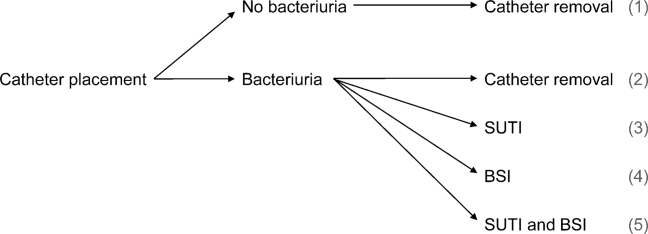
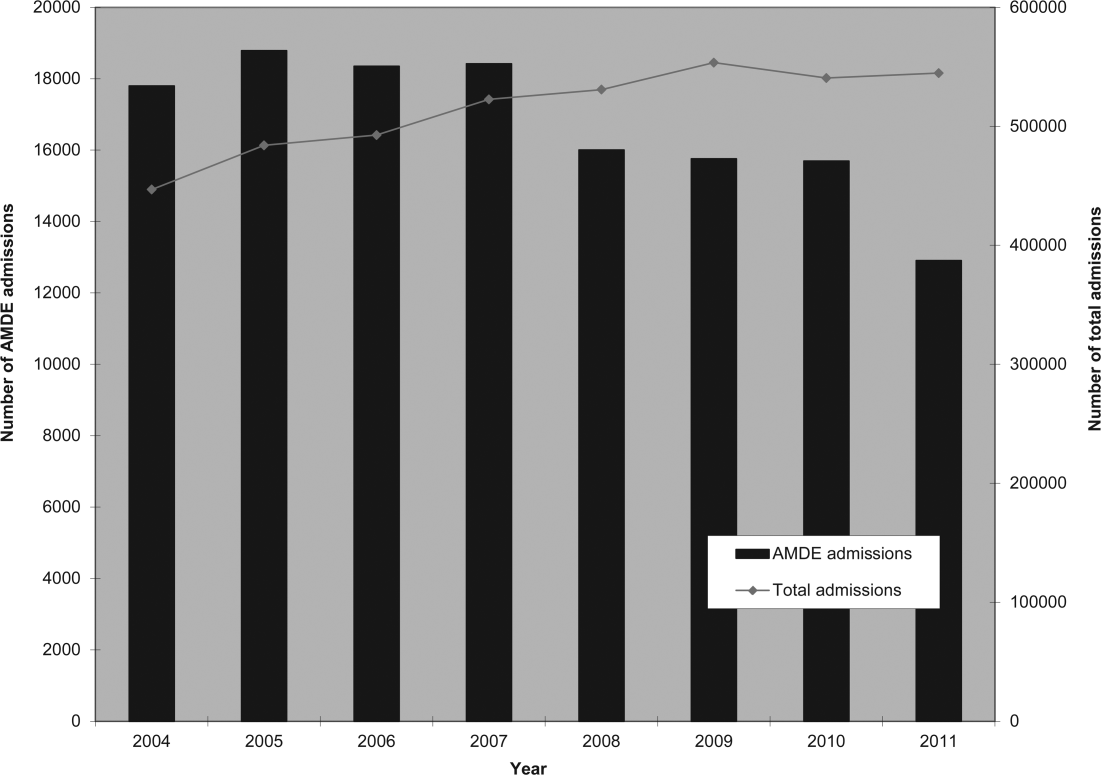
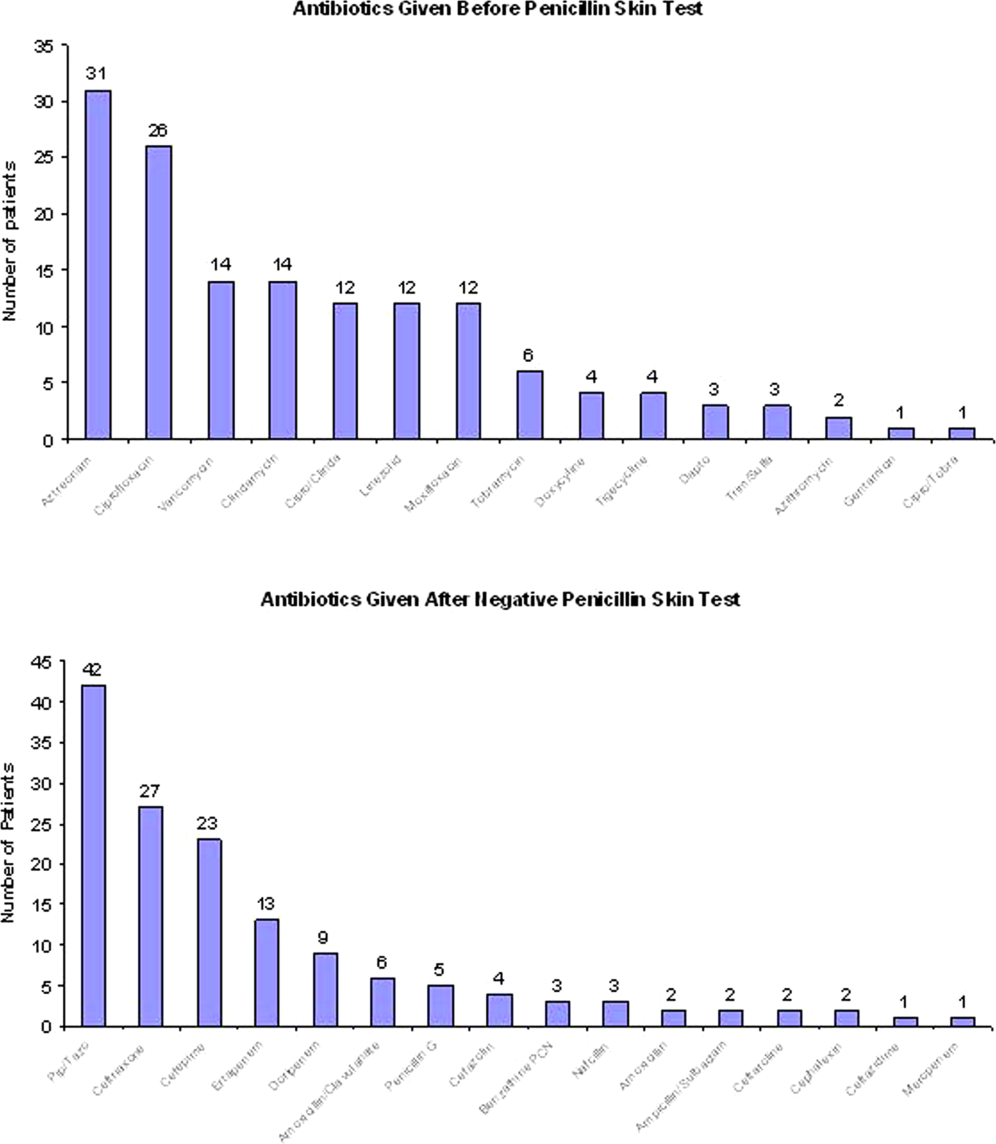
User login
AMA Documentation Analysis
Approximately 1% to 2% of inpatient stays result in discharges against medical advice (AMA).[1] Though relatively infrequent, AMA discharges warrant attention as they are associated with higher morbidity, increased risk of readmission, and greater 30‐day mortality.[2] A recent study found a 30‐day readmission rate among AMA patients of 24.5%, nearly twice that of matched non‐AMA patients, and a 30‐day mortality rate of 1.3%, also nearly double that of planned discharges.[3] Discharges AMA may be expected to decrease index length of stay, yet accounting for 30‐day readmissions they are estimated to increase costs 56% higher than expected from an initial hospitalization.[4] Patients note several possible reasons for leaving AMA including family emergencies, dissatisfaction with care, financial concerns, or simply feeling better, among others.[5, 6, 7] Risk factors for AMA discharges include previous AMA discharge, having no primary care physician, younger age, lack of insurance, male sex, substance abuse, and lower socioeconomic status.[4, 6, 7, 8]
A number of prior studies have assessed risk factors for AMA discharges, the long‐ and short‐term outcomes, patient reasons for leaving, and physician perceptions of why patients leave AMA.[3, 5, 7, 9] However, there is limited information about opportunities for discharge transition interventions in this potentially more vulnerable population. Because of the increased short‐term and long‐term risks to these patients, treatment and follow‐up plans at the time of discharge may carry even greater importance than follow‐up plans with standard discharges. This study analyzed AMA documentation and what interventions were carried out at the time of discharge.
METHODS
We reviewed the records of all adult patients, ages 18 years and older, admitted to a university‐affiliated tertiary care hospital in Dayton, Ohio (a 520‐bed hospital with approximately 17,000 adult patient encounters per year) over a 2‐year period, and who subsequently left AMA. A hospital database identified 351 adult AMA cases (1.0% of adult admissions). A single reviewer performed an in‐depth review of the 291 patient admissions to the general medical service between January 1, 2009 and December 31, 2010, and manually reviewed and abstracted the data of interest. The Wright State University institutional review board approved the study.
Documentation review focused on the presence of a specified AMA note, the presence of documentation addressing informed consent, patient decision‐making capacity, patient health literacy, follow‐up plans, whether or not medications were prescribed, and whether or not any warning indicators of impending AMA were apparent. These items represented key elements of the discharge policy and procedure in place at our institution during the period of study. We speculated that nurses may be more immediately available at the time of AMA discharge and thus might carry out AMA documentation more often than physicians. To assess this we recorded the role (nurse vs provider) of the writer of AMA notes. We also assessed patient gender, length of stay, prior AMA, 30‐day emergency department (ED) re‐encounters, and 30‐day hospital readmission after AMA discharge.
Informed consent was deemed present if patients signed the hospital's standardized AMA form. Decision‐making capacity was assessed as present if there was specific mention of the patient's capacity on the day of discharge. Any mention of health literacy or the patient's stated understanding of his medical condition at any time during the hospitalization was considered positive documentation of healthcare literacy. Follow‐up plans included any mention of where and when the patient would return. Discharge medications included prescribed medication or indication that no medications were warranted. Warning indicators included specific mention of the patient's desire to leave AMA. For example, patients who left the unit without informing staff were considered to have given no warning of AMA. Alternatively, when documentation was present stating that the patient had verbally expressed a desire to leave AMA, this was considered advanced warning of AMA.
Statistical Analysis
Continuous variables were reported as means and standard deviations. Categorical variables were reported as counts and percents. The independent samples t test was used for comparisons involving 2 groups and a second variable measured on a continuous scale. The 2 test was used to compare 2 categorical variables. Inferences were made at the 0.05 level of significance with no correction for multiple comparisons.
RESULTS
Mean age and gender distribution were similar to those reported in other AMA studies (Table 1).[3] Thirty‐day ED revisit and 30‐day hospital readmission frequencies for medical service patients were 121 (41.6%) and 88 (30.2%), respectively, also similar to those reported in other AMA studies.[3]
| Study Population, Mean SD or Count (%) | Hospital Population, Mean or Count (%) | |
|---|---|---|
| ||
| Age, y | 45.3 15.9 | 62.8 18.2 |
| Sex | ||
| Male | 168 (57.7) | 14,965 (43.6) |
| Female | 123 (42.3) | 19,333 (56.4) |
| Length of stay, d | 2.46 2.82 | 4.72 4.74 |
| 30‐day ED re‐encounter rate | 121 (41.6) | |
| 30‐day hospital readmission rate | 88 (30.2) | 4424 (12.9) |
| Prior AMA discharge | 49 (16.8) | |
Although our intent was to conduct a quantitative assessment of discharge interventions, we found stated reasons for leaving similar to those previously reported. In our study, AMA patients tended to be younger, more likely male, and at increased risk for AMA discharge if they had prior AMA discharges (Table 1). The most common reasons found in the medical record for leaving AMA were caring for sick family members, financial concerns, feeling better, and occasionally dissatisfaction with care, reasons similar to those reported in previous studies.[5, 7, 9, 10]
AMA notes were present in 276 (94.8%) charts. AMA notes were written by physicians in 163 (59.1%) and nurses in 110 (37.8%) encounters. The informed consent form was present in 88 (30.2%) charts, mentioned in the note but not present in the electronic medical record in 111 (38.1%), and not signed in 92 (31.6%) charts. Decision‐making capacity and health literacy were documented in 108 (37.1%) and 75 (25.8%) records, respectively. Warning of impending AMA was present in 217 (74.6%) charts. Medications prescribed and follow‐up plans were only documented in 71 (24.4%) and 91 (31.3%) charts, respectively (Table 2).
| Count (%) | |
|---|---|
| |
| AMA note present | 276 (94.8) |
| Primary AMA note author | |
| Physician | 163 (56) |
| Nurse, without physician note | 110 (37.8) |
| Other (ie, social worker) | 3 (1.0) |
| Warning of impending AMA | 217 (74.6) |
| Informed consent signed | |
| Yes | 88 (30.2) |
| No | 92 (31.6) |
| Absenta | 111 (38.1) |
| Documentation of decision‐making capacity | 108 (37.1) |
| Documentation of health literacy | 75 (25.8) |
| Documentation of follow‐up plan | 91 (31.3) |
| Documentation of medications at discharge | 71 (24.4) |
Patients with documentation of medications given did not have decreased 30‐day ED revisits (33.8% vs 44.3%, P = 0.12) or 30‐day hospital readmission (23.9% vs 32.4%, P = 0.18). Similarly, there was no relationship between documentation of follow‐up plans and 30‐day ED revisits (37.4% vs 43.7%, P = 0.31) or 30‐day hospital readmission (29.7% vs 30.7%, P = 0.87). Finally, there was no relationship between physician versus nurse authorship of AMA notes and 30‐day ED revisits (37.4% vs 46.4%, P = 0.14) or 30‐day hospital readmission (28.2% vs 31.8%, P = 0.52) (Table 3).
| Yes, % | No, % | P Value | |
|---|---|---|---|
| |||
| Documentation of discharge medicationsa | |||
| 30‐day ED revisit | 33.8 | 44.3 | 0.12 |
| 30‐day rehospitalization | 23.9 | 32.4 | 0.18 |
| Documentation of follow‐up | |||
| 30‐day ED revisit | 37.4 | 43.7 | 0.31 |
| 30‐day rehospitalization | 29.7 | 30.7 | 0.87 |
| Physician author of AMA note | |||
| 30‐day ED revisit | 37.4 | 46.4 | 0.14 |
| 30‐day rehospitalization | 28.2 | 31.8 | 0.52 |
| Documentation of medications | 36.2 | 10.0 | 0.001 |
| Documentation of follow‐up plan | 43.6 | 16.4 | 0.001 |
| Warning of AMA | |||
| Documentation of medications | 30.4 | 6.8 | 0.001 |
| Documentation of follow‐up plan | 37.3 | 13.5 | 0.001 |
Physician documentation of the AMA was associated with an increased frequency of discharge medication being prescribed (36.2% vs 10.0%, P 0.001) and with an increased finding of documented follow‐up plans (43.6% vs 16.4%, P 0.001). A documented warning of impending AMA was associated with an increased frequency of discharge medication being prescribed (30.4% vs 6.8%, P 0.001) and increased frequency of follow‐up plans being documented (37.3% vs 13.5%, P 0.001) (Table 3).
DISCUSSION
To gain insights into opportunities for discharge transition interventions in this potentially more vulnerable population,[1, 5] we analyzed AMA documentation and what interventions were carried out at the time of discharge. Our intent was a quantitative assessment of discharge interventions, but we also found stated reasons for leaving AMA that were similar to those previously reported.[5, 6, 10]
We identified several opportunities for improved documentation as well as targeted discharge intervention among AMA patients. Documentation in the charts of AMA patients was often suboptimal. In our study, a physician's AMA note was present only half of the time. Mention of the patient's mental status or health literacy was present in only one‐fourth of cases. Protection from litigation in AMA cases is enhanced when these elements and others, like informed consent, are present in the medical record.[11]
Physician documentation of the AMA was associated with an increased frequency of discharge medication being prescribed and with an increased finding of documented follow‐up plans. This association might be confounded by the fact that physicians can prescribe whereas most nurses cannot. The findings that a documented warning of impending AMA was associated with an increased frequency of discharge medication being prescribed (30.4% vs 6.8%, P 0.001) and increased frequency of follow‐up plans being documented (37.3% vs 13.5%, P 0.001) suggest opportunities for improvement through early inquiry about potential for AMA as well as early responses when patients threaten to leave AMA.
An important focus of our study was on documentation of discharge medications and follow‐up plans. These elements were documented in 31% and 25% of charts, respectively. A warning of impending AMA was present in 74.6% of encounters, yet medications and follow‐up plans were documented at a much lower rate. This represents an area where caregivers have the possibility to intervene, but are not documenting that they are doing so. We found no relationship between the documentation of giving prescriptions and giving explicit follow‐up plans with decreased rates of return to the ED or readmission, but that possibility may still warrant future prospective study.
Our study did not attempt to explain why only a minority of AMA discharges include medication prescription or follow‐up plans, but a number of potential explanations are possible. Some AMA discharges may occur unannounced with a patient simply walking off the ward giving little or no advance notice. It is also possible that provider perceptions and attitudes toward AMA patients may influence potential interventions.[12] An AMA discharge is against the caregiver's preferred advice for the patient, and it may seem illogical to offer patients second‐best advice. Perhaps some providers have the misconception that medications cannot or must not be prescribed for an AMA discharge. However, second‐best therapy may be better than no therapy, and some follow‐up better than no follow‐up plan.
Given the high rates of ED return and 30‐day readmission, the associated increased healthcare costs as well as increased morbidity and mortality associated with AMA dispositions, a continued search for effective intervention strategies and opportunities is warranted. Recently, programs for transition of care/discharge have demonstrated improved outcomes including reduced rates of readmission with standard discharges.[13] At the time of our study, effective programs such as Project BOOST (Better Outcomes for Older adult through Safe Transitions),[14] the Care Transitions Program,[15] and RED (Project Re‐engineered Discharge)[16] were not yet routinely employed, but their common elements may be applicable to the AMA population. In general, these programs focus on elements we investigated (patient understanding, follow‐up plans, medications prescribed) but add a number of additional components. Additional elements include written discharge instructions, patient education, teach‐back process, decision support, emergency plans, caregiver education, telephone follow‐up, and transition coaches to coordinate home and office follow‐up visits. Most potential interventions add significant time (and cost) to the discharge process. Thus, future studies applying these components to AMA discharges should emphasize timely identification of threatened AMA and prioritized interventions. Future studies should focus on which interventions are the most cost‐effective with AMA patients.
Limitations of our study include not being able to access information from area hospitals not in our hospital network, and thus we may not have identified all ED returns and readmissions. Additionally, interventions at the time of discharge (like prescription of medications or provider assessment of decision‐making capacity) may not have been documented and thus not available for our review. Also, our study was a retrospective review at a single institution and included a relatively small population of patients; consequently, our findings may not apply to other healthcare providers in other hospitals or settings. Our study was strengthened by reviewing all consecutive AMA cases over a 2‐year period encompassing a diverse group of healthcare providers.
CONCLUSION
In the majority of cases reviewed, some advance warning of impending AMA is apparent, affording an opportunity for interventions that may improve health outcomes. Despite this advance warning, only a minority of cases result in key interventions such as prescription of medications or development of follow‐up plans. Medical documentation of AMA dispositions is often inadequate, suggesting missed opportunities for potential intervention as well as suboptimal medicolegal scenarios. Future prospective studies examining cost‐effective interventions at the time of AMA discharge and transition of care may provide valuable insight into lowering rates of ED return and rehospitalization.
Disclosures: The views and opinions expressed in this article are those of the author(s) and do not reflect official policy or position of the United States Air Force, Department of Defense, or US government. The authors report no conflicts of interest.
Approximately 1% to 2% of inpatient stays result in discharges against medical advice (AMA).[1] Though relatively infrequent, AMA discharges warrant attention as they are associated with higher morbidity, increased risk of readmission, and greater 30‐day mortality.[2] A recent study found a 30‐day readmission rate among AMA patients of 24.5%, nearly twice that of matched non‐AMA patients, and a 30‐day mortality rate of 1.3%, also nearly double that of planned discharges.[3] Discharges AMA may be expected to decrease index length of stay, yet accounting for 30‐day readmissions they are estimated to increase costs 56% higher than expected from an initial hospitalization.[4] Patients note several possible reasons for leaving AMA including family emergencies, dissatisfaction with care, financial concerns, or simply feeling better, among others.[5, 6, 7] Risk factors for AMA discharges include previous AMA discharge, having no primary care physician, younger age, lack of insurance, male sex, substance abuse, and lower socioeconomic status.[4, 6, 7, 8]
A number of prior studies have assessed risk factors for AMA discharges, the long‐ and short‐term outcomes, patient reasons for leaving, and physician perceptions of why patients leave AMA.[3, 5, 7, 9] However, there is limited information about opportunities for discharge transition interventions in this potentially more vulnerable population. Because of the increased short‐term and long‐term risks to these patients, treatment and follow‐up plans at the time of discharge may carry even greater importance than follow‐up plans with standard discharges. This study analyzed AMA documentation and what interventions were carried out at the time of discharge.
METHODS
We reviewed the records of all adult patients, ages 18 years and older, admitted to a university‐affiliated tertiary care hospital in Dayton, Ohio (a 520‐bed hospital with approximately 17,000 adult patient encounters per year) over a 2‐year period, and who subsequently left AMA. A hospital database identified 351 adult AMA cases (1.0% of adult admissions). A single reviewer performed an in‐depth review of the 291 patient admissions to the general medical service between January 1, 2009 and December 31, 2010, and manually reviewed and abstracted the data of interest. The Wright State University institutional review board approved the study.
Documentation review focused on the presence of a specified AMA note, the presence of documentation addressing informed consent, patient decision‐making capacity, patient health literacy, follow‐up plans, whether or not medications were prescribed, and whether or not any warning indicators of impending AMA were apparent. These items represented key elements of the discharge policy and procedure in place at our institution during the period of study. We speculated that nurses may be more immediately available at the time of AMA discharge and thus might carry out AMA documentation more often than physicians. To assess this we recorded the role (nurse vs provider) of the writer of AMA notes. We also assessed patient gender, length of stay, prior AMA, 30‐day emergency department (ED) re‐encounters, and 30‐day hospital readmission after AMA discharge.
Informed consent was deemed present if patients signed the hospital's standardized AMA form. Decision‐making capacity was assessed as present if there was specific mention of the patient's capacity on the day of discharge. Any mention of health literacy or the patient's stated understanding of his medical condition at any time during the hospitalization was considered positive documentation of healthcare literacy. Follow‐up plans included any mention of where and when the patient would return. Discharge medications included prescribed medication or indication that no medications were warranted. Warning indicators included specific mention of the patient's desire to leave AMA. For example, patients who left the unit without informing staff were considered to have given no warning of AMA. Alternatively, when documentation was present stating that the patient had verbally expressed a desire to leave AMA, this was considered advanced warning of AMA.
Statistical Analysis
Continuous variables were reported as means and standard deviations. Categorical variables were reported as counts and percents. The independent samples t test was used for comparisons involving 2 groups and a second variable measured on a continuous scale. The 2 test was used to compare 2 categorical variables. Inferences were made at the 0.05 level of significance with no correction for multiple comparisons.
RESULTS
Mean age and gender distribution were similar to those reported in other AMA studies (Table 1).[3] Thirty‐day ED revisit and 30‐day hospital readmission frequencies for medical service patients were 121 (41.6%) and 88 (30.2%), respectively, also similar to those reported in other AMA studies.[3]
| Study Population, Mean SD or Count (%) | Hospital Population, Mean or Count (%) | |
|---|---|---|
| ||
| Age, y | 45.3 15.9 | 62.8 18.2 |
| Sex | ||
| Male | 168 (57.7) | 14,965 (43.6) |
| Female | 123 (42.3) | 19,333 (56.4) |
| Length of stay, d | 2.46 2.82 | 4.72 4.74 |
| 30‐day ED re‐encounter rate | 121 (41.6) | |
| 30‐day hospital readmission rate | 88 (30.2) | 4424 (12.9) |
| Prior AMA discharge | 49 (16.8) | |
Although our intent was to conduct a quantitative assessment of discharge interventions, we found stated reasons for leaving similar to those previously reported. In our study, AMA patients tended to be younger, more likely male, and at increased risk for AMA discharge if they had prior AMA discharges (Table 1). The most common reasons found in the medical record for leaving AMA were caring for sick family members, financial concerns, feeling better, and occasionally dissatisfaction with care, reasons similar to those reported in previous studies.[5, 7, 9, 10]
AMA notes were present in 276 (94.8%) charts. AMA notes were written by physicians in 163 (59.1%) and nurses in 110 (37.8%) encounters. The informed consent form was present in 88 (30.2%) charts, mentioned in the note but not present in the electronic medical record in 111 (38.1%), and not signed in 92 (31.6%) charts. Decision‐making capacity and health literacy were documented in 108 (37.1%) and 75 (25.8%) records, respectively. Warning of impending AMA was present in 217 (74.6%) charts. Medications prescribed and follow‐up plans were only documented in 71 (24.4%) and 91 (31.3%) charts, respectively (Table 2).
| Count (%) | |
|---|---|
| |
| AMA note present | 276 (94.8) |
| Primary AMA note author | |
| Physician | 163 (56) |
| Nurse, without physician note | 110 (37.8) |
| Other (ie, social worker) | 3 (1.0) |
| Warning of impending AMA | 217 (74.6) |
| Informed consent signed | |
| Yes | 88 (30.2) |
| No | 92 (31.6) |
| Absenta | 111 (38.1) |
| Documentation of decision‐making capacity | 108 (37.1) |
| Documentation of health literacy | 75 (25.8) |
| Documentation of follow‐up plan | 91 (31.3) |
| Documentation of medications at discharge | 71 (24.4) |
Patients with documentation of medications given did not have decreased 30‐day ED revisits (33.8% vs 44.3%, P = 0.12) or 30‐day hospital readmission (23.9% vs 32.4%, P = 0.18). Similarly, there was no relationship between documentation of follow‐up plans and 30‐day ED revisits (37.4% vs 43.7%, P = 0.31) or 30‐day hospital readmission (29.7% vs 30.7%, P = 0.87). Finally, there was no relationship between physician versus nurse authorship of AMA notes and 30‐day ED revisits (37.4% vs 46.4%, P = 0.14) or 30‐day hospital readmission (28.2% vs 31.8%, P = 0.52) (Table 3).
| Yes, % | No, % | P Value | |
|---|---|---|---|
| |||
| Documentation of discharge medicationsa | |||
| 30‐day ED revisit | 33.8 | 44.3 | 0.12 |
| 30‐day rehospitalization | 23.9 | 32.4 | 0.18 |
| Documentation of follow‐up | |||
| 30‐day ED revisit | 37.4 | 43.7 | 0.31 |
| 30‐day rehospitalization | 29.7 | 30.7 | 0.87 |
| Physician author of AMA note | |||
| 30‐day ED revisit | 37.4 | 46.4 | 0.14 |
| 30‐day rehospitalization | 28.2 | 31.8 | 0.52 |
| Documentation of medications | 36.2 | 10.0 | 0.001 |
| Documentation of follow‐up plan | 43.6 | 16.4 | 0.001 |
| Warning of AMA | |||
| Documentation of medications | 30.4 | 6.8 | 0.001 |
| Documentation of follow‐up plan | 37.3 | 13.5 | 0.001 |
Physician documentation of the AMA was associated with an increased frequency of discharge medication being prescribed (36.2% vs 10.0%, P 0.001) and with an increased finding of documented follow‐up plans (43.6% vs 16.4%, P 0.001). A documented warning of impending AMA was associated with an increased frequency of discharge medication being prescribed (30.4% vs 6.8%, P 0.001) and increased frequency of follow‐up plans being documented (37.3% vs 13.5%, P 0.001) (Table 3).
DISCUSSION
To gain insights into opportunities for discharge transition interventions in this potentially more vulnerable population,[1, 5] we analyzed AMA documentation and what interventions were carried out at the time of discharge. Our intent was a quantitative assessment of discharge interventions, but we also found stated reasons for leaving AMA that were similar to those previously reported.[5, 6, 10]
We identified several opportunities for improved documentation as well as targeted discharge intervention among AMA patients. Documentation in the charts of AMA patients was often suboptimal. In our study, a physician's AMA note was present only half of the time. Mention of the patient's mental status or health literacy was present in only one‐fourth of cases. Protection from litigation in AMA cases is enhanced when these elements and others, like informed consent, are present in the medical record.[11]
Physician documentation of the AMA was associated with an increased frequency of discharge medication being prescribed and with an increased finding of documented follow‐up plans. This association might be confounded by the fact that physicians can prescribe whereas most nurses cannot. The findings that a documented warning of impending AMA was associated with an increased frequency of discharge medication being prescribed (30.4% vs 6.8%, P 0.001) and increased frequency of follow‐up plans being documented (37.3% vs 13.5%, P 0.001) suggest opportunities for improvement through early inquiry about potential for AMA as well as early responses when patients threaten to leave AMA.
An important focus of our study was on documentation of discharge medications and follow‐up plans. These elements were documented in 31% and 25% of charts, respectively. A warning of impending AMA was present in 74.6% of encounters, yet medications and follow‐up plans were documented at a much lower rate. This represents an area where caregivers have the possibility to intervene, but are not documenting that they are doing so. We found no relationship between the documentation of giving prescriptions and giving explicit follow‐up plans with decreased rates of return to the ED or readmission, but that possibility may still warrant future prospective study.
Our study did not attempt to explain why only a minority of AMA discharges include medication prescription or follow‐up plans, but a number of potential explanations are possible. Some AMA discharges may occur unannounced with a patient simply walking off the ward giving little or no advance notice. It is also possible that provider perceptions and attitudes toward AMA patients may influence potential interventions.[12] An AMA discharge is against the caregiver's preferred advice for the patient, and it may seem illogical to offer patients second‐best advice. Perhaps some providers have the misconception that medications cannot or must not be prescribed for an AMA discharge. However, second‐best therapy may be better than no therapy, and some follow‐up better than no follow‐up plan.
Given the high rates of ED return and 30‐day readmission, the associated increased healthcare costs as well as increased morbidity and mortality associated with AMA dispositions, a continued search for effective intervention strategies and opportunities is warranted. Recently, programs for transition of care/discharge have demonstrated improved outcomes including reduced rates of readmission with standard discharges.[13] At the time of our study, effective programs such as Project BOOST (Better Outcomes for Older adult through Safe Transitions),[14] the Care Transitions Program,[15] and RED (Project Re‐engineered Discharge)[16] were not yet routinely employed, but their common elements may be applicable to the AMA population. In general, these programs focus on elements we investigated (patient understanding, follow‐up plans, medications prescribed) but add a number of additional components. Additional elements include written discharge instructions, patient education, teach‐back process, decision support, emergency plans, caregiver education, telephone follow‐up, and transition coaches to coordinate home and office follow‐up visits. Most potential interventions add significant time (and cost) to the discharge process. Thus, future studies applying these components to AMA discharges should emphasize timely identification of threatened AMA and prioritized interventions. Future studies should focus on which interventions are the most cost‐effective with AMA patients.
Limitations of our study include not being able to access information from area hospitals not in our hospital network, and thus we may not have identified all ED returns and readmissions. Additionally, interventions at the time of discharge (like prescription of medications or provider assessment of decision‐making capacity) may not have been documented and thus not available for our review. Also, our study was a retrospective review at a single institution and included a relatively small population of patients; consequently, our findings may not apply to other healthcare providers in other hospitals or settings. Our study was strengthened by reviewing all consecutive AMA cases over a 2‐year period encompassing a diverse group of healthcare providers.
CONCLUSION
In the majority of cases reviewed, some advance warning of impending AMA is apparent, affording an opportunity for interventions that may improve health outcomes. Despite this advance warning, only a minority of cases result in key interventions such as prescription of medications or development of follow‐up plans. Medical documentation of AMA dispositions is often inadequate, suggesting missed opportunities for potential intervention as well as suboptimal medicolegal scenarios. Future prospective studies examining cost‐effective interventions at the time of AMA discharge and transition of care may provide valuable insight into lowering rates of ED return and rehospitalization.
Disclosures: The views and opinions expressed in this article are those of the author(s) and do not reflect official policy or position of the United States Air Force, Department of Defense, or US government. The authors report no conflicts of interest.
Approximately 1% to 2% of inpatient stays result in discharges against medical advice (AMA).[1] Though relatively infrequent, AMA discharges warrant attention as they are associated with higher morbidity, increased risk of readmission, and greater 30‐day mortality.[2] A recent study found a 30‐day readmission rate among AMA patients of 24.5%, nearly twice that of matched non‐AMA patients, and a 30‐day mortality rate of 1.3%, also nearly double that of planned discharges.[3] Discharges AMA may be expected to decrease index length of stay, yet accounting for 30‐day readmissions they are estimated to increase costs 56% higher than expected from an initial hospitalization.[4] Patients note several possible reasons for leaving AMA including family emergencies, dissatisfaction with care, financial concerns, or simply feeling better, among others.[5, 6, 7] Risk factors for AMA discharges include previous AMA discharge, having no primary care physician, younger age, lack of insurance, male sex, substance abuse, and lower socioeconomic status.[4, 6, 7, 8]
A number of prior studies have assessed risk factors for AMA discharges, the long‐ and short‐term outcomes, patient reasons for leaving, and physician perceptions of why patients leave AMA.[3, 5, 7, 9] However, there is limited information about opportunities for discharge transition interventions in this potentially more vulnerable population. Because of the increased short‐term and long‐term risks to these patients, treatment and follow‐up plans at the time of discharge may carry even greater importance than follow‐up plans with standard discharges. This study analyzed AMA documentation and what interventions were carried out at the time of discharge.
METHODS
We reviewed the records of all adult patients, ages 18 years and older, admitted to a university‐affiliated tertiary care hospital in Dayton, Ohio (a 520‐bed hospital with approximately 17,000 adult patient encounters per year) over a 2‐year period, and who subsequently left AMA. A hospital database identified 351 adult AMA cases (1.0% of adult admissions). A single reviewer performed an in‐depth review of the 291 patient admissions to the general medical service between January 1, 2009 and December 31, 2010, and manually reviewed and abstracted the data of interest. The Wright State University institutional review board approved the study.
Documentation review focused on the presence of a specified AMA note, the presence of documentation addressing informed consent, patient decision‐making capacity, patient health literacy, follow‐up plans, whether or not medications were prescribed, and whether or not any warning indicators of impending AMA were apparent. These items represented key elements of the discharge policy and procedure in place at our institution during the period of study. We speculated that nurses may be more immediately available at the time of AMA discharge and thus might carry out AMA documentation more often than physicians. To assess this we recorded the role (nurse vs provider) of the writer of AMA notes. We also assessed patient gender, length of stay, prior AMA, 30‐day emergency department (ED) re‐encounters, and 30‐day hospital readmission after AMA discharge.
Informed consent was deemed present if patients signed the hospital's standardized AMA form. Decision‐making capacity was assessed as present if there was specific mention of the patient's capacity on the day of discharge. Any mention of health literacy or the patient's stated understanding of his medical condition at any time during the hospitalization was considered positive documentation of healthcare literacy. Follow‐up plans included any mention of where and when the patient would return. Discharge medications included prescribed medication or indication that no medications were warranted. Warning indicators included specific mention of the patient's desire to leave AMA. For example, patients who left the unit without informing staff were considered to have given no warning of AMA. Alternatively, when documentation was present stating that the patient had verbally expressed a desire to leave AMA, this was considered advanced warning of AMA.
Statistical Analysis
Continuous variables were reported as means and standard deviations. Categorical variables were reported as counts and percents. The independent samples t test was used for comparisons involving 2 groups and a second variable measured on a continuous scale. The 2 test was used to compare 2 categorical variables. Inferences were made at the 0.05 level of significance with no correction for multiple comparisons.
RESULTS
Mean age and gender distribution were similar to those reported in other AMA studies (Table 1).[3] Thirty‐day ED revisit and 30‐day hospital readmission frequencies for medical service patients were 121 (41.6%) and 88 (30.2%), respectively, also similar to those reported in other AMA studies.[3]
| Study Population, Mean SD or Count (%) | Hospital Population, Mean or Count (%) | |
|---|---|---|
| ||
| Age, y | 45.3 15.9 | 62.8 18.2 |
| Sex | ||
| Male | 168 (57.7) | 14,965 (43.6) |
| Female | 123 (42.3) | 19,333 (56.4) |
| Length of stay, d | 2.46 2.82 | 4.72 4.74 |
| 30‐day ED re‐encounter rate | 121 (41.6) | |
| 30‐day hospital readmission rate | 88 (30.2) | 4424 (12.9) |
| Prior AMA discharge | 49 (16.8) | |
Although our intent was to conduct a quantitative assessment of discharge interventions, we found stated reasons for leaving similar to those previously reported. In our study, AMA patients tended to be younger, more likely male, and at increased risk for AMA discharge if they had prior AMA discharges (Table 1). The most common reasons found in the medical record for leaving AMA were caring for sick family members, financial concerns, feeling better, and occasionally dissatisfaction with care, reasons similar to those reported in previous studies.[5, 7, 9, 10]
AMA notes were present in 276 (94.8%) charts. AMA notes were written by physicians in 163 (59.1%) and nurses in 110 (37.8%) encounters. The informed consent form was present in 88 (30.2%) charts, mentioned in the note but not present in the electronic medical record in 111 (38.1%), and not signed in 92 (31.6%) charts. Decision‐making capacity and health literacy were documented in 108 (37.1%) and 75 (25.8%) records, respectively. Warning of impending AMA was present in 217 (74.6%) charts. Medications prescribed and follow‐up plans were only documented in 71 (24.4%) and 91 (31.3%) charts, respectively (Table 2).
| Count (%) | |
|---|---|
| |
| AMA note present | 276 (94.8) |
| Primary AMA note author | |
| Physician | 163 (56) |
| Nurse, without physician note | 110 (37.8) |
| Other (ie, social worker) | 3 (1.0) |
| Warning of impending AMA | 217 (74.6) |
| Informed consent signed | |
| Yes | 88 (30.2) |
| No | 92 (31.6) |
| Absenta | 111 (38.1) |
| Documentation of decision‐making capacity | 108 (37.1) |
| Documentation of health literacy | 75 (25.8) |
| Documentation of follow‐up plan | 91 (31.3) |
| Documentation of medications at discharge | 71 (24.4) |
Patients with documentation of medications given did not have decreased 30‐day ED revisits (33.8% vs 44.3%, P = 0.12) or 30‐day hospital readmission (23.9% vs 32.4%, P = 0.18). Similarly, there was no relationship between documentation of follow‐up plans and 30‐day ED revisits (37.4% vs 43.7%, P = 0.31) or 30‐day hospital readmission (29.7% vs 30.7%, P = 0.87). Finally, there was no relationship between physician versus nurse authorship of AMA notes and 30‐day ED revisits (37.4% vs 46.4%, P = 0.14) or 30‐day hospital readmission (28.2% vs 31.8%, P = 0.52) (Table 3).
| Yes, % | No, % | P Value | |
|---|---|---|---|
| |||
| Documentation of discharge medicationsa | |||
| 30‐day ED revisit | 33.8 | 44.3 | 0.12 |
| 30‐day rehospitalization | 23.9 | 32.4 | 0.18 |
| Documentation of follow‐up | |||
| 30‐day ED revisit | 37.4 | 43.7 | 0.31 |
| 30‐day rehospitalization | 29.7 | 30.7 | 0.87 |
| Physician author of AMA note | |||
| 30‐day ED revisit | 37.4 | 46.4 | 0.14 |
| 30‐day rehospitalization | 28.2 | 31.8 | 0.52 |
| Documentation of medications | 36.2 | 10.0 | 0.001 |
| Documentation of follow‐up plan | 43.6 | 16.4 | 0.001 |
| Warning of AMA | |||
| Documentation of medications | 30.4 | 6.8 | 0.001 |
| Documentation of follow‐up plan | 37.3 | 13.5 | 0.001 |
Physician documentation of the AMA was associated with an increased frequency of discharge medication being prescribed (36.2% vs 10.0%, P 0.001) and with an increased finding of documented follow‐up plans (43.6% vs 16.4%, P 0.001). A documented warning of impending AMA was associated with an increased frequency of discharge medication being prescribed (30.4% vs 6.8%, P 0.001) and increased frequency of follow‐up plans being documented (37.3% vs 13.5%, P 0.001) (Table 3).
DISCUSSION
To gain insights into opportunities for discharge transition interventions in this potentially more vulnerable population,[1, 5] we analyzed AMA documentation and what interventions were carried out at the time of discharge. Our intent was a quantitative assessment of discharge interventions, but we also found stated reasons for leaving AMA that were similar to those previously reported.[5, 6, 10]
We identified several opportunities for improved documentation as well as targeted discharge intervention among AMA patients. Documentation in the charts of AMA patients was often suboptimal. In our study, a physician's AMA note was present only half of the time. Mention of the patient's mental status or health literacy was present in only one‐fourth of cases. Protection from litigation in AMA cases is enhanced when these elements and others, like informed consent, are present in the medical record.[11]
Physician documentation of the AMA was associated with an increased frequency of discharge medication being prescribed and with an increased finding of documented follow‐up plans. This association might be confounded by the fact that physicians can prescribe whereas most nurses cannot. The findings that a documented warning of impending AMA was associated with an increased frequency of discharge medication being prescribed (30.4% vs 6.8%, P 0.001) and increased frequency of follow‐up plans being documented (37.3% vs 13.5%, P 0.001) suggest opportunities for improvement through early inquiry about potential for AMA as well as early responses when patients threaten to leave AMA.
An important focus of our study was on documentation of discharge medications and follow‐up plans. These elements were documented in 31% and 25% of charts, respectively. A warning of impending AMA was present in 74.6% of encounters, yet medications and follow‐up plans were documented at a much lower rate. This represents an area where caregivers have the possibility to intervene, but are not documenting that they are doing so. We found no relationship between the documentation of giving prescriptions and giving explicit follow‐up plans with decreased rates of return to the ED or readmission, but that possibility may still warrant future prospective study.
Our study did not attempt to explain why only a minority of AMA discharges include medication prescription or follow‐up plans, but a number of potential explanations are possible. Some AMA discharges may occur unannounced with a patient simply walking off the ward giving little or no advance notice. It is also possible that provider perceptions and attitudes toward AMA patients may influence potential interventions.[12] An AMA discharge is against the caregiver's preferred advice for the patient, and it may seem illogical to offer patients second‐best advice. Perhaps some providers have the misconception that medications cannot or must not be prescribed for an AMA discharge. However, second‐best therapy may be better than no therapy, and some follow‐up better than no follow‐up plan.
Given the high rates of ED return and 30‐day readmission, the associated increased healthcare costs as well as increased morbidity and mortality associated with AMA dispositions, a continued search for effective intervention strategies and opportunities is warranted. Recently, programs for transition of care/discharge have demonstrated improved outcomes including reduced rates of readmission with standard discharges.[13] At the time of our study, effective programs such as Project BOOST (Better Outcomes for Older adult through Safe Transitions),[14] the Care Transitions Program,[15] and RED (Project Re‐engineered Discharge)[16] were not yet routinely employed, but their common elements may be applicable to the AMA population. In general, these programs focus on elements we investigated (patient understanding, follow‐up plans, medications prescribed) but add a number of additional components. Additional elements include written discharge instructions, patient education, teach‐back process, decision support, emergency plans, caregiver education, telephone follow‐up, and transition coaches to coordinate home and office follow‐up visits. Most potential interventions add significant time (and cost) to the discharge process. Thus, future studies applying these components to AMA discharges should emphasize timely identification of threatened AMA and prioritized interventions. Future studies should focus on which interventions are the most cost‐effective with AMA patients.
Limitations of our study include not being able to access information from area hospitals not in our hospital network, and thus we may not have identified all ED returns and readmissions. Additionally, interventions at the time of discharge (like prescription of medications or provider assessment of decision‐making capacity) may not have been documented and thus not available for our review. Also, our study was a retrospective review at a single institution and included a relatively small population of patients; consequently, our findings may not apply to other healthcare providers in other hospitals or settings. Our study was strengthened by reviewing all consecutive AMA cases over a 2‐year period encompassing a diverse group of healthcare providers.
CONCLUSION
In the majority of cases reviewed, some advance warning of impending AMA is apparent, affording an opportunity for interventions that may improve health outcomes. Despite this advance warning, only a minority of cases result in key interventions such as prescription of medications or development of follow‐up plans. Medical documentation of AMA dispositions is often inadequate, suggesting missed opportunities for potential intervention as well as suboptimal medicolegal scenarios. Future prospective studies examining cost‐effective interventions at the time of AMA discharge and transition of care may provide valuable insight into lowering rates of ED return and rehospitalization.
Disclosures: The views and opinions expressed in this article are those of the author(s) and do not reflect official policy or position of the United States Air Force, Department of Defense, or US government. The authors report no conflicts of interest.
Postdischarge Clinics
Transitions of care, which encompass the patient experience of hospital discharge to the community, are frequently associated with clinically and financially costly adverse events.[1, 2] One important element for reducing the risk of postdischarge adverse events is provision of timely follow‐up by a clinician familiar with the patient and hospital course.[3, 4]
However, achieving this ideal is becoming more difficult because of an increased demand for primary care services (due to expanding coverage of Medicare and Medicaid) and the decreased supply of primary care physicians.[5, 6] When a timely visit with a clinician is available postdischarge, the widening discontinuity between inpatient and outpatient care providers often means this clinician is lacking essential details of the hospitalization.[7, 8]
One increasingly common innovation to improve postdischarge care access and continuity is to extend the role of inpatient providers (usually hospitalists) to provide care after discharge in a postdischarge clinic (PDC).[9, 10, 11] These clinics require an expansion of a hospitalist's duties to the outpatient setting, a requirement that has met with hospitalist resistance in initial reports.[12] However, little is known about hospitalists' experience with PDCs or attitudes toward postdischarge care. We aimed to explore these attitudes and experiences surrounding postdischarge care and PDCs.
METHODS
We conducted a cross‐sectional 17‐question Web‐based survey of hospitalists at 20 academic and 17 VA medical centers across the United States. Hospital medicine faculty at each site were identified by their group leader; members of each group then received an email survey up to 3 times. To collect responses from nonacademic hospitalists, the survey was also distributed to a large national private hospitalist employer. Due to internal limitations at the employer site, sampling was not feasible, and thus a convenience sample was obtained. Hospitalists who were not clinically active or did not have computer access to complete the survey were excluded. Responses were initially gathered on a 4‐point Likert scale; for comparisons between groups the scale was collapsed to a binary comparison using Fisher exact or 2 tests. We included questions answered in partially completed surveys in both the numerator and denominator; questions not answered were excluded from both numerator and denominator. The denominator of all responses was noted. All analyses were conducted using SAS 9.3 (SAS Institute, Inc., Cary, NC). The study was approved by the Colorado Multiple Institutional Review Board.
RESULTS
Of 814 hospitalists, 228 responded to the survey (28.3%). Table 1 illustrates characteristics of responding hospitalists, who were divided between university hospitals, community teaching hospitals, and community nonteaching hospitals in diverse practices in terms of location and group size.
| Characteristic | Respondents, No. (%) |
|---|---|
| |
| Employing institution | |
| University hospital | 79 (37.4) |
| Community, nonteaching | 62 (29.4) |
| Community, teaching | 70 (33.2) |
| Care environment | |
| Hospitalist providers | 98 (46.4) |
| Housestaff providers | 94 (44.6) |
| Primary care providers | 10 (4.7) |
| Combination | 9 (4.3) |
| Hospitalist group size (number of hospitalists) | |
| 15 | 39 (18.5) |
| 610 | 50 (23.7) |
| 1120 | 52 (24.6) |
| 2150 | 59 (28.0) |
| >50 | 9 (4.3) |
| Hospital locationpopulation | |
| Rural | 20 (9.5) |
| Suburban | 47 (22.3) |
| Urban | 144 (68.2) |
| Hospital locationgeographic | |
| West Coast | 11 (5.2) |
| Midwest | 47 (22.3) |
| Southern | 57 (27.1) |
| East Coast | 21 (10) |
| Southwest | 36 (17.1) |
| Mountain | 32 (15.2) |
Sixty‐one percent of responding hospitalists believed most patient problems after discharge were due to poor follow‐up with primary care providers, and 55% found it difficult to arrange timely primary care follow up (Table 2). Despite this, 87% thought patient problems after discharge should be cared for by primary care physicians, and 62% opposed the idea of hospitalists seeing patients in the clinic after discharge.
| Agree, No. (%) | Disagree, No. (%) | |
|---|---|---|
| ||
| Hospitalists should see patients in clinic after discharge | 87 (38.2) | 141 (61.8) |
| Primary care responsible for problems after discharge | 198 (86.8) | 30 (13.2) |
| Hospitalists responsible for patients after discharge | 113 (49.6) | 115 (50.4) |
| Would welcome a PDC if employer required | 113 (49.6) | 115 (50.4) |
| Would require extra compensation to work in a PDC | 175 (76.8) | 53 (23.2) |
| Believe a PDC would reduce ED visits after discharge | 168 (73.7) | 60 (26.3) |
| Would discharge patients earlier if could see after discharge | 116 (50.9) | 112 (49.1) |
| Most postdischarge problems due to poor PCP access | 138 (60.5) | 90 (39.5) |
| Easy to arrange timely follow‐up with patient's PCP | 100 (44.2) | 126 (55.3) |
When asked if hospitalists were responsible for patients after discharge from the hospital, only 50% responded positively. However, when asked how long hospitalists were responsible for patients after discharge, 71% gave a response longer than hospital discharge, including 60% who believed this responsibility ended at 1 week or less following discharge. A minority (12%) felt it extended to 1 month following discharge (Table 3).
| Respondents, No. (%) | |
|---|---|
| |
| Length of time inpatient providers responsible after discharge | |
| Responsibility ends at time of discharge | 65 (28.5) |
| 13 days | 40 (17.5) |
| 47 days | 57 (25.0) |
| 2 weeks | 41 (18.0) |
| 4 weeks | 20 (8.8) |
| 3 months | 3 (1.3) |
| 3 months | 2 (0.9) |
| Postdischarge clinic present | 20 (8.8) |
| Considered starting a postdischarge clinica | 62 (30.5) |
| Starting in next yearb | 6 (3.3) |
| Are satisfied with experience in postdischarge clinicc | 17 (85) |
| Think patients are satisfied/highly satisfiedc | 14 (70) |
Responding hospitalists expressed confidence in a PDC to reduce postdischarge emergency department visits (74%). However, most felt they would require extra compensation to staff a PDC (77%). They were divided on whether they would discharge patients from the hospital earlier if they could see those patients in postdischarge follow‐up (51% would discharge patients earlier).
