User login
Genitourinary endometriosis: Diagnosis and management
Endometriosis is a benign disease characterized by endometrial glands and stroma outside of the uterine cavity. It is commonly associated with pelvic pain and infertility. Ectopic endometrial tissue is predominantly located in the pelvis, but it can appear anywhere in the body, where it is referred to as extragenital endometriosis. The bowel and urinary tract are the most common sites of extragenital endometriosis.1
Laparoscopic management of extragenital endometriosis has been described since the 1980s.2 However, laparoscopic management of genitourinary endometriosis is still not widespread.3,4 Physicians are often unfamiliar with the signs and symptoms of genitourinary endometriosis and fail to consider it when a patient presents with bladder pain or hematuria, which may or may not be cyclic. Furthermore, many gynecologists do not have the experience to correctly identify the various forms of endometriosis that may appear on the pelvic organ, including the serosa and peritoneum, as variable colored spots, blebs, lesions, or adhesions. Many surgeons are also not adequately trained in the advanced laparoscopic techniques required to treat genitourinary endometriosis.4
In this article, we describe the clinical presentation and diagnosis of genitourinary endometriosis and discuss laparoscopic management strategies with and without robotic assistance.
Clinical presentation and diagnosis of genitourinary endometriosis
While ureteral and bladder endometriosis are both diseases of the urinary tract, they are not always found together in the same patient. The bladder is the most commonly affected organ, followed by the ureter and kidney.3,5,6 Endometriosis of the bladder usually presents with significant lower urinary tract symptoms. In contrast, ureteral endometriosis is usually silent with no apparent urinary symptoms.
Ureteral endometriosis. Cyclic hematuria is present in less than 15% of patients with ureteral endometriosis. Some patients experience cyclic, nonspecific colicky flank pain.7-9 Otherwise, most patients present with the usual symptoms of endometriosis, such as pelvic pain and dysmenorrhea. In a systematic review, Cavaco-Gomes and colleagues described 700 patients with ureteral endometriosis; 81% reported dysmenorrhea, 70% had pelvic pain, and 66% had dyspareunia.10 Rarely, ureteral endometriosis can result in silent kidney loss if the ureter becomes severely obstructed without treatment.11,12
Continue to: The lack of symptoms makes...
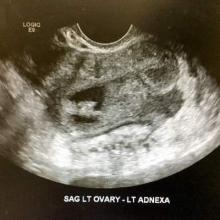
The lack of symptoms makes the early diagnosis of ureteral endometriosis difficult. As with all types of endometriosis, histologic evaluation of a biopsy sample is diagnostic. Multiple imaging modalities have been used to preoperatively diagnose ureteral involvement, including computed tomography,13 magnetic resonance imaging (MRI),14 intravenous pyelogram (IVP), and pelvic ultrasonography. However, each of these imaging modalities is limited in both sensitivity and the ability to characterize depth of tissue invasion.
In a study of 245 women undergoing pelvic ultrasonography, Pateman and colleagues reported that an experienced sonographer was able to visualize the bilateral ureters in 93% of cases.15 Renal ultrasonography is indicated in any woman suspected of having genitourinary tract involvement with the degree of hydroureter and level of obstruction noted during the exam.16
In our group, we perform renography to assess kidney function when hydroureter is noted preoperatively. Studies suggest that if greater than 10% of normal glomerular filtration rate remains, the kidney is considered salvageable, and near-normal function often returns following resection of disease. If preoperative kidney function is noted to be less than 10%, consultation with a nephrologist for possible nephrectomy is warranted.
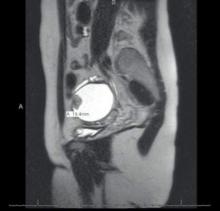
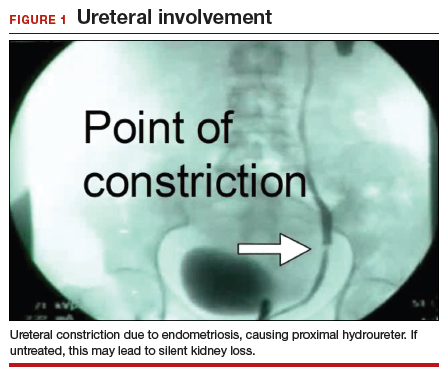
We find that IVP is often helpful for preoperatively identifying the level and degree of ureteral involvement, and it also can be used postoperatively to evaluate for ureteral continuity (FIGURE 1). Sillou and colleagues showed MRI to be adequately sensitive for the detection of intrinsic ureteral endometriosis, but they reported that MRI often overestimates the frequency of disease.17 Authors of a 2016 Cochrane review of imaging modalities for endometriosis, including 4,807 women in 49 studies, reported that no imaging test was superior to surgery for diagnosing endometriosis.18 However, the review notably excluded genitourinary tract endometriosis, as surgery is not an acceptable reference standard, given that many surgeons cannot reliably identify such lesions.18
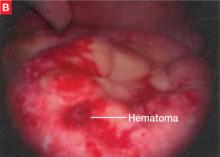
Bladder endometriosis. Unlike patients with ureteral endometriosis, those with bladder endometriosis are typically symptomatic and experience dysuria, hematuria, urinary frequency, and suprapubic tenderness.7,19 Urinary tract infection, interstitial cystitis, and cancer must be considered in the differential diagnosis. Urinalysis and urine culture should be performed, and other diagnostic procedures such as ultrasonography, MRI, and cystoscopy should be considered to evaluate for endometriosis of the bladder.
Ultrasound and MRI of the bladder both demonstrate a high specificity for detecting bladder endometriosis, but they lack sensitivity for lesions less than 3 cm.20 Deep infiltrating endometriosis of the bladder can be identified at the time of cystoscopy, which can assist in determining the need for ureteroneocystostomy if lesions are within 2 cm of the urethral opening.20 Cystoscopy also allows for biopsy to be performed if underlying malignancy is of concern.19
In our group, when bladder endometriosis is suspected, we routinely perform preoperative bladder ultrasonography to identify the lesion and plan to perform intraoperative cystoscopy at the time of laparoscopic resection.19,21
Continue to: Treatment...
Treatment
Medical management
Empiric medical therapies for endometriosis are centered around the idea that ectopic endometrial tissue responds to treatment in a similar manner as normal eutopic endometrium. The goals of treatment are to reduce or eliminate cyclic menstruation, thereby decreasing peritoneal seeding and suppressing the growth and activity of established ectopic implants. Medical therapy commonly involves the use of gonadotropin-releasing hormone agonists or antagonists, danazol, combined oral contraceptives, progestins, and aromatase inhibitors.
Medical therapy is commonly employed for patients with mild or early-stage disease and in those who are poor surgical candidates or decline surgery. Medical management alone clearly is contraindicated in the setting of ureteral obstruction and—in our opinion—may not be a good option for those with endometriosis of the ureter, given the potential for recurrence and potential serious sequelae of reduced renal function.22 Therefore, surgery has become the standard approach to therapy for mild to moderate cases of ureteral endometriosis.3
Medical therapy for patients with endometriosis of the bladder is generally considered a temporary solution as hormonal suppression, with its associated adverse effects, must be maintained throughout menopause. However, if endometriosis lesions lie within close proximity to the trigone, medical management is preferred, as surgical excision in the area of the trigone may predispose patients to neurogenic bladder and retrograde bladder reflux.23,24
Surgical management
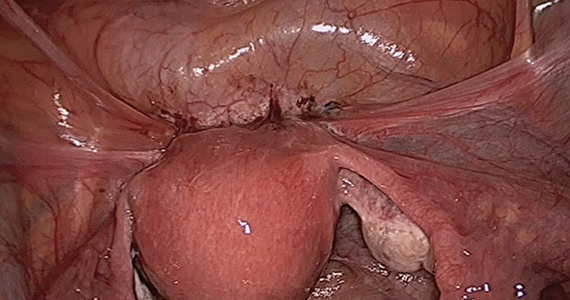
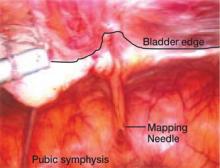
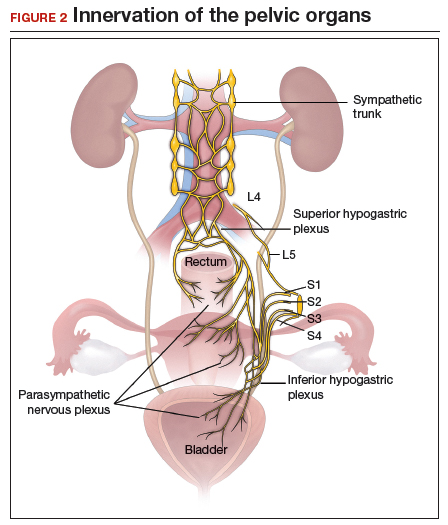
The objectives of surgical treatment for genitourinary tract endometriosis are to excise all visible disease, to prevent or delay recurrence of the disease to the extent possible, and to avoid any further morbidity—in particular, to preserve renal function in cases of ureteral endometriosis—and to avoid iatrogenic injury to surrounding pelvic nervous structures25-27 (FIGURE 2). The surgical approach depends on the technical expertise of the surgeon and the availability of necessary instrumentation. In our experience, laparoscopy with or without robotic assistance is the preferred surgical approach.3,4,6,11,28-32
Others have reported on the benefits of laparoscopy over laparotomy for the surgical management of genitourinary endometriosis. In a review of 61 patients who underwent either robot-assisted laparoscopic (n = 25) or open (n = 41) ureteroneocystostomy (n = 41), Isac and colleagues reported the procedure was longer in the laparoscopic group (279 min vs 200 min, P<.001), but the laparoscopic group had a shorter hospital stay (3 days vs 5 days, P<.001), used fewer narcotics postoperatively (P<.001), and had lower intraoperative blood loss (100 mL vs 150 mL, P<.001).32 No differences in long-term outcomes were observed in either cohort.
In a systematic review of 700 patients undergoing laparoscopic surgery for ureteral endometriosis, Cavaco-Gomes and colleagues reported that conversion to laparotomy occurred in only 3% to 7% of cases.10 In instances of ureteral endometriosis, laparoscopy provides greater visualization of the intraperitoneal contents over laparotomy, enabling better evaluation and treatment of lesions.3,29,33,34 Robot-assisted laparoscopy provides the additional advantages of 3D visualization, potential for an accelerated learning curve over traditional laparoscopy, improvement in dissection technique, and ease of suturing technique.6,35,36
Continue to: Extrinsic disease...
Extrinsic disease. In our group, we perform ureterolysis for extrinsic disease.25 The peritoneal incision is made in an area unaffected by endometriosis. Using the suction irrigator, a potential space is developed under the serosa of the ureter by injecting normal saline or lactated Ringer’s solution. By creating a fluid barrier between the serosa and underlying tissues, the depth of surgical incision and lateral thermal spread are minimized. Grasping forceps are used to peel the peritoneum away.25,37,38
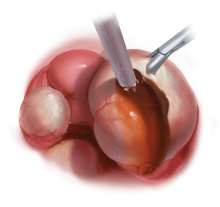
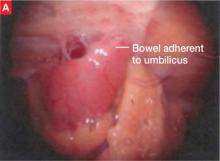
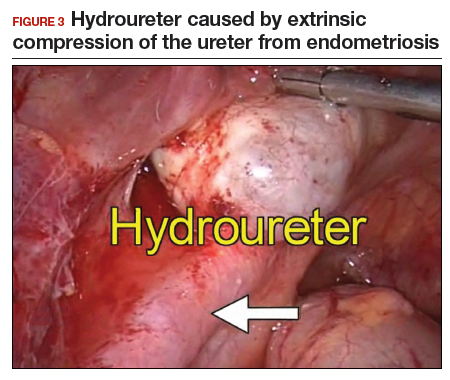
Intrinsic disease. Unlike extrinsic disease, intrinsic disease can infiltrate the muscularis, lamina propria, and ureteral lumen, resulting in proximal dilation of the ureter with strictures.8 In this situation, ureteral compromise is likely and resection of the ureter is indicated3,28 (FIGURE 3). Intrinsic disease can be suggested by preoperative imaging or when there is evidence of deep infiltrating disease on physical exam, such as rectovaginal nodularity.30,39 When intrinsic ureteral disease is known, consultation with a urologist to plan a joint procedure is advisable. The procedure chosen to re-establish a functional ureter following resection depends on the location and extent of the involved ureter. Resection in close proximity to the bladder may be repaired by ureteroneocystostomy with or without psoas hitch,30,39,40 whereas resection of more proximal ureter may be repaired using Boari flap, ileal interposition, or autotransplantation. Lesions in the upper third or middle ureter may be repaired using ureterouretral anastomosis.6,7,30,41-43
Continue to: Bladder endometriosis...
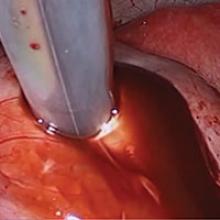
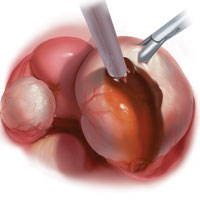
Bladder endometriosis. Surgical treatment for bladder endometriosis depends on the depth of invasion and the location of the lesion (FIGURE 4). Lesions of the superficial aspect of the bladder identified at the time of laparoscopy can be treated with either excision or fulguration28,35,44 In our group, we perform excision over fulguration to remove the entire lesion and obtain a pathologic diagnosis. Deeper lesions involving the detrusor muscle are likely to be an endometrioma of the bladder. In these cases, laparoscopic excision is recommended.7 Rarely, lesions close to the interureteric ridge may require ureteroneocystostomy.19,45
In our experience, laparoscopic resection of bladder endometriomas is associated with better results in terms of symptom relief, progression of disease, and recurrence risk compared with other approaches. When performing laparoscopic excision of bladder lesions, we concomitantly evaluate the bladder lesion via cystoscopy to ensure adequate margins are obtained. Double-J stent placement is advised when lesions are within 2 cm of the urethral meatus to ensure ureteral patency during the postoperative period.45 A postoperative cystogram routinely is performed 7 to 14 days after surgery to ensure adequate repair prior to removing the urinary catheter.9,25,46,47

Postsurgical follow-up
Follow-up after treatment of genitourinary tract endometriosis should include monitoring the patient for symptoms of recurrence. Regular history and physical examination, renal function testing, and, in some instances, pelvic ultrasonography, all have a role in surveillance for recurrent ureteric disease. IVP or MRI may be warranted if a recurrence is suspected. A high index of suspicion should be maintained on the part of the clinician to avoid the devastating consequences of silent kidney loss. Patients should be counseled about the risk of disease recurrence, especially in those not undergoing postoperative hormonal suppression.
In conclusion
While endometriosis of the genitourinary tract is rare, patients can experience significant morbidity. Medical management of the disease is often limited by substantial adverse effects that limit treatment duration and is best used postoperatively after excision. An adequate physical exam and preoperative diagnostic imaging can be employed to characterize the extent of disease. When extensive disease involving the ureter is suspected, preoperative consultation with a urologist is encouraged to plan a multidisciplinary approach to surgical resection.
The ideal approach to surgery is laparoscopic resection with or without robotic assistance. Treatment of ureteral disease most commonly involves ureterolysis for cases of extrinsic disease but may require total resection of the ureter with concurrent ureteral reconstruction when disease is intrinsic to the ureter. Surgery for bladder endometriosis depends on the depth of invasion and location of the lesion. Superficial bladder lesions can be treated with fulguration or excision, while deeper lesions involving the detrusor muscle require excision. Lesions in close proximity to the interureteric ridge may require ureteroneocystostomy. Follow-up after excisional procedures involves monitoring the patient for signs and symptoms of disease recurrence, especially in cases of ureteral involvement, to avoid the devastating consequences of silent kidney loss.
The definitive cause of endometriosis remains unknown, but several prominent theories have been proposed.
Sampson's theory of retrograde menstruation through the fallopian tubes is the most well-known theory,1 although Schron had acknowledged a similar thought 3 centuries before.2 This theory posits that refluxed endometrial cells enter the abdominal cavity and invade the peritoneum, developing a blood supply necessary for survival and growth. Early reports supported this theory by suggesting that women with genital tract obstruction have a higher incidence of endometriosis.3,4 However, it was later confirmed that women without genital tract obstruction have a similar incidence of retrograde menstruation. In fact, up to 90% of women are found to have retrograde menstruation, but only 10% develop endometriosis. This suggests that once endometrial cells have escaped the uterine cavity, other events are necessary for endometrial cells to implant and survive.3,5 Other evidence to support the theory of retrograde menstruation is the observation that endometriosis is most commonly observed in the dependent portions of the pelvis, on the ovaries, in the anterior and posterior cul-de-sacs, and on the uterosacral ligament.6
The coelomic metaplasia theory holds that endometriosis results from spontaneous metaplastic change to mesothelial cells derived from the coelomic epithelium (located in the peritoneum and the pleura) upon exposure to menstrual effluent or other stimuli.7 Evidence for this theory is supported by the observation that intact endometrial cells have no access to the thoracic cavity in the absence of anatomical defect; therefore, the implantation theory cannot explain cases of pleural or pulmonary endometriosis.
Immune dysregulation also has been invoked to explain endometriosis implants both inside and outside the genitourinary tract. Studies have shown a higher incidence of endometriosis in women with other autoimmune disorders, including hypothyroidism, chronic fatigue syndrome, rheumatoid arthritis, systemic lupus erythematosus, Sjogren syndrome, and multiple sclerosis as well as in women with allergies, asthma, and eczema.8 In such women, dysregulation of the innate and adaptive immune system might promote the disease by inhibiting apoptosis of ectopic endometrial cells and by promoting their attachment, invasion, and proliferation into healthy peritoneum through the secretion of various growth factors and cytokines.9,10
Other possible theories that might explain the pathogenesis of endometriosis exist.11-13 While each theory has documented supporting evidence, no single theory currently accounts for all cases of endometriosis. It is likely, then, that endometriosis is a multifactorial disease with a combination of immune dysregulation, molecular abnormalities, genetic and epigenetic factors, and environmental exposures involved in its pathogenesis.
References
- Sampson J. Peritoneal endometriosis due to the menstrual dissemination of endometrial tissue into the peritoneal cavity. Am J Obstet Gynecol. 1927;14:422-469.
- Nezhat C, Nezhat F, Nezhat C. Endometriosis: ancient disease, ancient treatments. Fertil Steril. 2012;98(6 suppl):S1-62.
- Halme J, Hammond MG, Hulka JF, et al. Retrograde menstruation in healthy women and in patients with endometriosis. Obstet Gynecol. 1984;64:151-154.
- Sanfilippo JS, Wakim NG, Schikler KN, et al. Endometriosis in association with uterine anomaly. Am J Obstet Gynecol. 1986;154:39-43.
- Burney RO, Giudice LC. Pathogenesis and pathophysiology of endometriosis. Fertil Steril. 2012;98:511-519.
- Vercellini P, Chapron C, Fedele L, et al. Evidence for asymmetric distribution of lower intestinal tract endometriosis. BJOG. 2004;111:1213-1217.
- Sourial S, Tempest N, Hapangama DK. Theories on the pathogenesis of endometriosis. Int J Reprod Med. 2014;2014:179515.
- Sinaii N, Cleary SD, Ballweg ML, et al. High rates of autoimmune and endocrine disorders, fibromyalgia, chronic fatigue syndrome and atopic diseases among women with endometriosis: a survey analysis. Hum Reprod. 2002;17:2715-2724.
- Lebovic DI, Mueller MD, Taylor RN. Immunobiology of endometriosis. Fertil Steril. 2001;75:1-10.
- Sidell N, Han SW, Parthasarathy S. Regulation and modulation of abnormal immune responses in endometriosis. Ann N Y Acad Sci. 2002;955: 159-173; discussion 199-200, 396-406.
- Burney RO, Giudice LC. The pathogenesis of endometriosis. In: Nezhat C, Nezhat F, Nezhat C, eds. Nezhat's Video-Assisted and Robotic-Assisted Laparoscopy and Hysteroscopy. 4th ed. New York, NY: Cambridge University Press; 2013;252-258.
- Buka NJ. Vesical endometriosis after cesarean section. Am J Obstet Gynecol. 1988;158:1117-1118.
- Price DT, Maloney KE, Ibrahim GK, et al. Vesical endometriosis: report of two cases and review of the literature. Urology. 1996;48:639-643.
- Veeraswamy A, Lewis M, Mann A, et al. Extragenital endometriosis. Clin Obstet Gynecol. 2010;53:449-466.
- Nezhat C, Crowgey SR, Garrison GP. Surgical treatment of endometriosis via laser laparoscopy. Fertil Steril. 1986;45:778-783.
- Bosev D, Nicoll LM, Bhagan L, et al. Laparoscopic management of ureteral endometriosis: the Stanford University hospital experience with 96 consecutive cases. J Urol. 2009;182:2748-2752.
- Nezhat C, Falik R, McKinney S, et al. Pathophysiology and management of urinary tract endometriosis. Nat Rev Urol. 2017;14:359-372.
- Shook TE, Nyberg LM. Endometriosis of the urinary tract. Urology. 1988;31:1-6.
- Nezhat C, Modest AM, King LP. The role of the robot in treating urinary tract endometriosis. Curr Opin Obstet Gynecol. 2013;25:308-311.
- Comiter CV. Endometriosis of the urinary tract. Urol Clin North Am. 2002;29:625-635.
- Gustilo-Ashby AM, Paraiso MF. Treatment of urinary tract endometriosis. J Minim Invasive Gynecol. 2006;13:559-565.
- Berlanda N, Somigliana E, Frattaruolo MP, et al. Surgery versus hormonal therapy for deep endometriosis: is it a choice of the physician? Eur J Obstet Gyneocol Reprod Biol. 2017;209:67-71.
- Cavaco-Gomes J, Martinho M, Gilabert-Aguilar J, et al. Laparoscopic management of ureteral endometriosis: a systematic review. Eur J Obstet Gynecol Reprod Biol. 2017;210:94-101.
- Nezhat C, Nezhat F, Green B. Laparoscopic treatment of obstructed ureter due to endometriosis by resection and ureteroureterostomy: a case report. J Urol. 1992;148:865-868.
- Nezhat C, Paka C, Gomaa M, et al. Silent loss of kidney secondary to ureteral endometriosis. JSLS. 2012;16:451-455.
- Iosca S, Lumia D, Bracchi E, et al. Multislice computed tomography with colon water distention (MSCT-c) in the study of intestinal and ureteral endometriosis. Clin Imaging. 2013;37(6):1061-1068.
- Medeiros LR, Rosa MI, Silva BR, et al. Accuracy of magnetic resonance in deeply infiltrating endometriosis: a systematic review and meta-analysis. Arch Gynecol Obstet. 2015;291:611-621.
- Pateman K, Mavrelos D, Hoo WL, et al. Visualization of ureters on standard gynecological transvaginal scan: a feasibility study. Ultrasound Obstet Gynecol. 2013;41:696-701.
- Guerriero S, Condous G, van den Bosch T, et al. Systematic approach to sonographic evaluation of the pelvis in women with suspected endometriosis, including terms, definitions and measurements: a consensus opinion from the International Deep Endometriosis Analysis (IDEA) group. Ultrasound Obstet Gynecol. 2016;48:318-332.
- Sillou S, Poirée S, Millischer AE, et al. Urinary endometriosis: MR imaging appearance with surgical and histological correlations. Diagn Interv Imaging. 2015;96:373-381.
- Nisenblat V, Bossuyt PM, Farquhar C, et al. Imaging modalities for the non-invasive diagnosis of endometriosis. Cochrane Database Syst Rev. 2016;2:CD009591.
- Nezhat CH, Malik S, Osias J, et al. Laparoscopic management of 15 patients with infiltrating endometriosis of the bladder and a case of primary intravesical endometrioid adenosarcoma. Fertil Steril. 2002;78:872-875.
- Kolodziej A, Krajewski W, Dolowy L, et al. Urinary tract endometriosis. Urol J. 2015;12:2213-2217.
- Nezhat C, Buescher E, Paka C, et al. Video-assisted laparoscopic treatment of endometriosis. In: Nezhat C, Nezhat F, Nezhat C, eds. Nezhat's Video-Assisted and Robotic-Assisted Laparoscopy and Hysteroscopy. 4th ed. New York, NY: Cambridge University Press; 2013;265.
- Al-Fozan H, Tulandi T. Left lateral predisposition of endometriosis and endometrioma. Obstet Gynecol. 2003;101:164-166.
- Hastings JC, Van Winkle W, Barker E, et al. The effect of suture materials on healing wounds of the bladder. Surg Gynecol Obstet. 1975;140:933-937.
- Cornell KK. Cystotomy, partial cystectomy, and tube cystostomy. Clin Tech Small Anim Pract. 2000;15:11-16.
- Nezhat C, Nezhat F, Nezhat C, eds. Nezhat's Video-Assisted and Robotic-Assisted Laparoscopy and Hysteroscopy. 4th ed. New York, NY: Cambridge University Press; 2013.
- Uccella S, Cromi A, Casarin J, et al. Laparoscopy for ureteral endometriosis: surgical details, long-term follow-up, and fertility outcomes. Fertil Steril. 2014;102:160-166.e2.
- Knabben L, Imboden S, Fellmann B, et al. Urinary tract endometriosis in patients with deep infiltrating endometriosis: prevalence, symptoms, management, and proposal for a new clinical classification. Fertil Steril. 2015;103:147-152.
- Nezhat C, Nezhat F, Nezhat CH, et al. Urinary tract endometriosis treated by laparoscopy. Fertil Steril. 1996;66:920-924.
- Nezhat CH, Nezhat F, Seidman D, et al. Laparoscopic ureteroureterostomy: a prospective follow-up of 9 patients. Prim Care Update Ob Gyns. 1998;5:200.
- Nezhat CH, Bracale U, Scala A, et al. Laparoscopic ureteroneocystostomy and vesicopsoas hitch for infiltrative endometriosis. JSLS. 2004;8:3-7.
- Nezhat C, Lewis M, Kotikela S, et al. Robotic versus standard laparoscopy for the treatment of endometriosis. Fertil Steril. 2010;94:2758-2760.
- Isac W, Kaouk J, Altunrende F, et al. Robotic-assisted ureteroneocytostomy: techniques and comparative outcomes. J Endourol. 2013;27:318-323.
- Nezhat C, Nezhat F. Laparoscopic repair of ureter resected during operative laparoscopy. Obstet Gynecol. 1992;80(3 pt 2):543-544.
- De Cicco C, Ussia A, Koninckx PR. Laparoscopic ureteral repair in gynaecological surgery. Curr Opin Obstet Gynecol. 2011;23:296-300.
- Nezhat C, Hajhosseini B, King LP. Robotic-assisted laparoscopic treatment of bowel, bladder, and ureteral endometriosis. JSLS. 2011;15:387-392.
- Fadhlaoui A, Gillon T, Lebbi I, et al. Endometriosis and vesico-sphincteral disorders. Front Surg. 2015;2:23.
- Nezhat C, Nezhat FR. Safe laser endoscopic excision or vaporization of peritoneal endometriosis. Fertil Steril. 1989;52:149-151.
- Nezhat C, Winer W, Nezhat FA. Comparison of the CO2, argon, and KTP/532 lasers in the videolaseroscopic treatment of endometriosis. J Gynecol Surg. 2009;41-47.
- Azioni G, Bracale U, Scala A, et al. Laparoscopic ureteroneocytostomy and vesicopsoas hitch for infiltrative ureteral endometriosis. Minim Invasive Ther Allied Technol. 2010;19:292-297.
- Stepniewska A, Grosso G, Molon A, et al. Ureteral endometriosis: clinical and radiological follow-up after laparoscopic ureterocystoneostomy. Hum Reprod. 2011;26:112-116.
- Nezhat CH, Nezhat FR, Freiha F, et al. Laparoscopic vesicopsoas hitch for infiltrative ureteral endometriosis. Fertil Steril. 1999;71:376-379.
- Scioscia M, Molon A, Grosso G, et al. Laparoscopic management of ureteral endometriosis. Curr Opin Obstet Gynecol. 2009;21:325-328.
- Antonelli A. Urinary tract endometriosis. Urologia. 2012;79:167-170.
- Camanni M, Bonino L, Delpiano EM, et al. Laparoscopic conservative management of ureteral endometriosis: a survey of eighty patients submitted to ureterolysis. Reprod Biol Endocrinol. 2009;7:109.
- Chapron C, Bourret A, Chopin N, et al. Surgery for bladder endometriosis: long-term results and concomitant management of associated posterior deep lesions. Hum Reprod. 2010;25:884-889.
- Nezhat CR, Nezhat FR. Laparoscopic segmental bladder resection for endometriosis: a report of two cases. Obstet Gynecol. 1993;81(5 pt 2):882-884.
- Bourdel N, Cognet S, Canis M, et al. Laparoscopic ureteroneocystostomy: be prepared! J Minim Invasive Gynecol. 2015;22:827-833.
