User login
50 years of gynecologic surgery: A large dose of ingenuity, a small dose of controversy
Over the past 50 years, there has been explosive change in gynecologic surgery. Ob.Gyn. News has been at the forefront of capturing and chronicling this paradigm shift in the treatment of the female patient.
Our beginnings
From antiquity, physicians and surgeons have struggled with pelvic prolapse, uterine fibroids, ovarian cysts, urinary incontinence, vesicovaginal fistulas, pelvic pain, and abnormal uterine bleeding. At the time of the first edition of Ob.Gyn. News, it had been less than a century since Thomas Edison invented the light bulb; just over 50 years since Hans Christian Jacobaeus first created air pneumoperitoneum using a trocar, followed by the Nitze cystoscope; about 40 years since Richard Zollikofer created a carbon dioxide pneumoperitoneum; 25 years since F.H. Powers and A.C. Barnes had first described laparoscopic tubal sterilization by cautery; and about 20 years since Raoul Palmer, considered the father of modern laparoscopy, had first described the technique – left upper quadrant entry, testing insufflation, Trendelenburg positioning, and simple laparoscopic instrumentation.
In the 1950s, Hans Frangenheim would bring monopolar electrosurgery to laparoscopy and Harold Hopkins would introduce fiber optics. It was not until 1967 that Patrick Steptoe would publish the first textbook on laparoscopy in the English language.
Although usage as a diagnostic tool and a method of sterilization increased popularity of laparoscopy in the 1960s and early 1970s, there were few advances. In fact, a review of early editions of Ob.Gyn. News during that time period shows that the majority of articles involving laparoscopy dealt with sterilization; including the introduction of clips for tubal sterilization by Jaroslav Hulka in 1972. This did not deter the efforts of Jordan Phillips, who along with Jacques Rioux, Louis Keith, Richard Soderstrom – four early laparoscopists – incorporated a new society, the American Association of Gynecologic Laparoscopists (the AAGL) in 1971.
Simultaneously, in 1979, James Daniell in the United States, Maurice Bruhart in France, and Yona Tadir in Israel were promoting efforts to couple the carbon dioxide laser to the laparoscope to treat pelvic adhesions and endometriosis. Later on, fiber lasers, KTP, Nd:YAG, and Argon lasers would be utilized in our field. Still, only a few extirpative procedures were being performed via a laparoscope route. This included linear salpingostomy for the treatment of ectopic pregnancy, championed by Professor Bruhart and H. Manhes in Europe, and Alan DeCherney in the United States.
During the 1980s, laparoscopic surgery was at its innovative best. Through the pioneering efforts of Professor Kurt Semm and his protégée, Liselotte Mettler, the gynecologic laparoscopist was introduced to endoloops, simple suturing techniques, and mechanical morcellation techniques.
Procedures such as salpingo-oophorectomy, appendectomy, and myomectomy could now be performed via the laparoscope. Dr. Camran Nezhat coupled the carbon dioxide laser, the laparoscope, and the television monitor, coining the term laparoscopy. Most importantly, the laparoscopic surgeon was liberated; he or she could remain upright and perform surgery with both hands. Through the 1980s and 1990s, Dr. Nezhat, Dr. Harry Reich, and other innovators pushed the envelope in increasing the ability to extirpate endometriosis, excise severe pelvic adhesions, and perform discoid and segmental bowel resection.
The day the earth stood still
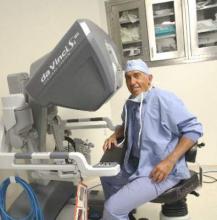
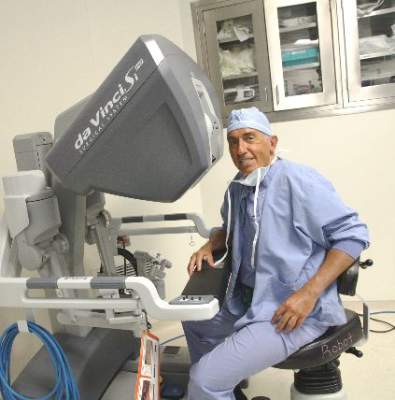
Every gynecologic laparoscopic surgeon should remember Jan. 26, 1988, as that was the date that Dr. Harry Reich performed the first total laparoscopic hysterectomy. Now, little more than 25 years later, in many parts of the country, a laparoscopic approach to hysterectomy is indeed the most common route. Over the years, with the evolution of instrumentation, including new energy systems (ultrasonic, advanced bipolar) and the introduction of barbed sutures, hysterectomy can now be performed via minilaparoscopy, single-site laparoscopy, robot-assisted, and robotic single site, all of which have been featured in the Ob.Gyn. News’ Master Class in Gynecologic Surgery.
But hysteroscopy came first
Abulkasim utilized a mirror to reflect light into the vaginal vault in 1,000 A.D. In 1806, Philipp Bozzini originated the idea of illuminating body cavities by an external light source. Through a system of mirrors and tubes, candlelight could be reflected into the body. In 1869, D.C. Pantaleoni used a cystoscope developed by Antoine Desormeaux – who has been called the father of endoscopy – to treat endometrial polyps with silver nitrate.
Through the 50 years of Ob.Gyn. News and over the past 12 years of the Master Class in Gynecologic Surgery, our community has been consistently updated as to advances in hysteroscopy, not only to enhance treatment efficacy, but safety as well. This has included such advances as the continuous flow hysteroscope, the Hamou contact hysteroscope, and fluid management systems to enhance visualization.
In 1978, Robert Neuwirth introduced loops to perform hysteroscopic myomectomy. The loop resectoscope was quickly followed by the rollerball to perform endometrial ablation. In the late 1990s, hysteroscopic bipolar cutting loops were introduced. This enabled use of ionic distension media saline, instead of nonionic media, thus decreasing risks related to hyponatremia.
In 2003, Mark Emanuel introduced hysteroscopic morcellation systems, which enabled more gynecologists to perform operative hysteroscopy safely. Resected tissue is removed immediately to allow superior visualization. The flexible hysteroscope coupled with vaginoscopy has enabled hysteroscopy to be done with minimal to no anesthesia in an in-office setting.
With advances in hysteroscopy over the past 35 years, hysteroscopic procedures such as polypectomy, myomectomy, lysis of adhesions, transection of endometriosis, evacuation of retained products of conception, and endometrial ablation/resection have become routine.
And now, the controversy
Since its inception, laparoscopic surgery has not been without controversy. In 1933, Karl Fervers described explosion and flashes of light from a combination of high frequency electric current and oxygen distension gas while performing laparoscopic adhesiolysis with the coagulation probe of the ureterocystoscope.
In the early 1970s, Professor Kurt Semm’s pioneering effort was not rewarded by his department, in Kiel, Germany, which instead recommended he schedule a brain scan and psychological testing.
Nearly 20 years later, in a 1992 edition of Current Science, Professor Semm, along with Alan DeCherney, stated that “over 80% of gynecological operations can now be performed by laparoscopy.” Shortly thereafter, however, Dr. Roy Pitkin, who at the time was president of the American College of Obstetricians and Gynecologists, wrote an editorial in the Journal of Obstetrics and Gynecology – “Operative Laparoscopy: Surgical Advance or Technical Gimmick?” (Obstet Gynecol. 1992 Mar;79[3]:441-2).
Fortunately, 18 years later, with the continued advances in laparoscopic surgery making it less expensive, safer, and more accessible, Dr. Pitkin did retract his statement (Obstet Gynecol. 2010 May;115[5]:890-1).
Currently, the gynecologic community is embroiled in controversies involving the use of the robot to assist in the performance of laparoscopic surgery, the incorporation of synthetic mesh to enhance urogynecologic procedures, the placement of Essure micro-inserts to occlude fallopian tubes, and the use of electronic power morcellation at time of laparoscopic or robot-assisted hysterectomy, myomectomy, or sacrocolpopexy.
After reading the 2013 article by Dr. Jason Wright, published in JAMA, comparing laparoscopic hysterectomy to robotic hysterectomy, no one can deny that the rise in a minimally invasive route to hysterectomy has coincided with the advent of the robot (JAMA. 2013 Feb 20;309[7]:689-98). On the other hand, many detractors, including Dr. James Breeden (past ACOG president 2012-2013), find the higher cost of robotic surgery very problematic. In fact, many of these detractors cite the paucity of data showing a significant advantage to use of robotics.
While certainly cost, more than ever, must be a major consideration, remember that during the 1990s, there were multiple articles in Ob.Gyn. News raising concerns about the cost of laparoscopic hysterectomy. Interestingly, studies over the past decade by Warren and Jonsdottir show a cost savings when hysterectomy is done laparoscopically as opposed to its being done by laparotomy. Thus, it certainly can be anticipated that with more physician experience, improved instrumentation, and robotic industry competition, the overall cost will become more comparable to a laparoscopic route.
In 1995, Ulf Ulmsten first described the use of tension-free tape (TVT) to treat stress urinary incontinence. In 1998, the Food and Drug Administration approved the use of the TVT sling in the United States. Since then, transobturator tension-free vaginal tape (TVT-O) and single incision mini-slings have been introduced. All of these techniques have been shown to be successful and have been well adapted into the armamentarium of physicians treating stress urinary incontinence.
With the success of synthetic mesh for the treatment of stress urinary incontinence, its use was extended to pelvic prolapse. In 2002, the first mesh device with indications for the treatment of pelvic organ prolapse was approved by the FDA. While the erosion rate utilizing synthetic mesh for stress urinary incontinence has been noted to be 2%, rates up to 8.3% have been noted in patients treated for pelvic prolapse.
In 2008, the FDA issued a warning regarding the use of mesh for prolapse and incontinence repair secondary to the sequelae of mesh erosion. Subsequently, in 2011, the concern was limited to vaginal mesh to correct pelvic organ prolapse. Finally, on Jan. 4, 2016, the FDA issued an order to reclassify surgical mesh to repair pelvic organ prolapse from class II, which includes moderate-risk devices, to class III, which includes high-risk devices. Moreover, the FDA issued a second order to manufacturers to submit a premarket approval application to support the safety and effectiveness of synthetic mesh for transvaginal repair of pelvic organ prolapse.
Essure micro-inserts for permanent birth control received initial approval from the FDA in November 2002. Despite the fact that Essure can be easily placed, is highly effective, and has seemingly low complication rates, concerns have been raised by the Facebook group “Essure Problems” and Erin Brockovich, the focus of the 2000 biographical film starring Julia Roberts.
After more than 5,000 women filed grievances with the FDA between November 2002 and May 2015, based on unintended pregnancies, miscarriages, stillbirths, severe pain, and bleeding, the FDA announced in 2016 that it would require a boxed warning label for Essure. The FDA also called upon Bayer, which makes and markets Essure, to conduct surveillance to assess “risks of the device in a real-world environment.” The agency stated it will use the results to “determine what, if any, further actions related to Essure are needed to protect public health.”
While Jan. 26, 1988, is a very special date in minimally invasive gynecologic surgery, April 17, 2014, is a day of infamy for the gynecologic laparoscopist. For on this day, the FDA announced a warning regarding electronic power morcellation. Many hospitals and hospital systems throughout the country issued bans on electronic power morcellation, leading to needless open laparotomy procedures and thus, introducing prolonged recovery times and increased risk.
At a time when the recent introduction of barbed suture had made both closure of the vaginal cuff at time of hysterectomy and repair of the hysterotomy at myomectomy easier and faster, the gynecologic laparoscopist was taking a step backward. The FDA based this decision and a subsequent boxed warning – issued in November 2014 – on a small number of studies showing potential upstaging of leiomyosarcoma post electronic power morcellation. Interestingly, many of the morcellation procedures cited did not use power morcellation. Furthermore, a more comprehensive meta-analysis by Elizabeth A. Pritts and colleagues, showed a far lower risk than suggested by the FDA (Gynecol Surg. 2015;12[3]:165-77).
Recently, an article by William Parker and colleagues recommended that the FDA reverse its position (Obstet Gynecol. 2016 Jan;127[1]:18-22). Many believe that ultimately, the solution will be morcellation in a containment bag, which I and my colleagues have been performing in virtually every power morcellation procedure since May 2014. During this current power morcellation controversy, the Master Class in Gynecologic Surgery has continued to update its readers with three different articles related to the subject.
And in conclusion
Without a doubt, the past 50 years of gynecologic surgery has been a time of unparalleled innovation with occasional controversy thrown in. Ob.Gyn. News and more recently, the Master Class in Gynecologic Surgery, has had a major leadership role in bringing this profound ingenuity to the gynecology community by introducing this explosion of surgical creativity to its readers.
And what will the next 50 years bring? I believe we will continue to see tremendous advancements in minimally invasive gynecologic surgery. There will be a definite impact of costs on the marketplace. Thus, many of the minor minimally invasive procedures currently performed in the hospital or surgery center will be brought into office settings. In addition, secondary to reimbursement, the more complex cases will be carried out by fewer gynecologic surgeons who have undergone more intense training in pelvic surgery and who can perform these cases more efficiently and with fewer complications. Our ability to perform surgery and what type of procedures we do will not only be based on randomized, controlled trials, but big data collection as well.
Dr. Miller is clinical associate professor at the University of Illinois at Chicago, and past president of the AAGL and the International Society for Gynecologic Endoscopy (ISGE). He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in private practice in Naperville and Schaumburg, Ill.; director of minimally invasive gynecologic surgery and the director of the AAGL/Society of Reproductive Surgery fellowship in minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill.; and the medical editor of this column, Master Class. Dr. Miller is a consultant and is on the speakers bureau for Ethicon.
Over the past 50 years, there has been explosive change in gynecologic surgery. Ob.Gyn. News has been at the forefront of capturing and chronicling this paradigm shift in the treatment of the female patient.
Our beginnings
From antiquity, physicians and surgeons have struggled with pelvic prolapse, uterine fibroids, ovarian cysts, urinary incontinence, vesicovaginal fistulas, pelvic pain, and abnormal uterine bleeding. At the time of the first edition of Ob.Gyn. News, it had been less than a century since Thomas Edison invented the light bulb; just over 50 years since Hans Christian Jacobaeus first created air pneumoperitoneum using a trocar, followed by the Nitze cystoscope; about 40 years since Richard Zollikofer created a carbon dioxide pneumoperitoneum; 25 years since F.H. Powers and A.C. Barnes had first described laparoscopic tubal sterilization by cautery; and about 20 years since Raoul Palmer, considered the father of modern laparoscopy, had first described the technique – left upper quadrant entry, testing insufflation, Trendelenburg positioning, and simple laparoscopic instrumentation.
In the 1950s, Hans Frangenheim would bring monopolar electrosurgery to laparoscopy and Harold Hopkins would introduce fiber optics. It was not until 1967 that Patrick Steptoe would publish the first textbook on laparoscopy in the English language.
Although usage as a diagnostic tool and a method of sterilization increased popularity of laparoscopy in the 1960s and early 1970s, there were few advances. In fact, a review of early editions of Ob.Gyn. News during that time period shows that the majority of articles involving laparoscopy dealt with sterilization; including the introduction of clips for tubal sterilization by Jaroslav Hulka in 1972. This did not deter the efforts of Jordan Phillips, who along with Jacques Rioux, Louis Keith, Richard Soderstrom – four early laparoscopists – incorporated a new society, the American Association of Gynecologic Laparoscopists (the AAGL) in 1971.
Simultaneously, in 1979, James Daniell in the United States, Maurice Bruhart in France, and Yona Tadir in Israel were promoting efforts to couple the carbon dioxide laser to the laparoscope to treat pelvic adhesions and endometriosis. Later on, fiber lasers, KTP, Nd:YAG, and Argon lasers would be utilized in our field. Still, only a few extirpative procedures were being performed via a laparoscope route. This included linear salpingostomy for the treatment of ectopic pregnancy, championed by Professor Bruhart and H. Manhes in Europe, and Alan DeCherney in the United States.
During the 1980s, laparoscopic surgery was at its innovative best. Through the pioneering efforts of Professor Kurt Semm and his protégée, Liselotte Mettler, the gynecologic laparoscopist was introduced to endoloops, simple suturing techniques, and mechanical morcellation techniques.
Procedures such as salpingo-oophorectomy, appendectomy, and myomectomy could now be performed via the laparoscope. Dr. Camran Nezhat coupled the carbon dioxide laser, the laparoscope, and the television monitor, coining the term laparoscopy. Most importantly, the laparoscopic surgeon was liberated; he or she could remain upright and perform surgery with both hands. Through the 1980s and 1990s, Dr. Nezhat, Dr. Harry Reich, and other innovators pushed the envelope in increasing the ability to extirpate endometriosis, excise severe pelvic adhesions, and perform discoid and segmental bowel resection.
The day the earth stood still
Every gynecologic laparoscopic surgeon should remember Jan. 26, 1988, as that was the date that Dr. Harry Reich performed the first total laparoscopic hysterectomy. Now, little more than 25 years later, in many parts of the country, a laparoscopic approach to hysterectomy is indeed the most common route. Over the years, with the evolution of instrumentation, including new energy systems (ultrasonic, advanced bipolar) and the introduction of barbed sutures, hysterectomy can now be performed via minilaparoscopy, single-site laparoscopy, robot-assisted, and robotic single site, all of which have been featured in the Ob.Gyn. News’ Master Class in Gynecologic Surgery.
But hysteroscopy came first
Abulkasim utilized a mirror to reflect light into the vaginal vault in 1,000 A.D. In 1806, Philipp Bozzini originated the idea of illuminating body cavities by an external light source. Through a system of mirrors and tubes, candlelight could be reflected into the body. In 1869, D.C. Pantaleoni used a cystoscope developed by Antoine Desormeaux – who has been called the father of endoscopy – to treat endometrial polyps with silver nitrate.
Through the 50 years of Ob.Gyn. News and over the past 12 years of the Master Class in Gynecologic Surgery, our community has been consistently updated as to advances in hysteroscopy, not only to enhance treatment efficacy, but safety as well. This has included such advances as the continuous flow hysteroscope, the Hamou contact hysteroscope, and fluid management systems to enhance visualization.
In 1978, Robert Neuwirth introduced loops to perform hysteroscopic myomectomy. The loop resectoscope was quickly followed by the rollerball to perform endometrial ablation. In the late 1990s, hysteroscopic bipolar cutting loops were introduced. This enabled use of ionic distension media saline, instead of nonionic media, thus decreasing risks related to hyponatremia.
In 2003, Mark Emanuel introduced hysteroscopic morcellation systems, which enabled more gynecologists to perform operative hysteroscopy safely. Resected tissue is removed immediately to allow superior visualization. The flexible hysteroscope coupled with vaginoscopy has enabled hysteroscopy to be done with minimal to no anesthesia in an in-office setting.
With advances in hysteroscopy over the past 35 years, hysteroscopic procedures such as polypectomy, myomectomy, lysis of adhesions, transection of endometriosis, evacuation of retained products of conception, and endometrial ablation/resection have become routine.
And now, the controversy
Since its inception, laparoscopic surgery has not been without controversy. In 1933, Karl Fervers described explosion and flashes of light from a combination of high frequency electric current and oxygen distension gas while performing laparoscopic adhesiolysis with the coagulation probe of the ureterocystoscope.
In the early 1970s, Professor Kurt Semm’s pioneering effort was not rewarded by his department, in Kiel, Germany, which instead recommended he schedule a brain scan and psychological testing.
Nearly 20 years later, in a 1992 edition of Current Science, Professor Semm, along with Alan DeCherney, stated that “over 80% of gynecological operations can now be performed by laparoscopy.” Shortly thereafter, however, Dr. Roy Pitkin, who at the time was president of the American College of Obstetricians and Gynecologists, wrote an editorial in the Journal of Obstetrics and Gynecology – “Operative Laparoscopy: Surgical Advance or Technical Gimmick?” (Obstet Gynecol. 1992 Mar;79[3]:441-2).
Fortunately, 18 years later, with the continued advances in laparoscopic surgery making it less expensive, safer, and more accessible, Dr. Pitkin did retract his statement (Obstet Gynecol. 2010 May;115[5]:890-1).
Currently, the gynecologic community is embroiled in controversies involving the use of the robot to assist in the performance of laparoscopic surgery, the incorporation of synthetic mesh to enhance urogynecologic procedures, the placement of Essure micro-inserts to occlude fallopian tubes, and the use of electronic power morcellation at time of laparoscopic or robot-assisted hysterectomy, myomectomy, or sacrocolpopexy.
After reading the 2013 article by Dr. Jason Wright, published in JAMA, comparing laparoscopic hysterectomy to robotic hysterectomy, no one can deny that the rise in a minimally invasive route to hysterectomy has coincided with the advent of the robot (JAMA. 2013 Feb 20;309[7]:689-98). On the other hand, many detractors, including Dr. James Breeden (past ACOG president 2012-2013), find the higher cost of robotic surgery very problematic. In fact, many of these detractors cite the paucity of data showing a significant advantage to use of robotics.
While certainly cost, more than ever, must be a major consideration, remember that during the 1990s, there were multiple articles in Ob.Gyn. News raising concerns about the cost of laparoscopic hysterectomy. Interestingly, studies over the past decade by Warren and Jonsdottir show a cost savings when hysterectomy is done laparoscopically as opposed to its being done by laparotomy. Thus, it certainly can be anticipated that with more physician experience, improved instrumentation, and robotic industry competition, the overall cost will become more comparable to a laparoscopic route.
In 1995, Ulf Ulmsten first described the use of tension-free tape (TVT) to treat stress urinary incontinence. In 1998, the Food and Drug Administration approved the use of the TVT sling in the United States. Since then, transobturator tension-free vaginal tape (TVT-O) and single incision mini-slings have been introduced. All of these techniques have been shown to be successful and have been well adapted into the armamentarium of physicians treating stress urinary incontinence.
With the success of synthetic mesh for the treatment of stress urinary incontinence, its use was extended to pelvic prolapse. In 2002, the first mesh device with indications for the treatment of pelvic organ prolapse was approved by the FDA. While the erosion rate utilizing synthetic mesh for stress urinary incontinence has been noted to be 2%, rates up to 8.3% have been noted in patients treated for pelvic prolapse.
In 2008, the FDA issued a warning regarding the use of mesh for prolapse and incontinence repair secondary to the sequelae of mesh erosion. Subsequently, in 2011, the concern was limited to vaginal mesh to correct pelvic organ prolapse. Finally, on Jan. 4, 2016, the FDA issued an order to reclassify surgical mesh to repair pelvic organ prolapse from class II, which includes moderate-risk devices, to class III, which includes high-risk devices. Moreover, the FDA issued a second order to manufacturers to submit a premarket approval application to support the safety and effectiveness of synthetic mesh for transvaginal repair of pelvic organ prolapse.
Essure micro-inserts for permanent birth control received initial approval from the FDA in November 2002. Despite the fact that Essure can be easily placed, is highly effective, and has seemingly low complication rates, concerns have been raised by the Facebook group “Essure Problems” and Erin Brockovich, the focus of the 2000 biographical film starring Julia Roberts.
After more than 5,000 women filed grievances with the FDA between November 2002 and May 2015, based on unintended pregnancies, miscarriages, stillbirths, severe pain, and bleeding, the FDA announced in 2016 that it would require a boxed warning label for Essure. The FDA also called upon Bayer, which makes and markets Essure, to conduct surveillance to assess “risks of the device in a real-world environment.” The agency stated it will use the results to “determine what, if any, further actions related to Essure are needed to protect public health.”
