User login
The Official Newspaper of the American Association for Thoracic Surgery
TAVR Usable in Older, High-Risk Patients
NEW ORLEANS Transcatheter aortic valve replacement was associated with the same survival rates at 1 year as was found in conventional surgery for older, high-risk patients who suffered from severe, symptomatic aortic stenosis, according to the results of the PARTNER trial.
All-cause mortality at 30 days favored the use of transcatheter aortic valve replacement (TAVR) at 3.4%, compared with 6.5% for open surgery, and was similar at 1 year (24% vs, 27%, respectively).
The difference at 1 year reached the trial's prespecified noninferiority margin with a P value of .001.
"We already learned from the previous cohort that TAVR is the standard of care for patients who can't tolerate surgery. So this [finding] opens up a new set of patients who may very well benefit as much with TAVR as with the gold standard surgery," coprincipal investigator Dr. Craig B. Smith said at the annual meeting of the American College of Cardiology.
However, The stroke rate with TAVR as seen in the industry-sponsored PARTNER (Placement of Aortic Transcatheter Valve) trial, turned out to be twice the rate for these patients as that observed with conventional surgical repair.
Stroke plus transient ischemic attack (TIA) rates were significantly more frequent with TAVR than surgery at both 30 days (5.5% vs. 2.4%) and 1 year (8.3% vs. 4.3%).
When only major strokes were compared, the difference was not significant between TAVR and convential surgery at 30 days (3.8% vs. 2.1%) or 1 year (5.1% vs. 2.4%), said Dr. Smith, chief of cardiothoracic surgery at New York Presbyterian Hospital/Columbia University Medical Center in New York City.
Notably, patients who un?derwent conventional surgery were significantly more likely than TAVR patients to experience major bleeding at 30 days (19.5% vs. 9.3%) and 1 year (26% vs. 15%), as well as new atrial fibrillation at both 30 days (16% vs. 8.6%) and 1 year (17% vs. 12%).
An examination of echocardiographic findings indicated a small hemodynamic benefit ocurred with TAVR vs. surgery at 1 year, but there was significantly increased paravalvular aortic reÂgurgitation at 30 days, 6 months, and 1 year.
"TAVR and AVR [aortic valve replacement] are both acceptable therapies in these high-risk patients; differing periprocedural hazards should influence case-based decision making," Dr. Smith said.
Dr. David Moliterno, chair of the intervention program (i2 Summit) at the meeting, told reporters that they were witnessing histoÂry in the making.
"This will probably be seen as one of the biggest steps in cardiovascular medicine, as far as intervention is concerned, potentially in our lifetime," said Dr. Moliterno, chief of cardiovascular medicine at the University of Kentucky, Lexington.
"If we look back to balloon angioÂplasty, the advent of stents and drug-eluting stents, this will be seen as the next major turning point."
Last year, the PARTNER trial investigators reported a 20% survival benefit at 1 year in a separate cohort of 358 inoperable patients with severe aortic stenosis who underwent transfemoral TAVR vs. standard therapy, including balloon aortic valvuloplasty (N. Engl. J. Med. 2010;363:1597-607). TAVR was associated with significantly more major vascular complications (16% vs. 1%) and a higher incidence of major strokes (5% vs. 1%).
The current portion of PARTNER randomly assigned 699 patients at 26 centers to surgery or TAVR. These patients were treated using the transfemoral approach in 492 cases and the transapical approach in 207.
Severe stenosis was defined as an aortic valve area of less than 0.8 cm2 and mean aortic valve gradient of more than 40 mm Hg or a peak aortic jet velocity of more than 4.0 m/second.
The patients in the trial had a New York Heart Association heart failure class II or greater, and they also had a predicted risk of operative mortality of at least 15% as determined by the site surgeon and cardiologist, coupled with a Society of Thoracic Surgery (STS) score of at least 10.
The mean age of the patients in the study was 83 years, and 94% of them were NYHA class III or IV. In all, 42 patients were not treated as assigned, Dr. Smith said.
All-cause mortality at 1 year was simÂilar for the transfemoral TAVR subgroup, at 22.2% and 26.4% for surgery, as well as for the transapical TAVR subgroup (29% vs. 28%, respectively).
A preliminary subgroup analysis sugÂgested that there may be a possible benefit found with TAVR in women and in patients without prior coronary artery bypass surgery, but Dr. Smith said that those results should be interÂpreted cautiously.
Overall, the TAVR mortality of 3.4% at 30 days was the lowest reported in any series, despite the use of an early-generation device and limited previous operator experience, he said, noting that nine of the participating centers had never performed TAVR before the trial.
Symptom improvement, including NYHA class and 6-minute walk distance, favored TAVR at 30 days and was similar to conventional surgery at 1 year, Dr. Moliterno said.
Coinvestigator Dr. Murat Tuzcu, vice chair of the department of cardiovascu?lar medicine at the Cleveland Clinic, stressed that the results were accomplished only through unprecedented teamwork among cardiologists, surgeons, and imaging experts, who conÂsulted on each case in the trial.
"I want to emphasize that if we fail to pay equal attention to what we have done in this trial after the device is approved, I don't think we will be able to replicate the same results," he said.
Several speakers at the press conference noted the balancing act that all clinicians will face regarding public demand for the new procedure.
Dr. Michael Crawford, chief of clinical cardiology at the University of California, San Francisco, observed that centers will face substantial up-front costs in the adoption of TAVR, including hybrid surgical/interventional suites, and that this will restrict initial uptake to high-volume centers of excellence.
The 3.4% mortality rate represents an “amazing effort, particularly in the use of the larger, first-generation device, said invited discussant Dr. Martyn Thomas, clinical director of cardiothoracic services at St. Thomas Hospital, London.
The delivery catheters used in PARTNER are sized at 22 and 24 French, which corresponds to an outside diamÂeter of 7-8 mm.
The size of catheters has been decreased down to 16-Fr in Europe, where more than 5,000 TAVR procedures have been done and where both the Edwards Lifesciences Corp.'s Sapien valve that was used in PARTNER and the Medtronic Inc.'s CoreValve are already approved for commercial use.
Recruitment began in February for the PARTNER II trial that is evaluating the smaller Edwards Sapien XT device and the later generation NovaFlex delivery system among inoperable patients with symptomatic severe aortic stenosis. The trial is expected to enroll 600 patients, and is targeted for primary completion in December 2011.
When asked whether the advent of newer, thinner devices will mean adoption in patients with lower risk, Dr. Smith replied that the adoption of TAVR will "march steadily down the risk categories."
Edwards Lifesciences sponsored the trial. Dr. Smith and his coauthors reported no conflicts of interest.
NEW ORLEANS Transcatheter aortic valve replacement was associated with the same survival rates at 1 year as was found in conventional surgery for older, high-risk patients who suffered from severe, symptomatic aortic stenosis, according to the results of the PARTNER trial.
All-cause mortality at 30 days favored the use of transcatheter aortic valve replacement (TAVR) at 3.4%, compared with 6.5% for open surgery, and was similar at 1 year (24% vs, 27%, respectively).
The difference at 1 year reached the trial's prespecified noninferiority margin with a P value of .001.
"We already learned from the previous cohort that TAVR is the standard of care for patients who can't tolerate surgery. So this [finding] opens up a new set of patients who may very well benefit as much with TAVR as with the gold standard surgery," coprincipal investigator Dr. Craig B. Smith said at the annual meeting of the American College of Cardiology.
However, The stroke rate with TAVR as seen in the industry-sponsored PARTNER (Placement of Aortic Transcatheter Valve) trial, turned out to be twice the rate for these patients as that observed with conventional surgical repair.
Stroke plus transient ischemic attack (TIA) rates were significantly more frequent with TAVR than surgery at both 30 days (5.5% vs. 2.4%) and 1 year (8.3% vs. 4.3%).
When only major strokes were compared, the difference was not significant between TAVR and convential surgery at 30 days (3.8% vs. 2.1%) or 1 year (5.1% vs. 2.4%), said Dr. Smith, chief of cardiothoracic surgery at New York Presbyterian Hospital/Columbia University Medical Center in New York City.
Notably, patients who un?derwent conventional surgery were significantly more likely than TAVR patients to experience major bleeding at 30 days (19.5% vs. 9.3%) and 1 year (26% vs. 15%), as well as new atrial fibrillation at both 30 days (16% vs. 8.6%) and 1 year (17% vs. 12%).
An examination of echocardiographic findings indicated a small hemodynamic benefit ocurred with TAVR vs. surgery at 1 year, but there was significantly increased paravalvular aortic reÂgurgitation at 30 days, 6 months, and 1 year.
"TAVR and AVR [aortic valve replacement] are both acceptable therapies in these high-risk patients; differing periprocedural hazards should influence case-based decision making," Dr. Smith said.
Dr. David Moliterno, chair of the intervention program (i2 Summit) at the meeting, told reporters that they were witnessing histoÂry in the making.
"This will probably be seen as one of the biggest steps in cardiovascular medicine, as far as intervention is concerned, potentially in our lifetime," said Dr. Moliterno, chief of cardiovascular medicine at the University of Kentucky, Lexington.
"If we look back to balloon angioÂplasty, the advent of stents and drug-eluting stents, this will be seen as the next major turning point."
Last year, the PARTNER trial investigators reported a 20% survival benefit at 1 year in a separate cohort of 358 inoperable patients with severe aortic stenosis who underwent transfemoral TAVR vs. standard therapy, including balloon aortic valvuloplasty (N. Engl. J. Med. 2010;363:1597-607). TAVR was associated with significantly more major vascular complications (16% vs. 1%) and a higher incidence of major strokes (5% vs. 1%).
The current portion of PARTNER randomly assigned 699 patients at 26 centers to surgery or TAVR. These patients were treated using the transfemoral approach in 492 cases and the transapical approach in 207.
Severe stenosis was defined as an aortic valve area of less than 0.8 cm2 and mean aortic valve gradient of more than 40 mm Hg or a peak aortic jet velocity of more than 4.0 m/second.
The patients in the trial had a New York Heart Association heart failure class II or greater, and they also had a predicted risk of operative mortality of at least 15% as determined by the site surgeon and cardiologist, coupled with a Society of Thoracic Surgery (STS) score of at least 10.
The mean age of the patients in the study was 83 years, and 94% of them were NYHA class III or IV. In all, 42 patients were not treated as assigned, Dr. Smith said.
All-cause mortality at 1 year was simÂilar for the transfemoral TAVR subgroup, at 22.2% and 26.4% for surgery, as well as for the transapical TAVR subgroup (29% vs. 28%, respectively).
A preliminary subgroup analysis sugÂgested that there may be a possible benefit found with TAVR in women and in patients without prior coronary artery bypass surgery, but Dr. Smith said that those results should be interÂpreted cautiously.
Overall, the TAVR mortality of 3.4% at 30 days was the lowest reported in any series, despite the use of an early-generation device and limited previous operator experience, he said, noting that nine of the participating centers had never performed TAVR before the trial.
Symptom improvement, including NYHA class and 6-minute walk distance, favored TAVR at 30 days and was similar to conventional surgery at 1 year, Dr. Moliterno said.
Coinvestigator Dr. Murat Tuzcu, vice chair of the department of cardiovascu?lar medicine at the Cleveland Clinic, stressed that the results were accomplished only through unprecedented teamwork among cardiologists, surgeons, and imaging experts, who conÂsulted on each case in the trial.
"I want to emphasize that if we fail to pay equal attention to what we have done in this trial after the device is approved, I don't think we will be able to replicate the same results," he said.
Several speakers at the press conference noted the balancing act that all clinicians will face regarding public demand for the new procedure.
Dr. Michael Crawford, chief of clinical cardiology at the University of California, San Francisco, observed that centers will face substantial up-front costs in the adoption of TAVR, including hybrid surgical/interventional suites, and that this will restrict initial uptake to high-volume centers of excellence.
The 3.4% mortality rate represents an “amazing effort, particularly in the use of the larger, first-generation device, said invited discussant Dr. Martyn Thomas, clinical director of cardiothoracic services at St. Thomas Hospital, London.
The delivery catheters used in PARTNER are sized at 22 and 24 French, which corresponds to an outside diamÂeter of 7-8 mm.
The size of catheters has been decreased down to 16-Fr in Europe, where more than 5,000 TAVR procedures have been done and where both the Edwards Lifesciences Corp.'s Sapien valve that was used in PARTNER and the Medtronic Inc.'s CoreValve are already approved for commercial use.
Recruitment began in February for the PARTNER II trial that is evaluating the smaller Edwards Sapien XT device and the later generation NovaFlex delivery system among inoperable patients with symptomatic severe aortic stenosis. The trial is expected to enroll 600 patients, and is targeted for primary completion in December 2011.
When asked whether the advent of newer, thinner devices will mean adoption in patients with lower risk, Dr. Smith replied that the adoption of TAVR will "march steadily down the risk categories."
Edwards Lifesciences sponsored the trial. Dr. Smith and his coauthors reported no conflicts of interest.
NEW ORLEANS Transcatheter aortic valve replacement was associated with the same survival rates at 1 year as was found in conventional surgery for older, high-risk patients who suffered from severe, symptomatic aortic stenosis, according to the results of the PARTNER trial.
All-cause mortality at 30 days favored the use of transcatheter aortic valve replacement (TAVR) at 3.4%, compared with 6.5% for open surgery, and was similar at 1 year (24% vs, 27%, respectively).
The difference at 1 year reached the trial's prespecified noninferiority margin with a P value of .001.
"We already learned from the previous cohort that TAVR is the standard of care for patients who can't tolerate surgery. So this [finding] opens up a new set of patients who may very well benefit as much with TAVR as with the gold standard surgery," coprincipal investigator Dr. Craig B. Smith said at the annual meeting of the American College of Cardiology.
However, The stroke rate with TAVR as seen in the industry-sponsored PARTNER (Placement of Aortic Transcatheter Valve) trial, turned out to be twice the rate for these patients as that observed with conventional surgical repair.
Stroke plus transient ischemic attack (TIA) rates were significantly more frequent with TAVR than surgery at both 30 days (5.5% vs. 2.4%) and 1 year (8.3% vs. 4.3%).
When only major strokes were compared, the difference was not significant between TAVR and convential surgery at 30 days (3.8% vs. 2.1%) or 1 year (5.1% vs. 2.4%), said Dr. Smith, chief of cardiothoracic surgery at New York Presbyterian Hospital/Columbia University Medical Center in New York City.
Notably, patients who un?derwent conventional surgery were significantly more likely than TAVR patients to experience major bleeding at 30 days (19.5% vs. 9.3%) and 1 year (26% vs. 15%), as well as new atrial fibrillation at both 30 days (16% vs. 8.6%) and 1 year (17% vs. 12%).
An examination of echocardiographic findings indicated a small hemodynamic benefit ocurred with TAVR vs. surgery at 1 year, but there was significantly increased paravalvular aortic reÂgurgitation at 30 days, 6 months, and 1 year.
"TAVR and AVR [aortic valve replacement] are both acceptable therapies in these high-risk patients; differing periprocedural hazards should influence case-based decision making," Dr. Smith said.
Dr. David Moliterno, chair of the intervention program (i2 Summit) at the meeting, told reporters that they were witnessing histoÂry in the making.
"This will probably be seen as one of the biggest steps in cardiovascular medicine, as far as intervention is concerned, potentially in our lifetime," said Dr. Moliterno, chief of cardiovascular medicine at the University of Kentucky, Lexington.
"If we look back to balloon angioÂplasty, the advent of stents and drug-eluting stents, this will be seen as the next major turning point."
Last year, the PARTNER trial investigators reported a 20% survival benefit at 1 year in a separate cohort of 358 inoperable patients with severe aortic stenosis who underwent transfemoral TAVR vs. standard therapy, including balloon aortic valvuloplasty (N. Engl. J. Med. 2010;363:1597-607). TAVR was associated with significantly more major vascular complications (16% vs. 1%) and a higher incidence of major strokes (5% vs. 1%).
The current portion of PARTNER randomly assigned 699 patients at 26 centers to surgery or TAVR. These patients were treated using the transfemoral approach in 492 cases and the transapical approach in 207.
Severe stenosis was defined as an aortic valve area of less than 0.8 cm2 and mean aortic valve gradient of more than 40 mm Hg or a peak aortic jet velocity of more than 4.0 m/second.
The patients in the trial had a New York Heart Association heart failure class II or greater, and they also had a predicted risk of operative mortality of at least 15% as determined by the site surgeon and cardiologist, coupled with a Society of Thoracic Surgery (STS) score of at least 10.
The mean age of the patients in the study was 83 years, and 94% of them were NYHA class III or IV. In all, 42 patients were not treated as assigned, Dr. Smith said.
All-cause mortality at 1 year was simÂilar for the transfemoral TAVR subgroup, at 22.2% and 26.4% for surgery, as well as for the transapical TAVR subgroup (29% vs. 28%, respectively).
A preliminary subgroup analysis sugÂgested that there may be a possible benefit found with TAVR in women and in patients without prior coronary artery bypass surgery, but Dr. Smith said that those results should be interÂpreted cautiously.
Overall, the TAVR mortality of 3.4% at 30 days was the lowest reported in any series, despite the use of an early-generation device and limited previous operator experience, he said, noting that nine of the participating centers had never performed TAVR before the trial.
Symptom improvement, including NYHA class and 6-minute walk distance, favored TAVR at 30 days and was similar to conventional surgery at 1 year, Dr. Moliterno said.
Coinvestigator Dr. Murat Tuzcu, vice chair of the department of cardiovascu?lar medicine at the Cleveland Clinic, stressed that the results were accomplished only through unprecedented teamwork among cardiologists, surgeons, and imaging experts, who conÂsulted on each case in the trial.
"I want to emphasize that if we fail to pay equal attention to what we have done in this trial after the device is approved, I don't think we will be able to replicate the same results," he said.
