User login
Expected spike in acute flaccid myelitis did not occur in 2020
suggested researchers at the Centers for Disease Control and Prevention.
Acute flaccid myelitis (AFM) is an uncommon but serious complication of some viral infections, including West Nile virus and nonpolio enteroviruses. It is “characterized by sudden onset of limb weakness and lesions in the gray matter of the spinal cord,” they said, and more than 90% of cases occur in young children.
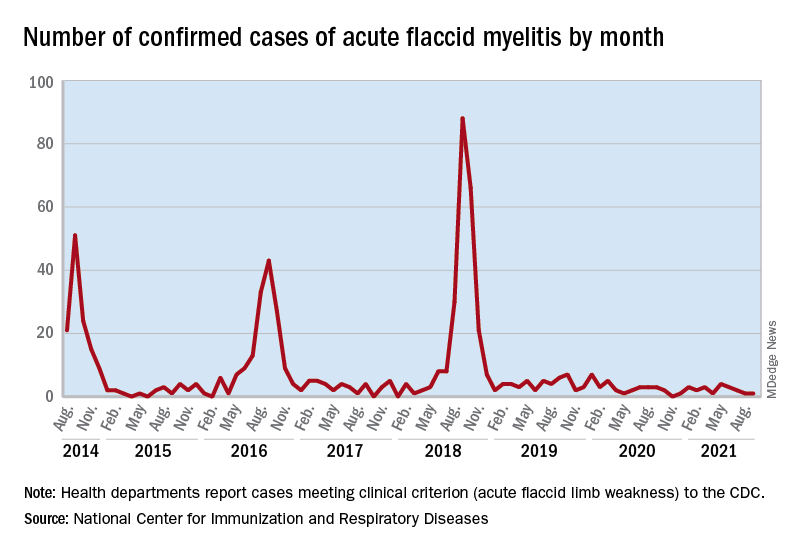
Cases of AFM, which can lead to respiratory insufficiency and permanent paralysis, spiked during the late summer and early fall in 2014, 2016, and 2018 and were expected to do so again in 2020, Sarah Kidd, MD, and associates at the division of viral diseases at the CDC’s National Center for Immunization and Respiratory Diseases, Atlanta, said in the Morbidity and Mortality Weekly Report.
Monthly peaks in those previous years – each occurring in September – reached 51 cases in 2014, 43 cases in 2016, and 88 cases in 2018, but in 2020 there was only 1 case reported in September, with a high of 4 coming in May, CDC data show. The total number of cases for 2020 (32) was, in fact, lower than in 2019, when 47 were reported.
The investigators’ main objective was to see if there were any differences between the 2018 and 2019-2020 cases. Reports from state health departments to the CDC showed that, in 2019-2020, “patients were older; more likely to have lower limb involvement; and less likely to have upper limb involvement, prodromal illness, [cerebrospinal fluid] pleocytosis, or specimens that tested positive for EV [enterovirus]-D68” than patients from 2018, Dr. Kidd and associates said.
Mask wearing and reduced in-school attendance may have decreased circulation of EV-D68 – the enterovirus type most often detected in the stool and respiratory specimens of AFM patients – as was seen with other respiratory viruses, such as influenza and respiratory syncytial virus, in 2020. Previous studies have suggested that EV-D68 drives the increases in cases during peak years, the researchers noted.
The absence of such an increase “in 2020 reflects a deviation from the previously observed biennial pattern, and it is unclear when the next increase in AFM should be expected. Clinicians should continue to maintain vigilance and suspect AFM in any child with acute flaccid limb weakness, particularly in the setting of recent febrile or respiratory illness,” they wrote.
suggested researchers at the Centers for Disease Control and Prevention.

Acute flaccid myelitis (AFM) is an uncommon but serious complication of some viral infections, including West Nile virus and nonpolio enteroviruses. It is “characterized by sudden onset of limb weakness and lesions in the gray matter of the spinal cord,” they said, and more than 90% of cases occur in young children.
Cases of AFM, which can lead to respiratory insufficiency and permanent paralysis, spiked during the late summer and early fall in 2014, 2016, and 2018 and were expected to do so again in 2020, Sarah Kidd, MD, and associates at the division of viral diseases at the CDC’s National Center for Immunization and Respiratory Diseases, Atlanta, said in the Morbidity and Mortality Weekly Report.
Monthly peaks in those previous years – each occurring in September – reached 51 cases in 2014, 43 cases in 2016, and 88 cases in 2018, but in 2020 there was only 1 case reported in September, with a high of 4 coming in May, CDC data show. The total number of cases for 2020 (32) was, in fact, lower than in 2019, when 47 were reported.
The investigators’ main objective was to see if there were any differences between the 2018 and 2019-2020 cases. Reports from state health departments to the CDC showed that, in 2019-2020, “patients were older; more likely to have lower limb involvement; and less likely to have upper limb involvement, prodromal illness, [cerebrospinal fluid] pleocytosis, or specimens that tested positive for EV [enterovirus]-D68” than patients from 2018, Dr. Kidd and associates said.
Mask wearing and reduced in-school attendance may have decreased circulation of EV-D68 – the enterovirus type most often detected in the stool and respiratory specimens of AFM patients – as was seen with other respiratory viruses, such as influenza and respiratory syncytial virus, in 2020. Previous studies have suggested that EV-D68 drives the increases in cases during peak years, the researchers noted.
The absence of such an increase “in 2020 reflects a deviation from the previously observed biennial pattern, and it is unclear when the next increase in AFM should be expected. Clinicians should continue to maintain vigilance and suspect AFM in any child with acute flaccid limb weakness, particularly in the setting of recent febrile or respiratory illness,” they wrote.
suggested researchers at the Centers for Disease Control and Prevention.

Acute flaccid myelitis (AFM) is an uncommon but serious complication of some viral infections, including West Nile virus and nonpolio enteroviruses. It is “characterized by sudden onset of limb weakness and lesions in the gray matter of the spinal cord,” they said, and more than 90% of cases occur in young children.
Cases of AFM, which can lead to respiratory insufficiency and permanent paralysis, spiked during the late summer and early fall in 2014, 2016, and 2018 and were expected to do so again in 2020, Sarah Kidd, MD, and associates at the division of viral diseases at the CDC’s National Center for Immunization and Respiratory Diseases, Atlanta, said in the Morbidity and Mortality Weekly Report.
Monthly peaks in those previous years – each occurring in September – reached 51 cases in 2014, 43 cases in 2016, and 88 cases in 2018, but in 2020 there was only 1 case reported in September, with a high of 4 coming in May, CDC data show. The total number of cases for 2020 (32) was, in fact, lower than in 2019, when 47 were reported.
The investigators’ main objective was to see if there were any differences between the 2018 and 2019-2020 cases. Reports from state health departments to the CDC showed that, in 2019-2020, “patients were older; more likely to have lower limb involvement; and less likely to have upper limb involvement, prodromal illness, [cerebrospinal fluid] pleocytosis, or specimens that tested positive for EV [enterovirus]-D68” than patients from 2018, Dr. Kidd and associates said.
Mask wearing and reduced in-school attendance may have decreased circulation of EV-D68 – the enterovirus type most often detected in the stool and respiratory specimens of AFM patients – as was seen with other respiratory viruses, such as influenza and respiratory syncytial virus, in 2020. Previous studies have suggested that EV-D68 drives the increases in cases during peak years, the researchers noted.
The absence of such an increase “in 2020 reflects a deviation from the previously observed biennial pattern, and it is unclear when the next increase in AFM should be expected. Clinicians should continue to maintain vigilance and suspect AFM in any child with acute flaccid limb weakness, particularly in the setting of recent febrile or respiratory illness,” they wrote.
FROM MMWR
Antihypertensives tied to lower Alzheimer’s disease pathology
new research shows.
Investigators found that use of any antihypertensive was associated with an 18% decrease in Alzheimer’s disease neuropathology, a 22% decrease in Lewy bodies, and a 40% decrease in TAR DNA-binding protein 43 (TDP-43), a protein relevant to several neurodegenerative diseases. Diuretics in particular appear to be driving the association.
Although diuretics might be a better option for preventing brain neuropathology, it’s too early to make firm recommendations solely on the basis of these results as to what blood pressure–lowering agent to prescribe a particular patient, said study investigator Ahmad Sajjadi, MD, assistant professor of neurology, University of California, Irvine.
“This is early stages and preliminary results,” said Dr. Sajjadi, “but it’s food for thought.”
The findings were presented at the 2021 annual meeting of the American Neurological Association.
Autopsy data
The study included 3,315 individuals who had donated their brains to research. The National Alzheimer’s Coordinating Center maintains a database that includes data from 32 Alzheimer’s disease research centers in the United States. Participants in the study must have visited one of these centers within 4 years of death. Each person whose brain was included in the study underwent two or more BP measurements on at least 50% of visits.
The mean age at death was 81.7 years, and the mean time between last visit and death was 13.1 months. About 44.4% of participants were women, 57.0% had at least a college degree, and 84.7% had cognitive impairment.
Researchers defined hypertension as systolic BP of at least 130 mm Hg, diastolic BP of at least 80 mm Hg, mean arterial pressure of at least 100 mm Hg, and pulse pressure of at least 60 mm Hg.
Antihypertensive medications that were evaluated included antiadrenergic agents, ACE inhibitors, angiotensin II receptor blockers, beta blockers, calcium channel blockers, diuretics, vasodilators, and combination therapies.
The investigators assessed the number of neuropathologies. In addition to Alzheimer’s disease neuropathology, which included amyloid-beta, tau, Lewy bodies, and TDP-43, they also assessed for atherosclerosis, arteriolosclerosis, cerebral amyloid angiopathy, frontotemporal lobar degeneration, and hippocampal sclerosis.
Results showed that use of any antihypertensive was associated with a lower likelihood of Alzheimer’s disease neuropathology (odds ratio, 0.822), Lewy bodies (OR, 0.786), and TDP 43 (OR, 0.597). Use of antihypertensives was also associated with increased odds of atherosclerosis (OR, 1.217) (all P < .5.)
The study showed that hypertensive systolic BP was associated with higher odds of Alzheimer’s disease neuropathology (OR, 1.28; P < .5).
Differences by drug type
Results differed in accordance with antihypertensive class. Angiotensin II receptor blockers decreased the odds of Alzheimer’s disease neuropathology by 40% (OR, 0.60; P < .5). Diuretics decreased the odds of Alzheimer’s disease by 36% (OR, 0.64; P < .001) and of hippocampal sclerosis by 32% (OR, 0.68; P < .5).
“We see diuretics are a main driver, especially for lower odds of Alzheimer’s disease and lower odds of hippocampal sclerosis,” said lead author Hanna L. Nguyen, a first-year medical student at the University of California, Irvine.
The results indicate that it is the medications, not BP levels, that account for these associations, she added.
One potential mechanism linking antihypertensives to brain pathology is that with these agents, BP is maintained in the target zone. Blood pressure that’s too high can damage blood vessels, whereas BP that’s too low may result in less than adequate perfusion, said Ms. Nguyen.
These medications may also alter pathways leading to degeneration and could, for example, affect the apo E mechanism of Alzheimer’s disease, she added.
The researchers plan to conduct subset analyses using apo E genetic status and age of death.
Although this is a “massive database,” it has limitations. For example, said Dr. Sajjadi, it does not reveal when patients started taking BP medication, how long they had been taking it, or why.
“We don’t know the exact the reason they were taking these medications. Was it just hypertension, or did they also have heart disease, stroke, a kidney problem, or was there another explanation,” he said.
Following the study presentation, session comoderator Krish Sathian, MBBS, PhD, professor of neurology, neural, and behavioral sciences, and psychology and director of the Neuroscience Institute, Penn State University, Hershey, called this work “fascinating. It provides a lot of data that really touches on everyday practice,” inasmuch as clinicians often prescribe antihypertensive medications and see patients with these kinds of brain disorders.
The investigators and Dr. Sathian reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research shows.
Investigators found that use of any antihypertensive was associated with an 18% decrease in Alzheimer’s disease neuropathology, a 22% decrease in Lewy bodies, and a 40% decrease in TAR DNA-binding protein 43 (TDP-43), a protein relevant to several neurodegenerative diseases. Diuretics in particular appear to be driving the association.
Although diuretics might be a better option for preventing brain neuropathology, it’s too early to make firm recommendations solely on the basis of these results as to what blood pressure–lowering agent to prescribe a particular patient, said study investigator Ahmad Sajjadi, MD, assistant professor of neurology, University of California, Irvine.
“This is early stages and preliminary results,” said Dr. Sajjadi, “but it’s food for thought.”
The findings were presented at the 2021 annual meeting of the American Neurological Association.
Autopsy data
The study included 3,315 individuals who had donated their brains to research. The National Alzheimer’s Coordinating Center maintains a database that includes data from 32 Alzheimer’s disease research centers in the United States. Participants in the study must have visited one of these centers within 4 years of death. Each person whose brain was included in the study underwent two or more BP measurements on at least 50% of visits.
The mean age at death was 81.7 years, and the mean time between last visit and death was 13.1 months. About 44.4% of participants were women, 57.0% had at least a college degree, and 84.7% had cognitive impairment.
Researchers defined hypertension as systolic BP of at least 130 mm Hg, diastolic BP of at least 80 mm Hg, mean arterial pressure of at least 100 mm Hg, and pulse pressure of at least 60 mm Hg.
Antihypertensive medications that were evaluated included antiadrenergic agents, ACE inhibitors, angiotensin II receptor blockers, beta blockers, calcium channel blockers, diuretics, vasodilators, and combination therapies.
The investigators assessed the number of neuropathologies. In addition to Alzheimer’s disease neuropathology, which included amyloid-beta, tau, Lewy bodies, and TDP-43, they also assessed for atherosclerosis, arteriolosclerosis, cerebral amyloid angiopathy, frontotemporal lobar degeneration, and hippocampal sclerosis.
