User login
FDA approves first targeted treatment for rare DMD mutation
, the agency has announced.
This particular mutation of the DMD gene “is amenable to exon 45 skipping,” the FDA noted in a press release. The agency added that this is its first approval of a targeted treatment for patients with the mutation.
“Developing drugs designed for patients with specific mutations is a critical part of personalized medicine,” Eric Bastings, MD, deputy director of the Office of Neuroscience at the FDA’s Center for Drug Evaluation and Research, said in a statement.
The approval was based on results from a 43-person randomized controlled trial. Patients who received casimersen had a greater increase in production of the muscle-fiber protein dystrophin compared with their counterparts who received placebo.
Approved – with cautions
The FDA noted that DMD prevalence worldwide is about 1 in 3,600 boys – although it can also affect girls in rare cases. Symptoms of the disorder are commonly first observed around age 3 years but worsen steadily over time. DMD gene mutations lead to a decrease in dystrophin.
As reported by Medscape Medical News in August, the FDA approved viltolarsen (Viltepso, NS Pharma) for the treatment of DMD in patients with a confirmed mutation amenable to exon 53 skipping, following approval of golodirsen injection (Vyondys 53, Sarepta Therapeutics) for the same indication in December 2019.
The DMD gene mutation that is amenable to exon 45 skipping is present in about 8% of patients with DMD.
The trial that carried weight with the FDA included 43 male participants with DMD aged 7-20 years. All were confirmed to have the exon 45-skipping gene mutation and all were randomly assigned 2:1 to received IV casimersen 30 mg/kg or matching placebo.
Results showed that, between baseline and 48 weeks post treatment, the casimersen group showed a significantly higher increase in levels of dystrophin protein than in the placebo group.
Upper respiratory tract infections, fever, joint and throat pain, headache, and cough were the most common adverse events experienced by the active-treatment group.
Although the clinical studies assessing casimersen did not show any reports of kidney toxicity, the adverse event was observed in some nonclinical studies. Therefore, clinicians should monitor kidney function in any patient receiving this treatment, the FDA recommended.
Overall, “the FDA has concluded that the data submitted by the applicant demonstrated an increase in dystrophin production that is reasonably likely to predict clinical benefit” in this patient population, the agency said in its press release.
However, it noted that definitive clinical benefits such as improved motor function were not “established.”
“In making this decision, the FDA considered the potential risks associated with the drug, the life-threatening and debilitating nature of the disease, and the lack of [other] available therapy,” the agency said.
It added that the manufacturer is currently conducting a multicenter study focused on the safety and efficacy of the drug in ambulatory patients with DMD.
The FDA approved casimersen using its Accelerated Approval pathway, granted Fast Track and Priority Review designations to its applications, and gave the treatment Orphan Drug designation.
A version of this article first appeared on Medscape.com.
, the agency has announced.
This particular mutation of the DMD gene “is amenable to exon 45 skipping,” the FDA noted in a press release. The agency added that this is its first approval of a targeted treatment for patients with the mutation.
“Developing drugs designed for patients with specific mutations is a critical part of personalized medicine,” Eric Bastings, MD, deputy director of the Office of Neuroscience at the FDA’s Center for Drug Evaluation and Research, said in a statement.
The approval was based on results from a 43-person randomized controlled trial. Patients who received casimersen had a greater increase in production of the muscle-fiber protein dystrophin compared with their counterparts who received placebo.
Approved – with cautions
The FDA noted that DMD prevalence worldwide is about 1 in 3,600 boys – although it can also affect girls in rare cases. Symptoms of the disorder are commonly first observed around age 3 years but worsen steadily over time. DMD gene mutations lead to a decrease in dystrophin.
As reported by Medscape Medical News in August, the FDA approved viltolarsen (Viltepso, NS Pharma) for the treatment of DMD in patients with a confirmed mutation amenable to exon 53 skipping, following approval of golodirsen injection (Vyondys 53, Sarepta Therapeutics) for the same indication in December 2019.
The DMD gene mutation that is amenable to exon 45 skipping is present in about 8% of patients with DMD.
The trial that carried weight with the FDA included 43 male participants with DMD aged 7-20 years. All were confirmed to have the exon 45-skipping gene mutation and all were randomly assigned 2:1 to received IV casimersen 30 mg/kg or matching placebo.
Results showed that, between baseline and 48 weeks post treatment, the casimersen group showed a significantly higher increase in levels of dystrophin protein than in the placebo group.
Upper respiratory tract infections, fever, joint and throat pain, headache, and cough were the most common adverse events experienced by the active-treatment group.
Although the clinical studies assessing casimersen did not show any reports of kidney toxicity, the adverse event was observed in some nonclinical studies. Therefore, clinicians should monitor kidney function in any patient receiving this treatment, the FDA recommended.
Overall, “the FDA has concluded that the data submitted by the applicant demonstrated an increase in dystrophin production that is reasonably likely to predict clinical benefit” in this patient population, the agency said in its press release.
However, it noted that definitive clinical benefits such as improved motor function were not “established.”
“In making this decision, the FDA considered the potential risks associated with the drug, the life-threatening and debilitating nature of the disease, and the lack of [other] available therapy,” the agency said.
It added that the manufacturer is currently conducting a multicenter study focused on the safety and efficacy of the drug in ambulatory patients with DMD.
The FDA approved casimersen using its Accelerated Approval pathway, granted Fast Track and Priority Review designations to its applications, and gave the treatment Orphan Drug designation.
A version of this article first appeared on Medscape.com.
, the agency has announced.
This particular mutation of the DMD gene “is amenable to exon 45 skipping,” the FDA noted in a press release. The agency added that this is its first approval of a targeted treatment for patients with the mutation.
“Developing drugs designed for patients with specific mutations is a critical part of personalized medicine,” Eric Bastings, MD, deputy director of the Office of Neuroscience at the FDA’s Center for Drug Evaluation and Research, said in a statement.
The approval was based on results from a 43-person randomized controlled trial. Patients who received casimersen had a greater increase in production of the muscle-fiber protein dystrophin compared with their counterparts who received placebo.
Approved – with cautions
The FDA noted that DMD prevalence worldwide is about 1 in 3,600 boys – although it can also affect girls in rare cases. Symptoms of the disorder are commonly first observed around age 3 years but worsen steadily over time. DMD gene mutations lead to a decrease in dystrophin.
As reported by Medscape Medical News in August, the FDA approved viltolarsen (Viltepso, NS Pharma) for the treatment of DMD in patients with a confirmed mutation amenable to exon 53 skipping, following approval of golodirsen injection (Vyondys 53, Sarepta Therapeutics) for the same indication in December 2019.
The DMD gene mutation that is amenable to exon 45 skipping is present in about 8% of patients with DMD.
The trial that carried weight with the FDA included 43 male participants with DMD aged 7-20 years. All were confirmed to have the exon 45-skipping gene mutation and all were randomly assigned 2:1 to received IV casimersen 30 mg/kg or matching placebo.
Results showed that, between baseline and 48 weeks post treatment, the casimersen group showed a significantly higher increase in levels of dystrophin protein than in the placebo group.
Upper respiratory tract infections, fever, joint and throat pain, headache, and cough were the most common adverse events experienced by the active-treatment group.
Although the clinical studies assessing casimersen did not show any reports of kidney toxicity, the adverse event was observed in some nonclinical studies. Therefore, clinicians should monitor kidney function in any patient receiving this treatment, the FDA recommended.
Overall, “the FDA has concluded that the data submitted by the applicant demonstrated an increase in dystrophin production that is reasonably likely to predict clinical benefit” in this patient population, the agency said in its press release.
However, it noted that definitive clinical benefits such as improved motor function were not “established.”
“In making this decision, the FDA considered the potential risks associated with the drug, the life-threatening and debilitating nature of the disease, and the lack of [other] available therapy,” the agency said.
It added that the manufacturer is currently conducting a multicenter study focused on the safety and efficacy of the drug in ambulatory patients with DMD.
The FDA approved casimersen using its Accelerated Approval pathway, granted Fast Track and Priority Review designations to its applications, and gave the treatment Orphan Drug designation.
A version of this article first appeared on Medscape.com.
Study: Central sleep apnea is common in ticagrelor users post ACS
The prevalence of asymptomatic central sleep apnea after acute coronary syndrome is high and may be associated with the use of ticagrelor, a new study finds.
Prior studies have suggested that ticagrelor is associated with an increased likelihood of central sleep apnea. The drug’s label notes that two respiratory conditions – central sleep apnea and Cheyne-Stokes respiration – are adverse reactions that were identified after the drug’s approval in the United States in 2011. “Because these reactions are reported voluntarily from a population of an unknown size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure,” the label says.
Among 80 patients receiving ticagrelor, 24 had central sleep apnea hypopnea syndrome (CSAHS), whereas of 41 patients not taking ticagrelor, 3 had this condition (30% vs. 7.3%, P = .004), in the new study published online Jan. 20, 2021, in Sleep Medicine. A multivariable analysis included in the paper found that age and ticagrelor administration were the only two factors associated with the occurrence of CSAHS.
Findings are ‘striking’
The different rates of central sleep apnea in the study are striking, but it is not clear that asymptomatic central sleep apnea in patients taking ticagrelor is a concern, Ofer Jacobowitz, MD, PhD, associate professor of otolaryngology at Hofstra University, Hempstead, N.Y, said in an interview.
“Whether this particular drug-induced central sleep apnea is consequential” is an open question, noted Dr. Jacobowitz. “There is no evidence that shows that this is definitely harmful.”
“The different types of central sleep apnea are caused by different mechanisms and this one, we don’t know,” Dr. Jacobwitz added.
Study author continues to prescribe ticagrelor
One of the study authors, Philippe Meurin, MD, said that he continues to prescribe ticagrelor every day and that the side effect is not necessarily important.
It is possible that central sleep apnea may resolve, although further studies would need to examine central sleep apnea over time to establish the duration of the condition, he added. Nevertheless, awareness of the association could have implications for clinical practice, Dr. Meurin said.
Central sleep apnea is rare, and if doctors detect it during a sleep study, they may perform extensive tests to assess for possible neurologic diseases, for example, when the cause may be attributed to the medication, he said. In addition, if a patient who is taking ticagrelor has dyspnea, the presence of central sleep apnea may suggest that dyspnea could be related to the drug, although this possibility needs further study, he noted.
Study included patients with ACS history, but no heart failure
Dr. Meurin, of Centre de Réadaptation Cardiaque de La Brie, Les Grands Prés, Villeneuve-Saint-Denis, France, and colleagues included in their study patients between 1 week and 1 year after acute coronary syndrome who did not have heart failure or a history of sleep apnea.
After an overnight sleep study, they classified patients as normal, as having CSAHS (i.e., an apnea-hypopnea index of 15 or greater, mostly with central sleep apneas), or as having obstructive sleep apnea hypopnea syndrome (OSAHS; i.e., an apnea-hypopnea index of 15 or greater, mostly with obstructive sleep apneas).
The prospective study included 121 consecutive patients between January 2018 and March 2020. Patients had a mean age of 56.8, and 88% were men.
Switching to another P2Y12 inhibitor ‘does not seem appropriate’
“CSAHS could be promoted by the use of ticagrelor, a relatively new drug that modifies the apneic threshold,” the study authors wrote. “Regarding underlying mechanisms, the most probable explanation seems to be increased chemosensitivity to hypercapnia by a direct P2Y12 inhibitory effect on the central nervous system.”
Doctors should not overestimate the severity of the adverse reaction or consider it the same way they do OSASH, they added.
Among patients with acute coronary syndrome in the PLATO study, ticagrelor, compared with clopidogrel, “significantly reduced the rate of death from vascular causes, myocardial infarction, or stroke,” Dr. Meurin and colleagues said. “Because in this study more than 9,000 patients received ticagrelor for 12 months, CSAHS (even if it seems frequent in our study) did not seem to impair the good efficacy/tolerance balance of the drug. Therefore, in asymptomatic CSAHS patients, switching from ticagrelor to another P2Y12 inhibitor does not seem appropriate.”
A recent analysis of data from randomized, controlled trials with ticagrelor did not find excess cases of sleep apnea with the drug. But an asymptomatic adverse event such as central sleep apnea “cannot emerge from a post hoc analysis,” Dr. Meurin and colleagues said.
The analysis of randomized trial data was conducted by Marc S. Sabatine, MD, MPH, chairman of the Thrombolysis in Myocardial Infarction (TIMI) Study Group at Brigham and Women’s Hospital, and coauthors. It was published in JACC: Cardiovascular Interventions in April 2020.
They “used the gold standard for medical evidence (randomized, placebo-controlled trials) and found 158 cases of sleep apnea reported, with absolutely no difference between ticagrelor and placebo,” Dr. Sabatine said in an interview. Their analysis examined clinically overt apnea, he noted.
“It is quite clear that when looking at large numbers in placebo-controlled trials, there is no excess,” Dr. Sabatine said. “Meurin et al. are examining a different outcome: the results of a lab test in what may be entirely asymptomatic patients.”
A randomized trial could confirm the association, he said.
“The association may be real, but also may be play of chance or confounded,” said Dr. Sabatine. “To convince the medical community, the next step would be for the investigators to do a randomized trial and test whether ticagrelor increases the risk of central sleep apnea.”
Dr. Meurin and the study coauthors had no disclosures. The analysis of randomized, controlled trial data by Dr. Sabatine and colleagues was funded by AstraZeneca, which distributes ticagrelor under the trade name Brilinta. Dr. Sabatine has been a consultant for AstraZeneca and received research grants through Brigham and Women’s Hospital from AstraZeneca. He has consulted for and received grants through the hospital from other companies as well. Dr. Jacobowitz had no relevant disclosures.
jremaly@mdedge.com
The prevalence of asymptomatic central sleep apnea after acute coronary syndrome is high and may be associated with the use of ticagrelor, a new study finds.
Prior studies have suggested that ticagrelor is associated with an increased likelihood of central sleep apnea. The drug’s label notes that two respiratory conditions – central sleep apnea and Cheyne-Stokes respiration – are adverse reactions that were identified after the drug’s approval in the United States in 2011. “Because these reactions are reported voluntarily from a population of an unknown size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure,” the label says.
Among 80 patients receiving ticagrelor, 24 had central sleep apnea hypopnea syndrome (CSAHS), whereas of 41 patients not taking ticagrelor, 3 had this condition (30% vs. 7.3%, P = .004), in the new study published online Jan. 20, 2021, in Sleep Medicine. A multivariable analysis included in the paper found that age and ticagrelor administration were the only two factors associated with the occurrence of CSAHS.
Findings are ‘striking’
The different rates of central sleep apnea in the study are striking, but it is not clear that asymptomatic central sleep apnea in patients taking ticagrelor is a concern, Ofer Jacobowitz, MD, PhD, associate professor of otolaryngology at Hofstra University, Hempstead, N.Y, said in an interview.
“Whether this particular drug-induced central sleep apnea is consequential” is an open question, noted Dr. Jacobowitz. “There is no evidence that shows that this is definitely harmful.”
“The different types of central sleep apnea are caused by different mechanisms and this one, we don’t know,” Dr. Jacobwitz added.
Study author continues to prescribe ticagrelor
One of the study authors, Philippe Meurin, MD, said that he continues to prescribe ticagrelor every day and that the side effect is not necessarily important.
It is possible that central sleep apnea may resolve, although further studies would need to examine central sleep apnea over time to establish the duration of the condition, he added. Nevertheless, awareness of the association could have implications for clinical practice, Dr. Meurin said.
Central sleep apnea is rare, and if doctors detect it during a sleep study, they may perform extensive tests to assess for possible neurologic diseases, for example, when the cause may be attributed to the medication, he said. In addition, if a patient who is taking ticagrelor has dyspnea, the presence of central sleep apnea may suggest that dyspnea could be related to the drug, although this possibility needs further study, he noted.
Study included patients with ACS history, but no heart failure
Dr. Meurin, of Centre de Réadaptation Cardiaque de La Brie, Les Grands Prés, Villeneuve-Saint-Denis, France, and colleagues included in their study patients between 1 week and 1 year after acute coronary syndrome who did not have heart failure or a history of sleep apnea.
After an overnight sleep study, they classified patients as normal, as having CSAHS (i.e., an apnea-hypopnea index of 15 or greater, mostly with central sleep apneas), or as having obstructive sleep apnea hypopnea syndrome (OSAHS; i.e., an apnea-hypopnea index of 15 or greater, mostly with obstructive sleep apneas).
The prospective study included 121 consecutive patients between January 2018 and March 2020. Patients had a mean age of 56.8, and 88% were men.
Switching to another P2Y12 inhibitor ‘does not seem appropriate’
“CSAHS could be promoted by the use of ticagrelor, a relatively new drug that modifies the apneic threshold,” the study authors wrote. “Regarding underlying mechanisms, the most probable explanation seems to be increased chemosensitivity to hypercapnia by a direct P2Y12 inhibitory effect on the central nervous system.”
Doctors should not overestimate the severity of the adverse reaction or consider it the same way they do OSASH, they added.
Among patients with acute coronary syndrome in the PLATO study, ticagrelor, compared with clopidogrel, “significantly reduced the rate of death from vascular causes, myocardial infarction, or stroke,” Dr. Meurin and colleagues said. “Because in this study more than 9,000 patients received ticagrelor for 12 months, CSAHS (even if it seems frequent in our study) did not seem to impair the good efficacy/tolerance balance of the drug. Therefore, in asymptomatic CSAHS patients, switching from ticagrelor to another P2Y12 inhibitor does not seem appropriate.”
A recent analysis of data from randomized, controlled trials with ticagrelor did not find excess cases of sleep apnea with the drug. But an asymptomatic adverse event such as central sleep apnea “cannot emerge from a post hoc analysis,” Dr. Meurin and colleagues said.
The analysis of randomized trial data was conducted by Marc S. Sabatine, MD, MPH, chairman of the Thrombolysis in Myocardial Infarction (TIMI) Study Group at Brigham and Women’s Hospital, and coauthors. It was published in JACC: Cardiovascular Interventions in April 2020.
They “used the gold standard for medical evidence (randomized, placebo-controlled trials) and found 158 cases of sleep apnea reported, with absolutely no difference between ticagrelor and placebo,” Dr. Sabatine said in an interview. Their analysis examined clinically overt apnea, he noted.
“It is quite clear that when looking at large numbers in placebo-controlled trials, there is no excess,” Dr. Sabatine said. “Meurin et al. are examining a different outcome: the results of a lab test in what may be entirely asymptomatic patients.”
A randomized trial could confirm the association, he said.
“The association may be real, but also may be play of chance or confounded,” said Dr. Sabatine. “To convince the medical community, the next step would be for the investigators to do a randomized trial and test whether ticagrelor increases the risk of central sleep apnea.”
Dr. Meurin and the study coauthors had no disclosures. The analysis of randomized, controlled trial data by Dr. Sabatine and colleagues was funded by AstraZeneca, which distributes ticagrelor under the trade name Brilinta. Dr. Sabatine has been a consultant for AstraZeneca and received research grants through Brigham and Women’s Hospital from AstraZeneca. He has consulted for and received grants through the hospital from other companies as well. Dr. Jacobowitz had no relevant disclosures.
jremaly@mdedge.com
The prevalence of asymptomatic central sleep apnea after acute coronary syndrome is high and may be associated with the use of ticagrelor, a new study finds.
Prior studies have suggested that ticagrelor is associated with an increased likelihood of central sleep apnea. The drug’s label notes that two respiratory conditions – central sleep apnea and Cheyne-Stokes respiration – are adverse reactions that were identified after the drug’s approval in the United States in 2011. “Because these reactions are reported voluntarily from a population of an unknown size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure,” the label says.
Among 80 patients receiving ticagrelor, 24 had central sleep apnea hypopnea syndrome (CSAHS), whereas of 41 patients not taking ticagrelor, 3 had this condition (30% vs. 7.3%, P = .004), in the new study published online Jan. 20, 2021, in Sleep Medicine. A multivariable analysis included in the paper found that age and ticagrelor administration were the only two factors associated with the occurrence of CSAHS.
Findings are ‘striking’
The different rates of central sleep apnea in the study are striking, but it is not clear that asymptomatic central sleep apnea in patients taking ticagrelor is a concern, Ofer Jacobowitz, MD, PhD, associate professor of otolaryngology at Hofstra University, Hempstead, N.Y, said in an interview.
“Whether this particular drug-induced central sleep apnea is consequential” is an open question, noted Dr. Jacobowitz. “There is no evidence that shows that this is definitely harmful.”
