User login
Team identifies potential target for T-ALL therapy

New research suggests T-cell acute lymphoblastic leukemia (T-ALL) cells use the tricarboxylic acid (TCA) cycle to support their growth and survival.
Investigators say the findings could aid the development of therapeutics that can kill T-ALL cells by targeting an enzyme that exists in the TCA cycle—dihydrolipoamide S-succinyltransferase (DLST).
The team described this research in the journal Leukemia.
“Researchers have wrongly assumed that cancer cells do not use the TCA cycle to support their growth,” said study author Hui Feng, MD, PhD, of Boston University Medical Center in Massachusetts.
“Our new findings provide solid evidence that these cancer cells depend on the TCA cycle for their survival. Additionally, we demonstrated the importance of DLST in T-cell leukemia development and have identified a targetable enzyme for T-cell leukemia treatment.”
For this study, the investigators set out to examine the mechanisms underlying MYC-mediated tumorigenesis in T-ALL.
They used a zebrafish model of MYC-induced T-ALL to screen for genes that contribute to disease onset. The results suggested the TCA-cycle enzyme DLST is an important contributor to T-ALL development.
And experiments showed that heterozygous inactivation of DLST significantly delayed disease onset in the zebrafish, apparently without affecting the development of the fish.
Further analysis revealed that inhibiting the activity of DLST could effectively kill human T-ALL cells. Specifically, RNAi knockdown of DLST decreased cell viability and induced apoptosis in human T-ALL cell
lines.
The investigators found that knockdown of DLST disrupted the TCA cycle in the human T-ALL cells. But adding succinate, the downstream TCA-cycle intermediate, to the cells rescued defects in cell viability caused by DLST knockdown.
The investigators said the therapeutic benefit of DLST inhibition may extend to cancers other than T-ALL as well. ![]()

New research suggests T-cell acute lymphoblastic leukemia (T-ALL) cells use the tricarboxylic acid (TCA) cycle to support their growth and survival.
Investigators say the findings could aid the development of therapeutics that can kill T-ALL cells by targeting an enzyme that exists in the TCA cycle—dihydrolipoamide S-succinyltransferase (DLST).
The team described this research in the journal Leukemia.
“Researchers have wrongly assumed that cancer cells do not use the TCA cycle to support their growth,” said study author Hui Feng, MD, PhD, of Boston University Medical Center in Massachusetts.
“Our new findings provide solid evidence that these cancer cells depend on the TCA cycle for their survival. Additionally, we demonstrated the importance of DLST in T-cell leukemia development and have identified a targetable enzyme for T-cell leukemia treatment.”
For this study, the investigators set out to examine the mechanisms underlying MYC-mediated tumorigenesis in T-ALL.
They used a zebrafish model of MYC-induced T-ALL to screen for genes that contribute to disease onset. The results suggested the TCA-cycle enzyme DLST is an important contributor to T-ALL development.
And experiments showed that heterozygous inactivation of DLST significantly delayed disease onset in the zebrafish, apparently without affecting the development of the fish.
Further analysis revealed that inhibiting the activity of DLST could effectively kill human T-ALL cells. Specifically, RNAi knockdown of DLST decreased cell viability and induced apoptosis in human T-ALL cell
lines.
The investigators found that knockdown of DLST disrupted the TCA cycle in the human T-ALL cells. But adding succinate, the downstream TCA-cycle intermediate, to the cells rescued defects in cell viability caused by DLST knockdown.
The investigators said the therapeutic benefit of DLST inhibition may extend to cancers other than T-ALL as well. ![]()

New research suggests T-cell acute lymphoblastic leukemia (T-ALL) cells use the tricarboxylic acid (TCA) cycle to support their growth and survival.
Investigators say the findings could aid the development of therapeutics that can kill T-ALL cells by targeting an enzyme that exists in the TCA cycle—dihydrolipoamide S-succinyltransferase (DLST).
The team described this research in the journal Leukemia.
“Researchers have wrongly assumed that cancer cells do not use the TCA cycle to support their growth,” said study author Hui Feng, MD, PhD, of Boston University Medical Center in Massachusetts.
“Our new findings provide solid evidence that these cancer cells depend on the TCA cycle for their survival. Additionally, we demonstrated the importance of DLST in T-cell leukemia development and have identified a targetable enzyme for T-cell leukemia treatment.”
For this study, the investigators set out to examine the mechanisms underlying MYC-mediated tumorigenesis in T-ALL.
They used a zebrafish model of MYC-induced T-ALL to screen for genes that contribute to disease onset. The results suggested the TCA-cycle enzyme DLST is an important contributor to T-ALL development.
And experiments showed that heterozygous inactivation of DLST significantly delayed disease onset in the zebrafish, apparently without affecting the development of the fish.
Further analysis revealed that inhibiting the activity of DLST could effectively kill human T-ALL cells. Specifically, RNAi knockdown of DLST decreased cell viability and induced apoptosis in human T-ALL cell
lines.
The investigators found that knockdown of DLST disrupted the TCA cycle in the human T-ALL cells. But adding succinate, the downstream TCA-cycle intermediate, to the cells rescued defects in cell viability caused by DLST knockdown.
The investigators said the therapeutic benefit of DLST inhibition may extend to cancers other than T-ALL as well. ![]()
Poverty tied to early relapse in kids with ALL

Photo by Logan Tuttle
A new study suggests children with acute lymphoblastic leukemia (ALL) are more likely to suffer early relapse if they live in high-poverty areas.
All of the children studied received the same treatment, and the rates of relapse were similar regardless of poverty level.
But early relapse was more common among children from poorer areas. These children also had a lower rate of 5-year overall survival, but the difference was not significant.
Kira Bona, MD, of Dana-Farber Cancer Institute in Boston, Massachusetts, and her colleagues reported these results in Pediatric Blood & Cancer.
The team examined outcomes for 575 children, ages 1 to 18, with newly diagnosed ALL who were treated on Dana-Farber Cancer Institute ALL Consortium Protocols at 7 major academic medical centers in the US between 2000 and 2010.
Using US Census Bureau criteria, the investigators defined high-poverty areas as zip codes where 20% or more of residents have incomes below the federal poverty level. For a family of 4, this translates to an annual income of $24,250 or less.
Dr Bona and her colleagues found the overall rates of relapse were similar between children from low-poverty areas and those from high-poverty areas.
However, the timing of relapse differed significantly. Ninety-two percent of children from high-poverty areas who relapsed suffered early relapse (less than 36 months after first achieving complete remission), while 48% of the other children who relapsed did so early (P=0.008).
The 5-year overall survival was 85% for children from high-poverty areas and 92% for children from low-poverty areas. This difference is statistically significant when considered on its own (P=0.02) but not when the analysis is adjusted for other factors (P=0.07).
Still, the investigators said this suggests a possible disparity in survival.
“These children are getting the same best possible care at well-resourced institutions from highly trained clinicians, and we’re still seeing disparities,” Dr Bona said. “In trying to improve cure rates, we, as a field, have focused almost exclusively on biology. If we want to move forward, we also have to look at social determinants.”
Next steps
Dr Bona and her colleagues are undertaking further research designed to delve deeper into the relationship between socioeconomic status and outcomes and to allow for the development of poverty-targeted interventions.
As part of a prospective trial for children with ALL, the researchers will investigate associations between disease outcomes and the socioeconomic status of patients’ families, using a targetable measure of socioeconomic status called material hardship (food, housing, and/or energy insecurity).
The researchers will also investigate possible mechanisms underlying the relationship between socioeconomic status and early relapse, including adherence to oral chemotherapy and delays or dose reductions in chemotherapy due to a child’s underlying health.
In another study, investigators will conduct in-depth interviews with patients’ families, probing their knowledge and experience to pinpoint factors that might explain the disparity in outcomes and identify factors that can be targeted with interventions.
“Doing these next 2 studies is incredibly important,” Dr Bona said. “This study told us that simply providing the current best treatment regimen is not good enough if our goal is to cure every child with cancer.”
“At the same time that we develop new drugs and new treatment protocols, we need to address social determinants of health. Findings from these next studies will help us develop specific interventions to address disparities in outcomes. That’s an amazing opportunity.” ![]()

