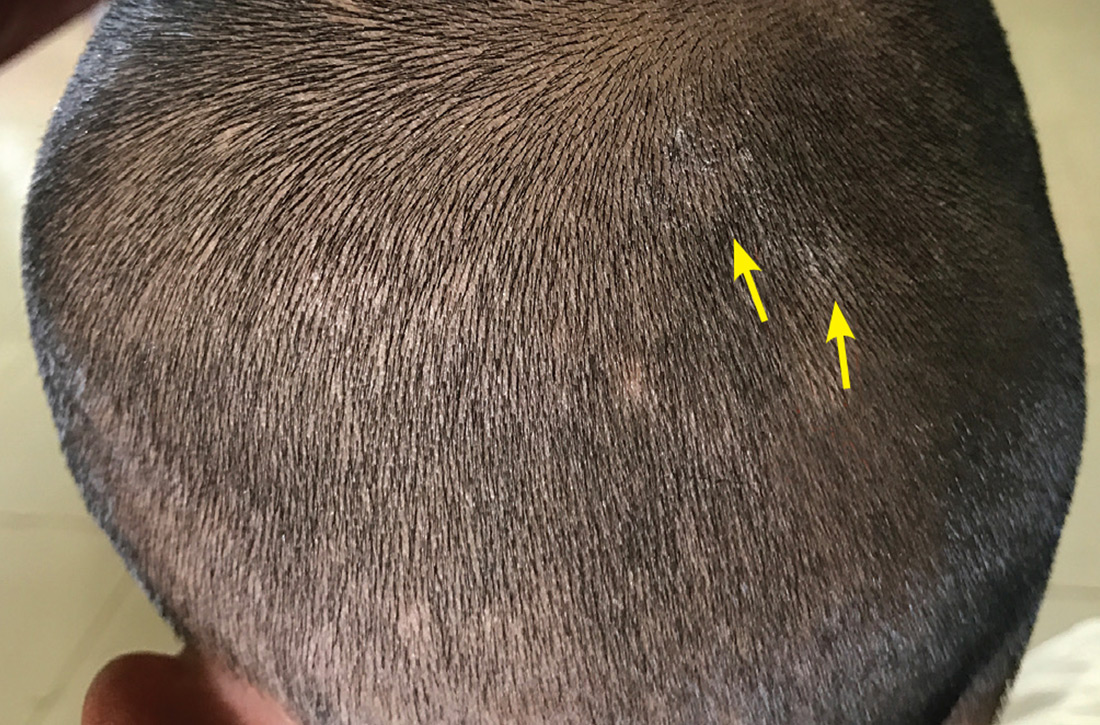
User login
COVID-19 may damage blood vessels in the brain
Until now, the neurological manifestations of COVID-19 have been believed to be a result of direct damage to nerve cells. However, a new study suggests that the virus might actually damage the brain’s small blood vessels rather than nerve cells themselves.
The findings add further weight to previous research into neurological complications from COVID-19, according to Anna Cervantes, MD. Dr. Cervantes is assistant professor of neurology at the Boston University and has been studying the neurological effects of COVID-19, though she was not involved in this study. “I can tell from my personal experience, and things we’ve published on and the literature that’s out there – there are patients that are having complications like stroke that aren’t even critically ill from COVID. We’re seeing that not in just the acute setting, but also in a delayed fashion. Even though most of the coagulopathy is largely venous and probably microvascular, this does affect the brain through a myriad of ways,” Dr. Cervantes said.
The research was published online Jan. 12 in the New England Journal of Medicine. Myoung‑Hwa Lee, PhD, was the lead author.
The study included high resolution magnetic resonance imaging and histopathological examination of 13 individuals with a median age of 50 years. Among 10 patients with brain alterations, the researchers conducted further studies in 5 individuals using multiplex fluorescence imaging and chromogenic immunostaining in all 10.
The team conducted conventional histopathology on the brains of 18 individuals. Fourteen had a history of chronic illness, including diabetes, and hypertension, and 11 had died unexpectedly or been found dead. Magnetic resonance microscopy revealed punctuate hypo-intensities in nine subjects, indicating microvascular injury and fibrinogen leakage. Histopathology using fluorescence imaging showed the same features. Collagen IV immunostaining showed thinning of the basal lamina of the endothelial cells in five patients. Ten patients had congested blood vessels and surrounding fibrinogen leakage, but comparatively intact vasculature. The researchers interpreted linear hypo-intensities as micro-hemorrhages.
The researchers found little perivascular inflammation, and no vascular occlusion. Thirteen subjects had perivascular-activated microglia, macrophage infiltrates, and hypertrophic astrocytes. Eight had CD3+ and CD8+ T cells in the perivascular spaces and in lumens next to endothelial cells, which could help explain vascular injury.
The researchers found no evidence of the SARS-CoV-2 virus itself, despite efforts using polymerase chain reaction with multiple primer sets, RNA sequencing within the brain, or RNA in situ hybridization and immunostaining. Subjects may have cleared the virus by the time they died, or viral copy numbers could have been below the detection limit of the assays.
The researchers also obtained a convenience sample of subjects who had died from COVID-19. Magnetic resonance microscopy, histopathology, and immunohistochemical analysis of sections revealed microvascular injury in the brain and olfactory bulb, despite no evidence of viral infection. The authors stressed that they could not draw conclusions about the neurological features of COVID-19 because of a lack of clinical information.
Dr. Cervantes noted that limitation: “We’re seeing a lot of patients with encephalopathy or alterations in their mental status. A lot of things can cause that, and some are common in patients who are critically ill, like medications and metabolic derangement.”
Still, the findings could help to inform future medical management. “There’s going to be a large number of patients who don’t have really bad pulmonary disease but still may have encephalopathy. So if there is small vessel involvement because of inflammation that we might not necessarily catch in a lumbar puncture or routine imaging, there’s still somebody we can make better (using) steroids. Having more information on what’s happening on a pathophysiologic level and on pathology is really helpful.”
The study was supported by internal funds from the National Institute of Neurological Disorders and Stroke. Dr. Cervantes has no relevant financial disclosures.
Until now, the neurological manifestations of COVID-19 have been believed to be a result of direct damage to nerve cells. However, a new study suggests that the virus might actually damage the brain’s small blood vessels rather than nerve cells themselves.
The findings add further weight to previous research into neurological complications from COVID-19, according to Anna Cervantes, MD. Dr. Cervantes is assistant professor of neurology at the Boston University and has been studying the neurological effects of COVID-19, though she was not involved in this study. “I can tell from my personal experience, and things we’ve published on and the literature that’s out there – there are patients that are having complications like stroke that aren’t even critically ill from COVID. We’re seeing that not in just the acute setting, but also in a delayed fashion. Even though most of the coagulopathy is largely venous and probably microvascular, this does affect the brain through a myriad of ways,” Dr. Cervantes said.
The research was published online Jan. 12 in the New England Journal of Medicine. Myoung‑Hwa Lee, PhD, was the lead author.
The study included high resolution magnetic resonance imaging and histopathological examination of 13 individuals with a median age of 50 years. Among 10 patients with brain alterations, the researchers conducted further studies in 5 individuals using multiplex fluorescence imaging and chromogenic immunostaining in all 10.
The team conducted conventional histopathology on the brains of 18 individuals. Fourteen had a history of chronic illness, including diabetes, and hypertension, and 11 had died unexpectedly or been found dead. Magnetic resonance microscopy revealed punctuate hypo-intensities in nine subjects, indicating microvascular injury and fibrinogen leakage. Histopathology using fluorescence imaging showed the same features. Collagen IV immunostaining showed thinning of the basal lamina of the endothelial cells in five patients. Ten patients had congested blood vessels and surrounding fibrinogen leakage, but comparatively intact vasculature. The researchers interpreted linear hypo-intensities as micro-hemorrhages.
The researchers found little perivascular inflammation, and no vascular occlusion. Thirteen subjects had perivascular-activated microglia, macrophage infiltrates, and hypertrophic astrocytes. Eight had CD3+ and CD8+ T cells in the perivascular spaces and in lumens next to endothelial cells, which could help explain vascular injury.
The researchers found no evidence of the SARS-CoV-2 virus itself, despite efforts using polymerase chain reaction with multiple primer sets, RNA sequencing within the brain, or RNA in situ hybridization and immunostaining. Subjects may have cleared the virus by the time they died, or viral copy numbers could have been below the detection limit of the assays.
The researchers also obtained a convenience sample of subjects who had died from COVID-19. Magnetic resonance microscopy, histopathology, and immunohistochemical analysis of sections revealed microvascular injury in the brain and olfactory bulb, despite no evidence of viral infection. The authors stressed that they could not draw conclusions about the neurological features of COVID-19 because of a lack of clinical information.
Dr. Cervantes noted that limitation: “We’re seeing a lot of patients with encephalopathy or alterations in their mental status. A lot of things can cause that, and some are common in patients who are critically ill, like medications and metabolic derangement.”
Still, the findings could help to inform future medical management. “There’s going to be a large number of patients who don’t have really bad pulmonary disease but still may have encephalopathy. So if there is small vessel involvement because of inflammation that we might not necessarily catch in a lumbar puncture or routine imaging, there’s still somebody we can make better (using) steroids. Having more information on what’s happening on a pathophysiologic level and on pathology is really helpful.”
The study was supported by internal funds from the National Institute of Neurological Disorders and Stroke. Dr. Cervantes has no relevant financial disclosures.
Until now, the neurological manifestations of COVID-19 have been believed to be a result of direct damage to nerve cells. However, a new study suggests that the virus might actually damage the brain’s small blood vessels rather than nerve cells themselves.
The findings add further weight to previous research into neurological complications from COVID-19, according to Anna Cervantes, MD. Dr. Cervantes is assistant professor of neurology at the Boston University and has been studying the neurological effects of COVID-19, though she was not involved in this study. “I can tell from my personal experience, and things we’ve published on and the literature that’s out there – there are patients that are having complications like stroke that aren’t even critically ill from COVID. We’re seeing that not in just the acute setting, but also in a delayed fashion. Even though most of the coagulopathy is largely venous and probably microvascular, this does affect the brain through a myriad of ways,” Dr. Cervantes said.
The research was published online Jan. 12 in the New England Journal of Medicine. Myoung‑Hwa Lee, PhD, was the lead author.
The study included high resolution magnetic resonance imaging and histopathological examination of 13 individuals with a median age of 50 years. Among 10 patients with brain alterations, the researchers conducted further studies in 5 individuals using multiplex fluorescence imaging and chromogenic immunostaining in all 10.
The team conducted conventional histopathology on the brains of 18 individuals. Fourteen had a history of chronic illness, including diabetes, and hypertension, and 11 had died unexpectedly or been found dead. Magnetic resonance microscopy revealed punctuate hypo-intensities in nine subjects, indicating microvascular injury and fibrinogen leakage. Histopathology using fluorescence imaging showed the same features. Collagen IV immunostaining showed thinning of the basal lamina of the endothelial cells in five patients. Ten patients had congested blood vessels and surrounding fibrinogen leakage, but comparatively intact vasculature. The researchers interpreted linear hypo-intensities as micro-hemorrhages.
The researchers found little perivascular inflammation, and no vascular occlusion. Thirteen subjects had perivascular-activated microglia, macrophage infiltrates, and hypertrophic astrocytes. Eight had CD3+ and CD8+ T cells in the perivascular spaces and in lumens next to endothelial cells, which could help explain vascular injury.
The researchers found no evidence of the SARS-CoV-2 virus itself, despite efforts using polymerase chain reaction with multiple primer sets, RNA sequencing within the brain, or RNA in situ hybridization and immunostaining. Subjects may have cleared the virus by the time they died, or viral copy numbers could have been below the detection limit of the assays.
The researchers also obtained a convenience sample of subjects who had died from COVID-19. Magnetic resonance microscopy, histopathology, and immunohistochemical analysis of sections revealed microvascular injury in the brain and olfactory bulb, despite no evidence of viral infection. The authors stressed that they could not draw conclusions about the neurological features of COVID-19 because of a lack of clinical information.
Dr. Cervantes noted that limitation: “We’re seeing a lot of patients with encephalopathy or alterations in their mental status. A lot of things can cause that, and some are common in patients who are critically ill, like medications and metabolic derangement.”
Still, the findings could help to inform future medical management. “There’s going to be a large number of patients who don’t have really bad pulmonary disease but still may have encephalopathy. So if there is small vessel involvement because of inflammation that we might not necessarily catch in a lumbar puncture or routine imaging, there’s still somebody we can make better (using) steroids. Having more information on what’s happening on a pathophysiologic level and on pathology is really helpful.”
The study was supported by internal funds from the National Institute of Neurological Disorders and Stroke. Dr. Cervantes has no relevant financial disclosures.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
HHS will drop buprenorphine waiver rule for most physicians
Federal officials on Thursday announced a plan to largely drop the so-called X-waiver requirement for buprenorphine prescriptions for physicians in a bid to remove an administrative procedure widely seen as a barrier to opioid use disorder (OUD) treatment.
The Department of Health & Human Services unveiled new practice guidelines that include an exemption from current certification requirements. The exemption applies to physicians already registered with the Drug Enforcement Administration.
A restriction included in the new HHS policy is a limit of treating no more than 30 patients with buprenorphine for OUD at any one time. There is an exception to this limit for hospital-based physicians, such as those working in emergency departments, HHS said.
, such as buprenorphine, and does not apply to methadone. The new guidelines say the date on which they will take effect will be added after publication in the Federal Register. HHS did not immediately answer a request from this news organization for a more specific timeline.
Welcomed change
The change in prescribing rule was widely welcomed, with the American Medical Association issuing a statement endorsing the revision. The AMA and many prescribers and researchers had seen the X-waiver as a hurdle to address the nation’s opioid epidemic.
There were more than 83,000 deaths attributed to drug overdoses in the United States in the 12 months ending in June 2020. This is the highest number of overdose deaths ever recorded in a 12-month period, HHS said in a press release, which cited data from the Centers for Disease Control and Prevention.
In a tweet about the new policy, Peter Grinspoon, MD, a Boston internist and author of the memoir “Free Refills: A Doctor Confronts His Addiction,” contrasted the relative ease with which clinicians can give medicines that carry a risk for abuse with the challenge that has existed in trying to provide patients with buprenorphine.
“Absolutely insane that we need a special waiver for buprenorphine to TREAT opioid addiction, but not to prescribe oxycodone, Vicodin, etc., which can get people in trouble in the first place!!” Dr. Grinspoon tweeted.
Patrice Harris, MD, chair of the AMA’s Opioid Task Force and the organization’s immediate past president, said removing the X-waiver requirement can help lessen the stigma associated with this OUD treatment. The AMA had urged HHS to change the regulation.
“With this change, office-based physicians and physician-led teams working with patients to manage their other medical conditions can also treat them for their opioid use disorder without being subjected to a separate and burdensome regulatory regime,” Dr. Harris said in the AMA statement.
Researchers have in recent years sought to highlight what they described as missed opportunities for OUD treatment because of the need for the X-waiver.
Buprenorphine is a cost-effective treatment for opioid use disorder, which reduces the risk of injection-related infections and mortality risk, notes a study published online last month in JAMA Network Open.
However, results showed that fewer than 2% of obstetrician-gynecologists who examined women enrolled in Medicaid were trained to prescribe buprenorphine. The study, which was based on data from 31, 211 ob.gyns. who accepted Medicaid insurance, was created to quantify how many were on the list of Drug Addiction Treatment Act buprenorphine-waived clinicians.
The Drug Addiction Treatment Act has required 8 hours of training for physicians and 24 hours for nurse practitioners and physician assistants for the X-waiver needed to prescribe buprenorphine, the investigators report.
‘X the X-waiver’
Only 10% of recent family residency graduates reported being adequately trained to prescribe buprenorphine and only 7% reported actually prescribing the drug, write Kevin Fiscella, MD, University of Rochester (N.Y.) Medical Center and colleagues in a 2018 Viewpoint article published in JAMA Psychiatry.
In the article, which was subtitled “X the X Waiver,” they called for deregulation of buprenorphine as a way of mainstreaming treatment for OUD.
“The DATA 2000 has failed – too few physicians have obtained X-waivers,” the authors write. “Regulations reinforce the stigma surrounding buprenorphine prescribers and patients who receive it while constraining access and discouraging patient engagement and retention in treatment.”
The change, announced Jan. 14, leaves in place restrictions on prescribing for clinicians other than physicians. On a call with reporters, Adm. Brett P. Giroir, MD, assistant secretary for health, suggested that federal officials should take further steps to remove hurdles to buprenorphine prescriptions.
“Many people will say this has gone too far,” Dr. Giroir said of the drive to end the X-waiver for clinicians. “But I believe more people will say this has not gone far enough.”
A version of this article first appeared on Medscape.com.
Federal officials on Thursday announced a plan to largely drop the so-called X-waiver requirement for buprenorphine prescriptions for physicians in a bid to remove an administrative procedure widely seen as a barrier to opioid use disorder (OUD) treatment.
The Department of Health & Human Services unveiled new practice guidelines that include an exemption from current certification requirements. The exemption applies to physicians already registered with the Drug Enforcement Administration.
A restriction included in the new HHS policy is a limit of treating no more than 30 patients with buprenorphine for OUD at any one time. There is an exception to this limit for hospital-based physicians, such as those working in emergency departments, HHS said.
, such as buprenorphine, and does not apply to methadone. The new guidelines say the date on which they will take effect will be added after publication in the Federal Register. HHS did not immediately answer a request from this news organization for a more specific timeline.
Welcomed change
The change in prescribing rule was widely welcomed, with the American Medical Association issuing a statement endorsing the revision. The AMA and many prescribers and researchers had seen the X-waiver as a hurdle to address the nation’s opioid epidemic.
There were more than 83,000 deaths attributed to drug overdoses in the United States in the 12 months ending in June 2020. This is the highest number of overdose deaths ever recorded in a 12-month period, HHS said in a press release, which cited data from the Centers for Disease Control and Prevention.
In a tweet about the new policy, Peter Grinspoon, MD, a Boston internist and author of the memoir “Free Refills: A Doctor Confronts His Addiction,” contrasted the relative ease with which clinicians can give medicines that carry a risk for abuse with the challenge that has existed in trying to provide patients with buprenorphine.
“Absolutely insane that we need a special waiver for buprenorphine to TREAT opioid addiction, but not to prescribe oxycodone, Vicodin, etc., which can get people in trouble in the first place!!” Dr. Grinspoon tweeted.
Patrice Harris, MD, chair of the AMA’s Opioid Task Force and the organization’s immediate past president, said removing the X-waiver requirement can help lessen the stigma associated with this OUD treatment. The AMA had urged HHS to change the regulation.
“With this change, office-based physicians and physician-led teams working with patients to manage their other medical conditions can also treat them for their opioid use disorder without being subjected to a separate and burdensome regulatory regime,” Dr. Harris said in the AMA statement.
Researchers have in recent years sought to highlight what they described as missed opportunities for OUD treatment because of the need for the X-waiver.
Buprenorphine is a cost-effective treatment for opioid use disorder, which reduces the risk of injection-related infections and mortality risk, notes a study published online last month in JAMA Network Open.
