patients had achieved a CR at the time of evaluation.
One patient with DLBCL experienced mild cytokine release syndrome (CRS) before achieving CR. However, the patient relapsed after a second CRS occurred (after 3 months).
Another DLBCL patient achieved a CR prior to T-cell infusion and remained in CR for 6 months before progressing.
One CLL patient and another DLBCL patient responded prior to T-cell infusion and remained in CR for more than 3 months. The CLL patient was still in CR at the time of the meeting.
One of the ALL patients achieved a CR after transient central nervous system toxicity but relapsed at 3 months with CD19-negative ALL. The other ALL patient was in CR for more than a month after experiencing CRS but ultimately progressed.
One CLL patient and 2 MCL patients had all progressed by 3 months.
The FL/DLBCL patient progressed after 1 month, with mild CRS. And the patient with Burkitt lymphoma had major CRS and progressive disease.
The investigators noted that 5 of the 6 patients who received pre-conditioning treatment had initial CRs.
The team is now analyzing whether there is any correlation between the level of immunosuppressive cells and patient response.
*Information presented at the meeting differs from the abstract.

NEW YORK—For the first time, according to researchers, chimeric antigen receptor (CAR) T-cell therapy has been tested in a clinical trial in Sweden.
Early results have shown the treatment can produce complete responses (CRs) in leukemia and lymphoma, although most patients ultimately progressed.
Hannah Karlsson, PhD, of Uppsala University in Sweden, presented data from the phase 1/2a trial of the third-generation CD19 CAR T-cell therapy (abstract A041*) at the inaugural CRI-CIMT-EATI-AACR International Cancer Immunotherapy Conference.
The trial is a collaboration between Uppsala University and Baylor College of Medicine and was funded by AFA Insurances AB and the Swedish Cancer Society.
“Third-generation CAR T cells are being tested in clinical trials for leukemia patients in the United States with success,” said senior study author Angelica Loskog, PhD, also of Uppsala University.
“[T]he main purpose of our clinical trial was to evaluate whether we could reproduce the successful results in leukemia patients in Sweden and to also test if patients with lymphoma will also respond to this treatment.”
So the investigators enrolled 13 patients, 11 of whom were evaluable for efficacy at 3 months after CAR T-cell infusion. All patients had relapsed or refractory, CD19-positive, B-cell disease.
Two patients had acute lymphoblastic leukemia (ALL), 2 had chronic lymphocytic leukemia (CLL), and 7 had lymphoma—3 with diffuse large B-cell lymphoma (DLBCL), 2 with mantle cell lymphoma (MCL), 1 with follicular lymphoma (FL)/DLBCL, and 1 with Burkitt lymphoma.
All of the lymphoma patients received chemotherapy before CAR T-cell infusion to shrink their tumors. Seven patients—3 with leukemia and 4 with lymphoma—received pre-conditioning with cyclophosphamide plus fludarabine to reduce their immunosuppressive cell counts.
The investigators used CAR T cells containing signaling domains from both CD28 and 4-1BB and manufactured using a gamma retrovirus.
Patients received a single infusion of the CAR T cells, 2 patients at a dose of 2 x 107 cells/m2, 4 at a dose of 1 x 108 cells/m2, and 5 at 2 x 108 cells/m2.
Response and toxicity
Six patients had achieved a CR at the time of evaluation.
One patient with DLBCL experienced mild cytokine release syndrome (CRS) before achieving CR. However, the patient relapsed after a second CRS occurred (after 3 months).
Another DLBCL patient achieved a CR prior to T-cell infusion and remained in CR for 6 months before progressing.
One CLL patient and another DLBCL patient responded prior to T-cell infusion and remained in CR for more than 3 months. The CLL patient was still in CR at the time of the meeting.
One of the ALL patients achieved a CR after transient central nervous system toxicity but relapsed at 3 months with CD19-negative ALL. The other ALL patient was in CR for more than a month after experiencing CRS but ultimately progressed.
One CLL patient and 2 MCL patients had all progressed by 3 months.
The FL/DLBCL patient progressed after 1 month, with mild CRS. And the patient with Burkitt lymphoma had major CRS and progressive disease.
The investigators noted that 5 of the 6 patients who received pre-conditioning treatment had initial CRs.
The team is now analyzing whether there is any correlation between the level of immunosuppressive cells and patient response.
*Information presented at the meeting differs from the abstract.

NEW YORK—For the first time, according to researchers, chimeric antigen receptor (CAR) T-cell therapy has been tested in a clinical trial in Sweden.
Early results have shown the treatment can produce complete responses (CRs) in leukemia and lymphoma, although most patients ultimately progressed.