Compared to those who had not experienced a PDC, responding hospitalists who had provided care in a PDC trended toward responding more positively that hospitalists should provide postdischarge care (P = 0.054). Few responding hospitalists had such exposure (8.8%) at the time of the survey. Although 31% had considering starting a PDC, only 3% were starting in the next year. Of responding hospitalists with exposure to a PDC, 70% were satisfied with the experience and 85% felt their patients were satisfied. Responses did not vary by type of practice (academic vs nonacademic), group size, geographic location, or by exposure to a PDC except as above.
DISCUSSION
Responding hospitalists reported encountering significant difficulty arranging appropriate postdischarge appointments with primary care providers and feel this contributes to postdischarge complications. Nearly 75% of those surveyed felt a hospitalist‐run PDC would be effective in reducing postdischarge emergency department visits, presumably in part due to improved access to postdischarge care. However, 62% of responding hospitalists opposed providing this type of care, though those who had experienced a PDC were somewhat more likely to view providing care in a PDC favorably. Survey responses largely reflect attitudes rather than experience with PDCs, because very few respondents had ever worked in a PDC.
The juxtaposition of the confidence expressed in PDCs to reduce postdischarge emergency department visits with the less enthusiastic views of respondents about providing care in a PDC was surprising. Several explanations are possible. First, providing such care is outside the usual scope of practice of most hospitalists, and preliminary reports indicate hospitalists, as self‐selected inpatient providers, may not initially welcome this opportunity.[12] Second, responding hospitalists identified the need for extra compensation for providing this care, suggesting they would see staffing a PDC as a burden requiring extra payment. Third, only 12% of respondents felt their responsibility to their discharged patients extended to 1 month following discharge. Given this, hospitalists may not feel enough personal ownership over 30‐day readmission rates to justify the additional clinical demand of staffing a PDC.[13]
In fact, 29% of responding hospitalists felt their responsibility to the patient ended at the time of discharge. Respondents may have interpreted responsibility differently, and we cannot rule out response bias given our lower‐than‐expected response rate. However, we had anticipated many fewer hospitalists would respond this way given professional hospitalist societies have endorsed guidelines for improved transitions of care, which clearly delineate the key role hospitalists play in care transitions.[14]
Although fewer than 10% of respondents had worked in a PDC, nearly one‐third reported considering starting such a practice in the future, underscoring the importance of understanding hospitalist attitudes and experiences when creating a PDC and the significant barriers to arranging appropriate postdischarge care identified by survey respondents. The barriers to establishing a PDC may explain why few planned to start a PDC in the next year.
This study should be interpreted in the context of its design. Due to limitations in survey delivery, more rigorous sampling designs could not be used, and efforts were instead made to deliver the survey to a diverse group of hospitalists. The survey response rate was lower than anticipated and this increases the risk of response bias. Though this response rate is characteristic of other surveys of hospitalists, responses may have been from a selected population and therefore not representative of all hospitalists.[15] We sampled from a variety of practice venues, locations, academic and community practices, and practice group sizes to try to minimize this bias. Due to the low exposure rate to PDCs, hospitalist responses to experiences with PDCs should be considered exploratory.
We asked about similar content areas in the survey in multiple questions to maximize content validity; this resulted in variations in the degree of agreement or disagreement to similar prompts. For example, 62% of hospitalists opposed seeing patients in the clinic after discharge when directly asked, but nearly 50% said they would welcome the opportunity to work in a PDC if their employer required it. In another example, 50% of respondents said their responsibility for the patient ended at time of discharge, but when asked about duration of responsibility, 30% identified time of discharge as the limit. When reporting and interpreting results, we have tried to highlight responses to questions that ask most clearly and directly about the content of interest (rather than general themes), but this interpretation may also be subject to bias.
The time after hospital discharge is one of heightened risk for adverse events for the recently discharged patient. Hospitalist‐run postdischarge clinics may offer improved postdischarge care access and continuity; more research is needed on the effects of such clinics on patient outcomes, including postdischarge utilization. Until then, physicians and hospitals considering establishing PDCs should consider the barriers responding hospitalists identified to working in such a clinic, as well as the confidence they expressed in PDCs to reduce subsequent utilization.
Disclosures: Dr. Burke had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. The views expressed in this article are those of the authors and do not necessarily represent the views of the Department of Veterans Affairs. Dr. Burke was supported by grant funding from the Colorado Research Enhancement Award Program to Improve Care Coordination for Veterans. Dr. Ryan has no conflicts of interest to disclose.
- , , , , . The incidence and severity of adverse events affecting patients after discharge from the hospital. Ann Intern Med. 2003;138(3):161–167.
- , , . Rehospitalizations among patients in the Medicare fee‐for‐service program. N Engl J Med. 2009;360(14):1418–1428.
- , , , et al. Relationship between early physician follow‐up and 30‐day readmission among Medicare beneficiaries hospitalized for heart failure. JAMA. 2010;303(17):1716–1722.
- , , . Post‐hospitalization transitions: Examining the effects of timing of primary care provider follow‐up. J Hosp Med. 2010;5(7):392–397.
- , , . Will generalist physician supply meet demands of an increasing and aging population? Health Aff (Millwood). 2008;27(3):w232–w241.
- Association of American Medical Colleges. June 2010.The impact of health care reform on the future supply and demand for physicians updated projections through 2025. Available at: http://www.aamc.org/download/158076/data/updated_projections_through_2025.pdf. Accessed May 1, 2012.
- , , , , , . Continuity of outpatient and inpatient care by primary care physicians for hospitalized older adults. JAMA. 2009;301(16):1671–1680.
- , , , , . Trends in inpatient continuity of care for a cohort of Medicare patients 1996–2006. J Hosp Med. 2011;6(8):438–444.
- . Is a post‐discharge clinic in your hospital's future? The Hospitalist. December 2011 Available at: http://www.the‐hospitalist.org/details/article/1409011/Is_a_Post_Discharge_Clinic_in_Your_Hospitals_future.html. Accessed May 1, 2013.
- , , , . Continuity of care and patient outcomes after hospital discharge. J Gen Intern Med. 2004;19(6):624–631.
- , , , . Effects of a postdischarge clinic on housestaff satisfaction and utilization of hospital services. J Gen Intern Med. 1996;11(3):179–181.
- . Interval examination: establishment of a hospitalist‐staffed discharge clinic. J Gen Intern Med. 2012;27(10):1377–1382.
- Patient Protection and Affordable Care Act (PPACA).Public Law 111–148 2010. Available at: http://www.gpo.gov/fdsys/pkg/PLAW‐111publ148/pdf/PLAW‐111publ148.pdf. Accessed January 10, 2013.
- , , , et al. Transitions of Care Consensus policy statement: American College of Physicians, Society of General Internal Medicine, Society of Hospital Medicine, American Geriatrics Society, American College of Emergency Physicians, and Society for Academic Emergency Medicine. J Hosp Med. 2009;4(6):364–370.
- , , , , . Person‐job fit: an exploratory cross‐sectional analysis of hospitalists. J Hosp Med. 2013;8(2):96–101.
Transitions of care, which encompass the patient experience of hospital discharge to the community, are frequently associated with clinically and financially costly adverse events.[1, 2] One important element for reducing the risk of postdischarge adverse events is provision of timely follow‐up by a clinician familiar with the patient and hospital course.[3, 4]
However, achieving this ideal is becoming more difficult because of an increased demand for primary care services (due to expanding coverage of Medicare and Medicaid) and the decreased supply of primary care physicians.[5, 6] When a timely visit with a clinician is available postdischarge, the widening discontinuity between inpatient and outpatient care providers often means this clinician is lacking essential details of the hospitalization.[7, 8]
One increasingly common innovation to improve postdischarge care access and continuity is to extend the role of inpatient providers (usually hospitalists) to provide care after discharge in a postdischarge clinic (PDC).[9, 10, 11] These clinics require an expansion of a hospitalist's duties to the outpatient setting, a requirement that has met with hospitalist resistance in initial reports.[12] However, little is known about hospitalists' experience with PDCs or attitudes toward postdischarge care. We aimed to explore these attitudes and experiences surrounding postdischarge care and PDCs.
METHODS
We conducted a cross‐sectional 17‐question Web‐based survey of hospitalists at 20 academic and 17 VA medical centers across the United States. Hospital medicine faculty at each site were identified by their group leader; members of each group then received an email survey up to 3 times. To collect responses from nonacademic hospitalists, the survey was also distributed to a large national private hospitalist employer. Due to internal limitations at the employer site, sampling was not feasible, and thus a convenience sample was obtained. Hospitalists who were not clinically active or did not have computer access to complete the survey were excluded. Responses were initially gathered on a 4‐point Likert scale; for comparisons between groups the scale was collapsed to a binary comparison using Fisher exact or 2 tests. We included questions answered in partially completed surveys in both the numerator and denominator; questions not answered were excluded from both numerator and denominator. The denominator of all responses was noted. All analyses were conducted using SAS 9.3 (SAS Institute, Inc., Cary, NC). The study was approved by the Colorado Multiple Institutional Review Board.
RESULTS
Of 814 hospitalists, 228 responded to the survey (28.3%). Table 1 illustrates characteristics of responding hospitalists, who were divided between university hospitals, community teaching hospitals, and community nonteaching hospitals in diverse practices in terms of location and group size.
| Characteristic | Respondents, No. (%) |
|---|---|
| |
| Employing institution | |
| University hospital | 79 (37.4) |
| Community, nonteaching | 62 (29.4) |
| Community, teaching | 70 (33.2) |
| Care environment | |
| Hospitalist providers | 98 (46.4) |
| Housestaff providers | 94 (44.6) |
| Primary care providers | 10 (4.7) |
| Combination | 9 (4.3) |
| Hospitalist group size (number of hospitalists) | |
| 15 | 39 (18.5) |
| 610 | 50 (23.7) |
| 1120 | 52 (24.6) |
| 2150 | 59 (28.0) |
| >50 | 9 (4.3) |
| Hospital locationpopulation | |
| Rural | 20 (9.5) |
| Suburban | 47 (22.3) |
| Urban | 144 (68.2) |
| Hospital locationgeographic | |
| West Coast | 11 (5.2) |
| Midwest | 47 (22.3) |
| Southern | 57 (27.1) |
| East Coast | 21 (10) |
| Southwest | 36 (17.1) |
| Mountain | 32 (15.2) |
Sixty‐one percent of responding hospitalists believed most patient problems after discharge were due to poor follow‐up with primary care providers, and 55% found it difficult to arrange timely primary care follow up (Table 2). Despite this, 87% thought patient problems after discharge should be cared for by primary care physicians, and 62% opposed the idea of hospitalists seeing patients in the clinic after discharge.
| Agree, No. (%) | Disagree, No. (%) | |
|---|---|---|
| ||
| Hospitalists should see patients in clinic after discharge | 87 (38.2) | 141 (61.8) |
| Primary care responsible for problems after discharge | 198 (86.8) | 30 (13.2) |
| Hospitalists responsible for patients after discharge | 113 (49.6) | 115 (50.4) |
| Would welcome a PDC if employer required | 113 (49.6) | 115 (50.4) |
| Would require extra compensation to work in a PDC | 175 (76.8) | 53 (23.2) |
| Believe a PDC would reduce ED visits after discharge | 168 (73.7) | 60 (26.3) |
| Would discharge patients earlier if could see after discharge | 116 (50.9) | 112 (49.1) |
| Most postdischarge problems due to poor PCP access | 138 (60.5) | 90 (39.5) |
| Easy to arrange timely follow‐up with patient's PCP | 100 (44.2) | 126 (55.3) |
When asked if hospitalists were responsible for patients after discharge from the hospital, only 50% responded positively. However, when asked how long hospitalists were responsible for patients after discharge, 71% gave a response longer than hospital discharge, including 60% who believed this responsibility ended at 1 week or less following discharge. A minority (12%) felt it extended to 1 month following discharge (Table 3).
| Respondents, No. (%) | |
|---|---|
| |
| Length of time inpatient providers responsible after discharge | |
| Responsibility ends at time of discharge | 65 (28.5) |
| 13 days | 40 (17.5) |
| 47 days | 57 (25.0) |
| 2 weeks | 41 (18.0) |
| 4 weeks | 20 (8.8) |
| 3 months | 3 (1.3) |
| 3 months | 2 (0.9) |
| Postdischarge clinic present | 20 (8.8) |
| Considered starting a postdischarge clinica | 62 (30.5) |
| Starting in next yearb | 6 (3.3) |
| Are satisfied with experience in postdischarge clinicc | 17 (85) |
| Think patients are satisfied/highly satisfiedc | 14 (70) |
Responding hospitalists expressed confidence in a PDC to reduce postdischarge emergency department visits (74%). However, most felt they would require extra compensation to staff a PDC (77%). They were divided on whether they would discharge patients from the hospital earlier if they could see those patients in postdischarge follow‐up (51% would discharge patients earlier).
Compared to those who had not experienced a PDC, responding hospitalists who had provided care in a PDC trended toward responding more positively that hospitalists should provide postdischarge care (P = 0.054). Few responding hospitalists had such exposure (8.8%) at the time of the survey. Although 31% had considering starting a PDC, only 3% were starting in the next year. Of responding hospitalists with exposure to a PDC, 70% were satisfied with the experience and 85% felt their patients were satisfied. Responses did not vary by type of practice (academic vs nonacademic), group size, geographic location, or by exposure to a PDC except as above.
DISCUSSION
Responding hospitalists reported encountering significant difficulty arranging appropriate postdischarge appointments with primary care providers and feel this contributes to postdischarge complications. Nearly 75% of those surveyed felt a hospitalist‐run PDC would be effective in reducing postdischarge emergency department visits, presumably in part due to improved access to postdischarge care. However, 62% of responding hospitalists opposed providing this type of care, though those who had experienced a PDC were somewhat more likely to view providing care in a PDC favorably. Survey responses largely reflect attitudes rather than experience with PDCs, because very few respondents had ever worked in a PDC.
The juxtaposition of the confidence expressed in PDCs to reduce postdischarge emergency department visits with the less enthusiastic views of respondents about providing care in a PDC was surprising. Several explanations are possible. First, providing such care is outside the usual scope of practice of most hospitalists, and preliminary reports indicate hospitalists, as self‐selected inpatient providers, may not initially welcome this opportunity.[12] Second, responding hospitalists identified the need for extra compensation for providing this care, suggesting they would see staffing a PDC as a burden requiring extra payment. Third, only 12% of respondents felt their responsibility to their discharged patients extended to 1 month following discharge. Given this, hospitalists may not feel enough personal ownership over 30‐day readmission rates to justify the additional clinical demand of staffing a PDC.[13]
In fact, 29% of responding hospitalists felt their responsibility to the patient ended at the time of discharge. Respondents may have interpreted responsibility differently, and we cannot rule out response bias given our lower‐than‐expected response rate. However, we had anticipated many fewer hospitalists would respond this way given professional hospitalist societies have endorsed guidelines for improved transitions of care, which clearly delineate the key role hospitalists play in care transitions.[14]
Although fewer than 10% of respondents had worked in a PDC, nearly one‐third reported considering starting such a practice in the future, underscoring the importance of understanding hospitalist attitudes and experiences when creating a PDC and the significant barriers to arranging appropriate postdischarge care identified by survey respondents. The barriers to establishing a PDC may explain why few planned to start a PDC in the next year.
This study should be interpreted in the context of its design. Due to limitations in survey delivery, more rigorous sampling designs could not be used, and efforts were instead made to deliver the survey to a diverse group of hospitalists. The survey response rate was lower than anticipated and this increases the risk of response bias. Though this response rate is characteristic of other surveys of hospitalists, responses may have been from a selected population and therefore not representative of all hospitalists.[15] We sampled from a variety of practice venues, locations, academic and community practices, and practice group sizes to try to minimize this bias. Due to the low exposure rate to PDCs, hospitalist responses to experiences with PDCs should be considered exploratory.
We asked about similar content areas in the survey in multiple questions to maximize content validity; this resulted in variations in the degree of agreement or disagreement to similar prompts. For example, 62% of hospitalists opposed seeing patients in the clinic after discharge when directly asked, but nearly 50% said they would welcome the opportunity to work in a PDC if their employer required it. In another example, 50% of respondents said their responsibility for the patient ended at time of discharge, but when asked about duration of responsibility, 30% identified time of discharge as the limit. When reporting and interpreting results, we have tried to highlight responses to questions that ask most clearly and directly about the content of interest (rather than general themes), but this interpretation may also be subject to bias.
The time after hospital discharge is one of heightened risk for adverse events for the recently discharged patient. Hospitalist‐run postdischarge clinics may offer improved postdischarge care access and continuity; more research is needed on the effects of such clinics on patient outcomes, including postdischarge utilization. Until then, physicians and hospitals considering establishing PDCs should consider the barriers responding hospitalists identified to working in such a clinic, as well as the confidence they expressed in PDCs to reduce subsequent utilization.
Disclosures: Dr. Burke had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. The views expressed in this article are those of the authors and do not necessarily represent the views of the Department of Veterans Affairs. Dr. Burke was supported by grant funding from the Colorado Research Enhancement Award Program to Improve Care Coordination for Veterans. Dr. Ryan has no conflicts of interest to disclose.
Transitions of care, which encompass the patient experience of hospital discharge to the community, are frequently associated with clinically and financially costly adverse events.[1, 2] One important element for reducing the risk of postdischarge adverse events is provision of timely follow‐up by a clinician familiar with the patient and hospital course.[3, 4]
However, achieving this ideal is becoming more difficult because of an increased demand for primary care services (due to expanding coverage of Medicare and Medicaid) and the decreased supply of primary care physicians.[5, 6] When a timely visit with a clinician is available postdischarge, the widening discontinuity between inpatient and outpatient care providers often means this clinician is lacking essential details of the hospitalization.[7, 8]
One increasingly common innovation to improve postdischarge care access and continuity is to extend the role of inpatient providers (usually hospitalists) to provide care after discharge in a postdischarge clinic (PDC).[9, 10, 11] These clinics require an expansion of a hospitalist's duties to the outpatient setting, a requirement that has met with hospitalist resistance in initial reports.[12] However, little is known about hospitalists' experience with PDCs or attitudes toward postdischarge care. We aimed to explore these attitudes and experiences surrounding postdischarge care and PDCs.
METHODS
We conducted a cross‐sectional 17‐question Web‐based survey of hospitalists at 20 academic and 17 VA medical centers across the United States. Hospital medicine faculty at each site were identified by their group leader; members of each group then received an email survey up to 3 times. To collect responses from nonacademic hospitalists, the survey was also distributed to a large national private hospitalist employer. Due to internal limitations at the employer site, sampling was not feasible, and thus a convenience sample was obtained. Hospitalists who were not clinically active or did not have computer access to complete the survey were excluded. Responses were initially gathered on a 4‐point Likert scale; for comparisons between groups the scale was collapsed to a binary comparison using Fisher exact or 2 tests. We included questions answered in partially completed surveys in both the numerator and denominator; questions not answered were excluded from both numerator and denominator. The denominator of all responses was noted. All analyses were conducted using SAS 9.3 (SAS Institute, Inc., Cary, NC). The study was approved by the Colorado Multiple Institutional Review Board.
RESULTS
Of 814 hospitalists, 228 responded to the survey (28.3%). Table 1 illustrates characteristics of responding hospitalists, who were divided between university hospitals, community teaching hospitals, and community nonteaching hospitals in diverse practices in terms of location and group size.
| Characteristic | Respondents, No. (%) |
|---|---|
| |
| Employing institution | |
| University hospital | 79 (37.4) |
| Community, nonteaching | 62 (29.4) |
| Community, teaching | 70 (33.2) |
| Care environment | |
| Hospitalist providers | 98 (46.4) |
| Housestaff providers | 94 (44.6) |
| Primary care providers | 10 (4.7) |
| Combination | 9 (4.3) |
| Hospitalist group size (number of hospitalists) | |
| 15 | 39 (18.5) |
| 610 | 50 (23.7) |
| 1120 | 52 (24.6) |
| 2150 | 59 (28.0) |
| >50 | 9 (4.3) |
| Hospital locationpopulation | |
| Rural | 20 (9.5) |
| Suburban | 47 (22.3) |
| Urban | 144 (68.2) |
| Hospital locationgeographic | |
| West Coast | 11 (5.2) |
| Midwest | 47 (22.3) |
| Southern | 57 (27.1) |
| East Coast | 21 (10) |
| Southwest | 36 (17.1) |
| Mountain | 32 (15.2) |
Sixty‐one percent of responding hospitalists believed most patient problems after discharge were due to poor follow‐up with primary care providers, and 55% found it difficult to arrange timely primary care follow up (Table 2). Despite this, 87% thought patient problems after discharge should be cared for by primary care physicians, and 62% opposed the idea of hospitalists seeing patients in the clinic after discharge.
| Agree, No. (%) | Disagree, No. (%) | |
|---|---|---|
| ||
| Hospitalists should see patients in clinic after discharge | 87 (38.2) | 141 (61.8) |
| Primary care responsible for problems after discharge | 198 (86.8) | 30 (13.2) |
| Hospitalists responsible for patients after discharge | 113 (49.6) | 115 (50.4) |
| Would welcome a PDC if employer required | 113 (49.6) | 115 (50.4) |
| Would require extra compensation to work in a PDC | 175 (76.8) | 53 (23.2) |
| Believe a PDC would reduce ED visits after discharge | 168 (73.7) | 60 (26.3) |
| Would discharge patients earlier if could see after discharge | 116 (50.9) | 112 (49.1) |
| Most postdischarge problems due to poor PCP access | 138 (60.5) | 90 (39.5) |
| Easy to arrange timely follow‐up with patient's PCP | 100 (44.2) | 126 (55.3) |
When asked if hospitalists were responsible for patients after discharge from the hospital, only 50% responded positively. However, when asked how long hospitalists were responsible for patients after discharge, 71% gave a response longer than hospital discharge, including 60% who believed this responsibility ended at 1 week or less following discharge. A minority (12%) felt it extended to 1 month following discharge (Table 3).
| Respondents, No. (%) | |
|---|---|
| |
| Length of time inpatient providers responsible after discharge | |
| Responsibility ends at time of discharge | 65 (28.5) |
| 13 days | 40 (17.5) |
| 47 days | 57 (25.0) |
| 2 weeks | 41 (18.0) |
| 4 weeks | 20 (8.8) |
| 3 months | 3 (1.3) |
| 3 months | 2 (0.9) |
| Postdischarge clinic present | 20 (8.8) |
| Considered starting a postdischarge clinica | 62 (30.5) |
| Starting in next yearb | 6 (3.3) |
| Are satisfied with experience in postdischarge clinicc | 17 (85) |
| Think patients are satisfied/highly satisfiedc | 14 (70) |
Responding hospitalists expressed confidence in a PDC to reduce postdischarge emergency department visits (74%). However, most felt they would require extra compensation to staff a PDC (77%). They were divided on whether they would discharge patients from the hospital earlier if they could see those patients in postdischarge follow‐up (51% would discharge patients earlier).
Compared to those who had not experienced a PDC, responding hospitalists who had provided care in a PDC trended toward responding more positively that hospitalists should provide postdischarge care (P = 0.054). Few responding hospitalists had such exposure (8.8%) at the time of the survey. Although 31% had considering starting a PDC, only 3% were starting in the next year. Of responding hospitalists with exposure to a PDC, 70% were satisfied with the experience and 85% felt their patients were satisfied. Responses did not vary by type of practice (academic vs nonacademic), group size, geographic location, or by exposure to a PDC except as above.
DISCUSSION
Responding hospitalists reported encountering significant difficulty arranging appropriate postdischarge appointments with primary care providers and feel this contributes to postdischarge complications. Nearly 75% of those surveyed felt a hospitalist‐run PDC would be effective in reducing postdischarge emergency department visits, presumably in part due to improved access to postdischarge care. However, 62% of responding hospitalists opposed providing this type of care, though those who had experienced a PDC were somewhat more likely to view providing care in a PDC favorably. Survey responses largely reflect attitudes rather than experience with PDCs, because very few respondents had ever worked in a PDC.
The juxtaposition of the confidence expressed in PDCs to reduce postdischarge emergency department visits with the less enthusiastic views of respondents about providing care in a PDC was surprising. Several explanations are possible. First, providing such care is outside the usual scope of practice of most hospitalists, and preliminary reports indicate hospitalists, as self‐selected inpatient providers, may not initially welcome this opportunity.[12] Second, responding hospitalists identified the need for extra compensation for providing this care, suggesting they would see staffing a PDC as a burden requiring extra payment. Third, only 12% of respondents felt their responsibility to their discharged patients extended to 1 month following discharge. Given this, hospitalists may not feel enough personal ownership over 30‐day readmission rates to justify the additional clinical demand of staffing a PDC.[13]
In fact, 29% of responding hospitalists felt their responsibility to the patient ended at the time of discharge. Respondents may have interpreted responsibility differently, and we cannot rule out response bias given our lower‐than‐expected response rate. However, we had anticipated many fewer hospitalists would respond this way given professional hospitalist societies have endorsed guidelines for improved transitions of care, which clearly delineate the key role hospitalists play in care transitions.[14]
Although fewer than 10% of respondents had worked in a PDC, nearly one‐third reported considering starting such a practice in the future, underscoring the importance of understanding hospitalist attitudes and experiences when creating a PDC and the significant barriers to arranging appropriate postdischarge care identified by survey respondents. The barriers to establishing a PDC may explain why few planned to start a PDC in the next year.
This study should be interpreted in the context of its design. Due to limitations in survey delivery, more rigorous sampling designs could not be used, and efforts were instead made to deliver the survey to a diverse group of hospitalists. The survey response rate was lower than anticipated and this increases the risk of response bias. Though this response rate is characteristic of other surveys of hospitalists, responses may have been from a selected population and therefore not representative of all hospitalists.[15] We sampled from a variety of practice venues, locations, academic and community practices, and practice group sizes to try to minimize this bias. Due to the low exposure rate to PDCs, hospitalist responses to experiences with PDCs should be considered exploratory.
We asked about similar content areas in the survey in multiple questions to maximize content validity; this resulted in variations in the degree of agreement or disagreement to similar prompts. For example, 62% of hospitalists opposed seeing patients in the clinic after discharge when directly asked, but nearly 50% said they would welcome the opportunity to work in a PDC if their employer required it. In another example, 50% of respondents said their responsibility for the patient ended at time of discharge, but when asked about duration of responsibility, 30% identified time of discharge as the limit. When reporting and interpreting results, we have tried to highlight responses to questions that ask most clearly and directly about the content of interest (rather than general themes), but this interpretation may also be subject to bias.
The time after hospital discharge is one of heightened risk for adverse events for the recently discharged patient. Hospitalist‐run postdischarge clinics may offer improved postdischarge care access and continuity; more research is needed on the effects of such clinics on patient outcomes, including postdischarge utilization. Until then, physicians and hospitals considering establishing PDCs should consider the barriers responding hospitalists identified to working in such a clinic, as well as the confidence they expressed in PDCs to reduce subsequent utilization.
Disclosures: Dr. Burke had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. The views expressed in this article are those of the authors and do not necessarily represent the views of the Department of Veterans Affairs. Dr. Burke was supported by grant funding from the Colorado Research Enhancement Award Program to Improve Care Coordination for Veterans. Dr. Ryan has no conflicts of interest to disclose.
- , , , , . The incidence and severity of adverse events affecting patients after discharge from the hospital. Ann Intern Med. 2003;138(3):161–167.
- , , . Rehospitalizations among patients in the Medicare fee‐for‐service program. N Engl J Med. 2009;360(14):1418–1428.
- , , , et al. Relationship between early physician follow‐up and 30‐day readmission among Medicare beneficiaries hospitalized for heart failure. JAMA. 2010;303(17):1716–1722.
- , , . Post‐hospitalization transitions: Examining the effects of timing of primary care provider follow‐up. J Hosp Med. 2010;5(7):392–397.
- , , . Will generalist physician supply meet demands of an increasing and aging population? Health Aff (Millwood). 2008;27(3):w232–w241.
- Association of American Medical Colleges. June 2010.The impact of health care reform on the future supply and demand for physicians updated projections through 2025. Available at: http://www.aamc.org/download/158076/data/updated_projections_through_2025.pdf. Accessed May 1, 2012.
- , , , , , . Continuity of outpatient and inpatient care by primary care physicians for hospitalized older adults. JAMA. 2009;301(16):1671–1680.
- , , , , . Trends in inpatient continuity of care for a cohort of Medicare patients 1996–2006. J Hosp Med. 2011;6(8):438–444.
- . Is a post‐discharge clinic in your hospital's future? The Hospitalist. December 2011 Available at: http://www.the‐hospitalist.org/details/article/1409011/Is_a_Post_Discharge_Clinic_in_Your_Hospitals_future.html. Accessed May 1, 2013.
- , , , . Continuity of care and patient outcomes after hospital discharge. J Gen Intern Med. 2004;19(6):624–631.
- , , , . Effects of a postdischarge clinic on housestaff satisfaction and utilization of hospital services. J Gen Intern Med. 1996;11(3):179–181.
- . Interval examination: establishment of a hospitalist‐staffed discharge clinic. J Gen Intern Med. 2012;27(10):1377–1382.
- Patient Protection and Affordable Care Act (PPACA).Public Law 111–148 2010. Available at: http://www.gpo.gov/fdsys/pkg/PLAW‐111publ148/pdf/PLAW‐111publ148.pdf. Accessed January 10, 2013.
- , , , et al. Transitions of Care Consensus policy statement: American College of Physicians, Society of General Internal Medicine, Society of Hospital Medicine, American Geriatrics Society, American College of Emergency Physicians, and Society for Academic Emergency Medicine. J Hosp Med. 2009;4(6):364–370.
- , , , , . Person‐job fit: an exploratory cross‐sectional analysis of hospitalists. J Hosp Med. 2013;8(2):96–101.
- , , , , . The incidence and severity of adverse events affecting patients after discharge from the hospital. Ann Intern Med. 2003;138(3):161–167.
- , , . Rehospitalizations among patients in the Medicare fee‐for‐service program. N Engl J Med. 2009;360(14):1418–1428.
- , , , et al. Relationship between early physician follow‐up and 30‐day readmission among Medicare beneficiaries hospitalized for heart failure. JAMA. 2010;303(17):1716–1722.
- , , . Post‐hospitalization transitions: Examining the effects of timing of primary care provider follow‐up. J Hosp Med. 2010;5(7):392–397.
- , , . Will generalist physician supply meet demands of an increasing and aging population? Health Aff (Millwood). 2008;27(3):w232–w241.
- Association of American Medical Colleges. June 2010.The impact of health care reform on the future supply and demand for physicians updated projections through 2025. Available at: http://www.aamc.org/download/158076/data/updated_projections_through_2025.pdf. Accessed May 1, 2012.
- , , , , , . Continuity of outpatient and inpatient care by primary care physicians for hospitalized older adults. JAMA. 2009;301(16):1671–1680.
- , , , , . Trends in inpatient continuity of care for a cohort of Medicare patients 1996–2006. J Hosp Med. 2011;6(8):438–444.
- . Is a post‐discharge clinic in your hospital's future? The Hospitalist. December 2011 Available at: http://www.the‐hospitalist.org/details/article/1409011/Is_a_Post_Discharge_Clinic_in_Your_Hospitals_future.html. Accessed May 1, 2013.
- , , , . Continuity of care and patient outcomes after hospital discharge. J Gen Intern Med. 2004;19(6):624–631.
- , , , . Effects of a postdischarge clinic on housestaff satisfaction and utilization of hospital services. J Gen Intern Med. 1996;11(3):179–181.
- . Interval examination: establishment of a hospitalist‐staffed discharge clinic. J Gen Intern Med. 2012;27(10):1377–1382.
- Patient Protection and Affordable Care Act (PPACA).Public Law 111–148 2010. Available at: http://www.gpo.gov/fdsys/pkg/PLAW‐111publ148/pdf/PLAW‐111publ148.pdf. Accessed January 10, 2013.
- , , , et al. Transitions of Care Consensus policy statement: American College of Physicians, Society of General Internal Medicine, Society of Hospital Medicine, American Geriatrics Society, American College of Emergency Physicians, and Society for Academic Emergency Medicine. J Hosp Med. 2009;4(6):364–370.
- , , , , . Person‐job fit: an exploratory cross‐sectional analysis of hospitalists. J Hosp Med. 2013;8(2):96–101.
Estimating Hospital Costs of CAUTI
Healthcare‐associated infections affect 5% to 10% of all hospitalized patients each year in the United States, account for nearly $45 billion in direct hospital costs, and cause nearly 100,000 deaths annually.[1, 2] Because catheter‐associated urinary tract infection (CAUTI) is one of the most common healthcare‐associated infections in the United States and is reasonably preventable, the Centers for Medicare and Medicaid Services stopped reimbursing hospitals in 2008 for the additional costs of caring for patients who develop CAUTI during hospitalization.[3] Still, strategies for reducing inappropriate urinary catheterization are infrequently implemented in practice; this is despite a consensus that such strategies are effective.[4]
To help motivate hospitals to reduce inappropriate urinary catheter use, we present a tool for estimating costs of CAUTI for individual hospitals. Although other tools for estimating the excess costs of healthcare‐associated infections are available (eg, the APIC Cost of Healthcare‐Associated Infections Model available at
METHODS
General Setup
We consider 4 possible events after urinary catheter placement: bacteriuria, symptomatic urinary tract infection (SUTI), bloodstream infection (BSI), and catheter removal. Conservatively, assuming that bacteriuria must precede SUTI and BSI, there are 5 possible trajectories for any hospitalized patient (Figure 1): (1) no infection, (2) only bacteriuria, (3) bacteriuria and SUTI, (4) bacteriuria and BSI, or (5) bacteriuria, SUTI, and BSI. The cost of CAUTI for a particular hospital is therefore the per‐patient cost of each trajectory multiplied by the number of patients experiencing each trajectory. Our approach for estimating hospital costs is based on factorizing the number of patients experiencing each trajectory into a product of terms for which estimates are available from the literature (see the Supporting Information, Appendix, in the online version of this article for all technical details).
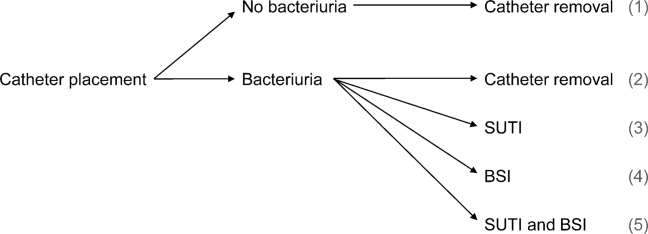
Deriving Estimates of Current Costs
We start with 2 minor simplifying assumptions. First, because the presence of asymptomatic bacteriuria is typically unknown, we only consider costs to the hospital due to SUTI and BSI[6]; in other words, we assume hospitals do not incur costs for patients with trajectories 1 or 2. This assumption should only bias cost estimates conservatively. Second, we assume that patients with both SUTI and BSI (trajectory 5) incur costs equal to those for patients with only BSI (trajectory 4). Further, because the joint risk of SUTI and BSI is unknown, we conservatively assume SUTI must precede BSI. Under these assumptions we can write: (total CAUTI costs)=(per‐patient SUTI cost) (number with SUTI but no BSI)+(perpatient BSI cost) (number with BSI).
We use per‐patient hospital costs of SUTI and BSI of $911 and $3824, respectively, which were determined using a microcosting approach[6] and adjusted for inflation using the general Consumer Price Index.[7] Although an alternative strategy for estimating costs would be to enter the hospital‐specific, per‐patient costs of SUTI and BSI into the above equation, these quantities are often difficult to measure or otherwise unavailable. Thus, it remains to factorize the number of hospitalized patients who develop SUTI and BSI into component terms for which we have accessible estimates. First note that the number with only SUTI (or any BSI) equals the total number of patients hospitalized times the proportion of hospitalizations with only SUTI (or any BSI). The former quantity depends on the particular hospital and so is specified as an input by the user. The latter quantity can be factorized further under our aforementioned conservative assumption that bacteriuria must precede SUTI and BSI.
Specifically, for SUTI:
(Proportion SUTI but no BSI)={(SUTI risk among those catheterized with bacteriuria)(BSI risk among those catheterized with bacteriuria)} (bacteriuria risk among those catheterized) (proportion catheterized).
And for BSI:
(Proportion BSI)=(BSI risk among those catheterized with bacteriuria) (bacteriuria risk among those catheterized) (proportion catheterized).
The risks of SUTI and BSI among those catheterized with bacteriuria, along with the risk of bacteriuria among those catheterized, have been estimated previously via a meta‐analytic approach.[6] The proportion catheterized depends on the particular hospital, such as the total number of patients hospitalized, and so is also specified as a user input. Therefore, we have now factorized the total hospital costs due to CAUTI as a product of either user‐specified terms or terms for which we have estimates from the literature. All estimates and corresponding standard errors derived from the literature are listed together in Table 1 (see the Supporting Information, Appendix Section 1, for further details in the online version of this article).
| Quantity | Estimate (SE) |
|---|---|
| |
| Overall risk of bacteriuria among those catheterized | 26.0% (1.53%) |
| Per‐day risk of bacteriuria among those catheterized | 5.0% |
| days | 6.68 |
| Risk of SUTI among those catheterized with bacteriuria | 24.0% (4.08%) |
| Risk of BSI among those catheterized with bacteriuria | 3.6% (0.10%) |
| Per‐patient SUTI cost | $911 ($911) |
| Per‐patient BSI cost | $3824 ($3824) |
Deriving Projected Costs After Intervention
The approach described above permits estimation of current costs for managing patients with CAUTI for a particular hospital. To estimate projected costs after participation in an intervention to reduce infection risk, we characterize interventions of interest and introduce additional factorization. Specifically, following previous work,[8] we consider interventions that reduce (1) placement (ie, the proportion catheterized) and (2) duration (ie, the mean duration of catheterization). Incorporating reductions in placement is straightforward, because our above expression for costs already contains a term for the proportion catheterized. However, incorporating reductions in duration requires further factorization. Under the assumptions of constant per‐day risks of bacteriuria and of catheter removal, we can write the postintervention risk of bacteriuria among the catheterized as a function of (1) the percent decrease in mean duration of catheterization due to intervention, and (2) the preintervention risk of bacteriuria among the catheterized (see the Supporting Information, Appendix Section 2, for further details in the online version of this article). This means we can fully characterize postintervention costs as a function of user‐specified quantities, quantities specific to the intervention (which are varied across plausible ranges), and quantities for which we have estimates from the literature. Therefore, we can estimate savings by subtracting postintervention costs from current costs.
Because our estimators of current costs, projected costs, and savings are all formulated as functions of other estimators, we use the standard delta method approach[9] to derive appropriate variance estimates (see the Supporting Information, Appendix Section 3, for further details in the online version of this article).
Online Implementation
Customized results (based on annual admissions, urinary catheter prevalence, and other inputs) can be computed using online implementation of our proposed method at
RESULTS
Figure 2 shows the projected savings in hospital costs due to CAUTI across a range of interventions defined by percent decreases in placement and duration, for a hypothetical hospital with 3000 total patients, 15% with urinary catheters preintervention, and with all other default values listed in Table 1. The current costs for this hospital (ie, the costs when the percent reduction in placement and duration is zero) are estimated to be $37,868 (95% confidence interval [CI]: $9159‐$156,564). After an intervention resulting in 40% reductions in both urinary catheter placement and duration, this hospital would be expected to save $22,653 (95% CI: $5479‐$93,656). A less effective intervention yielding a 10% reduction in both urinary catheter placement and duration would result in more modest savings of $6376 (95% CI: $1542‐$26,360).
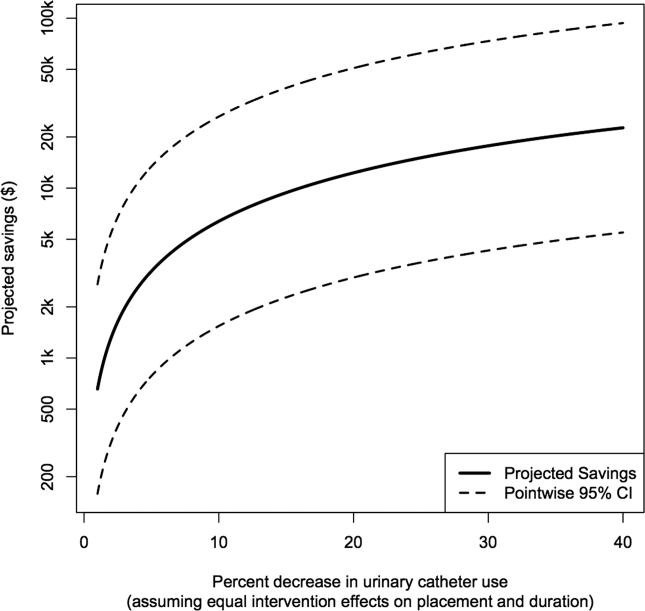
After an intervention resulting in 29% and 37% reductions in placement and duration, respectively, reflecting reductions seen in practice,[10, 11] our hypothetical hospital is estimated to save $19,126 (95% CI: $4626‐$79,074). This reflects an estimated savings of nearly 50%.
DISCUSSION
We have presented a tool for estimating customized hospital costs of CAUTI, both before and after a hypothetical intervention to reduce risk of infection. Our approach relies on mostly conservative assumptions, incorporates published risk estimates (properly accounting for their associated variability), and has easy‐to‐use online implementation. We believe this can play an important role in motivating hospitals to reduce inappropriate urinary catheter use.
The methodology employed here does have a few limitations. First and foremost, our results depend on the reliability of the input values, which are either provided by users or are based on estimates from the literature (see Table 1 for a complete list of suggested defaults). New information could potentially be incorporated if and when available. For example, substitution of more precise risk estimates could help reduce confidence interval length. Second, our approach essentially averages over hospital quality; we do not directly take into account quality of care or variation in underlying infection risk across hospitals in computing estimated costs. Finally, we only compute direct costs due to infection; other costs (eg, intervention costs) would typically also need to be considered for decision making.
Despite these limitations, we believe that our tool can help infection control professionals demonstrate the values of CAUTI prevention efforts to key administrators, particularly at a time where it has become increasingly necessary to develop a business case to initiate new interventions or justify the continued support for ongoing programs.[12] Additionally, we believe the proposed approach can be an important supplement to initiatives like the Society of Hospital Medicine's Choosing Wisely campaign, which aims to help reduce inappropriate urinary catheter use. Reducing catheter utilization has the potential to reduce costs associated with caring for CAUTI patients, but more importantly would help reduce CAUTI incidence as well as catheter‐related, noninfectious complications.[13, 14] We hope that our tool will greatly assist hospitals in promoting their CAUTI prevention efforts and improve the overall safety of hospitalized patients.
Disclosures
This project was supported by the Ann Arbor VA Medical Center/University of Michigan Patient Safety Enhancement Program (PSEP) and a subcontract to implement multistate CAUTI prevention with the Agency for Healthcare Research and Quality/Health Educational and Research Trust. Mr. Kennedy has no conflicts of interest to report. Drs. Saint and Greene are subcontracted to implement multistate CAUTI prevention with the Agency for Healthcare Research and Quality/Health Educational and Research Trust. Dr. Saint has received numerous honoraria and speaking fees for lectures on healthcare‐associated infection prevention, implementation science, and patient safety from hospitals, academic medical centers, professional societies, and nonprofit foundations. None of these activities are related to speaker's bureaus. Dr. Saint is also on the medical advisory board of Doximity, a new social networking site for physicians. The views expressed in this article are those of the authors and do not necessarily reflect the position or policy of the Department of Veterans Affairs.
- , , , et al. Estimating health care‐associated infections and deaths in U.S. hospitals, 2002. Public Health Rep. 2007;122:160–166.
- The direct medical costs of healthcare‐associated infections in US hospitals and the benefits of prevention. US Centers for Disease Control and Prevention Web site. Published 2009. Available at: http://www.cdc.gov/HAI/pdfs/hai/Scott_CostPaper.pdf. Accessed March 24, 2013.
- , , , , . Catheter‐associated urinary tract infection and the Medicare rule changes. Ann Intern Med. 2009;150(12):877–884.
- , . Improving use of the other catheter. Arch Intern Med. 2012;172(3):260–261.
- Choosing Wisely: five things patients and physicians should question. Society of Hospital Medicine. Published 2012. Available at: http://www.hospitalmedicine.org/AM/pdf/SHM‐Adult_5things_List_Web.pdf. Accessed March 24, 2013.
- . Clinical and economic consequences of nosocomial catheter‐related bacteriuria. Am J Infect Control. 2000;28(1):68–75.
- CPI Inflation Calculator. United States Department of Labor, Bureau of Labor Statistics Web site. Published 2013. Available at: http://www.bls.gov/data/inflation_calculator.htm. Accessed March 24, 2013.
- , , , et al. Introducing a population‐based outcome measure to evaluate the effect of interventions to reduce catheter‐associated urinary tract infection. Am J Infect Control. 2012;40(4):359–364.
- . Asymptotic Statistics. Cambridge, UK: Cambridge University Press; 2000.
- , , , et al. Effect of establishing guidelines on appropriate urinary catheter placement. Acad Emerg Med. 2010;17:337–340.
- , , , . Systematic review and meta‐analysis: reminder systems to reduce catheter‐associated urinary tract infections and urinary catheter use in hospitalized patients. Clin Infect Dis. 2010;51(5):550–560.
- , , , et al. Raising standards while watching the bottom line: making a business case for infection control. Infect Control Hosp Epidemiol. 2007;28:1121–1133.
- , , , , . Urinary catheters: what type do men and their nurses prefer? J Am Geriatr Soc. 1999;47(12):1453–1457.
- , , . Indwelling urinary catheters: a one‐point restraint? Ann Intern Med. 2002;137(2):125–127.
Healthcare‐associated infections affect 5% to 10% of all hospitalized patients each year in the United States, account for nearly $45 billion in direct hospital costs, and cause nearly 100,000 deaths annually.[1, 2] Because catheter‐associated urinary tract infection (CAUTI) is one of the most common healthcare‐associated infections in the United States and is reasonably preventable, the Centers for Medicare and Medicaid Services stopped reimbursing hospitals in 2008 for the additional costs of caring for patients who develop CAUTI during hospitalization.[3] Still, strategies for reducing inappropriate urinary catheterization are infrequently implemented in practice; this is despite a consensus that such strategies are effective.[4]
To help motivate hospitals to reduce inappropriate urinary catheter use, we present a tool for estimating costs of CAUTI for individual hospitals. Although other tools for estimating the excess costs of healthcare‐associated infections are available (eg, the APIC Cost of Healthcare‐Associated Infections Model available at
METHODS
General Setup
We consider 4 possible events after urinary catheter placement: bacteriuria, symptomatic urinary tract infection (SUTI), bloodstream infection (BSI), and catheter removal. Conservatively, assuming that bacteriuria must precede SUTI and BSI, there are 5 possible trajectories for any hospitalized patient (Figure 1): (1) no infection, (2) only bacteriuria, (3) bacteriuria and SUTI, (4) bacteriuria and BSI, or (5) bacteriuria, SUTI, and BSI. The cost of CAUTI for a particular hospital is therefore the per‐patient cost of each trajectory multiplied by the number of patients experiencing each trajectory. Our approach for estimating hospital costs is based on factorizing the number of patients experiencing each trajectory into a product of terms for which estimates are available from the literature (see the Supporting Information, Appendix, in the online version of this article for all technical details).

Deriving Estimates of Current Costs
We start with 2 minor simplifying assumptions. First, because the presence of asymptomatic bacteriuria is typically unknown, we only consider costs to the hospital due to SUTI and BSI[6]; in other words, we assume hospitals do not incur costs for patients with trajectories 1 or 2. This assumption should only bias cost estimates conservatively. Second, we assume that patients with both SUTI and BSI (trajectory 5) incur costs equal to those for patients with only BSI (trajectory 4). Further, because the joint risk of SUTI and BSI is unknown, we conservatively assume SUTI must precede BSI. Under these assumptions we can write: (total CAUTI costs)=(per‐patient SUTI cost) (number with SUTI but no BSI)+(perpatient BSI cost) (number with BSI).