- Page B. Camran Nezhat and the Advent of Advanced Operative Video-laparoscopy. In: Nezhat C, ed. Nezhat's History of Endoscopy. Tuttlingen, Germany: Endo Press; 2011:159-187.
- Podratz K. Degrees of Freedom: Advances in Gynecological and Obstetrical Surgery. Remembering Milestones and Achievements in Surgery: Inspiring Quality for a Hundred Years 1913-2012. Published by American College of Surgeons 2012. Tampa, FL: Faircount Media Group; 2013.
- Kelley WE. The evolution of laparoscopy and the revolution in surgery in the decade of the 1990s. JSLS: J Soc Laparoendoscopic Surgeons. 2008;12:351-357.
Endometriosis is a benign disease characterized by endometrial glands and stroma outside of the uterine cavity. It is commonly associated with pelvic pain and infertility. Ectopic endometrial tissue is predominantly located in the pelvis, but it can appear anywhere in the body, where it is referred to as extragenital endometriosis. The bowel and urinary tract are the most common sites of extragenital endometriosis.1
Laparoscopic management of extragenital endometriosis has been described since the 1980s.2 However, laparoscopic management of genitourinary endometriosis is still not widespread.3,4 Physicians are often unfamiliar with the signs and symptoms of genitourinary endometriosis and fail to consider it when a patient presents with bladder pain or hematuria, which may or may not be cyclic. Furthermore, many gynecologists do not have the experience to correctly identify the various forms of endometriosis that may appear on the pelvic organ, including the serosa and peritoneum, as variable colored spots, blebs, lesions, or adhesions. Many surgeons are also not adequately trained in the advanced laparoscopic techniques required to treat genitourinary endometriosis.4
In this article, we describe the clinical presentation and diagnosis of genitourinary endometriosis and discuss laparoscopic management strategies with and without robotic assistance.
Clinical presentation and diagnosis of genitourinary endometriosis
While ureteral and bladder endometriosis are both diseases of the urinary tract, they are not always found together in the same patient. The bladder is the most commonly affected organ, followed by the ureter and kidney.3,5,6 Endometriosis of the bladder usually presents with significant lower urinary tract symptoms. In contrast, ureteral endometriosis is usually silent with no apparent urinary symptoms.
Ureteral endometriosis. Cyclic hematuria is present in less than 15% of patients with ureteral endometriosis. Some patients experience cyclic, nonspecific colicky flank pain.7-9 Otherwise, most patients present with the usual symptoms of endometriosis, such as pelvic pain and dysmenorrhea. In a systematic review, Cavaco-Gomes and colleagues described 700 patients with ureteral endometriosis; 81% reported dysmenorrhea, 70% had pelvic pain, and 66% had dyspareunia.10 Rarely, ureteral endometriosis can result in silent kidney loss if the ureter becomes severely obstructed without treatment.11,12
Continue to: The lack of symptoms makes...
The lack of symptoms makes the early diagnosis of ureteral endometriosis difficult. As with all types of endometriosis, histologic evaluation of a biopsy sample is diagnostic. Multiple imaging modalities have been used to preoperatively diagnose ureteral involvement, including computed tomography,13 magnetic resonance imaging (MRI),14 intravenous pyelogram (IVP), and pelvic ultrasonography. However, each of these imaging modalities is limited in both sensitivity and the ability to characterize depth of tissue invasion.
In a study of 245 women undergoing pelvic ultrasonography, Pateman and colleagues reported that an experienced sonographer was able to visualize the bilateral ureters in 93% of cases.15 Renal ultrasonography is indicated in any woman suspected of having genitourinary tract involvement with the degree of hydroureter and level of obstruction noted during the exam.16
In our group, we perform renography to assess kidney function when hydroureter is noted preoperatively. Studies suggest that if greater than 10% of normal glomerular filtration rate remains, the kidney is considered salvageable, and near-normal function often returns following resection of disease. If preoperative kidney function is noted to be less than 10%, consultation with a nephrologist for possible nephrectomy is warranted.
We find that IVP is often helpful for preoperatively identifying the level and degree of ureteral involvement, and it also can be used postoperatively to evaluate for ureteral continuity (FIGURE 1). Sillou and colleagues showed MRI to be adequately sensitive for the detection of intrinsic ureteral endometriosis, but they reported that MRI often overestimates the frequency of disease.17 Authors of a 2016 Cochrane review of imaging modalities for endometriosis, including 4,807 women in 49 studies, reported that no imaging test was superior to surgery for diagnosing endometriosis.18 However, the review notably excluded genitourinary tract endometriosis, as surgery is not an acceptable reference standard, given that many surgeons cannot reliably identify such lesions.18

Bladder endometriosis. Unlike patients with ureteral endometriosis, those with bladder endometriosis are typically symptomatic and experience dysuria, hematuria, urinary frequency, and suprapubic tenderness.7,19 Urinary tract infection, interstitial cystitis, and cancer must be considered in the differential diagnosis. Urinalysis and urine culture should be performed, and other diagnostic procedures such as ultrasonography, MRI, and cystoscopy should be considered to evaluate for endometriosis of the bladder.
Ultrasound and MRI of the bladder both demonstrate a high specificity for detecting bladder endometriosis, but they lack sensitivity for lesions less than 3 cm.20 Deep infiltrating endometriosis of the bladder can be identified at the time of cystoscopy, which can assist in determining the need for ureteroneocystostomy if lesions are within 2 cm of the urethral opening.20 Cystoscopy also allows for biopsy to be performed if underlying malignancy is of concern.19
In our group, when bladder endometriosis is suspected, we routinely perform preoperative bladder ultrasonography to identify the lesion and plan to perform intraoperative cystoscopy at the time of laparoscopic resection.19,21
Continue to: Treatment...
Treatment
Medical management
Empiric medical therapies for endometriosis are centered around the idea that ectopic endometrial tissue responds to treatment in a similar manner as normal eutopic endometrium. The goals of treatment are to reduce or eliminate cyclic menstruation, thereby decreasing peritoneal seeding and suppressing the growth and activity of established ectopic implants. Medical therapy commonly involves the use of gonadotropin-releasing hormone agonists or antagonists, danazol, combined oral contraceptives, progestins, and aromatase inhibitors.
Medical therapy is commonly employed for patients with mild or early-stage disease and in those who are poor surgical candidates or decline surgery. Medical management alone clearly is contraindicated in the setting of ureteral obstruction and—in our opinion—may not be a good option for those with endometriosis of the ureter, given the potential for recurrence and potential serious sequelae of reduced renal function.22 Therefore, surgery has become the standard approach to therapy for mild to moderate cases of ureteral endometriosis.3
Medical therapy for patients with endometriosis of the bladder is generally considered a temporary solution as hormonal suppression, with its associated adverse effects, must be maintained throughout menopause. However, if endometriosis lesions lie within close proximity to the trigone, medical management is preferred, as surgical excision in the area of the trigone may predispose patients to neurogenic bladder and retrograde bladder reflux.23,24
Surgical management
The objectives of surgical treatment for genitourinary tract endometriosis are to excise all visible disease, to prevent or delay recurrence of the disease to the extent possible, and to avoid any further morbidity—in particular, to preserve renal function in cases of ureteral endometriosis—and to avoid iatrogenic injury to surrounding pelvic nervous structures25-27 (FIGURE 2). The surgical approach depends on the technical expertise of the surgeon and the availability of necessary instrumentation. In our experience, laparoscopy with or without robotic assistance is the preferred surgical approach.3,4,6,11,28-32
Others have reported on the benefits of laparoscopy over laparotomy for the surgical management of genitourinary endometriosis. In a review of 61 patients who underwent either robot-assisted laparoscopic (n = 25) or open (n = 41) ureteroneocystostomy (n = 41), Isac and colleagues reported the procedure was longer in the laparoscopic group (279 min vs 200 min, P<.001), but the laparoscopic group had a shorter hospital stay (3 days vs 5 days, P<.001), used fewer narcotics postoperatively (P<.001), and had lower intraoperative blood loss (100 mL vs 150 mL, P<.001).32 No differences in long-term outcomes were observed in either cohort.
In a systematic review of 700 patients undergoing laparoscopic surgery for ureteral endometriosis, Cavaco-Gomes and colleagues reported that conversion to laparotomy occurred in only 3% to 7% of cases.10 In instances of ureteral endometriosis, laparoscopy provides greater visualization of the intraperitoneal contents over laparotomy, enabling better evaluation and treatment of lesions.3,29,33,34 Robot-assisted laparoscopy provides the additional advantages of 3D visualization, potential for an accelerated learning curve over traditional laparoscopy, improvement in dissection technique, and ease of suturing technique.6,35,36

Continue to: Extrinsic disease...
Extrinsic disease. In our group, we perform ureterolysis for extrinsic disease.25 The peritoneal incision is made in an area unaffected by endometriosis. Using the suction irrigator, a potential space is developed under the serosa of the ureter by injecting normal saline or lactated Ringer’s solution. By creating a fluid barrier between the serosa and underlying tissues, the depth of surgical incision and lateral thermal spread are minimized. Grasping forceps are used to peel the peritoneum away.25,37,38
Intrinsic disease. Unlike extrinsic disease, intrinsic disease can infiltrate the muscularis, lamina propria, and ureteral lumen, resulting in proximal dilation of the ureter with strictures.8 In this situation, ureteral compromise is likely and resection of the ureter is indicated3,28 (FIGURE 3). Intrinsic disease can be suggested by preoperative imaging or when there is evidence of deep infiltrating disease on physical exam, such as rectovaginal nodularity.30,39 When intrinsic ureteral disease is known, consultation with a urologist to plan a joint procedure is advisable. The procedure chosen to re-establish a functional ureter following resection depends on the location and extent of the involved ureter. Resection in close proximity to the bladder may be repaired by ureteroneocystostomy with or without psoas hitch,30,39,40 whereas resection of more proximal ureter may be repaired using Boari flap, ileal interposition, or autotransplantation. Lesions in the upper third or middle ureter may be repaired using ureterouretral anastomosis.6,7,30,41-43

Continue to: Bladder endometriosis...
Bladder endometriosis. Surgical treatment for bladder endometriosis depends on the depth of invasion and the location of the lesion (FIGURE 4). Lesions of the superficial aspect of the bladder identified at the time of laparoscopy can be treated with either excision or fulguration28,35,44 In our group, we perform excision over fulguration to remove the entire lesion and obtain a pathologic diagnosis. Deeper lesions involving the detrusor muscle are likely to be an endometrioma of the bladder. In these cases, laparoscopic excision is recommended.7 Rarely, lesions close to the interureteric ridge may require ureteroneocystostomy.19,45
In our experience, laparoscopic resection of bladder endometriomas is associated with better results in terms of symptom relief, progression of disease, and recurrence risk compared with other approaches. When performing laparoscopic excision of bladder lesions, we concomitantly evaluate the bladder lesion via cystoscopy to ensure adequate margins are obtained. Double-J stent placement is advised when lesions are within 2 cm of the urethral meatus to ensure ureteral patency during the postoperative period.45 A postoperative cystogram routinely is performed 7 to 14 days after surgery to ensure adequate repair prior to removing the urinary catheter.9,25,46,47

Postsurgical follow-up
Follow-up after treatment of genitourinary tract endometriosis should include monitoring the patient for symptoms of recurrence. Regular history and physical examination, renal function testing, and, in some instances, pelvic ultrasonography, all have a role in surveillance for recurrent ureteric disease. IVP or MRI may be warranted if a recurrence is suspected. A high index of suspicion should be maintained on the part of the clinician to avoid the devastating consequences of silent kidney loss. Patients should be counseled about the risk of disease recurrence, especially in those not undergoing postoperative hormonal suppression.
In conclusion
While endometriosis of the genitourinary tract is rare, patients can experience significant morbidity. Medical management of the disease is often limited by substantial adverse effects that limit treatment duration and is best used postoperatively after excision. An adequate physical exam and preoperative diagnostic imaging can be employed to characterize the extent of disease. When extensive disease involving the ureter is suspected, preoperative consultation with a urologist is encouraged to plan a multidisciplinary approach to surgical resection.
The ideal approach to surgery is laparoscopic resection with or without robotic assistance. Treatment of ureteral disease most commonly involves ureterolysis for cases of extrinsic disease but may require total resection of the ureter with concurrent ureteral reconstruction when disease is intrinsic to the ureter. Surgery for bladder endometriosis depends on the depth of invasion and location of the lesion. Superficial bladder lesions can be treated with fulguration or excision, while deeper lesions involving the detrusor muscle require excision. Lesions in close proximity to the interureteric ridge may require ureteroneocystostomy. Follow-up after excisional procedures involves monitoring the patient for signs and symptoms of disease recurrence, especially in cases of ureteral involvement, to avoid the devastating consequences of silent kidney loss.
The definitive cause of endometriosis remains unknown, but several prominent theories have been proposed.
Sampson's theory of retrograde menstruation through the fallopian tubes is the most well-known theory,1 although Schron had acknowledged a similar thought 3 centuries before.2 This theory posits that refluxed endometrial cells enter the abdominal cavity and invade the peritoneum, developing a blood supply necessary for survival and growth. Early reports supported this theory by suggesting that women with genital tract obstruction have a higher incidence of endometriosis.3,4 However, it was later confirmed that women without genital tract obstruction have a similar incidence of retrograde menstruation. In fact, up to 90% of women are found to have retrograde menstruation, but only 10% develop endometriosis. This suggests that once endometrial cells have escaped the uterine cavity, other events are necessary for endometrial cells to implant and survive.3,5 Other evidence to support the theory of retrograde menstruation is the observation that endometriosis is most commonly observed in the dependent portions of the pelvis, on the ovaries, in the anterior and posterior cul-de-sacs, and on the uterosacral ligament.6
The coelomic metaplasia theory holds that endometriosis results from spontaneous metaplastic change to mesothelial cells derived from the coelomic epithelium (located in the peritoneum and the pleura) upon exposure to menstrual effluent or other stimuli.7 Evidence for this theory is supported by the observation that intact endometrial cells have no access to the thoracic cavity in the absence of anatomical defect; therefore, the implantation theory cannot explain cases of pleural or pulmonary endometriosis.
Immune dysregulation also has been invoked to explain endometriosis implants both inside and outside the genitourinary tract. Studies have shown a higher incidence of endometriosis in women with other autoimmune disorders, including hypothyroidism, chronic fatigue syndrome, rheumatoid arthritis, systemic lupus erythematosus, Sjogren syndrome, and multiple sclerosis as well as in women with allergies, asthma, and eczema.8 In such women, dysregulation of the innate and adaptive immune system might promote the disease by inhibiting apoptosis of ectopic endometrial cells and by promoting their attachment, invasion, and proliferation into healthy peritoneum through the secretion of various growth factors and cytokines.9,10
Other possible theories that might explain the pathogenesis of endometriosis exist.11-13 While each theory has documented supporting evidence, no single theory currently accounts for all cases of endometriosis. It is likely, then, that endometriosis is a multifactorial disease with a combination of immune dysregulation, molecular abnormalities, genetic and epigenetic factors, and environmental exposures involved in its pathogenesis.
References
- Sampson J. Peritoneal endometriosis due to the menstrual dissemination of endometrial tissue into the peritoneal cavity. Am J Obstet Gynecol. 1927;14:422-469.
- Nezhat C, Nezhat F, Nezhat C. Endometriosis: ancient disease, ancient treatments. Fertil Steril. 2012;98(6 suppl):S1-62.
- Halme J, Hammond MG, Hulka JF, et al. Retrograde menstruation in healthy women and in patients with endometriosis. Obstet Gynecol. 1984;64:151-154.
- Sanfilippo JS, Wakim NG, Schikler KN, et al. Endometriosis in association with uterine anomaly. Am J Obstet Gynecol. 1986;154:39-43.
- Burney RO, Giudice LC. Pathogenesis and pathophysiology of endometriosis. Fertil Steril. 2012;98:511-519.
- Vercellini P, Chapron C, Fedele L, et al. Evidence for asymmetric distribution of lower intestinal tract endometriosis. BJOG. 2004;111:1213-1217.
- Sourial S, Tempest N, Hapangama DK. Theories on the pathogenesis of endometriosis. Int J Reprod Med. 2014;2014:179515.
- Sinaii N, Cleary SD, Ballweg ML, et al. High rates of autoimmune and endocrine disorders, fibromyalgia, chronic fatigue syndrome and atopic diseases among women with endometriosis: a survey analysis. Hum Reprod. 2002;17:2715-2724.
- Lebovic DI, Mueller MD, Taylor RN. Immunobiology of endometriosis. Fertil Steril. 2001;75:1-10.
- Sidell N, Han SW, Parthasarathy S. Regulation and modulation of abnormal immune responses in endometriosis. Ann N Y Acad Sci. 2002;955: 159-173; discussion 199-200, 396-406.
- Burney RO, Giudice LC. The pathogenesis of endometriosis. In: Nezhat C, Nezhat F, Nezhat C, eds. Nezhat's Video-Assisted and Robotic-Assisted Laparoscopy and Hysteroscopy. 4th ed. New York, NY: Cambridge University Press; 2013;252-258.
- Buka NJ. Vesical endometriosis after cesarean section. Am J Obstet Gynecol. 1988;158:1117-1118.
- Price DT, Maloney KE, Ibrahim GK, et al. Vesical endometriosis: report of two cases and review of the literature. Urology. 1996;48:639-643.
Endometriosis is a benign disease characterized by endometrial glands and stroma outside of the uterine cavity. It is commonly associated with pelvic pain and infertility. Ectopic endometrial tissue is predominantly located in the pelvis, but it can appear anywhere in the body, where it is referred to as extragenital endometriosis. The bowel and urinary tract are the most common sites of extragenital endometriosis.1
Laparoscopic management of extragenital endometriosis has been described since the 1980s.2 However, laparoscopic management of genitourinary endometriosis is still not widespread.3,4 Physicians are often unfamiliar with the signs and symptoms of genitourinary endometriosis and fail to consider it when a patient presents with bladder pain or hematuria, which may or may not be cyclic. Furthermore, many gynecologists do not have the experience to correctly identify the various forms of endometriosis that may appear on the pelvic organ, including the serosa and peritoneum, as variable colored spots, blebs, lesions, or adhesions. Many surgeons are also not adequately trained in the advanced laparoscopic techniques required to treat genitourinary endometriosis.4
In this article, we describe the clinical presentation and diagnosis of genitourinary endometriosis and discuss laparoscopic management strategies with and without robotic assistance.
Clinical presentation and diagnosis of genitourinary endometriosis
While ureteral and bladder endometriosis are both diseases of the urinary tract, they are not always found together in the same patient. The bladder is the most commonly affected organ, followed by the ureter and kidney.3,5,6 Endometriosis of the bladder usually presents with significant lower urinary tract symptoms. In contrast, ureteral endometriosis is usually silent with no apparent urinary symptoms.
Ureteral endometriosis. Cyclic hematuria is present in less than 15% of patients with ureteral endometriosis. Some patients experience cyclic, nonspecific colicky flank pain.7-9 Otherwise, most patients present with the usual symptoms of endometriosis, such as pelvic pain and dysmenorrhea. In a systematic review, Cavaco-Gomes and colleagues described 700 patients with ureteral endometriosis; 81% reported dysmenorrhea, 70% had pelvic pain, and 66% had dyspareunia.10 Rarely, ureteral endometriosis can result in silent kidney loss if the ureter becomes severely obstructed without treatment.11,12
Continue to: The lack of symptoms makes...
The lack of symptoms makes the early diagnosis of ureteral endometriosis difficult. As with all types of endometriosis, histologic evaluation of a biopsy sample is diagnostic. Multiple imaging modalities have been used to preoperatively diagnose ureteral involvement, including computed tomography,13 magnetic resonance imaging (MRI),14 intravenous pyelogram (IVP), and pelvic ultrasonography. However, each of these imaging modalities is limited in both sensitivity and the ability to characterize depth of tissue invasion.
In a study of 245 women undergoing pelvic ultrasonography, Pateman and colleagues reported that an experienced sonographer was able to visualize the bilateral ureters in 93% of cases.15 Renal ultrasonography is indicated in any woman suspected of having genitourinary tract involvement with the degree of hydroureter and level of obstruction noted during the exam.16
In our group, we perform renography to assess kidney function when hydroureter is noted preoperatively. Studies suggest that if greater than 10% of normal glomerular filtration rate remains, the kidney is considered salvageable, and near-normal function often returns following resection of disease. If preoperative kidney function is noted to be less than 10%, consultation with a nephrologist for possible nephrectomy is warranted.
We find that IVP is often helpful for preoperatively identifying the level and degree of ureteral involvement, and it also can be used postoperatively to evaluate for ureteral continuity (FIGURE 1). Sillou and colleagues showed MRI to be adequately sensitive for the detection of intrinsic ureteral endometriosis, but they reported that MRI often overestimates the frequency of disease.17 Authors of a 2016 Cochrane review of imaging modalities for endometriosis, including 4,807 women in 49 studies, reported that no imaging test was superior to surgery for diagnosing endometriosis.18 However, the review notably excluded genitourinary tract endometriosis, as surgery is not an acceptable reference standard, given that many surgeons cannot reliably identify such lesions.18

Bladder endometriosis. Unlike patients with ureteral endometriosis, those with bladder endometriosis are typically symptomatic and experience dysuria, hematuria, urinary frequency, and suprapubic tenderness.7,19 Urinary tract infection, interstitial cystitis, and cancer must be considered in the differential diagnosis. Urinalysis and urine culture should be performed, and other diagnostic procedures such as ultrasonography, MRI, and cystoscopy should be considered to evaluate for endometriosis of the bladder.
Ultrasound and MRI of the bladder both demonstrate a high specificity for detecting bladder endometriosis, but they lack sensitivity for lesions less than 3 cm.20 Deep infiltrating endometriosis of the bladder can be identified at the time of cystoscopy, which can assist in determining the need for ureteroneocystostomy if lesions are within 2 cm of the urethral opening.20 Cystoscopy also allows for biopsy to be performed if underlying malignancy is of concern.19
In our group, when bladder endometriosis is suspected, we routinely perform preoperative bladder ultrasonography to identify the lesion and plan to perform intraoperative cystoscopy at the time of laparoscopic resection.19,21
Continue to: Treatment...
Treatment
Medical management
Empiric medical therapies for endometriosis are centered around the idea that ectopic endometrial tissue responds to treatment in a similar manner as normal eutopic endometrium. The goals of treatment are to reduce or eliminate cyclic menstruation, thereby decreasing peritoneal seeding and suppressing the growth and activity of established ectopic implants. Medical therapy commonly involves the use of gonadotropin-releasing hormone agonists or antagonists, danazol, combined oral contraceptives, progestins, and aromatase inhibitors.
Medical therapy is commonly employed for patients with mild or early-stage disease and in those who are poor surgical candidates or decline surgery. Medical management alone clearly is contraindicated in the setting of ureteral obstruction and—in our opinion—may not be a good option for those with endometriosis of the ureter, given the potential for recurrence and potential serious sequelae of reduced renal function.22 Therefore, surgery has become the standard approach to therapy for mild to moderate cases of ureteral endometriosis.3
Medical therapy for patients with endometriosis of the bladder is generally considered a temporary solution as hormonal suppression, with its associated adverse effects, must be maintained throughout menopause. However, if endometriosis lesions lie within close proximity to the trigone, medical management is preferred, as surgical excision in the area of the trigone may predispose patients to neurogenic bladder and retrograde bladder reflux.23,24
Surgical management
The objectives of surgical treatment for genitourinary tract endometriosis are to excise all visible disease, to prevent or delay recurrence of the disease to the extent possible, and to avoid any further morbidity—in particular, to preserve renal function in cases of ureteral endometriosis—and to avoid iatrogenic injury to surrounding pelvic nervous structures25-27 (FIGURE 2). The surgical approach depends on the technical expertise of the surgeon and the availability of necessary instrumentation. In our experience, laparoscopy with or without robotic assistance is the preferred surgical approach.3,4,6,11,28-32
Others have reported on the benefits of laparoscopy over laparotomy for the surgical management of genitourinary endometriosis. In a review of 61 patients who underwent either robot-assisted laparoscopic (n = 25) or open (n = 41) ureteroneocystostomy (n = 41), Isac and colleagues reported the procedure was longer in the laparoscopic group (279 min vs 200 min, P<.001), but the laparoscopic group had a shorter hospital stay (3 days vs 5 days, P<.001), used fewer narcotics postoperatively (P<.001), and had lower intraoperative blood loss (100 mL vs 150 mL, P<.001).32 No differences in long-term outcomes were observed in either cohort.
In a systematic review of 700 patients undergoing laparoscopic surgery for ureteral endometriosis, Cavaco-Gomes and colleagues reported that conversion to laparotomy occurred in only 3% to 7% of cases.10 In instances of ureteral endometriosis, laparoscopy provides greater visualization of the intraperitoneal contents over laparotomy, enabling better evaluation and treatment of lesions.3,29,33,34 Robot-assisted laparoscopy provides the additional advantages of 3D visualization, potential for an accelerated learning curve over traditional laparoscopy, improvement in dissection technique, and ease of suturing technique.6,35,36

Continue to: Extrinsic disease...
Extrinsic disease. In our group, we perform ureterolysis for extrinsic disease.25 The peritoneal incision is made in an area unaffected by endometriosis. Using the suction irrigator, a potential space is developed under the serosa of the ureter by injecting normal saline or lactated Ringer’s solution. By creating a fluid barrier between the serosa and underlying tissues, the depth of surgical incision and lateral thermal spread are minimized. Grasping forceps are used to peel the peritoneum away.25,37,38
Intrinsic disease. Unlike extrinsic disease, intrinsic disease can infiltrate the muscularis, lamina propria, and ureteral lumen, resulting in proximal dilation of the ureter with strictures.8 In this situation, ureteral compromise is likely and resection of the ureter is indicated3,28 (FIGURE 3). Intrinsic disease can be suggested by preoperative imaging or when there is evidence of deep infiltrating disease on physical exam, such as rectovaginal nodularity.30,39 When intrinsic ureteral disease is known, consultation with a urologist to plan a joint procedure is advisable. The procedure chosen to re-establish a functional ureter following resection depends on the location and extent of the involved ureter. Resection in close proximity to the bladder may be repaired by ureteroneocystostomy with or without psoas hitch,30,39,40 whereas resection of more proximal ureter may be repaired using Boari flap, ileal interposition, or autotransplantation. Lesions in the upper third or middle ureter may be repaired using ureterouretral anastomosis.6,7,30,41-43

Continue to: Bladder endometriosis...
Bladder endometriosis. Surgical treatment for bladder endometriosis depends on the depth of invasion and the location of the lesion (FIGURE 4). Lesions of the superficial aspect of the bladder identified at the time of laparoscopy can be treated with either excision or fulguration28,35,44 In our group, we perform excision over fulguration to remove the entire lesion and obtain a pathologic diagnosis. Deeper lesions involving the detrusor muscle are likely to be an endometrioma of the bladder. In these cases, laparoscopic excision is recommended.7 Rarely, lesions close to the interureteric ridge may require ureteroneocystostomy.19,45
In our experience, laparoscopic resection of bladder endometriomas is associated with better results in terms of symptom relief, progression of disease, and recurrence risk compared with other approaches. When performing laparoscopic excision of bladder lesions, we concomitantly evaluate the bladder lesion via cystoscopy to ensure adequate margins are obtained. Double-J stent placement is advised when lesions are within 2 cm of the urethral meatus to ensure ureteral patency during the postoperative period.45 A postoperative cystogram routinely is performed 7 to 14 days after surgery to ensure adequate repair prior to removing the urinary catheter.9,25,46,47

Postsurgical follow-up
Follow-up after treatment of genitourinary tract endometriosis should include monitoring the patient for symptoms of recurrence. Regular history and physical examination, renal function testing, and, in some instances, pelvic ultrasonography, all have a role in surveillance for recurrent ureteric disease. IVP or MRI may be warranted if a recurrence is suspected. A high index of suspicion should be maintained on the part of the clinician to avoid the devastating consequences of silent kidney loss. Patients should be counseled about the risk of disease recurrence, especially in those not undergoing postoperative hormonal suppression.
In conclusion
While endometriosis of the genitourinary tract is rare, patients can experience significant morbidity. Medical management of the disease is often limited by substantial adverse effects that limit treatment duration and is best used postoperatively after excision. An adequate physical exam and preoperative diagnostic imaging can be employed to characterize the extent of disease. When extensive disease involving the ureter is suspected, preoperative consultation with a urologist is encouraged to plan a multidisciplinary approach to surgical resection.
The ideal approach to surgery is laparoscopic resection with or without robotic assistance. Treatment of ureteral disease most commonly involves ureterolysis for cases of extrinsic disease but may require total resection of the ureter with concurrent ureteral reconstruction when disease is intrinsic to the ureter. Surgery for bladder endometriosis depends on the depth of invasion and location of the lesion. Superficial bladder lesions can be treated with fulguration or excision, while deeper lesions involving the detrusor muscle require excision. Lesions in close proximity to the interureteric ridge may require ureteroneocystostomy. Follow-up after excisional procedures involves monitoring the patient for signs and symptoms of disease recurrence, especially in cases of ureteral involvement, to avoid the devastating consequences of silent kidney loss.
