User login
New insight into how brain stimulation eases major depression
For the first time, researchers understand what happens to the brain when patients with treatment-resistant depression receive repetitive transcranial magnetic stimulation (rTMS).
Using functional magnetic resonance imaging (fMRI), they showed that rTMS induces widespread alterations in functional connectivity in brain regions involved in emotion and motor control.
“‘How does rTMS work?’ is one of the most frequent questions I get in clinic. Providing an accurate explanation and narrative to patients is critical,” senior investigator Fidel Vila-Rodriguez, MD, PhD, director of the Non-Invasive Neurostimulation Therapies Laboratory, University of British Columbia, Vancouver, told this news organization.
“Our findings suggest that rTMS might rely on the brain’s capacity for change (neuroplasticity) to exert its effects and that rTMS effects on the brain are widespread beyond the focal area stimulated (functional network effects),” Dr. Vila-Rodriguez added.
The study was published online in the American Journal of Psychiatry.
Mechanistic insights
Although rTMS has proven efficacy for treatment-resistant depression, the mechanisms behind how it affects the brain are not well understood.
In the current study, researchers used fMRI to assess changes in functional connectivity induced by a single rTMS session in 26 women and 12 men with treatment-resistant depression.
They found that – from managing emotional responses to memory and motor control.
Following a 4-week course of rTMS, these connectivity changes predicted about 30% of the variance of improvement in scores on the Montgomery-Åsberg Depression Rating Scale after rTMS treatment.
The most robust predictive associations involved connections between prefrontal regions and motor, parietal, and insular cortices and between bilateral regions of the thalamus.
“By demonstrating this principle and identifying regions of the brain that are activated by rTMS, we can now try to understand whether this pattern can be used as a biomarker,” Dr. Vila-Rodriguez said in a news release.
“This work provides a mechanistic explanation of what rTMS does to treat depression and supports the notion that for rTMS to treat depressive symptoms a distributed change in brain activity (network or circuit base) is necessary,” he told this news organization.
With funding from the Canadian Institutes of Health Research (CIHR), the team will next see if they can use fMRI to guide rTMS at the individual level, with the ultimate goal of “personalizing” rTMS using individualized functional targets, Dr. Vila-Rodriguez said.
New generation of tms researchers
Reached for comment, Jonathan Downar, MD, PhD, department of psychiatry, University of Toronto, noted that TMS can be “very effective” for treatment-resistant depression, and it has a “very clean side effect profile compared to medications.”
What the field is trying to figure out now is “who it works for and how we can predict more effectively who’s going to benefit from it,” Dr. Downar said in an interview.
He noted that the study’s investigators are part of a “new generation of TMS researchers who are bringing new ideas into the fold and figuring out how to use brain imaging to personalize the treatment.” This study represents “a step” in that direction.
“A challenge for the field is that it’s often pretty easy to demonstrate a change at the group level, but the question is whether we can use that at the individual level. That’s a higher bar to meet, and we’re still not there yet,” Dr. Downar added.
Support for the study was provided by Brain Canada, the Michael Smith Foundation for Health Research and the Vancouver Coastal Health Research Institute. Dr. Vila-Rodriguez has received research support from CIHR, Brain Canada, the Michael Smith Foundation for Health Research, the Vancouver Coastal Health Research Institute, and the Weston Brain Institute for investigator-initiated research and philanthropic support from the Seedlings Foundation; he received in-kind equipment support from MagVenture for this investigator-initiated trial; and he has received honoraria for participation on an advisory board for Janssen. Dr. Downar has served as an adviser for BrainCheck, NeuroStim TMS, and Salience Neuro Health; received research grant from CIHR, National Institute for Mental Health, Brain Canada, Canadian Biomarker Integration Network in Depression, Ontario Brain Institute, Klarman Family Foundation, Arrell Family Foundation and the Edgestone Foundation; received travel stipends from Lundbeck and ANT Neuro; and received in-kind equipment support for investigator-initiated trials from MagVenture.
A version of this article first appeared on Medscape.com.
For the first time, researchers understand what happens to the brain when patients with treatment-resistant depression receive repetitive transcranial magnetic stimulation (rTMS).
Using functional magnetic resonance imaging (fMRI), they showed that rTMS induces widespread alterations in functional connectivity in brain regions involved in emotion and motor control.
“‘How does rTMS work?’ is one of the most frequent questions I get in clinic. Providing an accurate explanation and narrative to patients is critical,” senior investigator Fidel Vila-Rodriguez, MD, PhD, director of the Non-Invasive Neurostimulation Therapies Laboratory, University of British Columbia, Vancouver, told this news organization.
“Our findings suggest that rTMS might rely on the brain’s capacity for change (neuroplasticity) to exert its effects and that rTMS effects on the brain are widespread beyond the focal area stimulated (functional network effects),” Dr. Vila-Rodriguez added.
The study was published online in the American Journal of Psychiatry.
Mechanistic insights
Although rTMS has proven efficacy for treatment-resistant depression, the mechanisms behind how it affects the brain are not well understood.
In the current study, researchers used fMRI to assess changes in functional connectivity induced by a single rTMS session in 26 women and 12 men with treatment-resistant depression.
They found that – from managing emotional responses to memory and motor control.
Following a 4-week course of rTMS, these connectivity changes predicted about 30% of the variance of improvement in scores on the Montgomery-Åsberg Depression Rating Scale after rTMS treatment.
The most robust predictive associations involved connections between prefrontal regions and motor, parietal, and insular cortices and between bilateral regions of the thalamus.
“By demonstrating this principle and identifying regions of the brain that are activated by rTMS, we can now try to understand whether this pattern can be used as a biomarker,” Dr. Vila-Rodriguez said in a news release.
“This work provides a mechanistic explanation of what rTMS does to treat depression and supports the notion that for rTMS to treat depressive symptoms a distributed change in brain activity (network or circuit base) is necessary,” he told this news organization.
With funding from the Canadian Institutes of Health Research (CIHR), the team will next see if they can use fMRI to guide rTMS at the individual level, with the ultimate goal of “personalizing” rTMS using individualized functional targets, Dr. Vila-Rodriguez said.
New generation of tms researchers
Reached for comment, Jonathan Downar, MD, PhD, department of psychiatry, University of Toronto, noted that TMS can be “very effective” for treatment-resistant depression, and it has a “very clean side effect profile compared to medications.”
What the field is trying to figure out now is “who it works for and how we can predict more effectively who’s going to benefit from it,” Dr. Downar said in an interview.
He noted that the study’s investigators are part of a “new generation of TMS researchers who are bringing new ideas into the fold and figuring out how to use brain imaging to personalize the treatment.” This study represents “a step” in that direction.
“A challenge for the field is that it’s often pretty easy to demonstrate a change at the group level, but the question is whether we can use that at the individual level. That’s a higher bar to meet, and we’re still not there yet,” Dr. Downar added.
Support for the study was provided by Brain Canada, the Michael Smith Foundation for Health Research and the Vancouver Coastal Health Research Institute. Dr. Vila-Rodriguez has received research support from CIHR, Brain Canada, the Michael Smith Foundation for Health Research, the Vancouver Coastal Health Research Institute, and the Weston Brain Institute for investigator-initiated research and philanthropic support from the Seedlings Foundation; he received in-kind equipment support from MagVenture for this investigator-initiated trial; and he has received honoraria for participation on an advisory board for Janssen. Dr. Downar has served as an adviser for BrainCheck, NeuroStim TMS, and Salience Neuro Health; received research grant from CIHR, National Institute for Mental Health, Brain Canada, Canadian Biomarker Integration Network in Depression, Ontario Brain Institute, Klarman Family Foundation, Arrell Family Foundation and the Edgestone Foundation; received travel stipends from Lundbeck and ANT Neuro; and received in-kind equipment support for investigator-initiated trials from MagVenture.
A version of this article first appeared on Medscape.com.
For the first time, researchers understand what happens to the brain when patients with treatment-resistant depression receive repetitive transcranial magnetic stimulation (rTMS).
Using functional magnetic resonance imaging (fMRI), they showed that rTMS induces widespread alterations in functional connectivity in brain regions involved in emotion and motor control.
“‘How does rTMS work?’ is one of the most frequent questions I get in clinic. Providing an accurate explanation and narrative to patients is critical,” senior investigator Fidel Vila-Rodriguez, MD, PhD, director of the Non-Invasive Neurostimulation Therapies Laboratory, University of British Columbia, Vancouver, told this news organization.
“Our findings suggest that rTMS might rely on the brain’s capacity for change (neuroplasticity) to exert its effects and that rTMS effects on the brain are widespread beyond the focal area stimulated (functional network effects),” Dr. Vila-Rodriguez added.
The study was published online in the American Journal of Psychiatry.
Mechanistic insights
Although rTMS has proven efficacy for treatment-resistant depression, the mechanisms behind how it affects the brain are not well understood.
In the current study, researchers used fMRI to assess changes in functional connectivity induced by a single rTMS session in 26 women and 12 men with treatment-resistant depression.
They found that – from managing emotional responses to memory and motor control.
Following a 4-week course of rTMS, these connectivity changes predicted about 30% of the variance of improvement in scores on the Montgomery-Åsberg Depression Rating Scale after rTMS treatment.
The most robust predictive associations involved connections between prefrontal regions and motor, parietal, and insular cortices and between bilateral regions of the thalamus.
“By demonstrating this principle and identifying regions of the brain that are activated by rTMS, we can now try to understand whether this pattern can be used as a biomarker,” Dr. Vila-Rodriguez said in a news release.
“This work provides a mechanistic explanation of what rTMS does to treat depression and supports the notion that for rTMS to treat depressive symptoms a distributed change in brain activity (network or circuit base) is necessary,” he told this news organization.
With funding from the Canadian Institutes of Health Research (CIHR), the team will next see if they can use fMRI to guide rTMS at the individual level, with the ultimate goal of “personalizing” rTMS using individualized functional targets, Dr. Vila-Rodriguez said.
New generation of tms researchers
Reached for comment, Jonathan Downar, MD, PhD, department of psychiatry, University of Toronto, noted that TMS can be “very effective” for treatment-resistant depression, and it has a “very clean side effect profile compared to medications.”
What the field is trying to figure out now is “who it works for and how we can predict more effectively who’s going to benefit from it,” Dr. Downar said in an interview.
He noted that the study’s investigators are part of a “new generation of TMS researchers who are bringing new ideas into the fold and figuring out how to use brain imaging to personalize the treatment.” This study represents “a step” in that direction.
“A challenge for the field is that it’s often pretty easy to demonstrate a change at the group level, but the question is whether we can use that at the individual level. That’s a higher bar to meet, and we’re still not there yet,” Dr. Downar added.
Support for the study was provided by Brain Canada, the Michael Smith Foundation for Health Research and the Vancouver Coastal Health Research Institute. Dr. Vila-Rodriguez has received research support from CIHR, Brain Canada, the Michael Smith Foundation for Health Research, the Vancouver Coastal Health Research Institute, and the Weston Brain Institute for investigator-initiated research and philanthropic support from the Seedlings Foundation; he received in-kind equipment support from MagVenture for this investigator-initiated trial; and he has received honoraria for participation on an advisory board for Janssen. Dr. Downar has served as an adviser for BrainCheck, NeuroStim TMS, and Salience Neuro Health; received research grant from CIHR, National Institute for Mental Health, Brain Canada, Canadian Biomarker Integration Network in Depression, Ontario Brain Institute, Klarman Family Foundation, Arrell Family Foundation and the Edgestone Foundation; received travel stipends from Lundbeck and ANT Neuro; and received in-kind equipment support for investigator-initiated trials from MagVenture.
A version of this article first appeared on Medscape.com.
Many Americans missing an opportunity to prevent dementia
(ADRD), including hypertension, low levels of physical activity, and obesity, new research shows.
Data from the Centers for Disease Control and Prevention reveal that among nearly 162,000 adults aged 45 and older who were surveyed in 2019 as part of the Behavioral Risk Factor Surveillance System (BRFSS), nearly half had high blood pressure and did not achieve aerobic physical activity recommendations. These were the two most common modifiable risk factors for ADRD.
In addition, more than one-third (35%) of adults were obese, 19% had diabetes, 18% had depression, 15% were smokers, 11% had hearing loss, and 10% were binge drinkers.
The findings were published online in the CDC’s Morbidity and Mortality Weekly Report.
A missed prevention opportunity
More than 1 in 10 (11.3%) adults surveyed reported subjective cognitive decline (SCD), an early indicator of possible future ADRD.
The prevalence of SCD increased from about 4% among adults with no modifiable risk factors for ADRD to 25% for those with four or more risk factors.
Adults with SCD were more apt to report having almost all modifiable risk factors and were more likely to report four or more risk factors (34%) than were peers without SCD (13%)
The prevalence of SCD ranged from a high of about 29% in those with depression and 25% in those with hearing loss to 11% in those who reported binge drinking.
In line with previous research, the findings indicate that American Indian or Alaska Native, Black or African American, and Hispanic populations were more likely to have modifiable risk factors for ADRD than other racial groups, the researchers reported.
The CDC’s National Healthy Brain Initiative supports culturally tailored interventions that address ADRD risk factors specifically in these populations.
In 2021, the federal government’s National Plan to Address Alzheimer’s Disease was updated to include a new goal to reduce risk factors for ADRD.
“Given the prevalence of modifiable risk factors for ADRD and anticipated growth of the older adult population and those with ADRD, this new goal has the potential to benefit a large proportion of U.S. adults,” the investigators wrote.
“In addition to helping patients discuss concerns about memory loss, health care professionals should also screen patients for modifiable risk factors, counsel patients with risk factors, and refer them to effective programs and interventions where recommended,” they advised.
A recent report from the Lancet Commission on Dementia Prevention, Intervention, and Care found that modifying 12 risk factors over the life course could delay or prevent 40% of dementia cases.
A version of this article first appeared on Medscape.com.
(ADRD), including hypertension, low levels of physical activity, and obesity, new research shows.
Data from the Centers for Disease Control and Prevention reveal that among nearly 162,000 adults aged 45 and older who were surveyed in 2019 as part of the Behavioral Risk Factor Surveillance System (BRFSS), nearly half had high blood pressure and did not achieve aerobic physical activity recommendations. These were the two most common modifiable risk factors for ADRD.
In addition, more than one-third (35%) of adults were obese, 19% had diabetes, 18% had depression, 15% were smokers, 11% had hearing loss, and 10% were binge drinkers.
The findings were published online in the CDC’s Morbidity and Mortality Weekly Report.
A missed prevention opportunity
More than 1 in 10 (11.3%) adults surveyed reported subjective cognitive decline (SCD), an early indicator of possible future ADRD.
The prevalence of SCD increased from about 4% among adults with no modifiable risk factors for ADRD to 25% for those with four or more risk factors.
Adults with SCD were more apt to report having almost all modifiable risk factors and were more likely to report four or more risk factors (34%) than were peers without SCD (13%)
The prevalence of SCD ranged from a high of about 29% in those with depression and 25% in those with hearing loss to 11% in those who reported binge drinking.
