User login
Non-White subjects are sparse in DMT trials for MS
NATIONAL HARBOR, MD. -- Over 25 years of clinical research, phase 3 trials of approved disease-modifying therapies (DMTs) for multiple sclerosis (MS) were overwhelmingly made up of White subjects, a new analysis finds, and many studies failed to report percentages of non-White subjects at all. Researchers also found that the websites of multiple major drug manufacturers don’t include any trial data about how medications may affect people of different races and ethnicities.
It’s clear that “non-White participants are significantly underreported and unrepresented,” said study corresponding author and Dell Medical School/The University of Texas at Austin neurologist Leorah Freeman, MD, PhD, in an interview.
The study was presented at the annual meeting of the Consortium of Multiple Sclerosis Centers and published in Neurology.
“The lack of diversity in MS research is something that has been sporadically discussed in the past. By conducting this systematic review of MS phase 3 trials, we wanted to put numbers on this issue and review the evidence systematically,” Dr. Freeman said. “By doing so, we hoped to raise awareness about the problem of underreporting and underrepresentation of non-White participants in trials so that we, as a community involved in MS research, can start having the difficult conversations needed for change to occur.”
25 years of clinical research
The researchers reviewed 44 phase 3 studies from 1995-2020 that represented 45 trials. “We wanted to capture data from the very first global trials being conducted for the approval of MS DMTs, and the first was published in 1995,” Dr. Freeman said. “We were interested in understanding the impact of trial globalization over a long period of time on diversity of enrollment.”
The studies include trials of mainstays of MS treatment such as interferon, glatiramer acetate, teriflunomide, dimethyl fumarate, diroximel fumarate, fingolimod, natalizumab, and others.
The researchers found that 17 (37.8%) of the trials did not report race or ethnicity, 14 (31.1%) reported race and ethnicity as proportion of White participants only, and 14 (31.1%) reported 2 or more races/ethnicities.
Of the 28 trials with racial breakdowns, the median percentage of White participants was 93.8% (range 78.5-99.6% across 28 studies), 1.9% for Black participants (range 0.1-8.1% across 14 studies), and 0.5% for Asian participants (range 0.1-14.5% across 11 studies).
The studies often failed to account for non-White subjects even though “Black people, in particular, have been shown to have a more severe disease course,” Dr. Freeman said.
A 2022 study of more than 2.6 million Southern California adults finds that prevalence of MS was similar among White and Black people at about 230 per 100,000. “Taken together with previous studies, these findings indicate that the burden of MS in the United States Black community has long been underrecognized,” the researchers wrote.
According to Dr. Freeman, it’s unclear why the studies were so dominated by White subjects. “Lack of awareness about the importance of this information likely explains why this information often goes unreported.”
She highlighted the TOWER (teriflunomide) and DEFINE and CONFIRM (dimethyl fumarate) studies as positive examples. “We noted the inclusion of trial sites in Asia and consequently a higher representation of Asian people with MS in those trials. We felt these studies were examples of how trial globalization can support better representation of underrepresented groups.”
And she noted that the ongoing CHIMES trial is examining the use of ocrelizumab in Black and Hispanic people with MS. “This study was designed in partnership with MS patients and advocacy groups to bridge gaps in clinical trial participation in these communities,” she said. “Innovative strategies were developed to increase participation of Black and Hispanic patients in this trial.”
What should happen next?
Going forward, Dr. Freeman said, “MS researchers, DMT manufacturers, sponsors, and publishers need to set better standards for racial and ethnic representation and reporting in trial publications.”
In an interview, epidemiologist Luisa N. Borrell, DDS, PhD, a professor who studies race and medicine at City University of New York, said the new study is valid and useful. She noted that it reflects the findings of a 2022 analysis of more than 20,500 clinical trials in the U.S. from 2000-2020: Only 43% reported racial/ethnic breakdowns, and the subjects were much more White than the population at large.
Possible reasons for the disparity include distrust among possible participants and lack of health literacy, she said, which are both “modifiable issues.”
Dr. Borrell added: “Clinical trials should aim to recruit populations affected by the outcome of interest. That would allow for the intervention to effectively treat those who need it the most. Moreover, the lack of diversity of trials brings issues of generalizability and transportability of the findings.”
No funding is reported. Dr. Freeman and some of the other study authors report various disclosures.
NATIONAL HARBOR, MD. -- Over 25 years of clinical research, phase 3 trials of approved disease-modifying therapies (DMTs) for multiple sclerosis (MS) were overwhelmingly made up of White subjects, a new analysis finds, and many studies failed to report percentages of non-White subjects at all. Researchers also found that the websites of multiple major drug manufacturers don’t include any trial data about how medications may affect people of different races and ethnicities.
It’s clear that “non-White participants are significantly underreported and unrepresented,” said study corresponding author and Dell Medical School/The University of Texas at Austin neurologist Leorah Freeman, MD, PhD, in an interview.
The study was presented at the annual meeting of the Consortium of Multiple Sclerosis Centers and published in Neurology.
“The lack of diversity in MS research is something that has been sporadically discussed in the past. By conducting this systematic review of MS phase 3 trials, we wanted to put numbers on this issue and review the evidence systematically,” Dr. Freeman said. “By doing so, we hoped to raise awareness about the problem of underreporting and underrepresentation of non-White participants in trials so that we, as a community involved in MS research, can start having the difficult conversations needed for change to occur.”
25 years of clinical research
The researchers reviewed 44 phase 3 studies from 1995-2020 that represented 45 trials. “We wanted to capture data from the very first global trials being conducted for the approval of MS DMTs, and the first was published in 1995,” Dr. Freeman said. “We were interested in understanding the impact of trial globalization over a long period of time on diversity of enrollment.”
The studies include trials of mainstays of MS treatment such as interferon, glatiramer acetate, teriflunomide, dimethyl fumarate, diroximel fumarate, fingolimod, natalizumab, and others.
The researchers found that 17 (37.8%) of the trials did not report race or ethnicity, 14 (31.1%) reported race and ethnicity as proportion of White participants only, and 14 (31.1%) reported 2 or more races/ethnicities.
Of the 28 trials with racial breakdowns, the median percentage of White participants was 93.8% (range 78.5-99.6% across 28 studies), 1.9% for Black participants (range 0.1-8.1% across 14 studies), and 0.5% for Asian participants (range 0.1-14.5% across 11 studies).
The studies often failed to account for non-White subjects even though “Black people, in particular, have been shown to have a more severe disease course,” Dr. Freeman said.
A 2022 study of more than 2.6 million Southern California adults finds that prevalence of MS was similar among White and Black people at about 230 per 100,000. “Taken together with previous studies, these findings indicate that the burden of MS in the United States Black community has long been underrecognized,” the researchers wrote.
According to Dr. Freeman, it’s unclear why the studies were so dominated by White subjects. “Lack of awareness about the importance of this information likely explains why this information often goes unreported.”
She highlighted the TOWER (teriflunomide) and DEFINE and CONFIRM (dimethyl fumarate) studies as positive examples. “We noted the inclusion of trial sites in Asia and consequently a higher representation of Asian people with MS in those trials. We felt these studies were examples of how trial globalization can support better representation of underrepresented groups.”
And she noted that the ongoing CHIMES trial is examining the use of ocrelizumab in Black and Hispanic people with MS. “This study was designed in partnership with MS patients and advocacy groups to bridge gaps in clinical trial participation in these communities,” she said. “Innovative strategies were developed to increase participation of Black and Hispanic patients in this trial.”
What should happen next?
Going forward, Dr. Freeman said, “MS researchers, DMT manufacturers, sponsors, and publishers need to set better standards for racial and ethnic representation and reporting in trial publications.”
In an interview, epidemiologist Luisa N. Borrell, DDS, PhD, a professor who studies race and medicine at City University of New York, said the new study is valid and useful. She noted that it reflects the findings of a 2022 analysis of more than 20,500 clinical trials in the U.S. from 2000-2020: Only 43% reported racial/ethnic breakdowns, and the subjects were much more White than the population at large.
Possible reasons for the disparity include distrust among possible participants and lack of health literacy, she said, which are both “modifiable issues.”
Dr. Borrell added: “Clinical trials should aim to recruit populations affected by the outcome of interest. That would allow for the intervention to effectively treat those who need it the most. Moreover, the lack of diversity of trials brings issues of generalizability and transportability of the findings.”
No funding is reported. Dr. Freeman and some of the other study authors report various disclosures.
NATIONAL HARBOR, MD. -- Over 25 years of clinical research, phase 3 trials of approved disease-modifying therapies (DMTs) for multiple sclerosis (MS) were overwhelmingly made up of White subjects, a new analysis finds, and many studies failed to report percentages of non-White subjects at all. Researchers also found that the websites of multiple major drug manufacturers don’t include any trial data about how medications may affect people of different races and ethnicities.
It’s clear that “non-White participants are significantly underreported and unrepresented,” said study corresponding author and Dell Medical School/The University of Texas at Austin neurologist Leorah Freeman, MD, PhD, in an interview.
The study was presented at the annual meeting of the Consortium of Multiple Sclerosis Centers and published in Neurology.
“The lack of diversity in MS research is something that has been sporadically discussed in the past. By conducting this systematic review of MS phase 3 trials, we wanted to put numbers on this issue and review the evidence systematically,” Dr. Freeman said. “By doing so, we hoped to raise awareness about the problem of underreporting and underrepresentation of non-White participants in trials so that we, as a community involved in MS research, can start having the difficult conversations needed for change to occur.”
25 years of clinical research
The researchers reviewed 44 phase 3 studies from 1995-2020 that represented 45 trials. “We wanted to capture data from the very first global trials being conducted for the approval of MS DMTs, and the first was published in 1995,” Dr. Freeman said. “We were interested in understanding the impact of trial globalization over a long period of time on diversity of enrollment.”
The studies include trials of mainstays of MS treatment such as interferon, glatiramer acetate, teriflunomide, dimethyl fumarate, diroximel fumarate, fingolimod, natalizumab, and others.
The researchers found that 17 (37.8%) of the trials did not report race or ethnicity, 14 (31.1%) reported race and ethnicity as proportion of White participants only, and 14 (31.1%) reported 2 or more races/ethnicities.
Of the 28 trials with racial breakdowns, the median percentage of White participants was 93.8% (range 78.5-99.6% across 28 studies), 1.9% for Black participants (range 0.1-8.1% across 14 studies), and 0.5% for Asian participants (range 0.1-14.5% across 11 studies).
The studies often failed to account for non-White subjects even though “Black people, in particular, have been shown to have a more severe disease course,” Dr. Freeman said.
A 2022 study of more than 2.6 million Southern California adults finds that prevalence of MS was similar among White and Black people at about 230 per 100,000. “Taken together with previous studies, these findings indicate that the burden of MS in the United States Black community has long been underrecognized,” the researchers wrote.
According to Dr. Freeman, it’s unclear why the studies were so dominated by White subjects. “Lack of awareness about the importance of this information likely explains why this information often goes unreported.”
She highlighted the TOWER (teriflunomide) and DEFINE and CONFIRM (dimethyl fumarate) studies as positive examples. “We noted the inclusion of trial sites in Asia and consequently a higher representation of Asian people with MS in those trials. We felt these studies were examples of how trial globalization can support better representation of underrepresented groups.”
And she noted that the ongoing CHIMES trial is examining the use of ocrelizumab in Black and Hispanic people with MS. “This study was designed in partnership with MS patients and advocacy groups to bridge gaps in clinical trial participation in these communities,” she said. “Innovative strategies were developed to increase participation of Black and Hispanic patients in this trial.”
What should happen next?
Going forward, Dr. Freeman said, “MS researchers, DMT manufacturers, sponsors, and publishers need to set better standards for racial and ethnic representation and reporting in trial publications.”
In an interview, epidemiologist Luisa N. Borrell, DDS, PhD, a professor who studies race and medicine at City University of New York, said the new study is valid and useful. She noted that it reflects the findings of a 2022 analysis of more than 20,500 clinical trials in the U.S. from 2000-2020: Only 43% reported racial/ethnic breakdowns, and the subjects were much more White than the population at large.
Possible reasons for the disparity include distrust among possible participants and lack of health literacy, she said, which are both “modifiable issues.”
Dr. Borrell added: “Clinical trials should aim to recruit populations affected by the outcome of interest. That would allow for the intervention to effectively treat those who need it the most. Moreover, the lack of diversity of trials brings issues of generalizability and transportability of the findings.”
No funding is reported. Dr. Freeman and some of the other study authors report various disclosures.
AT CMSC 2022
Autism ‘elopement’ raises summer drowning risk
It happens quickly: A child on the autism spectrum bolts from supervision and disappears – an emergency called “autism elopement.” While any child can wander off, children on the autism spectrum face particular risks. These include the lure of water and the risk of drowning.
Some youngsters on the spectrum will follow this strong attraction to water and head for a nearby pond, river, or swimming pool. Such circumstances have made drowning a leading cause of death for these missing youths.
Autism elopement can happen any time. Summer can be especially dangerous. When the weather warms, the risk of drowning death rises, says Lori McIlwain, cofounder of the National Autism Association.
“The fatality risk is higher in May, June, July for that child to exit the setting unnoticed, especially if there’s an outdoor gathering and then they go directly to water,” Ms. McIlwain says. For instance, she says children can dart away during outdoor play, barbecues, gatherings, and other activities. Or they might wander off while vacationing near a beach or hotel pool.
Autism elopement
Many people don’t know about this risk, including some families with youngsters on the autism spectrum. The National Center for Missing and Exploited Children is working to change that – and find solutions.
About 12 years ago, “we started noticing a very disturbing trend that children with autism were going missing and they were having grave results,” says John Bischoff, vice president of the Center’s Missing Children Division.
The Center analyzed a decade of data on accidental deaths of children on the autism spectrum. Drowning was the #1 cause, accounting for 84% of those deaths.
In 2012, researchers reported on autism and wandering in the journal Pediatrics. They analyzed answers from about 1,000 families to an online survey on the topic. Parents who had children on the spectrum and children not on the spectrum responded. Nearly half of the parents said their child with autism had tried to wander off after age 4, and 26% had gone missing long enough to cause concern.
“Of those who went missing, 24% were in danger of drowning and 65% were in danger of traffic injury,” the researchers wrote. Children on the spectrum might also be drawn to traffic signs, highways, fire trucks, and trains.
In comparison, brothers and sisters of all ages who were not on the spectrum were much less likely to have wandered off.
Seeking a quiet place
It’s not entirely clear why children with autism are so drawn to water, Ms. McIlwain says. But there are some clues.
“What we see is that these children exit settings that are usually bothersome,” Ms. McIlwain says. “[Those settings are] loud, with a high amount of stimuli or stress or commotion, and they go to a quiet place, usually water in a quiet area. It’s calm. It’s peaceful.”
Water isn’t the only dangerous draw. When autism elopement happens, “they also go to the woods, they go to abandoned vehicles,” she says. “So any quiet thing is usually where they will head.”
A family’s loss
Beth Dilg, a mother in Maryland, lost her 7-year-old daughter, Savannah Martin, who was on the autism spectrum, to drowning in 2011. Ms. Dilg had been living in Oklahoma and raising her three children alone after separating from her husband. On a chilly February day, Savannah and her 2-year-old brother left their house after Ms. Dilg had asked her 11-year-old son to keep watch while she went into the bathroom for a few minutes.
When Ms. Dilg realized the two younger kids had left, she searched the property frantically. She shouted Savannah’s name repeatedly, but the child, who had limited language, didn’t come when called. “I feel like she knew what her name was,” Ms. Dilg says, “but it wasn’t like you’d call her name and she’d come to you.”
Ms. Dilg ran to a pond near her property after her 11-year-old son said that the two siblings were in the water. Ms. Dilg entered the water and grabbed her toddler, who had survived after having been kept afloat by his bicycle helmet. But when Ms. Dilg reached Savannah, she was already unresponsive. A neighbor helped pull the children out.
It can happen in any family. Even when a parent takes precautions, a child can slip out in a moment, perhaps while the parent is asleep or taking care of personal needs or if the child is at school or elsewhere.
“It’s unrealistic to say that you’d never take your eyes off your kid,” Ms. Dilg says.
She had tried to protect Savannah by starting her on swimming lessons, installing high locks on the doors, and trying to teach her about how to stay safe.
Still, children can be skillful in finding ways to escape, Ms. Dilg says. “These kids with autism are so smart. They may not be verbal, but they have this level of intelligence,” she says. “You always have to stay a step ahead of them.”
Ms. Dilg has been a longtime volunteer with Team HOPE, a peer support group with the National Center for Missing and Exploited Children. She offers emotional support to parents whose children are missing or who have died, including the parents of youngsters with autism who have drowned.
Teaching first responders
If a child on the autism spectrum goes missing, searching for them can be complicated by their condition. For instance, some children cannot speak or aren’t able to respond to searchers calling their name. The National Center for Missing and Exploited Children offers training to law enforcement and provides search protocols for first responders.
The center has drawn on expertise from Laurie Reyes, an officer with Maryland’s Montgomery County Police Department. In 2005, Ms. Reyes created a special unit within the department to focus on safety for people at risk for wandering. They have conditions that include autism/intellectual and developmental disabilities, as well as Alzheimer’s and other forms of dementia.
“We have a culture of awareness here,” Ms. Reyes says. All Montgomery County recruits and officers receive training in how to interact with those on the autism spectrum, who may not respond to police commands. Police also learn how to search, including immediately checking bodies of water. “We’ve had many times where we’ve located individuals in bodies of water,” Ms. Reyes says.
Don’t wait to call 911. When a child goes missing, time matters. Ms. Reyes advises families not to search on their own. “Call 911 right away,” she says.
Top safety tips to help prevent autism elopement
Use these tips to help keep kids on the autism spectrum safe and prevent drownings.