Several speakers at the press conference noted the balancing act that all clinicians will face regarding public demand for the new procedure.
Dr. Michael Crawford, chief of clinical cardiology at the University of California, San Francisco, observed that centers will face substantial up-front costs in the adoption of TAVR, including hybrid surgical/interventional suites, and that this will restrict initial uptake to high-volume centers of excellence.
The 3.4% mortality rate represents an “amazing effort, particularly in the use of the larger, first-generation device, said invited discussant Dr. Martyn Thomas, clinical director of cardiothoracic services at St. Thomas Hospital, London.
The delivery catheters used in PARTNER are sized at 22 and 24 French, which corresponds to an outside diamÂeter of 7-8 mm.
The size of catheters has been decreased down to 16-Fr in Europe, where more than 5,000 TAVR procedures have been done and where both the Edwards Lifesciences Corp.'s Sapien valve that was used in PARTNER and the Medtronic Inc.'s CoreValve are already approved for commercial use.
Recruitment began in February for the PARTNER II trial that is evaluating the smaller Edwards Sapien XT device and the later generation NovaFlex delivery system among inoperable patients with symptomatic severe aortic stenosis. The trial is expected to enroll 600 patients, and is targeted for primary completion in December 2011.
When asked whether the advent of newer, thinner devices will mean adoption in patients with lower risk, Dr. Smith replied that the adoption of TAVR will "march steadily down the risk categories."
Edwards Lifesciences sponsored the trial. Dr. Smith and his coauthors reported no conflicts of interest.
Clinical Challenge
Images courtesy Dr. Michael Davidson
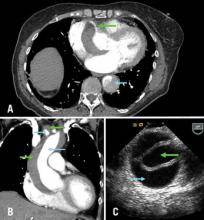
A) Axial CT chest at level of aortic root: green arrow = right coronary artery filling from true lumen, traversing false lumen; blue arrow = dissection in descending thoracic aorta; B) CT chest coronal reconstruction; blue arrows = true lumen in ascending aorta and innominate artery opacified with intravenous contrast; green arrows = false lumen; C) Transesophageal echocardiography of ascending aorta; green arrow= true lumen, blue arrow = false lumen.
BTX
A 71-year-old female with known ascending aortic aneurysm previously 4.5 cm presents to the emergency room with sharp chest pain of acute onset radiating to her back. She has diminished right radial and left femoral pulses. Chest x-ray shows a moderately widened mediastinum. Representative images are shown from the computed tomography of chest with contrast and a transesophageal echo with views of the ascending aorta.
Directed questions:
1. What is the diagnosis?
2. What are the two most common classification systems of this disease process?
3. What is the mortality associated with this diagnosis?
4. What is the best method of diagnosis in a stable patient? In an unstable patient?
5. What is the recommended treatment strategy?
6. What options are available for arterial cannulation?
Answers to the Clinical Challenge
1) Acute Aortic dissection of the ascending aorta, aortic arch and descending aorta
2) Stanford system (A=ascending arch, B=descending arch), Debakey system (I=ascending, arch and descending aorta, II - ascending aorta only, III - descending aorta only)
3) An acute type A dissection has been classically described as having a mortality of 1% per hour. Common causes of death are aortic free rupture, pericardial tamponade, acute aortic regurgitation, acute myocardial ischemia from coronary malperfusion.
4) In a stable patient, computed tomography with intravenous contrast is best due its ease and a sensitivity and a specificity nearing 90-100%. In unstable patients transesophageal echocardiography in the operating room is an excellent strategy for diagnosis to avoid delay in definitive surgical repair.
5) Initial medical management is aimed at control of blood pressure and pain. Esmolol and nitroprusside are good options. Unstable patients belong in the operating room and all delays should be avoided. Surgical repair is indicated in nearly all type A acute aortic dissections except maybe in the highest risk patients. At surgery, the ascending aorta, the aortic root, aortic valve, coronary arteries, aortic arch and arch vessels must all be assessed and repaired or replaced as necessary. As dissections frequently will extend into the arch, a short period of deep hypothermic circulatory arrest is common.
6) Most commonly, peripheral arterial cannulation is performed via the femoral or axillary artery. Care must be taken if dissection into the peripheral vessels is suspected. The ascending aorta may be cannulated but one must ensure perfusion through the true lumen. Transapical cannulation has been described as well. Transesophageal echocardiography can be used to guide cannulation placement into the ascending aorta.
Selected References and Additional Resources:
Bolman, R. Acute Type A Aortic Dissection. Operative Techniques in Thoracic and Cardiovascular Surgery. 14(2):124, 2009.
Coady MA, et al. Natural history, pathogenesis, and etiology of thoracic aortic aneurysms and dissections. Cardiology Clinics. 17:615, 1999.
Sabik JF, et al: Long-term effectiveness of operations for ascending aortic dissections. J Thorac Cardiovasc Surg. 119:946, 2000.
Safi HJ, Miller CC 3rd, Reardon MJ, et al: Operation for acute and chronic aortic dissection: Recent outcome with regard to neurologic deficit and early death. Ann Thorac Surg 1998; 66:402
TSDAWeekly Curriculum - "Acute Aortic Dissection" http://tsda.org/documents/PDF/Weekly%20Curricula/E-mail%20Archive/03.11.10.pdf
Images courtesy Dr. Michael Davidson
A) Axial CT chest at level of aortic root: green arrow = right coronary artery filling from true lumen, traversing false lumen; blue arrow = dissection in descending thoracic aorta; B) CT chest coronal reconstruction; blue arrows = true lumen in ascending aorta and innominate artery opacified with intravenous contrast; green arrows = false lumen; C) Transesophageal echocardiography of ascending aorta; green arrow= true lumen, blue arrow = false lumen.
BTX
A 71-year-old female with known ascending aortic aneurysm previously 4.5 cm presents to the emergency room with sharp chest pain of acute onset radiating to her back. She has diminished right radial and left femoral pulses. Chest x-ray shows a moderately widened mediastinum. Representative images are shown from the computed tomography of chest with contrast and a transesophageal echo with views of the ascending aorta.
Directed questions:
1. What is the diagnosis?
2. What are the two most common classification systems of this disease process?
3. What is the mortality associated with this diagnosis?
4. What is the best method of diagnosis in a stable patient? In an unstable patient?
5. What is the recommended treatment strategy?
6. What options are available for arterial cannulation?
Answers to the Clinical Challenge
1) Acute Aortic dissection of the ascending aorta, aortic arch and descending aorta
2) Stanford system (A=ascending arch, B=descending arch), Debakey system (I=ascending, arch and descending aorta, II - ascending aorta only, III - descending aorta only)
3) An acute type A dissection has been classically described as having a mortality of 1% per hour. Common causes of death are aortic free rupture, pericardial tamponade, acute aortic regurgitation, acute myocardial ischemia from coronary malperfusion.
4) In a stable patient, computed tomography with intravenous contrast is best due its ease and a sensitivity and a specificity nearing 90-100%. In unstable patients transesophageal echocardiography in the operating room is an excellent strategy for diagnosis to avoid delay in definitive surgical repair.
5) Initial medical management is aimed at control of blood pressure and pain. Esmolol and nitroprusside are good options. Unstable patients belong in the operating room and all delays should be avoided. Surgical repair is indicated in nearly all type A acute aortic dissections except maybe in the highest risk patients. At surgery, the ascending aorta, the aortic root, aortic valve, coronary arteries, aortic arch and arch vessels must all be assessed and repaired or replaced as necessary. As dissections frequently will extend into the arch, a short period of deep hypothermic circulatory arrest is common.
6) Most commonly, peripheral arterial cannulation is performed via the femoral or axillary artery. Care must be taken if dissection into the peripheral vessels is suspected. The ascending aorta may be cannulated but one must ensure perfusion through the true lumen. Transapical cannulation has been described as well. Transesophageal echocardiography can be used to guide cannulation placement into the ascending aorta.
Selected References and Additional Resources:
Bolman, R. Acute Type A Aortic Dissection. Operative Techniques in Thoracic and Cardiovascular Surgery. 14(2):124, 2009.
Coady MA, et al. Natural history, pathogenesis, and etiology of thoracic aortic aneurysms and dissections. Cardiology Clinics. 17:615, 1999.
Sabik JF, et al: Long-term effectiveness of operations for ascending aortic dissections. J Thorac Cardiovasc Surg. 119:946, 2000.
Safi HJ, Miller CC 3rd, Reardon MJ, et al: Operation for acute and chronic aortic dissection: Recent outcome with regard to neurologic deficit and early death. Ann Thorac Surg 1998; 66:402
TSDAWeekly Curriculum - "Acute Aortic Dissection" http://tsda.org/documents/PDF/Weekly%20Curricula/E-mail%20Archive/03.11.10.pdf
Images courtesy Dr. Michael Davidson
A) Axial CT chest at level of aortic root: green arrow = right coronary artery filling from true lumen, traversing false lumen; blue arrow = dissection in descending thoracic aorta; B) CT chest coronal reconstruction; blue arrows = true lumen in ascending aorta and innominate artery opacified with intravenous contrast; green arrows = false lumen; C) Transesophageal echocardiography of ascending aorta; green arrow= true lumen, blue arrow = false lumen.
BTX
A 71-year-old female with known ascending aortic aneurysm previously 4.5 cm presents to the emergency room with sharp chest pain of acute onset radiating to her back. She has diminished right radial and left femoral pulses. Chest x-ray shows a moderately widened mediastinum. Representative images are shown from the computed tomography of chest with contrast and a transesophageal echo with views of the ascending aorta.
Directed questions:
1. What is the diagnosis?
2. What are the two most common classification systems of this disease process?
3. What is the mortality associated with this diagnosis?
4. What is the best method of diagnosis in a stable patient? In an unstable patient?
5. What is the recommended treatment strategy?
6. What options are available for arterial cannulation?
Answers to the Clinical Challenge
1) Acute Aortic dissection of the ascending aorta, aortic arch and descending aorta
2) Stanford system (A=ascending arch, B=descending arch), Debakey system (I=ascending, arch and descending aorta, II - ascending aorta only, III - descending aorta only)
3) An acute type A dissection has been classically described as having a mortality of 1% per hour. Common causes of death are aortic free rupture, pericardial tamponade, acute aortic regurgitation, acute myocardial ischemia from coronary malperfusion.
4) In a stable patient, computed tomography with intravenous contrast is best due its ease and a sensitivity and a specificity nearing 90-100%. In unstable patients transesophageal echocardiography in the operating room is an excellent strategy for diagnosis to avoid delay in definitive surgical repair.
5) Initial medical management is aimed at control of blood pressure and pain. Esmolol and nitroprusside are good options. Unstable patients belong in the operating room and all delays should be avoided. Surgical repair is indicated in nearly all type A acute aortic dissections except maybe in the highest risk patients. At surgery, the ascending aorta, the aortic root, aortic valve, coronary arteries, aortic arch and arch vessels must all be assessed and repaired or replaced as necessary. As dissections frequently will extend into the arch, a short period of deep hypothermic circulatory arrest is common.
6) Most commonly, peripheral arterial cannulation is performed via the femoral or axillary artery. Care must be taken if dissection into the peripheral vessels is suspected. The ascending aorta may be cannulated but one must ensure perfusion through the true lumen. Transapical cannulation has been described as well. Transesophageal echocardiography can be used to guide cannulation placement into the ascending aorta.
Selected References and Additional Resources:
Bolman, R. Acute Type A Aortic Dissection. Operative Techniques in Thoracic and Cardiovascular Surgery. 14(2):124, 2009.
Coady MA, et al. Natural history, pathogenesis, and etiology of thoracic aortic aneurysms and dissections. Cardiology Clinics. 17:615, 1999.
Sabik JF, et al: Long-term effectiveness of operations for ascending aortic dissections. J Thorac Cardiovasc Surg. 119:946, 2000.
Safi HJ, Miller CC 3rd, Reardon MJ, et al: Operation for acute and chronic aortic dissection: Recent outcome with regard to neurologic deficit and early death. Ann Thorac Surg 1998; 66:402
TSDAWeekly Curriculum - "Acute Aortic Dissection" http://tsda.org/documents/PDF/Weekly%20Curricula/E-mail%20Archive/03.11.10.pdf
New Cardiothoracic Safety Reporting for 'Near Misses'
Many residents are unaware that a near miss reporting site for cardiothoracic surgery exists, the Cardiothoracic Safety Reporting System. This system, available through the CTSNet website (http://ctsrs.ctsnet.org/) allows anyone with a CTSNet login to submit descriptions of near misses and review the near misses others have submitted, ranging from labeling errors to a case of an ascending aortic graft igniting during a Bentall procedure.
By way of background, it has been estimated that about 100,000 people die avoidable deaths annually in U.S. hospitals, and many more incur injuries to the tune of an annual cost of approximately $9 billion; this exceeds the combined number of deaths and injuries from automobile and airplane crashes, suicides, poisonings, drownings and falls annually (BMJ 2000; 320(7237): 759-63). To reduce this number of errors requires learning not only from the times harm resulted to patients from errors, but also the cases where an error occurred but negative consequences were averted, a “near miss.”
As surgeons, we are accustomed to the discussion of our errors in regular morbidity and mortality conferences. Conferences such as these are invaluable for learning and practice improvement, but are limited in that they focus on actual adverse events, with near-misses rarely discussed. And, since only the participants present at the conferences learn the lessons to be gleaned from these events, the impact of conferences such as this are narrow.
In other disciplines, industry-wide schemes for reporting “close calls,” near misses, or sentinel (“warning”) events have been created. The near-miss reporting system in aviation, the Aviation Reporting Safety System (http://asrs.arc.nasa.gov/), is well-known, but similar systems are in place in the nuclear power industry, the military, petrochemical processing and steel production, high-reliability organizations which function in “high consequence” environments (www.ctsnet.org/sections/newsandviews/specialreports/article-10.html).
With these examples in mind, in 2007 the STS's Workforce for Patient Safety, at the time under the leadership of Dr. Thor Sundt, created the Cardiothoracic Safety Reporting System. Dr. Sundt, Dr. Marshall Blair, the current chair of the Workforce, and Dr. Emile Bacha, who was involved in the early planning and discussion of this project, recently approached the Thoracic Surgery Residents' Association (TSRA) regarding increasing resident participation in the effort. In TSRA internal discussions about the site, the chief concern was the anonymity of those who submitted cases.
Although CTSNet login information is required to access the site, it is electronically scrambled so that the users identity is “unobtainable and untraceable through any means, even if CTSNet wanted to try to find that information, they could not,” said Dr. Marshall. “It is scrambled to the extent that even if the Supreme Court ordered CTSNet to divulge the data, CTSNet would be unable to do so because they simply do not have that data; it doesn't exist anywhere,” explained Dr. Sundt.
Furthermore, the cases submitted are modified to remove any data that is identifiable and only those with CTSNet logins can review the cases. It is also important to recognize that by definition, no legally actionable material can be submitted; for legal reasons; no actual harm to a patient can have occurred for a case to be considered a “near miss.”
What is to be gained by submitting cases to the site? “Everybody makes mistakes … there's an old saying that a mistake is only a mistake when you don't learn from it. The purpose of a site like this is to share your mistakes and have other people benefit from it - that is something good that can come out of an otherwise bad situation. In a near miss, there is no harm to the patient, but we still can benefit.
“The idea is to move outside the team-centric morbidity and mortality conference type of sharing to a sharing with the rest of the field things that would benefit everybody and by doing this to end up avoiding that problem in a different place - how great is that?” said Dr. Bacha. “And, the more robust the site becomes, the more the field would benefit,” he continued.
Besides these altruistic benefits, those who review the site can also learn from the cases others have submitted. Visitors will see that previously submitted cases have been analyzed by a human factors expert, who provided useful commentary on the situations described. Dr. Marshall has analyzed all of the cases submitted to date and has found that they are split about 50/50 between labeling and device/equipment errors.
For instance, Dr. Marshall described a case of a labeling error in the context of transplantation. In this case, both the heart and lungs were procured at one institution but when the heart and lung teams arrived at their home institutions, they found they had brought the wrong cooler back with them; the heart team had the lungs and the lung team had the heart.
As it turned out, the organs went to neighboring institutions so the error was easily rectified, but this was an obvious near miss with a practical, easy to implement solution: label your coolers.
There is a lot to be gained from this project with very little downside; “it's very easy to submit cases, it is very straightforward and totally anonymous” said Dr. Marshall “and the more cases we have, the more we can learn.”
Many residents are unaware that a near miss reporting site for cardiothoracic surgery exists, the Cardiothoracic Safety Reporting System. This system, available through the CTSNet website (http://ctsrs.ctsnet.org/) allows anyone with a CTSNet login to submit descriptions of near misses and review the near misses others have submitted, ranging from labeling errors to a case of an ascending aortic graft igniting during a Bentall procedure.
By way of background, it has been estimated that about 100,000 people die avoidable deaths annually in U.S. hospitals, and many more incur injuries to the tune of an annual cost of approximately $9 billion; this exceeds the combined number of deaths and injuries from automobile and airplane crashes, suicides, poisonings, drownings and falls annually (BMJ 2000; 320(7237): 759-63). To reduce this number of errors requires learning not only from the times harm resulted to patients from errors, but also the cases where an error occurred but negative consequences were averted, a “near miss.”
As surgeons, we are accustomed to the discussion of our errors in regular morbidity and mortality conferences. Conferences such as these are invaluable for learning and practice improvement, but are limited in that they focus on actual adverse events, with near-misses rarely discussed. And, since only the participants present at the conferences learn the lessons to be gleaned from these events, the impact of conferences such as this are narrow.