“The different types of central sleep apnea are caused by different mechanisms and this one, we don’t know,” Dr. Jacobwitz added.
Study author continues to prescribe ticagrelor
One of the study authors, Philippe Meurin, MD, said that he continues to prescribe ticagrelor every day and that the side effect is not necessarily important.
It is possible that central sleep apnea may resolve, although further studies would need to examine central sleep apnea over time to establish the duration of the condition, he added. Nevertheless, awareness of the association could have implications for clinical practice, Dr. Meurin said.
Central sleep apnea is rare, and if doctors detect it during a sleep study, they may perform extensive tests to assess for possible neurologic diseases, for example, when the cause may be attributed to the medication, he said. In addition, if a patient who is taking ticagrelor has dyspnea, the presence of central sleep apnea may suggest that dyspnea could be related to the drug, although this possibility needs further study, he noted.
Study included patients with ACS history, but no heart failure
Dr. Meurin, of Centre de Réadaptation Cardiaque de La Brie, Les Grands Prés, Villeneuve-Saint-Denis, France, and colleagues included in their study patients between 1 week and 1 year after acute coronary syndrome who did not have heart failure or a history of sleep apnea.
After an overnight sleep study, they classified patients as normal, as having CSAHS (i.e., an apnea-hypopnea index of 15 or greater, mostly with central sleep apneas), or as having obstructive sleep apnea hypopnea syndrome (OSAHS; i.e., an apnea-hypopnea index of 15 or greater, mostly with obstructive sleep apneas).
The prospective study included 121 consecutive patients between January 2018 and March 2020. Patients had a mean age of 56.8, and 88% were men.
Switching to another P2Y12 inhibitor ‘does not seem appropriate’
“CSAHS could be promoted by the use of ticagrelor, a relatively new drug that modifies the apneic threshold,” the study authors wrote. “Regarding underlying mechanisms, the most probable explanation seems to be increased chemosensitivity to hypercapnia by a direct P2Y12 inhibitory effect on the central nervous system.”
Doctors should not overestimate the severity of the adverse reaction or consider it the same way they do OSASH, they added.
Among patients with acute coronary syndrome in the PLATO study, ticagrelor, compared with clopidogrel, “significantly reduced the rate of death from vascular causes, myocardial infarction, or stroke,” Dr. Meurin and colleagues said. “Because in this study more than 9,000 patients received ticagrelor for 12 months, CSAHS (even if it seems frequent in our study) did not seem to impair the good efficacy/tolerance balance of the drug. Therefore, in asymptomatic CSAHS patients, switching from ticagrelor to another P2Y12 inhibitor does not seem appropriate.”
A recent analysis of data from randomized, controlled trials with ticagrelor did not find excess cases of sleep apnea with the drug. But an asymptomatic adverse event such as central sleep apnea “cannot emerge from a post hoc analysis,” Dr. Meurin and colleagues said.
The analysis of randomized trial data was conducted by Marc S. Sabatine, MD, MPH, chairman of the Thrombolysis in Myocardial Infarction (TIMI) Study Group at Brigham and Women’s Hospital, and coauthors. It was published in JACC: Cardiovascular Interventions in April 2020.
They “used the gold standard for medical evidence (randomized, placebo-controlled trials) and found 158 cases of sleep apnea reported, with absolutely no difference between ticagrelor and placebo,” Dr. Sabatine said in an interview. Their analysis examined clinically overt apnea, he noted.
“It is quite clear that when looking at large numbers in placebo-controlled trials, there is no excess,” Dr. Sabatine said. “Meurin et al. are examining a different outcome: the results of a lab test in what may be entirely asymptomatic patients.”
A randomized trial could confirm the association, he said.
“The association may be real, but also may be play of chance or confounded,” said Dr. Sabatine. “To convince the medical community, the next step would be for the investigators to do a randomized trial and test whether ticagrelor increases the risk of central sleep apnea.”
Dr. Meurin and the study coauthors had no disclosures. The analysis of randomized, controlled trial data by Dr. Sabatine and colleagues was funded by AstraZeneca, which distributes ticagrelor under the trade name Brilinta. Dr. Sabatine has been a consultant for AstraZeneca and received research grants through Brigham and Women’s Hospital from AstraZeneca. He has consulted for and received grants through the hospital from other companies as well. Dr. Jacobowitz had no relevant disclosures.
jremaly@mdedge.com
FROM SLEEP MEDICINE
New data may help intercept head injuries in college football
Novel research from the Concussion Assessment, Research and Education (CARE) Consortium sheds new light on how to effectively reduce the incidence of concussion and head injury exposure in college football.
The study, led by neurotrauma experts Michael McCrea, PhD, and Brian Stemper, PhD, professors of neurosurgery at the Medical College of Wisconsin in Milwaukee, reports data from hundreds of college football players across five seasons and shows
The research also reveals that such injuries occur more often during practices than games.
“We think that with the findings from this paper, there’s a role for everybody to play in reducing injury,” Dr. McCrea said. “We hope these data help inform broad-based policy about practice and preseason training policies in collegiate football. We also think there’s a role for athletic administrators, coaches, and even athletes themselves.”
The study was published online Feb. 1 in JAMA Neurology.
More injuries in preseason
Concussion is one of the most common injuries in football. Beyond these harms are growing concerns that repetitive HIE may increase the risk of long-term neurologic health problems including chronic traumatic encephalopathy (CTE).
The CARE Consortium, which has been conducting research with college athletes across 26 sports and military cadets since 2014, has been interested in multiple facets of concussion and brain trauma.
“We’ve enrolled more than 50,000 athletes and service academy cadets into the consortium over the last 6 years to research all involved aspects including the clinical core, the imaging core, the blood biomarker core, and the genetic core, and we have a head impact measurement core.”
To investigate the pattern of concussion incidence across the football season in college players, the investigators used impact measurement technology across six Division I NCAA football programs participating in the CARE Consortium from 2015 to 2019.
A total of 658 players – all male, mean age 19 years – were fitted with the Head Impact Telemetry System (HITS) sensor arrays in their helmets to measure head impact frequency, location, and magnitude during play.
“This particular study had built-in algorithms that weeded out impacts that were below 10G of linear magnitude, because those have been determined not likely to be real impacts,” Dr. McCrea said.
Across the five seasons studied, 528,684 head impacts recorded met the quality standards for analysis. Players sustained a median of 415 (interquartile range [IQR], 190-727) impacts per season.
Of those, 68 players sustained a diagnosed concussion. In total, 48.5% of concussions occurred during preseason training, despite preseason representing only 20.8% of the football season. Total head injury exposure in the preseason occurred at twice the proportion of the regular season (324.9 vs. 162.4 impacts per team per day; mean difference, 162.6 impacts; 95% confidence interval, 110.9-214.3; P < .001).
“Preseason training often has a much higher intensity to it, in terms of the total hours, the actual training, and the heavy emphasis on full-contact drills like tackling and blocking,” said Dr. McCrea. “Even the volume of players that are participating is greater.”
Results also showed that in each of the five seasons, head injury exposure per athlete was highest in August (preseason) (median, 146.0 impacts; IQR, 63.0-247.8) and lowest in November (median, 80.0 impacts; IQR, 35.0-148.0). In the studied period, 72% of concussions and 66.9% of head injury exposure occurred in practice. Even within the regular season, total head injury exposure in practices was 84.2% higher than in games.
“This incredible dataset we have on head impact measurement also gives us the opportunity to compare it with our other research looking at the correlation between a single head impact and changes in brain structure and function on MRI, on blood biomarkers, giving us the ability to look at the connection between mechanism of effect of injury and recovery from injury,” said Dr. McCrea.
These findings also provide an opportunity to modify approaches to preseason training and football practices to keep players safer, said Dr. McCrea, noting that about half of the variance in head injury exposure is at the level of the individual athlete.
“With this large body of athletes we’ve instrumented, we can look at, for instance, all of the running backs and understand the athlete and what his head injury exposure looks like compared to all other running backs. If we find out that an athlete has a rate of head injury exposure that’s 300% higher than most other players that play the same position, we can take that data directly to the athlete to work on their technique and approach to the game.
“Every researcher wishes that their basic science or their clinical research findings will have some impact on the health and well-being of the population they’re studying. By modifying practices and preseason training, football teams could greatly reduce the risk of injury and exposure for their players, while still maintaining the competitive nature of game play,” he added.
Through a combination of policy and education, similar strategies could be implemented to help prevent concussion and HIE in high school and youth football too, said Dr. McCrea.
‘Shocking’ findings
In an accompanying editorial, Christopher J. Nowinski, PhD, of the Concussion Legacy Foundation, Boston, and Robert C. Cantu, MD, department of neurosurgery, Emerson Hospital, Concord, Massachusetts, said the findings could have significant policy implications and offer a valuable expansion of prior research.
“From 2005 to 2010, studies on college football revealed that about two-thirds of head impacts occurred in practice,” they noted. “We cited this data in 2010 when we proposed to the NFL Players Association that the most effective way to reduce the risks of negative neurological outcomes was to reduce hitting in practice. They agreed, and in 2011 collectively bargained for severe contact limits in practice, with 14 full-contact practices allowed during the 17-week season. Since that rule was implemented, only 18% of NFL concussions have occurred in practice.”
“Against this backdrop, the results of the study by McCrea et al. are shocking,” they added. “It reveals that college football players still experience 72% of their concussions and 67% of their total head injury exposure in practice.”
Even more shocking, noted Dr. Nowinski and Dr. Cantu, is that these numbers are almost certainly an underestimate of the dangers of practice.
“As a former college football player and a former team physician, respectively, we find this situation inexcusable. Concussions in games are inevitable, but concussions in practice are preventable,” they wrote.
“Laudably,” they added “the investigators call on the NCAA and football conferences to explore policy and rule changes to reduce concussion incidence and HIE and to create robust educational offerings to encourage change from coaches and college administrators.”
A version of this article first appeared on Medscape.com.
Novel research from the Concussion Assessment, Research and Education (CARE) Consortium sheds new light on how to effectively reduce the incidence of concussion and head injury exposure in college football.
The study, led by neurotrauma experts Michael McCrea, PhD, and Brian Stemper, PhD, professors of neurosurgery at the Medical College of Wisconsin in Milwaukee, reports data from hundreds of college football players across five seasons and shows
The research also reveals that such injuries occur more often during practices than games.
“We think that with the findings from this paper, there’s a role for everybody to play in reducing injury,” Dr. McCrea said. “We hope these data help inform broad-based policy about practice and preseason training policies in collegiate football. We also think there’s a role for athletic administrators, coaches, and even athletes themselves.”
The study was published online Feb. 1 in JAMA Neurology.
More injuries in preseason
Concussion is one of the most common injuries in football. Beyond these harms are growing concerns that repetitive HIE may increase the risk of long-term neurologic health problems including chronic traumatic encephalopathy (CTE).
The CARE Consortium, which has been conducting research with college athletes across 26 sports and military cadets since 2014, has been interested in multiple facets of concussion and brain trauma.
“We’ve enrolled more than 50,000 athletes and service academy cadets into the consortium over the last 6 years to research all involved aspects including the clinical core, the imaging core, the blood biomarker core, and the genetic core, and we have a head impact measurement core.”
To investigate the pattern of concussion incidence across the football season in college players, the investigators used impact measurement technology across six Division I NCAA football programs participating in the CARE Consortium from 2015 to 2019.
A total of 658 players – all male, mean age 19 years – were fitted with the Head Impact Telemetry System (HITS) sensor arrays in their helmets to measure head impact frequency, location, and magnitude during play.
“This particular study had built-in algorithms that weeded out impacts that were below 10G of linear magnitude, because those have been determined not likely to be real impacts,” Dr. McCrea said.
Across the five seasons studied, 528,684 head impacts recorded met the quality standards for analysis. Players sustained a median of 415 (interquartile range [IQR], 190-727) impacts per season.
Of those, 68 players sustained a diagnosed concussion. In total, 48.5% of concussions occurred during preseason training, despite preseason representing only 20.8% of the football season. Total head injury exposure in the preseason occurred at twice the proportion of the regular season (324.9 vs. 162.4 impacts per team per day; mean difference, 162.6 impacts; 95% confidence interval, 110.9-214.3; P < .001).
“Preseason training often has a much higher intensity to it, in terms of the total hours, the actual training, and the heavy emphasis on full-contact drills like tackling and blocking,” said Dr. McCrea. “Even the volume of players that are participating is greater.”
Results also showed that in each of the five seasons, head injury exposure per athlete was highest in August (preseason) (median, 146.0 impacts; IQR, 63.0-247.8) and lowest in November (median, 80.0 impacts; IQR, 35.0-148.0). In the studied period, 72% of concussions and 66.9% of head injury exposure occurred in practice. Even within the regular season, total head injury exposure in practices was 84.2% higher than in games.
“This incredible dataset we have on head impact measurement also gives us the opportunity to compare it with our other research looking at the correlation between a single head impact and changes in brain structure and function on MRI, on blood biomarkers, giving us the ability to look at the connection between mechanism of effect of injury and recovery from injury,” said Dr. McCrea.
These findings also provide an opportunity to modify approaches to preseason training and football practices to keep players safer, said Dr. McCrea, noting that about half of the variance in head injury exposure is at the level of the individual athlete.
“With this large body of athletes we’ve instrumented, we can look at, for instance, all of the running backs and understand the athlete and what his head injury exposure looks like compared to all other running backs. If we find out that an athlete has a rate of head injury exposure that’s 300% higher than most other players that play the same position, we can take that data directly to the athlete to work on their technique and approach to the game.
“Every researcher wishes that their basic science or their clinical research findings will have some impact on the health and well-being of the population they’re studying. By modifying practices and preseason training, football teams could greatly reduce the risk of injury and exposure for their players, while still maintaining the competitive nature of game play,” he added.
Through a combination of policy and education, similar strategies could be implemented to help prevent concussion and HIE in high school and youth football too, said Dr. McCrea.
‘Shocking’ findings
In an accompanying editorial, Christopher J. Nowinski, PhD, of the Concussion Legacy Foundation, Boston, and Robert C. Cantu, MD, department of neurosurgery, Emerson Hospital, Concord, Massachusetts, said the findings could have significant policy implications and offer a valuable expansion of prior research.
“From 2005 to 2010, studies on college football revealed that about two-thirds of head impacts occurred in practice,” they noted. “We cited this data in 2010 when we proposed to the NFL Players Association that the most effective way to reduce the risks of negative neurological outcomes was to reduce hitting in practice. They agreed, and in 2011 collectively bargained for severe contact limits in practice, with 14 full-contact practices allowed during the 17-week season. Since that rule was implemented, only 18% of NFL concussions have occurred in practice.”
“Against this backdrop, the results of the study by McCrea et al. are shocking,” they added. “It reveals that college football players still experience 72% of their concussions and 67% of their total head injury exposure in practice.”
Even more shocking, noted Dr. Nowinski and Dr. Cantu, is that these numbers are almost certainly an underestimate of the dangers of practice.
“As a former college football player and a former team physician, respectively, we find this situation inexcusable. Concussions in games are inevitable, but concussions in practice are preventable,” they wrote.
“Laudably,” they added “the investigators call on the NCAA and football conferences to explore policy and rule changes to reduce concussion incidence and HIE and to create robust educational offerings to encourage change from coaches and college administrators.”
A version of this article first appeared on Medscape.com.
Novel research from the Concussion Assessment, Research and Education (CARE) Consortium sheds new light on how to effectively reduce the incidence of concussion and head injury exposure in college football.
The study, led by neurotrauma experts Michael McCrea, PhD, and Brian Stemper, PhD, professors of neurosurgery at the Medical College of Wisconsin in Milwaukee, reports data from hundreds of college football players across five seasons and shows
The research also reveals that such injuries occur more often during practices than games.
“We think that with the findings from this paper, there’s a role for everybody to play in reducing injury,” Dr. McCrea said. “We hope these data help inform broad-based policy about practice and preseason training policies in collegiate football. We also think there’s a role for athletic administrators, coaches, and even athletes themselves.”
The study was published online Feb. 1 in JAMA Neurology.
More injuries in preseason
Concussion is one of the most common injuries in football. Beyond these harms are growing concerns that repetitive HIE may increase the risk of long-term neurologic health problems including chronic traumatic encephalopathy (CTE).
The CARE Consortium, which has been conducting research with college athletes across 26 sports and military cadets since 2014, has been interested in multiple facets of concussion and brain trauma.
“We’ve enrolled more than 50,000 athletes and service academy cadets into the consortium over the last 6 years to research all involved aspects including the clinical core, the imaging core, the blood biomarker core, and the genetic core, and we have a head impact measurement core.”
To investigate the pattern of concussion incidence across the football season in college players, the investigators used impact measurement technology across six Division I NCAA football programs participating in the CARE Consortium from 2015 to 2019.
A total of 658 players – all male, mean age 19 years – were fitted with the Head Impact Telemetry System (HITS) sensor arrays in their helmets to measure head impact frequency, location, and magnitude during play.
“This particular study had built-in algorithms that weeded out impacts that were below 10G of linear magnitude, because those have been determined not likely to be real impacts,” Dr. McCrea said.
Across the five seasons studied, 528,684 head impacts recorded met the quality standards for analysis. Players sustained a median of 415 (interquartile range [IQR], 190-727) impacts per season.
Of those, 68 players sustained a diagnosed concussion. In total, 48.5% of concussions occurred during preseason training, despite preseason representing only 20.8% of the football season. Total head injury exposure in the preseason occurred at twice the proportion of the regular season (324.9 vs. 162.4 impacts per team per day; mean difference, 162.6 impacts; 95% confidence interval, 110.9-214.3; P < .001).
“Preseason training often has a much higher intensity to it, in terms of the total hours, the actual training, and the heavy emphasis on full-contact drills like tackling and blocking,” said Dr. McCrea. “Even the volume of players that are participating is greater.”
Results also showed that in each of the five seasons, head injury exposure per athlete was highest in August (preseason) (median, 146.0 impacts; IQR, 63.0-247.8) and lowest in November (median, 80.0 impacts; IQR, 35.0-148.0). In the studied period, 72% of concussions and 66.9% of head injury exposure occurred in practice. Even within the regular season, total head injury exposure in practices was 84.2% higher than in games.
“This incredible dataset we have on head impact measurement also gives us the opportunity to compare it with our other research looking at the correlation between a single head impact and changes in brain structure and function on MRI, on blood biomarkers, giving us the ability to look at the connection between mechanism of effect of injury and recovery from injury,” said Dr. McCrea.
These findings also provide an opportunity to modify approaches to preseason training and football practices to keep players safer, said Dr. McCrea, noting that about half of the variance in head injury exposure is at the level of the individual athlete.
“With this large body of athletes we’ve instrumented, we can look at, for instance, all of the running backs and understand the athlete and what his head injury exposure looks like compared to all other running backs. If we find out that an athlete has a rate of head injury exposure that’s 300% higher than most other players that play the same position, we can take that data directly to the athlete to work on their technique and approach to the game.
“Every researcher wishes that their basic science or their clinical research findings will have some impact on the health and well-being of the population they’re studying. By modifying practices and preseason training, football teams could greatly reduce the risk of injury and exposure for their players, while still maintaining the competitive nature of game play,” he added.
Through a combination of policy and education, similar strategies could be implemented to help prevent concussion and HIE in high school and youth football too, said Dr. McCrea.
‘Shocking’ findings
In an accompanying editorial, Christopher J. Nowinski, PhD, of the Concussion Legacy Foundation, Boston, and Robert C. Cantu, MD, department of neurosurgery, Emerson Hospital, Concord, Massachusetts, said the findings could have significant policy implications and offer a valuable expansion of prior research.
“From 2005 to 2010, studies on college football revealed that about two-thirds of head impacts occurred in practice,” they noted. “We cited this data in 2010 when we proposed to the NFL Players Association that the most effective way to reduce the risks of negative neurological outcomes was to reduce hitting in practice. They agreed, and in 2011 collectively bargained for severe contact limits in practice, with 14 full-contact practices allowed during the 17-week season. Since that rule was implemented, only 18% of NFL concussions have occurred in practice.”
“Against this backdrop, the results of the study by McCrea et al. are shocking,” they added. “It reveals that college football players still experience 72% of their concussions and 67% of their total head injury exposure in practice.”
Even more shocking, noted Dr. Nowinski and Dr. Cantu, is that these numbers are almost certainly an underestimate of the dangers of practice.
“As a former college football player and a former team physician, respectively, we find this situation inexcusable. Concussions in games are inevitable, but concussions in practice are preventable,” they wrote.