Photo by Logan Tuttle
A new study suggests children with acute lymphoblastic leukemia (ALL) are more likely to suffer early relapse if they live in high-poverty areas.
All of the children studied received the same treatment, and the rates of relapse were similar regardless of poverty level.
But early relapse was more common among children from poorer areas. These children also had a lower rate of 5-year overall survival, but the difference was not significant.
Kira Bona, MD, of Dana-Farber Cancer Institute in Boston, Massachusetts, and her colleagues reported these results in Pediatric Blood & Cancer.
The team examined outcomes for 575 children, ages 1 to 18, with newly diagnosed ALL who were treated on Dana-Farber Cancer Institute ALL Consortium Protocols at 7 major academic medical centers in the US between 2000 and 2010.
Using US Census Bureau criteria, the investigators defined high-poverty areas as zip codes where 20% or more of residents have incomes below the federal poverty level. For a family of 4, this translates to an annual income of $24,250 or less.
Dr Bona and her colleagues found the overall rates of relapse were similar between children from low-poverty areas and those from high-poverty areas.
However, the timing of relapse differed significantly. Ninety-two percent of children from high-poverty areas who relapsed suffered early relapse (less than 36 months after first achieving complete remission), while 48% of the other children who relapsed did so early (P=0.008).
The 5-year overall survival was 85% for children from high-poverty areas and 92% for children from low-poverty areas. This difference is statistically significant when considered on its own (P=0.02) but not when the analysis is adjusted for other factors (P=0.07).
Still, the investigators said this suggests a possible disparity in survival.
“These children are getting the same best possible care at well-resourced institutions from highly trained clinicians, and we’re still seeing disparities,” Dr Bona said. “In trying to improve cure rates, we, as a field, have focused almost exclusively on biology. If we want to move forward, we also have to look at social determinants.”
Next steps
Dr Bona and her colleagues are undertaking further research designed to delve deeper into the relationship between socioeconomic status and outcomes and to allow for the development of poverty-targeted interventions.
As part of a prospective trial for children with ALL, the researchers will investigate associations between disease outcomes and the socioeconomic status of patients’ families, using a targetable measure of socioeconomic status called material hardship (food, housing, and/or energy insecurity).
The researchers will also investigate possible mechanisms underlying the relationship between socioeconomic status and early relapse, including adherence to oral chemotherapy and delays or dose reductions in chemotherapy due to a child’s underlying health.
In another study, investigators will conduct in-depth interviews with patients’ families, probing their knowledge and experience to pinpoint factors that might explain the disparity in outcomes and identify factors that can be targeted with interventions.
“Doing these next 2 studies is incredibly important,” Dr Bona said. “This study told us that simply providing the current best treatment regimen is not good enough if our goal is to cure every child with cancer.”
“At the same time that we develop new drugs and new treatment protocols, we need to address social determinants of health. Findings from these next studies will help us develop specific interventions to address disparities in outcomes. That’s an amazing opportunity.” ![]()

Photo by Logan Tuttle
A new study suggests children with acute lymphoblastic leukemia (ALL) are more likely to suffer early relapse if they live in high-poverty areas.
All of the children studied received the same treatment, and the rates of relapse were similar regardless of poverty level.
But early relapse was more common among children from poorer areas. These children also had a lower rate of 5-year overall survival, but the difference was not significant.
Kira Bona, MD, of Dana-Farber Cancer Institute in Boston, Massachusetts, and her colleagues reported these results in Pediatric Blood & Cancer.
The team examined outcomes for 575 children, ages 1 to 18, with newly diagnosed ALL who were treated on Dana-Farber Cancer Institute ALL Consortium Protocols at 7 major academic medical centers in the US between 2000 and 2010.
Using US Census Bureau criteria, the investigators defined high-poverty areas as zip codes where 20% or more of residents have incomes below the federal poverty level. For a family of 4, this translates to an annual income of $24,250 or less.
Dr Bona and her colleagues found the overall rates of relapse were similar between children from low-poverty areas and those from high-poverty areas.
However, the timing of relapse differed significantly. Ninety-two percent of children from high-poverty areas who relapsed suffered early relapse (less than 36 months after first achieving complete remission), while 48% of the other children who relapsed did so early (P=0.008).
The 5-year overall survival was 85% for children from high-poverty areas and 92% for children from low-poverty areas. This difference is statistically significant when considered on its own (P=0.02) but not when the analysis is adjusted for other factors (P=0.07).
Still, the investigators said this suggests a possible disparity in survival.
“These children are getting the same best possible care at well-resourced institutions from highly trained clinicians, and we’re still seeing disparities,” Dr Bona said. “In trying to improve cure rates, we, as a field, have focused almost exclusively on biology. If we want to move forward, we also have to look at social determinants.”
Next steps
Dr Bona and her colleagues are undertaking further research designed to delve deeper into the relationship between socioeconomic status and outcomes and to allow for the development of poverty-targeted interventions.
As part of a prospective trial for children with ALL, the researchers will investigate associations between disease outcomes and the socioeconomic status of patients’ families, using a targetable measure of socioeconomic status called material hardship (food, housing, and/or energy insecurity).
The researchers will also investigate possible mechanisms underlying the relationship between socioeconomic status and early relapse, including adherence to oral chemotherapy and delays or dose reductions in chemotherapy due to a child’s underlying health.
In another study, investigators will conduct in-depth interviews with patients’ families, probing their knowledge and experience to pinpoint factors that might explain the disparity in outcomes and identify factors that can be targeted with interventions.
“Doing these next 2 studies is incredibly important,” Dr Bona said. “This study told us that simply providing the current best treatment regimen is not good enough if our goal is to cure every child with cancer.”
“At the same time that we develop new drugs and new treatment protocols, we need to address social determinants of health. Findings from these next studies will help us develop specific interventions to address disparities in outcomes. That’s an amazing opportunity.” ![]()
Targeting EZH2 to treat ETP-ALL

The gene EZH2 is a driver of, and potential therapeutic target for, early T-cell precursor acute lymphoblastic leukemia (ETP-ALL), according to a new study.
A previous study, published in Nature in 2012, suggested that nearly half of ETP-ALLs have inactivating alterations in EZH2.
Loss of EZH2 function can inactivate Polycomb repressive complex 2 (PRC2), but it was not clear how PRC2 loss-of-function mutations would aid leukemia growth.
The new study, published in Cell Reports, provides some insight.
Tobias Neff, MD, of the University of Colorado Denver in Aurora, and his colleagues developed a mouse model of NRASQ61K-driven leukemia that recapitulates phenotypic and transcriptional features of ETP-ALL.
Experiments with this model revealed that inactivation of EZH2 helps accelerate leukemia development and enhances a stem-cell-related transcriptional program.
“We have 2 major features of [ETP-ALL]—stem-like cells and increased growth—and, now, we show an actor implicated in both—namely, EZH2/PRC2,” Dr Neff said.
“How exactly the stem-cell-like gene expression profile contributes to the aggressiveness of ETP-ALL is unknown, but we’ve known that these stem-like cells are associated with poor prognosis in acute leukemia.”
The researchers also found that EZH2 inactivation resulted in increased activation of STAT3 by tyrosine 705 phosphorylation. This led them to wonder whether the JAK/STAT pathway might be important in their ETP-ALL model.
The team tested the JAK1/2 inhibitor ruxolitinib in NRASQ61K cells with EZH2 deletion and observed inhibition of cell growth.
“Ruxolitinib is unlikely to treat the disease by itself, but this model will help us test possible drug combinations that could eventually benefit ETP-ALL patients,” Dr Neff said.
He and his colleagues also plan to test the activity of different drugs against other cell types with inactivated EZH2.
“In addition to our specific finding in this disease, we are excited to now have a model that allows us to explore consequences of EZH2 inactivation that may enrich our understanding of a number of other conditions with a similar set of genetic changes,” Dr Neff said.
He and his colleagues noted that EZH2 is known to be inactivated in myelodysplastic syndromes, myeloproliferative neoplasms, and other hematologic malignancies. ![]()

The gene EZH2 is a driver of, and potential therapeutic target for, early T-cell precursor acute lymphoblastic leukemia (ETP-ALL), according to a new study.
A previous study, published in Nature in 2012, suggested that nearly half of ETP-ALLs have inactivating alterations in EZH2.
Loss of EZH2 function can inactivate Polycomb repressive complex 2 (PRC2), but it was not clear how PRC2 loss-of-function mutations would aid leukemia growth.
The new study, published in Cell Reports, provides some insight.
Tobias Neff, MD, of the University of Colorado Denver in Aurora, and his colleagues developed a mouse model of NRASQ61K-driven leukemia that recapitulates phenotypic and transcriptional features of ETP-ALL.
Experiments with this model revealed that inactivation of EZH2 helps accelerate leukemia development and enhances a stem-cell-related transcriptional program.
“We have 2 major features of [ETP-ALL]—stem-like cells and increased growth—and, now, we show an actor implicated in both—namely, EZH2/PRC2,” Dr Neff said.
“How exactly the stem-cell-like gene expression profile contributes to the aggressiveness of ETP-ALL is unknown, but we’ve known that these stem-like cells are associated with poor prognosis in acute leukemia.”
The researchers also found that EZH2 inactivation resulted in increased activation of STAT3 by tyrosine 705 phosphorylation. This led them to wonder whether the JAK/STAT pathway might be important in their ETP-ALL model.
The team tested the JAK1/2 inhibitor ruxolitinib in NRASQ61K cells with EZH2 deletion and observed inhibition of cell growth.
“Ruxolitinib is unlikely to treat the disease by itself, but this model will help us test possible drug combinations that could eventually benefit ETP-ALL patients,” Dr Neff said.
He and his colleagues also plan to test the activity of different drugs against other cell types with inactivated EZH2.
“In addition to our specific finding in this disease, we are excited to now have a model that allows us to explore consequences of EZH2 inactivation that may enrich our understanding of a number of other conditions with a similar set of genetic changes,” Dr Neff said.
He and his colleagues noted that EZH2 is known to be inactivated in myelodysplastic syndromes, myeloproliferative neoplasms, and other hematologic malignancies. ![]()

