User login
Combatting the cough that won’t quit
• Always include postnasal drip, asthma, and gastroesophageal reflux disease in the differential diagnosis for persistent cough, regardless of clinical signs and symptoms. B
• Do not rely on a patient’s description of the character and timing of the cough or the absence (or presence) of sputum to narrow down the differential diagnosis. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE Margaret M, a 52-year-old nonsmoker, came to our clinic because of a persistent cough that had started about 4 weeks earlier. She had tried multiple over-the-counter cough suppressants, including dextromethorphan and guaifenesin, as well as cough drops, but none had been effective.
Margaret denied having had a cold or respiratory infection in the past few months or being in close contact with anyone with a chronic cough, and she had never had an asthma diagnosis. In response to a question about previous coughing episodes, the patient recalled having had several bouts of chronic cough in the past, including one about a year ago.
While Margaret had no known allergies, she did have occasional heartburn, which an antacid—or, at times, a drink of water—always relieved. Thyroid medication and calcium were the only things she took on a regular basis, separated by several hours to avoid problems with absorption.
Patients like Margaret, who seek help from their primary care physician only after attempting to combat a persistent cough on their own, may be quite frustrated by the time they arrive in your office. They’re counting on you to provide a cure. Fortunately, you’re likely to find it, as the differential diagnosis for subacute cough (a cough of 3-8 weeks’ duration) is limited.
Nonetheless, finding the cause of a subacute or chronic cough (lasting >8 weeks) is sometimes a matter of trial and error. Postnasal drip (also known as upper airway cough syndrome, or UACS), asthma, and gastroesophageal reflux disease (GERD) are the most common causes,1,2 followed by postinfectious cough, nonasthmatic eosinophilic bronchitis (NAEB), and pertussis.3 Although these conditions are all relatively well known, they are not always easy to detect: Some disorders, including UACS, asthma, and GERD, may be “silent,” with persistent cough the only presenting sign or symptom.4 In other cases, more than one condition may be contributing to the cough.

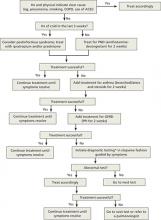
Starting with trials of empiric therapy for the most common causes of persistent cough—with sequential therapy and diagnostic tests, as needed—is far more effective than searching for relatively uncommon or obscure conditions. Following such a protocol, as detailed in the algorithm (FIGURE)4-7 we’ve developed and in the text that follows, can help you combat subacute and chronic cough in a cost-effective, timely way.
FIGURE
Dx and treatment when persistent cough is the only symptom4-7
*May include CXR, PPD, B pertussis IgG or IgA, spirometry with methacholine inhalation challenge, barium swallow, prolonged pH monitoring, sinus CT, and sputum eosinophil count, excluding any tests that have already been performed.
ACEI, angiotensin-converting enzyme inhibitor; CT, computed tomography; COPD, chronic obstructive pulmonary disease; CXR, chest x-ray; GERD, gastroesophageal reflux disease; IgA, immunoglobulin A; IgG, immunoglobulin G; PND, postnasal drip; PPD, purified protein derivative; PPI, proton pump inhibitor.
Treat all patients for upper airway cough syndrome
Postnasal drip—renamed UACS by the guideline committee of the American Association of Chest Physicians because it isn’t clear whether the cough is caused by irritation from direct contact with postnasal drip or by inflammation of cough receptors in the upper airway—is the most common cause of chronic cough.6
The differential diagnosis for UACS, which is implicated in about 34% of cases of persistent cough, includes allergic, postinfectious, and occupational rhinitis; rhinitis due to anatomic abnormalities or physical or chemical irritants, rhinitis medicamentosa, and rhinitis of pregnancy; bacterial sinusitis; and allergic fungal sinusitis.8
The signs and symptoms of UACS are nonspecific, and a definitive diagnosis typically cannot be made from the medical history and physical examination alone. What’s more, the absence of any of the usual clinical findings—eg, rhinorrhea and excess sputum production—should not preclude an empiric trial with a first-generation antihistamine-decongestant combination such as brompheniramine/sustained-release pseudoephedrine. Second-and third-generation combination products, such as fexofenadine/pseudoephedrine, should not be used, as they are not effective in treating UACS.4
CASE Margaret’s physical exam was unremarkable. Her vital signs were stable, she had no cervical lymphadenopathy, and her chest was clear on auscultation. She had a dry cough that occurred twice during the exam, but not on inspiration.
The patient’s work-up included office spirometry, which was normal; a nasopharyngeal culture for Bordetella pertussis was negative. We prescribed a 2-week course of therapy with brompheniramine/sustained-release pseudoephedrine and scheduled a return visit shortly after it was completed.
There is no gold standard diagnostic test to confirm or rule out postnasal drip as the cause of cough. CT scanning of sinuses has a poor positive predictive value and is no longer recommended as part of an initial work-up,9 but may be useful for patients whose symptoms persist longer than 3 weeks.
Consider bronchodilator Tx when asthma is suspected
Cough-variant asthma is the second most common cause of persistent cough, and is responsible for an estimated 28% of cases.6 Asthma is the easiest of the conditions included in the differential diagnosis for persistent cough to establish in an office setting. The challenge is to remember to consider it in patients who present with cough but no sign of the classic expiratory wheezing. When you suspect that a patient has asthma, consider empiric bronchodilator therapy—or conduct spirometry testing.
Spirometric values of forced expiratory volume in 1 second/forced vital capacity (FEV1/FVC) <70% and a positive bronchodilator response (≥12%) are consistent with an asthma diagnosis. Management of asthma depends on severity, and patients should be evaluated based on the National Heart, Lung, and Blood Institute’s National Asthma Education and Prevention Program Guidelines for the Diagnosis and Management of Asthma.10
It is crucial to ask patients with asthma (and, indeed, to ask all patients with a persistent cough) about exposure to secondhand smoke, and to stress the importance of avoiding smoking and secondary exposure. Individuals who are regularly exposed to secondhand smoke report more nasal symptoms and greater use of nasal decongestants compared with people with no exposure to smoke;11 they also have poor control of asthma.12-14
Cough unresolved? Add therapy for GERD
Although GERD is primarily associated with heartburn and gastrointestinal distress, it is not unusual for cough to be its only sign or symptom.15 In fact, GERD is the third most common cause of subacute cough—affecting about 21% of patients who seek help for cough at primary care practices.3
CASE Margaret returned to the clinic shortly after completion of a 2-week course of brompheniramine/sustained-release pseudoephedrine, and reported that she was still coughing frequently—and that the medication had brought little improvement. Because of her history of heartburn, we added a 2-week trial with a proton pump inhibitor (PPI)—omeprazole 20 mg/d.
While there are diagnostic tests for GERD, including a pH probe of the esophagus, a barium esophagogram, and manometry testing, empiric therapy with a PPI—starting with a trial of at least 2 weeks—often eliminates the troublesome cough.16 If the patient responds to treatment, the medication can be continued. Risks associated with long-term PPI therapy include osteoporosis and interference with calcium and magnesium absorption,17 so it is important to monitor patients taking them and to discontinue treatment as soon as the cough symptoms resolve.
Have you ruled out postinfectious cough?
If a patient has a cough that has lingered for 3 to 8 weeks after his or her recovery from an acute upper respiratory infection (URI), postinfectious cough may be the reason.18,19 Such a cough is subacute and self-limiting. (If the cough lasts >8 weeks after an acute illness, other diagnoses, such as chronic infection, are more likely.)
The pathogenesis for postinfectious cough may be related to postviral airway inflammation or bronchial hyperresponsiveness, and antibiotics are not indicated.4 Patients may be treated with a bronchodilator such as ipratropium rather than a beta-agonist or inhaled corticosteroids; oral tapered prednisone can be prescribed, if needed, for severe paroxysms, although there is limited evidence of its efficacy.20 Central antitussive agents such as codeine and dextromethorphan can be used when other measures fail to bring relief.
Nonasthmatic eosinophilic bronchitis does not impede airflow
NAEB is less well known than the conditions discussed thus far, but it is a relatively common cause of persistent cough.21-23 In some studies, up to 13% of patients with subacute cough were diagnosed with NAEB.6
Unlike asthma, NAEB is not associated with abnormalities in airway function; patients have no dyspnea and no wheezing, and no obstruction of airflow.24 Patients will have FEV1 >80% and FEV1/FVC >75% on spirometric examination, a negative response to bronchoprovocation, and, typically, an elevated sputum eosinophil count of >3%. Because induced sputum or bronchoscopic washings are difficult, exhaled nitric oxide testing is another option. If these tests are not available, a trial of inhaled steroids is indicated, even if neither spirometry nor bronchoprovocation testing was abnormal.9
Patients with NAEB respond well to inhaled corticosteroids, and budesonide 400 mcg twice a day or prednisolone 30 mg daily may be prescribed. It is also important to remove airway irritants. Long-term follow-up studies of patients with NAEB have had conflicting results. One study found that most cases resolve completely;23 another showed a need for long-term treatment, and suggested that patients with NAEB may be at increased risk for asthma and chronic obstructive pulmonary disease. 25
Paroxysmal cough, whoops point to pertussis
When a patient has paroxysms of cough, posttussive vomiting, and/or an inspiratory whooping sound, B pertussis infection is the likely culprit.26-28 A definitive diagnosis of pertussis, or whooping cough, may be based on a positive culture from a nasopharyngeal aspirate swab.29 Suspected cases can be confirmed with a polymerase chain reaction test, and a presumptive diagnosis may be made as a result of a 4-fold increase in immunoglobulin G or immunoglobulin A antibodies for B pertussis.4
A macrolide antibiotic, usually azithromycin, is the standard treatment for pertussis.30-32 Patients should be isolated for 5 days from the start of treatment. Antibiotic therapy will reduce the risk of transmission, but will not affect the duration of the cough, which may be 6 to 8 weeks. Long-acting beta-agonists, antihistamines, and corticosteroids should not be used to treat pertussis.4
CASE After a 2-week course of omeprazole 20 mg daily, Margaret was coughing much less. We extended the prescription, and by the end of the next 4 weeks, she was no longer coughing. After 2 months, both the PPI and the antihistamine/decongestant were discontinued. We advised her to institute antireflux measures, such as elevating her head at night and not eating after 6 pm, and she has not had a relapse.
CORRESPONDENCE Rebecca H. Gladu, MD, FAAFP, San Jacinto Methodist Hospital, 4401 Garth Road, Baytown, TX 77521; Rgladu@tmhs.org
1. Corrao WM. Chronic persistent cough: diagnosis and treatment update. Pediatr Ann. 1996;25:162-168.
2. Holmes RL, Fadden CT. Evaluation of the patient with chronic cough. Am Fam Physician. 2004;69:2159-2166.
3. Irwin RS, Curley FJ, French CL. Chronic cough. The spectrum and frequency of causes, key components of the diagnostic evaluation, and outcome of specific therapy. Am Rev Respir Dis. 1990;141:640-647.
4. Irwin RS, Baumann MH, Bolser DC, et al. Diagnosis and management of cough. Executive summary: ACCP evidence-based practice guideline. Chest. 2006;129(1 suppl):1S-23S.
5. Pratter MR, Bartter T, Akers S, et al. An algorithmic approach to chronic cough. Ann Intern Med. 1993;119:977-983.
6. Pratter MR, Brightling CE, Boulet LP, et al. An empiric integrative approach to the management of cough: ACCP evidence-based clinical practice guidelines. Chest. 2006;129 (1 suppl):222S-231S.
7. Irwin RS, Madison JM. Anatomical diagnostic protocol in evaluating chronic cough with specific reference to gastroesophageal reflux disease. Am J Med. 2000;108(suppl 4a):126S-130S.
8. Irwin RS, Corrao WM, Pratter MR. Chronic persistent cough in the adult: the spectrum and frequency of causes and successful outcome of specific therapy. Am Rev Respir Dis. 1981;123 (4 Pt 1):413-417.
9. Birring SS. Controversies in the evaluation and management of chronic cough. Am J Respir Crit Care Med. 2011;183:708-715.
10. National Asthma Education and Prevention Program. Expert Panel Report 3 (EPR-3): guidelines for the diagnosis and management of asthma-summary report 2007. J Allergy Clin Immunol. 2007;120(5 suppl):S94-S138.
11. Reh DD, Lin SY, Clipp SL, et al. Secondhand tobacco smoke exposure and chronic rhinosinusitis: a population-based case-control study. Am J Rhinol Allergy. 2009;23:562-567.
12. Stapleton M, Howard-Thompson A, George C, et al. Smoking and asthma. J Am Board Fam Med. 2011;24:313-322.
13. Hersoug LG, Husemoen LL, Sigsgaard T, et al. Indoor exposure to environmental cigarette smoke, but not other inhaled particulates associates with respiratory symptoms and diminished lung function in adults. Respirology. 2010;15:993-1000.
14. Self TH, Wallace JL, Gray LA, et al. Are we failing to document adequate smoking histories? A brief review 1999-2009. Curr Med Res Opin. 2010;26:1691-1696.
15. Sontag SJ. The spectrum of pulmonary symptoms due to gastroesophageal reflux. Thorac Surg Clin. 2005;15:353-368.
16. Irwin RS. Chronic cough due to gastroesophageal reflux. ACCP evidence-based clinical practice guidelines. Chest. 2006;129(suppl 1):80S-94S.
17. Chen J, Yuan YC, Leontiadis GI, et al. Recent safety concerns with proton pump inhibitors. J Clin Gastroenterol. 2012;46:93-114.
18. Braman SS. Postinfectious cough: ACCP evidence-based practice guidelines. Chest. 2006;129(suppl 1):138S-146S.
19. Pratter MR. Cough and the common cold: ACCP evidence-based practice guidelines. Chest. 2006;129(suppl 1):72S-74S.
20. Chang AB, McKean M, Morris P. Inhaled anticholinergics for prolonged non-specific cough in children. Cochrane Database Syst Rev. 2004;(1):CD004358.-
21. Brightling CE, Ward R, Goh KL, et al. Eosinophilic bronchitis is an important cause of chronic cough. Am J Respir Crit Care Med. 1999;160:406-410.
22. Gonlugur U, Gonlugur TE. Eosinophilic bronchitis without asthma. Int Arch Allergy Immunol. 2008;147:1-5.
23. Brightling CE. Cough due to asthma and nonasthmatic eosinophilic bronchitis. Lung. 2010;188 (suppl 1):S13-S17.
24. Gibson PG, Hargreave FE, Girgis-Gabardo, et al. Chronic cough with eosinophilic bronchitis: examination for variable airflow obstruction and response to corticosteroid. Clin Exp Allergy. 1995;25:127-132.
25. Berry MA, Hargadon B, McKenna S, et al. Observational study of the natural history of eosinophilic bronchitis. Clin Exp Allergy. 2005;35:598-601.
26. Antico A, Fabozzi F, Scipiotti C. Pertussis in adults. A study in an Italian population with chronic cough. Monaldi Arch Chest Dis. 2002;57:247-252.
27. Birkebaek NH, Kristiansen M, Seefeldt T, et al. Bordetella pertussis and chronic cough in adults. Clin Infect Dis. 1999;29:1239-1242.
28. Kapaskelis AM, Vouloumanou EK, Rafailidis PI, et al. High prevalence of antibody titers against Bordetella pertussis in an adult population with prolonged cough. Respir Med. 2008;102:1586-1591.
29. Cornia PB, Hersh AL, Lipsky BA, et al. Does this coughing adolescent or adult patient have pertussis? JAMA. 2010;304:890-896.
30. Devasia RA, Jones TF, Collier B, et al. Compliance with azithromycin versus erythromycin in the setting of a pertussis outbreak. Am J Med Sci. 2009;337:176-178.
31. Poe RH, Harder RV, Israel RH, et al. Chronic persistent cough. Experience in diagnosis and outcome using an anatomic diagnostic protocol. Chest. 1989;95:723-728.
32. Altunaji SM, Kukuruzovic RH, Curtis NC, et al. Antibiotics for whooping cough (pertussis). Cochrane Database Syst Rev. 2007;(3):CD004404.-
• Always include postnasal drip, asthma, and gastroesophageal reflux disease in the differential diagnosis for persistent cough, regardless of clinical signs and symptoms. B
• Do not rely on a patient’s description of the character and timing of the cough or the absence (or presence) of sputum to narrow down the differential diagnosis. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE Margaret M, a 52-year-old nonsmoker, came to our clinic because of a persistent cough that had started about 4 weeks earlier. She had tried multiple over-the-counter cough suppressants, including dextromethorphan and guaifenesin, as well as cough drops, but none had been effective.
Margaret denied having had a cold or respiratory infection in the past few months or being in close contact with anyone with a chronic cough, and she had never had an asthma diagnosis. In response to a question about previous coughing episodes, the patient recalled having had several bouts of chronic cough in the past, including one about a year ago.
While Margaret had no known allergies, she did have occasional heartburn, which an antacid—or, at times, a drink of water—always relieved. Thyroid medication and calcium were the only things she took on a regular basis, separated by several hours to avoid problems with absorption.
Patients like Margaret, who seek help from their primary care physician only after attempting to combat a persistent cough on their own, may be quite frustrated by the time they arrive in your office. They’re counting on you to provide a cure. Fortunately, you’re likely to find it, as the differential diagnosis for subacute cough (a cough of 3-8 weeks’ duration) is limited.
Nonetheless, finding the cause of a subacute or chronic cough (lasting >8 weeks) is sometimes a matter of trial and error. Postnasal drip (also known as upper airway cough syndrome, or UACS), asthma, and gastroesophageal reflux disease (GERD) are the most common causes,1,2 followed by postinfectious cough, nonasthmatic eosinophilic bronchitis (NAEB), and pertussis.3 Although these conditions are all relatively well known, they are not always easy to detect: Some disorders, including UACS, asthma, and GERD, may be “silent,” with persistent cough the only presenting sign or symptom.4 In other cases, more than one condition may be contributing to the cough.

Starting with trials of empiric therapy for the most common causes of persistent cough—with sequential therapy and diagnostic tests, as needed—is far more effective than searching for relatively uncommon or obscure conditions. Following such a protocol, as detailed in the algorithm (FIGURE)4-7 we’ve developed and in the text that follows, can help you combat subacute and chronic cough in a cost-effective, timely way.
FIGURE
Dx and treatment when persistent cough is the only symptom4-7
*May include CXR, PPD, B pertussis IgG or IgA, spirometry with methacholine inhalation challenge, barium swallow, prolonged pH monitoring, sinus CT, and sputum eosinophil count, excluding any tests that have already been performed.
ACEI, angiotensin-converting enzyme inhibitor; CT, computed tomography; COPD, chronic obstructive pulmonary disease; CXR, chest x-ray; GERD, gastroesophageal reflux disease; IgA, immunoglobulin A; IgG, immunoglobulin G; PND, postnasal drip; PPD, purified protein derivative; PPI, proton pump inhibitor.
Treat all patients for upper airway cough syndrome
Postnasal drip—renamed UACS by the guideline committee of the American Association of Chest Physicians because it isn’t clear whether the cough is caused by irritation from direct contact with postnasal drip or by inflammation of cough receptors in the upper airway—is the most common cause of chronic cough.6
The differential diagnosis for UACS, which is implicated in about 34% of cases of persistent cough, includes allergic, postinfectious, and occupational rhinitis; rhinitis due to anatomic abnormalities or physical or chemical irritants, rhinitis medicamentosa, and rhinitis of pregnancy; bacterial sinusitis; and allergic fungal sinusitis.8
The signs and symptoms of UACS are nonspecific, and a definitive diagnosis typically cannot be made from the medical history and physical examination alone. What’s more, the absence of any of the usual clinical findings—eg, rhinorrhea and excess sputum production—should not preclude an empiric trial with a first-generation antihistamine-decongestant combination such as brompheniramine/sustained-release pseudoephedrine. Second-and third-generation combination products, such as fexofenadine/pseudoephedrine, should not be used, as they are not effective in treating UACS.4
CASE Margaret’s physical exam was unremarkable. Her vital signs were stable, she had no cervical lymphadenopathy, and her chest was clear on auscultation. She had a dry cough that occurred twice during the exam, but not on inspiration.
