User login
For MD-IQ use only
Tools—and rules—to support behavior change
Changing behavior is hard. And at nearly every clinical encounter, we counsel/encourage/remind/help (choose a verb) our patients to make a change—to do something hard. We tell them they need to increase their physical activity, get more sleep, or alter their eating habits. We know that if they make the needed changes, they can improve their health and possibly lengthen their lives. But we also know (from the systematic reviews the US Preventive Services Task Force [USPSTF] uses to make its recommendations) that brief counseling in our offices is largely ineffective unless we connect patients to resources to support the recommended change.
As examples, the USPSTF currently recommends the following (both grade “B”):
- offer or refer adults with cardiovascular disease risk factors to behavioral counseling interventions to promote a healthy diet and physical activity.1
- offer or refer adults with a body mass index of 30 or higher to intensive, multicomponent behavioral interventions.2
To support our patients when making recommendations such as these, we might refer them to a dietitian for intensive counseling and meal-planning guidance. The American Diabetes Association says that patients seeking to manage their diabetes and prediabetes “can start by working with a registered dietitian nutritionist … to make an eating plan that works for [them].”3 However, this kind of resource is unavailable to many of our patients.
So what else can we do?
We can help patients decide what to buy in the grocery aisle. Nutrition labels are useful, but they are limited by their complexity and requisite level of health literacy.4 Even the concept of “calories” is not so intuitive. This challenge with interpreting calories led me (in some of my prior work) to explore a potentially more useful approach: conveying calorie information as physical activity equivalents.5
In this issue of The Journal of Family Practice, Dong and colleagues present their findings on whether a simple equation (the Altman Rule) that uses information on nutrition labels may be a reasonable proxy for an even more difficult concept—glycemic load.6 The idea is that consumers (eg, patients with diabetes) can use this rule to help them in their decision-making at the grocery store (or the convenience store or gas station, for that matter, where the high-glycemic-load carbohydrates may be even more tempting). The 2-step rule is tech-free and can be applied in a few seconds. Their research demonstrated that the rule is a reasonable proxy for glycemic load for packaged carbohydrates (eg, chips, cereals, crackers, granola bars). Caveats acknowledged, foods that meet the rule are likely to be healthier choices.
Looking ahead, I would like to see whether counseling patients about the Altman Rule leads to their use of it, and how that translates into healthier eating, lower A1C, and ideally better health. For now, the Altman Rule is worth learning about. It may serve as another tool that you can use to support your patients when you ask them to do the hard work of making healthier food choices.
1. US Preventive Services Task Force. Behavioral counseling interventions to promote a healthy diet and physical activity for cardiovascular disease prevention in adults with cardiovascular risk factors: US Preventive Services Task Force recommendation statement. JAMA. 2020;324:2069-2075. doi: 10.1001/jama.2020.21749
2. US Preventive Services Task Force. Behavioral weight loss interventions to prevent obesity-related morbidity and mortality in adults: US Preventive Services Task Force recommendation statement. JAMA. 2018;320:1163-1171. doi: 10.1001/jama.2018.13022
3. American Diabetes Association. Eating right doesn’t have to be boring. Accessed August 23, 2023. diabetes.org/healthy-living/recipes-nutrition
4. Weiss BD, Mays MZ, Martz W, et al. Quick assessment of literacy in primary care: the newest vital sign. Ann Fam Med. 2005;3:514-522. doi: 10.1370/afm.405
5. Viera AJ, Gizlice Z, Tuttle L, et al. Effect of calories-only vs physical activity calorie expenditure labeling on lunch calories purchased in worksite cafeterias. BMC Public Health. 2019;19:107. doi: 10.1186/s12889-019-6433-x
6. Dong KR, Eustis S, Hawkins K, et al. Is the Altman Rule a proxy for glycemic load? J Fam Pract. 2023;72:286-291. doi: 10.12788/jfp.0656
Changing behavior is hard. And at nearly every clinical encounter, we counsel/encourage/remind/help (choose a verb) our patients to make a change—to do something hard. We tell them they need to increase their physical activity, get more sleep, or alter their eating habits. We know that if they make the needed changes, they can improve their health and possibly lengthen their lives. But we also know (from the systematic reviews the US Preventive Services Task Force [USPSTF] uses to make its recommendations) that brief counseling in our offices is largely ineffective unless we connect patients to resources to support the recommended change.
As examples, the USPSTF currently recommends the following (both grade “B”):
- offer or refer adults with cardiovascular disease risk factors to behavioral counseling interventions to promote a healthy diet and physical activity.1
- offer or refer adults with a body mass index of 30 or higher to intensive, multicomponent behavioral interventions.2
To support our patients when making recommendations such as these, we might refer them to a dietitian for intensive counseling and meal-planning guidance. The American Diabetes Association says that patients seeking to manage their diabetes and prediabetes “can start by working with a registered dietitian nutritionist … to make an eating plan that works for [them].”3 However, this kind of resource is unavailable to many of our patients.
So what else can we do?
We can help patients decide what to buy in the grocery aisle. Nutrition labels are useful, but they are limited by their complexity and requisite level of health literacy.4 Even the concept of “calories” is not so intuitive. This challenge with interpreting calories led me (in some of my prior work) to explore a potentially more useful approach: conveying calorie information as physical activity equivalents.5
In this issue of The Journal of Family Practice, Dong and colleagues present their findings on whether a simple equation (the Altman Rule) that uses information on nutrition labels may be a reasonable proxy for an even more difficult concept—glycemic load.6 The idea is that consumers (eg, patients with diabetes) can use this rule to help them in their decision-making at the grocery store (or the convenience store or gas station, for that matter, where the high-glycemic-load carbohydrates may be even more tempting). The 2-step rule is tech-free and can be applied in a few seconds. Their research demonstrated that the rule is a reasonable proxy for glycemic load for packaged carbohydrates (eg, chips, cereals, crackers, granola bars). Caveats acknowledged, foods that meet the rule are likely to be healthier choices.
Looking ahead, I would like to see whether counseling patients about the Altman Rule leads to their use of it, and how that translates into healthier eating, lower A1C, and ideally better health. For now, the Altman Rule is worth learning about. It may serve as another tool that you can use to support your patients when you ask them to do the hard work of making healthier food choices.
Changing behavior is hard. And at nearly every clinical encounter, we counsel/encourage/remind/help (choose a verb) our patients to make a change—to do something hard. We tell them they need to increase their physical activity, get more sleep, or alter their eating habits. We know that if they make the needed changes, they can improve their health and possibly lengthen their lives. But we also know (from the systematic reviews the US Preventive Services Task Force [USPSTF] uses to make its recommendations) that brief counseling in our offices is largely ineffective unless we connect patients to resources to support the recommended change.
As examples, the USPSTF currently recommends the following (both grade “B”):
- offer or refer adults with cardiovascular disease risk factors to behavioral counseling interventions to promote a healthy diet and physical activity.1
- offer or refer adults with a body mass index of 30 or higher to intensive, multicomponent behavioral interventions.2
To support our patients when making recommendations such as these, we might refer them to a dietitian for intensive counseling and meal-planning guidance. The American Diabetes Association says that patients seeking to manage their diabetes and prediabetes “can start by working with a registered dietitian nutritionist … to make an eating plan that works for [them].”3 However, this kind of resource is unavailable to many of our patients.
So what else can we do?
We can help patients decide what to buy in the grocery aisle. Nutrition labels are useful, but they are limited by their complexity and requisite level of health literacy.4 Even the concept of “calories” is not so intuitive. This challenge with interpreting calories led me (in some of my prior work) to explore a potentially more useful approach: conveying calorie information as physical activity equivalents.5
In this issue of The Journal of Family Practice, Dong and colleagues present their findings on whether a simple equation (the Altman Rule) that uses information on nutrition labels may be a reasonable proxy for an even more difficult concept—glycemic load.6 The idea is that consumers (eg, patients with diabetes) can use this rule to help them in their decision-making at the grocery store (or the convenience store or gas station, for that matter, where the high-glycemic-load carbohydrates may be even more tempting). The 2-step rule is tech-free and can be applied in a few seconds. Their research demonstrated that the rule is a reasonable proxy for glycemic load for packaged carbohydrates (eg, chips, cereals, crackers, granola bars). Caveats acknowledged, foods that meet the rule are likely to be healthier choices.
Looking ahead, I would like to see whether counseling patients about the Altman Rule leads to their use of it, and how that translates into healthier eating, lower A1C, and ideally better health. For now, the Altman Rule is worth learning about. It may serve as another tool that you can use to support your patients when you ask them to do the hard work of making healthier food choices.
1. US Preventive Services Task Force. Behavioral counseling interventions to promote a healthy diet and physical activity for cardiovascular disease prevention in adults with cardiovascular risk factors: US Preventive Services Task Force recommendation statement. JAMA. 2020;324:2069-2075. doi: 10.1001/jama.2020.21749
2. US Preventive Services Task Force. Behavioral weight loss interventions to prevent obesity-related morbidity and mortality in adults: US Preventive Services Task Force recommendation statement. JAMA. 2018;320:1163-1171. doi: 10.1001/jama.2018.13022
3. American Diabetes Association. Eating right doesn’t have to be boring. Accessed August 23, 2023. diabetes.org/healthy-living/recipes-nutrition
4. Weiss BD, Mays MZ, Martz W, et al. Quick assessment of literacy in primary care: the newest vital sign. Ann Fam Med. 2005;3:514-522. doi: 10.1370/afm.405
5. Viera AJ, Gizlice Z, Tuttle L, et al. Effect of calories-only vs physical activity calorie expenditure labeling on lunch calories purchased in worksite cafeterias. BMC Public Health. 2019;19:107. doi: 10.1186/s12889-019-6433-x
6. Dong KR, Eustis S, Hawkins K, et al. Is the Altman Rule a proxy for glycemic load? J Fam Pract. 2023;72:286-291. doi: 10.12788/jfp.0656
1. US Preventive Services Task Force. Behavioral counseling interventions to promote a healthy diet and physical activity for cardiovascular disease prevention in adults with cardiovascular risk factors: US Preventive Services Task Force recommendation statement. JAMA. 2020;324:2069-2075. doi: 10.1001/jama.2020.21749
2. US Preventive Services Task Force. Behavioral weight loss interventions to prevent obesity-related morbidity and mortality in adults: US Preventive Services Task Force recommendation statement. JAMA. 2018;320:1163-1171. doi: 10.1001/jama.2018.13022
3. American Diabetes Association. Eating right doesn’t have to be boring. Accessed August 23, 2023. diabetes.org/healthy-living/recipes-nutrition
4. Weiss BD, Mays MZ, Martz W, et al. Quick assessment of literacy in primary care: the newest vital sign. Ann Fam Med. 2005;3:514-522. doi: 10.1370/afm.405
5. Viera AJ, Gizlice Z, Tuttle L, et al. Effect of calories-only vs physical activity calorie expenditure labeling on lunch calories purchased in worksite cafeterias. BMC Public Health. 2019;19:107. doi: 10.1186/s12889-019-6433-x
6. Dong KR, Eustis S, Hawkins K, et al. Is the Altman Rule a proxy for glycemic load? J Fam Pract. 2023;72:286-291. doi: 10.12788/jfp.0656
Is low-dose naltrexone effective in chronic pain management?
Evidence summary
Naltrexone is comparable to amitriptyline for diabetic neuropathy pain
A 2021 randomized, double-blind, active-comparator, crossover clinical trial conducted in India examined the efficacy of low-dose naltrexone vs standard-of-care amitriptyline in patients (N = 67) with painful diabetic neuropathy. Participants were adults (ages 18 to 75 years) with painful diabetic neuropathy who had been on a stable dose of nonopioid pain medication for at least 1 month.1
Patients were randomly assigned to start receiving naltrexone 2 mg (n = 33) or amitriptyline 10 mg (n = 34). They received their starting medication for 6 weeks (with follow-up every 2 weeks), then completed a 2-week washout period, and then switched to the other study medication for 6 weeks (same follow-up schedule). If patients reported < 20% pain reduction on the Visual Analog Scale (VAS; 0-100 scoring system with 0 = no pain and 100 = worst pain) at a follow-up visit, their medication dose was titrated up, to a maximum of 4 mg of naltrexone or 25 to 50 mg of amitriptyline.1
The primary outcome of interest was the mean change in VAS pain score following 6 weeks of treatment. There was no statistically different change from baseline VAS pain score between the amitriptyline and naltrexone groups (mean difference [MD] = 1.6; 95% CI, –0.9 to 4.2; P = 0.21). These findings were consistent across the secondary endpoints (Likert 5-point pain scale and McGill Pain Questionnaire scores). There was no statistically significant difference in Hamilton Depression Rating Scale scores (13 in the naltrexone group vs 11 in the amitriptyline group; P = .81), no reports of decreased sleep quality in either group, and no significant difference in Patients’ Global Impression of Change scores at 6-week evaluation.1
The naltrexone cohort experienced 8 adverse events (most commonly, mild diarrhea), while the amitriptyline cohort experienced 52 adverse events (most commonly, somnolence) (P < .001). The limitations of the study include the lack of a placebo arm and a relatively small sample size.1
Greater reduction in pain scores with naltrexone
A 2022 retrospective cohort study evaluated the effectiveness of naltrexone for patients treated at a single outpatient integrative pain management practice in Alaska between 2014 and 2019. The exposure group (n = 36) included patients who had completed at least a 2-month continuous regimen of oral naltrexone 4.5 mg. Controls (n = 42) were selected from the remaining practice population receiving standard care and were primarily matched by diagnosis code, followed by gender, then age +/– 5 years. Patients were divided into subgroups for inflammatory and neuropathic pain.2
The primary outcome measured was the mean change in VAS score or numeric rating score (NRS; both used a 1-10 rating system), which was assessed during a patient’s appointment from initiation of treatment to the most recent visit or at the termination of therapy (intervention interquartile range, 12-14 months). There was no statistically significant difference in VAS/NRS between the low-dose naltrexone and control groups at baseline (6.09 vs 6.38; P = .454). The low-dose naltrexone group experienced a greater reduction in VAS/NRS pain scores compared to the control group (–37.8% vs –4.3%; P < .001).2
Compared with control patients in each group, patients in the inflammatory pain subgroup and the neuropathic pain subgroup who received low-dose naltrexone reported reductions in pain scores of 32% (P < .001) and 44% (P = .048), respectively. There was no statistically significant difference in mean change in VAS/NRS scores between the inflammatory and neuropathic subgroups (P = .763). A multivariate linear regression analysis did not identify significant variables other than low-dose naltrexone that correlated with pain improvement. The number needed to treat to observe a ≥ 50% reduction in pain scores was 3.2.2
Continue to: Limitations for this study...
Limitations for this study include its small sample size and open-label design.2
Low-dose naltrexone is effective for fibromyalgia pain
A 2020 single-blind prospective dose-response study utilized the up-and-down method to identify effective naltrexone dose for patients in a Danish university hospital pain clinic. Patients were White women ages 18 to 60 years (N = 25) who had a diagnosis of fibromyalgia unresponsive to traditional pharmacologic treatment. All patients received treatment with low-dose naltrexone (ranging from 0.75 mg to 6.0 mg) but were blinded to dose.3
Patients were evaluated for improvement in fibromyalgia symptoms using the Patient Global Impression of Improvement (PGI-I) scale—which ranges from 1 (very much improved) to 7 (very much worse), with 4 being “no change”—at baseline and after 2 to 3 weeks of treatment with low-dose naltrexone. A patient was considered a responder if they scored 1 to 3 on the follow-up PGI-I scale or if they experienced a > 30% pain reduction on the VAS. If a patient did not respond to their dose, the next patient began treatment at a dose 0.75 mg higher than the previous patient’s ending dose. If a patient did respond to low-dose naltrexone treatment, the next patient’s starting dose was 0.75 mg less than the previous patient’s. Eleven of 25 patients were considered responders.3
The primary outcomes were effective dose for 50% of fibromyalgia patients (3.88 mg; 95% CI, 3.39-4.35) and effective dose for 95% of fibromyalgia patients (5.4 mg; 95% CI, 4.66-6.13). Secondary outcomes were fibromyalgia symptoms as evaluated on the Fibromyalgia Impact Questionnaire Revised. Five of the 11 responders reported a > 30% improvement in tenderness and 8 of the 11 responders reported a > 30% decrease in waking unrefreshed.3
Limitations of the study include the short time period of treatment before response was assessed and the decision to use low test doses, which may have hindered detection of effective doses > 6 mg in fibromyalgia.3
Editor’s takeaway
Low-dose naltrexone, a less-often-used form of pain management, is a welcome option. Studies show some effectiveness in a variety of pain conditions with few adverse effects. The small number of studies, the small sample sizes, and the limited follow-up duration should encourage more investigation into how to best use this intervention.
1. Srinivasan A, Dutta P, Bansal D, et al. Efficacy and safety of low-dose naltrexone in painful diabetic neuropathy: a randomized, double-blind, active-control, crossover clinical trial. J Diabetes. 2021;13:770-778. doi: 10.1111/1753-0407.13202
2. Martin SJ, McAnally HB, Okediji P, et al. Low-dose naltrexone, an opioid-receptor antagonist, is a broad-spectrum analgesic: a retrospective cohort study. Pain Management. 2022;12:699-709. doi: 10.2217/pmt-2021-0122
3. Bruun-Plesner K, Blichfeldt-Eckhardt MR, Vaegter HB, et al. Low-dose naltrexone for the treatment of fibromyalgia: investigation of dose-response relationships. Pain Med. 2020;21:2253-2261. doi: 10.1093/pm/pnaa001
Evidence summary
Naltrexone is comparable to amitriptyline for diabetic neuropathy pain
A 2021 randomized, double-blind, active-comparator, crossover clinical trial conducted in India examined the efficacy of low-dose naltrexone vs standard-of-care amitriptyline in patients (N = 67) with painful diabetic neuropathy. Participants were adults (ages 18 to 75 years) with painful diabetic neuropathy who had been on a stable dose of nonopioid pain medication for at least 1 month.1
Patients were randomly assigned to start receiving naltrexone 2 mg (n = 33) or amitriptyline 10 mg (n = 34). They received their starting medication for 6 weeks (with follow-up every 2 weeks), then completed a 2-week washout period, and then switched to the other study medication for 6 weeks (same follow-up schedule). If patients reported < 20% pain reduction on the Visual Analog Scale (VAS; 0-100 scoring system with 0 = no pain and 100 = worst pain) at a follow-up visit, their medication dose was titrated up, to a maximum of 4 mg of naltrexone or 25 to 50 mg of amitriptyline.1
The primary outcome of interest was the mean change in VAS pain score following 6 weeks of treatment. There was no statistically different change from baseline VAS pain score between the amitriptyline and naltrexone groups (mean difference [MD] = 1.6; 95% CI, –0.9 to 4.2; P = 0.21). These findings were consistent across the secondary endpoints (Likert 5-point pain scale and McGill Pain Questionnaire scores). There was no statistically significant difference in Hamilton Depression Rating Scale scores (13 in the naltrexone group vs 11 in the amitriptyline group; P = .81), no reports of decreased sleep quality in either group, and no significant difference in Patients’ Global Impression of Change scores at 6-week evaluation.1
The naltrexone cohort experienced 8 adverse events (most commonly, mild diarrhea), while the amitriptyline cohort experienced 52 adverse events (most commonly, somnolence) (P < .001). The limitations of the study include the lack of a placebo arm and a relatively small sample size.1
Greater reduction in pain scores with naltrexone
A 2022 retrospective cohort study evaluated the effectiveness of naltrexone for patients treated at a single outpatient integrative pain management practice in Alaska between 2014 and 2019. The exposure group (n = 36) included patients who had completed at least a 2-month continuous regimen of oral naltrexone 4.5 mg. Controls (n = 42) were selected from the remaining practice population receiving standard care and were primarily matched by diagnosis code, followed by gender, then age +/– 5 years. Patients were divided into subgroups for inflammatory and neuropathic pain.2
The primary outcome measured was the mean change in VAS score or numeric rating score (NRS; both used a 1-10 rating system), which was assessed during a patient’s appointment from initiation of treatment to the most recent visit or at the termination of therapy (intervention interquartile range, 12-14 months). There was no statistically significant difference in VAS/NRS between the low-dose naltrexone and control groups at baseline (6.09 vs 6.38; P = .454). The low-dose naltrexone group experienced a greater reduction in VAS/NRS pain scores compared to the control group (–37.8% vs –4.3%; P < .001).2
Compared with control patients in each group, patients in the inflammatory pain subgroup and the neuropathic pain subgroup who received low-dose naltrexone reported reductions in pain scores of 32% (P < .001) and 44% (P = .048), respectively. There was no statistically significant difference in mean change in VAS/NRS scores between the inflammatory and neuropathic subgroups (P = .763). A multivariate linear regression analysis did not identify significant variables other than low-dose naltrexone that correlated with pain improvement. The number needed to treat to observe a ≥ 50% reduction in pain scores was 3.2.2
Continue to: Limitations for this study...
Limitations for this study include its small sample size and open-label design.2
Low-dose naltrexone is effective for fibromyalgia pain
A 2020 single-blind prospective dose-response study utilized the up-and-down method to identify effective naltrexone dose for patients in a Danish university hospital pain clinic. Patients were White women ages 18 to 60 years (N = 25) who had a diagnosis of fibromyalgia unresponsive to traditional pharmacologic treatment. All patients received treatment with low-dose naltrexone (ranging from 0.75 mg to 6.0 mg) but were blinded to dose.3
Patients were evaluated for improvement in fibromyalgia symptoms using the Patient Global Impression of Improvement (PGI-I) scale—which ranges from 1 (very much improved) to 7 (very much worse), with 4 being “no change”—at baseline and after 2 to 3 weeks of treatment with low-dose naltrexone. A patient was considered a responder if they scored 1 to 3 on the follow-up PGI-I scale or if they experienced a > 30% pain reduction on the VAS. If a patient did not respond to their dose, the next patient began treatment at a dose 0.75 mg higher than the previous patient’s ending dose. If a patient did respond to low-dose naltrexone treatment, the next patient’s starting dose was 0.75 mg less than the previous patient’s. Eleven of 25 patients were considered responders.3
The primary outcomes were effective dose for 50% of fibromyalgia patients (3.88 mg; 95% CI, 3.39-4.35) and effective dose for 95% of fibromyalgia patients (5.4 mg; 95% CI, 4.66-6.13). Secondary outcomes were fibromyalgia symptoms as evaluated on the Fibromyalgia Impact Questionnaire Revised. Five of the 11 responders reported a > 30% improvement in tenderness and 8 of the 11 responders reported a > 30% decrease in waking unrefreshed.3
Limitations of the study include the short time period of treatment before response was assessed and the decision to use low test doses, which may have hindered detection of effective doses > 6 mg in fibromyalgia.3
Editor’s takeaway
Low-dose naltrexone, a less-often-used form of pain management, is a welcome option. Studies show some effectiveness in a variety of pain conditions with few adverse effects. The small number of studies, the small sample sizes, and the limited follow-up duration should encourage more investigation into how to best use this intervention.
Evidence summary
Naltrexone is comparable to amitriptyline for diabetic neuropathy pain
A 2021 randomized, double-blind, active-comparator, crossover clinical trial conducted in India examined the efficacy of low-dose naltrexone vs standard-of-care amitriptyline in patients (N = 67) with painful diabetic neuropathy. Participants were adults (ages 18 to 75 years) with painful diabetic neuropathy who had been on a stable dose of nonopioid pain medication for at least 1 month.1
Patients were randomly assigned to start receiving naltrexone 2 mg (n = 33) or amitriptyline 10 mg (n = 34). They received their starting medication for 6 weeks (with follow-up every 2 weeks), then completed a 2-week washout period, and then switched to the other study medication for 6 weeks (same follow-up schedule). If patients reported < 20% pain reduction on the Visual Analog Scale (VAS; 0-100 scoring system with 0 = no pain and 100 = worst pain) at a follow-up visit, their medication dose was titrated up, to a maximum of 4 mg of naltrexone or 25 to 50 mg of amitriptyline.1
The primary outcome of interest was the mean change in VAS pain score following 6 weeks of treatment. There was no statistically different change from baseline VAS pain score between the amitriptyline and naltrexone groups (mean difference [MD] = 1.6; 95% CI, –0.9 to 4.2; P = 0.21). These findings were consistent across the secondary endpoints (Likert 5-point pain scale and McGill Pain Questionnaire scores). There was no statistically significant difference in Hamilton Depression Rating Scale scores (13 in the naltrexone group vs 11 in the amitriptyline group; P = .81), no reports of decreased sleep quality in either group, and no significant difference in Patients’ Global Impression of Change scores at 6-week evaluation.1
The naltrexone cohort experienced 8 adverse events (most commonly, mild diarrhea), while the amitriptyline cohort experienced 52 adverse events (most commonly, somnolence) (P < .001). The limitations of the study include the lack of a placebo arm and a relatively small sample size.1
Greater reduction in pain scores with naltrexone
A 2022 retrospective cohort study evaluated the effectiveness of naltrexone for patients treated at a single outpatient integrative pain management practice in Alaska between 2014 and 2019. The exposure group (n = 36) included patients who had completed at least a 2-month continuous regimen of oral naltrexone 4.5 mg. Controls (n = 42) were selected from the remaining practice population receiving standard care and were primarily matched by diagnosis code, followed by gender, then age +/– 5 years. Patients were divided into subgroups for inflammatory and neuropathic pain.2
The primary outcome measured was the mean change in VAS score or numeric rating score (NRS; both used a 1-10 rating system), which was assessed during a patient’s appointment from initiation of treatment to the most recent visit or at the termination of therapy (intervention interquartile range, 12-14 months). There was no statistically significant difference in VAS/NRS between the low-dose naltrexone and control groups at baseline (6.09 vs 6.38; P = .454). The low-dose naltrexone group experienced a greater reduction in VAS/NRS pain scores compared to the control group (–37.8% vs –4.3%; P < .001).2
Compared with control patients in each group, patients in the inflammatory pain subgroup and the neuropathic pain subgroup who received low-dose naltrexone reported reductions in pain scores of 32% (P < .001) and 44% (P = .048), respectively. There was no statistically significant difference in mean change in VAS/NRS scores between the inflammatory and neuropathic subgroups (P = .763). A multivariate linear regression analysis did not identify significant variables other than low-dose naltrexone that correlated with pain improvement. The number needed to treat to observe a ≥ 50% reduction in pain scores was 3.2.2
Continue to: Limitations for this study...
Limitations for this study include its small sample size and open-label design.2
Low-dose naltrexone is effective for fibromyalgia pain
A 2020 single-blind prospective dose-response study utilized the up-and-down method to identify effective naltrexone dose for patients in a Danish university hospital pain clinic. Patients were White women ages 18 to 60 years (N = 25) who had a diagnosis of fibromyalgia unresponsive to traditional pharmacologic treatment. All patients received treatment with low-dose naltrexone (ranging from 0.75 mg to 6.0 mg) but were blinded to dose.3
Patients were evaluated for improvement in fibromyalgia symptoms using the Patient Global Impression of Improvement (PGI-I) scale—which ranges from 1 (very much improved) to 7 (very much worse), with 4 being “no change”—at baseline and after 2 to 3 weeks of treatment with low-dose naltrexone. A patient was considered a responder if they scored 1 to 3 on the follow-up PGI-I scale or if they experienced a > 30% pain reduction on the VAS. If a patient did not respond to their dose, the next patient began treatment at a dose 0.75 mg higher than the previous patient’s ending dose. If a patient did respond to low-dose naltrexone treatment, the next patient’s starting dose was 0.75 mg less than the previous patient’s. Eleven of 25 patients were considered responders.3
The primary outcomes were effective dose for 50% of fibromyalgia patients (3.88 mg; 95% CI, 3.39-4.35) and effective dose for 95% of fibromyalgia patients (5.4 mg; 95% CI, 4.66-6.13). Secondary outcomes were fibromyalgia symptoms as evaluated on the Fibromyalgia Impact Questionnaire Revised. Five of the 11 responders reported a > 30% improvement in tenderness and 8 of the 11 responders reported a > 30% decrease in waking unrefreshed.3
Limitations of the study include the short time period of treatment before response was assessed and the decision to use low test doses, which may have hindered detection of effective doses > 6 mg in fibromyalgia.3
Editor’s takeaway
Low-dose naltrexone, a less-often-used form of pain management, is a welcome option. Studies show some effectiveness in a variety of pain conditions with few adverse effects. The small number of studies, the small sample sizes, and the limited follow-up duration should encourage more investigation into how to best use this intervention.
1. Srinivasan A, Dutta P, Bansal D, et al. Efficacy and safety of low-dose naltrexone in painful diabetic neuropathy: a randomized, double-blind, active-control, crossover clinical trial. J Diabetes. 2021;13:770-778. doi: 10.1111/1753-0407.13202
2. Martin SJ, McAnally HB, Okediji P, et al. Low-dose naltrexone, an opioid-receptor antagonist, is a broad-spectrum analgesic: a retrospective cohort study. Pain Management. 2022;12:699-709. doi: 10.2217/pmt-2021-0122
3. Bruun-Plesner K, Blichfeldt-Eckhardt MR, Vaegter HB, et al. Low-dose naltrexone for the treatment of fibromyalgia: investigation of dose-response relationships. Pain Med. 2020;21:2253-2261. doi: 10.1093/pm/pnaa001
1. Srinivasan A, Dutta P, Bansal D, et al. Efficacy and safety of low-dose naltrexone in painful diabetic neuropathy: a randomized, double-blind, active-control, crossover clinical trial. J Diabetes. 2021;13:770-778. doi: 10.1111/1753-0407.13202
2. Martin SJ, McAnally HB, Okediji P, et al. Low-dose naltrexone, an opioid-receptor antagonist, is a broad-spectrum analgesic: a retrospective cohort study. Pain Management. 2022;12:699-709. doi: 10.2217/pmt-2021-0122
3. Bruun-Plesner K, Blichfeldt-Eckhardt MR, Vaegter HB, et al. Low-dose naltrexone for the treatment of fibromyalgia: investigation of dose-response relationships. Pain Med. 2020;21:2253-2261. doi: 10.1093/pm/pnaa001
EVIDENCE-BASED ANSWER:
YES. Low-dose naltrexone is as effective as amitriptyline in the treatment of painful diabetic neuropathy and has a superior safety profile (strength of recommendation [SOR], B; single randomized controlled trial [RCT]).
Low-dose naltrexone significantly reduced pain by 32% in inflammatory conditions and 44% in neuropathic conditions (SOR, B; single retrospective cohort study).
Doses as low as 5.4 mg were found to reduce pain in 95% of patients with fibromyalgia (SOR, B; single prospective dose-response study).
School avoidance: How to help when a child refuses to go
THE CASE
Juana*, a 10-year-old who identifies as a cisgender, Hispanic female, was referred to our integrated behavioral health program by her primary care physician. Her mother was concerned because Juana had been refusing to attend school due to complaints of gastrointestinal upset. This concern began when Juana was in first grade but had increased in severity over the past few months.
Upon further questioning, the patient reported that she initially did not want to attend school due to academic difficulties and bullying. However, since COVID-19, her fears of attending school had significantly worsened. Juana’s mother’s primary language was Spanish and she had limited English proficiency; she reported difficulty communicating with school personnel about Juana’s poor attendance.
Juana had recently had a complete medical work-up for her gastrointestinal concerns, with negative results. Since the negative work-up, Juana’s mother had told her daughter that she would be punished if she didn’t go to school.
●
* The patient’s name has been changed to protect her identity.
School avoidance, also referred to as school refusal, is a symptom of an emotional condition that manifests as a child refusing to go to school or having difficulty going to school or remaining in the classroom for the entire day. School avoidance is not a clinical diagnosis but often is related to an underlying disorder.1
School avoidance is common, affecting 5% to 28% of youth sometime in their school career.2 Available data are not specific to school avoidance but focus on chronic absenteeism (missing ≥ 15 days per school year). Rates of chronic absenteeism are high in elementary and middle school (about 14% each) and tend to increase in high school (about 21%).3 Students with disabilities are 1.5 times more likely to be chronically absent than students without disabilities.3 Compared to White students, American Indian and Pacific Islander students are > 50% more likely, Black students 40% more likely, and Hispanic students 17% more likely to miss ≥ 3 weeks of school.3 Rates of chronic absenteeism are similar (about 16%) for males and females.3
Absenteeism can have immediate and long-term negative effects.4 School attendance issues are correlated to negative life outcomes, such as delinquency, teen pregnancy, substance use, and poor academic achievement.5 According to the US Department of Education, individuals who chronically miss school are less likely to achieve educational milestones (particularly in younger years) and may be more likely to drop out of school.3
What school avoidance is (and what it isn’t)
It is important to distinguish school avoidance from truancy. Truancy often is associated with antisocial behavior such as lying and stealing, while school avoidance occurs in the absence of significant antisocial disorders.6 With truancy, the absence usually is hidden from the parent. In contrast, with school avoidance, the parents usually know where their child is; the child often spends the day secluded in their bedroom. Students who engage in truancy do not demonstrate excessive anxiety about attending school but may have decreased interest in schoolwork and academic performance.6 With school avoidance, the child exhibits severe emotional distress about attending school but is willing to complete schoolwork at home.
Why children may avoid school
School avoidance is a biopsychosocial condition with a multitude of underlying causes.4 It is associated most commonly with anxiety disorders and neurodevelopmental disorders, including but not limited to learning disabilities and attention-deficit/hyperactivity disorder.1 Depressive disorders also have been associated with school avoidance.7 Social concerns related to changes with school personnel or classes, academic challenges, bullying, health emergencies, and family stressors also can result in symptoms of school avoidance.1
Continue to: A child seeking to avoid...
A child seeking to avoid school may be motivated by potential negative and/or positive effects of doing so. Kearney and Silverman8 identified 4 primary functions of school refusal behaviors:
- avoiding stimuli at school that lend to negative affect (depression, anxiety)
- escaping the social interactions and/or situations for evaluation that occur at school
- gaining more attention from caregivers, and
- obtaining tangible rewards or benefits outside the school environment.
How school avoidance manifests
School avoidance has attributes of internalizing (depression, anxiety, somatic complaints) and externalizing (aggression, tantrums, running away, clinginess) behaviors. It can cause distress for the student, parents and caregivers, and school personnel.
The avoidance may manifest with behaviors such as crying, hiding, emotional outbursts, and refusing to move prior to the start of the school day. Additionally, the child may beg their parents not to make them go to school or, when at school, they may leave the classroom to go to a safe place such as the nurse’s or counselor’s office.
The avoidance may occur abruptly, such as after a break in the school schedule or a change of school. Or it may be the final result of the student’s gradual inability to cope with the underlying issue.
How to assess for school avoidance
Due to the multifactorial nature of this presenting concern, a comprehensive evaluation is recommended when school avoidance is reported.4 Often the child will present with physical symptoms, such as abdominal pain, nausea, vomiting, diarrhea, headaches, shortness of breath, dizziness, chest pain, and palpitations. A thorough medical examination should be performed to rule out a physiological cause. The medical visit should include clinical interviews with the patient and family members or guardians.
Continue to: To identify school avoidance...
To identify school avoidance in pediatric and adolescent populations, medical history and physical examination—along with social history to better understand familial, social, and academic concerns—should be a regular part of the medical encounter. The School Refusal Assessment Scale-Revised (SRAS-R) for both parents and their children was developed to assess for school avoidance and can be utilized within the primary care setting. Additional psychiatric history for both the family and patient may be beneficial, due to associations between parental mental health concerns and school avoidance in their children.9,10
Assessment for an underlying mental health condition, such as an anxiety or depressive disorder, should be completed when a patient presents with school avoidance.4 More than one-third of children with behavioral problems, such as school avoidance, have been diagnosed with anxiety.11 The 2020 National Survey of Children’s Health found that 7.8% of children and adolescents ages 3 to 17 years had a current anxiety disorder, leading the US Preventive Services Task Force to recommend screening for anxiety in children and adolescents ages 8 to 18 years.12,13 Furthermore, if academic achievement is of concern, then consideration of further assessment for neurodevelopmental disorders is warranted.1
Treatment is multimodal and multidisciplinary
Treatment for school avoidance is often multimodal and may involve interdisciplinary, team-based care including the medical provider, school system (eg, Child Study Team), family, and mental health care provider.1,4
Cognitive behavioral therapy (CBT) is the most-studied intervention for school avoidance, with behavioral, exposure-based interventions often central to therapeutic gains in treatment.1,14,15 The goals of treatment are to increase school attendance while decreasing emotional distress through various strategies, including exposure-based interventions, contingency management with parents and school staff, relaxation training, and/or social skills training.14,16 Collaborative involvement between the medical provider and the school system is key to successful treatment.
Medication may be considered alone or in combination with CBT when comorbid mental health conditions have been identified. Selective serotonin reuptake inhibitors (SSRIs)—including fluoxetine, sertraline, and escitalopram—are considered first-line treatment for anxiety in children and adolescents.17 Serotonin-norepinephrine reuptake inhibitors (SNRIs), such as duloxetine and venlafaxine, also have been shown to be effective. Duloxetine is the only medication approved by the US Food and Drug Administration (FDA) for treatment of generalized anxiety disorder in children ages 7 years and older.17
Continue to: SSRIs and SNRIs have a boxed warning...
SSRIs and SNRIs have a boxed warning from the FDA for increased suicidal thoughts and behaviors in children and adolescents. Although this risk is rare, it should be discussed with the patient and parent/guardian in order to obtain informed consent prior to treatment initiation.
Medication should be started at the lowest possible dose and increased gradually. Patients should remain on the medication for 6 to 12 months after symptom resolution and should be tapered during a nonstressful time, such as the summer break.
THE CASE
Based on the concerns of continued school refusal after negative gastrointestinal work-up, Juana’s physician screened her for anxiety and conducted a clinical interview to better understand any psychosocial concerns. Juana’s score of 10 on the General Anxiety Disorder-7 scale indicated moderate anxiety. She reported symptoms consistent with social anxiety disorder contributing to school avoidance.
The physician consulted with the clinic’s behavioral health consultant (BHC) to confirm the multimodal treatment plan, which was then discussed with Juana and her mother. The physician discussed medication options (SSRIs) and provided documentation (in both English and Spanish) from the visit to Juana’s mother so she could initiate a school-based intervention with the Child Study Team at Juana’s school. A plan for CBT—including a collaborative contingency management plan between the patient and her parent (eg, a reward chart for attending school) and exposure interventions (eg, a graduated plan to participate in school-based activities with the end goal to resume full school attendance)—was developed with the BHC. Biweekly follow-up appointments were scheduled with the BHC and monthly appointments were scheduled with the physician to reinforce the interventions.
CORRESPONDENCE
Meredith L. C. Williamson, PhD, 2900 East 29th Street, Suite 100, Bryan, TX 77840; meredith.williamson@tamu.edu
1. School Avoidance Alliance. School avoidance facts. Published September 16, 2021. Accessed July 27, 2023. https://schoolavoidance.org/school-avoidance-facts/
2. Kearney CA. School Refusal Behavior in Youth: A Functional Approach to Assessment and Treatment. American Psychological Association; 2001.
3. US Department of Education. Chronic absenteeism in the nation’s schools: a hidden educational crisis. Updated January 2019. Accessed August 3, 2023. www2.ed.gov/datastory/chronicabsenteeism.html
4. Allen CW, Diamond-Myrsten S, Rollins LK. School absenteeism in children and adolescents. Am Fam Physician. 2018;98:738-744.
5. Gonzálvez C, Díaz-Herrero Á, Vicent M, et al. School refusal behavior: latent class analysis approach and its relationship with psychopathological symptoms. Curr Psychology. 2022;41:2078-2088. doi: 10.1007/s12144-020-00711-6
6. Fremont WP. School refusal in children and adolescents. Am Fam Physician. 2003;68:1555-1560.
7. McShane G, Walter G, Rey JM. Characteristics of adolescents with school refusal. Aust N Z J Psychiatry. 2001;35:822-826. doi: 10.1046/j.1440-1614.2001.00955.x
8. Kearney CA, Silverman WK. The evolution and reconciliation of taxonomic strategies for school refusal behavior. Clin Psychology Sci Pract. 1996;3:339-354. doi: 10.1111/j.1468-2850.1996.tb00087.x
9. Kearney CA, Albano AM. School Refusal Assessment Scale-Revised C. Oxford University Press; 2007.
10. Heyne D. School refusal. In: Fisher JE, O’Donohue WT (eds). Practitioner’s Guide to Evidence-based Psychotherapy. Springer Science + Business Media. 2006;600-619. doi: 10.1007/978-0-387-28370-8_60
11. Ghandour RM, Sherman LJ, Vladutiu CJ, et al. Prevalence and treatment of depression, anxiety, and conduct problems in US children. J Pediatrics. 2019;206:256-267.e3. doi: 10.1016/j.jpeds.2018.09.021
12. US Census Bureau. 2020 National Survey of Children’s Health: Topical Frequencies. Published June 2, 2021. Accessed August 4, 2023. www2.census.gov/programs-surveys/nsch/technical-documentation/codebook/NSCH_2020_Topical_Frequencies.pdf
13. USPSTF. Anxiety in children and adolescents: screening. Final Recommendation Statement. Published October 11, 2022. Accessed August 4, 2023. www.uspreventiveservicestaskforce.org/uspstf/recommendation/screening-anxiety-children-adolescents
14. Maynard BR, Brendel KE, Bulanda JJ, et al. Psychosocial interventions for school refusal with primary and secondary school students: a systematic review. Campbell Systematic Rev. 2015;11:1-76. doi: 10.4073/csr.2015.12
15. Kearney CA, Albano AM. When Children Refuse School: Parent Workbook. 3rd ed. Oxford University Press; 2018. doi: 10.1093/med-psych/9780190604080.001.0001
16. Heyne DA, Sauter FM. School refusal. In: Essau CA, Ollendick TH. The Wiley-Blackwell Handbook of the Treatment of Childhood and Adolescent Anxiety. Wiley Blackwell; 2013:471-517.
17. Kowalchuk A, Gonzalez SJ, Zoorob RJ. Anxiety disorders in children and adolescents. Am Fam Physician. 2022;106:657-664.
THE CASE
Juana*, a 10-year-old who identifies as a cisgender, Hispanic female, was referred to our integrated behavioral health program by her primary care physician. Her mother was concerned because Juana had been refusing to attend school due to complaints of gastrointestinal upset. This concern began when Juana was in first grade but had increased in severity over the past few months.
Upon further questioning, the patient reported that she initially did not want to attend school due to academic difficulties and bullying. However, since COVID-19, her fears of attending school had significantly worsened. Juana’s mother’s primary language was Spanish and she had limited English proficiency; she reported difficulty communicating with school personnel about Juana’s poor attendance.
Juana had recently had a complete medical work-up for her gastrointestinal concerns, with negative results. Since the negative work-up, Juana’s mother had told her daughter that she would be punished if she didn’t go to school.
●
* The patient’s name has been changed to protect her identity.
School avoidance, also referred to as school refusal, is a symptom of an emotional condition that manifests as a child refusing to go to school or having difficulty going to school or remaining in the classroom for the entire day. School avoidance is not a clinical diagnosis but often is related to an underlying disorder.1
School avoidance is common, affecting 5% to 28% of youth sometime in their school career.2 Available data are not specific to school avoidance but focus on chronic absenteeism (missing ≥ 15 days per school year). Rates of chronic absenteeism are high in elementary and middle school (about 14% each) and tend to increase in high school (about 21%).3 Students with disabilities are 1.5 times more likely to be chronically absent than students without disabilities.3 Compared to White students, American Indian and Pacific Islander students are > 50% more likely, Black students 40% more likely, and Hispanic students 17% more likely to miss ≥ 3 weeks of school.3 Rates of chronic absenteeism are similar (about 16%) for males and females.3
Absenteeism can have immediate and long-term negative effects.4 School attendance issues are correlated to negative life outcomes, such as delinquency, teen pregnancy, substance use, and poor academic achievement.5 According to the US Department of Education, individuals who chronically miss school are less likely to achieve educational milestones (particularly in younger years) and may be more likely to drop out of school.3
What school avoidance is (and what it isn’t)
It is important to distinguish school avoidance from truancy. Truancy often is associated with antisocial behavior such as lying and stealing, while school avoidance occurs in the absence of significant antisocial disorders.6 With truancy, the absence usually is hidden from the parent. In contrast, with school avoidance, the parents usually know where their child is; the child often spends the day secluded in their bedroom. Students who engage in truancy do not demonstrate excessive anxiety about attending school but may have decreased interest in schoolwork and academic performance.6 With school avoidance, the child exhibits severe emotional distress about attending school but is willing to complete schoolwork at home.
Why children may avoid school
School avoidance is a biopsychosocial condition with a multitude of underlying causes.4 It is associated most commonly with anxiety disorders and neurodevelopmental disorders, including but not limited to learning disabilities and attention-deficit/hyperactivity disorder.1 Depressive disorders also have been associated with school avoidance.7 Social concerns related to changes with school personnel or classes, academic challenges, bullying, health emergencies, and family stressors also can result in symptoms of school avoidance.1
Continue to: A child seeking to avoid...
A child seeking to avoid school may be motivated by potential negative and/or positive effects of doing so. Kearney and Silverman8 identified 4 primary functions of school refusal behaviors:
- avoiding stimuli at school that lend to negative affect (depression, anxiety)
- escaping the social interactions and/or situations for evaluation that occur at school
- gaining more attention from caregivers, and
- obtaining tangible rewards or benefits outside the school environment.
How school avoidance manifests
School avoidance has attributes of internalizing (depression, anxiety, somatic complaints) and externalizing (aggression, tantrums, running away, clinginess) behaviors. It can cause distress for the student, parents and caregivers, and school personnel.
The avoidance may manifest with behaviors such as crying, hiding, emotional outbursts, and refusing to move prior to the start of the school day. Additionally, the child may beg their parents not to make them go to school or, when at school, they may leave the classroom to go to a safe place such as the nurse’s or counselor’s office.
The avoidance may occur abruptly, such as after a break in the school schedule or a change of school. Or it may be the final result of the student’s gradual inability to cope with the underlying issue.
How to assess for school avoidance
Due to the multifactorial nature of this presenting concern, a comprehensive evaluation is recommended when school avoidance is reported.4 Often the child will present with physical symptoms, such as abdominal pain, nausea, vomiting, diarrhea, headaches, shortness of breath, dizziness, chest pain, and palpitations. A thorough medical examination should be performed to rule out a physiological cause. The medical visit should include clinical interviews with the patient and family members or guardians.
Continue to: To identify school avoidance...
To identify school avoidance in pediatric and adolescent populations, medical history and physical examination—along with social history to better understand familial, social, and academic concerns—should be a regular part of the medical encounter. The School Refusal Assessment Scale-Revised (SRAS-R) for both parents and their children was developed to assess for school avoidance and can be utilized within the primary care setting. Additional psychiatric history for both the family and patient may be beneficial, due to associations between parental mental health concerns and school avoidance in their children.9,10
Assessment for an underlying mental health condition, such as an anxiety or depressive disorder, should be completed when a patient presents with school avoidance.4 More than one-third of children with behavioral problems, such as school avoidance, have been diagnosed with anxiety.11 The 2020 National Survey of Children’s Health found that 7.8% of children and adolescents ages 3 to 17 years had a current anxiety disorder, leading the US Preventive Services Task Force to recommend screening for anxiety in children and adolescents ages 8 to 18 years.12,13 Furthermore, if academic achievement is of concern, then consideration of further assessment for neurodevelopmental disorders is warranted.1
Treatment is multimodal and multidisciplinary
Treatment for school avoidance is often multimodal and may involve interdisciplinary, team-based care including the medical provider, school system (eg, Child Study Team), family, and mental health care provider.1,4
Cognitive behavioral therapy (CBT) is the most-studied intervention for school avoidance, with behavioral, exposure-based interventions often central to therapeutic gains in treatment.1,14,15 The goals of treatment are to increase school attendance while decreasing emotional distress through various strategies, including exposure-based interventions, contingency management with parents and school staff, relaxation training, and/or social skills training.14,16 Collaborative involvement between the medical provider and the school system is key to successful treatment.
Medication may be considered alone or in combination with CBT when comorbid mental health conditions have been identified. Selective serotonin reuptake inhibitors (SSRIs)—including fluoxetine, sertraline, and escitalopram—are considered first-line treatment for anxiety in children and adolescents.17 Serotonin-norepinephrine reuptake inhibitors (SNRIs), such as duloxetine and venlafaxine, also have been shown to be effective. Duloxetine is the only medication approved by the US Food and Drug Administration (FDA) for treatment of generalized anxiety disorder in children ages 7 years and older.17
Continue to: SSRIs and SNRIs have a boxed warning...
SSRIs and SNRIs have a boxed warning from the FDA for increased suicidal thoughts and behaviors in children and adolescents. Although this risk is rare, it should be discussed with the patient and parent/guardian in order to obtain informed consent prior to treatment initiation.
Medication should be started at the lowest possible dose and increased gradually. Patients should remain on the medication for 6 to 12 months after symptom resolution and should be tapered during a nonstressful time, such as the summer break.
THE CASE
Based on the concerns of continued school refusal after negative gastrointestinal work-up, Juana’s physician screened her for anxiety and conducted a clinical interview to better understand any psychosocial concerns. Juana’s score of 10 on the General Anxiety Disorder-7 scale indicated moderate anxiety. She reported symptoms consistent with social anxiety disorder contributing to school avoidance.
The physician consulted with the clinic’s behavioral health consultant (BHC) to confirm the multimodal treatment plan, which was then discussed with Juana and her mother. The physician discussed medication options (SSRIs) and provided documentation (in both English and Spanish) from the visit to Juana’s mother so she could initiate a school-based intervention with the Child Study Team at Juana’s school. A plan for CBT—including a collaborative contingency management plan between the patient and her parent (eg, a reward chart for attending school) and exposure interventions (eg, a graduated plan to participate in school-based activities with the end goal to resume full school attendance)—was developed with the BHC. Biweekly follow-up appointments were scheduled with the BHC and monthly appointments were scheduled with the physician to reinforce the interventions.
CORRESPONDENCE
Meredith L. C. Williamson, PhD, 2900 East 29th Street, Suite 100, Bryan, TX 77840; meredith.williamson@tamu.edu
THE CASE
Juana*, a 10-year-old who identifies as a cisgender, Hispanic female, was referred to our integrated behavioral health program by her primary care physician. Her mother was concerned because Juana had been refusing to attend school due to complaints of gastrointestinal upset. This concern began when Juana was in first grade but had increased in severity over the past few months.
Upon further questioning, the patient reported that she initially did not want to attend school due to academic difficulties and bullying. However, since COVID-19, her fears of attending school had significantly worsened. Juana’s mother’s primary language was Spanish and she had limited English proficiency; she reported difficulty communicating with school personnel about Juana’s poor attendance.
Juana had recently had a complete medical work-up for her gastrointestinal concerns, with negative results. Since the negative work-up, Juana’s mother had told her daughter that she would be punished if she didn’t go to school.
●
* The patient’s name has been changed to protect her identity.
School avoidance, also referred to as school refusal, is a symptom of an emotional condition that manifests as a child refusing to go to school or having difficulty going to school or remaining in the classroom for the entire day. School avoidance is not a clinical diagnosis but often is related to an underlying disorder.1
School avoidance is common, affecting 5% to 28% of youth sometime in their school career.2 Available data are not specific to school avoidance but focus on chronic absenteeism (missing ≥ 15 days per school year). Rates of chronic absenteeism are high in elementary and middle school (about 14% each) and tend to increase in high school (about 21%).3 Students with disabilities are 1.5 times more likely to be chronically absent than students without disabilities.3 Compared to White students, American Indian and Pacific Islander students are > 50% more likely, Black students 40% more likely, and Hispanic students 17% more likely to miss ≥ 3 weeks of school.3 Rates of chronic absenteeism are similar (about 16%) for males and females.3
Absenteeism can have immediate and long-term negative effects.4 School attendance issues are correlated to negative life outcomes, such as delinquency, teen pregnancy, substance use, and poor academic achievement.5 According to the US Department of Education, individuals who chronically miss school are less likely to achieve educational milestones (particularly in younger years) and may be more likely to drop out of school.3
What school avoidance is (and what it isn’t)
It is important to distinguish school avoidance from truancy. Truancy often is associated with antisocial behavior such as lying and stealing, while school avoidance occurs in the absence of significant antisocial disorders.6 With truancy, the absence usually is hidden from the parent. In contrast, with school avoidance, the parents usually know where their child is; the child often spends the day secluded in their bedroom. Students who engage in truancy do not demonstrate excessive anxiety about attending school but may have decreased interest in schoolwork and academic performance.6 With school avoidance, the child exhibits severe emotional distress about attending school but is willing to complete schoolwork at home.
Why children may avoid school
School avoidance is a biopsychosocial condition with a multitude of underlying causes.4 It is associated most commonly with anxiety disorders and neurodevelopmental disorders, including but not limited to learning disabilities and attention-deficit/hyperactivity disorder.1 Depressive disorders also have been associated with school avoidance.7 Social concerns related to changes with school personnel or classes, academic challenges, bullying, health emergencies, and family stressors also can result in symptoms of school avoidance.1
Continue to: A child seeking to avoid...
A child seeking to avoid school may be motivated by potential negative and/or positive effects of doing so. Kearney and Silverman8 identified 4 primary functions of school refusal behaviors:
- avoiding stimuli at school that lend to negative affect (depression, anxiety)
- escaping the social interactions and/or situations for evaluation that occur at school
- gaining more attention from caregivers, and
- obtaining tangible rewards or benefits outside the school environment.
How school avoidance manifests
School avoidance has attributes of internalizing (depression, anxiety, somatic complaints) and externalizing (aggression, tantrums, running away, clinginess) behaviors. It can cause distress for the student, parents and caregivers, and school personnel.
The avoidance may manifest with behaviors such as crying, hiding, emotional outbursts, and refusing to move prior to the start of the school day. Additionally, the child may beg their parents not to make them go to school or, when at school, they may leave the classroom to go to a safe place such as the nurse’s or counselor’s office.
The avoidance may occur abruptly, such as after a break in the school schedule or a change of school. Or it may be the final result of the student’s gradual inability to cope with the underlying issue.
How to assess for school avoidance
Due to the multifactorial nature of this presenting concern, a comprehensive evaluation is recommended when school avoidance is reported.4 Often the child will present with physical symptoms, such as abdominal pain, nausea, vomiting, diarrhea, headaches, shortness of breath, dizziness, chest pain, and palpitations. A thorough medical examination should be performed to rule out a physiological cause. The medical visit should include clinical interviews with the patient and family members or guardians.
Continue to: To identify school avoidance...
To identify school avoidance in pediatric and adolescent populations, medical history and physical examination—along with social history to better understand familial, social, and academic concerns—should be a regular part of the medical encounter. The School Refusal Assessment Scale-Revised (SRAS-R) for both parents and their children was developed to assess for school avoidance and can be utilized within the primary care setting. Additional psychiatric history for both the family and patient may be beneficial, due to associations between parental mental health concerns and school avoidance in their children.9,10
Assessment for an underlying mental health condition, such as an anxiety or depressive disorder, should be completed when a patient presents with school avoidance.4 More than one-third of children with behavioral problems, such as school avoidance, have been diagnosed with anxiety.11 The 2020 National Survey of Children’s Health found that 7.8% of children and adolescents ages 3 to 17 years had a current anxiety disorder, leading the US Preventive Services Task Force to recommend screening for anxiety in children and adolescents ages 8 to 18 years.12,13 Furthermore, if academic achievement is of concern, then consideration of further assessment for neurodevelopmental disorders is warranted.1
Treatment is multimodal and multidisciplinary
Treatment for school avoidance is often multimodal and may involve interdisciplinary, team-based care including the medical provider, school system (eg, Child Study Team), family, and mental health care provider.1,4
Cognitive behavioral therapy (CBT) is the most-studied intervention for school avoidance, with behavioral, exposure-based interventions often central to therapeutic gains in treatment.1,14,15 The goals of treatment are to increase school attendance while decreasing emotional distress through various strategies, including exposure-based interventions, contingency management with parents and school staff, relaxation training, and/or social skills training.14,16 Collaborative involvement between the medical provider and the school system is key to successful treatment.
Medication may be considered alone or in combination with CBT when comorbid mental health conditions have been identified. Selective serotonin reuptake inhibitors (SSRIs)—including fluoxetine, sertraline, and escitalopram—are considered first-line treatment for anxiety in children and adolescents.17 Serotonin-norepinephrine reuptake inhibitors (SNRIs), such as duloxetine and venlafaxine, also have been shown to be effective. Duloxetine is the only medication approved by the US Food and Drug Administration (FDA) for treatment of generalized anxiety disorder in children ages 7 years and older.17
Continue to: SSRIs and SNRIs have a boxed warning...
SSRIs and SNRIs have a boxed warning from the FDA for increased suicidal thoughts and behaviors in children and adolescents. Although this risk is rare, it should be discussed with the patient and parent/guardian in order to obtain informed consent prior to treatment initiation.
Medication should be started at the lowest possible dose and increased gradually. Patients should remain on the medication for 6 to 12 months after symptom resolution and should be tapered during a nonstressful time, such as the summer break.
THE CASE
Based on the concerns of continued school refusal after negative gastrointestinal work-up, Juana’s physician screened her for anxiety and conducted a clinical interview to better understand any psychosocial concerns. Juana’s score of 10 on the General Anxiety Disorder-7 scale indicated moderate anxiety. She reported symptoms consistent with social anxiety disorder contributing to school avoidance.
The physician consulted with the clinic’s behavioral health consultant (BHC) to confirm the multimodal treatment plan, which was then discussed with Juana and her mother. The physician discussed medication options (SSRIs) and provided documentation (in both English and Spanish) from the visit to Juana’s mother so she could initiate a school-based intervention with the Child Study Team at Juana’s school. A plan for CBT—including a collaborative contingency management plan between the patient and her parent (eg, a reward chart for attending school) and exposure interventions (eg, a graduated plan to participate in school-based activities with the end goal to resume full school attendance)—was developed with the BHC. Biweekly follow-up appointments were scheduled with the BHC and monthly appointments were scheduled with the physician to reinforce the interventions.
CORRESPONDENCE
Meredith L. C. Williamson, PhD, 2900 East 29th Street, Suite 100, Bryan, TX 77840; meredith.williamson@tamu.edu
1. School Avoidance Alliance. School avoidance facts. Published September 16, 2021. Accessed July 27, 2023. https://schoolavoidance.org/school-avoidance-facts/
2. Kearney CA. School Refusal Behavior in Youth: A Functional Approach to Assessment and Treatment. American Psychological Association; 2001.
3. US Department of Education. Chronic absenteeism in the nation’s schools: a hidden educational crisis. Updated January 2019. Accessed August 3, 2023. www2.ed.gov/datastory/chronicabsenteeism.html
4. Allen CW, Diamond-Myrsten S, Rollins LK. School absenteeism in children and adolescents. Am Fam Physician. 2018;98:738-744.
5. Gonzálvez C, Díaz-Herrero Á, Vicent M, et al. School refusal behavior: latent class analysis approach and its relationship with psychopathological symptoms. Curr Psychology. 2022;41:2078-2088. doi: 10.1007/s12144-020-00711-6
6. Fremont WP. School refusal in children and adolescents. Am Fam Physician. 2003;68:1555-1560.
7. McShane G, Walter G, Rey JM. Characteristics of adolescents with school refusal. Aust N Z J Psychiatry. 2001;35:822-826. doi: 10.1046/j.1440-1614.2001.00955.x
8. Kearney CA, Silverman WK. The evolution and reconciliation of taxonomic strategies for school refusal behavior. Clin Psychology Sci Pract. 1996;3:339-354. doi: 10.1111/j.1468-2850.1996.tb00087.x
9. Kearney CA, Albano AM. School Refusal Assessment Scale-Revised C. Oxford University Press; 2007.
10. Heyne D. School refusal. In: Fisher JE, O’Donohue WT (eds). Practitioner’s Guide to Evidence-based Psychotherapy. Springer Science + Business Media. 2006;600-619. doi: 10.1007/978-0-387-28370-8_60
11. Ghandour RM, Sherman LJ, Vladutiu CJ, et al. Prevalence and treatment of depression, anxiety, and conduct problems in US children. J Pediatrics. 2019;206:256-267.e3. doi: 10.1016/j.jpeds.2018.09.021
12. US Census Bureau. 2020 National Survey of Children’s Health: Topical Frequencies. Published June 2, 2021. Accessed August 4, 2023. www2.census.gov/programs-surveys/nsch/technical-documentation/codebook/NSCH_2020_Topical_Frequencies.pdf
13. USPSTF. Anxiety in children and adolescents: screening. Final Recommendation Statement. Published October 11, 2022. Accessed August 4, 2023. www.uspreventiveservicestaskforce.org/uspstf/recommendation/screening-anxiety-children-adolescents
14. Maynard BR, Brendel KE, Bulanda JJ, et al. Psychosocial interventions for school refusal with primary and secondary school students: a systematic review. Campbell Systematic Rev. 2015;11:1-76. doi: 10.4073/csr.2015.12
15. Kearney CA, Albano AM. When Children Refuse School: Parent Workbook. 3rd ed. Oxford University Press; 2018. doi: 10.1093/med-psych/9780190604080.001.0001
16. Heyne DA, Sauter FM. School refusal. In: Essau CA, Ollendick TH. The Wiley-Blackwell Handbook of the Treatment of Childhood and Adolescent Anxiety. Wiley Blackwell; 2013:471-517.
17. Kowalchuk A, Gonzalez SJ, Zoorob RJ. Anxiety disorders in children and adolescents. Am Fam Physician. 2022;106:657-664.
1. School Avoidance Alliance. School avoidance facts. Published September 16, 2021. Accessed July 27, 2023. https://schoolavoidance.org/school-avoidance-facts/
2. Kearney CA. School Refusal Behavior in Youth: A Functional Approach to Assessment and Treatment. American Psychological Association; 2001.
3. US Department of Education. Chronic absenteeism in the nation’s schools: a hidden educational crisis. Updated January 2019. Accessed August 3, 2023. www2.ed.gov/datastory/chronicabsenteeism.html
4. Allen CW, Diamond-Myrsten S, Rollins LK. School absenteeism in children and adolescents. Am Fam Physician. 2018;98:738-744.
5. Gonzálvez C, Díaz-Herrero Á, Vicent M, et al. School refusal behavior: latent class analysis approach and its relationship with psychopathological symptoms. Curr Psychology. 2022;41:2078-2088. doi: 10.1007/s12144-020-00711-6
6. Fremont WP. School refusal in children and adolescents. Am Fam Physician. 2003;68:1555-1560.
7. McShane G, Walter G, Rey JM. Characteristics of adolescents with school refusal. Aust N Z J Psychiatry. 2001;35:822-826. doi: 10.1046/j.1440-1614.2001.00955.x
8. Kearney CA, Silverman WK. The evolution and reconciliation of taxonomic strategies for school refusal behavior. Clin Psychology Sci Pract. 1996;3:339-354. doi: 10.1111/j.1468-2850.1996.tb00087.x
9. Kearney CA, Albano AM. School Refusal Assessment Scale-Revised C. Oxford University Press; 2007.
10. Heyne D. School refusal. In: Fisher JE, O’Donohue WT (eds). Practitioner’s Guide to Evidence-based Psychotherapy. Springer Science + Business Media. 2006;600-619. doi: 10.1007/978-0-387-28370-8_60
11. Ghandour RM, Sherman LJ, Vladutiu CJ, et al. Prevalence and treatment of depression, anxiety, and conduct problems in US children. J Pediatrics. 2019;206:256-267.e3. doi: 10.1016/j.jpeds.2018.09.021
12. US Census Bureau. 2020 National Survey of Children’s Health: Topical Frequencies. Published June 2, 2021. Accessed August 4, 2023. www2.census.gov/programs-surveys/nsch/technical-documentation/codebook/NSCH_2020_Topical_Frequencies.pdf
13. USPSTF. Anxiety in children and adolescents: screening. Final Recommendation Statement. Published October 11, 2022. Accessed August 4, 2023. www.uspreventiveservicestaskforce.org/uspstf/recommendation/screening-anxiety-children-adolescents
14. Maynard BR, Brendel KE, Bulanda JJ, et al. Psychosocial interventions for school refusal with primary and secondary school students: a systematic review. Campbell Systematic Rev. 2015;11:1-76. doi: 10.4073/csr.2015.12
15. Kearney CA, Albano AM. When Children Refuse School: Parent Workbook. 3rd ed. Oxford University Press; 2018. doi: 10.1093/med-psych/9780190604080.001.0001
16. Heyne DA, Sauter FM. School refusal. In: Essau CA, Ollendick TH. The Wiley-Blackwell Handbook of the Treatment of Childhood and Adolescent Anxiety. Wiley Blackwell; 2013:471-517.
17. Kowalchuk A, Gonzalez SJ, Zoorob RJ. Anxiety disorders in children and adolescents. Am Fam Physician. 2022;106:657-664.
Is the Altman Rule a proxy for glycemic load?
ABSTRACT
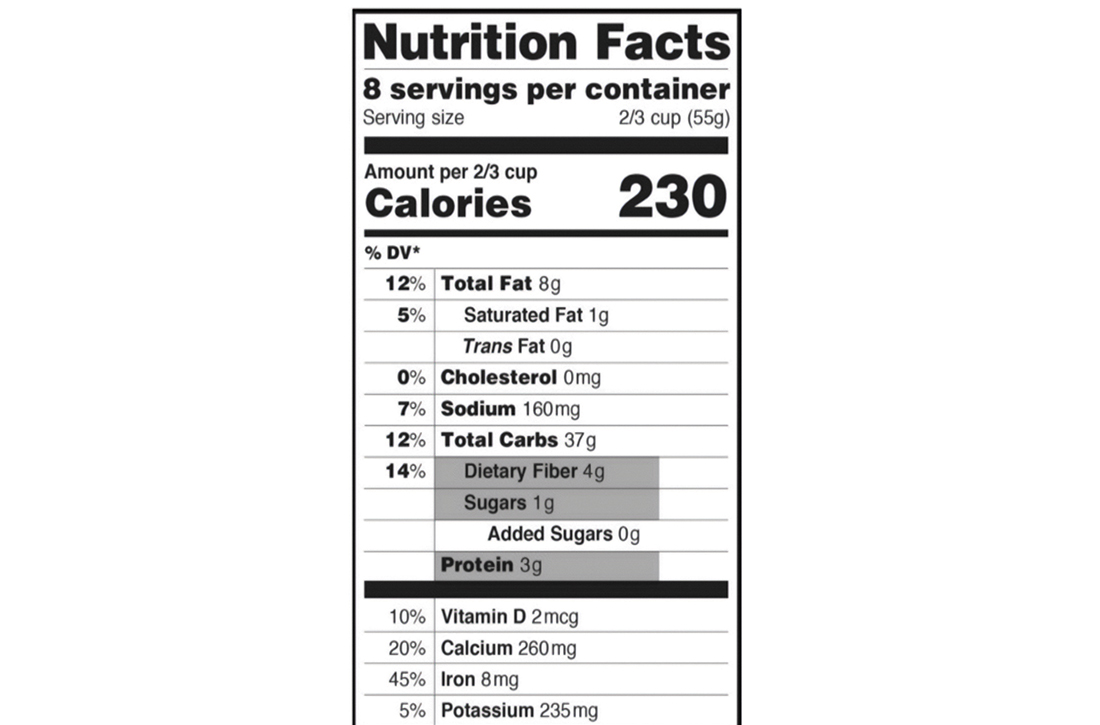
Background: The Altman Rule, a simple tool for consumers seeking to make healthier packaged food choices at the point of sale, applies to packaged carbohydrates. According to the Altman Rule, a food is a healthier option if it has at least 3 g of fiber per serving and the grams of fiber plus the grams of protein exceed the grams of sugar per serving. This study sought to evaluate whether the Altman Rule is a valid proxy for glycemic load (GL).
Methods: We compared the binary outcome of whether a food item meets the Altman Rule with the GL of all foods categorized as cereals, chips, crackers, and granola bars in the Nutrition Data System for Research Database (University of Minnesota, Version 2010). We examined the percentage of foods in low-, medium-, and high-GL categories that met the Altman Rule.
Results: There were 1235 foods (342 cereals, 305 chips, 379 crackers, and 209 granola bars) in this analysis. There was a significant relationship between the GL of foods and the Altman Rule (P < .001) in that most low-GL (68%), almost half of medium-GL (48%), and very few high-GL (7%) foods met the criteria of the rule.
Conclusions: The Altman Rule is a reasonable proxy for GL and can be a useful and accessible tool for consumers interested in buying healthier packaged carbohydrate foods.
Nutrition can be complicated for consumers interested in making healthier choices at the grocery store. Consumers may have difficulty identifying more nutritious options, especially when food labels are adorned with claims such as “Good Source of Fiber” or “Heart Healthy.”1 In addition, when reading food labels, consumers may find it difficult to decipher which data to prioritize when carbohydrates, total sugars, added sugars, total dietary fiber, soluble fiber, and insoluble fiber are all listed.
The concept of glycemic load (GL) is an important consideration, especially for people with diabetes. GL approximates the blood sugar response to different foods. A food with a high GL is digested quickly, and its carbohydrates are taken into the bloodstream rapidly. This leads to a spike and subsequent drop in blood sugars, which can cause symptoms of hyperglycemia and hypoglycemia in a person with diabetes.2,3 Despite its usefulness, GL may be too complicated for a consumer to understand, and it does not appear anywhere on the food label. Since GL is calculated using pooled blood sugar response from individuals after the ingestion of the particular food, estimation of the GL is not intuitable.4
Point-of-sale tools. People seeking to lose weight, control diabetes, improve dyslipidemia and/or blood pressure, and/or decrease their risk for heart disease may benefit from point-of-sale tools such as the Altman Rule, which simplifies and encourages the selection of more nutritious foods.1 Other tools—such as Guiding Stars (https://guidingstars.com), NuVal (www.nuval.com), and different variations of traffic lights—have been created to help consumers make more informed and healthier food choices.5-8 However, Guiding Stars and NuVal are based on complicated algorithms that are not entirely transparent and not accessible to the average consumer.6,7 Evaluations of these nutrition tools indicate that consumers tend to underrate the healthiness of some foods, such as raw almonds and salmon, and overrate the healthiness of others, such as fruit punch and diet soda, when using traffic light systems.6 Furthermore, these nutrition tools are not available in many supermarkets. Previous research suggests that the use of point-of-sale nutrition apps decreases with the time and effort involved in using an app.9
Continue to: The Altman Rule
The Altman Rule was developed by a family physician (author WA) to provide a more accessible tool for people interested in choosing healthier prepackaged carbohydrate foods while shopping. Since the user does not need to have a smartphone, and they are not required to download or understand an app for each purchase, the Altman Rule may be more usable compared with more complicated alternatives.