While Jan. 26, 1988, is a very special date in minimally invasive gynecologic surgery, April 17, 2014, is a day of infamy for the gynecologic laparoscopist. For on this day, the FDA announced a warning regarding electronic power morcellation. Many hospitals and hospital systems throughout the country issued bans on electronic power morcellation, leading to needless open laparotomy procedures and thus, introducing prolonged recovery times and increased risk.
At a time when the recent introduction of barbed suture had made both closure of the vaginal cuff at time of hysterectomy and repair of the hysterotomy at myomectomy easier and faster, the gynecologic laparoscopist was taking a step backward. The FDA based this decision and a subsequent boxed warning – issued in November 2014 – on a small number of studies showing potential upstaging of leiomyosarcoma post electronic power morcellation. Interestingly, many of the morcellation procedures cited did not use power morcellation. Furthermore, a more comprehensive meta-analysis by Elizabeth A. Pritts and colleagues, showed a far lower risk than suggested by the FDA (Gynecol Surg. 2015;12[3]:165-77).
Recently, an article by William Parker and colleagues recommended that the FDA reverse its position (Obstet Gynecol. 2016 Jan;127[1]:18-22). Many believe that ultimately, the solution will be morcellation in a containment bag, which I and my colleagues have been performing in virtually every power morcellation procedure since May 2014. During this current power morcellation controversy, the Master Class in Gynecologic Surgery has continued to update its readers with three different articles related to the subject.
And in conclusion
Without a doubt, the past 50 years of gynecologic surgery has been a time of unparalleled innovation with occasional controversy thrown in. Ob.Gyn. News and more recently, the Master Class in Gynecologic Surgery, has had a major leadership role in bringing this profound ingenuity to the gynecology community by introducing this explosion of surgical creativity to its readers.
And what will the next 50 years bring? I believe we will continue to see tremendous advancements in minimally invasive gynecologic surgery. There will be a definite impact of costs on the marketplace. Thus, many of the minor minimally invasive procedures currently performed in the hospital or surgery center will be brought into office settings. In addition, secondary to reimbursement, the more complex cases will be carried out by fewer gynecologic surgeons who have undergone more intense training in pelvic surgery and who can perform these cases more efficiently and with fewer complications. Our ability to perform surgery and what type of procedures we do will not only be based on randomized, controlled trials, but big data collection as well.
Dr. Miller is clinical associate professor at the University of Illinois at Chicago, and past president of the AAGL and the International Society for Gynecologic Endoscopy (ISGE). He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in private practice in Naperville and Schaumburg, Ill.; director of minimally invasive gynecologic surgery and the director of the AAGL/Society of Reproductive Surgery fellowship in minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill.; and the medical editor of this column, Master Class. Dr. Miller is a consultant and is on the speakers bureau for Ethicon.
Over the past 50 years, there has been explosive change in gynecologic surgery. Ob.Gyn. News has been at the forefront of capturing and chronicling this paradigm shift in the treatment of the female patient.
Our beginnings
From antiquity, physicians and surgeons have struggled with pelvic prolapse, uterine fibroids, ovarian cysts, urinary incontinence, vesicovaginal fistulas, pelvic pain, and abnormal uterine bleeding. At the time of the first edition of Ob.Gyn. News, it had been less than a century since Thomas Edison invented the light bulb; just over 50 years since Hans Christian Jacobaeus first created air pneumoperitoneum using a trocar, followed by the Nitze cystoscope; about 40 years since Richard Zollikofer created a carbon dioxide pneumoperitoneum; 25 years since F.H. Powers and A.C. Barnes had first described laparoscopic tubal sterilization by cautery; and about 20 years since Raoul Palmer, considered the father of modern laparoscopy, had first described the technique – left upper quadrant entry, testing insufflation, Trendelenburg positioning, and simple laparoscopic instrumentation.
In the 1950s, Hans Frangenheim would bring monopolar electrosurgery to laparoscopy and Harold Hopkins would introduce fiber optics. It was not until 1967 that Patrick Steptoe would publish the first textbook on laparoscopy in the English language.
Although usage as a diagnostic tool and a method of sterilization increased popularity of laparoscopy in the 1960s and early 1970s, there were few advances. In fact, a review of early editions of Ob.Gyn. News during that time period shows that the majority of articles involving laparoscopy dealt with sterilization; including the introduction of clips for tubal sterilization by Jaroslav Hulka in 1972. This did not deter the efforts of Jordan Phillips, who along with Jacques Rioux, Louis Keith, Richard Soderstrom – four early laparoscopists – incorporated a new society, the American Association of Gynecologic Laparoscopists (the AAGL) in 1971.
Simultaneously, in 1979, James Daniell in the United States, Maurice Bruhart in France, and Yona Tadir in Israel were promoting efforts to couple the carbon dioxide laser to the laparoscope to treat pelvic adhesions and endometriosis. Later on, fiber lasers, KTP, Nd:YAG, and Argon lasers would be utilized in our field. Still, only a few extirpative procedures were being performed via a laparoscope route. This included linear salpingostomy for the treatment of ectopic pregnancy, championed by Professor Bruhart and H. Manhes in Europe, and Alan DeCherney in the United States.
During the 1980s, laparoscopic surgery was at its innovative best. Through the pioneering efforts of Professor Kurt Semm and his protégée, Liselotte Mettler, the gynecologic laparoscopist was introduced to endoloops, simple suturing techniques, and mechanical morcellation techniques.
Procedures such as salpingo-oophorectomy, appendectomy, and myomectomy could now be performed via the laparoscope. Dr. Camran Nezhat coupled the carbon dioxide laser, the laparoscope, and the television monitor, coining the term laparoscopy. Most importantly, the laparoscopic surgeon was liberated; he or she could remain upright and perform surgery with both hands. Through the 1980s and 1990s, Dr. Nezhat, Dr. Harry Reich, and other innovators pushed the envelope in increasing the ability to extirpate endometriosis, excise severe pelvic adhesions, and perform discoid and segmental bowel resection.
The day the earth stood still
Every gynecologic laparoscopic surgeon should remember Jan. 26, 1988, as that was the date that Dr. Harry Reich performed the first total laparoscopic hysterectomy. Now, little more than 25 years later, in many parts of the country, a laparoscopic approach to hysterectomy is indeed the most common route. Over the years, with the evolution of instrumentation, including new energy systems (ultrasonic, advanced bipolar) and the introduction of barbed sutures, hysterectomy can now be performed via minilaparoscopy, single-site laparoscopy, robot-assisted, and robotic single site, all of which have been featured in the Ob.Gyn. News’ Master Class in Gynecologic Surgery.
But hysteroscopy came first
Abulkasim utilized a mirror to reflect light into the vaginal vault in 1,000 A.D. In 1806, Philipp Bozzini originated the idea of illuminating body cavities by an external light source. Through a system of mirrors and tubes, candlelight could be reflected into the body. In 1869, D.C. Pantaleoni used a cystoscope developed by Antoine Desormeaux – who has been called the father of endoscopy – to treat endometrial polyps with silver nitrate.
Through the 50 years of Ob.Gyn. News and over the past 12 years of the Master Class in Gynecologic Surgery, our community has been consistently updated as to advances in hysteroscopy, not only to enhance treatment efficacy, but safety as well. This has included such advances as the continuous flow hysteroscope, the Hamou contact hysteroscope, and fluid management systems to enhance visualization.
In 1978, Robert Neuwirth introduced loops to perform hysteroscopic myomectomy. The loop resectoscope was quickly followed by the rollerball to perform endometrial ablation. In the late 1990s, hysteroscopic bipolar cutting loops were introduced. This enabled use of ionic distension media saline, instead of nonionic media, thus decreasing risks related to hyponatremia.
In 2003, Mark Emanuel introduced hysteroscopic morcellation systems, which enabled more gynecologists to perform operative hysteroscopy safely. Resected tissue is removed immediately to allow superior visualization. The flexible hysteroscope coupled with vaginoscopy has enabled hysteroscopy to be done with minimal to no anesthesia in an in-office setting.
With advances in hysteroscopy over the past 35 years, hysteroscopic procedures such as polypectomy, myomectomy, lysis of adhesions, transection of endometriosis, evacuation of retained products of conception, and endometrial ablation/resection have become routine.
And now, the controversy
Since its inception, laparoscopic surgery has not been without controversy. In 1933, Karl Fervers described explosion and flashes of light from a combination of high frequency electric current and oxygen distension gas while performing laparoscopic adhesiolysis with the coagulation probe of the ureterocystoscope.
In the early 1970s, Professor Kurt Semm’s pioneering effort was not rewarded by his department, in Kiel, Germany, which instead recommended he schedule a brain scan and psychological testing.
Nearly 20 years later, in a 1992 edition of Current Science, Professor Semm, along with Alan DeCherney, stated that “over 80% of gynecological operations can now be performed by laparoscopy.” Shortly thereafter, however, Dr. Roy Pitkin, who at the time was president of the American College of Obstetricians and Gynecologists, wrote an editorial in the Journal of Obstetrics and Gynecology – “Operative Laparoscopy: Surgical Advance or Technical Gimmick?” (Obstet Gynecol. 1992 Mar;79[3]:441-2).
Fortunately, 18 years later, with the continued advances in laparoscopic surgery making it less expensive, safer, and more accessible, Dr. Pitkin did retract his statement (Obstet Gynecol. 2010 May;115[5]:890-1).
Currently, the gynecologic community is embroiled in controversies involving the use of the robot to assist in the performance of laparoscopic surgery, the incorporation of synthetic mesh to enhance urogynecologic procedures, the placement of Essure micro-inserts to occlude fallopian tubes, and the use of electronic power morcellation at time of laparoscopic or robot-assisted hysterectomy, myomectomy, or sacrocolpopexy.
After reading the 2013 article by Dr. Jason Wright, published in JAMA, comparing laparoscopic hysterectomy to robotic hysterectomy, no one can deny that the rise in a minimally invasive route to hysterectomy has coincided with the advent of the robot (JAMA. 2013 Feb 20;309[7]:689-98). On the other hand, many detractors, including Dr. James Breeden (past ACOG president 2012-2013), find the higher cost of robotic surgery very problematic. In fact, many of these detractors cite the paucity of data showing a significant advantage to use of robotics.
While certainly cost, more than ever, must be a major consideration, remember that during the 1990s, there were multiple articles in Ob.Gyn. News raising concerns about the cost of laparoscopic hysterectomy. Interestingly, studies over the past decade by Warren and Jonsdottir show a cost savings when hysterectomy is done laparoscopically as opposed to its being done by laparotomy. Thus, it certainly can be anticipated that with more physician experience, improved instrumentation, and robotic industry competition, the overall cost will become more comparable to a laparoscopic route.
In 1995, Ulf Ulmsten first described the use of tension-free tape (TVT) to treat stress urinary incontinence. In 1998, the Food and Drug Administration approved the use of the TVT sling in the United States. Since then, transobturator tension-free vaginal tape (TVT-O) and single incision mini-slings have been introduced. All of these techniques have been shown to be successful and have been well adapted into the armamentarium of physicians treating stress urinary incontinence.
With the success of synthetic mesh for the treatment of stress urinary incontinence, its use was extended to pelvic prolapse. In 2002, the first mesh device with indications for the treatment of pelvic organ prolapse was approved by the FDA. While the erosion rate utilizing synthetic mesh for stress urinary incontinence has been noted to be 2%, rates up to 8.3% have been noted in patients treated for pelvic prolapse.
In 2008, the FDA issued a warning regarding the use of mesh for prolapse and incontinence repair secondary to the sequelae of mesh erosion. Subsequently, in 2011, the concern was limited to vaginal mesh to correct pelvic organ prolapse. Finally, on Jan. 4, 2016, the FDA issued an order to reclassify surgical mesh to repair pelvic organ prolapse from class II, which includes moderate-risk devices, to class III, which includes high-risk devices. Moreover, the FDA issued a second order to manufacturers to submit a premarket approval application to support the safety and effectiveness of synthetic mesh for transvaginal repair of pelvic organ prolapse.
Essure micro-inserts for permanent birth control received initial approval from the FDA in November 2002. Despite the fact that Essure can be easily placed, is highly effective, and has seemingly low complication rates, concerns have been raised by the Facebook group “Essure Problems” and Erin Brockovich, the focus of the 2000 biographical film starring Julia Roberts.
After more than 5,000 women filed grievances with the FDA between November 2002 and May 2015, based on unintended pregnancies, miscarriages, stillbirths, severe pain, and bleeding, the FDA announced in 2016 that it would require a boxed warning label for Essure. The FDA also called upon Bayer, which makes and markets Essure, to conduct surveillance to assess “risks of the device in a real-world environment.” The agency stated it will use the results to “determine what, if any, further actions related to Essure are needed to protect public health.”
While Jan. 26, 1988, is a very special date in minimally invasive gynecologic surgery, April 17, 2014, is a day of infamy for the gynecologic laparoscopist. For on this day, the FDA announced a warning regarding electronic power morcellation. Many hospitals and hospital systems throughout the country issued bans on electronic power morcellation, leading to needless open laparotomy procedures and thus, introducing prolonged recovery times and increased risk.
At a time when the recent introduction of barbed suture had made both closure of the vaginal cuff at time of hysterectomy and repair of the hysterotomy at myomectomy easier and faster, the gynecologic laparoscopist was taking a step backward. The FDA based this decision and a subsequent boxed warning – issued in November 2014 – on a small number of studies showing potential upstaging of leiomyosarcoma post electronic power morcellation. Interestingly, many of the morcellation procedures cited did not use power morcellation. Furthermore, a more comprehensive meta-analysis by Elizabeth A. Pritts and colleagues, showed a far lower risk than suggested by the FDA (Gynecol Surg. 2015;12[3]:165-77).
Recently, an article by William Parker and colleagues recommended that the FDA reverse its position (Obstet Gynecol. 2016 Jan;127[1]:18-22). Many believe that ultimately, the solution will be morcellation in a containment bag, which I and my colleagues have been performing in virtually every power morcellation procedure since May 2014. During this current power morcellation controversy, the Master Class in Gynecologic Surgery has continued to update its readers with three different articles related to the subject.
And in conclusion
Without a doubt, the past 50 years of gynecologic surgery has been a time of unparalleled innovation with occasional controversy thrown in. Ob.Gyn. News and more recently, the Master Class in Gynecologic Surgery, has had a major leadership role in bringing this profound ingenuity to the gynecology community by introducing this explosion of surgical creativity to its readers.
And what will the next 50 years bring? I believe we will continue to see tremendous advancements in minimally invasive gynecologic surgery. There will be a definite impact of costs on the marketplace. Thus, many of the minor minimally invasive procedures currently performed in the hospital or surgery center will be brought into office settings. In addition, secondary to reimbursement, the more complex cases will be carried out by fewer gynecologic surgeons who have undergone more intense training in pelvic surgery and who can perform these cases more efficiently and with fewer complications. Our ability to perform surgery and what type of procedures we do will not only be based on randomized, controlled trials, but big data collection as well.
Dr. Miller is clinical associate professor at the University of Illinois at Chicago, and past president of the AAGL and the International Society for Gynecologic Endoscopy (ISGE). He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in private practice in Naperville and Schaumburg, Ill.; director of minimally invasive gynecologic surgery and the director of the AAGL/Society of Reproductive Surgery fellowship in minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill.; and the medical editor of this column, Master Class. Dr. Miller is a consultant and is on the speakers bureau for Ethicon.
Examining the fetal origins of obesity
The figures and trends behind the obesity epidemic are alarming: More than one-third of all adults in the United States are obese, as are 34% of women aged 20-39, and 17% of youth aged 2-19, according to data for 2011-2014 from the National Health and Nutrition Examination Survey.
In our ob.gyn. practices, many of us have witnessed the significant climb in national obesity rates over the past several decades. We’ve seen a continued increase in the prevalence of obesity among childbearing women, and a steady increase in the incidence of high-birth-weight babies. The percentage of women weighing 200 pounds has more than doubled since 1980, and up to 3-4 times as many children and teens in various age subsets are obese today as in the 1970s.
The obesity epidemic is often attributed to a high-fat and/or calorie-dense diet and decreased activity levels. However, this is only part of the picture. There has been growing recognition in recent years that obesity may be programmed by the in utero and newborn environment, particularly as it relates to nutritional permutations. We now have evidence, in fact, that developmental programming is likely a primary cause of the obesity epidemic.
Exposure to maternal obesity and being born with a low birth weight – especially a low birth weight paired with rapid catch-up growth – are both associated with a significantly increased risk of childhood and adult obesity.
Research has demonstrated that newborns may be programmed, in both of these scenarios, with an increased appetite and a predisposition to storing calories as fat. In addition, data are accumulating that exposure to bisphenol A and other endocrine-disruptive chemicals, other environmental toxins, and corticosteroids may exert similar programming effects.
This window into the origins of obesity has significant implications for the practice of ob.gyn., where we have the opportunity to address the programming effects of the in utero and early life environment. Most importantly, we must counsel women before pregnancy about the importance of losing weight, guide them during pregnancy to achieve optimal pregnancy nutrition and weight gain, and prepare them to adopt optimal newborn feeding strategies that will guard against overconsumption.
Programming of obesity
The current obesity epidemic is only minimally due to genetics. Although select genetic mutations may be associated with obesity, these mutations account for an exceedingly small proportion of the obese population. Instead, much of the obesity epidemic involves epigenetic change – in this case, largely epigenetic deregulation of gene expression – and more broadly what we call gestational, or developmental, programming.
Developmental programming is a process by which a stress or stimulus at a critical or sensitive period of development has long-term effects. The major part of the developmental process pertaining to cell division occurs during intrauterine life; more than 90% of the cell divisions necessary to make an adult human occur before birth. Although there are important effects of the early newborn period, developmental programming is therefore largely gestational programming. Depending on when an in utero stress or perturbation occurs, it may permanently change cell number and/or cell differentiation, organ structure, metabolic set points, and gene expression.
The late physician Dr. David Barker got us thinking about in utero programming when he demonstrated an association between low birth weight, rapid weight gain in early life, and adult cardiovascular mortality. His theory about how nutrition and growth before birth may affect cardiovascular health later on, as well as other adult chronic diseases and conditions, became known as the Barker Hypothesis.
Many studies, both animal research and human epidemiological studies, have since confirmed and expanded our understanding of this phenomena. Research has demonstrated associations, for instance, between low birth weight and later risks of insulin resistance, diabetes, fatty liver, and the often-underlying metabolic syndrome.
Obesity is also central to the development of the metabolic syndrome, and we now have irrefutable evidence to show that low birth weight infants have a higher risk of obesity than do normal weight infants. We also know, as Dr. Barker and his colleagues had surmised, that the greatest risks occur when there is rapid catch-up growth of low-birth-weight infants in the early years of life.
Moreover, we now understand that maternal obesity has programming effects that are similar to those of an in utero environment of undernutrition and growth restriction. In the past several decades, the marked increase in maternal obesity has resulted in this programming process having an ever-increasing impact.
Both animal and human studies have shown that infants born to obese mothers have the same increased risks for adult chronic disease – including the risk of becoming obese – as those of low birth weight infants. This increased risk is often, but not always, associated with high birth weight, and it is independent of whether the mother has gestational diabetes mellitus (GDM). Having a high birth weight is more likely in the setting of maternal obesity and itself raises the risk of eventual obesity (as does GDM), but an infant’s exposure to maternal obesity in and of itself is a risk factor.
The mechanisms
The programming mechanisms that predispose offspring to obesity are similar in infants of obese mothers and intrauterine growth restricted newborns, though they involve different epigenetic signals. Both involve dysregulation of appetite/satiety and of adipogenesis.
Appetite is primarily controlled by a complex circuit of neurons in the hypothalamus of the brain called the hypothalamic arcuate nucleus. Some neurons are orexigenic and stimulate or increase appetite, while others are anorexigenic and suppress appetite by promoting satiety.
During fetal development, hypothalamic neural stem cells proliferate and differentiate into various cell types. Neurons destined for the arcuate nucleus then differentiate into these so-called appetite neurons and satiety neurons. Though there is continued neural development and maturation during newborn life, hypothalamic control of appetite and satiety is largely set during this period.
Differentiation to appetite or satiety neurons is regulated by a complex interplay of pathways that may be significantly altered by the nutrient environment. Research in our laboratory and others has shown that both limited and excess nutrition can program the structure and function of the arcuate nucleus – changing its wiring, in essence – such that there is an increased ratio of appetite to satiety neurons (Clin Obstet Gynecol. 2013 Sep;56[3]:529-36).
There also appears to be a programmed down-regulation in the reward pathway of the brain, and some studies have shown that children of obese mothers and children who were born with low birth weights have a higher preference for sweet and high-calorie foods. This all begins at the neural stem cell level.
With more appetite neurons and fewer satiety neurons, as well as a down-regulation of reward – and an abundance of available food – a newborn is at high risk of becoming obese. Eating for this child will not only be pleasurable; it will be driven by an enhanced appetite, an inability to feel full after reasonable amounts of food, and a down-regulation of reward (potentially requiring greater amounts of food or a shift in preference for high fat/sweet food to achieve the pleasure from eating).
In addition to alterations in appetite/satiety, the nutrition environment in utero can alter adipose tissue development and function.
Like neural development, adipogenesis – the process by which preadipocytes proliferate and differentiate into mature adipocytes – is tightly regulated by a cascade of transcription factors that are expressed in response to stimuli, including nutrients. In animal studies we have found an up-regulation of adipogenic and lipogenic transcription factors in intrauterine growth restricted offspring as well as in offspring of obese mothers (Reprod Sci. 2008 Oct;15[8]:785-96 and Curr Diab Rep. 2013 Feb;13[1]:27-33).
This up-regulation leads to greater proliferation of preadipocytes and greater lipid synthesis and storage in mature adipocytes. Not only will the newborn have an increased number of adipocytes, but he or she will have an increased number of hypertrophic lipid-filled fat cells. The enhanced adipogenesis will contribute to the newborn’s programmed propensity for obesity, and the directive to “just eat less” will likely be ineffective throughout childhood and beyond.
Programmed offspring are resistant to both central and peripheral effects of leptin and insulin, resulting in impaired satiety (i.e., overeating) and manifestations of GDM. Responses to an array of additional energy regulatory factors (e.g., ghrelin) demonstrate a similar programmed dysfunction.
In practice
There are several approaches that ob.gyns. can take to prevent childhood and lifelong obesity. Most importantly, we must counsel our obese patients to lose weight before pregnancy. In doing so, it may be meaningful and effective to ask the patient to think about her baby’s future as an obese adult.
Patients who have experienced the challenges of trying to lose weight, and who are told about the developmental origins of obesity and how obesity can be programmed, may be more motivated to lose weight to avoid passing on to their children the burden and challenges that they’ve experienced. We can tell obese patients that their children may well be predisposed through the current in utero environment to have an increased appetite and a propensity to store body fat, and that they subsequently will face higher risks of diabetes and other serious chronic conditions.
We should also appropriately counsel women on healthy weight gain during pregnancy, and urge them not to gain excessive weight.
Newborn feeding strategies are also important for babies exposed to gestational programming of obesity, but especially small babies given the high risk of obesity when there is rapid catch-up growth. We must encourage good growth of both the low-birth-weight and macrosomic infant during the newborn period, but not overgrowth.
The importance of breastfeeding cannot be overestimated, as it has been demonstrated to reduce the occurrence of excessive newborn weight gain and improve long term infant health. We should encourage breastfeeding for the natural opportunity it provides to avoid excessive feeding, in addition to its other benefits. And for newborns who are bottle fed, we should counsel the new mother on optimal feeding and strategies for comforting a crying baby, which will protect against overfeeding.