Secure your home. Use window and door alarms to alert you if a door or window becomes ajar. “Those door alarms are essential,” Ms. McIlwain says.
You can buy alarms online or get them free from the National Autism Association through its Big Red Safety Box program.
You can also buy portable door alarms for travel and arrange to have door alarms at your child’s school.
Ms. McIlwain advises securing the home with adequate locks and using baby monitors. Installing visual prompts, such as a stop sign on the door, might also cue a child not to leave.
Use personal identification. Ms. McIlwain says that children with autism must wear identification, such as a wristband, that includes their name, autism diagnosis, and the name and phone number of a contact person.
If children won’t wear a wristband, IDs on shoelaces are an option, she says. But parents should be aware that kids might leave without shoes or take them off before entering water.
Parents can also weigh the pros and cons of using tracking and locater devices, Ms. McIlwain says.
Identify triggers. “What’s going to make the child want to leave the setting? Is it noise? Is it a certain thing that they fear?” Ms. McIlwain says. “There’s always a reason.”
If parents can identify particular triggers, they can use calming techniques, for example, or provide headphones to counteract bothersome noises.
Teach safety skills, such as swimming lessons. Swimming lessons are important, Ms. McIlwain says. However, children with autism are often bothered by noise and commotion. So a regular swim class might not work for them.
Instead, Ms. McIlwain encourages parents to ask their local YMCA about special-needs swimming lessons or to search for such lessons online. What usually turns out to be best is to give the child a few private swimming lessons “with somebody who understands autism.”
For the child’s final lesson, they should swim fully clothed and with shoes on, Ms. McIlwain says. “A lot of our kids go straight into water fully clothed, and they just need to be able to be familiar with how that feels and the weight of that and be able to swim like that as well.”
If a child is drawn to water, discuss a scheduled time to go so that the youngster can wait, Ms. McIlwain says. “They can see that they’re going to get that water time. They’re going to be able to go to that place. They’re going to wait instead of trying to go on their own.”
Keep a close watch and team up. “When there is a family gathering or an outdoor barbecue, a lot of times, we all think, there are more adults here, so there are going to be more eyes on all the kids. And that always ends up being opposite, right?” Ms. McIlwain says.
Be specific about who is monitoring the child’s safety.
“We encourage parents to do the ‘Tag, you’re it’ game with one another. So you basically tag an adult who is responsible for keeping an eye on that child for a period of time so that there’s always supervision.”
Be prepared
There are a few things you can do now to be ready in case your child slips away. These measures may help find the child quickly.
Take photos today. Keep a full-length shot and a head shot of your child and store them electronically. If your child wanders away, you can immediately send the images to law enforcement to help them search.
Write a 911 script. Have this document ready in case your child wanders. It describes, among other things, points of interest that might draw your child, as well as locations of nearby bodies of water. By having it all written down, you’ll be able to share the information quickly with first responders. The Montgomery County Police Department has a “Wandering 911 Script” that you can download and use.
A version of this article first appeared on Webmd.com.
It happens quickly: A child on the autism spectrum bolts from supervision and disappears – an emergency called “autism elopement.” While any child can wander off, children on the autism spectrum face particular risks. These include the lure of water and the risk of drowning.
Some youngsters on the spectrum will follow this strong attraction to water and head for a nearby pond, river, or swimming pool. Such circumstances have made drowning a leading cause of death for these missing youths.
Autism elopement can happen any time. Summer can be especially dangerous. When the weather warms, the risk of drowning death rises, says Lori McIlwain, cofounder of the National Autism Association.
“The fatality risk is higher in May, June, July for that child to exit the setting unnoticed, especially if there’s an outdoor gathering and then they go directly to water,” Ms. McIlwain says. For instance, she says children can dart away during outdoor play, barbecues, gatherings, and other activities. Or they might wander off while vacationing near a beach or hotel pool.
Autism elopement
Many people don’t know about this risk, including some families with youngsters on the autism spectrum. The National Center for Missing and Exploited Children is working to change that – and find solutions.
About 12 years ago, “we started noticing a very disturbing trend that children with autism were going missing and they were having grave results,” says John Bischoff, vice president of the Center’s Missing Children Division.
The Center analyzed a decade of data on accidental deaths of children on the autism spectrum. Drowning was the #1 cause, accounting for 84% of those deaths.
In 2012, researchers reported on autism and wandering in the journal Pediatrics. They analyzed answers from about 1,000 families to an online survey on the topic. Parents who had children on the spectrum and children not on the spectrum responded. Nearly half of the parents said their child with autism had tried to wander off after age 4, and 26% had gone missing long enough to cause concern.
“Of those who went missing, 24% were in danger of drowning and 65% were in danger of traffic injury,” the researchers wrote. Children on the spectrum might also be drawn to traffic signs, highways, fire trucks, and trains.
In comparison, brothers and sisters of all ages who were not on the spectrum were much less likely to have wandered off.
Seeking a quiet place
It’s not entirely clear why children with autism are so drawn to water, Ms. McIlwain says. But there are some clues.
“What we see is that these children exit settings that are usually bothersome,” Ms. McIlwain says. “[Those settings are] loud, with a high amount of stimuli or stress or commotion, and they go to a quiet place, usually water in a quiet area. It’s calm. It’s peaceful.”
Water isn’t the only dangerous draw. When autism elopement happens, “they also go to the woods, they go to abandoned vehicles,” she says. “So any quiet thing is usually where they will head.”
A family’s loss
Beth Dilg, a mother in Maryland, lost her 7-year-old daughter, Savannah Martin, who was on the autism spectrum, to drowning in 2011. Ms. Dilg had been living in Oklahoma and raising her three children alone after separating from her husband. On a chilly February day, Savannah and her 2-year-old brother left their house after Ms. Dilg had asked her 11-year-old son to keep watch while she went into the bathroom for a few minutes.
When Ms. Dilg realized the two younger kids had left, she searched the property frantically. She shouted Savannah’s name repeatedly, but the child, who had limited language, didn’t come when called. “I feel like she knew what her name was,” Ms. Dilg says, “but it wasn’t like you’d call her name and she’d come to you.”
Ms. Dilg ran to a pond near her property after her 11-year-old son said that the two siblings were in the water. Ms. Dilg entered the water and grabbed her toddler, who had survived after having been kept afloat by his bicycle helmet. But when Ms. Dilg reached Savannah, she was already unresponsive. A neighbor helped pull the children out.
It can happen in any family. Even when a parent takes precautions, a child can slip out in a moment, perhaps while the parent is asleep or taking care of personal needs or if the child is at school or elsewhere.
“It’s unrealistic to say that you’d never take your eyes off your kid,” Ms. Dilg says.
She had tried to protect Savannah by starting her on swimming lessons, installing high locks on the doors, and trying to teach her about how to stay safe.
Still, children can be skillful in finding ways to escape, Ms. Dilg says. “These kids with autism are so smart. They may not be verbal, but they have this level of intelligence,” she says. “You always have to stay a step ahead of them.”
Ms. Dilg has been a longtime volunteer with Team HOPE, a peer support group with the National Center for Missing and Exploited Children. She offers emotional support to parents whose children are missing or who have died, including the parents of youngsters with autism who have drowned.
Teaching first responders
If a child on the autism spectrum goes missing, searching for them can be complicated by their condition. For instance, some children cannot speak or aren’t able to respond to searchers calling their name. The National Center for Missing and Exploited Children offers training to law enforcement and provides search protocols for first responders.
The center has drawn on expertise from Laurie Reyes, an officer with Maryland’s Montgomery County Police Department. In 2005, Ms. Reyes created a special unit within the department to focus on safety for people at risk for wandering. They have conditions that include autism/intellectual and developmental disabilities, as well as Alzheimer’s and other forms of dementia.
“We have a culture of awareness here,” Ms. Reyes says. All Montgomery County recruits and officers receive training in how to interact with those on the autism spectrum, who may not respond to police commands. Police also learn how to search, including immediately checking bodies of water. “We’ve had many times where we’ve located individuals in bodies of water,” Ms. Reyes says.
Don’t wait to call 911. When a child goes missing, time matters. Ms. Reyes advises families not to search on their own. “Call 911 right away,” she says.
Top safety tips to help prevent autism elopement
Use these tips to help keep kids on the autism spectrum safe and prevent drownings.
Secure your home. Use window and door alarms to alert you if a door or window becomes ajar. “Those door alarms are essential,” Ms. McIlwain says.
You can buy alarms online or get them free from the National Autism Association through its Big Red Safety Box program.
You can also buy portable door alarms for travel and arrange to have door alarms at your child’s school.
Ms. McIlwain advises securing the home with adequate locks and using baby monitors. Installing visual prompts, such as a stop sign on the door, might also cue a child not to leave.
Use personal identification. Ms. McIlwain says that children with autism must wear identification, such as a wristband, that includes their name, autism diagnosis, and the name and phone number of a contact person.
If children won’t wear a wristband, IDs on shoelaces are an option, she says. But parents should be aware that kids might leave without shoes or take them off before entering water.
Parents can also weigh the pros and cons of using tracking and locater devices, Ms. McIlwain says.
Identify triggers. “What’s going to make the child want to leave the setting? Is it noise? Is it a certain thing that they fear?” Ms. McIlwain says. “There’s always a reason.”
If parents can identify particular triggers, they can use calming techniques, for example, or provide headphones to counteract bothersome noises.
Teach safety skills, such as swimming lessons. Swimming lessons are important, Ms. McIlwain says. However, children with autism are often bothered by noise and commotion. So a regular swim class might not work for them.
Instead, Ms. McIlwain encourages parents to ask their local YMCA about special-needs swimming lessons or to search for such lessons online. What usually turns out to be best is to give the child a few private swimming lessons “with somebody who understands autism.”
For the child’s final lesson, they should swim fully clothed and with shoes on, Ms. McIlwain says. “A lot of our kids go straight into water fully clothed, and they just need to be able to be familiar with how that feels and the weight of that and be able to swim like that as well.”
If a child is drawn to water, discuss a scheduled time to go so that the youngster can wait, Ms. McIlwain says. “They can see that they’re going to get that water time. They’re going to be able to go to that place. They’re going to wait instead of trying to go on their own.”
Keep a close watch and team up. “When there is a family gathering or an outdoor barbecue, a lot of times, we all think, there are more adults here, so there are going to be more eyes on all the kids. And that always ends up being opposite, right?” Ms. McIlwain says.
Be specific about who is monitoring the child’s safety.
“We encourage parents to do the ‘Tag, you’re it’ game with one another. So you basically tag an adult who is responsible for keeping an eye on that child for a period of time so that there’s always supervision.”
Be prepared
There are a few things you can do now to be ready in case your child slips away. These measures may help find the child quickly.
Take photos today. Keep a full-length shot and a head shot of your child and store them electronically. If your child wanders away, you can immediately send the images to law enforcement to help them search.
Write a 911 script. Have this document ready in case your child wanders. It describes, among other things, points of interest that might draw your child, as well as locations of nearby bodies of water. By having it all written down, you’ll be able to share the information quickly with first responders. The Montgomery County Police Department has a “Wandering 911 Script” that you can download and use.
A version of this article first appeared on Webmd.com.
It happens quickly: A child on the autism spectrum bolts from supervision and disappears – an emergency called “autism elopement.” While any child can wander off, children on the autism spectrum face particular risks. These include the lure of water and the risk of drowning.
Some youngsters on the spectrum will follow this strong attraction to water and head for a nearby pond, river, or swimming pool. Such circumstances have made drowning a leading cause of death for these missing youths.
Autism elopement can happen any time. Summer can be especially dangerous. When the weather warms, the risk of drowning death rises, says Lori McIlwain, cofounder of the National Autism Association.
“The fatality risk is higher in May, June, July for that child to exit the setting unnoticed, especially if there’s an outdoor gathering and then they go directly to water,” Ms. McIlwain says. For instance, she says children can dart away during outdoor play, barbecues, gatherings, and other activities. Or they might wander off while vacationing near a beach or hotel pool.
Autism elopement
Many people don’t know about this risk, including some families with youngsters on the autism spectrum. The National Center for Missing and Exploited Children is working to change that – and find solutions.
About 12 years ago, “we started noticing a very disturbing trend that children with autism were going missing and they were having grave results,” says John Bischoff, vice president of the Center’s Missing Children Division.
The Center analyzed a decade of data on accidental deaths of children on the autism spectrum. Drowning was the #1 cause, accounting for 84% of those deaths.
In 2012, researchers reported on autism and wandering in the journal Pediatrics. They analyzed answers from about 1,000 families to an online survey on the topic. Parents who had children on the spectrum and children not on the spectrum responded. Nearly half of the parents said their child with autism had tried to wander off after age 4, and 26% had gone missing long enough to cause concern.
“Of those who went missing, 24% were in danger of drowning and 65% were in danger of traffic injury,” the researchers wrote. Children on the spectrum might also be drawn to traffic signs, highways, fire trucks, and trains.
In comparison, brothers and sisters of all ages who were not on the spectrum were much less likely to have wandered off.
Seeking a quiet place
It’s not entirely clear why children with autism are so drawn to water, Ms. McIlwain says. But there are some clues.
“What we see is that these children exit settings that are usually bothersome,” Ms. McIlwain says. “[Those settings are] loud, with a high amount of stimuli or stress or commotion, and they go to a quiet place, usually water in a quiet area. It’s calm. It’s peaceful.”
Water isn’t the only dangerous draw. When autism elopement happens, “they also go to the woods, they go to abandoned vehicles,” she says. “So any quiet thing is usually where they will head.”
A family’s loss
Beth Dilg, a mother in Maryland, lost her 7-year-old daughter, Savannah Martin, who was on the autism spectrum, to drowning in 2011. Ms. Dilg had been living in Oklahoma and raising her three children alone after separating from her husband. On a chilly February day, Savannah and her 2-year-old brother left their house after Ms. Dilg had asked her 11-year-old son to keep watch while she went into the bathroom for a few minutes.
When Ms. Dilg realized the two younger kids had left, she searched the property frantically. She shouted Savannah’s name repeatedly, but the child, who had limited language, didn’t come when called. “I feel like she knew what her name was,” Ms. Dilg says, “but it wasn’t like you’d call her name and she’d come to you.”
Ms. Dilg ran to a pond near her property after her 11-year-old son said that the two siblings were in the water. Ms. Dilg entered the water and grabbed her toddler, who had survived after having been kept afloat by his bicycle helmet. But when Ms. Dilg reached Savannah, she was already unresponsive. A neighbor helped pull the children out.
It can happen in any family. Even when a parent takes precautions, a child can slip out in a moment, perhaps while the parent is asleep or taking care of personal needs or if the child is at school or elsewhere.
“It’s unrealistic to say that you’d never take your eyes off your kid,” Ms. Dilg says.
She had tried to protect Savannah by starting her on swimming lessons, installing high locks on the doors, and trying to teach her about how to stay safe.
Still, children can be skillful in finding ways to escape, Ms. Dilg says. “These kids with autism are so smart. They may not be verbal, but they have this level of intelligence,” she says. “You always have to stay a step ahead of them.”
Ms. Dilg has been a longtime volunteer with Team HOPE, a peer support group with the National Center for Missing and Exploited Children. She offers emotional support to parents whose children are missing or who have died, including the parents of youngsters with autism who have drowned.
Teaching first responders
If a child on the autism spectrum goes missing, searching for them can be complicated by their condition. For instance, some children cannot speak or aren’t able to respond to searchers calling their name. The National Center for Missing and Exploited Children offers training to law enforcement and provides search protocols for first responders.
The center has drawn on expertise from Laurie Reyes, an officer with Maryland’s Montgomery County Police Department. In 2005, Ms. Reyes created a special unit within the department to focus on safety for people at risk for wandering. They have conditions that include autism/intellectual and developmental disabilities, as well as Alzheimer’s and other forms of dementia.
“We have a culture of awareness here,” Ms. Reyes says. All Montgomery County recruits and officers receive training in how to interact with those on the autism spectrum, who may not respond to police commands. Police also learn how to search, including immediately checking bodies of water. “We’ve had many times where we’ve located individuals in bodies of water,” Ms. Reyes says.
Don’t wait to call 911. When a child goes missing, time matters. Ms. Reyes advises families not to search on their own. “Call 911 right away,” she says.
Top safety tips to help prevent autism elopement
Use these tips to help keep kids on the autism spectrum safe and prevent drownings.
Secure your home. Use window and door alarms to alert you if a door or window becomes ajar. “Those door alarms are essential,” Ms. McIlwain says.
You can buy alarms online or get them free from the National Autism Association through its Big Red Safety Box program.
You can also buy portable door alarms for travel and arrange to have door alarms at your child’s school.
Ms. McIlwain advises securing the home with adequate locks and using baby monitors. Installing visual prompts, such as a stop sign on the door, might also cue a child not to leave.
Use personal identification. Ms. McIlwain says that children with autism must wear identification, such as a wristband, that includes their name, autism diagnosis, and the name and phone number of a contact person.
If children won’t wear a wristband, IDs on shoelaces are an option, she says. But parents should be aware that kids might leave without shoes or take them off before entering water.
Parents can also weigh the pros and cons of using tracking and locater devices, Ms. McIlwain says.
Identify triggers. “What’s going to make the child want to leave the setting? Is it noise? Is it a certain thing that they fear?” Ms. McIlwain says. “There’s always a reason.”
If parents can identify particular triggers, they can use calming techniques, for example, or provide headphones to counteract bothersome noises.
Teach safety skills, such as swimming lessons. Swimming lessons are important, Ms. McIlwain says. However, children with autism are often bothered by noise and commotion. So a regular swim class might not work for them.
Instead, Ms. McIlwain encourages parents to ask their local YMCA about special-needs swimming lessons or to search for such lessons online. What usually turns out to be best is to give the child a few private swimming lessons “with somebody who understands autism.”