The patient’s work-up included office spirometry, which was normal; a nasopharyngeal culture for Bordetella pertussis was negative. We prescribed a 2-week course of therapy with brompheniramine/sustained-release pseudoephedrine and scheduled a return visit shortly after it was completed.
There is no gold standard diagnostic test to confirm or rule out postnasal drip as the cause of cough. CT scanning of sinuses has a poor positive predictive value and is no longer recommended as part of an initial work-up,9 but may be useful for patients whose symptoms persist longer than 3 weeks.
Consider bronchodilator Tx when asthma is suspected
Cough-variant asthma is the second most common cause of persistent cough, and is responsible for an estimated 28% of cases.6 Asthma is the easiest of the conditions included in the differential diagnosis for persistent cough to establish in an office setting. The challenge is to remember to consider it in patients who present with cough but no sign of the classic expiratory wheezing. When you suspect that a patient has asthma, consider empiric bronchodilator therapy—or conduct spirometry testing.
Spirometric values of forced expiratory volume in 1 second/forced vital capacity (FEV1/FVC) <70% and a positive bronchodilator response (≥12%) are consistent with an asthma diagnosis. Management of asthma depends on severity, and patients should be evaluated based on the National Heart, Lung, and Blood Institute’s National Asthma Education and Prevention Program Guidelines for the Diagnosis and Management of Asthma.10
It is crucial to ask patients with asthma (and, indeed, to ask all patients with a persistent cough) about exposure to secondhand smoke, and to stress the importance of avoiding smoking and secondary exposure. Individuals who are regularly exposed to secondhand smoke report more nasal symptoms and greater use of nasal decongestants compared with people with no exposure to smoke;11 they also have poor control of asthma.12-14
Cough unresolved? Add therapy for GERD
Although GERD is primarily associated with heartburn and gastrointestinal distress, it is not unusual for cough to be its only sign or symptom.15 In fact, GERD is the third most common cause of subacute cough—affecting about 21% of patients who seek help for cough at primary care practices.3
CASE Margaret returned to the clinic shortly after completion of a 2-week course of brompheniramine/sustained-release pseudoephedrine, and reported that she was still coughing frequently—and that the medication had brought little improvement. Because of her history of heartburn, we added a 2-week trial with a proton pump inhibitor (PPI)—omeprazole 20 mg/d.
While there are diagnostic tests for GERD, including a pH probe of the esophagus, a barium esophagogram, and manometry testing, empiric therapy with a PPI—starting with a trial of at least 2 weeks—often eliminates the troublesome cough.16 If the patient responds to treatment, the medication can be continued. Risks associated with long-term PPI therapy include osteoporosis and interference with calcium and magnesium absorption,17 so it is important to monitor patients taking them and to discontinue treatment as soon as the cough symptoms resolve.
Have you ruled out postinfectious cough?
If a patient has a cough that has lingered for 3 to 8 weeks after his or her recovery from an acute upper respiratory infection (URI), postinfectious cough may be the reason.18,19 Such a cough is subacute and self-limiting. (If the cough lasts >8 weeks after an acute illness, other diagnoses, such as chronic infection, are more likely.)
The pathogenesis for postinfectious cough may be related to postviral airway inflammation or bronchial hyperresponsiveness, and antibiotics are not indicated.4 Patients may be treated with a bronchodilator such as ipratropium rather than a beta-agonist or inhaled corticosteroids; oral tapered prednisone can be prescribed, if needed, for severe paroxysms, although there is limited evidence of its efficacy.20 Central antitussive agents such as codeine and dextromethorphan can be used when other measures fail to bring relief.
Nonasthmatic eosinophilic bronchitis does not impede airflow
NAEB is less well known than the conditions discussed thus far, but it is a relatively common cause of persistent cough.21-23 In some studies, up to 13% of patients with subacute cough were diagnosed with NAEB.6
Unlike asthma, NAEB is not associated with abnormalities in airway function; patients have no dyspnea and no wheezing, and no obstruction of airflow.24 Patients will have FEV1 >80% and FEV1/FVC >75% on spirometric examination, a negative response to bronchoprovocation, and, typically, an elevated sputum eosinophil count of >3%. Because induced sputum or bronchoscopic washings are difficult, exhaled nitric oxide testing is another option. If these tests are not available, a trial of inhaled steroids is indicated, even if neither spirometry nor bronchoprovocation testing was abnormal.9
Patients with NAEB respond well to inhaled corticosteroids, and budesonide 400 mcg twice a day or prednisolone 30 mg daily may be prescribed. It is also important to remove airway irritants. Long-term follow-up studies of patients with NAEB have had conflicting results. One study found that most cases resolve completely;23 another showed a need for long-term treatment, and suggested that patients with NAEB may be at increased risk for asthma and chronic obstructive pulmonary disease. 25
Paroxysmal cough, whoops point to pertussis
When a patient has paroxysms of cough, posttussive vomiting, and/or an inspiratory whooping sound, B pertussis infection is the likely culprit.26-28 A definitive diagnosis of pertussis, or whooping cough, may be based on a positive culture from a nasopharyngeal aspirate swab.29 Suspected cases can be confirmed with a polymerase chain reaction test, and a presumptive diagnosis may be made as a result of a 4-fold increase in immunoglobulin G or immunoglobulin A antibodies for B pertussis.4
A macrolide antibiotic, usually azithromycin, is the standard treatment for pertussis.30-32 Patients should be isolated for 5 days from the start of treatment. Antibiotic therapy will reduce the risk of transmission, but will not affect the duration of the cough, which may be 6 to 8 weeks. Long-acting beta-agonists, antihistamines, and corticosteroids should not be used to treat pertussis.4
CASE After a 2-week course of omeprazole 20 mg daily, Margaret was coughing much less. We extended the prescription, and by the end of the next 4 weeks, she was no longer coughing. After 2 months, both the PPI and the antihistamine/decongestant were discontinued. We advised her to institute antireflux measures, such as elevating her head at night and not eating after 6 pm, and she has not had a relapse.
CORRESPONDENCE Rebecca H. Gladu, MD, FAAFP, San Jacinto Methodist Hospital, 4401 Garth Road, Baytown, TX 77521; Rgladu@tmhs.org
• Always include postnasal drip, asthma, and gastroesophageal reflux disease in the differential diagnosis for persistent cough, regardless of clinical signs and symptoms. B
• Do not rely on a patient’s description of the character and timing of the cough or the absence (or presence) of sputum to narrow down the differential diagnosis. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE Margaret M, a 52-year-old nonsmoker, came to our clinic because of a persistent cough that had started about 4 weeks earlier. She had tried multiple over-the-counter cough suppressants, including dextromethorphan and guaifenesin, as well as cough drops, but none had been effective.
Margaret denied having had a cold or respiratory infection in the past few months or being in close contact with anyone with a chronic cough, and she had never had an asthma diagnosis. In response to a question about previous coughing episodes, the patient recalled having had several bouts of chronic cough in the past, including one about a year ago.
While Margaret had no known allergies, she did have occasional heartburn, which an antacid—or, at times, a drink of water—always relieved. Thyroid medication and calcium were the only things she took on a regular basis, separated by several hours to avoid problems with absorption.
Patients like Margaret, who seek help from their primary care physician only after attempting to combat a persistent cough on their own, may be quite frustrated by the time they arrive in your office. They’re counting on you to provide a cure. Fortunately, you’re likely to find it, as the differential diagnosis for subacute cough (a cough of 3-8 weeks’ duration) is limited.
Nonetheless, finding the cause of a subacute or chronic cough (lasting >8 weeks) is sometimes a matter of trial and error. Postnasal drip (also known as upper airway cough syndrome, or UACS), asthma, and gastroesophageal reflux disease (GERD) are the most common causes,1,2 followed by postinfectious cough, nonasthmatic eosinophilic bronchitis (NAEB), and pertussis.3 Although these conditions are all relatively well known, they are not always easy to detect: Some disorders, including UACS, asthma, and GERD, may be “silent,” with persistent cough the only presenting sign or symptom.4 In other cases, more than one condition may be contributing to the cough.

Starting with trials of empiric therapy for the most common causes of persistent cough—with sequential therapy and diagnostic tests, as needed—is far more effective than searching for relatively uncommon or obscure conditions. Following such a protocol, as detailed in the algorithm (FIGURE)4-7 we’ve developed and in the text that follows, can help you combat subacute and chronic cough in a cost-effective, timely way.
FIGURE
Dx and treatment when persistent cough is the only symptom4-7
*May include CXR, PPD, B pertussis IgG or IgA, spirometry with methacholine inhalation challenge, barium swallow, prolonged pH monitoring, sinus CT, and sputum eosinophil count, excluding any tests that have already been performed.
ACEI, angiotensin-converting enzyme inhibitor; CT, computed tomography; COPD, chronic obstructive pulmonary disease; CXR, chest x-ray; GERD, gastroesophageal reflux disease; IgA, immunoglobulin A; IgG, immunoglobulin G; PND, postnasal drip; PPD, purified protein derivative; PPI, proton pump inhibitor.
Treat all patients for upper airway cough syndrome
Postnasal drip—renamed UACS by the guideline committee of the American Association of Chest Physicians because it isn’t clear whether the cough is caused by irritation from direct contact with postnasal drip or by inflammation of cough receptors in the upper airway—is the most common cause of chronic cough.6
The differential diagnosis for UACS, which is implicated in about 34% of cases of persistent cough, includes allergic, postinfectious, and occupational rhinitis; rhinitis due to anatomic abnormalities or physical or chemical irritants, rhinitis medicamentosa, and rhinitis of pregnancy; bacterial sinusitis; and allergic fungal sinusitis.8
The signs and symptoms of UACS are nonspecific, and a definitive diagnosis typically cannot be made from the medical history and physical examination alone. What’s more, the absence of any of the usual clinical findings—eg, rhinorrhea and excess sputum production—should not preclude an empiric trial with a first-generation antihistamine-decongestant combination such as brompheniramine/sustained-release pseudoephedrine. Second-and third-generation combination products, such as fexofenadine/pseudoephedrine, should not be used, as they are not effective in treating UACS.4
CASE Margaret’s physical exam was unremarkable. Her vital signs were stable, she had no cervical lymphadenopathy, and her chest was clear on auscultation. She had a dry cough that occurred twice during the exam, but not on inspiration.
The patient’s work-up included office spirometry, which was normal; a nasopharyngeal culture for Bordetella pertussis was negative. We prescribed a 2-week course of therapy with brompheniramine/sustained-release pseudoephedrine and scheduled a return visit shortly after it was completed.
There is no gold standard diagnostic test to confirm or rule out postnasal drip as the cause of cough. CT scanning of sinuses has a poor positive predictive value and is no longer recommended as part of an initial work-up,9 but may be useful for patients whose symptoms persist longer than 3 weeks.
Consider bronchodilator Tx when asthma is suspected
Cough-variant asthma is the second most common cause of persistent cough, and is responsible for an estimated 28% of cases.6 Asthma is the easiest of the conditions included in the differential diagnosis for persistent cough to establish in an office setting. The challenge is to remember to consider it in patients who present with cough but no sign of the classic expiratory wheezing. When you suspect that a patient has asthma, consider empiric bronchodilator therapy—or conduct spirometry testing.
Spirometric values of forced expiratory volume in 1 second/forced vital capacity (FEV1/FVC) <70% and a positive bronchodilator response (≥12%) are consistent with an asthma diagnosis. Management of asthma depends on severity, and patients should be evaluated based on the National Heart, Lung, and Blood Institute’s National Asthma Education and Prevention Program Guidelines for the Diagnosis and Management of Asthma.10
It is crucial to ask patients with asthma (and, indeed, to ask all patients with a persistent cough) about exposure to secondhand smoke, and to stress the importance of avoiding smoking and secondary exposure. Individuals who are regularly exposed to secondhand smoke report more nasal symptoms and greater use of nasal decongestants compared with people with no exposure to smoke;11 they also have poor control of asthma.12-14
Cough unresolved? Add therapy for GERD
Although GERD is primarily associated with heartburn and gastrointestinal distress, it is not unusual for cough to be its only sign or symptom.15 In fact, GERD is the third most common cause of subacute cough—affecting about 21% of patients who seek help for cough at primary care practices.3
CASE Margaret returned to the clinic shortly after completion of a 2-week course of brompheniramine/sustained-release pseudoephedrine, and reported that she was still coughing frequently—and that the medication had brought little improvement. Because of her history of heartburn, we added a 2-week trial with a proton pump inhibitor (PPI)—omeprazole 20 mg/d.
While there are diagnostic tests for GERD, including a pH probe of the esophagus, a barium esophagogram, and manometry testing, empiric therapy with a PPI—starting with a trial of at least 2 weeks—often eliminates the troublesome cough.16 If the patient responds to treatment, the medication can be continued. Risks associated with long-term PPI therapy include osteoporosis and interference with calcium and magnesium absorption,17 so it is important to monitor patients taking them and to discontinue treatment as soon as the cough symptoms resolve.
Have you ruled out postinfectious cough?
If a patient has a cough that has lingered for 3 to 8 weeks after his or her recovery from an acute upper respiratory infection (URI), postinfectious cough may be the reason.18,19 Such a cough is subacute and self-limiting. (If the cough lasts >8 weeks after an acute illness, other diagnoses, such as chronic infection, are more likely.)
The pathogenesis for postinfectious cough may be related to postviral airway inflammation or bronchial hyperresponsiveness, and antibiotics are not indicated.4 Patients may be treated with a bronchodilator such as ipratropium rather than a beta-agonist or inhaled corticosteroids; oral tapered prednisone can be prescribed, if needed, for severe paroxysms, although there is limited evidence of its efficacy.20 Central antitussive agents such as codeine and dextromethorphan can be used when other measures fail to bring relief.
Nonasthmatic eosinophilic bronchitis does not impede airflow
NAEB is less well known than the conditions discussed thus far, but it is a relatively common cause of persistent cough.21-23 In some studies, up to 13% of patients with subacute cough were diagnosed with NAEB.6
Unlike asthma, NAEB is not associated with abnormalities in airway function; patients have no dyspnea and no wheezing, and no obstruction of airflow.24 Patients will have FEV1 >80% and FEV1/FVC >75% on spirometric examination, a negative response to bronchoprovocation, and, typically, an elevated sputum eosinophil count of >3%. Because induced sputum or bronchoscopic washings are difficult, exhaled nitric oxide testing is another option. If these tests are not available, a trial of inhaled steroids is indicated, even if neither spirometry nor bronchoprovocation testing was abnormal.9
Patients with NAEB respond well to inhaled corticosteroids, and budesonide 400 mcg twice a day or prednisolone 30 mg daily may be prescribed. It is also important to remove airway irritants. Long-term follow-up studies of patients with NAEB have had conflicting results. One study found that most cases resolve completely;23 another showed a need for long-term treatment, and suggested that patients with NAEB may be at increased risk for asthma and chronic obstructive pulmonary disease. 25
Paroxysmal cough, whoops point to pertussis
When a patient has paroxysms of cough, posttussive vomiting, and/or an inspiratory whooping sound, B pertussis infection is the likely culprit.26-28 A definitive diagnosis of pertussis, or whooping cough, may be based on a positive culture from a nasopharyngeal aspirate swab.29 Suspected cases can be confirmed with a polymerase chain reaction test, and a presumptive diagnosis may be made as a result of a 4-fold increase in immunoglobulin G or immunoglobulin A antibodies for B pertussis.4
A macrolide antibiotic, usually azithromycin, is the standard treatment for pertussis.30-32 Patients should be isolated for 5 days from the start of treatment. Antibiotic therapy will reduce the risk of transmission, but will not affect the duration of the cough, which may be 6 to 8 weeks. Long-acting beta-agonists, antihistamines, and corticosteroids should not be used to treat pertussis.4
CASE After a 2-week course of omeprazole 20 mg daily, Margaret was coughing much less. We extended the prescription, and by the end of the next 4 weeks, she was no longer coughing. After 2 months, both the PPI and the antihistamine/decongestant were discontinued. We advised her to institute antireflux measures, such as elevating her head at night and not eating after 6 pm, and she has not had a relapse.
CORRESPONDENCE Rebecca H. Gladu, MD, FAAFP, San Jacinto Methodist Hospital, 4401 Garth Road, Baytown, TX 77521; Rgladu@tmhs.org
1. Corrao WM. Chronic persistent cough: diagnosis and treatment update. Pediatr Ann. 1996;25:162-168.
2. Holmes RL, Fadden CT. Evaluation of the patient with chronic cough. Am Fam Physician. 2004;69:2159-2166.
3. Irwin RS, Curley FJ, French CL. Chronic cough. The spectrum and frequency of causes, key components of the diagnostic evaluation, and outcome of specific therapy. Am Rev Respir Dis. 1990;141:640-647.
4. Irwin RS, Baumann MH, Bolser DC, et al. Diagnosis and management of cough. Executive summary: ACCP evidence-based practice guideline. Chest. 2006;129(1 suppl):1S-23S.
5. Pratter MR, Bartter T, Akers S, et al. An algorithmic approach to chronic cough. Ann Intern Med. 1993;119:977-983.
6. Pratter MR, Brightling CE, Boulet LP, et al. An empiric integrative approach to the management of cough: ACCP evidence-based clinical practice guidelines. Chest. 2006;129 (1 suppl):222S-231S.
7. Irwin RS, Madison JM. Anatomical diagnostic protocol in evaluating chronic cough with specific reference to gastroesophageal reflux disease. Am J Med. 2000;108(suppl 4a):126S-130S.
8. Irwin RS, Corrao WM, Pratter MR. Chronic persistent cough in the adult: the spectrum and frequency of causes and successful outcome of specific therapy. Am Rev Respir Dis. 1981;123 (4 Pt 1):413-417.
9. Birring SS. Controversies in the evaluation and management of chronic cough. Am J Respir Crit Care Med. 2011;183:708-715.
10. National Asthma Education and Prevention Program. Expert Panel Report 3 (EPR-3): guidelines for the diagnosis and management of asthma-summary report 2007. J Allergy Clin Immunol. 2007;120(5 suppl):S94-S138.
11. Reh DD, Lin SY, Clipp SL, et al. Secondhand tobacco smoke exposure and chronic rhinosinusitis: a population-based case-control study. Am J Rhinol Allergy. 2009;23:562-567.
12. Stapleton M, Howard-Thompson A, George C, et al. Smoking and asthma. J Am Board Fam Med. 2011;24:313-322.
13. Hersoug LG, Husemoen LL, Sigsgaard T, et al. Indoor exposure to environmental cigarette smoke, but not other inhaled particulates associates with respiratory symptoms and diminished lung function in adults. Respirology. 2010;15:993-1000.
14. Self TH, Wallace JL, Gray LA, et al. Are we failing to document adequate smoking histories? A brief review 1999-2009. Curr Med Res Opin. 2010;26:1691-1696.