Results showed that use of any antihypertensive was associated with a lower likelihood of Alzheimer’s disease neuropathology (odds ratio, 0.822), Lewy bodies (OR, 0.786), and TDP 43 (OR, 0.597). Use of antihypertensives was also associated with increased odds of atherosclerosis (OR, 1.217) (all P < .5.)
The study showed that hypertensive systolic BP was associated with higher odds of Alzheimer’s disease neuropathology (OR, 1.28; P < .5).
Differences by drug type
Results differed in accordance with antihypertensive class. Angiotensin II receptor blockers decreased the odds of Alzheimer’s disease neuropathology by 40% (OR, 0.60; P < .5). Diuretics decreased the odds of Alzheimer’s disease by 36% (OR, 0.64; P < .001) and of hippocampal sclerosis by 32% (OR, 0.68; P < .5).
“We see diuretics are a main driver, especially for lower odds of Alzheimer’s disease and lower odds of hippocampal sclerosis,” said lead author Hanna L. Nguyen, a first-year medical student at the University of California, Irvine.
The results indicate that it is the medications, not BP levels, that account for these associations, she added.
One potential mechanism linking antihypertensives to brain pathology is that with these agents, BP is maintained in the target zone. Blood pressure that’s too high can damage blood vessels, whereas BP that’s too low may result in less than adequate perfusion, said Ms. Nguyen.
These medications may also alter pathways leading to degeneration and could, for example, affect the apo E mechanism of Alzheimer’s disease, she added.
The researchers plan to conduct subset analyses using apo E genetic status and age of death.
Although this is a “massive database,” it has limitations. For example, said Dr. Sajjadi, it does not reveal when patients started taking BP medication, how long they had been taking it, or why.
“We don’t know the exact the reason they were taking these medications. Was it just hypertension, or did they also have heart disease, stroke, a kidney problem, or was there another explanation,” he said.
Following the study presentation, session comoderator Krish Sathian, MBBS, PhD, professor of neurology, neural, and behavioral sciences, and psychology and director of the Neuroscience Institute, Penn State University, Hershey, called this work “fascinating. It provides a lot of data that really touches on everyday practice,” inasmuch as clinicians often prescribe antihypertensive medications and see patients with these kinds of brain disorders.
The investigators and Dr. Sathian reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research shows.
Investigators found that use of any antihypertensive was associated with an 18% decrease in Alzheimer’s disease neuropathology, a 22% decrease in Lewy bodies, and a 40% decrease in TAR DNA-binding protein 43 (TDP-43), a protein relevant to several neurodegenerative diseases. Diuretics in particular appear to be driving the association.
Although diuretics might be a better option for preventing brain neuropathology, it’s too early to make firm recommendations solely on the basis of these results as to what blood pressure–lowering agent to prescribe a particular patient, said study investigator Ahmad Sajjadi, MD, assistant professor of neurology, University of California, Irvine.
“This is early stages and preliminary results,” said Dr. Sajjadi, “but it’s food for thought.”
The findings were presented at the 2021 annual meeting of the American Neurological Association.
Autopsy data
The study included 3,315 individuals who had donated their brains to research. The National Alzheimer’s Coordinating Center maintains a database that includes data from 32 Alzheimer’s disease research centers in the United States. Participants in the study must have visited one of these centers within 4 years of death. Each person whose brain was included in the study underwent two or more BP measurements on at least 50% of visits.
The mean age at death was 81.7 years, and the mean time between last visit and death was 13.1 months. About 44.4% of participants were women, 57.0% had at least a college degree, and 84.7% had cognitive impairment.
Researchers defined hypertension as systolic BP of at least 130 mm Hg, diastolic BP of at least 80 mm Hg, mean arterial pressure of at least 100 mm Hg, and pulse pressure of at least 60 mm Hg.
Antihypertensive medications that were evaluated included antiadrenergic agents, ACE inhibitors, angiotensin II receptor blockers, beta blockers, calcium channel blockers, diuretics, vasodilators, and combination therapies.
The investigators assessed the number of neuropathologies. In addition to Alzheimer’s disease neuropathology, which included amyloid-beta, tau, Lewy bodies, and TDP-43, they also assessed for atherosclerosis, arteriolosclerosis, cerebral amyloid angiopathy, frontotemporal lobar degeneration, and hippocampal sclerosis.
Results showed that use of any antihypertensive was associated with a lower likelihood of Alzheimer’s disease neuropathology (odds ratio, 0.822), Lewy bodies (OR, 0.786), and TDP 43 (OR, 0.597). Use of antihypertensives was also associated with increased odds of atherosclerosis (OR, 1.217) (all P < .5.)
The study showed that hypertensive systolic BP was associated with higher odds of Alzheimer’s disease neuropathology (OR, 1.28; P < .5).
Differences by drug type
Results differed in accordance with antihypertensive class. Angiotensin II receptor blockers decreased the odds of Alzheimer’s disease neuropathology by 40% (OR, 0.60; P < .5). Diuretics decreased the odds of Alzheimer’s disease by 36% (OR, 0.64; P < .001) and of hippocampal sclerosis by 32% (OR, 0.68; P < .5).
“We see diuretics are a main driver, especially for lower odds of Alzheimer’s disease and lower odds of hippocampal sclerosis,” said lead author Hanna L. Nguyen, a first-year medical student at the University of California, Irvine.
The results indicate that it is the medications, not BP levels, that account for these associations, she added.
One potential mechanism linking antihypertensives to brain pathology is that with these agents, BP is maintained in the target zone. Blood pressure that’s too high can damage blood vessels, whereas BP that’s too low may result in less than adequate perfusion, said Ms. Nguyen.
These medications may also alter pathways leading to degeneration and could, for example, affect the apo E mechanism of Alzheimer’s disease, she added.
The researchers plan to conduct subset analyses using apo E genetic status and age of death.
Although this is a “massive database,” it has limitations. For example, said Dr. Sajjadi, it does not reveal when patients started taking BP medication, how long they had been taking it, or why.
“We don’t know the exact the reason they were taking these medications. Was it just hypertension, or did they also have heart disease, stroke, a kidney problem, or was there another explanation,” he said.
Following the study presentation, session comoderator Krish Sathian, MBBS, PhD, professor of neurology, neural, and behavioral sciences, and psychology and director of the Neuroscience Institute, Penn State University, Hershey, called this work “fascinating. It provides a lot of data that really touches on everyday practice,” inasmuch as clinicians often prescribe antihypertensive medications and see patients with these kinds of brain disorders.
The investigators and Dr. Sathian reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ANA 2021
Alopecia tied to a threefold increased risk for dementia
Alopecia areata (AA) has been linked to a significantly increased risk for dementia, new research shows.
After controlling for an array of potential confounders, investigators found a threefold higher risk of developing any form of dementia and a fourfold higher risk of developing Alzheimer’s disease (AD) in those with AA versus the controls.
“AA shares a similar inflammatory signature with dementia and has great psychological impacts that lead to poor social engagement,” lead author Cheng-Yuan Li, MD, MSc, of the department of dermatology, Taipei (Taiwan) Veterans General Hospital.
“Poor social engagement and shared inflammatory cytokines might both be important links between AA and dementia,” said Dr. Li, who is also affiliated with the School of Medicine and the Institute of Brain Science at National Yang Ming Chiao Tung University, Taipei.
The study was published online Oct. 26, 2021, in the Journal of Clinical Psychiatry (doi: 10.4088/JCP.21m13931).
Significant psychological impact
Patients with AA often experience anxiety and depression, possibly caused by the negative emotional and psychological impact of the hair loss and partial or even complete baldness associated with the disease, the authors noted.
However, AA is also associated with an array of other atopic and autoimmune diseases, including psoriasis and systemic lupus erythematosus (SLE).
Epidemiologic research has suggested a link between dementia and autoimmune diseases such as psoriasis and SLE, with some evidence suggesting that autoimmune and inflammatory mechanisms may “play a role” in the development of AD.
Dementia in general and AD in particular, “have been shown to include an inflammatory component” that may share some of the same mediators seen in AA (eg, IL-1 beta, IL-6, and tumor necrosis factor–alpha).
Moreover, “the great negative psychosocial impact of AA might result in poor social engagement, a typical risk factor for dementia,” said Dr. Li. The investigators sought to investigate whether patients with AA actually do have a higher dementia risk than individuals without AA.
The researchers used data from the Taiwan National Health Insurance Research Database, comparing 2,534 patients with AA against 25,340 controls matched for age, sex, residence, income, dementia-related comorbidities, systemic steroid use, and annual outpatient visits. Participants were enrolled between 1998 and 2011 and followed to the end of 2013.
The mean age of the cohort was 53.9 years, and a little over half (57.6%) were female. The most common comorbidity was hypertension (32.3%), followed by dyslipidemia (27%) and diabetes (15.4%).
Dual intervention
After adjusting for potential confounders, those with AA were more likely to develop dementia, AD, and unspecified dementia, compared with controls. They also had a numerically higher risk for vascular dementia, compared with controls, but it was not statistically significant.
When participants were stratified by age, investigators found a significant association between AA and higher risk for any dementia as well as unspecified dementia in individuals of all ages and an increased risk for AD in patients with dementia age at onset of 65 years and older.
The mean age of dementia diagnosis was considerably younger in patients with AA versus controls (73.4 vs. 78.9 years, P = .002). The risk for any dementia and unspecified dementia was higher in patients of both sexes, but the risk for AD was higher only in male patients.
Sensitivity analyses that excluded the first year or first 3 years of observation yielded similar and consistent findings.
“Intervention targeting poor social engagement and inflammatory cytokines may be beneficial to AA-associated dementia,” said Dr. Li.
“Physicians should be more aware of this possible association, help reduce disease discrimination among the public, and encourage more social engagement for AA patients,” he said.
“Further studies are needed to elucidate the underlying pathophysiology between AA and dementia risk,” he added.
No cause and effect
Commenting on the study, Heather M. Snyder, PhD, vice president of medical and scientific affairs, Alzheimer’s Association, said, “We continue to learn about and better understand factors that may increase or decrease a person’s risk of dementia.”
“While we know the immune system plays a role in Alzheimer’s and other dementia, we are still investigating links between, and impact of, autoimmune diseases – like alopecia areata, rheumatoid arthritis, and others – on our overall health and our brains, [which] may eventually give us important information on risk reduction strategies as well,” said Dr. Snyder, who was not involved in the research.
She cautioned that although the study did show a correlation between AA and dementia risk, this does not equate to a demonstration of cause and effect.
At present, “the message for clinicians is that when a patient comes to your office with complaints about their memory, they should, No. 1, be taken seriously; and, No. 2, receive a thorough evaluation that takes into account the many factors that may lead to cognitive decline,” Dr. Snyder said.
The study was supported by a grant from Taipei Veterans General Hospital and the Ministry of Science and Technology, Taiwan. Dr. Li, coauthors, and Dr. Snyder disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Alopecia areata (AA) has been linked to a significantly increased risk for dementia, new research shows.
After controlling for an array of potential confounders, investigators found a threefold higher risk of developing any form of dementia and a fourfold higher risk of developing Alzheimer’s disease (AD) in those with AA versus the controls.
“AA shares a similar inflammatory signature with dementia and has great psychological impacts that lead to poor social engagement,” lead author Cheng-Yuan Li, MD, MSc, of the department of dermatology, Taipei (Taiwan) Veterans General Hospital.
“Poor social engagement and shared inflammatory cytokines might both be important links between AA and dementia,” said Dr. Li, who is also affiliated with the School of Medicine and the Institute of Brain Science at National Yang Ming Chiao Tung University, Taipei.
The study was published online Oct. 26, 2021, in the Journal of Clinical Psychiatry (doi: 10.4088/JCP.21m13931).
Significant psychological impact
Patients with AA often experience anxiety and depression, possibly caused by the negative emotional and psychological impact of the hair loss and partial or even complete baldness associated with the disease, the authors noted.
However, AA is also associated with an array of other atopic and autoimmune diseases, including psoriasis and systemic lupus erythematosus (SLE).
Epidemiologic research has suggested a link between dementia and autoimmune diseases such as psoriasis and SLE, with some evidence suggesting that autoimmune and inflammatory mechanisms may “play a role” in the development of AD.
Dementia in general and AD in particular, “have been shown to include an inflammatory component” that may share some of the same mediators seen in AA (eg, IL-1 beta, IL-6, and tumor necrosis factor–alpha).
Moreover, “the great negative psychosocial impact of AA might result in poor social engagement, a typical risk factor for dementia,” said Dr. Li. The investigators sought to investigate whether patients with AA actually do have a higher dementia risk than individuals without AA.
The researchers used data from the Taiwan National Health Insurance Research Database, comparing 2,534 patients with AA against 25,340 controls matched for age, sex, residence, income, dementia-related comorbidities, systemic steroid use, and annual outpatient visits. Participants were enrolled between 1998 and 2011 and followed to the end of 2013.
The mean age of the cohort was 53.9 years, and a little over half (57.6%) were female. The most common comorbidity was hypertension (32.3%), followed by dyslipidemia (27%) and diabetes (15.4%).
Dual intervention
After adjusting for potential confounders, those with AA were more likely to develop dementia, AD, and unspecified dementia, compared with controls. They also had a numerically higher risk for vascular dementia, compared with controls, but it was not statistically significant.
When participants were stratified by age, investigators found a significant association between AA and higher risk for any dementia as well as unspecified dementia in individuals of all ages and an increased risk for AD in patients with dementia age at onset of 65 years and older.
The mean age of dementia diagnosis was considerably younger in patients with AA versus controls (73.4 vs. 78.9 years, P = .002). The risk for any dementia and unspecified dementia was higher in patients of both sexes, but the risk for AD was higher only in male patients.
Sensitivity analyses that excluded the first year or first 3 years of observation yielded similar and consistent findings.
