User login
Positive phase 3 topline results for early Parkinson’s drug
Topline results from a phase 3 trial of P2B001, a fixed-dose combination of extended release (ER) formulations of pramipexole and rasagiline, showed it was superior to its individual components as a first-line treatment for early Parkinson’s disease.
Study participants also reported less daytime sleepiness with P2B001, according to a statement from the drug’s manufacturer.
while mitigating the side effects typically associated with this class of medicine such as somnolence, orthostatic hypotension, and hallucinations,” Sheila Oren, MD, chief executive officer of Pharma Two B, said in a statement.
“This is important for PD patients of all ages and is critical for the elderly, who typically do not tolerate side effects of dopamine agonists,” Dr. Oren added.
Promising results
The 12-week, international, randomized, double-blind trial was designed to study the efficacy, safety, and tolerability of P2B001 compared to its individual components and to a calibration arm of pramipexole ER in 544 patients with early PD.
Participants received P2B001, a once-daily ER combination product composed of pramipexole 0.6 mg and rasagiline 0.75 mg; pramipexole ER capsule 0.6 mg once daily; rasagiline ER capsule 0.75 mg once daily; or the currently marketed product pramipexole ER capsules titrated to an optimal dose for each individual patient (1.5-4.5 mg).
The adjusted mean change from baseline in total Unified Parkinson’s Disease Rating Scale (UPDRS) score was –2.66 points for P2B001 versus pramipexole (P = .0018) and –3.30 points for P2B001 versus rasagiline (P = .0001). There was no significant difference in UPDRS scores between P2B001 and pramipexole ER.
The adjusted mean change from baseline in the Epworth Sleepiness Scale score for P2B001 versus pramipexole ER was –2.66 points (P < .0001).
Treatment-related adverse events were mostly mild or moderate and were similar among groups.
“The initiation of treatment of patients with Parkinson’s disease represents an area of unmet need due to the side effects associated with current treatments,” Warren Olanow, MD, professor emeritus of neurology and neuroscience at the Icahn School of Medicine at Mount Sinai in New York, said in a statement from the manufacturer.
“Based on the data from this well-designed, rigorous, active-controlled study, P2B001 has the potential to become a leading treatment option for PD, particularly as first line therapy for early-stage patients of all ages,” Dr. Olanow added.
The company plans to file a new drug application in 2022.
A version of this article first appeared on Medscape.com.
Topline results from a phase 3 trial of P2B001, a fixed-dose combination of extended release (ER) formulations of pramipexole and rasagiline, showed it was superior to its individual components as a first-line treatment for early Parkinson’s disease.
Study participants also reported less daytime sleepiness with P2B001, according to a statement from the drug’s manufacturer.
while mitigating the side effects typically associated with this class of medicine such as somnolence, orthostatic hypotension, and hallucinations,” Sheila Oren, MD, chief executive officer of Pharma Two B, said in a statement.
“This is important for PD patients of all ages and is critical for the elderly, who typically do not tolerate side effects of dopamine agonists,” Dr. Oren added.
Promising results
The 12-week, international, randomized, double-blind trial was designed to study the efficacy, safety, and tolerability of P2B001 compared to its individual components and to a calibration arm of pramipexole ER in 544 patients with early PD.
Participants received P2B001, a once-daily ER combination product composed of pramipexole 0.6 mg and rasagiline 0.75 mg; pramipexole ER capsule 0.6 mg once daily; rasagiline ER capsule 0.75 mg once daily; or the currently marketed product pramipexole ER capsules titrated to an optimal dose for each individual patient (1.5-4.5 mg).
The adjusted mean change from baseline in total Unified Parkinson’s Disease Rating Scale (UPDRS) score was –2.66 points for P2B001 versus pramipexole (P = .0018) and –3.30 points for P2B001 versus rasagiline (P = .0001). There was no significant difference in UPDRS scores between P2B001 and pramipexole ER.
The adjusted mean change from baseline in the Epworth Sleepiness Scale score for P2B001 versus pramipexole ER was –2.66 points (P < .0001).
Treatment-related adverse events were mostly mild or moderate and were similar among groups.
“The initiation of treatment of patients with Parkinson’s disease represents an area of unmet need due to the side effects associated with current treatments,” Warren Olanow, MD, professor emeritus of neurology and neuroscience at the Icahn School of Medicine at Mount Sinai in New York, said in a statement from the manufacturer.
“Based on the data from this well-designed, rigorous, active-controlled study, P2B001 has the potential to become a leading treatment option for PD, particularly as first line therapy for early-stage patients of all ages,” Dr. Olanow added.
The company plans to file a new drug application in 2022.
A version of this article first appeared on Medscape.com.
Topline results from a phase 3 trial of P2B001, a fixed-dose combination of extended release (ER) formulations of pramipexole and rasagiline, showed it was superior to its individual components as a first-line treatment for early Parkinson’s disease.
Study participants also reported less daytime sleepiness with P2B001, according to a statement from the drug’s manufacturer.
while mitigating the side effects typically associated with this class of medicine such as somnolence, orthostatic hypotension, and hallucinations,” Sheila Oren, MD, chief executive officer of Pharma Two B, said in a statement.
“This is important for PD patients of all ages and is critical for the elderly, who typically do not tolerate side effects of dopamine agonists,” Dr. Oren added.
Promising results
The 12-week, international, randomized, double-blind trial was designed to study the efficacy, safety, and tolerability of P2B001 compared to its individual components and to a calibration arm of pramipexole ER in 544 patients with early PD.
Participants received P2B001, a once-daily ER combination product composed of pramipexole 0.6 mg and rasagiline 0.75 mg; pramipexole ER capsule 0.6 mg once daily; rasagiline ER capsule 0.75 mg once daily; or the currently marketed product pramipexole ER capsules titrated to an optimal dose for each individual patient (1.5-4.5 mg).
The adjusted mean change from baseline in total Unified Parkinson’s Disease Rating Scale (UPDRS) score was –2.66 points for P2B001 versus pramipexole (P = .0018) and –3.30 points for P2B001 versus rasagiline (P = .0001). There was no significant difference in UPDRS scores between P2B001 and pramipexole ER.
The adjusted mean change from baseline in the Epworth Sleepiness Scale score for P2B001 versus pramipexole ER was –2.66 points (P < .0001).
Treatment-related adverse events were mostly mild or moderate and were similar among groups.
“The initiation of treatment of patients with Parkinson’s disease represents an area of unmet need due to the side effects associated with current treatments,” Warren Olanow, MD, professor emeritus of neurology and neuroscience at the Icahn School of Medicine at Mount Sinai in New York, said in a statement from the manufacturer.
“Based on the data from this well-designed, rigorous, active-controlled study, P2B001 has the potential to become a leading treatment option for PD, particularly as first line therapy for early-stage patients of all ages,” Dr. Olanow added.
The company plans to file a new drug application in 2022.
A version of this article first appeared on Medscape.com.
Formaldehyde exposure tied to cognitive impairment
Long-term exposure to formaldehyde on the job is linked to cognitive impairment down the road, new research suggests.
In a large observational study of adults aged 45-70 years, researchers found a 17% higher risk for cognitive problems in those with occupational formaldehyde exposure – and higher risks for those with longer duration of exposure.
“The effect of formaldehyde on the brain has been previously shown mainly in animal experiments, but very few studies have been done on humans,” lead author Noemie Letellier, PhD, Institute for Neurosciences of Montpellier, University of Montpellier (France), said in an interview.
“Our results show that being or having been occupationally exposed to formaldehyde is associated with cognitive impairment in a relatively young population,” Dr. Letellier said.
The findings were published online Dec. 22, 2021, in the journal Neurology.
Dose-effect relationship
The investigators assessed a representative sample of 75,322 adults in France (median age, 57.5 years; 53% women). All were part of the CONSTANCES cohort, an observational cohort with a focus on occupational and environmental factors.
A total of 6,026 participants (8%) were exposed to formaldehyde during their careers. Their occupations included nurses, caregivers, medical technicians, workers in the textile, chemistry and metal industries, carpenters, and cleaners.
The researchers calculated lifetime formaldehyde exposure using a French job-exposure matrix created to estimate a person’s exposure to potential health hazards in different occupations.
Individuals were divided into three equal groups according to their years of exposure to formaldehyde. “Low” was considered to be 6 or fewer years of exposure, “medium” was 7-21 years, and “high” was 22 or more years.
Participants were also split into three groups according to their cumulative exposure (total lifetime formaldehyde exposure based on the probability, intensity, and frequency of exposure).
Prevention efforts needed
After adjusting for age, sex, education and other confounders, participants exposed to formaldehyde were at higher risk for global cognitive impairment (adjusted relative risk, 1.17; 95% confidence interval, 1.1-1.2).
Longer duration of exposure and high cumulative lifetime exposure were associated with worse cognitive impairment, “with a dose-effect relationship for exposure duration,” the researchers reported.
Those exposed to formaldehyde for 22 years or more had a 21% higher risk of global cognitive impairment and workers with the highest cumulative exposure had a 19% higher risk of cognitive impairment, compared with workers with no exposure.
Although workers with recent exposure showed higher cognitive impairment, “time may not fully attenuate formaldehyde-associated cognitive deficits, especially in highly exposed but also in moderately exposed workers,” the researchers wrote.
They caution that their findings only show an association and does not prove that exposure to formaldehyde causes cognitive impairment.
Nonetheless, Dr. Letellier encourages health care providers to “be aware of lifetime occupational exposure to target prevention efforts to the identified occupational groups.” This especially includes the care sector where the most people are exposed to formaldehyde, such as nurses, caregivers, and medical technicians.
“Despite the restrictions on the use of formaldehyde due to the better knowledge of its toxicity, especially its carcinogenic effect, formaldehyde is still widely used in many sectors. These results encourage prevention efforts to further limit worker exposure to formaldehyde,” Dr. Letellier said.
Relevant to health care workers
Commenting on the study, Shaheen E. Lakhan, MD, PhD, a neurologist in Newton, Mass., said in an interview that exposure to some degree of formaldehyde is found in every home and workplace, “from the floors to furniture.”
“If you have cigarette smoke in the environment, your exposure rises sharply. When limiting your exposure, it’s not only cancer that you are preventing, but also your brain health,” added Dr. Lakhan, who was not involved with the research.
He said the disturbances in cognitive function noted in the current study were “particularly relevant to health care workers, given the use of formaldehyde in sterilization, tissue pathology processing, and embalming.”
“Interestingly, with only past exposure, there seems to be some degree of cognitive recovery,” but it does not return to a level before any exposure when corrected for age and other factors, Dr. Lakhan said.
Some caveats should also be noted, he pointed out. The study included a French population, but regulators such as the U.S. Occupational Safety and Health Administration and the California Office of Environmental Health Hazard Assessment have strict standards on formaldehyde use in a variety of work settings.
On the flip side, given the COVID-19 pandemic, there has been greater use of chemical disinfectants in and out the workplace, some of which contain formaldehyde, Dr. Lakhan said.
In addition, he noted the study assessed data from 1950 to 2018, so prepandemic.
“A word of advice from a brain doc: Check with your employer on the level of occupational exposure to formaldehyde, heavy metals, and other toxic substances – and cross-reference with your local environmental standards,” Dr. Lakhan concluded.
The research was supported by a grant from the French Agency for Food, Environmental, and Occupational Health & Safety. The investigators and Dr. Lakhan disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Long-term exposure to formaldehyde on the job is linked to cognitive impairment down the road, new research suggests.
In a large observational study of adults aged 45-70 years, researchers found a 17% higher risk for cognitive problems in those with occupational formaldehyde exposure – and higher risks for those with longer duration of exposure.
“The effect of formaldehyde on the brain has been previously shown mainly in animal experiments, but very few studies have been done on humans,” lead author Noemie Letellier, PhD, Institute for Neurosciences of Montpellier, University of Montpellier (France), said in an interview.
“Our results show that being or having been occupationally exposed to formaldehyde is associated with cognitive impairment in a relatively young population,” Dr. Letellier said.
The findings were published online Dec. 22, 2021, in the journal Neurology.
Dose-effect relationship
The investigators assessed a representative sample of 75,322 adults in France (median age, 57.5 years; 53% women). All were part of the CONSTANCES cohort, an observational cohort with a focus on occupational and environmental factors.
A total of 6,026 participants (8%) were exposed to formaldehyde during their careers. Their occupations included nurses, caregivers, medical technicians, workers in the textile, chemistry and metal industries, carpenters, and cleaners.
The researchers calculated lifetime formaldehyde exposure using a French job-exposure matrix created to estimate a person’s exposure to potential health hazards in different occupations.
Individuals were divided into three equal groups according to their years of exposure to formaldehyde. “Low” was considered to be 6 or fewer years of exposure, “medium” was 7-21 years, and “high” was 22 or more years.
Participants were also split into three groups according to their cumulative exposure (total lifetime formaldehyde exposure based on the probability, intensity, and frequency of exposure).
Prevention efforts needed
After adjusting for age, sex, education and other confounders, participants exposed to formaldehyde were at higher risk for global cognitive impairment (adjusted relative risk, 1.17; 95% confidence interval, 1.1-1.2).
Longer duration of exposure and high cumulative lifetime exposure were associated with worse cognitive impairment, “with a dose-effect relationship for exposure duration,” the researchers reported.
Those exposed to formaldehyde for 22 years or more had a 21% higher risk of global cognitive impairment and workers with the highest cumulative exposure had a 19% higher risk of cognitive impairment, compared with workers with no exposure.
Although workers with recent exposure showed higher cognitive impairment, “time may not fully attenuate formaldehyde-associated cognitive deficits, especially in highly exposed but also in moderately exposed workers,” the researchers wrote.
They caution that their findings only show an association and does not prove that exposure to formaldehyde causes cognitive impairment.
Nonetheless, Dr. Letellier encourages health care providers to “be aware of lifetime occupational exposure to target prevention efforts to the identified occupational groups.” This especially includes the care sector where the most people are exposed to formaldehyde, such as nurses, caregivers, and medical technicians.
“Despite the restrictions on the use of formaldehyde due to the better knowledge of its toxicity, especially its carcinogenic effect, formaldehyde is still widely used in many sectors. These results encourage prevention efforts to further limit worker exposure to formaldehyde,” Dr. Letellier said.
Relevant to health care workers
Commenting on the study, Shaheen E. Lakhan, MD, PhD, a neurologist in Newton, Mass., said in an interview that exposure to some degree of formaldehyde is found in every home and workplace, “from the floors to furniture.”
“If you have cigarette smoke in the environment, your exposure rises sharply. When limiting your exposure, it’s not only cancer that you are preventing, but also your brain health,” added Dr. Lakhan, who was not involved with the research.
He said the disturbances in cognitive function noted in the current study were “particularly relevant to health care workers, given the use of formaldehyde in sterilization, tissue pathology processing, and embalming.”
“Interestingly, with only past exposure, there seems to be some degree of cognitive recovery,” but it does not return to a level before any exposure when corrected for age and other factors, Dr. Lakhan said.