Hannah Karlsson, PhD, of Uppsala University in Sweden, presented data from the phase 1/2a trial of the third-generation CD19 CAR T-cell therapy (abstract A041*) at the inaugural CRI-CIMT-EATI-AACR International Cancer Immunotherapy Conference.
The trial is a collaboration between Uppsala University and Baylor College of Medicine and was funded by AFA Insurances AB and the Swedish Cancer Society.
“Third-generation CAR T cells are being tested in clinical trials for leukemia patients in the United States with success,” said senior study author Angelica Loskog, PhD, also of Uppsala University.
“[T]he main purpose of our clinical trial was to evaluate whether we could reproduce the successful results in leukemia patients in Sweden and to also test if patients with lymphoma will also respond to this treatment.”
So the investigators enrolled 13 patients, 11 of whom were evaluable for efficacy at 3 months after CAR T-cell infusion. All patients had relapsed or refractory, CD19-positive, B-cell disease.
Two patients had acute lymphoblastic leukemia (ALL), 2 had chronic lymphocytic leukemia (CLL), and 7 had lymphoma—3 with diffuse large B-cell lymphoma (DLBCL), 2 with mantle cell lymphoma (MCL), 1 with follicular lymphoma (FL)/DLBCL, and 1 with Burkitt lymphoma.
All of the lymphoma patients received chemotherapy before CAR T-cell infusion to shrink their tumors. Seven patients—3 with leukemia and 4 with lymphoma—received pre-conditioning with cyclophosphamide plus fludarabine to reduce their immunosuppressive cell counts.
The investigators used CAR T cells containing signaling domains from both CD28 and 4-1BB and manufactured using a gamma retrovirus.
Patients received a single infusion of the CAR T cells, 2 patients at a dose of 2 x 107 cells/m2, 4 at a dose of 1 x 108 cells/m2, and 5 at 2 x 108 cells/m2.
Response and toxicity
Six patients had achieved a CR at the time of evaluation.
One patient with DLBCL experienced mild cytokine release syndrome (CRS) before achieving CR. However, the patient relapsed after a second CRS occurred (after 3 months).
Another DLBCL patient achieved a CR prior to T-cell infusion and remained in CR for 6 months before progressing.
One CLL patient and another DLBCL patient responded prior to T-cell infusion and remained in CR for more than 3 months. The CLL patient was still in CR at the time of the meeting.
One of the ALL patients achieved a CR after transient central nervous system toxicity but relapsed at 3 months with CD19-negative ALL. The other ALL patient was in CR for more than a month after experiencing CRS but ultimately progressed.
One CLL patient and 2 MCL patients had all progressed by 3 months.
The FL/DLBCL patient progressed after 1 month, with mild CRS. And the patient with Burkitt lymphoma had major CRS and progressive disease.
The investigators noted that 5 of the 6 patients who received pre-conditioning treatment had initial CRs.
The team is now analyzing whether there is any correlation between the level of immunosuppressive cells and patient response.
*Information presented at the meeting differs from the abstract.
Cancer report highlights progress, makes predictions

Photo courtesy of the FDA
Despite recent progress in the fight against cancers, these diseases continue to exert “an immense toll” in the US, according to the AACR Cancer Progress Report 2015.
The report highlights the recent approval by the US Food and Drug Administration (FDA) of several anticancer therapies, a vaccine, and 2 diagnostic aids.
But the report also includes data suggesting that cancer cases, and costs related to cancer care, are on the rise.
The report states that, between Aug. 1, 2014, and July 31, 2015, the FDA approved 9 anticancer therapies, either for the first time or for new indications.
During the same period, the FDA approved a new cancer vaccine, a new cancer screening test, and a new use for a previously approved imaging agent.
| Cancer-related products approved from Aug. 1, 2014 to July 31, 2015 | |
| Drug | Approved indication |
| bevacizumab (Avastin) | cervical, ovarian, fallopian
tube, and peritoneal cancers |
| blinatumomab (Blincyto) | acute lymphoblastic leukemia |
| denosumab (Xgeva) | potentially lethal complication
of advanced cancers |
| dinutuximab (Unituxin) | neuroblastoma |
| gefitinib (Iressa) | lung cancer |
| ibrutinib (Imbruvica) | Waldenstrom macroglobulinemia |
| lenvatinib (Lenvima) | thyroid cancer |
| nivolumab (Opdivo) | melanoma, lung cancer |
| olaparib (Lynparza) | ovarian cancer |
| palbociclib (Ibrance) | breast cancer |
| panobinostat (Farydak) | multiple myeloma |
| pembrolizumab (Keytruda) | melanoma |
| ramucirumab (Cyramza) | colorectal and lung cancers |
| sonidegib (Odomzo) | skin cancer |
| Imaging agent | Approved indication |
| technetium 99m tilmanocept
(Lymphoseek) |
lymphatic mapping in solid tumors |
| Vaccine | Approved indication |
| human papillomavirus
9-valent vaccine (Gardasil 9) |
cervical, vulvar,
vaginal, and anal cancers |
| Screening test | Approved indication |
| Cologuard (no generic name) | colorectal cancer |
Despite these advances, cancers continue to exert personal and economic tolls, according to the report.