We use per‐patient hospital costs of SUTI and BSI of $911 and $3824, respectively, which were determined using a microcosting approach[6] and adjusted for inflation using the general Consumer Price Index.[7] Although an alternative strategy for estimating costs would be to enter the hospital‐specific, per‐patient costs of SUTI and BSI into the above equation, these quantities are often difficult to measure or otherwise unavailable. Thus, it remains to factorize the number of hospitalized patients who develop SUTI and BSI into component terms for which we have accessible estimates. First note that the number with only SUTI (or any BSI) equals the total number of patients hospitalized times the proportion of hospitalizations with only SUTI (or any BSI). The former quantity depends on the particular hospital and so is specified as an input by the user. The latter quantity can be factorized further under our aforementioned conservative assumption that bacteriuria must precede SUTI and BSI.
Specifically, for SUTI:
(Proportion SUTI but no BSI)={(SUTI risk among those catheterized with bacteriuria)(BSI risk among those catheterized with bacteriuria)} (bacteriuria risk among those catheterized) (proportion catheterized).
And for BSI:
(Proportion BSI)=(BSI risk among those catheterized with bacteriuria) (bacteriuria risk among those catheterized) (proportion catheterized).
The risks of SUTI and BSI among those catheterized with bacteriuria, along with the risk of bacteriuria among those catheterized, have been estimated previously via a meta‐analytic approach.[6] The proportion catheterized depends on the particular hospital, such as the total number of patients hospitalized, and so is also specified as a user input. Therefore, we have now factorized the total hospital costs due to CAUTI as a product of either user‐specified terms or terms for which we have estimates from the literature. All estimates and corresponding standard errors derived from the literature are listed together in Table 1 (see the Supporting Information, Appendix Section 1, for further details in the online version of this article).
| Quantity | Estimate (SE) |
|---|---|
| |
| Overall risk of bacteriuria among those catheterized | 26.0% (1.53%) |
| Per‐day risk of bacteriuria among those catheterized | 5.0% |
| days | 6.68 |
| Risk of SUTI among those catheterized with bacteriuria | 24.0% (4.08%) |
| Risk of BSI among those catheterized with bacteriuria | 3.6% (0.10%) |
| Per‐patient SUTI cost | $911 ($911) |
| Per‐patient BSI cost | $3824 ($3824) |
Deriving Projected Costs After Intervention
The approach described above permits estimation of current costs for managing patients with CAUTI for a particular hospital. To estimate projected costs after participation in an intervention to reduce infection risk, we characterize interventions of interest and introduce additional factorization. Specifically, following previous work,[8] we consider interventions that reduce (1) placement (ie, the proportion catheterized) and (2) duration (ie, the mean duration of catheterization). Incorporating reductions in placement is straightforward, because our above expression for costs already contains a term for the proportion catheterized. However, incorporating reductions in duration requires further factorization. Under the assumptions of constant per‐day risks of bacteriuria and of catheter removal, we can write the postintervention risk of bacteriuria among the catheterized as a function of (1) the percent decrease in mean duration of catheterization due to intervention, and (2) the preintervention risk of bacteriuria among the catheterized (see the Supporting Information, Appendix Section 2, for further details in the online version of this article). This means we can fully characterize postintervention costs as a function of user‐specified quantities, quantities specific to the intervention (which are varied across plausible ranges), and quantities for which we have estimates from the literature. Therefore, we can estimate savings by subtracting postintervention costs from current costs.
Because our estimators of current costs, projected costs, and savings are all formulated as functions of other estimators, we use the standard delta method approach[9] to derive appropriate variance estimates (see the Supporting Information, Appendix Section 3, for further details in the online version of this article).
Online Implementation
Customized results (based on annual admissions, urinary catheter prevalence, and other inputs) can be computed using online implementation of our proposed method at
RESULTS
Figure 2 shows the projected savings in hospital costs due to CAUTI across a range of interventions defined by percent decreases in placement and duration, for a hypothetical hospital with 3000 total patients, 15% with urinary catheters preintervention, and with all other default values listed in Table 1. The current costs for this hospital (ie, the costs when the percent reduction in placement and duration is zero) are estimated to be $37,868 (95% confidence interval [CI]: $9159‐$156,564). After an intervention resulting in 40% reductions in both urinary catheter placement and duration, this hospital would be expected to save $22,653 (95% CI: $5479‐$93,656). A less effective intervention yielding a 10% reduction in both urinary catheter placement and duration would result in more modest savings of $6376 (95% CI: $1542‐$26,360).

After an intervention resulting in 29% and 37% reductions in placement and duration, respectively, reflecting reductions seen in practice,[10, 11] our hypothetical hospital is estimated to save $19,126 (95% CI: $4626‐$79,074). This reflects an estimated savings of nearly 50%.
DISCUSSION
We have presented a tool for estimating customized hospital costs of CAUTI, both before and after a hypothetical intervention to reduce risk of infection. Our approach relies on mostly conservative assumptions, incorporates published risk estimates (properly accounting for their associated variability), and has easy‐to‐use online implementation. We believe this can play an important role in motivating hospitals to reduce inappropriate urinary catheter use.
The methodology employed here does have a few limitations. First and foremost, our results depend on the reliability of the input values, which are either provided by users or are based on estimates from the literature (see Table 1 for a complete list of suggested defaults). New information could potentially be incorporated if and when available. For example, substitution of more precise risk estimates could help reduce confidence interval length. Second, our approach essentially averages over hospital quality; we do not directly take into account quality of care or variation in underlying infection risk across hospitals in computing estimated costs. Finally, we only compute direct costs due to infection; other costs (eg, intervention costs) would typically also need to be considered for decision making.
Despite these limitations, we believe that our tool can help infection control professionals demonstrate the values of CAUTI prevention efforts to key administrators, particularly at a time where it has become increasingly necessary to develop a business case to initiate new interventions or justify the continued support for ongoing programs.[12] Additionally, we believe the proposed approach can be an important supplement to initiatives like the Society of Hospital Medicine's Choosing Wisely campaign, which aims to help reduce inappropriate urinary catheter use. Reducing catheter utilization has the potential to reduce costs associated with caring for CAUTI patients, but more importantly would help reduce CAUTI incidence as well as catheter‐related, noninfectious complications.[13, 14] We hope that our tool will greatly assist hospitals in promoting their CAUTI prevention efforts and improve the overall safety of hospitalized patients.
Disclosures
This project was supported by the Ann Arbor VA Medical Center/University of Michigan Patient Safety Enhancement Program (PSEP) and a subcontract to implement multistate CAUTI prevention with the Agency for Healthcare Research and Quality/Health Educational and Research Trust. Mr. Kennedy has no conflicts of interest to report. Drs. Saint and Greene are subcontracted to implement multistate CAUTI prevention with the Agency for Healthcare Research and Quality/Health Educational and Research Trust. Dr. Saint has received numerous honoraria and speaking fees for lectures on healthcare‐associated infection prevention, implementation science, and patient safety from hospitals, academic medical centers, professional societies, and nonprofit foundations. None of these activities are related to speaker's bureaus. Dr. Saint is also on the medical advisory board of Doximity, a new social networking site for physicians. The views expressed in this article are those of the authors and do not necessarily reflect the position or policy of the Department of Veterans Affairs.
Healthcare‐associated infections affect 5% to 10% of all hospitalized patients each year in the United States, account for nearly $45 billion in direct hospital costs, and cause nearly 100,000 deaths annually.[1, 2] Because catheter‐associated urinary tract infection (CAUTI) is one of the most common healthcare‐associated infections in the United States and is reasonably preventable, the Centers for Medicare and Medicaid Services stopped reimbursing hospitals in 2008 for the additional costs of caring for patients who develop CAUTI during hospitalization.[3] Still, strategies for reducing inappropriate urinary catheterization are infrequently implemented in practice; this is despite a consensus that such strategies are effective.[4]
To help motivate hospitals to reduce inappropriate urinary catheter use, we present a tool for estimating costs of CAUTI for individual hospitals. Although other tools for estimating the excess costs of healthcare‐associated infections are available (eg, the APIC Cost of Healthcare‐Associated Infections Model available at
METHODS
General Setup
We consider 4 possible events after urinary catheter placement: bacteriuria, symptomatic urinary tract infection (SUTI), bloodstream infection (BSI), and catheter removal. Conservatively, assuming that bacteriuria must precede SUTI and BSI, there are 5 possible trajectories for any hospitalized patient (Figure 1): (1) no infection, (2) only bacteriuria, (3) bacteriuria and SUTI, (4) bacteriuria and BSI, or (5) bacteriuria, SUTI, and BSI. The cost of CAUTI for a particular hospital is therefore the per‐patient cost of each trajectory multiplied by the number of patients experiencing each trajectory. Our approach for estimating hospital costs is based on factorizing the number of patients experiencing each trajectory into a product of terms for which estimates are available from the literature (see the Supporting Information, Appendix, in the online version of this article for all technical details).

Deriving Estimates of Current Costs
We start with 2 minor simplifying assumptions. First, because the presence of asymptomatic bacteriuria is typically unknown, we only consider costs to the hospital due to SUTI and BSI[6]; in other words, we assume hospitals do not incur costs for patients with trajectories 1 or 2. This assumption should only bias cost estimates conservatively. Second, we assume that patients with both SUTI and BSI (trajectory 5) incur costs equal to those for patients with only BSI (trajectory 4). Further, because the joint risk of SUTI and BSI is unknown, we conservatively assume SUTI must precede BSI. Under these assumptions we can write: (total CAUTI costs)=(per‐patient SUTI cost) (number with SUTI but no BSI)+(perpatient BSI cost) (number with BSI).
We use per‐patient hospital costs of SUTI and BSI of $911 and $3824, respectively, which were determined using a microcosting approach[6] and adjusted for inflation using the general Consumer Price Index.[7] Although an alternative strategy for estimating costs would be to enter the hospital‐specific, per‐patient costs of SUTI and BSI into the above equation, these quantities are often difficult to measure or otherwise unavailable. Thus, it remains to factorize the number of hospitalized patients who develop SUTI and BSI into component terms for which we have accessible estimates. First note that the number with only SUTI (or any BSI) equals the total number of patients hospitalized times the proportion of hospitalizations with only SUTI (or any BSI). The former quantity depends on the particular hospital and so is specified as an input by the user. The latter quantity can be factorized further under our aforementioned conservative assumption that bacteriuria must precede SUTI and BSI.
Specifically, for SUTI:
(Proportion SUTI but no BSI)={(SUTI risk among those catheterized with bacteriuria)(BSI risk among those catheterized with bacteriuria)} (bacteriuria risk among those catheterized) (proportion catheterized).
And for BSI:
(Proportion BSI)=(BSI risk among those catheterized with bacteriuria) (bacteriuria risk among those catheterized) (proportion catheterized).
The risks of SUTI and BSI among those catheterized with bacteriuria, along with the risk of bacteriuria among those catheterized, have been estimated previously via a meta‐analytic approach.[6] The proportion catheterized depends on the particular hospital, such as the total number of patients hospitalized, and so is also specified as a user input. Therefore, we have now factorized the total hospital costs due to CAUTI as a product of either user‐specified terms or terms for which we have estimates from the literature. All estimates and corresponding standard errors derived from the literature are listed together in Table 1 (see the Supporting Information, Appendix Section 1, for further details in the online version of this article).
| Quantity | Estimate (SE) |
|---|---|
| |
| Overall risk of bacteriuria among those catheterized | 26.0% (1.53%) |
| Per‐day risk of bacteriuria among those catheterized | 5.0% |
| days | 6.68 |
| Risk of SUTI among those catheterized with bacteriuria | 24.0% (4.08%) |
| Risk of BSI among those catheterized with bacteriuria | 3.6% (0.10%) |
| Per‐patient SUTI cost | $911 ($911) |
| Per‐patient BSI cost | $3824 ($3824) |
Deriving Projected Costs After Intervention
The approach described above permits estimation of current costs for managing patients with CAUTI for a particular hospital. To estimate projected costs after participation in an intervention to reduce infection risk, we characterize interventions of interest and introduce additional factorization. Specifically, following previous work,[8] we consider interventions that reduce (1) placement (ie, the proportion catheterized) and (2) duration (ie, the mean duration of catheterization). Incorporating reductions in placement is straightforward, because our above expression for costs already contains a term for the proportion catheterized. However, incorporating reductions in duration requires further factorization. Under the assumptions of constant per‐day risks of bacteriuria and of catheter removal, we can write the postintervention risk of bacteriuria among the catheterized as a function of (1) the percent decrease in mean duration of catheterization due to intervention, and (2) the preintervention risk of bacteriuria among the catheterized (see the Supporting Information, Appendix Section 2, for further details in the online version of this article). This means we can fully characterize postintervention costs as a function of user‐specified quantities, quantities specific to the intervention (which are varied across plausible ranges), and quantities for which we have estimates from the literature. Therefore, we can estimate savings by subtracting postintervention costs from current costs.
Because our estimators of current costs, projected costs, and savings are all formulated as functions of other estimators, we use the standard delta method approach[9] to derive appropriate variance estimates (see the Supporting Information, Appendix Section 3, for further details in the online version of this article).
Online Implementation
Customized results (based on annual admissions, urinary catheter prevalence, and other inputs) can be computed using online implementation of our proposed method at
RESULTS
Figure 2 shows the projected savings in hospital costs due to CAUTI across a range of interventions defined by percent decreases in placement and duration, for a hypothetical hospital with 3000 total patients, 15% with urinary catheters preintervention, and with all other default values listed in Table 1. The current costs for this hospital (ie, the costs when the percent reduction in placement and duration is zero) are estimated to be $37,868 (95% confidence interval [CI]: $9159‐$156,564). After an intervention resulting in 40% reductions in both urinary catheter placement and duration, this hospital would be expected to save $22,653 (95% CI: $5479‐$93,656). A less effective intervention yielding a 10% reduction in both urinary catheter placement and duration would result in more modest savings of $6376 (95% CI: $1542‐$26,360).

After an intervention resulting in 29% and 37% reductions in placement and duration, respectively, reflecting reductions seen in practice,[10, 11] our hypothetical hospital is estimated to save $19,126 (95% CI: $4626‐$79,074). This reflects an estimated savings of nearly 50%.
DISCUSSION
We have presented a tool for estimating customized hospital costs of CAUTI, both before and after a hypothetical intervention to reduce risk of infection. Our approach relies on mostly conservative assumptions, incorporates published risk estimates (properly accounting for their associated variability), and has easy‐to‐use online implementation. We believe this can play an important role in motivating hospitals to reduce inappropriate urinary catheter use.
The methodology employed here does have a few limitations. First and foremost, our results depend on the reliability of the input values, which are either provided by users or are based on estimates from the literature (see Table 1 for a complete list of suggested defaults). New information could potentially be incorporated if and when available. For example, substitution of more precise risk estimates could help reduce confidence interval length. Second, our approach essentially averages over hospital quality; we do not directly take into account quality of care or variation in underlying infection risk across hospitals in computing estimated costs. Finally, we only compute direct costs due to infection; other costs (eg, intervention costs) would typically also need to be considered for decision making.
Despite these limitations, we believe that our tool can help infection control professionals demonstrate the values of CAUTI prevention efforts to key administrators, particularly at a time where it has become increasingly necessary to develop a business case to initiate new interventions or justify the continued support for ongoing programs.[12] Additionally, we believe the proposed approach can be an important supplement to initiatives like the Society of Hospital Medicine's Choosing Wisely campaign, which aims to help reduce inappropriate urinary catheter use. Reducing catheter utilization has the potential to reduce costs associated with caring for CAUTI patients, but more importantly would help reduce CAUTI incidence as well as catheter‐related, noninfectious complications.[13, 14] We hope that our tool will greatly assist hospitals in promoting their CAUTI prevention efforts and improve the overall safety of hospitalized patients.
Disclosures
This project was supported by the Ann Arbor VA Medical Center/University of Michigan Patient Safety Enhancement Program (PSEP) and a subcontract to implement multistate CAUTI prevention with the Agency for Healthcare Research and Quality/Health Educational and Research Trust. Mr. Kennedy has no conflicts of interest to report. Drs. Saint and Greene are subcontracted to implement multistate CAUTI prevention with the Agency for Healthcare Research and Quality/Health Educational and Research Trust. Dr. Saint has received numerous honoraria and speaking fees for lectures on healthcare‐associated infection prevention, implementation science, and patient safety from hospitals, academic medical centers, professional societies, and nonprofit foundations. None of these activities are related to speaker's bureaus. Dr. Saint is also on the medical advisory board of Doximity, a new social networking site for physicians. The views expressed in this article are those of the authors and do not necessarily reflect the position or policy of the Department of Veterans Affairs.
- , , , et al. Estimating health care‐associated infections and deaths in U.S. hospitals, 2002. Public Health Rep. 2007;122:160–166.
- The direct medical costs of healthcare‐associated infections in US hospitals and the benefits of prevention. US Centers for Disease Control and Prevention Web site. Published 2009. Available at: http://www.cdc.gov/HAI/pdfs/hai/Scott_CostPaper.pdf. Accessed March 24, 2013.
- , , , , . Catheter‐associated urinary tract infection and the Medicare rule changes. Ann Intern Med. 2009;150(12):877–884.
- , . Improving use of the other catheter. Arch Intern Med. 2012;172(3):260–261.
- Choosing Wisely: five things patients and physicians should question. Society of Hospital Medicine. Published 2012. Available at: http://www.hospitalmedicine.org/AM/pdf/SHM‐Adult_5things_List_Web.pdf. Accessed March 24, 2013.
- . Clinical and economic consequences of nosocomial catheter‐related bacteriuria. Am J Infect Control. 2000;28(1):68–75.
- CPI Inflation Calculator. United States Department of Labor, Bureau of Labor Statistics Web site. Published 2013. Available at: http://www.bls.gov/data/inflation_calculator.htm. Accessed March 24, 2013.
- , , , et al. Introducing a population‐based outcome measure to evaluate the effect of interventions to reduce catheter‐associated urinary tract infection. Am J Infect Control. 2012;40(4):359–364.
- . Asymptotic Statistics. Cambridge, UK: Cambridge University Press; 2000.
- , , , et al. Effect of establishing guidelines on appropriate urinary catheter placement. Acad Emerg Med. 2010;17:337–340.
- , , , . Systematic review and meta‐analysis: reminder systems to reduce catheter‐associated urinary tract infections and urinary catheter use in hospitalized patients. Clin Infect Dis. 2010;51(5):550–560.
- , , , et al. Raising standards while watching the bottom line: making a business case for infection control. Infect Control Hosp Epidemiol. 2007;28:1121–1133.
- , , , , . Urinary catheters: what type do men and their nurses prefer? J Am Geriatr Soc. 1999;47(12):1453–1457.
- , , . Indwelling urinary catheters: a one‐point restraint? Ann Intern Med. 2002;137(2):125–127.
- , , , et al. Estimating health care‐associated infections and deaths in U.S. hospitals, 2002. Public Health Rep. 2007;122:160–166.
- The direct medical costs of healthcare‐associated infections in US hospitals and the benefits of prevention. US Centers for Disease Control and Prevention Web site. Published 2009. Available at: http://www.cdc.gov/HAI/pdfs/hai/Scott_CostPaper.pdf. Accessed March 24, 2013.
- , , , , . Catheter‐associated urinary tract infection and the Medicare rule changes. Ann Intern Med. 2009;150(12):877–884.
- , . Improving use of the other catheter. Arch Intern Med. 2012;172(3):260–261.
- Choosing Wisely: five things patients and physicians should question. Society of Hospital Medicine. Published 2012. Available at: http://www.hospitalmedicine.org/AM/pdf/SHM‐Adult_5things_List_Web.pdf. Accessed March 24, 2013.
- . Clinical and economic consequences of nosocomial catheter‐related bacteriuria. Am J Infect Control. 2000;28(1):68–75.
- CPI Inflation Calculator. United States Department of Labor, Bureau of Labor Statistics Web site. Published 2013. Available at: http://www.bls.gov/data/inflation_calculator.htm. Accessed March 24, 2013.
- , , , et al. Introducing a population‐based outcome measure to evaluate the effect of interventions to reduce catheter‐associated urinary tract infection. Am J Infect Control. 2012;40(4):359–364.
- . Asymptotic Statistics. Cambridge, UK: Cambridge University Press; 2000.
- , , , et al. Effect of establishing guidelines on appropriate urinary catheter placement. Acad Emerg Med. 2010;17:337–340.
- , , , . Systematic review and meta‐analysis: reminder systems to reduce catheter‐associated urinary tract infections and urinary catheter use in hospitalized patients. Clin Infect Dis. 2010;51(5):550–560.
- , , , et al. Raising standards while watching the bottom line: making a business case for infection control. Infect Control Hosp Epidemiol. 2007;28:1121–1133.
- , , , , . Urinary catheters: what type do men and their nurses prefer? J Am Geriatr Soc. 1999;47(12):1453–1457.
- , , . Indwelling urinary catheters: a one‐point restraint? Ann Intern Med. 2002;137(2):125–127.
Medicare Beneficiaries Likely Readmitted
For at least 25 years, approximately 20% of Medicare fee‐for‐service discharges have been followed by a hospital readmission within 30 days.[1, 2] Section 3025 of the Patient Protection and Affordable Care Act (ACA)[3] created escalating penalties for hospitals with higher than expected 30‐day readmission rates, and the Congressional Budget Office estimated this will reduce Medicare spending by over $7 billion between 2010 and 2019.[4]
Hospitals and physicians have begun developing strategies to identify which Medicare beneficiaries are most likely to be readmitted and use this information to design programs to reduce their readmission rate. Initially, penalties will be based on readmission rates after an index discharge with heart failure, myocardial infarction, and pneumonia.[5] Recently, the Centers for Medicare and Medicaid Services (CMS) released the Inpatient Prospective Payment System FY2014 proposed rule, which proposes to add 2 new readmission penalties beginning in FY2015: readmissions for hip/knee arthroplasty and chronic obstructive pulmonary disease.[6] Other countries are already penalizing hospitals with high readmission rates; for example, Germany is penalizing all readmissions that occur within a 30‐day period following admission.[7] In this brief report, we examine the characteristics of Medicare beneficiaries most likely to be readmitted within 30 days. We focus on readmission rates for all discharge conditions and all patient readmission rates, because we believe the language in the ACA ultimately points to an all‐inclusive approach.
METHODS
We used a nationally random 5% sample of all Medicare beneficiaries for the period between January 1, 2008 and September 30, 2008. To be included, beneficiaries must have both Part A and B coverage and live within the United States. Medicare Advantage patients were excluded because Medicare Advantage plans do not report the data in the same way as fee for service. We calculated the readmission rate as the number of admissions that were preceded by an at‐risk discharge within 30 days divided by the total number of at‐risk discharges. This definition included admissions to and discharges from sole community providers, Medicare‐dependent small rural hospitals, and critical access hospitals. We counted as at risk all live discharges from short‐term acute care hospitals that were not discharged against medical advice, discharged to a rehabilitation unit within an acute care hospital, or readmitted on day 0 (due to inconsistency with use of transfer coding). We only included discharges and readmissions to acute care hospitals and excluded hospitalizations in long‐term care facilities, rehabilitation facilities, skilled nursing homes, and other non‐acute care hospital facilities from being an index hospitalization. However, if the beneficiary was discharged to 1 of these facilities and then readmitted to an acute care hospital, the readmission was counted.
Each discharge was recorded as an independent event and we reset the readmission clock for a fresh 30‐day count each time the beneficiary was discharged. We examined the admission and readmission rate to determine if the rates varied by age, gender, reason for entitlement, racial characteristics, region of the country, number of chronic conditions, and whether the beneficiary is also enrolled in Medicaid (dual eligibles). We calculated the mean readmission rate for each diagnosis‐related group (DRG) and then used the probability of having a readmission for each DRG to calculate a case mix adjustment for each hospital. To calculate the chronic illness burden, we used a previously developed methodology for counting the number of chronic disease categories reported for the patient in the preceding year (2007).[8, 9] The classification system is maintained by the Agency for Health Care Research and Quality. We then used logistic regression to calculate the odds ratio of a discharge being readmitted based on these factors. We preformed statistical analysis using SAS version 9.1.3 (SAS Institute Inc., Cary, NC).
RESULTS
There were 434,999 hospital discharges that occurred in the first 9 months of 2008 in the 5% sample. There were 20.6% of Medicare beneficiaries hospitalized, and the overall readmission rate was 19.5%. Table 1 shows the odds ratios and 95% confidence intervals for the probability that a Medicare beneficiary will be readmitted within 30 days for variables including: age, sex, race, dual‐eligibility status, number of comorbid conditions, geographic region, and reason for entitlement. Of note, beneficiaries with 10 or more chronic conditions were more than 6 times more likely, and beneficiaries with 5 to 9 chronic conditions were more than 2.5 times more likely, to be readmitted than beneficiaries with 1 to 4 chronic conditions.
| Variable | Estimate | 95% Confidence Limits |
|---|---|---|
| ||
| Age 144 years | 1.634 | 1.5071.771 |
| Age 4564 years | 1.231 | 1.1421.327 |
| Age 7584 years | 1.048 | 1.0271.069 |
| Age 85+ years | 1.141 | 1.1151.168 |
| Age 6574 years | REF | |
| Male | 1.201 | 1.1831.220 |
| Black | 1.250 | 1.2211.280 |
| Other race | 1.071 | 1.0331.111 |
| White | REF | |
| Dual eligibles | 1.173 | 1.1511.195 |
| Northeast region | 1.146 | 1.1151.178 |
| Midwest region | 1.092 | 1.0631.122 |
| South region | 1.037 | 1.0111.063 |
| West region | REF | |
| 0 comorbidities | 0.255 | 0.1480.441 |
| 59 comorbidities | 2.533 | 2.4492.621 |
| 10+ comorbidities | 6.119 | 5.9136.332 |
| 14 comorbidities | REF | |
| Disabled | 0.817 | 0.7570.880 |
| ESRD | 1.327 | 1.2231.440 |
| Age >64 years | REF | |
DISCUSSION
The most interesting finding is that beneficiaries with 10 or more chronic conditions were more than 6 times more likely to be readmitted than beneficiaries with 1 to 4 chronic conditions. Beneficiaries with 10 or more chronic conditions represent only 8.9% of all Medicare beneficiaries (31.0% of all hospitalizations), but they were responsible for 50.2% of all readmissions. The 31.8% of beneficiaries with 5 to 9 chronic conditions (55.5% of all hospitalizations) had the second highest odds ratio (2.5) and were responsible for 45% of all readmissions. The 59.3% of beneficiaries with 5 comorbidities (13.6% of all hospitalizations) were associated with only 4.7% of all readmissions. This strongly suggests that hospitals focus their attention on beneficiaries with 10 or more comorbidities. These results were despite correction for DRG diagnosis in the model.
We recognize that the number of chronic conditions is a crude measure of health status because it weighs hundreds of different clinical conditions equally; however, it seems a good proxy for 3 closely allied concepts: (1) the overall burden of chronic illness carried by the patient, (2) the patient's level of engagement with the healthcare system (including number of unique providers), and (3) the number of conditions being treated. By providing a 1‐year window of a patient's health status, it is a more complete picture than any single hospital claim submission or indices based solely on hospital discharge data.
The other variables are less predictive of 30‐day readmissions. Beneficiaries over 85 years old are only 14% more likely, whereas disabled Medicare beneficiaries 44 years old are 63% more likely to be readmitted than beneficiaries between 65 and 74 years old. Men are 20% more likely to be readmitted than women. Black race and dual‐eligibility slightly increase rates of readmission. Beneficiaries located in the West have the lowest readmission rates. In comparison to those who are aged, those with end‐stage renal disease (ESRD) have a higher rate of readmission, and those with a disability have a lower rate of readmission. In considering the age and reason for entitlement findings, one would assume that ESRD was the driver of higher readmission rates in the younger Medicare population.
CMS will need to analyze which hospitals have higher than expected readmission rates, and this will require risk adjustment at each hospital. In addition to the number of chronic conditions and other variables shown in Table 1, other factors CMS might want to include when it starts doing readmissions for all discharges is the discharge diagnosis (because our results suggest there are significant differences in the probability of a readmission across DRGs). In addition, CMS will need to consider how to capture additional data not currently in the claims data, such as social factors like homelessness.
We recognize significant limitations to these findings. First, this analysis uses only information that is available from Medicare claims and administrative data. Claims give almost no information on how well the hospital planned the discharge, instructed the patient and family, or engaged follow‐up providers. Also, claims data tell us virtually nothing about a patient's health literacy or social situation. Second, the analysis relies on claims data, but this has little clinical detail. Third, these data are limited to persons enrolled in fee‐for‐service Medicare. Fourth, we included all readmissions, including some readmissions (such as chemotherapy and staged percutaneous coronary interventions) that were part of a planned treatment protocol.[10] Fifth, we were unable to distinguish same‐day readmissions versus transfers, and therefore excluded all same‐day readmissions from measurement.
As hospitals and physicians begin to plan for the regulations that will penalize hospitals with high readmission rates, they will need to strongly consider targeting beneficiaries with more than 10 chronic conditions.
Acknowledgments
The Commonwealth Fund provided a grant to Dr. Anderson to help support this work. The authors report no conflicts of interest.
- , . Hospital readmissions in the Medicare population. N Engl J Med. 1984;311:1349–1353.
- , , . Rehospitalizations among patients in the Medicare fee‐for‐service program. N Engl J Med. 2009;360:1418–1428.
- Patient Protection and Affordable Care Act. Section 3025. Available at: http://www.gpo.gov/fdsys/pkg/PLAW‐111publ148/pdf/PLAW‐111publ148.pdf. Accessed April 8, 2013.
- Congressional Budget Office.Patient Protection and Affordable Care Act. Available at: http://www.cbo.gov/doc.cfm?index=10868. Accessed April 8, 2013.
- , , , et al.2012 measures maintenance technical report: acute myocardial infarction, heart failure, and pneumonia 30‐day risk‐standardized readmission measures. Available at: http://www.qualitynet.org/dcs/ContentServer?c=Page78:27597–27599.
- , , , , . Hospital payment based on diagnosis‐related groups differs in Europe and holds lessons for the United States. Health Aff (Millwood). 2013;32:713–723.
- , , , . Out‐of‐pocket medical spending for care of chronic conditions. Health Aff (Millwood). 2001;20:267–278.
- MEPS data documentation HC‐006: 1996 medical conditions. Pub. no. 99‐DP06. Rockville, MD: AHRQ; 1999.
- , . Planned readmissions: a potential solution. Arch Intern Med. 2012;172:269–270.
For at least 25 years, approximately 20% of Medicare fee‐for‐service discharges have been followed by a hospital readmission within 30 days.[1, 2] Section 3025 of the Patient Protection and Affordable Care Act (ACA)[3] created escalating penalties for hospitals with higher than expected 30‐day readmission rates, and the Congressional Budget Office estimated this will reduce Medicare spending by over $7 billion between 2010 and 2019.[4]
Hospitals and physicians have begun developing strategies to identify which Medicare beneficiaries are most likely to be readmitted and use this information to design programs to reduce their readmission rate. Initially, penalties will be based on readmission rates after an index discharge with heart failure, myocardial infarction, and pneumonia.[5] Recently, the Centers for Medicare and Medicaid Services (CMS) released the Inpatient Prospective Payment System FY2014 proposed rule, which proposes to add 2 new readmission penalties beginning in FY2015: readmissions for hip/knee arthroplasty and chronic obstructive pulmonary disease.[6] Other countries are already penalizing hospitals with high readmission rates; for example, Germany is penalizing all readmissions that occur within a 30‐day period following admission.[7] In this brief report, we examine the characteristics of Medicare beneficiaries most likely to be readmitted within 30 days. We focus on readmission rates for all discharge conditions and all patient readmission rates, because we believe the language in the ACA ultimately points to an all‐inclusive approach.
METHODS
We used a nationally random 5% sample of all Medicare beneficiaries for the period between January 1, 2008 and September 30, 2008. To be included, beneficiaries must have both Part A and B coverage and live within the United States. Medicare Advantage patients were excluded because Medicare Advantage plans do not report the data in the same way as fee for service. We calculated the readmission rate as the number of admissions that were preceded by an at‐risk discharge within 30 days divided by the total number of at‐risk discharges. This definition included admissions to and discharges from sole community providers, Medicare‐dependent small rural hospitals, and critical access hospitals. We counted as at risk all live discharges from short‐term acute care hospitals that were not discharged against medical advice, discharged to a rehabilitation unit within an acute care hospital, or readmitted on day 0 (due to inconsistency with use of transfer coding). We only included discharges and readmissions to acute care hospitals and excluded hospitalizations in long‐term care facilities, rehabilitation facilities, skilled nursing homes, and other non‐acute care hospital facilities from being an index hospitalization. However, if the beneficiary was discharged to 1 of these facilities and then readmitted to an acute care hospital, the readmission was counted.
Each discharge was recorded as an independent event and we reset the readmission clock for a fresh 30‐day count each time the beneficiary was discharged. We examined the admission and readmission rate to determine if the rates varied by age, gender, reason for entitlement, racial characteristics, region of the country, number of chronic conditions, and whether the beneficiary is also enrolled in Medicaid (dual eligibles). We calculated the mean readmission rate for each diagnosis‐related group (DRG) and then used the probability of having a readmission for each DRG to calculate a case mix adjustment for each hospital. To calculate the chronic illness burden, we used a previously developed methodology for counting the number of chronic disease categories reported for the patient in the preceding year (2007).[8, 9] The classification system is maintained by the Agency for Health Care Research and Quality. We then used logistic regression to calculate the odds ratio of a discharge being readmitted based on these factors. We preformed statistical analysis using SAS version 9.1.3 (SAS Institute Inc., Cary, NC).
RESULTS
There were 434,999 hospital discharges that occurred in the first 9 months of 2008 in the 5% sample. There were 20.6% of Medicare beneficiaries hospitalized, and the overall readmission rate was 19.5%. Table 1 shows the odds ratios and 95% confidence intervals for the probability that a Medicare beneficiary will be readmitted within 30 days for variables including: age, sex, race, dual‐eligibility status, number of comorbid conditions, geographic region, and reason for entitlement. Of note, beneficiaries with 10 or more chronic conditions were more than 6 times more likely, and beneficiaries with 5 to 9 chronic conditions were more than 2.5 times more likely, to be readmitted than beneficiaries with 1 to 4 chronic conditions.
| Variable | Estimate | 95% Confidence Limits |
|---|---|---|
| ||
| Age 144 years | 1.634 | 1.5071.771 |
| Age 4564 years | 1.231 | 1.1421.327 |
| Age 7584 years | 1.048 | 1.0271.069 |
| Age 85+ years | 1.141 | 1.1151.168 |
| Age 6574 years | REF | |
| Male | 1.201 | 1.1831.220 |
| Black | 1.250 | 1.2211.280 |
| Other race | 1.071 | 1.0331.111 |
| White | REF | |
| Dual eligibles | 1.173 | 1.1511.195 |
| Northeast region | 1.146 | 1.1151.178 |
| Midwest region | 1.092 | 1.0631.122 |
| South region | 1.037 | 1.0111.063 |
| West region | REF | |
| 0 comorbidities | 0.255 | 0.1480.441 |
| 59 comorbidities | 2.533 | 2.4492.621 |
| 10+ comorbidities | 6.119 | 5.9136.332 |
| 14 comorbidities | REF | |
| Disabled | 0.817 | 0.7570.880 |
| ESRD | 1.327 | 1.2231.440 |
| Age >64 years | REF | |
DISCUSSION
The most interesting finding is that beneficiaries with 10 or more chronic conditions were more than 6 times more likely to be readmitted than beneficiaries with 1 to 4 chronic conditions. Beneficiaries with 10 or more chronic conditions represent only 8.9% of all Medicare beneficiaries (31.0% of all hospitalizations), but they were responsible for 50.2% of all readmissions. The 31.8% of beneficiaries with 5 to 9 chronic conditions (55.5% of all hospitalizations) had the second highest odds ratio (2.5) and were responsible for 45% of all readmissions. The 59.3% of beneficiaries with 5 comorbidities (13.6% of all hospitalizations) were associated with only 4.7% of all readmissions. This strongly suggests that hospitals focus their attention on beneficiaries with 10 or more comorbidities. These results were despite correction for DRG diagnosis in the model.
We recognize that the number of chronic conditions is a crude measure of health status because it weighs hundreds of different clinical conditions equally; however, it seems a good proxy for 3 closely allied concepts: (1) the overall burden of chronic illness carried by the patient, (2) the patient's level of engagement with the healthcare system (including number of unique providers), and (3) the number of conditions being treated. By providing a 1‐year window of a patient's health status, it is a more complete picture than any single hospital claim submission or indices based solely on hospital discharge data.
The other variables are less predictive of 30‐day readmissions. Beneficiaries over 85 years old are only 14% more likely, whereas disabled Medicare beneficiaries 44 years old are 63% more likely to be readmitted than beneficiaries between 65 and 74 years old. Men are 20% more likely to be readmitted than women. Black race and dual‐eligibility slightly increase rates of readmission. Beneficiaries located in the West have the lowest readmission rates. In comparison to those who are aged, those with end‐stage renal disease (ESRD) have a higher rate of readmission, and those with a disability have a lower rate of readmission. In considering the age and reason for entitlement findings, one would assume that ESRD was the driver of higher readmission rates in the younger Medicare population.
CMS will need to analyze which hospitals have higher than expected readmission rates, and this will require risk adjustment at each hospital. In addition to the number of chronic conditions and other variables shown in Table 1, other factors CMS might want to include when it starts doing readmissions for all discharges is the discharge diagnosis (because our results suggest there are significant differences in the probability of a readmission across DRGs). In addition, CMS will need to consider how to capture additional data not currently in the claims data, such as social factors like homelessness.
We recognize significant limitations to these findings. First, this analysis uses only information that is available from Medicare claims and administrative data. Claims give almost no information on how well the hospital planned the discharge, instructed the patient and family, or engaged follow‐up providers. Also, claims data tell us virtually nothing about a patient's health literacy or social situation. Second, the analysis relies on claims data, but this has little clinical detail. Third, these data are limited to persons enrolled in fee‐for‐service Medicare. Fourth, we included all readmissions, including some readmissions (such as chemotherapy and staged percutaneous coronary interventions) that were part of a planned treatment protocol.[10] Fifth, we were unable to distinguish same‐day readmissions versus transfers, and therefore excluded all same‐day readmissions from measurement.
As hospitals and physicians begin to plan for the regulations that will penalize hospitals with high readmission rates, they will need to strongly consider targeting beneficiaries with more than 10 chronic conditions.
Acknowledgments
The Commonwealth Fund provided a grant to Dr. Anderson to help support this work. The authors report no conflicts of interest.
For at least 25 years, approximately 20% of Medicare fee‐for‐service discharges have been followed by a hospital readmission within 30 days.[1, 2] Section 3025 of the Patient Protection and Affordable Care Act (ACA)[3] created escalating penalties for hospitals with higher than expected 30‐day readmission rates, and the Congressional Budget Office estimated this will reduce Medicare spending by over $7 billion between 2010 and 2019.[4]
Hospitals and physicians have begun developing strategies to identify which Medicare beneficiaries are most likely to be readmitted and use this information to design programs to reduce their readmission rate. Initially, penalties will be based on readmission rates after an index discharge with heart failure, myocardial infarction, and pneumonia.[5] Recently, the Centers for Medicare and Medicaid Services (CMS) released the Inpatient Prospective Payment System FY2014 proposed rule, which proposes to add 2 new readmission penalties beginning in FY2015: readmissions for hip/knee arthroplasty and chronic obstructive pulmonary disease.[6] Other countries are already penalizing hospitals with high readmission rates; for example, Germany is penalizing all readmissions that occur within a 30‐day period following admission.[7] In this brief report, we examine the characteristics of Medicare beneficiaries most likely to be readmitted within 30 days. We focus on readmission rates for all discharge conditions and all patient readmission rates, because we believe the language in the ACA ultimately points to an all‐inclusive approach.
METHODS
We used a nationally random 5% sample of all Medicare beneficiaries for the period between January 1, 2008 and September 30, 2008. To be included, beneficiaries must have both Part A and B coverage and live within the United States. Medicare Advantage patients were excluded because Medicare Advantage plans do not report the data in the same way as fee for service. We calculated the readmission rate as the number of admissions that were preceded by an at‐risk discharge within 30 days divided by the total number of at‐risk discharges. This definition included admissions to and discharges from sole community providers, Medicare‐dependent small rural hospitals, and critical access hospitals. We counted as at risk all live discharges from short‐term acute care hospitals that were not discharged against medical advice, discharged to a rehabilitation unit within an acute care hospital, or readmitted on day 0 (due to inconsistency with use of transfer coding). We only included discharges and readmissions to acute care hospitals and excluded hospitalizations in long‐term care facilities, rehabilitation facilities, skilled nursing homes, and other non‐acute care hospital facilities from being an index hospitalization. However, if the beneficiary was discharged to 1 of these facilities and then readmitted to an acute care hospital, the readmission was counted.
Each discharge was recorded as an independent event and we reset the readmission clock for a fresh 30‐day count each time the beneficiary was discharged. We examined the admission and readmission rate to determine if the rates varied by age, gender, reason for entitlement, racial characteristics, region of the country, number of chronic conditions, and whether the beneficiary is also enrolled in Medicaid (dual eligibles). We calculated the mean readmission rate for each diagnosis‐related group (DRG) and then used the probability of having a readmission for each DRG to calculate a case mix adjustment for each hospital. To calculate the chronic illness burden, we used a previously developed methodology for counting the number of chronic disease categories reported for the patient in the preceding year (2007).[8, 9] The classification system is maintained by the Agency for Health Care Research and Quality. We then used logistic regression to calculate the odds ratio of a discharge being readmitted based on these factors. We preformed statistical analysis using SAS version 9.1.3 (SAS Institute Inc., Cary, NC).
RESULTS
There were 434,999 hospital discharges that occurred in the first 9 months of 2008 in the 5% sample. There were 20.6% of Medicare beneficiaries hospitalized, and the overall readmission rate was 19.5%. Table 1 shows the odds ratios and 95% confidence intervals for the probability that a Medicare beneficiary will be readmitted within 30 days for variables including: age, sex, race, dual‐eligibility status, number of comorbid conditions, geographic region, and reason for entitlement. Of note, beneficiaries with 10 or more chronic conditions were more than 6 times more likely, and beneficiaries with 5 to 9 chronic conditions were more than 2.5 times more likely, to be readmitted than beneficiaries with 1 to 4 chronic conditions.
| Variable | Estimate | 95% Confidence Limits |
|---|---|---|
| ||
| Age 144 years | 1.634 | 1.5071.771 |
| Age 4564 years | 1.231 | 1.1421.327 |
| Age 7584 years | 1.048 | 1.0271.069 |
| Age 85+ years | 1.141 | 1.1151.168 |
| Age 6574 years | REF | |
| Male | 1.201 | 1.1831.220 |
| Black | 1.250 | 1.2211.280 |
| Other race | 1.071 | 1.0331.111 |
| White | REF | |
| Dual eligibles | 1.173 | 1.1511.195 |
| Northeast region | 1.146 | 1.1151.178 |
| Midwest region | 1.092 | 1.0631.122 |
| South region | 1.037 | 1.0111.063 |
| West region | REF | |
| 0 comorbidities | 0.255 | 0.1480.441 |
| 59 comorbidities | 2.533 | 2.4492.621 |
| 10+ comorbidities | 6.119 | 5.9136.332 |
| 14 comorbidities | REF | |
| Disabled | 0.817 | 0.7570.880 |
| ESRD | 1.327 | 1.2231.440 |
| Age >64 years | REF | |
DISCUSSION
The most interesting finding is that beneficiaries with 10 or more chronic conditions were more than 6 times more likely to be readmitted than beneficiaries with 1 to 4 chronic conditions. Beneficiaries with 10 or more chronic conditions represent only 8.9% of all Medicare beneficiaries (31.0% of all hospitalizations), but they were responsible for 50.2% of all readmissions. The 31.8% of beneficiaries with 5 to 9 chronic conditions (55.5% of all hospitalizations) had the second highest odds ratio (2.5) and were responsible for 45% of all readmissions. The 59.3% of beneficiaries with 5 comorbidities (13.6% of all hospitalizations) were associated with only 4.7% of all readmissions. This strongly suggests that hospitals focus their attention on beneficiaries with 10 or more comorbidities. These results were despite correction for DRG diagnosis in the model.
We recognize that the number of chronic conditions is a crude measure of health status because it weighs hundreds of different clinical conditions equally; however, it seems a good proxy for 3 closely allied concepts: (1) the overall burden of chronic illness carried by the patient, (2) the patient's level of engagement with the healthcare system (including number of unique providers), and (3) the number of conditions being treated. By providing a 1‐year window of a patient's health status, it is a more complete picture than any single hospital claim submission or indices based solely on hospital discharge data.
The other variables are less predictive of 30‐day readmissions. Beneficiaries over 85 years old are only 14% more likely, whereas disabled Medicare beneficiaries 44 years old are 63% more likely to be readmitted than beneficiaries between 65 and 74 years old. Men are 20% more likely to be readmitted than women. Black race and dual‐eligibility slightly increase rates of readmission. Beneficiaries located in the West have the lowest readmission rates. In comparison to those who are aged, those with end‐stage renal disease (ESRD) have a higher rate of readmission, and those with a disability have a lower rate of readmission. In considering the age and reason for entitlement findings, one would assume that ESRD was the driver of higher readmission rates in the younger Medicare population.
CMS will need to analyze which hospitals have higher than expected readmission rates, and this will require risk adjustment at each hospital. In addition to the number of chronic conditions and other variables shown in Table 1, other factors CMS might want to include when it starts doing readmissions for all discharges is the discharge diagnosis (because our results suggest there are significant differences in the probability of a readmission across DRGs). In addition, CMS will need to consider how to capture additional data not currently in the claims data, such as social factors like homelessness.
We recognize significant limitations to these findings. First, this analysis uses only information that is available from Medicare claims and administrative data. Claims give almost no information on how well the hospital planned the discharge, instructed the patient and family, or engaged follow‐up providers. Also, claims data tell us virtually nothing about a patient's health literacy or social situation. Second, the analysis relies on claims data, but this has little clinical detail. Third, these data are limited to persons enrolled in fee‐for‐service Medicare. Fourth, we included all readmissions, including some readmissions (such as chemotherapy and staged percutaneous coronary interventions) that were part of a planned treatment protocol.[10] Fifth, we were unable to distinguish same‐day readmissions versus transfers, and therefore excluded all same‐day readmissions from measurement.
As hospitals and physicians begin to plan for the regulations that will penalize hospitals with high readmission rates, they will need to strongly consider targeting beneficiaries with more than 10 chronic conditions.
Acknowledgments
The Commonwealth Fund provided a grant to Dr. Anderson to help support this work. The authors report no conflicts of interest.
- , . Hospital readmissions in the Medicare population. N Engl J Med. 1984;311:1349–1353.
- , , . Rehospitalizations among patients in the Medicare fee‐for‐service program. N Engl J Med. 2009;360:1418–1428.
- Patient Protection and Affordable Care Act. Section 3025. Available at: http://www.gpo.gov/fdsys/pkg/PLAW‐111publ148/pdf/PLAW‐111publ148.pdf. Accessed April 8, 2013.
- Congressional Budget Office.Patient Protection and Affordable Care Act. Available at: http://www.cbo.gov/doc.cfm?index=10868. Accessed April 8, 2013.
- , , , et al.2012 measures maintenance technical report: acute myocardial infarction, heart failure, and pneumonia 30‐day risk‐standardized readmission measures. Available at: http://www.qualitynet.org/dcs/ContentServer?c=Page78:27597–27599.
- , , , , . Hospital payment based on diagnosis‐related groups differs in Europe and holds lessons for the United States. Health Aff (Millwood). 2013;32:713–723.
- , , , . Out‐of‐pocket medical spending for care of chronic conditions. Health Aff (Millwood). 2001;20:267–278.
- MEPS data documentation HC‐006: 1996 medical conditions. Pub. no. 99‐DP06. Rockville, MD: AHRQ; 1999.
- , . Planned readmissions: a potential solution. Arch Intern Med. 2012;172:269–270.
- , . Hospital readmissions in the Medicare population. N Engl J Med. 1984;311:1349–1353.
- , , . Rehospitalizations among patients in the Medicare fee‐for‐service program. N Engl J Med. 2009;360:1418–1428.
- Patient Protection and Affordable Care Act. Section 3025. Available at: http://www.gpo.gov/fdsys/pkg/PLAW‐111publ148/pdf/PLAW‐111publ148.pdf. Accessed April 8, 2013.
- Congressional Budget Office.Patient Protection and Affordable Care Act. Available at: http://www.cbo.gov/doc.cfm?index=10868. Accessed April 8, 2013.
- , , , et al.2012 measures maintenance technical report: acute myocardial infarction, heart failure, and pneumonia 30‐day risk‐standardized readmission measures. Available at: http://www.qualitynet.org/dcs/ContentServer?c=Page78:27597–27599.
- , , , , . Hospital payment based on diagnosis‐related groups differs in Europe and holds lessons for the United States. Health Aff (Millwood). 2013;32:713–723.
- , , , . Out‐of‐pocket medical spending for care of chronic conditions. Health Aff (Millwood). 2001;20:267–278.
- MEPS data documentation HC‐006: 1996 medical conditions. Pub. no. 99‐DP06. Rockville, MD: AHRQ; 1999.
- , . Planned readmissions: a potential solution. Arch Intern Med. 2012;172:269–270.
