User login
The Official Newspaper of the American Association for Thoracic Surgery
PROACT Trial: Lower INR goal acceptable
MINNEAPOLIS – High-risk patients receiving the On-X mechanical aortic valve can be safely managed with less aggressive anticoagulation than currently recommended, interim results of the PROACT trial suggest.*
The lower target international normalized ratio (INR) in the trial resulted in a decline of more than 50% in bleeding events and did not increase the risk of thromboembolism, reported Dr. John Puskas, international principal investigator for PROACT (Prospective Randomized On-X Anticoagulation Clinical Trial) and associate chief of cardiothoracic surgery at Emory University in Atlanta.
"This aortic bileaflet mechanical valve may be safely managed in these select patients at an INR of 1.5 to 2.0, with daily low-dose aspirin," he said at the annual meeting of the American Association for Thoracic Surgery.
Current American College of Cardiology and American Heart Association guidelines recommend that warfarin be dosed to achieve an INR of 2.0 to 3.0 after implantation of a bileaflet mechanical valve, and that once-daily aspirin 75-100 mg be added for all patients with mechanical heart valves.
Dr. Puskas and his associates analyzed data from 375 high-risk patients randomly assigned to lower-dose warfarin (INR 1.5-2.0) or to continue standard-dose warfarin (INR 2.0-3.0), 3 months after implantation with the On-X bileaflet mechanical heart valve. All patients received aspirin 81 mg daily. INR was adjusted by rigorous home self-monitoring, with an average of 9 days between readings and at least 96% compliance.
High-risk patients included those with chronic atrial fibrillation, left ventricular ejection fraction less than 30%, ventricular aneurysm, left atrium diameter greater than 50 mm, prior neurological events, on estrogen replacement therapy, hypercoagulability, or inadequate platelet response to aspirin or clopidogrel (Plavix). There were 185 patients in the experimental, test arm and 190 in the control arm. After randomization, 11 test patients had a neurological event (5 strokes, 6 transient ischemic attacks) and crossed over to the control group, per protocol.
After an average follow-up of 3.82 years, patients managed with a lower target INR had a significant benefit compared with controls with respect to number of major bleeding events (10 vs. 25, respectively), minor bleeds (8 vs. 25), total bleeds (18 vs. 50), and all bleeding and thrombus (38 vs. 64), Dr. Puskas said. The corresponding rate ratios (RRs) were 0.45, 0.36, 0.40, and 0.66.
There was no difference between groups in the composite primary endpoint of major bleed, stroke, transient ischemic attack, thromboembolic events and thrombosis (30 events vs. 39 events; RR, 0.86; P = .54), he said.
Specifically, hemorrhagic stroke occurred in 1 test patient and 2 controls (RR, 0.56) and ischemic stroke in 5 patients in each group (RR, 1.12), he said.
Valve-related mortality was also similar in the test and control groups (5 deaths vs. 4 deaths), as was total mortality (10 vs. 11), Dr. Puskas said.
Invited discussant Dr. A. Pieter Kappetein, a member of the RE-ALIGN trial steering committee and professor of thoracic surgery at Erasmus Medical Center in Rotterdam, the Netherlands, said the number of patients in the analysis was extremely low and questioned the validity of combining bleeding and thromboembolic events in the primary endpoint.
"In this study, you mix the efficacy endpoint with the safety endpoints," he said, observing that they move in opposite directions.
In light of such large-scale trials as ARISTOTLE and RELY, he asked whether PROACT should be considered a pilot trial and whether a new trial, designed with roughly 8,000 patients, should be performed that would also include newer anticoagulation agents to adequately evaluate reduced anticoagulation in mechanical valves.
"Is it not potentially dangerous if we do not know what the increase is for thrombosis and follow your conclusions?" he added.
Dr. Puskas said he shared Dr. Kappetein’s concern about the noninferiority design of the trial and that it was a topic of great discussion with the Food and Drug Administration (FDA). He also agreed that thrombotic events and bleeding events move in the opposite direction.
"What we are really looking for is to determine the sweet spot where those two curves intersect," he said. "While it is theoretically and intellectually correct to say that thrombosis is the efficacy issue and hemorrhage is the safety issue, we are obliged to combine those for two reasons.
"The first is practical; no company will sponsor an 8,000-patient trial, and second, this is, in fact, a trade-off in the minds of patients and clinicians. So, it is a relevant clinical endpoint – the unholy composite, if you will – of thrombosis and hemorrhage."
Finally, a member of the audience asked whether the results would hold up with standard management because universal point-of-care home testing is not the "real world" in the United States.
Dr. Puskas replied that it is in Scandinavia and other parts of the world, and admonished American clinicians, including himself, "to catch up to what should be standard of care." He noted that, based on the roughly 53,000 INR readings in PROACT, controlling INR within your range was more important in terms of adverse events, particularly hemorrhagic events, than what arm patients were assigned to.
"Home monitoring is available, it’s not high tech and it’s much easier for patients," he said. "To be perfectly blunt, there’s really no excuse for us not using it uniformly in America. Quite frankly, it is a conflict of interest between local caregivers and their patients’ well-being.
"There is a small revenue stream to cardiology offices and primary care doctors running Coumadin clinics, and that is keeping us in the system that we have now rather than home monitoring through bigger, centralized Coumadin clinics."
Dr. Puskas did not report data on PROACT’s low-risk arm managed with clopidogrel 75 mg/day plus aspirin 325 mg/day, or a third arm managed on warfarin at an INR of 2.0-2.5 plus aspirin 81 mg/day. The low-risk data will not be available for at least one more year, although the investigators are in discussion with the FDA about a possible interim analysis, he said in an interview.
The evaluable high-risk patients were 79% male, 93% were in sinus rhythm preoperatively, and concomitant procedures included coronary artery bypass grafting in 27%, aortic aneurysm repair in 14%, and other procedures in 25%. Their average age was 55 years.
Life Technologies sponsored the study. Dr. Puskas reported having no financial relationship with Life Technologies.
Correction, 6/18/2013: An earlier version of this article stated that the On-X aortic valve is investigational. This was a misstatement. The valve itself has been approved in the United States since 2001. The PROACT trial applications of lowered INR and an aspirin/Plavix regimen are not approved.
MINNEAPOLIS – High-risk patients receiving the On-X mechanical aortic valve can be safely managed with less aggressive anticoagulation than currently recommended, interim results of the PROACT trial suggest.*
The lower target international normalized ratio (INR) in the trial resulted in a decline of more than 50% in bleeding events and did not increase the risk of thromboembolism, reported Dr. John Puskas, international principal investigator for PROACT (Prospective Randomized On-X Anticoagulation Clinical Trial) and associate chief of cardiothoracic surgery at Emory University in Atlanta.
"This aortic bileaflet mechanical valve may be safely managed in these select patients at an INR of 1.5 to 2.0, with daily low-dose aspirin," he said at the annual meeting of the American Association for Thoracic Surgery.
Current American College of Cardiology and American Heart Association guidelines recommend that warfarin be dosed to achieve an INR of 2.0 to 3.0 after implantation of a bileaflet mechanical valve, and that once-daily aspirin 75-100 mg be added for all patients with mechanical heart valves.
Dr. Puskas and his associates analyzed data from 375 high-risk patients randomly assigned to lower-dose warfarin (INR 1.5-2.0) or to continue standard-dose warfarin (INR 2.0-3.0), 3 months after implantation with the On-X bileaflet mechanical heart valve. All patients received aspirin 81 mg daily. INR was adjusted by rigorous home self-monitoring, with an average of 9 days between readings and at least 96% compliance.
High-risk patients included those with chronic atrial fibrillation, left ventricular ejection fraction less than 30%, ventricular aneurysm, left atrium diameter greater than 50 mm, prior neurological events, on estrogen replacement therapy, hypercoagulability, or inadequate platelet response to aspirin or clopidogrel (Plavix). There were 185 patients in the experimental, test arm and 190 in the control arm. After randomization, 11 test patients had a neurological event (5 strokes, 6 transient ischemic attacks) and crossed over to the control group, per protocol.
After an average follow-up of 3.82 years, patients managed with a lower target INR had a significant benefit compared with controls with respect to number of major bleeding events (10 vs. 25, respectively), minor bleeds (8 vs. 25), total bleeds (18 vs. 50), and all bleeding and thrombus (38 vs. 64), Dr. Puskas said. The corresponding rate ratios (RRs) were 0.45, 0.36, 0.40, and 0.66.
There was no difference between groups in the composite primary endpoint of major bleed, stroke, transient ischemic attack, thromboembolic events and thrombosis (30 events vs. 39 events; RR, 0.86; P = .54), he said.
Specifically, hemorrhagic stroke occurred in 1 test patient and 2 controls (RR, 0.56) and ischemic stroke in 5 patients in each group (RR, 1.12), he said.
Valve-related mortality was also similar in the test and control groups (5 deaths vs. 4 deaths), as was total mortality (10 vs. 11), Dr. Puskas said.
Invited discussant Dr. A. Pieter Kappetein, a member of the RE-ALIGN trial steering committee and professor of thoracic surgery at Erasmus Medical Center in Rotterdam, the Netherlands, said the number of patients in the analysis was extremely low and questioned the validity of combining bleeding and thromboembolic events in the primary endpoint.
"In this study, you mix the efficacy endpoint with the safety endpoints," he said, observing that they move in opposite directions.
In light of such large-scale trials as ARISTOTLE and RELY, he asked whether PROACT should be considered a pilot trial and whether a new trial, designed with roughly 8,000 patients, should be performed that would also include newer anticoagulation agents to adequately evaluate reduced anticoagulation in mechanical valves.
"Is it not potentially dangerous if we do not know what the increase is for thrombosis and follow your conclusions?" he added.
Dr. Puskas said he shared Dr. Kappetein’s concern about the noninferiority design of the trial and that it was a topic of great discussion with the Food and Drug Administration (FDA). He also agreed that thrombotic events and bleeding events move in the opposite direction.
"What we are really looking for is to determine the sweet spot where those two curves intersect," he said. "While it is theoretically and intellectually correct to say that thrombosis is the efficacy issue and hemorrhage is the safety issue, we are obliged to combine those for two reasons.
"The first is practical; no company will sponsor an 8,000-patient trial, and second, this is, in fact, a trade-off in the minds of patients and clinicians. So, it is a relevant clinical endpoint – the unholy composite, if you will – of thrombosis and hemorrhage."
Finally, a member of the audience asked whether the results would hold up with standard management because universal point-of-care home testing is not the "real world" in the United States.
Dr. Puskas replied that it is in Scandinavia and other parts of the world, and admonished American clinicians, including himself, "to catch up to what should be standard of care." He noted that, based on the roughly 53,000 INR readings in PROACT, controlling INR within your range was more important in terms of adverse events, particularly hemorrhagic events, than what arm patients were assigned to.
"Home monitoring is available, it’s not high tech and it’s much easier for patients," he said. "To be perfectly blunt, there’s really no excuse for us not using it uniformly in America. Quite frankly, it is a conflict of interest between local caregivers and their patients’ well-being.
"There is a small revenue stream to cardiology offices and primary care doctors running Coumadin clinics, and that is keeping us in the system that we have now rather than home monitoring through bigger, centralized Coumadin clinics."
Dr. Puskas did not report data on PROACT’s low-risk arm managed with clopidogrel 75 mg/day plus aspirin 325 mg/day, or a third arm managed on warfarin at an INR of 2.0-2.5 plus aspirin 81 mg/day. The low-risk data will not be available for at least one more year, although the investigators are in discussion with the FDA about a possible interim analysis, he said in an interview.
The evaluable high-risk patients were 79% male, 93% were in sinus rhythm preoperatively, and concomitant procedures included coronary artery bypass grafting in 27%, aortic aneurysm repair in 14%, and other procedures in 25%. Their average age was 55 years.
Life Technologies sponsored the study. Dr. Puskas reported having no financial relationship with Life Technologies.
Correction, 6/18/2013: An earlier version of this article stated that the On-X aortic valve is investigational. This was a misstatement. The valve itself has been approved in the United States since 2001. The PROACT trial applications of lowered INR and an aspirin/Plavix regimen are not approved.
MINNEAPOLIS – High-risk patients receiving the On-X mechanical aortic valve can be safely managed with less aggressive anticoagulation than currently recommended, interim results of the PROACT trial suggest.*
The lower target international normalized ratio (INR) in the trial resulted in a decline of more than 50% in bleeding events and did not increase the risk of thromboembolism, reported Dr. John Puskas, international principal investigator for PROACT (Prospective Randomized On-X Anticoagulation Clinical Trial) and associate chief of cardiothoracic surgery at Emory University in Atlanta.
"This aortic bileaflet mechanical valve may be safely managed in these select patients at an INR of 1.5 to 2.0, with daily low-dose aspirin," he said at the annual meeting of the American Association for Thoracic Surgery.
Current American College of Cardiology and American Heart Association guidelines recommend that warfarin be dosed to achieve an INR of 2.0 to 3.0 after implantation of a bileaflet mechanical valve, and that once-daily aspirin 75-100 mg be added for all patients with mechanical heart valves.
Dr. Puskas and his associates analyzed data from 375 high-risk patients randomly assigned to lower-dose warfarin (INR 1.5-2.0) or to continue standard-dose warfarin (INR 2.0-3.0), 3 months after implantation with the On-X bileaflet mechanical heart valve. All patients received aspirin 81 mg daily. INR was adjusted by rigorous home self-monitoring, with an average of 9 days between readings and at least 96% compliance.