Some caveats should also be noted, he pointed out. The study included a French population, but regulators such as the U.S. Occupational Safety and Health Administration and the California Office of Environmental Health Hazard Assessment have strict standards on formaldehyde use in a variety of work settings.
On the flip side, given the COVID-19 pandemic, there has been greater use of chemical disinfectants in and out the workplace, some of which contain formaldehyde, Dr. Lakhan said.
In addition, he noted the study assessed data from 1950 to 2018, so prepandemic.
“A word of advice from a brain doc: Check with your employer on the level of occupational exposure to formaldehyde, heavy metals, and other toxic substances – and cross-reference with your local environmental standards,” Dr. Lakhan concluded.
The research was supported by a grant from the French Agency for Food, Environmental, and Occupational Health & Safety. The investigators and Dr. Lakhan disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Long-term exposure to formaldehyde on the job is linked to cognitive impairment down the road, new research suggests.
In a large observational study of adults aged 45-70 years, researchers found a 17% higher risk for cognitive problems in those with occupational formaldehyde exposure – and higher risks for those with longer duration of exposure.
“The effect of formaldehyde on the brain has been previously shown mainly in animal experiments, but very few studies have been done on humans,” lead author Noemie Letellier, PhD, Institute for Neurosciences of Montpellier, University of Montpellier (France), said in an interview.
“Our results show that being or having been occupationally exposed to formaldehyde is associated with cognitive impairment in a relatively young population,” Dr. Letellier said.
The findings were published online Dec. 22, 2021, in the journal Neurology.
Dose-effect relationship
The investigators assessed a representative sample of 75,322 adults in France (median age, 57.5 years; 53% women). All were part of the CONSTANCES cohort, an observational cohort with a focus on occupational and environmental factors.
A total of 6,026 participants (8%) were exposed to formaldehyde during their careers. Their occupations included nurses, caregivers, medical technicians, workers in the textile, chemistry and metal industries, carpenters, and cleaners.
The researchers calculated lifetime formaldehyde exposure using a French job-exposure matrix created to estimate a person’s exposure to potential health hazards in different occupations.
Individuals were divided into three equal groups according to their years of exposure to formaldehyde. “Low” was considered to be 6 or fewer years of exposure, “medium” was 7-21 years, and “high” was 22 or more years.
Participants were also split into three groups according to their cumulative exposure (total lifetime formaldehyde exposure based on the probability, intensity, and frequency of exposure).
Prevention efforts needed
After adjusting for age, sex, education and other confounders, participants exposed to formaldehyde were at higher risk for global cognitive impairment (adjusted relative risk, 1.17; 95% confidence interval, 1.1-1.2).
Longer duration of exposure and high cumulative lifetime exposure were associated with worse cognitive impairment, “with a dose-effect relationship for exposure duration,” the researchers reported.
Those exposed to formaldehyde for 22 years or more had a 21% higher risk of global cognitive impairment and workers with the highest cumulative exposure had a 19% higher risk of cognitive impairment, compared with workers with no exposure.
Although workers with recent exposure showed higher cognitive impairment, “time may not fully attenuate formaldehyde-associated cognitive deficits, especially in highly exposed but also in moderately exposed workers,” the researchers wrote.
They caution that their findings only show an association and does not prove that exposure to formaldehyde causes cognitive impairment.
Nonetheless, Dr. Letellier encourages health care providers to “be aware of lifetime occupational exposure to target prevention efforts to the identified occupational groups.” This especially includes the care sector where the most people are exposed to formaldehyde, such as nurses, caregivers, and medical technicians.
“Despite the restrictions on the use of formaldehyde due to the better knowledge of its toxicity, especially its carcinogenic effect, formaldehyde is still widely used in many sectors. These results encourage prevention efforts to further limit worker exposure to formaldehyde,” Dr. Letellier said.
Relevant to health care workers
Commenting on the study, Shaheen E. Lakhan, MD, PhD, a neurologist in Newton, Mass., said in an interview that exposure to some degree of formaldehyde is found in every home and workplace, “from the floors to furniture.”
“If you have cigarette smoke in the environment, your exposure rises sharply. When limiting your exposure, it’s not only cancer that you are preventing, but also your brain health,” added Dr. Lakhan, who was not involved with the research.
He said the disturbances in cognitive function noted in the current study were “particularly relevant to health care workers, given the use of formaldehyde in sterilization, tissue pathology processing, and embalming.”
“Interestingly, with only past exposure, there seems to be some degree of cognitive recovery,” but it does not return to a level before any exposure when corrected for age and other factors, Dr. Lakhan said.
Some caveats should also be noted, he pointed out. The study included a French population, but regulators such as the U.S. Occupational Safety and Health Administration and the California Office of Environmental Health Hazard Assessment have strict standards on formaldehyde use in a variety of work settings.
On the flip side, given the COVID-19 pandemic, there has been greater use of chemical disinfectants in and out the workplace, some of which contain formaldehyde, Dr. Lakhan said.
In addition, he noted the study assessed data from 1950 to 2018, so prepandemic.
“A word of advice from a brain doc: Check with your employer on the level of occupational exposure to formaldehyde, heavy metals, and other toxic substances – and cross-reference with your local environmental standards,” Dr. Lakhan concluded.
The research was supported by a grant from the French Agency for Food, Environmental, and Occupational Health & Safety. The investigators and Dr. Lakhan disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NEUROLOGY
Epilepsy in older adults: Misdiagnosis and case complexity are common
, a neurologist told an audience at the annual meeting of the American Epilepsy Society. She urged colleagues to focus on possible interactions with other neurological conditions, consider various complicating factors, and embrace a team strategy.
“There are lots of nuances,” said Rebecca O’Dwyer, MD, an adult epilepsy specialist with Rush Epilepsy Center in Chicago. “It takes a lot of time and requires a multidisciplinary approach. Taking care of older individuals with epilepsy truly is a team sport.”
According to a 2014 report highlighted by Dr. O’Dwyer, “nearly 25% of new-onset seizures occur after age 65. The incidence of epilepsy in this age group is almost twice the rate in children, and in people over age 80, it is triple the rate in children.”
Research suggests it can take up to 2 years to correctly diagnose epilepsy in older people, Dr. O’Dwyer said, and nearly two-thirds of cases may be misdiagnosed. “Some of it is just limited awareness. There’s this perception in the public that epilepsy is something that occurs in younger adults or young children, and that when you come to a certain age, you cannot have epilepsy. Also, there are differences in the clinical manifestations of their seizures, and many comorbid possibilities could also present in similar fashion to epilepsy. Some of our usual tools that we use to come to the diagnosis such as EEG are also known to be less sensitive in this age group.”
According to the 2014 report, research finds that the elderly are much more likely than young adults to have postictal sleepiness or unresponsiveness and seizures manifesting as brief moments of subtle confusion. They’re much less likely to have epileptic aura and generalized tonic seizures.
“An epileptic seizure in an older adult tends to be less dramatic with fewer motor manifestations, and they often tend to be monophasic. They may be so subtle that they’re missed by family members and other medical providers,” Dr. O’Dwyer said. “I had a patient whose seizure consisted of her tapping her left shoulder. She had been doing this for at least 6 months, and she came to my clinic after her daughter realized that she was a little confused afterward. She’d already seen a behavioral neurologist and been given the diagnosis of dementia. We were fortunate enough to catch one of these episodes while we were doing an EEG, and we diagnosed her with focal epilepsy. With one antiseizure medication, we stopped the seizures, and her memory came back.”
Make sure to take detailed histories and keep an eye out for descriptions of behaviors that are episodic but perhaps not typical of seizures, she said.
Epilepsy can be misdiagnosed as a variety of conditions, she said, such as syncope, Alzheimer’s disease, stroke, Parkinson’s disease, and atrial fibrillation. “When you do diagnose somebody older with new-onset epilepsy, you should work them up for a stroke. Because we know that within the first 4 weeks after their first seizure the likelihood that they could have a stroke is three times higher.”
It’s also possible that neurological conditions can be followed by new-onset epilepsy, she said, making dementia even worse. Low-dose antiepileptic drugs can be helpful in these patients.
But seniors are especially vulnerable to side effects of antiepileptic drugs such as sedation, dizziness, and cardiac-conduction abnormalities. “You must adhere to the mantra of going low and going slow because they are exquisitely susceptible,” Dr. O’Dwyer said.
She recommends lamotrigine, which is well tolerated with helpful mood-stabilizing effects, and levetiracetam, which attenuates cognitive decline in dementia but may cause side effects such as irritable mood. Zonisamide is showing promise in patients with parkinsonian syndromes, she said, and it may be helpful to maximize drugs that patients are already taking such as gabapentin or pregabalin.
Finally, Dr. O’Dwyer urged colleagues to work in teams that include caregivers, primary care doctors, social workers, and pharmacists. “Sometimes in all this,” she said, “my job is the easiest.”
Dr. O’Dwyer discloses research support from the Shapiro Foundation.
, a neurologist told an audience at the annual meeting of the American Epilepsy Society. She urged colleagues to focus on possible interactions with other neurological conditions, consider various complicating factors, and embrace a team strategy.
“There are lots of nuances,” said Rebecca O’Dwyer, MD, an adult epilepsy specialist with Rush Epilepsy Center in Chicago. “It takes a lot of time and requires a multidisciplinary approach. Taking care of older individuals with epilepsy truly is a team sport.”
According to a 2014 report highlighted by Dr. O’Dwyer, “nearly 25% of new-onset seizures occur after age 65. The incidence of epilepsy in this age group is almost twice the rate in children, and in people over age 80, it is triple the rate in children.”
Research suggests it can take up to 2 years to correctly diagnose epilepsy in older people, Dr. O’Dwyer said, and nearly two-thirds of cases may be misdiagnosed. “Some of it is just limited awareness. There’s this perception in the public that epilepsy is something that occurs in younger adults or young children, and that when you come to a certain age, you cannot have epilepsy. Also, there are differences in the clinical manifestations of their seizures, and many comorbid possibilities could also present in similar fashion to epilepsy. Some of our usual tools that we use to come to the diagnosis such as EEG are also known to be less sensitive in this age group.”
According to the 2014 report, research finds that the elderly are much more likely than young adults to have postictal sleepiness or unresponsiveness and seizures manifesting as brief moments of subtle confusion. They’re much less likely to have epileptic aura and generalized tonic seizures.
“An epileptic seizure in an older adult tends to be less dramatic with fewer motor manifestations, and they often tend to be monophasic. They may be so subtle that they’re missed by family members and other medical providers,” Dr. O’Dwyer said. “I had a patient whose seizure consisted of her tapping her left shoulder. She had been doing this for at least 6 months, and she came to my clinic after her daughter realized that she was a little confused afterward. She’d already seen a behavioral neurologist and been given the diagnosis of dementia. We were fortunate enough to catch one of these episodes while we were doing an EEG, and we diagnosed her with focal epilepsy. With one antiseizure medication, we stopped the seizures, and her memory came back.”
Make sure to take detailed histories and keep an eye out for descriptions of behaviors that are episodic but perhaps not typical of seizures, she said.
Epilepsy can be misdiagnosed as a variety of conditions, she said, such as syncope, Alzheimer’s disease, stroke, Parkinson’s disease, and atrial fibrillation. “When you do diagnose somebody older with new-onset epilepsy, you should work them up for a stroke. Because we know that within the first 4 weeks after their first seizure the likelihood that they could have a stroke is three times higher.”
It’s also possible that neurological conditions can be followed by new-onset epilepsy, she said, making dementia even worse. Low-dose antiepileptic drugs can be helpful in these patients.
But seniors are especially vulnerable to side effects of antiepileptic drugs such as sedation, dizziness, and cardiac-conduction abnormalities. “You must adhere to the mantra of going low and going slow because they are exquisitely susceptible,” Dr. O’Dwyer said.
She recommends lamotrigine, which is well tolerated with helpful mood-stabilizing effects, and levetiracetam, which attenuates cognitive decline in dementia but may cause side effects such as irritable mood. Zonisamide is showing promise in patients with parkinsonian syndromes, she said, and it may be helpful to maximize drugs that patients are already taking such as gabapentin or pregabalin.
Finally, Dr. O’Dwyer urged colleagues to work in teams that include caregivers, primary care doctors, social workers, and pharmacists. “Sometimes in all this,” she said, “my job is the easiest.”
Dr. O’Dwyer discloses research support from the Shapiro Foundation.
, a neurologist told an audience at the annual meeting of the American Epilepsy Society. She urged colleagues to focus on possible interactions with other neurological conditions, consider various complicating factors, and embrace a team strategy.
“There are lots of nuances,” said Rebecca O’Dwyer, MD, an adult epilepsy specialist with Rush Epilepsy Center in Chicago. “It takes a lot of time and requires a multidisciplinary approach. Taking care of older individuals with epilepsy truly is a team sport.”
According to a 2014 report highlighted by Dr. O’Dwyer, “nearly 25% of new-onset seizures occur after age 65. The incidence of epilepsy in this age group is almost twice the rate in children, and in people over age 80, it is triple the rate in children.”
Research suggests it can take up to 2 years to correctly diagnose epilepsy in older people, Dr. O’Dwyer said, and nearly two-thirds of cases may be misdiagnosed. “Some of it is just limited awareness. There’s this perception in the public that epilepsy is something that occurs in younger adults or young children, and that when you come to a certain age, you cannot have epilepsy. Also, there are differences in the clinical manifestations of their seizures, and many comorbid possibilities could also present in similar fashion to epilepsy. Some of our usual tools that we use to come to the diagnosis such as EEG are also known to be less sensitive in this age group.”
According to the 2014 report, research finds that the elderly are much more likely than young adults to have postictal sleepiness or unresponsiveness and seizures manifesting as brief moments of subtle confusion. They’re much less likely to have epileptic aura and generalized tonic seizures.
“An epileptic seizure in an older adult tends to be less dramatic with fewer motor manifestations, and they often tend to be monophasic. They may be so subtle that they’re missed by family members and other medical providers,” Dr. O’Dwyer said. “I had a patient whose seizure consisted of her tapping her left shoulder. She had been doing this for at least 6 months, and she came to my clinic after her daughter realized that she was a little confused afterward. She’d already seen a behavioral neurologist and been given the diagnosis of dementia. We were fortunate enough to catch one of these episodes while we were doing an EEG, and we diagnosed her with focal epilepsy. With one antiseizure medication, we stopped the seizures, and her memory came back.”
Make sure to take detailed histories and keep an eye out for descriptions of behaviors that are episodic but perhaps not typical of seizures, she said.
Epilepsy can be misdiagnosed as a variety of conditions, she said, such as syncope, Alzheimer’s disease, stroke, Parkinson’s disease, and atrial fibrillation. “When you do diagnose somebody older with new-onset epilepsy, you should work them up for a stroke. Because we know that within the first 4 weeks after their first seizure the likelihood that they could have a stroke is three times higher.”