In other disciplines, industry-wide schemes for reporting “close calls,” near misses, or sentinel (“warning”) events have been created. The near-miss reporting system in aviation, the Aviation Reporting Safety System (http://asrs.arc.nasa.gov/), is well-known, but similar systems are in place in the nuclear power industry, the military, petrochemical processing and steel production, high-reliability organizations which function in “high consequence” environments (www.ctsnet.org/sections/newsandviews/specialreports/article-10.html).
With these examples in mind, in 2007 the STS's Workforce for Patient Safety, at the time under the leadership of Dr. Thor Sundt, created the Cardiothoracic Safety Reporting System. Dr. Sundt, Dr. Marshall Blair, the current chair of the Workforce, and Dr. Emile Bacha, who was involved in the early planning and discussion of this project, recently approached the Thoracic Surgery Residents' Association (TSRA) regarding increasing resident participation in the effort. In TSRA internal discussions about the site, the chief concern was the anonymity of those who submitted cases.
Although CTSNet login information is required to access the site, it is electronically scrambled so that the users identity is “unobtainable and untraceable through any means, even if CTSNet wanted to try to find that information, they could not,” said Dr. Marshall. “It is scrambled to the extent that even if the Supreme Court ordered CTSNet to divulge the data, CTSNet would be unable to do so because they simply do not have that data; it doesn't exist anywhere,” explained Dr. Sundt.
Furthermore, the cases submitted are modified to remove any data that is identifiable and only those with CTSNet logins can review the cases. It is also important to recognize that by definition, no legally actionable material can be submitted; for legal reasons; no actual harm to a patient can have occurred for a case to be considered a “near miss.”
What is to be gained by submitting cases to the site? “Everybody makes mistakes … there's an old saying that a mistake is only a mistake when you don't learn from it. The purpose of a site like this is to share your mistakes and have other people benefit from it - that is something good that can come out of an otherwise bad situation. In a near miss, there is no harm to the patient, but we still can benefit.
“The idea is to move outside the team-centric morbidity and mortality conference type of sharing to a sharing with the rest of the field things that would benefit everybody and by doing this to end up avoiding that problem in a different place - how great is that?” said Dr. Bacha. “And, the more robust the site becomes, the more the field would benefit,” he continued.
Besides these altruistic benefits, those who review the site can also learn from the cases others have submitted. Visitors will see that previously submitted cases have been analyzed by a human factors expert, who provided useful commentary on the situations described. Dr. Marshall has analyzed all of the cases submitted to date and has found that they are split about 50/50 between labeling and device/equipment errors.
For instance, Dr. Marshall described a case of a labeling error in the context of transplantation. In this case, both the heart and lungs were procured at one institution but when the heart and lung teams arrived at their home institutions, they found they had brought the wrong cooler back with them; the heart team had the lungs and the lung team had the heart.
As it turned out, the organs went to neighboring institutions so the error was easily rectified, but this was an obvious near miss with a practical, easy to implement solution: label your coolers.
There is a lot to be gained from this project with very little downside; “it's very easy to submit cases, it is very straightforward and totally anonymous” said Dr. Marshall “and the more cases we have, the more we can learn.”
Many residents are unaware that a near miss reporting site for cardiothoracic surgery exists, the Cardiothoracic Safety Reporting System. This system, available through the CTSNet website (http://ctsrs.ctsnet.org/) allows anyone with a CTSNet login to submit descriptions of near misses and review the near misses others have submitted, ranging from labeling errors to a case of an ascending aortic graft igniting during a Bentall procedure.
By way of background, it has been estimated that about 100,000 people die avoidable deaths annually in U.S. hospitals, and many more incur injuries to the tune of an annual cost of approximately $9 billion; this exceeds the combined number of deaths and injuries from automobile and airplane crashes, suicides, poisonings, drownings and falls annually (BMJ 2000; 320(7237): 759-63). To reduce this number of errors requires learning not only from the times harm resulted to patients from errors, but also the cases where an error occurred but negative consequences were averted, a “near miss.”
As surgeons, we are accustomed to the discussion of our errors in regular morbidity and mortality conferences. Conferences such as these are invaluable for learning and practice improvement, but are limited in that they focus on actual adverse events, with near-misses rarely discussed. And, since only the participants present at the conferences learn the lessons to be gleaned from these events, the impact of conferences such as this are narrow.
In other disciplines, industry-wide schemes for reporting “close calls,” near misses, or sentinel (“warning”) events have been created. The near-miss reporting system in aviation, the Aviation Reporting Safety System (http://asrs.arc.nasa.gov/), is well-known, but similar systems are in place in the nuclear power industry, the military, petrochemical processing and steel production, high-reliability organizations which function in “high consequence” environments (www.ctsnet.org/sections/newsandviews/specialreports/article-10.html).
With these examples in mind, in 2007 the STS's Workforce for Patient Safety, at the time under the leadership of Dr. Thor Sundt, created the Cardiothoracic Safety Reporting System. Dr. Sundt, Dr. Marshall Blair, the current chair of the Workforce, and Dr. Emile Bacha, who was involved in the early planning and discussion of this project, recently approached the Thoracic Surgery Residents' Association (TSRA) regarding increasing resident participation in the effort. In TSRA internal discussions about the site, the chief concern was the anonymity of those who submitted cases.
Although CTSNet login information is required to access the site, it is electronically scrambled so that the users identity is “unobtainable and untraceable through any means, even if CTSNet wanted to try to find that information, they could not,” said Dr. Marshall. “It is scrambled to the extent that even if the Supreme Court ordered CTSNet to divulge the data, CTSNet would be unable to do so because they simply do not have that data; it doesn't exist anywhere,” explained Dr. Sundt.
Furthermore, the cases submitted are modified to remove any data that is identifiable and only those with CTSNet logins can review the cases. It is also important to recognize that by definition, no legally actionable material can be submitted; for legal reasons; no actual harm to a patient can have occurred for a case to be considered a “near miss.”
What is to be gained by submitting cases to the site? “Everybody makes mistakes … there's an old saying that a mistake is only a mistake when you don't learn from it. The purpose of a site like this is to share your mistakes and have other people benefit from it - that is something good that can come out of an otherwise bad situation. In a near miss, there is no harm to the patient, but we still can benefit.
“The idea is to move outside the team-centric morbidity and mortality conference type of sharing to a sharing with the rest of the field things that would benefit everybody and by doing this to end up avoiding that problem in a different place - how great is that?” said Dr. Bacha. “And, the more robust the site becomes, the more the field would benefit,” he continued.
Besides these altruistic benefits, those who review the site can also learn from the cases others have submitted. Visitors will see that previously submitted cases have been analyzed by a human factors expert, who provided useful commentary on the situations described. Dr. Marshall has analyzed all of the cases submitted to date and has found that they are split about 50/50 between labeling and device/equipment errors.
For instance, Dr. Marshall described a case of a labeling error in the context of transplantation. In this case, both the heart and lungs were procured at one institution but when the heart and lung teams arrived at their home institutions, they found they had brought the wrong cooler back with them; the heart team had the lungs and the lung team had the heart.
As it turned out, the organs went to neighboring institutions so the error was easily rectified, but this was an obvious near miss with a practical, easy to implement solution: label your coolers.
There is a lot to be gained from this project with very little downside; “it's very easy to submit cases, it is very straightforward and totally anonymous” said Dr. Marshall “and the more cases we have, the more we can learn.”
MedPac Seeks SGR Alternatives
WASHINGTON - Multiple options exist to permanently fix the Medicare Sustainable Growth Rate formula, but each has its cost to physicians, patients, and the program, according to staff analysts for the Medicare Payment Advisory Commission.
Among the options MedPAC staff presented to commissioners at a recent MedPAC meeting were adjusting the SGR's spending targets so that they are no longer cumulative, but are calculated on an annual basis and allowing some flexibility in the target. Both of those options would forgive any excess over the target, removing the annual pay cut threat doctors have endured since 2002 under the SGR, according to Cristina Boccuti, a principal policy analyst for MedPAC. However, forgiving any overage will lead to higher costs for the Medicare program. Neither option would leave any room to offer incentives for improved quality and efficiency, she added.
In the past, MedPAC has recommended setting target growth rates - and payment rates - according to particular service categories; the commission is looking in this direction again. For example, separate categories could be established for primary care, imaging, minor procedures, and anesthesia, allowing rates to more closely track volume of services. But the system might also provide incentives for physicians to order higher-volume (and more highly paid) services, according to Kevin Hayes, another MedPAC principal policy analyst.
Two options that seemed to pique commissioners' interest: exempting certain providers (such as accountable care organizations) from the current SGR target but holding them accountable for other targets, and penalizing physicians who are outliers in resource use.
MedPAC has two contractors working on projects to better determine the valuation of providers' time and resource use; more information will be available at the next commission meeting, Ms. Boccuti said.
Every year since 2002, Medicare has failed to meet the SGR targets, causing physician pay, by law, to be reduced. However, every year, and more recently, two or three times a year, Congress has stepped in to legislate a way to avoid those cuts. Cumulatively, the avoided cuts are becoming an ever-growing debt on the federal ledger.
The White House, in its fiscal 2012 budget proposal, is proposing to reduce that debt over the next 10 years, at a cost of $370 billion.
But the administration has figured out only how to pay for that fix for the first 2 years. The reality is that there's a declining pool of Medicare-specific offsets - required by law - to pay for fixing the SGR, Glenn Hackbarth, MedPAC chairman, said at the meeting. "We're in a deteriorating situation here; we're spiraling down," said Mr. Hackbarth. "This isn't going to get better; it's going to get worse."
He envisions a future where lawmakers will have to take money from education or roads or some other non-health-related area of the budget to pay for the Medicare fix. "And that bothers me," he said.
MedPAC in 2001 saw trouble with the SGR and recommended that it be scrapped. Now, the commission is looking again at ways to overhaul the formula so that physicians do not have to face a constantly shifting landscape. The uncertainty is undermining physician confidence in Medicare and leading some to stop seeing beneficiaries, Mr. Hackbarth noted.
He said the time might be right to work out a "quid pro quo" with physicians: an end to the yearly exercise to avert the SGR cuts in exchange for a payment system that has volume constraints and rewards efficiency and improved quality, or, alternatively penalizes those who fail to meet such targets.
WASHINGTON - Multiple options exist to permanently fix the Medicare Sustainable Growth Rate formula, but each has its cost to physicians, patients, and the program, according to staff analysts for the Medicare Payment Advisory Commission.
Among the options MedPAC staff presented to commissioners at a recent MedPAC meeting were adjusting the SGR's spending targets so that they are no longer cumulative, but are calculated on an annual basis and allowing some flexibility in the target. Both of those options would forgive any excess over the target, removing the annual pay cut threat doctors have endured since 2002 under the SGR, according to Cristina Boccuti, a principal policy analyst for MedPAC. However, forgiving any overage will lead to higher costs for the Medicare program. Neither option would leave any room to offer incentives for improved quality and efficiency, she added.
In the past, MedPAC has recommended setting target growth rates - and payment rates - according to particular service categories; the commission is looking in this direction again. For example, separate categories could be established for primary care, imaging, minor procedures, and anesthesia, allowing rates to more closely track volume of services. But the system might also provide incentives for physicians to order higher-volume (and more highly paid) services, according to Kevin Hayes, another MedPAC principal policy analyst.
Two options that seemed to pique commissioners' interest: exempting certain providers (such as accountable care organizations) from the current SGR target but holding them accountable for other targets, and penalizing physicians who are outliers in resource use.
MedPAC has two contractors working on projects to better determine the valuation of providers' time and resource use; more information will be available at the next commission meeting, Ms. Boccuti said.
Every year since 2002, Medicare has failed to meet the SGR targets, causing physician pay, by law, to be reduced. However, every year, and more recently, two or three times a year, Congress has stepped in to legislate a way to avoid those cuts. Cumulatively, the avoided cuts are becoming an ever-growing debt on the federal ledger.
The White House, in its fiscal 2012 budget proposal, is proposing to reduce that debt over the next 10 years, at a cost of $370 billion.
But the administration has figured out only how to pay for that fix for the first 2 years. The reality is that there's a declining pool of Medicare-specific offsets - required by law - to pay for fixing the SGR, Glenn Hackbarth, MedPAC chairman, said at the meeting. "We're in a deteriorating situation here; we're spiraling down," said Mr. Hackbarth. "This isn't going to get better; it's going to get worse."
He envisions a future where lawmakers will have to take money from education or roads or some other non-health-related area of the budget to pay for the Medicare fix. "And that bothers me," he said.
MedPAC in 2001 saw trouble with the SGR and recommended that it be scrapped. Now, the commission is looking again at ways to overhaul the formula so that physicians do not have to face a constantly shifting landscape. The uncertainty is undermining physician confidence in Medicare and leading some to stop seeing beneficiaries, Mr. Hackbarth noted.
He said the time might be right to work out a "quid pro quo" with physicians: an end to the yearly exercise to avert the SGR cuts in exchange for a payment system that has volume constraints and rewards efficiency and improved quality, or, alternatively penalizes those who fail to meet such targets.
WASHINGTON - Multiple options exist to permanently fix the Medicare Sustainable Growth Rate formula, but each has its cost to physicians, patients, and the program, according to staff analysts for the Medicare Payment Advisory Commission.
Among the options MedPAC staff presented to commissioners at a recent MedPAC meeting were adjusting the SGR's spending targets so that they are no longer cumulative, but are calculated on an annual basis and allowing some flexibility in the target. Both of those options would forgive any excess over the target, removing the annual pay cut threat doctors have endured since 2002 under the SGR, according to Cristina Boccuti, a principal policy analyst for MedPAC. However, forgiving any overage will lead to higher costs for the Medicare program. Neither option would leave any room to offer incentives for improved quality and efficiency, she added.
In the past, MedPAC has recommended setting target growth rates - and payment rates - according to particular service categories; the commission is looking in this direction again. For example, separate categories could be established for primary care, imaging, minor procedures, and anesthesia, allowing rates to more closely track volume of services. But the system might also provide incentives for physicians to order higher-volume (and more highly paid) services, according to Kevin Hayes, another MedPAC principal policy analyst.
Two options that seemed to pique commissioners' interest: exempting certain providers (such as accountable care organizations) from the current SGR target but holding them accountable for other targets, and penalizing physicians who are outliers in resource use.
MedPAC has two contractors working on projects to better determine the valuation of providers' time and resource use; more information will be available at the next commission meeting, Ms. Boccuti said.
Every year since 2002, Medicare has failed to meet the SGR targets, causing physician pay, by law, to be reduced. However, every year, and more recently, two or three times a year, Congress has stepped in to legislate a way to avoid those cuts. Cumulatively, the avoided cuts are becoming an ever-growing debt on the federal ledger.
The White House, in its fiscal 2012 budget proposal, is proposing to reduce that debt over the next 10 years, at a cost of $370 billion.
But the administration has figured out only how to pay for that fix for the first 2 years. The reality is that there's a declining pool of Medicare-specific offsets - required by law - to pay for fixing the SGR, Glenn Hackbarth, MedPAC chairman, said at the meeting. "We're in a deteriorating situation here; we're spiraling down," said Mr. Hackbarth. "This isn't going to get better; it's going to get worse."
He envisions a future where lawmakers will have to take money from education or roads or some other non-health-related area of the budget to pay for the Medicare fix. "And that bothers me," he said.
MedPAC in 2001 saw trouble with the SGR and recommended that it be scrapped. Now, the commission is looking again at ways to overhaul the formula so that physicians do not have to face a constantly shifting landscape. The uncertainty is undermining physician confidence in Medicare and leading some to stop seeing beneficiaries, Mr. Hackbarth noted.
He said the time might be right to work out a "quid pro quo" with physicians: an end to the yearly exercise to avert the SGR cuts in exchange for a payment system that has volume constraints and rewards efficiency and improved quality, or, alternatively penalizes those who fail to meet such targets.
Delaying Esophagectomy After CXRT May Be Okay, Study Finds
FROM THE ANNUAL MEETING OF THE SOCIETY OF THORACIC SURGEONS
SAN DIEGO - An interval between neoadjuvant chemoradiation and esophagectomy that extends beyond 8 weeks is not associated with increased perioperative complications, increased pathological complete response, or change in overall survival, results from a long-term single-center study showed.
"For patients who have not yet recovered from neoadjuvant chemoradiation, it is safe to delay surgery to allow them to improve their performance status," Dr. Jae Y. Kim said at the meeting.
Traditionally, he said, surgery has been recommended within 8 weeks after completing neoadjuvant chemoradiation for esophageal cancer, yet many patients choose to delay their surgery.
Some patients have not yet recovered from chemoradiation, while others are delayed for personal or logistical reasons, explained Dr. Kim, a thoracic surgery fellow at the University of Texas M.D. Anderson Cancer Center, Houston. "The impact of this delay on outcomes is unknown." Radiation-induced tumor necrosis increases over time, he said, and there is evidence from rectal cancer that a longer interval may increase the rate of pathological complete response. "On the other hand, there are theoretical concerns that this delay may lead to increased radiation fibrosis and cause a more difficult operation. It is also possible that a delay would allow for tumor regrowth."
In an effort to determine whether an increased interval between chemoradiation and surgery is associated with risk of major perioperative complications or overall survival, Dr. Kim and his associates conducted a retrospective study of 266 patients with esophageal cancer followed by neoadjuvant chemoradiation who were treated at M.D. Anderson in 2002-2008. They divided the patients into two groups: a "short-interval" group of 150 who underwent esophagectomy within 8 weeks of chemoradiation, and a "delayed" group of 116 who underwent esophagectomy more than 8 weeks following chemoradiation.
"Most patients were clustered around 4-11 weeks," Dr. Kim said. "No patient had surgery more than 46 weeks after completing neoadjuvant chemoradiation."
The median interval from completion of neoadjuvant therapy to surgery was 46 days in the short-interval group and 76 days in the delayed group. In both groups, more than 95% of patients were staged with PET-CT and endoscopic ultrasound.