High-risk patients included those with chronic atrial fibrillation, left ventricular ejection fraction less than 30%, ventricular aneurysm, left atrium diameter greater than 50 mm, prior neurological events, on estrogen replacement therapy, hypercoagulability, or inadequate platelet response to aspirin or clopidogrel (Plavix). There were 185 patients in the experimental, test arm and 190 in the control arm. After randomization, 11 test patients had a neurological event (5 strokes, 6 transient ischemic attacks) and crossed over to the control group, per protocol.
After an average follow-up of 3.82 years, patients managed with a lower target INR had a significant benefit compared with controls with respect to number of major bleeding events (10 vs. 25, respectively), minor bleeds (8 vs. 25), total bleeds (18 vs. 50), and all bleeding and thrombus (38 vs. 64), Dr. Puskas said. The corresponding rate ratios (RRs) were 0.45, 0.36, 0.40, and 0.66.
There was no difference between groups in the composite primary endpoint of major bleed, stroke, transient ischemic attack, thromboembolic events and thrombosis (30 events vs. 39 events; RR, 0.86; P = .54), he said.
Specifically, hemorrhagic stroke occurred in 1 test patient and 2 controls (RR, 0.56) and ischemic stroke in 5 patients in each group (RR, 1.12), he said.
Valve-related mortality was also similar in the test and control groups (5 deaths vs. 4 deaths), as was total mortality (10 vs. 11), Dr. Puskas said.
Invited discussant Dr. A. Pieter Kappetein, a member of the RE-ALIGN trial steering committee and professor of thoracic surgery at Erasmus Medical Center in Rotterdam, the Netherlands, said the number of patients in the analysis was extremely low and questioned the validity of combining bleeding and thromboembolic events in the primary endpoint.
"In this study, you mix the efficacy endpoint with the safety endpoints," he said, observing that they move in opposite directions.
In light of such large-scale trials as ARISTOTLE and RELY, he asked whether PROACT should be considered a pilot trial and whether a new trial, designed with roughly 8,000 patients, should be performed that would also include newer anticoagulation agents to adequately evaluate reduced anticoagulation in mechanical valves.
"Is it not potentially dangerous if we do not know what the increase is for thrombosis and follow your conclusions?" he added.
Dr. Puskas said he shared Dr. Kappetein’s concern about the noninferiority design of the trial and that it was a topic of great discussion with the Food and Drug Administration (FDA). He also agreed that thrombotic events and bleeding events move in the opposite direction.
"What we are really looking for is to determine the sweet spot where those two curves intersect," he said. "While it is theoretically and intellectually correct to say that thrombosis is the efficacy issue and hemorrhage is the safety issue, we are obliged to combine those for two reasons.
"The first is practical; no company will sponsor an 8,000-patient trial, and second, this is, in fact, a trade-off in the minds of patients and clinicians. So, it is a relevant clinical endpoint – the unholy composite, if you will – of thrombosis and hemorrhage."
Finally, a member of the audience asked whether the results would hold up with standard management because universal point-of-care home testing is not the "real world" in the United States.
Dr. Puskas replied that it is in Scandinavia and other parts of the world, and admonished American clinicians, including himself, "to catch up to what should be standard of care." He noted that, based on the roughly 53,000 INR readings in PROACT, controlling INR within your range was more important in terms of adverse events, particularly hemorrhagic events, than what arm patients were assigned to.
"Home monitoring is available, it’s not high tech and it’s much easier for patients," he said. "To be perfectly blunt, there’s really no excuse for us not using it uniformly in America. Quite frankly, it is a conflict of interest between local caregivers and their patients’ well-being.
"There is a small revenue stream to cardiology offices and primary care doctors running Coumadin clinics, and that is keeping us in the system that we have now rather than home monitoring through bigger, centralized Coumadin clinics."
Dr. Puskas did not report data on PROACT’s low-risk arm managed with clopidogrel 75 mg/day plus aspirin 325 mg/day, or a third arm managed on warfarin at an INR of 2.0-2.5 plus aspirin 81 mg/day. The low-risk data will not be available for at least one more year, although the investigators are in discussion with the FDA about a possible interim analysis, he said in an interview.
The evaluable high-risk patients were 79% male, 93% were in sinus rhythm preoperatively, and concomitant procedures included coronary artery bypass grafting in 27%, aortic aneurysm repair in 14%, and other procedures in 25%. Their average age was 55 years.
Life Technologies sponsored the study. Dr. Puskas reported having no financial relationship with Life Technologies.
Correction, 6/18/2013: An earlier version of this article stated that the On-X aortic valve is investigational. This was a misstatement. The valve itself has been approved in the United States since 2001. The PROACT trial applications of lowered INR and an aspirin/Plavix regimen are not approved.
AT THE AATS ANNUAL MEETING
Major finding: The composite primary endpoint of major bleed, stroke, transient ischemic attack, thromboembolic events, and thrombosis occurred in 30 patients managed with less aggressive anticoagulation and in 39 managed with standard warfarin anticoagulation (rate ratio, 0.86; P = .54).
Data source: Interim analysis of 375 high-risk aortic valve replacement patients in the Prospective Randomized On-X Anticoagulation Trial.
Disclosures: Life Technologies sponsored the study. Dr. Puskas reported having no financial relationship with Life Technologies.
Commission proposes shifting away from fee-for-service to alternatives
The fee-for-service payment system has contributed to high health care costs and inconsistent quality of care and should be replaced with a blended payment model that includes fixed payments, according to a new report released by a panel of physicians and health care experts.
In a recently published report, the National Commission on Physician Payment Reform recommended phasing out the current fee-for-service system over 5 years in favor of bundled payments, capitation, and increased financial risk-sharing.
"We can’t control runaway medical spending without changing how doctors get paid," Dr. Bill Frist, honorary chair of the commission and former Senate majority leader, said in a statement.
"This is a bipartisan issue. We all want to get the most from our health care dollars and that requires rethinking the way we pay for health care."
But the 14-member commission predicted that fee-for-service would continue to play a large role. By the end of the decade, they called for a blended system of fee-for-service, fixed payments, and salary.
The commission also recommended eliminating the Sustainable Growth Rate (SGR) formula, which ties Medicare physician payments to changes in the gross domestic product (GDP). The Congressional Budget Office (CBO) recently estimated the price of eliminating the SGR at $138 billion over 10 years, which the commission said could be paid for by reducing the overutilization of Medicare services and cutting down on fraud.
The commission, which was convened by the Society of General Internal Medicine last March, is chaired by Dr. Steven A. Schroeder, former president of the Robert Wood Johnson Foundation.
The other members include physicians from various specialties, as well as experts in health care policy. The commission is funded in part by the Robert Wood Johnson Foundation and the California HealthCare Foundation.
Some of the commission’s other recommendations include:
• Increasing payments for evaluation and management codes, while freezing procedural diagnosis codes for 3 years.
• Eliminating higher payments for facility-based services that can be performed in lower-cost settings of care.
• Incorporating quality metrics into fee-for-service contracts.
• Using fixed payment models in areas such as the management of multiple chronic diseases and in-hospital procedures and follow-up.
• Changing the membership of the Relative Value Scale Update Committee (RUC) to make it more representative of the medical profession.
The fee-for-service payment system has contributed to high health care costs and inconsistent quality of care and should be replaced with a blended payment model that includes fixed payments, according to a new report released by a panel of physicians and health care experts.
In a recently published report, the National Commission on Physician Payment Reform recommended phasing out the current fee-for-service system over 5 years in favor of bundled payments, capitation, and increased financial risk-sharing.
"We can’t control runaway medical spending without changing how doctors get paid," Dr. Bill Frist, honorary chair of the commission and former Senate majority leader, said in a statement.
"This is a bipartisan issue. We all want to get the most from our health care dollars and that requires rethinking the way we pay for health care."
But the 14-member commission predicted that fee-for-service would continue to play a large role. By the end of the decade, they called for a blended system of fee-for-service, fixed payments, and salary.
The commission also recommended eliminating the Sustainable Growth Rate (SGR) formula, which ties Medicare physician payments to changes in the gross domestic product (GDP). The Congressional Budget Office (CBO) recently estimated the price of eliminating the SGR at $138 billion over 10 years, which the commission said could be paid for by reducing the overutilization of Medicare services and cutting down on fraud.
The commission, which was convened by the Society of General Internal Medicine last March, is chaired by Dr. Steven A. Schroeder, former president of the Robert Wood Johnson Foundation.
The other members include physicians from various specialties, as well as experts in health care policy. The commission is funded in part by the Robert Wood Johnson Foundation and the California HealthCare Foundation.
Some of the commission’s other recommendations include:
• Increasing payments for evaluation and management codes, while freezing procedural diagnosis codes for 3 years.
• Eliminating higher payments for facility-based services that can be performed in lower-cost settings of care.
• Incorporating quality metrics into fee-for-service contracts.
• Using fixed payment models in areas such as the management of multiple chronic diseases and in-hospital procedures and follow-up.
• Changing the membership of the Relative Value Scale Update Committee (RUC) to make it more representative of the medical profession.
The fee-for-service payment system has contributed to high health care costs and inconsistent quality of care and should be replaced with a blended payment model that includes fixed payments, according to a new report released by a panel of physicians and health care experts.
In a recently published report, the National Commission on Physician Payment Reform recommended phasing out the current fee-for-service system over 5 years in favor of bundled payments, capitation, and increased financial risk-sharing.
"We can’t control runaway medical spending without changing how doctors get paid," Dr. Bill Frist, honorary chair of the commission and former Senate majority leader, said in a statement.
"This is a bipartisan issue. We all want to get the most from our health care dollars and that requires rethinking the way we pay for health care."
But the 14-member commission predicted that fee-for-service would continue to play a large role. By the end of the decade, they called for a blended system of fee-for-service, fixed payments, and salary.
The commission also recommended eliminating the Sustainable Growth Rate (SGR) formula, which ties Medicare physician payments to changes in the gross domestic product (GDP). The Congressional Budget Office (CBO) recently estimated the price of eliminating the SGR at $138 billion over 10 years, which the commission said could be paid for by reducing the overutilization of Medicare services and cutting down on fraud.
The commission, which was convened by the Society of General Internal Medicine last March, is chaired by Dr. Steven A. Schroeder, former president of the Robert Wood Johnson Foundation.
The other members include physicians from various specialties, as well as experts in health care policy. The commission is funded in part by the Robert Wood Johnson Foundation and the California HealthCare Foundation.
Some of the commission’s other recommendations include:
• Increasing payments for evaluation and management codes, while freezing procedural diagnosis codes for 3 years.
• Eliminating higher payments for facility-based services that can be performed in lower-cost settings of care.
• Incorporating quality metrics into fee-for-service contracts.
• Using fixed payment models in areas such as the management of multiple chronic diseases and in-hospital procedures and follow-up.
• Changing the membership of the Relative Value Scale Update Committee (RUC) to make it more representative of the medical profession.
Progress, obstacles cited in building STEMI networks
SNOWMASS, COLO. – Competition among hospitals and between cardiology groups constitutes the greatest barrier to well-functioning regional networks for ST-elevation myocardial infarction therapy, according to Dr. Bernard J. Gersh.
"We’ve got the resources in this country, but we are competitive. That’s the name of the game. So this is a real challenge," said Dr. Gersh, professor of medicine at the Mayo Clinic, Rochester, Minn.
The 2013 ACC/American Heart Association STEMI (ST-elevation myocardial infarction) guidelines list as a class I recommendation that "each community should develop a STEMI system of care."
But a one-size-fits-all approach won’t work. Creating an efficient network to deliver reperfusion therapy to as many STEMI patients as quickly as possible in Los Angeles, where there is seemingly a percutaneous coronary intervention (PCI) center every few blocks, poses a very different set of challenges than in, say, Wyoming, with two cardiac catheterization laboratories to serve nearly a 100,000-quare-mile area, he said.
He recalled a recent conversation with a colleague from a midsize Eastern city with four PCI hospitals. All four run three call schedules per 24 hours so an interventional cardiologist is always available. But collectively the hospitals handle an average of only five or six STEMIs per week.
"Can you really justify that? Furthermore, if you look at all the epidemiology coming out of the U.S. and the Western World, STEMI is in decline. Only about 30% of MIs now are STEMIs, and it’s going to be less and less," Dr. Gersh continued.
Dr. Gersh noted that the American Heart Association Mission: Lifeline program, which was created to increase timely access to PCI for STEMI patients, recently published the first-ever national survey of regional STEMI systems. The purpose was to identify best practices, financing strategies, and barriers to system implementation. Responses were obtained from 381 STEMI networks with 899 PCI hospitals.
The single most commonly cited barrier to network implementation and optimal functioning was hospital competition, identified as a significant problem in 37% of the systems. Next came emergency medical services (EMS) transport and finances, cited by 26% of respondents. The third most common barrier was competition between cardiology groups, which was an issue in 21% of networks.
The predominant funding sources for STEMI systems were PCI hospitals and cardiology practices.
Based on his favorable personal experience with the Mayo Clinic STEMI network, which uses three helicopters, an airplane, and ground ambulances to serve 28 hospitals as far as 150 miles away, Dr. Gersh said it’s clear from the national survey results that most STEMI systems around the country are doing a lot of the important things right.
For example, 92% of systems activate the cath lab with a single phone call, 97% of PCI hospitals accept a STEMI patient 24/7 regardless of bed availability, and 84% of programs operate a data registry with continuous audit.