For the child’s final lesson, they should swim fully clothed and with shoes on, Ms. McIlwain says. “A lot of our kids go straight into water fully clothed, and they just need to be able to be familiar with how that feels and the weight of that and be able to swim like that as well.”
If a child is drawn to water, discuss a scheduled time to go so that the youngster can wait, Ms. McIlwain says. “They can see that they’re going to get that water time. They’re going to be able to go to that place. They’re going to wait instead of trying to go on their own.”
Keep a close watch and team up. “When there is a family gathering or an outdoor barbecue, a lot of times, we all think, there are more adults here, so there are going to be more eyes on all the kids. And that always ends up being opposite, right?” Ms. McIlwain says.
Be specific about who is monitoring the child’s safety.
“We encourage parents to do the ‘Tag, you’re it’ game with one another. So you basically tag an adult who is responsible for keeping an eye on that child for a period of time so that there’s always supervision.”
Be prepared
There are a few things you can do now to be ready in case your child slips away. These measures may help find the child quickly.
Take photos today. Keep a full-length shot and a head shot of your child and store them electronically. If your child wanders away, you can immediately send the images to law enforcement to help them search.
Write a 911 script. Have this document ready in case your child wanders. It describes, among other things, points of interest that might draw your child, as well as locations of nearby bodies of water. By having it all written down, you’ll be able to share the information quickly with first responders. The Montgomery County Police Department has a “Wandering 911 Script” that you can download and use.
A version of this article first appeared on Webmd.com.
Blood-based assay may offer new way of diagnosing Parkinson’s disease
A novel blood-based assay could one day be used to diagnose Parkinson’s disease and possibly other chronic inflammatory conditions, according to investigators. In addition to being highly accurate, the assay, which detects changes in expression of cytochrome P450s, is faster and easier to perform than other Parkinson’s disease assays under investigation, reported lead author Kohei Ihara, PhD, of Kobe University, Japan, and colleagues.
“Effective diagnostic systems and biomarkers for patients without subjective motor symptoms have not yet been established,” the investigators wrote in Nature Scientific Reports. “Consequently, the poor diagnostic options for Parkinson’s disease delay the development of therapeutic approaches and medication. Therefore, the development of efficient diagnostic systems and biomarkers is crucial for overcoming Parkinson’s disease.”
According to Dr. Ihara and colleagues, various cytochrome P450 expression patterns and associated serum metabolites correlate with chronic conditions, making them possible markers of disease. To detect these changes, they developed the present assay. It relies upon recombinant P450s expressed on the surface of Escherichia coli. By mixing the E. coli with serum and Vivid, a fluorescent substrate, the investigators can measure “the inhibition rate of the Vivid decomposition reaction” that was driven by “serum metabolites associated with P450s,” revealing underlying expression and, if present, disease.
After some promising initial experiments with mouse models of ulcerative colitis and diabetes, Dr. Ihara and colleagues focused on a rat model of Parkinson’s disease. Evaluating inhibition rates associated with four P450s revealed area-under-the-curve (AUC) values of 0.814-0.914. Two of those P450s were also associated with progression of disease symptoms.
“Therefore, we concluded that the P450 inhibition assay could discriminate between Parkinson’s disease model rats and control rats,” the investigators wrote.
Next, the investigators tested the approach with a case-control study involving 20 patients with Parkinson’s disease and 20 healthy volunteers. Twelve P450s were analyzed, three of which revealed significant differences between patients with Parkinson’s disease and controls, with AUCs ranging from 0.740-0.775. Each of the three P450 enzymes also correlated significantly with stage of disease on the Hoehn & Yahr scale, although severity and frequency of symptoms were not reported.
To increase accuracy of the technique, the investigators developed a logistic regression model using two of the three P450s, generating an AUC of 0.910. Further testing showed that the P450 inhibition assay could distinguish between patients with Parkinson’s disease and Alzheimer’s disease, as well as other chronic inflammatory diseases.
“The P450 inhibition assay is easier to perform and is faster than other assays because this assay does not require pretreatment, such as purification of exosomes, and it involves a single enzymatic reaction,” the investigators wrote, suggesting that the assay may be suitable for real-world diagnosis.
‘Promising’ findings need replication
According to Douglas Galasko, MD, a neurologist and professor of neurosciences at UC San Diego Health, the reported accuracy of the assay “seems spectacular,” and the findings are “promising,” but they need to be replicated, “particularly in early-stage patients where the diagnosis [of Parkinson’s disease] is more difficult and important to make.” In practice, the assay would likely see greatest usage for “early diagnosis or diagnosis of unusual or challenging cases,” so accuracy testing needs to be conducted in this setting, he said.
Dr. Galasko, who was not involved in the study, predicted that liquid biopsy for detecting Parkinson’s disease is unlikely to hit the clinic floor anytime soon. “We’re not really close with blood-based biomarkers for Parkinson’s disease,” he said, “unlike the situation for Alzheimer’s disease, where there are several promising blood-based biomarkers.”
For diagnosing Parkinson’s disease, Dr. Galasko suggested that assays using skin biopsies to measure alpha-synuclein accumulation may be closer to approval.
The study was supported by JSPS KAKENHI Grant Number 20K20223 and the Sumitomo Electric Industries Group Corporate Social Responsibility Foundation. The investigators disclosed no conflicts of interest.
A novel blood-based assay could one day be used to diagnose Parkinson’s disease and possibly other chronic inflammatory conditions, according to investigators. In addition to being highly accurate, the assay, which detects changes in expression of cytochrome P450s, is faster and easier to perform than other Parkinson’s disease assays under investigation, reported lead author Kohei Ihara, PhD, of Kobe University, Japan, and colleagues.
“Effective diagnostic systems and biomarkers for patients without subjective motor symptoms have not yet been established,” the investigators wrote in Nature Scientific Reports. “Consequently, the poor diagnostic options for Parkinson’s disease delay the development of therapeutic approaches and medication. Therefore, the development of efficient diagnostic systems and biomarkers is crucial for overcoming Parkinson’s disease.”
According to Dr. Ihara and colleagues, various cytochrome P450 expression patterns and associated serum metabolites correlate with chronic conditions, making them possible markers of disease. To detect these changes, they developed the present assay. It relies upon recombinant P450s expressed on the surface of Escherichia coli. By mixing the E. coli with serum and Vivid, a fluorescent substrate, the investigators can measure “the inhibition rate of the Vivid decomposition reaction” that was driven by “serum metabolites associated with P450s,” revealing underlying expression and, if present, disease.
After some promising initial experiments with mouse models of ulcerative colitis and diabetes, Dr. Ihara and colleagues focused on a rat model of Parkinson’s disease. Evaluating inhibition rates associated with four P450s revealed area-under-the-curve (AUC) values of 0.814-0.914. Two of those P450s were also associated with progression of disease symptoms.
“Therefore, we concluded that the P450 inhibition assay could discriminate between Parkinson’s disease model rats and control rats,” the investigators wrote.
Next, the investigators tested the approach with a case-control study involving 20 patients with Parkinson’s disease and 20 healthy volunteers. Twelve P450s were analyzed, three of which revealed significant differences between patients with Parkinson’s disease and controls, with AUCs ranging from 0.740-0.775. Each of the three P450 enzymes also correlated significantly with stage of disease on the Hoehn & Yahr scale, although severity and frequency of symptoms were not reported.
To increase accuracy of the technique, the investigators developed a logistic regression model using two of the three P450s, generating an AUC of 0.910. Further testing showed that the P450 inhibition assay could distinguish between patients with Parkinson’s disease and Alzheimer’s disease, as well as other chronic inflammatory diseases.
“The P450 inhibition assay is easier to perform and is faster than other assays because this assay does not require pretreatment, such as purification of exosomes, and it involves a single enzymatic reaction,” the investigators wrote, suggesting that the assay may be suitable for real-world diagnosis.
‘Promising’ findings need replication
According to Douglas Galasko, MD, a neurologist and professor of neurosciences at UC San Diego Health, the reported accuracy of the assay “seems spectacular,” and the findings are “promising,” but they need to be replicated, “particularly in early-stage patients where the diagnosis [of Parkinson’s disease] is more difficult and important to make.” In practice, the assay would likely see greatest usage for “early diagnosis or diagnosis of unusual or challenging cases,” so accuracy testing needs to be conducted in this setting, he said.
Dr. Galasko, who was not involved in the study, predicted that liquid biopsy for detecting Parkinson’s disease is unlikely to hit the clinic floor anytime soon. “We’re not really close with blood-based biomarkers for Parkinson’s disease,” he said, “unlike the situation for Alzheimer’s disease, where there are several promising blood-based biomarkers.”
For diagnosing Parkinson’s disease, Dr. Galasko suggested that assays using skin biopsies to measure alpha-synuclein accumulation may be closer to approval.
The study was supported by JSPS KAKENHI Grant Number 20K20223 and the Sumitomo Electric Industries Group Corporate Social Responsibility Foundation. The investigators disclosed no conflicts of interest.
A novel blood-based assay could one day be used to diagnose Parkinson’s disease and possibly other chronic inflammatory conditions, according to investigators. In addition to being highly accurate, the assay, which detects changes in expression of cytochrome P450s, is faster and easier to perform than other Parkinson’s disease assays under investigation, reported lead author Kohei Ihara, PhD, of Kobe University, Japan, and colleagues.
“Effective diagnostic systems and biomarkers for patients without subjective motor symptoms have not yet been established,” the investigators wrote in Nature Scientific Reports. “Consequently, the poor diagnostic options for Parkinson’s disease delay the development of therapeutic approaches and medication. Therefore, the development of efficient diagnostic systems and biomarkers is crucial for overcoming Parkinson’s disease.”
According to Dr. Ihara and colleagues, various cytochrome P450 expression patterns and associated serum metabolites correlate with chronic conditions, making them possible markers of disease. To detect these changes, they developed the present assay. It relies upon recombinant P450s expressed on the surface of Escherichia coli. By mixing the E. coli with serum and Vivid, a fluorescent substrate, the investigators can measure “the inhibition rate of the Vivid decomposition reaction” that was driven by “serum metabolites associated with P450s,” revealing underlying expression and, if present, disease.
After some promising initial experiments with mouse models of ulcerative colitis and diabetes, Dr. Ihara and colleagues focused on a rat model of Parkinson’s disease. Evaluating inhibition rates associated with four P450s revealed area-under-the-curve (AUC) values of 0.814-0.914. Two of those P450s were also associated with progression of disease symptoms.
“Therefore, we concluded that the P450 inhibition assay could discriminate between Parkinson’s disease model rats and control rats,” the investigators wrote.
Next, the investigators tested the approach with a case-control study involving 20 patients with Parkinson’s disease and 20 healthy volunteers. Twelve P450s were analyzed, three of which revealed significant differences between patients with Parkinson’s disease and controls, with AUCs ranging from 0.740-0.775. Each of the three P450 enzymes also correlated significantly with stage of disease on the Hoehn & Yahr scale, although severity and frequency of symptoms were not reported.
To increase accuracy of the technique, the investigators developed a logistic regression model using two of the three P450s, generating an AUC of 0.910. Further testing showed that the P450 inhibition assay could distinguish between patients with Parkinson’s disease and Alzheimer’s disease, as well as other chronic inflammatory diseases.
“The P450 inhibition assay is easier to perform and is faster than other assays because this assay does not require pretreatment, such as purification of exosomes, and it involves a single enzymatic reaction,” the investigators wrote, suggesting that the assay may be suitable for real-world diagnosis.
‘Promising’ findings need replication
According to Douglas Galasko, MD, a neurologist and professor of neurosciences at UC San Diego Health, the reported accuracy of the assay “seems spectacular,” and the findings are “promising,” but they need to be replicated, “particularly in early-stage patients where the diagnosis [of Parkinson’s disease] is more difficult and important to make.” In practice, the assay would likely see greatest usage for “early diagnosis or diagnosis of unusual or challenging cases,” so accuracy testing needs to be conducted in this setting, he said.
Dr. Galasko, who was not involved in the study, predicted that liquid biopsy for detecting Parkinson’s disease is unlikely to hit the clinic floor anytime soon. “We’re not really close with blood-based biomarkers for Parkinson’s disease,” he said, “unlike the situation for Alzheimer’s disease, where there are several promising blood-based biomarkers.”
For diagnosing Parkinson’s disease, Dr. Galasko suggested that assays using skin biopsies to measure alpha-synuclein accumulation may be closer to approval.
The study was supported by JSPS KAKENHI Grant Number 20K20223 and the Sumitomo Electric Industries Group Corporate Social Responsibility Foundation. The investigators disclosed no conflicts of interest.
FROM NATURE SCIENTIFIC REPORTS
Can lasers be used to measure nerve sensitivity in the skin?
SAN DIEGO – In a 2006 report of complications from laser dermatologic surgery, one of the authors, Dieter Manstein, MD, PhD, who had subjected his forearm to treatment with a fractional laser skin resurfacing prototype device, was included as 1 of the 19 featured cases.
Dr. Manstein, of the Cutaneous Biology Research Center in the department of dermatology at Massachusetts General Hospital, Boston, was exposed to three test spots in the evaluation of the effects of different microscopic thermal zone densities for the prototype device, emitting at 1,450 nm and an energy per MTZ of 3 mJ.
Two years later, hypopigmentation persisted at the test site treated with the highest MTZ density, while two other sites treated with the lower MTZ densities did not show any dyspigmentation. But he noticed something else during the experiment: He felt minimal to no pain as each test site was being treated.
“It took 7 minutes without any cooling or anesthesia,” Dr. Manstein recalled at the annual meeting of the American Society for Laser Medicine and Surgery. “It was not completely painless, but each time the laser was applied, sometimes I felt a little prick, sometimes I felt nothing.” Essentially, he added, “we created cell injury with a focused laser beam without anesthesia,” but this could also indicate that if skin is treated with a fractional laser very slowly, anesthesia is not needed. “Current devices are meant to treat very quickly, but if we [treat] slowly, maybe you could remove lesions painlessly without anesthesia.”
The observation from that experiment also led Dr. Manstein and colleagues to wonder: Could a focused laser beam pattern be used to assess cutaneous innervation? If so, they postulated, perhaps it could be used to not only assess nerve sensitivity of candidates for dermatologic surgery, but as a tool to help diagnose small fiber neuropathies such as diabetic neuropathy, and neuropathies in patients with HIV and sarcoidosis.
The current gold standard for making these diagnoses involves a skin biopsy, immunohistochemical analysis, and nerve fiber quantification, which is not widely available. It also requires strict histologic processing and nerve counting rules. Confocal microscopy of nerve fibers in the cornea is another approach, but is very difficult to perform, “so it would be nice if there was a simple way” to determine nerve fiber density in the skin using a focused laser beam, Dr. Manstein said.
With help from Payal Patel, MD, a dermatology research fellow at MGH, records each subject’s perception of a stimulus, and maps the areas of stimulus response. Current diameters being studied range from 0.076-1.15 mm and depths less than 0.71 mm. “We can focus the laser beam, preset the beam diameter, and very slowly, in a controlled manner, make a rectangular pattern, and after each time, inquire if the subject felt the pulse or not,” Dr. Manstein explained.
“This laser could become a new method for diagnosing nerve fiber neuropathies. If this works well, I think we can miniaturize the device,” he added.
Dr. Manstein disclosed that he is a consultant for Blossom Innovations, R2 Dermatology, and AVAVA. He is also a member of the advisory board for Blossom Innovations.
SAN DIEGO – In a 2006 report of complications from laser dermatologic surgery, one of the authors, Dieter Manstein, MD, PhD, who had subjected his forearm to treatment with a fractional laser skin resurfacing prototype device, was included as 1 of the 19 featured cases.
Dr. Manstein, of the Cutaneous Biology Research Center in the department of dermatology at Massachusetts General Hospital, Boston, was exposed to three test spots in the evaluation of the effects of different microscopic thermal zone densities for the prototype device, emitting at 1,450 nm and an energy per MTZ of 3 mJ.
Two years later, hypopigmentation persisted at the test site treated with the highest MTZ density, while two other sites treated with the lower MTZ densities did not show any dyspigmentation. But he noticed something else during the experiment: He felt minimal to no pain as each test site was being treated.
“It took 7 minutes without any cooling or anesthesia,” Dr. Manstein recalled at the annual meeting of the American Society for Laser Medicine and Surgery. “It was not completely painless, but each time the laser was applied, sometimes I felt a little prick, sometimes I felt nothing.” Essentially, he added, “we created cell injury with a focused laser beam without anesthesia,” but this could also indicate that if skin is treated with a fractional laser very slowly, anesthesia is not needed. “Current devices are meant to treat very quickly, but if we [treat] slowly, maybe you could remove lesions painlessly without anesthesia.”
The observation from that experiment also led Dr. Manstein and colleagues to wonder: Could a focused laser beam pattern be used to assess cutaneous innervation? If so, they postulated, perhaps it could be used to not only assess nerve sensitivity of candidates for dermatologic surgery, but as a tool to help diagnose small fiber neuropathies such as diabetic neuropathy, and neuropathies in patients with HIV and sarcoidosis.
The current gold standard for making these diagnoses involves a skin biopsy, immunohistochemical analysis, and nerve fiber quantification, which is not widely available. It also requires strict histologic processing and nerve counting rules. Confocal microscopy of nerve fibers in the cornea is another approach, but is very difficult to perform, “so it would be nice if there was a simple way” to determine nerve fiber density in the skin using a focused laser beam, Dr. Manstein said.
With help from Payal Patel, MD, a dermatology research fellow at MGH, records each subject’s perception of a stimulus, and maps the areas of stimulus response. Current diameters being studied range from 0.076-1.15 mm and depths less than 0.71 mm. “We can focus the laser beam, preset the beam diameter, and very slowly, in a controlled manner, make a rectangular pattern, and after each time, inquire if the subject felt the pulse or not,” Dr. Manstein explained.