The definitive cause of endometriosis remains unknown, but several prominent theories have been proposed.
Sampson's theory of retrograde menstruation through the fallopian tubes is the most well-known theory,1 although Schron had acknowledged a similar thought 3 centuries before.2 This theory posits that refluxed endometrial cells enter the abdominal cavity and invade the peritoneum, developing a blood supply necessary for survival and growth. Early reports supported this theory by suggesting that women with genital tract obstruction have a higher incidence of endometriosis.3,4 However, it was later confirmed that women without genital tract obstruction have a similar incidence of retrograde menstruation. In fact, up to 90% of women are found to have retrograde menstruation, but only 10% develop endometriosis. This suggests that once endometrial cells have escaped the uterine cavity, other events are necessary for endometrial cells to implant and survive.3,5 Other evidence to support the theory of retrograde menstruation is the observation that endometriosis is most commonly observed in the dependent portions of the pelvis, on the ovaries, in the anterior and posterior cul-de-sacs, and on the uterosacral ligament.6
The coelomic metaplasia theory holds that endometriosis results from spontaneous metaplastic change to mesothelial cells derived from the coelomic epithelium (located in the peritoneum and the pleura) upon exposure to menstrual effluent or other stimuli.7 Evidence for this theory is supported by the observation that intact endometrial cells have no access to the thoracic cavity in the absence of anatomical defect; therefore, the implantation theory cannot explain cases of pleural or pulmonary endometriosis.
Immune dysregulation also has been invoked to explain endometriosis implants both inside and outside the genitourinary tract. Studies have shown a higher incidence of endometriosis in women with other autoimmune disorders, including hypothyroidism, chronic fatigue syndrome, rheumatoid arthritis, systemic lupus erythematosus, Sjogren syndrome, and multiple sclerosis as well as in women with allergies, asthma, and eczema.8 In such women, dysregulation of the innate and adaptive immune system might promote the disease by inhibiting apoptosis of ectopic endometrial cells and by promoting their attachment, invasion, and proliferation into healthy peritoneum through the secretion of various growth factors and cytokines.9,10
Other possible theories that might explain the pathogenesis of endometriosis exist.11-13 While each theory has documented supporting evidence, no single theory currently accounts for all cases of endometriosis. It is likely, then, that endometriosis is a multifactorial disease with a combination of immune dysregulation, molecular abnormalities, genetic and epigenetic factors, and environmental exposures involved in its pathogenesis.
References
- Sampson J. Peritoneal endometriosis due to the menstrual dissemination of endometrial tissue into the peritoneal cavity. Am J Obstet Gynecol. 1927;14:422-469.
- Nezhat C, Nezhat F, Nezhat C. Endometriosis: ancient disease, ancient treatments. Fertil Steril. 2012;98(6 suppl):S1-62.
- Halme J, Hammond MG, Hulka JF, et al. Retrograde menstruation in healthy women and in patients with endometriosis. Obstet Gynecol. 1984;64:151-154.
- Sanfilippo JS, Wakim NG, Schikler KN, et al. Endometriosis in association with uterine anomaly. Am J Obstet Gynecol. 1986;154:39-43.
- Burney RO, Giudice LC. Pathogenesis and pathophysiology of endometriosis. Fertil Steril. 2012;98:511-519.
- Vercellini P, Chapron C, Fedele L, et al. Evidence for asymmetric distribution of lower intestinal tract endometriosis. BJOG. 2004;111:1213-1217.
- Sourial S, Tempest N, Hapangama DK. Theories on the pathogenesis of endometriosis. Int J Reprod Med. 2014;2014:179515.
- Sinaii N, Cleary SD, Ballweg ML, et al. High rates of autoimmune and endocrine disorders, fibromyalgia, chronic fatigue syndrome and atopic diseases among women with endometriosis: a survey analysis. Hum Reprod. 2002;17:2715-2724.
- Lebovic DI, Mueller MD, Taylor RN. Immunobiology of endometriosis. Fertil Steril. 2001;75:1-10.
- Sidell N, Han SW, Parthasarathy S. Regulation and modulation of abnormal immune responses in endometriosis. Ann N Y Acad Sci. 2002;955: 159-173; discussion 199-200, 396-406.
- Burney RO, Giudice LC. The pathogenesis of endometriosis. In: Nezhat C, Nezhat F, Nezhat C, eds. Nezhat's Video-Assisted and Robotic-Assisted Laparoscopy and Hysteroscopy. 4th ed. New York, NY: Cambridge University Press; 2013;252-258.
- Buka NJ. Vesical endometriosis after cesarean section. Am J Obstet Gynecol. 1988;158:1117-1118.
- Price DT, Maloney KE, Ibrahim GK, et al. Vesical endometriosis: report of two cases and review of the literature. Urology. 1996;48:639-643.
- Veeraswamy A, Lewis M, Mann A, et al. Extragenital endometriosis. Clin Obstet Gynecol. 2010;53:449-466.
- Nezhat C, Crowgey SR, Garrison GP. Surgical treatment of endometriosis via laser laparoscopy. Fertil Steril. 1986;45:778-783.
- Bosev D, Nicoll LM, Bhagan L, et al. Laparoscopic management of ureteral endometriosis: the Stanford University hospital experience with 96 consecutive cases. J Urol. 2009;182:2748-2752.
- Nezhat C, Falik R, McKinney S, et al. Pathophysiology and management of urinary tract endometriosis. Nat Rev Urol. 2017;14:359-372.
- Shook TE, Nyberg LM. Endometriosis of the urinary tract. Urology. 1988;31:1-6.
- Nezhat C, Modest AM, King LP. The role of the robot in treating urinary tract endometriosis. Curr Opin Obstet Gynecol. 2013;25:308-311.
- Comiter CV. Endometriosis of the urinary tract. Urol Clin North Am. 2002;29:625-635.
- Gustilo-Ashby AM, Paraiso MF. Treatment of urinary tract endometriosis. J Minim Invasive Gynecol. 2006;13:559-565.
- Berlanda N, Somigliana E, Frattaruolo MP, et al. Surgery versus hormonal therapy for deep endometriosis: is it a choice of the physician? Eur J Obstet Gyneocol Reprod Biol. 2017;209:67-71.
- Cavaco-Gomes J, Martinho M, Gilabert-Aguilar J, et al. Laparoscopic management of ureteral endometriosis: a systematic review. Eur J Obstet Gynecol Reprod Biol. 2017;210:94-101.
- Nezhat C, Nezhat F, Green B. Laparoscopic treatment of obstructed ureter due to endometriosis by resection and ureteroureterostomy: a case report. J Urol. 1992;148:865-868.
- Nezhat C, Paka C, Gomaa M, et al. Silent loss of kidney secondary to ureteral endometriosis. JSLS. 2012;16:451-455.
- Iosca S, Lumia D, Bracchi E, et al. Multislice computed tomography with colon water distention (MSCT-c) in the study of intestinal and ureteral endometriosis. Clin Imaging. 2013;37(6):1061-1068.
- Medeiros LR, Rosa MI, Silva BR, et al. Accuracy of magnetic resonance in deeply infiltrating endometriosis: a systematic review and meta-analysis. Arch Gynecol Obstet. 2015;291:611-621.
- Pateman K, Mavrelos D, Hoo WL, et al. Visualization of ureters on standard gynecological transvaginal scan: a feasibility study. Ultrasound Obstet Gynecol. 2013;41:696-701.
- Guerriero S, Condous G, van den Bosch T, et al. Systematic approach to sonographic evaluation of the pelvis in women with suspected endometriosis, including terms, definitions and measurements: a consensus opinion from the International Deep Endometriosis Analysis (IDEA) group. Ultrasound Obstet Gynecol. 2016;48:318-332.
- Sillou S, Poirée S, Millischer AE, et al. Urinary endometriosis: MR imaging appearance with surgical and histological correlations. Diagn Interv Imaging. 2015;96:373-381.
- Nisenblat V, Bossuyt PM, Farquhar C, et al. Imaging modalities for the non-invasive diagnosis of endometriosis. Cochrane Database Syst Rev. 2016;2:CD009591.
- Nezhat CH, Malik S, Osias J, et al. Laparoscopic management of 15 patients with infiltrating endometriosis of the bladder and a case of primary intravesical endometrioid adenosarcoma. Fertil Steril. 2002;78:872-875.
- Kolodziej A, Krajewski W, Dolowy L, et al. Urinary tract endometriosis. Urol J. 2015;12:2213-2217.
- Nezhat C, Buescher E, Paka C, et al. Video-assisted laparoscopic treatment of endometriosis. In: Nezhat C, Nezhat F, Nezhat C, eds. Nezhat's Video-Assisted and Robotic-Assisted Laparoscopy and Hysteroscopy. 4th ed. New York, NY: Cambridge University Press; 2013;265.
- Al-Fozan H, Tulandi T. Left lateral predisposition of endometriosis and endometrioma. Obstet Gynecol. 2003;101:164-166.
- Hastings JC, Van Winkle W, Barker E, et al. The effect of suture materials on healing wounds of the bladder. Surg Gynecol Obstet. 1975;140:933-937.
- Cornell KK. Cystotomy, partial cystectomy, and tube cystostomy. Clin Tech Small Anim Pract. 2000;15:11-16.
- Nezhat C, Nezhat F, Nezhat C, eds. Nezhat's Video-Assisted and Robotic-Assisted Laparoscopy and Hysteroscopy. 4th ed. New York, NY: Cambridge University Press; 2013.
- Uccella S, Cromi A, Casarin J, et al. Laparoscopy for ureteral endometriosis: surgical details, long-term follow-up, and fertility outcomes. Fertil Steril. 2014;102:160-166.e2.
- Knabben L, Imboden S, Fellmann B, et al. Urinary tract endometriosis in patients with deep infiltrating endometriosis: prevalence, symptoms, management, and proposal for a new clinical classification. Fertil Steril. 2015;103:147-152.
- Nezhat C, Nezhat F, Nezhat CH, et al. Urinary tract endometriosis treated by laparoscopy. Fertil Steril. 1996;66:920-924.
- Nezhat CH, Nezhat F, Seidman D, et al. Laparoscopic ureteroureterostomy: a prospective follow-up of 9 patients. Prim Care Update Ob Gyns. 1998;5:200.
- Nezhat CH, Bracale U, Scala A, et al. Laparoscopic ureteroneocystostomy and vesicopsoas hitch for infiltrative endometriosis. JSLS. 2004;8:3-7.
- Nezhat C, Lewis M, Kotikela S, et al. Robotic versus standard laparoscopy for the treatment of endometriosis. Fertil Steril. 2010;94:2758-2760.
- Isac W, Kaouk J, Altunrende F, et al. Robotic-assisted ureteroneocytostomy: techniques and comparative outcomes. J Endourol. 2013;27:318-323.
- Nezhat C, Nezhat F. Laparoscopic repair of ureter resected during operative laparoscopy. Obstet Gynecol. 1992;80(3 pt 2):543-544.
- De Cicco C, Ussia A, Koninckx PR. Laparoscopic ureteral repair in gynaecological surgery. Curr Opin Obstet Gynecol. 2011;23:296-300.
- Nezhat C, Hajhosseini B, King LP. Robotic-assisted laparoscopic treatment of bowel, bladder, and ureteral endometriosis. JSLS. 2011;15:387-392.
- Fadhlaoui A, Gillon T, Lebbi I, et al. Endometriosis and vesico-sphincteral disorders. Front Surg. 2015;2:23.
- Nezhat C, Nezhat FR. Safe laser endoscopic excision or vaporization of peritoneal endometriosis. Fertil Steril. 1989;52:149-151.
- Nezhat C, Winer W, Nezhat FA. Comparison of the CO2, argon, and KTP/532 lasers in the videolaseroscopic treatment of endometriosis. J Gynecol Surg. 2009;41-47.
- Azioni G, Bracale U, Scala A, et al. Laparoscopic ureteroneocytostomy and vesicopsoas hitch for infiltrative ureteral endometriosis. Minim Invasive Ther Allied Technol. 2010;19:292-297.
- Stepniewska A, Grosso G, Molon A, et al. Ureteral endometriosis: clinical and radiological follow-up after laparoscopic ureterocystoneostomy. Hum Reprod. 2011;26:112-116.
- Nezhat CH, Nezhat FR, Freiha F, et al. Laparoscopic vesicopsoas hitch for infiltrative ureteral endometriosis. Fertil Steril. 1999;71:376-379.
- Scioscia M, Molon A, Grosso G, et al. Laparoscopic management of ureteral endometriosis. Curr Opin Obstet Gynecol. 2009;21:325-328.
- Antonelli A. Urinary tract endometriosis. Urologia. 2012;79:167-170.
- Camanni M, Bonino L, Delpiano EM, et al. Laparoscopic conservative management of ureteral endometriosis: a survey of eighty patients submitted to ureterolysis. Reprod Biol Endocrinol. 2009;7:109.
- Chapron C, Bourret A, Chopin N, et al. Surgery for bladder endometriosis: long-term results and concomitant management of associated posterior deep lesions. Hum Reprod. 2010;25:884-889.
- Nezhat CR, Nezhat FR. Laparoscopic segmental bladder resection for endometriosis: a report of two cases. Obstet Gynecol. 1993;81(5 pt 2):882-884.
- Bourdel N, Cognet S, Canis M, et al. Laparoscopic ureteroneocystostomy: be prepared! J Minim Invasive Gynecol. 2015;22:827-833.
- Page B. Camran Nezhat and the Advent of Advanced Operative Video-laparoscopy. In: Nezhat C, ed. Nezhat's History of Endoscopy. Tuttlingen, Germany: Endo Press; 2011:159-187.
- Podratz K. Degrees of Freedom: Advances in Gynecological and Obstetrical Surgery. Remembering Milestones and Achievements in Surgery: Inspiring Quality for a Hundred Years 1913-2012. Published by American College of Surgeons 2012. Tampa, FL: Faircount Media Group; 2013.
- Kelley WE. The evolution of laparoscopy and the revolution in surgery in the decade of the 1990s. JSLS: J Soc Laparoendoscopic Surgeons. 2008;12:351-357.
- Veeraswamy A, Lewis M, Mann A, et al. Extragenital endometriosis. Clin Obstet Gynecol. 2010;53:449-466.
- Nezhat C, Crowgey SR, Garrison GP. Surgical treatment of endometriosis via laser laparoscopy. Fertil Steril. 1986;45:778-783.
- Bosev D, Nicoll LM, Bhagan L, et al. Laparoscopic management of ureteral endometriosis: the Stanford University hospital experience with 96 consecutive cases. J Urol. 2009;182:2748-2752.
- Nezhat C, Falik R, McKinney S, et al. Pathophysiology and management of urinary tract endometriosis. Nat Rev Urol. 2017;14:359-372.
- Shook TE, Nyberg LM. Endometriosis of the urinary tract. Urology. 1988;31:1-6.
- Nezhat C, Modest AM, King LP. The role of the robot in treating urinary tract endometriosis. Curr Opin Obstet Gynecol. 2013;25:308-311.
- Comiter CV. Endometriosis of the urinary tract. Urol Clin North Am. 2002;29:625-635.
- Gustilo-Ashby AM, Paraiso MF. Treatment of urinary tract endometriosis. J Minim Invasive Gynecol. 2006;13:559-565.
- Berlanda N, Somigliana E, Frattaruolo MP, et al. Surgery versus hormonal therapy for deep endometriosis: is it a choice of the physician? Eur J Obstet Gyneocol Reprod Biol. 2017;209:67-71.
- Cavaco-Gomes J, Martinho M, Gilabert-Aguilar J, et al. Laparoscopic management of ureteral endometriosis: a systematic review. Eur J Obstet Gynecol Reprod Biol. 2017;210:94-101.
- Nezhat C, Nezhat F, Green B. Laparoscopic treatment of obstructed ureter due to endometriosis by resection and ureteroureterostomy: a case report. J Urol. 1992;148:865-868.
- Nezhat C, Paka C, Gomaa M, et al. Silent loss of kidney secondary to ureteral endometriosis. JSLS. 2012;16:451-455.
- Iosca S, Lumia D, Bracchi E, et al. Multislice computed tomography with colon water distention (MSCT-c) in the study of intestinal and ureteral endometriosis. Clin Imaging. 2013;37(6):1061-1068.
- Medeiros LR, Rosa MI, Silva BR, et al. Accuracy of magnetic resonance in deeply infiltrating endometriosis: a systematic review and meta-analysis. Arch Gynecol Obstet. 2015;291:611-621.
- Pateman K, Mavrelos D, Hoo WL, et al. Visualization of ureters on standard gynecological transvaginal scan: a feasibility study. Ultrasound Obstet Gynecol. 2013;41:696-701.
- Guerriero S, Condous G, van den Bosch T, et al. Systematic approach to sonographic evaluation of the pelvis in women with suspected endometriosis, including terms, definitions and measurements: a consensus opinion from the International Deep Endometriosis Analysis (IDEA) group. Ultrasound Obstet Gynecol. 2016;48:318-332.
- Sillou S, Poirée S, Millischer AE, et al. Urinary endometriosis: MR imaging appearance with surgical and histological correlations. Diagn Interv Imaging. 2015;96:373-381.
- Nisenblat V, Bossuyt PM, Farquhar C, et al. Imaging modalities for the non-invasive diagnosis of endometriosis. Cochrane Database Syst Rev. 2016;2:CD009591.
- Nezhat CH, Malik S, Osias J, et al. Laparoscopic management of 15 patients with infiltrating endometriosis of the bladder and a case of primary intravesical endometrioid adenosarcoma. Fertil Steril. 2002;78:872-875.
- Kolodziej A, Krajewski W, Dolowy L, et al. Urinary tract endometriosis. Urol J. 2015;12:2213-2217.
- Nezhat C, Buescher E, Paka C, et al. Video-assisted laparoscopic treatment of endometriosis. In: Nezhat C, Nezhat F, Nezhat C, eds. Nezhat's Video-Assisted and Robotic-Assisted Laparoscopy and Hysteroscopy. 4th ed. New York, NY: Cambridge University Press; 2013;265.
- Al-Fozan H, Tulandi T. Left lateral predisposition of endometriosis and endometrioma. Obstet Gynecol. 2003;101:164-166.
- Hastings JC, Van Winkle W, Barker E, et al. The effect of suture materials on healing wounds of the bladder. Surg Gynecol Obstet. 1975;140:933-937.
- Cornell KK. Cystotomy, partial cystectomy, and tube cystostomy. Clin Tech Small Anim Pract. 2000;15:11-16.
- Nezhat C, Nezhat F, Nezhat C, eds. Nezhat's Video-Assisted and Robotic-Assisted Laparoscopy and Hysteroscopy. 4th ed. New York, NY: Cambridge University Press; 2013.
- Uccella S, Cromi A, Casarin J, et al. Laparoscopy for ureteral endometriosis: surgical details, long-term follow-up, and fertility outcomes. Fertil Steril. 2014;102:160-166.e2.
- Knabben L, Imboden S, Fellmann B, et al. Urinary tract endometriosis in patients with deep infiltrating endometriosis: prevalence, symptoms, management, and proposal for a new clinical classification. Fertil Steril. 2015;103:147-152.
- Nezhat C, Nezhat F, Nezhat CH, et al. Urinary tract endometriosis treated by laparoscopy. Fertil Steril. 1996;66:920-924.
- Nezhat CH, Nezhat F, Seidman D, et al. Laparoscopic ureteroureterostomy: a prospective follow-up of 9 patients. Prim Care Update Ob Gyns. 1998;5:200.
- Nezhat CH, Bracale U, Scala A, et al. Laparoscopic ureteroneocystostomy and vesicopsoas hitch for infiltrative endometriosis. JSLS. 2004;8:3-7.
- Nezhat C, Lewis M, Kotikela S, et al. Robotic versus standard laparoscopy for the treatment of endometriosis. Fertil Steril. 2010;94:2758-2760.
- Isac W, Kaouk J, Altunrende F, et al. Robotic-assisted ureteroneocytostomy: techniques and comparative outcomes. J Endourol. 2013;27:318-323.
- Nezhat C, Nezhat F. Laparoscopic repair of ureter resected during operative laparoscopy. Obstet Gynecol. 1992;80(3 pt 2):543-544.
- De Cicco C, Ussia A, Koninckx PR. Laparoscopic ureteral repair in gynaecological surgery. Curr Opin Obstet Gynecol. 2011;23:296-300.
- Nezhat C, Hajhosseini B, King LP. Robotic-assisted laparoscopic treatment of bowel, bladder, and ureteral endometriosis. JSLS. 2011;15:387-392.
- Fadhlaoui A, Gillon T, Lebbi I, et al. Endometriosis and vesico-sphincteral disorders. Front Surg. 2015;2:23.
- Nezhat C, Nezhat FR. Safe laser endoscopic excision or vaporization of peritoneal endometriosis. Fertil Steril. 1989;52:149-151.
- Nezhat C, Winer W, Nezhat FA. Comparison of the CO2, argon, and KTP/532 lasers in the videolaseroscopic treatment of endometriosis. J Gynecol Surg. 2009;41-47.
- Azioni G, Bracale U, Scala A, et al. Laparoscopic ureteroneocytostomy and vesicopsoas hitch for infiltrative ureteral endometriosis. Minim Invasive Ther Allied Technol. 2010;19:292-297.
- Stepniewska A, Grosso G, Molon A, et al. Ureteral endometriosis: clinical and radiological follow-up after laparoscopic ureterocystoneostomy. Hum Reprod. 2011;26:112-116.
- Nezhat CH, Nezhat FR, Freiha F, et al. Laparoscopic vesicopsoas hitch for infiltrative ureteral endometriosis. Fertil Steril. 1999;71:376-379.
- Scioscia M, Molon A, Grosso G, et al. Laparoscopic management of ureteral endometriosis. Curr Opin Obstet Gynecol. 2009;21:325-328.
- Antonelli A. Urinary tract endometriosis. Urologia. 2012;79:167-170.
- Camanni M, Bonino L, Delpiano EM, et al. Laparoscopic conservative management of ureteral endometriosis: a survey of eighty patients submitted to ureterolysis. Reprod Biol Endocrinol. 2009;7:109.
- Chapron C, Bourret A, Chopin N, et al. Surgery for bladder endometriosis: long-term results and concomitant management of associated posterior deep lesions. Hum Reprod. 2010;25:884-889.
- Nezhat CR, Nezhat FR. Laparoscopic segmental bladder resection for endometriosis: a report of two cases. Obstet Gynecol. 1993;81(5 pt 2):882-884.
- Bourdel N, Cognet S, Canis M, et al. Laparoscopic ureteroneocystostomy: be prepared! J Minim Invasive Gynecol. 2015;22:827-833.
- Page B. Camran Nezhat and the Advent of Advanced Operative Video-laparoscopy. In: Nezhat C, ed. Nezhat's History of Endoscopy. Tuttlingen, Germany: Endo Press; 2011:159-187.
- Podratz K. Degrees of Freedom: Advances in Gynecological and Obstetrical Surgery. Remembering Milestones and Achievements in Surgery: Inspiring Quality for a Hundred Years 1913-2012. Published by American College of Surgeons 2012. Tampa, FL: Faircount Media Group; 2013.
- Kelley WE. The evolution of laparoscopy and the revolution in surgery in the decade of the 1990s. JSLS: J Soc Laparoendoscopic Surgeons. 2008;12:351-357.
Endometriomas: Classification and surgical management
Related article:
Endometriosis: Expert answers to 7 crucial questions on diagnosis
Etiology
Endometriomas are extensively described in the literature, and their origin is the subject of several theories. In 1921, Sampson noted luteal membrane and ovarian epithelial tissues within endometriomas and was the first to indicate that endometriomas may result from the invasion of functional cysts by endometrial tissue.2,4,5 In 1979, Czernobilsky and Morris6 found endometrial and oviduct-like epithelium in ovarian endometriosis and concluded that ovarian tissue may be a common histologic precursor. Several other authors subsequently have reported finding different types of tissue within ovarian endometriomas, and not all of these chocolate cysts showed histologic evidence of endometriosis.4,7,8
Read about the classification of endometriomas
Disease classification
Our classification system identifies 2 types of endometriomas on the basis of their etiologies and characteristics. Type I, which arise from endometrial tissue implanted on the ovarian surface, are also called true endometriomas. Invagination of cortex and subsequent hemorrhage from endometrial tissue result in cyst formation. Endometrial tissue (endometrial stroma and glands) is histologically present in all type I endometriomas.1,4,9 These endometriomas usually are small (<5 cm in diameter) and have a densely adherent fibrous capsule.4 Often, there is no clear plane between cyst wall and ovarian stroma.3
Type II endometriomas arise from functional cysts involved in or invaded by cortical or pelvic side-wall endometrial implants or by type I endometriomas. Type II endometriomas are subclassified by the extent of endometrial implant involvement in the cyst wall. Type IIA endometriomas are hemorrhagic cysts with less than 10% of endometrial tissue within the cyst wall. Similar to the functional cysts from which they originate, type IIA endometriomas have a cyst wall that is separated easily from ovarian tissue during surgery.4,7,9 Although type II endometriomas tend to be larger than their type I counterparts, in some cases they are identified at an early stage of 2 to 5 cm. Endometriomas larger than 5 cm are almost always type II.4
Type IIB and IIC endometriomas have endometrial implants and fibrosis within their cyst walls, with progressively more endometrial invasion in type IIC endometriomas (>50%) than in type IIB (10% to 50%). Consequently, type IIB cysts are relatively easy to dissect from ovarian tissue, except adjacent to an endometriotic area where the cyst densely adheres to the ovarian stroma. In type IIC, endometrial tissue more extensively penetrates the capsule, making dissection of diseased tissue from the ovarian stroma more difficult; in fact, separating type IIC cyst wall from ovarian stroma can be as challenging as excising a type I endometrioma.7 In most cases, a type IIC cyst is attached by adhesions and fibrosis to the pelvic side wall or uterus and ruptures during mobilization (TABLE).

Related article:
Imaging the endometrioma and mature cystic teratoma
Presentation and diagnosis
Almost all patients with an endometrioma concurrently have peritoneal endometriosis, which is characterized by dysmenorrhea, dyspareunia, chronic pelvic pain, infertility, and, in some cases, gastrointestinal or genitourinary dysfunction.1 Pelvic examination may reveal an adnexal mass that is an endometrioma, or an endometrioma may appear on imaging obtained in a pelvic pain or infertility work-up. Given its 73% sensitivity, 94% specificity, safety, and low cost, transvaginal ultrasonography is the preferred imaging modality for endometrioma.3 The characteristic ultrasonographic appearance is that of a round, homogeneous, fluid-filled mass with low-level echoes.1 Magnetic resonance imaging is appropriate when a more sensitive imaging modality is indicated, as for a patient with risk factors for malignancy.3,10–12
Read about the surgical management of endometriomas
Surgical management
Indications for surgical excision of endometriomas include pelvic pain, infertility, and prevention and diagnosis of malignancy. Endometriomas may be excised prior to use of assisted reproductive technology.13–15 Medical therapy, such as oral contraceptives, can be used to reduce the size of endometriomas but does not improve fertility.3 Certain ovarian cancers are more common in women with endometriosis, and ovarian tumors are thought to develop in about 1% of ovarian endometriosis cases.1,12 Therefore, endometrioma excision may reduce the risk of malignancy. As with other ovarian cysts, large endometriomas may be excised to reduce the risks of rupture and torsion.
Approach
Laparoscopy is the preferred approach for endometrioma excision. Controversy exists regarding the ideal procedure: complete excision (with stripping of the cyst capsule) or drainage and ablation of the cyst wall. Compared with drainage and ablation, excision reduces recurrence of endometriomas; relieves dysmenorrhea, dyspareunia, pelvic pain, and other symptoms; and improves fertility.13,16 The recurrence rate may be as low as 5.8% with complete excision but is 90% with simple transvaginal aspiration.17,18 If not performed properly, however, cyst capsule stripping may damage nearby ovarian stroma and decrease the ovarian reserve.14 Some authors have advocated combining excision and ablation—performing cystectomy until there is no longer a clear plane between capsule and ovarian stroma and then ablating any remaining endometrial tissue.8
With type I and IIC endometriomas, we have seen the endometrial cyst wall infiltrating the ovarian stroma so deeply there is not always a definable plane. By contrast, type IIA and IIB endometriomas typically have a plane between the cyst wall and the ovarian cortex. In type II endometriomas, endometrial implants on the ovarian cortex infiltrate the plane of the cyst wall such that the juxtaposing lipomatous follicular cyst detaches with minimal intraoperative traction. Portions of type II endometriomas containing fibrosis and adhesions may become more difficult to peel off the cyst wall. For most endometriomas, at least 1 spot is difficult to peel off the ovary, and extra care must be taken at the hilum of ovary to avoid excising healthy ovarian cortex.4,5,7,8
Our surgical approach accounts for the described variations in type I and II endometriomas. Endometrial contents often spill as the endometrioma is dissected off neighboring structures. When possible, endometriomas should be aspirated and irrigated prior to cystectomy to avoid seeding the pelvis and abdomen with spilled endometriotic contents. We use hydrodissection, the injection of dilute vasopressin with a laparoscopic needle, to create a plane between cyst wall and ovarian stroma and strip the cyst capsule with laparoscopic graspers. Type I endometriomas adhere densely to the ovary. Given the presence of fibrosis and adhesions, the cyst is excised in a piecemeal fashion. Care is taken to remove any endometrial implants from the ovary while preserving as much of the ovarian tissue as possible.1
Type II endometriomas are larger cysts originating from the invasion of endometrial implants or type I endometrioma into functional cysts. The difficulty of capsule excision varies according to the extent of endometrial invasion. Type IIA endometriomas contain less than 10% endometrial tissue within the cyst capsule. Thus, the standard ovarian cystectomy stripping technique is successful in removing more than 90% of the cyst capsule. Special care is taken in stripping the residual small portion that involves the endometrial glands and stroma and adheres densely to the ovary.