Two-thirds of STEMI systems have the capability to transmit ECGs from at least some of their ambulances (Circ. Cardiovasc. Qual. Outcomes 2012;5:423-8).
In 87% of the networks nationwide, an emergency department physician can activate the cath lab without cardiology consultation. However, the Mayo Clinic network takes a different approach: Transferred patients bypass the emergency department and are taken straight to the coronary care unit or cath lab.
In an editorial accompanying the report, Dr. Timothy D. Henry, who in 2002 helped organize the nation’s first regional STEMI system at the Minneapolis Heart Institute, said: "Public policy changes to provide financial incentives for more rational use of resources to support regional STEMI systems rather than building more catheterization laboratories would ... be helpful" (Circulation 2012;126:166-8).
Dr. Gersh reported that he serves as a consultant to a number of device and pharmaceutical companies.
SNOWMASS, COLO. – Competition among hospitals and between cardiology groups constitutes the greatest barrier to well-functioning regional networks for ST-elevation myocardial infarction therapy, according to Dr. Bernard J. Gersh.
"We’ve got the resources in this country, but we are competitive. That’s the name of the game. So this is a real challenge," said Dr. Gersh, professor of medicine at the Mayo Clinic, Rochester, Minn.
The 2013 ACC/American Heart Association STEMI (ST-elevation myocardial infarction) guidelines list as a class I recommendation that "each community should develop a STEMI system of care."
But a one-size-fits-all approach won’t work. Creating an efficient network to deliver reperfusion therapy to as many STEMI patients as quickly as possible in Los Angeles, where there is seemingly a percutaneous coronary intervention (PCI) center every few blocks, poses a very different set of challenges than in, say, Wyoming, with two cardiac catheterization laboratories to serve nearly a 100,000-quare-mile area, he said.
He recalled a recent conversation with a colleague from a midsize Eastern city with four PCI hospitals. All four run three call schedules per 24 hours so an interventional cardiologist is always available. But collectively the hospitals handle an average of only five or six STEMIs per week.
"Can you really justify that? Furthermore, if you look at all the epidemiology coming out of the U.S. and the Western World, STEMI is in decline. Only about 30% of MIs now are STEMIs, and it’s going to be less and less," Dr. Gersh continued.
Dr. Gersh noted that the American Heart Association Mission: Lifeline program, which was created to increase timely access to PCI for STEMI patients, recently published the first-ever national survey of regional STEMI systems. The purpose was to identify best practices, financing strategies, and barriers to system implementation. Responses were obtained from 381 STEMI networks with 899 PCI hospitals.
The single most commonly cited barrier to network implementation and optimal functioning was hospital competition, identified as a significant problem in 37% of the systems. Next came emergency medical services (EMS) transport and finances, cited by 26% of respondents. The third most common barrier was competition between cardiology groups, which was an issue in 21% of networks.
The predominant funding sources for STEMI systems were PCI hospitals and cardiology practices.
Based on his favorable personal experience with the Mayo Clinic STEMI network, which uses three helicopters, an airplane, and ground ambulances to serve 28 hospitals as far as 150 miles away, Dr. Gersh said it’s clear from the national survey results that most STEMI systems around the country are doing a lot of the important things right.
For example, 92% of systems activate the cath lab with a single phone call, 97% of PCI hospitals accept a STEMI patient 24/7 regardless of bed availability, and 84% of programs operate a data registry with continuous audit.
Two-thirds of STEMI systems have the capability to transmit ECGs from at least some of their ambulances (Circ. Cardiovasc. Qual. Outcomes 2012;5:423-8).
In 87% of the networks nationwide, an emergency department physician can activate the cath lab without cardiology consultation. However, the Mayo Clinic network takes a different approach: Transferred patients bypass the emergency department and are taken straight to the coronary care unit or cath lab.
In an editorial accompanying the report, Dr. Timothy D. Henry, who in 2002 helped organize the nation’s first regional STEMI system at the Minneapolis Heart Institute, said: "Public policy changes to provide financial incentives for more rational use of resources to support regional STEMI systems rather than building more catheterization laboratories would ... be helpful" (Circulation 2012;126:166-8).
Dr. Gersh reported that he serves as a consultant to a number of device and pharmaceutical companies.
SNOWMASS, COLO. – Competition among hospitals and between cardiology groups constitutes the greatest barrier to well-functioning regional networks for ST-elevation myocardial infarction therapy, according to Dr. Bernard J. Gersh.
"We’ve got the resources in this country, but we are competitive. That’s the name of the game. So this is a real challenge," said Dr. Gersh, professor of medicine at the Mayo Clinic, Rochester, Minn.
The 2013 ACC/American Heart Association STEMI (ST-elevation myocardial infarction) guidelines list as a class I recommendation that "each community should develop a STEMI system of care."
But a one-size-fits-all approach won’t work. Creating an efficient network to deliver reperfusion therapy to as many STEMI patients as quickly as possible in Los Angeles, where there is seemingly a percutaneous coronary intervention (PCI) center every few blocks, poses a very different set of challenges than in, say, Wyoming, with two cardiac catheterization laboratories to serve nearly a 100,000-quare-mile area, he said.
He recalled a recent conversation with a colleague from a midsize Eastern city with four PCI hospitals. All four run three call schedules per 24 hours so an interventional cardiologist is always available. But collectively the hospitals handle an average of only five or six STEMIs per week.
"Can you really justify that? Furthermore, if you look at all the epidemiology coming out of the U.S. and the Western World, STEMI is in decline. Only about 30% of MIs now are STEMIs, and it’s going to be less and less," Dr. Gersh continued.
Dr. Gersh noted that the American Heart Association Mission: Lifeline program, which was created to increase timely access to PCI for STEMI patients, recently published the first-ever national survey of regional STEMI systems. The purpose was to identify best practices, financing strategies, and barriers to system implementation. Responses were obtained from 381 STEMI networks with 899 PCI hospitals.
The single most commonly cited barrier to network implementation and optimal functioning was hospital competition, identified as a significant problem in 37% of the systems. Next came emergency medical services (EMS) transport and finances, cited by 26% of respondents. The third most common barrier was competition between cardiology groups, which was an issue in 21% of networks.
The predominant funding sources for STEMI systems were PCI hospitals and cardiology practices.
Based on his favorable personal experience with the Mayo Clinic STEMI network, which uses three helicopters, an airplane, and ground ambulances to serve 28 hospitals as far as 150 miles away, Dr. Gersh said it’s clear from the national survey results that most STEMI systems around the country are doing a lot of the important things right.
For example, 92% of systems activate the cath lab with a single phone call, 97% of PCI hospitals accept a STEMI patient 24/7 regardless of bed availability, and 84% of programs operate a data registry with continuous audit.
Two-thirds of STEMI systems have the capability to transmit ECGs from at least some of their ambulances (Circ. Cardiovasc. Qual. Outcomes 2012;5:423-8).
In 87% of the networks nationwide, an emergency department physician can activate the cath lab without cardiology consultation. However, the Mayo Clinic network takes a different approach: Transferred patients bypass the emergency department and are taken straight to the coronary care unit or cath lab.
In an editorial accompanying the report, Dr. Timothy D. Henry, who in 2002 helped organize the nation’s first regional STEMI system at the Minneapolis Heart Institute, said: "Public policy changes to provide financial incentives for more rational use of resources to support regional STEMI systems rather than building more catheterization laboratories would ... be helpful" (Circulation 2012;126:166-8).
Dr. Gersh reported that he serves as a consultant to a number of device and pharmaceutical companies.
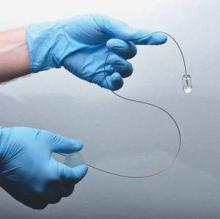
Laser pill shows potential benefits over upper GI endoscopy
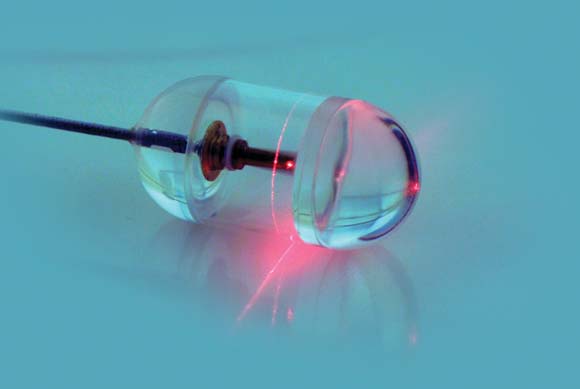
A small, swallowed, laser imaging capsule provides full-thickness imaging of the upper gastro- intestinal tract without biopsy, and is quicker and less invasive than traditional endoscopy, according to the Harvard University research-ers who are developing it.
About the size of a large multivitamin pill, the transparent capsule generates a near-infrared beam that spins rapidly about its circumference during transit. Changes in the reflected light allow cross-sectional imaging of the esophagus in a few minutes. Sequential cross-sections can be compiled into three-dimensional models of the entire lumen (Nat. Med. 2013 Jan. 13 [doi: 10.1038/nm.3052]).
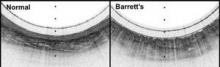
"This system gives us a convenient way to screen for Barrett?s [esophagus] that doesn?t require patient sedation, a specialized setting and equipment, or a physician who has been trained in endoscopy. By showing the three-dimensional, microscopic structure of the esophageal lining, it reveals much more detail than can be seen with even high-resolution endoscopy. The images produced have been some of the best we have seen of the esophagus," investigator Dr. Guillermo Tearney, a Harvard Medical School pathology professor and the associate director of the Wellman Center for Photomedicine at Massachusetts General Hospital, Boston, said in a statement.
The capsule is on a tether, which carries its fiber optic line and laser driveshaft and helps with positioning. The capsule is pulled up and out after use, and disinfected for the next patient.
In early testing, 15 cm of esophagus in seven healthy and six Barrett?s esophagus patients was imaged in a mean of 58 seconds; it took about 6 minutes to make two down- and two up-transits. The technique, dubbed tethered capsule endomicroscopy, clearly distinguished the cellular abnormalities of Barrett?s. Standard upper GI endoscopy takes about 90 minutes.
"We originally were concerned that we might miss a lot of data because of the small size of the capsule, but we were surprised to find that, once the pill has been swallowed, it is firmly grasped by the esophagus, allowing complete microscopic imaging of the entire wall," Dr. Tearney said.
There were no complications, and 12 of the 13 subjects said they preferred the capsule to previous endoscopies.
"Because the tethered endomicroscopy pill traverses the gastrointestinal tract without visual guidance, the training required to conduct the procedure is minimal. This fact, combined with the brevity and ease with which the procedure is performed, will enable internal microscopic imaging in almost any health care setting, including in the office of the primary care physician," Dr. Tearney and his colleagues wrote in their paper.
The research was supported by grants from the National Institutes of Health. The researchers said they had no disclosures.
A small, swallowed, laser imaging capsule provides full-thickness imaging of the upper gastro- intestinal tract without biopsy, and is quicker and less invasive than traditional endoscopy, according to the Harvard University research-ers who are developing it.
About the size of a large multivitamin pill, the transparent capsule generates a near-infrared beam that spins rapidly about its circumference during transit. Changes in the reflected light allow cross-sectional imaging of the esophagus in a few minutes. Sequential cross-sections can be compiled into three-dimensional models of the entire lumen (Nat. Med. 2013 Jan. 13 [doi: 10.1038/nm.3052]).
"This system gives us a convenient way to screen for Barrett?s [esophagus] that doesn?t require patient sedation, a specialized setting and equipment, or a physician who has been trained in endoscopy. By showing the three-dimensional, microscopic structure of the esophageal lining, it reveals much more detail than can be seen with even high-resolution endoscopy. The images produced have been some of the best we have seen of the esophagus," investigator Dr. Guillermo Tearney, a Harvard Medical School pathology professor and the associate director of the Wellman Center for Photomedicine at Massachusetts General Hospital, Boston, said in a statement.
The capsule is on a tether, which carries its fiber optic line and laser driveshaft and helps with positioning. The capsule is pulled up and out after use, and disinfected for the next patient.
In early testing, 15 cm of esophagus in seven healthy and six Barrett?s esophagus patients was imaged in a mean of 58 seconds; it took about 6 minutes to make two down- and two up-transits. The technique, dubbed tethered capsule endomicroscopy, clearly distinguished the cellular abnormalities of Barrett?s. Standard upper GI endoscopy takes about 90 minutes.
"We originally were concerned that we might miss a lot of data because of the small size of the capsule, but we were surprised to find that, once the pill has been swallowed, it is firmly grasped by the esophagus, allowing complete microscopic imaging of the entire wall," Dr. Tearney said.
There were no complications, and 12 of the 13 subjects said they preferred the capsule to previous endoscopies.
"Because the tethered endomicroscopy pill traverses the gastrointestinal tract without visual guidance, the training required to conduct the procedure is minimal. This fact, combined with the brevity and ease with which the procedure is performed, will enable internal microscopic imaging in almost any health care setting, including in the office of the primary care physician," Dr. Tearney and his colleagues wrote in their paper.
The research was supported by grants from the National Institutes of Health. The researchers said they had no disclosures.
A small, swallowed, laser imaging capsule provides full-thickness imaging of the upper gastro- intestinal tract without biopsy, and is quicker and less invasive than traditional endoscopy, according to the Harvard University research-ers who are developing it.