It’s also possible that neurological conditions can be followed by new-onset epilepsy, she said, making dementia even worse. Low-dose antiepileptic drugs can be helpful in these patients.
But seniors are especially vulnerable to side effects of antiepileptic drugs such as sedation, dizziness, and cardiac-conduction abnormalities. “You must adhere to the mantra of going low and going slow because they are exquisitely susceptible,” Dr. O’Dwyer said.
She recommends lamotrigine, which is well tolerated with helpful mood-stabilizing effects, and levetiracetam, which attenuates cognitive decline in dementia but may cause side effects such as irritable mood. Zonisamide is showing promise in patients with parkinsonian syndromes, she said, and it may be helpful to maximize drugs that patients are already taking such as gabapentin or pregabalin.
Finally, Dr. O’Dwyer urged colleagues to work in teams that include caregivers, primary care doctors, social workers, and pharmacists. “Sometimes in all this,” she said, “my job is the easiest.”
Dr. O’Dwyer discloses research support from the Shapiro Foundation.
FROM AES 2021
Even light physical activity linked to lower dementia risk
Older adults who participate in even light physical activity (LPA) may have a lower risk of developing dementia, new research suggests.
In a retrospective analysis of more than 62,000 individuals aged 65 or older without preexisting dementia, 6% developed dementia.
Compared with inactive individuals, “insufficiently active,” “active,” and “highly active” individuals all had a 10%, 20%, and 28% lower risk for dementia, respectively. And this association was consistent regardless of age, sex, other comorbidities, or after the researchers censored for stroke.
Even the lowest amount of LPA was associated with reduced dementia risk, investigators noted.
“In older adults, an increased physical activity level, including a low amount of LPA, was associated with a reduced risk of dementia,” Minjae Yoon, MD, division of cardiology, Severance Cardiovascular Hospital, Yonsei University, Seoul, South Korea, and colleagues wrote.
“Promotion of LPA might reduce the risk of dementia in older adults,” they added.
The findings were published online in JAMA Network Open.
Reverse causation?
Physical activity has been shown previously to be associated with reduced dementia risk. Current World Health Organization guidelines recommend that adults with normal cognition should engage in PA to reduce their risk for cognitive decline.
However, some studies have not yielded this result, “suggesting that previous findings showing a lower risk of dementia in physically active people could be attributed to reverse causation,” the investigators noted. Additionally, previous research regarding exercise intensity has been “inconsistent” concerning the role of LPA in reducing dementia risk.
Many older adults with frailty and comorbidity cannot perform intense or even moderate PA, therefore “these adults would have to gain the benefits of physical activity from LPA,” the researchers noted.
To clarify the potential association between PA and new-onset dementia, they focused specifically on the “dose-response association” between PA and dementia – especially LPA.
Between 2009 and 2012, the investigators enrolled 62,286 older individuals (60.4% women; mean age, 73.2 years) with available health checkup data from the National Health Insurance Service–Senior Database of Korea. All had no history of dementia.
Leisure-time PA was assessed with self-report questionnaires that used a 7-day recall method and included three questions regarding usual frequency (in days per week):
- Vigorous PA (VPA) for at least 20 minutes
- Moderate-intensity PA (MPA) for at least 30 minutes
- LPA for at least 30 minutes
VPA was defined as “intense exercise that caused severe shortness of breath, MPA was defined as activity causing mild shortness of breath, and LPA was defined as “walking at a slow or leisurely pace.”
PA-related energy expenditure was also calculated in metabolic equivalent (MET) minutes per week by “summing the product of frequency, intensity, and duration,” the investigators noted.
Participants were stratified on the basis of their weekly total PA levels into the following groups:
- Inactive (no LPA beyond basic movements)
- Insufficiently active (less than the recommended target range of 1-499 MET-min/wk)
- Active (meeting the recommended target range of 500-999 MET-min/wk)
- Highly active (exceeding the recommended target range of at least 1,000 MET-min/wk)
Of all participants, 35% were categorized as inactive, 25% were insufficiently active, 24.4% were active, and 15.2% were highly active.
Controversy remains
During the total median follow-up of 42 months, 6% of participants had all-cause dementia. After the researchers excluded the first 2 years, incidence of dementia was 21.6 per 1000 person-years during follow-up.
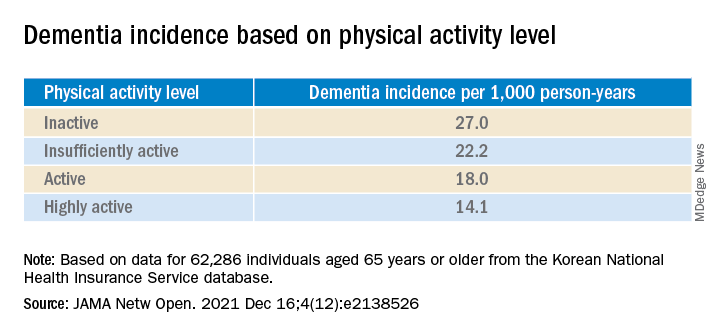
“The cumulative incidence of dementia was associated with a progressively decreasing trend with increasing physical activity” (P = .001 for trend), the investigators reported.
When using a competing-risk multivariable regression model, they found that higher levels of PA were associated with lower risk for dementia, compared with the inactive group.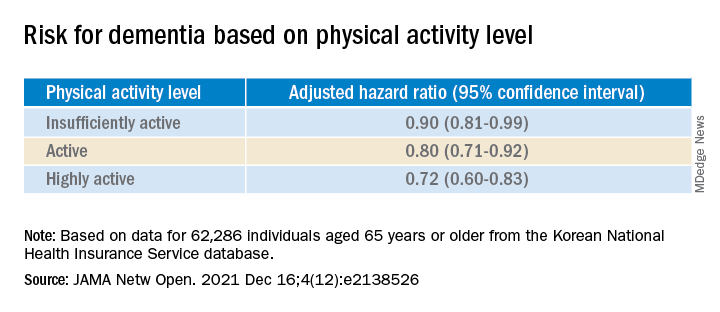
Similar findings were obtained after censoring for stroke, and were consistent for all follow-up periods. In subgroup analysis, the association between PA level and dementia risk remained consistent, regardless of age, sex, and comorbidities.
Even a low amount of LPA (1-299 MET-min/wk) was linked to reduced risk for dementia versus total sedentary behavior (adjusted HR, 0.86; 95% CI, 0.74-0.99).
The investigators noted that some “controversy” remains regarding the possibility of reverse causation and, because their study was observational in nature, “it cannot be used to establish causal relationship.”
Nevertheless, the study had important strengths, including the large number of older adults with available data, the assessment of dose-response association between PA and dementia, and the sensitivity analyses they performed, the researchers added.
Piece of important evidence
Commenting on the findings, Takashi Tarumi, PhD, senior research investigator, National Institute of Advanced Industrial Science and Technology, Ibaraki, Japan, said previous studies have suggested “an inverse association between physical activity and dementia risk, such that older adults performing a higher dose of exercise may have a greater benefit for reducing the dementia risk.”
Dr. Tarumi, an associate editor at the Journal of Alzheimer’s Disease, added the current study “significantly extends our knowledge by showing that dementia risk can also be reduced by light physical activities when they are performed for longer hours.”
This provides “another piece of important evidence” to support clinicians recommending regular physical activity for the prevention of dementia in later life, said Dr. Tarumi, who was not involved with the research.
Also commenting, Martin Underwood, MD, Warwick Medical School, Coventry, England, described the association between reduced physical inactivity and dementia as well established – and noted the current study “appears to confirm earlier observational data showing this relationship.”
The current results have “still not been able to fully exclude the possibility of reverse causation,” said Dr. Underwood, who was also not associated with the study.
However, the finding that more physically active individuals are less likely to develop dementia “only becomes of real interest if we can show that increased physical activity prevents the onset, or slows the progression, of dementia,” he noted.
“To my knowledge this has not yet been established” in randomized clinical trials, Dr. Underwood added.
The study was supported by grants from the Patient-Centered Clinical Research Coordinating Center, funded by the Ministry of Health & Welfare, Republic of Korea; and by a research grant from Yonsei University. One coauthor reported serving as a speaker for Bayer, Bristol-Myers Squibb/Pfizer, Medtronic, and Daiichi-Sankyo, and receiving research funds from Medtronic and Abbott. No other author disclosures were reported. Dr. Tarumi and Dr. Underwood have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Older adults who participate in even light physical activity (LPA) may have a lower risk of developing dementia, new research suggests.
In a retrospective analysis of more than 62,000 individuals aged 65 or older without preexisting dementia, 6% developed dementia.
Compared with inactive individuals, “insufficiently active,” “active,” and “highly active” individuals all had a 10%, 20%, and 28% lower risk for dementia, respectively. And this association was consistent regardless of age, sex, other comorbidities, or after the researchers censored for stroke.
Even the lowest amount of LPA was associated with reduced dementia risk, investigators noted.
“In older adults, an increased physical activity level, including a low amount of LPA, was associated with a reduced risk of dementia,” Minjae Yoon, MD, division of cardiology, Severance Cardiovascular Hospital, Yonsei University, Seoul, South Korea, and colleagues wrote.
“Promotion of LPA might reduce the risk of dementia in older adults,” they added.
The findings were published online in JAMA Network Open.
Reverse causation?
Physical activity has been shown previously to be associated with reduced dementia risk. Current World Health Organization guidelines recommend that adults with normal cognition should engage in PA to reduce their risk for cognitive decline.
However, some studies have not yielded this result, “suggesting that previous findings showing a lower risk of dementia in physically active people could be attributed to reverse causation,” the investigators noted. Additionally, previous research regarding exercise intensity has been “inconsistent” concerning the role of LPA in reducing dementia risk.
Many older adults with frailty and comorbidity cannot perform intense or even moderate PA, therefore “these adults would have to gain the benefits of physical activity from LPA,” the researchers noted.
To clarify the potential association between PA and new-onset dementia, they focused specifically on the “dose-response association” between PA and dementia – especially LPA.
Between 2009 and 2012, the investigators enrolled 62,286 older individuals (60.4% women; mean age, 73.2 years) with available health checkup data from the National Health Insurance Service–Senior Database of Korea. All had no history of dementia.
Leisure-time PA was assessed with self-report questionnaires that used a 7-day recall method and included three questions regarding usual frequency (in days per week):
- Vigorous PA (VPA) for at least 20 minutes
- Moderate-intensity PA (MPA) for at least 30 minutes
- LPA for at least 30 minutes
VPA was defined as “intense exercise that caused severe shortness of breath, MPA was defined as activity causing mild shortness of breath, and LPA was defined as “walking at a slow or leisurely pace.”
PA-related energy expenditure was also calculated in metabolic equivalent (MET) minutes per week by “summing the product of frequency, intensity, and duration,” the investigators noted.
Participants were stratified on the basis of their weekly total PA levels into the following groups:
- Inactive (no LPA beyond basic movements)
- Insufficiently active (less than the recommended target range of 1-499 MET-min/wk)
- Active (meeting the recommended target range of 500-999 MET-min/wk)
- Highly active (exceeding the recommended target range of at least 1,000 MET-min/wk)
Of all participants, 35% were categorized as inactive, 25% were insufficiently active, 24.4% were active, and 15.2% were highly active.
Controversy remains
During the total median follow-up of 42 months, 6% of participants had all-cause dementia. After the researchers excluded the first 2 years, incidence of dementia was 21.6 per 1000 person-years during follow-up.

“The cumulative incidence of dementia was associated with a progressively decreasing trend with increasing physical activity” (P = .001 for trend), the investigators reported.
When using a competing-risk multivariable regression model, they found that higher levels of PA were associated with lower risk for dementia, compared with the inactive group.
Similar findings were obtained after censoring for stroke, and were consistent for all follow-up periods. In subgroup analysis, the association between PA level and dementia risk remained consistent, regardless of age, sex, and comorbidities.
Even a low amount of LPA (1-299 MET-min/wk) was linked to reduced risk for dementia versus total sedentary behavior (adjusted HR, 0.86; 95% CI, 0.74-0.99).
The investigators noted that some “controversy” remains regarding the possibility of reverse causation and, because their study was observational in nature, “it cannot be used to establish causal relationship.”
Nevertheless, the study had important strengths, including the large number of older adults with available data, the assessment of dose-response association between PA and dementia, and the sensitivity analyses they performed, the researchers added.
Piece of important evidence
Commenting on the findings, Takashi Tarumi, PhD, senior research investigator, National Institute of Advanced Industrial Science and Technology, Ibaraki, Japan, said previous studies have suggested “an inverse association between physical activity and dementia risk, such that older adults performing a higher dose of exercise may have a greater benefit for reducing the dementia risk.”
Dr. Tarumi, an associate editor at the Journal of Alzheimer’s Disease, added the current study “significantly extends our knowledge by showing that dementia risk can also be reduced by light physical activities when they are performed for longer hours.”
This provides “another piece of important evidence” to support clinicians recommending regular physical activity for the prevention of dementia in later life, said Dr. Tarumi, who was not involved with the research.
Also commenting, Martin Underwood, MD, Warwick Medical School, Coventry, England, described the association between reduced physical inactivity and dementia as well established – and noted the current study “appears to confirm earlier observational data showing this relationship.”
The current results have “still not been able to fully exclude the possibility of reverse causation,” said Dr. Underwood, who was also not associated with the study.
However, the finding that more physically active individuals are less likely to develop dementia “only becomes of real interest if we can show that increased physical activity prevents the onset, or slows the progression, of dementia,” he noted.
“To my knowledge this has not yet been established” in randomized clinical trials, Dr. Underwood added.
The study was supported by grants from the Patient-Centered Clinical Research Coordinating Center, funded by the Ministry of Health & Welfare, Republic of Korea; and by a research grant from Yonsei University. One coauthor reported serving as a speaker for Bayer, Bristol-Myers Squibb/Pfizer, Medtronic, and Daiichi-Sankyo, and receiving research funds from Medtronic and Abbott. No other author disclosures were reported. Dr. Tarumi and Dr. Underwood have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Older adults who participate in even light physical activity (LPA) may have a lower risk of developing dementia, new research suggests.
In a retrospective analysis of more than 62,000 individuals aged 65 or older without preexisting dementia, 6% developed dementia.
Compared with inactive individuals, “insufficiently active,” “active,” and “highly active” individuals all had a 10%, 20%, and 28% lower risk for dementia, respectively. And this association was consistent regardless of age, sex, other comorbidities, or after the researchers censored for stroke.
Even the lowest amount of LPA was associated with reduced dementia risk, investigators noted.
“In older adults, an increased physical activity level, including a low amount of LPA, was associated with a reduced risk of dementia,” Minjae Yoon, MD, division of cardiology, Severance Cardiovascular Hospital, Yonsei University, Seoul, South Korea, and colleagues wrote.
“Promotion of LPA might reduce the risk of dementia in older adults,” they added.
The findings were published online in JAMA Network Open.
Reverse causation?