Promoting Professionalism
Unprofessional behavior in the inpatient setting has the potential to impact care delivery and the quality of trainee's educational experience. These behaviors, from disparaging colleagues to blocking admissions, can negatively impact the learning environment. The learning environment or conditions created by the patient care team's actions play a critical role in the development of trainees.[1, 2] The rising presence of hospitalists in the inpatient setting raises the question of how their actions impact the learning environment. Professional behavior has been defined as a core competency for hospitalists by the Society of Hospital Medicine.[3] Professional behavior of all team members, from faculty to trainee, can impact the learning environment and patient safety.[4, 5] However, few educational materials exist to train faculty and housestaff on recognizing and ameliorating unprofessional behaviors.
A prior assessment regarding hospitalists' lapses in professionalism identified scenarios that demonstrated increased participation by hospitalists at 3 institutions.[6] Participants reported observation or participation in specific unprofessional behaviors and rated their perception of these behaviors. Additional work within those residency environments demonstrated that residents' perceptions of and participation in these behaviors increased throughout training, with environmental characteristics, specifically faculty behavior, influencing trainee professional development and acclimation of these behaviors.[7, 8]
Although overall participation in egregious behavior was low, resident participation in 3 categories of unprofessional behavior increased during internship. Those scenarios included disparaging the emergency room or primary care physician for missed findings or management decisions, blocking or not taking admissions appropriate for the service in question, and misrepresenting a test as urgent to expedite obtaining the test. We developed our intervention focused on these areas to address professionalism lapses that occur during internship. Our earlier work showed faculty role models influenced trainee behavior. For this reason, we provided education to both residents and hospitalists to maximize the impact of the intervention.
We present here a novel, interactive, video‐based workshop curriculum for faculty and trainees that aims to illustrate unprofessional behaviors and outlines the role faculty may play in promoting such behaviors. In addition, we review the result of postworkshop evaluation on intent to change behavior and satisfaction.
METHODS
A grant from the American Board of Internal Medicine Foundation supported this project. The working group that resulted, the Chicago Professional Practice Project and Outcomes, included faculty representation from 3 Chicago‐area hospitals: the University of Chicago, Northwestern University, and NorthShore University HealthSystem. Academic hospitalists at these sites were invited to participate. Each site also has an internal medicine residency program in which hospitalists were expected to attend the teaching service. Given this, resident trainees at all participating sites, and 1 community teaching affiliate program (Mercy Hospital and Medical Center) where academic hospitalists at the University of Chicago rotate, were recruited for participation. Faculty champions were identified for each site, and 1 internal and external faculty representative from the working group served to debrief and facilitate. Trainee workshops were administered by 1 internal and external collaborator, and for the community site, 2 external faculty members. Workshops were held during established educational conference times, and lunch was provided.
Scripts highlighting each of the behaviors identified in the prior survey were developed and peer reviewed for clarity and face validity across the 3 sites. Medical student and resident actors were trained utilizing the finalized scripts, and a performance artist affiliated with the Screen Actors Guild assisted in their preparation for filming. All videos were filmed at the University of Chicago Pritzker School of Medicine Clinical Performance Center. The final videos ranged in length from 4 to 7 minutes and included title, cast, and funding source. As an example, 1 video highlighted the unprofessional behavior of misrepresenting a test as urgent to prioritize one's patient in the queue. This video included a resident, intern, and attending on inpatient rounds during which the resident encouraged the intern to misrepresent the patient's status to expedite obtaining the study and facilitate the patient's discharge. The resident stressed that he would be in the clinic and had many patients to see, highlighting the impact of workload on unprofessional behavior, and aggressively persuaded the intern to sell her test to have it performed the same day. When this occurred, the attending applauded the intern for her strong work.
A moderator guide and debriefing tools were developed to facilitate discussion. The duration of each of the workshops was approximately 60 minutes. After welcoming remarks, participants were provided tools to utilize during the viewing of each video. These checklists noted the roles of those depicted in the video, asked to identify positive or negative behaviors displayed, and included questions regarding how behaviors could be detrimental and how the situation could have been prevented. After viewing the videos, participants divided into small groups to discuss the individual exhibiting the unprofessional behavior, their perceived motivation for said behavior, and its impact on the team culture and patient care. Following a small‐group discussion, large‐group debriefing was performed, addressing the barriers and facilitators to professional behavior. Two videos were shown at each workshop, and participants completed a postworkshop evaluation. Videos chosen for viewing were based upon preworkshop survey results that highlighted areas of concern at that specific site.
Postworkshop paper‐based evaluations assessed participants' perception of displayed behaviors on a Likert‐type scale (1=unprofessional to 5=professional) utilizing items validated in prior work,[6, 7, 8] their level of agreement regarding the impact of video‐based exercises, and intent to change behavior using a Likert‐type scale (1=strongly disagree to 5=strongly agree). A constructed‐response section for comments regarding their experience was included. Descriptive statistics and Wilcoxon rank sum analyses were performed.
RESULTS
Forty‐four academic hospitalist faculty members (44/83; 53%) and 244 resident trainees (244/356; 68%) participated. When queried regarding their perception of the displayed behaviors in the videos, nearly 100% of faculty and trainees felt disparaging the emergency department or primary care physician for missed findings or clinical decisions was somewhat unprofessional or unprofessional. Ninety percent of hospitalists and 93% of trainees rated celebrating a blocked admission as somewhat unprofessional or unprofessional (Table 1).
| Behavior | Faculty Rated as Unprofessional or Somewhat Unprofessional (n = 44) | Housestaff Rated as Unprofessional or Somewhat Unprofessional (n=244) |
|---|---|---|
| ||
| Disparaging the ED/PCP to colleagues for findings later discovered on the floor or patient care management decisions | 95.6% | 97.5% |
| Refusing an admission that could be considered appropriate for your service (eg, blocking) | 86.4% | 95.1% |
| Celebrating a blocked admission | 90.1% | 93.0% |
| Ordering a routine test as urgent to get it expedited | 77.2% | 80.3% |
The scenarios portrayed were well received, with more than 85% of faculty and trainees agreeing that the behaviors displayed were realistic. Those who perceived videos as very realistic were more likely to report intent to change behavior (93% vs 53%, P=0.01). Nearly two‐thirds of faculty and 67% of housestaff expressed agreement that they intended to change behavior based upon the experience (Table 2).
| Evaluation Item | Faculty Level of Agreement (StronglyAgree or Agree) (n=44) | Housestaff Level of Agreement (Strongly Agree or Agree) (n=244) |
|---|---|---|
| The scenarios portrayed in the videos were realistic | 86.4% | 86.9% |
| I will change my behavior as a result of this exercise | 65.9% | 67.2% |
| I feel that this was a useful and effective exercise | 65.9% | 77.1% |
Qualitative comments in the constructed‐response portion of the evaluation noted the effectiveness of the interactive materials. In addition, the need for focused faculty development was identified by 1 respondent who stated: If unprofessional behavior is the unwritten curriculum, there needs to be an explicit, written curriculum to address it. Finally, the aim of facilitating self‐reflection is echoed in this faculty respondent's comment: Always good to be reminded of our behaviors and the influence they have on others and from this resident physician It helps to re‐evaluate how you talk to people.
CONCLUSIONS
Faculty can be a large determinant of the learning environment and impact trainees' professional development.[9] Hospitalists should be encouraged to embrace faculty role‐modeling of effective professional behaviors, especially given their increased presence in the inpatient learning environment. In addition, resident trainees and their behaviors contribute to the learning environment and influence the further professional development of more junior trainees.[10] Targeting professionalism education toward previously identified and prevalent unprofessional behaviors in the inpatient care of patients may serve to affect the most change among providers who practice in this setting. Individualized assessment of the learning environment may aid in identifying common scenarios that may plague a specific learning culture, allowing for relevant and targeted discussion of factors that promote and perpetuate such behaviors.[11]
Interactive, video‐based modules provided an effective way to promote interactive reflection and robust discussion. This model of experiential learning is an effective form of professional development as it engages the learner and stimulates ongoing incorporation of the topics addressed.[12, 13] Creating a shared concrete experience among targeted learners, using the video‐based scenarios, stimulates reflective observation, and ultimately experimentation, or incorporation into practice.[14]
There are several limitations to our evaluation including that we focused solely on academic hospitalist programs, and our sample size for faculty and residents was small. Also, we only addressed a small, though representative, sample of unprofessional behaviors and have not yet linked intervention to actual behavior change. Finally, the script scenarios that we used in this study were not previously published as they were created specifically for this intervention. Validity evidence for these scenarios include that they were based upon the results of earlier work from our institutions and underwent thorough peer review for content and clarity. Further studies will be required to do this. However, we do believe that these are positive findings for utilizing this type of interactive curriculum for professionalism education to promote self‐reflection and behavior change.
Video‐based professionalism education is a feasible, interactive mechanism to encourage self‐reflection and intent to change behavior among faculty and resident physicians. Future study is underway to conduct longitudinal assessments of the learning environments at the participating institutions to assess culture change, perceptions of behaviors, and sustainability of this type of intervention.
Disclosures: The authors acknowledge funding from the American Board of Internal Medicine. The funders had no role in the design of the study; the collection, analysis, and interpretation of the data; or the decision to approve publication of the finished manuscript. Results from this work have been presented at the Midwest Society of General Internal Medicine Regional Meeting, Chicago, Illinois, September 2011; Midwest Society of Hospital Medicine Regional Meeting, Chicago, Illinois, October 2011, and Society of Hospital Medicine Annual Meeting, San Diego, California, April 2012. The authors declare that they do not have any conflicts of interest to disclose.
- Liaison Committee on Medical Education. Functions and structure of a medical school. Available at: http://www.lcme.org/functions.pdf. Accessed October 10, 2012.
- , , , , . Residents' perceptions of their own professionalism and the professionalism of their learning environment. J Grad Med Educ. 2009;1:208–215.
- Society of Hospital Medicine. The core competencies in hospital medicine. http://www.hospitalmedicine.org/Content/NavigationMenu/Education/CoreCurriculum/Core_Competencies.htm. Accessed October 10, 2012.
- The Joint Commission. Behaviors that undermine a culture of safety. Sentinel Event Alert. 2008;(40):1–3. http://www.jointcommission.org/assets/1/18/SEA_40.pdf. Accessed October 10, 2012.
- , . A survey of the impact of disruptive behaviors and communication defects on patient safety. Jt Comm J Qual Patient Saf. 2008;34:464–471.
- , , , et al. Participation in unprofessional behaviors among hospitalists: a multicenter study. J Hosp Med. 2012;7(7):543–550.
- , , et al. Participation in and perceptions of unprofessional behaviors among incoming internal medicine interns. JAMA. 2008;300:1132–1134.
- , , , et al., Changes in perception of and participation in unprofessional behaviors during internship. Acad Med. 2010;85:S76–S80.
- , , , et al. Perspective: beyond counting hours: the importance of supervision, professionalism, transitions of care, and workload in residency training. Acad Med. 2012;87(7):883–888.
- , . The role of the student‐teacher relationship in the formation of physicians: the hidden curriculum as process. J Gen Intern Med. 2006;21:S16–S20.
- , , , et al. Evidence for validity of a survey to measure the learning environment for professionalism. Med Teach. 2011;33(12):e683–e688.
- . Experiential Learning: Experience as the Source of Learning and Development. Englewood Cliffs, NJ: Prentice Hall; 1984.
- , . How can physicians' learning style drive educational planning? Acad Med. 2005;80:680–84.
- , . Twenty years of experience using trigger films as a teaching tool. Acad Med. 2001;76:656–658.
Unprofessional behavior in the inpatient setting has the potential to impact care delivery and the quality of trainee's educational experience. These behaviors, from disparaging colleagues to blocking admissions, can negatively impact the learning environment. The learning environment or conditions created by the patient care team's actions play a critical role in the development of trainees.[1, 2] The rising presence of hospitalists in the inpatient setting raises the question of how their actions impact the learning environment. Professional behavior has been defined as a core competency for hospitalists by the Society of Hospital Medicine.[3] Professional behavior of all team members, from faculty to trainee, can impact the learning environment and patient safety.[4, 5] However, few educational materials exist to train faculty and housestaff on recognizing and ameliorating unprofessional behaviors.
A prior assessment regarding hospitalists' lapses in professionalism identified scenarios that demonstrated increased participation by hospitalists at 3 institutions.[6] Participants reported observation or participation in specific unprofessional behaviors and rated their perception of these behaviors. Additional work within those residency environments demonstrated that residents' perceptions of and participation in these behaviors increased throughout training, with environmental characteristics, specifically faculty behavior, influencing trainee professional development and acclimation of these behaviors.[7, 8]
Although overall participation in egregious behavior was low, resident participation in 3 categories of unprofessional behavior increased during internship. Those scenarios included disparaging the emergency room or primary care physician for missed findings or management decisions, blocking or not taking admissions appropriate for the service in question, and misrepresenting a test as urgent to expedite obtaining the test. We developed our intervention focused on these areas to address professionalism lapses that occur during internship. Our earlier work showed faculty role models influenced trainee behavior. For this reason, we provided education to both residents and hospitalists to maximize the impact of the intervention.
We present here a novel, interactive, video‐based workshop curriculum for faculty and trainees that aims to illustrate unprofessional behaviors and outlines the role faculty may play in promoting such behaviors. In addition, we review the result of postworkshop evaluation on intent to change behavior and satisfaction.
METHODS
A grant from the American Board of Internal Medicine Foundation supported this project. The working group that resulted, the Chicago Professional Practice Project and Outcomes, included faculty representation from 3 Chicago‐area hospitals: the University of Chicago, Northwestern University, and NorthShore University HealthSystem. Academic hospitalists at these sites were invited to participate. Each site also has an internal medicine residency program in which hospitalists were expected to attend the teaching service. Given this, resident trainees at all participating sites, and 1 community teaching affiliate program (Mercy Hospital and Medical Center) where academic hospitalists at the University of Chicago rotate, were recruited for participation. Faculty champions were identified for each site, and 1 internal and external faculty representative from the working group served to debrief and facilitate. Trainee workshops were administered by 1 internal and external collaborator, and for the community site, 2 external faculty members. Workshops were held during established educational conference times, and lunch was provided.
Scripts highlighting each of the behaviors identified in the prior survey were developed and peer reviewed for clarity and face validity across the 3 sites. Medical student and resident actors were trained utilizing the finalized scripts, and a performance artist affiliated with the Screen Actors Guild assisted in their preparation for filming. All videos were filmed at the University of Chicago Pritzker School of Medicine Clinical Performance Center. The final videos ranged in length from 4 to 7 minutes and included title, cast, and funding source. As an example, 1 video highlighted the unprofessional behavior of misrepresenting a test as urgent to prioritize one's patient in the queue. This video included a resident, intern, and attending on inpatient rounds during which the resident encouraged the intern to misrepresent the patient's status to expedite obtaining the study and facilitate the patient's discharge. The resident stressed that he would be in the clinic and had many patients to see, highlighting the impact of workload on unprofessional behavior, and aggressively persuaded the intern to sell her test to have it performed the same day. When this occurred, the attending applauded the intern for her strong work.
A moderator guide and debriefing tools were developed to facilitate discussion. The duration of each of the workshops was approximately 60 minutes. After welcoming remarks, participants were provided tools to utilize during the viewing of each video. These checklists noted the roles of those depicted in the video, asked to identify positive or negative behaviors displayed, and included questions regarding how behaviors could be detrimental and how the situation could have been prevented. After viewing the videos, participants divided into small groups to discuss the individual exhibiting the unprofessional behavior, their perceived motivation for said behavior, and its impact on the team culture and patient care. Following a small‐group discussion, large‐group debriefing was performed, addressing the barriers and facilitators to professional behavior. Two videos were shown at each workshop, and participants completed a postworkshop evaluation. Videos chosen for viewing were based upon preworkshop survey results that highlighted areas of concern at that specific site.
Postworkshop paper‐based evaluations assessed participants' perception of displayed behaviors on a Likert‐type scale (1=unprofessional to 5=professional) utilizing items validated in prior work,[6, 7, 8] their level of agreement regarding the impact of video‐based exercises, and intent to change behavior using a Likert‐type scale (1=strongly disagree to 5=strongly agree). A constructed‐response section for comments regarding their experience was included. Descriptive statistics and Wilcoxon rank sum analyses were performed.
RESULTS
Forty‐four academic hospitalist faculty members (44/83; 53%) and 244 resident trainees (244/356; 68%) participated. When queried regarding their perception of the displayed behaviors in the videos, nearly 100% of faculty and trainees felt disparaging the emergency department or primary care physician for missed findings or clinical decisions was somewhat unprofessional or unprofessional. Ninety percent of hospitalists and 93% of trainees rated celebrating a blocked admission as somewhat unprofessional or unprofessional (Table 1).
| Behavior | Faculty Rated as Unprofessional or Somewhat Unprofessional (n = 44) | Housestaff Rated as Unprofessional or Somewhat Unprofessional (n=244) |
|---|---|---|
| ||
| Disparaging the ED/PCP to colleagues for findings later discovered on the floor or patient care management decisions | 95.6% | 97.5% |
| Refusing an admission that could be considered appropriate for your service (eg, blocking) | 86.4% | 95.1% |
| Celebrating a blocked admission | 90.1% | 93.0% |
| Ordering a routine test as urgent to get it expedited | 77.2% | 80.3% |
The scenarios portrayed were well received, with more than 85% of faculty and trainees agreeing that the behaviors displayed were realistic. Those who perceived videos as very realistic were more likely to report intent to change behavior (93% vs 53%, P=0.01). Nearly two‐thirds of faculty and 67% of housestaff expressed agreement that they intended to change behavior based upon the experience (Table 2).
| Evaluation Item | Faculty Level of Agreement (StronglyAgree or Agree) (n=44) | Housestaff Level of Agreement (Strongly Agree or Agree) (n=244) |
|---|---|---|
| The scenarios portrayed in the videos were realistic | 86.4% | 86.9% |
| I will change my behavior as a result of this exercise | 65.9% | 67.2% |
| I feel that this was a useful and effective exercise | 65.9% | 77.1% |
Qualitative comments in the constructed‐response portion of the evaluation noted the effectiveness of the interactive materials. In addition, the need for focused faculty development was identified by 1 respondent who stated: If unprofessional behavior is the unwritten curriculum, there needs to be an explicit, written curriculum to address it. Finally, the aim of facilitating self‐reflection is echoed in this faculty respondent's comment: Always good to be reminded of our behaviors and the influence they have on others and from this resident physician It helps to re‐evaluate how you talk to people.
CONCLUSIONS
Faculty can be a large determinant of the learning environment and impact trainees' professional development.[9] Hospitalists should be encouraged to embrace faculty role‐modeling of effective professional behaviors, especially given their increased presence in the inpatient learning environment. In addition, resident trainees and their behaviors contribute to the learning environment and influence the further professional development of more junior trainees.[10] Targeting professionalism education toward previously identified and prevalent unprofessional behaviors in the inpatient care of patients may serve to affect the most change among providers who practice in this setting. Individualized assessment of the learning environment may aid in identifying common scenarios that may plague a specific learning culture, allowing for relevant and targeted discussion of factors that promote and perpetuate such behaviors.[11]
Interactive, video‐based modules provided an effective way to promote interactive reflection and robust discussion. This model of experiential learning is an effective form of professional development as it engages the learner and stimulates ongoing incorporation of the topics addressed.[12, 13] Creating a shared concrete experience among targeted learners, using the video‐based scenarios, stimulates reflective observation, and ultimately experimentation, or incorporation into practice.[14]
There are several limitations to our evaluation including that we focused solely on academic hospitalist programs, and our sample size for faculty and residents was small. Also, we only addressed a small, though representative, sample of unprofessional behaviors and have not yet linked intervention to actual behavior change. Finally, the script scenarios that we used in this study were not previously published as they were created specifically for this intervention. Validity evidence for these scenarios include that they were based upon the results of earlier work from our institutions and underwent thorough peer review for content and clarity. Further studies will be required to do this. However, we do believe that these are positive findings for utilizing this type of interactive curriculum for professionalism education to promote self‐reflection and behavior change.
Video‐based professionalism education is a feasible, interactive mechanism to encourage self‐reflection and intent to change behavior among faculty and resident physicians. Future study is underway to conduct longitudinal assessments of the learning environments at the participating institutions to assess culture change, perceptions of behaviors, and sustainability of this type of intervention.
Disclosures: The authors acknowledge funding from the American Board of Internal Medicine. The funders had no role in the design of the study; the collection, analysis, and interpretation of the data; or the decision to approve publication of the finished manuscript. Results from this work have been presented at the Midwest Society of General Internal Medicine Regional Meeting, Chicago, Illinois, September 2011; Midwest Society of Hospital Medicine Regional Meeting, Chicago, Illinois, October 2011, and Society of Hospital Medicine Annual Meeting, San Diego, California, April 2012. The authors declare that they do not have any conflicts of interest to disclose.
Unprofessional behavior in the inpatient setting has the potential to impact care delivery and the quality of trainee's educational experience. These behaviors, from disparaging colleagues to blocking admissions, can negatively impact the learning environment. The learning environment or conditions created by the patient care team's actions play a critical role in the development of trainees.[1, 2] The rising presence of hospitalists in the inpatient setting raises the question of how their actions impact the learning environment. Professional behavior has been defined as a core competency for hospitalists by the Society of Hospital Medicine.[3] Professional behavior of all team members, from faculty to trainee, can impact the learning environment and patient safety.[4, 5] However, few educational materials exist to train faculty and housestaff on recognizing and ameliorating unprofessional behaviors.
A prior assessment regarding hospitalists' lapses in professionalism identified scenarios that demonstrated increased participation by hospitalists at 3 institutions.[6] Participants reported observation or participation in specific unprofessional behaviors and rated their perception of these behaviors. Additional work within those residency environments demonstrated that residents' perceptions of and participation in these behaviors increased throughout training, with environmental characteristics, specifically faculty behavior, influencing trainee professional development and acclimation of these behaviors.[7, 8]
Although overall participation in egregious behavior was low, resident participation in 3 categories of unprofessional behavior increased during internship. Those scenarios included disparaging the emergency room or primary care physician for missed findings or management decisions, blocking or not taking admissions appropriate for the service in question, and misrepresenting a test as urgent to expedite obtaining the test. We developed our intervention focused on these areas to address professionalism lapses that occur during internship. Our earlier work showed faculty role models influenced trainee behavior. For this reason, we provided education to both residents and hospitalists to maximize the impact of the intervention.
We present here a novel, interactive, video‐based workshop curriculum for faculty and trainees that aims to illustrate unprofessional behaviors and outlines the role faculty may play in promoting such behaviors. In addition, we review the result of postworkshop evaluation on intent to change behavior and satisfaction.
METHODS
A grant from the American Board of Internal Medicine Foundation supported this project. The working group that resulted, the Chicago Professional Practice Project and Outcomes, included faculty representation from 3 Chicago‐area hospitals: the University of Chicago, Northwestern University, and NorthShore University HealthSystem. Academic hospitalists at these sites were invited to participate. Each site also has an internal medicine residency program in which hospitalists were expected to attend the teaching service. Given this, resident trainees at all participating sites, and 1 community teaching affiliate program (Mercy Hospital and Medical Center) where academic hospitalists at the University of Chicago rotate, were recruited for participation. Faculty champions were identified for each site, and 1 internal and external faculty representative from the working group served to debrief and facilitate. Trainee workshops were administered by 1 internal and external collaborator, and for the community site, 2 external faculty members. Workshops were held during established educational conference times, and lunch was provided.
Scripts highlighting each of the behaviors identified in the prior survey were developed and peer reviewed for clarity and face validity across the 3 sites. Medical student and resident actors were trained utilizing the finalized scripts, and a performance artist affiliated with the Screen Actors Guild assisted in their preparation for filming. All videos were filmed at the University of Chicago Pritzker School of Medicine Clinical Performance Center. The final videos ranged in length from 4 to 7 minutes and included title, cast, and funding source. As an example, 1 video highlighted the unprofessional behavior of misrepresenting a test as urgent to prioritize one's patient in the queue. This video included a resident, intern, and attending on inpatient rounds during which the resident encouraged the intern to misrepresent the patient's status to expedite obtaining the study and facilitate the patient's discharge. The resident stressed that he would be in the clinic and had many patients to see, highlighting the impact of workload on unprofessional behavior, and aggressively persuaded the intern to sell her test to have it performed the same day. When this occurred, the attending applauded the intern for her strong work.
A moderator guide and debriefing tools were developed to facilitate discussion. The duration of each of the workshops was approximately 60 minutes. After welcoming remarks, participants were provided tools to utilize during the viewing of each video. These checklists noted the roles of those depicted in the video, asked to identify positive or negative behaviors displayed, and included questions regarding how behaviors could be detrimental and how the situation could have been prevented. After viewing the videos, participants divided into small groups to discuss the individual exhibiting the unprofessional behavior, their perceived motivation for said behavior, and its impact on the team culture and patient care. Following a small‐group discussion, large‐group debriefing was performed, addressing the barriers and facilitators to professional behavior. Two videos were shown at each workshop, and participants completed a postworkshop evaluation. Videos chosen for viewing were based upon preworkshop survey results that highlighted areas of concern at that specific site.
Postworkshop paper‐based evaluations assessed participants' perception of displayed behaviors on a Likert‐type scale (1=unprofessional to 5=professional) utilizing items validated in prior work,[6, 7, 8] their level of agreement regarding the impact of video‐based exercises, and intent to change behavior using a Likert‐type scale (1=strongly disagree to 5=strongly agree). A constructed‐response section for comments regarding their experience was included. Descriptive statistics and Wilcoxon rank sum analyses were performed.
RESULTS
Forty‐four academic hospitalist faculty members (44/83; 53%) and 244 resident trainees (244/356; 68%) participated. When queried regarding their perception of the displayed behaviors in the videos, nearly 100% of faculty and trainees felt disparaging the emergency department or primary care physician for missed findings or clinical decisions was somewhat unprofessional or unprofessional. Ninety percent of hospitalists and 93% of trainees rated celebrating a blocked admission as somewhat unprofessional or unprofessional (Table 1).
| Behavior | Faculty Rated as Unprofessional or Somewhat Unprofessional (n = 44) | Housestaff Rated as Unprofessional or Somewhat Unprofessional (n=244) |
|---|---|---|
| ||
| Disparaging the ED/PCP to colleagues for findings later discovered on the floor or patient care management decisions | 95.6% | 97.5% |
| Refusing an admission that could be considered appropriate for your service (eg, blocking) | 86.4% | 95.1% |
| Celebrating a blocked admission | 90.1% | 93.0% |
| Ordering a routine test as urgent to get it expedited | 77.2% | 80.3% |
The scenarios portrayed were well received, with more than 85% of faculty and trainees agreeing that the behaviors displayed were realistic. Those who perceived videos as very realistic were more likely to report intent to change behavior (93% vs 53%, P=0.01). Nearly two‐thirds of faculty and 67% of housestaff expressed agreement that they intended to change behavior based upon the experience (Table 2).
| Evaluation Item | Faculty Level of Agreement (StronglyAgree or Agree) (n=44) | Housestaff Level of Agreement (Strongly Agree or Agree) (n=244) |
|---|---|---|
| The scenarios portrayed in the videos were realistic | 86.4% | 86.9% |
| I will change my behavior as a result of this exercise | 65.9% | 67.2% |
| I feel that this was a useful and effective exercise | 65.9% | 77.1% |
Qualitative comments in the constructed‐response portion of the evaluation noted the effectiveness of the interactive materials. In addition, the need for focused faculty development was identified by 1 respondent who stated: If unprofessional behavior is the unwritten curriculum, there needs to be an explicit, written curriculum to address it. Finally, the aim of facilitating self‐reflection is echoed in this faculty respondent's comment: Always good to be reminded of our behaviors and the influence they have on others and from this resident physician It helps to re‐evaluate how you talk to people.
CONCLUSIONS
Faculty can be a large determinant of the learning environment and impact trainees' professional development.[9] Hospitalists should be encouraged to embrace faculty role‐modeling of effective professional behaviors, especially given their increased presence in the inpatient learning environment. In addition, resident trainees and their behaviors contribute to the learning environment and influence the further professional development of more junior trainees.[10] Targeting professionalism education toward previously identified and prevalent unprofessional behaviors in the inpatient care of patients may serve to affect the most change among providers who practice in this setting. Individualized assessment of the learning environment may aid in identifying common scenarios that may plague a specific learning culture, allowing for relevant and targeted discussion of factors that promote and perpetuate such behaviors.[11]
Interactive, video‐based modules provided an effective way to promote interactive reflection and robust discussion. This model of experiential learning is an effective form of professional development as it engages the learner and stimulates ongoing incorporation of the topics addressed.[12, 13] Creating a shared concrete experience among targeted learners, using the video‐based scenarios, stimulates reflective observation, and ultimately experimentation, or incorporation into practice.[14]
There are several limitations to our evaluation including that we focused solely on academic hospitalist programs, and our sample size for faculty and residents was small. Also, we only addressed a small, though representative, sample of unprofessional behaviors and have not yet linked intervention to actual behavior change. Finally, the script scenarios that we used in this study were not previously published as they were created specifically for this intervention. Validity evidence for these scenarios include that they were based upon the results of earlier work from our institutions and underwent thorough peer review for content and clarity. Further studies will be required to do this. However, we do believe that these are positive findings for utilizing this type of interactive curriculum for professionalism education to promote self‐reflection and behavior change.
Video‐based professionalism education is a feasible, interactive mechanism to encourage self‐reflection and intent to change behavior among faculty and resident physicians. Future study is underway to conduct longitudinal assessments of the learning environments at the participating institutions to assess culture change, perceptions of behaviors, and sustainability of this type of intervention.
Disclosures: The authors acknowledge funding from the American Board of Internal Medicine. The funders had no role in the design of the study; the collection, analysis, and interpretation of the data; or the decision to approve publication of the finished manuscript. Results from this work have been presented at the Midwest Society of General Internal Medicine Regional Meeting, Chicago, Illinois, September 2011; Midwest Society of Hospital Medicine Regional Meeting, Chicago, Illinois, October 2011, and Society of Hospital Medicine Annual Meeting, San Diego, California, April 2012. The authors declare that they do not have any conflicts of interest to disclose.
- Liaison Committee on Medical Education. Functions and structure of a medical school. Available at: http://www.lcme.org/functions.pdf. Accessed October 10, 2012.
- , , , , . Residents' perceptions of their own professionalism and the professionalism of their learning environment. J Grad Med Educ. 2009;1:208–215.
- Society of Hospital Medicine. The core competencies in hospital medicine. http://www.hospitalmedicine.org/Content/NavigationMenu/Education/CoreCurriculum/Core_Competencies.htm. Accessed October 10, 2012.
- The Joint Commission. Behaviors that undermine a culture of safety. Sentinel Event Alert. 2008;(40):1–3. http://www.jointcommission.org/assets/1/18/SEA_40.pdf. Accessed October 10, 2012.
- , . A survey of the impact of disruptive behaviors and communication defects on patient safety. Jt Comm J Qual Patient Saf. 2008;34:464–471.
- , , , et al. Participation in unprofessional behaviors among hospitalists: a multicenter study. J Hosp Med. 2012;7(7):543–550.
- , , et al. Participation in and perceptions of unprofessional behaviors among incoming internal medicine interns. JAMA. 2008;300:1132–1134.
- , , , et al., Changes in perception of and participation in unprofessional behaviors during internship. Acad Med. 2010;85:S76–S80.
- , , , et al. Perspective: beyond counting hours: the importance of supervision, professionalism, transitions of care, and workload in residency training. Acad Med. 2012;87(7):883–888.
- , . The role of the student‐teacher relationship in the formation of physicians: the hidden curriculum as process. J Gen Intern Med. 2006;21:S16–S20.
- , , , et al. Evidence for validity of a survey to measure the learning environment for professionalism. Med Teach. 2011;33(12):e683–e688.
- . Experiential Learning: Experience as the Source of Learning and Development. Englewood Cliffs, NJ: Prentice Hall; 1984.
- , . How can physicians' learning style drive educational planning? Acad Med. 2005;80:680–84.
- , . Twenty years of experience using trigger films as a teaching tool. Acad Med. 2001;76:656–658.
- Liaison Committee on Medical Education. Functions and structure of a medical school. Available at: http://www.lcme.org/functions.pdf. Accessed October 10, 2012.
- , , , , . Residents' perceptions of their own professionalism and the professionalism of their learning environment. J Grad Med Educ. 2009;1:208–215.
- Society of Hospital Medicine. The core competencies in hospital medicine. http://www.hospitalmedicine.org/Content/NavigationMenu/Education/CoreCurriculum/Core_Competencies.htm. Accessed October 10, 2012.
- The Joint Commission. Behaviors that undermine a culture of safety. Sentinel Event Alert. 2008;(40):1–3. http://www.jointcommission.org/assets/1/18/SEA_40.pdf. Accessed October 10, 2012.
- , . A survey of the impact of disruptive behaviors and communication defects on patient safety. Jt Comm J Qual Patient Saf. 2008;34:464–471.
- , , , et al. Participation in unprofessional behaviors among hospitalists: a multicenter study. J Hosp Med. 2012;7(7):543–550.
- , , et al. Participation in and perceptions of unprofessional behaviors among incoming internal medicine interns. JAMA. 2008;300:1132–1134.
- , , , et al., Changes in perception of and participation in unprofessional behaviors during internship. Acad Med. 2010;85:S76–S80.
- , , , et al. Perspective: beyond counting hours: the importance of supervision, professionalism, transitions of care, and workload in residency training. Acad Med. 2012;87(7):883–888.
- , . The role of the student‐teacher relationship in the formation of physicians: the hidden curriculum as process. J Gen Intern Med. 2006;21:S16–S20.
- , , , et al. Evidence for validity of a survey to measure the learning environment for professionalism. Med Teach. 2011;33(12):e683–e688.
- . Experiential Learning: Experience as the Source of Learning and Development. Englewood Cliffs, NJ: Prentice Hall; 1984.
- , . How can physicians' learning style drive educational planning? Acad Med. 2005;80:680–84.
- , . Twenty years of experience using trigger films as a teaching tool. Acad Med. 2001;76:656–658.
AMDEs in Children
Children with complex chronic conditions comprise an increasing proportion of hospital admissions, readmissions, and resource use.[1, 2, 3] Dependence on technology or medical devices is a frequent characteristic of children in this group.[4] Adverse medical device events (AMDEs) are estimated to occur in as many as 8% of all adult admissions, depending on the methods used to identify them.[5] These events may result in hospitalizations or complicate hospital stays. To date, however, the burden of AMDEs among hospitalized children is little described, even though children may be at increased risk for device events as compared to adults.[6] Although some medical devices are intended solely or primarily for use with children, most devices used with children have been initially developed for, tested with, and most frequently employed to treat adults.[6] Assessing the continued safety and effectiveness of medical devices marketed in the Unites States is the responsibility of the Center for Devices and Radiologic Health of the US Food and Drug Administration (FDA). Its existing mechanisms for postmarket device surveillance rely primarily on passive reporting systems and specific observational studies.[7]
The objective of this study was to utilize administrative data from children's hospitals to explore the prevalence and nature of AMDEs in tertiary care children's hospitals that treat significant numbers of children with complex needs requiring medical devices.
METHODS
Data were obtained from the Pediatric Health Information System (PHIS), an administrative database containing inpatient data from 44 not‐for‐profit, tertiary care, pediatric hospitals affiliated with the Children's Hospital Association. Data are deidentified at the time of submission, and are subjected to a number of reliability and validity checks.[8] Individual admission records have both a deidentified visit identification (ID) and patient ID, allowing for linkage of multiple admissions by the same patient.
AMDEs were defined by International Classification of Diseases, Ninth Revision (ICD‐9) codes, using a methodology developed by Samore et al., who identified a set of such codes that specified devices in their definitions and therefore were considered to have a high likelihood of indicating a device problem (see Supporting Information, Table S1, in the online version of this article).[5] The diagnosis codes were grouped into device categories (eg, nervous system, orthopedic, cardiac).
From the 44 hospitals, the primary study cohort consisted of any patient with an admission between January 1, 2004 and December 31, 2011 with 1 AMDE ICD‐9 code as a primary or secondary diagnosis.
Descriptive statistics for patient demographics and visit characteristics of AMDE admissions were generated and stratified by device category. We reported these as counts and percentages for categorical variables and as median and interquartile range for length of stay. We also reported on how frequently patients with AMDEs have a top 10 most common diagnosis and top 10 most common procedure during the AMDE admission. We also reported the presence or absence of a complex chronic condition.[9] We generated the list of most common principal diagnoses and procedures by a separate query of PHIS from 2004 to 2009. Our top 10 most common diagnoses included ICD‐9 codes 486 (pneumonia), 466.11 (acute bronchiolitis due to respiratory syncytial virus), V58.11 (chemotherapy encounter), 493.92 (asthma exacerbation), 493.91 (asthma with status asthmaticus), 466.19 (acute bronchiolitis due to other organism), 780.39 (other convulsions), 540.9 (acute appendicitis), 282.62 (sickle cell disease with crisis), and 276.51 (dehydration). Our top 10 most common procedures included ICD‐9 codes 38.93 (venous catheterization), 03.31 (spinal tap), 99.04 (packed blood cell transfusion), 99.15 (parenteral nutrition), 99.25 (cancer chemotherapy), 96.71 (invasive mechanical ventilation, 96 hours), 96.04 (endotracheal intubation), 96.72 (invasive mechanical ventilation,95 hours), 96.6 (enteral nutrition), and 99.05 (platelet transfusion). Analyses were performed using SAS Enterprise Guide version 4.2 for Windows (SAS Institute, Cary, NC).
This study was approved by Cincinnati Children's Hospital Medical Center Institutional Review Board.
RESULTS
Of the 4,115,755 admissions during the study period, 136,465 (3.3%) had at least 1 AMDE. Over our study period, AMDEs were associated with a mean 17,058 inpatient stays annually. The number of AMDE‐related admissions decreased the last 4 years of our study period despite generally increasing admissions at PHIS hospitals (Figure 1). For 55% of the admissions (75,206/136,465), this AMDE code represented the primary diagnosis. Of these visits with a primary AMDE diagnosis, 39,874 (53%) were related to nervous system devices. The visits associated with AMDEs were comprised of 88,908 unique patients, 55% of whom were male (Table 1). The median age on admission was 6 years, and the interquartile range was 1 to 14 years of age.
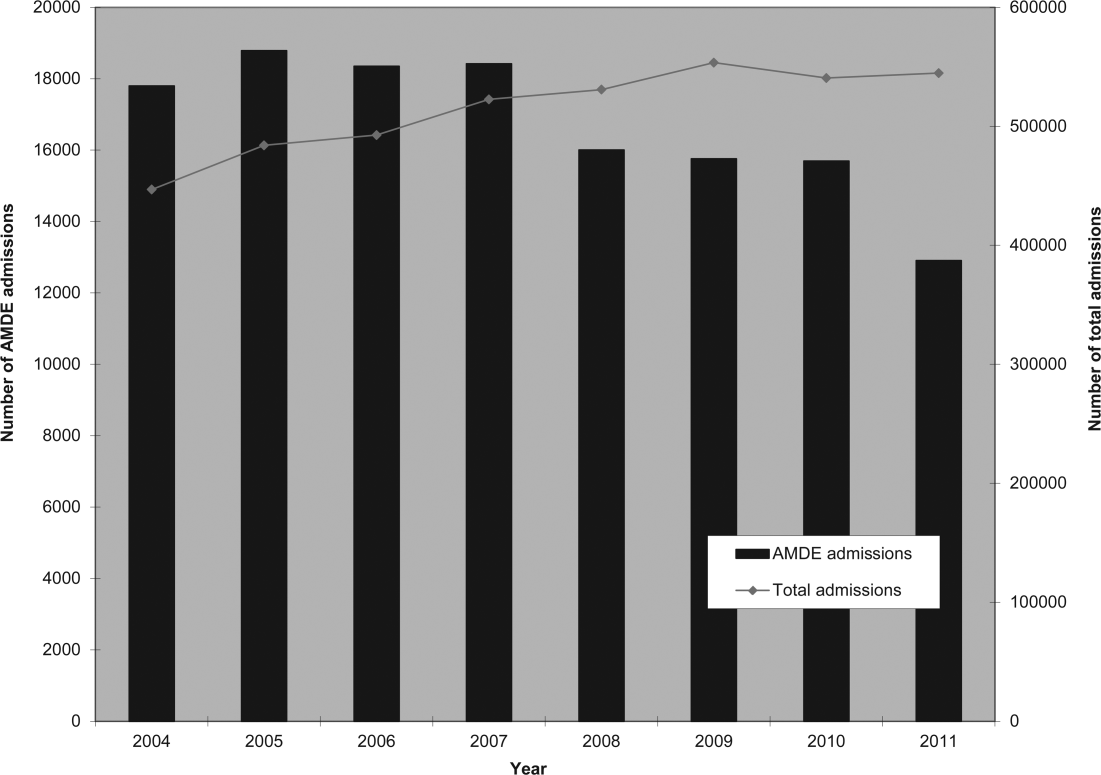
| Total, N=88,908 | |
|---|---|
| Age at first admission | |
| 2 years | 35,160 (40.0%) |
| 35 years | 9,352 (10.5%) |
| 611 years | 16,148 (18.2%) |
| 1218 years | 22,483 (25.3%) |
| 19 years | 5,765 (6.5%) |
| Gender | |
| Male | 49,172 (55.3%) |
| Female | 39,730 (44.7%) |
| Race | |
| White | 59,842 (67.3%) |
| Black | 14,747 (16.6%) |
| Asian | 1,910 (2.2%) |
| American Indian | 900 (1.0%) |
| Other | 8,732 (9.8%) |
| Missing | 2,777 (3.1%) |
| Number of admissions by patient | |
| 1 | 66,814 (75.1%) |
| 2 | 12,520 (14.1%) |
| 3 | 4,504 (5.1%) |
| 4 | 5,071 (5.7%) |
Among admissions with AMDEs, 2.9% ended in death. The mortality was 0.5% when an AMDE was the primary diagnosis and 5.7% when the AMDE was a secondary diagnosis. The median length of inpatient stays was 6 days, with an interquartile range of 2 to 17 days.
Vascular access AMDEs were the most common event associated with admissions (26.6%), followed by nervous system devices (17.8%) (Table 2). The majority (75.5%) of patients admitted with AMDEs had a complex chronic condition. Less than half (46.8%) of AMDE admissions had an associated code for 1 of the 10 most common principal procedures. A minority (14.3%) of admissions had an associated ICD‐9 code for 1 of the top 10 most common principal diagnoses.
| Device Category | Admissions, n=136,465 | Presence of Top 10 Most Common Principal Procedures, n=63,801 | Presence of Top 10 Most Common Principal Diagnoses, n=19,472 | Presence of 1 Complex Chronic Condition, n=103,003 |
|---|---|---|---|---|
| ||||
| Only 1 AMDE diagnosis | ||||
| Vascular access | 36,257 (26.6%) | 26,658 (41.8%) | 6,518 (33.5%) | 26,022 (25.3%) |
| Nervous system | 24,243 (17.8%) | 4,266 (6.7%) | 3,567 (18.3%) | 21,516 (20.9%) |
| Unspecified device | 21,222 (15.6%) | 11,368 (17.8%) | 2,512 (12.9%) | 13,826 (13.4%) |
| Cardiac | 4,384 (3.2%) | 1,959 (3.1%) | 309 (1.6%) | 3,962 (3.8%) |
| Orthopedic | 3,064 (2.2%) | 874 (1.4%) | 179 (0.9%) | 1,235 (1.2%) |
| Dialysis | 2,426 (1.8%) | 836 (1.3%) | 281 (1.4%) | 1,462 (1.4%) |
| Genitourinary | 1,165 (0.9%) | 388 (0.6%) | 166 (0.9%) | 668 (0.6%) |
| Prosthetic cardiac valve | 518 (0.4%) | 236 (0.4%) | 33 (0.2%) | 411 (0.4%) |
| Urologic catheters | 379 (0.3%) | 228 (0.4%) | 93 (0.5%) | 223 (0.2%) |
| Defibrillator | 197 (0.1%) | 11 (0.02%) | 4 (0.02%) | 18 (0.02%) |
| Ocular | 3 (0.002%) | 1 (0.002%) | 1 (0.005%) | 1 (0.001%) |
| Only 1 AMDE diagnosis subtotal | 93,861 (68.8%) | 46,825 (73.4%) | 13,663 (70.2%) | 69,344 (67.3%) |
| 2 AMDE diagnoses | 39,557 (29.0%) | 15,003 (23.5%) | 5,312 (27.3%) | 31,091 (30.2%) |
| >2 AMDE diagnoses | 3,047 (2.2%) | 1,973 (3.1%) | 497 (2.6%) | 2,568 (2.5%) |
DISCUSSION
To our knowledge, our study is the first to report the burden of AMDEs among children requiring hospitalization. AMDEs are common in this population of children cared for at tertiary care children's hospitals, accounting for or complicating 3.3% of inpatient stays in these 44 hospitals. AMDEs were associated with a mean of >17,000 total visits per year. Vascular access devices and nervous system devices were the most common device categories linked to AMDEs. Similar to published literature, we found that the youngest children accounted for the highest proportion of AMDEs.[10, 11]
The majority (>75%) of children with an AMDE admission had diagnoses indicating complex chronic conditions during the admission. Over a partially overlapping study period, Feudtner and colleagues found 25.2% of patients admitted to PHIS hospitals had complex chronic conditions.[12] This finding, combined with the uncommon association of the most prevalent diagnoses and procedures, suggests that the burden of AMDEs falls disproportionately on this population of children. Death occurred considerably less commonly when AMDE diagnosis was the primary versus a secondary diagnosis (0.5% vs 5.7%). This finding likely illustrates 2 distinct populations: children with an AMDE that causes admission who have a relatively low risk of mortality and a second group who have AMDE‐complicated hospitalizations that may have an already high risk of mortality.
Our findings complement those of Wang and colleagues who employed the National Electronic Injury Surveillance System All Injury Program database to provide national estimates of medical device‐associated adverse events.[11] Importantly, this group used a different population (patients presenting to the emergency department) and a different methodology. These authors reported on device‐associated events, as they did not collect information to discriminate the device's role in the event. A walker that malfunctioned leading to patient injury would be a device‐related event; however, a patient who has a walker suffering a fall would be device‐associated, even if the walker's role in the injury was uncertain. We believe our methodology, established by Samore et al., more accurately identifies device‐related events.[5] Wang et al. found that 6.3% of pediatric patients who presented to emergency departments with medical device‐associated events were admitted to the hospital.[11] This resulted in national estimates of 9,082 events with 95% confidence intervals of 2,990 to 25,373 hospitalizations. Our findings of >17,000 AMDE‐related inpatient stays per annum included not only AMDEs leading to admissions but also those that were complications during stays.
Our study has several limitations, most related to the possibility of misclassification present in administrative data. Our approach only captured events that led to or complicated admissions. We suspect that ICD‐9 codes likely missed some AMDEs and that our estimates may therefore under‐represent this problem in our population. Future studies should compare our methodology, which has produced the first across‐center estimates of AMDE admissions, to alternative event capture techniques. We were unable to determine which events were present on admission and which complicated hospital stays, and it is likely that differing interventions would be required to reduce these 2 types of AMDEs. Another important limitation is that the PHIS database, comprised of data on children receiving care at tertiary academic medical centers with large numbers of pediatric subspecialists, is not representative of the population of children overall. The individual ICD‐9 codes for AMDEs are sufficiently nonspecific to limit the ability to characterize device events from administrative data alone. The high prevalence of unspecified device‐related admissions is an additional limitation. Although the estimates of these types of AMDEs are important in describing the frequency of these events, the unspecified category limits the ability to fully stratify based on device type and then implement monitoring strategies and interventions based on each.
To our knowledge, this study is the first multicenter analysis of the spectrum of pediatric AMDEs in hospitalized children. The AMDE prevalence is substantial, and the burden of these events largely falls on children with complex chronic conditions. Despite its limitations, this study complements recent efforts to enhance postmarket surveillance of pediatric devices including that of the FDA's Office of Pediatric Therapeutics, the recent FDA report Strengthening Our National System for Medical Device Postmarket Surveillance (
Our description of AMDEs by device category and patient characteristics is a first and necessary step to understanding the public health burden associated with device use in the pediatric population. Further developments in refined coding and device designation (eg, UDI systems) are needed to refine these estimates.
Acknowledgments
The authors thank Amy Liu, with the Data Management Center, and Colleen Mangeot, with the Biostatistical Consulting Unit in the Division of Epidemiology and Biostatistics, for their assistance with the data pull and creation of the analytic dataset. The authors also thank Lilliam Ambroggio, PhD, and Joshua Schaffzin, MD, PhD, for their thoughtful review of draft manuscripts.
Disclosures: Dr. Brady was supported by funds from the Bureau of Health Professions (BHPr), Health Resources and Services Administration (HRSA), and Department of Health and Human Services (DHHS) under grant T32 HP10027. This project was supported by cooperative agreement number U18 HS016957‐03 from the Agency for Healthcare Research and Quality. The content is solely the responsibility of the authors and does not necessarily represent the official views of the Agency for Healthcare Research and Quality. The study sponsors had no role in the study design; the collection, analysis, and interpretation of data; the writing of the report; or the decision to submit the article for publication. The opinions and assertions presented herein are the private views of the authors and are not to be construed as conveying either an official endorsement or criticism by the US Department of Health and Human Services, The Public Health Service, or the US Food and Drug Administration.
- , , , et al. Hospital utilization and characteristics of patients experiencing recurrent readmissions within children's hospitals. JAMA. 2011;305(7):682–690.
- , , , et al. Children with complex chronic conditions in inpatient hospital settings in the United States. Pediatrics. 2010;126(4):647–655.
- , , , , , . Increasing prevalence of medically complex children in US hospitals. Pediatrics. 2010;126(4):638–646.