However, results showed that fewer than 2% of obstetrician-gynecologists who examined women enrolled in Medicaid were trained to prescribe buprenorphine. The study, which was based on data from 31, 211 ob.gyns. who accepted Medicaid insurance, was created to quantify how many were on the list of Drug Addiction Treatment Act buprenorphine-waived clinicians.
The Drug Addiction Treatment Act has required 8 hours of training for physicians and 24 hours for nurse practitioners and physician assistants for the X-waiver needed to prescribe buprenorphine, the investigators report.
‘X the X-waiver’
Only 10% of recent family residency graduates reported being adequately trained to prescribe buprenorphine and only 7% reported actually prescribing the drug, write Kevin Fiscella, MD, University of Rochester (N.Y.) Medical Center and colleagues in a 2018 Viewpoint article published in JAMA Psychiatry.
In the article, which was subtitled “X the X Waiver,” they called for deregulation of buprenorphine as a way of mainstreaming treatment for OUD.
“The DATA 2000 has failed – too few physicians have obtained X-waivers,” the authors write. “Regulations reinforce the stigma surrounding buprenorphine prescribers and patients who receive it while constraining access and discouraging patient engagement and retention in treatment.”
The change, announced Jan. 14, leaves in place restrictions on prescribing for clinicians other than physicians. On a call with reporters, Adm. Brett P. Giroir, MD, assistant secretary for health, suggested that federal officials should take further steps to remove hurdles to buprenorphine prescriptions.
“Many people will say this has gone too far,” Dr. Giroir said of the drive to end the X-waiver for clinicians. “But I believe more people will say this has not gone far enough.”
A version of this article first appeared on Medscape.com.
Federal officials on Thursday announced a plan to largely drop the so-called X-waiver requirement for buprenorphine prescriptions for physicians in a bid to remove an administrative procedure widely seen as a barrier to opioid use disorder (OUD) treatment.
The Department of Health & Human Services unveiled new practice guidelines that include an exemption from current certification requirements. The exemption applies to physicians already registered with the Drug Enforcement Administration.
A restriction included in the new HHS policy is a limit of treating no more than 30 patients with buprenorphine for OUD at any one time. There is an exception to this limit for hospital-based physicians, such as those working in emergency departments, HHS said.
, such as buprenorphine, and does not apply to methadone. The new guidelines say the date on which they will take effect will be added after publication in the Federal Register. HHS did not immediately answer a request from this news organization for a more specific timeline.
Welcomed change
The change in prescribing rule was widely welcomed, with the American Medical Association issuing a statement endorsing the revision. The AMA and many prescribers and researchers had seen the X-waiver as a hurdle to address the nation’s opioid epidemic.
There were more than 83,000 deaths attributed to drug overdoses in the United States in the 12 months ending in June 2020. This is the highest number of overdose deaths ever recorded in a 12-month period, HHS said in a press release, which cited data from the Centers for Disease Control and Prevention.
In a tweet about the new policy, Peter Grinspoon, MD, a Boston internist and author of the memoir “Free Refills: A Doctor Confronts His Addiction,” contrasted the relative ease with which clinicians can give medicines that carry a risk for abuse with the challenge that has existed in trying to provide patients with buprenorphine.
“Absolutely insane that we need a special waiver for buprenorphine to TREAT opioid addiction, but not to prescribe oxycodone, Vicodin, etc., which can get people in trouble in the first place!!” Dr. Grinspoon tweeted.
Patrice Harris, MD, chair of the AMA’s Opioid Task Force and the organization’s immediate past president, said removing the X-waiver requirement can help lessen the stigma associated with this OUD treatment. The AMA had urged HHS to change the regulation.
“With this change, office-based physicians and physician-led teams working with patients to manage their other medical conditions can also treat them for their opioid use disorder without being subjected to a separate and burdensome regulatory regime,” Dr. Harris said in the AMA statement.
Researchers have in recent years sought to highlight what they described as missed opportunities for OUD treatment because of the need for the X-waiver.
Buprenorphine is a cost-effective treatment for opioid use disorder, which reduces the risk of injection-related infections and mortality risk, notes a study published online last month in JAMA Network Open.
However, results showed that fewer than 2% of obstetrician-gynecologists who examined women enrolled in Medicaid were trained to prescribe buprenorphine. The study, which was based on data from 31, 211 ob.gyns. who accepted Medicaid insurance, was created to quantify how many were on the list of Drug Addiction Treatment Act buprenorphine-waived clinicians.
The Drug Addiction Treatment Act has required 8 hours of training for physicians and 24 hours for nurse practitioners and physician assistants for the X-waiver needed to prescribe buprenorphine, the investigators report.
‘X the X-waiver’
Only 10% of recent family residency graduates reported being adequately trained to prescribe buprenorphine and only 7% reported actually prescribing the drug, write Kevin Fiscella, MD, University of Rochester (N.Y.) Medical Center and colleagues in a 2018 Viewpoint article published in JAMA Psychiatry.
In the article, which was subtitled “X the X Waiver,” they called for deregulation of buprenorphine as a way of mainstreaming treatment for OUD.
“The DATA 2000 has failed – too few physicians have obtained X-waivers,” the authors write. “Regulations reinforce the stigma surrounding buprenorphine prescribers and patients who receive it while constraining access and discouraging patient engagement and retention in treatment.”
The change, announced Jan. 14, leaves in place restrictions on prescribing for clinicians other than physicians. On a call with reporters, Adm. Brett P. Giroir, MD, assistant secretary for health, suggested that federal officials should take further steps to remove hurdles to buprenorphine prescriptions.
“Many people will say this has gone too far,” Dr. Giroir said of the drive to end the X-waiver for clinicians. “But I believe more people will say this has not gone far enough.”
A version of this article first appeared on Medscape.com.
Covert stroke after noncardiac surgery linked with cognitive decline
Background: Prior studies have established an increased risk of overt stroke after noncardiac surgery, with significant associated morbidity and mortality. Similarly, covert stroke in the nonsurgical population is well described and has been shown to be associated with cognitive decline.
Study design: Prospective cohort study.
Setting: Academic centers in nine countries.
Synopsis: This study evaluated 1,114 patients older than 65 years who were hospitalized for noncardiac surgery, excluding patients with carotid and neurosurgical procedures. All enrolled participants completed diffusion-weight MRI of the brain within 9 days of surgery. Follow-up rates for clinical outcomes (1,112; greater than 99%) were excellent, and the primary outcome measure, follow-up Montreal Cognitive Assessment (MOCA) at 1 year, was defined in 1,001 (90%) of the study subjects.
Covert stroke was detected in 78 (7%) of the study participants. Those with covert stroke had a higher incidence of cognitive decline at 1 year (adjusted odds ratio, 1.98; 95% confidence interval, 1.22-3.2) with an absolute risk increase of 13%. Patients with covert stroke also had a higher rate of delirium within 3 days of surgery (hazard ratio, 2.24; 95% CI, 1.06-4.73) and a higher rate of overt stroke and transient ischemic attack at 1 year (HR, 4.13; 95% CI, 1.14-14.99).
This study helps to establish the incidence of covert stroke after noncardiac surgery and provides support for covert stroke as a risk factor for cognitive impairment.
Bottom line: Covert stroke following noncardiac surgery is common, affecting 1 in 14 patients in this study, and it is associated with an increased risk of cognitive decline, perioperative delirium, and subsequent overt stroke.
Citation: The NeuroVISION Investigators (Mrkobrada M et al.). Perioperative covert stroke in patients undergoing noncardiac surgery (NeuroVISION): a prospective cohort study. Lancet. 2019;394(10203):1022-9.
Dr. Herrle is a hospitalist at Maine Medical Center in Portland and at Stephens Memorial Hospital in Norway, Maine.
Background: Prior studies have established an increased risk of overt stroke after noncardiac surgery, with significant associated morbidity and mortality. Similarly, covert stroke in the nonsurgical population is well described and has been shown to be associated with cognitive decline.
Study design: Prospective cohort study.
Setting: Academic centers in nine countries.
Synopsis: This study evaluated 1,114 patients older than 65 years who were hospitalized for noncardiac surgery, excluding patients with carotid and neurosurgical procedures. All enrolled participants completed diffusion-weight MRI of the brain within 9 days of surgery. Follow-up rates for clinical outcomes (1,112; greater than 99%) were excellent, and the primary outcome measure, follow-up Montreal Cognitive Assessment (MOCA) at 1 year, was defined in 1,001 (90%) of the study subjects.
Covert stroke was detected in 78 (7%) of the study participants. Those with covert stroke had a higher incidence of cognitive decline at 1 year (adjusted odds ratio, 1.98; 95% confidence interval, 1.22-3.2) with an absolute risk increase of 13%. Patients with covert stroke also had a higher rate of delirium within 3 days of surgery (hazard ratio, 2.24; 95% CI, 1.06-4.73) and a higher rate of overt stroke and transient ischemic attack at 1 year (HR, 4.13; 95% CI, 1.14-14.99).
This study helps to establish the incidence of covert stroke after noncardiac surgery and provides support for covert stroke as a risk factor for cognitive impairment.
Bottom line: Covert stroke following noncardiac surgery is common, affecting 1 in 14 patients in this study, and it is associated with an increased risk of cognitive decline, perioperative delirium, and subsequent overt stroke.
Citation: The NeuroVISION Investigators (Mrkobrada M et al.). Perioperative covert stroke in patients undergoing noncardiac surgery (NeuroVISION): a prospective cohort study. Lancet. 2019;394(10203):1022-9.
Dr. Herrle is a hospitalist at Maine Medical Center in Portland and at Stephens Memorial Hospital in Norway, Maine.
Background: Prior studies have established an increased risk of overt stroke after noncardiac surgery, with significant associated morbidity and mortality. Similarly, covert stroke in the nonsurgical population is well described and has been shown to be associated with cognitive decline.
Study design: Prospective cohort study.
Setting: Academic centers in nine countries.
Synopsis: This study evaluated 1,114 patients older than 65 years who were hospitalized for noncardiac surgery, excluding patients with carotid and neurosurgical procedures. All enrolled participants completed diffusion-weight MRI of the brain within 9 days of surgery. Follow-up rates for clinical outcomes (1,112; greater than 99%) were excellent, and the primary outcome measure, follow-up Montreal Cognitive Assessment (MOCA) at 1 year, was defined in 1,001 (90%) of the study subjects.
Covert stroke was detected in 78 (7%) of the study participants. Those with covert stroke had a higher incidence of cognitive decline at 1 year (adjusted odds ratio, 1.98; 95% confidence interval, 1.22-3.2) with an absolute risk increase of 13%. Patients with covert stroke also had a higher rate of delirium within 3 days of surgery (hazard ratio, 2.24; 95% CI, 1.06-4.73) and a higher rate of overt stroke and transient ischemic attack at 1 year (HR, 4.13; 95% CI, 1.14-14.99).
This study helps to establish the incidence of covert stroke after noncardiac surgery and provides support for covert stroke as a risk factor for cognitive impairment.
Bottom line: Covert stroke following noncardiac surgery is common, affecting 1 in 14 patients in this study, and it is associated with an increased risk of cognitive decline, perioperative delirium, and subsequent overt stroke.
Citation: The NeuroVISION Investigators (Mrkobrada M et al.). Perioperative covert stroke in patients undergoing noncardiac surgery (NeuroVISION): a prospective cohort study. Lancet. 2019;394(10203):1022-9.
Dr. Herrle is a hospitalist at Maine Medical Center in Portland and at Stephens Memorial Hospital in Norway, Maine.
Overcoming the challenges of COVID-19 for Alzheimer’s patients in long-term care, research
An alarming number of additional Alzheimer’s disease (AD) deaths have been reported across various states within the past several months, according to the Alzheimer’s Association. Centers for Disease Control and Prevention data indicate that no less than 31,000 additional people with the neurodegenerative condition had died from the beginning of the pandemic through the end of September 2020. We know that long-term care facilities have been hit hardest, and access to adequate and/or prompt testing has been cited as the most pressing issue during the onset of the pandemic.1
When ADLs become a matter of survival
For individuals affected with Alzheimer’s disease and other types of dementia, performing routine tasks may seem cumbersome and overwhelming. Many of these patients are dependent upon caregivers and family support to facilitate their activities of daily living (ADLs).
Transitioning into the “new normal” set by the pandemic milieu is not an easy task for the average AD individual, because they are now expected to comply with numerous safety instructions (for example, maintaining hygiene, social distancing, etc.). They are also expected to monitor and communicate information about the onset of any suspected symptom to their caregiver or health care clinician.
The additional tasks added to their list of ADLs are particularly distressing given their already compromised short-term memory and overall cognitive decline. Individuals with AD may also be dealing with a host of psychobehavioral challenges, such as the presence of depression, anxiety, and/or agitation amid self-isolation. Enforced social isolation tied to COVID-19 may compound those issues.
Resource diversion and mitigation strategies
Unfortunately, any disruption in services within a long-term care setting may lead to a suboptimal therapeutic environment for patients. The Washington State LTC, for example, reported experiencing a case fatality rate (CFR) exceeding more than a third of its residents; essential staff and health care clinicians were duly affected from exposure to the virus (the risk of transmission increases considerably during transport between facilities). Access to personal protective equipment (PPE) might have been hindered by availability.
Another issue with far-reaching consequences is diversion of resources for urgent care. Health care professionals may simply not be readily available for those with chronic care needs because of the enormous scale of the impact of COVID-19 upon health care systems.
Continuity of therapy might include evaluations or follow-up services via teleconferencing modalities, but an exhaustive initial onsite physical examination is often necessary for accurate diagnostics and care. Medication management for the newly diagnosed AD or dementia patient necessitates a thorough screening process involving appropriate in-person blood or laboratory work. It is for this reason that clinicians will need to plan ahead by preparing a contingency plan with the corresponding mitigation strategies (for example, telemedicine, proposed solutions to anticipated disruption of services, extended support, and feedback from family members, etc.).2
Resilience and recovery in a retrospective study
A research team from Wuhan Red Cross Hospital in China performed a retrospective cohort study on a sample of patients (n = 42) to determine the severity and prognostic features of COVID-19 pneumonia; 19 AD patients (as per National Institute on Aging/Alzheimer’s Association diagnostic guidelines) were directly compared with 23 age-matched non-AD COVID-19 patients in a similar treatment context.
The study yielded some rather unexpected findings, namely, AD patients experienced remarkably shorter hospital stays and better appetites, especially with respect to their non-AD counterparts. This is even more puzzling when considering that previous studies indicated that dementia patients with concomitant COVID-19 pneumonia are twice as likely to die as those without neurodegenerative compromise.
Aside from a seemingly inexplicable interest in food, the observable positive changes may be attributable to such factors that are particular to the nursing home – residents have immediate access to health care services, which generally allows for timely diagnosis and care. However, the authors of the study speculate that the pathophysiological response of angiotensin-converting enzyme 2 (ACE2) confers to AD patients a therapeutic advantage as they have reduced expression.3 Despite the notoriously high mortality rates of COVID-19 pneumonia among the elderly population, , which underscores the importance of early diagnosis and intervention.
Genetic and environmental susceptibility
One of the more devastating observations about the ongoing pandemic environment is that a whopping 80% of dying patients committed to a long-term facility also include those with AD; it has been reported that almost half of all patients in nursing homes and related services have the neurodegenerative condition. The grim scenario is brought about by several factors, chief of which is the proximity of shared living arrangements within the context of a residential care setting. It should be noted that patients with AD exhibit comorbid conditions (for example, diabetes, cardiovascular disease, and/or respiratory issues) that immediately put them at high risk for COVID exposure. Interestingly enough, the ApoE4 genotype, which is associated with an increased susceptibility for AD, is also correlated with COVID-19 prognosis and severity. Although exact numbers are difficult to come by, it is of utmost importance for clinicians to evaluate the overall scope of the situation, identify at-risk patients such as individuals with AD and related dementias, and work with caregivers to afford care to patients who need it the most.4
Transcending research design
The elderly population, unsurprisingly, experiences the highest COVID-19 mortality rate because of the presence of multiple risk factors, namely, compromised immunity and difficulties maintaining ADLs, and thereby adhering to safety protocols. As far as Alzheimer’s patients are concerned, numerous hurdles affect the domain of neurodegenerative research.
To safeguard the health and well-being of the participants and caregivers, site sponsors and investigators must explore various communication avenues with the goal of facilitating health education (for example, mitigation strategies, adverse effects monitoring, etc.), as well as implementing contingency plans in the event that a site becomes inaccessible (for example, site closure, traveling regulations, lockdowns, etc.).
Alternatives such as telemedicine present viable solutions for ensuring completion of studies. Given the nature of the pandemic, there is a possibility that a research participant may contract the virus, necessitating a break from the established protocol. It is for this reason that site sponsors and corresponding regulatory bodies are advised to proactively engage in dialogue and transparent communications with respect to ensuing protocol deviations. Institutional Review Boards can expedite the review process by making the necessary changes in a timely manner.5
References
1. Ritchie K. KJZZ. 2020 Nov 16.
2. Brown EE et al. Am J Geriatr Psychiatry. 2020 Jul;28(7):712-21.
3. Li J et al. J Alzheimers Dis. 2020;77(1):67-73.
4. Perry G. J Alzheimers Dis. 2020 Jan 1;76(1):1.
5. Alzheimers Dement. 2020 Apr;16(4):587-8.
Dr. Islam is a medical adviser for the International Maternal and Child Health Foundation, Montreal, and is based in New York. He also is a postdoctoral fellow, psychopharmacologist, and a board-certified medical affairs specialist. Dr. Islam disclosed no relevant financial relationships.
Dr. Dhillon is a staff neurologist at Brigham and Women’s Hospital in Boston. Dr. Dhillon is currently on the speaker bureau/advisory board for Biogen, Bristol Myers Squibb, Genzyme, and Teva Neuroscience.
Dr. Choudhry is the chief scientific officer and head of the department of mental health and clinical research at the International Maternal and Child Health Foundation. He has no disclosures.
An alarming number of additional Alzheimer’s disease (AD) deaths have been reported across various states within the past several months, according to the Alzheimer’s Association. Centers for Disease Control and Prevention data indicate that no less than 31,000 additional people with the neurodegenerative condition had died from the beginning of the pandemic through the end of September 2020. We know that long-term care facilities have been hit hardest, and access to adequate and/or prompt testing has been cited as the most pressing issue during the onset of the pandemic.1
When ADLs become a matter of survival
For individuals affected with Alzheimer’s disease and other types of dementia, performing routine tasks may seem cumbersome and overwhelming. Many of these patients are dependent upon caregivers and family support to facilitate their activities of daily living (ADLs).
Transitioning into the “new normal” set by the pandemic milieu is not an easy task for the average AD individual, because they are now expected to comply with numerous safety instructions (for example, maintaining hygiene, social distancing, etc.). They are also expected to monitor and communicate information about the onset of any suspected symptom to their caregiver or health care clinician.
The additional tasks added to their list of ADLs are particularly distressing given their already compromised short-term memory and overall cognitive decline. Individuals with AD may also be dealing with a host of psychobehavioral challenges, such as the presence of depression, anxiety, and/or agitation amid self-isolation. Enforced social isolation tied to COVID-19 may compound those issues.