The gene EZH2 is a driver of, and potential therapeutic target for, early T-cell precursor acute lymphoblastic leukemia (ETP-ALL), according to a new study.
A previous study, published in Nature in 2012, suggested that nearly half of ETP-ALLs have inactivating alterations in EZH2.
Loss of EZH2 function can inactivate Polycomb repressive complex 2 (PRC2), but it was not clear how PRC2 loss-of-function mutations would aid leukemia growth.
The new study, published in Cell Reports, provides some insight.
Tobias Neff, MD, of the University of Colorado Denver in Aurora, and his colleagues developed a mouse model of NRASQ61K-driven leukemia that recapitulates phenotypic and transcriptional features of ETP-ALL.
Experiments with this model revealed that inactivation of EZH2 helps accelerate leukemia development and enhances a stem-cell-related transcriptional program.
“We have 2 major features of [ETP-ALL]—stem-like cells and increased growth—and, now, we show an actor implicated in both—namely, EZH2/PRC2,” Dr Neff said.
“How exactly the stem-cell-like gene expression profile contributes to the aggressiveness of ETP-ALL is unknown, but we’ve known that these stem-like cells are associated with poor prognosis in acute leukemia.”
The researchers also found that EZH2 inactivation resulted in increased activation of STAT3 by tyrosine 705 phosphorylation. This led them to wonder whether the JAK/STAT pathway might be important in their ETP-ALL model.
The team tested the JAK1/2 inhibitor ruxolitinib in NRASQ61K cells with EZH2 deletion and observed inhibition of cell growth.
“Ruxolitinib is unlikely to treat the disease by itself, but this model will help us test possible drug combinations that could eventually benefit ETP-ALL patients,” Dr Neff said.
He and his colleagues also plan to test the activity of different drugs against other cell types with inactivated EZH2.
“In addition to our specific finding in this disease, we are excited to now have a model that allows us to explore consequences of EZH2 inactivation that may enrich our understanding of a number of other conditions with a similar set of genetic changes,” Dr Neff said.
He and his colleagues noted that EZH2 is known to be inactivated in myelodysplastic syndromes, myeloproliferative neoplasms, and other hematologic malignancies. ![]()
Gene variants linked to drug intolerance

Photo courtesy of the CDC
New research has revealed inherited genetic variations that may predispose patients to severe toxicity from thiopurines, a class of medications used as anticancer and immunosuppressive drugs.
Investigators identified 4 variations in the NUDT15 gene that alter thiopurine metabolism, leaving patients particularly sensitive to the drugs and at risk for toxicity.
One in 3 Japanese patients in this study carried the variations.
And evidence suggests the variations are common in other populations across Asia and in individuals of Hispanic ethnicity.
Jun J. Yang, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues reported these findings in Nature Genetics.
In 2015, Dr Yang and his colleagues published evidence linking a NUDT15 variant to reduced tolerance of mercaptopurine and reported the variant was more common in patients of East Asian ancestry.
With the current study, the investigators identified 3 additional NUDT15 variants and found that all 4 variants—p.Arg139Cys, p.Arg139His, p.Val18Ile, and p.Val18_Val19insGlyVal—were associated with lower levels of enzymatic activity and imbalance of thiopurine metabolism.
In a group of 270 children with acute lymphoblastic leukemia (ALL), the variants caused a 74.4% to 100% loss of NUDT15 function. They also predicted enzyme activity and mercaptopurine tolerance. In Singapore and Japan, for example, patients with the 2 highest risk variants had the lowest level of enzyme activity.
“These patients had excessive levels of the active drug metabolites per mercaptopurine dose, which suggests we may reduce the drug dose to achieve the level necessary to kill leukemia cells without causing toxicity,” Dr Yang said, adding that the NUDT15 variants have no other known health consequences.
The investigators also checked leukemic cells from 285 children newly diagnosed with ALL and found that patients with NUDT15 variants were more sensitive to thiopurines.
“That suggests we can screen for NUDT15 variants and potentially plan mercaptopurine doses according to each patient’s genotype before the therapy starts,” Dr Yang said. “This way, we hope to avoid toxicity without compromising treatment effectiveness.”
The investigators noted that future studies are needed to determine optimal thiopurine doses for patients with different NUDT15 variants.
Meanwhile, the search continues for variants in NUDT15 or other genes that influence chemotherapy effectiveness and safety. The NUDT15 variants and previously identified TPMT variants could not fully explain why Guatemalan patients in this study tolerated the lowest doses of mercaptopurine. ![]()

Photo courtesy of the CDC
New research has revealed inherited genetic variations that may predispose patients to severe toxicity from thiopurines, a class of medications used as anticancer and immunosuppressive drugs.
Investigators identified 4 variations in the NUDT15 gene that alter thiopurine metabolism, leaving patients particularly sensitive to the drugs and at risk for toxicity.
One in 3 Japanese patients in this study carried the variations.
And evidence suggests the variations are common in other populations across Asia and in individuals of Hispanic ethnicity.
Jun J. Yang, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues reported these findings in Nature Genetics.
In 2015, Dr Yang and his colleagues published evidence linking a NUDT15 variant to reduced tolerance of mercaptopurine and reported the variant was more common in patients of East Asian ancestry.
With the current study, the investigators identified 3 additional NUDT15 variants and found that all 4 variants—p.Arg139Cys, p.Arg139His, p.Val18Ile, and p.Val18_Val19insGlyVal—were associated with lower levels of enzymatic activity and imbalance of thiopurine metabolism.
In a group of 270 children with acute lymphoblastic leukemia (ALL), the variants caused a 74.4% to 100% loss of NUDT15 function. They also predicted enzyme activity and mercaptopurine tolerance. In Singapore and Japan, for example, patients with the 2 highest risk variants had the lowest level of enzyme activity.
“These patients had excessive levels of the active drug metabolites per mercaptopurine dose, which suggests we may reduce the drug dose to achieve the level necessary to kill leukemia cells without causing toxicity,” Dr Yang said, adding that the NUDT15 variants have no other known health consequences.
The investigators also checked leukemic cells from 285 children newly diagnosed with ALL and found that patients with NUDT15 variants were more sensitive to thiopurines.
“That suggests we can screen for NUDT15 variants and potentially plan mercaptopurine doses according to each patient’s genotype before the therapy starts,” Dr Yang said. “This way, we hope to avoid toxicity without compromising treatment effectiveness.”
The investigators noted that future studies are needed to determine optimal thiopurine doses for patients with different NUDT15 variants.
Meanwhile, the search continues for variants in NUDT15 or other genes that influence chemotherapy effectiveness and safety. The NUDT15 variants and previously identified TPMT variants could not fully explain why Guatemalan patients in this study tolerated the lowest doses of mercaptopurine. ![]()

Photo courtesy of the CDC
New research has revealed inherited genetic variations that may predispose patients to severe toxicity from thiopurines, a class of medications used as anticancer and immunosuppressive drugs.
Investigators identified 4 variations in the NUDT15 gene that alter thiopurine metabolism, leaving patients particularly sensitive to the drugs and at risk for toxicity.
One in 3 Japanese patients in this study carried the variations.
And evidence suggests the variations are common in other populations across Asia and in individuals of Hispanic ethnicity.
Jun J. Yang, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues reported these findings in Nature Genetics.
In 2015, Dr Yang and his colleagues published evidence linking a NUDT15 variant to reduced tolerance of mercaptopurine and reported the variant was more common in patients of East Asian ancestry.
With the current study, the investigators identified 3 additional NUDT15 variants and found that all 4 variants—p.Arg139Cys, p.Arg139His, p.Val18Ile, and p.Val18_Val19insGlyVal—were associated with lower levels of enzymatic activity and imbalance of thiopurine metabolism.
In a group of 270 children with acute lymphoblastic leukemia (ALL), the variants caused a 74.4% to 100% loss of NUDT15 function. They also predicted enzyme activity and mercaptopurine tolerance. In Singapore and Japan, for example, patients with the 2 highest risk variants had the lowest level of enzyme activity.
“These patients had excessive levels of the active drug metabolites per mercaptopurine dose, which suggests we may reduce the drug dose to achieve the level necessary to kill leukemia cells without causing toxicity,” Dr Yang said, adding that the NUDT15 variants have no other known health consequences.
The investigators also checked leukemic cells from 285 children newly diagnosed with ALL and found that patients with NUDT15 variants were more sensitive to thiopurines.
“That suggests we can screen for NUDT15 variants and potentially plan mercaptopurine doses according to each patient’s genotype before the therapy starts,” Dr Yang said. “This way, we hope to avoid toxicity without compromising treatment effectiveness.”
The investigators noted that future studies are needed to determine optimal thiopurine doses for patients with different NUDT15 variants.
Meanwhile, the search continues for variants in NUDT15 or other genes that influence chemotherapy effectiveness and safety. The NUDT15 variants and previously identified TPMT variants could not fully explain why Guatemalan patients in this study tolerated the lowest doses of mercaptopurine. ![]()
Chemo linked to long-term learning problems in ALL survivors