It states that cancer is the number 1 cause of disease-related death among US children. And more than 589,000 people in the US are projected to die from cancer in 2015.
The number of new cancer cases in the US is predicted to rise from 1.7 million in 2015 to 2.4 million in 2035.
In addition, estimates suggest the direct medical costs of cancer care in the US in 2010 were nearly $125 billion, and these costs are predicted to rise to $156 billion in 2020.
These data underscore the need for more research to develop new approaches to cancer prevention and treatment, according to the report.
Its authors call for Congress and the administration to provide the National Institutes of Health, National Cancer Institute, and FDA with annual funding increases.
“We have made spectacular progress against cancer, which has saved the lives of millions of individuals in the United States and around the world,” said Margaret Foti, PhD, MD, chief executive officer of the AACR.
“However, without increased federal funding for cancer research, we will not be able to realize the promise of recent discoveries and technological advances.”

Photo courtesy of the FDA
Despite recent progress in the fight against cancers, these diseases continue to exert “an immense toll” in the US, according to the AACR Cancer Progress Report 2015.
The report highlights the recent approval by the US Food and Drug Administration (FDA) of several anticancer therapies, a vaccine, and 2 diagnostic aids.
But the report also includes data suggesting that cancer cases, and costs related to cancer care, are on the rise.
The report states that, between Aug. 1, 2014, and July 31, 2015, the FDA approved 9 anticancer therapies, either for the first time or for new indications.
During the same period, the FDA approved a new cancer vaccine, a new cancer screening test, and a new use for a previously approved imaging agent.
| Cancer-related products approved from Aug. 1, 2014 to July 31, 2015 | |
| Drug | Approved indication |
| bevacizumab (Avastin) | cervical, ovarian, fallopian
tube, and peritoneal cancers |
| blinatumomab (Blincyto) | acute lymphoblastic leukemia |
| denosumab (Xgeva) | potentially lethal complication
of advanced cancers |
| dinutuximab (Unituxin) | neuroblastoma |
| gefitinib (Iressa) | lung cancer |
| ibrutinib (Imbruvica) | Waldenstrom macroglobulinemia |
| lenvatinib (Lenvima) | thyroid cancer |
| nivolumab (Opdivo) | melanoma, lung cancer |
| olaparib (Lynparza) | ovarian cancer |
| palbociclib (Ibrance) | breast cancer |
| panobinostat (Farydak) | multiple myeloma |
| pembrolizumab (Keytruda) | melanoma |
| ramucirumab (Cyramza) | colorectal and lung cancers |
| sonidegib (Odomzo) | skin cancer |
| Imaging agent | Approved indication |
| technetium 99m tilmanocept
(Lymphoseek) |
lymphatic mapping in solid tumors |
| Vaccine | Approved indication |
| human papillomavirus
9-valent vaccine (Gardasil 9) |
cervical, vulvar,
vaginal, and anal cancers |
| Screening test | Approved indication |
| Cologuard (no generic name) | colorectal cancer |
Despite these advances, cancers continue to exert personal and economic tolls, according to the report.
It states that cancer is the number 1 cause of disease-related death among US children. And more than 589,000 people in the US are projected to die from cancer in 2015.
The number of new cancer cases in the US is predicted to rise from 1.7 million in 2015 to 2.4 million in 2035.
In addition, estimates suggest the direct medical costs of cancer care in the US in 2010 were nearly $125 billion, and these costs are predicted to rise to $156 billion in 2020.
These data underscore the need for more research to develop new approaches to cancer prevention and treatment, according to the report.
Its authors call for Congress and the administration to provide the National Institutes of Health, National Cancer Institute, and FDA with annual funding increases.
“We have made spectacular progress against cancer, which has saved the lives of millions of individuals in the United States and around the world,” said Margaret Foti, PhD, MD, chief executive officer of the AACR.
“However, without increased federal funding for cancer research, we will not be able to realize the promise of recent discoveries and technological advances.”

Photo courtesy of the FDA
Despite recent progress in the fight against cancers, these diseases continue to exert “an immense toll” in the US, according to the AACR Cancer Progress Report 2015.
The report highlights the recent approval by the US Food and Drug Administration (FDA) of several anticancer therapies, a vaccine, and 2 diagnostic aids.