“Laudably,” they added “the investigators call on the NCAA and football conferences to explore policy and rule changes to reduce concussion incidence and HIE and to create robust educational offerings to encourage change from coaches and college administrators.”
A version of this article first appeared on Medscape.com.
FROM JAMA NEUROLOGY
Loss of smell lingers post COVID-19
The findings illustrate that olfactory problems are common not only during the acute COVID-19 phase but also “in the long run” and that these problems should be “taken into consideration” when following up these patients, study investigator Johannes Frasnelli, MD, professor, department of anatomy, Université du Québec à Trois-Rivières, said in an interview.
Loss of the sense of smell can affect quality of life because it affects eating and drinking, and may even be dangerous, said Dr. Frasnelli. “If your sense of smell is impaired, you may unknowingly eat spoiled food, or you may not smell smoke or gas in your home,” he said. In addition, Dr. Frasnelli noted that an impaired sense of smell is associated with higher rates of depression. The findings will be presented at the annual meeting of the American Academy of Neurology in April.
‘Striking’ finding
Research shows that about 60% of patients with COVID-19 lose their sense of smell to some degree during the acute phase of the disease. “But we wanted to go further and look at the longer-term effects of loss of smell and taste,” said Dr. Frasnelli.
The analysis included 813 health care workers in the province of Quebec. For all the patients, SARS-CoV-2 infection was confirmed through testing with a nasopharyngeal viral swab.
Participants completed a 64-item online questionnaire that asked about three senses: olfactory; gustatory, which includes tastes such as sweet, sour, bitter, salty, savory and umami; and trigeminal, which includes sensations such as spiciness of hot peppers and “coolness” of mint.
They were asked to rate these on a scale of 0 (no perception) to 10 (very strong perception) before the infection, during the infection, and currently. They were also asked about other symptoms, including fatigue.
Most respondents had been infected in the first wave of the virus in March and April of 2020 and responded to the questionnaire an average of 5 months later.
The vast majority of respondents (84.1%) were women, which Dr. Frasnelli said was not surprising because women predominate in the health care field.
The analysis showed that average smell ratings were 8.98 before infection, 2.85 during the acute phase, and 7.41 when respondents answered the questionnaire. The sense of taste was less affected and recovered faster than did the sense of smell. Results for taste were 9.20 before infection, 3.59 during the acute phase, and 8.05 after COVID-19.
Among 580 respondents who indicated a compromised sense of smell during the acute phase, the average smell rating when answering the questionnaire was 6.89, compared to 9.03 before the infection. More than half (51.2%) reported not regaining full olfactory function.
The fact that the sense of smell had not returned to normal for half the participants so long after being infected is “novel and quite striking,” said Dr. Frasnelli.
However, he noted, this doesn’t necessarily mean all those with a compromised sense of smell “have huge problems.” In some cases, he said, the problem “is more subtle.”
Not a CNS problem?
Respondents also completed a chemosensory dysfunction home test (CD-HT). They were asked to prepare common household food items, such as peanut butter, sugar, salt, and vinegar, in a particular way – for example, to add sugar or salt to water – and provide feedback on how they smell and taste.
For this CD-HT analysis, 18.4% of respondents reported having persistent loss of smell. This, Dr. Frasnelli said, adds to evidence from self-reported responses and suggests that in some cases, the problem is more than senses not returning to normal.
“From the questionnaires, roughly 50% said their sense of smell is still not back to normal, and when we look at the CD home test, we see that almost 20% of subjects indeed have pretty strong impairment of their sense of smell,” he said.
The results showed no sex differences, although Dr. Frasnelli noted that most of the sample were women. “It’s tricky to look at the data with regard to sex because it’s a bit skewed,” he said.
Male respondents were older than female participants, but there was no difference in impairment between age groups. Dr. Frasnelli said this was “quite interesting,” inasmuch as older people usually lose some sense of smell.
The researchers have not yet examined whether the results differ by type of health care worker.
They also have not examined in detail whether infection severity affects the risk for extended olfactory impairment. Although some research suggests that the problem with smell is more common in less severe cases, Dr. Frasnelli noted this could be because loss of smell is not a huge problem for patients battling grave health problems.
As for other symptoms, many respondents reported lingering fatigue; some reported debilitating fatigue, said Dr. Frasnelli. However, he cautioned that this is difficult to interpret, because the participants were health care workers, many of whom returned to work during the pandemic and perhaps had not fully rested.
He also noted that he and his colleagues have not “made the link” between impaired smell and the degree of fatigue.
The COVID-19 virus appears to attack supporting sustentacular cells in the olfactory epithelium, not nerve cells.
“Right now, it seems that the smell problem is not a central nervous system problem but a peripheral problem,” said Dr. Frasnelli. “But we don’t know for sure; it may be that the virus somehow gets into the brain and some symptoms are caused by the effects of the infection on the brain.”
The researchers will extend their research with another questionnaire to assess senses 10-12 months after COVID-19.
Limitations of the study include the subjective nature of the smell and taste ratings and the single time point at which data were collected.
Confirmatory findings
Commenting on the research in an interview, Thomas Hummel, MD, professor, smell and taste clinic, department of otorhinolaryngology, Technische Universität Dresden (Germany), said the new results regarding loss of smell after COVID-19 are “very congruent” with what he and his colleagues have observed.
Research shows that up to one in five of those infected with SARS-CoV-2 experience olfactory loss. “While the numbers may vary a bit from study to study or lab to lab, I think 5% to 20% of post–COVID-19 patients exhibit long-term olfactory loss,” Dr. Hummel said.
His group has observed that “many more are not back to normal,” which conforms with what Dr. Frasnelli’s study reveals, said Dr. Hummel.
Also commenting on the research, Kenneth L. Tyler, MD, professor of neurology, University of Colorado at Denver, Aurora, and a fellow of the American Academy of Neurology, said the study was relatively large and the results “interesting.”
Although it “provides more evidence there’s a subset of patients with symptoms even well past the acute phase” of COVID-19, the results are “mostly confirmatory” and include “nothing super surprising,” Dr. Tyler said in an interview.
However, the investigators did attempt to make the study “a little more quantitative” and “to confirm the self-reporting with their validated CD home test,” he said.
Dr. Tyler wondered how representative the sample was and whether the study drew more participants with impaired senses. “If I had a loss of smell or taste, maybe I would be more likely to respond to such a survey,” he said.
He also noted the difficulty of separating loss of smell from loss of taste.
“If you lose your sense of smell, things don’t taste right, so it can be confounding as to how to separate out those two,” he noted.
The study was supported by the Foundation of the Université du Québec à Trois-Rivières and the Province of Quebec. Dr. Frasnelli has received royalties from Styriabooks in Austria for a book on olfaction published in 2019 and has received honoraria for speaking engagements. Dr. Hummel and Dr. Tyler have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The findings illustrate that olfactory problems are common not only during the acute COVID-19 phase but also “in the long run” and that these problems should be “taken into consideration” when following up these patients, study investigator Johannes Frasnelli, MD, professor, department of anatomy, Université du Québec à Trois-Rivières, said in an interview.
Loss of the sense of smell can affect quality of life because it affects eating and drinking, and may even be dangerous, said Dr. Frasnelli. “If your sense of smell is impaired, you may unknowingly eat spoiled food, or you may not smell smoke or gas in your home,” he said. In addition, Dr. Frasnelli noted that an impaired sense of smell is associated with higher rates of depression. The findings will be presented at the annual meeting of the American Academy of Neurology in April.
‘Striking’ finding
Research shows that about 60% of patients with COVID-19 lose their sense of smell to some degree during the acute phase of the disease. “But we wanted to go further and look at the longer-term effects of loss of smell and taste,” said Dr. Frasnelli.
The analysis included 813 health care workers in the province of Quebec. For all the patients, SARS-CoV-2 infection was confirmed through testing with a nasopharyngeal viral swab.
Participants completed a 64-item online questionnaire that asked about three senses: olfactory; gustatory, which includes tastes such as sweet, sour, bitter, salty, savory and umami; and trigeminal, which includes sensations such as spiciness of hot peppers and “coolness” of mint.
They were asked to rate these on a scale of 0 (no perception) to 10 (very strong perception) before the infection, during the infection, and currently. They were also asked about other symptoms, including fatigue.
Most respondents had been infected in the first wave of the virus in March and April of 2020 and responded to the questionnaire an average of 5 months later.
The vast majority of respondents (84.1%) were women, which Dr. Frasnelli said was not surprising because women predominate in the health care field.
The analysis showed that average smell ratings were 8.98 before infection, 2.85 during the acute phase, and 7.41 when respondents answered the questionnaire. The sense of taste was less affected and recovered faster than did the sense of smell. Results for taste were 9.20 before infection, 3.59 during the acute phase, and 8.05 after COVID-19.
Among 580 respondents who indicated a compromised sense of smell during the acute phase, the average smell rating when answering the questionnaire was 6.89, compared to 9.03 before the infection. More than half (51.2%) reported not regaining full olfactory function.
The fact that the sense of smell had not returned to normal for half the participants so long after being infected is “novel and quite striking,” said Dr. Frasnelli.
However, he noted, this doesn’t necessarily mean all those with a compromised sense of smell “have huge problems.” In some cases, he said, the problem “is more subtle.”
Not a CNS problem?
Respondents also completed a chemosensory dysfunction home test (CD-HT). They were asked to prepare common household food items, such as peanut butter, sugar, salt, and vinegar, in a particular way – for example, to add sugar or salt to water – and provide feedback on how they smell and taste.
For this CD-HT analysis, 18.4% of respondents reported having persistent loss of smell. This, Dr. Frasnelli said, adds to evidence from self-reported responses and suggests that in some cases, the problem is more than senses not returning to normal.
“From the questionnaires, roughly 50% said their sense of smell is still not back to normal, and when we look at the CD home test, we see that almost 20% of subjects indeed have pretty strong impairment of their sense of smell,” he said.
The results showed no sex differences, although Dr. Frasnelli noted that most of the sample were women. “It’s tricky to look at the data with regard to sex because it’s a bit skewed,” he said.
Male respondents were older than female participants, but there was no difference in impairment between age groups. Dr. Frasnelli said this was “quite interesting,” inasmuch as older people usually lose some sense of smell.
The researchers have not yet examined whether the results differ by type of health care worker.
They also have not examined in detail whether infection severity affects the risk for extended olfactory impairment. Although some research suggests that the problem with smell is more common in less severe cases, Dr. Frasnelli noted this could be because loss of smell is not a huge problem for patients battling grave health problems.
As for other symptoms, many respondents reported lingering fatigue; some reported debilitating fatigue, said Dr. Frasnelli. However, he cautioned that this is difficult to interpret, because the participants were health care workers, many of whom returned to work during the pandemic and perhaps had not fully rested.
He also noted that he and his colleagues have not “made the link” between impaired smell and the degree of fatigue.
The COVID-19 virus appears to attack supporting sustentacular cells in the olfactory epithelium, not nerve cells.
“Right now, it seems that the smell problem is not a central nervous system problem but a peripheral problem,” said Dr. Frasnelli. “But we don’t know for sure; it may be that the virus somehow gets into the brain and some symptoms are caused by the effects of the infection on the brain.”
The researchers will extend their research with another questionnaire to assess senses 10-12 months after COVID-19.
Limitations of the study include the subjective nature of the smell and taste ratings and the single time point at which data were collected.
Confirmatory findings
Commenting on the research in an interview, Thomas Hummel, MD, professor, smell and taste clinic, department of otorhinolaryngology, Technische Universität Dresden (Germany), said the new results regarding loss of smell after COVID-19 are “very congruent” with what he and his colleagues have observed.
Research shows that up to one in five of those infected with SARS-CoV-2 experience olfactory loss. “While the numbers may vary a bit from study to study or lab to lab, I think 5% to 20% of post–COVID-19 patients exhibit long-term olfactory loss,” Dr. Hummel said.
His group has observed that “many more are not back to normal,” which conforms with what Dr. Frasnelli’s study reveals, said Dr. Hummel.
Also commenting on the research, Kenneth L. Tyler, MD, professor of neurology, University of Colorado at Denver, Aurora, and a fellow of the American Academy of Neurology, said the study was relatively large and the results “interesting.”
Although it “provides more evidence there’s a subset of patients with symptoms even well past the acute phase” of COVID-19, the results are “mostly confirmatory” and include “nothing super surprising,” Dr. Tyler said in an interview.
However, the investigators did attempt to make the study “a little more quantitative” and “to confirm the self-reporting with their validated CD home test,” he said.
Dr. Tyler wondered how representative the sample was and whether the study drew more participants with impaired senses. “If I had a loss of smell or taste, maybe I would be more likely to respond to such a survey,” he said.
He also noted the difficulty of separating loss of smell from loss of taste.
“If you lose your sense of smell, things don’t taste right, so it can be confounding as to how to separate out those two,” he noted.
The study was supported by the Foundation of the Université du Québec à Trois-Rivières and the Province of Quebec. Dr. Frasnelli has received royalties from Styriabooks in Austria for a book on olfaction published in 2019 and has received honoraria for speaking engagements. Dr. Hummel and Dr. Tyler have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The findings illustrate that olfactory problems are common not only during the acute COVID-19 phase but also “in the long run” and that these problems should be “taken into consideration” when following up these patients, study investigator Johannes Frasnelli, MD, professor, department of anatomy, Université du Québec à Trois-Rivières, said in an interview.
Loss of the sense of smell can affect quality of life because it affects eating and drinking, and may even be dangerous, said Dr. Frasnelli. “If your sense of smell is impaired, you may unknowingly eat spoiled food, or you may not smell smoke or gas in your home,” he said. In addition, Dr. Frasnelli noted that an impaired sense of smell is associated with higher rates of depression. The findings will be presented at the annual meeting of the American Academy of Neurology in April.
‘Striking’ finding
Research shows that about 60% of patients with COVID-19 lose their sense of smell to some degree during the acute phase of the disease. “But we wanted to go further and look at the longer-term effects of loss of smell and taste,” said Dr. Frasnelli.
The analysis included 813 health care workers in the province of Quebec. For all the patients, SARS-CoV-2 infection was confirmed through testing with a nasopharyngeal viral swab.
Participants completed a 64-item online questionnaire that asked about three senses: olfactory; gustatory, which includes tastes such as sweet, sour, bitter, salty, savory and umami; and trigeminal, which includes sensations such as spiciness of hot peppers and “coolness” of mint.
They were asked to rate these on a scale of 0 (no perception) to 10 (very strong perception) before the infection, during the infection, and currently. They were also asked about other symptoms, including fatigue.
Most respondents had been infected in the first wave of the virus in March and April of 2020 and responded to the questionnaire an average of 5 months later.
The vast majority of respondents (84.1%) were women, which Dr. Frasnelli said was not surprising because women predominate in the health care field.
The analysis showed that average smell ratings were 8.98 before infection, 2.85 during the acute phase, and 7.41 when respondents answered the questionnaire. The sense of taste was less affected and recovered faster than did the sense of smell. Results for taste were 9.20 before infection, 3.59 during the acute phase, and 8.05 after COVID-19.
Among 580 respondents who indicated a compromised sense of smell during the acute phase, the average smell rating when answering the questionnaire was 6.89, compared to 9.03 before the infection. More than half (51.2%) reported not regaining full olfactory function.
The fact that the sense of smell had not returned to normal for half the participants so long after being infected is “novel and quite striking,” said Dr. Frasnelli.
However, he noted, this doesn’t necessarily mean all those with a compromised sense of smell “have huge problems.” In some cases, he said, the problem “is more subtle.”
Not a CNS problem?
Respondents also completed a chemosensory dysfunction home test (CD-HT). They were asked to prepare common household food items, such as peanut butter, sugar, salt, and vinegar, in a particular way – for example, to add sugar or salt to water – and provide feedback on how they smell and taste.
For this CD-HT analysis, 18.4% of respondents reported having persistent loss of smell. This, Dr. Frasnelli said, adds to evidence from self-reported responses and suggests that in some cases, the problem is more than senses not returning to normal.
“From the questionnaires, roughly 50% said their sense of smell is still not back to normal, and when we look at the CD home test, we see that almost 20% of subjects indeed have pretty strong impairment of their sense of smell,” he said.
The results showed no sex differences, although Dr. Frasnelli noted that most of the sample were women. “It’s tricky to look at the data with regard to sex because it’s a bit skewed,” he said.
Male respondents were older than female participants, but there was no difference in impairment between age groups. Dr. Frasnelli said this was “quite interesting,” inasmuch as older people usually lose some sense of smell.
The researchers have not yet examined whether the results differ by type of health care worker.
They also have not examined in detail whether infection severity affects the risk for extended olfactory impairment. Although some research suggests that the problem with smell is more common in less severe cases, Dr. Frasnelli noted this could be because loss of smell is not a huge problem for patients battling grave health problems.
As for other symptoms, many respondents reported lingering fatigue; some reported debilitating fatigue, said Dr. Frasnelli. However, he cautioned that this is difficult to interpret, because the participants were health care workers, many of whom returned to work during the pandemic and perhaps had not fully rested.
He also noted that he and his colleagues have not “made the link” between impaired smell and the degree of fatigue.
The COVID-19 virus appears to attack supporting sustentacular cells in the olfactory epithelium, not nerve cells.
“Right now, it seems that the smell problem is not a central nervous system problem but a peripheral problem,” said Dr. Frasnelli. “But we don’t know for sure; it may be that the virus somehow gets into the brain and some symptoms are caused by the effects of the infection on the brain.”
The researchers will extend their research with another questionnaire to assess senses 10-12 months after COVID-19.
Limitations of the study include the subjective nature of the smell and taste ratings and the single time point at which data were collected.
Confirmatory findings
Commenting on the research in an interview, Thomas Hummel, MD, professor, smell and taste clinic, department of otorhinolaryngology, Technische Universität Dresden (Germany), said the new results regarding loss of smell after COVID-19 are “very congruent” with what he and his colleagues have observed.
Research shows that up to one in five of those infected with SARS-CoV-2 experience olfactory loss. “While the numbers may vary a bit from study to study or lab to lab, I think 5% to 20% of post–COVID-19 patients exhibit long-term olfactory loss,” Dr. Hummel said.
His group has observed that “many more are not back to normal,” which conforms with what Dr. Frasnelli’s study reveals, said Dr. Hummel.
Also commenting on the research, Kenneth L. Tyler, MD, professor of neurology, University of Colorado at Denver, Aurora, and a fellow of the American Academy of Neurology, said the study was relatively large and the results “interesting.”
Although it “provides more evidence there’s a subset of patients with symptoms even well past the acute phase” of COVID-19, the results are “mostly confirmatory” and include “nothing super surprising,” Dr. Tyler said in an interview.
However, the investigators did attempt to make the study “a little more quantitative” and “to confirm the self-reporting with their validated CD home test,” he said.
Dr. Tyler wondered how representative the sample was and whether the study drew more participants with impaired senses. “If I had a loss of smell or taste, maybe I would be more likely to respond to such a survey,” he said.
He also noted the difficulty of separating loss of smell from loss of taste.
“If you lose your sense of smell, things don’t taste right, so it can be confounding as to how to separate out those two,” he noted.
The study was supported by the Foundation of the Université du Québec à Trois-Rivières and the Province of Quebec. Dr. Frasnelli has received royalties from Styriabooks in Austria for a book on olfaction published in 2019 and has received honoraria for speaking engagements. Dr. Hummel and Dr. Tyler have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Strep A and tic worsening: Final word?
Exposure to Group A streptococcus (GAS) does not appear to worsen symptoms of Tourette syndrome and other chronic tic disorders (CTDs) in children and adolescents, new research suggests.
Investigators studied over 700 children and teenagers with CTDs, one-third of whom also had attention deficit hyperactivity disorder and one-third who had obsessive-compulsive disorder (OCD).
The youngsters were followed for an average of 16 months and evaluated at 4-month intervals to see if they were infected with GAS. Tic severity was monitored through telephone interviews, in-person visits, and parental reports.
A little less than half the children experienced worsening of tics during the study period, but the researchers found no association between these exacerbations and GAS exposure.
There was also no link between GAS and worsening OCD. However, researchers did find an association between GAS exposure and an increase in hyperactivity and impulsivity in patients with ADHD.
“This study does not support GAS exposures as contributing factors for tic exacerbations in children with CTD,” the authors note.
“Specific work-up or active management of GAS infections is unlikely to help modifying the course of tics in CTD and is therefore not recommended,” they conclude.
