User login
Treatment of opioid use disorder in hospitalized patients
An opportunity for impact
Case
A 35-year-old woman with opioid use disorder (OUD) presents with fever, left arm redness, and swelling. She is admitted to the hospital for cellulitis treatment. On the day after admission she becomes agitated and develops nausea, diarrhea, and generalized pain. Opioid withdrawal is suspected. How should her opioid use be addressed while in the hospital?
Brief overview of the issue
Since 1999, there have been more than 800,000 deaths related to drug overdose in the United States, and in 2019 more than 70% of drug overdose deaths involved an opioid.1,2 Although effective treatments for OUD exist, less than 20% of those with OUD are engaged in treatment.3
In America, 4%-11% of hospitalized patients have OUD. Hospitalized patients with OUD often experience stigma surrounding their disease, and many inpatient clinicians lack knowledge regarding the care of patients with OUD. As a result, withdrawal symptoms may go untreated, which can erode trust in the medical system and contribute to patients’ leaving the hospital before their primary medical issue is fully addressed. Therefore, it is essential that inpatient clinicians be familiar with the management of this complex and vulnerable patient population. Initiating treatment for OUD in the hospital setting is feasible and effective, and can lead to increased engagement in OUD treatment even after the hospital stay.
Overview of the data
Assessing patients with suspected OUD
Assessment for OUD starts with an in-depth opioid use history including frequency, amount, and method of administration. Clinicians should gather information regarding use of other substances or nonprescribed medications, and take thorough psychiatric and social histories. A formal diagnosis of OUD can be made using the Fifth Edition Diagnostic and Statistical Manual for Mental Disorders (DSM-5) diagnostic criteria.
Recognizing and managing opioid withdrawal
OUD in hospitalized patients often becomes apparent when patients develop signs and symptoms of withdrawal. Decreasing physical discomfort related to withdrawal can allow inpatient clinicians to address the condition for which the patient was hospitalized, help to strengthen the patient-clinician relationship, and provide an opportunity to discuss long-term OUD treatment.
Signs and symptoms of opioid withdrawal include anxiety, restlessness, irritability, generalized pain, rhinorrhea, yawning, lacrimation, piloerection, anorexia, and nausea. Withdrawal can last days to weeks, depending on the half-life of the opioid that was used. Opioids with shorter half-lives, such as heroin or oxycodone, cause withdrawal with earlier onset and shorter duration than do opioids with longer half-lives, such as methadone. The degree of withdrawal can be quantified with validated tools, such as the Clinical Opiate Withdrawal Scale (COWS).
Treatment of opioid withdrawal should generally include the use of an opioid agonist such as methadone or buprenorphine. A 2017 Cochrane meta-analysis found methadone or buprenorphine to be more effective than clonidine in alleviating symptoms of withdrawal and in retaining patients in treatment.4 Clonidine, an alpha2-adrenergic agonist that binds to receptors in the locus coeruleus, does not alleviate opioid cravings, but may be used as an adjunctive treatment for associated autonomic withdrawal symptoms. Other adjunctive medications include analgesics, antiemetics, antidiarrheals, and antihistamines.
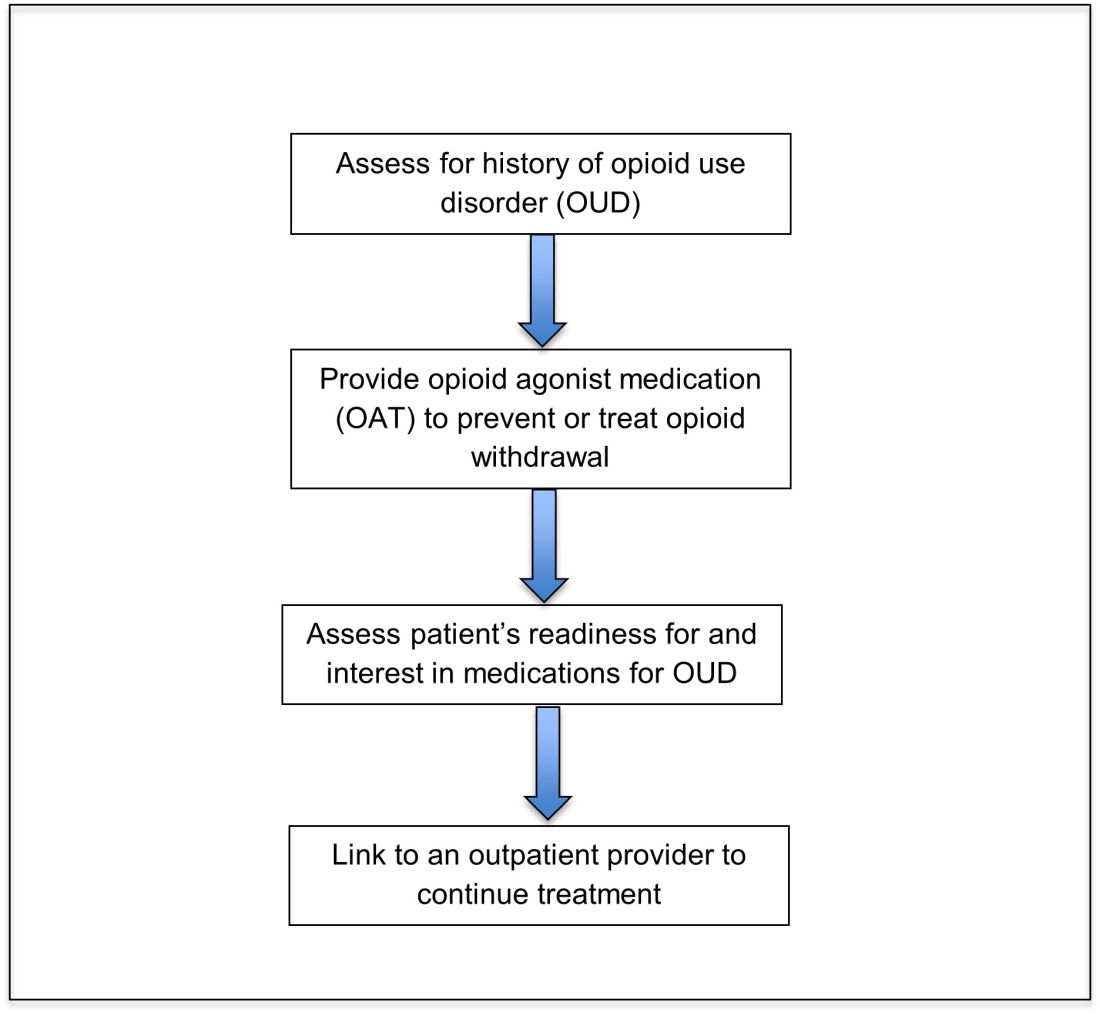
Opioid agonist treatment for opioid use disorder
Opioid agonist treatment (OAT) with methadone or buprenorphine is associated with decreased mortality, opioid use, and infectious complications, but remains underutilized.5 Hospitalized patients with OUD are frequently managed with a rapid opioid detoxification and then discharged without continued OUD treatment. Detoxification alone can lead to a relapse rate as high as 90%.6 Patients are at increased risk for overdose after withdrawal due to loss of tolerance. Inpatient clinicians can close this OUD treatment gap by familiarizing themselves with OAT and offering to initiate OAT for maintenance treatment in interested patients. In one study, patients started on buprenorphine while hospitalized were more likely to be engaged in treatment and less likely to report drug use at follow-up, compared to patients who were referred without starting the medication.7
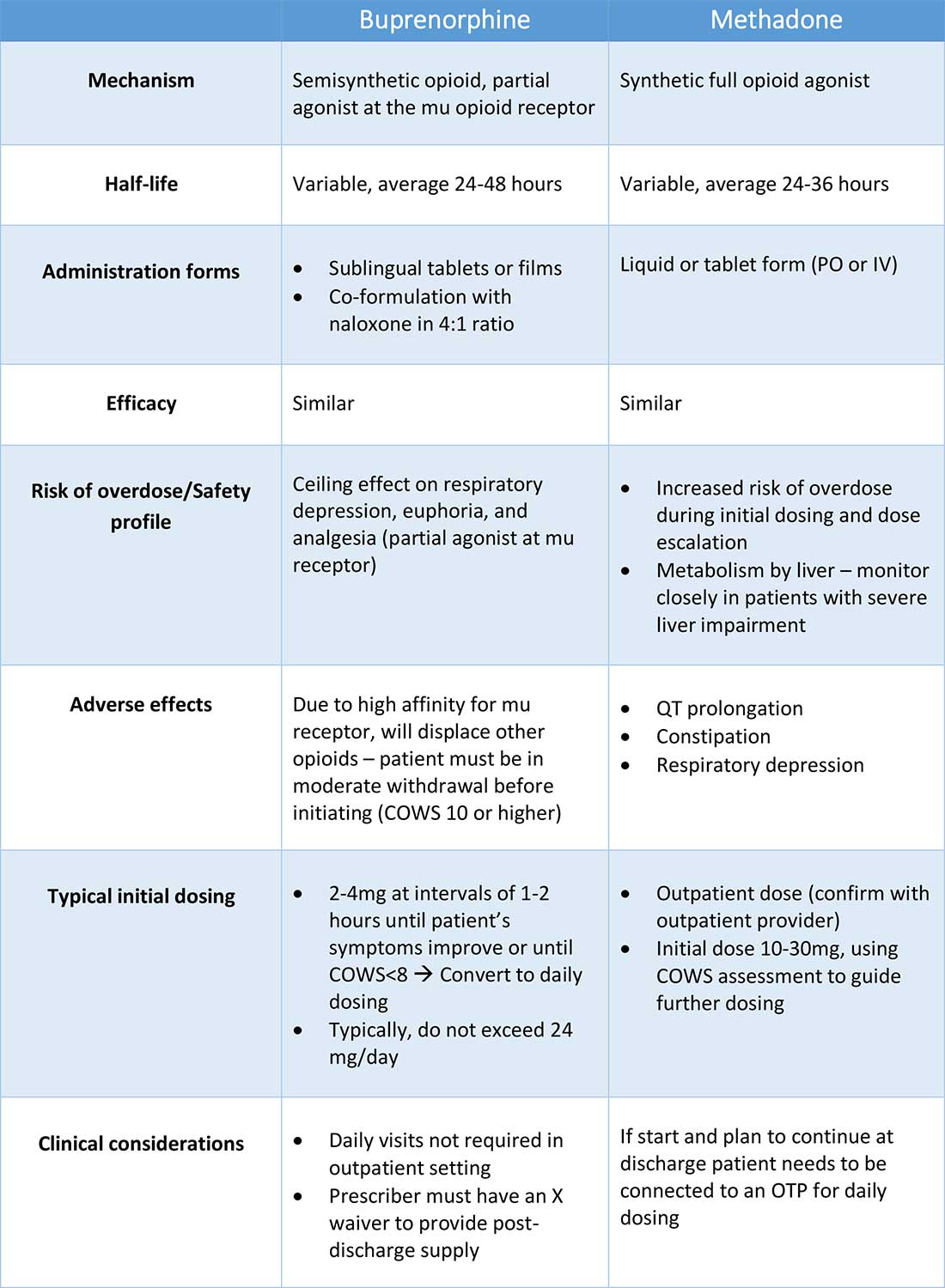
Buprenorphine
Buprenorphine is a partial agonist at the mu opioid receptor that can be ordered in the inpatient setting by any clinician. In the outpatient setting only DATA 2000 waivered clinicians can prescribe buprenorphine.8 Buprenorphine is most commonly coformulated with naloxone, an opioid antagonist, and is available in sublingual films or tablets. The naloxone component is not bioavailable when taken sublingually but becomes bioavailable if the drug is injected intravenously, leading to acute withdrawal.
Buprenorphine has a higher affinity for the mu opioid receptor than most opioids. If administered while other opioids are still present, it will displace the other opioid from the receptor but only partially stimulate the receptor, which can cause precipitated withdrawal. Buprenorphine initiation can start when the COWS score reflects moderate withdrawal. Many institutions use a threshold of 8-12 on the COWS scale. Typical dosing is 2-4 mg of buprenorphine at intervals of 1-2 hours as needed until the COWS score is less than 8, up to a maximum of 16 mg on day 1. The total dose from day 1 may be given as a daily dose beginning on day 2, up to a maximum total daily dose of 24 mg.
In recent years, a method of initiating buprenorphine called “micro-dosing” has gained traction. Very small doses of buprenorphine are given while a patient is receiving other opioids, thereby reducing the risk of precipitated withdrawal. This method can be helpful for patients who cannot tolerate withdrawal or who have recently taken long-acting opioids such as methadone. Such protocols should be utilized only at centers where consultation with an addiction specialist or experienced clinician is possible.
Despite evidence of buprenorphine’s efficacy, there are barriers to prescribing it. Physicians and advanced practitioners must be granted a waiver from the Drug Enforcement Administration to prescribe buprenorphine to outpatients. As of 2017, less than 10% of primary care physicians had obtained waivers.9 However, inpatient clinicians without a waiver can order buprenorphine and initiate treatment. Best practice is to do so with a specific plan for continuation at discharge. We encourage inpatient clinicians to obtain a waiver, so that a prescription can be given at discharge to bridge the patient to a first appointment with a community clinician who can continue treatment. As of April 27, 2021, providers treating fewer than 30 patients with OUD at one time may obtain a waiver without additional training.10
Methadone
Methadone is a full agonist at the mu opioid receptor. In the hospital setting, methadone can be ordered by any clinician to prevent and treat withdrawal. Commonly, doses of 10 mg can be given using the COWS score to guide the need for additional dosing. The patient can be reassessed every 1-2 hours to ensure that symptoms are improving, and that there is no sign of oversedation before giving additional methadone. For most patients, withdrawal can be managed with 20-40 mg of methadone daily.
In contrast to buprenorphine, methadone will not precipitate withdrawal and can be initiated even when patients are not yet showing withdrawal symptoms. Outpatient methadone treatment for OUD is federally regulated and can be delivered only in opioid treatment programs (OTPs).
Choosing methadone or buprenorphine in the inpatient setting
The choice between buprenorphine and methadone should take into consideration several factors, including patient preference, treatment history, and available outpatient treatment programs, which may vary widely by geographic region. Some patients benefit from the higher level of support and counseling available at OTPs. Methadone is available at all OTPs, and the availability of buprenorphine in this setting is increasing. Other patients may prefer the convenience and flexibility of buprenorphine treatment in an outpatient office setting.
Some patients have prior negative experiences with OAT. These can include prior precipitated withdrawal with buprenorphine induction, or negative experiences with the structure of OTPs. Clinicians are encouraged to provide counseling if patients have a history of precipitated withdrawal to assure them that this can be avoided with proper dosing. Clinicians should be familiar with available treatment options in their community and can refer to the Substance Abuse and Mental Health Services Administration (SAMHSA) website to locate OTPs and buprenorphine prescribers.
Polypharmacy and safety
If combined with benzodiazepines, alcohol, or other sedating agents, methadone or buprenorphine can increase risk of overdose. However, OUD treatment should not be withheld because of other substance use. Clinicians initiating treatment should counsel patients on the risk of concomitant substance use and provide overdose prevention education.
A brief note on naltrexone
Naltrexone, an opioid antagonist, is used more commonly in outpatient addiction treatment than in the inpatient setting, but inpatient clinicians should be aware of its use. It is available in oral and long-acting injectable formulations. Its utility in the inpatient setting may be limited as safe administration requires 7-10 days of opioid abstinence.
Discharge planning
Patients with OUD or who are started on OAT during a hospitalization should be linked to continued outpatient treatment. Before discharge it is best to ensure vaccinations for HAV, HBV, pneumococcus, and tetanus are up to date, and perform screening for HIV, hepatitis C, tuberculosis, and sexually transmitted infections if appropriate. All patients with OUD should be prescribed or provided with take-home naloxone for overdose reversal. Patients can also be referred to syringe service programs for additional harm reduction counseling and services.
Application of the data to our patient
For our patient, either methadone or buprenorphine could be used to treat her withdrawal. The COWS score should be used to assess withdrawal severity, and to guide appropriate timing of medication initiation. If she wishes to continue OAT after discharge, she should be linked to a clinician who can engage her in ongoing medical care. Prior to discharge she should also receive relevant vaccines and screening for infectious diseases as outlined above, as well as take-home naloxone (or a prescription).
Bottom line
Inpatient clinicians can play a pivotal role in patients’ lives by ensuring that patients with OUD receive OAT and are connected to outpatient care at discharge.
Dr. Linker is assistant professor in the division of hospital medicine, Icahn School of Medicine at Mount Sinai, New York. Ms. Hirt, Mr. Fine, and Mr. Villasanivis are medical students at the Icahn School of Medicine at Mount Sinai. Dr. Wang is assistant professor in the division of general internal medicine, Icahn School of Medicine at Mount Sinai. Dr. Herscher is assistant professor in the division of hospital medicine, Icahn School of Medicine at Mount Sinai.
References
1. Wide-ranging online data for epidemiologic research (WONDER). Atlanta, GA: CDC, National Center for Health Statistics; 2020. Available at http://wonder.cdc.gov.
2. Mattson CL et al. Trends and geographic patterns in drug and synthetic opioid overdose deaths – United States, 2013-2019. MMWR Morb Mortal Wkly Rep. 2021;70:202-7. doi: 10.15585/mmwr.mm7006a4.
3. Wakeman SE et al. Comparative effectiveness of different treatment pathways for opioid use disorder. JAMA Netw Open. 2020 Feb 5;3(2):e1920622. doi: 10.1001/jamanetworkopen.2019.20622.
4. Gowing L et al. Buprenorphine for managing opioid withdrawal. Cochrane Database Syst Rev. 2017 Feb;2017(2):CD002025. doi: 10.1002/14651858.CD002025.pub5.
5. Sordo L et al. Mortality risk during and after opioid substitution treatment: Systematic review and meta-analysis of cohort studies. BMJ. 2017 Apr 26;357:j1550. doi: 10.1136/bmj.j1550.
6. Smyth BP et al. Lapse and relapse following inpatient treatment of opiate dependence. Ir Med J. 2010 Jun;103(6):176-9. Available at www.drugsandalcohol.ie/13405.
7. Liebschutz JM. Buprenorphine treatment for hospitalized, opioid-dependent patients: A randomized clinical trial. JAMA Intern Med. 2014 Aug;174(8):1369-76. doi: 10.1001/jamainternmed.2014.2556.
8. Substance Abuse and Mental Health Services Administration. (Aug 20, 2020) Statutes, Regulations, and Guidelines.
9. McBain RK et al. Growth and distribution of buprenorphine-waivered providers in the United States, 2007-2017. Ann Intern Med. 2020;172(7):504-6. doi: 10.7326/M19-2403.
10. HHS releases new buprenorphine practice guidelines, expanding access to treatment for opioid use disorder. Apr 27, 2021.
11. Herscher M et al. Diagnosis and management of opioid use disorder in hospitalized patients. Med Clin North Am. 2020 Jul;104(4):695-708. doi: 10.1016/j.mcna.2020.03.003.
Additional reading
Winetsky D. Expanding treatment opportunities for hospitalized patients with opioid use disorders. J Hosp Med. 2018 Jan;13(1):62-4. doi: 10.12788/jhm.2861.
Donroe JH. Caring for patients with opioid use disorder in the hospital. Can Med Assoc J. 2016 Dec 6;188(17-18):1232-9. doi: 10.1503/cmaj.160290.
Herscher M et al. Diagnosis and management of opioid use disorder in hospitalized patients. Med Clin North Am. 2020 Jul;104(4):695-708. doi: 10.1016/j.mcna.2020.03.003.
Key points
- Most patients with OUD are not engaged in evidence-based treatment. Clinicians have an opportunity to utilize the inpatient stay as a ‘reachable moment’ to engage patients with OUD in evidence-based treatment.
- Buprenorphine and methadone are effective opioid agonist medications used to treat OUD, and clinicians with the appropriate knowledge base can initiate either during the inpatient encounter, and link the patient to OUD treatment after the hospital stay.
Quiz
Caring for hospitalized patients with OUD
Most patients with OUD are not engaged in effective treatment. Hospitalization can be a ‘reachable moment’ to engage patients with OUD in evidence-based treatment.
1. Which is an effective and evidence-based medication for treating opioid withdrawal and OUD?
a) Naltrexone.
b) Buprenorphine.
c) Opioid detoxification.
d) Clonidine.
Explanation: Buprenorphine is effective for alleviating symptoms of withdrawal as well as for the long-term treatment of OUD. While naltrexone is also used to treat OUD, it is not useful for treating withdrawal. Clonidine can be a useful adjunctive medication for treating withdrawal but is not a long-term treatment for OUD. Nonpharmacologic detoxification is not an effective treatment for OUD and is associated with high relapse rates.
2. What scale can be used during a hospital stay to monitor patients with OUD at risk of opioid withdrawal, and to aid in buprenorphine initiation?
a) CIWA score.
b) PADUA score.
c) COWS score.
d) 4T score.
Explanation: COWS is the “clinical opiate withdrawal scale.” The COWS score should be calculated by a trained provider, and includes objective parameters (such as pulse) and subjective symptoms (such as GI upset, bone/joint aches.) It is recommended that agonist therapy be started when the COWS score is consistent with moderate withdrawal.