About the size of a large multivitamin pill, the transparent capsule generates a near-infrared beam that spins rapidly about its circumference during transit. Changes in the reflected light allow cross-sectional imaging of the esophagus in a few minutes. Sequential cross-sections can be compiled into three-dimensional models of the entire lumen (Nat. Med. 2013 Jan. 13 [doi: 10.1038/nm.3052]).
"This system gives us a convenient way to screen for Barrett?s [esophagus] that doesn?t require patient sedation, a specialized setting and equipment, or a physician who has been trained in endoscopy. By showing the three-dimensional, microscopic structure of the esophageal lining, it reveals much more detail than can be seen with even high-resolution endoscopy. The images produced have been some of the best we have seen of the esophagus," investigator Dr. Guillermo Tearney, a Harvard Medical School pathology professor and the associate director of the Wellman Center for Photomedicine at Massachusetts General Hospital, Boston, said in a statement.
The capsule is on a tether, which carries its fiber optic line and laser driveshaft and helps with positioning. The capsule is pulled up and out after use, and disinfected for the next patient.
In early testing, 15 cm of esophagus in seven healthy and six Barrett?s esophagus patients was imaged in a mean of 58 seconds; it took about 6 minutes to make two down- and two up-transits. The technique, dubbed tethered capsule endomicroscopy, clearly distinguished the cellular abnormalities of Barrett?s. Standard upper GI endoscopy takes about 90 minutes.
"We originally were concerned that we might miss a lot of data because of the small size of the capsule, but we were surprised to find that, once the pill has been swallowed, it is firmly grasped by the esophagus, allowing complete microscopic imaging of the entire wall," Dr. Tearney said.
There were no complications, and 12 of the 13 subjects said they preferred the capsule to previous endoscopies.
"Because the tethered endomicroscopy pill traverses the gastrointestinal tract without visual guidance, the training required to conduct the procedure is minimal. This fact, combined with the brevity and ease with which the procedure is performed, will enable internal microscopic imaging in almost any health care setting, including in the office of the primary care physician," Dr. Tearney and his colleagues wrote in their paper.
The research was supported by grants from the National Institutes of Health. The researchers said they had no disclosures.
Major Finding: In early testing, 15 cm of esophagus was imaged in a mean of 58 seconds and clearly distinguished the cellular abnormalities of Barrett?s esophagus; it took about 6 minutes to make two down- and two up-transits.
Data Source: A pilot study to image the esophagus in seven healthy patients and six with Barrett?s esophagus.
Disclosures: The research was supported by grants from the National Institutes of Health. The investigators said they had no disclosures.
Study: Early esophageal cancer staging suspect
LOS ANGELES – Clinical staging of T2N0 esophageal cancer remains unreliable, despite advances in staging techniques, a study has shown.
The addition of endoscopic ultrasound and PET/CT has improved the ability to clinically stage esophageal cancer overall, but clinical staging of T2N0 disease has generally been less reliable than staging of more advanced disease. The subset of patients with T2N0 disease, which accounts for only a small portion of esophageal resections, has been a difficult group to study because single-center investigations involve a limited number of patients, Dr. Traves D. Crabtree said at the annual meeting of the Society of Thoracic Surgeons in the prestigious Richard E. Clark Paper for General Thoracic Surgery.
Dr. Crabtree of Washington University, St. Louis, and his colleagues examined the adequacy of clinical staging of T2N0 disease using the Society of Thoracic Surgeons (STS) General Thoracic Surgery Database.
The researchers identified 810 patients clinically staged as T2N0 from 2002 to 2011 and excluded 58 because of inadequate pathologic staging data. Clinical stage, pathologic stage, and preoperative characteristics were recorded for each patient and multivariable analysis was used to identify factors associated with upstaging at the time of surgery.
Among 752 patients with clinically staged T2N0, the investigators found that 482 went directly to surgery. Of these, 27.4% (132) were confirmed as pathologic T2N0. A total of 25.9% (125) were downstaged (T1N0), while 46.7% (225) were upstaged (T3-4N0 or TanyN1-3).
Tumor depth (pT3-4) accounted for 18.2% of upstaging while nodal upstaging occurred in approximately 82%. When logistic regression was used, male sex was associated with upstaging (odds ratio = 1.85, P = .024). By analyzing the part of the database that included tumor grade (between the years 2009 and 2011), the investigators found that a higher histologic grade was significantly associated with upstaging (P = .004).
"Over one-third of surgeons have opted to treat T2N0 disease with induction therapy, despite the fact that one-quarter of these patients will be pT1N0," he said.
"This is the first large-scale multi-institutional study of clinical T2N0 patients using the STS General Thoracic Surgery Database. These data highlight the inaccuracy associated with clinical staging of T2N0 esophageal cancer and may influence the surgeon’s decision-making process in choosing a treatment regimen for these patients," Dr. Crabtree added in an interview.
"Given the current limitations of clinical staging of T2N0 patients, the incidence of occult nodal disease, and the similar perioperative outcomes among patients treated with and without induction therapy, these patients may more likely be treated with induction therapy in the future. Additional studies are needed to compare long-term outcomes between patients receiving induction therapy, vs. those clinical T2N0 patients going directly to surgery, before a definitive recommendation can be made," he concluded.
Dr. Crabtree reported that he had no relevant financial disclosures.
LOS ANGELES – Clinical staging of T2N0 esophageal cancer remains unreliable, despite advances in staging techniques, a study has shown.
The addition of endoscopic ultrasound and PET/CT has improved the ability to clinically stage esophageal cancer overall, but clinical staging of T2N0 disease has generally been less reliable than staging of more advanced disease. The subset of patients with T2N0 disease, which accounts for only a small portion of esophageal resections, has been a difficult group to study because single-center investigations involve a limited number of patients, Dr. Traves D. Crabtree said at the annual meeting of the Society of Thoracic Surgeons in the prestigious Richard E. Clark Paper for General Thoracic Surgery.
Dr. Crabtree of Washington University, St. Louis, and his colleagues examined the adequacy of clinical staging of T2N0 disease using the Society of Thoracic Surgeons (STS) General Thoracic Surgery Database.
The researchers identified 810 patients clinically staged as T2N0 from 2002 to 2011 and excluded 58 because of inadequate pathologic staging data. Clinical stage, pathologic stage, and preoperative characteristics were recorded for each patient and multivariable analysis was used to identify factors associated with upstaging at the time of surgery.
Among 752 patients with clinically staged T2N0, the investigators found that 482 went directly to surgery. Of these, 27.4% (132) were confirmed as pathologic T2N0. A total of 25.9% (125) were downstaged (T1N0), while 46.7% (225) were upstaged (T3-4N0 or TanyN1-3).
Tumor depth (pT3-4) accounted for 18.2% of upstaging while nodal upstaging occurred in approximately 82%. When logistic regression was used, male sex was associated with upstaging (odds ratio = 1.85, P = .024). By analyzing the part of the database that included tumor grade (between the years 2009 and 2011), the investigators found that a higher histologic grade was significantly associated with upstaging (P = .004).
"Over one-third of surgeons have opted to treat T2N0 disease with induction therapy, despite the fact that one-quarter of these patients will be pT1N0," he said.
"This is the first large-scale multi-institutional study of clinical T2N0 patients using the STS General Thoracic Surgery Database. These data highlight the inaccuracy associated with clinical staging of T2N0 esophageal cancer and may influence the surgeon’s decision-making process in choosing a treatment regimen for these patients," Dr. Crabtree added in an interview.
"Given the current limitations of clinical staging of T2N0 patients, the incidence of occult nodal disease, and the similar perioperative outcomes among patients treated with and without induction therapy, these patients may more likely be treated with induction therapy in the future. Additional studies are needed to compare long-term outcomes between patients receiving induction therapy, vs. those clinical T2N0 patients going directly to surgery, before a definitive recommendation can be made," he concluded.
Dr. Crabtree reported that he had no relevant financial disclosures.
LOS ANGELES – Clinical staging of T2N0 esophageal cancer remains unreliable, despite advances in staging techniques, a study has shown.
The addition of endoscopic ultrasound and PET/CT has improved the ability to clinically stage esophageal cancer overall, but clinical staging of T2N0 disease has generally been less reliable than staging of more advanced disease. The subset of patients with T2N0 disease, which accounts for only a small portion of esophageal resections, has been a difficult group to study because single-center investigations involve a limited number of patients, Dr. Traves D. Crabtree said at the annual meeting of the Society of Thoracic Surgeons in the prestigious Richard E. Clark Paper for General Thoracic Surgery.
Dr. Crabtree of Washington University, St. Louis, and his colleagues examined the adequacy of clinical staging of T2N0 disease using the Society of Thoracic Surgeons (STS) General Thoracic Surgery Database.
The researchers identified 810 patients clinically staged as T2N0 from 2002 to 2011 and excluded 58 because of inadequate pathologic staging data. Clinical stage, pathologic stage, and preoperative characteristics were recorded for each patient and multivariable analysis was used to identify factors associated with upstaging at the time of surgery.
Among 752 patients with clinically staged T2N0, the investigators found that 482 went directly to surgery. Of these, 27.4% (132) were confirmed as pathologic T2N0. A total of 25.9% (125) were downstaged (T1N0), while 46.7% (225) were upstaged (T3-4N0 or TanyN1-3).
Tumor depth (pT3-4) accounted for 18.2% of upstaging while nodal upstaging occurred in approximately 82%. When logistic regression was used, male sex was associated with upstaging (odds ratio = 1.85, P = .024). By analyzing the part of the database that included tumor grade (between the years 2009 and 2011), the investigators found that a higher histologic grade was significantly associated with upstaging (P = .004).
"Over one-third of surgeons have opted to treat T2N0 disease with induction therapy, despite the fact that one-quarter of these patients will be pT1N0," he said.
"This is the first large-scale multi-institutional study of clinical T2N0 patients using the STS General Thoracic Surgery Database. These data highlight the inaccuracy associated with clinical staging of T2N0 esophageal cancer and may influence the surgeon’s decision-making process in choosing a treatment regimen for these patients," Dr. Crabtree added in an interview.
"Given the current limitations of clinical staging of T2N0 patients, the incidence of occult nodal disease, and the similar perioperative outcomes among patients treated with and without induction therapy, these patients may more likely be treated with induction therapy in the future. Additional studies are needed to compare long-term outcomes between patients receiving induction therapy, vs. those clinical T2N0 patients going directly to surgery, before a definitive recommendation can be made," he concluded.
Dr. Crabtree reported that he had no relevant financial disclosures.
AT THE ANNUAL MEETING OF THE SOCIETY OF THORACIC SURGEONS
Major Finding: Of 482 patients who went directly to surgery, 26% were downstaged, while 47% were upstaged.
Data Source: A retrospective, database analysis of 810 patients clinically staged as T2N0 from 2002 to 2011.
Disclosures: Dr. Crabtree reported that he had no relevant financial disclosures.
CMS rules on reporting physician COI
Federal health officials have finally released the details on how online public reporting of industry payments to physicians will work.
Under the final rule released by the Centers for Medicare and Medicaid Services (CMS), drug, device, and medical supply manufacturers who participate in Medicare, Medicaid, or the Children’s Health Insurance Program will be required to submit annual reports to the federal government on any payments of $10 or more that they made to physicians and teaching hospitals. They also will be required to report on all payments if the payments and transfers of value to a single physician reach $100 in aggregate value for a year.
Manufacturers and group purchasing organizations (GPOs) must also report on physician ownership and investment interests each year. CMS will post the information on a public website. The requirements are mandated under the Affordable Care Act (ACA).
"You should know when your doctor has a financial relationship with the companies that manufacture or supply the medicines or medical devices you may need," Dr. Peter Budetti, CMS deputy administrator for Program Integrity, said in a statement. "Disclosure of these relationships allows patients to have more informed discussions with their doctors."
Manufacturers and GPOs have until Aug. 1 to begin collecting data. They must submit their reports on payments made in 2013 by March 31, 2014. CMS will post the data online by Sept. 30, 2014.
CMS did not meet the deadline set by law for issuing this final regulation: Under the ACA, data collection was supposed to begin in January 2012.
The final rule contains plenty of exceptions, however. For instance, reporting is not required for gifts between individuals with an existing personal relationship. Other exclusions include small payments of less than $10, educational materials that directly benefit patients or are intended for patient use, discounts for rebates for drugs and devices, in-kind items for charity care, and samples.
Indirect payments made to speakers at accredited or certified continuing medical education (CME) events also do not need to be reported as long as the manufacturer doesn’t suggest speakers.
The final rule also clarifies that companies sponsoring large-scale conferences do not need to track and report on small gifts and food items worth less than $10 such as pens and bottles of water. These items also won’t count toward the minimum yearly reporting threshold of $100, according to CMS.
"I think this will make life easier, because it will contribute toward a more relaxed atmosphere at meetings so that attendees won’t have to worry every time they pick up a bottle of water or a granola bar," said Dr. Daniel Carlat, project director for the Pew Prescription Project, which works for greater transparency in physician-industry relationships.
Dr. Carlat said the final rule strikes the right balance between increasing payment transparency and not overburdening physicians with the requirements.
Although the data collection and reporting requirements are on the drug and device industry, physicians are responsible for reviewing their information before publication. Under the final rule, physicians will have 45 days to review the reports and another 15 days to work with the manufacturers to correct any disputed reports. After that, if there are still disputes, the information will be posted publicly but will include a disclaimer that it is disputed, according to the final rule.
