User login
Study tests ways to increase autism screening and referrals
To improve autism screening rates, researchers in Utah tried a range of interventions.
They added automatic reminders to the electronic health record (EHR). They started using a shorter, more sensitive screening instrument. And they trained clinicians to perform autism-specific evaluations in a primary care clinic.
The researchers found that these interventions were associated with increased rates of autism screening and referrals.
At the same time, they looked at screening and referral rates at other community clinics in their health care system. These clinics incorporated EHR reminders but not all of the other changes.
“The community clinics had an increase in screening frequency with only automatic reminders,” the researchers reported. At the two intervention clinics, however, screening rates increased more than they did at the community clinics. Referrals did not significantly increase at the community clinics.
Kathleen Campbell, MD, MHSc, a pediatric resident at the University of Utah in Salt Lake City, and colleagues described their research in a study published in Pediatrics.
Three phases
They examined more than 12,000 well-child visits for children aged 16-30 months between July 2017 and June 2019.
In all, 4,155 visits occurred at the 2 intervention clinics, and 8,078 visits occurred at the 27 community clinics in the University of Utah health care system.
From baseline through the interventions, the proportion of visits with screening increased by 51% in the intervention clinics (from 58.6% to 88.8%), and by 21% in the community clinics (from 43.4% to 52.4%). The proportion of referrals increased 1.5-fold in intervention clinics, from 1.3% to 3.3%, the authors said.
The American Academy of Pediatrics (AAP) supports screening for autism in all children starting at age 18 months, but “only 44% of children with autism have had a comprehensive autism evaluation before age 36 months,” Dr. Campbell and colleagues wrote.
In their system, about half of the children were being screened for autism, and 0.5% had autism diagnosed.
In an effort to increase the proportion of visits with screening for autism and the proportion of visits with referrals for autism evaluation, Dr. Campbell and colleagues designed a quality improvement study.
Following a baseline period, they implemented interventions in three phases.
Initially, all clinics used the Modified Checklist for Autism in Toddlers, Revised (M-CHAT-R) for autism screening. For the first phase starting in July 2018, the researchers changed the screening instrument at the two intervention clinics to the Parent’s Observation of Social Interaction (POSI). This instrument “is embedded in a broadband developmental screen, is shorter than the M-CHAT-R, and includes questions about the consistency of the child’s behavior,” the authors said. “The POSI has greater sensitivity than the M-CHAT-R ... and similar, although somewhat lower, specificity.”
In intervention phase 2 starting in November 2018, the researchers “added an automatic reminder for autism screening to the EHR health maintenance screen.” Both the intervention clinics and the community clinics received the automatic reminders.
In intervention phase 3 starting in February 2019, they “added a referral option that clinicians could use for rapid access to autism-specific evaluation ... for children who had a POSI result suggestive of autism and for whom the clinician had sufficient concerns about autism that would indicate the need for referral for autism evaluation,” the researchers said.
“Using an online tutorial, we trained three clinicians in the intervention clinics to administer an observational assessment known as the Screening Tool for Autism in Toddlers (STAT),” which requires a 30-minute visit, they said. “Children who had a STAT result suggestive of autism were referred for expedited autism diagnostic evaluation, which was performed by a multidisciplinary team in our university-based developmental assessment clinic. Children who had a STAT result that did not suggest autism did not receive further autism evaluations unless the clinician felt they still needed further evaluation at the developmental clinic.”
After the switch to POSI, the percentage of visits with a positive screen result increased from 4.7% to 13.5% in the intervention clinics.
Furthermore, referrals were 3.4 times more frequent for visits during phase 3 in the intervention clinics, relative to the baseline period.
Potential to overwhelm
“The change to a more sensitive screening instrument increased the frequency of screening results suggestive of autism and informed our improvement team of the need to implement autism evaluation in primary care to avoid overwhelming our referral system,” Dr. Campbell and coauthors reported.
Future studies may assess whether increased screening and referrals speed the time to diagnosis and treatment and improve long-term functional abilities of children with autism. Some children in the study have received an autism diagnosis, while others have not yet been evaluated.
The use of STAT in primary care may be limited by “the barriers of training providers and purchasing materials,” the authors noted. “However, the time-based billing for lengthier appointments and billing for developmental testing help to cover cost.”
The intervention clinics and community clinics were staffed by pediatric providers, including residents and attendings, said Dr. Campbell.
“The staffing is similar at the community and intervention clinics, with mostly pediatricians and some nurse practitioners,” Dr. Campbell said. “One difference is that there are a few family medicine physicians in the community clinics, but we did not study whether that made a difference in screening. At the beginning of the study the approach to screening was the same.”
From the start, the community clinics were screening for autism and referring for further autism evaluation less often than the intervention clinics. “I don’t know why they were screening less, but they did improve with the automatic reminders,” said Dr. Campbell. “We didn’t examine type of provider or type of practice in this study, but the literature suggests that family physicians do not screen for autism as often as pediatricians.”
Payment and referral challenges
In theory, the approach in the study is a great idea, but it may not be feasible to implement for many private practices, said Herschel Lessin, MD. Dr. Lessin is a senior partner of the Children’s Medical Group in New York.
“We desperately need autism screening in a primary care setting,” Dr. Lessin said. “These authors found that wasn’t being done as recommended by the AAP Bright Futures, which is a problem.”
However, the researchers incorporated the interventions in a health care system with “far more resources than most people in practice would ever have” and substituted a less familiar screening tool.
In addition, the ability to use confirmatory STAT for primary care evaluations may be limited. “Unless you can find pediatricians willing to commit 30 to 45 minutes on one of these evaluations ... few are going to do that,” he said.
“The whole problem is that there are no referrals available or very few referrals available, and that insurance payments so underpay for developmental screening and evaluation that it does not justify the time doing it, so a lot of doctors are unable to do it,” said Dr. Lessin. When a referral is warranted, developmental pediatricians may have 6- to 12-month waiting lists, he said.
“For people in clinical practice, this is not news,” Dr. Lessin said. “We know we should screen for autism. The problem is it’s time consuming. Nobody pays for it. We have no place to send them even when we are suspicious.”
From screening to diagnosis to treatment
“Autism screen approaches vary but with educational efforts on the part of the AAP, CDC, and family organizations the rates for autism screening have dramatically improved,” said Susan L. Hyman, MD, professor of pediatrics at the University of Rochester in New York. “I do not know if screening rates have been impacted by COVID.”
Dr. Hyman and coauthors wrote an AAP clinical report on the identification, evaluation, and management of children with autism spectrum disorder. The report was published in the January 2020 issue of Pediatrics.
After screening and diagnostic testing, patients most importantly need to be able to access “timely and equitable evidence-based intervention,” which should be available, said Dr. Hyman.
Although researchers have proposed training primary care providers in autism diagnostics, “older, more complex patients with co-occurring behavioral health or other developmental disorders may need more specialized diagnostic assessment than could be accomplished in a primary care setting,” Dr. Hyman added.
“However, it is very important to identify children with therapeutic needs as early as possible and move them through the continuum from screening to diagnosis to treatment in a timely fashion. It would be wonderful if symptoms could be addressed without the need for diagnosis in the very youngest children,” Dr. Hyman said. “Early symptoms, even if not autism, are likely to be appropriate for intervention – whether it is speech therapy, attention to food selectivity, sleep problems – things that impact quality of life and potential future symptoms.”
The research was supported by the Utah Stimulating Access to Research in Residency Transition Scholar award, which is funded by the National Institutes of Health.
Dr. Campbell is an inventor on a patent related to screening for autism. The study authors otherwise had no disclosures. Dr. Lessin is on the editorial advisory board for Pediatric News and is on an advisory board for Cognoa, which is developing a medical device to diagnose autism and he is also the co-editor of the AAP's current ADHD Toolkit. Dr. Hyman had no relevant financial disclosures.
*This story was updated on Feb. 11, 2021.
To improve autism screening rates, researchers in Utah tried a range of interventions.
They added automatic reminders to the electronic health record (EHR). They started using a shorter, more sensitive screening instrument. And they trained clinicians to perform autism-specific evaluations in a primary care clinic.
The researchers found that these interventions were associated with increased rates of autism screening and referrals.
At the same time, they looked at screening and referral rates at other community clinics in their health care system. These clinics incorporated EHR reminders but not all of the other changes.
“The community clinics had an increase in screening frequency with only automatic reminders,” the researchers reported. At the two intervention clinics, however, screening rates increased more than they did at the community clinics. Referrals did not significantly increase at the community clinics.
Kathleen Campbell, MD, MHSc, a pediatric resident at the University of Utah in Salt Lake City, and colleagues described their research in a study published in Pediatrics.
Three phases
They examined more than 12,000 well-child visits for children aged 16-30 months between July 2017 and June 2019.
In all, 4,155 visits occurred at the 2 intervention clinics, and 8,078 visits occurred at the 27 community clinics in the University of Utah health care system.
From baseline through the interventions, the proportion of visits with screening increased by 51% in the intervention clinics (from 58.6% to 88.8%), and by 21% in the community clinics (from 43.4% to 52.4%). The proportion of referrals increased 1.5-fold in intervention clinics, from 1.3% to 3.3%, the authors said.
The American Academy of Pediatrics (AAP) supports screening for autism in all children starting at age 18 months, but “only 44% of children with autism have had a comprehensive autism evaluation before age 36 months,” Dr. Campbell and colleagues wrote.
In their system, about half of the children were being screened for autism, and 0.5% had autism diagnosed.
In an effort to increase the proportion of visits with screening for autism and the proportion of visits with referrals for autism evaluation, Dr. Campbell and colleagues designed a quality improvement study.
Following a baseline period, they implemented interventions in three phases.
Initially, all clinics used the Modified Checklist for Autism in Toddlers, Revised (M-CHAT-R) for autism screening. For the first phase starting in July 2018, the researchers changed the screening instrument at the two intervention clinics to the Parent’s Observation of Social Interaction (POSI). This instrument “is embedded in a broadband developmental screen, is shorter than the M-CHAT-R, and includes questions about the consistency of the child’s behavior,” the authors said. “The POSI has greater sensitivity than the M-CHAT-R ... and similar, although somewhat lower, specificity.”
In intervention phase 2 starting in November 2018, the researchers “added an automatic reminder for autism screening to the EHR health maintenance screen.” Both the intervention clinics and the community clinics received the automatic reminders.
In intervention phase 3 starting in February 2019, they “added a referral option that clinicians could use for rapid access to autism-specific evaluation ... for children who had a POSI result suggestive of autism and for whom the clinician had sufficient concerns about autism that would indicate the need for referral for autism evaluation,” the researchers said.
“Using an online tutorial, we trained three clinicians in the intervention clinics to administer an observational assessment known as the Screening Tool for Autism in Toddlers (STAT),” which requires a 30-minute visit, they said. “Children who had a STAT result suggestive of autism were referred for expedited autism diagnostic evaluation, which was performed by a multidisciplinary team in our university-based developmental assessment clinic. Children who had a STAT result that did not suggest autism did not receive further autism evaluations unless the clinician felt they still needed further evaluation at the developmental clinic.”
After the switch to POSI, the percentage of visits with a positive screen result increased from 4.7% to 13.5% in the intervention clinics.
Furthermore, referrals were 3.4 times more frequent for visits during phase 3 in the intervention clinics, relative to the baseline period.
Potential to overwhelm
“The change to a more sensitive screening instrument increased the frequency of screening results suggestive of autism and informed our improvement team of the need to implement autism evaluation in primary care to avoid overwhelming our referral system,” Dr. Campbell and coauthors reported.
Future studies may assess whether increased screening and referrals speed the time to diagnosis and treatment and improve long-term functional abilities of children with autism. Some children in the study have received an autism diagnosis, while others have not yet been evaluated.
The use of STAT in primary care may be limited by “the barriers of training providers and purchasing materials,” the authors noted. “However, the time-based billing for lengthier appointments and billing for developmental testing help to cover cost.”
The intervention clinics and community clinics were staffed by pediatric providers, including residents and attendings, said Dr. Campbell.
“The staffing is similar at the community and intervention clinics, with mostly pediatricians and some nurse practitioners,” Dr. Campbell said. “One difference is that there are a few family medicine physicians in the community clinics, but we did not study whether that made a difference in screening. At the beginning of the study the approach to screening was the same.”
From the start, the community clinics were screening for autism and referring for further autism evaluation less often than the intervention clinics. “I don’t know why they were screening less, but they did improve with the automatic reminders,” said Dr. Campbell. “We didn’t examine type of provider or type of practice in this study, but the literature suggests that family physicians do not screen for autism as often as pediatricians.”
Payment and referral challenges
In theory, the approach in the study is a great idea, but it may not be feasible to implement for many private practices, said Herschel Lessin, MD. Dr. Lessin is a senior partner of the Children’s Medical Group in New York.
“We desperately need autism screening in a primary care setting,” Dr. Lessin said. “These authors found that wasn’t being done as recommended by the AAP Bright Futures, which is a problem.”
However, the researchers incorporated the interventions in a health care system with “far more resources than most people in practice would ever have” and substituted a less familiar screening tool.
In addition, the ability to use confirmatory STAT for primary care evaluations may be limited. “Unless you can find pediatricians willing to commit 30 to 45 minutes on one of these evaluations ... few are going to do that,” he said.
“The whole problem is that there are no referrals available or very few referrals available, and that insurance payments so underpay for developmental screening and evaluation that it does not justify the time doing it, so a lot of doctors are unable to do it,” said Dr. Lessin. When a referral is warranted, developmental pediatricians may have 6- to 12-month waiting lists, he said.
“For people in clinical practice, this is not news,” Dr. Lessin said. “We know we should screen for autism. The problem is it’s time consuming. Nobody pays for it. We have no place to send them even when we are suspicious.”
From screening to diagnosis to treatment
“Autism screen approaches vary but with educational efforts on the part of the AAP, CDC, and family organizations the rates for autism screening have dramatically improved,” said Susan L. Hyman, MD, professor of pediatrics at the University of Rochester in New York. “I do not know if screening rates have been impacted by COVID.”
Dr. Hyman and coauthors wrote an AAP clinical report on the identification, evaluation, and management of children with autism spectrum disorder. The report was published in the January 2020 issue of Pediatrics.
After screening and diagnostic testing, patients most importantly need to be able to access “timely and equitable evidence-based intervention,” which should be available, said Dr. Hyman.
Although researchers have proposed training primary care providers in autism diagnostics, “older, more complex patients with co-occurring behavioral health or other developmental disorders may need more specialized diagnostic assessment than could be accomplished in a primary care setting,” Dr. Hyman added.
“However, it is very important to identify children with therapeutic needs as early as possible and move them through the continuum from screening to diagnosis to treatment in a timely fashion. It would be wonderful if symptoms could be addressed without the need for diagnosis in the very youngest children,” Dr. Hyman said. “Early symptoms, even if not autism, are likely to be appropriate for intervention – whether it is speech therapy, attention to food selectivity, sleep problems – things that impact quality of life and potential future symptoms.”
The research was supported by the Utah Stimulating Access to Research in Residency Transition Scholar award, which is funded by the National Institutes of Health.
Dr. Campbell is an inventor on a patent related to screening for autism. The study authors otherwise had no disclosures. Dr. Lessin is on the editorial advisory board for Pediatric News and is on an advisory board for Cognoa, which is developing a medical device to diagnose autism and he is also the co-editor of the AAP's current ADHD Toolkit. Dr. Hyman had no relevant financial disclosures.
*This story was updated on Feb. 11, 2021.
To improve autism screening rates, researchers in Utah tried a range of interventions.
They added automatic reminders to the electronic health record (EHR). They started using a shorter, more sensitive screening instrument. And they trained clinicians to perform autism-specific evaluations in a primary care clinic.
The researchers found that these interventions were associated with increased rates of autism screening and referrals.
At the same time, they looked at screening and referral rates at other community clinics in their health care system. These clinics incorporated EHR reminders but not all of the other changes.
“The community clinics had an increase in screening frequency with only automatic reminders,” the researchers reported. At the two intervention clinics, however, screening rates increased more than they did at the community clinics. Referrals did not significantly increase at the community clinics.
Kathleen Campbell, MD, MHSc, a pediatric resident at the University of Utah in Salt Lake City, and colleagues described their research in a study published in Pediatrics.
Three phases
They examined more than 12,000 well-child visits for children aged 16-30 months between July 2017 and June 2019.
In all, 4,155 visits occurred at the 2 intervention clinics, and 8,078 visits occurred at the 27 community clinics in the University of Utah health care system.
From baseline through the interventions, the proportion of visits with screening increased by 51% in the intervention clinics (from 58.6% to 88.8%), and by 21% in the community clinics (from 43.4% to 52.4%). The proportion of referrals increased 1.5-fold in intervention clinics, from 1.3% to 3.3%, the authors said.
The American Academy of Pediatrics (AAP) supports screening for autism in all children starting at age 18 months, but “only 44% of children with autism have had a comprehensive autism evaluation before age 36 months,” Dr. Campbell and colleagues wrote.
In their system, about half of the children were being screened for autism, and 0.5% had autism diagnosed.
In an effort to increase the proportion of visits with screening for autism and the proportion of visits with referrals for autism evaluation, Dr. Campbell and colleagues designed a quality improvement study.
Following a baseline period, they implemented interventions in three phases.
Initially, all clinics used the Modified Checklist for Autism in Toddlers, Revised (M-CHAT-R) for autism screening. For the first phase starting in July 2018, the researchers changed the screening instrument at the two intervention clinics to the Parent’s Observation of Social Interaction (POSI). This instrument “is embedded in a broadband developmental screen, is shorter than the M-CHAT-R, and includes questions about the consistency of the child’s behavior,” the authors said. “The POSI has greater sensitivity than the M-CHAT-R ... and similar, although somewhat lower, specificity.”
In intervention phase 2 starting in November 2018, the researchers “added an automatic reminder for autism screening to the EHR health maintenance screen.” Both the intervention clinics and the community clinics received the automatic reminders.
In intervention phase 3 starting in February 2019, they “added a referral option that clinicians could use for rapid access to autism-specific evaluation ... for children who had a POSI result suggestive of autism and for whom the clinician had sufficient concerns about autism that would indicate the need for referral for autism evaluation,” the researchers said.
“Using an online tutorial, we trained three clinicians in the intervention clinics to administer an observational assessment known as the Screening Tool for Autism in Toddlers (STAT),” which requires a 30-minute visit, they said. “Children who had a STAT result suggestive of autism were referred for expedited autism diagnostic evaluation, which was performed by a multidisciplinary team in our university-based developmental assessment clinic. Children who had a STAT result that did not suggest autism did not receive further autism evaluations unless the clinician felt they still needed further evaluation at the developmental clinic.”
After the switch to POSI, the percentage of visits with a positive screen result increased from 4.7% to 13.5% in the intervention clinics.
Furthermore, referrals were 3.4 times more frequent for visits during phase 3 in the intervention clinics, relative to the baseline period.
Potential to overwhelm
“The change to a more sensitive screening instrument increased the frequency of screening results suggestive of autism and informed our improvement team of the need to implement autism evaluation in primary care to avoid overwhelming our referral system,” Dr. Campbell and coauthors reported.
Future studies may assess whether increased screening and referrals speed the time to diagnosis and treatment and improve long-term functional abilities of children with autism. Some children in the study have received an autism diagnosis, while others have not yet been evaluated.
The use of STAT in primary care may be limited by “the barriers of training providers and purchasing materials,” the authors noted. “However, the time-based billing for lengthier appointments and billing for developmental testing help to cover cost.”
The intervention clinics and community clinics were staffed by pediatric providers, including residents and attendings, said Dr. Campbell.
“The staffing is similar at the community and intervention clinics, with mostly pediatricians and some nurse practitioners,” Dr. Campbell said. “One difference is that there are a few family medicine physicians in the community clinics, but we did not study whether that made a difference in screening. At the beginning of the study the approach to screening was the same.”
From the start, the community clinics were screening for autism and referring for further autism evaluation less often than the intervention clinics. “I don’t know why they were screening less, but they did improve with the automatic reminders,” said Dr. Campbell. “We didn’t examine type of provider or type of practice in this study, but the literature suggests that family physicians do not screen for autism as often as pediatricians.”
Payment and referral challenges
In theory, the approach in the study is a great idea, but it may not be feasible to implement for many private practices, said Herschel Lessin, MD. Dr. Lessin is a senior partner of the Children’s Medical Group in New York.
“We desperately need autism screening in a primary care setting,” Dr. Lessin said. “These authors found that wasn’t being done as recommended by the AAP Bright Futures, which is a problem.”
However, the researchers incorporated the interventions in a health care system with “far more resources than most people in practice would ever have” and substituted a less familiar screening tool.
In addition, the ability to use confirmatory STAT for primary care evaluations may be limited. “Unless you can find pediatricians willing to commit 30 to 45 minutes on one of these evaluations ... few are going to do that,” he said.
“The whole problem is that there are no referrals available or very few referrals available, and that insurance payments so underpay for developmental screening and evaluation that it does not justify the time doing it, so a lot of doctors are unable to do it,” said Dr. Lessin. When a referral is warranted, developmental pediatricians may have 6- to 12-month waiting lists, he said.
“For people in clinical practice, this is not news,” Dr. Lessin said. “We know we should screen for autism. The problem is it’s time consuming. Nobody pays for it. We have no place to send them even when we are suspicious.”
From screening to diagnosis to treatment
“Autism screen approaches vary but with educational efforts on the part of the AAP, CDC, and family organizations the rates for autism screening have dramatically improved,” said Susan L. Hyman, MD, professor of pediatrics at the University of Rochester in New York. “I do not know if screening rates have been impacted by COVID.”
Dr. Hyman and coauthors wrote an AAP clinical report on the identification, evaluation, and management of children with autism spectrum disorder. The report was published in the January 2020 issue of Pediatrics.
After screening and diagnostic testing, patients most importantly need to be able to access “timely and equitable evidence-based intervention,” which should be available, said Dr. Hyman.
Although researchers have proposed training primary care providers in autism diagnostics, “older, more complex patients with co-occurring behavioral health or other developmental disorders may need more specialized diagnostic assessment than could be accomplished in a primary care setting,” Dr. Hyman added.
“However, it is very important to identify children with therapeutic needs as early as possible and move them through the continuum from screening to diagnosis to treatment in a timely fashion. It would be wonderful if symptoms could be addressed without the need for diagnosis in the very youngest children,” Dr. Hyman said. “Early symptoms, even if not autism, are likely to be appropriate for intervention – whether it is speech therapy, attention to food selectivity, sleep problems – things that impact quality of life and potential future symptoms.”
The research was supported by the Utah Stimulating Access to Research in Residency Transition Scholar award, which is funded by the National Institutes of Health.
Dr. Campbell is an inventor on a patent related to screening for autism. The study authors otherwise had no disclosures. Dr. Lessin is on the editorial advisory board for Pediatric News and is on an advisory board for Cognoa, which is developing a medical device to diagnose autism and he is also the co-editor of the AAP's current ADHD Toolkit. Dr. Hyman had no relevant financial disclosures.
*This story was updated on Feb. 11, 2021.
FROM PEDIATRICS
FDA approves intramuscular administration for peginterferon beta-1a in MS
“The new IM administration offers people living with relapsing MS the well-characterized efficacy and safety of Plegridy with the potential for significantly reduced injection site reactions,” Biogen said in a news release announcing the FDA action.
Plegridy is a pegylated version of interferon beta-1a, which prolongs the circulation time of the molecule in the body by increasing its size. The process extends the drug’s half-life, allowing for a less-frequent dosing schedule.
Peginterferon beta-1a administered subcutaneously was first approved by the FDA in 2014 based on data showing it significantly reduces MS relapses, disability progression, and brain lesions.
The FDA approved IM administration for peginterferon beta-1a based on data evaluating bioequivalence and adverse reactions associated with IM administration compared with subcutaneous (SC) administration in healthy volunteers.
Bioequivalence of the IM and SC dosing regimens was confirmed and volunteers receiving the drug through IM administration experienced fewer injection site reactions relative to those receiving SC administration (14.4% vs. 32.1%), the company said.
The overall safety profiles of IM and SC administration were generally similar, with no new safety signals.
The European Commission allowed marketing authorization for IM administration of peginterferon beta-1a in December 2020.
A version of this article first appeared on Medscape.com.
“The new IM administration offers people living with relapsing MS the well-characterized efficacy and safety of Plegridy with the potential for significantly reduced injection site reactions,” Biogen said in a news release announcing the FDA action.
Plegridy is a pegylated version of interferon beta-1a, which prolongs the circulation time of the molecule in the body by increasing its size. The process extends the drug’s half-life, allowing for a less-frequent dosing schedule.
Peginterferon beta-1a administered subcutaneously was first approved by the FDA in 2014 based on data showing it significantly reduces MS relapses, disability progression, and brain lesions.
The FDA approved IM administration for peginterferon beta-1a based on data evaluating bioequivalence and adverse reactions associated with IM administration compared with subcutaneous (SC) administration in healthy volunteers.
Bioequivalence of the IM and SC dosing regimens was confirmed and volunteers receiving the drug through IM administration experienced fewer injection site reactions relative to those receiving SC administration (14.4% vs. 32.1%), the company said.
The overall safety profiles of IM and SC administration were generally similar, with no new safety signals.
The European Commission allowed marketing authorization for IM administration of peginterferon beta-1a in December 2020.
A version of this article first appeared on Medscape.com.
“The new IM administration offers people living with relapsing MS the well-characterized efficacy and safety of Plegridy with the potential for significantly reduced injection site reactions,” Biogen said in a news release announcing the FDA action.
Plegridy is a pegylated version of interferon beta-1a, which prolongs the circulation time of the molecule in the body by increasing its size. The process extends the drug’s half-life, allowing for a less-frequent dosing schedule.
Peginterferon beta-1a administered subcutaneously was first approved by the FDA in 2014 based on data showing it significantly reduces MS relapses, disability progression, and brain lesions.
The FDA approved IM administration for peginterferon beta-1a based on data evaluating bioequivalence and adverse reactions associated with IM administration compared with subcutaneous (SC) administration in healthy volunteers.
Bioequivalence of the IM and SC dosing regimens was confirmed and volunteers receiving the drug through IM administration experienced fewer injection site reactions relative to those receiving SC administration (14.4% vs. 32.1%), the company said.
The overall safety profiles of IM and SC administration were generally similar, with no new safety signals.
The European Commission allowed marketing authorization for IM administration of peginterferon beta-1a in December 2020.
A version of this article first appeared on Medscape.com.
Afternoon napping associated with better cognition in elderly, study shows
according to a new study in General Psychiatry.
The findings add to those seen in other observational studies showing afternoon napping promotes cognitive function, said the authors of the paper, published in General Psychiatry.
“The prevalence of afternoon napping has been increasing in older adults much more than in younger individuals,” wrote Han Cai, MS, of the department of geriatrics at The Fourth People’s Hospital of Wuhu, Anhui, China, and coauthors. “The elderly individuals who took afternoon naps showed significantly higher cognitive performance compared with those who did not nap.”
The researchers enrolled 2,214 people in the study – all Han Chinese and aged 60 or older. Afternoon napping was considered any period of inactivity of at least 5 minutes but less than 2 hours after lunch and outside of the person’s main sleep schedule. Those who reported ever napping – 1,534 subjects – were included in the napping group, and the others – 680 – in the nonnapping group. Patients with major physical conditions were excluded.
The Montreal Cognitive Assessment (MoCA), the Mini-Mental State Examination (MMSE), and the Neuropsychological Test Battery (NTB) were used to measure cognitive function, and 739 patients agreed to blood tests for lipid values.
The average total MMSE score was higher for the napping group at 25.3 points out of 30, than for the nonnapping group, at 24.56 (P = .003). Those in the napping group also had significantly higher scores in the orientation portion of the MoCA test, at 5.55 out of 6 points, compared with 5.41 for the nonnapping group (P = .006).
Those in the napping group scored significantly higher on the digit span and language fluency parts of the Neuropsychological Test Battery (P = .009 and .020, respectively).
Dementia was assessed with face-to-face visits with clinicians, but diagnoses of dementia were not different between the groups.
Triglycerides were found to be higher – though still in the normal range – in the napping group compared with the nonnapping group, 1.80 mmol/L to 1.75 mmol/L, the researchers found (P = .001). No differences were seen for HDL or LDL cholesterol levels, or in hypertension or diabetes, the researchers reported.
The authors noted that inflammation is likely an important feature in the relationship between napping and cognitive function. Inflammatory cytokines have been found to play a role in sleep disorders, and strong inflammatory responses can lead to adverse events, including cognitive impairment.