“Intervention targeting poor social engagement and inflammatory cytokines may be beneficial to AA-associated dementia,” said Dr. Li.
“Physicians should be more aware of this possible association, help reduce disease discrimination among the public, and encourage more social engagement for AA patients,” he said.
“Further studies are needed to elucidate the underlying pathophysiology between AA and dementia risk,” he added.
No cause and effect
Commenting on the study, Heather M. Snyder, PhD, vice president of medical and scientific affairs, Alzheimer’s Association, said, “We continue to learn about and better understand factors that may increase or decrease a person’s risk of dementia.”
“While we know the immune system plays a role in Alzheimer’s and other dementia, we are still investigating links between, and impact of, autoimmune diseases – like alopecia areata, rheumatoid arthritis, and others – on our overall health and our brains, [which] may eventually give us important information on risk reduction strategies as well,” said Dr. Snyder, who was not involved in the research.
She cautioned that although the study did show a correlation between AA and dementia risk, this does not equate to a demonstration of cause and effect.
At present, “the message for clinicians is that when a patient comes to your office with complaints about their memory, they should, No. 1, be taken seriously; and, No. 2, receive a thorough evaluation that takes into account the many factors that may lead to cognitive decline,” Dr. Snyder said.
The study was supported by a grant from Taipei Veterans General Hospital and the Ministry of Science and Technology, Taiwan. Dr. Li, coauthors, and Dr. Snyder disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Alopecia areata (AA) has been linked to a significantly increased risk for dementia, new research shows.
After controlling for an array of potential confounders, investigators found a threefold higher risk of developing any form of dementia and a fourfold higher risk of developing Alzheimer’s disease (AD) in those with AA versus the controls.
“AA shares a similar inflammatory signature with dementia and has great psychological impacts that lead to poor social engagement,” lead author Cheng-Yuan Li, MD, MSc, of the department of dermatology, Taipei (Taiwan) Veterans General Hospital.
“Poor social engagement and shared inflammatory cytokines might both be important links between AA and dementia,” said Dr. Li, who is also affiliated with the School of Medicine and the Institute of Brain Science at National Yang Ming Chiao Tung University, Taipei.
The study was published online Oct. 26, 2021, in the Journal of Clinical Psychiatry (doi: 10.4088/JCP.21m13931).
Significant psychological impact
Patients with AA often experience anxiety and depression, possibly caused by the negative emotional and psychological impact of the hair loss and partial or even complete baldness associated with the disease, the authors noted.
However, AA is also associated with an array of other atopic and autoimmune diseases, including psoriasis and systemic lupus erythematosus (SLE).
Epidemiologic research has suggested a link between dementia and autoimmune diseases such as psoriasis and SLE, with some evidence suggesting that autoimmune and inflammatory mechanisms may “play a role” in the development of AD.
Dementia in general and AD in particular, “have been shown to include an inflammatory component” that may share some of the same mediators seen in AA (eg, IL-1 beta, IL-6, and tumor necrosis factor–alpha).
Moreover, “the great negative psychosocial impact of AA might result in poor social engagement, a typical risk factor for dementia,” said Dr. Li. The investigators sought to investigate whether patients with AA actually do have a higher dementia risk than individuals without AA.
The researchers used data from the Taiwan National Health Insurance Research Database, comparing 2,534 patients with AA against 25,340 controls matched for age, sex, residence, income, dementia-related comorbidities, systemic steroid use, and annual outpatient visits. Participants were enrolled between 1998 and 2011 and followed to the end of 2013.
The mean age of the cohort was 53.9 years, and a little over half (57.6%) were female. The most common comorbidity was hypertension (32.3%), followed by dyslipidemia (27%) and diabetes (15.4%).
Dual intervention
After adjusting for potential confounders, those with AA were more likely to develop dementia, AD, and unspecified dementia, compared with controls. They also had a numerically higher risk for vascular dementia, compared with controls, but it was not statistically significant.
When participants were stratified by age, investigators found a significant association between AA and higher risk for any dementia as well as unspecified dementia in individuals of all ages and an increased risk for AD in patients with dementia age at onset of 65 years and older.
The mean age of dementia diagnosis was considerably younger in patients with AA versus controls (73.4 vs. 78.9 years, P = .002). The risk for any dementia and unspecified dementia was higher in patients of both sexes, but the risk for AD was higher only in male patients.
Sensitivity analyses that excluded the first year or first 3 years of observation yielded similar and consistent findings.
“Intervention targeting poor social engagement and inflammatory cytokines may be beneficial to AA-associated dementia,” said Dr. Li.
“Physicians should be more aware of this possible association, help reduce disease discrimination among the public, and encourage more social engagement for AA patients,” he said.
“Further studies are needed to elucidate the underlying pathophysiology between AA and dementia risk,” he added.
No cause and effect
Commenting on the study, Heather M. Snyder, PhD, vice president of medical and scientific affairs, Alzheimer’s Association, said, “We continue to learn about and better understand factors that may increase or decrease a person’s risk of dementia.”
“While we know the immune system plays a role in Alzheimer’s and other dementia, we are still investigating links between, and impact of, autoimmune diseases – like alopecia areata, rheumatoid arthritis, and others – on our overall health and our brains, [which] may eventually give us important information on risk reduction strategies as well,” said Dr. Snyder, who was not involved in the research.
She cautioned that although the study did show a correlation between AA and dementia risk, this does not equate to a demonstration of cause and effect.
At present, “the message for clinicians is that when a patient comes to your office with complaints about their memory, they should, No. 1, be taken seriously; and, No. 2, receive a thorough evaluation that takes into account the many factors that may lead to cognitive decline,” Dr. Snyder said.
The study was supported by a grant from Taipei Veterans General Hospital and the Ministry of Science and Technology, Taiwan. Dr. Li, coauthors, and Dr. Snyder disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FDA not recognizing efficacy of psychopharmacologic therapies
Many years ago, drug development in psychiatry turned to control of specific symptoms across disorders rather than within disorders, but regulatory agencies are still not yet on board, according to an expert psychopharmacologist outlining the ongoing evolution at the virtual Psychopharmacology Update presented by Current Psychiatry and the American Academy of Clinical Psychiatrists, sponsored by Medscape Live.
If this reorientation is going to lead to the broad indications the newer drugs likely deserve, which is control of specific types of symptoms regardless of the diagnosis, “we have to move the [Food and Drug Administration] along,” said Stephen M. Stahl, MD, PhD, chairman of the Neuroscience Institute and an adjunct professor of psychiatry at the University of California, San Diego.
On the side of drug development and clinical practice, the reorientation has already taken place. Dr. Stahl described numerous brain circuits known to produce symptoms when function is altered that are now treatment targets. This includes the ventral medial prefrontal cortex where deficient information processing leads to depression and the orbital frontal cortex where altered function leads to impulsivity.
“It is not like each part of the brain does a little bit of everything. Rather, each part of the brain has an assignment and duty and function,” Dr. Stahl explained. By addressing the disturbed signaling in brain circuits that lead to depression, impulsivity, agitation, or other symptoms, there is an opportunity for control, regardless of the psychiatric diagnosis with which the symptom is associated.
For example, Dr. Stahl predicted that pimavanserin, a highly selective 5-HT2A inverse agonist that is already approved for psychosis in Parkinson’s disease, is now likely to be approved for psychosis associated with other conditions on the basis of recent positive clinical studies in these other disorders.
Brexpiprazole, a serotonin-dopamine activity modulator already known to be useful for control of the agitation characteristic of schizophrenia, is now showing the same type of activity against agitation when it is associated with Alzheimer’s disease. Again, Dr. Stahl thinks this drug is on course for an indication across diseases once studies are conducted in each disease individually.
Another drug being evaluated for agitation, the N-methyl-D-aspartate receptor antagonist dextromethorphan bupropion, is also being tested for treatment of symptoms across multiple disorders, he reported.
However, the FDA has so far taken the position that each drug must be tested separately for a given symptom in each disorder for which it is being considered despite the underlying premise that it is the symptom, not the disease, that is important.
Unlike physiological diseases where symptoms, like a fever or abdominal cramps, are the product of a disease, psychiatric symptoms are the disease and a fundamental target – regardless of the DSM-based diagnosis.
To some degree, the symptoms of psychiatric disorders have always been the focus of treatment, but a pivot toward developing therapies that will control a symptom regardless of the underlying diagnosis is an important conceptual change. It is being made possible by advances in the detail with which the neuropathology of these symptoms is understood .
“By my count, 79 symptoms are described in DSM-5, but they are spread across hundreds of syndromes because they are grouped together in different ways,” Dr. Stahl observed.
He noted that clinicians make a diagnosis on the basis symptom groupings, but their interventions are selected to address the manifestations of the disease, not the disease itself.
“If you are a real psychopharmacologist treating real patients, you are treating the specific symptoms of the specific patient,” according to Dr. Stahl.
So far, the FDA has not made this leap, insisting on trials in these categorical disorders rather than permitting trial designs that allow benefit to be demonstrated against a symptom regardless of the syndrome with which it is associated.
Of egregious examples, Dr. Stahl recounted a recent trial of a 5-HT2 antagonist that looked so promising against psychosis in Alzheimer’s disease that the trialists enrolled patients with psychosis regardless of type of dementia, such as vascular dementia and Lewy body disease. The efficacy was impressive.
“It worked so well that they stopped the trial, but the FDA declined to approve it,” Dr. Stahl recounted. Despite clear evidence of benefit, the regulators insisted that the investigators needed to show a significant benefit in each condition individually.
While the trial investigators acknowledged that there was not enough power in the trial to show a statistically significant benefit in each category, they argued that the overall benefit and the consistent response across categories required them to stop the trial for ethical reasons.
“That’s your problem, the FDA said to the investigators,” according to Dr. Stahl.
The failure of the FDA to recognize the efficacy of psychopharmacologic therapies across symptoms regardless of the associated disease is a failure to stay current with an important evolution in medicine, Dr. Stahl indicated.
“What we have come to understand is the neurobiology of any given symptom is likely to be the same across disorders,” he said.
Agency’s arbitrary decisions cited
“I completely agree with Dr. Stahl,” said Henry A. Nasrallah, MD, professor of psychiatry, neurology, and neuroscience, University of Cincinnati.
In addition to the fact that symptoms are present across multiple categories, many patients manifest multiple symptoms at one time, Dr. Nasrallah pointed out. For neurodegenerative disorders associated with psychosis, depression, anxiety, aggression, and other symptoms, it is already well known that the heterogeneous symptoms “cannot be treated with a single drug,” he said. Rather different drugs targeting each symptom individually is essential for effective management.
Dr. Nasrallah, who chaired the Psychopharmacology Update meeting, has made this point many times in the past, including in his role as the editor of Current Psychiatry. In one editorial 10 years ago, he wrote that “it makes little sense for the FDA to mandate that a drug must work for a DSM diagnosis instead of specific symptoms.”
“The FDA must update its old policy, which has led to the widespread off-label use of psychiatric drugs, an artificial concept, simply because the FDA arbitrarily decided a long time ago that new drugs must be approved for a specific DSM diagnosis,” Dr. Nasrallah said.
Dr. Stahl reported financial relationships with more than 20 pharmaceutical companies, including those that are involved in the development of drugs included in his talk. Medscape Live and this news organization are owned by the same parent company.
Many years ago, drug development in psychiatry turned to control of specific symptoms across disorders rather than within disorders, but regulatory agencies are still not yet on board, according to an expert psychopharmacologist outlining the ongoing evolution at the virtual Psychopharmacology Update presented by Current Psychiatry and the American Academy of Clinical Psychiatrists, sponsored by Medscape Live.
If this reorientation is going to lead to the broad indications the newer drugs likely deserve, which is control of specific types of symptoms regardless of the diagnosis, “we have to move the [Food and Drug Administration] along,” said Stephen M. Stahl, MD, PhD, chairman of the Neuroscience Institute and an adjunct professor of psychiatry at the University of California, San Diego.
On the side of drug development and clinical practice, the reorientation has already taken place. Dr. Stahl described numerous brain circuits known to produce symptoms when function is altered that are now treatment targets. This includes the ventral medial prefrontal cortex where deficient information processing leads to depression and the orbital frontal cortex where altered function leads to impulsivity.
“It is not like each part of the brain does a little bit of everything. Rather, each part of the brain has an assignment and duty and function,” Dr. Stahl explained. By addressing the disturbed signaling in brain circuits that lead to depression, impulsivity, agitation, or other symptoms, there is an opportunity for control, regardless of the psychiatric diagnosis with which the symptom is associated.
For example, Dr. Stahl predicted that pimavanserin, a highly selective 5-HT2A inverse agonist that is already approved for psychosis in Parkinson’s disease, is now likely to be approved for psychosis associated with other conditions on the basis of recent positive clinical studies in these other disorders.
Brexpiprazole, a serotonin-dopamine activity modulator already known to be useful for control of the agitation characteristic of schizophrenia, is now showing the same type of activity against agitation when it is associated with Alzheimer’s disease. Again, Dr. Stahl thinks this drug is on course for an indication across diseases once studies are conducted in each disease individually.
Another drug being evaluated for agitation, the N-methyl-D-aspartate receptor antagonist dextromethorphan bupropion, is also being tested for treatment of symptoms across multiple disorders, he reported.
However, the FDA has so far taken the position that each drug must be tested separately for a given symptom in each disorder for which it is being considered despite the underlying premise that it is the symptom, not the disease, that is important.
Unlike physiological diseases where symptoms, like a fever or abdominal cramps, are the product of a disease, psychiatric symptoms are the disease and a fundamental target – regardless of the DSM-based diagnosis.
To some degree, the symptoms of psychiatric disorders have always been the focus of treatment, but a pivot toward developing therapies that will control a symptom regardless of the underlying diagnosis is an important conceptual change. It is being made possible by advances in the detail with which the neuropathology of these symptoms is understood .
“By my count, 79 symptoms are described in DSM-5, but they are spread across hundreds of syndromes because they are grouped together in different ways,” Dr. Stahl observed.