“This laser could become a new method for diagnosing nerve fiber neuropathies. If this works well, I think we can miniaturize the device,” he added.
Dr. Manstein disclosed that he is a consultant for Blossom Innovations, R2 Dermatology, and AVAVA. He is also a member of the advisory board for Blossom Innovations.
SAN DIEGO – In a 2006 report of complications from laser dermatologic surgery, one of the authors, Dieter Manstein, MD, PhD, who had subjected his forearm to treatment with a fractional laser skin resurfacing prototype device, was included as 1 of the 19 featured cases.
Dr. Manstein, of the Cutaneous Biology Research Center in the department of dermatology at Massachusetts General Hospital, Boston, was exposed to three test spots in the evaluation of the effects of different microscopic thermal zone densities for the prototype device, emitting at 1,450 nm and an energy per MTZ of 3 mJ.
Two years later, hypopigmentation persisted at the test site treated with the highest MTZ density, while two other sites treated with the lower MTZ densities did not show any dyspigmentation. But he noticed something else during the experiment: He felt minimal to no pain as each test site was being treated.
“It took 7 minutes without any cooling or anesthesia,” Dr. Manstein recalled at the annual meeting of the American Society for Laser Medicine and Surgery. “It was not completely painless, but each time the laser was applied, sometimes I felt a little prick, sometimes I felt nothing.” Essentially, he added, “we created cell injury with a focused laser beam without anesthesia,” but this could also indicate that if skin is treated with a fractional laser very slowly, anesthesia is not needed. “Current devices are meant to treat very quickly, but if we [treat] slowly, maybe you could remove lesions painlessly without anesthesia.”
The observation from that experiment also led Dr. Manstein and colleagues to wonder: Could a focused laser beam pattern be used to assess cutaneous innervation? If so, they postulated, perhaps it could be used to not only assess nerve sensitivity of candidates for dermatologic surgery, but as a tool to help diagnose small fiber neuropathies such as diabetic neuropathy, and neuropathies in patients with HIV and sarcoidosis.
The current gold standard for making these diagnoses involves a skin biopsy, immunohistochemical analysis, and nerve fiber quantification, which is not widely available. It also requires strict histologic processing and nerve counting rules. Confocal microscopy of nerve fibers in the cornea is another approach, but is very difficult to perform, “so it would be nice if there was a simple way” to determine nerve fiber density in the skin using a focused laser beam, Dr. Manstein said.
With help from Payal Patel, MD, a dermatology research fellow at MGH, records each subject’s perception of a stimulus, and maps the areas of stimulus response. Current diameters being studied range from 0.076-1.15 mm and depths less than 0.71 mm. “We can focus the laser beam, preset the beam diameter, and very slowly, in a controlled manner, make a rectangular pattern, and after each time, inquire if the subject felt the pulse or not,” Dr. Manstein explained.
“This laser could become a new method for diagnosing nerve fiber neuropathies. If this works well, I think we can miniaturize the device,” he added.
Dr. Manstein disclosed that he is a consultant for Blossom Innovations, R2 Dermatology, and AVAVA. He is also a member of the advisory board for Blossom Innovations.
AT ASLMS 2022
Hearing, vision loss combo a colossal risk for cognitive decline
The combination of hearing loss and vision loss is linked to an eightfold increased risk of cognitive impairment, new research shows.
Investigators analyzed data on more than 5 million U.S. seniors. Adjusted results show that participants with hearing impairment alone had more than twice the odds of also having cognitive impairment, while those with vision impairment alone had more than triple the odds of cognitive impairment.
However, those with dual sensory impairment (DSI) had an eightfold higher risk for cognitive impairment.
In addition, half of the participants with DSI also had cognitive impairment. Of those with cognitive impairment, 16% had DSI, compared with only about 2% of their peers without cognitive impairment.
“The findings of the present study may inform interventions that can support older people with concurrent sensory impairment and cognitive impairment,” said lead author Esme Fuller-Thomson, PhD, professor, Factor-Inwentash Faculty of Social Work, University of Toronto.
“Special attention, in particular, should be given to those aged 65-74 who have serious hearing and/or vision impairment [because], if the relationship with dementia is found to be causal, such interventions can potentially mitigate the development of cognitive impairment,” said Dr. Fuller-Thomson, who is also director of the Institute for Life Course and Aging and a professor in the department of family and community medicine and faculty of nursing, all at the University of Toronto.
The findings were published online in the Journal of Alzheimer’s Disease Reports.
Sensory isolation
Hearing and vision impairment increase with age; it is estimated that one-third of U.S. adults between the ages of 65 and 74 experience hearing loss, and 4% experience vision impairment, the investigators note.
“The link between dual hearing loss and seeing loss and mental health problems such as depression and social isolation have been well researched, but we were very interested in the link between dual sensory loss and cognitive problems,” Dr. Fuller-Thomson said.
Additionally, “there have been several studies in the past decade linking hearing loss to dementia and cognitive decline, but less attention has been paid to cognitive problems among those with DSI, despite this group being particularly isolated,” she said. Existing research into DSI suggests an association with cognitive decline; the current investigators sought to expand on this previous work.
To do so, they used merged data from 10 consecutive waves from 2008 to 2017 of the American Community Survey (ACS), which was conducted by the U.S. Census Bureau. The ACS is a nationally representative sample of 3.5 million randomly selected U.S. addresses and includes community-dwelling adults and those residing in institutional settings.
Participants aged 65 or older (n = 5,405,135; 56.4% women) were asked yes/no questions regarding serious cognitive impairment, hearing impairment, and vision impairment. A proxy, such as a family member or nursing home staff member, provided answers for individuals not capable of self-report.
Potential confounding variables included age, race/ethnicity, sex, education, and household income.
Potential mechanisms
Results showed that, among those with cognitive impairment, there was a higher prevalence of hearing impairment, vision impairment, and DSI than among their peers without cognitive impairment; in addition, a lower percentage of these persons had no sensory impairment (P < .001).
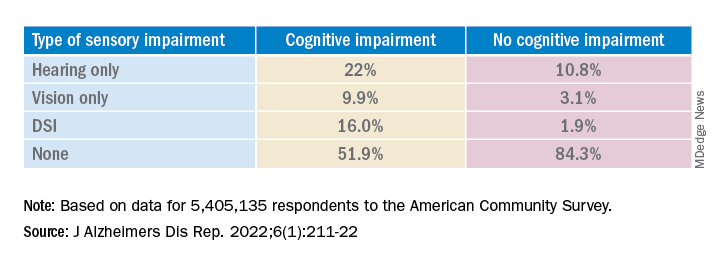
The prevalence of DSI climbed with age, from 1.5% for respondents aged 65-74 years to 2.6% for those aged 75-84 and to 10.8% in those 85 years and older.
Individuals with higher levels of poverty also had higher levels of DSI. Among those who had not completed high school, the prevalence of DSI was higher, compared with high school or university graduates (6.3% vs. 3.1% and 1.85, respectively).
After controlling for age, race, education, and income, the researchers found “substantially” higher odds of cognitive impairment in those with vs. those without sensory impairments.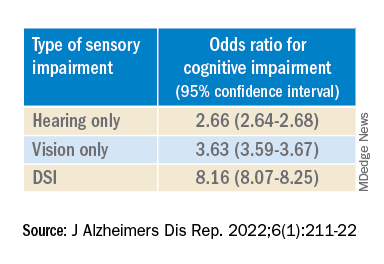
“The magnitude of the odds of cognitive impairment by sensory impairment was greatest for the youngest cohort (age 65-74) and lowest for the oldest cohort (age 85+),” the investigators wrote. Among participants in the youngest cohort, there was a “dose-response relationship” for those with hearing impairment only, visual impairment only, and DSI.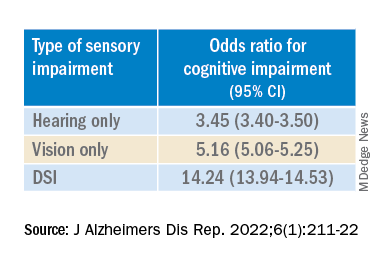
Because the study was observational, it “does not provide sufficient information to determine the reasons behind the observed link between sensory loss and cognitive problems,” Dr. Fuller-Thomson said. However, there are “several potential causal mechanisms [that] warrant future research.”
The “sensory deprivation hypothesis” suggests that DSI could cause cognitive deterioration because of decreased auditory and visual input. The “resource allocation hypothesis” posits that hearing- or vision-impaired older adults “may use more cognitive resources to accommodate for sensory deficits, allocating fewer cognitive resources for higher-order memory processes,” the researchers wrote. Hearing impairment “may also lead to social disengagement among older adults, hastening cognitive decline due to isolation and lack of stimulation,” they added.
Reverse causality is also possible. In the “cognitive load on perception” hypothesis, cognitive decline may lead to declines in hearing and vision because of “decreased resources for sensory processing.”
In addition, the association may be noncausal. “The ‘common cause hypothesis’ theorizes that sensory impairment and cognitive impairment may be due to shared age-related degeneration of the central nervous system ... or frailty,” Dr. Fuller-Thomson said.
Parallel findings
The results are similar to those from a study conducted by Phillip Hwang, PhD, of the department of anatomy and neurobiology, Boston University, and colleagues that was published online in JAMA Network Open.
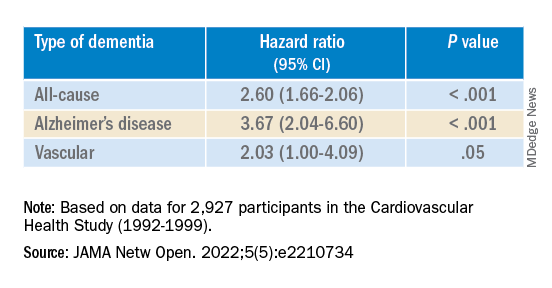
They analyzed data on 8 years of follow-up of 2,927 participants in the Cardiovascular Health Study (mean age, 74.6 years; 58.2% women).
Compared with no sensory impairment, DSI was associated with increased risk for all-cause dementia and Alzheimer’s disease, but not with vascular dementia.
“Future work in health care guidelines could consider incorporating screening of sensory impairment in older adults as part of risk assessment for dementia,” Nicholas Reed, AuD, and Esther Oh, MD, PhD, both of Johns Hopkins University, Baltimore, wrote in an accompanying editorial.
Accurate testing
Commenting on both studies, Heather Whitson, MD, professor of medicine (geriatrics) and ophthalmology and director at the Duke University Center for the Study of Aging and Human Development, Durham, N.C., said both “add further strength to the evidence base, which has really converged in the last few years to support that there is a link between sensory health and cognitive health.”
However, “we still don’t know whether hearing/vision loss causes cognitive decline, though there are plausible ways that sensory loss could affect cognitive abilities like memory, language, and executive function,” she said
Dr. Whitson, who was not involved with the research, is also codirector of the Duke/University of North Carolina Alzheimer’s Disease Research Center at Duke University, Durham, N.C., and the Durham VA Medical Center.
“The big question is whether we can improve patients’ cognitive performance by treating or accommodating their sensory impairments,” she said. “If safe and feasible things like hearing aids or cataract surgery improve cognitive health, even a little bit, it would be a huge benefit to society, because sensory loss is very common, and there are many treatment options,” Dr. Whitson added.
Dr. Fuller-Thomson emphasized that practitioners should “consider the full impact of sensory impairment on cognitive testing methods, as both auditory and visual testing methods may fail to take hearing and vision impairment into account.”
Thus, “when performing cognitive tests on older adults with sensory impairments, practitioners should ensure they are communicating audibly and/or using visual speech cues for hearing-impaired individuals, eliminating items from cognitive tests that rely on vision for those who are visually impaired, and using physical cues for individuals with hearing or dual sensory impairment, as this can help increase the accuracy of testing and prevent confounding,” she said.
The study by Fuller-Thomson et al. was funded by a donation from Janis Rotman. Its investigators have reported no relevant financial relationships. The study by Hwang et al. was funded by contracts from the National Heart, Lung, and Blood Institute, the National Institute of Neurological Disorders and Stroke, and the National Institute on Aging. Dr. Hwang reports no relevant financial relationships. The other investigators’ disclosures are listed in the original article. Dr. Reed received grants from the National Institute on Aging during the conduct of the study and has served on the advisory board of Neosensory outside the submitted work. Dr. Oh and Dr. Whitson report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The combination of hearing loss and vision loss is linked to an eightfold increased risk of cognitive impairment, new research shows.
Investigators analyzed data on more than 5 million U.S. seniors. Adjusted results show that participants with hearing impairment alone had more than twice the odds of also having cognitive impairment, while those with vision impairment alone had more than triple the odds of cognitive impairment.
However, those with dual sensory impairment (DSI) had an eightfold higher risk for cognitive impairment.
In addition, half of the participants with DSI also had cognitive impairment. Of those with cognitive impairment, 16% had DSI, compared with only about 2% of their peers without cognitive impairment.
“The findings of the present study may inform interventions that can support older people with concurrent sensory impairment and cognitive impairment,” said lead author Esme Fuller-Thomson, PhD, professor, Factor-Inwentash Faculty of Social Work, University of Toronto.
“Special attention, in particular, should be given to those aged 65-74 who have serious hearing and/or vision impairment [because], if the relationship with dementia is found to be causal, such interventions can potentially mitigate the development of cognitive impairment,” said Dr. Fuller-Thomson, who is also director of the Institute for Life Course and Aging and a professor in the department of family and community medicine and faculty of nursing, all at the University of Toronto.
The findings were published online in the Journal of Alzheimer’s Disease Reports.
Sensory isolation
Hearing and vision impairment increase with age; it is estimated that one-third of U.S. adults between the ages of 65 and 74 experience hearing loss, and 4% experience vision impairment, the investigators note.
“The link between dual hearing loss and seeing loss and mental health problems such as depression and social isolation have been well researched, but we were very interested in the link between dual sensory loss and cognitive problems,” Dr. Fuller-Thomson said.
Additionally, “there have been several studies in the past decade linking hearing loss to dementia and cognitive decline, but less attention has been paid to cognitive problems among those with DSI, despite this group being particularly isolated,” she said. Existing research into DSI suggests an association with cognitive decline; the current investigators sought to expand on this previous work.
To do so, they used merged data from 10 consecutive waves from 2008 to 2017 of the American Community Survey (ACS), which was conducted by the U.S. Census Bureau. The ACS is a nationally representative sample of 3.5 million randomly selected U.S. addresses and includes community-dwelling adults and those residing in institutional settings.
Participants aged 65 or older (n = 5,405,135; 56.4% women) were asked yes/no questions regarding serious cognitive impairment, hearing impairment, and vision impairment. A proxy, such as a family member or nursing home staff member, provided answers for individuals not capable of self-report.
Potential confounding variables included age, race/ethnicity, sex, education, and household income.
Potential mechanisms
Results showed that, among those with cognitive impairment, there was a higher prevalence of hearing impairment, vision impairment, and DSI than among their peers without cognitive impairment; in addition, a lower percentage of these persons had no sensory impairment (P < .001).

The prevalence of DSI climbed with age, from 1.5% for respondents aged 65-74 years to 2.6% for those aged 75-84 and to 10.8% in those 85 years and older.
Individuals with higher levels of poverty also had higher levels of DSI. Among those who had not completed high school, the prevalence of DSI was higher, compared with high school or university graduates (6.3% vs. 3.1% and 1.85, respectively).
After controlling for age, race, education, and income, the researchers found “substantially” higher odds of cognitive impairment in those with vs. those without sensory impairments.
“The magnitude of the odds of cognitive impairment by sensory impairment was greatest for the youngest cohort (age 65-74) and lowest for the oldest cohort (age 85+),” the investigators wrote. Among participants in the youngest cohort, there was a “dose-response relationship” for those with hearing impairment only, visual impairment only, and DSI.
Because the study was observational, it “does not provide sufficient information to determine the reasons behind the observed link between sensory loss and cognitive problems,” Dr. Fuller-Thomson said. However, there are “several potential causal mechanisms [that] warrant future research.”
The “sensory deprivation hypothesis” suggests that DSI could cause cognitive deterioration because of decreased auditory and visual input. The “resource allocation hypothesis” posits that hearing- or vision-impaired older adults “may use more cognitive resources to accommodate for sensory deficits, allocating fewer cognitive resources for higher-order memory processes,” the researchers wrote. Hearing impairment “may also lead to social disengagement among older adults, hastening cognitive decline due to isolation and lack of stimulation,” they added.
Reverse causality is also possible. In the “cognitive load on perception” hypothesis, cognitive decline may lead to declines in hearing and vision because of “decreased resources for sensory processing.”
In addition, the association may be noncausal. “The ‘common cause hypothesis’ theorizes that sensory impairment and cognitive impairment may be due to shared age-related degeneration of the central nervous system ... or frailty,” Dr. Fuller-Thomson said.
Parallel findings
The results are similar to those from a study conducted by Phillip Hwang, PhD, of the department of anatomy and neurobiology, Boston University, and colleagues that was published online in JAMA Network Open.

They analyzed data on 8 years of follow-up of 2,927 participants in the Cardiovascular Health Study (mean age, 74.6 years; 58.2% women).
Compared with no sensory impairment, DSI was associated with increased risk for all-cause dementia and Alzheimer’s disease, but not with vascular dementia.
“Future work in health care guidelines could consider incorporating screening of sensory impairment in older adults as part of risk assessment for dementia,” Nicholas Reed, AuD, and Esther Oh, MD, PhD, both of Johns Hopkins University, Baltimore, wrote in an accompanying editorial.
Accurate testing
Commenting on both studies, Heather Whitson, MD, professor of medicine (geriatrics) and ophthalmology and director at the Duke University Center for the Study of Aging and Human Development, Durham, N.C., said both “add further strength to the evidence base, which has really converged in the last few years to support that there is a link between sensory health and cognitive health.”