In line with previous research, the findings indicate that American Indian or Alaska Native, Black or African American, and Hispanic populations were more likely to have modifiable risk factors for ADRD than other racial groups, the researchers reported.
The CDC’s National Healthy Brain Initiative supports culturally tailored interventions that address ADRD risk factors specifically in these populations.
In 2021, the federal government’s National Plan to Address Alzheimer’s Disease was updated to include a new goal to reduce risk factors for ADRD.
“Given the prevalence of modifiable risk factors for ADRD and anticipated growth of the older adult population and those with ADRD, this new goal has the potential to benefit a large proportion of U.S. adults,” the investigators wrote.
“In addition to helping patients discuss concerns about memory loss, health care professionals should also screen patients for modifiable risk factors, counsel patients with risk factors, and refer them to effective programs and interventions where recommended,” they advised.
A recent report from the Lancet Commission on Dementia Prevention, Intervention, and Care found that modifying 12 risk factors over the life course could delay or prevent 40% of dementia cases.
A version of this article first appeared on Medscape.com.
(ADRD), including hypertension, low levels of physical activity, and obesity, new research shows.
Data from the Centers for Disease Control and Prevention reveal that among nearly 162,000 adults aged 45 and older who were surveyed in 2019 as part of the Behavioral Risk Factor Surveillance System (BRFSS), nearly half had high blood pressure and did not achieve aerobic physical activity recommendations. These were the two most common modifiable risk factors for ADRD.
In addition, more than one-third (35%) of adults were obese, 19% had diabetes, 18% had depression, 15% were smokers, 11% had hearing loss, and 10% were binge drinkers.
The findings were published online in the CDC’s Morbidity and Mortality Weekly Report.
A missed prevention opportunity
More than 1 in 10 (11.3%) adults surveyed reported subjective cognitive decline (SCD), an early indicator of possible future ADRD.
The prevalence of SCD increased from about 4% among adults with no modifiable risk factors for ADRD to 25% for those with four or more risk factors.
Adults with SCD were more apt to report having almost all modifiable risk factors and were more likely to report four or more risk factors (34%) than were peers without SCD (13%)
The prevalence of SCD ranged from a high of about 29% in those with depression and 25% in those with hearing loss to 11% in those who reported binge drinking.
In line with previous research, the findings indicate that American Indian or Alaska Native, Black or African American, and Hispanic populations were more likely to have modifiable risk factors for ADRD than other racial groups, the researchers reported.
The CDC’s National Healthy Brain Initiative supports culturally tailored interventions that address ADRD risk factors specifically in these populations.
In 2021, the federal government’s National Plan to Address Alzheimer’s Disease was updated to include a new goal to reduce risk factors for ADRD.
“Given the prevalence of modifiable risk factors for ADRD and anticipated growth of the older adult population and those with ADRD, this new goal has the potential to benefit a large proportion of U.S. adults,” the investigators wrote.
“In addition to helping patients discuss concerns about memory loss, health care professionals should also screen patients for modifiable risk factors, counsel patients with risk factors, and refer them to effective programs and interventions where recommended,” they advised.
A recent report from the Lancet Commission on Dementia Prevention, Intervention, and Care found that modifying 12 risk factors over the life course could delay or prevent 40% of dementia cases.
A version of this article first appeared on Medscape.com.
FROM MMWR
SCAI issues guidelines for PFO management, makes case for expansion
The first-ever guidelines for interventional cardiologists using percutaneous patent foramen ovale closure recommend expanding the use of the procedure beyond the Food and Drug Administration–approved indication following PFO-associated ischemic stroke, adding clarification about the use of PFO with anticoagulation and hedging against abuse and overuse of the procedure, said the chair of the guideline writing committee.
“The most important things surrounding these guidelines are to help clinicians and policymakers – third-party payers – to address PFO in patient subsets that were not included in the large randomized clinical trials that led to FDA approval,” said writing group chair Clifford J. Kavinsky, MD, PhD, chief of structural and interventional cardiology at Rush University Medical Center, Chicago.
The Society for Cardiovascular Angiography & Interventions issued the guidelines at its annual scientific sessions meeting in Atlanta and published them simultaneously in the society’s journal.
The guidelines issue strong and conditional recommendations. The former means clinicians should order the intervention for most patients; the latter means decisionmaking is more nuanced and should consider contributing factors.
The guidelines clarify patient selection for PFO closure outside the “pretty narrow” indication the FDA approved, Dr. Kavinsky said, which is for PFO-associated ischemic stroke in patients aged 18-60 years.
“So what about patients who are older than 60? What about patients who had their stroke 10 years ago?” Dr. Kavinsky asked. “Those are issues that were unanswered in the randomized clinical trials.”
The guidelines also refine recommendations about anticoagulation in these patients, including its use after PFO closure in selected patients, Dr. Kavinsky noted. “It’s the opinion of the panel that although anticoagulants may be effective, because of issues of noncompliance, because of issues of interruption of therapy by physicians for a variety of reasons, including surgery or noncompliance, that it is preferable to do a PFO device closure to giving anticoagulant therapy.”
Many of the recommendations cover PFO closure alongside antiplatelet or anticoagulation therapy. Key conditional recommendations for patients who haven’t had a PFO-related stroke are:
- Avoiding its routine use in patients with chronic migraines, prior decompression illness (DCI), thrombophilia, atrial septal aneurysm, transient ischemic attack (TIA), or deep vein thrombosis (DVT).
- Considering PFO closure in patients with platypnea-orthodeoxia syndrome (POS) with no other discernible cause of hypoxia or systemic embolism in whom other embolic causes have been ruled out.
In patients who’ve had a PFO-related stroke, the guidelines strongly recommend PFO closure versus antiplatelet therapy alone, but conditionally, not in patients with atrial fibrillation who’ve had an ischemic stroke. They also conditionally suggest PFO closure rather than long-term antiplatelet therapy alone in PFO stroke patients aged 60 and older, as well as those with thrombophilia already on antiplatelet therapy but not anticoagulation. However, the guidelines make no recommendation on PFO closure based on how much time has passed since the previous stroke.
“Furthermore,” Dr. Kavinsky said, “in patients who require lifelong anticoagulation because of recurrent DVT or recurrent pulmonary emboli or thrombopenia, if they’ve had a PFO-mediated stroke, then it’s our opinion that they should have their PFO closed in addition to taking lifelong anticoagulation because of the same issues of noncompliance and interruption of therapy.” Those are conditional recommendations.
The guideline also checks a box in the FDA labeling that mandated agreement between cardiology and neurology in patient selection. The American Academy of Neurology (AAN) issued its own guideline in 2020 for patients with stroke and PFO. In Europe, the European Society of Cardiology issued two position papers on expanded applications of PFO closure.
The recommendations on when PFO closure shouldn’t be done are noteworthy, Dr. Kavinsky said. “PFOs are present in 25% of the adult population, so the number of patients with PFO is huge and the indication for the FDA is really narrow: to reduce the risk of recurrent stroke in patients with PFO-mediated stroke. So, there’s the tremendous potential for abuse out there, of excessive procedures, of doing unnecessary procedures.”
The guidelines are a follow-up to the operator institutional requirements document SCAI issued in 2019 that set requirements for hospital offering and physicians performing PFO closure, Dr. Kavinsky added.
In an editorial accompanying the published guideline, Robert J. Sommer, MD, and Jamil A. Aboulhosn, MD, wrote that they support the recommendations “which help spotlight and clarify the growing list of potential indications for PFO closure.” They noted that the guidelines panel’s “strong” recommendations were for indications validated by randomized trials and that “conditional” recommendations were based on panelists’ experience and observational data.
“It is critical to recognize that most of these guidelines represent consensus opinion only,” wrote Dr. Sommer, who specializes in adult congenital and pediatric cardiology at Columbia University Irving Medical Center, New York, and Dr. Aboulhosn, an interventional cardiologist at Ronald Reagan University of California, Los Angeles, Medical Center. They emphasized the guidelines’ “heavy emphasis” on shared decisionmaking with patients.
Dr. Kavinsky is a principal investigator for Edwards Lifesciences, W.L. Gore and Associates, Medtronic, and Abbott. Dr. Sommer is a principal investigator and investigator in studies sponsored by W.L. Gore & Associates. Dr. Aboulhosn is a consultant to Abbott Medical.
The first-ever guidelines for interventional cardiologists using percutaneous patent foramen ovale closure recommend expanding the use of the procedure beyond the Food and Drug Administration–approved indication following PFO-associated ischemic stroke, adding clarification about the use of PFO with anticoagulation and hedging against abuse and overuse of the procedure, said the chair of the guideline writing committee.
“The most important things surrounding these guidelines are to help clinicians and policymakers – third-party payers – to address PFO in patient subsets that were not included in the large randomized clinical trials that led to FDA approval,” said writing group chair Clifford J. Kavinsky, MD, PhD, chief of structural and interventional cardiology at Rush University Medical Center, Chicago.
The Society for Cardiovascular Angiography & Interventions issued the guidelines at its annual scientific sessions meeting in Atlanta and published them simultaneously in the society’s journal.
The guidelines issue strong and conditional recommendations. The former means clinicians should order the intervention for most patients; the latter means decisionmaking is more nuanced and should consider contributing factors.
The guidelines clarify patient selection for PFO closure outside the “pretty narrow” indication the FDA approved, Dr. Kavinsky said, which is for PFO-associated ischemic stroke in patients aged 18-60 years.
“So what about patients who are older than 60? What about patients who had their stroke 10 years ago?” Dr. Kavinsky asked. “Those are issues that were unanswered in the randomized clinical trials.”
The guidelines also refine recommendations about anticoagulation in these patients, including its use after PFO closure in selected patients, Dr. Kavinsky noted. “It’s the opinion of the panel that although anticoagulants may be effective, because of issues of noncompliance, because of issues of interruption of therapy by physicians for a variety of reasons, including surgery or noncompliance, that it is preferable to do a PFO device closure to giving anticoagulant therapy.”
Many of the recommendations cover PFO closure alongside antiplatelet or anticoagulation therapy. Key conditional recommendations for patients who haven’t had a PFO-related stroke are:
- Avoiding its routine use in patients with chronic migraines, prior decompression illness (DCI), thrombophilia, atrial septal aneurysm, transient ischemic attack (TIA), or deep vein thrombosis (DVT).
- Considering PFO closure in patients with platypnea-orthodeoxia syndrome (POS) with no other discernible cause of hypoxia or systemic embolism in whom other embolic causes have been ruled out.
In patients who’ve had a PFO-related stroke, the guidelines strongly recommend PFO closure versus antiplatelet therapy alone, but conditionally, not in patients with atrial fibrillation who’ve had an ischemic stroke. They also conditionally suggest PFO closure rather than long-term antiplatelet therapy alone in PFO stroke patients aged 60 and older, as well as those with thrombophilia already on antiplatelet therapy but not anticoagulation. However, the guidelines make no recommendation on PFO closure based on how much time has passed since the previous stroke.
“Furthermore,” Dr. Kavinsky said, “in patients who require lifelong anticoagulation because of recurrent DVT or recurrent pulmonary emboli or thrombopenia, if they’ve had a PFO-mediated stroke, then it’s our opinion that they should have their PFO closed in addition to taking lifelong anticoagulation because of the same issues of noncompliance and interruption of therapy.” Those are conditional recommendations.
The guideline also checks a box in the FDA labeling that mandated agreement between cardiology and neurology in patient selection. The American Academy of Neurology (AAN) issued its own guideline in 2020 for patients with stroke and PFO. In Europe, the European Society of Cardiology issued two position papers on expanded applications of PFO closure.
The recommendations on when PFO closure shouldn’t be done are noteworthy, Dr. Kavinsky said. “PFOs are present in 25% of the adult population, so the number of patients with PFO is huge and the indication for the FDA is really narrow: to reduce the risk of recurrent stroke in patients with PFO-mediated stroke. So, there’s the tremendous potential for abuse out there, of excessive procedures, of doing unnecessary procedures.”
The guidelines are a follow-up to the operator institutional requirements document SCAI issued in 2019 that set requirements for hospital offering and physicians performing PFO closure, Dr. Kavinsky added.
In an editorial accompanying the published guideline, Robert J. Sommer, MD, and Jamil A. Aboulhosn, MD, wrote that they support the recommendations “which help spotlight and clarify the growing list of potential indications for PFO closure.” They noted that the guidelines panel’s “strong” recommendations were for indications validated by randomized trials and that “conditional” recommendations were based on panelists’ experience and observational data.
“It is critical to recognize that most of these guidelines represent consensus opinion only,” wrote Dr. Sommer, who specializes in adult congenital and pediatric cardiology at Columbia University Irving Medical Center, New York, and Dr. Aboulhosn, an interventional cardiologist at Ronald Reagan University of California, Los Angeles, Medical Center. They emphasized the guidelines’ “heavy emphasis” on shared decisionmaking with patients.
Dr. Kavinsky is a principal investigator for Edwards Lifesciences, W.L. Gore and Associates, Medtronic, and Abbott. Dr. Sommer is a principal investigator and investigator in studies sponsored by W.L. Gore & Associates. Dr. Aboulhosn is a consultant to Abbott Medical.
The first-ever guidelines for interventional cardiologists using percutaneous patent foramen ovale closure recommend expanding the use of the procedure beyond the Food and Drug Administration–approved indication following PFO-associated ischemic stroke, adding clarification about the use of PFO with anticoagulation and hedging against abuse and overuse of the procedure, said the chair of the guideline writing committee.
“The most important things surrounding these guidelines are to help clinicians and policymakers – third-party payers – to address PFO in patient subsets that were not included in the large randomized clinical trials that led to FDA approval,” said writing group chair Clifford J. Kavinsky, MD, PhD, chief of structural and interventional cardiology at Rush University Medical Center, Chicago.
The Society for Cardiovascular Angiography & Interventions issued the guidelines at its annual scientific sessions meeting in Atlanta and published them simultaneously in the society’s journal.
The guidelines issue strong and conditional recommendations. The former means clinicians should order the intervention for most patients; the latter means decisionmaking is more nuanced and should consider contributing factors.
The guidelines clarify patient selection for PFO closure outside the “pretty narrow” indication the FDA approved, Dr. Kavinsky said, which is for PFO-associated ischemic stroke in patients aged 18-60 years.
“So what about patients who are older than 60? What about patients who had their stroke 10 years ago?” Dr. Kavinsky asked. “Those are issues that were unanswered in the randomized clinical trials.”
The guidelines also refine recommendations about anticoagulation in these patients, including its use after PFO closure in selected patients, Dr. Kavinsky noted. “It’s the opinion of the panel that although anticoagulants may be effective, because of issues of noncompliance, because of issues of interruption of therapy by physicians for a variety of reasons, including surgery or noncompliance, that it is preferable to do a PFO device closure to giving anticoagulant therapy.”
Many of the recommendations cover PFO closure alongside antiplatelet or anticoagulation therapy. Key conditional recommendations for patients who haven’t had a PFO-related stroke are:
- Avoiding its routine use in patients with chronic migraines, prior decompression illness (DCI), thrombophilia, atrial septal aneurysm, transient ischemic attack (TIA), or deep vein thrombosis (DVT).