Physical activity has been shown previously to be associated with reduced dementia risk. Current World Health Organization guidelines recommend that adults with normal cognition should engage in PA to reduce their risk for cognitive decline.
However, some studies have not yielded this result, “suggesting that previous findings showing a lower risk of dementia in physically active people could be attributed to reverse causation,” the investigators noted. Additionally, previous research regarding exercise intensity has been “inconsistent” concerning the role of LPA in reducing dementia risk.
Many older adults with frailty and comorbidity cannot perform intense or even moderate PA, therefore “these adults would have to gain the benefits of physical activity from LPA,” the researchers noted.
To clarify the potential association between PA and new-onset dementia, they focused specifically on the “dose-response association” between PA and dementia – especially LPA.
Between 2009 and 2012, the investigators enrolled 62,286 older individuals (60.4% women; mean age, 73.2 years) with available health checkup data from the National Health Insurance Service–Senior Database of Korea. All had no history of dementia.
Leisure-time PA was assessed with self-report questionnaires that used a 7-day recall method and included three questions regarding usual frequency (in days per week):
- Vigorous PA (VPA) for at least 20 minutes
- Moderate-intensity PA (MPA) for at least 30 minutes
- LPA for at least 30 minutes
VPA was defined as “intense exercise that caused severe shortness of breath, MPA was defined as activity causing mild shortness of breath, and LPA was defined as “walking at a slow or leisurely pace.”
PA-related energy expenditure was also calculated in metabolic equivalent (MET) minutes per week by “summing the product of frequency, intensity, and duration,” the investigators noted.
Participants were stratified on the basis of their weekly total PA levels into the following groups:
- Inactive (no LPA beyond basic movements)
- Insufficiently active (less than the recommended target range of 1-499 MET-min/wk)
- Active (meeting the recommended target range of 500-999 MET-min/wk)
- Highly active (exceeding the recommended target range of at least 1,000 MET-min/wk)
Of all participants, 35% were categorized as inactive, 25% were insufficiently active, 24.4% were active, and 15.2% were highly active.
Controversy remains
During the total median follow-up of 42 months, 6% of participants had all-cause dementia. After the researchers excluded the first 2 years, incidence of dementia was 21.6 per 1000 person-years during follow-up.

“The cumulative incidence of dementia was associated with a progressively decreasing trend with increasing physical activity” (P = .001 for trend), the investigators reported.
When using a competing-risk multivariable regression model, they found that higher levels of PA were associated with lower risk for dementia, compared with the inactive group.
Similar findings were obtained after censoring for stroke, and were consistent for all follow-up periods. In subgroup analysis, the association between PA level and dementia risk remained consistent, regardless of age, sex, and comorbidities.
Even a low amount of LPA (1-299 MET-min/wk) was linked to reduced risk for dementia versus total sedentary behavior (adjusted HR, 0.86; 95% CI, 0.74-0.99).
The investigators noted that some “controversy” remains regarding the possibility of reverse causation and, because their study was observational in nature, “it cannot be used to establish causal relationship.”
Nevertheless, the study had important strengths, including the large number of older adults with available data, the assessment of dose-response association between PA and dementia, and the sensitivity analyses they performed, the researchers added.
Piece of important evidence
Commenting on the findings, Takashi Tarumi, PhD, senior research investigator, National Institute of Advanced Industrial Science and Technology, Ibaraki, Japan, said previous studies have suggested “an inverse association between physical activity and dementia risk, such that older adults performing a higher dose of exercise may have a greater benefit for reducing the dementia risk.”
Dr. Tarumi, an associate editor at the Journal of Alzheimer’s Disease, added the current study “significantly extends our knowledge by showing that dementia risk can also be reduced by light physical activities when they are performed for longer hours.”
This provides “another piece of important evidence” to support clinicians recommending regular physical activity for the prevention of dementia in later life, said Dr. Tarumi, who was not involved with the research.
Also commenting, Martin Underwood, MD, Warwick Medical School, Coventry, England, described the association between reduced physical inactivity and dementia as well established – and noted the current study “appears to confirm earlier observational data showing this relationship.”
The current results have “still not been able to fully exclude the possibility of reverse causation,” said Dr. Underwood, who was also not associated with the study.
However, the finding that more physically active individuals are less likely to develop dementia “only becomes of real interest if we can show that increased physical activity prevents the onset, or slows the progression, of dementia,” he noted.
“To my knowledge this has not yet been established” in randomized clinical trials, Dr. Underwood added.
The study was supported by grants from the Patient-Centered Clinical Research Coordinating Center, funded by the Ministry of Health & Welfare, Republic of Korea; and by a research grant from Yonsei University. One coauthor reported serving as a speaker for Bayer, Bristol-Myers Squibb/Pfizer, Medtronic, and Daiichi-Sankyo, and receiving research funds from Medtronic and Abbott. No other author disclosures were reported. Dr. Tarumi and Dr. Underwood have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Ophthalmologist who developed medical botox dies at 89
his family confirmed to National Public Radio.
Four decades ago, Alan Brown Scott, MD, a native of Berkeley, Calif., turned the drug, once a deadly poison, into a revolutionary treatment for obscure eye diseases. It later became a well-known blockbuster treatment for reducing the appearance of wrinkles and treating hyperhidrosis (excessive sweating). Other approved medical uses include treatment of overactive bladder and urinary incontinence.
According to the American Society of Plastic Surgeons, its popularity for cosmetic use was boosted further during the pandemic and it was the No. 1 minimally invasive cosmetic procedure performed in 2020. Among the 13.3 million procedures, 4.4 million involved Botox.
According to Bloomberg Businessweek, Ed Schantz, who was working in the military’s biological weapons program, was the one to first send the toxin to Dr. Scott, who wanted to explore its properties for medical use.
The same Bloomberg article also noted that the original botulinum toxin itself “is so powerful that a tiny amount can suffocate a person by paralyzing the muscles used for breathing.”
Dr. Scott was looking for a way to help his patients avoid extensive surgeries.
“Specifically, he was aiming to treat people with strabismus, or cross-eyes, and blepharospasm, which is an uncontrollable closure of eyes. Today, it’s also used as a treatment to help with migraines, hair loss, and drooling,” NPR reported.
The New York Times once described Botox as “medicine’s answer to duct tape.”
Dr. Scott was the executive director of the Smith-Kettlewell Eye Research Institute in San Francisco when he did his pioneering research with botulinum toxin in the 1970s and 1980s, according to a 2002 article in SFGate.
In 1991, Dr. Scott sold the drug to Allergan, when it was called Oculinum. The next year, the name was officially changed to Botox.
In 2002, Dr. Scott told SFGate, when asked about the more popular use for the drug, “I think that’s a charming, slightly frivolous use,” adding, “but it’s not along the lines of what I was into, applications for serious disorders.”
According to Scientific American in 2016, Dr. Scott, then age 83, kept working on the noncosmetic benefits of botulism-toxin injections for eye-related disorders at the Strabismus Research Foundation,
He told Scientific American he was proud that his efforts “are directly helpful to people.”
“There are interesting and difficult problems still to be solved, and I’m a practicing physician and I see them every day,” he said.
Dr. Scott’s daughter, Ann Scott, told NPR: “He definitely loved his work and he was also a really great father.” She said her dad involved his children in his research and work.
She added, “He was a really calm, more of a quiet reserved person,” and said he was committed to teaching his students, many of them international students.
“That was what he really loved,” she said.
Dr. Scott, who died Dec. 16, was in intensive care for the last 10 days from an unspecified illness, his daughter told NPR.
A version of this article first appeared on Medscape.com.
his family confirmed to National Public Radio.
Four decades ago, Alan Brown Scott, MD, a native of Berkeley, Calif., turned the drug, once a deadly poison, into a revolutionary treatment for obscure eye diseases. It later became a well-known blockbuster treatment for reducing the appearance of wrinkles and treating hyperhidrosis (excessive sweating). Other approved medical uses include treatment of overactive bladder and urinary incontinence.
According to the American Society of Plastic Surgeons, its popularity for cosmetic use was boosted further during the pandemic and it was the No. 1 minimally invasive cosmetic procedure performed in 2020. Among the 13.3 million procedures, 4.4 million involved Botox.
According to Bloomberg Businessweek, Ed Schantz, who was working in the military’s biological weapons program, was the one to first send the toxin to Dr. Scott, who wanted to explore its properties for medical use.
The same Bloomberg article also noted that the original botulinum toxin itself “is so powerful that a tiny amount can suffocate a person by paralyzing the muscles used for breathing.”
Dr. Scott was looking for a way to help his patients avoid extensive surgeries.
“Specifically, he was aiming to treat people with strabismus, or cross-eyes, and blepharospasm, which is an uncontrollable closure of eyes. Today, it’s also used as a treatment to help with migraines, hair loss, and drooling,” NPR reported.
The New York Times once described Botox as “medicine’s answer to duct tape.”
Dr. Scott was the executive director of the Smith-Kettlewell Eye Research Institute in San Francisco when he did his pioneering research with botulinum toxin in the 1970s and 1980s, according to a 2002 article in SFGate.
In 1991, Dr. Scott sold the drug to Allergan, when it was called Oculinum. The next year, the name was officially changed to Botox.
In 2002, Dr. Scott told SFGate, when asked about the more popular use for the drug, “I think that’s a charming, slightly frivolous use,” adding, “but it’s not along the lines of what I was into, applications for serious disorders.”
According to Scientific American in 2016, Dr. Scott, then age 83, kept working on the noncosmetic benefits of botulism-toxin injections for eye-related disorders at the Strabismus Research Foundation,
He told Scientific American he was proud that his efforts “are directly helpful to people.”
“There are interesting and difficult problems still to be solved, and I’m a practicing physician and I see them every day,” he said.
Dr. Scott’s daughter, Ann Scott, told NPR: “He definitely loved his work and he was also a really great father.” She said her dad involved his children in his research and work.
She added, “He was a really calm, more of a quiet reserved person,” and said he was committed to teaching his students, many of them international students.
“That was what he really loved,” she said.
Dr. Scott, who died Dec. 16, was in intensive care for the last 10 days from an unspecified illness, his daughter told NPR.
A version of this article first appeared on Medscape.com.
his family confirmed to National Public Radio.
Four decades ago, Alan Brown Scott, MD, a native of Berkeley, Calif., turned the drug, once a deadly poison, into a revolutionary treatment for obscure eye diseases. It later became a well-known blockbuster treatment for reducing the appearance of wrinkles and treating hyperhidrosis (excessive sweating). Other approved medical uses include treatment of overactive bladder and urinary incontinence.
According to the American Society of Plastic Surgeons, its popularity for cosmetic use was boosted further during the pandemic and it was the No. 1 minimally invasive cosmetic procedure performed in 2020. Among the 13.3 million procedures, 4.4 million involved Botox.
According to Bloomberg Businessweek, Ed Schantz, who was working in the military’s biological weapons program, was the one to first send the toxin to Dr. Scott, who wanted to explore its properties for medical use.
The same Bloomberg article also noted that the original botulinum toxin itself “is so powerful that a tiny amount can suffocate a person by paralyzing the muscles used for breathing.”
Dr. Scott was looking for a way to help his patients avoid extensive surgeries.
“Specifically, he was aiming to treat people with strabismus, or cross-eyes, and blepharospasm, which is an uncontrollable closure of eyes. Today, it’s also used as a treatment to help with migraines, hair loss, and drooling,” NPR reported.
The New York Times once described Botox as “medicine’s answer to duct tape.”
Dr. Scott was the executive director of the Smith-Kettlewell Eye Research Institute in San Francisco when he did his pioneering research with botulinum toxin in the 1970s and 1980s, according to a 2002 article in SFGate.
In 1991, Dr. Scott sold the drug to Allergan, when it was called Oculinum. The next year, the name was officially changed to Botox.
In 2002, Dr. Scott told SFGate, when asked about the more popular use for the drug, “I think that’s a charming, slightly frivolous use,” adding, “but it’s not along the lines of what I was into, applications for serious disorders.”
According to Scientific American in 2016, Dr. Scott, then age 83, kept working on the noncosmetic benefits of botulism-toxin injections for eye-related disorders at the Strabismus Research Foundation,
He told Scientific American he was proud that his efforts “are directly helpful to people.”
“There are interesting and difficult problems still to be solved, and I’m a practicing physician and I see them every day,” he said.
Dr. Scott’s daughter, Ann Scott, told NPR: “He definitely loved his work and he was also a really great father.” She said her dad involved his children in his research and work.
She added, “He was a really calm, more of a quiet reserved person,” and said he was committed to teaching his students, many of them international students.
“That was what he really loved,” she said.
Dr. Scott, who died Dec. 16, was in intensive care for the last 10 days from an unspecified illness, his daughter told NPR.
A version of this article first appeared on Medscape.com.
FDA approves new myasthenia gravis drug
“There are significant unmet medical needs for people living with myasthenia gravis, as with many other rare diseases,” Billy Dunn, MD, director, office of neuroscience, FDA Center for Drug Evaluation and Research, said in a news release.
This approval represents “an important step in providing a novel therapy option for patients and underscores the agency’s commitment to help make new treatment options available for people living with rare diseases,” Dr. Dunn added.
Effective, well tolerated
The rare and chronic autoimmune neuromuscular disorder of gMG causes debilitating and potentially life-threatening muscle weakness and significantly impaired independence and quality of life. Most patients with gMG have IgG antibodies, which are most often directed against skeletal muscle nicotinic acetylcholine receptors.
Efgartigimod is an antibody fragment designed to reduce pathogenic IgG antibodies and block the IgG recycling process in patients with gMG.
The novel agent binds to the neonatal Fc receptor (FcRn), which is widely expressed throughout the body and plays a central role in rescuing IgG antibodies from degradation. Blocking FcRn reduces IgG antibody levels.
As previously reported, efgartigimod was effective and well tolerated in the phase 3, randomized, placebo-controlled ADAPT trial, which enrolled 187 adults with gMG regardless of acetylcholine receptor antibody status. All had a Myasthenia Gravis–Activities of Daily Living score of at least 5 (>50% nonocular) on a background of a stable dose of at least one MG drug.
For 26 weeks, 84 patients were randomly assigned to receive efgartigimod 10 mg/kg and 83 to receive matching placebo. Both treatments were administered as four infusions per cycle at one infusion per week. The process was repeated as needed, depending on clinical response no sooner than 8 weeks after initiation of the previous cycle.
Treatment with efgartigimod reduced disease burden and improved strength and quality of life in patients with gMG across four MG-specific scales. In addition, these benefits were observed early and were reproducible and durable.
The results were published in Lancet Neurology.
‘Important new advance’
Efgartigimod is a “very rapidly acting drug relative to other treatments that may take 4, 6, sometimes 10 months before they start to work; and the side-effect profile is much like placebo,” said principal investigator James Howard Jr., MD, department of neurology, University of North Carolina at Chapel Hill.
The FDA granted efgartigimod fast track and orphan drug designation.