The Altman Rule can be used with nutrition labels that feature serving information and calories in enlarged and bold type, in compliance with the most recent US Food and Drug Administration (FDA) guideline from 2016. Many foods with high fiber also have high amounts of sugar, so the criteria of the Altman Rule includes a 2-step process requiring (1) a minimum of 3 g of total dietary fiber per serving and (2) the sum of the grams of fiber plus the grams of protein per serving to be greater than the total grams of sugar (not grams of added sugar or grams of carbohydrate) per serving (FIGURE 1A). Unlike the relatively complicated formula related to GL, this 2-part rule can be applied in seconds while shopping (FIGURE 1B).
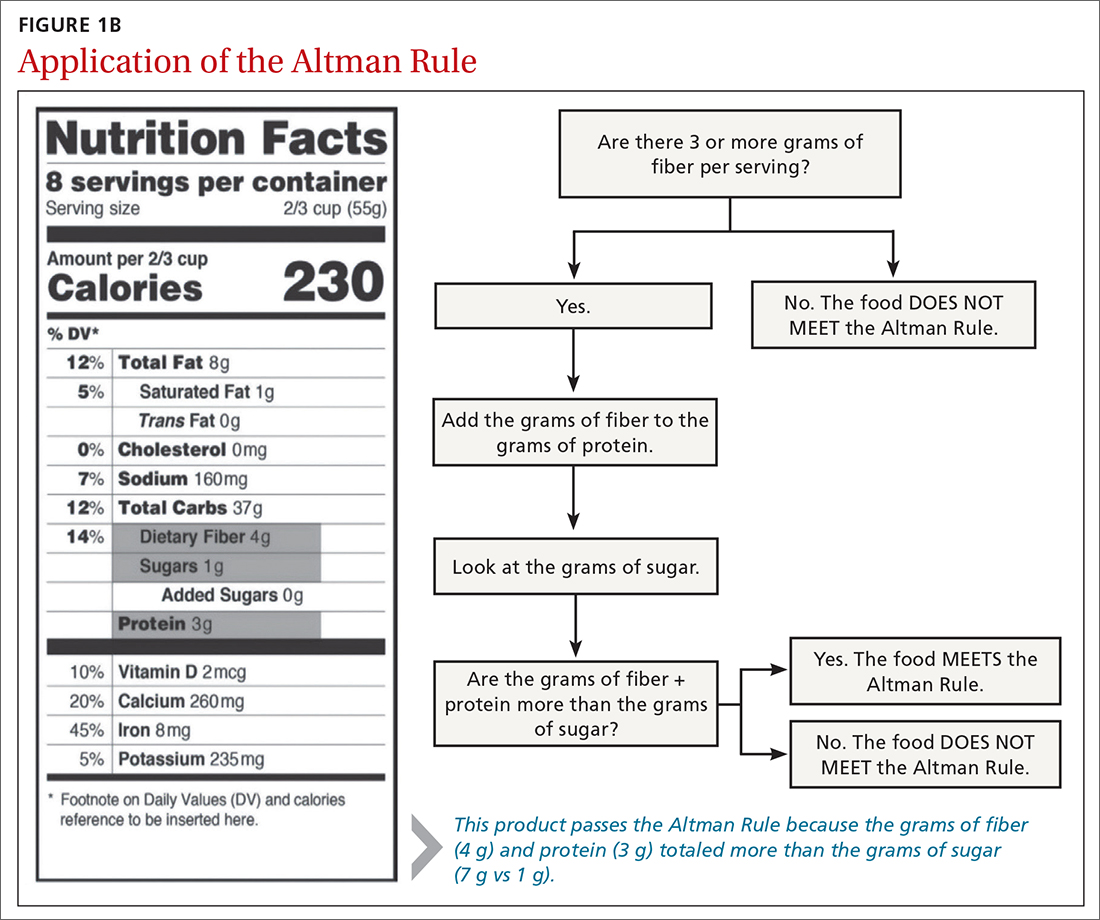
The rule is intended only to be used for packaged carbohydrate products, such as bread, muffins, bagels, pasta, rice, oatmeal, cereals, snack bars, chips, and crackers. It does not apply to whole foods, such as meat, dairy, fruits, or vegetables. These foods are excluded to prevent any consumer confusion related to the nutritional content of whole foods (eg, an apple may have more sugar than fiber and protein combined, but it is still a nutritious option).
This study aimed to determine if the Altman Rule is a reasonable proxy for the more complicated concept of GL. We calculated the relationship between the GL of commercially available packaged carbohydrate foods and whether those foods met the Altman Rule.
METHODS
The Altman Rule was tested by comparing the binary outcome of the rule (meets/does not meet) with data on all foods categorized as cereals, chips, crackers, and granola bars in the Nutrition Data System for Research (NDSR) Database (University of Minnesota, Version 2010).
Continue to: To account for differences...
To account for differences in serving size, we used the standard of 50 g for each product as 1 serving. We used 50 g (about 1.7 oz) to help compare the different foods and between foods within the same group. Additionally, 50 g is close to 1 serving for most foods in these groups; it is about the size of a typical granola bar, three-quarters to 2 cups of cereal, 10 to 12 crackers, and 15 to 25 chips. We determined the GL for each product by multiplying the number of available carbohydrates (total carbohydrate – dietary fiber) by the product’s glycemic index/100. In general, GL is categorized as low (≤ 10), medium (11-19), or high (≥ 20).
We applied the Altman Rule to categorize each product as meeting or not meeting the rule. We compared the proportion of foods meeting the Altman Rule, stratified by GL and by specific foods, and used chi-square to determine if differences were statistically significant. These data were collected and analyzed in the summer of 2019.
RESULTS
There were 1235 foods (342 breakfast cereals, 305 chips, 379 crackers, and 209 granola bars) used for this analysis. There is a significant relationship between the GL of foods and the Altman Rule in that most low-GL (68%), almost half of medium-GL (48%), and only a few high-GL foods (7%) met the rule (P < .001) (TABLE 1). There was also a significant relationship between “meeting the Altman Rule” and GL within each food type (P < .001) (TABLE 2).
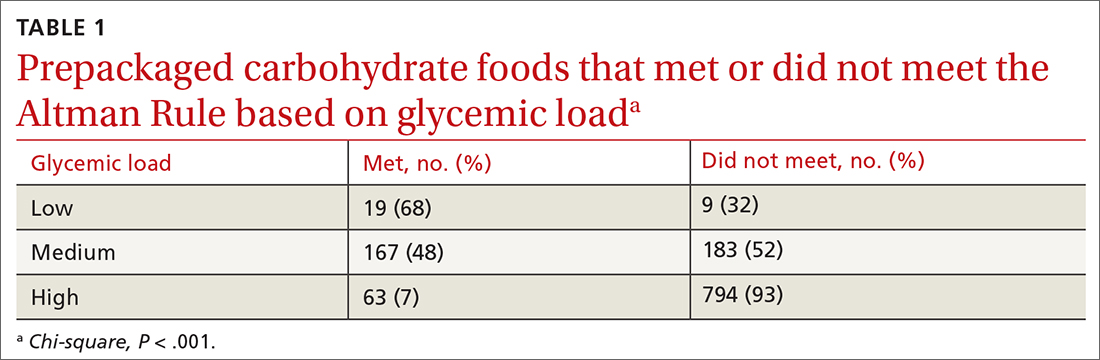
The medium-GL foods were the second largest category of foods we calculated; thus we further broke them into binary categories of
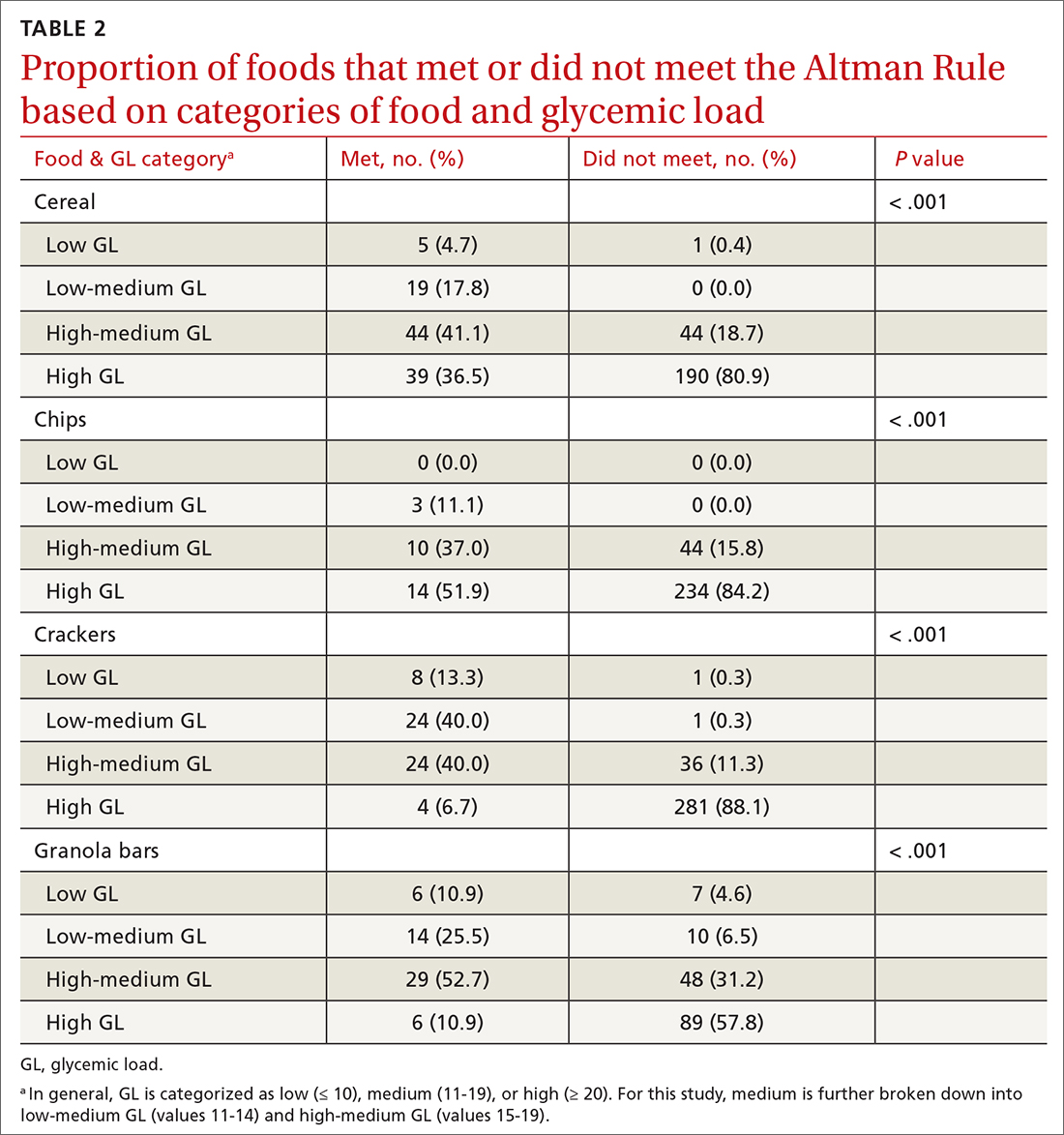
Foods that met the rule were more likely to be low GL and foods that did not pass the rule were more likely high GL. Within the medium-GL category, foods that met the rule were more likely to be low-medium GL.
Continue to: The findings within food categories...
The findings within food categories showed that very few cereals, chips, crackers, and granola bars were low GL. For every food category, except granola bars, far more low-GL foods met the Altman Rule than those that did not. At the same time, very few high-GL foods met the Altman Rule. The category with the most individual high-GL food items meeting the Altman Rule was cereal. This was also the subcategory with the largest percentage of high-GL food items meeting the Altman Rule. Thirty-nine cereals that were high GL met the rule, but more than 4 times as many high-GL cereals did not (n = 190).
DISCUSSION
Marketing and nutrition messaging create consumer confusion that makes it challenging to identify packaged food items that are more nutrient dense. The Altman Rule simplifies food choices that have become unnecessarily complex. Our findings suggest this 2-step rule is a reasonable proxy for the more complicated and less accessible GL for packaged carbohydrates, such as cereals, chips, crackers, and snack bars. Foods that meet the rule are likely low or low-medium GL and thus are foods that are likely to be healthier choices.
Of note, only 9% of chips (n = 27) passed the Altman Rule, likely due to their low dietary fiber content, which was typical of chips. If a food item does not have at least 3 grams of total dietary fiber per serving, it does not pass the Altman Rule, regardless of how much protein or sugar is in the product. This may be considered a strength or a weakness of the Altman Rule. Few nutrition-dense foods are low in fiber, but some foods could be nutritious but do not meet the Altman Rule due to having < 3 g of fiber.
With the high prevalence of chronic diseases such as hypertension, diabetes, hyperlipidemia, and cardiovascular disease, it is essential to help consumers prevent chronic disease altogether or manage their chronic disease by providing tools to identify healthier food choices. The tool also has a place in clinical medicine for use by physicians and other health care professionals. Research shows that physicians find both time and lack of knowledge/resources to be a barrier to providing nutritional counseling to patients.10 Since the Altman Rule can be shared and explained with very little time and without extensive nutritional knowledge, it meets these needs.
Limitations
Glycemic load. We acknowledge that the Altman Rule is not foolproof and that assessing this rule based on GL has some limitations. GL is not a perfect or comprehensive way to measure the nutritional value of a food. For example, fruits such as watermelon and grapes are nutritionally dense. However, they contain high amounts of natural sugars—and as such, their GL is relatively high, which could lead a consumer to perceive them as unhealthy. Nevertheless, GL is both a useful and accepted tool and a reasonable way to assess the validity of the rule, specifically when assessing packaged carbohydrates. The simplicity of the Altman Rule and its relationship with GL makes it such that consumers are more likely to make a healthier food choice using it.9
Continue to: Specificity and sensitivity
Specificity and sensitivity. There are other limitations to the Altman Rule, given that a small number of high-GL foods meet the rule. For example, some granola bars had high dietary protein, which offset a high sugar content just enough to pass the rule despite a higher GL. As such, concluding that a snack bar is a healthier choice because it meets the Altman Rule when it has high amounts of sugar may not be appropriate. This limitation could be considered a lack of specificity (the rule includes food it ought not to include). Another limitation to consider would be a lack of sensitivity, given that only 68% of low-GL foods passed the Altman Rule. Since GL is associated with carbohydrate content, foods with a low carbohydrate count often have little to no fiber and thus would fall into the category of foods that did not meet the Altman Rule but had low GL. In this case, however, the low amount of fiber may render the Altman Rule a better indicator of a healthier food choice than the GL.
Hidden sugars. Foods with sugar alcohols and artificial sweeteners may be as deleterious as caloric alternatives while not being accounted for when reporting the grams of sugar per serving on the nutrition label.7 This may represent an exception to the Altman Rule, as foods that are not healthier choices may pass the rule because the sugar content on the nutrition label is, in a sense, artificially lowered. Future research may investigate the hypothesis that these foods are nutritionally inferior despite meeting the Altman Rule.
The sample. Our study also was limited to working only with foods that were included in the NDSR database up to 2010. This limitation is mitigated by the fact that the sample size was large (> 1000 packaged food items were included in our analyses). The study also could be limited by the food categories that were analyzed; food categories such as bread, rice, pasta, and bagels were not included.
The objective of this research was to investigate the relationship between GL and the Altman Rule, rather than to conduct an exhaustive analysis of the Altman Rule for every possible food category. Studying the relationship between the Altman Rule and GL in other categories of food is an objective for future research. The data so far support a relationship between these entities. The likelihood of the nutrition facts of foods changing without the GL changing (or vice versa) is very low. As such, the Altman Rule still seems to be a reasonable proxy of GL.
CONCLUSIONS
Research indicates that point-of-sale tools, such as Guiding Stars, NuVal, and other stoplight tools, can successfully alter consumers’ behaviors.9 These tools can be helpful but are not available in many supermarkets. Despite the limitations, the Altman Rule is a useful decision aid that is accessible to all consumers no matter where they live or shop and is easy to use and remember.
The Altman rule can be used in clinical practice by health care professionals, such as physicians, nurse practitioners, physician assistants, dietitians, and health coaches. It also has the potential to be used in commercial settings, such as grocery stores, to help consumers easily identify healthier convenience foods. This has public health implications, as the rule can both empower consumers and potentially incentivize food manufacturers to upgrade their products nutritionally.
Additional research would be useful to evaluate consumers’ preferences and perceptions about how user-friendly the Altman Rule is at the point of sale with packaged carbohydrate foods. This would help to further understand how the use of information on food packaging can motivate healthier decisions—thereby helping to alleviate the burden of chronic disease.
CORRESPONDENCE
Kimberly R. Dong, DrPH, MS, RDN, Tufts University School of Medicine, Department of Public Health and Community Medicine, 136 Harrison Avenue, MV Building, Boston, MA 02111; kimberly.dong@tufts.edu
1. Hersey JC, Wohlgenant KC, Arsenault JE, et al. Effects of front-of-package and shelf nutrition labeling systems on consumers. Nutr Rev. 2013;71:1-14. doi: 10.1111/nure.12000
2. Jenkins DJA, Dehghan M, Mente A, et al. Glycemic index, glycemic load, and cardiovascular disease and mortality. N Engl J Med. 2021;384:1312-1322. doi: 10.1056/NEJMoa2007123
3. Brand-Miller J, Hayne S, Petocz P, et al. Low–glycemic index diets in the management of diabetes. Diabetes Care. 2003;26:2261-2267. doi: 10.2337/diacare.26.8.2261
4. Matthan NR, Ausman LM, Meng H, et al. Estimating the reliability of glycemic index values and potential sources of methodological and biological variability. Am J Clin Nutr. 2016;104:1004-1013. doi: 10.3945/ajcn.116.137208
5. Sonnenberg L, Gelsomin E, Levy DE, et al. A traffic light food labeling intervention increases consumer awareness of health and healthy choices at the point-of-purchase. Prev Med. 2013;57:253-257. doi: 10.1016/j.ypmed.2013.07.001
6. Savoie N, Barlow K, Harvey KL, et al. Consumer perceptions of front-of-package labelling systems and healthiness of foods. Can J Public Health. 2013;104:e359-e363. doi: 10.17269/cjph.104.4027
7. Fischer LM, Sutherland LA, Kaley LA, et al. Development and implementation of the Guiding Stars nutrition guidance program. Am J Health Promot. 2011;26:e55-e63. doi: 10.4278/ajhp.100709-QUAL-238
8. Maubach N, Hoek J, Mather D. Interpretive front-of-pack nutrition labels. Comparing competing recommendations. Appetite. 2014;82:67-77. doi: 10.1016/j.appet.2014.07.006
9. Chan J, McMahon E, Brimblecombe J. Point‐of‐sale nutrition information interventions in food retail stores to promote healthier food purchase and intake: a systematic review. Obes Rev. 2021;22. doi: 10.1111/obr.13311
10. Mathioudakis N, Bashura H, Boyér L, et al. Development, implementation, and evaluation of a physician-targeted inpatient glycemic management curriculum. J Med Educ Curric Dev. 2019;6:238212051986134. doi: 10.1177/2382120519861342
ABSTRACT
Background: The Altman Rule, a simple tool for consumers seeking to make healthier packaged food choices at the point of sale, applies to packaged carbohydrates. According to the Altman Rule, a food is a healthier option if it has at least 3 g of fiber per serving and the grams of fiber plus the grams of protein exceed the grams of sugar per serving. This study sought to evaluate whether the Altman Rule is a valid proxy for glycemic load (GL).
Methods: We compared the binary outcome of whether a food item meets the Altman Rule with the GL of all foods categorized as cereals, chips, crackers, and granola bars in the Nutrition Data System for Research Database (University of Minnesota, Version 2010). We examined the percentage of foods in low-, medium-, and high-GL categories that met the Altman Rule.
Results: There were 1235 foods (342 cereals, 305 chips, 379 crackers, and 209 granola bars) in this analysis. There was a significant relationship between the GL of foods and the Altman Rule (P < .001) in that most low-GL (68%), almost half of medium-GL (48%), and very few high-GL (7%) foods met the criteria of the rule.
Conclusions: The Altman Rule is a reasonable proxy for GL and can be a useful and accessible tool for consumers interested in buying healthier packaged carbohydrate foods.
Nutrition can be complicated for consumers interested in making healthier choices at the grocery store. Consumers may have difficulty identifying more nutritious options, especially when food labels are adorned with claims such as “Good Source of Fiber” or “Heart Healthy.”1 In addition, when reading food labels, consumers may find it difficult to decipher which data to prioritize when carbohydrates, total sugars, added sugars, total dietary fiber, soluble fiber, and insoluble fiber are all listed.
The concept of glycemic load (GL) is an important consideration, especially for people with diabetes. GL approximates the blood sugar response to different foods. A food with a high GL is digested quickly, and its carbohydrates are taken into the bloodstream rapidly. This leads to a spike and subsequent drop in blood sugars, which can cause symptoms of hyperglycemia and hypoglycemia in a person with diabetes.2,3 Despite its usefulness, GL may be too complicated for a consumer to understand, and it does not appear anywhere on the food label. Since GL is calculated using pooled blood sugar response from individuals after the ingestion of the particular food, estimation of the GL is not intuitable.4
Point-of-sale tools. People seeking to lose weight, control diabetes, improve dyslipidemia and/or blood pressure, and/or decrease their risk for heart disease may benefit from point-of-sale tools such as the Altman Rule, which simplifies and encourages the selection of more nutritious foods.1 Other tools—such as Guiding Stars (https://guidingstars.com), NuVal (www.nuval.com), and different variations of traffic lights—have been created to help consumers make more informed and healthier food choices.5-8 However, Guiding Stars and NuVal are based on complicated algorithms that are not entirely transparent and not accessible to the average consumer.6,7 Evaluations of these nutrition tools indicate that consumers tend to underrate the healthiness of some foods, such as raw almonds and salmon, and overrate the healthiness of others, such as fruit punch and diet soda, when using traffic light systems.6 Furthermore, these nutrition tools are not available in many supermarkets. Previous research suggests that the use of point-of-sale nutrition apps decreases with the time and effort involved in using an app.9
Continue to: The Altman Rule
The Altman Rule was developed by a family physician (author WA) to provide a more accessible tool for people interested in choosing healthier prepackaged carbohydrate foods while shopping. Since the user does not need to have a smartphone, and they are not required to download or understand an app for each purchase, the Altman Rule may be more usable compared with more complicated alternatives.