- Considering PFO closure in patients with platypnea-orthodeoxia syndrome (POS) with no other discernible cause of hypoxia or systemic embolism in whom other embolic causes have been ruled out.
In patients who’ve had a PFO-related stroke, the guidelines strongly recommend PFO closure versus antiplatelet therapy alone, but conditionally, not in patients with atrial fibrillation who’ve had an ischemic stroke. They also conditionally suggest PFO closure rather than long-term antiplatelet therapy alone in PFO stroke patients aged 60 and older, as well as those with thrombophilia already on antiplatelet therapy but not anticoagulation. However, the guidelines make no recommendation on PFO closure based on how much time has passed since the previous stroke.
“Furthermore,” Dr. Kavinsky said, “in patients who require lifelong anticoagulation because of recurrent DVT or recurrent pulmonary emboli or thrombopenia, if they’ve had a PFO-mediated stroke, then it’s our opinion that they should have their PFO closed in addition to taking lifelong anticoagulation because of the same issues of noncompliance and interruption of therapy.” Those are conditional recommendations.
The guideline also checks a box in the FDA labeling that mandated agreement between cardiology and neurology in patient selection. The American Academy of Neurology (AAN) issued its own guideline in 2020 for patients with stroke and PFO. In Europe, the European Society of Cardiology issued two position papers on expanded applications of PFO closure.
The recommendations on when PFO closure shouldn’t be done are noteworthy, Dr. Kavinsky said. “PFOs are present in 25% of the adult population, so the number of patients with PFO is huge and the indication for the FDA is really narrow: to reduce the risk of recurrent stroke in patients with PFO-mediated stroke. So, there’s the tremendous potential for abuse out there, of excessive procedures, of doing unnecessary procedures.”
The guidelines are a follow-up to the operator institutional requirements document SCAI issued in 2019 that set requirements for hospital offering and physicians performing PFO closure, Dr. Kavinsky added.
In an editorial accompanying the published guideline, Robert J. Sommer, MD, and Jamil A. Aboulhosn, MD, wrote that they support the recommendations “which help spotlight and clarify the growing list of potential indications for PFO closure.” They noted that the guidelines panel’s “strong” recommendations were for indications validated by randomized trials and that “conditional” recommendations were based on panelists’ experience and observational data.
“It is critical to recognize that most of these guidelines represent consensus opinion only,” wrote Dr. Sommer, who specializes in adult congenital and pediatric cardiology at Columbia University Irving Medical Center, New York, and Dr. Aboulhosn, an interventional cardiologist at Ronald Reagan University of California, Los Angeles, Medical Center. They emphasized the guidelines’ “heavy emphasis” on shared decisionmaking with patients.
Dr. Kavinsky is a principal investigator for Edwards Lifesciences, W.L. Gore and Associates, Medtronic, and Abbott. Dr. Sommer is a principal investigator and investigator in studies sponsored by W.L. Gore & Associates. Dr. Aboulhosn is a consultant to Abbott Medical.
FROM SCAI 2022
Advances In Neurologic Care
- Pharmacist Impact on Access to Care in an Epilepsy Clinic
- MRI Protocols for Veterans With Multiple Sclerosis
- Neuroimaging in the Era of Artificial Intelligence
- Autonomic Dysfunction in CADASIL Syndrome
- Pharmacist Impact on Access to Care in an Epilepsy Clinic
- MRI Protocols for Veterans With Multiple Sclerosis
- Neuroimaging in the Era of Artificial Intelligence
- Autonomic Dysfunction in CADASIL Syndrome
- Pharmacist Impact on Access to Care in an Epilepsy Clinic
- MRI Protocols for Veterans With Multiple Sclerosis
- Neuroimaging in the Era of Artificial Intelligence
- Autonomic Dysfunction in CADASIL Syndrome
Updated AHA/ASA guideline changes care for spontaneous intracerebral hemorrhage
Many strategies widely considered “standard care” for managing spontaneous intracerebral hemorrhage (ICH) are not as effective as previously thought and are no longer recommended in updated guidelines from the American Heart Association/American Stroke Association (ASA).
Compression stockings, antiseizure medication, and steroid treatment are among the treatments with uncertain effectiveness, the writing group says.
The 2022 Guideline for the Management of Patients With Spontaneous ICH was published online in Stroke. The 80-page document contains major changes and refinements to the 2015 guideline on ICH management.
“Advances have been made in an array of fields related to ICH, including the organization of regional health care systems, reversal of the negative effects of blood thinners, minimally invasive surgical procedures, and the underlying disease in small blood vessels,” Steven M. Greenberg, MD, PhD, chair of the guideline writing group with Harvard Medical School and Massachusetts General Hospital, both in Boston, said in a news release.
“We’ve updated sections across the board. There’s probably no area that went untouched with some tweaking and new evidence added that led to some changes in level of evidence or strength of a recommendation,” Dr. Greenberg added in an interview with this news organization.
“Each section comes with knowledge gaps, and it wasn’t hard to come up with knowledge gaps in every section,” Dr. Greenberg acknowledged.
Time-honored treatments no more?
Among the key updates are changes to some “time-honored” treatments that continue to be used with some “regularity” for patients with ICH, yet appear to confer either no benefit or harm, Dr. Greenberg said.
For example, for emergency or critical care treatment of ICH, prophylactic corticosteroids or continuous hyperosmolar therapy is not recommended, because it appears to have no benefit for outcome, while use of platelet transfusions outside the setting of emergency surgery or severe thrombocytopenia appears to worsen outcome, the authors say.
Use of graduated knee- or thigh-high compression stockings alone is not an effective prophylactic therapy for prevention of deep vein thrombosis (DVT). Instead, intermittent pneumatic compression (IPC) starting on the day of diagnosis is now recommended for DVT prophylaxis.
“This is an area where we still have a lot of exploration to do. It is unclear whether even specialized compression devices reduce the risks of deep vein thrombosis or improve the overall health of people with a brain bleed,” Dr. Greenberg said in the release.
The new guidance advises against use of antiseizure or antidepressant medications for ICH patients in whom there is no evidence of seizures or depression.
In clinical trials, antiseizure medication did not contribute to improvements in functionality or long-term seizure control, and the use of antidepressants increased the chance of bone fractures, the authors say.
The guideline also provides updated recommendations for acute reversal of anticoagulation after ICH. It highlights the use of protein complex concentrate for reversal of vitamin K antagonists, such as warfarin; idarucizumab for reversal of the thrombin inhibitor dabigatran; and andexanet alfa for reversal of factor Xa inhibitors, such as rivaroxaban, apixaban, and edoxaban.
For acute blood pressure lowering after mild to moderate ICH, treatment regimens that limit blood pressure variability and achieve smooth, sustained blood pressure control appear to reduce hematoma expansion and yield better functional outcome, the guideline says.
It also notes that minimally invasive approaches for hematoma evacuation, compared with medical management alone‚ have been shown to reduce mortality.
For patients with cerebellar hemorrhage, indications for immediate surgical evacuation with or without an external ventricular drain to reduce mortality now include larger volume (> 15 mL) in addition to previously recommended indications of neurologic deterioration, brainstem compression, and hydrocephalus, the authors note.
However, a “major knowledge gap is whether we can improve functional outcome with hematoma evacuation,” Dr. Greenberg said.
Multidisciplinary care
For rehabilitation after ICH, the guideline reinforces the importance of having a multidisciplinary team to develop a comprehensive plan for recovery.
Starting rehabilitation activities such as stretching and functional task training may be considered 24 to 48 hours following mild or moderate ICH. However, early aggressive mobilization within the first 24 hours has been linked to an increased risk of death within 14 days after an ICH, the guideline says.
Knowledge gaps include how soon it’s safe to return to work, drive, and participate in other social engagements. Recommendations on sexual activity and exercise levels that are safe after a stroke are also needed.
“People need additional help with these lifestyle changes, whether it’s moving around more, curbing their alcohol use, or eating healthier foods. This all happens after they leave the hospital, and we need to be sure we are empowering families with the information they may need to be properly supportive,” Dr. Greenberg says in the release.
The guideline points to the patient’s home caregiver as a “key and sometimes overlooked” member of the care team. It recommends psychosocial education, practical support, and training for the caregiver to improve the patient’s balance, activity level, and overall quality of life.
Opportunity for prevention?
The guideline also suggests there may be an opportunity to prevent ICH in some people through neuroimaging markers.
While neuroimaging is not routinely performed as a part of risk stratification for primary ICH risk, damage to small blood vessels that is associated with ICH may be evident on MRI that could signal future ICH risk, the guideline says.
“We added to the guidelines for the first time a section on mostly imaging markers of risk for having a first-ever hemorrhage,” Dr. Greenberg said in an interview.
“We don’t make any recommendations as to how to act on these markers because there is a knowledge gap. The hope is that we’ll see growth in our ability to predict first-ever hemorrhage and be able to do things to prevent first-ever hemorrhage,” he said.
“We believe the wide range of knowledge set forth in the new guideline will translate into meaningful improvements in ICH care,” Dr. Greenberg adds in the release.
The updated guideline has been endorsed by the American Association of Neurological Surgeons and Congress of Neurological Surgeons, the Society of Vascular and Interventional Neurology, and the Neurocritical Care Society. The American Academy of Neurology has affirmed the value of this statement as an educational tool for neurologists.
This research had no commercial funding. Dr. Greenberg has disclosed no relevant financial relationships. A complete list of disclosures for the guideline group is available with the original article.
A version of this article first appeared on Medscape.com.
Many strategies widely considered “standard care” for managing spontaneous intracerebral hemorrhage (ICH) are not as effective as previously thought and are no longer recommended in updated guidelines from the American Heart Association/American Stroke Association (ASA).
Compression stockings, antiseizure medication, and steroid treatment are among the treatments with uncertain effectiveness, the writing group says.
The 2022 Guideline for the Management of Patients With Spontaneous ICH was published online in Stroke. The 80-page document contains major changes and refinements to the 2015 guideline on ICH management.
“Advances have been made in an array of fields related to ICH, including the organization of regional health care systems, reversal of the negative effects of blood thinners, minimally invasive surgical procedures, and the underlying disease in small blood vessels,” Steven M. Greenberg, MD, PhD, chair of the guideline writing group with Harvard Medical School and Massachusetts General Hospital, both in Boston, said in a news release.
“We’ve updated sections across the board. There’s probably no area that went untouched with some tweaking and new evidence added that led to some changes in level of evidence or strength of a recommendation,” Dr. Greenberg added in an interview with this news organization.
“Each section comes with knowledge gaps, and it wasn’t hard to come up with knowledge gaps in every section,” Dr. Greenberg acknowledged.
Time-honored treatments no more?
Among the key updates are changes to some “time-honored” treatments that continue to be used with some “regularity” for patients with ICH, yet appear to confer either no benefit or harm, Dr. Greenberg said.
For example, for emergency or critical care treatment of ICH, prophylactic corticosteroids or continuous hyperosmolar therapy is not recommended, because it appears to have no benefit for outcome, while use of platelet transfusions outside the setting of emergency surgery or severe thrombocytopenia appears to worsen outcome, the authors say.
Use of graduated knee- or thigh-high compression stockings alone is not an effective prophylactic therapy for prevention of deep vein thrombosis (DVT). Instead, intermittent pneumatic compression (IPC) starting on the day of diagnosis is now recommended for DVT prophylaxis.
“This is an area where we still have a lot of exploration to do. It is unclear whether even specialized compression devices reduce the risks of deep vein thrombosis or improve the overall health of people with a brain bleed,” Dr. Greenberg said in the release.
The new guidance advises against use of antiseizure or antidepressant medications for ICH patients in whom there is no evidence of seizures or depression.
In clinical trials, antiseizure medication did not contribute to improvements in functionality or long-term seizure control, and the use of antidepressants increased the chance of bone fractures, the authors say.
The guideline also provides updated recommendations for acute reversal of anticoagulation after ICH. It highlights the use of protein complex concentrate for reversal of vitamin K antagonists, such as warfarin; idarucizumab for reversal of the thrombin inhibitor dabigatran; and andexanet alfa for reversal of factor Xa inhibitors, such as rivaroxaban, apixaban, and edoxaban.
For acute blood pressure lowering after mild to moderate ICH, treatment regimens that limit blood pressure variability and achieve smooth, sustained blood pressure control appear to reduce hematoma expansion and yield better functional outcome, the guideline says.
It also notes that minimally invasive approaches for hematoma evacuation, compared with medical management alone‚ have been shown to reduce mortality.
For patients with cerebellar hemorrhage, indications for immediate surgical evacuation with or without an external ventricular drain to reduce mortality now include larger volume (> 15 mL) in addition to previously recommended indications of neurologic deterioration, brainstem compression, and hydrocephalus, the authors note.
However, a “major knowledge gap is whether we can improve functional outcome with hematoma evacuation,” Dr. Greenberg said.
Multidisciplinary care
For rehabilitation after ICH, the guideline reinforces the importance of having a multidisciplinary team to develop a comprehensive plan for recovery.
Starting rehabilitation activities such as stretching and functional task training may be considered 24 to 48 hours following mild or moderate ICH. However, early aggressive mobilization within the first 24 hours has been linked to an increased risk of death within 14 days after an ICH, the guideline says.
Knowledge gaps include how soon it’s safe to return to work, drive, and participate in other social engagements. Recommendations on sexual activity and exercise levels that are safe after a stroke are also needed.
“People need additional help with these lifestyle changes, whether it’s moving around more, curbing their alcohol use, or eating healthier foods. This all happens after they leave the hospital, and we need to be sure we are empowering families with the information they may need to be properly supportive,” Dr. Greenberg says in the release.
The guideline points to the patient’s home caregiver as a “key and sometimes overlooked” member of the care team. It recommends psychosocial education, practical support, and training for the caregiver to improve the patient’s balance, activity level, and overall quality of life.
Opportunity for prevention?
The guideline also suggests there may be an opportunity to prevent ICH in some people through neuroimaging markers.
While neuroimaging is not routinely performed as a part of risk stratification for primary ICH risk, damage to small blood vessels that is associated with ICH may be evident on MRI that could signal future ICH risk, the guideline says.
“We added to the guidelines for the first time a section on mostly imaging markers of risk for having a first-ever hemorrhage,” Dr. Greenberg said in an interview.
“We don’t make any recommendations as to how to act on these markers because there is a knowledge gap. The hope is that we’ll see growth in our ability to predict first-ever hemorrhage and be able to do things to prevent first-ever hemorrhage,” he said.
“We believe the wide range of knowledge set forth in the new guideline will translate into meaningful improvements in ICH care,” Dr. Greenberg adds in the release.
The updated guideline has been endorsed by the American Association of Neurological Surgeons and Congress of Neurological Surgeons, the Society of Vascular and Interventional Neurology, and the Neurocritical Care Society. The American Academy of Neurology has affirmed the value of this statement as an educational tool for neurologists.