Resource diversion and mitigation strategies
Unfortunately, any disruption in services within a long-term care setting may lead to a suboptimal therapeutic environment for patients. The Washington State LTC, for example, reported experiencing a case fatality rate (CFR) exceeding more than a third of its residents; essential staff and health care clinicians were duly affected from exposure to the virus (the risk of transmission increases considerably during transport between facilities). Access to personal protective equipment (PPE) might have been hindered by availability.
Another issue with far-reaching consequences is diversion of resources for urgent care. Health care professionals may simply not be readily available for those with chronic care needs because of the enormous scale of the impact of COVID-19 upon health care systems.
Continuity of therapy might include evaluations or follow-up services via teleconferencing modalities, but an exhaustive initial onsite physical examination is often necessary for accurate diagnostics and care. Medication management for the newly diagnosed AD or dementia patient necessitates a thorough screening process involving appropriate in-person blood or laboratory work. It is for this reason that clinicians will need to plan ahead by preparing a contingency plan with the corresponding mitigation strategies (for example, telemedicine, proposed solutions to anticipated disruption of services, extended support, and feedback from family members, etc.).2
Resilience and recovery in a retrospective study
A research team from Wuhan Red Cross Hospital in China performed a retrospective cohort study on a sample of patients (n = 42) to determine the severity and prognostic features of COVID-19 pneumonia; 19 AD patients (as per National Institute on Aging/Alzheimer’s Association diagnostic guidelines) were directly compared with 23 age-matched non-AD COVID-19 patients in a similar treatment context.
The study yielded some rather unexpected findings, namely, AD patients experienced remarkably shorter hospital stays and better appetites, especially with respect to their non-AD counterparts. This is even more puzzling when considering that previous studies indicated that dementia patients with concomitant COVID-19 pneumonia are twice as likely to die as those without neurodegenerative compromise.
Aside from a seemingly inexplicable interest in food, the observable positive changes may be attributable to such factors that are particular to the nursing home – residents have immediate access to health care services, which generally allows for timely diagnosis and care. However, the authors of the study speculate that the pathophysiological response of angiotensin-converting enzyme 2 (ACE2) confers to AD patients a therapeutic advantage as they have reduced expression.3 Despite the notoriously high mortality rates of COVID-19 pneumonia among the elderly population, , which underscores the importance of early diagnosis and intervention.
Genetic and environmental susceptibility
One of the more devastating observations about the ongoing pandemic environment is that a whopping 80% of dying patients committed to a long-term facility also include those with AD; it has been reported that almost half of all patients in nursing homes and related services have the neurodegenerative condition. The grim scenario is brought about by several factors, chief of which is the proximity of shared living arrangements within the context of a residential care setting. It should be noted that patients with AD exhibit comorbid conditions (for example, diabetes, cardiovascular disease, and/or respiratory issues) that immediately put them at high risk for COVID exposure. Interestingly enough, the ApoE4 genotype, which is associated with an increased susceptibility for AD, is also correlated with COVID-19 prognosis and severity. Although exact numbers are difficult to come by, it is of utmost importance for clinicians to evaluate the overall scope of the situation, identify at-risk patients such as individuals with AD and related dementias, and work with caregivers to afford care to patients who need it the most.4
Transcending research design
The elderly population, unsurprisingly, experiences the highest COVID-19 mortality rate because of the presence of multiple risk factors, namely, compromised immunity and difficulties maintaining ADLs, and thereby adhering to safety protocols. As far as Alzheimer’s patients are concerned, numerous hurdles affect the domain of neurodegenerative research.
To safeguard the health and well-being of the participants and caregivers, site sponsors and investigators must explore various communication avenues with the goal of facilitating health education (for example, mitigation strategies, adverse effects monitoring, etc.), as well as implementing contingency plans in the event that a site becomes inaccessible (for example, site closure, traveling regulations, lockdowns, etc.).
Alternatives such as telemedicine present viable solutions for ensuring completion of studies. Given the nature of the pandemic, there is a possibility that a research participant may contract the virus, necessitating a break from the established protocol. It is for this reason that site sponsors and corresponding regulatory bodies are advised to proactively engage in dialogue and transparent communications with respect to ensuing protocol deviations. Institutional Review Boards can expedite the review process by making the necessary changes in a timely manner.5
References
1. Ritchie K. KJZZ. 2020 Nov 16.
2. Brown EE et al. Am J Geriatr Psychiatry. 2020 Jul;28(7):712-21.
3. Li J et al. J Alzheimers Dis. 2020;77(1):67-73.
4. Perry G. J Alzheimers Dis. 2020 Jan 1;76(1):1.
5. Alzheimers Dement. 2020 Apr;16(4):587-8.
Dr. Islam is a medical adviser for the International Maternal and Child Health Foundation, Montreal, and is based in New York. He also is a postdoctoral fellow, psychopharmacologist, and a board-certified medical affairs specialist. Dr. Islam disclosed no relevant financial relationships.
Dr. Dhillon is a staff neurologist at Brigham and Women’s Hospital in Boston. Dr. Dhillon is currently on the speaker bureau/advisory board for Biogen, Bristol Myers Squibb, Genzyme, and Teva Neuroscience.
Dr. Choudhry is the chief scientific officer and head of the department of mental health and clinical research at the International Maternal and Child Health Foundation. He has no disclosures.
An alarming number of additional Alzheimer’s disease (AD) deaths have been reported across various states within the past several months, according to the Alzheimer’s Association. Centers for Disease Control and Prevention data indicate that no less than 31,000 additional people with the neurodegenerative condition had died from the beginning of the pandemic through the end of September 2020. We know that long-term care facilities have been hit hardest, and access to adequate and/or prompt testing has been cited as the most pressing issue during the onset of the pandemic.1
When ADLs become a matter of survival
For individuals affected with Alzheimer’s disease and other types of dementia, performing routine tasks may seem cumbersome and overwhelming. Many of these patients are dependent upon caregivers and family support to facilitate their activities of daily living (ADLs).
Transitioning into the “new normal” set by the pandemic milieu is not an easy task for the average AD individual, because they are now expected to comply with numerous safety instructions (for example, maintaining hygiene, social distancing, etc.). They are also expected to monitor and communicate information about the onset of any suspected symptom to their caregiver or health care clinician.
The additional tasks added to their list of ADLs are particularly distressing given their already compromised short-term memory and overall cognitive decline. Individuals with AD may also be dealing with a host of psychobehavioral challenges, such as the presence of depression, anxiety, and/or agitation amid self-isolation. Enforced social isolation tied to COVID-19 may compound those issues.
Resource diversion and mitigation strategies
Unfortunately, any disruption in services within a long-term care setting may lead to a suboptimal therapeutic environment for patients. The Washington State LTC, for example, reported experiencing a case fatality rate (CFR) exceeding more than a third of its residents; essential staff and health care clinicians were duly affected from exposure to the virus (the risk of transmission increases considerably during transport between facilities). Access to personal protective equipment (PPE) might have been hindered by availability.
Another issue with far-reaching consequences is diversion of resources for urgent care. Health care professionals may simply not be readily available for those with chronic care needs because of the enormous scale of the impact of COVID-19 upon health care systems.
Continuity of therapy might include evaluations or follow-up services via teleconferencing modalities, but an exhaustive initial onsite physical examination is often necessary for accurate diagnostics and care. Medication management for the newly diagnosed AD or dementia patient necessitates a thorough screening process involving appropriate in-person blood or laboratory work. It is for this reason that clinicians will need to plan ahead by preparing a contingency plan with the corresponding mitigation strategies (for example, telemedicine, proposed solutions to anticipated disruption of services, extended support, and feedback from family members, etc.).2
Resilience and recovery in a retrospective study
A research team from Wuhan Red Cross Hospital in China performed a retrospective cohort study on a sample of patients (n = 42) to determine the severity and prognostic features of COVID-19 pneumonia; 19 AD patients (as per National Institute on Aging/Alzheimer’s Association diagnostic guidelines) were directly compared with 23 age-matched non-AD COVID-19 patients in a similar treatment context.
The study yielded some rather unexpected findings, namely, AD patients experienced remarkably shorter hospital stays and better appetites, especially with respect to their non-AD counterparts. This is even more puzzling when considering that previous studies indicated that dementia patients with concomitant COVID-19 pneumonia are twice as likely to die as those without neurodegenerative compromise.
Aside from a seemingly inexplicable interest in food, the observable positive changes may be attributable to such factors that are particular to the nursing home – residents have immediate access to health care services, which generally allows for timely diagnosis and care. However, the authors of the study speculate that the pathophysiological response of angiotensin-converting enzyme 2 (ACE2) confers to AD patients a therapeutic advantage as they have reduced expression.3 Despite the notoriously high mortality rates of COVID-19 pneumonia among the elderly population, , which underscores the importance of early diagnosis and intervention.
Genetic and environmental susceptibility
One of the more devastating observations about the ongoing pandemic environment is that a whopping 80% of dying patients committed to a long-term facility also include those with AD; it has been reported that almost half of all patients in nursing homes and related services have the neurodegenerative condition. The grim scenario is brought about by several factors, chief of which is the proximity of shared living arrangements within the context of a residential care setting. It should be noted that patients with AD exhibit comorbid conditions (for example, diabetes, cardiovascular disease, and/or respiratory issues) that immediately put them at high risk for COVID exposure. Interestingly enough, the ApoE4 genotype, which is associated with an increased susceptibility for AD, is also correlated with COVID-19 prognosis and severity. Although exact numbers are difficult to come by, it is of utmost importance for clinicians to evaluate the overall scope of the situation, identify at-risk patients such as individuals with AD and related dementias, and work with caregivers to afford care to patients who need it the most.4
Transcending research design
The elderly population, unsurprisingly, experiences the highest COVID-19 mortality rate because of the presence of multiple risk factors, namely, compromised immunity and difficulties maintaining ADLs, and thereby adhering to safety protocols. As far as Alzheimer’s patients are concerned, numerous hurdles affect the domain of neurodegenerative research.
To safeguard the health and well-being of the participants and caregivers, site sponsors and investigators must explore various communication avenues with the goal of facilitating health education (for example, mitigation strategies, adverse effects monitoring, etc.), as well as implementing contingency plans in the event that a site becomes inaccessible (for example, site closure, traveling regulations, lockdowns, etc.).
Alternatives such as telemedicine present viable solutions for ensuring completion of studies. Given the nature of the pandemic, there is a possibility that a research participant may contract the virus, necessitating a break from the established protocol. It is for this reason that site sponsors and corresponding regulatory bodies are advised to proactively engage in dialogue and transparent communications with respect to ensuing protocol deviations. Institutional Review Boards can expedite the review process by making the necessary changes in a timely manner.5
References
1. Ritchie K. KJZZ. 2020 Nov 16.
2. Brown EE et al. Am J Geriatr Psychiatry. 2020 Jul;28(7):712-21.
3. Li J et al. J Alzheimers Dis. 2020;77(1):67-73.
4. Perry G. J Alzheimers Dis. 2020 Jan 1;76(1):1.
5. Alzheimers Dement. 2020 Apr;16(4):587-8.
Dr. Islam is a medical adviser for the International Maternal and Child Health Foundation, Montreal, and is based in New York. He also is a postdoctoral fellow, psychopharmacologist, and a board-certified medical affairs specialist. Dr. Islam disclosed no relevant financial relationships.
Dr. Dhillon is a staff neurologist at Brigham and Women’s Hospital in Boston. Dr. Dhillon is currently on the speaker bureau/advisory board for Biogen, Bristol Myers Squibb, Genzyme, and Teva Neuroscience.
Dr. Choudhry is the chief scientific officer and head of the department of mental health and clinical research at the International Maternal and Child Health Foundation. He has no disclosures.
Microvascular injury of brain, olfactory bulb seen in COVID-19
new research suggests.
Postmortem MRI brain scans of 13 patients who died from COVID-19 showed abnormalities in 10 of the participants. Of these, nine showed punctate hyperintensities, “which represented areas of microvascular injury and fibrinogen leakage,” the investigators reported. Immunostaining also showed a thinning of the basal lamina in five of these patients.
Further analyses showed punctate hypointensities linked to congested blood vessels in 10 patients. These areas were “interpreted as microhemorrhages,” the researchers noted.
There was no evidence of viral infection, including SARS-CoV-2.
“These findings may inform the interpretation of changes observed on [MRI] of punctate hyperintensities and linear hypointensities in patients with COVID-19,” wrote Myoung-Hwa Lee, PhD, a research fellow at the National Institute of Neurological Disorders and Stroke, and colleagues. The findings were published online Dec. 30 in a “correspondence” piece in the New England Journal of Medicine.
Interpret with caution
The investigators examined brains from a convenience sample of 19 patients (mean age, 50 years), all of whom died from COVID-19 between March and July 2020.
An 11.7-tesla scanner was used to obtain magnetic resonance microscopy images for 13 of the patients. In order to scan the olfactory bulb, the scanner was set at a resolution of 25 mcm; for the brain, it was set at 100 mcm.
Chromogenic immunostaining was used to assess brain abnormalities found in 10 of the patients. Multiplex fluorescence imaging was also used for some of the patients.
For 18 study participants, a histopathological brain examination was performed. In the patients who also had medical histories available to the researchers, five had mild respiratory syndrome, four had acute respiratory distress syndrome, two had pulmonary embolism, one had delirium, and three had unknown symptoms.
The punctate hyperintensities found on magnetic resonance microscopy were also found on histopathological exam. Collagen IV immunostaining showed a thinning in the basal lamina of endothelial cells in these areas.
In addition to congested blood vessels, punctate hypointensities were linked to areas of fibrinogen leakage – but also to “relatively intact vasculature,” the investigators reported.
“There was minimal perivascular inflammation in the specimens examined, but there was no vascular occlusion,” they added.
SARS-CoV-2 was also not found in any of the participants. “It is possible that the virus was cleared by the time of death or that viral copy numbers were below the level of detection by our assays,” the researchers noted.
In 13 of the patients, hypertrophic astrocytes, macrophage infiltrates, and perivascular-activated microglia were found. Eight patients showed CD3+ and CD8+ T cells in spaces and lumens next to endothelial cells.
Finally, five patients showed activated microglia next to neurons. This is “suggestive of neuronophagia in the olfactory bulb, substantial nigra, dorsal motor nucleus of the vagal nerve, and the pre-Bötzinger complex in the medulla, which is involved in the generation of spontaneous rhythmic breathing,” wrote the investigators.
In summary, vascular pathology was found in 10 cases, perivascular infiltrates were present in 13 cases, acute ischemic hypoxic neurons were present in 6 cases, and changes suggestive of neuronophagia were present in 5 cases.
The researchers noted that, although the study findings may be helpful when interpreting brain changes on MRI scan in this patient population, availability of clinical information for the participants was limited.
Therefore, “no conclusions can be drawn in relation to neurologic features of COVID-19,” they wrote.
The study was funded by NINDS. Dr. Lee and all but one of the other investigators reported no relevant financial relationships; the remaining investigator reported having received grants from NINDS during the conduct of this study.
A version of this article first appeared on Medscape.com.
new research suggests.
Postmortem MRI brain scans of 13 patients who died from COVID-19 showed abnormalities in 10 of the participants. Of these, nine showed punctate hyperintensities, “which represented areas of microvascular injury and fibrinogen leakage,” the investigators reported. Immunostaining also showed a thinning of the basal lamina in five of these patients.
Further analyses showed punctate hypointensities linked to congested blood vessels in 10 patients. These areas were “interpreted as microhemorrhages,” the researchers noted.
There was no evidence of viral infection, including SARS-CoV-2.
“These findings may inform the interpretation of changes observed on [MRI] of punctate hyperintensities and linear hypointensities in patients with COVID-19,” wrote Myoung-Hwa Lee, PhD, a research fellow at the National Institute of Neurological Disorders and Stroke, and colleagues. The findings were published online Dec. 30 in a “correspondence” piece in the New England Journal of Medicine.
Interpret with caution
The investigators examined brains from a convenience sample of 19 patients (mean age, 50 years), all of whom died from COVID-19 between March and July 2020.
An 11.7-tesla scanner was used to obtain magnetic resonance microscopy images for 13 of the patients. In order to scan the olfactory bulb, the scanner was set at a resolution of 25 mcm; for the brain, it was set at 100 mcm.
Chromogenic immunostaining was used to assess brain abnormalities found in 10 of the patients. Multiplex fluorescence imaging was also used for some of the patients.
For 18 study participants, a histopathological brain examination was performed. In the patients who also had medical histories available to the researchers, five had mild respiratory syndrome, four had acute respiratory distress syndrome, two had pulmonary embolism, one had delirium, and three had unknown symptoms.
The punctate hyperintensities found on magnetic resonance microscopy were also found on histopathological exam. Collagen IV immunostaining showed a thinning in the basal lamina of endothelial cells in these areas.
In addition to congested blood vessels, punctate hypointensities were linked to areas of fibrinogen leakage – but also to “relatively intact vasculature,” the investigators reported.
“There was minimal perivascular inflammation in the specimens examined, but there was no vascular occlusion,” they added.
SARS-CoV-2 was also not found in any of the participants. “It is possible that the virus was cleared by the time of death or that viral copy numbers were below the level of detection by our assays,” the researchers noted.
In 13 of the patients, hypertrophic astrocytes, macrophage infiltrates, and perivascular-activated microglia were found. Eight patients showed CD3+ and CD8+ T cells in spaces and lumens next to endothelial cells.
Finally, five patients showed activated microglia next to neurons. This is “suggestive of neuronophagia in the olfactory bulb, substantial nigra, dorsal motor nucleus of the vagal nerve, and the pre-Bötzinger complex in the medulla, which is involved in the generation of spontaneous rhythmic breathing,” wrote the investigators.
In summary, vascular pathology was found in 10 cases, perivascular infiltrates were present in 13 cases, acute ischemic hypoxic neurons were present in 6 cases, and changes suggestive of neuronophagia were present in 5 cases.
The researchers noted that, although the study findings may be helpful when interpreting brain changes on MRI scan in this patient population, availability of clinical information for the participants was limited.
Therefore, “no conclusions can be drawn in relation to neurologic features of COVID-19,” they wrote.
The study was funded by NINDS. Dr. Lee and all but one of the other investigators reported no relevant financial relationships; the remaining investigator reported having received grants from NINDS during the conduct of this study.
A version of this article first appeared on Medscape.com.
new research suggests.
Postmortem MRI brain scans of 13 patients who died from COVID-19 showed abnormalities in 10 of the participants. Of these, nine showed punctate hyperintensities, “which represented areas of microvascular injury and fibrinogen leakage,” the investigators reported. Immunostaining also showed a thinning of the basal lamina in five of these patients.
Further analyses showed punctate hypointensities linked to congested blood vessels in 10 patients. These areas were “interpreted as microhemorrhages,” the researchers noted.
There was no evidence of viral infection, including SARS-CoV-2.
“These findings may inform the interpretation of changes observed on [MRI] of punctate hyperintensities and linear hypointensities in patients with COVID-19,” wrote Myoung-Hwa Lee, PhD, a research fellow at the National Institute of Neurological Disorders and Stroke, and colleagues. The findings were published online Dec. 30 in a “correspondence” piece in the New England Journal of Medicine.
Interpret with caution
The investigators examined brains from a convenience sample of 19 patients (mean age, 50 years), all of whom died from COVID-19 between March and July 2020.