However, “we still don’t know whether hearing/vision loss causes cognitive decline, though there are plausible ways that sensory loss could affect cognitive abilities like memory, language, and executive function,” she said
Dr. Whitson, who was not involved with the research, is also codirector of the Duke/University of North Carolina Alzheimer’s Disease Research Center at Duke University, Durham, N.C., and the Durham VA Medical Center.
“The big question is whether we can improve patients’ cognitive performance by treating or accommodating their sensory impairments,” she said. “If safe and feasible things like hearing aids or cataract surgery improve cognitive health, even a little bit, it would be a huge benefit to society, because sensory loss is very common, and there are many treatment options,” Dr. Whitson added.
Dr. Fuller-Thomson emphasized that practitioners should “consider the full impact of sensory impairment on cognitive testing methods, as both auditory and visual testing methods may fail to take hearing and vision impairment into account.”
Thus, “when performing cognitive tests on older adults with sensory impairments, practitioners should ensure they are communicating audibly and/or using visual speech cues for hearing-impaired individuals, eliminating items from cognitive tests that rely on vision for those who are visually impaired, and using physical cues for individuals with hearing or dual sensory impairment, as this can help increase the accuracy of testing and prevent confounding,” she said.
The study by Fuller-Thomson et al. was funded by a donation from Janis Rotman. Its investigators have reported no relevant financial relationships. The study by Hwang et al. was funded by contracts from the National Heart, Lung, and Blood Institute, the National Institute of Neurological Disorders and Stroke, and the National Institute on Aging. Dr. Hwang reports no relevant financial relationships. The other investigators’ disclosures are listed in the original article. Dr. Reed received grants from the National Institute on Aging during the conduct of the study and has served on the advisory board of Neosensory outside the submitted work. Dr. Oh and Dr. Whitson report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The combination of hearing loss and vision loss is linked to an eightfold increased risk of cognitive impairment, new research shows.
Investigators analyzed data on more than 5 million U.S. seniors. Adjusted results show that participants with hearing impairment alone had more than twice the odds of also having cognitive impairment, while those with vision impairment alone had more than triple the odds of cognitive impairment.
However, those with dual sensory impairment (DSI) had an eightfold higher risk for cognitive impairment.
In addition, half of the participants with DSI also had cognitive impairment. Of those with cognitive impairment, 16% had DSI, compared with only about 2% of their peers without cognitive impairment.
“The findings of the present study may inform interventions that can support older people with concurrent sensory impairment and cognitive impairment,” said lead author Esme Fuller-Thomson, PhD, professor, Factor-Inwentash Faculty of Social Work, University of Toronto.
“Special attention, in particular, should be given to those aged 65-74 who have serious hearing and/or vision impairment [because], if the relationship with dementia is found to be causal, such interventions can potentially mitigate the development of cognitive impairment,” said Dr. Fuller-Thomson, who is also director of the Institute for Life Course and Aging and a professor in the department of family and community medicine and faculty of nursing, all at the University of Toronto.
The findings were published online in the Journal of Alzheimer’s Disease Reports.
Sensory isolation
Hearing and vision impairment increase with age; it is estimated that one-third of U.S. adults between the ages of 65 and 74 experience hearing loss, and 4% experience vision impairment, the investigators note.
“The link between dual hearing loss and seeing loss and mental health problems such as depression and social isolation have been well researched, but we were very interested in the link between dual sensory loss and cognitive problems,” Dr. Fuller-Thomson said.
Additionally, “there have been several studies in the past decade linking hearing loss to dementia and cognitive decline, but less attention has been paid to cognitive problems among those with DSI, despite this group being particularly isolated,” she said. Existing research into DSI suggests an association with cognitive decline; the current investigators sought to expand on this previous work.
To do so, they used merged data from 10 consecutive waves from 2008 to 2017 of the American Community Survey (ACS), which was conducted by the U.S. Census Bureau. The ACS is a nationally representative sample of 3.5 million randomly selected U.S. addresses and includes community-dwelling adults and those residing in institutional settings.
Participants aged 65 or older (n = 5,405,135; 56.4% women) were asked yes/no questions regarding serious cognitive impairment, hearing impairment, and vision impairment. A proxy, such as a family member or nursing home staff member, provided answers for individuals not capable of self-report.
Potential confounding variables included age, race/ethnicity, sex, education, and household income.
Potential mechanisms
Results showed that, among those with cognitive impairment, there was a higher prevalence of hearing impairment, vision impairment, and DSI than among their peers without cognitive impairment; in addition, a lower percentage of these persons had no sensory impairment (P < .001).

The prevalence of DSI climbed with age, from 1.5% for respondents aged 65-74 years to 2.6% for those aged 75-84 and to 10.8% in those 85 years and older.
Individuals with higher levels of poverty also had higher levels of DSI. Among those who had not completed high school, the prevalence of DSI was higher, compared with high school or university graduates (6.3% vs. 3.1% and 1.85, respectively).
After controlling for age, race, education, and income, the researchers found “substantially” higher odds of cognitive impairment in those with vs. those without sensory impairments.
“The magnitude of the odds of cognitive impairment by sensory impairment was greatest for the youngest cohort (age 65-74) and lowest for the oldest cohort (age 85+),” the investigators wrote. Among participants in the youngest cohort, there was a “dose-response relationship” for those with hearing impairment only, visual impairment only, and DSI.
Because the study was observational, it “does not provide sufficient information to determine the reasons behind the observed link between sensory loss and cognitive problems,” Dr. Fuller-Thomson said. However, there are “several potential causal mechanisms [that] warrant future research.”
The “sensory deprivation hypothesis” suggests that DSI could cause cognitive deterioration because of decreased auditory and visual input. The “resource allocation hypothesis” posits that hearing- or vision-impaired older adults “may use more cognitive resources to accommodate for sensory deficits, allocating fewer cognitive resources for higher-order memory processes,” the researchers wrote. Hearing impairment “may also lead to social disengagement among older adults, hastening cognitive decline due to isolation and lack of stimulation,” they added.
Reverse causality is also possible. In the “cognitive load on perception” hypothesis, cognitive decline may lead to declines in hearing and vision because of “decreased resources for sensory processing.”
In addition, the association may be noncausal. “The ‘common cause hypothesis’ theorizes that sensory impairment and cognitive impairment may be due to shared age-related degeneration of the central nervous system ... or frailty,” Dr. Fuller-Thomson said.
Parallel findings
The results are similar to those from a study conducted by Phillip Hwang, PhD, of the department of anatomy and neurobiology, Boston University, and colleagues that was published online in JAMA Network Open.

They analyzed data on 8 years of follow-up of 2,927 participants in the Cardiovascular Health Study (mean age, 74.6 years; 58.2% women).
Compared with no sensory impairment, DSI was associated with increased risk for all-cause dementia and Alzheimer’s disease, but not with vascular dementia.
“Future work in health care guidelines could consider incorporating screening of sensory impairment in older adults as part of risk assessment for dementia,” Nicholas Reed, AuD, and Esther Oh, MD, PhD, both of Johns Hopkins University, Baltimore, wrote in an accompanying editorial.
Accurate testing
Commenting on both studies, Heather Whitson, MD, professor of medicine (geriatrics) and ophthalmology and director at the Duke University Center for the Study of Aging and Human Development, Durham, N.C., said both “add further strength to the evidence base, which has really converged in the last few years to support that there is a link between sensory health and cognitive health.”
However, “we still don’t know whether hearing/vision loss causes cognitive decline, though there are plausible ways that sensory loss could affect cognitive abilities like memory, language, and executive function,” she said
Dr. Whitson, who was not involved with the research, is also codirector of the Duke/University of North Carolina Alzheimer’s Disease Research Center at Duke University, Durham, N.C., and the Durham VA Medical Center.
“The big question is whether we can improve patients’ cognitive performance by treating or accommodating their sensory impairments,” she said. “If safe and feasible things like hearing aids or cataract surgery improve cognitive health, even a little bit, it would be a huge benefit to society, because sensory loss is very common, and there are many treatment options,” Dr. Whitson added.
Dr. Fuller-Thomson emphasized that practitioners should “consider the full impact of sensory impairment on cognitive testing methods, as both auditory and visual testing methods may fail to take hearing and vision impairment into account.”
Thus, “when performing cognitive tests on older adults with sensory impairments, practitioners should ensure they are communicating audibly and/or using visual speech cues for hearing-impaired individuals, eliminating items from cognitive tests that rely on vision for those who are visually impaired, and using physical cues for individuals with hearing or dual sensory impairment, as this can help increase the accuracy of testing and prevent confounding,” she said.
The study by Fuller-Thomson et al. was funded by a donation from Janis Rotman. Its investigators have reported no relevant financial relationships. The study by Hwang et al. was funded by contracts from the National Heart, Lung, and Blood Institute, the National Institute of Neurological Disorders and Stroke, and the National Institute on Aging. Dr. Hwang reports no relevant financial relationships. The other investigators’ disclosures are listed in the original article. Dr. Reed received grants from the National Institute on Aging during the conduct of the study and has served on the advisory board of Neosensory outside the submitted work. Dr. Oh and Dr. Whitson report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF ALZHEIMER’S DISEASE REPORTS
Long COVID neuropsychiatric deficits greater than expected
NEW ORLEANS – , adding to mounting evidence of the significant toll the chronic condition can have on mental health.
“Many clinicians have observed the symptoms we describe in this study, however this report is among the first which identify the specific deficits using neuropsychological testing to better characterize the syndrome,” Sean T. Lynch, MD, first author of a study on the issue presented at the annual meeting of the American Psychiatric Association, said in an interview.
Dr. Lynch, of the department of psychiatry, Westchester Medical Center Health System, Valhalla, N.Y., and his colleagues enrolled 60 participants who had experienced acute COVID-19 disease 6-8 months earlier and had undergone neuropsychological, psychiatric, medical, functional, and quality-of-life assessments. Results from the study were published online in the Journal of the Academy of Consultation–Liaison Psychiatry (2022 Jan 25. doi: 10.1016/j.jaclp.2022.01.003).
Among the study participants, 32 were seeking treatment for brain fog in a clinical program for survivors of COVID-19, while the remaining 28 were part of an ongoing longitudinal investigation of neuropsychological, medical, and psychiatric sequelae of COVID-19, but were not seeking care for the persistent symptoms.
Assessments for neurocognitive impairment included a battery of tests used in infectious and other diseases, including the Test of Premorbid Function, the Patient Assessment of Own Function, the Trail Making Test parts A and B, the Stroop Color and Word Test, and others.
Overall, the battery of assessments showed that 37 (62%) of participants had neuropsychological test impairment, with results below the 16th percentile in two tests, while 16 (27%) showed scores indicative of severe impairment (below the second percentile in at least one test and below the 16th percentile in one test).
Those reporting brain fog had scores that were even lower than expected on tests of attention, processing speed, memory, and executive function. And among those reporting brain fog, significantly more had scores reflecting severe impairment compared with the controls (38% vs. 14%; P < .04).
“Based on what we’ve observed in our patients and what others have previously reported, we did expect to find some impairment in this study sample,” Dr. Lynch noted.
“However, we were surprised to find that 27% of the study sample had extremely low neuropsychological test scores, meaning that they scored at least two standard deviations below the expected score on at least one neuropsychological test based on their age and level of education.”
The brain fog group also reported significantly higher levels of depression, fatigue, PTSD, and functional difficulties, and lower quality of life.
Severe impairment on the neuropsychological tests correlated with the extent of acute COVID-19 symptoms, as well as depression scores, number of medical comorbidities, and subjective cognitive complaints.
An analysis of serum levels of the inflammatory markers among 50 of the 60 participants showed that 45% of the patients had an elevated IL-6, 20% had elevated TNF-alpha, and 41% had elevated CRP, compared with reference ranges.
IL-6 levels were found to correlate with acute COVID-19 symptoms, the number of medical comorbidities, fatigue, and measures of executive function, while C-reactive protein (CRP) correlated with current COVID-19 symptoms and depression scores.
In terms of clinical factors that might predict low neuropsychological test scores, Dr. Lynch noted that the “markers that we found to be significant included severity of acute COVID-19 illness, current post-COVID-19 symptoms, measures of depression and anxiety, level of fatigue, and number of medical comorbidities.”
Dr. Lynch noted that the ongoing study will include up to 18-month follow-ups that are currently underway. “The [follow-ups] will examine if symptoms improve over time and evaluate if any intervention that took place was successful,” he said.
Survey supports findings
The detrimental effects of mental health symptoms in long COVID were further supported in another study at the APA meeting, an online survey of 787 survivors of acute COVID-19.
In the community survey, presented by Michael Van Ameringen, MD, a professor in the department of psychiatry and behavioral neurosciences at McMaster University, in Hamilton, Ont., all respondents (100%) reported having persistent symptoms of the virus, and as many as 68% indicated that they had not returned to normal functioning, despite only 15% of the respondents having been hospitalized with COVID-19.
A large proportion showed significant depression, anxiety, and posttraumatic stress disorder (PTSD), and the most commonly reported persistent symptoms were fatigue in 75.9% of respondents, brain fog in 67.9%, concentration difficulties in 61.1%, and weakness in 51.2%.
As many as 88.2% of patients said they experienced persistent neurocognitive symptoms, with poor memory and concentration; 56% reported problems with word finding; and 54.1% had slowed thinking.
The respondents showed high rates of anxiety (41.7%) as well as depression (61.4%) as determined by scores above 9 on the Generalized Anxiety Disorder–7 (GAD-7) and Patient Health Questionnaires (PHQ-9).
As many as 40.5% of respondents showed probable PTSD, with scores above 30 on the PTSD checklist (PCL-5). Their mean resilience score on the Brief Resilient Coping Scale was 13.5, suggesting low resilience.
Among the respondents, 43.3% said they had received past treatment for mental health, while 33.5% were currently receiving mental health treatment.
Dr. Van Ameringen noted the important limitation of the study being an online survey with no control group, but said the responses nevertheless raise the question of the role of prior psychiatric disorders in long COVID.
“In our sample, 40% of respondents had a past psychiatric history, so you wonder if that also makes you vulnerable to long COVID,” he said in an interview.
“About a third were getting psychiatric help, but I think the more impaired you are, the more likely you are to seek help.”
Those who were hospitalized with COVID-19 were at a higher risk of PTSD compared with those not hospitalized (P < .001), as were those under the age of 30 (P < .05) or between 31 and 50 vs. over 50 (P < .01).
Dr. Van Ameringen noted that the survey’s high rate of subjects who had not returned to normal functioning was especially striking.
“This is not a minor issue – these are people who are no longer functioning in society,” he said.
In pandemics, the brain tends to be ‘overlooked’
Further addressing the neurological effects of COVID-19 at the APA meeting, Avindra Nath, MD, clinical director of the National Institutes of Neurologic Disorders and Stroke in Bethesda, Md., noted that the persisting cognitive and psychiatric symptoms after illness, such as brain fog and depression and anxiety, are not necessarily unique to COVID-19.
“We have seen this before,” he said. “There have been at least seven or eight human coronaviruses, and the interesting thing is each one affects the brain and causes neurological complications.”
The effects are classified differently and have slightly different receptors, “but the consequences are the same.”
Of note, however, research published in The Lancet Psychiatry (2021 May. doi: 10.1016/S2215-0366[21]00084-5) revealed that symptoms such as dementia, mood, and anxiety are significantly higher after COVID-19 compared with other respiratory infections, with the differences increasing at 180 days since the index event.
Dr. Nath noted that, over the decades, he has observed that in pandemics “the brain tends to get overlooked.” He explained that “what can be most important in the end is what happened in the brain, because those are the things that really cause the long-term consequences.”
“These patients are depressed; they have dementia, they have brain fog, and even now that we recognize these issues, we haven’t done a very good job of studying them,” he said. “There’s so much we still don’t know, and a lot of patients are left with these symptoms and nowhere to go.”
Dr. Lynch, Dr. Van Ameringen, and Dr. Nath had no disclosures to report.
NEW ORLEANS – , adding to mounting evidence of the significant toll the chronic condition can have on mental health.
“Many clinicians have observed the symptoms we describe in this study, however this report is among the first which identify the specific deficits using neuropsychological testing to better characterize the syndrome,” Sean T. Lynch, MD, first author of a study on the issue presented at the annual meeting of the American Psychiatric Association, said in an interview.
Dr. Lynch, of the department of psychiatry, Westchester Medical Center Health System, Valhalla, N.Y., and his colleagues enrolled 60 participants who had experienced acute COVID-19 disease 6-8 months earlier and had undergone neuropsychological, psychiatric, medical, functional, and quality-of-life assessments. Results from the study were published online in the Journal of the Academy of Consultation–Liaison Psychiatry (2022 Jan 25. doi: 10.1016/j.jaclp.2022.01.003).
Among the study participants, 32 were seeking treatment for brain fog in a clinical program for survivors of COVID-19, while the remaining 28 were part of an ongoing longitudinal investigation of neuropsychological, medical, and psychiatric sequelae of COVID-19, but were not seeking care for the persistent symptoms.
Assessments for neurocognitive impairment included a battery of tests used in infectious and other diseases, including the Test of Premorbid Function, the Patient Assessment of Own Function, the Trail Making Test parts A and B, the Stroop Color and Word Test, and others.
Overall, the battery of assessments showed that 37 (62%) of participants had neuropsychological test impairment, with results below the 16th percentile in two tests, while 16 (27%) showed scores indicative of severe impairment (below the second percentile in at least one test and below the 16th percentile in one test).
Those reporting brain fog had scores that were even lower than expected on tests of attention, processing speed, memory, and executive function. And among those reporting brain fog, significantly more had scores reflecting severe impairment compared with the controls (38% vs. 14%; P < .04).