This research had no commercial funding. Dr. Greenberg has disclosed no relevant financial relationships. A complete list of disclosures for the guideline group is available with the original article.
A version of this article first appeared on Medscape.com.
Many strategies widely considered “standard care” for managing spontaneous intracerebral hemorrhage (ICH) are not as effective as previously thought and are no longer recommended in updated guidelines from the American Heart Association/American Stroke Association (ASA).
Compression stockings, antiseizure medication, and steroid treatment are among the treatments with uncertain effectiveness, the writing group says.
The 2022 Guideline for the Management of Patients With Spontaneous ICH was published online in Stroke. The 80-page document contains major changes and refinements to the 2015 guideline on ICH management.
“Advances have been made in an array of fields related to ICH, including the organization of regional health care systems, reversal of the negative effects of blood thinners, minimally invasive surgical procedures, and the underlying disease in small blood vessels,” Steven M. Greenberg, MD, PhD, chair of the guideline writing group with Harvard Medical School and Massachusetts General Hospital, both in Boston, said in a news release.
“We’ve updated sections across the board. There’s probably no area that went untouched with some tweaking and new evidence added that led to some changes in level of evidence or strength of a recommendation,” Dr. Greenberg added in an interview with this news organization.
“Each section comes with knowledge gaps, and it wasn’t hard to come up with knowledge gaps in every section,” Dr. Greenberg acknowledged.
Time-honored treatments no more?
Among the key updates are changes to some “time-honored” treatments that continue to be used with some “regularity” for patients with ICH, yet appear to confer either no benefit or harm, Dr. Greenberg said.
For example, for emergency or critical care treatment of ICH, prophylactic corticosteroids or continuous hyperosmolar therapy is not recommended, because it appears to have no benefit for outcome, while use of platelet transfusions outside the setting of emergency surgery or severe thrombocytopenia appears to worsen outcome, the authors say.
Use of graduated knee- or thigh-high compression stockings alone is not an effective prophylactic therapy for prevention of deep vein thrombosis (DVT). Instead, intermittent pneumatic compression (IPC) starting on the day of diagnosis is now recommended for DVT prophylaxis.
“This is an area where we still have a lot of exploration to do. It is unclear whether even specialized compression devices reduce the risks of deep vein thrombosis or improve the overall health of people with a brain bleed,” Dr. Greenberg said in the release.
The new guidance advises against use of antiseizure or antidepressant medications for ICH patients in whom there is no evidence of seizures or depression.
In clinical trials, antiseizure medication did not contribute to improvements in functionality or long-term seizure control, and the use of antidepressants increased the chance of bone fractures, the authors say.
The guideline also provides updated recommendations for acute reversal of anticoagulation after ICH. It highlights the use of protein complex concentrate for reversal of vitamin K antagonists, such as warfarin; idarucizumab for reversal of the thrombin inhibitor dabigatran; and andexanet alfa for reversal of factor Xa inhibitors, such as rivaroxaban, apixaban, and edoxaban.
For acute blood pressure lowering after mild to moderate ICH, treatment regimens that limit blood pressure variability and achieve smooth, sustained blood pressure control appear to reduce hematoma expansion and yield better functional outcome, the guideline says.
It also notes that minimally invasive approaches for hematoma evacuation, compared with medical management alone‚ have been shown to reduce mortality.
For patients with cerebellar hemorrhage, indications for immediate surgical evacuation with or without an external ventricular drain to reduce mortality now include larger volume (> 15 mL) in addition to previously recommended indications of neurologic deterioration, brainstem compression, and hydrocephalus, the authors note.
However, a “major knowledge gap is whether we can improve functional outcome with hematoma evacuation,” Dr. Greenberg said.
Multidisciplinary care
For rehabilitation after ICH, the guideline reinforces the importance of having a multidisciplinary team to develop a comprehensive plan for recovery.
Starting rehabilitation activities such as stretching and functional task training may be considered 24 to 48 hours following mild or moderate ICH. However, early aggressive mobilization within the first 24 hours has been linked to an increased risk of death within 14 days after an ICH, the guideline says.
Knowledge gaps include how soon it’s safe to return to work, drive, and participate in other social engagements. Recommendations on sexual activity and exercise levels that are safe after a stroke are also needed.
“People need additional help with these lifestyle changes, whether it’s moving around more, curbing their alcohol use, or eating healthier foods. This all happens after they leave the hospital, and we need to be sure we are empowering families with the information they may need to be properly supportive,” Dr. Greenberg says in the release.
The guideline points to the patient’s home caregiver as a “key and sometimes overlooked” member of the care team. It recommends psychosocial education, practical support, and training for the caregiver to improve the patient’s balance, activity level, and overall quality of life.
Opportunity for prevention?
The guideline also suggests there may be an opportunity to prevent ICH in some people through neuroimaging markers.
While neuroimaging is not routinely performed as a part of risk stratification for primary ICH risk, damage to small blood vessels that is associated with ICH may be evident on MRI that could signal future ICH risk, the guideline says.
“We added to the guidelines for the first time a section on mostly imaging markers of risk for having a first-ever hemorrhage,” Dr. Greenberg said in an interview.
“We don’t make any recommendations as to how to act on these markers because there is a knowledge gap. The hope is that we’ll see growth in our ability to predict first-ever hemorrhage and be able to do things to prevent first-ever hemorrhage,” he said.
“We believe the wide range of knowledge set forth in the new guideline will translate into meaningful improvements in ICH care,” Dr. Greenberg adds in the release.
The updated guideline has been endorsed by the American Association of Neurological Surgeons and Congress of Neurological Surgeons, the Society of Vascular and Interventional Neurology, and the Neurocritical Care Society. The American Academy of Neurology has affirmed the value of this statement as an educational tool for neurologists.
This research had no commercial funding. Dr. Greenberg has disclosed no relevant financial relationships. A complete list of disclosures for the guideline group is available with the original article.
A version of this article first appeared on Medscape.com.
The psychopathic brain: New insight
Using MRI, researchers found that the striatum was about 10% larger on average in adults with psychopathic traits than in matched control persons and that this relationship was mediated by stimulation seeking and impulsivity.
The striatum is a subcortical region of the forebrain involved in the cognitive processing of reward-related information and motivational aspects of behavior.
“Our study’s results help advance our knowledge about what underlies antisocial behavior such as psychopathy,” co-author and neurocriminologist Olivia Choy, PhD, with Nanyang Technological University, Singapore, said in a news release.
“In addition to social environmental influences, it is important to consider that there can be differences in biology – in this case, the size of brain structures – between antisocial and non-antisocial individuals,” Dr. Choy added.
The study was published online in the Journal of Psychiatric Research.
Antisocial, egocentric
Individuals with psychopathic traits typically have an egocentric and antisocial personality. They generally lack remorse for their actions or empathy for others and often have criminal tendencies.
Some prior research suggests links between psychopathy and an overactive striatum, but it was unclear what role striatal volume plays in this behavior.
For the study, investigators assessed striatal volume using MRI in 120 adults living in the community, and they assessed psychopathy using the Psychopathy Checklist – Revised.
Correlational analyses showed that increased striatal volumes were associated with more psychopathic traits (P = .001) in both men and women.
Volumetric increases were found for all subregions of the striatum in psychopathic individuals, after controlling for age, substance dependence, substance abuse, antisocial personality disorder, attention-deficit/hyperactivity disorder, social adversity, and total brain volume.
An analysis of 18 psychopathic individuals showed that striatal volumes were increased 9.4%, compared with 18 propensity-matched control persons (P = .01).
Abnormal reward processing
Stimulation seeking and impulsivity partly mediated the striatal-psychopathy relationship, accounting for 49.4% of this association.
These findings “replicate and build on initial studies indicating striatal enlargement in adults with psychopathy, yielding an updated effect size of d = 0.48,” the researchers note.
The results are “consistent with the notion that striatal abnormalities in individuals with psychopathy partly reflect increased sensation-seeking and impulsivity and support the hypothesis of abnormal reward processing in psychopathy,” they add.
“We have always known that psychopaths go to extreme lengths to seek out rewards, including criminal activities that involve property, sex, and drugs,” co-author Adrian Raine, DPhil, department of criminology, psychiatry, and psychology, University of Pennsylvania, Philadelphia, said in a news release.
“We are now finding out a neurobiological underpinning of this impulsive and stimulating behavior in the form of enlargement to the striatum, a key brain area involved in rewards,” Dr. Raine added.
What causes striatal enlargement in individuals with psychopathy still needs to be determined.
In human development, the striatum typically becomes smaller as a child matures, suggesting that psychopathy is associated with differences in brain development, the researchers suggest.
“Because biological traits, such as the size of one’s striatum, can be inherited to child from parent, these findings give added support to neurodevelopmental perspectives of psychopathy – that the brains of these offenders do not develop normally throughout childhood and adolescence,” said Dr. Raine.
Larger studies needed
Commenting on the findings for this news organization, Terrie E. Moffitt, PhD, professor of psychology, Duke University, Durham, N.C., noted that there is “general consensus among brain-imaging researchers that testing brain-behavior relations requires very large samples in the thousands and also samples of research participants who represent the full extent of variation in the population as well as possible – from rich to poor, from well to unwell, from high IQ to low IQ, from strong mental health to mental illness, etc.
“It would be grand to see this study’s provocative finding replicated in a large, representative sampling design,” Dr. Moffitt said.
The study was supported in part by the National Institutes of Health. Dr. Choy, Dr. Raine, and Dr. Moffitt have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Using MRI, researchers found that the striatum was about 10% larger on average in adults with psychopathic traits than in matched control persons and that this relationship was mediated by stimulation seeking and impulsivity.
The striatum is a subcortical region of the forebrain involved in the cognitive processing of reward-related information and motivational aspects of behavior.
“Our study’s results help advance our knowledge about what underlies antisocial behavior such as psychopathy,” co-author and neurocriminologist Olivia Choy, PhD, with Nanyang Technological University, Singapore, said in a news release.
“In addition to social environmental influences, it is important to consider that there can be differences in biology – in this case, the size of brain structures – between antisocial and non-antisocial individuals,” Dr. Choy added.
The study was published online in the Journal of Psychiatric Research.
Antisocial, egocentric
Individuals with psychopathic traits typically have an egocentric and antisocial personality. They generally lack remorse for their actions or empathy for others and often have criminal tendencies.
Some prior research suggests links between psychopathy and an overactive striatum, but it was unclear what role striatal volume plays in this behavior.
For the study, investigators assessed striatal volume using MRI in 120 adults living in the community, and they assessed psychopathy using the Psychopathy Checklist – Revised.
Correlational analyses showed that increased striatal volumes were associated with more psychopathic traits (P = .001) in both men and women.
Volumetric increases were found for all subregions of the striatum in psychopathic individuals, after controlling for age, substance dependence, substance abuse, antisocial personality disorder, attention-deficit/hyperactivity disorder, social adversity, and total brain volume.
An analysis of 18 psychopathic individuals showed that striatal volumes were increased 9.4%, compared with 18 propensity-matched control persons (P = .01).
Abnormal reward processing
Stimulation seeking and impulsivity partly mediated the striatal-psychopathy relationship, accounting for 49.4% of this association.
These findings “replicate and build on initial studies indicating striatal enlargement in adults with psychopathy, yielding an updated effect size of d = 0.48,” the researchers note.
The results are “consistent with the notion that striatal abnormalities in individuals with psychopathy partly reflect increased sensation-seeking and impulsivity and support the hypothesis of abnormal reward processing in psychopathy,” they add.
“We have always known that psychopaths go to extreme lengths to seek out rewards, including criminal activities that involve property, sex, and drugs,” co-author Adrian Raine, DPhil, department of criminology, psychiatry, and psychology, University of Pennsylvania, Philadelphia, said in a news release.
“We are now finding out a neurobiological underpinning of this impulsive and stimulating behavior in the form of enlargement to the striatum, a key brain area involved in rewards,” Dr. Raine added.
What causes striatal enlargement in individuals with psychopathy still needs to be determined.
In human development, the striatum typically becomes smaller as a child matures, suggesting that psychopathy is associated with differences in brain development, the researchers suggest.
“Because biological traits, such as the size of one’s striatum, can be inherited to child from parent, these findings give added support to neurodevelopmental perspectives of psychopathy – that the brains of these offenders do not develop normally throughout childhood and adolescence,” said Dr. Raine.
Larger studies needed
Commenting on the findings for this news organization, Terrie E. Moffitt, PhD, professor of psychology, Duke University, Durham, N.C., noted that there is “general consensus among brain-imaging researchers that testing brain-behavior relations requires very large samples in the thousands and also samples of research participants who represent the full extent of variation in the population as well as possible – from rich to poor, from well to unwell, from high IQ to low IQ, from strong mental health to mental illness, etc.
“It would be grand to see this study’s provocative finding replicated in a large, representative sampling design,” Dr. Moffitt said.
The study was supported in part by the National Institutes of Health. Dr. Choy, Dr. Raine, and Dr. Moffitt have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Using MRI, researchers found that the striatum was about 10% larger on average in adults with psychopathic traits than in matched control persons and that this relationship was mediated by stimulation seeking and impulsivity.
The striatum is a subcortical region of the forebrain involved in the cognitive processing of reward-related information and motivational aspects of behavior.
“Our study’s results help advance our knowledge about what underlies antisocial behavior such as psychopathy,” co-author and neurocriminologist Olivia Choy, PhD, with Nanyang Technological University, Singapore, said in a news release.
“In addition to social environmental influences, it is important to consider that there can be differences in biology – in this case, the size of brain structures – between antisocial and non-antisocial individuals,” Dr. Choy added.
The study was published online in the Journal of Psychiatric Research.
Antisocial, egocentric
Individuals with psychopathic traits typically have an egocentric and antisocial personality. They generally lack remorse for their actions or empathy for others and often have criminal tendencies.
Some prior research suggests links between psychopathy and an overactive striatum, but it was unclear what role striatal volume plays in this behavior.
For the study, investigators assessed striatal volume using MRI in 120 adults living in the community, and they assessed psychopathy using the Psychopathy Checklist – Revised.
Correlational analyses showed that increased striatal volumes were associated with more psychopathic traits (P = .001) in both men and women.
Volumetric increases were found for all subregions of the striatum in psychopathic individuals, after controlling for age, substance dependence, substance abuse, antisocial personality disorder, attention-deficit/hyperactivity disorder, social adversity, and total brain volume.
An analysis of 18 psychopathic individuals showed that striatal volumes were increased 9.4%, compared with 18 propensity-matched control persons (P = .01).
Abnormal reward processing
Stimulation seeking and impulsivity partly mediated the striatal-psychopathy relationship, accounting for 49.4% of this association.
These findings “replicate and build on initial studies indicating striatal enlargement in adults with psychopathy, yielding an updated effect size of d = 0.48,” the researchers note.
The results are “consistent with the notion that striatal abnormalities in individuals with psychopathy partly reflect increased sensation-seeking and impulsivity and support the hypothesis of abnormal reward processing in psychopathy,” they add.