He noted that clinicians make a diagnosis on the basis symptom groupings, but their interventions are selected to address the manifestations of the disease, not the disease itself.
“If you are a real psychopharmacologist treating real patients, you are treating the specific symptoms of the specific patient,” according to Dr. Stahl.
So far, the FDA has not made this leap, insisting on trials in these categorical disorders rather than permitting trial designs that allow benefit to be demonstrated against a symptom regardless of the syndrome with which it is associated.
Of egregious examples, Dr. Stahl recounted a recent trial of a 5-HT2 antagonist that looked so promising against psychosis in Alzheimer’s disease that the trialists enrolled patients with psychosis regardless of type of dementia, such as vascular dementia and Lewy body disease. The efficacy was impressive.
“It worked so well that they stopped the trial, but the FDA declined to approve it,” Dr. Stahl recounted. Despite clear evidence of benefit, the regulators insisted that the investigators needed to show a significant benefit in each condition individually.
While the trial investigators acknowledged that there was not enough power in the trial to show a statistically significant benefit in each category, they argued that the overall benefit and the consistent response across categories required them to stop the trial for ethical reasons.
“That’s your problem, the FDA said to the investigators,” according to Dr. Stahl.
The failure of the FDA to recognize the efficacy of psychopharmacologic therapies across symptoms regardless of the associated disease is a failure to stay current with an important evolution in medicine, Dr. Stahl indicated.
“What we have come to understand is the neurobiology of any given symptom is likely to be the same across disorders,” he said.
Agency’s arbitrary decisions cited
“I completely agree with Dr. Stahl,” said Henry A. Nasrallah, MD, professor of psychiatry, neurology, and neuroscience, University of Cincinnati.
In addition to the fact that symptoms are present across multiple categories, many patients manifest multiple symptoms at one time, Dr. Nasrallah pointed out. For neurodegenerative disorders associated with psychosis, depression, anxiety, aggression, and other symptoms, it is already well known that the heterogeneous symptoms “cannot be treated with a single drug,” he said. Rather different drugs targeting each symptom individually is essential for effective management.
Dr. Nasrallah, who chaired the Psychopharmacology Update meeting, has made this point many times in the past, including in his role as the editor of Current Psychiatry. In one editorial 10 years ago, he wrote that “it makes little sense for the FDA to mandate that a drug must work for a DSM diagnosis instead of specific symptoms.”
“The FDA must update its old policy, which has led to the widespread off-label use of psychiatric drugs, an artificial concept, simply because the FDA arbitrarily decided a long time ago that new drugs must be approved for a specific DSM diagnosis,” Dr. Nasrallah said.
Dr. Stahl reported financial relationships with more than 20 pharmaceutical companies, including those that are involved in the development of drugs included in his talk. Medscape Live and this news organization are owned by the same parent company.
Many years ago, drug development in psychiatry turned to control of specific symptoms across disorders rather than within disorders, but regulatory agencies are still not yet on board, according to an expert psychopharmacologist outlining the ongoing evolution at the virtual Psychopharmacology Update presented by Current Psychiatry and the American Academy of Clinical Psychiatrists, sponsored by Medscape Live.
If this reorientation is going to lead to the broad indications the newer drugs likely deserve, which is control of specific types of symptoms regardless of the diagnosis, “we have to move the [Food and Drug Administration] along,” said Stephen M. Stahl, MD, PhD, chairman of the Neuroscience Institute and an adjunct professor of psychiatry at the University of California, San Diego.
On the side of drug development and clinical practice, the reorientation has already taken place. Dr. Stahl described numerous brain circuits known to produce symptoms when function is altered that are now treatment targets. This includes the ventral medial prefrontal cortex where deficient information processing leads to depression and the orbital frontal cortex where altered function leads to impulsivity.
“It is not like each part of the brain does a little bit of everything. Rather, each part of the brain has an assignment and duty and function,” Dr. Stahl explained. By addressing the disturbed signaling in brain circuits that lead to depression, impulsivity, agitation, or other symptoms, there is an opportunity for control, regardless of the psychiatric diagnosis with which the symptom is associated.
For example, Dr. Stahl predicted that pimavanserin, a highly selective 5-HT2A inverse agonist that is already approved for psychosis in Parkinson’s disease, is now likely to be approved for psychosis associated with other conditions on the basis of recent positive clinical studies in these other disorders.
Brexpiprazole, a serotonin-dopamine activity modulator already known to be useful for control of the agitation characteristic of schizophrenia, is now showing the same type of activity against agitation when it is associated with Alzheimer’s disease. Again, Dr. Stahl thinks this drug is on course for an indication across diseases once studies are conducted in each disease individually.
Another drug being evaluated for agitation, the N-methyl-D-aspartate receptor antagonist dextromethorphan bupropion, is also being tested for treatment of symptoms across multiple disorders, he reported.
However, the FDA has so far taken the position that each drug must be tested separately for a given symptom in each disorder for which it is being considered despite the underlying premise that it is the symptom, not the disease, that is important.
Unlike physiological diseases where symptoms, like a fever or abdominal cramps, are the product of a disease, psychiatric symptoms are the disease and a fundamental target – regardless of the DSM-based diagnosis.
To some degree, the symptoms of psychiatric disorders have always been the focus of treatment, but a pivot toward developing therapies that will control a symptom regardless of the underlying diagnosis is an important conceptual change. It is being made possible by advances in the detail with which the neuropathology of these symptoms is understood .
“By my count, 79 symptoms are described in DSM-5, but they are spread across hundreds of syndromes because they are grouped together in different ways,” Dr. Stahl observed.
He noted that clinicians make a diagnosis on the basis symptom groupings, but their interventions are selected to address the manifestations of the disease, not the disease itself.
“If you are a real psychopharmacologist treating real patients, you are treating the specific symptoms of the specific patient,” according to Dr. Stahl.
So far, the FDA has not made this leap, insisting on trials in these categorical disorders rather than permitting trial designs that allow benefit to be demonstrated against a symptom regardless of the syndrome with which it is associated.
Of egregious examples, Dr. Stahl recounted a recent trial of a 5-HT2 antagonist that looked so promising against psychosis in Alzheimer’s disease that the trialists enrolled patients with psychosis regardless of type of dementia, such as vascular dementia and Lewy body disease. The efficacy was impressive.
“It worked so well that they stopped the trial, but the FDA declined to approve it,” Dr. Stahl recounted. Despite clear evidence of benefit, the regulators insisted that the investigators needed to show a significant benefit in each condition individually.
While the trial investigators acknowledged that there was not enough power in the trial to show a statistically significant benefit in each category, they argued that the overall benefit and the consistent response across categories required them to stop the trial for ethical reasons.
“That’s your problem, the FDA said to the investigators,” according to Dr. Stahl.
The failure of the FDA to recognize the efficacy of psychopharmacologic therapies across symptoms regardless of the associated disease is a failure to stay current with an important evolution in medicine, Dr. Stahl indicated.
“What we have come to understand is the neurobiology of any given symptom is likely to be the same across disorders,” he said.
Agency’s arbitrary decisions cited
“I completely agree with Dr. Stahl,” said Henry A. Nasrallah, MD, professor of psychiatry, neurology, and neuroscience, University of Cincinnati.
In addition to the fact that symptoms are present across multiple categories, many patients manifest multiple symptoms at one time, Dr. Nasrallah pointed out. For neurodegenerative disorders associated with psychosis, depression, anxiety, aggression, and other symptoms, it is already well known that the heterogeneous symptoms “cannot be treated with a single drug,” he said. Rather different drugs targeting each symptom individually is essential for effective management.
Dr. Nasrallah, who chaired the Psychopharmacology Update meeting, has made this point many times in the past, including in his role as the editor of Current Psychiatry. In one editorial 10 years ago, he wrote that “it makes little sense for the FDA to mandate that a drug must work for a DSM diagnosis instead of specific symptoms.”
“The FDA must update its old policy, which has led to the widespread off-label use of psychiatric drugs, an artificial concept, simply because the FDA arbitrarily decided a long time ago that new drugs must be approved for a specific DSM diagnosis,” Dr. Nasrallah said.
Dr. Stahl reported financial relationships with more than 20 pharmaceutical companies, including those that are involved in the development of drugs included in his talk. Medscape Live and this news organization are owned by the same parent company.
FROM PSYCHOPHARMACOLOGY UPDATE
Patients given NSAIDs over antiemetics for headaches spend less time in the ED
based on data from approximately 7,000 patients.
Headache is the fourth-most common chief complaint in the ED, accounting for approximately 3% of all ED visits, said Philip Wang, a medical student at the Cleveland Clinic, in a presentation at the annual meeting of the American College of Emergency Physicians.
A variety of pharmacotherapies are used to manage headache, which leads to a range of resource use, he said.
To understand the association between route of drug administration and length of ED stay, Mr. Wang and colleagues reviewed data from 7,233 visits by 6,715 patients at any of the 21 Cleveland Clinic Health System EDs in 2018 with headache as the primary discharge diagnosis. Patients admitted to the hospital were excluded; those treated with opioids, antiemetics, and/or NSAIDs were included. The average age of the study population was 31 years, 57% were White, and approximately half were Medicaid or Medicare patients.
Approximately 68% of patients received antiemetics, 66.8% received NSAIDs, and 9.8% received opioids. Approximately 42% of patients received parenteral-only treatment and 42% received oral-only treatment; 15% received mixed treatment. The average length of ED stay was 202 minutes.
In a multivariate analysis adjusted for sex, age, income, race, insurance status, ED type, and arrival time, treatment with oral drugs only was associated with an 11% reduction of length of stay, compared with treatment with parenteral medication only (P < .001). However, the length of stay for patients treated with mixed route of administration was 10% longer, compared with parenteral only (P < .001).
In terms of drug class (a secondary outcome), patients treated with opioids had a 10% increase in length of stay (P < .01) and those treated with antiemetics had a 14% increase in length of stay; however, patients treated with NSAIDs had a 7% decrease in length of stay.
The study findings were limited in part by the challenge of isolating patients presenting with a primary headache diagnosis, Mr. Wang noted in the presentation.
The challenge of controlling for all the potential factors impacting length of stay, which is “provider, resource, and situation dependent,” is an additional limitation, he said.
However, the results show that route of administration has a significant impact on length of ED stay in patients presenting with headache, he concluded.
The study received no outside funding. The researchers disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
based on data from approximately 7,000 patients.
Headache is the fourth-most common chief complaint in the ED, accounting for approximately 3% of all ED visits, said Philip Wang, a medical student at the Cleveland Clinic, in a presentation at the annual meeting of the American College of Emergency Physicians.
A variety of pharmacotherapies are used to manage headache, which leads to a range of resource use, he said.
To understand the association between route of drug administration and length of ED stay, Mr. Wang and colleagues reviewed data from 7,233 visits by 6,715 patients at any of the 21 Cleveland Clinic Health System EDs in 2018 with headache as the primary discharge diagnosis. Patients admitted to the hospital were excluded; those treated with opioids, antiemetics, and/or NSAIDs were included. The average age of the study population was 31 years, 57% were White, and approximately half were Medicaid or Medicare patients.
Approximately 68% of patients received antiemetics, 66.8% received NSAIDs, and 9.8% received opioids. Approximately 42% of patients received parenteral-only treatment and 42% received oral-only treatment; 15% received mixed treatment. The average length of ED stay was 202 minutes.
In a multivariate analysis adjusted for sex, age, income, race, insurance status, ED type, and arrival time, treatment with oral drugs only was associated with an 11% reduction of length of stay, compared with treatment with parenteral medication only (P < .001). However, the length of stay for patients treated with mixed route of administration was 10% longer, compared with parenteral only (P < .001).
In terms of drug class (a secondary outcome), patients treated with opioids had a 10% increase in length of stay (P < .01) and those treated with antiemetics had a 14% increase in length of stay; however, patients treated with NSAIDs had a 7% decrease in length of stay.
The study findings were limited in part by the challenge of isolating patients presenting with a primary headache diagnosis, Mr. Wang noted in the presentation.
The challenge of controlling for all the potential factors impacting length of stay, which is “provider, resource, and situation dependent,” is an additional limitation, he said.
However, the results show that route of administration has a significant impact on length of ED stay in patients presenting with headache, he concluded.
The study received no outside funding. The researchers disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
based on data from approximately 7,000 patients.
Headache is the fourth-most common chief complaint in the ED, accounting for approximately 3% of all ED visits, said Philip Wang, a medical student at the Cleveland Clinic, in a presentation at the annual meeting of the American College of Emergency Physicians.
A variety of pharmacotherapies are used to manage headache, which leads to a range of resource use, he said.
To understand the association between route of drug administration and length of ED stay, Mr. Wang and colleagues reviewed data from 7,233 visits by 6,715 patients at any of the 21 Cleveland Clinic Health System EDs in 2018 with headache as the primary discharge diagnosis. Patients admitted to the hospital were excluded; those treated with opioids, antiemetics, and/or NSAIDs were included. The average age of the study population was 31 years, 57% were White, and approximately half were Medicaid or Medicare patients.
Approximately 68% of patients received antiemetics, 66.8% received NSAIDs, and 9.8% received opioids. Approximately 42% of patients received parenteral-only treatment and 42% received oral-only treatment; 15% received mixed treatment. The average length of ED stay was 202 minutes.
In a multivariate analysis adjusted for sex, age, income, race, insurance status, ED type, and arrival time, treatment with oral drugs only was associated with an 11% reduction of length of stay, compared with treatment with parenteral medication only (P < .001). However, the length of stay for patients treated with mixed route of administration was 10% longer, compared with parenteral only (P < .001).
In terms of drug class (a secondary outcome), patients treated with opioids had a 10% increase in length of stay (P < .01) and those treated with antiemetics had a 14% increase in length of stay; however, patients treated with NSAIDs had a 7% decrease in length of stay.