The researchers compared the two groups in terms of perioperative complications, rate of pathological complete response, and overall survival.
The two groups were similar in 18 of 22 baseline characteristics examined, but they were different in four areas. Compared with their counterparts in the short-interval group, the patients in the delayed group were slightly older (mean age, 60 years vs. 57 years, respectively), had more coronary artery disease (17% vs. 7%), had less adenocarcinoma histology (87% vs. 97%), and weighed less (53% with a body mass index of 25 kg/m2 or greater vs. 75% of their counterparts in the short-interval group).
By any objective measure used to gauge the difficulty of the operation, the two groups were similar, including mean OR time (390 minutes in the short-interval group vs. 398 minutes in the delayed group), mean number of lymph nodes removed (21% vs. 20%), and mean estimated blood loss (505 mL vs. 478 mL).
The rates of major complications also were similar between the two groups, including perioperative mortality (2% in the short-interval group vs. 3% in the delayed group), median length of stay (11 days in each group), and rate of anastomotic leak (11% vs. 16%).
The rate of pathological complete response was similar between the two groups (21% vs. 23%).
Overall 5-year survival in the short-interval group was 46%, compared with 36% in the delayed-surgery group, a nonsignificant difference. Disease-free 5-year survival in the short-interval group was 44%, compared with 36% in the delayed group.
"The timing of surgery, both as a continuous and a dichotomous variable, was not associated" with perioperative complication or death, pathological complete response, or overall survival, Dr. Kim added.
On multivariable analysis, older age, more involved lymph nodes, and advanced pathological stage were independently associated with decreased survival.
The researchers performed a subgroup analysis of patients with adenocarcinoma histology and found that the results were similar.
Dr. Kim said that he had no relevant financial disclosures.
The authors point out that it is often difficult to standardize timing of esophagectomy in this setting because of a variety of difficult-to-anticipate events, particularly induction therapy toxicities. That most patients still had their esophagectomies performed by 3 months following the end of therapy suggests that the two groups might not be expected to be vastly different, although it is comforting to now have data to support this contention. Perhaps a future study might be to compare morbidity/mortality of esophagectomy following induction therapy versus that performed as a salvage strategy for patients who have failed chemoradiotherapy as definitive treatment. Those two groups are far more distinct. Late local recurrence following definitive chemoradiation therapy is, unfortunately, not that uncommon and has few other treatment options.
Dr. Sudish Murthy is an ACS Fellow and surgical director of the Center for Major Airway Disease, Cleveland Clinic.
The authors point out that it is often difficult to standardize timing of esophagectomy in this setting because of a variety of difficult-to-anticipate events, particularly induction therapy toxicities. That most patients still had their esophagectomies performed by 3 months following the end of therapy suggests that the two groups might not be expected to be vastly different, although it is comforting to now have data to support this contention. Perhaps a future study might be to compare morbidity/mortality of esophagectomy following induction therapy versus that performed as a salvage strategy for patients who have failed chemoradiotherapy as definitive treatment. Those two groups are far more distinct. Late local recurrence following definitive chemoradiation therapy is, unfortunately, not that uncommon and has few other treatment options.
Dr. Sudish Murthy is an ACS Fellow and surgical director of the Center for Major Airway Disease, Cleveland Clinic.
The authors point out that it is often difficult to standardize timing of esophagectomy in this setting because of a variety of difficult-to-anticipate events, particularly induction therapy toxicities. That most patients still had their esophagectomies performed by 3 months following the end of therapy suggests that the two groups might not be expected to be vastly different, although it is comforting to now have data to support this contention. Perhaps a future study might be to compare morbidity/mortality of esophagectomy following induction therapy versus that performed as a salvage strategy for patients who have failed chemoradiotherapy as definitive treatment. Those two groups are far more distinct. Late local recurrence following definitive chemoradiation therapy is, unfortunately, not that uncommon and has few other treatment options.
Dr. Sudish Murthy is an ACS Fellow and surgical director of the Center for Major Airway Disease, Cleveland Clinic.
FROM THE ANNUAL MEETING OF THE SOCIETY OF THORACIC SURGEONS
SAN DIEGO - An interval between neoadjuvant chemoradiation and esophagectomy that extends beyond 8 weeks is not associated with increased perioperative complications, increased pathological complete response, or change in overall survival, results from a long-term single-center study showed.
"For patients who have not yet recovered from neoadjuvant chemoradiation, it is safe to delay surgery to allow them to improve their performance status," Dr. Jae Y. Kim said at the meeting.
Traditionally, he said, surgery has been recommended within 8 weeks after completing neoadjuvant chemoradiation for esophageal cancer, yet many patients choose to delay their surgery.
Some patients have not yet recovered from chemoradiation, while others are delayed for personal or logistical reasons, explained Dr. Kim, a thoracic surgery fellow at the University of Texas M.D. Anderson Cancer Center, Houston. "The impact of this delay on outcomes is unknown." Radiation-induced tumor necrosis increases over time, he said, and there is evidence from rectal cancer that a longer interval may increase the rate of pathological complete response. "On the other hand, there are theoretical concerns that this delay may lead to increased radiation fibrosis and cause a more difficult operation. It is also possible that a delay would allow for tumor regrowth."
In an effort to determine whether an increased interval between chemoradiation and surgery is associated with risk of major perioperative complications or overall survival, Dr. Kim and his associates conducted a retrospective study of 266 patients with esophageal cancer followed by neoadjuvant chemoradiation who were treated at M.D. Anderson in 2002-2008. They divided the patients into two groups: a "short-interval" group of 150 who underwent esophagectomy within 8 weeks of chemoradiation, and a "delayed" group of 116 who underwent esophagectomy more than 8 weeks following chemoradiation.
"Most patients were clustered around 4-11 weeks," Dr. Kim said. "No patient had surgery more than 46 weeks after completing neoadjuvant chemoradiation."
The median interval from completion of neoadjuvant therapy to surgery was 46 days in the short-interval group and 76 days in the delayed group. In both groups, more than 95% of patients were staged with PET-CT and endoscopic ultrasound.
The researchers compared the two groups in terms of perioperative complications, rate of pathological complete response, and overall survival.
The two groups were similar in 18 of 22 baseline characteristics examined, but they were different in four areas. Compared with their counterparts in the short-interval group, the patients in the delayed group were slightly older (mean age, 60 years vs. 57 years, respectively), had more coronary artery disease (17% vs. 7%), had less adenocarcinoma histology (87% vs. 97%), and weighed less (53% with a body mass index of 25 kg/m2 or greater vs. 75% of their counterparts in the short-interval group).
By any objective measure used to gauge the difficulty of the operation, the two groups were similar, including mean OR time (390 minutes in the short-interval group vs. 398 minutes in the delayed group), mean number of lymph nodes removed (21% vs. 20%), and mean estimated blood loss (505 mL vs. 478 mL).
The rates of major complications also were similar between the two groups, including perioperative mortality (2% in the short-interval group vs. 3% in the delayed group), median length of stay (11 days in each group), and rate of anastomotic leak (11% vs. 16%).
The rate of pathological complete response was similar between the two groups (21% vs. 23%).
Overall 5-year survival in the short-interval group was 46%, compared with 36% in the delayed-surgery group, a nonsignificant difference. Disease-free 5-year survival in the short-interval group was 44%, compared with 36% in the delayed group.
"The timing of surgery, both as a continuous and a dichotomous variable, was not associated" with perioperative complication or death, pathological complete response, or overall survival, Dr. Kim added.
On multivariable analysis, older age, more involved lymph nodes, and advanced pathological stage were independently associated with decreased survival.
The researchers performed a subgroup analysis of patients with adenocarcinoma histology and found that the results were similar.
Dr. Kim said that he had no relevant financial disclosures.
FROM THE ANNUAL MEETING OF THE SOCIETY OF THORACIC SURGEONS
SAN DIEGO - An interval between neoadjuvant chemoradiation and esophagectomy that extends beyond 8 weeks is not associated with increased perioperative complications, increased pathological complete response, or change in overall survival, results from a long-term single-center study showed.
"For patients who have not yet recovered from neoadjuvant chemoradiation, it is safe to delay surgery to allow them to improve their performance status," Dr. Jae Y. Kim said at the meeting.
Traditionally, he said, surgery has been recommended within 8 weeks after completing neoadjuvant chemoradiation for esophageal cancer, yet many patients choose to delay their surgery.
Some patients have not yet recovered from chemoradiation, while others are delayed for personal or logistical reasons, explained Dr. Kim, a thoracic surgery fellow at the University of Texas M.D. Anderson Cancer Center, Houston. "The impact of this delay on outcomes is unknown." Radiation-induced tumor necrosis increases over time, he said, and there is evidence from rectal cancer that a longer interval may increase the rate of pathological complete response. "On the other hand, there are theoretical concerns that this delay may lead to increased radiation fibrosis and cause a more difficult operation. It is also possible that a delay would allow for tumor regrowth."
In an effort to determine whether an increased interval between chemoradiation and surgery is associated with risk of major perioperative complications or overall survival, Dr. Kim and his associates conducted a retrospective study of 266 patients with esophageal cancer followed by neoadjuvant chemoradiation who were treated at M.D. Anderson in 2002-2008. They divided the patients into two groups: a "short-interval" group of 150 who underwent esophagectomy within 8 weeks of chemoradiation, and a "delayed" group of 116 who underwent esophagectomy more than 8 weeks following chemoradiation.
"Most patients were clustered around 4-11 weeks," Dr. Kim said. "No patient had surgery more than 46 weeks after completing neoadjuvant chemoradiation."
The median interval from completion of neoadjuvant therapy to surgery was 46 days in the short-interval group and 76 days in the delayed group. In both groups, more than 95% of patients were staged with PET-CT and endoscopic ultrasound.
The researchers compared the two groups in terms of perioperative complications, rate of pathological complete response, and overall survival.
The two groups were similar in 18 of 22 baseline characteristics examined, but they were different in four areas. Compared with their counterparts in the short-interval group, the patients in the delayed group were slightly older (mean age, 60 years vs. 57 years, respectively), had more coronary artery disease (17% vs. 7%), had less adenocarcinoma histology (87% vs. 97%), and weighed less (53% with a body mass index of 25 kg/m2 or greater vs. 75% of their counterparts in the short-interval group).
By any objective measure used to gauge the difficulty of the operation, the two groups were similar, including mean OR time (390 minutes in the short-interval group vs. 398 minutes in the delayed group), mean number of lymph nodes removed (21% vs. 20%), and mean estimated blood loss (505 mL vs. 478 mL).
The rates of major complications also were similar between the two groups, including perioperative mortality (2% in the short-interval group vs. 3% in the delayed group), median length of stay (11 days in each group), and rate of anastomotic leak (11% vs. 16%).
The rate of pathological complete response was similar between the two groups (21% vs. 23%).
Overall 5-year survival in the short-interval group was 46%, compared with 36% in the delayed-surgery group, a nonsignificant difference. Disease-free 5-year survival in the short-interval group was 44%, compared with 36% in the delayed group.
"The timing of surgery, both as a continuous and a dichotomous variable, was not associated" with perioperative complication or death, pathological complete response, or overall survival, Dr. Kim added.
On multivariable analysis, older age, more involved lymph nodes, and advanced pathological stage were independently associated with decreased survival.
The researchers performed a subgroup analysis of patients with adenocarcinoma histology and found that the results were similar.
Dr. Kim said that he had no relevant financial disclosures.
Delaying Esophagectomy After CXRT May Be Okay, Study Finds
FROM THE ANNUAL MEETING OF THE SOCIETY OF THORACIC SURGEONS
SAN DIEGO - An interval between neoadjuvant chemoradiation and esophagectomy that extends beyond 8 weeks is not associated with increased perioperative complications, increased pathological complete response, or change in overall survival, results from a long-term single-center study showed.
"For patients who have not yet recovered from neoadjuvant chemoradiation, it is safe to delay surgery to allow them to improve their performance status," Dr. Jae Y. Kim said at the meeting.
Traditionally, he said, surgery has been recommended within 8 weeks after completing neoadjuvant chemoradiation for esophageal cancer, yet many patients choose to delay their surgery.
Some patients have not yet recovered from chemoradiation, while others are delayed for personal or logistical reasons, explained Dr. Kim, a thoracic surgery fellow at the University of Texas M.D. Anderson Cancer Center, Houston. "The impact of this delay on outcomes is unknown." Radiation-induced tumor necrosis increases over time, he said, and there is evidence from rectal cancer that a longer interval may increase the rate of pathological complete response. "On the other hand, there are theoretical concerns that this delay may lead to increased radiation fibrosis and cause a more difficult operation. It is also possible that a delay would allow for tumor regrowth."
In an effort to determine whether an increased interval between chemoradiation and surgery is associated with risk of major perioperative complications or overall survival, Dr. Kim and his associates conducted a retrospective study of 266 patients with esophageal cancer followed by neoadjuvant chemoradiation who were treated at M.D. Anderson in 2002-2008. They divided the patients into two groups: a "short-interval" group of 150 who underwent esophagectomy within 8 weeks of chemoradiation, and a "delayed" group of 116 who underwent esophagectomy more than 8 weeks following chemoradiation.
"Most patients were clustered around 4-11 weeks," Dr. Kim said. "No patient had surgery more than 46 weeks after completing neoadjuvant chemoradiation."
The median interval from completion of neoadjuvant therapy to surgery was 46 days in the short-interval group and 76 days in the delayed group. In both groups, more than 95% of patients were staged with PET-CT and endoscopic ultrasound.
The researchers compared the two groups in terms of perioperative complications, rate of pathological complete response, and overall survival.
The two groups were similar in 18 of 22 baseline characteristics examined, but they were different in four areas. Compared with their counterparts in the short-interval group, the patients in the delayed group were slightly older (mean age, 60 years vs. 57 years, respectively), had more coronary artery disease (17% vs. 7%), had less adenocarcinoma histology (87% vs. 97%), and weighed less (53% with a body mass index of 25 kg/m2 or greater vs. 75% of their counterparts in the short-interval group).
By any objective measure used to gauge the difficulty of the operation, the two groups were similar, including mean OR time (390 minutes in the short-interval group vs. 398 minutes in the delayed group), mean number of lymph nodes removed (21% vs. 20%), and mean estimated blood loss (505 mL vs. 478 mL).
The rates of major complications also were similar between the two groups, including perioperative mortality (2% in the short-interval group vs. 3% in the delayed group), median length of stay (11 days in each group), and rate of anastomotic leak (11% vs. 16%).
The rate of pathological complete response was similar between the two groups (21% vs. 23%).
Overall 5-year survival in the short-interval group was 46%, compared with 36% in the delayed-surgery group, a nonsignificant difference. Disease-free 5-year survival in the short-interval group was 44%, compared with 36% in the delayed group.
"The timing of surgery, both as a continuous and a dichotomous variable, was not associated" with perioperative complication or death, pathological complete response, or overall survival, Dr. Kim added.
On multivariable analysis, older age, more involved lymph nodes, and advanced pathological stage were independently associated with decreased survival.
The researchers performed a subgroup analysis of patients with adenocarcinoma histology and found that the results were similar.
Dr. Kim said that he had no relevant financial disclosures.
The authors point out that it is often difficult to standardize timing of esophagectomy in this setting because of a variety of difficult-to-anticipate events, particularly induction therapy toxicities. That most patients still had their esophagectomies performed by 3 months following the end of therapy suggests that the two groups might not be expected to be vastly different, although it is comforting to now have data to support this contention. Perhaps a future study might be to compare morbidity/mortality of esophagectomy following induction therapy versus that performed as a salvage strategy for patients who have failed chemoradiotherapy as definitive treatment. Those two groups are far more distinct. Late local recurrence following definitive chemoradiation therapy is, unfortunately, not that uncommon and has few other treatment options.
Dr. Sudish Murthy is an ACS Fellow and surgical director of the Center for Major Airway Disease, Cleveland Clinic.
The authors point out that it is often difficult to standardize timing of esophagectomy in this setting because of a variety of difficult-to-anticipate events, particularly induction therapy toxicities. That most patients still had their esophagectomies performed by 3 months following the end of therapy suggests that the two groups might not be expected to be vastly different, although it is comforting to now have data to support this contention. Perhaps a future study might be to compare morbidity/mortality of esophagectomy following induction therapy versus that performed as a salvage strategy for patients who have failed chemoradiotherapy as definitive treatment. Those two groups are far more distinct. Late local recurrence following definitive chemoradiation therapy is, unfortunately, not that uncommon and has few other treatment options.
Dr. Sudish Murthy is an ACS Fellow and surgical director of the Center for Major Airway Disease, Cleveland Clinic.
The authors point out that it is often difficult to standardize timing of esophagectomy in this setting because of a variety of difficult-to-anticipate events, particularly induction therapy toxicities. That most patients still had their esophagectomies performed by 3 months following the end of therapy suggests that the two groups might not be expected to be vastly different, although it is comforting to now have data to support this contention. Perhaps a future study might be to compare morbidity/mortality of esophagectomy following induction therapy versus that performed as a salvage strategy for patients who have failed chemoradiotherapy as definitive treatment. Those two groups are far more distinct. Late local recurrence following definitive chemoradiation therapy is, unfortunately, not that uncommon and has few other treatment options.
Dr. Sudish Murthy is an ACS Fellow and surgical director of the Center for Major Airway Disease, Cleveland Clinic.
FROM THE ANNUAL MEETING OF THE SOCIETY OF THORACIC SURGEONS
SAN DIEGO - An interval between neoadjuvant chemoradiation and esophagectomy that extends beyond 8 weeks is not associated with increased perioperative complications, increased pathological complete response, or change in overall survival, results from a long-term single-center study showed.
"For patients who have not yet recovered from neoadjuvant chemoradiation, it is safe to delay surgery to allow them to improve their performance status," Dr. Jae Y. Kim said at the meeting.