3. How can clinicians reliably find out if there are outpatient resources/clinics for patients with OUD in their area?
a) No way to find this out without personal knowledge.
b) Hospital providers and patients can visit www.samhsa.gov/find-help/national-helpline or call 1-800-662-HELP (4357) to find options for treatment for substance use disorders in their areas.
c) Dial “0” on any phone and ask.
d) Ask around at your hospital.
Explanation: The Substance Abuse and Mental Health Services Administration (SAMHSA) is an agency in the U.S. Department of Health and Human Services that is engaged in public health efforts to reduce the impact of substance abuse and mental illness on local communities. The agency’s website has helpful information about resources for substance use treatment.
4. Patients with OUD should be prescribed and given training about what medication that can be lifesaving when given during an opioid overdose?
a) Aspirin.
b) Naloxone.
c) Naltrexone.
d) Clonidine.
Explanation: Naloxone can be life-saving in the setting of an overdose. Best practice is to provide naloxone and training to patients with OUD.
5. When patients take buprenorphine soon after taking other opioids, there is concern for the development of which reaction:
a) Precipitated withdrawal.
b) Opioid overdose.
c) Allergic reaction.
d) Intoxication.
Explanation: Administering buprenorphine soon after taking other opioids can cause precipitated withdrawal, as buprenorphine binds with higher affinity to the mu receptor than many opioids. Precipitated withdrawal causes severe discomfort and can be dangerous for patients.
An opportunity for impact
An opportunity for impact
Case
A 35-year-old woman with opioid use disorder (OUD) presents with fever, left arm redness, and swelling. She is admitted to the hospital for cellulitis treatment. On the day after admission she becomes agitated and develops nausea, diarrhea, and generalized pain. Opioid withdrawal is suspected. How should her opioid use be addressed while in the hospital?
Brief overview of the issue
Since 1999, there have been more than 800,000 deaths related to drug overdose in the United States, and in 2019 more than 70% of drug overdose deaths involved an opioid.1,2 Although effective treatments for OUD exist, less than 20% of those with OUD are engaged in treatment.3
In America, 4%-11% of hospitalized patients have OUD. Hospitalized patients with OUD often experience stigma surrounding their disease, and many inpatient clinicians lack knowledge regarding the care of patients with OUD. As a result, withdrawal symptoms may go untreated, which can erode trust in the medical system and contribute to patients’ leaving the hospital before their primary medical issue is fully addressed. Therefore, it is essential that inpatient clinicians be familiar with the management of this complex and vulnerable patient population. Initiating treatment for OUD in the hospital setting is feasible and effective, and can lead to increased engagement in OUD treatment even after the hospital stay.
Overview of the data
Assessing patients with suspected OUD
Assessment for OUD starts with an in-depth opioid use history including frequency, amount, and method of administration. Clinicians should gather information regarding use of other substances or nonprescribed medications, and take thorough psychiatric and social histories. A formal diagnosis of OUD can be made using the Fifth Edition Diagnostic and Statistical Manual for Mental Disorders (DSM-5) diagnostic criteria.
Recognizing and managing opioid withdrawal
OUD in hospitalized patients often becomes apparent when patients develop signs and symptoms of withdrawal. Decreasing physical discomfort related to withdrawal can allow inpatient clinicians to address the condition for which the patient was hospitalized, help to strengthen the patient-clinician relationship, and provide an opportunity to discuss long-term OUD treatment.
Signs and symptoms of opioid withdrawal include anxiety, restlessness, irritability, generalized pain, rhinorrhea, yawning, lacrimation, piloerection, anorexia, and nausea. Withdrawal can last days to weeks, depending on the half-life of the opioid that was used. Opioids with shorter half-lives, such as heroin or oxycodone, cause withdrawal with earlier onset and shorter duration than do opioids with longer half-lives, such as methadone. The degree of withdrawal can be quantified with validated tools, such as the Clinical Opiate Withdrawal Scale (COWS).
Treatment of opioid withdrawal should generally include the use of an opioid agonist such as methadone or buprenorphine. A 2017 Cochrane meta-analysis found methadone or buprenorphine to be more effective than clonidine in alleviating symptoms of withdrawal and in retaining patients in treatment.4 Clonidine, an alpha2-adrenergic agonist that binds to receptors in the locus coeruleus, does not alleviate opioid cravings, but may be used as an adjunctive treatment for associated autonomic withdrawal symptoms. Other adjunctive medications include analgesics, antiemetics, antidiarrheals, and antihistamines.

Opioid agonist treatment for opioid use disorder
Opioid agonist treatment (OAT) with methadone or buprenorphine is associated with decreased mortality, opioid use, and infectious complications, but remains underutilized.5 Hospitalized patients with OUD are frequently managed with a rapid opioid detoxification and then discharged without continued OUD treatment. Detoxification alone can lead to a relapse rate as high as 90%.6 Patients are at increased risk for overdose after withdrawal due to loss of tolerance. Inpatient clinicians can close this OUD treatment gap by familiarizing themselves with OAT and offering to initiate OAT for maintenance treatment in interested patients. In one study, patients started on buprenorphine while hospitalized were more likely to be engaged in treatment and less likely to report drug use at follow-up, compared to patients who were referred without starting the medication.7
Buprenorphine
Buprenorphine is a partial agonist at the mu opioid receptor that can be ordered in the inpatient setting by any clinician. In the outpatient setting only DATA 2000 waivered clinicians can prescribe buprenorphine.8 Buprenorphine is most commonly coformulated with naloxone, an opioid antagonist, and is available in sublingual films or tablets. The naloxone component is not bioavailable when taken sublingually but becomes bioavailable if the drug is injected intravenously, leading to acute withdrawal.
Buprenorphine has a higher affinity for the mu opioid receptor than most opioids. If administered while other opioids are still present, it will displace the other opioid from the receptor but only partially stimulate the receptor, which can cause precipitated withdrawal. Buprenorphine initiation can start when the COWS score reflects moderate withdrawal. Many institutions use a threshold of 8-12 on the COWS scale. Typical dosing is 2-4 mg of buprenorphine at intervals of 1-2 hours as needed until the COWS score is less than 8, up to a maximum of 16 mg on day 1. The total dose from day 1 may be given as a daily dose beginning on day 2, up to a maximum total daily dose of 24 mg.
In recent years, a method of initiating buprenorphine called “micro-dosing” has gained traction. Very small doses of buprenorphine are given while a patient is receiving other opioids, thereby reducing the risk of precipitated withdrawal. This method can be helpful for patients who cannot tolerate withdrawal or who have recently taken long-acting opioids such as methadone. Such protocols should be utilized only at centers where consultation with an addiction specialist or experienced clinician is possible.
Despite evidence of buprenorphine’s efficacy, there are barriers to prescribing it. Physicians and advanced practitioners must be granted a waiver from the Drug Enforcement Administration to prescribe buprenorphine to outpatients. As of 2017, less than 10% of primary care physicians had obtained waivers.9 However, inpatient clinicians without a waiver can order buprenorphine and initiate treatment. Best practice is to do so with a specific plan for continuation at discharge. We encourage inpatient clinicians to obtain a waiver, so that a prescription can be given at discharge to bridge the patient to a first appointment with a community clinician who can continue treatment. As of April 27, 2021, providers treating fewer than 30 patients with OUD at one time may obtain a waiver without additional training.10
Methadone
Methadone is a full agonist at the mu opioid receptor. In the hospital setting, methadone can be ordered by any clinician to prevent and treat withdrawal. Commonly, doses of 10 mg can be given using the COWS score to guide the need for additional dosing. The patient can be reassessed every 1-2 hours to ensure that symptoms are improving, and that there is no sign of oversedation before giving additional methadone. For most patients, withdrawal can be managed with 20-40 mg of methadone daily.
In contrast to buprenorphine, methadone will not precipitate withdrawal and can be initiated even when patients are not yet showing withdrawal symptoms. Outpatient methadone treatment for OUD is federally regulated and can be delivered only in opioid treatment programs (OTPs).
Choosing methadone or buprenorphine in the inpatient setting
The choice between buprenorphine and methadone should take into consideration several factors, including patient preference, treatment history, and available outpatient treatment programs, which may vary widely by geographic region. Some patients benefit from the higher level of support and counseling available at OTPs. Methadone is available at all OTPs, and the availability of buprenorphine in this setting is increasing. Other patients may prefer the convenience and flexibility of buprenorphine treatment in an outpatient office setting.
Some patients have prior negative experiences with OAT. These can include prior precipitated withdrawal with buprenorphine induction, or negative experiences with the structure of OTPs. Clinicians are encouraged to provide counseling if patients have a history of precipitated withdrawal to assure them that this can be avoided with proper dosing. Clinicians should be familiar with available treatment options in their community and can refer to the Substance Abuse and Mental Health Services Administration (SAMHSA) website to locate OTPs and buprenorphine prescribers.

Polypharmacy and safety
If combined with benzodiazepines, alcohol, or other sedating agents, methadone or buprenorphine can increase risk of overdose. However, OUD treatment should not be withheld because of other substance use. Clinicians initiating treatment should counsel patients on the risk of concomitant substance use and provide overdose prevention education.
A brief note on naltrexone
Naltrexone, an opioid antagonist, is used more commonly in outpatient addiction treatment than in the inpatient setting, but inpatient clinicians should be aware of its use. It is available in oral and long-acting injectable formulations. Its utility in the inpatient setting may be limited as safe administration requires 7-10 days of opioid abstinence.
Discharge planning
Patients with OUD or who are started on OAT during a hospitalization should be linked to continued outpatient treatment. Before discharge it is best to ensure vaccinations for HAV, HBV, pneumococcus, and tetanus are up to date, and perform screening for HIV, hepatitis C, tuberculosis, and sexually transmitted infections if appropriate. All patients with OUD should be prescribed or provided with take-home naloxone for overdose reversal. Patients can also be referred to syringe service programs for additional harm reduction counseling and services.
Application of the data to our patient
For our patient, either methadone or buprenorphine could be used to treat her withdrawal. The COWS score should be used to assess withdrawal severity, and to guide appropriate timing of medication initiation. If she wishes to continue OAT after discharge, she should be linked to a clinician who can engage her in ongoing medical care. Prior to discharge she should also receive relevant vaccines and screening for infectious diseases as outlined above, as well as take-home naloxone (or a prescription).
Bottom line
Inpatient clinicians can play a pivotal role in patients’ lives by ensuring that patients with OUD receive OAT and are connected to outpatient care at discharge.
Dr. Linker is assistant professor in the division of hospital medicine, Icahn School of Medicine at Mount Sinai, New York. Ms. Hirt, Mr. Fine, and Mr. Villasanivis are medical students at the Icahn School of Medicine at Mount Sinai. Dr. Wang is assistant professor in the division of general internal medicine, Icahn School of Medicine at Mount Sinai. Dr. Herscher is assistant professor in the division of hospital medicine, Icahn School of Medicine at Mount Sinai.
References
1. Wide-ranging online data for epidemiologic research (WONDER). Atlanta, GA: CDC, National Center for Health Statistics; 2020. Available at http://wonder.cdc.gov.
2. Mattson CL et al. Trends and geographic patterns in drug and synthetic opioid overdose deaths – United States, 2013-2019. MMWR Morb Mortal Wkly Rep. 2021;70:202-7. doi: 10.15585/mmwr.mm7006a4.
3. Wakeman SE et al. Comparative effectiveness of different treatment pathways for opioid use disorder. JAMA Netw Open. 2020 Feb 5;3(2):e1920622. doi: 10.1001/jamanetworkopen.2019.20622.
4. Gowing L et al. Buprenorphine for managing opioid withdrawal. Cochrane Database Syst Rev. 2017 Feb;2017(2):CD002025. doi: 10.1002/14651858.CD002025.pub5.
5. Sordo L et al. Mortality risk during and after opioid substitution treatment: Systematic review and meta-analysis of cohort studies. BMJ. 2017 Apr 26;357:j1550. doi: 10.1136/bmj.j1550.
6. Smyth BP et al. Lapse and relapse following inpatient treatment of opiate dependence. Ir Med J. 2010 Jun;103(6):176-9. Available at www.drugsandalcohol.ie/13405.
7. Liebschutz JM. Buprenorphine treatment for hospitalized, opioid-dependent patients: A randomized clinical trial. JAMA Intern Med. 2014 Aug;174(8):1369-76. doi: 10.1001/jamainternmed.2014.2556.
8. Substance Abuse and Mental Health Services Administration. (Aug 20, 2020) Statutes, Regulations, and Guidelines.
9. McBain RK et al. Growth and distribution of buprenorphine-waivered providers in the United States, 2007-2017. Ann Intern Med. 2020;172(7):504-6. doi: 10.7326/M19-2403.
10. HHS releases new buprenorphine practice guidelines, expanding access to treatment for opioid use disorder. Apr 27, 2021.
11. Herscher M et al. Diagnosis and management of opioid use disorder in hospitalized patients. Med Clin North Am. 2020 Jul;104(4):695-708. doi: 10.1016/j.mcna.2020.03.003.
Additional reading
Winetsky D. Expanding treatment opportunities for hospitalized patients with opioid use disorders. J Hosp Med. 2018 Jan;13(1):62-4. doi: 10.12788/jhm.2861.
Donroe JH. Caring for patients with opioid use disorder in the hospital. Can Med Assoc J. 2016 Dec 6;188(17-18):1232-9. doi: 10.1503/cmaj.160290.
Herscher M et al. Diagnosis and management of opioid use disorder in hospitalized patients. Med Clin North Am. 2020 Jul;104(4):695-708. doi: 10.1016/j.mcna.2020.03.003.
Key points
- Most patients with OUD are not engaged in evidence-based treatment. Clinicians have an opportunity to utilize the inpatient stay as a ‘reachable moment’ to engage patients with OUD in evidence-based treatment.
- Buprenorphine and methadone are effective opioid agonist medications used to treat OUD, and clinicians with the appropriate knowledge base can initiate either during the inpatient encounter, and link the patient to OUD treatment after the hospital stay.
Quiz
Caring for hospitalized patients with OUD
Most patients with OUD are not engaged in effective treatment. Hospitalization can be a ‘reachable moment’ to engage patients with OUD in evidence-based treatment.
1. Which is an effective and evidence-based medication for treating opioid withdrawal and OUD?
a) Naltrexone.
b) Buprenorphine.
c) Opioid detoxification.
d) Clonidine.
Explanation: Buprenorphine is effective for alleviating symptoms of withdrawal as well as for the long-term treatment of OUD. While naltrexone is also used to treat OUD, it is not useful for treating withdrawal. Clonidine can be a useful adjunctive medication for treating withdrawal but is not a long-term treatment for OUD. Nonpharmacologic detoxification is not an effective treatment for OUD and is associated with high relapse rates.
2. What scale can be used during a hospital stay to monitor patients with OUD at risk of opioid withdrawal, and to aid in buprenorphine initiation?
a) CIWA score.
b) PADUA score.
c) COWS score.
d) 4T score.
Explanation: COWS is the “clinical opiate withdrawal scale.” The COWS score should be calculated by a trained provider, and includes objective parameters (such as pulse) and subjective symptoms (such as GI upset, bone/joint aches.) It is recommended that agonist therapy be started when the COWS score is consistent with moderate withdrawal.
3. How can clinicians reliably find out if there are outpatient resources/clinics for patients with OUD in their area?
a) No way to find this out without personal knowledge.
b) Hospital providers and patients can visit www.samhsa.gov/find-help/national-helpline or call 1-800-662-HELP (4357) to find options for treatment for substance use disorders in their areas.
c) Dial “0” on any phone and ask.
d) Ask around at your hospital.
Explanation: The Substance Abuse and Mental Health Services Administration (SAMHSA) is an agency in the U.S. Department of Health and Human Services that is engaged in public health efforts to reduce the impact of substance abuse and mental illness on local communities. The agency’s website has helpful information about resources for substance use treatment.
4. Patients with OUD should be prescribed and given training about what medication that can be lifesaving when given during an opioid overdose?
a) Aspirin.
b) Naloxone.
c) Naltrexone.
d) Clonidine.
Explanation: Naloxone can be life-saving in the setting of an overdose. Best practice is to provide naloxone and training to patients with OUD.
5. When patients take buprenorphine soon after taking other opioids, there is concern for the development of which reaction:
a) Precipitated withdrawal.
b) Opioid overdose.
c) Allergic reaction.
d) Intoxication.
Explanation: Administering buprenorphine soon after taking other opioids can cause precipitated withdrawal, as buprenorphine binds with higher affinity to the mu receptor than many opioids. Precipitated withdrawal causes severe discomfort and can be dangerous for patients.
Case
A 35-year-old woman with opioid use disorder (OUD) presents with fever, left arm redness, and swelling. She is admitted to the hospital for cellulitis treatment. On the day after admission she becomes agitated and develops nausea, diarrhea, and generalized pain. Opioid withdrawal is suspected. How should her opioid use be addressed while in the hospital?
Brief overview of the issue
Since 1999, there have been more than 800,000 deaths related to drug overdose in the United States, and in 2019 more than 70% of drug overdose deaths involved an opioid.1,2 Although effective treatments for OUD exist, less than 20% of those with OUD are engaged in treatment.3
In America, 4%-11% of hospitalized patients have OUD. Hospitalized patients with OUD often experience stigma surrounding their disease, and many inpatient clinicians lack knowledge regarding the care of patients with OUD. As a result, withdrawal symptoms may go untreated, which can erode trust in the medical system and contribute to patients’ leaving the hospital before their primary medical issue is fully addressed. Therefore, it is essential that inpatient clinicians be familiar with the management of this complex and vulnerable patient population. Initiating treatment for OUD in the hospital setting is feasible and effective, and can lead to increased engagement in OUD treatment even after the hospital stay.
Overview of the data
Assessing patients with suspected OUD
Assessment for OUD starts with an in-depth opioid use history including frequency, amount, and method of administration. Clinicians should gather information regarding use of other substances or nonprescribed medications, and take thorough psychiatric and social histories. A formal diagnosis of OUD can be made using the Fifth Edition Diagnostic and Statistical Manual for Mental Disorders (DSM-5) diagnostic criteria.
Recognizing and managing opioid withdrawal
OUD in hospitalized patients often becomes apparent when patients develop signs and symptoms of withdrawal. Decreasing physical discomfort related to withdrawal can allow inpatient clinicians to address the condition for which the patient was hospitalized, help to strengthen the patient-clinician relationship, and provide an opportunity to discuss long-term OUD treatment.
Signs and symptoms of opioid withdrawal include anxiety, restlessness, irritability, generalized pain, rhinorrhea, yawning, lacrimation, piloerection, anorexia, and nausea. Withdrawal can last days to weeks, depending on the half-life of the opioid that was used. Opioids with shorter half-lives, such as heroin or oxycodone, cause withdrawal with earlier onset and shorter duration than do opioids with longer half-lives, such as methadone. The degree of withdrawal can be quantified with validated tools, such as the Clinical Opiate Withdrawal Scale (COWS).
Treatment of opioid withdrawal should generally include the use of an opioid agonist such as methadone or buprenorphine. A 2017 Cochrane meta-analysis found methadone or buprenorphine to be more effective than clonidine in alleviating symptoms of withdrawal and in retaining patients in treatment.4 Clonidine, an alpha2-adrenergic agonist that binds to receptors in the locus coeruleus, does not alleviate opioid cravings, but may be used as an adjunctive treatment for associated autonomic withdrawal symptoms. Other adjunctive medications include analgesics, antiemetics, antidiarrheals, and antihistamines.

Opioid agonist treatment for opioid use disorder
Opioid agonist treatment (OAT) with methadone or buprenorphine is associated with decreased mortality, opioid use, and infectious complications, but remains underutilized.5 Hospitalized patients with OUD are frequently managed with a rapid opioid detoxification and then discharged without continued OUD treatment. Detoxification alone can lead to a relapse rate as high as 90%.6 Patients are at increased risk for overdose after withdrawal due to loss of tolerance. Inpatient clinicians can close this OUD treatment gap by familiarizing themselves with OAT and offering to initiate OAT for maintenance treatment in interested patients. In one study, patients started on buprenorphine while hospitalized were more likely to be engaged in treatment and less likely to report drug use at follow-up, compared to patients who were referred without starting the medication.7
Buprenorphine
Buprenorphine is a partial agonist at the mu opioid receptor that can be ordered in the inpatient setting by any clinician. In the outpatient setting only DATA 2000 waivered clinicians can prescribe buprenorphine.8 Buprenorphine is most commonly coformulated with naloxone, an opioid antagonist, and is available in sublingual films or tablets. The naloxone component is not bioavailable when taken sublingually but becomes bioavailable if the drug is injected intravenously, leading to acute withdrawal.
Buprenorphine has a higher affinity for the mu opioid receptor than most opioids. If administered while other opioids are still present, it will displace the other opioid from the receptor but only partially stimulate the receptor, which can cause precipitated withdrawal. Buprenorphine initiation can start when the COWS score reflects moderate withdrawal. Many institutions use a threshold of 8-12 on the COWS scale. Typical dosing is 2-4 mg of buprenorphine at intervals of 1-2 hours as needed until the COWS score is less than 8, up to a maximum of 16 mg on day 1. The total dose from day 1 may be given as a daily dose beginning on day 2, up to a maximum total daily dose of 24 mg.