The study was published online in Neurology.
‘Intense debate’
The association between GAS and CTD stems from the description of Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal infection (PANDAS) – a condition that is now incorporated in the pediatric acute neuropsychiatric syndromes (PANS), the authors note. Tics constitute an “accompanying feature” of this condition.
However, neither population-based nor longitudinal clinical studies “could definitely establish if tic exacerbations in CTD are associated with GAS infections,” they note.
“The link between streptococcus and tics in children is still a matter of intense debate,” said study author Davide Martino, MD, PhD, director of the Movement Disorders Program at the University of Calgary (Alta.), in a press release.
“We wanted to look at that question, as well as a possible link between strep and behavioral symptoms like obsessive-compulsive disorder and attention deficit hyperactivity disorder,” he said.
The researchers followed 715 children with CTD (mean age 10.7 years, 76.8% male) who were drawn from 16 specialist clinics in nine countries. Almost all (90.8%) had a diagnosis of Tourette syndrome (TS); 31.7% had OCD, and 36.1% had ADHD.
Participants received a throat swab at baseline, and of these, 8.4% tested positive for GAS.
Participants were evaluated over a 16- to 18-month period, consisting of:
- Face-to-face interviews and collection of throat swabs and serum at 4-month intervals.
- Telephone interviews at 4-month intervals, which took place at 2 months between study visit.
- Weekly diaries: Parents were asked to indicate any worsening of tics and focus on detecting the earliest possible tic exacerbation.
Beyond the regularly scheduled visits, parents were instructed to report, by phone or email, any noticeable increase in tic severity and then attend an in-person visit.
Tic exacerbations were defined as an increase of greater than or equal to 6 points on the Yale Global Tic Severity Scale-Total Tic Severity Score (YGTSS-TTS), compared with the previous assessment.
OCD and ADHD symptoms were assessed according to the Yale-Brown Obsessive-Compulsive Scale and the parent-reported Swanson, Nolan, and Pelham-IV (SNAP-IV) questionnaire.
The researchers divided GAS exposures into four categories: new definite exposure; new possible exposure; ongoing definite exposure; and ongoing possible exposure.
Unlikely trigger
During the follow-up period, 43.1% (n = 308) of participants experienced tic exacerbations. Of these, 218 participants experienced one exacerbation, while 90 participants experienced two, three, or four exacerbations.
The researchers did not find a significant association between GAS exposure status and tic exacerbation.
Participants who did develop a GAS-associated exacerbation (n = 49) were younger at study exit (9.63 vs. 11.4 years, P < .0001) and were more likely to be male (46/49 vs. 210/259, Fisher’s = .035), compared with participants who developed a non-GAS-associated tic exacerbation (n = 259).
Additional analyses were adjusted for sex, age at onset, exposure to psychotropic medications, exposures to antibiotics, geographical regions, and number of visits in the time interval of interest. These analyses continued to yield no significant association between new or ongoing concurrent GAS exposure episodes and tic exacerbation events.
Of the children in the study, 103 had a positive throat swab, indicating a new definite GAS exposure, whereas 46 had a positive throat swab indicating an ongoing definite exposure (n = 149 visits). Of these visits, only 20 corresponded to tic exacerbations.
There was also no association between GAS exposure and OCD symptom severity. However, it was associated with longitudinal changes (between 17% and 21%, depending on GAS exposure definition) in the severity of hyperactivity-impulsivity symptoms in children with ADHD.
“It is known that immune activation may concur with tic severity in youth with CTDs and that psychosocial stress levels may predict short-term future tic severity in these patients,” the authors write.
“Our findings suggest that GAS is unlikely to be the main trigger for immune activation in these patients,” they add.
Brick or cornerstone?
Commenting on the study for this news organization, Margo Thienemann, MD, clinical professor of psychiatry, Stanford (Calif.) University, said that in the clinic population they treat, GAS, other pathogens, and other stresses can “each be associated with PANS symptom exacerbations.”
However, these “would not be likely to cause PANS symptoms exacerbations in the vast majority of individuals, only individuals with genetic backgrounds and immunologic dysfunctions creating susceptibility,” said Dr. Thienemann, who also directs the Pediatric Acute-Onset Neuropsychiatric Syndrome (PANS) Clinic at Stanford Children’s Health. She was not involved with the study.
In an accompanying editorial, Andrea Cavanna, MD, PhD, honorary reader in neuropsychiatry, Birmingham (England) Medical School and Keith Coffman, MD, director, Tourette Syndrome Center of Excellence, Children’s Mercy Hospital, Kansas City, Mo., suggest that perhaps the “interaction of psychosocial stress and GAS infections contributes more to tic exacerbation than psychosocial stress alone.”
“Time will tell whether this study stands as another brick – a cornerstone? – in the wall that separates streptococcus from tics,” they write.
The study was supported by the European Union’s Seventh Framework Program. Dr. Martino has received honoraria for lecturing from the Movement Disorders Society, Tourette Syndrome Association of America, and Dystonia Medical Research Foundation Canada; research funding support from Dystonia Medical Research Foundation Canada, the University of Calgary (Alta.), the Michael P. Smith Family, the Owerko Foundation, Ipsen Corporate, the Parkinson Association of Alberta, and the Canadian Institutes for Health Research; and royalties from Springer-Verlag. The other authors’ disclosures are listed in the original article. Dr. Cavanna, Dr. Coffman, and Dr. Thienemann have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Exposure to Group A streptococcus (GAS) does not appear to worsen symptoms of Tourette syndrome and other chronic tic disorders (CTDs) in children and adolescents, new research suggests.
Investigators studied over 700 children and teenagers with CTDs, one-third of whom also had attention deficit hyperactivity disorder and one-third who had obsessive-compulsive disorder (OCD).
The youngsters were followed for an average of 16 months and evaluated at 4-month intervals to see if they were infected with GAS. Tic severity was monitored through telephone interviews, in-person visits, and parental reports.
A little less than half the children experienced worsening of tics during the study period, but the researchers found no association between these exacerbations and GAS exposure.
There was also no link between GAS and worsening OCD. However, researchers did find an association between GAS exposure and an increase in hyperactivity and impulsivity in patients with ADHD.
“This study does not support GAS exposures as contributing factors for tic exacerbations in children with CTD,” the authors note.
“Specific work-up or active management of GAS infections is unlikely to help modifying the course of tics in CTD and is therefore not recommended,” they conclude.
The study was published online in Neurology.
‘Intense debate’
The association between GAS and CTD stems from the description of Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal infection (PANDAS) – a condition that is now incorporated in the pediatric acute neuropsychiatric syndromes (PANS), the authors note. Tics constitute an “accompanying feature” of this condition.
However, neither population-based nor longitudinal clinical studies “could definitely establish if tic exacerbations in CTD are associated with GAS infections,” they note.
“The link between streptococcus and tics in children is still a matter of intense debate,” said study author Davide Martino, MD, PhD, director of the Movement Disorders Program at the University of Calgary (Alta.), in a press release.
“We wanted to look at that question, as well as a possible link between strep and behavioral symptoms like obsessive-compulsive disorder and attention deficit hyperactivity disorder,” he said.
The researchers followed 715 children with CTD (mean age 10.7 years, 76.8% male) who were drawn from 16 specialist clinics in nine countries. Almost all (90.8%) had a diagnosis of Tourette syndrome (TS); 31.7% had OCD, and 36.1% had ADHD.
Participants received a throat swab at baseline, and of these, 8.4% tested positive for GAS.
Participants were evaluated over a 16- to 18-month period, consisting of:
- Face-to-face interviews and collection of throat swabs and serum at 4-month intervals.
- Telephone interviews at 4-month intervals, which took place at 2 months between study visit.
- Weekly diaries: Parents were asked to indicate any worsening of tics and focus on detecting the earliest possible tic exacerbation.
Beyond the regularly scheduled visits, parents were instructed to report, by phone or email, any noticeable increase in tic severity and then attend an in-person visit.
Tic exacerbations were defined as an increase of greater than or equal to 6 points on the Yale Global Tic Severity Scale-Total Tic Severity Score (YGTSS-TTS), compared with the previous assessment.
OCD and ADHD symptoms were assessed according to the Yale-Brown Obsessive-Compulsive Scale and the parent-reported Swanson, Nolan, and Pelham-IV (SNAP-IV) questionnaire.
The researchers divided GAS exposures into four categories: new definite exposure; new possible exposure; ongoing definite exposure; and ongoing possible exposure.
Unlikely trigger
During the follow-up period, 43.1% (n = 308) of participants experienced tic exacerbations. Of these, 218 participants experienced one exacerbation, while 90 participants experienced two, three, or four exacerbations.
The researchers did not find a significant association between GAS exposure status and tic exacerbation.
Participants who did develop a GAS-associated exacerbation (n = 49) were younger at study exit (9.63 vs. 11.4 years, P < .0001) and were more likely to be male (46/49 vs. 210/259, Fisher’s = .035), compared with participants who developed a non-GAS-associated tic exacerbation (n = 259).
Additional analyses were adjusted for sex, age at onset, exposure to psychotropic medications, exposures to antibiotics, geographical regions, and number of visits in the time interval of interest. These analyses continued to yield no significant association between new or ongoing concurrent GAS exposure episodes and tic exacerbation events.
Of the children in the study, 103 had a positive throat swab, indicating a new definite GAS exposure, whereas 46 had a positive throat swab indicating an ongoing definite exposure (n = 149 visits). Of these visits, only 20 corresponded to tic exacerbations.
There was also no association between GAS exposure and OCD symptom severity. However, it was associated with longitudinal changes (between 17% and 21%, depending on GAS exposure definition) in the severity of hyperactivity-impulsivity symptoms in children with ADHD.
“It is known that immune activation may concur with tic severity in youth with CTDs and that psychosocial stress levels may predict short-term future tic severity in these patients,” the authors write.
“Our findings suggest that GAS is unlikely to be the main trigger for immune activation in these patients,” they add.
Brick or cornerstone?
Commenting on the study for this news organization, Margo Thienemann, MD, clinical professor of psychiatry, Stanford (Calif.) University, said that in the clinic population they treat, GAS, other pathogens, and other stresses can “each be associated with PANS symptom exacerbations.”
However, these “would not be likely to cause PANS symptoms exacerbations in the vast majority of individuals, only individuals with genetic backgrounds and immunologic dysfunctions creating susceptibility,” said Dr. Thienemann, who also directs the Pediatric Acute-Onset Neuropsychiatric Syndrome (PANS) Clinic at Stanford Children’s Health. She was not involved with the study.
In an accompanying editorial, Andrea Cavanna, MD, PhD, honorary reader in neuropsychiatry, Birmingham (England) Medical School and Keith Coffman, MD, director, Tourette Syndrome Center of Excellence, Children’s Mercy Hospital, Kansas City, Mo., suggest that perhaps the “interaction of psychosocial stress and GAS infections contributes more to tic exacerbation than psychosocial stress alone.”
“Time will tell whether this study stands as another brick – a cornerstone? – in the wall that separates streptococcus from tics,” they write.
The study was supported by the European Union’s Seventh Framework Program. Dr. Martino has received honoraria for lecturing from the Movement Disorders Society, Tourette Syndrome Association of America, and Dystonia Medical Research Foundation Canada; research funding support from Dystonia Medical Research Foundation Canada, the University of Calgary (Alta.), the Michael P. Smith Family, the Owerko Foundation, Ipsen Corporate, the Parkinson Association of Alberta, and the Canadian Institutes for Health Research; and royalties from Springer-Verlag. The other authors’ disclosures are listed in the original article. Dr. Cavanna, Dr. Coffman, and Dr. Thienemann have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Exposure to Group A streptococcus (GAS) does not appear to worsen symptoms of Tourette syndrome and other chronic tic disorders (CTDs) in children and adolescents, new research suggests.
Investigators studied over 700 children and teenagers with CTDs, one-third of whom also had attention deficit hyperactivity disorder and one-third who had obsessive-compulsive disorder (OCD).
The youngsters were followed for an average of 16 months and evaluated at 4-month intervals to see if they were infected with GAS. Tic severity was monitored through telephone interviews, in-person visits, and parental reports.
A little less than half the children experienced worsening of tics during the study period, but the researchers found no association between these exacerbations and GAS exposure.
There was also no link between GAS and worsening OCD. However, researchers did find an association between GAS exposure and an increase in hyperactivity and impulsivity in patients with ADHD.
“This study does not support GAS exposures as contributing factors for tic exacerbations in children with CTD,” the authors note.
“Specific work-up or active management of GAS infections is unlikely to help modifying the course of tics in CTD and is therefore not recommended,” they conclude.
The study was published online in Neurology.
‘Intense debate’
The association between GAS and CTD stems from the description of Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal infection (PANDAS) – a condition that is now incorporated in the pediatric acute neuropsychiatric syndromes (PANS), the authors note. Tics constitute an “accompanying feature” of this condition.
However, neither population-based nor longitudinal clinical studies “could definitely establish if tic exacerbations in CTD are associated with GAS infections,” they note.
“The link between streptococcus and tics in children is still a matter of intense debate,” said study author Davide Martino, MD, PhD, director of the Movement Disorders Program at the University of Calgary (Alta.), in a press release.
“We wanted to look at that question, as well as a possible link between strep and behavioral symptoms like obsessive-compulsive disorder and attention deficit hyperactivity disorder,” he said.
The researchers followed 715 children with CTD (mean age 10.7 years, 76.8% male) who were drawn from 16 specialist clinics in nine countries. Almost all (90.8%) had a diagnosis of Tourette syndrome (TS); 31.7% had OCD, and 36.1% had ADHD.
Participants received a throat swab at baseline, and of these, 8.4% tested positive for GAS.
Participants were evaluated over a 16- to 18-month period, consisting of:
- Face-to-face interviews and collection of throat swabs and serum at 4-month intervals.
- Telephone interviews at 4-month intervals, which took place at 2 months between study visit.
- Weekly diaries: Parents were asked to indicate any worsening of tics and focus on detecting the earliest possible tic exacerbation.
Beyond the regularly scheduled visits, parents were instructed to report, by phone or email, any noticeable increase in tic severity and then attend an in-person visit.
Tic exacerbations were defined as an increase of greater than or equal to 6 points on the Yale Global Tic Severity Scale-Total Tic Severity Score (YGTSS-TTS), compared with the previous assessment.
OCD and ADHD symptoms were assessed according to the Yale-Brown Obsessive-Compulsive Scale and the parent-reported Swanson, Nolan, and Pelham-IV (SNAP-IV) questionnaire.
The researchers divided GAS exposures into four categories: new definite exposure; new possible exposure; ongoing definite exposure; and ongoing possible exposure.
Unlikely trigger
During the follow-up period, 43.1% (n = 308) of participants experienced tic exacerbations. Of these, 218 participants experienced one exacerbation, while 90 participants experienced two, three, or four exacerbations.
The researchers did not find a significant association between GAS exposure status and tic exacerbation.
Participants who did develop a GAS-associated exacerbation (n = 49) were younger at study exit (9.63 vs. 11.4 years, P < .0001) and were more likely to be male (46/49 vs. 210/259, Fisher’s = .035), compared with participants who developed a non-GAS-associated tic exacerbation (n = 259).
Additional analyses were adjusted for sex, age at onset, exposure to psychotropic medications, exposures to antibiotics, geographical regions, and number of visits in the time interval of interest. These analyses continued to yield no significant association between new or ongoing concurrent GAS exposure episodes and tic exacerbation events.
Of the children in the study, 103 had a positive throat swab, indicating a new definite GAS exposure, whereas 46 had a positive throat swab indicating an ongoing definite exposure (n = 149 visits). Of these visits, only 20 corresponded to tic exacerbations.
There was also no association between GAS exposure and OCD symptom severity. However, it was associated with longitudinal changes (between 17% and 21%, depending on GAS exposure definition) in the severity of hyperactivity-impulsivity symptoms in children with ADHD.
“It is known that immune activation may concur with tic severity in youth with CTDs and that psychosocial stress levels may predict short-term future tic severity in these patients,” the authors write.
“Our findings suggest that GAS is unlikely to be the main trigger for immune activation in these patients,” they add.
Brick or cornerstone?
Commenting on the study for this news organization, Margo Thienemann, MD, clinical professor of psychiatry, Stanford (Calif.) University, said that in the clinic population they treat, GAS, other pathogens, and other stresses can “each be associated with PANS symptom exacerbations.”
However, these “would not be likely to cause PANS symptoms exacerbations in the vast majority of individuals, only individuals with genetic backgrounds and immunologic dysfunctions creating susceptibility,” said Dr. Thienemann, who also directs the Pediatric Acute-Onset Neuropsychiatric Syndrome (PANS) Clinic at Stanford Children’s Health. She was not involved with the study.
In an accompanying editorial, Andrea Cavanna, MD, PhD, honorary reader in neuropsychiatry, Birmingham (England) Medical School and Keith Coffman, MD, director, Tourette Syndrome Center of Excellence, Children’s Mercy Hospital, Kansas City, Mo., suggest that perhaps the “interaction of psychosocial stress and GAS infections contributes more to tic exacerbation than psychosocial stress alone.”
“Time will tell whether this study stands as another brick – a cornerstone? – in the wall that separates streptococcus from tics,” they write.
The study was supported by the European Union’s Seventh Framework Program. Dr. Martino has received honoraria for lecturing from the Movement Disorders Society, Tourette Syndrome Association of America, and Dystonia Medical Research Foundation Canada; research funding support from Dystonia Medical Research Foundation Canada, the University of Calgary (Alta.), the Michael P. Smith Family, the Owerko Foundation, Ipsen Corporate, the Parkinson Association of Alberta, and the Canadian Institutes for Health Research; and royalties from Springer-Verlag. The other authors’ disclosures are listed in the original article. Dr. Cavanna, Dr. Coffman, and Dr. Thienemann have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Short sleep predicts incident dementia and all-cause mortality
More evidence has emerged linking sleep deficiency, dementia, and mortality.
“Sleep disturbance and insufficiency have been shown to be associated with both the development and progression of Alzheimer’s disease and with all-cause mortality,” wrote Rebecca S. Robbins, PhD, of Brigham and Women’s Hospital, Boston, and colleagues. However, research on this topic has yielded conflicting results, and “few studies have included a comprehensive set of sleep characteristics in a single examination of incident dementia and all-cause mortality.”
In a study published in Aging, the researchers identified 2,812 adults aged 65 years and older from the National Health and Aging Trends Study (NHATS), a nationally representative longitudinal study of Medicare beneficiaries aged 65 years and older in the United States.
Participants completed surveys about sleep disturbance and duration in 2013 (1,575 individuals) and in 2014 (1,237 individuals), and the researchers examined the relationship between sleep disturbance and deficiency and incident dementia and all-cause mortality over the next 5 years. The average age of the study participants was 76.9 years, 60% were women, and 72% were White.
Overall, approximately 60% of the participants reported never or rarely having problems with alertness, approximately half said that they rarely or never napped, and more than half said they fell asleep in 15 minutes or less. Approximately 70% rated their sleep quality as good or very good, and more than 90% said they rarely or never snored.
The researchers examined the relationships between sleep characteristics and the development of incident dementia over 5 years. In a fully adjusted Cox multivariate analysis, individuals who slept 5 hours or less per night had approximately twice the risk for incident dementia as those who slept longer (hazard ratio, 2.04); risk of dementia also was higher among those who took 30 minutes or longer to fall asleep (HR, 1.45).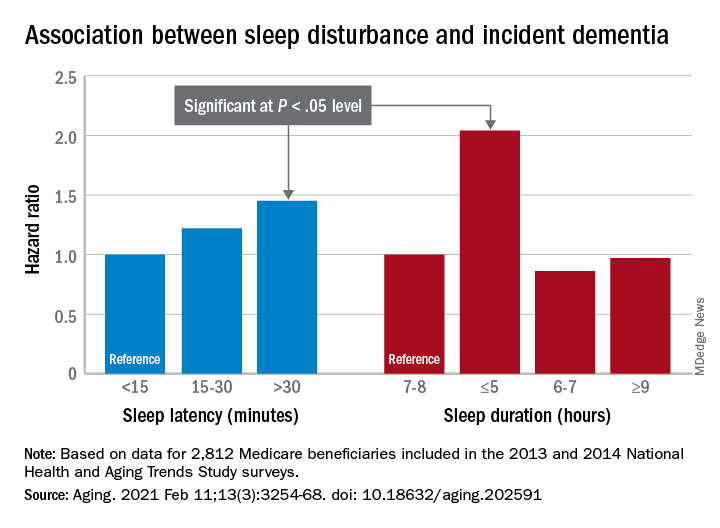
In addition, the risk of all-cause mortality was significantly higher among individuals who reported difficulty maintaining alertness some days or most days/every day (HR, 1.49 and HR, 1.65, respectively), routinely napping some days or most days/every day (HR, 1.38 and HR, 1.73, respectively), poor or very poor sleep quality (HR, 1.75), and sleeping 5 hours or less each night (HR, 2.38).