Traditionally, he said, surgery has been recommended within 8 weeks after completing neoadjuvant chemoradiation for esophageal cancer, yet many patients choose to delay their surgery.
Some patients have not yet recovered from chemoradiation, while others are delayed for personal or logistical reasons, explained Dr. Kim, a thoracic surgery fellow at the University of Texas M.D. Anderson Cancer Center, Houston. "The impact of this delay on outcomes is unknown." Radiation-induced tumor necrosis increases over time, he said, and there is evidence from rectal cancer that a longer interval may increase the rate of pathological complete response. "On the other hand, there are theoretical concerns that this delay may lead to increased radiation fibrosis and cause a more difficult operation. It is also possible that a delay would allow for tumor regrowth."
In an effort to determine whether an increased interval between chemoradiation and surgery is associated with risk of major perioperative complications or overall survival, Dr. Kim and his associates conducted a retrospective study of 266 patients with esophageal cancer followed by neoadjuvant chemoradiation who were treated at M.D. Anderson in 2002-2008. They divided the patients into two groups: a "short-interval" group of 150 who underwent esophagectomy within 8 weeks of chemoradiation, and a "delayed" group of 116 who underwent esophagectomy more than 8 weeks following chemoradiation.
"Most patients were clustered around 4-11 weeks," Dr. Kim said. "No patient had surgery more than 46 weeks after completing neoadjuvant chemoradiation."
The median interval from completion of neoadjuvant therapy to surgery was 46 days in the short-interval group and 76 days in the delayed group. In both groups, more than 95% of patients were staged with PET-CT and endoscopic ultrasound.
The researchers compared the two groups in terms of perioperative complications, rate of pathological complete response, and overall survival.
The two groups were similar in 18 of 22 baseline characteristics examined, but they were different in four areas. Compared with their counterparts in the short-interval group, the patients in the delayed group were slightly older (mean age, 60 years vs. 57 years, respectively), had more coronary artery disease (17% vs. 7%), had less adenocarcinoma histology (87% vs. 97%), and weighed less (53% with a body mass index of 25 kg/m2 or greater vs. 75% of their counterparts in the short-interval group).
By any objective measure used to gauge the difficulty of the operation, the two groups were similar, including mean OR time (390 minutes in the short-interval group vs. 398 minutes in the delayed group), mean number of lymph nodes removed (21% vs. 20%), and mean estimated blood loss (505 mL vs. 478 mL).
The rates of major complications also were similar between the two groups, including perioperative mortality (2% in the short-interval group vs. 3% in the delayed group), median length of stay (11 days in each group), and rate of anastomotic leak (11% vs. 16%).
The rate of pathological complete response was similar between the two groups (21% vs. 23%).
Overall 5-year survival in the short-interval group was 46%, compared with 36% in the delayed-surgery group, a nonsignificant difference. Disease-free 5-year survival in the short-interval group was 44%, compared with 36% in the delayed group.
"The timing of surgery, both as a continuous and a dichotomous variable, was not associated" with perioperative complication or death, pathological complete response, or overall survival, Dr. Kim added.
On multivariable analysis, older age, more involved lymph nodes, and advanced pathological stage were independently associated with decreased survival.
The researchers performed a subgroup analysis of patients with adenocarcinoma histology and found that the results were similar.
Dr. Kim said that he had no relevant financial disclosures.
FROM THE ANNUAL MEETING OF THE SOCIETY OF THORACIC SURGEONS
SAN DIEGO - An interval between neoadjuvant chemoradiation and esophagectomy that extends beyond 8 weeks is not associated with increased perioperative complications, increased pathological complete response, or change in overall survival, results from a long-term single-center study showed.
"For patients who have not yet recovered from neoadjuvant chemoradiation, it is safe to delay surgery to allow them to improve their performance status," Dr. Jae Y. Kim said at the meeting.
Traditionally, he said, surgery has been recommended within 8 weeks after completing neoadjuvant chemoradiation for esophageal cancer, yet many patients choose to delay their surgery.
Some patients have not yet recovered from chemoradiation, while others are delayed for personal or logistical reasons, explained Dr. Kim, a thoracic surgery fellow at the University of Texas M.D. Anderson Cancer Center, Houston. "The impact of this delay on outcomes is unknown." Radiation-induced tumor necrosis increases over time, he said, and there is evidence from rectal cancer that a longer interval may increase the rate of pathological complete response. "On the other hand, there are theoretical concerns that this delay may lead to increased radiation fibrosis and cause a more difficult operation. It is also possible that a delay would allow for tumor regrowth."
In an effort to determine whether an increased interval between chemoradiation and surgery is associated with risk of major perioperative complications or overall survival, Dr. Kim and his associates conducted a retrospective study of 266 patients with esophageal cancer followed by neoadjuvant chemoradiation who were treated at M.D. Anderson in 2002-2008. They divided the patients into two groups: a "short-interval" group of 150 who underwent esophagectomy within 8 weeks of chemoradiation, and a "delayed" group of 116 who underwent esophagectomy more than 8 weeks following chemoradiation.
"Most patients were clustered around 4-11 weeks," Dr. Kim said. "No patient had surgery more than 46 weeks after completing neoadjuvant chemoradiation."
The median interval from completion of neoadjuvant therapy to surgery was 46 days in the short-interval group and 76 days in the delayed group. In both groups, more than 95% of patients were staged with PET-CT and endoscopic ultrasound.
The researchers compared the two groups in terms of perioperative complications, rate of pathological complete response, and overall survival.
The two groups were similar in 18 of 22 baseline characteristics examined, but they were different in four areas. Compared with their counterparts in the short-interval group, the patients in the delayed group were slightly older (mean age, 60 years vs. 57 years, respectively), had more coronary artery disease (17% vs. 7%), had less adenocarcinoma histology (87% vs. 97%), and weighed less (53% with a body mass index of 25 kg/m2 or greater vs. 75% of their counterparts in the short-interval group).
By any objective measure used to gauge the difficulty of the operation, the two groups were similar, including mean OR time (390 minutes in the short-interval group vs. 398 minutes in the delayed group), mean number of lymph nodes removed (21% vs. 20%), and mean estimated blood loss (505 mL vs. 478 mL).
The rates of major complications also were similar between the two groups, including perioperative mortality (2% in the short-interval group vs. 3% in the delayed group), median length of stay (11 days in each group), and rate of anastomotic leak (11% vs. 16%).
The rate of pathological complete response was similar between the two groups (21% vs. 23%).
Overall 5-year survival in the short-interval group was 46%, compared with 36% in the delayed-surgery group, a nonsignificant difference. Disease-free 5-year survival in the short-interval group was 44%, compared with 36% in the delayed group.
"The timing of surgery, both as a continuous and a dichotomous variable, was not associated" with perioperative complication or death, pathological complete response, or overall survival, Dr. Kim added.
On multivariable analysis, older age, more involved lymph nodes, and advanced pathological stage were independently associated with decreased survival.
The researchers performed a subgroup analysis of patients with adenocarcinoma histology and found that the results were similar.
Dr. Kim said that he had no relevant financial disclosures.
Delaying Esophagectomy After CXRT May Be Okay, Study Finds
FROM THE ANNUAL MEETING OF THE SOCIETY OF THORACIC SURGEONS
SAN DIEGO - An interval between neoadjuvant chemoradiation and esophagectomy that extends beyond 8 weeks is not associated with increased perioperative complications, increased pathological complete response, or change in overall survival, results from a long-term single-center study showed.
"For patients who have not yet recovered from neoadjuvant chemoradiation, it is safe to delay surgery to allow them to improve their performance status," Dr. Jae Y. Kim said at the meeting.
Traditionally, he said, surgery has been recommended within 8 weeks after completing neoadjuvant chemoradiation for esophageal cancer, yet many patients choose to delay their surgery.
Some patients have not yet recovered from chemoradiation, while others are delayed for personal or logistical reasons, explained Dr. Kim, a thoracic surgery fellow at the University of Texas M.D. Anderson Cancer Center, Houston. "The impact of this delay on outcomes is unknown." Radiation-induced tumor necrosis increases over time, he said, and there is evidence from rectal cancer that a longer interval may increase the rate of pathological complete response. "On the other hand, there are theoretical concerns that this delay may lead to increased radiation fibrosis and cause a more difficult operation. It is also possible that a delay would allow for tumor regrowth."
In an effort to determine whether an increased interval between chemoradiation and surgery is associated with risk of major perioperative complications or overall survival, Dr. Kim and his associates conducted a retrospective study of 266 patients with esophageal cancer followed by neoadjuvant chemoradiation who were treated at M.D. Anderson in 2002-2008. They divided the patients into two groups: a "short-interval" group of 150 who underwent esophagectomy within 8 weeks of chemoradiation, and a "delayed" group of 116 who underwent esophagectomy more than 8 weeks following chemoradiation.
"Most patients were clustered around 4-11 weeks," Dr. Kim said. "No patient had surgery more than 46 weeks after completing neoadjuvant chemoradiation."
The median interval from completion of neoadjuvant therapy to surgery was 46 days in the short-interval group and 76 days in the delayed group. In both groups, more than 95% of patients were staged with PET-CT and endoscopic ultrasound.
The researchers compared the two groups in terms of perioperative complications, rate of pathological complete response, and overall survival.
The two groups were similar in 18 of 22 baseline characteristics examined, but they were different in four areas. Compared with their counterparts in the short-interval group, the patients in the delayed group were slightly older (mean age, 60 years vs. 57 years, respectively), had more coronary artery disease (17% vs. 7%), had less adenocarcinoma histology (87% vs. 97%), and weighed less (53% with a body mass index of 25 kg/m2 or greater vs. 75% of their counterparts in the short-interval group).
By any objective measure used to gauge the difficulty of the operation, the two groups were similar, including mean OR time (390 minutes in the short-interval group vs. 398 minutes in the delayed group), mean number of lymph nodes removed (21% vs. 20%), and mean estimated blood loss (505 mL vs. 478 mL).
The rates of major complications also were similar between the two groups, including perioperative mortality (2% in the short-interval group vs. 3% in the delayed group), median length of stay (11 days in each group), and rate of anastomotic leak (11% vs. 16%).
The rate of pathological complete response was similar between the two groups (21% vs. 23%).
Overall 5-year survival in the short-interval group was 46%, compared with 36% in the delayed-surgery group, a nonsignificant difference. Disease-free 5-year survival in the short-interval group was 44%, compared with 36% in the delayed group.
"The timing of surgery, both as a continuous and a dichotomous variable, was not associated" with perioperative complication or death, pathological complete response, or overall survival, Dr. Kim added.
On multivariable analysis, older age, more involved lymph nodes, and advanced pathological stage were independently associated with decreased survival.
The researchers performed a subgroup analysis of patients with adenocarcinoma histology and found that the results were similar.
Dr. Kim said that he had no relevant financial disclosures.
The authors point out that it is often difficult to standardize timing of esophagectomy in this setting because of a variety of difficult-to-anticipate events, particularly induction therapy toxicities. That most patients still had their esophagectomies performed by 3 months following the end of therapy suggests that the two groups might not be expected to be vastly different, although it is comforting to now have data to support this contention. Perhaps a future study might be to compare morbidity/mortality of esophagectomy following induction therapy versus that performed as a salvage strategy for patients who have failed chemoradiotherapy as definitive treatment. Those two groups are far more distinct. Late local recurrence following definitive chemoradiation therapy is, unfortunately, not that uncommon and has few other treatment options.
Dr. Sudish Murthy is an ACS Fellow and surgical director of the Center for Major Airway Disease, Cleveland Clinic.
The authors point out that it is often difficult to standardize timing of esophagectomy in this setting because of a variety of difficult-to-anticipate events, particularly induction therapy toxicities. That most patients still had their esophagectomies performed by 3 months following the end of therapy suggests that the two groups might not be expected to be vastly different, although it is comforting to now have data to support this contention. Perhaps a future study might be to compare morbidity/mortality of esophagectomy following induction therapy versus that performed as a salvage strategy for patients who have failed chemoradiotherapy as definitive treatment. Those two groups are far more distinct. Late local recurrence following definitive chemoradiation therapy is, unfortunately, not that uncommon and has few other treatment options.
Dr. Sudish Murthy is an ACS Fellow and surgical director of the Center for Major Airway Disease, Cleveland Clinic.
The authors point out that it is often difficult to standardize timing of esophagectomy in this setting because of a variety of difficult-to-anticipate events, particularly induction therapy toxicities. That most patients still had their esophagectomies performed by 3 months following the end of therapy suggests that the two groups might not be expected to be vastly different, although it is comforting to now have data to support this contention. Perhaps a future study might be to compare morbidity/mortality of esophagectomy following induction therapy versus that performed as a salvage strategy for patients who have failed chemoradiotherapy as definitive treatment. Those two groups are far more distinct. Late local recurrence following definitive chemoradiation therapy is, unfortunately, not that uncommon and has few other treatment options.
Dr. Sudish Murthy is an ACS Fellow and surgical director of the Center for Major Airway Disease, Cleveland Clinic.
FROM THE ANNUAL MEETING OF THE SOCIETY OF THORACIC SURGEONS
SAN DIEGO - An interval between neoadjuvant chemoradiation and esophagectomy that extends beyond 8 weeks is not associated with increased perioperative complications, increased pathological complete response, or change in overall survival, results from a long-term single-center study showed.
"For patients who have not yet recovered from neoadjuvant chemoradiation, it is safe to delay surgery to allow them to improve their performance status," Dr. Jae Y. Kim said at the meeting.
Traditionally, he said, surgery has been recommended within 8 weeks after completing neoadjuvant chemoradiation for esophageal cancer, yet many patients choose to delay their surgery.
Some patients have not yet recovered from chemoradiation, while others are delayed for personal or logistical reasons, explained Dr. Kim, a thoracic surgery fellow at the University of Texas M.D. Anderson Cancer Center, Houston. "The impact of this delay on outcomes is unknown." Radiation-induced tumor necrosis increases over time, he said, and there is evidence from rectal cancer that a longer interval may increase the rate of pathological complete response. "On the other hand, there are theoretical concerns that this delay may lead to increased radiation fibrosis and cause a more difficult operation. It is also possible that a delay would allow for tumor regrowth."
In an effort to determine whether an increased interval between chemoradiation and surgery is associated with risk of major perioperative complications or overall survival, Dr. Kim and his associates conducted a retrospective study of 266 patients with esophageal cancer followed by neoadjuvant chemoradiation who were treated at M.D. Anderson in 2002-2008. They divided the patients into two groups: a "short-interval" group of 150 who underwent esophagectomy within 8 weeks of chemoradiation, and a "delayed" group of 116 who underwent esophagectomy more than 8 weeks following chemoradiation.
"Most patients were clustered around 4-11 weeks," Dr. Kim said. "No patient had surgery more than 46 weeks after completing neoadjuvant chemoradiation."
The median interval from completion of neoadjuvant therapy to surgery was 46 days in the short-interval group and 76 days in the delayed group. In both groups, more than 95% of patients were staged with PET-CT and endoscopic ultrasound.
The researchers compared the two groups in terms of perioperative complications, rate of pathological complete response, and overall survival.
The two groups were similar in 18 of 22 baseline characteristics examined, but they were different in four areas. Compared with their counterparts in the short-interval group, the patients in the delayed group were slightly older (mean age, 60 years vs. 57 years, respectively), had more coronary artery disease (17% vs. 7%), had less adenocarcinoma histology (87% vs. 97%), and weighed less (53% with a body mass index of 25 kg/m2 or greater vs. 75% of their counterparts in the short-interval group).
By any objective measure used to gauge the difficulty of the operation, the two groups were similar, including mean OR time (390 minutes in the short-interval group vs. 398 minutes in the delayed group), mean number of lymph nodes removed (21% vs. 20%), and mean estimated blood loss (505 mL vs. 478 mL).
The rates of major complications also were similar between the two groups, including perioperative mortality (2% in the short-interval group vs. 3% in the delayed group), median length of stay (11 days in each group), and rate of anastomotic leak (11% vs. 16%).
The rate of pathological complete response was similar between the two groups (21% vs. 23%).
Overall 5-year survival in the short-interval group was 46%, compared with 36% in the delayed-surgery group, a nonsignificant difference. Disease-free 5-year survival in the short-interval group was 44%, compared with 36% in the delayed group.
"The timing of surgery, both as a continuous and a dichotomous variable, was not associated" with perioperative complication or death, pathological complete response, or overall survival, Dr. Kim added.
On multivariable analysis, older age, more involved lymph nodes, and advanced pathological stage were independently associated with decreased survival.
The researchers performed a subgroup analysis of patients with adenocarcinoma histology and found that the results were similar.
Dr. Kim said that he had no relevant financial disclosures.
FROM THE ANNUAL MEETING OF THE SOCIETY OF THORACIC SURGEONS
SAN DIEGO - An interval between neoadjuvant chemoradiation and esophagectomy that extends beyond 8 weeks is not associated with increased perioperative complications, increased pathological complete response, or change in overall survival, results from a long-term single-center study showed.
"For patients who have not yet recovered from neoadjuvant chemoradiation, it is safe to delay surgery to allow them to improve their performance status," Dr. Jae Y. Kim said at the meeting.
Traditionally, he said, surgery has been recommended within 8 weeks after completing neoadjuvant chemoradiation for esophageal cancer, yet many patients choose to delay their surgery.
Some patients have not yet recovered from chemoradiation, while others are delayed for personal or logistical reasons, explained Dr. Kim, a thoracic surgery fellow at the University of Texas M.D. Anderson Cancer Center, Houston. "The impact of this delay on outcomes is unknown." Radiation-induced tumor necrosis increases over time, he said, and there is evidence from rectal cancer that a longer interval may increase the rate of pathological complete response. "On the other hand, there are theoretical concerns that this delay may lead to increased radiation fibrosis and cause a more difficult operation. It is also possible that a delay would allow for tumor regrowth."