“Based on what we’ve observed in our patients and what others have previously reported, we did expect to find some impairment in this study sample,” Dr. Lynch noted.
“However, we were surprised to find that 27% of the study sample had extremely low neuropsychological test scores, meaning that they scored at least two standard deviations below the expected score on at least one neuropsychological test based on their age and level of education.”
The brain fog group also reported significantly higher levels of depression, fatigue, PTSD, and functional difficulties, and lower quality of life.
Severe impairment on the neuropsychological tests correlated with the extent of acute COVID-19 symptoms, as well as depression scores, number of medical comorbidities, and subjective cognitive complaints.
An analysis of serum levels of the inflammatory markers among 50 of the 60 participants showed that 45% of the patients had an elevated IL-6, 20% had elevated TNF-alpha, and 41% had elevated CRP, compared with reference ranges.
IL-6 levels were found to correlate with acute COVID-19 symptoms, the number of medical comorbidities, fatigue, and measures of executive function, while C-reactive protein (CRP) correlated with current COVID-19 symptoms and depression scores.
In terms of clinical factors that might predict low neuropsychological test scores, Dr. Lynch noted that the “markers that we found to be significant included severity of acute COVID-19 illness, current post-COVID-19 symptoms, measures of depression and anxiety, level of fatigue, and number of medical comorbidities.”
Dr. Lynch noted that the ongoing study will include up to 18-month follow-ups that are currently underway. “The [follow-ups] will examine if symptoms improve over time and evaluate if any intervention that took place was successful,” he said.
Survey supports findings
The detrimental effects of mental health symptoms in long COVID were further supported in another study at the APA meeting, an online survey of 787 survivors of acute COVID-19.
In the community survey, presented by Michael Van Ameringen, MD, a professor in the department of psychiatry and behavioral neurosciences at McMaster University, in Hamilton, Ont., all respondents (100%) reported having persistent symptoms of the virus, and as many as 68% indicated that they had not returned to normal functioning, despite only 15% of the respondents having been hospitalized with COVID-19.
A large proportion showed significant depression, anxiety, and posttraumatic stress disorder (PTSD), and the most commonly reported persistent symptoms were fatigue in 75.9% of respondents, brain fog in 67.9%, concentration difficulties in 61.1%, and weakness in 51.2%.
As many as 88.2% of patients said they experienced persistent neurocognitive symptoms, with poor memory and concentration; 56% reported problems with word finding; and 54.1% had slowed thinking.
The respondents showed high rates of anxiety (41.7%) as well as depression (61.4%) as determined by scores above 9 on the Generalized Anxiety Disorder–7 (GAD-7) and Patient Health Questionnaires (PHQ-9).
As many as 40.5% of respondents showed probable PTSD, with scores above 30 on the PTSD checklist (PCL-5). Their mean resilience score on the Brief Resilient Coping Scale was 13.5, suggesting low resilience.
Among the respondents, 43.3% said they had received past treatment for mental health, while 33.5% were currently receiving mental health treatment.
Dr. Van Ameringen noted the important limitation of the study being an online survey with no control group, but said the responses nevertheless raise the question of the role of prior psychiatric disorders in long COVID.
“In our sample, 40% of respondents had a past psychiatric history, so you wonder if that also makes you vulnerable to long COVID,” he said in an interview.
“About a third were getting psychiatric help, but I think the more impaired you are, the more likely you are to seek help.”
Those who were hospitalized with COVID-19 were at a higher risk of PTSD compared with those not hospitalized (P < .001), as were those under the age of 30 (P < .05) or between 31 and 50 vs. over 50 (P < .01).
Dr. Van Ameringen noted that the survey’s high rate of subjects who had not returned to normal functioning was especially striking.
“This is not a minor issue – these are people who are no longer functioning in society,” he said.
In pandemics, the brain tends to be ‘overlooked’
Further addressing the neurological effects of COVID-19 at the APA meeting, Avindra Nath, MD, clinical director of the National Institutes of Neurologic Disorders and Stroke in Bethesda, Md., noted that the persisting cognitive and psychiatric symptoms after illness, such as brain fog and depression and anxiety, are not necessarily unique to COVID-19.
“We have seen this before,” he said. “There have been at least seven or eight human coronaviruses, and the interesting thing is each one affects the brain and causes neurological complications.”
The effects are classified differently and have slightly different receptors, “but the consequences are the same.”
Of note, however, research published in The Lancet Psychiatry (2021 May. doi: 10.1016/S2215-0366[21]00084-5) revealed that symptoms such as dementia, mood, and anxiety are significantly higher after COVID-19 compared with other respiratory infections, with the differences increasing at 180 days since the index event.
Dr. Nath noted that, over the decades, he has observed that in pandemics “the brain tends to get overlooked.” He explained that “what can be most important in the end is what happened in the brain, because those are the things that really cause the long-term consequences.”
“These patients are depressed; they have dementia, they have brain fog, and even now that we recognize these issues, we haven’t done a very good job of studying them,” he said. “There’s so much we still don’t know, and a lot of patients are left with these symptoms and nowhere to go.”
Dr. Lynch, Dr. Van Ameringen, and Dr. Nath had no disclosures to report.
NEW ORLEANS – , adding to mounting evidence of the significant toll the chronic condition can have on mental health.
“Many clinicians have observed the symptoms we describe in this study, however this report is among the first which identify the specific deficits using neuropsychological testing to better characterize the syndrome,” Sean T. Lynch, MD, first author of a study on the issue presented at the annual meeting of the American Psychiatric Association, said in an interview.
Dr. Lynch, of the department of psychiatry, Westchester Medical Center Health System, Valhalla, N.Y., and his colleagues enrolled 60 participants who had experienced acute COVID-19 disease 6-8 months earlier and had undergone neuropsychological, psychiatric, medical, functional, and quality-of-life assessments. Results from the study were published online in the Journal of the Academy of Consultation–Liaison Psychiatry (2022 Jan 25. doi: 10.1016/j.jaclp.2022.01.003).
Among the study participants, 32 were seeking treatment for brain fog in a clinical program for survivors of COVID-19, while the remaining 28 were part of an ongoing longitudinal investigation of neuropsychological, medical, and psychiatric sequelae of COVID-19, but were not seeking care for the persistent symptoms.
Assessments for neurocognitive impairment included a battery of tests used in infectious and other diseases, including the Test of Premorbid Function, the Patient Assessment of Own Function, the Trail Making Test parts A and B, the Stroop Color and Word Test, and others.
Overall, the battery of assessments showed that 37 (62%) of participants had neuropsychological test impairment, with results below the 16th percentile in two tests, while 16 (27%) showed scores indicative of severe impairment (below the second percentile in at least one test and below the 16th percentile in one test).
Those reporting brain fog had scores that were even lower than expected on tests of attention, processing speed, memory, and executive function. And among those reporting brain fog, significantly more had scores reflecting severe impairment compared with the controls (38% vs. 14%; P < .04).
“Based on what we’ve observed in our patients and what others have previously reported, we did expect to find some impairment in this study sample,” Dr. Lynch noted.
“However, we were surprised to find that 27% of the study sample had extremely low neuropsychological test scores, meaning that they scored at least two standard deviations below the expected score on at least one neuropsychological test based on their age and level of education.”
The brain fog group also reported significantly higher levels of depression, fatigue, PTSD, and functional difficulties, and lower quality of life.
Severe impairment on the neuropsychological tests correlated with the extent of acute COVID-19 symptoms, as well as depression scores, number of medical comorbidities, and subjective cognitive complaints.
An analysis of serum levels of the inflammatory markers among 50 of the 60 participants showed that 45% of the patients had an elevated IL-6, 20% had elevated TNF-alpha, and 41% had elevated CRP, compared with reference ranges.
IL-6 levels were found to correlate with acute COVID-19 symptoms, the number of medical comorbidities, fatigue, and measures of executive function, while C-reactive protein (CRP) correlated with current COVID-19 symptoms and depression scores.
In terms of clinical factors that might predict low neuropsychological test scores, Dr. Lynch noted that the “markers that we found to be significant included severity of acute COVID-19 illness, current post-COVID-19 symptoms, measures of depression and anxiety, level of fatigue, and number of medical comorbidities.”
Dr. Lynch noted that the ongoing study will include up to 18-month follow-ups that are currently underway. “The [follow-ups] will examine if symptoms improve over time and evaluate if any intervention that took place was successful,” he said.
Survey supports findings
The detrimental effects of mental health symptoms in long COVID were further supported in another study at the APA meeting, an online survey of 787 survivors of acute COVID-19.
In the community survey, presented by Michael Van Ameringen, MD, a professor in the department of psychiatry and behavioral neurosciences at McMaster University, in Hamilton, Ont., all respondents (100%) reported having persistent symptoms of the virus, and as many as 68% indicated that they had not returned to normal functioning, despite only 15% of the respondents having been hospitalized with COVID-19.
A large proportion showed significant depression, anxiety, and posttraumatic stress disorder (PTSD), and the most commonly reported persistent symptoms were fatigue in 75.9% of respondents, brain fog in 67.9%, concentration difficulties in 61.1%, and weakness in 51.2%.
As many as 88.2% of patients said they experienced persistent neurocognitive symptoms, with poor memory and concentration; 56% reported problems with word finding; and 54.1% had slowed thinking.
The respondents showed high rates of anxiety (41.7%) as well as depression (61.4%) as determined by scores above 9 on the Generalized Anxiety Disorder–7 (GAD-7) and Patient Health Questionnaires (PHQ-9).
As many as 40.5% of respondents showed probable PTSD, with scores above 30 on the PTSD checklist (PCL-5). Their mean resilience score on the Brief Resilient Coping Scale was 13.5, suggesting low resilience.
Among the respondents, 43.3% said they had received past treatment for mental health, while 33.5% were currently receiving mental health treatment.
Dr. Van Ameringen noted the important limitation of the study being an online survey with no control group, but said the responses nevertheless raise the question of the role of prior psychiatric disorders in long COVID.
“In our sample, 40% of respondents had a past psychiatric history, so you wonder if that also makes you vulnerable to long COVID,” he said in an interview.
“About a third were getting psychiatric help, but I think the more impaired you are, the more likely you are to seek help.”
Those who were hospitalized with COVID-19 were at a higher risk of PTSD compared with those not hospitalized (P < .001), as were those under the age of 30 (P < .05) or between 31 and 50 vs. over 50 (P < .01).
Dr. Van Ameringen noted that the survey’s high rate of subjects who had not returned to normal functioning was especially striking.
“This is not a minor issue – these are people who are no longer functioning in society,” he said.
In pandemics, the brain tends to be ‘overlooked’
Further addressing the neurological effects of COVID-19 at the APA meeting, Avindra Nath, MD, clinical director of the National Institutes of Neurologic Disorders and Stroke in Bethesda, Md., noted that the persisting cognitive and psychiatric symptoms after illness, such as brain fog and depression and anxiety, are not necessarily unique to COVID-19.
“We have seen this before,” he said. “There have been at least seven or eight human coronaviruses, and the interesting thing is each one affects the brain and causes neurological complications.”
The effects are classified differently and have slightly different receptors, “but the consequences are the same.”
Of note, however, research published in The Lancet Psychiatry (2021 May. doi: 10.1016/S2215-0366[21]00084-5) revealed that symptoms such as dementia, mood, and anxiety are significantly higher after COVID-19 compared with other respiratory infections, with the differences increasing at 180 days since the index event.
Dr. Nath noted that, over the decades, he has observed that in pandemics “the brain tends to get overlooked.” He explained that “what can be most important in the end is what happened in the brain, because those are the things that really cause the long-term consequences.”
“These patients are depressed; they have dementia, they have brain fog, and even now that we recognize these issues, we haven’t done a very good job of studying them,” he said. “There’s so much we still don’t know, and a lot of patients are left with these symptoms and nowhere to go.”
Dr. Lynch, Dr. Van Ameringen, and Dr. Nath had no disclosures to report.
AT APA 2022
FDA expands indication for spinal muscular atrophy drug
As previously reported, the FDA first approved oral risdiplam for SMA in children older than age 2 years in 2020.
The FDA expanded the indication for risdiplam to include babies younger than 2 months old because of interim safety and efficacy data from the ongoing RAINBOWFISH study. It includes 25 babies from birth to 6 weeks of age at first dose, all of whom have genetically diagnosed SMA but are not yet presenting with symptoms.
After 12 months of risdiplam treatment, the majority of presymptomatic infants with SMA reached key motor milestones, Genentech said in a news release.
Of the six babies with two or three copies of the SMN2 gene, all were able to sit after 1 year of active treatment, roughly two-thirds could stand, and half could walk independently.
All babies were alive at 12 months without permanent ventilation.
“The approval of Evrysdi for presymptomatic babies is particularly important, as early treatment of SMA, before symptoms start to arise, can help babies to achieve motor milestones,” Richard Finkel, MD, principal investigator of the trial, said in the release.
“With the inclusion of SMA in newborn screening programs, this approval provides the opportunity to start treating at home with Evrysdi soon after the diagnosis is confirmed,” added Dr. Finkel, who is director of the experimental neuroscience program, St. Jude Children’s Research Hospital, Memphis.
From newborns to older adults?
SMA is a rare and often fatal genetic disease that causes muscle weakness and progressive loss of movement.
SMA, which affects about 1 in 10,000 babies, is caused by a mutation in the survival motor neuron 1 (SMN1) gene. The gene encodes the SMN protein, which is critical for the maintenance and function of motor neurons.
Risdiplam is an orally administered, centrally and peripherally distributed small molecule that modulates survival motor neuron 2 (SMN2) premessenger RNA splicing to increase SMN protein levels.
As part of the label extension, the prescribing information for risdiplam has also been updated to include 2-year pooled data from parts 1 and 2 of the FIREFISH study, which demonstrated long-term efficacy and safety in symptomatic infants with Type 1 SMA, the company noted.
“Because of its efficacy in multiple settings, Evrysdi is now available for people with SMA, from presymptomatic newborns to older adults,” Levi Garraway, MD, PhD, chief medical officer and head of global product development at Genentech, said in the release.
“We are proud of this achievement, which has the potential to make a real difference to those living with SMA and their caregivers,” Dr. Garraway added.
A version of this article first appeared on Medscape.com.
As previously reported, the FDA first approved oral risdiplam for SMA in children older than age 2 years in 2020.
The FDA expanded the indication for risdiplam to include babies younger than 2 months old because of interim safety and efficacy data from the ongoing RAINBOWFISH study. It includes 25 babies from birth to 6 weeks of age at first dose, all of whom have genetically diagnosed SMA but are not yet presenting with symptoms.
After 12 months of risdiplam treatment, the majority of presymptomatic infants with SMA reached key motor milestones, Genentech said in a news release.
Of the six babies with two or three copies of the SMN2 gene, all were able to sit after 1 year of active treatment, roughly two-thirds could stand, and half could walk independently.
All babies were alive at 12 months without permanent ventilation.
“The approval of Evrysdi for presymptomatic babies is particularly important, as early treatment of SMA, before symptoms start to arise, can help babies to achieve motor milestones,” Richard Finkel, MD, principal investigator of the trial, said in the release.
“With the inclusion of SMA in newborn screening programs, this approval provides the opportunity to start treating at home with Evrysdi soon after the diagnosis is confirmed,” added Dr. Finkel, who is director of the experimental neuroscience program, St. Jude Children’s Research Hospital, Memphis.
From newborns to older adults?
SMA is a rare and often fatal genetic disease that causes muscle weakness and progressive loss of movement.
SMA, which affects about 1 in 10,000 babies, is caused by a mutation in the survival motor neuron 1 (SMN1) gene. The gene encodes the SMN protein, which is critical for the maintenance and function of motor neurons.
Risdiplam is an orally administered, centrally and peripherally distributed small molecule that modulates survival motor neuron 2 (SMN2) premessenger RNA splicing to increase SMN protein levels.
As part of the label extension, the prescribing information for risdiplam has also been updated to include 2-year pooled data from parts 1 and 2 of the FIREFISH study, which demonstrated long-term efficacy and safety in symptomatic infants with Type 1 SMA, the company noted.
“Because of its efficacy in multiple settings, Evrysdi is now available for people with SMA, from presymptomatic newborns to older adults,” Levi Garraway, MD, PhD, chief medical officer and head of global product development at Genentech, said in the release.
“We are proud of this achievement, which has the potential to make a real difference to those living with SMA and their caregivers,” Dr. Garraway added.
A version of this article first appeared on Medscape.com.
As previously reported, the FDA first approved oral risdiplam for SMA in children older than age 2 years in 2020.
The FDA expanded the indication for risdiplam to include babies younger than 2 months old because of interim safety and efficacy data from the ongoing RAINBOWFISH study. It includes 25 babies from birth to 6 weeks of age at first dose, all of whom have genetically diagnosed SMA but are not yet presenting with symptoms.
After 12 months of risdiplam treatment, the majority of presymptomatic infants with SMA reached key motor milestones, Genentech said in a news release.
Of the six babies with two or three copies of the SMN2 gene, all were able to sit after 1 year of active treatment, roughly two-thirds could stand, and half could walk independently.
All babies were alive at 12 months without permanent ventilation.
“The approval of Evrysdi for presymptomatic babies is particularly important, as early treatment of SMA, before symptoms start to arise, can help babies to achieve motor milestones,” Richard Finkel, MD, principal investigator of the trial, said in the release.
“With the inclusion of SMA in newborn screening programs, this approval provides the opportunity to start treating at home with Evrysdi soon after the diagnosis is confirmed,” added Dr. Finkel, who is director of the experimental neuroscience program, St. Jude Children’s Research Hospital, Memphis.
From newborns to older adults?
SMA is a rare and often fatal genetic disease that causes muscle weakness and progressive loss of movement.