The study findings were limited in part by the challenge of isolating patients presenting with a primary headache diagnosis, Mr. Wang noted in the presentation.
The challenge of controlling for all the potential factors impacting length of stay, which is “provider, resource, and situation dependent,” is an additional limitation, he said.
However, the results show that route of administration has a significant impact on length of ED stay in patients presenting with headache, he concluded.
The study received no outside funding. The researchers disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Influenza tied to long-term increased risk for Parkinson’s disease
Influenza infection is linked to a subsequent diagnosis of Parkinson’s disease (PD) more than 10 years later, resurfacing a long-held debate about whether infection increases the risk for movement disorders over the long term.
In a large case-control study, investigators found and by more than 70% for PD occurring more than 10 years after the flu.
“This study is not definitive by any means, but it certainly suggests there are potential long-term consequences from influenza,” study investigator Noelle M. Cocoros, DSc, research scientist at Harvard Pilgrim Health Care Institute and Harvard Medical School, Boston, said in an interview.
The study was published online Oct. 25 in JAMA Neurology.
Ongoing debate
The debate about whether influenza is associated with PD has been going on as far back as the 1918 influenza pandemic, when experts documented parkinsonism in affected individuals.
Using data from the Danish patient registry, researchers identified 10,271 subjects diagnosed with PD during a 17-year period (2000-2016). Of these, 38.7% were female, and the mean age was 71.4 years.
They matched these subjects for age and sex to 51,355 controls without PD. Compared with controls, slightly fewer individuals with PD had chronic obstructive pulmonary disease (COPD) or emphysema, but there was a similar distribution of cardiovascular disease and various other conditions.
Researchers collected data on influenza diagnoses from inpatient and outpatient hospital clinics from 1977 to 2016. They plotted these by month and year on a graph, calculated the median number of diagnoses per month, and identified peaks as those with more than threefold the median.
They categorized cases in groups related to the time between the infection and PD: More than 10 years, 10-15 years, and more than 15 years.
The time lapse accounts for a rather long “run-up” to PD, said Dr. Cocoros. There’s a sometimes decades-long preclinical phase before patients develop typical motor signs and a prodromal phase where they may present with nonmotor symptoms such as sleep disorders and constipation.
“We expected there would be at least 10 years between any infection and PD if there was an association present,” said Dr. Cocoros.
Investigators found an association between influenza exposure and PD diagnosis “that held up over time,” she said.
For more than 10 years before PD, the likelihood of a diagnosis for the infected compared with the unexposed was increased 73% (odds ratio [OR] 1.73; 95% confidence interval, 1.11-2.71; P = .02) after adjustment for cardiovascular disease, diabetes, chronic obstructive pulmonary disease, emphysema, lung cancer, Crohn’s disease, and ulcerative colitis.
The odds increased with more time from infection. For more than 15 years, the adjusted OR was 1.91 (95% CI, 1.14 - 3.19; P =.01).
However, for the 10- to 15-year time frame, the point estimate was reduced and the CI nonsignificant (OR, 1.33; 95% CI, 0.54-3.27; P = .53). This “is a little hard to interpret,” but could be a result of the small numbers, exposure misclassification, or because “the longer time interval is what’s meaningful,” said Dr. Cocoros.
Potential COVID-19–related PD surge?
In a sensitivity analysis, researchers looked at peak infection activity. “We wanted to increase the likelihood of these diagnoses representing actual infection,” Dr. Cocoros noted.
Here, the OR was still elevated at more than 10 years, but the CI was quite wide and included 1 (OR, 1.52; 95% CI, 0.80-2.89; P = .21). “So the association holds up, but the estimates are quite unstable,” said Dr. Cocoros.
Researchers examined associations with numerous other infection types, but did not see the same trend over time. Some infections – for example, gastrointestinal infections and septicemia – were associated with PD within 5 years, but most associations appeared to be null after more than 10 years.
“There seemed to be associations earlier between the infection and PD, which we interpret to suggest there’s actually not a meaningful association,” said Dr. Cocoros.
An exception might be urinary tract infections (UTIs), where after 10 years, the adjusted OR was 1.19 (95% CI, 1.01-1.40). Research suggests patients with PD often have UTIs and neurogenic bladder.
“It’s possible that UTIs could be an early symptom of PD rather than a causative factor,” said Dr. Cocoros.
It’s unclear how influenza might lead to PD but it could be that the virus gets into the central nervous system, resulting in neuroinflammation. Cytokines generated in response to the influenza infection might damage the brain.
“The infection could be a ‘primer’ or an initial ‘hit’ to the system, maybe setting people up for PD,” said Dr. Cocoros.
As for the current COVID-19 pandemic, some experts are concerned about a potential surge in PD cases in decades to come, and are calling for prospective monitoring of patients with this infection, said Dr. Cocoros.
However, she noted that infections don’t account for all PD cases and that genetic and environmental factors also influence risk.
Many individuals who contract influenza don’t seek medical care or get tested, so it’s possible the study counted those who had the infection as unexposed. Another potential study limitation was that small numbers for some infections, for example, Helicobacter pylori and hepatitis C, limited the ability to interpret results.
‘Exciting and important’ findings
Commenting on the research for this news organization, Aparna Wagle Shukla, MD, professor, Norman Fixel Institute for Neurological Diseases, University of Florida, Gainesville, said the results amid the current pandemic are “exciting and important” and “have reinvigorated interest” in the role of infection in PD.
However, the study had some limitations, an important one being lack of accounting for confounding factors, including environmental factors, she said. Exposure to pesticides, living in a rural area, drinking well water, and having had a head injury may increase PD risk, whereas high intake of caffeine, nicotine, alcohol, and nonsteroidal anti-inflammatory drugs might lower the risk.
The researchers did not take into account exposure to multiple microbes or “infection burden,” said Dr. Wagle Shukla, who was not involved in the current study. In addition, as the data are from a single country with exposure to specific influenza strains, application of the findings elsewhere may be limited.
Dr. Wagle Shukla noted that a case-control design “isn’t ideal” from an epidemiological perspective. “Future studies should involve large cohorts followed longitudinally.”
The study was supported by grants from the Lundbeck Foundation and the Augustinus Foundation. Dr. Cocoros has disclosed no relevant financial relationships. Several coauthors have disclosed relationships with industry. The full list can be found with the original article.
A version of this article first appeared on Medscape.com.
Influenza infection is linked to a subsequent diagnosis of Parkinson’s disease (PD) more than 10 years later, resurfacing a long-held debate about whether infection increases the risk for movement disorders over the long term.
In a large case-control study, investigators found and by more than 70% for PD occurring more than 10 years after the flu.
“This study is not definitive by any means, but it certainly suggests there are potential long-term consequences from influenza,” study investigator Noelle M. Cocoros, DSc, research scientist at Harvard Pilgrim Health Care Institute and Harvard Medical School, Boston, said in an interview.
The study was published online Oct. 25 in JAMA Neurology.
Ongoing debate
The debate about whether influenza is associated with PD has been going on as far back as the 1918 influenza pandemic, when experts documented parkinsonism in affected individuals.
Using data from the Danish patient registry, researchers identified 10,271 subjects diagnosed with PD during a 17-year period (2000-2016). Of these, 38.7% were female, and the mean age was 71.4 years.
They matched these subjects for age and sex to 51,355 controls without PD. Compared with controls, slightly fewer individuals with PD had chronic obstructive pulmonary disease (COPD) or emphysema, but there was a similar distribution of cardiovascular disease and various other conditions.
Researchers collected data on influenza diagnoses from inpatient and outpatient hospital clinics from 1977 to 2016. They plotted these by month and year on a graph, calculated the median number of diagnoses per month, and identified peaks as those with more than threefold the median.
They categorized cases in groups related to the time between the infection and PD: More than 10 years, 10-15 years, and more than 15 years.
The time lapse accounts for a rather long “run-up” to PD, said Dr. Cocoros. There’s a sometimes decades-long preclinical phase before patients develop typical motor signs and a prodromal phase where they may present with nonmotor symptoms such as sleep disorders and constipation.
“We expected there would be at least 10 years between any infection and PD if there was an association present,” said Dr. Cocoros.
Investigators found an association between influenza exposure and PD diagnosis “that held up over time,” she said.
For more than 10 years before PD, the likelihood of a diagnosis for the infected compared with the unexposed was increased 73% (odds ratio [OR] 1.73; 95% confidence interval, 1.11-2.71; P = .02) after adjustment for cardiovascular disease, diabetes, chronic obstructive pulmonary disease, emphysema, lung cancer, Crohn’s disease, and ulcerative colitis.
The odds increased with more time from infection. For more than 15 years, the adjusted OR was 1.91 (95% CI, 1.14 - 3.19; P =.01).
However, for the 10- to 15-year time frame, the point estimate was reduced and the CI nonsignificant (OR, 1.33; 95% CI, 0.54-3.27; P = .53). This “is a little hard to interpret,” but could be a result of the small numbers, exposure misclassification, or because “the longer time interval is what’s meaningful,” said Dr. Cocoros.
Potential COVID-19–related PD surge?
In a sensitivity analysis, researchers looked at peak infection activity. “We wanted to increase the likelihood of these diagnoses representing actual infection,” Dr. Cocoros noted.
Here, the OR was still elevated at more than 10 years, but the CI was quite wide and included 1 (OR, 1.52; 95% CI, 0.80-2.89; P = .21). “So the association holds up, but the estimates are quite unstable,” said Dr. Cocoros.
Researchers examined associations with numerous other infection types, but did not see the same trend over time. Some infections – for example, gastrointestinal infections and septicemia – were associated with PD within 5 years, but most associations appeared to be null after more than 10 years.
“There seemed to be associations earlier between the infection and PD, which we interpret to suggest there’s actually not a meaningful association,” said Dr. Cocoros.
An exception might be urinary tract infections (UTIs), where after 10 years, the adjusted OR was 1.19 (95% CI, 1.01-1.40). Research suggests patients with PD often have UTIs and neurogenic bladder.
“It’s possible that UTIs could be an early symptom of PD rather than a causative factor,” said Dr. Cocoros.
It’s unclear how influenza might lead to PD but it could be that the virus gets into the central nervous system, resulting in neuroinflammation. Cytokines generated in response to the influenza infection might damage the brain.
“The infection could be a ‘primer’ or an initial ‘hit’ to the system, maybe setting people up for PD,” said Dr. Cocoros.
As for the current COVID-19 pandemic, some experts are concerned about a potential surge in PD cases in decades to come, and are calling for prospective monitoring of patients with this infection, said Dr. Cocoros.
However, she noted that infections don’t account for all PD cases and that genetic and environmental factors also influence risk.
Many individuals who contract influenza don’t seek medical care or get tested, so it’s possible the study counted those who had the infection as unexposed. Another potential study limitation was that small numbers for some infections, for example, Helicobacter pylori and hepatitis C, limited the ability to interpret results.
‘Exciting and important’ findings
Commenting on the research for this news organization, Aparna Wagle Shukla, MD, professor, Norman Fixel Institute for Neurological Diseases, University of Florida, Gainesville, said the results amid the current pandemic are “exciting and important” and “have reinvigorated interest” in the role of infection in PD.
However, the study had some limitations, an important one being lack of accounting for confounding factors, including environmental factors, she said. Exposure to pesticides, living in a rural area, drinking well water, and having had a head injury may increase PD risk, whereas high intake of caffeine, nicotine, alcohol, and nonsteroidal anti-inflammatory drugs might lower the risk.
The researchers did not take into account exposure to multiple microbes or “infection burden,” said Dr. Wagle Shukla, who was not involved in the current study. In addition, as the data are from a single country with exposure to specific influenza strains, application of the findings elsewhere may be limited.
Dr. Wagle Shukla noted that a case-control design “isn’t ideal” from an epidemiological perspective. “Future studies should involve large cohorts followed longitudinally.”
The study was supported by grants from the Lundbeck Foundation and the Augustinus Foundation. Dr. Cocoros has disclosed no relevant financial relationships. Several coauthors have disclosed relationships with industry. The full list can be found with the original article.
A version of this article first appeared on Medscape.com.
Influenza infection is linked to a subsequent diagnosis of Parkinson’s disease (PD) more than 10 years later, resurfacing a long-held debate about whether infection increases the risk for movement disorders over the long term.
In a large case-control study, investigators found and by more than 70% for PD occurring more than 10 years after the flu.
“This study is not definitive by any means, but it certainly suggests there are potential long-term consequences from influenza,” study investigator Noelle M. Cocoros, DSc, research scientist at Harvard Pilgrim Health Care Institute and Harvard Medical School, Boston, said in an interview.
The study was published online Oct. 25 in JAMA Neurology.
Ongoing debate
The debate about whether influenza is associated with PD has been going on as far back as the 1918 influenza pandemic, when experts documented parkinsonism in affected individuals.
Using data from the Danish patient registry, researchers identified 10,271 subjects diagnosed with PD during a 17-year period (2000-2016). Of these, 38.7% were female, and the mean age was 71.4 years.
They matched these subjects for age and sex to 51,355 controls without PD. Compared with controls, slightly fewer individuals with PD had chronic obstructive pulmonary disease (COPD) or emphysema, but there was a similar distribution of cardiovascular disease and various other conditions.
Researchers collected data on influenza diagnoses from inpatient and outpatient hospital clinics from 1977 to 2016. They plotted these by month and year on a graph, calculated the median number of diagnoses per month, and identified peaks as those with more than threefold the median.
They categorized cases in groups related to the time between the infection and PD: More than 10 years, 10-15 years, and more than 15 years.
The time lapse accounts for a rather long “run-up” to PD, said Dr. Cocoros. There’s a sometimes decades-long preclinical phase before patients develop typical motor signs and a prodromal phase where they may present with nonmotor symptoms such as sleep disorders and constipation.
“We expected there would be at least 10 years between any infection and PD if there was an association present,” said Dr. Cocoros.