15. Sontag SJ. The spectrum of pulmonary symptoms due to gastroesophageal reflux. Thorac Surg Clin. 2005;15:353-368.
16. Irwin RS. Chronic cough due to gastroesophageal reflux. ACCP evidence-based clinical practice guidelines. Chest. 2006;129(suppl 1):80S-94S.
17. Chen J, Yuan YC, Leontiadis GI, et al. Recent safety concerns with proton pump inhibitors. J Clin Gastroenterol. 2012;46:93-114.
18. Braman SS. Postinfectious cough: ACCP evidence-based practice guidelines. Chest. 2006;129(suppl 1):138S-146S.
19. Pratter MR. Cough and the common cold: ACCP evidence-based practice guidelines. Chest. 2006;129(suppl 1):72S-74S.
20. Chang AB, McKean M, Morris P. Inhaled anticholinergics for prolonged non-specific cough in children. Cochrane Database Syst Rev. 2004;(1):CD004358.-
21. Brightling CE, Ward R, Goh KL, et al. Eosinophilic bronchitis is an important cause of chronic cough. Am J Respir Crit Care Med. 1999;160:406-410.
22. Gonlugur U, Gonlugur TE. Eosinophilic bronchitis without asthma. Int Arch Allergy Immunol. 2008;147:1-5.
23. Brightling CE. Cough due to asthma and nonasthmatic eosinophilic bronchitis. Lung. 2010;188 (suppl 1):S13-S17.
24. Gibson PG, Hargreave FE, Girgis-Gabardo, et al. Chronic cough with eosinophilic bronchitis: examination for variable airflow obstruction and response to corticosteroid. Clin Exp Allergy. 1995;25:127-132.
25. Berry MA, Hargadon B, McKenna S, et al. Observational study of the natural history of eosinophilic bronchitis. Clin Exp Allergy. 2005;35:598-601.
26. Antico A, Fabozzi F, Scipiotti C. Pertussis in adults. A study in an Italian population with chronic cough. Monaldi Arch Chest Dis. 2002;57:247-252.
27. Birkebaek NH, Kristiansen M, Seefeldt T, et al. Bordetella pertussis and chronic cough in adults. Clin Infect Dis. 1999;29:1239-1242.
28. Kapaskelis AM, Vouloumanou EK, Rafailidis PI, et al. High prevalence of antibody titers against Bordetella pertussis in an adult population with prolonged cough. Respir Med. 2008;102:1586-1591.
29. Cornia PB, Hersh AL, Lipsky BA, et al. Does this coughing adolescent or adult patient have pertussis? JAMA. 2010;304:890-896.
30. Devasia RA, Jones TF, Collier B, et al. Compliance with azithromycin versus erythromycin in the setting of a pertussis outbreak. Am J Med Sci. 2009;337:176-178.
31. Poe RH, Harder RV, Israel RH, et al. Chronic persistent cough. Experience in diagnosis and outcome using an anatomic diagnostic protocol. Chest. 1989;95:723-728.
32. Altunaji SM, Kukuruzovic RH, Curtis NC, et al. Antibiotics for whooping cough (pertussis). Cochrane Database Syst Rev. 2007;(3):CD004404.-
1. Corrao WM. Chronic persistent cough: diagnosis and treatment update. Pediatr Ann. 1996;25:162-168.
2. Holmes RL, Fadden CT. Evaluation of the patient with chronic cough. Am Fam Physician. 2004;69:2159-2166.
3. Irwin RS, Curley FJ, French CL. Chronic cough. The spectrum and frequency of causes, key components of the diagnostic evaluation, and outcome of specific therapy. Am Rev Respir Dis. 1990;141:640-647.
4. Irwin RS, Baumann MH, Bolser DC, et al. Diagnosis and management of cough. Executive summary: ACCP evidence-based practice guideline. Chest. 2006;129(1 suppl):1S-23S.
5. Pratter MR, Bartter T, Akers S, et al. An algorithmic approach to chronic cough. Ann Intern Med. 1993;119:977-983.
6. Pratter MR, Brightling CE, Boulet LP, et al. An empiric integrative approach to the management of cough: ACCP evidence-based clinical practice guidelines. Chest. 2006;129 (1 suppl):222S-231S.
7. Irwin RS, Madison JM. Anatomical diagnostic protocol in evaluating chronic cough with specific reference to gastroesophageal reflux disease. Am J Med. 2000;108(suppl 4a):126S-130S.
8. Irwin RS, Corrao WM, Pratter MR. Chronic persistent cough in the adult: the spectrum and frequency of causes and successful outcome of specific therapy. Am Rev Respir Dis. 1981;123 (4 Pt 1):413-417.
9. Birring SS. Controversies in the evaluation and management of chronic cough. Am J Respir Crit Care Med. 2011;183:708-715.
10. National Asthma Education and Prevention Program. Expert Panel Report 3 (EPR-3): guidelines for the diagnosis and management of asthma-summary report 2007. J Allergy Clin Immunol. 2007;120(5 suppl):S94-S138.
11. Reh DD, Lin SY, Clipp SL, et al. Secondhand tobacco smoke exposure and chronic rhinosinusitis: a population-based case-control study. Am J Rhinol Allergy. 2009;23:562-567.
12. Stapleton M, Howard-Thompson A, George C, et al. Smoking and asthma. J Am Board Fam Med. 2011;24:313-322.
13. Hersoug LG, Husemoen LL, Sigsgaard T, et al. Indoor exposure to environmental cigarette smoke, but not other inhaled particulates associates with respiratory symptoms and diminished lung function in adults. Respirology. 2010;15:993-1000.
14. Self TH, Wallace JL, Gray LA, et al. Are we failing to document adequate smoking histories? A brief review 1999-2009. Curr Med Res Opin. 2010;26:1691-1696.
15. Sontag SJ. The spectrum of pulmonary symptoms due to gastroesophageal reflux. Thorac Surg Clin. 2005;15:353-368.
16. Irwin RS. Chronic cough due to gastroesophageal reflux. ACCP evidence-based clinical practice guidelines. Chest. 2006;129(suppl 1):80S-94S.
17. Chen J, Yuan YC, Leontiadis GI, et al. Recent safety concerns with proton pump inhibitors. J Clin Gastroenterol. 2012;46:93-114.
18. Braman SS. Postinfectious cough: ACCP evidence-based practice guidelines. Chest. 2006;129(suppl 1):138S-146S.
19. Pratter MR. Cough and the common cold: ACCP evidence-based practice guidelines. Chest. 2006;129(suppl 1):72S-74S.
20. Chang AB, McKean M, Morris P. Inhaled anticholinergics for prolonged non-specific cough in children. Cochrane Database Syst Rev. 2004;(1):CD004358.-
21. Brightling CE, Ward R, Goh KL, et al. Eosinophilic bronchitis is an important cause of chronic cough. Am J Respir Crit Care Med. 1999;160:406-410.
22. Gonlugur U, Gonlugur TE. Eosinophilic bronchitis without asthma. Int Arch Allergy Immunol. 2008;147:1-5.
23. Brightling CE. Cough due to asthma and nonasthmatic eosinophilic bronchitis. Lung. 2010;188 (suppl 1):S13-S17.
24. Gibson PG, Hargreave FE, Girgis-Gabardo, et al. Chronic cough with eosinophilic bronchitis: examination for variable airflow obstruction and response to corticosteroid. Clin Exp Allergy. 1995;25:127-132.
25. Berry MA, Hargadon B, McKenna S, et al. Observational study of the natural history of eosinophilic bronchitis. Clin Exp Allergy. 2005;35:598-601.
26. Antico A, Fabozzi F, Scipiotti C. Pertussis in adults. A study in an Italian population with chronic cough. Monaldi Arch Chest Dis. 2002;57:247-252.
27. Birkebaek NH, Kristiansen M, Seefeldt T, et al. Bordetella pertussis and chronic cough in adults. Clin Infect Dis. 1999;29:1239-1242.
28. Kapaskelis AM, Vouloumanou EK, Rafailidis PI, et al. High prevalence of antibody titers against Bordetella pertussis in an adult population with prolonged cough. Respir Med. 2008;102:1586-1591.
29. Cornia PB, Hersh AL, Lipsky BA, et al. Does this coughing adolescent or adult patient have pertussis? JAMA. 2010;304:890-896.
30. Devasia RA, Jones TF, Collier B, et al. Compliance with azithromycin versus erythromycin in the setting of a pertussis outbreak. Am J Med Sci. 2009;337:176-178.
31. Poe RH, Harder RV, Israel RH, et al. Chronic persistent cough. Experience in diagnosis and outcome using an anatomic diagnostic protocol. Chest. 1989;95:723-728.
32. Altunaji SM, Kukuruzovic RH, Curtis NC, et al. Antibiotics for whooping cough (pertussis). Cochrane Database Syst Rev. 2007;(3):CD004404.-
Step by step: Obliterating the vaginal canal to correct pelvic organ prolapse
- LeFort partial colpocleisis
- Colpectomy and colpocleisis
- Colpectomy and colpocleisis after two previously failed obliterative procedures
These videos were selected by Mickey Karram, MD, and are presented courtesy of the International Academy of Pelvic Surgery (IAPS)
This article, with accompanying video footage, is presented with the support of the International Academy of Pelvic Surgery.
As women live longer, on average, pelvic floor disorders are, as a whole, becoming more prevalent and a greater health and social problem. Many women entering the eighth and ninth decades of life display symptomatic pelvic organ prolapse (POP)—often after an unsuccessful trial of a pessary or even surgery.
These elderly patients often have other concomitant medical issues and are not sexually active, making extensive surgery for them less than an ideal solution. Instead, surgical procedures that obliterate the vaginal canal can alleviate their symptoms of POP.
In this article, we provide a step-by-step description of:
- LeFort partial colpocleisis in a woman who still has her uterus in place
- partial or complete colpectomy and colpocleisis in a woman who has post-hysterectomy prolapse
- levator plication and perineorrhaphy, as essential concluding steps in these procedures.
LeFort partial colpocleisis
An obliterative procedure in the form of a LeFort partial colpocleisis is an option when a patient 1) has her uterus and 2) is no longer sexually active. Because the uterus is retained in this procedure, however, keep in mind that it will be difficult to evaluate any uterine bleeding or cervical pathology in the future. Endovaginal ultrasonography or an endometrial biopsy, and a Pap smear, must be done before LeFort surgery.
The ideal candidate for LeFort partial colpocleisis is a woman who has complete uterine prolapse, or procidentia (FIGURE 1), which is characterized by symmetric eversion of the anterior and posterior vaginal walls.

FIGURE 1 Pelvic organ prolapse, preoperatively
Top: Uterine procidentia. A patient who has this condition is an ideal candidate for LeFort partial colpocleisis. Bottom: Asymmetric anterior vaginal prolapse.
LeFort partial colpocleisis: Key step by key step
![]()
![]()
![]()
![]()
![]()
![]()
![]()
FIGURE 2 shows key steps in performing LeFort partial colpocleisis. See Video #1 at www.obgmanagement.com for demonstrations of how to perform LeFort partial colpocleisis.
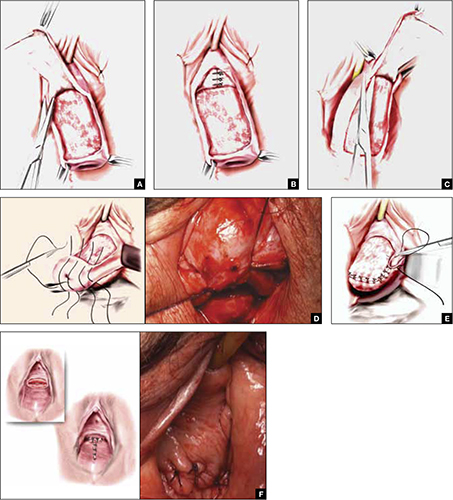
FIGURE 2 Steps: LeFort partial colpocleisis
A. Denude the anterior vaginal epithelium. B. Plicate the neck of the bladder. C. Next, denude the posterior vaginal epithelium. D. Approximate most proximal surfaces. E. Place lateral sutures to allow for drainage canals. F. The uterus has been replaced and most of the distal incisions closed.
Total colpectomy and colpocleisis: Key step by key step
In a patient who has post-hysterectomy prolapse and is not interested in continued sexual function, total colpectomy and colpocleisis provide a highly minimally invasive, durable option to correct her prolapse.
If there is complete eversion of the vagina then, truly, total colpectomy and colpocleisis is the procedure of choice. If there is significant prolapse of only one segment of the pelvic floor, however—for example, the anterior vaginal wall (FIGURE 1)—then aggressive repair of this variant with a narrowing down of the genital hiatus accomplishes the same result without requiring complete removal of what appears to be fairly well supported vaginal mucosa.
Here are key steps for performing partial or complete colpectomy and colpocleisis.
![]()
![]()
Completely remove the vaginal epithelium (FIGURES 3A and 3B); your goal is to leave most of the muscularis of the vaginal wall on the prolapse.
Avoid the peritoneal cavity if at all possible; when the main portion of the prolapse is secondary to an enterocele and the vaginal epithelium is very thin, however, formal excision of the enterocele sac, with closing of the defect, may be required.
![]()
If at all possible, avoid the peritoneum and the wall of the viscera, whether bladder or bowel. Invert the apex of the soft tissue, using the tip of forceps, as each purse-string suture is tied.
There is a variation of this procedure: Perform a separate anterior and posterior colporrhaphy, with two purse-string sutures used to approximate the anterior and posterior segments, thus obliterating any dead space.
![]()
![]()
See Video #2 and Video #3 for a demonstration of how to perform a complete colpectomy and colpocleisis. FIGURE 3D shows the completed colpocleisis.
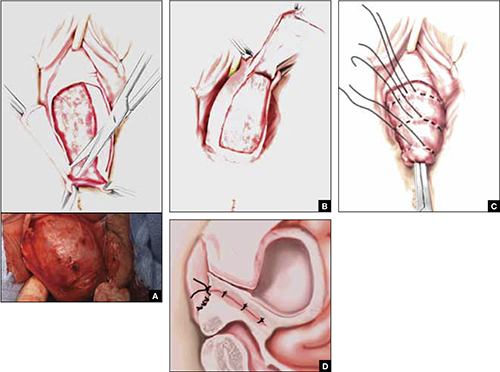
FIGURE 3 Steps: Total colpectomy and colpocleisis
Denude the anterior vaginal epithelium (A) and then the posterior epithelium (B). C. Place sequential purse-string sutures. D. The completed colpocleisis, in cross-section.
Distal levatoroplasty with high perineorrhaphy: Key step by key step
![]()
![]()
![]()
![]()
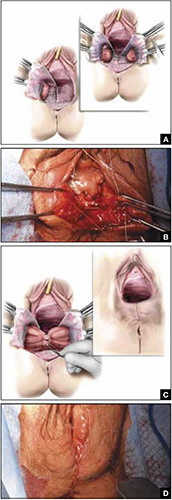
FIGURE 4 Steps: Distal levatoroplasty with high perineorrhaphy
A. Lateral dissection to the levator ani muscles. Inset: levator ani plicated with sequential sutures. B. Place three sutures to plicate the levator ani. C. Secure the plication sutures. Inset C, and D: Completed levatoroplasty.
Our experience
We are often asked questions about the procedures that we’ve just described, including patients’ satisfaction with the outcome, complications, and the risk that prolapse will recur. In the accompanying box, “Questions we’re asked (and answers we give) about obliterative surgery,” opposite, we give our responses to eight common inquiries.
about obliterative surgery
Q1 How satisfied are women with the outcome of these procedures—do many regret having their vaginal canal obliterated?
A Overall, studies indicate that 85% to 100% of patients are “satisfied” or “very satisfied” with the outcomes of obliterative procedures.1 There are rare reports of regret after colpocleisis over loss of coital ability; in one study of a series of procedures,2 5% of subjects expressed regret postoperatively.
Q2 Why is levatoroplasty and perineorrhaphy such an important part of both the LeFort partial colpocleisis and colpectomy and colpocleisis?
A The aim of both these procedures is to reduce prolapsed tissue. The true durability of repair comes from significantly decreasing the caliber of the genital hiatus, with the hope of closing off the bulk of the distal vaginal canal. This can really only be accomplished by utilizing an aggressive levatoroplasty and perineorrhaphy, described in the text.
Q3 How often do patients develop de novo stress incontinence or significant voiding dysfunction, or both, after an obliterative procedure?
A The risk of developing urinary incontinence after an obliterative procedure is difficult to ascertain. In general, patients who had retention or a high postvoid residual volume preoperatively have a good outcome in regard to correcting their voiding dysfunction. This is because, in most cases, the voiding dysfunction is directly related to the anatomic distortion created by the prolapse.
Q4 What is the rate of prolapse recurrence after these procedures, and how is a recurrence managed?
A Multiple studies have documented an excellent anatomic outcome after these procedures, with a prolapse recurrence rate of only 1% to 8%.3 Very little has been written about how to best manage recurrent prolapse after an obliterative procedure. Most surgeons would, most likely, recommend repeat colpocleisis or aggressive levatoroplasty and perineorrhaphy. (Note: The patient whose colpectomy and colpocleisis is shown in Video #3 failed two previous colpectomy and colpocleisis procedures.)
Q5 Can these procedures be performed under local anesthesia, with some intravenous sedation, or under regional anesthesia—thereby avoiding intubation?
A Yes. We have utilized IV sedation and bilateral block successfully to perform these procedures. (Note: Video #3 of LeFort partial colpocleisis shows the procedure performed under local anesthesia.)
Q6 What does the literature say about common complications after these procedures?
A Postoperative morbidity and mortality in the elderly surgical population is a considerable concern. Significant postoperative complications occur in approximately 5% of patients in modern series4—often attributed to the effects of age and to the frail condition of patients who are commonly selected for colpocleisis.
Specifically, approximately 5% of patients experience a postoperative cardiac, thromboembolic, pulmonary, or cerebrovascular event. Transfusion is the most commonly reported major complication related to the procedure itself. Other complications include:
- fever and its associated morbidity
- pneumonia
- ongoing vaginal bleeding
- pyelonephritis
- hematoma
- cystotomy
- ureteral occlusion.
Minor surgical complications occur at a rate of approximately 15%. Surgical mortality is about 1 in 400 cases.
Q7 Do you routinely undertake urodynamic study of patients who are scheduled to undergo an obliterative procedure?
A At minimum, a lower urinary tract evaluation should include a postvoid residual volume study and, we believe, some kind of a filling study and stress test, with reduction of the prolapse. Beyond that, we recommend that you conduct more detailed urodynamic tests on a patient-by-patient basis, when you think that the findings will add to the clinical picture.
Q8 Would you ever perform a vaginal hysterectomy and then proceed with a colpectomy and colpocleisis?
A The principal rationale for performing hysterectomy at the time of colpocleisis is to eliminate the risk of endometrial or cervical carcinoma. Hysterectomy also eliminates the risk of pyometra, a rare but serious complication that can occur when the lateral canals become obstructed after a LeFort procedure.
A recent study5 looked at 1) concomitant hysterectomy in conjunction with colpectomy and colpocleisis and 2) traditional LeFort partial colpocleisis. In this retrospective review, objective and subjective success rates were high, but patients who underwent hysterectomy had a statistically significantly greater decline in postoperative hematocrit and a significant increase in the need for transfusion, compared with patients who did not undergo hysterectomy (35% vs. 13%).
References
1. Fitzgerald MP, Richter HE, Bradley CS, et al. Pelvic support, pelvic symptoms and patient satisfaction after colpocleisis. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19(12):1603-1609.