Regarding environmental exposures, this area of developmental programming is continuing to evolve at a rapid rate. Both animal research and epidemiological studies support the association of developmental exposure to BPA and other chemicals with obesity.
For the present, we should educate our patients regarding optimal nutrition prior to and during pregnancy, and the avoidance of potentially toxic or metabolically-active chemicals or drugs. We look forward to continued research into the mechanisms and preventive/therapeutic strategies for optimization of childhood and adult health.
Dr. Ross is professor of obstetrics and gynecology at the University of California, Los Angeles. Dr. Desai is assistant professor of ob.gyn. at the university. They reported having no relevant financial disclosures.
The figures and trends behind the obesity epidemic are alarming: More than one-third of all adults in the United States are obese, as are 34% of women aged 20-39, and 17% of youth aged 2-19, according to data for 2011-2014 from the National Health and Nutrition Examination Survey.
In our ob.gyn. practices, many of us have witnessed the significant climb in national obesity rates over the past several decades. We’ve seen a continued increase in the prevalence of obesity among childbearing women, and a steady increase in the incidence of high-birth-weight babies. The percentage of women weighing 200 pounds has more than doubled since 1980, and up to 3-4 times as many children and teens in various age subsets are obese today as in the 1970s.
The obesity epidemic is often attributed to a high-fat and/or calorie-dense diet and decreased activity levels. However, this is only part of the picture. There has been growing recognition in recent years that obesity may be programmed by the in utero and newborn environment, particularly as it relates to nutritional permutations. We now have evidence, in fact, that developmental programming is likely a primary cause of the obesity epidemic.
Exposure to maternal obesity and being born with a low birth weight – especially a low birth weight paired with rapid catch-up growth – are both associated with a significantly increased risk of childhood and adult obesity.
Research has demonstrated that newborns may be programmed, in both of these scenarios, with an increased appetite and a predisposition to storing calories as fat. In addition, data are accumulating that exposure to bisphenol A and other endocrine-disruptive chemicals, other environmental toxins, and corticosteroids may exert similar programming effects.
This window into the origins of obesity has significant implications for the practice of ob.gyn., where we have the opportunity to address the programming effects of the in utero and early life environment. Most importantly, we must counsel women before pregnancy about the importance of losing weight, guide them during pregnancy to achieve optimal pregnancy nutrition and weight gain, and prepare them to adopt optimal newborn feeding strategies that will guard against overconsumption.
Programming of obesity
The current obesity epidemic is only minimally due to genetics. Although select genetic mutations may be associated with obesity, these mutations account for an exceedingly small proportion of the obese population. Instead, much of the obesity epidemic involves epigenetic change – in this case, largely epigenetic deregulation of gene expression – and more broadly what we call gestational, or developmental, programming.
Developmental programming is a process by which a stress or stimulus at a critical or sensitive period of development has long-term effects. The major part of the developmental process pertaining to cell division occurs during intrauterine life; more than 90% of the cell divisions necessary to make an adult human occur before birth. Although there are important effects of the early newborn period, developmental programming is therefore largely gestational programming. Depending on when an in utero stress or perturbation occurs, it may permanently change cell number and/or cell differentiation, organ structure, metabolic set points, and gene expression.
The late physician Dr. David Barker got us thinking about in utero programming when he demonstrated an association between low birth weight, rapid weight gain in early life, and adult cardiovascular mortality. His theory about how nutrition and growth before birth may affect cardiovascular health later on, as well as other adult chronic diseases and conditions, became known as the Barker Hypothesis.
Many studies, both animal research and human epidemiological studies, have since confirmed and expanded our understanding of this phenomena. Research has demonstrated associations, for instance, between low birth weight and later risks of insulin resistance, diabetes, fatty liver, and the often-underlying metabolic syndrome.
Obesity is also central to the development of the metabolic syndrome, and we now have irrefutable evidence to show that low birth weight infants have a higher risk of obesity than do normal weight infants. We also know, as Dr. Barker and his colleagues had surmised, that the greatest risks occur when there is rapid catch-up growth of low-birth-weight infants in the early years of life.
Moreover, we now understand that maternal obesity has programming effects that are similar to those of an in utero environment of undernutrition and growth restriction. In the past several decades, the marked increase in maternal obesity has resulted in this programming process having an ever-increasing impact.
Both animal and human studies have shown that infants born to obese mothers have the same increased risks for adult chronic disease – including the risk of becoming obese – as those of low birth weight infants. This increased risk is often, but not always, associated with high birth weight, and it is independent of whether the mother has gestational diabetes mellitus (GDM). Having a high birth weight is more likely in the setting of maternal obesity and itself raises the risk of eventual obesity (as does GDM), but an infant’s exposure to maternal obesity in and of itself is a risk factor.
The mechanisms
The programming mechanisms that predispose offspring to obesity are similar in infants of obese mothers and intrauterine growth restricted newborns, though they involve different epigenetic signals. Both involve dysregulation of appetite/satiety and of adipogenesis.
Appetite is primarily controlled by a complex circuit of neurons in the hypothalamus of the brain called the hypothalamic arcuate nucleus. Some neurons are orexigenic and stimulate or increase appetite, while others are anorexigenic and suppress appetite by promoting satiety.
During fetal development, hypothalamic neural stem cells proliferate and differentiate into various cell types. Neurons destined for the arcuate nucleus then differentiate into these so-called appetite neurons and satiety neurons. Though there is continued neural development and maturation during newborn life, hypothalamic control of appetite and satiety is largely set during this period.
Differentiation to appetite or satiety neurons is regulated by a complex interplay of pathways that may be significantly altered by the nutrient environment. Research in our laboratory and others has shown that both limited and excess nutrition can program the structure and function of the arcuate nucleus – changing its wiring, in essence – such that there is an increased ratio of appetite to satiety neurons (Clin Obstet Gynecol. 2013 Sep;56[3]:529-36).
There also appears to be a programmed down-regulation in the reward pathway of the brain, and some studies have shown that children of obese mothers and children who were born with low birth weights have a higher preference for sweet and high-calorie foods. This all begins at the neural stem cell level.
With more appetite neurons and fewer satiety neurons, as well as a down-regulation of reward – and an abundance of available food – a newborn is at high risk of becoming obese. Eating for this child will not only be pleasurable; it will be driven by an enhanced appetite, an inability to feel full after reasonable amounts of food, and a down-regulation of reward (potentially requiring greater amounts of food or a shift in preference for high fat/sweet food to achieve the pleasure from eating).
In addition to alterations in appetite/satiety, the nutrition environment in utero can alter adipose tissue development and function.
Like neural development, adipogenesis – the process by which preadipocytes proliferate and differentiate into mature adipocytes – is tightly regulated by a cascade of transcription factors that are expressed in response to stimuli, including nutrients. In animal studies we have found an up-regulation of adipogenic and lipogenic transcription factors in intrauterine growth restricted offspring as well as in offspring of obese mothers (Reprod Sci. 2008 Oct;15[8]:785-96 and Curr Diab Rep. 2013 Feb;13[1]:27-33).
This up-regulation leads to greater proliferation of preadipocytes and greater lipid synthesis and storage in mature adipocytes. Not only will the newborn have an increased number of adipocytes, but he or she will have an increased number of hypertrophic lipid-filled fat cells. The enhanced adipogenesis will contribute to the newborn’s programmed propensity for obesity, and the directive to “just eat less” will likely be ineffective throughout childhood and beyond.
Programmed offspring are resistant to both central and peripheral effects of leptin and insulin, resulting in impaired satiety (i.e., overeating) and manifestations of GDM. Responses to an array of additional energy regulatory factors (e.g., ghrelin) demonstrate a similar programmed dysfunction.
In practice
There are several approaches that ob.gyns. can take to prevent childhood and lifelong obesity. Most importantly, we must counsel our obese patients to lose weight before pregnancy. In doing so, it may be meaningful and effective to ask the patient to think about her baby’s future as an obese adult.
Patients who have experienced the challenges of trying to lose weight, and who are told about the developmental origins of obesity and how obesity can be programmed, may be more motivated to lose weight to avoid passing on to their children the burden and challenges that they’ve experienced. We can tell obese patients that their children may well be predisposed through the current in utero environment to have an increased appetite and a propensity to store body fat, and that they subsequently will face higher risks of diabetes and other serious chronic conditions.
We should also appropriately counsel women on healthy weight gain during pregnancy, and urge them not to gain excessive weight.
Newborn feeding strategies are also important for babies exposed to gestational programming of obesity, but especially small babies given the high risk of obesity when there is rapid catch-up growth. We must encourage good growth of both the low-birth-weight and macrosomic infant during the newborn period, but not overgrowth.
The importance of breastfeeding cannot be overestimated, as it has been demonstrated to reduce the occurrence of excessive newborn weight gain and improve long term infant health. We should encourage breastfeeding for the natural opportunity it provides to avoid excessive feeding, in addition to its other benefits. And for newborns who are bottle fed, we should counsel the new mother on optimal feeding and strategies for comforting a crying baby, which will protect against overfeeding.
Regarding environmental exposures, this area of developmental programming is continuing to evolve at a rapid rate. Both animal research and epidemiological studies support the association of developmental exposure to BPA and other chemicals with obesity.
For the present, we should educate our patients regarding optimal nutrition prior to and during pregnancy, and the avoidance of potentially toxic or metabolically-active chemicals or drugs. We look forward to continued research into the mechanisms and preventive/therapeutic strategies for optimization of childhood and adult health.
Dr. Ross is professor of obstetrics and gynecology at the University of California, Los Angeles. Dr. Desai is assistant professor of ob.gyn. at the university. They reported having no relevant financial disclosures.
The figures and trends behind the obesity epidemic are alarming: More than one-third of all adults in the United States are obese, as are 34% of women aged 20-39, and 17% of youth aged 2-19, according to data for 2011-2014 from the National Health and Nutrition Examination Survey.
In our ob.gyn. practices, many of us have witnessed the significant climb in national obesity rates over the past several decades. We’ve seen a continued increase in the prevalence of obesity among childbearing women, and a steady increase in the incidence of high-birth-weight babies. The percentage of women weighing 200 pounds has more than doubled since 1980, and up to 3-4 times as many children and teens in various age subsets are obese today as in the 1970s.
The obesity epidemic is often attributed to a high-fat and/or calorie-dense diet and decreased activity levels. However, this is only part of the picture. There has been growing recognition in recent years that obesity may be programmed by the in utero and newborn environment, particularly as it relates to nutritional permutations. We now have evidence, in fact, that developmental programming is likely a primary cause of the obesity epidemic.
Exposure to maternal obesity and being born with a low birth weight – especially a low birth weight paired with rapid catch-up growth – are both associated with a significantly increased risk of childhood and adult obesity.
Research has demonstrated that newborns may be programmed, in both of these scenarios, with an increased appetite and a predisposition to storing calories as fat. In addition, data are accumulating that exposure to bisphenol A and other endocrine-disruptive chemicals, other environmental toxins, and corticosteroids may exert similar programming effects.
This window into the origins of obesity has significant implications for the practice of ob.gyn., where we have the opportunity to address the programming effects of the in utero and early life environment. Most importantly, we must counsel women before pregnancy about the importance of losing weight, guide them during pregnancy to achieve optimal pregnancy nutrition and weight gain, and prepare them to adopt optimal newborn feeding strategies that will guard against overconsumption.
Programming of obesity
The current obesity epidemic is only minimally due to genetics. Although select genetic mutations may be associated with obesity, these mutations account for an exceedingly small proportion of the obese population. Instead, much of the obesity epidemic involves epigenetic change – in this case, largely epigenetic deregulation of gene expression – and more broadly what we call gestational, or developmental, programming.
Developmental programming is a process by which a stress or stimulus at a critical or sensitive period of development has long-term effects. The major part of the developmental process pertaining to cell division occurs during intrauterine life; more than 90% of the cell divisions necessary to make an adult human occur before birth. Although there are important effects of the early newborn period, developmental programming is therefore largely gestational programming. Depending on when an in utero stress or perturbation occurs, it may permanently change cell number and/or cell differentiation, organ structure, metabolic set points, and gene expression.
The late physician Dr. David Barker got us thinking about in utero programming when he demonstrated an association between low birth weight, rapid weight gain in early life, and adult cardiovascular mortality. His theory about how nutrition and growth before birth may affect cardiovascular health later on, as well as other adult chronic diseases and conditions, became known as the Barker Hypothesis.
Many studies, both animal research and human epidemiological studies, have since confirmed and expanded our understanding of this phenomena. Research has demonstrated associations, for instance, between low birth weight and later risks of insulin resistance, diabetes, fatty liver, and the often-underlying metabolic syndrome.
Obesity is also central to the development of the metabolic syndrome, and we now have irrefutable evidence to show that low birth weight infants have a higher risk of obesity than do normal weight infants. We also know, as Dr. Barker and his colleagues had surmised, that the greatest risks occur when there is rapid catch-up growth of low-birth-weight infants in the early years of life.
Moreover, we now understand that maternal obesity has programming effects that are similar to those of an in utero environment of undernutrition and growth restriction. In the past several decades, the marked increase in maternal obesity has resulted in this programming process having an ever-increasing impact.
Both animal and human studies have shown that infants born to obese mothers have the same increased risks for adult chronic disease – including the risk of becoming obese – as those of low birth weight infants. This increased risk is often, but not always, associated with high birth weight, and it is independent of whether the mother has gestational diabetes mellitus (GDM). Having a high birth weight is more likely in the setting of maternal obesity and itself raises the risk of eventual obesity (as does GDM), but an infant’s exposure to maternal obesity in and of itself is a risk factor.
The mechanisms
The programming mechanisms that predispose offspring to obesity are similar in infants of obese mothers and intrauterine growth restricted newborns, though they involve different epigenetic signals. Both involve dysregulation of appetite/satiety and of adipogenesis.
Appetite is primarily controlled by a complex circuit of neurons in the hypothalamus of the brain called the hypothalamic arcuate nucleus. Some neurons are orexigenic and stimulate or increase appetite, while others are anorexigenic and suppress appetite by promoting satiety.
During fetal development, hypothalamic neural stem cells proliferate and differentiate into various cell types. Neurons destined for the arcuate nucleus then differentiate into these so-called appetite neurons and satiety neurons. Though there is continued neural development and maturation during newborn life, hypothalamic control of appetite and satiety is largely set during this period.
Differentiation to appetite or satiety neurons is regulated by a complex interplay of pathways that may be significantly altered by the nutrient environment. Research in our laboratory and others has shown that both limited and excess nutrition can program the structure and function of the arcuate nucleus – changing its wiring, in essence – such that there is an increased ratio of appetite to satiety neurons (Clin Obstet Gynecol. 2013 Sep;56[3]:529-36).
There also appears to be a programmed down-regulation in the reward pathway of the brain, and some studies have shown that children of obese mothers and children who were born with low birth weights have a higher preference for sweet and high-calorie foods. This all begins at the neural stem cell level.
With more appetite neurons and fewer satiety neurons, as well as a down-regulation of reward – and an abundance of available food – a newborn is at high risk of becoming obese. Eating for this child will not only be pleasurable; it will be driven by an enhanced appetite, an inability to feel full after reasonable amounts of food, and a down-regulation of reward (potentially requiring greater amounts of food or a shift in preference for high fat/sweet food to achieve the pleasure from eating).
In addition to alterations in appetite/satiety, the nutrition environment in utero can alter adipose tissue development and function.
Like neural development, adipogenesis – the process by which preadipocytes proliferate and differentiate into mature adipocytes – is tightly regulated by a cascade of transcription factors that are expressed in response to stimuli, including nutrients. In animal studies we have found an up-regulation of adipogenic and lipogenic transcription factors in intrauterine growth restricted offspring as well as in offspring of obese mothers (Reprod Sci. 2008 Oct;15[8]:785-96 and Curr Diab Rep. 2013 Feb;13[1]:27-33).
This up-regulation leads to greater proliferation of preadipocytes and greater lipid synthesis and storage in mature adipocytes. Not only will the newborn have an increased number of adipocytes, but he or she will have an increased number of hypertrophic lipid-filled fat cells. The enhanced adipogenesis will contribute to the newborn’s programmed propensity for obesity, and the directive to “just eat less” will likely be ineffective throughout childhood and beyond.
Programmed offspring are resistant to both central and peripheral effects of leptin and insulin, resulting in impaired satiety (i.e., overeating) and manifestations of GDM. Responses to an array of additional energy regulatory factors (e.g., ghrelin) demonstrate a similar programmed dysfunction.
In practice
There are several approaches that ob.gyns. can take to prevent childhood and lifelong obesity. Most importantly, we must counsel our obese patients to lose weight before pregnancy. In doing so, it may be meaningful and effective to ask the patient to think about her baby’s future as an obese adult.
Patients who have experienced the challenges of trying to lose weight, and who are told about the developmental origins of obesity and how obesity can be programmed, may be more motivated to lose weight to avoid passing on to their children the burden and challenges that they’ve experienced. We can tell obese patients that their children may well be predisposed through the current in utero environment to have an increased appetite and a propensity to store body fat, and that they subsequently will face higher risks of diabetes and other serious chronic conditions.
We should also appropriately counsel women on healthy weight gain during pregnancy, and urge them not to gain excessive weight.
Newborn feeding strategies are also important for babies exposed to gestational programming of obesity, but especially small babies given the high risk of obesity when there is rapid catch-up growth. We must encourage good growth of both the low-birth-weight and macrosomic infant during the newborn period, but not overgrowth.
The importance of breastfeeding cannot be overestimated, as it has been demonstrated to reduce the occurrence of excessive newborn weight gain and improve long term infant health. We should encourage breastfeeding for the natural opportunity it provides to avoid excessive feeding, in addition to its other benefits. And for newborns who are bottle fed, we should counsel the new mother on optimal feeding and strategies for comforting a crying baby, which will protect against overfeeding.
Regarding environmental exposures, this area of developmental programming is continuing to evolve at a rapid rate. Both animal research and epidemiological studies support the association of developmental exposure to BPA and other chemicals with obesity.
For the present, we should educate our patients regarding optimal nutrition prior to and during pregnancy, and the avoidance of potentially toxic or metabolically-active chemicals or drugs. We look forward to continued research into the mechanisms and preventive/therapeutic strategies for optimization of childhood and adult health.
Dr. Ross is professor of obstetrics and gynecology at the University of California, Los Angeles. Dr. Desai is assistant professor of ob.gyn. at the university. They reported having no relevant financial disclosures.
The far-reaching implications of weight gain in pregnancy
It is not surprising that a pregnant woman’s actions heavily influence her developing baby. Ob.gyns. advise patients to stop smoking or to stop using illicit drugs, and limit their alcohol consumption during pregnancy because we know that these substances can cause serious, even fatal, consequences for the fetus. Although we routinely provide nutrition information and guidelines on healthy weight gain in pregnancy, we may not stress the importance of healthy eating to the same degree as we may emphasize the need to eliminate tobacco use. But should we?
In 2011, a study by researchers at Yale University, the University of Texas, and Arizona State University suggested that food can have effects on the brain similar to those of addictive substances (Arch Gen Psychiatry. 2011 Aug;68[8]:808-16). Using MRI, the investigators examined which areas of the brain became active in response to the consumption of a chocolate milkshake, and compared these results to brain scans of people addicted to opioids. The study enrolled 48 women who were lean to obese, based on body mass index. The researchers found that people who were obese had brain activity patterns in response to food that were similar to patterns that people with drug addiction had in response to opioids. Although the sample size was small, the investigators showed, in essence, that food is a “drug.”
Ob.gyns. working with patients who are overweight or obese typically encourage weight loss prior to pregnancy, or suggest limited weight gain during gestation, because obesity increases complications for both the pregnant mother and her unborn baby. If, as the 2011 study suggests, we were to think of food addiction as we do any other drug addiction – tobacco, opioids, alcohol – that should be curbed out of concern for the developing baby, ob.gyns. might tell our patients to reduce or completely eliminate their “trans-fat food habit” before and during pregnancy.
Importantly, a mother’s nutrition, or lack thereof, may exert harmful effects on her child’s long-term health. This idea was intimated decades ago when Dr. David Barker proposed that a person’s future risk for disease began during pregnancy. Exactly how this type of early programming may occur remains to be determined. Therefore, this month we examine the fetal origins of obesity, and have invited Dr. Michael G. Ross, professor of obstetrics and gynecology, and Mina Desai, Ph.D., assistant professor of obstetrics and gynecology, at the University of California, Los Angeles, to discuss this important topic.
Dr. Reece, who specializes in maternal-fetal medicine, is vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. Dr. Reece said he had no relevant financial disclosures. He is the medical editor of this column. Contact him at obnews@frontlinemedcom.com.
It is not surprising that a pregnant woman’s actions heavily influence her developing baby. Ob.gyns. advise patients to stop smoking or to stop using illicit drugs, and limit their alcohol consumption during pregnancy because we know that these substances can cause serious, even fatal, consequences for the fetus. Although we routinely provide nutrition information and guidelines on healthy weight gain in pregnancy, we may not stress the importance of healthy eating to the same degree as we may emphasize the need to eliminate tobacco use. But should we?
In 2011, a study by researchers at Yale University, the University of Texas, and Arizona State University suggested that food can have effects on the brain similar to those of addictive substances (Arch Gen Psychiatry. 2011 Aug;68[8]:808-16). Using MRI, the investigators examined which areas of the brain became active in response to the consumption of a chocolate milkshake, and compared these results to brain scans of people addicted to opioids. The study enrolled 48 women who were lean to obese, based on body mass index. The researchers found that people who were obese had brain activity patterns in response to food that were similar to patterns that people with drug addiction had in response to opioids. Although the sample size was small, the investigators showed, in essence, that food is a “drug.”
Ob.gyns. working with patients who are overweight or obese typically encourage weight loss prior to pregnancy, or suggest limited weight gain during gestation, because obesity increases complications for both the pregnant mother and her unborn baby. If, as the 2011 study suggests, we were to think of food addiction as we do any other drug addiction – tobacco, opioids, alcohol – that should be curbed out of concern for the developing baby, ob.gyns. might tell our patients to reduce or completely eliminate their “trans-fat food habit” before and during pregnancy.
Importantly, a mother’s nutrition, or lack thereof, may exert harmful effects on her child’s long-term health. This idea was intimated decades ago when Dr. David Barker proposed that a person’s future risk for disease began during pregnancy. Exactly how this type of early programming may occur remains to be determined. Therefore, this month we examine the fetal origins of obesity, and have invited Dr. Michael G. Ross, professor of obstetrics and gynecology, and Mina Desai, Ph.D., assistant professor of obstetrics and gynecology, at the University of California, Los Angeles, to discuss this important topic.
Dr. Reece, who specializes in maternal-fetal medicine, is vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. Dr. Reece said he had no relevant financial disclosures. He is the medical editor of this column. Contact him at obnews@frontlinemedcom.com.
It is not surprising that a pregnant woman’s actions heavily influence her developing baby. Ob.gyns. advise patients to stop smoking or to stop using illicit drugs, and limit their alcohol consumption during pregnancy because we know that these substances can cause serious, even fatal, consequences for the fetus. Although we routinely provide nutrition information and guidelines on healthy weight gain in pregnancy, we may not stress the importance of healthy eating to the same degree as we may emphasize the need to eliminate tobacco use. But should we?