In an effort to determine whether an increased interval between chemoradiation and surgery is associated with risk of major perioperative complications or overall survival, Dr. Kim and his associates conducted a retrospective study of 266 patients with esophageal cancer followed by neoadjuvant chemoradiation who were treated at M.D. Anderson in 2002-2008. They divided the patients into two groups: a "short-interval" group of 150 who underwent esophagectomy within 8 weeks of chemoradiation, and a "delayed" group of 116 who underwent esophagectomy more than 8 weeks following chemoradiation.
"Most patients were clustered around 4-11 weeks," Dr. Kim said. "No patient had surgery more than 46 weeks after completing neoadjuvant chemoradiation."
The median interval from completion of neoadjuvant therapy to surgery was 46 days in the short-interval group and 76 days in the delayed group. In both groups, more than 95% of patients were staged with PET-CT and endoscopic ultrasound.
The researchers compared the two groups in terms of perioperative complications, rate of pathological complete response, and overall survival.
The two groups were similar in 18 of 22 baseline characteristics examined, but they were different in four areas. Compared with their counterparts in the short-interval group, the patients in the delayed group were slightly older (mean age, 60 years vs. 57 years, respectively), had more coronary artery disease (17% vs. 7%), had less adenocarcinoma histology (87% vs. 97%), and weighed less (53% with a body mass index of 25 kg/m2 or greater vs. 75% of their counterparts in the short-interval group).
By any objective measure used to gauge the difficulty of the operation, the two groups were similar, including mean OR time (390 minutes in the short-interval group vs. 398 minutes in the delayed group), mean number of lymph nodes removed (21% vs. 20%), and mean estimated blood loss (505 mL vs. 478 mL).
The rates of major complications also were similar between the two groups, including perioperative mortality (2% in the short-interval group vs. 3% in the delayed group), median length of stay (11 days in each group), and rate of anastomotic leak (11% vs. 16%).
The rate of pathological complete response was similar between the two groups (21% vs. 23%).
Overall 5-year survival in the short-interval group was 46%, compared with 36% in the delayed-surgery group, a nonsignificant difference. Disease-free 5-year survival in the short-interval group was 44%, compared with 36% in the delayed group.
"The timing of surgery, both as a continuous and a dichotomous variable, was not associated" with perioperative complication or death, pathological complete response, or overall survival, Dr. Kim added.
On multivariable analysis, older age, more involved lymph nodes, and advanced pathological stage were independently associated with decreased survival.
The researchers performed a subgroup analysis of patients with adenocarcinoma histology and found that the results were similar.
Dr. Kim said that he had no relevant financial disclosures.
MIE Oncologically Safe, Gave Better Postop Outcomes
A minimally invasive esophagectomy resulted in improved postoperative outcomes and a complete resection rate (R0) that was just as high as with open surgery, plus a significantly greater harvest of lymph nodes.
Compared with those who had open surgery, patients who underwent the minimally invasive surgery had significantly less blood loss, shorter hospital stays, and fewer major complications, said Dr. Adam C. Berger at the annual meeting of the Southern Surgical Association.
Although further studies are needed to confirm the benefits of such an approach, "Our current practice is to attempt minimally invasive esophagectomies in all patients," said Dr. Berger of the Kimmel Cancer Center at the Thomas Jefferson University Hospital in Philadelphia.
He and his colleagues presented a retrospective study comparing surgical and oncologic outcomes in two groups of patients with esophageal cancer. This comprised 65 patients who underwent esophagectomy with a thoracoscopic component (11 thoracoscopy/laparotomy, 2 Ivor-Lewis thoracoscopy/laparoscopy, and 52 three-hole thoracoscopy/laparoscopy) compared with 53 patients who underwent open surgery (15 Ivor-Lewis and 38 three-hole esophagectomy) during the same time period.
Mean patient age was 61 years. Neoadjuvant therapy was administered in 28 (43%) of the minimally invasive esophagectomy (MIE) group and 43 (81%) of the open surgery group. Adenocarcinoma was present in most patients (85% MIE and 74% open). Squamous cell carcinoma occurred in 6% of the MIE group and 26% of the open group; there were five cases (8%) of high-grade dysplasia in the MIE group. One patient had MIE for esophageal melanoma.
During the thoracoscopy, Dr. Berger uses 10-mm ports in the anterior axillary line at the fourth and eighth intercostal spaces and at the posterior axillary line. "A 5-mm port is placed at the tip of the scapula, and a suction port can be placed between the ports in the anterior axillary line," he said. "Finally, a suture is placed in the tendinous portion of the diaphragm to maintain retraction."
He uses a Penrose drain to encircle and retract the esophagus. The azygous vein is divided, and the aortic branches are divided or clipped. "The subcarinal lymph node package is dissected under direct vision and removed, and paraesophageal lymph nodes and tissue dissected up with the specimen."
During the subsequent laparoscopy, he places a 5-mm port in the right lateral subcostal area to accommodate a liver retractor, a 12-mm port in the umbilicus for the camera, an additional 12-mm working port in the right subcostal region, and a 5-mm port in the left subcostal region. "Once the gastric mobilization is complete, we often enlarge the 12-mm right subcostal port to about 5 cm and place a Lap Disk."
After dissecting the neck, he isolates and divides the esophagus. The completed reconstruction involves sewing the tip of the gastric tube to the chest tube and pulling that tube back up into the neck for a side-to-side esophagectomy. Anastomosis is in the neck with the gastric tube in the posterior mediastinum.
Mortality was similar in both groups of patients (8%). While the overall rate of complications was not significantly different (48% MIE vs. 60% open), the rate of major complications was (20% vs. 41%). Respiratory failure or acute respiratory distress syndrome occurred in a significantly smaller percentage of MIE patients (8% vs. 21%). There were also fewer cases of pneumonia among the MIE patients, but this was not a significant difference (8% vs. 18%). "Interestingly, there was a significant increase in the number of patients with a deep venous thrombosis or pulmonary embolism in the open group (2% vs. 11%)," Dr. Berger said. "This may have something to do with earlier postoperative mobilization for patients with MIE."
MIE patients also fared significantly better in terms of blood loss (182 mL vs. 619 mL) and median length of stay (9 vs. 16 days). Oncologic outcomes were as good as, or better than, those seen in open surgery. "There was no difference in R0 resection rates," Dr. Berger pointed out (97% MIE vs. 94% open). "We also saw a more than doubling of the number of examined lymph nodes in the MIE group, which was highly significant (20 vs. 9; P less than .0001)."
Dr. Berger pointed out a major limitation of the study. Because his group attempts the MIE approach in all patients, "It's impossible to find a concurrent group of patients who had open surgery for comparison." Also, he noted, "Pathology standards have changed and, thus, the importance of lymph nodes has become more recognized. Higher node yields are shown to be an important prognostic factor for survival. Therefore, our pathologists are now more diligent in finding the lymph nodes in the specimen."
Dr. Berger had no disclosures.
The authors report that the complete resection rates (R0) were similar between patients who underwent minimally invasive esophagectomy (MIE) and those who underwent an open transthoracic esophagectomy (more than 94%). However, the number of lymph nodes resected was significantly higher in the MIE group, which had fewer serious complications and a shorter length of hospital stay. In their conclusion, the authors highlight similar R0 resection rates, improved lymphadenectomy, and improved perioperative outcomes with a minimally invasive approach.
This is an interesting study, but it has some important limitations. First, it is a retrospective comparison, not a randomized study. The approaches were adopted in different time periods with potential differences in practice patterns - such as pathologic evaluation of specimens or policies regarding hospital discharge - that may have accounted for some of the differences observed. Second, although the open and MIE groups were matched for age and gender, there may be important differences between them. Neoadjuvant chemoradiation was used nearly twice as often in the open transthoracic group as in the MIE group, and this different rate might confound the analysis because it may reflect a more advanced clinical stage and be associated with an increase in perioperative complications.
The 8% mortality appears high, compared with recent series. For example, mortality was 2% in the multicenter MIE trial conducted by the Eastern Cooperative Oncology Group (ECOG 2202). Although the authors describe oncologic efficacy in terms of completeness of resection, the lack of longer-term outcome data on recurrence and survival is a major limitation.
Dr. James D. Luketich is Henry T. Bahnson Professor and chairman, department of cardiothoracic surgery, University of Pittsburgh Medical Center.
The authors report that the complete resection rates (R0) were similar between patients who underwent minimally invasive esophagectomy (MIE) and those who underwent an open transthoracic esophagectomy (more than 94%). However, the number of lymph nodes resected was significantly higher in the MIE group, which had fewer serious complications and a shorter length of hospital stay. In their conclusion, the authors highlight similar R0 resection rates, improved lymphadenectomy, and improved perioperative outcomes with a minimally invasive approach.
This is an interesting study, but it has some important limitations. First, it is a retrospective comparison, not a randomized study. The approaches were adopted in different time periods with potential differences in practice patterns - such as pathologic evaluation of specimens or policies regarding hospital discharge - that may have accounted for some of the differences observed. Second, although the open and MIE groups were matched for age and gender, there may be important differences between them. Neoadjuvant chemoradiation was used nearly twice as often in the open transthoracic group as in the MIE group, and this different rate might confound the analysis because it may reflect a more advanced clinical stage and be associated with an increase in perioperative complications.
The 8% mortality appears high, compared with recent series. For example, mortality was 2% in the multicenter MIE trial conducted by the Eastern Cooperative Oncology Group (ECOG 2202). Although the authors describe oncologic efficacy in terms of completeness of resection, the lack of longer-term outcome data on recurrence and survival is a major limitation.
Dr. James D. Luketich is Henry T. Bahnson Professor and chairman, department of cardiothoracic surgery, University of Pittsburgh Medical Center.
The authors report that the complete resection rates (R0) were similar between patients who underwent minimally invasive esophagectomy (MIE) and those who underwent an open transthoracic esophagectomy (more than 94%). However, the number of lymph nodes resected was significantly higher in the MIE group, which had fewer serious complications and a shorter length of hospital stay. In their conclusion, the authors highlight similar R0 resection rates, improved lymphadenectomy, and improved perioperative outcomes with a minimally invasive approach.
This is an interesting study, but it has some important limitations. First, it is a retrospective comparison, not a randomized study. The approaches were adopted in different time periods with potential differences in practice patterns - such as pathologic evaluation of specimens or policies regarding hospital discharge - that may have accounted for some of the differences observed. Second, although the open and MIE groups were matched for age and gender, there may be important differences between them. Neoadjuvant chemoradiation was used nearly twice as often in the open transthoracic group as in the MIE group, and this different rate might confound the analysis because it may reflect a more advanced clinical stage and be associated with an increase in perioperative complications.
The 8% mortality appears high, compared with recent series. For example, mortality was 2% in the multicenter MIE trial conducted by the Eastern Cooperative Oncology Group (ECOG 2202). Although the authors describe oncologic efficacy in terms of completeness of resection, the lack of longer-term outcome data on recurrence and survival is a major limitation.
Dr. James D. Luketich is Henry T. Bahnson Professor and chairman, department of cardiothoracic surgery, University of Pittsburgh Medical Center.
A minimally invasive esophagectomy resulted in improved postoperative outcomes and a complete resection rate (R0) that was just as high as with open surgery, plus a significantly greater harvest of lymph nodes.
Compared with those who had open surgery, patients who underwent the minimally invasive surgery had significantly less blood loss, shorter hospital stays, and fewer major complications, said Dr. Adam C. Berger at the annual meeting of the Southern Surgical Association.
Although further studies are needed to confirm the benefits of such an approach, "Our current practice is to attempt minimally invasive esophagectomies in all patients," said Dr. Berger of the Kimmel Cancer Center at the Thomas Jefferson University Hospital in Philadelphia.
He and his colleagues presented a retrospective study comparing surgical and oncologic outcomes in two groups of patients with esophageal cancer. This comprised 65 patients who underwent esophagectomy with a thoracoscopic component (11 thoracoscopy/laparotomy, 2 Ivor-Lewis thoracoscopy/laparoscopy, and 52 three-hole thoracoscopy/laparoscopy) compared with 53 patients who underwent open surgery (15 Ivor-Lewis and 38 three-hole esophagectomy) during the same time period.
Mean patient age was 61 years. Neoadjuvant therapy was administered in 28 (43%) of the minimally invasive esophagectomy (MIE) group and 43 (81%) of the open surgery group. Adenocarcinoma was present in most patients (85% MIE and 74% open). Squamous cell carcinoma occurred in 6% of the MIE group and 26% of the open group; there were five cases (8%) of high-grade dysplasia in the MIE group. One patient had MIE for esophageal melanoma.
During the thoracoscopy, Dr. Berger uses 10-mm ports in the anterior axillary line at the fourth and eighth intercostal spaces and at the posterior axillary line. "A 5-mm port is placed at the tip of the scapula, and a suction port can be placed between the ports in the anterior axillary line," he said. "Finally, a suture is placed in the tendinous portion of the diaphragm to maintain retraction."
He uses a Penrose drain to encircle and retract the esophagus. The azygous vein is divided, and the aortic branches are divided or clipped. "The subcarinal lymph node package is dissected under direct vision and removed, and paraesophageal lymph nodes and tissue dissected up with the specimen."
During the subsequent laparoscopy, he places a 5-mm port in the right lateral subcostal area to accommodate a liver retractor, a 12-mm port in the umbilicus for the camera, an additional 12-mm working port in the right subcostal region, and a 5-mm port in the left subcostal region. "Once the gastric mobilization is complete, we often enlarge the 12-mm right subcostal port to about 5 cm and place a Lap Disk."
After dissecting the neck, he isolates and divides the esophagus. The completed reconstruction involves sewing the tip of the gastric tube to the chest tube and pulling that tube back up into the neck for a side-to-side esophagectomy. Anastomosis is in the neck with the gastric tube in the posterior mediastinum.
Mortality was similar in both groups of patients (8%). While the overall rate of complications was not significantly different (48% MIE vs. 60% open), the rate of major complications was (20% vs. 41%). Respiratory failure or acute respiratory distress syndrome occurred in a significantly smaller percentage of MIE patients (8% vs. 21%). There were also fewer cases of pneumonia among the MIE patients, but this was not a significant difference (8% vs. 18%). "Interestingly, there was a significant increase in the number of patients with a deep venous thrombosis or pulmonary embolism in the open group (2% vs. 11%)," Dr. Berger said. "This may have something to do with earlier postoperative mobilization for patients with MIE."
MIE patients also fared significantly better in terms of blood loss (182 mL vs. 619 mL) and median length of stay (9 vs. 16 days). Oncologic outcomes were as good as, or better than, those seen in open surgery. "There was no difference in R0 resection rates," Dr. Berger pointed out (97% MIE vs. 94% open). "We also saw a more than doubling of the number of examined lymph nodes in the MIE group, which was highly significant (20 vs. 9; P less than .0001)."
Dr. Berger pointed out a major limitation of the study. Because his group attempts the MIE approach in all patients, "It's impossible to find a concurrent group of patients who had open surgery for comparison." Also, he noted, "Pathology standards have changed and, thus, the importance of lymph nodes has become more recognized. Higher node yields are shown to be an important prognostic factor for survival. Therefore, our pathologists are now more diligent in finding the lymph nodes in the specimen."
Dr. Berger had no disclosures.
A minimally invasive esophagectomy resulted in improved postoperative outcomes and a complete resection rate (R0) that was just as high as with open surgery, plus a significantly greater harvest of lymph nodes.
Compared with those who had open surgery, patients who underwent the minimally invasive surgery had significantly less blood loss, shorter hospital stays, and fewer major complications, said Dr. Adam C. Berger at the annual meeting of the Southern Surgical Association.
Although further studies are needed to confirm the benefits of such an approach, "Our current practice is to attempt minimally invasive esophagectomies in all patients," said Dr. Berger of the Kimmel Cancer Center at the Thomas Jefferson University Hospital in Philadelphia.
He and his colleagues presented a retrospective study comparing surgical and oncologic outcomes in two groups of patients with esophageal cancer. This comprised 65 patients who underwent esophagectomy with a thoracoscopic component (11 thoracoscopy/laparotomy, 2 Ivor-Lewis thoracoscopy/laparoscopy, and 52 three-hole thoracoscopy/laparoscopy) compared with 53 patients who underwent open surgery (15 Ivor-Lewis and 38 three-hole esophagectomy) during the same time period.
Mean patient age was 61 years. Neoadjuvant therapy was administered in 28 (43%) of the minimally invasive esophagectomy (MIE) group and 43 (81%) of the open surgery group. Adenocarcinoma was present in most patients (85% MIE and 74% open). Squamous cell carcinoma occurred in 6% of the MIE group and 26% of the open group; there were five cases (8%) of high-grade dysplasia in the MIE group. One patient had MIE for esophageal melanoma.
During the thoracoscopy, Dr. Berger uses 10-mm ports in the anterior axillary line at the fourth and eighth intercostal spaces and at the posterior axillary line. "A 5-mm port is placed at the tip of the scapula, and a suction port can be placed between the ports in the anterior axillary line," he said. "Finally, a suture is placed in the tendinous portion of the diaphragm to maintain retraction."
He uses a Penrose drain to encircle and retract the esophagus. The azygous vein is divided, and the aortic branches are divided or clipped. "The subcarinal lymph node package is dissected under direct vision and removed, and paraesophageal lymph nodes and tissue dissected up with the specimen."
During the subsequent laparoscopy, he places a 5-mm port in the right lateral subcostal area to accommodate a liver retractor, a 12-mm port in the umbilicus for the camera, an additional 12-mm working port in the right subcostal region, and a 5-mm port in the left subcostal region. "Once the gastric mobilization is complete, we often enlarge the 12-mm right subcostal port to about 5 cm and place a Lap Disk."