The larger proportion of endometrial tissue present in type IIB and IIC endometriomas degrades the plane between the cyst capsule and the ovarian stroma, making excision more difficult. Similar to the type I excision, a piecemeal approach is often necessary. If complete stripping of the cyst capsule would result in extensive loss of healthy ovarian tissue, then electrocautery, plasma energy, or laser ablation can be selectively used to destroy focal areas of endometrial invasion. Complete ablation may be difficult, as the endometrioma wall can be up to 5 mm thick.19 For these thick-walled endometriomas, we recommend excision (vs ablation), which lowers the risk of endometrioma recurrence.
Related article:
Endometriosis and pain: Expert answers to 6 questions targeting your management options
- Endometriomas are common adnexal masses in women affected by endometriosis and may exacerbate pelvic pain and impair fertility. Classification of endometriomas into type I and type II,depending on their etiology and characteristics, can guide minimally invasive surgical management.
- Type I endometriomas arise from invagination of endometrial implants on the ovarian cortex, resulting in dense fibrosis and adhesions. These lesions typically require piecemeal excision in order to completely remove the cyst capsule.
- Type II endometriomas result from invasion of endometrial tissue into preexisting functional cysts and are further subclassified by the proportion of cyst capsule containing endometrial tissue (IIA <10%, IIB 10% to 50%, IIC >50%).
- The difficulty of excising type II endometriomas correlates with the degree of endometrial invasion, with type IIA being relatively straightforward and type IIC being as challenging and piecemeal as type I.
- We generally favor complete excision rather than ablation of the cyst capsule, except for when excision would result in an unacceptable loss of healthy ovarian tissue.
Conclusion
Endometriomas, common adnexal masses in women affected by endometriosis, may exacerbate pelvic pain and impair fertility. Gynecologists should be prepared to excise endometriomas completely and exercise care in preserving as much of the ovarian stroma as possible. We classify endometriomas into 2 types: type I, which develop from invagination of endometrial implants in the ovarian cortex, and type II, which stem from invasion of functional cysts by endometrial implants or type I endometrioma. This distinction guides surgical management. We hope this article and its accompanying video will be helpful in guiding laparoscopic excision of type I and II endometriomas.
Share your thoughts! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
- Nezhat C, Buescher E, Paka C, et al. Video-assisted laparoscopic treatment of endometriosis. In: Nezhat C, Nezhat F, Nezhat C, eds. Nezhat’s Video-Assisted and Robotic-Assisted Laparoscopy and Hysteroscopy. 4th ed. New York, NY: Cambridge University Press; 2013:265–302.
- Burney RO, Giudice LC. Pathogenesis and pathophysiology of endometriosis. Fertil Steril. 2012;98(3):511–519.
- Keyhan S, Hughes C, Price T, Muasher S. An update on surgical versus expectant management of ovarian endometriomas in infertile women. Biomed Res Int. 2015;2015:204792.
- Nezhat F, Nezhat C, Allan CJ, Metzger DA, Sears DL. Clinical and histologic classification of endometriomas. Implications for a mechanism of pathogenesis. J Reprod Med. 1992;37(9):771–776.
- Burney RO, Giudice LC. The pathogenesis of endometriosis. In: Nezhat C, Nezhat F, Nezhat C, eds. Nezhat’s Video-Assisted and Robotic-Assisted Laparoscopy and Hysteroscopy. 4th ed. New York, NY: Cambridge University Press; 2013:252–258.
- Czernobilsky B, Morris WJ. A histologic study of ovarian endometriosis with emphasis on hyperplastic and atypical changes. Obstet Gynecol. 1979;53(3):318–323.
- Nezhat F, Nezhat C, Nezhat C, Admon D. A fresh look at ovarian endometriomas. Contemp Ob Gyn. 1994;39(11):81–94.
- Donnez J, Lousse JC, Jadoul P, Donnez O, Squifflet J. Laparoscopic management of endometriomas using a combined technique of excisional (cystectomy) and ablative surgery. Fertil Steril. 2010;94(1):28–32.
- Nezhat C, Nezhat F, Nezhat C, Seidman DS. Classification of endometriosis. Improving the classification of endometriotic ovarian cysts. Hum Reprod. 1994;9(12):2212–2213.
- Nezhat FR, Pejovic T, Reis FM, Guo SW. The link between endometriosis and ovarian cancer: clinical implications. Int J Gynecol Cancer. 2014;24(4):623–628.
- Nezhat F, Apostol R, Mahmoud M, el Daouk M. Malignant transformation of endometriosis and its clinical significance. Fertil Steril. 2014;102(2):342–344.
- Nezhat FR, Apostal R, Nezhat C, Pejovic T. New insights in the pathophysiology of ovarian cancer and implications for screening and prevention. Am J Obstet Gynecol. 2015;213(3):262–267.
- Hart RJ, Hickey M, Maouris P, Buckett W. Excisional surgery versus ablative surgery for ovarian endometriomata. Cochrane Database Syst Rev. 2008;(2):CD004992.
- Yates J. Endometriosis and infertility: expert answers to 6 questions to help pinpoint the best route to pregnancy. OBG Manag. 2015;27(6):30–35.
- Littman E, Giudice L, Lathi R, Berker B, Milki A, Nezhat C. Role of laparoscopic treatment of endometriosis in patients with failed in vitro fertilization cycles. Fertil Steril. 2005;84(6):1574–1578.
- Exacoustos C, Zupi E, Amadio A, et al. Laparoscopic removal of endometriomas: sonographic evaluation of residual functioning ovarian tissue. Am J Obstet Gynecol. 2004;191(1):68–72.
- Gonçalves FC, Andres MP, Passman LJ, Gonçalves MO, Podgaec S. A systematic review of ultrasonography-guided transvaginal aspiration of recurrent ovarian endometrioma. Int J Gynaecol Obstet. 2016;134(1):3–7.
- Alborzi S, Momtahan M, Parsanezhad ME, Dehbashi S, Zolghadri J, Alborzi S. A prospective, randomized study comparing laparoscopic ovarian cystectomy versus fenestration and coagulation in patients with endometriomas. Fertil Steril. 2004;82(6):1633–1637.
- Nezhat C, Crowgey SR, Garrison CP. Surgical treatment of endometriosis via laser laparoscopy. Fertil Steril. 1986;45(6):778–783.
Related article:
Endometriosis: Expert answers to 7 crucial questions on diagnosis
Etiology
Endometriomas are extensively described in the literature, and their origin is the subject of several theories. In 1921, Sampson noted luteal membrane and ovarian epithelial tissues within endometriomas and was the first to indicate that endometriomas may result from the invasion of functional cysts by endometrial tissue.2,4,5 In 1979, Czernobilsky and Morris6 found endometrial and oviduct-like epithelium in ovarian endometriosis and concluded that ovarian tissue may be a common histologic precursor. Several other authors subsequently have reported finding different types of tissue within ovarian endometriomas, and not all of these chocolate cysts showed histologic evidence of endometriosis.4,7,8
Read about the classification of endometriomas
Disease classification
Our classification system identifies 2 types of endometriomas on the basis of their etiologies and characteristics. Type I, which arise from endometrial tissue implanted on the ovarian surface, are also called true endometriomas. Invagination of cortex and subsequent hemorrhage from endometrial tissue result in cyst formation. Endometrial tissue (endometrial stroma and glands) is histologically present in all type I endometriomas.1,4,9 These endometriomas usually are small (<5 cm in diameter) and have a densely adherent fibrous capsule.4 Often, there is no clear plane between cyst wall and ovarian stroma.3
Type II endometriomas arise from functional cysts involved in or invaded by cortical or pelvic side-wall endometrial implants or by type I endometriomas. Type II endometriomas are subclassified by the extent of endometrial implant involvement in the cyst wall. Type IIA endometriomas are hemorrhagic cysts with less than 10% of endometrial tissue within the cyst wall. Similar to the functional cysts from which they originate, type IIA endometriomas have a cyst wall that is separated easily from ovarian tissue during surgery.4,7,9 Although type II endometriomas tend to be larger than their type I counterparts, in some cases they are identified at an early stage of 2 to 5 cm. Endometriomas larger than 5 cm are almost always type II.4
Type IIB and IIC endometriomas have endometrial implants and fibrosis within their cyst walls, with progressively more endometrial invasion in type IIC endometriomas (>50%) than in type IIB (10% to 50%). Consequently, type IIB cysts are relatively easy to dissect from ovarian tissue, except adjacent to an endometriotic area where the cyst densely adheres to the ovarian stroma. In type IIC, endometrial tissue more extensively penetrates the capsule, making dissection of diseased tissue from the ovarian stroma more difficult; in fact, separating type IIC cyst wall from ovarian stroma can be as challenging as excising a type I endometrioma.7 In most cases, a type IIC cyst is attached by adhesions and fibrosis to the pelvic side wall or uterus and ruptures during mobilization (TABLE).

Related article:
Imaging the endometrioma and mature cystic teratoma
Presentation and diagnosis
Almost all patients with an endometrioma concurrently have peritoneal endometriosis, which is characterized by dysmenorrhea, dyspareunia, chronic pelvic pain, infertility, and, in some cases, gastrointestinal or genitourinary dysfunction.1 Pelvic examination may reveal an adnexal mass that is an endometrioma, or an endometrioma may appear on imaging obtained in a pelvic pain or infertility work-up. Given its 73% sensitivity, 94% specificity, safety, and low cost, transvaginal ultrasonography is the preferred imaging modality for endometrioma.3 The characteristic ultrasonographic appearance is that of a round, homogeneous, fluid-filled mass with low-level echoes.1 Magnetic resonance imaging is appropriate when a more sensitive imaging modality is indicated, as for a patient with risk factors for malignancy.3,10–12
Read about the surgical management of endometriomas
Surgical management
Indications for surgical excision of endometriomas include pelvic pain, infertility, and prevention and diagnosis of malignancy. Endometriomas may be excised prior to use of assisted reproductive technology.13–15 Medical therapy, such as oral contraceptives, can be used to reduce the size of endometriomas but does not improve fertility.3 Certain ovarian cancers are more common in women with endometriosis, and ovarian tumors are thought to develop in about 1% of ovarian endometriosis cases.1,12 Therefore, endometrioma excision may reduce the risk of malignancy. As with other ovarian cysts, large endometriomas may be excised to reduce the risks of rupture and torsion.
Approach
Laparoscopy is the preferred approach for endometrioma excision. Controversy exists regarding the ideal procedure: complete excision (with stripping of the cyst capsule) or drainage and ablation of the cyst wall. Compared with drainage and ablation, excision reduces recurrence of endometriomas; relieves dysmenorrhea, dyspareunia, pelvic pain, and other symptoms; and improves fertility.13,16 The recurrence rate may be as low as 5.8% with complete excision but is 90% with simple transvaginal aspiration.17,18 If not performed properly, however, cyst capsule stripping may damage nearby ovarian stroma and decrease the ovarian reserve.14 Some authors have advocated combining excision and ablation—performing cystectomy until there is no longer a clear plane between capsule and ovarian stroma and then ablating any remaining endometrial tissue.8
With type I and IIC endometriomas, we have seen the endometrial cyst wall infiltrating the ovarian stroma so deeply there is not always a definable plane. By contrast, type IIA and IIB endometriomas typically have a plane between the cyst wall and the ovarian cortex. In type II endometriomas, endometrial implants on the ovarian cortex infiltrate the plane of the cyst wall such that the juxtaposing lipomatous follicular cyst detaches with minimal intraoperative traction. Portions of type II endometriomas containing fibrosis and adhesions may become more difficult to peel off the cyst wall. For most endometriomas, at least 1 spot is difficult to peel off the ovary, and extra care must be taken at the hilum of ovary to avoid excising healthy ovarian cortex.4,5,7,8
Our surgical approach accounts for the described variations in type I and II endometriomas. Endometrial contents often spill as the endometrioma is dissected off neighboring structures. When possible, endometriomas should be aspirated and irrigated prior to cystectomy to avoid seeding the pelvis and abdomen with spilled endometriotic contents. We use hydrodissection, the injection of dilute vasopressin with a laparoscopic needle, to create a plane between cyst wall and ovarian stroma and strip the cyst capsule with laparoscopic graspers. Type I endometriomas adhere densely to the ovary. Given the presence of fibrosis and adhesions, the cyst is excised in a piecemeal fashion. Care is taken to remove any endometrial implants from the ovary while preserving as much of the ovarian tissue as possible.1
Type II endometriomas are larger cysts originating from the invasion of endometrial implants or type I endometrioma into functional cysts. The difficulty of capsule excision varies according to the extent of endometrial invasion. Type IIA endometriomas contain less than 10% endometrial tissue within the cyst capsule. Thus, the standard ovarian cystectomy stripping technique is successful in removing more than 90% of the cyst capsule. Special care is taken in stripping the residual small portion that involves the endometrial glands and stroma and adheres densely to the ovary.
The larger proportion of endometrial tissue present in type IIB and IIC endometriomas degrades the plane between the cyst capsule and the ovarian stroma, making excision more difficult. Similar to the type I excision, a piecemeal approach is often necessary. If complete stripping of the cyst capsule would result in extensive loss of healthy ovarian tissue, then electrocautery, plasma energy, or laser ablation can be selectively used to destroy focal areas of endometrial invasion. Complete ablation may be difficult, as the endometrioma wall can be up to 5 mm thick.19 For these thick-walled endometriomas, we recommend excision (vs ablation), which lowers the risk of endometrioma recurrence.
Related article:
Endometriosis and pain: Expert answers to 6 questions targeting your management options
- Endometriomas are common adnexal masses in women affected by endometriosis and may exacerbate pelvic pain and impair fertility. Classification of endometriomas into type I and type II,depending on their etiology and characteristics, can guide minimally invasive surgical management.
- Type I endometriomas arise from invagination of endometrial implants on the ovarian cortex, resulting in dense fibrosis and adhesions. These lesions typically require piecemeal excision in order to completely remove the cyst capsule.
- Type II endometriomas result from invasion of endometrial tissue into preexisting functional cysts and are further subclassified by the proportion of cyst capsule containing endometrial tissue (IIA <10%, IIB 10% to 50%, IIC >50%).
- The difficulty of excising type II endometriomas correlates with the degree of endometrial invasion, with type IIA being relatively straightforward and type IIC being as challenging and piecemeal as type I.
- We generally favor complete excision rather than ablation of the cyst capsule, except for when excision would result in an unacceptable loss of healthy ovarian tissue.
Conclusion
Endometriomas, common adnexal masses in women affected by endometriosis, may exacerbate pelvic pain and impair fertility. Gynecologists should be prepared to excise endometriomas completely and exercise care in preserving as much of the ovarian stroma as possible. We classify endometriomas into 2 types: type I, which develop from invagination of endometrial implants in the ovarian cortex, and type II, which stem from invasion of functional cysts by endometrial implants or type I endometrioma. This distinction guides surgical management. We hope this article and its accompanying video will be helpful in guiding laparoscopic excision of type I and II endometriomas.
Share your thoughts! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
Related article:
Endometriosis: Expert answers to 7 crucial questions on diagnosis
Etiology
Endometriomas are extensively described in the literature, and their origin is the subject of several theories. In 1921, Sampson noted luteal membrane and ovarian epithelial tissues within endometriomas and was the first to indicate that endometriomas may result from the invasion of functional cysts by endometrial tissue.2,4,5 In 1979, Czernobilsky and Morris6 found endometrial and oviduct-like epithelium in ovarian endometriosis and concluded that ovarian tissue may be a common histologic precursor. Several other authors subsequently have reported finding different types of tissue within ovarian endometriomas, and not all of these chocolate cysts showed histologic evidence of endometriosis.4,7,8
Read about the classification of endometriomas
Disease classification
Our classification system identifies 2 types of endometriomas on the basis of their etiologies and characteristics. Type I, which arise from endometrial tissue implanted on the ovarian surface, are also called true endometriomas. Invagination of cortex and subsequent hemorrhage from endometrial tissue result in cyst formation. Endometrial tissue (endometrial stroma and glands) is histologically present in all type I endometriomas.1,4,9 These endometriomas usually are small (<5 cm in diameter) and have a densely adherent fibrous capsule.4 Often, there is no clear plane between cyst wall and ovarian stroma.3
Type II endometriomas arise from functional cysts involved in or invaded by cortical or pelvic side-wall endometrial implants or by type I endometriomas. Type II endometriomas are subclassified by the extent of endometrial implant involvement in the cyst wall. Type IIA endometriomas are hemorrhagic cysts with less than 10% of endometrial tissue within the cyst wall. Similar to the functional cysts from which they originate, type IIA endometriomas have a cyst wall that is separated easily from ovarian tissue during surgery.4,7,9 Although type II endometriomas tend to be larger than their type I counterparts, in some cases they are identified at an early stage of 2 to 5 cm. Endometriomas larger than 5 cm are almost always type II.4
Type IIB and IIC endometriomas have endometrial implants and fibrosis within their cyst walls, with progressively more endometrial invasion in type IIC endometriomas (>50%) than in type IIB (10% to 50%). Consequently, type IIB cysts are relatively easy to dissect from ovarian tissue, except adjacent to an endometriotic area where the cyst densely adheres to the ovarian stroma. In type IIC, endometrial tissue more extensively penetrates the capsule, making dissection of diseased tissue from the ovarian stroma more difficult; in fact, separating type IIC cyst wall from ovarian stroma can be as challenging as excising a type I endometrioma.7 In most cases, a type IIC cyst is attached by adhesions and fibrosis to the pelvic side wall or uterus and ruptures during mobilization (TABLE).

Related article:
Imaging the endometrioma and mature cystic teratoma
Presentation and diagnosis
Almost all patients with an endometrioma concurrently have peritoneal endometriosis, which is characterized by dysmenorrhea, dyspareunia, chronic pelvic pain, infertility, and, in some cases, gastrointestinal or genitourinary dysfunction.1 Pelvic examination may reveal an adnexal mass that is an endometrioma, or an endometrioma may appear on imaging obtained in a pelvic pain or infertility work-up. Given its 73% sensitivity, 94% specificity, safety, and low cost, transvaginal ultrasonography is the preferred imaging modality for endometrioma.3 The characteristic ultrasonographic appearance is that of a round, homogeneous, fluid-filled mass with low-level echoes.1 Magnetic resonance imaging is appropriate when a more sensitive imaging modality is indicated, as for a patient with risk factors for malignancy.3,10–12
Read about the surgical management of endometriomas
Surgical management
Indications for surgical excision of endometriomas include pelvic pain, infertility, and prevention and diagnosis of malignancy. Endometriomas may be excised prior to use of assisted reproductive technology.13–15 Medical therapy, such as oral contraceptives, can be used to reduce the size of endometriomas but does not improve fertility.3 Certain ovarian cancers are more common in women with endometriosis, and ovarian tumors are thought to develop in about 1% of ovarian endometriosis cases.1,12 Therefore, endometrioma excision may reduce the risk of malignancy. As with other ovarian cysts, large endometriomas may be excised to reduce the risks of rupture and torsion.
Approach
Laparoscopy is the preferred approach for endometrioma excision. Controversy exists regarding the ideal procedure: complete excision (with stripping of the cyst capsule) or drainage and ablation of the cyst wall. Compared with drainage and ablation, excision reduces recurrence of endometriomas; relieves dysmenorrhea, dyspareunia, pelvic pain, and other symptoms; and improves fertility.13,16 The recurrence rate may be as low as 5.8% with complete excision but is 90% with simple transvaginal aspiration.17,18 If not performed properly, however, cyst capsule stripping may damage nearby ovarian stroma and decrease the ovarian reserve.14 Some authors have advocated combining excision and ablation—performing cystectomy until there is no longer a clear plane between capsule and ovarian stroma and then ablating any remaining endometrial tissue.8
With type I and IIC endometriomas, we have seen the endometrial cyst wall infiltrating the ovarian stroma so deeply there is not always a definable plane. By contrast, type IIA and IIB endometriomas typically have a plane between the cyst wall and the ovarian cortex. In type II endometriomas, endometrial implants on the ovarian cortex infiltrate the plane of the cyst wall such that the juxtaposing lipomatous follicular cyst detaches with minimal intraoperative traction. Portions of type II endometriomas containing fibrosis and adhesions may become more difficult to peel off the cyst wall. For most endometriomas, at least 1 spot is difficult to peel off the ovary, and extra care must be taken at the hilum of ovary to avoid excising healthy ovarian cortex.4,5,7,8
Our surgical approach accounts for the described variations in type I and II endometriomas. Endometrial contents often spill as the endometrioma is dissected off neighboring structures. When possible, endometriomas should be aspirated and irrigated prior to cystectomy to avoid seeding the pelvis and abdomen with spilled endometriotic contents. We use hydrodissection, the injection of dilute vasopressin with a laparoscopic needle, to create a plane between cyst wall and ovarian stroma and strip the cyst capsule with laparoscopic graspers. Type I endometriomas adhere densely to the ovary. Given the presence of fibrosis and adhesions, the cyst is excised in a piecemeal fashion. Care is taken to remove any endometrial implants from the ovary while preserving as much of the ovarian tissue as possible.1
Type II endometriomas are larger cysts originating from the invasion of endometrial implants or type I endometrioma into functional cysts. The difficulty of capsule excision varies according to the extent of endometrial invasion. Type IIA endometriomas contain less than 10% endometrial tissue within the cyst capsule. Thus, the standard ovarian cystectomy stripping technique is successful in removing more than 90% of the cyst capsule. Special care is taken in stripping the residual small portion that involves the endometrial glands and stroma and adheres densely to the ovary.
The larger proportion of endometrial tissue present in type IIB and IIC endometriomas degrades the plane between the cyst capsule and the ovarian stroma, making excision more difficult. Similar to the type I excision, a piecemeal approach is often necessary. If complete stripping of the cyst capsule would result in extensive loss of healthy ovarian tissue, then electrocautery, plasma energy, or laser ablation can be selectively used to destroy focal areas of endometrial invasion. Complete ablation may be difficult, as the endometrioma wall can be up to 5 mm thick.19 For these thick-walled endometriomas, we recommend excision (vs ablation), which lowers the risk of endometrioma recurrence.
Related article:
Endometriosis and pain: Expert answers to 6 questions targeting your management options
- Endometriomas are common adnexal masses in women affected by endometriosis and may exacerbate pelvic pain and impair fertility. Classification of endometriomas into type I and type II,depending on their etiology and characteristics, can guide minimally invasive surgical management.
- Type I endometriomas arise from invagination of endometrial implants on the ovarian cortex, resulting in dense fibrosis and adhesions. These lesions typically require piecemeal excision in order to completely remove the cyst capsule.
- Type II endometriomas result from invasion of endometrial tissue into preexisting functional cysts and are further subclassified by the proportion of cyst capsule containing endometrial tissue (IIA <10%, IIB 10% to 50%, IIC >50%).
- The difficulty of excising type II endometriomas correlates with the degree of endometrial invasion, with type IIA being relatively straightforward and type IIC being as challenging and piecemeal as type I.
- We generally favor complete excision rather than ablation of the cyst capsule, except for when excision would result in an unacceptable loss of healthy ovarian tissue.
Conclusion
Endometriomas, common adnexal masses in women affected by endometriosis, may exacerbate pelvic pain and impair fertility. Gynecologists should be prepared to excise endometriomas completely and exercise care in preserving as much of the ovarian stroma as possible. We classify endometriomas into 2 types: type I, which develop from invagination of endometrial implants in the ovarian cortex, and type II, which stem from invasion of functional cysts by endometrial implants or type I endometrioma. This distinction guides surgical management. We hope this article and its accompanying video will be helpful in guiding laparoscopic excision of type I and II endometriomas.
Share your thoughts! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
- Nezhat C, Buescher E, Paka C, et al. Video-assisted laparoscopic treatment of endometriosis. In: Nezhat C, Nezhat F, Nezhat C, eds. Nezhat’s Video-Assisted and Robotic-Assisted Laparoscopy and Hysteroscopy. 4th ed. New York, NY: Cambridge University Press; 2013:265–302.
- Burney RO, Giudice LC. Pathogenesis and pathophysiology of endometriosis. Fertil Steril. 2012;98(3):511–519.
- Keyhan S, Hughes C, Price T, Muasher S. An update on surgical versus expectant management of ovarian endometriomas in infertile women. Biomed Res Int. 2015;2015:204792.
- Nezhat F, Nezhat C, Allan CJ, Metzger DA, Sears DL. Clinical and histologic classification of endometriomas. Implications for a mechanism of pathogenesis. J Reprod Med. 1992;37(9):771–776.
- Burney RO, Giudice LC. The pathogenesis of endometriosis. In: Nezhat C, Nezhat F, Nezhat C, eds. Nezhat’s Video-Assisted and Robotic-Assisted Laparoscopy and Hysteroscopy. 4th ed. New York, NY: Cambridge University Press; 2013:252–258.
- Czernobilsky B, Morris WJ. A histologic study of ovarian endometriosis with emphasis on hyperplastic and atypical changes. Obstet Gynecol. 1979;53(3):318–323.
- Nezhat F, Nezhat C, Nezhat C, Admon D. A fresh look at ovarian endometriomas. Contemp Ob Gyn. 1994;39(11):81–94.
- Donnez J, Lousse JC, Jadoul P, Donnez O, Squifflet J. Laparoscopic management of endometriomas using a combined technique of excisional (cystectomy) and ablative surgery. Fertil Steril. 2010;94(1):28–32.
- Nezhat C, Nezhat F, Nezhat C, Seidman DS. Classification of endometriosis. Improving the classification of endometriotic ovarian cysts. Hum Reprod. 1994;9(12):2212–2213.
- Nezhat FR, Pejovic T, Reis FM, Guo SW. The link between endometriosis and ovarian cancer: clinical implications. Int J Gynecol Cancer. 2014;24(4):623–628.
- Nezhat F, Apostol R, Mahmoud M, el Daouk M. Malignant transformation of endometriosis and its clinical significance. Fertil Steril. 2014;102(2):342–344.
- Nezhat FR, Apostal R, Nezhat C, Pejovic T. New insights in the pathophysiology of ovarian cancer and implications for screening and prevention. Am J Obstet Gynecol. 2015;213(3):262–267.
- Hart RJ, Hickey M, Maouris P, Buckett W. Excisional surgery versus ablative surgery for ovarian endometriomata. Cochrane Database Syst Rev. 2008;(2):CD004992.
- Yates J. Endometriosis and infertility: expert answers to 6 questions to help pinpoint the best route to pregnancy. OBG Manag. 2015;27(6):30–35.
- Littman E, Giudice L, Lathi R, Berker B, Milki A, Nezhat C. Role of laparoscopic treatment of endometriosis in patients with failed in vitro fertilization cycles. Fertil Steril. 2005;84(6):1574–1578.
- Exacoustos C, Zupi E, Amadio A, et al. Laparoscopic removal of endometriomas: sonographic evaluation of residual functioning ovarian tissue. Am J Obstet Gynecol. 2004;191(1):68–72.
- Gonçalves FC, Andres MP, Passman LJ, Gonçalves MO, Podgaec S. A systematic review of ultrasonography-guided transvaginal aspiration of recurrent ovarian endometrioma. Int J Gynaecol Obstet. 2016;134(1):3–7.
- Alborzi S, Momtahan M, Parsanezhad ME, Dehbashi S, Zolghadri J, Alborzi S. A prospective, randomized study comparing laparoscopic ovarian cystectomy versus fenestration and coagulation in patients with endometriomas. Fertil Steril. 2004;82(6):1633–1637.
- Nezhat C, Crowgey SR, Garrison CP. Surgical treatment of endometriosis via laser laparoscopy. Fertil Steril. 1986;45(6):778–783.