The new transparency initiative will likely enhance the public’s trust and confidence in their physicians, Dr. Carlat said. Consumers often hear about the worst-case scenarios, where physicians are taking millions of dollars that may cause conflicts of interest, but the new reporting is likely to show that is rare, he said.
"I think we’ll find with these transparency reports that the vast majority of payments and gifts are of very low value and are the equivalent of $50 to $100 or a few hundred dollars a year," Dr. Carlat said. "I think when patients see these figures, their concerns about relationships between doctors and companies will be to some extent allayed."
Federal health officials have finally released the details on how online public reporting of industry payments to physicians will work.
Under the final rule released by the Centers for Medicare and Medicaid Services (CMS), drug, device, and medical supply manufacturers who participate in Medicare, Medicaid, or the Children’s Health Insurance Program will be required to submit annual reports to the federal government on any payments of $10 or more that they made to physicians and teaching hospitals. They also will be required to report on all payments if the payments and transfers of value to a single physician reach $100 in aggregate value for a year.
Manufacturers and group purchasing organizations (GPOs) must also report on physician ownership and investment interests each year. CMS will post the information on a public website. The requirements are mandated under the Affordable Care Act (ACA).
"You should know when your doctor has a financial relationship with the companies that manufacture or supply the medicines or medical devices you may need," Dr. Peter Budetti, CMS deputy administrator for Program Integrity, said in a statement. "Disclosure of these relationships allows patients to have more informed discussions with their doctors."
Manufacturers and GPOs have until Aug. 1 to begin collecting data. They must submit their reports on payments made in 2013 by March 31, 2014. CMS will post the data online by Sept. 30, 2014.
CMS did not meet the deadline set by law for issuing this final regulation: Under the ACA, data collection was supposed to begin in January 2012.
The final rule contains plenty of exceptions, however. For instance, reporting is not required for gifts between individuals with an existing personal relationship. Other exclusions include small payments of less than $10, educational materials that directly benefit patients or are intended for patient use, discounts for rebates for drugs and devices, in-kind items for charity care, and samples.
Indirect payments made to speakers at accredited or certified continuing medical education (CME) events also do not need to be reported as long as the manufacturer doesn’t suggest speakers.
The final rule also clarifies that companies sponsoring large-scale conferences do not need to track and report on small gifts and food items worth less than $10 such as pens and bottles of water. These items also won’t count toward the minimum yearly reporting threshold of $100, according to CMS.
"I think this will make life easier, because it will contribute toward a more relaxed atmosphere at meetings so that attendees won’t have to worry every time they pick up a bottle of water or a granola bar," said Dr. Daniel Carlat, project director for the Pew Prescription Project, which works for greater transparency in physician-industry relationships.
Dr. Carlat said the final rule strikes the right balance between increasing payment transparency and not overburdening physicians with the requirements.
Although the data collection and reporting requirements are on the drug and device industry, physicians are responsible for reviewing their information before publication. Under the final rule, physicians will have 45 days to review the reports and another 15 days to work with the manufacturers to correct any disputed reports. After that, if there are still disputes, the information will be posted publicly but will include a disclaimer that it is disputed, according to the final rule.
The new transparency initiative will likely enhance the public’s trust and confidence in their physicians, Dr. Carlat said. Consumers often hear about the worst-case scenarios, where physicians are taking millions of dollars that may cause conflicts of interest, but the new reporting is likely to show that is rare, he said.
"I think we’ll find with these transparency reports that the vast majority of payments and gifts are of very low value and are the equivalent of $50 to $100 or a few hundred dollars a year," Dr. Carlat said. "I think when patients see these figures, their concerns about relationships between doctors and companies will be to some extent allayed."
Federal health officials have finally released the details on how online public reporting of industry payments to physicians will work.
Under the final rule released by the Centers for Medicare and Medicaid Services (CMS), drug, device, and medical supply manufacturers who participate in Medicare, Medicaid, or the Children’s Health Insurance Program will be required to submit annual reports to the federal government on any payments of $10 or more that they made to physicians and teaching hospitals. They also will be required to report on all payments if the payments and transfers of value to a single physician reach $100 in aggregate value for a year.
Manufacturers and group purchasing organizations (GPOs) must also report on physician ownership and investment interests each year. CMS will post the information on a public website. The requirements are mandated under the Affordable Care Act (ACA).
"You should know when your doctor has a financial relationship with the companies that manufacture or supply the medicines or medical devices you may need," Dr. Peter Budetti, CMS deputy administrator for Program Integrity, said in a statement. "Disclosure of these relationships allows patients to have more informed discussions with their doctors."
Manufacturers and GPOs have until Aug. 1 to begin collecting data. They must submit their reports on payments made in 2013 by March 31, 2014. CMS will post the data online by Sept. 30, 2014.
CMS did not meet the deadline set by law for issuing this final regulation: Under the ACA, data collection was supposed to begin in January 2012.
The final rule contains plenty of exceptions, however. For instance, reporting is not required for gifts between individuals with an existing personal relationship. Other exclusions include small payments of less than $10, educational materials that directly benefit patients or are intended for patient use, discounts for rebates for drugs and devices, in-kind items for charity care, and samples.
Indirect payments made to speakers at accredited or certified continuing medical education (CME) events also do not need to be reported as long as the manufacturer doesn’t suggest speakers.
The final rule also clarifies that companies sponsoring large-scale conferences do not need to track and report on small gifts and food items worth less than $10 such as pens and bottles of water. These items also won’t count toward the minimum yearly reporting threshold of $100, according to CMS.
"I think this will make life easier, because it will contribute toward a more relaxed atmosphere at meetings so that attendees won’t have to worry every time they pick up a bottle of water or a granola bar," said Dr. Daniel Carlat, project director for the Pew Prescription Project, which works for greater transparency in physician-industry relationships.
Dr. Carlat said the final rule strikes the right balance between increasing payment transparency and not overburdening physicians with the requirements.
Although the data collection and reporting requirements are on the drug and device industry, physicians are responsible for reviewing their information before publication. Under the final rule, physicians will have 45 days to review the reports and another 15 days to work with the manufacturers to correct any disputed reports. After that, if there are still disputes, the information will be posted publicly but will include a disclaimer that it is disputed, according to the final rule.
The new transparency initiative will likely enhance the public’s trust and confidence in their physicians, Dr. Carlat said. Consumers often hear about the worst-case scenarios, where physicians are taking millions of dollars that may cause conflicts of interest, but the new reporting is likely to show that is rare, he said.
"I think we’ll find with these transparency reports that the vast majority of payments and gifts are of very low value and are the equivalent of $50 to $100 or a few hundred dollars a year," Dr. Carlat said. "I think when patients see these figures, their concerns about relationships between doctors and companies will be to some extent allayed."
Beta-blocker use fails as a CABG quality metric
LOS ANGELES – Single-institution reports regarding the benefits of beta-blocker use are conflicting, despite the fact that preoperative beta-blockade for coronary artery bypass grafting has become an accepted hospital quality metric, according to Dr. Damien J. LaPar and his colleagues.
Dr. LaPar of the University of Virginia, Charlottesville reported the results of research undertaken to assess this issue based on a study of patient records from a statewide, multi-institutional Society of Thoracic Surgeons (STS)–certified database for isolated coronary artery bypass grafting (CABG) operations (2001-2011). He and his colleagues found that there was no difference seen in mortality, length of stay, or readmission comparing patients with or without preoperative beta-blocker use.
Their prestigious Richard E. Clark Paper for Adult Cardiac Surgery, presented at the annual meeting of the Society of Thoracic Surgeons, utilized the STS Adult Cardiac Surgery Database to demonstrate that the perceived benefits of using beta-blockers before CABG do not stand up as statistically significant improvements in outcomes.
"Preoperative beta-blocker use is not associated with improved patient outcomes or hospital resource utilization following" CABG, said Dr. LaPar.
Patients were stratified by preoperative beta-blocker use and the influence of preoperative beta-blockers on risk-adjusted outcomes was assessed by hierarchical regression modeling with adjustment for preoperative risk using calculated STS predictive risk indices.
A total of 43,747 patients with a mean age of around 64 years were included in the study; 80.2% of these patients were treated with beta-blockers. The median STS-predicted risk of mortality scores for beta-blocker patients were incrementally lower, compared with non–beta blocker patients (1.2% vs. 1.4%, P less than .001). Non–beta blocker patients more frequently developed pneumonia (3.5% vs. 2.8%, P = .001), while beta-blocker patients had surprisingly greater intraoperative blood usage (16.0% vs. 11.2%, P less than .001).
There was, however, no difference in unadjusted mortality (beta-blocker, 1.9%, vs. non beta-blocker, 2.2%; P = 0.15). After risk adjustment, preoperative beta-blocker use was not associated with mortality (P = .63), morbidity, length of stay (P = .79), or hospital readmission (P = .97).
"These data suggest that the use of preoperative beta-blockers for [CABG] operations should not be used as a measure of surgical quality," Dr. LaPar said.
"In an era of increasing pressure on individual hospital and surgical outcomes, the identification of appropriate measures of surgical quality remains critical," Dr. LaPar added in an interview.
"More importantly, as public reporting of surgeon outcomes becomes more common, the cardiothoracic surgical community must play a central role in providing updated data from which to base health care policy, hospital and surgeon reimbursement strategies, and referral patterns for cardiac surgical patients," he stated.
"The results of our study provide an updated reexamination of an issue of increasing debate and provide current clinical estimates on the adjusted impact of preoperative beta-blocker use on outcomes following isolated CABG operations. Future randomized controlled trials are needed to more clearly define a cause-effect relationship between preoperative beta-blocker therapy and coronary artery bypass grafting outcomes before the routine use of beta-blockade should be adopted as a quality performance measure by the cardiothoracic surgical community," Dr. LaPar concluded.
Dr. LaPar reported having no relevant conflicts of interest with regard to this paper; two of his colleagues reported serving as speakers or receiving funding from a variety of drug and device companies.
LOS ANGELES – Single-institution reports regarding the benefits of beta-blocker use are conflicting, despite the fact that preoperative beta-blockade for coronary artery bypass grafting has become an accepted hospital quality metric, according to Dr. Damien J. LaPar and his colleagues.
Dr. LaPar of the University of Virginia, Charlottesville reported the results of research undertaken to assess this issue based on a study of patient records from a statewide, multi-institutional Society of Thoracic Surgeons (STS)–certified database for isolated coronary artery bypass grafting (CABG) operations (2001-2011). He and his colleagues found that there was no difference seen in mortality, length of stay, or readmission comparing patients with or without preoperative beta-blocker use.
Their prestigious Richard E. Clark Paper for Adult Cardiac Surgery, presented at the annual meeting of the Society of Thoracic Surgeons, utilized the STS Adult Cardiac Surgery Database to demonstrate that the perceived benefits of using beta-blockers before CABG do not stand up as statistically significant improvements in outcomes.
"Preoperative beta-blocker use is not associated with improved patient outcomes or hospital resource utilization following" CABG, said Dr. LaPar.
Patients were stratified by preoperative beta-blocker use and the influence of preoperative beta-blockers on risk-adjusted outcomes was assessed by hierarchical regression modeling with adjustment for preoperative risk using calculated STS predictive risk indices.
A total of 43,747 patients with a mean age of around 64 years were included in the study; 80.2% of these patients were treated with beta-blockers. The median STS-predicted risk of mortality scores for beta-blocker patients were incrementally lower, compared with non–beta blocker patients (1.2% vs. 1.4%, P less than .001). Non–beta blocker patients more frequently developed pneumonia (3.5% vs. 2.8%, P = .001), while beta-blocker patients had surprisingly greater intraoperative blood usage (16.0% vs. 11.2%, P less than .001).
There was, however, no difference in unadjusted mortality (beta-blocker, 1.9%, vs. non beta-blocker, 2.2%; P = 0.15). After risk adjustment, preoperative beta-blocker use was not associated with mortality (P = .63), morbidity, length of stay (P = .79), or hospital readmission (P = .97).
"These data suggest that the use of preoperative beta-blockers for [CABG] operations should not be used as a measure of surgical quality," Dr. LaPar said.
"In an era of increasing pressure on individual hospital and surgical outcomes, the identification of appropriate measures of surgical quality remains critical," Dr. LaPar added in an interview.
"More importantly, as public reporting of surgeon outcomes becomes more common, the cardiothoracic surgical community must play a central role in providing updated data from which to base health care policy, hospital and surgeon reimbursement strategies, and referral patterns for cardiac surgical patients," he stated.
"The results of our study provide an updated reexamination of an issue of increasing debate and provide current clinical estimates on the adjusted impact of preoperative beta-blocker use on outcomes following isolated CABG operations. Future randomized controlled trials are needed to more clearly define a cause-effect relationship between preoperative beta-blocker therapy and coronary artery bypass grafting outcomes before the routine use of beta-blockade should be adopted as a quality performance measure by the cardiothoracic surgical community," Dr. LaPar concluded.
Dr. LaPar reported having no relevant conflicts of interest with regard to this paper; two of his colleagues reported serving as speakers or receiving funding from a variety of drug and device companies.
LOS ANGELES – Single-institution reports regarding the benefits of beta-blocker use are conflicting, despite the fact that preoperative beta-blockade for coronary artery bypass grafting has become an accepted hospital quality metric, according to Dr. Damien J. LaPar and his colleagues.