But the report also includes data suggesting that cancer cases, and costs related to cancer care, are on the rise.
The report states that, between Aug. 1, 2014, and July 31, 2015, the FDA approved 9 anticancer therapies, either for the first time or for new indications.
During the same period, the FDA approved a new cancer vaccine, a new cancer screening test, and a new use for a previously approved imaging agent.
| Cancer-related products approved from Aug. 1, 2014 to July 31, 2015 | |
| Drug | Approved indication |
| bevacizumab (Avastin) | cervical, ovarian, fallopian
tube, and peritoneal cancers |
| blinatumomab (Blincyto) | acute lymphoblastic leukemia |
| denosumab (Xgeva) | potentially lethal complication
of advanced cancers |
| dinutuximab (Unituxin) | neuroblastoma |
| gefitinib (Iressa) | lung cancer |
| ibrutinib (Imbruvica) | Waldenstrom macroglobulinemia |
| lenvatinib (Lenvima) | thyroid cancer |
| nivolumab (Opdivo) | melanoma, lung cancer |
| olaparib (Lynparza) | ovarian cancer |
| palbociclib (Ibrance) | breast cancer |
| panobinostat (Farydak) | multiple myeloma |
| pembrolizumab (Keytruda) | melanoma |
| ramucirumab (Cyramza) | colorectal and lung cancers |
| sonidegib (Odomzo) | skin cancer |
| Imaging agent | Approved indication |
| technetium 99m tilmanocept
(Lymphoseek) |
lymphatic mapping in solid tumors |
| Vaccine | Approved indication |
| human papillomavirus
9-valent vaccine (Gardasil 9) |
cervical, vulvar,
vaginal, and anal cancers |
| Screening test | Approved indication |
| Cologuard (no generic name) | colorectal cancer |
Despite these advances, cancers continue to exert personal and economic tolls, according to the report.
It states that cancer is the number 1 cause of disease-related death among US children. And more than 589,000 people in the US are projected to die from cancer in 2015.
The number of new cancer cases in the US is predicted to rise from 1.7 million in 2015 to 2.4 million in 2035.
In addition, estimates suggest the direct medical costs of cancer care in the US in 2010 were nearly $125 billion, and these costs are predicted to rise to $156 billion in 2020.
These data underscore the need for more research to develop new approaches to cancer prevention and treatment, according to the report.
Its authors call for Congress and the administration to provide the National Institutes of Health, National Cancer Institute, and FDA with annual funding increases.
“We have made spectacular progress against cancer, which has saved the lives of millions of individuals in the United States and around the world,” said Margaret Foti, PhD, MD, chief executive officer of the AACR.
“However, without increased federal funding for cancer research, we will not be able to realize the promise of recent discoveries and technological advances.”
Blood cancer drugs set to be removed from CDF

Photo courtesy of CDC
England’s National Health Service (NHS) plans to remove several drugs used to treat hematologic malignancies from the Cancer Drugs Fund (CDF).
The plan is that, as of November 4, 2015, pomalidomide, lenalidomide, ibrutinib, dasatinib, brentuximab, bosutinib, and bendamustine will no longer be funded via the CDF for certain indications.
Ofatumumab was removed from the CDF list yesterday but is now available through the NHS.
Drugs used to treat solid tumor malignancies are set to be de-funded through CDF in November as well.
However, the NHS said the proposal to remove a drug from the CDF is not necessarily a final decision.
In cases where a drug offers enough clinical benefit, the pharmaceutical company developing that drug has the opportunity to reduce the price they are asking the NHS to pay to ensure that it achieves a satisfactory level of value for money. The NHS said a number of such negotiations are underway.
In addition, patients who are currently receiving the drugs set to be removed from the CDF will continue to have access to those drugs.
About the CDF and the NHS
The CDF—set up in 2010 and currently due to run until March 2016—is money the government has set aside to pay for cancer drugs that haven’t been approved by the National Institute for Health and Care Excellence (NICE) and aren’t available within the NHS in England. Most cancer drugs are routinely funded outside of the CDF.
NHS England and NICE are planning to consult on a proposed new system for commissioning cancer drugs. The NHS said the new system will be designed to provide the agency with a more systematic approach to getting the best price for cancer drugs.
Reason for drug removals
The NHS previously increased the budget for the CDF from £200 million in 2013/14, to £280 million in 2014/15, and £340 million from April 2015. This represents a total increase of 70% since August 2014.
However, current projections suggest that spending would rise to around £410 million for this year, an over-spend of £70 million, in the absence of further prioritization. The NHS said this money could be used for other aspects of cancer treatment or NHS services for other patient groups.