Pediatric acute lymphoblastic leukemia (ALL) patients treated with chemotherapy alone are at risk for attention and learning problems that persist after treatment ends, according to a study published in the Journal of Clinical Oncology.
Researchers said this study is the largest assessment to date of neurocognitive outcomes in pediatric ALL survivors treated with intensive chemotherapy alone rather than in combination with cranial radiation.
“This is an important contribution to the literature because the smaller size and design of previous studies made examining the impact of treatment difficult,” said study author Lisa Jacola, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
“The findings underscore the need for neurocognitive and academic screening to be included as part of routine survivorship care for all pediatric ALL survivors.”
The study included patients enrolled in the St. Jude Total Therapy Study XV. They underwent neurocognitive assessments at the beginning of induction (n=142), end of maintenance (n=243), and 2 years after completing treatment (n=211).
The subjects completed standardized tests of overall intelligence, attention, learning, and academic performance. In addition, parents and other caregivers rated the subjects’ attention, learning, and behavior.
Two years after treatment completion, subjects performed as expected for their age (compared to national data) on measures of overall intelligence, learning, and memory. However, study subjects had significant attention deficits and a significantly greater frequency of learning problems (all P≤0.005).
The risk of such problems was greatest for survivors who were less than 5 years old when diagnosed with ALL and for those who received more intensive chemotherapy.
Specifically, the younger subjects had a greater risk of difficulties with attention, learning, working memory, and processing speed (all P≤0.05). And subjects who received higher-intensity, CNS-directed chemotherapy had a greater risk of difficulties in attention, processing speed, and academics (all P≤0.01).
Researchers also found that subjects with attention problems at the end of therapy had lower academic scores 2 years later (all P≤0.05).
“These findings provide additional evidence that neurocognitive functioning has improved in survivors of childhood ALL since cranial irradiation was replaced with intensified chemotherapy,” Dr Jacola said.
“But we also show these young people are at an elevated risk for attention problems that have real-world consequences, particularly for learning and school performance. Attention is a building block for learning, and, in this study, attention difficulties predicted academic problems later. If we know attention problems seen at the end of therapy continue and contribute to academic problems, then our goal is to intervene earlier to reduce or prevent such difficulties.” ![]()

Pediatric acute lymphoblastic leukemia (ALL) patients treated with chemotherapy alone are at risk for attention and learning problems that persist after treatment ends, according to a study published in the Journal of Clinical Oncology.
Researchers said this study is the largest assessment to date of neurocognitive outcomes in pediatric ALL survivors treated with intensive chemotherapy alone rather than in combination with cranial radiation.
“This is an important contribution to the literature because the smaller size and design of previous studies made examining the impact of treatment difficult,” said study author Lisa Jacola, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
“The findings underscore the need for neurocognitive and academic screening to be included as part of routine survivorship care for all pediatric ALL survivors.”
The study included patients enrolled in the St. Jude Total Therapy Study XV. They underwent neurocognitive assessments at the beginning of induction (n=142), end of maintenance (n=243), and 2 years after completing treatment (n=211).
The subjects completed standardized tests of overall intelligence, attention, learning, and academic performance. In addition, parents and other caregivers rated the subjects’ attention, learning, and behavior.
Two years after treatment completion, subjects performed as expected for their age (compared to national data) on measures of overall intelligence, learning, and memory. However, study subjects had significant attention deficits and a significantly greater frequency of learning problems (all P≤0.005).
The risk of such problems was greatest for survivors who were less than 5 years old when diagnosed with ALL and for those who received more intensive chemotherapy.
Specifically, the younger subjects had a greater risk of difficulties with attention, learning, working memory, and processing speed (all P≤0.05). And subjects who received higher-intensity, CNS-directed chemotherapy had a greater risk of difficulties in attention, processing speed, and academics (all P≤0.01).
Researchers also found that subjects with attention problems at the end of therapy had lower academic scores 2 years later (all P≤0.05).
“These findings provide additional evidence that neurocognitive functioning has improved in survivors of childhood ALL since cranial irradiation was replaced with intensified chemotherapy,” Dr Jacola said.
“But we also show these young people are at an elevated risk for attention problems that have real-world consequences, particularly for learning and school performance. Attention is a building block for learning, and, in this study, attention difficulties predicted academic problems later. If we know attention problems seen at the end of therapy continue and contribute to academic problems, then our goal is to intervene earlier to reduce or prevent such difficulties.” ![]()

Pediatric acute lymphoblastic leukemia (ALL) patients treated with chemotherapy alone are at risk for attention and learning problems that persist after treatment ends, according to a study published in the Journal of Clinical Oncology.
Researchers said this study is the largest assessment to date of neurocognitive outcomes in pediatric ALL survivors treated with intensive chemotherapy alone rather than in combination with cranial radiation.
“This is an important contribution to the literature because the smaller size and design of previous studies made examining the impact of treatment difficult,” said study author Lisa Jacola, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
“The findings underscore the need for neurocognitive and academic screening to be included as part of routine survivorship care for all pediatric ALL survivors.”
The study included patients enrolled in the St. Jude Total Therapy Study XV. They underwent neurocognitive assessments at the beginning of induction (n=142), end of maintenance (n=243), and 2 years after completing treatment (n=211).
The subjects completed standardized tests of overall intelligence, attention, learning, and academic performance. In addition, parents and other caregivers rated the subjects’ attention, learning, and behavior.
Two years after treatment completion, subjects performed as expected for their age (compared to national data) on measures of overall intelligence, learning, and memory. However, study subjects had significant attention deficits and a significantly greater frequency of learning problems (all P≤0.005).
The risk of such problems was greatest for survivors who were less than 5 years old when diagnosed with ALL and for those who received more intensive chemotherapy.
Specifically, the younger subjects had a greater risk of difficulties with attention, learning, working memory, and processing speed (all P≤0.05). And subjects who received higher-intensity, CNS-directed chemotherapy had a greater risk of difficulties in attention, processing speed, and academics (all P≤0.01).
Researchers also found that subjects with attention problems at the end of therapy had lower academic scores 2 years later (all P≤0.05).
“These findings provide additional evidence that neurocognitive functioning has improved in survivors of childhood ALL since cranial irradiation was replaced with intensified chemotherapy,” Dr Jacola said.
“But we also show these young people are at an elevated risk for attention problems that have real-world consequences, particularly for learning and school performance. Attention is a building block for learning, and, in this study, attention difficulties predicted academic problems later. If we know attention problems seen at the end of therapy continue and contribute to academic problems, then our goal is to intervene earlier to reduce or prevent such difficulties.” ![]()
Dendritic cells appear to promote T-ALL

Dendritic cells may play a key role in T-cell acute lymphoblastic leukemia (T-ALL), according to research published in PNAS.
Investigators identified tumor-associated dendritic cells that appeared to promote T-ALL growth and survival at primary and metastatic tumor sites in mice.
Analyses of samples from patients with T-ALL suggested dendritic cells are positioned to support T-ALL growth in humans as well.
“It’s only more recently that people have really appreciated that tumors are complex organs in and of themselves, with all of the heterogenous cell types that can talk to each other and promote each other’s survival and proliferation,” said study author Lauren Ehrlich, PhD, of the University of Texas at Austin.
Dr Ehrlich and her colleagues first found that primary T-ALL cells required tumor stroma for survival ex vivo. When T-ALL cells were cultured alone or in wild-type thymic stroma, the cells died off. Only T-ALL cells cultured with tumor-associated stroma survived.
Subsequent experiments suggested it was tumor-associated dendritic cells that spurred T-ALL growth, both for newly developing T-ALL cells and tumors that had spread to distant organs in mouse models. Tissue samples from pediatric T-ALL patients had similar growth environments with abundant dendritic cells.
To determine the mechanism by which dendritic cells support T-ALL, the investigators performed gene expression profiling. They found upregulation of PDGFRB and IGF1R on T-ALL cells, with concomitant expression of their ligands by tumor-associated dendritic cells.
The team said PDGFRB and IGF1R were activated in T-ALL cells ex vivo. And when they cocultured T-ALL cells with tumor-associated dendritic cells, they observed sustained IGF1R activation. However, they did not see this activation when they cocultured T-ALL cells with normal thymic dendritic cells.
Finally, the investigators found that IGF1R signaling was necessary for dendritic cell-mediated T-ALL survival.
The team said this is the first evidence that endogenous tumor-associated dendritic cells supply signals driving T-ALL growth.
“We hope this study will be a catalyst to spur other research groups to further elucidate the roles of dendritic cells in supporting T-ALL,” said study author Todd Triplett, PhD, also of the University of Texas at Austin.
“[T]hat could ultimately lead to the discovery of novel therapeutic targets that are more effective and less toxic than current treatment regimens.” ![]()