In recent years, a method of initiating buprenorphine called “micro-dosing” has gained traction. Very small doses of buprenorphine are given while a patient is receiving other opioids, thereby reducing the risk of precipitated withdrawal. This method can be helpful for patients who cannot tolerate withdrawal or who have recently taken long-acting opioids such as methadone. Such protocols should be utilized only at centers where consultation with an addiction specialist or experienced clinician is possible.
Despite evidence of buprenorphine’s efficacy, there are barriers to prescribing it. Physicians and advanced practitioners must be granted a waiver from the Drug Enforcement Administration to prescribe buprenorphine to outpatients. As of 2017, less than 10% of primary care physicians had obtained waivers.9 However, inpatient clinicians without a waiver can order buprenorphine and initiate treatment. Best practice is to do so with a specific plan for continuation at discharge. We encourage inpatient clinicians to obtain a waiver, so that a prescription can be given at discharge to bridge the patient to a first appointment with a community clinician who can continue treatment. As of April 27, 2021, providers treating fewer than 30 patients with OUD at one time may obtain a waiver without additional training.10
Methadone
Methadone is a full agonist at the mu opioid receptor. In the hospital setting, methadone can be ordered by any clinician to prevent and treat withdrawal. Commonly, doses of 10 mg can be given using the COWS score to guide the need for additional dosing. The patient can be reassessed every 1-2 hours to ensure that symptoms are improving, and that there is no sign of oversedation before giving additional methadone. For most patients, withdrawal can be managed with 20-40 mg of methadone daily.
In contrast to buprenorphine, methadone will not precipitate withdrawal and can be initiated even when patients are not yet showing withdrawal symptoms. Outpatient methadone treatment for OUD is federally regulated and can be delivered only in opioid treatment programs (OTPs).
Choosing methadone or buprenorphine in the inpatient setting
The choice between buprenorphine and methadone should take into consideration several factors, including patient preference, treatment history, and available outpatient treatment programs, which may vary widely by geographic region. Some patients benefit from the higher level of support and counseling available at OTPs. Methadone is available at all OTPs, and the availability of buprenorphine in this setting is increasing. Other patients may prefer the convenience and flexibility of buprenorphine treatment in an outpatient office setting.
Some patients have prior negative experiences with OAT. These can include prior precipitated withdrawal with buprenorphine induction, or negative experiences with the structure of OTPs. Clinicians are encouraged to provide counseling if patients have a history of precipitated withdrawal to assure them that this can be avoided with proper dosing. Clinicians should be familiar with available treatment options in their community and can refer to the Substance Abuse and Mental Health Services Administration (SAMHSA) website to locate OTPs and buprenorphine prescribers.

Polypharmacy and safety
If combined with benzodiazepines, alcohol, or other sedating agents, methadone or buprenorphine can increase risk of overdose. However, OUD treatment should not be withheld because of other substance use. Clinicians initiating treatment should counsel patients on the risk of concomitant substance use and provide overdose prevention education.
A brief note on naltrexone
Naltrexone, an opioid antagonist, is used more commonly in outpatient addiction treatment than in the inpatient setting, but inpatient clinicians should be aware of its use. It is available in oral and long-acting injectable formulations. Its utility in the inpatient setting may be limited as safe administration requires 7-10 days of opioid abstinence.
Discharge planning
Patients with OUD or who are started on OAT during a hospitalization should be linked to continued outpatient treatment. Before discharge it is best to ensure vaccinations for HAV, HBV, pneumococcus, and tetanus are up to date, and perform screening for HIV, hepatitis C, tuberculosis, and sexually transmitted infections if appropriate. All patients with OUD should be prescribed or provided with take-home naloxone for overdose reversal. Patients can also be referred to syringe service programs for additional harm reduction counseling and services.
Application of the data to our patient
For our patient, either methadone or buprenorphine could be used to treat her withdrawal. The COWS score should be used to assess withdrawal severity, and to guide appropriate timing of medication initiation. If she wishes to continue OAT after discharge, she should be linked to a clinician who can engage her in ongoing medical care. Prior to discharge she should also receive relevant vaccines and screening for infectious diseases as outlined above, as well as take-home naloxone (or a prescription).
Bottom line
Inpatient clinicians can play a pivotal role in patients’ lives by ensuring that patients with OUD receive OAT and are connected to outpatient care at discharge.
Dr. Linker is assistant professor in the division of hospital medicine, Icahn School of Medicine at Mount Sinai, New York. Ms. Hirt, Mr. Fine, and Mr. Villasanivis are medical students at the Icahn School of Medicine at Mount Sinai. Dr. Wang is assistant professor in the division of general internal medicine, Icahn School of Medicine at Mount Sinai. Dr. Herscher is assistant professor in the division of hospital medicine, Icahn School of Medicine at Mount Sinai.
References
1. Wide-ranging online data for epidemiologic research (WONDER). Atlanta, GA: CDC, National Center for Health Statistics; 2020. Available at http://wonder.cdc.gov.
2. Mattson CL et al. Trends and geographic patterns in drug and synthetic opioid overdose deaths – United States, 2013-2019. MMWR Morb Mortal Wkly Rep. 2021;70:202-7. doi: 10.15585/mmwr.mm7006a4.
3. Wakeman SE et al. Comparative effectiveness of different treatment pathways for opioid use disorder. JAMA Netw Open. 2020 Feb 5;3(2):e1920622. doi: 10.1001/jamanetworkopen.2019.20622.
4. Gowing L et al. Buprenorphine for managing opioid withdrawal. Cochrane Database Syst Rev. 2017 Feb;2017(2):CD002025. doi: 10.1002/14651858.CD002025.pub5.
5. Sordo L et al. Mortality risk during and after opioid substitution treatment: Systematic review and meta-analysis of cohort studies. BMJ. 2017 Apr 26;357:j1550. doi: 10.1136/bmj.j1550.
6. Smyth BP et al. Lapse and relapse following inpatient treatment of opiate dependence. Ir Med J. 2010 Jun;103(6):176-9. Available at www.drugsandalcohol.ie/13405.
7. Liebschutz JM. Buprenorphine treatment for hospitalized, opioid-dependent patients: A randomized clinical trial. JAMA Intern Med. 2014 Aug;174(8):1369-76. doi: 10.1001/jamainternmed.2014.2556.
8. Substance Abuse and Mental Health Services Administration. (Aug 20, 2020) Statutes, Regulations, and Guidelines.
9. McBain RK et al. Growth and distribution of buprenorphine-waivered providers in the United States, 2007-2017. Ann Intern Med. 2020;172(7):504-6. doi: 10.7326/M19-2403.
10. HHS releases new buprenorphine practice guidelines, expanding access to treatment for opioid use disorder. Apr 27, 2021.
11. Herscher M et al. Diagnosis and management of opioid use disorder in hospitalized patients. Med Clin North Am. 2020 Jul;104(4):695-708. doi: 10.1016/j.mcna.2020.03.003.
Additional reading
Winetsky D. Expanding treatment opportunities for hospitalized patients with opioid use disorders. J Hosp Med. 2018 Jan;13(1):62-4. doi: 10.12788/jhm.2861.
Donroe JH. Caring for patients with opioid use disorder in the hospital. Can Med Assoc J. 2016 Dec 6;188(17-18):1232-9. doi: 10.1503/cmaj.160290.
Herscher M et al. Diagnosis and management of opioid use disorder in hospitalized patients. Med Clin North Am. 2020 Jul;104(4):695-708. doi: 10.1016/j.mcna.2020.03.003.
Key points
- Most patients with OUD are not engaged in evidence-based treatment. Clinicians have an opportunity to utilize the inpatient stay as a ‘reachable moment’ to engage patients with OUD in evidence-based treatment.
- Buprenorphine and methadone are effective opioid agonist medications used to treat OUD, and clinicians with the appropriate knowledge base can initiate either during the inpatient encounter, and link the patient to OUD treatment after the hospital stay.
Quiz
Caring for hospitalized patients with OUD
Most patients with OUD are not engaged in effective treatment. Hospitalization can be a ‘reachable moment’ to engage patients with OUD in evidence-based treatment.
1. Which is an effective and evidence-based medication for treating opioid withdrawal and OUD?
a) Naltrexone.
b) Buprenorphine.
c) Opioid detoxification.
d) Clonidine.
Explanation: Buprenorphine is effective for alleviating symptoms of withdrawal as well as for the long-term treatment of OUD. While naltrexone is also used to treat OUD, it is not useful for treating withdrawal. Clonidine can be a useful adjunctive medication for treating withdrawal but is not a long-term treatment for OUD. Nonpharmacologic detoxification is not an effective treatment for OUD and is associated with high relapse rates.
2. What scale can be used during a hospital stay to monitor patients with OUD at risk of opioid withdrawal, and to aid in buprenorphine initiation?
a) CIWA score.
b) PADUA score.
c) COWS score.
d) 4T score.
Explanation: COWS is the “clinical opiate withdrawal scale.” The COWS score should be calculated by a trained provider, and includes objective parameters (such as pulse) and subjective symptoms (such as GI upset, bone/joint aches.) It is recommended that agonist therapy be started when the COWS score is consistent with moderate withdrawal.
3. How can clinicians reliably find out if there are outpatient resources/clinics for patients with OUD in their area?
a) No way to find this out without personal knowledge.
b) Hospital providers and patients can visit www.samhsa.gov/find-help/national-helpline or call 1-800-662-HELP (4357) to find options for treatment for substance use disorders in their areas.
c) Dial “0” on any phone and ask.
d) Ask around at your hospital.
Explanation: The Substance Abuse and Mental Health Services Administration (SAMHSA) is an agency in the U.S. Department of Health and Human Services that is engaged in public health efforts to reduce the impact of substance abuse and mental illness on local communities. The agency’s website has helpful information about resources for substance use treatment.
4. Patients with OUD should be prescribed and given training about what medication that can be lifesaving when given during an opioid overdose?
a) Aspirin.
b) Naloxone.
c) Naltrexone.
d) Clonidine.
Explanation: Naloxone can be life-saving in the setting of an overdose. Best practice is to provide naloxone and training to patients with OUD.
5. When patients take buprenorphine soon after taking other opioids, there is concern for the development of which reaction:
a) Precipitated withdrawal.
b) Opioid overdose.
c) Allergic reaction.
d) Intoxication.
Explanation: Administering buprenorphine soon after taking other opioids can cause precipitated withdrawal, as buprenorphine binds with higher affinity to the mu receptor than many opioids. Precipitated withdrawal causes severe discomfort and can be dangerous for patients.
Yoga effective adjunct therapy in recurrent vasovagal syncope
Yoga added to conventional therapy for vasovagal syncope (VVS), when patients faint after a sudden drop in heart rate and blood pressure, can reduce symptoms and improve quality of life, new research suggests.
A small, open-label trial conducted in New Delhi showed that participants practicing yoga reported an improvement in VVS symptoms after only 6 weeks, with a reduction of 1.82 events at 12 months. All those practicing yoga also showed significantly improved quality of life (QoL) scores by the end of the trial.
“Yoga as add-on therapy in VVS is superior to medical therapy in reducing syncopal and presyncopal events and in improving the QoL,” report Gautam Sharma, MD, DM, Centre for Integrative Medicine and Research, All India Institute of Medical Sciences, New Delhi, and colleagues. “It may be useful to integrate a cost-effective and safe intervention such as yoga into the management of VVS.”
Results of the LIVE-Yoga study were published online in JACC: Clinical Electrophysiology.
Vasovagal syncope is a common and non–life-threatening condition, but given the severity and frequency of recurrence it can result in significant deterioration in a patient’s quality of life, the authors note. “Existing management therapies have been largely ineffective,” they write.
Recent trials have suggested some efficacy for yoga in diseases of autonomic imbalance, suggesting a possible use in VVS. To find out, the researchers enrolled adults with VVS between the ages of 15-70 years who had a positive head-up tilt test (HUTT) and at least two syncope or presyncope events within 3 months of enrollment. They also needed to be willing and able to practice yoga. Those with structural heart disease, accelerated hypertension, and underlying neurologic disorders were not included in the study.
A total of 55 patients were randomly assigned to receive either a specialized yoga training program in addition to guideline-based therapy, or guideline-based therapy alone. Standard care included physical counterpressure maneuvers, avoidance of known triggers, increased salt and water intake, and drug therapy or pacing at the discretion of the treating physician.
The primary outcome was a composite of the number of episodes of syncope and presyncope at 12 months.
Secondary outcomes including QoL, assessed using the World Health Organization Quality of Life Brief Field questionnaire (WHOQoL-BREF) and the Syncope Functional Status Questionnaire (SFSQ) at 12 months, a head-up tilt test, and heart rate variability at 6 weeks.
For the first 2 weeks, patients in the intervention group were enrolled in eight supervised yoga sessions conducted at the Centre for Integrative Medicine and Research at the All India Institute of Medical Sciences. For the remainder of the trial, they continued a daily yoga practice at home at least 5 days a week.
The yoga module created for participants was designed with a view to the pathophysiology of VVS and featured postures, breathing, and relaxation techniques. Yoga classes were taught by qualified therapists under the guidance of physicians.
In addition to a booklet with a pictorial of the yoga regimen, participants received twice-monthly calls from the yoga center to encourage compliance. Results show that all participants adhered to their yoga routine for more than 80% of the 12-month trial.
At 12 months, the mean number of syncopal or presyncopal events was 0.7 ± 0.7 with the yoga intervention versus 2.52 ± 1.93 among patients in the control arm (P < .05). The reduction in events started as early as 6 weeks and continued to separate out to 12 months, the researchers note.
Thirteen of 30 (43.3%) intervention patients and 4 of 25 (16%) control patients remained event-free at 12 months, a statistically significant difference (P = .02). There was a trend toward fewer positive head-up tilt tests between groups that did not reach significance, and there was no difference in heart rate variability at 6 weeks.
No adverse events as a result of the yoga practice were reported, and no patient started drug therapy or received pacing therapy during the trial, they note.
The researchers point out that yoga postures can enhance vascular and muscle tone, especially in the lower limbs.
“Yoga breathing and relaxation techniques have been shown to increase vagal tone and improve autonomic balance, which could potentially curtail the sympathetic overdrive phase and interrupt the activation of the c-mechanoreceptors, which is a critical step in the syncope cascade,” they note.
“We postulate that positive effects of yoga in this study could be related to a multidimensional effect of this intervention acting through both central and peripheral mechanisms, including physical, psychological, and autonomic pathways,” the authors conclude.
Comprehensive regimen
Dhanunjaya Lakkireddy, MD, medical director for the Kansas City Heart Rhythm Institute, Overland Park, Kansas, says these results are in line with previous research indicating the benefits of yoga in improving cardiovascular function.
“All of this clearly shows that when you [include] a systematic diet of yoga for a reasonable amount of time to improve the plasticity of parasympathetic inputs into the chest and thereby the cardiovascular system ... you can help patients to improve their symptoms,” he said in an interview.
He already prescribes yoga in his own practice as part of a comprehensive therapeutic regimen, he said. “We have a handful of practitioners all around the city who work with us,” Dr. Lakkireddy said.
Both he and the study authors point the economic burden of VVS both in management and in loss of patient productivity. “A low-cost intervention in the form of yoga, which essentially requires only a mat, can reduce both direct and indirect costs significantly,” note the authors.
The trial was supported under the extramural research (EMR) scheme by the Ministry of AYUSH, Government of India. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Yoga added to conventional therapy for vasovagal syncope (VVS), when patients faint after a sudden drop in heart rate and blood pressure, can reduce symptoms and improve quality of life, new research suggests.
A small, open-label trial conducted in New Delhi showed that participants practicing yoga reported an improvement in VVS symptoms after only 6 weeks, with a reduction of 1.82 events at 12 months. All those practicing yoga also showed significantly improved quality of life (QoL) scores by the end of the trial.
“Yoga as add-on therapy in VVS is superior to medical therapy in reducing syncopal and presyncopal events and in improving the QoL,” report Gautam Sharma, MD, DM, Centre for Integrative Medicine and Research, All India Institute of Medical Sciences, New Delhi, and colleagues. “It may be useful to integrate a cost-effective and safe intervention such as yoga into the management of VVS.”
Results of the LIVE-Yoga study were published online in JACC: Clinical Electrophysiology.
Vasovagal syncope is a common and non–life-threatening condition, but given the severity and frequency of recurrence it can result in significant deterioration in a patient’s quality of life, the authors note. “Existing management therapies have been largely ineffective,” they write.
Recent trials have suggested some efficacy for yoga in diseases of autonomic imbalance, suggesting a possible use in VVS. To find out, the researchers enrolled adults with VVS between the ages of 15-70 years who had a positive head-up tilt test (HUTT) and at least two syncope or presyncope events within 3 months of enrollment. They also needed to be willing and able to practice yoga. Those with structural heart disease, accelerated hypertension, and underlying neurologic disorders were not included in the study.
A total of 55 patients were randomly assigned to receive either a specialized yoga training program in addition to guideline-based therapy, or guideline-based therapy alone. Standard care included physical counterpressure maneuvers, avoidance of known triggers, increased salt and water intake, and drug therapy or pacing at the discretion of the treating physician.
The primary outcome was a composite of the number of episodes of syncope and presyncope at 12 months.
Secondary outcomes including QoL, assessed using the World Health Organization Quality of Life Brief Field questionnaire (WHOQoL-BREF) and the Syncope Functional Status Questionnaire (SFSQ) at 12 months, a head-up tilt test, and heart rate variability at 6 weeks.
For the first 2 weeks, patients in the intervention group were enrolled in eight supervised yoga sessions conducted at the Centre for Integrative Medicine and Research at the All India Institute of Medical Sciences. For the remainder of the trial, they continued a daily yoga practice at home at least 5 days a week.
The yoga module created for participants was designed with a view to the pathophysiology of VVS and featured postures, breathing, and relaxation techniques. Yoga classes were taught by qualified therapists under the guidance of physicians.
In addition to a booklet with a pictorial of the yoga regimen, participants received twice-monthly calls from the yoga center to encourage compliance. Results show that all participants adhered to their yoga routine for more than 80% of the 12-month trial.
At 12 months, the mean number of syncopal or presyncopal events was 0.7 ± 0.7 with the yoga intervention versus 2.52 ± 1.93 among patients in the control arm (P < .05). The reduction in events started as early as 6 weeks and continued to separate out to 12 months, the researchers note.
Thirteen of 30 (43.3%) intervention patients and 4 of 25 (16%) control patients remained event-free at 12 months, a statistically significant difference (P = .02). There was a trend toward fewer positive head-up tilt tests between groups that did not reach significance, and there was no difference in heart rate variability at 6 weeks.
No adverse events as a result of the yoga practice were reported, and no patient started drug therapy or received pacing therapy during the trial, they note.
The researchers point out that yoga postures can enhance vascular and muscle tone, especially in the lower limbs.
“Yoga breathing and relaxation techniques have been shown to increase vagal tone and improve autonomic balance, which could potentially curtail the sympathetic overdrive phase and interrupt the activation of the c-mechanoreceptors, which is a critical step in the syncope cascade,” they note.
“We postulate that positive effects of yoga in this study could be related to a multidimensional effect of this intervention acting through both central and peripheral mechanisms, including physical, psychological, and autonomic pathways,” the authors conclude.
Comprehensive regimen
Dhanunjaya Lakkireddy, MD, medical director for the Kansas City Heart Rhythm Institute, Overland Park, Kansas, says these results are in line with previous research indicating the benefits of yoga in improving cardiovascular function.
“All of this clearly shows that when you [include] a systematic diet of yoga for a reasonable amount of time to improve the plasticity of parasympathetic inputs into the chest and thereby the cardiovascular system ... you can help patients to improve their symptoms,” he said in an interview.
He already prescribes yoga in his own practice as part of a comprehensive therapeutic regimen, he said. “We have a handful of practitioners all around the city who work with us,” Dr. Lakkireddy said.
Both he and the study authors point the economic burden of VVS both in management and in loss of patient productivity. “A low-cost intervention in the form of yoga, which essentially requires only a mat, can reduce both direct and indirect costs significantly,” note the authors.
The trial was supported under the extramural research (EMR) scheme by the Ministry of AYUSH, Government of India. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Yoga added to conventional therapy for vasovagal syncope (VVS), when patients faint after a sudden drop in heart rate and blood pressure, can reduce symptoms and improve quality of life, new research suggests.
A small, open-label trial conducted in New Delhi showed that participants practicing yoga reported an improvement in VVS symptoms after only 6 weeks, with a reduction of 1.82 events at 12 months. All those practicing yoga also showed significantly improved quality of life (QoL) scores by the end of the trial.
“Yoga as add-on therapy in VVS is superior to medical therapy in reducing syncopal and presyncopal events and in improving the QoL,” report Gautam Sharma, MD, DM, Centre for Integrative Medicine and Research, All India Institute of Medical Sciences, New Delhi, and colleagues. “It may be useful to integrate a cost-effective and safe intervention such as yoga into the management of VVS.”
Results of the LIVE-Yoga study were published online in JACC: Clinical Electrophysiology.
Vasovagal syncope is a common and non–life-threatening condition, but given the severity and frequency of recurrence it can result in significant deterioration in a patient’s quality of life, the authors note. “Existing management therapies have been largely ineffective,” they write.
Recent trials have suggested some efficacy for yoga in diseases of autonomic imbalance, suggesting a possible use in VVS. To find out, the researchers enrolled adults with VVS between the ages of 15-70 years who had a positive head-up tilt test (HUTT) and at least two syncope or presyncope events within 3 months of enrollment. They also needed to be willing and able to practice yoga. Those with structural heart disease, accelerated hypertension, and underlying neurologic disorders were not included in the study.