SMA, which affects about 1 in 10,000 babies, is caused by a mutation in the survival motor neuron 1 (SMN1) gene. The gene encodes the SMN protein, which is critical for the maintenance and function of motor neurons.
Risdiplam is an orally administered, centrally and peripherally distributed small molecule that modulates survival motor neuron 2 (SMN2) premessenger RNA splicing to increase SMN protein levels.
As part of the label extension, the prescribing information for risdiplam has also been updated to include 2-year pooled data from parts 1 and 2 of the FIREFISH study, which demonstrated long-term efficacy and safety in symptomatic infants with Type 1 SMA, the company noted.
“Because of its efficacy in multiple settings, Evrysdi is now available for people with SMA, from presymptomatic newborns to older adults,” Levi Garraway, MD, PhD, chief medical officer and head of global product development at Genentech, said in the release.
“We are proud of this achievement, which has the potential to make a real difference to those living with SMA and their caregivers,” Dr. Garraway added.
A version of this article first appeared on Medscape.com.
‘Double-edged’ impact of sparring on the brains of MMA fighters
, early research suggests.
Investigators found sparring, defined as strategically hitting opponents with kicks, punches, and other strikes during practice sessions, is linked to increased white matter hyperintensities in the brain, pointing to possible vascular damage from repeated head trauma. However, the study results also show sparring was associated with a larger bilateral caudate which, in theory, is neuroprotective.
“From our preliminary study, sparring practice in MMA fighters may have a ‘double-edged sword’ effect on the brain,” study investigator Aaron Esagoff, a second-year medical student at Johns Hopkins University School of Medicine, Baltimore, told this news organization.
“The combination of complex movements along with constant strategy and anticipation of your opponent’s next move may provide a neuroprotective effect on the caudate,” Mr. Esagoff said. However, he added, more research is needed into understanding this particular finding.
The study results were presented at the American Psychiatric Association (APA) 2022 Annual Meeting.
Growing popularity
MMA is a full-contact combat sport that has become increasingly popular over the past 15 years. It combines techniques from boxing, wrestling, karate, judo, and jujitsu.
To prepare for fights, MMA practitioners incorporate sparring and grappling, which use techniques such as chokes and locks to submit an opponent. Head protection is sometimes incorporated during practice, but is not the norm during a fight, said Mr. Esagoff.
The study investigated sparring during practice rather than fights because, he said, MMA competitors only fight a few times a year but spend hundreds of hours training. “So the health effects of training are going to be really important,” he said.
As with other combat sports, MMA involves hits to the head. Previous research has shown repetitive head trauma can lead to neurodegenerative diseases, including chronic traumatic encephalopathy (CTE) and Alzheimer’s disease, Mr. Esagoff noted.
Previous studies have also linked more professional fights and years of fighting to a decrease in brain volume among MMA fighters, he added.
The new analysis was conducted as part of the Professional Fighters Brain Health Study, a longitudinal cohort study of MMA professional fighters. It included 92 fighters with data available on MRI and habits regarding practicing. The mean age of the participants was 30 years, 62% were White, and 85% were men.
The study examined sparring but did not include grappling because of “several challenges” with the current data analysis, Mr. Esagoff said. Researchers adjusted for age, sex, education, race, number of fights, total intracranial volume, and type of MRI scanner used.
A ‘highly strategic’ sport
Results showed a strong association between the number of sparring rounds per week and increased white matter hyperintensity volume (mcL) on MRI (P = .039).
This suggests white matter damage, possibly a result of direct neuronal injury, vascular damage, or immune modulation, said Mr. Esagoff. However, another mechanism may be involved, he added.
There was also a significant association between sparring and increased size of the caudate nucleus, an area of the brain involved in movement, learning, and memory (P = .014 for right caudate volume, P = .012 for left caudate volume).
There are some theories that might explain this finding, said Mr. Esagoff. For example, individuals who spar more may get better at avoiding impacts and injuries during a fight, which might in turn affect the size of the caudate.
The controlled movements and techniques used during sparring could also affect the caudate. “Some research has shown that behavior, learning, and/or exercise may increase the size of certain brain regions,” Mr. Esagoff said.
He noted the “highly strategic” nature of combat sports – and used the example of Brazilian jiu-jitsu. That sport “is known as human chess because it takes a thoughtful approach to defeat a larger opponent with base, leverage, and technique,” he said.
However, Mr. Esagoff stressed that while it is possible movements involved in MMA increase caudate size, this is just a theory at this point.
A study limitation was that fighters volunteered to participate and may not represent all fighters. As well, the study was cross-sectional and looked at only one point in time, so it cannot infer causation.
Overall, the new findings should help inform fighters, governing bodies, and the public about the potential risks and benefits of different styles of MMA fighting and practice, although more research is needed, said Mr. Esagoff.
He and his team now plan to conduct a longer-term study and investigate effects of grappling on brain structure and function in addition to sparring.
Jury still out
Commenting on the study, Howard Liu, MD, chair of the University of Nebraska Medical Center department of psychiatry and incoming chair of the APA’s Council on Communications, said the jury “is clearly still out” when it comes to the investigation of brain impacts.
“We don’t know quite what these changes fully correlate to,” said Dr. Liu, who moderated a press briefing highlighting the study.
He underlined the importance of protecting athletes vulnerable to head trauma, be they professionals or those involved at the youth sports level.
Dr. Liu also noted the “extreme popularity” and rapid growth of MMA around the world, which he said provides an opportunity for researchers to study these professional fighters.
“This is a unique population that signed up in the midst of hundreds of hours of sparring to advance neuroscience, and that’s quite amazing,” he said.
A version of this article first appeared on Medscape.com.
, early research suggests.
Investigators found sparring, defined as strategically hitting opponents with kicks, punches, and other strikes during practice sessions, is linked to increased white matter hyperintensities in the brain, pointing to possible vascular damage from repeated head trauma. However, the study results also show sparring was associated with a larger bilateral caudate which, in theory, is neuroprotective.
“From our preliminary study, sparring practice in MMA fighters may have a ‘double-edged sword’ effect on the brain,” study investigator Aaron Esagoff, a second-year medical student at Johns Hopkins University School of Medicine, Baltimore, told this news organization.
“The combination of complex movements along with constant strategy and anticipation of your opponent’s next move may provide a neuroprotective effect on the caudate,” Mr. Esagoff said. However, he added, more research is needed into understanding this particular finding.
The study results were presented at the American Psychiatric Association (APA) 2022 Annual Meeting.
Growing popularity
MMA is a full-contact combat sport that has become increasingly popular over the past 15 years. It combines techniques from boxing, wrestling, karate, judo, and jujitsu.
To prepare for fights, MMA practitioners incorporate sparring and grappling, which use techniques such as chokes and locks to submit an opponent. Head protection is sometimes incorporated during practice, but is not the norm during a fight, said Mr. Esagoff.
The study investigated sparring during practice rather than fights because, he said, MMA competitors only fight a few times a year but spend hundreds of hours training. “So the health effects of training are going to be really important,” he said.
As with other combat sports, MMA involves hits to the head. Previous research has shown repetitive head trauma can lead to neurodegenerative diseases, including chronic traumatic encephalopathy (CTE) and Alzheimer’s disease, Mr. Esagoff noted.
Previous studies have also linked more professional fights and years of fighting to a decrease in brain volume among MMA fighters, he added.
The new analysis was conducted as part of the Professional Fighters Brain Health Study, a longitudinal cohort study of MMA professional fighters. It included 92 fighters with data available on MRI and habits regarding practicing. The mean age of the participants was 30 years, 62% were White, and 85% were men.
The study examined sparring but did not include grappling because of “several challenges” with the current data analysis, Mr. Esagoff said. Researchers adjusted for age, sex, education, race, number of fights, total intracranial volume, and type of MRI scanner used.
A ‘highly strategic’ sport
Results showed a strong association between the number of sparring rounds per week and increased white matter hyperintensity volume (mcL) on MRI (P = .039).
This suggests white matter damage, possibly a result of direct neuronal injury, vascular damage, or immune modulation, said Mr. Esagoff. However, another mechanism may be involved, he added.
There was also a significant association between sparring and increased size of the caudate nucleus, an area of the brain involved in movement, learning, and memory (P = .014 for right caudate volume, P = .012 for left caudate volume).
There are some theories that might explain this finding, said Mr. Esagoff. For example, individuals who spar more may get better at avoiding impacts and injuries during a fight, which might in turn affect the size of the caudate.
The controlled movements and techniques used during sparring could also affect the caudate. “Some research has shown that behavior, learning, and/or exercise may increase the size of certain brain regions,” Mr. Esagoff said.
He noted the “highly strategic” nature of combat sports – and used the example of Brazilian jiu-jitsu. That sport “is known as human chess because it takes a thoughtful approach to defeat a larger opponent with base, leverage, and technique,” he said.
However, Mr. Esagoff stressed that while it is possible movements involved in MMA increase caudate size, this is just a theory at this point.
A study limitation was that fighters volunteered to participate and may not represent all fighters. As well, the study was cross-sectional and looked at only one point in time, so it cannot infer causation.
Overall, the new findings should help inform fighters, governing bodies, and the public about the potential risks and benefits of different styles of MMA fighting and practice, although more research is needed, said Mr. Esagoff.
He and his team now plan to conduct a longer-term study and investigate effects of grappling on brain structure and function in addition to sparring.
Jury still out
Commenting on the study, Howard Liu, MD, chair of the University of Nebraska Medical Center department of psychiatry and incoming chair of the APA’s Council on Communications, said the jury “is clearly still out” when it comes to the investigation of brain impacts.
“We don’t know quite what these changes fully correlate to,” said Dr. Liu, who moderated a press briefing highlighting the study.
He underlined the importance of protecting athletes vulnerable to head trauma, be they professionals or those involved at the youth sports level.
Dr. Liu also noted the “extreme popularity” and rapid growth of MMA around the world, which he said provides an opportunity for researchers to study these professional fighters.
“This is a unique population that signed up in the midst of hundreds of hours of sparring to advance neuroscience, and that’s quite amazing,” he said.
A version of this article first appeared on Medscape.com.
, early research suggests.
Investigators found sparring, defined as strategically hitting opponents with kicks, punches, and other strikes during practice sessions, is linked to increased white matter hyperintensities in the brain, pointing to possible vascular damage from repeated head trauma. However, the study results also show sparring was associated with a larger bilateral caudate which, in theory, is neuroprotective.
“From our preliminary study, sparring practice in MMA fighters may have a ‘double-edged sword’ effect on the brain,” study investigator Aaron Esagoff, a second-year medical student at Johns Hopkins University School of Medicine, Baltimore, told this news organization.
“The combination of complex movements along with constant strategy and anticipation of your opponent’s next move may provide a neuroprotective effect on the caudate,” Mr. Esagoff said. However, he added, more research is needed into understanding this particular finding.
The study results were presented at the American Psychiatric Association (APA) 2022 Annual Meeting.
Growing popularity
MMA is a full-contact combat sport that has become increasingly popular over the past 15 years. It combines techniques from boxing, wrestling, karate, judo, and jujitsu.
To prepare for fights, MMA practitioners incorporate sparring and grappling, which use techniques such as chokes and locks to submit an opponent. Head protection is sometimes incorporated during practice, but is not the norm during a fight, said Mr. Esagoff.
The study investigated sparring during practice rather than fights because, he said, MMA competitors only fight a few times a year but spend hundreds of hours training. “So the health effects of training are going to be really important,” he said.
As with other combat sports, MMA involves hits to the head. Previous research has shown repetitive head trauma can lead to neurodegenerative diseases, including chronic traumatic encephalopathy (CTE) and Alzheimer’s disease, Mr. Esagoff noted.
Previous studies have also linked more professional fights and years of fighting to a decrease in brain volume among MMA fighters, he added.
The new analysis was conducted as part of the Professional Fighters Brain Health Study, a longitudinal cohort study of MMA professional fighters. It included 92 fighters with data available on MRI and habits regarding practicing. The mean age of the participants was 30 years, 62% were White, and 85% were men.
The study examined sparring but did not include grappling because of “several challenges” with the current data analysis, Mr. Esagoff said. Researchers adjusted for age, sex, education, race, number of fights, total intracranial volume, and type of MRI scanner used.
A ‘highly strategic’ sport
Results showed a strong association between the number of sparring rounds per week and increased white matter hyperintensity volume (mcL) on MRI (P = .039).
This suggests white matter damage, possibly a result of direct neuronal injury, vascular damage, or immune modulation, said Mr. Esagoff. However, another mechanism may be involved, he added.
There was also a significant association between sparring and increased size of the caudate nucleus, an area of the brain involved in movement, learning, and memory (P = .014 for right caudate volume, P = .012 for left caudate volume).
There are some theories that might explain this finding, said Mr. Esagoff. For example, individuals who spar more may get better at avoiding impacts and injuries during a fight, which might in turn affect the size of the caudate.
The controlled movements and techniques used during sparring could also affect the caudate. “Some research has shown that behavior, learning, and/or exercise may increase the size of certain brain regions,” Mr. Esagoff said.
He noted the “highly strategic” nature of combat sports – and used the example of Brazilian jiu-jitsu. That sport “is known as human chess because it takes a thoughtful approach to defeat a larger opponent with base, leverage, and technique,” he said.
However, Mr. Esagoff stressed that while it is possible movements involved in MMA increase caudate size, this is just a theory at this point.
A study limitation was that fighters volunteered to participate and may not represent all fighters. As well, the study was cross-sectional and looked at only one point in time, so it cannot infer causation.
Overall, the new findings should help inform fighters, governing bodies, and the public about the potential risks and benefits of different styles of MMA fighting and practice, although more research is needed, said Mr. Esagoff.
He and his team now plan to conduct a longer-term study and investigate effects of grappling on brain structure and function in addition to sparring.
Jury still out
Commenting on the study, Howard Liu, MD, chair of the University of Nebraska Medical Center department of psychiatry and incoming chair of the APA’s Council on Communications, said the jury “is clearly still out” when it comes to the investigation of brain impacts.
“We don’t know quite what these changes fully correlate to,” said Dr. Liu, who moderated a press briefing highlighting the study.
He underlined the importance of protecting athletes vulnerable to head trauma, be they professionals or those involved at the youth sports level.
Dr. Liu also noted the “extreme popularity” and rapid growth of MMA around the world, which he said provides an opportunity for researchers to study these professional fighters.
“This is a unique population that signed up in the midst of hundreds of hours of sparring to advance neuroscience, and that’s quite amazing,” he said.
A version of this article first appeared on Medscape.com.
FROM APA 2022
Duration of breastfeeding associated with cognition in children
Breastfeeding duration is associated with improved cognitive scores at ages 5-14, even after controlling for maternal socioeconomic position and cognitive ability, said the researchers behind a new study.
Despite previous studies demonstrating an association between breastfeeding and standardized intelligence test scores – with breastfed infants scoring higher on intelligence tests than non-breastfed infants – a causal relationship is still contested.
“There is some debate about whether breastfeeding a baby for a longer period of time improves their cognitive development,” the authors of the new study said. They went on to explain how improved cognitive outcomes in breastfed infants could potentially be explained by other characteristics of the women, such as “socioeconomics and maternal intelligence.”
Important at the population level
For the study, published in the open-access journal PLOS ONE, researchers from the University of Oxford (England) set out to investigate how much these confounders influenced the association between breastfeeding duration and cognitive development.
They analyzed data from the U.K. Millennium Cohort Study on 7,855 infants born in 2000 to 2002 and followed until age 14. They highlighted that although the cohort was not specifically designed to address the association between breastfeeding and cognition, it included information on duration of any breastfeeding, duration of exclusive breastfeeding, verbal cognitive scores at ages 5, 7, 11, and 14, spatial cognitive scores at ages 5, 7, and 11, as well as potential confounders, including socioeconomic characteristics and maternal cognition, based on a vocabulary test.
The researchers discovered that longer breastfeeding durations were associated with higher verbal and spatial cognitive scores at all ages up to 14 and 11, respectively.
After taking the differences in socioeconomic position and maternal cognitive ability into account, those children who were breastfed for longer scored higher in cognitive measures up to age 14, compared with children who were not breastfed. They also found that longer breastfeeding durations were associated with mean cognitive scores 0.08-0.26 standard deviations higher than the mean cognitive score of those who were never breastfed. “This difference may seem small for an individual child but could be important at the population level,” the authors commented.
Modest effect
In the United Kingdom, women who have more educational qualifications and are more economically advantaged tend to breastfeed for longer, said the authors. In addition, they added, this group tends to “score more highly on cognitive tests.”
These differences could explain why babies who breastfeed for longer do better in cognitive assessments. However, they said that in their study, “we found that even after taking these differences into account, children breastfed for longer scored higher in cognitive measures up to age 14, in comparison to children who were not breastfed.”
The authors explained that the association between breastfeeding duration and cognitive scores “persists after adjusting for socioeconomics and maternal intelligence.” However, they pointed out that “the effect was modest.”
A version of this article first appeared on Medscape UK.
Breastfeeding duration is associated with improved cognitive scores at ages 5-14, even after controlling for maternal socioeconomic position and cognitive ability, said the researchers behind a new study.
Despite previous studies demonstrating an association between breastfeeding and standardized intelligence test scores – with breastfed infants scoring higher on intelligence tests than non-breastfed infants – a causal relationship is still contested.
“There is some debate about whether breastfeeding a baby for a longer period of time improves their cognitive development,” the authors of the new study said. They went on to explain how improved cognitive outcomes in breastfed infants could potentially be explained by other characteristics of the women, such as “socioeconomics and maternal intelligence.”
Important at the population level
For the study, published in the open-access journal PLOS ONE, researchers from the University of Oxford (England) set out to investigate how much these confounders influenced the association between breastfeeding duration and cognitive development.