- , , , et al. Children with medical complexity: an emerging population for clinical and research initiatives. Pediatrics. 2011;127(3):529–538.
- , , , et al. Surveillance of medical device‐related hazards and adverse events in hospitalized patients. JAMA. 2004;291(3):325–334.
- Institute of Medicine (U.S.). Committee on Postmarket Surveillance of Pediatric Medical Devices. , . Safe medical devices for children. Washington, DC: National Academies Press; 2006.
- , . Pharmacovigilance. 2nd ed. Chichester, England ; Hoboken, NJ: John Wiley 2007.
- , , , . Corticosteroids and mortality in children with bacterial meningitis. JAMA. 2008;299(17):2048–2055.
- , , . Pediatric deaths attributable to complex chronic conditions: a population‐based study of Washington State, 1980–1997. Pediatrics. 2000;106(1 pt 2):205–209.
- , , , et al. Infection rates following initial cerebrospinal fluid shunt placement across pediatric hospitals in the United States. Clinical article. J Neurosurg Pediatr. 2009;4(2):156–165.
- , , , et al. Emergency department visits for medical device‐associated adverse events among children. Pediatrics. 2010;126(2):247–259.
- , , , et al. How well can hospital readmission be predicted in a cohort of hospitalized children? A retrospective, multicenter study. Pediatrics. 2009;123(1):286–293.
- , . Unique device identification in the service of public health. N Engl J Med. 2012;367(17):1583–1585.
Children with complex chronic conditions comprise an increasing proportion of hospital admissions, readmissions, and resource use.[1, 2, 3] Dependence on technology or medical devices is a frequent characteristic of children in this group.[4] Adverse medical device events (AMDEs) are estimated to occur in as many as 8% of all adult admissions, depending on the methods used to identify them.[5] These events may result in hospitalizations or complicate hospital stays. To date, however, the burden of AMDEs among hospitalized children is little described, even though children may be at increased risk for device events as compared to adults.[6] Although some medical devices are intended solely or primarily for use with children, most devices used with children have been initially developed for, tested with, and most frequently employed to treat adults.[6] Assessing the continued safety and effectiveness of medical devices marketed in the Unites States is the responsibility of the Center for Devices and Radiologic Health of the US Food and Drug Administration (FDA). Its existing mechanisms for postmarket device surveillance rely primarily on passive reporting systems and specific observational studies.[7]
The objective of this study was to utilize administrative data from children's hospitals to explore the prevalence and nature of AMDEs in tertiary care children's hospitals that treat significant numbers of children with complex needs requiring medical devices.
METHODS
Data were obtained from the Pediatric Health Information System (PHIS), an administrative database containing inpatient data from 44 not‐for‐profit, tertiary care, pediatric hospitals affiliated with the Children's Hospital Association. Data are deidentified at the time of submission, and are subjected to a number of reliability and validity checks.[8] Individual admission records have both a deidentified visit identification (ID) and patient ID, allowing for linkage of multiple admissions by the same patient.
AMDEs were defined by International Classification of Diseases, Ninth Revision (ICD‐9) codes, using a methodology developed by Samore et al., who identified a set of such codes that specified devices in their definitions and therefore were considered to have a high likelihood of indicating a device problem (see Supporting Information, Table S1, in the online version of this article).[5] The diagnosis codes were grouped into device categories (eg, nervous system, orthopedic, cardiac).
From the 44 hospitals, the primary study cohort consisted of any patient with an admission between January 1, 2004 and December 31, 2011 with 1 AMDE ICD‐9 code as a primary or secondary diagnosis.
Descriptive statistics for patient demographics and visit characteristics of AMDE admissions were generated and stratified by device category. We reported these as counts and percentages for categorical variables and as median and interquartile range for length of stay. We also reported on how frequently patients with AMDEs have a top 10 most common diagnosis and top 10 most common procedure during the AMDE admission. We also reported the presence or absence of a complex chronic condition.[9] We generated the list of most common principal diagnoses and procedures by a separate query of PHIS from 2004 to 2009. Our top 10 most common diagnoses included ICD‐9 codes 486 (pneumonia), 466.11 (acute bronchiolitis due to respiratory syncytial virus), V58.11 (chemotherapy encounter), 493.92 (asthma exacerbation), 493.91 (asthma with status asthmaticus), 466.19 (acute bronchiolitis due to other organism), 780.39 (other convulsions), 540.9 (acute appendicitis), 282.62 (sickle cell disease with crisis), and 276.51 (dehydration). Our top 10 most common procedures included ICD‐9 codes 38.93 (venous catheterization), 03.31 (spinal tap), 99.04 (packed blood cell transfusion), 99.15 (parenteral nutrition), 99.25 (cancer chemotherapy), 96.71 (invasive mechanical ventilation, 96 hours), 96.04 (endotracheal intubation), 96.72 (invasive mechanical ventilation,95 hours), 96.6 (enteral nutrition), and 99.05 (platelet transfusion). Analyses were performed using SAS Enterprise Guide version 4.2 for Windows (SAS Institute, Cary, NC).
This study was approved by Cincinnati Children's Hospital Medical Center Institutional Review Board.
RESULTS
Of the 4,115,755 admissions during the study period, 136,465 (3.3%) had at least 1 AMDE. Over our study period, AMDEs were associated with a mean 17,058 inpatient stays annually. The number of AMDE‐related admissions decreased the last 4 years of our study period despite generally increasing admissions at PHIS hospitals (Figure 1). For 55% of the admissions (75,206/136,465), this AMDE code represented the primary diagnosis. Of these visits with a primary AMDE diagnosis, 39,874 (53%) were related to nervous system devices. The visits associated with AMDEs were comprised of 88,908 unique patients, 55% of whom were male (Table 1). The median age on admission was 6 years, and the interquartile range was 1 to 14 years of age.

| Total, N=88,908 | |
|---|---|
| Age at first admission | |
| 2 years | 35,160 (40.0%) |
| 35 years | 9,352 (10.5%) |
| 611 years | 16,148 (18.2%) |
| 1218 years | 22,483 (25.3%) |
| 19 years | 5,765 (6.5%) |
| Gender | |
| Male | 49,172 (55.3%) |
| Female | 39,730 (44.7%) |
| Race | |
| White | 59,842 (67.3%) |
| Black | 14,747 (16.6%) |
| Asian | 1,910 (2.2%) |
| American Indian | 900 (1.0%) |
| Other | 8,732 (9.8%) |
| Missing | 2,777 (3.1%) |
| Number of admissions by patient | |
| 1 | 66,814 (75.1%) |
| 2 | 12,520 (14.1%) |
| 3 | 4,504 (5.1%) |
| 4 | 5,071 (5.7%) |
Among admissions with AMDEs, 2.9% ended in death. The mortality was 0.5% when an AMDE was the primary diagnosis and 5.7% when the AMDE was a secondary diagnosis. The median length of inpatient stays was 6 days, with an interquartile range of 2 to 17 days.
Vascular access AMDEs were the most common event associated with admissions (26.6%), followed by nervous system devices (17.8%) (Table 2). The majority (75.5%) of patients admitted with AMDEs had a complex chronic condition. Less than half (46.8%) of AMDE admissions had an associated code for 1 of the 10 most common principal procedures. A minority (14.3%) of admissions had an associated ICD‐9 code for 1 of the top 10 most common principal diagnoses.
| Device Category | Admissions, n=136,465 | Presence of Top 10 Most Common Principal Procedures, n=63,801 | Presence of Top 10 Most Common Principal Diagnoses, n=19,472 | Presence of 1 Complex Chronic Condition, n=103,003 |
|---|---|---|---|---|
| ||||
| Only 1 AMDE diagnosis | ||||
| Vascular access | 36,257 (26.6%) | 26,658 (41.8%) | 6,518 (33.5%) | 26,022 (25.3%) |
| Nervous system | 24,243 (17.8%) | 4,266 (6.7%) | 3,567 (18.3%) | 21,516 (20.9%) |
| Unspecified device | 21,222 (15.6%) | 11,368 (17.8%) | 2,512 (12.9%) | 13,826 (13.4%) |
| Cardiac | 4,384 (3.2%) | 1,959 (3.1%) | 309 (1.6%) | 3,962 (3.8%) |
| Orthopedic | 3,064 (2.2%) | 874 (1.4%) | 179 (0.9%) | 1,235 (1.2%) |
| Dialysis | 2,426 (1.8%) | 836 (1.3%) | 281 (1.4%) | 1,462 (1.4%) |
| Genitourinary | 1,165 (0.9%) | 388 (0.6%) | 166 (0.9%) | 668 (0.6%) |
| Prosthetic cardiac valve | 518 (0.4%) | 236 (0.4%) | 33 (0.2%) | 411 (0.4%) |
| Urologic catheters | 379 (0.3%) | 228 (0.4%) | 93 (0.5%) | 223 (0.2%) |
| Defibrillator | 197 (0.1%) | 11 (0.02%) | 4 (0.02%) | 18 (0.02%) |
| Ocular | 3 (0.002%) | 1 (0.002%) | 1 (0.005%) | 1 (0.001%) |
| Only 1 AMDE diagnosis subtotal | 93,861 (68.8%) | 46,825 (73.4%) | 13,663 (70.2%) | 69,344 (67.3%) |
| 2 AMDE diagnoses | 39,557 (29.0%) | 15,003 (23.5%) | 5,312 (27.3%) | 31,091 (30.2%) |
| >2 AMDE diagnoses | 3,047 (2.2%) | 1,973 (3.1%) | 497 (2.6%) | 2,568 (2.5%) |
DISCUSSION
To our knowledge, our study is the first to report the burden of AMDEs among children requiring hospitalization. AMDEs are common in this population of children cared for at tertiary care children's hospitals, accounting for or complicating 3.3% of inpatient stays in these 44 hospitals. AMDEs were associated with a mean of >17,000 total visits per year. Vascular access devices and nervous system devices were the most common device categories linked to AMDEs. Similar to published literature, we found that the youngest children accounted for the highest proportion of AMDEs.[10, 11]
The majority (>75%) of children with an AMDE admission had diagnoses indicating complex chronic conditions during the admission. Over a partially overlapping study period, Feudtner and colleagues found 25.2% of patients admitted to PHIS hospitals had complex chronic conditions.[12] This finding, combined with the uncommon association of the most prevalent diagnoses and procedures, suggests that the burden of AMDEs falls disproportionately on this population of children. Death occurred considerably less commonly when AMDE diagnosis was the primary versus a secondary diagnosis (0.5% vs 5.7%). This finding likely illustrates 2 distinct populations: children with an AMDE that causes admission who have a relatively low risk of mortality and a second group who have AMDE‐complicated hospitalizations that may have an already high risk of mortality.
Our findings complement those of Wang and colleagues who employed the National Electronic Injury Surveillance System All Injury Program database to provide national estimates of medical device‐associated adverse events.[11] Importantly, this group used a different population (patients presenting to the emergency department) and a different methodology. These authors reported on device‐associated events, as they did not collect information to discriminate the device's role in the event. A walker that malfunctioned leading to patient injury would be a device‐related event; however, a patient who has a walker suffering a fall would be device‐associated, even if the walker's role in the injury was uncertain. We believe our methodology, established by Samore et al., more accurately identifies device‐related events.[5] Wang et al. found that 6.3% of pediatric patients who presented to emergency departments with medical device‐associated events were admitted to the hospital.[11] This resulted in national estimates of 9,082 events with 95% confidence intervals of 2,990 to 25,373 hospitalizations. Our findings of >17,000 AMDE‐related inpatient stays per annum included not only AMDEs leading to admissions but also those that were complications during stays.
Our study has several limitations, most related to the possibility of misclassification present in administrative data. Our approach only captured events that led to or complicated admissions. We suspect that ICD‐9 codes likely missed some AMDEs and that our estimates may therefore under‐represent this problem in our population. Future studies should compare our methodology, which has produced the first across‐center estimates of AMDE admissions, to alternative event capture techniques. We were unable to determine which events were present on admission and which complicated hospital stays, and it is likely that differing interventions would be required to reduce these 2 types of AMDEs. Another important limitation is that the PHIS database, comprised of data on children receiving care at tertiary academic medical centers with large numbers of pediatric subspecialists, is not representative of the population of children overall. The individual ICD‐9 codes for AMDEs are sufficiently nonspecific to limit the ability to characterize device events from administrative data alone. The high prevalence of unspecified device‐related admissions is an additional limitation. Although the estimates of these types of AMDEs are important in describing the frequency of these events, the unspecified category limits the ability to fully stratify based on device type and then implement monitoring strategies and interventions based on each.
To our knowledge, this study is the first multicenter analysis of the spectrum of pediatric AMDEs in hospitalized children. The AMDE prevalence is substantial, and the burden of these events largely falls on children with complex chronic conditions. Despite its limitations, this study complements recent efforts to enhance postmarket surveillance of pediatric devices including that of the FDA's Office of Pediatric Therapeutics, the recent FDA report Strengthening Our National System for Medical Device Postmarket Surveillance (
Our description of AMDEs by device category and patient characteristics is a first and necessary step to understanding the public health burden associated with device use in the pediatric population. Further developments in refined coding and device designation (eg, UDI systems) are needed to refine these estimates.
Acknowledgments
The authors thank Amy Liu, with the Data Management Center, and Colleen Mangeot, with the Biostatistical Consulting Unit in the Division of Epidemiology and Biostatistics, for their assistance with the data pull and creation of the analytic dataset. The authors also thank Lilliam Ambroggio, PhD, and Joshua Schaffzin, MD, PhD, for their thoughtful review of draft manuscripts.
Disclosures: Dr. Brady was supported by funds from the Bureau of Health Professions (BHPr), Health Resources and Services Administration (HRSA), and Department of Health and Human Services (DHHS) under grant T32 HP10027. This project was supported by cooperative agreement number U18 HS016957‐03 from the Agency for Healthcare Research and Quality. The content is solely the responsibility of the authors and does not necessarily represent the official views of the Agency for Healthcare Research and Quality. The study sponsors had no role in the study design; the collection, analysis, and interpretation of data; the writing of the report; or the decision to submit the article for publication. The opinions and assertions presented herein are the private views of the authors and are not to be construed as conveying either an official endorsement or criticism by the US Department of Health and Human Services, The Public Health Service, or the US Food and Drug Administration.
Children with complex chronic conditions comprise an increasing proportion of hospital admissions, readmissions, and resource use.[1, 2, 3] Dependence on technology or medical devices is a frequent characteristic of children in this group.[4] Adverse medical device events (AMDEs) are estimated to occur in as many as 8% of all adult admissions, depending on the methods used to identify them.[5] These events may result in hospitalizations or complicate hospital stays. To date, however, the burden of AMDEs among hospitalized children is little described, even though children may be at increased risk for device events as compared to adults.[6] Although some medical devices are intended solely or primarily for use with children, most devices used with children have been initially developed for, tested with, and most frequently employed to treat adults.[6] Assessing the continued safety and effectiveness of medical devices marketed in the Unites States is the responsibility of the Center for Devices and Radiologic Health of the US Food and Drug Administration (FDA). Its existing mechanisms for postmarket device surveillance rely primarily on passive reporting systems and specific observational studies.[7]
The objective of this study was to utilize administrative data from children's hospitals to explore the prevalence and nature of AMDEs in tertiary care children's hospitals that treat significant numbers of children with complex needs requiring medical devices.
METHODS
Data were obtained from the Pediatric Health Information System (PHIS), an administrative database containing inpatient data from 44 not‐for‐profit, tertiary care, pediatric hospitals affiliated with the Children's Hospital Association. Data are deidentified at the time of submission, and are subjected to a number of reliability and validity checks.[8] Individual admission records have both a deidentified visit identification (ID) and patient ID, allowing for linkage of multiple admissions by the same patient.
AMDEs were defined by International Classification of Diseases, Ninth Revision (ICD‐9) codes, using a methodology developed by Samore et al., who identified a set of such codes that specified devices in their definitions and therefore were considered to have a high likelihood of indicating a device problem (see Supporting Information, Table S1, in the online version of this article).[5] The diagnosis codes were grouped into device categories (eg, nervous system, orthopedic, cardiac).
From the 44 hospitals, the primary study cohort consisted of any patient with an admission between January 1, 2004 and December 31, 2011 with 1 AMDE ICD‐9 code as a primary or secondary diagnosis.
Descriptive statistics for patient demographics and visit characteristics of AMDE admissions were generated and stratified by device category. We reported these as counts and percentages for categorical variables and as median and interquartile range for length of stay. We also reported on how frequently patients with AMDEs have a top 10 most common diagnosis and top 10 most common procedure during the AMDE admission. We also reported the presence or absence of a complex chronic condition.[9] We generated the list of most common principal diagnoses and procedures by a separate query of PHIS from 2004 to 2009. Our top 10 most common diagnoses included ICD‐9 codes 486 (pneumonia), 466.11 (acute bronchiolitis due to respiratory syncytial virus), V58.11 (chemotherapy encounter), 493.92 (asthma exacerbation), 493.91 (asthma with status asthmaticus), 466.19 (acute bronchiolitis due to other organism), 780.39 (other convulsions), 540.9 (acute appendicitis), 282.62 (sickle cell disease with crisis), and 276.51 (dehydration). Our top 10 most common procedures included ICD‐9 codes 38.93 (venous catheterization), 03.31 (spinal tap), 99.04 (packed blood cell transfusion), 99.15 (parenteral nutrition), 99.25 (cancer chemotherapy), 96.71 (invasive mechanical ventilation, 96 hours), 96.04 (endotracheal intubation), 96.72 (invasive mechanical ventilation,95 hours), 96.6 (enteral nutrition), and 99.05 (platelet transfusion). Analyses were performed using SAS Enterprise Guide version 4.2 for Windows (SAS Institute, Cary, NC).
This study was approved by Cincinnati Children's Hospital Medical Center Institutional Review Board.
RESULTS
Of the 4,115,755 admissions during the study period, 136,465 (3.3%) had at least 1 AMDE. Over our study period, AMDEs were associated with a mean 17,058 inpatient stays annually. The number of AMDE‐related admissions decreased the last 4 years of our study period despite generally increasing admissions at PHIS hospitals (Figure 1). For 55% of the admissions (75,206/136,465), this AMDE code represented the primary diagnosis. Of these visits with a primary AMDE diagnosis, 39,874 (53%) were related to nervous system devices. The visits associated with AMDEs were comprised of 88,908 unique patients, 55% of whom were male (Table 1). The median age on admission was 6 years, and the interquartile range was 1 to 14 years of age.

| Total, N=88,908 | |
|---|---|
| Age at first admission | |
| 2 years | 35,160 (40.0%) |
| 35 years | 9,352 (10.5%) |
| 611 years | 16,148 (18.2%) |
| 1218 years | 22,483 (25.3%) |
| 19 years | 5,765 (6.5%) |
| Gender | |
| Male | 49,172 (55.3%) |
| Female | 39,730 (44.7%) |
| Race | |
| White | 59,842 (67.3%) |
| Black | 14,747 (16.6%) |
| Asian | 1,910 (2.2%) |
| American Indian | 900 (1.0%) |
| Other | 8,732 (9.8%) |
| Missing | 2,777 (3.1%) |
| Number of admissions by patient | |
| 1 | 66,814 (75.1%) |
| 2 | 12,520 (14.1%) |
| 3 | 4,504 (5.1%) |
| 4 | 5,071 (5.7%) |
Among admissions with AMDEs, 2.9% ended in death. The mortality was 0.5% when an AMDE was the primary diagnosis and 5.7% when the AMDE was a secondary diagnosis. The median length of inpatient stays was 6 days, with an interquartile range of 2 to 17 days.
Vascular access AMDEs were the most common event associated with admissions (26.6%), followed by nervous system devices (17.8%) (Table 2). The majority (75.5%) of patients admitted with AMDEs had a complex chronic condition. Less than half (46.8%) of AMDE admissions had an associated code for 1 of the 10 most common principal procedures. A minority (14.3%) of admissions had an associated ICD‐9 code for 1 of the top 10 most common principal diagnoses.
| Device Category | Admissions, n=136,465 | Presence of Top 10 Most Common Principal Procedures, n=63,801 | Presence of Top 10 Most Common Principal Diagnoses, n=19,472 | Presence of 1 Complex Chronic Condition, n=103,003 |
|---|---|---|---|---|
| ||||
| Only 1 AMDE diagnosis | ||||
| Vascular access | 36,257 (26.6%) | 26,658 (41.8%) | 6,518 (33.5%) | 26,022 (25.3%) |
| Nervous system | 24,243 (17.8%) | 4,266 (6.7%) | 3,567 (18.3%) | 21,516 (20.9%) |
| Unspecified device | 21,222 (15.6%) | 11,368 (17.8%) | 2,512 (12.9%) | 13,826 (13.4%) |
| Cardiac | 4,384 (3.2%) | 1,959 (3.1%) | 309 (1.6%) | 3,962 (3.8%) |
| Orthopedic | 3,064 (2.2%) | 874 (1.4%) | 179 (0.9%) | 1,235 (1.2%) |
| Dialysis | 2,426 (1.8%) | 836 (1.3%) | 281 (1.4%) | 1,462 (1.4%) |
| Genitourinary | 1,165 (0.9%) | 388 (0.6%) | 166 (0.9%) | 668 (0.6%) |
| Prosthetic cardiac valve | 518 (0.4%) | 236 (0.4%) | 33 (0.2%) | 411 (0.4%) |
| Urologic catheters | 379 (0.3%) | 228 (0.4%) | 93 (0.5%) | 223 (0.2%) |
| Defibrillator | 197 (0.1%) | 11 (0.02%) | 4 (0.02%) | 18 (0.02%) |
| Ocular | 3 (0.002%) | 1 (0.002%) | 1 (0.005%) | 1 (0.001%) |
| Only 1 AMDE diagnosis subtotal | 93,861 (68.8%) | 46,825 (73.4%) | 13,663 (70.2%) | 69,344 (67.3%) |
| 2 AMDE diagnoses | 39,557 (29.0%) | 15,003 (23.5%) | 5,312 (27.3%) | 31,091 (30.2%) |
| >2 AMDE diagnoses | 3,047 (2.2%) | 1,973 (3.1%) | 497 (2.6%) | 2,568 (2.5%) |
DISCUSSION
To our knowledge, our study is the first to report the burden of AMDEs among children requiring hospitalization. AMDEs are common in this population of children cared for at tertiary care children's hospitals, accounting for or complicating 3.3% of inpatient stays in these 44 hospitals. AMDEs were associated with a mean of >17,000 total visits per year. Vascular access devices and nervous system devices were the most common device categories linked to AMDEs. Similar to published literature, we found that the youngest children accounted for the highest proportion of AMDEs.[10, 11]
The majority (>75%) of children with an AMDE admission had diagnoses indicating complex chronic conditions during the admission. Over a partially overlapping study period, Feudtner and colleagues found 25.2% of patients admitted to PHIS hospitals had complex chronic conditions.[12] This finding, combined with the uncommon association of the most prevalent diagnoses and procedures, suggests that the burden of AMDEs falls disproportionately on this population of children. Death occurred considerably less commonly when AMDE diagnosis was the primary versus a secondary diagnosis (0.5% vs 5.7%). This finding likely illustrates 2 distinct populations: children with an AMDE that causes admission who have a relatively low risk of mortality and a second group who have AMDE‐complicated hospitalizations that may have an already high risk of mortality.
Our findings complement those of Wang and colleagues who employed the National Electronic Injury Surveillance System All Injury Program database to provide national estimates of medical device‐associated adverse events.[11] Importantly, this group used a different population (patients presenting to the emergency department) and a different methodology. These authors reported on device‐associated events, as they did not collect information to discriminate the device's role in the event. A walker that malfunctioned leading to patient injury would be a device‐related event; however, a patient who has a walker suffering a fall would be device‐associated, even if the walker's role in the injury was uncertain. We believe our methodology, established by Samore et al., more accurately identifies device‐related events.[5] Wang et al. found that 6.3% of pediatric patients who presented to emergency departments with medical device‐associated events were admitted to the hospital.[11] This resulted in national estimates of 9,082 events with 95% confidence intervals of 2,990 to 25,373 hospitalizations. Our findings of >17,000 AMDE‐related inpatient stays per annum included not only AMDEs leading to admissions but also those that were complications during stays.
Our study has several limitations, most related to the possibility of misclassification present in administrative data. Our approach only captured events that led to or complicated admissions. We suspect that ICD‐9 codes likely missed some AMDEs and that our estimates may therefore under‐represent this problem in our population. Future studies should compare our methodology, which has produced the first across‐center estimates of AMDE admissions, to alternative event capture techniques. We were unable to determine which events were present on admission and which complicated hospital stays, and it is likely that differing interventions would be required to reduce these 2 types of AMDEs. Another important limitation is that the PHIS database, comprised of data on children receiving care at tertiary academic medical centers with large numbers of pediatric subspecialists, is not representative of the population of children overall. The individual ICD‐9 codes for AMDEs are sufficiently nonspecific to limit the ability to characterize device events from administrative data alone. The high prevalence of unspecified device‐related admissions is an additional limitation. Although the estimates of these types of AMDEs are important in describing the frequency of these events, the unspecified category limits the ability to fully stratify based on device type and then implement monitoring strategies and interventions based on each.
To our knowledge, this study is the first multicenter analysis of the spectrum of pediatric AMDEs in hospitalized children. The AMDE prevalence is substantial, and the burden of these events largely falls on children with complex chronic conditions. Despite its limitations, this study complements recent efforts to enhance postmarket surveillance of pediatric devices including that of the FDA's Office of Pediatric Therapeutics, the recent FDA report Strengthening Our National System for Medical Device Postmarket Surveillance (
Our description of AMDEs by device category and patient characteristics is a first and necessary step to understanding the public health burden associated with device use in the pediatric population. Further developments in refined coding and device designation (eg, UDI systems) are needed to refine these estimates.
Acknowledgments
The authors thank Amy Liu, with the Data Management Center, and Colleen Mangeot, with the Biostatistical Consulting Unit in the Division of Epidemiology and Biostatistics, for their assistance with the data pull and creation of the analytic dataset. The authors also thank Lilliam Ambroggio, PhD, and Joshua Schaffzin, MD, PhD, for their thoughtful review of draft manuscripts.
Disclosures: Dr. Brady was supported by funds from the Bureau of Health Professions (BHPr), Health Resources and Services Administration (HRSA), and Department of Health and Human Services (DHHS) under grant T32 HP10027. This project was supported by cooperative agreement number U18 HS016957‐03 from the Agency for Healthcare Research and Quality. The content is solely the responsibility of the authors and does not necessarily represent the official views of the Agency for Healthcare Research and Quality. The study sponsors had no role in the study design; the collection, analysis, and interpretation of data; the writing of the report; or the decision to submit the article for publication. The opinions and assertions presented herein are the private views of the authors and are not to be construed as conveying either an official endorsement or criticism by the US Department of Health and Human Services, The Public Health Service, or the US Food and Drug Administration.
- , , , et al. Hospital utilization and characteristics of patients experiencing recurrent readmissions within children's hospitals. JAMA. 2011;305(7):682–690.
- , , , et al. Children with complex chronic conditions in inpatient hospital settings in the United States. Pediatrics. 2010;126(4):647–655.
- , , , , , . Increasing prevalence of medically complex children in US hospitals. Pediatrics. 2010;126(4):638–646.
- , , , et al. Children with medical complexity: an emerging population for clinical and research initiatives. Pediatrics. 2011;127(3):529–538.
- , , , et al. Surveillance of medical device‐related hazards and adverse events in hospitalized patients. JAMA. 2004;291(3):325–334.
- Institute of Medicine (U.S.). Committee on Postmarket Surveillance of Pediatric Medical Devices. , . Safe medical devices for children. Washington, DC: National Academies Press; 2006.
- , . Pharmacovigilance. 2nd ed. Chichester, England ; Hoboken, NJ: John Wiley 2007.
- , , , . Corticosteroids and mortality in children with bacterial meningitis. JAMA. 2008;299(17):2048–2055.
- , , . Pediatric deaths attributable to complex chronic conditions: a population‐based study of Washington State, 1980–1997. Pediatrics. 2000;106(1 pt 2):205–209.
- , , , et al. Infection rates following initial cerebrospinal fluid shunt placement across pediatric hospitals in the United States. Clinical article. J Neurosurg Pediatr. 2009;4(2):156–165.
- , , , et al. Emergency department visits for medical device‐associated adverse events among children. Pediatrics. 2010;126(2):247–259.
- , , , et al. How well can hospital readmission be predicted in a cohort of hospitalized children? A retrospective, multicenter study. Pediatrics. 2009;123(1):286–293.
- , . Unique device identification in the service of public health. N Engl J Med. 2012;367(17):1583–1585.
- , , , et al. Hospital utilization and characteristics of patients experiencing recurrent readmissions within children's hospitals. JAMA. 2011;305(7):682–690.
- , , , et al. Children with complex chronic conditions in inpatient hospital settings in the United States. Pediatrics. 2010;126(4):647–655.
- , , , , , . Increasing prevalence of medically complex children in US hospitals. Pediatrics. 2010;126(4):638–646.
- , , , et al. Children with medical complexity: an emerging population for clinical and research initiatives. Pediatrics. 2011;127(3):529–538.
- , , , et al. Surveillance of medical device‐related hazards and adverse events in hospitalized patients. JAMA. 2004;291(3):325–334.
- Institute of Medicine (U.S.). Committee on Postmarket Surveillance of Pediatric Medical Devices. , . Safe medical devices for children. Washington, DC: National Academies Press; 2006.
- , . Pharmacovigilance. 2nd ed. Chichester, England ; Hoboken, NJ: John Wiley 2007.
- , , , . Corticosteroids and mortality in children with bacterial meningitis. JAMA. 2008;299(17):2048–2055.
- , , . Pediatric deaths attributable to complex chronic conditions: a population‐based study of Washington State, 1980–1997. Pediatrics. 2000;106(1 pt 2):205–209.
- , , , et al. Infection rates following initial cerebrospinal fluid shunt placement across pediatric hospitals in the United States. Clinical article. J Neurosurg Pediatr. 2009;4(2):156–165.
- , , , et al. Emergency department visits for medical device‐associated adverse events among children. Pediatrics. 2010;126(2):247–259.
- , , , et al. How well can hospital readmission be predicted in a cohort of hospitalized children? A retrospective, multicenter study. Pediatrics. 2009;123(1):286–293.
- , . Unique device identification in the service of public health. N Engl J Med. 2012;367(17):1583–1585.
Impact of Penicillin Skin Testing
Self‐reported penicillin allergy is common and frequently limits the available antimicrobial agents to choose from. This often results in the use of more expensive, potentially more toxic, and possibly less efficacious agents.[1, 2]
For over 30 years, penicilloyl‐polylysine (PPL) penicillin skin testing (PST) was widely used to diagnose penicillin allergy with a negative predictive value (NPV) of about 97% to 99%.[3] After being off the market for 5 years, PPL PST was reapproved in 2009 as PRE‐PEN.[4] However, many clinicians still fail to utilize PST despite its simplicity and substantial clinical impact. The main purpose of this study was to describe the predictive value of PST and impact on antibiotic selection in a sample of hospitalized patients with a reported history of penicillin allergy.
METHODS
In 2010, PST was introduced as a quality‐improvement measure after approval and support from the chief of professional services and the medical staff executive committee at Vidant Medical Center, an 861‐bed tertiary care and teaching hospital. Our antimicrobial stewardship program is regularly contacted for approval of alternative therapies in penicillin allergic patients. The PST quality‐improvement intervention was implemented to avoid resorting to less appropriate therapies in these situations. Following approval by the University and Medical Center Institutional Review Board, we designed a 4‐month study to assess the impact of this ongoing quality improvement measure from March 2012 to July 2012.
Hospitalized patients of all ages with reported penicillin allergies were obtained from our antimicrobial stewardship database. Their charts were reviewed for demographics, antibiotic use, clinical infection, and allergic description. Deciding whether to alter antibiotic therapy to a ‐lactam regimen was based on microbiologic results, laboratory values, clinical infection, and history of immunoglobulin E (IgE)‐mediated reactions, as defined by the updated drug allergy practice parameters.[5] IgE‐mediated reactions included: (1) immediate urticaria, laryngeal edema, or hypotension; (2) anemia; and (3) fever, arthralgias, lymphadenopathy, and an urticarial rash after 7 to 21 days.[5, 6, 7] We defined anaphylaxis as the development of angioedema or hemodynamic instability within 1 hour of penicillin administration. A true negative reaction was a lack of an IgE‐mediated reaction to all the drug challenges.
Patients in the medical, surgical, labor, and delivery wards; intensive care units; and emergency department underwent testing. The ‐lactam agent used after a negative PST was recorded, and the patients were followed for 24 hours after transitioning their therapy to a ‐lactam regimen. Excluded subjects included those with (1) nonIgE‐mediated reactions, (2) skin conditions that can give false positive results, (3) medications that may interfere with anaphylactic therapy, (4) history of severe exfoliative reactions to ‐lactams, (5) anaphylaxis less than 4 weeks prior, (6) allergies to antibiotics other than penicillin, and (7) uncertain allergy history.
PST Reagents/Procedure
Our benzylpenicilloyl major determinant molecule, commercially produced as PPL, was purchased as a PRE‐PEN from ALK‐Abello, Round Rock, Texas. Penicillin G potassium, purchased from Pfizer, New York, New York, is the only commercially available minor determinant and can improve identification of penicillin allergy by up to 97%.[2] The PST panel also included histamine (positive control) and normal saline (negative control).
Skin Testing Procedure
An infectious diseases fellow (R.H.R. or B.K.) was supervised in preparing for potential anaphylaxis, applying the reagents and interpreting the results based on drug allergy practice parameters.[5] The preliminary epicutaneous prick/puncture test was performed with a lancet in subjects without prior anaphylaxis using full‐strength PPL and penicillin G potassium reagents. If there was no response within 15 minutes, which we defined as a lack of wheal formation 3 mm or greater than that of the negative control, 0.02 to 0.03 mL of each reagent was injected intradermally using a tuberculin syringe and examined for 15 minutes.[5] If there was no response, patients were then challenged with either a single oral dose of penicillin V potassium 250 mg or whichever oral penicillin agent they previously reported an allergy to. If no reaction was appreciated within 2 hours, their therapy was changed to a ‐lactam agent including penicillins, cephalosporins, and carbapenems for the remaining duration of therapy (Figure 1) An estimate of NPV was obtained after 24 hours follow‐up.
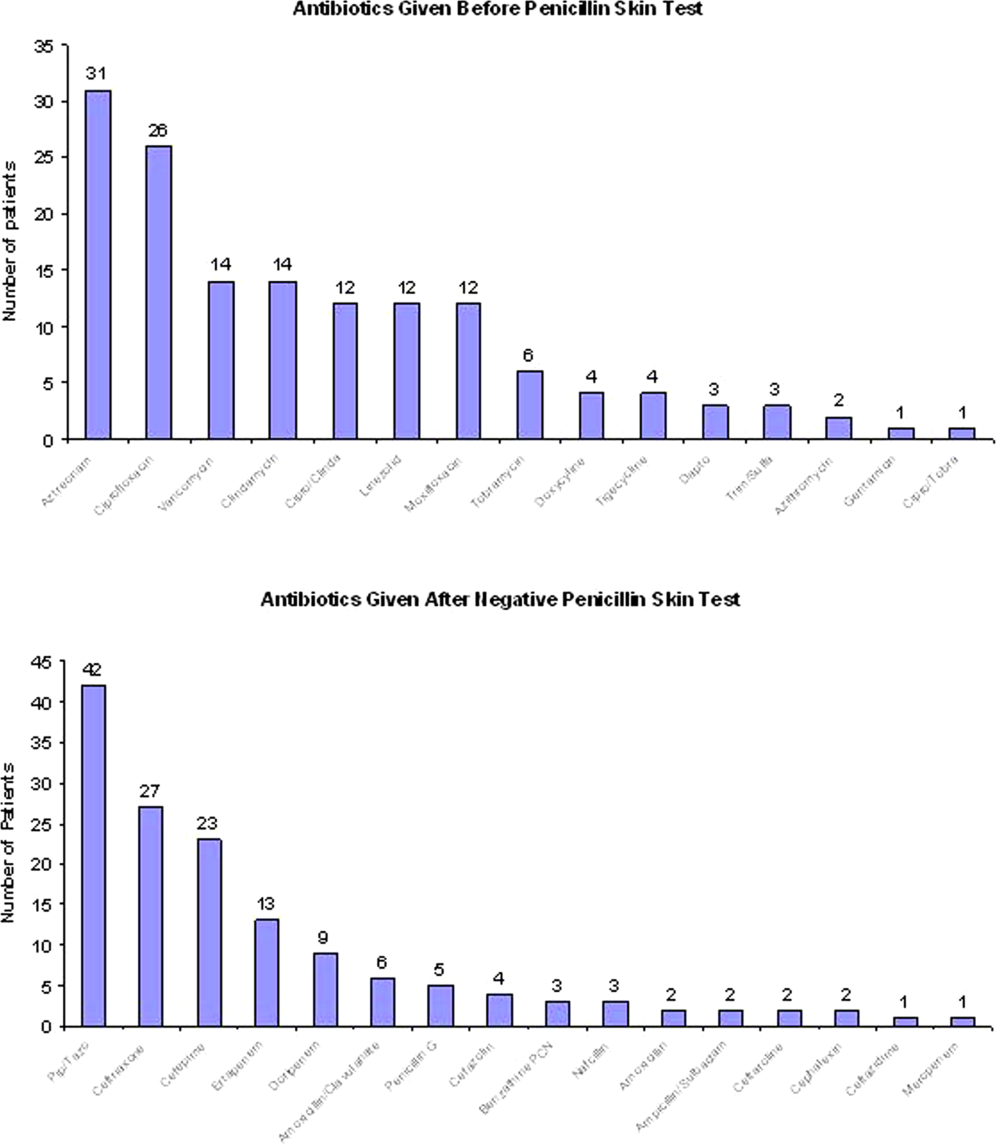
Statistical Analysis
We designed a study to estimate whether the reapproved PST achieves an NPV of at least 95%.[3] We hypothesized that clinicians will be willing to utilize PST even if it has an NPV of slightly less than 98% compared to the current standard of treating patients without PST.[7] Assuming an equivalence margin of 3%, we estimated a sample size of 146 to achieve at least 82% power to test a hypothesis of NPV 95% using a 1‐sided Z test with a type‐I error rate of 5%.[8] Once the sample size of 146 subjects was reached, we stopped recruiting patients.
Sample characteristics of the subjects who underwent testing were summarized using descriptive statistics. Sample proportions were calculated to summarize categorical variables. Mean and standard deviation were calculated to summarize continuous variables. Cost analysis of antibiotic therapy was estimated from the Vidant Medical Center antibiotic pharmaceutical acquisition costs. Estimated cost of peripherally inserted central catheter (PICC) placement and removal as well as laboratory testing costs were obtained from our institution's medical billing department. Marketing costs of pharmacist drug calibration and nursing assessments with dressing changes were obtained from hospital‐affiliated outpatient antibiotic infusion companies.
RESULTS
A total of 4031 allergy histories were reviewed during the 5‐month study period to achieve the sample size of 146 patients (Table 1). Of those, 3885 were excluded (Figure 2). Common infections included pneumonias (26%) and urinary tract infections (20%) (Table 2) Only 1 subject had a positive reaction with hives, edema, and itching approximately 6 minutes after the agents were injected intradermally. The remaining 145 (99%) had negative reactions to the PST and oral challenge and were then successfully transitioned to a ‐lactam agent without any reaction at 24 hours, giving an NPV of 100%. Ten subjects were switched from intravenous to oral ‐lactam agents (Figure 1). Avoidance of PICC placement ($1,200) and removal ($65), dressing changes, weekly drug‐level testing, laboratory technician, and pharmaceutical drug calibration costs allowed for a healthcare reduction of $5,233 ($520/patient) based on the 146 patients studied. The total cost of therapy would have been $113,991 if the PST had not been performed. However, the cost of altered therapy following a negative PST was $81,180, a difference of $32,811 ($225/per patient) in a 5‐month period. The total estimated annual difference, including antibiotic alteration and associated drug‐costs, would be $82,000.
| Antibiotic | No. of Patients Reporting An Allergy | % Per Total Charts Reviewed |
|---|---|---|
| Penicillin | 428 | 10.6 |
| Sulfonamide | 271 | 6.7 |
| Quinolone | 108 | 2.7 |
| Cephalosporin | 81 | 2.0 |
| Macrolide | 65 | 1.6 |
| Vancomycin | 39 | 0.9 |
| Tetracycline | 20 | 0.5 |
| Clindamycin | 18 | 0.4 |
| Metronidazole | 9 | 0.2 |
| Linezolid | 2 | 0.05 |
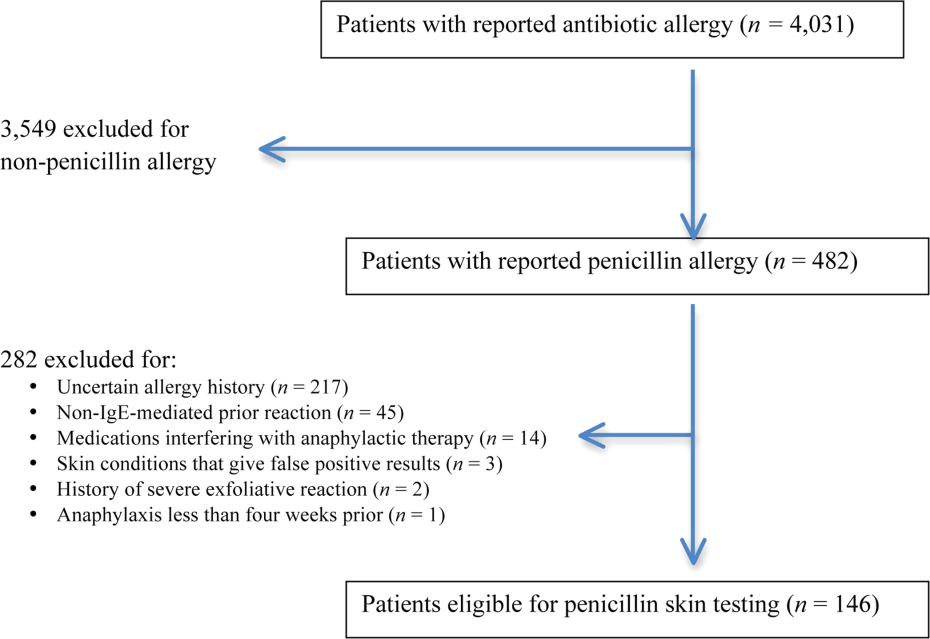
| Categories | No. of Patients (%) |
|---|---|
| |
| Time since last reported penicillin use | |
| 1 month1 year | 6 (4) |
| 25 years | 39 (27) |
| 610 years | 23 (16) |
| >10 years | 78 (53) |
| Reported IgE‐mediated reactions | |
| Bronchospasm | 23 (16) |
| Urticarial rash | 100 (68) |
| Edema | 32 (22) |
| Anaphylaxis | 21 (14) |
| Age on admission, y | |
| 2050 | 28 (19) |
| 5160 | 29 (20) |
| 6170 | 41 (28) |
| 7180 | 24 (16) |
| >80 | 24 (16) |
| Gender | |
| Male | 55 (40) |
| Female | 88 (60) |
| Race | |
| White | 82 (56) |
| Black | 61 (42) |
| Hispanic | 3 (2) |
| Infections being treated | |
| Bacteremia | 7 (4.8) |
| Catheter‐related bloodstream infection | 2 (1.4) |
| Empyema | 1 (0.7) |
| Epidural abscess | 2 (1.4) |
| Infective endocarditis | 4 (2.7) |
| Intra‐abdominal infection | 24 (16.4) |
| Meningitis | 1 (0.7) |
| Neutropenic fever | 1 (0.7) |
| Osteomyelitis | 6 (4.1) |
| Pericardial effusion | 1 (0.7) |
| Prosthetic joint infection | 5 (3.4) |
| Pneumonia | 40 (27.4) |
| Skin and soft‐tissue infection | 20 (13.7) |
| Syphilis | 3 (2.1) |
| Urinary tract infection | 29 (19.7) |
DISCUSSION
PST is the most rapid, sensitive, and cost‐effective modality for evaluating patients with immediate allergic reactions to penicillin. Over 90% of individuals with a true history of penicillin allergy have confirmed sensitivity with a PST, implying most patients who are skin tested negative are truly not allergic.[7, 9, 10, 11, 12] Our study shows that the reapproved PST with the PPL and penicillin G determinants continues to have a high NPV. A patient with a negative PST result is generally at a low risk of developing an immediate‐type hypersensitivity reaction to penicillin.[2, 11] PST frequently allowed for less expensive agents that would have been avoided due to a reported allergy. The estimated annual savings of $82,000 dollars from antibiotic alteration with successful transition to a ‐lactam agent after a negative PST illustrates its value, supports its validity, and makes this study novel.
Many ‐lactamase inhibitors (ie, piperacillin‐tazobactam), fourth generation cephalosporins (ie, cefepime), and carbapenems still remain costly. Despite this, we were still able to achieve a significant reduction in overall cost. In addition to financial benefits, PST allowed for the use of more appropriate agents with less potential adverse effects. Narrow‐spectrum, non‐lactam agents were sometimes altered to a broader‐spectrum ‐lactam agent. We also frequently tailored 2 agents to just 1 broad‐spectrum ‐lactam. This led to more patients being given broad‐spectrum agents after the PST (72 vs 89 patients). However, we were able to avoid using second‐line agents, such as aztreonam, vancomycin, linezolid, daptomycin, and tobramycin, in many patients with infections that are often best treated with penicillin‐based antibiotics (ie, syphilis, group B Streptococcus infections). With increasing incidence and recovery of multidrug‐resistant bacteria, PST may also allow use of potentially more effective antimicrobial agents.
A possible limitation is that our prevalence of a true penicillin allergy was 1%, whereas Bousquet et al. illustrate a higher prevalence of about 20%.[7] Although our prevalence may not be generalizable, Bousquet's study only assessed patients with allergies 5 years prior.
The introduction of PST into clinical practice will allow trained healthcare providers to prescribe cheaper, more appropriate, less toxic antimicrobial agents. The overall benefit of reintroducing penicillin agents when needed in the future is far more cost‐effective than what is described here. PST should become a standard of care when prescribing antibiotics to patients with a history of penicillin allergy. Medical providers should be aware of its utility, acquire training, and incorporate it into their practice.
Acknowledgment
Disclosures: Paul P. Cook, MD, has potential conflicts of interest with Gilead (investigator), Pfizer (investigator), Merck (investigator and speakers' bureau), and Forest (speakers' bureau). Neither he nor any of the other authors has received any sources of funding for this article. For the remaining authors, no conflicts were declared. The corresponding author, Ramzy Rimawi, MD, had full access to all of the data in the study and had final responsibility for the decision to submit for publication.
- , , . Elective penicillin skin testing in a pediatric outpatient setting. Ann Allergy Asthma Immunol. 2006;97(6):807–812.
- US Department of Veterans Affairs Web site. Benzypenicilloyl polylisine (PRE‐PEN) national drug monograph. May 2012. Available at: http://www.pbm.va.gov/DrugMonograph.aspx. Accessed September 1, 2012.
- , . Diagnosis and management of penicillin allergy. Mayo Clin Proc. 2005;80(3):405–410.
- PRE‐PEN penicillin skin test antigen. Available at: http://www.alk‐abello.com/us/products/pre‐pen/Pages/PREPEN.aspx. Accessed September 1, 2012.
- , , , et al.; Joint Task Force on Practice Parameters; American Academy of Allergy, Asthma and Immunology; American College of Allergy, Asthma and Immunology; Joint Council of Allergy, Asthma and Immunology. Drug allergy: an updated practice parameter. Ann Allergy Asthma Immunol. 2010;105:259–273.
- . Immunochemical mechanisms in penicillin allergy. Fed Proc. 1965;24:51–54.
- , , , et al. Oral challenges are needed in the diagnosis of beta‐lactam hypersensitivity. Clin Exp Allergy. 2008;38(1):185–190.
- , , . Sample Size Calculations in Clinical Research. New York, NY: Chapman 2003.
- , , , et al. Retrospective case series analysis of penicillin allergy testing in a UK specialist regional allergy clinic. J Clin Pathol. 2011;64:1014–1018.
- . Prevalence of skin test reactivity in patients with convincing, vague and unnacceptible histories of penicillin allergy. Allergy Asthma Proc. 2005;26(1):59–64.
- , . Frequency of systemic reactions to penicillin skin tests. Ann Allergy Asthma Immunol. 2000;85:363–365.
- , , , et al. The incidence of antimicrobial allergies in hospitalized patients. Arch Intern Med. 2000;160;2819–2822.
Self‐reported penicillin allergy is common and frequently limits the available antimicrobial agents to choose from. This often results in the use of more expensive, potentially more toxic, and possibly less efficacious agents.[1, 2]
For over 30 years, penicilloyl‐polylysine (PPL) penicillin skin testing (PST) was widely used to diagnose penicillin allergy with a negative predictive value (NPV) of about 97% to 99%.[3] After being off the market for 5 years, PPL PST was reapproved in 2009 as PRE‐PEN.[4] However, many clinicians still fail to utilize PST despite its simplicity and substantial clinical impact. The main purpose of this study was to describe the predictive value of PST and impact on antibiotic selection in a sample of hospitalized patients with a reported history of penicillin allergy.