“We have always known that psychopaths go to extreme lengths to seek out rewards, including criminal activities that involve property, sex, and drugs,” co-author Adrian Raine, DPhil, department of criminology, psychiatry, and psychology, University of Pennsylvania, Philadelphia, said in a news release.
“We are now finding out a neurobiological underpinning of this impulsive and stimulating behavior in the form of enlargement to the striatum, a key brain area involved in rewards,” Dr. Raine added.
What causes striatal enlargement in individuals with psychopathy still needs to be determined.
In human development, the striatum typically becomes smaller as a child matures, suggesting that psychopathy is associated with differences in brain development, the researchers suggest.
“Because biological traits, such as the size of one’s striatum, can be inherited to child from parent, these findings give added support to neurodevelopmental perspectives of psychopathy – that the brains of these offenders do not develop normally throughout childhood and adolescence,” said Dr. Raine.
Larger studies needed
Commenting on the findings for this news organization, Terrie E. Moffitt, PhD, professor of psychology, Duke University, Durham, N.C., noted that there is “general consensus among brain-imaging researchers that testing brain-behavior relations requires very large samples in the thousands and also samples of research participants who represent the full extent of variation in the population as well as possible – from rich to poor, from well to unwell, from high IQ to low IQ, from strong mental health to mental illness, etc.
“It would be grand to see this study’s provocative finding replicated in a large, representative sampling design,” Dr. Moffitt said.
The study was supported in part by the National Institutes of Health. Dr. Choy, Dr. Raine, and Dr. Moffitt have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Neuropsychiatric risks of COVID-19: New data
The neuropsychiatric ramifications of severe COVID-19 infection appear to be no different than for other severe acute respiratory infections (SARI).
This suggests that disease severity, rather than pathogen, is the most relevant factor in new-onset neuropsychiatric illness, the investigators note.
The risk of new-onset neuropsychological illness after severe COVID-19 infection are “substantial, but similar to those after other severe respiratory infections,” study investigator Peter Watkinson, MD, Nuffield Department of Clinical Neurosciences, University of Oxford, and John Radcliffe Hospital, Oxford, England, told this news organization.
The study was published online in JAMA Psychiatry.
Significant mental health burden
Research has shown a significant burden of neuropsychological illness after severe COVID-19 infection. However, it’s unclear how this risk compares to SARI.
To investigate, Dr. Watkinson and colleagues evaluated electronic health record data on more than 8.3 million adults, including 16,679 (0.02%) who survived a hospital admission for SARI and 32,525 (0.03%) who survived a hospital stay for COVID-19.
Compared with the remaining population, risks of new anxiety disorder, dementia, psychotic disorder, depression, and bipolar disorder diagnoses were significantly and similarly increased in adults surviving hospitalization for either COVID-19 or SARI.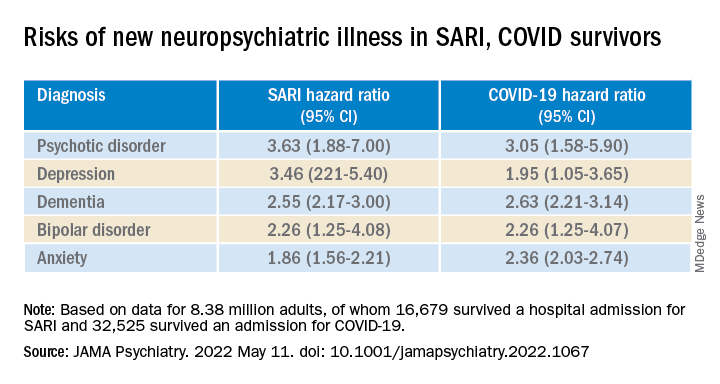
Compared with the wider population, survivors of severe SARI or COVID-19 were also at increased risk of starting treatment with antidepressants, hypnotics/anxiolytics, or antipsychotics.
When comparing survivors of SARI hospitalization to survivors of COVID-19 hospitalization, no significant differences were observed in the postdischarge rates of new-onset anxiety disorder, dementia, depression, or bipolar affective disorder.
The SARI and COVID groups also did not differ in terms of their postdischarge risks of antidepressant or hypnotic/anxiolytic use, but the COVID survivors had a 20% lower risk of starting an antipsychotic.
“In this cohort study, SARI were found to be associated with significant postacute neuropsychiatric morbidity, for which COVID-19 is not distinctly different,” Dr. Watkinson and colleagues write.
“These results may help refine our understanding of the post–severe COVID-19 phenotype and may inform post-discharge support for patients requiring hospital-based and intensive care for SARI regardless of causative pathogen,” they write.
Caveats, cautionary notes
Kevin McConway, PhD, emeritus professor of applied statistics at the Open University in Milton Keynes, England, described the study as “impressive.” However, he pointed out that the study’s observational design is a limitation.
“One can never be absolutely certain about the interpretation of findings of an observational study. What the research can’t tell us is what caused the increased psychiatric risks for people hospitalized with COVID-19 or some other serious respiratory disease,” Dr. McConway said.
“It can’t tell us what might happen in the future, when, we all hope, many fewer are being hospitalized with COVID-19 than was the case in those first two waves, and the current backlog of provision of some health services has decreased,” he added.
“So we can’t just say that, in general, serious COVID-19 has much the same neuropsychiatric consequences as other very serious respiratory illness. Maybe it does, maybe it doesn’t,” Dr. McConway cautioned.
Max Taquet, PhD, with the University of Oxford, noted that the study is limited to hospitalized adult patients, leaving open the question of risk in nonhospitalized individuals – which is the overwhelming majority of patients with COVID-19 – or in children.
Whether the neuropsychiatric risks have remained the same since the emergence of the Omicron variant also remains “an open question since all patients in this study were diagnosed before July 2021,” Dr. Taquet said in statement.
The study was funded by the Wellcome Trust, the John Fell Oxford University Press Research Fund, the Oxford Wellcome Institutional Strategic Support Fund and Cancer Research UK, through the Cancer Research UK Oxford Centre. Dr. Watkinson disclosed grants from the National Institute for Health Research and Sensyne Health outside the submitted work; and serving as chief medical officer for Sensyne Health prior to this work, as well as holding shares in the company. Dr. McConway is a trustee of the UK Science Media Centre and a member of its advisory committee. His comments were provided in his capacity as an independent professional statistician. Dr. Taquet has worked on similar studies trying to identify, quantify, and specify the neurological and psychiatric consequences of COVID-19.
A version of this article first appeared on Medscape.com.
The neuropsychiatric ramifications of severe COVID-19 infection appear to be no different than for other severe acute respiratory infections (SARI).
This suggests that disease severity, rather than pathogen, is the most relevant factor in new-onset neuropsychiatric illness, the investigators note.
The risk of new-onset neuropsychological illness after severe COVID-19 infection are “substantial, but similar to those after other severe respiratory infections,” study investigator Peter Watkinson, MD, Nuffield Department of Clinical Neurosciences, University of Oxford, and John Radcliffe Hospital, Oxford, England, told this news organization.
The study was published online in JAMA Psychiatry.
Significant mental health burden
Research has shown a significant burden of neuropsychological illness after severe COVID-19 infection. However, it’s unclear how this risk compares to SARI.
To investigate, Dr. Watkinson and colleagues evaluated electronic health record data on more than 8.3 million adults, including 16,679 (0.02%) who survived a hospital admission for SARI and 32,525 (0.03%) who survived a hospital stay for COVID-19.
Compared with the remaining population, risks of new anxiety disorder, dementia, psychotic disorder, depression, and bipolar disorder diagnoses were significantly and similarly increased in adults surviving hospitalization for either COVID-19 or SARI.
Compared with the wider population, survivors of severe SARI or COVID-19 were also at increased risk of starting treatment with antidepressants, hypnotics/anxiolytics, or antipsychotics.
When comparing survivors of SARI hospitalization to survivors of COVID-19 hospitalization, no significant differences were observed in the postdischarge rates of new-onset anxiety disorder, dementia, depression, or bipolar affective disorder.
The SARI and COVID groups also did not differ in terms of their postdischarge risks of antidepressant or hypnotic/anxiolytic use, but the COVID survivors had a 20% lower risk of starting an antipsychotic.
“In this cohort study, SARI were found to be associated with significant postacute neuropsychiatric morbidity, for which COVID-19 is not distinctly different,” Dr. Watkinson and colleagues write.
“These results may help refine our understanding of the post–severe COVID-19 phenotype and may inform post-discharge support for patients requiring hospital-based and intensive care for SARI regardless of causative pathogen,” they write.
Caveats, cautionary notes
Kevin McConway, PhD, emeritus professor of applied statistics at the Open University in Milton Keynes, England, described the study as “impressive.” However, he pointed out that the study’s observational design is a limitation.
“One can never be absolutely certain about the interpretation of findings of an observational study. What the research can’t tell us is what caused the increased psychiatric risks for people hospitalized with COVID-19 or some other serious respiratory disease,” Dr. McConway said.
“It can’t tell us what might happen in the future, when, we all hope, many fewer are being hospitalized with COVID-19 than was the case in those first two waves, and the current backlog of provision of some health services has decreased,” he added.
“So we can’t just say that, in general, serious COVID-19 has much the same neuropsychiatric consequences as other very serious respiratory illness. Maybe it does, maybe it doesn’t,” Dr. McConway cautioned.
Max Taquet, PhD, with the University of Oxford, noted that the study is limited to hospitalized adult patients, leaving open the question of risk in nonhospitalized individuals – which is the overwhelming majority of patients with COVID-19 – or in children.
Whether the neuropsychiatric risks have remained the same since the emergence of the Omicron variant also remains “an open question since all patients in this study were diagnosed before July 2021,” Dr. Taquet said in statement.
The study was funded by the Wellcome Trust, the John Fell Oxford University Press Research Fund, the Oxford Wellcome Institutional Strategic Support Fund and Cancer Research UK, through the Cancer Research UK Oxford Centre. Dr. Watkinson disclosed grants from the National Institute for Health Research and Sensyne Health outside the submitted work; and serving as chief medical officer for Sensyne Health prior to this work, as well as holding shares in the company. Dr. McConway is a trustee of the UK Science Media Centre and a member of its advisory committee. His comments were provided in his capacity as an independent professional statistician. Dr. Taquet has worked on similar studies trying to identify, quantify, and specify the neurological and psychiatric consequences of COVID-19.
A version of this article first appeared on Medscape.com.
The neuropsychiatric ramifications of severe COVID-19 infection appear to be no different than for other severe acute respiratory infections (SARI).
This suggests that disease severity, rather than pathogen, is the most relevant factor in new-onset neuropsychiatric illness, the investigators note.
The risk of new-onset neuropsychological illness after severe COVID-19 infection are “substantial, but similar to those after other severe respiratory infections,” study investigator Peter Watkinson, MD, Nuffield Department of Clinical Neurosciences, University of Oxford, and John Radcliffe Hospital, Oxford, England, told this news organization.
The study was published online in JAMA Psychiatry.
Significant mental health burden
Research has shown a significant burden of neuropsychological illness after severe COVID-19 infection. However, it’s unclear how this risk compares to SARI.
To investigate, Dr. Watkinson and colleagues evaluated electronic health record data on more than 8.3 million adults, including 16,679 (0.02%) who survived a hospital admission for SARI and 32,525 (0.03%) who survived a hospital stay for COVID-19.
Compared with the remaining population, risks of new anxiety disorder, dementia, psychotic disorder, depression, and bipolar disorder diagnoses were significantly and similarly increased in adults surviving hospitalization for either COVID-19 or SARI.
Compared with the wider population, survivors of severe SARI or COVID-19 were also at increased risk of starting treatment with antidepressants, hypnotics/anxiolytics, or antipsychotics.
When comparing survivors of SARI hospitalization to survivors of COVID-19 hospitalization, no significant differences were observed in the postdischarge rates of new-onset anxiety disorder, dementia, depression, or bipolar affective disorder.
The SARI and COVID groups also did not differ in terms of their postdischarge risks of antidepressant or hypnotic/anxiolytic use, but the COVID survivors had a 20% lower risk of starting an antipsychotic.
“In this cohort study, SARI were found to be associated with significant postacute neuropsychiatric morbidity, for which COVID-19 is not distinctly different,” Dr. Watkinson and colleagues write.
“These results may help refine our understanding of the post–severe COVID-19 phenotype and may inform post-discharge support for patients requiring hospital-based and intensive care for SARI regardless of causative pathogen,” they write.
Caveats, cautionary notes
Kevin McConway, PhD, emeritus professor of applied statistics at the Open University in Milton Keynes, England, described the study as “impressive.” However, he pointed out that the study’s observational design is a limitation.
“One can never be absolutely certain about the interpretation of findings of an observational study. What the research can’t tell us is what caused the increased psychiatric risks for people hospitalized with COVID-19 or some other serious respiratory disease,” Dr. McConway said.
“It can’t tell us what might happen in the future, when, we all hope, many fewer are being hospitalized with COVID-19 than was the case in those first two waves, and the current backlog of provision of some health services has decreased,” he added.
“So we can’t just say that, in general, serious COVID-19 has much the same neuropsychiatric consequences as other very serious respiratory illness. Maybe it does, maybe it doesn’t,” Dr. McConway cautioned.
Max Taquet, PhD, with the University of Oxford, noted that the study is limited to hospitalized adult patients, leaving open the question of risk in nonhospitalized individuals – which is the overwhelming majority of patients with COVID-19 – or in children.
Whether the neuropsychiatric risks have remained the same since the emergence of the Omicron variant also remains “an open question since all patients in this study were diagnosed before July 2021,” Dr. Taquet said in statement.
The study was funded by the Wellcome Trust, the John Fell Oxford University Press Research Fund, the Oxford Wellcome Institutional Strategic Support Fund and Cancer Research UK, through the Cancer Research UK Oxford Centre. Dr. Watkinson disclosed grants from the National Institute for Health Research and Sensyne Health outside the submitted work; and serving as chief medical officer for Sensyne Health prior to this work, as well as holding shares in the company. Dr. McConway is a trustee of the UK Science Media Centre and a member of its advisory committee. His comments were provided in his capacity as an independent professional statistician. Dr. Taquet has worked on similar studies trying to identify, quantify, and specify the neurological and psychiatric consequences of COVID-19.
A version of this article first appeared on Medscape.com.
Most COVID-19 survivors return to work within 2 years
The burden of persistent COVID-19 symptoms appeared to improve over time, but a higher percentage of former patients reported poor health, compared with the general population. This suggests that some patients need more time to completely recover from COVID-19, wrote the authors of the new study, which was published in The Lancet Respiratory Medicine. Previous research has shown that the health effects of COVID-19 last for up to a year, but data from longer-term studies are limited, said Lixue Huang, MD, of Capital Medical University, Beijing, one of the study authors, and colleagues.