Dendritic cells may play a key role in T-cell acute lymphoblastic leukemia (T-ALL), according to research published in PNAS.
Investigators identified tumor-associated dendritic cells that appeared to promote T-ALL growth and survival at primary and metastatic tumor sites in mice.
Analyses of samples from patients with T-ALL suggested dendritic cells are positioned to support T-ALL growth in humans as well.
“It’s only more recently that people have really appreciated that tumors are complex organs in and of themselves, with all of the heterogenous cell types that can talk to each other and promote each other’s survival and proliferation,” said study author Lauren Ehrlich, PhD, of the University of Texas at Austin.
Dr Ehrlich and her colleagues first found that primary T-ALL cells required tumor stroma for survival ex vivo. When T-ALL cells were cultured alone or in wild-type thymic stroma, the cells died off. Only T-ALL cells cultured with tumor-associated stroma survived.
Subsequent experiments suggested it was tumor-associated dendritic cells that spurred T-ALL growth, both for newly developing T-ALL cells and tumors that had spread to distant organs in mouse models. Tissue samples from pediatric T-ALL patients had similar growth environments with abundant dendritic cells.
To determine the mechanism by which dendritic cells support T-ALL, the investigators performed gene expression profiling. They found upregulation of PDGFRB and IGF1R on T-ALL cells, with concomitant expression of their ligands by tumor-associated dendritic cells.
The team said PDGFRB and IGF1R were activated in T-ALL cells ex vivo. And when they cocultured T-ALL cells with tumor-associated dendritic cells, they observed sustained IGF1R activation. However, they did not see this activation when they cocultured T-ALL cells with normal thymic dendritic cells.
Finally, the investigators found that IGF1R signaling was necessary for dendritic cell-mediated T-ALL survival.
The team said this is the first evidence that endogenous tumor-associated dendritic cells supply signals driving T-ALL growth.
“We hope this study will be a catalyst to spur other research groups to further elucidate the roles of dendritic cells in supporting T-ALL,” said study author Todd Triplett, PhD, also of the University of Texas at Austin.
“[T]hat could ultimately lead to the discovery of novel therapeutic targets that are more effective and less toxic than current treatment regimens.” ![]()

Dendritic cells may play a key role in T-cell acute lymphoblastic leukemia (T-ALL), according to research published in PNAS.
Investigators identified tumor-associated dendritic cells that appeared to promote T-ALL growth and survival at primary and metastatic tumor sites in mice.
Analyses of samples from patients with T-ALL suggested dendritic cells are positioned to support T-ALL growth in humans as well.
“It’s only more recently that people have really appreciated that tumors are complex organs in and of themselves, with all of the heterogenous cell types that can talk to each other and promote each other’s survival and proliferation,” said study author Lauren Ehrlich, PhD, of the University of Texas at Austin.
Dr Ehrlich and her colleagues first found that primary T-ALL cells required tumor stroma for survival ex vivo. When T-ALL cells were cultured alone or in wild-type thymic stroma, the cells died off. Only T-ALL cells cultured with tumor-associated stroma survived.
Subsequent experiments suggested it was tumor-associated dendritic cells that spurred T-ALL growth, both for newly developing T-ALL cells and tumors that had spread to distant organs in mouse models. Tissue samples from pediatric T-ALL patients had similar growth environments with abundant dendritic cells.
To determine the mechanism by which dendritic cells support T-ALL, the investigators performed gene expression profiling. They found upregulation of PDGFRB and IGF1R on T-ALL cells, with concomitant expression of their ligands by tumor-associated dendritic cells.
The team said PDGFRB and IGF1R were activated in T-ALL cells ex vivo. And when they cocultured T-ALL cells with tumor-associated dendritic cells, they observed sustained IGF1R activation. However, they did not see this activation when they cocultured T-ALL cells with normal thymic dendritic cells.
Finally, the investigators found that IGF1R signaling was necessary for dendritic cell-mediated T-ALL survival.
The team said this is the first evidence that endogenous tumor-associated dendritic cells supply signals driving T-ALL growth.
“We hope this study will be a catalyst to spur other research groups to further elucidate the roles of dendritic cells in supporting T-ALL,” said study author Todd Triplett, PhD, also of the University of Texas at Austin.
“[T]hat could ultimately lead to the discovery of novel therapeutic targets that are more effective and less toxic than current treatment regimens.” ![]()
New insight into Ph-like ALL could lead to new treatment

Photo courtesy of St. Jude
Children’s Research Hospital
Research published in Cancer Cell appears to explain how the abnormal breakage and rearrangement of chromosomes in white blood cells triggers Philadelphia chromosome-like (Ph-like) acute lymphoblastic leukemia (ALL).
Genomic analysis revealed 4 chromosomal rearrangements that all resulted in a truncated version of the erythropoietin receptor (EPOR) gene and drove white blood cells to proliferate out of control.
“To our knowledge, this is a previously unknown mechanism for leukemia,” said study author Charles Mullighan, MBBS, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
“Our search of cancer genomic data has shown that there are many other examples of chromosomal rearrangements that alter genes’ structure, but this type—where a truncating rearrangement leads to activation—is new.”
Although Dr Mullighan and his colleagues had previously identified an abnormal chromosome rearrangement in Ph-like ALL, little was known about the biological effects of that rearrangement. So they set out to pinpoint those effects by studying human leukemic cells and mouse cells engineered to mimic Ph-like ALL.
The investigators discovered the 4 rearrangements of EPOR, all of which resulted in truncation of the cytoplasmic tail of EPOR at residues similar to those mutated in primary familial congenital polycythemia. The proximal tyrosine essential for receptor activation was preserved, but distal regulatory residues were lost.
The team said these rearrangements resulted in deregulated EPOR expression, hypersensitivity to erythropoietin stimulation, and heightened JAK-STAT activation.
The investigators noted that the rearrangements were present in all of the leukemic cells from patients, which suggests these changes were fundamental to Ph-like ALL development. The team also showed that introducing truncated EPOR in mouse B-cell progenitors gave rise to ALL in mice.
Further investigation revealed that EPOR rearrangements arise early in the development of Ph-like ALL and persist as the disease progresses.
“That finding was important because it suggests that treatments for this leukemia targeting this receptor won’t just impact a subset of the leukemia cells, allowing others to keep proliferating,” said study author Ilaria Iacobucci, PhD, of St. Jude Children’s Research Hospital.
The investigators then found that human leukemic cells with EPOR rearrangements were sensitive to JAK-STAT inhibition via treatment with ruxolitinib.
The team also cited the case of an adult patient treated at MD Anderson Cancer Research Center in Houston, Texas, whose genetic analysis revealed EPOR-rearranged ALL. That patient had not responded significantly to other chemotherapy drugs. But, when given ruxolitinib, the patient showed a major drop in leukemia cells.
In experiments with leukemic cells, the investigators found that ruxolitinib worked synergistically with 3 chemotherapeutic agents—dexamethasone, vincristine, and daunorubicin.
“We think these findings provide a useful road map for planning more accurate testing of combination chemotherapies,” Dr Mullighan said.
“These findings expand the number of ALL patients who should be amenable to precision medicine therapies that add targeted inhibitors to chemotherapy for ALL patents with specific genetic changes in the leukemia cells,” added study author Stephen Hunger, MD, of Children’s Hospital of Philadelphia in Pennsylvania.
Dr Hunger said the Children’s Oncology Group has developed a clinical trial testing this strategy with ruxolitinib, which will begin treating patients in mid-2016. Based on the results of the Cancer Cell research, the trial will include children with ALL and EPOR rearrangements. ![]()

Photo courtesy of St. Jude
Children’s Research Hospital
Research published in Cancer Cell appears to explain how the abnormal breakage and rearrangement of chromosomes in white blood cells triggers Philadelphia chromosome-like (Ph-like) acute lymphoblastic leukemia (ALL).
Genomic analysis revealed 4 chromosomal rearrangements that all resulted in a truncated version of the erythropoietin receptor (EPOR) gene and drove white blood cells to proliferate out of control.
“To our knowledge, this is a previously unknown mechanism for leukemia,” said study author Charles Mullighan, MBBS, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
“Our search of cancer genomic data has shown that there are many other examples of chromosomal rearrangements that alter genes’ structure, but this type—where a truncating rearrangement leads to activation—is new.”
Although Dr Mullighan and his colleagues had previously identified an abnormal chromosome rearrangement in Ph-like ALL, little was known about the biological effects of that rearrangement. So they set out to pinpoint those effects by studying human leukemic cells and mouse cells engineered to mimic Ph-like ALL.
The investigators discovered the 4 rearrangements of EPOR, all of which resulted in truncation of the cytoplasmic tail of EPOR at residues similar to those mutated in primary familial congenital polycythemia. The proximal tyrosine essential for receptor activation was preserved, but distal regulatory residues were lost.
The team said these rearrangements resulted in deregulated EPOR expression, hypersensitivity to erythropoietin stimulation, and heightened JAK-STAT activation.
The investigators noted that the rearrangements were present in all of the leukemic cells from patients, which suggests these changes were fundamental to Ph-like ALL development. The team also showed that introducing truncated EPOR in mouse B-cell progenitors gave rise to ALL in mice.
Further investigation revealed that EPOR rearrangements arise early in the development of Ph-like ALL and persist as the disease progresses.
“That finding was important because it suggests that treatments for this leukemia targeting this receptor won’t just impact a subset of the leukemia cells, allowing others to keep proliferating,” said study author Ilaria Iacobucci, PhD, of St. Jude Children’s Research Hospital.
The investigators then found that human leukemic cells with EPOR rearrangements were sensitive to JAK-STAT inhibition via treatment with ruxolitinib.
The team also cited the case of an adult patient treated at MD Anderson Cancer Research Center in Houston, Texas, whose genetic analysis revealed EPOR-rearranged ALL. That patient had not responded significantly to other chemotherapy drugs. But, when given ruxolitinib, the patient showed a major drop in leukemia cells.
In experiments with leukemic cells, the investigators found that ruxolitinib worked synergistically with 3 chemotherapeutic agents—dexamethasone, vincristine, and daunorubicin.
“We think these findings provide a useful road map for planning more accurate testing of combination chemotherapies,” Dr Mullighan said.
“These findings expand the number of ALL patients who should be amenable to precision medicine therapies that add targeted inhibitors to chemotherapy for ALL patents with specific genetic changes in the leukemia cells,” added study author Stephen Hunger, MD, of Children’s Hospital of Philadelphia in Pennsylvania.
Dr Hunger said the Children’s Oncology Group has developed a clinical trial testing this strategy with ruxolitinib, which will begin treating patients in mid-2016. Based on the results of the Cancer Cell research, the trial will include children with ALL and EPOR rearrangements. ![]()