METHODS
In 2010, PST was introduced as a quality‐improvement measure after approval and support from the chief of professional services and the medical staff executive committee at Vidant Medical Center, an 861‐bed tertiary care and teaching hospital. Our antimicrobial stewardship program is regularly contacted for approval of alternative therapies in penicillin allergic patients. The PST quality‐improvement intervention was implemented to avoid resorting to less appropriate therapies in these situations. Following approval by the University and Medical Center Institutional Review Board, we designed a 4‐month study to assess the impact of this ongoing quality improvement measure from March 2012 to July 2012.
Hospitalized patients of all ages with reported penicillin allergies were obtained from our antimicrobial stewardship database. Their charts were reviewed for demographics, antibiotic use, clinical infection, and allergic description. Deciding whether to alter antibiotic therapy to a ‐lactam regimen was based on microbiologic results, laboratory values, clinical infection, and history of immunoglobulin E (IgE)‐mediated reactions, as defined by the updated drug allergy practice parameters.[5] IgE‐mediated reactions included: (1) immediate urticaria, laryngeal edema, or hypotension; (2) anemia; and (3) fever, arthralgias, lymphadenopathy, and an urticarial rash after 7 to 21 days.[5, 6, 7] We defined anaphylaxis as the development of angioedema or hemodynamic instability within 1 hour of penicillin administration. A true negative reaction was a lack of an IgE‐mediated reaction to all the drug challenges.
Patients in the medical, surgical, labor, and delivery wards; intensive care units; and emergency department underwent testing. The ‐lactam agent used after a negative PST was recorded, and the patients were followed for 24 hours after transitioning their therapy to a ‐lactam regimen. Excluded subjects included those with (1) nonIgE‐mediated reactions, (2) skin conditions that can give false positive results, (3) medications that may interfere with anaphylactic therapy, (4) history of severe exfoliative reactions to ‐lactams, (5) anaphylaxis less than 4 weeks prior, (6) allergies to antibiotics other than penicillin, and (7) uncertain allergy history.
PST Reagents/Procedure
Our benzylpenicilloyl major determinant molecule, commercially produced as PPL, was purchased as a PRE‐PEN from ALK‐Abello, Round Rock, Texas. Penicillin G potassium, purchased from Pfizer, New York, New York, is the only commercially available minor determinant and can improve identification of penicillin allergy by up to 97%.[2] The PST panel also included histamine (positive control) and normal saline (negative control).
Skin Testing Procedure
An infectious diseases fellow (R.H.R. or B.K.) was supervised in preparing for potential anaphylaxis, applying the reagents and interpreting the results based on drug allergy practice parameters.[5] The preliminary epicutaneous prick/puncture test was performed with a lancet in subjects without prior anaphylaxis using full‐strength PPL and penicillin G potassium reagents. If there was no response within 15 minutes, which we defined as a lack of wheal formation 3 mm or greater than that of the negative control, 0.02 to 0.03 mL of each reagent was injected intradermally using a tuberculin syringe and examined for 15 minutes.[5] If there was no response, patients were then challenged with either a single oral dose of penicillin V potassium 250 mg or whichever oral penicillin agent they previously reported an allergy to. If no reaction was appreciated within 2 hours, their therapy was changed to a ‐lactam agent including penicillins, cephalosporins, and carbapenems for the remaining duration of therapy (Figure 1) An estimate of NPV was obtained after 24 hours follow‐up.

Statistical Analysis
We designed a study to estimate whether the reapproved PST achieves an NPV of at least 95%.[3] We hypothesized that clinicians will be willing to utilize PST even if it has an NPV of slightly less than 98% compared to the current standard of treating patients without PST.[7] Assuming an equivalence margin of 3%, we estimated a sample size of 146 to achieve at least 82% power to test a hypothesis of NPV 95% using a 1‐sided Z test with a type‐I error rate of 5%.[8] Once the sample size of 146 subjects was reached, we stopped recruiting patients.
Sample characteristics of the subjects who underwent testing were summarized using descriptive statistics. Sample proportions were calculated to summarize categorical variables. Mean and standard deviation were calculated to summarize continuous variables. Cost analysis of antibiotic therapy was estimated from the Vidant Medical Center antibiotic pharmaceutical acquisition costs. Estimated cost of peripherally inserted central catheter (PICC) placement and removal as well as laboratory testing costs were obtained from our institution's medical billing department. Marketing costs of pharmacist drug calibration and nursing assessments with dressing changes were obtained from hospital‐affiliated outpatient antibiotic infusion companies.
RESULTS
A total of 4031 allergy histories were reviewed during the 5‐month study period to achieve the sample size of 146 patients (Table 1). Of those, 3885 were excluded (Figure 2). Common infections included pneumonias (26%) and urinary tract infections (20%) (Table 2) Only 1 subject had a positive reaction with hives, edema, and itching approximately 6 minutes after the agents were injected intradermally. The remaining 145 (99%) had negative reactions to the PST and oral challenge and were then successfully transitioned to a ‐lactam agent without any reaction at 24 hours, giving an NPV of 100%. Ten subjects were switched from intravenous to oral ‐lactam agents (Figure 1). Avoidance of PICC placement ($1,200) and removal ($65), dressing changes, weekly drug‐level testing, laboratory technician, and pharmaceutical drug calibration costs allowed for a healthcare reduction of $5,233 ($520/patient) based on the 146 patients studied. The total cost of therapy would have been $113,991 if the PST had not been performed. However, the cost of altered therapy following a negative PST was $81,180, a difference of $32,811 ($225/per patient) in a 5‐month period. The total estimated annual difference, including antibiotic alteration and associated drug‐costs, would be $82,000.
| Antibiotic | No. of Patients Reporting An Allergy | % Per Total Charts Reviewed |
|---|---|---|
| Penicillin | 428 | 10.6 |
| Sulfonamide | 271 | 6.7 |
| Quinolone | 108 | 2.7 |
| Cephalosporin | 81 | 2.0 |
| Macrolide | 65 | 1.6 |
| Vancomycin | 39 | 0.9 |
| Tetracycline | 20 | 0.5 |
| Clindamycin | 18 | 0.4 |
| Metronidazole | 9 | 0.2 |
| Linezolid | 2 | 0.05 |

| Categories | No. of Patients (%) |
|---|---|
| |
| Time since last reported penicillin use | |
| 1 month1 year | 6 (4) |
| 25 years | 39 (27) |
| 610 years | 23 (16) |
| >10 years | 78 (53) |
| Reported IgE‐mediated reactions | |
| Bronchospasm | 23 (16) |
| Urticarial rash | 100 (68) |
| Edema | 32 (22) |
| Anaphylaxis | 21 (14) |
| Age on admission, y | |
| 2050 | 28 (19) |
| 5160 | 29 (20) |
| 6170 | 41 (28) |
| 7180 | 24 (16) |
| >80 | 24 (16) |
| Gender | |
| Male | 55 (40) |
| Female | 88 (60) |
| Race | |
| White | 82 (56) |
| Black | 61 (42) |
| Hispanic | 3 (2) |
| Infections being treated | |
| Bacteremia | 7 (4.8) |
| Catheter‐related bloodstream infection | 2 (1.4) |
| Empyema | 1 (0.7) |
| Epidural abscess | 2 (1.4) |
| Infective endocarditis | 4 (2.7) |
| Intra‐abdominal infection | 24 (16.4) |
| Meningitis | 1 (0.7) |
| Neutropenic fever | 1 (0.7) |
| Osteomyelitis | 6 (4.1) |
| Pericardial effusion | 1 (0.7) |
| Prosthetic joint infection | 5 (3.4) |
| Pneumonia | 40 (27.4) |
| Skin and soft‐tissue infection | 20 (13.7) |
| Syphilis | 3 (2.1) |
| Urinary tract infection | 29 (19.7) |
DISCUSSION
PST is the most rapid, sensitive, and cost‐effective modality for evaluating patients with immediate allergic reactions to penicillin. Over 90% of individuals with a true history of penicillin allergy have confirmed sensitivity with a PST, implying most patients who are skin tested negative are truly not allergic.[7, 9, 10, 11, 12] Our study shows that the reapproved PST with the PPL and penicillin G determinants continues to have a high NPV. A patient with a negative PST result is generally at a low risk of developing an immediate‐type hypersensitivity reaction to penicillin.[2, 11] PST frequently allowed for less expensive agents that would have been avoided due to a reported allergy. The estimated annual savings of $82,000 dollars from antibiotic alteration with successful transition to a ‐lactam agent after a negative PST illustrates its value, supports its validity, and makes this study novel.
Many ‐lactamase inhibitors (ie, piperacillin‐tazobactam), fourth generation cephalosporins (ie, cefepime), and carbapenems still remain costly. Despite this, we were still able to achieve a significant reduction in overall cost. In addition to financial benefits, PST allowed for the use of more appropriate agents with less potential adverse effects. Narrow‐spectrum, non‐lactam agents were sometimes altered to a broader‐spectrum ‐lactam agent. We also frequently tailored 2 agents to just 1 broad‐spectrum ‐lactam. This led to more patients being given broad‐spectrum agents after the PST (72 vs 89 patients). However, we were able to avoid using second‐line agents, such as aztreonam, vancomycin, linezolid, daptomycin, and tobramycin, in many patients with infections that are often best treated with penicillin‐based antibiotics (ie, syphilis, group B Streptococcus infections). With increasing incidence and recovery of multidrug‐resistant bacteria, PST may also allow use of potentially more effective antimicrobial agents.
A possible limitation is that our prevalence of a true penicillin allergy was 1%, whereas Bousquet et al. illustrate a higher prevalence of about 20%.[7] Although our prevalence may not be generalizable, Bousquet's study only assessed patients with allergies 5 years prior.
The introduction of PST into clinical practice will allow trained healthcare providers to prescribe cheaper, more appropriate, less toxic antimicrobial agents. The overall benefit of reintroducing penicillin agents when needed in the future is far more cost‐effective than what is described here. PST should become a standard of care when prescribing antibiotics to patients with a history of penicillin allergy. Medical providers should be aware of its utility, acquire training, and incorporate it into their practice.
Acknowledgment
Disclosures: Paul P. Cook, MD, has potential conflicts of interest with Gilead (investigator), Pfizer (investigator), Merck (investigator and speakers' bureau), and Forest (speakers' bureau). Neither he nor any of the other authors has received any sources of funding for this article. For the remaining authors, no conflicts were declared. The corresponding author, Ramzy Rimawi, MD, had full access to all of the data in the study and had final responsibility for the decision to submit for publication.
Self‐reported penicillin allergy is common and frequently limits the available antimicrobial agents to choose from. This often results in the use of more expensive, potentially more toxic, and possibly less efficacious agents.[1, 2]
For over 30 years, penicilloyl‐polylysine (PPL) penicillin skin testing (PST) was widely used to diagnose penicillin allergy with a negative predictive value (NPV) of about 97% to 99%.[3] After being off the market for 5 years, PPL PST was reapproved in 2009 as PRE‐PEN.[4] However, many clinicians still fail to utilize PST despite its simplicity and substantial clinical impact. The main purpose of this study was to describe the predictive value of PST and impact on antibiotic selection in a sample of hospitalized patients with a reported history of penicillin allergy.
METHODS
In 2010, PST was introduced as a quality‐improvement measure after approval and support from the chief of professional services and the medical staff executive committee at Vidant Medical Center, an 861‐bed tertiary care and teaching hospital. Our antimicrobial stewardship program is regularly contacted for approval of alternative therapies in penicillin allergic patients. The PST quality‐improvement intervention was implemented to avoid resorting to less appropriate therapies in these situations. Following approval by the University and Medical Center Institutional Review Board, we designed a 4‐month study to assess the impact of this ongoing quality improvement measure from March 2012 to July 2012.
Hospitalized patients of all ages with reported penicillin allergies were obtained from our antimicrobial stewardship database. Their charts were reviewed for demographics, antibiotic use, clinical infection, and allergic description. Deciding whether to alter antibiotic therapy to a ‐lactam regimen was based on microbiologic results, laboratory values, clinical infection, and history of immunoglobulin E (IgE)‐mediated reactions, as defined by the updated drug allergy practice parameters.[5] IgE‐mediated reactions included: (1) immediate urticaria, laryngeal edema, or hypotension; (2) anemia; and (3) fever, arthralgias, lymphadenopathy, and an urticarial rash after 7 to 21 days.[5, 6, 7] We defined anaphylaxis as the development of angioedema or hemodynamic instability within 1 hour of penicillin administration. A true negative reaction was a lack of an IgE‐mediated reaction to all the drug challenges.
Patients in the medical, surgical, labor, and delivery wards; intensive care units; and emergency department underwent testing. The ‐lactam agent used after a negative PST was recorded, and the patients were followed for 24 hours after transitioning their therapy to a ‐lactam regimen. Excluded subjects included those with (1) nonIgE‐mediated reactions, (2) skin conditions that can give false positive results, (3) medications that may interfere with anaphylactic therapy, (4) history of severe exfoliative reactions to ‐lactams, (5) anaphylaxis less than 4 weeks prior, (6) allergies to antibiotics other than penicillin, and (7) uncertain allergy history.
PST Reagents/Procedure
Our benzylpenicilloyl major determinant molecule, commercially produced as PPL, was purchased as a PRE‐PEN from ALK‐Abello, Round Rock, Texas. Penicillin G potassium, purchased from Pfizer, New York, New York, is the only commercially available minor determinant and can improve identification of penicillin allergy by up to 97%.[2] The PST panel also included histamine (positive control) and normal saline (negative control).
Skin Testing Procedure
An infectious diseases fellow (R.H.R. or B.K.) was supervised in preparing for potential anaphylaxis, applying the reagents and interpreting the results based on drug allergy practice parameters.[5] The preliminary epicutaneous prick/puncture test was performed with a lancet in subjects without prior anaphylaxis using full‐strength PPL and penicillin G potassium reagents. If there was no response within 15 minutes, which we defined as a lack of wheal formation 3 mm or greater than that of the negative control, 0.02 to 0.03 mL of each reagent was injected intradermally using a tuberculin syringe and examined for 15 minutes.[5] If there was no response, patients were then challenged with either a single oral dose of penicillin V potassium 250 mg or whichever oral penicillin agent they previously reported an allergy to. If no reaction was appreciated within 2 hours, their therapy was changed to a ‐lactam agent including penicillins, cephalosporins, and carbapenems for the remaining duration of therapy (Figure 1) An estimate of NPV was obtained after 24 hours follow‐up.

Statistical Analysis
We designed a study to estimate whether the reapproved PST achieves an NPV of at least 95%.[3] We hypothesized that clinicians will be willing to utilize PST even if it has an NPV of slightly less than 98% compared to the current standard of treating patients without PST.[7] Assuming an equivalence margin of 3%, we estimated a sample size of 146 to achieve at least 82% power to test a hypothesis of NPV 95% using a 1‐sided Z test with a type‐I error rate of 5%.[8] Once the sample size of 146 subjects was reached, we stopped recruiting patients.
Sample characteristics of the subjects who underwent testing were summarized using descriptive statistics. Sample proportions were calculated to summarize categorical variables. Mean and standard deviation were calculated to summarize continuous variables. Cost analysis of antibiotic therapy was estimated from the Vidant Medical Center antibiotic pharmaceutical acquisition costs. Estimated cost of peripherally inserted central catheter (PICC) placement and removal as well as laboratory testing costs were obtained from our institution's medical billing department. Marketing costs of pharmacist drug calibration and nursing assessments with dressing changes were obtained from hospital‐affiliated outpatient antibiotic infusion companies.
RESULTS
A total of 4031 allergy histories were reviewed during the 5‐month study period to achieve the sample size of 146 patients (Table 1). Of those, 3885 were excluded (Figure 2). Common infections included pneumonias (26%) and urinary tract infections (20%) (Table 2) Only 1 subject had a positive reaction with hives, edema, and itching approximately 6 minutes after the agents were injected intradermally. The remaining 145 (99%) had negative reactions to the PST and oral challenge and were then successfully transitioned to a ‐lactam agent without any reaction at 24 hours, giving an NPV of 100%. Ten subjects were switched from intravenous to oral ‐lactam agents (Figure 1). Avoidance of PICC placement ($1,200) and removal ($65), dressing changes, weekly drug‐level testing, laboratory technician, and pharmaceutical drug calibration costs allowed for a healthcare reduction of $5,233 ($520/patient) based on the 146 patients studied. The total cost of therapy would have been $113,991 if the PST had not been performed. However, the cost of altered therapy following a negative PST was $81,180, a difference of $32,811 ($225/per patient) in a 5‐month period. The total estimated annual difference, including antibiotic alteration and associated drug‐costs, would be $82,000.
| Antibiotic | No. of Patients Reporting An Allergy | % Per Total Charts Reviewed |
|---|---|---|
| Penicillin | 428 | 10.6 |
| Sulfonamide | 271 | 6.7 |
| Quinolone | 108 | 2.7 |
| Cephalosporin | 81 | 2.0 |
| Macrolide | 65 | 1.6 |
| Vancomycin | 39 | 0.9 |
| Tetracycline | 20 | 0.5 |
| Clindamycin | 18 | 0.4 |
| Metronidazole | 9 | 0.2 |
| Linezolid | 2 | 0.05 |

| Categories | No. of Patients (%) |
|---|---|
| |
| Time since last reported penicillin use | |
| 1 month1 year | 6 (4) |
| 25 years | 39 (27) |
| 610 years | 23 (16) |
| >10 years | 78 (53) |
| Reported IgE‐mediated reactions | |
| Bronchospasm | 23 (16) |
| Urticarial rash | 100 (68) |
| Edema | 32 (22) |
| Anaphylaxis | 21 (14) |
| Age on admission, y | |
| 2050 | 28 (19) |
| 5160 | 29 (20) |
| 6170 | 41 (28) |
| 7180 | 24 (16) |
| >80 | 24 (16) |
| Gender | |
| Male | 55 (40) |
| Female | 88 (60) |
| Race | |
| White | 82 (56) |
| Black | 61 (42) |
| Hispanic | 3 (2) |
| Infections being treated | |
| Bacteremia | 7 (4.8) |
| Catheter‐related bloodstream infection | 2 (1.4) |
| Empyema | 1 (0.7) |
| Epidural abscess | 2 (1.4) |
| Infective endocarditis | 4 (2.7) |
| Intra‐abdominal infection | 24 (16.4) |
| Meningitis | 1 (0.7) |
| Neutropenic fever | 1 (0.7) |
| Osteomyelitis | 6 (4.1) |
| Pericardial effusion | 1 (0.7) |
| Prosthetic joint infection | 5 (3.4) |
| Pneumonia | 40 (27.4) |
| Skin and soft‐tissue infection | 20 (13.7) |
| Syphilis | 3 (2.1) |
| Urinary tract infection | 29 (19.7) |
DISCUSSION
PST is the most rapid, sensitive, and cost‐effective modality for evaluating patients with immediate allergic reactions to penicillin. Over 90% of individuals with a true history of penicillin allergy have confirmed sensitivity with a PST, implying most patients who are skin tested negative are truly not allergic.[7, 9, 10, 11, 12] Our study shows that the reapproved PST with the PPL and penicillin G determinants continues to have a high NPV. A patient with a negative PST result is generally at a low risk of developing an immediate‐type hypersensitivity reaction to penicillin.[2, 11] PST frequently allowed for less expensive agents that would have been avoided due to a reported allergy. The estimated annual savings of $82,000 dollars from antibiotic alteration with successful transition to a ‐lactam agent after a negative PST illustrates its value, supports its validity, and makes this study novel.
Many ‐lactamase inhibitors (ie, piperacillin‐tazobactam), fourth generation cephalosporins (ie, cefepime), and carbapenems still remain costly. Despite this, we were still able to achieve a significant reduction in overall cost. In addition to financial benefits, PST allowed for the use of more appropriate agents with less potential adverse effects. Narrow‐spectrum, non‐lactam agents were sometimes altered to a broader‐spectrum ‐lactam agent. We also frequently tailored 2 agents to just 1 broad‐spectrum ‐lactam. This led to more patients being given broad‐spectrum agents after the PST (72 vs 89 patients). However, we were able to avoid using second‐line agents, such as aztreonam, vancomycin, linezolid, daptomycin, and tobramycin, in many patients with infections that are often best treated with penicillin‐based antibiotics (ie, syphilis, group B Streptococcus infections). With increasing incidence and recovery of multidrug‐resistant bacteria, PST may also allow use of potentially more effective antimicrobial agents.
A possible limitation is that our prevalence of a true penicillin allergy was 1%, whereas Bousquet et al. illustrate a higher prevalence of about 20%.[7] Although our prevalence may not be generalizable, Bousquet's study only assessed patients with allergies 5 years prior.
The introduction of PST into clinical practice will allow trained healthcare providers to prescribe cheaper, more appropriate, less toxic antimicrobial agents. The overall benefit of reintroducing penicillin agents when needed in the future is far more cost‐effective than what is described here. PST should become a standard of care when prescribing antibiotics to patients with a history of penicillin allergy. Medical providers should be aware of its utility, acquire training, and incorporate it into their practice.
Acknowledgment
Disclosures: Paul P. Cook, MD, has potential conflicts of interest with Gilead (investigator), Pfizer (investigator), Merck (investigator and speakers' bureau), and Forest (speakers' bureau). Neither he nor any of the other authors has received any sources of funding for this article. For the remaining authors, no conflicts were declared. The corresponding author, Ramzy Rimawi, MD, had full access to all of the data in the study and had final responsibility for the decision to submit for publication.
- , , . Elective penicillin skin testing in a pediatric outpatient setting. Ann Allergy Asthma Immunol. 2006;97(6):807–812.
- US Department of Veterans Affairs Web site. Benzypenicilloyl polylisine (PRE‐PEN) national drug monograph. May 2012. Available at: http://www.pbm.va.gov/DrugMonograph.aspx. Accessed September 1, 2012.
- , . Diagnosis and management of penicillin allergy. Mayo Clin Proc. 2005;80(3):405–410.
- PRE‐PEN penicillin skin test antigen. Available at: http://www.alk‐abello.com/us/products/pre‐pen/Pages/PREPEN.aspx. Accessed September 1, 2012.
- , , , et al.; Joint Task Force on Practice Parameters; American Academy of Allergy, Asthma and Immunology; American College of Allergy, Asthma and Immunology; Joint Council of Allergy, Asthma and Immunology. Drug allergy: an updated practice parameter. Ann Allergy Asthma Immunol. 2010;105:259–273.
- . Immunochemical mechanisms in penicillin allergy. Fed Proc. 1965;24:51–54.
- , , , et al. Oral challenges are needed in the diagnosis of beta‐lactam hypersensitivity. Clin Exp Allergy. 2008;38(1):185–190.
- , , . Sample Size Calculations in Clinical Research. New York, NY: Chapman 2003.
- , , , et al. Retrospective case series analysis of penicillin allergy testing in a UK specialist regional allergy clinic. J Clin Pathol. 2011;64:1014–1018.
- . Prevalence of skin test reactivity in patients with convincing, vague and unnacceptible histories of penicillin allergy. Allergy Asthma Proc. 2005;26(1):59–64.
- , . Frequency of systemic reactions to penicillin skin tests. Ann Allergy Asthma Immunol. 2000;85:363–365.
- , , , et al. The incidence of antimicrobial allergies in hospitalized patients. Arch Intern Med. 2000;160;2819–2822.
- , , . Elective penicillin skin testing in a pediatric outpatient setting. Ann Allergy Asthma Immunol. 2006;97(6):807–812.
- US Department of Veterans Affairs Web site. Benzypenicilloyl polylisine (PRE‐PEN) national drug monograph. May 2012. Available at: http://www.pbm.va.gov/DrugMonograph.aspx. Accessed September 1, 2012.
- , . Diagnosis and management of penicillin allergy. Mayo Clin Proc. 2005;80(3):405–410.
- PRE‐PEN penicillin skin test antigen. Available at: http://www.alk‐abello.com/us/products/pre‐pen/Pages/PREPEN.aspx. Accessed September 1, 2012.
- , , , et al.; Joint Task Force on Practice Parameters; American Academy of Allergy, Asthma and Immunology; American College of Allergy, Asthma and Immunology; Joint Council of Allergy, Asthma and Immunology. Drug allergy: an updated practice parameter. Ann Allergy Asthma Immunol. 2010;105:259–273.
- . Immunochemical mechanisms in penicillin allergy. Fed Proc. 1965;24:51–54.
- , , , et al. Oral challenges are needed in the diagnosis of beta‐lactam hypersensitivity. Clin Exp Allergy. 2008;38(1):185–190.
- , , . Sample Size Calculations in Clinical Research. New York, NY: Chapman 2003.
- , , , et al. Retrospective case series analysis of penicillin allergy testing in a UK specialist regional allergy clinic. J Clin Pathol. 2011;64:1014–1018.
- . Prevalence of skin test reactivity in patients with convincing, vague and unnacceptible histories of penicillin allergy. Allergy Asthma Proc. 2005;26(1):59–64.
- , . Frequency of systemic reactions to penicillin skin tests. Ann Allergy Asthma Immunol. 2000;85:363–365.
- , , , et al. The incidence of antimicrobial allergies in hospitalized patients. Arch Intern Med. 2000;160;2819–2822.
FDR and Telemetry Rhythm at Time of IHCA
In‐hospital cardiac arrest (IHCA) research often relies on the first documented cardiac rhythm (FDR) on resuscitation records at the time of cardiopulmonary resuscitation (CPR) initiation as a surrogate for arrest etiology.[1] Over 1000 hospitals report the FDR and associated cardiac arrest data to national registries annually.[2, 3] These data are subsequently used to report national IHCA epidemiology, as well as to develop and refine guidelines for in‐hospital resuscitation.[4]
Suspecting that the FDR might represent the later stage of a progressive cardiopulmonary process rather than a sudden dysrhythmia, we sought to compare the first rhythm documented on resuscitation records at the time of CPR initiation with the telemetry rhythm at the time of the code blue call. We hypothesized that the agreement between FDR and telemetry rhythm would be 80% beyond that predicted by chance (kappa0.8).[5]
METHODS
Design
Between June 2008 and February 2010, we performed a cross‐sectional study at a 750‐bed adult tertiary care hospital (Christiana Hospital) and a 240‐bed adult inner city community hospital (Wilmington Hospital). Both hospitals included teaching and nonteaching inpatient services. The Christiana Care Health System Institutional Review Board approved the study.
Study Population
Eligible subjects included a convenience sample of adult inpatients aged 18 years who were monitored on the hospital's telemetry system during the 2 minutes prior to a code blue call from a nonintensive care, noncardiac care inpatient ward for IHCA. Intensive care unit (ICU) locations were excluded because they are not captured in our central telemetry recording system. We defined IHCA as a resuscitation event requiring >1 minute of chest compressions and/or defibrillation. We excluded patients with do not attempt resuscitation orders at the time of the IHCA. For patients with multiple IHCAs, only their first event was included in the analysis. International Classification of Diseases, 9th Revision admission diagnoses were categorized into infectious, oncology, endocrine/metabolic; cardiovascular, renal, or other disease categories. The decision to place patients on telemetry monitoring was not part of the study and was entirely at the discretion of the physicians caring for the patients.
Variables and Measurements
We reviewed the paper resuscitation records of each IHCA during the study period and identified the FDR. To create groups that would allow comparison between telemetry and resuscitation record rhythms, we placed each rhythm into 1 of the following 3 categories: asystole, ventricular tachyarrhythmia (VTA), or other organized rhythms (Table 1). It was not possible to retrospectively ascertain the presence of pulses to determine if an organized rhythm identified on telemetry tracings was pulseless electrical activity (PEA) or a perfusing rhythm. Therefore, we elected to take a conservative approach that would bias toward agreement (the opposite direction of our hypothesis that the rhythms are discrepant) and consider all other organized rhythms in agreement with one another. We reviewed printouts of telemetry electrocardiographic records for each patient. Minute 0 was defined as the time of the code blue call. Two physician investigators (C.C. and U.B.) independently reviewed telemetry data for each patient at minute 0 and the 2 minutes preceding the code blue call (minutes 1 and 2). Rhythms at each minute mark were assigned to 1 of the following categories according to the classification scheme in Table 1: asystole, VTA, or other organized rhythms. Leads off and uninterpretable telemetry were also noted. Discrepancies in rhythm categorization between reviewers were resolved by a third investigator (M.Z.) blinded to rhythm category assignment. We used the telemetry rhythm at minute 0 for analysis whenever possible. If the leads were off or the telemetry was uninterpretable at minute 0, we used minute 1. If minute 1 was also unusable, we used minute 2. If there were no usable data at minutes 0, 1, or 2, we excluded the patient.
| Category | Rhythm |
|---|---|
| Asystole | Asystole |
| Ventricular tachyarrhythmia | Ventricular fibrillation, ventricular tachycardia |
| Other organized rhythms | Atrial fibrillation, bradycardia, paced pulseless electrical activity, sinus, idioventricular, other |
Statistical Analysis
We determined the percent agreement between the resuscitation record rhythm category and the last interpretable telemetry rhythm category. We then calculated an unweighted kappa for the agreement between the resuscitation record rhythm category and the last interpretable telemetry rhythm category.
RESULTS
During the study period, there were 135 code blue calls for urgent assistance among telemetry‐monitored non‐ICU patients. Of the 135 calls, we excluded 4 events (3%) that did not meet the definition of IHCA, 9 events (7%) with missing or uninterpretable data, and 53 events (39%) with unobtainable data due to automatic purging from the telemetry server. Therefore, 69 events in 69 different patients remained for analysis. Twelve of the 69 included arrests that occurred at Wilmington Hospital and 57 at Christiana Hospital. The characteristics of the patients are shown in Table 2.
| n | % | |
|---|---|---|
| Age, y | ||
| 3039 | 1 | 1.4 |
| 4049 | 4 | 5.8 |
| 5059 | 11 | 15.9 |
| 6069 | 15 | 21.7 |
| 7079 | 16 | 23.2 |
| 8089 | 18 | 26.1 |
| 90+ | 4 | 5.8 |
| Sex | ||
| Male | 26 | 37.7 |
| Female | 43 | 62.3 |
| Race/ethnicity | ||
| White | 51 | 73.9 |
| Black | 17 | 24.6 |
| Hispanic | 1 | 1.4 |
| Admission body mass index | ||
| Underweight (18.5) | 3 | 4.3 |
| Normal (18.525) | 15 | 21.7 |
| Overweight (2530) 24 | 24 | 34.8 |
| Obese (3035) | 17 | 24.6 |
| Very obese (35) | 9 | 13.0 |
| Unknown | 1 | 1.4 |
| Admission diagnosis category | ||
| Infectious | 29 | 42.0 |
| Oncology | 4 | 5.8 |
| Endocrine/metabolic | 22 | 31.9 |
| Cardiovascular | 7 | 10.1 |
| Renal | 2 | 2.8 |
| Other | 5 | 7.2 |
Of the 69 arrests, we used the telemetry rhythm at minute 0 in 42 patients (61%), minute 1 in 22 patients (32%), and minute 2 in 5 patients (7%). Agreement between telemetry and FDR was 65% (kappa=0.37, 95% confidence interval: 0.17‐0.56) (Table 3). Agreement did not vary significantly by sex, race, hospital, weekday, time of day, or minute used in the analysis. Agreement was not associated with survival to hospital discharge.
| Telemetry | Resuscitation Record | |||
|---|---|---|---|---|
| Asystole | Ventricular Tachyarrhythmia | Other Organized Rhythms | Total | |
| ||||
| Asystole | 3 | 0 | 2 | 5 |
| Ventricular tachyarrhythmia | 1 | 12 | 8 | 21 |
| Other organized rhythms | 8 | 5 | 30 | 43 |
| Total | 12 | 17 | 40 | 69 |
Of the 69 IHCA events, the FDRs vs telemetry rhythms at the time of IHCA were: asystole 17% vs 7%, VTA 25% vs 31%, and other organized rhythms 58% vs 62%. Among the 12 events with FDR recorded as asystole, telemetry at the time of the code call was asystole in 3 (25%), VTA in 1 (8%), and other organized rhythms in 8 (67%). Among the 17 events with FDR recorded as VTA, telemetry at the time of the code call was VTA in 12 (71%) and other organized rhythms in 5 (29%). Among the 40 events with FDR recorded as other organized rhythms, telemetry at the time of the code call was asystole in 2 (5%), VTA in 8 (20%), and other organized rhythms in 30 (75%). Among the 8 patients with VTA on telemetry and other organized rhythms on the resuscitation record, the other organized rhythms were documented as PEA (n=6), sinus (n=1), and bradycardia (n=1). Of the 12 patients with VTA on telemetry and on the resuscitation record, 8 (67%) had ventricular tachycardia on telemetry. Four of the 8 (50%) who had ventricular tachycardia on telemetry had deteriorated into ventricular fibrillation by the time the FDR was recorded. Of the 4 who had ventricular fibrillation on telemetry, all had ventricular fibrillation as the FDR on the resuscitation record.
DISCUSSION
These results establish that FDRs often differ from the telemetry rhythms at the time of the code blue call. This is important because national registries such as the American Heart Association's Get with the GuidelinesResuscitation[2] database use the FDR as a surrogate for arrest etiology, and use their findings to report national IHCA outcomes as well as to develop and refine evidence‐based guidelines for in‐hospital resuscitation. Our findings suggest that using the FDR may be an oversimplification of the complex progression of cardiac rhythms that occurs in the periarrest period. Adding preceding telemetry rhythms to the data elements collected may shed additional light on etiology. Furthermore, our results demonstrate that, among adults with VTA or asystole documented upon arrival of the code blue team, other organized rhythms are often present at the time the staff recognized a life‐threatening condition and called for immediate assistance. This suggests that the VTA and asystole FDRs may represent the later stages of progressive cardiopulmonary processes. This is in contrast to out‐of‐hospital cardiac arrests typically attributed to sudden catastrophic dysrhythmias that often progress to asystole unless rapidly defibrillated.[6, 7, 8] Out‐of‐hospital and in‐hospital arrests are likely different (but overlapping) entities that might benefit from different resuscitation strategies.[9, 10] We hypothesize that, for a subset of these patients, progressive respiratory insufficiency and circulatory shockconditions classically associated more strongly with pediatric than adult IHCAmay have been directly responsible for the event.[1] If future research supports the concept that progressive respiratory insufficiency and circulatory shock are responsible for more adult IHCA than previously recognized, more robust monitoring may be indicated for a larger subset of adult patients hospitalized on general wards. This could include pulse oximetry (wave form can be a surrogate for perfusion), respiratory rate, and/or end‐tidal CO2 monitoring. In addition, if future research confirms that there is a greater distinction between in‐hospital and out‐of‐hospital cardiac arrest etiology, the expert panels that develop resuscitation guidelines should consider including setting of resuscitation as a branch point in future algorithms.
Our study had several limitations. First, the sample size was small due to uninterpretable rhythm strips, and for 39% of the total code events, the telemetry data had already been purged from the system by the time research staff attempted to retrieve it. Although we do not believe that there was any systematic bias to the data analyzed, the possibility cannot be completely excluded. Second, we were constrained by the inability to retrospectively ascertain the presence of pulses to determine if an organized rhythm identified on telemetry tracings was PEA. Thus, we categorized rhythms into large groups. Although this limited the granularity of the rhythm groups, it was a conservative approach that likely biased toward agreement (the opposite direction of our hypothesis). Third, the lack of perfect time synchronization between the telemetry system, wall clocks in the hospital, and wrist watches that may be referenced when documenting resuscitative efforts on the resuscitation record means that the rhythms we used may have reflected physiology after interventions had already commenced. Thus, in some situations, minute 1, 2, or earlier minutes may more accurately reflect the preintervention rhythm. Highly accurate time synchronization should be a central component of future prospective work in this area.
CONCLUSIONS
The FDR had only fair agreement with the telemetry rhythm at the time of the code blue call. Among those with VTA or asystole documented on CPR initiation, telemetry often revealed other organized rhythms present at the time hospital staff recognized a life‐threatening condition. In contrast to out‐of‐hospital cardiac arrest, FDR of asystole was only rarely preceded by VTA, and FDR of VTA was often preceded by an organized rhythm.[8, 11] Future studies should examine antecedent rhythms in combination with respiratory and perfusion status to more precisely determine arrest etiology.
Acknowledgments
The authors thank the staff at Flex Monitoring at Christiana Care Health System for their vital contributions to the study.
Disclosures
Dr. Zubrow had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. The authors report no conflicts of interest.
- , , , et al. First documented rhythm and clinical outcome from in‐hospital cardiac arrest among children and adults. JAMA. 2006;295(1):50–57.
- Get With The Guidelines–Resuscitation (GWTG‐R) overview. Available at: http://www.heart.org/HEARTORG/HealthcareResearch/GetWithTheGuidelines‐Resuscitation/Get‐With‐The‐Guidelines‐ResuscitationOverview_UCM_314497_Article.jsp. Accessed May 8, 2012.
- , , , et al. Recommended guidelines for reviewing, reporting, and conducting research on in‐hospital resuscitation: the in‐hospital “Utstein Style”. Circulation. 1997;95:2213–2239.
- , , , et al. Cardiopulmonary resuscitation of adults in the hospital: a report of 14,720 cardiac arrests from the National Registry of Cardiopulmonary Resuscitation. Resuscitation. 2003;58:297–308.
- , . The measurement of observer agreement for categorical data. Biometrics. 1977;33(1):159–174.
- , , , et al. Characteristics and outcome among patients suffering in‐hospital cardiac arrest in monitored and nonmonitored areas. Resuscitation. 2001;48:125–135.
- , , , et al. A comparison between patients suffering in‐hospital and out‐of hospital cardiac arrest in terms of treatment and outcome. J Intern Med. 2000;248:53–60.
- , , , et al. Cardiac arrest outside and inside hospital in a community: mechanisms behind the differences in outcomes and outcome in relation to time of arrest. Am Heart J. 2010;159:749–756.
- , , , et al.; Resuscitation Outcomes Consortium Investigators. Ventricular tachyarrhythmias after cardiac arrest in public versus at home. N Engl J Med. 2011;364:313–321.
- , . In‐hospital cardiac arrest. Emerg Med Clin North Am. 2012;30:25–34.
- , , , et al. Analysis of initial rhythm, witnessed status and delay to treatment among survivors of out‐of‐hospital cardiac arrest in Sweden. Heart. 2010;96:1826–1830.
In‐hospital cardiac arrest (IHCA) research often relies on the first documented cardiac rhythm (FDR) on resuscitation records at the time of cardiopulmonary resuscitation (CPR) initiation as a surrogate for arrest etiology.[1] Over 1000 hospitals report the FDR and associated cardiac arrest data to national registries annually.[2, 3] These data are subsequently used to report national IHCA epidemiology, as well as to develop and refine guidelines for in‐hospital resuscitation.[4]
Suspecting that the FDR might represent the later stage of a progressive cardiopulmonary process rather than a sudden dysrhythmia, we sought to compare the first rhythm documented on resuscitation records at the time of CPR initiation with the telemetry rhythm at the time of the code blue call. We hypothesized that the agreement between FDR and telemetry rhythm would be 80% beyond that predicted by chance (kappa0.8).[5]
METHODS
Design
Between June 2008 and February 2010, we performed a cross‐sectional study at a 750‐bed adult tertiary care hospital (Christiana Hospital) and a 240‐bed adult inner city community hospital (Wilmington Hospital). Both hospitals included teaching and nonteaching inpatient services. The Christiana Care Health System Institutional Review Board approved the study.
Study Population
Eligible subjects included a convenience sample of adult inpatients aged 18 years who were monitored on the hospital's telemetry system during the 2 minutes prior to a code blue call from a nonintensive care, noncardiac care inpatient ward for IHCA. Intensive care unit (ICU) locations were excluded because they are not captured in our central telemetry recording system. We defined IHCA as a resuscitation event requiring >1 minute of chest compressions and/or defibrillation. We excluded patients with do not attempt resuscitation orders at the time of the IHCA. For patients with multiple IHCAs, only their first event was included in the analysis. International Classification of Diseases, 9th Revision admission diagnoses were categorized into infectious, oncology, endocrine/metabolic; cardiovascular, renal, or other disease categories. The decision to place patients on telemetry monitoring was not part of the study and was entirely at the discretion of the physicians caring for the patients.
Variables and Measurements
We reviewed the paper resuscitation records of each IHCA during the study period and identified the FDR. To create groups that would allow comparison between telemetry and resuscitation record rhythms, we placed each rhythm into 1 of the following 3 categories: asystole, ventricular tachyarrhythmia (VTA), or other organized rhythms (Table 1). It was not possible to retrospectively ascertain the presence of pulses to determine if an organized rhythm identified on telemetry tracings was pulseless electrical activity (PEA) or a perfusing rhythm. Therefore, we elected to take a conservative approach that would bias toward agreement (the opposite direction of our hypothesis that the rhythms are discrepant) and consider all other organized rhythms in agreement with one another. We reviewed printouts of telemetry electrocardiographic records for each patient. Minute 0 was defined as the time of the code blue call. Two physician investigators (C.C. and U.B.) independently reviewed telemetry data for each patient at minute 0 and the 2 minutes preceding the code blue call (minutes 1 and 2). Rhythms at each minute mark were assigned to 1 of the following categories according to the classification scheme in Table 1: asystole, VTA, or other organized rhythms. Leads off and uninterpretable telemetry were also noted. Discrepancies in rhythm categorization between reviewers were resolved by a third investigator (M.Z.) blinded to rhythm category assignment. We used the telemetry rhythm at minute 0 for analysis whenever possible. If the leads were off or the telemetry was uninterpretable at minute 0, we used minute 1. If minute 1 was also unusable, we used minute 2. If there were no usable data at minutes 0, 1, or 2, we excluded the patient.
| Category | Rhythm |
|---|---|
| Asystole | Asystole |
| Ventricular tachyarrhythmia | Ventricular fibrillation, ventricular tachycardia |
| Other organized rhythms | Atrial fibrillation, bradycardia, paced pulseless electrical activity, sinus, idioventricular, other |
Statistical Analysis
We determined the percent agreement between the resuscitation record rhythm category and the last interpretable telemetry rhythm category. We then calculated an unweighted kappa for the agreement between the resuscitation record rhythm category and the last interpretable telemetry rhythm category.
RESULTS
During the study period, there were 135 code blue calls for urgent assistance among telemetry‐monitored non‐ICU patients. Of the 135 calls, we excluded 4 events (3%) that did not meet the definition of IHCA, 9 events (7%) with missing or uninterpretable data, and 53 events (39%) with unobtainable data due to automatic purging from the telemetry server. Therefore, 69 events in 69 different patients remained for analysis. Twelve of the 69 included arrests that occurred at Wilmington Hospital and 57 at Christiana Hospital. The characteristics of the patients are shown in Table 2.
| n | % | |
|---|---|---|
| Age, y | ||
| 3039 | 1 | 1.4 |
| 4049 | 4 | 5.8 |
| 5059 | 11 | 15.9 |
| 6069 | 15 | 21.7 |
| 7079 | 16 | 23.2 |
| 8089 | 18 | 26.1 |
| 90+ | 4 | 5.8 |
| Sex | ||
| Male | 26 | 37.7 |
| Female | 43 | 62.3 |
| Race/ethnicity | ||
| White | 51 | 73.9 |
| Black | 17 | 24.6 |
| Hispanic | 1 | 1.4 |
| Admission body mass index | ||
| Underweight (18.5) | 3 | 4.3 |
| Normal (18.525) | 15 | 21.7 |
| Overweight (2530) 24 | 24 | 34.8 |
| Obese (3035) | 17 | 24.6 |
| Very obese (35) | 9 | 13.0 |
| Unknown | 1 | 1.4 |
| Admission diagnosis category | ||
| Infectious | 29 | 42.0 |
| Oncology | 4 | 5.8 |
| Endocrine/metabolic | 22 | 31.9 |
| Cardiovascular | 7 | 10.1 |
| Renal | 2 | 2.8 |
| Other | 5 | 7.2 |
Of the 69 arrests, we used the telemetry rhythm at minute 0 in 42 patients (61%), minute 1 in 22 patients (32%), and minute 2 in 5 patients (7%). Agreement between telemetry and FDR was 65% (kappa=0.37, 95% confidence interval: 0.17‐0.56) (Table 3). Agreement did not vary significantly by sex, race, hospital, weekday, time of day, or minute used in the analysis. Agreement was not associated with survival to hospital discharge.
| Telemetry | Resuscitation Record | |||
|---|---|---|---|---|
| Asystole | Ventricular Tachyarrhythmia | Other Organized Rhythms | Total | |
| ||||
| Asystole | 3 | 0 | 2 | 5 |
| Ventricular tachyarrhythmia | 1 | 12 | 8 | 21 |
| Other organized rhythms | 8 | 5 | 30 | 43 |
| Total | 12 | 17 | 40 | 69 |
Of the 69 IHCA events, the FDRs vs telemetry rhythms at the time of IHCA were: asystole 17% vs 7%, VTA 25% vs 31%, and other organized rhythms 58% vs 62%. Among the 12 events with FDR recorded as asystole, telemetry at the time of the code call was asystole in 3 (25%), VTA in 1 (8%), and other organized rhythms in 8 (67%). Among the 17 events with FDR recorded as VTA, telemetry at the time of the code call was VTA in 12 (71%) and other organized rhythms in 5 (29%). Among the 40 events with FDR recorded as other organized rhythms, telemetry at the time of the code call was asystole in 2 (5%), VTA in 8 (20%), and other organized rhythms in 30 (75%). Among the 8 patients with VTA on telemetry and other organized rhythms on the resuscitation record, the other organized rhythms were documented as PEA (n=6), sinus (n=1), and bradycardia (n=1). Of the 12 patients with VTA on telemetry and on the resuscitation record, 8 (67%) had ventricular tachycardia on telemetry. Four of the 8 (50%) who had ventricular tachycardia on telemetry had deteriorated into ventricular fibrillation by the time the FDR was recorded. Of the 4 who had ventricular fibrillation on telemetry, all had ventricular fibrillation as the FDR on the resuscitation record.
DISCUSSION
These results establish that FDRs often differ from the telemetry rhythms at the time of the code blue call. This is important because national registries such as the American Heart Association's Get with the GuidelinesResuscitation[2] database use the FDR as a surrogate for arrest etiology, and use their findings to report national IHCA outcomes as well as to develop and refine evidence‐based guidelines for in‐hospital resuscitation. Our findings suggest that using the FDR may be an oversimplification of the complex progression of cardiac rhythms that occurs in the periarrest period. Adding preceding telemetry rhythms to the data elements collected may shed additional light on etiology. Furthermore, our results demonstrate that, among adults with VTA or asystole documented upon arrival of the code blue team, other organized rhythms are often present at the time the staff recognized a life‐threatening condition and called for immediate assistance. This suggests that the VTA and asystole FDRs may represent the later stages of progressive cardiopulmonary processes. This is in contrast to out‐of‐hospital cardiac arrests typically attributed to sudden catastrophic dysrhythmias that often progress to asystole unless rapidly defibrillated.[6, 7, 8] Out‐of‐hospital and in‐hospital arrests are likely different (but overlapping) entities that might benefit from different resuscitation strategies.[9, 10] We hypothesize that, for a subset of these patients, progressive respiratory insufficiency and circulatory shockconditions classically associated more strongly with pediatric than adult IHCAmay have been directly responsible for the event.[1] If future research supports the concept that progressive respiratory insufficiency and circulatory shock are responsible for more adult IHCA than previously recognized, more robust monitoring may be indicated for a larger subset of adult patients hospitalized on general wards. This could include pulse oximetry (wave form can be a surrogate for perfusion), respiratory rate, and/or end‐tidal CO2 monitoring. In addition, if future research confirms that there is a greater distinction between in‐hospital and out‐of‐hospital cardiac arrest etiology, the expert panels that develop resuscitation guidelines should consider including setting of resuscitation as a branch point in future algorithms.
Our study had several limitations. First, the sample size was small due to uninterpretable rhythm strips, and for 39% of the total code events, the telemetry data had already been purged from the system by the time research staff attempted to retrieve it. Although we do not believe that there was any systematic bias to the data analyzed, the possibility cannot be completely excluded. Second, we were constrained by the inability to retrospectively ascertain the presence of pulses to determine if an organized rhythm identified on telemetry tracings was PEA. Thus, we categorized rhythms into large groups. Although this limited the granularity of the rhythm groups, it was a conservative approach that likely biased toward agreement (the opposite direction of our hypothesis). Third, the lack of perfect time synchronization between the telemetry system, wall clocks in the hospital, and wrist watches that may be referenced when documenting resuscitative efforts on the resuscitation record means that the rhythms we used may have reflected physiology after interventions had already commenced. Thus, in some situations, minute 1, 2, or earlier minutes may more accurately reflect the preintervention rhythm. Highly accurate time synchronization should be a central component of future prospective work in this area.
CONCLUSIONS
The FDR had only fair agreement with the telemetry rhythm at the time of the code blue call. Among those with VTA or asystole documented on CPR initiation, telemetry often revealed other organized rhythms present at the time hospital staff recognized a life‐threatening condition. In contrast to out‐of‐hospital cardiac arrest, FDR of asystole was only rarely preceded by VTA, and FDR of VTA was often preceded by an organized rhythm.[8, 11] Future studies should examine antecedent rhythms in combination with respiratory and perfusion status to more precisely determine arrest etiology.
Acknowledgments
The authors thank the staff at Flex Monitoring at Christiana Care Health System for their vital contributions to the study.
Disclosures
Dr. Zubrow had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. The authors report no conflicts of interest.