The study findings were limited by several factors including a population representing only one-quarter of the NHATS cohort, which prevented nationally representative estimates, the availability of only 2 years of sleep data, and small sample size for certain response categories, the researchers noted.
However, “our study offers a contribution to the literature on sleep among aging populations in its assessment of incident dementia and all-cause mortality and a range of sleep characteristics among older adults,” they said. In particular, “short sleep duration was a strong predictor of both incident dementia and all-cause mortality, suggesting this may be a sleep characteristic that is important – over and above the other predictors – of adverse outcomes among older adults,” and future areas for research include the development of novel behavioral interventions to improve sleep in this population.
The study was supported in part by the National Institute for Occupational Safety and Health; the National Heart, Lung, and Blood Institute; the National Institute on Aging; and the Brigham Research Institute Fund to Sustain Research Excellence. Lead author Dr. Robbins disclosed fees from Denihan Hospitality, Rituals Cosmetics, Dagmejan, Asystem, and SleepCycle. Several coauthors disclosed relationships with multiple pharmaceutical companies, and support from various philanthropic organizations.
More evidence has emerged linking sleep deficiency, dementia, and mortality.
“Sleep disturbance and insufficiency have been shown to be associated with both the development and progression of Alzheimer’s disease and with all-cause mortality,” wrote Rebecca S. Robbins, PhD, of Brigham and Women’s Hospital, Boston, and colleagues. However, research on this topic has yielded conflicting results, and “few studies have included a comprehensive set of sleep characteristics in a single examination of incident dementia and all-cause mortality.”
In a study published in Aging, the researchers identified 2,812 adults aged 65 years and older from the National Health and Aging Trends Study (NHATS), a nationally representative longitudinal study of Medicare beneficiaries aged 65 years and older in the United States.
Participants completed surveys about sleep disturbance and duration in 2013 (1,575 individuals) and in 2014 (1,237 individuals), and the researchers examined the relationship between sleep disturbance and deficiency and incident dementia and all-cause mortality over the next 5 years. The average age of the study participants was 76.9 years, 60% were women, and 72% were White.
Overall, approximately 60% of the participants reported never or rarely having problems with alertness, approximately half said that they rarely or never napped, and more than half said they fell asleep in 15 minutes or less. Approximately 70% rated their sleep quality as good or very good, and more than 90% said they rarely or never snored.
The researchers examined the relationships between sleep characteristics and the development of incident dementia over 5 years. In a fully adjusted Cox multivariate analysis, individuals who slept 5 hours or less per night had approximately twice the risk for incident dementia as those who slept longer (hazard ratio, 2.04); risk of dementia also was higher among those who took 30 minutes or longer to fall asleep (HR, 1.45).
In addition, the risk of all-cause mortality was significantly higher among individuals who reported difficulty maintaining alertness some days or most days/every day (HR, 1.49 and HR, 1.65, respectively), routinely napping some days or most days/every day (HR, 1.38 and HR, 1.73, respectively), poor or very poor sleep quality (HR, 1.75), and sleeping 5 hours or less each night (HR, 2.38).
The study findings were limited by several factors including a population representing only one-quarter of the NHATS cohort, which prevented nationally representative estimates, the availability of only 2 years of sleep data, and small sample size for certain response categories, the researchers noted.
However, “our study offers a contribution to the literature on sleep among aging populations in its assessment of incident dementia and all-cause mortality and a range of sleep characteristics among older adults,” they said. In particular, “short sleep duration was a strong predictor of both incident dementia and all-cause mortality, suggesting this may be a sleep characteristic that is important – over and above the other predictors – of adverse outcomes among older adults,” and future areas for research include the development of novel behavioral interventions to improve sleep in this population.
The study was supported in part by the National Institute for Occupational Safety and Health; the National Heart, Lung, and Blood Institute; the National Institute on Aging; and the Brigham Research Institute Fund to Sustain Research Excellence. Lead author Dr. Robbins disclosed fees from Denihan Hospitality, Rituals Cosmetics, Dagmejan, Asystem, and SleepCycle. Several coauthors disclosed relationships with multiple pharmaceutical companies, and support from various philanthropic organizations.
More evidence has emerged linking sleep deficiency, dementia, and mortality.
“Sleep disturbance and insufficiency have been shown to be associated with both the development and progression of Alzheimer’s disease and with all-cause mortality,” wrote Rebecca S. Robbins, PhD, of Brigham and Women’s Hospital, Boston, and colleagues. However, research on this topic has yielded conflicting results, and “few studies have included a comprehensive set of sleep characteristics in a single examination of incident dementia and all-cause mortality.”
In a study published in Aging, the researchers identified 2,812 adults aged 65 years and older from the National Health and Aging Trends Study (NHATS), a nationally representative longitudinal study of Medicare beneficiaries aged 65 years and older in the United States.
Participants completed surveys about sleep disturbance and duration in 2013 (1,575 individuals) and in 2014 (1,237 individuals), and the researchers examined the relationship between sleep disturbance and deficiency and incident dementia and all-cause mortality over the next 5 years. The average age of the study participants was 76.9 years, 60% were women, and 72% were White.
Overall, approximately 60% of the participants reported never or rarely having problems with alertness, approximately half said that they rarely or never napped, and more than half said they fell asleep in 15 minutes or less. Approximately 70% rated their sleep quality as good or very good, and more than 90% said they rarely or never snored.
The researchers examined the relationships between sleep characteristics and the development of incident dementia over 5 years. In a fully adjusted Cox multivariate analysis, individuals who slept 5 hours or less per night had approximately twice the risk for incident dementia as those who slept longer (hazard ratio, 2.04); risk of dementia also was higher among those who took 30 minutes or longer to fall asleep (HR, 1.45).
In addition, the risk of all-cause mortality was significantly higher among individuals who reported difficulty maintaining alertness some days or most days/every day (HR, 1.49 and HR, 1.65, respectively), routinely napping some days or most days/every day (HR, 1.38 and HR, 1.73, respectively), poor or very poor sleep quality (HR, 1.75), and sleeping 5 hours or less each night (HR, 2.38).
The study findings were limited by several factors including a population representing only one-quarter of the NHATS cohort, which prevented nationally representative estimates, the availability of only 2 years of sleep data, and small sample size for certain response categories, the researchers noted.
However, “our study offers a contribution to the literature on sleep among aging populations in its assessment of incident dementia and all-cause mortality and a range of sleep characteristics among older adults,” they said. In particular, “short sleep duration was a strong predictor of both incident dementia and all-cause mortality, suggesting this may be a sleep characteristic that is important – over and above the other predictors – of adverse outcomes among older adults,” and future areas for research include the development of novel behavioral interventions to improve sleep in this population.
The study was supported in part by the National Institute for Occupational Safety and Health; the National Heart, Lung, and Blood Institute; the National Institute on Aging; and the Brigham Research Institute Fund to Sustain Research Excellence. Lead author Dr. Robbins disclosed fees from Denihan Hospitality, Rituals Cosmetics, Dagmejan, Asystem, and SleepCycle. Several coauthors disclosed relationships with multiple pharmaceutical companies, and support from various philanthropic organizations.
FROM AGING
When the X-Waiver gets X’ed: Implications for hospitalists
There are two pandemics permeating the United States: COVID-19 and addiction. To date, more than 468,000 people have died from COVID-19 in the U.S. In the 12-month period ending in May 2020, over 80,000 died from a drug related cause – the highest number ever recorded in a year. Many of these deaths involved opioids.
COVID-19 has worsened outcomes for people with addiction. There is less access to treatment, increased isolation, and worsening psychosocial and economic stressors. These factors may drive new, increased, or more risky substance use and return to use for people in recovery. As hospitalists, we have been responders in both COVID-19 and our country’s worsening overdose and addiction crisis.
In December 2020’s Journal of Hospital Medicine article “Converging Crises: Caring for hospitalized adults with substance use disorder in the time of COVID-19”, Dr. Honora Englander and her coauthors called on hospitalists to actively engage patients with substance use disorders during hospitalization. The article highlights the colliding crises of addiction and COVID-19 and provides eight practical approaches for hospitalists to address substance use disorders during the pandemic, including initiating buprenorphine for opioid withdrawal and prescribing it for opioid use disorder (OUD) treatment.
Buprenorphine effectively treats opioid withdrawal, reduces OUD-related mortality, and decreases hospital readmissions related to OUD. To prescribe buprenorphine for OUD in the outpatient setting or on hospital discharge, providers need an X-Waiver. The X-Waiver is a result of the Drug Addiction Treatment Act 2000 (DATA 2000), which was enacted in 2000. It permits physicians to prescribe buprenorphine for OUD treatment after an 8-hour training. In 2016, the Comprehensive Addiction and Recovery Act extended buprenorphine prescribing to physician assistants (PAs) and advanced-practice nurses (APNs). However, PAs and APNs are required to complete a 24-hour training to receive the waiver.
On Jan. 14, 2021, the U.S. Department of Health and Human Services under the Trump administration announced it was removing the X-Waiver training previously required for physicians to prescribe this life-saving medication. However, on Jan. 20, 2021, the Biden administration froze the training requirement removal pending a 60-day review. The excitement about the waiver’s eradication further dampened on Jan. 25, when the plan was halted due to procedural factors coupled with the concern that HHS may not have the authority to void requirements mandated by Congress.
Many of us continue to be hopeful that the X-Waiver will soon be gone. The Substance Abuse and Mental Health Services Administration has committed to working with federal agencies to increase access to buprenorphine. The Biden administration also committed to addressing our country’s addiction crisis, including a plan to “make effective prevention, treatment, and recovery services available to all, including through a $125 billion federal investment.”
Despite the pause on HHS’s recent attempt to “X the X-Waiver,” we now have renewed attention and interest in this critical issue and an opportunity for greater and longer-lasting legislative impact. SHM supports that Congress repeal the legislative requirement for buprenorphine training dictated by DATA 2000 so that it cannot be rolled back by future administrations. To further increase access to buprenorphine treatment, the training requirement should be removed for all providers who care for individuals with OUD.
The X-Waiver has been a barrier to hospitalist adoption of this critical, life-saving medication. HHS’s stance to nix the waiver, though fleeting, should be interpreted as an urgent call to the medical community, including us as hospitalists, to learn about buprenorphine with the many resources available (see table 1). As hospital medicine providers, we can order buprenorphine for patients with OUD during hospitalization. It is discharge prescriptions that have been limited to providers with an X-Waiver.
What can we do now to prepare for the eventual X-Waiver training removal? We can start by educating ourselves with the resources listed in table 1. Those of us who are already buprenorphine champions could lead trainings in our home institutions. In a future without the waiver there will be more flexibility to develop hospitalist-focused buprenorphine trainings, as the previous ones were geared for outpatient providers. Hospitalist organizations could support hospitalist-specific buprenorphine trainings and extend the models to include additional medications for addiction.
There is a large body of evidence regarding buprenorphine’s safety and efficacy in OUD treatment. With a worsening overdose crisis, there have been increasing opioid-related hospitalizations. When new medications for diabetes, hypertension, or DVT treatment become available, as hospitalists we incorporate them into our toolbox. As buprenorphine becomes more accessible, we can be leaders in further adopting it (and other substance use disorder medications while we are at it) as our standard of care for people with OUD.
Dr. Bottner is a physician assistant in the Division of Hospital Medicine at Dell Medical School at The University of Texas at Austin and director of the hospital’s Buprenorphine Team. Dr. Martin is a board-certified addiction medicine physician and hospitalist at University of California, San Francisco, and director of the Addiction Care Team at San Francisco General Hospital. Dr. Bottner and Dr. Martin colead the SHM Substance Use Disorder Special Interest Group.
There are two pandemics permeating the United States: COVID-19 and addiction. To date, more than 468,000 people have died from COVID-19 in the U.S. In the 12-month period ending in May 2020, over 80,000 died from a drug related cause – the highest number ever recorded in a year. Many of these deaths involved opioids.
COVID-19 has worsened outcomes for people with addiction. There is less access to treatment, increased isolation, and worsening psychosocial and economic stressors. These factors may drive new, increased, or more risky substance use and return to use for people in recovery. As hospitalists, we have been responders in both COVID-19 and our country’s worsening overdose and addiction crisis.
In December 2020’s Journal of Hospital Medicine article “Converging Crises: Caring for hospitalized adults with substance use disorder in the time of COVID-19”, Dr. Honora Englander and her coauthors called on hospitalists to actively engage patients with substance use disorders during hospitalization. The article highlights the colliding crises of addiction and COVID-19 and provides eight practical approaches for hospitalists to address substance use disorders during the pandemic, including initiating buprenorphine for opioid withdrawal and prescribing it for opioid use disorder (OUD) treatment.
Buprenorphine effectively treats opioid withdrawal, reduces OUD-related mortality, and decreases hospital readmissions related to OUD. To prescribe buprenorphine for OUD in the outpatient setting or on hospital discharge, providers need an X-Waiver. The X-Waiver is a result of the Drug Addiction Treatment Act 2000 (DATA 2000), which was enacted in 2000. It permits physicians to prescribe buprenorphine for OUD treatment after an 8-hour training. In 2016, the Comprehensive Addiction and Recovery Act extended buprenorphine prescribing to physician assistants (PAs) and advanced-practice nurses (APNs). However, PAs and APNs are required to complete a 24-hour training to receive the waiver.
On Jan. 14, 2021, the U.S. Department of Health and Human Services under the Trump administration announced it was removing the X-Waiver training previously required for physicians to prescribe this life-saving medication. However, on Jan. 20, 2021, the Biden administration froze the training requirement removal pending a 60-day review. The excitement about the waiver’s eradication further dampened on Jan. 25, when the plan was halted due to procedural factors coupled with the concern that HHS may not have the authority to void requirements mandated by Congress.
Many of us continue to be hopeful that the X-Waiver will soon be gone. The Substance Abuse and Mental Health Services Administration has committed to working with federal agencies to increase access to buprenorphine. The Biden administration also committed to addressing our country’s addiction crisis, including a plan to “make effective prevention, treatment, and recovery services available to all, including through a $125 billion federal investment.”
Despite the pause on HHS’s recent attempt to “X the X-Waiver,” we now have renewed attention and interest in this critical issue and an opportunity for greater and longer-lasting legislative impact. SHM supports that Congress repeal the legislative requirement for buprenorphine training dictated by DATA 2000 so that it cannot be rolled back by future administrations. To further increase access to buprenorphine treatment, the training requirement should be removed for all providers who care for individuals with OUD.
The X-Waiver has been a barrier to hospitalist adoption of this critical, life-saving medication. HHS’s stance to nix the waiver, though fleeting, should be interpreted as an urgent call to the medical community, including us as hospitalists, to learn about buprenorphine with the many resources available (see table 1). As hospital medicine providers, we can order buprenorphine for patients with OUD during hospitalization. It is discharge prescriptions that have been limited to providers with an X-Waiver.
What can we do now to prepare for the eventual X-Waiver training removal? We can start by educating ourselves with the resources listed in table 1. Those of us who are already buprenorphine champions could lead trainings in our home institutions. In a future without the waiver there will be more flexibility to develop hospitalist-focused buprenorphine trainings, as the previous ones were geared for outpatient providers. Hospitalist organizations could support hospitalist-specific buprenorphine trainings and extend the models to include additional medications for addiction.
There is a large body of evidence regarding buprenorphine’s safety and efficacy in OUD treatment. With a worsening overdose crisis, there have been increasing opioid-related hospitalizations. When new medications for diabetes, hypertension, or DVT treatment become available, as hospitalists we incorporate them into our toolbox. As buprenorphine becomes more accessible, we can be leaders in further adopting it (and other substance use disorder medications while we are at it) as our standard of care for people with OUD.
Dr. Bottner is a physician assistant in the Division of Hospital Medicine at Dell Medical School at The University of Texas at Austin and director of the hospital’s Buprenorphine Team. Dr. Martin is a board-certified addiction medicine physician and hospitalist at University of California, San Francisco, and director of the Addiction Care Team at San Francisco General Hospital. Dr. Bottner and Dr. Martin colead the SHM Substance Use Disorder Special Interest Group.
There are two pandemics permeating the United States: COVID-19 and addiction. To date, more than 468,000 people have died from COVID-19 in the U.S. In the 12-month period ending in May 2020, over 80,000 died from a drug related cause – the highest number ever recorded in a year. Many of these deaths involved opioids.
COVID-19 has worsened outcomes for people with addiction. There is less access to treatment, increased isolation, and worsening psychosocial and economic stressors. These factors may drive new, increased, or more risky substance use and return to use for people in recovery. As hospitalists, we have been responders in both COVID-19 and our country’s worsening overdose and addiction crisis.
In December 2020’s Journal of Hospital Medicine article “Converging Crises: Caring for hospitalized adults with substance use disorder in the time of COVID-19”, Dr. Honora Englander and her coauthors called on hospitalists to actively engage patients with substance use disorders during hospitalization. The article highlights the colliding crises of addiction and COVID-19 and provides eight practical approaches for hospitalists to address substance use disorders during the pandemic, including initiating buprenorphine for opioid withdrawal and prescribing it for opioid use disorder (OUD) treatment.
Buprenorphine effectively treats opioid withdrawal, reduces OUD-related mortality, and decreases hospital readmissions related to OUD. To prescribe buprenorphine for OUD in the outpatient setting or on hospital discharge, providers need an X-Waiver. The X-Waiver is a result of the Drug Addiction Treatment Act 2000 (DATA 2000), which was enacted in 2000. It permits physicians to prescribe buprenorphine for OUD treatment after an 8-hour training. In 2016, the Comprehensive Addiction and Recovery Act extended buprenorphine prescribing to physician assistants (PAs) and advanced-practice nurses (APNs). However, PAs and APNs are required to complete a 24-hour training to receive the waiver.
On Jan. 14, 2021, the U.S. Department of Health and Human Services under the Trump administration announced it was removing the X-Waiver training previously required for physicians to prescribe this life-saving medication. However, on Jan. 20, 2021, the Biden administration froze the training requirement removal pending a 60-day review. The excitement about the waiver’s eradication further dampened on Jan. 25, when the plan was halted due to procedural factors coupled with the concern that HHS may not have the authority to void requirements mandated by Congress.
Many of us continue to be hopeful that the X-Waiver will soon be gone. The Substance Abuse and Mental Health Services Administration has committed to working with federal agencies to increase access to buprenorphine. The Biden administration also committed to addressing our country’s addiction crisis, including a plan to “make effective prevention, treatment, and recovery services available to all, including through a $125 billion federal investment.”
Despite the pause on HHS’s recent attempt to “X the X-Waiver,” we now have renewed attention and interest in this critical issue and an opportunity for greater and longer-lasting legislative impact. SHM supports that Congress repeal the legislative requirement for buprenorphine training dictated by DATA 2000 so that it cannot be rolled back by future administrations. To further increase access to buprenorphine treatment, the training requirement should be removed for all providers who care for individuals with OUD.
The X-Waiver has been a barrier to hospitalist adoption of this critical, life-saving medication. HHS’s stance to nix the waiver, though fleeting, should be interpreted as an urgent call to the medical community, including us as hospitalists, to learn about buprenorphine with the many resources available (see table 1). As hospital medicine providers, we can order buprenorphine for patients with OUD during hospitalization. It is discharge prescriptions that have been limited to providers with an X-Waiver.
What can we do now to prepare for the eventual X-Waiver training removal? We can start by educating ourselves with the resources listed in table 1. Those of us who are already buprenorphine champions could lead trainings in our home institutions. In a future without the waiver there will be more flexibility to develop hospitalist-focused buprenorphine trainings, as the previous ones were geared for outpatient providers. Hospitalist organizations could support hospitalist-specific buprenorphine trainings and extend the models to include additional medications for addiction.
There is a large body of evidence regarding buprenorphine’s safety and efficacy in OUD treatment. With a worsening overdose crisis, there have been increasing opioid-related hospitalizations. When new medications for diabetes, hypertension, or DVT treatment become available, as hospitalists we incorporate them into our toolbox. As buprenorphine becomes more accessible, we can be leaders in further adopting it (and other substance use disorder medications while we are at it) as our standard of care for people with OUD.
