User login
For MD-IQ use only
TIL for Melanoma: What Are the Costs and Other Challenges to Getting It to Patients?
The US Food and Drug Administration (FDA) recently approved the tumor-infiltrating lymphocyte cell therapy (TIL) for use in certain adults with unresectable or metastatic melanoma. This marks the first time the FDA has allowed a cellular therapy to be marketed for a solid tumor cancer.
Lifileucel is made from a patient’s surgically removed tumor. Tissue from that tumor is then sent to a manufacturing center. Turnaround time to when the drug is ready to be sent back to the cancer center for use is approximately 34 days, according to the drug’s manufacturer, Iovance.
Insurance Adjustments
The cost of the one-time lifileucel treatment is $515,000, according to the manufacturer.
Two investigators in the clinical trials of lifileucel, Allison Betof Warner, MD, of Stanford University, Stanford, California, and Igor Puzanov, MD, of Roswell Park Comprehensive Cancer Center, Buffalo, New York, shared their expectations regarding factors that would contribute to how much a patient paid for the drug.
Given the drug’s recent approval, the logistical details are still being worked out between cancer centers and insurers regarding how much patients will pay out of pocket for lifileucel, said Dr. Betof Warner, who is assistant professor in the Department of Medicine, Division of Medical Oncology at Stanford University.
The associated costs, including the surgery that is needed to procure the TIL cells for expansion into the final drug product, will be different for each patient, she told this publication.
Patients’ costs for lifileucel will vary based on their insurance, explained Dr. Puzanov, chief of melanoma and professor of oncology at Roswell Park Comprehensive Cancer Center.
At Roswell Park, “we will work with our regionally-based payers on a case-by-case basis to seek approval for those patients we believe can most benefit from lifileucel,” he said in an interview. Preauthorization will be required, as is standard for many cancer treatments, he added.
Once payer approval is in place, Dr. Puzanov said, he did not anticipate significant delays in access for patients.
Certified centers such as the multidisciplinary team at Roswell Park are ready to treat patients now. Other centers are similarly prepared, especially those involved in the clinical trials of lifileucel, he said.
Logistics and Infrastructure
A position article and guidelines on the management of and best practices for TIL was published in the Journal for ImmunoTherapy of Cancer on February 29. The paper, of which both Dr. Betof Warner and Dr. Puzanov served as authors, noted that one of the barriers to the use of TIL cell therapy in clinical practice is the need for state-of-the art infrastructure at centers that want to offer the treatment. Scheduling, patient referrals, and surgery, as well as the production and infusion of TIL, must be organized and streamlined for successful treatment, the authors wrote.
The two supply chains involved in TIL — the transportation of the tumor tissue from the treatment center to the manufacturer and transport of the TIL infusion product back to the treatment center — must be timely and precise, they emphasized.
Docs Hope TIL Improves in Several Ways
Although the TIL technology is a breakthrough, “we hope to see even better efficacy and lower toxicity as further research looks at ways to improve on the current TIL standard,” Dr. Puzanov said.
More research and dose adjustments may impact patient costs and side effects, he noted. “I am looking to see TILs used in the front line, with or without checkpoint inhibitors.”
Research is needed to explore how to lower the chemotherapy doses and possibly the associated toxicity, he added. Finally, researchers must consider whether high-dose IL-2 therapy — given as part of the TIL cell therapy — could be replaced with other cytokines, or whether the number of doses could be lowered. Another avenue of exploration is engineering genes for cytokines into TILs, he said.
“The key is to think about TIL therapy before you need it — ideally, when the patient is still doing well on their frontline checkpoint inhibition immunotherapy,” Dr. Puzanov said in an interview. That is the time for evaluation, and specialty centers can provide an expert assessment, he said.
“We are constantly working to improve TIL therapy,” Dr. Betof Warner told this publication. More research is needed optimize the regimen to reduce side effects, which would not only make treatment easier for currently eligible patients, but might allow treatment for patients not currently eligible.
“For example, we are looking for ways to reduce the dose of preparative chemotherapy, which prepares the body for the cells to maximize their longevity and efficacy, and to reduce or eliminate the need to give IL-2 after the cell administration,” continued Dr. Betof Warner, who is also Director of Melanoma Medical Oncology, Director of Solid Tumor Cellular Therapy, and Codirector of the Pigmented Lesion and Melanoma Program at Stanford University. “We are also actively studying next-generation TIL therapies to try to increase the efficacy.”
“Lifileucel has about a 30% success rate for melanoma that has progressed after standard therapy; we are working hard to do better than that,” she noted.
In a press release, Iovance summarized the results of the trial that supported the FDA’s accelerated approval of lifileucel. In an open-label single-arm study, including multiple sites worldwide, 73 adults with unresectable or metastatic melanoma who had received at least one previous systemic therapy underwent a lymphodepleting regimen followed by treatments with fludarabine and aldesleukin. Patients then received lifileucel at a median dose of 21.1 x 109 viable cells; the recommended dose ranges from 7.5 x 109 to 72 x 109 cells.
The primary efficacy outcome was objective response rate (ORR). The ORR in the study was 31.5%, and the median time to initial lifileucel response was 1.5 months.
The clinical trials of lifileucel for which Dr. Betof Warner and Dr. Puzanov served as investigators were sponsored by Iovance.
The US Food and Drug Administration (FDA) recently approved the tumor-infiltrating lymphocyte cell therapy (TIL) for use in certain adults with unresectable or metastatic melanoma. This marks the first time the FDA has allowed a cellular therapy to be marketed for a solid tumor cancer.
Lifileucel is made from a patient’s surgically removed tumor. Tissue from that tumor is then sent to a manufacturing center. Turnaround time to when the drug is ready to be sent back to the cancer center for use is approximately 34 days, according to the drug’s manufacturer, Iovance.