“People living with gMG have been in need of new treatment options that are targeted to the underlying pathogenesis of the disease and supported by clinical data,” Dr. Howard said in a company news release issued upon approval.
This approval “represents an important new advance for gMG patients and families affected by this debilitating disease. This therapy has the potential to reduce the disease burden of gMG and transform the way we treat this disease,” Dr. Howard added.
A version of this article first appeared on Medscape.com.
“There are significant unmet medical needs for people living with myasthenia gravis, as with many other rare diseases,” Billy Dunn, MD, director, office of neuroscience, FDA Center for Drug Evaluation and Research, said in a news release.
This approval represents “an important step in providing a novel therapy option for patients and underscores the agency’s commitment to help make new treatment options available for people living with rare diseases,” Dr. Dunn added.
Effective, well tolerated
The rare and chronic autoimmune neuromuscular disorder of gMG causes debilitating and potentially life-threatening muscle weakness and significantly impaired independence and quality of life. Most patients with gMG have IgG antibodies, which are most often directed against skeletal muscle nicotinic acetylcholine receptors.
Efgartigimod is an antibody fragment designed to reduce pathogenic IgG antibodies and block the IgG recycling process in patients with gMG.
The novel agent binds to the neonatal Fc receptor (FcRn), which is widely expressed throughout the body and plays a central role in rescuing IgG antibodies from degradation. Blocking FcRn reduces IgG antibody levels.
As previously reported, efgartigimod was effective and well tolerated in the phase 3, randomized, placebo-controlled ADAPT trial, which enrolled 187 adults with gMG regardless of acetylcholine receptor antibody status. All had a Myasthenia Gravis–Activities of Daily Living score of at least 5 (>50% nonocular) on a background of a stable dose of at least one MG drug.
For 26 weeks, 84 patients were randomly assigned to receive efgartigimod 10 mg/kg and 83 to receive matching placebo. Both treatments were administered as four infusions per cycle at one infusion per week. The process was repeated as needed, depending on clinical response no sooner than 8 weeks after initiation of the previous cycle.
Treatment with efgartigimod reduced disease burden and improved strength and quality of life in patients with gMG across four MG-specific scales. In addition, these benefits were observed early and were reproducible and durable.
The results were published in Lancet Neurology.
‘Important new advance’
Efgartigimod is a “very rapidly acting drug relative to other treatments that may take 4, 6, sometimes 10 months before they start to work; and the side-effect profile is much like placebo,” said principal investigator James Howard Jr., MD, department of neurology, University of North Carolina at Chapel Hill.
The FDA granted efgartigimod fast track and orphan drug designation.
“People living with gMG have been in need of new treatment options that are targeted to the underlying pathogenesis of the disease and supported by clinical data,” Dr. Howard said in a company news release issued upon approval.
This approval “represents an important new advance for gMG patients and families affected by this debilitating disease. This therapy has the potential to reduce the disease burden of gMG and transform the way we treat this disease,” Dr. Howard added.
A version of this article first appeared on Medscape.com.
“There are significant unmet medical needs for people living with myasthenia gravis, as with many other rare diseases,” Billy Dunn, MD, director, office of neuroscience, FDA Center for Drug Evaluation and Research, said in a news release.
This approval represents “an important step in providing a novel therapy option for patients and underscores the agency’s commitment to help make new treatment options available for people living with rare diseases,” Dr. Dunn added.
Effective, well tolerated
The rare and chronic autoimmune neuromuscular disorder of gMG causes debilitating and potentially life-threatening muscle weakness and significantly impaired independence and quality of life. Most patients with gMG have IgG antibodies, which are most often directed against skeletal muscle nicotinic acetylcholine receptors.
Efgartigimod is an antibody fragment designed to reduce pathogenic IgG antibodies and block the IgG recycling process in patients with gMG.
The novel agent binds to the neonatal Fc receptor (FcRn), which is widely expressed throughout the body and plays a central role in rescuing IgG antibodies from degradation. Blocking FcRn reduces IgG antibody levels.
As previously reported, efgartigimod was effective and well tolerated in the phase 3, randomized, placebo-controlled ADAPT trial, which enrolled 187 adults with gMG regardless of acetylcholine receptor antibody status. All had a Myasthenia Gravis–Activities of Daily Living score of at least 5 (>50% nonocular) on a background of a stable dose of at least one MG drug.
For 26 weeks, 84 patients were randomly assigned to receive efgartigimod 10 mg/kg and 83 to receive matching placebo. Both treatments were administered as four infusions per cycle at one infusion per week. The process was repeated as needed, depending on clinical response no sooner than 8 weeks after initiation of the previous cycle.
Treatment with efgartigimod reduced disease burden and improved strength and quality of life in patients with gMG across four MG-specific scales. In addition, these benefits were observed early and were reproducible and durable.
The results were published in Lancet Neurology.
‘Important new advance’
Efgartigimod is a “very rapidly acting drug relative to other treatments that may take 4, 6, sometimes 10 months before they start to work; and the side-effect profile is much like placebo,” said principal investigator James Howard Jr., MD, department of neurology, University of North Carolina at Chapel Hill.
The FDA granted efgartigimod fast track and orphan drug designation.
“People living with gMG have been in need of new treatment options that are targeted to the underlying pathogenesis of the disease and supported by clinical data,” Dr. Howard said in a company news release issued upon approval.
This approval “represents an important new advance for gMG patients and families affected by this debilitating disease. This therapy has the potential to reduce the disease burden of gMG and transform the way we treat this disease,” Dr. Howard added.
A version of this article first appeared on Medscape.com.
Genetic tests prompt therapy adjustments in children with epilepsy
Physicians at a Boston hospital adjusted medical management for nearly three-quarters of patients with infantile- or childhood-onset epilepsy who were diagnosed with genetic epilepsy, researchers reported at the annual meeting of the American Epilepsy Society. The findings provide new insight into the usefulness of genetic tests in children with epilepsy of unknown cause.
“. Genetic testing should be included as part of the standard evaluation of individuals with unexplained pediatric epilepsy as a means of achieving diagnostic precision and informing clinical management,” study lead author Isabel Haviland, MD, a neurologist with Boston Children’s Hospital/Harvard Medical School, said in an interview.
According to Dr. Haviland, the causes of epilepsy are unexplained in an estimated two-thirds of pediatric epilepsy cases. “Increasingly, when genetic testing is available, previously unexplained cases of pediatric epilepsy are being found to have single-gene etiologies,” she said. “Though a genetic diagnosis in this population has implications for medical care, the direct impact on medical management in a clinical setting has not been measured. We aimed to describe the impact of genetic diagnosis on medical management in a cohort of individuals with pediatric epilepsy.”
Researchers tracked 602 patients at Boston Children’s Hospital who received next-generation gene sequencing testing from 2012 to 2019. Of those, Dr. Haviland said, 152 (25%) had a positive result that indicated genetic epilepsy (46% female, median age of onset = 6 months [2-15 months]). These patients were included in the study.
“We documented an impact on medical management in nearly three-fourths of participants (72%),” Dr. Haviland said. “A genetic diagnosis affected at least one of four categories of medical management, including care coordination (48%), treatment (45%), counseling about a change in prognosis (28%), and change in diagnosis for a few individuals who had a prior established diagnosis (1%).”
As examples, she mentioned three cases:
- Testing revealed that a subject has a disease-causing genetic variant in a gene called PRRT2. “This gene is involved in the release of neurotransmitters in the brain,” Dr. Haviland said. “Thanks to his diagnosis, he was treated with the antiseizure medication oxcarbazepine, which is often effective for epilepsy caused by variants in this gene. He had excellent response to the medication and later became seizure free.”
- A subject had a variation in the SCN1A gene that causes types of epilepsy. “At the time of his diagnosis, there was a trial for a medication called fenfluramine being offered for individuals with SCN1A variants, and his family elected to participate,” she said. “This medication was later approved by the [Food and Drug Administration] for SCN1A-related epilepsy.”
- Testing identified disease-causing variant in the GRIN2A gene in another subject. “This gene is involved in brain cell communication,” Dr. Haviland said. “This individual was treated with memantine, which acts on the specific biological pathway affected by the gene. This treatment would not have been considered without the genetic diagnosis as it is currently only approved for Alzheimer’s disease.”
In addition, Dr. Haviland said, researchers found that “there was impact on medical management both in those with earlier age of epilepsy onset (under 2 years) and those with later age of onset, as well as both in those with developmental disorders (such as autism spectrum disorder, intellectual disability and developmental delay) and those with normal development.
As for the cost of genetic tests, Dr. Haviland pointed to a 2019 study that she said estimated epilepsy panel testing runs from $1,500 to $7,500, and the whole exome sequencing from $4,500 to $7,000. “Insurers sometimes cover testing, but not always,” she said. “In some cases, insurance will only cover testing if it is documented that results will directly alter medical management, which highlights the importance of our findings.”
No study funding was reported. Dr. Haviland and several other authors report no disclosures. One author reports consulting fees from Takeda, Zogenix, Marinus, and FOXG1 Research Foundation. Another author reports research support from the International Foundation for CDKL5 Research.
Physicians at a Boston hospital adjusted medical management for nearly three-quarters of patients with infantile- or childhood-onset epilepsy who were diagnosed with genetic epilepsy, researchers reported at the annual meeting of the American Epilepsy Society. The findings provide new insight into the usefulness of genetic tests in children with epilepsy of unknown cause.
“. Genetic testing should be included as part of the standard evaluation of individuals with unexplained pediatric epilepsy as a means of achieving diagnostic precision and informing clinical management,” study lead author Isabel Haviland, MD, a neurologist with Boston Children’s Hospital/Harvard Medical School, said in an interview.
According to Dr. Haviland, the causes of epilepsy are unexplained in an estimated two-thirds of pediatric epilepsy cases. “Increasingly, when genetic testing is available, previously unexplained cases of pediatric epilepsy are being found to have single-gene etiologies,” she said. “Though a genetic diagnosis in this population has implications for medical care, the direct impact on medical management in a clinical setting has not been measured. We aimed to describe the impact of genetic diagnosis on medical management in a cohort of individuals with pediatric epilepsy.”
Researchers tracked 602 patients at Boston Children’s Hospital who received next-generation gene sequencing testing from 2012 to 2019. Of those, Dr. Haviland said, 152 (25%) had a positive result that indicated genetic epilepsy (46% female, median age of onset = 6 months [2-15 months]). These patients were included in the study.
“We documented an impact on medical management in nearly three-fourths of participants (72%),” Dr. Haviland said. “A genetic diagnosis affected at least one of four categories of medical management, including care coordination (48%), treatment (45%), counseling about a change in prognosis (28%), and change in diagnosis for a few individuals who had a prior established diagnosis (1%).”
As examples, she mentioned three cases:
- Testing revealed that a subject has a disease-causing genetic variant in a gene called PRRT2. “This gene is involved in the release of neurotransmitters in the brain,” Dr. Haviland said. “Thanks to his diagnosis, he was treated with the antiseizure medication oxcarbazepine, which is often effective for epilepsy caused by variants in this gene. He had excellent response to the medication and later became seizure free.”
- A subject had a variation in the SCN1A gene that causes types of epilepsy. “At the time of his diagnosis, there was a trial for a medication called fenfluramine being offered for individuals with SCN1A variants, and his family elected to participate,” she said. “This medication was later approved by the [Food and Drug Administration] for SCN1A-related epilepsy.”
- Testing identified disease-causing variant in the GRIN2A gene in another subject. “This gene is involved in brain cell communication,” Dr. Haviland said. “This individual was treated with memantine, which acts on the specific biological pathway affected by the gene. This treatment would not have been considered without the genetic diagnosis as it is currently only approved for Alzheimer’s disease.”
In addition, Dr. Haviland said, researchers found that “there was impact on medical management both in those with earlier age of epilepsy onset (under 2 years) and those with later age of onset, as well as both in those with developmental disorders (such as autism spectrum disorder, intellectual disability and developmental delay) and those with normal development.
As for the cost of genetic tests, Dr. Haviland pointed to a 2019 study that she said estimated epilepsy panel testing runs from $1,500 to $7,500, and the whole exome sequencing from $4,500 to $7,000. “Insurers sometimes cover testing, but not always,” she said. “In some cases, insurance will only cover testing if it is documented that results will directly alter medical management, which highlights the importance of our findings.”
No study funding was reported. Dr. Haviland and several other authors report no disclosures. One author reports consulting fees from Takeda, Zogenix, Marinus, and FOXG1 Research Foundation. Another author reports research support from the International Foundation for CDKL5 Research.
Physicians at a Boston hospital adjusted medical management for nearly three-quarters of patients with infantile- or childhood-onset epilepsy who were diagnosed with genetic epilepsy, researchers reported at the annual meeting of the American Epilepsy Society. The findings provide new insight into the usefulness of genetic tests in children with epilepsy of unknown cause.
“. Genetic testing should be included as part of the standard evaluation of individuals with unexplained pediatric epilepsy as a means of achieving diagnostic precision and informing clinical management,” study lead author Isabel Haviland, MD, a neurologist with Boston Children’s Hospital/Harvard Medical School, said in an interview.
According to Dr. Haviland, the causes of epilepsy are unexplained in an estimated two-thirds of pediatric epilepsy cases. “Increasingly, when genetic testing is available, previously unexplained cases of pediatric epilepsy are being found to have single-gene etiologies,” she said. “Though a genetic diagnosis in this population has implications for medical care, the direct impact on medical management in a clinical setting has not been measured. We aimed to describe the impact of genetic diagnosis on medical management in a cohort of individuals with pediatric epilepsy.”
Researchers tracked 602 patients at Boston Children’s Hospital who received next-generation gene sequencing testing from 2012 to 2019. Of those, Dr. Haviland said, 152 (25%) had a positive result that indicated genetic epilepsy (46% female, median age of onset = 6 months [2-15 months]). These patients were included in the study.
“We documented an impact on medical management in nearly three-fourths of participants (72%),” Dr. Haviland said. “A genetic diagnosis affected at least one of four categories of medical management, including care coordination (48%), treatment (45%), counseling about a change in prognosis (28%), and change in diagnosis for a few individuals who had a prior established diagnosis (1%).”
As examples, she mentioned three cases:
- Testing revealed that a subject has a disease-causing genetic variant in a gene called PRRT2. “This gene is involved in the release of neurotransmitters in the brain,” Dr. Haviland said. “Thanks to his diagnosis, he was treated with the antiseizure medication oxcarbazepine, which is often effective for epilepsy caused by variants in this gene. He had excellent response to the medication and later became seizure free.”
- A subject had a variation in the SCN1A gene that causes types of epilepsy. “At the time of his diagnosis, there was a trial for a medication called fenfluramine being offered for individuals with SCN1A variants, and his family elected to participate,” she said. “This medication was later approved by the [Food and Drug Administration] for SCN1A-related epilepsy.”