An 11.7-tesla scanner was used to obtain magnetic resonance microscopy images for 13 of the patients. In order to scan the olfactory bulb, the scanner was set at a resolution of 25 mcm; for the brain, it was set at 100 mcm.
Chromogenic immunostaining was used to assess brain abnormalities found in 10 of the patients. Multiplex fluorescence imaging was also used for some of the patients.
For 18 study participants, a histopathological brain examination was performed. In the patients who also had medical histories available to the researchers, five had mild respiratory syndrome, four had acute respiratory distress syndrome, two had pulmonary embolism, one had delirium, and three had unknown symptoms.
The punctate hyperintensities found on magnetic resonance microscopy were also found on histopathological exam. Collagen IV immunostaining showed a thinning in the basal lamina of endothelial cells in these areas.
In addition to congested blood vessels, punctate hypointensities were linked to areas of fibrinogen leakage – but also to “relatively intact vasculature,” the investigators reported.
“There was minimal perivascular inflammation in the specimens examined, but there was no vascular occlusion,” they added.
SARS-CoV-2 was also not found in any of the participants. “It is possible that the virus was cleared by the time of death or that viral copy numbers were below the level of detection by our assays,” the researchers noted.
In 13 of the patients, hypertrophic astrocytes, macrophage infiltrates, and perivascular-activated microglia were found. Eight patients showed CD3+ and CD8+ T cells in spaces and lumens next to endothelial cells.
Finally, five patients showed activated microglia next to neurons. This is “suggestive of neuronophagia in the olfactory bulb, substantial nigra, dorsal motor nucleus of the vagal nerve, and the pre-Bötzinger complex in the medulla, which is involved in the generation of spontaneous rhythmic breathing,” wrote the investigators.
In summary, vascular pathology was found in 10 cases, perivascular infiltrates were present in 13 cases, acute ischemic hypoxic neurons were present in 6 cases, and changes suggestive of neuronophagia were present in 5 cases.
The researchers noted that, although the study findings may be helpful when interpreting brain changes on MRI scan in this patient population, availability of clinical information for the participants was limited.
Therefore, “no conclusions can be drawn in relation to neurologic features of COVID-19,” they wrote.
The study was funded by NINDS. Dr. Lee and all but one of the other investigators reported no relevant financial relationships; the remaining investigator reported having received grants from NINDS during the conduct of this study.
A version of this article first appeared on Medscape.com.
The top pediatric articles of 2019
Updates in pediatric hospital medicine
The expansion of the field of pediatric hospital medicine in the past 30 years has resulted in improved health care outcomes for hospitalized children1,2 and has been accompanied by a robust increase in the amount of scholarly work related to the field.3 We performed a review of the literature published in 2019 to identify the 10 articles that had the most impact on pediatric hospital medicine, and presented the findings at HM20 Virtual, the 2020 annual conference of the Society of Hospital Medicine. Five of the selected articles are highlighted here.
STUDY 1
Wechsler ME et al. Step-up therapy in black children and adults with poorly controlled asthma. N Engl J Med. 2019 Sep 26;381(13):1227-39.
Background
Current pediatric asthma guidelines suggest adding a long-acting beta-agonist (LABA) to inhaled corticosteroid (ICS) therapy, rather than increasing the ICS dose, for children with poorly controlled asthma. However, these data are based on trials with disproportionately few Black subjects. This study aimed to determine the best step-up therapy for Black patients whose asthma was poorly controlled on ICS monotherapy.
Study overview and results
The authors reported two parallel double-blind, randomized, controlled trials, one in children and one in adolescents and adults. The study of children included 280 subjects ranging in age from 5 to 11, with at least one Black grandparent, and with poorly controlled asthma on low-dose ICS therapy. It used a four-way crossover design in which each subject was treated with four different 14-week treatment regimens: either double (medium-dose) or quintuple (high-dose) their baseline ICS dose, with or without the addition of a LABA. A superior response was defined by the composite outcome of at least one fewer asthma exacerbation, more asthma-control days, or a 5–percentage point difference in predicted FEV1. Forty-six percent of children had improved asthma outcomes when the ICS dose was increased rather than with the addition of a LABA. In contrast, Black adolescents and Black adults had superior responses to the addition of a LABA. There was no significant interaction between the percentage of African ancestry as determined by DNA genotyping and the primary composite outcome. High-dose ICS was associated with a decrease in the ratio of urinary cortisol to creatinine in children younger than 8 years.
Limitations
Approximately 25% of children dropped out of the study, with disproportionately more children dropping out while on a high-dose ICS regimen. Additionally, the difference in the composite outcome was primarily driven by differences in FEV1, with few subjects demonstrating a difference in asthma exacerbations or asthma-control days. Although a decrease in urinary cortisol to creatinine ratio was noted in children under 8 on high-dose ICS, the study period was not long enough to determine the clinical implications of this finding.
Important findings and implications
While studies with a majority of white children have suggested a superior response from adding a LABA compared to increasing the dose of an ICS, almost half of Black children showed a superior response when the dose of an ICS was increased rather than adding a LABA. It is important to note that current guidelines are based on studies with a disproportionate majority of white subjects and may not accurately reflect optimal care for patients in other racial groups. This study underscores the need to include a diverse patient population in research studies.
STUDY 2
Chang PW, Newman TB. A simpler prediction rule for rebound hyperbilirubinemia. Pediatrics. 2019 Jul;144(1):e20183712.
Background
Hyperbilirubinemia (jaundice) is estimated to affect 50%-60% of all newborns. Rebound hyperbilirubinemia – a rise in bilirubin after cessation of phototherapy – is common and can lead to recently discharged infants being readmitted for additional therapy. Lack of clear guidelines regarding when to discharge infants with hyperbilirubinemia has likely contributed to practice variation and some trepidation regarding whether a bilirubin level is “low enough” to discontinue therapy.
Study overview and results
The authors had previously proposed a three-factor hyperbilirubinemia risk model and sought to simplify their rule further.4 They examined a retrospective cohort of 7,048 infants greater than or equal to 35 weeks’ gestation using a random split sample. The authors derived a two-factor model using the same methods and compared its performance to the three-factor model. The two-factor formula was shown to be a good fit as a logistic regression model (Hosmer-Lemeshow test 9.21; P = .33), and the AUROC (area under the receiver operating characteristic) curves for the derivation and validation cohorts were similar between the two-factor (0.877 and 0.876, respectively) and three-factor risk models (0.887 and 0.881, respectively).
Limitations
These data are limited to infants receiving their first treatment of phototherapy and have not been externally validated. An important variable, serum bilirubin at phototherapy termination, was estimated in most subjects, which may have affected the accuracy of the prediction rule. Whether infants received home phototherapy was based only on equipment orders, and some infants may have received phototherapy unbeknownst to investigators. Last, infants with rebound hyperbilirubinemia at less than 72 hours after phototherapy discontinuation may have been missed.
Important findings and implications
This prediction model provides evidence-based, concrete data that can be used in making joint decisions with families regarding discharge timing of infants with hyperbilirubinemia. It also could be beneficial when deciding appropriate follow-up time after discharge.
STUDY 3
Ramgopal S et al. Risk of serious bacterial infection in infants aged ≤60 days presenting to emergency departments with a history of fever only. J Pediatr. 2019 Jan;204:191-195. doi: 10.1016/j.jpeds.2018.08.043.
Background
Febrile infants aged 60 days and younger are at risk for serious bacterial infections (SBI) including urinary tract infections (UTI), bacteremia, and meningitis. As physical exam is a poor discriminator of SBI in this age group, providers frequently rely on laboratory values and risk factors to guide management. Infants presenting with documented fevers by caregivers but found to have no fever in the emergency department are a challenge, and there are limited data regarding SBI frequency in this population.
Study overview and results
The authors performed a secondary analysis of a prospectively gathered cohort of infants aged 60 days and younger within the Pediatric Emergency Care Applied Research Network (PECARN) who had blood, urine, and CSF data available. Notable exclusions included infants who were premature, had a focal infection, were clinically ill, had recent antibiotic use, did not have blood, urine, and CSF data available, or were lost to telephone follow-up at 7 days to ensure wellness. The study cohort included 6,014 infants, 1,233 (32%) who were febrile by history alone. Rates of overall SBI were lower in the afebrile group (8.8% vs. 12.8%). For infants 0-28 days, rates of UTI were lower for the afebrile group (9.5% vs. 14.5%), but there was no difference in the rates of bacteremia or meningitis. For infants 29-60 days, rates of UTI (6.6% vs. 9.3%) and bacteremia (.5% vs. 1.7%) were lower in the afebrile group.
Limitations
Neither the use of home antipyretics nor the method of temperature taking at home were studied. Also, as this was a secondary analysis, it is possible that not all infants who presented with history of fever only were captured, as work-up was dictated by individual treating providers who may have chosen not to work up certain afebrile infants.
Important findings and implications
Nearly one-third of infants presenting for fever evaluation are afebrile on arrival. Although overall rates of SBI were lower in the group with fever by history only, this difference is largely accounted for by differing rates of UTI. Rates of bacteremia and meningitis remained substantial between groups, particularly for infants aged 0-28 days. Because of the significant morbidity associated with these infections, it is reasonable to suggest that absence of fever on presentation alone should not alter clinical or laboratory work-up, particularly in infants 0-28 days.
STUDY 4
Humphrey-Murto S et al. The influence of prior performance information on ratings of current performance and implications for learner handover: A scoping review. Acad Med. 2019 Jul;94(7):1050-7.
Background
Learner Handover (LH) or “forward feeding” occurs when information about trainees is shared between faculty supervisors. Although this can be helpful to tailor educational experiences and build upon previous assessments, it risks stigmatizing trainees and adding bias to future feedback and assessments as the trainee never really has a “clean slate.” In this study, the authors sought to uncover the key concepts of how prior performance information (PPI) influences assessments and any implications for medical education.
Study overview and results
The authors performed a cross-disciplinary scoping review looking at over 17,000 articles published between 1980 and 2017 across the domains of psychology, sports, business, and education. Seven themes were identified with the following notable findings. Raters exposed to positive PPI scored a learner’s performance higher, and vice versa. There was a dose-response relationship with more positive and more negative PPI resulting in higher and lower assessments, respectively. General standards, such as a direction to complete all work in a timely manner, caused an assimilation effect, while specific standards, such as a direction to complete a certain task by a certain day, did not. More motivated and more experienced raters are less affected by PPI, and those who believe that people can change (incremental theorists) are less affected by PPI while those who believe personal attributes are fixed (entity theorists) are more affected.
Limitations
The heterogeneity of the studies and the fact that they were largely conducted in experimental settings may limit generalizability to medical education. Slightly less than half of the studies included a control arm. Last, most of the studies looked at the ratings of only one target performance, not multiple performances over time.
Important findings and implications
Ratings of current performance displace toward PPI direction, with negative PPI more influential than positive PPI. In a formative setting, PPI may help the assessor focus on areas of possible weakness. In contrast, for a summative assessment, PPI may be prejudicial and have an impact on the rating given to the student. Clinicians should be mindful of the information they share with future raters about learners and the potential bias on future assessments that can manifest as a result.
STUDY 5
McCann ME et al. Neurodevelopmental outcome at 5 years of age after general anaesthesia or awake-regional anaesthesia in infancy (GAS): An international, multicentre, randomised, controlled equivalence trial. The Lancet. 2019 Feb;393:664-77.
Background
Animal models and observational studies have suggested a link between early anesthesia exposure and adverse neurocognitive outcomes; however, findings have been mixed and studies are prone to confounding. This study is the first randomized controlled trial to compare neurocognitive outcomes for infants exposed to general anesthesia versus awake-regional anesthesia.
Study overview and results
In this international, multicenter, assessor-masked trial, 722 infants undergoing inguinal hernia repair were randomized to awake-regional anesthesia or single-agent sevoflurane-based general anesthesia. Infants born at greater than 26 weeks’ gestational age were eligible, while those with prior anesthesia exposure or risks for neurocognitive delay were excluded. The primary outcome was full-scale intelligence quotient (FSIQ) testing at 5 years of age on the Wechsler Preschool and Primary Scale of Intelligence, third edition (WPPSI-III). Seven additional neurodevelopmental assessments and parental questionnaires regarding behavior were administered as secondary outcomes. Average anesthesia exposure was 54 minutes and no infant had exposure greater than 120 minutes. There was no significant difference in mean scores on WPPSI-III FSIQ testing, and no difference in the additional neurocognitive assessments or parent-reported outcomes used as secondary outcomes.
Limitations
This study was limited to single, short periods of single-agent anesthesia exposure in children with no additional neurologic risk factors, so caution should be used in extrapolating these data to children with medical complexity and children undergoing multiple procedures, longer surgeries, or multidrug anesthetic regimens. The study population was majority male because of the surgical pathology selected and included only children in the narrow range of postmenstrual age 60 weeks or less. While this population represents a suspected a period of high cerebral vulnerability based on animal models, the implications of anesthesia exposure at other ages are unclear.
Important findings and implications
An estimated 10% of children from developed countries are exposed to general anesthesia during the first 3 years of life. While hospitalists do not typically select the route of anesthesia, they frequently care for patients undergoing procedures and must address parental concerns regarding the safety of anesthesia exposure. Given the rigorous study methods and long-term follow up in the current study, these data should provide reassurance that, for healthy infants undergoing short, single-agent anesthetic exposure, there is no evidence of future adverse neurologic outcomes.
Dr. Russo is director of pediatrics, medical director for quality and innovation, at WellSpan Health, York, Pa. Dr. Money is a pediatric hospitalist at Primary Children’s Hospital, University of Utah School of Medicine, Salt Lake City. Dr. Steed is instructor of hospital medicine, Northwestern Memorial Hospital and Ann and Robert H. Lurie Children’s Hospital of Chicago, Northwestern University School of Medicine, Chicago. The authors would like to thank Dr. Klint M. Schwenk and the Society for Hospital Medicine Pediatric Special Interest Group Executive Council.
References
1. Roberts KB, Fisher ER, and Rauch DA. The history of pediatric hospital medicine in the United States, 1996-2019. J Hosp Med. 2020 Jul;15(7):424-7.
2. Mussman GM and Conway PH. Pediatric hospitalist systems versus traditional models of care: Effect on quality and cost outcomes. J Hosp Med. 2012 Apr;7(4):350-7.
3. Wang ME, Shaughnessy EE, and Leyenaar JK. The future of pediatric hospital medicine: Challenges and opportunities. J Hosp Med. 2020 Jul;15(7):428-30.
4. Chang PW et al. A clinical prediction rule for rebound hyperbilirubinemia following inpatient phototherapy. Pediatrics. 2017;139 Mar;139(3):e20162896.
Updates in pediatric hospital medicine
Updates in pediatric hospital medicine
The expansion of the field of pediatric hospital medicine in the past 30 years has resulted in improved health care outcomes for hospitalized children1,2 and has been accompanied by a robust increase in the amount of scholarly work related to the field.3 We performed a review of the literature published in 2019 to identify the 10 articles that had the most impact on pediatric hospital medicine, and presented the findings at HM20 Virtual, the 2020 annual conference of the Society of Hospital Medicine. Five of the selected articles are highlighted here.
STUDY 1
Wechsler ME et al. Step-up therapy in black children and adults with poorly controlled asthma. N Engl J Med. 2019 Sep 26;381(13):1227-39.
Background
Current pediatric asthma guidelines suggest adding a long-acting beta-agonist (LABA) to inhaled corticosteroid (ICS) therapy, rather than increasing the ICS dose, for children with poorly controlled asthma. However, these data are based on trials with disproportionately few Black subjects. This study aimed to determine the best step-up therapy for Black patients whose asthma was poorly controlled on ICS monotherapy.
Study overview and results
The authors reported two parallel double-blind, randomized, controlled trials, one in children and one in adolescents and adults. The study of children included 280 subjects ranging in age from 5 to 11, with at least one Black grandparent, and with poorly controlled asthma on low-dose ICS therapy. It used a four-way crossover design in which each subject was treated with four different 14-week treatment regimens: either double (medium-dose) or quintuple (high-dose) their baseline ICS dose, with or without the addition of a LABA. A superior response was defined by the composite outcome of at least one fewer asthma exacerbation, more asthma-control days, or a 5–percentage point difference in predicted FEV1. Forty-six percent of children had improved asthma outcomes when the ICS dose was increased rather than with the addition of a LABA. In contrast, Black adolescents and Black adults had superior responses to the addition of a LABA. There was no significant interaction between the percentage of African ancestry as determined by DNA genotyping and the primary composite outcome. High-dose ICS was associated with a decrease in the ratio of urinary cortisol to creatinine in children younger than 8 years.
Limitations
Approximately 25% of children dropped out of the study, with disproportionately more children dropping out while on a high-dose ICS regimen. Additionally, the difference in the composite outcome was primarily driven by differences in FEV1, with few subjects demonstrating a difference in asthma exacerbations or asthma-control days. Although a decrease in urinary cortisol to creatinine ratio was noted in children under 8 on high-dose ICS, the study period was not long enough to determine the clinical implications of this finding.
Important findings and implications
While studies with a majority of white children have suggested a superior response from adding a LABA compared to increasing the dose of an ICS, almost half of Black children showed a superior response when the dose of an ICS was increased rather than adding a LABA. It is important to note that current guidelines are based on studies with a disproportionate majority of white subjects and may not accurately reflect optimal care for patients in other racial groups. This study underscores the need to include a diverse patient population in research studies.
STUDY 2
Chang PW, Newman TB. A simpler prediction rule for rebound hyperbilirubinemia. Pediatrics. 2019 Jul;144(1):e20183712.
Background
Hyperbilirubinemia (jaundice) is estimated to affect 50%-60% of all newborns. Rebound hyperbilirubinemia – a rise in bilirubin after cessation of phototherapy – is common and can lead to recently discharged infants being readmitted for additional therapy. Lack of clear guidelines regarding when to discharge infants with hyperbilirubinemia has likely contributed to practice variation and some trepidation regarding whether a bilirubin level is “low enough” to discontinue therapy.
Study overview and results
The authors had previously proposed a three-factor hyperbilirubinemia risk model and sought to simplify their rule further.4 They examined a retrospective cohort of 7,048 infants greater than or equal to 35 weeks’ gestation using a random split sample. The authors derived a two-factor model using the same methods and compared its performance to the three-factor model. The two-factor formula was shown to be a good fit as a logistic regression model (Hosmer-Lemeshow test 9.21; P = .33), and the AUROC (area under the receiver operating characteristic) curves for the derivation and validation cohorts were similar between the two-factor (0.877 and 0.876, respectively) and three-factor risk models (0.887 and 0.881, respectively).
Limitations
These data are limited to infants receiving their first treatment of phototherapy and have not been externally validated. An important variable, serum bilirubin at phototherapy termination, was estimated in most subjects, which may have affected the accuracy of the prediction rule. Whether infants received home phototherapy was based only on equipment orders, and some infants may have received phototherapy unbeknownst to investigators. Last, infants with rebound hyperbilirubinemia at less than 72 hours after phototherapy discontinuation may have been missed.
Important findings and implications
This prediction model provides evidence-based, concrete data that can be used in making joint decisions with families regarding discharge timing of infants with hyperbilirubinemia. It also could be beneficial when deciding appropriate follow-up time after discharge.