- Nezhat C, Buescher E, Paka C, et al. Video-assisted laparoscopic treatment of endometriosis. In: Nezhat C, Nezhat F, Nezhat C, eds. Nezhat’s Video-Assisted and Robotic-Assisted Laparoscopy and Hysteroscopy. 4th ed. New York, NY: Cambridge University Press; 2013:265–302.
- Burney RO, Giudice LC. Pathogenesis and pathophysiology of endometriosis. Fertil Steril. 2012;98(3):511–519.
- Keyhan S, Hughes C, Price T, Muasher S. An update on surgical versus expectant management of ovarian endometriomas in infertile women. Biomed Res Int. 2015;2015:204792.
- Nezhat F, Nezhat C, Allan CJ, Metzger DA, Sears DL. Clinical and histologic classification of endometriomas. Implications for a mechanism of pathogenesis. J Reprod Med. 1992;37(9):771–776.
- Burney RO, Giudice LC. The pathogenesis of endometriosis. In: Nezhat C, Nezhat F, Nezhat C, eds. Nezhat’s Video-Assisted and Robotic-Assisted Laparoscopy and Hysteroscopy. 4th ed. New York, NY: Cambridge University Press; 2013:252–258.
- Czernobilsky B, Morris WJ. A histologic study of ovarian endometriosis with emphasis on hyperplastic and atypical changes. Obstet Gynecol. 1979;53(3):318–323.
- Nezhat F, Nezhat C, Nezhat C, Admon D. A fresh look at ovarian endometriomas. Contemp Ob Gyn. 1994;39(11):81–94.
- Donnez J, Lousse JC, Jadoul P, Donnez O, Squifflet J. Laparoscopic management of endometriomas using a combined technique of excisional (cystectomy) and ablative surgery. Fertil Steril. 2010;94(1):28–32.
- Nezhat C, Nezhat F, Nezhat C, Seidman DS. Classification of endometriosis. Improving the classification of endometriotic ovarian cysts. Hum Reprod. 1994;9(12):2212–2213.
- Nezhat FR, Pejovic T, Reis FM, Guo SW. The link between endometriosis and ovarian cancer: clinical implications. Int J Gynecol Cancer. 2014;24(4):623–628.
- Nezhat F, Apostol R, Mahmoud M, el Daouk M. Malignant transformation of endometriosis and its clinical significance. Fertil Steril. 2014;102(2):342–344.
- Nezhat FR, Apostal R, Nezhat C, Pejovic T. New insights in the pathophysiology of ovarian cancer and implications for screening and prevention. Am J Obstet Gynecol. 2015;213(3):262–267.
- Hart RJ, Hickey M, Maouris P, Buckett W. Excisional surgery versus ablative surgery for ovarian endometriomata. Cochrane Database Syst Rev. 2008;(2):CD004992.
- Yates J. Endometriosis and infertility: expert answers to 6 questions to help pinpoint the best route to pregnancy. OBG Manag. 2015;27(6):30–35.
- Littman E, Giudice L, Lathi R, Berker B, Milki A, Nezhat C. Role of laparoscopic treatment of endometriosis in patients with failed in vitro fertilization cycles. Fertil Steril. 2005;84(6):1574–1578.
- Exacoustos C, Zupi E, Amadio A, et al. Laparoscopic removal of endometriomas: sonographic evaluation of residual functioning ovarian tissue. Am J Obstet Gynecol. 2004;191(1):68–72.
- Gonçalves FC, Andres MP, Passman LJ, Gonçalves MO, Podgaec S. A systematic review of ultrasonography-guided transvaginal aspiration of recurrent ovarian endometrioma. Int J Gynaecol Obstet. 2016;134(1):3–7.
- Alborzi S, Momtahan M, Parsanezhad ME, Dehbashi S, Zolghadri J, Alborzi S. A prospective, randomized study comparing laparoscopic ovarian cystectomy versus fenestration and coagulation in patients with endometriomas. Fertil Steril. 2004;82(6):1633–1637.
- Nezhat C, Crowgey SR, Garrison CP. Surgical treatment of endometriosis via laser laparoscopy. Fertil Steril. 1986;45(6):778–783.
IN THIS ARTICLE
Managing endometriosis to prevent ovarian cancer
Endometriosis is a common condition, occurring in this country in 1 of 10 women of reproductive age. An association between endometriosis and subsequent ovarian carcinoma has been reported for decades, yet it is only recently that our knowledge has deepened enough to support more rational methods for preventing the malignancy.
Each year, approximately 22,000 new cases of ovarian cancer are diagnosed. The lifetime risk of developing this malignancy is low, but it is the deadliest of the gynecologic malignancies, with diagnosis usually made in advanced stages when prognosis is poor.
Endometriosis shows some characteristics of malignancy, such as the development of local and distant foci, and attachment to and invasion of other tissues with subsequent damage to these tissues. Endometriosis also is characterized by recurrent, unregulated cell proliferation and estrogen-dependent growth.
Our attempts during the past 2 decades to detect ovarian carcinoma at the early stages through a combined screening modality involving transvaginal ultrasound and a test for the serum level of cancer antigen 125 have failed to provide any survival benefit or even any measurable reduction in morbidity. Today, early-stage ovarian carcinoma, which has a 5-year survival rate of more than 90%, is diagnosed in only a minority of women.
There is good news, however. In recent years our insight into the pathophysiology of ovarian cancer has deepened, providing us with a new paradigm for ovarian cancer pathogenesis that divides ovarian epithelial carcinoma into two distinct types with distinct molecular profiles – one which originates largely in the distal portion of the fallopian tube and the other which traces back to endometriosis.
This new paradigm strengthens and helps to explain the reported association between endometriosis and ovarian cancer. It also has important clinical implications for current practice. While we have much more to learn about the etiology of endometriosis and the causes of malignant transformation, our current knowledge provides a strong rationale for identification and close monitoring of some patients with endometriosis deemed at risk for ovarian cancer, risk-reducing medical management, earlier and more meticulous surgical treatment, and close monitoring.
By combining this new approach to endometriosis with consideration of salpingectomy after completion of childbearing, we have an unprecedented opportunity to reduce the incidence of epithelial ovarian cancer.
Dual pathogenesis
The majority of ovarian cancers are of epithelial origin and fall into four histologic categories: serous, endometrioid, clear cell, and mucinous. In recent years, we have gained a deeper understanding of the pathogenesis of ovarian carcinoma, with an array of epidemiologic, histologic, and molecular data showing us that epithelial ovarian cancers are also of two distinct types (Am J Obstet Gynecol. 2015 Sep;213[3]:262-7).
One of these types, a high-grade serous carcinoma, appears to arise in many cases in the epithelium of the fallopian tube. The other type of tumor is a low-grade carcinoma – particularly of the endometrioid and clear cell histologic subtypes – that originates largely from ovarian endometriotic lesions or from borderline serous tumors in the case of serous histology.
The majority of diagnosed stage 1 ovarian cancers are carcinomas of this low-grade type and not high-grade serous carcinomas. In a study of 76 consecutive stage 1 carcinomas, investigators found that ovarian endometriosis was present in 40 of the 76 cases. More than two-thirds of the 76 cases (71%) were nonserous cancers, and almost all of these cases were associated with endometriosis based on histologic examination (Fertil Steril. 2007 Oct;88[4]:906-10).
This study was among the first to show that the majority of stage 1 ovarian carcinomas are not high-grade serous carcinomas, but rather nonserous, primarily endometrioid and clear cell, cancers. The research demonstrated that endometriosis should be viewed as a potential precursor lesion to specific subtypes of ovarian cancer.
The malignant transformation of endometriosis was first suggested by Dr. J. A. Sampson in 1925, and a number of studies – in addition to the 2007 landmark study – have since described ovarian cancer arising from endometriosis, based on the frequent co-occurrence in surgical specimens.
Most recently, a study from the Ovarian Cancer Association Consortium (OCAC) found that women who reported a history of endometriosis had a significantly higher risk of developing ovarian cancer than the general population (odds ratio, 1.46).
Investigators of this critical study pooled data from 13 ovarian cancer case-control studies involving more than 13,226 controls and 7,911 women with invasive epithelial ovarian cancer – 818 (6.2%) and 738 (9.3%) of whom, respectively, reported a history of endometriosis. Specifically, they determined that self-reported endometriosis was associated with a 3.05-fold increased risk for clear cell invasive ovarian cancer and a 2.04-fold increased risk of endometrioid ovarian cancer.
Moreover, a significant association between preexisting endometriosis and low-grade serous invasive ovarian cancer (OR, 2.11) was demonstrated, while no association was found between endometriosis and the risk of high-grade serous invasive ovarian cancer (Lancet Oncol. 2012 Apr;13[4]:385-94).
A second recently published report – a meta-analysis of 20 case-control and 15 cohort studies published between 1990 and 2012 and involving more than 444,000 patients – found that endometriosis increased cancer risk in case-control or two-arm cohort studies by 27% (relative risk, 1.265) and by approximately 80% in single-arm cohort studies (standard incidence ratio, 1.797). Endometrioid and clear cell carcinomas were more common in endometriosis-associated ovarian cancer, while serous carcinoma was less frequent (Br J Cancer. 2014 Apr 2;110[7]:1878-90).
Findings of both of these large studies have served to clarify the association between endometriosis and specific histologic subtypes and suggested that there are important differences in the pathogenesis of low-grade and high-grade serous ovarian carcinomas.
Clinical implications
It is not clear what causes malignant transformation or what predisposes some patients with endometriosis to develop ovarian cancer, but the risk likely involves genetic and epigenetic influences as well as immunologic, inflammatory, and hormonal factors.
The molecular profiles of the main two types of ovarian cancer are different: While the majority of high-grade serous ovarian tumors are characterized by TP53 mutations, the low-grade carcinomas are characterized by a variety of mutations, including KRAS, BRAF, ERBB2, CTNNB1, and BCL2 mutations.
There currently are not enough data to recommend genetic screening tests in patients with endometriosis, but our hope is that we eventually will be able to screen for “high-risk” endometriotic lesions by testing for genes specific to various histologic subtypes of low-grade ovarian cancer, or by finding and utilizing other biomarkers.
In the meantime, we believe it is important to more thoroughly treat endometriosis and to identify and follow women with a history of the condition, especially those with a long-standing history, those with a history of endometriosis associated with infertility, and those with ovarian endometrioma. Each of these factors predisposes patients to a higher risk of malignant transformation.
Complete surgical resection of all visible endometriosis is the most effective treatment and will afford the best cancer prevention, even in women who are asymptomatic. In a recent Swedish national registry case-control study, women who underwent radical surgical excision of all visible endometriosis were significantly less likely (OR, 0.30) to develop ovarian cancer (Acta Obstet Gynecol Scand. 2013 May;92[5]:546-54).
Suppressive hormonal therapy is another treatment option for patients with no interest in conceiving. Most large endometriomas are functional ovarian cysts that have been invaded by cortical ovarian endometriosis or by small primary endometriomas (J Reprod Med. 1992 Sep;37[9]:771-6).
While hormonal therapy will not always result in complete regression of endometriotic lesions, it will decrease the recurrence rate of endometriomas and can be considered for long-term prevention of potentially premalignant lesions. It is most effective when it follows surgical excision of endometriomas and associated endometriosis.
A patient who has completed childbearing at the time of surgical resection may be offered bilateral salpingectomy, regardless of menopausal status. Salpingectomy in both average and high-risk populations (e.g., BRCA 1/2 carriers) not only prevents high-grade serous carcinoma by eliminating the site of origin, but also may decrease the risk of endometrioid and clear cell carcinoma by blocking the passageway that enables the flow of endometrium and factors that induce inflammation. It is estimated that the procedure reduces the risk of ovarian cancer by 40%.
Interestingly, tubal ligation has historically been shown to decrease the risk of ovarian cancer, and recent data have shown that the risk of endometrioid and clear cell carcinoma is cut even more than the risk of high-grade serous carcinoma (Int J Epidemiol. 2013 Apr;42[2]:579-89).
The Society of Gynecologic Oncology recommends that risk-reducing salpingectomy be considered at the time of hysterectomy or other abdominal or pelvic surgery, and in lieu of tubal ligation. The American College of Obstetricians and Gynecologists similarly has stated that prophylactic salpingectomy may offer clinicians the opportunity to prevent ovarian cancer in their patients. Salpingectomy is an important option for all patients, but is especially important when the fallopian tubes are found to be damaged by endometriosis and/or pelvic inflammatory disease. When imaging studies show that endometriomas are present and resection is not performed, pelvic ultrasound should become part of the patient’s routine examination.
Most endometriomas have a homogeneous appearance; any significant increase in size or a change in the homogeneous cystic characteristics to a more heterogeneous appearance with mural components should raise suspicion about malignant change.
It can be difficult to detect relatively small endocystic components with ultrasound, so if there is any doubt about whether there is some heterogeneous consistency, an MRI should be performed. MRI is showing more promise in detecting malignant change. Hyperdense mural nodules within the ovary and rapid growth of an endometrioma have both been associated with malignant transformation and can be seen on these images.
In a cohort study comparing MRI findings of 10 patients with ovarian adenocarcinoma to 10 patients with benign endometriomas, investigators found mural nodules in all 10 malignancies but in only three of the benign cases (AJR Am J Roentgenol. 2000 Nov;175[5]:1423-30).
Long-term follow-up is necessary to understand the timeline of transformation in patients with mural nodules. This together with increasing knowledge of molecular events underpinning evolution of endometriosis will lead to better screening and preventive strategies.
Dr. Nezhat is the director of minimally invasive gynecologic surgery and robotics at Winthrop University Hospital in Mineola, N.Y., and an adjunct professor of obstetrics, gynecology, and reproductive medicine at the State University of New York at Stony Brook. He reported having no financial disclosures.
Endometriosis is a common condition, occurring in this country in 1 of 10 women of reproductive age. An association between endometriosis and subsequent ovarian carcinoma has been reported for decades, yet it is only recently that our knowledge has deepened enough to support more rational methods for preventing the malignancy.
Each year, approximately 22,000 new cases of ovarian cancer are diagnosed. The lifetime risk of developing this malignancy is low, but it is the deadliest of the gynecologic malignancies, with diagnosis usually made in advanced stages when prognosis is poor.
Endometriosis shows some characteristics of malignancy, such as the development of local and distant foci, and attachment to and invasion of other tissues with subsequent damage to these tissues. Endometriosis also is characterized by recurrent, unregulated cell proliferation and estrogen-dependent growth.
Our attempts during the past 2 decades to detect ovarian carcinoma at the early stages through a combined screening modality involving transvaginal ultrasound and a test for the serum level of cancer antigen 125 have failed to provide any survival benefit or even any measurable reduction in morbidity. Today, early-stage ovarian carcinoma, which has a 5-year survival rate of more than 90%, is diagnosed in only a minority of women.
There is good news, however. In recent years our insight into the pathophysiology of ovarian cancer has deepened, providing us with a new paradigm for ovarian cancer pathogenesis that divides ovarian epithelial carcinoma into two distinct types with distinct molecular profiles – one which originates largely in the distal portion of the fallopian tube and the other which traces back to endometriosis.
This new paradigm strengthens and helps to explain the reported association between endometriosis and ovarian cancer. It also has important clinical implications for current practice. While we have much more to learn about the etiology of endometriosis and the causes of malignant transformation, our current knowledge provides a strong rationale for identification and close monitoring of some patients with endometriosis deemed at risk for ovarian cancer, risk-reducing medical management, earlier and more meticulous surgical treatment, and close monitoring.
By combining this new approach to endometriosis with consideration of salpingectomy after completion of childbearing, we have an unprecedented opportunity to reduce the incidence of epithelial ovarian cancer.
Dual pathogenesis
The majority of ovarian cancers are of epithelial origin and fall into four histologic categories: serous, endometrioid, clear cell, and mucinous. In recent years, we have gained a deeper understanding of the pathogenesis of ovarian carcinoma, with an array of epidemiologic, histologic, and molecular data showing us that epithelial ovarian cancers are also of two distinct types (Am J Obstet Gynecol. 2015 Sep;213[3]:262-7).
One of these types, a high-grade serous carcinoma, appears to arise in many cases in the epithelium of the fallopian tube. The other type of tumor is a low-grade carcinoma – particularly of the endometrioid and clear cell histologic subtypes – that originates largely from ovarian endometriotic lesions or from borderline serous tumors in the case of serous histology.
The majority of diagnosed stage 1 ovarian cancers are carcinomas of this low-grade type and not high-grade serous carcinomas. In a study of 76 consecutive stage 1 carcinomas, investigators found that ovarian endometriosis was present in 40 of the 76 cases. More than two-thirds of the 76 cases (71%) were nonserous cancers, and almost all of these cases were associated with endometriosis based on histologic examination (Fertil Steril. 2007 Oct;88[4]:906-10).
This study was among the first to show that the majority of stage 1 ovarian carcinomas are not high-grade serous carcinomas, but rather nonserous, primarily endometrioid and clear cell, cancers. The research demonstrated that endometriosis should be viewed as a potential precursor lesion to specific subtypes of ovarian cancer.
The malignant transformation of endometriosis was first suggested by Dr. J. A. Sampson in 1925, and a number of studies – in addition to the 2007 landmark study – have since described ovarian cancer arising from endometriosis, based on the frequent co-occurrence in surgical specimens.
Most recently, a study from the Ovarian Cancer Association Consortium (OCAC) found that women who reported a history of endometriosis had a significantly higher risk of developing ovarian cancer than the general population (odds ratio, 1.46).
Investigators of this critical study pooled data from 13 ovarian cancer case-control studies involving more than 13,226 controls and 7,911 women with invasive epithelial ovarian cancer – 818 (6.2%) and 738 (9.3%) of whom, respectively, reported a history of endometriosis. Specifically, they determined that self-reported endometriosis was associated with a 3.05-fold increased risk for clear cell invasive ovarian cancer and a 2.04-fold increased risk of endometrioid ovarian cancer.
Moreover, a significant association between preexisting endometriosis and low-grade serous invasive ovarian cancer (OR, 2.11) was demonstrated, while no association was found between endometriosis and the risk of high-grade serous invasive ovarian cancer (Lancet Oncol. 2012 Apr;13[4]:385-94).
A second recently published report – a meta-analysis of 20 case-control and 15 cohort studies published between 1990 and 2012 and involving more than 444,000 patients – found that endometriosis increased cancer risk in case-control or two-arm cohort studies by 27% (relative risk, 1.265) and by approximately 80% in single-arm cohort studies (standard incidence ratio, 1.797). Endometrioid and clear cell carcinomas were more common in endometriosis-associated ovarian cancer, while serous carcinoma was less frequent (Br J Cancer. 2014 Apr 2;110[7]:1878-90).
Findings of both of these large studies have served to clarify the association between endometriosis and specific histologic subtypes and suggested that there are important differences in the pathogenesis of low-grade and high-grade serous ovarian carcinomas.
Clinical implications
It is not clear what causes malignant transformation or what predisposes some patients with endometriosis to develop ovarian cancer, but the risk likely involves genetic and epigenetic influences as well as immunologic, inflammatory, and hormonal factors.
The molecular profiles of the main two types of ovarian cancer are different: While the majority of high-grade serous ovarian tumors are characterized by TP53 mutations, the low-grade carcinomas are characterized by a variety of mutations, including KRAS, BRAF, ERBB2, CTNNB1, and BCL2 mutations.
There currently are not enough data to recommend genetic screening tests in patients with endometriosis, but our hope is that we eventually will be able to screen for “high-risk” endometriotic lesions by testing for genes specific to various histologic subtypes of low-grade ovarian cancer, or by finding and utilizing other biomarkers.
In the meantime, we believe it is important to more thoroughly treat endometriosis and to identify and follow women with a history of the condition, especially those with a long-standing history, those with a history of endometriosis associated with infertility, and those with ovarian endometrioma. Each of these factors predisposes patients to a higher risk of malignant transformation.
Complete surgical resection of all visible endometriosis is the most effective treatment and will afford the best cancer prevention, even in women who are asymptomatic. In a recent Swedish national registry case-control study, women who underwent radical surgical excision of all visible endometriosis were significantly less likely (OR, 0.30) to develop ovarian cancer (Acta Obstet Gynecol Scand. 2013 May;92[5]:546-54).
Suppressive hormonal therapy is another treatment option for patients with no interest in conceiving. Most large endometriomas are functional ovarian cysts that have been invaded by cortical ovarian endometriosis or by small primary endometriomas (J Reprod Med. 1992 Sep;37[9]:771-6).
While hormonal therapy will not always result in complete regression of endometriotic lesions, it will decrease the recurrence rate of endometriomas and can be considered for long-term prevention of potentially premalignant lesions. It is most effective when it follows surgical excision of endometriomas and associated endometriosis.
A patient who has completed childbearing at the time of surgical resection may be offered bilateral salpingectomy, regardless of menopausal status. Salpingectomy in both average and high-risk populations (e.g., BRCA 1/2 carriers) not only prevents high-grade serous carcinoma by eliminating the site of origin, but also may decrease the risk of endometrioid and clear cell carcinoma by blocking the passageway that enables the flow of endometrium and factors that induce inflammation. It is estimated that the procedure reduces the risk of ovarian cancer by 40%.
Interestingly, tubal ligation has historically been shown to decrease the risk of ovarian cancer, and recent data have shown that the risk of endometrioid and clear cell carcinoma is cut even more than the risk of high-grade serous carcinoma (Int J Epidemiol. 2013 Apr;42[2]:579-89).
The Society of Gynecologic Oncology recommends that risk-reducing salpingectomy be considered at the time of hysterectomy or other abdominal or pelvic surgery, and in lieu of tubal ligation. The American College of Obstetricians and Gynecologists similarly has stated that prophylactic salpingectomy may offer clinicians the opportunity to prevent ovarian cancer in their patients. Salpingectomy is an important option for all patients, but is especially important when the fallopian tubes are found to be damaged by endometriosis and/or pelvic inflammatory disease. When imaging studies show that endometriomas are present and resection is not performed, pelvic ultrasound should become part of the patient’s routine examination.
Most endometriomas have a homogeneous appearance; any significant increase in size or a change in the homogeneous cystic characteristics to a more heterogeneous appearance with mural components should raise suspicion about malignant change.
It can be difficult to detect relatively small endocystic components with ultrasound, so if there is any doubt about whether there is some heterogeneous consistency, an MRI should be performed. MRI is showing more promise in detecting malignant change. Hyperdense mural nodules within the ovary and rapid growth of an endometrioma have both been associated with malignant transformation and can be seen on these images.
In a cohort study comparing MRI findings of 10 patients with ovarian adenocarcinoma to 10 patients with benign endometriomas, investigators found mural nodules in all 10 malignancies but in only three of the benign cases (AJR Am J Roentgenol. 2000 Nov;175[5]:1423-30).
Long-term follow-up is necessary to understand the timeline of transformation in patients with mural nodules. This together with increasing knowledge of molecular events underpinning evolution of endometriosis will lead to better screening and preventive strategies.
Dr. Nezhat is the director of minimally invasive gynecologic surgery and robotics at Winthrop University Hospital in Mineola, N.Y., and an adjunct professor of obstetrics, gynecology, and reproductive medicine at the State University of New York at Stony Brook. He reported having no financial disclosures.
Endometriosis is a common condition, occurring in this country in 1 of 10 women of reproductive age. An association between endometriosis and subsequent ovarian carcinoma has been reported for decades, yet it is only recently that our knowledge has deepened enough to support more rational methods for preventing the malignancy.
Each year, approximately 22,000 new cases of ovarian cancer are diagnosed. The lifetime risk of developing this malignancy is low, but it is the deadliest of the gynecologic malignancies, with diagnosis usually made in advanced stages when prognosis is poor.
Endometriosis shows some characteristics of malignancy, such as the development of local and distant foci, and attachment to and invasion of other tissues with subsequent damage to these tissues. Endometriosis also is characterized by recurrent, unregulated cell proliferation and estrogen-dependent growth.
Our attempts during the past 2 decades to detect ovarian carcinoma at the early stages through a combined screening modality involving transvaginal ultrasound and a test for the serum level of cancer antigen 125 have failed to provide any survival benefit or even any measurable reduction in morbidity. Today, early-stage ovarian carcinoma, which has a 5-year survival rate of more than 90%, is diagnosed in only a minority of women.
There is good news, however. In recent years our insight into the pathophysiology of ovarian cancer has deepened, providing us with a new paradigm for ovarian cancer pathogenesis that divides ovarian epithelial carcinoma into two distinct types with distinct molecular profiles – one which originates largely in the distal portion of the fallopian tube and the other which traces back to endometriosis.
This new paradigm strengthens and helps to explain the reported association between endometriosis and ovarian cancer. It also has important clinical implications for current practice. While we have much more to learn about the etiology of endometriosis and the causes of malignant transformation, our current knowledge provides a strong rationale for identification and close monitoring of some patients with endometriosis deemed at risk for ovarian cancer, risk-reducing medical management, earlier and more meticulous surgical treatment, and close monitoring.
By combining this new approach to endometriosis with consideration of salpingectomy after completion of childbearing, we have an unprecedented opportunity to reduce the incidence of epithelial ovarian cancer.
Dual pathogenesis
The majority of ovarian cancers are of epithelial origin and fall into four histologic categories: serous, endometrioid, clear cell, and mucinous. In recent years, we have gained a deeper understanding of the pathogenesis of ovarian carcinoma, with an array of epidemiologic, histologic, and molecular data showing us that epithelial ovarian cancers are also of two distinct types (Am J Obstet Gynecol. 2015 Sep;213[3]:262-7).
One of these types, a high-grade serous carcinoma, appears to arise in many cases in the epithelium of the fallopian tube. The other type of tumor is a low-grade carcinoma – particularly of the endometrioid and clear cell histologic subtypes – that originates largely from ovarian endometriotic lesions or from borderline serous tumors in the case of serous histology.
The majority of diagnosed stage 1 ovarian cancers are carcinomas of this low-grade type and not high-grade serous carcinomas. In a study of 76 consecutive stage 1 carcinomas, investigators found that ovarian endometriosis was present in 40 of the 76 cases. More than two-thirds of the 76 cases (71%) were nonserous cancers, and almost all of these cases were associated with endometriosis based on histologic examination (Fertil Steril. 2007 Oct;88[4]:906-10).
This study was among the first to show that the majority of stage 1 ovarian carcinomas are not high-grade serous carcinomas, but rather nonserous, primarily endometrioid and clear cell, cancers. The research demonstrated that endometriosis should be viewed as a potential precursor lesion to specific subtypes of ovarian cancer.
The malignant transformation of endometriosis was first suggested by Dr. J. A. Sampson in 1925, and a number of studies – in addition to the 2007 landmark study – have since described ovarian cancer arising from endometriosis, based on the frequent co-occurrence in surgical specimens.
Most recently, a study from the Ovarian Cancer Association Consortium (OCAC) found that women who reported a history of endometriosis had a significantly higher risk of developing ovarian cancer than the general population (odds ratio, 1.46).
Investigators of this critical study pooled data from 13 ovarian cancer case-control studies involving more than 13,226 controls and 7,911 women with invasive epithelial ovarian cancer – 818 (6.2%) and 738 (9.3%) of whom, respectively, reported a history of endometriosis. Specifically, they determined that self-reported endometriosis was associated with a 3.05-fold increased risk for clear cell invasive ovarian cancer and a 2.04-fold increased risk of endometrioid ovarian cancer.
Moreover, a significant association between preexisting endometriosis and low-grade serous invasive ovarian cancer (OR, 2.11) was demonstrated, while no association was found between endometriosis and the risk of high-grade serous invasive ovarian cancer (Lancet Oncol. 2012 Apr;13[4]:385-94).
A second recently published report – a meta-analysis of 20 case-control and 15 cohort studies published between 1990 and 2012 and involving more than 444,000 patients – found that endometriosis increased cancer risk in case-control or two-arm cohort studies by 27% (relative risk, 1.265) and by approximately 80% in single-arm cohort studies (standard incidence ratio, 1.797). Endometrioid and clear cell carcinomas were more common in endometriosis-associated ovarian cancer, while serous carcinoma was less frequent (Br J Cancer. 2014 Apr 2;110[7]:1878-90).
Findings of both of these large studies have served to clarify the association between endometriosis and specific histologic subtypes and suggested that there are important differences in the pathogenesis of low-grade and high-grade serous ovarian carcinomas.
Clinical implications
It is not clear what causes malignant transformation or what predisposes some patients with endometriosis to develop ovarian cancer, but the risk likely involves genetic and epigenetic influences as well as immunologic, inflammatory, and hormonal factors.
The molecular profiles of the main two types of ovarian cancer are different: While the majority of high-grade serous ovarian tumors are characterized by TP53 mutations, the low-grade carcinomas are characterized by a variety of mutations, including KRAS, BRAF, ERBB2, CTNNB1, and BCL2 mutations.
There currently are not enough data to recommend genetic screening tests in patients with endometriosis, but our hope is that we eventually will be able to screen for “high-risk” endometriotic lesions by testing for genes specific to various histologic subtypes of low-grade ovarian cancer, or by finding and utilizing other biomarkers.