Therefore, some drugs are set to be removed from the CDF. The NHS said all decisions on drugs to be maintained in the CDF were based on the advice of clinicians, the best available evidence, and the cost of the treatment.
“There is no escaping the fact that we face a difficult set of choices, but it is our duty to ensure we get maximum value from every penny available on behalf of patients,” said Peter Clark, chair of the CDF.
“We must ensure we invest in those treatments that offer the most benefit, based on rigorous evidence-based clinical analysis and an assessment of the cost of those treatments.”
While de-funding certain drugs will reduce costs, the CDF is not expected to be back on budget this financial year. The NHS does expect the CDF will be operating within its budget during 2016/17.
Blood cancer drugs to be removed
The following drugs are currently on the CDF list for the following indications, but they are set to be de-listed on November 4, 2015.
Bendamustine
For the treatment of chronic lymphocytic leukemia (CLL) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- CLL (not licensed in this indication)
- Second-line indication, third-line indication, or fourth-line indication
- To be used within the treating Trust’s governance framework, as bendamustine is not licensed in this indication
For the treatment of relapsed mantle cell lymphoma (MCL) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- MCL
- Option for second- or subsequent-line chemotherapy
- No previous treatment with bendamustine
- To be used within the treating Trust’s governance framework, as bendamustine is not licensed in this indication
*Bendamustine will remain on the CDF for other indications.
Bosutinib
For the treatment of refractory, chronic phase chronic myeloid leukemia (CML) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Chronic phase CML
- Refractory to nilotinib or dasatinib (if dasatinib accessed via a clinical trial or via its current approved CDF indication)
For the treatment of refractory, accelerated phase CML where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Accelerated phase CML
- Refractory to nilotinib or dasatinib (if dasatinib accessed via a clinical trial or via its current approved CDF indication)
- Significant intolerance to nilotinib (grade 3 or 4 events)
For the treatment of accelerated phase CML where there is intolerance of treatments and where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Accelerated phase CML
- Significant intolerance to dasatinib (grade 3 or 4 adverse events; if dasatinib accessed via its current approved CDF indication)
- Significant intolerance to nilotinib (grade 3 or 4 events)
*Bosutinib will still be available through the CDF for patients with chronic phase CML that is intolerant of other treatments.
Brentuximab
For the treatment of refractory, systemic anaplastic lymphoma where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Relapsed or refractory systemic anaplastic large-cell lymphoma
For the treatment of relapsed or refractory CD30+ Hodgkin lymphoma where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Relapsed or refractory CD30+ Hodgkin lymphoma
- Following autologous stem cell transplant or following at least 2 prior therapies when autologous stem cell transplant or multi-agent chemotherapy is not an option
Dasatinib
For the treatment of Philadelphia-chromosome-positive (Ph+) acute lymphoblastic leukemia where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Refractory or significant intolerance or resistance to prior therapy including imatinib (grade 3 or 4 adverse events)
- Second-line indication or third-line indication
*Dasatinib will still be available for chronic phase and accelerated phase CML.
Ibrutinib
For the treatment of relapsed/refractory CLL where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Confirmed CLL
- Must have received at least 1 prior therapy for CLL
- Considered not appropriate for treatment or retreatment with purine-analogue-based therapy due to:
- Failure to respond to chemo-immunotherapy or
- A progression-free interval of less than 3 years or
- Age of 70 years or more or
- Age of 65 years or more plus the presence of comorbidities or
- A 17p or TP53 deletion
- ECOG performance status of 0-2
- A neutrophil count of ≥0.75 x 10⁹/L
- A platelet count of ≥30 x 10⁹/L
- Patient not on warfarin or CYP3A4/5 inhibitors
- No prior treatment with idelalisib
For the treatment of relapsed/refractory MCL where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Confirmed MCL with cyclin D1 overexpression or translocation breakpoints at t(11;14)
- Failure to achieve at least partial response with, or documented disease progression disease after, the most recent treatment regimen
- ECOG performance status of 0-2
- At least 1 but no more than 5 previous lines of treatment
Lenalidomide
For the second-line treatment of multiple myeloma (MM) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- MM
- Second-line indication
- Contraindication to bortezomib or previously received bortezomib in the first-line setting
*Lenalidomide will still be available for patients with myelodysplastic syndromes with 5q deletion.
Pomalidomide
For the treatment of relapsed and refractory MM where the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically
- MM
- Performance status of 0-2
- Previously received treatment with adequate trials of at least all of the following options of therapy: bortezomib, lenalidomide, and alkylating agents
- Failed treatment with bortezomib or lenalidomide, as defined by: progression on or before 60 days of treatment, progressive disease 6 months or less after achieving a partial response, or intolerance to bortezomib
- Refractory disease to previous treatment
- No resistance to high-dose dexamethasone used in the last line of therapy
- No peripheral neuropathy of grade 2 or more
A complete list of proposed changes to the CDF, as well as the drugs that were de-listed on March 12, 2015, is available on the NHS website.