STUDY 3
Ramgopal S et al. Risk of serious bacterial infection in infants aged ≤60 days presenting to emergency departments with a history of fever only. J Pediatr. 2019 Jan;204:191-195. doi: 10.1016/j.jpeds.2018.08.043.
Background
Febrile infants aged 60 days and younger are at risk for serious bacterial infections (SBI) including urinary tract infections (UTI), bacteremia, and meningitis. As physical exam is a poor discriminator of SBI in this age group, providers frequently rely on laboratory values and risk factors to guide management. Infants presenting with documented fevers by caregivers but found to have no fever in the emergency department are a challenge, and there are limited data regarding SBI frequency in this population.
Study overview and results
The authors performed a secondary analysis of a prospectively gathered cohort of infants aged 60 days and younger within the Pediatric Emergency Care Applied Research Network (PECARN) who had blood, urine, and CSF data available. Notable exclusions included infants who were premature, had a focal infection, were clinically ill, had recent antibiotic use, did not have blood, urine, and CSF data available, or were lost to telephone follow-up at 7 days to ensure wellness. The study cohort included 6,014 infants, 1,233 (32%) who were febrile by history alone. Rates of overall SBI were lower in the afebrile group (8.8% vs. 12.8%). For infants 0-28 days, rates of UTI were lower for the afebrile group (9.5% vs. 14.5%), but there was no difference in the rates of bacteremia or meningitis. For infants 29-60 days, rates of UTI (6.6% vs. 9.3%) and bacteremia (.5% vs. 1.7%) were lower in the afebrile group.
Limitations
Neither the use of home antipyretics nor the method of temperature taking at home were studied. Also, as this was a secondary analysis, it is possible that not all infants who presented with history of fever only were captured, as work-up was dictated by individual treating providers who may have chosen not to work up certain afebrile infants.
Important findings and implications
Nearly one-third of infants presenting for fever evaluation are afebrile on arrival. Although overall rates of SBI were lower in the group with fever by history only, this difference is largely accounted for by differing rates of UTI. Rates of bacteremia and meningitis remained substantial between groups, particularly for infants aged 0-28 days. Because of the significant morbidity associated with these infections, it is reasonable to suggest that absence of fever on presentation alone should not alter clinical or laboratory work-up, particularly in infants 0-28 days.
STUDY 4
Humphrey-Murto S et al. The influence of prior performance information on ratings of current performance and implications for learner handover: A scoping review. Acad Med. 2019 Jul;94(7):1050-7.
Background
Learner Handover (LH) or “forward feeding” occurs when information about trainees is shared between faculty supervisors. Although this can be helpful to tailor educational experiences and build upon previous assessments, it risks stigmatizing trainees and adding bias to future feedback and assessments as the trainee never really has a “clean slate.” In this study, the authors sought to uncover the key concepts of how prior performance information (PPI) influences assessments and any implications for medical education.
Study overview and results
The authors performed a cross-disciplinary scoping review looking at over 17,000 articles published between 1980 and 2017 across the domains of psychology, sports, business, and education. Seven themes were identified with the following notable findings. Raters exposed to positive PPI scored a learner’s performance higher, and vice versa. There was a dose-response relationship with more positive and more negative PPI resulting in higher and lower assessments, respectively. General standards, such as a direction to complete all work in a timely manner, caused an assimilation effect, while specific standards, such as a direction to complete a certain task by a certain day, did not. More motivated and more experienced raters are less affected by PPI, and those who believe that people can change (incremental theorists) are less affected by PPI while those who believe personal attributes are fixed (entity theorists) are more affected.
Limitations
The heterogeneity of the studies and the fact that they were largely conducted in experimental settings may limit generalizability to medical education. Slightly less than half of the studies included a control arm. Last, most of the studies looked at the ratings of only one target performance, not multiple performances over time.
Important findings and implications
Ratings of current performance displace toward PPI direction, with negative PPI more influential than positive PPI. In a formative setting, PPI may help the assessor focus on areas of possible weakness. In contrast, for a summative assessment, PPI may be prejudicial and have an impact on the rating given to the student. Clinicians should be mindful of the information they share with future raters about learners and the potential bias on future assessments that can manifest as a result.
STUDY 5
McCann ME et al. Neurodevelopmental outcome at 5 years of age after general anaesthesia or awake-regional anaesthesia in infancy (GAS): An international, multicentre, randomised, controlled equivalence trial. The Lancet. 2019 Feb;393:664-77.
Background
Animal models and observational studies have suggested a link between early anesthesia exposure and adverse neurocognitive outcomes; however, findings have been mixed and studies are prone to confounding. This study is the first randomized controlled trial to compare neurocognitive outcomes for infants exposed to general anesthesia versus awake-regional anesthesia.
Study overview and results
In this international, multicenter, assessor-masked trial, 722 infants undergoing inguinal hernia repair were randomized to awake-regional anesthesia or single-agent sevoflurane-based general anesthesia. Infants born at greater than 26 weeks’ gestational age were eligible, while those with prior anesthesia exposure or risks for neurocognitive delay were excluded. The primary outcome was full-scale intelligence quotient (FSIQ) testing at 5 years of age on the Wechsler Preschool and Primary Scale of Intelligence, third edition (WPPSI-III). Seven additional neurodevelopmental assessments and parental questionnaires regarding behavior were administered as secondary outcomes. Average anesthesia exposure was 54 minutes and no infant had exposure greater than 120 minutes. There was no significant difference in mean scores on WPPSI-III FSIQ testing, and no difference in the additional neurocognitive assessments or parent-reported outcomes used as secondary outcomes.
Limitations
This study was limited to single, short periods of single-agent anesthesia exposure in children with no additional neurologic risk factors, so caution should be used in extrapolating these data to children with medical complexity and children undergoing multiple procedures, longer surgeries, or multidrug anesthetic regimens. The study population was majority male because of the surgical pathology selected and included only children in the narrow range of postmenstrual age 60 weeks or less. While this population represents a suspected a period of high cerebral vulnerability based on animal models, the implications of anesthesia exposure at other ages are unclear.
Important findings and implications
An estimated 10% of children from developed countries are exposed to general anesthesia during the first 3 years of life. While hospitalists do not typically select the route of anesthesia, they frequently care for patients undergoing procedures and must address parental concerns regarding the safety of anesthesia exposure. Given the rigorous study methods and long-term follow up in the current study, these data should provide reassurance that, for healthy infants undergoing short, single-agent anesthetic exposure, there is no evidence of future adverse neurologic outcomes.
Dr. Russo is director of pediatrics, medical director for quality and innovation, at WellSpan Health, York, Pa. Dr. Money is a pediatric hospitalist at Primary Children’s Hospital, University of Utah School of Medicine, Salt Lake City. Dr. Steed is instructor of hospital medicine, Northwestern Memorial Hospital and Ann and Robert H. Lurie Children’s Hospital of Chicago, Northwestern University School of Medicine, Chicago. The authors would like to thank Dr. Klint M. Schwenk and the Society for Hospital Medicine Pediatric Special Interest Group Executive Council.
References
1. Roberts KB, Fisher ER, and Rauch DA. The history of pediatric hospital medicine in the United States, 1996-2019. J Hosp Med. 2020 Jul;15(7):424-7.
2. Mussman GM and Conway PH. Pediatric hospitalist systems versus traditional models of care: Effect on quality and cost outcomes. J Hosp Med. 2012 Apr;7(4):350-7.
3. Wang ME, Shaughnessy EE, and Leyenaar JK. The future of pediatric hospital medicine: Challenges and opportunities. J Hosp Med. 2020 Jul;15(7):428-30.
4. Chang PW et al. A clinical prediction rule for rebound hyperbilirubinemia following inpatient phototherapy. Pediatrics. 2017;139 Mar;139(3):e20162896.
The expansion of the field of pediatric hospital medicine in the past 30 years has resulted in improved health care outcomes for hospitalized children1,2 and has been accompanied by a robust increase in the amount of scholarly work related to the field.3 We performed a review of the literature published in 2019 to identify the 10 articles that had the most impact on pediatric hospital medicine, and presented the findings at HM20 Virtual, the 2020 annual conference of the Society of Hospital Medicine. Five of the selected articles are highlighted here.
STUDY 1
Wechsler ME et al. Step-up therapy in black children and adults with poorly controlled asthma. N Engl J Med. 2019 Sep 26;381(13):1227-39.
Background
Current pediatric asthma guidelines suggest adding a long-acting beta-agonist (LABA) to inhaled corticosteroid (ICS) therapy, rather than increasing the ICS dose, for children with poorly controlled asthma. However, these data are based on trials with disproportionately few Black subjects. This study aimed to determine the best step-up therapy for Black patients whose asthma was poorly controlled on ICS monotherapy.
Study overview and results
The authors reported two parallel double-blind, randomized, controlled trials, one in children and one in adolescents and adults. The study of children included 280 subjects ranging in age from 5 to 11, with at least one Black grandparent, and with poorly controlled asthma on low-dose ICS therapy. It used a four-way crossover design in which each subject was treated with four different 14-week treatment regimens: either double (medium-dose) or quintuple (high-dose) their baseline ICS dose, with or without the addition of a LABA. A superior response was defined by the composite outcome of at least one fewer asthma exacerbation, more asthma-control days, or a 5–percentage point difference in predicted FEV1. Forty-six percent of children had improved asthma outcomes when the ICS dose was increased rather than with the addition of a LABA. In contrast, Black adolescents and Black adults had superior responses to the addition of a LABA. There was no significant interaction between the percentage of African ancestry as determined by DNA genotyping and the primary composite outcome. High-dose ICS was associated with a decrease in the ratio of urinary cortisol to creatinine in children younger than 8 years.
Limitations
Approximately 25% of children dropped out of the study, with disproportionately more children dropping out while on a high-dose ICS regimen. Additionally, the difference in the composite outcome was primarily driven by differences in FEV1, with few subjects demonstrating a difference in asthma exacerbations or asthma-control days. Although a decrease in urinary cortisol to creatinine ratio was noted in children under 8 on high-dose ICS, the study period was not long enough to determine the clinical implications of this finding.
Important findings and implications
While studies with a majority of white children have suggested a superior response from adding a LABA compared to increasing the dose of an ICS, almost half of Black children showed a superior response when the dose of an ICS was increased rather than adding a LABA. It is important to note that current guidelines are based on studies with a disproportionate majority of white subjects and may not accurately reflect optimal care for patients in other racial groups. This study underscores the need to include a diverse patient population in research studies.
STUDY 2
Chang PW, Newman TB. A simpler prediction rule for rebound hyperbilirubinemia. Pediatrics. 2019 Jul;144(1):e20183712.
Background
Hyperbilirubinemia (jaundice) is estimated to affect 50%-60% of all newborns. Rebound hyperbilirubinemia – a rise in bilirubin after cessation of phototherapy – is common and can lead to recently discharged infants being readmitted for additional therapy. Lack of clear guidelines regarding when to discharge infants with hyperbilirubinemia has likely contributed to practice variation and some trepidation regarding whether a bilirubin level is “low enough” to discontinue therapy.
Study overview and results
The authors had previously proposed a three-factor hyperbilirubinemia risk model and sought to simplify their rule further.4 They examined a retrospective cohort of 7,048 infants greater than or equal to 35 weeks’ gestation using a random split sample. The authors derived a two-factor model using the same methods and compared its performance to the three-factor model. The two-factor formula was shown to be a good fit as a logistic regression model (Hosmer-Lemeshow test 9.21; P = .33), and the AUROC (area under the receiver operating characteristic) curves for the derivation and validation cohorts were similar between the two-factor (0.877 and 0.876, respectively) and three-factor risk models (0.887 and 0.881, respectively).
Limitations
These data are limited to infants receiving their first treatment of phototherapy and have not been externally validated. An important variable, serum bilirubin at phototherapy termination, was estimated in most subjects, which may have affected the accuracy of the prediction rule. Whether infants received home phototherapy was based only on equipment orders, and some infants may have received phototherapy unbeknownst to investigators. Last, infants with rebound hyperbilirubinemia at less than 72 hours after phototherapy discontinuation may have been missed.
Important findings and implications
This prediction model provides evidence-based, concrete data that can be used in making joint decisions with families regarding discharge timing of infants with hyperbilirubinemia. It also could be beneficial when deciding appropriate follow-up time after discharge.
STUDY 3
Ramgopal S et al. Risk of serious bacterial infection in infants aged ≤60 days presenting to emergency departments with a history of fever only. J Pediatr. 2019 Jan;204:191-195. doi: 10.1016/j.jpeds.2018.08.043.
Background
Febrile infants aged 60 days and younger are at risk for serious bacterial infections (SBI) including urinary tract infections (UTI), bacteremia, and meningitis. As physical exam is a poor discriminator of SBI in this age group, providers frequently rely on laboratory values and risk factors to guide management. Infants presenting with documented fevers by caregivers but found to have no fever in the emergency department are a challenge, and there are limited data regarding SBI frequency in this population.
Study overview and results
The authors performed a secondary analysis of a prospectively gathered cohort of infants aged 60 days and younger within the Pediatric Emergency Care Applied Research Network (PECARN) who had blood, urine, and CSF data available. Notable exclusions included infants who were premature, had a focal infection, were clinically ill, had recent antibiotic use, did not have blood, urine, and CSF data available, or were lost to telephone follow-up at 7 days to ensure wellness. The study cohort included 6,014 infants, 1,233 (32%) who were febrile by history alone. Rates of overall SBI were lower in the afebrile group (8.8% vs. 12.8%). For infants 0-28 days, rates of UTI were lower for the afebrile group (9.5% vs. 14.5%), but there was no difference in the rates of bacteremia or meningitis. For infants 29-60 days, rates of UTI (6.6% vs. 9.3%) and bacteremia (.5% vs. 1.7%) were lower in the afebrile group.
Limitations
Neither the use of home antipyretics nor the method of temperature taking at home were studied. Also, as this was a secondary analysis, it is possible that not all infants who presented with history of fever only were captured, as work-up was dictated by individual treating providers who may have chosen not to work up certain afebrile infants.
Important findings and implications
Nearly one-third of infants presenting for fever evaluation are afebrile on arrival. Although overall rates of SBI were lower in the group with fever by history only, this difference is largely accounted for by differing rates of UTI. Rates of bacteremia and meningitis remained substantial between groups, particularly for infants aged 0-28 days. Because of the significant morbidity associated with these infections, it is reasonable to suggest that absence of fever on presentation alone should not alter clinical or laboratory work-up, particularly in infants 0-28 days.
STUDY 4
Humphrey-Murto S et al. The influence of prior performance information on ratings of current performance and implications for learner handover: A scoping review. Acad Med. 2019 Jul;94(7):1050-7.
Background
Learner Handover (LH) or “forward feeding” occurs when information about trainees is shared between faculty supervisors. Although this can be helpful to tailor educational experiences and build upon previous assessments, it risks stigmatizing trainees and adding bias to future feedback and assessments as the trainee never really has a “clean slate.” In this study, the authors sought to uncover the key concepts of how prior performance information (PPI) influences assessments and any implications for medical education.
Study overview and results
The authors performed a cross-disciplinary scoping review looking at over 17,000 articles published between 1980 and 2017 across the domains of psychology, sports, business, and education. Seven themes were identified with the following notable findings. Raters exposed to positive PPI scored a learner’s performance higher, and vice versa. There was a dose-response relationship with more positive and more negative PPI resulting in higher and lower assessments, respectively. General standards, such as a direction to complete all work in a timely manner, caused an assimilation effect, while specific standards, such as a direction to complete a certain task by a certain day, did not. More motivated and more experienced raters are less affected by PPI, and those who believe that people can change (incremental theorists) are less affected by PPI while those who believe personal attributes are fixed (entity theorists) are more affected.
Limitations
The heterogeneity of the studies and the fact that they were largely conducted in experimental settings may limit generalizability to medical education. Slightly less than half of the studies included a control arm. Last, most of the studies looked at the ratings of only one target performance, not multiple performances over time.
Important findings and implications
Ratings of current performance displace toward PPI direction, with negative PPI more influential than positive PPI. In a formative setting, PPI may help the assessor focus on areas of possible weakness. In contrast, for a summative assessment, PPI may be prejudicial and have an impact on the rating given to the student. Clinicians should be mindful of the information they share with future raters about learners and the potential bias on future assessments that can manifest as a result.
STUDY 5
McCann ME et al. Neurodevelopmental outcome at 5 years of age after general anaesthesia or awake-regional anaesthesia in infancy (GAS): An international, multicentre, randomised, controlled equivalence trial. The Lancet. 2019 Feb;393:664-77.
Background
Animal models and observational studies have suggested a link between early anesthesia exposure and adverse neurocognitive outcomes; however, findings have been mixed and studies are prone to confounding. This study is the first randomized controlled trial to compare neurocognitive outcomes for infants exposed to general anesthesia versus awake-regional anesthesia.
Study overview and results
In this international, multicenter, assessor-masked trial, 722 infants undergoing inguinal hernia repair were randomized to awake-regional anesthesia or single-agent sevoflurane-based general anesthesia. Infants born at greater than 26 weeks’ gestational age were eligible, while those with prior anesthesia exposure or risks for neurocognitive delay were excluded. The primary outcome was full-scale intelligence quotient (FSIQ) testing at 5 years of age on the Wechsler Preschool and Primary Scale of Intelligence, third edition (WPPSI-III). Seven additional neurodevelopmental assessments and parental questionnaires regarding behavior were administered as secondary outcomes. Average anesthesia exposure was 54 minutes and no infant had exposure greater than 120 minutes. There was no significant difference in mean scores on WPPSI-III FSIQ testing, and no difference in the additional neurocognitive assessments or parent-reported outcomes used as secondary outcomes.
Limitations
This study was limited to single, short periods of single-agent anesthesia exposure in children with no additional neurologic risk factors, so caution should be used in extrapolating these data to children with medical complexity and children undergoing multiple procedures, longer surgeries, or multidrug anesthetic regimens. The study population was majority male because of the surgical pathology selected and included only children in the narrow range of postmenstrual age 60 weeks or less. While this population represents a suspected a period of high cerebral vulnerability based on animal models, the implications of anesthesia exposure at other ages are unclear.
Important findings and implications
An estimated 10% of children from developed countries are exposed to general anesthesia during the first 3 years of life. While hospitalists do not typically select the route of anesthesia, they frequently care for patients undergoing procedures and must address parental concerns regarding the safety of anesthesia exposure. Given the rigorous study methods and long-term follow up in the current study, these data should provide reassurance that, for healthy infants undergoing short, single-agent anesthetic exposure, there is no evidence of future adverse neurologic outcomes.
Dr. Russo is director of pediatrics, medical director for quality and innovation, at WellSpan Health, York, Pa. Dr. Money is a pediatric hospitalist at Primary Children’s Hospital, University of Utah School of Medicine, Salt Lake City. Dr. Steed is instructor of hospital medicine, Northwestern Memorial Hospital and Ann and Robert H. Lurie Children’s Hospital of Chicago, Northwestern University School of Medicine, Chicago. The authors would like to thank Dr. Klint M. Schwenk and the Society for Hospital Medicine Pediatric Special Interest Group Executive Council.