2. Hullfish K, Bobbjerg B, Steers W. Colpocleisis for pelvic organ prolapse Patient Goals Quality of life and Satisfaction. Obstet Gynecol. 2007;110(2 Pt 1):341-345.
3. Fitzgerald MP, Brubaker L. Colpocleisis and urinary incontinence. Am J Obstet Gynecol. 2003;189(5):1241-1244.
4. von Pechmann WS, Muton M, Fyffe J, Hale DS. Total colpocleisis with high levator placation for the treatment of advanced organ prolapse. Am J Obstet Gynecol. 2003;189(1):121-126.
5. Kohli NE, Sze E, Karram M. Pyometra following LeFort colpocleisis. Int Urogyn J. 1996;7(5):264-266.
We want to hear from you! Tell us what you think.
- LeFort partial colpocleisis
- Colpectomy and colpocleisis
- Colpectomy and colpocleisis after two previously failed obliterative procedures
These videos were selected by Mickey Karram, MD, and are presented courtesy of the International Academy of Pelvic Surgery (IAPS)
This article, with accompanying video footage, is presented with the support of the International Academy of Pelvic Surgery.
As women live longer, on average, pelvic floor disorders are, as a whole, becoming more prevalent and a greater health and social problem. Many women entering the eighth and ninth decades of life display symptomatic pelvic organ prolapse (POP)—often after an unsuccessful trial of a pessary or even surgery.
These elderly patients often have other concomitant medical issues and are not sexually active, making extensive surgery for them less than an ideal solution. Instead, surgical procedures that obliterate the vaginal canal can alleviate their symptoms of POP.
In this article, we provide a step-by-step description of:
- LeFort partial colpocleisis in a woman who still has her uterus in place
- partial or complete colpectomy and colpocleisis in a woman who has post-hysterectomy prolapse
- levator plication and perineorrhaphy, as essential concluding steps in these procedures.
LeFort partial colpocleisis
An obliterative procedure in the form of a LeFort partial colpocleisis is an option when a patient 1) has her uterus and 2) is no longer sexually active. Because the uterus is retained in this procedure, however, keep in mind that it will be difficult to evaluate any uterine bleeding or cervical pathology in the future. Endovaginal ultrasonography or an endometrial biopsy, and a Pap smear, must be done before LeFort surgery.
The ideal candidate for LeFort partial colpocleisis is a woman who has complete uterine prolapse, or procidentia (FIGURE 1), which is characterized by symmetric eversion of the anterior and posterior vaginal walls.

FIGURE 1 Pelvic organ prolapse, preoperatively
Top: Uterine procidentia. A patient who has this condition is an ideal candidate for LeFort partial colpocleisis. Bottom: Asymmetric anterior vaginal prolapse.
LeFort partial colpocleisis: Key step by key step
![]()
![]()
![]()
![]()
![]()
![]()
![]()
FIGURE 2 shows key steps in performing LeFort partial colpocleisis. See Video #1 at www.obgmanagement.com for demonstrations of how to perform LeFort partial colpocleisis.

FIGURE 2 Steps: LeFort partial colpocleisis
A. Denude the anterior vaginal epithelium. B. Plicate the neck of the bladder. C. Next, denude the posterior vaginal epithelium. D. Approximate most proximal surfaces. E. Place lateral sutures to allow for drainage canals. F. The uterus has been replaced and most of the distal incisions closed.
Total colpectomy and colpocleisis: Key step by key step
In a patient who has post-hysterectomy prolapse and is not interested in continued sexual function, total colpectomy and colpocleisis provide a highly minimally invasive, durable option to correct her prolapse.
If there is complete eversion of the vagina then, truly, total colpectomy and colpocleisis is the procedure of choice. If there is significant prolapse of only one segment of the pelvic floor, however—for example, the anterior vaginal wall (FIGURE 1)—then aggressive repair of this variant with a narrowing down of the genital hiatus accomplishes the same result without requiring complete removal of what appears to be fairly well supported vaginal mucosa.
Here are key steps for performing partial or complete colpectomy and colpocleisis.
![]()
![]()
Completely remove the vaginal epithelium (FIGURES 3A and 3B); your goal is to leave most of the muscularis of the vaginal wall on the prolapse.
Avoid the peritoneal cavity if at all possible; when the main portion of the prolapse is secondary to an enterocele and the vaginal epithelium is very thin, however, formal excision of the enterocele sac, with closing of the defect, may be required.
![]()
If at all possible, avoid the peritoneum and the wall of the viscera, whether bladder or bowel. Invert the apex of the soft tissue, using the tip of forceps, as each purse-string suture is tied.
There is a variation of this procedure: Perform a separate anterior and posterior colporrhaphy, with two purse-string sutures used to approximate the anterior and posterior segments, thus obliterating any dead space.
![]()
![]()
See Video #2 and Video #3 for a demonstration of how to perform a complete colpectomy and colpocleisis. FIGURE 3D shows the completed colpocleisis.

FIGURE 3 Steps: Total colpectomy and colpocleisis
Denude the anterior vaginal epithelium (A) and then the posterior epithelium (B). C. Place sequential purse-string sutures. D. The completed colpocleisis, in cross-section.
Distal levatoroplasty with high perineorrhaphy: Key step by key step
![]()
![]()
![]()
![]()

FIGURE 4 Steps: Distal levatoroplasty with high perineorrhaphy
A. Lateral dissection to the levator ani muscles. Inset: levator ani plicated with sequential sutures. B. Place three sutures to plicate the levator ani. C. Secure the plication sutures. Inset C, and D: Completed levatoroplasty.
Our experience
We are often asked questions about the procedures that we’ve just described, including patients’ satisfaction with the outcome, complications, and the risk that prolapse will recur. In the accompanying box, “Questions we’re asked (and answers we give) about obliterative surgery,” opposite, we give our responses to eight common inquiries.
about obliterative surgery
Q1 How satisfied are women with the outcome of these procedures—do many regret having their vaginal canal obliterated?
A Overall, studies indicate that 85% to 100% of patients are “satisfied” or “very satisfied” with the outcomes of obliterative procedures.1 There are rare reports of regret after colpocleisis over loss of coital ability; in one study of a series of procedures,2 5% of subjects expressed regret postoperatively.
Q2 Why is levatoroplasty and perineorrhaphy such an important part of both the LeFort partial colpocleisis and colpectomy and colpocleisis?
A The aim of both these procedures is to reduce prolapsed tissue. The true durability of repair comes from significantly decreasing the caliber of the genital hiatus, with the hope of closing off the bulk of the distal vaginal canal. This can really only be accomplished by utilizing an aggressive levatoroplasty and perineorrhaphy, described in the text.
Q3 How often do patients develop de novo stress incontinence or significant voiding dysfunction, or both, after an obliterative procedure?
A The risk of developing urinary incontinence after an obliterative procedure is difficult to ascertain. In general, patients who had retention or a high postvoid residual volume preoperatively have a good outcome in regard to correcting their voiding dysfunction. This is because, in most cases, the voiding dysfunction is directly related to the anatomic distortion created by the prolapse.
Q4 What is the rate of prolapse recurrence after these procedures, and how is a recurrence managed?
A Multiple studies have documented an excellent anatomic outcome after these procedures, with a prolapse recurrence rate of only 1% to 8%.3 Very little has been written about how to best manage recurrent prolapse after an obliterative procedure. Most surgeons would, most likely, recommend repeat colpocleisis or aggressive levatoroplasty and perineorrhaphy. (Note: The patient whose colpectomy and colpocleisis is shown in Video #3 failed two previous colpectomy and colpocleisis procedures.)
Q5 Can these procedures be performed under local anesthesia, with some intravenous sedation, or under regional anesthesia—thereby avoiding intubation?
A Yes. We have utilized IV sedation and bilateral block successfully to perform these procedures. (Note: Video #3 of LeFort partial colpocleisis shows the procedure performed under local anesthesia.)
Q6 What does the literature say about common complications after these procedures?
A Postoperative morbidity and mortality in the elderly surgical population is a considerable concern. Significant postoperative complications occur in approximately 5% of patients in modern series4—often attributed to the effects of age and to the frail condition of patients who are commonly selected for colpocleisis.
Specifically, approximately 5% of patients experience a postoperative cardiac, thromboembolic, pulmonary, or cerebrovascular event. Transfusion is the most commonly reported major complication related to the procedure itself. Other complications include:
- fever and its associated morbidity
- pneumonia
- ongoing vaginal bleeding
- pyelonephritis
- hematoma
- cystotomy
- ureteral occlusion.
Minor surgical complications occur at a rate of approximately 15%. Surgical mortality is about 1 in 400 cases.
Q7 Do you routinely undertake urodynamic study of patients who are scheduled to undergo an obliterative procedure?
A At minimum, a lower urinary tract evaluation should include a postvoid residual volume study and, we believe, some kind of a filling study and stress test, with reduction of the prolapse. Beyond that, we recommend that you conduct more detailed urodynamic tests on a patient-by-patient basis, when you think that the findings will add to the clinical picture.
Q8 Would you ever perform a vaginal hysterectomy and then proceed with a colpectomy and colpocleisis?
A The principal rationale for performing hysterectomy at the time of colpocleisis is to eliminate the risk of endometrial or cervical carcinoma. Hysterectomy also eliminates the risk of pyometra, a rare but serious complication that can occur when the lateral canals become obstructed after a LeFort procedure.
A recent study5 looked at 1) concomitant hysterectomy in conjunction with colpectomy and colpocleisis and 2) traditional LeFort partial colpocleisis. In this retrospective review, objective and subjective success rates were high, but patients who underwent hysterectomy had a statistically significantly greater decline in postoperative hematocrit and a significant increase in the need for transfusion, compared with patients who did not undergo hysterectomy (35% vs. 13%).
References
1. Fitzgerald MP, Richter HE, Bradley CS, et al. Pelvic support, pelvic symptoms and patient satisfaction after colpocleisis. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19(12):1603-1609.
2. Hullfish K, Bobbjerg B, Steers W. Colpocleisis for pelvic organ prolapse Patient Goals Quality of life and Satisfaction. Obstet Gynecol. 2007;110(2 Pt 1):341-345.
3. Fitzgerald MP, Brubaker L. Colpocleisis and urinary incontinence. Am J Obstet Gynecol. 2003;189(5):1241-1244.
4. von Pechmann WS, Muton M, Fyffe J, Hale DS. Total colpocleisis with high levator placation for the treatment of advanced organ prolapse. Am J Obstet Gynecol. 2003;189(1):121-126.
5. Kohli NE, Sze E, Karram M. Pyometra following LeFort colpocleisis. Int Urogyn J. 1996;7(5):264-266.
We want to hear from you! Tell us what you think.
- LeFort partial colpocleisis
- Colpectomy and colpocleisis
- Colpectomy and colpocleisis after two previously failed obliterative procedures
These videos were selected by Mickey Karram, MD, and are presented courtesy of the International Academy of Pelvic Surgery (IAPS)
This article, with accompanying video footage, is presented with the support of the International Academy of Pelvic Surgery.
As women live longer, on average, pelvic floor disorders are, as a whole, becoming more prevalent and a greater health and social problem. Many women entering the eighth and ninth decades of life display symptomatic pelvic organ prolapse (POP)—often after an unsuccessful trial of a pessary or even surgery.
These elderly patients often have other concomitant medical issues and are not sexually active, making extensive surgery for them less than an ideal solution. Instead, surgical procedures that obliterate the vaginal canal can alleviate their symptoms of POP.
In this article, we provide a step-by-step description of:
- LeFort partial colpocleisis in a woman who still has her uterus in place
- partial or complete colpectomy and colpocleisis in a woman who has post-hysterectomy prolapse
- levator plication and perineorrhaphy, as essential concluding steps in these procedures.
LeFort partial colpocleisis
An obliterative procedure in the form of a LeFort partial colpocleisis is an option when a patient 1) has her uterus and 2) is no longer sexually active. Because the uterus is retained in this procedure, however, keep in mind that it will be difficult to evaluate any uterine bleeding or cervical pathology in the future. Endovaginal ultrasonography or an endometrial biopsy, and a Pap smear, must be done before LeFort surgery.
The ideal candidate for LeFort partial colpocleisis is a woman who has complete uterine prolapse, or procidentia (FIGURE 1), which is characterized by symmetric eversion of the anterior and posterior vaginal walls.

FIGURE 1 Pelvic organ prolapse, preoperatively
Top: Uterine procidentia. A patient who has this condition is an ideal candidate for LeFort partial colpocleisis. Bottom: Asymmetric anterior vaginal prolapse.
LeFort partial colpocleisis: Key step by key step
![]()
![]()
![]()
![]()
![]()
![]()
![]()
FIGURE 2 shows key steps in performing LeFort partial colpocleisis. See Video #1 at www.obgmanagement.com for demonstrations of how to perform LeFort partial colpocleisis.

FIGURE 2 Steps: LeFort partial colpocleisis
A. Denude the anterior vaginal epithelium. B. Plicate the neck of the bladder. C. Next, denude the posterior vaginal epithelium. D. Approximate most proximal surfaces. E. Place lateral sutures to allow for drainage canals. F. The uterus has been replaced and most of the distal incisions closed.
Total colpectomy and colpocleisis: Key step by key step
In a patient who has post-hysterectomy prolapse and is not interested in continued sexual function, total colpectomy and colpocleisis provide a highly minimally invasive, durable option to correct her prolapse.
If there is complete eversion of the vagina then, truly, total colpectomy and colpocleisis is the procedure of choice. If there is significant prolapse of only one segment of the pelvic floor, however—for example, the anterior vaginal wall (FIGURE 1)—then aggressive repair of this variant with a narrowing down of the genital hiatus accomplishes the same result without requiring complete removal of what appears to be fairly well supported vaginal mucosa.
Here are key steps for performing partial or complete colpectomy and colpocleisis.
![]()
![]()
Completely remove the vaginal epithelium (FIGURES 3A and 3B); your goal is to leave most of the muscularis of the vaginal wall on the prolapse.
Avoid the peritoneal cavity if at all possible; when the main portion of the prolapse is secondary to an enterocele and the vaginal epithelium is very thin, however, formal excision of the enterocele sac, with closing of the defect, may be required.
![]()
If at all possible, avoid the peritoneum and the wall of the viscera, whether bladder or bowel. Invert the apex of the soft tissue, using the tip of forceps, as each purse-string suture is tied.
There is a variation of this procedure: Perform a separate anterior and posterior colporrhaphy, with two purse-string sutures used to approximate the anterior and posterior segments, thus obliterating any dead space.
![]()
![]()
See Video #2 and Video #3 for a demonstration of how to perform a complete colpectomy and colpocleisis. FIGURE 3D shows the completed colpocleisis.

FIGURE 3 Steps: Total colpectomy and colpocleisis
Denude the anterior vaginal epithelium (A) and then the posterior epithelium (B). C. Place sequential purse-string sutures. D. The completed colpocleisis, in cross-section.
Distal levatoroplasty with high perineorrhaphy: Key step by key step
![]()
![]()
![]()
![]()

FIGURE 4 Steps: Distal levatoroplasty with high perineorrhaphy
A. Lateral dissection to the levator ani muscles. Inset: levator ani plicated with sequential sutures. B. Place three sutures to plicate the levator ani. C. Secure the plication sutures. Inset C, and D: Completed levatoroplasty.
Our experience
We are often asked questions about the procedures that we’ve just described, including patients’ satisfaction with the outcome, complications, and the risk that prolapse will recur. In the accompanying box, “Questions we’re asked (and answers we give) about obliterative surgery,” opposite, we give our responses to eight common inquiries.
about obliterative surgery
Q1 How satisfied are women with the outcome of these procedures—do many regret having their vaginal canal obliterated?
A Overall, studies indicate that 85% to 100% of patients are “satisfied” or “very satisfied” with the outcomes of obliterative procedures.1 There are rare reports of regret after colpocleisis over loss of coital ability; in one study of a series of procedures,2 5% of subjects expressed regret postoperatively.
Q2 Why is levatoroplasty and perineorrhaphy such an important part of both the LeFort partial colpocleisis and colpectomy and colpocleisis?
A The aim of both these procedures is to reduce prolapsed tissue. The true durability of repair comes from significantly decreasing the caliber of the genital hiatus, with the hope of closing off the bulk of the distal vaginal canal. This can really only be accomplished by utilizing an aggressive levatoroplasty and perineorrhaphy, described in the text.
Q3 How often do patients develop de novo stress incontinence or significant voiding dysfunction, or both, after an obliterative procedure?
A The risk of developing urinary incontinence after an obliterative procedure is difficult to ascertain. In general, patients who had retention or a high postvoid residual volume preoperatively have a good outcome in regard to correcting their voiding dysfunction. This is because, in most cases, the voiding dysfunction is directly related to the anatomic distortion created by the prolapse.
Q4 What is the rate of prolapse recurrence after these procedures, and how is a recurrence managed?
A Multiple studies have documented an excellent anatomic outcome after these procedures, with a prolapse recurrence rate of only 1% to 8%.3 Very little has been written about how to best manage recurrent prolapse after an obliterative procedure. Most surgeons would, most likely, recommend repeat colpocleisis or aggressive levatoroplasty and perineorrhaphy. (Note: The patient whose colpectomy and colpocleisis is shown in Video #3 failed two previous colpectomy and colpocleisis procedures.)
Q5 Can these procedures be performed under local anesthesia, with some intravenous sedation, or under regional anesthesia—thereby avoiding intubation?
A Yes. We have utilized IV sedation and bilateral block successfully to perform these procedures. (Note: Video #3 of LeFort partial colpocleisis shows the procedure performed under local anesthesia.)
Q6 What does the literature say about common complications after these procedures?
A Postoperative morbidity and mortality in the elderly surgical population is a considerable concern. Significant postoperative complications occur in approximately 5% of patients in modern series4—often attributed to the effects of age and to the frail condition of patients who are commonly selected for colpocleisis.
Specifically, approximately 5% of patients experience a postoperative cardiac, thromboembolic, pulmonary, or cerebrovascular event. Transfusion is the most commonly reported major complication related to the procedure itself. Other complications include:
- fever and its associated morbidity
- pneumonia
- ongoing vaginal bleeding
- pyelonephritis
- hematoma
- cystotomy
- ureteral occlusion.
Minor surgical complications occur at a rate of approximately 15%. Surgical mortality is about 1 in 400 cases.
Q7 Do you routinely undertake urodynamic study of patients who are scheduled to undergo an obliterative procedure?
A At minimum, a lower urinary tract evaluation should include a postvoid residual volume study and, we believe, some kind of a filling study and stress test, with reduction of the prolapse. Beyond that, we recommend that you conduct more detailed urodynamic tests on a patient-by-patient basis, when you think that the findings will add to the clinical picture.
Q8 Would you ever perform a vaginal hysterectomy and then proceed with a colpectomy and colpocleisis?
A The principal rationale for performing hysterectomy at the time of colpocleisis is to eliminate the risk of endometrial or cervical carcinoma. Hysterectomy also eliminates the risk of pyometra, a rare but serious complication that can occur when the lateral canals become obstructed after a LeFort procedure.
A recent study5 looked at 1) concomitant hysterectomy in conjunction with colpectomy and colpocleisis and 2) traditional LeFort partial colpocleisis. In this retrospective review, objective and subjective success rates were high, but patients who underwent hysterectomy had a statistically significantly greater decline in postoperative hematocrit and a significant increase in the need for transfusion, compared with patients who did not undergo hysterectomy (35% vs. 13%).