Dr. Bottner is a physician assistant in the Division of Hospital Medicine at Dell Medical School at The University of Texas at Austin and director of the hospital’s Buprenorphine Team. Dr. Martin is a board-certified addiction medicine physician and hospitalist at University of California, San Francisco, and director of the Addiction Care Team at San Francisco General Hospital. Dr. Bottner and Dr. Martin colead the SHM Substance Use Disorder Special Interest Group.
Opioids prescribed for diabetic neuropathy pain, against advice
Prescriptions for opioids as a first-line treatment for painful diabetic peripheral neuropathy (DPN) outnumbered those for other medications between 2014 and 2018, despite the fact that the former is not recommended, new research indicates.
“We know that for any kind of chronic pain, opioids are not ideal. They’re not very effective for chronic pain in general, and they’re definitely not safe,” senior author Rozalina G. McCoy, MD, an endocrinologist and primary care clinician at the Mayo Clinic in Rochester, Minn., told this news organization.
That’s true even for severe DPN pain or painful exacerbations, she added.
“There’s a myth that opioids are the strongest pain meds possible ... For painful neuropathic pain, duloxetine [Cymbalta], pregabalin [Lyrica], and gabapentin [Neurontin] are the most effective pain medications based on multiple studies and extensive experience using them,” she explained. “But I think the public perception is that opioids are the strongest. When a patient comes with severe pain, I think there’s that kind of gut feeling that if the pain is severe, I need to give opioids.”
What’s more, she noted, “evidence is emerging for other harms, not only the potential for dependency and potential overdose, but also the potential for opioid-induced hyperalgesia. Opioids themselves can cause chronic pain. When we think about using opioids for chronic pain, we are really shooting ourselves in the foot. We’re going to harm patients.”
The American Diabetes Association DPN guidelines essentially say as much, advising opioids only as a tertiary option for refractory pain, she observed.
The new findings, from a retrospective study of Mayo Clinic electronic health data, were published online in JAMA Network Open by Jungwei Fan, PhD, also of Mayo Clinic, and colleagues.
Are fewer patients with DPN receiving any treatment now?
The data also reveal that, while opioid prescribing dropped over the study period, there wasn’t a comparable rise in prescriptions of recommended pain medications, suggesting that recent efforts to minimize opioid prescribing may have resulted in less overall treatment of significant pain. (The study had to be stopped in 2018 when Mayo switched to a new electronic health record system, Dr. McCoy explained.)
“The proportion of opioids among new prescriptions has been decreasing. I’m hopeful that the rates are even lower now than they were 2 years ago. What was concerning to me was the proportion of people receiving treatment overall had gone down,” Dr. McCoy noted.
“So, while it’s great that opioids aren’t being used, it’s doubtful that people with DPN are any less symptomatic. So I worry that there’s a proportion of patients who have pain who aren’t getting the treatment they need just because we don’t want to give them opioids. There are other options,” Dr. McCoy said, including nonpharmacologic approaches.
Opioids dominated in new-onset DPN prescribing during 2014-2018
The study involved 3,495 adults with newly diagnosed DPN from all three Mayo Clinic locations in Rochester, Minn.; Phoenix, Ariz.; and Jacksonville, Fla. during the period 2014-2018. Of those, 40.2% (1,406) were prescribed a new pain medication after diagnosis. However, that proportion dropped from 45.6% in 2014 to 35.2% in 2018.
The odds of initiating any treatment were significantly greater among patients with depression (odds ratio, 1.61), arthritis (OR, 1.21), and back pain (OR, 1.34), but decreased over time among all patients.
Among those receiving drug treatment, opioids were prescribed to 43.8%, whereas guideline-recommended medications (gabapentin, pregabalin, and serotonin norepinephrine reuptake inhibitors including duloxetine) were prescribed to 42.9%.
Another 20.6% received medications deemed “acceptable” for treating neuropathic pain, including topical analgesics, tricyclic antidepressants, and other anticonvulsants.
Males were significantly more likely than females to receive opioids (OR, 1.26), while individuals diagnosed with comorbid fibromyalgia were less likely (OR, 0.67). Those with comorbid arthritis were less likely to receive recommended DPN medications (OR, 0.76).
Use of opioids was 29% less likely in 2018, compared with 2014, although this difference did not achieve significance. Similarly, use of recommended medications was 25% more likely in 2018, compared with 2014, also not a significant difference.
Dr. McCoy offers clinical pearls for treating pain in DPN
Clinically, Dr. McCoy said that she individualizes treatment for painful DPN.
“I tend to use duloxetine if the patient also has a mood disorder including depression or anxiety, because it can also help with that. Gabapentin can also be helpful for radiculopathy or for chronic low-back pain. It can even help with degenerative joint disease like arthritis of the knees. So, you maximize benefit if you use one drug to treat multiple things.”
All three recommended medications are generic now, although pregabalin still tends to be more expensive, she noted. Gabapentin can cause drowsiness, which makes it ideal for a patient with insomnia but much less so for a long-haul truck driver. Duloxetine doesn’t cause sleepiness. Pregabalin can, but less so than gabapentin.
“I think that’s why it’s so important to talk to your patient and ask how the neuropathy is affecting them. What other comorbidities do they have? What is their life like? I think you have to figure out what drug works for each individual person.”
Importantly, she advised, if one of the three doesn’t work, stop it and try another. “It doesn’t mean that none of these meds work. All three should be tried to see if they give relief.”
Nonpharmacologic measures such as cognitive behavioral therapy, acupuncture, or physical therapy may help some patients as well.
Supplements such as vitamin B12 – which can also help with metformin-induced B12 deficiency – or alpha-lipoic acid may also be worth a try as long as the patient is made aware of potential risks, she noted.
Dr. McCoy hopes to repeat this study using national data. “I don’t think this is isolated to Mayo ... I think it affects all practices,” she said.
Since the study, “we [Mayo Clinic] have implemented practice changes to limit use of opioids for chronic pain ... so I hope it’s getting better. It’s important to be aware of our patterns in prescribing.”
The study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. McCoy reported receiving grants from the AARP Quality Measure Innovation program through a collaboration with OptumLabs and the Mayo Clinic’s Robert D. and Patricia E. Kern Center for the Science of Health Care Delivery.
A version of this article first appeared on Medscape.com.
Prescriptions for opioids as a first-line treatment for painful diabetic peripheral neuropathy (DPN) outnumbered those for other medications between 2014 and 2018, despite the fact that the former is not recommended, new research indicates.
“We know that for any kind of chronic pain, opioids are not ideal. They’re not very effective for chronic pain in general, and they’re definitely not safe,” senior author Rozalina G. McCoy, MD, an endocrinologist and primary care clinician at the Mayo Clinic in Rochester, Minn., told this news organization.
That’s true even for severe DPN pain or painful exacerbations, she added.
“There’s a myth that opioids are the strongest pain meds possible ... For painful neuropathic pain, duloxetine [Cymbalta], pregabalin [Lyrica], and gabapentin [Neurontin] are the most effective pain medications based on multiple studies and extensive experience using them,” she explained. “But I think the public perception is that opioids are the strongest. When a patient comes with severe pain, I think there’s that kind of gut feeling that if the pain is severe, I need to give opioids.”
What’s more, she noted, “evidence is emerging for other harms, not only the potential for dependency and potential overdose, but also the potential for opioid-induced hyperalgesia. Opioids themselves can cause chronic pain. When we think about using opioids for chronic pain, we are really shooting ourselves in the foot. We’re going to harm patients.”
The American Diabetes Association DPN guidelines essentially say as much, advising opioids only as a tertiary option for refractory pain, she observed.
The new findings, from a retrospective study of Mayo Clinic electronic health data, were published online in JAMA Network Open by Jungwei Fan, PhD, also of Mayo Clinic, and colleagues.
Are fewer patients with DPN receiving any treatment now?
The data also reveal that, while opioid prescribing dropped over the study period, there wasn’t a comparable rise in prescriptions of recommended pain medications, suggesting that recent efforts to minimize opioid prescribing may have resulted in less overall treatment of significant pain. (The study had to be stopped in 2018 when Mayo switched to a new electronic health record system, Dr. McCoy explained.)
“The proportion of opioids among new prescriptions has been decreasing. I’m hopeful that the rates are even lower now than they were 2 years ago. What was concerning to me was the proportion of people receiving treatment overall had gone down,” Dr. McCoy noted.
“So, while it’s great that opioids aren’t being used, it’s doubtful that people with DPN are any less symptomatic. So I worry that there’s a proportion of patients who have pain who aren’t getting the treatment they need just because we don’t want to give them opioids. There are other options,” Dr. McCoy said, including nonpharmacologic approaches.
Opioids dominated in new-onset DPN prescribing during 2014-2018
The study involved 3,495 adults with newly diagnosed DPN from all three Mayo Clinic locations in Rochester, Minn.; Phoenix, Ariz.; and Jacksonville, Fla. during the period 2014-2018. Of those, 40.2% (1,406) were prescribed a new pain medication after diagnosis. However, that proportion dropped from 45.6% in 2014 to 35.2% in 2018.
The odds of initiating any treatment were significantly greater among patients with depression (odds ratio, 1.61), arthritis (OR, 1.21), and back pain (OR, 1.34), but decreased over time among all patients.
Among those receiving drug treatment, opioids were prescribed to 43.8%, whereas guideline-recommended medications (gabapentin, pregabalin, and serotonin norepinephrine reuptake inhibitors including duloxetine) were prescribed to 42.9%.
Another 20.6% received medications deemed “acceptable” for treating neuropathic pain, including topical analgesics, tricyclic antidepressants, and other anticonvulsants.
Males were significantly more likely than females to receive opioids (OR, 1.26), while individuals diagnosed with comorbid fibromyalgia were less likely (OR, 0.67). Those with comorbid arthritis were less likely to receive recommended DPN medications (OR, 0.76).
Use of opioids was 29% less likely in 2018, compared with 2014, although this difference did not achieve significance. Similarly, use of recommended medications was 25% more likely in 2018, compared with 2014, also not a significant difference.
Dr. McCoy offers clinical pearls for treating pain in DPN
Clinically, Dr. McCoy said that she individualizes treatment for painful DPN.
“I tend to use duloxetine if the patient also has a mood disorder including depression or anxiety, because it can also help with that. Gabapentin can also be helpful for radiculopathy or for chronic low-back pain. It can even help with degenerative joint disease like arthritis of the knees. So, you maximize benefit if you use one drug to treat multiple things.”
All three recommended medications are generic now, although pregabalin still tends to be more expensive, she noted. Gabapentin can cause drowsiness, which makes it ideal for a patient with insomnia but much less so for a long-haul truck driver. Duloxetine doesn’t cause sleepiness. Pregabalin can, but less so than gabapentin.
“I think that’s why it’s so important to talk to your patient and ask how the neuropathy is affecting them. What other comorbidities do they have? What is their life like? I think you have to figure out what drug works for each individual person.”
Importantly, she advised, if one of the three doesn’t work, stop it and try another. “It doesn’t mean that none of these meds work. All three should be tried to see if they give relief.”
Nonpharmacologic measures such as cognitive behavioral therapy, acupuncture, or physical therapy may help some patients as well.
Supplements such as vitamin B12 – which can also help with metformin-induced B12 deficiency – or alpha-lipoic acid may also be worth a try as long as the patient is made aware of potential risks, she noted.
Dr. McCoy hopes to repeat this study using national data. “I don’t think this is isolated to Mayo ... I think it affects all practices,” she said.
Since the study, “we [Mayo Clinic] have implemented practice changes to limit use of opioids for chronic pain ... so I hope it’s getting better. It’s important to be aware of our patterns in prescribing.”
The study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. McCoy reported receiving grants from the AARP Quality Measure Innovation program through a collaboration with OptumLabs and the Mayo Clinic’s Robert D. and Patricia E. Kern Center for the Science of Health Care Delivery.
A version of this article first appeared on Medscape.com.
Prescriptions for opioids as a first-line treatment for painful diabetic peripheral neuropathy (DPN) outnumbered those for other medications between 2014 and 2018, despite the fact that the former is not recommended, new research indicates.
“We know that for any kind of chronic pain, opioids are not ideal. They’re not very effective for chronic pain in general, and they’re definitely not safe,” senior author Rozalina G. McCoy, MD, an endocrinologist and primary care clinician at the Mayo Clinic in Rochester, Minn., told this news organization.
That’s true even for severe DPN pain or painful exacerbations, she added.
“There’s a myth that opioids are the strongest pain meds possible ... For painful neuropathic pain, duloxetine [Cymbalta], pregabalin [Lyrica], and gabapentin [Neurontin] are the most effective pain medications based on multiple studies and extensive experience using them,” she explained. “But I think the public perception is that opioids are the strongest. When a patient comes with severe pain, I think there’s that kind of gut feeling that if the pain is severe, I need to give opioids.”
What’s more, she noted, “evidence is emerging for other harms, not only the potential for dependency and potential overdose, but also the potential for opioid-induced hyperalgesia. Opioids themselves can cause chronic pain. When we think about using opioids for chronic pain, we are really shooting ourselves in the foot. We’re going to harm patients.”
The American Diabetes Association DPN guidelines essentially say as much, advising opioids only as a tertiary option for refractory pain, she observed.
The new findings, from a retrospective study of Mayo Clinic electronic health data, were published online in JAMA Network Open by Jungwei Fan, PhD, also of Mayo Clinic, and colleagues.
Are fewer patients with DPN receiving any treatment now?
The data also reveal that, while opioid prescribing dropped over the study period, there wasn’t a comparable rise in prescriptions of recommended pain medications, suggesting that recent efforts to minimize opioid prescribing may have resulted in less overall treatment of significant pain. (The study had to be stopped in 2018 when Mayo switched to a new electronic health record system, Dr. McCoy explained.)
“The proportion of opioids among new prescriptions has been decreasing. I’m hopeful that the rates are even lower now than they were 2 years ago. What was concerning to me was the proportion of people receiving treatment overall had gone down,” Dr. McCoy noted.
“So, while it’s great that opioids aren’t being used, it’s doubtful that people with DPN are any less symptomatic. So I worry that there’s a proportion of patients who have pain who aren’t getting the treatment they need just because we don’t want to give them opioids. There are other options,” Dr. McCoy said, including nonpharmacologic approaches.
Opioids dominated in new-onset DPN prescribing during 2014-2018
The study involved 3,495 adults with newly diagnosed DPN from all three Mayo Clinic locations in Rochester, Minn.; Phoenix, Ariz.; and Jacksonville, Fla. during the period 2014-2018. Of those, 40.2% (1,406) were prescribed a new pain medication after diagnosis. However, that proportion dropped from 45.6% in 2014 to 35.2% in 2018.
The odds of initiating any treatment were significantly greater among patients with depression (odds ratio, 1.61), arthritis (OR, 1.21), and back pain (OR, 1.34), but decreased over time among all patients.
Among those receiving drug treatment, opioids were prescribed to 43.8%, whereas guideline-recommended medications (gabapentin, pregabalin, and serotonin norepinephrine reuptake inhibitors including duloxetine) were prescribed to 42.9%.
Another 20.6% received medications deemed “acceptable” for treating neuropathic pain, including topical analgesics, tricyclic antidepressants, and other anticonvulsants.
Males were significantly more likely than females to receive opioids (OR, 1.26), while individuals diagnosed with comorbid fibromyalgia were less likely (OR, 0.67). Those with comorbid arthritis were less likely to receive recommended DPN medications (OR, 0.76).
Use of opioids was 29% less likely in 2018, compared with 2014, although this difference did not achieve significance. Similarly, use of recommended medications was 25% more likely in 2018, compared with 2014, also not a significant difference.
Dr. McCoy offers clinical pearls for treating pain in DPN
Clinically, Dr. McCoy said that she individualizes treatment for painful DPN.
“I tend to use duloxetine if the patient also has a mood disorder including depression or anxiety, because it can also help with that. Gabapentin can also be helpful for radiculopathy or for chronic low-back pain. It can even help with degenerative joint disease like arthritis of the knees. So, you maximize benefit if you use one drug to treat multiple things.”
All three recommended medications are generic now, although pregabalin still tends to be more expensive, she noted. Gabapentin can cause drowsiness, which makes it ideal for a patient with insomnia but much less so for a long-haul truck driver. Duloxetine doesn’t cause sleepiness. Pregabalin can, but less so than gabapentin.
“I think that’s why it’s so important to talk to your patient and ask how the neuropathy is affecting them. What other comorbidities do they have? What is their life like? I think you have to figure out what drug works for each individual person.”
Importantly, she advised, if one of the three doesn’t work, stop it and try another. “It doesn’t mean that none of these meds work. All three should be tried to see if they give relief.”
Nonpharmacologic measures such as cognitive behavioral therapy, acupuncture, or physical therapy may help some patients as well.
Supplements such as vitamin B12 – which can also help with metformin-induced B12 deficiency – or alpha-lipoic acid may also be worth a try as long as the patient is made aware of potential risks, she noted.
Dr. McCoy hopes to repeat this study using national data. “I don’t think this is isolated to Mayo ... I think it affects all practices,” she said.
Since the study, “we [Mayo Clinic] have implemented practice changes to limit use of opioids for chronic pain ... so I hope it’s getting better. It’s important to be aware of our patterns in prescribing.”
The study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. McCoy reported receiving grants from the AARP Quality Measure Innovation program through a collaboration with OptumLabs and the Mayo Clinic’s Robert D. and Patricia E. Kern Center for the Science of Health Care Delivery.
A version of this article first appeared on Medscape.com.
Prostate drugs tied to lower risk for Parkinson’s disease
new research suggests. Treatment of BPH with terazosin (Hytrin), doxazosin (Cardura), or alfuzosin (Uroxatral), all of which enhance glycolysis, was associated with a lower risk of developing Parkinson’s disease than patients taking a drug used for the same indication, tamsulosin (Flomax), which does not affect glycolysis.
“If giving someone terazosin or similar medications truly reduces their risk of disease, these results could have significant clinical implications for neurologists,” said lead author Jacob E. Simmering, PhD, assistant professor of internal medicine at the University of Iowa, Iowa City.
There are few reliable neuroprotective treatments for Parkinson’s disease, he said. “We can manage some of the symptoms, but we can’t stop it from progressing. If a randomized trial finds the same result, this will provide a new option to slow progression of Parkinson’s disease.”
The pathogenesis of Parkinson’s disease is heterogeneous, however, and not all patients may benefit from glycolysis-enhancing drugs, the investigators noted. Future research will be needed to identify potential candidates for this treatment, and clarify the effects of these drugs, they wrote.
The findings were published online Feb. 1, 2021, in JAMA Neurology.
Time-dependent effects
The major risk factor for Parkinson’s disease is age, which is associated with impaired energy metabolism. Glycolysis is decreased among patients with Parkinson’s disease, yet impaired energy metabolism has not been investigated widely as a pathogenic factor in the disease, the authors wrote.
Studies have indicated that terazosin increases the activity of an enzyme important in glycolysis. Doxazosin and alfuzosin have a similar mechanism of action and enhance energy metabolism. Tamsulosin, a structurally unrelated drug, has the same mechanism of action as the other three drugs, but does not enhance energy metabolism.
In this report, the researchers investigated the hypothesis that patients who received therapy with terazosin, doxazosin, or alfuzosin would have a lower risk of developing Parkinson’s disease than patients receiving tamsulosin. To do that, they used health care utilization data from Denmark and the United States, including the Danish National Prescription Registry, the Danish National Patient Registry, the Danish Civil Registration System, and the Truven Health Analytics MarketScan database.
The investigators searched the records for patients who filled prescriptions for any of the four drugs of interest. They excluded any patients who developed Parkinson’s disease within 1 year of starting medication. Because use of these drugs is rare among women, they included only men in their analysis.
They looked at patient outcomes beginning at 1 year after the initiation of treatment. They also required patients to fill at least two prescriptions before the beginning of follow-up. Patients who switched from tamsulosin to any of the other drugs, or vice versa, were excluded from analysis.
The investigators used propensity-score matching to ensure that patients in the tamsulosin and terazosin/doxazosin/alfuzosin groups were similar in terms of their other potential risk factors. The primary outcome was the development of Parkinson’s disease.
They identified 52,365 propensity score–matched pairs in the Danish registries and 94,883 pairs in the Truven database. The mean age was 67.9 years in the Danish registries and 63.8 years in the Truven database, and follow-up was approximately 5 years and 3 years respectively. Baseline covariates were well balanced between cohorts.