A total of 55 patients were randomly assigned to receive either a specialized yoga training program in addition to guideline-based therapy, or guideline-based therapy alone. Standard care included physical counterpressure maneuvers, avoidance of known triggers, increased salt and water intake, and drug therapy or pacing at the discretion of the treating physician.
The primary outcome was a composite of the number of episodes of syncope and presyncope at 12 months.
Secondary outcomes including QoL, assessed using the World Health Organization Quality of Life Brief Field questionnaire (WHOQoL-BREF) and the Syncope Functional Status Questionnaire (SFSQ) at 12 months, a head-up tilt test, and heart rate variability at 6 weeks.
For the first 2 weeks, patients in the intervention group were enrolled in eight supervised yoga sessions conducted at the Centre for Integrative Medicine and Research at the All India Institute of Medical Sciences. For the remainder of the trial, they continued a daily yoga practice at home at least 5 days a week.
The yoga module created for participants was designed with a view to the pathophysiology of VVS and featured postures, breathing, and relaxation techniques. Yoga classes were taught by qualified therapists under the guidance of physicians.
In addition to a booklet with a pictorial of the yoga regimen, participants received twice-monthly calls from the yoga center to encourage compliance. Results show that all participants adhered to their yoga routine for more than 80% of the 12-month trial.
At 12 months, the mean number of syncopal or presyncopal events was 0.7 ± 0.7 with the yoga intervention versus 2.52 ± 1.93 among patients in the control arm (P < .05). The reduction in events started as early as 6 weeks and continued to separate out to 12 months, the researchers note.
Thirteen of 30 (43.3%) intervention patients and 4 of 25 (16%) control patients remained event-free at 12 months, a statistically significant difference (P = .02). There was a trend toward fewer positive head-up tilt tests between groups that did not reach significance, and there was no difference in heart rate variability at 6 weeks.
No adverse events as a result of the yoga practice were reported, and no patient started drug therapy or received pacing therapy during the trial, they note.
The researchers point out that yoga postures can enhance vascular and muscle tone, especially in the lower limbs.
“Yoga breathing and relaxation techniques have been shown to increase vagal tone and improve autonomic balance, which could potentially curtail the sympathetic overdrive phase and interrupt the activation of the c-mechanoreceptors, which is a critical step in the syncope cascade,” they note.
“We postulate that positive effects of yoga in this study could be related to a multidimensional effect of this intervention acting through both central and peripheral mechanisms, including physical, psychological, and autonomic pathways,” the authors conclude.
Comprehensive regimen
Dhanunjaya Lakkireddy, MD, medical director for the Kansas City Heart Rhythm Institute, Overland Park, Kansas, says these results are in line with previous research indicating the benefits of yoga in improving cardiovascular function.
“All of this clearly shows that when you [include] a systematic diet of yoga for a reasonable amount of time to improve the plasticity of parasympathetic inputs into the chest and thereby the cardiovascular system ... you can help patients to improve their symptoms,” he said in an interview.
He already prescribes yoga in his own practice as part of a comprehensive therapeutic regimen, he said. “We have a handful of practitioners all around the city who work with us,” Dr. Lakkireddy said.
Both he and the study authors point the economic burden of VVS both in management and in loss of patient productivity. “A low-cost intervention in the form of yoga, which essentially requires only a mat, can reduce both direct and indirect costs significantly,” note the authors.
The trial was supported under the extramural research (EMR) scheme by the Ministry of AYUSH, Government of India. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New insights into psychogenic seizures in teens
, results of a small study suggest.
The school experience of teens with PNES is overwhelmingly negative, study investigator Andrea Tanner, PhD, a postdoctoral fellow at Indiana University School of Nursing, Indianapolis.
She hopes this research will spur a collaborative effort between students, schools, families, and health care providers “to develop an effective plan to help these adolescents cope, to manage this condition, and hopefully reach seizure freedom.”
The findings were presented at the annual meeting of the American Epilepsy Society.
Anxiety, perfectionism
Although psychogenic seizures resemble epileptic seizures, they have a psychological basis and, unlike epilepsy, are not caused by abnormal electrical brain activity.
While the school experience has previously been identified as a source of predisposing, precipitating, and perpetuating factors for PNES, little is known about the school experience of adolescents with the disorder and the role it may play in PNES management, the investigators noted.
During her 20 years as a school nurse, Dr. Tanner saw firsthand how school staff struggled with responding appropriately to teens with PNES. “They wanted to call 911 every time; they wanted to respond as if it [were] an epileptic seizure.”
For the study, she interviewed 10 teens with PNES, aged 12 to 19 years, whom she found mostly through Facebook support groups but also through flyers. All participants had undergone video EEG and been diagnosed with PNES.
From the interviews, Dr. Tanner and colleagues conducted a qualitative content analysis and uncovered “overarching” themes.
A main theme was stress, some of which focused on bullying by peers or harassment by school personnel, much of which was related to accusations of the children “faking” seizures to get attention, said Dr. Tanner.
Some teens reported being banned from school events, such as field trips, out of concern they would be a “distraction,” which led to feelings of isolation and exclusion, said Dr. Tanner.
Research points to a growing incidence of PNES among adolescents. This may be because it is now better recognized, or it may stem from the unique stressors today’s teens face, said Dr. Tanner.
Adolescents discussed the pressures they feel to be the best at everything. “They wanted to be good in athletics; they wanted to be good in academics; they wanted to get into a good college,” said Dr. Tanner.
Some study participants had undergone psychotherapy, including cognitive-behavioral therapy, and others had investigated mindfulness-based therapy. However, not all were receiving treatment. For some, such care was inaccessible, while others had tried a mental health care intervention but had abandoned it.
Although all the study participants were female, Dr. Tanner has interviewed males outside this study and found their experiences are similar.
Her next research step is to try to quantify the findings. “I would like to begin to look at what would be the appropriate outcomes if I were to do an intervention to improve the school experience.”
Her message for doctors is to see school nurses as a “partner” or “liaison” who “can bridge the world of health care and education.”
Important, novel research
Commenting on the research, Barbara Dworetzky, MD, Chief, Epilepsy, Brigham and Women’s Hospital, and professor of neurology, Harvard Medical School, said it’s “important and novel.”
The study focuses on the main factors – or themes – that lead to increased stress, such as bullying, isolation, and “not being believed,” that are likely triggers for PNES, said Dr. Dworetzky.
The study is also important because it focuses on factors that help make the girls “feel supported and protected” – for example, having staff “take the episodes seriously,” she said.
The study’s qualitative measures “are a valid way of understanding these girls and giving them a voice,” said Dr. Dworetzky. She added the study provides “practical information” that could help target treatments to improve outcomes in this group.
A limitation of the study was that the very small cohort of teenage girls was selected only through families in Facebook support groups or flyers to school nurses, said Dr. Dworetzky.
“There are likely many other groups who don’t even have families trying to help them. Larger cohorts without this type of bias may be next steps.”
A version of this article first appeared on Medscape.com.
, results of a small study suggest.
The school experience of teens with PNES is overwhelmingly negative, study investigator Andrea Tanner, PhD, a postdoctoral fellow at Indiana University School of Nursing, Indianapolis.
She hopes this research will spur a collaborative effort between students, schools, families, and health care providers “to develop an effective plan to help these adolescents cope, to manage this condition, and hopefully reach seizure freedom.”
The findings were presented at the annual meeting of the American Epilepsy Society.
Anxiety, perfectionism
Although psychogenic seizures resemble epileptic seizures, they have a psychological basis and, unlike epilepsy, are not caused by abnormal electrical brain activity.
While the school experience has previously been identified as a source of predisposing, precipitating, and perpetuating factors for PNES, little is known about the school experience of adolescents with the disorder and the role it may play in PNES management, the investigators noted.
During her 20 years as a school nurse, Dr. Tanner saw firsthand how school staff struggled with responding appropriately to teens with PNES. “They wanted to call 911 every time; they wanted to respond as if it [were] an epileptic seizure.”
For the study, she interviewed 10 teens with PNES, aged 12 to 19 years, whom she found mostly through Facebook support groups but also through flyers. All participants had undergone video EEG and been diagnosed with PNES.
From the interviews, Dr. Tanner and colleagues conducted a qualitative content analysis and uncovered “overarching” themes.
A main theme was stress, some of which focused on bullying by peers or harassment by school personnel, much of which was related to accusations of the children “faking” seizures to get attention, said Dr. Tanner.
Some teens reported being banned from school events, such as field trips, out of concern they would be a “distraction,” which led to feelings of isolation and exclusion, said Dr. Tanner.
Research points to a growing incidence of PNES among adolescents. This may be because it is now better recognized, or it may stem from the unique stressors today’s teens face, said Dr. Tanner.
Adolescents discussed the pressures they feel to be the best at everything. “They wanted to be good in athletics; they wanted to be good in academics; they wanted to get into a good college,” said Dr. Tanner.
Some study participants had undergone psychotherapy, including cognitive-behavioral therapy, and others had investigated mindfulness-based therapy. However, not all were receiving treatment. For some, such care was inaccessible, while others had tried a mental health care intervention but had abandoned it.
Although all the study participants were female, Dr. Tanner has interviewed males outside this study and found their experiences are similar.
Her next research step is to try to quantify the findings. “I would like to begin to look at what would be the appropriate outcomes if I were to do an intervention to improve the school experience.”
Her message for doctors is to see school nurses as a “partner” or “liaison” who “can bridge the world of health care and education.”
Important, novel research
Commenting on the research, Barbara Dworetzky, MD, Chief, Epilepsy, Brigham and Women’s Hospital, and professor of neurology, Harvard Medical School, said it’s “important and novel.”
The study focuses on the main factors – or themes – that lead to increased stress, such as bullying, isolation, and “not being believed,” that are likely triggers for PNES, said Dr. Dworetzky.
The study is also important because it focuses on factors that help make the girls “feel supported and protected” – for example, having staff “take the episodes seriously,” she said.
The study’s qualitative measures “are a valid way of understanding these girls and giving them a voice,” said Dr. Dworetzky. She added the study provides “practical information” that could help target treatments to improve outcomes in this group.
A limitation of the study was that the very small cohort of teenage girls was selected only through families in Facebook support groups or flyers to school nurses, said Dr. Dworetzky.
“There are likely many other groups who don’t even have families trying to help them. Larger cohorts without this type of bias may be next steps.”
A version of this article first appeared on Medscape.com.
, results of a small study suggest.
The school experience of teens with PNES is overwhelmingly negative, study investigator Andrea Tanner, PhD, a postdoctoral fellow at Indiana University School of Nursing, Indianapolis.
She hopes this research will spur a collaborative effort between students, schools, families, and health care providers “to develop an effective plan to help these adolescents cope, to manage this condition, and hopefully reach seizure freedom.”
The findings were presented at the annual meeting of the American Epilepsy Society.
Anxiety, perfectionism
Although psychogenic seizures resemble epileptic seizures, they have a psychological basis and, unlike epilepsy, are not caused by abnormal electrical brain activity.
While the school experience has previously been identified as a source of predisposing, precipitating, and perpetuating factors for PNES, little is known about the school experience of adolescents with the disorder and the role it may play in PNES management, the investigators noted.
During her 20 years as a school nurse, Dr. Tanner saw firsthand how school staff struggled with responding appropriately to teens with PNES. “They wanted to call 911 every time; they wanted to respond as if it [were] an epileptic seizure.”
For the study, she interviewed 10 teens with PNES, aged 12 to 19 years, whom she found mostly through Facebook support groups but also through flyers. All participants had undergone video EEG and been diagnosed with PNES.
From the interviews, Dr. Tanner and colleagues conducted a qualitative content analysis and uncovered “overarching” themes.
A main theme was stress, some of which focused on bullying by peers or harassment by school personnel, much of which was related to accusations of the children “faking” seizures to get attention, said Dr. Tanner.
Some teens reported being banned from school events, such as field trips, out of concern they would be a “distraction,” which led to feelings of isolation and exclusion, said Dr. Tanner.
Research points to a growing incidence of PNES among adolescents. This may be because it is now better recognized, or it may stem from the unique stressors today’s teens face, said Dr. Tanner.
Adolescents discussed the pressures they feel to be the best at everything. “They wanted to be good in athletics; they wanted to be good in academics; they wanted to get into a good college,” said Dr. Tanner.
Some study participants had undergone psychotherapy, including cognitive-behavioral therapy, and others had investigated mindfulness-based therapy. However, not all were receiving treatment. For some, such care was inaccessible, while others had tried a mental health care intervention but had abandoned it.
Although all the study participants were female, Dr. Tanner has interviewed males outside this study and found their experiences are similar.
Her next research step is to try to quantify the findings. “I would like to begin to look at what would be the appropriate outcomes if I were to do an intervention to improve the school experience.”
Her message for doctors is to see school nurses as a “partner” or “liaison” who “can bridge the world of health care and education.”
Important, novel research
Commenting on the research, Barbara Dworetzky, MD, Chief, Epilepsy, Brigham and Women’s Hospital, and professor of neurology, Harvard Medical School, said it’s “important and novel.”
The study focuses on the main factors – or themes – that lead to increased stress, such as bullying, isolation, and “not being believed,” that are likely triggers for PNES, said Dr. Dworetzky.
The study is also important because it focuses on factors that help make the girls “feel supported and protected” – for example, having staff “take the episodes seriously,” she said.
The study’s qualitative measures “are a valid way of understanding these girls and giving them a voice,” said Dr. Dworetzky. She added the study provides “practical information” that could help target treatments to improve outcomes in this group.
A limitation of the study was that the very small cohort of teenage girls was selected only through families in Facebook support groups or flyers to school nurses, said Dr. Dworetzky.
“There are likely many other groups who don’t even have families trying to help them. Larger cohorts without this type of bias may be next steps.”
A version of this article first appeared on Medscape.com.
From AES 2021
‘Alarming’ rate of abuse in pregnant women with epilepsy
, new research shows.
Study investigator Naveed Chaudhry, MD, a recent epilepsy fellow and assistant professor of neurology, University of Colorado School of Medicine, described the finding as “alarming” and called for more support for this patient population.
Investigators found that women with epilepsy are also more likely to report other stressors, including divorce, illness, lost pay, and partner discord, while expecting.
“As epilepsy physicians, it’s important that we ask the right questions and dive a little bit deeper with these patients, even if it’s uncomfortable and not something we’re used to,” said Dr. Chaudhry.
The findings were presented at the annual meeting of the American Epilepsy Society.
Cause for concern
Women with epilepsy may be under stress for a variety of social and economic reasons. In some women, stress can trigger seizures, and during pregnancy, this can lead to complications such as preterm labor and low birth weight.
For the study, researchers tapped into the Center for Disease Control and Prevention Pregnancy Risk Assessment and Monitoring System (PRAMS). This database includes information from surveys asking women across the U.S. about their pregnancy and postpartum period.
Thirteen states collected data on stresses in women with and without epilepsy. Respondents were asked about 14 economic and other worries in the year prior to their baby’s birth, including the pregnancy period.
The analysis included 64,951 women, 1,140 of whom had epilepsy, who were included in surveys from 2012-2020. There were no significant demographic differences between those with and those without the disorder.
After adjusting for maternal age, race, ethnicity, marital status, education, and socioeconomic status, the study found that women with epilepsy experienced an average of 2.41 of the stressors compared with 1.72 for women without epilepsy.
Women with epilepsy were more likely to have experienced family illness, divorce, homelessness, partner job loss, reduced work or pay, increased arguments, having a partner in jail, drug use, and the death of someone close to them.
The results showed that unmarried and younger women as well as those with lower incomes were particularly prone to experience stress during pregnancy.
It’s not clear why women with epilepsy report more stressors. “Looking at the literature, no one has really looked at the exact reason for this, but we postulate it could be a lack of supports and support systems,” said Dr. Chaudhry.
Women were asked about physical, sexual, and emotional abuse. Results showed that substantially more women with epilepsy than those without the disorder reported such abuse during pregnancy – 10.6% versus 4.1%. The adjusted odds ratio for women with epilepsy reporting abuse was 2.78 (95% CI, 2.07-3.74).
“That raises our concern and needs to be looked at in more detail,” said Dr. Chaudhry.
It is unclear whether some women might have had psychogenic non-epileptic seizures (PNES), which are linked to a higher rate of abuse, said Dr. Chaudhry. “But the prevalence of PNES in the general population is quite low, so we don’t think it’s contributing to a large extent to this finding.”
The findings highlight the importance of addressing stress in women with epilepsy during pregnancy, he said. “We need to have good support services and we need to counsel women to optimize good outcomes.”
This applies to all women of childbearing age. “We suspect abuse and stressors are going to be going on throughout that period,” said Dr. Chaudhry. “It’s important to ask about it and have appropriate support staff and social work and people available to help when an issue is identified.”
Stress a common seizure trigger
Commenting on the research, Kimford Meador, MD, professor, Department of Neurology and Neurological Sciences, Stanford University School of Medicine, noted the study was well conducted and had a large sample size.
The findings are important, as stress is a common trigger for seizures in people with epilepsy and is associated with mood and anxiety, which can affect quality of life, said Dr. Meador.
Results of his analysis from the Maternal Outcomes and Neurodevelopmental Effects of Antiepileptic Drugs (MONEAD) study, also presented at this year’s AES meeting, showed that women with epilepsy had more depressive symptoms during the postpartum period and more anxiety symptoms during pregnancy and postpartum in comparison with those without epilepsy.
Dr. Meador’s group also recently conducted a study that was published in JAMA Neurology, showing that in women with epilepsy during the postpartum period, anxiety is associated with lower cognitive ability in their children at age 2 years.
“All these findings highlight the importance of assessing and managing stress, anxiety, and mood in women with epilepsy,” said Dr. Meador. “Interventions could impact seizures and quality of life in pregnant women with epilepsy and long-term outcomes in their children.”
A version of this article first appeared on Medscape.com.
, new research shows.
Study investigator Naveed Chaudhry, MD, a recent epilepsy fellow and assistant professor of neurology, University of Colorado School of Medicine, described the finding as “alarming” and called for more support for this patient population.
Investigators found that women with epilepsy are also more likely to report other stressors, including divorce, illness, lost pay, and partner discord, while expecting.
“As epilepsy physicians, it’s important that we ask the right questions and dive a little bit deeper with these patients, even if it’s uncomfortable and not something we’re used to,” said Dr. Chaudhry.
The findings were presented at the annual meeting of the American Epilepsy Society.
Cause for concern
Women with epilepsy may be under stress for a variety of social and economic reasons. In some women, stress can trigger seizures, and during pregnancy, this can lead to complications such as preterm labor and low birth weight.
For the study, researchers tapped into the Center for Disease Control and Prevention Pregnancy Risk Assessment and Monitoring System (PRAMS). This database includes information from surveys asking women across the U.S. about their pregnancy and postpartum period.
Thirteen states collected data on stresses in women with and without epilepsy. Respondents were asked about 14 economic and other worries in the year prior to their baby’s birth, including the pregnancy period.
The analysis included 64,951 women, 1,140 of whom had epilepsy, who were included in surveys from 2012-2020. There were no significant demographic differences between those with and those without the disorder.
After adjusting for maternal age, race, ethnicity, marital status, education, and socioeconomic status, the study found that women with epilepsy experienced an average of 2.41 of the stressors compared with 1.72 for women without epilepsy.
Women with epilepsy were more likely to have experienced family illness, divorce, homelessness, partner job loss, reduced work or pay, increased arguments, having a partner in jail, drug use, and the death of someone close to them.
The results showed that unmarried and younger women as well as those with lower incomes were particularly prone to experience stress during pregnancy.
It’s not clear why women with epilepsy report more stressors. “Looking at the literature, no one has really looked at the exact reason for this, but we postulate it could be a lack of supports and support systems,” said Dr. Chaudhry.
Women were asked about physical, sexual, and emotional abuse. Results showed that substantially more women with epilepsy than those without the disorder reported such abuse during pregnancy – 10.6% versus 4.1%. The adjusted odds ratio for women with epilepsy reporting abuse was 2.78 (95% CI, 2.07-3.74).
“That raises our concern and needs to be looked at in more detail,” said Dr. Chaudhry.
It is unclear whether some women might have had psychogenic non-epileptic seizures (PNES), which are linked to a higher rate of abuse, said Dr. Chaudhry. “But the prevalence of PNES in the general population is quite low, so we don’t think it’s contributing to a large extent to this finding.”
The findings highlight the importance of addressing stress in women with epilepsy during pregnancy, he said. “We need to have good support services and we need to counsel women to optimize good outcomes.”
This applies to all women of childbearing age. “We suspect abuse and stressors are going to be going on throughout that period,” said Dr. Chaudhry. “It’s important to ask about it and have appropriate support staff and social work and people available to help when an issue is identified.”
Stress a common seizure trigger
Commenting on the research, Kimford Meador, MD, professor, Department of Neurology and Neurological Sciences, Stanford University School of Medicine, noted the study was well conducted and had a large sample size.
The findings are important, as stress is a common trigger for seizures in people with epilepsy and is associated with mood and anxiety, which can affect quality of life, said Dr. Meador.