Photo courtesy of CDC
England’s National Health Service (NHS) plans to remove several drugs used to treat hematologic malignancies from the Cancer Drugs Fund (CDF).
The plan is that, as of November 4, 2015, pomalidomide, lenalidomide, ibrutinib, dasatinib, brentuximab, bosutinib, and bendamustine will no longer be funded via the CDF for certain indications.
Ofatumumab was removed from the CDF list yesterday but is now available through the NHS.
Drugs used to treat solid tumor malignancies are set to be de-funded through CDF in November as well.
However, the NHS said the proposal to remove a drug from the CDF is not necessarily a final decision.
In cases where a drug offers enough clinical benefit, the pharmaceutical company developing that drug has the opportunity to reduce the price they are asking the NHS to pay to ensure that it achieves a satisfactory level of value for money. The NHS said a number of such negotiations are underway.
In addition, patients who are currently receiving the drugs set to be removed from the CDF will continue to have access to those drugs.
About the CDF and the NHS
The CDF—set up in 2010 and currently due to run until March 2016—is money the government has set aside to pay for cancer drugs that haven’t been approved by the National Institute for Health and Care Excellence (NICE) and aren’t available within the NHS in England. Most cancer drugs are routinely funded outside of the CDF.
NHS England and NICE are planning to consult on a proposed new system for commissioning cancer drugs. The NHS said the new system will be designed to provide the agency with a more systematic approach to getting the best price for cancer drugs.
Reason for drug removals
The NHS previously increased the budget for the CDF from £200 million in 2013/14, to £280 million in 2014/15, and £340 million from April 2015. This represents a total increase of 70% since August 2014.
However, current projections suggest that spending would rise to around £410 million for this year, an over-spend of £70 million, in the absence of further prioritization. The NHS said this money could be used for other aspects of cancer treatment or NHS services for other patient groups.
Therefore, some drugs are set to be removed from the CDF. The NHS said all decisions on drugs to be maintained in the CDF were based on the advice of clinicians, the best available evidence, and the cost of the treatment.
“There is no escaping the fact that we face a difficult set of choices, but it is our duty to ensure we get maximum value from every penny available on behalf of patients,” said Peter Clark, chair of the CDF.
“We must ensure we invest in those treatments that offer the most benefit, based on rigorous evidence-based clinical analysis and an assessment of the cost of those treatments.”
While de-funding certain drugs will reduce costs, the CDF is not expected to be back on budget this financial year. The NHS does expect the CDF will be operating within its budget during 2016/17.
Blood cancer drugs to be removed
The following drugs are currently on the CDF list for the following indications, but they are set to be de-listed on November 4, 2015.
Bendamustine
For the treatment of chronic lymphocytic leukemia (CLL) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- CLL (not licensed in this indication)
- Second-line indication, third-line indication, or fourth-line indication
- To be used within the treating Trust’s governance framework, as bendamustine is not licensed in this indication
For the treatment of relapsed mantle cell lymphoma (MCL) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- MCL
- Option for second- or subsequent-line chemotherapy
- No previous treatment with bendamustine
- To be used within the treating Trust’s governance framework, as bendamustine is not licensed in this indication
*Bendamustine will remain on the CDF for other indications.
Bosutinib
For the treatment of refractory, chronic phase chronic myeloid leukemia (CML) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Chronic phase CML
- Refractory to nilotinib or dasatinib (if dasatinib accessed via a clinical trial or via its current approved CDF indication)
For the treatment of refractory, accelerated phase CML where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Accelerated phase CML
- Refractory to nilotinib or dasatinib (if dasatinib accessed via a clinical trial or via its current approved CDF indication)
- Significant intolerance to nilotinib (grade 3 or 4 events)
For the treatment of accelerated phase CML where there is intolerance of treatments and where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Accelerated phase CML
- Significant intolerance to dasatinib (grade 3 or 4 adverse events; if dasatinib accessed via its current approved CDF indication)
- Significant intolerance to nilotinib (grade 3 or 4 events)
*Bosutinib will still be available through the CDF for patients with chronic phase CML that is intolerant of other treatments.