The Altman Rule can be used with nutrition labels that feature serving information and calories in enlarged and bold type, in compliance with the most recent US Food and Drug Administration (FDA) guideline from 2016. Many foods with high fiber also have high amounts of sugar, so the criteria of the Altman Rule includes a 2-step process requiring (1) a minimum of 3 g of total dietary fiber per serving and (2) the sum of the grams of fiber plus the grams of protein per serving to be greater than the total grams of sugar (not grams of added sugar or grams of carbohydrate) per serving (FIGURE 1A). Unlike the relatively complicated formula related to GL, this 2-part rule can be applied in seconds while shopping (FIGURE 1B).

The rule is intended only to be used for packaged carbohydrate products, such as bread, muffins, bagels, pasta, rice, oatmeal, cereals, snack bars, chips, and crackers. It does not apply to whole foods, such as meat, dairy, fruits, or vegetables. These foods are excluded to prevent any consumer confusion related to the nutritional content of whole foods (eg, an apple may have more sugar than fiber and protein combined, but it is still a nutritious option).
This study aimed to determine if the Altman Rule is a reasonable proxy for the more complicated concept of GL. We calculated the relationship between the GL of commercially available packaged carbohydrate foods and whether those foods met the Altman Rule.
METHODS
The Altman Rule was tested by comparing the binary outcome of the rule (meets/does not meet) with data on all foods categorized as cereals, chips, crackers, and granola bars in the Nutrition Data System for Research (NDSR) Database (University of Minnesota, Version 2010).
Continue to: To account for differences...
To account for differences in serving size, we used the standard of 50 g for each product as 1 serving. We used 50 g (about 1.7 oz) to help compare the different foods and between foods within the same group. Additionally, 50 g is close to 1 serving for most foods in these groups; it is about the size of a typical granola bar, three-quarters to 2 cups of cereal, 10 to 12 crackers, and 15 to 25 chips. We determined the GL for each product by multiplying the number of available carbohydrates (total carbohydrate – dietary fiber) by the product’s glycemic index/100. In general, GL is categorized as low (≤ 10), medium (11-19), or high (≥ 20).
We applied the Altman Rule to categorize each product as meeting or not meeting the rule. We compared the proportion of foods meeting the Altman Rule, stratified by GL and by specific foods, and used chi-square to determine if differences were statistically significant. These data were collected and analyzed in the summer of 2019.
RESULTS
There were 1235 foods (342 breakfast cereals, 305 chips, 379 crackers, and 209 granola bars) used for this analysis. There is a significant relationship between the GL of foods and the Altman Rule in that most low-GL (68%), almost half of medium-GL (48%), and only a few high-GL foods (7%) met the rule (P < .001) (TABLE 1). There was also a significant relationship between “meeting the Altman Rule” and GL within each food type (P < .001) (TABLE 2).