“Sleep is known to be a regulator of the immune response that counters these inflammatory mediators, whereas napping, in particular, is thought to be an evolved response to inflammation,” they said.
The average age of patients in the napping group was 72.8 years, slightly older than those in the nonnapping group at 71.3 years, and this was a significant difference (P = .016).
The researchers acknowledged that the study “could not show direct causality of napping, whether beneficial or harmful,” and that “a lack of detailed information regarding napping duration ... also limited the description of napping status.”
Junxin Li, PhD, RN, assistant professor at Johns Hopkins School of Nursing, Baltimore, who has studied napping and cognition, said that previous research generally supports a U-shaped relationship between napping and mental acuity, with shorter or medium-length naps benefiting cognition and no naps or naps that are too long being detrimental.
“This study looked at no nap versus naps of less than 2 hours and may not be able to capture this potential U-shaped association,” she said.
For clinicians, the duration, timing, frequency, and purpose of naps are important factors in making recommendations to patients, she said.
“For example, timing – napping in the early evening close to older adult’s bedtime may delay their bedtime and interfere with their nighttime sleep quality. Taking naps after lunchtime is hypothesized to provide the most therapeutic values to the health and usually recommended,” she said. Regular napping is better than “randomly dozing off,” Dr. Li added.
There are also cultural considerations – in east Asia, napping tends to be considered part of a healthy lifestyle, while in western countries it is not – and this could impact napping behaviors and how these behaviors affect cognition, she said.
Phyllis C. Zee, MD, PhD, director of the Center for Circadian and Sleep Medicine at the Northwestern University, Chicago, said the results are consistent with early cross-sectional studies that showed that regular, scheduled naps in the afternoon were associated with positive cognitive performance and lower cardiometabolic disease risk.
Dr. Zee noted that it’s important to recognize that the positive data are associated with naps that are planned, while older adults napping because of excess sleepiness are at a higher risk for cognitive impairment and other health issues.
The study authors, Dr. Li, and Dr. Zee reported no relevant financial disclosures.
according to a new study in General Psychiatry.
The findings add to those seen in other observational studies showing afternoon napping promotes cognitive function, said the authors of the paper, published in General Psychiatry.
“The prevalence of afternoon napping has been increasing in older adults much more than in younger individuals,” wrote Han Cai, MS, of the department of geriatrics at The Fourth People’s Hospital of Wuhu, Anhui, China, and coauthors. “The elderly individuals who took afternoon naps showed significantly higher cognitive performance compared with those who did not nap.”
The researchers enrolled 2,214 people in the study – all Han Chinese and aged 60 or older. Afternoon napping was considered any period of inactivity of at least 5 minutes but less than 2 hours after lunch and outside of the person’s main sleep schedule. Those who reported ever napping – 1,534 subjects – were included in the napping group, and the others – 680 – in the nonnapping group. Patients with major physical conditions were excluded.
The Montreal Cognitive Assessment (MoCA), the Mini-Mental State Examination (MMSE), and the Neuropsychological Test Battery (NTB) were used to measure cognitive function, and 739 patients agreed to blood tests for lipid values.
The average total MMSE score was higher for the napping group at 25.3 points out of 30, than for the nonnapping group, at 24.56 (P = .003). Those in the napping group also had significantly higher scores in the orientation portion of the MoCA test, at 5.55 out of 6 points, compared with 5.41 for the nonnapping group (P = .006).
Those in the napping group scored significantly higher on the digit span and language fluency parts of the Neuropsychological Test Battery (P = .009 and .020, respectively).
Dementia was assessed with face-to-face visits with clinicians, but diagnoses of dementia were not different between the groups.
Triglycerides were found to be higher – though still in the normal range – in the napping group compared with the nonnapping group, 1.80 mmol/L to 1.75 mmol/L, the researchers found (P = .001). No differences were seen for HDL or LDL cholesterol levels, or in hypertension or diabetes, the researchers reported.
The authors noted that inflammation is likely an important feature in the relationship between napping and cognitive function. Inflammatory cytokines have been found to play a role in sleep disorders, and strong inflammatory responses can lead to adverse events, including cognitive impairment.
“Sleep is known to be a regulator of the immune response that counters these inflammatory mediators, whereas napping, in particular, is thought to be an evolved response to inflammation,” they said.
The average age of patients in the napping group was 72.8 years, slightly older than those in the nonnapping group at 71.3 years, and this was a significant difference (P = .016).
The researchers acknowledged that the study “could not show direct causality of napping, whether beneficial or harmful,” and that “a lack of detailed information regarding napping duration ... also limited the description of napping status.”
Junxin Li, PhD, RN, assistant professor at Johns Hopkins School of Nursing, Baltimore, who has studied napping and cognition, said that previous research generally supports a U-shaped relationship between napping and mental acuity, with shorter or medium-length naps benefiting cognition and no naps or naps that are too long being detrimental.
“This study looked at no nap versus naps of less than 2 hours and may not be able to capture this potential U-shaped association,” she said.
For clinicians, the duration, timing, frequency, and purpose of naps are important factors in making recommendations to patients, she said.
“For example, timing – napping in the early evening close to older adult’s bedtime may delay their bedtime and interfere with their nighttime sleep quality. Taking naps after lunchtime is hypothesized to provide the most therapeutic values to the health and usually recommended,” she said. Regular napping is better than “randomly dozing off,” Dr. Li added.
There are also cultural considerations – in east Asia, napping tends to be considered part of a healthy lifestyle, while in western countries it is not – and this could impact napping behaviors and how these behaviors affect cognition, she said.
Phyllis C. Zee, MD, PhD, director of the Center for Circadian and Sleep Medicine at the Northwestern University, Chicago, said the results are consistent with early cross-sectional studies that showed that regular, scheduled naps in the afternoon were associated with positive cognitive performance and lower cardiometabolic disease risk.
Dr. Zee noted that it’s important to recognize that the positive data are associated with naps that are planned, while older adults napping because of excess sleepiness are at a higher risk for cognitive impairment and other health issues.
The study authors, Dr. Li, and Dr. Zee reported no relevant financial disclosures.
according to a new study in General Psychiatry.
The findings add to those seen in other observational studies showing afternoon napping promotes cognitive function, said the authors of the paper, published in General Psychiatry.
“The prevalence of afternoon napping has been increasing in older adults much more than in younger individuals,” wrote Han Cai, MS, of the department of geriatrics at The Fourth People’s Hospital of Wuhu, Anhui, China, and coauthors. “The elderly individuals who took afternoon naps showed significantly higher cognitive performance compared with those who did not nap.”
The researchers enrolled 2,214 people in the study – all Han Chinese and aged 60 or older. Afternoon napping was considered any period of inactivity of at least 5 minutes but less than 2 hours after lunch and outside of the person’s main sleep schedule. Those who reported ever napping – 1,534 subjects – were included in the napping group, and the others – 680 – in the nonnapping group. Patients with major physical conditions were excluded.
The Montreal Cognitive Assessment (MoCA), the Mini-Mental State Examination (MMSE), and the Neuropsychological Test Battery (NTB) were used to measure cognitive function, and 739 patients agreed to blood tests for lipid values.
The average total MMSE score was higher for the napping group at 25.3 points out of 30, than for the nonnapping group, at 24.56 (P = .003). Those in the napping group also had significantly higher scores in the orientation portion of the MoCA test, at 5.55 out of 6 points, compared with 5.41 for the nonnapping group (P = .006).
Those in the napping group scored significantly higher on the digit span and language fluency parts of the Neuropsychological Test Battery (P = .009 and .020, respectively).
Dementia was assessed with face-to-face visits with clinicians, but diagnoses of dementia were not different between the groups.
Triglycerides were found to be higher – though still in the normal range – in the napping group compared with the nonnapping group, 1.80 mmol/L to 1.75 mmol/L, the researchers found (P = .001). No differences were seen for HDL or LDL cholesterol levels, or in hypertension or diabetes, the researchers reported.
The authors noted that inflammation is likely an important feature in the relationship between napping and cognitive function. Inflammatory cytokines have been found to play a role in sleep disorders, and strong inflammatory responses can lead to adverse events, including cognitive impairment.
“Sleep is known to be a regulator of the immune response that counters these inflammatory mediators, whereas napping, in particular, is thought to be an evolved response to inflammation,” they said.
The average age of patients in the napping group was 72.8 years, slightly older than those in the nonnapping group at 71.3 years, and this was a significant difference (P = .016).
The researchers acknowledged that the study “could not show direct causality of napping, whether beneficial or harmful,” and that “a lack of detailed information regarding napping duration ... also limited the description of napping status.”
Junxin Li, PhD, RN, assistant professor at Johns Hopkins School of Nursing, Baltimore, who has studied napping and cognition, said that previous research generally supports a U-shaped relationship between napping and mental acuity, with shorter or medium-length naps benefiting cognition and no naps or naps that are too long being detrimental.
“This study looked at no nap versus naps of less than 2 hours and may not be able to capture this potential U-shaped association,” she said.
For clinicians, the duration, timing, frequency, and purpose of naps are important factors in making recommendations to patients, she said.
“For example, timing – napping in the early evening close to older adult’s bedtime may delay their bedtime and interfere with their nighttime sleep quality. Taking naps after lunchtime is hypothesized to provide the most therapeutic values to the health and usually recommended,” she said. Regular napping is better than “randomly dozing off,” Dr. Li added.
There are also cultural considerations – in east Asia, napping tends to be considered part of a healthy lifestyle, while in western countries it is not – and this could impact napping behaviors and how these behaviors affect cognition, she said.
Phyllis C. Zee, MD, PhD, director of the Center for Circadian and Sleep Medicine at the Northwestern University, Chicago, said the results are consistent with early cross-sectional studies that showed that regular, scheduled naps in the afternoon were associated with positive cognitive performance and lower cardiometabolic disease risk.
Dr. Zee noted that it’s important to recognize that the positive data are associated with naps that are planned, while older adults napping because of excess sleepiness are at a higher risk for cognitive impairment and other health issues.
The study authors, Dr. Li, and Dr. Zee reported no relevant financial disclosures.
Advanced Imaging Study Reveals How COVID-19 Attacks the Brain
Researchers from the National Institute of Neurological Disorders and Stroke studying the brains of patients who died from COVID-19, “consistently” found microvascular damage—but no signs of COVID-19 infection. Of the 19 patients in the study, 14 had chronic illnesses, including diabetes mellitus and hypertension, and 11 had ben found dead or had died unexpectedly. Of the 16 with available medical histories, one had delirium and the others had respiratory or unknown symptoms. Two had pulmonary embolism.
Patients with COVID-19 often have neurological problems, such as headaches, delirium, and dizziness. Some have strokes. Several studies have shown that COVID-19 can cause inflammation and blood vessel damage, but the precise mode of action is still unclear. In this study, the researchers used a magnetic resonance imaging (MRI) scanner 4 to 10 times more sensitive than most MRI scanners to examine samples of the olfactory bulbs and brainstems from the samples.
In 9 patients, the MRI scan showed punctate hyperintensities (bright spots representing areas of microvascular injury and fibrinogen leakage) that often indicate inflammation. In 10 brains, they found punctate hypointensities (dark spots) that corresponded to congested blood vessels, with surrounding areas of fibrinogen leakage and relatively intact vasculature. Areas of linear hypointensities (dark spots) were interpreted as microhemorrhages.
Using the scans as a guide, the researchers examined the spots more closely under a microscope. They found that the bright spots contained blood vessels that were thinner than normal and sometimes leaked blood proteins into the brain. This, the researchers say, seemed to trigger an immune reaction. The spots were surrounded by T cells from the blood and the brain’s own immune cells. In contrast, the dark spots contained clotted and leaky blood vessels but no immune response.
Moreover, although they used several methods for detecting genetic material or proteins from SAS-CoV-2, they found none. It’s possible, the researchers say, that the virus was cleared by the time of death or that viral copy numbers were undetectable by their assays.
We were completely surprised,” said Avindra Nath, MD, NINDS clinical director. “Originally, we expected to see damage that is caused by a lack of oxygen. Instead, we saw multifocal areas of damage that is usually associated with strokes and neuroinflammatory diseases.”
In future, Nath says, they plan to study how COVID-19 harms the blood vessels and whether that produces some of the short- and long-term symptoms seen. “We hope these results will help doctors understand the full spectrum of problems patients may suffer so that we can come up with better treatments.”
Researchers from the National Institute of Neurological Disorders and Stroke studying the brains of patients who died from COVID-19, “consistently” found microvascular damage—but no signs of COVID-19 infection. Of the 19 patients in the study, 14 had chronic illnesses, including diabetes mellitus and hypertension, and 11 had ben found dead or had died unexpectedly. Of the 16 with available medical histories, one had delirium and the others had respiratory or unknown symptoms. Two had pulmonary embolism.
Patients with COVID-19 often have neurological problems, such as headaches, delirium, and dizziness. Some have strokes. Several studies have shown that COVID-19 can cause inflammation and blood vessel damage, but the precise mode of action is still unclear. In this study, the researchers used a magnetic resonance imaging (MRI) scanner 4 to 10 times more sensitive than most MRI scanners to examine samples of the olfactory bulbs and brainstems from the samples.
In 9 patients, the MRI scan showed punctate hyperintensities (bright spots representing areas of microvascular injury and fibrinogen leakage) that often indicate inflammation. In 10 brains, they found punctate hypointensities (dark spots) that corresponded to congested blood vessels, with surrounding areas of fibrinogen leakage and relatively intact vasculature. Areas of linear hypointensities (dark spots) were interpreted as microhemorrhages.
Using the scans as a guide, the researchers examined the spots more closely under a microscope. They found that the bright spots contained blood vessels that were thinner than normal and sometimes leaked blood proteins into the brain. This, the researchers say, seemed to trigger an immune reaction. The spots were surrounded by T cells from the blood and the brain’s own immune cells. In contrast, the dark spots contained clotted and leaky blood vessels but no immune response.
Moreover, although they used several methods for detecting genetic material or proteins from SAS-CoV-2, they found none. It’s possible, the researchers say, that the virus was cleared by the time of death or that viral copy numbers were undetectable by their assays.
We were completely surprised,” said Avindra Nath, MD, NINDS clinical director. “Originally, we expected to see damage that is caused by a lack of oxygen. Instead, we saw multifocal areas of damage that is usually associated with strokes and neuroinflammatory diseases.”
In future, Nath says, they plan to study how COVID-19 harms the blood vessels and whether that produces some of the short- and long-term symptoms seen. “We hope these results will help doctors understand the full spectrum of problems patients may suffer so that we can come up with better treatments.”
Researchers from the National Institute of Neurological Disorders and Stroke studying the brains of patients who died from COVID-19, “consistently” found microvascular damage—but no signs of COVID-19 infection. Of the 19 patients in the study, 14 had chronic illnesses, including diabetes mellitus and hypertension, and 11 had ben found dead or had died unexpectedly. Of the 16 with available medical histories, one had delirium and the others had respiratory or unknown symptoms. Two had pulmonary embolism.
Patients with COVID-19 often have neurological problems, such as headaches, delirium, and dizziness. Some have strokes. Several studies have shown that COVID-19 can cause inflammation and blood vessel damage, but the precise mode of action is still unclear. In this study, the researchers used a magnetic resonance imaging (MRI) scanner 4 to 10 times more sensitive than most MRI scanners to examine samples of the olfactory bulbs and brainstems from the samples.
In 9 patients, the MRI scan showed punctate hyperintensities (bright spots representing areas of microvascular injury and fibrinogen leakage) that often indicate inflammation. In 10 brains, they found punctate hypointensities (dark spots) that corresponded to congested blood vessels, with surrounding areas of fibrinogen leakage and relatively intact vasculature. Areas of linear hypointensities (dark spots) were interpreted as microhemorrhages.
Using the scans as a guide, the researchers examined the spots more closely under a microscope. They found that the bright spots contained blood vessels that were thinner than normal and sometimes leaked blood proteins into the brain. This, the researchers say, seemed to trigger an immune reaction. The spots were surrounded by T cells from the blood and the brain’s own immune cells. In contrast, the dark spots contained clotted and leaky blood vessels but no immune response.
Moreover, although they used several methods for detecting genetic material or proteins from SAS-CoV-2, they found none. It’s possible, the researchers say, that the virus was cleared by the time of death or that viral copy numbers were undetectable by their assays.
We were completely surprised,” said Avindra Nath, MD, NINDS clinical director. “Originally, we expected to see damage that is caused by a lack of oxygen. Instead, we saw multifocal areas of damage that is usually associated with strokes and neuroinflammatory diseases.”
In future, Nath says, they plan to study how COVID-19 harms the blood vessels and whether that produces some of the short- and long-term symptoms seen. “We hope these results will help doctors understand the full spectrum of problems patients may suffer so that we can come up with better treatments.”
Is the incidence of depressive disorders increased following cerebral concussion?
EVIDENCE SUMMARY
Higher odds of depression in youth and adolescents with concussion
A 2019 prospective cohort study used data from the 2017 Nevada Youth Risk Behavior Surveillance Survey (YRBSS) to evaluate the relationship between concussion and depression in high school students.1 Included students were physically active for at least 60 minutes on 5 or more days per week or played on at least 1 sports team (N = 3427; 9th-12th grade students from 98 schools). When compared to the total population of included students and controlled for covariates, those who self-reported a concussion within the past 12 months (N = 664) had a higher adjusted odds ratio (aOR) of depressive symptoms (aOR = 1.5; 95% confidence interval [CI], 1.1-1.9). Depressive symptoms were reported in 38.1% of patients with a history of concussion, compared to 29.2% of patients who did not report a concussion in the past 12 months.
A 2014 retrospective cohort study examined data from the 2007-2008 National Survey of Children’s Health and evaluated the association between previous concussion and current depression diagnosis in youth ages 12 to 17 years without a current concussion (N = 36,060).2 Parents were contacted by random-digit dialing, prompted with a description of depression, and asked if their child currently had a clinical diagnosis of depression and whether a concussion had ever been diagnosed. A prior diagnosis of concussion was associated with greater risk for current depression compared to youth with no concussion history (aOR = 3.3; 95% CI, 2-5.5). Current depression was reported in 10.1% of patients with a history of concussion compared to 3.4% of patients with no history of concussion.
Findings vary among college athletes
A 2015 case-control study examined the prevalence of depressive symptoms in college athletes diagnosed with concussion compared to an athletic control group.3 The intervention group (N = 84; 77% male; average age, 18.4 years) received a concussion diagnosis from the team physician or certified athletic trainer. The athletic control group (N = 42; 55% male; average age, 18.9 years) reported no concussions in the past year.
The Beck Depression Inventory–Fast Screen (BDI-FS) was administered to the concussion group at baseline and postconcussion, and to the control group at 2 time points, with an average interval of 6.8 weeks. A score of ≥ 4 on the BDI-FS (scoring range, 0-21; higher score suggestive of more severe depression) indicated athletes at risk for depression. Concussed athletes exhibited a statistically significant increase in depression symptoms compared to control participants (20% vs 5%; x21 = 5.2; P = .02).
A 2018 cross-sectional study examined the association between concussion and adverse health outcomes in former college football players who played at least 1 year in college (1999-2001) but had no professional football experience.4 The cohort (N = 204; average age, 35) self-reported (15 years after their college career ended) the number of concussions sustained during high school and college sports performance. Reports were then stratified into 3 categories: no concussions, 1 or 2 concussions, and ≥ 3 concussions. The Patient Health Questionnaire (PHQ-9) was used to screen for depression, with scores categorized to no or mild depression (< 10) and moderate-to-severe depression (≥ 10).
Controlling for body mass index, athletes reporting ≥ 3 concussions had a higher prevalence of depression compared to those reporting no concussions (prevalence ratio [PR] = 4.2; 95% CI, 1.0-16.3) or 1 to 2 concussions (PR = 2.8; 95% CI, 1.3-6.0). No statistically significant association between concussion and depression was observed with athletes reporting 1 to 2 concussions compared to 0 concussions.
A 2015 prospective longitudinal cohort study examined postinjury depressive symptoms in 3 groups of Division 1 male and female college student athletes (N = 21; ages 18-22).5 Physician-diagnosed concussed (N = 7) and injured but nonconcussed (N = 7) athletes completed the Center for Epidemiological Studies Depression Scale (CES-D) at baseline and at 1 week, 1 month, and 3 months postinjury. Sport-matched healthy athletes (N = 7) completed it only at baseline. A CES-D score of ≥ 16 (range, 0-60) indicated a risk for clinical depression. Participants with a history of depression or other injury resulting in ≥ 1 day of time lost within the past 3 months were excluded.
Continue to: While both groups...
While both groups showed a significant increase from baseline CES-D scores, there were no significant differences in depressive symptoms between concussed (mean CES-D score ± standard deviation [SD]: baseline, 6.7 ± 3.9; 1 week, 11 ± 5.3; 1 month, 8.3 ± 5; 3 months, 6.4 ± 5.4) and injured but nonconcussed participants (mean CES-D score ± SD: baseline, 5.7 ± 2.8; 1 week, 9.1 ± 4; 1 month, 8.9 ± 4.6; 3 months, 6.9 ± 2.8) at any of the postinjury time points.
Findings among semipro and pro athletes appear to vary by sport
A 2016 prospective cohort study assessed the impact of concussive events on incidence of depression in active semiprofessional and professional football players who had previously sustained ≥ 1 concussions.6 Participants (N = 27) answered an anonymous online survey that included the revised version of the CES-D (CESD-R) to determine level of depression (a score of ≥ 16 defined clinical depression). Players with a CESD-R score ≥ 16 (N = 16) sustained a significantly greater average number of concussions compared to those who scored < 16 (N = 11; 3.8 vs. 1.6, P = .0004). Players who sustained ≥ 3 concussions scored significantly higher on the CESD-R than players with ≤ 2 concussions (average score, 24 vs 15.6; P = .03).
A 2017 case-control study examined the long-term health outcomes of retired Scottish male rugby players (N = 52; mean age, 54 years) with a history of mild concussion compared to males of similar age with no previous history of concussion (N = 29; mean age, 55).7 The Hospital Anxiety and Depression Scale (HADS) was used to assess depression on a 21-point scale (normal = 0-7; borderline, 8-10; abnormal, 11-21). There was no significant difference observed in mean HADS scores between the rugby players and controls, respectively (2.8 ± 2.1 vs 2.6 ± 2 .8; P = .941).
A 2013 case-control study of 30 retired NFL players with 29 controls matched for age, estimated IQ, and education examined the relationship between a remote history of concussion and current symptoms of depression.8 Concussion history was self-reported by the retired players. Controls with a history of concussion were excluded from the study. The Beck Depression Inventory-II (BDI-II) was used to measure depression symptoms, with a score of 1 to 9 designating minimal depression and ≥ 10 mild-to-moderate depression. Retired players scored significantly higher on the BDI-II compared to the controls (8.8 vs 2.8; P = .001).
Editor’s takeaway
Concussions include cognitive compromise. An astute clinician’s concern for depression as a sequela makes sense. This evidence contributes to that conjecture. However, the authors of this Clinical Inquiry correctly outline the limitations, inconsistencies, and biases of the evidence. The exact relationship—degree and context—between concussion and depression remains vague.
1. Yang MN, Clements-Nolle K, Parrish B, et al. Adolescent concussion and mental health outcomes: a population-based study. Am J Health Behav. 2019;43:258-265.
2. Chrisman SPD, Richardson LP. Prevalence of diagnosed depression in adolescents with history of concussion. J Adolesc Health. 2014;54:582-586.
3. Vargas G, Rabinowitz A, Meyer J, et al. Predictors and prevalence of postconcussion depression symptoms in collegiate athletes. J Athl Train. 2015;50:250-255.
4. Kerr ZY, Thomas LC, Simon JE, et al. Association between history of multiple concussions and health outcomes among former college football players. Am J Sports Med. 2018;46:1733-1741.
5. Roiger T, Weidauer L, Kern B. A longitudinal pilot study of depressive symptoms in concussed and injured/nonconcussed National Collegiate Athletic Association Division I student-athletes. J Athl Train. 2015;50:256-261.
6. Pryor J, Larson A, DeBeliso M. The prevalence of depression and concussions in a sample of active North American semi-professional and professional football players. J Lifestyle Med. 2016;6:7-15.
7. McMillan TM, McSkimming P, Wainman-Lefley J, et al. Long-term health outcomes after exposure to repeated concussion in elite level: rugby union players. J Neurol Neurosurg Psychiatry. 2017;88:505-511.
8. Didehbani N, Munro Cullum C, Mansinghani S, et al. Depressive symptoms and concussions in aging retired NFL players. Arch Clin Neuropsychol. 2013;28:418-424.
EVIDENCE SUMMARY
Higher odds of depression in youth and adolescents with concussion
A 2019 prospective cohort study used data from the 2017 Nevada Youth Risk Behavior Surveillance Survey (YRBSS) to evaluate the relationship between concussion and depression in high school students.1 Included students were physically active for at least 60 minutes on 5 or more days per week or played on at least 1 sports team (N = 3427; 9th-12th grade students from 98 schools). When compared to the total population of included students and controlled for covariates, those who self-reported a concussion within the past 12 months (N = 664) had a higher adjusted odds ratio (aOR) of depressive symptoms (aOR = 1.5; 95% confidence interval [CI], 1.1-1.9). Depressive symptoms were reported in 38.1% of patients with a history of concussion, compared to 29.2% of patients who did not report a concussion in the past 12 months.
A 2014 retrospective cohort study examined data from the 2007-2008 National Survey of Children’s Health and evaluated the association between previous concussion and current depression diagnosis in youth ages 12 to 17 years without a current concussion (N = 36,060).2 Parents were contacted by random-digit dialing, prompted with a description of depression, and asked if their child currently had a clinical diagnosis of depression and whether a concussion had ever been diagnosed. A prior diagnosis of concussion was associated with greater risk for current depression compared to youth with no concussion history (aOR = 3.3; 95% CI, 2-5.5). Current depression was reported in 10.1% of patients with a history of concussion compared to 3.4% of patients with no history of concussion.
Findings vary among college athletes
A 2015 case-control study examined the prevalence of depressive symptoms in college athletes diagnosed with concussion compared to an athletic control group.3 The intervention group (N = 84; 77% male; average age, 18.4 years) received a concussion diagnosis from the team physician or certified athletic trainer. The athletic control group (N = 42; 55% male; average age, 18.9 years) reported no concussions in the past year.
The Beck Depression Inventory–Fast Screen (BDI-FS) was administered to the concussion group at baseline and postconcussion, and to the control group at 2 time points, with an average interval of 6.8 weeks. A score of ≥ 4 on the BDI-FS (scoring range, 0-21; higher score suggestive of more severe depression) indicated athletes at risk for depression. Concussed athletes exhibited a statistically significant increase in depression symptoms compared to control participants (20% vs 5%; x21 = 5.2; P = .02).
A 2018 cross-sectional study examined the association between concussion and adverse health outcomes in former college football players who played at least 1 year in college (1999-2001) but had no professional football experience.4 The cohort (N = 204; average age, 35) self-reported (15 years after their college career ended) the number of concussions sustained during high school and college sports performance. Reports were then stratified into 3 categories: no concussions, 1 or 2 concussions, and ≥ 3 concussions. The Patient Health Questionnaire (PHQ-9) was used to screen for depression, with scores categorized to no or mild depression (< 10) and moderate-to-severe depression (≥ 10).
Controlling for body mass index, athletes reporting ≥ 3 concussions had a higher prevalence of depression compared to those reporting no concussions (prevalence ratio [PR] = 4.2; 95% CI, 1.0-16.3) or 1 to 2 concussions (PR = 2.8; 95% CI, 1.3-6.0). No statistically significant association between concussion and depression was observed with athletes reporting 1 to 2 concussions compared to 0 concussions.
A 2015 prospective longitudinal cohort study examined postinjury depressive symptoms in 3 groups of Division 1 male and female college student athletes (N = 21; ages 18-22).5 Physician-diagnosed concussed (N = 7) and injured but nonconcussed (N = 7) athletes completed the Center for Epidemiological Studies Depression Scale (CES-D) at baseline and at 1 week, 1 month, and 3 months postinjury. Sport-matched healthy athletes (N = 7) completed it only at baseline. A CES-D score of ≥ 16 (range, 0-60) indicated a risk for clinical depression. Participants with a history of depression or other injury resulting in ≥ 1 day of time lost within the past 3 months were excluded.
Continue to: While both groups...