- Testing identified disease-causing variant in the GRIN2A gene in another subject. “This gene is involved in brain cell communication,” Dr. Haviland said. “This individual was treated with memantine, which acts on the specific biological pathway affected by the gene. This treatment would not have been considered without the genetic diagnosis as it is currently only approved for Alzheimer’s disease.”
In addition, Dr. Haviland said, researchers found that “there was impact on medical management both in those with earlier age of epilepsy onset (under 2 years) and those with later age of onset, as well as both in those with developmental disorders (such as autism spectrum disorder, intellectual disability and developmental delay) and those with normal development.
As for the cost of genetic tests, Dr. Haviland pointed to a 2019 study that she said estimated epilepsy panel testing runs from $1,500 to $7,500, and the whole exome sequencing from $4,500 to $7,000. “Insurers sometimes cover testing, but not always,” she said. “In some cases, insurance will only cover testing if it is documented that results will directly alter medical management, which highlights the importance of our findings.”
No study funding was reported. Dr. Haviland and several other authors report no disclosures. One author reports consulting fees from Takeda, Zogenix, Marinus, and FOXG1 Research Foundation. Another author reports research support from the International Foundation for CDKL5 Research.
FROM AES 2021
Ginger for migraine: A new review
in patients who do not want to use or don’t have access to prescription medications, new data suggest.
Conducted by investigators at the National Institute of Mental Health and Neurosciences, Bangalore, India, the review showed ginger root can relieve migraine-related pain, nausea, and vomiting. However, the evidence does not support ginger’s use as a first-line therapy for acute migraine or for migraine prevention.
Study author Chittaranjan Andrade, MD, professor of clinical psychopharmacology and neurotoxicology at the institute, said in an interview that the evidence base is still “too small” to support formal clinical recommendations. However, he added, ginger can be considered as a viable “home-remedy option” for acute migraine.
The review was published online Dec. 2 in The Journal of Clinical Psychiatry.
Potential uses
Used for centuries in traditional medicine, much of the preclinical and clinical research has examined the potential of raw ginger, ginger extracts, and ginger constituents to prevent and treat a wide range of medical conditions. These include nausea and vomiting associated with pregnancy, chemotherapy, postoperative states, motion sickness, and other diseases and disorders, said Dr. Andrade.
Ginger has “long been recommended as an effective home remedy for the acute treatment of migraine, relieving both headache and the associated nausea,” Dr. Andrade noted.
One recommended recipe is stirring half a teaspoon of ground ginger into a glass of water and drinking the “ginger juice,” while another is to drink hot tea made from a teaspoon of freshly ground ginger.
“Patients with a number of common ailments, including migraine, are sometimes caught without medicines; or they may have poor access to medicines,” Dr. Andrade said. “I came across a reference to the use of ginger for migraine in a book on home remedies and I thought that if the research literature supports the use of ginger for migraine episodes, such patients could benefit.”
Large treatment gap
The review and meta-analysis included three randomized controlled trials with 227 patients looking at ginger versus placebo for the treatment.
One of the studies investigated the therapeutic efficacy of a specific proprietary formulation of ginger, combined with feverfew, while two trials were independent of industry.
Of these two, one examined the benefit of add-on dry ginger extract (400 mg; 5% active gingerols) in 50 patients who were also taking ketoprofen to treat migraine episodes, while the other examined the 3-month efficacy of daily dry ginger extract for migraine prophylaxis in 107 patients.
The two studies that examined the therapeutic efficacy of ginger versus placebo showed ginger reduced mean pain scores at 2 hours (mean difference, –1.27 [95% confidence interval, –1.46 to 1,07]) and also increased the proportion of patients who were pain free at 2 hours (RR, 1.79 [1.04 to 3.09]). In addition, compared to placebo, ginger halved the risk of migraine-related nausea and vomiting in all of the studies and was not associated with an increased risk of adverse events.
One RCT investigated prophylactic efficacy and found it to be more effective than placebo in bringing a ≥ 50% reduction in the frequency of monthly migraine episodes (in 42% versus 39% of patients, respectively), but the difference was not deemed statistically significant. In addition, there were no significant differences between the groups in days of pain, severe pain, days requiring use of analgesics, number of migraine episodes, and maximum duration of migraine episodes.
Dr. Andrade noted that ginger has many chemical constituents, including phenolic compounds, terpenes, polysaccharides, lipids, and organic acids of which 6-shogaol, 6-gingerol, and 10-dehydrogingerdione “may be important.”
It also has antioxidant and anti-inflammatory effects, lowering prostaglandins, and reducing several serum lipid and glycemic measures. Additionally, it has “putative” vasculoprotective effects, he added.
“Ginger has a large number of chemical constituents and we do not know which of these, separately or in combination, will help relieve migraine,” he said. “We won’t know the answer unless clinical trials are conducted with the individual constituents rather than with ginger extract.” He compared this to the study of omega-3 fatty acids rather than fish and nuts for various neuropsychiatric or cardiovascular indications.
Nevertheless, given the high global prevalence of migraine and the “large treatment gap [of migraine] in primary care,” it could be common for many affected patients to experience episodes of migraine headache “without recourse to recommended pharmacologic relief,” he noted. “In such cases, the availability of a simple home remedy, such as ginger, could be helpful.”
‘Good additional tool’
Commenting on the study for this news organization, Jessica Ailani, MD, director, MedStar Georgetown Headache Center and professor of clinical neurology, MedStar Georgetown University Hospital, Washington, said that for “people with migraine who are seeking treatment with minimal side effects that they can obtain without counsel of a health care provider, ginger is a good additional tool to have.”
Dr. Ailani, vice cochair of strategic planning in the MedStar department of neurology, who was not involved with the study, said that clinicians can “consider suggesting ginger to patients with migraine that have associated nausea who are interested in nonpharmacologic ways to treat symptoms.”
Since there are “many other effective ways to treat migraine,” she advises “conversing with the patient about speed of onset of efficacy, along with tolerability, and return of migraine symptoms as important factors to evaluate when choosing and staying with a treatment.”
Also commenting on the study for this news organization, Nada Hindiyeh, MD, clinical associate professor, department of neurology, Stanford (Calif.) University, called it a “nice summary of the objective research available for the use of ginger in acute and preventive treatment of migraine.”
Although there is insufficient literature evaluating ginger alone in migraine treatment, so “no definitive conclusions can be drawn,” since it appears to be safe and “somewhat helpful for migraine-associated nausea and vomiting and possibly in frequency of migraine reduction, it remains a considerable alternative for those seeking nonprescription options,” said Dr. Hindiyeh, who was not involved with the study.
Dr. Andrade publishes an e-newsletter supported by Sun Pharmaceuticals, with payments made to charities. He has received payments for developing educational materials for scientific initiatives and programs. Dr. Ailani reports honoraria for independent consulting from various pharmaceutical companies and clinical trial grants to her institution from the American Migraine Foundation, Allergan, Biohaven, Eli Lilly, Satsuma, and Zosano. Dr. Hindiyeh discloses no relevant financial relationships.
A version of this article first appeared on Medscape.com.
in patients who do not want to use or don’t have access to prescription medications, new data suggest.
Conducted by investigators at the National Institute of Mental Health and Neurosciences, Bangalore, India, the review showed ginger root can relieve migraine-related pain, nausea, and vomiting. However, the evidence does not support ginger’s use as a first-line therapy for acute migraine or for migraine prevention.
Study author Chittaranjan Andrade, MD, professor of clinical psychopharmacology and neurotoxicology at the institute, said in an interview that the evidence base is still “too small” to support formal clinical recommendations. However, he added, ginger can be considered as a viable “home-remedy option” for acute migraine.
The review was published online Dec. 2 in The Journal of Clinical Psychiatry.
Potential uses
Used for centuries in traditional medicine, much of the preclinical and clinical research has examined the potential of raw ginger, ginger extracts, and ginger constituents to prevent and treat a wide range of medical conditions. These include nausea and vomiting associated with pregnancy, chemotherapy, postoperative states, motion sickness, and other diseases and disorders, said Dr. Andrade.
Ginger has “long been recommended as an effective home remedy for the acute treatment of migraine, relieving both headache and the associated nausea,” Dr. Andrade noted.
One recommended recipe is stirring half a teaspoon of ground ginger into a glass of water and drinking the “ginger juice,” while another is to drink hot tea made from a teaspoon of freshly ground ginger.
“Patients with a number of common ailments, including migraine, are sometimes caught without medicines; or they may have poor access to medicines,” Dr. Andrade said. “I came across a reference to the use of ginger for migraine in a book on home remedies and I thought that if the research literature supports the use of ginger for migraine episodes, such patients could benefit.”
Large treatment gap
The review and meta-analysis included three randomized controlled trials with 227 patients looking at ginger versus placebo for the treatment.
One of the studies investigated the therapeutic efficacy of a specific proprietary formulation of ginger, combined with feverfew, while two trials were independent of industry.
Of these two, one examined the benefit of add-on dry ginger extract (400 mg; 5% active gingerols) in 50 patients who were also taking ketoprofen to treat migraine episodes, while the other examined the 3-month efficacy of daily dry ginger extract for migraine prophylaxis in 107 patients.
The two studies that examined the therapeutic efficacy of ginger versus placebo showed ginger reduced mean pain scores at 2 hours (mean difference, –1.27 [95% confidence interval, –1.46 to 1,07]) and also increased the proportion of patients who were pain free at 2 hours (RR, 1.79 [1.04 to 3.09]). In addition, compared to placebo, ginger halved the risk of migraine-related nausea and vomiting in all of the studies and was not associated with an increased risk of adverse events.
One RCT investigated prophylactic efficacy and found it to be more effective than placebo in bringing a ≥ 50% reduction in the frequency of monthly migraine episodes (in 42% versus 39% of patients, respectively), but the difference was not deemed statistically significant. In addition, there were no significant differences between the groups in days of pain, severe pain, days requiring use of analgesics, number of migraine episodes, and maximum duration of migraine episodes.
Dr. Andrade noted that ginger has many chemical constituents, including phenolic compounds, terpenes, polysaccharides, lipids, and organic acids of which 6-shogaol, 6-gingerol, and 10-dehydrogingerdione “may be important.”
It also has antioxidant and anti-inflammatory effects, lowering prostaglandins, and reducing several serum lipid and glycemic measures. Additionally, it has “putative” vasculoprotective effects, he added.
“Ginger has a large number of chemical constituents and we do not know which of these, separately or in combination, will help relieve migraine,” he said. “We won’t know the answer unless clinical trials are conducted with the individual constituents rather than with ginger extract.” He compared this to the study of omega-3 fatty acids rather than fish and nuts for various neuropsychiatric or cardiovascular indications.
Nevertheless, given the high global prevalence of migraine and the “large treatment gap [of migraine] in primary care,” it could be common for many affected patients to experience episodes of migraine headache “without recourse to recommended pharmacologic relief,” he noted. “In such cases, the availability of a simple home remedy, such as ginger, could be helpful.”
‘Good additional tool’
Commenting on the study for this news organization, Jessica Ailani, MD, director, MedStar Georgetown Headache Center and professor of clinical neurology, MedStar Georgetown University Hospital, Washington, said that for “people with migraine who are seeking treatment with minimal side effects that they can obtain without counsel of a health care provider, ginger is a good additional tool to have.”
Dr. Ailani, vice cochair of strategic planning in the MedStar department of neurology, who was not involved with the study, said that clinicians can “consider suggesting ginger to patients with migraine that have associated nausea who are interested in nonpharmacologic ways to treat symptoms.”
Since there are “many other effective ways to treat migraine,” she advises “conversing with the patient about speed of onset of efficacy, along with tolerability, and return of migraine symptoms as important factors to evaluate when choosing and staying with a treatment.”
Also commenting on the study for this news organization, Nada Hindiyeh, MD, clinical associate professor, department of neurology, Stanford (Calif.) University, called it a “nice summary of the objective research available for the use of ginger in acute and preventive treatment of migraine.”
Although there is insufficient literature evaluating ginger alone in migraine treatment, so “no definitive conclusions can be drawn,” since it appears to be safe and “somewhat helpful for migraine-associated nausea and vomiting and possibly in frequency of migraine reduction, it remains a considerable alternative for those seeking nonprescription options,” said Dr. Hindiyeh, who was not involved with the study.
Dr. Andrade publishes an e-newsletter supported by Sun Pharmaceuticals, with payments made to charities. He has received payments for developing educational materials for scientific initiatives and programs. Dr. Ailani reports honoraria for independent consulting from various pharmaceutical companies and clinical trial grants to her institution from the American Migraine Foundation, Allergan, Biohaven, Eli Lilly, Satsuma, and Zosano. Dr. Hindiyeh discloses no relevant financial relationships.
A version of this article first appeared on Medscape.com.
in patients who do not want to use or don’t have access to prescription medications, new data suggest.
Conducted by investigators at the National Institute of Mental Health and Neurosciences, Bangalore, India, the review showed ginger root can relieve migraine-related pain, nausea, and vomiting. However, the evidence does not support ginger’s use as a first-line therapy for acute migraine or for migraine prevention.
Study author Chittaranjan Andrade, MD, professor of clinical psychopharmacology and neurotoxicology at the institute, said in an interview that the evidence base is still “too small” to support formal clinical recommendations. However, he added, ginger can be considered as a viable “home-remedy option” for acute migraine.
The review was published online Dec. 2 in The Journal of Clinical Psychiatry.
Potential uses
Used for centuries in traditional medicine, much of the preclinical and clinical research has examined the potential of raw ginger, ginger extracts, and ginger constituents to prevent and treat a wide range of medical conditions. These include nausea and vomiting associated with pregnancy, chemotherapy, postoperative states, motion sickness, and other diseases and disorders, said Dr. Andrade.
Ginger has “long been recommended as an effective home remedy for the acute treatment of migraine, relieving both headache and the associated nausea,” Dr. Andrade noted.
One recommended recipe is stirring half a teaspoon of ground ginger into a glass of water and drinking the “ginger juice,” while another is to drink hot tea made from a teaspoon of freshly ground ginger.
“Patients with a number of common ailments, including migraine, are sometimes caught without medicines; or they may have poor access to medicines,” Dr. Andrade said. “I came across a reference to the use of ginger for migraine in a book on home remedies and I thought that if the research literature supports the use of ginger for migraine episodes, such patients could benefit.”
Large treatment gap
The review and meta-analysis included three randomized controlled trials with 227 patients looking at ginger versus placebo for the treatment.
One of the studies investigated the therapeutic efficacy of a specific proprietary formulation of ginger, combined with feverfew, while two trials were independent of industry.
Of these two, one examined the benefit of add-on dry ginger extract (400 mg; 5% active gingerols) in 50 patients who were also taking ketoprofen to treat migraine episodes, while the other examined the 3-month efficacy of daily dry ginger extract for migraine prophylaxis in 107 patients.
The two studies that examined the therapeutic efficacy of ginger versus placebo showed ginger reduced mean pain scores at 2 hours (mean difference, –1.27 [95% confidence interval, –1.46 to 1,07]) and also increased the proportion of patients who were pain free at 2 hours (RR, 1.79 [1.04 to 3.09]). In addition, compared to placebo, ginger halved the risk of migraine-related nausea and vomiting in all of the studies and was not associated with an increased risk of adverse events.