Among Danish patients, those who took terazosin, doxazosin, or alfuzosin had a lower risk of developing Parkinson’s disease versus those who took tamsulosin (hazard ratio, 0.88). Similarly, patients in the Truven database who took terazosin, doxazosin, or alfuzosin had a lower risk of developing Parkinson’s disease than those who took tamsulosin (HR, 0.63).
In both cohorts, the risk for Parkinson’s disease among patients receiving terazosin, doxazosin, or alfuzosin, compared with those receiving tamsulosin, decreased with increasing numbers of prescriptions filled. Long-term treatment with any of the three glycolysis-enhancing drugs was associated with greater risk reduction in the Danish (HR, 0.79) and Truven (HR, 0.46) cohorts versus tamsulosin.
Differences in case definitions, which may reflect how Parkinson’s disease was managed, complicate comparisons between the Danish and Truven cohorts, said Dr. Simmering. Another challenge is the source of the data. “The Truven data set was derived from insurance claims from people with private insurance or Medicare supplemental plans,” he said. “This group is quite large but may not be representative of everyone in the United States. We would also only be able to follow people while they were on one insurance plan. If they switched coverage to a company that doesn’t contribute data, we would lose them.”
The Danish database, however, includes all residents of Denmark. Only people who left the country were lost to follow-up.
The results support the hypothesis that increasing energy in cells slows disease progression, Dr. Simmering added. “There are a few conditions, mostly REM sleep disorders, that are associated with future diagnosis of Parkinson’s disease. Right now, we don’t have anything to offer people at elevated risk of Parkinson’s disease that might prevent the disease. If a controlled trial finds that terazosin slows or prevents Parkinson’s disease, we would have something truly protective to offer these patients.”
Biomarker needed
Commenting on the results, Alberto J. Espay, MD, MSc, professor of neurology at the University of Cincinnati Academic Health Center, was cautious. “These findings are of unclear applicability to any particular patient without a biomarker for a deficit of glycolysis that these drugs are presumed to affect,” Dr. Espay said. “Hence, there is no feasible or warranted change in practice as a result of this study.”
Pathogenic mechanisms are heterogeneous among patients with Parkinson’s disease, Dr. Espay added. “We will need to understand who among the large biological universe of Parkinson’s patients may have impaired energy metabolism as a pathogenic mechanism to be selected for a future clinical trial evaluating terazosin, doxazosin, or alfuzosin as a potential disease-modifying intervention.”
Parkinson’s disease is not one disease, but a group of disorders with unique biological abnormalities, said Dr. Espay. “We know so much about ‘Parkinson’s disease’ and next to nothing about the biology of individuals with Parkinson’s disease.”
This situation has enabled the development of symptomatic treatments, such as dopaminergic therapies, but failed to yield disease-modifying treatments, he said.
The University of Iowa contributed funds for this study. Dr. Simmering has received pilot funding from the University of Iowa Institute for Clinical and Translational Science. He had no conflicts of interest to disclose. Dr. Espay disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests. Treatment of BPH with terazosin (Hytrin), doxazosin (Cardura), or alfuzosin (Uroxatral), all of which enhance glycolysis, was associated with a lower risk of developing Parkinson’s disease than patients taking a drug used for the same indication, tamsulosin (Flomax), which does not affect glycolysis.
“If giving someone terazosin or similar medications truly reduces their risk of disease, these results could have significant clinical implications for neurologists,” said lead author Jacob E. Simmering, PhD, assistant professor of internal medicine at the University of Iowa, Iowa City.
There are few reliable neuroprotective treatments for Parkinson’s disease, he said. “We can manage some of the symptoms, but we can’t stop it from progressing. If a randomized trial finds the same result, this will provide a new option to slow progression of Parkinson’s disease.”
The pathogenesis of Parkinson’s disease is heterogeneous, however, and not all patients may benefit from glycolysis-enhancing drugs, the investigators noted. Future research will be needed to identify potential candidates for this treatment, and clarify the effects of these drugs, they wrote.
The findings were published online Feb. 1, 2021, in JAMA Neurology.
Time-dependent effects
The major risk factor for Parkinson’s disease is age, which is associated with impaired energy metabolism. Glycolysis is decreased among patients with Parkinson’s disease, yet impaired energy metabolism has not been investigated widely as a pathogenic factor in the disease, the authors wrote.
Studies have indicated that terazosin increases the activity of an enzyme important in glycolysis. Doxazosin and alfuzosin have a similar mechanism of action and enhance energy metabolism. Tamsulosin, a structurally unrelated drug, has the same mechanism of action as the other three drugs, but does not enhance energy metabolism.
In this report, the researchers investigated the hypothesis that patients who received therapy with terazosin, doxazosin, or alfuzosin would have a lower risk of developing Parkinson’s disease than patients receiving tamsulosin. To do that, they used health care utilization data from Denmark and the United States, including the Danish National Prescription Registry, the Danish National Patient Registry, the Danish Civil Registration System, and the Truven Health Analytics MarketScan database.
The investigators searched the records for patients who filled prescriptions for any of the four drugs of interest. They excluded any patients who developed Parkinson’s disease within 1 year of starting medication. Because use of these drugs is rare among women, they included only men in their analysis.
They looked at patient outcomes beginning at 1 year after the initiation of treatment. They also required patients to fill at least two prescriptions before the beginning of follow-up. Patients who switched from tamsulosin to any of the other drugs, or vice versa, were excluded from analysis.
The investigators used propensity-score matching to ensure that patients in the tamsulosin and terazosin/doxazosin/alfuzosin groups were similar in terms of their other potential risk factors. The primary outcome was the development of Parkinson’s disease.
They identified 52,365 propensity score–matched pairs in the Danish registries and 94,883 pairs in the Truven database. The mean age was 67.9 years in the Danish registries and 63.8 years in the Truven database, and follow-up was approximately 5 years and 3 years respectively. Baseline covariates were well balanced between cohorts.
Among Danish patients, those who took terazosin, doxazosin, or alfuzosin had a lower risk of developing Parkinson’s disease versus those who took tamsulosin (hazard ratio, 0.88). Similarly, patients in the Truven database who took terazosin, doxazosin, or alfuzosin had a lower risk of developing Parkinson’s disease than those who took tamsulosin (HR, 0.63).
In both cohorts, the risk for Parkinson’s disease among patients receiving terazosin, doxazosin, or alfuzosin, compared with those receiving tamsulosin, decreased with increasing numbers of prescriptions filled. Long-term treatment with any of the three glycolysis-enhancing drugs was associated with greater risk reduction in the Danish (HR, 0.79) and Truven (HR, 0.46) cohorts versus tamsulosin.
Differences in case definitions, which may reflect how Parkinson’s disease was managed, complicate comparisons between the Danish and Truven cohorts, said Dr. Simmering. Another challenge is the source of the data. “The Truven data set was derived from insurance claims from people with private insurance or Medicare supplemental plans,” he said. “This group is quite large but may not be representative of everyone in the United States. We would also only be able to follow people while they were on one insurance plan. If they switched coverage to a company that doesn’t contribute data, we would lose them.”
The Danish database, however, includes all residents of Denmark. Only people who left the country were lost to follow-up.
The results support the hypothesis that increasing energy in cells slows disease progression, Dr. Simmering added. “There are a few conditions, mostly REM sleep disorders, that are associated with future diagnosis of Parkinson’s disease. Right now, we don’t have anything to offer people at elevated risk of Parkinson’s disease that might prevent the disease. If a controlled trial finds that terazosin slows or prevents Parkinson’s disease, we would have something truly protective to offer these patients.”
Biomarker needed
Commenting on the results, Alberto J. Espay, MD, MSc, professor of neurology at the University of Cincinnati Academic Health Center, was cautious. “These findings are of unclear applicability to any particular patient without a biomarker for a deficit of glycolysis that these drugs are presumed to affect,” Dr. Espay said. “Hence, there is no feasible or warranted change in practice as a result of this study.”
Pathogenic mechanisms are heterogeneous among patients with Parkinson’s disease, Dr. Espay added. “We will need to understand who among the large biological universe of Parkinson’s patients may have impaired energy metabolism as a pathogenic mechanism to be selected for a future clinical trial evaluating terazosin, doxazosin, or alfuzosin as a potential disease-modifying intervention.”
Parkinson’s disease is not one disease, but a group of disorders with unique biological abnormalities, said Dr. Espay. “We know so much about ‘Parkinson’s disease’ and next to nothing about the biology of individuals with Parkinson’s disease.”
This situation has enabled the development of symptomatic treatments, such as dopaminergic therapies, but failed to yield disease-modifying treatments, he said.
The University of Iowa contributed funds for this study. Dr. Simmering has received pilot funding from the University of Iowa Institute for Clinical and Translational Science. He had no conflicts of interest to disclose. Dr. Espay disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests. Treatment of BPH with terazosin (Hytrin), doxazosin (Cardura), or alfuzosin (Uroxatral), all of which enhance glycolysis, was associated with a lower risk of developing Parkinson’s disease than patients taking a drug used for the same indication, tamsulosin (Flomax), which does not affect glycolysis.
“If giving someone terazosin or similar medications truly reduces their risk of disease, these results could have significant clinical implications for neurologists,” said lead author Jacob E. Simmering, PhD, assistant professor of internal medicine at the University of Iowa, Iowa City.
There are few reliable neuroprotective treatments for Parkinson’s disease, he said. “We can manage some of the symptoms, but we can’t stop it from progressing. If a randomized trial finds the same result, this will provide a new option to slow progression of Parkinson’s disease.”
The pathogenesis of Parkinson’s disease is heterogeneous, however, and not all patients may benefit from glycolysis-enhancing drugs, the investigators noted. Future research will be needed to identify potential candidates for this treatment, and clarify the effects of these drugs, they wrote.
The findings were published online Feb. 1, 2021, in JAMA Neurology.
Time-dependent effects
The major risk factor for Parkinson’s disease is age, which is associated with impaired energy metabolism. Glycolysis is decreased among patients with Parkinson’s disease, yet impaired energy metabolism has not been investigated widely as a pathogenic factor in the disease, the authors wrote.
Studies have indicated that terazosin increases the activity of an enzyme important in glycolysis. Doxazosin and alfuzosin have a similar mechanism of action and enhance energy metabolism. Tamsulosin, a structurally unrelated drug, has the same mechanism of action as the other three drugs, but does not enhance energy metabolism.
In this report, the researchers investigated the hypothesis that patients who received therapy with terazosin, doxazosin, or alfuzosin would have a lower risk of developing Parkinson’s disease than patients receiving tamsulosin. To do that, they used health care utilization data from Denmark and the United States, including the Danish National Prescription Registry, the Danish National Patient Registry, the Danish Civil Registration System, and the Truven Health Analytics MarketScan database.
The investigators searched the records for patients who filled prescriptions for any of the four drugs of interest. They excluded any patients who developed Parkinson’s disease within 1 year of starting medication. Because use of these drugs is rare among women, they included only men in their analysis.
They looked at patient outcomes beginning at 1 year after the initiation of treatment. They also required patients to fill at least two prescriptions before the beginning of follow-up. Patients who switched from tamsulosin to any of the other drugs, or vice versa, were excluded from analysis.
The investigators used propensity-score matching to ensure that patients in the tamsulosin and terazosin/doxazosin/alfuzosin groups were similar in terms of their other potential risk factors. The primary outcome was the development of Parkinson’s disease.
They identified 52,365 propensity score–matched pairs in the Danish registries and 94,883 pairs in the Truven database. The mean age was 67.9 years in the Danish registries and 63.8 years in the Truven database, and follow-up was approximately 5 years and 3 years respectively. Baseline covariates were well balanced between cohorts.
Among Danish patients, those who took terazosin, doxazosin, or alfuzosin had a lower risk of developing Parkinson’s disease versus those who took tamsulosin (hazard ratio, 0.88). Similarly, patients in the Truven database who took terazosin, doxazosin, or alfuzosin had a lower risk of developing Parkinson’s disease than those who took tamsulosin (HR, 0.63).
In both cohorts, the risk for Parkinson’s disease among patients receiving terazosin, doxazosin, or alfuzosin, compared with those receiving tamsulosin, decreased with increasing numbers of prescriptions filled. Long-term treatment with any of the three glycolysis-enhancing drugs was associated with greater risk reduction in the Danish (HR, 0.79) and Truven (HR, 0.46) cohorts versus tamsulosin.
Differences in case definitions, which may reflect how Parkinson’s disease was managed, complicate comparisons between the Danish and Truven cohorts, said Dr. Simmering. Another challenge is the source of the data. “The Truven data set was derived from insurance claims from people with private insurance or Medicare supplemental plans,” he said. “This group is quite large but may not be representative of everyone in the United States. We would also only be able to follow people while they were on one insurance plan. If they switched coverage to a company that doesn’t contribute data, we would lose them.”
The Danish database, however, includes all residents of Denmark. Only people who left the country were lost to follow-up.
The results support the hypothesis that increasing energy in cells slows disease progression, Dr. Simmering added. “There are a few conditions, mostly REM sleep disorders, that are associated with future diagnosis of Parkinson’s disease. Right now, we don’t have anything to offer people at elevated risk of Parkinson’s disease that might prevent the disease. If a controlled trial finds that terazosin slows or prevents Parkinson’s disease, we would have something truly protective to offer these patients.”
Biomarker needed
Commenting on the results, Alberto J. Espay, MD, MSc, professor of neurology at the University of Cincinnati Academic Health Center, was cautious. “These findings are of unclear applicability to any particular patient without a biomarker for a deficit of glycolysis that these drugs are presumed to affect,” Dr. Espay said. “Hence, there is no feasible or warranted change in practice as a result of this study.”
Pathogenic mechanisms are heterogeneous among patients with Parkinson’s disease, Dr. Espay added. “We will need to understand who among the large biological universe of Parkinson’s patients may have impaired energy metabolism as a pathogenic mechanism to be selected for a future clinical trial evaluating terazosin, doxazosin, or alfuzosin as a potential disease-modifying intervention.”
Parkinson’s disease is not one disease, but a group of disorders with unique biological abnormalities, said Dr. Espay. “We know so much about ‘Parkinson’s disease’ and next to nothing about the biology of individuals with Parkinson’s disease.”
This situation has enabled the development of symptomatic treatments, such as dopaminergic therapies, but failed to yield disease-modifying treatments, he said.
The University of Iowa contributed funds for this study. Dr. Simmering has received pilot funding from the University of Iowa Institute for Clinical and Translational Science. He had no conflicts of interest to disclose. Dr. Espay disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NEUROLOGY
Researchers examine factors associated with opioid use among migraineurs
Among patients with migraine who use prescription medications, the increasing use of prescription opioids is associated with chronic migraine, more severe disability, and anxiety and depression, according to an analysis published in the January issue of Headache . The use of prescription opioids also is associated with treatment-related variables such as poor acute treatment optimization and treatment in a pain clinic. The results indicate the continued need to educate patients and clinicians about the potential risks of opioids for migraineurs, according to the researchers.
In the Migraine in America Symptoms and Treatment (MAST) study, which the researchers analyzed for their investigation, one-third of migraineurs who use acute prescriptions reported using opioids. Among opioid users, 42% took opioids on 4 or more days per month. “These findings are like [those of] a previous report from the American Migraine Prevalence and Prevention study and more recent findings from the Observational Survey of the Epidemiology, Treatment, and Care of Migraine (OVERCOME) study,” said Richard Lipton, MD, Edwin S. Lowe professor and vice chair of neurology at Albert Einstein College of Medicine in the Bronx, New York. “High rates of opioid use are problematic because opioid use is associated with worsening of migraine over time.”
Opioids remain in widespread use for migraine, even though guidelines recommend against this treatment. Among migraineurs, opioid use is associated with more severe headache-related disability and greater use of health care resources. Opioid use also increases the risk of progressing from episodic migraine to chronic migraine.
A review of MAST data
Dr. Lipton and colleagues set out to identify the variables associated with the frequency of opioid use in people with migraine. Among the variables that they sought to examine were demographic characteristics, comorbidities, headache characteristics, medication use, and patterns of health care use. Dr. Lipton’s group hypothesized that migraine-related severity and burden would increase with increasing frequency of opioid use.
To conduct their research, the investigators examined data from the MAST study, a nationwide sample of American adults with migraine. They focused specifically on participants who reported receiving prescription acute medications. Participants eligible for this analysis reported 3 or more headache days in the previous 3 months and at least 1 monthly headache day in the previous month. In all, 15,133 participants met these criteria.
Dr. Lipton and colleagues categorized participants into four groups based on their frequency of opioid use. The groups had no opioid use, 3 or fewer monthly days of opioid use, 4 to 9 monthly days of opioid use, and 10 or more days of monthly opioid use. The last category is consistent with the International Classification of Headache Disorders-3 criteria for overuse of opioids in migraine.
At baseline, MAST participants provided information about variables such as gender, age, marital status, smoking status, education, and income. Participants also reported how many times in the previous 6 months they had visited a primary care doctor, a neurologist, a headache specialist, or a pain specialist. Dr. Lipton’s group calculated monthly headache days using the number of days during the previous 3 months affected by headache. The Migraine Disability Assessment (MIDAS) questionnaire was used to measure headache-related disability. The four-item Patient Health Questionnaire (PHQ-4) was used to screen for anxiety and depression, and the Migraine Treatment Optimization Questionnaire (mTOQ-4) evaluated participants’ treatment optimization.
Men predominated among opioid users
The investigators included 4,701 MAST participants in their analysis. The population’s mean age was 45 years, and 71.6% of participants were women. Of the entire sample, 67.5% reported no opioid use, and 32.5% reported opioid use. Of the total study population, 18.7% of patients took opioids 3 or fewer days per month, 6.5% took opioids 4 to 9 days per month, and 7.3% took opioids on 10 or more days per month.
Opioid users did not differ from nonusers on race or marital status. Men were overrepresented among all groups of opioid users, however. In addition, opioid use was more prevalent among participants with fewer than 4 years of college education (34.9%) than among participants with 4 or more years of college (30.8%). The proportion of participants with fewer than 4 years of college increased with increasing monthly opioid use. Furthermore, opioid use increased with decreasing household income. As opioid use increased, rates of employment decreased. Approximately 33% of the entire sample were obese, and the proportion of obese participants increased with increasing days per month of opioid use.
The most frequent setting during the previous 6 months for participants seeking care was primary care (49.7%). The next most frequent setting was neurology units (20.9%), pain clinics (8.3%), and headache clinics (7.7%). The prevalence of opioid use was 37.5% among participants with primary care visits, 37.3% among participants with neurologist visits, 43.0% among participants with headache clinic visits, and 53.5% with pain clinic visits.
About 15% of the population had chronic migraine. The prevalence of chronic migraine increased with increasing frequency of opioid use. About 49% of the sample had allodynia, and the prevalence of allodynia increased with increasing frequency of opioid use. Overall, disability was moderate to severe in 57.3% of participants. Participants who used opioids on 3 or fewer days per month had the lowest prevalence of moderate to severe disability (50.2%), and participants who used opioids on 10 or more days per month had the highest prevalence of moderate to severe disability (83.8%).
Approximately 21% of participants had anxiety or depression. The lowest prevalence of anxiety or depression was among participants who took opioids on 3 or fewer days per month (17.4%), and the highest prevalence was among participants who took opioids on 10 or more days per month (43.2%). About 39% of the population had very poor to poor treatment optimization. Among opioid nonusers, 35.6% had very poor to poor treatment optimization, and 59.4% of participants who used opioids on 10 or more days per month had very poor to poor treatment optimization.
Dr. Lipton and colleagues also examined the study population’s use of triptans. Overall, 51.5% of participants reported taking triptans. The prevalence of triptan use was highest among participants who did not use opioids (64.1%) and lowest among participants who used opioids on 3 or fewer days per month (20.5%). Triptan use increased as monthly days of opioid use increased.
Pain clinics and opioid prescription
“In the general population, women are more likely to receive opioids than men,” said Dr. Lipton. “This [finding] could reflect, in part, that women have more pain disorders than men and are more likely to seek medical care for pain than men.” In the current study, however, men with migraine were more likely to receive opioid prescriptions than were women with migraine. One potential explanation for this finding is that men with migraine are less likely to receive a migraine diagnosis, which might attenuate opioid prescribing, than women with migraine. “It may be that opioids are perceived to be serious drugs for serious pain, and that some physicians may be more likely to prescribe opioids to men because the disorder is taken more seriously in men than women,” said Dr. Lipton.