The medium-GL foods were the second largest category of foods we calculated; thus we further broke them into binary categories of

Foods that met the rule were more likely to be low GL and foods that did not pass the rule were more likely high GL. Within the medium-GL category, foods that met the rule were more likely to be low-medium GL.
Continue to: The findings within food categories...
The findings within food categories showed that very few cereals, chips, crackers, and granola bars were low GL. For every food category, except granola bars, far more low-GL foods met the Altman Rule than those that did not. At the same time, very few high-GL foods met the Altman Rule. The category with the most individual high-GL food items meeting the Altman Rule was cereal. This was also the subcategory with the largest percentage of high-GL food items meeting the Altman Rule. Thirty-nine cereals that were high GL met the rule, but more than 4 times as many high-GL cereals did not (n = 190).
DISCUSSION
Marketing and nutrition messaging create consumer confusion that makes it challenging to identify packaged food items that are more nutrient dense. The Altman Rule simplifies food choices that have become unnecessarily complex. Our findings suggest this 2-step rule is a reasonable proxy for the more complicated and less accessible GL for packaged carbohydrates, such as cereals, chips, crackers, and snack bars. Foods that meet the rule are likely low or low-medium GL and thus are foods that are likely to be healthier choices.
Of note, only 9% of chips (n = 27) passed the Altman Rule, likely due to their low dietary fiber content, which was typical of chips. If a food item does not have at least 3 grams of total dietary fiber per serving, it does not pass the Altman Rule, regardless of how much protein or sugar is in the product. This may be considered a strength or a weakness of the Altman Rule. Few nutrition-dense foods are low in fiber, but some foods could be nutritious but do not meet the Altman Rule due to having < 3 g of fiber.
With the high prevalence of chronic diseases such as hypertension, diabetes, hyperlipidemia, and cardiovascular disease, it is essential to help consumers prevent chronic disease altogether or manage their chronic disease by providing tools to identify healthier food choices. The tool also has a place in clinical medicine for use by physicians and other health care professionals. Research shows that physicians find both time and lack of knowledge/resources to be a barrier to providing nutritional counseling to patients.10 Since the Altman Rule can be shared and explained with very little time and without extensive nutritional knowledge, it meets these needs.
Limitations
Glycemic load. We acknowledge that the Altman Rule is not foolproof and that assessing this rule based on GL has some limitations. GL is not a perfect or comprehensive way to measure the nutritional value of a food. For example, fruits such as watermelon and grapes are nutritionally dense. However, they contain high amounts of natural sugars—and as such, their GL is relatively high, which could lead a consumer to perceive them as unhealthy. Nevertheless, GL is both a useful and accepted tool and a reasonable way to assess the validity of the rule, specifically when assessing packaged carbohydrates. The simplicity of the Altman Rule and its relationship with GL makes it such that consumers are more likely to make a healthier food choice using it.9
Continue to: Specificity and sensitivity
Specificity and sensitivity. There are other limitations to the Altman Rule, given that a small number of high-GL foods meet the rule. For example, some granola bars had high dietary protein, which offset a high sugar content just enough to pass the rule despite a higher GL. As such, concluding that a snack bar is a healthier choice because it meets the Altman Rule when it has high amounts of sugar may not be appropriate. This limitation could be considered a lack of specificity (the rule includes food it ought not to include). Another limitation to consider would be a lack of sensitivity, given that only 68% of low-GL foods passed the Altman Rule. Since GL is associated with carbohydrate content, foods with a low carbohydrate count often have little to no fiber and thus would fall into the category of foods that did not meet the Altman Rule but had low GL. In this case, however, the low amount of fiber may render the Altman Rule a better indicator of a healthier food choice than the GL.
Hidden sugars. Foods with sugar alcohols and artificial sweeteners may be as deleterious as caloric alternatives while not being accounted for when reporting the grams of sugar per serving on the nutrition label.7 This may represent an exception to the Altman Rule, as foods that are not healthier choices may pass the rule because the sugar content on the nutrition label is, in a sense, artificially lowered. Future research may investigate the hypothesis that these foods are nutritionally inferior despite meeting the Altman Rule.
The sample. Our study also was limited to working only with foods that were included in the NDSR database up to 2010. This limitation is mitigated by the fact that the sample size was large (> 1000 packaged food items were included in our analyses). The study also could be limited by the food categories that were analyzed; food categories such as bread, rice, pasta, and bagels were not included.
The objective of this research was to investigate the relationship between GL and the Altman Rule, rather than to conduct an exhaustive analysis of the Altman Rule for every possible food category. Studying the relationship between the Altman Rule and GL in other categories of food is an objective for future research. The data so far support a relationship between these entities. The likelihood of the nutrition facts of foods changing without the GL changing (or vice versa) is very low. As such, the Altman Rule still seems to be a reasonable proxy of GL.
CONCLUSIONS
Research indicates that point-of-sale tools, such as Guiding Stars, NuVal, and other stoplight tools, can successfully alter consumers’ behaviors.9 These tools can be helpful but are not available in many supermarkets. Despite the limitations, the Altman Rule is a useful decision aid that is accessible to all consumers no matter where they live or shop and is easy to use and remember.
The Altman rule can be used in clinical practice by health care professionals, such as physicians, nurse practitioners, physician assistants, dietitians, and health coaches. It also has the potential to be used in commercial settings, such as grocery stores, to help consumers easily identify healthier convenience foods. This has public health implications, as the rule can both empower consumers and potentially incentivize food manufacturers to upgrade their products nutritionally.
Additional research would be useful to evaluate consumers’ preferences and perceptions about how user-friendly the Altman Rule is at the point of sale with packaged carbohydrate foods. This would help to further understand how the use of information on food packaging can motivate healthier decisions—thereby helping to alleviate the burden of chronic disease.
CORRESPONDENCE
Kimberly R. Dong, DrPH, MS, RDN, Tufts University School of Medicine, Department of Public Health and Community Medicine, 136 Harrison Avenue, MV Building, Boston, MA 02111; kimberly.dong@tufts.edu
ABSTRACT
Background: The Altman Rule, a simple tool for consumers seeking to make healthier packaged food choices at the point of sale, applies to packaged carbohydrates. According to the Altman Rule, a food is a healthier option if it has at least 3 g of fiber per serving and the grams of fiber plus the grams of protein exceed the grams of sugar per serving. This study sought to evaluate whether the Altman Rule is a valid proxy for glycemic load (GL).
Methods: We compared the binary outcome of whether a food item meets the Altman Rule with the GL of all foods categorized as cereals, chips, crackers, and granola bars in the Nutrition Data System for Research Database (University of Minnesota, Version 2010). We examined the percentage of foods in low-, medium-, and high-GL categories that met the Altman Rule.
Results: There were 1235 foods (342 cereals, 305 chips, 379 crackers, and 209 granola bars) in this analysis. There was a significant relationship between the GL of foods and the Altman Rule (P < .001) in that most low-GL (68%), almost half of medium-GL (48%), and very few high-GL (7%) foods met the criteria of the rule.
Conclusions: The Altman Rule is a reasonable proxy for GL and can be a useful and accessible tool for consumers interested in buying healthier packaged carbohydrate foods.
Nutrition can be complicated for consumers interested in making healthier choices at the grocery store. Consumers may have difficulty identifying more nutritious options, especially when food labels are adorned with claims such as “Good Source of Fiber” or “Heart Healthy.”1 In addition, when reading food labels, consumers may find it difficult to decipher which data to prioritize when carbohydrates, total sugars, added sugars, total dietary fiber, soluble fiber, and insoluble fiber are all listed.
The concept of glycemic load (GL) is an important consideration, especially for people with diabetes. GL approximates the blood sugar response to different foods. A food with a high GL is digested quickly, and its carbohydrates are taken into the bloodstream rapidly. This leads to a spike and subsequent drop in blood sugars, which can cause symptoms of hyperglycemia and hypoglycemia in a person with diabetes.2,3 Despite its usefulness, GL may be too complicated for a consumer to understand, and it does not appear anywhere on the food label. Since GL is calculated using pooled blood sugar response from individuals after the ingestion of the particular food, estimation of the GL is not intuitable.4
Point-of-sale tools. People seeking to lose weight, control diabetes, improve dyslipidemia and/or blood pressure, and/or decrease their risk for heart disease may benefit from point-of-sale tools such as the Altman Rule, which simplifies and encourages the selection of more nutritious foods.1 Other tools—such as Guiding Stars (https://guidingstars.com), NuVal (www.nuval.com), and different variations of traffic lights—have been created to help consumers make more informed and healthier food choices.5-8 However, Guiding Stars and NuVal are based on complicated algorithms that are not entirely transparent and not accessible to the average consumer.6,7 Evaluations of these nutrition tools indicate that consumers tend to underrate the healthiness of some foods, such as raw almonds and salmon, and overrate the healthiness of others, such as fruit punch and diet soda, when using traffic light systems.6 Furthermore, these nutrition tools are not available in many supermarkets. Previous research suggests that the use of point-of-sale nutrition apps decreases with the time and effort involved in using an app.9
Continue to: The Altman Rule
The Altman Rule was developed by a family physician (author WA) to provide a more accessible tool for people interested in choosing healthier prepackaged carbohydrate foods while shopping. Since the user does not need to have a smartphone, and they are not required to download or understand an app for each purchase, the Altman Rule may be more usable compared with more complicated alternatives.

The Altman Rule can be used with nutrition labels that feature serving information and calories in enlarged and bold type, in compliance with the most recent US Food and Drug Administration (FDA) guideline from 2016. Many foods with high fiber also have high amounts of sugar, so the criteria of the Altman Rule includes a 2-step process requiring (1) a minimum of 3 g of total dietary fiber per serving and (2) the sum of the grams of fiber plus the grams of protein per serving to be greater than the total grams of sugar (not grams of added sugar or grams of carbohydrate) per serving (FIGURE 1A). Unlike the relatively complicated formula related to GL, this 2-part rule can be applied in seconds while shopping (FIGURE 1B).

The rule is intended only to be used for packaged carbohydrate products, such as bread, muffins, bagels, pasta, rice, oatmeal, cereals, snack bars, chips, and crackers. It does not apply to whole foods, such as meat, dairy, fruits, or vegetables. These foods are excluded to prevent any consumer confusion related to the nutritional content of whole foods (eg, an apple may have more sugar than fiber and protein combined, but it is still a nutritious option).
This study aimed to determine if the Altman Rule is a reasonable proxy for the more complicated concept of GL. We calculated the relationship between the GL of commercially available packaged carbohydrate foods and whether those foods met the Altman Rule.
METHODS
The Altman Rule was tested by comparing the binary outcome of the rule (meets/does not meet) with data on all foods categorized as cereals, chips, crackers, and granola bars in the Nutrition Data System for Research (NDSR) Database (University of Minnesota, Version 2010).
Continue to: To account for differences...
To account for differences in serving size, we used the standard of 50 g for each product as 1 serving. We used 50 g (about 1.7 oz) to help compare the different foods and between foods within the same group. Additionally, 50 g is close to 1 serving for most foods in these groups; it is about the size of a typical granola bar, three-quarters to 2 cups of cereal, 10 to 12 crackers, and 15 to 25 chips. We determined the GL for each product by multiplying the number of available carbohydrates (total carbohydrate – dietary fiber) by the product’s glycemic index/100. In general, GL is categorized as low (≤ 10), medium (11-19), or high (≥ 20).
We applied the Altman Rule to categorize each product as meeting or not meeting the rule. We compared the proportion of foods meeting the Altman Rule, stratified by GL and by specific foods, and used chi-square to determine if differences were statistically significant. These data were collected and analyzed in the summer of 2019.
RESULTS
There were 1235 foods (342 breakfast cereals, 305 chips, 379 crackers, and 209 granola bars) used for this analysis. There is a significant relationship between the GL of foods and the Altman Rule in that most low-GL (68%), almost half of medium-GL (48%), and only a few high-GL foods (7%) met the rule (P < .001) (TABLE 1). There was also a significant relationship between “meeting the Altman Rule” and GL within each food type (P < .001) (TABLE 2).

The medium-GL foods were the second largest category of foods we calculated; thus we further broke them into binary categories of

Foods that met the rule were more likely to be low GL and foods that did not pass the rule were more likely high GL. Within the medium-GL category, foods that met the rule were more likely to be low-medium GL.
Continue to: The findings within food categories...
The findings within food categories showed that very few cereals, chips, crackers, and granola bars were low GL. For every food category, except granola bars, far more low-GL foods met the Altman Rule than those that did not. At the same time, very few high-GL foods met the Altman Rule. The category with the most individual high-GL food items meeting the Altman Rule was cereal. This was also the subcategory with the largest percentage of high-GL food items meeting the Altman Rule. Thirty-nine cereals that were high GL met the rule, but more than 4 times as many high-GL cereals did not (n = 190).
DISCUSSION
Marketing and nutrition messaging create consumer confusion that makes it challenging to identify packaged food items that are more nutrient dense. The Altman Rule simplifies food choices that have become unnecessarily complex. Our findings suggest this 2-step rule is a reasonable proxy for the more complicated and less accessible GL for packaged carbohydrates, such as cereals, chips, crackers, and snack bars. Foods that meet the rule are likely low or low-medium GL and thus are foods that are likely to be healthier choices.
Of note, only 9% of chips (n = 27) passed the Altman Rule, likely due to their low dietary fiber content, which was typical of chips. If a food item does not have at least 3 grams of total dietary fiber per serving, it does not pass the Altman Rule, regardless of how much protein or sugar is in the product. This may be considered a strength or a weakness of the Altman Rule. Few nutrition-dense foods are low in fiber, but some foods could be nutritious but do not meet the Altman Rule due to having < 3 g of fiber.
With the high prevalence of chronic diseases such as hypertension, diabetes, hyperlipidemia, and cardiovascular disease, it is essential to help consumers prevent chronic disease altogether or manage their chronic disease by providing tools to identify healthier food choices. The tool also has a place in clinical medicine for use by physicians and other health care professionals. Research shows that physicians find both time and lack of knowledge/resources to be a barrier to providing nutritional counseling to patients.10 Since the Altman Rule can be shared and explained with very little time and without extensive nutritional knowledge, it meets these needs.
Limitations
Glycemic load. We acknowledge that the Altman Rule is not foolproof and that assessing this rule based on GL has some limitations. GL is not a perfect or comprehensive way to measure the nutritional value of a food. For example, fruits such as watermelon and grapes are nutritionally dense. However, they contain high amounts of natural sugars—and as such, their GL is relatively high, which could lead a consumer to perceive them as unhealthy. Nevertheless, GL is both a useful and accepted tool and a reasonable way to assess the validity of the rule, specifically when assessing packaged carbohydrates. The simplicity of the Altman Rule and its relationship with GL makes it such that consumers are more likely to make a healthier food choice using it.9
Continue to: Specificity and sensitivity
Specificity and sensitivity. There are other limitations to the Altman Rule, given that a small number of high-GL foods meet the rule. For example, some granola bars had high dietary protein, which offset a high sugar content just enough to pass the rule despite a higher GL. As such, concluding that a snack bar is a healthier choice because it meets the Altman Rule when it has high amounts of sugar may not be appropriate. This limitation could be considered a lack of specificity (the rule includes food it ought not to include). Another limitation to consider would be a lack of sensitivity, given that only 68% of low-GL foods passed the Altman Rule. Since GL is associated with carbohydrate content, foods with a low carbohydrate count often have little to no fiber and thus would fall into the category of foods that did not meet the Altman Rule but had low GL. In this case, however, the low amount of fiber may render the Altman Rule a better indicator of a healthier food choice than the GL.
Hidden sugars. Foods with sugar alcohols and artificial sweeteners may be as deleterious as caloric alternatives while not being accounted for when reporting the grams of sugar per serving on the nutrition label.7 This may represent an exception to the Altman Rule, as foods that are not healthier choices may pass the rule because the sugar content on the nutrition label is, in a sense, artificially lowered. Future research may investigate the hypothesis that these foods are nutritionally inferior despite meeting the Altman Rule.
The sample. Our study also was limited to working only with foods that were included in the NDSR database up to 2010. This limitation is mitigated by the fact that the sample size was large (> 1000 packaged food items were included in our analyses). The study also could be limited by the food categories that were analyzed; food categories such as bread, rice, pasta, and bagels were not included.
The objective of this research was to investigate the relationship between GL and the Altman Rule, rather than to conduct an exhaustive analysis of the Altman Rule for every possible food category. Studying the relationship between the Altman Rule and GL in other categories of food is an objective for future research. The data so far support a relationship between these entities. The likelihood of the nutrition facts of foods changing without the GL changing (or vice versa) is very low. As such, the Altman Rule still seems to be a reasonable proxy of GL.
CONCLUSIONS
Research indicates that point-of-sale tools, such as Guiding Stars, NuVal, and other stoplight tools, can successfully alter consumers’ behaviors.9 These tools can be helpful but are not available in many supermarkets. Despite the limitations, the Altman Rule is a useful decision aid that is accessible to all consumers no matter where they live or shop and is easy to use and remember.
The Altman rule can be used in clinical practice by health care professionals, such as physicians, nurse practitioners, physician assistants, dietitians, and health coaches. It also has the potential to be used in commercial settings, such as grocery stores, to help consumers easily identify healthier convenience foods. This has public health implications, as the rule can both empower consumers and potentially incentivize food manufacturers to upgrade their products nutritionally.
Additional research would be useful to evaluate consumers’ preferences and perceptions about how user-friendly the Altman Rule is at the point of sale with packaged carbohydrate foods. This would help to further understand how the use of information on food packaging can motivate healthier decisions—thereby helping to alleviate the burden of chronic disease.
CORRESPONDENCE
Kimberly R. Dong, DrPH, MS, RDN, Tufts University School of Medicine, Department of Public Health and Community Medicine, 136 Harrison Avenue, MV Building, Boston, MA 02111; kimberly.dong@tufts.edu
1. Hersey JC, Wohlgenant KC, Arsenault JE, et al. Effects of front-of-package and shelf nutrition labeling systems on consumers. Nutr Rev. 2013;71:1-14. doi: 10.1111/nure.12000
2. Jenkins DJA, Dehghan M, Mente A, et al. Glycemic index, glycemic load, and cardiovascular disease and mortality. N Engl J Med. 2021;384:1312-1322. doi: 10.1056/NEJMoa2007123
3. Brand-Miller J, Hayne S, Petocz P, et al. Low–glycemic index diets in the management of diabetes. Diabetes Care. 2003;26:2261-2267. doi: 10.2337/diacare.26.8.2261
4. Matthan NR, Ausman LM, Meng H, et al. Estimating the reliability of glycemic index values and potential sources of methodological and biological variability. Am J Clin Nutr. 2016;104:1004-1013. doi: 10.3945/ajcn.116.137208
5. Sonnenberg L, Gelsomin E, Levy DE, et al. A traffic light food labeling intervention increases consumer awareness of health and healthy choices at the point-of-purchase. Prev Med. 2013;57:253-257. doi: 10.1016/j.ypmed.2013.07.001
6. Savoie N, Barlow K, Harvey KL, et al. Consumer perceptions of front-of-package labelling systems and healthiness of foods. Can J Public Health. 2013;104:e359-e363. doi: 10.17269/cjph.104.4027
7. Fischer LM, Sutherland LA, Kaley LA, et al. Development and implementation of the Guiding Stars nutrition guidance program. Am J Health Promot. 2011;26:e55-e63. doi: 10.4278/ajhp.100709-QUAL-238
8. Maubach N, Hoek J, Mather D. Interpretive front-of-pack nutrition labels. Comparing competing recommendations. Appetite. 2014;82:67-77. doi: 10.1016/j.appet.2014.07.006
9. Chan J, McMahon E, Brimblecombe J. Point‐of‐sale nutrition information interventions in food retail stores to promote healthier food purchase and intake: a systematic review. Obes Rev. 2021;22. doi: 10.1111/obr.13311
10. Mathioudakis N, Bashura H, Boyér L, et al. Development, implementation, and evaluation of a physician-targeted inpatient glycemic management curriculum. J Med Educ Curric Dev. 2019;6:238212051986134. doi: 10.1177/2382120519861342
1. Hersey JC, Wohlgenant KC, Arsenault JE, et al. Effects of front-of-package and shelf nutrition labeling systems on consumers. Nutr Rev. 2013;71:1-14. doi: 10.1111/nure.12000
2. Jenkins DJA, Dehghan M, Mente A, et al. Glycemic index, glycemic load, and cardiovascular disease and mortality. N Engl J Med. 2021;384:1312-1322. doi: 10.1056/NEJMoa2007123
3. Brand-Miller J, Hayne S, Petocz P, et al. Low–glycemic index diets in the management of diabetes. Diabetes Care. 2003;26:2261-2267. doi: 10.2337/diacare.26.8.2261
4. Matthan NR, Ausman LM, Meng H, et al. Estimating the reliability of glycemic index values and potential sources of methodological and biological variability. Am J Clin Nutr. 2016;104:1004-1013. doi: 10.3945/ajcn.116.137208
5. Sonnenberg L, Gelsomin E, Levy DE, et al. A traffic light food labeling intervention increases consumer awareness of health and healthy choices at the point-of-purchase. Prev Med. 2013;57:253-257. doi: 10.1016/j.ypmed.2013.07.001
6. Savoie N, Barlow K, Harvey KL, et al. Consumer perceptions of front-of-package labelling systems and healthiness of foods. Can J Public Health. 2013;104:e359-e363. doi: 10.17269/cjph.104.4027
7. Fischer LM, Sutherland LA, Kaley LA, et al. Development and implementation of the Guiding Stars nutrition guidance program. Am J Health Promot. 2011;26:e55-e63. doi: 10.4278/ajhp.100709-QUAL-238
8. Maubach N, Hoek J, Mather D. Interpretive front-of-pack nutrition labels. Comparing competing recommendations. Appetite. 2014;82:67-77. doi: 10.1016/j.appet.2014.07.006
9. Chan J, McMahon E, Brimblecombe J. Point‐of‐sale nutrition information interventions in food retail stores to promote healthier food purchase and intake: a systematic review. Obes Rev. 2021;22. doi: 10.1111/obr.13311
10. Mathioudakis N, Bashura H, Boyér L, et al. Development, implementation, and evaluation of a physician-targeted inpatient glycemic management curriculum. J Med Educ Curric Dev. 2019;6:238212051986134. doi: 10.1177/2382120519861342
Migraine headache: When to consider these newer agents
Migraine is a headache disorder that often causes unilateral pain, photophobia, phonophobia, nausea, and vomiting. More than 70% of office visits for migraine are made to primary care physicians.1 Recent data suggest migraine may be caused primarily by neuronal dysfunction and only secondarily by vasodilation.2 Although there are numerous classes of drugs used for migraine prevention and treatment, their success has been limited by inadequate efficacy, tolerability, and patient adherence.3 The discovery of pro-inflammatory markers such as calcitonin gene-related peptide (CGRP) has led to the development of new medications to prevent and treat migraine.4

Pathophysiology, Dx and triggers, indications for pharmacotherapy
Pathophysiology. A migraine is thought to be caused by cortical spreading depression (CSD), a depolarization of glial and neuronal cell membranes.5
Dx and triggers. In 2018, the International Headache Society revised its guidelines for the diagnosis of migraine.7 According to the 3rd edition of The International Classification of Headache Disorders (ICHD-3), the diagnosis of migraine is made when a patient has at least 5 headache attacks that last 4 to 72 hours and have at least 2 of the following characteristics: (1) unilateral location, (2) pulsating quality, (3) moderate-to-severe pain intensity, and (4) aggravated by or causing avoidance of routine physical activity.7 The headache attacks also should have (1) associated nausea or vomiting or (2) photophobia and phonophobia.7 The presence of atypical signs or symptoms as indicated by the SNNOOP10 mnemonic raises concerns for secondary headaches and the need for further investigation into the cause of the headache (TABLE 1).8 It is not possible to detect every secondary headache with standard neuroimaging, but the SNNOOP10 red flags can help determine when imaging may be indicated.8 Potential triggers for migraine can be found in TABLE 2.9
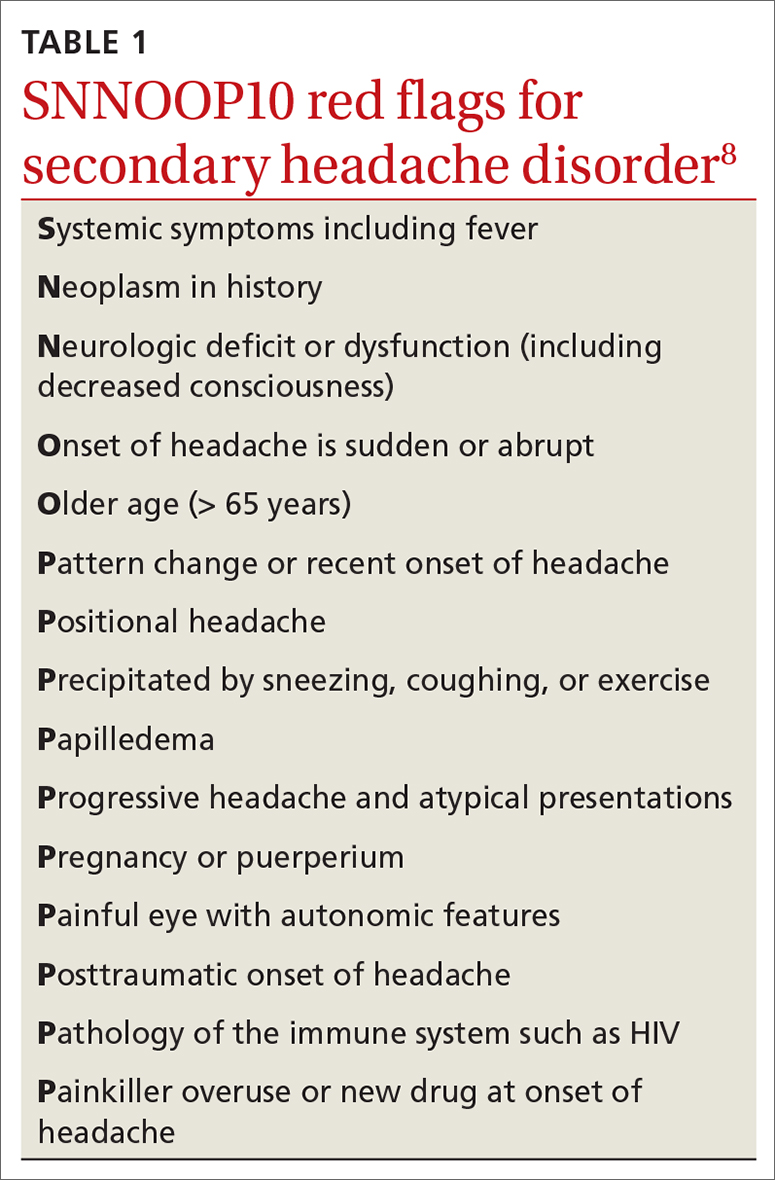
Indications for pharmacotherapy. All patients receiving a diagnosis of migraine should be offered acute pharmacologic treatment. Consider preventive therapy anytime there are ≥ 4 headache days per month, debilitating attacks despite acute therapy, overuse of acute medication (> 2 d/wk), difficulty tolerating acute medication, patient preference, or presence of certain migraine subtypes.7,10