References
1. Fitzgerald MP, Richter HE, Bradley CS, et al. Pelvic support, pelvic symptoms and patient satisfaction after colpocleisis. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19(12):1603-1609.
2. Hullfish K, Bobbjerg B, Steers W. Colpocleisis for pelvic organ prolapse Patient Goals Quality of life and Satisfaction. Obstet Gynecol. 2007;110(2 Pt 1):341-345.
3. Fitzgerald MP, Brubaker L. Colpocleisis and urinary incontinence. Am J Obstet Gynecol. 2003;189(5):1241-1244.
4. von Pechmann WS, Muton M, Fyffe J, Hale DS. Total colpocleisis with high levator placation for the treatment of advanced organ prolapse. Am J Obstet Gynecol. 2003;189(1):121-126.
5. Kohli NE, Sze E, Karram M. Pyometra following LeFort colpocleisis. Int Urogyn J. 1996;7(5):264-266.
We want to hear from you! Tell us what you think.
Have you tried a progestin for your patient’s pelvic pain?
The date of the changeover to the 10th revision of International Classification of Diseases (ICD-10-CM) codes is incorrectly stated in the November 2011 Reimbursement Adviser, page 51. The date should be October 1, 2013.
To read the corrected version of this article, Click here
—The Editors
CASE
Your patient is a 26-year-old G0 woman who has a long history of progressively worsening dysmenorrhea, pelvic pain, and dyspareunia. In the recent past, she was treated with nonsteroidal anti-inflammatory drugs, a cyclic estrogen-progestin contraceptive, and a continuous estrogen-progestin contraceptive—in that order, and without appreciable relief of the pain.
Recently, the woman underwent laparoscopy, which demonstrated Stage-II endometriosis, which was ablated.
What would you prescribe for her postoperatively to alleviate symptoms?
Endometriosis will be diagnosed in approximately 8% of women of reproductive age.1 Pelvic pain, dysmenorrhea, and deep dyspareunia are common symptoms of endometriosis that interfere with quality of life.
Endometriosis is a chronic disease best managed by developing a life-long treatment plan. Following laparoscopic diagnosis and treatment, many experts strongly recommend postoperative hormone-suppressive therapy to reduce the risk that severe pelvic pain will recur, requiring re-operation.
Options for postoperative hormonal treatment of endometriosis include:
- an estrogen–progestin contraceptive
- a progestin (norethindrone acetate [NEA]; depot medroxyprogesterone acetate [DMPA]; oral medroxyprogesterone acetate; the levonorgestrel-releasing intrauterine system [LNG-IUS; Mirena]; and the progestin-releasing implant [Implanon])
- a gonadotropin-releasing hormone (GnRH) agonist (depot leuprolide [Depot Lupron]; nafarelin nasal spray [Synarel]).
CASE Continued
Considering that both cyclic and continuous estrogen-progestin contraceptives have already failed to provide adequate pain relief for your patient, you know that you should offer an alternative to her. Taking into account that progestins are significantly less costly than a GnRH agonist, a progestin formulation might, for her, be considered a first-line postoperative treatment of symptoms of endometriosis.
Options when considering a progestin
Norethindrone acetate
This agent is available in a single formulation: a 5-mg tablet; however, dosages ranging from 2.5 mg/d (half of a tablet) to 15 mg/d have been reported to be effective for relieving pain caused by endometriosis.
What is it? NEA is an androgenic progestin that suppresses luteinizing hormone and follicle-stimulating hormone, thus reducing production of ovarian estrogen. In the absence of ovarian estrogen, endometriosis lesions atrophy. In addition, NEA binds to, and stimulates, endometrial progestin and androgen receptors, resulting in decidualization and atrophy of both eutopic and ectopic endometrial tissue.
Importantly, NEA does not appear to cause bone loss, a phenomenon that is common with agents such as the GnRH agonists or DPMA.2-4
The research record. One randomized study, two pilot studies, and one large observational study have reported that NEA is effective for pelvic pain caused by endometriosis.
In the randomized trial, 90 women who had moderate or severe pelvic pain and rectovaginal endometriosis, and who remained symptomatic after conservative surgery, were randomized to receive NEA, 2.5 mg/d, or a low-dose estrogen-progestin contraceptive (ethinyl estradiol, 10 μg, plus cyproterone acetate, 3 mg) daily for 12 months.5 Both treatment groups reported significant and similar decreases in dysmenorrhea, deep dyspareunia, non-menstrual pain and dyschezia.
In a small pilot study, 40 women who had pelvic pain and colorectal endometriosis were treated with NEA 2.5 mg/d for 12 months. The drug produced significant improvement in dysmenorrhea, pelvic pain, deep dyspareunia, dyschezia, and cyclic rectal bleeding.6
In another pilot study, women who had pelvic pain and rectovaginal endometriosis were treated with either an aromatase inhibitor (letrozole, 2.5 mg/d) plus NEA (2.5 mg/d) or NEA (2.5 mg/d) alone for 6 months. Both treatments resulted in a significant improvement in pelvic pain and deep dyspareunia. Improvement in pain scores was greater with letrozole plus NEA; patients were more satisfied with NEA monotherapy than with the combined letrozole-NEA treatment, however, because the former was associated with fewer side effects.7
In a large (n=194) observational study of the postoperative use of NEA in young women with pelvic pain and endometriosis, NEA at dosages as high as 15 mg/d significantly diminished pelvic pain and self-reported menstrual bleeding. All subjects were started on a dosage of 5 mg/d, which was increased in 2.5-mg increments every 2 weeks to achieve the goals of amenorrhea and a lessening of pelvic pain; the maximum dosage administered was 15 mg/d. Mean duration of NEA use was 13 months; 75% of subjects took the maximum prescribed dosage of 15 mg at some point during treatment. The most commonly reported side effects were weight gain (16% of women); acne (10%); mood lability (9%); and vasomotor symptoms (8%).8
In summary. NEA is effective for treating pelvic pain caused by endometriosis at dosages from 2.5 mg/d to 15 mg/d. An important goal of treatment is a decrease in pain symptoms and amenorrhea; a dosage of 2.5 mg is often insufficient to reliably achieve both of those objectives.
In my practice I begin therapy at a dosage of 5 mg/d; the drug is effective for most patients at that dosage. If 5 mg/d does not reduce pain, I increase the dosage by 2.5 mg (half of a tablet) daily every 4 weeks, to a maximum dosage of 10 mg/d (two tablets). If that dosage is ineffective, I usually discontinue NEA and switch to a GnRH agonist.
Depot medroxyprogesterone acetate; oral medroxy-progesterone acetate
DMPA is available in two FDA-approved formulations:
- a 150-mg dose given by intramuscular injection every 3 months
- a 104-mg dose given by subcutaneous injection every 3 months.
Research. The results of two large clinical trials, comprising a total of more than 550 subjects, showed that DMPA (104 mg, SC, every 3 months) and depot leuprolide (11.25 mg, IM, every 3 months or 3.75 mg, monthly) were each equally effective in relieving dysmenorrhea, dyspareunia, pelvic pain, pelvic tenderness, and pelvic induration in women who had endometriosis.9,10
DMPA was associated with a greater rate of episodes of irregular bleeding than depot leuprolide; conversely, depot leuprolide was associated with greater loss of bone density and a higher incidence of vasomotor symptoms. Weight gain was in the range of 0.6 kg in both groups.
Of note, DPMA is much less expensive than depot leuprolide.
Another study showed that increasing the dosage of DMPA did not improve efficacy over the standard dosage11: DMPA, 150 mg IM, monthly, and DMPA, 150 mg IM, every 3 months produced similar relief of pelvic pain.
Oral medroxyprogesterone acetate, prescribed at high dosages, is also effective for pelvic pain caused by endometriosis. In a pilot study (n=21), oral MPA, 50 mg/d for 4 months, alleviated dysmenorrhea, dyspareunia, pelvic pain, dyschezia, and pelvic tenderness and decreased pelvic nodularity. Sixty percent of subjects reported weight gain— 1.5 kg, on average.12
Progestin-releasing devices: Mirena and Implanon
Many pilot studies have reported that the levonorgestrel-releasing intrauterine system (LNG-IUS) is effective for pelvic pain caused by endometriosis.13-17 For example:
Research. In a small clinical trial, 30 women who had pelvic pain and endometriosis were randomized to receive an LNG-IUS (Mirena) or DMPA, 150 mg IM, every 3 months for 3 years.13 Both therapies were effective at reducing pelvic pain.
At the conclusion of the study, more women opted to retain the LNG-IUS (87%) than to continue DMPA injection (47%). Bone density was maintained in women who had the LNG-IUS placed but slightly diminished in women receiving DMPA.
In a pilot study of an etonogestrel releasing implant (Implanon), 41 women who had pelvic pain and endometriosis were randomized to receive the implant or DMPA, 150 mg IM, every 3 months for 1 year.18 Both therapies were similarly effective at reducing pelvic pain.
Notably, irregular uterine bleeding is a common problem when the etonogestrel-releasing implant is used to treat endometriosis. Achieving amenorrhea or oligomenorrhea is an important goal for women who suffer from pelvic pain caused by endometriosis.
My recommendation
Most ObGyns see patients who are suffering from difficult-to-treat pelvic pain caused by endometriosis. Many of these patients have not had a trial of a progestin, such as NEA, DMPA, or the LNG-IUS that I use in my practice.
Progestins are, as I’ve described, effective for pelvic pain. They are also relatively inexpensive and have a side-effect profile that most patients find acceptable. I recommend that you try a progestin for your patients who have refractory pelvic pain.
What is your preferred hormone treatment for women with unrelieved pelvic pain from endometriosis?
1. Missmer SA, Hankinson S, Spiegelman D, et al. The incidence of laparoscopically confirmed endometriosis by demographic, anthropomorphic and lifestyle factors. Am J Epidemiol. 2004;160(8):784-796.
2. Abdalla HI, Hart DM, Lindsay R, Leggate I, Hooke A. Prevention of bone mineral loss in postmenopausal women by norethisterone. Obstet Gynecol. 1985;66(6):789-792.
3. Riss BJ, Lehmann HJ, Christiansen C. Norethisterone acetate in combination with estrogen: effects on the skeleton and other organs. Am J Obstet Gynecol. 2002;187(4):1101-1116.
4. Hornstein MD, Surrey ES, Weisberg GW, Casino LA. Leuprolide acetate depot and hormonal add-back in endometriosis: a 12-month study. Lupron Add-back Study Group. Obstet Gynecol. 1998;91(1):16-24.
5. Vercellini P, Pietropauolo G, De Giorgi O, Pasin R, Chiodini A, Crosignani PG. Treatment of symptomatic rectovaginal endometriosis with an estrogen-progestogen combination versus low-dose norethindrone acetate. Fertil Steril. 2005;84(5):1375-1387.
6. Ferrero S, Camerini G, Ragni N, Venturini PL, Biscaldi E, Remorgida V. Norethisterone acetate in the treatment of colorectal endometriosis: a pilot study. Hum Reprod. 2010;25(1):94-100.
7. Ferrero S, Camerini G, Seracchioli R, Ragni N, Venturini PL, Remorgida V. Letrozole combined with norethisterone acetate compared with norethisterone acetate alone in the treatment of pain symptoms caused by endometriosis. Hum Reprod. 2009;24(12):3033-3341.
8. Kaser DJ, Missmer SA, Berry KF, Laufer MR. Use of norethindrone acetate alone for postoperative suppression of endometriosis symptoms [published online ahead of print December 9 2011]. J Pediatr Adolesc Gynecol. doi:10.1016/j.jpag.2011.09.013.
9. Schlaff WD, Carson SA, Luciano A, Ross D, Bergqvist A. Subcutaneous injection of depot medroxyprogesterone acetate compared with leuprolide acetate in the treatment of endometriosis associated pain. Fertil Steril. 2006;85(2):314-325.
10. Crosignani PG, Luciano A, Ray A, Bergqvist A. Subcutaneous depot medroxyprogesterone acetate versus leuprolide acetate in the treatment of endometriosis-associated pain. Hum Reprod. 2006;21(1):248-256.
11. Cheewadhanaraks S, Peeyananjarassri K, Choksuchat C, Dhanaworavibul K, Choobun T, Bunyapipat S. Interval of injections of intramuscular depot medroxyprogesterone acetate in the long-term treatment of endometriosis-associated pain: a randomized clinical trial. Gynecol Obstet Invest. 2009;68(2):116-121.
12. Luciano AA, Turksoy RN, Carleo J. Evaluation of oral medroxyprogesterone acetate in the treatment of endometriosis. Obstet Gynecol. 1988;72(3 Pt 1):323-327.
13. Wong AY, Tang LC, Chin RK. Levonorgestrel-releasing intrauterine system (Mirena) and depot medroxyprogesterone acetate (Depoprovera) as long-term maintenance therapy for patients with moderate and severe endometriosis: a randomized controlled trial. Aust N Z J Obstet Gynaecol. 2010;50(3):273-279.
14. Lockhat FB, Emembolu JO, Konje JC. The efficacy side-effects and continuation rates in women with symptomatic endometriosis undergoing treatment with an intrauterine administered progestogen (levonorgestrel): a 3 year follow-up. Hum Reprod. 2005;20(3):789-793.
15. Petta CA, Ferriani RA, Abrao MS, et al. Randomized clinical trial of a levonorgestrel-releasing intrauterine system and a depot GnRH analogue for the treatment of chronic pelvic pain in women with endometriosis. Hum Reprod. 2005;20(7):1993-1998.
16. Vercellini P, Aimi G, Panazza S, De Giorgi O, Pesole A, Crosignani PG. A levonorgestrel-releasing intrauterine system for the treatment of dysmenorrhea associated with endometriosis: a pilot study. Fertil Steril. 1999;72(3):505-508.
17. Vercellini P, Frontino G, De Giorgi O, Aimi G, Zaina B, Crosignani PG. Comparison of a levonorgestrel-releasing intrauterine device versus expectant management after conservative surgery for symptomatic endometriosis: a pilot study. Fertil Steril. 2003;80(2):305-309.
18. Walch K, Unfried G, Huber J, Kurz C, van Trotsenburg M, Pernicka E, Wenzl R. Implanon versus medroxyprogesterone acetate: effects on pain scores in patients with symptomatic endometriosis—a pilot study. Contraception. 2009;79(1):29-34.
The date of the changeover to the 10th revision of International Classification of Diseases (ICD-10-CM) codes is incorrectly stated in the November 2011 Reimbursement Adviser, page 51. The date should be October 1, 2013.
To read the corrected version of this article, Click here
—The Editors
CASE
Your patient is a 26-year-old G0 woman who has a long history of progressively worsening dysmenorrhea, pelvic pain, and dyspareunia. In the recent past, she was treated with nonsteroidal anti-inflammatory drugs, a cyclic estrogen-progestin contraceptive, and a continuous estrogen-progestin contraceptive—in that order, and without appreciable relief of the pain.
Recently, the woman underwent laparoscopy, which demonstrated Stage-II endometriosis, which was ablated.
What would you prescribe for her postoperatively to alleviate symptoms?
Endometriosis will be diagnosed in approximately 8% of women of reproductive age.1 Pelvic pain, dysmenorrhea, and deep dyspareunia are common symptoms of endometriosis that interfere with quality of life.
Endometriosis is a chronic disease best managed by developing a life-long treatment plan. Following laparoscopic diagnosis and treatment, many experts strongly recommend postoperative hormone-suppressive therapy to reduce the risk that severe pelvic pain will recur, requiring re-operation.
Options for postoperative hormonal treatment of endometriosis include:
- an estrogen–progestin contraceptive
- a progestin (norethindrone acetate [NEA]; depot medroxyprogesterone acetate [DMPA]; oral medroxyprogesterone acetate; the levonorgestrel-releasing intrauterine system [LNG-IUS; Mirena]; and the progestin-releasing implant [Implanon])
- a gonadotropin-releasing hormone (GnRH) agonist (depot leuprolide [Depot Lupron]; nafarelin nasal spray [Synarel]).
CASE Continued
Considering that both cyclic and continuous estrogen-progestin contraceptives have already failed to provide adequate pain relief for your patient, you know that you should offer an alternative to her. Taking into account that progestins are significantly less costly than a GnRH agonist, a progestin formulation might, for her, be considered a first-line postoperative treatment of symptoms of endometriosis.
Options when considering a progestin
Norethindrone acetate
This agent is available in a single formulation: a 5-mg tablet; however, dosages ranging from 2.5 mg/d (half of a tablet) to 15 mg/d have been reported to be effective for relieving pain caused by endometriosis.
What is it? NEA is an androgenic progestin that suppresses luteinizing hormone and follicle-stimulating hormone, thus reducing production of ovarian estrogen. In the absence of ovarian estrogen, endometriosis lesions atrophy. In addition, NEA binds to, and stimulates, endometrial progestin and androgen receptors, resulting in decidualization and atrophy of both eutopic and ectopic endometrial tissue.
Importantly, NEA does not appear to cause bone loss, a phenomenon that is common with agents such as the GnRH agonists or DPMA.2-4
The research record. One randomized study, two pilot studies, and one large observational study have reported that NEA is effective for pelvic pain caused by endometriosis.
In the randomized trial, 90 women who had moderate or severe pelvic pain and rectovaginal endometriosis, and who remained symptomatic after conservative surgery, were randomized to receive NEA, 2.5 mg/d, or a low-dose estrogen-progestin contraceptive (ethinyl estradiol, 10 μg, plus cyproterone acetate, 3 mg) daily for 12 months.5 Both treatment groups reported significant and similar decreases in dysmenorrhea, deep dyspareunia, non-menstrual pain and dyschezia.
In a small pilot study, 40 women who had pelvic pain and colorectal endometriosis were treated with NEA 2.5 mg/d for 12 months. The drug produced significant improvement in dysmenorrhea, pelvic pain, deep dyspareunia, dyschezia, and cyclic rectal bleeding.6
In another pilot study, women who had pelvic pain and rectovaginal endometriosis were treated with either an aromatase inhibitor (letrozole, 2.5 mg/d) plus NEA (2.5 mg/d) or NEA (2.5 mg/d) alone for 6 months. Both treatments resulted in a significant improvement in pelvic pain and deep dyspareunia. Improvement in pain scores was greater with letrozole plus NEA; patients were more satisfied with NEA monotherapy than with the combined letrozole-NEA treatment, however, because the former was associated with fewer side effects.7
In a large (n=194) observational study of the postoperative use of NEA in young women with pelvic pain and endometriosis, NEA at dosages as high as 15 mg/d significantly diminished pelvic pain and self-reported menstrual bleeding. All subjects were started on a dosage of 5 mg/d, which was increased in 2.5-mg increments every 2 weeks to achieve the goals of amenorrhea and a lessening of pelvic pain; the maximum dosage administered was 15 mg/d. Mean duration of NEA use was 13 months; 75% of subjects took the maximum prescribed dosage of 15 mg at some point during treatment. The most commonly reported side effects were weight gain (16% of women); acne (10%); mood lability (9%); and vasomotor symptoms (8%).8
In summary. NEA is effective for treating pelvic pain caused by endometriosis at dosages from 2.5 mg/d to 15 mg/d. An important goal of treatment is a decrease in pain symptoms and amenorrhea; a dosage of 2.5 mg is often insufficient to reliably achieve both of those objectives.
In my practice I begin therapy at a dosage of 5 mg/d; the drug is effective for most patients at that dosage. If 5 mg/d does not reduce pain, I increase the dosage by 2.5 mg (half of a tablet) daily every 4 weeks, to a maximum dosage of 10 mg/d (two tablets). If that dosage is ineffective, I usually discontinue NEA and switch to a GnRH agonist.