In 2011, a study by researchers at Yale University, the University of Texas, and Arizona State University suggested that food can have effects on the brain similar to those of addictive substances (Arch Gen Psychiatry. 2011 Aug;68[8]:808-16). Using MRI, the investigators examined which areas of the brain became active in response to the consumption of a chocolate milkshake, and compared these results to brain scans of people addicted to opioids. The study enrolled 48 women who were lean to obese, based on body mass index. The researchers found that people who were obese had brain activity patterns in response to food that were similar to patterns that people with drug addiction had in response to opioids. Although the sample size was small, the investigators showed, in essence, that food is a “drug.”
Ob.gyns. working with patients who are overweight or obese typically encourage weight loss prior to pregnancy, or suggest limited weight gain during gestation, because obesity increases complications for both the pregnant mother and her unborn baby. If, as the 2011 study suggests, we were to think of food addiction as we do any other drug addiction – tobacco, opioids, alcohol – that should be curbed out of concern for the developing baby, ob.gyns. might tell our patients to reduce or completely eliminate their “trans-fat food habit” before and during pregnancy.
Importantly, a mother’s nutrition, or lack thereof, may exert harmful effects on her child’s long-term health. This idea was intimated decades ago when Dr. David Barker proposed that a person’s future risk for disease began during pregnancy. Exactly how this type of early programming may occur remains to be determined. Therefore, this month we examine the fetal origins of obesity, and have invited Dr. Michael G. Ross, professor of obstetrics and gynecology, and Mina Desai, Ph.D., assistant professor of obstetrics and gynecology, at the University of California, Los Angeles, to discuss this important topic.
Dr. Reece, who specializes in maternal-fetal medicine, is vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. Dr. Reece said he had no relevant financial disclosures. He is the medical editor of this column. Contact him at obnews@frontlinemedcom.com.
Managing endometriosis to prevent ovarian cancer
Endometriosis is a common condition, occurring in this country in 1 of 10 women of reproductive age. An association between endometriosis and subsequent ovarian carcinoma has been reported for decades, yet it is only recently that our knowledge has deepened enough to support more rational methods for preventing the malignancy.
Each year, approximately 22,000 new cases of ovarian cancer are diagnosed. The lifetime risk of developing this malignancy is low, but it is the deadliest of the gynecologic malignancies, with diagnosis usually made in advanced stages when prognosis is poor.
Endometriosis shows some characteristics of malignancy, such as the development of local and distant foci, and attachment to and invasion of other tissues with subsequent damage to these tissues. Endometriosis also is characterized by recurrent, unregulated cell proliferation and estrogen-dependent growth.
Our attempts during the past 2 decades to detect ovarian carcinoma at the early stages through a combined screening modality involving transvaginal ultrasound and a test for the serum level of cancer antigen 125 have failed to provide any survival benefit or even any measurable reduction in morbidity. Today, early-stage ovarian carcinoma, which has a 5-year survival rate of more than 90%, is diagnosed in only a minority of women.
There is good news, however. In recent years our insight into the pathophysiology of ovarian cancer has deepened, providing us with a new paradigm for ovarian cancer pathogenesis that divides ovarian epithelial carcinoma into two distinct types with distinct molecular profiles – one which originates largely in the distal portion of the fallopian tube and the other which traces back to endometriosis.
This new paradigm strengthens and helps to explain the reported association between endometriosis and ovarian cancer. It also has important clinical implications for current practice. While we have much more to learn about the etiology of endometriosis and the causes of malignant transformation, our current knowledge provides a strong rationale for identification and close monitoring of some patients with endometriosis deemed at risk for ovarian cancer, risk-reducing medical management, earlier and more meticulous surgical treatment, and close monitoring.
By combining this new approach to endometriosis with consideration of salpingectomy after completion of childbearing, we have an unprecedented opportunity to reduce the incidence of epithelial ovarian cancer.
Dual pathogenesis
The majority of ovarian cancers are of epithelial origin and fall into four histologic categories: serous, endometrioid, clear cell, and mucinous. In recent years, we have gained a deeper understanding of the pathogenesis of ovarian carcinoma, with an array of epidemiologic, histologic, and molecular data showing us that epithelial ovarian cancers are also of two distinct types (Am J Obstet Gynecol. 2015 Sep;213[3]:262-7).
One of these types, a high-grade serous carcinoma, appears to arise in many cases in the epithelium of the fallopian tube. The other type of tumor is a low-grade carcinoma – particularly of the endometrioid and clear cell histologic subtypes – that originates largely from ovarian endometriotic lesions or from borderline serous tumors in the case of serous histology.
The majority of diagnosed stage 1 ovarian cancers are carcinomas of this low-grade type and not high-grade serous carcinomas. In a study of 76 consecutive stage 1 carcinomas, investigators found that ovarian endometriosis was present in 40 of the 76 cases. More than two-thirds of the 76 cases (71%) were nonserous cancers, and almost all of these cases were associated with endometriosis based on histologic examination (Fertil Steril. 2007 Oct;88[4]:906-10).
This study was among the first to show that the majority of stage 1 ovarian carcinomas are not high-grade serous carcinomas, but rather nonserous, primarily endometrioid and clear cell, cancers. The research demonstrated that endometriosis should be viewed as a potential precursor lesion to specific subtypes of ovarian cancer.
The malignant transformation of endometriosis was first suggested by Dr. J. A. Sampson in 1925, and a number of studies – in addition to the 2007 landmark study – have since described ovarian cancer arising from endometriosis, based on the frequent co-occurrence in surgical specimens.
Most recently, a study from the Ovarian Cancer Association Consortium (OCAC) found that women who reported a history of endometriosis had a significantly higher risk of developing ovarian cancer than the general population (odds ratio, 1.46).
Investigators of this critical study pooled data from 13 ovarian cancer case-control studies involving more than 13,226 controls and 7,911 women with invasive epithelial ovarian cancer – 818 (6.2%) and 738 (9.3%) of whom, respectively, reported a history of endometriosis. Specifically, they determined that self-reported endometriosis was associated with a 3.05-fold increased risk for clear cell invasive ovarian cancer and a 2.04-fold increased risk of endometrioid ovarian cancer.
Moreover, a significant association between preexisting endometriosis and low-grade serous invasive ovarian cancer (OR, 2.11) was demonstrated, while no association was found between endometriosis and the risk of high-grade serous invasive ovarian cancer (Lancet Oncol. 2012 Apr;13[4]:385-94).
A second recently published report – a meta-analysis of 20 case-control and 15 cohort studies published between 1990 and 2012 and involving more than 444,000 patients – found that endometriosis increased cancer risk in case-control or two-arm cohort studies by 27% (relative risk, 1.265) and by approximately 80% in single-arm cohort studies (standard incidence ratio, 1.797). Endometrioid and clear cell carcinomas were more common in endometriosis-associated ovarian cancer, while serous carcinoma was less frequent (Br J Cancer. 2014 Apr 2;110[7]:1878-90).
Findings of both of these large studies have served to clarify the association between endometriosis and specific histologic subtypes and suggested that there are important differences in the pathogenesis of low-grade and high-grade serous ovarian carcinomas.
Clinical implications
It is not clear what causes malignant transformation or what predisposes some patients with endometriosis to develop ovarian cancer, but the risk likely involves genetic and epigenetic influences as well as immunologic, inflammatory, and hormonal factors.
The molecular profiles of the main two types of ovarian cancer are different: While the majority of high-grade serous ovarian tumors are characterized by TP53 mutations, the low-grade carcinomas are characterized by a variety of mutations, including KRAS, BRAF, ERBB2, CTNNB1, and BCL2 mutations.
There currently are not enough data to recommend genetic screening tests in patients with endometriosis, but our hope is that we eventually will be able to screen for “high-risk” endometriotic lesions by testing for genes specific to various histologic subtypes of low-grade ovarian cancer, or by finding and utilizing other biomarkers.
In the meantime, we believe it is important to more thoroughly treat endometriosis and to identify and follow women with a history of the condition, especially those with a long-standing history, those with a history of endometriosis associated with infertility, and those with ovarian endometrioma. Each of these factors predisposes patients to a higher risk of malignant transformation.
Complete surgical resection of all visible endometriosis is the most effective treatment and will afford the best cancer prevention, even in women who are asymptomatic. In a recent Swedish national registry case-control study, women who underwent radical surgical excision of all visible endometriosis were significantly less likely (OR, 0.30) to develop ovarian cancer (Acta Obstet Gynecol Scand. 2013 May;92[5]:546-54).
Suppressive hormonal therapy is another treatment option for patients with no interest in conceiving. Most large endometriomas are functional ovarian cysts that have been invaded by cortical ovarian endometriosis or by small primary endometriomas (J Reprod Med. 1992 Sep;37[9]:771-6).
While hormonal therapy will not always result in complete regression of endometriotic lesions, it will decrease the recurrence rate of endometriomas and can be considered for long-term prevention of potentially premalignant lesions. It is most effective when it follows surgical excision of endometriomas and associated endometriosis.
A patient who has completed childbearing at the time of surgical resection may be offered bilateral salpingectomy, regardless of menopausal status. Salpingectomy in both average and high-risk populations (e.g., BRCA 1/2 carriers) not only prevents high-grade serous carcinoma by eliminating the site of origin, but also may decrease the risk of endometrioid and clear cell carcinoma by blocking the passageway that enables the flow of endometrium and factors that induce inflammation. It is estimated that the procedure reduces the risk of ovarian cancer by 40%.
Interestingly, tubal ligation has historically been shown to decrease the risk of ovarian cancer, and recent data have shown that the risk of endometrioid and clear cell carcinoma is cut even more than the risk of high-grade serous carcinoma (Int J Epidemiol. 2013 Apr;42[2]:579-89).
The Society of Gynecologic Oncology recommends that risk-reducing salpingectomy be considered at the time of hysterectomy or other abdominal or pelvic surgery, and in lieu of tubal ligation. The American College of Obstetricians and Gynecologists similarly has stated that prophylactic salpingectomy may offer clinicians the opportunity to prevent ovarian cancer in their patients. Salpingectomy is an important option for all patients, but is especially important when the fallopian tubes are found to be damaged by endometriosis and/or pelvic inflammatory disease. When imaging studies show that endometriomas are present and resection is not performed, pelvic ultrasound should become part of the patient’s routine examination.
Most endometriomas have a homogeneous appearance; any significant increase in size or a change in the homogeneous cystic characteristics to a more heterogeneous appearance with mural components should raise suspicion about malignant change.
It can be difficult to detect relatively small endocystic components with ultrasound, so if there is any doubt about whether there is some heterogeneous consistency, an MRI should be performed. MRI is showing more promise in detecting malignant change. Hyperdense mural nodules within the ovary and rapid growth of an endometrioma have both been associated with malignant transformation and can be seen on these images.
In a cohort study comparing MRI findings of 10 patients with ovarian adenocarcinoma to 10 patients with benign endometriomas, investigators found mural nodules in all 10 malignancies but in only three of the benign cases (AJR Am J Roentgenol. 2000 Nov;175[5]:1423-30).
Long-term follow-up is necessary to understand the timeline of transformation in patients with mural nodules. This together with increasing knowledge of molecular events underpinning evolution of endometriosis will lead to better screening and preventive strategies.
Dr. Nezhat is the director of minimally invasive gynecologic surgery and robotics at Winthrop University Hospital in Mineola, N.Y., and an adjunct professor of obstetrics, gynecology, and reproductive medicine at the State University of New York at Stony Brook. He reported having no financial disclosures.
Endometriosis is a common condition, occurring in this country in 1 of 10 women of reproductive age. An association between endometriosis and subsequent ovarian carcinoma has been reported for decades, yet it is only recently that our knowledge has deepened enough to support more rational methods for preventing the malignancy.
Each year, approximately 22,000 new cases of ovarian cancer are diagnosed. The lifetime risk of developing this malignancy is low, but it is the deadliest of the gynecologic malignancies, with diagnosis usually made in advanced stages when prognosis is poor.
Endometriosis shows some characteristics of malignancy, such as the development of local and distant foci, and attachment to and invasion of other tissues with subsequent damage to these tissues. Endometriosis also is characterized by recurrent, unregulated cell proliferation and estrogen-dependent growth.
Our attempts during the past 2 decades to detect ovarian carcinoma at the early stages through a combined screening modality involving transvaginal ultrasound and a test for the serum level of cancer antigen 125 have failed to provide any survival benefit or even any measurable reduction in morbidity. Today, early-stage ovarian carcinoma, which has a 5-year survival rate of more than 90%, is diagnosed in only a minority of women.
There is good news, however. In recent years our insight into the pathophysiology of ovarian cancer has deepened, providing us with a new paradigm for ovarian cancer pathogenesis that divides ovarian epithelial carcinoma into two distinct types with distinct molecular profiles – one which originates largely in the distal portion of the fallopian tube and the other which traces back to endometriosis.
This new paradigm strengthens and helps to explain the reported association between endometriosis and ovarian cancer. It also has important clinical implications for current practice. While we have much more to learn about the etiology of endometriosis and the causes of malignant transformation, our current knowledge provides a strong rationale for identification and close monitoring of some patients with endometriosis deemed at risk for ovarian cancer, risk-reducing medical management, earlier and more meticulous surgical treatment, and close monitoring.
By combining this new approach to endometriosis with consideration of salpingectomy after completion of childbearing, we have an unprecedented opportunity to reduce the incidence of epithelial ovarian cancer.
Dual pathogenesis
The majority of ovarian cancers are of epithelial origin and fall into four histologic categories: serous, endometrioid, clear cell, and mucinous. In recent years, we have gained a deeper understanding of the pathogenesis of ovarian carcinoma, with an array of epidemiologic, histologic, and molecular data showing us that epithelial ovarian cancers are also of two distinct types (Am J Obstet Gynecol. 2015 Sep;213[3]:262-7).
One of these types, a high-grade serous carcinoma, appears to arise in many cases in the epithelium of the fallopian tube. The other type of tumor is a low-grade carcinoma – particularly of the endometrioid and clear cell histologic subtypes – that originates largely from ovarian endometriotic lesions or from borderline serous tumors in the case of serous histology.
The majority of diagnosed stage 1 ovarian cancers are carcinomas of this low-grade type and not high-grade serous carcinomas. In a study of 76 consecutive stage 1 carcinomas, investigators found that ovarian endometriosis was present in 40 of the 76 cases. More than two-thirds of the 76 cases (71%) were nonserous cancers, and almost all of these cases were associated with endometriosis based on histologic examination (Fertil Steril. 2007 Oct;88[4]:906-10).
This study was among the first to show that the majority of stage 1 ovarian carcinomas are not high-grade serous carcinomas, but rather nonserous, primarily endometrioid and clear cell, cancers. The research demonstrated that endometriosis should be viewed as a potential precursor lesion to specific subtypes of ovarian cancer.
The malignant transformation of endometriosis was first suggested by Dr. J. A. Sampson in 1925, and a number of studies – in addition to the 2007 landmark study – have since described ovarian cancer arising from endometriosis, based on the frequent co-occurrence in surgical specimens.
Most recently, a study from the Ovarian Cancer Association Consortium (OCAC) found that women who reported a history of endometriosis had a significantly higher risk of developing ovarian cancer than the general population (odds ratio, 1.46).
Investigators of this critical study pooled data from 13 ovarian cancer case-control studies involving more than 13,226 controls and 7,911 women with invasive epithelial ovarian cancer – 818 (6.2%) and 738 (9.3%) of whom, respectively, reported a history of endometriosis. Specifically, they determined that self-reported endometriosis was associated with a 3.05-fold increased risk for clear cell invasive ovarian cancer and a 2.04-fold increased risk of endometrioid ovarian cancer.
Moreover, a significant association between preexisting endometriosis and low-grade serous invasive ovarian cancer (OR, 2.11) was demonstrated, while no association was found between endometriosis and the risk of high-grade serous invasive ovarian cancer (Lancet Oncol. 2012 Apr;13[4]:385-94).
A second recently published report – a meta-analysis of 20 case-control and 15 cohort studies published between 1990 and 2012 and involving more than 444,000 patients – found that endometriosis increased cancer risk in case-control or two-arm cohort studies by 27% (relative risk, 1.265) and by approximately 80% in single-arm cohort studies (standard incidence ratio, 1.797). Endometrioid and clear cell carcinomas were more common in endometriosis-associated ovarian cancer, while serous carcinoma was less frequent (Br J Cancer. 2014 Apr 2;110[7]:1878-90).
Findings of both of these large studies have served to clarify the association between endometriosis and specific histologic subtypes and suggested that there are important differences in the pathogenesis of low-grade and high-grade serous ovarian carcinomas.
Clinical implications
It is not clear what causes malignant transformation or what predisposes some patients with endometriosis to develop ovarian cancer, but the risk likely involves genetic and epigenetic influences as well as immunologic, inflammatory, and hormonal factors.
The molecular profiles of the main two types of ovarian cancer are different: While the majority of high-grade serous ovarian tumors are characterized by TP53 mutations, the low-grade carcinomas are characterized by a variety of mutations, including KRAS, BRAF, ERBB2, CTNNB1, and BCL2 mutations.
There currently are not enough data to recommend genetic screening tests in patients with endometriosis, but our hope is that we eventually will be able to screen for “high-risk” endometriotic lesions by testing for genes specific to various histologic subtypes of low-grade ovarian cancer, or by finding and utilizing other biomarkers.
In the meantime, we believe it is important to more thoroughly treat endometriosis and to identify and follow women with a history of the condition, especially those with a long-standing history, those with a history of endometriosis associated with infertility, and those with ovarian endometrioma. Each of these factors predisposes patients to a higher risk of malignant transformation.
Complete surgical resection of all visible endometriosis is the most effective treatment and will afford the best cancer prevention, even in women who are asymptomatic. In a recent Swedish national registry case-control study, women who underwent radical surgical excision of all visible endometriosis were significantly less likely (OR, 0.30) to develop ovarian cancer (Acta Obstet Gynecol Scand. 2013 May;92[5]:546-54).
Suppressive hormonal therapy is another treatment option for patients with no interest in conceiving. Most large endometriomas are functional ovarian cysts that have been invaded by cortical ovarian endometriosis or by small primary endometriomas (J Reprod Med. 1992 Sep;37[9]:771-6).
While hormonal therapy will not always result in complete regression of endometriotic lesions, it will decrease the recurrence rate of endometriomas and can be considered for long-term prevention of potentially premalignant lesions. It is most effective when it follows surgical excision of endometriomas and associated endometriosis.
A patient who has completed childbearing at the time of surgical resection may be offered bilateral salpingectomy, regardless of menopausal status. Salpingectomy in both average and high-risk populations (e.g., BRCA 1/2 carriers) not only prevents high-grade serous carcinoma by eliminating the site of origin, but also may decrease the risk of endometrioid and clear cell carcinoma by blocking the passageway that enables the flow of endometrium and factors that induce inflammation. It is estimated that the procedure reduces the risk of ovarian cancer by 40%.
Interestingly, tubal ligation has historically been shown to decrease the risk of ovarian cancer, and recent data have shown that the risk of endometrioid and clear cell carcinoma is cut even more than the risk of high-grade serous carcinoma (Int J Epidemiol. 2013 Apr;42[2]:579-89).
The Society of Gynecologic Oncology recommends that risk-reducing salpingectomy be considered at the time of hysterectomy or other abdominal or pelvic surgery, and in lieu of tubal ligation. The American College of Obstetricians and Gynecologists similarly has stated that prophylactic salpingectomy may offer clinicians the opportunity to prevent ovarian cancer in their patients. Salpingectomy is an important option for all patients, but is especially important when the fallopian tubes are found to be damaged by endometriosis and/or pelvic inflammatory disease. When imaging studies show that endometriomas are present and resection is not performed, pelvic ultrasound should become part of the patient’s routine examination.
Most endometriomas have a homogeneous appearance; any significant increase in size or a change in the homogeneous cystic characteristics to a more heterogeneous appearance with mural components should raise suspicion about malignant change.
It can be difficult to detect relatively small endocystic components with ultrasound, so if there is any doubt about whether there is some heterogeneous consistency, an MRI should be performed. MRI is showing more promise in detecting malignant change. Hyperdense mural nodules within the ovary and rapid growth of an endometrioma have both been associated with malignant transformation and can be seen on these images.
In a cohort study comparing MRI findings of 10 patients with ovarian adenocarcinoma to 10 patients with benign endometriomas, investigators found mural nodules in all 10 malignancies but in only three of the benign cases (AJR Am J Roentgenol. 2000 Nov;175[5]:1423-30).
Long-term follow-up is necessary to understand the timeline of transformation in patients with mural nodules. This together with increasing knowledge of molecular events underpinning evolution of endometriosis will lead to better screening and preventive strategies.
Dr. Nezhat is the director of minimally invasive gynecologic surgery and robotics at Winthrop University Hospital in Mineola, N.Y., and an adjunct professor of obstetrics, gynecology, and reproductive medicine at the State University of New York at Stony Brook. He reported having no financial disclosures.
Endometriosis is a common condition, occurring in this country in 1 of 10 women of reproductive age. An association between endometriosis and subsequent ovarian carcinoma has been reported for decades, yet it is only recently that our knowledge has deepened enough to support more rational methods for preventing the malignancy.
Each year, approximately 22,000 new cases of ovarian cancer are diagnosed. The lifetime risk of developing this malignancy is low, but it is the deadliest of the gynecologic malignancies, with diagnosis usually made in advanced stages when prognosis is poor.
Endometriosis shows some characteristics of malignancy, such as the development of local and distant foci, and attachment to and invasion of other tissues with subsequent damage to these tissues. Endometriosis also is characterized by recurrent, unregulated cell proliferation and estrogen-dependent growth.
Our attempts during the past 2 decades to detect ovarian carcinoma at the early stages through a combined screening modality involving transvaginal ultrasound and a test for the serum level of cancer antigen 125 have failed to provide any survival benefit or even any measurable reduction in morbidity. Today, early-stage ovarian carcinoma, which has a 5-year survival rate of more than 90%, is diagnosed in only a minority of women.
There is good news, however. In recent years our insight into the pathophysiology of ovarian cancer has deepened, providing us with a new paradigm for ovarian cancer pathogenesis that divides ovarian epithelial carcinoma into two distinct types with distinct molecular profiles – one which originates largely in the distal portion of the fallopian tube and the other which traces back to endometriosis.
This new paradigm strengthens and helps to explain the reported association between endometriosis and ovarian cancer. It also has important clinical implications for current practice. While we have much more to learn about the etiology of endometriosis and the causes of malignant transformation, our current knowledge provides a strong rationale for identification and close monitoring of some patients with endometriosis deemed at risk for ovarian cancer, risk-reducing medical management, earlier and more meticulous surgical treatment, and close monitoring.
By combining this new approach to endometriosis with consideration of salpingectomy after completion of childbearing, we have an unprecedented opportunity to reduce the incidence of epithelial ovarian cancer.
Dual pathogenesis
The majority of ovarian cancers are of epithelial origin and fall into four histologic categories: serous, endometrioid, clear cell, and mucinous. In recent years, we have gained a deeper understanding of the pathogenesis of ovarian carcinoma, with an array of epidemiologic, histologic, and molecular data showing us that epithelial ovarian cancers are also of two distinct types (Am J Obstet Gynecol. 2015 Sep;213[3]:262-7).
One of these types, a high-grade serous carcinoma, appears to arise in many cases in the epithelium of the fallopian tube. The other type of tumor is a low-grade carcinoma – particularly of the endometrioid and clear cell histologic subtypes – that originates largely from ovarian endometriotic lesions or from borderline serous tumors in the case of serous histology.