Investigators found an association between influenza exposure and PD diagnosis “that held up over time,” she said.
For more than 10 years before PD, the likelihood of a diagnosis for the infected compared with the unexposed was increased 73% (odds ratio [OR] 1.73; 95% confidence interval, 1.11-2.71; P = .02) after adjustment for cardiovascular disease, diabetes, chronic obstructive pulmonary disease, emphysema, lung cancer, Crohn’s disease, and ulcerative colitis.
The odds increased with more time from infection. For more than 15 years, the adjusted OR was 1.91 (95% CI, 1.14 - 3.19; P =.01).
However, for the 10- to 15-year time frame, the point estimate was reduced and the CI nonsignificant (OR, 1.33; 95% CI, 0.54-3.27; P = .53). This “is a little hard to interpret,” but could be a result of the small numbers, exposure misclassification, or because “the longer time interval is what’s meaningful,” said Dr. Cocoros.
Potential COVID-19–related PD surge?
In a sensitivity analysis, researchers looked at peak infection activity. “We wanted to increase the likelihood of these diagnoses representing actual infection,” Dr. Cocoros noted.
Here, the OR was still elevated at more than 10 years, but the CI was quite wide and included 1 (OR, 1.52; 95% CI, 0.80-2.89; P = .21). “So the association holds up, but the estimates are quite unstable,” said Dr. Cocoros.
Researchers examined associations with numerous other infection types, but did not see the same trend over time. Some infections – for example, gastrointestinal infections and septicemia – were associated with PD within 5 years, but most associations appeared to be null after more than 10 years.
“There seemed to be associations earlier between the infection and PD, which we interpret to suggest there’s actually not a meaningful association,” said Dr. Cocoros.
An exception might be urinary tract infections (UTIs), where after 10 years, the adjusted OR was 1.19 (95% CI, 1.01-1.40). Research suggests patients with PD often have UTIs and neurogenic bladder.
“It’s possible that UTIs could be an early symptom of PD rather than a causative factor,” said Dr. Cocoros.
It’s unclear how influenza might lead to PD but it could be that the virus gets into the central nervous system, resulting in neuroinflammation. Cytokines generated in response to the influenza infection might damage the brain.
“The infection could be a ‘primer’ or an initial ‘hit’ to the system, maybe setting people up for PD,” said Dr. Cocoros.
As for the current COVID-19 pandemic, some experts are concerned about a potential surge in PD cases in decades to come, and are calling for prospective monitoring of patients with this infection, said Dr. Cocoros.
However, she noted that infections don’t account for all PD cases and that genetic and environmental factors also influence risk.
Many individuals who contract influenza don’t seek medical care or get tested, so it’s possible the study counted those who had the infection as unexposed. Another potential study limitation was that small numbers for some infections, for example, Helicobacter pylori and hepatitis C, limited the ability to interpret results.
‘Exciting and important’ findings
Commenting on the research for this news organization, Aparna Wagle Shukla, MD, professor, Norman Fixel Institute for Neurological Diseases, University of Florida, Gainesville, said the results amid the current pandemic are “exciting and important” and “have reinvigorated interest” in the role of infection in PD.
However, the study had some limitations, an important one being lack of accounting for confounding factors, including environmental factors, she said. Exposure to pesticides, living in a rural area, drinking well water, and having had a head injury may increase PD risk, whereas high intake of caffeine, nicotine, alcohol, and nonsteroidal anti-inflammatory drugs might lower the risk.
The researchers did not take into account exposure to multiple microbes or “infection burden,” said Dr. Wagle Shukla, who was not involved in the current study. In addition, as the data are from a single country with exposure to specific influenza strains, application of the findings elsewhere may be limited.
Dr. Wagle Shukla noted that a case-control design “isn’t ideal” from an epidemiological perspective. “Future studies should involve large cohorts followed longitudinally.”
The study was supported by grants from the Lundbeck Foundation and the Augustinus Foundation. Dr. Cocoros has disclosed no relevant financial relationships. Several coauthors have disclosed relationships with industry. The full list can be found with the original article.
A version of this article first appeared on Medscape.com.
Sleep time ‘sweet spot’ to slow cognitive decline identified?
In a longitudinal study, investigators found older adults who slept less than 4.5 hours or more than 6.5 hours a night reported significant cognitive decline over time, but cognitive scores for those with sleep duration in between that range remained stable.
“This really suggests that there’s this middle range, a ‘sweet spot,’ where your sleep is really optimal,” lead author Brendan Lucey, MD, MSCI, associate professor of neurology and director of the Washington University Sleep Medicine Center, St. Louis, said in an interview.
The study, published online Oct. 20, 2021, in the journal Brain, is part of a growing body of research that seeks to determine if sleep can be used as a marker of Alzheimer’s disease progression.
A complex relationship
Studies suggest a strong relationship between sleep patterns and Alzheimer’s disease, which affects nearly 6 million Americans. The challenge, Dr. Lucey said, is unwinding the complex links between sleep, AD, and cognitive function.
An earlier study by Dr. Lucey and colleagues found that poor sleep quality is associated with early signs of AD, and a report published in September found that elderly people who slept less than 6 hours a night had a greater burden of amyloid-beta, a hallmark sign of AD.
For this new study, researchers monitored sleep-wake activity over 4-6 nights in 100 participants who underwent annual cognitive assessments and clinical studies, including APOE genotyping, as part of a longitudinal study at the Knight Alzheimer Disease Research Center at Washington University.
Participants also provided cerebrospinal fluid (CSF) total tau and amyloid-beta 42 and wore a small EEG device on their forehead while they slept.
The majority of participants had a clinical dementia rating (CDR) score of 0, indicating no cognitive impairment. Twelve individuals had a CDR greater than 0, with most reporting mild cognitive impairment.
As expected, CSF analysis showed greater evidence of AD pathology in those with a baseline CDR greater than 0.
Changes in cognitive function were measured using a Preclinical Alzheimer Cognitive Composite (PACC) score, a composite of results from a neuropsychological testing battery that included the Free and Cued Selective Reminding Test, the Logical Memory Delayed Recall Test from the Wechsler Memory Scale–Revised, the Digit Symbol Substitution Test from the Wechsler Adult Intelligence Scale–Revised, and the Mini-Mental State Examination.
Researchers found an upside-down U-shaped relationship between PACC scores and sleep duration, with dramatic cognitive decline in those who slept less than 4.5 hours or more than 6.5 hours a night (P < .001 for both).
The U-shaped relationship was also found with measures of sleep phases, including time spent in rapid eye movement and in non-REM sleep (P < .001 for both).
The findings persisted even after controlling for confounders that can affect sleep and cognition, such as age, CSF total tau/amyloid-beta 42 ratio, apo E four-allele carrier status, years of education, and sex.
Understanding how sleep changes at different stages of AD could help researchers determine if sleep can be used as a marker of disease progression, Dr. Lucey said. That could lead to interventions to slow that process.
“We’re not at the point yet where we can say that we need to monitor someone’s sleep time and then do an intervention to see if it would improve their risk for cognitive decline,” said Dr. Lucey, who plans to repeat this sleep study with the same cohort to track changes in sleep patterns and cognitive function over time. “But that’s a question I’m very excited to try to answer.”
A component of cognitive health
Commenting on the findings for this news organization, Heather Snyder, PhD, vice president of medical and scientific relations for the Alzheimer’s Association, noted that the study adds to a body of evidence linking sleep and cognition, especially how sleep quality can optimize brain function.
“We’ve seen previous research that’s shown poor sleep contributes to dementia risk, as well as research showing sleep duration may play a role in cognition,” she said.
“We also need studies that look at sleep as an intervention for cognitive health,” Dr. Snyder said. “Sleep is an important aspect of our overall health. Clinicians should have conversations with their patients about sleep as part of standard discussions about their health habits and wellness.”
The study was funded by the National Institutes of Health, the American Sleep Medicine Foundation, the Roger and Paula Riney Fund, and the Daniel J. Brennan, MD Fund. Dr. Lucey consults for Merck and Eli Lilly. Dr. Snyder has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a longitudinal study, investigators found older adults who slept less than 4.5 hours or more than 6.5 hours a night reported significant cognitive decline over time, but cognitive scores for those with sleep duration in between that range remained stable.
“This really suggests that there’s this middle range, a ‘sweet spot,’ where your sleep is really optimal,” lead author Brendan Lucey, MD, MSCI, associate professor of neurology and director of the Washington University Sleep Medicine Center, St. Louis, said in an interview.
The study, published online Oct. 20, 2021, in the journal Brain, is part of a growing body of research that seeks to determine if sleep can be used as a marker of Alzheimer’s disease progression.
A complex relationship
Studies suggest a strong relationship between sleep patterns and Alzheimer’s disease, which affects nearly 6 million Americans. The challenge, Dr. Lucey said, is unwinding the complex links between sleep, AD, and cognitive function.
An earlier study by Dr. Lucey and colleagues found that poor sleep quality is associated with early signs of AD, and a report published in September found that elderly people who slept less than 6 hours a night had a greater burden of amyloid-beta, a hallmark sign of AD.
For this new study, researchers monitored sleep-wake activity over 4-6 nights in 100 participants who underwent annual cognitive assessments and clinical studies, including APOE genotyping, as part of a longitudinal study at the Knight Alzheimer Disease Research Center at Washington University.
Participants also provided cerebrospinal fluid (CSF) total tau and amyloid-beta 42 and wore a small EEG device on their forehead while they slept.
The majority of participants had a clinical dementia rating (CDR) score of 0, indicating no cognitive impairment. Twelve individuals had a CDR greater than 0, with most reporting mild cognitive impairment.
As expected, CSF analysis showed greater evidence of AD pathology in those with a baseline CDR greater than 0.
Changes in cognitive function were measured using a Preclinical Alzheimer Cognitive Composite (PACC) score, a composite of results from a neuropsychological testing battery that included the Free and Cued Selective Reminding Test, the Logical Memory Delayed Recall Test from the Wechsler Memory Scale–Revised, the Digit Symbol Substitution Test from the Wechsler Adult Intelligence Scale–Revised, and the Mini-Mental State Examination.
Researchers found an upside-down U-shaped relationship between PACC scores and sleep duration, with dramatic cognitive decline in those who slept less than 4.5 hours or more than 6.5 hours a night (P < .001 for both).
The U-shaped relationship was also found with measures of sleep phases, including time spent in rapid eye movement and in non-REM sleep (P < .001 for both).
The findings persisted even after controlling for confounders that can affect sleep and cognition, such as age, CSF total tau/amyloid-beta 42 ratio, apo E four-allele carrier status, years of education, and sex.
Understanding how sleep changes at different stages of AD could help researchers determine if sleep can be used as a marker of disease progression, Dr. Lucey said. That could lead to interventions to slow that process.
“We’re not at the point yet where we can say that we need to monitor someone’s sleep time and then do an intervention to see if it would improve their risk for cognitive decline,” said Dr. Lucey, who plans to repeat this sleep study with the same cohort to track changes in sleep patterns and cognitive function over time. “But that’s a question I’m very excited to try to answer.”
A component of cognitive health
Commenting on the findings for this news organization, Heather Snyder, PhD, vice president of medical and scientific relations for the Alzheimer’s Association, noted that the study adds to a body of evidence linking sleep and cognition, especially how sleep quality can optimize brain function.
“We’ve seen previous research that’s shown poor sleep contributes to dementia risk, as well as research showing sleep duration may play a role in cognition,” she said.
“We also need studies that look at sleep as an intervention for cognitive health,” Dr. Snyder said. “Sleep is an important aspect of our overall health. Clinicians should have conversations with their patients about sleep as part of standard discussions about their health habits and wellness.”
The study was funded by the National Institutes of Health, the American Sleep Medicine Foundation, the Roger and Paula Riney Fund, and the Daniel J. Brennan, MD Fund. Dr. Lucey consults for Merck and Eli Lilly. Dr. Snyder has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a longitudinal study, investigators found older adults who slept less than 4.5 hours or more than 6.5 hours a night reported significant cognitive decline over time, but cognitive scores for those with sleep duration in between that range remained stable.
“This really suggests that there’s this middle range, a ‘sweet spot,’ where your sleep is really optimal,” lead author Brendan Lucey, MD, MSCI, associate professor of neurology and director of the Washington University Sleep Medicine Center, St. Louis, said in an interview.
The study, published online Oct. 20, 2021, in the journal Brain, is part of a growing body of research that seeks to determine if sleep can be used as a marker of Alzheimer’s disease progression.
A complex relationship
Studies suggest a strong relationship between sleep patterns and Alzheimer’s disease, which affects nearly 6 million Americans. The challenge, Dr. Lucey said, is unwinding the complex links between sleep, AD, and cognitive function.
An earlier study by Dr. Lucey and colleagues found that poor sleep quality is associated with early signs of AD, and a report published in September found that elderly people who slept less than 6 hours a night had a greater burden of amyloid-beta, a hallmark sign of AD.
For this new study, researchers monitored sleep-wake activity over 4-6 nights in 100 participants who underwent annual cognitive assessments and clinical studies, including APOE genotyping, as part of a longitudinal study at the Knight Alzheimer Disease Research Center at Washington University.
Participants also provided cerebrospinal fluid (CSF) total tau and amyloid-beta 42 and wore a small EEG device on their forehead while they slept.
The majority of participants had a clinical dementia rating (CDR) score of 0, indicating no cognitive impairment. Twelve individuals had a CDR greater than 0, with most reporting mild cognitive impairment.
As expected, CSF analysis showed greater evidence of AD pathology in those with a baseline CDR greater than 0.
Changes in cognitive function were measured using a Preclinical Alzheimer Cognitive Composite (PACC) score, a composite of results from a neuropsychological testing battery that included the Free and Cued Selective Reminding Test, the Logical Memory Delayed Recall Test from the Wechsler Memory Scale–Revised, the Digit Symbol Substitution Test from the Wechsler Adult Intelligence Scale–Revised, and the Mini-Mental State Examination.