Depot medroxyprogesterone acetate; oral medroxy-progesterone acetate
DMPA is available in two FDA-approved formulations:
- a 150-mg dose given by intramuscular injection every 3 months
- a 104-mg dose given by subcutaneous injection every 3 months.
Research. The results of two large clinical trials, comprising a total of more than 550 subjects, showed that DMPA (104 mg, SC, every 3 months) and depot leuprolide (11.25 mg, IM, every 3 months or 3.75 mg, monthly) were each equally effective in relieving dysmenorrhea, dyspareunia, pelvic pain, pelvic tenderness, and pelvic induration in women who had endometriosis.9,10
DMPA was associated with a greater rate of episodes of irregular bleeding than depot leuprolide; conversely, depot leuprolide was associated with greater loss of bone density and a higher incidence of vasomotor symptoms. Weight gain was in the range of 0.6 kg in both groups.
Of note, DPMA is much less expensive than depot leuprolide.
Another study showed that increasing the dosage of DMPA did not improve efficacy over the standard dosage11: DMPA, 150 mg IM, monthly, and DMPA, 150 mg IM, every 3 months produced similar relief of pelvic pain.
Oral medroxyprogesterone acetate, prescribed at high dosages, is also effective for pelvic pain caused by endometriosis. In a pilot study (n=21), oral MPA, 50 mg/d for 4 months, alleviated dysmenorrhea, dyspareunia, pelvic pain, dyschezia, and pelvic tenderness and decreased pelvic nodularity. Sixty percent of subjects reported weight gain— 1.5 kg, on average.12
Progestin-releasing devices: Mirena and Implanon
Many pilot studies have reported that the levonorgestrel-releasing intrauterine system (LNG-IUS) is effective for pelvic pain caused by endometriosis.13-17 For example:
Research. In a small clinical trial, 30 women who had pelvic pain and endometriosis were randomized to receive an LNG-IUS (Mirena) or DMPA, 150 mg IM, every 3 months for 3 years.13 Both therapies were effective at reducing pelvic pain.
At the conclusion of the study, more women opted to retain the LNG-IUS (87%) than to continue DMPA injection (47%). Bone density was maintained in women who had the LNG-IUS placed but slightly diminished in women receiving DMPA.
In a pilot study of an etonogestrel releasing implant (Implanon), 41 women who had pelvic pain and endometriosis were randomized to receive the implant or DMPA, 150 mg IM, every 3 months for 1 year.18 Both therapies were similarly effective at reducing pelvic pain.
Notably, irregular uterine bleeding is a common problem when the etonogestrel-releasing implant is used to treat endometriosis. Achieving amenorrhea or oligomenorrhea is an important goal for women who suffer from pelvic pain caused by endometriosis.
My recommendation
Most ObGyns see patients who are suffering from difficult-to-treat pelvic pain caused by endometriosis. Many of these patients have not had a trial of a progestin, such as NEA, DMPA, or the LNG-IUS that I use in my practice.
Progestins are, as I’ve described, effective for pelvic pain. They are also relatively inexpensive and have a side-effect profile that most patients find acceptable. I recommend that you try a progestin for your patients who have refractory pelvic pain.
What is your preferred hormone treatment for women with unrelieved pelvic pain from endometriosis?
The date of the changeover to the 10th revision of International Classification of Diseases (ICD-10-CM) codes is incorrectly stated in the November 2011 Reimbursement Adviser, page 51. The date should be October 1, 2013.
To read the corrected version of this article, Click here
—The Editors
CASE
Your patient is a 26-year-old G0 woman who has a long history of progressively worsening dysmenorrhea, pelvic pain, and dyspareunia. In the recent past, she was treated with nonsteroidal anti-inflammatory drugs, a cyclic estrogen-progestin contraceptive, and a continuous estrogen-progestin contraceptive—in that order, and without appreciable relief of the pain.
Recently, the woman underwent laparoscopy, which demonstrated Stage-II endometriosis, which was ablated.
What would you prescribe for her postoperatively to alleviate symptoms?
Endometriosis will be diagnosed in approximately 8% of women of reproductive age.1 Pelvic pain, dysmenorrhea, and deep dyspareunia are common symptoms of endometriosis that interfere with quality of life.
Endometriosis is a chronic disease best managed by developing a life-long treatment plan. Following laparoscopic diagnosis and treatment, many experts strongly recommend postoperative hormone-suppressive therapy to reduce the risk that severe pelvic pain will recur, requiring re-operation.
Options for postoperative hormonal treatment of endometriosis include:
- an estrogen–progestin contraceptive
- a progestin (norethindrone acetate [NEA]; depot medroxyprogesterone acetate [DMPA]; oral medroxyprogesterone acetate; the levonorgestrel-releasing intrauterine system [LNG-IUS; Mirena]; and the progestin-releasing implant [Implanon])
- a gonadotropin-releasing hormone (GnRH) agonist (depot leuprolide [Depot Lupron]; nafarelin nasal spray [Synarel]).
CASE Continued
Considering that both cyclic and continuous estrogen-progestin contraceptives have already failed to provide adequate pain relief for your patient, you know that you should offer an alternative to her. Taking into account that progestins are significantly less costly than a GnRH agonist, a progestin formulation might, for her, be considered a first-line postoperative treatment of symptoms of endometriosis.
Options when considering a progestin
Norethindrone acetate
This agent is available in a single formulation: a 5-mg tablet; however, dosages ranging from 2.5 mg/d (half of a tablet) to 15 mg/d have been reported to be effective for relieving pain caused by endometriosis.
What is it? NEA is an androgenic progestin that suppresses luteinizing hormone and follicle-stimulating hormone, thus reducing production of ovarian estrogen. In the absence of ovarian estrogen, endometriosis lesions atrophy. In addition, NEA binds to, and stimulates, endometrial progestin and androgen receptors, resulting in decidualization and atrophy of both eutopic and ectopic endometrial tissue.
Importantly, NEA does not appear to cause bone loss, a phenomenon that is common with agents such as the GnRH agonists or DPMA.2-4
The research record. One randomized study, two pilot studies, and one large observational study have reported that NEA is effective for pelvic pain caused by endometriosis.
In the randomized trial, 90 women who had moderate or severe pelvic pain and rectovaginal endometriosis, and who remained symptomatic after conservative surgery, were randomized to receive NEA, 2.5 mg/d, or a low-dose estrogen-progestin contraceptive (ethinyl estradiol, 10 μg, plus cyproterone acetate, 3 mg) daily for 12 months.5 Both treatment groups reported significant and similar decreases in dysmenorrhea, deep dyspareunia, non-menstrual pain and dyschezia.
In a small pilot study, 40 women who had pelvic pain and colorectal endometriosis were treated with NEA 2.5 mg/d for 12 months. The drug produced significant improvement in dysmenorrhea, pelvic pain, deep dyspareunia, dyschezia, and cyclic rectal bleeding.6
In another pilot study, women who had pelvic pain and rectovaginal endometriosis were treated with either an aromatase inhibitor (letrozole, 2.5 mg/d) plus NEA (2.5 mg/d) or NEA (2.5 mg/d) alone for 6 months. Both treatments resulted in a significant improvement in pelvic pain and deep dyspareunia. Improvement in pain scores was greater with letrozole plus NEA; patients were more satisfied with NEA monotherapy than with the combined letrozole-NEA treatment, however, because the former was associated with fewer side effects.7
In a large (n=194) observational study of the postoperative use of NEA in young women with pelvic pain and endometriosis, NEA at dosages as high as 15 mg/d significantly diminished pelvic pain and self-reported menstrual bleeding. All subjects were started on a dosage of 5 mg/d, which was increased in 2.5-mg increments every 2 weeks to achieve the goals of amenorrhea and a lessening of pelvic pain; the maximum dosage administered was 15 mg/d. Mean duration of NEA use was 13 months; 75% of subjects took the maximum prescribed dosage of 15 mg at some point during treatment. The most commonly reported side effects were weight gain (16% of women); acne (10%); mood lability (9%); and vasomotor symptoms (8%).8
In summary. NEA is effective for treating pelvic pain caused by endometriosis at dosages from 2.5 mg/d to 15 mg/d. An important goal of treatment is a decrease in pain symptoms and amenorrhea; a dosage of 2.5 mg is often insufficient to reliably achieve both of those objectives.
In my practice I begin therapy at a dosage of 5 mg/d; the drug is effective for most patients at that dosage. If 5 mg/d does not reduce pain, I increase the dosage by 2.5 mg (half of a tablet) daily every 4 weeks, to a maximum dosage of 10 mg/d (two tablets). If that dosage is ineffective, I usually discontinue NEA and switch to a GnRH agonist.
Depot medroxyprogesterone acetate; oral medroxy-progesterone acetate
DMPA is available in two FDA-approved formulations:
- a 150-mg dose given by intramuscular injection every 3 months
- a 104-mg dose given by subcutaneous injection every 3 months.
Research. The results of two large clinical trials, comprising a total of more than 550 subjects, showed that DMPA (104 mg, SC, every 3 months) and depot leuprolide (11.25 mg, IM, every 3 months or 3.75 mg, monthly) were each equally effective in relieving dysmenorrhea, dyspareunia, pelvic pain, pelvic tenderness, and pelvic induration in women who had endometriosis.9,10
DMPA was associated with a greater rate of episodes of irregular bleeding than depot leuprolide; conversely, depot leuprolide was associated with greater loss of bone density and a higher incidence of vasomotor symptoms. Weight gain was in the range of 0.6 kg in both groups.
Of note, DPMA is much less expensive than depot leuprolide.
Another study showed that increasing the dosage of DMPA did not improve efficacy over the standard dosage11: DMPA, 150 mg IM, monthly, and DMPA, 150 mg IM, every 3 months produced similar relief of pelvic pain.
Oral medroxyprogesterone acetate, prescribed at high dosages, is also effective for pelvic pain caused by endometriosis. In a pilot study (n=21), oral MPA, 50 mg/d for 4 months, alleviated dysmenorrhea, dyspareunia, pelvic pain, dyschezia, and pelvic tenderness and decreased pelvic nodularity. Sixty percent of subjects reported weight gain— 1.5 kg, on average.12
Progestin-releasing devices: Mirena and Implanon
Many pilot studies have reported that the levonorgestrel-releasing intrauterine system (LNG-IUS) is effective for pelvic pain caused by endometriosis.13-17 For example:
Research. In a small clinical trial, 30 women who had pelvic pain and endometriosis were randomized to receive an LNG-IUS (Mirena) or DMPA, 150 mg IM, every 3 months for 3 years.13 Both therapies were effective at reducing pelvic pain.
At the conclusion of the study, more women opted to retain the LNG-IUS (87%) than to continue DMPA injection (47%). Bone density was maintained in women who had the LNG-IUS placed but slightly diminished in women receiving DMPA.
In a pilot study of an etonogestrel releasing implant (Implanon), 41 women who had pelvic pain and endometriosis were randomized to receive the implant or DMPA, 150 mg IM, every 3 months for 1 year.18 Both therapies were similarly effective at reducing pelvic pain.
Notably, irregular uterine bleeding is a common problem when the etonogestrel-releasing implant is used to treat endometriosis. Achieving amenorrhea or oligomenorrhea is an important goal for women who suffer from pelvic pain caused by endometriosis.
My recommendation
Most ObGyns see patients who are suffering from difficult-to-treat pelvic pain caused by endometriosis. Many of these patients have not had a trial of a progestin, such as NEA, DMPA, or the LNG-IUS that I use in my practice.
Progestins are, as I’ve described, effective for pelvic pain. They are also relatively inexpensive and have a side-effect profile that most patients find acceptable. I recommend that you try a progestin for your patients who have refractory pelvic pain.
What is your preferred hormone treatment for women with unrelieved pelvic pain from endometriosis?
1. Missmer SA, Hankinson S, Spiegelman D, et al. The incidence of laparoscopically confirmed endometriosis by demographic, anthropomorphic and lifestyle factors. Am J Epidemiol. 2004;160(8):784-796.
2. Abdalla HI, Hart DM, Lindsay R, Leggate I, Hooke A. Prevention of bone mineral loss in postmenopausal women by norethisterone. Obstet Gynecol. 1985;66(6):789-792.
3. Riss BJ, Lehmann HJ, Christiansen C. Norethisterone acetate in combination with estrogen: effects on the skeleton and other organs. Am J Obstet Gynecol. 2002;187(4):1101-1116.
4. Hornstein MD, Surrey ES, Weisberg GW, Casino LA. Leuprolide acetate depot and hormonal add-back in endometriosis: a 12-month study. Lupron Add-back Study Group. Obstet Gynecol. 1998;91(1):16-24.
5. Vercellini P, Pietropauolo G, De Giorgi O, Pasin R, Chiodini A, Crosignani PG. Treatment of symptomatic rectovaginal endometriosis with an estrogen-progestogen combination versus low-dose norethindrone acetate. Fertil Steril. 2005;84(5):1375-1387.
6. Ferrero S, Camerini G, Ragni N, Venturini PL, Biscaldi E, Remorgida V. Norethisterone acetate in the treatment of colorectal endometriosis: a pilot study. Hum Reprod. 2010;25(1):94-100.
7. Ferrero S, Camerini G, Seracchioli R, Ragni N, Venturini PL, Remorgida V. Letrozole combined with norethisterone acetate compared with norethisterone acetate alone in the treatment of pain symptoms caused by endometriosis. Hum Reprod. 2009;24(12):3033-3341.
8. Kaser DJ, Missmer SA, Berry KF, Laufer MR. Use of norethindrone acetate alone for postoperative suppression of endometriosis symptoms [published online ahead of print December 9 2011]. J Pediatr Adolesc Gynecol. doi:10.1016/j.jpag.2011.09.013.
9. Schlaff WD, Carson SA, Luciano A, Ross D, Bergqvist A. Subcutaneous injection of depot medroxyprogesterone acetate compared with leuprolide acetate in the treatment of endometriosis associated pain. Fertil Steril. 2006;85(2):314-325.
10. Crosignani PG, Luciano A, Ray A, Bergqvist A. Subcutaneous depot medroxyprogesterone acetate versus leuprolide acetate in the treatment of endometriosis-associated pain. Hum Reprod. 2006;21(1):248-256.
11. Cheewadhanaraks S, Peeyananjarassri K, Choksuchat C, Dhanaworavibul K, Choobun T, Bunyapipat S. Interval of injections of intramuscular depot medroxyprogesterone acetate in the long-term treatment of endometriosis-associated pain: a randomized clinical trial. Gynecol Obstet Invest. 2009;68(2):116-121.
12. Luciano AA, Turksoy RN, Carleo J. Evaluation of oral medroxyprogesterone acetate in the treatment of endometriosis. Obstet Gynecol. 1988;72(3 Pt 1):323-327.
13. Wong AY, Tang LC, Chin RK. Levonorgestrel-releasing intrauterine system (Mirena) and depot medroxyprogesterone acetate (Depoprovera) as long-term maintenance therapy for patients with moderate and severe endometriosis: a randomized controlled trial. Aust N Z J Obstet Gynaecol. 2010;50(3):273-279.
14. Lockhat FB, Emembolu JO, Konje JC. The efficacy side-effects and continuation rates in women with symptomatic endometriosis undergoing treatment with an intrauterine administered progestogen (levonorgestrel): a 3 year follow-up. Hum Reprod. 2005;20(3):789-793.
15. Petta CA, Ferriani RA, Abrao MS, et al. Randomized clinical trial of a levonorgestrel-releasing intrauterine system and a depot GnRH analogue for the treatment of chronic pelvic pain in women with endometriosis. Hum Reprod. 2005;20(7):1993-1998.
16. Vercellini P, Aimi G, Panazza S, De Giorgi O, Pesole A, Crosignani PG. A levonorgestrel-releasing intrauterine system for the treatment of dysmenorrhea associated with endometriosis: a pilot study. Fertil Steril. 1999;72(3):505-508.
17. Vercellini P, Frontino G, De Giorgi O, Aimi G, Zaina B, Crosignani PG. Comparison of a levonorgestrel-releasing intrauterine device versus expectant management after conservative surgery for symptomatic endometriosis: a pilot study. Fertil Steril. 2003;80(2):305-309.
18. Walch K, Unfried G, Huber J, Kurz C, van Trotsenburg M, Pernicka E, Wenzl R. Implanon versus medroxyprogesterone acetate: effects on pain scores in patients with symptomatic endometriosis—a pilot study. Contraception. 2009;79(1):29-34.
1. Missmer SA, Hankinson S, Spiegelman D, et al. The incidence of laparoscopically confirmed endometriosis by demographic, anthropomorphic and lifestyle factors. Am J Epidemiol. 2004;160(8):784-796.
2. Abdalla HI, Hart DM, Lindsay R, Leggate I, Hooke A. Prevention of bone mineral loss in postmenopausal women by norethisterone. Obstet Gynecol. 1985;66(6):789-792.
3. Riss BJ, Lehmann HJ, Christiansen C. Norethisterone acetate in combination with estrogen: effects on the skeleton and other organs. Am J Obstet Gynecol. 2002;187(4):1101-1116.
4. Hornstein MD, Surrey ES, Weisberg GW, Casino LA. Leuprolide acetate depot and hormonal add-back in endometriosis: a 12-month study. Lupron Add-back Study Group. Obstet Gynecol. 1998;91(1):16-24.
5. Vercellini P, Pietropauolo G, De Giorgi O, Pasin R, Chiodini A, Crosignani PG. Treatment of symptomatic rectovaginal endometriosis with an estrogen-progestogen combination versus low-dose norethindrone acetate. Fertil Steril. 2005;84(5):1375-1387.
6. Ferrero S, Camerini G, Ragni N, Venturini PL, Biscaldi E, Remorgida V. Norethisterone acetate in the treatment of colorectal endometriosis: a pilot study. Hum Reprod. 2010;25(1):94-100.
7. Ferrero S, Camerini G, Seracchioli R, Ragni N, Venturini PL, Remorgida V. Letrozole combined with norethisterone acetate compared with norethisterone acetate alone in the treatment of pain symptoms caused by endometriosis. Hum Reprod. 2009;24(12):3033-3341.
8. Kaser DJ, Missmer SA, Berry KF, Laufer MR. Use of norethindrone acetate alone for postoperative suppression of endometriosis symptoms [published online ahead of print December 9 2011]. J Pediatr Adolesc Gynecol. doi:10.1016/j.jpag.2011.09.013.
9. Schlaff WD, Carson SA, Luciano A, Ross D, Bergqvist A. Subcutaneous injection of depot medroxyprogesterone acetate compared with leuprolide acetate in the treatment of endometriosis associated pain. Fertil Steril. 2006;85(2):314-325.
10. Crosignani PG, Luciano A, Ray A, Bergqvist A. Subcutaneous depot medroxyprogesterone acetate versus leuprolide acetate in the treatment of endometriosis-associated pain. Hum Reprod. 2006;21(1):248-256.
11. Cheewadhanaraks S, Peeyananjarassri K, Choksuchat C, Dhanaworavibul K, Choobun T, Bunyapipat S. Interval of injections of intramuscular depot medroxyprogesterone acetate in the long-term treatment of endometriosis-associated pain: a randomized clinical trial. Gynecol Obstet Invest. 2009;68(2):116-121.
12. Luciano AA, Turksoy RN, Carleo J. Evaluation of oral medroxyprogesterone acetate in the treatment of endometriosis. Obstet Gynecol. 1988;72(3 Pt 1):323-327.