They analyzed data from the U.K. Millennium Cohort Study on 7,855 infants born in 2000 to 2002 and followed until age 14. They highlighted that although the cohort was not specifically designed to address the association between breastfeeding and cognition, it included information on duration of any breastfeeding, duration of exclusive breastfeeding, verbal cognitive scores at ages 5, 7, 11, and 14, spatial cognitive scores at ages 5, 7, and 11, as well as potential confounders, including socioeconomic characteristics and maternal cognition, based on a vocabulary test.
The researchers discovered that longer breastfeeding durations were associated with higher verbal and spatial cognitive scores at all ages up to 14 and 11, respectively.
After taking the differences in socioeconomic position and maternal cognitive ability into account, those children who were breastfed for longer scored higher in cognitive measures up to age 14, compared with children who were not breastfed. They also found that longer breastfeeding durations were associated with mean cognitive scores 0.08-0.26 standard deviations higher than the mean cognitive score of those who were never breastfed. “This difference may seem small for an individual child but could be important at the population level,” the authors commented.
Modest effect
In the United Kingdom, women who have more educational qualifications and are more economically advantaged tend to breastfeed for longer, said the authors. In addition, they added, this group tends to “score more highly on cognitive tests.”
These differences could explain why babies who breastfeed for longer do better in cognitive assessments. However, they said that in their study, “we found that even after taking these differences into account, children breastfed for longer scored higher in cognitive measures up to age 14, in comparison to children who were not breastfed.”
The authors explained that the association between breastfeeding duration and cognitive scores “persists after adjusting for socioeconomics and maternal intelligence.” However, they pointed out that “the effect was modest.”
A version of this article first appeared on Medscape UK.
Breastfeeding duration is associated with improved cognitive scores at ages 5-14, even after controlling for maternal socioeconomic position and cognitive ability, said the researchers behind a new study.
Despite previous studies demonstrating an association between breastfeeding and standardized intelligence test scores – with breastfed infants scoring higher on intelligence tests than non-breastfed infants – a causal relationship is still contested.
“There is some debate about whether breastfeeding a baby for a longer period of time improves their cognitive development,” the authors of the new study said. They went on to explain how improved cognitive outcomes in breastfed infants could potentially be explained by other characteristics of the women, such as “socioeconomics and maternal intelligence.”
Important at the population level
For the study, published in the open-access journal PLOS ONE, researchers from the University of Oxford (England) set out to investigate how much these confounders influenced the association between breastfeeding duration and cognitive development.
They analyzed data from the U.K. Millennium Cohort Study on 7,855 infants born in 2000 to 2002 and followed until age 14. They highlighted that although the cohort was not specifically designed to address the association between breastfeeding and cognition, it included information on duration of any breastfeeding, duration of exclusive breastfeeding, verbal cognitive scores at ages 5, 7, 11, and 14, spatial cognitive scores at ages 5, 7, and 11, as well as potential confounders, including socioeconomic characteristics and maternal cognition, based on a vocabulary test.
The researchers discovered that longer breastfeeding durations were associated with higher verbal and spatial cognitive scores at all ages up to 14 and 11, respectively.
After taking the differences in socioeconomic position and maternal cognitive ability into account, those children who were breastfed for longer scored higher in cognitive measures up to age 14, compared with children who were not breastfed. They also found that longer breastfeeding durations were associated with mean cognitive scores 0.08-0.26 standard deviations higher than the mean cognitive score of those who were never breastfed. “This difference may seem small for an individual child but could be important at the population level,” the authors commented.
Modest effect
In the United Kingdom, women who have more educational qualifications and are more economically advantaged tend to breastfeed for longer, said the authors. In addition, they added, this group tends to “score more highly on cognitive tests.”
These differences could explain why babies who breastfeed for longer do better in cognitive assessments. However, they said that in their study, “we found that even after taking these differences into account, children breastfed for longer scored higher in cognitive measures up to age 14, in comparison to children who were not breastfed.”
The authors explained that the association between breastfeeding duration and cognitive scores “persists after adjusting for socioeconomics and maternal intelligence.” However, they pointed out that “the effect was modest.”
A version of this article first appeared on Medscape UK.
FROM PLOS ONE
Thrombolysis is safe in stroke patients on oral anticoagulants
, a new observational study suggests, prompting researchers to ask whether guidelines that restrict its use should be updated.
Researchers found that DOAC users were significantly less likely to develop symptomatic intracerebral hemorrhage (sICH) after IVT, and there was no difference in functional independence at 3 months, compared with patients who received IVT but who did not receive DOAC.
“At the moment, the guidelines really pose a barrier and stop sign in front of the most important medical reperfusion therapy, which is thrombolysis,” said principal investigator Jan Purrucker, MD, professor of neurology at Heidelberg University Hospital.
“The main question we have to answer is, is IVT safe in patients with acute ischemic stroke who were pretreated with direct oral anticoagulants or not?”
The findings were presented at the European Stroke Organisation Conference (ESOC) 2022, Lyon, France.
A ‘daily clinical problem’
As many as 20% of patients with atrial fibrillation experience ischemic stroke while receiving DOAC therapy. Reperfusion therapy with intravenous alteplase is considered standard of care for acute ischemic stroke, but current guidelines recommend against the use of IVT for patients who have recently received a DOAC, owing to safety concerns that researchers say are not backed by strong clinical evidence.
A recent study found no significant difference in sICH among patients who received IV alteplase for acute ischemic stroke within 7 days of receiving therapy with non–vitamin K antagonist oral anticoagulants.
“In our daily clinical practice, we face a lot of patients who have received oral anticoagulation, many with atrial fibrillation, but a lot of other indicators as well, and they suffer from ischemic stroke,” Dr. Purrucker said. “They usually are ineligible for medical reperfusion therapy because of quite strict guideline recommendations at the moment. This is a daily clinical problem.”
Dr. Purrucker and colleagues in New Zealand and Switzerland launched an international, observational, multicenter cohort study to examine the issue.
Researchers collected data on patients with ischemic stroke who had last received DOAC therapy 48 hours or less before the event or whose last intake was unknown and who had received IVT. They included 20,448 patients, 830 of whom were receiving DOAC therapy at the time of stroke onset.
Among the DOAC users, 30% received DOAC reversal prior to IVT, 27% had their DOAC level measured, and 42% received IVT without reversal treatment or knowledge of DOAC levels.
Overall, 4.5% of patients developed sICH. Compared with the control group, DOAC users were half as likely to develop sICH (adjusted odds ratio, 0.47; P = .003).
There was no significant difference between groups in independent outcome at 3 months, defined as a Modified Rankin Scale score of 1 to 3 (aOR, 1.21; 95% confidence interval, 0.99-1.49).
This finding held across patient subgroups, including patients for whom selection methods differed and patients with very recent intake of less than 12 hours.
“The question is whether we are so confident in these data that we would change our clinical practice now,” Dr. Purrucker said.
Infrastructure needed
While the findings are promising, more data are needed to strengthen the argument for revising current IVT guidelines, said Ho-Yan Yvonne Chun, PhD, honorary senior clinical lecturer with the Centre for Clinical Brain Sciences at the University of Edinburgh and a consultant in stroke medicine for NHS Lothian and Borders General Hospital, who commented on the findings.
“The study sample are a highly selected group of patients from selected centers that have the infrastructure to offer DOAC level checking and DOAC reversal,” Dr. Chun said. “The selected centers are not representative of the majority of hospitals that offer IVT to stroke patients with acute stroke.”
Most hospitals lack the equipment necessary to test DOAC levels and don’t have immediate access to DOAC reversal agents, Dr. Chun said. In those centers, she added, the administration of IVT could be delayed, which might affect clinical outcomes.
“Infrastructure needs to be in place to ensure timely delivery of IVT to these patients,” Dr. Chun added. “This means that in real-world practice, hospitals need to have right logistical pathway in place in order to provide timely DOAC level checking and DOAC reversal agents.”
Dr. Chun added that “large pragmatic clinical trials, preferably multicentered, are needed to provide the definitive evidence on the safety and effectiveness of using these approaches to select patients with prior DOAC use for IVT.”
But such a study may not be feasible, Dr. Purrucker said. Among the hurdles he noted are the large sample size needed for such a trial, uncertainty regarding funding, and patient selection bias, resulting from the fact that such studies would likely exclude patients eligible for mechanical thrombectomy or those eligible for reversal treatment.
In light of earlier studies, including preclinical data that support the safety of DOACs in IVT, and these new data, Dr. Purrucker said he hopes a change in guidelines might be taken up in the future.
“But it should be good academic practice to first let the results be externally evaluated, for example, during the manuscript submission process,” he said. “But once published, guideline working groups will have to evaluate the recent and new evidence and might reconsider previous recommendations.”
The study received no commercial funding. Dr. Purrucker and Dr. Chun reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, a new observational study suggests, prompting researchers to ask whether guidelines that restrict its use should be updated.
Researchers found that DOAC users were significantly less likely to develop symptomatic intracerebral hemorrhage (sICH) after IVT, and there was no difference in functional independence at 3 months, compared with patients who received IVT but who did not receive DOAC.
“At the moment, the guidelines really pose a barrier and stop sign in front of the most important medical reperfusion therapy, which is thrombolysis,” said principal investigator Jan Purrucker, MD, professor of neurology at Heidelberg University Hospital.
“The main question we have to answer is, is IVT safe in patients with acute ischemic stroke who were pretreated with direct oral anticoagulants or not?”
The findings were presented at the European Stroke Organisation Conference (ESOC) 2022, Lyon, France.
A ‘daily clinical problem’
As many as 20% of patients with atrial fibrillation experience ischemic stroke while receiving DOAC therapy. Reperfusion therapy with intravenous alteplase is considered standard of care for acute ischemic stroke, but current guidelines recommend against the use of IVT for patients who have recently received a DOAC, owing to safety concerns that researchers say are not backed by strong clinical evidence.
A recent study found no significant difference in sICH among patients who received IV alteplase for acute ischemic stroke within 7 days of receiving therapy with non–vitamin K antagonist oral anticoagulants.
“In our daily clinical practice, we face a lot of patients who have received oral anticoagulation, many with atrial fibrillation, but a lot of other indicators as well, and they suffer from ischemic stroke,” Dr. Purrucker said. “They usually are ineligible for medical reperfusion therapy because of quite strict guideline recommendations at the moment. This is a daily clinical problem.”
Dr. Purrucker and colleagues in New Zealand and Switzerland launched an international, observational, multicenter cohort study to examine the issue.
Researchers collected data on patients with ischemic stroke who had last received DOAC therapy 48 hours or less before the event or whose last intake was unknown and who had received IVT. They included 20,448 patients, 830 of whom were receiving DOAC therapy at the time of stroke onset.
Among the DOAC users, 30% received DOAC reversal prior to IVT, 27% had their DOAC level measured, and 42% received IVT without reversal treatment or knowledge of DOAC levels.
Overall, 4.5% of patients developed sICH. Compared with the control group, DOAC users were half as likely to develop sICH (adjusted odds ratio, 0.47; P = .003).
There was no significant difference between groups in independent outcome at 3 months, defined as a Modified Rankin Scale score of 1 to 3 (aOR, 1.21; 95% confidence interval, 0.99-1.49).
This finding held across patient subgroups, including patients for whom selection methods differed and patients with very recent intake of less than 12 hours.
“The question is whether we are so confident in these data that we would change our clinical practice now,” Dr. Purrucker said.
Infrastructure needed
While the findings are promising, more data are needed to strengthen the argument for revising current IVT guidelines, said Ho-Yan Yvonne Chun, PhD, honorary senior clinical lecturer with the Centre for Clinical Brain Sciences at the University of Edinburgh and a consultant in stroke medicine for NHS Lothian and Borders General Hospital, who commented on the findings.
“The study sample are a highly selected group of patients from selected centers that have the infrastructure to offer DOAC level checking and DOAC reversal,” Dr. Chun said. “The selected centers are not representative of the majority of hospitals that offer IVT to stroke patients with acute stroke.”
Most hospitals lack the equipment necessary to test DOAC levels and don’t have immediate access to DOAC reversal agents, Dr. Chun said. In those centers, she added, the administration of IVT could be delayed, which might affect clinical outcomes.
“Infrastructure needs to be in place to ensure timely delivery of IVT to these patients,” Dr. Chun added. “This means that in real-world practice, hospitals need to have right logistical pathway in place in order to provide timely DOAC level checking and DOAC reversal agents.”
Dr. Chun added that “large pragmatic clinical trials, preferably multicentered, are needed to provide the definitive evidence on the safety and effectiveness of using these approaches to select patients with prior DOAC use for IVT.”
But such a study may not be feasible, Dr. Purrucker said. Among the hurdles he noted are the large sample size needed for such a trial, uncertainty regarding funding, and patient selection bias, resulting from the fact that such studies would likely exclude patients eligible for mechanical thrombectomy or those eligible for reversal treatment.
In light of earlier studies, including preclinical data that support the safety of DOACs in IVT, and these new data, Dr. Purrucker said he hopes a change in guidelines might be taken up in the future.
“But it should be good academic practice to first let the results be externally evaluated, for example, during the manuscript submission process,” he said. “But once published, guideline working groups will have to evaluate the recent and new evidence and might reconsider previous recommendations.”
The study received no commercial funding. Dr. Purrucker and Dr. Chun reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, a new observational study suggests, prompting researchers to ask whether guidelines that restrict its use should be updated.
Researchers found that DOAC users were significantly less likely to develop symptomatic intracerebral hemorrhage (sICH) after IVT, and there was no difference in functional independence at 3 months, compared with patients who received IVT but who did not receive DOAC.
“At the moment, the guidelines really pose a barrier and stop sign in front of the most important medical reperfusion therapy, which is thrombolysis,” said principal investigator Jan Purrucker, MD, professor of neurology at Heidelberg University Hospital.
“The main question we have to answer is, is IVT safe in patients with acute ischemic stroke who were pretreated with direct oral anticoagulants or not?”
The findings were presented at the European Stroke Organisation Conference (ESOC) 2022, Lyon, France.
A ‘daily clinical problem’
As many as 20% of patients with atrial fibrillation experience ischemic stroke while receiving DOAC therapy. Reperfusion therapy with intravenous alteplase is considered standard of care for acute ischemic stroke, but current guidelines recommend against the use of IVT for patients who have recently received a DOAC, owing to safety concerns that researchers say are not backed by strong clinical evidence.
A recent study found no significant difference in sICH among patients who received IV alteplase for acute ischemic stroke within 7 days of receiving therapy with non–vitamin K antagonist oral anticoagulants.
“In our daily clinical practice, we face a lot of patients who have received oral anticoagulation, many with atrial fibrillation, but a lot of other indicators as well, and they suffer from ischemic stroke,” Dr. Purrucker said. “They usually are ineligible for medical reperfusion therapy because of quite strict guideline recommendations at the moment. This is a daily clinical problem.”
Dr. Purrucker and colleagues in New Zealand and Switzerland launched an international, observational, multicenter cohort study to examine the issue.
Researchers collected data on patients with ischemic stroke who had last received DOAC therapy 48 hours or less before the event or whose last intake was unknown and who had received IVT. They included 20,448 patients, 830 of whom were receiving DOAC therapy at the time of stroke onset.
Among the DOAC users, 30% received DOAC reversal prior to IVT, 27% had their DOAC level measured, and 42% received IVT without reversal treatment or knowledge of DOAC levels.
Overall, 4.5% of patients developed sICH. Compared with the control group, DOAC users were half as likely to develop sICH (adjusted odds ratio, 0.47; P = .003).
There was no significant difference between groups in independent outcome at 3 months, defined as a Modified Rankin Scale score of 1 to 3 (aOR, 1.21; 95% confidence interval, 0.99-1.49).
This finding held across patient subgroups, including patients for whom selection methods differed and patients with very recent intake of less than 12 hours.
“The question is whether we are so confident in these data that we would change our clinical practice now,” Dr. Purrucker said.
Infrastructure needed
While the findings are promising, more data are needed to strengthen the argument for revising current IVT guidelines, said Ho-Yan Yvonne Chun, PhD, honorary senior clinical lecturer with the Centre for Clinical Brain Sciences at the University of Edinburgh and a consultant in stroke medicine for NHS Lothian and Borders General Hospital, who commented on the findings.
“The study sample are a highly selected group of patients from selected centers that have the infrastructure to offer DOAC level checking and DOAC reversal,” Dr. Chun said. “The selected centers are not representative of the majority of hospitals that offer IVT to stroke patients with acute stroke.”
Most hospitals lack the equipment necessary to test DOAC levels and don’t have immediate access to DOAC reversal agents, Dr. Chun said. In those centers, she added, the administration of IVT could be delayed, which might affect clinical outcomes.
“Infrastructure needs to be in place to ensure timely delivery of IVT to these patients,” Dr. Chun added. “This means that in real-world practice, hospitals need to have right logistical pathway in place in order to provide timely DOAC level checking and DOAC reversal agents.”
Dr. Chun added that “large pragmatic clinical trials, preferably multicentered, are needed to provide the definitive evidence on the safety and effectiveness of using these approaches to select patients with prior DOAC use for IVT.”
But such a study may not be feasible, Dr. Purrucker said. Among the hurdles he noted are the large sample size needed for such a trial, uncertainty regarding funding, and patient selection bias, resulting from the fact that such studies would likely exclude patients eligible for mechanical thrombectomy or those eligible for reversal treatment.
In light of earlier studies, including preclinical data that support the safety of DOACs in IVT, and these new data, Dr. Purrucker said he hopes a change in guidelines might be taken up in the future.
“But it should be good academic practice to first let the results be externally evaluated, for example, during the manuscript submission process,” he said. “But once published, guideline working groups will have to evaluate the recent and new evidence and might reconsider previous recommendations.”
The study received no commercial funding. Dr. Purrucker and Dr. Chun reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ESOC 2022