One RCT investigated prophylactic efficacy and found it to be more effective than placebo in bringing a ≥ 50% reduction in the frequency of monthly migraine episodes (in 42% versus 39% of patients, respectively), but the difference was not deemed statistically significant. In addition, there were no significant differences between the groups in days of pain, severe pain, days requiring use of analgesics, number of migraine episodes, and maximum duration of migraine episodes.
Dr. Andrade noted that ginger has many chemical constituents, including phenolic compounds, terpenes, polysaccharides, lipids, and organic acids of which 6-shogaol, 6-gingerol, and 10-dehydrogingerdione “may be important.”
It also has antioxidant and anti-inflammatory effects, lowering prostaglandins, and reducing several serum lipid and glycemic measures. Additionally, it has “putative” vasculoprotective effects, he added.
“Ginger has a large number of chemical constituents and we do not know which of these, separately or in combination, will help relieve migraine,” he said. “We won’t know the answer unless clinical trials are conducted with the individual constituents rather than with ginger extract.” He compared this to the study of omega-3 fatty acids rather than fish and nuts for various neuropsychiatric or cardiovascular indications.
Nevertheless, given the high global prevalence of migraine and the “large treatment gap [of migraine] in primary care,” it could be common for many affected patients to experience episodes of migraine headache “without recourse to recommended pharmacologic relief,” he noted. “In such cases, the availability of a simple home remedy, such as ginger, could be helpful.”
‘Good additional tool’
Commenting on the study for this news organization, Jessica Ailani, MD, director, MedStar Georgetown Headache Center and professor of clinical neurology, MedStar Georgetown University Hospital, Washington, said that for “people with migraine who are seeking treatment with minimal side effects that they can obtain without counsel of a health care provider, ginger is a good additional tool to have.”
Dr. Ailani, vice cochair of strategic planning in the MedStar department of neurology, who was not involved with the study, said that clinicians can “consider suggesting ginger to patients with migraine that have associated nausea who are interested in nonpharmacologic ways to treat symptoms.”
Since there are “many other effective ways to treat migraine,” she advises “conversing with the patient about speed of onset of efficacy, along with tolerability, and return of migraine symptoms as important factors to evaluate when choosing and staying with a treatment.”
Also commenting on the study for this news organization, Nada Hindiyeh, MD, clinical associate professor, department of neurology, Stanford (Calif.) University, called it a “nice summary of the objective research available for the use of ginger in acute and preventive treatment of migraine.”
Although there is insufficient literature evaluating ginger alone in migraine treatment, so “no definitive conclusions can be drawn,” since it appears to be safe and “somewhat helpful for migraine-associated nausea and vomiting and possibly in frequency of migraine reduction, it remains a considerable alternative for those seeking nonprescription options,” said Dr. Hindiyeh, who was not involved with the study.
Dr. Andrade publishes an e-newsletter supported by Sun Pharmaceuticals, with payments made to charities. He has received payments for developing educational materials for scientific initiatives and programs. Dr. Ailani reports honoraria for independent consulting from various pharmaceutical companies and clinical trial grants to her institution from the American Migraine Foundation, Allergan, Biohaven, Eli Lilly, Satsuma, and Zosano. Dr. Hindiyeh discloses no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF CLINICAL PSYCHIATRY
Diabetes tied to Parkinson’s risk, more rapid disease progression
Diabetes mellitus (DM) is associated with Parkinson’s disease (PD) development, as well as more severe symptoms and more rapid disease progression, new research suggests.
In a systematic review, patients with type 2 diabetes were 34% more likely to develop PD than those without comorbid DM. In addition, patients with both conditions had significantly worse scores on the Unified Parkinson’s Disease Rating Scale (UPDRS) and worse cognitive performance.
Together, the results suggest that “DM may be a facilitating factor of neurodegeneration,” wrote the investigators, led by Gennaro Pagano, MD, PhD, expert medical director at Roche Pharma Research and Early Development, in Basel, Switzerland.
The findings were published in a recent issue of the Journal of Parkinson’s Disease.
Unanswered questions
Researchers have long proposed a potential relationship between diabetes and PD. However, case-control studies have yielded conflicting results about this relationship – and previous systematic reviews have failed to clarify the question.
In the current systematic review and meta-analysis, investigators identified relevant studies in databases such as MEDLINE/PubMed, Cochrane CENTRAL, and Scopus.
Eligible studies reported prevalence of DM in patients with PD, reported incidence of PD in those with and those without DM, and analyzed Parkinson’s phenotype and progression in those with and those without DM.
The researchers identified 3,829 articles in their initial search, evaluated 90 articles in detail, and included 43 studies in their analysis. Study quality was judged to be moderate or good, and the investigators did not find significant publication bias.
Twenty-one studies that encompassed 11,396 patients were examined to determine prevalence of DM in PD. This prevalence was calculated to be 10.02%, which is similar to the global prevalence of 9.3% reported in 2019.
The researchers also analyzed 12 cohort studies that included 17,797,221 patients to calculate risk for PD in patients with comorbid diabetes. The pooled summary odds ratio for incident PD among patients with type 2 diabetes was 1.34.
The evaluation of the effect of diabetes on PD severity was based on 10 studies that included 603 patients with both diseases. Because data on motor symptoms were not available for all studies, the researchers considered Hoehn and Yahr stage, UPDRS score, and cognitive impairment.
Patients with both conditions had a worse Hoehn and Yahr stage (standardized mean difference, 0.36; P < .001), and higher UPDRS score (SMD, 0.60; P < .001). In 7 of the 10 studies, diabetes was associated with worse cognitive performance in patients with PD.
Mechanisms uncertain
The mechanisms of the effect of diabetes on risk for and severity of PD are uncertain, but the researchers have developed hypotheses.
“Overlapping mechanisms between insulin resistance, mitochondrial dysfunction, oxidative stress, and alpha-synuclein expression could influence the development of the neurodegeneration process,” they wrote.
Because the current analysis demonstrated a trend toward more pronounced cognitive decline in patients with the comorbidities, clinicians should pay particular attention to the progression of motor and cognitive symptoms in patients with these diseases, the investigators noted.
“Additional studies are needed in order to better define the clinical phenotype of PD-DM patients and explore the role of antidiabetic drugs on PD progression,” they wrote.
They add that future studies also are needed to evaluate whether antidiabetic drugs might reduce risk for PD in these patients.
The investigators noted several limitations of their research. In many of the studies they examined, for example, diagnostic criteria of type 2 diabetes and PD were based only on medical records or self-reported health questionnaires. The diagnoses were rarely confirmed.
In addition, not all studies clearly stated that their populations presented with type 2 diabetes. Finally, patients with diabetes may be at increased risk for cardiovascular death, which could affect follow-up related to the development of PD, the investigators noted.
A version of this article first appeared on Medscape.com.
Diabetes mellitus (DM) is associated with Parkinson’s disease (PD) development, as well as more severe symptoms and more rapid disease progression, new research suggests.
In a systematic review, patients with type 2 diabetes were 34% more likely to develop PD than those without comorbid DM. In addition, patients with both conditions had significantly worse scores on the Unified Parkinson’s Disease Rating Scale (UPDRS) and worse cognitive performance.
Together, the results suggest that “DM may be a facilitating factor of neurodegeneration,” wrote the investigators, led by Gennaro Pagano, MD, PhD, expert medical director at Roche Pharma Research and Early Development, in Basel, Switzerland.
The findings were published in a recent issue of the Journal of Parkinson’s Disease.
Unanswered questions
Researchers have long proposed a potential relationship between diabetes and PD. However, case-control studies have yielded conflicting results about this relationship – and previous systematic reviews have failed to clarify the question.
In the current systematic review and meta-analysis, investigators identified relevant studies in databases such as MEDLINE/PubMed, Cochrane CENTRAL, and Scopus.
Eligible studies reported prevalence of DM in patients with PD, reported incidence of PD in those with and those without DM, and analyzed Parkinson’s phenotype and progression in those with and those without DM.
The researchers identified 3,829 articles in their initial search, evaluated 90 articles in detail, and included 43 studies in their analysis. Study quality was judged to be moderate or good, and the investigators did not find significant publication bias.
Twenty-one studies that encompassed 11,396 patients were examined to determine prevalence of DM in PD. This prevalence was calculated to be 10.02%, which is similar to the global prevalence of 9.3% reported in 2019.
The researchers also analyzed 12 cohort studies that included 17,797,221 patients to calculate risk for PD in patients with comorbid diabetes. The pooled summary odds ratio for incident PD among patients with type 2 diabetes was 1.34.
The evaluation of the effect of diabetes on PD severity was based on 10 studies that included 603 patients with both diseases. Because data on motor symptoms were not available for all studies, the researchers considered Hoehn and Yahr stage, UPDRS score, and cognitive impairment.
Patients with both conditions had a worse Hoehn and Yahr stage (standardized mean difference, 0.36; P < .001), and higher UPDRS score (SMD, 0.60; P < .001). In 7 of the 10 studies, diabetes was associated with worse cognitive performance in patients with PD.
Mechanisms uncertain
The mechanisms of the effect of diabetes on risk for and severity of PD are uncertain, but the researchers have developed hypotheses.
“Overlapping mechanisms between insulin resistance, mitochondrial dysfunction, oxidative stress, and alpha-synuclein expression could influence the development of the neurodegeneration process,” they wrote.
Because the current analysis demonstrated a trend toward more pronounced cognitive decline in patients with the comorbidities, clinicians should pay particular attention to the progression of motor and cognitive symptoms in patients with these diseases, the investigators noted.
“Additional studies are needed in order to better define the clinical phenotype of PD-DM patients and explore the role of antidiabetic drugs on PD progression,” they wrote.
They add that future studies also are needed to evaluate whether antidiabetic drugs might reduce risk for PD in these patients.
The investigators noted several limitations of their research. In many of the studies they examined, for example, diagnostic criteria of type 2 diabetes and PD were based only on medical records or self-reported health questionnaires. The diagnoses were rarely confirmed.
In addition, not all studies clearly stated that their populations presented with type 2 diabetes. Finally, patients with diabetes may be at increased risk for cardiovascular death, which could affect follow-up related to the development of PD, the investigators noted.
A version of this article first appeared on Medscape.com.
Diabetes mellitus (DM) is associated with Parkinson’s disease (PD) development, as well as more severe symptoms and more rapid disease progression, new research suggests.
In a systematic review, patients with type 2 diabetes were 34% more likely to develop PD than those without comorbid DM. In addition, patients with both conditions had significantly worse scores on the Unified Parkinson’s Disease Rating Scale (UPDRS) and worse cognitive performance.
Together, the results suggest that “DM may be a facilitating factor of neurodegeneration,” wrote the investigators, led by Gennaro Pagano, MD, PhD, expert medical director at Roche Pharma Research and Early Development, in Basel, Switzerland.
The findings were published in a recent issue of the Journal of Parkinson’s Disease.
Unanswered questions
Researchers have long proposed a potential relationship between diabetes and PD. However, case-control studies have yielded conflicting results about this relationship – and previous systematic reviews have failed to clarify the question.
In the current systematic review and meta-analysis, investigators identified relevant studies in databases such as MEDLINE/PubMed, Cochrane CENTRAL, and Scopus.
Eligible studies reported prevalence of DM in patients with PD, reported incidence of PD in those with and those without DM, and analyzed Parkinson’s phenotype and progression in those with and those without DM.
The researchers identified 3,829 articles in their initial search, evaluated 90 articles in detail, and included 43 studies in their analysis. Study quality was judged to be moderate or good, and the investigators did not find significant publication bias.
Twenty-one studies that encompassed 11,396 patients were examined to determine prevalence of DM in PD. This prevalence was calculated to be 10.02%, which is similar to the global prevalence of 9.3% reported in 2019.
The researchers also analyzed 12 cohort studies that included 17,797,221 patients to calculate risk for PD in patients with comorbid diabetes. The pooled summary odds ratio for incident PD among patients with type 2 diabetes was 1.34.
The evaluation of the effect of diabetes on PD severity was based on 10 studies that included 603 patients with both diseases. Because data on motor symptoms were not available for all studies, the researchers considered Hoehn and Yahr stage, UPDRS score, and cognitive impairment.
Patients with both conditions had a worse Hoehn and Yahr stage (standardized mean difference, 0.36; P < .001), and higher UPDRS score (SMD, 0.60; P < .001). In 7 of the 10 studies, diabetes was associated with worse cognitive performance in patients with PD.
Mechanisms uncertain
The mechanisms of the effect of diabetes on risk for and severity of PD are uncertain, but the researchers have developed hypotheses.
“Overlapping mechanisms between insulin resistance, mitochondrial dysfunction, oxidative stress, and alpha-synuclein expression could influence the development of the neurodegeneration process,” they wrote.
Because the current analysis demonstrated a trend toward more pronounced cognitive decline in patients with the comorbidities, clinicians should pay particular attention to the progression of motor and cognitive symptoms in patients with these diseases, the investigators noted.
“Additional studies are needed in order to better define the clinical phenotype of PD-DM patients and explore the role of antidiabetic drugs on PD progression,” they wrote.
They add that future studies also are needed to evaluate whether antidiabetic drugs might reduce risk for PD in these patients.
The investigators noted several limitations of their research. In many of the studies they examined, for example, diagnostic criteria of type 2 diabetes and PD were based only on medical records or self-reported health questionnaires. The diagnoses were rarely confirmed.
In addition, not all studies clearly stated that their populations presented with type 2 diabetes. Finally, patients with diabetes may be at increased risk for cardiovascular death, which could affect follow-up related to the development of PD, the investigators noted.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF PARKINSON’S DISEASE
Are newer migraine therapies better? It depends
The findings, published in JAMA Network Open, “may imply that triptans will remain the current mainstay of specific acute migraine treatment,” suggested senior author Shuu-Jiun Wang, MD, from the National Yang Ming Chiao Tung University, and the Taipei Veterans General Hospital, both in Taipei, Taiwan, and his coauthors. However, lasmiditan (a 5-hydroxytryptamine1F receptor agonist) and rimegepant and ubrogepant (both calcitonin gene-related peptide [CGRP] antagonists) might still have unique advantages, since triptans are contraindicated for patients with cardiovascular risks, they said.
The systemic review and meta-analysis showed that, for the outcome of pain freedom and pain relief at 2 hours after the dose, the three newer agents worked better than placebo, but were inferior to most triptans. However, ubrogepant and rimegepant, which received U.S. Food and Drug Administration approval for the treatment of acute migraine in adults in December 2019 and February 2020, respectively, might be associated with fewer risks of adverse events (AEs), compared with triptans. “These new effective therapeutic options enrich the therapeutic categories of specific acute migraine treatments and may provide an opportunity to decrease the risks of barbiturate or opioid overuse or addiction,” they wrote.