Brentuximab
For the treatment of refractory, systemic anaplastic lymphoma where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Relapsed or refractory systemic anaplastic large-cell lymphoma
For the treatment of relapsed or refractory CD30+ Hodgkin lymphoma where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Relapsed or refractory CD30+ Hodgkin lymphoma
- Following autologous stem cell transplant or following at least 2 prior therapies when autologous stem cell transplant or multi-agent chemotherapy is not an option
Dasatinib
For the treatment of Philadelphia-chromosome-positive (Ph+) acute lymphoblastic leukemia where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Refractory or significant intolerance or resistance to prior therapy including imatinib (grade 3 or 4 adverse events)
- Second-line indication or third-line indication
*Dasatinib will still be available for chronic phase and accelerated phase CML.
Ibrutinib
For the treatment of relapsed/refractory CLL where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Confirmed CLL
- Must have received at least 1 prior therapy for CLL
- Considered not appropriate for treatment or retreatment with purine-analogue-based therapy due to:
- Failure to respond to chemo-immunotherapy or
- A progression-free interval of less than 3 years or
- Age of 70 years or more or
- Age of 65 years or more plus the presence of comorbidities or
- A 17p or TP53 deletion
- ECOG performance status of 0-2
- A neutrophil count of ≥0.75 x 10⁹/L
- A platelet count of ≥30 x 10⁹/L
- Patient not on warfarin or CYP3A4/5 inhibitors
- No prior treatment with idelalisib
For the treatment of relapsed/refractory MCL where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Confirmed MCL with cyclin D1 overexpression or translocation breakpoints at t(11;14)
- Failure to achieve at least partial response with, or documented disease progression disease after, the most recent treatment regimen
- ECOG performance status of 0-2
- At least 1 but no more than 5 previous lines of treatment
Lenalidomide
For the second-line treatment of multiple myeloma (MM) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- MM
- Second-line indication
- Contraindication to bortezomib or previously received bortezomib in the first-line setting
*Lenalidomide will still be available for patients with myelodysplastic syndromes with 5q deletion.
Pomalidomide
For the treatment of relapsed and refractory MM where the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically
- MM
- Performance status of 0-2
- Previously received treatment with adequate trials of at least all of the following options of therapy: bortezomib, lenalidomide, and alkylating agents
- Failed treatment with bortezomib or lenalidomide, as defined by: progression on or before 60 days of treatment, progressive disease 6 months or less after achieving a partial response, or intolerance to bortezomib
- Refractory disease to previous treatment
- No resistance to high-dose dexamethasone used in the last line of therapy
- No peripheral neuropathy of grade 2 or more
A complete list of proposed changes to the CDF, as well as the drugs that were de-listed on March 12, 2015, is available on the NHS website.

Photo courtesy of CDC
England’s National Health Service (NHS) plans to remove several drugs used to treat hematologic malignancies from the Cancer Drugs Fund (CDF).
The plan is that, as of November 4, 2015, pomalidomide, lenalidomide, ibrutinib, dasatinib, brentuximab, bosutinib, and bendamustine will no longer be funded via the CDF for certain indications.
Ofatumumab was removed from the CDF list yesterday but is now available through the NHS.
Drugs used to treat solid tumor malignancies are set to be de-funded through CDF in November as well.
However, the NHS said the proposal to remove a drug from the CDF is not necessarily a final decision.
In cases where a drug offers enough clinical benefit, the pharmaceutical company developing that drug has the opportunity to reduce the price they are asking the NHS to pay to ensure that it achieves a satisfactory level of value for money. The NHS said a number of such negotiations are underway.
In addition, patients who are currently receiving the drugs set to be removed from the CDF will continue to have access to those drugs.
About the CDF and the NHS
The CDF—set up in 2010 and currently due to run until March 2016—is money the government has set aside to pay for cancer drugs that haven’t been approved by the National Institute for Health and Care Excellence (NICE) and aren’t available within the NHS in England. Most cancer drugs are routinely funded outside of the CDF.
NHS England and NICE are planning to consult on a proposed new system for commissioning cancer drugs. The NHS said the new system will be designed to provide the agency with a more systematic approach to getting the best price for cancer drugs.
Reason for drug removals
The NHS previously increased the budget for the CDF from £200 million in 2013/14, to £280 million in 2014/15, and £340 million from April 2015. This represents a total increase of 70% since August 2014.
However, current projections suggest that spending would rise to around £410 million for this year, an over-spend of £70 million, in the absence of further prioritization. The NHS said this money could be used for other aspects of cancer treatment or NHS services for other patient groups.
Therefore, some drugs are set to be removed from the CDF. The NHS said all decisions on drugs to be maintained in the CDF were based on the advice of clinicians, the best available evidence, and the cost of the treatment.
“There is no escaping the fact that we face a difficult set of choices, but it is our duty to ensure we get maximum value from every penny available on behalf of patients,” said Peter Clark, chair of the CDF.
“We must ensure we invest in those treatments that offer the most benefit, based on rigorous evidence-based clinical analysis and an assessment of the cost of those treatments.”