In the meantime, we believe it is important to more thoroughly treat endometriosis and to identify and follow women with a history of the condition, especially those with a long-standing history, those with a history of endometriosis associated with infertility, and those with ovarian endometrioma. Each of these factors predisposes patients to a higher risk of malignant transformation.
Complete surgical resection of all visible endometriosis is the most effective treatment and will afford the best cancer prevention, even in women who are asymptomatic. In a recent Swedish national registry case-control study, women who underwent radical surgical excision of all visible endometriosis were significantly less likely (OR, 0.30) to develop ovarian cancer (Acta Obstet Gynecol Scand. 2013 May;92[5]:546-54).
Suppressive hormonal therapy is another treatment option for patients with no interest in conceiving. Most large endometriomas are functional ovarian cysts that have been invaded by cortical ovarian endometriosis or by small primary endometriomas (J Reprod Med. 1992 Sep;37[9]:771-6).
While hormonal therapy will not always result in complete regression of endometriotic lesions, it will decrease the recurrence rate of endometriomas and can be considered for long-term prevention of potentially premalignant lesions. It is most effective when it follows surgical excision of endometriomas and associated endometriosis.
A patient who has completed childbearing at the time of surgical resection may be offered bilateral salpingectomy, regardless of menopausal status. Salpingectomy in both average and high-risk populations (e.g., BRCA 1/2 carriers) not only prevents high-grade serous carcinoma by eliminating the site of origin, but also may decrease the risk of endometrioid and clear cell carcinoma by blocking the passageway that enables the flow of endometrium and factors that induce inflammation. It is estimated that the procedure reduces the risk of ovarian cancer by 40%.
Interestingly, tubal ligation has historically been shown to decrease the risk of ovarian cancer, and recent data have shown that the risk of endometrioid and clear cell carcinoma is cut even more than the risk of high-grade serous carcinoma (Int J Epidemiol. 2013 Apr;42[2]:579-89).
The Society of Gynecologic Oncology recommends that risk-reducing salpingectomy be considered at the time of hysterectomy or other abdominal or pelvic surgery, and in lieu of tubal ligation. The American College of Obstetricians and Gynecologists similarly has stated that prophylactic salpingectomy may offer clinicians the opportunity to prevent ovarian cancer in their patients. Salpingectomy is an important option for all patients, but is especially important when the fallopian tubes are found to be damaged by endometriosis and/or pelvic inflammatory disease. When imaging studies show that endometriomas are present and resection is not performed, pelvic ultrasound should become part of the patient’s routine examination.
Most endometriomas have a homogeneous appearance; any significant increase in size or a change in the homogeneous cystic characteristics to a more heterogeneous appearance with mural components should raise suspicion about malignant change.
It can be difficult to detect relatively small endocystic components with ultrasound, so if there is any doubt about whether there is some heterogeneous consistency, an MRI should be performed. MRI is showing more promise in detecting malignant change. Hyperdense mural nodules within the ovary and rapid growth of an endometrioma have both been associated with malignant transformation and can be seen on these images.
In a cohort study comparing MRI findings of 10 patients with ovarian adenocarcinoma to 10 patients with benign endometriomas, investigators found mural nodules in all 10 malignancies but in only three of the benign cases (AJR Am J Roentgenol. 2000 Nov;175[5]:1423-30).
Long-term follow-up is necessary to understand the timeline of transformation in patients with mural nodules. This together with increasing knowledge of molecular events underpinning evolution of endometriosis will lead to better screening and preventive strategies.
Dr. Nezhat is the director of minimally invasive gynecologic surgery and robotics at Winthrop University Hospital in Mineola, N.Y., and an adjunct professor of obstetrics, gynecology, and reproductive medicine at the State University of New York at Stony Brook. He reported having no financial disclosures.
Averting complications of laparoscopy: Pearls from 5 patients
To view three clips of surgical pearls for laparoscopy, visit the Video Library.
<huc>Q.</huc>What is the only surgical procedure that is completely safe?
<huc>A.</huc>The surgical procedure that is not performed.
The unfortunate truth is that complications can occur during any operative procedure, despite our best efforts—and laparoscopy is no exception. Being vigilant for iatrogenic injuries, both during and after surgery, and ensuring that repairs are both thorough and timely, are two of our best weapons against major complications, along with meticulous technique and adequate experience.
This article features five cases that illustrate some of the most serious complications of laparoscopy—and how to prevent and manage them.
CASE 1: Surgical patient returns with signs of ureteral injury
A 42-year-old woman with a history of endometriosis undergoes laparoscopic hysterectomy and bilateral salpingo-oophorectomy. She is discharged 2 days later. Two days after that, she returns to the hospital complaining of fluid leaking from the vagina. She has no fever or any other significant complaint or physical findings other than abdominal tenderness, which is to be expected after surgery. A computed tomography (CT) scan with intravenous (IV) contrast reveals left ureteral obstruction near the bladder, with extravasation of contrast media into the abdominal cavity. Further investigation reveals a left ureteral transection.
Could this injury have been avoided? How should it be managed?
Postoperative diagnosis of ureteral injury can be challenging, in part because up to 50% of unilateral cases are asymptomatic. Be on the lookout for this complication in women who have undergone pelvic sidewall dissection or laparoscopic hysterectomy, such as the patient in the case just described. As the number of laparoscopic hysterectomies and retroperitoneal procedures has risen in recent years, so has the rate of ureteral injury, with an incidence of 0.3% to 2%.1,2
Ureteral injury can be caused by ligation, ischemia, resection, transection, crushing, or angulation. Three sites are particularly troublesome: the infundibulopelvic ligament, ovarian fossa, and ureteral tunnel.3,4 In Case 1, injury to the ureter was proximal to the bladder and probably occurred during transection of the uterosacral cardinal ligament complex.
What’s the best preventive strategy?
Meticulous technique is imperative to protect the ureters. This includes adequate visualization, intraperitoneal or retroperitoneal dissection, and early identification of the ureter. In a high-risk patient likely to have distorted anatomy due to severe endometriosis and fibrosis, retroperitoneal dissection of any adhesions or tumor and identification of the ureter are the best ways to avoid injury.
Intraperitoneal identification and dissection of the ureters can be enhanced by hydrodissection and resection of the affected peritoneum.3,4 To create a safe operating plane, make a small opening in the peritoneum below the ureter and inject 50 to 100 mL of lactated Ringer’s solution along the course of the ureter, which will displace it laterally.5
Although neither IV indigo carmine nor ureteral catheterization has been shown to reduce the risk of ureteral injury or identify ligation or thermal injury,3,6 both can help the surgeon identify intraoperative perforation of the ureter. Liberal use of cystoscopy with indigo carmine administration for identification of ureteral flow and ureteral catheterization can be used in potentially high-risk patients. If there is suspicion for devascularization or thermal injury, use prophylactic ureteral stents postoperatively for 2 to 4 weeks.
Don’t hesitate to consult a urologist
In Case 1, the surgeon sought immediate urologic consultation and the patient underwent laparotomy with ureteroneocystotomy without sequelae.
In general, management of ureteral injury depends on its severity and location, as well as the comfort level of the surgeon. Minor injuries are sometimes managed with cystoscopic stent placement, but more severe cases may require operative ureteral repair.
In cases like this one, where ureteral injury occurred in close proximity to the bladder, a ureteroneocystotomy is possible. However, in more cephalad injuries, there may be insufficient residual ureter to allow such a repair. In these cases, a Boari flap may be attempted to use bladder tissue to bridge the gap to the ureteral edge. Rarely, in high ureteral injuries, trans-ureteroureterostomy may be appropriate. This procedure carries the greatest risk, given that both kidneys are reliant on one ureter.
Is laparoscopic repair reasonable?
When surgical intervention is necessary, the choice between laparoscopy and laparotomy depends on the skill and comfort level of the surgeon and the availability of instruments and support team.6,7 That said, ureteral injury is usually treated via laparotomy.1 As operative laparoscopy becomes even more commonplace, reconstruction of the urinary system will increasingly be managed laparoscopically.
Depending on the size and location of the injury, reconstruction may involve ureteral reimplantation with or without a psoas hitch, Boari flap, or primary endtoend anastomosis.8-10
CASE 2: Postoperative symptoms lead to rehospitalization
A 35-year-old patient undergoes laparoscopic ovarian cystectomy and returns home the same day. She is readmitted 72 hours later because of lower abdominal tenderness, worsening nausea and vomiting, and urine-like drainage from her midline suprapubic trocar site. Analysis of the leaking fluid shows high creatinine levels consistent with urine. The patient has no fever and is hemodynamically stable. Examination reveals a moderately distended abdomen with decreased bowel sounds. Hematuria is evident on urine analysis.
Urologic consultation is obtained, and the patient undergoes simultaneous laparoscopy and cystoscopy, during which perforation of the bladder dome is discovered, apparently caused by the mid suprapubic trocar. The bladder is mobilized anteriorly, and both anterior and posterior aspects of the perforation are repaired in one layer laparoscopically.
After continuous drainage with a transurethral Foley catheter for 7 days, cystography shows complete healing of the bladder, and the Foley catheter is removed. The patient recovers completely.
Vesical injury sometimes occurs in patients who have a history of laparotomy, a full bladder at the time of surgery, or displaced anatomy due to pelvic adhesions.11 Although bladder injury is rare, laparoscopy increases the risk. Trocars, uterine manipulators, and blunt instruments can perforate or lacerate the bladder, and energy devices can cause thermal injury. The risk of bladder injury increases during laparoscopic hysterectomy.
Be vigilant about trocar placement and dissection techniques
Accessory trocars can injure a full bladder. Injury can also occur when distorted anatomy from a previous pelvic operation obscures bladder boundaries, making insertion of the midline trocar potentially perilous (FIGURE 1). The Veress needle and Rubin’s cannula can perforate the bladder.11-13 And in the anterior cul-de-sac, adhesiolysis, deep coagulation, laser ablation, or sharp excision of endometriosis implants can predispose a patient to bladder injury.
In women with severe endometriosis, lower-segment myoma, or a history of cesarean section, the bladder is vulnerable to laceration when blunt dissection is used during laparoscopic hysterectomy or laparoscopically assisted vaginal hysterectomy (LAVH). A vesical injury also can occur at the time of laparoscopic bladder-neck suspension upon entry into, and dissection of, the space of Retzius.
FIGURE 1 A bladder at risk
In this patient with a previous cesarean section, the bladder is adherent to the anterior abdominal wall. Needle mapping in the conventional midline trocar position indicates that the trocar must be relocated to avoid bladder injury.
Intraoperative findings that suggest bladder injury include air in the urinary catheter, hematuria, trocar site drainage of urine, or indigo carmine leakage. Postoperative signs and symptoms include leaking from incisional sites, a mass in the abdominal wall, and abdominal swelling.
Liberal use of cystoscopy or distension of the bladder with 300 to 500 mL of normal saline is recommended whenever there is a suspicion of bladder injury, especially during laparoscopic hysterectomy or LAVH. When a trocar causes the injury, look for both entry and exit punctures, both of which should be treated.
No matter how much care is taken, some bladder injuries, such as vesicovaginal fistulae, become apparent only postoperatively. More rarely, peritonitis or pseudoascites herald the injury. Retrograde cystography may aid identification.
Treatment of bladder injuries
Small perforations recognized intraoperatively may be conservatively managed by postoperative bladder drainage for 5 to 7 days. Most other bladder injuries require prompt intervention. For example, trocar injury to the bladder dome requires one- or two-layer closure followed by 5 to 7 days of urinary drainage. (Both closing and healing are promoted by drainage.)
Laparoscopy or laparotomy? Laparoscopic repair has become increasingly common, and bladder injury is a common complication of LAVH.13,14
CASE 3: Postop pain, tachycardia
A 41-year-old obese woman undergoes laparoscopic cystectomy for an 8-cm left ovarian mass. The abdomen is entered on the second attempt with a long Veress needle. The umbilical trocar is reinserted “several” times because of difficulty opening the peritoneum with the tip of the trocar sheath. The surgical procedure is completed within 2 hours, and the patient is discharged 23 hours later.
The next day, she experiences increasing abdominal pain and presents to the emergency room. Upon admission she reports intermittent chills, but denies nausea and vomiting. She is in mild distress, pale and tachycardic, with a temperature of 96.4°, pulse of 117, respiration rate of 20, blood pressure of 106/64 mm Hg, and oxygen saturation of 92%. She also has a diffusely tender abdomen but normal blood work. Abdominal and chest x-rays show a large right subphrenic air-fluid level that is consistent with free intraperitoneal air, unsurprising given her recent surgery. Bibasilar atelectasis and consolidation are noted on the initial chest x-ray.
During observation over the next 2 days, she remains afebrile and tachycardic, but her shortness of breath becomes progressively worse. Neither spiral CT nor lower-extremity Doppler suggests pulmonary embolism or deep venous thrombosis. Supplemental oxygen, aggressive pain management, albuterol, ipratropium, and acetylcysteine are initiated after pulmonary consultation.
The patient tolerates a regular diet on postoperative day 3 and has a bowel movement on day 5. However, the same day she begins vomiting and reports worsening abdominal pain. CT imaging of the abdomen and pelvis reveals free air in the abdomen and loculated fluid with air bubbles suspicious for intra-abdominal infection and perforated bowel.
Exploratory laparotomy reveals diffuse feculent peritonitis, as well as food particles and contrast media. There is a perforation in the antimesenteric side of the ileum approximately 1.5 feet proximal to the ileocecal valve. This perforation measures approximately 1 cm in diameter and is freely spilling intestinal contents. Small bowel resection is performed to treat the perforation.
Following the surgery, the patient recovers slowly.
Could the bowel perforation have been detected sooner?
Intestinal tract injury is a serious complication, particularly with postoperative diagnosis.15 Damage can occur during insertion of the Veress needle or trocar when the bowel is immobilized by adhesions, or during enterolysis.16 Unrecognized thermal injury can cause delayed bowel injury.
Small-bowel damage often occurs during uncontrolled insertion of the Veress needle or primary umbilical trocar. It also may result from sharp dissection or thermal injury.17,18 Abrasions and lacerations can occur if traction is exerted on the bowel using serrated graspers. When adhesions are dense and tissue planes poorly defined, the risk of laceration due to energy sources or sharp dissection increases.
Be cautious during bowel manipulation. Avoid blunt dissection. Be especially careful when the small bowel is adherent to the anterior abdominal wall (FIGURE 2A), particularly during evaluation of patients with a history of bowel resection, exploratory laparotomy for trauma-related peritonitis, or tumor debulking.
Remove the primary and ancillary cannulas under direct visualization with the laparoscope to prevent formation of a vacuum that can draw bowel into the incision and cause herniation.19
FIGURE 2 Adherent bowel, minor bleeding
A: Veress needle pressure measurements are persistently elevated before primary trocar insertion in this patient, raising the suspicion of adhesive disease from earlier surgery. As a result, the primary trocar is relocated to the left upper quadrant. Inspection confirms that small bowel is adherent to the anterior abdominal wall.
FIGURE 2 Adherent bowel, minor bleeding
B: After the small-bowel adhesions are dissected off the anterior abdominal wall via laparoscopy, a small hematoma is discovered, likely caused by the Veress needle. The patient is managed conservatively and recovers.
The value of open laparoscopy
In open laparoscopy, an abdominal incision is made into the peritoneal cavity so that the trocar can be placed under direct vision, after which the abdomen is insufflated. This approach can prevent bowel injury only when the adhesions and attachments are to the anterior abdominal wall and away from the entry site. When the attachment lies directly beneath the umbilicus, however, open laparoscopy is no guarantee against injury.
When bowel adhesions are severe, use alternative trocar sites such as the left upper quadrant (Palmer’s point) for the Veress needle and primary trocars.5,20,21
The likelihood of perforation can be reduced with preoperative bowel prep when there is a risk of bowel adhesions.
Identifying bowel injury
We recommend routine inspection of the structures beneath the primary trocar upon insertion of the laparoscope to look for injury to the bowel, mesentery, or vascular structures. If adhesions are found, evaluate the area carefully to rule out injury to the bowel or omentum. It may be necessary to change the position of the laparoscope to assess the patient.
Trauma to the intestinal tract can be mechanical or electrical in nature, and each type of trauma creates a distinctive, characteristic pattern. Thermal injury can be subtle and present as simple blanching or a distinct burn and charring. A small hole or obvious tear in the bowel wall can be the result of mechanical injury.22
Benign-appearing, superficial thermal bowel injuries may be managed conservatively.22 Minimal serosal burns (smaller than 5 mm in diameter) can be managed expectantly. Immediate surgical intervention is needed if the area of blanching on the intestinal serosa exceeds 5 mm in diameter or if the burn appears to involve more than the serosa.23
Small-bowel injuries that escape notice intraoperatively generally become apparent 2 to 4 days later, when the patient develops fever, nausea, lower abdominal pain, and anorexia. On postoperative day 5 or 6, the white blood cell (WBC) count rises and earlier symptoms may become worse. Radiography may reveal multiple air and fluid levels—another sign of bowel injury. Be aware that if the patient has clinical symptoms of gastrointestinal injury, even if the WBC count is normal, exploratory laparoscopy or laparotomy is necessary for accurate diagnosis.
Intraoperatively discovered injury
Careful inspection may reveal no leakage or bleeding in the affected area. Small punctures or superficial lacerations seal readily and may not require further treatment (FIGURE 2B), but larger perforations require repair. Straightforward repair is not always possible when the injury is extensive and considerable time has elapsed before it is discovered.
Inspect the intestine thoroughly at the conclusion of a procedure; obvious leakage requires intervention. Repair the small intestine in one or two layers, using the initial row of interrupted sutures to approximate the mucosa and muscularis.24 To lessen the risk of stenosis, close all lacerations transversely when they are smaller than one half the diameter of the bowel. If the laceration exceeds that size, segmental resection and anastomosis are necessary. Resection is prudent if the mesenteric blood supply is compromised.25
When performing one-layer repair of the small bowel, delayed absorbable suture (eg, Vicryl or PDS) or nonabsorbable suture (eg, silk) is recommended.26
At the conclusion of a repair, copiously irrigate the entire abdomen. Place a nasogastric tube only if ileus is anticipated; the tube can be removed when drainage diminishes and active bowel sounds and flatus appear. Do not give anything by mouth until the patient has return of bowel function and active peristalsis. Prescribe prophylactic antibiotics.
Note that peritonitis sometimes develops after repair of the bowel.25 This can be managed with prolonged bowel rest and peripheral or total parenteral nutrition.
Conservative management may be possible
Patients whose symptoms of bowel laceration become apparent after discharge can sometimes be managed conservatively. More than 50% of patients treated conservatively require no surgery.23 Inpatient management consists of monitoring the WBC count, providing hydration and IV antibiotics, and examining the patient every 6 hours, giving nothing by mouth.
When injury is discovered later
If conservative management with observation and bowel rest fails, or the patient complains of severe abdominal pain, vomiting, nausea, obstipation, or signs and symptoms of peritonitis, such as the patient in Case 3, immediate surgical intervention is necessary. When an injury is not detected until some time after initial surgery, resection of all necrotic tissue is mandatory. In most cases, the perforation is managed by segmental resection and reanastomosis. Evaluate the entire small and large bowel to rule out any other injury, and irrigate generously. Bowel rest, parenteral nutrition, and IV antibiotics also are indicated.
Of 36,928 procedures reported by members of the American Association of Gynecologic Laparoscopists, there were two deaths—both caused by unrecognized bowel injury.15
CASE 4: Large-bowel injury precipitates lengthy recovery
A surgeon performs a left laparoscopic salpingo-oophorectomy to remove an 8-cm ovarian endometrioma that is adherent to the rectosigmoid colon of a 40-year-old diabetic woman. Sharp and electrosurgical scissors are used to separate the adnexa from the rectosigmoid colon. No injury is observed, and she is discharged the same day. Four days later, she returns with severe abdominal pain, nausea, vomiting, and fatigue. Lab tests reveal a WBC count of 17,000; a CT scan shows pockets of air beneath the diaphragm, as well as fluid collection suggestive of a pelvic abscess.
Immediate laparotomy is performed, during which the surgeon discovers contamination of the abdominal viscera by bowel contents, as well as a 0.5-cm perforation of the rectosigmoid colon. The perforation is repaired in two layers after its edges are trimmed, and a diverting colostomy is performed. The patient is admitted to the ICU and requires antibiotic treatment, total parenteral nutrition, and bowel rest due to severe peritonitis. She gradually recovers and is discharged 3 weeks later. The diverting colostomy is reversed 3 months later.
Even small perforations in the large bowel can cause infection and abscess due to the high bacterial content of the colon. The most common cause of injury to the rectosigmoid colon is pelvic adhesiolysis during cul-de-sac dissection, treatment of pelvic endometriosis, and resection of adherent pelvic masses.
Sharp dissection with scissors or high-powered lasers is relatively safe near the bowel. When dissecting the cul-de-sac, identify the vagina and rectum by placing a probe or finger in each area. Begin dissection from the unaffected pararectal space, and proceed toward the obliterated cul-de-sac.27,28
Bowel prep is indicated before extensive pelvic surgery and when the history suggests endometriosis or significant pelvic adhesions. Some general surgeons base their decision to perform colostomy (or not) on whether the bowel was prepped preoperatively.29
If the large bowel is perforated by the Veress needle, the saline aspiration test will yield brownish fluid. When significant pelvic adhesiolysis or pelvic or endometriotic tumor resection is performed, inject air into the rectum afterward via a sigmoidoscope or bulb syringe and assess the submerged rectum and rectosigmoid colon for bubbling. The rectal wall may be weakened during these types of procedures, so instruct the patient to use oral stool softeners and avoid enemas.30
Delay in detection can have serious ramifications
When a large-bowel injury goes undetected at the time of operation, the patient generally presents on the third or fourth postoperative day with mild fever, occasionally sudden sharp epigastric pain, lower abdominal pain, slight nausea, and anorexia. By the fifth or sixth day, these symptoms have become more severe and are accompanied by peritonitis and an elevated WBC count.
Whenever a patient complains of abdominal pain and a deteriorating condition, assume that bowel injury is the cause until it is proved otherwise.
Intraoperative management
Repair small trocar wounds using primary suture closure. Copious lavage of the peritoneal cavity, drainage, and a broad-spectrum antibiotic minimize the risk of infection. Manage deep electrical injury to the right colon by resecting the injured segment and performing primary anastomosis. Primary closure or resection and reanastomosis may not be adequate when the vascular supply of the descending colon or rectum is compromised. In that case, perform a diverting colostomy or ileostomy, which can be reversed 6 to 12 weeks later.25,26
CASE 5: Vascular injury
A tall, thin, athletic 19-year-old undergoes diagnostic laparoscopy to rule out pelvic pathology after she complains of severe, monthly abdominal pain. Upon insertion of the laparoscope, the surgeon observes a large hematoma forming at the right pelvic sidewall. At the same time, the anesthesiologist reports a significant drop in blood pressure, and vascular injury is diagnosed. The surgeon attempts to control the bleeding using bipolar coagulation, but the problem only becomes worse. He decides to switch to laparotomy.
A vascular surgeon is called in, and injury to the right common iliac artery and vein—apparently caused during insertion of the primary umbilical trocar—is repaired. The patient is given 5 U of red blood cells. She goes home 10 days later, but returns with thrombophlebitis and rejection of the graft. After several surgeries, she finally recovers, with some sequelae, such as unilateral leg edema.
Management of vascular injury depends on the source and type of injury. On major vessels, electrocoagulation is contraindicated. After immediate atraumatic compression with tamponade to control bleeding, vascular repair, in consultation with a vascular surgeon, is indicated. At times, a vascular graft may be required.
Smaller vessels, such as the infundibulopelvic ligament or uterine vessels, can be managed by clips, suture, or loop ligatures. If thermal energy is used in the repair, be careful to avoid injury to surrounding structures.
Most emergency laparotomies are performed for uncontrolled bleeding.30,31 Lack of control or a wrong angle at insertion of the Veress needle and trocars is a major cause of large-vessel injury. Sharp dissection of adhesions, uterosacral ablation, transection of vascular pedicles without adequate dessication, and rough handling of tissues can all cause bleeding. Distorted anatomy is a main cause of vascular injury and can compound injury in areas more prone to bleeding, such as the oviduct, infundibulopelvic ligament, mesosalpinx, and pelvic sidewall vessels.
The return of pressure gradients to normal levels at the end of a procedure can be accompanied by bleeding into the retroperitoneal space, so evaluate the patient in a supine position after intra-abdominal pressure is reduced.
A vascular surgeon may be required
Depending on the type of vessel, size and location of the injury, and degree of bleeding, you may use unipolar or bipolar electrocoagulation, suture, clips, vasopressin, or loop ligatures to control bleeding. Although diluted vasopressin (10 U in 60 mL of lactated Ringer’s saline) can decrease oozing from raw peritoneal areas, injury to a major vessel, such as the iliac vessels, vena cava, or aorta, needs immediate control and proper repair. The decision to perform laparoscopy or laparotomy depends on your preference and experience. In any case, a vascular surgeon may be consulted for major vascular injuries.32
If a major vessel is injured, do not crush-clamp it. If possible (and if your laparoscopic skills are advanced), insert a sponge via a 10-mm trocar and apply pressure to the vessel to minimize bleeding and enhance visualization. The decision to repair the injury laparoscopically or by laparotomy should be made judiciously and promptly. n
The authors acknowledge the editorial contributions of Kristina Petrasek and Barbara Page, of the University of California, Berkeley, to the manuscript of this article.
1. Ostrzenski A, Radolinski B, Ostrzenska K. A review of laparoscopic ureteral injury in pelvic surgery. Obstet Gynecol Surv. 2003;58:794-799.
2. Kabalin J. Chapter 1—Surgical anatomy of the retroperitoneum, kidneys, and ureters. In: Walsh P, Retik A, Wein A, eds. Campbell’s Urology. 8th ed. Philadelphia: Saunders; 2002 36-40.
3. Chan J, Morrow J, Manetta A. Prevention of ureteral injuries in gynecologic surgery. Am J Obstet Gynecol. 2003;188:1273-1277.
4. Grainger DA, Soderstrom RM, Schiff SF, Glickman MG, DeCherney AH, Diamond MP. Ureteral injuries at laparoscopy: insights into diagnosis, management, and prevention. Obstet Gynecol. 1990;75:839-843.
5. Nezhat C. Chapter 20. Operative Gynecologic Laparoscopy: Principles and Techniques. 2nd ed. New York: McGraw–Hill; 2000.
6. Ou CS, Huang IA, Rowbotham R. Laparoscopic ureteroureteral anastomosis for repair of ureteral injury involving stricture. Int Urogynecol J Pelvic Floor Dysfunct. 2005;16:155-157.
7. Modi P, Goel R, Dodiya S. Laparoscopic ureteroneocystostomy for distal ureteral injuries. Urology. 2005;66:751-753.
8. Nezhat C, Nezhat F, Nezhat CH, et al. Urinary tract endometriosis treated by laparoscopy. Fertil Steril. 1996;66:920-924.
9. Nezhat C, Nezhat F. Laparoscopic repair of ureter resected during operative laparoscopy. Obstet Gynecol. 1992;80:543-544.
10. Nezhat CH, Nezhat FR, Freiha F, Nezhat CR. Laparoscopic vesicopsoas hitch for infiltrative ureteral endometriosis. Fertil Steril. 1999;71:376-379.
11. Georgy FM, Fettman HH, Chefetz MD. Complications of laparoscopy: two cases of perforated urinary bladder. Am J Obstet Gynecol. 1974;120:1121-1122.
12. Sherer DM. Inadvertent transvaginal cystotomy during laparoscopy. Int J Gynaecol Obstet. 1990;32:77-79.
13. Nezhat CH, Seidman DS, Nezhat F, et al. Laparoscopic management of internal and unintentional cystotomy. J Urol. 1996;156:1400-1402.
14. Lee CL, Lai YM, Soong YK. Management of urinary bladder injuries in laparoscopic assisted vaginal hysterectomy. Acta Obstet Gynecol Scand. 1996;75:174-177.
15. Peterson HB, et al. American Association of Gynecologic Laparoscopists’ 1988 membership survey on operative laparoscopy. J Reprod Med. 1990;35:587-589.
16. Phillips JM, Hulka JF, Hulka B, et al. 1978 AAGL membership survey. J Reprod Med. 1981;26:529-533.
17. Chapron C, Pierre F, et al. Gastrointestinal injuries during laparoscopy. Hum Reprod. 1999;14:333-337.
18. Schrenk P, Woisetschlager R, Rieger R, et al. Mechanism, management, and prevention of laparoscopic bowel injuries. Gastrointest Endosc. 1996;43:572-574.
19. Sauer M, Jarrett JC. Small bowel obstruction following diagnostic laparoscopy. Fertil Steril. 1984;42:653-654.
20. Penfield AJ. How to prevent complications of open laparoscopy. J Reprod Med. 1985;30:660-663.
21. Brill A, Nezhat F, Nezhat CH, Nezhat C. The incidence of adhesions after prior laparatomy: a laparoscopic appraisal. Obstet Gynecol. 1995;85:269-272.