Dr. LaPar of the University of Virginia, Charlottesville reported the results of research undertaken to assess this issue based on a study of patient records from a statewide, multi-institutional Society of Thoracic Surgeons (STS)–certified database for isolated coronary artery bypass grafting (CABG) operations (2001-2011). He and his colleagues found that there was no difference seen in mortality, length of stay, or readmission comparing patients with or without preoperative beta-blocker use.
Their prestigious Richard E. Clark Paper for Adult Cardiac Surgery, presented at the annual meeting of the Society of Thoracic Surgeons, utilized the STS Adult Cardiac Surgery Database to demonstrate that the perceived benefits of using beta-blockers before CABG do not stand up as statistically significant improvements in outcomes.
"Preoperative beta-blocker use is not associated with improved patient outcomes or hospital resource utilization following" CABG, said Dr. LaPar.
Patients were stratified by preoperative beta-blocker use and the influence of preoperative beta-blockers on risk-adjusted outcomes was assessed by hierarchical regression modeling with adjustment for preoperative risk using calculated STS predictive risk indices.
A total of 43,747 patients with a mean age of around 64 years were included in the study; 80.2% of these patients were treated with beta-blockers. The median STS-predicted risk of mortality scores for beta-blocker patients were incrementally lower, compared with non–beta blocker patients (1.2% vs. 1.4%, P less than .001). Non–beta blocker patients more frequently developed pneumonia (3.5% vs. 2.8%, P = .001), while beta-blocker patients had surprisingly greater intraoperative blood usage (16.0% vs. 11.2%, P less than .001).
There was, however, no difference in unadjusted mortality (beta-blocker, 1.9%, vs. non beta-blocker, 2.2%; P = 0.15). After risk adjustment, preoperative beta-blocker use was not associated with mortality (P = .63), morbidity, length of stay (P = .79), or hospital readmission (P = .97).
"These data suggest that the use of preoperative beta-blockers for [CABG] operations should not be used as a measure of surgical quality," Dr. LaPar said.
"In an era of increasing pressure on individual hospital and surgical outcomes, the identification of appropriate measures of surgical quality remains critical," Dr. LaPar added in an interview.
"More importantly, as public reporting of surgeon outcomes becomes more common, the cardiothoracic surgical community must play a central role in providing updated data from which to base health care policy, hospital and surgeon reimbursement strategies, and referral patterns for cardiac surgical patients," he stated.
"The results of our study provide an updated reexamination of an issue of increasing debate and provide current clinical estimates on the adjusted impact of preoperative beta-blocker use on outcomes following isolated CABG operations. Future randomized controlled trials are needed to more clearly define a cause-effect relationship between preoperative beta-blocker therapy and coronary artery bypass grafting outcomes before the routine use of beta-blockade should be adopted as a quality performance measure by the cardiothoracic surgical community," Dr. LaPar concluded.
Dr. LaPar reported having no relevant conflicts of interest with regard to this paper; two of his colleagues reported serving as speakers or receiving funding from a variety of drug and device companies.
AT THE STS ANNUAL MEETING
Major Finding: Preoperative beta-blocker use was not associated with mortality, morbidity, length of stay, or readmission.
Data Source: A retrospective database analysis of 43,747 CABG patients, 80% of whom were treated with beta-blockers.
Disclosures: Dr. LaPar reported having no relevant conflicts of interest; two of his colleagues reported serving as speakers or receiving funding from a variety of drug and device companies.
Neonates at highest risk for Ebstein’s malformation treatment
LOS ANGELES – Neonates in particular are at risk for poor outcomes from Ebstein’s malformation, showing significantly higher mortality than infants, children, and adults, according to a database study presented by Dr. Ryan R. Davies at the annual meeting of the Society of Thoracic Surgeons.
Ebstein’s malformation is a rare congenital cardiac anomaly. Surgery for Ebstein’s involves a range of procedures, and with low institutional volumes, the only available data on treatment have been limited to individual reports demonstrating highly variable approaches.
Dr. Davies of the Nemours/A.I. duPont Hospital for Children, Wilmington, Del., and his colleagues performed a retrospective study of procedures performed on patients with a primary diagnosis of Ebstein’s malformation (2002-2009) in the STS Congenital Heart Surgery Database.
A total of 595 operations on patients with Ebstein’s were included: 116 on neonates (19%), 122 on infants (21%), 264 on children (44%), and 93 on adults (16%). The authors found that average annual institutional case volumes were low (median, 1 case/year), and procedures varied according to age. Neonates had a high rate of palliative procedures: tricuspid valve (TV) closure (16%) and systemic-to-pulmonary shunts with or without TV closure (37%) and isolated TV closure (8.6%), with Ebstein’s repair or TV valvuloplasty performed in 32%.
Infants usually underwent superior cavopulmonary connections (52%).
Among older patients, procedures were mostly in three categories: TV surgery (children, 55%; adults, 69%), arrhythmia procedures (children, 9%; adults, 17%), and Fontan (children, 16%). In-hospital mortality was higher among neonatal patients (23%) than in infants (4%), children (0.8%), and adults (1.1%).
Among neonates, 36 subsequent procedures were performed during the same hospitalization in 27 patients (23%); including TV closure (11%); shunt (15%); Ebstein’s repair (17%) or TV replacement (15%); and heart transplantation (7.4%). Mortality was similar among neonates who had a second procedure and those who did not (27% vs. 23%, respectively). ECMO (extracorporeal membrane oxygenation) was used in 9% of neonates but in less than 2% of patients in other age groups.
"This study represents a broad overview of the diverse options for surgical treatment of Ebstein’s anomaly. It shows the challenges faced in caring for extremely ill neonatal patients. We have also shown that repair of Ebstein’s anomaly is performed infrequently at most centers, limiting the ability of individual series to define optimal management strategies," Dr. Davies said in an interview.
"Unfortunately, currently available databases do not contain information that may be important in defining such strategies (both surgical and nonsurgical), including anatomic and physiologic variables – whether they are neonates presenting in severe heart failure or older patients presenting for tricuspid valve repair or replacement," he added.
"We feel that in this setting, a prospective multi-institutional study would be of significant value. It should include operative and nonoperative patients, as well as precise diagnostic information and procedural details, to evaluate long-term outcomes including survival, reoperation and other reinterventions, as well as neurodevelopmental outcomes, functional health status, and quality of life," Dr. Davies concluded.
Dr. Davies and his colleagues reported having no relevant disclosures.
LOS ANGELES – Neonates in particular are at risk for poor outcomes from Ebstein’s malformation, showing significantly higher mortality than infants, children, and adults, according to a database study presented by Dr. Ryan R. Davies at the annual meeting of the Society of Thoracic Surgeons.
Ebstein’s malformation is a rare congenital cardiac anomaly. Surgery for Ebstein’s involves a range of procedures, and with low institutional volumes, the only available data on treatment have been limited to individual reports demonstrating highly variable approaches.
Dr. Davies of the Nemours/A.I. duPont Hospital for Children, Wilmington, Del., and his colleagues performed a retrospective study of procedures performed on patients with a primary diagnosis of Ebstein’s malformation (2002-2009) in the STS Congenital Heart Surgery Database.
A total of 595 operations on patients with Ebstein’s were included: 116 on neonates (19%), 122 on infants (21%), 264 on children (44%), and 93 on adults (16%). The authors found that average annual institutional case volumes were low (median, 1 case/year), and procedures varied according to age. Neonates had a high rate of palliative procedures: tricuspid valve (TV) closure (16%) and systemic-to-pulmonary shunts with or without TV closure (37%) and isolated TV closure (8.6%), with Ebstein’s repair or TV valvuloplasty performed in 32%.
Infants usually underwent superior cavopulmonary connections (52%).
Among older patients, procedures were mostly in three categories: TV surgery (children, 55%; adults, 69%), arrhythmia procedures (children, 9%; adults, 17%), and Fontan (children, 16%). In-hospital mortality was higher among neonatal patients (23%) than in infants (4%), children (0.8%), and adults (1.1%).
Among neonates, 36 subsequent procedures were performed during the same hospitalization in 27 patients (23%); including TV closure (11%); shunt (15%); Ebstein’s repair (17%) or TV replacement (15%); and heart transplantation (7.4%). Mortality was similar among neonates who had a second procedure and those who did not (27% vs. 23%, respectively). ECMO (extracorporeal membrane oxygenation) was used in 9% of neonates but in less than 2% of patients in other age groups.
"This study represents a broad overview of the diverse options for surgical treatment of Ebstein’s anomaly. It shows the challenges faced in caring for extremely ill neonatal patients. We have also shown that repair of Ebstein’s anomaly is performed infrequently at most centers, limiting the ability of individual series to define optimal management strategies," Dr. Davies said in an interview.
"Unfortunately, currently available databases do not contain information that may be important in defining such strategies (both surgical and nonsurgical), including anatomic and physiologic variables – whether they are neonates presenting in severe heart failure or older patients presenting for tricuspid valve repair or replacement," he added.
"We feel that in this setting, a prospective multi-institutional study would be of significant value. It should include operative and nonoperative patients, as well as precise diagnostic information and procedural details, to evaluate long-term outcomes including survival, reoperation and other reinterventions, as well as neurodevelopmental outcomes, functional health status, and quality of life," Dr. Davies concluded.
Dr. Davies and his colleagues reported having no relevant disclosures.
LOS ANGELES – Neonates in particular are at risk for poor outcomes from Ebstein’s malformation, showing significantly higher mortality than infants, children, and adults, according to a database study presented by Dr. Ryan R. Davies at the annual meeting of the Society of Thoracic Surgeons.
Ebstein’s malformation is a rare congenital cardiac anomaly. Surgery for Ebstein’s involves a range of procedures, and with low institutional volumes, the only available data on treatment have been limited to individual reports demonstrating highly variable approaches.
Dr. Davies of the Nemours/A.I. duPont Hospital for Children, Wilmington, Del., and his colleagues performed a retrospective study of procedures performed on patients with a primary diagnosis of Ebstein’s malformation (2002-2009) in the STS Congenital Heart Surgery Database.
A total of 595 operations on patients with Ebstein’s were included: 116 on neonates (19%), 122 on infants (21%), 264 on children (44%), and 93 on adults (16%). The authors found that average annual institutional case volumes were low (median, 1 case/year), and procedures varied according to age. Neonates had a high rate of palliative procedures: tricuspid valve (TV) closure (16%) and systemic-to-pulmonary shunts with or without TV closure (37%) and isolated TV closure (8.6%), with Ebstein’s repair or TV valvuloplasty performed in 32%.
Infants usually underwent superior cavopulmonary connections (52%).
Among older patients, procedures were mostly in three categories: TV surgery (children, 55%; adults, 69%), arrhythmia procedures (children, 9%; adults, 17%), and Fontan (children, 16%). In-hospital mortality was higher among neonatal patients (23%) than in infants (4%), children (0.8%), and adults (1.1%).
Among neonates, 36 subsequent procedures were performed during the same hospitalization in 27 patients (23%); including TV closure (11%); shunt (15%); Ebstein’s repair (17%) or TV replacement (15%); and heart transplantation (7.4%). Mortality was similar among neonates who had a second procedure and those who did not (27% vs. 23%, respectively). ECMO (extracorporeal membrane oxygenation) was used in 9% of neonates but in less than 2% of patients in other age groups.
"This study represents a broad overview of the diverse options for surgical treatment of Ebstein’s anomaly. It shows the challenges faced in caring for extremely ill neonatal patients. We have also shown that repair of Ebstein’s anomaly is performed infrequently at most centers, limiting the ability of individual series to define optimal management strategies," Dr. Davies said in an interview.
"Unfortunately, currently available databases do not contain information that may be important in defining such strategies (both surgical and nonsurgical), including anatomic and physiologic variables – whether they are neonates presenting in severe heart failure or older patients presenting for tricuspid valve repair or replacement," he added.
"We feel that in this setting, a prospective multi-institutional study would be of significant value. It should include operative and nonoperative patients, as well as precise diagnostic information and procedural details, to evaluate long-term outcomes including survival, reoperation and other reinterventions, as well as neurodevelopmental outcomes, functional health status, and quality of life," Dr. Davies concluded.
Dr. Davies and his colleagues reported having no relevant disclosures.
AT THE STS ANNUAL MEETING
Major Finding: In-hospital mortality was higher among neonatal patients (23%) than in infants (4%), children (0.8%), and adults (1.1%).
Data Source: A retrospective database analysis of 595 operations on patients with Ebstein’s malformation.
Disclosures: Dr. Davies and his colleagues reported having no relevant disclosures.
PFOs raise stroke risk from devices
LOS ANGELES – Patients with a patent foramen ovale and an implanted defibrillator or pacemaker may be good candidates for targeted closure, based on a review of more than 6,000 patients.
During an average follow-up of nearly 5 years, patients with a PFO who received an implantable cardioverter defibrillator (ICD) or a permanent pacemaker were more than fourfold more likely to develop stroke or transient ischemic attack (TIA) compared with implanted device recipients who did not have a PFO, Dr. Christopher V. DeSimone said at the annual scientific sessions of the American Heart Association.
"We think that this is a high-risk population that might benefit from PFO closure," said Dr. DeSimone, an internal medicine physician at the Mayo Clinic in Rochester, Minn. He acknowledged the poor efficacy of PFO closure for stroke prevention in several recent randomized trials, but noted that patients with a PFO who receive an ICD or permanent pacemaker may constitute a special subgroup that stands to benefit from PFO closure. "If a patient has a right atrial or ventricular lead and a clot forms and sits there next to the PFO, they would be at high risk" for a stroke or TIA, he said in an interview. In fact, trials that have assessed the efficacy of PFO closure explicitly excluded patients with permanent pacemakers as well as many ICD recipients because of their substantially impaired left ventricular function, such as in the CLOSURE I trial (N. Engl. J. Med. 2012;366:991-9). "This needs to be studied prospectively," he added, noting that his study was limited by being retrospective.