Acute treatments
Abortive therapies for migraine include analgesics such as nonsteroidal anti-inflammatory drugs (NSAIDs) and acetaminophen, and ergot alkaloids, triptans, or small-molecule CGRP receptor antagonists (gepants). Prompt administration increases the chance of success with acute therapy. Medications with the highest levels of efficacy based on the 2015 guidelines from the American Headache Society (AHS) are given in TABLE 3.11 Lasmiditan (Reyvow) is not included in the 2015 guidelines, as it was approved after publication of the guidelines.
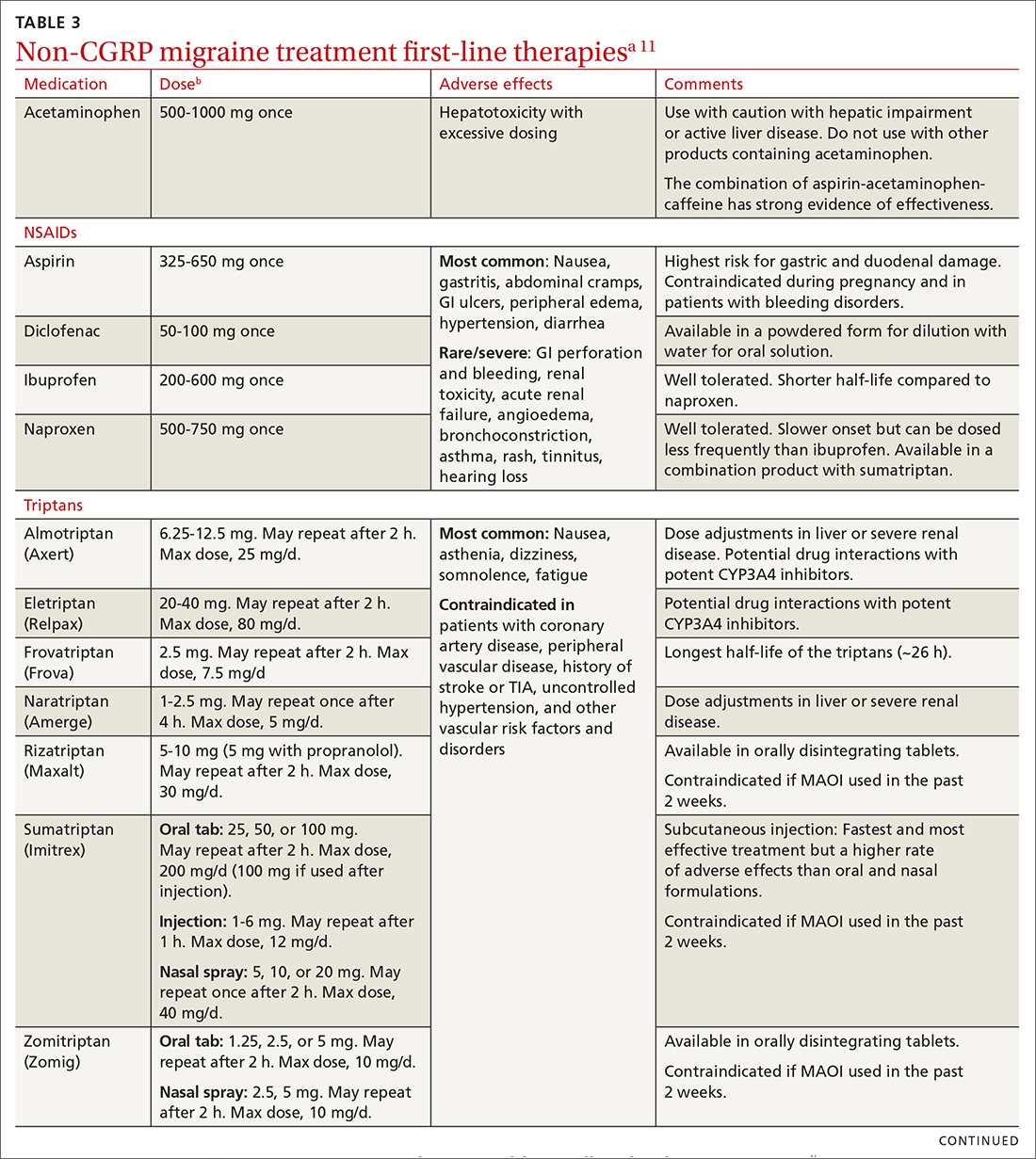
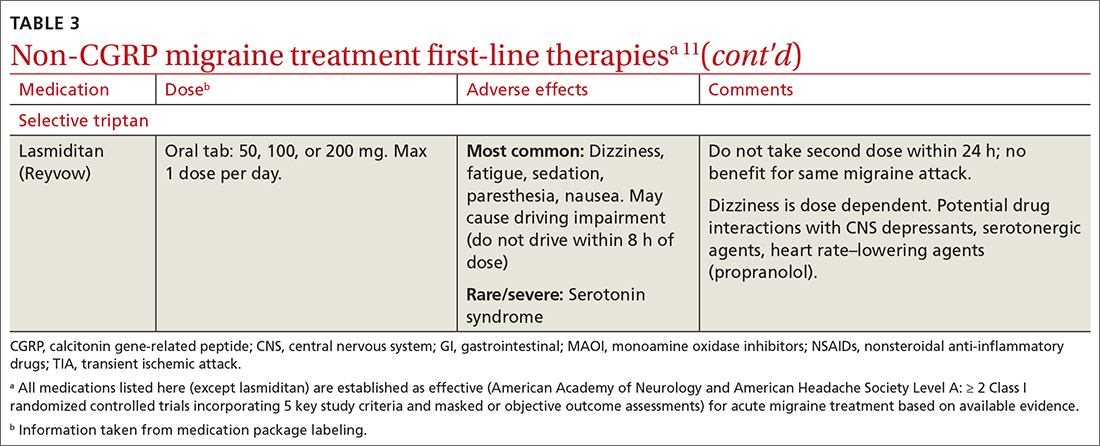
Non-CGRP first-line therapies
NSAIDs and acetaminophen. NSAIDs such as aspirin, diclofenac, ibuprofen, and naproxen have a high level of evidence to support their use as first-line treatments for mild-to-moderate migraine attacks. Trials consistently demonstrate their superiority to placebo in headache relief and complete pain relief at 2 hours. There is no recommendation for selecting one NSAID over another; however, consider their frequency of dosing and adverse effect profiles. The number needed to treat for complete pain relief at 2 hours ranges from 7 to 10 for most NSAIDs.11,12 In some placebo-controlled studies, acetaminophen was less effective than NSAIDs, but was safer because it did not cause gastric irritation or antiplatelet effects.12
Triptans inhibit 5-HT1B/1D receptors. Consider formulation, route of administration, cost, and pharmacokinetics when selecting a triptan. Patients who do not respond well to one triptan may respond favorably to another. A meta-analysis of the effectiveness of the 7 available agents found that triptans at standard doses provided pain relief within 2 hours in 42% to 76% of patients, and sustained freedom from pain for 2 hours in 18% to 50% of patients.13 Lasmiditan is a selective serotonin receptor (5-HT1F) agonist that lacks vasoconstrictor activity. This is an option for patients with relative contraindications to triptans due to cardiovascular risk factors.10
Continue to: Second-line therapies
Second-line therapies
Intranasal dihydroergotamine has a favorable adverse event profile and greater evidence for efficacy compared with ergotamine. Compared with triptans, intranasal dihydroergotamine has a high level of efficacy but causes more adverse effects.14 Severe nausea is common, and dihydroergotamine often is used in combination with an antiemetic drug. Dihydroergotamine should not be used within 24 hours of taking a triptan, and it is contraindicated for patients who have hypertension or ischemic heart disease or who are pregnant or breastfeeding. There is also the potential for adverse drug interactions.15
Antiemetics may be helpful for migraine associated with severe nausea or vomiting. The dopamine antagonists metoclopramide, prochlorperazine, and chlorpromazine have demonstrated benefit in randomized placebo-controlled trials.11 Ondansetron has not been studied extensively, but sometimes is used in clinical practice. Nonoral routes of administration may be useful in patients having trouble swallowing medications or in those experiencing significant nausea or vomiting early during migraine attacks.
Due to the high potential for abuse, opioids should not be used routinely for the treatment of migraine.12 There is no high-quality evidence supporting the efficacy of barbiturates (ie, butalbital-containing compounds) for acute migraine treatment.11 Moreover, use of these agents may increase the likelihood of progression from episodic to chronic migraine.16
Gepants for acute migraine treatment
Neuropeptide CGRP is released from trigeminal nerves and is a potent dilator of cerebral and dural vessels, playing a key role in regulating blood flow to the brain. Other roles of CGRP include the release of inflammatory agents from mast cells and the transmission of painful stimuli from intracranial vessels.17 The CGRP receptor or ligand can be targeted by small-molecule receptor antagonists for acute and preventive migraine treatment (and by monoclonal antibodies solely for prevention, discussed later). It has been theorized that gepants bind to CGRP receptors, resulting in decreased blood flow to the brain, inhibition of neurogenic inflammation, and reduced pain signaling.17 Unlike triptans and ergotamine derivatives, these novel treatments do not constrict blood vessels and may have a unique role in patients with contraindications to triptans.
The 3 gepants approved for acute treatment—ubrogepant (Ubrelvy),18 rimegepant (Nurtec),19 and zavegepant (Zavzpret)20—were compared with placebo in clinical trials and were shown to increase the number of patients who were completely pain free at 2 hours, were free of the most bothersome associated symptom (photophobia, phonophobia, or nausea) at 2 hours, and remained pain free at 24 hours (TABLE 418-24).
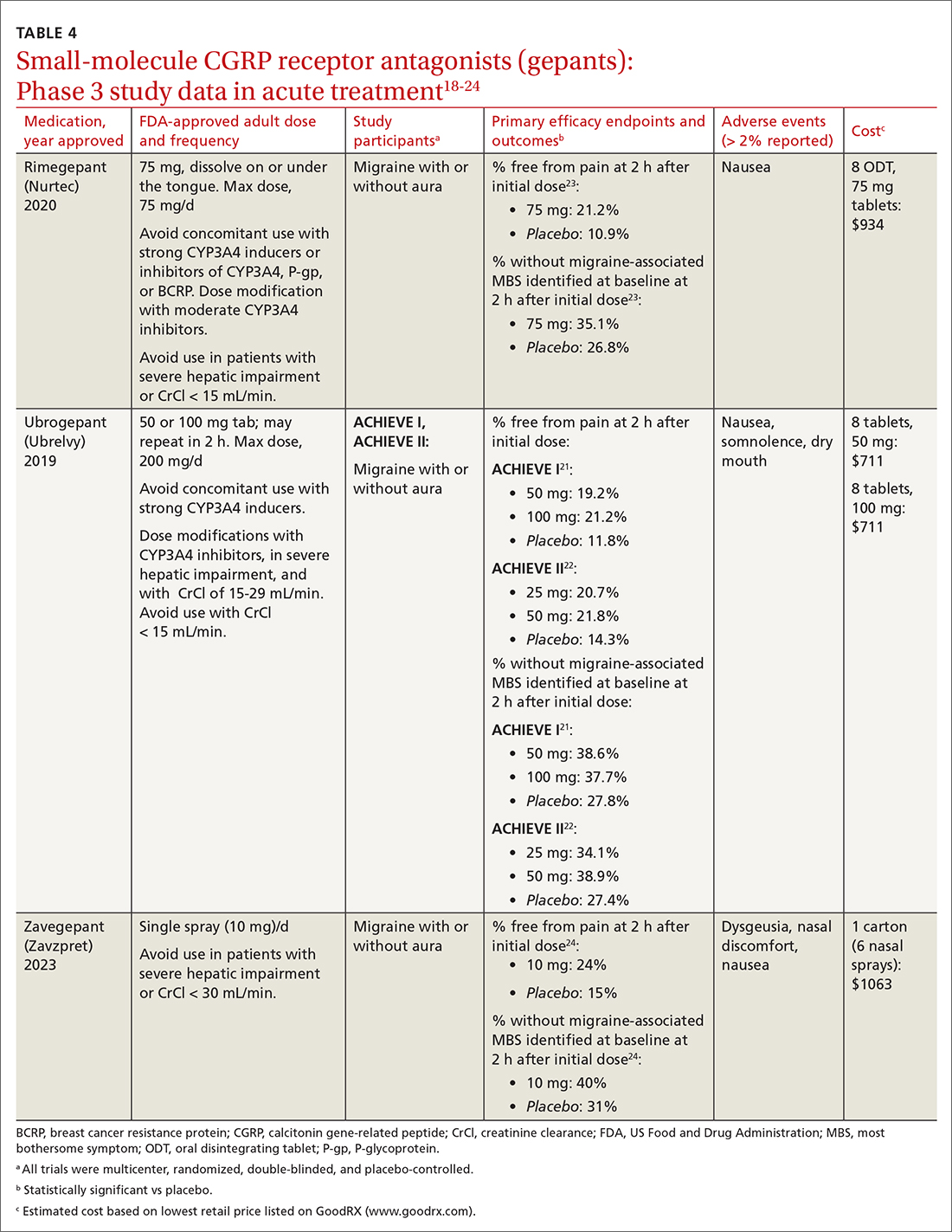
Continue to: Ubrogrepant
Ubrogepant, in 2 Phase 3 trials (ACHIEVE I and ACHIEVE II) demonstrated effectiveness compared with placebo.21,22 The most common adverse effects reported were nausea and somnolence at very low rates. Pain-relief rates at 2 hours post dose (> 60% of participants) were higher than pain-free rates, and a significantly higher percentage (> 40%) of ubrogepant-treated participants reported ability to function normally on the Functional Disability Scale.25
Rimegepant was also superior to placebo (59% vs 43%) in pain relief at 2 hours post dose and other secondary endpoints.23 Rimegepant also has potential drug interactions
Zavegepant, approved in March 2023, is administered once daily as a 10-mg nasal spray. In its Phase 3 trial, zavegepant was significantly superior to placebo at 2 hours post dose in freedom from pain (24% v 15%), and in freedom from the most bothersome symptom (40% v 31%).24 Dosage modifications are not needed with mild-to-moderate renal or hepatic disease.20
Worth noting. The safety of using ubrogepant to treat more than 8 migraine episodes in a 30-day period has not been established. The safety of using more than 18 doses of zavegepant in a 30-day period also has not been established. With ubrogepant and rimegepant, there are dosing modifications for concomitant use with specific drugs (CYP3A4 inhibitors and inducers) due to potential interactions and in patients with hepatic or renal impairment.18,19
There are no trials comparing efficacy of CGRP antagonists to triptans. Recognizing that these newer medications would be costly, the AHS position statement released in 2019 recommends that gepants be considered for those with contraindications to triptans or for whom at least 2 oral triptans have failed (as determined by a validated patient outcome questionnaire).10 Step therapy with documentation of previous trials and therapy failures is often required by insurance companies prior to gepant coverage.
Continue to: Preventive therapies
Preventive therapies
Preventive migraine therapies are used to reduce duration, frequency, and severity of attacks, the need for acute treatment, and overall headache disability.26 Medications typically are chosen based on efficacy, adverse effect profile, and patient comorbidities. Barriers to successful use include poor patient adherence and tolerability, the need for slow dose titration, and long-term use (minimum of 2 months) at maximum tolerated or minimum effective doses. Medications with established efficacy (Level Aa) based on the 2012 guidelines from the American Academy of Neurology (AAN) and the AHS are given in TABLE 5.27-29
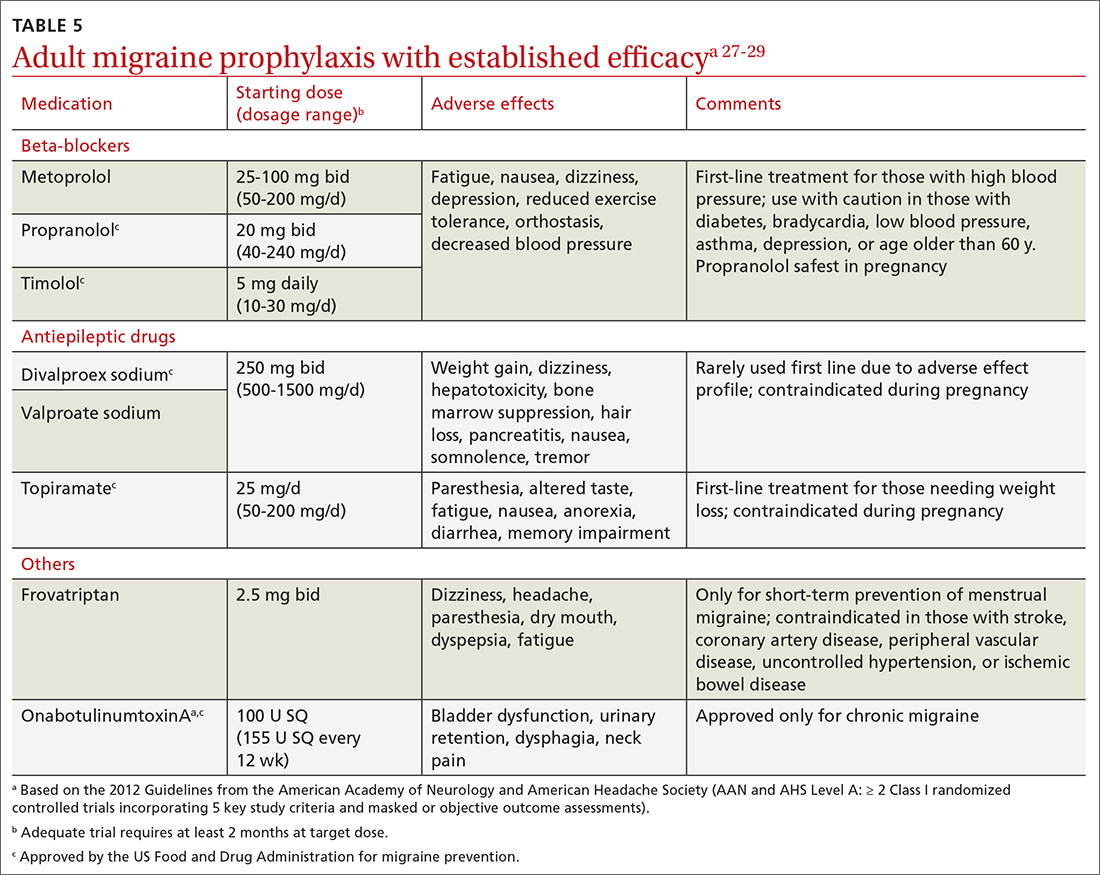
Drugs having received the strongest level of evidence for migraine prevention are metoprolol, propranolol, timolol, topiramate, valproate sodium, divalproex sodium, and onabotulinumtoxinA (Botox), and frovatriptan for menstrual migraine prevention. Because these guidelines were last updated in 2012, they did not cover gepants (which will be discussed shortly). The AHS released a position statement in 2019 supporting the use of
CGRP-targeted prevention
Four anti-CGRP mAbs and 2 gepants have been approved for migraine prevention in the United States. Differences between products include targets (ligand vs receptor), antibody IgG subtype, bioavailability, route of administration, and frequency of administration.28 As noted in the Phase 3 studies (TABLE 619,30-47), these therapies are highly efficacious, safe, and tolerable.
Gepants. Rimegepant, discussed earlier for migraine treatment, is one of the CGRP receptor antagonists approved for prevention. The other is atogepant (Qulipta), approved only for prevention. Ubrogepant is not approved for prevention.
Anti-CGRP mAb is the only medication class specifically created for migraine prevention.10,26 As already noted, several efficacious non-CGRP treatment options are available for migraine prevention. However, higher doses of those agents, if needed,
Continue to: The targeted anti-CGRP approach...
The targeted anti-CGRP approach, which can be used by patients with liver or kidney disease, results in decreased toxicity and minimal drug interactions. Long half-lives allow for monthly or quarterly injections, possibly resulting in increased compliance.28 Dose titration is not needed, allowing for more rapid symptom management. The large molecular size of a mAb limits its transfer across the blood-brain barrier, making central nervous system adverse effects unlikely.28 Despite the compelling mAb pharmacologic properties, their use may be limited by a lack of long-term safety data and the need for parenteral administration. Although immunogenicity—the development of neutralizing antibodies—can limit long-term tolerability or efficacy of mAbs generally,26,28 anti-CGRP mAbs were engineered to minimally activate the immune system and have not been associated with immune suppression, opportunistic infections, malignancies, or decreased efficacy.28
A pooled meta-analysis including 4 trials (3166 patients) found that CGRP mAbs compared with placebo significantly improved patient response rates, defined as at least a 50% and 75% reduction in monthly headache/migraine days from baseline to Weeks 9 to 12.48 Another meta-analysis including 8 trials (2292 patients) found a significant reduction from baseline in monthly migraine days and monthly acute migraine medication consumption among patients taking CGRP mAbs compared with those taking placebo.49 Open-label extension studies have shown progressive and cumulative benefits in individuals who respond to anti-CGRP mAbs. Therefore, several treatment cycles may be necessary to determine overall efficacy of therapy.10,28
Cost initially can be a barrier. Insurance companies often require step therapy before agreeing to cover mAb therapy, which aligns with the 2019 AHS position statement.10
When combination treatment may be appropriate
Monotherapy is the usual approach to preventing migraine due to advantages of efficacy, simplified regimens, lower cost, and reduced adverse effects.51 However, if a patient does not benefit from monotherapy even after trying dose titrations as tolerated or switching therapies, trying complementary combination therapy is appropriate. Despite a shortage of clinical trials supporting the use of 2 or more preventive medications with different mechanisms of action, this strategy is used clinically.10 Consider combination therapy in those with refractory disease, partial responses, or intolerance to recommended doses.52 Articles reporting on case study reviews have rationalized the combined use of onabotulinumtoxinA and anti-CGRP mAbs, noting better migraine control.51,53 The 2019 AHS position statement recommends adding a mAb to an existing preventive treatment regimen with no other changes until mAb effectiveness is determined, as the risk for drug interactions on dual therapy is low.10 Safety and efficacy also have been demonstrated with the combination of preventive anti-CGRP mAbs and acute treatment with gepants as needed.54
CORRESPONDENCE
Emily Peterson, PharmD, BCACP, 3640 Middlebury Road, Iowa City, IA 52242; Emily-a-peterson@uiowa.edu
1. Lipton RB, Nicholson RA, Reed ML, et al. Diagnosis, consultation, treatment, and impact of migraine in the US: results of the OVERCOME (US) study. Headache. 2022;62:122-140. doi: 10.1111/head.14259
2. Burstein R, Noseda R, Borsook D. Migraine: multiple processes; complext pathophysiology. J Neurosci. 2015;35:6619-6629. doi: 10.1523/JNEUROSCI.0373-15.2015
3. Edvinsson L, Haanes KA, Warfvinge K, et al. CGRP as the target of new migraine therapies - successful translation from bench to clinic. Nat Rev Neurol. 2018;14:338-350. doi: 10.1038/s41582-018-0003-1
4. McGrath K, Rague A, Thesing C, et al. Migraine: expanding our Tx arsenal. J Fam Pract. 2019;68:10-14;16-24.
5. Dodick DW. Migraine. Lancet. 2018;391:1315-1330. doi: 10.1016/S0140-6736(18)30478-1
6. Agostoni EC, Barbanti P, Calabresi P, et al. Current and emerging evidence-based treatment options in chronic migraine: a narrative review. J Headache Pain. 2019;20:92. doi: 10.1186/s10194-019-1038-4
7. IHS. Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211. doi: 10.1177/0333102417738202
8. Do TP, Remmers A, Schytz HW, et al. Red and orange flags for secondary headaches in clinical practice: SNNOOP10 list. Neurology. 2019;92:134-144. doi: 10.1212/WNL.0000000000006697
9. NIH. Migraine. Accessed July 30, 2023.
10. AHS. The American Headache Society position statement on integrating new migraine treatments into clinical practice. Headache. 2019;59:1-18. doi: 10.1111/head.13456
11. Marmura MJ, Silberstein SD, Schwedt TJ. The acute treatment of migraine in adults: the American Headache Society evidence assessment of migraine pharmacotherapies. Headache. 2015;55:3-20. doi: 10.1111/head.12499
12. Mayans L, Walling A. Acute migraine headache: treatment strategies. Am Fam Physician. 2018;97:243-251.
13. Cameron C, Kelly S, Hsieh SC, et al. Triptans in the acute treatment of migraine: a systematic review and network meta-analysis. Headache. 2015;55(suppl 4):221-235. doi: 10.1111/head.12601
14. Becker WJ. Acute migraine treatment. Continuum (Minneap Minn). 2015;21:953-972. doi: 10.1212/CON.0000000000000192
15. Migranal (dihydroergotamine mesylate) Package insert. Valeant Pharmaceuticals North America; 2019. Accessed June 17, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2019/020148Orig1s025lbl.pdf
16. Minen MT, Tanev K, Friedman BW. Evaluation and treatment of migraine in the emergency department: a review. Headache. 2014;54:1131-45. doi: 10.1111/head.12399
17. Durham PL. CGRP-receptor antagonists--a fresh approach to migraine therapy? N Engl J Med. 2004;350:1073-1075. doi: 10.1056/NEJMp048016
18. Ubrelvy (ubrogepant). Package insert. Allergan, Inc.; 2019. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2019/211765s000lbl.pdf
19. Nurtec ODT (rimegepant sulfate). Package insert. Biohaven Pharmaceuticals, Inc.; 2021. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2021/212728s006lbl.pdf
20. Zavzpret (zavegepant). Package insert. Pfizer Labs.; 2023. Accessed July 15, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2023/216386s000lbl.pdf
21. Dodick DW, Lipton RB, Ailani J, et al. Ubrogepant for the treatment of migraine. N Engl J Med. 2019;381:2230-2241. doi: 10.1056/NEJMoa1813049
22. Lipton RB, Dodick DW, Ailani J, et al. Effect of ubrogepant vs placebo on pain and the most bothersome associated symptom in the acute treatment of migraine: the ACHIEVE II randomized clinical trial. JAMA. 2019;322:1887-1898. doi: 10.1001/jama.2019.16711
23. Croop R, Goadsby PJ, Stock DA, et al. Efficacy, safety, and tolerability of rimegepant orally disintegrating tablet for the acute treatment of migraine: a randomised, phase 3, double-blind, placebo-controlled trial. Lancet. 2019;394:737-745. doi: 10.1016/S0140-6736(19)31606-X
24. Lipton RB, Croop R, Stock DA, et al. Safety, tolerability, and efficacy of zavegepant 10 mg nasal spray for the acute treatment of migraine in the USA: a phase 3, double-blind, randomised, placebo-controlled multicentre trial. Lancet Neurol. 2023;22:209-217. doi: 10.1016/S1474-4422(22)00517-8
25. Dodick DW, Lipton RB, Ailani J, et al. Ubrogepant, an acute treatment for migraine, improved patient-reported functional disability and satisfaction in 2 single-attack phase 3 randomized trials, ACHIEVE I and II. Headache. 2020;60:686-700. doi: 10.1111/head.13766
26. Burch R. Migraine and tension-type headache: diagnosis and treatment. Med Clin North Am. 2019;103:215-233. doi:10.1016/j.mcna.2018.10.003
27. Silberstein SD, Holland S, Freitag F, et al. Evidence-based guideline update: pharmacologic treatment for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology. 2012;78:1337-1345. doi: 10.1212/WNL.0b013e3182535d20
28. Dodick DW. CGRP ligand and receptor monoclonal antibodies for migraine prevention: evidence review and clinical implications. Cephalalgia. 2019;39:445-458. doi: 10.1177/ 0333102418821662
29. Pringsheim T, Davenport WJ, Becker WJ. Prophylaxis of migraine headache. CMAJ. 2010;182:E269-276. doi: 10.1503/cmaj.081657
30. Vyepti (eptinezumab-jjmr). Package insert. Lundbeck Pharmaceuticals LLV; 2020. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2020/761119s000lbl.pdf
31. Aimovig (erenumab-aooe). Package insert. Amgen Inc.; 2021. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2021/761077s009lbl.pdf
32. Ajovy (fremanezumab-vfrm). Package insert. Teva Pharmaceuticals USA, Inc.; 2018. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2018/761089s000lbl.pdf
33. Emgality (galcanezumab-gnlm). Package insert. Eli Lilly and Company; 2018. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2018/761063s000lbl.pdf
34. Ashina M, Saper J, Cady R, et al. Eptinezumab in episodic migraine: a randomized, double-blind, placebo-controlled study (PROMISE-1). Cephalalgia. 2020;40:241-254. doi: 10.1177/0333102420905132
35. Lipton RB, Goadsby PJ, Smith J, et al. Efficacy and safety of eptinezumab in patients with chronic migraine: PROMISE-2. Neurology. 2020;94:e1365-e1377. doi: 10.1212/WNL.0000000000009169
36. Dodick DW, Ashina M, Brandes JL, et al. ARISE: a phase 3 randomized trial of erenumab for episodic migraine. Cephalalgia. 2018;38:1026-1037. doi: 10.1177/0333102418759786
37. Goadsby PJ, Reuter U, Hallström Y, et al. A controlled trial of erenumab for episodic migraine. N Engl J Med. 2017;377:2123-2132. doi: 10.1056/NEJMoa1705848
38. Reuter U, Goadsby PJ, Lanteri-Minet M, et al. Efficacy and tolerability of erenumab in patients with episodic migraine in whom two-to-four previous preventive treatments were unsuccessful: a randomised, double-blind, placebo-controlled, phase 3b study. Lancet. 2018;392:2280-2287. doi: 10.1016/S0140-6736(18)32534-0
39. Silberstein SD, Dodick DW, Bigal ME, et al. Fremanezumab for the preventive treatment of chronic migraine. N Engl J Med. 2017; 377:2113-2122. doi: 10.1056/NEJMoa1709038
40. Dodick DW, Silberstein SD, Bigal ME, et al. Effect of fremanezumab compared with placebo for prevention of episodic migraine: a randomized clinical trial. JAMA. 2018;319:1999-2008. doi: 10.1001/jama.2018.4853
41. Stauffer VL, Dodick DW, Zhang Q, et al. Evaluation of galcanezumab for the prevention of episodic migraine: the EVOLVE-1 randomized clinical trial. JAMA Neurol. 2018;75:1080-1088. doi: 10.1001/jamaneurol.2018.1212
42. Skljarevski V, Matharu M, Millen BA, et al. Efficacy and safety of galcanezumab for the prevention of episodic migraine: results of the EVOLVE-2 phase 3 randomized controlled clinical trial. Cephalalgia. 2018;38:1442-1454. doi: 10.1177/0333102418779543
43. Detke HC, Goadsby PJ, Wang S, et al. Galcanezumab in chronic migraine: the randomized, double-blind, placebo-controlled REGAIN study. Neurology. 2018;91:e2211-e2221. doi: 10.1212/WNL.0000000000006640
44. Goadsby PJ, Dodick DW, Leone M, at al. Trial of galcanezumab in prevention of episodic cluster headache. N Engl J Med. 2019; 381:132-141. doi: 10.1056/NEJMoa1813440
45. Croop R, Lipton RB, Kudrow D, et al. Oral rimegepant for preventive treatment of migraine: a phase 2/3, randomised, double-blind, placebo-controlled trial. Lancet. 2021;397:51-60. doi: 10.1016/S0140-6736(20)32544-7
46. Ailani J, Lipton RB, Goadsby PJ, et al. Atogepant for the preventive treatment of migraine. N Engl J Med. 2021;385:695-706. doi: 10.1056/NEJMoa2035908
47. Qulipta (atogepant). Package insert. AbbVie; 2021. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2021/215206Orig1s000lbl.pdf
48. Han L, Liu Y, Xiong H, et al. CGRP monoclonal antibody for preventive treatment of chronic migraine: an update of meta-analysis. Brain Behav. 2019;9:e01215. doi: 10.1002/brb3.1215
49. Zhu Y, Liu Y, Zhao J, et al. The efficacy and safety of calcitonin gene-related peptide monoclonal antibody for episodic migraine: a meta-analysis. Neurol Sci. 2018;39:2097-2106. doi: 10.1007/s10072-018-3547-3
50. Szperka CL, VanderPluym J, Orr SL, et al. Recommendations on the use of anti-CGRP monoclonal antibodies in children and adolescents. Headache. 2018;58:1658-1669. doi: 10.1111/head.13414
51. Pellesi L, Do TP, Ashina H, et al. Dual therapy with anti-CGRP monoclonal antibodies and botulinum toxin for migraine prevention: is there a rationale? Headache. 2020;60:1056-1065. doi: 10.1111/head.13843
52. D’Antona L, Matharu M. Identifying and managing refractory migraine: barriers and opportunities? J Headache Pain. 2019;20:89. doi: 10.1186/s10194-019-1040-x
53. Cohen F, Armand C, Lipton RB, et al. Efficacy and tolerability of calcitonin gene-related peptide targeted monoclonal antibody medications as add-on therapy to onabotulinumtoxinA in patients with chronic migraine. Pain Med. 2021;1857-1863. doi: 10.1093/pm/pnab093
54. Berman G, Croop R, Kudrow D, et al. Safety of rimegepant, an oral CGRP receptor antagonist, plus CGRP monoclonal antibodies for migraine. Headache. 2020;60:1734-1742. doi: 10.1111/head.13930
Migraine is a headache disorder that often causes unilateral pain, photophobia, phonophobia, nausea, and vomiting. More than 70% of office visits for migraine are made to primary care physicians.1 Recent data suggest migraine may be caused primarily by neuronal dysfunction and only secondarily by vasodilation.2 Although there are numerous classes of drugs used for migraine prevention and treatment, their success has been limited by inadequate efficacy, tolerability, and patient adherence.3 The discovery of pro-inflammatory markers such as calcitonin gene-related peptide (CGRP) has led to the development of new medications to prevent and treat migraine.4

Pathophysiology, Dx and triggers, indications for pharmacotherapy
Pathophysiology. A migraine is thought to be caused by cortical spreading depression (CSD), a depolarization of glial and neuronal cell membranes.5
Dx and triggers. In 2018, the International Headache Society revised its guidelines for the diagnosis of migraine.7 According to the 3rd edition of The International Classification of Headache Disorders (ICHD-3), the diagnosis of migraine is made when a patient has at least 5 headache attacks that last 4 to 72 hours and have at least 2 of the following characteristics: (1) unilateral location, (2) pulsating quality, (3) moderate-to-severe pain intensity, and (4) aggravated by or causing avoidance of routine physical activity.7 The headache attacks also should have (1) associated nausea or vomiting or (2) photophobia and phonophobia.7 The presence of atypical signs or symptoms as indicated by the SNNOOP10 mnemonic raises concerns for secondary headaches and the need for further investigation into the cause of the headache (TABLE 1).8 It is not possible to detect every secondary headache with standard neuroimaging, but the SNNOOP10 red flags can help determine when imaging may be indicated.8 Potential triggers for migraine can be found in TABLE 2.9

Indications for pharmacotherapy. All patients receiving a diagnosis of migraine should be offered acute pharmacologic treatment. Consider preventive therapy anytime there are ≥ 4 headache days per month, debilitating attacks despite acute therapy, overuse of acute medication (> 2 d/wk), difficulty tolerating acute medication, patient preference, or presence of certain migraine subtypes.7,10

Acute treatments
Abortive therapies for migraine include analgesics such as nonsteroidal anti-inflammatory drugs (NSAIDs) and acetaminophen, and ergot alkaloids, triptans, or small-molecule CGRP receptor antagonists (gepants). Prompt administration increases the chance of success with acute therapy. Medications with the highest levels of efficacy based on the 2015 guidelines from the American Headache Society (AHS) are given in TABLE 3.11 Lasmiditan (Reyvow) is not included in the 2015 guidelines, as it was approved after publication of the guidelines.


Non-CGRP first-line therapies
NSAIDs and acetaminophen. NSAIDs such as aspirin, diclofenac, ibuprofen, and naproxen have a high level of evidence to support their use as first-line treatments for mild-to-moderate migraine attacks. Trials consistently demonstrate their superiority to placebo in headache relief and complete pain relief at 2 hours. There is no recommendation for selecting one NSAID over another; however, consider their frequency of dosing and adverse effect profiles. The number needed to treat for complete pain relief at 2 hours ranges from 7 to 10 for most NSAIDs.11,12 In some placebo-controlled studies, acetaminophen was less effective than NSAIDs, but was safer because it did not cause gastric irritation or antiplatelet effects.12
Triptans inhibit 5-HT1B/1D receptors. Consider formulation, route of administration, cost, and pharmacokinetics when selecting a triptan. Patients who do not respond well to one triptan may respond favorably to another. A meta-analysis of the effectiveness of the 7 available agents found that triptans at standard doses provided pain relief within 2 hours in 42% to 76% of patients, and sustained freedom from pain for 2 hours in 18% to 50% of patients.13 Lasmiditan is a selective serotonin receptor (5-HT1F) agonist that lacks vasoconstrictor activity. This is an option for patients with relative contraindications to triptans due to cardiovascular risk factors.10
Continue to: Second-line therapies
Second-line therapies
Intranasal dihydroergotamine has a favorable adverse event profile and greater evidence for efficacy compared with ergotamine. Compared with triptans, intranasal dihydroergotamine has a high level of efficacy but causes more adverse effects.14 Severe nausea is common, and dihydroergotamine often is used in combination with an antiemetic drug. Dihydroergotamine should not be used within 24 hours of taking a triptan, and it is contraindicated for patients who have hypertension or ischemic heart disease or who are pregnant or breastfeeding. There is also the potential for adverse drug interactions.15
Antiemetics may be helpful for migraine associated with severe nausea or vomiting. The dopamine antagonists metoclopramide, prochlorperazine, and chlorpromazine have demonstrated benefit in randomized placebo-controlled trials.11 Ondansetron has not been studied extensively, but sometimes is used in clinical practice. Nonoral routes of administration may be useful in patients having trouble swallowing medications or in those experiencing significant nausea or vomiting early during migraine attacks.
Due to the high potential for abuse, opioids should not be used routinely for the treatment of migraine.12 There is no high-quality evidence supporting the efficacy of barbiturates (ie, butalbital-containing compounds) for acute migraine treatment.11 Moreover, use of these agents may increase the likelihood of progression from episodic to chronic migraine.16
Gepants for acute migraine treatment
Neuropeptide CGRP is released from trigeminal nerves and is a potent dilator of cerebral and dural vessels, playing a key role in regulating blood flow to the brain. Other roles of CGRP include the release of inflammatory agents from mast cells and the transmission of painful stimuli from intracranial vessels.17 The CGRP receptor or ligand can be targeted by small-molecule receptor antagonists for acute and preventive migraine treatment (and by monoclonal antibodies solely for prevention, discussed later). It has been theorized that gepants bind to CGRP receptors, resulting in decreased blood flow to the brain, inhibition of neurogenic inflammation, and reduced pain signaling.17 Unlike triptans and ergotamine derivatives, these novel treatments do not constrict blood vessels and may have a unique role in patients with contraindications to triptans.
The 3 gepants approved for acute treatment—ubrogepant (Ubrelvy),18 rimegepant (Nurtec),19 and zavegepant (Zavzpret)20—were compared with placebo in clinical trials and were shown to increase the number of patients who were completely pain free at 2 hours, were free of the most bothersome associated symptom (photophobia, phonophobia, or nausea) at 2 hours, and remained pain free at 24 hours (TABLE 418-24).