While both groups showed a significant increase from baseline CES-D scores, there were no significant differences in depressive symptoms between concussed (mean CES-D score ± standard deviation [SD]: baseline, 6.7 ± 3.9; 1 week, 11 ± 5.3; 1 month, 8.3 ± 5; 3 months, 6.4 ± 5.4) and injured but nonconcussed participants (mean CES-D score ± SD: baseline, 5.7 ± 2.8; 1 week, 9.1 ± 4; 1 month, 8.9 ± 4.6; 3 months, 6.9 ± 2.8) at any of the postinjury time points.
Findings among semipro and pro athletes appear to vary by sport
A 2016 prospective cohort study assessed the impact of concussive events on incidence of depression in active semiprofessional and professional football players who had previously sustained ≥ 1 concussions.6 Participants (N = 27) answered an anonymous online survey that included the revised version of the CES-D (CESD-R) to determine level of depression (a score of ≥ 16 defined clinical depression). Players with a CESD-R score ≥ 16 (N = 16) sustained a significantly greater average number of concussions compared to those who scored < 16 (N = 11; 3.8 vs. 1.6, P = .0004). Players who sustained ≥ 3 concussions scored significantly higher on the CESD-R than players with ≤ 2 concussions (average score, 24 vs 15.6; P = .03).
A 2017 case-control study examined the long-term health outcomes of retired Scottish male rugby players (N = 52; mean age, 54 years) with a history of mild concussion compared to males of similar age with no previous history of concussion (N = 29; mean age, 55).7 The Hospital Anxiety and Depression Scale (HADS) was used to assess depression on a 21-point scale (normal = 0-7; borderline, 8-10; abnormal, 11-21). There was no significant difference observed in mean HADS scores between the rugby players and controls, respectively (2.8 ± 2.1 vs 2.6 ± 2 .8; P = .941).
A 2013 case-control study of 30 retired NFL players with 29 controls matched for age, estimated IQ, and education examined the relationship between a remote history of concussion and current symptoms of depression.8 Concussion history was self-reported by the retired players. Controls with a history of concussion were excluded from the study. The Beck Depression Inventory-II (BDI-II) was used to measure depression symptoms, with a score of 1 to 9 designating minimal depression and ≥ 10 mild-to-moderate depression. Retired players scored significantly higher on the BDI-II compared to the controls (8.8 vs 2.8; P = .001).
Editor’s takeaway
Concussions include cognitive compromise. An astute clinician’s concern for depression as a sequela makes sense. This evidence contributes to that conjecture. However, the authors of this Clinical Inquiry correctly outline the limitations, inconsistencies, and biases of the evidence. The exact relationship—degree and context—between concussion and depression remains vague.
EVIDENCE SUMMARY
Higher odds of depression in youth and adolescents with concussion
A 2019 prospective cohort study used data from the 2017 Nevada Youth Risk Behavior Surveillance Survey (YRBSS) to evaluate the relationship between concussion and depression in high school students.1 Included students were physically active for at least 60 minutes on 5 or more days per week or played on at least 1 sports team (N = 3427; 9th-12th grade students from 98 schools). When compared to the total population of included students and controlled for covariates, those who self-reported a concussion within the past 12 months (N = 664) had a higher adjusted odds ratio (aOR) of depressive symptoms (aOR = 1.5; 95% confidence interval [CI], 1.1-1.9). Depressive symptoms were reported in 38.1% of patients with a history of concussion, compared to 29.2% of patients who did not report a concussion in the past 12 months.
A 2014 retrospective cohort study examined data from the 2007-2008 National Survey of Children’s Health and evaluated the association between previous concussion and current depression diagnosis in youth ages 12 to 17 years without a current concussion (N = 36,060).2 Parents were contacted by random-digit dialing, prompted with a description of depression, and asked if their child currently had a clinical diagnosis of depression and whether a concussion had ever been diagnosed. A prior diagnosis of concussion was associated with greater risk for current depression compared to youth with no concussion history (aOR = 3.3; 95% CI, 2-5.5). Current depression was reported in 10.1% of patients with a history of concussion compared to 3.4% of patients with no history of concussion.
Findings vary among college athletes
A 2015 case-control study examined the prevalence of depressive symptoms in college athletes diagnosed with concussion compared to an athletic control group.3 The intervention group (N = 84; 77% male; average age, 18.4 years) received a concussion diagnosis from the team physician or certified athletic trainer. The athletic control group (N = 42; 55% male; average age, 18.9 years) reported no concussions in the past year.
The Beck Depression Inventory–Fast Screen (BDI-FS) was administered to the concussion group at baseline and postconcussion, and to the control group at 2 time points, with an average interval of 6.8 weeks. A score of ≥ 4 on the BDI-FS (scoring range, 0-21; higher score suggestive of more severe depression) indicated athletes at risk for depression. Concussed athletes exhibited a statistically significant increase in depression symptoms compared to control participants (20% vs 5%; x21 = 5.2; P = .02).
A 2018 cross-sectional study examined the association between concussion and adverse health outcomes in former college football players who played at least 1 year in college (1999-2001) but had no professional football experience.4 The cohort (N = 204; average age, 35) self-reported (15 years after their college career ended) the number of concussions sustained during high school and college sports performance. Reports were then stratified into 3 categories: no concussions, 1 or 2 concussions, and ≥ 3 concussions. The Patient Health Questionnaire (PHQ-9) was used to screen for depression, with scores categorized to no or mild depression (< 10) and moderate-to-severe depression (≥ 10).
Controlling for body mass index, athletes reporting ≥ 3 concussions had a higher prevalence of depression compared to those reporting no concussions (prevalence ratio [PR] = 4.2; 95% CI, 1.0-16.3) or 1 to 2 concussions (PR = 2.8; 95% CI, 1.3-6.0). No statistically significant association between concussion and depression was observed with athletes reporting 1 to 2 concussions compared to 0 concussions.
A 2015 prospective longitudinal cohort study examined postinjury depressive symptoms in 3 groups of Division 1 male and female college student athletes (N = 21; ages 18-22).5 Physician-diagnosed concussed (N = 7) and injured but nonconcussed (N = 7) athletes completed the Center for Epidemiological Studies Depression Scale (CES-D) at baseline and at 1 week, 1 month, and 3 months postinjury. Sport-matched healthy athletes (N = 7) completed it only at baseline. A CES-D score of ≥ 16 (range, 0-60) indicated a risk for clinical depression. Participants with a history of depression or other injury resulting in ≥ 1 day of time lost within the past 3 months were excluded.
Continue to: While both groups...
While both groups showed a significant increase from baseline CES-D scores, there were no significant differences in depressive symptoms between concussed (mean CES-D score ± standard deviation [SD]: baseline, 6.7 ± 3.9; 1 week, 11 ± 5.3; 1 month, 8.3 ± 5; 3 months, 6.4 ± 5.4) and injured but nonconcussed participants (mean CES-D score ± SD: baseline, 5.7 ± 2.8; 1 week, 9.1 ± 4; 1 month, 8.9 ± 4.6; 3 months, 6.9 ± 2.8) at any of the postinjury time points.
Findings among semipro and pro athletes appear to vary by sport
A 2016 prospective cohort study assessed the impact of concussive events on incidence of depression in active semiprofessional and professional football players who had previously sustained ≥ 1 concussions.6 Participants (N = 27) answered an anonymous online survey that included the revised version of the CES-D (CESD-R) to determine level of depression (a score of ≥ 16 defined clinical depression). Players with a CESD-R score ≥ 16 (N = 16) sustained a significantly greater average number of concussions compared to those who scored < 16 (N = 11; 3.8 vs. 1.6, P = .0004). Players who sustained ≥ 3 concussions scored significantly higher on the CESD-R than players with ≤ 2 concussions (average score, 24 vs 15.6; P = .03).
A 2017 case-control study examined the long-term health outcomes of retired Scottish male rugby players (N = 52; mean age, 54 years) with a history of mild concussion compared to males of similar age with no previous history of concussion (N = 29; mean age, 55).7 The Hospital Anxiety and Depression Scale (HADS) was used to assess depression on a 21-point scale (normal = 0-7; borderline, 8-10; abnormal, 11-21). There was no significant difference observed in mean HADS scores between the rugby players and controls, respectively (2.8 ± 2.1 vs 2.6 ± 2 .8; P = .941).
A 2013 case-control study of 30 retired NFL players with 29 controls matched for age, estimated IQ, and education examined the relationship between a remote history of concussion and current symptoms of depression.8 Concussion history was self-reported by the retired players. Controls with a history of concussion were excluded from the study. The Beck Depression Inventory-II (BDI-II) was used to measure depression symptoms, with a score of 1 to 9 designating minimal depression and ≥ 10 mild-to-moderate depression. Retired players scored significantly higher on the BDI-II compared to the controls (8.8 vs 2.8; P = .001).
Editor’s takeaway
Concussions include cognitive compromise. An astute clinician’s concern for depression as a sequela makes sense. This evidence contributes to that conjecture. However, the authors of this Clinical Inquiry correctly outline the limitations, inconsistencies, and biases of the evidence. The exact relationship—degree and context—between concussion and depression remains vague.
1. Yang MN, Clements-Nolle K, Parrish B, et al. Adolescent concussion and mental health outcomes: a population-based study. Am J Health Behav. 2019;43:258-265.
2. Chrisman SPD, Richardson LP. Prevalence of diagnosed depression in adolescents with history of concussion. J Adolesc Health. 2014;54:582-586.
3. Vargas G, Rabinowitz A, Meyer J, et al. Predictors and prevalence of postconcussion depression symptoms in collegiate athletes. J Athl Train. 2015;50:250-255.
4. Kerr ZY, Thomas LC, Simon JE, et al. Association between history of multiple concussions and health outcomes among former college football players. Am J Sports Med. 2018;46:1733-1741.
5. Roiger T, Weidauer L, Kern B. A longitudinal pilot study of depressive symptoms in concussed and injured/nonconcussed National Collegiate Athletic Association Division I student-athletes. J Athl Train. 2015;50:256-261.
6. Pryor J, Larson A, DeBeliso M. The prevalence of depression and concussions in a sample of active North American semi-professional and professional football players. J Lifestyle Med. 2016;6:7-15.
7. McMillan TM, McSkimming P, Wainman-Lefley J, et al. Long-term health outcomes after exposure to repeated concussion in elite level: rugby union players. J Neurol Neurosurg Psychiatry. 2017;88:505-511.
8. Didehbani N, Munro Cullum C, Mansinghani S, et al. Depressive symptoms and concussions in aging retired NFL players. Arch Clin Neuropsychol. 2013;28:418-424.
1. Yang MN, Clements-Nolle K, Parrish B, et al. Adolescent concussion and mental health outcomes: a population-based study. Am J Health Behav. 2019;43:258-265.
2. Chrisman SPD, Richardson LP. Prevalence of diagnosed depression in adolescents with history of concussion. J Adolesc Health. 2014;54:582-586.
3. Vargas G, Rabinowitz A, Meyer J, et al. Predictors and prevalence of postconcussion depression symptoms in collegiate athletes. J Athl Train. 2015;50:250-255.
4. Kerr ZY, Thomas LC, Simon JE, et al. Association between history of multiple concussions and health outcomes among former college football players. Am J Sports Med. 2018;46:1733-1741.
5. Roiger T, Weidauer L, Kern B. A longitudinal pilot study of depressive symptoms in concussed and injured/nonconcussed National Collegiate Athletic Association Division I student-athletes. J Athl Train. 2015;50:256-261.
6. Pryor J, Larson A, DeBeliso M. The prevalence of depression and concussions in a sample of active North American semi-professional and professional football players. J Lifestyle Med. 2016;6:7-15.
7. McMillan TM, McSkimming P, Wainman-Lefley J, et al. Long-term health outcomes after exposure to repeated concussion in elite level: rugby union players. J Neurol Neurosurg Psychiatry. 2017;88:505-511.
8. Didehbani N, Munro Cullum C, Mansinghani S, et al. Depressive symptoms and concussions in aging retired NFL players. Arch Clin Neuropsychol. 2013;28:418-424.
EVIDENCE-BASED ANSWER
Yes, in some populations. Youth and adolescents with self-reported history of concussion had increased risk of depressive disorders (strength of recommendation [SOR]: B, based on a prospective cohort study and a retrospective cohort study). Evidence was inconsistent for college athletes. Athletes with ≥ 3 concussions exhibited more depressive disorders, but no association was observed for those with 1 or 2 concussions compared to nonconcussion injuries (SOR: B, based on a cross-sectional study, a small prospective cohort study, and a case-control study).
In semiprofessional and professional athletes, evidence was variable and may be sport related. Retired rugby players with a history of concussion showed no increase in depression compared to controls with no concussion history (SOR: B, based on a case-control study). Retired football players with previous concussions displayed increased incidence of depression, especially after ≥ 3 concussions (SOR: B, based on a prospective cohort study and a small case-control study).
There is a significant risk of bias in these studies because of their reliance on self-reported concussions, differing definitions of depression, and possible unmeasured confounders in the study designs, making a causative relationship between concussion and depression unclear.
Tough pain relief choices in the COVID-19 pandemic
More people with fever and body aches are turning to NSAIDs to ease symptoms, but the drugs have come under new scrutiny as investigators work to determine whether they are a safe way to relieve the pain of COVID-19 vaccination or symptoms of the disease.
Early on in the pandemic, French health officials warned that NSAIDs, such as ibuprofen, could worsen coronavirus disease, and they recommended switching to acetaminophen instead.
The National Health Service in the United Kingdom followed with a similar recommendation for acetaminophen.
But the European Medicines Agency took a different approach, reporting “no scientific evidence” that NSAIDs could worsen COVID-19. The U.S. Food and Drug Administration also opted not to take a stance.
The debate prompted discussion on social media, with various reactions from around the world. It also inspired Craig Wilen, MD, PhD, from Yale University, New Haven, Conn., and associates to examine the effect of NSAIDs on COVID-19 infection and immune response. Their findings were published online Jan.20 in the Journal of Virology.
“It really bothered me that non–evidence-based decisions were driving the conversation,” Dr. Wilen said. “Millions of people are taking NSAIDs every day and clinical decisions about their care shouldn’t be made on a hypothesis.”
One theory is that NSAIDs alter susceptibility to infection by modifying ACE2. The drugs might also change the cell entry receptor for SARS-CoV-2, alter virus replication, or even modify the immune response.
British researchers, also questioning the safety of NSAIDs in patients with COVID-19, delved into National Health Service records to study two large groups of patients, some of whom were taking the pain relievers.
“We were watching the controversy and the lack of evidence and wanted to contribute,” lead investigator Angel Wong, PhD, from the London School of Hygiene and Tropical Medicine, said in an interview.
And with nearly 11 million NSAID prescriptions dispensed in primary care in England alone in the past 12 months, the inconsistency was concerning.
The team compared COVID-19–related deaths in two groups: one group of more than 700,000 people taking NSAIDs, including patients with rheumatoid arthritis and osteoarthritis; and another of almost 3.5 million people not on the medication.
NSAIDs work by inhibiting cyclooxygenase-1 and COX-2 enzymes in the body, which are crucial for the generation of prostaglandins. These lipid molecules play a role in inflammation and are blocked by NSAIDs.
The investigators found no evidence of a harmful effect of NSAIDs on COVID-19-related deaths; their results were published online Jan. 21 in the Annals of the Rheumatic Diseases.
The results, they pointed out, are in line with a Danish study that also showed no evidence of a higher risk for severe COVID-19 outcomes with NSAID use.
“It’s reassuring,” Dr. Wong said, “that patients can safely continue treatment.”
More new evidence
Dr. Wilen’s team found that SARS-CoV-2 infection stimulated COX-2 expression in human and mice cells. However, suppression of COX-2 by two commonly used NSAIDs, ibuprofen and meloxicam, had no effect on ACE2 expression, viral entry, or viral replication.
In their mouse model of SARS-CoV-2 infection, the investigators saw that NSAIDs impaired the production of proinflammatory cytokines and neutralizing antibodies. The findings suggest that NSAIDs influence COVID-19 outcomes by dampening the inflammatory response and production of protective antibodies, rather than modifying susceptibility to infection or viral replication.
Understanding the effect of NSAIDs on cytokine production is critical, Dr. Wilen pointed out, because they might be protective early in COVID-19 but pathologic at later stages.
Timing is crucial in the case of other immunomodulatory drugs. For example, dexamethasone lowers mortality in COVID-19 patients on respiratory support but is potentially harmful for those with milder disease.
There still is a lot to learn, Dr. Wilen acknowledged. “We may be seeing something similar going on with NSAIDs, where the timing of treatment is important.”
A version of this article first appeared on Medscape.com.
More people with fever and body aches are turning to NSAIDs to ease symptoms, but the drugs have come under new scrutiny as investigators work to determine whether they are a safe way to relieve the pain of COVID-19 vaccination or symptoms of the disease.
Early on in the pandemic, French health officials warned that NSAIDs, such as ibuprofen, could worsen coronavirus disease, and they recommended switching to acetaminophen instead.
The National Health Service in the United Kingdom followed with a similar recommendation for acetaminophen.
But the European Medicines Agency took a different approach, reporting “no scientific evidence” that NSAIDs could worsen COVID-19. The U.S. Food and Drug Administration also opted not to take a stance.
The debate prompted discussion on social media, with various reactions from around the world. It also inspired Craig Wilen, MD, PhD, from Yale University, New Haven, Conn., and associates to examine the effect of NSAIDs on COVID-19 infection and immune response. Their findings were published online Jan.20 in the Journal of Virology.
“It really bothered me that non–evidence-based decisions were driving the conversation,” Dr. Wilen said. “Millions of people are taking NSAIDs every day and clinical decisions about their care shouldn’t be made on a hypothesis.”
One theory is that NSAIDs alter susceptibility to infection by modifying ACE2. The drugs might also change the cell entry receptor for SARS-CoV-2, alter virus replication, or even modify the immune response.
British researchers, also questioning the safety of NSAIDs in patients with COVID-19, delved into National Health Service records to study two large groups of patients, some of whom were taking the pain relievers.
“We were watching the controversy and the lack of evidence and wanted to contribute,” lead investigator Angel Wong, PhD, from the London School of Hygiene and Tropical Medicine, said in an interview.
And with nearly 11 million NSAID prescriptions dispensed in primary care in England alone in the past 12 months, the inconsistency was concerning.
The team compared COVID-19–related deaths in two groups: one group of more than 700,000 people taking NSAIDs, including patients with rheumatoid arthritis and osteoarthritis; and another of almost 3.5 million people not on the medication.
NSAIDs work by inhibiting cyclooxygenase-1 and COX-2 enzymes in the body, which are crucial for the generation of prostaglandins. These lipid molecules play a role in inflammation and are blocked by NSAIDs.
The investigators found no evidence of a harmful effect of NSAIDs on COVID-19-related deaths; their results were published online Jan. 21 in the Annals of the Rheumatic Diseases.
The results, they pointed out, are in line with a Danish study that also showed no evidence of a higher risk for severe COVID-19 outcomes with NSAID use.
“It’s reassuring,” Dr. Wong said, “that patients can safely continue treatment.”
More new evidence
Dr. Wilen’s team found that SARS-CoV-2 infection stimulated COX-2 expression in human and mice cells. However, suppression of COX-2 by two commonly used NSAIDs, ibuprofen and meloxicam, had no effect on ACE2 expression, viral entry, or viral replication.
In their mouse model of SARS-CoV-2 infection, the investigators saw that NSAIDs impaired the production of proinflammatory cytokines and neutralizing antibodies. The findings suggest that NSAIDs influence COVID-19 outcomes by dampening the inflammatory response and production of protective antibodies, rather than modifying susceptibility to infection or viral replication.
Understanding the effect of NSAIDs on cytokine production is critical, Dr. Wilen pointed out, because they might be protective early in COVID-19 but pathologic at later stages.
Timing is crucial in the case of other immunomodulatory drugs. For example, dexamethasone lowers mortality in COVID-19 patients on respiratory support but is potentially harmful for those with milder disease.
There still is a lot to learn, Dr. Wilen acknowledged. “We may be seeing something similar going on with NSAIDs, where the timing of treatment is important.”
A version of this article first appeared on Medscape.com.
More people with fever and body aches are turning to NSAIDs to ease symptoms, but the drugs have come under new scrutiny as investigators work to determine whether they are a safe way to relieve the pain of COVID-19 vaccination or symptoms of the disease.
Early on in the pandemic, French health officials warned that NSAIDs, such as ibuprofen, could worsen coronavirus disease, and they recommended switching to acetaminophen instead.
The National Health Service in the United Kingdom followed with a similar recommendation for acetaminophen.
But the European Medicines Agency took a different approach, reporting “no scientific evidence” that NSAIDs could worsen COVID-19. The U.S. Food and Drug Administration also opted not to take a stance.
The debate prompted discussion on social media, with various reactions from around the world. It also inspired Craig Wilen, MD, PhD, from Yale University, New Haven, Conn., and associates to examine the effect of NSAIDs on COVID-19 infection and immune response. Their findings were published online Jan.20 in the Journal of Virology.
“It really bothered me that non–evidence-based decisions were driving the conversation,” Dr. Wilen said. “Millions of people are taking NSAIDs every day and clinical decisions about their care shouldn’t be made on a hypothesis.”
One theory is that NSAIDs alter susceptibility to infection by modifying ACE2. The drugs might also change the cell entry receptor for SARS-CoV-2, alter virus replication, or even modify the immune response.
British researchers, also questioning the safety of NSAIDs in patients with COVID-19, delved into National Health Service records to study two large groups of patients, some of whom were taking the pain relievers.
“We were watching the controversy and the lack of evidence and wanted to contribute,” lead investigator Angel Wong, PhD, from the London School of Hygiene and Tropical Medicine, said in an interview.
And with nearly 11 million NSAID prescriptions dispensed in primary care in England alone in the past 12 months, the inconsistency was concerning.
The team compared COVID-19–related deaths in two groups: one group of more than 700,000 people taking NSAIDs, including patients with rheumatoid arthritis and osteoarthritis; and another of almost 3.5 million people not on the medication.
NSAIDs work by inhibiting cyclooxygenase-1 and COX-2 enzymes in the body, which are crucial for the generation of prostaglandins. These lipid molecules play a role in inflammation and are blocked by NSAIDs.
The investigators found no evidence of a harmful effect of NSAIDs on COVID-19-related deaths; their results were published online Jan. 21 in the Annals of the Rheumatic Diseases.
The results, they pointed out, are in line with a Danish study that also showed no evidence of a higher risk for severe COVID-19 outcomes with NSAID use.
“It’s reassuring,” Dr. Wong said, “that patients can safely continue treatment.”
More new evidence
Dr. Wilen’s team found that SARS-CoV-2 infection stimulated COX-2 expression in human and mice cells. However, suppression of COX-2 by two commonly used NSAIDs, ibuprofen and meloxicam, had no effect on ACE2 expression, viral entry, or viral replication.
In their mouse model of SARS-CoV-2 infection, the investigators saw that NSAIDs impaired the production of proinflammatory cytokines and neutralizing antibodies. The findings suggest that NSAIDs influence COVID-19 outcomes by dampening the inflammatory response and production of protective antibodies, rather than modifying susceptibility to infection or viral replication.
Understanding the effect of NSAIDs on cytokine production is critical, Dr. Wilen pointed out, because they might be protective early in COVID-19 but pathologic at later stages.
Timing is crucial in the case of other immunomodulatory drugs. For example, dexamethasone lowers mortality in COVID-19 patients on respiratory support but is potentially harmful for those with milder disease.
There still is a lot to learn, Dr. Wilen acknowledged. “We may be seeing something similar going on with NSAIDs, where the timing of treatment is important.”
A version of this article first appeared on Medscape.com.
Stem cell transplant shows long-term benefit in MS
The benefits of autologous hematopoietic stem cell transplant (AHSCT) for patients with multiple sclerosis (MS) persist for more than 10 years in the majority of patents, new data show. The study reports on 210 Italian patients who underwent AHSCT between 2007 and 2019. Among the entire study cohort, 79.5% of patients had not experienced worsening of disability at 5 years, and 65.5% had not experienced it at 10 years.
Patients with relapsing remitting MS had better results, with 85.5% experiencing no worsening of disability at 5 years, and 71.3% at 10 years. Among patients with progressive MS, 71.0% showed no worsening of disability at 5 years, and 57.2% at 10 years.
“This is the longest follow-up of AHSCT in MS patients so far to be reported,” said study author Matilde Inglese, MD, University of Genoa (Italy). “We have shown AHSCT to be highly effective to prevent long-term disability worsening in most treated patients.”
The study was published online Jan. 20 in Neurology.
“We suggest that AHSCT should be considered as a treatment strategy for MS not responding to conventional therapy,” the authors concluded.
The study had no control group, so a direct comparison is not possible. Nevertheless, Dr. Inglese said she believed these results are better than those that would be achieved with disease-modifying drug therapy for similar patients.
“The best patient candidates for this procedure are those with highly active multiple sclerosis who are not responsive to high-efficacy drugs, such as alemtuzumab or ocrelizumab,” Dr. Inglese commented. “Younger patients with an aggressive form of relapsing remitting MS tend to do the best, although patients with progressive forms of MS who still have active lesions on MRI also benefit.”
Renewing the immune system
The transplant procedure involves giving high-dose cyclophosphamide to stimulate mobilization of bone marrow stem cells, which are collected from peripheral blood. Patients then undergo intense immunosuppression with a cocktail of drugs to remove the autoreactive T cells, and the stem cells, which are not autoreactive, are reinfused.
“We are effectively renewing the immune system,” Dr. Inglese said. “While it is not correct to call it a cure, as we are not eliminating the etiology of the disease, it is the closest to complete suppression of the disease that we can get.”
Other results from the study show that among patients with relapsing remitting MS, rates of relapse-free survival were 78.1% at 5 years and 63.5% at 10 years.
Better results were achieved for patients who received the BEAM+ATG conditioning regimen for immunosuppression. That regimen includes carmustine, cytosine-arabinoside, etoposide, and melphalan, followed by rabbit antithymocyte globulin. Among patients with relapsing remitting disease who were treated with this protocol, rates of relapse-free survival were 86.4% at 5 years and 77.0% at 10 years.
For patients with relapsing remitting MS, the probability of achieving NEDA-3 status (no evidence of disease activity, including the absence of clinical relapses, disability worsening, and MRI inflammatory activity) was 62.2% at 5 years and 40.5% at 10 years.
Among those patients with relapsing remitting MS who received the BEAM+ATG conditioning protocol, NEDA-3 status was achieved in 67.7% at 5 years and in 54.9% at 10 years.
Three deaths occurred within 100 days following AHSCT (1.4% of the entire study population). One patient developed pulmonary thromboembolism, received fibrinolytic treatment, and died 48 hours later after intracranial hemorrhage. The second patient experienced engraftment failure and died 24 days after transplant because of an opportunistic infection. The third patient died 1 month after transplant from Wernicke-like encephalopathy. All the patients who died received the BEAM+ATG conditioning regimen. No transplant-related deaths occurred in patients who underwent transplant after 2007.
Dr. Inglese noted that the mortality rate associated with AHSCT has been greatly reduced in recent years. “We are seeing a very low mortality rate – about 0.3% – thanks to improvements in the procedure and better patient selection. This seems acceptable, given that we are treating patients with very aggressive disease who have a high risk of becoming significantly disabled relatively early in life,” she commented.
However, it is vitally important that the procedure be conducted in a specialized center with a highly experienced multidisciplinary team, she stressed.
In the Neurology article, the authors concluded: “Although patients with RRMS [relapsing remitting MS] are those who benefit the most from transplant, AHSCT has been also shown to prevent disability worsening in a large proportion of patients with active progressive MS.
“The BEAM+ATG conditioning protocol, although associated with a higher transplant mortality rate, was associated with a more pronounced suppression of clinical relapses and MRI inflammatory activity, allowing complete disease control in a higher proportion of patients,” they wrote.
Potent and durable efficacy, with caveats
Commenting on these latest findings, Jeffrey A. Cohen, MD, of the Mellen Center for Multiple Sclerosis at the Cleveland Clinic, said: “AHSCT appears to have potent and durable efficacy in MS but is associated with significant risk and cost.”
The patients who are most likely to benefit are young and have experienced the onset of disease relatively recently. They are still ambulatory with highly active MS and have experienced recent clinical relapses and/or MRI lesion activity, and such activity continues despite disease-modifying therapy, Dr. Cohen noted. He added that “AHSCT is a reasonable option for such patients who have essentially failed the available disease-modifying therapy options.”
He pointed out that the key question is where AHSCT belongs in the overall MS algorithm relative to other high-efficacy therapies. “We need to know whether it should be used more broadly rather than as a last resort.”