Results of his analysis from the Maternal Outcomes and Neurodevelopmental Effects of Antiepileptic Drugs (MONEAD) study, also presented at this year’s AES meeting, showed that women with epilepsy had more depressive symptoms during the postpartum period and more anxiety symptoms during pregnancy and postpartum in comparison with those without epilepsy.
Dr. Meador’s group also recently conducted a study that was published in JAMA Neurology, showing that in women with epilepsy during the postpartum period, anxiety is associated with lower cognitive ability in their children at age 2 years.
“All these findings highlight the importance of assessing and managing stress, anxiety, and mood in women with epilepsy,” said Dr. Meador. “Interventions could impact seizures and quality of life in pregnant women with epilepsy and long-term outcomes in their children.”
A version of this article first appeared on Medscape.com.
, new research shows.
Study investigator Naveed Chaudhry, MD, a recent epilepsy fellow and assistant professor of neurology, University of Colorado School of Medicine, described the finding as “alarming” and called for more support for this patient population.
Investigators found that women with epilepsy are also more likely to report other stressors, including divorce, illness, lost pay, and partner discord, while expecting.
“As epilepsy physicians, it’s important that we ask the right questions and dive a little bit deeper with these patients, even if it’s uncomfortable and not something we’re used to,” said Dr. Chaudhry.
The findings were presented at the annual meeting of the American Epilepsy Society.
Cause for concern
Women with epilepsy may be under stress for a variety of social and economic reasons. In some women, stress can trigger seizures, and during pregnancy, this can lead to complications such as preterm labor and low birth weight.
For the study, researchers tapped into the Center for Disease Control and Prevention Pregnancy Risk Assessment and Monitoring System (PRAMS). This database includes information from surveys asking women across the U.S. about their pregnancy and postpartum period.
Thirteen states collected data on stresses in women with and without epilepsy. Respondents were asked about 14 economic and other worries in the year prior to their baby’s birth, including the pregnancy period.
The analysis included 64,951 women, 1,140 of whom had epilepsy, who were included in surveys from 2012-2020. There were no significant demographic differences between those with and those without the disorder.
After adjusting for maternal age, race, ethnicity, marital status, education, and socioeconomic status, the study found that women with epilepsy experienced an average of 2.41 of the stressors compared with 1.72 for women without epilepsy.
Women with epilepsy were more likely to have experienced family illness, divorce, homelessness, partner job loss, reduced work or pay, increased arguments, having a partner in jail, drug use, and the death of someone close to them.
The results showed that unmarried and younger women as well as those with lower incomes were particularly prone to experience stress during pregnancy.
It’s not clear why women with epilepsy report more stressors. “Looking at the literature, no one has really looked at the exact reason for this, but we postulate it could be a lack of supports and support systems,” said Dr. Chaudhry.
Women were asked about physical, sexual, and emotional abuse. Results showed that substantially more women with epilepsy than those without the disorder reported such abuse during pregnancy – 10.6% versus 4.1%. The adjusted odds ratio for women with epilepsy reporting abuse was 2.78 (95% CI, 2.07-3.74).
“That raises our concern and needs to be looked at in more detail,” said Dr. Chaudhry.
It is unclear whether some women might have had psychogenic non-epileptic seizures (PNES), which are linked to a higher rate of abuse, said Dr. Chaudhry. “But the prevalence of PNES in the general population is quite low, so we don’t think it’s contributing to a large extent to this finding.”
The findings highlight the importance of addressing stress in women with epilepsy during pregnancy, he said. “We need to have good support services and we need to counsel women to optimize good outcomes.”
This applies to all women of childbearing age. “We suspect abuse and stressors are going to be going on throughout that period,” said Dr. Chaudhry. “It’s important to ask about it and have appropriate support staff and social work and people available to help when an issue is identified.”
Stress a common seizure trigger
Commenting on the research, Kimford Meador, MD, professor, Department of Neurology and Neurological Sciences, Stanford University School of Medicine, noted the study was well conducted and had a large sample size.
The findings are important, as stress is a common trigger for seizures in people with epilepsy and is associated with mood and anxiety, which can affect quality of life, said Dr. Meador.
Results of his analysis from the Maternal Outcomes and Neurodevelopmental Effects of Antiepileptic Drugs (MONEAD) study, also presented at this year’s AES meeting, showed that women with epilepsy had more depressive symptoms during the postpartum period and more anxiety symptoms during pregnancy and postpartum in comparison with those without epilepsy.
Dr. Meador’s group also recently conducted a study that was published in JAMA Neurology, showing that in women with epilepsy during the postpartum period, anxiety is associated with lower cognitive ability in their children at age 2 years.
“All these findings highlight the importance of assessing and managing stress, anxiety, and mood in women with epilepsy,” said Dr. Meador. “Interventions could impact seizures and quality of life in pregnant women with epilepsy and long-term outcomes in their children.”
A version of this article first appeared on Medscape.com.
From AES 2021
Could Viagra help prevent Alzheimer’s?
published in the journal Nature Aging.
Patients who used the drug sildenafil, the generic name for Viagra, were 69% less likely to develop the disease than were nonusers.
“Sildenafil, which has been shown to significantly improve cognition and memory in preclinical models, presented as the best drug candidate,” Feixiong Cheng, PhD, the lead study author in the Cleveland Clinic’s Genomic Medicine Institute, said in a statement.
“Notably, we found that sildenafil use reduced the likelihood of Alzheimer’s in individuals with coronary artery disease, hypertension, and type 2 diabetes, all of which are comorbidities significantly associated with risk of the disease, as well as in those without,” he said.
Alzheimer’s, which is the most common form of age-related dementia, affects hundreds of millions of people worldwide. The disease is expected to affect nearly 14 million Americans by 2050. There is no approved treatment for it.
Dr. Cheng and colleagues at the Cleveland Clinic used a large gene-mapping network to analyze whether more than 1,600 Food and Drug Administration–approved drugs could work against Alzheimer’s. They gave higher scores to drugs that target both amyloid and tau proteins in the brain, which are two hallmarks of the disease. Sildenafil appeared at the top of the list.
Then the researchers used a database of health insurance claims for more than 7 million people in the U.S. to understand the relationship between sildenafil and Alzheimer’s disease outcomes. They compared sildenafil users to nonusers and found that those who used the drug were 69% less likely to have the neurodegenerative disease, even after 6 years of follow-up.
After that, the research team came up with a lab model that showed the sildenafil increased brain cell growth and targeted tau proteins. The lab model could indicate how the drug influences disease-related brain changes.
But Dr. Cheng cautioned against drawing strong conclusions. The study doesn’t demonstrate a causal relationship between sildenafil and Alzheimer’s disease. Researchers will need to conduct clinical trials with a placebo control to see how well the drug works.
Other researchers said the findings offer a new avenue for research but don’t yet provide solid answers.
“Being able to repurpose a drug already licensed for health conditions could help speed up the drug discovery process and bring about life-changing dementia treatments sooner,” Susan Kohlhaas, PhD, director of research at Alzheimer’s Research UK, told the Science Media Centre.
“Importantly, this research doesn’t prove that sildenafil is responsible for reducing dementia risk, or that it slows or stops the disease,” she continued. “If you want to discuss any treatments you are receiving, the first port of call is to speak to your doctor.”
And doctors won’t likely recommend it as a treatment just yet either.
“While these data are interesting scientifically, based on this study, I would not rush out to start taking sildenafil as a prevention for Alzheimer’s disease,” Tara Spires-Jones, PhD, deputy director of the Centre for Discovery Brain Sciences at the University of Edinburgh, told the Science Media Centre.
A version of this article first appeared on WebMD.com.
published in the journal Nature Aging.
Patients who used the drug sildenafil, the generic name for Viagra, were 69% less likely to develop the disease than were nonusers.
“Sildenafil, which has been shown to significantly improve cognition and memory in preclinical models, presented as the best drug candidate,” Feixiong Cheng, PhD, the lead study author in the Cleveland Clinic’s Genomic Medicine Institute, said in a statement.
“Notably, we found that sildenafil use reduced the likelihood of Alzheimer’s in individuals with coronary artery disease, hypertension, and type 2 diabetes, all of which are comorbidities significantly associated with risk of the disease, as well as in those without,” he said.
Alzheimer’s, which is the most common form of age-related dementia, affects hundreds of millions of people worldwide. The disease is expected to affect nearly 14 million Americans by 2050. There is no approved treatment for it.
Dr. Cheng and colleagues at the Cleveland Clinic used a large gene-mapping network to analyze whether more than 1,600 Food and Drug Administration–approved drugs could work against Alzheimer’s. They gave higher scores to drugs that target both amyloid and tau proteins in the brain, which are two hallmarks of the disease. Sildenafil appeared at the top of the list.
Then the researchers used a database of health insurance claims for more than 7 million people in the U.S. to understand the relationship between sildenafil and Alzheimer’s disease outcomes. They compared sildenafil users to nonusers and found that those who used the drug were 69% less likely to have the neurodegenerative disease, even after 6 years of follow-up.
After that, the research team came up with a lab model that showed the sildenafil increased brain cell growth and targeted tau proteins. The lab model could indicate how the drug influences disease-related brain changes.
But Dr. Cheng cautioned against drawing strong conclusions. The study doesn’t demonstrate a causal relationship between sildenafil and Alzheimer’s disease. Researchers will need to conduct clinical trials with a placebo control to see how well the drug works.
Other researchers said the findings offer a new avenue for research but don’t yet provide solid answers.
“Being able to repurpose a drug already licensed for health conditions could help speed up the drug discovery process and bring about life-changing dementia treatments sooner,” Susan Kohlhaas, PhD, director of research at Alzheimer’s Research UK, told the Science Media Centre.
“Importantly, this research doesn’t prove that sildenafil is responsible for reducing dementia risk, or that it slows or stops the disease,” she continued. “If you want to discuss any treatments you are receiving, the first port of call is to speak to your doctor.”
And doctors won’t likely recommend it as a treatment just yet either.
“While these data are interesting scientifically, based on this study, I would not rush out to start taking sildenafil as a prevention for Alzheimer’s disease,” Tara Spires-Jones, PhD, deputy director of the Centre for Discovery Brain Sciences at the University of Edinburgh, told the Science Media Centre.
A version of this article first appeared on WebMD.com.
published in the journal Nature Aging.
Patients who used the drug sildenafil, the generic name for Viagra, were 69% less likely to develop the disease than were nonusers.
“Sildenafil, which has been shown to significantly improve cognition and memory in preclinical models, presented as the best drug candidate,” Feixiong Cheng, PhD, the lead study author in the Cleveland Clinic’s Genomic Medicine Institute, said in a statement.
“Notably, we found that sildenafil use reduced the likelihood of Alzheimer’s in individuals with coronary artery disease, hypertension, and type 2 diabetes, all of which are comorbidities significantly associated with risk of the disease, as well as in those without,” he said.
Alzheimer’s, which is the most common form of age-related dementia, affects hundreds of millions of people worldwide. The disease is expected to affect nearly 14 million Americans by 2050. There is no approved treatment for it.
Dr. Cheng and colleagues at the Cleveland Clinic used a large gene-mapping network to analyze whether more than 1,600 Food and Drug Administration–approved drugs could work against Alzheimer’s. They gave higher scores to drugs that target both amyloid and tau proteins in the brain, which are two hallmarks of the disease. Sildenafil appeared at the top of the list.
Then the researchers used a database of health insurance claims for more than 7 million people in the U.S. to understand the relationship between sildenafil and Alzheimer’s disease outcomes. They compared sildenafil users to nonusers and found that those who used the drug were 69% less likely to have the neurodegenerative disease, even after 6 years of follow-up.
After that, the research team came up with a lab model that showed the sildenafil increased brain cell growth and targeted tau proteins. The lab model could indicate how the drug influences disease-related brain changes.
But Dr. Cheng cautioned against drawing strong conclusions. The study doesn’t demonstrate a causal relationship between sildenafil and Alzheimer’s disease. Researchers will need to conduct clinical trials with a placebo control to see how well the drug works.
Other researchers said the findings offer a new avenue for research but don’t yet provide solid answers.
“Being able to repurpose a drug already licensed for health conditions could help speed up the drug discovery process and bring about life-changing dementia treatments sooner,” Susan Kohlhaas, PhD, director of research at Alzheimer’s Research UK, told the Science Media Centre.
“Importantly, this research doesn’t prove that sildenafil is responsible for reducing dementia risk, or that it slows or stops the disease,” she continued. “If you want to discuss any treatments you are receiving, the first port of call is to speak to your doctor.”
And doctors won’t likely recommend it as a treatment just yet either.
“While these data are interesting scientifically, based on this study, I would not rush out to start taking sildenafil as a prevention for Alzheimer’s disease,” Tara Spires-Jones, PhD, deputy director of the Centre for Discovery Brain Sciences at the University of Edinburgh, told the Science Media Centre.
A version of this article first appeared on WebMD.com.
FROM NATURE AGING
Higher resting heart rate tied to increased dementia risk
independent of the presence of cardiovascular disease (CVD) risk factors, new research shows.
“RHR is easy to measure and might be used to identify older people potentially at high risk of dementia and cognitive decline for early interventions,” Yume Imahori, MD, PhD, with the Aging Research Center, Karolinska Institutet, Stockholm, said in an interview.
“Health care professionals should be aware of potential cognitive consequences associated with elevated RHR in older people and may advise older people with high RHR to have a follow-up assessment of cognitive function,” Dr. Imahori said.
The study was published online Dec. 3, 2021, in Alzheimer’s & Dementia.
Heart-brain connection
The findings are based on 2,147 adults (62% women) aged 60 years and older (mean age, 70.6 years) from the population-based Swedish National Aging and Care in Kungsholmen (SNAC-K) study. All were free of dementia at baseline and were followed regularly from 2001-2004 to 2013-2016.
The average RHR at baseline was 65.7 bpm. Individuals in higher RHR groups were older, less educated, and were more likely to be smokers and sedentary and to have hypertension. There were no differences among RHR groups in the prevalence of CVD at baseline.
During a median follow-up of 11.4 years, 289 participants were diagnosed with dementia.
In the fully adjusted model, participants with RHR of 80 bpm or higher had a 55% increased risk of developing dementia, compared with peers with lower RHR of 60 to 69 bpm (hazard ratio, 1.55; 95% CI, 1.06-2.27).
“This association was not due to underlying cardiovascular diseases such as atrial fibrillation and heart failure, which is important because elevated RHR is often related to heart disease,” Dr. Imahori said in an interview.
Regarding cognitive function, Mini-Mental State Examination scores declined over time during the follow-up period in all RHR groups, but participants with RHR 70-79 and 80+ bpm had a greater decline, compared with those with lower RHR of 60-69 bpm.
Dr. Imahori said these findings are in line with data from the U.S. Atherosclerosis Risk in Communities study linking elevated RHR of 80+ bpm in midlife to dementia and cognitive decline in late life.
Public health implications
Reached for comment, Claire Sexton, DPhil, Alzheimer’s Association director of scientific programs and outreach, said this study adds to the “growing body of research showing the health of the heart and brain are closely connected. However, this study only shows a correlation between resting heart rate and cognition, not causation. More research is needed.
“Evidence shows that other risk factors for cardiovascular disease and stroke – obesity, high blood pressure, and diabetes – negatively impact your cognitive health,” Dr. Sexton said in an interview.
“The Alzheimer’s Association believes the conversation about heart health management is something everyone should be having with their doctor,” she said.
“There are things you can do today to lower your risk for cardiovascular disease, including regular exercise and maintaining a healthy diet. Improving your heart health is an important step to maintaining your brain health as you age,” Dr. Sexton added.
SNAC-K is supported by the Swedish Ministry of Health and Social Affairs and the participating county councils and municipalities and in part by additional grants from the Swedish Research Council and the Swedish Research Council for Health, Working Life and Welfare. Dr. Imahori and Dr. Sexton disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
independent of the presence of cardiovascular disease (CVD) risk factors, new research shows.
“RHR is easy to measure and might be used to identify older people potentially at high risk of dementia and cognitive decline for early interventions,” Yume Imahori, MD, PhD, with the Aging Research Center, Karolinska Institutet, Stockholm, said in an interview.
“Health care professionals should be aware of potential cognitive consequences associated with elevated RHR in older people and may advise older people with high RHR to have a follow-up assessment of cognitive function,” Dr. Imahori said.
The study was published online Dec. 3, 2021, in Alzheimer’s & Dementia.
Heart-brain connection
The findings are based on 2,147 adults (62% women) aged 60 years and older (mean age, 70.6 years) from the population-based Swedish National Aging and Care in Kungsholmen (SNAC-K) study. All were free of dementia at baseline and were followed regularly from 2001-2004 to 2013-2016.
The average RHR at baseline was 65.7 bpm. Individuals in higher RHR groups were older, less educated, and were more likely to be smokers and sedentary and to have hypertension. There were no differences among RHR groups in the prevalence of CVD at baseline.
During a median follow-up of 11.4 years, 289 participants were diagnosed with dementia.
In the fully adjusted model, participants with RHR of 80 bpm or higher had a 55% increased risk of developing dementia, compared with peers with lower RHR of 60 to 69 bpm (hazard ratio, 1.55; 95% CI, 1.06-2.27).
“This association was not due to underlying cardiovascular diseases such as atrial fibrillation and heart failure, which is important because elevated RHR is often related to heart disease,” Dr. Imahori said in an interview.
Regarding cognitive function, Mini-Mental State Examination scores declined over time during the follow-up period in all RHR groups, but participants with RHR 70-79 and 80+ bpm had a greater decline, compared with those with lower RHR of 60-69 bpm.
Dr. Imahori said these findings are in line with data from the U.S. Atherosclerosis Risk in Communities study linking elevated RHR of 80+ bpm in midlife to dementia and cognitive decline in late life.
Public health implications
Reached for comment, Claire Sexton, DPhil, Alzheimer’s Association director of scientific programs and outreach, said this study adds to the “growing body of research showing the health of the heart and brain are closely connected. However, this study only shows a correlation between resting heart rate and cognition, not causation. More research is needed.
“Evidence shows that other risk factors for cardiovascular disease and stroke – obesity, high blood pressure, and diabetes – negatively impact your cognitive health,” Dr. Sexton said in an interview.
“The Alzheimer’s Association believes the conversation about heart health management is something everyone should be having with their doctor,” she said.
“There are things you can do today to lower your risk for cardiovascular disease, including regular exercise and maintaining a healthy diet. Improving your heart health is an important step to maintaining your brain health as you age,” Dr. Sexton added.
SNAC-K is supported by the Swedish Ministry of Health and Social Affairs and the participating county councils and municipalities and in part by additional grants from the Swedish Research Council and the Swedish Research Council for Health, Working Life and Welfare. Dr. Imahori and Dr. Sexton disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
independent of the presence of cardiovascular disease (CVD) risk factors, new research shows.
“RHR is easy to measure and might be used to identify older people potentially at high risk of dementia and cognitive decline for early interventions,” Yume Imahori, MD, PhD, with the Aging Research Center, Karolinska Institutet, Stockholm, said in an interview.
“Health care professionals should be aware of potential cognitive consequences associated with elevated RHR in older people and may advise older people with high RHR to have a follow-up assessment of cognitive function,” Dr. Imahori said.
The study was published online Dec. 3, 2021, in Alzheimer’s & Dementia.
Heart-brain connection
The findings are based on 2,147 adults (62% women) aged 60 years and older (mean age, 70.6 years) from the population-based Swedish National Aging and Care in Kungsholmen (SNAC-K) study. All were free of dementia at baseline and were followed regularly from 2001-2004 to 2013-2016.
The average RHR at baseline was 65.7 bpm. Individuals in higher RHR groups were older, less educated, and were more likely to be smokers and sedentary and to have hypertension. There were no differences among RHR groups in the prevalence of CVD at baseline.
During a median follow-up of 11.4 years, 289 participants were diagnosed with dementia.
In the fully adjusted model, participants with RHR of 80 bpm or higher had a 55% increased risk of developing dementia, compared with peers with lower RHR of 60 to 69 bpm (hazard ratio, 1.55; 95% CI, 1.06-2.27).
“This association was not due to underlying cardiovascular diseases such as atrial fibrillation and heart failure, which is important because elevated RHR is often related to heart disease,” Dr. Imahori said in an interview.
Regarding cognitive function, Mini-Mental State Examination scores declined over time during the follow-up period in all RHR groups, but participants with RHR 70-79 and 80+ bpm had a greater decline, compared with those with lower RHR of 60-69 bpm.
Dr. Imahori said these findings are in line with data from the U.S. Atherosclerosis Risk in Communities study linking elevated RHR of 80+ bpm in midlife to dementia and cognitive decline in late life.
Public health implications
Reached for comment, Claire Sexton, DPhil, Alzheimer’s Association director of scientific programs and outreach, said this study adds to the “growing body of research showing the health of the heart and brain are closely connected. However, this study only shows a correlation between resting heart rate and cognition, not causation. More research is needed.
“Evidence shows that other risk factors for cardiovascular disease and stroke – obesity, high blood pressure, and diabetes – negatively impact your cognitive health,” Dr. Sexton said in an interview.
“The Alzheimer’s Association believes the conversation about heart health management is something everyone should be having with their doctor,” she said.
“There are things you can do today to lower your risk for cardiovascular disease, including regular exercise and maintaining a healthy diet. Improving your heart health is an important step to maintaining your brain health as you age,” Dr. Sexton added.