22. Levy BS, Soderstrom RM, Dail DH. Bowel injuries during laparoscopy: gross anatomy and histology. J Reprod Med. 1985;30:168-172.
23. Wheeless CR. Gastrointestinal injuries associated with laparoscopy. In: Phillips JM, ed. Endoscopy in Gynecology. Santa Fe Springs, Calif: American Association of Gynecologic Laparoscopists; 1978.
24. Borton M. Laparoscopic Complications: Prevention and Management. Philadelphia: Decker; 1986.
25. DeCherney AH. Laparoscopy with unexpected viscus penetration. In: Nichols DH, ed. Clinical Problems, Injuries, and Complications of Gynecologic Surgery. Baltimore: Williams & Wilkins; 1988.
26. Nezhat C, Nezhat F, Ambroze W, Pennington E. Laparoscopic repair of small bowel and colon: a report of 26 cases. Surg Endosc. 1993;7:88-89.
27. Redwine D. Laparoscopic en bloc resection for treatment of the obliterated cul-de-sac in endometriosis. J Reprod Med. 1992;37:696-698.
28. Nezhat C, Nezhat F, et al. Laparoscopic treatment of infiltrative rectosigmoid colon and rectovaginal septum endometriosis by the technique of videolaseroscopy and the CO2 laser. Br J Obstet Gynaecol. 1992;99:664-667.
29. Nezhat C, Seidman D, Nezhat F, et al. The role of intraoperative proctosigmoidoscopy in laparoscopic pelvic surgery. J Am Assoc Gynecol Laparosc. 2004;11:47-49.
30. Chapron CM, et al. Major vascular injuries during gynecologic laparoscopy. J Am Coll Surg. 1997;185:461-465.
31. Geers J, Holden C. Major vascular injury as a complication of laparoscopic surgery: a report of three cases and review of the literature. Am Surg. 1996;62:377-379.
32. Nezhat C, Childers J, Nezhat F, et al. Major retroperitoneal vascular injury during laparoscopic surgery. Hum Reprod. 1997;12:480-483.
Dr. Farr Nezhat reports no relevant financial relationships. Dr. Ceana Nezhat is a speaker and consultant for Karl Storz, Johnson & Johnson, Valleylab, US Surgical, and Viking. Dr. Camran Nezhat is a speaker for or receives educational support from Karl Storz, Stryker, Johnson & Johnson, Valleylab, and Baxter.
To view three clips of surgical pearls for laparoscopy, visit the Video Library.
<huc>Q.</huc>What is the only surgical procedure that is completely safe?
<huc>A.</huc>The surgical procedure that is not performed.
The unfortunate truth is that complications can occur during any operative procedure, despite our best efforts—and laparoscopy is no exception. Being vigilant for iatrogenic injuries, both during and after surgery, and ensuring that repairs are both thorough and timely, are two of our best weapons against major complications, along with meticulous technique and adequate experience.
This article features five cases that illustrate some of the most serious complications of laparoscopy—and how to prevent and manage them.
CASE 1: Surgical patient returns with signs of ureteral injury
A 42-year-old woman with a history of endometriosis undergoes laparoscopic hysterectomy and bilateral salpingo-oophorectomy. She is discharged 2 days later. Two days after that, she returns to the hospital complaining of fluid leaking from the vagina. She has no fever or any other significant complaint or physical findings other than abdominal tenderness, which is to be expected after surgery. A computed tomography (CT) scan with intravenous (IV) contrast reveals left ureteral obstruction near the bladder, with extravasation of contrast media into the abdominal cavity. Further investigation reveals a left ureteral transection.
Could this injury have been avoided? How should it be managed?
Postoperative diagnosis of ureteral injury can be challenging, in part because up to 50% of unilateral cases are asymptomatic. Be on the lookout for this complication in women who have undergone pelvic sidewall dissection or laparoscopic hysterectomy, such as the patient in the case just described. As the number of laparoscopic hysterectomies and retroperitoneal procedures has risen in recent years, so has the rate of ureteral injury, with an incidence of 0.3% to 2%.1,2
Ureteral injury can be caused by ligation, ischemia, resection, transection, crushing, or angulation. Three sites are particularly troublesome: the infundibulopelvic ligament, ovarian fossa, and ureteral tunnel.3,4 In Case 1, injury to the ureter was proximal to the bladder and probably occurred during transection of the uterosacral cardinal ligament complex.
What’s the best preventive strategy?
Meticulous technique is imperative to protect the ureters. This includes adequate visualization, intraperitoneal or retroperitoneal dissection, and early identification of the ureter. In a high-risk patient likely to have distorted anatomy due to severe endometriosis and fibrosis, retroperitoneal dissection of any adhesions or tumor and identification of the ureter are the best ways to avoid injury.
Intraperitoneal identification and dissection of the ureters can be enhanced by hydrodissection and resection of the affected peritoneum.3,4 To create a safe operating plane, make a small opening in the peritoneum below the ureter and inject 50 to 100 mL of lactated Ringer’s solution along the course of the ureter, which will displace it laterally.5
Although neither IV indigo carmine nor ureteral catheterization has been shown to reduce the risk of ureteral injury or identify ligation or thermal injury,3,6 both can help the surgeon identify intraoperative perforation of the ureter. Liberal use of cystoscopy with indigo carmine administration for identification of ureteral flow and ureteral catheterization can be used in potentially high-risk patients. If there is suspicion for devascularization or thermal injury, use prophylactic ureteral stents postoperatively for 2 to 4 weeks.
Don’t hesitate to consult a urologist
In Case 1, the surgeon sought immediate urologic consultation and the patient underwent laparotomy with ureteroneocystotomy without sequelae.
In general, management of ureteral injury depends on its severity and location, as well as the comfort level of the surgeon. Minor injuries are sometimes managed with cystoscopic stent placement, but more severe cases may require operative ureteral repair.
In cases like this one, where ureteral injury occurred in close proximity to the bladder, a ureteroneocystotomy is possible. However, in more cephalad injuries, there may be insufficient residual ureter to allow such a repair. In these cases, a Boari flap may be attempted to use bladder tissue to bridge the gap to the ureteral edge. Rarely, in high ureteral injuries, trans-ureteroureterostomy may be appropriate. This procedure carries the greatest risk, given that both kidneys are reliant on one ureter.
Is laparoscopic repair reasonable?
When surgical intervention is necessary, the choice between laparoscopy and laparotomy depends on the skill and comfort level of the surgeon and the availability of instruments and support team.6,7 That said, ureteral injury is usually treated via laparotomy.1 As operative laparoscopy becomes even more commonplace, reconstruction of the urinary system will increasingly be managed laparoscopically.
Depending on the size and location of the injury, reconstruction may involve ureteral reimplantation with or without a psoas hitch, Boari flap, or primary endtoend anastomosis.8-10
CASE 2: Postoperative symptoms lead to rehospitalization
A 35-year-old patient undergoes laparoscopic ovarian cystectomy and returns home the same day. She is readmitted 72 hours later because of lower abdominal tenderness, worsening nausea and vomiting, and urine-like drainage from her midline suprapubic trocar site. Analysis of the leaking fluid shows high creatinine levels consistent with urine. The patient has no fever and is hemodynamically stable. Examination reveals a moderately distended abdomen with decreased bowel sounds. Hematuria is evident on urine analysis.
Urologic consultation is obtained, and the patient undergoes simultaneous laparoscopy and cystoscopy, during which perforation of the bladder dome is discovered, apparently caused by the mid suprapubic trocar. The bladder is mobilized anteriorly, and both anterior and posterior aspects of the perforation are repaired in one layer laparoscopically.
After continuous drainage with a transurethral Foley catheter for 7 days, cystography shows complete healing of the bladder, and the Foley catheter is removed. The patient recovers completely.
Vesical injury sometimes occurs in patients who have a history of laparotomy, a full bladder at the time of surgery, or displaced anatomy due to pelvic adhesions.11 Although bladder injury is rare, laparoscopy increases the risk. Trocars, uterine manipulators, and blunt instruments can perforate or lacerate the bladder, and energy devices can cause thermal injury. The risk of bladder injury increases during laparoscopic hysterectomy.
Be vigilant about trocar placement and dissection techniques
Accessory trocars can injure a full bladder. Injury can also occur when distorted anatomy from a previous pelvic operation obscures bladder boundaries, making insertion of the midline trocar potentially perilous (FIGURE 1). The Veress needle and Rubin’s cannula can perforate the bladder.11-13 And in the anterior cul-de-sac, adhesiolysis, deep coagulation, laser ablation, or sharp excision of endometriosis implants can predispose a patient to bladder injury.
In women with severe endometriosis, lower-segment myoma, or a history of cesarean section, the bladder is vulnerable to laceration when blunt dissection is used during laparoscopic hysterectomy or laparoscopically assisted vaginal hysterectomy (LAVH). A vesical injury also can occur at the time of laparoscopic bladder-neck suspension upon entry into, and dissection of, the space of Retzius.
FIGURE 1 A bladder at risk
In this patient with a previous cesarean section, the bladder is adherent to the anterior abdominal wall. Needle mapping in the conventional midline trocar position indicates that the trocar must be relocated to avoid bladder injury.
Intraoperative findings that suggest bladder injury include air in the urinary catheter, hematuria, trocar site drainage of urine, or indigo carmine leakage. Postoperative signs and symptoms include leaking from incisional sites, a mass in the abdominal wall, and abdominal swelling.
Liberal use of cystoscopy or distension of the bladder with 300 to 500 mL of normal saline is recommended whenever there is a suspicion of bladder injury, especially during laparoscopic hysterectomy or LAVH. When a trocar causes the injury, look for both entry and exit punctures, both of which should be treated.
No matter how much care is taken, some bladder injuries, such as vesicovaginal fistulae, become apparent only postoperatively. More rarely, peritonitis or pseudoascites herald the injury. Retrograde cystography may aid identification.
Treatment of bladder injuries
Small perforations recognized intraoperatively may be conservatively managed by postoperative bladder drainage for 5 to 7 days. Most other bladder injuries require prompt intervention. For example, trocar injury to the bladder dome requires one- or two-layer closure followed by 5 to 7 days of urinary drainage. (Both closing and healing are promoted by drainage.)
Laparoscopy or laparotomy? Laparoscopic repair has become increasingly common, and bladder injury is a common complication of LAVH.13,14
CASE 3: Postop pain, tachycardia
A 41-year-old obese woman undergoes laparoscopic cystectomy for an 8-cm left ovarian mass. The abdomen is entered on the second attempt with a long Veress needle. The umbilical trocar is reinserted “several” times because of difficulty opening the peritoneum with the tip of the trocar sheath. The surgical procedure is completed within 2 hours, and the patient is discharged 23 hours later.
The next day, she experiences increasing abdominal pain and presents to the emergency room. Upon admission she reports intermittent chills, but denies nausea and vomiting. She is in mild distress, pale and tachycardic, with a temperature of 96.4°, pulse of 117, respiration rate of 20, blood pressure of 106/64 mm Hg, and oxygen saturation of 92%. She also has a diffusely tender abdomen but normal blood work. Abdominal and chest x-rays show a large right subphrenic air-fluid level that is consistent with free intraperitoneal air, unsurprising given her recent surgery. Bibasilar atelectasis and consolidation are noted on the initial chest x-ray.
During observation over the next 2 days, she remains afebrile and tachycardic, but her shortness of breath becomes progressively worse. Neither spiral CT nor lower-extremity Doppler suggests pulmonary embolism or deep venous thrombosis. Supplemental oxygen, aggressive pain management, albuterol, ipratropium, and acetylcysteine are initiated after pulmonary consultation.
The patient tolerates a regular diet on postoperative day 3 and has a bowel movement on day 5. However, the same day she begins vomiting and reports worsening abdominal pain. CT imaging of the abdomen and pelvis reveals free air in the abdomen and loculated fluid with air bubbles suspicious for intra-abdominal infection and perforated bowel.
Exploratory laparotomy reveals diffuse feculent peritonitis, as well as food particles and contrast media. There is a perforation in the antimesenteric side of the ileum approximately 1.5 feet proximal to the ileocecal valve. This perforation measures approximately 1 cm in diameter and is freely spilling intestinal contents. Small bowel resection is performed to treat the perforation.
Following the surgery, the patient recovers slowly.
Could the bowel perforation have been detected sooner?
Intestinal tract injury is a serious complication, particularly with postoperative diagnosis.15 Damage can occur during insertion of the Veress needle or trocar when the bowel is immobilized by adhesions, or during enterolysis.16 Unrecognized thermal injury can cause delayed bowel injury.
Small-bowel damage often occurs during uncontrolled insertion of the Veress needle or primary umbilical trocar. It also may result from sharp dissection or thermal injury.17,18 Abrasions and lacerations can occur if traction is exerted on the bowel using serrated graspers. When adhesions are dense and tissue planes poorly defined, the risk of laceration due to energy sources or sharp dissection increases.
Be cautious during bowel manipulation. Avoid blunt dissection. Be especially careful when the small bowel is adherent to the anterior abdominal wall (FIGURE 2A), particularly during evaluation of patients with a history of bowel resection, exploratory laparotomy for trauma-related peritonitis, or tumor debulking.
Remove the primary and ancillary cannulas under direct visualization with the laparoscope to prevent formation of a vacuum that can draw bowel into the incision and cause herniation.19
FIGURE 2 Adherent bowel, minor bleeding
A: Veress needle pressure measurements are persistently elevated before primary trocar insertion in this patient, raising the suspicion of adhesive disease from earlier surgery. As a result, the primary trocar is relocated to the left upper quadrant. Inspection confirms that small bowel is adherent to the anterior abdominal wall.
FIGURE 2 Adherent bowel, minor bleeding
B: After the small-bowel adhesions are dissected off the anterior abdominal wall via laparoscopy, a small hematoma is discovered, likely caused by the Veress needle. The patient is managed conservatively and recovers.
The value of open laparoscopy
In open laparoscopy, an abdominal incision is made into the peritoneal cavity so that the trocar can be placed under direct vision, after which the abdomen is insufflated. This approach can prevent bowel injury only when the adhesions and attachments are to the anterior abdominal wall and away from the entry site. When the attachment lies directly beneath the umbilicus, however, open laparoscopy is no guarantee against injury.
When bowel adhesions are severe, use alternative trocar sites such as the left upper quadrant (Palmer’s point) for the Veress needle and primary trocars.5,20,21
The likelihood of perforation can be reduced with preoperative bowel prep when there is a risk of bowel adhesions.
Identifying bowel injury
We recommend routine inspection of the structures beneath the primary trocar upon insertion of the laparoscope to look for injury to the bowel, mesentery, or vascular structures. If adhesions are found, evaluate the area carefully to rule out injury to the bowel or omentum. It may be necessary to change the position of the laparoscope to assess the patient.
Trauma to the intestinal tract can be mechanical or electrical in nature, and each type of trauma creates a distinctive, characteristic pattern. Thermal injury can be subtle and present as simple blanching or a distinct burn and charring. A small hole or obvious tear in the bowel wall can be the result of mechanical injury.22
Benign-appearing, superficial thermal bowel injuries may be managed conservatively.22 Minimal serosal burns (smaller than 5 mm in diameter) can be managed expectantly. Immediate surgical intervention is needed if the area of blanching on the intestinal serosa exceeds 5 mm in diameter or if the burn appears to involve more than the serosa.23
Small-bowel injuries that escape notice intraoperatively generally become apparent 2 to 4 days later, when the patient develops fever, nausea, lower abdominal pain, and anorexia. On postoperative day 5 or 6, the white blood cell (WBC) count rises and earlier symptoms may become worse. Radiography may reveal multiple air and fluid levels—another sign of bowel injury. Be aware that if the patient has clinical symptoms of gastrointestinal injury, even if the WBC count is normal, exploratory laparoscopy or laparotomy is necessary for accurate diagnosis.
Intraoperatively discovered injury
Careful inspection may reveal no leakage or bleeding in the affected area. Small punctures or superficial lacerations seal readily and may not require further treatment (FIGURE 2B), but larger perforations require repair. Straightforward repair is not always possible when the injury is extensive and considerable time has elapsed before it is discovered.
Inspect the intestine thoroughly at the conclusion of a procedure; obvious leakage requires intervention. Repair the small intestine in one or two layers, using the initial row of interrupted sutures to approximate the mucosa and muscularis.24 To lessen the risk of stenosis, close all lacerations transversely when they are smaller than one half the diameter of the bowel. If the laceration exceeds that size, segmental resection and anastomosis are necessary. Resection is prudent if the mesenteric blood supply is compromised.25
When performing one-layer repair of the small bowel, delayed absorbable suture (eg, Vicryl or PDS) or nonabsorbable suture (eg, silk) is recommended.26
At the conclusion of a repair, copiously irrigate the entire abdomen. Place a nasogastric tube only if ileus is anticipated; the tube can be removed when drainage diminishes and active bowel sounds and flatus appear. Do not give anything by mouth until the patient has return of bowel function and active peristalsis. Prescribe prophylactic antibiotics.
Note that peritonitis sometimes develops after repair of the bowel.25 This can be managed with prolonged bowel rest and peripheral or total parenteral nutrition.
Conservative management may be possible
Patients whose symptoms of bowel laceration become apparent after discharge can sometimes be managed conservatively. More than 50% of patients treated conservatively require no surgery.23 Inpatient management consists of monitoring the WBC count, providing hydration and IV antibiotics, and examining the patient every 6 hours, giving nothing by mouth.
When injury is discovered later
If conservative management with observation and bowel rest fails, or the patient complains of severe abdominal pain, vomiting, nausea, obstipation, or signs and symptoms of peritonitis, such as the patient in Case 3, immediate surgical intervention is necessary. When an injury is not detected until some time after initial surgery, resection of all necrotic tissue is mandatory. In most cases, the perforation is managed by segmental resection and reanastomosis. Evaluate the entire small and large bowel to rule out any other injury, and irrigate generously. Bowel rest, parenteral nutrition, and IV antibiotics also are indicated.
Of 36,928 procedures reported by members of the American Association of Gynecologic Laparoscopists, there were two deaths—both caused by unrecognized bowel injury.15
CASE 4: Large-bowel injury precipitates lengthy recovery
A surgeon performs a left laparoscopic salpingo-oophorectomy to remove an 8-cm ovarian endometrioma that is adherent to the rectosigmoid colon of a 40-year-old diabetic woman. Sharp and electrosurgical scissors are used to separate the adnexa from the rectosigmoid colon. No injury is observed, and she is discharged the same day. Four days later, she returns with severe abdominal pain, nausea, vomiting, and fatigue. Lab tests reveal a WBC count of 17,000; a CT scan shows pockets of air beneath the diaphragm, as well as fluid collection suggestive of a pelvic abscess.
Immediate laparotomy is performed, during which the surgeon discovers contamination of the abdominal viscera by bowel contents, as well as a 0.5-cm perforation of the rectosigmoid colon. The perforation is repaired in two layers after its edges are trimmed, and a diverting colostomy is performed. The patient is admitted to the ICU and requires antibiotic treatment, total parenteral nutrition, and bowel rest due to severe peritonitis. She gradually recovers and is discharged 3 weeks later. The diverting colostomy is reversed 3 months later.
Even small perforations in the large bowel can cause infection and abscess due to the high bacterial content of the colon. The most common cause of injury to the rectosigmoid colon is pelvic adhesiolysis during cul-de-sac dissection, treatment of pelvic endometriosis, and resection of adherent pelvic masses.
Sharp dissection with scissors or high-powered lasers is relatively safe near the bowel. When dissecting the cul-de-sac, identify the vagina and rectum by placing a probe or finger in each area. Begin dissection from the unaffected pararectal space, and proceed toward the obliterated cul-de-sac.27,28
Bowel prep is indicated before extensive pelvic surgery and when the history suggests endometriosis or significant pelvic adhesions. Some general surgeons base their decision to perform colostomy (or not) on whether the bowel was prepped preoperatively.29
If the large bowel is perforated by the Veress needle, the saline aspiration test will yield brownish fluid. When significant pelvic adhesiolysis or pelvic or endometriotic tumor resection is performed, inject air into the rectum afterward via a sigmoidoscope or bulb syringe and assess the submerged rectum and rectosigmoid colon for bubbling. The rectal wall may be weakened during these types of procedures, so instruct the patient to use oral stool softeners and avoid enemas.30
Delay in detection can have serious ramifications
When a large-bowel injury goes undetected at the time of operation, the patient generally presents on the third or fourth postoperative day with mild fever, occasionally sudden sharp epigastric pain, lower abdominal pain, slight nausea, and anorexia. By the fifth or sixth day, these symptoms have become more severe and are accompanied by peritonitis and an elevated WBC count.
Whenever a patient complains of abdominal pain and a deteriorating condition, assume that bowel injury is the cause until it is proved otherwise.
Intraoperative management
Repair small trocar wounds using primary suture closure. Copious lavage of the peritoneal cavity, drainage, and a broad-spectrum antibiotic minimize the risk of infection. Manage deep electrical injury to the right colon by resecting the injured segment and performing primary anastomosis. Primary closure or resection and reanastomosis may not be adequate when the vascular supply of the descending colon or rectum is compromised. In that case, perform a diverting colostomy or ileostomy, which can be reversed 6 to 12 weeks later.25,26
CASE 5: Vascular injury
A tall, thin, athletic 19-year-old undergoes diagnostic laparoscopy to rule out pelvic pathology after she complains of severe, monthly abdominal pain. Upon insertion of the laparoscope, the surgeon observes a large hematoma forming at the right pelvic sidewall. At the same time, the anesthesiologist reports a significant drop in blood pressure, and vascular injury is diagnosed. The surgeon attempts to control the bleeding using bipolar coagulation, but the problem only becomes worse. He decides to switch to laparotomy.
A vascular surgeon is called in, and injury to the right common iliac artery and vein—apparently caused during insertion of the primary umbilical trocar—is repaired. The patient is given 5 U of red blood cells. She goes home 10 days later, but returns with thrombophlebitis and rejection of the graft. After several surgeries, she finally recovers, with some sequelae, such as unilateral leg edema.
Management of vascular injury depends on the source and type of injury. On major vessels, electrocoagulation is contraindicated. After immediate atraumatic compression with tamponade to control bleeding, vascular repair, in consultation with a vascular surgeon, is indicated. At times, a vascular graft may be required.
Smaller vessels, such as the infundibulopelvic ligament or uterine vessels, can be managed by clips, suture, or loop ligatures. If thermal energy is used in the repair, be careful to avoid injury to surrounding structures.
Most emergency laparotomies are performed for uncontrolled bleeding.30,31 Lack of control or a wrong angle at insertion of the Veress needle and trocars is a major cause of large-vessel injury. Sharp dissection of adhesions, uterosacral ablation, transection of vascular pedicles without adequate dessication, and rough handling of tissues can all cause bleeding. Distorted anatomy is a main cause of vascular injury and can compound injury in areas more prone to bleeding, such as the oviduct, infundibulopelvic ligament, mesosalpinx, and pelvic sidewall vessels.
The return of pressure gradients to normal levels at the end of a procedure can be accompanied by bleeding into the retroperitoneal space, so evaluate the patient in a supine position after intra-abdominal pressure is reduced.
A vascular surgeon may be required
Depending on the type of vessel, size and location of the injury, and degree of bleeding, you may use unipolar or bipolar electrocoagulation, suture, clips, vasopressin, or loop ligatures to control bleeding. Although diluted vasopressin (10 U in 60 mL of lactated Ringer’s saline) can decrease oozing from raw peritoneal areas, injury to a major vessel, such as the iliac vessels, vena cava, or aorta, needs immediate control and proper repair. The decision to perform laparoscopy or laparotomy depends on your preference and experience. In any case, a vascular surgeon may be consulted for major vascular injuries.32
If a major vessel is injured, do not crush-clamp it. If possible (and if your laparoscopic skills are advanced), insert a sponge via a 10-mm trocar and apply pressure to the vessel to minimize bleeding and enhance visualization. The decision to repair the injury laparoscopically or by laparotomy should be made judiciously and promptly. n
The authors acknowledge the editorial contributions of Kristina Petrasek and Barbara Page, of the University of California, Berkeley, to the manuscript of this article.
To view three clips of surgical pearls for laparoscopy, visit the Video Library.
<huc>Q.</huc>What is the only surgical procedure that is completely safe?
<huc>A.</huc>The surgical procedure that is not performed.
The unfortunate truth is that complications can occur during any operative procedure, despite our best efforts—and laparoscopy is no exception. Being vigilant for iatrogenic injuries, both during and after surgery, and ensuring that repairs are both thorough and timely, are two of our best weapons against major complications, along with meticulous technique and adequate experience.
This article features five cases that illustrate some of the most serious complications of laparoscopy—and how to prevent and manage them.
CASE 1: Surgical patient returns with signs of ureteral injury
A 42-year-old woman with a history of endometriosis undergoes laparoscopic hysterectomy and bilateral salpingo-oophorectomy. She is discharged 2 days later. Two days after that, she returns to the hospital complaining of fluid leaking from the vagina. She has no fever or any other significant complaint or physical findings other than abdominal tenderness, which is to be expected after surgery. A computed tomography (CT) scan with intravenous (IV) contrast reveals left ureteral obstruction near the bladder, with extravasation of contrast media into the abdominal cavity. Further investigation reveals a left ureteral transection.
Could this injury have been avoided? How should it be managed?
Postoperative diagnosis of ureteral injury can be challenging, in part because up to 50% of unilateral cases are asymptomatic. Be on the lookout for this complication in women who have undergone pelvic sidewall dissection or laparoscopic hysterectomy, such as the patient in the case just described. As the number of laparoscopic hysterectomies and retroperitoneal procedures has risen in recent years, so has the rate of ureteral injury, with an incidence of 0.3% to 2%.1,2
Ureteral injury can be caused by ligation, ischemia, resection, transection, crushing, or angulation. Three sites are particularly troublesome: the infundibulopelvic ligament, ovarian fossa, and ureteral tunnel.3,4 In Case 1, injury to the ureter was proximal to the bladder and probably occurred during transection of the uterosacral cardinal ligament complex.
What’s the best preventive strategy?
Meticulous technique is imperative to protect the ureters. This includes adequate visualization, intraperitoneal or retroperitoneal dissection, and early identification of the ureter. In a high-risk patient likely to have distorted anatomy due to severe endometriosis and fibrosis, retroperitoneal dissection of any adhesions or tumor and identification of the ureter are the best ways to avoid injury.
Intraperitoneal identification and dissection of the ureters can be enhanced by hydrodissection and resection of the affected peritoneum.3,4 To create a safe operating plane, make a small opening in the peritoneum below the ureter and inject 50 to 100 mL of lactated Ringer’s solution along the course of the ureter, which will displace it laterally.5
Although neither IV indigo carmine nor ureteral catheterization has been shown to reduce the risk of ureteral injury or identify ligation or thermal injury,3,6 both can help the surgeon identify intraoperative perforation of the ureter. Liberal use of cystoscopy with indigo carmine administration for identification of ureteral flow and ureteral catheterization can be used in potentially high-risk patients. If there is suspicion for devascularization or thermal injury, use prophylactic ureteral stents postoperatively for 2 to 4 weeks.
Don’t hesitate to consult a urologist
In Case 1, the surgeon sought immediate urologic consultation and the patient underwent laparotomy with ureteroneocystotomy without sequelae.
In general, management of ureteral injury depends on its severity and location, as well as the comfort level of the surgeon. Minor injuries are sometimes managed with cystoscopic stent placement, but more severe cases may require operative ureteral repair.
In cases like this one, where ureteral injury occurred in close proximity to the bladder, a ureteroneocystotomy is possible. However, in more cephalad injuries, there may be insufficient residual ureter to allow such a repair. In these cases, a Boari flap may be attempted to use bladder tissue to bridge the gap to the ureteral edge. Rarely, in high ureteral injuries, trans-ureteroureterostomy may be appropriate. This procedure carries the greatest risk, given that both kidneys are reliant on one ureter.
Is laparoscopic repair reasonable?
When surgical intervention is necessary, the choice between laparoscopy and laparotomy depends on the skill and comfort level of the surgeon and the availability of instruments and support team.6,7 That said, ureteral injury is usually treated via laparotomy.1 As operative laparoscopy becomes even more commonplace, reconstruction of the urinary system will increasingly be managed laparoscopically.
Depending on the size and location of the injury, reconstruction may involve ureteral reimplantation with or without a psoas hitch, Boari flap, or primary endtoend anastomosis.8-10
CASE 2: Postoperative symptoms lead to rehospitalization
A 35-year-old patient undergoes laparoscopic ovarian cystectomy and returns home the same day. She is readmitted 72 hours later because of lower abdominal tenderness, worsening nausea and vomiting, and urine-like drainage from her midline suprapubic trocar site. Analysis of the leaking fluid shows high creatinine levels consistent with urine. The patient has no fever and is hemodynamically stable. Examination reveals a moderately distended abdomen with decreased bowel sounds. Hematuria is evident on urine analysis.