The meta-analysis included 64 randomized, controlled trials involving 46,442 participants (74%-87% female across studies; age range, 36-43 years). All studies examined clinically relevant outcomes in patients with International Headache Society criteria for migraine, and compared currently available migraine-specific acute treatments with each other or placebo. The drugs were examined at doses with widespread clinical use and included: ergotamine, dihydroergotamine, sumatriptan, zolmitriptan, naratriptan, rizatriptan, almotriptan, eletriptan, frovatriptan, lasmiditan, rimegepant, and ubrogepant.
The findings showed that all drug treatments were associated with a higher odds ratio for pain freedom, compared with placebo, except for sumatriptan, 10-mg nasal spray. The most effective drug was eletriptan 40 mg (OR, 5.59), and the least effective was lasmiditan 50 mg (OR, 1.65). Most triptans were associated with higher ORs for both pain freedom and pain relief at 2 hours, compared with lasmiditan, rimegepant, or ubrogepant, while comparisons between lasmiditan, rimegepant, and ubrogepant for these outcomes showed no statistically significant difference, they reported.
Lasmiditan was associated with the highest risk of any AEs, “however, the AEs were tolerable and were not considered serious. … Therefore, we suggest that the benefits should be weighed against the risk of its AEs when considering the clinical application of lasmiditan,” they wrote. Certain triptans (rizatriptan, sumatriptan, and zolmitriptan) were also associated with a higher risk of any AEs, compared with the CGRP antagonists. “Nevertheless, most of the AEs were mild to moderate, and the percentages of serious AEs were low (0.0%-2.1%).”
Finally, the authors noted that their observations of successful treatment with 5-hydroxytriptamine1F receptor agonists and CGRP antagonists “reveals that vasoconstriction is not essential for antimigraine therapy.” which could have implications for future pharmaceutical development.
Older and newer medications each have advantages
“Triptans will be around for a long time, but the newer medications are here to stay,” said Alan M. Rapoport, MD, in reaction to the study. “Before this publication, we knew that the 2-hour efficacy results of the newer medications were not quite as good as the faster-acting triptans; and after this network meta-analysis we are more sure of that,” said Dr. Rapoport, of the department of neurology at University of California, Los Angeles. “But the fact that the three newer medications do not constrict blood vessels and can easily be given even to patients with contraindications to triptans, or patients that simply are at greater risk due to obesity, smoking history, family history, diabetes, lack of exercise, or higher lipid levels, puts them into a desirable category.”
Calling it a “very carefully done” systematic review, Dr. Rapoport had a few caveats about the strength of the research. The trials that were included were not identically designed and were performed in different areas, by different investigators, on different patients, he noted. They were also not head-to-head trials “which ensures that the resultant data are more pure.” The studies also looked only at rapid results at 2 hours after dosing. “In my experience, patients are often satisfied with the response times from these newer agents; and doctors and patients both are happy that they are not vasoconstrictive,” he said. “The researchers also omitted studies looking at zolmitriptan nasal spray, which I have found to be rapid in onset and efficacious with few adverse events.”
Finally, Dr. Rapoport noted that one condition not examined in the review was medication overuse headache (MOH), which is “a major problem with patients that have high-frequency episodic migraine and chronic migraine. To our knowledge thus far, the two gepants (ubrogepant and rimegepant) do not appear to cause MOH when taken frequently, and these agents may end up being a treatment for this condition.”
Dr Wang reported receiving personal fees from Eli Lilly, Daiichi-Sankyo, Norvatis Taiwan, Biogen, Pfizer, and Bayer; and grants from AbbVie, Norvatis, Eli Lilly, Taiwan Ministry of Technology and Science, Brain Research Center, National Yang Ming Chiao Tung University, and Taipei Veterans General Hospital outside the submitted work. No other disclosures were reported. Dr. Rapoport serves as an advisor for AbbVie, Amgen, Biohaven, Cala Health, Satsuma, Teva Pharmaceutical Industries, Theranica, Xoc and Zosano; he is on the Speakers Bureau of AbbVie, Amgen, Biohaven, Lundbeck and Teva Pharmaceutical Industries. He is Editor-in-Chief of Neurology Reviews.
The study was funded by the Ministry of Science and Technology, Taiwan, Ministry of Education, Taiwan, and the Brain Research Center, National Yang Ming Chiao Tung University.
The findings, published in JAMA Network Open, “may imply that triptans will remain the current mainstay of specific acute migraine treatment,” suggested senior author Shuu-Jiun Wang, MD, from the National Yang Ming Chiao Tung University, and the Taipei Veterans General Hospital, both in Taipei, Taiwan, and his coauthors. However, lasmiditan (a 5-hydroxytryptamine1F receptor agonist) and rimegepant and ubrogepant (both calcitonin gene-related peptide [CGRP] antagonists) might still have unique advantages, since triptans are contraindicated for patients with cardiovascular risks, they said.
The systemic review and meta-analysis showed that, for the outcome of pain freedom and pain relief at 2 hours after the dose, the three newer agents worked better than placebo, but were inferior to most triptans. However, ubrogepant and rimegepant, which received U.S. Food and Drug Administration approval for the treatment of acute migraine in adults in December 2019 and February 2020, respectively, might be associated with fewer risks of adverse events (AEs), compared with triptans. “These new effective therapeutic options enrich the therapeutic categories of specific acute migraine treatments and may provide an opportunity to decrease the risks of barbiturate or opioid overuse or addiction,” they wrote.
The meta-analysis included 64 randomized, controlled trials involving 46,442 participants (74%-87% female across studies; age range, 36-43 years). All studies examined clinically relevant outcomes in patients with International Headache Society criteria for migraine, and compared currently available migraine-specific acute treatments with each other or placebo. The drugs were examined at doses with widespread clinical use and included: ergotamine, dihydroergotamine, sumatriptan, zolmitriptan, naratriptan, rizatriptan, almotriptan, eletriptan, frovatriptan, lasmiditan, rimegepant, and ubrogepant.
The findings showed that all drug treatments were associated with a higher odds ratio for pain freedom, compared with placebo, except for sumatriptan, 10-mg nasal spray. The most effective drug was eletriptan 40 mg (OR, 5.59), and the least effective was lasmiditan 50 mg (OR, 1.65). Most triptans were associated with higher ORs for both pain freedom and pain relief at 2 hours, compared with lasmiditan, rimegepant, or ubrogepant, while comparisons between lasmiditan, rimegepant, and ubrogepant for these outcomes showed no statistically significant difference, they reported.
Lasmiditan was associated with the highest risk of any AEs, “however, the AEs were tolerable and were not considered serious. … Therefore, we suggest that the benefits should be weighed against the risk of its AEs when considering the clinical application of lasmiditan,” they wrote. Certain triptans (rizatriptan, sumatriptan, and zolmitriptan) were also associated with a higher risk of any AEs, compared with the CGRP antagonists. “Nevertheless, most of the AEs were mild to moderate, and the percentages of serious AEs were low (0.0%-2.1%).”
Finally, the authors noted that their observations of successful treatment with 5-hydroxytriptamine1F receptor agonists and CGRP antagonists “reveals that vasoconstriction is not essential for antimigraine therapy.” which could have implications for future pharmaceutical development.
Older and newer medications each have advantages
“Triptans will be around for a long time, but the newer medications are here to stay,” said Alan M. Rapoport, MD, in reaction to the study. “Before this publication, we knew that the 2-hour efficacy results of the newer medications were not quite as good as the faster-acting triptans; and after this network meta-analysis we are more sure of that,” said Dr. Rapoport, of the department of neurology at University of California, Los Angeles. “But the fact that the three newer medications do not constrict blood vessels and can easily be given even to patients with contraindications to triptans, or patients that simply are at greater risk due to obesity, smoking history, family history, diabetes, lack of exercise, or higher lipid levels, puts them into a desirable category.”
Calling it a “very carefully done” systematic review, Dr. Rapoport had a few caveats about the strength of the research. The trials that were included were not identically designed and were performed in different areas, by different investigators, on different patients, he noted. They were also not head-to-head trials “which ensures that the resultant data are more pure.” The studies also looked only at rapid results at 2 hours after dosing. “In my experience, patients are often satisfied with the response times from these newer agents; and doctors and patients both are happy that they are not vasoconstrictive,” he said. “The researchers also omitted studies looking at zolmitriptan nasal spray, which I have found to be rapid in onset and efficacious with few adverse events.”
Finally, Dr. Rapoport noted that one condition not examined in the review was medication overuse headache (MOH), which is “a major problem with patients that have high-frequency episodic migraine and chronic migraine. To our knowledge thus far, the two gepants (ubrogepant and rimegepant) do not appear to cause MOH when taken frequently, and these agents may end up being a treatment for this condition.”
Dr Wang reported receiving personal fees from Eli Lilly, Daiichi-Sankyo, Norvatis Taiwan, Biogen, Pfizer, and Bayer; and grants from AbbVie, Norvatis, Eli Lilly, Taiwan Ministry of Technology and Science, Brain Research Center, National Yang Ming Chiao Tung University, and Taipei Veterans General Hospital outside the submitted work. No other disclosures were reported. Dr. Rapoport serves as an advisor for AbbVie, Amgen, Biohaven, Cala Health, Satsuma, Teva Pharmaceutical Industries, Theranica, Xoc and Zosano; he is on the Speakers Bureau of AbbVie, Amgen, Biohaven, Lundbeck and Teva Pharmaceutical Industries. He is Editor-in-Chief of Neurology Reviews.
The study was funded by the Ministry of Science and Technology, Taiwan, Ministry of Education, Taiwan, and the Brain Research Center, National Yang Ming Chiao Tung University.
The findings, published in JAMA Network Open, “may imply that triptans will remain the current mainstay of specific acute migraine treatment,” suggested senior author Shuu-Jiun Wang, MD, from the National Yang Ming Chiao Tung University, and the Taipei Veterans General Hospital, both in Taipei, Taiwan, and his coauthors. However, lasmiditan (a 5-hydroxytryptamine1F receptor agonist) and rimegepant and ubrogepant (both calcitonin gene-related peptide [CGRP] antagonists) might still have unique advantages, since triptans are contraindicated for patients with cardiovascular risks, they said.
The systemic review and meta-analysis showed that, for the outcome of pain freedom and pain relief at 2 hours after the dose, the three newer agents worked better than placebo, but were inferior to most triptans. However, ubrogepant and rimegepant, which received U.S. Food and Drug Administration approval for the treatment of acute migraine in adults in December 2019 and February 2020, respectively, might be associated with fewer risks of adverse events (AEs), compared with triptans. “These new effective therapeutic options enrich the therapeutic categories of specific acute migraine treatments and may provide an opportunity to decrease the risks of barbiturate or opioid overuse or addiction,” they wrote.
The meta-analysis included 64 randomized, controlled trials involving 46,442 participants (74%-87% female across studies; age range, 36-43 years). All studies examined clinically relevant outcomes in patients with International Headache Society criteria for migraine, and compared currently available migraine-specific acute treatments with each other or placebo. The drugs were examined at doses with widespread clinical use and included: ergotamine, dihydroergotamine, sumatriptan, zolmitriptan, naratriptan, rizatriptan, almotriptan, eletriptan, frovatriptan, lasmiditan, rimegepant, and ubrogepant.
The findings showed that all drug treatments were associated with a higher odds ratio for pain freedom, compared with placebo, except for sumatriptan, 10-mg nasal spray. The most effective drug was eletriptan 40 mg (OR, 5.59), and the least effective was lasmiditan 50 mg (OR, 1.65). Most triptans were associated with higher ORs for both pain freedom and pain relief at 2 hours, compared with lasmiditan, rimegepant, or ubrogepant, while comparisons between lasmiditan, rimegepant, and ubrogepant for these outcomes showed no statistically significant difference, they reported.
Lasmiditan was associated with the highest risk of any AEs, “however, the AEs were tolerable and were not considered serious. … Therefore, we suggest that the benefits should be weighed against the risk of its AEs when considering the clinical application of lasmiditan,” they wrote. Certain triptans (rizatriptan, sumatriptan, and zolmitriptan) were also associated with a higher risk of any AEs, compared with the CGRP antagonists. “Nevertheless, most of the AEs were mild to moderate, and the percentages of serious AEs were low (0.0%-2.1%).”
Finally, the authors noted that their observations of successful treatment with 5-hydroxytriptamine1F receptor agonists and CGRP antagonists “reveals that vasoconstriction is not essential for antimigraine therapy.” which could have implications for future pharmaceutical development.
Older and newer medications each have advantages
“Triptans will be around for a long time, but the newer medications are here to stay,” said Alan M. Rapoport, MD, in reaction to the study. “Before this publication, we knew that the 2-hour efficacy results of the newer medications were not quite as good as the faster-acting triptans; and after this network meta-analysis we are more sure of that,” said Dr. Rapoport, of the department of neurology at University of California, Los Angeles. “But the fact that the three newer medications do not constrict blood vessels and can easily be given even to patients with contraindications to triptans, or patients that simply are at greater risk due to obesity, smoking history, family history, diabetes, lack of exercise, or higher lipid levels, puts them into a desirable category.”
Calling it a “very carefully done” systematic review, Dr. Rapoport had a few caveats about the strength of the research. The trials that were included were not identically designed and were performed in different areas, by different investigators, on different patients, he noted. They were also not head-to-head trials “which ensures that the resultant data are more pure.” The studies also looked only at rapid results at 2 hours after dosing. “In my experience, patients are often satisfied with the response times from these newer agents; and doctors and patients both are happy that they are not vasoconstrictive,” he said. “The researchers also omitted studies looking at zolmitriptan nasal spray, which I have found to be rapid in onset and efficacious with few adverse events.”
Finally, Dr. Rapoport noted that one condition not examined in the review was medication overuse headache (MOH), which is “a major problem with patients that have high-frequency episodic migraine and chronic migraine. To our knowledge thus far, the two gepants (ubrogepant and rimegepant) do not appear to cause MOH when taken frequently, and these agents may end up being a treatment for this condition.”
Dr Wang reported receiving personal fees from Eli Lilly, Daiichi-Sankyo, Norvatis Taiwan, Biogen, Pfizer, and Bayer; and grants from AbbVie, Norvatis, Eli Lilly, Taiwan Ministry of Technology and Science, Brain Research Center, National Yang Ming Chiao Tung University, and Taipei Veterans General Hospital outside the submitted work. No other disclosures were reported. Dr. Rapoport serves as an advisor for AbbVie, Amgen, Biohaven, Cala Health, Satsuma, Teva Pharmaceutical Industries, Theranica, Xoc and Zosano; he is on the Speakers Bureau of AbbVie, Amgen, Biohaven, Lundbeck and Teva Pharmaceutical Industries. He is Editor-in-Chief of Neurology Reviews.
The study was funded by the Ministry of Science and Technology, Taiwan, Ministry of Education, Taiwan, and the Brain Research Center, National Yang Ming Chiao Tung University.
FROM JAMA NETWORK OPEN