To address that question, several randomized trials comparing AHSCT with high-efficacy disease-modifying therapy are in progress, including the National Institutes of Health–sponsored BEAT-MS trial in the United States (for which Dr. Cohen is the lead investigator) and four European trials – NET-MS (for which Dr. Inglese is the lead investigator), STAR-MS, RAM-MS, and COAST-MS.
The current study was partially funded and supported by the Italian Multiple Sclerosis Foundation. Dr. Inglese disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The benefits of autologous hematopoietic stem cell transplant (AHSCT) for patients with multiple sclerosis (MS) persist for more than 10 years in the majority of patents, new data show. The study reports on 210 Italian patients who underwent AHSCT between 2007 and 2019. Among the entire study cohort, 79.5% of patients had not experienced worsening of disability at 5 years, and 65.5% had not experienced it at 10 years.
Patients with relapsing remitting MS had better results, with 85.5% experiencing no worsening of disability at 5 years, and 71.3% at 10 years. Among patients with progressive MS, 71.0% showed no worsening of disability at 5 years, and 57.2% at 10 years.
“This is the longest follow-up of AHSCT in MS patients so far to be reported,” said study author Matilde Inglese, MD, University of Genoa (Italy). “We have shown AHSCT to be highly effective to prevent long-term disability worsening in most treated patients.”
The study was published online Jan. 20 in Neurology.
“We suggest that AHSCT should be considered as a treatment strategy for MS not responding to conventional therapy,” the authors concluded.
The study had no control group, so a direct comparison is not possible. Nevertheless, Dr. Inglese said she believed these results are better than those that would be achieved with disease-modifying drug therapy for similar patients.
“The best patient candidates for this procedure are those with highly active multiple sclerosis who are not responsive to high-efficacy drugs, such as alemtuzumab or ocrelizumab,” Dr. Inglese commented. “Younger patients with an aggressive form of relapsing remitting MS tend to do the best, although patients with progressive forms of MS who still have active lesions on MRI also benefit.”
Renewing the immune system
The transplant procedure involves giving high-dose cyclophosphamide to stimulate mobilization of bone marrow stem cells, which are collected from peripheral blood. Patients then undergo intense immunosuppression with a cocktail of drugs to remove the autoreactive T cells, and the stem cells, which are not autoreactive, are reinfused.
“We are effectively renewing the immune system,” Dr. Inglese said. “While it is not correct to call it a cure, as we are not eliminating the etiology of the disease, it is the closest to complete suppression of the disease that we can get.”
Other results from the study show that among patients with relapsing remitting MS, rates of relapse-free survival were 78.1% at 5 years and 63.5% at 10 years.
Better results were achieved for patients who received the BEAM+ATG conditioning regimen for immunosuppression. That regimen includes carmustine, cytosine-arabinoside, etoposide, and melphalan, followed by rabbit antithymocyte globulin. Among patients with relapsing remitting disease who were treated with this protocol, rates of relapse-free survival were 86.4% at 5 years and 77.0% at 10 years.
For patients with relapsing remitting MS, the probability of achieving NEDA-3 status (no evidence of disease activity, including the absence of clinical relapses, disability worsening, and MRI inflammatory activity) was 62.2% at 5 years and 40.5% at 10 years.
Among those patients with relapsing remitting MS who received the BEAM+ATG conditioning protocol, NEDA-3 status was achieved in 67.7% at 5 years and in 54.9% at 10 years.
Three deaths occurred within 100 days following AHSCT (1.4% of the entire study population). One patient developed pulmonary thromboembolism, received fibrinolytic treatment, and died 48 hours later after intracranial hemorrhage. The second patient experienced engraftment failure and died 24 days after transplant because of an opportunistic infection. The third patient died 1 month after transplant from Wernicke-like encephalopathy. All the patients who died received the BEAM+ATG conditioning regimen. No transplant-related deaths occurred in patients who underwent transplant after 2007.
Dr. Inglese noted that the mortality rate associated with AHSCT has been greatly reduced in recent years. “We are seeing a very low mortality rate – about 0.3% – thanks to improvements in the procedure and better patient selection. This seems acceptable, given that we are treating patients with very aggressive disease who have a high risk of becoming significantly disabled relatively early in life,” she commented.
However, it is vitally important that the procedure be conducted in a specialized center with a highly experienced multidisciplinary team, she stressed.
In the Neurology article, the authors concluded: “Although patients with RRMS [relapsing remitting MS] are those who benefit the most from transplant, AHSCT has been also shown to prevent disability worsening in a large proportion of patients with active progressive MS.
“The BEAM+ATG conditioning protocol, although associated with a higher transplant mortality rate, was associated with a more pronounced suppression of clinical relapses and MRI inflammatory activity, allowing complete disease control in a higher proportion of patients,” they wrote.
Potent and durable efficacy, with caveats
Commenting on these latest findings, Jeffrey A. Cohen, MD, of the Mellen Center for Multiple Sclerosis at the Cleveland Clinic, said: “AHSCT appears to have potent and durable efficacy in MS but is associated with significant risk and cost.”
The patients who are most likely to benefit are young and have experienced the onset of disease relatively recently. They are still ambulatory with highly active MS and have experienced recent clinical relapses and/or MRI lesion activity, and such activity continues despite disease-modifying therapy, Dr. Cohen noted. He added that “AHSCT is a reasonable option for such patients who have essentially failed the available disease-modifying therapy options.”
He pointed out that the key question is where AHSCT belongs in the overall MS algorithm relative to other high-efficacy therapies. “We need to know whether it should be used more broadly rather than as a last resort.”
To address that question, several randomized trials comparing AHSCT with high-efficacy disease-modifying therapy are in progress, including the National Institutes of Health–sponsored BEAT-MS trial in the United States (for which Dr. Cohen is the lead investigator) and four European trials – NET-MS (for which Dr. Inglese is the lead investigator), STAR-MS, RAM-MS, and COAST-MS.
The current study was partially funded and supported by the Italian Multiple Sclerosis Foundation. Dr. Inglese disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The benefits of autologous hematopoietic stem cell transplant (AHSCT) for patients with multiple sclerosis (MS) persist for more than 10 years in the majority of patents, new data show. The study reports on 210 Italian patients who underwent AHSCT between 2007 and 2019. Among the entire study cohort, 79.5% of patients had not experienced worsening of disability at 5 years, and 65.5% had not experienced it at 10 years.
Patients with relapsing remitting MS had better results, with 85.5% experiencing no worsening of disability at 5 years, and 71.3% at 10 years. Among patients with progressive MS, 71.0% showed no worsening of disability at 5 years, and 57.2% at 10 years.
“This is the longest follow-up of AHSCT in MS patients so far to be reported,” said study author Matilde Inglese, MD, University of Genoa (Italy). “We have shown AHSCT to be highly effective to prevent long-term disability worsening in most treated patients.”
The study was published online Jan. 20 in Neurology.
“We suggest that AHSCT should be considered as a treatment strategy for MS not responding to conventional therapy,” the authors concluded.
The study had no control group, so a direct comparison is not possible. Nevertheless, Dr. Inglese said she believed these results are better than those that would be achieved with disease-modifying drug therapy for similar patients.
“The best patient candidates for this procedure are those with highly active multiple sclerosis who are not responsive to high-efficacy drugs, such as alemtuzumab or ocrelizumab,” Dr. Inglese commented. “Younger patients with an aggressive form of relapsing remitting MS tend to do the best, although patients with progressive forms of MS who still have active lesions on MRI also benefit.”
Renewing the immune system
The transplant procedure involves giving high-dose cyclophosphamide to stimulate mobilization of bone marrow stem cells, which are collected from peripheral blood. Patients then undergo intense immunosuppression with a cocktail of drugs to remove the autoreactive T cells, and the stem cells, which are not autoreactive, are reinfused.
“We are effectively renewing the immune system,” Dr. Inglese said. “While it is not correct to call it a cure, as we are not eliminating the etiology of the disease, it is the closest to complete suppression of the disease that we can get.”
Other results from the study show that among patients with relapsing remitting MS, rates of relapse-free survival were 78.1% at 5 years and 63.5% at 10 years.
Better results were achieved for patients who received the BEAM+ATG conditioning regimen for immunosuppression. That regimen includes carmustine, cytosine-arabinoside, etoposide, and melphalan, followed by rabbit antithymocyte globulin. Among patients with relapsing remitting disease who were treated with this protocol, rates of relapse-free survival were 86.4% at 5 years and 77.0% at 10 years.
For patients with relapsing remitting MS, the probability of achieving NEDA-3 status (no evidence of disease activity, including the absence of clinical relapses, disability worsening, and MRI inflammatory activity) was 62.2% at 5 years and 40.5% at 10 years.
Among those patients with relapsing remitting MS who received the BEAM+ATG conditioning protocol, NEDA-3 status was achieved in 67.7% at 5 years and in 54.9% at 10 years.
Three deaths occurred within 100 days following AHSCT (1.4% of the entire study population). One patient developed pulmonary thromboembolism, received fibrinolytic treatment, and died 48 hours later after intracranial hemorrhage. The second patient experienced engraftment failure and died 24 days after transplant because of an opportunistic infection. The third patient died 1 month after transplant from Wernicke-like encephalopathy. All the patients who died received the BEAM+ATG conditioning regimen. No transplant-related deaths occurred in patients who underwent transplant after 2007.
Dr. Inglese noted that the mortality rate associated with AHSCT has been greatly reduced in recent years. “We are seeing a very low mortality rate – about 0.3% – thanks to improvements in the procedure and better patient selection. This seems acceptable, given that we are treating patients with very aggressive disease who have a high risk of becoming significantly disabled relatively early in life,” she commented.
However, it is vitally important that the procedure be conducted in a specialized center with a highly experienced multidisciplinary team, she stressed.
In the Neurology article, the authors concluded: “Although patients with RRMS [relapsing remitting MS] are those who benefit the most from transplant, AHSCT has been also shown to prevent disability worsening in a large proportion of patients with active progressive MS.
“The BEAM+ATG conditioning protocol, although associated with a higher transplant mortality rate, was associated with a more pronounced suppression of clinical relapses and MRI inflammatory activity, allowing complete disease control in a higher proportion of patients,” they wrote.
Potent and durable efficacy, with caveats
Commenting on these latest findings, Jeffrey A. Cohen, MD, of the Mellen Center for Multiple Sclerosis at the Cleveland Clinic, said: “AHSCT appears to have potent and durable efficacy in MS but is associated with significant risk and cost.”
The patients who are most likely to benefit are young and have experienced the onset of disease relatively recently. They are still ambulatory with highly active MS and have experienced recent clinical relapses and/or MRI lesion activity, and such activity continues despite disease-modifying therapy, Dr. Cohen noted. He added that “AHSCT is a reasonable option for such patients who have essentially failed the available disease-modifying therapy options.”
He pointed out that the key question is where AHSCT belongs in the overall MS algorithm relative to other high-efficacy therapies. “We need to know whether it should be used more broadly rather than as a last resort.”
To address that question, several randomized trials comparing AHSCT with high-efficacy disease-modifying therapy are in progress, including the National Institutes of Health–sponsored BEAT-MS trial in the United States (for which Dr. Cohen is the lead investigator) and four European trials – NET-MS (for which Dr. Inglese is the lead investigator), STAR-MS, RAM-MS, and COAST-MS.
The current study was partially funded and supported by the Italian Multiple Sclerosis Foundation. Dr. Inglese disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NEUROLOGY
Blood biomarker may predict Alzheimer’s disease progression
new research suggests.
In a study of more than 1,000 participants, changes over time in levels of p-tau181 were associated with prospective neurodegeneration and cognitive decline characteristic of Alzheimer’s disease. These results have implications for investigative trials as well as clinical practice, the investigators noted.
Like p-tau181, neurofilament light chain (NfL) is associated with imaging markers of degeneration and cognitive decline; in contrast to the findings related to p-tau181, however, the associations between NfL and these outcomes are not specific to Alzheimer’s disease. Using both biomarkers could improve prediction of outcomes and patient monitoring, according to the researchers.
“These findings demonstrate that p-tau181 and NfL in blood have individual and complementary potential roles in the diagnosis and the monitoring of neurodegenerative disease,” said coinvestigator Michael Schöll, PhD, senior lecturer in psychiatry and neurochemistry at the University of Gothenburg (Sweden).
“With the reservation that we did not assess domain-specific cognitive impairment, p-tau181 was also more strongly associated with cognitive decline than was NfL,” Dr. Schöll added.
The findings were published online Jan. 11 in JAMA Neurology.
Biomarker-tracked neurodegeneration
Monitoring a patient’s neurodegenerative changes is important for tracking Alzheimer’s disease progression. Although clinicians can detect amyloid-beta and tau pathology using PET and cerebrospinal fluid (CSF) biomarkers, the widespread use of the latter has been hampered by cost and limited availability of necessary equipment. The use of blood-based biomarkers is not limited in these ways, and so they could aid in diagnosis and patient monitoring.
Previous studies have suggested that p-tau181 is a marker of Alzheimer’s disease status.
In the current study, investigators examined whether baseline and longitudinal levels of p-tau181 in plasma were associated with progressive neurodegeneration related to the disease. They analyzed data from the Alzheimer’s Disease Neuroimaging Initiative, a multicenter study designed to identify biomarkers for the detection and tracking of Alzheimer’s disease.
The researchers selected data for cognitively unimpaired and cognitively impaired participants who participated in the initiative between Feb. 1, 2007, and June 6, 2016. Participants were eligible for inclusion if plasma p-tau181 and NfL data were available for them and if they had undergone at least one 18fluorodeoxyglucose (FDG)–PET scan or structural T1 MRI at the same study visit. Most had also undergone imaging with 18florbetapir, which detects amyloid-beta.
A single-molecule array was used to analyze concentrations of p-tau181 and NfL in participants’ blood samples. Outliers for p-tau181 and NfL concentrations were excluded from further analysis. Using participants’ FDG-PET scans, the investigators measured glucose hypometabolism characteristic of Alzheimer’s disease. They used T1-weighted MRI scans to measure gray-matter volume.
Cognitively unimpaired participants responded to the Preclinical Alzheimer Cognitive Composite, a measure designed to detect early cognitive changes in cognitively normal patients with Alzheimer’s disease pathology. Cognitively impaired participants underwent the Alzheimer Disease Assessment Scale–Cognitive Subscale with 13 tasks to assess the severity of cognitive impairment.
The researchers included 1,113 participants (54% men; 89% non-Hispanic Whites; mean age, 74 years) in their analysis. In all, 378 participants were cognitively unimpaired, and 735 were cognitively impaired. Of the latter group, 73% had mild cognitive impairment, and 27% had Alzheimer’s disease dementia.
Atrophy predictor
Results showed that higher plasma p-tau181 levels at baseline were associated with more rapid progression of hypometabolism and atrophy in areas vulnerable to Alzheimer’s disease among cognitively impaired participants (FDG-PET standardized uptake value ratio change, r = –0.28; P < .001; gray-matter volume change, r = –0.28; P < .001).
The association with atrophy progression in cognitively impaired participants was stronger for p-tau181 than for NfL.
Plasma p-tau181 levels at baseline also predicted atrophy in temporoparietal regions vulnerable to Alzheimer’s disease among cognitively unimpaired participants (r = –0.11; P = .03). NfL, however, was associated with progressive atrophy in frontal regions among cognitively unimpaired participants.
At baseline, plasma p-tau181 levels were associated with prospective cognitive decline in both the cognitively unimpaired group (r = −0.12; P = .04) and the cognitively impaired group (r = 0.35; P < .001). However, plasma NfL was linked to cognitive decline only among those who were cognitively impaired (r = 0.26; P < .001).
Additional analyses showed that p-tau181, unlike NfL, was associated with hypometabolism and atrophy only in participants with amyloid-beta, regardless of cognitive status.
Between 25% and 45% of the association between baseline p-tau181 level and cognitive decline was mediated by baseline imaging markers of neurodegeneration. This finding suggests that another factor, such as regional tau pathology, might have an independent and direct effect on cognition, Dr. Schöll noted.
Furthermore, changes over time in p-tau181 levels were associated with cognitive decline in the cognitively unimpaired (r = –0.24; P < .001) and cognitively impaired (r = 0.34; P < .001) participants. Longitudinal changes in this biomarker also were associated with a prospective decrease in glucose metabolism in cognitively unimpaired (r = –0.05; P = .48) and cognitively impaired (r = –0.27; P < .001) participants, but the association was only significant in the latter group.
Changes over time in p-tau181 levels were linked to prospective decreases in gray-matter volume in brain regions highly characteristic of Alzheimer’s disease in those who were cognitively unimpaired (r = –0.19; P < .001) and those who were cognitively impaired (r = –0.31, P < .001). However, these associations were obtained only in patients with amyloid-beta.
Dr. Schöll noted that blood-based biomarkers that are sensitive to Alzheimer’s disease could greatly expand patients’ access to a diagnostic workup and could improve screening for clinical trials.
“While the final validation of the existence and the monitoring of potential changes of neuropathology in vivo is likely to be conducted using neuroimaging modalities such as PET, our results suggest that at least a part of these examinations could be replaced by regular blood tests,” Dr. Schöll said.
Lead author Alexis Moscoso, PhD, a postdoctoral researcher in psychiatry and neurochemistry at the University of Gothenburg, reported that the researchers will continue validating blood-based biomarkers, especially against established and well-validated neuroimaging methods. “We are also hoping to be able to compare existing and novel blood-based Alzheimer’s disease biomarkers head to head to establish the individual roles each of these play in the research and diagnosis of Alzheimer’s disease,” Dr. Moscoso said.
‘Outstanding study’
Commenting on the findings, David S. Knopman, MD, professor of neurology at Mayo Clinic, Rochester, Minn., said that this is “an outstanding study” because of its large number of participants and because the investigators are “world leaders in the technology of measuring plasma p-tau and NfL.”
Dr. Knopman, who was not involved with the research, noted that the study had no substantive weaknesses.
“The biggest advantages of a blood-based biomarker over CSF- and PET-based biomarkers of Alzheimer disease are the obvious ones of accessibility, cost, portability, and ease of repeatability,” he said.
“As CSF and PET exams are largely limited to major medical centers, valid blood-based biomarkers of Alzheimer disease that are reasonably specific make large-scale epidemiological studies that investigate dementia etiologies in rural or urban and diverse communities feasible,” he added.
Whereas p-tau181 appears to be specific for plaque and tangle disease, NfL is a nonspecific marker of neurodegeneration.
“Each has a role that could be valuable, depending on the circumstance,” said Dr. Knopman. “Plasma NfL has already proved itself useful in frontotemporal degeneration and chronic traumatic encephalopathy, for example.”
He noted that future studies should examine how closely p-tau181 and NfL align with more granular and direct measures of Alzheimer’s disease–related brain pathologies.
“There has got to be some loss of fidelity in detecting abnormality in going from brain tissue to blood, which might siphon off some time-related and severity-related information,” said Dr. Knopman.
“The exact role that plasma p-tau and NfL will play remains to be seen, because the diagnostic information that these biomarkers provide is contingent on the existence of interventions that require specific or nonspecific information about progressive neurodegeneration due to Alzheimer disease,” he added.
The study was funded by grants from the Spanish Instituto de Salud Carlos III, the Brightfocus Foundation, the Swedish Alzheimer Foundation, and the Swedish Brain Foundation. Dr. Schöll reported serving on a scientific advisory board for Servier on matters unrelated to this study. Dr. Moscoso and Dr. Knopman have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests.
In a study of more than 1,000 participants, changes over time in levels of p-tau181 were associated with prospective neurodegeneration and cognitive decline characteristic of Alzheimer’s disease. These results have implications for investigative trials as well as clinical practice, the investigators noted.
Like p-tau181, neurofilament light chain (NfL) is associated with imaging markers of degeneration and cognitive decline; in contrast to the findings related to p-tau181, however, the associations between NfL and these outcomes are not specific to Alzheimer’s disease. Using both biomarkers could improve prediction of outcomes and patient monitoring, according to the researchers.
“These findings demonstrate that p-tau181 and NfL in blood have individual and complementary potential roles in the diagnosis and the monitoring of neurodegenerative disease,” said coinvestigator Michael Schöll, PhD, senior lecturer in psychiatry and neurochemistry at the University of Gothenburg (Sweden).
“With the reservation that we did not assess domain-specific cognitive impairment, p-tau181 was also more strongly associated with cognitive decline than was NfL,” Dr. Schöll added.
The findings were published online Jan. 11 in JAMA Neurology.
Biomarker-tracked neurodegeneration
Monitoring a patient’s neurodegenerative changes is important for tracking Alzheimer’s disease progression. Although clinicians can detect amyloid-beta and tau pathology using PET and cerebrospinal fluid (CSF) biomarkers, the widespread use of the latter has been hampered by cost and limited availability of necessary equipment. The use of blood-based biomarkers is not limited in these ways, and so they could aid in diagnosis and patient monitoring.
Previous studies have suggested that p-tau181 is a marker of Alzheimer’s disease status.
In the current study, investigators examined whether baseline and longitudinal levels of p-tau181 in plasma were associated with progressive neurodegeneration related to the disease. They analyzed data from the Alzheimer’s Disease Neuroimaging Initiative, a multicenter study designed to identify biomarkers for the detection and tracking of Alzheimer’s disease.
The researchers selected data for cognitively unimpaired and cognitively impaired participants who participated in the initiative between Feb. 1, 2007, and June 6, 2016. Participants were eligible for inclusion if plasma p-tau181 and NfL data were available for them and if they had undergone at least one 18fluorodeoxyglucose (FDG)–PET scan or structural T1 MRI at the same study visit. Most had also undergone imaging with 18florbetapir, which detects amyloid-beta.
A single-molecule array was used to analyze concentrations of p-tau181 and NfL in participants’ blood samples. Outliers for p-tau181 and NfL concentrations were excluded from further analysis. Using participants’ FDG-PET scans, the investigators measured glucose hypometabolism characteristic of Alzheimer’s disease. They used T1-weighted MRI scans to measure gray-matter volume.
Cognitively unimpaired participants responded to the Preclinical Alzheimer Cognitive Composite, a measure designed to detect early cognitive changes in cognitively normal patients with Alzheimer’s disease pathology. Cognitively impaired participants underwent the Alzheimer Disease Assessment Scale–Cognitive Subscale with 13 tasks to assess the severity of cognitive impairment.
The researchers included 1,113 participants (54% men; 89% non-Hispanic Whites; mean age, 74 years) in their analysis. In all, 378 participants were cognitively unimpaired, and 735 were cognitively impaired. Of the latter group, 73% had mild cognitive impairment, and 27% had Alzheimer’s disease dementia.
Atrophy predictor
Results showed that higher plasma p-tau181 levels at baseline were associated with more rapid progression of hypometabolism and atrophy in areas vulnerable to Alzheimer’s disease among cognitively impaired participants (FDG-PET standardized uptake value ratio change, r = –0.28; P < .001; gray-matter volume change, r = –0.28; P < .001).
The association with atrophy progression in cognitively impaired participants was stronger for p-tau181 than for NfL.
Plasma p-tau181 levels at baseline also predicted atrophy in temporoparietal regions vulnerable to Alzheimer’s disease among cognitively unimpaired participants (r = –0.11; P = .03). NfL, however, was associated with progressive atrophy in frontal regions among cognitively unimpaired participants.
At baseline, plasma p-tau181 levels were associated with prospective cognitive decline in both the cognitively unimpaired group (r = −0.12; P = .04) and the cognitively impaired group (r = 0.35; P < .001). However, plasma NfL was linked to cognitive decline only among those who were cognitively impaired (r = 0.26; P < .001).
Additional analyses showed that p-tau181, unlike NfL, was associated with hypometabolism and atrophy only in participants with amyloid-beta, regardless of cognitive status.
Between 25% and 45% of the association between baseline p-tau181 level and cognitive decline was mediated by baseline imaging markers of neurodegeneration. This finding suggests that another factor, such as regional tau pathology, might have an independent and direct effect on cognition, Dr. Schöll noted.
Furthermore, changes over time in p-tau181 levels were associated with cognitive decline in the cognitively unimpaired (r = –0.24; P < .001) and cognitively impaired (r = 0.34; P < .001) participants. Longitudinal changes in this biomarker also were associated with a prospective decrease in glucose metabolism in cognitively unimpaired (r = –0.05; P = .48) and cognitively impaired (r = –0.27; P < .001) participants, but the association was only significant in the latter group.
Changes over time in p-tau181 levels were linked to prospective decreases in gray-matter volume in brain regions highly characteristic of Alzheimer’s disease in those who were cognitively unimpaired (r = –0.19; P < .001) and those who were cognitively impaired (r = –0.31, P < .001). However, these associations were obtained only in patients with amyloid-beta.
Dr. Schöll noted that blood-based biomarkers that are sensitive to Alzheimer’s disease could greatly expand patients’ access to a diagnostic workup and could improve screening for clinical trials.
“While the final validation of the existence and the monitoring of potential changes of neuropathology in vivo is likely to be conducted using neuroimaging modalities such as PET, our results suggest that at least a part of these examinations could be replaced by regular blood tests,” Dr. Schöll said.
Lead author Alexis Moscoso, PhD, a postdoctoral researcher in psychiatry and neurochemistry at the University of Gothenburg, reported that the researchers will continue validating blood-based biomarkers, especially against established and well-validated neuroimaging methods. “We are also hoping to be able to compare existing and novel blood-based Alzheimer’s disease biomarkers head to head to establish the individual roles each of these play in the research and diagnosis of Alzheimer’s disease,” Dr. Moscoso said.
‘Outstanding study’
Commenting on the findings, David S. Knopman, MD, professor of neurology at Mayo Clinic, Rochester, Minn., said that this is “an outstanding study” because of its large number of participants and because the investigators are “world leaders in the technology of measuring plasma p-tau and NfL.”
Dr. Knopman, who was not involved with the research, noted that the study had no substantive weaknesses.
“The biggest advantages of a blood-based biomarker over CSF- and PET-based biomarkers of Alzheimer disease are the obvious ones of accessibility, cost, portability, and ease of repeatability,” he said.
“As CSF and PET exams are largely limited to major medical centers, valid blood-based biomarkers of Alzheimer disease that are reasonably specific make large-scale epidemiological studies that investigate dementia etiologies in rural or urban and diverse communities feasible,” he added.
Whereas p-tau181 appears to be specific for plaque and tangle disease, NfL is a nonspecific marker of neurodegeneration.
“Each has a role that could be valuable, depending on the circumstance,” said Dr. Knopman. “Plasma NfL has already proved itself useful in frontotemporal degeneration and chronic traumatic encephalopathy, for example.”
He noted that future studies should examine how closely p-tau181 and NfL align with more granular and direct measures of Alzheimer’s disease–related brain pathologies.
“There has got to be some loss of fidelity in detecting abnormality in going from brain tissue to blood, which might siphon off some time-related and severity-related information,” said Dr. Knopman.
“The exact role that plasma p-tau and NfL will play remains to be seen, because the diagnostic information that these biomarkers provide is contingent on the existence of interventions that require specific or nonspecific information about progressive neurodegeneration due to Alzheimer disease,” he added.
The study was funded by grants from the Spanish Instituto de Salud Carlos III, the Brightfocus Foundation, the Swedish Alzheimer Foundation, and the Swedish Brain Foundation. Dr. Schöll reported serving on a scientific advisory board for Servier on matters unrelated to this study. Dr. Moscoso and Dr. Knopman have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests.
In a study of more than 1,000 participants, changes over time in levels of p-tau181 were associated with prospective neurodegeneration and cognitive decline characteristic of Alzheimer’s disease. These results have implications for investigative trials as well as clinical practice, the investigators noted.
Like p-tau181, neurofilament light chain (NfL) is associated with imaging markers of degeneration and cognitive decline; in contrast to the findings related to p-tau181, however, the associations between NfL and these outcomes are not specific to Alzheimer’s disease. Using both biomarkers could improve prediction of outcomes and patient monitoring, according to the researchers.
“These findings demonstrate that p-tau181 and NfL in blood have individual and complementary potential roles in the diagnosis and the monitoring of neurodegenerative disease,” said coinvestigator Michael Schöll, PhD, senior lecturer in psychiatry and neurochemistry at the University of Gothenburg (Sweden).
“With the reservation that we did not assess domain-specific cognitive impairment, p-tau181 was also more strongly associated with cognitive decline than was NfL,” Dr. Schöll added.
The findings were published online Jan. 11 in JAMA Neurology.
Biomarker-tracked neurodegeneration
Monitoring a patient’s neurodegenerative changes is important for tracking Alzheimer’s disease progression. Although clinicians can detect amyloid-beta and tau pathology using PET and cerebrospinal fluid (CSF) biomarkers, the widespread use of the latter has been hampered by cost and limited availability of necessary equipment. The use of blood-based biomarkers is not limited in these ways, and so they could aid in diagnosis and patient monitoring.