Researchers found an upside-down U-shaped relationship between PACC scores and sleep duration, with dramatic cognitive decline in those who slept less than 4.5 hours or more than 6.5 hours a night (P < .001 for both).
The U-shaped relationship was also found with measures of sleep phases, including time spent in rapid eye movement and in non-REM sleep (P < .001 for both).
The findings persisted even after controlling for confounders that can affect sleep and cognition, such as age, CSF total tau/amyloid-beta 42 ratio, apo E four-allele carrier status, years of education, and sex.
Understanding how sleep changes at different stages of AD could help researchers determine if sleep can be used as a marker of disease progression, Dr. Lucey said. That could lead to interventions to slow that process.
“We’re not at the point yet where we can say that we need to monitor someone’s sleep time and then do an intervention to see if it would improve their risk for cognitive decline,” said Dr. Lucey, who plans to repeat this sleep study with the same cohort to track changes in sleep patterns and cognitive function over time. “But that’s a question I’m very excited to try to answer.”
A component of cognitive health
Commenting on the findings for this news organization, Heather Snyder, PhD, vice president of medical and scientific relations for the Alzheimer’s Association, noted that the study adds to a body of evidence linking sleep and cognition, especially how sleep quality can optimize brain function.
“We’ve seen previous research that’s shown poor sleep contributes to dementia risk, as well as research showing sleep duration may play a role in cognition,” she said.
“We also need studies that look at sleep as an intervention for cognitive health,” Dr. Snyder said. “Sleep is an important aspect of our overall health. Clinicians should have conversations with their patients about sleep as part of standard discussions about their health habits and wellness.”
The study was funded by the National Institutes of Health, the American Sleep Medicine Foundation, the Roger and Paula Riney Fund, and the Daniel J. Brennan, MD Fund. Dr. Lucey consults for Merck and Eli Lilly. Dr. Snyder has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
MS and COVID: Docs switched DMTs but maybe didn’t need to
These are the messages of a pair of new studies that examine the impact of the pandemic on the treatment of MS.
One report finds that 80% of specialists surveyed in the summer of 2020 said the pandemic may have changed how they prescribe disease-modifying therapies (DMTs). However, the other report finds no evidence that choice of DMT affects risk of COVID-19 infection. Both studies were presented at the 2021 Annual Meeting of the Consortium of Multiple Sclerosis Centers (CMSC).
For the survey, researchers led by neurologist Elizabeth H. Morrison-Banks, MD, of the University of California, Riverside, sent questions to 188 clinicians who serve on regional National Multiple Sclerosis Society Healthcare Provider Councils. A total of 86 people responded: 45 physicians, 18 rehabilitation therapists, 7 psychologists, 8 advanced practice clinicians, 4 social workers, 2 nurses, a pharmacist, and a researcher.
The results, which were published earlier in 2021 in Multiple Sclerosis and Related Disorders, revealed that the survey participants were prescribing certain DMTs more often: beta-interferons (prescribed more by 28.6% of prescribers), natalizumab (23.8%), and glatiramer acetate (21.4%). Those prescribed less included alemtuzumab (64.2% prescribed it less), cladribine (52.4%), and B cell–depleting agents including ocrelizumab and rituximab (50%). Some specialists suspended drugs entirely (21.4% for alemtuzumab, 16.7% for B cell–depleting agents) or extending dosing intervals (38.1% for natalizumab, 11.9% for fingolimod and siponimod).
“We suspect that some of the lower-efficacy therapies were prescribed more often because these therapies were much less immunosuppressive, and because they did not require in-person visits that would increase risk of viral exposure from infusion center staff, or from other infusion patients,” Dr. Morrison-Banks said in an interview. “We also suspect that some of our survey respondents may have increased the dosing intervals for higher-efficacy therapies such as B cell–modulating agents – or even avoided these therapies altogether – because they were concerned that immunosuppressive agents might trigger severe complications from COVID-19.”
As she noted, “in retrospect, at least some of the concerns expressed in our survey may not have been entirely warranted, but then again, we all knew even less then about COVID-19.”
Indeed, researchers led by neurologist Tyler E. Smith, MD, of New York University Langone Multiple Sclerosis Care Center are reporting that they couldn’t find any link between the following DMTs and higher rates of COVID-19 at the New York City center: rituximab, ocrelizumab, fumerate (dimethyl fumarate, monomethyl fumarate, diroximel fumarate), sphingosine-1-phosphate modulators (fingolimod, siponimod), and natalizumab.
The researchers tracked 1,439 patients with MS who were taking the DMTs from March 2020 to March 2021. Of those, 16.0% were infected with COVID-19 (75% lab confirmed), 6.5% were hospitalized, and 0.9% died.
“We did not find an association between the choice of disease-modifying therapy and developing COVID-19 infection, nor having increased disease severity,” Dr. Smith said in an interview. “We are still analyzing data and hope to publish an updated analysis, but at this point, we don’t have conclusive evidence that DMTs, including anti-CD20 agents, need to be changed to lower the risk of COVID-19.”
Instead, he said, “at this point, we feel our energies should be spent on educating our patients on importance of vaccines and boosters. I don’t think it is necessary to switch DMTs because of COVID-19 concerns. However, this should be reviewed on a case-by-case basis.”
No funding is reported for the survey study, and the authors reported various disclosures. The DMT study was funded by an investigator-initiated grant from the Consortium of Multiple Sclerosis Centers, and the authors reported various disclosures.
These are the messages of a pair of new studies that examine the impact of the pandemic on the treatment of MS.
One report finds that 80% of specialists surveyed in the summer of 2020 said the pandemic may have changed how they prescribe disease-modifying therapies (DMTs). However, the other report finds no evidence that choice of DMT affects risk of COVID-19 infection. Both studies were presented at the 2021 Annual Meeting of the Consortium of Multiple Sclerosis Centers (CMSC).
For the survey, researchers led by neurologist Elizabeth H. Morrison-Banks, MD, of the University of California, Riverside, sent questions to 188 clinicians who serve on regional National Multiple Sclerosis Society Healthcare Provider Councils. A total of 86 people responded: 45 physicians, 18 rehabilitation therapists, 7 psychologists, 8 advanced practice clinicians, 4 social workers, 2 nurses, a pharmacist, and a researcher.
The results, which were published earlier in 2021 in Multiple Sclerosis and Related Disorders, revealed that the survey participants were prescribing certain DMTs more often: beta-interferons (prescribed more by 28.6% of prescribers), natalizumab (23.8%), and glatiramer acetate (21.4%). Those prescribed less included alemtuzumab (64.2% prescribed it less), cladribine (52.4%), and B cell–depleting agents including ocrelizumab and rituximab (50%). Some specialists suspended drugs entirely (21.4% for alemtuzumab, 16.7% for B cell–depleting agents) or extending dosing intervals (38.1% for natalizumab, 11.9% for fingolimod and siponimod).
“We suspect that some of the lower-efficacy therapies were prescribed more often because these therapies were much less immunosuppressive, and because they did not require in-person visits that would increase risk of viral exposure from infusion center staff, or from other infusion patients,” Dr. Morrison-Banks said in an interview. “We also suspect that some of our survey respondents may have increased the dosing intervals for higher-efficacy therapies such as B cell–modulating agents – or even avoided these therapies altogether – because they were concerned that immunosuppressive agents might trigger severe complications from COVID-19.”
As she noted, “in retrospect, at least some of the concerns expressed in our survey may not have been entirely warranted, but then again, we all knew even less then about COVID-19.”
Indeed, researchers led by neurologist Tyler E. Smith, MD, of New York University Langone Multiple Sclerosis Care Center are reporting that they couldn’t find any link between the following DMTs and higher rates of COVID-19 at the New York City center: rituximab, ocrelizumab, fumerate (dimethyl fumarate, monomethyl fumarate, diroximel fumarate), sphingosine-1-phosphate modulators (fingolimod, siponimod), and natalizumab.
The researchers tracked 1,439 patients with MS who were taking the DMTs from March 2020 to March 2021. Of those, 16.0% were infected with COVID-19 (75% lab confirmed), 6.5% were hospitalized, and 0.9% died.
“We did not find an association between the choice of disease-modifying therapy and developing COVID-19 infection, nor having increased disease severity,” Dr. Smith said in an interview. “We are still analyzing data and hope to publish an updated analysis, but at this point, we don’t have conclusive evidence that DMTs, including anti-CD20 agents, need to be changed to lower the risk of COVID-19.”
Instead, he said, “at this point, we feel our energies should be spent on educating our patients on importance of vaccines and boosters. I don’t think it is necessary to switch DMTs because of COVID-19 concerns. However, this should be reviewed on a case-by-case basis.”
No funding is reported for the survey study, and the authors reported various disclosures. The DMT study was funded by an investigator-initiated grant from the Consortium of Multiple Sclerosis Centers, and the authors reported various disclosures.
These are the messages of a pair of new studies that examine the impact of the pandemic on the treatment of MS.
One report finds that 80% of specialists surveyed in the summer of 2020 said the pandemic may have changed how they prescribe disease-modifying therapies (DMTs). However, the other report finds no evidence that choice of DMT affects risk of COVID-19 infection. Both studies were presented at the 2021 Annual Meeting of the Consortium of Multiple Sclerosis Centers (CMSC).
For the survey, researchers led by neurologist Elizabeth H. Morrison-Banks, MD, of the University of California, Riverside, sent questions to 188 clinicians who serve on regional National Multiple Sclerosis Society Healthcare Provider Councils. A total of 86 people responded: 45 physicians, 18 rehabilitation therapists, 7 psychologists, 8 advanced practice clinicians, 4 social workers, 2 nurses, a pharmacist, and a researcher.
The results, which were published earlier in 2021 in Multiple Sclerosis and Related Disorders, revealed that the survey participants were prescribing certain DMTs more often: beta-interferons (prescribed more by 28.6% of prescribers), natalizumab (23.8%), and glatiramer acetate (21.4%). Those prescribed less included alemtuzumab (64.2% prescribed it less), cladribine (52.4%), and B cell–depleting agents including ocrelizumab and rituximab (50%). Some specialists suspended drugs entirely (21.4% for alemtuzumab, 16.7% for B cell–depleting agents) or extending dosing intervals (38.1% for natalizumab, 11.9% for fingolimod and siponimod).
“We suspect that some of the lower-efficacy therapies were prescribed more often because these therapies were much less immunosuppressive, and because they did not require in-person visits that would increase risk of viral exposure from infusion center staff, or from other infusion patients,” Dr. Morrison-Banks said in an interview. “We also suspect that some of our survey respondents may have increased the dosing intervals for higher-efficacy therapies such as B cell–modulating agents – or even avoided these therapies altogether – because they were concerned that immunosuppressive agents might trigger severe complications from COVID-19.”
As she noted, “in retrospect, at least some of the concerns expressed in our survey may not have been entirely warranted, but then again, we all knew even less then about COVID-19.”
Indeed, researchers led by neurologist Tyler E. Smith, MD, of New York University Langone Multiple Sclerosis Care Center are reporting that they couldn’t find any link between the following DMTs and higher rates of COVID-19 at the New York City center: rituximab, ocrelizumab, fumerate (dimethyl fumarate, monomethyl fumarate, diroximel fumarate), sphingosine-1-phosphate modulators (fingolimod, siponimod), and natalizumab.
The researchers tracked 1,439 patients with MS who were taking the DMTs from March 2020 to March 2021. Of those, 16.0% were infected with COVID-19 (75% lab confirmed), 6.5% were hospitalized, and 0.9% died.
“We did not find an association between the choice of disease-modifying therapy and developing COVID-19 infection, nor having increased disease severity,” Dr. Smith said in an interview. “We are still analyzing data and hope to publish an updated analysis, but at this point, we don’t have conclusive evidence that DMTs, including anti-CD20 agents, need to be changed to lower the risk of COVID-19.”
Instead, he said, “at this point, we feel our energies should be spent on educating our patients on importance of vaccines and boosters. I don’t think it is necessary to switch DMTs because of COVID-19 concerns. However, this should be reviewed on a case-by-case basis.”
No funding is reported for the survey study, and the authors reported various disclosures. The DMT study was funded by an investigator-initiated grant from the Consortium of Multiple Sclerosis Centers, and the authors reported various disclosures.
FROM CMSC 2021
Autism prevalence in children as high as 10% in some New Jersey communities
Investigators found that up to 10% of children in some of the state’s school districts have an ASD diagnosis vs. the national average of just under 2%.
School districts with higher ASD prevalence in the study have expansive health and educational programs in place to diagnose and support children with ASD, which likely contributed to the higher caseloads, senior investigator Walter Zahorodny, PhD, associate professor of pediatrics at New Jersey Medical School in Newark, said in an interview.
“When you have those players on the ground, it’s likely we’re going to be finding more, if not almost all, of the children with autism in a given district,” said Dr. Zahorodny, director of the New Jersey Autism Study for the Centers for Disease Control and Prevention.
The study was published online Oct. 21 in the journal Autism Research.
Local analysis
Researchers used the Autism and Developmental Disabilities Monitoring (ADDM) Network, a surveillance method developed by the CDC that includes data collected from health and special education records of children living in 11 communities across the United States. New Jersey is one of the ADDM participating sites.
National data are important, but Dr. Zahorodny and colleagues wanted to examine ASD prevalence at a more granular level, comparing prevalence district by district.
They examined data from 5,453 children who were 8 years old in 2016 and attended public school in Essex, Hudson, Ocean, and Union counties.
The prevalence of ASD was 36 children per 1,000 overall. Hudson County reported the lowest rate, at 31 cases per 1,000 children, and Ocean County reported the highest, at 54 cases per 1,000 children.
Across the region, ASD prevalence was four times higher in boys vs. girls, mirroring national statistics.