The majority of diagnosed stage 1 ovarian cancers are carcinomas of this low-grade type and not high-grade serous carcinomas. In a study of 76 consecutive stage 1 carcinomas, investigators found that ovarian endometriosis was present in 40 of the 76 cases. More than two-thirds of the 76 cases (71%) were nonserous cancers, and almost all of these cases were associated with endometriosis based on histologic examination (Fertil Steril. 2007 Oct;88[4]:906-10).
This study was among the first to show that the majority of stage 1 ovarian carcinomas are not high-grade serous carcinomas, but rather nonserous, primarily endometrioid and clear cell, cancers. The research demonstrated that endometriosis should be viewed as a potential precursor lesion to specific subtypes of ovarian cancer.
The malignant transformation of endometriosis was first suggested by Dr. J. A. Sampson in 1925, and a number of studies – in addition to the 2007 landmark study – have since described ovarian cancer arising from endometriosis, based on the frequent co-occurrence in surgical specimens.
Most recently, a study from the Ovarian Cancer Association Consortium (OCAC) found that women who reported a history of endometriosis had a significantly higher risk of developing ovarian cancer than the general population (odds ratio, 1.46).
Investigators of this critical study pooled data from 13 ovarian cancer case-control studies involving more than 13,226 controls and 7,911 women with invasive epithelial ovarian cancer – 818 (6.2%) and 738 (9.3%) of whom, respectively, reported a history of endometriosis. Specifically, they determined that self-reported endometriosis was associated with a 3.05-fold increased risk for clear cell invasive ovarian cancer and a 2.04-fold increased risk of endometrioid ovarian cancer.
Moreover, a significant association between preexisting endometriosis and low-grade serous invasive ovarian cancer (OR, 2.11) was demonstrated, while no association was found between endometriosis and the risk of high-grade serous invasive ovarian cancer (Lancet Oncol. 2012 Apr;13[4]:385-94).
A second recently published report – a meta-analysis of 20 case-control and 15 cohort studies published between 1990 and 2012 and involving more than 444,000 patients – found that endometriosis increased cancer risk in case-control or two-arm cohort studies by 27% (relative risk, 1.265) and by approximately 80% in single-arm cohort studies (standard incidence ratio, 1.797). Endometrioid and clear cell carcinomas were more common in endometriosis-associated ovarian cancer, while serous carcinoma was less frequent (Br J Cancer. 2014 Apr 2;110[7]:1878-90).
Findings of both of these large studies have served to clarify the association between endometriosis and specific histologic subtypes and suggested that there are important differences in the pathogenesis of low-grade and high-grade serous ovarian carcinomas.
Clinical implications
It is not clear what causes malignant transformation or what predisposes some patients with endometriosis to develop ovarian cancer, but the risk likely involves genetic and epigenetic influences as well as immunologic, inflammatory, and hormonal factors.
The molecular profiles of the main two types of ovarian cancer are different: While the majority of high-grade serous ovarian tumors are characterized by TP53 mutations, the low-grade carcinomas are characterized by a variety of mutations, including KRAS, BRAF, ERBB2, CTNNB1, and BCL2 mutations.
There currently are not enough data to recommend genetic screening tests in patients with endometriosis, but our hope is that we eventually will be able to screen for “high-risk” endometriotic lesions by testing for genes specific to various histologic subtypes of low-grade ovarian cancer, or by finding and utilizing other biomarkers.
In the meantime, we believe it is important to more thoroughly treat endometriosis and to identify and follow women with a history of the condition, especially those with a long-standing history, those with a history of endometriosis associated with infertility, and those with ovarian endometrioma. Each of these factors predisposes patients to a higher risk of malignant transformation.
Complete surgical resection of all visible endometriosis is the most effective treatment and will afford the best cancer prevention, even in women who are asymptomatic. In a recent Swedish national registry case-control study, women who underwent radical surgical excision of all visible endometriosis were significantly less likely (OR, 0.30) to develop ovarian cancer (Acta Obstet Gynecol Scand. 2013 May;92[5]:546-54).
Suppressive hormonal therapy is another treatment option for patients with no interest in conceiving. Most large endometriomas are functional ovarian cysts that have been invaded by cortical ovarian endometriosis or by small primary endometriomas (J Reprod Med. 1992 Sep;37[9]:771-6).
While hormonal therapy will not always result in complete regression of endometriotic lesions, it will decrease the recurrence rate of endometriomas and can be considered for long-term prevention of potentially premalignant lesions. It is most effective when it follows surgical excision of endometriomas and associated endometriosis.
A patient who has completed childbearing at the time of surgical resection may be offered bilateral salpingectomy, regardless of menopausal status. Salpingectomy in both average and high-risk populations (e.g., BRCA 1/2 carriers) not only prevents high-grade serous carcinoma by eliminating the site of origin, but also may decrease the risk of endometrioid and clear cell carcinoma by blocking the passageway that enables the flow of endometrium and factors that induce inflammation. It is estimated that the procedure reduces the risk of ovarian cancer by 40%.
Interestingly, tubal ligation has historically been shown to decrease the risk of ovarian cancer, and recent data have shown that the risk of endometrioid and clear cell carcinoma is cut even more than the risk of high-grade serous carcinoma (Int J Epidemiol. 2013 Apr;42[2]:579-89).
The Society of Gynecologic Oncology recommends that risk-reducing salpingectomy be considered at the time of hysterectomy or other abdominal or pelvic surgery, and in lieu of tubal ligation. The American College of Obstetricians and Gynecologists similarly has stated that prophylactic salpingectomy may offer clinicians the opportunity to prevent ovarian cancer in their patients. Salpingectomy is an important option for all patients, but is especially important when the fallopian tubes are found to be damaged by endometriosis and/or pelvic inflammatory disease. When imaging studies show that endometriomas are present and resection is not performed, pelvic ultrasound should become part of the patient’s routine examination.
Most endometriomas have a homogeneous appearance; any significant increase in size or a change in the homogeneous cystic characteristics to a more heterogeneous appearance with mural components should raise suspicion about malignant change.
It can be difficult to detect relatively small endocystic components with ultrasound, so if there is any doubt about whether there is some heterogeneous consistency, an MRI should be performed. MRI is showing more promise in detecting malignant change. Hyperdense mural nodules within the ovary and rapid growth of an endometrioma have both been associated with malignant transformation and can be seen on these images.
In a cohort study comparing MRI findings of 10 patients with ovarian adenocarcinoma to 10 patients with benign endometriomas, investigators found mural nodules in all 10 malignancies but in only three of the benign cases (AJR Am J Roentgenol. 2000 Nov;175[5]:1423-30).
Long-term follow-up is necessary to understand the timeline of transformation in patients with mural nodules. This together with increasing knowledge of molecular events underpinning evolution of endometriosis will lead to better screening and preventive strategies.
Dr. Nezhat is the director of minimally invasive gynecologic surgery and robotics at Winthrop University Hospital in Mineola, N.Y., and an adjunct professor of obstetrics, gynecology, and reproductive medicine at the State University of New York at Stony Brook. He reported having no financial disclosures.
The impact of endometriosis on ovarian cancer
During an ob.gyn. rotation, a medical student quickly learns the risks related to endometriosis; that is, pelvic pain, abnormal uterine bleeding, and infertility. With more experience, the young practitioner realizes the concern of unopposed estrogen therapy in patients with a history of endometriosis (i.e., cancer).
Now, in this excellent discussion by Dr. Farr Nezhat, for the current edition of the Master Class in Gynecologic Surgery, he describes the risk of endometriosis and ovarian cancer. Not only does Dr. Nezhat present data revealing the increased association between ovarian cancer and endometriosis, but he goes on to describe the usual type of epithelial ovarian cancer that is noted in the patient with endometriosis.
Dr. Nezhat describes women who appear to be predisposed to malignant transformation and provides current recommendations to lower the risk of malignancy in patients with endometriosis. This includes complete surgical resection of endometriosis, routine ultrasound/MRI if endometriosis is not resected, suppressive hormonal therapy, and bilateral salpingectomy. Moreover, Dr. Nezhat looks to the future and the possibility of genetic screening tests.
Dr. Nezhat is board certified in gynecologic oncology and is world renowned for his work with advanced laparoscopic and robotic surgery for the treatment of gynecologic cancers and complex benign conditions. He is the director of minimally invasive gynecologic surgery and robotics at Winthrop University Hospital in Mineola, N.Y., and an adjunct professor of obstetrics, gynecology, and reproductive medicine at State University of New York at Stony Brook.
His main areas of interest and research include early detection and treatment of early and advanced ovarian cancer, as well as cancer arising in endometriosis. Dr. Nezhat has authored and coauthored more than 200 medical and scientific manuscripts and book chapters.
Dr. Miller is a clinical associate professor at the University of Illinois at Chicago, and a past president of the AAGL and the International Society for Gynecologic Endoscopy (ISGE). He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in private practice in Naperville and Schaumburg, Ill.; director of minimally invasive gynecologic surgery and the director of the AAGL/Society of Reproductive Surgery fellowship in minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill.; and the medical editor of this column, Master Class. Dr. Miller reported having no financial disclosures relevant to this column.
During an ob.gyn. rotation, a medical student quickly learns the risks related to endometriosis; that is, pelvic pain, abnormal uterine bleeding, and infertility. With more experience, the young practitioner realizes the concern of unopposed estrogen therapy in patients with a history of endometriosis (i.e., cancer).
Now, in this excellent discussion by Dr. Farr Nezhat, for the current edition of the Master Class in Gynecologic Surgery, he describes the risk of endometriosis and ovarian cancer. Not only does Dr. Nezhat present data revealing the increased association between ovarian cancer and endometriosis, but he goes on to describe the usual type of epithelial ovarian cancer that is noted in the patient with endometriosis.
Dr. Nezhat describes women who appear to be predisposed to malignant transformation and provides current recommendations to lower the risk of malignancy in patients with endometriosis. This includes complete surgical resection of endometriosis, routine ultrasound/MRI if endometriosis is not resected, suppressive hormonal therapy, and bilateral salpingectomy. Moreover, Dr. Nezhat looks to the future and the possibility of genetic screening tests.
Dr. Nezhat is board certified in gynecologic oncology and is world renowned for his work with advanced laparoscopic and robotic surgery for the treatment of gynecologic cancers and complex benign conditions. He is the director of minimally invasive gynecologic surgery and robotics at Winthrop University Hospital in Mineola, N.Y., and an adjunct professor of obstetrics, gynecology, and reproductive medicine at State University of New York at Stony Brook.
His main areas of interest and research include early detection and treatment of early and advanced ovarian cancer, as well as cancer arising in endometriosis. Dr. Nezhat has authored and coauthored more than 200 medical and scientific manuscripts and book chapters.
Dr. Miller is a clinical associate professor at the University of Illinois at Chicago, and a past president of the AAGL and the International Society for Gynecologic Endoscopy (ISGE). He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in private practice in Naperville and Schaumburg, Ill.; director of minimally invasive gynecologic surgery and the director of the AAGL/Society of Reproductive Surgery fellowship in minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill.; and the medical editor of this column, Master Class. Dr. Miller reported having no financial disclosures relevant to this column.
During an ob.gyn. rotation, a medical student quickly learns the risks related to endometriosis; that is, pelvic pain, abnormal uterine bleeding, and infertility. With more experience, the young practitioner realizes the concern of unopposed estrogen therapy in patients with a history of endometriosis (i.e., cancer).
Now, in this excellent discussion by Dr. Farr Nezhat, for the current edition of the Master Class in Gynecologic Surgery, he describes the risk of endometriosis and ovarian cancer. Not only does Dr. Nezhat present data revealing the increased association between ovarian cancer and endometriosis, but he goes on to describe the usual type of epithelial ovarian cancer that is noted in the patient with endometriosis.
Dr. Nezhat describes women who appear to be predisposed to malignant transformation and provides current recommendations to lower the risk of malignancy in patients with endometriosis. This includes complete surgical resection of endometriosis, routine ultrasound/MRI if endometriosis is not resected, suppressive hormonal therapy, and bilateral salpingectomy. Moreover, Dr. Nezhat looks to the future and the possibility of genetic screening tests.
Dr. Nezhat is board certified in gynecologic oncology and is world renowned for his work with advanced laparoscopic and robotic surgery for the treatment of gynecologic cancers and complex benign conditions. He is the director of minimally invasive gynecologic surgery and robotics at Winthrop University Hospital in Mineola, N.Y., and an adjunct professor of obstetrics, gynecology, and reproductive medicine at State University of New York at Stony Brook.
His main areas of interest and research include early detection and treatment of early and advanced ovarian cancer, as well as cancer arising in endometriosis. Dr. Nezhat has authored and coauthored more than 200 medical and scientific manuscripts and book chapters.
Dr. Miller is a clinical associate professor at the University of Illinois at Chicago, and a past president of the AAGL and the International Society for Gynecologic Endoscopy (ISGE). He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in private practice in Naperville and Schaumburg, Ill.; director of minimally invasive gynecologic surgery and the director of the AAGL/Society of Reproductive Surgery fellowship in minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill.; and the medical editor of this column, Master Class. Dr. Miller reported having no financial disclosures relevant to this column.
New data points to slower course of labor
Only recently has evidence emerged that challenges our long-held understanding of “normal” and “abnormal” labor. We now know there is a much wider range of normal labor progress in women who go on to have good labor outcomes. We have a new labor curve to guide us – one that shows us, for example, that active labor occurs most commonly after 6 cm dilation rather than 4 cm as we’d previously thought.
By appreciating this new labor paradigm, we can potentially have a significant impact on the cesarean rate in the United States. While our use of the older labor curve is not the only reason for the rise in cesarean deliveries over the last 30 years, it very likely has played a role. A study published in 2011 of more than 32,000 live births at a major academic hospital demonstrated that one of the most common reasons for primary cesarean is abnormal labor or arrest (Obstet Gynecol. 2011 Jul;118[1]:29-38).
Another study by the Consortium on Safe Labor – an analysis of labor and delivery information from more than 228,000 women across the United States – showed that half of the cesarean deliveries performed for dystocia in women undergoing labor induction were performed before 6 cm of cervical dilation and relatively soon after the previous cervical examination (Am J Obstet Gynecol. 2010 Oct; 203[4]: 326.e1–326.e10).
Our new labor paradigm brings to the forefront a host of new issues and questions about how we can best manage labor to optimize outcomes. In a way, recent discoveries about labor progress have highlighted a dearth of evidence and made “old” issues in labor management seem new and urgent.
As we strive to learn more, however, we are challenged to change our practices and behavior at the bedside with the evidence we currently have. By appreciating both the new labor curve and our current understanding of how labor induction, obesity, and other patient characteristics and clinical conditions can affect labor progress, we can expect that many women will simply progress much more slowly than was historically expected.
As long as we have indications of the well-being of the baby and the well-being of the mother, a slower but progressive labor in the first stage should not prompt us to intervene. We should no longer apply the standards of active-phase progress – standards that have traditionally driven our diagnoses of labor dystocia – until the patient has achieved 6 cm of dilation.
The labor curve that had shaped our thinking about normal and abnormal labor progress until recently was developed by Dr. Emanuel Friedman. Based on findings from a prospective cohort study of 500 nulliparous women, Dr. Friedman plotted labor progress with centimeters of cervical dilation on the Y-axis and time on the X-axis, and divided labor into several stages and phases. In this curve, the rate of change of cervical dilation over time started increasing significantly at 4 cm; this period of increasing slope defined the active phase of labor.
Abnormal labor progress in the active phase was then defined, based on the 95th percentile, as cervical dilation of less than 1.2 cm per hour for nulliparous women and less than 1.5 cm per hour for multiparous women. Based on Dr. Friedman’s work, a woman was deemed to be in active-phase arrest when she had no cervical changes for 2 hours or more while having adequate uterine contractions and cervical dilation of at least 4 cm. These concepts came to govern labor management.
The paradigm shifted when the Consortium on Safe Labor reported in 2010 on a retrospective cohort study of more than 62,000 women at 19 U.S. hospitals. The women had a singleton term gestation, spontaneous labor, vertex presentation, vaginal delivery, and a normal perinatal outcome. In their analysis of labor and delivery information, Dr. Jun Zhang of the National Institutes of Health’s Eunice Kennedy Shriver National Institute of Child Health and Human Development and his colleagues accounted for the fact that the exact times of cervical change are unknown.
They used modern statistical methods and analytical tools that took into account the specific nature of cervical dilation data – that cervical measurements are interval-censored (we never know the exact time when a woman’s cervix changes) and that multiple exams of the cervix in the same patient are not independent (Obstet Gynecol. 2010 Dec;116[6]:1281-7).
The methodology used in the Consortium study accounted for both the interval-censored and repeated-measures nature of cervical dilation data. It thus addressed analytical flaws in the previous approach to labor data, which was purely descriptive of the exam findings and did not consider the nature of the data itself.
Under the new analysis and in the larger, contemporary population of patients, the period of increasing slope was found to occur most commonly after 6 cm, not 4 cm. The slowest 5% of nulliparous women had cervical dilation of 0.4 cm per hour (with the median at 1.9 cm per hour), compared with 1.2 cm per hour (with a median of 3.0 cm per hour) as in the Friedman data.
Dr. Zhang’s study showed us that labor may take more than 6 hours to progress from 4 to 5 cm dilation, and more than 3 hours to progress from 5 to 6 cm dilation – a rate of progress that is significantly slower than what Dr. Friedman had described. The new data showed us, moreover, that from 4 cm-6 cm dilation, nulliparous and multiparous women progressed similarly slowly. Beyond 6 cm, multiparous women dilated more rapidly, with a steeper acceleration phase than previously described.
A consensus statement published in 2014 by the American College of Obstetricians and Gynecologists (ACOG) and the Society for Maternal-Fetal Medicine (SMFM) on “Safe Prevention of the Primary Cesarean Delivery” encourages use of the Consortium data to revisit the definition of labor dystocia. While the data “do not directly address an optimal duration for the diagnosis of active-phase protraction or labor arrest, [they] do suggest that neither should be diagnosed before 6 cm dilation” (Obstet Gynecol. 2014 Mar;123[3]:693-711).
The ACOG-SMFM statement makes a series of recommendations for managing the first and second stages of labor, based not only on the Consortium data but on a broader literature review. It recommends that if mother and fetus appear well, cesarean delivery for active-phase arrest in the first stage of labor be reserved for women of at least 6 cm of dilation with ruptured membranes who fail to progress despite 4 hours of adequate uterine activity, or at least 6 hours of oxytocin administration with inadequate uterine activity and no cervical change.
Regarding the latent phase of labor, the statement says that most women with a prolonged latent phase ultimately will enter the active phase with expectant management. It advises that a prolonged latent phase (for example, greater than 20 hours in nulliparous women and greater than 14 hours in multiparous women) should not be an isolated indication for cesarean delivery.
The consensus statement also recognizes recent data showing that women who undergo labor induction have an even slower “normal” course of labor, particularly a longer latent phase, than women who labor spontaneously. A retrospective cohort study of more than 5,000 women, for instance, found that before 6 cm, women whose labor is induced can spend up to 10 hours to achieve each 1 cm of dilation (Obstet Gynecol. 2012 Jun;119[6]:1113-8).
As long as maternal and fetal status are reassuring, the statement says, cesarean deliveries for failed induction of labor in the latent phase can be avoided by allowing longer durations of the latent phase (up to 24 hours) and by requiring that oxytocin be administered for 12-18 hours after membrane rupture before deeming induction a failure.
Each of these described recommendations were graded in the ACOG-SMFM consensus document as “strong” recommendations with “moderate quality evidence.”
Examining our standards
Moving forward, we must further develop and define our thresholds for identifying who will most benefit from a cesarean delivery. We have many specific aspects of labor management to address as well, such as the optimal timing of artificial membrane rupture and the safety and efficacy of different oxytocin protocols. We may also want to revisit recommendations for serial cervical assessment, possibly adjusting the intervals given our understanding of the new labor curve.
Under the new labor paradigm, moreover, we must think not only about the clinical decisions we make at the bedside, but about the decisions we make early in the labor management process.
The timing of admission is one such decision. A statement published in 2012 on “Preventing the First Cesarean Delivery” by ACOG, SMFM, and the Eunice Kennedy Shriver National Institute of Child Health and Human Development advises us to avoid admittance of women during the early latent phase of labor (Obstet Gynecol. 2012 Nov;120[5]:1181-93).
It may even be advisable that we consider admittance at higher cervical dilation. A study published this year shows that women admitted at less than 6 cm of dilation had an increased risk of cesarean delivery, compared with women admitted at higher cervical dilation (Am J Perinatol. 2016 Jan;33[2]:188-94). We have more to learn, but certainly, given what we know now about labor progress and the start of active labor, the timing of admission is an important factor to consider.
The second stage of labor, defined as the interval from complete cervical dilation through delivery of the fetus, presents many questions as well. There is a paucity of quality published data concerning what is normal, how long the stage should last, and how we should manage it. Historically, we have been taught to allow 2 hours of pushing for nulliparous women and 1 hour for multiparous women, when epidural anesthesia has not been administered, and to add an additional hour when epidural is used.
The 2014 ACOG-SMFM consensus statement recommends extending each of these limits by an hour, if maternal and fetal conditions permit, so that we allow at least 3 hours of pushing for nulliparous women and at least 2 hours for multiparous women before diagnosing arrest of labor in the second stage. Longer durations may be appropriate with the use of epidural anesthesia and on an individualized basis.
At this time, it is unclear whether there is any absolute maximum length of time beyond which all women in the second stage of labor should undergo cesarean delivery. We also still do not know the optimal technique for managing maternal pushing during the second stage. Should women with an epidural push right away or should they allow for a period of spontaneous descent? Many of the high-quality studies reported thus far that compare delayed and immediate pushing have limited applicability to current practice because they involved now-obsolete midpelvic forceps deliveries. A large multicenter randomized trial currently underway should provide us with some answers.
Dr. Cahill is an associate professor and chief of the division of maternal-fetal medicine in the department of obstetrics and gynecology at Washington University School of Medicine in St. Louis. She reported having no relevant financial disclosures.
Only recently has evidence emerged that challenges our long-held understanding of “normal” and “abnormal” labor. We now know there is a much wider range of normal labor progress in women who go on to have good labor outcomes. We have a new labor curve to guide us – one that shows us, for example, that active labor occurs most commonly after 6 cm dilation rather than 4 cm as we’d previously thought.
By appreciating this new labor paradigm, we can potentially have a significant impact on the cesarean rate in the United States. While our use of the older labor curve is not the only reason for the rise in cesarean deliveries over the last 30 years, it very likely has played a role. A study published in 2011 of more than 32,000 live births at a major academic hospital demonstrated that one of the most common reasons for primary cesarean is abnormal labor or arrest (Obstet Gynecol. 2011 Jul;118[1]:29-38).
Another study by the Consortium on Safe Labor – an analysis of labor and delivery information from more than 228,000 women across the United States – showed that half of the cesarean deliveries performed for dystocia in women undergoing labor induction were performed before 6 cm of cervical dilation and relatively soon after the previous cervical examination (Am J Obstet Gynecol. 2010 Oct; 203[4]: 326.e1–326.e10).
Our new labor paradigm brings to the forefront a host of new issues and questions about how we can best manage labor to optimize outcomes. In a way, recent discoveries about labor progress have highlighted a dearth of evidence and made “old” issues in labor management seem new and urgent.