Photo courtesy of St. Jude
Children’s Research Hospital
Research published in Cancer Cell appears to explain how the abnormal breakage and rearrangement of chromosomes in white blood cells triggers Philadelphia chromosome-like (Ph-like) acute lymphoblastic leukemia (ALL).
Genomic analysis revealed 4 chromosomal rearrangements that all resulted in a truncated version of the erythropoietin receptor (EPOR) gene and drove white blood cells to proliferate out of control.
“To our knowledge, this is a previously unknown mechanism for leukemia,” said study author Charles Mullighan, MBBS, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
“Our search of cancer genomic data has shown that there are many other examples of chromosomal rearrangements that alter genes’ structure, but this type—where a truncating rearrangement leads to activation—is new.”
Although Dr Mullighan and his colleagues had previously identified an abnormal chromosome rearrangement in Ph-like ALL, little was known about the biological effects of that rearrangement. So they set out to pinpoint those effects by studying human leukemic cells and mouse cells engineered to mimic Ph-like ALL.
The investigators discovered the 4 rearrangements of EPOR, all of which resulted in truncation of the cytoplasmic tail of EPOR at residues similar to those mutated in primary familial congenital polycythemia. The proximal tyrosine essential for receptor activation was preserved, but distal regulatory residues were lost.
The team said these rearrangements resulted in deregulated EPOR expression, hypersensitivity to erythropoietin stimulation, and heightened JAK-STAT activation.
The investigators noted that the rearrangements were present in all of the leukemic cells from patients, which suggests these changes were fundamental to Ph-like ALL development. The team also showed that introducing truncated EPOR in mouse B-cell progenitors gave rise to ALL in mice.
Further investigation revealed that EPOR rearrangements arise early in the development of Ph-like ALL and persist as the disease progresses.
“That finding was important because it suggests that treatments for this leukemia targeting this receptor won’t just impact a subset of the leukemia cells, allowing others to keep proliferating,” said study author Ilaria Iacobucci, PhD, of St. Jude Children’s Research Hospital.
The investigators then found that human leukemic cells with EPOR rearrangements were sensitive to JAK-STAT inhibition via treatment with ruxolitinib.
The team also cited the case of an adult patient treated at MD Anderson Cancer Research Center in Houston, Texas, whose genetic analysis revealed EPOR-rearranged ALL. That patient had not responded significantly to other chemotherapy drugs. But, when given ruxolitinib, the patient showed a major drop in leukemia cells.
In experiments with leukemic cells, the investigators found that ruxolitinib worked synergistically with 3 chemotherapeutic agents—dexamethasone, vincristine, and daunorubicin.
“We think these findings provide a useful road map for planning more accurate testing of combination chemotherapies,” Dr Mullighan said.
“These findings expand the number of ALL patients who should be amenable to precision medicine therapies that add targeted inhibitors to chemotherapy for ALL patents with specific genetic changes in the leukemia cells,” added study author Stephen Hunger, MD, of Children’s Hospital of Philadelphia in Pennsylvania.
Dr Hunger said the Children’s Oncology Group has developed a clinical trial testing this strategy with ruxolitinib, which will begin treating patients in mid-2016. Based on the results of the Cancer Cell research, the trial will include children with ALL and EPOR rearrangements.
Survivors of childhood ALL show significant attention problems
Survivors of childhood acute lymphoblastic leukemia (ALL) who underwent CNS-directed chemotherapy show significant attention problems that impair their functioning 2 years later, according to a report published online Feb. 8 in the Journal of Clinical Oncology.
The attention problems were isolated from intelligence and academic performance, but they still “significantly and negatively impact real-world functioning,” said Lisa M. Jacola, Ph.D. of the department of psychology and her associates at St. Jude Children’s Research Hospital, Memphis (J Clin Oncol. 2016 Feb 8. [doi: 10.1200/JCO.2015.64.3205]).
Intrathecal chemotherapy has largely replaced cranial radiation therapy to protect the CNS in childhood ALL, but most research concerning neurocognitive outcomes after this treatment has been retrospective and has involved small sample sizes. So investigators performed a prospective study using data from an ongoing trial involving a representative cohort of more than 400 patients aged 1-18 years who were treated at a single center during a 7-year period.
These patients were classified as low- or high-risk based on comprehensive biologic and clinical factors, such as blast cell immunophenotype and genotype, presenting clinical features, and early treatment response. They then received intrathecal methotrexate, cytarabine, and hydrocortisone in doses appropriate to their risk status, as well as standardized leucovorin followed by mercaptopurine and vincristine plus dexamethasone.
The researchers focused on the 211 patients who underwent comprehensive neurocognitive assessment 2 years after diagnosis and treatment. “The overall group did not significantly differ from normative expectations on measures of global intelligence (estimated IQ), academic skills (reading, math, or spelling), and learning and memory.” However, half of them were rated as below average on several measures of attention. In addition, caregivers reported a significantly greater than expected frequency of hyperactivity, impulsivity, and learning problems in the children.
This adverse effect was strongest among children who were younger than age 5 at diagnosis and among those at high risk who received more aggressive treatment.
“Our findings ... emphasize the importance of routine neurocognitive monitoring of all survivors treated with contemporary therapy.” But early detection will only be helpful if effective interventions are developed to remediate these attention and behavior problems. Research has shown that pharmacologic therapies “have reduced acceptability among survivors of childhood cancer,” so nonpharmacologic approaches would be especially useful, the researchers added.
Survivors of childhood acute lymphoblastic leukemia (ALL) who underwent CNS-directed chemotherapy show significant attention problems that impair their functioning 2 years later, according to a report published online Feb. 8 in the Journal of Clinical Oncology.
The attention problems were isolated from intelligence and academic performance, but they still “significantly and negatively impact real-world functioning,” said Lisa M. Jacola, Ph.D. of the department of psychology and her associates at St. Jude Children’s Research Hospital, Memphis (J Clin Oncol. 2016 Feb 8. [doi: 10.1200/JCO.2015.64.3205]).
Intrathecal chemotherapy has largely replaced cranial radiation therapy to protect the CNS in childhood ALL, but most research concerning neurocognitive outcomes after this treatment has been retrospective and has involved small sample sizes. So investigators performed a prospective study using data from an ongoing trial involving a representative cohort of more than 400 patients aged 1-18 years who were treated at a single center during a 7-year period.
These patients were classified as low- or high-risk based on comprehensive biologic and clinical factors, such as blast cell immunophenotype and genotype, presenting clinical features, and early treatment response. They then received intrathecal methotrexate, cytarabine, and hydrocortisone in doses appropriate to their risk status, as well as standardized leucovorin followed by mercaptopurine and vincristine plus dexamethasone.
The researchers focused on the 211 patients who underwent comprehensive neurocognitive assessment 2 years after diagnosis and treatment. “The overall group did not significantly differ from normative expectations on measures of global intelligence (estimated IQ), academic skills (reading, math, or spelling), and learning and memory.” However, half of them were rated as below average on several measures of attention. In addition, caregivers reported a significantly greater than expected frequency of hyperactivity, impulsivity, and learning problems in the children.
This adverse effect was strongest among children who were younger than age 5 at diagnosis and among those at high risk who received more aggressive treatment.
“Our findings ... emphasize the importance of routine neurocognitive monitoring of all survivors treated with contemporary therapy.” But early detection will only be helpful if effective interventions are developed to remediate these attention and behavior problems. Research has shown that pharmacologic therapies “have reduced acceptability among survivors of childhood cancer,” so nonpharmacologic approaches would be especially useful, the researchers added.
Survivors of childhood acute lymphoblastic leukemia (ALL) who underwent CNS-directed chemotherapy show significant attention problems that impair their functioning 2 years later, according to a report published online Feb. 8 in the Journal of Clinical Oncology.
The attention problems were isolated from intelligence and academic performance, but they still “significantly and negatively impact real-world functioning,” said Lisa M. Jacola, Ph.D. of the department of psychology and her associates at St. Jude Children’s Research Hospital, Memphis (J Clin Oncol. 2016 Feb 8. [doi: 10.1200/JCO.2015.64.3205]).
Intrathecal chemotherapy has largely replaced cranial radiation therapy to protect the CNS in childhood ALL, but most research concerning neurocognitive outcomes after this treatment has been retrospective and has involved small sample sizes. So investigators performed a prospective study using data from an ongoing trial involving a representative cohort of more than 400 patients aged 1-18 years who were treated at a single center during a 7-year period.
These patients were classified as low- or high-risk based on comprehensive biologic and clinical factors, such as blast cell immunophenotype and genotype, presenting clinical features, and early treatment response. They then received intrathecal methotrexate, cytarabine, and hydrocortisone in doses appropriate to their risk status, as well as standardized leucovorin followed by mercaptopurine and vincristine plus dexamethasone.
The researchers focused on the 211 patients who underwent comprehensive neurocognitive assessment 2 years after diagnosis and treatment. “The overall group did not significantly differ from normative expectations on measures of global intelligence (estimated IQ), academic skills (reading, math, or spelling), and learning and memory.” However, half of them were rated as below average on several measures of attention. In addition, caregivers reported a significantly greater than expected frequency of hyperactivity, impulsivity, and learning problems in the children.
This adverse effect was strongest among children who were younger than age 5 at diagnosis and among those at high risk who received more aggressive treatment.
“Our findings ... emphasize the importance of routine neurocognitive monitoring of all survivors treated with contemporary therapy.” But early detection will only be helpful if effective interventions are developed to remediate these attention and behavior problems. Research has shown that pharmacologic therapies “have reduced acceptability among survivors of childhood cancer,” so nonpharmacologic approaches would be especially useful, the researchers added.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Survivors of childhood ALL who underwent CNS-directed chemotherapy show significant attention problems that impair their functioning 2 years later.
Major finding: 50% of the study participants were rated as below average on several measures of attention, and caregivers reported a significantly greater than expected frequency of hyperactivity, impulsivity, and learning problems.
Data source: A prospective longitudinal analysis of neurocognitive function in 211 ALL survivors assessed 2 years after diagnosis and treatment.
Disclosures: This study was supported by the National Cancer Institute and American Lebanese Syrian Associated Charities. Dr. Jacola reported having no relevant financial disclosures; one of her associates reported receiving research funding from Sigma Tau Pharmaceuticals.
Induction ALL treatment can cause bone loss
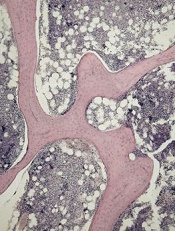
of cancellous bone
Patients with acute lymphoblastic leukemia (ALL) may experience significant bone loss much earlier than previously assumed, according to a study published in the journal Bone.
Investigators analyzed a cohort of adolescent and young adult ALL patients before and after their first month of chemotherapy and observed “significant alterations” to cancellous and cortical bone in this short period of time.
Previous studies to determine the changes to bone density during ALL therapy had focused on the cumulative effects of chemotherapy after months or even years of treatment.
“In clinic, we would see patients with fractures and vertebral compression during the very first few weeks of treatment,” said study author Etan Orgel, MD, of Children’s Hospital Los Angeles in California.
“But we were unaware of any study that specifically examined bone before chemotherapy and immediately after the first 30 days of treatment, which would allow us to understand the impact of this early treatment phase.”
So Dr Orgel and his colleagues conducted a prospective study of 38 patients, ages 10 to 21, who were newly diagnosed with ALL.
The team used quantitative computerized tomography (QCT) to assess leukemia-related changes to bone at diagnosis and then the subsequent effects of the induction phase of chemotherapy.
All of the patients received a 28-day induction regimen consisting of vincristine, pegylated L-asparaginase, anthracycline (daunorubicin or doxorubicin), and a glucocorticoid (either prednisone at 60 mg/m2/day for 28 days or dexamethasone at 10 mg/m2/day for 14 days).
The investigators compared the patients to age- and sex-matched controls and found that leukemia did not dramatically alter the properties of bone before chemotherapy.
However, QCT revealed significant changes during the 30-day induction phase in the 35 patients who were well enough to undergo imaging after treatment.
The patients experienced a significant decrease in cancellous volumetric bone mineral density, which was measured in the spine. The median decrease was 27% (P<0.001).
There was no significant change in cortical volumetric bone mineral density, which was measured in the tibia (−0.0%, P=0.860) or femur (−0.7%, P=0.290).
But there was significant cortical thinning in the tibia. The average cortical thickness decreased 1.2% (P<0.001), and the cortical area decreased 0.4% (P=0.014).
The femur was less affected, the investigators said. There was a decrease in average cortical thickness, but this was not significant (-0.3%, P=0.740).
To help clinicians relate to these findings, the investigators also measured bone mineral density using the older but more widely available technique of dual-energy x-ray absorptiometry. They found that it underestimated these changes as compared to QCT measurements.
“Now that we know how soon bone toxicity occurs, we need to re-evaluate our approaches to managing these changes and focus research efforts on new ways to mitigate this common yet significant adverse effect,” said study author Steven Mittelman, MD, PhD, of Children’s Hospital Los Angeles.