Methods and results
In the new study, the researchers reviewed data from 1,192 adult patients who were discharged from the hospital after surviving COVID-19 between Jan. 7, 2020, and May 29, 2020. The researchers measured the participants’ health outcomes at 6 months, 12 months, and 2 years after their onset of symptoms. A community-based dataset of 3,383 adults with no history of COVID-19 served as controls to measure the recovery of the COVID-19 patients. The median age of the patients at the time of hospital discharge was 57 years, and 46% were women. The median follow-up time after the onset of symptoms was 185 days, 349 days, and 685 days for the 6-month, 12-month, and 2-year visits, respectively. The researchers measured health outcomes using a 6-min walking distance (6MWD) test, laboratory tests, and questionnaires about symptoms, mental health, health-related quality of life, returning to work, and health care use since leaving the hospital.
Overall, the proportion of COVID-19 survivors with at least one symptom decreased from 68% at 6 months to 55% at 2 years (P < .0001). The most frequent symptoms were fatigue and muscle weakness, reported by approximately one-third of the patients (31%); sleep problems also were reported by 31% of the patients.
The proportion of individuals with poor results on the 6MWD decreased continuously over time, not only in COVID-19 survivors overall, but also in three subgroups of varying initial disease severity. Of the 494 survivors who reported working before becoming ill, 438 (89%) had returned to their original jobs 2 years later. The most common reasons for not returning to work were decreased physical function, unwillingness to return, and unemployment, the researchers noted.
However, at 2 years, COVID-19 survivors reported more pain and discomfort, as well as more anxiety and depression, compared with the controls (23% vs. 5% and 12% vs. 5%, respectively).
In addition, significantly more survivors who needed high levels of respiratory support while hospitalized had lung diffusion impairment (65%), reduced residual volume (62%), and total lung capacity (39%), compared with matched controls (36%, 20%, and 6%, respectively) at 2 years.
Long-COVID concerns
Approximately half of the survivors had symptoms of long COVID at 2 years. These individuals were more likely to report pain or discomfort or anxiety or depression, as well as mobility problems, compared to survivors without long COVID. Participants with long-COVID symptoms were more than twice as likely to have an outpatient clinic visit (odds ratio, 2.82), and not quite twice as likely to be rehospitalized (OR, 1.64).
“We found that [health-related quality of life], exercise capacity, and mental health continued to improve throughout the 2 years regardless of initial disease severity, but about half still had symptomatic sequelae at 2 years,” the researchers wrote in their paper.
Findings can inform doctor-patient discussions
“We are increasingly recognizing that the health effects of COVID-19 may persist beyond acute illness, therefore this is a timely study to assess the long-term impact of COVID-19 with a long follow-up period,” said Suman Pal, MD, an internal medicine physician at the University of New Mexico, Albuquerque, in an interview.
The findings are consistent with the existing literature, said Dr. Pal, who was not involved in the study. The data from the study “can help clinicians have discussions regarding expected recovery and long-term prognosis for patients with COVID-19,” he noted.
What patients should know is that “studies such as this can help COVID-19 survivors understand and monitor persistent symptoms they may experience, and bring them to the attention of their clinicians,” said Dr. Pal.
However, “As a single-center study with high attrition of subjects during the study period, the findings may not be generalizable,” Dr. Pal emphasized. “Larger-scale studies and patient registries distributed over different geographical areas and time periods will help obtain a better understanding of the nature and prevalence of long COVID,” he said.
The study findings were limited by several factors, including the lack of formerly hospitalized controls with respiratory infections other than COVID-19 to determine which outcomes are COVID-19 specific, the researchers noted. Other limitations included the use of data from only patients at a single center, and from the early stages of the pandemic, as well as the use of self-reports for comorbidities and health outcomes, they said.
However, the results represent the longest-known published longitudinal follow-up of patients who recovered from acute COVID-19, the researchers emphasized. Study strengths included the large sample size, longitudinal design, and long-term follow-up with non-COVID controls to determine outcomes. The researchers noted their plans to conduct annual follow-ups in the current study population. They added that more research is needed to explore rehabilitation programs to promote recovery for COVID-19 survivors and to reduce the effects of long COVID.
The study was supported by the Chinese Academy of Medical Sciences, National Natural Science Foundation of China, National Key Research and Development Program of China, National Administration of Traditional Chinese Medicine, Major Projects of National Science and Technology on New Drug Creation and Development of Pulmonary Tuberculosis, China Evergrande Group, Jack Ma Foundation, Sino Biopharmaceutical, Ping An Insurance (Group), and New Sunshine Charity Foundation. The researchers and Dr. Pal had no financial conflicts to disclose.
This article was updated on 5/16/2022.
The burden of persistent COVID-19 symptoms appeared to improve over time, but a higher percentage of former patients reported poor health, compared with the general population. This suggests that some patients need more time to completely recover from COVID-19, wrote the authors of the new study, which was published in The Lancet Respiratory Medicine. Previous research has shown that the health effects of COVID-19 last for up to a year, but data from longer-term studies are limited, said Lixue Huang, MD, of Capital Medical University, Beijing, one of the study authors, and colleagues.
Methods and results
In the new study, the researchers reviewed data from 1,192 adult patients who were discharged from the hospital after surviving COVID-19 between Jan. 7, 2020, and May 29, 2020. The researchers measured the participants’ health outcomes at 6 months, 12 months, and 2 years after their onset of symptoms. A community-based dataset of 3,383 adults with no history of COVID-19 served as controls to measure the recovery of the COVID-19 patients. The median age of the patients at the time of hospital discharge was 57 years, and 46% were women. The median follow-up time after the onset of symptoms was 185 days, 349 days, and 685 days for the 6-month, 12-month, and 2-year visits, respectively. The researchers measured health outcomes using a 6-min walking distance (6MWD) test, laboratory tests, and questionnaires about symptoms, mental health, health-related quality of life, returning to work, and health care use since leaving the hospital.
Overall, the proportion of COVID-19 survivors with at least one symptom decreased from 68% at 6 months to 55% at 2 years (P < .0001). The most frequent symptoms were fatigue and muscle weakness, reported by approximately one-third of the patients (31%); sleep problems also were reported by 31% of the patients.
The proportion of individuals with poor results on the 6MWD decreased continuously over time, not only in COVID-19 survivors overall, but also in three subgroups of varying initial disease severity. Of the 494 survivors who reported working before becoming ill, 438 (89%) had returned to their original jobs 2 years later. The most common reasons for not returning to work were decreased physical function, unwillingness to return, and unemployment, the researchers noted.
However, at 2 years, COVID-19 survivors reported more pain and discomfort, as well as more anxiety and depression, compared with the controls (23% vs. 5% and 12% vs. 5%, respectively).
In addition, significantly more survivors who needed high levels of respiratory support while hospitalized had lung diffusion impairment (65%), reduced residual volume (62%), and total lung capacity (39%), compared with matched controls (36%, 20%, and 6%, respectively) at 2 years.
Long-COVID concerns
Approximately half of the survivors had symptoms of long COVID at 2 years. These individuals were more likely to report pain or discomfort or anxiety or depression, as well as mobility problems, compared to survivors without long COVID. Participants with long-COVID symptoms were more than twice as likely to have an outpatient clinic visit (odds ratio, 2.82), and not quite twice as likely to be rehospitalized (OR, 1.64).
“We found that [health-related quality of life], exercise capacity, and mental health continued to improve throughout the 2 years regardless of initial disease severity, but about half still had symptomatic sequelae at 2 years,” the researchers wrote in their paper.
Findings can inform doctor-patient discussions
“We are increasingly recognizing that the health effects of COVID-19 may persist beyond acute illness, therefore this is a timely study to assess the long-term impact of COVID-19 with a long follow-up period,” said Suman Pal, MD, an internal medicine physician at the University of New Mexico, Albuquerque, in an interview.
The findings are consistent with the existing literature, said Dr. Pal, who was not involved in the study. The data from the study “can help clinicians have discussions regarding expected recovery and long-term prognosis for patients with COVID-19,” he noted.
What patients should know is that “studies such as this can help COVID-19 survivors understand and monitor persistent symptoms they may experience, and bring them to the attention of their clinicians,” said Dr. Pal.
However, “As a single-center study with high attrition of subjects during the study period, the findings may not be generalizable,” Dr. Pal emphasized. “Larger-scale studies and patient registries distributed over different geographical areas and time periods will help obtain a better understanding of the nature and prevalence of long COVID,” he said.
The study findings were limited by several factors, including the lack of formerly hospitalized controls with respiratory infections other than COVID-19 to determine which outcomes are COVID-19 specific, the researchers noted. Other limitations included the use of data from only patients at a single center, and from the early stages of the pandemic, as well as the use of self-reports for comorbidities and health outcomes, they said.
However, the results represent the longest-known published longitudinal follow-up of patients who recovered from acute COVID-19, the researchers emphasized. Study strengths included the large sample size, longitudinal design, and long-term follow-up with non-COVID controls to determine outcomes. The researchers noted their plans to conduct annual follow-ups in the current study population. They added that more research is needed to explore rehabilitation programs to promote recovery for COVID-19 survivors and to reduce the effects of long COVID.
The study was supported by the Chinese Academy of Medical Sciences, National Natural Science Foundation of China, National Key Research and Development Program of China, National Administration of Traditional Chinese Medicine, Major Projects of National Science and Technology on New Drug Creation and Development of Pulmonary Tuberculosis, China Evergrande Group, Jack Ma Foundation, Sino Biopharmaceutical, Ping An Insurance (Group), and New Sunshine Charity Foundation. The researchers and Dr. Pal had no financial conflicts to disclose.
This article was updated on 5/16/2022.
The burden of persistent COVID-19 symptoms appeared to improve over time, but a higher percentage of former patients reported poor health, compared with the general population. This suggests that some patients need more time to completely recover from COVID-19, wrote the authors of the new study, which was published in The Lancet Respiratory Medicine. Previous research has shown that the health effects of COVID-19 last for up to a year, but data from longer-term studies are limited, said Lixue Huang, MD, of Capital Medical University, Beijing, one of the study authors, and colleagues.
Methods and results
In the new study, the researchers reviewed data from 1,192 adult patients who were discharged from the hospital after surviving COVID-19 between Jan. 7, 2020, and May 29, 2020. The researchers measured the participants’ health outcomes at 6 months, 12 months, and 2 years after their onset of symptoms. A community-based dataset of 3,383 adults with no history of COVID-19 served as controls to measure the recovery of the COVID-19 patients. The median age of the patients at the time of hospital discharge was 57 years, and 46% were women. The median follow-up time after the onset of symptoms was 185 days, 349 days, and 685 days for the 6-month, 12-month, and 2-year visits, respectively. The researchers measured health outcomes using a 6-min walking distance (6MWD) test, laboratory tests, and questionnaires about symptoms, mental health, health-related quality of life, returning to work, and health care use since leaving the hospital.
Overall, the proportion of COVID-19 survivors with at least one symptom decreased from 68% at 6 months to 55% at 2 years (P < .0001). The most frequent symptoms were fatigue and muscle weakness, reported by approximately one-third of the patients (31%); sleep problems also were reported by 31% of the patients.
The proportion of individuals with poor results on the 6MWD decreased continuously over time, not only in COVID-19 survivors overall, but also in three subgroups of varying initial disease severity. Of the 494 survivors who reported working before becoming ill, 438 (89%) had returned to their original jobs 2 years later. The most common reasons for not returning to work were decreased physical function, unwillingness to return, and unemployment, the researchers noted.
However, at 2 years, COVID-19 survivors reported more pain and discomfort, as well as more anxiety and depression, compared with the controls (23% vs. 5% and 12% vs. 5%, respectively).
In addition, significantly more survivors who needed high levels of respiratory support while hospitalized had lung diffusion impairment (65%), reduced residual volume (62%), and total lung capacity (39%), compared with matched controls (36%, 20%, and 6%, respectively) at 2 years.
Long-COVID concerns
Approximately half of the survivors had symptoms of long COVID at 2 years. These individuals were more likely to report pain or discomfort or anxiety or depression, as well as mobility problems, compared to survivors without long COVID. Participants with long-COVID symptoms were more than twice as likely to have an outpatient clinic visit (odds ratio, 2.82), and not quite twice as likely to be rehospitalized (OR, 1.64).
“We found that [health-related quality of life], exercise capacity, and mental health continued to improve throughout the 2 years regardless of initial disease severity, but about half still had symptomatic sequelae at 2 years,” the researchers wrote in their paper.
Findings can inform doctor-patient discussions
“We are increasingly recognizing that the health effects of COVID-19 may persist beyond acute illness, therefore this is a timely study to assess the long-term impact of COVID-19 with a long follow-up period,” said Suman Pal, MD, an internal medicine physician at the University of New Mexico, Albuquerque, in an interview.
The findings are consistent with the existing literature, said Dr. Pal, who was not involved in the study. The data from the study “can help clinicians have discussions regarding expected recovery and long-term prognosis for patients with COVID-19,” he noted.
What patients should know is that “studies such as this can help COVID-19 survivors understand and monitor persistent symptoms they may experience, and bring them to the attention of their clinicians,” said Dr. Pal.
However, “As a single-center study with high attrition of subjects during the study period, the findings may not be generalizable,” Dr. Pal emphasized. “Larger-scale studies and patient registries distributed over different geographical areas and time periods will help obtain a better understanding of the nature and prevalence of long COVID,” he said.
The study findings were limited by several factors, including the lack of formerly hospitalized controls with respiratory infections other than COVID-19 to determine which outcomes are COVID-19 specific, the researchers noted. Other limitations included the use of data from only patients at a single center, and from the early stages of the pandemic, as well as the use of self-reports for comorbidities and health outcomes, they said.
However, the results represent the longest-known published longitudinal follow-up of patients who recovered from acute COVID-19, the researchers emphasized. Study strengths included the large sample size, longitudinal design, and long-term follow-up with non-COVID controls to determine outcomes. The researchers noted their plans to conduct annual follow-ups in the current study population. They added that more research is needed to explore rehabilitation programs to promote recovery for COVID-19 survivors and to reduce the effects of long COVID.
The study was supported by the Chinese Academy of Medical Sciences, National Natural Science Foundation of China, National Key Research and Development Program of China, National Administration of Traditional Chinese Medicine, Major Projects of National Science and Technology on New Drug Creation and Development of Pulmonary Tuberculosis, China Evergrande Group, Jack Ma Foundation, Sino Biopharmaceutical, Ping An Insurance (Group), and New Sunshine Charity Foundation. The researchers and Dr. Pal had no financial conflicts to disclose.
This article was updated on 5/16/2022.
FROM THE LANCET RESPIRATORY MEDICINE
Myositis guidelines aim to standardize adult and pediatric care
All patients with idiopathic inflammatory myopathies (IIM) should be screened for swallowing difficulties, according to the first evidence-based guideline to be produced.
The guideline, which has been developed by a working group of the British Society for Rheumatology (BSR), also advises that all diagnosed patients should have their myositis antibody levels checked and have their overall well-being assessed. Other recommendations for all patients include the use of glucocorticoids to reduce muscle inflammation and conventional synthetic disease-modifying antirheumatic drugs (csDMARDs) for long-term treatment.
“Finally, now, we’re able to standardize the way we treat adults and children with IIM,” senior guideline author Hector Chinoy, PhD, said at the society’s annual meeting.