13. Wong AY, Tang LC, Chin RK. Levonorgestrel-releasing intrauterine system (Mirena) and depot medroxyprogesterone acetate (Depoprovera) as long-term maintenance therapy for patients with moderate and severe endometriosis: a randomized controlled trial. Aust N Z J Obstet Gynaecol. 2010;50(3):273-279.
14. Lockhat FB, Emembolu JO, Konje JC. The efficacy side-effects and continuation rates in women with symptomatic endometriosis undergoing treatment with an intrauterine administered progestogen (levonorgestrel): a 3 year follow-up. Hum Reprod. 2005;20(3):789-793.
15. Petta CA, Ferriani RA, Abrao MS, et al. Randomized clinical trial of a levonorgestrel-releasing intrauterine system and a depot GnRH analogue for the treatment of chronic pelvic pain in women with endometriosis. Hum Reprod. 2005;20(7):1993-1998.
16. Vercellini P, Aimi G, Panazza S, De Giorgi O, Pesole A, Crosignani PG. A levonorgestrel-releasing intrauterine system for the treatment of dysmenorrhea associated with endometriosis: a pilot study. Fertil Steril. 1999;72(3):505-508.
17. Vercellini P, Frontino G, De Giorgi O, Aimi G, Zaina B, Crosignani PG. Comparison of a levonorgestrel-releasing intrauterine device versus expectant management after conservative surgery for symptomatic endometriosis: a pilot study. Fertil Steril. 2003;80(2):305-309.
18. Walch K, Unfried G, Huber J, Kurz C, van Trotsenburg M, Pernicka E, Wenzl R. Implanon versus medroxyprogesterone acetate: effects on pain scores in patients with symptomatic endometriosis—a pilot study. Contraception. 2009;79(1):29-34.
Survivorship: Evaluating needs and the integrated model
Who is a survivor? Am I a survivor? Are you a survivor? What does a survivor need? How can community oncologists help? These are among the many questions a community oncologist can expect to hear during the course of treating a cancer patient, from the diagnosis, through the decisions about therapy, at all stages of treatment, and well into aftercare and follow-up. A “patient” used to be someone in active treatment, and a “survivor” was someone who had been free of disease for 5 years. More recently, Ellen Stoval of the National Coalition for Cancer Survivorship noted that the term “cancer survivor” can be used “to describe anyone who has been diagnosed with cancer as well as caregivers and loved ones of those diagnosed with the disease.”1 Her broader perspective more accurately captures the multifaceted nature of survivorship...
*For a PDF of the full article, click on the link to the left of this introduction.
Who is a survivor? Am I a survivor? Are you a survivor? What does a survivor need? How can community oncologists help? These are among the many questions a community oncologist can expect to hear during the course of treating a cancer patient, from the diagnosis, through the decisions about therapy, at all stages of treatment, and well into aftercare and follow-up. A “patient” used to be someone in active treatment, and a “survivor” was someone who had been free of disease for 5 years. More recently, Ellen Stoval of the National Coalition for Cancer Survivorship noted that the term “cancer survivor” can be used “to describe anyone who has been diagnosed with cancer as well as caregivers and loved ones of those diagnosed with the disease.”1 Her broader perspective more accurately captures the multifaceted nature of survivorship...
*For a PDF of the full article, click on the link to the left of this introduction.
Who is a survivor? Am I a survivor? Are you a survivor? What does a survivor need? How can community oncologists help? These are among the many questions a community oncologist can expect to hear during the course of treating a cancer patient, from the diagnosis, through the decisions about therapy, at all stages of treatment, and well into aftercare and follow-up. A “patient” used to be someone in active treatment, and a “survivor” was someone who had been free of disease for 5 years. More recently, Ellen Stoval of the National Coalition for Cancer Survivorship noted that the term “cancer survivor” can be used “to describe anyone who has been diagnosed with cancer as well as caregivers and loved ones of those diagnosed with the disease.”1 Her broader perspective more accurately captures the multifaceted nature of survivorship...
*For a PDF of the full article, click on the link to the left of this introduction.
Implications of Improved Survival in Patients With Chronic Myeloid Leukemia: A Nursing Perspective
With the introduction of tyrosine kinase inhibitor (TKI) therapy and the development of more sensitive monitoring techniques, the management of patients with chronic myeloid leukemia (CML) has evolved considerably over the last decade. In this review, we summarize the available literature evaluating the safety and efficacy of the TKIs imatinib, dasatinib, and nilotinib for information relevant to patient management to provide insight into long-term management of CML patients who receive TKI therapy. We suggest that these developments in treatment have expanded the role of oncology nurses, who can help address new issues that have arisen for patients learning to adapt to a chronic condition. The essential practice of monitoring, the critical importance of medication adherence, the safety profile of the three available TKIs, strategies for supportive care related to adverse events, drugdrug and drug-food interactions, and family planning are important aspects of long-term patient management...
*For a PDF of the full article, click on the link to the left of this introduction.
With the introduction of tyrosine kinase inhibitor (TKI) therapy and the development of more sensitive monitoring techniques, the management of patients with chronic myeloid leukemia (CML) has evolved considerably over the last decade. In this review, we summarize the available literature evaluating the safety and efficacy of the TKIs imatinib, dasatinib, and nilotinib for information relevant to patient management to provide insight into long-term management of CML patients who receive TKI therapy. We suggest that these developments in treatment have expanded the role of oncology nurses, who can help address new issues that have arisen for patients learning to adapt to a chronic condition. The essential practice of monitoring, the critical importance of medication adherence, the safety profile of the three available TKIs, strategies for supportive care related to adverse events, drugdrug and drug-food interactions, and family planning are important aspects of long-term patient management...
*For a PDF of the full article, click on the link to the left of this introduction.
With the introduction of tyrosine kinase inhibitor (TKI) therapy and the development of more sensitive monitoring techniques, the management of patients with chronic myeloid leukemia (CML) has evolved considerably over the last decade. In this review, we summarize the available literature evaluating the safety and efficacy of the TKIs imatinib, dasatinib, and nilotinib for information relevant to patient management to provide insight into long-term management of CML patients who receive TKI therapy. We suggest that these developments in treatment have expanded the role of oncology nurses, who can help address new issues that have arisen for patients learning to adapt to a chronic condition. The essential practice of monitoring, the critical importance of medication adherence, the safety profile of the three available TKIs, strategies for supportive care related to adverse events, drugdrug and drug-food interactions, and family planning are important aspects of long-term patient management...
*For a PDF of the full article, click on the link to the left of this introduction.
Maximizing clinical outcomes with axitinib therapy in advanced renal cell carcinoma through proactive side-effect management
Renal cell carcinoma (RCC) continues to exert a substantial disease burden. Increasing knowledge of the molecular signaling pathways associated with renal cancer has led to the development of targeted therapies for advanced RCC, including several antiangiogenic agents designed to inhibit development of abnormal blood vessels that sustain tumor growth. Axitinib is an investigational antiangiogenic agent that targets vascular endothelial growth factor receptors 1, 2, and 3. In phase II studies, axitinib elicited significant response rates in patients with advanced RCC refractory to cytokines or sorafenib. In a phase III study of axitinib versus sorafenib in patients with metastatic RCC, axitinib demonstrated clinically significant improvement in progression-free survival compared with sorafenib. As with other targeted agents, side effects associated with axitinib, such as hypertension, fatigue, and diarrhea, can negatively affect the patient’s physical and emotional states and quality of life, thus jeopardizing adherence to and the effectiveness of the treatment plan. Clinicians should be aware of side effects that may occur during treatment and manage them proactively. Nurses should educate patients about possible side effects and their management before axitinib treatment is initiated. Management strategies include early reporting of the symptoms, regular clinic visits and laboratory tests, ongoing review of concomitant medications, and prompt treatment of side effects and follow-up to assess the effectiveness of interventions, which could include treatment interruption and/or dose reduction. These approaches would help maximize the patient adherence to therapy, quality of life, and clinical outcomes.
*For a PDF of the full article, click on the link to the left of this introduction.
Renal cell carcinoma (RCC) continues to exert a substantial disease burden. Increasing knowledge of the molecular signaling pathways associated with renal cancer has led to the development of targeted therapies for advanced RCC, including several antiangiogenic agents designed to inhibit development of abnormal blood vessels that sustain tumor growth. Axitinib is an investigational antiangiogenic agent that targets vascular endothelial growth factor receptors 1, 2, and 3. In phase II studies, axitinib elicited significant response rates in patients with advanced RCC refractory to cytokines or sorafenib. In a phase III study of axitinib versus sorafenib in patients with metastatic RCC, axitinib demonstrated clinically significant improvement in progression-free survival compared with sorafenib. As with other targeted agents, side effects associated with axitinib, such as hypertension, fatigue, and diarrhea, can negatively affect the patient’s physical and emotional states and quality of life, thus jeopardizing adherence to and the effectiveness of the treatment plan. Clinicians should be aware of side effects that may occur during treatment and manage them proactively. Nurses should educate patients about possible side effects and their management before axitinib treatment is initiated. Management strategies include early reporting of the symptoms, regular clinic visits and laboratory tests, ongoing review of concomitant medications, and prompt treatment of side effects and follow-up to assess the effectiveness of interventions, which could include treatment interruption and/or dose reduction. These approaches would help maximize the patient adherence to therapy, quality of life, and clinical outcomes.
*For a PDF of the full article, click on the link to the left of this introduction.
Renal cell carcinoma (RCC) continues to exert a substantial disease burden. Increasing knowledge of the molecular signaling pathways associated with renal cancer has led to the development of targeted therapies for advanced RCC, including several antiangiogenic agents designed to inhibit development of abnormal blood vessels that sustain tumor growth. Axitinib is an investigational antiangiogenic agent that targets vascular endothelial growth factor receptors 1, 2, and 3. In phase II studies, axitinib elicited significant response rates in patients with advanced RCC refractory to cytokines or sorafenib. In a phase III study of axitinib versus sorafenib in patients with metastatic RCC, axitinib demonstrated clinically significant improvement in progression-free survival compared with sorafenib. As with other targeted agents, side effects associated with axitinib, such as hypertension, fatigue, and diarrhea, can negatively affect the patient’s physical and emotional states and quality of life, thus jeopardizing adherence to and the effectiveness of the treatment plan. Clinicians should be aware of side effects that may occur during treatment and manage them proactively. Nurses should educate patients about possible side effects and their management before axitinib treatment is initiated. Management strategies include early reporting of the symptoms, regular clinic visits and laboratory tests, ongoing review of concomitant medications, and prompt treatment of side effects and follow-up to assess the effectiveness of interventions, which could include treatment interruption and/or dose reduction. These approaches would help maximize the patient adherence to therapy, quality of life, and clinical outcomes.
*For a PDF of the full article, click on the link to the left of this introduction.
Smartphone Use During Rounds Comes Under Fire
Residents and attendings have divergent attitudes toward smartphone use during inpatient attending rounds, according to a researcher studying the topic.
Rachel Katz-Sidlow, MD, department of pediatrics, Jacobi Medical Center, Bronx, N.Y., is looking into the potential disadvantages of the phones if users are sending or receiving texts, emails, or pictures during rounds. Her preliminary research shows that a majority of faculty members believe a policy should be put in place to codify smartphone use.
“I truly think that policies to regulate smartphone use during patient management sessions are necessary, and will become commonplace in the near future until there are technology-driven solutions put into place,” says Dr. Katz-Sidlow.
Her research, currently in peer review for publication in the Journal of Hospital Medicine, comes as the ubiquity of smartphone technology has begun to be questioned by observers in the mainstream media. She says the majority of attendings and residents who responded to her initial questioning agree that smartphones “can be a serious source of distraction” during rounds.
The policy Dr. Katz-Sidlow is testing at Jacobi Medical Center defines a “smartphone” as any personal mobile communication device, including basic cellphones, Internet-enabled cellphones, and tablet computers. The beta policy restricts smartphone use during rounds to patient-care tasks, and would require all team members’ devices to be silenced or turned off at the beginning of rounds.
“Smartphones are here to stay and will become even more common in the future,” says Dr. Katz-Sidlow. “They’re such a valuable tool in medical education and patient care ... but there needs to be balance.”
Residents and attendings have divergent attitudes toward smartphone use during inpatient attending rounds, according to a researcher studying the topic.
Rachel Katz-Sidlow, MD, department of pediatrics, Jacobi Medical Center, Bronx, N.Y., is looking into the potential disadvantages of the phones if users are sending or receiving texts, emails, or pictures during rounds. Her preliminary research shows that a majority of faculty members believe a policy should be put in place to codify smartphone use.
“I truly think that policies to regulate smartphone use during patient management sessions are necessary, and will become commonplace in the near future until there are technology-driven solutions put into place,” says Dr. Katz-Sidlow.
Her research, currently in peer review for publication in the Journal of Hospital Medicine, comes as the ubiquity of smartphone technology has begun to be questioned by observers in the mainstream media. She says the majority of attendings and residents who responded to her initial questioning agree that smartphones “can be a serious source of distraction” during rounds.
The policy Dr. Katz-Sidlow is testing at Jacobi Medical Center defines a “smartphone” as any personal mobile communication device, including basic cellphones, Internet-enabled cellphones, and tablet computers. The beta policy restricts smartphone use during rounds to patient-care tasks, and would require all team members’ devices to be silenced or turned off at the beginning of rounds.
“Smartphones are here to stay and will become even more common in the future,” says Dr. Katz-Sidlow. “They’re such a valuable tool in medical education and patient care ... but there needs to be balance.”
Residents and attendings have divergent attitudes toward smartphone use during inpatient attending rounds, according to a researcher studying the topic.
Rachel Katz-Sidlow, MD, department of pediatrics, Jacobi Medical Center, Bronx, N.Y., is looking into the potential disadvantages of the phones if users are sending or receiving texts, emails, or pictures during rounds. Her preliminary research shows that a majority of faculty members believe a policy should be put in place to codify smartphone use.
“I truly think that policies to regulate smartphone use during patient management sessions are necessary, and will become commonplace in the near future until there are technology-driven solutions put into place,” says Dr. Katz-Sidlow.
Her research, currently in peer review for publication in the Journal of Hospital Medicine, comes as the ubiquity of smartphone technology has begun to be questioned by observers in the mainstream media. She says the majority of attendings and residents who responded to her initial questioning agree that smartphones “can be a serious source of distraction” during rounds.
The policy Dr. Katz-Sidlow is testing at Jacobi Medical Center defines a “smartphone” as any personal mobile communication device, including basic cellphones, Internet-enabled cellphones, and tablet computers. The beta policy restricts smartphone use during rounds to patient-care tasks, and would require all team members’ devices to be silenced or turned off at the beginning of rounds.
“Smartphones are here to stay and will become even more common in the future,” says Dr. Katz-Sidlow. “They’re such a valuable tool in medical education and patient care ... but there needs to be balance.”
Hospitalist/Palliative-Care Collaboration Aims to Reduce Readmissions
A poster presented at the Center to Advance Palliative Care national seminar in San Diego in November described a growing collaboration between the HM service and the palliative-care team at a Wisconsin medical center as part of efforts to control readmissions using tools from SHM’s Project BOOST (Better Outcomes for Older Adults through Safe Transitions).
On admission to 227-bed Aurora West Allis (Wis.) Medical Center, all patients assigned to a hospitalist are assessed for readmission risk factors. If a risk factor is identified, the hospital has established a set of specific interventions, including a palliative-care consultation.
“Basically, we took the BOOST tools and developed a physician action plan behind those risk factors,” says Andrew McDonagh, MD, head of the center’s hospitalist service.
Dr. McDonagh started Aurora West Allis’ HM service in 2008, and Timothy Jessick, DO, initiated the palliative-care service in 2010. “As our programs grew together, it became apparent that there were significant synergies between the two specialties, so we took the opportunity to work together in several ways,” Dr. McDonagh says.
A Palliative Care Quality Indicators Checklist, which looks for four key clinical indicators, triggers hospitalists and unit nurses to order the palliative consult. If an elderly patient has multiple admissions for the same diagnosis, the second admission triggers a geriatric consultation, and the third admission gets a palliative-care consult.
Hospitalists at the medical center are given education and modeling on how to hold family conferences with patients and their families to elicit their goals of care. Collaboration between HM and palliative care is spreading to the hospital’s ICUs, to patients transitioning out of the hospital to nursing homes, and to two other Aurora hospitals in the Milwaukee area, Dr. McDonagh explains. Since the BOOST tools have been implemented, preliminary evidence points to reduced readmissions, increased patient satisfaction, and increased palliative-care consults at the hospital.
“In the future, doing our job well as hospitalists will be more than just addressing medical needs but tailoring our care plans to the individual patient. Palliative care helps us better define appropriate care for these patients, looking beyond the trees for the forest,” he says. “I believe I’m a better clinician for being part of this relationship.”
A poster presented at the Center to Advance Palliative Care national seminar in San Diego in November described a growing collaboration between the HM service and the palliative-care team at a Wisconsin medical center as part of efforts to control readmissions using tools from SHM’s Project BOOST (Better Outcomes for Older Adults through Safe Transitions).
On admission to 227-bed Aurora West Allis (Wis.) Medical Center, all patients assigned to a hospitalist are assessed for readmission risk factors. If a risk factor is identified, the hospital has established a set of specific interventions, including a palliative-care consultation.
“Basically, we took the BOOST tools and developed a physician action plan behind those risk factors,” says Andrew McDonagh, MD, head of the center’s hospitalist service.
Dr. McDonagh started Aurora West Allis’ HM service in 2008, and Timothy Jessick, DO, initiated the palliative-care service in 2010. “As our programs grew together, it became apparent that there were significant synergies between the two specialties, so we took the opportunity to work together in several ways,” Dr. McDonagh says.
A Palliative Care Quality Indicators Checklist, which looks for four key clinical indicators, triggers hospitalists and unit nurses to order the palliative consult. If an elderly patient has multiple admissions for the same diagnosis, the second admission triggers a geriatric consultation, and the third admission gets a palliative-care consult.
Hospitalists at the medical center are given education and modeling on how to hold family conferences with patients and their families to elicit their goals of care. Collaboration between HM and palliative care is spreading to the hospital’s ICUs, to patients transitioning out of the hospital to nursing homes, and to two other Aurora hospitals in the Milwaukee area, Dr. McDonagh explains. Since the BOOST tools have been implemented, preliminary evidence points to reduced readmissions, increased patient satisfaction, and increased palliative-care consults at the hospital.
“In the future, doing our job well as hospitalists will be more than just addressing medical needs but tailoring our care plans to the individual patient. Palliative care helps us better define appropriate care for these patients, looking beyond the trees for the forest,” he says. “I believe I’m a better clinician for being part of this relationship.”
A poster presented at the Center to Advance Palliative Care national seminar in San Diego in November described a growing collaboration between the HM service and the palliative-care team at a Wisconsin medical center as part of efforts to control readmissions using tools from SHM’s Project BOOST (Better Outcomes for Older Adults through Safe Transitions).
On admission to 227-bed Aurora West Allis (Wis.) Medical Center, all patients assigned to a hospitalist are assessed for readmission risk factors. If a risk factor is identified, the hospital has established a set of specific interventions, including a palliative-care consultation.
“Basically, we took the BOOST tools and developed a physician action plan behind those risk factors,” says Andrew McDonagh, MD, head of the center’s hospitalist service.
Dr. McDonagh started Aurora West Allis’ HM service in 2008, and Timothy Jessick, DO, initiated the palliative-care service in 2010. “As our programs grew together, it became apparent that there were significant synergies between the two specialties, so we took the opportunity to work together in several ways,” Dr. McDonagh says.