Dr. DeSimone and his associates reviewed 6,086 patients who received an ICD or permanent pacemaker at the Mayo Clinic during January 2000 to October 2010. The group included 375 patients with PFOs. Average age of the patients was 67 years; nearly two-thirds were men. About 15% had a history of stroke or TIA, about 44% had atrial fibrillation, and their average CHA2D2-VASc score was 3.1.
During an average follow-up of 4.7 years, the incidence of stroke or TIA was 11% in the PFO patients and 2% in the patients without a PFO. In a multivariate analysis that controlled for baseline demographic and clinical differences, including atrial fibrillation and aspirin and warfarin use, patients with a PFO were 4.6-fold more likely to have a stroke or TIA than were patients without a PFO, a statistically significant difference.
Additional analyses showed that the stroke and TIA rate remained significantly elevated in the PFO patients regardless of whether patients were on treatment with aspirin or on warfarin, and also regardless of whether or not they were older than age 65 or had a history of stroke or TIA, and regardless of whether they had a low or high CHA2D2-VASc score, Dr. DeSimone said. They saw no significant link between a PFO present and all-cause mortality.
The PFO-related difference in the incidence of stroke and TIA first became apparent about 1 year after device placement. The event curves continued to diverge more and more over time. Micro-emboli that originate on the device leads may pass through the PFO and into pulmonary circulation, causing increased pulmonary-artery pressures during the year after device placement. The increased right-sided pressure then favors a right-to-left shunt and increased embolization.
The stroke risk in this analysis may have underestimated the true risk because the methods used to find PFOs and stroke may not have been optimal.
Dr. DeSimone had no disclosures.
LOS ANGELES – Patients with a patent foramen ovale and an implanted defibrillator or pacemaker may be good candidates for targeted closure, based on a review of more than 6,000 patients.
During an average follow-up of nearly 5 years, patients with a PFO who received an implantable cardioverter defibrillator (ICD) or a permanent pacemaker were more than fourfold more likely to develop stroke or transient ischemic attack (TIA) compared with implanted device recipients who did not have a PFO, Dr. Christopher V. DeSimone said at the annual scientific sessions of the American Heart Association.
"We think that this is a high-risk population that might benefit from PFO closure," said Dr. DeSimone, an internal medicine physician at the Mayo Clinic in Rochester, Minn. He acknowledged the poor efficacy of PFO closure for stroke prevention in several recent randomized trials, but noted that patients with a PFO who receive an ICD or permanent pacemaker may constitute a special subgroup that stands to benefit from PFO closure. "If a patient has a right atrial or ventricular lead and a clot forms and sits there next to the PFO, they would be at high risk" for a stroke or TIA, he said in an interview. In fact, trials that have assessed the efficacy of PFO closure explicitly excluded patients with permanent pacemakers as well as many ICD recipients because of their substantially impaired left ventricular function, such as in the CLOSURE I trial (N. Engl. J. Med. 2012;366:991-9). "This needs to be studied prospectively," he added, noting that his study was limited by being retrospective.
Dr. DeSimone and his associates reviewed 6,086 patients who received an ICD or permanent pacemaker at the Mayo Clinic during January 2000 to October 2010. The group included 375 patients with PFOs. Average age of the patients was 67 years; nearly two-thirds were men. About 15% had a history of stroke or TIA, about 44% had atrial fibrillation, and their average CHA2D2-VASc score was 3.1.
During an average follow-up of 4.7 years, the incidence of stroke or TIA was 11% in the PFO patients and 2% in the patients without a PFO. In a multivariate analysis that controlled for baseline demographic and clinical differences, including atrial fibrillation and aspirin and warfarin use, patients with a PFO were 4.6-fold more likely to have a stroke or TIA than were patients without a PFO, a statistically significant difference.
Additional analyses showed that the stroke and TIA rate remained significantly elevated in the PFO patients regardless of whether patients were on treatment with aspirin or on warfarin, and also regardless of whether or not they were older than age 65 or had a history of stroke or TIA, and regardless of whether they had a low or high CHA2D2-VASc score, Dr. DeSimone said. They saw no significant link between a PFO present and all-cause mortality.
The PFO-related difference in the incidence of stroke and TIA first became apparent about 1 year after device placement. The event curves continued to diverge more and more over time. Micro-emboli that originate on the device leads may pass through the PFO and into pulmonary circulation, causing increased pulmonary-artery pressures during the year after device placement. The increased right-sided pressure then favors a right-to-left shunt and increased embolization.
The stroke risk in this analysis may have underestimated the true risk because the methods used to find PFOs and stroke may not have been optimal.
Dr. DeSimone had no disclosures.
LOS ANGELES – Patients with a patent foramen ovale and an implanted defibrillator or pacemaker may be good candidates for targeted closure, based on a review of more than 6,000 patients.
During an average follow-up of nearly 5 years, patients with a PFO who received an implantable cardioverter defibrillator (ICD) or a permanent pacemaker were more than fourfold more likely to develop stroke or transient ischemic attack (TIA) compared with implanted device recipients who did not have a PFO, Dr. Christopher V. DeSimone said at the annual scientific sessions of the American Heart Association.
"We think that this is a high-risk population that might benefit from PFO closure," said Dr. DeSimone, an internal medicine physician at the Mayo Clinic in Rochester, Minn. He acknowledged the poor efficacy of PFO closure for stroke prevention in several recent randomized trials, but noted that patients with a PFO who receive an ICD or permanent pacemaker may constitute a special subgroup that stands to benefit from PFO closure. "If a patient has a right atrial or ventricular lead and a clot forms and sits there next to the PFO, they would be at high risk" for a stroke or TIA, he said in an interview. In fact, trials that have assessed the efficacy of PFO closure explicitly excluded patients with permanent pacemakers as well as many ICD recipients because of their substantially impaired left ventricular function, such as in the CLOSURE I trial (N. Engl. J. Med. 2012;366:991-9). "This needs to be studied prospectively," he added, noting that his study was limited by being retrospective.
Dr. DeSimone and his associates reviewed 6,086 patients who received an ICD or permanent pacemaker at the Mayo Clinic during January 2000 to October 2010. The group included 375 patients with PFOs. Average age of the patients was 67 years; nearly two-thirds were men. About 15% had a history of stroke or TIA, about 44% had atrial fibrillation, and their average CHA2D2-VASc score was 3.1.
During an average follow-up of 4.7 years, the incidence of stroke or TIA was 11% in the PFO patients and 2% in the patients without a PFO. In a multivariate analysis that controlled for baseline demographic and clinical differences, including atrial fibrillation and aspirin and warfarin use, patients with a PFO were 4.6-fold more likely to have a stroke or TIA than were patients without a PFO, a statistically significant difference.
Additional analyses showed that the stroke and TIA rate remained significantly elevated in the PFO patients regardless of whether patients were on treatment with aspirin or on warfarin, and also regardless of whether or not they were older than age 65 or had a history of stroke or TIA, and regardless of whether they had a low or high CHA2D2-VASc score, Dr. DeSimone said. They saw no significant link between a PFO present and all-cause mortality.
The PFO-related difference in the incidence of stroke and TIA first became apparent about 1 year after device placement. The event curves continued to diverge more and more over time. Micro-emboli that originate on the device leads may pass through the PFO and into pulmonary circulation, causing increased pulmonary-artery pressures during the year after device placement. The increased right-sided pressure then favors a right-to-left shunt and increased embolization.
The stroke risk in this analysis may have underestimated the true risk because the methods used to find PFOs and stroke may not have been optimal.
Dr. DeSimone had no disclosures.
Major Finding: ICD or pacemaker recipients with a PFO had a 4.6-fold increased risk of stroke or TIA.
Data Source: A review of 6,086 patients at the Mayo Clinic.
Disclosures: Dr. DeSimone said that he had no disclosures.
The 2013 outlook in medicine: Is SGR action possible?
Could 2013 finally be the year to eliminate the Sustainable Growth Rate formula?
Officials at the American Medical Association say there's a chance that Congress could decide to permanently scrap the unpopular formula, which drives payment under the Medicare physician fee schedule, as part of a larger deal to cut the federal deficit.
"The fact that we've got this big potential deficit-reduction package would make us more optimistic that we can get [the SGR] taken care of this coming year," said Dr. Jeremy A. Lazarus, president of the American Medical Association.
On Jan. 1, lawmakers passed legislation providing a short-term, 1-year delay to the scheduled 26.5% SGR cut. The bill also included a 2-month delay to scheduled tax hikes and federal spending cuts that were planned as part of a deficit- reduction process known as sequestration. That gives Congress several weeks to craft a new plan to deal with the nation’s debt and the growth in Medicare spending.
It wouldn't be unprecedented for a permanent SGR fix to be considered as part of comprehensive deficit-reduction legislation. SGR repeal was included in bipartisan plans created by outside groups several times, including the Simpson-Bowles Commission, the Senate Gang of Six, and others, Dr. Lazarus said..
Although complete SGR repeal carries a 10-year price tag of nearly $300 billion, physicians argue that, since Congress always acts to avert the pay cuts triggered by the formula, the federal government does not save any money by keeping it on the books. The large cost of repeal, however, means that it may be easier to get the SGR fix inserted into a larger bill than to get lawmakers to approve it separately, Dr. Lazarus said.
The AMA is asking Congress to not only repeal the SGR but also to establish a period of stable Medicare payments so that physicians can begin to transition to a new payment system that focuses on quality of care, Dr. Lazarus said. In the meantime, the AMA and other groups have been working on developing new delivery and payment reform options that could offer an alternative to the current fee for service system.
"We do hope we can start changing the equation on reimbursement and going from fee for service to accounting for quality," said Dr. William A. Zoghbi, president of the American College of Cardiology.
ACC officials are eager to move away from the SGR but they are concerned about where the money to do so might come from. Dr. Zoghbi said that he doesn't want to see lawmakers robbing other health care priorities to pay for the fix. For instance, in December, lawmakers considered a proposal to pay for a 1-year SGR fix using money that was slated for increasing Medicaid payments to physicians providing primary care services. Instead, lawmakers financed the 1-year SGR fix mainly through cuts to hospital payments.
"These fixes cannot be on the backs of the professionals providing care," Dr. Zoghbi said.
ACA milestones
This year also will see some practice-impacting milestones under the Affordable Care Act.
Federal money now helps pay for preventive services for Medicaid patients, and many primary care services provided under Medicaid now are paid at the higher Medicare rate. Under the ACA, Medicaid payment increases to 100% of Medicare rates for family physicians, internists, and pediatricians when they provide certain primary care services. Subspecialists in these areas are also eligible for increased payments. The pay hike is for 2013 and 2014.
The law also provides an additional 2 years of funding to the Children's Health Insurance Program to continue coverage for those children for eligible under the Medicaid program.
The Independent Payment Advisory Board is slated to start work this year, even though its members have yet to be named by President Obama. The controversial 15-member panel is charged with making recommendations on how to reduce Medicare spending. Dr. Lazarus said the AMA will continue to work toward eliminating the IPAB.
Some of the biggest changes under the ACA - the expansion of Medicaid eligibility and the creation of state-based health insurance exchanges - are coming in 2014, but physician leaders said that doctors need to start preparing this year.
Exactly how to get ready will vary by state since both the Medicaid expansion and the exchanges will be largely state-run. The AMA is pushing to give physicians greater say by getting them seats on the boards of state exchanges. But even as physicians await more information on these changes, they can prepare by becoming more familiar with the Medicaid program since they are likely to see more of those patients, said Robert Doherty, senior vice president for governmental affairs and public policy at the American College of Physicians.
Penalties kick in
This year the Physician Quality Reporting System (PQRS) transitions from a pure incentive program to a mixed incentive/disincentive program. Previously, PQRS offered small bonus payments to physicians for successfully reporting on quality measures. Now, physicians who don't participate in the program will be assessed a penalty. The 1.5% cut to Medicare fees won’t come until 2015, but it will be based on participation this year. Physicians will see a 2% penalty in 2016 if they don't successfully report data during 2014.
"People don't realize that if they get past 2013, they won't have an opportunity to fix it for the next year," said Dr. Bruce Bagley, medical director for quality improvement at the American Academy of Family Physicians.
There are also penalties coming in Medicare's Electronic Prescribing (eRx) Incentive Program. To avoid a 2% penalty in 2014, physicians must meet Medicare's e-prescribing requirements by June 30, 2013.
Penalties from the Medicare Electronic Health Record (EHR) Incentive Program aren't coming until 2015, but Dr. Bagley said that physicians should take a good look at this program now to try to earn some money to offset the cost of EHR implementation. "The sooner you get going on this stuff, the better," he said.
A physician who starts participating this year can earn up to $39,000 over 4 years. Start next year and the bonus drops to $24,000. A 1% penalty takes effect in 2015, increasing to 2% the following year.
The transition to the ICD-10 coding set is another requirement that physicians need to keep in mind, ACP's Mr. Doherty said. The Department of Health and Human Services delayed the move to ICD-10 until October 2014, but Mr. Doherty said physicians can't afford to wait that long to prepare. The ACP is trying to convince federal officials to accept some alternative ways of coding that would both satisfy the ICD-10 requirements and be clinically relevant, he said.