It has been a long labor of love, however, taking 4 years to get the guideline published, said Dr. Chinoy, professor of rheumatology and neuromuscular disease at the University of Manchester (England), and a consultant at Salford (England) Royal Hospital.
“We’re not covering diagnosis, classification, or the investigation of suspected IIM,” said Dr. Chinoy. Inclusion body myositis also is not included.
Altogether, there are 13 recommendations that have been developed using a PICO (patient or population, intervention, comparison, outcome) format, graded based on the quality of the available evidence, and then voted on by the working group members to give a score of the strength of agreement. Dr. Chinoy noted that there was a checklist included in the Supplementary Data section of the guideline to help follow the recommendations.
“The target audience for the guideline reflects the variety of clinicians caring for patients with IIM,” Dr. Chinoy said. So that is not just pediatric and adult rheumatologists, but also neurologists, dermatologists, respiratory physicians, oncologists, gastroenterologists, cardiologists, and of course other health care professionals. This includes rheumatology and neurology nurses, psychologists, speech and language therapists, and podiatrists, as well as rheumatology specialist pharmacists, physiotherapists, and occupational therapists.
With reference to the latter, Liza McCann, MBBS, who co-led the development of the guideline, said in a statement released by the BSR that the guideline “highlights the importance of exercise, led and monitored by specialist physiotherapists and occupational therapists.”
Dr. McCann, a consultant pediatric rheumatologist at Alder Hey Hospital, Liverpool, England, and Honorary Clinical Lecturer at the University of Liverpool, added that the guidelines also cover “the need to address psychological wellbeing as an integral part of treatment, in parallel with pharmacological therapies.”
Recommendation highlights
Some of the highlights of the recommendations include the use of high-dose glucocorticoids to manage skeletal muscle inflammation at the time of treatment induction, with specific guidance on the different doses to use in adults and in children. There also is guidance on the use of csDMARDs in both populations and what to use if there is refractory disease – with the strongest evidence supporting the use of intravenous immunoglobulin (IVIG) or cyclophosphamide, and possibly rituximab and abatacept.
“There is insufficient evidence to recommend JAK inhibition,” Dr. Chinoy said. The data search used to develop the guideline had a cutoff of October 2020, but even now there is only anecdotal evidence from case studies, he added.
Importantly, the guidelines recognize that childhood IIM differs from adult disease and call for children to be managed by pediatric specialists.
“Routine assessment of dysphagia should be considered in all patients,” Dr. Chinoy said, “so ask the question.” The recommendation is that a swallowing assessment should involve a speech and language therapist or gastroenterologist, and that IVIG be considered for active disease and dysphagia that is resistant to other treatments.
There also are recommendations to screen adult patients for interstitial lung disease, consider fracture risk, and screen adult patients for cancer if they have specific risk factors that include older age at onset, male gender, dysphagia, and rapid disease onset, among others.
Separate cancer screening guidelines on cards
“Around one in four patients with myositis will develop cancer within the 3 years either before or after myositis onset,” Alexander Oldroyd, MBChB, PhD, said in a separate presentation at the BSR annual meeting.
“It’s a hugely increased risk compared to the general population, and a great worry for patients,” he added. Exactly why there is an increased risk is not known, but “there’s a big link between the biological onset of cancer and myositis.”
Dr. Oldroyd, who is an NIHR Academic Clinical Lecturer at the University of Manchester in England and a coauthor of the BSR myositis guideline, is part of a special interest group set up by the International Myositis Assessment and Clinical Studies Group (IMACS) that is in the process of developing separate guidelines for cancer screening in people newly diagnosed with IIM.
The aim was to produce evidence-based recommendations that were both “pragmatic and practical,” that could help clinicians answer patient’s questions on their risk and how best and how often to screen them, Dr. Oldroyd explained. Importantly, IMACS has endeavored to create recommendations that should be applicable across different countries and health care systems.
“We had to acknowledge that there’s not a lot of evidence base there,” Dr. Oldroyd said, noting that he and colleagues conducted a systematic literature review and meta-analysis and used a Delphi process to draft 20 recommendations. These cover identifying risk factors for cancer in people with myositis and categorizing people into low, medium, and high-risk categories. The recommendations also cover what should constitute basic and enhanced screening, and how often someone should be screened.
Moreover, the authors make recommendations on the use of imaging modalities such as PET and CT scans, as well as upper and lower gastrointestinal endoscopy and naso-endoscopy.
“As rheumatologists, we don’t talk about cancer a lot,” Dr. Oldroyd said. “We pick up a lot of incidental cancers, but we don’t usually talk about cancer screening with patients.” That’s something that needs to change, he said.
“It’s important – just get it out in the open, talk to people about it,” Dr. Oldroyd said.
“Tell them what you’re wanting to do, how you’re wanting to investigate for it, clearly communicate their risk,” he said. “But also acknowledge the limited evidence as well, and clearly communicate the results.”
Dr. Chinoy acknowledged he had received fees for presentations (UCB, Biogen), consultancy (Alexion, Novartis, Eli Lilly, Orphazyme, AstraZeneca), or grant support (Eli Lilly, UCB) that had been paid via his institution for the purpose of furthering myositis research. Dr. Oldroyd had no conflicts of interest to disclose.
All patients with idiopathic inflammatory myopathies (IIM) should be screened for swallowing difficulties, according to the first evidence-based guideline to be produced.
The guideline, which has been developed by a working group of the British Society for Rheumatology (BSR), also advises that all diagnosed patients should have their myositis antibody levels checked and have their overall well-being assessed. Other recommendations for all patients include the use of glucocorticoids to reduce muscle inflammation and conventional synthetic disease-modifying antirheumatic drugs (csDMARDs) for long-term treatment.
“Finally, now, we’re able to standardize the way we treat adults and children with IIM,” senior guideline author Hector Chinoy, PhD, said at the society’s annual meeting.
It has been a long labor of love, however, taking 4 years to get the guideline published, said Dr. Chinoy, professor of rheumatology and neuromuscular disease at the University of Manchester (England), and a consultant at Salford (England) Royal Hospital.
“We’re not covering diagnosis, classification, or the investigation of suspected IIM,” said Dr. Chinoy. Inclusion body myositis also is not included.
Altogether, there are 13 recommendations that have been developed using a PICO (patient or population, intervention, comparison, outcome) format, graded based on the quality of the available evidence, and then voted on by the working group members to give a score of the strength of agreement. Dr. Chinoy noted that there was a checklist included in the Supplementary Data section of the guideline to help follow the recommendations.
“The target audience for the guideline reflects the variety of clinicians caring for patients with IIM,” Dr. Chinoy said. So that is not just pediatric and adult rheumatologists, but also neurologists, dermatologists, respiratory physicians, oncologists, gastroenterologists, cardiologists, and of course other health care professionals. This includes rheumatology and neurology nurses, psychologists, speech and language therapists, and podiatrists, as well as rheumatology specialist pharmacists, physiotherapists, and occupational therapists.
With reference to the latter, Liza McCann, MBBS, who co-led the development of the guideline, said in a statement released by the BSR that the guideline “highlights the importance of exercise, led and monitored by specialist physiotherapists and occupational therapists.”
Dr. McCann, a consultant pediatric rheumatologist at Alder Hey Hospital, Liverpool, England, and Honorary Clinical Lecturer at the University of Liverpool, added that the guidelines also cover “the need to address psychological wellbeing as an integral part of treatment, in parallel with pharmacological therapies.”
Recommendation highlights
Some of the highlights of the recommendations include the use of high-dose glucocorticoids to manage skeletal muscle inflammation at the time of treatment induction, with specific guidance on the different doses to use in adults and in children. There also is guidance on the use of csDMARDs in both populations and what to use if there is refractory disease – with the strongest evidence supporting the use of intravenous immunoglobulin (IVIG) or cyclophosphamide, and possibly rituximab and abatacept.
“There is insufficient evidence to recommend JAK inhibition,” Dr. Chinoy said. The data search used to develop the guideline had a cutoff of October 2020, but even now there is only anecdotal evidence from case studies, he added.
Importantly, the guidelines recognize that childhood IIM differs from adult disease and call for children to be managed by pediatric specialists.
“Routine assessment of dysphagia should be considered in all patients,” Dr. Chinoy said, “so ask the question.” The recommendation is that a swallowing assessment should involve a speech and language therapist or gastroenterologist, and that IVIG be considered for active disease and dysphagia that is resistant to other treatments.
There also are recommendations to screen adult patients for interstitial lung disease, consider fracture risk, and screen adult patients for cancer if they have specific risk factors that include older age at onset, male gender, dysphagia, and rapid disease onset, among others.
Separate cancer screening guidelines on cards
“Around one in four patients with myositis will develop cancer within the 3 years either before or after myositis onset,” Alexander Oldroyd, MBChB, PhD, said in a separate presentation at the BSR annual meeting.
“It’s a hugely increased risk compared to the general population, and a great worry for patients,” he added. Exactly why there is an increased risk is not known, but “there’s a big link between the biological onset of cancer and myositis.”
Dr. Oldroyd, who is an NIHR Academic Clinical Lecturer at the University of Manchester in England and a coauthor of the BSR myositis guideline, is part of a special interest group set up by the International Myositis Assessment and Clinical Studies Group (IMACS) that is in the process of developing separate guidelines for cancer screening in people newly diagnosed with IIM.
The aim was to produce evidence-based recommendations that were both “pragmatic and practical,” that could help clinicians answer patient’s questions on their risk and how best and how often to screen them, Dr. Oldroyd explained. Importantly, IMACS has endeavored to create recommendations that should be applicable across different countries and health care systems.
“We had to acknowledge that there’s not a lot of evidence base there,” Dr. Oldroyd said, noting that he and colleagues conducted a systematic literature review and meta-analysis and used a Delphi process to draft 20 recommendations. These cover identifying risk factors for cancer in people with myositis and categorizing people into low, medium, and high-risk categories. The recommendations also cover what should constitute basic and enhanced screening, and how often someone should be screened.
Moreover, the authors make recommendations on the use of imaging modalities such as PET and CT scans, as well as upper and lower gastrointestinal endoscopy and naso-endoscopy.
“As rheumatologists, we don’t talk about cancer a lot,” Dr. Oldroyd said. “We pick up a lot of incidental cancers, but we don’t usually talk about cancer screening with patients.” That’s something that needs to change, he said.
“It’s important – just get it out in the open, talk to people about it,” Dr. Oldroyd said.
“Tell them what you’re wanting to do, how you’re wanting to investigate for it, clearly communicate their risk,” he said. “But also acknowledge the limited evidence as well, and clearly communicate the results.”
Dr. Chinoy acknowledged he had received fees for presentations (UCB, Biogen), consultancy (Alexion, Novartis, Eli Lilly, Orphazyme, AstraZeneca), or grant support (Eli Lilly, UCB) that had been paid via his institution for the purpose of furthering myositis research. Dr. Oldroyd had no conflicts of interest to disclose.
All patients with idiopathic inflammatory myopathies (IIM) should be screened for swallowing difficulties, according to the first evidence-based guideline to be produced.
The guideline, which has been developed by a working group of the British Society for Rheumatology (BSR), also advises that all diagnosed patients should have their myositis antibody levels checked and have their overall well-being assessed. Other recommendations for all patients include the use of glucocorticoids to reduce muscle inflammation and conventional synthetic disease-modifying antirheumatic drugs (csDMARDs) for long-term treatment.
“Finally, now, we’re able to standardize the way we treat adults and children with IIM,” senior guideline author Hector Chinoy, PhD, said at the society’s annual meeting.
It has been a long labor of love, however, taking 4 years to get the guideline published, said Dr. Chinoy, professor of rheumatology and neuromuscular disease at the University of Manchester (England), and a consultant at Salford (England) Royal Hospital.
“We’re not covering diagnosis, classification, or the investigation of suspected IIM,” said Dr. Chinoy. Inclusion body myositis also is not included.
Altogether, there are 13 recommendations that have been developed using a PICO (patient or population, intervention, comparison, outcome) format, graded based on the quality of the available evidence, and then voted on by the working group members to give a score of the strength of agreement. Dr. Chinoy noted that there was a checklist included in the Supplementary Data section of the guideline to help follow the recommendations.
“The target audience for the guideline reflects the variety of clinicians caring for patients with IIM,” Dr. Chinoy said. So that is not just pediatric and adult rheumatologists, but also neurologists, dermatologists, respiratory physicians, oncologists, gastroenterologists, cardiologists, and of course other health care professionals. This includes rheumatology and neurology nurses, psychologists, speech and language therapists, and podiatrists, as well as rheumatology specialist pharmacists, physiotherapists, and occupational therapists.
With reference to the latter, Liza McCann, MBBS, who co-led the development of the guideline, said in a statement released by the BSR that the guideline “highlights the importance of exercise, led and monitored by specialist physiotherapists and occupational therapists.”
Dr. McCann, a consultant pediatric rheumatologist at Alder Hey Hospital, Liverpool, England, and Honorary Clinical Lecturer at the University of Liverpool, added that the guidelines also cover “the need to address psychological wellbeing as an integral part of treatment, in parallel with pharmacological therapies.”
Recommendation highlights
Some of the highlights of the recommendations include the use of high-dose glucocorticoids to manage skeletal muscle inflammation at the time of treatment induction, with specific guidance on the different doses to use in adults and in children. There also is guidance on the use of csDMARDs in both populations and what to use if there is refractory disease – with the strongest evidence supporting the use of intravenous immunoglobulin (IVIG) or cyclophosphamide, and possibly rituximab and abatacept.
“There is insufficient evidence to recommend JAK inhibition,” Dr. Chinoy said. The data search used to develop the guideline had a cutoff of October 2020, but even now there is only anecdotal evidence from case studies, he added.
Importantly, the guidelines recognize that childhood IIM differs from adult disease and call for children to be managed by pediatric specialists.
“Routine assessment of dysphagia should be considered in all patients,” Dr. Chinoy said, “so ask the question.” The recommendation is that a swallowing assessment should involve a speech and language therapist or gastroenterologist, and that IVIG be considered for active disease and dysphagia that is resistant to other treatments.
There also are recommendations to screen adult patients for interstitial lung disease, consider fracture risk, and screen adult patients for cancer if they have specific risk factors that include older age at onset, male gender, dysphagia, and rapid disease onset, among others.
Separate cancer screening guidelines on cards
“Around one in four patients with myositis will develop cancer within the 3 years either before or after myositis onset,” Alexander Oldroyd, MBChB, PhD, said in a separate presentation at the BSR annual meeting.
“It’s a hugely increased risk compared to the general population, and a great worry for patients,” he added. Exactly why there is an increased risk is not known, but “there’s a big link between the biological onset of cancer and myositis.”
Dr. Oldroyd, who is an NIHR Academic Clinical Lecturer at the University of Manchester in England and a coauthor of the BSR myositis guideline, is part of a special interest group set up by the International Myositis Assessment and Clinical Studies Group (IMACS) that is in the process of developing separate guidelines for cancer screening in people newly diagnosed with IIM.