Continue to: Ubrogrepant
Ubrogepant, in 2 Phase 3 trials (ACHIEVE I and ACHIEVE II) demonstrated effectiveness compared with placebo.21,22 The most common adverse effects reported were nausea and somnolence at very low rates. Pain-relief rates at 2 hours post dose (> 60% of participants) were higher than pain-free rates, and a significantly higher percentage (> 40%) of ubrogepant-treated participants reported ability to function normally on the Functional Disability Scale.25
Rimegepant was also superior to placebo (59% vs 43%) in pain relief at 2 hours post dose and other secondary endpoints.23 Rimegepant also has potential drug interactions
Zavegepant, approved in March 2023, is administered once daily as a 10-mg nasal spray. In its Phase 3 trial, zavegepant was significantly superior to placebo at 2 hours post dose in freedom from pain (24% v 15%), and in freedom from the most bothersome symptom (40% v 31%).24 Dosage modifications are not needed with mild-to-moderate renal or hepatic disease.20
Worth noting. The safety of using ubrogepant to treat more than 8 migraine episodes in a 30-day period has not been established. The safety of using more than 18 doses of zavegepant in a 30-day period also has not been established. With ubrogepant and rimegepant, there are dosing modifications for concomitant use with specific drugs (CYP3A4 inhibitors and inducers) due to potential interactions and in patients with hepatic or renal impairment.18,19
There are no trials comparing efficacy of CGRP antagonists to triptans. Recognizing that these newer medications would be costly, the AHS position statement released in 2019 recommends that gepants be considered for those with contraindications to triptans or for whom at least 2 oral triptans have failed (as determined by a validated patient outcome questionnaire).10 Step therapy with documentation of previous trials and therapy failures is often required by insurance companies prior to gepant coverage.
Continue to: Preventive therapies
Preventive therapies
Preventive migraine therapies are used to reduce duration, frequency, and severity of attacks, the need for acute treatment, and overall headache disability.26 Medications typically are chosen based on efficacy, adverse effect profile, and patient comorbidities. Barriers to successful use include poor patient adherence and tolerability, the need for slow dose titration, and long-term use (minimum of 2 months) at maximum tolerated or minimum effective doses. Medications with established efficacy (Level Aa) based on the 2012 guidelines from the American Academy of Neurology (AAN) and the AHS are given in TABLE 5.27-29

Drugs having received the strongest level of evidence for migraine prevention are metoprolol, propranolol, timolol, topiramate, valproate sodium, divalproex sodium, and onabotulinumtoxinA (Botox), and frovatriptan for menstrual migraine prevention. Because these guidelines were last updated in 2012, they did not cover gepants (which will be discussed shortly). The AHS released a position statement in 2019 supporting the use of
CGRP-targeted prevention
Four anti-CGRP mAbs and 2 gepants have been approved for migraine prevention in the United States. Differences between products include targets (ligand vs receptor), antibody IgG subtype, bioavailability, route of administration, and frequency of administration.28 As noted in the Phase 3 studies (TABLE 619,30-47), these therapies are highly efficacious, safe, and tolerable.
Gepants. Rimegepant, discussed earlier for migraine treatment, is one of the CGRP receptor antagonists approved for prevention. The other is atogepant (Qulipta), approved only for prevention. Ubrogepant is not approved for prevention.
Anti-CGRP mAb is the only medication class specifically created for migraine prevention.10,26 As already noted, several efficacious non-CGRP treatment options are available for migraine prevention. However, higher doses of those agents, if needed,
Continue to: The targeted anti-CGRP approach...
The targeted anti-CGRP approach, which can be used by patients with liver or kidney disease, results in decreased toxicity and minimal drug interactions. Long half-lives allow for monthly or quarterly injections, possibly resulting in increased compliance.28 Dose titration is not needed, allowing for more rapid symptom management. The large molecular size of a mAb limits its transfer across the blood-brain barrier, making central nervous system adverse effects unlikely.28 Despite the compelling mAb pharmacologic properties, their use may be limited by a lack of long-term safety data and the need for parenteral administration. Although immunogenicity—the development of neutralizing antibodies—can limit long-term tolerability or efficacy of mAbs generally,26,28 anti-CGRP mAbs were engineered to minimally activate the immune system and have not been associated with immune suppression, opportunistic infections, malignancies, or decreased efficacy.28
A pooled meta-analysis including 4 trials (3166 patients) found that CGRP mAbs compared with placebo significantly improved patient response rates, defined as at least a 50% and 75% reduction in monthly headache/migraine days from baseline to Weeks 9 to 12.48 Another meta-analysis including 8 trials (2292 patients) found a significant reduction from baseline in monthly migraine days and monthly acute migraine medication consumption among patients taking CGRP mAbs compared with those taking placebo.49 Open-label extension studies have shown progressive and cumulative benefits in individuals who respond to anti-CGRP mAbs. Therefore, several treatment cycles may be necessary to determine overall efficacy of therapy.10,28
Cost initially can be a barrier. Insurance companies often require step therapy before agreeing to cover mAb therapy, which aligns with the 2019 AHS position statement.10
When combination treatment may be appropriate
Monotherapy is the usual approach to preventing migraine due to advantages of efficacy, simplified regimens, lower cost, and reduced adverse effects.51 However, if a patient does not benefit from monotherapy even after trying dose titrations as tolerated or switching therapies, trying complementary combination therapy is appropriate. Despite a shortage of clinical trials supporting the use of 2 or more preventive medications with different mechanisms of action, this strategy is used clinically.10 Consider combination therapy in those with refractory disease, partial responses, or intolerance to recommended doses.52 Articles reporting on case study reviews have rationalized the combined use of onabotulinumtoxinA and anti-CGRP mAbs, noting better migraine control.51,53 The 2019 AHS position statement recommends adding a mAb to an existing preventive treatment regimen with no other changes until mAb effectiveness is determined, as the risk for drug interactions on dual therapy is low.10 Safety and efficacy also have been demonstrated with the combination of preventive anti-CGRP mAbs and acute treatment with gepants as needed.54
CORRESPONDENCE
Emily Peterson, PharmD, BCACP, 3640 Middlebury Road, Iowa City, IA 52242; Emily-a-peterson@uiowa.edu
Migraine is a headache disorder that often causes unilateral pain, photophobia, phonophobia, nausea, and vomiting. More than 70% of office visits for migraine are made to primary care physicians.1 Recent data suggest migraine may be caused primarily by neuronal dysfunction and only secondarily by vasodilation.2 Although there are numerous classes of drugs used for migraine prevention and treatment, their success has been limited by inadequate efficacy, tolerability, and patient adherence.3 The discovery of pro-inflammatory markers such as calcitonin gene-related peptide (CGRP) has led to the development of new medications to prevent and treat migraine.4

Pathophysiology, Dx and triggers, indications for pharmacotherapy
Pathophysiology. A migraine is thought to be caused by cortical spreading depression (CSD), a depolarization of glial and neuronal cell membranes.5
Dx and triggers. In 2018, the International Headache Society revised its guidelines for the diagnosis of migraine.7 According to the 3rd edition of The International Classification of Headache Disorders (ICHD-3), the diagnosis of migraine is made when a patient has at least 5 headache attacks that last 4 to 72 hours and have at least 2 of the following characteristics: (1) unilateral location, (2) pulsating quality, (3) moderate-to-severe pain intensity, and (4) aggravated by or causing avoidance of routine physical activity.7 The headache attacks also should have (1) associated nausea or vomiting or (2) photophobia and phonophobia.7 The presence of atypical signs or symptoms as indicated by the SNNOOP10 mnemonic raises concerns for secondary headaches and the need for further investigation into the cause of the headache (TABLE 1).8 It is not possible to detect every secondary headache with standard neuroimaging, but the SNNOOP10 red flags can help determine when imaging may be indicated.8 Potential triggers for migraine can be found in TABLE 2.9

Indications for pharmacotherapy. All patients receiving a diagnosis of migraine should be offered acute pharmacologic treatment. Consider preventive therapy anytime there are ≥ 4 headache days per month, debilitating attacks despite acute therapy, overuse of acute medication (> 2 d/wk), difficulty tolerating acute medication, patient preference, or presence of certain migraine subtypes.7,10

Acute treatments
Abortive therapies for migraine include analgesics such as nonsteroidal anti-inflammatory drugs (NSAIDs) and acetaminophen, and ergot alkaloids, triptans, or small-molecule CGRP receptor antagonists (gepants). Prompt administration increases the chance of success with acute therapy. Medications with the highest levels of efficacy based on the 2015 guidelines from the American Headache Society (AHS) are given in TABLE 3.11 Lasmiditan (Reyvow) is not included in the 2015 guidelines, as it was approved after publication of the guidelines.


Non-CGRP first-line therapies
NSAIDs and acetaminophen. NSAIDs such as aspirin, diclofenac, ibuprofen, and naproxen have a high level of evidence to support their use as first-line treatments for mild-to-moderate migraine attacks. Trials consistently demonstrate their superiority to placebo in headache relief and complete pain relief at 2 hours. There is no recommendation for selecting one NSAID over another; however, consider their frequency of dosing and adverse effect profiles. The number needed to treat for complete pain relief at 2 hours ranges from 7 to 10 for most NSAIDs.11,12 In some placebo-controlled studies, acetaminophen was less effective than NSAIDs, but was safer because it did not cause gastric irritation or antiplatelet effects.12
Triptans inhibit 5-HT1B/1D receptors. Consider formulation, route of administration, cost, and pharmacokinetics when selecting a triptan. Patients who do not respond well to one triptan may respond favorably to another. A meta-analysis of the effectiveness of the 7 available agents found that triptans at standard doses provided pain relief within 2 hours in 42% to 76% of patients, and sustained freedom from pain for 2 hours in 18% to 50% of patients.13 Lasmiditan is a selective serotonin receptor (5-HT1F) agonist that lacks vasoconstrictor activity. This is an option for patients with relative contraindications to triptans due to cardiovascular risk factors.10
Continue to: Second-line therapies
Second-line therapies
Intranasal dihydroergotamine has a favorable adverse event profile and greater evidence for efficacy compared with ergotamine. Compared with triptans, intranasal dihydroergotamine has a high level of efficacy but causes more adverse effects.14 Severe nausea is common, and dihydroergotamine often is used in combination with an antiemetic drug. Dihydroergotamine should not be used within 24 hours of taking a triptan, and it is contraindicated for patients who have hypertension or ischemic heart disease or who are pregnant or breastfeeding. There is also the potential for adverse drug interactions.15
Antiemetics may be helpful for migraine associated with severe nausea or vomiting. The dopamine antagonists metoclopramide, prochlorperazine, and chlorpromazine have demonstrated benefit in randomized placebo-controlled trials.11 Ondansetron has not been studied extensively, but sometimes is used in clinical practice. Nonoral routes of administration may be useful in patients having trouble swallowing medications or in those experiencing significant nausea or vomiting early during migraine attacks.
Due to the high potential for abuse, opioids should not be used routinely for the treatment of migraine.12 There is no high-quality evidence supporting the efficacy of barbiturates (ie, butalbital-containing compounds) for acute migraine treatment.11 Moreover, use of these agents may increase the likelihood of progression from episodic to chronic migraine.16
Gepants for acute migraine treatment
Neuropeptide CGRP is released from trigeminal nerves and is a potent dilator of cerebral and dural vessels, playing a key role in regulating blood flow to the brain. Other roles of CGRP include the release of inflammatory agents from mast cells and the transmission of painful stimuli from intracranial vessels.17 The CGRP receptor or ligand can be targeted by small-molecule receptor antagonists for acute and preventive migraine treatment (and by monoclonal antibodies solely for prevention, discussed later). It has been theorized that gepants bind to CGRP receptors, resulting in decreased blood flow to the brain, inhibition of neurogenic inflammation, and reduced pain signaling.17 Unlike triptans and ergotamine derivatives, these novel treatments do not constrict blood vessels and may have a unique role in patients with contraindications to triptans.
The 3 gepants approved for acute treatment—ubrogepant (Ubrelvy),18 rimegepant (Nurtec),19 and zavegepant (Zavzpret)20—were compared with placebo in clinical trials and were shown to increase the number of patients who were completely pain free at 2 hours, were free of the most bothersome associated symptom (photophobia, phonophobia, or nausea) at 2 hours, and remained pain free at 24 hours (TABLE 418-24).

Continue to: Ubrogrepant
Ubrogepant, in 2 Phase 3 trials (ACHIEVE I and ACHIEVE II) demonstrated effectiveness compared with placebo.21,22 The most common adverse effects reported were nausea and somnolence at very low rates. Pain-relief rates at 2 hours post dose (> 60% of participants) were higher than pain-free rates, and a significantly higher percentage (> 40%) of ubrogepant-treated participants reported ability to function normally on the Functional Disability Scale.25
Rimegepant was also superior to placebo (59% vs 43%) in pain relief at 2 hours post dose and other secondary endpoints.23 Rimegepant also has potential drug interactions
Zavegepant, approved in March 2023, is administered once daily as a 10-mg nasal spray. In its Phase 3 trial, zavegepant was significantly superior to placebo at 2 hours post dose in freedom from pain (24% v 15%), and in freedom from the most bothersome symptom (40% v 31%).24 Dosage modifications are not needed with mild-to-moderate renal or hepatic disease.20
Worth noting. The safety of using ubrogepant to treat more than 8 migraine episodes in a 30-day period has not been established. The safety of using more than 18 doses of zavegepant in a 30-day period also has not been established. With ubrogepant and rimegepant, there are dosing modifications for concomitant use with specific drugs (CYP3A4 inhibitors and inducers) due to potential interactions and in patients with hepatic or renal impairment.18,19
There are no trials comparing efficacy of CGRP antagonists to triptans. Recognizing that these newer medications would be costly, the AHS position statement released in 2019 recommends that gepants be considered for those with contraindications to triptans or for whom at least 2 oral triptans have failed (as determined by a validated patient outcome questionnaire).10 Step therapy with documentation of previous trials and therapy failures is often required by insurance companies prior to gepant coverage.
Continue to: Preventive therapies
Preventive therapies
Preventive migraine therapies are used to reduce duration, frequency, and severity of attacks, the need for acute treatment, and overall headache disability.26 Medications typically are chosen based on efficacy, adverse effect profile, and patient comorbidities. Barriers to successful use include poor patient adherence and tolerability, the need for slow dose titration, and long-term use (minimum of 2 months) at maximum tolerated or minimum effective doses. Medications with established efficacy (Level Aa) based on the 2012 guidelines from the American Academy of Neurology (AAN) and the AHS are given in TABLE 5.27-29