of cancellous bone
Patients with acute lymphoblastic leukemia (ALL) may experience significant bone loss much earlier than previously assumed, according to a study published in the journal Bone.
Investigators analyzed a cohort of adolescent and young adult ALL patients before and after their first month of chemotherapy and observed “significant alterations” to cancellous and cortical bone in this short period of time.
Previous studies to determine the changes to bone density during ALL therapy had focused on the cumulative effects of chemotherapy after months or even years of treatment.
“In clinic, we would see patients with fractures and vertebral compression during the very first few weeks of treatment,” said study author Etan Orgel, MD, of Children’s Hospital Los Angeles in California.
“But we were unaware of any study that specifically examined bone before chemotherapy and immediately after the first 30 days of treatment, which would allow us to understand the impact of this early treatment phase.”
So Dr Orgel and his colleagues conducted a prospective study of 38 patients, ages 10 to 21, who were newly diagnosed with ALL.
The team used quantitative computerized tomography (QCT) to assess leukemia-related changes to bone at diagnosis and then the subsequent effects of the induction phase of chemotherapy.
All of the patients received a 28-day induction regimen consisting of vincristine, pegylated L-asparaginase, anthracycline (daunorubicin or doxorubicin), and a glucocorticoid (either prednisone at 60 mg/m2/day for 28 days or dexamethasone at 10 mg/m2/day for 14 days).
The investigators compared the patients to age- and sex-matched controls and found that leukemia did not dramatically alter the properties of bone before chemotherapy.
However, QCT revealed significant changes during the 30-day induction phase in the 35 patients who were well enough to undergo imaging after treatment.
The patients experienced a significant decrease in cancellous volumetric bone mineral density, which was measured in the spine. The median decrease was 27% (P<0.001).
There was no significant change in cortical volumetric bone mineral density, which was measured in the tibia (−0.0%, P=0.860) or femur (−0.7%, P=0.290).
But there was significant cortical thinning in the tibia. The average cortical thickness decreased 1.2% (P<0.001), and the cortical area decreased 0.4% (P=0.014).
The femur was less affected, the investigators said. There was a decrease in average cortical thickness, but this was not significant (-0.3%, P=0.740).
To help clinicians relate to these findings, the investigators also measured bone mineral density using the older but more widely available technique of dual-energy x-ray absorptiometry. They found that it underestimated these changes as compared to QCT measurements.
“Now that we know how soon bone toxicity occurs, we need to re-evaluate our approaches to managing these changes and focus research efforts on new ways to mitigate this common yet significant adverse effect,” said study author Steven Mittelman, MD, PhD, of Children’s Hospital Los Angeles.

of cancellous bone
Patients with acute lymphoblastic leukemia (ALL) may experience significant bone loss much earlier than previously assumed, according to a study published in the journal Bone.
Investigators analyzed a cohort of adolescent and young adult ALL patients before and after their first month of chemotherapy and observed “significant alterations” to cancellous and cortical bone in this short period of time.
Previous studies to determine the changes to bone density during ALL therapy had focused on the cumulative effects of chemotherapy after months or even years of treatment.
“In clinic, we would see patients with fractures and vertebral compression during the very first few weeks of treatment,” said study author Etan Orgel, MD, of Children’s Hospital Los Angeles in California.
“But we were unaware of any study that specifically examined bone before chemotherapy and immediately after the first 30 days of treatment, which would allow us to understand the impact of this early treatment phase.”
So Dr Orgel and his colleagues conducted a prospective study of 38 patients, ages 10 to 21, who were newly diagnosed with ALL.
The team used quantitative computerized tomography (QCT) to assess leukemia-related changes to bone at diagnosis and then the subsequent effects of the induction phase of chemotherapy.
All of the patients received a 28-day induction regimen consisting of vincristine, pegylated L-asparaginase, anthracycline (daunorubicin or doxorubicin), and a glucocorticoid (either prednisone at 60 mg/m2/day for 28 days or dexamethasone at 10 mg/m2/day for 14 days).
The investigators compared the patients to age- and sex-matched controls and found that leukemia did not dramatically alter the properties of bone before chemotherapy.
However, QCT revealed significant changes during the 30-day induction phase in the 35 patients who were well enough to undergo imaging after treatment.
The patients experienced a significant decrease in cancellous volumetric bone mineral density, which was measured in the spine. The median decrease was 27% (P<0.001).
There was no significant change in cortical volumetric bone mineral density, which was measured in the tibia (−0.0%, P=0.860) or femur (−0.7%, P=0.290).
But there was significant cortical thinning in the tibia. The average cortical thickness decreased 1.2% (P<0.001), and the cortical area decreased 0.4% (P=0.014).
The femur was less affected, the investigators said. There was a decrease in average cortical thickness, but this was not significant (-0.3%, P=0.740).
To help clinicians relate to these findings, the investigators also measured bone mineral density using the older but more widely available technique of dual-energy x-ray absorptiometry. They found that it underestimated these changes as compared to QCT measurements.
“Now that we know how soon bone toxicity occurs, we need to re-evaluate our approaches to managing these changes and focus research efforts on new ways to mitigate this common yet significant adverse effect,” said study author Steven Mittelman, MD, PhD, of Children’s Hospital Los Angeles.
Generic imatinib launched with savings program