The aim was to produce evidence-based recommendations that were both “pragmatic and practical,” that could help clinicians answer patient’s questions on their risk and how best and how often to screen them, Dr. Oldroyd explained. Importantly, IMACS has endeavored to create recommendations that should be applicable across different countries and health care systems.
“We had to acknowledge that there’s not a lot of evidence base there,” Dr. Oldroyd said, noting that he and colleagues conducted a systematic literature review and meta-analysis and used a Delphi process to draft 20 recommendations. These cover identifying risk factors for cancer in people with myositis and categorizing people into low, medium, and high-risk categories. The recommendations also cover what should constitute basic and enhanced screening, and how often someone should be screened.
Moreover, the authors make recommendations on the use of imaging modalities such as PET and CT scans, as well as upper and lower gastrointestinal endoscopy and naso-endoscopy.
“As rheumatologists, we don’t talk about cancer a lot,” Dr. Oldroyd said. “We pick up a lot of incidental cancers, but we don’t usually talk about cancer screening with patients.” That’s something that needs to change, he said.
“It’s important – just get it out in the open, talk to people about it,” Dr. Oldroyd said.
“Tell them what you’re wanting to do, how you’re wanting to investigate for it, clearly communicate their risk,” he said. “But also acknowledge the limited evidence as well, and clearly communicate the results.”
Dr. Chinoy acknowledged he had received fees for presentations (UCB, Biogen), consultancy (Alexion, Novartis, Eli Lilly, Orphazyme, AstraZeneca), or grant support (Eli Lilly, UCB) that had been paid via his institution for the purpose of furthering myositis research. Dr. Oldroyd had no conflicts of interest to disclose.
FROM BSR 2022
Exenatide linked to less hyperglycemia after stroke
Treatment with the diabetes drug exenatide was associated with a significant decrease in hyperglycemia in acute stroke patients, a new study shows.
The research could offer clinicians an alternative to insulin therapy to treat hyperglycemia and reduce glucose levels, which are elevated in up to 60% of stroke patients and associated with worse outcomes after stroke.
“Use of these diabetes drugs to control glucose in acute stroke has enormous potential,” said lead researcher Christopher Bladin, PhD, professor of neurology at Monash University and Eastern Health Clinical School, Australia.
The findings were presented at the European Stroke Organisation Conference (ESOC) 2022 annual meeting in Lyon, France.
A better fix than insulin?
Hyperglycemia is common in stroke patients, including those who have no prior history of diabetes. Among stroke patients with normal blood glucose upon admission, about 30% will develop hyperglycemia within 48 hours of stroke onset.
Previous research suggests that hyperglycemia is a poor prognostic factor in patients with stroke and may reduce the efficacy of reperfusion therapies such as thrombolysis and mechanical thrombectomy.
“We’ve been looking for different ways of treating hyperglycemia for quite some time, and one of the obvious ways is to use insulin therapy,” Dr. Bladin said. “But as we’ve seen from multiple studies, insulin therapy is difficult.”
Insulin treatment is resource-heavy, significantly increases the risk for hypoglycemia, and some studies suggest the therapy isn’t associated with better outcomes.
An advantage to a GLP-1 agonist-like exenatide, Dr. Bladin added, is that it’s glucose-dependent. As the glucose level falls, the drug’s efficacy diminishes. It is delivered via an autoinjector and easy to administer.
A case for more study
To study exenatide’s efficacy in reducing hyperglycemia and improving neurologic outcomes, researchers developed the phase 2, international, multicenter, randomized controlled TEXAIS trial.
The study enrolled 350 patients following an ischemic stroke. Within 9 hours of stroke onset, patients received either standard care or a subcutaneous injection of 5 mg of exenatide twice daily for 5 days.
On admission, 42% of patients had hyperglycemia, defined as blood glucose > 7.0 mmol/L.
The study’s primary outcome was at least an 8-point improvement in National Institutes of Health Stroke Scale (NIHSS) score by 7 days after treatment with exenatide. Although there was a trend toward better scores with exenatide, the score was not significantly different between groups (56.7% with standard care versus 61.2% with exenatide; adjusted odds ratio, 1.22; P = .38).
However, when the researchers examined hyperglycemia frequency, they found significantly lower incidence in patients treated with exenatide (P = .002).
There were no cases of hypoglycemia in either group, and only 4% of the study group reported nausea or vomiting.
“Clearly exenatide is having some benefit in terms of keeping glucose under control, reducing hyperglycemia,” Dr. Bladin said. “It certainly lends itself to a larger phase 3 study which can look at this more completely.”
Value to clinicians
Commenting on the findings, Yvonne Chun, PhD, honorary senior clinical lecturer at University of Edinburgh, noted that, even though the study didn’t find a significant association with improved neurological outcomes, the reduced risk for hypoglycemia makes exenatide an attractive alternative to insulin therapy in stroke patients.
“The results are of value to clinicians, as exenatide could potentially be a safer medication to administer than an insulin infusion in acute stroke patients with hyperglycemia,” Dr. Chun said. “There is less risk of hypoglycemia with exenatide compared to standard care.”
However, Dr. Chun noted that more study is needed before exenatide can replace standard care. Dr. Bladin agrees and would like to pursue a phase 3 trial with a modified design to answer questions raised by Dr. Chun and others.
“The next phase could consider changing the primary outcome to an ordinal shift analysis on modified Rankin Scale – a very commonly used primary outcome in stroke clinical trials to assess improvement in disability,” Dr. Chun said. “The primary outcome used in the presented trial – an 8-point improvement on NIHSS – seemed too ambitious and does not inform disability of the patient post stroke.”
Dr. Bladin said he would also like to see the next phase enroll more patients, examine a higher dose of exenatide, and include better stratification of patients with a history of diabetes. Such a trial could yield findings demonstrating the drug’s effectiveness at reducing hyperglycemia and improving outcomes after stroke, he said.
“I can see the day patients will come in with acute stroke, and as they’re coming into the emergency department, they’ll simply get their shot of exenatide because we know it’s safe to use, and it doesn’t cause hypoglycemia,” Dr. Bladin said. “And from the moment that patient arrives the glucose control is underway.”
Dr. Bladin and Dr. Chun reported no relevant financial relationships. Study funding was not disclosed.
A version of this article first appeared on Medscape.com.
Treatment with the diabetes drug exenatide was associated with a significant decrease in hyperglycemia in acute stroke patients, a new study shows.
The research could offer clinicians an alternative to insulin therapy to treat hyperglycemia and reduce glucose levels, which are elevated in up to 60% of stroke patients and associated with worse outcomes after stroke.
“Use of these diabetes drugs to control glucose in acute stroke has enormous potential,” said lead researcher Christopher Bladin, PhD, professor of neurology at Monash University and Eastern Health Clinical School, Australia.
The findings were presented at the European Stroke Organisation Conference (ESOC) 2022 annual meeting in Lyon, France.
A better fix than insulin?
Hyperglycemia is common in stroke patients, including those who have no prior history of diabetes. Among stroke patients with normal blood glucose upon admission, about 30% will develop hyperglycemia within 48 hours of stroke onset.
Previous research suggests that hyperglycemia is a poor prognostic factor in patients with stroke and may reduce the efficacy of reperfusion therapies such as thrombolysis and mechanical thrombectomy.
“We’ve been looking for different ways of treating hyperglycemia for quite some time, and one of the obvious ways is to use insulin therapy,” Dr. Bladin said. “But as we’ve seen from multiple studies, insulin therapy is difficult.”
Insulin treatment is resource-heavy, significantly increases the risk for hypoglycemia, and some studies suggest the therapy isn’t associated with better outcomes.
An advantage to a GLP-1 agonist-like exenatide, Dr. Bladin added, is that it’s glucose-dependent. As the glucose level falls, the drug’s efficacy diminishes. It is delivered via an autoinjector and easy to administer.
A case for more study
To study exenatide’s efficacy in reducing hyperglycemia and improving neurologic outcomes, researchers developed the phase 2, international, multicenter, randomized controlled TEXAIS trial.
The study enrolled 350 patients following an ischemic stroke. Within 9 hours of stroke onset, patients received either standard care or a subcutaneous injection of 5 mg of exenatide twice daily for 5 days.
On admission, 42% of patients had hyperglycemia, defined as blood glucose > 7.0 mmol/L.
The study’s primary outcome was at least an 8-point improvement in National Institutes of Health Stroke Scale (NIHSS) score by 7 days after treatment with exenatide. Although there was a trend toward better scores with exenatide, the score was not significantly different between groups (56.7% with standard care versus 61.2% with exenatide; adjusted odds ratio, 1.22; P = .38).
However, when the researchers examined hyperglycemia frequency, they found significantly lower incidence in patients treated with exenatide (P = .002).
There were no cases of hypoglycemia in either group, and only 4% of the study group reported nausea or vomiting.
“Clearly exenatide is having some benefit in terms of keeping glucose under control, reducing hyperglycemia,” Dr. Bladin said. “It certainly lends itself to a larger phase 3 study which can look at this more completely.”
Value to clinicians
Commenting on the findings, Yvonne Chun, PhD, honorary senior clinical lecturer at University of Edinburgh, noted that, even though the study didn’t find a significant association with improved neurological outcomes, the reduced risk for hypoglycemia makes exenatide an attractive alternative to insulin therapy in stroke patients.
“The results are of value to clinicians, as exenatide could potentially be a safer medication to administer than an insulin infusion in acute stroke patients with hyperglycemia,” Dr. Chun said. “There is less risk of hypoglycemia with exenatide compared to standard care.”
However, Dr. Chun noted that more study is needed before exenatide can replace standard care. Dr. Bladin agrees and would like to pursue a phase 3 trial with a modified design to answer questions raised by Dr. Chun and others.
“The next phase could consider changing the primary outcome to an ordinal shift analysis on modified Rankin Scale – a very commonly used primary outcome in stroke clinical trials to assess improvement in disability,” Dr. Chun said. “The primary outcome used in the presented trial – an 8-point improvement on NIHSS – seemed too ambitious and does not inform disability of the patient post stroke.”
Dr. Bladin said he would also like to see the next phase enroll more patients, examine a higher dose of exenatide, and include better stratification of patients with a history of diabetes. Such a trial could yield findings demonstrating the drug’s effectiveness at reducing hyperglycemia and improving outcomes after stroke, he said.
“I can see the day patients will come in with acute stroke, and as they’re coming into the emergency department, they’ll simply get their shot of exenatide because we know it’s safe to use, and it doesn’t cause hypoglycemia,” Dr. Bladin said. “And from the moment that patient arrives the glucose control is underway.”
Dr. Bladin and Dr. Chun reported no relevant financial relationships. Study funding was not disclosed.
A version of this article first appeared on Medscape.com.
Treatment with the diabetes drug exenatide was associated with a significant decrease in hyperglycemia in acute stroke patients, a new study shows.
The research could offer clinicians an alternative to insulin therapy to treat hyperglycemia and reduce glucose levels, which are elevated in up to 60% of stroke patients and associated with worse outcomes after stroke.
“Use of these diabetes drugs to control glucose in acute stroke has enormous potential,” said lead researcher Christopher Bladin, PhD, professor of neurology at Monash University and Eastern Health Clinical School, Australia.
The findings were presented at the European Stroke Organisation Conference (ESOC) 2022 annual meeting in Lyon, France.
A better fix than insulin?
Hyperglycemia is common in stroke patients, including those who have no prior history of diabetes. Among stroke patients with normal blood glucose upon admission, about 30% will develop hyperglycemia within 48 hours of stroke onset.
Previous research suggests that hyperglycemia is a poor prognostic factor in patients with stroke and may reduce the efficacy of reperfusion therapies such as thrombolysis and mechanical thrombectomy.
“We’ve been looking for different ways of treating hyperglycemia for quite some time, and one of the obvious ways is to use insulin therapy,” Dr. Bladin said. “But as we’ve seen from multiple studies, insulin therapy is difficult.”
Insulin treatment is resource-heavy, significantly increases the risk for hypoglycemia, and some studies suggest the therapy isn’t associated with better outcomes.
An advantage to a GLP-1 agonist-like exenatide, Dr. Bladin added, is that it’s glucose-dependent. As the glucose level falls, the drug’s efficacy diminishes. It is delivered via an autoinjector and easy to administer.
A case for more study
To study exenatide’s efficacy in reducing hyperglycemia and improving neurologic outcomes, researchers developed the phase 2, international, multicenter, randomized controlled TEXAIS trial.
The study enrolled 350 patients following an ischemic stroke. Within 9 hours of stroke onset, patients received either standard care or a subcutaneous injection of 5 mg of exenatide twice daily for 5 days.
On admission, 42% of patients had hyperglycemia, defined as blood glucose > 7.0 mmol/L.
The study’s primary outcome was at least an 8-point improvement in National Institutes of Health Stroke Scale (NIHSS) score by 7 days after treatment with exenatide. Although there was a trend toward better scores with exenatide, the score was not significantly different between groups (56.7% with standard care versus 61.2% with exenatide; adjusted odds ratio, 1.22; P = .38).
However, when the researchers examined hyperglycemia frequency, they found significantly lower incidence in patients treated with exenatide (P = .002).
There were no cases of hypoglycemia in either group, and only 4% of the study group reported nausea or vomiting.
“Clearly exenatide is having some benefit in terms of keeping glucose under control, reducing hyperglycemia,” Dr. Bladin said. “It certainly lends itself to a larger phase 3 study which can look at this more completely.”
Value to clinicians
Commenting on the findings, Yvonne Chun, PhD, honorary senior clinical lecturer at University of Edinburgh, noted that, even though the study didn’t find a significant association with improved neurological outcomes, the reduced risk for hypoglycemia makes exenatide an attractive alternative to insulin therapy in stroke patients.
“The results are of value to clinicians, as exenatide could potentially be a safer medication to administer than an insulin infusion in acute stroke patients with hyperglycemia,” Dr. Chun said. “There is less risk of hypoglycemia with exenatide compared to standard care.”
However, Dr. Chun noted that more study is needed before exenatide can replace standard care. Dr. Bladin agrees and would like to pursue a phase 3 trial with a modified design to answer questions raised by Dr. Chun and others.
“The next phase could consider changing the primary outcome to an ordinal shift analysis on modified Rankin Scale – a very commonly used primary outcome in stroke clinical trials to assess improvement in disability,” Dr. Chun said. “The primary outcome used in the presented trial – an 8-point improvement on NIHSS – seemed too ambitious and does not inform disability of the patient post stroke.”
Dr. Bladin said he would also like to see the next phase enroll more patients, examine a higher dose of exenatide, and include better stratification of patients with a history of diabetes. Such a trial could yield findings demonstrating the drug’s effectiveness at reducing hyperglycemia and improving outcomes after stroke, he said.
“I can see the day patients will come in with acute stroke, and as they’re coming into the emergency department, they’ll simply get their shot of exenatide because we know it’s safe to use, and it doesn’t cause hypoglycemia,” Dr. Bladin said. “And from the moment that patient arrives the glucose control is underway.”
Dr. Bladin and Dr. Chun reported no relevant financial relationships. Study funding was not disclosed.
A version of this article first appeared on Medscape.com.
FROM ESOC 2022