Urologic consultation is obtained, and the patient undergoes simultaneous laparoscopy and cystoscopy, during which perforation of the bladder dome is discovered, apparently caused by the mid suprapubic trocar. The bladder is mobilized anteriorly, and both anterior and posterior aspects of the perforation are repaired in one layer laparoscopically.
After continuous drainage with a transurethral Foley catheter for 7 days, cystography shows complete healing of the bladder, and the Foley catheter is removed. The patient recovers completely.
Vesical injury sometimes occurs in patients who have a history of laparotomy, a full bladder at the time of surgery, or displaced anatomy due to pelvic adhesions.11 Although bladder injury is rare, laparoscopy increases the risk. Trocars, uterine manipulators, and blunt instruments can perforate or lacerate the bladder, and energy devices can cause thermal injury. The risk of bladder injury increases during laparoscopic hysterectomy.
Be vigilant about trocar placement and dissection techniques
Accessory trocars can injure a full bladder. Injury can also occur when distorted anatomy from a previous pelvic operation obscures bladder boundaries, making insertion of the midline trocar potentially perilous (FIGURE 1). The Veress needle and Rubin’s cannula can perforate the bladder.11-13 And in the anterior cul-de-sac, adhesiolysis, deep coagulation, laser ablation, or sharp excision of endometriosis implants can predispose a patient to bladder injury.
In women with severe endometriosis, lower-segment myoma, or a history of cesarean section, the bladder is vulnerable to laceration when blunt dissection is used during laparoscopic hysterectomy or laparoscopically assisted vaginal hysterectomy (LAVH). A vesical injury also can occur at the time of laparoscopic bladder-neck suspension upon entry into, and dissection of, the space of Retzius.
FIGURE 1 A bladder at risk
In this patient with a previous cesarean section, the bladder is adherent to the anterior abdominal wall. Needle mapping in the conventional midline trocar position indicates that the trocar must be relocated to avoid bladder injury.
Intraoperative findings that suggest bladder injury include air in the urinary catheter, hematuria, trocar site drainage of urine, or indigo carmine leakage. Postoperative signs and symptoms include leaking from incisional sites, a mass in the abdominal wall, and abdominal swelling.
Liberal use of cystoscopy or distension of the bladder with 300 to 500 mL of normal saline is recommended whenever there is a suspicion of bladder injury, especially during laparoscopic hysterectomy or LAVH. When a trocar causes the injury, look for both entry and exit punctures, both of which should be treated.
No matter how much care is taken, some bladder injuries, such as vesicovaginal fistulae, become apparent only postoperatively. More rarely, peritonitis or pseudoascites herald the injury. Retrograde cystography may aid identification.
Treatment of bladder injuries
Small perforations recognized intraoperatively may be conservatively managed by postoperative bladder drainage for 5 to 7 days. Most other bladder injuries require prompt intervention. For example, trocar injury to the bladder dome requires one- or two-layer closure followed by 5 to 7 days of urinary drainage. (Both closing and healing are promoted by drainage.)
Laparoscopy or laparotomy? Laparoscopic repair has become increasingly common, and bladder injury is a common complication of LAVH.13,14
CASE 3: Postop pain, tachycardia
A 41-year-old obese woman undergoes laparoscopic cystectomy for an 8-cm left ovarian mass. The abdomen is entered on the second attempt with a long Veress needle. The umbilical trocar is reinserted “several” times because of difficulty opening the peritoneum with the tip of the trocar sheath. The surgical procedure is completed within 2 hours, and the patient is discharged 23 hours later.
The next day, she experiences increasing abdominal pain and presents to the emergency room. Upon admission she reports intermittent chills, but denies nausea and vomiting. She is in mild distress, pale and tachycardic, with a temperature of 96.4°, pulse of 117, respiration rate of 20, blood pressure of 106/64 mm Hg, and oxygen saturation of 92%. She also has a diffusely tender abdomen but normal blood work. Abdominal and chest x-rays show a large right subphrenic air-fluid level that is consistent with free intraperitoneal air, unsurprising given her recent surgery. Bibasilar atelectasis and consolidation are noted on the initial chest x-ray.
During observation over the next 2 days, she remains afebrile and tachycardic, but her shortness of breath becomes progressively worse. Neither spiral CT nor lower-extremity Doppler suggests pulmonary embolism or deep venous thrombosis. Supplemental oxygen, aggressive pain management, albuterol, ipratropium, and acetylcysteine are initiated after pulmonary consultation.
The patient tolerates a regular diet on postoperative day 3 and has a bowel movement on day 5. However, the same day she begins vomiting and reports worsening abdominal pain. CT imaging of the abdomen and pelvis reveals free air in the abdomen and loculated fluid with air bubbles suspicious for intra-abdominal infection and perforated bowel.
Exploratory laparotomy reveals diffuse feculent peritonitis, as well as food particles and contrast media. There is a perforation in the antimesenteric side of the ileum approximately 1.5 feet proximal to the ileocecal valve. This perforation measures approximately 1 cm in diameter and is freely spilling intestinal contents. Small bowel resection is performed to treat the perforation.
Following the surgery, the patient recovers slowly.
Could the bowel perforation have been detected sooner?
Intestinal tract injury is a serious complication, particularly with postoperative diagnosis.15 Damage can occur during insertion of the Veress needle or trocar when the bowel is immobilized by adhesions, or during enterolysis.16 Unrecognized thermal injury can cause delayed bowel injury.
Small-bowel damage often occurs during uncontrolled insertion of the Veress needle or primary umbilical trocar. It also may result from sharp dissection or thermal injury.17,18 Abrasions and lacerations can occur if traction is exerted on the bowel using serrated graspers. When adhesions are dense and tissue planes poorly defined, the risk of laceration due to energy sources or sharp dissection increases.
Be cautious during bowel manipulation. Avoid blunt dissection. Be especially careful when the small bowel is adherent to the anterior abdominal wall (FIGURE 2A), particularly during evaluation of patients with a history of bowel resection, exploratory laparotomy for trauma-related peritonitis, or tumor debulking.
Remove the primary and ancillary cannulas under direct visualization with the laparoscope to prevent formation of a vacuum that can draw bowel into the incision and cause herniation.19
FIGURE 2 Adherent bowel, minor bleeding
A: Veress needle pressure measurements are persistently elevated before primary trocar insertion in this patient, raising the suspicion of adhesive disease from earlier surgery. As a result, the primary trocar is relocated to the left upper quadrant. Inspection confirms that small bowel is adherent to the anterior abdominal wall.
FIGURE 2 Adherent bowel, minor bleeding
B: After the small-bowel adhesions are dissected off the anterior abdominal wall via laparoscopy, a small hematoma is discovered, likely caused by the Veress needle. The patient is managed conservatively and recovers.
The value of open laparoscopy
In open laparoscopy, an abdominal incision is made into the peritoneal cavity so that the trocar can be placed under direct vision, after which the abdomen is insufflated. This approach can prevent bowel injury only when the adhesions and attachments are to the anterior abdominal wall and away from the entry site. When the attachment lies directly beneath the umbilicus, however, open laparoscopy is no guarantee against injury.
When bowel adhesions are severe, use alternative trocar sites such as the left upper quadrant (Palmer’s point) for the Veress needle and primary trocars.5,20,21
The likelihood of perforation can be reduced with preoperative bowel prep when there is a risk of bowel adhesions.
Identifying bowel injury
We recommend routine inspection of the structures beneath the primary trocar upon insertion of the laparoscope to look for injury to the bowel, mesentery, or vascular structures. If adhesions are found, evaluate the area carefully to rule out injury to the bowel or omentum. It may be necessary to change the position of the laparoscope to assess the patient.
Trauma to the intestinal tract can be mechanical or electrical in nature, and each type of trauma creates a distinctive, characteristic pattern. Thermal injury can be subtle and present as simple blanching or a distinct burn and charring. A small hole or obvious tear in the bowel wall can be the result of mechanical injury.22
Benign-appearing, superficial thermal bowel injuries may be managed conservatively.22 Minimal serosal burns (smaller than 5 mm in diameter) can be managed expectantly. Immediate surgical intervention is needed if the area of blanching on the intestinal serosa exceeds 5 mm in diameter or if the burn appears to involve more than the serosa.23
Small-bowel injuries that escape notice intraoperatively generally become apparent 2 to 4 days later, when the patient develops fever, nausea, lower abdominal pain, and anorexia. On postoperative day 5 or 6, the white blood cell (WBC) count rises and earlier symptoms may become worse. Radiography may reveal multiple air and fluid levels—another sign of bowel injury. Be aware that if the patient has clinical symptoms of gastrointestinal injury, even if the WBC count is normal, exploratory laparoscopy or laparotomy is necessary for accurate diagnosis.
Intraoperatively discovered injury
Careful inspection may reveal no leakage or bleeding in the affected area. Small punctures or superficial lacerations seal readily and may not require further treatment (FIGURE 2B), but larger perforations require repair. Straightforward repair is not always possible when the injury is extensive and considerable time has elapsed before it is discovered.
Inspect the intestine thoroughly at the conclusion of a procedure; obvious leakage requires intervention. Repair the small intestine in one or two layers, using the initial row of interrupted sutures to approximate the mucosa and muscularis.24 To lessen the risk of stenosis, close all lacerations transversely when they are smaller than one half the diameter of the bowel. If the laceration exceeds that size, segmental resection and anastomosis are necessary. Resection is prudent if the mesenteric blood supply is compromised.25
When performing one-layer repair of the small bowel, delayed absorbable suture (eg, Vicryl or PDS) or nonabsorbable suture (eg, silk) is recommended.26
At the conclusion of a repair, copiously irrigate the entire abdomen. Place a nasogastric tube only if ileus is anticipated; the tube can be removed when drainage diminishes and active bowel sounds and flatus appear. Do not give anything by mouth until the patient has return of bowel function and active peristalsis. Prescribe prophylactic antibiotics.
Note that peritonitis sometimes develops after repair of the bowel.25 This can be managed with prolonged bowel rest and peripheral or total parenteral nutrition.
Conservative management may be possible
Patients whose symptoms of bowel laceration become apparent after discharge can sometimes be managed conservatively. More than 50% of patients treated conservatively require no surgery.23 Inpatient management consists of monitoring the WBC count, providing hydration and IV antibiotics, and examining the patient every 6 hours, giving nothing by mouth.
When injury is discovered later
If conservative management with observation and bowel rest fails, or the patient complains of severe abdominal pain, vomiting, nausea, obstipation, or signs and symptoms of peritonitis, such as the patient in Case 3, immediate surgical intervention is necessary. When an injury is not detected until some time after initial surgery, resection of all necrotic tissue is mandatory. In most cases, the perforation is managed by segmental resection and reanastomosis. Evaluate the entire small and large bowel to rule out any other injury, and irrigate generously. Bowel rest, parenteral nutrition, and IV antibiotics also are indicated.
Of 36,928 procedures reported by members of the American Association of Gynecologic Laparoscopists, there were two deaths—both caused by unrecognized bowel injury.15
CASE 4: Large-bowel injury precipitates lengthy recovery
A surgeon performs a left laparoscopic salpingo-oophorectomy to remove an 8-cm ovarian endometrioma that is adherent to the rectosigmoid colon of a 40-year-old diabetic woman. Sharp and electrosurgical scissors are used to separate the adnexa from the rectosigmoid colon. No injury is observed, and she is discharged the same day. Four days later, she returns with severe abdominal pain, nausea, vomiting, and fatigue. Lab tests reveal a WBC count of 17,000; a CT scan shows pockets of air beneath the diaphragm, as well as fluid collection suggestive of a pelvic abscess.
Immediate laparotomy is performed, during which the surgeon discovers contamination of the abdominal viscera by bowel contents, as well as a 0.5-cm perforation of the rectosigmoid colon. The perforation is repaired in two layers after its edges are trimmed, and a diverting colostomy is performed. The patient is admitted to the ICU and requires antibiotic treatment, total parenteral nutrition, and bowel rest due to severe peritonitis. She gradually recovers and is discharged 3 weeks later. The diverting colostomy is reversed 3 months later.
Even small perforations in the large bowel can cause infection and abscess due to the high bacterial content of the colon. The most common cause of injury to the rectosigmoid colon is pelvic adhesiolysis during cul-de-sac dissection, treatment of pelvic endometriosis, and resection of adherent pelvic masses.
Sharp dissection with scissors or high-powered lasers is relatively safe near the bowel. When dissecting the cul-de-sac, identify the vagina and rectum by placing a probe or finger in each area. Begin dissection from the unaffected pararectal space, and proceed toward the obliterated cul-de-sac.27,28
Bowel prep is indicated before extensive pelvic surgery and when the history suggests endometriosis or significant pelvic adhesions. Some general surgeons base their decision to perform colostomy (or not) on whether the bowel was prepped preoperatively.29
If the large bowel is perforated by the Veress needle, the saline aspiration test will yield brownish fluid. When significant pelvic adhesiolysis or pelvic or endometriotic tumor resection is performed, inject air into the rectum afterward via a sigmoidoscope or bulb syringe and assess the submerged rectum and rectosigmoid colon for bubbling. The rectal wall may be weakened during these types of procedures, so instruct the patient to use oral stool softeners and avoid enemas.30
Delay in detection can have serious ramifications
When a large-bowel injury goes undetected at the time of operation, the patient generally presents on the third or fourth postoperative day with mild fever, occasionally sudden sharp epigastric pain, lower abdominal pain, slight nausea, and anorexia. By the fifth or sixth day, these symptoms have become more severe and are accompanied by peritonitis and an elevated WBC count.
Whenever a patient complains of abdominal pain and a deteriorating condition, assume that bowel injury is the cause until it is proved otherwise.
Intraoperative management
Repair small trocar wounds using primary suture closure. Copious lavage of the peritoneal cavity, drainage, and a broad-spectrum antibiotic minimize the risk of infection. Manage deep electrical injury to the right colon by resecting the injured segment and performing primary anastomosis. Primary closure or resection and reanastomosis may not be adequate when the vascular supply of the descending colon or rectum is compromised. In that case, perform a diverting colostomy or ileostomy, which can be reversed 6 to 12 weeks later.25,26
CASE 5: Vascular injury
A tall, thin, athletic 19-year-old undergoes diagnostic laparoscopy to rule out pelvic pathology after she complains of severe, monthly abdominal pain. Upon insertion of the laparoscope, the surgeon observes a large hematoma forming at the right pelvic sidewall. At the same time, the anesthesiologist reports a significant drop in blood pressure, and vascular injury is diagnosed. The surgeon attempts to control the bleeding using bipolar coagulation, but the problem only becomes worse. He decides to switch to laparotomy.
A vascular surgeon is called in, and injury to the right common iliac artery and vein—apparently caused during insertion of the primary umbilical trocar—is repaired. The patient is given 5 U of red blood cells. She goes home 10 days later, but returns with thrombophlebitis and rejection of the graft. After several surgeries, she finally recovers, with some sequelae, such as unilateral leg edema.
Management of vascular injury depends on the source and type of injury. On major vessels, electrocoagulation is contraindicated. After immediate atraumatic compression with tamponade to control bleeding, vascular repair, in consultation with a vascular surgeon, is indicated. At times, a vascular graft may be required.
Smaller vessels, such as the infundibulopelvic ligament or uterine vessels, can be managed by clips, suture, or loop ligatures. If thermal energy is used in the repair, be careful to avoid injury to surrounding structures.
Most emergency laparotomies are performed for uncontrolled bleeding.30,31 Lack of control or a wrong angle at insertion of the Veress needle and trocars is a major cause of large-vessel injury. Sharp dissection of adhesions, uterosacral ablation, transection of vascular pedicles without adequate dessication, and rough handling of tissues can all cause bleeding. Distorted anatomy is a main cause of vascular injury and can compound injury in areas more prone to bleeding, such as the oviduct, infundibulopelvic ligament, mesosalpinx, and pelvic sidewall vessels.
The return of pressure gradients to normal levels at the end of a procedure can be accompanied by bleeding into the retroperitoneal space, so evaluate the patient in a supine position after intra-abdominal pressure is reduced.
A vascular surgeon may be required
Depending on the type of vessel, size and location of the injury, and degree of bleeding, you may use unipolar or bipolar electrocoagulation, suture, clips, vasopressin, or loop ligatures to control bleeding. Although diluted vasopressin (10 U in 60 mL of lactated Ringer’s saline) can decrease oozing from raw peritoneal areas, injury to a major vessel, such as the iliac vessels, vena cava, or aorta, needs immediate control and proper repair. The decision to perform laparoscopy or laparotomy depends on your preference and experience. In any case, a vascular surgeon may be consulted for major vascular injuries.32
If a major vessel is injured, do not crush-clamp it. If possible (and if your laparoscopic skills are advanced), insert a sponge via a 10-mm trocar and apply pressure to the vessel to minimize bleeding and enhance visualization. The decision to repair the injury laparoscopically or by laparotomy should be made judiciously and promptly. n
The authors acknowledge the editorial contributions of Kristina Petrasek and Barbara Page, of the University of California, Berkeley, to the manuscript of this article.
1. Ostrzenski A, Radolinski B, Ostrzenska K. A review of laparoscopic ureteral injury in pelvic surgery. Obstet Gynecol Surv. 2003;58:794-799.
2. Kabalin J. Chapter 1—Surgical anatomy of the retroperitoneum, kidneys, and ureters. In: Walsh P, Retik A, Wein A, eds. Campbell’s Urology. 8th ed. Philadelphia: Saunders; 2002 36-40.
3. Chan J, Morrow J, Manetta A. Prevention of ureteral injuries in gynecologic surgery. Am J Obstet Gynecol. 2003;188:1273-1277.
4. Grainger DA, Soderstrom RM, Schiff SF, Glickman MG, DeCherney AH, Diamond MP. Ureteral injuries at laparoscopy: insights into diagnosis, management, and prevention. Obstet Gynecol. 1990;75:839-843.
5. Nezhat C. Chapter 20. Operative Gynecologic Laparoscopy: Principles and Techniques. 2nd ed. New York: McGraw–Hill; 2000.
6. Ou CS, Huang IA, Rowbotham R. Laparoscopic ureteroureteral anastomosis for repair of ureteral injury involving stricture. Int Urogynecol J Pelvic Floor Dysfunct. 2005;16:155-157.
7. Modi P, Goel R, Dodiya S. Laparoscopic ureteroneocystostomy for distal ureteral injuries. Urology. 2005;66:751-753.
8. Nezhat C, Nezhat F, Nezhat CH, et al. Urinary tract endometriosis treated by laparoscopy. Fertil Steril. 1996;66:920-924.
9. Nezhat C, Nezhat F. Laparoscopic repair of ureter resected during operative laparoscopy. Obstet Gynecol. 1992;80:543-544.
10. Nezhat CH, Nezhat FR, Freiha F, Nezhat CR. Laparoscopic vesicopsoas hitch for infiltrative ureteral endometriosis. Fertil Steril. 1999;71:376-379.
11. Georgy FM, Fettman HH, Chefetz MD. Complications of laparoscopy: two cases of perforated urinary bladder. Am J Obstet Gynecol. 1974;120:1121-1122.
12. Sherer DM. Inadvertent transvaginal cystotomy during laparoscopy. Int J Gynaecol Obstet. 1990;32:77-79.
13. Nezhat CH, Seidman DS, Nezhat F, et al. Laparoscopic management of internal and unintentional cystotomy. J Urol. 1996;156:1400-1402.
14. Lee CL, Lai YM, Soong YK. Management of urinary bladder injuries in laparoscopic assisted vaginal hysterectomy. Acta Obstet Gynecol Scand. 1996;75:174-177.
15. Peterson HB, et al. American Association of Gynecologic Laparoscopists’ 1988 membership survey on operative laparoscopy. J Reprod Med. 1990;35:587-589.
16. Phillips JM, Hulka JF, Hulka B, et al. 1978 AAGL membership survey. J Reprod Med. 1981;26:529-533.
17. Chapron C, Pierre F, et al. Gastrointestinal injuries during laparoscopy. Hum Reprod. 1999;14:333-337.
18. Schrenk P, Woisetschlager R, Rieger R, et al. Mechanism, management, and prevention of laparoscopic bowel injuries. Gastrointest Endosc. 1996;43:572-574.
19. Sauer M, Jarrett JC. Small bowel obstruction following diagnostic laparoscopy. Fertil Steril. 1984;42:653-654.
20. Penfield AJ. How to prevent complications of open laparoscopy. J Reprod Med. 1985;30:660-663.
21. Brill A, Nezhat F, Nezhat CH, Nezhat C. The incidence of adhesions after prior laparatomy: a laparoscopic appraisal. Obstet Gynecol. 1995;85:269-272.
22. Levy BS, Soderstrom RM, Dail DH. Bowel injuries during laparoscopy: gross anatomy and histology. J Reprod Med. 1985;30:168-172.
23. Wheeless CR. Gastrointestinal injuries associated with laparoscopy. In: Phillips JM, ed. Endoscopy in Gynecology. Santa Fe Springs, Calif: American Association of Gynecologic Laparoscopists; 1978.
24. Borton M. Laparoscopic Complications: Prevention and Management. Philadelphia: Decker; 1986.
25. DeCherney AH. Laparoscopy with unexpected viscus penetration. In: Nichols DH, ed. Clinical Problems, Injuries, and Complications of Gynecologic Surgery. Baltimore: Williams & Wilkins; 1988.
26. Nezhat C, Nezhat F, Ambroze W, Pennington E. Laparoscopic repair of small bowel and colon: a report of 26 cases. Surg Endosc. 1993;7:88-89.
27. Redwine D. Laparoscopic en bloc resection for treatment of the obliterated cul-de-sac in endometriosis. J Reprod Med. 1992;37:696-698.
28. Nezhat C, Nezhat F, et al. Laparoscopic treatment of infiltrative rectosigmoid colon and rectovaginal septum endometriosis by the technique of videolaseroscopy and the CO2 laser. Br J Obstet Gynaecol. 1992;99:664-667.
29. Nezhat C, Seidman D, Nezhat F, et al. The role of intraoperative proctosigmoidoscopy in laparoscopic pelvic surgery. J Am Assoc Gynecol Laparosc. 2004;11:47-49.
30. Chapron CM, et al. Major vascular injuries during gynecologic laparoscopy. J Am Coll Surg. 1997;185:461-465.
31. Geers J, Holden C. Major vascular injury as a complication of laparoscopic surgery: a report of three cases and review of the literature. Am Surg. 1996;62:377-379.
32. Nezhat C, Childers J, Nezhat F, et al. Major retroperitoneal vascular injury during laparoscopic surgery. Hum Reprod. 1997;12:480-483.
Dr. Farr Nezhat reports no relevant financial relationships. Dr. Ceana Nezhat is a speaker and consultant for Karl Storz, Johnson & Johnson, Valleylab, US Surgical, and Viking. Dr. Camran Nezhat is a speaker for or receives educational support from Karl Storz, Stryker, Johnson & Johnson, Valleylab, and Baxter.
1. Ostrzenski A, Radolinski B, Ostrzenska K. A review of laparoscopic ureteral injury in pelvic surgery. Obstet Gynecol Surv. 2003;58:794-799.
2. Kabalin J. Chapter 1—Surgical anatomy of the retroperitoneum, kidneys, and ureters. In: Walsh P, Retik A, Wein A, eds. Campbell’s Urology. 8th ed. Philadelphia: Saunders; 2002 36-40.
3. Chan J, Morrow J, Manetta A. Prevention of ureteral injuries in gynecologic surgery. Am J Obstet Gynecol. 2003;188:1273-1277.
4. Grainger DA, Soderstrom RM, Schiff SF, Glickman MG, DeCherney AH, Diamond MP. Ureteral injuries at laparoscopy: insights into diagnosis, management, and prevention. Obstet Gynecol. 1990;75:839-843.
5. Nezhat C. Chapter 20. Operative Gynecologic Laparoscopy: Principles and Techniques. 2nd ed. New York: McGraw–Hill; 2000.
6. Ou CS, Huang IA, Rowbotham R. Laparoscopic ureteroureteral anastomosis for repair of ureteral injury involving stricture. Int Urogynecol J Pelvic Floor Dysfunct. 2005;16:155-157.
7. Modi P, Goel R, Dodiya S. Laparoscopic ureteroneocystostomy for distal ureteral injuries. Urology. 2005;66:751-753.
8. Nezhat C, Nezhat F, Nezhat CH, et al. Urinary tract endometriosis treated by laparoscopy. Fertil Steril. 1996;66:920-924.
9. Nezhat C, Nezhat F. Laparoscopic repair of ureter resected during operative laparoscopy. Obstet Gynecol. 1992;80:543-544.
10. Nezhat CH, Nezhat FR, Freiha F, Nezhat CR. Laparoscopic vesicopsoas hitch for infiltrative ureteral endometriosis. Fertil Steril. 1999;71:376-379.
11. Georgy FM, Fettman HH, Chefetz MD. Complications of laparoscopy: two cases of perforated urinary bladder. Am J Obstet Gynecol. 1974;120:1121-1122.
12. Sherer DM. Inadvertent transvaginal cystotomy during laparoscopy. Int J Gynaecol Obstet. 1990;32:77-79.
13. Nezhat CH, Seidman DS, Nezhat F, et al. Laparoscopic management of internal and unintentional cystotomy. J Urol. 1996;156:1400-1402.
14. Lee CL, Lai YM, Soong YK. Management of urinary bladder injuries in laparoscopic assisted vaginal hysterectomy. Acta Obstet Gynecol Scand. 1996;75:174-177.
15. Peterson HB, et al. American Association of Gynecologic Laparoscopists’ 1988 membership survey on operative laparoscopy. J Reprod Med. 1990;35:587-589.
16. Phillips JM, Hulka JF, Hulka B, et al. 1978 AAGL membership survey. J Reprod Med. 1981;26:529-533.
17. Chapron C, Pierre F, et al. Gastrointestinal injuries during laparoscopy. Hum Reprod. 1999;14:333-337.
18. Schrenk P, Woisetschlager R, Rieger R, et al. Mechanism, management, and prevention of laparoscopic bowel injuries. Gastrointest Endosc. 1996;43:572-574.
19. Sauer M, Jarrett JC. Small bowel obstruction following diagnostic laparoscopy. Fertil Steril. 1984;42:653-654.
20. Penfield AJ. How to prevent complications of open laparoscopy. J Reprod Med. 1985;30:660-663.
21. Brill A, Nezhat F, Nezhat CH, Nezhat C. The incidence of adhesions after prior laparatomy: a laparoscopic appraisal. Obstet Gynecol. 1995;85:269-272.
22. Levy BS, Soderstrom RM, Dail DH. Bowel injuries during laparoscopy: gross anatomy and histology. J Reprod Med. 1985;30:168-172.
23. Wheeless CR. Gastrointestinal injuries associated with laparoscopy. In: Phillips JM, ed. Endoscopy in Gynecology. Santa Fe Springs, Calif: American Association of Gynecologic Laparoscopists; 1978.
24. Borton M. Laparoscopic Complications: Prevention and Management. Philadelphia: Decker; 1986.
25. DeCherney AH. Laparoscopy with unexpected viscus penetration. In: Nichols DH, ed. Clinical Problems, Injuries, and Complications of Gynecologic Surgery. Baltimore: Williams & Wilkins; 1988.
26. Nezhat C, Nezhat F, Ambroze W, Pennington E. Laparoscopic repair of small bowel and colon: a report of 26 cases. Surg Endosc. 1993;7:88-89.
27. Redwine D. Laparoscopic en bloc resection for treatment of the obliterated cul-de-sac in endometriosis. J Reprod Med. 1992;37:696-698.
28. Nezhat C, Nezhat F, et al. Laparoscopic treatment of infiltrative rectosigmoid colon and rectovaginal septum endometriosis by the technique of videolaseroscopy and the CO2 laser. Br J Obstet Gynaecol. 1992;99:664-667.
29. Nezhat C, Seidman D, Nezhat F, et al. The role of intraoperative proctosigmoidoscopy in laparoscopic pelvic surgery. J Am Assoc Gynecol Laparosc. 2004;11:47-49.
30. Chapron CM, et al. Major vascular injuries during gynecologic laparoscopy. J Am Coll Surg. 1997;185:461-465.
31. Geers J, Holden C. Major vascular injury as a complication of laparoscopic surgery: a report of three cases and review of the literature. Am Surg. 1996;62:377-379.
32. Nezhat C, Childers J, Nezhat F, et al. Major retroperitoneal vascular injury during laparoscopic surgery. Hum Reprod. 1997;12:480-483.
Dr. Farr Nezhat reports no relevant financial relationships. Dr. Ceana Nezhat is a speaker and consultant for Karl Storz, Johnson & Johnson, Valleylab, US Surgical, and Viking. Dr. Camran Nezhat is a speaker for or receives educational support from Karl Storz, Stryker, Johnson & Johnson, Valleylab, and Baxter.