Drugs having received the strongest level of evidence for migraine prevention are metoprolol, propranolol, timolol, topiramate, valproate sodium, divalproex sodium, and onabotulinumtoxinA (Botox), and frovatriptan for menstrual migraine prevention. Because these guidelines were last updated in 2012, they did not cover gepants (which will be discussed shortly). The AHS released a position statement in 2019 supporting the use of
CGRP-targeted prevention
Four anti-CGRP mAbs and 2 gepants have been approved for migraine prevention in the United States. Differences between products include targets (ligand vs receptor), antibody IgG subtype, bioavailability, route of administration, and frequency of administration.28 As noted in the Phase 3 studies (TABLE 619,30-47), these therapies are highly efficacious, safe, and tolerable.
Gepants. Rimegepant, discussed earlier for migraine treatment, is one of the CGRP receptor antagonists approved for prevention. The other is atogepant (Qulipta), approved only for prevention. Ubrogepant is not approved for prevention.
Anti-CGRP mAb is the only medication class specifically created for migraine prevention.10,26 As already noted, several efficacious non-CGRP treatment options are available for migraine prevention. However, higher doses of those agents, if needed,
Continue to: The targeted anti-CGRP approach...
The targeted anti-CGRP approach, which can be used by patients with liver or kidney disease, results in decreased toxicity and minimal drug interactions. Long half-lives allow for monthly or quarterly injections, possibly resulting in increased compliance.28 Dose titration is not needed, allowing for more rapid symptom management. The large molecular size of a mAb limits its transfer across the blood-brain barrier, making central nervous system adverse effects unlikely.28 Despite the compelling mAb pharmacologic properties, their use may be limited by a lack of long-term safety data and the need for parenteral administration. Although immunogenicity—the development of neutralizing antibodies—can limit long-term tolerability or efficacy of mAbs generally,26,28 anti-CGRP mAbs were engineered to minimally activate the immune system and have not been associated with immune suppression, opportunistic infections, malignancies, or decreased efficacy.28
A pooled meta-analysis including 4 trials (3166 patients) found that CGRP mAbs compared with placebo significantly improved patient response rates, defined as at least a 50% and 75% reduction in monthly headache/migraine days from baseline to Weeks 9 to 12.48 Another meta-analysis including 8 trials (2292 patients) found a significant reduction from baseline in monthly migraine days and monthly acute migraine medication consumption among patients taking CGRP mAbs compared with those taking placebo.49 Open-label extension studies have shown progressive and cumulative benefits in individuals who respond to anti-CGRP mAbs. Therefore, several treatment cycles may be necessary to determine overall efficacy of therapy.10,28
Cost initially can be a barrier. Insurance companies often require step therapy before agreeing to cover mAb therapy, which aligns with the 2019 AHS position statement.10
When combination treatment may be appropriate
Monotherapy is the usual approach to preventing migraine due to advantages of efficacy, simplified regimens, lower cost, and reduced adverse effects.51 However, if a patient does not benefit from monotherapy even after trying dose titrations as tolerated or switching therapies, trying complementary combination therapy is appropriate. Despite a shortage of clinical trials supporting the use of 2 or more preventive medications with different mechanisms of action, this strategy is used clinically.10 Consider combination therapy in those with refractory disease, partial responses, or intolerance to recommended doses.52 Articles reporting on case study reviews have rationalized the combined use of onabotulinumtoxinA and anti-CGRP mAbs, noting better migraine control.51,53 The 2019 AHS position statement recommends adding a mAb to an existing preventive treatment regimen with no other changes until mAb effectiveness is determined, as the risk for drug interactions on dual therapy is low.10 Safety and efficacy also have been demonstrated with the combination of preventive anti-CGRP mAbs and acute treatment with gepants as needed.54
CORRESPONDENCE
Emily Peterson, PharmD, BCACP, 3640 Middlebury Road, Iowa City, IA 52242; Emily-a-peterson@uiowa.edu
1. Lipton RB, Nicholson RA, Reed ML, et al. Diagnosis, consultation, treatment, and impact of migraine in the US: results of the OVERCOME (US) study. Headache. 2022;62:122-140. doi: 10.1111/head.14259
2. Burstein R, Noseda R, Borsook D. Migraine: multiple processes; complext pathophysiology. J Neurosci. 2015;35:6619-6629. doi: 10.1523/JNEUROSCI.0373-15.2015
3. Edvinsson L, Haanes KA, Warfvinge K, et al. CGRP as the target of new migraine therapies - successful translation from bench to clinic. Nat Rev Neurol. 2018;14:338-350. doi: 10.1038/s41582-018-0003-1
4. McGrath K, Rague A, Thesing C, et al. Migraine: expanding our Tx arsenal. J Fam Pract. 2019;68:10-14;16-24.
5. Dodick DW. Migraine. Lancet. 2018;391:1315-1330. doi: 10.1016/S0140-6736(18)30478-1
6. Agostoni EC, Barbanti P, Calabresi P, et al. Current and emerging evidence-based treatment options in chronic migraine: a narrative review. J Headache Pain. 2019;20:92. doi: 10.1186/s10194-019-1038-4
7. IHS. Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211. doi: 10.1177/0333102417738202
8. Do TP, Remmers A, Schytz HW, et al. Red and orange flags for secondary headaches in clinical practice: SNNOOP10 list. Neurology. 2019;92:134-144. doi: 10.1212/WNL.0000000000006697
9. NIH. Migraine. Accessed July 30, 2023.
10. AHS. The American Headache Society position statement on integrating new migraine treatments into clinical practice. Headache. 2019;59:1-18. doi: 10.1111/head.13456
11. Marmura MJ, Silberstein SD, Schwedt TJ. The acute treatment of migraine in adults: the American Headache Society evidence assessment of migraine pharmacotherapies. Headache. 2015;55:3-20. doi: 10.1111/head.12499
12. Mayans L, Walling A. Acute migraine headache: treatment strategies. Am Fam Physician. 2018;97:243-251.
13. Cameron C, Kelly S, Hsieh SC, et al. Triptans in the acute treatment of migraine: a systematic review and network meta-analysis. Headache. 2015;55(suppl 4):221-235. doi: 10.1111/head.12601
14. Becker WJ. Acute migraine treatment. Continuum (Minneap Minn). 2015;21:953-972. doi: 10.1212/CON.0000000000000192
15. Migranal (dihydroergotamine mesylate) Package insert. Valeant Pharmaceuticals North America; 2019. Accessed June 17, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2019/020148Orig1s025lbl.pdf
16. Minen MT, Tanev K, Friedman BW. Evaluation and treatment of migraine in the emergency department: a review. Headache. 2014;54:1131-45. doi: 10.1111/head.12399
17. Durham PL. CGRP-receptor antagonists--a fresh approach to migraine therapy? N Engl J Med. 2004;350:1073-1075. doi: 10.1056/NEJMp048016
18. Ubrelvy (ubrogepant). Package insert. Allergan, Inc.; 2019. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2019/211765s000lbl.pdf
19. Nurtec ODT (rimegepant sulfate). Package insert. Biohaven Pharmaceuticals, Inc.; 2021. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2021/212728s006lbl.pdf
20. Zavzpret (zavegepant). Package insert. Pfizer Labs.; 2023. Accessed July 15, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2023/216386s000lbl.pdf
21. Dodick DW, Lipton RB, Ailani J, et al. Ubrogepant for the treatment of migraine. N Engl J Med. 2019;381:2230-2241. doi: 10.1056/NEJMoa1813049
22. Lipton RB, Dodick DW, Ailani J, et al. Effect of ubrogepant vs placebo on pain and the most bothersome associated symptom in the acute treatment of migraine: the ACHIEVE II randomized clinical trial. JAMA. 2019;322:1887-1898. doi: 10.1001/jama.2019.16711
23. Croop R, Goadsby PJ, Stock DA, et al. Efficacy, safety, and tolerability of rimegepant orally disintegrating tablet for the acute treatment of migraine: a randomised, phase 3, double-blind, placebo-controlled trial. Lancet. 2019;394:737-745. doi: 10.1016/S0140-6736(19)31606-X
24. Lipton RB, Croop R, Stock DA, et al. Safety, tolerability, and efficacy of zavegepant 10 mg nasal spray for the acute treatment of migraine in the USA: a phase 3, double-blind, randomised, placebo-controlled multicentre trial. Lancet Neurol. 2023;22:209-217. doi: 10.1016/S1474-4422(22)00517-8
25. Dodick DW, Lipton RB, Ailani J, et al. Ubrogepant, an acute treatment for migraine, improved patient-reported functional disability and satisfaction in 2 single-attack phase 3 randomized trials, ACHIEVE I and II. Headache. 2020;60:686-700. doi: 10.1111/head.13766
26. Burch R. Migraine and tension-type headache: diagnosis and treatment. Med Clin North Am. 2019;103:215-233. doi:10.1016/j.mcna.2018.10.003
27. Silberstein SD, Holland S, Freitag F, et al. Evidence-based guideline update: pharmacologic treatment for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology. 2012;78:1337-1345. doi: 10.1212/WNL.0b013e3182535d20
28. Dodick DW. CGRP ligand and receptor monoclonal antibodies for migraine prevention: evidence review and clinical implications. Cephalalgia. 2019;39:445-458. doi: 10.1177/ 0333102418821662
29. Pringsheim T, Davenport WJ, Becker WJ. Prophylaxis of migraine headache. CMAJ. 2010;182:E269-276. doi: 10.1503/cmaj.081657
30. Vyepti (eptinezumab-jjmr). Package insert. Lundbeck Pharmaceuticals LLV; 2020. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2020/761119s000lbl.pdf
31. Aimovig (erenumab-aooe). Package insert. Amgen Inc.; 2021. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2021/761077s009lbl.pdf
32. Ajovy (fremanezumab-vfrm). Package insert. Teva Pharmaceuticals USA, Inc.; 2018. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2018/761089s000lbl.pdf
33. Emgality (galcanezumab-gnlm). Package insert. Eli Lilly and Company; 2018. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2018/761063s000lbl.pdf
34. Ashina M, Saper J, Cady R, et al. Eptinezumab in episodic migraine: a randomized, double-blind, placebo-controlled study (PROMISE-1). Cephalalgia. 2020;40:241-254. doi: 10.1177/0333102420905132
35. Lipton RB, Goadsby PJ, Smith J, et al. Efficacy and safety of eptinezumab in patients with chronic migraine: PROMISE-2. Neurology. 2020;94:e1365-e1377. doi: 10.1212/WNL.0000000000009169
36. Dodick DW, Ashina M, Brandes JL, et al. ARISE: a phase 3 randomized trial of erenumab for episodic migraine. Cephalalgia. 2018;38:1026-1037. doi: 10.1177/0333102418759786
37. Goadsby PJ, Reuter U, Hallström Y, et al. A controlled trial of erenumab for episodic migraine. N Engl J Med. 2017;377:2123-2132. doi: 10.1056/NEJMoa1705848
38. Reuter U, Goadsby PJ, Lanteri-Minet M, et al. Efficacy and tolerability of erenumab in patients with episodic migraine in whom two-to-four previous preventive treatments were unsuccessful: a randomised, double-blind, placebo-controlled, phase 3b study. Lancet. 2018;392:2280-2287. doi: 10.1016/S0140-6736(18)32534-0
39. Silberstein SD, Dodick DW, Bigal ME, et al. Fremanezumab for the preventive treatment of chronic migraine. N Engl J Med. 2017; 377:2113-2122. doi: 10.1056/NEJMoa1709038
40. Dodick DW, Silberstein SD, Bigal ME, et al. Effect of fremanezumab compared with placebo for prevention of episodic migraine: a randomized clinical trial. JAMA. 2018;319:1999-2008. doi: 10.1001/jama.2018.4853
41. Stauffer VL, Dodick DW, Zhang Q, et al. Evaluation of galcanezumab for the prevention of episodic migraine: the EVOLVE-1 randomized clinical trial. JAMA Neurol. 2018;75:1080-1088. doi: 10.1001/jamaneurol.2018.1212
42. Skljarevski V, Matharu M, Millen BA, et al. Efficacy and safety of galcanezumab for the prevention of episodic migraine: results of the EVOLVE-2 phase 3 randomized controlled clinical trial. Cephalalgia. 2018;38:1442-1454. doi: 10.1177/0333102418779543
43. Detke HC, Goadsby PJ, Wang S, et al. Galcanezumab in chronic migraine: the randomized, double-blind, placebo-controlled REGAIN study. Neurology. 2018;91:e2211-e2221. doi: 10.1212/WNL.0000000000006640
44. Goadsby PJ, Dodick DW, Leone M, at al. Trial of galcanezumab in prevention of episodic cluster headache. N Engl J Med. 2019; 381:132-141. doi: 10.1056/NEJMoa1813440
45. Croop R, Lipton RB, Kudrow D, et al. Oral rimegepant for preventive treatment of migraine: a phase 2/3, randomised, double-blind, placebo-controlled trial. Lancet. 2021;397:51-60. doi: 10.1016/S0140-6736(20)32544-7
46. Ailani J, Lipton RB, Goadsby PJ, et al. Atogepant for the preventive treatment of migraine. N Engl J Med. 2021;385:695-706. doi: 10.1056/NEJMoa2035908
47. Qulipta (atogepant). Package insert. AbbVie; 2021. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2021/215206Orig1s000lbl.pdf
48. Han L, Liu Y, Xiong H, et al. CGRP monoclonal antibody for preventive treatment of chronic migraine: an update of meta-analysis. Brain Behav. 2019;9:e01215. doi: 10.1002/brb3.1215
49. Zhu Y, Liu Y, Zhao J, et al. The efficacy and safety of calcitonin gene-related peptide monoclonal antibody for episodic migraine: a meta-analysis. Neurol Sci. 2018;39:2097-2106. doi: 10.1007/s10072-018-3547-3
50. Szperka CL, VanderPluym J, Orr SL, et al. Recommendations on the use of anti-CGRP monoclonal antibodies in children and adolescents. Headache. 2018;58:1658-1669. doi: 10.1111/head.13414
51. Pellesi L, Do TP, Ashina H, et al. Dual therapy with anti-CGRP monoclonal antibodies and botulinum toxin for migraine prevention: is there a rationale? Headache. 2020;60:1056-1065. doi: 10.1111/head.13843
52. D’Antona L, Matharu M. Identifying and managing refractory migraine: barriers and opportunities? J Headache Pain. 2019;20:89. doi: 10.1186/s10194-019-1040-x
53. Cohen F, Armand C, Lipton RB, et al. Efficacy and tolerability of calcitonin gene-related peptide targeted monoclonal antibody medications as add-on therapy to onabotulinumtoxinA in patients with chronic migraine. Pain Med. 2021;1857-1863. doi: 10.1093/pm/pnab093
54. Berman G, Croop R, Kudrow D, et al. Safety of rimegepant, an oral CGRP receptor antagonist, plus CGRP monoclonal antibodies for migraine. Headache. 2020;60:1734-1742. doi: 10.1111/head.13930
1. Lipton RB, Nicholson RA, Reed ML, et al. Diagnosis, consultation, treatment, and impact of migraine in the US: results of the OVERCOME (US) study. Headache. 2022;62:122-140. doi: 10.1111/head.14259
2. Burstein R, Noseda R, Borsook D. Migraine: multiple processes; complext pathophysiology. J Neurosci. 2015;35:6619-6629. doi: 10.1523/JNEUROSCI.0373-15.2015
3. Edvinsson L, Haanes KA, Warfvinge K, et al. CGRP as the target of new migraine therapies - successful translation from bench to clinic. Nat Rev Neurol. 2018;14:338-350. doi: 10.1038/s41582-018-0003-1
4. McGrath K, Rague A, Thesing C, et al. Migraine: expanding our Tx arsenal. J Fam Pract. 2019;68:10-14;16-24.
5. Dodick DW. Migraine. Lancet. 2018;391:1315-1330. doi: 10.1016/S0140-6736(18)30478-1
6. Agostoni EC, Barbanti P, Calabresi P, et al. Current and emerging evidence-based treatment options in chronic migraine: a narrative review. J Headache Pain. 2019;20:92. doi: 10.1186/s10194-019-1038-4
7. IHS. Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211. doi: 10.1177/0333102417738202
8. Do TP, Remmers A, Schytz HW, et al. Red and orange flags for secondary headaches in clinical practice: SNNOOP10 list. Neurology. 2019;92:134-144. doi: 10.1212/WNL.0000000000006697
9. NIH. Migraine. Accessed July 30, 2023.
10. AHS. The American Headache Society position statement on integrating new migraine treatments into clinical practice. Headache. 2019;59:1-18. doi: 10.1111/head.13456
11. Marmura MJ, Silberstein SD, Schwedt TJ. The acute treatment of migraine in adults: the American Headache Society evidence assessment of migraine pharmacotherapies. Headache. 2015;55:3-20. doi: 10.1111/head.12499
12. Mayans L, Walling A. Acute migraine headache: treatment strategies. Am Fam Physician. 2018;97:243-251.
13. Cameron C, Kelly S, Hsieh SC, et al. Triptans in the acute treatment of migraine: a systematic review and network meta-analysis. Headache. 2015;55(suppl 4):221-235. doi: 10.1111/head.12601
14. Becker WJ. Acute migraine treatment. Continuum (Minneap Minn). 2015;21:953-972. doi: 10.1212/CON.0000000000000192
15. Migranal (dihydroergotamine mesylate) Package insert. Valeant Pharmaceuticals North America; 2019. Accessed June 17, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2019/020148Orig1s025lbl.pdf
16. Minen MT, Tanev K, Friedman BW. Evaluation and treatment of migraine in the emergency department: a review. Headache. 2014;54:1131-45. doi: 10.1111/head.12399
17. Durham PL. CGRP-receptor antagonists--a fresh approach to migraine therapy? N Engl J Med. 2004;350:1073-1075. doi: 10.1056/NEJMp048016
18. Ubrelvy (ubrogepant). Package insert. Allergan, Inc.; 2019. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2019/211765s000lbl.pdf
19. Nurtec ODT (rimegepant sulfate). Package insert. Biohaven Pharmaceuticals, Inc.; 2021. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2021/212728s006lbl.pdf
20. Zavzpret (zavegepant). Package insert. Pfizer Labs.; 2023. Accessed July 15, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2023/216386s000lbl.pdf
21. Dodick DW, Lipton RB, Ailani J, et al. Ubrogepant for the treatment of migraine. N Engl J Med. 2019;381:2230-2241. doi: 10.1056/NEJMoa1813049
22. Lipton RB, Dodick DW, Ailani J, et al. Effect of ubrogepant vs placebo on pain and the most bothersome associated symptom in the acute treatment of migraine: the ACHIEVE II randomized clinical trial. JAMA. 2019;322:1887-1898. doi: 10.1001/jama.2019.16711
23. Croop R, Goadsby PJ, Stock DA, et al. Efficacy, safety, and tolerability of rimegepant orally disintegrating tablet for the acute treatment of migraine: a randomised, phase 3, double-blind, placebo-controlled trial. Lancet. 2019;394:737-745. doi: 10.1016/S0140-6736(19)31606-X
24. Lipton RB, Croop R, Stock DA, et al. Safety, tolerability, and efficacy of zavegepant 10 mg nasal spray for the acute treatment of migraine in the USA: a phase 3, double-blind, randomised, placebo-controlled multicentre trial. Lancet Neurol. 2023;22:209-217. doi: 10.1016/S1474-4422(22)00517-8
25. Dodick DW, Lipton RB, Ailani J, et al. Ubrogepant, an acute treatment for migraine, improved patient-reported functional disability and satisfaction in 2 single-attack phase 3 randomized trials, ACHIEVE I and II. Headache. 2020;60:686-700. doi: 10.1111/head.13766
26. Burch R. Migraine and tension-type headache: diagnosis and treatment. Med Clin North Am. 2019;103:215-233. doi:10.1016/j.mcna.2018.10.003
27. Silberstein SD, Holland S, Freitag F, et al. Evidence-based guideline update: pharmacologic treatment for episodic migraine prevention in adults: report of the Quality Standards Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology. 2012;78:1337-1345. doi: 10.1212/WNL.0b013e3182535d20
28. Dodick DW. CGRP ligand and receptor monoclonal antibodies for migraine prevention: evidence review and clinical implications. Cephalalgia. 2019;39:445-458. doi: 10.1177/ 0333102418821662
29. Pringsheim T, Davenport WJ, Becker WJ. Prophylaxis of migraine headache. CMAJ. 2010;182:E269-276. doi: 10.1503/cmaj.081657
30. Vyepti (eptinezumab-jjmr). Package insert. Lundbeck Pharmaceuticals LLV; 2020. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2020/761119s000lbl.pdf
31. Aimovig (erenumab-aooe). Package insert. Amgen Inc.; 2021. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2021/761077s009lbl.pdf
32. Ajovy (fremanezumab-vfrm). Package insert. Teva Pharmaceuticals USA, Inc.; 2018. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2018/761089s000lbl.pdf
33. Emgality (galcanezumab-gnlm). Package insert. Eli Lilly and Company; 2018. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2018/761063s000lbl.pdf
34. Ashina M, Saper J, Cady R, et al. Eptinezumab in episodic migraine: a randomized, double-blind, placebo-controlled study (PROMISE-1). Cephalalgia. 2020;40:241-254. doi: 10.1177/0333102420905132
35. Lipton RB, Goadsby PJ, Smith J, et al. Efficacy and safety of eptinezumab in patients with chronic migraine: PROMISE-2. Neurology. 2020;94:e1365-e1377. doi: 10.1212/WNL.0000000000009169
36. Dodick DW, Ashina M, Brandes JL, et al. ARISE: a phase 3 randomized trial of erenumab for episodic migraine. Cephalalgia. 2018;38:1026-1037. doi: 10.1177/0333102418759786
37. Goadsby PJ, Reuter U, Hallström Y, et al. A controlled trial of erenumab for episodic migraine. N Engl J Med. 2017;377:2123-2132. doi: 10.1056/NEJMoa1705848
38. Reuter U, Goadsby PJ, Lanteri-Minet M, et al. Efficacy and tolerability of erenumab in patients with episodic migraine in whom two-to-four previous preventive treatments were unsuccessful: a randomised, double-blind, placebo-controlled, phase 3b study. Lancet. 2018;392:2280-2287. doi: 10.1016/S0140-6736(18)32534-0
39. Silberstein SD, Dodick DW, Bigal ME, et al. Fremanezumab for the preventive treatment of chronic migraine. N Engl J Med. 2017; 377:2113-2122. doi: 10.1056/NEJMoa1709038
40. Dodick DW, Silberstein SD, Bigal ME, et al. Effect of fremanezumab compared with placebo for prevention of episodic migraine: a randomized clinical trial. JAMA. 2018;319:1999-2008. doi: 10.1001/jama.2018.4853
41. Stauffer VL, Dodick DW, Zhang Q, et al. Evaluation of galcanezumab for the prevention of episodic migraine: the EVOLVE-1 randomized clinical trial. JAMA Neurol. 2018;75:1080-1088. doi: 10.1001/jamaneurol.2018.1212
42. Skljarevski V, Matharu M, Millen BA, et al. Efficacy and safety of galcanezumab for the prevention of episodic migraine: results of the EVOLVE-2 phase 3 randomized controlled clinical trial. Cephalalgia. 2018;38:1442-1454. doi: 10.1177/0333102418779543
43. Detke HC, Goadsby PJ, Wang S, et al. Galcanezumab in chronic migraine: the randomized, double-blind, placebo-controlled REGAIN study. Neurology. 2018;91:e2211-e2221. doi: 10.1212/WNL.0000000000006640
44. Goadsby PJ, Dodick DW, Leone M, at al. Trial of galcanezumab in prevention of episodic cluster headache. N Engl J Med. 2019; 381:132-141. doi: 10.1056/NEJMoa1813440
45. Croop R, Lipton RB, Kudrow D, et al. Oral rimegepant for preventive treatment of migraine: a phase 2/3, randomised, double-blind, placebo-controlled trial. Lancet. 2021;397:51-60. doi: 10.1016/S0140-6736(20)32544-7
46. Ailani J, Lipton RB, Goadsby PJ, et al. Atogepant for the preventive treatment of migraine. N Engl J Med. 2021;385:695-706. doi: 10.1056/NEJMoa2035908
47. Qulipta (atogepant). Package insert. AbbVie; 2021. Accessed June 19, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2021/215206Orig1s000lbl.pdf
48. Han L, Liu Y, Xiong H, et al. CGRP monoclonal antibody for preventive treatment of chronic migraine: an update of meta-analysis. Brain Behav. 2019;9:e01215. doi: 10.1002/brb3.1215
49. Zhu Y, Liu Y, Zhao J, et al. The efficacy and safety of calcitonin gene-related peptide monoclonal antibody for episodic migraine: a meta-analysis. Neurol Sci. 2018;39:2097-2106. doi: 10.1007/s10072-018-3547-3
50. Szperka CL, VanderPluym J, Orr SL, et al. Recommendations on the use of anti-CGRP monoclonal antibodies in children and adolescents. Headache. 2018;58:1658-1669. doi: 10.1111/head.13414
51. Pellesi L, Do TP, Ashina H, et al. Dual therapy with anti-CGRP monoclonal antibodies and botulinum toxin for migraine prevention: is there a rationale? Headache. 2020;60:1056-1065. doi: 10.1111/head.13843
52. D’Antona L, Matharu M. Identifying and managing refractory migraine: barriers and opportunities? J Headache Pain. 2019;20:89. doi: 10.1186/s10194-019-1040-x
53. Cohen F, Armand C, Lipton RB, et al. Efficacy and tolerability of calcitonin gene-related peptide targeted monoclonal antibody medications as add-on therapy to onabotulinumtoxinA in patients with chronic migraine. Pain Med. 2021;1857-1863. doi: 10.1093/pm/pnab093
54. Berman G, Croop R, Kudrow D, et al. Safety of rimegepant, an oral CGRP receptor antagonist, plus CGRP monoclonal antibodies for migraine. Headache. 2020;60:1734-1742. doi: 10.1111/head.13930
PRACTICE RECOMMENDATIONS
› Consider small-molecule calcitonin gene-related peptide (CGRP) receptor antagonists (gepants) for acute migraine treatment after treatment failure of at least 2 non-CGRP first-line therapies. A
› Consider anti-CGRP monoclonal antibodies or gepants for migraine prevention if traditional therapies have proven ineffective or are contraindicated or intolerable to the patient. A
› Add an anti-CGRP monoclonal antibody or gepant to existing preventive treatment if the patient continues to experience migraine. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Using JAK inhibitors for myelofibrosis
“We are thankfully starting to be blessed with more options than we’ve ever had,” he said, but “in the front-line proliferative setting, ruxolitinib has remained the standard of care.” It’s “well established in higher-risk patients and very much an option for very symptomatic lower-risk patients.”
Dr. Hunter helped his colleagues navigate the evolving field of JAK inhibition for myelofibrosis in a presentation titled “Choosing and Properly Using a JAK Inhibitor in Myelofibrosis,”at the Society of Hematologic Oncology annual meeting.
Ruxolitinib was the first JAK inhibitor for myelofibrosis on the U.S. market, approved in 2011. Two more have followed, fedratinib in 2019 and pacritinib in 2022.
A fourth JAK inhibitor for myelofibrosis, momelotinib, is under Food and Drug Administration review with a decision expected shortly.
JAK inhibitors disrupt a key pathogenic pathway in myelofibrosis and are a mainstay of treatment, but Dr. Hunter noted that they should not replace allogeneic transplants in patients who are candidates because transplants remain “the best way to achieve long term survival, especially in higher risk patients.”
He noted that not every patient needs a JAK inhibitor, especially “lower-risk, more asymptomatic patients who are predominantly manifesting with cytopenias. [They] are less likely to benefit.”
Dr. Hunter said that although ruxolitinib remains a treatment of choice, fedratinib “is certainly an option” with comparable rates of symptom control and splenomegaly reduction. Also, while ruxolitinib is dosed according to platelet levels, fedratinib allows for full dosing down to a platelet count of 50 x 109/L.
“But there’s more GI toxicity than with ruxolitinib, especially in the first couple of months,” he said, as well as a black box warning of Wernicke’s encephalopathy. “I generally put all my [fedratinib] patients on thiamine repletion as a precaution.”
One of the most challenging aspects of using JAK inhibitors for myelofibrosis is their tendency to cause cytopenia, particularly anemia and thrombocytopenia, which, ironically, are also hallmarks of myelofibrosis itself.
Although there’s an alternative low-dose ruxolitinib regimen that can be effective in anemic settings, the approval of pacritinib and most likely momelotinib is particularly helpful for cytopenic patients, “a population which historically has been very hard to treat with our prior agents,” Dr. Hunter said.
Pacritinib is approved specifically for patients with platelet counts below 50 x 109/L; momelotinib also included lower platelet counts in several studies. Both agents indirectly boost erythropoiesis with subsequent amelioration of anemia.
“Momelotinib is an important emerging agent for these more anemic patients,” with a spleen response comparable to ruxolitinib and significantly higher rates of transfusion independence, but with lower rates of symptom control, Dr. Hunter said.
Pacritinib “really helps extend the benefit of JAK inhibitors to a group of thrombocytopenic patients who have been hard to treat with ruxolitinib,” with the added potential of improving anemia, although, like fedratinib, it has more GI toxicity, he said.
There are multiple add-on options for JAK inhibitor patients with anemia, including luspatercept, an erythropoiesis-stimulating agent approved for anemia in patients with myelodysplastic syndromes; promising results were reported recently for myelofibrosis.
Fedratinib, pacritinib, and momelotinib all have activity in the second line after ruxolitinib failure, Dr. Hunter noted, but he cautioned that ruxolitinib must be tapered over a few weeks, not stopped abruptly, to avoid withdrawal symptoms. Some clinicians overlap JAK inhibitors a day or two to avoid issues.
“Clinical trials should still be considered in many of these settings,” he said, adding that emerging agents are under development, including multiple combination therapies, often with JAK inhibitors as the background.
No disclosure information was reported.
“We are thankfully starting to be blessed with more options than we’ve ever had,” he said, but “in the front-line proliferative setting, ruxolitinib has remained the standard of care.” It’s “well established in higher-risk patients and very much an option for very symptomatic lower-risk patients.”
Dr. Hunter helped his colleagues navigate the evolving field of JAK inhibition for myelofibrosis in a presentation titled “Choosing and Properly Using a JAK Inhibitor in Myelofibrosis,”at the Society of Hematologic Oncology annual meeting.
Ruxolitinib was the first JAK inhibitor for myelofibrosis on the U.S. market, approved in 2011. Two more have followed, fedratinib in 2019 and pacritinib in 2022.
A fourth JAK inhibitor for myelofibrosis, momelotinib, is under Food and Drug Administration review with a decision expected shortly.
JAK inhibitors disrupt a key pathogenic pathway in myelofibrosis and are a mainstay of treatment, but Dr. Hunter noted that they should not replace allogeneic transplants in patients who are candidates because transplants remain “the best way to achieve long term survival, especially in higher risk patients.”
He noted that not every patient needs a JAK inhibitor, especially “lower-risk, more asymptomatic patients who are predominantly manifesting with cytopenias. [They] are less likely to benefit.”
Dr. Hunter said that although ruxolitinib remains a treatment of choice, fedratinib “is certainly an option” with comparable rates of symptom control and splenomegaly reduction. Also, while ruxolitinib is dosed according to platelet levels, fedratinib allows for full dosing down to a platelet count of 50 x 109/L.
“But there’s more GI toxicity than with ruxolitinib, especially in the first couple of months,” he said, as well as a black box warning of Wernicke’s encephalopathy. “I generally put all my [fedratinib] patients on thiamine repletion as a precaution.”
One of the most challenging aspects of using JAK inhibitors for myelofibrosis is their tendency to cause cytopenia, particularly anemia and thrombocytopenia, which, ironically, are also hallmarks of myelofibrosis itself.
Although there’s an alternative low-dose ruxolitinib regimen that can be effective in anemic settings, the approval of pacritinib and most likely momelotinib is particularly helpful for cytopenic patients, “a population which historically has been very hard to treat with our prior agents,” Dr. Hunter said.
Pacritinib is approved specifically for patients with platelet counts below 50 x 109/L; momelotinib also included lower platelet counts in several studies. Both agents indirectly boost erythropoiesis with subsequent amelioration of anemia.
“Momelotinib is an important emerging agent for these more anemic patients,” with a spleen response comparable to ruxolitinib and significantly higher rates of transfusion independence, but with lower rates of symptom control, Dr. Hunter said.
Pacritinib “really helps extend the benefit of JAK inhibitors to a group of thrombocytopenic patients who have been hard to treat with ruxolitinib,” with the added potential of improving anemia, although, like fedratinib, it has more GI toxicity, he said.
There are multiple add-on options for JAK inhibitor patients with anemia, including luspatercept, an erythropoiesis-stimulating agent approved for anemia in patients with myelodysplastic syndromes; promising results were reported recently for myelofibrosis.
Fedratinib, pacritinib, and momelotinib all have activity in the second line after ruxolitinib failure, Dr. Hunter noted, but he cautioned that ruxolitinib must be tapered over a few weeks, not stopped abruptly, to avoid withdrawal symptoms. Some clinicians overlap JAK inhibitors a day or two to avoid issues.
“Clinical trials should still be considered in many of these settings,” he said, adding that emerging agents are under development, including multiple combination therapies, often with JAK inhibitors as the background.
No disclosure information was reported.
“We are thankfully starting to be blessed with more options than we’ve ever had,” he said, but “in the front-line proliferative setting, ruxolitinib has remained the standard of care.” It’s “well established in higher-risk patients and very much an option for very symptomatic lower-risk patients.”
Dr. Hunter helped his colleagues navigate the evolving field of JAK inhibition for myelofibrosis in a presentation titled “Choosing and Properly Using a JAK Inhibitor in Myelofibrosis,”at the Society of Hematologic Oncology annual meeting.
Ruxolitinib was the first JAK inhibitor for myelofibrosis on the U.S. market, approved in 2011. Two more have followed, fedratinib in 2019 and pacritinib in 2022.
A fourth JAK inhibitor for myelofibrosis, momelotinib, is under Food and Drug Administration review with a decision expected shortly.
JAK inhibitors disrupt a key pathogenic pathway in myelofibrosis and are a mainstay of treatment, but Dr. Hunter noted that they should not replace allogeneic transplants in patients who are candidates because transplants remain “the best way to achieve long term survival, especially in higher risk patients.”
He noted that not every patient needs a JAK inhibitor, especially “lower-risk, more asymptomatic patients who are predominantly manifesting with cytopenias. [They] are less likely to benefit.”
Dr. Hunter said that although ruxolitinib remains a treatment of choice, fedratinib “is certainly an option” with comparable rates of symptom control and splenomegaly reduction. Also, while ruxolitinib is dosed according to platelet levels, fedratinib allows for full dosing down to a platelet count of 50 x 109/L.
“But there’s more GI toxicity than with ruxolitinib, especially in the first couple of months,” he said, as well as a black box warning of Wernicke’s encephalopathy. “I generally put all my [fedratinib] patients on thiamine repletion as a precaution.”
One of the most challenging aspects of using JAK inhibitors for myelofibrosis is their tendency to cause cytopenia, particularly anemia and thrombocytopenia, which, ironically, are also hallmarks of myelofibrosis itself.
Although there’s an alternative low-dose ruxolitinib regimen that can be effective in anemic settings, the approval of pacritinib and most likely momelotinib is particularly helpful for cytopenic patients, “a population which historically has been very hard to treat with our prior agents,” Dr. Hunter said.
Pacritinib is approved specifically for patients with platelet counts below 50 x 109/L; momelotinib also included lower platelet counts in several studies. Both agents indirectly boost erythropoiesis with subsequent amelioration of anemia.
“Momelotinib is an important emerging agent for these more anemic patients,” with a spleen response comparable to ruxolitinib and significantly higher rates of transfusion independence, but with lower rates of symptom control, Dr. Hunter said.
Pacritinib “really helps extend the benefit of JAK inhibitors to a group of thrombocytopenic patients who have been hard to treat with ruxolitinib,” with the added potential of improving anemia, although, like fedratinib, it has more GI toxicity, he said.
There are multiple add-on options for JAK inhibitor patients with anemia, including luspatercept, an erythropoiesis-stimulating agent approved for anemia in patients with myelodysplastic syndromes; promising results were reported recently for myelofibrosis.
Fedratinib, pacritinib, and momelotinib all have activity in the second line after ruxolitinib failure, Dr. Hunter noted, but he cautioned that ruxolitinib must be tapered over a few weeks, not stopped abruptly, to avoid withdrawal symptoms. Some clinicians overlap JAK inhibitors a day or two to avoid issues.
“Clinical trials should still be considered in many of these settings,” he said, adding that emerging agents are under development, including multiple combination therapies, often with JAK inhibitors as the background.
No disclosure information was reported.
FROM SOHO 2023
Implementation and Evaluation of a Clinical Pharmacist Practitioner-Led Pharmacogenomics Service in a Veterans Affairs Hematology and Oncology Clinic
BACKGROUND
The Pharmacogenomic Testing for Veterans (PHASER) program provides preemptive pharmacogenomic testing for Veterans nationally. Program implementation at the Madison VA began in the hematology and oncology (hem/onc) clinics. In these clinics, PHASER test results are reviewed by the hem/onc clinical pharmacist practitioner (CPP) who provides recommendations regarding therapy via an electronic health record note. The purpose of this retrospective chart review was to assess the impact of the CPP on medication management informed by pharmacogenomics.
METHODS
A retrospective chart review was completed for all Veterans enrolled in hem/onc services and offered PHASER testing between April 1, 2022 and November 1, 2022. The number and type of interventions recommended by the hem/onc CPP, acceptance of recommended interventions, and hem/onc CPP time spent were collected for all patients who accepted and completed PHASER testing. Interventions were categorized and descriptive statistics were used to summarize data.
RESULTS
Of the 98 patients reviewed by the CPP, 75 (77%) were prescribed a medication with potential pharmacogenomic implications. At least one actionable recommendation for medication therapy adjustment was identified for 40 (53%) of those patients based on their pharmacogenomic test results. The CPP spent an average of 12 minutes per patient review (range 5 to 30 minutes) and 100% of CPP recommendations were accepted.
CONCLUSIONS
The CPP efficiently reviewed pharmacogenomic test results and made meaningful recommendations for medication therapy adjustments. CPP recommendations were highly accepted in the hem/onc setting.
BACKGROUND
The Pharmacogenomic Testing for Veterans (PHASER) program provides preemptive pharmacogenomic testing for Veterans nationally. Program implementation at the Madison VA began in the hematology and oncology (hem/onc) clinics. In these clinics, PHASER test results are reviewed by the hem/onc clinical pharmacist practitioner (CPP) who provides recommendations regarding therapy via an electronic health record note. The purpose of this retrospective chart review was to assess the impact of the CPP on medication management informed by pharmacogenomics.
METHODS
A retrospective chart review was completed for all Veterans enrolled in hem/onc services and offered PHASER testing between April 1, 2022 and November 1, 2022. The number and type of interventions recommended by the hem/onc CPP, acceptance of recommended interventions, and hem/onc CPP time spent were collected for all patients who accepted and completed PHASER testing. Interventions were categorized and descriptive statistics were used to summarize data.
RESULTS
Of the 98 patients reviewed by the CPP, 75 (77%) were prescribed a medication with potential pharmacogenomic implications. At least one actionable recommendation for medication therapy adjustment was identified for 40 (53%) of those patients based on their pharmacogenomic test results. The CPP spent an average of 12 minutes per patient review (range 5 to 30 minutes) and 100% of CPP recommendations were accepted.
CONCLUSIONS
The CPP efficiently reviewed pharmacogenomic test results and made meaningful recommendations for medication therapy adjustments. CPP recommendations were highly accepted in the hem/onc setting.
BACKGROUND
The Pharmacogenomic Testing for Veterans (PHASER) program provides preemptive pharmacogenomic testing for Veterans nationally. Program implementation at the Madison VA began in the hematology and oncology (hem/onc) clinics. In these clinics, PHASER test results are reviewed by the hem/onc clinical pharmacist practitioner (CPP) who provides recommendations regarding therapy via an electronic health record note. The purpose of this retrospective chart review was to assess the impact of the CPP on medication management informed by pharmacogenomics.
METHODS
A retrospective chart review was completed for all Veterans enrolled in hem/onc services and offered PHASER testing between April 1, 2022 and November 1, 2022. The number and type of interventions recommended by the hem/onc CPP, acceptance of recommended interventions, and hem/onc CPP time spent were collected for all patients who accepted and completed PHASER testing. Interventions were categorized and descriptive statistics were used to summarize data.
RESULTS
Of the 98 patients reviewed by the CPP, 75 (77%) were prescribed a medication with potential pharmacogenomic implications. At least one actionable recommendation for medication therapy adjustment was identified for 40 (53%) of those patients based on their pharmacogenomic test results. The CPP spent an average of 12 minutes per patient review (range 5 to 30 minutes) and 100% of CPP recommendations were accepted.
CONCLUSIONS
The CPP efficiently reviewed pharmacogenomic test results and made meaningful recommendations for medication therapy adjustments. CPP recommendations were highly accepted in the hem/onc setting.
‘Promising’ new txs for most common adult leukemia
The rapid rise of chimeric antigen receptor T (CAR T-cell) therapy has allowed hematologists to make great strides in treating aggressive cases of multiple myeloma and several types of lymphoma and leukemia. But patients with chronic lymphocytic leukemia (CLL), the most common leukemia in adults, have been left out.
“These are the two immunotherapies that have the most potential right now,” said Ohio State University, Columbus, hematologist Kerry A. Rogers, MD, in an interview. She went on to say that these treatments could be a boon for patients with CLL who don’t respond well to targeted therapy drugs or are so young that those medications may not retain effectiveness throughout the patients’ lifespans.
As the American Cancer Society explains, CAR T therapy is a way to get T cells “to fight cancer by changing them in the lab so they can find and destroy cancer cells.” The cells are then returned to the patient.
As the National Cancer Institute says, “If all goes as planned, the CAR T cells will continue to multiply in the patient’s body and, with guidance from their engineered receptor, recognize and kill any cancer cells that harbor the target antigen on their surfaces.”
According to Dr. Rogers, CAR T therapy is less toxic than stem cell transplantation, a related treatment. That means older people can better tolerate it, including many CLL patients in their late 60s and beyond, she said. (Side effects of CAR T therapy include cytokine release syndrome, nervous system impairment, and weakening of the immune system.)
Thus far, CAR T therapy has been approved by the U.S. Food and Drug Administration to treat lymphomas, some forms of leukemia, and multiple myeloma. “Despite the excitement around these therapies, they lead to long-term survival in fewer than half of the patients treated,” cautions the National Cancer Institute, which also notes their high cost: more than $450,000 in one case.
CAR T therapy is not FDA-approved for CLL. “There are many reasons why CAR T is less effective in patients with CLL versus other lymphomas,” said Lee Greenberger, PhD, chief scientific officer of the Leukemia & Lymphoma Society, in an interview. “For one, many patients with heavily pretreated CLL – prior to any use of CAR T – have mutations that are known to be difficult to treat. Dysfunctional T cells are also common in patients with CLL, and there’s often a lower number of available T-cells to manufacture.”
The results of a phase 1/2 trial released in August 2023 offered new insight about CAR T for CLL. In the open-label trial reported in The Lancet, 117 U.S. patients with CLL or small lymphocytic lymphoma underwent a form of CAR T therapy called lisocabtagene maraleucel after failing treatment with two lines of therapy, including a Bruton´s tyrosine kinase inhibitor. Among 49 patients at a specific dose, “the rate of complete response or remission (including with incomplete marrow recovery) was statistically significant at 18%,” the researchers reported. A total of 51 patients in the entire study died.
The rate of undetectable minimal residual disease blood was 64%. That rate is impressive, said University of Texas MD Anderson Cancer Center leukemia specialist Nitin Jain, MD, in an interview. It’s not nearly as high as researchers have seen in other disease settings, but it’s “a good, good thing for these patients. We’ll have to see in the longer follow-up how these patients fare 2, 3, or 4 years down the line.”
Dr. Rogers, the Ohio physician, said doctors had hoped durable benefit in the Lancet study would be more impressive. An important factor limiting its value may be the aggressiveness of the disease in patients who have already failed several treatments, she said. “The efficacy of CAR T might be improved by giving it as an earlier line of therapy before the CLL has become this aggressive. But it’s difficult to propose that you should use this before a Bruton´s tyrosine kinase inhibitor or venetoclax because it’s expensive and difficult.”
What’s next for CART T research in CLL? Understanding the best timing for treatment will be key, Dr. Rogers said.
The Leukemia & Lymphoma Society’s Dr. Greenberger predicted that “we will begin to see CAR T explored in CLL patients whose disease has a high risk of failing approved agents, such as Bruton´s tyrosine kinase and B cell lymphoma 2 inhibitors. However, CLL patients may still receive prior therapy with more effective Bruton’s tyrosine kinase or B cell lymphoma 2 inhibitors in the future before using CAR T. This will likely be heightened as more Bruton´s tyrosine kinase inhibitors become generic in the next 5 to 10 years and, hopefully, less expensive than CAR T therapy.”