In‐hospital cardiac arrest (IHCA) research often relies on the first documented cardiac rhythm (FDR) on resuscitation records at the time of cardiopulmonary resuscitation (CPR) initiation as a surrogate for arrest etiology.[1] Over 1000 hospitals report the FDR and associated cardiac arrest data to national registries annually.[2, 3] These data are subsequently used to report national IHCA epidemiology, as well as to develop and refine guidelines for in‐hospital resuscitation.[4]
Suspecting that the FDR might represent the later stage of a progressive cardiopulmonary process rather than a sudden dysrhythmia, we sought to compare the first rhythm documented on resuscitation records at the time of CPR initiation with the telemetry rhythm at the time of the code blue call. We hypothesized that the agreement between FDR and telemetry rhythm would be 80% beyond that predicted by chance (kappa0.8).[5]
METHODS
Design
Between June 2008 and February 2010, we performed a cross‐sectional study at a 750‐bed adult tertiary care hospital (Christiana Hospital) and a 240‐bed adult inner city community hospital (Wilmington Hospital). Both hospitals included teaching and nonteaching inpatient services. The Christiana Care Health System Institutional Review Board approved the study.
Study Population
Eligible subjects included a convenience sample of adult inpatients aged 18 years who were monitored on the hospital's telemetry system during the 2 minutes prior to a code blue call from a nonintensive care, noncardiac care inpatient ward for IHCA. Intensive care unit (ICU) locations were excluded because they are not captured in our central telemetry recording system. We defined IHCA as a resuscitation event requiring >1 minute of chest compressions and/or defibrillation. We excluded patients with do not attempt resuscitation orders at the time of the IHCA. For patients with multiple IHCAs, only their first event was included in the analysis. International Classification of Diseases, 9th Revision admission diagnoses were categorized into infectious, oncology, endocrine/metabolic; cardiovascular, renal, or other disease categories. The decision to place patients on telemetry monitoring was not part of the study and was entirely at the discretion of the physicians caring for the patients.
Variables and Measurements
We reviewed the paper resuscitation records of each IHCA during the study period and identified the FDR. To create groups that would allow comparison between telemetry and resuscitation record rhythms, we placed each rhythm into 1 of the following 3 categories: asystole, ventricular tachyarrhythmia (VTA), or other organized rhythms (Table 1). It was not possible to retrospectively ascertain the presence of pulses to determine if an organized rhythm identified on telemetry tracings was pulseless electrical activity (PEA) or a perfusing rhythm. Therefore, we elected to take a conservative approach that would bias toward agreement (the opposite direction of our hypothesis that the rhythms are discrepant) and consider all other organized rhythms in agreement with one another. We reviewed printouts of telemetry electrocardiographic records for each patient. Minute 0 was defined as the time of the code blue call. Two physician investigators (C.C. and U.B.) independently reviewed telemetry data for each patient at minute 0 and the 2 minutes preceding the code blue call (minutes 1 and 2). Rhythms at each minute mark were assigned to 1 of the following categories according to the classification scheme in Table 1: asystole, VTA, or other organized rhythms. Leads off and uninterpretable telemetry were also noted. Discrepancies in rhythm categorization between reviewers were resolved by a third investigator (M.Z.) blinded to rhythm category assignment. We used the telemetry rhythm at minute 0 for analysis whenever possible. If the leads were off or the telemetry was uninterpretable at minute 0, we used minute 1. If minute 1 was also unusable, we used minute 2. If there were no usable data at minutes 0, 1, or 2, we excluded the patient.
| Category | Rhythm |
|---|---|
| Asystole | Asystole |
| Ventricular tachyarrhythmia | Ventricular fibrillation, ventricular tachycardia |
| Other organized rhythms | Atrial fibrillation, bradycardia, paced pulseless electrical activity, sinus, idioventricular, other |
Statistical Analysis
We determined the percent agreement between the resuscitation record rhythm category and the last interpretable telemetry rhythm category. We then calculated an unweighted kappa for the agreement between the resuscitation record rhythm category and the last interpretable telemetry rhythm category.
RESULTS
During the study period, there were 135 code blue calls for urgent assistance among telemetry‐monitored non‐ICU patients. Of the 135 calls, we excluded 4 events (3%) that did not meet the definition of IHCA, 9 events (7%) with missing or uninterpretable data, and 53 events (39%) with unobtainable data due to automatic purging from the telemetry server. Therefore, 69 events in 69 different patients remained for analysis. Twelve of the 69 included arrests that occurred at Wilmington Hospital and 57 at Christiana Hospital. The characteristics of the patients are shown in Table 2.
| n | % | |
|---|---|---|
| Age, y | ||
| 3039 | 1 | 1.4 |
| 4049 | 4 | 5.8 |
| 5059 | 11 | 15.9 |
| 6069 | 15 | 21.7 |
| 7079 | 16 | 23.2 |
| 8089 | 18 | 26.1 |
| 90+ | 4 | 5.8 |
| Sex | ||
| Male | 26 | 37.7 |
| Female | 43 | 62.3 |
| Race/ethnicity | ||
| White | 51 | 73.9 |
| Black | 17 | 24.6 |
| Hispanic | 1 | 1.4 |
| Admission body mass index | ||
| Underweight (18.5) | 3 | 4.3 |
| Normal (18.525) | 15 | 21.7 |
| Overweight (2530) 24 | 24 | 34.8 |
| Obese (3035) | 17 | 24.6 |
| Very obese (35) | 9 | 13.0 |
| Unknown | 1 | 1.4 |
| Admission diagnosis category | ||
| Infectious | 29 | 42.0 |
| Oncology | 4 | 5.8 |
| Endocrine/metabolic | 22 | 31.9 |
| Cardiovascular | 7 | 10.1 |
| Renal | 2 | 2.8 |
| Other | 5 | 7.2 |
Of the 69 arrests, we used the telemetry rhythm at minute 0 in 42 patients (61%), minute 1 in 22 patients (32%), and minute 2 in 5 patients (7%). Agreement between telemetry and FDR was 65% (kappa=0.37, 95% confidence interval: 0.17‐0.56) (Table 3). Agreement did not vary significantly by sex, race, hospital, weekday, time of day, or minute used in the analysis. Agreement was not associated with survival to hospital discharge.
| Telemetry | Resuscitation Record | |||
|---|---|---|---|---|
| Asystole | Ventricular Tachyarrhythmia | Other Organized Rhythms | Total | |
| ||||
| Asystole | 3 | 0 | 2 | 5 |
| Ventricular tachyarrhythmia | 1 | 12 | 8 | 21 |
| Other organized rhythms | 8 | 5 | 30 | 43 |
| Total | 12 | 17 | 40 | 69 |
Of the 69 IHCA events, the FDRs vs telemetry rhythms at the time of IHCA were: asystole 17% vs 7%, VTA 25% vs 31%, and other organized rhythms 58% vs 62%. Among the 12 events with FDR recorded as asystole, telemetry at the time of the code call was asystole in 3 (25%), VTA in 1 (8%), and other organized rhythms in 8 (67%). Among the 17 events with FDR recorded as VTA, telemetry at the time of the code call was VTA in 12 (71%) and other organized rhythms in 5 (29%). Among the 40 events with FDR recorded as other organized rhythms, telemetry at the time of the code call was asystole in 2 (5%), VTA in 8 (20%), and other organized rhythms in 30 (75%). Among the 8 patients with VTA on telemetry and other organized rhythms on the resuscitation record, the other organized rhythms were documented as PEA (n=6), sinus (n=1), and bradycardia (n=1). Of the 12 patients with VTA on telemetry and on the resuscitation record, 8 (67%) had ventricular tachycardia on telemetry. Four of the 8 (50%) who had ventricular tachycardia on telemetry had deteriorated into ventricular fibrillation by the time the FDR was recorded. Of the 4 who had ventricular fibrillation on telemetry, all had ventricular fibrillation as the FDR on the resuscitation record.
DISCUSSION
These results establish that FDRs often differ from the telemetry rhythms at the time of the code blue call. This is important because national registries such as the American Heart Association's Get with the GuidelinesResuscitation[2] database use the FDR as a surrogate for arrest etiology, and use their findings to report national IHCA outcomes as well as to develop and refine evidence‐based guidelines for in‐hospital resuscitation. Our findings suggest that using the FDR may be an oversimplification of the complex progression of cardiac rhythms that occurs in the periarrest period. Adding preceding telemetry rhythms to the data elements collected may shed additional light on etiology. Furthermore, our results demonstrate that, among adults with VTA or asystole documented upon arrival of the code blue team, other organized rhythms are often present at the time the staff recognized a life‐threatening condition and called for immediate assistance. This suggests that the VTA and asystole FDRs may represent the later stages of progressive cardiopulmonary processes. This is in contrast to out‐of‐hospital cardiac arrests typically attributed to sudden catastrophic dysrhythmias that often progress to asystole unless rapidly defibrillated.[6, 7, 8] Out‐of‐hospital and in‐hospital arrests are likely different (but overlapping) entities that might benefit from different resuscitation strategies.[9, 10] We hypothesize that, for a subset of these patients, progressive respiratory insufficiency and circulatory shockconditions classically associated more strongly with pediatric than adult IHCAmay have been directly responsible for the event.[1] If future research supports the concept that progressive respiratory insufficiency and circulatory shock are responsible for more adult IHCA than previously recognized, more robust monitoring may be indicated for a larger subset of adult patients hospitalized on general wards. This could include pulse oximetry (wave form can be a surrogate for perfusion), respiratory rate, and/or end‐tidal CO2 monitoring. In addition, if future research confirms that there is a greater distinction between in‐hospital and out‐of‐hospital cardiac arrest etiology, the expert panels that develop resuscitation guidelines should consider including setting of resuscitation as a branch point in future algorithms.
Our study had several limitations. First, the sample size was small due to uninterpretable rhythm strips, and for 39% of the total code events, the telemetry data had already been purged from the system by the time research staff attempted to retrieve it. Although we do not believe that there was any systematic bias to the data analyzed, the possibility cannot be completely excluded. Second, we were constrained by the inability to retrospectively ascertain the presence of pulses to determine if an organized rhythm identified on telemetry tracings was PEA. Thus, we categorized rhythms into large groups. Although this limited the granularity of the rhythm groups, it was a conservative approach that likely biased toward agreement (the opposite direction of our hypothesis). Third, the lack of perfect time synchronization between the telemetry system, wall clocks in the hospital, and wrist watches that may be referenced when documenting resuscitative efforts on the resuscitation record means that the rhythms we used may have reflected physiology after interventions had already commenced. Thus, in some situations, minute 1, 2, or earlier minutes may more accurately reflect the preintervention rhythm. Highly accurate time synchronization should be a central component of future prospective work in this area.
CONCLUSIONS
The FDR had only fair agreement with the telemetry rhythm at the time of the code blue call. Among those with VTA or asystole documented on CPR initiation, telemetry often revealed other organized rhythms present at the time hospital staff recognized a life‐threatening condition. In contrast to out‐of‐hospital cardiac arrest, FDR of asystole was only rarely preceded by VTA, and FDR of VTA was often preceded by an organized rhythm.[8, 11] Future studies should examine antecedent rhythms in combination with respiratory and perfusion status to more precisely determine arrest etiology.
Acknowledgments
The authors thank the staff at Flex Monitoring at Christiana Care Health System for their vital contributions to the study.
Disclosures
Dr. Zubrow had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. The authors report no conflicts of interest.
- , , , et al. First documented rhythm and clinical outcome from in‐hospital cardiac arrest among children and adults. JAMA. 2006;295(1):50–57.
- Get With The Guidelines–Resuscitation (GWTG‐R) overview. Available at: http://www.heart.org/HEARTORG/HealthcareResearch/GetWithTheGuidelines‐Resuscitation/Get‐With‐The‐Guidelines‐ResuscitationOverview_UCM_314497_Article.jsp. Accessed May 8, 2012.
- , , , et al. Recommended guidelines for reviewing, reporting, and conducting research on in‐hospital resuscitation: the in‐hospital “Utstein Style”. Circulation. 1997;95:2213–2239.
- , , , et al. Cardiopulmonary resuscitation of adults in the hospital: a report of 14,720 cardiac arrests from the National Registry of Cardiopulmonary Resuscitation. Resuscitation. 2003;58:297–308.
- , . The measurement of observer agreement for categorical data. Biometrics. 1977;33(1):159–174.
- , , , et al. Characteristics and outcome among patients suffering in‐hospital cardiac arrest in monitored and nonmonitored areas. Resuscitation. 2001;48:125–135.
- , , , et al. A comparison between patients suffering in‐hospital and out‐of hospital cardiac arrest in terms of treatment and outcome. J Intern Med. 2000;248:53–60.
- , , , et al. Cardiac arrest outside and inside hospital in a community: mechanisms behind the differences in outcomes and outcome in relation to time of arrest. Am Heart J. 2010;159:749–756.
- , , , et al.; Resuscitation Outcomes Consortium Investigators. Ventricular tachyarrhythmias after cardiac arrest in public versus at home. N Engl J Med. 2011;364:313–321.
- , . In‐hospital cardiac arrest. Emerg Med Clin North Am. 2012;30:25–34.
- , , , et al. Analysis of initial rhythm, witnessed status and delay to treatment among survivors of out‐of‐hospital cardiac arrest in Sweden. Heart. 2010;96:1826–1830.
- , , , et al. First documented rhythm and clinical outcome from in‐hospital cardiac arrest among children and adults. JAMA. 2006;295(1):50–57.
- Get With The Guidelines–Resuscitation (GWTG‐R) overview. Available at: http://www.heart.org/HEARTORG/HealthcareResearch/GetWithTheGuidelines‐Resuscitation/Get‐With‐The‐Guidelines‐ResuscitationOverview_UCM_314497_Article.jsp. Accessed May 8, 2012.
- , , , et al. Recommended guidelines for reviewing, reporting, and conducting research on in‐hospital resuscitation: the in‐hospital “Utstein Style”. Circulation. 1997;95:2213–2239.
- , , , et al. Cardiopulmonary resuscitation of adults in the hospital: a report of 14,720 cardiac arrests from the National Registry of Cardiopulmonary Resuscitation. Resuscitation. 2003;58:297–308.
- , . The measurement of observer agreement for categorical data. Biometrics. 1977;33(1):159–174.
- , , , et al. Characteristics and outcome among patients suffering in‐hospital cardiac arrest in monitored and nonmonitored areas. Resuscitation. 2001;48:125–135.
- , , , et al. A comparison between patients suffering in‐hospital and out‐of hospital cardiac arrest in terms of treatment and outcome. J Intern Med. 2000;248:53–60.
- , , , et al. Cardiac arrest outside and inside hospital in a community: mechanisms behind the differences in outcomes and outcome in relation to time of arrest. Am Heart J. 2010;159:749–756.
- , , , et al.; Resuscitation Outcomes Consortium Investigators. Ventricular tachyarrhythmias after cardiac arrest in public versus at home. N Engl J Med. 2011;364:313–321.
- , . In‐hospital cardiac arrest. Emerg Med Clin North Am. 2012;30:25–34.
- , , , et al. Analysis of initial rhythm, witnessed status and delay to treatment among survivors of out‐of‐hospital cardiac arrest in Sweden. Heart. 2010;96:1826–1830.
Bacterial Contamination of Smart Phones
Mobile phones are now widely used. Healthcare workers, in particular, use them for rapid communication in many hospital settings. As mobile phones increase in popularity, a number of concerns have been raised, including noise and distraction in the clinical environment, confidentiality of patient information, and data security among others.[1]
Of the various concerns regarding mobile phone use in hospitals, one of the most important is that mobile phones may serve as vehicles for nosocomial transmission of micro‐organisms.[2, 3] One report showed that over 90% of healthcare workers' cell phones were contaminated with micro‐organisms, and 14.3% of cell phones were contaminated with bacteria that can cause nosocomial infection.[2]
Smart phones, which are rapidly flooding the mobile phone market, are useful in the hospital setting, as they could provide rapid access to medical information, quicker consultation and responding, feedback of results to the patient, and ongoing monitoring of chronic diseases (eg, asthma and diabetes).[4, 5, 6, 7, 8]
However, as most smart phones have wide, full, touch screens and are used more often by their owners than non‐smart phones are, bacterial contamination rates may be higher than those of non‐smart phones. The aim of this study was to compare the contamination rates by bacteria with pathogenic potential in smart phones versus non‐smart phones.
MATERIALS AND METHODS
Study Design and Participants
This cross‐sectional study was conducted from March 1, 2011 to June 30, 2011, in 3 teaching hospitals affiliated with Seoul National University School of Medicine, namely Seoul National University Hospital, Bundang Seoul National University Hospital, and Seoul National University Boramae Medical Center. Hospital staff working in general wards as well as in intensive care units of the 3 hospitals were invited to participate in this study. The study protocol was approved by the institutional review board of each of the 3 participating hospitals. Informed consent was obtained from all participants.
Questionnaire
We designed a questionnaire inquiring about demographics (age, gender, occupation) as well as behavior regarding cell phone use (type of cell phone, frequency and reasons for use, cleaning of cell phones).
Bacterial Culture, Identification, and Drug Susceptibility Testing
Both the anterior and posterior surfaces of each participant's mobile phone were touched onto blood agar plates. The sampled culture plates were subsequently incubated aerobically at 36C for 48 hours. To identify cultivated micro‐organisms and for the assessment of antibiotic susceptibility, VITEK2 (bioMrieux, Inc., Durham, NC) systems were used.
Classification of Isolated Micro‐organisms
We classified the micro‐organisms isolated from healthcare workers' mobile phones as bacteria with pathogenic potential (probable pathogens) or nonpathogens.[4, 9] Among probable pathogenic micro‐organisms, representative drug‐resistant strains such as methicillin‐resistant Staphylococcus aureus (MRSA), vancomycin‐resistant Enterococcus (VRE), and imipenem‐resistant Acinetobacter baumannii (IRAB) were categorized as drug‐resistant pathogens.
Classification of Smart Phones Versus Non‐Smart Phones
Mobile phones that ran complete mobile operating systems and software that provided a standardized interface and a platform for application developers, were classified as smart phones.[10] All others were classified as non‐smart phones.
Statistical Analysis
The participants' clinical variables were analyzed using descriptive statistics. The results are expressed as meanstandard deviation or median value with range. Variables were compared between the smart phone and non‐smart phone users. Categorical variables were compared by [2] analysis, and continuous variables were compared using Student t test or the Mann‐Whitney test. Variables with P0.20 after univariate analysis or clinically significant variables were subjected to multiple logistic regression to determine the risk factors for contamination of cell phones with potentially pathogenic bacteria. For all analyses, P values 0.05 were considered significant. Homer‐Lemeshow goodness of fit (GOF) test was performed to confirm the fitness of the final model. The Statistical Package for the Social Sciences version 17.0 (IBM SPSS, Armonk, NY) was used for all statistical analysis.
RESULTS
Participants and Their Behaviors Regarding Cell Phone Use
In total, 203 healthcare workers participated in this study; 80 (39.4%) were physicians, 106 (52.2%) were nurses, and 17 (8.4%) were assistants. The median age of the participants was 29 years, 43 (21.2%) were males, 115 (56.7%) participants used smart phones, and 88 (43.3%) were non‐smart phone users (Table 1).
| Smart Phone Users (N=115) | Non‐Smart Phone Users (N=88) | P Valuea | |
|---|---|---|---|
| |||
| Age, median (range), y | 28 (2048) | 29 (1952) | 0.03 |
| Gender, female | 92 (80.0%) | 68 (77.3%) | 0.64 |
| ICU workers | 78 (67.8%) | 57 (64.8%) | 0.65 |
| Occupation | 0.93 | ||
| Physicians | 45 (39.1%) | 35 (39.8%) | |
| Nurses | 63 (54.8%) | 43 (48.9%) | |
| Others | 7 (6.1%) | 10 (11.4%) | |
| Direct contact with patients | 110 (95.7%) | 84 (95.5%) | 0.95 |
| Using phones during work hours | 53 (46.1%) | 45 (51.1%) | 0.48 |
| Frequency of using phones during working | 0.46 | ||
| 13 | 8 (7.0%) | 10 (11.4%) | |
| 46 | 11 (9.6%) | 6 (6.8%) | |
| 79 | 8 (7.0%) | 1 (1.1%) | |
| Over 10 times | 26 (22.6%) | 28 (31.8%) | |
| None | 62 (53.9%) | 43 (48.9%) | |
| Reason of using phones | 0.04 | ||
| Calling | 30 (26.1%) | 42 (47.7%) | |
| Mail check or searching information | 3 (2.6%) | 0 | |
| Both reasons | 20 (17.4%) | 3 (3.4%) | |
| None | 62 (53.9%) | 43 (48.9%) | |
| Routine cleaning of phones | 15 (13.2%) | 11 (12.8%) | 0.94 |
| Frequency of cleaning hands (times/day) | 0.21 | ||
| 03 | 1 (0.9%) | 4 (4.5%) | |
| 46 | 18 (15.7%) | 14 (15.9%) | |
| 710 | 13 (11.3%) | 13 (14.8%) | |
| Over 10 | 83 (72.2%) | 57 (64.8%) | |
| Methods of cleaning hands | 0.72 | ||
| Washing with soaps | 48 (41.7%) | 34 (38.6%) | |
| Disinfectant | 42 (36.5%) | 33 (37.5%) | |
| Both | 25 (21.8%) | 21 (23.8%) | |
Smart phone users were slightly younger than non‐smart phone users. The distribution of occupations did not differ between the two groups. The frequency of use, reasons for using cell phones, the proportion of participants who routinely cleaned their phone, and the frequency of hand washing were also similar (Table 1).
Bacteria Isolated From Cell Phones
Bacteria were isolated from all 203 mobile phones; 3 or more different types of bacteria were isolated from 155 (76.4%) phones, 2 types from 39 (19.2%) phones, and 1 type from 9 (4.4%) phones. The most commonly cultured micro‐organism was coagulase‐negative Staphylococcus, which was isolated from 194 (95.6%) cell phones. The isolation of Gram‐positive bacilli and Micrococcus species was also frequent.
Probable pathogenic bacteria were isolated from 58 (28.6%) mobile phones. Among probable pathogens, Staphylococcus aureus (S. aureus) was the most commonly isolated. Of the 50 mobile phones that were contaminated with S. aureus, 8 were contaminated with a methicillin‐resistant strain. Five (2.4%) phones yielded Acinetobacter baumannii (Table 2).
| Organisms | Total, N=203 | No. of Drug Resistant Strains |
|---|---|---|
| Probable pathogen | ||
| Gram‐positive bacteria | ||
| Staphylococcus aureus | 50 (24.6%) | 8 (16%) |
| Streptococcus agalactiae | 1 (0.5%) | 0 |
| Enterococcus faecium | 1 (0.5%) | 1 (100%) |
| Gram‐negative bacteria | ||
| Acinetobacter baumannii | 5 (2.4%) | 1 (20%) |
| Pseudomonas aeruginosa | 1 (0.5%) | 0 |
| Enterobacter cloacae | 2 (1.0%) | 0 |
Although all mobile phones were contaminated with bacteria, probable pathogens were isolated more often from smart phones (34.8% vs 20.5% of non‐smart phones, P=0.03). The total colony count of probable pathogens from smart phones was also higher (average, 5.5 vs 5.0 from non‐smart phones, P=0.01). The isolation rate of drug‐resistant pathogens appeared to be higher from smart phones (7.0% vs 2.3% from non‐smart phones), but this difference did not reach statistical significance (P=0.19).
Risk Factors for Contamination With Probable Pathogens
In the final model constructed to determine the risk factors for contamination with probable pathogenic bacteria, data regarding cell phone users' age, gender, occupation (ie, physician or not), duration of working in the same place, daily work hours, whether the phone was a smart phone, and frequency of cell phone use during working hours were included. Among these factors, only the phone's being a smart phone was found to be a risk factor for contamination by bacteria with pathogenic potential (adjusted odds ratio (OR), 4.02; 95% CI, 1.43‐11.31; P=0.01). The fitness of this model was confirmed with the Hosmer‐Lemeshow GOF test (P=0.94). Using the cell phone more than 10 times during working hours appeared to be associated with pathogen contamination; however, this correlation failed to reach statistical significance (OR, 2.9; 95% CI, 0.9‐9.3; P=0.07).
DISCUSSION
Our study showed that smart phones were more frequently contaminated with bacteria than were non‐smart phones. In addition, total colony count of probable pathogens from smart phones was also higher. The colony count as well as contamination rate of pathogens are clinically relevant, because both factors can attribute to increased transmission of pathogens.[11]
Previous studies have attempted to identify user risk factors associated with bacterial contamination of cell phones.[12, 13, 14] Many variables, including gender, frequency of use, type of phone, work time, and the medical specialty of the user were considered; however, none of these factors was associated with an increased risk of bacterial contamination.[2, 14, 15]
In our study, none of the above‐mentioned factors was associated with contamination of cell phones by potentially pathogenic bacteria. Smart phones were the sole predictor of contamination by such bacteria. The reason that smart phones were more frequently contaminated with bacteria with pathogenic potential than were non‐smart phones is not clear. We propose two hypotheses to explain this observation. First, smart phones generally have wide screens, whereas non‐smart phones have relatively small screens with keypads. Larger screens may afford more opportunity for contamination by micro‐organisms. The mean size of a monitor in the smart phone group was 6633.2 340.1 cm2 and 5729.4564.7 cm2 in the non‐smart phone group (P0.01). However, in a multivariate model including size with other variables above, the smart phone remained a significant risk factor for the pathogen contamination (odds ratio [OR], 4.17; 95% CI, 1.06‐16.33; P=0.04). Cell phones are manufactured in a standardized form and the size cannot be changed or controlled. Therefore, we did not include the size in the final model of logistic regression in the main result. Our second explanation is in regard to the pattern of use of smart phones. Considering a single use, smart phones are used for longer periods and require a higher number of finger touches compared with non‐smart phones. The intensive use of phones with large screens could facilitate contamination of smart phones by pathogens from the healthcare workers' fingers or palms.
A recent study showed that cleaning cell phones on a daily basis decreased contamination rates. However, it did not decrease contamination by potentially pathogenic bacteria.[12] The role of the hospital environment as a reservoir of nosocomial pathogens and the effect of sanitization on decreasing clinical infection are still controversial.[16, 17, 18, 19] Thus, further studies are needed to recommend routine cell phone sanitizing and to declare that it is relevant in terms of reduction of hospital‐acquired infections potentially associated with the mobile phones of healthcare workers.
Our study is subject to limitation. Lack of association between hand washing and pathogen contamination might be a result of false reporting on hand washing behavior as well as the small number of participants. The bacterial contamination rate of the folding type of non‐smart phones may have been underestimated, as their keypads could not contact agar plates because they would not opened flatly (we touched the exterior surface of the folding type phones, which did not harbor the keypad, to the agar plate). However, given that folding phones are usually stored in their folded position, bacteria on the outside of the phones are likely more relevant than those within keypads insofar as transmission is concerned.
In summary, our data showed that over one‐fourth of the mobile phones examined in this study were found to harbor potentially pathogenic micro‐organisms. In particular, smart phones of healthcare workers were more frequently contaminated with potentially pathogenic bacteria than were non‐smart phones even after adjusting for the phone size. Preventive measures to minimize the possibility of bacterial transmission via cell phones should be devised.
Disclosure
This study was funded by grant 04‐2011‐1020 from the Seoul National University College of Medicine Research Fund (Seoul, South Korea). The sponsor of the study had no role in the tudy design, data collection, data analysis, data interpretation, or writing of the report. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication. The authors declare that no potential conflicts of interest exist with any companies/organizations whose products or services may be discussed in this article.
- , , , et al. Evidence‐based policy? The use of mobile phones in hospital. J Public Health (Oxf). 2006;28:299–303.
- , , , , . Is your phone bugged? The incidence of bacteria known to cause nosocomial infection on healthcare workers' mobile phones. J Hosp Infect. 2006;62:123–125.
- , , , , . Bacterial contamination of mobile communication devices in the operative environment. J Hosp Infect. 2007;66:397–398.
- , , , . Review of mobile communication devices as potential reservoirs of nosocomial pathogens. J Hosp Infect. 2009;71:295–300.
- , , . Use of SMS text messaging to improve outpatient attendance. Med J Aust. 2005;183:366–368.
- , , , et al. The use of text messaging to improve attendance in primary care: a randomized controlled trial. Fam Pract. 2006;23:699–705.
- , , , . Mobile phone text messaging in the management of diabetes. J Telemed Telecare. 2004;10:282–285.
- , , , , . Mobile phone text messaging can help young people manage asthma. BMJ. 2002;325:600.
- , , , et al. Use of cellular telephones and transmission of pathogens by medical staff in New York and Israel. Infect Control Hosp Epidemiol. 2007;28:500–503.
- Feature phone. Phone Scoop Web site. Available at: http://www.phonescoop.com/glossary/term.php?gid=310. Accessed June 22, 2011.
- , , . Comparison of two possible routes of pathogen contamination of spinach leaves in a hydroponic cultivation system. J Food Prot 2011;74:1536–1542.
- , , , et al. Use of mobile phones by medical staff at Queen Elizabeth Hospital, Barbados: evidence for both benefit and harm. J Hosp Infect. 2008;70:160–165.
- , , , . Pathogenic bacteria on personal pagers. Am J Infect Control. 2000;28:387–388.
- , , , , , . Bacterial contamination of health care workers' pagers and the efficacy of various disinfecting agents. Pediatr Infect Dis J. 2006;25: 1074–1075.
- , , . Preventing transmission of multidrug‐resistant bacteria in health care settings: a tale of 2 guidelines. Clin Infect Dis. 2006;42:828–835.
- . Environmental contamination makes an important contribution to hospital infection. J Hosp Infect. 2007;65(suppl 2):50–54.
- , , , . Widespread environmental contamination associated with patients with diarrhea and methicillin‐resistant Staphylococcus aureus colonization of the gastrointestinal tract. Infect Control Hosp Epidemiol. 2007;28:1142–1147.
- , , , , . Computer keyboards and faucet handles as reservoirs of nosocomial pathogens in the intensive care unit. Am J Infect Control. 2000;28:465–471.
- , , , , , . Routine disinfection of patients' environmental surfaces. Myth or reality? J Hosp Infect. 1999;42:113–117.
Mobile phones are now widely used. Healthcare workers, in particular, use them for rapid communication in many hospital settings. As mobile phones increase in popularity, a number of concerns have been raised, including noise and distraction in the clinical environment, confidentiality of patient information, and data security among others.[1]
Of the various concerns regarding mobile phone use in hospitals, one of the most important is that mobile phones may serve as vehicles for nosocomial transmission of micro‐organisms.[2, 3] One report showed that over 90% of healthcare workers' cell phones were contaminated with micro‐organisms, and 14.3% of cell phones were contaminated with bacteria that can cause nosocomial infection.[2]
Smart phones, which are rapidly flooding the mobile phone market, are useful in the hospital setting, as they could provide rapid access to medical information, quicker consultation and responding, feedback of results to the patient, and ongoing monitoring of chronic diseases (eg, asthma and diabetes).[4, 5, 6, 7, 8]
However, as most smart phones have wide, full, touch screens and are used more often by their owners than non‐smart phones are, bacterial contamination rates may be higher than those of non‐smart phones. The aim of this study was to compare the contamination rates by bacteria with pathogenic potential in smart phones versus non‐smart phones.
MATERIALS AND METHODS
Study Design and Participants
This cross‐sectional study was conducted from March 1, 2011 to June 30, 2011, in 3 teaching hospitals affiliated with Seoul National University School of Medicine, namely Seoul National University Hospital, Bundang Seoul National University Hospital, and Seoul National University Boramae Medical Center. Hospital staff working in general wards as well as in intensive care units of the 3 hospitals were invited to participate in this study. The study protocol was approved by the institutional review board of each of the 3 participating hospitals. Informed consent was obtained from all participants.
Questionnaire
We designed a questionnaire inquiring about demographics (age, gender, occupation) as well as behavior regarding cell phone use (type of cell phone, frequency and reasons for use, cleaning of cell phones).
Bacterial Culture, Identification, and Drug Susceptibility Testing
Both the anterior and posterior surfaces of each participant's mobile phone were touched onto blood agar plates. The sampled culture plates were subsequently incubated aerobically at 36C for 48 hours. To identify cultivated micro‐organisms and for the assessment of antibiotic susceptibility, VITEK2 (bioMrieux, Inc., Durham, NC) systems were used.
Classification of Isolated Micro‐organisms
We classified the micro‐organisms isolated from healthcare workers' mobile phones as bacteria with pathogenic potential (probable pathogens) or nonpathogens.[4, 9] Among probable pathogenic micro‐organisms, representative drug‐resistant strains such as methicillin‐resistant Staphylococcus aureus (MRSA), vancomycin‐resistant Enterococcus (VRE), and imipenem‐resistant Acinetobacter baumannii (IRAB) were categorized as drug‐resistant pathogens.
Classification of Smart Phones Versus Non‐Smart Phones
Mobile phones that ran complete mobile operating systems and software that provided a standardized interface and a platform for application developers, were classified as smart phones.[10] All others were classified as non‐smart phones.
Statistical Analysis
The participants' clinical variables were analyzed using descriptive statistics. The results are expressed as meanstandard deviation or median value with range. Variables were compared between the smart phone and non‐smart phone users. Categorical variables were compared by [2] analysis, and continuous variables were compared using Student t test or the Mann‐Whitney test. Variables with P0.20 after univariate analysis or clinically significant variables were subjected to multiple logistic regression to determine the risk factors for contamination of cell phones with potentially pathogenic bacteria. For all analyses, P values 0.05 were considered significant. Homer‐Lemeshow goodness of fit (GOF) test was performed to confirm the fitness of the final model. The Statistical Package for the Social Sciences version 17.0 (IBM SPSS, Armonk, NY) was used for all statistical analysis.
RESULTS
Participants and Their Behaviors Regarding Cell Phone Use
In total, 203 healthcare workers participated in this study; 80 (39.4%) were physicians, 106 (52.2%) were nurses, and 17 (8.4%) were assistants. The median age of the participants was 29 years, 43 (21.2%) were males, 115 (56.7%) participants used smart phones, and 88 (43.3%) were non‐smart phone users (Table 1).
| Smart Phone Users (N=115) | Non‐Smart Phone Users (N=88) | P Valuea | |
|---|---|---|---|
| |||
| Age, median (range), y | 28 (2048) | 29 (1952) | 0.03 |
| Gender, female | 92 (80.0%) | 68 (77.3%) | 0.64 |
| ICU workers | 78 (67.8%) | 57 (64.8%) | 0.65 |
| Occupation | 0.93 | ||
| Physicians | 45 (39.1%) | 35 (39.8%) | |
| Nurses | 63 (54.8%) | 43 (48.9%) | |
| Others | 7 (6.1%) | 10 (11.4%) | |
| Direct contact with patients | 110 (95.7%) | 84 (95.5%) | 0.95 |
| Using phones during work hours | 53 (46.1%) | 45 (51.1%) | 0.48 |
| Frequency of using phones during working | 0.46 | ||
| 13 | 8 (7.0%) | 10 (11.4%) | |
| 46 | 11 (9.6%) | 6 (6.8%) | |
| 79 | 8 (7.0%) | 1 (1.1%) | |
| Over 10 times | 26 (22.6%) | 28 (31.8%) | |
| None | 62 (53.9%) | 43 (48.9%) | |
| Reason of using phones | 0.04 | ||
| Calling | 30 (26.1%) | 42 (47.7%) | |
| Mail check or searching information | 3 (2.6%) | 0 | |
| Both reasons | 20 (17.4%) | 3 (3.4%) | |
| None | 62 (53.9%) | 43 (48.9%) | |
| Routine cleaning of phones | 15 (13.2%) | 11 (12.8%) | 0.94 |
| Frequency of cleaning hands (times/day) | 0.21 | ||
| 03 | 1 (0.9%) | 4 (4.5%) | |
| 46 | 18 (15.7%) | 14 (15.9%) | |
| 710 | 13 (11.3%) | 13 (14.8%) | |
| Over 10 | 83 (72.2%) | 57 (64.8%) | |
| Methods of cleaning hands | 0.72 | ||
| Washing with soaps | 48 (41.7%) | 34 (38.6%) | |
| Disinfectant | 42 (36.5%) | 33 (37.5%) | |
| Both | 25 (21.8%) | 21 (23.8%) | |
Smart phone users were slightly younger than non‐smart phone users. The distribution of occupations did not differ between the two groups. The frequency of use, reasons for using cell phones, the proportion of participants who routinely cleaned their phone, and the frequency of hand washing were also similar (Table 1).
Bacteria Isolated From Cell Phones
Bacteria were isolated from all 203 mobile phones; 3 or more different types of bacteria were isolated from 155 (76.4%) phones, 2 types from 39 (19.2%) phones, and 1 type from 9 (4.4%) phones. The most commonly cultured micro‐organism was coagulase‐negative Staphylococcus, which was isolated from 194 (95.6%) cell phones. The isolation of Gram‐positive bacilli and Micrococcus species was also frequent.
Probable pathogenic bacteria were isolated from 58 (28.6%) mobile phones. Among probable pathogens, Staphylococcus aureus (S. aureus) was the most commonly isolated. Of the 50 mobile phones that were contaminated with S. aureus, 8 were contaminated with a methicillin‐resistant strain. Five (2.4%) phones yielded Acinetobacter baumannii (Table 2).
| Organisms | Total, N=203 | No. of Drug Resistant Strains |
|---|---|---|
| Probable pathogen | ||
| Gram‐positive bacteria | ||
| Staphylococcus aureus | 50 (24.6%) | 8 (16%) |
| Streptococcus agalactiae | 1 (0.5%) | 0 |
| Enterococcus faecium | 1 (0.5%) | 1 (100%) |
| Gram‐negative bacteria | ||
| Acinetobacter baumannii | 5 (2.4%) | 1 (20%) |
| Pseudomonas aeruginosa | 1 (0.5%) | 0 |
| Enterobacter cloacae | 2 (1.0%) | 0 |
Although all mobile phones were contaminated with bacteria, probable pathogens were isolated more often from smart phones (34.8% vs 20.5% of non‐smart phones, P=0.03). The total colony count of probable pathogens from smart phones was also higher (average, 5.5 vs 5.0 from non‐smart phones, P=0.01). The isolation rate of drug‐resistant pathogens appeared to be higher from smart phones (7.0% vs 2.3% from non‐smart phones), but this difference did not reach statistical significance (P=0.19).
Risk Factors for Contamination With Probable Pathogens
In the final model constructed to determine the risk factors for contamination with probable pathogenic bacteria, data regarding cell phone users' age, gender, occupation (ie, physician or not), duration of working in the same place, daily work hours, whether the phone was a smart phone, and frequency of cell phone use during working hours were included. Among these factors, only the phone's being a smart phone was found to be a risk factor for contamination by bacteria with pathogenic potential (adjusted odds ratio (OR), 4.02; 95% CI, 1.43‐11.31; P=0.01). The fitness of this model was confirmed with the Hosmer‐Lemeshow GOF test (P=0.94). Using the cell phone more than 10 times during working hours appeared to be associated with pathogen contamination; however, this correlation failed to reach statistical significance (OR, 2.9; 95% CI, 0.9‐9.3; P=0.07).
DISCUSSION
Our study showed that smart phones were more frequently contaminated with bacteria than were non‐smart phones. In addition, total colony count of probable pathogens from smart phones was also higher. The colony count as well as contamination rate of pathogens are clinically relevant, because both factors can attribute to increased transmission of pathogens.[11]
Previous studies have attempted to identify user risk factors associated with bacterial contamination of cell phones.[12, 13, 14] Many variables, including gender, frequency of use, type of phone, work time, and the medical specialty of the user were considered; however, none of these factors was associated with an increased risk of bacterial contamination.[2, 14, 15]
In our study, none of the above‐mentioned factors was associated with contamination of cell phones by potentially pathogenic bacteria. Smart phones were the sole predictor of contamination by such bacteria. The reason that smart phones were more frequently contaminated with bacteria with pathogenic potential than were non‐smart phones is not clear. We propose two hypotheses to explain this observation. First, smart phones generally have wide screens, whereas non‐smart phones have relatively small screens with keypads. Larger screens may afford more opportunity for contamination by micro‐organisms. The mean size of a monitor in the smart phone group was 6633.2 340.1 cm2 and 5729.4564.7 cm2 in the non‐smart phone group (P0.01). However, in a multivariate model including size with other variables above, the smart phone remained a significant risk factor for the pathogen contamination (odds ratio [OR], 4.17; 95% CI, 1.06‐16.33; P=0.04). Cell phones are manufactured in a standardized form and the size cannot be changed or controlled. Therefore, we did not include the size in the final model of logistic regression in the main result. Our second explanation is in regard to the pattern of use of smart phones. Considering a single use, smart phones are used for longer periods and require a higher number of finger touches compared with non‐smart phones. The intensive use of phones with large screens could facilitate contamination of smart phones by pathogens from the healthcare workers' fingers or palms.
A recent study showed that cleaning cell phones on a daily basis decreased contamination rates. However, it did not decrease contamination by potentially pathogenic bacteria.[12] The role of the hospital environment as a reservoir of nosocomial pathogens and the effect of sanitization on decreasing clinical infection are still controversial.[16, 17, 18, 19] Thus, further studies are needed to recommend routine cell phone sanitizing and to declare that it is relevant in terms of reduction of hospital‐acquired infections potentially associated with the mobile phones of healthcare workers.
Our study is subject to limitation. Lack of association between hand washing and pathogen contamination might be a result of false reporting on hand washing behavior as well as the small number of participants. The bacterial contamination rate of the folding type of non‐smart phones may have been underestimated, as their keypads could not contact agar plates because they would not opened flatly (we touched the exterior surface of the folding type phones, which did not harbor the keypad, to the agar plate). However, given that folding phones are usually stored in their folded position, bacteria on the outside of the phones are likely more relevant than those within keypads insofar as transmission is concerned.
In summary, our data showed that over one‐fourth of the mobile phones examined in this study were found to harbor potentially pathogenic micro‐organisms. In particular, smart phones of healthcare workers were more frequently contaminated with potentially pathogenic bacteria than were non‐smart phones even after adjusting for the phone size. Preventive measures to minimize the possibility of bacterial transmission via cell phones should be devised.
Disclosure
This study was funded by grant 04‐2011‐1020 from the Seoul National University College of Medicine Research Fund (Seoul, South Korea). The sponsor of the study had no role in the tudy design, data collection, data analysis, data interpretation, or writing of the report. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication. The authors declare that no potential conflicts of interest exist with any companies/organizations whose products or services may be discussed in this article.
Mobile phones are now widely used. Healthcare workers, in particular, use them for rapid communication in many hospital settings. As mobile phones increase in popularity, a number of concerns have been raised, including noise and distraction in the clinical environment, confidentiality of patient information, and data security among others.[1]
Of the various concerns regarding mobile phone use in hospitals, one of the most important is that mobile phones may serve as vehicles for nosocomial transmission of micro‐organisms.[2, 3] One report showed that over 90% of healthcare workers' cell phones were contaminated with micro‐organisms, and 14.3% of cell phones were contaminated with bacteria that can cause nosocomial infection.[2]
Smart phones, which are rapidly flooding the mobile phone market, are useful in the hospital setting, as they could provide rapid access to medical information, quicker consultation and responding, feedback of results to the patient, and ongoing monitoring of chronic diseases (eg, asthma and diabetes).[4, 5, 6, 7, 8]
However, as most smart phones have wide, full, touch screens and are used more often by their owners than non‐smart phones are, bacterial contamination rates may be higher than those of non‐smart phones. The aim of this study was to compare the contamination rates by bacteria with pathogenic potential in smart phones versus non‐smart phones.
MATERIALS AND METHODS
Study Design and Participants
This cross‐sectional study was conducted from March 1, 2011 to June 30, 2011, in 3 teaching hospitals affiliated with Seoul National University School of Medicine, namely Seoul National University Hospital, Bundang Seoul National University Hospital, and Seoul National University Boramae Medical Center. Hospital staff working in general wards as well as in intensive care units of the 3 hospitals were invited to participate in this study. The study protocol was approved by the institutional review board of each of the 3 participating hospitals. Informed consent was obtained from all participants.
Questionnaire
We designed a questionnaire inquiring about demographics (age, gender, occupation) as well as behavior regarding cell phone use (type of cell phone, frequency and reasons for use, cleaning of cell phones).
Bacterial Culture, Identification, and Drug Susceptibility Testing
Both the anterior and posterior surfaces of each participant's mobile phone were touched onto blood agar plates. The sampled culture plates were subsequently incubated aerobically at 36C for 48 hours. To identify cultivated micro‐organisms and for the assessment of antibiotic susceptibility, VITEK2 (bioMrieux, Inc., Durham, NC) systems were used.
Classification of Isolated Micro‐organisms
We classified the micro‐organisms isolated from healthcare workers' mobile phones as bacteria with pathogenic potential (probable pathogens) or nonpathogens.[4, 9] Among probable pathogenic micro‐organisms, representative drug‐resistant strains such as methicillin‐resistant Staphylococcus aureus (MRSA), vancomycin‐resistant Enterococcus (VRE), and imipenem‐resistant Acinetobacter baumannii (IRAB) were categorized as drug‐resistant pathogens.
Classification of Smart Phones Versus Non‐Smart Phones
Mobile phones that ran complete mobile operating systems and software that provided a standardized interface and a platform for application developers, were classified as smart phones.[10] All others were classified as non‐smart phones.
Statistical Analysis
The participants' clinical variables were analyzed using descriptive statistics. The results are expressed as meanstandard deviation or median value with range. Variables were compared between the smart phone and non‐smart phone users. Categorical variables were compared by [2] analysis, and continuous variables were compared using Student t test or the Mann‐Whitney test. Variables with P0.20 after univariate analysis or clinically significant variables were subjected to multiple logistic regression to determine the risk factors for contamination of cell phones with potentially pathogenic bacteria. For all analyses, P values 0.05 were considered significant. Homer‐Lemeshow goodness of fit (GOF) test was performed to confirm the fitness of the final model. The Statistical Package for the Social Sciences version 17.0 (IBM SPSS, Armonk, NY) was used for all statistical analysis.
RESULTS
Participants and Their Behaviors Regarding Cell Phone Use
In total, 203 healthcare workers participated in this study; 80 (39.4%) were physicians, 106 (52.2%) were nurses, and 17 (8.4%) were assistants. The median age of the participants was 29 years, 43 (21.2%) were males, 115 (56.7%) participants used smart phones, and 88 (43.3%) were non‐smart phone users (Table 1).
| Smart Phone Users (N=115) | Non‐Smart Phone Users (N=88) | P Valuea | |
|---|---|---|---|
| |||
| Age, median (range), y | 28 (2048) | 29 (1952) | 0.03 |
| Gender, female | 92 (80.0%) | 68 (77.3%) | 0.64 |
| ICU workers | 78 (67.8%) | 57 (64.8%) | 0.65 |
| Occupation | 0.93 | ||
| Physicians | 45 (39.1%) | 35 (39.8%) | |
| Nurses | 63 (54.8%) | 43 (48.9%) | |
| Others | 7 (6.1%) | 10 (11.4%) | |
| Direct contact with patients | 110 (95.7%) | 84 (95.5%) | 0.95 |
| Using phones during work hours | 53 (46.1%) | 45 (51.1%) | 0.48 |
| Frequency of using phones during working | 0.46 | ||
| 13 | 8 (7.0%) | 10 (11.4%) | |
| 46 | 11 (9.6%) | 6 (6.8%) | |
| 79 | 8 (7.0%) | 1 (1.1%) | |
| Over 10 times | 26 (22.6%) | 28 (31.8%) | |
| None | 62 (53.9%) | 43 (48.9%) | |
| Reason of using phones | 0.04 | ||
| Calling | 30 (26.1%) | 42 (47.7%) | |
| Mail check or searching information | 3 (2.6%) | 0 | |
| Both reasons | 20 (17.4%) | 3 (3.4%) | |
| None | 62 (53.9%) | 43 (48.9%) | |
| Routine cleaning of phones | 15 (13.2%) | 11 (12.8%) | 0.94 |
| Frequency of cleaning hands (times/day) | 0.21 | ||
| 03 | 1 (0.9%) | 4 (4.5%) | |
| 46 | 18 (15.7%) | 14 (15.9%) | |
| 710 | 13 (11.3%) | 13 (14.8%) | |
| Over 10 | 83 (72.2%) | 57 (64.8%) | |
| Methods of cleaning hands | 0.72 | ||
| Washing with soaps | 48 (41.7%) | 34 (38.6%) | |
| Disinfectant | 42 (36.5%) | 33 (37.5%) | |
| Both | 25 (21.8%) | 21 (23.8%) | |
Smart phone users were slightly younger than non‐smart phone users. The distribution of occupations did not differ between the two groups. The frequency of use, reasons for using cell phones, the proportion of participants who routinely cleaned their phone, and the frequency of hand washing were also similar (Table 1).
Bacteria Isolated From Cell Phones
Bacteria were isolated from all 203 mobile phones; 3 or more different types of bacteria were isolated from 155 (76.4%) phones, 2 types from 39 (19.2%) phones, and 1 type from 9 (4.4%) phones. The most commonly cultured micro‐organism was coagulase‐negative Staphylococcus, which was isolated from 194 (95.6%) cell phones. The isolation of Gram‐positive bacilli and Micrococcus species was also frequent.