References
1. Roberts KB, Fisher ER, and Rauch DA. The history of pediatric hospital medicine in the United States, 1996-2019. J Hosp Med. 2020 Jul;15(7):424-7.
2. Mussman GM and Conway PH. Pediatric hospitalist systems versus traditional models of care: Effect on quality and cost outcomes. J Hosp Med. 2012 Apr;7(4):350-7.
3. Wang ME, Shaughnessy EE, and Leyenaar JK. The future of pediatric hospital medicine: Challenges and opportunities. J Hosp Med. 2020 Jul;15(7):428-30.
4. Chang PW et al. A clinical prediction rule for rebound hyperbilirubinemia following inpatient phototherapy. Pediatrics. 2017;139 Mar;139(3):e20162896.
High blood pressure at any age speeds cognitive decline
, new research shows. In a retrospective study of more than 15,000 participants, hypertension during middle age was associated with memory decline, and onset at later ages was linked to worsening memory and global cognition.
The investigators found that prehypertension, defined as systolic pressure of 120-139 mm Hg or diastolic pressure of 80-89 mm Hg, was also linked to accelerated cognitive decline.
Although duration of hypertension was not associated with any marker of cognitive decline, blood pressure control “can substantially reduce hypertension’s deleterious effect on the pace of cognitive decline,” said study investigator Sandhi M. Barreto, MD, PhD, professor of medicine at Universidade Federal de Minas Gerais, Belo Horizonte, Brazil.
The findings were published online Dec. 14 in Hypertension.
Unanswered questions
Hypertension is an established and highly prevalent risk factor for cognitive decline, but the age at which it begins to affect cognition is unclear. Previous research suggests that onset during middle age is associated with more harmful cognitive effects than onset in later life. One reason for this apparent difference may be that the duration of hypertension influences the magnitude of cognitive decline, the researchers noted.
Other studies have shown that prehypertension is associated with damage to certain organs, but its effects on cognition are uncertain. In addition, the effect of good blood pressure control with antihypertensive medications and the impact on cognition are also unclear.
To investigate, the researchers examined data from the ongoing, multicenter ELSA-Brasil study. ELSA-Brasil follows 15,105 civil servants between the ages of 35 and 74 years. Dr. Barreto and team assessed data from visit 1, which was conducted between 2008 and 2010, and visit 2, which was conducted between 2012 and 2014.
At each visit, participants underwent a memory test, a verbal fluency test, and the Trail Making Test Part B. The investigators calculated Z scores for these tests to derive a global cognitive score.
Blood pressure was measured on the right arm, and hypertension status, age at the time of hypertension diagnosis, duration of hypertension diagnosis, hypertension treatment, and control status were recorded. Other covariables included sex, education, race, smoking status, physical activity, body mass index, and total cholesterol level.
The researchers excluded patients who did not undergo cognitive testing at visit 2, those who had a history of stroke at baseline, and those who initiated antihypertensive medications despite having normotension. After exclusions, the analysis included 7,063 participants (approximately 55% were women, 15% were Black).
At visit 1, the mean age of the group was 58.9 years, and 53.4% of participants had 14 or more years of education. In addition, 22% had prehypertension, and 46.8% had hypertension. The median duration of hypertension was 7 years; 29.8% of participants with hypertension were diagnosed with the condition during middle age.
Of those who reported having hypertension at visit 1, 7.3% were not taking any antihypertensive medication. Among participants with hypertension who were taking antihypertensives, 31.2% had uncontrolled blood pressure.
Independent predictor
Results showed that prehypertension independently predicted a significantly greater decline in verbal fluency (Z score, –0.0095; P < .01) and global cognitive score (Z score, –0.0049; P < .05) compared with normal blood pressure.
At middle age, hypertension was associated with a steeper decline in memory (Z score, –0.0072; P < .05) compared with normal blood pressure. At older ages, hypertension was linked to a steeper decline in both memory (Z score, –0.0151; P < .001) and global cognitive score (Z score, –0.0080; P < .01). Duration of hypertension, however, did not significantly predict changes in cognition (P < .109).
Among those with hypertension who were taking antihypertensive medications, those with uncontrolled blood pressure experienced greater declines in rapid memory (Z score, –0.0126; P < .01) and global cognitive score (Z score, –0.0074; P < .01) than did those with controlled blood pressure.
The investigators noted that the study participants had a comparatively high level of education, which has been shown to “boost cognitive reserve and lessen the speed of age-related cognitive decline,” Dr. Barreto said. However, “our results indicate that the effect of hypertension on cognitive decline affects individuals of all educational levels similarly,” she said.
Dr. Barreto noted that the findings have two major clinical implications. First, “maintaining blood pressure below prehypertension levels is important to preserve cognitive function or delay cognitive decline,” she said. Secondly, “in hypertensive individuals, keeping blood pressure under control is essential to reduce the speed of cognitive decline.”
The researchers plan to conduct further analyses of the data to clarify the observed relationship between memory and verbal fluency. They also plan to examine how hypertension affects long-term executive function.
‘Continuum of risk’
Commenting on the study, Philip B. Gorelick, MD, MPH, adjunct professor of neurology (stroke and neurocritical care) at Northwestern University, Chicago, noted that, so far, research suggests that the risk for stroke associated with blood pressure levels should be understood as representing a continuum rather than as being associated with several discrete points.
“The same may hold true for cognitive decline and dementia. There may be a continuum of risk whereby persons even at so-called elevated but relatively lower levels of blood pressure based on a continuous scale are at risk,” said Dr. Gorelick, who was not involved with the current study.
The investigators relied on a large and well-studied population of civil servants. However, the population’s relative youth and high level of education may limit the generalizability of the findings, he noted. In addition, the follow-up time was relatively short.
“The hard endpoint of dementia was not studied but would be of interest to enhance our understanding of the influence of blood pressure elevation on cognitive decline or dementia during a longer follow-up of the cohort,” Dr. Gorelick said.
The findings also suggest the need to better understand mechanisms that link blood pressure elevation with cognitive decline, he added.
They indicate “the need for additional clinical trials to better elucidate blood pressure lowering targets for cognitive preservation in different groups of persons at risk,” such as those with normal cognition, those with mild cognitive impairment, and those with dementia, said Dr. Gorelick. “For example, is it safe and efficacious to lower blood pressure in persons with more advanced cognitive impairment or dementia?” he asked.
The study was funded by the Brazilian Coordination for the Improvement of Higher Education Personnel. Dr. Barreto has received support from the Research Agency of the State of Minas Gerais. Although Dr. Gorelick was not involved in the ELSA-Brasil cohort study, he serves on a data monitoring committee for a trial of a blood pressure–lowering agent in the preservation of cognition.
A version of this article first appeared on Medscape.com.
, new research shows. In a retrospective study of more than 15,000 participants, hypertension during middle age was associated with memory decline, and onset at later ages was linked to worsening memory and global cognition.
The investigators found that prehypertension, defined as systolic pressure of 120-139 mm Hg or diastolic pressure of 80-89 mm Hg, was also linked to accelerated cognitive decline.
Although duration of hypertension was not associated with any marker of cognitive decline, blood pressure control “can substantially reduce hypertension’s deleterious effect on the pace of cognitive decline,” said study investigator Sandhi M. Barreto, MD, PhD, professor of medicine at Universidade Federal de Minas Gerais, Belo Horizonte, Brazil.
The findings were published online Dec. 14 in Hypertension.
Unanswered questions
Hypertension is an established and highly prevalent risk factor for cognitive decline, but the age at which it begins to affect cognition is unclear. Previous research suggests that onset during middle age is associated with more harmful cognitive effects than onset in later life. One reason for this apparent difference may be that the duration of hypertension influences the magnitude of cognitive decline, the researchers noted.
Other studies have shown that prehypertension is associated with damage to certain organs, but its effects on cognition are uncertain. In addition, the effect of good blood pressure control with antihypertensive medications and the impact on cognition are also unclear.
To investigate, the researchers examined data from the ongoing, multicenter ELSA-Brasil study. ELSA-Brasil follows 15,105 civil servants between the ages of 35 and 74 years. Dr. Barreto and team assessed data from visit 1, which was conducted between 2008 and 2010, and visit 2, which was conducted between 2012 and 2014.
At each visit, participants underwent a memory test, a verbal fluency test, and the Trail Making Test Part B. The investigators calculated Z scores for these tests to derive a global cognitive score.
Blood pressure was measured on the right arm, and hypertension status, age at the time of hypertension diagnosis, duration of hypertension diagnosis, hypertension treatment, and control status were recorded. Other covariables included sex, education, race, smoking status, physical activity, body mass index, and total cholesterol level.
The researchers excluded patients who did not undergo cognitive testing at visit 2, those who had a history of stroke at baseline, and those who initiated antihypertensive medications despite having normotension. After exclusions, the analysis included 7,063 participants (approximately 55% were women, 15% were Black).
At visit 1, the mean age of the group was 58.9 years, and 53.4% of participants had 14 or more years of education. In addition, 22% had prehypertension, and 46.8% had hypertension. The median duration of hypertension was 7 years; 29.8% of participants with hypertension were diagnosed with the condition during middle age.
Of those who reported having hypertension at visit 1, 7.3% were not taking any antihypertensive medication. Among participants with hypertension who were taking antihypertensives, 31.2% had uncontrolled blood pressure.
Independent predictor
Results showed that prehypertension independently predicted a significantly greater decline in verbal fluency (Z score, –0.0095; P < .01) and global cognitive score (Z score, –0.0049; P < .05) compared with normal blood pressure.
At middle age, hypertension was associated with a steeper decline in memory (Z score, –0.0072; P < .05) compared with normal blood pressure. At older ages, hypertension was linked to a steeper decline in both memory (Z score, –0.0151; P < .001) and global cognitive score (Z score, –0.0080; P < .01). Duration of hypertension, however, did not significantly predict changes in cognition (P < .109).
Among those with hypertension who were taking antihypertensive medications, those with uncontrolled blood pressure experienced greater declines in rapid memory (Z score, –0.0126; P < .01) and global cognitive score (Z score, –0.0074; P < .01) than did those with controlled blood pressure.
The investigators noted that the study participants had a comparatively high level of education, which has been shown to “boost cognitive reserve and lessen the speed of age-related cognitive decline,” Dr. Barreto said. However, “our results indicate that the effect of hypertension on cognitive decline affects individuals of all educational levels similarly,” she said.
Dr. Barreto noted that the findings have two major clinical implications. First, “maintaining blood pressure below prehypertension levels is important to preserve cognitive function or delay cognitive decline,” she said. Secondly, “in hypertensive individuals, keeping blood pressure under control is essential to reduce the speed of cognitive decline.”
The researchers plan to conduct further analyses of the data to clarify the observed relationship between memory and verbal fluency. They also plan to examine how hypertension affects long-term executive function.
‘Continuum of risk’
Commenting on the study, Philip B. Gorelick, MD, MPH, adjunct professor of neurology (stroke and neurocritical care) at Northwestern University, Chicago, noted that, so far, research suggests that the risk for stroke associated with blood pressure levels should be understood as representing a continuum rather than as being associated with several discrete points.
“The same may hold true for cognitive decline and dementia. There may be a continuum of risk whereby persons even at so-called elevated but relatively lower levels of blood pressure based on a continuous scale are at risk,” said Dr. Gorelick, who was not involved with the current study.
The investigators relied on a large and well-studied population of civil servants. However, the population’s relative youth and high level of education may limit the generalizability of the findings, he noted. In addition, the follow-up time was relatively short.
“The hard endpoint of dementia was not studied but would be of interest to enhance our understanding of the influence of blood pressure elevation on cognitive decline or dementia during a longer follow-up of the cohort,” Dr. Gorelick said.
The findings also suggest the need to better understand mechanisms that link blood pressure elevation with cognitive decline, he added.
They indicate “the need for additional clinical trials to better elucidate blood pressure lowering targets for cognitive preservation in different groups of persons at risk,” such as those with normal cognition, those with mild cognitive impairment, and those with dementia, said Dr. Gorelick. “For example, is it safe and efficacious to lower blood pressure in persons with more advanced cognitive impairment or dementia?” he asked.
The study was funded by the Brazilian Coordination for the Improvement of Higher Education Personnel. Dr. Barreto has received support from the Research Agency of the State of Minas Gerais. Although Dr. Gorelick was not involved in the ELSA-Brasil cohort study, he serves on a data monitoring committee for a trial of a blood pressure–lowering agent in the preservation of cognition.
A version of this article first appeared on Medscape.com.
, new research shows. In a retrospective study of more than 15,000 participants, hypertension during middle age was associated with memory decline, and onset at later ages was linked to worsening memory and global cognition.
The investigators found that prehypertension, defined as systolic pressure of 120-139 mm Hg or diastolic pressure of 80-89 mm Hg, was also linked to accelerated cognitive decline.
Although duration of hypertension was not associated with any marker of cognitive decline, blood pressure control “can substantially reduce hypertension’s deleterious effect on the pace of cognitive decline,” said study investigator Sandhi M. Barreto, MD, PhD, professor of medicine at Universidade Federal de Minas Gerais, Belo Horizonte, Brazil.
The findings were published online Dec. 14 in Hypertension.
Unanswered questions
Hypertension is an established and highly prevalent risk factor for cognitive decline, but the age at which it begins to affect cognition is unclear. Previous research suggests that onset during middle age is associated with more harmful cognitive effects than onset in later life. One reason for this apparent difference may be that the duration of hypertension influences the magnitude of cognitive decline, the researchers noted.
Other studies have shown that prehypertension is associated with damage to certain organs, but its effects on cognition are uncertain. In addition, the effect of good blood pressure control with antihypertensive medications and the impact on cognition are also unclear.
To investigate, the researchers examined data from the ongoing, multicenter ELSA-Brasil study. ELSA-Brasil follows 15,105 civil servants between the ages of 35 and 74 years. Dr. Barreto and team assessed data from visit 1, which was conducted between 2008 and 2010, and visit 2, which was conducted between 2012 and 2014.
At each visit, participants underwent a memory test, a verbal fluency test, and the Trail Making Test Part B. The investigators calculated Z scores for these tests to derive a global cognitive score.
Blood pressure was measured on the right arm, and hypertension status, age at the time of hypertension diagnosis, duration of hypertension diagnosis, hypertension treatment, and control status were recorded. Other covariables included sex, education, race, smoking status, physical activity, body mass index, and total cholesterol level.
The researchers excluded patients who did not undergo cognitive testing at visit 2, those who had a history of stroke at baseline, and those who initiated antihypertensive medications despite having normotension. After exclusions, the analysis included 7,063 participants (approximately 55% were women, 15% were Black).
At visit 1, the mean age of the group was 58.9 years, and 53.4% of participants had 14 or more years of education. In addition, 22% had prehypertension, and 46.8% had hypertension. The median duration of hypertension was 7 years; 29.8% of participants with hypertension were diagnosed with the condition during middle age.
Of those who reported having hypertension at visit 1, 7.3% were not taking any antihypertensive medication. Among participants with hypertension who were taking antihypertensives, 31.2% had uncontrolled blood pressure.
Independent predictor
Results showed that prehypertension independently predicted a significantly greater decline in verbal fluency (Z score, –0.0095; P < .01) and global cognitive score (Z score, –0.0049; P < .05) compared with normal blood pressure.
At middle age, hypertension was associated with a steeper decline in memory (Z score, –0.0072; P < .05) compared with normal blood pressure. At older ages, hypertension was linked to a steeper decline in both memory (Z score, –0.0151; P < .001) and global cognitive score (Z score, –0.0080; P < .01). Duration of hypertension, however, did not significantly predict changes in cognition (P < .109).
Among those with hypertension who were taking antihypertensive medications, those with uncontrolled blood pressure experienced greater declines in rapid memory (Z score, –0.0126; P < .01) and global cognitive score (Z score, –0.0074; P < .01) than did those with controlled blood pressure.
The investigators noted that the study participants had a comparatively high level of education, which has been shown to “boost cognitive reserve and lessen the speed of age-related cognitive decline,” Dr. Barreto said. However, “our results indicate that the effect of hypertension on cognitive decline affects individuals of all educational levels similarly,” she said.
Dr. Barreto noted that the findings have two major clinical implications. First, “maintaining blood pressure below prehypertension levels is important to preserve cognitive function or delay cognitive decline,” she said. Secondly, “in hypertensive individuals, keeping blood pressure under control is essential to reduce the speed of cognitive decline.”
The researchers plan to conduct further analyses of the data to clarify the observed relationship between memory and verbal fluency. They also plan to examine how hypertension affects long-term executive function.
‘Continuum of risk’
Commenting on the study, Philip B. Gorelick, MD, MPH, adjunct professor of neurology (stroke and neurocritical care) at Northwestern University, Chicago, noted that, so far, research suggests that the risk for stroke associated with blood pressure levels should be understood as representing a continuum rather than as being associated with several discrete points.
“The same may hold true for cognitive decline and dementia. There may be a continuum of risk whereby persons even at so-called elevated but relatively lower levels of blood pressure based on a continuous scale are at risk,” said Dr. Gorelick, who was not involved with the current study.
The investigators relied on a large and well-studied population of civil servants. However, the population’s relative youth and high level of education may limit the generalizability of the findings, he noted. In addition, the follow-up time was relatively short.
“The hard endpoint of dementia was not studied but would be of interest to enhance our understanding of the influence of blood pressure elevation on cognitive decline or dementia during a longer follow-up of the cohort,” Dr. Gorelick said.
The findings also suggest the need to better understand mechanisms that link blood pressure elevation with cognitive decline, he added.
They indicate “the need for additional clinical trials to better elucidate blood pressure lowering targets for cognitive preservation in different groups of persons at risk,” such as those with normal cognition, those with mild cognitive impairment, and those with dementia, said Dr. Gorelick. “For example, is it safe and efficacious to lower blood pressure in persons with more advanced cognitive impairment or dementia?” he asked.
The study was funded by the Brazilian Coordination for the Improvement of Higher Education Personnel. Dr. Barreto has received support from the Research Agency of the State of Minas Gerais. Although Dr. Gorelick was not involved in the ELSA-Brasil cohort study, he serves on a data monitoring committee for a trial of a blood pressure–lowering agent in the preservation of cognition.
A version of this article first appeared on Medscape.com.
FROM HYPERTENSION
Urgent recall for Penumbra JET 7 Xtra Flex reperfusion catheters
“All users should stop using this device, and facilities should remove these devices from inventory,” the recall notice, posted on the U.S. Food and Drug Administration website, advises.
The recall covers the JET 7 Xtra Flex catheter, which was cleared for use in June 2019, and the JET 7MAX configuration (which includes the JET 7 Xtra Flex catheter and MAX delivery device), which was cleared in February of this year.
The recall does not apply to the Penumbra JET 7 reperfusion catheter with standard tip.
The FDA says it has received over 200 medical device reports (MDRs) associated with the JET 7 Xtra Flex catheter, including reports of deaths, serious injuries, and malfunctions.
Twenty of these MDRs describe 14 unique patient deaths. Other MDRs describe serious patient injury, such as vessel damage, hemorrhage, and cerebral infarction.
Device malfunctions described in the reports include ballooning, expansion, rupture, breakage or complete separation, and exposure of internal support coils near the distal tip region of the JET 7 Xtra Flex catheter.