While de-funding certain drugs will reduce costs, the CDF is not expected to be back on budget this financial year. The NHS does expect the CDF will be operating within its budget during 2016/17.
Blood cancer drugs to be removed
The following drugs are currently on the CDF list for the following indications, but they are set to be de-listed on November 4, 2015.
Bendamustine
For the treatment of chronic lymphocytic leukemia (CLL) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- CLL (not licensed in this indication)
- Second-line indication, third-line indication, or fourth-line indication
- To be used within the treating Trust’s governance framework, as bendamustine is not licensed in this indication
For the treatment of relapsed mantle cell lymphoma (MCL) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- MCL
- Option for second- or subsequent-line chemotherapy
- No previous treatment with bendamustine
- To be used within the treating Trust’s governance framework, as bendamustine is not licensed in this indication
*Bendamustine will remain on the CDF for other indications.
Bosutinib
For the treatment of refractory, chronic phase chronic myeloid leukemia (CML) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Chronic phase CML
- Refractory to nilotinib or dasatinib (if dasatinib accessed via a clinical trial or via its current approved CDF indication)
For the treatment of refractory, accelerated phase CML where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Accelerated phase CML
- Refractory to nilotinib or dasatinib (if dasatinib accessed via a clinical trial or via its current approved CDF indication)
- Significant intolerance to nilotinib (grade 3 or 4 events)
For the treatment of accelerated phase CML where there is intolerance of treatments and where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Accelerated phase CML
- Significant intolerance to dasatinib (grade 3 or 4 adverse events; if dasatinib accessed via its current approved CDF indication)
- Significant intolerance to nilotinib (grade 3 or 4 events)
*Bosutinib will still be available through the CDF for patients with chronic phase CML that is intolerant of other treatments.
Brentuximab
For the treatment of refractory, systemic anaplastic lymphoma where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Relapsed or refractory systemic anaplastic large-cell lymphoma
For the treatment of relapsed or refractory CD30+ Hodgkin lymphoma where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Relapsed or refractory CD30+ Hodgkin lymphoma
- Following autologous stem cell transplant or following at least 2 prior therapies when autologous stem cell transplant or multi-agent chemotherapy is not an option
Dasatinib
For the treatment of Philadelphia-chromosome-positive (Ph+) acute lymphoblastic leukemia where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Refractory or significant intolerance or resistance to prior therapy including imatinib (grade 3 or 4 adverse events)
- Second-line indication or third-line indication
*Dasatinib will still be available for chronic phase and accelerated phase CML.
Ibrutinib
For the treatment of relapsed/refractory CLL where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Confirmed CLL
- Must have received at least 1 prior therapy for CLL
- Considered not appropriate for treatment or retreatment with purine-analogue-based therapy due to:
- Failure to respond to chemo-immunotherapy or
- A progression-free interval of less than 3 years or
- Age of 70 years or more or
- Age of 65 years or more plus the presence of comorbidities or
- A 17p or TP53 deletion
- ECOG performance status of 0-2
- A neutrophil count of ≥0.75 x 10⁹/L
- A platelet count of ≥30 x 10⁹/L
- Patient not on warfarin or CYP3A4/5 inhibitors
- No prior treatment with idelalisib
For the treatment of relapsed/refractory MCL where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- Confirmed MCL with cyclin D1 overexpression or translocation breakpoints at t(11;14)
- Failure to achieve at least partial response with, or documented disease progression disease after, the most recent treatment regimen
- ECOG performance status of 0-2
- At least 1 but no more than 5 previous lines of treatment
Lenalidomide
For the second-line treatment of multiple myeloma (MM) where all the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically trained and accredited in the use of systemic anticancer therapy
- MM
- Second-line indication
- Contraindication to bortezomib or previously received bortezomib in the first-line setting
*Lenalidomide will still be available for patients with myelodysplastic syndromes with 5q deletion.
Pomalidomide
For the treatment of relapsed and refractory MM where the following criteria are met:
- Application made by and first cycle of systemic anticancer therapy to be prescribed by a consultant specialist specifically
- MM
- Performance status of 0-2
- Previously received treatment with adequate trials of at least all of the following options of therapy: bortezomib, lenalidomide, and alkylating agents
- Failed treatment with bortezomib or lenalidomide, as defined by: progression on or before 60 days of treatment, progressive disease 6 months or less after achieving a partial response, or intolerance to bortezomib
- Refractory disease to previous treatment
- No resistance to high-dose dexamethasone used in the last line of therapy
- No peripheral neuropathy of grade 2 or more
A complete list of proposed changes to the CDF, as well as the drugs that were de-listed on March 12, 2015, is available on the NHS website.