High ASD prevalence was more likely in mid-socioeconomic status districts (prevalence ratio [PR], 1.2; P = .01) and in larger school districts (PR, 1.3; P = .004).
Hispanic children had significantly lower ASD prevalence overall compared with White children (PR, 0.6; P < .001). In fact, prevalence was 30%-60% lower among Hispanic children in three of four counties compared with White children.
Another study is underway to better understand why autism rates were lower in specific districts and Hispanic children overall, but Dr. Zahorodny said one possibility is a lack of resources in those districts.
Will new methodology miss cases?
The study’s methodology was used by the CDC from 2000 to 2016 and includes assessment of children who have an ASD diagnosis, and children who haven’t received a diagnosis but have documented behaviors consistent with ASD.
In 2018, it was replaced with a less comprehensive approach that relies only on children with an ASD diagnosis. Data using this new methodology have not yet been reported.
In the new study from New Jersey, 767 children with autism were diagnosed by a pediatrician, neurologist, or other community provider. The remaining 175 children with autism, 18.6% of the total cohort, did not have an ASD diagnosis but met the ADDM case definition.
Under the new methodology, those children would not be counted.
“Something could be lost in the new methodology in terms of usefulness of the information because when the estimates are incomplete or low, that might lead people to make the wrong judgments when they make decisions about resources,” Dr. Zahorodny said.
The study was funded by the Centers for Disease Control and Prevention and the National Institutes of Health. The study authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Investigators found that up to 10% of children in some of the state’s school districts have an ASD diagnosis vs. the national average of just under 2%.
School districts with higher ASD prevalence in the study have expansive health and educational programs in place to diagnose and support children with ASD, which likely contributed to the higher caseloads, senior investigator Walter Zahorodny, PhD, associate professor of pediatrics at New Jersey Medical School in Newark, said in an interview.
“When you have those players on the ground, it’s likely we’re going to be finding more, if not almost all, of the children with autism in a given district,” said Dr. Zahorodny, director of the New Jersey Autism Study for the Centers for Disease Control and Prevention.
The study was published online Oct. 21 in the journal Autism Research.
Local analysis
Researchers used the Autism and Developmental Disabilities Monitoring (ADDM) Network, a surveillance method developed by the CDC that includes data collected from health and special education records of children living in 11 communities across the United States. New Jersey is one of the ADDM participating sites.
National data are important, but Dr. Zahorodny and colleagues wanted to examine ASD prevalence at a more granular level, comparing prevalence district by district.
They examined data from 5,453 children who were 8 years old in 2016 and attended public school in Essex, Hudson, Ocean, and Union counties.
The prevalence of ASD was 36 children per 1,000 overall. Hudson County reported the lowest rate, at 31 cases per 1,000 children, and Ocean County reported the highest, at 54 cases per 1,000 children.
Across the region, ASD prevalence was four times higher in boys vs. girls, mirroring national statistics.
High ASD prevalence was more likely in mid-socioeconomic status districts (prevalence ratio [PR], 1.2; P = .01) and in larger school districts (PR, 1.3; P = .004).
Hispanic children had significantly lower ASD prevalence overall compared with White children (PR, 0.6; P < .001). In fact, prevalence was 30%-60% lower among Hispanic children in three of four counties compared with White children.
Another study is underway to better understand why autism rates were lower in specific districts and Hispanic children overall, but Dr. Zahorodny said one possibility is a lack of resources in those districts.
Will new methodology miss cases?
The study’s methodology was used by the CDC from 2000 to 2016 and includes assessment of children who have an ASD diagnosis, and children who haven’t received a diagnosis but have documented behaviors consistent with ASD.
In 2018, it was replaced with a less comprehensive approach that relies only on children with an ASD diagnosis. Data using this new methodology have not yet been reported.
In the new study from New Jersey, 767 children with autism were diagnosed by a pediatrician, neurologist, or other community provider. The remaining 175 children with autism, 18.6% of the total cohort, did not have an ASD diagnosis but met the ADDM case definition.
Under the new methodology, those children would not be counted.
“Something could be lost in the new methodology in terms of usefulness of the information because when the estimates are incomplete or low, that might lead people to make the wrong judgments when they make decisions about resources,” Dr. Zahorodny said.
The study was funded by the Centers for Disease Control and Prevention and the National Institutes of Health. The study authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Investigators found that up to 10% of children in some of the state’s school districts have an ASD diagnosis vs. the national average of just under 2%.
School districts with higher ASD prevalence in the study have expansive health and educational programs in place to diagnose and support children with ASD, which likely contributed to the higher caseloads, senior investigator Walter Zahorodny, PhD, associate professor of pediatrics at New Jersey Medical School in Newark, said in an interview.
“When you have those players on the ground, it’s likely we’re going to be finding more, if not almost all, of the children with autism in a given district,” said Dr. Zahorodny, director of the New Jersey Autism Study for the Centers for Disease Control and Prevention.
The study was published online Oct. 21 in the journal Autism Research.
Local analysis
Researchers used the Autism and Developmental Disabilities Monitoring (ADDM) Network, a surveillance method developed by the CDC that includes data collected from health and special education records of children living in 11 communities across the United States. New Jersey is one of the ADDM participating sites.
National data are important, but Dr. Zahorodny and colleagues wanted to examine ASD prevalence at a more granular level, comparing prevalence district by district.
They examined data from 5,453 children who were 8 years old in 2016 and attended public school in Essex, Hudson, Ocean, and Union counties.
The prevalence of ASD was 36 children per 1,000 overall. Hudson County reported the lowest rate, at 31 cases per 1,000 children, and Ocean County reported the highest, at 54 cases per 1,000 children.
Across the region, ASD prevalence was four times higher in boys vs. girls, mirroring national statistics.
High ASD prevalence was more likely in mid-socioeconomic status districts (prevalence ratio [PR], 1.2; P = .01) and in larger school districts (PR, 1.3; P = .004).
Hispanic children had significantly lower ASD prevalence overall compared with White children (PR, 0.6; P < .001). In fact, prevalence was 30%-60% lower among Hispanic children in three of four counties compared with White children.
Another study is underway to better understand why autism rates were lower in specific districts and Hispanic children overall, but Dr. Zahorodny said one possibility is a lack of resources in those districts.
Will new methodology miss cases?
The study’s methodology was used by the CDC from 2000 to 2016 and includes assessment of children who have an ASD diagnosis, and children who haven’t received a diagnosis but have documented behaviors consistent with ASD.
In 2018, it was replaced with a less comprehensive approach that relies only on children with an ASD diagnosis. Data using this new methodology have not yet been reported.
In the new study from New Jersey, 767 children with autism were diagnosed by a pediatrician, neurologist, or other community provider. The remaining 175 children with autism, 18.6% of the total cohort, did not have an ASD diagnosis but met the ADDM case definition.
Under the new methodology, those children would not be counted.
“Something could be lost in the new methodology in terms of usefulness of the information because when the estimates are incomplete or low, that might lead people to make the wrong judgments when they make decisions about resources,” Dr. Zahorodny said.
The study was funded by the Centers for Disease Control and Prevention and the National Institutes of Health. The study authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Ohio records more deaths than births for first time
In 2020, around 143,661 Ohioans died and 129,313 Ohioans were born, according to The Columbus Dispatch. The trend appears to have continued so far this year, with 107,462 deaths and 100,781 births reported to date.
Deaths haven’t surpassed births in the 112 years since the state began compiling data in 1909, the newspaper reported. The state’s birth rate has been declining for years while the number of deaths has risen, though data shows that the COVID-19 pandemic accelerated the shift.
“It doesn’t surprise me at all,” Joseph Gastaldo, MD, the medical director of infectious diseases for OhioHealth, told the newspaper.
Ohio’s birth rate fell by 4% in 2020, which defied public expectations about a pandemic birth boom. In addition, the state reported 13,927 COVID-19 deaths throughout the year.
“It’s COVID, clearly,” he noted.
Alabama also recorded more deaths than births for the first time last year, according to The New York Times. The state reported 64,714 deaths and 57,641 births in 2020.
“Our state literally shrunk in 2020,” Scott Harris, MD, the state health officer for Alabama, said at a news conference in September.
The state had never recorded a gap that large, even during World War I, World War II, and the 1918 flu pandemic, he said. Alabama has kept records on the numbers since 1900.
“We’ve never had a time when deaths exceeded births,” Dr. Harris said.
In fact, about half of U.S. states reported death rates higher than birth rates in 2020, according to a recent study from researchers at the University of New Hampshire. In 2019, only five states --Maine, New Hampshire, Rhode Island, Vermont, and West Virginia -- reported more deaths than births.
In 2020, the United States reported a record of nearly 3.4 million deaths, which was 18% more than in 2019, the researchers found. COVID-19 was the primary reason for the increase in deaths, accounting for about 11% of total deaths. Meanwhile, births dropped by 4% to about 3.6 million.
The surplus of births over deaths added 229,000 people to the U.S. population in 2020, as compared to 892,000 in 2019, which means the country’s population growth slowed last year. The decline, paired with lower immigration rates during the pandemic, led to the smallest annual percentage population gain in at least 100 years.
“Deaths will likely exceed births again in many states in 2021,” Kenneth Johnson, PhD, a senior demographer and study author, wrote in a statement.
“How large or protracted these fertility declines and mortality increases will be remains to be seen, but they have already dramatically reduced population growth in the United States,” he said.
A version of this article first appeared on WebMD.com.
In 2020, around 143,661 Ohioans died and 129,313 Ohioans were born, according to The Columbus Dispatch. The trend appears to have continued so far this year, with 107,462 deaths and 100,781 births reported to date.
Deaths haven’t surpassed births in the 112 years since the state began compiling data in 1909, the newspaper reported. The state’s birth rate has been declining for years while the number of deaths has risen, though data shows that the COVID-19 pandemic accelerated the shift.
“It doesn’t surprise me at all,” Joseph Gastaldo, MD, the medical director of infectious diseases for OhioHealth, told the newspaper.
Ohio’s birth rate fell by 4% in 2020, which defied public expectations about a pandemic birth boom. In addition, the state reported 13,927 COVID-19 deaths throughout the year.
“It’s COVID, clearly,” he noted.
Alabama also recorded more deaths than births for the first time last year, according to The New York Times. The state reported 64,714 deaths and 57,641 births in 2020.
“Our state literally shrunk in 2020,” Scott Harris, MD, the state health officer for Alabama, said at a news conference in September.
The state had never recorded a gap that large, even during World War I, World War II, and the 1918 flu pandemic, he said. Alabama has kept records on the numbers since 1900.
“We’ve never had a time when deaths exceeded births,” Dr. Harris said.
In fact, about half of U.S. states reported death rates higher than birth rates in 2020, according to a recent study from researchers at the University of New Hampshire. In 2019, only five states --Maine, New Hampshire, Rhode Island, Vermont, and West Virginia -- reported more deaths than births.
In 2020, the United States reported a record of nearly 3.4 million deaths, which was 18% more than in 2019, the researchers found. COVID-19 was the primary reason for the increase in deaths, accounting for about 11% of total deaths. Meanwhile, births dropped by 4% to about 3.6 million.
The surplus of births over deaths added 229,000 people to the U.S. population in 2020, as compared to 892,000 in 2019, which means the country’s population growth slowed last year. The decline, paired with lower immigration rates during the pandemic, led to the smallest annual percentage population gain in at least 100 years.
“Deaths will likely exceed births again in many states in 2021,” Kenneth Johnson, PhD, a senior demographer and study author, wrote in a statement.
“How large or protracted these fertility declines and mortality increases will be remains to be seen, but they have already dramatically reduced population growth in the United States,” he said.
A version of this article first appeared on WebMD.com.
In 2020, around 143,661 Ohioans died and 129,313 Ohioans were born, according to The Columbus Dispatch. The trend appears to have continued so far this year, with 107,462 deaths and 100,781 births reported to date.
Deaths haven’t surpassed births in the 112 years since the state began compiling data in 1909, the newspaper reported. The state’s birth rate has been declining for years while the number of deaths has risen, though data shows that the COVID-19 pandemic accelerated the shift.
“It doesn’t surprise me at all,” Joseph Gastaldo, MD, the medical director of infectious diseases for OhioHealth, told the newspaper.
Ohio’s birth rate fell by 4% in 2020, which defied public expectations about a pandemic birth boom. In addition, the state reported 13,927 COVID-19 deaths throughout the year.
“It’s COVID, clearly,” he noted.
Alabama also recorded more deaths than births for the first time last year, according to The New York Times. The state reported 64,714 deaths and 57,641 births in 2020.
“Our state literally shrunk in 2020,” Scott Harris, MD, the state health officer for Alabama, said at a news conference in September.
The state had never recorded a gap that large, even during World War I, World War II, and the 1918 flu pandemic, he said. Alabama has kept records on the numbers since 1900.
“We’ve never had a time when deaths exceeded births,” Dr. Harris said.
In fact, about half of U.S. states reported death rates higher than birth rates in 2020, according to a recent study from researchers at the University of New Hampshire. In 2019, only five states --Maine, New Hampshire, Rhode Island, Vermont, and West Virginia -- reported more deaths than births.
In 2020, the United States reported a record of nearly 3.4 million deaths, which was 18% more than in 2019, the researchers found. COVID-19 was the primary reason for the increase in deaths, accounting for about 11% of total deaths. Meanwhile, births dropped by 4% to about 3.6 million.
The surplus of births over deaths added 229,000 people to the U.S. population in 2020, as compared to 892,000 in 2019, which means the country’s population growth slowed last year. The decline, paired with lower immigration rates during the pandemic, led to the smallest annual percentage population gain in at least 100 years.
“Deaths will likely exceed births again in many states in 2021,” Kenneth Johnson, PhD, a senior demographer and study author, wrote in a statement.
“How large or protracted these fertility declines and mortality increases will be remains to be seen, but they have already dramatically reduced population growth in the United States,” he said.
A version of this article first appeared on WebMD.com.