According to the FDA, bench testing by the manufacturer, in which the catheter distal tip is plugged and pressurized to failure, indicates that the JET 7 Xtra Flex catheter is not able to withstand the same burst pressures to failure as the manufacturer’s other large-bore aspiration catheters used to remove thrombus for patients with acute ischemic stroke.
Penumbra’s urgent medical device recall letter advises health care providers and facilities to remove and quarantine all unused devices covered by this recall, to complete the product identification and return form, and to return all products to Penumbra in accordance with instructions provided.
For questions regarding this recall, contact Penumbra customer service by phone at 888-272-4606 or by email at notification@penumbrainc.com.
A version of this article first appeared on Medscape.com.
“All users should stop using this device, and facilities should remove these devices from inventory,” the recall notice, posted on the U.S. Food and Drug Administration website, advises.
The recall covers the JET 7 Xtra Flex catheter, which was cleared for use in June 2019, and the JET 7MAX configuration (which includes the JET 7 Xtra Flex catheter and MAX delivery device), which was cleared in February of this year.
The recall does not apply to the Penumbra JET 7 reperfusion catheter with standard tip.
The FDA says it has received over 200 medical device reports (MDRs) associated with the JET 7 Xtra Flex catheter, including reports of deaths, serious injuries, and malfunctions.
Twenty of these MDRs describe 14 unique patient deaths. Other MDRs describe serious patient injury, such as vessel damage, hemorrhage, and cerebral infarction.
Device malfunctions described in the reports include ballooning, expansion, rupture, breakage or complete separation, and exposure of internal support coils near the distal tip region of the JET 7 Xtra Flex catheter.
According to the FDA, bench testing by the manufacturer, in which the catheter distal tip is plugged and pressurized to failure, indicates that the JET 7 Xtra Flex catheter is not able to withstand the same burst pressures to failure as the manufacturer’s other large-bore aspiration catheters used to remove thrombus for patients with acute ischemic stroke.
Penumbra’s urgent medical device recall letter advises health care providers and facilities to remove and quarantine all unused devices covered by this recall, to complete the product identification and return form, and to return all products to Penumbra in accordance with instructions provided.
For questions regarding this recall, contact Penumbra customer service by phone at 888-272-4606 or by email at notification@penumbrainc.com.
A version of this article first appeared on Medscape.com.
“All users should stop using this device, and facilities should remove these devices from inventory,” the recall notice, posted on the U.S. Food and Drug Administration website, advises.
The recall covers the JET 7 Xtra Flex catheter, which was cleared for use in June 2019, and the JET 7MAX configuration (which includes the JET 7 Xtra Flex catheter and MAX delivery device), which was cleared in February of this year.
The recall does not apply to the Penumbra JET 7 reperfusion catheter with standard tip.
The FDA says it has received over 200 medical device reports (MDRs) associated with the JET 7 Xtra Flex catheter, including reports of deaths, serious injuries, and malfunctions.
Twenty of these MDRs describe 14 unique patient deaths. Other MDRs describe serious patient injury, such as vessel damage, hemorrhage, and cerebral infarction.
Device malfunctions described in the reports include ballooning, expansion, rupture, breakage or complete separation, and exposure of internal support coils near the distal tip region of the JET 7 Xtra Flex catheter.
According to the FDA, bench testing by the manufacturer, in which the catheter distal tip is plugged and pressurized to failure, indicates that the JET 7 Xtra Flex catheter is not able to withstand the same burst pressures to failure as the manufacturer’s other large-bore aspiration catheters used to remove thrombus for patients with acute ischemic stroke.
Penumbra’s urgent medical device recall letter advises health care providers and facilities to remove and quarantine all unused devices covered by this recall, to complete the product identification and return form, and to return all products to Penumbra in accordance with instructions provided.
For questions regarding this recall, contact Penumbra customer service by phone at 888-272-4606 or by email at notification@penumbrainc.com.
A version of this article first appeared on Medscape.com.
COVID-19 neurologic fallout not limited to the severely ill
Serious neurologic complications in patients with COVID-19 are not limited to the severely ill, new research confirms.
“We found a range of neurologic diagnoses, including stroke and seizures, among hospitalized patients with COVID-19 and the majority were not critically ill, suggesting that these complications are not limited just to those patients who require ICU care or a ventilator,” study investigator Pria Anand, MD, division of neuro-infectious diseases, Boston University, said in an interview.
The study was published online Dec. 9 in Neurology Clinical Practice.
‘Moderately severe’ disability
For the study, the investigators reviewed the medical records of 74 adults (mean age, 64 years) who were hospitalized with COVID-19 and evaluated for neurologic conditions at Boston Medical Center, a safety-net hospital caring primarily for underserved, low-income, racial and ethnic minority populations.
The most common COVID-19 symptoms on arrival to the hospital were cough (39%), dyspnea (36%), and fever (34%). Eleven patients required intubation (15%) and 28 required some form of supplemental oxygen (38%). Thirty-four patients required intensive care (46%).
The most common neurologic COVID-19 symptoms at presentation were altered mental status (53%), myalgia (24%), fatigue (24%), and headache (18%).
After neurologic assessment, the most common final neurologic diagnosis was multifactorial or toxic-metabolic encephalopathy (35%), followed by seizure (20%), ischemic stroke (20%), primary movement disorder (9%), peripheral neuropathy (8%), and hemorrhagic stroke (4%).
Three patients (4%) suffered traumatic brain injuries after falling in their homes after developing COVID-19.
Ten (14%) patients died in the hospital. Survivors had “moderately severe” disability at discharge (median modified Rankin Scale score of 4 from a preadmission mRS score of 2) and many were discharged to nursing facilities or rehabilitation hospitals.
“Although we do not have data on their posthospital course, this suggests that patients with neurologic complications of COVID-19 are likely to require ongoing rehabilitation, even after they leave the hospital,” Dr. Anand, a member of the American Academy of Neurology, said in an interview.
“There are a diverse range of mechanisms by which COVID-19 can cause neurologic complications,” Dr. Anand said.
“These complications can result from the body’s immunological response to the virus (e.g., Guillain-Barré syndrome, an autoimmune disorder affecting the nerves), from having a systemic severe illness (e.g., brain injury as a result of insufficient oxygenation), from the increased tendency to form blood clots (e.g., stroke), from worsening of preexisting neurologic disorders, and possibly from involvement of the nervous system by the virus itself,” she explained.
The researchers said more study is needed to characterize the infectious and postinfectious neurologic complications of COVID-19 in diverse patient populations.
Lingering issues
In an interview, Kenneth L. Tyler, MD, chair of neurology, University of Colorado, Denver, noted that this is one of the larger series published to date of the neurologic complications associated with COVID-19, and the first to come from a U.S. safety-net hospital in a large metropolitan area.
“Overall, the types and categories of neurological complications reported including encephalopathy (35%) and acute cerebrovascular events (~20%) are similar to those reported elsewhere,” said Dr. Tyler.
However, the frequency of stroke (~20%) is higher than in some other reports, “likely reflecting the comorbidities such as diabetes, hypertension, limited access to care [that are] present in this population,” he said.
Dr. Tyler also noted that the “relatively high frequency” of primary movement disorders, notably myoclonus, “hasn’t been particularly well recognized or described, although one of the authors has written on this in COVID-19, so perhaps there is a bit of an ‘ascertainment bias’ – as they were looking harder for it?”
Finally, he noted, it’s important to understand that all the published studies “vary tremendously in the populations they examine, so direct comparisons can be difficult.”
Also weighing in on the report in an interview, Richard Temes, MD, director, Northwell Health’s Center for Neurocritical Care in Manhasset, N.Y., said neurologic problems have been noted since the start of COVID-19 and have been well described.
“It’s common for patients to present with very nonspecific neurological complaints like confusion, disorientation, altered mental status, lethargy, but also neurological disease such as strokes, brain hemorrhages, and seizures are quite common as well,” said Dr. Temes.
He also noted that a number of patients with COVID-19 will have “lingering effects, especially patients who are hospitalized, that can range from memory deficit, cognitive slowing, and trouble with activities of daily living and depression.
“These effects can occur with any patient who is hospitalized for a [significant] period of time, especially in the intensive care unit, so it’s hard to tease out whether or not this is truly from COVID itself or if it’s just being a survivor from a very severe, critical illness. We don’t know yet. We need more data on that,” he cautioned.
A version of this article originally appeared on Medscape.com.
Serious neurologic complications in patients with COVID-19 are not limited to the severely ill, new research confirms.
“We found a range of neurologic diagnoses, including stroke and seizures, among hospitalized patients with COVID-19 and the majority were not critically ill, suggesting that these complications are not limited just to those patients who require ICU care or a ventilator,” study investigator Pria Anand, MD, division of neuro-infectious diseases, Boston University, said in an interview.
The study was published online Dec. 9 in Neurology Clinical Practice.
‘Moderately severe’ disability
For the study, the investigators reviewed the medical records of 74 adults (mean age, 64 years) who were hospitalized with COVID-19 and evaluated for neurologic conditions at Boston Medical Center, a safety-net hospital caring primarily for underserved, low-income, racial and ethnic minority populations.
The most common COVID-19 symptoms on arrival to the hospital were cough (39%), dyspnea (36%), and fever (34%). Eleven patients required intubation (15%) and 28 required some form of supplemental oxygen (38%). Thirty-four patients required intensive care (46%).
The most common neurologic COVID-19 symptoms at presentation were altered mental status (53%), myalgia (24%), fatigue (24%), and headache (18%).
After neurologic assessment, the most common final neurologic diagnosis was multifactorial or toxic-metabolic encephalopathy (35%), followed by seizure (20%), ischemic stroke (20%), primary movement disorder (9%), peripheral neuropathy (8%), and hemorrhagic stroke (4%).
Three patients (4%) suffered traumatic brain injuries after falling in their homes after developing COVID-19.
Ten (14%) patients died in the hospital. Survivors had “moderately severe” disability at discharge (median modified Rankin Scale score of 4 from a preadmission mRS score of 2) and many were discharged to nursing facilities or rehabilitation hospitals.
“Although we do not have data on their posthospital course, this suggests that patients with neurologic complications of COVID-19 are likely to require ongoing rehabilitation, even after they leave the hospital,” Dr. Anand, a member of the American Academy of Neurology, said in an interview.
“There are a diverse range of mechanisms by which COVID-19 can cause neurologic complications,” Dr. Anand said.
“These complications can result from the body’s immunological response to the virus (e.g., Guillain-Barré syndrome, an autoimmune disorder affecting the nerves), from having a systemic severe illness (e.g., brain injury as a result of insufficient oxygenation), from the increased tendency to form blood clots (e.g., stroke), from worsening of preexisting neurologic disorders, and possibly from involvement of the nervous system by the virus itself,” she explained.
The researchers said more study is needed to characterize the infectious and postinfectious neurologic complications of COVID-19 in diverse patient populations.
Lingering issues
In an interview, Kenneth L. Tyler, MD, chair of neurology, University of Colorado, Denver, noted that this is one of the larger series published to date of the neurologic complications associated with COVID-19, and the first to come from a U.S. safety-net hospital in a large metropolitan area.
“Overall, the types and categories of neurological complications reported including encephalopathy (35%) and acute cerebrovascular events (~20%) are similar to those reported elsewhere,” said Dr. Tyler.
However, the frequency of stroke (~20%) is higher than in some other reports, “likely reflecting the comorbidities such as diabetes, hypertension, limited access to care [that are] present in this population,” he said.
Dr. Tyler also noted that the “relatively high frequency” of primary movement disorders, notably myoclonus, “hasn’t been particularly well recognized or described, although one of the authors has written on this in COVID-19, so perhaps there is a bit of an ‘ascertainment bias’ – as they were looking harder for it?”
Finally, he noted, it’s important to understand that all the published studies “vary tremendously in the populations they examine, so direct comparisons can be difficult.”
Also weighing in on the report in an interview, Richard Temes, MD, director, Northwell Health’s Center for Neurocritical Care in Manhasset, N.Y., said neurologic problems have been noted since the start of COVID-19 and have been well described.
“It’s common for patients to present with very nonspecific neurological complaints like confusion, disorientation, altered mental status, lethargy, but also neurological disease such as strokes, brain hemorrhages, and seizures are quite common as well,” said Dr. Temes.
He also noted that a number of patients with COVID-19 will have “lingering effects, especially patients who are hospitalized, that can range from memory deficit, cognitive slowing, and trouble with activities of daily living and depression.
“These effects can occur with any patient who is hospitalized for a [significant] period of time, especially in the intensive care unit, so it’s hard to tease out whether or not this is truly from COVID itself or if it’s just being a survivor from a very severe, critical illness. We don’t know yet. We need more data on that,” he cautioned.
A version of this article originally appeared on Medscape.com.
Serious neurologic complications in patients with COVID-19 are not limited to the severely ill, new research confirms.
“We found a range of neurologic diagnoses, including stroke and seizures, among hospitalized patients with COVID-19 and the majority were not critically ill, suggesting that these complications are not limited just to those patients who require ICU care or a ventilator,” study investigator Pria Anand, MD, division of neuro-infectious diseases, Boston University, said in an interview.
The study was published online Dec. 9 in Neurology Clinical Practice.
‘Moderately severe’ disability
For the study, the investigators reviewed the medical records of 74 adults (mean age, 64 years) who were hospitalized with COVID-19 and evaluated for neurologic conditions at Boston Medical Center, a safety-net hospital caring primarily for underserved, low-income, racial and ethnic minority populations.
The most common COVID-19 symptoms on arrival to the hospital were cough (39%), dyspnea (36%), and fever (34%). Eleven patients required intubation (15%) and 28 required some form of supplemental oxygen (38%). Thirty-four patients required intensive care (46%).
The most common neurologic COVID-19 symptoms at presentation were altered mental status (53%), myalgia (24%), fatigue (24%), and headache (18%).
After neurologic assessment, the most common final neurologic diagnosis was multifactorial or toxic-metabolic encephalopathy (35%), followed by seizure (20%), ischemic stroke (20%), primary movement disorder (9%), peripheral neuropathy (8%), and hemorrhagic stroke (4%).
Three patients (4%) suffered traumatic brain injuries after falling in their homes after developing COVID-19.
Ten (14%) patients died in the hospital. Survivors had “moderately severe” disability at discharge (median modified Rankin Scale score of 4 from a preadmission mRS score of 2) and many were discharged to nursing facilities or rehabilitation hospitals.
“Although we do not have data on their posthospital course, this suggests that patients with neurologic complications of COVID-19 are likely to require ongoing rehabilitation, even after they leave the hospital,” Dr. Anand, a member of the American Academy of Neurology, said in an interview.
“There are a diverse range of mechanisms by which COVID-19 can cause neurologic complications,” Dr. Anand said.
“These complications can result from the body’s immunological response to the virus (e.g., Guillain-Barré syndrome, an autoimmune disorder affecting the nerves), from having a systemic severe illness (e.g., brain injury as a result of insufficient oxygenation), from the increased tendency to form blood clots (e.g., stroke), from worsening of preexisting neurologic disorders, and possibly from involvement of the nervous system by the virus itself,” she explained.
The researchers said more study is needed to characterize the infectious and postinfectious neurologic complications of COVID-19 in diverse patient populations.
Lingering issues
In an interview, Kenneth L. Tyler, MD, chair of neurology, University of Colorado, Denver, noted that this is one of the larger series published to date of the neurologic complications associated with COVID-19, and the first to come from a U.S. safety-net hospital in a large metropolitan area.
“Overall, the types and categories of neurological complications reported including encephalopathy (35%) and acute cerebrovascular events (~20%) are similar to those reported elsewhere,” said Dr. Tyler.
However, the frequency of stroke (~20%) is higher than in some other reports, “likely reflecting the comorbidities such as diabetes, hypertension, limited access to care [that are] present in this population,” he said.
Dr. Tyler also noted that the “relatively high frequency” of primary movement disorders, notably myoclonus, “hasn’t been particularly well recognized or described, although one of the authors has written on this in COVID-19, so perhaps there is a bit of an ‘ascertainment bias’ – as they were looking harder for it?”
Finally, he noted, it’s important to understand that all the published studies “vary tremendously in the populations they examine, so direct comparisons can be difficult.”
Also weighing in on the report in an interview, Richard Temes, MD, director, Northwell Health’s Center for Neurocritical Care in Manhasset, N.Y., said neurologic problems have been noted since the start of COVID-19 and have been well described.
“It’s common for patients to present with very nonspecific neurological complaints like confusion, disorientation, altered mental status, lethargy, but also neurological disease such as strokes, brain hemorrhages, and seizures are quite common as well,” said Dr. Temes.
He also noted that a number of patients with COVID-19 will have “lingering effects, especially patients who are hospitalized, that can range from memory deficit, cognitive slowing, and trouble with activities of daily living and depression.
“These effects can occur with any patient who is hospitalized for a [significant] period of time, especially in the intensive care unit, so it’s hard to tease out whether or not this is truly from COVID itself or if it’s just being a survivor from a very severe, critical illness. We don’t know yet. We need more data on that,” he cautioned.
A version of this article originally appeared on Medscape.com.
Itchy scalp with scale
An 11-year-old boy sought care at a small village’s health center in Panama for scalp itching and subtle hair loss. He was seen by a family physician (RU) and a team of medical students who were there as part of a humanitarian trip. The patient denied any hair pulling. He had a history of treatment for head lice.
Our physical examination revealed mild alopecia and scaling on the scalp (FIGURE 1), but what we saw through the dermatoscope (FIGURE 2) made the diagnosis clear.