Primary care gets a boost
Overall, the 2013 outlook will probably vary by specialty. The 2013 Medicare Physician Fee Schedule dealt some tough blows to subspecialists, making deep payment cuts in interventional cardiology, among others..
The 2013 fee schedule included two new transitional care management services codes (99495 and 99496) that will pay physicians for managing complex patients who have recently been discharged from a hospital or skilled nursing facility.
CPT code 99495 requires physicians or their staffs to make direct contact, by phone or electronically, with the patient or caregiver within 2 business days of discharge. A face-to-face visit with the patient is required within 14 calendar days of discharge. CPT code 99496 requires direct contact with the patient or caregiver within 2 business days and a face-to-face visit within 7 calendar days.
The ACP, the AMA, and other groups are developing a series of proposals calling on Medicare to begin paying for more of the non-face-to-face work involved in chronic care management.
Could 2013 finally be the year to eliminate the Sustainable Growth Rate formula?
Officials at the American Medical Association say there's a chance that Congress could decide to permanently scrap the unpopular formula, which drives payment under the Medicare physician fee schedule, as part of a larger deal to cut the federal deficit.
"The fact that we've got this big potential deficit-reduction package would make us more optimistic that we can get [the SGR] taken care of this coming year," said Dr. Jeremy A. Lazarus, president of the American Medical Association.
On Jan. 1, lawmakers passed legislation providing a short-term, 1-year delay to the scheduled 26.5% SGR cut. The bill also included a 2-month delay to scheduled tax hikes and federal spending cuts that were planned as part of a deficit- reduction process known as sequestration. That gives Congress several weeks to craft a new plan to deal with the nation’s debt and the growth in Medicare spending.
It wouldn't be unprecedented for a permanent SGR fix to be considered as part of comprehensive deficit-reduction legislation. SGR repeal was included in bipartisan plans created by outside groups several times, including the Simpson-Bowles Commission, the Senate Gang of Six, and others, Dr. Lazarus said..
Although complete SGR repeal carries a 10-year price tag of nearly $300 billion, physicians argue that, since Congress always acts to avert the pay cuts triggered by the formula, the federal government does not save any money by keeping it on the books. The large cost of repeal, however, means that it may be easier to get the SGR fix inserted into a larger bill than to get lawmakers to approve it separately, Dr. Lazarus said.
The AMA is asking Congress to not only repeal the SGR but also to establish a period of stable Medicare payments so that physicians can begin to transition to a new payment system that focuses on quality of care, Dr. Lazarus said. In the meantime, the AMA and other groups have been working on developing new delivery and payment reform options that could offer an alternative to the current fee for service system.
"We do hope we can start changing the equation on reimbursement and going from fee for service to accounting for quality," said Dr. William A. Zoghbi, president of the American College of Cardiology.
ACC officials are eager to move away from the SGR but they are concerned about where the money to do so might come from. Dr. Zoghbi said that he doesn't want to see lawmakers robbing other health care priorities to pay for the fix. For instance, in December, lawmakers considered a proposal to pay for a 1-year SGR fix using money that was slated for increasing Medicaid payments to physicians providing primary care services. Instead, lawmakers financed the 1-year SGR fix mainly through cuts to hospital payments.
"These fixes cannot be on the backs of the professionals providing care," Dr. Zoghbi said.
ACA milestones
This year also will see some practice-impacting milestones under the Affordable Care Act.
Federal money now helps pay for preventive services for Medicaid patients, and many primary care services provided under Medicaid now are paid at the higher Medicare rate. Under the ACA, Medicaid payment increases to 100% of Medicare rates for family physicians, internists, and pediatricians when they provide certain primary care services. Subspecialists in these areas are also eligible for increased payments. The pay hike is for 2013 and 2014.
The law also provides an additional 2 years of funding to the Children's Health Insurance Program to continue coverage for those children for eligible under the Medicaid program.
The Independent Payment Advisory Board is slated to start work this year, even though its members have yet to be named by President Obama. The controversial 15-member panel is charged with making recommendations on how to reduce Medicare spending. Dr. Lazarus said the AMA will continue to work toward eliminating the IPAB.
Some of the biggest changes under the ACA - the expansion of Medicaid eligibility and the creation of state-based health insurance exchanges - are coming in 2014, but physician leaders said that doctors need to start preparing this year.
Exactly how to get ready will vary by state since both the Medicaid expansion and the exchanges will be largely state-run. The AMA is pushing to give physicians greater say by getting them seats on the boards of state exchanges. But even as physicians await more information on these changes, they can prepare by becoming more familiar with the Medicaid program since they are likely to see more of those patients, said Robert Doherty, senior vice president for governmental affairs and public policy at the American College of Physicians.
Penalties kick in
This year the Physician Quality Reporting System (PQRS) transitions from a pure incentive program to a mixed incentive/disincentive program. Previously, PQRS offered small bonus payments to physicians for successfully reporting on quality measures. Now, physicians who don't participate in the program will be assessed a penalty. The 1.5% cut to Medicare fees won’t come until 2015, but it will be based on participation this year. Physicians will see a 2% penalty in 2016 if they don't successfully report data during 2014.
"People don't realize that if they get past 2013, they won't have an opportunity to fix it for the next year," said Dr. Bruce Bagley, medical director for quality improvement at the American Academy of Family Physicians.
There are also penalties coming in Medicare's Electronic Prescribing (eRx) Incentive Program. To avoid a 2% penalty in 2014, physicians must meet Medicare's e-prescribing requirements by June 30, 2013.
Penalties from the Medicare Electronic Health Record (EHR) Incentive Program aren't coming until 2015, but Dr. Bagley said that physicians should take a good look at this program now to try to earn some money to offset the cost of EHR implementation. "The sooner you get going on this stuff, the better," he said.
A physician who starts participating this year can earn up to $39,000 over 4 years. Start next year and the bonus drops to $24,000. A 1% penalty takes effect in 2015, increasing to 2% the following year.
The transition to the ICD-10 coding set is another requirement that physicians need to keep in mind, ACP's Mr. Doherty said. The Department of Health and Human Services delayed the move to ICD-10 until October 2014, but Mr. Doherty said physicians can't afford to wait that long to prepare. The ACP is trying to convince federal officials to accept some alternative ways of coding that would both satisfy the ICD-10 requirements and be clinically relevant, he said.
Primary care gets a boost
Overall, the 2013 outlook will probably vary by specialty. The 2013 Medicare Physician Fee Schedule dealt some tough blows to subspecialists, making deep payment cuts in interventional cardiology, among others..
The 2013 fee schedule included two new transitional care management services codes (99495 and 99496) that will pay physicians for managing complex patients who have recently been discharged from a hospital or skilled nursing facility.
CPT code 99495 requires physicians or their staffs to make direct contact, by phone or electronically, with the patient or caregiver within 2 business days of discharge. A face-to-face visit with the patient is required within 14 calendar days of discharge. CPT code 99496 requires direct contact with the patient or caregiver within 2 business days and a face-to-face visit within 7 calendar days.
The ACP, the AMA, and other groups are developing a series of proposals calling on Medicare to begin paying for more of the non-face-to-face work involved in chronic care management.
Could 2013 finally be the year to eliminate the Sustainable Growth Rate formula?
Officials at the American Medical Association say there's a chance that Congress could decide to permanently scrap the unpopular formula, which drives payment under the Medicare physician fee schedule, as part of a larger deal to cut the federal deficit.
"The fact that we've got this big potential deficit-reduction package would make us more optimistic that we can get [the SGR] taken care of this coming year," said Dr. Jeremy A. Lazarus, president of the American Medical Association.
On Jan. 1, lawmakers passed legislation providing a short-term, 1-year delay to the scheduled 26.5% SGR cut. The bill also included a 2-month delay to scheduled tax hikes and federal spending cuts that were planned as part of a deficit- reduction process known as sequestration. That gives Congress several weeks to craft a new plan to deal with the nation’s debt and the growth in Medicare spending.
It wouldn't be unprecedented for a permanent SGR fix to be considered as part of comprehensive deficit-reduction legislation. SGR repeal was included in bipartisan plans created by outside groups several times, including the Simpson-Bowles Commission, the Senate Gang of Six, and others, Dr. Lazarus said..
Although complete SGR repeal carries a 10-year price tag of nearly $300 billion, physicians argue that, since Congress always acts to avert the pay cuts triggered by the formula, the federal government does not save any money by keeping it on the books. The large cost of repeal, however, means that it may be easier to get the SGR fix inserted into a larger bill than to get lawmakers to approve it separately, Dr. Lazarus said.
The AMA is asking Congress to not only repeal the SGR but also to establish a period of stable Medicare payments so that physicians can begin to transition to a new payment system that focuses on quality of care, Dr. Lazarus said. In the meantime, the AMA and other groups have been working on developing new delivery and payment reform options that could offer an alternative to the current fee for service system.
"We do hope we can start changing the equation on reimbursement and going from fee for service to accounting for quality," said Dr. William A. Zoghbi, president of the American College of Cardiology.
ACC officials are eager to move away from the SGR but they are concerned about where the money to do so might come from. Dr. Zoghbi said that he doesn't want to see lawmakers robbing other health care priorities to pay for the fix. For instance, in December, lawmakers considered a proposal to pay for a 1-year SGR fix using money that was slated for increasing Medicaid payments to physicians providing primary care services. Instead, lawmakers financed the 1-year SGR fix mainly through cuts to hospital payments.
"These fixes cannot be on the backs of the professionals providing care," Dr. Zoghbi said.
ACA milestones
This year also will see some practice-impacting milestones under the Affordable Care Act.
Federal money now helps pay for preventive services for Medicaid patients, and many primary care services provided under Medicaid now are paid at the higher Medicare rate. Under the ACA, Medicaid payment increases to 100% of Medicare rates for family physicians, internists, and pediatricians when they provide certain primary care services. Subspecialists in these areas are also eligible for increased payments. The pay hike is for 2013 and 2014.
The law also provides an additional 2 years of funding to the Children's Health Insurance Program to continue coverage for those children for eligible under the Medicaid program.
The Independent Payment Advisory Board is slated to start work this year, even though its members have yet to be named by President Obama. The controversial 15-member panel is charged with making recommendations on how to reduce Medicare spending. Dr. Lazarus said the AMA will continue to work toward eliminating the IPAB.
Some of the biggest changes under the ACA - the expansion of Medicaid eligibility and the creation of state-based health insurance exchanges - are coming in 2014, but physician leaders said that doctors need to start preparing this year.
Exactly how to get ready will vary by state since both the Medicaid expansion and the exchanges will be largely state-run. The AMA is pushing to give physicians greater say by getting them seats on the boards of state exchanges. But even as physicians await more information on these changes, they can prepare by becoming more familiar with the Medicaid program since they are likely to see more of those patients, said Robert Doherty, senior vice president for governmental affairs and public policy at the American College of Physicians.
Penalties kick in
This year the Physician Quality Reporting System (PQRS) transitions from a pure incentive program to a mixed incentive/disincentive program. Previously, PQRS offered small bonus payments to physicians for successfully reporting on quality measures. Now, physicians who don't participate in the program will be assessed a penalty. The 1.5% cut to Medicare fees won’t come until 2015, but it will be based on participation this year. Physicians will see a 2% penalty in 2016 if they don't successfully report data during 2014.
"People don't realize that if they get past 2013, they won't have an opportunity to fix it for the next year," said Dr. Bruce Bagley, medical director for quality improvement at the American Academy of Family Physicians.
There are also penalties coming in Medicare's Electronic Prescribing (eRx) Incentive Program. To avoid a 2% penalty in 2014, physicians must meet Medicare's e-prescribing requirements by June 30, 2013.
Penalties from the Medicare Electronic Health Record (EHR) Incentive Program aren't coming until 2015, but Dr. Bagley said that physicians should take a good look at this program now to try to earn some money to offset the cost of EHR implementation. "The sooner you get going on this stuff, the better," he said.
A physician who starts participating this year can earn up to $39,000 over 4 years. Start next year and the bonus drops to $24,000. A 1% penalty takes effect in 2015, increasing to 2% the following year.
The transition to the ICD-10 coding set is another requirement that physicians need to keep in mind, ACP's Mr. Doherty said. The Department of Health and Human Services delayed the move to ICD-10 until October 2014, but Mr. Doherty said physicians can't afford to wait that long to prepare. The ACP is trying to convince federal officials to accept some alternative ways of coding that would both satisfy the ICD-10 requirements and be clinically relevant, he said.
Primary care gets a boost
Overall, the 2013 outlook will probably vary by specialty. The 2013 Medicare Physician Fee Schedule dealt some tough blows to subspecialists, making deep payment cuts in interventional cardiology, among others..
The 2013 fee schedule included two new transitional care management services codes (99495 and 99496) that will pay physicians for managing complex patients who have recently been discharged from a hospital or skilled nursing facility.
CPT code 99495 requires physicians or their staffs to make direct contact, by phone or electronically, with the patient or caregiver within 2 business days of discharge. A face-to-face visit with the patient is required within 14 calendar days of discharge. CPT code 99496 requires direct contact with the patient or caregiver within 2 business days and a face-to-face visit within 7 calendar days.
The ACP, the AMA, and other groups are developing a series of proposals calling on Medicare to begin paying for more of the non-face-to-face work involved in chronic care management.