SNAC-K is supported by the Swedish Ministry of Health and Social Affairs and the participating county councils and municipalities and in part by additional grants from the Swedish Research Council and the Swedish Research Council for Health, Working Life and Welfare. Dr. Imahori and Dr. Sexton disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ALZHEIMER’S & DEMENTIA
Is mindfulness key to helping physicians with mental health?
In 2011, the Mayo Clinic began surveying physicians about burnout and found 45% of physicians experienced at least one symptom, such as emotional exhaustion, finding work no longer meaningful, feelings of ineffectiveness, and depersonalizing patients. Associated manifestations can range from headache and insomnia to impaired memory and decreased attention.
Fast forward 10 years to the Medscape National Physician Burnout and Suicide Report, which found that a similar number of physicians (42%) feel burned out. The COVID-19 pandemic only added insult to injury. A Medscape survey that included nearly 5,000 U.S. physicians revealed that about two-thirds (64%) of them reported burnout had intensified during the crisis.
These elevated numbers are being labeled as “a public health crisis” for the impact widespread physician burnout could have on the health of the doctor and patient safety. The relatively consistent levels across the decade seem to suggest that, if health organizations are attempting to improve physician well-being, it doesn’t appear to be working, forcing doctors to find solutions for themselves.
Jill Wener, MD, considers herself part of the 45% burned out 10 years ago. She was working as an internist at Rush University Medical Center in Chicago, but the “existential reality of being a doctor in this world” was wearing on her. “Staying up with the literature, knowing that every day you’re going to go into work without knowing what you’re going to find, threats of lawsuits, the pressure of perfectionism,” Dr. Wener told this news organization. “By the time I hit burnout, everything made me feel like the world was crashing down on me.”
When Dr. Wener encountered someone who meditated twice a day, she was intrigued, even though the self-described “most Type-A, inside-the-box, nonspiritual type, anxious, linear-path doctor” didn’t think people like her could meditate. Dr. Wener is not alone in her hesitation to explore meditation as a means to help prevent burnout because the causes of burnout are primarily linked to external rather than internal factors. Issues including a loss of autonomy, the burden and distraction of electronic health records, and the intense pressure to comply with rules from the government are not things mindfulness can fix.
And because the sources of burnout are primarily environmental and inherent to the current medical system, the suggestion that physicians need to fix themselves with meditation can come as a slap in the face. However, when up against a system slow to change, mindfulness can provide physicians access to the one thing they can control: How they perceive and react to what’s in front of them.
At the recommendation of an acquaintance, Dr. Wener enrolled in a Vedic Meditation (also known as Conscious Health Meditation) course taught by Light Watkins, a well-known traveling instructor, author, and speaker. By the second meeting she was successfully practicing 20 minutes twice a day. This form of mediation traces its roots to the Vedas, ancient Indian texts (also the foundation for yoga), and uses a mantra to settle the mind, transitioning to an awake state of inner contentment.
Three weeks later, Dr. Wener’s daily crying jags ended as did her propensity for road rage. “I felt like I was on the cusp of something life-changing, I just didn’t understand it,” she recalled. “But I knew I was never going to give it up.”
Defining mindfulness
“Mindfulness is being able to be present in the moment that you’re in with acceptance of what it is and without judging it,” said Donna Rockwell, PsyD, a leading mindfulness meditation teacher. The practice of mindfulness is really meditation. Dr. Rockwell explained that the noise of our mind is most often focused on either the past or the future. “We’re either bemoaning something that happened earlier or we’re catastrophizing the future,” she said, which prevents us from being present in the moment.
Meditation allows you to notice when your mind has drifted from the present moment into the past or future. “You gently notice it, label it with a lot of self-compassion, and then bring your mind back by focusing on your breath – going out, going in – and the incoming stimuli through your five senses,” said Dr. Rockwell. “When you’re doing that, you can’t be in the past or future.”
Dr. Rockwell also pointed out that we constantly categorize incoming data of the moment as either “good for me or bad for me,” which gets in the way of simply being present for what you’re facing. “When you’re more fully present, you become more skillful and able to do what this moment is asking of you,” she said. Being mindful allows us to better navigate incoming stimuli, which could be a “code blue” in the ED or a patient who needs another 2 minutes during an office visit.
When Dr. Wener was burned out, she felt unable to adapt whenever something unexpected happened. “When you have no emotional reserves, everything feels like a big deal,” she said. “The meditation gave me what we call adaptation energy; it filled up my tank and kept me from feeling like I was going to lose it at 10 o’clock in the morning.”
Dr. Rockwell explained burnout as an overactive fight or flight response activated by the amygdala. It starts pumping cortisol, our pupils dilate, and our pores open. The prefrontal cortex is offline when we’re experiencing this physiological response because they both can’t be operational at the same time. “When we’re constantly in a ‘fight or flight’ response and don’t have any access to our prefrontal cortex, we are coming from a brain that is pumping cortisol and that leads to burnout,” said Dr. Rockwell.
“Any fight or flight response leaves a mark on your body,” Dr. Wener echoed. “When we go into our state of deep rest in the meditation practice, which is two to five times more restful than sleep, it heals those stress scars.”
Making time for mindfulness
Prescribing mindfulness for physicians is not new. Molecular biologist Jon Kabat-Zinn, PhD, developed Mindfulness-Based Stress Reduction (MBSR) in 1979, a practice that incorporates mindfulness exercises to help people become familiar with their behavior patterns in stressful situations. Thus, instead of reacting, they can respond with a clearer understanding of the circumstance. Dr. Kabat-Zinn initially targeted people with chronic health problems to help them cope with the effects of pain and the condition of their illness, but it has expanded to anyone experiencing challenges in their life, including physicians. A standard MBSR course runs 8 weeks, making it a commitment for most people.
Mindfulness training requires that physicians use what they already have so little of: time.
Dr. Wener was able to take a sabbatical, embarking on a 3-month trip to India to immerse herself in the study of Vedic Meditation. Upon her return, Dr. Wener took a position at Emory University, Atlanta, and has launched a number of CME-accredited meditation courses and retreats. Unlike Dr. Kabat-Zinn, her programs are by physicians and for physicians. She also created an online version of the meditation course to make it more accessible.
For these reasons, Kara Pepper, MD, an internist in outpatient primary care in Atlanta, was drawn to the meditation course. Dr. Pepper was 7 years into practice when she burned out. “The program dovetailed into my burnout recovery,” she said. “It allowed me space to separate myself from the thoughts I was having about work and just recognize them as just that – as thoughts.”
In the course, Dr. Wener teaches the REST Technique, which she says is different than mindfulness in that she encourages the mind to run rampant. “Trying to control the mind can feel very uncomfortable because we always have thoughts,” she says. “We can’t tell the mind to stop thinking just like we can’t tell the heart to stop beating.” Dr. Wener said the REST Technique lets “the mind swim downstream,” allowing the brain to go into a deep state of rest and start to heal from the scars caused by stress.
Dr. Pepper said the self-paced online course gave her all the tools she needed, and it was pragmatic and evidence based. “I didn’t feel ‘woo’ or like another gimmick,” she said. Pepper, who continues to practice medicine, became a life coach in 2019 to teach others the skills she uses daily.
An integrated strategy
In a review published in The American Journal of Medicine in 2019, Scott Yates, MD, MBA, from the Center for Executive Medicine in Plano, Tex., found that physicians who had adopted mediation and mindfulness training to decrease anxiety and perceived work stress only experienced modest benefits. In fact, Dr. Yates claims that there’s little data to suggest the long-term benefit of any particular stress management intervention in the prevention of burnout symptoms.
“The often-repeated goals of the Triple Aim [enhancing patient experience, improving population health, and reducing costs] may be unreachable until we recognize and address burnout in health care providers,” Dr. Yates wrote. He recommends adding a fourth goal to specifically address physician wellness, which certainly could include mindfulness training and meditation.
Burnout coach, trainer, and consultant Dike Drummond, MD, also professes that physician wellness must be added as the key fourth ingredient to improving health care. “Burnout is a dilemma, a balancing act,” he said. “It takes an integrated strategy.” The CEO and founder of TheHappyMD.com, Dr. Drummond’s integrated strategy to stop physician burnout has been taught to more than 40,000 physicians in 175 organizations, and one element of that strategy can be mindfulness training.
Dr. Drummond said he doesn’t use the word meditation “because that scares most people”; it takes a commitment and isn’t accessible for a lot of doctors. Instead, he coaches doctors to use a ‘single-breath’ technique to help them reset multiple times throughout the day. “I teach people how to breathe up to the top of their head and then down to the bottom of their feet,” Dr. Drummond said. He calls it the Squeegee Breath Technique because when they exhale, they “wipe away” anything that doesn’t need to be there right now. “If you happen to have a mindfulness practice like meditation, they work synergistically because the calmness you feel in your mediation is available to you at the bottom of these releasing breaths.”
Various studies and surveys provide great detail as to the “why” of physician burnout. And while mindfulness is not the sole answer, it’s something physicians can explore for themselves while health care as an industry looks for a more comprehensive solution.
“It’s not rocket science,” Dr. Drummond insisted. “You want a different result? You’re not satisfied with the way things are now and you want to feel different? You absolutely must do something different.”
A version of this article first appeared on Medscape.com.
In 2011, the Mayo Clinic began surveying physicians about burnout and found 45% of physicians experienced at least one symptom, such as emotional exhaustion, finding work no longer meaningful, feelings of ineffectiveness, and depersonalizing patients. Associated manifestations can range from headache and insomnia to impaired memory and decreased attention.
Fast forward 10 years to the Medscape National Physician Burnout and Suicide Report, which found that a similar number of physicians (42%) feel burned out. The COVID-19 pandemic only added insult to injury. A Medscape survey that included nearly 5,000 U.S. physicians revealed that about two-thirds (64%) of them reported burnout had intensified during the crisis.
These elevated numbers are being labeled as “a public health crisis” for the impact widespread physician burnout could have on the health of the doctor and patient safety. The relatively consistent levels across the decade seem to suggest that, if health organizations are attempting to improve physician well-being, it doesn’t appear to be working, forcing doctors to find solutions for themselves.
Jill Wener, MD, considers herself part of the 45% burned out 10 years ago. She was working as an internist at Rush University Medical Center in Chicago, but the “existential reality of being a doctor in this world” was wearing on her. “Staying up with the literature, knowing that every day you’re going to go into work without knowing what you’re going to find, threats of lawsuits, the pressure of perfectionism,” Dr. Wener told this news organization. “By the time I hit burnout, everything made me feel like the world was crashing down on me.”
When Dr. Wener encountered someone who meditated twice a day, she was intrigued, even though the self-described “most Type-A, inside-the-box, nonspiritual type, anxious, linear-path doctor” didn’t think people like her could meditate. Dr. Wener is not alone in her hesitation to explore meditation as a means to help prevent burnout because the causes of burnout are primarily linked to external rather than internal factors. Issues including a loss of autonomy, the burden and distraction of electronic health records, and the intense pressure to comply with rules from the government are not things mindfulness can fix.
And because the sources of burnout are primarily environmental and inherent to the current medical system, the suggestion that physicians need to fix themselves with meditation can come as a slap in the face. However, when up against a system slow to change, mindfulness can provide physicians access to the one thing they can control: How they perceive and react to what’s in front of them.
At the recommendation of an acquaintance, Dr. Wener enrolled in a Vedic Meditation (also known as Conscious Health Meditation) course taught by Light Watkins, a well-known traveling instructor, author, and speaker. By the second meeting she was successfully practicing 20 minutes twice a day. This form of mediation traces its roots to the Vedas, ancient Indian texts (also the foundation for yoga), and uses a mantra to settle the mind, transitioning to an awake state of inner contentment.
Three weeks later, Dr. Wener’s daily crying jags ended as did her propensity for road rage. “I felt like I was on the cusp of something life-changing, I just didn’t understand it,” she recalled. “But I knew I was never going to give it up.”
Defining mindfulness
“Mindfulness is being able to be present in the moment that you’re in with acceptance of what it is and without judging it,” said Donna Rockwell, PsyD, a leading mindfulness meditation teacher. The practice of mindfulness is really meditation. Dr. Rockwell explained that the noise of our mind is most often focused on either the past or the future. “We’re either bemoaning something that happened earlier or we’re catastrophizing the future,” she said, which prevents us from being present in the moment.
Meditation allows you to notice when your mind has drifted from the present moment into the past or future. “You gently notice it, label it with a lot of self-compassion, and then bring your mind back by focusing on your breath – going out, going in – and the incoming stimuli through your five senses,” said Dr. Rockwell. “When you’re doing that, you can’t be in the past or future.”
Dr. Rockwell also pointed out that we constantly categorize incoming data of the moment as either “good for me or bad for me,” which gets in the way of simply being present for what you’re facing. “When you’re more fully present, you become more skillful and able to do what this moment is asking of you,” she said. Being mindful allows us to better navigate incoming stimuli, which could be a “code blue” in the ED or a patient who needs another 2 minutes during an office visit.
When Dr. Wener was burned out, she felt unable to adapt whenever something unexpected happened. “When you have no emotional reserves, everything feels like a big deal,” she said. “The meditation gave me what we call adaptation energy; it filled up my tank and kept me from feeling like I was going to lose it at 10 o’clock in the morning.”
Dr. Rockwell explained burnout as an overactive fight or flight response activated by the amygdala. It starts pumping cortisol, our pupils dilate, and our pores open. The prefrontal cortex is offline when we’re experiencing this physiological response because they both can’t be operational at the same time. “When we’re constantly in a ‘fight or flight’ response and don’t have any access to our prefrontal cortex, we are coming from a brain that is pumping cortisol and that leads to burnout,” said Dr. Rockwell.
“Any fight or flight response leaves a mark on your body,” Dr. Wener echoed. “When we go into our state of deep rest in the meditation practice, which is two to five times more restful than sleep, it heals those stress scars.”
Making time for mindfulness
Prescribing mindfulness for physicians is not new. Molecular biologist Jon Kabat-Zinn, PhD, developed Mindfulness-Based Stress Reduction (MBSR) in 1979, a practice that incorporates mindfulness exercises to help people become familiar with their behavior patterns in stressful situations. Thus, instead of reacting, they can respond with a clearer understanding of the circumstance. Dr. Kabat-Zinn initially targeted people with chronic health problems to help them cope with the effects of pain and the condition of their illness, but it has expanded to anyone experiencing challenges in their life, including physicians. A standard MBSR course runs 8 weeks, making it a commitment for most people.
Mindfulness training requires that physicians use what they already have so little of: time.
Dr. Wener was able to take a sabbatical, embarking on a 3-month trip to India to immerse herself in the study of Vedic Meditation. Upon her return, Dr. Wener took a position at Emory University, Atlanta, and has launched a number of CME-accredited meditation courses and retreats. Unlike Dr. Kabat-Zinn, her programs are by physicians and for physicians. She also created an online version of the meditation course to make it more accessible.
For these reasons, Kara Pepper, MD, an internist in outpatient primary care in Atlanta, was drawn to the meditation course. Dr. Pepper was 7 years into practice when she burned out. “The program dovetailed into my burnout recovery,” she said. “It allowed me space to separate myself from the thoughts I was having about work and just recognize them as just that – as thoughts.”
In the course, Dr. Wener teaches the REST Technique, which she says is different than mindfulness in that she encourages the mind to run rampant. “Trying to control the mind can feel very uncomfortable because we always have thoughts,” she says. “We can’t tell the mind to stop thinking just like we can’t tell the heart to stop beating.” Dr. Wener said the REST Technique lets “the mind swim downstream,” allowing the brain to go into a deep state of rest and start to heal from the scars caused by stress.
Dr. Pepper said the self-paced online course gave her all the tools she needed, and it was pragmatic and evidence based. “I didn’t feel ‘woo’ or like another gimmick,” she said. Pepper, who continues to practice medicine, became a life coach in 2019 to teach others the skills she uses daily.
An integrated strategy
In a review published in The American Journal of Medicine in 2019, Scott Yates, MD, MBA, from the Center for Executive Medicine in Plano, Tex., found that physicians who had adopted mediation and mindfulness training to decrease anxiety and perceived work stress only experienced modest benefits. In fact, Dr. Yates claims that there’s little data to suggest the long-term benefit of any particular stress management intervention in the prevention of burnout symptoms.
“The often-repeated goals of the Triple Aim [enhancing patient experience, improving population health, and reducing costs] may be unreachable until we recognize and address burnout in health care providers,” Dr. Yates wrote. He recommends adding a fourth goal to specifically address physician wellness, which certainly could include mindfulness training and meditation.
Burnout coach, trainer, and consultant Dike Drummond, MD, also professes that physician wellness must be added as the key fourth ingredient to improving health care. “Burnout is a dilemma, a balancing act,” he said. “It takes an integrated strategy.” The CEO and founder of TheHappyMD.com, Dr. Drummond’s integrated strategy to stop physician burnout has been taught to more than 40,000 physicians in 175 organizations, and one element of that strategy can be mindfulness training.
Dr. Drummond said he doesn’t use the word meditation “because that scares most people”; it takes a commitment and isn’t accessible for a lot of doctors. Instead, he coaches doctors to use a ‘single-breath’ technique to help them reset multiple times throughout the day. “I teach people how to breathe up to the top of their head and then down to the bottom of their feet,” Dr. Drummond said. He calls it the Squeegee Breath Technique because when they exhale, they “wipe away” anything that doesn’t need to be there right now. “If you happen to have a mindfulness practice like meditation, they work synergistically because the calmness you feel in your mediation is available to you at the bottom of these releasing breaths.”
Various studies and surveys provide great detail as to the “why” of physician burnout. And while mindfulness is not the sole answer, it’s something physicians can explore for themselves while health care as an industry looks for a more comprehensive solution.
“It’s not rocket science,” Dr. Drummond insisted. “You want a different result? You’re not satisfied with the way things are now and you want to feel different? You absolutely must do something different.”
A version of this article first appeared on Medscape.com.
In 2011, the Mayo Clinic began surveying physicians about burnout and found 45% of physicians experienced at least one symptom, such as emotional exhaustion, finding work no longer meaningful, feelings of ineffectiveness, and depersonalizing patients. Associated manifestations can range from headache and insomnia to impaired memory and decreased attention.
Fast forward 10 years to the Medscape National Physician Burnout and Suicide Report, which found that a similar number of physicians (42%) feel burned out. The COVID-19 pandemic only added insult to injury. A Medscape survey that included nearly 5,000 U.S. physicians revealed that about two-thirds (64%) of them reported burnout had intensified during the crisis.
These elevated numbers are being labeled as “a public health crisis” for the impact widespread physician burnout could have on the health of the doctor and patient safety. The relatively consistent levels across the decade seem to suggest that, if health organizations are attempting to improve physician well-being, it doesn’t appear to be working, forcing doctors to find solutions for themselves.
Jill Wener, MD, considers herself part of the 45% burned out 10 years ago. She was working as an internist at Rush University Medical Center in Chicago, but the “existential reality of being a doctor in this world” was wearing on her. “Staying up with the literature, knowing that every day you’re going to go into work without knowing what you’re going to find, threats of lawsuits, the pressure of perfectionism,” Dr. Wener told this news organization. “By the time I hit burnout, everything made me feel like the world was crashing down on me.”
When Dr. Wener encountered someone who meditated twice a day, she was intrigued, even though the self-described “most Type-A, inside-the-box, nonspiritual type, anxious, linear-path doctor” didn’t think people like her could meditate. Dr. Wener is not alone in her hesitation to explore meditation as a means to help prevent burnout because the causes of burnout are primarily linked to external rather than internal factors. Issues including a loss of autonomy, the burden and distraction of electronic health records, and the intense pressure to comply with rules from the government are not things mindfulness can fix.
And because the sources of burnout are primarily environmental and inherent to the current medical system, the suggestion that physicians need to fix themselves with meditation can come as a slap in the face. However, when up against a system slow to change, mindfulness can provide physicians access to the one thing they can control: How they perceive and react to what’s in front of them.
At the recommendation of an acquaintance, Dr. Wener enrolled in a Vedic Meditation (also known as Conscious Health Meditation) course taught by Light Watkins, a well-known traveling instructor, author, and speaker. By the second meeting she was successfully practicing 20 minutes twice a day. This form of mediation traces its roots to the Vedas, ancient Indian texts (also the foundation for yoga), and uses a mantra to settle the mind, transitioning to an awake state of inner contentment.
Three weeks later, Dr. Wener’s daily crying jags ended as did her propensity for road rage. “I felt like I was on the cusp of something life-changing, I just didn’t understand it,” she recalled. “But I knew I was never going to give it up.”
Defining mindfulness
“Mindfulness is being able to be present in the moment that you’re in with acceptance of what it is and without judging it,” said Donna Rockwell, PsyD, a leading mindfulness meditation teacher. The practice of mindfulness is really meditation. Dr. Rockwell explained that the noise of our mind is most often focused on either the past or the future. “We’re either bemoaning something that happened earlier or we’re catastrophizing the future,” she said, which prevents us from being present in the moment.
Meditation allows you to notice when your mind has drifted from the present moment into the past or future. “You gently notice it, label it with a lot of self-compassion, and then bring your mind back by focusing on your breath – going out, going in – and the incoming stimuli through your five senses,” said Dr. Rockwell. “When you’re doing that, you can’t be in the past or future.”