Previous studies have suggested that p-tau181 is a marker of Alzheimer’s disease status.
In the current study, investigators examined whether baseline and longitudinal levels of p-tau181 in plasma were associated with progressive neurodegeneration related to the disease. They analyzed data from the Alzheimer’s Disease Neuroimaging Initiative, a multicenter study designed to identify biomarkers for the detection and tracking of Alzheimer’s disease.
The researchers selected data for cognitively unimpaired and cognitively impaired participants who participated in the initiative between Feb. 1, 2007, and June 6, 2016. Participants were eligible for inclusion if plasma p-tau181 and NfL data were available for them and if they had undergone at least one 18fluorodeoxyglucose (FDG)–PET scan or structural T1 MRI at the same study visit. Most had also undergone imaging with 18florbetapir, which detects amyloid-beta.
A single-molecule array was used to analyze concentrations of p-tau181 and NfL in participants’ blood samples. Outliers for p-tau181 and NfL concentrations were excluded from further analysis. Using participants’ FDG-PET scans, the investigators measured glucose hypometabolism characteristic of Alzheimer’s disease. They used T1-weighted MRI scans to measure gray-matter volume.
Cognitively unimpaired participants responded to the Preclinical Alzheimer Cognitive Composite, a measure designed to detect early cognitive changes in cognitively normal patients with Alzheimer’s disease pathology. Cognitively impaired participants underwent the Alzheimer Disease Assessment Scale–Cognitive Subscale with 13 tasks to assess the severity of cognitive impairment.
The researchers included 1,113 participants (54% men; 89% non-Hispanic Whites; mean age, 74 years) in their analysis. In all, 378 participants were cognitively unimpaired, and 735 were cognitively impaired. Of the latter group, 73% had mild cognitive impairment, and 27% had Alzheimer’s disease dementia.
Atrophy predictor
Results showed that higher plasma p-tau181 levels at baseline were associated with more rapid progression of hypometabolism and atrophy in areas vulnerable to Alzheimer’s disease among cognitively impaired participants (FDG-PET standardized uptake value ratio change, r = –0.28; P < .001; gray-matter volume change, r = –0.28; P < .001).
The association with atrophy progression in cognitively impaired participants was stronger for p-tau181 than for NfL.
Plasma p-tau181 levels at baseline also predicted atrophy in temporoparietal regions vulnerable to Alzheimer’s disease among cognitively unimpaired participants (r = –0.11; P = .03). NfL, however, was associated with progressive atrophy in frontal regions among cognitively unimpaired participants.
At baseline, plasma p-tau181 levels were associated with prospective cognitive decline in both the cognitively unimpaired group (r = −0.12; P = .04) and the cognitively impaired group (r = 0.35; P < .001). However, plasma NfL was linked to cognitive decline only among those who were cognitively impaired (r = 0.26; P < .001).
Additional analyses showed that p-tau181, unlike NfL, was associated with hypometabolism and atrophy only in participants with amyloid-beta, regardless of cognitive status.
Between 25% and 45% of the association between baseline p-tau181 level and cognitive decline was mediated by baseline imaging markers of neurodegeneration. This finding suggests that another factor, such as regional tau pathology, might have an independent and direct effect on cognition, Dr. Schöll noted.
Furthermore, changes over time in p-tau181 levels were associated with cognitive decline in the cognitively unimpaired (r = –0.24; P < .001) and cognitively impaired (r = 0.34; P < .001) participants. Longitudinal changes in this biomarker also were associated with a prospective decrease in glucose metabolism in cognitively unimpaired (r = –0.05; P = .48) and cognitively impaired (r = –0.27; P < .001) participants, but the association was only significant in the latter group.
Changes over time in p-tau181 levels were linked to prospective decreases in gray-matter volume in brain regions highly characteristic of Alzheimer’s disease in those who were cognitively unimpaired (r = –0.19; P < .001) and those who were cognitively impaired (r = –0.31, P < .001). However, these associations were obtained only in patients with amyloid-beta.
Dr. Schöll noted that blood-based biomarkers that are sensitive to Alzheimer’s disease could greatly expand patients’ access to a diagnostic workup and could improve screening for clinical trials.
“While the final validation of the existence and the monitoring of potential changes of neuropathology in vivo is likely to be conducted using neuroimaging modalities such as PET, our results suggest that at least a part of these examinations could be replaced by regular blood tests,” Dr. Schöll said.
Lead author Alexis Moscoso, PhD, a postdoctoral researcher in psychiatry and neurochemistry at the University of Gothenburg, reported that the researchers will continue validating blood-based biomarkers, especially against established and well-validated neuroimaging methods. “We are also hoping to be able to compare existing and novel blood-based Alzheimer’s disease biomarkers head to head to establish the individual roles each of these play in the research and diagnosis of Alzheimer’s disease,” Dr. Moscoso said.
‘Outstanding study’
Commenting on the findings, David S. Knopman, MD, professor of neurology at Mayo Clinic, Rochester, Minn., said that this is “an outstanding study” because of its large number of participants and because the investigators are “world leaders in the technology of measuring plasma p-tau and NfL.”
Dr. Knopman, who was not involved with the research, noted that the study had no substantive weaknesses.
“The biggest advantages of a blood-based biomarker over CSF- and PET-based biomarkers of Alzheimer disease are the obvious ones of accessibility, cost, portability, and ease of repeatability,” he said.
“As CSF and PET exams are largely limited to major medical centers, valid blood-based biomarkers of Alzheimer disease that are reasonably specific make large-scale epidemiological studies that investigate dementia etiologies in rural or urban and diverse communities feasible,” he added.
Whereas p-tau181 appears to be specific for plaque and tangle disease, NfL is a nonspecific marker of neurodegeneration.
“Each has a role that could be valuable, depending on the circumstance,” said Dr. Knopman. “Plasma NfL has already proved itself useful in frontotemporal degeneration and chronic traumatic encephalopathy, for example.”
He noted that future studies should examine how closely p-tau181 and NfL align with more granular and direct measures of Alzheimer’s disease–related brain pathologies.
“There has got to be some loss of fidelity in detecting abnormality in going from brain tissue to blood, which might siphon off some time-related and severity-related information,” said Dr. Knopman.
“The exact role that plasma p-tau and NfL will play remains to be seen, because the diagnostic information that these biomarkers provide is contingent on the existence of interventions that require specific or nonspecific information about progressive neurodegeneration due to Alzheimer disease,” he added.
The study was funded by grants from the Spanish Instituto de Salud Carlos III, the Brightfocus Foundation, the Swedish Alzheimer Foundation, and the Swedish Brain Foundation. Dr. Schöll reported serving on a scientific advisory board for Servier on matters unrelated to this study. Dr. Moscoso and Dr. Knopman have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Generalized pruritic blisters and bullous lesions
A 62-year-old man presented to our skin clinic with multiple pruritic, tense, bullous lesions that manifested on his arms, abdomen, back, and upper thighs over a 1-month period. There were no lesions in his oral cavity or around his eyes, nose, or penile region. He denied dysphagia.
The patient had multiple comorbidities, including diabetes, hypertension, recent stroke, and end-stage renal disease. He was being prepared for dialysis. His medications included torsemide, warfarin, amiodarone, metoprolol, pantoprozole, atorvastatin, and nifedipine. About 3 months prior to this presentation, he was started on oral linaglipton 5 mg/d, an oral antihyperglycemic medication. He had no history of skin disease or cancer, and his family history was not significant.
Physical examination showed multiple 5-mm to 2-cm blisters and bullae on the flexural surface of both of his arms (FIGURE), back, lower abdomen, and upper thighs. His palms and soles were not involved. The lesions were nontender, tense, and filled with clear fluid. Some were intact and others were rupturing. There was no mucocutaneous involvement. Nikolsky sign was negative. There were no signs of bleeding.
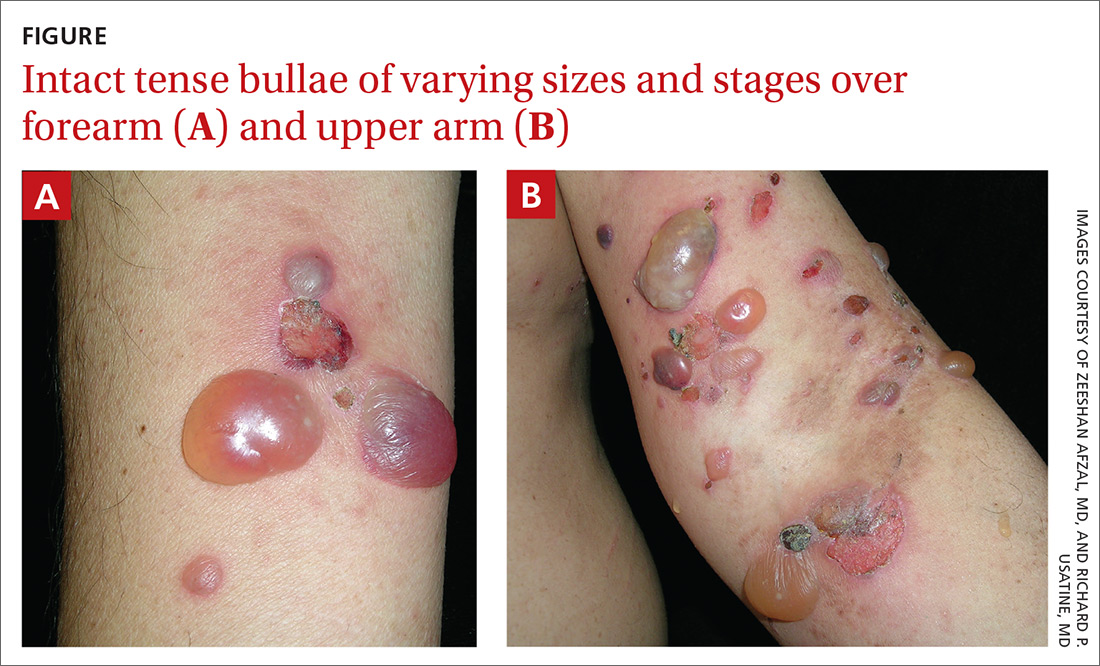
The family physician (FP) obtained a 4-mm punch biopsy at the edge of a 6-mm blister for light microscopy and a 3-mm perilesional punch biopsy for direct immunofluorescence (DIF) microscopy.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Dx: Bullous pemphigoid secondary to linagliptin use
DIF of the biopsy sample demonstrated linear deposition of complement 3 (C3) and immunoglobulin (Ig) G along the basement membrane zone. Indirect immunofluorescence on salt-split skin demonstrated linear deposition of IgG and C3 on both the roof and floor of the induced blisters. These findings and the patient’s clinical presentation met the criteria for bullous pemphigoid (BP), which is the most common autoimmune skin-blistering disease.1
BP is associated with subepidermal blistering, which can occur in reaction to a variety of triggers. Pathogenesis of this condition involves IgG anti-basement membrane autoantibody complex formation with the hemidesmosomal antigens BP230 and BP180—a process that activates C3 and the release of proteases that can be destructive to tissue along the dermo-epidermal junction.1
Growing incidence. BP usually occurs in patients > 60 years, with no racial or gender preference.1 The incidence rate of BP ranges from 2.4 to 21.7 new cases per 1 million individuals among various worldwide populations.2 The incidence appears to have increased 1.9- to 4.3-fold over the past 2 decades.2
What you’ll see, who’s at risk
Symptoms of BP include localized areas of erythema or pruritic urticarial plaques that gradually become more extensive. A patient may have pruritis alone for an extended period prior to developing blisters and bullae. The bullae are tense and normally 1 to 7 cm in size.1 Eruption is generalized, mostly affecting the lower abdomen, as well as the flexural parts of the extremities. The palms and soles also can be affected.
FPs should be aware of the atypical clinical variants of BP. In a review by Kridin and Ludwig, variants can be prurigo-like, eczema-like, urticaria-like, dyshidrosiform type, erosive type, and erythema annulare centrifugum–like type.2 At-risk populations, such as elderly patients (> 70 years), whose pruritis manifests with or without bullous formation, should be screened for BP.3,4
Continue to: Risk factors for BP
Risk factors for BP. Certain conditions linked to developing BP include neurologic disorders (dementia and Parkinson disease) and psychiatric disorders (unipolar and bipolar disorder).4 Further, it is important to note any medications that could be the cause of a patient’s BP, including dipeptidyl peptidase-4 (DPP-4) inhibitors, psychotropic medications, spironolactone, furosemide, beta-blockers, and antibiotics.3 This patient was taking a beta-blocker (metoprolol) and a DPP-4 inhibitor (linagliptin). Because he was most recently started on linagliptin, we suspected it may have had a causal role in the development of BP.
The association of DPP-4 inhibitors and BP
FPs are increasingly using DPP-4 inhibitors—including sitagliptin, vildagliptin, and linagliptin—as oral antihyperglycemic agents for type 2 diabetes mellitus. Therefore, it’s important to recognize this medication class’s association with BP.5 In a case-control study of 165 patients with BP, Benzaquen et al reported that 28 patients who were taking DPP-4 inhibitors had an associated increased risk for BP (adjusted odds ratio = 2.64; 95% confidence interval [CI], 1.19-5.85).3
The pathophysiology of BP associated with DPP-4 inhibitors remains unclear, but mechanisms have been proposed. The DPP-4 enzyme is expressed on many cells, including keratinocytes, T cells, and endothelial cells.3 It is possible that DPP-4 inhibition at these cells could stimulate activity of inflammatory cytokines, which can lead to enhanced local eosinophil activation and trigger bullous formation. DPP-4 enzymes are also involved in forming plasmin, which is a protease that cleaves BP180.3 Inhibition of this process can affect proper cleavage of BP180, impacting its function and antigenicity.3,6
Other conditions that also exhibit blisters
There are some skin conditions with similar presentations that need to be ruled out in the work-up.
Bullous diabeticorum is a rare, spontaneous, noninflammatory condition found in patients with diabetes.1 Blisters usually manifest as large, tense, asymmetrical, mildly tender lesions that commonly affect the feet and lower legs but can involve the trunk. These usually develop overnight without preceding trauma. Biopsy would show both intra-epidermal and subepidermal bulla with normal DIF findings.1 This condition usually has an excellent prognosis.
Continue to: Pemphigus vulgaris
Pemphigus vulgaris is characterized by nonpruritic, flaccid, painful blisters. This condition usually begins with manifestation of painful oral lesions that evolve into skin blisters. Some patients can develop mucocutaneous lesions.1 Nikolsky sign is positive in these cases. Light microscopy would show intra-epidermal bullae.
Dermatitis herpetiformis. This condition—usually affecting middle-age patients—is associated with severe pruritis and burning. It may start with a few pruritic papules or vesicles that later evolve into urticarial papules, vesicles, or bullae. Dermatitis herpetiformis can resemble herpes simplex virus. It can also be associated with gluten-sensitive enteropathy and small bowel lymphoma.1 DIF of a biopsy sample would show granular deposition of IgA within the tips of the dermal papillae and along the basement membrane of perilesional skin.1
Epidermolysis bullosa acquisita is a rare, severe, chronic condition with subepidermal mucocutaneous blistering.1 It is associated with skin fragility and spontaneous trauma-induced blisters that heal with scar formation and milia. IgG autoantibodies reacting to proteins in the basement membrane zone can cause the disease. It is also associated with Crohn disease.1 DIF findings are similar in BP, but they are differentiated by location of IgG deposits; they can be found on the dermal side of separation in epidermolysis bullosa acquisita, as compared with the epidermal side in BP.1
How to make the Dx in 3 steps
To effectively diagnose and classify BP, use the following 3-step method:
- Establish the presence of 3 of 4 clinical characteristics: patient’s age > 60 years, absence of atrophic scars, absence of mucosal involvement, and absence of bullous lesions on the head and neck.
- Order light microscopy. Findings should be consistent with eosinophils and neutrophils containing subepidermal bullae.
- Order a punch biopsy to obtain a perilesional specimen. DIF of the biopsy findings should feature linear deposits of IgG with or without C3 along the dermo-epidermal junction. This step is essential for an accurate diagnosis.
There also is benefit in ordering supplemental studies, such as an enzyme-linked immunosorbent assay for the detection of anti-BP180 or anti-BP230 IgG autoantibodies.7 However, for this patient, we did not order this study.
Continue to: Management focuses on steroids
Management focuses on steroids
The offending agent should be discontinued immediately. Depending on the severity of disease, treatment can include the use of potent topical corticosteroids alone or in combination with systemic corticosteroids and anti-inflammatory antibiotics (eg, doxycycline, minocycline, erythromycin).1,7 For patients with resistant or refractory disease, consider azathioprine, methotrexate, dapsone, and chlorambucil.1,7 Exceptional cases may benefit from the use of mycophenolate mofetil, intravenous immunoglobulin, or plasmapheresis.1,7
For this patient, initial treatment included discontinuation of linagliption and introduction of topical clobetasol 0.05% and oral prednisone 40 mg/d for 7 days, followed by prednisone 20 mg for 7 days. He was also started on oral doxycycline 100 mg bid and oral nicotinamide 500 mg bid.
1. Habif TP. Vesicular and bullous diseases. In: Habif TP, ed. Clinical Dermatology: a Color Guide to Diagnosis and Therapy. 6th ed. Elsevier; 2016:635-666.
2. Kridin K, Ludwig RJ. The growing incidence of bullous pemphigoid: overview and potential explanations. Front Med (Lausanne). 2018;5:220.
3. Benzaquen M, Borradori L, Berbis P, et al. Dipeptidyl peptidase IV inhibitors, a risk factor for bullous pemphigoid: retrospective multicenter case-control study from France and Switzerland. J Am Acad Dermatol. 2017;78:1090-1096.
4. Bastuji-Garin S, Joly P, Lemordant P, et al. Risk factors for bullous pemphigoid in the elderly: a prospective case-control study. J Invest Dermatol. 2011;131:637-643.
5. Kridin K, Bergman R. Association of bullous pemphigoid with dipeptidyl-peptidase 4 inhibitors in patients with diabetes: estimating the risk of the new agents and characterizing the patients. JAMA Dermatol. 2018;154:1152-1158.
6. Haber R, Fayad AM, Stephan F, et al. Bullous pemphigoid associated with linagliptin treatment. JAMA Dermatol. 2016;152:224-226.Management of bullous pemphigoid: the European Dermatology Forum consensus in collaboration with the European Academy of Dermatology and Venereology. Br J Dermatol. 2015;172:867-877.
A 62-year-old man presented to our skin clinic with multiple pruritic, tense, bullous lesions that manifested on his arms, abdomen, back, and upper thighs over a 1-month period. There were no lesions in his oral cavity or around his eyes, nose, or penile region. He denied dysphagia.
The patient had multiple comorbidities, including diabetes, hypertension, recent stroke, and end-stage renal disease. He was being prepared for dialysis. His medications included torsemide, warfarin, amiodarone, metoprolol, pantoprozole, atorvastatin, and nifedipine. About 3 months prior to this presentation, he was started on oral linaglipton 5 mg/d, an oral antihyperglycemic medication. He had no history of skin disease or cancer, and his family history was not significant.
Physical examination showed multiple 5-mm to 2-cm blisters and bullae on the flexural surface of both of his arms (FIGURE), back, lower abdomen, and upper thighs. His palms and soles were not involved. The lesions were nontender, tense, and filled with clear fluid. Some were intact and others were rupturing. There was no mucocutaneous involvement. Nikolsky sign was negative. There were no signs of bleeding.

The family physician (FP) obtained a 4-mm punch biopsy at the edge of a 6-mm blister for light microscopy and a 3-mm perilesional punch biopsy for direct immunofluorescence (DIF) microscopy.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Dx: Bullous pemphigoid secondary to linagliptin use
DIF of the biopsy sample demonstrated linear deposition of complement 3 (C3) and immunoglobulin (Ig) G along the basement membrane zone. Indirect immunofluorescence on salt-split skin demonstrated linear deposition of IgG and C3 on both the roof and floor of the induced blisters. These findings and the patient’s clinical presentation met the criteria for bullous pemphigoid (BP), which is the most common autoimmune skin-blistering disease.1
BP is associated with subepidermal blistering, which can occur in reaction to a variety of triggers. Pathogenesis of this condition involves IgG anti-basement membrane autoantibody complex formation with the hemidesmosomal antigens BP230 and BP180—a process that activates C3 and the release of proteases that can be destructive to tissue along the dermo-epidermal junction.1
Growing incidence. BP usually occurs in patients > 60 years, with no racial or gender preference.1 The incidence rate of BP ranges from 2.4 to 21.7 new cases per 1 million individuals among various worldwide populations.2 The incidence appears to have increased 1.9- to 4.3-fold over the past 2 decades.2
What you’ll see, who’s at risk
Symptoms of BP include localized areas of erythema or pruritic urticarial plaques that gradually become more extensive. A patient may have pruritis alone for an extended period prior to developing blisters and bullae. The bullae are tense and normally 1 to 7 cm in size.1 Eruption is generalized, mostly affecting the lower abdomen, as well as the flexural parts of the extremities. The palms and soles also can be affected.
FPs should be aware of the atypical clinical variants of BP. In a review by Kridin and Ludwig, variants can be prurigo-like, eczema-like, urticaria-like, dyshidrosiform type, erosive type, and erythema annulare centrifugum–like type.2 At-risk populations, such as elderly patients (> 70 years), whose pruritis manifests with or without bullous formation, should be screened for BP.3,4
Continue to: Risk factors for BP
Risk factors for BP. Certain conditions linked to developing BP include neurologic disorders (dementia and Parkinson disease) and psychiatric disorders (unipolar and bipolar disorder).4 Further, it is important to note any medications that could be the cause of a patient’s BP, including dipeptidyl peptidase-4 (DPP-4) inhibitors, psychotropic medications, spironolactone, furosemide, beta-blockers, and antibiotics.3 This patient was taking a beta-blocker (metoprolol) and a DPP-4 inhibitor (linagliptin). Because he was most recently started on linagliptin, we suspected it may have had a causal role in the development of BP.
The association of DPP-4 inhibitors and BP
FPs are increasingly using DPP-4 inhibitors—including sitagliptin, vildagliptin, and linagliptin—as oral antihyperglycemic agents for type 2 diabetes mellitus. Therefore, it’s important to recognize this medication class’s association with BP.5 In a case-control study of 165 patients with BP, Benzaquen et al reported that 28 patients who were taking DPP-4 inhibitors had an associated increased risk for BP (adjusted odds ratio = 2.64; 95% confidence interval [CI], 1.19-5.85).3
The pathophysiology of BP associated with DPP-4 inhibitors remains unclear, but mechanisms have been proposed. The DPP-4 enzyme is expressed on many cells, including keratinocytes, T cells, and endothelial cells.3 It is possible that DPP-4 inhibition at these cells could stimulate activity of inflammatory cytokines, which can lead to enhanced local eosinophil activation and trigger bullous formation. DPP-4 enzymes are also involved in forming plasmin, which is a protease that cleaves BP180.3 Inhibition of this process can affect proper cleavage of BP180, impacting its function and antigenicity.3,6
Other conditions that also exhibit blisters
There are some skin conditions with similar presentations that need to be ruled out in the work-up.
Bullous diabeticorum is a rare, spontaneous, noninflammatory condition found in patients with diabetes.1 Blisters usually manifest as large, tense, asymmetrical, mildly tender lesions that commonly affect the feet and lower legs but can involve the trunk. These usually develop overnight without preceding trauma. Biopsy would show both intra-epidermal and subepidermal bulla with normal DIF findings.1 This condition usually has an excellent prognosis.
Continue to: Pemphigus vulgaris
Pemphigus vulgaris is characterized by nonpruritic, flaccid, painful blisters. This condition usually begins with manifestation of painful oral lesions that evolve into skin blisters. Some patients can develop mucocutaneous lesions.1 Nikolsky sign is positive in these cases. Light microscopy would show intra-epidermal bullae.
Dermatitis herpetiformis. This condition—usually affecting middle-age patients—is associated with severe pruritis and burning. It may start with a few pruritic papules or vesicles that later evolve into urticarial papules, vesicles, or bullae. Dermatitis herpetiformis can resemble herpes simplex virus. It can also be associated with gluten-sensitive enteropathy and small bowel lymphoma.1 DIF of a biopsy sample would show granular deposition of IgA within the tips of the dermal papillae and along the basement membrane of perilesional skin.1
Epidermolysis bullosa acquisita is a rare, severe, chronic condition with subepidermal mucocutaneous blistering.1 It is associated with skin fragility and spontaneous trauma-induced blisters that heal with scar formation and milia. IgG autoantibodies reacting to proteins in the basement membrane zone can cause the disease. It is also associated with Crohn disease.1 DIF findings are similar in BP, but they are differentiated by location of IgG deposits; they can be found on the dermal side of separation in epidermolysis bullosa acquisita, as compared with the epidermal side in BP.1
How to make the Dx in 3 steps
To effectively diagnose and classify BP, use the following 3-step method:
- Establish the presence of 3 of 4 clinical characteristics: patient’s age > 60 years, absence of atrophic scars, absence of mucosal involvement, and absence of bullous lesions on the head and neck.
- Order light microscopy. Findings should be consistent with eosinophils and neutrophils containing subepidermal bullae.
- Order a punch biopsy to obtain a perilesional specimen. DIF of the biopsy findings should feature linear deposits of IgG with or without C3 along the dermo-epidermal junction. This step is essential for an accurate diagnosis.
There also is benefit in ordering supplemental studies, such as an enzyme-linked immunosorbent assay for the detection of anti-BP180 or anti-BP230 IgG autoantibodies.7 However, for this patient, we did not order this study.
Continue to: Management focuses on steroids
Management focuses on steroids
The offending agent should be discontinued immediately. Depending on the severity of disease, treatment can include the use of potent topical corticosteroids alone or in combination with systemic corticosteroids and anti-inflammatory antibiotics (eg, doxycycline, minocycline, erythromycin).1,7 For patients with resistant or refractory disease, consider azathioprine, methotrexate, dapsone, and chlorambucil.1,7 Exceptional cases may benefit from the use of mycophenolate mofetil, intravenous immunoglobulin, or plasmapheresis.1,7
For this patient, initial treatment included discontinuation of linagliption and introduction of topical clobetasol 0.05% and oral prednisone 40 mg/d for 7 days, followed by prednisone 20 mg for 7 days. He was also started on oral doxycycline 100 mg bid and oral nicotinamide 500 mg bid.
A 62-year-old man presented to our skin clinic with multiple pruritic, tense, bullous lesions that manifested on his arms, abdomen, back, and upper thighs over a 1-month period. There were no lesions in his oral cavity or around his eyes, nose, or penile region. He denied dysphagia.
The patient had multiple comorbidities, including diabetes, hypertension, recent stroke, and end-stage renal disease. He was being prepared for dialysis. His medications included torsemide, warfarin, amiodarone, metoprolol, pantoprozole, atorvastatin, and nifedipine. About 3 months prior to this presentation, he was started on oral linaglipton 5 mg/d, an oral antihyperglycemic medication. He had no history of skin disease or cancer, and his family history was not significant.
Physical examination showed multiple 5-mm to 2-cm blisters and bullae on the flexural surface of both of his arms (FIGURE), back, lower abdomen, and upper thighs. His palms and soles were not involved. The lesions were nontender, tense, and filled with clear fluid. Some were intact and others were rupturing. There was no mucocutaneous involvement. Nikolsky sign was negative. There were no signs of bleeding.

The family physician (FP) obtained a 4-mm punch biopsy at the edge of a 6-mm blister for light microscopy and a 3-mm perilesional punch biopsy for direct immunofluorescence (DIF) microscopy.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Dx: Bullous pemphigoid secondary to linagliptin use
DIF of the biopsy sample demonstrated linear deposition of complement 3 (C3) and immunoglobulin (Ig) G along the basement membrane zone. Indirect immunofluorescence on salt-split skin demonstrated linear deposition of IgG and C3 on both the roof and floor of the induced blisters. These findings and the patient’s clinical presentation met the criteria for bullous pemphigoid (BP), which is the most common autoimmune skin-blistering disease.1
BP is associated with subepidermal blistering, which can occur in reaction to a variety of triggers. Pathogenesis of this condition involves IgG anti-basement membrane autoantibody complex formation with the hemidesmosomal antigens BP230 and BP180—a process that activates C3 and the release of proteases that can be destructive to tissue along the dermo-epidermal junction.1
Growing incidence. BP usually occurs in patients > 60 years, with no racial or gender preference.1 The incidence rate of BP ranges from 2.4 to 21.7 new cases per 1 million individuals among various worldwide populations.2 The incidence appears to have increased 1.9- to 4.3-fold over the past 2 decades.2
What you’ll see, who’s at risk
Symptoms of BP include localized areas of erythema or pruritic urticarial plaques that gradually become more extensive. A patient may have pruritis alone for an extended period prior to developing blisters and bullae. The bullae are tense and normally 1 to 7 cm in size.1 Eruption is generalized, mostly affecting the lower abdomen, as well as the flexural parts of the extremities. The palms and soles also can be affected.