The observation that opioids were more likely to be prescribed for people treated in pain clinics “is consistent with my understanding of practice patterns,” he added. “Generally, neurologists strive to find effective acute treatment alternatives to opioids. The emergence of [drug classes known as] gepants and ditans provides a helpful set of alternatives to tritpans.”
Dr. Lipton and his colleagues plan further research into the treatment of migraineurs. “In a claims analysis, we showed that when people with migraine fail a triptan, they are most likely to get an opioid as their next drug,” he said. “Reasonable [clinicians] might disagree on the next step. The next step, in the absence of contraindications, could be a different oral triptan, a nonoral triptan, or a gepant or ditan. We are planning a randomized trial to probe this question.”
Why are opioids still being used?
The study’s reliance on patients’ self-report and its retrospective design are two of its weaknesses, said Alan M. Rapoport, MD, clinical professor of neurology at the University of California, Los Angeles, and editor-in-chief of Neurology Reviews. One strength, however, is that the stratified sampling methodology produced a study population that accurately reflects the demographic characteristics of the U.S. adult population, he added. Another strength is the investigators’ examination of opioid use by patient characteristics such as marital status, education, income, obesity, and smoking.
Given the harmful effects of opioids in migraine, it is hard to understand why as much as one-third of study participants using acute care medication for migraine were using opioids, said Dr. Rapoport. Using opioids for the acute treatment of migraine attacks often indicates inadequate treatment optimization, which leads to ongoing headache. As a consequence, patients may take more medication, which can increase headache frequency and lead to diagnoses of chronic migraine and medication overuse headache. Although the study found an association between the increased use of opioids and decreased household income and increased unemployment, smoking, and obesity, “it is not possible to assign causality to any of these associations, even though some would argue that decreased socioeconomic status was somehow related to more headache, disability, obesity, smoking, and unemployment,” he added.
“The paper suggests that future research should look at the risk factors for use of opioids and should determine if depression is a risk factor for or a consequence of opioid use,” said Dr. Rapoport. “Interventional studies designed to improve the acute care of migraine attacks might be able to reduce the use of opioids. I have not used opioids or butalbital-containing medication in my office for many years.”
This study was funded and sponsored by Dr. Reddy’s Laboratories group of companies, Princeton, N.J. Dr. Lipton has received grant support from the National Institutes of Health, the National Headache Foundation, and the Migraine Research Fund. He serves as a consultant, serves as an advisory board member, or has received honoraria from Alder, Allergan, American Headache Society, Autonomic Technologies, Biohaven, Dr. Reddy’s Laboratories, Eli Lilly, eNeura Therapeutics, Merck, Novartis, Pfizer, and Teva, Inc. He receives royalties from Wolff’s Headache, 8th Edition (New York: Oxford University Press, 2009) and holds stock options in eNeura Therapeutics and Biohaven.
SOURCE: Lipton RB, et al. Headache. https://doi.org/10.1111/head.14018. 2020;61(1):103-16.
Among patients with migraine who use prescription medications, the increasing use of prescription opioids is associated with chronic migraine, more severe disability, and anxiety and depression, according to an analysis published in the January issue of Headache . The use of prescription opioids also is associated with treatment-related variables such as poor acute treatment optimization and treatment in a pain clinic. The results indicate the continued need to educate patients and clinicians about the potential risks of opioids for migraineurs, according to the researchers.
In the Migraine in America Symptoms and Treatment (MAST) study, which the researchers analyzed for their investigation, one-third of migraineurs who use acute prescriptions reported using opioids. Among opioid users, 42% took opioids on 4 or more days per month. “These findings are like [those of] a previous report from the American Migraine Prevalence and Prevention study and more recent findings from the Observational Survey of the Epidemiology, Treatment, and Care of Migraine (OVERCOME) study,” said Richard Lipton, MD, Edwin S. Lowe professor and vice chair of neurology at Albert Einstein College of Medicine in the Bronx, New York. “High rates of opioid use are problematic because opioid use is associated with worsening of migraine over time.”
Opioids remain in widespread use for migraine, even though guidelines recommend against this treatment. Among migraineurs, opioid use is associated with more severe headache-related disability and greater use of health care resources. Opioid use also increases the risk of progressing from episodic migraine to chronic migraine.
A review of MAST data
Dr. Lipton and colleagues set out to identify the variables associated with the frequency of opioid use in people with migraine. Among the variables that they sought to examine were demographic characteristics, comorbidities, headache characteristics, medication use, and patterns of health care use. Dr. Lipton’s group hypothesized that migraine-related severity and burden would increase with increasing frequency of opioid use.
To conduct their research, the investigators examined data from the MAST study, a nationwide sample of American adults with migraine. They focused specifically on participants who reported receiving prescription acute medications. Participants eligible for this analysis reported 3 or more headache days in the previous 3 months and at least 1 monthly headache day in the previous month. In all, 15,133 participants met these criteria.
Dr. Lipton and colleagues categorized participants into four groups based on their frequency of opioid use. The groups had no opioid use, 3 or fewer monthly days of opioid use, 4 to 9 monthly days of opioid use, and 10 or more days of monthly opioid use. The last category is consistent with the International Classification of Headache Disorders-3 criteria for overuse of opioids in migraine.
At baseline, MAST participants provided information about variables such as gender, age, marital status, smoking status, education, and income. Participants also reported how many times in the previous 6 months they had visited a primary care doctor, a neurologist, a headache specialist, or a pain specialist. Dr. Lipton’s group calculated monthly headache days using the number of days during the previous 3 months affected by headache. The Migraine Disability Assessment (MIDAS) questionnaire was used to measure headache-related disability. The four-item Patient Health Questionnaire (PHQ-4) was used to screen for anxiety and depression, and the Migraine Treatment Optimization Questionnaire (mTOQ-4) evaluated participants’ treatment optimization.
Men predominated among opioid users
The investigators included 4,701 MAST participants in their analysis. The population’s mean age was 45 years, and 71.6% of participants were women. Of the entire sample, 67.5% reported no opioid use, and 32.5% reported opioid use. Of the total study population, 18.7% of patients took opioids 3 or fewer days per month, 6.5% took opioids 4 to 9 days per month, and 7.3% took opioids on 10 or more days per month.
Opioid users did not differ from nonusers on race or marital status. Men were overrepresented among all groups of opioid users, however. In addition, opioid use was more prevalent among participants with fewer than 4 years of college education (34.9%) than among participants with 4 or more years of college (30.8%). The proportion of participants with fewer than 4 years of college increased with increasing monthly opioid use. Furthermore, opioid use increased with decreasing household income. As opioid use increased, rates of employment decreased. Approximately 33% of the entire sample were obese, and the proportion of obese participants increased with increasing days per month of opioid use.
The most frequent setting during the previous 6 months for participants seeking care was primary care (49.7%). The next most frequent setting was neurology units (20.9%), pain clinics (8.3%), and headache clinics (7.7%). The prevalence of opioid use was 37.5% among participants with primary care visits, 37.3% among participants with neurologist visits, 43.0% among participants with headache clinic visits, and 53.5% with pain clinic visits.
About 15% of the population had chronic migraine. The prevalence of chronic migraine increased with increasing frequency of opioid use. About 49% of the sample had allodynia, and the prevalence of allodynia increased with increasing frequency of opioid use. Overall, disability was moderate to severe in 57.3% of participants. Participants who used opioids on 3 or fewer days per month had the lowest prevalence of moderate to severe disability (50.2%), and participants who used opioids on 10 or more days per month had the highest prevalence of moderate to severe disability (83.8%).
Approximately 21% of participants had anxiety or depression. The lowest prevalence of anxiety or depression was among participants who took opioids on 3 or fewer days per month (17.4%), and the highest prevalence was among participants who took opioids on 10 or more days per month (43.2%). About 39% of the population had very poor to poor treatment optimization. Among opioid nonusers, 35.6% had very poor to poor treatment optimization, and 59.4% of participants who used opioids on 10 or more days per month had very poor to poor treatment optimization.
Dr. Lipton and colleagues also examined the study population’s use of triptans. Overall, 51.5% of participants reported taking triptans. The prevalence of triptan use was highest among participants who did not use opioids (64.1%) and lowest among participants who used opioids on 3 or fewer days per month (20.5%). Triptan use increased as monthly days of opioid use increased.
Pain clinics and opioid prescription
“In the general population, women are more likely to receive opioids than men,” said Dr. Lipton. “This [finding] could reflect, in part, that women have more pain disorders than men and are more likely to seek medical care for pain than men.” In the current study, however, men with migraine were more likely to receive opioid prescriptions than were women with migraine. One potential explanation for this finding is that men with migraine are less likely to receive a migraine diagnosis, which might attenuate opioid prescribing, than women with migraine. “It may be that opioids are perceived to be serious drugs for serious pain, and that some physicians may be more likely to prescribe opioids to men because the disorder is taken more seriously in men than women,” said Dr. Lipton.
The observation that opioids were more likely to be prescribed for people treated in pain clinics “is consistent with my understanding of practice patterns,” he added. “Generally, neurologists strive to find effective acute treatment alternatives to opioids. The emergence of [drug classes known as] gepants and ditans provides a helpful set of alternatives to tritpans.”
Dr. Lipton and his colleagues plan further research into the treatment of migraineurs. “In a claims analysis, we showed that when people with migraine fail a triptan, they are most likely to get an opioid as their next drug,” he said. “Reasonable [clinicians] might disagree on the next step. The next step, in the absence of contraindications, could be a different oral triptan, a nonoral triptan, or a gepant or ditan. We are planning a randomized trial to probe this question.”
Why are opioids still being used?
The study’s reliance on patients’ self-report and its retrospective design are two of its weaknesses, said Alan M. Rapoport, MD, clinical professor of neurology at the University of California, Los Angeles, and editor-in-chief of Neurology Reviews. One strength, however, is that the stratified sampling methodology produced a study population that accurately reflects the demographic characteristics of the U.S. adult population, he added. Another strength is the investigators’ examination of opioid use by patient characteristics such as marital status, education, income, obesity, and smoking.
Given the harmful effects of opioids in migraine, it is hard to understand why as much as one-third of study participants using acute care medication for migraine were using opioids, said Dr. Rapoport. Using opioids for the acute treatment of migraine attacks often indicates inadequate treatment optimization, which leads to ongoing headache. As a consequence, patients may take more medication, which can increase headache frequency and lead to diagnoses of chronic migraine and medication overuse headache. Although the study found an association between the increased use of opioids and decreased household income and increased unemployment, smoking, and obesity, “it is not possible to assign causality to any of these associations, even though some would argue that decreased socioeconomic status was somehow related to more headache, disability, obesity, smoking, and unemployment,” he added.
“The paper suggests that future research should look at the risk factors for use of opioids and should determine if depression is a risk factor for or a consequence of opioid use,” said Dr. Rapoport. “Interventional studies designed to improve the acute care of migraine attacks might be able to reduce the use of opioids. I have not used opioids or butalbital-containing medication in my office for many years.”
This study was funded and sponsored by Dr. Reddy’s Laboratories group of companies, Princeton, N.J. Dr. Lipton has received grant support from the National Institutes of Health, the National Headache Foundation, and the Migraine Research Fund. He serves as a consultant, serves as an advisory board member, or has received honoraria from Alder, Allergan, American Headache Society, Autonomic Technologies, Biohaven, Dr. Reddy’s Laboratories, Eli Lilly, eNeura Therapeutics, Merck, Novartis, Pfizer, and Teva, Inc. He receives royalties from Wolff’s Headache, 8th Edition (New York: Oxford University Press, 2009) and holds stock options in eNeura Therapeutics and Biohaven.
SOURCE: Lipton RB, et al. Headache. https://doi.org/10.1111/head.14018. 2020;61(1):103-16.
Among patients with migraine who use prescription medications, the increasing use of prescription opioids is associated with chronic migraine, more severe disability, and anxiety and depression, according to an analysis published in the January issue of Headache . The use of prescription opioids also is associated with treatment-related variables such as poor acute treatment optimization and treatment in a pain clinic. The results indicate the continued need to educate patients and clinicians about the potential risks of opioids for migraineurs, according to the researchers.
In the Migraine in America Symptoms and Treatment (MAST) study, which the researchers analyzed for their investigation, one-third of migraineurs who use acute prescriptions reported using opioids. Among opioid users, 42% took opioids on 4 or more days per month. “These findings are like [those of] a previous report from the American Migraine Prevalence and Prevention study and more recent findings from the Observational Survey of the Epidemiology, Treatment, and Care of Migraine (OVERCOME) study,” said Richard Lipton, MD, Edwin S. Lowe professor and vice chair of neurology at Albert Einstein College of Medicine in the Bronx, New York. “High rates of opioid use are problematic because opioid use is associated with worsening of migraine over time.”
Opioids remain in widespread use for migraine, even though guidelines recommend against this treatment. Among migraineurs, opioid use is associated with more severe headache-related disability and greater use of health care resources. Opioid use also increases the risk of progressing from episodic migraine to chronic migraine.
A review of MAST data
Dr. Lipton and colleagues set out to identify the variables associated with the frequency of opioid use in people with migraine. Among the variables that they sought to examine were demographic characteristics, comorbidities, headache characteristics, medication use, and patterns of health care use. Dr. Lipton’s group hypothesized that migraine-related severity and burden would increase with increasing frequency of opioid use.
To conduct their research, the investigators examined data from the MAST study, a nationwide sample of American adults with migraine. They focused specifically on participants who reported receiving prescription acute medications. Participants eligible for this analysis reported 3 or more headache days in the previous 3 months and at least 1 monthly headache day in the previous month. In all, 15,133 participants met these criteria.
Dr. Lipton and colleagues categorized participants into four groups based on their frequency of opioid use. The groups had no opioid use, 3 or fewer monthly days of opioid use, 4 to 9 monthly days of opioid use, and 10 or more days of monthly opioid use. The last category is consistent with the International Classification of Headache Disorders-3 criteria for overuse of opioids in migraine.
At baseline, MAST participants provided information about variables such as gender, age, marital status, smoking status, education, and income. Participants also reported how many times in the previous 6 months they had visited a primary care doctor, a neurologist, a headache specialist, or a pain specialist. Dr. Lipton’s group calculated monthly headache days using the number of days during the previous 3 months affected by headache. The Migraine Disability Assessment (MIDAS) questionnaire was used to measure headache-related disability. The four-item Patient Health Questionnaire (PHQ-4) was used to screen for anxiety and depression, and the Migraine Treatment Optimization Questionnaire (mTOQ-4) evaluated participants’ treatment optimization.
Men predominated among opioid users
The investigators included 4,701 MAST participants in their analysis. The population’s mean age was 45 years, and 71.6% of participants were women. Of the entire sample, 67.5% reported no opioid use, and 32.5% reported opioid use. Of the total study population, 18.7% of patients took opioids 3 or fewer days per month, 6.5% took opioids 4 to 9 days per month, and 7.3% took opioids on 10 or more days per month.
Opioid users did not differ from nonusers on race or marital status. Men were overrepresented among all groups of opioid users, however. In addition, opioid use was more prevalent among participants with fewer than 4 years of college education (34.9%) than among participants with 4 or more years of college (30.8%). The proportion of participants with fewer than 4 years of college increased with increasing monthly opioid use. Furthermore, opioid use increased with decreasing household income. As opioid use increased, rates of employment decreased. Approximately 33% of the entire sample were obese, and the proportion of obese participants increased with increasing days per month of opioid use.
The most frequent setting during the previous 6 months for participants seeking care was primary care (49.7%). The next most frequent setting was neurology units (20.9%), pain clinics (8.3%), and headache clinics (7.7%). The prevalence of opioid use was 37.5% among participants with primary care visits, 37.3% among participants with neurologist visits, 43.0% among participants with headache clinic visits, and 53.5% with pain clinic visits.
About 15% of the population had chronic migraine. The prevalence of chronic migraine increased with increasing frequency of opioid use. About 49% of the sample had allodynia, and the prevalence of allodynia increased with increasing frequency of opioid use. Overall, disability was moderate to severe in 57.3% of participants. Participants who used opioids on 3 or fewer days per month had the lowest prevalence of moderate to severe disability (50.2%), and participants who used opioids on 10 or more days per month had the highest prevalence of moderate to severe disability (83.8%).
Approximately 21% of participants had anxiety or depression. The lowest prevalence of anxiety or depression was among participants who took opioids on 3 or fewer days per month (17.4%), and the highest prevalence was among participants who took opioids on 10 or more days per month (43.2%). About 39% of the population had very poor to poor treatment optimization. Among opioid nonusers, 35.6% had very poor to poor treatment optimization, and 59.4% of participants who used opioids on 10 or more days per month had very poor to poor treatment optimization.
Dr. Lipton and colleagues also examined the study population’s use of triptans. Overall, 51.5% of participants reported taking triptans. The prevalence of triptan use was highest among participants who did not use opioids (64.1%) and lowest among participants who used opioids on 3 or fewer days per month (20.5%). Triptan use increased as monthly days of opioid use increased.
Pain clinics and opioid prescription
“In the general population, women are more likely to receive opioids than men,” said Dr. Lipton. “This [finding] could reflect, in part, that women have more pain disorders than men and are more likely to seek medical care for pain than men.” In the current study, however, men with migraine were more likely to receive opioid prescriptions than were women with migraine. One potential explanation for this finding is that men with migraine are less likely to receive a migraine diagnosis, which might attenuate opioid prescribing, than women with migraine. “It may be that opioids are perceived to be serious drugs for serious pain, and that some physicians may be more likely to prescribe opioids to men because the disorder is taken more seriously in men than women,” said Dr. Lipton.
The observation that opioids were more likely to be prescribed for people treated in pain clinics “is consistent with my understanding of practice patterns,” he added. “Generally, neurologists strive to find effective acute treatment alternatives to opioids. The emergence of [drug classes known as] gepants and ditans provides a helpful set of alternatives to tritpans.”
Dr. Lipton and his colleagues plan further research into the treatment of migraineurs. “In a claims analysis, we showed that when people with migraine fail a triptan, they are most likely to get an opioid as their next drug,” he said. “Reasonable [clinicians] might disagree on the next step. The next step, in the absence of contraindications, could be a different oral triptan, a nonoral triptan, or a gepant or ditan. We are planning a randomized trial to probe this question.”
Why are opioids still being used?
The study’s reliance on patients’ self-report and its retrospective design are two of its weaknesses, said Alan M. Rapoport, MD, clinical professor of neurology at the University of California, Los Angeles, and editor-in-chief of Neurology Reviews. One strength, however, is that the stratified sampling methodology produced a study population that accurately reflects the demographic characteristics of the U.S. adult population, he added. Another strength is the investigators’ examination of opioid use by patient characteristics such as marital status, education, income, obesity, and smoking.
Given the harmful effects of opioids in migraine, it is hard to understand why as much as one-third of study participants using acute care medication for migraine were using opioids, said Dr. Rapoport. Using opioids for the acute treatment of migraine attacks often indicates inadequate treatment optimization, which leads to ongoing headache. As a consequence, patients may take more medication, which can increase headache frequency and lead to diagnoses of chronic migraine and medication overuse headache. Although the study found an association between the increased use of opioids and decreased household income and increased unemployment, smoking, and obesity, “it is not possible to assign causality to any of these associations, even though some would argue that decreased socioeconomic status was somehow related to more headache, disability, obesity, smoking, and unemployment,” he added.
“The paper suggests that future research should look at the risk factors for use of opioids and should determine if depression is a risk factor for or a consequence of opioid use,” said Dr. Rapoport. “Interventional studies designed to improve the acute care of migraine attacks might be able to reduce the use of opioids. I have not used opioids or butalbital-containing medication in my office for many years.”
This study was funded and sponsored by Dr. Reddy’s Laboratories group of companies, Princeton, N.J. Dr. Lipton has received grant support from the National Institutes of Health, the National Headache Foundation, and the Migraine Research Fund. He serves as a consultant, serves as an advisory board member, or has received honoraria from Alder, Allergan, American Headache Society, Autonomic Technologies, Biohaven, Dr. Reddy’s Laboratories, Eli Lilly, eNeura Therapeutics, Merck, Novartis, Pfizer, and Teva, Inc. He receives royalties from Wolff’s Headache, 8th Edition (New York: Oxford University Press, 2009) and holds stock options in eNeura Therapeutics and Biohaven.
SOURCE: Lipton RB, et al. Headache. https://doi.org/10.1111/head.14018. 2020;61(1):103-16.
FROM HEADACHE