As we strive to learn more, however, we are challenged to change our practices and behavior at the bedside with the evidence we currently have. By appreciating both the new labor curve and our current understanding of how labor induction, obesity, and other patient characteristics and clinical conditions can affect labor progress, we can expect that many women will simply progress much more slowly than was historically expected.
As long as we have indications of the well-being of the baby and the well-being of the mother, a slower but progressive labor in the first stage should not prompt us to intervene. We should no longer apply the standards of active-phase progress – standards that have traditionally driven our diagnoses of labor dystocia – until the patient has achieved 6 cm of dilation.
The labor curve that had shaped our thinking about normal and abnormal labor progress until recently was developed by Dr. Emanuel Friedman. Based on findings from a prospective cohort study of 500 nulliparous women, Dr. Friedman plotted labor progress with centimeters of cervical dilation on the Y-axis and time on the X-axis, and divided labor into several stages and phases. In this curve, the rate of change of cervical dilation over time started increasing significantly at 4 cm; this period of increasing slope defined the active phase of labor.
Abnormal labor progress in the active phase was then defined, based on the 95th percentile, as cervical dilation of less than 1.2 cm per hour for nulliparous women and less than 1.5 cm per hour for multiparous women. Based on Dr. Friedman’s work, a woman was deemed to be in active-phase arrest when she had no cervical changes for 2 hours or more while having adequate uterine contractions and cervical dilation of at least 4 cm. These concepts came to govern labor management.
The paradigm shifted when the Consortium on Safe Labor reported in 2010 on a retrospective cohort study of more than 62,000 women at 19 U.S. hospitals. The women had a singleton term gestation, spontaneous labor, vertex presentation, vaginal delivery, and a normal perinatal outcome. In their analysis of labor and delivery information, Dr. Jun Zhang of the National Institutes of Health’s Eunice Kennedy Shriver National Institute of Child Health and Human Development and his colleagues accounted for the fact that the exact times of cervical change are unknown.
They used modern statistical methods and analytical tools that took into account the specific nature of cervical dilation data – that cervical measurements are interval-censored (we never know the exact time when a woman’s cervix changes) and that multiple exams of the cervix in the same patient are not independent (Obstet Gynecol. 2010 Dec;116[6]:1281-7).
The methodology used in the Consortium study accounted for both the interval-censored and repeated-measures nature of cervical dilation data. It thus addressed analytical flaws in the previous approach to labor data, which was purely descriptive of the exam findings and did not consider the nature of the data itself.
Under the new analysis and in the larger, contemporary population of patients, the period of increasing slope was found to occur most commonly after 6 cm, not 4 cm. The slowest 5% of nulliparous women had cervical dilation of 0.4 cm per hour (with the median at 1.9 cm per hour), compared with 1.2 cm per hour (with a median of 3.0 cm per hour) as in the Friedman data.
Dr. Zhang’s study showed us that labor may take more than 6 hours to progress from 4 to 5 cm dilation, and more than 3 hours to progress from 5 to 6 cm dilation – a rate of progress that is significantly slower than what Dr. Friedman had described. The new data showed us, moreover, that from 4 cm-6 cm dilation, nulliparous and multiparous women progressed similarly slowly. Beyond 6 cm, multiparous women dilated more rapidly, with a steeper acceleration phase than previously described.
A consensus statement published in 2014 by the American College of Obstetricians and Gynecologists (ACOG) and the Society for Maternal-Fetal Medicine (SMFM) on “Safe Prevention of the Primary Cesarean Delivery” encourages use of the Consortium data to revisit the definition of labor dystocia. While the data “do not directly address an optimal duration for the diagnosis of active-phase protraction or labor arrest, [they] do suggest that neither should be diagnosed before 6 cm dilation” (Obstet Gynecol. 2014 Mar;123[3]:693-711).
The ACOG-SMFM statement makes a series of recommendations for managing the first and second stages of labor, based not only on the Consortium data but on a broader literature review. It recommends that if mother and fetus appear well, cesarean delivery for active-phase arrest in the first stage of labor be reserved for women of at least 6 cm of dilation with ruptured membranes who fail to progress despite 4 hours of adequate uterine activity, or at least 6 hours of oxytocin administration with inadequate uterine activity and no cervical change.
Regarding the latent phase of labor, the statement says that most women with a prolonged latent phase ultimately will enter the active phase with expectant management. It advises that a prolonged latent phase (for example, greater than 20 hours in nulliparous women and greater than 14 hours in multiparous women) should not be an isolated indication for cesarean delivery.
The consensus statement also recognizes recent data showing that women who undergo labor induction have an even slower “normal” course of labor, particularly a longer latent phase, than women who labor spontaneously. A retrospective cohort study of more than 5,000 women, for instance, found that before 6 cm, women whose labor is induced can spend up to 10 hours to achieve each 1 cm of dilation (Obstet Gynecol. 2012 Jun;119[6]:1113-8).
As long as maternal and fetal status are reassuring, the statement says, cesarean deliveries for failed induction of labor in the latent phase can be avoided by allowing longer durations of the latent phase (up to 24 hours) and by requiring that oxytocin be administered for 12-18 hours after membrane rupture before deeming induction a failure.
Each of these described recommendations were graded in the ACOG-SMFM consensus document as “strong” recommendations with “moderate quality evidence.”
Examining our standards
Moving forward, we must further develop and define our thresholds for identifying who will most benefit from a cesarean delivery. We have many specific aspects of labor management to address as well, such as the optimal timing of artificial membrane rupture and the safety and efficacy of different oxytocin protocols. We may also want to revisit recommendations for serial cervical assessment, possibly adjusting the intervals given our understanding of the new labor curve.
Under the new labor paradigm, moreover, we must think not only about the clinical decisions we make at the bedside, but about the decisions we make early in the labor management process.
The timing of admission is one such decision. A statement published in 2012 on “Preventing the First Cesarean Delivery” by ACOG, SMFM, and the Eunice Kennedy Shriver National Institute of Child Health and Human Development advises us to avoid admittance of women during the early latent phase of labor (Obstet Gynecol. 2012 Nov;120[5]:1181-93).
It may even be advisable that we consider admittance at higher cervical dilation. A study published this year shows that women admitted at less than 6 cm of dilation had an increased risk of cesarean delivery, compared with women admitted at higher cervical dilation (Am J Perinatol. 2016 Jan;33[2]:188-94). We have more to learn, but certainly, given what we know now about labor progress and the start of active labor, the timing of admission is an important factor to consider.
The second stage of labor, defined as the interval from complete cervical dilation through delivery of the fetus, presents many questions as well. There is a paucity of quality published data concerning what is normal, how long the stage should last, and how we should manage it. Historically, we have been taught to allow 2 hours of pushing for nulliparous women and 1 hour for multiparous women, when epidural anesthesia has not been administered, and to add an additional hour when epidural is used.
The 2014 ACOG-SMFM consensus statement recommends extending each of these limits by an hour, if maternal and fetal conditions permit, so that we allow at least 3 hours of pushing for nulliparous women and at least 2 hours for multiparous women before diagnosing arrest of labor in the second stage. Longer durations may be appropriate with the use of epidural anesthesia and on an individualized basis.
At this time, it is unclear whether there is any absolute maximum length of time beyond which all women in the second stage of labor should undergo cesarean delivery. We also still do not know the optimal technique for managing maternal pushing during the second stage. Should women with an epidural push right away or should they allow for a period of spontaneous descent? Many of the high-quality studies reported thus far that compare delayed and immediate pushing have limited applicability to current practice because they involved now-obsolete midpelvic forceps deliveries. A large multicenter randomized trial currently underway should provide us with some answers.
Dr. Cahill is an associate professor and chief of the division of maternal-fetal medicine in the department of obstetrics and gynecology at Washington University School of Medicine in St. Louis. She reported having no relevant financial disclosures.
Only recently has evidence emerged that challenges our long-held understanding of “normal” and “abnormal” labor. We now know there is a much wider range of normal labor progress in women who go on to have good labor outcomes. We have a new labor curve to guide us – one that shows us, for example, that active labor occurs most commonly after 6 cm dilation rather than 4 cm as we’d previously thought.
By appreciating this new labor paradigm, we can potentially have a significant impact on the cesarean rate in the United States. While our use of the older labor curve is not the only reason for the rise in cesarean deliveries over the last 30 years, it very likely has played a role. A study published in 2011 of more than 32,000 live births at a major academic hospital demonstrated that one of the most common reasons for primary cesarean is abnormal labor or arrest (Obstet Gynecol. 2011 Jul;118[1]:29-38).
Another study by the Consortium on Safe Labor – an analysis of labor and delivery information from more than 228,000 women across the United States – showed that half of the cesarean deliveries performed for dystocia in women undergoing labor induction were performed before 6 cm of cervical dilation and relatively soon after the previous cervical examination (Am J Obstet Gynecol. 2010 Oct; 203[4]: 326.e1–326.e10).
Our new labor paradigm brings to the forefront a host of new issues and questions about how we can best manage labor to optimize outcomes. In a way, recent discoveries about labor progress have highlighted a dearth of evidence and made “old” issues in labor management seem new and urgent.
As we strive to learn more, however, we are challenged to change our practices and behavior at the bedside with the evidence we currently have. By appreciating both the new labor curve and our current understanding of how labor induction, obesity, and other patient characteristics and clinical conditions can affect labor progress, we can expect that many women will simply progress much more slowly than was historically expected.
As long as we have indications of the well-being of the baby and the well-being of the mother, a slower but progressive labor in the first stage should not prompt us to intervene. We should no longer apply the standards of active-phase progress – standards that have traditionally driven our diagnoses of labor dystocia – until the patient has achieved 6 cm of dilation.
The labor curve that had shaped our thinking about normal and abnormal labor progress until recently was developed by Dr. Emanuel Friedman. Based on findings from a prospective cohort study of 500 nulliparous women, Dr. Friedman plotted labor progress with centimeters of cervical dilation on the Y-axis and time on the X-axis, and divided labor into several stages and phases. In this curve, the rate of change of cervical dilation over time started increasing significantly at 4 cm; this period of increasing slope defined the active phase of labor.
Abnormal labor progress in the active phase was then defined, based on the 95th percentile, as cervical dilation of less than 1.2 cm per hour for nulliparous women and less than 1.5 cm per hour for multiparous women. Based on Dr. Friedman’s work, a woman was deemed to be in active-phase arrest when she had no cervical changes for 2 hours or more while having adequate uterine contractions and cervical dilation of at least 4 cm. These concepts came to govern labor management.
The paradigm shifted when the Consortium on Safe Labor reported in 2010 on a retrospective cohort study of more than 62,000 women at 19 U.S. hospitals. The women had a singleton term gestation, spontaneous labor, vertex presentation, vaginal delivery, and a normal perinatal outcome. In their analysis of labor and delivery information, Dr. Jun Zhang of the National Institutes of Health’s Eunice Kennedy Shriver National Institute of Child Health and Human Development and his colleagues accounted for the fact that the exact times of cervical change are unknown.
They used modern statistical methods and analytical tools that took into account the specific nature of cervical dilation data – that cervical measurements are interval-censored (we never know the exact time when a woman’s cervix changes) and that multiple exams of the cervix in the same patient are not independent (Obstet Gynecol. 2010 Dec;116[6]:1281-7).
The methodology used in the Consortium study accounted for both the interval-censored and repeated-measures nature of cervical dilation data. It thus addressed analytical flaws in the previous approach to labor data, which was purely descriptive of the exam findings and did not consider the nature of the data itself.
Under the new analysis and in the larger, contemporary population of patients, the period of increasing slope was found to occur most commonly after 6 cm, not 4 cm. The slowest 5% of nulliparous women had cervical dilation of 0.4 cm per hour (with the median at 1.9 cm per hour), compared with 1.2 cm per hour (with a median of 3.0 cm per hour) as in the Friedman data.
Dr. Zhang’s study showed us that labor may take more than 6 hours to progress from 4 to 5 cm dilation, and more than 3 hours to progress from 5 to 6 cm dilation – a rate of progress that is significantly slower than what Dr. Friedman had described. The new data showed us, moreover, that from 4 cm-6 cm dilation, nulliparous and multiparous women progressed similarly slowly. Beyond 6 cm, multiparous women dilated more rapidly, with a steeper acceleration phase than previously described.
A consensus statement published in 2014 by the American College of Obstetricians and Gynecologists (ACOG) and the Society for Maternal-Fetal Medicine (SMFM) on “Safe Prevention of the Primary Cesarean Delivery” encourages use of the Consortium data to revisit the definition of labor dystocia. While the data “do not directly address an optimal duration for the diagnosis of active-phase protraction or labor arrest, [they] do suggest that neither should be diagnosed before 6 cm dilation” (Obstet Gynecol. 2014 Mar;123[3]:693-711).
The ACOG-SMFM statement makes a series of recommendations for managing the first and second stages of labor, based not only on the Consortium data but on a broader literature review. It recommends that if mother and fetus appear well, cesarean delivery for active-phase arrest in the first stage of labor be reserved for women of at least 6 cm of dilation with ruptured membranes who fail to progress despite 4 hours of adequate uterine activity, or at least 6 hours of oxytocin administration with inadequate uterine activity and no cervical change.
Regarding the latent phase of labor, the statement says that most women with a prolonged latent phase ultimately will enter the active phase with expectant management. It advises that a prolonged latent phase (for example, greater than 20 hours in nulliparous women and greater than 14 hours in multiparous women) should not be an isolated indication for cesarean delivery.
The consensus statement also recognizes recent data showing that women who undergo labor induction have an even slower “normal” course of labor, particularly a longer latent phase, than women who labor spontaneously. A retrospective cohort study of more than 5,000 women, for instance, found that before 6 cm, women whose labor is induced can spend up to 10 hours to achieve each 1 cm of dilation (Obstet Gynecol. 2012 Jun;119[6]:1113-8).
As long as maternal and fetal status are reassuring, the statement says, cesarean deliveries for failed induction of labor in the latent phase can be avoided by allowing longer durations of the latent phase (up to 24 hours) and by requiring that oxytocin be administered for 12-18 hours after membrane rupture before deeming induction a failure.
Each of these described recommendations were graded in the ACOG-SMFM consensus document as “strong” recommendations with “moderate quality evidence.”
Examining our standards
Moving forward, we must further develop and define our thresholds for identifying who will most benefit from a cesarean delivery. We have many specific aspects of labor management to address as well, such as the optimal timing of artificial membrane rupture and the safety and efficacy of different oxytocin protocols. We may also want to revisit recommendations for serial cervical assessment, possibly adjusting the intervals given our understanding of the new labor curve.
Under the new labor paradigm, moreover, we must think not only about the clinical decisions we make at the bedside, but about the decisions we make early in the labor management process.
The timing of admission is one such decision. A statement published in 2012 on “Preventing the First Cesarean Delivery” by ACOG, SMFM, and the Eunice Kennedy Shriver National Institute of Child Health and Human Development advises us to avoid admittance of women during the early latent phase of labor (Obstet Gynecol. 2012 Nov;120[5]:1181-93).
It may even be advisable that we consider admittance at higher cervical dilation. A study published this year shows that women admitted at less than 6 cm of dilation had an increased risk of cesarean delivery, compared with women admitted at higher cervical dilation (Am J Perinatol. 2016 Jan;33[2]:188-94). We have more to learn, but certainly, given what we know now about labor progress and the start of active labor, the timing of admission is an important factor to consider.
The second stage of labor, defined as the interval from complete cervical dilation through delivery of the fetus, presents many questions as well. There is a paucity of quality published data concerning what is normal, how long the stage should last, and how we should manage it. Historically, we have been taught to allow 2 hours of pushing for nulliparous women and 1 hour for multiparous women, when epidural anesthesia has not been administered, and to add an additional hour when epidural is used.
The 2014 ACOG-SMFM consensus statement recommends extending each of these limits by an hour, if maternal and fetal conditions permit, so that we allow at least 3 hours of pushing for nulliparous women and at least 2 hours for multiparous women before diagnosing arrest of labor in the second stage. Longer durations may be appropriate with the use of epidural anesthesia and on an individualized basis.
At this time, it is unclear whether there is any absolute maximum length of time beyond which all women in the second stage of labor should undergo cesarean delivery. We also still do not know the optimal technique for managing maternal pushing during the second stage. Should women with an epidural push right away or should they allow for a period of spontaneous descent? Many of the high-quality studies reported thus far that compare delayed and immediate pushing have limited applicability to current practice because they involved now-obsolete midpelvic forceps deliveries. A large multicenter randomized trial currently underway should provide us with some answers.
Dr. Cahill is an associate professor and chief of the division of maternal-fetal medicine in the department of obstetrics and gynecology at Washington University School of Medicine in St. Louis. She reported having no relevant financial disclosures.
Rethinking the management of labor
Over the last 50 years, we have witnessed some incredible advancements that have vastly improved maternal and fetal outcomes, even in the face of the most complex obstetrical dilemmas. As our practice and the research continues to evolve, it is increasingly important that we carefully review our practice standards to ensure that every woman and her baby receives the most up-to-date medical care.
This month’s Master Class highlights a critical area of obstetrics where the convergence of technology, clinical observation, and research stimulated a change in practice guidelines: the use of the labor curve to monitor normal versus abnormal labor. Until quite recently, ob.gyns. had based labor criteria on the “Friedman Curve,” first established in the mid-1950s, and supported by other smaller and less comprehensive studies. This work was adopted by the American College of Obstetricians and Gynecologists.
For more than half a century, we used these parameters to determine if a woman had entered active-phase arrest, and to make the very important decision of whether to perform a cesarean section. However, work in the early 2000s strongly suggested that the old criteria no longer applied to the full course of labor in contemporary patients (Am J Obstet Gynecol. 2002 Oct;187[4]:824-8). A 2010 comprehensive study showed that we needed to consider a new approach to labor management (Am J Obstet Gynecol. 2010 Oct;203[4]:326.e1-326.e10).
It may seem incredible that it took such a long time to update our thinking about what constitutes normal versus abnormal labor progression. However, we must keep in mind that many studies supported the original labor curve, and advanced tools to assess fetal health during labor were just being developed. The first commercially available fetal heart rate monitor would not be produced until 1968, and debates about the utility of these devices would continue into the early 1990s.
Additionally, our patient population has changed. As we have discussed in previous columns, the incidence and severity of other chronic conditions, such as diabetes and obesity, has increased significantly and deeply impacted labor progression.
Just as technology has advanced and our patients’ needs have changed, so, too, must our practice standards. We have invited Dr. Alison G. Cahill, associate professor and chief of the division of maternal-fetal medicine in the department of obstetrics and gynecology at Washington University, St. Louis, to discuss the importance and implications of the new labor curve.
Dr. Reece, who specializes in maternal-fetal medicine, is vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. Dr. Reece said he had no relevant financial disclosures. He is the medical editor of this column. Contact him at obnews@frontlinemedcom.com.
Over the last 50 years, we have witnessed some incredible advancements that have vastly improved maternal and fetal outcomes, even in the face of the most complex obstetrical dilemmas. As our practice and the research continues to evolve, it is increasingly important that we carefully review our practice standards to ensure that every woman and her baby receives the most up-to-date medical care.
This month’s Master Class highlights a critical area of obstetrics where the convergence of technology, clinical observation, and research stimulated a change in practice guidelines: the use of the labor curve to monitor normal versus abnormal labor. Until quite recently, ob.gyns. had based labor criteria on the “Friedman Curve,” first established in the mid-1950s, and supported by other smaller and less comprehensive studies. This work was adopted by the American College of Obstetricians and Gynecologists.
For more than half a century, we used these parameters to determine if a woman had entered active-phase arrest, and to make the very important decision of whether to perform a cesarean section. However, work in the early 2000s strongly suggested that the old criteria no longer applied to the full course of labor in contemporary patients (Am J Obstet Gynecol. 2002 Oct;187[4]:824-8). A 2010 comprehensive study showed that we needed to consider a new approach to labor management (Am J Obstet Gynecol. 2010 Oct;203[4]:326.e1-326.e10).
It may seem incredible that it took such a long time to update our thinking about what constitutes normal versus abnormal labor progression. However, we must keep in mind that many studies supported the original labor curve, and advanced tools to assess fetal health during labor were just being developed. The first commercially available fetal heart rate monitor would not be produced until 1968, and debates about the utility of these devices would continue into the early 1990s.
Additionally, our patient population has changed. As we have discussed in previous columns, the incidence and severity of other chronic conditions, such as diabetes and obesity, has increased significantly and deeply impacted labor progression.
Just as technology has advanced and our patients’ needs have changed, so, too, must our practice standards. We have invited Dr. Alison G. Cahill, associate professor and chief of the division of maternal-fetal medicine in the department of obstetrics and gynecology at Washington University, St. Louis, to discuss the importance and implications of the new labor curve.
Dr. Reece, who specializes in maternal-fetal medicine, is vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. Dr. Reece said he had no relevant financial disclosures. He is the medical editor of this column. Contact him at obnews@frontlinemedcom.com.
Over the last 50 years, we have witnessed some incredible advancements that have vastly improved maternal and fetal outcomes, even in the face of the most complex obstetrical dilemmas. As our practice and the research continues to evolve, it is increasingly important that we carefully review our practice standards to ensure that every woman and her baby receives the most up-to-date medical care.
This month’s Master Class highlights a critical area of obstetrics where the convergence of technology, clinical observation, and research stimulated a change in practice guidelines: the use of the labor curve to monitor normal versus abnormal labor. Until quite recently, ob.gyns. had based labor criteria on the “Friedman Curve,” first established in the mid-1950s, and supported by other smaller and less comprehensive studies. This work was adopted by the American College of Obstetricians and Gynecologists.
For more than half a century, we used these parameters to determine if a woman had entered active-phase arrest, and to make the very important decision of whether to perform a cesarean section. However, work in the early 2000s strongly suggested that the old criteria no longer applied to the full course of labor in contemporary patients (Am J Obstet Gynecol. 2002 Oct;187[4]:824-8). A 2010 comprehensive study showed that we needed to consider a new approach to labor management (Am J Obstet Gynecol. 2010 Oct;203[4]:326.e1-326.e10).
It may seem incredible that it took such a long time to update our thinking about what constitutes normal versus abnormal labor progression. However, we must keep in mind that many studies supported the original labor curve, and advanced tools to assess fetal health during labor were just being developed. The first commercially available fetal heart rate monitor would not be produced until 1968, and debates about the utility of these devices would continue into the early 1990s.
Additionally, our patient population has changed. As we have discussed in previous columns, the incidence and severity of other chronic conditions, such as diabetes and obesity, has increased significantly and deeply impacted labor progression.
Just as technology has advanced and our patients’ needs have changed, so, too, must our practice standards. We have invited Dr. Alison G. Cahill, associate professor and chief of the division of maternal-fetal medicine in the department of obstetrics and gynecology at Washington University, St. Louis, to discuss the importance and implications of the new labor curve.
Dr. Reece, who specializes in maternal-fetal medicine, is vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. Dr. Reece said he had no relevant financial disclosures. He is the medical editor of this column. Contact him at obnews@frontlinemedcom.com.
Understanding stillbirth
When a couple learns that “they are pregnant,” it is often one of the most joyous moments in their lives. However, despite the modern prenatal care available to women in the United States, pregnancy loss remains a real concern. Miscarriage is estimated to occur in 15%-20% of pregnancies; recurrent pregnancy loss in about 1%-2% of pregnancies; and stillbirth in as many as 1% of pregnancies. The causes of pregnancy loss can range from those we can diagnose, such as genetic factors, anatomic complications, and thrombophilia, to those that elude us completely.