Photo by Rhoda Baer
Sun Pharma has announced the US launch of imatinib mesylate tablets, which are a generic version of Novartis’s Gleevec, for indications approved by the US Food and Drug Administration (FDA).
As part of this launch, Sun Pharma has rolled out a savings card program. The goal is to provide greater access to imatinib mesylate tablets for patients who have commercial insurance, but their out-of-pocket cost may exceed an affordable amount.
Sun Pharma’s Imatinib Mesylate Savings Card will reduce patient’s co-payment to $10. The card will also offer patients an additional savings benefit of up to $700 for a 30-day fill to offset any additional out-of-pocket cost should they be required to meet their deductible or co-insurance.
Participating pharmacies across the US can use the patient’s card as part of this program.
Eligible patients can participate in Sun Pharma’s Imatinib Mesylate Savings Card program by registering at www.imatinibrx.com or by requesting a savings card from their oncologist. Sun Pharma will be supplying its Imatinib Mesylate Savings Cards to more than 4500 oncologists.
Sun Pharma has established a Hub service so patients can call and speak with a trained healthcare professional about imatinib mesylate. The number is 1-844-502-5950.
In addition, qualifying patients can receive Sun Pharma’s imatinib mesylate at no cost. Based on qualifications for applying and including a doctor’s prescription, the Hub service will determine if a patient is qualified to receive imatinib mesylate for free. Upon acceptance, the prescription will be processed and delivered to the qualifying patient at no cost.
Sun Pharma’s imatinib mesylate was approved by the FDA in December 2015 and was granted 180 days of marketing exclusivity from the time of its launch. The drug is available in 100 mg and 400 mg tablets.
It is approved to treat:
- Newly diagnosed adult and pediatric patients with Philadelphia-chromosome-positive chronic myeloid leukemia (Ph+ CML) in chronic phase
- Patients with Ph+ CML in blast crisis, accelerated phase, or in chronic phase after failure of interferon-alpha therapy
- Adults with relapsed or refractory Ph+ acute lymphoblastic leukemia
- Adults with myelodysplastic/myeloproliferative diseases associated with PDGFR gene re-arrangements
- Adults with aggressive systemic mastocytosis without the D816V c-Kit mutation or with c-Kit mutational status unknown
- Adults with hypereosinophilic syndrome and/or chronic eosinophilic leukemia, including those who have the FIP1L1-PDGFRα fusion kinase
- Adult patients with unresectable, recurrent, and/or metastatic dermatofibrosarcoma protuberans.
Sun Pharma’s imatinib mesylate is not approved to treat patients with KIT (CD117)-positive unresectable and/or metastatic malignant gastrointestinal stromal tumors.

Photo by Rhoda Baer
Sun Pharma has announced the US launch of imatinib mesylate tablets, which are a generic version of Novartis’s Gleevec, for indications approved by the US Food and Drug Administration (FDA).
As part of this launch, Sun Pharma has rolled out a savings card program. The goal is to provide greater access to imatinib mesylate tablets for patients who have commercial insurance, but their out-of-pocket cost may exceed an affordable amount.
Sun Pharma’s Imatinib Mesylate Savings Card will reduce patient’s co-payment to $10. The card will also offer patients an additional savings benefit of up to $700 for a 30-day fill to offset any additional out-of-pocket cost should they be required to meet their deductible or co-insurance.
Participating pharmacies across the US can use the patient’s card as part of this program.
Eligible patients can participate in Sun Pharma’s Imatinib Mesylate Savings Card program by registering at www.imatinibrx.com or by requesting a savings card from their oncologist. Sun Pharma will be supplying its Imatinib Mesylate Savings Cards to more than 4500 oncologists.
Sun Pharma has established a Hub service so patients can call and speak with a trained healthcare professional about imatinib mesylate. The number is 1-844-502-5950.
In addition, qualifying patients can receive Sun Pharma’s imatinib mesylate at no cost. Based on qualifications for applying and including a doctor’s prescription, the Hub service will determine if a patient is qualified to receive imatinib mesylate for free. Upon acceptance, the prescription will be processed and delivered to the qualifying patient at no cost.
Sun Pharma’s imatinib mesylate was approved by the FDA in December 2015 and was granted 180 days of marketing exclusivity from the time of its launch. The drug is available in 100 mg and 400 mg tablets.
It is approved to treat:
- Newly diagnosed adult and pediatric patients with Philadelphia-chromosome-positive chronic myeloid leukemia (Ph+ CML) in chronic phase
- Patients with Ph+ CML in blast crisis, accelerated phase, or in chronic phase after failure of interferon-alpha therapy
- Adults with relapsed or refractory Ph+ acute lymphoblastic leukemia
- Adults with myelodysplastic/myeloproliferative diseases associated with PDGFR gene re-arrangements
- Adults with aggressive systemic mastocytosis without the D816V c-Kit mutation or with c-Kit mutational status unknown
- Adults with hypereosinophilic syndrome and/or chronic eosinophilic leukemia, including those who have the FIP1L1-PDGFRα fusion kinase
- Adult patients with unresectable, recurrent, and/or metastatic dermatofibrosarcoma protuberans.
Sun Pharma’s imatinib mesylate is not approved to treat patients with KIT (CD117)-positive unresectable and/or metastatic malignant gastrointestinal stromal tumors.

Photo by Rhoda Baer
Sun Pharma has announced the US launch of imatinib mesylate tablets, which are a generic version of Novartis’s Gleevec, for indications approved by the US Food and Drug Administration (FDA).
As part of this launch, Sun Pharma has rolled out a savings card program. The goal is to provide greater access to imatinib mesylate tablets for patients who have commercial insurance, but their out-of-pocket cost may exceed an affordable amount.
Sun Pharma’s Imatinib Mesylate Savings Card will reduce patient’s co-payment to $10. The card will also offer patients an additional savings benefit of up to $700 for a 30-day fill to offset any additional out-of-pocket cost should they be required to meet their deductible or co-insurance.
Participating pharmacies across the US can use the patient’s card as part of this program.
Eligible patients can participate in Sun Pharma’s Imatinib Mesylate Savings Card program by registering at www.imatinibrx.com or by requesting a savings card from their oncologist. Sun Pharma will be supplying its Imatinib Mesylate Savings Cards to more than 4500 oncologists.
Sun Pharma has established a Hub service so patients can call and speak with a trained healthcare professional about imatinib mesylate. The number is 1-844-502-5950.
In addition, qualifying patients can receive Sun Pharma’s imatinib mesylate at no cost. Based on qualifications for applying and including a doctor’s prescription, the Hub service will determine if a patient is qualified to receive imatinib mesylate for free. Upon acceptance, the prescription will be processed and delivered to the qualifying patient at no cost.
Sun Pharma’s imatinib mesylate was approved by the FDA in December 2015 and was granted 180 days of marketing exclusivity from the time of its launch. The drug is available in 100 mg and 400 mg tablets.
It is approved to treat:
- Newly diagnosed adult and pediatric patients with Philadelphia-chromosome-positive chronic myeloid leukemia (Ph+ CML) in chronic phase
- Patients with Ph+ CML in blast crisis, accelerated phase, or in chronic phase after failure of interferon-alpha therapy
- Adults with relapsed or refractory Ph+ acute lymphoblastic leukemia
- Adults with myelodysplastic/myeloproliferative diseases associated with PDGFR gene re-arrangements
- Adults with aggressive systemic mastocytosis without the D816V c-Kit mutation or with c-Kit mutational status unknown
- Adults with hypereosinophilic syndrome and/or chronic eosinophilic leukemia, including those who have the FIP1L1-PDGFRα fusion kinase
- Adult patients with unresectable, recurrent, and/or metastatic dermatofibrosarcoma protuberans.
Sun Pharma’s imatinib mesylate is not approved to treat patients with KIT (CD117)-positive unresectable and/or metastatic malignant gastrointestinal stromal tumors.