FPs should be aware of the atypical clinical variants of BP. In a review by Kridin and Ludwig, variants can be prurigo-like, eczema-like, urticaria-like, dyshidrosiform type, erosive type, and erythema annulare centrifugum–like type.2 At-risk populations, such as elderly patients (> 70 years), whose pruritis manifests with or without bullous formation, should be screened for BP.3,4
Continue to: Risk factors for BP
Risk factors for BP. Certain conditions linked to developing BP include neurologic disorders (dementia and Parkinson disease) and psychiatric disorders (unipolar and bipolar disorder).4 Further, it is important to note any medications that could be the cause of a patient’s BP, including dipeptidyl peptidase-4 (DPP-4) inhibitors, psychotropic medications, spironolactone, furosemide, beta-blockers, and antibiotics.3 This patient was taking a beta-blocker (metoprolol) and a DPP-4 inhibitor (linagliptin). Because he was most recently started on linagliptin, we suspected it may have had a causal role in the development of BP.
The association of DPP-4 inhibitors and BP
FPs are increasingly using DPP-4 inhibitors—including sitagliptin, vildagliptin, and linagliptin—as oral antihyperglycemic agents for type 2 diabetes mellitus. Therefore, it’s important to recognize this medication class’s association with BP.5 In a case-control study of 165 patients with BP, Benzaquen et al reported that 28 patients who were taking DPP-4 inhibitors had an associated increased risk for BP (adjusted odds ratio = 2.64; 95% confidence interval [CI], 1.19-5.85).3
The pathophysiology of BP associated with DPP-4 inhibitors remains unclear, but mechanisms have been proposed. The DPP-4 enzyme is expressed on many cells, including keratinocytes, T cells, and endothelial cells.3 It is possible that DPP-4 inhibition at these cells could stimulate activity of inflammatory cytokines, which can lead to enhanced local eosinophil activation and trigger bullous formation. DPP-4 enzymes are also involved in forming plasmin, which is a protease that cleaves BP180.3 Inhibition of this process can affect proper cleavage of BP180, impacting its function and antigenicity.3,6
Other conditions that also exhibit blisters
There are some skin conditions with similar presentations that need to be ruled out in the work-up.
Bullous diabeticorum is a rare, spontaneous, noninflammatory condition found in patients with diabetes.1 Blisters usually manifest as large, tense, asymmetrical, mildly tender lesions that commonly affect the feet and lower legs but can involve the trunk. These usually develop overnight without preceding trauma. Biopsy would show both intra-epidermal and subepidermal bulla with normal DIF findings.1 This condition usually has an excellent prognosis.
Continue to: Pemphigus vulgaris
Pemphigus vulgaris is characterized by nonpruritic, flaccid, painful blisters. This condition usually begins with manifestation of painful oral lesions that evolve into skin blisters. Some patients can develop mucocutaneous lesions.1 Nikolsky sign is positive in these cases. Light microscopy would show intra-epidermal bullae.
Dermatitis herpetiformis. This condition—usually affecting middle-age patients—is associated with severe pruritis and burning. It may start with a few pruritic papules or vesicles that later evolve into urticarial papules, vesicles, or bullae. Dermatitis herpetiformis can resemble herpes simplex virus. It can also be associated with gluten-sensitive enteropathy and small bowel lymphoma.1 DIF of a biopsy sample would show granular deposition of IgA within the tips of the dermal papillae and along the basement membrane of perilesional skin.1
Epidermolysis bullosa acquisita is a rare, severe, chronic condition with subepidermal mucocutaneous blistering.1 It is associated with skin fragility and spontaneous trauma-induced blisters that heal with scar formation and milia. IgG autoantibodies reacting to proteins in the basement membrane zone can cause the disease. It is also associated with Crohn disease.1 DIF findings are similar in BP, but they are differentiated by location of IgG deposits; they can be found on the dermal side of separation in epidermolysis bullosa acquisita, as compared with the epidermal side in BP.1
How to make the Dx in 3 steps
To effectively diagnose and classify BP, use the following 3-step method:
- Establish the presence of 3 of 4 clinical characteristics: patient’s age > 60 years, absence of atrophic scars, absence of mucosal involvement, and absence of bullous lesions on the head and neck.
- Order light microscopy. Findings should be consistent with eosinophils and neutrophils containing subepidermal bullae.
- Order a punch biopsy to obtain a perilesional specimen. DIF of the biopsy findings should feature linear deposits of IgG with or without C3 along the dermo-epidermal junction. This step is essential for an accurate diagnosis.
There also is benefit in ordering supplemental studies, such as an enzyme-linked immunosorbent assay for the detection of anti-BP180 or anti-BP230 IgG autoantibodies.7 However, for this patient, we did not order this study.
Continue to: Management focuses on steroids
Management focuses on steroids
The offending agent should be discontinued immediately. Depending on the severity of disease, treatment can include the use of potent topical corticosteroids alone or in combination with systemic corticosteroids and anti-inflammatory antibiotics (eg, doxycycline, minocycline, erythromycin).1,7 For patients with resistant or refractory disease, consider azathioprine, methotrexate, dapsone, and chlorambucil.1,7 Exceptional cases may benefit from the use of mycophenolate mofetil, intravenous immunoglobulin, or plasmapheresis.1,7
For this patient, initial treatment included discontinuation of linagliption and introduction of topical clobetasol 0.05% and oral prednisone 40 mg/d for 7 days, followed by prednisone 20 mg for 7 days. He was also started on oral doxycycline 100 mg bid and oral nicotinamide 500 mg bid.
1. Habif TP. Vesicular and bullous diseases. In: Habif TP, ed. Clinical Dermatology: a Color Guide to Diagnosis and Therapy. 6th ed. Elsevier; 2016:635-666.
2. Kridin K, Ludwig RJ. The growing incidence of bullous pemphigoid: overview and potential explanations. Front Med (Lausanne). 2018;5:220.
3. Benzaquen M, Borradori L, Berbis P, et al. Dipeptidyl peptidase IV inhibitors, a risk factor for bullous pemphigoid: retrospective multicenter case-control study from France and Switzerland. J Am Acad Dermatol. 2017;78:1090-1096.
4. Bastuji-Garin S, Joly P, Lemordant P, et al. Risk factors for bullous pemphigoid in the elderly: a prospective case-control study. J Invest Dermatol. 2011;131:637-643.
5. Kridin K, Bergman R. Association of bullous pemphigoid with dipeptidyl-peptidase 4 inhibitors in patients with diabetes: estimating the risk of the new agents and characterizing the patients. JAMA Dermatol. 2018;154:1152-1158.
6. Haber R, Fayad AM, Stephan F, et al. Bullous pemphigoid associated with linagliptin treatment. JAMA Dermatol. 2016;152:224-226.Management of bullous pemphigoid: the European Dermatology Forum consensus in collaboration with the European Academy of Dermatology and Venereology. Br J Dermatol. 2015;172:867-877.
1. Habif TP. Vesicular and bullous diseases. In: Habif TP, ed. Clinical Dermatology: a Color Guide to Diagnosis and Therapy. 6th ed. Elsevier; 2016:635-666.
2. Kridin K, Ludwig RJ. The growing incidence of bullous pemphigoid: overview and potential explanations. Front Med (Lausanne). 2018;5:220.
3. Benzaquen M, Borradori L, Berbis P, et al. Dipeptidyl peptidase IV inhibitors, a risk factor for bullous pemphigoid: retrospective multicenter case-control study from France and Switzerland. J Am Acad Dermatol. 2017;78:1090-1096.
4. Bastuji-Garin S, Joly P, Lemordant P, et al. Risk factors for bullous pemphigoid in the elderly: a prospective case-control study. J Invest Dermatol. 2011;131:637-643.
5. Kridin K, Bergman R. Association of bullous pemphigoid with dipeptidyl-peptidase 4 inhibitors in patients with diabetes: estimating the risk of the new agents and characterizing the patients. JAMA Dermatol. 2018;154:1152-1158.
6. Haber R, Fayad AM, Stephan F, et al. Bullous pemphigoid associated with linagliptin treatment. JAMA Dermatol. 2016;152:224-226.Management of bullous pemphigoid: the European Dermatology Forum consensus in collaboration with the European Academy of Dermatology and Venereology. Br J Dermatol. 2015;172:867-877.
Breaking the cycle of medication overuse headache
Medication overuse headache (MOH), a secondary headache diagnosis, is a prevalent phenomenon that complicates headache diagnosis and treatment, increases the cost of care, and reduces quality of life. Effective abortive medication is essential for the headache sufferer; when an abortive is used too frequently, however, headache frequency increases—potentially beginning a cycle in which the patient then takes more medication to abort the headache. Over time, the patient suffers from an ever-increasing number of headaches, takes even more abortive medication, and so on. In the presence of MOH, there is a reduction in pain response to preventive and abortive treatments; when medication overuse is eliminated, pain response improves.1
Although MOH is well recognized among headache specialists, the condition is often overlooked in primary care. Since headache is a top complaint in primary care, however, and prevention is a major goal in family medicine, the opportunity for you to recognize, treat, and prevent MOH is significant. In fact, a randomized controlled trial showed that brief patient education about headache care and MOH provided by a primary care physician can lead to a significant reduction in headache frequency among patients with MOH.2
This article reviews the recognition and diagnosis of MOH, based on historical features and current criteria; addresses risk factors for abortive medication overuse and how to withdraw an offending agent; and explores the value of bridging and preventive therapies to reduce the overall frequency of headache.

What defines MOH?
Typically, MOH is a chronification of a primary headache disorder. However, in patients with a history of migraine who are undergoing treatment for another chronic pain condition with an opioid or other analgesic, MOH can be induced.3 An increase in the frequency of headache raises the specter of a concomitant increase in the level of disability4; psychiatric comorbidity5; and more headache days, with time lost from school and work.
The Migraine Disability Assessment (MIDAS) questionnaire, a validated instrument that helps the provider (1) measure the impact that headache has on a patient’s life and (2) follow treatment progress, also provides information to employers and insurance companies on treatment coverage and the need for work modification. The MIDAS score is 3 times higher in patients with MOH than in patients with episodic migraine.6,7
The annual associated cost per person of MOH has been estimated at $4000, resulting in billions of dollars in associated costs8; most of these costs are related to absenteeism and disability. After detoxification for MOH, annual outpatient medication costs are reduced by approximately 24%.9
Efforts to solve a common problem create another
Headache affects nearly 50% of the general population worldwide,10 accounting for about 4% of primary care visits11 and approximately 20% of outpatient neurology consultations.12 Although inpatient stays for headache are approximately half the duration of the overall average hospital stay, headache accounts for 3% of admissions.13 According to the Global Burden of Disease study, tension-type headache, migraine, and MOH are the 3 most common headache disorders.10 Headache is the second leading cause of disability among people 15 to 49 years of age.10
Continue to: The prevalence of MOH...
The prevalence of MOH in the general population is 2%.7,14,15 A population-based study showed that the rate of progression from episodic headache (< 15 d/mo) to chronic headache (≥ 15 d/mo) in the general population is 2.5% per year16; however, progression to chronic headache is 14% per year in patients with medication overuse. One-third of the general population with chronic migraine overuses symptomatic medication; in US headache clinics, roughly one-half of patients with chronic headache overuse acute medication.6
Definitions and diagnosis
MOH is a secondary headache diagnosis in the third edition of the International Classification of Headache Disorders (ICHD-3) (TABLE 1),17 which lists diagnostic criteria for recognized headache disorders.

Terminology. MOH has also been called rebound headache, drug-induced headache, and transformed migraine, but these terms are outdated and are not formal diagnoses. Patients sometimes refer to substance-withdrawal headaches (not discussed in this article) as rebound headaches, so clarity is important when discussing headache with patients: namely, that MOH is an exacerbation of an existing headache condition caused by overuse of abortive headache medications, including analgesics, combination analgesics, triptans, barbiturates, and opioids.
MOH was recognized in the early 1950s and fully differentiated as a diagnosis in 2005 in the second edition of the ICHD. The disorder is subcategorized by offending abortive agent (TABLE 217) because the frequency of analgesic use required to develop MOH differs by agent.

Risk factors for MOH and chronification of a primary headache disorder. There are several risk factors for developing MOH, and others that contribute to increasing headache frequency in general (TABLE 35,14,18-23). Some risk factors are common to each. All are important to address because some are modifiable.
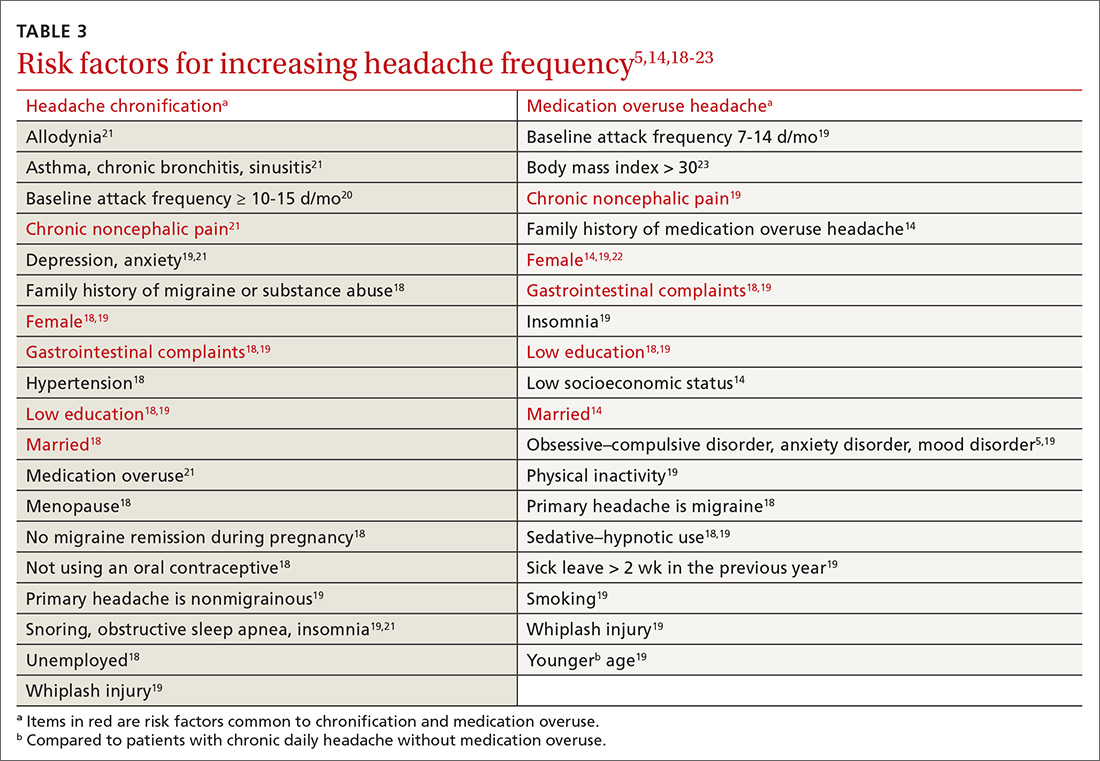
Continue to: Pathophysiology
Pathophysiology. The pathophysiology and psychology behind MOH are largely unknown. Physiologic changes in pain processing and functional imaging changes have been demonstrated in patients with MOH, both of which are reversible upon withdrawal of medication.23 Genetic factors and changes in hormone and neurotransmitter levels are found in MOH patients; this is not the case in patients who have an episodic headache pattern only.24
Presentation. Diagnostic criteria for MOH do not include clinical characteristics. Typically, the phenotype of MOH in a given patient is similar to the underlying primary headache25—although this principle can be complicated to tease out because these medications can suppress some symptoms. Diagnosis of a primary headache disorder should be documented along with the diagnosis of MOH.
Medication overuse can exist without MOH: Not every patient who frequently uses an abortive medication develops MOH.
Treatment is multifaceted—and can become complex
Mainstays of treatment of MOH are education about the disorder and detoxification from the overused agent, although specific treatments can differ depending on the agent involved, the frequency and duration of its use, and a patient’s behavioral patterns and psychiatric comorbidities. Often, a daily medication to prevent headache is considered upon, or after, withdrawal of the offending agent. The timing of introducing a preventive might impact its effectiveness. Some refractory cases require more intensive therapy, including hospitalization at a specialized tertiary center.
But before we look at detoxification from an overused agent, it’s important to review one of the best strategies of all in combatting MOH.
Continue to: First and best strategy
First and best strategy: Avoid onset of MOH
Select an appropriate abortive to reduce the risk of MOH. With regard to specific acute headache medications, some nuances other than type of headache should be considered. Nonsteroidal anti-inflammatory drugs (NSAIDs) are recommended as abortive therapy by the American Headache Society for their efficacy, favorable adverse effect profile, and low cost. NSAIDs are protective against development of MOH if a patient’s baseline headache frequency is < 10/mo; at a frequency of 10 to 14 d/mo, however, the risk of MOH increases when using an NSAID.6 A similar effect has been seen with triptans.16 Longer-acting NSAIDs, such as nabumetone and naproxen, have been proposed as less likely to cause MOH, and are even used as bridging therapy sometimes (as long as neither of these was the overused medication).26
The time it takes to develop MOH is shortest with triptans, followed by ergots, then analgesics.27
Prospective cohort studies6,16 have shown that barbiturates and opioids are more likely to induce MOH; for that reason, agents in these analgesic classes are almost universally avoided unless no other medically acceptable options exist. Using barbiturate-containing compounds or opioids > 4 d/mo exponentially increases the likelihood of MOH.
Promising preclinical data demonstrate that the gepant, or small-molecule calcitonin gene-related peptide (CGRP) receptor antagonist, class of medications used as abortive therapy does not induce medication overuse cutaneous allodynia.28
Provide education. Primary prevention of MOH involves (1) increasing patients’ awareness of how to take medications appropriately and (2) restricting intake of over-the-counter abortive medications. Often, the expert recommendation is to limit abortives to approximately 2 d/wk because more frequent use places patients at risk of further increased use and subsequent MOH.
Continue to: A randomized controlled trial in Norway...
A randomized controlled trial in Norway compared outcomes in 2 groups of patients with MOH: One group was given advice on the disorder by a physician; the other group was not provided with advice. In the “business-as-usual” group, there was no significant improvement; however, when general practitioners provided simple advice (lasting roughly 9 minutes) about reducing abortive medication use to a safe level and cautioned patients that they would be “feeling worse before feeling better,” headache days were reduced by approximately 8 per month and medication days, by 16 per month.2
A subsequent, long-term follow-up study29 of patients from the Norway trial2 who had been given advice and education showed a relapse rate (ie, into overuse of headache medication) of only 8% and sustained reduction of headache days and medication use at 16 months.
Offer support and other nondrug interventions. A recent review of 3 studies23 recommended that extra support for patients from a headache nurse, close follow-up, keeping an electronic diary that provides feedback, and undertaking a short course of psychotherapy can reduce medication overuse and prevent relapse.
If MOH develops, initiate withdrawal, introduce a preventive
Withdraw overused medication. Most current evidence suggests that withdrawal of the offending agent is the most effective factor in reducing headache days and improving quality of life. A randomized controlled trial compared the effects of (1) complete and immediate withdrawal of an abortive medication with (2) reducing its use (ie, limiting intake to 2 d/wk), on headache frequency, disability, and quality of life.30 There was a reduction of headache days in both groups; however, reduction was much greater at 2 months in the complete withdrawal group than in the restricted intake group (respectively, a 41% and a 26% reduction in headache days per month). This effect was sustained at 6 and 12 months in both groups. The study confirmed the results of earlier research2,15: Abrupt withdrawal leads to reversion to an episodic pattern at 2 to 6 months in approximately 40% to 60% of patients.
More studies are needed to determine the most appropriate treatment course for MOH; however, complete withdrawal of the causative drug is the most important intervention.
Continue to: Consider withdrawal plus preventive treatment
Consider withdrawal plus preventive treatment. Use of sodium valproate, in addition to medication overuse detoxification, led to a significant reduction in headache days and improvement in quality of life at 12 weeks but no difference after 24 weeks, compared with detoxification alone in a randomized, double-blind, placebo-controlled study.31
A study of 61 patients showed a larger reduction (by 7.2 d/mo) in headache frequency with any preventive medication in addition to medication withdrawal, compared to withdrawal alone (by 4.1 d/mo) after 3 months; however, the relative benefit was gone at 6 months.32
A study of 98 patients compared immediate and delayed initiation of preventive medication upon withdrawal of overused abortive medication.33 Response was defined as a > 50% reduction in headache frequency and was similar in both groups; results showed a 28% response with immediate initiation of a preventive; a 23% response with delayed (ie, 2 months after withdrawal) initiation; and a 48% response in both groups at 12 months.
Collectively, these studies suggest that adding a preventive medication at the time of withdrawal has the potential to reduce headache frequency more quickly than withdrawal alone. However, after 3 to 6 months, the outcome of reduced headache frequency is the same whether or not a preventive medication is used—as long as the offending agent has been withdrawn.
Do preventives work without withdrawing overused medication? Patients with MOH often show little or no improvement with addition of a preventive medication only; their response to a preventive improves after withdrawal of the overused medication. Patients without previous headache improvement after addition of a preventive, who also did not improve 2 months after withdrawal, then demonstrated an overall reduction in headache by 26% when a preventive was reintroduced after withdrawal.2
Continue to: The research evidence for preventives
The research evidence for preventives. Medications for headache prevention have not been extensively evaluated specifically for treating MOH. Here is what’s known:
- Flunarizine, amitriptyline, and beta-blockers usually are ineffective for MOH.24
- Results for topiramate are mixed: A small, double-blind, placebo-controlled chronic migraine study in Europe showed that, in a subgroup of patients with MOH, topiramate led to a small but significant reduction (3.5 d/mo) in headache frequency, compared to placebo.27 A similar study done in the United States did not show a significant difference between the active-treatment and placebo groups.34
- Findings regarding onabotulinumtoxinA are intriguing: In a posthoc analysis of onabotulinumtoxinA to treat chronic migraine, patients with MOH who did not undergo detoxification had an 8 d/mo greater reduction in headache, compared to placebo.35 However, when compared to placebo in conjunction with detoxification, onabotulinumtoxinA demonstrated no benefit.36
- Newer CGRP antagonist and CGRP receptor antagonist monoclonal antibodies are successful preventive medications that have demonstrated a reduction in acute medication use days per month and headache days per month37; these compounds have not been compared to withdrawal alone.
Reducing the severity and duration of withdrawal symptoms
Withdrawal from overused abortive headache medications can lead to worsening headache, nausea, vomiting, hypotension, tachycardia, sleep disturbances, restlessness, anxiety, and nervousness. Symptoms usually last 2 to 10 days but can persist for as long as 4 weeks; duration of withdrawal symptoms varies with the medication that is being overused. In patients who have used a triptan, for example, mean duration of withdrawal is 4.1 days; ergotamine, 6.7 days; and NSAIDs, 9.5 days.23 Tapered withdrawal is sometimes recommended with opioids and barbiturates to reduce withdrawal symptoms. It is unclear whether starting a preventive medication during withdrawal assists in reducing withdrawal symptoms.38
Bridging therapy to reduce symptoms of withdrawal is often provided despite debatable utility. Available evidence does not favor one agent or method but suggests some strategies that could be helpful:
- A prednisone taper has a potential role during the first 6 days of withdrawal by reducing rebound headache and withdrawal symptoms39; however, oral prednisolone has been shown to have no benefit.40
- Alone, IV methylprednisolone seems not to be of benefit; however, in a retrospective study of 94 patients, IV methylprednisolone plus diazepam for 5 days led to a significant reduction in headache frequency and drug consumption that was sustained after 3 months.41
- Celecoxib was compared to prednisone over a 20-day course: a celecoxib dosage of 400 mg/d for the first 5 days, tapered by 100 mg every 5 days, and an oral prednisone dosage of 75 mg/d for the first 5 days, then tapered every 5 days. Patients taking celecoxib had lower headache intensity but there was no difference in headache frequency and acute medication intake between the groups.42
Other strategies. Using antiemetics and NSAIDs to reduce withdrawal symptoms is widely practiced, but no placebo-controlled trials have been conducted to support this strategy.
Patients in withdrawal might be more likely to benefit from inpatient care if they have a severe comorbidity, such as opioid or barbiturate use; failure to respond to, tolerate, or adhere to treatment; or relapse after withdrawal.38
Continue to: Cognitive behavioral therapy...
Cognitive behavioral therapy, exercise, a headache diary, and biofeedback should be considered in every patient’s treatment strategy because a multidisciplinary approach increases adherence and leads to improvement in headache frequency and a decrease in disability and medication use.43
Predictors of Tx success
A prospective cohort study determined that the rate of MOH relapse is 31% at 6 months, 41% at 1 year, and 45% at 4 years, with the highest risk of relapse during the first year.44 Looking at the correlation between type of medication overused and relapse rate, the research indicates that
- triptans have the lowest risk of relapse,44
- simple analgesics have a higher risk of relapse than triptans,22,44 and
- opioids have the highest risk of relapse.22
Where the data don’t agree. Data on combination analgesics and on ergots are conflicting.22 In addition, data on whether the primary type of headache predicts relapse rate conflict; however, migraine might predict a better outcome than tension-type headache.22
To recap and expand: Management pearls
The major goals of headache management generally are to rule out secondary headache, reach a correct diagnosis, reduce overall headache frequency, and provide effective abortive medication. A large component of reducing headache frequency is addressing and treating medication overuse.
Seek to understand the nature of the patient’s headache disorder. Components of the history are key in identifying the underlying headache diagnosis and ruling out other, more concerning secondary headache diagnoses. The ICHD-3 is an excellent resource for treating headache disorders because the classification lists specific diagnostic criteria for all recognized headache diagnoses.
Continue to: Medication withdrawal...
Medication withdrawal—with or without preventive medication—should reduce the frequency of MOH in 2 or 3 months. If headache does not become less frequent, however, the headache diagnosis might need to be reconsidered. Minimizing the use of abortive medication is generally recommended, but reduction or withdrawal of these medications does not guarantee that patients will revert to an episodic pattern of headache.
Treating withdrawal symptoms is a reasonable approach in some patients, but evidence does not support routinely providing bridging therapy.
Apply preventives carefully. Abortive medication withdrawal should generally be completed before initiating preventive medication; however, over the short term, starting preventive therapy while withdrawing the overused medication could assist in reducing headache frequency rapidly. This strategy can put patients at risk of medication adverse effects and using the medications longer than necessary, yet might be reasonable in certain patients, given their comorbidities, risk of relapse, and physician and patient preference. A preventive medication for an individual patient should generally be chosen in line with recommendations of the American Academy of Neurology45 and on the basis of the history and comorbidities.
Provide education, which is essential to lowering barriers to success. Patients with MOH must be counseled to understand that (1) a headache treatment that is supposed to be making them feel better is, in fact, making them feel worse and (2) they will get worse before they get better. Many patients are afraid to be without medication to use as needed. It is helpful to educate them on the different types of treatments (abortive, preventive); how MOH interferes with headache prophylaxis and medication efficacy; how MOH alters brain function (ie, aforementioned physiologic changes in pain processing and functional imaging changes23); and that such change is reversible when medication is withdrawn.
ACKNOWLEDGEMENT
The author thanks Jeffrey Curtis, MD, MPH, for his support and editing assistance with the manuscript.
CORRESPONDENCE
Allison Crain, MD, 2927 N 7th Avenue, Phoenix, AZ 85013; Allison.Crain@dignityhealth.org.
1. Zeeberg P, Olesen J, Jensen R. Discontinuation of medication overuse in headache patients: recovery of therapeutic responsiveness. Cephalalgia. 2006;26:1192-1198.
2. Kristoffersen ES, Straand J, Vetvik KG, et al. Brief intervention for medication-overuse headache in primary care. The BIMOH study: a double-blind pragmatic cluster randomised parallel controlled trial. J Neurol Neurosurg Psychiatry. 2015;86:505-512.