In December 2015, investigators from Karolinska Institutet in Stockholm published a study indicating that women who gained weight between their first and second pregnancies, but who were a healthy weight prior to their first pregnancy, had an increased risk of experiencing a stillbirth (30%-50%), or having an infant who died within the first year (27%-60%) (Lancet 2015. doi: 10.1016/S0140-6736(15)00990-3). We have devoted a number of Master Class columns to the link between obesity and pregnancy complications, and this study further reinforces the influence of a healthy weight on pregnancy outcomes.
In addition to lifestyle modifications, evidence has suggested that low-molecular-weight heparin, aspirin, or vitamin supplements, in combination with appropriate surveillance and management, may reduce risk of pregnancy loss. However, more work is needed to fully understand why fetal death occurs if we are to better equip ourselves, and our patients, with all the information necessary to prevent loss from happening.
For this reason, we have invited Dr. Uma M. Reddy of the Pregnancy and Perinatology Branch of the Eunice Kennedy Shriver National Institute of Child Health and Human Development at the National Institutes of Health, to address one of the most devastating types of pregnancy losses: stillbirth. As a program scientist for large research studies, such as the Stillbirth Collaborative Research Network, Dr. Reddy’s unique perspective will add greatly to our understanding of pregnancy loss.
Dr. Reece, who specializes in maternal-fetal medicine, is vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. Dr. Reece reported having no relevant financial disclosures. He is the medical editor of this column.
When a couple learns that “they are pregnant,” it is often one of the most joyous moments in their lives. However, despite the modern prenatal care available to women in the United States, pregnancy loss remains a real concern. Miscarriage is estimated to occur in 15%-20% of pregnancies; recurrent pregnancy loss in about 1%-2% of pregnancies; and stillbirth in as many as 1% of pregnancies. The causes of pregnancy loss can range from those we can diagnose, such as genetic factors, anatomic complications, and thrombophilia, to those that elude us completely.
In December 2015, investigators from Karolinska Institutet in Stockholm published a study indicating that women who gained weight between their first and second pregnancies, but who were a healthy weight prior to their first pregnancy, had an increased risk of experiencing a stillbirth (30%-50%), or having an infant who died within the first year (27%-60%) (Lancet 2015. doi: 10.1016/S0140-6736(15)00990-3). We have devoted a number of Master Class columns to the link between obesity and pregnancy complications, and this study further reinforces the influence of a healthy weight on pregnancy outcomes.
In addition to lifestyle modifications, evidence has suggested that low-molecular-weight heparin, aspirin, or vitamin supplements, in combination with appropriate surveillance and management, may reduce risk of pregnancy loss. However, more work is needed to fully understand why fetal death occurs if we are to better equip ourselves, and our patients, with all the information necessary to prevent loss from happening.
For this reason, we have invited Dr. Uma M. Reddy of the Pregnancy and Perinatology Branch of the Eunice Kennedy Shriver National Institute of Child Health and Human Development at the National Institutes of Health, to address one of the most devastating types of pregnancy losses: stillbirth. As a program scientist for large research studies, such as the Stillbirth Collaborative Research Network, Dr. Reddy’s unique perspective will add greatly to our understanding of pregnancy loss.
Dr. Reece, who specializes in maternal-fetal medicine, is vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. Dr. Reece reported having no relevant financial disclosures. He is the medical editor of this column.
When a couple learns that “they are pregnant,” it is often one of the most joyous moments in their lives. However, despite the modern prenatal care available to women in the United States, pregnancy loss remains a real concern. Miscarriage is estimated to occur in 15%-20% of pregnancies; recurrent pregnancy loss in about 1%-2% of pregnancies; and stillbirth in as many as 1% of pregnancies. The causes of pregnancy loss can range from those we can diagnose, such as genetic factors, anatomic complications, and thrombophilia, to those that elude us completely.
In December 2015, investigators from Karolinska Institutet in Stockholm published a study indicating that women who gained weight between their first and second pregnancies, but who were a healthy weight prior to their first pregnancy, had an increased risk of experiencing a stillbirth (30%-50%), or having an infant who died within the first year (27%-60%) (Lancet 2015. doi: 10.1016/S0140-6736(15)00990-3). We have devoted a number of Master Class columns to the link between obesity and pregnancy complications, and this study further reinforces the influence of a healthy weight on pregnancy outcomes.
In addition to lifestyle modifications, evidence has suggested that low-molecular-weight heparin, aspirin, or vitamin supplements, in combination with appropriate surveillance and management, may reduce risk of pregnancy loss. However, more work is needed to fully understand why fetal death occurs if we are to better equip ourselves, and our patients, with all the information necessary to prevent loss from happening.
For this reason, we have invited Dr. Uma M. Reddy of the Pregnancy and Perinatology Branch of the Eunice Kennedy Shriver National Institute of Child Health and Human Development at the National Institutes of Health, to address one of the most devastating types of pregnancy losses: stillbirth. As a program scientist for large research studies, such as the Stillbirth Collaborative Research Network, Dr. Reddy’s unique perspective will add greatly to our understanding of pregnancy loss.
Dr. Reece, who specializes in maternal-fetal medicine, is vice president for medical affairs at the University of Maryland, Baltimore, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. Dr. Reece reported having no relevant financial disclosures. He is the medical editor of this column.
Research adds insight on stillbirth risk factors
Stillbirth is a major public health problem, occurring in approximately 1 of every 160 pregnancies in the United States. The rate has remained stagnant since 2006. Prior to that time, from 1990 to 2006, the rate declined somewhat, but only half as much as the decline in infant mortality during this time period. Racial disparities also have persisted, with non-Hispanic black women having more than a twofold increase in risk (Natl Vital Stat Rep. 2012;60:1-22).
Research conducted by the Stillbirth Collaborative Research Network (SCRN) and others has provided us with insight on risk factors and on probable and possible causes of death among stillbirths, which are defined as fetal deaths at 20 or more weeks’ gestation. We know from SCRN data, for instance, that black women are more likely to have stillbirths associated with obstetric complications and infections than white and Hispanic women. However, we still cannot explain a substantial proportion of stillbirths, despite a complete evaluation, or predict who will have a stillbirth.
What we can do as obstetricians is be aware that stillbirth is one of the most common adverse pregnancy outcomes in the United States and counsel women regarding risk factors that are modifiable. Moreover, when stillbirth happens, a complete postmortem evaluation that includes autopsy, placental pathology, karyotype or microarray analysis, and fetal-maternal hemorrhage testing is recommended (Obstet Gynecol. 2009;113[3]:748-61). Recent data show that each of these four components is valuable and should be considered the basic work-up for stillbirth.
Risks and causes
Pregnancy history was the strongest baseline risk factor for stillbirth in an analysis of 614 stillbirths and 1,816 live births in the SCRN’s population-based, case-control study conducted between 2006 and 2008. The SCRN was initiated by the Eunice Kennedy Shriver National Institute of Child Health and Human Development in 2003. This critical population-based study was conducted at 59 U.S. tertiary care and community hospitals in five catchment areas and has been analyzed in more than 15 published reports.
Women with a previous stillbirth have been known to be at 5- to 10-fold increased risk of a recurrence of stillbirth, and the SCRN findings confirmed this. The study added to our knowledge, however, with the finding that even a prior pregnancy loss at less than 20 weeks’ gestation increased the risk for stillbirth.
Other risk factors identified in the study, in addition to race, included having a multifetal pregnancy (adjusted odds ratio of 4.59), diabetes (AOR of 2.50), maternal age of 40 years or older (AOR of 2.41), maternal AB blood type (AOR of 1.96, compared with type O), a history of drug addiction (AOR of 2.08), smoking during the 3 months prior to pregnancy (AOR of 1.55-1.57, depending on amount), and being unmarried and not cohabitating (AOR of 1.69). Regarding racial disparity, the study showed that elevated risk of stillbirth for non-Hispanic blacks occurred predominantly prior to 24 weeks of gestation.
As in prior research, overweight and obesity also conferred elevated risks in the SCRN study (AORs of 1.43 and 1.72, respectively), and these risks were not explained by either diabetes or hypertension (JAMA. 2011;306:2469-79).
The use of assisted reproductive technology was not included in the study’s multivariate model, but previous research has shown a fourfold increased risk of stillbirth for singleton IVF/ICSI pregnancies. The reason is unclear, but the risk appears to be more related to IVF/ICSI rather than the underlying infertility (Hum Reprod. 2010 May;25[5]:1312-6).
A previous preterm or small-for-gestational-age birth has also been shown in prior research to be a significant risk factor for stillbirth. Less clear is the role of previous cesarean delivery in stillbirth risk. An association has been demonstrated in several studies, however, including one involving about 180,000 singleton pregnancies of 23 or more weeks’ gestation. Women in this cohort who had a previous cesarean delivery had a 1.3-fold increased risk of antepartum stillbirth, after controlling for important factors such as race, body mass index (BMI), and maternal disease (Obstet Gynecol. 2010 Nov;116[5]:1119-26).
In another analysis of the SCRN study looking specifically at causes of stillbirth, a “probable” cause of death was found in 61% of cases and a “possible or probable” cause of death in more than 76% of cases. The most common causes were obstetric complications (29.3%), placental abnormalities (23.6%), fetal genetic/structural abnormalities (13.7%), infection (12.9%), umbilical cord abnormalities (10.4%), hypertensive disorders (9.2%), and other maternal medical conditions (7.8%).
A higher proportion of stillbirths in non-Hispanic black women, compared with non-Hispanic white women and Hispanic women was associated with obstetric complications (43.5%) and infections (25.2%). This finding combined with the finding that stillbirth in black women often occurs at less than 24 weeks’ gestation suggests that measures aimed at reducing the rate of spontaneous preterm birth in black women could potentially reduce the rate of stillbirth as well (JAMA. 2011 Dec 14;306[22]:2459-68).
Work-up and prevention
Prevention of stillbirth requires that we identify the women at highest risk, and thus far this ability still eludes us. Apart from occurrence of previous stillbirth or pregnancy loss, other risk factors have had limited predictive value in the SCRN analyses and other research.
Biomarkers such as a low PAPP-A during the first trimester and a high AFP in the second trimester – as well as Doppler imaging of the uterine artery – have also been associated with stillbirth, but again, the positive predictive value has been shown to be low (Clin Obstet Gynecol. 2010 Sep;53[3]:597-606). More research is needed to determine if some combination of biochemical markers, imaging, and other risk factors can predict which women are at highest risk.
In the meantime, attention can be paid – in the preconception period if possible – to modifiable risk factors such as maternal obesity, diabetes, and smoking. About 10% of stillbirths are associated with maternal conditions such as hypertension and diabetes, and late stillbirths in particular (28 weeks or later) are associated with maternal medical conditions that are potentially preventable.
Normalization of prepregnancy weight should be a goal, since the overall risk of stillbirth appears to increase independently with increasing BMI. Glycemic control should also be achieved: A recent meta-analysis of preconception and prenatal care of diabetic women estimated “conservatively” that 10% of diabetes-associated stillbirths could be prevented with early detection and glycemic control (BMC Public Health. 2011;11 Suppl 3:S2). Research has also shown that women who quit smoking between their first and second pregnancy reduce their stillbirth risk to that of nonsmokers in the second pregnancy (BJOG. 2007 Jun;114[6]:699-704).
When stillbirth happens, a thorough work-up is recommended in order to counsel for future pregnancies and decrease the risk of recurrence. Evaluations for causes of stillbirth are too often incomplete in the United States for various reasons, including emotional, cultural, and resource factors. Even if a cause is not found, many families appreciate knowing that every effort has been made to determine a cause of death.
Four components of evaluation – autopsy, placental examination, karyotype or microarray analysis, and fetal-maternal hemorrhage testing – have proven to be high-yield tests when performed in all cases of stillbirth.
In the SCRN study, of 512 stillbirths undergoing a complete evaluation, 66.4% had a positive result – defined as abnormalities contributing to a probable or possible cause – for at least one of the first three tests (JAMA. 2011 Dec 14;306[22]:2459-68).
A Dutch study of 1,025 stillbirths similarly demonstrated that all four tests are justified. A test was defined as valuable in this study if it established or excluded a cause of stillbirth. Placental examination was determined to be the most valuable test, helping to determine a cause of death in 95.7% of cases. Autopsy was valuable 72.6% of the time, and cytogenetic analysis was valuable in 29% of cases.
Kleihauer-Betke testing for fetal-maternal hemorrhage was positive in 11.9% of women. However, fetal maternal hemorrhage was considered the cause of death in only 1.3%.of cases because, beyond a positive Kleihauer-Betke test, evidence of fetal anemia confirmed by placental examination and/or autopsy was required for hemorrhage to be considered the cause of death (Am. J. Obstet. Gynecol. 2012;206:53.e1-12). Because Kleihauer-Betke testing is ideally performed before induction, authors of both the SCRN study and the Dutch study believe it is a valuable test to be offered in all cases.
In both studies, the yield of other stillbirth diagnostic tests (for example, maternal serology, hormone assessment, and toxicology screen) was low, indicating that these tests are considered sequential and can be performed only when the clinical history or findings of the four core tests raise suspicion of particular potential causes. Antinuclear antibody testing and TORCH (toxoplasmosis, rubella, cytomegalovirus, herpes simplex) titers have an extremely low yield and are generally not useful.
For detecting genetic abnormalities after stillbirth, it appears that microarray analysis is superior to karyotype analysis. In a SCRN analysis of samples from 532 stillbirths, microarray yielded results more often and identified more genetic abnormalities. Unlike karyotype, it does not require live cells, which makes it preferable for stillbirth evaluation (N Engl J Med. 2012 Dec 6;367[23]:2185-93).
Current research
One of the more significant studies underway on prevention is looking at labor induction as an intervention for reducing stillbirths and improving other perinatal outcomes. The ARRIVE trial (“A Randomized Trial of Induction Versus Expectant Management”), currently in the recruitment stage, will examine outcomes after induction at 39 weeks’ gestation, compared with expectant management in 6,000 patients (clinicaltrials.gov/ct2/show/NCT01990612).
Common wisdom informed by retrospective cohort studies has long told us that inducing labor prior to 41 weeks’ gestation is associated with a higher risk of cesarean delivery in nulliparous women. However, recent observational data have suggested that women whose labor is induced actually have fewer cesarean deliveries and better perinatal outcomes, including a lower risk of stillbirth (AJOG 2012;207:502.e1-8).
In addition, a meta-analysis published in 2014, as the ARRIVE trial was taking shape, reported a 12% reduction in cesarean delivery, and a reduced risk of stillbirth, among women whose labor was induced. The initial cervical score did not impact the main findings (CMAJ. 2014 Jun 10;186[9]:665-73). If these findings are confirmed in the ARRIVE trial, we could see a new opportunity for stillbirth prevention.
Another ongoing study of 10,000 singleton pregnancies – the Nulliparous Pregnancy Outcomes: Monitoring Mothers-to-Be (nuMoM2b) study – may also lead to prevention strategies in women for whom the current pregnancy will lead to their first delivery. Among the questions being examined in this eight-site study are whether sleep-disordered breathing, or apnea, and a supine sleep position are risk factors for adverse pregnancy outcomes including stillbirth.
Supine sleeping in the last month of pregnancy was strongly associated with stillbirth in a recent analysis from the Sydney Stillbirth Study (Obstet Gynecol. 2015 Feb;125[2]:347-55), and an early analysis of a nuMoM2b subset has shown associations between sleep-disordered breathing in midpregnancy and the development of hypertensive disorders of pregnancy, and between sleep-disordered breathing in early- and mid-pregnancy and gestational diabetes (Am J Obstet Gynecol. 2015;212:S424-425).
The possible role of low-dose aspirin in preventing stillbirth also needs more exploration. A recent randomized trial of women attempting to become pregnant after having had one or two prior pregnancy losses found no difference overall in live birth rates between those who took low-dose aspirin and those assigned to placebo. However, there was one subgroup – women with a single loss at less than 20 weeks’ gestation during the previous year – in which live birth rates were higher in the aspirin group (Lancet. 2014 Jul 5;384[9937]:29-36). More research is necessary to determine if low-dose aspirin administration in women with a previous stillbirth improves pregnancy outcome.
Dr. Reddy is a member at the Pregnancy and Perinatology Branch of the Eunice Kennedy Shriver National Institute of Child Health and Human Development. She is a board-certified ob.gyn. and maternal-fetal medicine specialist. She is the program scientist for the Maternal-Fetal Medicine Units Network and for the Stillbirth Collaborative Research Network. The comments and views of the author do not necessarily represent the views of the NICHD.
Stillbirth is a major public health problem, occurring in approximately 1 of every 160 pregnancies in the United States. The rate has remained stagnant since 2006. Prior to that time, from 1990 to 2006, the rate declined somewhat, but only half as much as the decline in infant mortality during this time period. Racial disparities also have persisted, with non-Hispanic black women having more than a twofold increase in risk (Natl Vital Stat Rep. 2012;60:1-22).
Research conducted by the Stillbirth Collaborative Research Network (SCRN) and others has provided us with insight on risk factors and on probable and possible causes of death among stillbirths, which are defined as fetal deaths at 20 or more weeks’ gestation. We know from SCRN data, for instance, that black women are more likely to have stillbirths associated with obstetric complications and infections than white and Hispanic women. However, we still cannot explain a substantial proportion of stillbirths, despite a complete evaluation, or predict who will have a stillbirth.
What we can do as obstetricians is be aware that stillbirth is one of the most common adverse pregnancy outcomes in the United States and counsel women regarding risk factors that are modifiable. Moreover, when stillbirth happens, a complete postmortem evaluation that includes autopsy, placental pathology, karyotype or microarray analysis, and fetal-maternal hemorrhage testing is recommended (Obstet Gynecol. 2009;113[3]:748-61). Recent data show that each of these four components is valuable and should be considered the basic work-up for stillbirth.
Risks and causes
Pregnancy history was the strongest baseline risk factor for stillbirth in an analysis of 614 stillbirths and 1,816 live births in the SCRN’s population-based, case-control study conducted between 2006 and 2008. The SCRN was initiated by the Eunice Kennedy Shriver National Institute of Child Health and Human Development in 2003. This critical population-based study was conducted at 59 U.S. tertiary care and community hospitals in five catchment areas and has been analyzed in more than 15 published reports.
Women with a previous stillbirth have been known to be at 5- to 10-fold increased risk of a recurrence of stillbirth, and the SCRN findings confirmed this. The study added to our knowledge, however, with the finding that even a prior pregnancy loss at less than 20 weeks’ gestation increased the risk for stillbirth.
Other risk factors identified in the study, in addition to race, included having a multifetal pregnancy (adjusted odds ratio of 4.59), diabetes (AOR of 2.50), maternal age of 40 years or older (AOR of 2.41), maternal AB blood type (AOR of 1.96, compared with type O), a history of drug addiction (AOR of 2.08), smoking during the 3 months prior to pregnancy (AOR of 1.55-1.57, depending on amount), and being unmarried and not cohabitating (AOR of 1.69). Regarding racial disparity, the study showed that elevated risk of stillbirth for non-Hispanic blacks occurred predominantly prior to 24 weeks of gestation.
As in prior research, overweight and obesity also conferred elevated risks in the SCRN study (AORs of 1.43 and 1.72, respectively), and these risks were not explained by either diabetes or hypertension (JAMA. 2011;306:2469-79).
The use of assisted reproductive technology was not included in the study’s multivariate model, but previous research has shown a fourfold increased risk of stillbirth for singleton IVF/ICSI pregnancies. The reason is unclear, but the risk appears to be more related to IVF/ICSI rather than the underlying infertility (Hum Reprod. 2010 May;25[5]:1312-6).
A previous preterm or small-for-gestational-age birth has also been shown in prior research to be a significant risk factor for stillbirth. Less clear is the role of previous cesarean delivery in stillbirth risk. An association has been demonstrated in several studies, however, including one involving about 180,000 singleton pregnancies of 23 or more weeks’ gestation. Women in this cohort who had a previous cesarean delivery had a 1.3-fold increased risk of antepartum stillbirth, after controlling for important factors such as race, body mass index (BMI), and maternal disease (Obstet Gynecol. 2010 Nov;116[5]:1119-26).
In another analysis of the SCRN study looking specifically at causes of stillbirth, a “probable” cause of death was found in 61% of cases and a “possible or probable” cause of death in more than 76% of cases. The most common causes were obstetric complications (29.3%), placental abnormalities (23.6%), fetal genetic/structural abnormalities (13.7%), infection (12.9%), umbilical cord abnormalities (10.4%), hypertensive disorders (9.2%), and other maternal medical conditions (7.8%).
A higher proportion of stillbirths in non-Hispanic black women, compared with non-Hispanic white women and Hispanic women was associated with obstetric complications (43.5%) and infections (25.2%). This finding combined with the finding that stillbirth in black women often occurs at less than 24 weeks’ gestation suggests that measures aimed at reducing the rate of spontaneous preterm birth in black women could potentially reduce the rate of stillbirth as well (JAMA. 2011 Dec 14;306[22]:2459-68).
Work-up and prevention
Prevention of stillbirth requires that we identify the women at highest risk, and thus far this ability still eludes us. Apart from occurrence of previous stillbirth or pregnancy loss, other risk factors have had limited predictive value in the SCRN analyses and other research.
Biomarkers such as a low PAPP-A during the first trimester and a high AFP in the second trimester – as well as Doppler imaging of the uterine artery – have also been associated with stillbirth, but again, the positive predictive value has been shown to be low (Clin Obstet Gynecol. 2010 Sep;53[3]:597-606). More research is needed to determine if some combination of biochemical markers, imaging, and other risk factors can predict which women are at highest risk.
In the meantime, attention can be paid – in the preconception period if possible – to modifiable risk factors such as maternal obesity, diabetes, and smoking. About 10% of stillbirths are associated with maternal conditions such as hypertension and diabetes, and late stillbirths in particular (28 weeks or later) are associated with maternal medical conditions that are potentially preventable.
Normalization of prepregnancy weight should be a goal, since the overall risk of stillbirth appears to increase independently with increasing BMI. Glycemic control should also be achieved: A recent meta-analysis of preconception and prenatal care of diabetic women estimated “conservatively” that 10% of diabetes-associated stillbirths could be prevented with early detection and glycemic control (BMC Public Health. 2011;11 Suppl 3:S2). Research has also shown that women who quit smoking between their first and second pregnancy reduce their stillbirth risk to that of nonsmokers in the second pregnancy (BJOG. 2007 Jun;114[6]:699-704).
When stillbirth happens, a thorough work-up is recommended in order to counsel for future pregnancies and decrease the risk of recurrence. Evaluations for causes of stillbirth are too often incomplete in the United States for various reasons, including emotional, cultural, and resource factors. Even if a cause is not found, many families appreciate knowing that every effort has been made to determine a cause of death.
Four components of evaluation – autopsy, placental examination, karyotype or microarray analysis, and fetal-maternal hemorrhage testing – have proven to be high-yield tests when performed in all cases of stillbirth.
In the SCRN study, of 512 stillbirths undergoing a complete evaluation, 66.4% had a positive result – defined as abnormalities contributing to a probable or possible cause – for at least one of the first three tests (JAMA. 2011 Dec 14;306[22]:2459-68).
A Dutch study of 1,025 stillbirths similarly demonstrated that all four tests are justified. A test was defined as valuable in this study if it established or excluded a cause of stillbirth. Placental examination was determined to be the most valuable test, helping to determine a cause of death in 95.7% of cases. Autopsy was valuable 72.6% of the time, and cytogenetic analysis was valuable in 29% of cases.