Dr. Rockwell also pointed out that we constantly categorize incoming data of the moment as either “good for me or bad for me,” which gets in the way of simply being present for what you’re facing. “When you’re more fully present, you become more skillful and able to do what this moment is asking of you,” she said. Being mindful allows us to better navigate incoming stimuli, which could be a “code blue” in the ED or a patient who needs another 2 minutes during an office visit.
When Dr. Wener was burned out, she felt unable to adapt whenever something unexpected happened. “When you have no emotional reserves, everything feels like a big deal,” she said. “The meditation gave me what we call adaptation energy; it filled up my tank and kept me from feeling like I was going to lose it at 10 o’clock in the morning.”
Dr. Rockwell explained burnout as an overactive fight or flight response activated by the amygdala. It starts pumping cortisol, our pupils dilate, and our pores open. The prefrontal cortex is offline when we’re experiencing this physiological response because they both can’t be operational at the same time. “When we’re constantly in a ‘fight or flight’ response and don’t have any access to our prefrontal cortex, we are coming from a brain that is pumping cortisol and that leads to burnout,” said Dr. Rockwell.
“Any fight or flight response leaves a mark on your body,” Dr. Wener echoed. “When we go into our state of deep rest in the meditation practice, which is two to five times more restful than sleep, it heals those stress scars.”
Making time for mindfulness
Prescribing mindfulness for physicians is not new. Molecular biologist Jon Kabat-Zinn, PhD, developed Mindfulness-Based Stress Reduction (MBSR) in 1979, a practice that incorporates mindfulness exercises to help people become familiar with their behavior patterns in stressful situations. Thus, instead of reacting, they can respond with a clearer understanding of the circumstance. Dr. Kabat-Zinn initially targeted people with chronic health problems to help them cope with the effects of pain and the condition of their illness, but it has expanded to anyone experiencing challenges in their life, including physicians. A standard MBSR course runs 8 weeks, making it a commitment for most people.
Mindfulness training requires that physicians use what they already have so little of: time.
Dr. Wener was able to take a sabbatical, embarking on a 3-month trip to India to immerse herself in the study of Vedic Meditation. Upon her return, Dr. Wener took a position at Emory University, Atlanta, and has launched a number of CME-accredited meditation courses and retreats. Unlike Dr. Kabat-Zinn, her programs are by physicians and for physicians. She also created an online version of the meditation course to make it more accessible.
For these reasons, Kara Pepper, MD, an internist in outpatient primary care in Atlanta, was drawn to the meditation course. Dr. Pepper was 7 years into practice when she burned out. “The program dovetailed into my burnout recovery,” she said. “It allowed me space to separate myself from the thoughts I was having about work and just recognize them as just that – as thoughts.”
In the course, Dr. Wener teaches the REST Technique, which she says is different than mindfulness in that she encourages the mind to run rampant. “Trying to control the mind can feel very uncomfortable because we always have thoughts,” she says. “We can’t tell the mind to stop thinking just like we can’t tell the heart to stop beating.” Dr. Wener said the REST Technique lets “the mind swim downstream,” allowing the brain to go into a deep state of rest and start to heal from the scars caused by stress.
Dr. Pepper said the self-paced online course gave her all the tools she needed, and it was pragmatic and evidence based. “I didn’t feel ‘woo’ or like another gimmick,” she said. Pepper, who continues to practice medicine, became a life coach in 2019 to teach others the skills she uses daily.
An integrated strategy
In a review published in The American Journal of Medicine in 2019, Scott Yates, MD, MBA, from the Center for Executive Medicine in Plano, Tex., found that physicians who had adopted mediation and mindfulness training to decrease anxiety and perceived work stress only experienced modest benefits. In fact, Dr. Yates claims that there’s little data to suggest the long-term benefit of any particular stress management intervention in the prevention of burnout symptoms.
“The often-repeated goals of the Triple Aim [enhancing patient experience, improving population health, and reducing costs] may be unreachable until we recognize and address burnout in health care providers,” Dr. Yates wrote. He recommends adding a fourth goal to specifically address physician wellness, which certainly could include mindfulness training and meditation.
Burnout coach, trainer, and consultant Dike Drummond, MD, also professes that physician wellness must be added as the key fourth ingredient to improving health care. “Burnout is a dilemma, a balancing act,” he said. “It takes an integrated strategy.” The CEO and founder of TheHappyMD.com, Dr. Drummond’s integrated strategy to stop physician burnout has been taught to more than 40,000 physicians in 175 organizations, and one element of that strategy can be mindfulness training.
Dr. Drummond said he doesn’t use the word meditation “because that scares most people”; it takes a commitment and isn’t accessible for a lot of doctors. Instead, he coaches doctors to use a ‘single-breath’ technique to help them reset multiple times throughout the day. “I teach people how to breathe up to the top of their head and then down to the bottom of their feet,” Dr. Drummond said. He calls it the Squeegee Breath Technique because when they exhale, they “wipe away” anything that doesn’t need to be there right now. “If you happen to have a mindfulness practice like meditation, they work synergistically because the calmness you feel in your mediation is available to you at the bottom of these releasing breaths.”
Various studies and surveys provide great detail as to the “why” of physician burnout. And while mindfulness is not the sole answer, it’s something physicians can explore for themselves while health care as an industry looks for a more comprehensive solution.
“It’s not rocket science,” Dr. Drummond insisted. “You want a different result? You’re not satisfied with the way things are now and you want to feel different? You absolutely must do something different.”
A version of this article first appeared on Medscape.com.
An expensive lesson
In mid-July my son strained his neck working out at the gym.
It was an obvious generic muscle pull. I told him to take some ibuprofen and use a heating pad. My wife, a nurse, told him the same thing.
Regrettably, while my medical training (hopefully) counts for something with my patients, it doesn’t mean much to my kids. The unqualified opinions of their friends and Google are far more worthwhile, convincing him he had any number of serious injuries.
As a result, while we were at work he went to the emergency department to get checked out. He was evaluated by one of my colleagues who did x-rays and a cervical spine CT. (I figure the last one was because my son kept reminding them I was a doctor). After all the results were in, the ED physician told him he had a muscle strain, and to take ibuprofen and use a heating pad.
Big surprise, huh? I’m sure he was shocked to find out that his old man knew what he was doing. Of course, I didn’t order any tests so the ED doc tops me for that in my son’s mind.
But kids not believing their parents is nothing new, and I can’t claim innocence either from what I remember of being a teenager.
Fast-forward to today. From what I can see, the total bills for his little adventure in modern medicine were around $4,000-$5,000. Granted, I’m well aware that what gets charged has no relationship to what’s actually going to be collected but I’m not going to write about modern medical charges or collections or even defensive medicine. I understand all those, and certainly don’t fault my ED colleague for how he handled it.
Reassurance isn’t cheap, though. When it’s all over, our out-of-pocket share will be roughly $1,000, which we certainly hadn’t planned for in the usually money-tight months of December and January.
That’s a lot of money for ibuprofen and a heating pad (we had both at home, and they’re around $20 total at Target, anyway).
There’s certainly no shortage of research on unnecessary ED visits for minor things, but to me this is a classic example of it. Beyond just the financial cost (which, admittedly, I’m pretty irritated with him about) he tied up a bed and ED staff that someone in more dire circumstances may have needed.
His injury could have been handled at an urgent care, or, even better, just by staying home, listening to us, and using ibuprofen and a heating pad.
, and clarify what constitutes an emergency in the first place. There’s no shortage of urgent cares and other walk-in clinics that are there specifically to handle such things during daylight hours, if they need to be seen at all.
Of course, I can’t change the results of Google searches, or the age-old teenage belief that parents are morons.
But he is going to learn about what constitutes an emergency, and what else that $1,000 could have been used for.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
In mid-July my son strained his neck working out at the gym.
It was an obvious generic muscle pull. I told him to take some ibuprofen and use a heating pad. My wife, a nurse, told him the same thing.
Regrettably, while my medical training (hopefully) counts for something with my patients, it doesn’t mean much to my kids. The unqualified opinions of their friends and Google are far more worthwhile, convincing him he had any number of serious injuries.
As a result, while we were at work he went to the emergency department to get checked out. He was evaluated by one of my colleagues who did x-rays and a cervical spine CT. (I figure the last one was because my son kept reminding them I was a doctor). After all the results were in, the ED physician told him he had a muscle strain, and to take ibuprofen and use a heating pad.
Big surprise, huh? I’m sure he was shocked to find out that his old man knew what he was doing. Of course, I didn’t order any tests so the ED doc tops me for that in my son’s mind.
But kids not believing their parents is nothing new, and I can’t claim innocence either from what I remember of being a teenager.
Fast-forward to today. From what I can see, the total bills for his little adventure in modern medicine were around $4,000-$5,000. Granted, I’m well aware that what gets charged has no relationship to what’s actually going to be collected but I’m not going to write about modern medical charges or collections or even defensive medicine. I understand all those, and certainly don’t fault my ED colleague for how he handled it.
Reassurance isn’t cheap, though. When it’s all over, our out-of-pocket share will be roughly $1,000, which we certainly hadn’t planned for in the usually money-tight months of December and January.
That’s a lot of money for ibuprofen and a heating pad (we had both at home, and they’re around $20 total at Target, anyway).
There’s certainly no shortage of research on unnecessary ED visits for minor things, but to me this is a classic example of it. Beyond just the financial cost (which, admittedly, I’m pretty irritated with him about) he tied up a bed and ED staff that someone in more dire circumstances may have needed.
His injury could have been handled at an urgent care, or, even better, just by staying home, listening to us, and using ibuprofen and a heating pad.
, and clarify what constitutes an emergency in the first place. There’s no shortage of urgent cares and other walk-in clinics that are there specifically to handle such things during daylight hours, if they need to be seen at all.
Of course, I can’t change the results of Google searches, or the age-old teenage belief that parents are morons.
But he is going to learn about what constitutes an emergency, and what else that $1,000 could have been used for.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
In mid-July my son strained his neck working out at the gym.
It was an obvious generic muscle pull. I told him to take some ibuprofen and use a heating pad. My wife, a nurse, told him the same thing.
Regrettably, while my medical training (hopefully) counts for something with my patients, it doesn’t mean much to my kids. The unqualified opinions of their friends and Google are far more worthwhile, convincing him he had any number of serious injuries.
As a result, while we were at work he went to the emergency department to get checked out. He was evaluated by one of my colleagues who did x-rays and a cervical spine CT. (I figure the last one was because my son kept reminding them I was a doctor). After all the results were in, the ED physician told him he had a muscle strain, and to take ibuprofen and use a heating pad.
Big surprise, huh? I’m sure he was shocked to find out that his old man knew what he was doing. Of course, I didn’t order any tests so the ED doc tops me for that in my son’s mind.
But kids not believing their parents is nothing new, and I can’t claim innocence either from what I remember of being a teenager.
Fast-forward to today. From what I can see, the total bills for his little adventure in modern medicine were around $4,000-$5,000. Granted, I’m well aware that what gets charged has no relationship to what’s actually going to be collected but I’m not going to write about modern medical charges or collections or even defensive medicine. I understand all those, and certainly don’t fault my ED colleague for how he handled it.
Reassurance isn’t cheap, though. When it’s all over, our out-of-pocket share will be roughly $1,000, which we certainly hadn’t planned for in the usually money-tight months of December and January.
That’s a lot of money for ibuprofen and a heating pad (we had both at home, and they’re around $20 total at Target, anyway).
There’s certainly no shortage of research on unnecessary ED visits for minor things, but to me this is a classic example of it. Beyond just the financial cost (which, admittedly, I’m pretty irritated with him about) he tied up a bed and ED staff that someone in more dire circumstances may have needed.
His injury could have been handled at an urgent care, or, even better, just by staying home, listening to us, and using ibuprofen and a heating pad.
, and clarify what constitutes an emergency in the first place. There’s no shortage of urgent cares and other walk-in clinics that are there specifically to handle such things during daylight hours, if they need to be seen at all.
Of course, I can’t change the results of Google searches, or the age-old teenage belief that parents are morons.
But he is going to learn about what constitutes an emergency, and what else that $1,000 could have been used for.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Record-breaking autism rates reported with new CDC criteria
Childhood autism rates are at the highest level since the Centers for Disease Control and Prevention began tracking the disorder in 2000, new data released Dec. 2 show.
The increase likely reflects improvements in diagnosis and identification of autism spectrum disorder (ASD), not an increase in incidence, study authors with the CDC’s Autism and Developmental Disabilities Monitoring (ADDM) Network told this news organization.
Using a new surveillance methodology, researchers found that 2.3% of 8-year-olds in communities in 11 states across the United States had an autism diagnosis in 2018, up from 1.9% in 2016.
A separate report on early identification in 4-year-olds shows that children born in 2014 were 50% more likely to receive an autism diagnosis or ASD special education classification by 48 months of age than those born in 2010, signaling improved early diagnosis.
Taken together, the data suggest efforts to raise awareness about autism are working, though researchers were quick to say much work remains.
“It was not surprising to me and in fact it was reassuring that the number of children diagnosed with autism is higher and is actually approaching prevalence of autism that has been noted in some national surveys of parents,” Stuart Shapira, MD, PhD, associate director for science in CDC’s National Center on Birth Defects and Developmental Disability, told this news organization.
“It means we’re doing a better job of identifying children, which helps to get them into services earlier so they can achieve their best developmental outcome.”
The studies, published online in Morbidity and Mortality Weekly Report, are the first to use a new ASD surveillance protocol that relies on ASD diagnosis or special education classification and billing codes and eliminates comprehensive records analysis by trained clinician reviewers.
Racial disparities
The updated methodology was less labor intensive and reduced the time it took to produce the report, but it is not without its critics, who claim the new protocol will undercount the number of children with ASD.
Created in 2000 and funded by the CDC, the ADDM Network is the only surveillance program in the United States that tracks the number and characteristics of children with ASD in multiple communities in the U.S.
When ADDM released its first report in 2007 from six states and based on data from the year 2000, ASD prevalence was 6.7 per 1,000 children, or 1 in 150 children.
In the latest report, which includes data from 2018, the autism prevalence rate across 11 states was 23.0 per 1,000 children, or 1 in 44 children.
That rate is closer to reported autism prevalence from the National Survey of Children’s Health and the National Health Interview Survey, both of which rely on parent-reported ASD diagnoses.
in Arizona, Arkansas, California, Georgia, Maryland, Minnesota, Missouri, New Jersey, Tennessee, Utah, and Wisconsin.
Children were counted as having autism if their records included an ASD diagnosis, a special education classification of ASD, or an ASD International Classification of Diseases (ICD) code. A total of 5,058 children met those criteria.
Rates of ASD ranged from a low of 1.7% in Missouri to 3.9% in California and were 4.2 times higher in boys than in girls. Just under half of the children with ASD were evaluated by age 36 months.
Although the overall ASD prevalence was similar among White, Black, Hispanic, and Asian/Pacific Islander children, the report highlighted a number of other racial disparities overall and in individual states.
For example, among those with ASD and data on cognitive ability, 35.2% had an intelligence quotient score of 70 or lower. Black children with ASD were far more likely to have an IQ of 70 or less (49.8%) than Hispanic (33.1%) or White (29.7%) children.
“The persistent disparities in co-occurring intellectual disabilities in children with autism is something that we continue to see and suggests that we need to better understand exactly what’s happening,” Matthew Maenner, PhD, an epidemiologist and autism surveillance team lead with the CDC’s National Center on Birth Defects and Developmental Disabilities, told this news organization.
Another long-standing trend observed again in the new report on prevalence among 8-year-olds is low ASD prevalence among Hispanic children. While the overall estimate showed similar autism rates, a closer review of state-level data reveals a different picture.
“In almost half of the sites, Hispanic children were less likely to be identified as having ASD,” he said. “This gets lost if you look only at the overall estimate.”
New methodology
When ADDM released its first report in 2007, autism diagnosis was widely inadequate in the United States. Relying on only confirmed ASD diagnoses would significantly underestimate the number of children with the disorder, so the CDC added “active case finding” to the protocol.
Trained clinician reviewers analyzed individual notes from medical and educational records for every 8-year-old in ADDM Network sites, looking for evidence of characteristics and behaviors associated with autism. The process was labor- and time-intensive and took up to 4 years to complete.
In 2018, the CDC began investigating ways to speed the process and came up with the strategy used in the latest report. The new protocol was faster, easier, and less expensive. Although he says cost was never the deciding factor, Dr. Maenner acknowledges that had they stuck with the original protocol, they would have been forced to reduce the number of ADDM Network sites.
Dr. Maenner argues that a comparison of the two protocols shows the new method doesn’t compromise accuracy and may actually capture children who lacked the medical or educational records the previous protocol required for a count. But not everyone agrees.
“I thought the point was to be as accurate and complete as possible in doing the surveillance,” Walter Zahorodny, PhD, associate professor of pediatrics at Rutgers University, New Brunswick, N.J., and principal investigator of the New Jersey ADDM Network site, told this news organization. “In states where there’s a high detail of information in records, like New Jersey, it’s going to underestimate the count.”
Dr. Zahorodny says the latest data prove his point. In 2016, under the old methodology, ASD prevalence was 3.1% in the state. In 2018, under the new protocol, prevalence was 2.84%, a decrease of about 20% that Dr. Zahorodny pins squarely on the elimination of ADDM clinical reviewers.
But New Jersey is the only state that participated in both the 2016 and 2018 surveillance periods to report a decrease in ASD prevalence. The other eight states all found autism rates in their states went up.
Sydney Pettygrove, PhD, associate professor of public health and pediatrics at the University of Arizona, Tucson, and a principal investigator for the ADDM site in Arizona, told this news organization that when she first learned the CDC was rolling out a new methodology, she and other investigators were concerned.
“People were really upset. I was really upset,” she said. “I had formed an opinion based on the earlier data that this would not be a good idea.”
In 2000, when ASD surveillance began in Arizona, nearly 30% of children identified by ADDM clinical reviewers as having autism had no mention of the disorder in their records. Today, that percentage is closer to 5%.
“In 2000 it would have been catastrophic to try to estimate the prevalence of autism with the new protocol,” said Dr. Pettygrove. As it turns out, under the new protocol, prevalence rates in Arizona increased from 16.0 per 1,000 children in 2016 to 24.9 in 2018.
Built-in bias eliminated?
In addition to speeding up the process, the new methodology might have other benefits as well. Under the old ADDM surveillance protocol, children who lacked certain medical or educational records did not meet the ASD case definition and weren’t counted.
A 2019 study showed that this disproportionately affected Black and Hispanic children, who had significantly less access to health care professionals than White children.
As a result, “the old methodology had a bias built into it,” Maureen Durkin, PhD, DrPH, coauthor of that study and chair of population health sciences at the University of Wisconsin–Madison and principal investigator for the ADDM site in Wisconsin, told this news organization.
“Clinician reviewers ended up putting these children in the ‘suspected ASD’ category because they couldn’t call it a case under the case definition,” Dr. Durkin said. “There was a fairly large percentage of suspected cases and a disproportionate number of those kids were children of color.”
Although she can’t say for sure, Dr. Durkin said it’s possible the new protocol could eliminate some of that bias.
CDC researchers also attribute the new method to an expanded study of early diagnosis among 4-year-olds. In previous years, only a handful of the ADDM Network sites participating in the 8-year-old surveillance project also studied early diagnosis in 4-year-olds.
This year, all 11 sites took part in the early diagnosis analysis, tripling the number of children included in the analysis. That made it possible to include, for the first time, Asian/Pacific Islander children in this analysis.
In the past, ASD prevalence has trended higher in White children, compared with other racial groups. The new data found that ASD prevalence among 4-year-olds was significantly lower in White children (12.9 per 1,000 children) than in Black, Hispanic, or Asian/Pacific Islander children (16.6, 21.1, and 22.7 per 1,000, respectively). Prevalence in American Indian/Alaska Native children was the lowest among all racial groups (11.5 per 1,000).
It’s the first time researchers have seen this pattern in any ADDM report, Kelly Shaw, PhD, lead author of that study and an epidemiologist with the National Center on Birth Defects and Developmental Disability at the CDC, told this news organization.
These data don’t provide clues about the potential cause of that disparity, Dr. Shaw said. It’s likely an indication of better identification of ASD in those communities, she said, and not a sign of increased incidence of autism among Black, Hispanic, or Asian/Pacific Islander children.
“We don’t have any evidence to suggest or expect that autism would be increasing differentially among groups,” Dr. Shaw said.
The data suggest “we are making some progress but there certainly is still room for improvement,” Dr. Shaw said.
Study authors report no conflicts of interest.
A version of this article first appeared on Medscape.com.
Childhood autism rates are at the highest level since the Centers for Disease Control and Prevention began tracking the disorder in 2000, new data released Dec. 2 show.
The increase likely reflects improvements in diagnosis and identification of autism spectrum disorder (ASD), not an increase in incidence, study authors with the CDC’s Autism and Developmental Disabilities Monitoring (ADDM) Network told this news organization.
Using a new surveillance methodology, researchers found that 2.3% of 8-year-olds in communities in 11 states across the United States had an autism diagnosis in 2018, up from 1.9% in 2016.
A separate report on early identification in 4-year-olds shows that children born in 2014 were 50% more likely to receive an autism diagnosis or ASD special education classification by 48 months of age than those born in 2010, signaling improved early diagnosis.
Taken together, the data suggest efforts to raise awareness about autism are working, though researchers were quick to say much work remains.