After dissecting the neck, he isolates and divides the esophagus. The completed reconstruction involves sewing the tip of the gastric tube to the chest tube and pulling that tube back up into the neck for a side-to-side esophagectomy. Anastomosis is in the neck with the gastric tube in the posterior mediastinum.
Mortality was similar in both groups of patients (8%). While the overall rate of complications was not significantly different (48% MIE vs. 60% open), the rate of major complications was (20% vs. 41%). Respiratory failure or acute respiratory distress syndrome occurred in a significantly smaller percentage of MIE patients (8% vs. 21%). There were also fewer cases of pneumonia among the MIE patients, but this was not a significant difference (8% vs. 18%). "Interestingly, there was a significant increase in the number of patients with a deep venous thrombosis or pulmonary embolism in the open group (2% vs. 11%)," Dr. Berger said. "This may have something to do with earlier postoperative mobilization for patients with MIE."
MIE patients also fared significantly better in terms of blood loss (182 mL vs. 619 mL) and median length of stay (9 vs. 16 days). Oncologic outcomes were as good as, or better than, those seen in open surgery. "There was no difference in R0 resection rates," Dr. Berger pointed out (97% MIE vs. 94% open). "We also saw a more than doubling of the number of examined lymph nodes in the MIE group, which was highly significant (20 vs. 9; P less than .0001)."
Dr. Berger pointed out a major limitation of the study. Because his group attempts the MIE approach in all patients, "It's impossible to find a concurrent group of patients who had open surgery for comparison." Also, he noted, "Pathology standards have changed and, thus, the importance of lymph nodes has become more recognized. Higher node yields are shown to be an important prognostic factor for survival. Therefore, our pathologists are now more diligent in finding the lymph nodes in the specimen."
Dr. Berger had no disclosures.
MIE Oncologically Safe, Gave Better Postop Outcomes
A minimally invasive esophagectomy resulted in improved postoperative outcomes and a complete resection rate (R0) that was just as high as with open surgery, plus a significantly greater harvest of lymph nodes.
Compared with those who had open surgery, patients who underwent the minimally invasive surgery had significantly less blood loss, shorter hospital stays, and fewer major complications, said Dr. Adam C. Berger at the annual meeting of the Southern Surgical Association.
Although further studies are needed to confirm the benefits of such an approach, "Our current practice is to attempt minimally invasive esophagectomies in all patients," said Dr. Berger of the Kimmel Cancer Center at the Thomas Jefferson University Hospital in Philadelphia.
He and his colleagues presented a retrospective study comparing surgical and oncologic outcomes in two groups of patients with esophageal cancer. This comprised 65 patients who underwent esophagectomy with a thoracoscopic component (11 thoracoscopy/laparotomy, 2 Ivor-Lewis thoracoscopy/laparoscopy, and 52 three-hole thoracoscopy/laparoscopy) compared with 53 patients who underwent open surgery (15 Ivor-Lewis and 38 three-hole esophagectomy) during the same time period.
Mean patient age was 61 years. Neoadjuvant therapy was administered in 28 (43%) of the minimally invasive esophagectomy (MIE) group and 43 (81%) of the open surgery group. Adenocarcinoma was present in most patients (85% MIE and 74% open). Squamous cell carcinoma occurred in 6% of the MIE group and 26% of the open group; there were five cases (8%) of high-grade dysplasia in the MIE group. One patient had MIE for esophageal melanoma.
During the thoracoscopy, Dr. Berger uses 10-mm ports in the anterior axillary line at the fourth and eighth intercostal spaces and at the posterior axillary line. "A 5-mm port is placed at the tip of the scapula, and a suction port can be placed between the ports in the anterior axillary line," he said. "Finally, a suture is placed in the tendinous portion of the diaphragm to maintain retraction."
He uses a Penrose drain to encircle and retract the esophagus. The azygous vein is divided, and the aortic branches are divided or clipped. "The subcarinal lymph node package is dissected under direct vision and removed, and paraesophageal lymph nodes and tissue dissected up with the specimen."
During the subsequent laparoscopy, he places a 5-mm port in the right lateral subcostal area to accommodate a liver retractor, a 12-mm port in the umbilicus for the camera, an additional 12-mm working port in the right subcostal region, and a 5-mm port in the left subcostal region. "Once the gastric mobilization is complete, we often enlarge the 12-mm right subcostal port to about 5 cm and place a Lap Disk."
After dissecting the neck, he isolates and divides the esophagus. The completed reconstruction involves sewing the tip of the gastric tube to the chest tube and pulling that tube back up into the neck for a side-to-side esophagectomy. Anastomosis is in the neck with the gastric tube in the posterior mediastinum.
Mortality was similar in both groups of patients (8%). While the overall rate of complications was not significantly different (48% MIE vs. 60% open), the rate of major complications was (20% vs. 41%). Respiratory failure or acute respiratory distress syndrome occurred in a significantly smaller percentage of MIE patients (8% vs. 21%). There were also fewer cases of pneumonia among the MIE patients, but this was not a significant difference (8% vs. 18%). "Interestingly, there was a significant increase in the number of patients with a deep venous thrombosis or pulmonary embolism in the open group (2% vs. 11%)," Dr. Berger said. "This may have something to do with earlier postoperative mobilization for patients with MIE."
MIE patients also fared significantly better in terms of blood loss (182 mL vs. 619 mL) and median length of stay (9 vs. 16 days). Oncologic outcomes were as good as, or better than, those seen in open surgery. "There was no difference in R0 resection rates," Dr. Berger pointed out (97% MIE vs. 94% open). "We also saw a more than doubling of the number of examined lymph nodes in the MIE group, which was highly significant (20 vs. 9; P less than .0001)."
Dr. Berger pointed out a major limitation of the study. Because his group attempts the MIE approach in all patients, "It's impossible to find a concurrent group of patients who had open surgery for comparison." Also, he noted, "Pathology standards have changed and, thus, the importance of lymph nodes has become more recognized. Higher node yields are shown to be an important prognostic factor for survival. Therefore, our pathologists are now more diligent in finding the lymph nodes in the specimen."
Dr. Berger had no disclosures.
The authors report that the complete resection rates (R0) were similar between patients who underwent minimally invasive esophagectomy (MIE) and those who underwent an open transthoracic esophagectomy (more than 94%). However, the number of lymph nodes resected was significantly higher in the MIE group, which had fewer serious complications and a shorter length of hospital stay. In their conclusion, the authors highlight similar R0 resection rates, improved lymphadenectomy, and improved perioperative outcomes with a minimally invasive approach.
This is an interesting study, but it has some important limitations. First, it is a retrospective comparison, not a randomized study. The approaches were adopted in different time periods with potential differences in practice patterns - such as pathologic evaluation of specimens or policies regarding hospital discharge - that may have accounted for some of the differences observed. Second, although the open and MIE groups were matched for age and gender, there may be important differences between them. Neoadjuvant chemoradiation was used nearly twice as often in the open transthoracic group as in the MIE group, and this different rate might confound the analysis because it may reflect a more advanced clinical stage and be associated with an increase in perioperative complications.
The 8% mortality appears high, compared with recent series. For example, mortality was 2% in the multicenter MIE trial conducted by the Eastern Cooperative Oncology Group (ECOG 2202). Although the authors describe oncologic efficacy in terms of completeness of resection, the lack of longer-term outcome data on recurrence and survival is a major limitation.
Dr. James D. Luketich is Henry T. Bahnson Professor and chairman, department of cardiothoracic surgery, University of Pittsburgh Medical Center.
The authors report that the complete resection rates (R0) were similar between patients who underwent minimally invasive esophagectomy (MIE) and those who underwent an open transthoracic esophagectomy (more than 94%). However, the number of lymph nodes resected was significantly higher in the MIE group, which had fewer serious complications and a shorter length of hospital stay. In their conclusion, the authors highlight similar R0 resection rates, improved lymphadenectomy, and improved perioperative outcomes with a minimally invasive approach.
This is an interesting study, but it has some important limitations. First, it is a retrospective comparison, not a randomized study. The approaches were adopted in different time periods with potential differences in practice patterns - such as pathologic evaluation of specimens or policies regarding hospital discharge - that may have accounted for some of the differences observed. Second, although the open and MIE groups were matched for age and gender, there may be important differences between them. Neoadjuvant chemoradiation was used nearly twice as often in the open transthoracic group as in the MIE group, and this different rate might confound the analysis because it may reflect a more advanced clinical stage and be associated with an increase in perioperative complications.
The 8% mortality appears high, compared with recent series. For example, mortality was 2% in the multicenter MIE trial conducted by the Eastern Cooperative Oncology Group (ECOG 2202). Although the authors describe oncologic efficacy in terms of completeness of resection, the lack of longer-term outcome data on recurrence and survival is a major limitation.
Dr. James D. Luketich is Henry T. Bahnson Professor and chairman, department of cardiothoracic surgery, University of Pittsburgh Medical Center.
The authors report that the complete resection rates (R0) were similar between patients who underwent minimally invasive esophagectomy (MIE) and those who underwent an open transthoracic esophagectomy (more than 94%). However, the number of lymph nodes resected was significantly higher in the MIE group, which had fewer serious complications and a shorter length of hospital stay. In their conclusion, the authors highlight similar R0 resection rates, improved lymphadenectomy, and improved perioperative outcomes with a minimally invasive approach.
This is an interesting study, but it has some important limitations. First, it is a retrospective comparison, not a randomized study. The approaches were adopted in different time periods with potential differences in practice patterns - such as pathologic evaluation of specimens or policies regarding hospital discharge - that may have accounted for some of the differences observed. Second, although the open and MIE groups were matched for age and gender, there may be important differences between them. Neoadjuvant chemoradiation was used nearly twice as often in the open transthoracic group as in the MIE group, and this different rate might confound the analysis because it may reflect a more advanced clinical stage and be associated with an increase in perioperative complications.
The 8% mortality appears high, compared with recent series. For example, mortality was 2% in the multicenter MIE trial conducted by the Eastern Cooperative Oncology Group (ECOG 2202). Although the authors describe oncologic efficacy in terms of completeness of resection, the lack of longer-term outcome data on recurrence and survival is a major limitation.
Dr. James D. Luketich is Henry T. Bahnson Professor and chairman, department of cardiothoracic surgery, University of Pittsburgh Medical Center.
A minimally invasive esophagectomy resulted in improved postoperative outcomes and a complete resection rate (R0) that was just as high as with open surgery, plus a significantly greater harvest of lymph nodes.
Compared with those who had open surgery, patients who underwent the minimally invasive surgery had significantly less blood loss, shorter hospital stays, and fewer major complications, said Dr. Adam C. Berger at the annual meeting of the Southern Surgical Association.
Although further studies are needed to confirm the benefits of such an approach, "Our current practice is to attempt minimally invasive esophagectomies in all patients," said Dr. Berger of the Kimmel Cancer Center at the Thomas Jefferson University Hospital in Philadelphia.
He and his colleagues presented a retrospective study comparing surgical and oncologic outcomes in two groups of patients with esophageal cancer. This comprised 65 patients who underwent esophagectomy with a thoracoscopic component (11 thoracoscopy/laparotomy, 2 Ivor-Lewis thoracoscopy/laparoscopy, and 52 three-hole thoracoscopy/laparoscopy) compared with 53 patients who underwent open surgery (15 Ivor-Lewis and 38 three-hole esophagectomy) during the same time period.
Mean patient age was 61 years. Neoadjuvant therapy was administered in 28 (43%) of the minimally invasive esophagectomy (MIE) group and 43 (81%) of the open surgery group. Adenocarcinoma was present in most patients (85% MIE and 74% open). Squamous cell carcinoma occurred in 6% of the MIE group and 26% of the open group; there were five cases (8%) of high-grade dysplasia in the MIE group. One patient had MIE for esophageal melanoma.
During the thoracoscopy, Dr. Berger uses 10-mm ports in the anterior axillary line at the fourth and eighth intercostal spaces and at the posterior axillary line. "A 5-mm port is placed at the tip of the scapula, and a suction port can be placed between the ports in the anterior axillary line," he said. "Finally, a suture is placed in the tendinous portion of the diaphragm to maintain retraction."
He uses a Penrose drain to encircle and retract the esophagus. The azygous vein is divided, and the aortic branches are divided or clipped. "The subcarinal lymph node package is dissected under direct vision and removed, and paraesophageal lymph nodes and tissue dissected up with the specimen."
During the subsequent laparoscopy, he places a 5-mm port in the right lateral subcostal area to accommodate a liver retractor, a 12-mm port in the umbilicus for the camera, an additional 12-mm working port in the right subcostal region, and a 5-mm port in the left subcostal region. "Once the gastric mobilization is complete, we often enlarge the 12-mm right subcostal port to about 5 cm and place a Lap Disk."
After dissecting the neck, he isolates and divides the esophagus. The completed reconstruction involves sewing the tip of the gastric tube to the chest tube and pulling that tube back up into the neck for a side-to-side esophagectomy. Anastomosis is in the neck with the gastric tube in the posterior mediastinum.
Mortality was similar in both groups of patients (8%). While the overall rate of complications was not significantly different (48% MIE vs. 60% open), the rate of major complications was (20% vs. 41%). Respiratory failure or acute respiratory distress syndrome occurred in a significantly smaller percentage of MIE patients (8% vs. 21%). There were also fewer cases of pneumonia among the MIE patients, but this was not a significant difference (8% vs. 18%). "Interestingly, there was a significant increase in the number of patients with a deep venous thrombosis or pulmonary embolism in the open group (2% vs. 11%)," Dr. Berger said. "This may have something to do with earlier postoperative mobilization for patients with MIE."
MIE patients also fared significantly better in terms of blood loss (182 mL vs. 619 mL) and median length of stay (9 vs. 16 days). Oncologic outcomes were as good as, or better than, those seen in open surgery. "There was no difference in R0 resection rates," Dr. Berger pointed out (97% MIE vs. 94% open). "We also saw a more than doubling of the number of examined lymph nodes in the MIE group, which was highly significant (20 vs. 9; P less than .0001)."
Dr. Berger pointed out a major limitation of the study. Because his group attempts the MIE approach in all patients, "It's impossible to find a concurrent group of patients who had open surgery for comparison." Also, he noted, "Pathology standards have changed and, thus, the importance of lymph nodes has become more recognized. Higher node yields are shown to be an important prognostic factor for survival. Therefore, our pathologists are now more diligent in finding the lymph nodes in the specimen."
Dr. Berger had no disclosures.
A minimally invasive esophagectomy resulted in improved postoperative outcomes and a complete resection rate (R0) that was just as high as with open surgery, plus a significantly greater harvest of lymph nodes.
Compared with those who had open surgery, patients who underwent the minimally invasive surgery had significantly less blood loss, shorter hospital stays, and fewer major complications, said Dr. Adam C. Berger at the annual meeting of the Southern Surgical Association.
Although further studies are needed to confirm the benefits of such an approach, "Our current practice is to attempt minimally invasive esophagectomies in all patients," said Dr. Berger of the Kimmel Cancer Center at the Thomas Jefferson University Hospital in Philadelphia.
He and his colleagues presented a retrospective study comparing surgical and oncologic outcomes in two groups of patients with esophageal cancer. This comprised 65 patients who underwent esophagectomy with a thoracoscopic component (11 thoracoscopy/laparotomy, 2 Ivor-Lewis thoracoscopy/laparoscopy, and 52 three-hole thoracoscopy/laparoscopy) compared with 53 patients who underwent open surgery (15 Ivor-Lewis and 38 three-hole esophagectomy) during the same time period.
Mean patient age was 61 years. Neoadjuvant therapy was administered in 28 (43%) of the minimally invasive esophagectomy (MIE) group and 43 (81%) of the open surgery group. Adenocarcinoma was present in most patients (85% MIE and 74% open). Squamous cell carcinoma occurred in 6% of the MIE group and 26% of the open group; there were five cases (8%) of high-grade dysplasia in the MIE group. One patient had MIE for esophageal melanoma.
During the thoracoscopy, Dr. Berger uses 10-mm ports in the anterior axillary line at the fourth and eighth intercostal spaces and at the posterior axillary line. "A 5-mm port is placed at the tip of the scapula, and a suction port can be placed between the ports in the anterior axillary line," he said. "Finally, a suture is placed in the tendinous portion of the diaphragm to maintain retraction."
He uses a Penrose drain to encircle and retract the esophagus. The azygous vein is divided, and the aortic branches are divided or clipped. "The subcarinal lymph node package is dissected under direct vision and removed, and paraesophageal lymph nodes and tissue dissected up with the specimen."
During the subsequent laparoscopy, he places a 5-mm port in the right lateral subcostal area to accommodate a liver retractor, a 12-mm port in the umbilicus for the camera, an additional 12-mm working port in the right subcostal region, and a 5-mm port in the left subcostal region. "Once the gastric mobilization is complete, we often enlarge the 12-mm right subcostal port to about 5 cm and place a Lap Disk."
After dissecting the neck, he isolates and divides the esophagus. The completed reconstruction involves sewing the tip of the gastric tube to the chest tube and pulling that tube back up into the neck for a side-to-side esophagectomy. Anastomosis is in the neck with the gastric tube in the posterior mediastinum.
Mortality was similar in both groups of patients (8%). While the overall rate of complications was not significantly different (48% MIE vs. 60% open), the rate of major complications was (20% vs. 41%). Respiratory failure or acute respiratory distress syndrome occurred in a significantly smaller percentage of MIE patients (8% vs. 21%). There were also fewer cases of pneumonia among the MIE patients, but this was not a significant difference (8% vs. 18%). "Interestingly, there was a significant increase in the number of patients with a deep venous thrombosis or pulmonary embolism in the open group (2% vs. 11%)," Dr. Berger said. "This may have something to do with earlier postoperative mobilization for patients with MIE."