In the big picture, he said, “treatment of CLL with CAR T is possible, but still needs significant improvements if it is to become a mainline therapy in the future.”
CAR T therapy remains available via clinical trials, and Dr. Rogers said it is “currently an important option for patients whose CLL has become resistant to standard targeted agents. We can certainly expect to extend someone’s expected survival by years if they have a favorable response.” She acknowledged that the cost is quite high, but noted that targeted therapies are also expensive, especially over the long term. They can run to $10,000-$20,000 a month. Bispecific antibodies are also being explored as potential therapy for CLL. “They’re really exciting,” Dr. Rogers said, with the potential to spur responses similar to those from CAR T therapy.
A 2022 review described these drugs as “molecules that combine antibody-directed therapies with cellular mediated immunotherapy.” The FDA explains that “by targeting two antigens or epitopes, they can cause multiple physiological or antitumor responses, which may be independent or connected.”
According to Dr. Greenberger, many bispecifics are in clinical trials now. However, “in the context of CLL, actually, the data is actually very, very limited. The development is just starting, and there are phase 1 and phase 2 trials ongoing.”
But data from lymphoma trials are encouraging, he said, and bispecifics “are actually looking as good as CAR T in some settings.”
Regimens can be a challenge for patients taking bispecifics, Dr. Greenberger said. “Repeat dosing with a step-up dosing approach to start is typically required when treating lymphoma.”
On the other hand, Dr. Rogers noted that antibody treatment can be easier for hematologists to arrange than CAR T therapy and stem cell transplants. “From an administrative side, there’s not as many things you need to have set up. So it’s able to be administered in a wider variety of settings,” she said,
Bispecific side effects include cytokine release syndrome and neurotoxicity as well as infusion reactions, Dr. Greenberger said, adding that “I would not exclude cost as a challenge.”
According to Formulary Watch, the bispecific Columvi (glofitamab-gxbm), which recently gained FDA approval to treat diffuse large B-cell lymphoma, is estimated to cost $350,000 for an 8.5-month round of treatment. Reuters reported that the bispecific Talvey (talquetamab-tgvs), which just received FDA approval to treat multiple myeloma, is estimated to cost $270,000-$360,000 for 6-8 months of treatment.
For now, bispecific trials “are mostly now reserved for patients with CLL who become resistant to our current standard targeted agents,” Dr. Rogers said. “It’s a little unclear if you can do CAR T therapy first and then bispecifics, or bispecifics and then CAR T therapy.”
What’s coming next for bispecifics? “On the horizon is better ease of administration, which is already being addressed by subcutaneous dosing for some bispecifics in lymphomas,” Dr. Greenberger said. “There’s also the possibility of combining bispecifics with conventional therapy.”
Dr. Rogers discloses ties with Genentech, AbbVie, Novartis, AstraZeneca, Janssen, Pharmacyclics, Beigene, and LOXO@Lilly. Dr. Greenberger discloses employment with the Leukemia & Lymphoma Society, which supports academic grants and a venture philanthropy via the Therapy Acceleration Program.
Dr. Jain reports ties with Pharmacyclics, AbbVie, Genentech, AstraZeneca, Pfizer, and numerous other disclosures.
The rapid rise of chimeric antigen receptor T (CAR T-cell) therapy has allowed hematologists to make great strides in treating aggressive cases of multiple myeloma and several types of lymphoma and leukemia. But patients with chronic lymphocytic leukemia (CLL), the most common leukemia in adults, have been left out.
“These are the two immunotherapies that have the most potential right now,” said Ohio State University, Columbus, hematologist Kerry A. Rogers, MD, in an interview. She went on to say that these treatments could be a boon for patients with CLL who don’t respond well to targeted therapy drugs or are so young that those medications may not retain effectiveness throughout the patients’ lifespans.
As the American Cancer Society explains, CAR T therapy is a way to get T cells “to fight cancer by changing them in the lab so they can find and destroy cancer cells.” The cells are then returned to the patient.
As the National Cancer Institute says, “If all goes as planned, the CAR T cells will continue to multiply in the patient’s body and, with guidance from their engineered receptor, recognize and kill any cancer cells that harbor the target antigen on their surfaces.”
According to Dr. Rogers, CAR T therapy is less toxic than stem cell transplantation, a related treatment. That means older people can better tolerate it, including many CLL patients in their late 60s and beyond, she said. (Side effects of CAR T therapy include cytokine release syndrome, nervous system impairment, and weakening of the immune system.)
Thus far, CAR T therapy has been approved by the U.S. Food and Drug Administration to treat lymphomas, some forms of leukemia, and multiple myeloma. “Despite the excitement around these therapies, they lead to long-term survival in fewer than half of the patients treated,” cautions the National Cancer Institute, which also notes their high cost: more than $450,000 in one case.
CAR T therapy is not FDA-approved for CLL. “There are many reasons why CAR T is less effective in patients with CLL versus other lymphomas,” said Lee Greenberger, PhD, chief scientific officer of the Leukemia & Lymphoma Society, in an interview. “For one, many patients with heavily pretreated CLL – prior to any use of CAR T – have mutations that are known to be difficult to treat. Dysfunctional T cells are also common in patients with CLL, and there’s often a lower number of available T-cells to manufacture.”
The results of a phase 1/2 trial released in August 2023 offered new insight about CAR T for CLL. In the open-label trial reported in The Lancet, 117 U.S. patients with CLL or small lymphocytic lymphoma underwent a form of CAR T therapy called lisocabtagene maraleucel after failing treatment with two lines of therapy, including a Bruton´s tyrosine kinase inhibitor. Among 49 patients at a specific dose, “the rate of complete response or remission (including with incomplete marrow recovery) was statistically significant at 18%,” the researchers reported. A total of 51 patients in the entire study died.
The rate of undetectable minimal residual disease blood was 64%. That rate is impressive, said University of Texas MD Anderson Cancer Center leukemia specialist Nitin Jain, MD, in an interview. It’s not nearly as high as researchers have seen in other disease settings, but it’s “a good, good thing for these patients. We’ll have to see in the longer follow-up how these patients fare 2, 3, or 4 years down the line.”
Dr. Rogers, the Ohio physician, said doctors had hoped durable benefit in the Lancet study would be more impressive. An important factor limiting its value may be the aggressiveness of the disease in patients who have already failed several treatments, she said. “The efficacy of CAR T might be improved by giving it as an earlier line of therapy before the CLL has become this aggressive. But it’s difficult to propose that you should use this before a Bruton´s tyrosine kinase inhibitor or venetoclax because it’s expensive and difficult.”
What’s next for CART T research in CLL? Understanding the best timing for treatment will be key, Dr. Rogers said.
The Leukemia & Lymphoma Society’s Dr. Greenberger predicted that “we will begin to see CAR T explored in CLL patients whose disease has a high risk of failing approved agents, such as Bruton´s tyrosine kinase and B cell lymphoma 2 inhibitors. However, CLL patients may still receive prior therapy with more effective Bruton’s tyrosine kinase or B cell lymphoma 2 inhibitors in the future before using CAR T. This will likely be heightened as more Bruton´s tyrosine kinase inhibitors become generic in the next 5 to 10 years and, hopefully, less expensive than CAR T therapy.”
In the big picture, he said, “treatment of CLL with CAR T is possible, but still needs significant improvements if it is to become a mainline therapy in the future.”
CAR T therapy remains available via clinical trials, and Dr. Rogers said it is “currently an important option for patients whose CLL has become resistant to standard targeted agents. We can certainly expect to extend someone’s expected survival by years if they have a favorable response.” She acknowledged that the cost is quite high, but noted that targeted therapies are also expensive, especially over the long term. They can run to $10,000-$20,000 a month. Bispecific antibodies are also being explored as potential therapy for CLL. “They’re really exciting,” Dr. Rogers said, with the potential to spur responses similar to those from CAR T therapy.
A 2022 review described these drugs as “molecules that combine antibody-directed therapies with cellular mediated immunotherapy.” The FDA explains that “by targeting two antigens or epitopes, they can cause multiple physiological or antitumor responses, which may be independent or connected.”
According to Dr. Greenberger, many bispecifics are in clinical trials now. However, “in the context of CLL, actually, the data is actually very, very limited. The development is just starting, and there are phase 1 and phase 2 trials ongoing.”
But data from lymphoma trials are encouraging, he said, and bispecifics “are actually looking as good as CAR T in some settings.”
Regimens can be a challenge for patients taking bispecifics, Dr. Greenberger said. “Repeat dosing with a step-up dosing approach to start is typically required when treating lymphoma.”
On the other hand, Dr. Rogers noted that antibody treatment can be easier for hematologists to arrange than CAR T therapy and stem cell transplants. “From an administrative side, there’s not as many things you need to have set up. So it’s able to be administered in a wider variety of settings,” she said,
Bispecific side effects include cytokine release syndrome and neurotoxicity as well as infusion reactions, Dr. Greenberger said, adding that “I would not exclude cost as a challenge.”
According to Formulary Watch, the bispecific Columvi (glofitamab-gxbm), which recently gained FDA approval to treat diffuse large B-cell lymphoma, is estimated to cost $350,000 for an 8.5-month round of treatment. Reuters reported that the bispecific Talvey (talquetamab-tgvs), which just received FDA approval to treat multiple myeloma, is estimated to cost $270,000-$360,000 for 6-8 months of treatment.
For now, bispecific trials “are mostly now reserved for patients with CLL who become resistant to our current standard targeted agents,” Dr. Rogers said. “It’s a little unclear if you can do CAR T therapy first and then bispecifics, or bispecifics and then CAR T therapy.”
What’s coming next for bispecifics? “On the horizon is better ease of administration, which is already being addressed by subcutaneous dosing for some bispecifics in lymphomas,” Dr. Greenberger said. “There’s also the possibility of combining bispecifics with conventional therapy.”
Dr. Rogers discloses ties with Genentech, AbbVie, Novartis, AstraZeneca, Janssen, Pharmacyclics, Beigene, and LOXO@Lilly. Dr. Greenberger discloses employment with the Leukemia & Lymphoma Society, which supports academic grants and a venture philanthropy via the Therapy Acceleration Program.
Dr. Jain reports ties with Pharmacyclics, AbbVie, Genentech, AstraZeneca, Pfizer, and numerous other disclosures.
The rapid rise of chimeric antigen receptor T (CAR T-cell) therapy has allowed hematologists to make great strides in treating aggressive cases of multiple myeloma and several types of lymphoma and leukemia. But patients with chronic lymphocytic leukemia (CLL), the most common leukemia in adults, have been left out.
“These are the two immunotherapies that have the most potential right now,” said Ohio State University, Columbus, hematologist Kerry A. Rogers, MD, in an interview. She went on to say that these treatments could be a boon for patients with CLL who don’t respond well to targeted therapy drugs or are so young that those medications may not retain effectiveness throughout the patients’ lifespans.
As the American Cancer Society explains, CAR T therapy is a way to get T cells “to fight cancer by changing them in the lab so they can find and destroy cancer cells.” The cells are then returned to the patient.
As the National Cancer Institute says, “If all goes as planned, the CAR T cells will continue to multiply in the patient’s body and, with guidance from their engineered receptor, recognize and kill any cancer cells that harbor the target antigen on their surfaces.”
According to Dr. Rogers, CAR T therapy is less toxic than stem cell transplantation, a related treatment. That means older people can better tolerate it, including many CLL patients in their late 60s and beyond, she said. (Side effects of CAR T therapy include cytokine release syndrome, nervous system impairment, and weakening of the immune system.)
Thus far, CAR T therapy has been approved by the U.S. Food and Drug Administration to treat lymphomas, some forms of leukemia, and multiple myeloma. “Despite the excitement around these therapies, they lead to long-term survival in fewer than half of the patients treated,” cautions the National Cancer Institute, which also notes their high cost: more than $450,000 in one case.
CAR T therapy is not FDA-approved for CLL. “There are many reasons why CAR T is less effective in patients with CLL versus other lymphomas,” said Lee Greenberger, PhD, chief scientific officer of the Leukemia & Lymphoma Society, in an interview. “For one, many patients with heavily pretreated CLL – prior to any use of CAR T – have mutations that are known to be difficult to treat. Dysfunctional T cells are also common in patients with CLL, and there’s often a lower number of available T-cells to manufacture.”
The results of a phase 1/2 trial released in August 2023 offered new insight about CAR T for CLL. In the open-label trial reported in The Lancet, 117 U.S. patients with CLL or small lymphocytic lymphoma underwent a form of CAR T therapy called lisocabtagene maraleucel after failing treatment with two lines of therapy, including a Bruton´s tyrosine kinase inhibitor. Among 49 patients at a specific dose, “the rate of complete response or remission (including with incomplete marrow recovery) was statistically significant at 18%,” the researchers reported. A total of 51 patients in the entire study died.
The rate of undetectable minimal residual disease blood was 64%. That rate is impressive, said University of Texas MD Anderson Cancer Center leukemia specialist Nitin Jain, MD, in an interview. It’s not nearly as high as researchers have seen in other disease settings, but it’s “a good, good thing for these patients. We’ll have to see in the longer follow-up how these patients fare 2, 3, or 4 years down the line.”
Dr. Rogers, the Ohio physician, said doctors had hoped durable benefit in the Lancet study would be more impressive. An important factor limiting its value may be the aggressiveness of the disease in patients who have already failed several treatments, she said. “The efficacy of CAR T might be improved by giving it as an earlier line of therapy before the CLL has become this aggressive. But it’s difficult to propose that you should use this before a Bruton´s tyrosine kinase inhibitor or venetoclax because it’s expensive and difficult.”
What’s next for CART T research in CLL? Understanding the best timing for treatment will be key, Dr. Rogers said.
The Leukemia & Lymphoma Society’s Dr. Greenberger predicted that “we will begin to see CAR T explored in CLL patients whose disease has a high risk of failing approved agents, such as Bruton´s tyrosine kinase and B cell lymphoma 2 inhibitors. However, CLL patients may still receive prior therapy with more effective Bruton’s tyrosine kinase or B cell lymphoma 2 inhibitors in the future before using CAR T. This will likely be heightened as more Bruton´s tyrosine kinase inhibitors become generic in the next 5 to 10 years and, hopefully, less expensive than CAR T therapy.”
In the big picture, he said, “treatment of CLL with CAR T is possible, but still needs significant improvements if it is to become a mainline therapy in the future.”
CAR T therapy remains available via clinical trials, and Dr. Rogers said it is “currently an important option for patients whose CLL has become resistant to standard targeted agents. We can certainly expect to extend someone’s expected survival by years if they have a favorable response.” She acknowledged that the cost is quite high, but noted that targeted therapies are also expensive, especially over the long term. They can run to $10,000-$20,000 a month. Bispecific antibodies are also being explored as potential therapy for CLL. “They’re really exciting,” Dr. Rogers said, with the potential to spur responses similar to those from CAR T therapy.
A 2022 review described these drugs as “molecules that combine antibody-directed therapies with cellular mediated immunotherapy.” The FDA explains that “by targeting two antigens or epitopes, they can cause multiple physiological or antitumor responses, which may be independent or connected.”
According to Dr. Greenberger, many bispecifics are in clinical trials now. However, “in the context of CLL, actually, the data is actually very, very limited. The development is just starting, and there are phase 1 and phase 2 trials ongoing.”
But data from lymphoma trials are encouraging, he said, and bispecifics “are actually looking as good as CAR T in some settings.”
Regimens can be a challenge for patients taking bispecifics, Dr. Greenberger said. “Repeat dosing with a step-up dosing approach to start is typically required when treating lymphoma.”
On the other hand, Dr. Rogers noted that antibody treatment can be easier for hematologists to arrange than CAR T therapy and stem cell transplants. “From an administrative side, there’s not as many things you need to have set up. So it’s able to be administered in a wider variety of settings,” she said,
Bispecific side effects include cytokine release syndrome and neurotoxicity as well as infusion reactions, Dr. Greenberger said, adding that “I would not exclude cost as a challenge.”
According to Formulary Watch, the bispecific Columvi (glofitamab-gxbm), which recently gained FDA approval to treat diffuse large B-cell lymphoma, is estimated to cost $350,000 for an 8.5-month round of treatment. Reuters reported that the bispecific Talvey (talquetamab-tgvs), which just received FDA approval to treat multiple myeloma, is estimated to cost $270,000-$360,000 for 6-8 months of treatment.
For now, bispecific trials “are mostly now reserved for patients with CLL who become resistant to our current standard targeted agents,” Dr. Rogers said. “It’s a little unclear if you can do CAR T therapy first and then bispecifics, or bispecifics and then CAR T therapy.”
What’s coming next for bispecifics? “On the horizon is better ease of administration, which is already being addressed by subcutaneous dosing for some bispecifics in lymphomas,” Dr. Greenberger said. “There’s also the possibility of combining bispecifics with conventional therapy.”
Dr. Rogers discloses ties with Genentech, AbbVie, Novartis, AstraZeneca, Janssen, Pharmacyclics, Beigene, and LOXO@Lilly. Dr. Greenberger discloses employment with the Leukemia & Lymphoma Society, which supports academic grants and a venture philanthropy via the Therapy Acceleration Program.
Dr. Jain reports ties with Pharmacyclics, AbbVie, Genentech, AstraZeneca, Pfizer, and numerous other disclosures.
Barbie has an anxiety disorder
And it’s a great time to be a therapist
The Barbie movie is generating a lot of feelings, ranging from praise to vitriol. However one feels about the movie, let’s all pause and reflect for a moment on the fact that the number-one grossing film of 2023 is about our childhood doll trying to treat her anxiety disorder.
“Life imitates art more than art imitates life.” So said Oscar Wilde in 1889.
When my adult daughter, a childhood Barbie enthusiast, asked me to see the film, we put on pink and went. Twice. Little did I know that it would stir up so many thoughts and feelings. The one I want to share is how blessed I feel at this moment in time to be a mental health care provider! No longer is mental health something to be whispered about at the water cooler; instead, even Barbie is suffering. And with all the controversy in the press about the movie, no one seems at all surprised by this storyline.
I was raised by two child psychiatrists and have been practicing as an adult psychiatrist since 1991. The start of the pandemic was the most difficult time of my career, as almost every patient was struggling simultaneously, as was I. Three long years later, we are gradually emerging from our shared trauma. How ironic, now with the opportunity to go back to work, I have elected to maintain the majority of my practice online from home. It seems that most patients and providers prefer this mode of treatment, with a full 90 percent of practitioners saying they are using a hybrid model.
As mental health professionals, we know that anywhere from 3% to 49% of those experiencing trauma will develop posttraumatic stress disorder (PTSD), and we have been trained to treat them.
But what happens when an entire global population is exposed simultaneously to trauma? Historians and social scientists refer to such events by many different names, such as: Singularity, Black Swan Event, and Tipping Point. These events are incredibly rare, and afterwards everything is different. These global traumas always lead to massive change.
I think we are at that tipping point. This is the singularity. This is our Black Swan Event. Within a 3-year span, we have experienced the following:
- A global traumatic event (COVID-19).
- A sudden and seemingly permanent shift from office to remote video meetings mostly from home.
- Upending of traditional fundamentals of the stock market as the game literally stopped in January 2021.
- Rapid and widespread availability of Artificial Intelligence.
- The first generation to be fully raised on the Internet and social media (Gen Z) is now entering the workforce.
- Ongoing war in Ukraine.
That’s already an overwhelming list, and I could go on, but let’s get back to Barbie’s anxiety disorder.
The awareness about and acceptance of mental health issues has never been higher. The access to treatment never greater. There are now more online therapy options than ever. Treatment options have dramatically expanded in recent years, from Transcranial Magnetic Stimulation (TMS) to ketamine centers and psychedelics, as well as more mainstream options such as dialectical behavior therapy (DBT), cognitive behavioral therapy (CBT), selective serotonin reuptake inhibitors (SSRIs), and so many more.
What is particularly unique about this moment is the direct access to care. Self-help books abound with many making it to the New York Times bestseller list. YouTube is loaded with fantastic content on overcoming many mental health issues, although one should be careful with selecting reliable sources. Apps like HeadSpace and Calm are being downloaded by millions of people around the globe. Investors provided a record-breaking $1.5 billion to mental health startups in 2020 alone.
For most practitioners, our phones have been ringing off the hook since 2020. Applications to psychology, psychiatric residency, social work, and counseling degree programs are on the rise, with workforce shortages expected to continue for decades. Psychological expertise has been embraced by businesses especially for DEI (diversity, equity, and inclusion). Mental health experts are the most asked-for experts through media request services. Elite athletes are talking openly about bringing us on their teams.
In this unique moment, when everything seems set to transform into something else, it is time for mental health professionals to exert some agency and influence over where mental health will go from here. I think the next frontier for mental health specialists is to figure out how to speak collectively and help guide society.
Neil Howe, in his sweeping book “The Fourth Turning is Here,” says we have another 10 years in this “Millennial Crisis” phase. He calls this our “winter,” and it remains to be seen how we will emerge from our current challenges. I think we can make a difference.
If the Barbie movie is indeed a canary in the coal mine, I see positive trends ahead as we move past some of the societal and structural issues facing us, and work together to create a more open and egalitarian society. We must find creative solutions that will solve truly massive problems threatening our well-being and perhaps even our existence.
I am so grateful to be able to continue to practice and share my thoughts with you here from my home office, and I hope you can take a break and see this movie, which is not only entertaining but also thought- and emotion-provoking.
Dr. Ritvo has almost 30 years’ experience in psychiatry and is currently practicing telemedicine. She is the author of “Bekindr – The Transformative Power of Kindness” (Hellertown, Pa.: Momosa Publishing, 2018). She has no conflicts of interest.
And it’s a great time to be a therapist
And it’s a great time to be a therapist
The Barbie movie is generating a lot of feelings, ranging from praise to vitriol. However one feels about the movie, let’s all pause and reflect for a moment on the fact that the number-one grossing film of 2023 is about our childhood doll trying to treat her anxiety disorder.
“Life imitates art more than art imitates life.” So said Oscar Wilde in 1889.
When my adult daughter, a childhood Barbie enthusiast, asked me to see the film, we put on pink and went. Twice. Little did I know that it would stir up so many thoughts and feelings. The one I want to share is how blessed I feel at this moment in time to be a mental health care provider! No longer is mental health something to be whispered about at the water cooler; instead, even Barbie is suffering. And with all the controversy in the press about the movie, no one seems at all surprised by this storyline.
I was raised by two child psychiatrists and have been practicing as an adult psychiatrist since 1991. The start of the pandemic was the most difficult time of my career, as almost every patient was struggling simultaneously, as was I. Three long years later, we are gradually emerging from our shared trauma. How ironic, now with the opportunity to go back to work, I have elected to maintain the majority of my practice online from home. It seems that most patients and providers prefer this mode of treatment, with a full 90 percent of practitioners saying they are using a hybrid model.
As mental health professionals, we know that anywhere from 3% to 49% of those experiencing trauma will develop posttraumatic stress disorder (PTSD), and we have been trained to treat them.
But what happens when an entire global population is exposed simultaneously to trauma? Historians and social scientists refer to such events by many different names, such as: Singularity, Black Swan Event, and Tipping Point. These events are incredibly rare, and afterwards everything is different. These global traumas always lead to massive change.
I think we are at that tipping point. This is the singularity. This is our Black Swan Event. Within a 3-year span, we have experienced the following:
- A global traumatic event (COVID-19).
- A sudden and seemingly permanent shift from office to remote video meetings mostly from home.
- Upending of traditional fundamentals of the stock market as the game literally stopped in January 2021.
- Rapid and widespread availability of Artificial Intelligence.
- The first generation to be fully raised on the Internet and social media (Gen Z) is now entering the workforce.
- Ongoing war in Ukraine.
That’s already an overwhelming list, and I could go on, but let’s get back to Barbie’s anxiety disorder.
The awareness about and acceptance of mental health issues has never been higher. The access to treatment never greater. There are now more online therapy options than ever. Treatment options have dramatically expanded in recent years, from Transcranial Magnetic Stimulation (TMS) to ketamine centers and psychedelics, as well as more mainstream options such as dialectical behavior therapy (DBT), cognitive behavioral therapy (CBT), selective serotonin reuptake inhibitors (SSRIs), and so many more.
What is particularly unique about this moment is the direct access to care. Self-help books abound with many making it to the New York Times bestseller list. YouTube is loaded with fantastic content on overcoming many mental health issues, although one should be careful with selecting reliable sources. Apps like HeadSpace and Calm are being downloaded by millions of people around the globe. Investors provided a record-breaking $1.5 billion to mental health startups in 2020 alone.
For most practitioners, our phones have been ringing off the hook since 2020. Applications to psychology, psychiatric residency, social work, and counseling degree programs are on the rise, with workforce shortages expected to continue for decades. Psychological expertise has been embraced by businesses especially for DEI (diversity, equity, and inclusion). Mental health experts are the most asked-for experts through media request services. Elite athletes are talking openly about bringing us on their teams.
In this unique moment, when everything seems set to transform into something else, it is time for mental health professionals to exert some agency and influence over where mental health will go from here. I think the next frontier for mental health specialists is to figure out how to speak collectively and help guide society.
Neil Howe, in his sweeping book “The Fourth Turning is Here,” says we have another 10 years in this “Millennial Crisis” phase. He calls this our “winter,” and it remains to be seen how we will emerge from our current challenges. I think we can make a difference.
If the Barbie movie is indeed a canary in the coal mine, I see positive trends ahead as we move past some of the societal and structural issues facing us, and work together to create a more open and egalitarian society. We must find creative solutions that will solve truly massive problems threatening our well-being and perhaps even our existence.
I am so grateful to be able to continue to practice and share my thoughts with you here from my home office, and I hope you can take a break and see this movie, which is not only entertaining but also thought- and emotion-provoking.
Dr. Ritvo has almost 30 years’ experience in psychiatry and is currently practicing telemedicine. She is the author of “Bekindr – The Transformative Power of Kindness” (Hellertown, Pa.: Momosa Publishing, 2018). She has no conflicts of interest.
The Barbie movie is generating a lot of feelings, ranging from praise to vitriol. However one feels about the movie, let’s all pause and reflect for a moment on the fact that the number-one grossing film of 2023 is about our childhood doll trying to treat her anxiety disorder.
“Life imitates art more than art imitates life.” So said Oscar Wilde in 1889.
When my adult daughter, a childhood Barbie enthusiast, asked me to see the film, we put on pink and went. Twice. Little did I know that it would stir up so many thoughts and feelings. The one I want to share is how blessed I feel at this moment in time to be a mental health care provider! No longer is mental health something to be whispered about at the water cooler; instead, even Barbie is suffering. And with all the controversy in the press about the movie, no one seems at all surprised by this storyline.
I was raised by two child psychiatrists and have been practicing as an adult psychiatrist since 1991. The start of the pandemic was the most difficult time of my career, as almost every patient was struggling simultaneously, as was I. Three long years later, we are gradually emerging from our shared trauma. How ironic, now with the opportunity to go back to work, I have elected to maintain the majority of my practice online from home. It seems that most patients and providers prefer this mode of treatment, with a full 90 percent of practitioners saying they are using a hybrid model.
As mental health professionals, we know that anywhere from 3% to 49% of those experiencing trauma will develop posttraumatic stress disorder (PTSD), and we have been trained to treat them.
But what happens when an entire global population is exposed simultaneously to trauma? Historians and social scientists refer to such events by many different names, such as: Singularity, Black Swan Event, and Tipping Point. These events are incredibly rare, and afterwards everything is different. These global traumas always lead to massive change.
I think we are at that tipping point. This is the singularity. This is our Black Swan Event. Within a 3-year span, we have experienced the following:
- A global traumatic event (COVID-19).
- A sudden and seemingly permanent shift from office to remote video meetings mostly from home.
- Upending of traditional fundamentals of the stock market as the game literally stopped in January 2021.
- Rapid and widespread availability of Artificial Intelligence.
- The first generation to be fully raised on the Internet and social media (Gen Z) is now entering the workforce.
- Ongoing war in Ukraine.
That’s already an overwhelming list, and I could go on, but let’s get back to Barbie’s anxiety disorder.
The awareness about and acceptance of mental health issues has never been higher. The access to treatment never greater. There are now more online therapy options than ever. Treatment options have dramatically expanded in recent years, from Transcranial Magnetic Stimulation (TMS) to ketamine centers and psychedelics, as well as more mainstream options such as dialectical behavior therapy (DBT), cognitive behavioral therapy (CBT), selective serotonin reuptake inhibitors (SSRIs), and so many more.
What is particularly unique about this moment is the direct access to care. Self-help books abound with many making it to the New York Times bestseller list. YouTube is loaded with fantastic content on overcoming many mental health issues, although one should be careful with selecting reliable sources. Apps like HeadSpace and Calm are being downloaded by millions of people around the globe. Investors provided a record-breaking $1.5 billion to mental health startups in 2020 alone.
For most practitioners, our phones have been ringing off the hook since 2020. Applications to psychology, psychiatric residency, social work, and counseling degree programs are on the rise, with workforce shortages expected to continue for decades. Psychological expertise has been embraced by businesses especially for DEI (diversity, equity, and inclusion). Mental health experts are the most asked-for experts through media request services. Elite athletes are talking openly about bringing us on their teams.
In this unique moment, when everything seems set to transform into something else, it is time for mental health professionals to exert some agency and influence over where mental health will go from here. I think the next frontier for mental health specialists is to figure out how to speak collectively and help guide society.
Neil Howe, in his sweeping book “The Fourth Turning is Here,” says we have another 10 years in this “Millennial Crisis” phase. He calls this our “winter,” and it remains to be seen how we will emerge from our current challenges. I think we can make a difference.
If the Barbie movie is indeed a canary in the coal mine, I see positive trends ahead as we move past some of the societal and structural issues facing us, and work together to create a more open and egalitarian society. We must find creative solutions that will solve truly massive problems threatening our well-being and perhaps even our existence.
I am so grateful to be able to continue to practice and share my thoughts with you here from my home office, and I hope you can take a break and see this movie, which is not only entertaining but also thought- and emotion-provoking.
Dr. Ritvo has almost 30 years’ experience in psychiatry and is currently practicing telemedicine. She is the author of “Bekindr – The Transformative Power of Kindness” (Hellertown, Pa.: Momosa Publishing, 2018). She has no conflicts of interest.
CHP/CCUS: Low blood cancer risk for most patients
The reason is that patients will inevitably “go online and see that [the conditions are] associated with lots of bad things; it can really cause patients psychosocial harm if there is no one to explain what their risk is and also provide risk-specific management,” Dr. Weeks said at the annual meeting of the Society of Hematologic Oncology in Houston.
CHIP and CCUS are precursors of myeloid malignancies but for most patients, the risk of progression is less than 1%. CHIPS and CCUS are also associated with cardiovascular, rheumatologic, hepatic, and other diseases.
CHIP is defined by somatic mutations in myeloid malignancy driver genes with a variant allele fraction of 2% or more; CCUS is when those molecular features are accompanied by an unexplained and persistent anemia, thrombocytopenia, or neutropenia.
A small 2017 study suggested that about a third of patients with otherwise unexplained cytopenias have CCUS.
With the increasing use of next generation sequencing for tissue and liquid biopsies and other uses, the incidental diagnosis of both conditions is increasing.
Fortunately, Dr. Weeks’ group recently published a tool for predicting the risk of progression to myeloid malignancy.
Their “clonal hematopoiesis risk score” (CHRS) was developed and validated in over 400,000 healthy volunteers in the UK Biobank, with additional validation in cohorts from Dana Farber and the University of Pavia, Italy.
The CHRS incorporates eight high-risk genetic and clinical prognostic factors, including the type and number of genetic mutations in blood cells, factors related to red blood cell volume, and age over 65. It’s available online.
“You just input the patient’s information and it spits out if the patient is low, intermediate, or high risk for progression to any myeloid malignancy,” Dr. Weeks told her audience.
High-risk patients have about a 50% 10-year cumulative incidence of myeloid malignancy. The large majority of patients are low risk, however, and have a 10-year cumulative incidence of less than 1%. Patients in the middle have a 10-year risk of about 8%.
The low-risk group “is the population of people who probably don’t need to see a specialist,” and can be followed with an annual CBC with their primary care doctors plus further workup with any clinical change. Patients should also be evaluated for cardiovascular and other comorbidity risks.
“It’s the high-risk group we worry most about,” Dr. Weeks said. “We see them more often and repeat the next-generation sequencing” annually with a CBC at least every 6 months and a bone marrow biopsy with any clinical change.
“This is the population we would shuttle towards a clinical trial, as this is the population most likely to benefit,” she said.
The overarching goal of the several ongoing studies in CHIP/CCUS is to find a way to prevent progression to blood cancer. They range from prospective cohorts and single arm pilot studies to randomized clinical trials. One trial is evaluating canakinumab to prevent progression. “Intervention in clonal hematopoiesis might have the dual benefit of both preventing hematologic malignancy as well as reducing [the] inflammatory comorbidities,” Dr. Weeks said.
The reason is that patients will inevitably “go online and see that [the conditions are] associated with lots of bad things; it can really cause patients psychosocial harm if there is no one to explain what their risk is and also provide risk-specific management,” Dr. Weeks said at the annual meeting of the Society of Hematologic Oncology in Houston.
CHIP and CCUS are precursors of myeloid malignancies but for most patients, the risk of progression is less than 1%. CHIPS and CCUS are also associated with cardiovascular, rheumatologic, hepatic, and other diseases.
CHIP is defined by somatic mutations in myeloid malignancy driver genes with a variant allele fraction of 2% or more; CCUS is when those molecular features are accompanied by an unexplained and persistent anemia, thrombocytopenia, or neutropenia.
A small 2017 study suggested that about a third of patients with otherwise unexplained cytopenias have CCUS.
With the increasing use of next generation sequencing for tissue and liquid biopsies and other uses, the incidental diagnosis of both conditions is increasing.
Fortunately, Dr. Weeks’ group recently published a tool for predicting the risk of progression to myeloid malignancy.
Their “clonal hematopoiesis risk score” (CHRS) was developed and validated in over 400,000 healthy volunteers in the UK Biobank, with additional validation in cohorts from Dana Farber and the University of Pavia, Italy.
The CHRS incorporates eight high-risk genetic and clinical prognostic factors, including the type and number of genetic mutations in blood cells, factors related to red blood cell volume, and age over 65. It’s available online.
“You just input the patient’s information and it spits out if the patient is low, intermediate, or high risk for progression to any myeloid malignancy,” Dr. Weeks told her audience.
High-risk patients have about a 50% 10-year cumulative incidence of myeloid malignancy. The large majority of patients are low risk, however, and have a 10-year cumulative incidence of less than 1%. Patients in the middle have a 10-year risk of about 8%.
The low-risk group “is the population of people who probably don’t need to see a specialist,” and can be followed with an annual CBC with their primary care doctors plus further workup with any clinical change. Patients should also be evaluated for cardiovascular and other comorbidity risks.
“It’s the high-risk group we worry most about,” Dr. Weeks said. “We see them more often and repeat the next-generation sequencing” annually with a CBC at least every 6 months and a bone marrow biopsy with any clinical change.
“This is the population we would shuttle towards a clinical trial, as this is the population most likely to benefit,” she said.
The overarching goal of the several ongoing studies in CHIP/CCUS is to find a way to prevent progression to blood cancer. They range from prospective cohorts and single arm pilot studies to randomized clinical trials. One trial is evaluating canakinumab to prevent progression. “Intervention in clonal hematopoiesis might have the dual benefit of both preventing hematologic malignancy as well as reducing [the] inflammatory comorbidities,” Dr. Weeks said.
The reason is that patients will inevitably “go online and see that [the conditions are] associated with lots of bad things; it can really cause patients psychosocial harm if there is no one to explain what their risk is and also provide risk-specific management,” Dr. Weeks said at the annual meeting of the Society of Hematologic Oncology in Houston.
CHIP and CCUS are precursors of myeloid malignancies but for most patients, the risk of progression is less than 1%. CHIPS and CCUS are also associated with cardiovascular, rheumatologic, hepatic, and other diseases.
CHIP is defined by somatic mutations in myeloid malignancy driver genes with a variant allele fraction of 2% or more; CCUS is when those molecular features are accompanied by an unexplained and persistent anemia, thrombocytopenia, or neutropenia.
A small 2017 study suggested that about a third of patients with otherwise unexplained cytopenias have CCUS.
With the increasing use of next generation sequencing for tissue and liquid biopsies and other uses, the incidental diagnosis of both conditions is increasing.
Fortunately, Dr. Weeks’ group recently published a tool for predicting the risk of progression to myeloid malignancy.
Their “clonal hematopoiesis risk score” (CHRS) was developed and validated in over 400,000 healthy volunteers in the UK Biobank, with additional validation in cohorts from Dana Farber and the University of Pavia, Italy.
The CHRS incorporates eight high-risk genetic and clinical prognostic factors, including the type and number of genetic mutations in blood cells, factors related to red blood cell volume, and age over 65. It’s available online.
“You just input the patient’s information and it spits out if the patient is low, intermediate, or high risk for progression to any myeloid malignancy,” Dr. Weeks told her audience.
High-risk patients have about a 50% 10-year cumulative incidence of myeloid malignancy. The large majority of patients are low risk, however, and have a 10-year cumulative incidence of less than 1%. Patients in the middle have a 10-year risk of about 8%.
The low-risk group “is the population of people who probably don’t need to see a specialist,” and can be followed with an annual CBC with their primary care doctors plus further workup with any clinical change. Patients should also be evaluated for cardiovascular and other comorbidity risks.
“It’s the high-risk group we worry most about,” Dr. Weeks said. “We see them more often and repeat the next-generation sequencing” annually with a CBC at least every 6 months and a bone marrow biopsy with any clinical change.
“This is the population we would shuttle towards a clinical trial, as this is the population most likely to benefit,” she said.
The overarching goal of the several ongoing studies in CHIP/CCUS is to find a way to prevent progression to blood cancer. They range from prospective cohorts and single arm pilot studies to randomized clinical trials. One trial is evaluating canakinumab to prevent progression. “Intervention in clonal hematopoiesis might have the dual benefit of both preventing hematologic malignancy as well as reducing [the] inflammatory comorbidities,” Dr. Weeks said.
FROM SOHO 2023