Kleihauer-Betke testing for fetal-maternal hemorrhage was positive in 11.9% of women. However, fetal maternal hemorrhage was considered the cause of death in only 1.3%.of cases because, beyond a positive Kleihauer-Betke test, evidence of fetal anemia confirmed by placental examination and/or autopsy was required for hemorrhage to be considered the cause of death (Am. J. Obstet. Gynecol. 2012;206:53.e1-12). Because Kleihauer-Betke testing is ideally performed before induction, authors of both the SCRN study and the Dutch study believe it is a valuable test to be offered in all cases.
In both studies, the yield of other stillbirth diagnostic tests (for example, maternal serology, hormone assessment, and toxicology screen) was low, indicating that these tests are considered sequential and can be performed only when the clinical history or findings of the four core tests raise suspicion of particular potential causes. Antinuclear antibody testing and TORCH (toxoplasmosis, rubella, cytomegalovirus, herpes simplex) titers have an extremely low yield and are generally not useful.
For detecting genetic abnormalities after stillbirth, it appears that microarray analysis is superior to karyotype analysis. In a SCRN analysis of samples from 532 stillbirths, microarray yielded results more often and identified more genetic abnormalities. Unlike karyotype, it does not require live cells, which makes it preferable for stillbirth evaluation (N Engl J Med. 2012 Dec 6;367[23]:2185-93).
Current research
One of the more significant studies underway on prevention is looking at labor induction as an intervention for reducing stillbirths and improving other perinatal outcomes. The ARRIVE trial (“A Randomized Trial of Induction Versus Expectant Management”), currently in the recruitment stage, will examine outcomes after induction at 39 weeks’ gestation, compared with expectant management in 6,000 patients (clinicaltrials.gov/ct2/show/NCT01990612).
Common wisdom informed by retrospective cohort studies has long told us that inducing labor prior to 41 weeks’ gestation is associated with a higher risk of cesarean delivery in nulliparous women. However, recent observational data have suggested that women whose labor is induced actually have fewer cesarean deliveries and better perinatal outcomes, including a lower risk of stillbirth (AJOG 2012;207:502.e1-8).
In addition, a meta-analysis published in 2014, as the ARRIVE trial was taking shape, reported a 12% reduction in cesarean delivery, and a reduced risk of stillbirth, among women whose labor was induced. The initial cervical score did not impact the main findings (CMAJ. 2014 Jun 10;186[9]:665-73). If these findings are confirmed in the ARRIVE trial, we could see a new opportunity for stillbirth prevention.
Another ongoing study of 10,000 singleton pregnancies – the Nulliparous Pregnancy Outcomes: Monitoring Mothers-to-Be (nuMoM2b) study – may also lead to prevention strategies in women for whom the current pregnancy will lead to their first delivery. Among the questions being examined in this eight-site study are whether sleep-disordered breathing, or apnea, and a supine sleep position are risk factors for adverse pregnancy outcomes including stillbirth.
Supine sleeping in the last month of pregnancy was strongly associated with stillbirth in a recent analysis from the Sydney Stillbirth Study (Obstet Gynecol. 2015 Feb;125[2]:347-55), and an early analysis of a nuMoM2b subset has shown associations between sleep-disordered breathing in midpregnancy and the development of hypertensive disorders of pregnancy, and between sleep-disordered breathing in early- and mid-pregnancy and gestational diabetes (Am J Obstet Gynecol. 2015;212:S424-425).
The possible role of low-dose aspirin in preventing stillbirth also needs more exploration. A recent randomized trial of women attempting to become pregnant after having had one or two prior pregnancy losses found no difference overall in live birth rates between those who took low-dose aspirin and those assigned to placebo. However, there was one subgroup – women with a single loss at less than 20 weeks’ gestation during the previous year – in which live birth rates were higher in the aspirin group (Lancet. 2014 Jul 5;384[9937]:29-36). More research is necessary to determine if low-dose aspirin administration in women with a previous stillbirth improves pregnancy outcome.
Dr. Reddy is a member at the Pregnancy and Perinatology Branch of the Eunice Kennedy Shriver National Institute of Child Health and Human Development. She is a board-certified ob.gyn. and maternal-fetal medicine specialist. She is the program scientist for the Maternal-Fetal Medicine Units Network and for the Stillbirth Collaborative Research Network. The comments and views of the author do not necessarily represent the views of the NICHD.
Stillbirth is a major public health problem, occurring in approximately 1 of every 160 pregnancies in the United States. The rate has remained stagnant since 2006. Prior to that time, from 1990 to 2006, the rate declined somewhat, but only half as much as the decline in infant mortality during this time period. Racial disparities also have persisted, with non-Hispanic black women having more than a twofold increase in risk (Natl Vital Stat Rep. 2012;60:1-22).
Research conducted by the Stillbirth Collaborative Research Network (SCRN) and others has provided us with insight on risk factors and on probable and possible causes of death among stillbirths, which are defined as fetal deaths at 20 or more weeks’ gestation. We know from SCRN data, for instance, that black women are more likely to have stillbirths associated with obstetric complications and infections than white and Hispanic women. However, we still cannot explain a substantial proportion of stillbirths, despite a complete evaluation, or predict who will have a stillbirth.
What we can do as obstetricians is be aware that stillbirth is one of the most common adverse pregnancy outcomes in the United States and counsel women regarding risk factors that are modifiable. Moreover, when stillbirth happens, a complete postmortem evaluation that includes autopsy, placental pathology, karyotype or microarray analysis, and fetal-maternal hemorrhage testing is recommended (Obstet Gynecol. 2009;113[3]:748-61). Recent data show that each of these four components is valuable and should be considered the basic work-up for stillbirth.
Risks and causes
Pregnancy history was the strongest baseline risk factor for stillbirth in an analysis of 614 stillbirths and 1,816 live births in the SCRN’s population-based, case-control study conducted between 2006 and 2008. The SCRN was initiated by the Eunice Kennedy Shriver National Institute of Child Health and Human Development in 2003. This critical population-based study was conducted at 59 U.S. tertiary care and community hospitals in five catchment areas and has been analyzed in more than 15 published reports.
Women with a previous stillbirth have been known to be at 5- to 10-fold increased risk of a recurrence of stillbirth, and the SCRN findings confirmed this. The study added to our knowledge, however, with the finding that even a prior pregnancy loss at less than 20 weeks’ gestation increased the risk for stillbirth.
Other risk factors identified in the study, in addition to race, included having a multifetal pregnancy (adjusted odds ratio of 4.59), diabetes (AOR of 2.50), maternal age of 40 years or older (AOR of 2.41), maternal AB blood type (AOR of 1.96, compared with type O), a history of drug addiction (AOR of 2.08), smoking during the 3 months prior to pregnancy (AOR of 1.55-1.57, depending on amount), and being unmarried and not cohabitating (AOR of 1.69). Regarding racial disparity, the study showed that elevated risk of stillbirth for non-Hispanic blacks occurred predominantly prior to 24 weeks of gestation.
As in prior research, overweight and obesity also conferred elevated risks in the SCRN study (AORs of 1.43 and 1.72, respectively), and these risks were not explained by either diabetes or hypertension (JAMA. 2011;306:2469-79).
The use of assisted reproductive technology was not included in the study’s multivariate model, but previous research has shown a fourfold increased risk of stillbirth for singleton IVF/ICSI pregnancies. The reason is unclear, but the risk appears to be more related to IVF/ICSI rather than the underlying infertility (Hum Reprod. 2010 May;25[5]:1312-6).
A previous preterm or small-for-gestational-age birth has also been shown in prior research to be a significant risk factor for stillbirth. Less clear is the role of previous cesarean delivery in stillbirth risk. An association has been demonstrated in several studies, however, including one involving about 180,000 singleton pregnancies of 23 or more weeks’ gestation. Women in this cohort who had a previous cesarean delivery had a 1.3-fold increased risk of antepartum stillbirth, after controlling for important factors such as race, body mass index (BMI), and maternal disease (Obstet Gynecol. 2010 Nov;116[5]:1119-26).
In another analysis of the SCRN study looking specifically at causes of stillbirth, a “probable” cause of death was found in 61% of cases and a “possible or probable” cause of death in more than 76% of cases. The most common causes were obstetric complications (29.3%), placental abnormalities (23.6%), fetal genetic/structural abnormalities (13.7%), infection (12.9%), umbilical cord abnormalities (10.4%), hypertensive disorders (9.2%), and other maternal medical conditions (7.8%).
A higher proportion of stillbirths in non-Hispanic black women, compared with non-Hispanic white women and Hispanic women was associated with obstetric complications (43.5%) and infections (25.2%). This finding combined with the finding that stillbirth in black women often occurs at less than 24 weeks’ gestation suggests that measures aimed at reducing the rate of spontaneous preterm birth in black women could potentially reduce the rate of stillbirth as well (JAMA. 2011 Dec 14;306[22]:2459-68).
Work-up and prevention
Prevention of stillbirth requires that we identify the women at highest risk, and thus far this ability still eludes us. Apart from occurrence of previous stillbirth or pregnancy loss, other risk factors have had limited predictive value in the SCRN analyses and other research.
Biomarkers such as a low PAPP-A during the first trimester and a high AFP in the second trimester – as well as Doppler imaging of the uterine artery – have also been associated with stillbirth, but again, the positive predictive value has been shown to be low (Clin Obstet Gynecol. 2010 Sep;53[3]:597-606). More research is needed to determine if some combination of biochemical markers, imaging, and other risk factors can predict which women are at highest risk.
In the meantime, attention can be paid – in the preconception period if possible – to modifiable risk factors such as maternal obesity, diabetes, and smoking. About 10% of stillbirths are associated with maternal conditions such as hypertension and diabetes, and late stillbirths in particular (28 weeks or later) are associated with maternal medical conditions that are potentially preventable.
Normalization of prepregnancy weight should be a goal, since the overall risk of stillbirth appears to increase independently with increasing BMI. Glycemic control should also be achieved: A recent meta-analysis of preconception and prenatal care of diabetic women estimated “conservatively” that 10% of diabetes-associated stillbirths could be prevented with early detection and glycemic control (BMC Public Health. 2011;11 Suppl 3:S2). Research has also shown that women who quit smoking between their first and second pregnancy reduce their stillbirth risk to that of nonsmokers in the second pregnancy (BJOG. 2007 Jun;114[6]:699-704).
When stillbirth happens, a thorough work-up is recommended in order to counsel for future pregnancies and decrease the risk of recurrence. Evaluations for causes of stillbirth are too often incomplete in the United States for various reasons, including emotional, cultural, and resource factors. Even if a cause is not found, many families appreciate knowing that every effort has been made to determine a cause of death.
Four components of evaluation – autopsy, placental examination, karyotype or microarray analysis, and fetal-maternal hemorrhage testing – have proven to be high-yield tests when performed in all cases of stillbirth.
In the SCRN study, of 512 stillbirths undergoing a complete evaluation, 66.4% had a positive result – defined as abnormalities contributing to a probable or possible cause – for at least one of the first three tests (JAMA. 2011 Dec 14;306[22]:2459-68).
A Dutch study of 1,025 stillbirths similarly demonstrated that all four tests are justified. A test was defined as valuable in this study if it established or excluded a cause of stillbirth. Placental examination was determined to be the most valuable test, helping to determine a cause of death in 95.7% of cases. Autopsy was valuable 72.6% of the time, and cytogenetic analysis was valuable in 29% of cases.
Kleihauer-Betke testing for fetal-maternal hemorrhage was positive in 11.9% of women. However, fetal maternal hemorrhage was considered the cause of death in only 1.3%.of cases because, beyond a positive Kleihauer-Betke test, evidence of fetal anemia confirmed by placental examination and/or autopsy was required for hemorrhage to be considered the cause of death (Am. J. Obstet. Gynecol. 2012;206:53.e1-12). Because Kleihauer-Betke testing is ideally performed before induction, authors of both the SCRN study and the Dutch study believe it is a valuable test to be offered in all cases.
In both studies, the yield of other stillbirth diagnostic tests (for example, maternal serology, hormone assessment, and toxicology screen) was low, indicating that these tests are considered sequential and can be performed only when the clinical history or findings of the four core tests raise suspicion of particular potential causes. Antinuclear antibody testing and TORCH (toxoplasmosis, rubella, cytomegalovirus, herpes simplex) titers have an extremely low yield and are generally not useful.
For detecting genetic abnormalities after stillbirth, it appears that microarray analysis is superior to karyotype analysis. In a SCRN analysis of samples from 532 stillbirths, microarray yielded results more often and identified more genetic abnormalities. Unlike karyotype, it does not require live cells, which makes it preferable for stillbirth evaluation (N Engl J Med. 2012 Dec 6;367[23]:2185-93).
Current research
One of the more significant studies underway on prevention is looking at labor induction as an intervention for reducing stillbirths and improving other perinatal outcomes. The ARRIVE trial (“A Randomized Trial of Induction Versus Expectant Management”), currently in the recruitment stage, will examine outcomes after induction at 39 weeks’ gestation, compared with expectant management in 6,000 patients (clinicaltrials.gov/ct2/show/NCT01990612).
Common wisdom informed by retrospective cohort studies has long told us that inducing labor prior to 41 weeks’ gestation is associated with a higher risk of cesarean delivery in nulliparous women. However, recent observational data have suggested that women whose labor is induced actually have fewer cesarean deliveries and better perinatal outcomes, including a lower risk of stillbirth (AJOG 2012;207:502.e1-8).
In addition, a meta-analysis published in 2014, as the ARRIVE trial was taking shape, reported a 12% reduction in cesarean delivery, and a reduced risk of stillbirth, among women whose labor was induced. The initial cervical score did not impact the main findings (CMAJ. 2014 Jun 10;186[9]:665-73). If these findings are confirmed in the ARRIVE trial, we could see a new opportunity for stillbirth prevention.
Another ongoing study of 10,000 singleton pregnancies – the Nulliparous Pregnancy Outcomes: Monitoring Mothers-to-Be (nuMoM2b) study – may also lead to prevention strategies in women for whom the current pregnancy will lead to their first delivery. Among the questions being examined in this eight-site study are whether sleep-disordered breathing, or apnea, and a supine sleep position are risk factors for adverse pregnancy outcomes including stillbirth.
Supine sleeping in the last month of pregnancy was strongly associated with stillbirth in a recent analysis from the Sydney Stillbirth Study (Obstet Gynecol. 2015 Feb;125[2]:347-55), and an early analysis of a nuMoM2b subset has shown associations between sleep-disordered breathing in midpregnancy and the development of hypertensive disorders of pregnancy, and between sleep-disordered breathing in early- and mid-pregnancy and gestational diabetes (Am J Obstet Gynecol. 2015;212:S424-425).
The possible role of low-dose aspirin in preventing stillbirth also needs more exploration. A recent randomized trial of women attempting to become pregnant after having had one or two prior pregnancy losses found no difference overall in live birth rates between those who took low-dose aspirin and those assigned to placebo. However, there was one subgroup – women with a single loss at less than 20 weeks’ gestation during the previous year – in which live birth rates were higher in the aspirin group (Lancet. 2014 Jul 5;384[9937]:29-36). More research is necessary to determine if low-dose aspirin administration in women with a previous stillbirth improves pregnancy outcome.
Dr. Reddy is a member at the Pregnancy and Perinatology Branch of the Eunice Kennedy Shriver National Institute of Child Health and Human Development. She is a board-certified ob.gyn. and maternal-fetal medicine specialist. She is the program scientist for the Maternal-Fetal Medicine Units Network and for the Stillbirth Collaborative Research Network. The comments and views of the author do not necessarily represent the views of the NICHD.
Radiofrequency volumetric thermal ablation for symptomatic uterine fibroids
In 2002, Dr. Bruce B. Lee first described a laparoscopic technique to ablate symptomatic uterine fibroids utilizing radiofrequency under ultrasound guidance. Since this time, several papers have documented the procedure’s feasibility and efficacy, including reduction in menstrual blood loss, fibroid volume decrease, and improvement in quality of life.
In a randomized, prospective, single-center, longitudinal study that compared laparoscopic radiofrequency volumetric thermal ablation (RFVTA) of fibroids with laparoscopic myomectomy, Dr. Sara Y. Brucker and her colleagues concluded that RFVTA resulted in the treatment of more fibroids, a significantly shorter hospital stay, and less intraoperative blood loss than did laparoscopic myomectomy (Int J Gynaecol Obstet. 2014 Jun;125[3]:261-5).
More recently, in the literature and at the 2015 American Association of Gynecologic Laparoscopists (AAGL) Global Congress in November, viable, full-term pregnancies have been reported in patients previously treated for symptomatic fibroids via RFVTA (J Reprod Med. 2015 May-Jun;60[5-6]:194-8).
The system for performing RFVTA of symptomatic fibroids – the Acessa System (Halt Medical) – has continued to improve. Earlier this year, Dr. Donald I. Galen described the use of electromagnetic image guidance, which has been cleared by the Food and Drug Administration and incorporated into the Acessa Guidance System. Dr. Galen’s feasibility study showed that the guidance system enhances the ultrasonic image of Acessa’s handpiece to facilitate accurate tip placement during the targeting and ablation of uterine fibroids (Biomed Eng Online. 2015 Oct 15;14:90).
In this edition of the Master Class in Gynecologic Surgery, Dr. Jay M. Berman discusses the use of RFVTA for the treatment of symptomatic uterine fibroids. Dr. Berman is interim chairman of Wayne State University’s department of obstetrics and gynecology and interim specialist in chief for obstetrics and gynecology at the Detroit Medical Center. He served as a principal investigator of the pivotal trial of Acessa and has reported on reproductive outcomes. Dr. Berman has long been interested in alternatives to hysterectomy for fibroid management and has incorporated RFVTA into his armamentarium of therapies.
Dr. Miller is a clinical associate professor at the University of Illinois at Chicago, and a past president of the AAGL and the International Society for Gynecologic Endoscopy (ISGE). He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in private practice in Naperville and Schaumburg, Ill.; director of minimally invasive gynecologic surgery and the director of the AAGL/Society of Reproductive Surgery fellowship in minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill.; and the medical editor of this column, Master Class. Dr. Miller reported that he is a consultant for Halt Medical Inc., which developed the Acessa System.
In 2002, Dr. Bruce B. Lee first described a laparoscopic technique to ablate symptomatic uterine fibroids utilizing radiofrequency under ultrasound guidance. Since this time, several papers have documented the procedure’s feasibility and efficacy, including reduction in menstrual blood loss, fibroid volume decrease, and improvement in quality of life.
In a randomized, prospective, single-center, longitudinal study that compared laparoscopic radiofrequency volumetric thermal ablation (RFVTA) of fibroids with laparoscopic myomectomy, Dr. Sara Y. Brucker and her colleagues concluded that RFVTA resulted in the treatment of more fibroids, a significantly shorter hospital stay, and less intraoperative blood loss than did laparoscopic myomectomy (Int J Gynaecol Obstet. 2014 Jun;125[3]:261-5).
More recently, in the literature and at the 2015 American Association of Gynecologic Laparoscopists (AAGL) Global Congress in November, viable, full-term pregnancies have been reported in patients previously treated for symptomatic fibroids via RFVTA (J Reprod Med. 2015 May-Jun;60[5-6]:194-8).
The system for performing RFVTA of symptomatic fibroids – the Acessa System (Halt Medical) – has continued to improve. Earlier this year, Dr. Donald I. Galen described the use of electromagnetic image guidance, which has been cleared by the Food and Drug Administration and incorporated into the Acessa Guidance System. Dr. Galen’s feasibility study showed that the guidance system enhances the ultrasonic image of Acessa’s handpiece to facilitate accurate tip placement during the targeting and ablation of uterine fibroids (Biomed Eng Online. 2015 Oct 15;14:90).
In this edition of the Master Class in Gynecologic Surgery, Dr. Jay M. Berman discusses the use of RFVTA for the treatment of symptomatic uterine fibroids. Dr. Berman is interim chairman of Wayne State University’s department of obstetrics and gynecology and interim specialist in chief for obstetrics and gynecology at the Detroit Medical Center. He served as a principal investigator of the pivotal trial of Acessa and has reported on reproductive outcomes. Dr. Berman has long been interested in alternatives to hysterectomy for fibroid management and has incorporated RFVTA into his armamentarium of therapies.
Dr. Miller is a clinical associate professor at the University of Illinois at Chicago, and a past president of the AAGL and the International Society for Gynecologic Endoscopy (ISGE). He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in private practice in Naperville and Schaumburg, Ill.; director of minimally invasive gynecologic surgery and the director of the AAGL/Society of Reproductive Surgery fellowship in minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill.; and the medical editor of this column, Master Class. Dr. Miller reported that he is a consultant for Halt Medical Inc., which developed the Acessa System.
In 2002, Dr. Bruce B. Lee first described a laparoscopic technique to ablate symptomatic uterine fibroids utilizing radiofrequency under ultrasound guidance. Since this time, several papers have documented the procedure’s feasibility and efficacy, including reduction in menstrual blood loss, fibroid volume decrease, and improvement in quality of life.
In a randomized, prospective, single-center, longitudinal study that compared laparoscopic radiofrequency volumetric thermal ablation (RFVTA) of fibroids with laparoscopic myomectomy, Dr. Sara Y. Brucker and her colleagues concluded that RFVTA resulted in the treatment of more fibroids, a significantly shorter hospital stay, and less intraoperative blood loss than did laparoscopic myomectomy (Int J Gynaecol Obstet. 2014 Jun;125[3]:261-5).
More recently, in the literature and at the 2015 American Association of Gynecologic Laparoscopists (AAGL) Global Congress in November, viable, full-term pregnancies have been reported in patients previously treated for symptomatic fibroids via RFVTA (J Reprod Med. 2015 May-Jun;60[5-6]:194-8).
The system for performing RFVTA of symptomatic fibroids – the Acessa System (Halt Medical) – has continued to improve. Earlier this year, Dr. Donald I. Galen described the use of electromagnetic image guidance, which has been cleared by the Food and Drug Administration and incorporated into the Acessa Guidance System. Dr. Galen’s feasibility study showed that the guidance system enhances the ultrasonic image of Acessa’s handpiece to facilitate accurate tip placement during the targeting and ablation of uterine fibroids (Biomed Eng Online. 2015 Oct 15;14:90).
In this edition of the Master Class in Gynecologic Surgery, Dr. Jay M. Berman discusses the use of RFVTA for the treatment of symptomatic uterine fibroids. Dr. Berman is interim chairman of Wayne State University’s department of obstetrics and gynecology and interim specialist in chief for obstetrics and gynecology at the Detroit Medical Center. He served as a principal investigator of the pivotal trial of Acessa and has reported on reproductive outcomes. Dr. Berman has long been interested in alternatives to hysterectomy for fibroid management and has incorporated RFVTA into his armamentarium of therapies.
Dr. Miller is a clinical associate professor at the University of Illinois at Chicago, and a past president of the AAGL and the International Society for Gynecologic Endoscopy (ISGE). He is a reproductive endocrinologist and minimally invasive gynecologic surgeon in private practice in Naperville and Schaumburg, Ill.; director of minimally invasive gynecologic surgery and the director of the AAGL/Society of Reproductive Surgery fellowship in minimally invasive gynecologic surgery at Advocate Lutheran General Hospital, Park Ridge, Ill.; and the medical editor of this column, Master Class. Dr. Miller reported that he is a consultant for Halt Medical Inc., which developed the Acessa System.