MIE patients also fared significantly better in terms of blood loss (182 mL vs. 619 mL) and median length of stay (9 vs. 16 days). Oncologic outcomes were as good as, or better than, those seen in open surgery. "There was no difference in R0 resection rates," Dr. Berger pointed out (97% MIE vs. 94% open). "We also saw a more than doubling of the number of examined lymph nodes in the MIE group, which was highly significant (20 vs. 9; P less than .0001)."
Dr. Berger pointed out a major limitation of the study. Because his group attempts the MIE approach in all patients, "It's impossible to find a concurrent group of patients who had open surgery for comparison." Also, he noted, "Pathology standards have changed and, thus, the importance of lymph nodes has become more recognized. Higher node yields are shown to be an important prognostic factor for survival. Therefore, our pathologists are now more diligent in finding the lymph nodes in the specimen."
Dr. Berger had no disclosures.
MIE Oncologically Safe, Gave Better Postop Outcomes
A minimally invasive esophagectomy resulted in improved postoperative outcomes and a complete resection rate (R0) that was just as high as with open surgery, plus a significantly greater harvest of lymph nodes.
Compared with those who had open surgery, patients who underwent the minimally invasive surgery had significantly less blood loss, shorter hospital stays, and fewer major complications, said Dr. Adam C. Berger at the annual meeting of the Southern Surgical Association.
Although further studies are needed to confirm the benefits of such an approach, "Our current practice is to attempt minimally invasive esophagectomies in all patients," said Dr. Berger of the Kimmel Cancer Center at the Thomas Jefferson University Hospital in Philadelphia.
He and his colleagues presented a retrospective study comparing surgical and oncologic outcomes in two groups of patients with esophageal cancer. This comprised 65 patients who underwent esophagectomy with a thoracoscopic component (11 thoracoscopy/laparotomy, 2 Ivor-Lewis thoracoscopy/laparoscopy, and 52 three-hole thoracoscopy/laparoscopy) compared with 53 patients who underwent open surgery (15 Ivor-Lewis and 38 three-hole esophagectomy) during the same time period.
Mean patient age was 61 years. Neoadjuvant therapy was administered in 28 (43%) of the minimally invasive esophagectomy (MIE) group and 43 (81%) of the open surgery group. Adenocarcinoma was present in most patients (85% MIE and 74% open). Squamous cell carcinoma occurred in 6% of the MIE group and 26% of the open group; there were five cases (8%) of high-grade dysplasia in the MIE group. One patient had MIE for esophageal melanoma.
During the thoracoscopy, Dr. Berger uses 10-mm ports in the anterior axillary line at the fourth and eighth intercostal spaces and at the posterior axillary line. "A 5-mm port is placed at the tip of the scapula, and a suction port can be placed between the ports in the anterior axillary line," he said. "Finally, a suture is placed in the tendinous portion of the diaphragm to maintain retraction."
He uses a Penrose drain to encircle and retract the esophagus. The azygous vein is divided, and the aortic branches are divided or clipped. "The subcarinal lymph node package is dissected under direct vision and removed, and paraesophageal lymph nodes and tissue dissected up with the specimen."
During the subsequent laparoscopy, he places a 5-mm port in the right lateral subcostal area to accommodate a liver retractor, a 12-mm port in the umbilicus for the camera, an additional 12-mm working port in the right subcostal region, and a 5-mm port in the left subcostal region. "Once the gastric mobilization is complete, we often enlarge the 12-mm right subcostal port to about 5 cm and place a Lap Disk."
After dissecting the neck, he isolates and divides the esophagus. The completed reconstruction involves sewing the tip of the gastric tube to the chest tube and pulling that tube back up into the neck for a side-to-side esophagectomy. Anastomosis is in the neck with the gastric tube in the posterior mediastinum.
Mortality was similar in both groups of patients (8%). While the overall rate of complications was not significantly different (48% MIE vs. 60% open), the rate of major complications was (20% vs. 41%). Respiratory failure or acute respiratory distress syndrome occurred in a significantly smaller percentage of MIE patients (8% vs. 21%). There were also fewer cases of pneumonia among the MIE patients, but this was not a significant difference (8% vs. 18%). "Interestingly, there was a significant increase in the number of patients with a deep venous thrombosis or pulmonary embolism in the open group (2% vs. 11%)," Dr. Berger said. "This may have something to do with earlier postoperative mobilization for patients with MIE."
MIE patients also fared significantly better in terms of blood loss (182 mL vs. 619 mL) and median length of stay (9 vs. 16 days). Oncologic outcomes were as good as, or better than, those seen in open surgery. "There was no difference in R0 resection rates," Dr. Berger pointed out (97% MIE vs. 94% open). "We also saw a more than doubling of the number of examined lymph nodes in the MIE group, which was highly significant (20 vs. 9; P less than .0001)."
Dr. Berger pointed out a major limitation of the study. Because his group attempts the MIE approach in all patients, "It's impossible to find a concurrent group of patients who had open surgery for comparison." Also, he noted, "Pathology standards have changed and, thus, the importance of lymph nodes has become more recognized. Higher node yields are shown to be an important prognostic factor for survival. Therefore, our pathologists are now more diligent in finding the lymph nodes in the specimen."
Dr. Berger had no disclosures.
The authors report that the complete resection rates (R0) were similar between patients who underwent minimally invasive esophagectomy (MIE) and those who underwent an open transthoracic esophagectomy (more than 94%). However, the number of lymph nodes resected was significantly higher in the MIE group, which had fewer serious complications and a shorter length of hospital stay. In their conclusion, the authors highlight similar R0 resection rates, improved lymphadenectomy, and improved perioperative outcomes with a minimally invasive approach.
This is an interesting study, but it has some important limitations. First, it is a retrospective comparison, not a randomized study. The approaches were adopted in different time periods with potential differences in practice patterns - such as pathologic evaluation of specimens or policies regarding hospital discharge - that may have accounted for some of the differences observed. Second, although the open and MIE groups were matched for age and gender, there may be important differences between them. Neoadjuvant chemoradiation was used nearly twice as often in the open transthoracic group as in the MIE group, and this different rate might confound the analysis because it may reflect a more advanced clinical stage and be associated with an increase in perioperative complications.
The 8% mortality appears high, compared with recent series. For example, mortality was 2% in the multicenter MIE trial conducted by the Eastern Cooperative Oncology Group (ECOG 2202). Although the authors describe oncologic efficacy in terms of completeness of resection, the lack of longer-term outcome data on recurrence and survival is a major limitation.
Dr. James D. Luketich is Henry T. Bahnson Professor and chairman, department of cardiothoracic surgery, University of Pittsburgh Medical Center.
The authors report that the complete resection rates (R0) were similar between patients who underwent minimally invasive esophagectomy (MIE) and those who underwent an open transthoracic esophagectomy (more than 94%). However, the number of lymph nodes resected was significantly higher in the MIE group, which had fewer serious complications and a shorter length of hospital stay. In their conclusion, the authors highlight similar R0 resection rates, improved lymphadenectomy, and improved perioperative outcomes with a minimally invasive approach.
This is an interesting study, but it has some important limitations. First, it is a retrospective comparison, not a randomized study. The approaches were adopted in different time periods with potential differences in practice patterns - such as pathologic evaluation of specimens or policies regarding hospital discharge - that may have accounted for some of the differences observed. Second, although the open and MIE groups were matched for age and gender, there may be important differences between them. Neoadjuvant chemoradiation was used nearly twice as often in the open transthoracic group as in the MIE group, and this different rate might confound the analysis because it may reflect a more advanced clinical stage and be associated with an increase in perioperative complications.
The 8% mortality appears high, compared with recent series. For example, mortality was 2% in the multicenter MIE trial conducted by the Eastern Cooperative Oncology Group (ECOG 2202). Although the authors describe oncologic efficacy in terms of completeness of resection, the lack of longer-term outcome data on recurrence and survival is a major limitation.
Dr. James D. Luketich is Henry T. Bahnson Professor and chairman, department of cardiothoracic surgery, University of Pittsburgh Medical Center.
The authors report that the complete resection rates (R0) were similar between patients who underwent minimally invasive esophagectomy (MIE) and those who underwent an open transthoracic esophagectomy (more than 94%). However, the number of lymph nodes resected was significantly higher in the MIE group, which had fewer serious complications and a shorter length of hospital stay. In their conclusion, the authors highlight similar R0 resection rates, improved lymphadenectomy, and improved perioperative outcomes with a minimally invasive approach.
This is an interesting study, but it has some important limitations. First, it is a retrospective comparison, not a randomized study. The approaches were adopted in different time periods with potential differences in practice patterns - such as pathologic evaluation of specimens or policies regarding hospital discharge - that may have accounted for some of the differences observed. Second, although the open and MIE groups were matched for age and gender, there may be important differences between them. Neoadjuvant chemoradiation was used nearly twice as often in the open transthoracic group as in the MIE group, and this different rate might confound the analysis because it may reflect a more advanced clinical stage and be associated with an increase in perioperative complications.
The 8% mortality appears high, compared with recent series. For example, mortality was 2% in the multicenter MIE trial conducted by the Eastern Cooperative Oncology Group (ECOG 2202). Although the authors describe oncologic efficacy in terms of completeness of resection, the lack of longer-term outcome data on recurrence and survival is a major limitation.
Dr. James D. Luketich is Henry T. Bahnson Professor and chairman, department of cardiothoracic surgery, University of Pittsburgh Medical Center.
A minimally invasive esophagectomy resulted in improved postoperative outcomes and a complete resection rate (R0) that was just as high as with open surgery, plus a significantly greater harvest of lymph nodes.
Compared with those who had open surgery, patients who underwent the minimally invasive surgery had significantly less blood loss, shorter hospital stays, and fewer major complications, said Dr. Adam C. Berger at the annual meeting of the Southern Surgical Association.
Although further studies are needed to confirm the benefits of such an approach, "Our current practice is to attempt minimally invasive esophagectomies in all patients," said Dr. Berger of the Kimmel Cancer Center at the Thomas Jefferson University Hospital in Philadelphia.
He and his colleagues presented a retrospective study comparing surgical and oncologic outcomes in two groups of patients with esophageal cancer. This comprised 65 patients who underwent esophagectomy with a thoracoscopic component (11 thoracoscopy/laparotomy, 2 Ivor-Lewis thoracoscopy/laparoscopy, and 52 three-hole thoracoscopy/laparoscopy) compared with 53 patients who underwent open surgery (15 Ivor-Lewis and 38 three-hole esophagectomy) during the same time period.
Mean patient age was 61 years. Neoadjuvant therapy was administered in 28 (43%) of the minimally invasive esophagectomy (MIE) group and 43 (81%) of the open surgery group. Adenocarcinoma was present in most patients (85% MIE and 74% open). Squamous cell carcinoma occurred in 6% of the MIE group and 26% of the open group; there were five cases (8%) of high-grade dysplasia in the MIE group. One patient had MIE for esophageal melanoma.
During the thoracoscopy, Dr. Berger uses 10-mm ports in the anterior axillary line at the fourth and eighth intercostal spaces and at the posterior axillary line. "A 5-mm port is placed at the tip of the scapula, and a suction port can be placed between the ports in the anterior axillary line," he said. "Finally, a suture is placed in the tendinous portion of the diaphragm to maintain retraction."
He uses a Penrose drain to encircle and retract the esophagus. The azygous vein is divided, and the aortic branches are divided or clipped. "The subcarinal lymph node package is dissected under direct vision and removed, and paraesophageal lymph nodes and tissue dissected up with the specimen."
During the subsequent laparoscopy, he places a 5-mm port in the right lateral subcostal area to accommodate a liver retractor, a 12-mm port in the umbilicus for the camera, an additional 12-mm working port in the right subcostal region, and a 5-mm port in the left subcostal region. "Once the gastric mobilization is complete, we often enlarge the 12-mm right subcostal port to about 5 cm and place a Lap Disk."
After dissecting the neck, he isolates and divides the esophagus. The completed reconstruction involves sewing the tip of the gastric tube to the chest tube and pulling that tube back up into the neck for a side-to-side esophagectomy. Anastomosis is in the neck with the gastric tube in the posterior mediastinum.
Mortality was similar in both groups of patients (8%). While the overall rate of complications was not significantly different (48% MIE vs. 60% open), the rate of major complications was (20% vs. 41%). Respiratory failure or acute respiratory distress syndrome occurred in a significantly smaller percentage of MIE patients (8% vs. 21%). There were also fewer cases of pneumonia among the MIE patients, but this was not a significant difference (8% vs. 18%). "Interestingly, there was a significant increase in the number of patients with a deep venous thrombosis or pulmonary embolism in the open group (2% vs. 11%)," Dr. Berger said. "This may have something to do with earlier postoperative mobilization for patients with MIE."
MIE patients also fared significantly better in terms of blood loss (182 mL vs. 619 mL) and median length of stay (9 vs. 16 days). Oncologic outcomes were as good as, or better than, those seen in open surgery. "There was no difference in R0 resection rates," Dr. Berger pointed out (97% MIE vs. 94% open). "We also saw a more than doubling of the number of examined lymph nodes in the MIE group, which was highly significant (20 vs. 9; P less than .0001)."
Dr. Berger pointed out a major limitation of the study. Because his group attempts the MIE approach in all patients, "It's impossible to find a concurrent group of patients who had open surgery for comparison." Also, he noted, "Pathology standards have changed and, thus, the importance of lymph nodes has become more recognized. Higher node yields are shown to be an important prognostic factor for survival. Therefore, our pathologists are now more diligent in finding the lymph nodes in the specimen."
Dr. Berger had no disclosures.
A minimally invasive esophagectomy resulted in improved postoperative outcomes and a complete resection rate (R0) that was just as high as with open surgery, plus a significantly greater harvest of lymph nodes.
Compared with those who had open surgery, patients who underwent the minimally invasive surgery had significantly less blood loss, shorter hospital stays, and fewer major complications, said Dr. Adam C. Berger at the annual meeting of the Southern Surgical Association.
Although further studies are needed to confirm the benefits of such an approach, "Our current practice is to attempt minimally invasive esophagectomies in all patients," said Dr. Berger of the Kimmel Cancer Center at the Thomas Jefferson University Hospital in Philadelphia.
He and his colleagues presented a retrospective study comparing surgical and oncologic outcomes in two groups of patients with esophageal cancer. This comprised 65 patients who underwent esophagectomy with a thoracoscopic component (11 thoracoscopy/laparotomy, 2 Ivor-Lewis thoracoscopy/laparoscopy, and 52 three-hole thoracoscopy/laparoscopy) compared with 53 patients who underwent open surgery (15 Ivor-Lewis and 38 three-hole esophagectomy) during the same time period.
Mean patient age was 61 years. Neoadjuvant therapy was administered in 28 (43%) of the minimally invasive esophagectomy (MIE) group and 43 (81%) of the open surgery group. Adenocarcinoma was present in most patients (85% MIE and 74% open). Squamous cell carcinoma occurred in 6% of the MIE group and 26% of the open group; there were five cases (8%) of high-grade dysplasia in the MIE group. One patient had MIE for esophageal melanoma.
During the thoracoscopy, Dr. Berger uses 10-mm ports in the anterior axillary line at the fourth and eighth intercostal spaces and at the posterior axillary line. "A 5-mm port is placed at the tip of the scapula, and a suction port can be placed between the ports in the anterior axillary line," he said. "Finally, a suture is placed in the tendinous portion of the diaphragm to maintain retraction."
He uses a Penrose drain to encircle and retract the esophagus. The azygous vein is divided, and the aortic branches are divided or clipped. "The subcarinal lymph node package is dissected under direct vision and removed, and paraesophageal lymph nodes and tissue dissected up with the specimen."
During the subsequent laparoscopy, he places a 5-mm port in the right lateral subcostal area to accommodate a liver retractor, a 12-mm port in the umbilicus for the camera, an additional 12-mm working port in the right subcostal region, and a 5-mm port in the left subcostal region. "Once the gastric mobilization is complete, we often enlarge the 12-mm right subcostal port to about 5 cm and place a Lap Disk."
After dissecting the neck, he isolates and divides the esophagus. The completed reconstruction involves sewing the tip of the gastric tube to the chest tube and pulling that tube back up into the neck for a side-to-side esophagectomy. Anastomosis is in the neck with the gastric tube in the posterior mediastinum.
Mortality was similar in both groups of patients (8%). While the overall rate of complications was not significantly different (48% MIE vs. 60% open), the rate of major complications was (20% vs. 41%). Respiratory failure or acute respiratory distress syndrome occurred in a significantly smaller percentage of MIE patients (8% vs. 21%). There were also fewer cases of pneumonia among the MIE patients, but this was not a significant difference (8% vs. 18%). "Interestingly, there was a significant increase in the number of patients with a deep venous thrombosis or pulmonary embolism in the open group (2% vs. 11%)," Dr. Berger said. "This may have something to do with earlier postoperative mobilization for patients with MIE."
MIE patients also fared significantly better in terms of blood loss (182 mL vs. 619 mL) and median length of stay (9 vs. 16 days). Oncologic outcomes were as good as, or better than, those seen in open surgery. "There was no difference in R0 resection rates," Dr. Berger pointed out (97% MIE vs. 94% open). "We also saw a more than doubling of the number of examined lymph nodes in the MIE group, which was highly significant (20 vs. 9; P less than .0001)."
Dr. Berger pointed out a major limitation of the study. Because his group attempts the MIE approach in all patients, "It's impossible to find a concurrent group of patients who had open surgery for comparison." Also, he noted, "Pathology standards have changed and, thus, the importance of lymph nodes has become more recognized. Higher node yields are shown to be an important prognostic factor for survival. Therefore, our pathologists are now more diligent in finding the lymph nodes in the specimen."
Dr. Berger had no disclosures.