“It was not surprising to me and in fact it was reassuring that the number of children diagnosed with autism is higher and is actually approaching prevalence of autism that has been noted in some national surveys of parents,” Stuart Shapira, MD, PhD, associate director for science in CDC’s National Center on Birth Defects and Developmental Disability, told this news organization.
“It means we’re doing a better job of identifying children, which helps to get them into services earlier so they can achieve their best developmental outcome.”
The studies, published online in Morbidity and Mortality Weekly Report, are the first to use a new ASD surveillance protocol that relies on ASD diagnosis or special education classification and billing codes and eliminates comprehensive records analysis by trained clinician reviewers.
Racial disparities
The updated methodology was less labor intensive and reduced the time it took to produce the report, but it is not without its critics, who claim the new protocol will undercount the number of children with ASD.
Created in 2000 and funded by the CDC, the ADDM Network is the only surveillance program in the United States that tracks the number and characteristics of children with ASD in multiple communities in the U.S.
When ADDM released its first report in 2007 from six states and based on data from the year 2000, ASD prevalence was 6.7 per 1,000 children, or 1 in 150 children.
In the latest report, which includes data from 2018, the autism prevalence rate across 11 states was 23.0 per 1,000 children, or 1 in 44 children.
That rate is closer to reported autism prevalence from the National Survey of Children’s Health and the National Health Interview Survey, both of which rely on parent-reported ASD diagnoses.
in Arizona, Arkansas, California, Georgia, Maryland, Minnesota, Missouri, New Jersey, Tennessee, Utah, and Wisconsin.
Children were counted as having autism if their records included an ASD diagnosis, a special education classification of ASD, or an ASD International Classification of Diseases (ICD) code. A total of 5,058 children met those criteria.
Rates of ASD ranged from a low of 1.7% in Missouri to 3.9% in California and were 4.2 times higher in boys than in girls. Just under half of the children with ASD were evaluated by age 36 months.
Although the overall ASD prevalence was similar among White, Black, Hispanic, and Asian/Pacific Islander children, the report highlighted a number of other racial disparities overall and in individual states.
For example, among those with ASD and data on cognitive ability, 35.2% had an intelligence quotient score of 70 or lower. Black children with ASD were far more likely to have an IQ of 70 or less (49.8%) than Hispanic (33.1%) or White (29.7%) children.
“The persistent disparities in co-occurring intellectual disabilities in children with autism is something that we continue to see and suggests that we need to better understand exactly what’s happening,” Matthew Maenner, PhD, an epidemiologist and autism surveillance team lead with the CDC’s National Center on Birth Defects and Developmental Disabilities, told this news organization.
Another long-standing trend observed again in the new report on prevalence among 8-year-olds is low ASD prevalence among Hispanic children. While the overall estimate showed similar autism rates, a closer review of state-level data reveals a different picture.
“In almost half of the sites, Hispanic children were less likely to be identified as having ASD,” he said. “This gets lost if you look only at the overall estimate.”
New methodology
When ADDM released its first report in 2007, autism diagnosis was widely inadequate in the United States. Relying on only confirmed ASD diagnoses would significantly underestimate the number of children with the disorder, so the CDC added “active case finding” to the protocol.
Trained clinician reviewers analyzed individual notes from medical and educational records for every 8-year-old in ADDM Network sites, looking for evidence of characteristics and behaviors associated with autism. The process was labor- and time-intensive and took up to 4 years to complete.
In 2018, the CDC began investigating ways to speed the process and came up with the strategy used in the latest report. The new protocol was faster, easier, and less expensive. Although he says cost was never the deciding factor, Dr. Maenner acknowledges that had they stuck with the original protocol, they would have been forced to reduce the number of ADDM Network sites.
Dr. Maenner argues that a comparison of the two protocols shows the new method doesn’t compromise accuracy and may actually capture children who lacked the medical or educational records the previous protocol required for a count. But not everyone agrees.
“I thought the point was to be as accurate and complete as possible in doing the surveillance,” Walter Zahorodny, PhD, associate professor of pediatrics at Rutgers University, New Brunswick, N.J., and principal investigator of the New Jersey ADDM Network site, told this news organization. “In states where there’s a high detail of information in records, like New Jersey, it’s going to underestimate the count.”
Dr. Zahorodny says the latest data prove his point. In 2016, under the old methodology, ASD prevalence was 3.1% in the state. In 2018, under the new protocol, prevalence was 2.84%, a decrease of about 20% that Dr. Zahorodny pins squarely on the elimination of ADDM clinical reviewers.
But New Jersey is the only state that participated in both the 2016 and 2018 surveillance periods to report a decrease in ASD prevalence. The other eight states all found autism rates in their states went up.
Sydney Pettygrove, PhD, associate professor of public health and pediatrics at the University of Arizona, Tucson, and a principal investigator for the ADDM site in Arizona, told this news organization that when she first learned the CDC was rolling out a new methodology, she and other investigators were concerned.
“People were really upset. I was really upset,” she said. “I had formed an opinion based on the earlier data that this would not be a good idea.”
In 2000, when ASD surveillance began in Arizona, nearly 30% of children identified by ADDM clinical reviewers as having autism had no mention of the disorder in their records. Today, that percentage is closer to 5%.
“In 2000 it would have been catastrophic to try to estimate the prevalence of autism with the new protocol,” said Dr. Pettygrove. As it turns out, under the new protocol, prevalence rates in Arizona increased from 16.0 per 1,000 children in 2016 to 24.9 in 2018.
Built-in bias eliminated?
In addition to speeding up the process, the new methodology might have other benefits as well. Under the old ADDM surveillance protocol, children who lacked certain medical or educational records did not meet the ASD case definition and weren’t counted.
A 2019 study showed that this disproportionately affected Black and Hispanic children, who had significantly less access to health care professionals than White children.
As a result, “the old methodology had a bias built into it,” Maureen Durkin, PhD, DrPH, coauthor of that study and chair of population health sciences at the University of Wisconsin–Madison and principal investigator for the ADDM site in Wisconsin, told this news organization.
“Clinician reviewers ended up putting these children in the ‘suspected ASD’ category because they couldn’t call it a case under the case definition,” Dr. Durkin said. “There was a fairly large percentage of suspected cases and a disproportionate number of those kids were children of color.”
Although she can’t say for sure, Dr. Durkin said it’s possible the new protocol could eliminate some of that bias.
CDC researchers also attribute the new method to an expanded study of early diagnosis among 4-year-olds. In previous years, only a handful of the ADDM Network sites participating in the 8-year-old surveillance project also studied early diagnosis in 4-year-olds.
This year, all 11 sites took part in the early diagnosis analysis, tripling the number of children included in the analysis. That made it possible to include, for the first time, Asian/Pacific Islander children in this analysis.
In the past, ASD prevalence has trended higher in White children, compared with other racial groups. The new data found that ASD prevalence among 4-year-olds was significantly lower in White children (12.9 per 1,000 children) than in Black, Hispanic, or Asian/Pacific Islander children (16.6, 21.1, and 22.7 per 1,000, respectively). Prevalence in American Indian/Alaska Native children was the lowest among all racial groups (11.5 per 1,000).
It’s the first time researchers have seen this pattern in any ADDM report, Kelly Shaw, PhD, lead author of that study and an epidemiologist with the National Center on Birth Defects and Developmental Disability at the CDC, told this news organization.
These data don’t provide clues about the potential cause of that disparity, Dr. Shaw said. It’s likely an indication of better identification of ASD in those communities, she said, and not a sign of increased incidence of autism among Black, Hispanic, or Asian/Pacific Islander children.
“We don’t have any evidence to suggest or expect that autism would be increasing differentially among groups,” Dr. Shaw said.
The data suggest “we are making some progress but there certainly is still room for improvement,” Dr. Shaw said.
Study authors report no conflicts of interest.
A version of this article first appeared on Medscape.com.
Childhood autism rates are at the highest level since the Centers for Disease Control and Prevention began tracking the disorder in 2000, new data released Dec. 2 show.
The increase likely reflects improvements in diagnosis and identification of autism spectrum disorder (ASD), not an increase in incidence, study authors with the CDC’s Autism and Developmental Disabilities Monitoring (ADDM) Network told this news organization.
Using a new surveillance methodology, researchers found that 2.3% of 8-year-olds in communities in 11 states across the United States had an autism diagnosis in 2018, up from 1.9% in 2016.
A separate report on early identification in 4-year-olds shows that children born in 2014 were 50% more likely to receive an autism diagnosis or ASD special education classification by 48 months of age than those born in 2010, signaling improved early diagnosis.
Taken together, the data suggest efforts to raise awareness about autism are working, though researchers were quick to say much work remains.
“It was not surprising to me and in fact it was reassuring that the number of children diagnosed with autism is higher and is actually approaching prevalence of autism that has been noted in some national surveys of parents,” Stuart Shapira, MD, PhD, associate director for science in CDC’s National Center on Birth Defects and Developmental Disability, told this news organization.
“It means we’re doing a better job of identifying children, which helps to get them into services earlier so they can achieve their best developmental outcome.”
The studies, published online in Morbidity and Mortality Weekly Report, are the first to use a new ASD surveillance protocol that relies on ASD diagnosis or special education classification and billing codes and eliminates comprehensive records analysis by trained clinician reviewers.
Racial disparities
The updated methodology was less labor intensive and reduced the time it took to produce the report, but it is not without its critics, who claim the new protocol will undercount the number of children with ASD.
Created in 2000 and funded by the CDC, the ADDM Network is the only surveillance program in the United States that tracks the number and characteristics of children with ASD in multiple communities in the U.S.
When ADDM released its first report in 2007 from six states and based on data from the year 2000, ASD prevalence was 6.7 per 1,000 children, or 1 in 150 children.
In the latest report, which includes data from 2018, the autism prevalence rate across 11 states was 23.0 per 1,000 children, or 1 in 44 children.
That rate is closer to reported autism prevalence from the National Survey of Children’s Health and the National Health Interview Survey, both of which rely on parent-reported ASD diagnoses.
in Arizona, Arkansas, California, Georgia, Maryland, Minnesota, Missouri, New Jersey, Tennessee, Utah, and Wisconsin.
Children were counted as having autism if their records included an ASD diagnosis, a special education classification of ASD, or an ASD International Classification of Diseases (ICD) code. A total of 5,058 children met those criteria.
Rates of ASD ranged from a low of 1.7% in Missouri to 3.9% in California and were 4.2 times higher in boys than in girls. Just under half of the children with ASD were evaluated by age 36 months.
Although the overall ASD prevalence was similar among White, Black, Hispanic, and Asian/Pacific Islander children, the report highlighted a number of other racial disparities overall and in individual states.
For example, among those with ASD and data on cognitive ability, 35.2% had an intelligence quotient score of 70 or lower. Black children with ASD were far more likely to have an IQ of 70 or less (49.8%) than Hispanic (33.1%) or White (29.7%) children.
“The persistent disparities in co-occurring intellectual disabilities in children with autism is something that we continue to see and suggests that we need to better understand exactly what’s happening,” Matthew Maenner, PhD, an epidemiologist and autism surveillance team lead with the CDC’s National Center on Birth Defects and Developmental Disabilities, told this news organization.
Another long-standing trend observed again in the new report on prevalence among 8-year-olds is low ASD prevalence among Hispanic children. While the overall estimate showed similar autism rates, a closer review of state-level data reveals a different picture.
“In almost half of the sites, Hispanic children were less likely to be identified as having ASD,” he said. “This gets lost if you look only at the overall estimate.”
New methodology
When ADDM released its first report in 2007, autism diagnosis was widely inadequate in the United States. Relying on only confirmed ASD diagnoses would significantly underestimate the number of children with the disorder, so the CDC added “active case finding” to the protocol.
Trained clinician reviewers analyzed individual notes from medical and educational records for every 8-year-old in ADDM Network sites, looking for evidence of characteristics and behaviors associated with autism. The process was labor- and time-intensive and took up to 4 years to complete.
In 2018, the CDC began investigating ways to speed the process and came up with the strategy used in the latest report. The new protocol was faster, easier, and less expensive. Although he says cost was never the deciding factor, Dr. Maenner acknowledges that had they stuck with the original protocol, they would have been forced to reduce the number of ADDM Network sites.
Dr. Maenner argues that a comparison of the two protocols shows the new method doesn’t compromise accuracy and may actually capture children who lacked the medical or educational records the previous protocol required for a count. But not everyone agrees.
“I thought the point was to be as accurate and complete as possible in doing the surveillance,” Walter Zahorodny, PhD, associate professor of pediatrics at Rutgers University, New Brunswick, N.J., and principal investigator of the New Jersey ADDM Network site, told this news organization. “In states where there’s a high detail of information in records, like New Jersey, it’s going to underestimate the count.”
Dr. Zahorodny says the latest data prove his point. In 2016, under the old methodology, ASD prevalence was 3.1% in the state. In 2018, under the new protocol, prevalence was 2.84%, a decrease of about 20% that Dr. Zahorodny pins squarely on the elimination of ADDM clinical reviewers.
But New Jersey is the only state that participated in both the 2016 and 2018 surveillance periods to report a decrease in ASD prevalence. The other eight states all found autism rates in their states went up.
Sydney Pettygrove, PhD, associate professor of public health and pediatrics at the University of Arizona, Tucson, and a principal investigator for the ADDM site in Arizona, told this news organization that when she first learned the CDC was rolling out a new methodology, she and other investigators were concerned.
“People were really upset. I was really upset,” she said. “I had formed an opinion based on the earlier data that this would not be a good idea.”
In 2000, when ASD surveillance began in Arizona, nearly 30% of children identified by ADDM clinical reviewers as having autism had no mention of the disorder in their records. Today, that percentage is closer to 5%.
“In 2000 it would have been catastrophic to try to estimate the prevalence of autism with the new protocol,” said Dr. Pettygrove. As it turns out, under the new protocol, prevalence rates in Arizona increased from 16.0 per 1,000 children in 2016 to 24.9 in 2018.
Built-in bias eliminated?
In addition to speeding up the process, the new methodology might have other benefits as well. Under the old ADDM surveillance protocol, children who lacked certain medical or educational records did not meet the ASD case definition and weren’t counted.
A 2019 study showed that this disproportionately affected Black and Hispanic children, who had significantly less access to health care professionals than White children.
As a result, “the old methodology had a bias built into it,” Maureen Durkin, PhD, DrPH, coauthor of that study and chair of population health sciences at the University of Wisconsin–Madison and principal investigator for the ADDM site in Wisconsin, told this news organization.
“Clinician reviewers ended up putting these children in the ‘suspected ASD’ category because they couldn’t call it a case under the case definition,” Dr. Durkin said. “There was a fairly large percentage of suspected cases and a disproportionate number of those kids were children of color.”
Although she can’t say for sure, Dr. Durkin said it’s possible the new protocol could eliminate some of that bias.
CDC researchers also attribute the new method to an expanded study of early diagnosis among 4-year-olds. In previous years, only a handful of the ADDM Network sites participating in the 8-year-old surveillance project also studied early diagnosis in 4-year-olds.
This year, all 11 sites took part in the early diagnosis analysis, tripling the number of children included in the analysis. That made it possible to include, for the first time, Asian/Pacific Islander children in this analysis.
In the past, ASD prevalence has trended higher in White children, compared with other racial groups. The new data found that ASD prevalence among 4-year-olds was significantly lower in White children (12.9 per 1,000 children) than in Black, Hispanic, or Asian/Pacific Islander children (16.6, 21.1, and 22.7 per 1,000, respectively). Prevalence in American Indian/Alaska Native children was the lowest among all racial groups (11.5 per 1,000).
It’s the first time researchers have seen this pattern in any ADDM report, Kelly Shaw, PhD, lead author of that study and an epidemiologist with the National Center on Birth Defects and Developmental Disability at the CDC, told this news organization.
These data don’t provide clues about the potential cause of that disparity, Dr. Shaw said. It’s likely an indication of better identification of ASD in those communities, she said, and not a sign of increased incidence of autism among Black, Hispanic, or Asian/Pacific Islander children.
“We don’t have any evidence to suggest or expect that autism would be increasing differentially among groups,” Dr. Shaw said.
The data suggest “we are making some progress but there certainly is still room for improvement,” Dr. Shaw said.
Study authors report no conflicts of interest.
A version of this article first appeared on Medscape.com.
Intranasal vs. intramuscular naloxone in reversing opioid overdose
Background: Naloxone is an opioid antagonist that works to treat opioid overdose. Few randomized trials have assessed the efficacy of intranasal administration, whereas more data have been published supporting use of intramuscular naloxone. This prospective trial examines the ability of the same dose (800 mcg per 1 mL solution) of intranasal naloxone vs. intramuscular naloxone at managing opioid overdose.
Study design: Double-blind double-dummy randomized clinical trial.
Setting: Single supervised injection center in Sydney.
Synopsis: In this study, 197 participants with opioid overdose were randomized to intramuscular or intranasal naloxone. If the patient did not respond to either (GSC score less than 13, RR less than 10, or oxygen saturation less than 95%), a rescue dose of intramuscular naloxone was given. Participants who received the intramuscular naloxone were less likely to need the rescue dose (8.6% vs. 23.1%; odds ratio, 0.35; P = .002). The time to achieve an RR greater than 10 (15 vs. 8 minutes) and GSC score greater than 13 (17 vs. 8 minutes) was longer in the intranasal than the intramuscular group. Limitations include the setting of a controlled environment. Also, this protocol called for an initial 5 minutes of ventilation prior to randomization, which selected for more severe overdose cases in the overall study population. More studies are needed to assess efficacy in the field, needlestick injuries, and larger intranasal doses.
Bottom line: Intranasal naloxone effectively reverses opioid overdose but not as effectively as intramuscular naloxone at the same dose.
Citation: Dietze P et al. Effect of intranasal vs intramuscular naloxone on opioid overdose: A randomized clinical trial. JAMA Netw Open. 2019;2:e1914977. doi: 1
Dr. Welter is a hospitalist at Northwestern Memorial Hospital and instructor of medicine, Feinberg School of Medicine, both in Chicago.
Background: Naloxone is an opioid antagonist that works to treat opioid overdose. Few randomized trials have assessed the efficacy of intranasal administration, whereas more data have been published supporting use of intramuscular naloxone. This prospective trial examines the ability of the same dose (800 mcg per 1 mL solution) of intranasal naloxone vs. intramuscular naloxone at managing opioid overdose.
Study design: Double-blind double-dummy randomized clinical trial.
Setting: Single supervised injection center in Sydney.
Synopsis: In this study, 197 participants with opioid overdose were randomized to intramuscular or intranasal naloxone. If the patient did not respond to either (GSC score less than 13, RR less than 10, or oxygen saturation less than 95%), a rescue dose of intramuscular naloxone was given. Participants who received the intramuscular naloxone were less likely to need the rescue dose (8.6% vs. 23.1%; odds ratio, 0.35; P = .002). The time to achieve an RR greater than 10 (15 vs. 8 minutes) and GSC score greater than 13 (17 vs. 8 minutes) was longer in the intranasal than the intramuscular group. Limitations include the setting of a controlled environment. Also, this protocol called for an initial 5 minutes of ventilation prior to randomization, which selected for more severe overdose cases in the overall study population. More studies are needed to assess efficacy in the field, needlestick injuries, and larger intranasal doses.
Bottom line: Intranasal naloxone effectively reverses opioid overdose but not as effectively as intramuscular naloxone at the same dose.
Citation: Dietze P et al. Effect of intranasal vs intramuscular naloxone on opioid overdose: A randomized clinical trial. JAMA Netw Open. 2019;2:e1914977. doi: 1
Dr. Welter is a hospitalist at Northwestern Memorial Hospital and instructor of medicine, Feinberg School of Medicine, both in Chicago.
Background: Naloxone is an opioid antagonist that works to treat opioid overdose. Few randomized trials have assessed the efficacy of intranasal administration, whereas more data have been published supporting use of intramuscular naloxone. This prospective trial examines the ability of the same dose (800 mcg per 1 mL solution) of intranasal naloxone vs. intramuscular naloxone at managing opioid overdose.
Study design: Double-blind double-dummy randomized clinical trial.
Setting: Single supervised injection center in Sydney.
Synopsis: In this study, 197 participants with opioid overdose were randomized to intramuscular or intranasal naloxone. If the patient did not respond to either (GSC score less than 13, RR less than 10, or oxygen saturation less than 95%), a rescue dose of intramuscular naloxone was given. Participants who received the intramuscular naloxone were less likely to need the rescue dose (8.6% vs. 23.1%; odds ratio, 0.35; P = .002). The time to achieve an RR greater than 10 (15 vs. 8 minutes) and GSC score greater than 13 (17 vs. 8 minutes) was longer in the intranasal than the intramuscular group. Limitations include the setting of a controlled environment. Also, this protocol called for an initial 5 minutes of ventilation prior to randomization, which selected for more severe overdose cases in the overall study population. More studies are needed to assess efficacy in the field, needlestick injuries, and larger intranasal doses.
Bottom line: Intranasal naloxone effectively reverses opioid overdose but not as effectively as intramuscular naloxone at the same dose.
Citation: Dietze P et al. Effect of intranasal vs intramuscular naloxone on opioid overdose: A randomized clinical trial. JAMA Netw Open. 2019;2:e1914977. doi: 1
Dr. Welter is a hospitalist at Northwestern Memorial Hospital and instructor of medicine, Feinberg School of Medicine, both in Chicago.