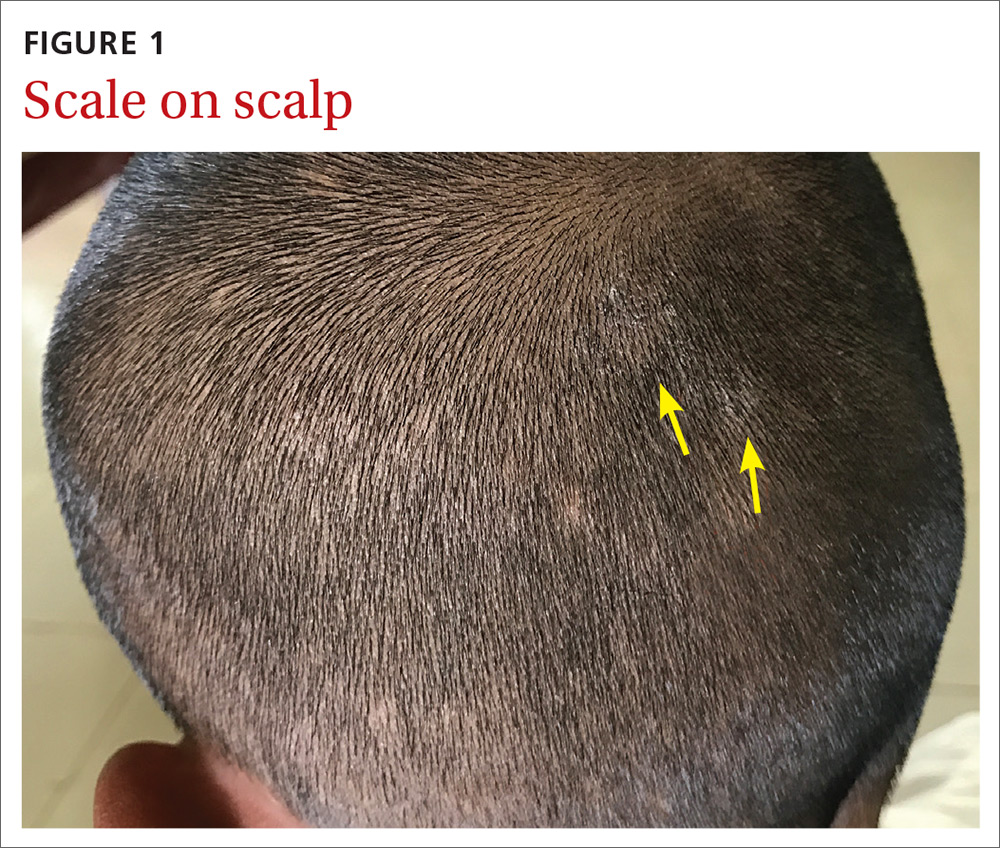
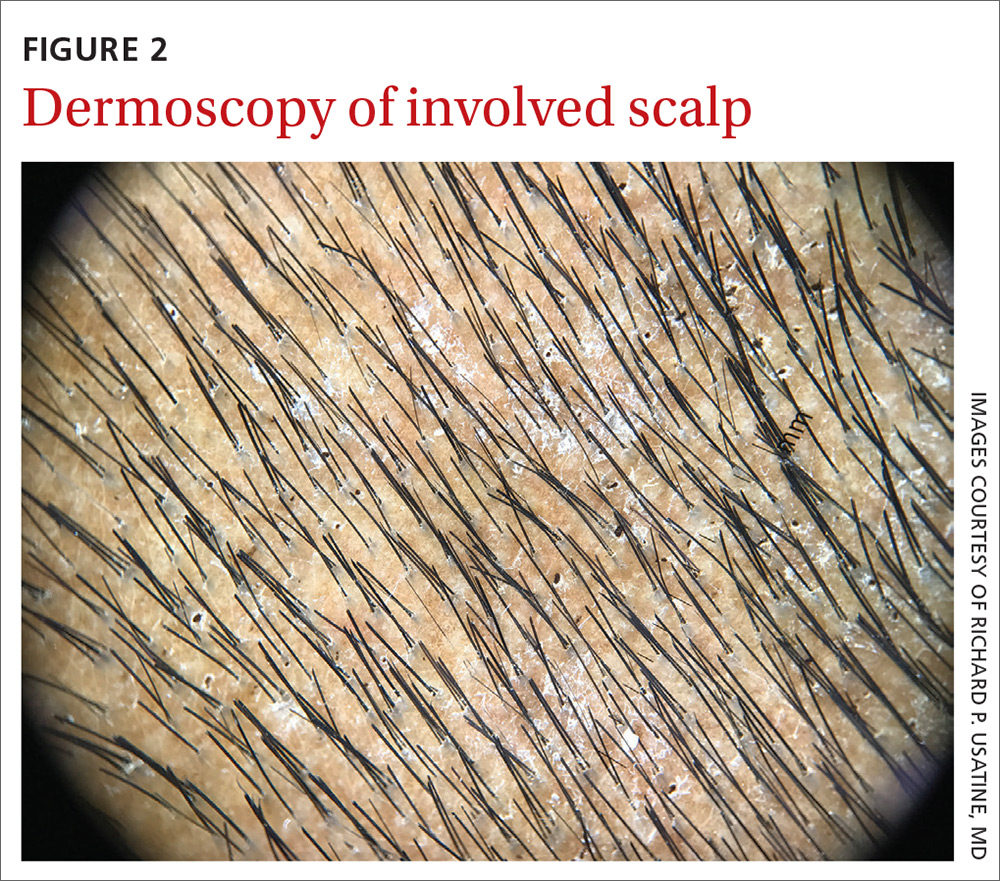
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Tinea capitis
On dermatoscopic examination (10× magnification), there were numerous “black dots” or broken hair shafts within patches of hair loss (FIGURE 3), which is indicative of tinea capitis.1,2 This condition causes hair shafts to break, creating “comma hairs” and black dots. The hairs are uniform in thickness and color and bend distally, like a comma.3
Tinea capitis (commonly called ringworm of the scalp) is a fungal infection caused by Trichophyton and Microsporum dermatophytes. It is the most common pediatric dermatophyte infection in the world; the usual age of onset is 5 to 10 years.2 The incidence of tinea capitis in the United States is not known because cases are no longer registered by public health agencies. That said, a Northern California study that tracked occurrences in children younger than 15 years from 1998 to 2007 found that the incidence was on the decline and lower in girls compared to boys (111.9 vs 146.4, respectively, in 1998; 27.9 vs 39.9, respectively, in 2007).4 Incidence rates were calculated per 10,000 eligible children.4
Tinea capitis can spread by contact with infected individuals and contaminated objects, including combs, towels, toys, and bedding.1 Fungal spores can remain viable on these surfaces for months.
In a study of 69 patients with tinea capitis (23 females, 46 males; mean age, 12 years), the risk factors for spreading infection included participation in sports, contact with an animal, a recent haircut, and use of a swimming pool.5
4 conditions you’ll want to rule out
The following conditions should be considered as part of the differential when a patient presents with an itchy scalp and/or hair loss.
Continue to: Psoriasis of the scalp...
Psoriasis of the scalp is characterized by scaling of the scalp along with crusted plaques. It is often accompanied by similar psoriatic plaques on the elbows, knees, and other areas of the body. Examination of our patient showed no psoriatic plaques.
Seborrhea of the scalp (also known as dandruff) is a very common diagnosis. However, it is unlikely to cause hair loss. It has widespread involvement of the scalp compared to tinea capitis, which is local and patchy. Our patient’s patches of hair loss indicated that seborrhea was unlikely.
Alopecia areata. Individuals develop this condition due to an autoimmune process affecting hair follicles. However, the resulting hair loss does not cause significant scaling, inflammation, scarring, or pain in the affected area. Further, this condition can cause the loss of the entire hair shaft.
Trichotillomania is an impulse control disorder that causes patients to pull out their own hair. There is no scaling of the scalp in this condition.
A dermatoscope can beuseful in making the Dx
Although clinical appearance and patient presentation are adequate to establish the diagnosis of tinea capitis, this case demonstrates the utility of a dermatoscope in making the diagnosis of tinea capitis. Previous studies have shown that dermoscopy allows for rapid identification of the broken hair shafts, which are a key distinction from alopecia areata.3,6
Microscopic inspection. Samples from the scaling of the scalp can be examined with potassium hydroxide (KOH) on a microscope slide. Hyphae, spores, and endo/ectothrix invasion can be seen through the microsope.
Continue to: Laboratory testing is helpful, but not needed.
Laboratory testing is helpful, but not needed. Testing for tinea capitis would require that you obtain a sample from the affected area using a swab, edge of a scalpel blade, or scalp brush.7 Because treatment can require weeks of medication, diagnosis should be confirmed with a KOH or culture when possible.
Newer antifungalsprovide a Tx advantage
Oral antifungal medications are the treatment of choice for tinea capitis. Newer antifungals, such as terbinafine and fluconazole, require a 3- to 6-week course compared to the standard 6- to 8-week course of griseofulvin.1 Also, antifungal shampoos—such as those that contain selenium sulfide—may be used for topical treatment but only as adjuvant therapy.1,2
For our patient, we dispensed a 3-week course of oral fluconazole, 3 to 6 mg/kg, to be given daily by his parents. We also recommended the use of an antidandruff shampoo, if possible. The treatment outcome was not known because our team’s humanitarian global health trip had ended.
1. Usatine R, Smith MA, Mayeaux Jr EJ, Chumley HS. The Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019.
2. Handler MZ. Tinea capitis. Medscape. https://emedicine.medscape.com/article/1091351-overview. Updated February 21, 2020. Accessed November 30, 2020.
3. Hernández-Bel P, Malvehy J, Crocker A, et al. Comma hairs: a new dermoscopic marker for tinea capitis [in Spanish]. Actas Dermosifiliogr. 2012;103:836-837.
4. Mirmirani P, Lue-Yen T. Epidemiologic trends in pediatric tinea capitis: a population-based study from Kaiser Permanente Northern California. J Am Acad Dermatol. 2013;69:916-921.
5. Mikaeili A, Kavaoussi H, Hashemian AH, et al. Clinico-mycological profile of tinea capitis and its comparative response to griseofulvin versus terbinafine. Curr Med Mycol. 2019;5:15-20.
6. Slowinska M, Rudnicka L, Schwartz RA, et al. Comma hairs: a dermatoscopic marker for tinea capitis: a rapid diagnostic method. Journal of the American Academy of Dermatology. 2008;59(suppl 5):S77-S79.
An 11-year-old boy sought care at a small village’s health center in Panama for scalp itching and subtle hair loss. He was seen by a family physician (RU) and a team of medical students who were there as part of a humanitarian trip. The patient denied any hair pulling. He had a history of treatment for head lice.
Our physical examination revealed mild alopecia and scaling on the scalp (FIGURE 1), but what we saw through the dermatoscope (FIGURE 2) made the diagnosis clear.


WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Tinea capitis
On dermatoscopic examination (10× magnification), there were numerous “black dots” or broken hair shafts within patches of hair loss (FIGURE 3), which is indicative of tinea capitis.1,2 This condition causes hair shafts to break, creating “comma hairs” and black dots. The hairs are uniform in thickness and color and bend distally, like a comma.3
Tinea capitis (commonly called ringworm of the scalp) is a fungal infection caused by Trichophyton and Microsporum dermatophytes. It is the most common pediatric dermatophyte infection in the world; the usual age of onset is 5 to 10 years.2 The incidence of tinea capitis in the United States is not known because cases are no longer registered by public health agencies. That said, a Northern California study that tracked occurrences in children younger than 15 years from 1998 to 2007 found that the incidence was on the decline and lower in girls compared to boys (111.9 vs 146.4, respectively, in 1998; 27.9 vs 39.9, respectively, in 2007).4 Incidence rates were calculated per 10,000 eligible children.4

Tinea capitis can spread by contact with infected individuals and contaminated objects, including combs, towels, toys, and bedding.1 Fungal spores can remain viable on these surfaces for months.
In a study of 69 patients with tinea capitis (23 females, 46 males; mean age, 12 years), the risk factors for spreading infection included participation in sports, contact with an animal, a recent haircut, and use of a swimming pool.5
4 conditions you’ll want to rule out
The following conditions should be considered as part of the differential when a patient presents with an itchy scalp and/or hair loss.
Continue to: Psoriasis of the scalp...
Psoriasis of the scalp is characterized by scaling of the scalp along with crusted plaques. It is often accompanied by similar psoriatic plaques on the elbows, knees, and other areas of the body. Examination of our patient showed no psoriatic plaques.
Seborrhea of the scalp (also known as dandruff) is a very common diagnosis. However, it is unlikely to cause hair loss. It has widespread involvement of the scalp compared to tinea capitis, which is local and patchy. Our patient’s patches of hair loss indicated that seborrhea was unlikely.
Alopecia areata. Individuals develop this condition due to an autoimmune process affecting hair follicles. However, the resulting hair loss does not cause significant scaling, inflammation, scarring, or pain in the affected area. Further, this condition can cause the loss of the entire hair shaft.
Trichotillomania is an impulse control disorder that causes patients to pull out their own hair. There is no scaling of the scalp in this condition.
A dermatoscope can beuseful in making the Dx
Although clinical appearance and patient presentation are adequate to establish the diagnosis of tinea capitis, this case demonstrates the utility of a dermatoscope in making the diagnosis of tinea capitis. Previous studies have shown that dermoscopy allows for rapid identification of the broken hair shafts, which are a key distinction from alopecia areata.3,6
Microscopic inspection. Samples from the scaling of the scalp can be examined with potassium hydroxide (KOH) on a microscope slide. Hyphae, spores, and endo/ectothrix invasion can be seen through the microsope.
Continue to: Laboratory testing is helpful, but not needed.
Laboratory testing is helpful, but not needed. Testing for tinea capitis would require that you obtain a sample from the affected area using a swab, edge of a scalpel blade, or scalp brush.7 Because treatment can require weeks of medication, diagnosis should be confirmed with a KOH or culture when possible.
Newer antifungalsprovide a Tx advantage
Oral antifungal medications are the treatment of choice for tinea capitis. Newer antifungals, such as terbinafine and fluconazole, require a 3- to 6-week course compared to the standard 6- to 8-week course of griseofulvin.1 Also, antifungal shampoos—such as those that contain selenium sulfide—may be used for topical treatment but only as adjuvant therapy.1,2
For our patient, we dispensed a 3-week course of oral fluconazole, 3 to 6 mg/kg, to be given daily by his parents. We also recommended the use of an antidandruff shampoo, if possible. The treatment outcome was not known because our team’s humanitarian global health trip had ended.
An 11-year-old boy sought care at a small village’s health center in Panama for scalp itching and subtle hair loss. He was seen by a family physician (RU) and a team of medical students who were there as part of a humanitarian trip. The patient denied any hair pulling. He had a history of treatment for head lice.
Our physical examination revealed mild alopecia and scaling on the scalp (FIGURE 1), but what we saw through the dermatoscope (FIGURE 2) made the diagnosis clear.


WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Tinea capitis
On dermatoscopic examination (10× magnification), there were numerous “black dots” or broken hair shafts within patches of hair loss (FIGURE 3), which is indicative of tinea capitis.1,2 This condition causes hair shafts to break, creating “comma hairs” and black dots. The hairs are uniform in thickness and color and bend distally, like a comma.3
Tinea capitis (commonly called ringworm of the scalp) is a fungal infection caused by Trichophyton and Microsporum dermatophytes. It is the most common pediatric dermatophyte infection in the world; the usual age of onset is 5 to 10 years.2 The incidence of tinea capitis in the United States is not known because cases are no longer registered by public health agencies. That said, a Northern California study that tracked occurrences in children younger than 15 years from 1998 to 2007 found that the incidence was on the decline and lower in girls compared to boys (111.9 vs 146.4, respectively, in 1998; 27.9 vs 39.9, respectively, in 2007).4 Incidence rates were calculated per 10,000 eligible children.4

Tinea capitis can spread by contact with infected individuals and contaminated objects, including combs, towels, toys, and bedding.1 Fungal spores can remain viable on these surfaces for months.
In a study of 69 patients with tinea capitis (23 females, 46 males; mean age, 12 years), the risk factors for spreading infection included participation in sports, contact with an animal, a recent haircut, and use of a swimming pool.5
4 conditions you’ll want to rule out
The following conditions should be considered as part of the differential when a patient presents with an itchy scalp and/or hair loss.
Continue to: Psoriasis of the scalp...
Psoriasis of the scalp is characterized by scaling of the scalp along with crusted plaques. It is often accompanied by similar psoriatic plaques on the elbows, knees, and other areas of the body. Examination of our patient showed no psoriatic plaques.
Seborrhea of the scalp (also known as dandruff) is a very common diagnosis. However, it is unlikely to cause hair loss. It has widespread involvement of the scalp compared to tinea capitis, which is local and patchy. Our patient’s patches of hair loss indicated that seborrhea was unlikely.
Alopecia areata. Individuals develop this condition due to an autoimmune process affecting hair follicles. However, the resulting hair loss does not cause significant scaling, inflammation, scarring, or pain in the affected area. Further, this condition can cause the loss of the entire hair shaft.
Trichotillomania is an impulse control disorder that causes patients to pull out their own hair. There is no scaling of the scalp in this condition.
A dermatoscope can beuseful in making the Dx
Although clinical appearance and patient presentation are adequate to establish the diagnosis of tinea capitis, this case demonstrates the utility of a dermatoscope in making the diagnosis of tinea capitis. Previous studies have shown that dermoscopy allows for rapid identification of the broken hair shafts, which are a key distinction from alopecia areata.3,6
Microscopic inspection. Samples from the scaling of the scalp can be examined with potassium hydroxide (KOH) on a microscope slide. Hyphae, spores, and endo/ectothrix invasion can be seen through the microsope.
Continue to: Laboratory testing is helpful, but not needed.
Laboratory testing is helpful, but not needed. Testing for tinea capitis would require that you obtain a sample from the affected area using a swab, edge of a scalpel blade, or scalp brush.7 Because treatment can require weeks of medication, diagnosis should be confirmed with a KOH or culture when possible.
Newer antifungalsprovide a Tx advantage
Oral antifungal medications are the treatment of choice for tinea capitis. Newer antifungals, such as terbinafine and fluconazole, require a 3- to 6-week course compared to the standard 6- to 8-week course of griseofulvin.1 Also, antifungal shampoos—such as those that contain selenium sulfide—may be used for topical treatment but only as adjuvant therapy.1,2
For our patient, we dispensed a 3-week course of oral fluconazole, 3 to 6 mg/kg, to be given daily by his parents. We also recommended the use of an antidandruff shampoo, if possible. The treatment outcome was not known because our team’s humanitarian global health trip had ended.
1. Usatine R, Smith MA, Mayeaux Jr EJ, Chumley HS. The Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019.
2. Handler MZ. Tinea capitis. Medscape. https://emedicine.medscape.com/article/1091351-overview. Updated February 21, 2020. Accessed November 30, 2020.
3. Hernández-Bel P, Malvehy J, Crocker A, et al. Comma hairs: a new dermoscopic marker for tinea capitis [in Spanish]. Actas Dermosifiliogr. 2012;103:836-837.
4. Mirmirani P, Lue-Yen T. Epidemiologic trends in pediatric tinea capitis: a population-based study from Kaiser Permanente Northern California. J Am Acad Dermatol. 2013;69:916-921.
5. Mikaeili A, Kavaoussi H, Hashemian AH, et al. Clinico-mycological profile of tinea capitis and its comparative response to griseofulvin versus terbinafine. Curr Med Mycol. 2019;5:15-20.
6. Slowinska M, Rudnicka L, Schwartz RA, et al. Comma hairs: a dermatoscopic marker for tinea capitis: a rapid diagnostic method. Journal of the American Academy of Dermatology. 2008;59(suppl 5):S77-S79.
1. Usatine R, Smith MA, Mayeaux Jr EJ, Chumley HS. The Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019.
2. Handler MZ. Tinea capitis. Medscape. https://emedicine.medscape.com/article/1091351-overview. Updated February 21, 2020. Accessed November 30, 2020.
3. Hernández-Bel P, Malvehy J, Crocker A, et al. Comma hairs: a new dermoscopic marker for tinea capitis [in Spanish]. Actas Dermosifiliogr. 2012;103:836-837.
4. Mirmirani P, Lue-Yen T. Epidemiologic trends in pediatric tinea capitis: a population-based study from Kaiser Permanente Northern California. J Am Acad Dermatol. 2013;69:916-921.
5. Mikaeili A, Kavaoussi H, Hashemian AH, et al. Clinico-mycological profile of tinea capitis and its comparative response to griseofulvin versus terbinafine. Curr Med Mycol. 2019;5:15-20.
6. Slowinska M, Rudnicka L, Schwartz RA, et al. Comma hairs: a dermatoscopic marker for tinea capitis: a rapid diagnostic method. Journal of the American Academy of Dermatology. 2008;59(suppl 5):S77-S79.