Probable pathogenic bacteria were isolated from 58 (28.6%) mobile phones. Among probable pathogens, Staphylococcus aureus (S. aureus) was the most commonly isolated. Of the 50 mobile phones that were contaminated with S. aureus, 8 were contaminated with a methicillin‐resistant strain. Five (2.4%) phones yielded Acinetobacter baumannii (Table 2).
| Organisms | Total, N=203 | No. of Drug Resistant Strains |
|---|---|---|
| Probable pathogen | ||
| Gram‐positive bacteria | ||
| Staphylococcus aureus | 50 (24.6%) | 8 (16%) |
| Streptococcus agalactiae | 1 (0.5%) | 0 |
| Enterococcus faecium | 1 (0.5%) | 1 (100%) |
| Gram‐negative bacteria | ||
| Acinetobacter baumannii | 5 (2.4%) | 1 (20%) |
| Pseudomonas aeruginosa | 1 (0.5%) | 0 |
| Enterobacter cloacae | 2 (1.0%) | 0 |
Although all mobile phones were contaminated with bacteria, probable pathogens were isolated more often from smart phones (34.8% vs 20.5% of non‐smart phones, P=0.03). The total colony count of probable pathogens from smart phones was also higher (average, 5.5 vs 5.0 from non‐smart phones, P=0.01). The isolation rate of drug‐resistant pathogens appeared to be higher from smart phones (7.0% vs 2.3% from non‐smart phones), but this difference did not reach statistical significance (P=0.19).
Risk Factors for Contamination With Probable Pathogens
In the final model constructed to determine the risk factors for contamination with probable pathogenic bacteria, data regarding cell phone users' age, gender, occupation (ie, physician or not), duration of working in the same place, daily work hours, whether the phone was a smart phone, and frequency of cell phone use during working hours were included. Among these factors, only the phone's being a smart phone was found to be a risk factor for contamination by bacteria with pathogenic potential (adjusted odds ratio (OR), 4.02; 95% CI, 1.43‐11.31; P=0.01). The fitness of this model was confirmed with the Hosmer‐Lemeshow GOF test (P=0.94). Using the cell phone more than 10 times during working hours appeared to be associated with pathogen contamination; however, this correlation failed to reach statistical significance (OR, 2.9; 95% CI, 0.9‐9.3; P=0.07).
DISCUSSION
Our study showed that smart phones were more frequently contaminated with bacteria than were non‐smart phones. In addition, total colony count of probable pathogens from smart phones was also higher. The colony count as well as contamination rate of pathogens are clinically relevant, because both factors can attribute to increased transmission of pathogens.[11]
Previous studies have attempted to identify user risk factors associated with bacterial contamination of cell phones.[12, 13, 14] Many variables, including gender, frequency of use, type of phone, work time, and the medical specialty of the user were considered; however, none of these factors was associated with an increased risk of bacterial contamination.[2, 14, 15]
In our study, none of the above‐mentioned factors was associated with contamination of cell phones by potentially pathogenic bacteria. Smart phones were the sole predictor of contamination by such bacteria. The reason that smart phones were more frequently contaminated with bacteria with pathogenic potential than were non‐smart phones is not clear. We propose two hypotheses to explain this observation. First, smart phones generally have wide screens, whereas non‐smart phones have relatively small screens with keypads. Larger screens may afford more opportunity for contamination by micro‐organisms. The mean size of a monitor in the smart phone group was 6633.2 340.1 cm2 and 5729.4564.7 cm2 in the non‐smart phone group (P0.01). However, in a multivariate model including size with other variables above, the smart phone remained a significant risk factor for the pathogen contamination (odds ratio [OR], 4.17; 95% CI, 1.06‐16.33; P=0.04). Cell phones are manufactured in a standardized form and the size cannot be changed or controlled. Therefore, we did not include the size in the final model of logistic regression in the main result. Our second explanation is in regard to the pattern of use of smart phones. Considering a single use, smart phones are used for longer periods and require a higher number of finger touches compared with non‐smart phones. The intensive use of phones with large screens could facilitate contamination of smart phones by pathogens from the healthcare workers' fingers or palms.
A recent study showed that cleaning cell phones on a daily basis decreased contamination rates. However, it did not decrease contamination by potentially pathogenic bacteria.[12] The role of the hospital environment as a reservoir of nosocomial pathogens and the effect of sanitization on decreasing clinical infection are still controversial.[16, 17, 18, 19] Thus, further studies are needed to recommend routine cell phone sanitizing and to declare that it is relevant in terms of reduction of hospital‐acquired infections potentially associated with the mobile phones of healthcare workers.
Our study is subject to limitation. Lack of association between hand washing and pathogen contamination might be a result of false reporting on hand washing behavior as well as the small number of participants. The bacterial contamination rate of the folding type of non‐smart phones may have been underestimated, as their keypads could not contact agar plates because they would not opened flatly (we touched the exterior surface of the folding type phones, which did not harbor the keypad, to the agar plate). However, given that folding phones are usually stored in their folded position, bacteria on the outside of the phones are likely more relevant than those within keypads insofar as transmission is concerned.
In summary, our data showed that over one‐fourth of the mobile phones examined in this study were found to harbor potentially pathogenic micro‐organisms. In particular, smart phones of healthcare workers were more frequently contaminated with potentially pathogenic bacteria than were non‐smart phones even after adjusting for the phone size. Preventive measures to minimize the possibility of bacterial transmission via cell phones should be devised.
Disclosure
This study was funded by grant 04‐2011‐1020 from the Seoul National University College of Medicine Research Fund (Seoul, South Korea). The sponsor of the study had no role in the tudy design, data collection, data analysis, data interpretation, or writing of the report. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication. The authors declare that no potential conflicts of interest exist with any companies/organizations whose products or services may be discussed in this article.
- , , , et al. Evidence‐based policy? The use of mobile phones in hospital. J Public Health (Oxf). 2006;28:299–303.
- , , , , . Is your phone bugged? The incidence of bacteria known to cause nosocomial infection on healthcare workers' mobile phones. J Hosp Infect. 2006;62:123–125.
- , , , , . Bacterial contamination of mobile communication devices in the operative environment. J Hosp Infect. 2007;66:397–398.
- , , , . Review of mobile communication devices as potential reservoirs of nosocomial pathogens. J Hosp Infect. 2009;71:295–300.
- , , . Use of SMS text messaging to improve outpatient attendance. Med J Aust. 2005;183:366–368.
- , , , et al. The use of text messaging to improve attendance in primary care: a randomized controlled trial. Fam Pract. 2006;23:699–705.
- , , , . Mobile phone text messaging in the management of diabetes. J Telemed Telecare. 2004;10:282–285.
- , , , , . Mobile phone text messaging can help young people manage asthma. BMJ. 2002;325:600.
- , , , et al. Use of cellular telephones and transmission of pathogens by medical staff in New York and Israel. Infect Control Hosp Epidemiol. 2007;28:500–503.
- Feature phone. Phone Scoop Web site. Available at: http://www.phonescoop.com/glossary/term.php?gid=310. Accessed June 22, 2011.
- , , . Comparison of two possible routes of pathogen contamination of spinach leaves in a hydroponic cultivation system. J Food Prot 2011;74:1536–1542.
- , , , et al. Use of mobile phones by medical staff at Queen Elizabeth Hospital, Barbados: evidence for both benefit and harm. J Hosp Infect. 2008;70:160–165.
- , , , . Pathogenic bacteria on personal pagers. Am J Infect Control. 2000;28:387–388.
- , , , , , . Bacterial contamination of health care workers' pagers and the efficacy of various disinfecting agents. Pediatr Infect Dis J. 2006;25: 1074–1075.
- , , . Preventing transmission of multidrug‐resistant bacteria in health care settings: a tale of 2 guidelines. Clin Infect Dis. 2006;42:828–835.
- . Environmental contamination makes an important contribution to hospital infection. J Hosp Infect. 2007;65(suppl 2):50–54.
- , , , . Widespread environmental contamination associated with patients with diarrhea and methicillin‐resistant Staphylococcus aureus colonization of the gastrointestinal tract. Infect Control Hosp Epidemiol. 2007;28:1142–1147.
- , , , , . Computer keyboards and faucet handles as reservoirs of nosocomial pathogens in the intensive care unit. Am J Infect Control. 2000;28:465–471.
- , , , , , . Routine disinfection of patients' environmental surfaces. Myth or reality? J Hosp Infect. 1999;42:113–117.
- , , , et al. Evidence‐based policy? The use of mobile phones in hospital. J Public Health (Oxf). 2006;28:299–303.
- , , , , . Is your phone bugged? The incidence of bacteria known to cause nosocomial infection on healthcare workers' mobile phones. J Hosp Infect. 2006;62:123–125.
- , , , , . Bacterial contamination of mobile communication devices in the operative environment. J Hosp Infect. 2007;66:397–398.
- , , , . Review of mobile communication devices as potential reservoirs of nosocomial pathogens. J Hosp Infect. 2009;71:295–300.
- , , . Use of SMS text messaging to improve outpatient attendance. Med J Aust. 2005;183:366–368.
- , , , et al. The use of text messaging to improve attendance in primary care: a randomized controlled trial. Fam Pract. 2006;23:699–705.
- , , , . Mobile phone text messaging in the management of diabetes. J Telemed Telecare. 2004;10:282–285.
- , , , , . Mobile phone text messaging can help young people manage asthma. BMJ. 2002;325:600.
- , , , et al. Use of cellular telephones and transmission of pathogens by medical staff in New York and Israel. Infect Control Hosp Epidemiol. 2007;28:500–503.
- Feature phone. Phone Scoop Web site. Available at: http://www.phonescoop.com/glossary/term.php?gid=310. Accessed June 22, 2011.
- , , . Comparison of two possible routes of pathogen contamination of spinach leaves in a hydroponic cultivation system. J Food Prot 2011;74:1536–1542.
- , , , et al. Use of mobile phones by medical staff at Queen Elizabeth Hospital, Barbados: evidence for both benefit and harm. J Hosp Infect. 2008;70:160–165.
- , , , . Pathogenic bacteria on personal pagers. Am J Infect Control. 2000;28:387–388.
- , , , , , . Bacterial contamination of health care workers' pagers and the efficacy of various disinfecting agents. Pediatr Infect Dis J. 2006;25: 1074–1075.
- , , . Preventing transmission of multidrug‐resistant bacteria in health care settings: a tale of 2 guidelines. Clin Infect Dis. 2006;42:828–835.
- . Environmental contamination makes an important contribution to hospital infection. J Hosp Infect. 2007;65(suppl 2):50–54.
- , , , . Widespread environmental contamination associated with patients with diarrhea and methicillin‐resistant Staphylococcus aureus colonization of the gastrointestinal tract. Infect Control Hosp Epidemiol. 2007;28:1142–1147.
- , , , , . Computer keyboards and faucet handles as reservoirs of nosocomial pathogens in the intensive care unit. Am J Infect Control. 2000;28:465–471.
- , , , , , . Routine disinfection of patients' environmental surfaces. Myth or reality? J Hosp Infect. 1999;42:113–117.
Causes of Sudden Unexpected Death
The sudden unexpected death of a hospitalized patient is extremely distressing to the family and the healthcare team. It is also distressingly common. Over 200,000 treated cardiac arrests are estimated to occur each year in US hospital patients.1 Most of these patients die, and studies have shown that physicians often incorrectly diagnose the causes of death when autopsies are performed to determine the causes of death.2, 3 This study was undertaken to elucidate the causes of sudden unexpected death of adult hospital patients as determined by autopsy.
METHODS
One hundred seventy‐five consecutive cases with autopsies by the senior author (L.N.) of adult hospital patients in the University of Pittsburgh Medical Center (UPMC) Health System, who died within 1 hour after onset of symptoms and in which death was unexpected, were retrospectively analyzed. Patients under 18, or dead on arrival, or on comfort measures only, were excluded. The unexpectedness of the deaths in this series was determined by review of the medical record, usually confirmed by a pre‐autopsy discussion with a clinician (following UPMC policy), and by the fact that attempted resuscitation was carried out in all but a few cases. Patient age, sex, race, and causes of death were obtained from the autopsy report. The medical record was reviewed to determine if the patient was on cardiac monitoring at the time of death. The study was approved by the University of Pittsburgh Medical Center Committee for Oversight of Research Involving the Dead.
RESULTS
The 175 autopsies in this study included 98 male patients and 77 female patients. Their ages ranged from 19 to 95 years, with an average age of 63.8 years. Categorized by race, 139 were white, 34 black, 1 Hispanic, and 1 Filipino. The autopsies were done over a 14‐year period from 1992 to 2006, during which the autopsy rate gradually decreased from 19% to 8%. Seeking authorization for autopsy from family was the responsibility of clinicians.
The most common immediate cause of death was judged to be a cardiac arrhythmia, usually presumptive; this was the immediate cause of death in 58 (33.1%) cases, as shown in Table 1. Second most common was hemorrhage, which was the immediate cause of death in 38 (21.7%) cases, and third was pulmonary thromboembolism in 27 (15.4%) cases, as shown in Table 1. Other conditions judged to be the immediate cause of death were cardiogenic pulmonary edema/congestive heart failure in 13 cases (7.4%), sepsis in 11 cases (6.3%), pulmonary edema due to acute lung injury in 4 cases (3 with associated pneumonia), and acute respiratory failure due to pneumonia (in 3 cases), usual interstitial pneumonia (2), emphysema (1), chronic obstructive pulmonary disease (1), herpes simplex virus bronchitis (1), carbon dioxide narcosis (1), and undiagnosed massive metastatic pancreatic carcinoma infiltrating and pushing up the diaphragm (1). Miscellaneous other conditions judged to be the immediate cause of death were brain stem compression in 2 cases, aspiration in 2 cases, andin 1 case eachsubarachnoid hemorrhage, hemorrhagic cerebral infarction, fat embolism, amniotic fluid embolism, bilateral pneumothoraces, massive hemolysis, sickle cell vaso‐occlusive crisis, cardiopulmonary decompensation, cardiac tamponade (due to pericardial metastases), and shock and systemic inflammatory response syndrome (due to volvulus).
| Immediate Cause of Death | No. (n = 175) |
|---|---|
| Cardiac arrhythmia | 58 (33.1%) |
| With 75% coronary stenosis | 36 |
| With myocardial infarction | 31 |
| Remote | 23 |
| Acute or subacute | 13 |
| Hemorrhage | 38 (21.7%) |
| With endogenous coagulopathy | 13 |
| With oral or injected anticoagulation | 12 |
| With antiplatelet therapy | 6 |
| Site: Pericardial (tamponade) | 15 |
| Retroperitoneal | 6 |
| Airway | 4 |
| Gastrointestinal | 4 |
| Pleural space | 2 |
| Thoracoabdominal | 2 |
| Intra‐abdominal | 1 |
| Mediastinal | 1 |
| Nasopharyngeal | 1 |
| Retroperitoneal and pleural | 1 |
| Multi‐organ | 1 |
| Pulmonary thromboembolism | 27 (15.4%) |
The majority, 36 of the 58 patients (62%), with sudden death judged due to arrhythmias, had 75% or greater stenosis of 1 or more coronary arteries (average 2); 11 of these patients were on cardiac monitoring with their fatal arrhythmia displayed on the monitor, and 1 patient was wearing a Holter monitor at the time of her sudden death. The frequency of cardiac arrhythmia as a cause of sudden death did not change over the course of the 14 years of this study. Among the 31 of the 58 patients (53.4%) with histologically confirmed myocardial infarctions, 15 (25.9%) had a remote or subacute myocardial infarction without a history of myocardial infarction.
The most common underlying cause of death was severe coronary atherosclerosis, as shown in Table 2, but there were 14 patients whose underlying cause of death was a diverse group of other heart diseases. Five patients died postoperatively following heart surgery. One patient had a mitral valve papillary fibroelastoma, and another patient's arrhythmia was preceded by a right bundle branch block and a new first‐degree atrioventricular block attributable to mitral annular calcification. One patient with sickle cell disease had a myocardial bridge over a coronary artery. Three patients had heart transplants, 2 alcoholic cardiomyopathy, and 1 idiopathic dilated cardiomyopathy.
| Underlying Cause of Death | No. (%) n = 175 |
|---|---|
| Severe coronary atherosclerosis | 43 (24.6) |
| Neoplastic disease | 30 (17.1) |
| Various heart diseases (see text) | 14 (8.0) |
| Digestive system disorders | 14 (8.0) |
| Aortic aneurysm or dissection | 12 (6.9) |
| Chronic lung disease | 8 (4.6) |
| Infectious diseases | 7 (4.0) |
| Autoimmune diseases | 5 (2.9) |
| Diabetes mellitus | 4 (2.3) |
| Deep vein thrombosis | 3 (1.7) |
| Morbid obesity | 3 (1.7) |
| Other | 32 (18.3) |
The initial cardiac rhythm during attempted cardiopulmonary resuscitation was obtainable for 120 cases. As shown in Table 3, a higher proportion of patients judged to have died of arrhythmias had an initial rhythm of ventricular tachycardia or fibrillation than the proportion of those judged to have died of hemorrhage, pulmonary embolism, or other immediate causes of death.
| Immediate Cause of Death | Initial Cardiac Rhythm, No. (%) | ||||
|---|---|---|---|---|---|
| Ventricular Tachycardia/Fibrillation n = 28 | Bradycardia n = 33 | Pulseless Electrical Activity n = 21 | Asystole n = 31 | Total | |
| |||||
| Arrhythmia | 14 (50) | 6 (18) | 7 (33) | 16 (52) | 43 |
| Hemorrhage | 6 (21) | 10 (30) | 10 (48) | 6 (19) | 32 |
| Thromboembolism | 2 (7) | 8 (24) | 2 (10) | 2 (6) | 15* |
| Other | 6 (21) | 9 (27) | 2 (10) | 7 (23) | 30 |
DISCUSSION
The patients who had an initial cardiac rhythm of asystole during attempted resuscitation, and were judged to have died of an arrhythmia, most likely had ventricular tachycardia or some other arrhythmia before asystole. The majority of them were found in cardiac arrest at night, not on cardiac monitoring. In other studies, there has been an unexplained continuing decline in the prevalence of sudden cardiac arrest cases presenting with ventricular fibrillation and corresponding rise in the prevalence of pulseless electrical activity (PEA) arrests.4 The 7 patients who had an initial cardiac rhythm of PEA and were judged to have died of an arrhythmia were all, except 1, off cardiac monitoring. Four had myocardial infarctions, 3 heart block pre‐arrest, 1 postoperative intramyocardial hematoma, 1 myocardial metastatic melanoma, 1 acute heart transplant rejection, and 3 ventricular tachycardia or fibrillation following PEA, suggesting that PEA was a phase in the evolution of an arrhythmogenic death. The patients who had an initial cardiac rhythm of ventricular tachycardia or fibrillation, and were judged to have died of hemorrhage or pulmonary thromboembolism, generally had coronary artery disease. The hemorrhage or embolism presumably caused terminal myocardial ischemia, but the cardiac disease was judged to be a contributing cause of death, less important than the immediate cause.
The conclusion that a cardiac arrhythmia was the most common mechanism of death in this series fits with the conclusion that cardiac arrhythmias were the most common immediate cause of cardiac arrest, specifically of 49% of them, in a study of 14,720 cardiac arrests of adult inpatients.5 It is accepted by convention that the presence of 75% or greater cross‐sectional luminal narrowing of a coronary artery, even without thrombosis, can be a cause of sudden cardiac death.6 Cardiac arrhythmias commonly occur during myocardial ischemia prior to the irreversible necrosis of myocardial infarction.7 Fatal cardiac arrhythmias can also be caused by old myocardial infarctions.8 The finding that 15 of the patients in this study, 25.9% of the 58 whose sudden death was attributable to a cardiac arrhythmia, had a remote or subacute myocardial infarction without a history of myocardial infarction fits with evidence that 25%30% of myocardial infarctions are unrecognized.
Hemorrhage has been an underpublicized cause of unexpected sudden death in hospital patients. Intracranial hemorrhages, due to ruptured berry aneurysms, hypertension, tumors, or arteriovenous malformations, are well‐recognized causes of sudden death, but not specifically in hospital patients.9 In a Scottish series of 111 unexpected sudden deaths due to an acute abdomen in patients aged 70 or older, 24 died of acute gastrointestinal hemorrhage, but cases of ruptured abdominal aortic aneurysm were excluded from the study because they would have dominated the analysis.10 Retroperitoneal hemorrhage is a particularly insidious cause of sudden death. It commonly causes little or no pain, and proceeds asymptomatically until the patient reaches the limits of cardiopulmonary compensation, which can mask the hemodynamic effect of the bleeding until sudden death.
The limitations of this study include the presumptive nature of the arrhythmias in the majority of patients judged most likely to have died of arrhythmias, and a potential selection bias in the cases coming to autopsy. No data on the 81% to 92% of deaths not investigated by autopsy is available, so the possibility of some sort of selection bias in this case series cannot be excluded. The causes of death determined by autopsy also inevitably represent a judgment or opinion about causation (as opposed to mere correlation), just as the assessment of the causes of death without autopsy does, but autopsy adds the knowledge of conditions undiagnosed prior to death and the exclusion of some suspected diagnoses, substantially improving the unavoidably judgmental conclusion.
There are implications for preventing the sudden unexpected death of hospital patients from the results of this study. They suggest that more cardiac rhythm monitoring might be helpful.11 More prophylactic antiarrhythmic medication and automatic implanted cardiac defibrillators might also be helpful.12 Some UPMC intensivists believe that the sort of fatal arrhythmias seen in this study are caused by hypoxemia, which suggests that more pulse oximetry oxygen saturation monitoring might allow preventative intervention.13 More frequent and possibly automated monitoring of vital signs might provide early warning of hemorrhage or pulmonary embolization.14 Keeping a wide differential diagnosis is taught for resuscitation with an initial rhythm of PEA, but keeping a wide differential including hemorrhage in cases with other initial rhythms, especially bradycardia, for example, may be important. This study suggests the importance of keeping hemorrhage in the differential diagnosis of sudden unexpected cardiac arrest of hospital patients.
- , , , et al. Incidence of treated cardiac arrest in hospitalized patients in the United States. Crit Care Med. 2011;39:2401–2406.
- , , , . Discrepancies between clinical and autopsy diagnoses. A comparison of university, community and private autopsy practices. Am J Clin Pathol. 2008;129:102–109.
- , , , . The four horsemen: clinicopathological correlation in 407 hospital autopsies. Intern Med J. 2010;40:626–632.
- , , , et al. Factors associated with pulseless electric activity versus ventricular fibrillation: the Oregon sudden unexpected death study. Circulation. 2010;122(21):2116–2122.
- , , , et al. Cardiopulmonary resuscitation of adults in the hospital: a report of 14,720 cardiac arrests from the national registry of cardiopulmonary resuscitation. Resuscitation. 2003;58:297–308.
- , , . Coronary thrombosis: what's new? Pathol Case Rev. 2001;6:244–252.
- , , , . Myocardial ischemia and ventricular fibrillation: pathophysiology and clinical implications. Int J Cardiol. 2007;119:283–290.
- , , , . Sudden death in coronary artery disease: acute ischemia versus myocardial substrate. Circulation. 1997;96:3215–3223.
- , . Sudden unexplained death in adults. Curr Top Pathol. 2001;95:125–148.
- , , . Acute abdomen as a cause of death in sudden, unexpected deaths in the elderly. Scott Med J. 2006;52:20–23.
- . Survival after tachyarrhythmic arrest—what are we waiting for? N Engl J Med. 2008;358:77–79.
- , , , et al. Circumstances and outcomes of sudden unexpected death in patients with high‐risk myocardial infarction: implications for prevention. Circulation. 2011;123:2674–2680.
- , , , , , . Use of pulse oximetry to predict in‐hospital complications in normotensive patients with pulmonary embolism. Am J Med. 2003;115(3):203–208.
- , , , , , . Telemetry‐based vital sign monitoring for ambulatory hospital patients. Conf Proc IEEE Eng Med Biol Soc. 2009;2009:4650–4653.
The sudden unexpected death of a hospitalized patient is extremely distressing to the family and the healthcare team. It is also distressingly common. Over 200,000 treated cardiac arrests are estimated to occur each year in US hospital patients.1 Most of these patients die, and studies have shown that physicians often incorrectly diagnose the causes of death when autopsies are performed to determine the causes of death.2, 3 This study was undertaken to elucidate the causes of sudden unexpected death of adult hospital patients as determined by autopsy.
METHODS
One hundred seventy‐five consecutive cases with autopsies by the senior author (L.N.) of adult hospital patients in the University of Pittsburgh Medical Center (UPMC) Health System, who died within 1 hour after onset of symptoms and in which death was unexpected, were retrospectively analyzed. Patients under 18, or dead on arrival, or on comfort measures only, were excluded. The unexpectedness of the deaths in this series was determined by review of the medical record, usually confirmed by a pre‐autopsy discussion with a clinician (following UPMC policy), and by the fact that attempted resuscitation was carried out in all but a few cases. Patient age, sex, race, and causes of death were obtained from the autopsy report. The medical record was reviewed to determine if the patient was on cardiac monitoring at the time of death. The study was approved by the University of Pittsburgh Medical Center Committee for Oversight of Research Involving the Dead.
RESULTS
The 175 autopsies in this study included 98 male patients and 77 female patients. Their ages ranged from 19 to 95 years, with an average age of 63.8 years. Categorized by race, 139 were white, 34 black, 1 Hispanic, and 1 Filipino. The autopsies were done over a 14‐year period from 1992 to 2006, during which the autopsy rate gradually decreased from 19% to 8%. Seeking authorization for autopsy from family was the responsibility of clinicians.
The most common immediate cause of death was judged to be a cardiac arrhythmia, usually presumptive; this was the immediate cause of death in 58 (33.1%) cases, as shown in Table 1. Second most common was hemorrhage, which was the immediate cause of death in 38 (21.7%) cases, and third was pulmonary thromboembolism in 27 (15.4%) cases, as shown in Table 1. Other conditions judged to be the immediate cause of death were cardiogenic pulmonary edema/congestive heart failure in 13 cases (7.4%), sepsis in 11 cases (6.3%), pulmonary edema due to acute lung injury in 4 cases (3 with associated pneumonia), and acute respiratory failure due to pneumonia (in 3 cases), usual interstitial pneumonia (2), emphysema (1), chronic obstructive pulmonary disease (1), herpes simplex virus bronchitis (1), carbon dioxide narcosis (1), and undiagnosed massive metastatic pancreatic carcinoma infiltrating and pushing up the diaphragm (1). Miscellaneous other conditions judged to be the immediate cause of death were brain stem compression in 2 cases, aspiration in 2 cases, andin 1 case eachsubarachnoid hemorrhage, hemorrhagic cerebral infarction, fat embolism, amniotic fluid embolism, bilateral pneumothoraces, massive hemolysis, sickle cell vaso‐occlusive crisis, cardiopulmonary decompensation, cardiac tamponade (due to pericardial metastases), and shock and systemic inflammatory response syndrome (due to volvulus).
| Immediate Cause of Death | No. (n = 175) |
|---|---|
| Cardiac arrhythmia | 58 (33.1%) |
| With 75% coronary stenosis | 36 |
| With myocardial infarction | 31 |
| Remote | 23 |
| Acute or subacute | 13 |
| Hemorrhage | 38 (21.7%) |
| With endogenous coagulopathy | 13 |
| With oral or injected anticoagulation | 12 |
| With antiplatelet therapy | 6 |
| Site: Pericardial (tamponade) | 15 |
| Retroperitoneal | 6 |
| Airway | 4 |
| Gastrointestinal | 4 |
| Pleural space | 2 |
| Thoracoabdominal | 2 |
| Intra‐abdominal | 1 |
| Mediastinal | 1 |
| Nasopharyngeal | 1 |
| Retroperitoneal and pleural | 1 |
| Multi‐organ | 1 |
| Pulmonary thromboembolism | 27 (15.4%) |
The majority, 36 of the 58 patients (62%), with sudden death judged due to arrhythmias, had 75% or greater stenosis of 1 or more coronary arteries (average 2); 11 of these patients were on cardiac monitoring with their fatal arrhythmia displayed on the monitor, and 1 patient was wearing a Holter monitor at the time of her sudden death. The frequency of cardiac arrhythmia as a cause of sudden death did not change over the course of the 14 years of this study. Among the 31 of the 58 patients (53.4%) with histologically confirmed myocardial infarctions, 15 (25.9%) had a remote or subacute myocardial infarction without a history of myocardial infarction.
The most common underlying cause of death was severe coronary atherosclerosis, as shown in Table 2, but there were 14 patients whose underlying cause of death was a diverse group of other heart diseases. Five patients died postoperatively following heart surgery. One patient had a mitral valve papillary fibroelastoma, and another patient's arrhythmia was preceded by a right bundle branch block and a new first‐degree atrioventricular block attributable to mitral annular calcification. One patient with sickle cell disease had a myocardial bridge over a coronary artery. Three patients had heart transplants, 2 alcoholic cardiomyopathy, and 1 idiopathic dilated cardiomyopathy.
| Underlying Cause of Death | No. (%) n = 175 |
|---|---|
| Severe coronary atherosclerosis | 43 (24.6) |
| Neoplastic disease | 30 (17.1) |
| Various heart diseases (see text) | 14 (8.0) |
| Digestive system disorders | 14 (8.0) |
| Aortic aneurysm or dissection | 12 (6.9) |
| Chronic lung disease | 8 (4.6) |
| Infectious diseases | 7 (4.0) |
| Autoimmune diseases | 5 (2.9) |
| Diabetes mellitus | 4 (2.3) |
| Deep vein thrombosis | 3 (1.7) |
| Morbid obesity | 3 (1.7) |
| Other | 32 (18.3) |
The initial cardiac rhythm during attempted cardiopulmonary resuscitation was obtainable for 120 cases. As shown in Table 3, a higher proportion of patients judged to have died of arrhythmias had an initial rhythm of ventricular tachycardia or fibrillation than the proportion of those judged to have died of hemorrhage, pulmonary embolism, or other immediate causes of death.
| Immediate Cause of Death | Initial Cardiac Rhythm, No. (%) | ||||
|---|---|---|---|---|---|
| Ventricular Tachycardia/Fibrillation n = 28 | Bradycardia n = 33 | Pulseless Electrical Activity n = 21 | Asystole n = 31 | Total | |
| |||||
| Arrhythmia | 14 (50) | 6 (18) | 7 (33) | 16 (52) | 43 |
| Hemorrhage | 6 (21) | 10 (30) | 10 (48) | 6 (19) | 32 |
| Thromboembolism | 2 (7) | 8 (24) | 2 (10) | 2 (6) | 15* |
| Other | 6 (21) | 9 (27) | 2 (10) | 7 (23) | 30 |
DISCUSSION
The patients who had an initial cardiac rhythm of asystole during attempted resuscitation, and were judged to have died of an arrhythmia, most likely had ventricular tachycardia or some other arrhythmia before asystole. The majority of them were found in cardiac arrest at night, not on cardiac monitoring. In other studies, there has been an unexplained continuing decline in the prevalence of sudden cardiac arrest cases presenting with ventricular fibrillation and corresponding rise in the prevalence of pulseless electrical activity (PEA) arrests.4 The 7 patients who had an initial cardiac rhythm of PEA and were judged to have died of an arrhythmia were all, except 1, off cardiac monitoring. Four had myocardial infarctions, 3 heart block pre‐arrest, 1 postoperative intramyocardial hematoma, 1 myocardial metastatic melanoma, 1 acute heart transplant rejection, and 3 ventricular tachycardia or fibrillation following PEA, suggesting that PEA was a phase in the evolution of an arrhythmogenic death. The patients who had an initial cardiac rhythm of ventricular tachycardia or fibrillation, and were judged to have died of hemorrhage or pulmonary thromboembolism, generally had coronary artery disease. The hemorrhage or embolism presumably caused terminal myocardial ischemia, but the cardiac disease was judged to be a contributing cause of death, less important than the immediate cause.
The conclusion that a cardiac arrhythmia was the most common mechanism of death in this series fits with the conclusion that cardiac arrhythmias were the most common immediate cause of cardiac arrest, specifically of 49% of them, in a study of 14,720 cardiac arrests of adult inpatients.5 It is accepted by convention that the presence of 75% or greater cross‐sectional luminal narrowing of a coronary artery, even without thrombosis, can be a cause of sudden cardiac death.6 Cardiac arrhythmias commonly occur during myocardial ischemia prior to the irreversible necrosis of myocardial infarction.7 Fatal cardiac arrhythmias can also be caused by old myocardial infarctions.8 The finding that 15 of the patients in this study, 25.9% of the 58 whose sudden death was attributable to a cardiac arrhythmia, had a remote or subacute myocardial infarction without a history of myocardial infarction fits with evidence that 25%30% of myocardial infarctions are unrecognized.
Hemorrhage has been an underpublicized cause of unexpected sudden death in hospital patients. Intracranial hemorrhages, due to ruptured berry aneurysms, hypertension, tumors, or arteriovenous malformations, are well‐recognized causes of sudden death, but not specifically in hospital patients.9 In a Scottish series of 111 unexpected sudden deaths due to an acute abdomen in patients aged 70 or older, 24 died of acute gastrointestinal hemorrhage, but cases of ruptured abdominal aortic aneurysm were excluded from the study because they would have dominated the analysis.10 Retroperitoneal hemorrhage is a particularly insidious cause of sudden death. It commonly causes little or no pain, and proceeds asymptomatically until the patient reaches the limits of cardiopulmonary compensation, which can mask the hemodynamic effect of the bleeding until sudden death.
The limitations of this study include the presumptive nature of the arrhythmias in the majority of patients judged most likely to have died of arrhythmias, and a potential selection bias in the cases coming to autopsy. No data on the 81% to 92% of deaths not investigated by autopsy is available, so the possibility of some sort of selection bias in this case series cannot be excluded. The causes of death determined by autopsy also inevitably represent a judgment or opinion about causation (as opposed to mere correlation), just as the assessment of the causes of death without autopsy does, but autopsy adds the knowledge of conditions undiagnosed prior to death and the exclusion of some suspected diagnoses, substantially improving the unavoidably judgmental conclusion.
There are implications for preventing the sudden unexpected death of hospital patients from the results of this study. They suggest that more cardiac rhythm monitoring might be helpful.11 More prophylactic antiarrhythmic medication and automatic implanted cardiac defibrillators might also be helpful.12 Some UPMC intensivists believe that the sort of fatal arrhythmias seen in this study are caused by hypoxemia, which suggests that more pulse oximetry oxygen saturation monitoring might allow preventative intervention.13 More frequent and possibly automated monitoring of vital signs might provide early warning of hemorrhage or pulmonary embolization.14 Keeping a wide differential diagnosis is taught for resuscitation with an initial rhythm of PEA, but keeping a wide differential including hemorrhage in cases with other initial rhythms, especially bradycardia, for example, may be important. This study suggests the importance of keeping hemorrhage in the differential diagnosis of sudden unexpected cardiac arrest of hospital patients.
The sudden unexpected death of a hospitalized patient is extremely distressing to the family and the healthcare team. It is also distressingly common. Over 200,000 treated cardiac arrests are estimated to occur each year in US hospital patients.1 Most of these patients die, and studies have shown that physicians often incorrectly diagnose the causes of death when autopsies are performed to determine the causes of death.2, 3 This study was undertaken to elucidate the causes of sudden unexpected death of adult hospital patients as determined by autopsy.
METHODS
One hundred seventy‐five consecutive cases with autopsies by the senior author (L.N.) of adult hospital patients in the University of Pittsburgh Medical Center (UPMC) Health System, who died within 1 hour after onset of symptoms and in which death was unexpected, were retrospectively analyzed. Patients under 18, or dead on arrival, or on comfort measures only, were excluded. The unexpectedness of the deaths in this series was determined by review of the medical record, usually confirmed by a pre‐autopsy discussion with a clinician (following UPMC policy), and by the fact that attempted resuscitation was carried out in all but a few cases. Patient age, sex, race, and causes of death were obtained from the autopsy report. The medical record was reviewed to determine if the patient was on cardiac monitoring at the time of death. The study was approved by the University of Pittsburgh Medical Center Committee for Oversight of Research Involving the Dead.
RESULTS
The 175 autopsies in this study included 98 male patients and 77 female patients. Their ages ranged from 19 to 95 years, with an average age of 63.8 years. Categorized by race, 139 were white, 34 black, 1 Hispanic, and 1 Filipino. The autopsies were done over a 14‐year period from 1992 to 2006, during which the autopsy rate gradually decreased from 19% to 8%. Seeking authorization for autopsy from family was the responsibility of clinicians.
The most common immediate cause of death was judged to be a cardiac arrhythmia, usually presumptive; this was the immediate cause of death in 58 (33.1%) cases, as shown in Table 1. Second most common was hemorrhage, which was the immediate cause of death in 38 (21.7%) cases, and third was pulmonary thromboembolism in 27 (15.4%) cases, as shown in Table 1. Other conditions judged to be the immediate cause of death were cardiogenic pulmonary edema/congestive heart failure in 13 cases (7.4%), sepsis in 11 cases (6.3%), pulmonary edema due to acute lung injury in 4 cases (3 with associated pneumonia), and acute respiratory failure due to pneumonia (in 3 cases), usual interstitial pneumonia (2), emphysema (1), chronic obstructive pulmonary disease (1), herpes simplex virus bronchitis (1), carbon dioxide narcosis (1), and undiagnosed massive metastatic pancreatic carcinoma infiltrating and pushing up the diaphragm (1). Miscellaneous other conditions judged to be the immediate cause of death were brain stem compression in 2 cases, aspiration in 2 cases, andin 1 case eachsubarachnoid hemorrhage, hemorrhagic cerebral infarction, fat embolism, amniotic fluid embolism, bilateral pneumothoraces, massive hemolysis, sickle cell vaso‐occlusive crisis, cardiopulmonary decompensation, cardiac tamponade (due to pericardial metastases), and shock and systemic inflammatory response syndrome (due to volvulus).
| Immediate Cause of Death | No. (n = 175) |
|---|---|
| Cardiac arrhythmia | 58 (33.1%) |
| With 75% coronary stenosis | 36 |
| With myocardial infarction | 31 |
| Remote | 23 |
| Acute or subacute | 13 |
| Hemorrhage | 38 (21.7%) |
| With endogenous coagulopathy | 13 |
| With oral or injected anticoagulation | 12 |
| With antiplatelet therapy | 6 |
| Site: Pericardial (tamponade) | 15 |
| Retroperitoneal | 6 |
| Airway | 4 |
| Gastrointestinal | 4 |
| Pleural space | 2 |
| Thoracoabdominal | 2 |
| Intra‐abdominal | 1 |
| Mediastinal | 1 |
| Nasopharyngeal | 1 |
| Retroperitoneal and pleural | 1 |
| Multi‐organ | 1 |
| Pulmonary thromboembolism | 27 (15.4%) |
The majority, 36 of the 58 patients (62%), with sudden death judged due to arrhythmias, had 75% or greater stenosis of 1 or more coronary arteries (average 2); 11 of these patients were on cardiac monitoring with their fatal arrhythmia displayed on the monitor, and 1 patient was wearing a Holter monitor at the time of her sudden death. The frequency of cardiac arrhythmia as a cause of sudden death did not change over the course of the 14 years of this study. Among the 31 of the 58 patients (53.4%) with histologically confirmed myocardial infarctions, 15 (25.9%) had a remote or subacute myocardial infarction without a history of myocardial infarction.
The most common underlying cause of death was severe coronary atherosclerosis, as shown in Table 2, but there were 14 patients whose underlying cause of death was a diverse group of other heart diseases. Five patients died postoperatively following heart surgery. One patient had a mitral valve papillary fibroelastoma, and another patient's arrhythmia was preceded by a right bundle branch block and a new first‐degree atrioventricular block attributable to mitral annular calcification. One patient with sickle cell disease had a myocardial bridge over a coronary artery. Three patients had heart transplants, 2 alcoholic cardiomyopathy, and 1 idiopathic dilated cardiomyopathy.
| Underlying Cause of Death | No. (%) n = 175 |
|---|---|
| Severe coronary atherosclerosis | 43 (24.6) |
| Neoplastic disease | 30 (17.1) |
| Various heart diseases (see text) | 14 (8.0) |
| Digestive system disorders | 14 (8.0) |
| Aortic aneurysm or dissection | 12 (6.9) |
| Chronic lung disease | 8 (4.6) |
| Infectious diseases | 7 (4.0) |
| Autoimmune diseases | 5 (2.9) |
| Diabetes mellitus | 4 (2.3) |
| Deep vein thrombosis | 3 (1.7) |
| Morbid obesity | 3 (1.7) |
| Other | 32 (18.3) |
The initial cardiac rhythm during attempted cardiopulmonary resuscitation was obtainable for 120 cases. As shown in Table 3, a higher proportion of patients judged to have died of arrhythmias had an initial rhythm of ventricular tachycardia or fibrillation than the proportion of those judged to have died of hemorrhage, pulmonary embolism, or other immediate causes of death.
| Immediate Cause of Death | Initial Cardiac Rhythm, No. (%) | ||||
|---|---|---|---|---|---|
| Ventricular Tachycardia/Fibrillation n = 28 | Bradycardia n = 33 | Pulseless Electrical Activity n = 21 | Asystole n = 31 | Total | |
| |||||
| Arrhythmia | 14 (50) | 6 (18) | 7 (33) | 16 (52) | 43 |
| Hemorrhage | 6 (21) | 10 (30) | 10 (48) | 6 (19) | 32 |
| Thromboembolism | 2 (7) | 8 (24) | 2 (10) | 2 (6) | 15* |
| Other | 6 (21) | 9 (27) | 2 (10) | 7 (23) | 30 |
DISCUSSION
The patients who had an initial cardiac rhythm of asystole during attempted resuscitation, and were judged to have died of an arrhythmia, most likely had ventricular tachycardia or some other arrhythmia before asystole. The majority of them were found in cardiac arrest at night, not on cardiac monitoring. In other studies, there has been an unexplained continuing decline in the prevalence of sudden cardiac arrest cases presenting with ventricular fibrillation and corresponding rise in the prevalence of pulseless electrical activity (PEA) arrests.4 The 7 patients who had an initial cardiac rhythm of PEA and were judged to have died of an arrhythmia were all, except 1, off cardiac monitoring. Four had myocardial infarctions, 3 heart block pre‐arrest, 1 postoperative intramyocardial hematoma, 1 myocardial metastatic melanoma, 1 acute heart transplant rejection, and 3 ventricular tachycardia or fibrillation following PEA, suggesting that PEA was a phase in the evolution of an arrhythmogenic death. The patients who had an initial cardiac rhythm of ventricular tachycardia or fibrillation, and were judged to have died of hemorrhage or pulmonary thromboembolism, generally had coronary artery disease. The hemorrhage or embolism presumably caused terminal myocardial ischemia, but the cardiac disease was judged to be a contributing cause of death, less important than the immediate cause.
The conclusion that a cardiac arrhythmia was the most common mechanism of death in this series fits with the conclusion that cardiac arrhythmias were the most common immediate cause of cardiac arrest, specifically of 49% of them, in a study of 14,720 cardiac arrests of adult inpatients.5 It is accepted by convention that the presence of 75% or greater cross‐sectional luminal narrowing of a coronary artery, even without thrombosis, can be a cause of sudden cardiac death.6 Cardiac arrhythmias commonly occur during myocardial ischemia prior to the irreversible necrosis of myocardial infarction.7 Fatal cardiac arrhythmias can also be caused by old myocardial infarctions.8 The finding that 15 of the patients in this study, 25.9% of the 58 whose sudden death was attributable to a cardiac arrhythmia, had a remote or subacute myocardial infarction without a history of myocardial infarction fits with evidence that 25%30% of myocardial infarctions are unrecognized.
Hemorrhage has been an underpublicized cause of unexpected sudden death in hospital patients. Intracranial hemorrhages, due to ruptured berry aneurysms, hypertension, tumors, or arteriovenous malformations, are well‐recognized causes of sudden death, but not specifically in hospital patients.9 In a Scottish series of 111 unexpected sudden deaths due to an acute abdomen in patients aged 70 or older, 24 died of acute gastrointestinal hemorrhage, but cases of ruptured abdominal aortic aneurysm were excluded from the study because they would have dominated the analysis.10 Retroperitoneal hemorrhage is a particularly insidious cause of sudden death. It commonly causes little or no pain, and proceeds asymptomatically until the patient reaches the limits of cardiopulmonary compensation, which can mask the hemodynamic effect of the bleeding until sudden death.
The limitations of this study include the presumptive nature of the arrhythmias in the majority of patients judged most likely to have died of arrhythmias, and a potential selection bias in the cases coming to autopsy. No data on the 81% to 92% of deaths not investigated by autopsy is available, so the possibility of some sort of selection bias in this case series cannot be excluded. The causes of death determined by autopsy also inevitably represent a judgment or opinion about causation (as opposed to mere correlation), just as the assessment of the causes of death without autopsy does, but autopsy adds the knowledge of conditions undiagnosed prior to death and the exclusion of some suspected diagnoses, substantially improving the unavoidably judgmental conclusion.
There are implications for preventing the sudden unexpected death of hospital patients from the results of this study. They suggest that more cardiac rhythm monitoring might be helpful.11 More prophylactic antiarrhythmic medication and automatic implanted cardiac defibrillators might also be helpful.12 Some UPMC intensivists believe that the sort of fatal arrhythmias seen in this study are caused by hypoxemia, which suggests that more pulse oximetry oxygen saturation monitoring might allow preventative intervention.13 More frequent and possibly automated monitoring of vital signs might provide early warning of hemorrhage or pulmonary embolization.14 Keeping a wide differential diagnosis is taught for resuscitation with an initial rhythm of PEA, but keeping a wide differential including hemorrhage in cases with other initial rhythms, especially bradycardia, for example, may be important. This study suggests the importance of keeping hemorrhage in the differential diagnosis of sudden unexpected cardiac arrest of hospital patients.
- , , , et al. Incidence of treated cardiac arrest in hospitalized patients in the United States. Crit Care Med. 2011;39:2401–2406.
- , , , . Discrepancies between clinical and autopsy diagnoses. A comparison of university, community and private autopsy practices. Am J Clin Pathol. 2008;129:102–109.
- , , , . The four horsemen: clinicopathological correlation in 407 hospital autopsies. Intern Med J. 2010;40:626–632.
- , , , et al. Factors associated with pulseless electric activity versus ventricular fibrillation: the Oregon sudden unexpected death study. Circulation. 2010;122(21):2116–2122.
- , , , et al. Cardiopulmonary resuscitation of adults in the hospital: a report of 14,720 cardiac arrests from the national registry of cardiopulmonary resuscitation. Resuscitation. 2003;58:297–308.
- , , . Coronary thrombosis: what's new? Pathol Case Rev. 2001;6:244–252.
- , , , . Myocardial ischemia and ventricular fibrillation: pathophysiology and clinical implications. Int J Cardiol. 2007;119:283–290.
- , , , . Sudden death in coronary artery disease: acute ischemia versus myocardial substrate. Circulation. 1997;96:3215–3223.
- , . Sudden unexplained death in adults. Curr Top Pathol. 2001;95:125–148.
- , , . Acute abdomen as a cause of death in sudden, unexpected deaths in the elderly. Scott Med J. 2006;52:20–23.
- . Survival after tachyarrhythmic arrest—what are we waiting for? N Engl J Med. 2008;358:77–79.
- , , , et al. Circumstances and outcomes of sudden unexpected death in patients with high‐risk myocardial infarction: implications for prevention. Circulation. 2011;123:2674–2680.
- , , , , , . Use of pulse oximetry to predict in‐hospital complications in normotensive patients with pulmonary embolism. Am J Med. 2003;115(3):203–208.
- , , , , , . Telemetry‐based vital sign monitoring for ambulatory hospital patients. Conf Proc IEEE Eng Med Biol Soc. 2009;2009:4650–4653.
- , , , et al. Incidence of treated cardiac arrest in hospitalized patients in the United States. Crit Care Med. 2011;39:2401–2406.
- , , , . Discrepancies between clinical and autopsy diagnoses. A comparison of university, community and private autopsy practices. Am J Clin Pathol. 2008;129:102–109.
- , , , . The four horsemen: clinicopathological correlation in 407 hospital autopsies. Intern Med J. 2010;40:626–632.
- , , , et al. Factors associated with pulseless electric activity versus ventricular fibrillation: the Oregon sudden unexpected death study. Circulation. 2010;122(21):2116–2122.
- , , , et al. Cardiopulmonary resuscitation of adults in the hospital: a report of 14,720 cardiac arrests from the national registry of cardiopulmonary resuscitation. Resuscitation. 2003;58:297–308.
- , , . Coronary thrombosis: what's new? Pathol Case Rev. 2001;6:244–252.
- , , , . Myocardial ischemia and ventricular fibrillation: pathophysiology and clinical implications. Int J Cardiol. 2007;119:283–290.
- , , , . Sudden death in coronary artery disease: acute ischemia versus myocardial substrate. Circulation. 1997;96:3215–3223.
- , . Sudden unexplained death in adults. Curr Top Pathol. 2001;95:125–148.
- , , . Acute abdomen as a cause of death in sudden, unexpected deaths in the elderly. Scott Med J. 2006;52:20–23.
- . Survival after tachyarrhythmic arrest—what are we waiting for? N Engl J Med. 2008;358:77–79.
- , , , et al. Circumstances and outcomes of sudden unexpected death in patients with high‐risk myocardial infarction: implications for prevention. Circulation. 2011;123:2674–2680.
- , , , , , . Use of pulse oximetry to predict in‐hospital complications in normotensive patients with pulmonary embolism. Am J Med. 2003;115(3):203–208.
- , , , , , . Telemetry‐based vital sign monitoring for ambulatory hospital patients. Conf Proc IEEE Eng Med Biol Soc. 2009;2009:4650–4653.