3. Bahra A, Walsh M, Menon S, et al. Does chronic daily headache arise de novo in association with regular use of analgesics? Headache. 2003;43:179-190.
4. Blumenfeld AM, Varon SF, Wilcox TK, et al. Disability, HRQoL and resource use among chronic and episodic migraineurs: results from the International Burden of Migraine Study (IBMS) Cephalalgia. 2011;31:301-315.
5. Chu H-T, Liang C-S, Lee J-T, et al. Associations between depression/anxiety and headache frequency in migraineurs: a cross-sectional study. Headache. 2018;58:407-415.
6. Bigal ME, Lipton RB. Excessive acute migraine medication use and migraine progression. Neurology. 2008;71:1821-1828.
7. Colás R, Muñoz P, Temprano R, et al. Chronic daily headache with analgesic overuse: epidemiology and impact on quality of life. Neurology. 2004;62:1338-1342.
8. Linde M, Gustavsson A, Stovner LJ, et al. The cost of headache disorders in Europe: the Eurolight project. Eur J Neurol. 2012;19:703-711.
9. Shah AM, Bendtsen L, Zeeberg P, et al. Reduction of medication costs after detoxification for medication-overuse headache. Headache. 2013;53:665-672.
10. . Global, regional, and national burden of migraine and tension-type headache, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018;17:954-976.
11. Kernick D, Stapley S, Goadsby PJ, et al. What happens to new-onset headache presenting to primary care? A case–cohort study using electronic primary care records. Cephalalgia. 2008;28:1188-1195.
12. Stone J, Carson A, Duncan R, et al. Who is referred to neurology clinics?—the diagnoses made in 3781 new patients. Clin Neurol Neurosurg. 2010;112:747-751.
13. Munoz-Ceron J, Marin-Careaga V, L, et al. Headache at the emergency room: etiologies, diagnostic usefulness of the ICHD 3 criteria, red and green flags. PloS One. 2019;14:e0208728.
14. Evers S, Marziniak M. Clinical features, pathophysiology, and treatment of medication-overuse headache. Lancet Neurol. 2010;9:391-401.
15. Tassorelli C, Jensen R, Allena M, et al; the . A consensus protocol for the management of medication-overuse headache: evaluation in a multicentric, multinational study. Cephalalgia. 2014;34:645-655.
16. Bigal ME, Serrano D, Buse D, et al. Acute migraine medications and evolution from episodic to chronic migraine: a longitudinal population-based study. Headache. 2008;48:1157-1168.
17. Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211.
18. Ferrari A, Leone S, Vergoni AV, et al. Similarities and differences between chronic migraine and episodic migraine. Headache. 2007;47:65-72.
19. Hagen K, Linde M, Steiner TJ, et al. Risk factors for medication-overuse headache: an 11-year follow-up study. The Nord-Trøndelag Health Studies. Pain. 2012;153:56-61.
20. Katsarava Z, Schneewiess S, Kurth T, et al. Incidence and predictors for chronicity of headache in patients with episodic migraine. Neurology. 2004;62:788-790.
21. Lipton RB, Fanning KM, Buse DC, et al. Migraine progression in subgroups of migraine based on comorbidities: results of the CaMEO study. Neurology. 2019;93:e2224-e2236.
22. Munksgaard SB, Madsen SK, Wienecke T. Treatment of medication overuse headache—a review. Acta Neurol Scand. 2019;139:405-414.
23. Ferraro S, Grazzi L, Mandelli M, et al. Pain processing in medication overuse headache: a functional magnetic resonance imaging (fMRI) study. Pain Med. 2012;13:255-262.
24. Diener H-C, Holle D, Solbach K, et al. Medication-overuse headache: risk factors, pathophysiology and management. Nat Rev Neurol. 2016;12:575-583.
25. Limmroth V, Katsarava Z, Fritsche G, et al. Features of medication overuse headache following overuse of different acute headache drugs. Neurology. 2002;59:1011-1014.
26. Mauskop A, ed. Migraine and Headache. 2nd ed. Oxford University Press; 2013.
27. Diener H-C, Bussone G, Van Oene JC, et al; . Topiramate reduces headache days in chronic migraine: a randomized, double-blind, placebo-controlled study. Cephalalgia. 2007;27:814-823.
28. Navratilova E, Behravesh S, Oyarzo J, et al. Ubrogepant does not induce latent sensitization in a preclinical model of medication overuse headache Cephalalgia. 2020;40:892-902.
29. Kristoffersen ES, Straand J, Russell MB, et al. Lasting improvement of medication-overuse headache after brief intervention—a long-term follow-up in primary care. Eur J Neurol. 2017;24:883-891.
30. Carlsen LN, Munksgaard SB, Jensen RH, et al. Complete detoxification is the most effective treatment of medication-overuse headache: a randomized controlled open-label trial. Cephalalgia. 2018;38:225-236.
31. Sarchielli P, Messina P, Cupini LM, et al; SAMOHA Study Group. Sodium valproate in migraine without aura and medication overuse headache: a randomized controlled trial. Eur Neuropsychopharmacol. 2014;24:1289-1297.
32. Hagen K, Stovner LJ. A randomized controlled trial on medication-overuse headache: outcome after 1 and 4 years. Acta Neurol Scand Suppl. 2011;124(suppl 191):38-43.
33. Munksgaard SB, Bendtsen L, Jensen RH. Detoxification of medication-overuse headache by a multidisciplinary treatment programme is highly effective: a comparison of two consecutive treatment methods in an open-label design. Cephalalgia. 2012;32:834-844.
34. Silberstein S, Lipton R, Dodick D, et al. Topiramate treatment of chronic migraine: a randomized, placebo-controlled trial of quality of life and other efficacy measures. Headache. 2009;49:1153-1162.
35. Silberstein SD, Blumenfeld AM, Cady RK, et al. OnabotulinumtoxinA for treatment of chronic migraine: PREEMPT 24-week pooled subgroup analysis of patients who had acute headache medication overuse at baseline. J Neurol Sci. 2013;331:48-56.
36. Sandrini G, Perrotta A, Tassorelli C, et al. Botulinum toxin type-A in the prophylactic treatment of medication-overuse headache: a multicenter, double-blind, randomized, placebo-controlled, parallel group study. J Headache Pain. 2011;12:427-433.
37. Tepper SJ. CGRP and headache: a brief review. Neurol Sci. 2019;40(suppl 1):99-105.
38. Diener H-C, Dodick D, Evers S, et al. Pathophysiology, prevention and treatment of medication overuse headache. Lancet Neurol. 2019;18:891-902.
39. Krymchantowski AV, Barbosa JS. Prednisone as initial treatment of analgesic-induced daily headache. Cephalalgia. 2000;20:107-113.
40. Bøe MG, Mygland A, Salvesen R. Prednisolone does not reduce withdrawal headache: a randomized, double-blind study. Neurology. 2007;69:26-31.
41. Paolucci M, Altamura C, Brunelli N, et al. Methylprednisolone plus diazepam i.v. as bridge therapy for medication overuse headache. Neurol Sci. 2017;38:2025-2029.
42. Taghdiri F, Togha M, Razeghi Jahromi S, et al. Celecoxib vs prednisone for the treatment of withdrawal headache in patients with medication overuse headache: a randomized, double-blind clinical trial. Headache. 2015;55:128-135.
43. Ramsey RR, Ryan JL, Hershey AD, et al. Treatment adherence in patients with headache: a systematic review. Headache. 2014;54:795-816.
44. Katsarava Z, Muessig M, Dzagnidze A, et al. Medication overuse headache: rates and predictors for relapse in a 4-year prospective study. Cephalalgia. 2005;25:12-15.
45. Silberstein SD, Holland S, Freitag F, et al; . Evidence-based guideline update: pharmacologic treatment for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology. 2012; 78:1137-1145.
Medication overuse headache (MOH), a secondary headache diagnosis, is a prevalent phenomenon that complicates headache diagnosis and treatment, increases the cost of care, and reduces quality of life. Effective abortive medication is essential for the headache sufferer; when an abortive is used too frequently, however, headache frequency increases—potentially beginning a cycle in which the patient then takes more medication to abort the headache. Over time, the patient suffers from an ever-increasing number of headaches, takes even more abortive medication, and so on. In the presence of MOH, there is a reduction in pain response to preventive and abortive treatments; when medication overuse is eliminated, pain response improves.1
Although MOH is well recognized among headache specialists, the condition is often overlooked in primary care. Since headache is a top complaint in primary care, however, and prevention is a major goal in family medicine, the opportunity for you to recognize, treat, and prevent MOH is significant. In fact, a randomized controlled trial showed that brief patient education about headache care and MOH provided by a primary care physician can lead to a significant reduction in headache frequency among patients with MOH.2
This article reviews the recognition and diagnosis of MOH, based on historical features and current criteria; addresses risk factors for abortive medication overuse and how to withdraw an offending agent; and explores the value of bridging and preventive therapies to reduce the overall frequency of headache.

What defines MOH?
Typically, MOH is a chronification of a primary headache disorder. However, in patients with a history of migraine who are undergoing treatment for another chronic pain condition with an opioid or other analgesic, MOH can be induced.3 An increase in the frequency of headache raises the specter of a concomitant increase in the level of disability4; psychiatric comorbidity5; and more headache days, with time lost from school and work.
The Migraine Disability Assessment (MIDAS) questionnaire, a validated instrument that helps the provider (1) measure the impact that headache has on a patient’s life and (2) follow treatment progress, also provides information to employers and insurance companies on treatment coverage and the need for work modification. The MIDAS score is 3 times higher in patients with MOH than in patients with episodic migraine.6,7
The annual associated cost per person of MOH has been estimated at $4000, resulting in billions of dollars in associated costs8; most of these costs are related to absenteeism and disability. After detoxification for MOH, annual outpatient medication costs are reduced by approximately 24%.9
Efforts to solve a common problem create another
Headache affects nearly 50% of the general population worldwide,10 accounting for about 4% of primary care visits11 and approximately 20% of outpatient neurology consultations.12 Although inpatient stays for headache are approximately half the duration of the overall average hospital stay, headache accounts for 3% of admissions.13 According to the Global Burden of Disease study, tension-type headache, migraine, and MOH are the 3 most common headache disorders.10 Headache is the second leading cause of disability among people 15 to 49 years of age.10
Continue to: The prevalence of MOH...
The prevalence of MOH in the general population is 2%.7,14,15 A population-based study showed that the rate of progression from episodic headache (< 15 d/mo) to chronic headache (≥ 15 d/mo) in the general population is 2.5% per year16; however, progression to chronic headache is 14% per year in patients with medication overuse. One-third of the general population with chronic migraine overuses symptomatic medication; in US headache clinics, roughly one-half of patients with chronic headache overuse acute medication.6
Definitions and diagnosis
MOH is a secondary headache diagnosis in the third edition of the International Classification of Headache Disorders (ICHD-3) (TABLE 1),17 which lists diagnostic criteria for recognized headache disorders.

Terminology. MOH has also been called rebound headache, drug-induced headache, and transformed migraine, but these terms are outdated and are not formal diagnoses. Patients sometimes refer to substance-withdrawal headaches (not discussed in this article) as rebound headaches, so clarity is important when discussing headache with patients: namely, that MOH is an exacerbation of an existing headache condition caused by overuse of abortive headache medications, including analgesics, combination analgesics, triptans, barbiturates, and opioids.
MOH was recognized in the early 1950s and fully differentiated as a diagnosis in 2005 in the second edition of the ICHD. The disorder is subcategorized by offending abortive agent (TABLE 217) because the frequency of analgesic use required to develop MOH differs by agent.

Risk factors for MOH and chronification of a primary headache disorder. There are several risk factors for developing MOH, and others that contribute to increasing headache frequency in general (TABLE 35,14,18-23). Some risk factors are common to each. All are important to address because some are modifiable.

Continue to: Pathophysiology
Pathophysiology. The pathophysiology and psychology behind MOH are largely unknown. Physiologic changes in pain processing and functional imaging changes have been demonstrated in patients with MOH, both of which are reversible upon withdrawal of medication.23 Genetic factors and changes in hormone and neurotransmitter levels are found in MOH patients; this is not the case in patients who have an episodic headache pattern only.24
Presentation. Diagnostic criteria for MOH do not include clinical characteristics. Typically, the phenotype of MOH in a given patient is similar to the underlying primary headache25—although this principle can be complicated to tease out because these medications can suppress some symptoms. Diagnosis of a primary headache disorder should be documented along with the diagnosis of MOH.
Medication overuse can exist without MOH: Not every patient who frequently uses an abortive medication develops MOH.
Treatment is multifaceted—and can become complex
Mainstays of treatment of MOH are education about the disorder and detoxification from the overused agent, although specific treatments can differ depending on the agent involved, the frequency and duration of its use, and a patient’s behavioral patterns and psychiatric comorbidities. Often, a daily medication to prevent headache is considered upon, or after, withdrawal of the offending agent. The timing of introducing a preventive might impact its effectiveness. Some refractory cases require more intensive therapy, including hospitalization at a specialized tertiary center.
But before we look at detoxification from an overused agent, it’s important to review one of the best strategies of all in combatting MOH.
Continue to: First and best strategy
First and best strategy: Avoid onset of MOH
Select an appropriate abortive to reduce the risk of MOH. With regard to specific acute headache medications, some nuances other than type of headache should be considered. Nonsteroidal anti-inflammatory drugs (NSAIDs) are recommended as abortive therapy by the American Headache Society for their efficacy, favorable adverse effect profile, and low cost. NSAIDs are protective against development of MOH if a patient’s baseline headache frequency is < 10/mo; at a frequency of 10 to 14 d/mo, however, the risk of MOH increases when using an NSAID.6 A similar effect has been seen with triptans.16 Longer-acting NSAIDs, such as nabumetone and naproxen, have been proposed as less likely to cause MOH, and are even used as bridging therapy sometimes (as long as neither of these was the overused medication).26
The time it takes to develop MOH is shortest with triptans, followed by ergots, then analgesics.27
Prospective cohort studies6,16 have shown that barbiturates and opioids are more likely to induce MOH; for that reason, agents in these analgesic classes are almost universally avoided unless no other medically acceptable options exist. Using barbiturate-containing compounds or opioids > 4 d/mo exponentially increases the likelihood of MOH.
Promising preclinical data demonstrate that the gepant, or small-molecule calcitonin gene-related peptide (CGRP) receptor antagonist, class of medications used as abortive therapy does not induce medication overuse cutaneous allodynia.28
Provide education. Primary prevention of MOH involves (1) increasing patients’ awareness of how to take medications appropriately and (2) restricting intake of over-the-counter abortive medications. Often, the expert recommendation is to limit abortives to approximately 2 d/wk because more frequent use places patients at risk of further increased use and subsequent MOH.
Continue to: A randomized controlled trial in Norway...
A randomized controlled trial in Norway compared outcomes in 2 groups of patients with MOH: One group was given advice on the disorder by a physician; the other group was not provided with advice. In the “business-as-usual” group, there was no significant improvement; however, when general practitioners provided simple advice (lasting roughly 9 minutes) about reducing abortive medication use to a safe level and cautioned patients that they would be “feeling worse before feeling better,” headache days were reduced by approximately 8 per month and medication days, by 16 per month.2
A subsequent, long-term follow-up study29 of patients from the Norway trial2 who had been given advice and education showed a relapse rate (ie, into overuse of headache medication) of only 8% and sustained reduction of headache days and medication use at 16 months.
Offer support and other nondrug interventions. A recent review of 3 studies23 recommended that extra support for patients from a headache nurse, close follow-up, keeping an electronic diary that provides feedback, and undertaking a short course of psychotherapy can reduce medication overuse and prevent relapse.
If MOH develops, initiate withdrawal, introduce a preventive
Withdraw overused medication. Most current evidence suggests that withdrawal of the offending agent is the most effective factor in reducing headache days and improving quality of life. A randomized controlled trial compared the effects of (1) complete and immediate withdrawal of an abortive medication with (2) reducing its use (ie, limiting intake to 2 d/wk), on headache frequency, disability, and quality of life.30 There was a reduction of headache days in both groups; however, reduction was much greater at 2 months in the complete withdrawal group than in the restricted intake group (respectively, a 41% and a 26% reduction in headache days per month). This effect was sustained at 6 and 12 months in both groups. The study confirmed the results of earlier research2,15: Abrupt withdrawal leads to reversion to an episodic pattern at 2 to 6 months in approximately 40% to 60% of patients.
More studies are needed to determine the most appropriate treatment course for MOH; however, complete withdrawal of the causative drug is the most important intervention.
Continue to: Consider withdrawal plus preventive treatment
Consider withdrawal plus preventive treatment. Use of sodium valproate, in addition to medication overuse detoxification, led to a significant reduction in headache days and improvement in quality of life at 12 weeks but no difference after 24 weeks, compared with detoxification alone in a randomized, double-blind, placebo-controlled study.31
A study of 61 patients showed a larger reduction (by 7.2 d/mo) in headache frequency with any preventive medication in addition to medication withdrawal, compared to withdrawal alone (by 4.1 d/mo) after 3 months; however, the relative benefit was gone at 6 months.32
A study of 98 patients compared immediate and delayed initiation of preventive medication upon withdrawal of overused abortive medication.33 Response was defined as a > 50% reduction in headache frequency and was similar in both groups; results showed a 28% response with immediate initiation of a preventive; a 23% response with delayed (ie, 2 months after withdrawal) initiation; and a 48% response in both groups at 12 months.
Collectively, these studies suggest that adding a preventive medication at the time of withdrawal has the potential to reduce headache frequency more quickly than withdrawal alone. However, after 3 to 6 months, the outcome of reduced headache frequency is the same whether or not a preventive medication is used—as long as the offending agent has been withdrawn.
Do preventives work without withdrawing overused medication? Patients with MOH often show little or no improvement with addition of a preventive medication only; their response to a preventive improves after withdrawal of the overused medication. Patients without previous headache improvement after addition of a preventive, who also did not improve 2 months after withdrawal, then demonstrated an overall reduction in headache by 26% when a preventive was reintroduced after withdrawal.2
Continue to: The research evidence for preventives
The research evidence for preventives. Medications for headache prevention have not been extensively evaluated specifically for treating MOH. Here is what’s known:
- Flunarizine, amitriptyline, and beta-blockers usually are ineffective for MOH.24
- Results for topiramate are mixed: A small, double-blind, placebo-controlled chronic migraine study in Europe showed that, in a subgroup of patients with MOH, topiramate led to a small but significant reduction (3.5 d/mo) in headache frequency, compared to placebo.27 A similar study done in the United States did not show a significant difference between the active-treatment and placebo groups.34
- Findings regarding onabotulinumtoxinA are intriguing: In a posthoc analysis of onabotulinumtoxinA to treat chronic migraine, patients with MOH who did not undergo detoxification had an 8 d/mo greater reduction in headache, compared to placebo.35 However, when compared to placebo in conjunction with detoxification, onabotulinumtoxinA demonstrated no benefit.36
- Newer CGRP antagonist and CGRP receptor antagonist monoclonal antibodies are successful preventive medications that have demonstrated a reduction in acute medication use days per month and headache days per month37; these compounds have not been compared to withdrawal alone.
Reducing the severity and duration of withdrawal symptoms
Withdrawal from overused abortive headache medications can lead to worsening headache, nausea, vomiting, hypotension, tachycardia, sleep disturbances, restlessness, anxiety, and nervousness. Symptoms usually last 2 to 10 days but can persist for as long as 4 weeks; duration of withdrawal symptoms varies with the medication that is being overused. In patients who have used a triptan, for example, mean duration of withdrawal is 4.1 days; ergotamine, 6.7 days; and NSAIDs, 9.5 days.23 Tapered withdrawal is sometimes recommended with opioids and barbiturates to reduce withdrawal symptoms. It is unclear whether starting a preventive medication during withdrawal assists in reducing withdrawal symptoms.38
Bridging therapy to reduce symptoms of withdrawal is often provided despite debatable utility. Available evidence does not favor one agent or method but suggests some strategies that could be helpful:
- A prednisone taper has a potential role during the first 6 days of withdrawal by reducing rebound headache and withdrawal symptoms39; however, oral prednisolone has been shown to have no benefit.40
- Alone, IV methylprednisolone seems not to be of benefit; however, in a retrospective study of 94 patients, IV methylprednisolone plus diazepam for 5 days led to a significant reduction in headache frequency and drug consumption that was sustained after 3 months.41
- Celecoxib was compared to prednisone over a 20-day course: a celecoxib dosage of 400 mg/d for the first 5 days, tapered by 100 mg every 5 days, and an oral prednisone dosage of 75 mg/d for the first 5 days, then tapered every 5 days. Patients taking celecoxib had lower headache intensity but there was no difference in headache frequency and acute medication intake between the groups.42
Other strategies. Using antiemetics and NSAIDs to reduce withdrawal symptoms is widely practiced, but no placebo-controlled trials have been conducted to support this strategy.
Patients in withdrawal might be more likely to benefit from inpatient care if they have a severe comorbidity, such as opioid or barbiturate use; failure to respond to, tolerate, or adhere to treatment; or relapse after withdrawal.38
Continue to: Cognitive behavioral therapy...
Cognitive behavioral therapy, exercise, a headache diary, and biofeedback should be considered in every patient’s treatment strategy because a multidisciplinary approach increases adherence and leads to improvement in headache frequency and a decrease in disability and medication use.43
Predictors of Tx success
A prospective cohort study determined that the rate of MOH relapse is 31% at 6 months, 41% at 1 year, and 45% at 4 years, with the highest risk of relapse during the first year.44 Looking at the correlation between type of medication overused and relapse rate, the research indicates that
- triptans have the lowest risk of relapse,44
- simple analgesics have a higher risk of relapse than triptans,22,44 and
- opioids have the highest risk of relapse.22
Where the data don’t agree. Data on combination analgesics and on ergots are conflicting.22 In addition, data on whether the primary type of headache predicts relapse rate conflict; however, migraine might predict a better outcome than tension-type headache.22
To recap and expand: Management pearls
The major goals of headache management generally are to rule out secondary headache, reach a correct diagnosis, reduce overall headache frequency, and provide effective abortive medication. A large component of reducing headache frequency is addressing and treating medication overuse.
Seek to understand the nature of the patient’s headache disorder. Components of the history are key in identifying the underlying headache diagnosis and ruling out other, more concerning secondary headache diagnoses. The ICHD-3 is an excellent resource for treating headache disorders because the classification lists specific diagnostic criteria for all recognized headache diagnoses.
Continue to: Medication withdrawal...
Medication withdrawal—with or without preventive medication—should reduce the frequency of MOH in 2 or 3 months. If headache does not become less frequent, however, the headache diagnosis might need to be reconsidered. Minimizing the use of abortive medication is generally recommended, but reduction or withdrawal of these medications does not guarantee that patients will revert to an episodic pattern of headache.
Treating withdrawal symptoms is a reasonable approach in some patients, but evidence does not support routinely providing bridging therapy.
Apply preventives carefully. Abortive medication withdrawal should generally be completed before initiating preventive medication; however, over the short term, starting preventive therapy while withdrawing the overused medication could assist in reducing headache frequency rapidly. This strategy can put patients at risk of medication adverse effects and using the medications longer than necessary, yet might be reasonable in certain patients, given their comorbidities, risk of relapse, and physician and patient preference. A preventive medication for an individual patient should generally be chosen in line with recommendations of the American Academy of Neurology45 and on the basis of the history and comorbidities.
Provide education, which is essential to lowering barriers to success. Patients with MOH must be counseled to understand that (1) a headache treatment that is supposed to be making them feel better is, in fact, making them feel worse and (2) they will get worse before they get better. Many patients are afraid to be without medication to use as needed. It is helpful to educate them on the different types of treatments (abortive, preventive); how MOH interferes with headache prophylaxis and medication efficacy; how MOH alters brain function (ie, aforementioned physiologic changes in pain processing and functional imaging changes23); and that such change is reversible when medication is withdrawn.
ACKNOWLEDGEMENT
The author thanks Jeffrey Curtis, MD, MPH, for his support and editing assistance with the manuscript.
CORRESPONDENCE
Allison Crain, MD, 2927 N 7th Avenue, Phoenix, AZ 85013; Allison.Crain@dignityhealth.org.
Medication overuse headache (MOH), a secondary headache diagnosis, is a prevalent phenomenon that complicates headache diagnosis and treatment, increases the cost of care, and reduces quality of life. Effective abortive medication is essential for the headache sufferer; when an abortive is used too frequently, however, headache frequency increases—potentially beginning a cycle in which the patient then takes more medication to abort the headache. Over time, the patient suffers from an ever-increasing number of headaches, takes even more abortive medication, and so on. In the presence of MOH, there is a reduction in pain response to preventive and abortive treatments; when medication overuse is eliminated, pain response improves.1
Although MOH is well recognized among headache specialists, the condition is often overlooked in primary care. Since headache is a top complaint in primary care, however, and prevention is a major goal in family medicine, the opportunity for you to recognize, treat, and prevent MOH is significant. In fact, a randomized controlled trial showed that brief patient education about headache care and MOH provided by a primary care physician can lead to a significant reduction in headache frequency among patients with MOH.2
This article reviews the recognition and diagnosis of MOH, based on historical features and current criteria; addresses risk factors for abortive medication overuse and how to withdraw an offending agent; and explores the value of bridging and preventive therapies to reduce the overall frequency of headache.

What defines MOH?
Typically, MOH is a chronification of a primary headache disorder. However, in patients with a history of migraine who are undergoing treatment for another chronic pain condition with an opioid or other analgesic, MOH can be induced.3 An increase in the frequency of headache raises the specter of a concomitant increase in the level of disability4; psychiatric comorbidity5; and more headache days, with time lost from school and work.
The Migraine Disability Assessment (MIDAS) questionnaire, a validated instrument that helps the provider (1) measure the impact that headache has on a patient’s life and (2) follow treatment progress, also provides information to employers and insurance companies on treatment coverage and the need for work modification. The MIDAS score is 3 times higher in patients with MOH than in patients with episodic migraine.6,7
The annual associated cost per person of MOH has been estimated at $4000, resulting in billions of dollars in associated costs8; most of these costs are related to absenteeism and disability. After detoxification for MOH, annual outpatient medication costs are reduced by approximately 24%.9
Efforts to solve a common problem create another
Headache affects nearly 50% of the general population worldwide,10 accounting for about 4% of primary care visits11 and approximately 20% of outpatient neurology consultations.12 Although inpatient stays for headache are approximately half the duration of the overall average hospital stay, headache accounts for 3% of admissions.13 According to the Global Burden of Disease study, tension-type headache, migraine, and MOH are the 3 most common headache disorders.10 Headache is the second leading cause of disability among people 15 to 49 years of age.10
Continue to: The prevalence of MOH...
The prevalence of MOH in the general population is 2%.7,14,15 A population-based study showed that the rate of progression from episodic headache (< 15 d/mo) to chronic headache (≥ 15 d/mo) in the general population is 2.5% per year16; however, progression to chronic headache is 14% per year in patients with medication overuse. One-third of the general population with chronic migraine overuses symptomatic medication; in US headache clinics, roughly one-half of patients with chronic headache overuse acute medication.6
Definitions and diagnosis
MOH is a secondary headache diagnosis in the third edition of the International Classification of Headache Disorders (ICHD-3) (TABLE 1),17 which lists diagnostic criteria for recognized headache disorders.

Terminology. MOH has also been called rebound headache, drug-induced headache, and transformed migraine, but these terms are outdated and are not formal diagnoses. Patients sometimes refer to substance-withdrawal headaches (not discussed in this article) as rebound headaches, so clarity is important when discussing headache with patients: namely, that MOH is an exacerbation of an existing headache condition caused by overuse of abortive headache medications, including analgesics, combination analgesics, triptans, barbiturates, and opioids.
MOH was recognized in the early 1950s and fully differentiated as a diagnosis in 2005 in the second edition of the ICHD. The disorder is subcategorized by offending abortive agent (TABLE 217) because the frequency of analgesic use required to develop MOH differs by agent.

Risk factors for MOH and chronification of a primary headache disorder. There are several risk factors for developing MOH, and others that contribute to increasing headache frequency in general (TABLE 35,14,18-23). Some risk factors are common to each. All are important to address because some are modifiable.

Continue to: Pathophysiology
Pathophysiology. The pathophysiology and psychology behind MOH are largely unknown. Physiologic changes in pain processing and functional imaging changes have been demonstrated in patients with MOH, both of which are reversible upon withdrawal of medication.23 Genetic factors and changes in hormone and neurotransmitter levels are found in MOH patients; this is not the case in patients who have an episodic headache pattern only.24
Presentation. Diagnostic criteria for MOH do not include clinical characteristics. Typically, the phenotype of MOH in a given patient is similar to the underlying primary headache25—although this principle can be complicated to tease out because these medications can suppress some symptoms. Diagnosis of a primary headache disorder should be documented along with the diagnosis of MOH.