A Palliative Care Quality Indicators Checklist, which looks for four key clinical indicators, triggers hospitalists and unit nurses to order the palliative consult. If an elderly patient has multiple admissions for the same diagnosis, the second admission triggers a geriatric consultation, and the third admission gets a palliative-care consult.
Hospitalists at the medical center are given education and modeling on how to hold family conferences with patients and their families to elicit their goals of care. Collaboration between HM and palliative care is spreading to the hospital’s ICUs, to patients transitioning out of the hospital to nursing homes, and to two other Aurora hospitals in the Milwaukee area, Dr. McDonagh explains. Since the BOOST tools have been implemented, preliminary evidence points to reduced readmissions, increased patient satisfaction, and increased palliative-care consults at the hospital.
“In the future, doing our job well as hospitalists will be more than just addressing medical needs but tailoring our care plans to the individual patient. Palliative care helps us better define appropriate care for these patients, looking beyond the trees for the forest,” he says. “I believe I’m a better clinician for being part of this relationship.”
FDA Approves Linagliptin/Metformin Combo For Type 2 Diabetes
The Food and Drug Administration on Jan. 30 approved a type 2 diabetes therapy that combines linagliptin and metformin in a single tablet taken twice daily.*
The approval was announced by Eli Lilly and Boehringer Ingelheim. The companies jointly developed the tablet, to be marketed as Jentadueto. Linagliptin is a dipeptidyl peptidase–4 (DPP-4) inhibitor that was approved for marketing in May 2011.
The linagliptin/metformin combination is intended to be used in conjunction with diet and exercise to improve glycemic control. Jentadueto was approved based on a 24-week, double-blind, placebo-controlled study that compared six arms: placebo, linagliptin once daily; 500 mg metformin twice daily; 2.5 mg linagliptin plus a 500-mg tablet of metformin twice daily; 1,000 mg metformin twice daily; and 2.5 mg linagliptin plus 1,000 mg metformin twice daily.
The actual combination tablet was not tested, but the FDA approved it based on bioequivalence studies, according to Lilly. The 2.5-mg linagliptin plus 1,000-mg metformin arm had the best results, reducing hemoglobin A1c by up to 1.7%. The two tablets did not cause any meaningful changes in body weight, according to a company statement.
Lilly said that Jentadueto can be used alone or in combination with a sulfonylurea. It is not indicated for type 1 diabetes or diabetic ketoacidosis. Jentadueto has a boxed warning for the risk of lactic acidosis.
*Correction, 1/31/12: An earlier version of this story noted the incorrect approval date.
The Food and Drug Administration on Jan. 30 approved a type 2 diabetes therapy that combines linagliptin and metformin in a single tablet taken twice daily.*
The approval was announced by Eli Lilly and Boehringer Ingelheim. The companies jointly developed the tablet, to be marketed as Jentadueto. Linagliptin is a dipeptidyl peptidase–4 (DPP-4) inhibitor that was approved for marketing in May 2011.
The linagliptin/metformin combination is intended to be used in conjunction with diet and exercise to improve glycemic control. Jentadueto was approved based on a 24-week, double-blind, placebo-controlled study that compared six arms: placebo, linagliptin once daily; 500 mg metformin twice daily; 2.5 mg linagliptin plus a 500-mg tablet of metformin twice daily; 1,000 mg metformin twice daily; and 2.5 mg linagliptin plus 1,000 mg metformin twice daily.
The actual combination tablet was not tested, but the FDA approved it based on bioequivalence studies, according to Lilly. The 2.5-mg linagliptin plus 1,000-mg metformin arm had the best results, reducing hemoglobin A1c by up to 1.7%. The two tablets did not cause any meaningful changes in body weight, according to a company statement.
Lilly said that Jentadueto can be used alone or in combination with a sulfonylurea. It is not indicated for type 1 diabetes or diabetic ketoacidosis. Jentadueto has a boxed warning for the risk of lactic acidosis.
*Correction, 1/31/12: An earlier version of this story noted the incorrect approval date.
The Food and Drug Administration on Jan. 30 approved a type 2 diabetes therapy that combines linagliptin and metformin in a single tablet taken twice daily.*
The approval was announced by Eli Lilly and Boehringer Ingelheim. The companies jointly developed the tablet, to be marketed as Jentadueto. Linagliptin is a dipeptidyl peptidase–4 (DPP-4) inhibitor that was approved for marketing in May 2011.
The linagliptin/metformin combination is intended to be used in conjunction with diet and exercise to improve glycemic control. Jentadueto was approved based on a 24-week, double-blind, placebo-controlled study that compared six arms: placebo, linagliptin once daily; 500 mg metformin twice daily; 2.5 mg linagliptin plus a 500-mg tablet of metformin twice daily; 1,000 mg metformin twice daily; and 2.5 mg linagliptin plus 1,000 mg metformin twice daily.
The actual combination tablet was not tested, but the FDA approved it based on bioequivalence studies, according to Lilly. The 2.5-mg linagliptin plus 1,000-mg metformin arm had the best results, reducing hemoglobin A1c by up to 1.7%. The two tablets did not cause any meaningful changes in body weight, according to a company statement.
Lilly said that Jentadueto can be used alone or in combination with a sulfonylurea. It is not indicated for type 1 diabetes or diabetic ketoacidosis. Jentadueto has a boxed warning for the risk of lactic acidosis.
*Correction, 1/31/12: An earlier version of this story noted the incorrect approval date.
Burn Size Remains Strongest Predictor of Pediatric Survival
For pediatric burn patients, the percentage of total body surface area affected remains the strongest predictor of survival, according to a report published online Jan. 31 in the Lancet.
"We have established that, in a modern pediatric burn care setting, a burn size of roughly 60% total body surface area is a crucial threshold for postburn morbidity and mortality," wrote Dr. Robert Kraft of Shriners Hospitals for Children, Galveston, Texas, and his associates.
For decades, burn size has been the main prognostic factor for both adults and children with burn injuries. But recent improvements in burn care – including novel drug treatments, new grafting techniques and materials, and improved life-support systems and monitoring methods – have dramatically improved survival, according to the investigators. Because treatment decisions, including whether to transfer a patient to a specialized burn center, are based on the probability of survival, it was important to determine whether burn size was still predictive of major complications and mortality under current treatment conditions.
To do so, Dr. Kraft and his colleagues examined outcomes in all 952 patients admitted over a 1-year period to Galveston’s Shriners Hospital with burns involving 30% or more of their total body surface area. Most of the study patients were burned on 40%-49% of their bodies.
Burn size proved to be the strongest predictor of survival, with mortality rising significantly as burn size increased. "In patient groups with burns smaller than 60% total body surface area, there were only minor increases in mortality starting at 3% and reaching up to 7%," the investigators wrote.
A large increase in mortality was noted when burn size reached 62% of the total body surface area. Such patients had a tenfold higher risk of death, compared with those who had smaller burns, the investigators reported (Lancet 2012 [doi:10.1016/S0140-6736(11)61345-7]).
Large burns also were associated with significant increases in multiorgan failure, infection during ICU stay, and the need for more surgeries to excise tissue. The rate of multiorgan failure was 6%-12% and the rate of sepsis was only 2%-6% for burns involving up to 59% of the body surface area. These rates ballooned as high as 27%-45% for multiorgan failure and 15%-26% for sepsis when burns involved 60% or more of the body surface area.
In addition, blood glucose and insulin levels rose significantly with burns involving 60% or more of the total body surface area, as did resting energy expenditure. Together with concomitant changes in liver structure and function, these findings reflect a massive hypermetabolic response to burn injury, Dr. Kraft and his associates reported.
Cytokine levels also differed by size of burn. With burns involving 60% or more of the body surface area, the levels of interleukin-6, -8, -10, and -13; macrophage inflammatory protein 1beta; tumor necrosis factor–alpha; granulocyte colony-stimulating factor; interferon-gamma; granulocyte-macrophage colony stimulating factor; and C-reactive protein rose substantially for an extended period of time.
Given these findings, "we recommend that pediatric patients with greater than 60% total body surface area burns be immediately transferred to a specialized burn center. Furthermore, at the burn center, patients should be treated with increased vigilance and improved therapies, in view of the increased risk of poor outcome associated with this burn size," the researchers wrote.
After burn size, the presence of inhalation injury in addition to external burn injury was a significant predictor of mortality. Patients who sustained inhalation injury had a threefold higher risk of death than did patients with no inhalation injuries. "We recommend that the treating physician [keep] in mind that the presence of inhalation injury significantly affects postburn outcomes," they noted.
Patient age and sex showed no association with survival, and neither did the time interval between sustaining the burn and presenting for admission.
It was noteworthy that mortality rates in this study were relatively low, even among patients who had the most extensive burns. "In our opinion, [high survival was] attributable to an improved and aggressive treatment regimen," they added.
This study was supported by Shriners Hospitals for Children, the National Institutes of Health, the National Institute on Disability and Rehabilitation Research, the Institute for Translational Sciences, the CFI Leaders Opportunity fund, and Physicians’ Services Incorporated Foundation. The investigators reported having no financial conflicts of interest.
This study shows that children with burns involving less than 62% of total body surface area can be treated successfully with standard procedures, but larger burns require the novel surgical wound management and advanced monitoring available at specialized burn centers, commented Dr. Ronald G. Tompkins.
It is also important to note that overall survival in this study was quite high. Even among children with burns involving 90% of total body surface area, half survived their injuries. And even the youngest children can be expected to survive as well as older children, now that pediatric intensive care has learned to compensate for their physiological and anatomical differences.
This means it is time to think beyond mere survival to survivors’ quality of life, he noted.
Dr. Tompkins is chief of the burn service at Massachusetts General Hospital and the Sumner M. Redstone Professor of Surgery at Harvard Medical School, both in Boston. He reported no financial conflicts of interest. These remarks were adapted from his editorial comment accompanying Dr. Kraft’s report (Lancet 2012 [doi:10.1016/S0140-6736(11)61626-7]).
This study shows that children with burns involving less than 62% of total body surface area can be treated successfully with standard procedures, but larger burns require the novel surgical wound management and advanced monitoring available at specialized burn centers, commented Dr. Ronald G. Tompkins.
It is also important to note that overall survival in this study was quite high. Even among children with burns involving 90% of total body surface area, half survived their injuries. And even the youngest children can be expected to survive as well as older children, now that pediatric intensive care has learned to compensate for their physiological and anatomical differences.
This means it is time to think beyond mere survival to survivors’ quality of life, he noted.
Dr. Tompkins is chief of the burn service at Massachusetts General Hospital and the Sumner M. Redstone Professor of Surgery at Harvard Medical School, both in Boston. He reported no financial conflicts of interest. These remarks were adapted from his editorial comment accompanying Dr. Kraft’s report (Lancet 2012 [doi:10.1016/S0140-6736(11)61626-7]).
This study shows that children with burns involving less than 62% of total body surface area can be treated successfully with standard procedures, but larger burns require the novel surgical wound management and advanced monitoring available at specialized burn centers, commented Dr. Ronald G. Tompkins.
It is also important to note that overall survival in this study was quite high. Even among children with burns involving 90% of total body surface area, half survived their injuries. And even the youngest children can be expected to survive as well as older children, now that pediatric intensive care has learned to compensate for their physiological and anatomical differences.
This means it is time to think beyond mere survival to survivors’ quality of life, he noted.
Dr. Tompkins is chief of the burn service at Massachusetts General Hospital and the Sumner M. Redstone Professor of Surgery at Harvard Medical School, both in Boston. He reported no financial conflicts of interest. These remarks were adapted from his editorial comment accompanying Dr. Kraft’s report (Lancet 2012 [doi:10.1016/S0140-6736(11)61626-7]).
For pediatric burn patients, the percentage of total body surface area affected remains the strongest predictor of survival, according to a report published online Jan. 31 in the Lancet.
"We have established that, in a modern pediatric burn care setting, a burn size of roughly 60% total body surface area is a crucial threshold for postburn morbidity and mortality," wrote Dr. Robert Kraft of Shriners Hospitals for Children, Galveston, Texas, and his associates.
For decades, burn size has been the main prognostic factor for both adults and children with burn injuries. But recent improvements in burn care – including novel drug treatments, new grafting techniques and materials, and improved life-support systems and monitoring methods – have dramatically improved survival, according to the investigators. Because treatment decisions, including whether to transfer a patient to a specialized burn center, are based on the probability of survival, it was important to determine whether burn size was still predictive of major complications and mortality under current treatment conditions.
To do so, Dr. Kraft and his colleagues examined outcomes in all 952 patients admitted over a 1-year period to Galveston’s Shriners Hospital with burns involving 30% or more of their total body surface area. Most of the study patients were burned on 40%-49% of their bodies.
Burn size proved to be the strongest predictor of survival, with mortality rising significantly as burn size increased. "In patient groups with burns smaller than 60% total body surface area, there were only minor increases in mortality starting at 3% and reaching up to 7%," the investigators wrote.
A large increase in mortality was noted when burn size reached 62% of the total body surface area. Such patients had a tenfold higher risk of death, compared with those who had smaller burns, the investigators reported (Lancet 2012 [doi:10.1016/S0140-6736(11)61345-7]).
Large burns also were associated with significant increases in multiorgan failure, infection during ICU stay, and the need for more surgeries to excise tissue. The rate of multiorgan failure was 6%-12% and the rate of sepsis was only 2%-6% for burns involving up to 59% of the body surface area. These rates ballooned as high as 27%-45% for multiorgan failure and 15%-26% for sepsis when burns involved 60% or more of the body surface area.
In addition, blood glucose and insulin levels rose significantly with burns involving 60% or more of the total body surface area, as did resting energy expenditure. Together with concomitant changes in liver structure and function, these findings reflect a massive hypermetabolic response to burn injury, Dr. Kraft and his associates reported.
Cytokine levels also differed by size of burn. With burns involving 60% or more of the body surface area, the levels of interleukin-6, -8, -10, and -13; macrophage inflammatory protein 1beta; tumor necrosis factor–alpha; granulocyte colony-stimulating factor; interferon-gamma; granulocyte-macrophage colony stimulating factor; and C-reactive protein rose substantially for an extended period of time.
Given these findings, "we recommend that pediatric patients with greater than 60% total body surface area burns be immediately transferred to a specialized burn center. Furthermore, at the burn center, patients should be treated with increased vigilance and improved therapies, in view of the increased risk of poor outcome associated with this burn size," the researchers wrote.
After burn size, the presence of inhalation injury in addition to external burn injury was a significant predictor of mortality. Patients who sustained inhalation injury had a threefold higher risk of death than did patients with no inhalation injuries. "We recommend that the treating physician [keep] in mind that the presence of inhalation injury significantly affects postburn outcomes," they noted.
Patient age and sex showed no association with survival, and neither did the time interval between sustaining the burn and presenting for admission.
It was noteworthy that mortality rates in this study were relatively low, even among patients who had the most extensive burns. "In our opinion, [high survival was] attributable to an improved and aggressive treatment regimen," they added.
This study was supported by Shriners Hospitals for Children, the National Institutes of Health, the National Institute on Disability and Rehabilitation Research, the Institute for Translational Sciences, the CFI Leaders Opportunity fund, and Physicians’ Services Incorporated Foundation. The investigators reported having no financial conflicts of interest.
For pediatric burn patients, the percentage of total body surface area affected remains the strongest predictor of survival, according to a report published online Jan. 31 in the Lancet.
"We have established that, in a modern pediatric burn care setting, a burn size of roughly 60% total body surface area is a crucial threshold for postburn morbidity and mortality," wrote Dr. Robert Kraft of Shriners Hospitals for Children, Galveston, Texas, and his associates.
For decades, burn size has been the main prognostic factor for both adults and children with burn injuries. But recent improvements in burn care – including novel drug treatments, new grafting techniques and materials, and improved life-support systems and monitoring methods – have dramatically improved survival, according to the investigators. Because treatment decisions, including whether to transfer a patient to a specialized burn center, are based on the probability of survival, it was important to determine whether burn size was still predictive of major complications and mortality under current treatment conditions.
To do so, Dr. Kraft and his colleagues examined outcomes in all 952 patients admitted over a 1-year period to Galveston’s Shriners Hospital with burns involving 30% or more of their total body surface area. Most of the study patients were burned on 40%-49% of their bodies.
Burn size proved to be the strongest predictor of survival, with mortality rising significantly as burn size increased. "In patient groups with burns smaller than 60% total body surface area, there were only minor increases in mortality starting at 3% and reaching up to 7%," the investigators wrote.
A large increase in mortality was noted when burn size reached 62% of the total body surface area. Such patients had a tenfold higher risk of death, compared with those who had smaller burns, the investigators reported (Lancet 2012 [doi:10.1016/S0140-6736(11)61345-7]).
Large burns also were associated with significant increases in multiorgan failure, infection during ICU stay, and the need for more surgeries to excise tissue. The rate of multiorgan failure was 6%-12% and the rate of sepsis was only 2%-6% for burns involving up to 59% of the body surface area. These rates ballooned as high as 27%-45% for multiorgan failure and 15%-26% for sepsis when burns involved 60% or more of the body surface area.
In addition, blood glucose and insulin levels rose significantly with burns involving 60% or more of the total body surface area, as did resting energy expenditure. Together with concomitant changes in liver structure and function, these findings reflect a massive hypermetabolic response to burn injury, Dr. Kraft and his associates reported.
Cytokine levels also differed by size of burn. With burns involving 60% or more of the body surface area, the levels of interleukin-6, -8, -10, and -13; macrophage inflammatory protein 1beta; tumor necrosis factor–alpha; granulocyte colony-stimulating factor; interferon-gamma; granulocyte-macrophage colony stimulating factor; and C-reactive protein rose substantially for an extended period of time.
Given these findings, "we recommend that pediatric patients with greater than 60% total body surface area burns be immediately transferred to a specialized burn center. Furthermore, at the burn center, patients should be treated with increased vigilance and improved therapies, in view of the increased risk of poor outcome associated with this burn size," the researchers wrote.
After burn size, the presence of inhalation injury in addition to external burn injury was a significant predictor of mortality. Patients who sustained inhalation injury had a threefold higher risk of death than did patients with no inhalation injuries. "We recommend that the treating physician [keep] in mind that the presence of inhalation injury significantly affects postburn outcomes," they noted.
Patient age and sex showed no association with survival, and neither did the time interval between sustaining the burn and presenting for admission.
It was noteworthy that mortality rates in this study were relatively low, even among patients who had the most extensive burns. "In our opinion, [high survival was] attributable to an improved and aggressive treatment regimen," they added.
This study was supported by Shriners Hospitals for Children, the National Institutes of Health, the National Institute on Disability and Rehabilitation Research, the Institute for Translational Sciences, the CFI Leaders Opportunity fund, and Physicians’ Services Incorporated Foundation. The investigators reported having no financial conflicts of interest.
FROM LANCET
Major Finding: Larger burn size correlated with poorer prognosis, with mortality rising tenfold in patients who had 62% or more of total body surface area involved.
Data Source: A single-center, observational cohort study of 952 pediatric patients with burns covering at least 30% of their total body surface area who were treated over a 1-year period.
Disclosures: This study was supported by Shriners Hospitals for Children, the National Institutes of Health, the National Institute on Disability and Rehabilitation Research, the Institute for Translational Sciences, the CFI Leaders Opportunity fund, and Physicians’ Services Incorporated Foundation. The investigators reported having no financial conflicts of interest.