Medication overuse can exist without MOH: Not every patient who frequently uses an abortive medication develops MOH.
Treatment is multifaceted—and can become complex
Mainstays of treatment of MOH are education about the disorder and detoxification from the overused agent, although specific treatments can differ depending on the agent involved, the frequency and duration of its use, and a patient’s behavioral patterns and psychiatric comorbidities. Often, a daily medication to prevent headache is considered upon, or after, withdrawal of the offending agent. The timing of introducing a preventive might impact its effectiveness. Some refractory cases require more intensive therapy, including hospitalization at a specialized tertiary center.
But before we look at detoxification from an overused agent, it’s important to review one of the best strategies of all in combatting MOH.
Continue to: First and best strategy
First and best strategy: Avoid onset of MOH
Select an appropriate abortive to reduce the risk of MOH. With regard to specific acute headache medications, some nuances other than type of headache should be considered. Nonsteroidal anti-inflammatory drugs (NSAIDs) are recommended as abortive therapy by the American Headache Society for their efficacy, favorable adverse effect profile, and low cost. NSAIDs are protective against development of MOH if a patient’s baseline headache frequency is < 10/mo; at a frequency of 10 to 14 d/mo, however, the risk of MOH increases when using an NSAID.6 A similar effect has been seen with triptans.16 Longer-acting NSAIDs, such as nabumetone and naproxen, have been proposed as less likely to cause MOH, and are even used as bridging therapy sometimes (as long as neither of these was the overused medication).26
The time it takes to develop MOH is shortest with triptans, followed by ergots, then analgesics.27
Prospective cohort studies6,16 have shown that barbiturates and opioids are more likely to induce MOH; for that reason, agents in these analgesic classes are almost universally avoided unless no other medically acceptable options exist. Using barbiturate-containing compounds or opioids > 4 d/mo exponentially increases the likelihood of MOH.
Promising preclinical data demonstrate that the gepant, or small-molecule calcitonin gene-related peptide (CGRP) receptor antagonist, class of medications used as abortive therapy does not induce medication overuse cutaneous allodynia.28
Provide education. Primary prevention of MOH involves (1) increasing patients’ awareness of how to take medications appropriately and (2) restricting intake of over-the-counter abortive medications. Often, the expert recommendation is to limit abortives to approximately 2 d/wk because more frequent use places patients at risk of further increased use and subsequent MOH.
Continue to: A randomized controlled trial in Norway...
A randomized controlled trial in Norway compared outcomes in 2 groups of patients with MOH: One group was given advice on the disorder by a physician; the other group was not provided with advice. In the “business-as-usual” group, there was no significant improvement; however, when general practitioners provided simple advice (lasting roughly 9 minutes) about reducing abortive medication use to a safe level and cautioned patients that they would be “feeling worse before feeling better,” headache days were reduced by approximately 8 per month and medication days, by 16 per month.2
A subsequent, long-term follow-up study29 of patients from the Norway trial2 who had been given advice and education showed a relapse rate (ie, into overuse of headache medication) of only 8% and sustained reduction of headache days and medication use at 16 months.
Offer support and other nondrug interventions. A recent review of 3 studies23 recommended that extra support for patients from a headache nurse, close follow-up, keeping an electronic diary that provides feedback, and undertaking a short course of psychotherapy can reduce medication overuse and prevent relapse.
If MOH develops, initiate withdrawal, introduce a preventive
Withdraw overused medication. Most current evidence suggests that withdrawal of the offending agent is the most effective factor in reducing headache days and improving quality of life. A randomized controlled trial compared the effects of (1) complete and immediate withdrawal of an abortive medication with (2) reducing its use (ie, limiting intake to 2 d/wk), on headache frequency, disability, and quality of life.30 There was a reduction of headache days in both groups; however, reduction was much greater at 2 months in the complete withdrawal group than in the restricted intake group (respectively, a 41% and a 26% reduction in headache days per month). This effect was sustained at 6 and 12 months in both groups. The study confirmed the results of earlier research2,15: Abrupt withdrawal leads to reversion to an episodic pattern at 2 to 6 months in approximately 40% to 60% of patients.
More studies are needed to determine the most appropriate treatment course for MOH; however, complete withdrawal of the causative drug is the most important intervention.
Continue to: Consider withdrawal plus preventive treatment
Consider withdrawal plus preventive treatment. Use of sodium valproate, in addition to medication overuse detoxification, led to a significant reduction in headache days and improvement in quality of life at 12 weeks but no difference after 24 weeks, compared with detoxification alone in a randomized, double-blind, placebo-controlled study.31
A study of 61 patients showed a larger reduction (by 7.2 d/mo) in headache frequency with any preventive medication in addition to medication withdrawal, compared to withdrawal alone (by 4.1 d/mo) after 3 months; however, the relative benefit was gone at 6 months.32
A study of 98 patients compared immediate and delayed initiation of preventive medication upon withdrawal of overused abortive medication.33 Response was defined as a > 50% reduction in headache frequency and was similar in both groups; results showed a 28% response with immediate initiation of a preventive; a 23% response with delayed (ie, 2 months after withdrawal) initiation; and a 48% response in both groups at 12 months.
Collectively, these studies suggest that adding a preventive medication at the time of withdrawal has the potential to reduce headache frequency more quickly than withdrawal alone. However, after 3 to 6 months, the outcome of reduced headache frequency is the same whether or not a preventive medication is used—as long as the offending agent has been withdrawn.
Do preventives work without withdrawing overused medication? Patients with MOH often show little or no improvement with addition of a preventive medication only; their response to a preventive improves after withdrawal of the overused medication. Patients without previous headache improvement after addition of a preventive, who also did not improve 2 months after withdrawal, then demonstrated an overall reduction in headache by 26% when a preventive was reintroduced after withdrawal.2
Continue to: The research evidence for preventives
The research evidence for preventives. Medications for headache prevention have not been extensively evaluated specifically for treating MOH. Here is what’s known:
- Flunarizine, amitriptyline, and beta-blockers usually are ineffective for MOH.24
- Results for topiramate are mixed: A small, double-blind, placebo-controlled chronic migraine study in Europe showed that, in a subgroup of patients with MOH, topiramate led to a small but significant reduction (3.5 d/mo) in headache frequency, compared to placebo.27 A similar study done in the United States did not show a significant difference between the active-treatment and placebo groups.34
- Findings regarding onabotulinumtoxinA are intriguing: In a posthoc analysis of onabotulinumtoxinA to treat chronic migraine, patients with MOH who did not undergo detoxification had an 8 d/mo greater reduction in headache, compared to placebo.35 However, when compared to placebo in conjunction with detoxification, onabotulinumtoxinA demonstrated no benefit.36
- Newer CGRP antagonist and CGRP receptor antagonist monoclonal antibodies are successful preventive medications that have demonstrated a reduction in acute medication use days per month and headache days per month37; these compounds have not been compared to withdrawal alone.
Reducing the severity and duration of withdrawal symptoms
Withdrawal from overused abortive headache medications can lead to worsening headache, nausea, vomiting, hypotension, tachycardia, sleep disturbances, restlessness, anxiety, and nervousness. Symptoms usually last 2 to 10 days but can persist for as long as 4 weeks; duration of withdrawal symptoms varies with the medication that is being overused. In patients who have used a triptan, for example, mean duration of withdrawal is 4.1 days; ergotamine, 6.7 days; and NSAIDs, 9.5 days.23 Tapered withdrawal is sometimes recommended with opioids and barbiturates to reduce withdrawal symptoms. It is unclear whether starting a preventive medication during withdrawal assists in reducing withdrawal symptoms.38
Bridging therapy to reduce symptoms of withdrawal is often provided despite debatable utility. Available evidence does not favor one agent or method but suggests some strategies that could be helpful:
- A prednisone taper has a potential role during the first 6 days of withdrawal by reducing rebound headache and withdrawal symptoms39; however, oral prednisolone has been shown to have no benefit.40
- Alone, IV methylprednisolone seems not to be of benefit; however, in a retrospective study of 94 patients, IV methylprednisolone plus diazepam for 5 days led to a significant reduction in headache frequency and drug consumption that was sustained after 3 months.41
- Celecoxib was compared to prednisone over a 20-day course: a celecoxib dosage of 400 mg/d for the first 5 days, tapered by 100 mg every 5 days, and an oral prednisone dosage of 75 mg/d for the first 5 days, then tapered every 5 days. Patients taking celecoxib had lower headache intensity but there was no difference in headache frequency and acute medication intake between the groups.42
Other strategies. Using antiemetics and NSAIDs to reduce withdrawal symptoms is widely practiced, but no placebo-controlled trials have been conducted to support this strategy.
Patients in withdrawal might be more likely to benefit from inpatient care if they have a severe comorbidity, such as opioid or barbiturate use; failure to respond to, tolerate, or adhere to treatment; or relapse after withdrawal.38
Continue to: Cognitive behavioral therapy...
Cognitive behavioral therapy, exercise, a headache diary, and biofeedback should be considered in every patient’s treatment strategy because a multidisciplinary approach increases adherence and leads to improvement in headache frequency and a decrease in disability and medication use.43
Predictors of Tx success
A prospective cohort study determined that the rate of MOH relapse is 31% at 6 months, 41% at 1 year, and 45% at 4 years, with the highest risk of relapse during the first year.44 Looking at the correlation between type of medication overused and relapse rate, the research indicates that
- triptans have the lowest risk of relapse,44
- simple analgesics have a higher risk of relapse than triptans,22,44 and
- opioids have the highest risk of relapse.22
Where the data don’t agree. Data on combination analgesics and on ergots are conflicting.22 In addition, data on whether the primary type of headache predicts relapse rate conflict; however, migraine might predict a better outcome than tension-type headache.22
To recap and expand: Management pearls
The major goals of headache management generally are to rule out secondary headache, reach a correct diagnosis, reduce overall headache frequency, and provide effective abortive medication. A large component of reducing headache frequency is addressing and treating medication overuse.
Seek to understand the nature of the patient’s headache disorder. Components of the history are key in identifying the underlying headache diagnosis and ruling out other, more concerning secondary headache diagnoses. The ICHD-3 is an excellent resource for treating headache disorders because the classification lists specific diagnostic criteria for all recognized headache diagnoses.
Continue to: Medication withdrawal...
Medication withdrawal—with or without preventive medication—should reduce the frequency of MOH in 2 or 3 months. If headache does not become less frequent, however, the headache diagnosis might need to be reconsidered. Minimizing the use of abortive medication is generally recommended, but reduction or withdrawal of these medications does not guarantee that patients will revert to an episodic pattern of headache.
Treating withdrawal symptoms is a reasonable approach in some patients, but evidence does not support routinely providing bridging therapy.
Apply preventives carefully. Abortive medication withdrawal should generally be completed before initiating preventive medication; however, over the short term, starting preventive therapy while withdrawing the overused medication could assist in reducing headache frequency rapidly. This strategy can put patients at risk of medication adverse effects and using the medications longer than necessary, yet might be reasonable in certain patients, given their comorbidities, risk of relapse, and physician and patient preference. A preventive medication for an individual patient should generally be chosen in line with recommendations of the American Academy of Neurology45 and on the basis of the history and comorbidities.
Provide education, which is essential to lowering barriers to success. Patients with MOH must be counseled to understand that (1) a headache treatment that is supposed to be making them feel better is, in fact, making them feel worse and (2) they will get worse before they get better. Many patients are afraid to be without medication to use as needed. It is helpful to educate them on the different types of treatments (abortive, preventive); how MOH interferes with headache prophylaxis and medication efficacy; how MOH alters brain function (ie, aforementioned physiologic changes in pain processing and functional imaging changes23); and that such change is reversible when medication is withdrawn.
ACKNOWLEDGEMENT
The author thanks Jeffrey Curtis, MD, MPH, for his support and editing assistance with the manuscript.
CORRESPONDENCE
Allison Crain, MD, 2927 N 7th Avenue, Phoenix, AZ 85013; Allison.Crain@dignityhealth.org.
1. Zeeberg P, Olesen J, Jensen R. Discontinuation of medication overuse in headache patients: recovery of therapeutic responsiveness. Cephalalgia. 2006;26:1192-1198.
2. Kristoffersen ES, Straand J, Vetvik KG, et al. Brief intervention for medication-overuse headache in primary care. The BIMOH study: a double-blind pragmatic cluster randomised parallel controlled trial. J Neurol Neurosurg Psychiatry. 2015;86:505-512.
3. Bahra A, Walsh M, Menon S, et al. Does chronic daily headache arise de novo in association with regular use of analgesics? Headache. 2003;43:179-190.
4. Blumenfeld AM, Varon SF, Wilcox TK, et al. Disability, HRQoL and resource use among chronic and episodic migraineurs: results from the International Burden of Migraine Study (IBMS) Cephalalgia. 2011;31:301-315.
5. Chu H-T, Liang C-S, Lee J-T, et al. Associations between depression/anxiety and headache frequency in migraineurs: a cross-sectional study. Headache. 2018;58:407-415.
6. Bigal ME, Lipton RB. Excessive acute migraine medication use and migraine progression. Neurology. 2008;71:1821-1828.
7. Colás R, Muñoz P, Temprano R, et al. Chronic daily headache with analgesic overuse: epidemiology and impact on quality of life. Neurology. 2004;62:1338-1342.
8. Linde M, Gustavsson A, Stovner LJ, et al. The cost of headache disorders in Europe: the Eurolight project. Eur J Neurol. 2012;19:703-711.
9. Shah AM, Bendtsen L, Zeeberg P, et al. Reduction of medication costs after detoxification for medication-overuse headache. Headache. 2013;53:665-672.
10. . Global, regional, and national burden of migraine and tension-type headache, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018;17:954-976.
11. Kernick D, Stapley S, Goadsby PJ, et al. What happens to new-onset headache presenting to primary care? A case–cohort study using electronic primary care records. Cephalalgia. 2008;28:1188-1195.
12. Stone J, Carson A, Duncan R, et al. Who is referred to neurology clinics?—the diagnoses made in 3781 new patients. Clin Neurol Neurosurg. 2010;112:747-751.
13. Munoz-Ceron J, Marin-Careaga V, L, et al. Headache at the emergency room: etiologies, diagnostic usefulness of the ICHD 3 criteria, red and green flags. PloS One. 2019;14:e0208728.
14. Evers S, Marziniak M. Clinical features, pathophysiology, and treatment of medication-overuse headache. Lancet Neurol. 2010;9:391-401.
15. Tassorelli C, Jensen R, Allena M, et al; the . A consensus protocol for the management of medication-overuse headache: evaluation in a multicentric, multinational study. Cephalalgia. 2014;34:645-655.
16. Bigal ME, Serrano D, Buse D, et al. Acute migraine medications and evolution from episodic to chronic migraine: a longitudinal population-based study. Headache. 2008;48:1157-1168.
17. Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211.
18. Ferrari A, Leone S, Vergoni AV, et al. Similarities and differences between chronic migraine and episodic migraine. Headache. 2007;47:65-72.
19. Hagen K, Linde M, Steiner TJ, et al. Risk factors for medication-overuse headache: an 11-year follow-up study. The Nord-Trøndelag Health Studies. Pain. 2012;153:56-61.
20. Katsarava Z, Schneewiess S, Kurth T, et al. Incidence and predictors for chronicity of headache in patients with episodic migraine. Neurology. 2004;62:788-790.
21. Lipton RB, Fanning KM, Buse DC, et al. Migraine progression in subgroups of migraine based on comorbidities: results of the CaMEO study. Neurology. 2019;93:e2224-e2236.
22. Munksgaard SB, Madsen SK, Wienecke T. Treatment of medication overuse headache—a review. Acta Neurol Scand. 2019;139:405-414.
23. Ferraro S, Grazzi L, Mandelli M, et al. Pain processing in medication overuse headache: a functional magnetic resonance imaging (fMRI) study. Pain Med. 2012;13:255-262.
24. Diener H-C, Holle D, Solbach K, et al. Medication-overuse headache: risk factors, pathophysiology and management. Nat Rev Neurol. 2016;12:575-583.
25. Limmroth V, Katsarava Z, Fritsche G, et al. Features of medication overuse headache following overuse of different acute headache drugs. Neurology. 2002;59:1011-1014.
26. Mauskop A, ed. Migraine and Headache. 2nd ed. Oxford University Press; 2013.
27. Diener H-C, Bussone G, Van Oene JC, et al; . Topiramate reduces headache days in chronic migraine: a randomized, double-blind, placebo-controlled study. Cephalalgia. 2007;27:814-823.
28. Navratilova E, Behravesh S, Oyarzo J, et al. Ubrogepant does not induce latent sensitization in a preclinical model of medication overuse headache Cephalalgia. 2020;40:892-902.
29. Kristoffersen ES, Straand J, Russell MB, et al. Lasting improvement of medication-overuse headache after brief intervention—a long-term follow-up in primary care. Eur J Neurol. 2017;24:883-891.
30. Carlsen LN, Munksgaard SB, Jensen RH, et al. Complete detoxification is the most effective treatment of medication-overuse headache: a randomized controlled open-label trial. Cephalalgia. 2018;38:225-236.
31. Sarchielli P, Messina P, Cupini LM, et al; SAMOHA Study Group. Sodium valproate in migraine without aura and medication overuse headache: a randomized controlled trial. Eur Neuropsychopharmacol. 2014;24:1289-1297.
32. Hagen K, Stovner LJ. A randomized controlled trial on medication-overuse headache: outcome after 1 and 4 years. Acta Neurol Scand Suppl. 2011;124(suppl 191):38-43.
33. Munksgaard SB, Bendtsen L, Jensen RH. Detoxification of medication-overuse headache by a multidisciplinary treatment programme is highly effective: a comparison of two consecutive treatment methods in an open-label design. Cephalalgia. 2012;32:834-844.
34. Silberstein S, Lipton R, Dodick D, et al. Topiramate treatment of chronic migraine: a randomized, placebo-controlled trial of quality of life and other efficacy measures. Headache. 2009;49:1153-1162.
35. Silberstein SD, Blumenfeld AM, Cady RK, et al. OnabotulinumtoxinA for treatment of chronic migraine: PREEMPT 24-week pooled subgroup analysis of patients who had acute headache medication overuse at baseline. J Neurol Sci. 2013;331:48-56.
36. Sandrini G, Perrotta A, Tassorelli C, et al. Botulinum toxin type-A in the prophylactic treatment of medication-overuse headache: a multicenter, double-blind, randomized, placebo-controlled, parallel group study. J Headache Pain. 2011;12:427-433.
37. Tepper SJ. CGRP and headache: a brief review. Neurol Sci. 2019;40(suppl 1):99-105.
38. Diener H-C, Dodick D, Evers S, et al. Pathophysiology, prevention and treatment of medication overuse headache. Lancet Neurol. 2019;18:891-902.
39. Krymchantowski AV, Barbosa JS. Prednisone as initial treatment of analgesic-induced daily headache. Cephalalgia. 2000;20:107-113.
40. Bøe MG, Mygland A, Salvesen R. Prednisolone does not reduce withdrawal headache: a randomized, double-blind study. Neurology. 2007;69:26-31.
41. Paolucci M, Altamura C, Brunelli N, et al. Methylprednisolone plus diazepam i.v. as bridge therapy for medication overuse headache. Neurol Sci. 2017;38:2025-2029.
42. Taghdiri F, Togha M, Razeghi Jahromi S, et al. Celecoxib vs prednisone for the treatment of withdrawal headache in patients with medication overuse headache: a randomized, double-blind clinical trial. Headache. 2015;55:128-135.
43. Ramsey RR, Ryan JL, Hershey AD, et al. Treatment adherence in patients with headache: a systematic review. Headache. 2014;54:795-816.
44. Katsarava Z, Muessig M, Dzagnidze A, et al. Medication overuse headache: rates and predictors for relapse in a 4-year prospective study. Cephalalgia. 2005;25:12-15.
45. Silberstein SD, Holland S, Freitag F, et al; . Evidence-based guideline update: pharmacologic treatment for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology. 2012; 78:1137-1145.
1. Zeeberg P, Olesen J, Jensen R. Discontinuation of medication overuse in headache patients: recovery of therapeutic responsiveness. Cephalalgia. 2006;26:1192-1198.
2. Kristoffersen ES, Straand J, Vetvik KG, et al. Brief intervention for medication-overuse headache in primary care. The BIMOH study: a double-blind pragmatic cluster randomised parallel controlled trial. J Neurol Neurosurg Psychiatry. 2015;86:505-512.
3. Bahra A, Walsh M, Menon S, et al. Does chronic daily headache arise de novo in association with regular use of analgesics? Headache. 2003;43:179-190.
4. Blumenfeld AM, Varon SF, Wilcox TK, et al. Disability, HRQoL and resource use among chronic and episodic migraineurs: results from the International Burden of Migraine Study (IBMS) Cephalalgia. 2011;31:301-315.
5. Chu H-T, Liang C-S, Lee J-T, et al. Associations between depression/anxiety and headache frequency in migraineurs: a cross-sectional study. Headache. 2018;58:407-415.
6. Bigal ME, Lipton RB. Excessive acute migraine medication use and migraine progression. Neurology. 2008;71:1821-1828.
7. Colás R, Muñoz P, Temprano R, et al. Chronic daily headache with analgesic overuse: epidemiology and impact on quality of life. Neurology. 2004;62:1338-1342.
8. Linde M, Gustavsson A, Stovner LJ, et al. The cost of headache disorders in Europe: the Eurolight project. Eur J Neurol. 2012;19:703-711.
9. Shah AM, Bendtsen L, Zeeberg P, et al. Reduction of medication costs after detoxification for medication-overuse headache. Headache. 2013;53:665-672.
10. . Global, regional, and national burden of migraine and tension-type headache, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018;17:954-976.
11. Kernick D, Stapley S, Goadsby PJ, et al. What happens to new-onset headache presenting to primary care? A case–cohort study using electronic primary care records. Cephalalgia. 2008;28:1188-1195.
12. Stone J, Carson A, Duncan R, et al. Who is referred to neurology clinics?—the diagnoses made in 3781 new patients. Clin Neurol Neurosurg. 2010;112:747-751.
13. Munoz-Ceron J, Marin-Careaga V, L, et al. Headache at the emergency room: etiologies, diagnostic usefulness of the ICHD 3 criteria, red and green flags. PloS One. 2019;14:e0208728.
14. Evers S, Marziniak M. Clinical features, pathophysiology, and treatment of medication-overuse headache. Lancet Neurol. 2010;9:391-401.
15. Tassorelli C, Jensen R, Allena M, et al; the . A consensus protocol for the management of medication-overuse headache: evaluation in a multicentric, multinational study. Cephalalgia. 2014;34:645-655.
16. Bigal ME, Serrano D, Buse D, et al. Acute migraine medications and evolution from episodic to chronic migraine: a longitudinal population-based study. Headache. 2008;48:1157-1168.
17. Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211.
18. Ferrari A, Leone S, Vergoni AV, et al. Similarities and differences between chronic migraine and episodic migraine. Headache. 2007;47:65-72.
19. Hagen K, Linde M, Steiner TJ, et al. Risk factors for medication-overuse headache: an 11-year follow-up study. The Nord-Trøndelag Health Studies. Pain. 2012;153:56-61.
20. Katsarava Z, Schneewiess S, Kurth T, et al. Incidence and predictors for chronicity of headache in patients with episodic migraine. Neurology. 2004;62:788-790.
21. Lipton RB, Fanning KM, Buse DC, et al. Migraine progression in subgroups of migraine based on comorbidities: results of the CaMEO study. Neurology. 2019;93:e2224-e2236.
22. Munksgaard SB, Madsen SK, Wienecke T. Treatment of medication overuse headache—a review. Acta Neurol Scand. 2019;139:405-414.
23. Ferraro S, Grazzi L, Mandelli M, et al. Pain processing in medication overuse headache: a functional magnetic resonance imaging (fMRI) study. Pain Med. 2012;13:255-262.
24. Diener H-C, Holle D, Solbach K, et al. Medication-overuse headache: risk factors, pathophysiology and management. Nat Rev Neurol. 2016;12:575-583.
25. Limmroth V, Katsarava Z, Fritsche G, et al. Features of medication overuse headache following overuse of different acute headache drugs. Neurology. 2002;59:1011-1014.
26. Mauskop A, ed. Migraine and Headache. 2nd ed. Oxford University Press; 2013.
27. Diener H-C, Bussone G, Van Oene JC, et al; . Topiramate reduces headache days in chronic migraine: a randomized, double-blind, placebo-controlled study. Cephalalgia. 2007;27:814-823.
28. Navratilova E, Behravesh S, Oyarzo J, et al. Ubrogepant does not induce latent sensitization in a preclinical model of medication overuse headache Cephalalgia. 2020;40:892-902.
29. Kristoffersen ES, Straand J, Russell MB, et al. Lasting improvement of medication-overuse headache after brief intervention—a long-term follow-up in primary care. Eur J Neurol. 2017;24:883-891.
30. Carlsen LN, Munksgaard SB, Jensen RH, et al. Complete detoxification is the most effective treatment of medication-overuse headache: a randomized controlled open-label trial. Cephalalgia. 2018;38:225-236.
31. Sarchielli P, Messina P, Cupini LM, et al; SAMOHA Study Group. Sodium valproate in migraine without aura and medication overuse headache: a randomized controlled trial. Eur Neuropsychopharmacol. 2014;24:1289-1297.
32. Hagen K, Stovner LJ. A randomized controlled trial on medication-overuse headache: outcome after 1 and 4 years. Acta Neurol Scand Suppl. 2011;124(suppl 191):38-43.
33. Munksgaard SB, Bendtsen L, Jensen RH. Detoxification of medication-overuse headache by a multidisciplinary treatment programme is highly effective: a comparison of two consecutive treatment methods in an open-label design. Cephalalgia. 2012;32:834-844.
34. Silberstein S, Lipton R, Dodick D, et al. Topiramate treatment of chronic migraine: a randomized, placebo-controlled trial of quality of life and other efficacy measures. Headache. 2009;49:1153-1162.
35. Silberstein SD, Blumenfeld AM, Cady RK, et al. OnabotulinumtoxinA for treatment of chronic migraine: PREEMPT 24-week pooled subgroup analysis of patients who had acute headache medication overuse at baseline. J Neurol Sci. 2013;331:48-56.
36. Sandrini G, Perrotta A, Tassorelli C, et al. Botulinum toxin type-A in the prophylactic treatment of medication-overuse headache: a multicenter, double-blind, randomized, placebo-controlled, parallel group study. J Headache Pain. 2011;12:427-433.
37. Tepper SJ. CGRP and headache: a brief review. Neurol Sci. 2019;40(suppl 1):99-105.
38. Diener H-C, Dodick D, Evers S, et al. Pathophysiology, prevention and treatment of medication overuse headache. Lancet Neurol. 2019;18:891-902.
39. Krymchantowski AV, Barbosa JS. Prednisone as initial treatment of analgesic-induced daily headache. Cephalalgia. 2000;20:107-113.
40. Bøe MG, Mygland A, Salvesen R. Prednisolone does not reduce withdrawal headache: a randomized, double-blind study. Neurology. 2007;69:26-31.
41. Paolucci M, Altamura C, Brunelli N, et al. Methylprednisolone plus diazepam i.v. as bridge therapy for medication overuse headache. Neurol Sci. 2017;38:2025-2029.
42. Taghdiri F, Togha M, Razeghi Jahromi S, et al. Celecoxib vs prednisone for the treatment of withdrawal headache in patients with medication overuse headache: a randomized, double-blind clinical trial. Headache. 2015;55:128-135.
43. Ramsey RR, Ryan JL, Hershey AD, et al. Treatment adherence in patients with headache: a systematic review. Headache. 2014;54:795-816.
44. Katsarava Z, Muessig M, Dzagnidze A, et al. Medication overuse headache: rates and predictors for relapse in a 4-year prospective study. Cephalalgia. 2005;25:12-15.
45. Silberstein SD, Holland S, Freitag F, et al; . Evidence-based guideline update: pharmacologic treatment for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology. 2012; 78:1137-1145.
PRACTICE RECOMMENDATIONS
› Avoid prescribing barbiturates or opioids for a headache disorder. A
› Limit use of a headache-abortive medication to twice a week when starting a patient on the drug. C
› Consider providing bridging therapy during detoxification of the overused medication. C
› Do not provide a preventive medication without withdrawing the overused agent. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series