Insurance Adjustments
The cost of the one-time lifileucel treatment is $515,000, according to the manufacturer.
Two investigators in the clinical trials of lifileucel, Allison Betof Warner, MD, of Stanford University, Stanford, California, and Igor Puzanov, MD, of Roswell Park Comprehensive Cancer Center, Buffalo, New York, shared their expectations regarding factors that would contribute to how much a patient paid for the drug.
Given the drug’s recent approval, the logistical details are still being worked out between cancer centers and insurers regarding how much patients will pay out of pocket for lifileucel, said Dr. Betof Warner, who is assistant professor in the Department of Medicine, Division of Medical Oncology at Stanford University.
The associated costs, including the surgery that is needed to procure the TIL cells for expansion into the final drug product, will be different for each patient, she told this publication.
Patients’ costs for lifileucel will vary based on their insurance, explained Dr. Puzanov, chief of melanoma and professor of oncology at Roswell Park Comprehensive Cancer Center.
At Roswell Park, “we will work with our regionally-based payers on a case-by-case basis to seek approval for those patients we believe can most benefit from lifileucel,” he said in an interview. Preauthorization will be required, as is standard for many cancer treatments, he added.
Once payer approval is in place, Dr. Puzanov said, he did not anticipate significant delays in access for patients.
Certified centers such as the multidisciplinary team at Roswell Park are ready to treat patients now. Other centers are similarly prepared, especially those involved in the clinical trials of lifileucel, he said.
Logistics and Infrastructure
A position article and guidelines on the management of and best practices for TIL was published in the Journal for ImmunoTherapy of Cancer on February 29. The paper, of which both Dr. Betof Warner and Dr. Puzanov served as authors, noted that one of the barriers to the use of TIL cell therapy in clinical practice is the need for state-of-the art infrastructure at centers that want to offer the treatment. Scheduling, patient referrals, and surgery, as well as the production and infusion of TIL, must be organized and streamlined for successful treatment, the authors wrote.
The two supply chains involved in TIL — the transportation of the tumor tissue from the treatment center to the manufacturer and transport of the TIL infusion product back to the treatment center — must be timely and precise, they emphasized.
Docs Hope TIL Improves in Several Ways
Although the TIL technology is a breakthrough, “we hope to see even better efficacy and lower toxicity as further research looks at ways to improve on the current TIL standard,” Dr. Puzanov said.
More research and dose adjustments may impact patient costs and side effects, he noted. “I am looking to see TILs used in the front line, with or without checkpoint inhibitors.”
Research is needed to explore how to lower the chemotherapy doses and possibly the associated toxicity, he added. Finally, researchers must consider whether high-dose IL-2 therapy — given as part of the TIL cell therapy — could be replaced with other cytokines, or whether the number of doses could be lowered. Another avenue of exploration is engineering genes for cytokines into TILs, he said.
“The key is to think about TIL therapy before you need it — ideally, when the patient is still doing well on their frontline checkpoint inhibition immunotherapy,” Dr. Puzanov said in an interview. That is the time for evaluation, and specialty centers can provide an expert assessment, he said.
“We are constantly working to improve TIL therapy,” Dr. Betof Warner told this publication. More research is needed optimize the regimen to reduce side effects, which would not only make treatment easier for currently eligible patients, but might allow treatment for patients not currently eligible.
“For example, we are looking for ways to reduce the dose of preparative chemotherapy, which prepares the body for the cells to maximize their longevity and efficacy, and to reduce or eliminate the need to give IL-2 after the cell administration,” continued Dr. Betof Warner, who is also Director of Melanoma Medical Oncology, Director of Solid Tumor Cellular Therapy, and Codirector of the Pigmented Lesion and Melanoma Program at Stanford University. “We are also actively studying next-generation TIL therapies to try to increase the efficacy.”
“Lifileucel has about a 30% success rate for melanoma that has progressed after standard therapy; we are working hard to do better than that,” she noted.
In a press release, Iovance summarized the results of the trial that supported the FDA’s accelerated approval of lifileucel. In an open-label single-arm study, including multiple sites worldwide, 73 adults with unresectable or metastatic melanoma who had received at least one previous systemic therapy underwent a lymphodepleting regimen followed by treatments with fludarabine and aldesleukin. Patients then received lifileucel at a median dose of 21.1 x 109 viable cells; the recommended dose ranges from 7.5 x 109 to 72 x 109 cells.
The primary efficacy outcome was objective response rate (ORR). The ORR in the study was 31.5%, and the median time to initial lifileucel response was 1.5 months.
The clinical trials of lifileucel for which Dr. Betof Warner and Dr. Puzanov served as investigators were sponsored by Iovance.
The US Food and Drug Administration (FDA) recently approved the tumor-infiltrating lymphocyte cell therapy (TIL) for use in certain adults with unresectable or metastatic melanoma. This marks the first time the FDA has allowed a cellular therapy to be marketed for a solid tumor cancer.
Lifileucel is made from a patient’s surgically removed tumor. Tissue from that tumor is then sent to a manufacturing center. Turnaround time to when the drug is ready to be sent back to the cancer center for use is approximately 34 days, according to the drug’s manufacturer, Iovance.
Insurance Adjustments
The cost of the one-time lifileucel treatment is $515,000, according to the manufacturer.
Two investigators in the clinical trials of lifileucel, Allison Betof Warner, MD, of Stanford University, Stanford, California, and Igor Puzanov, MD, of Roswell Park Comprehensive Cancer Center, Buffalo, New York, shared their expectations regarding factors that would contribute to how much a patient paid for the drug.
Given the drug’s recent approval, the logistical details are still being worked out between cancer centers and insurers regarding how much patients will pay out of pocket for lifileucel, said Dr. Betof Warner, who is assistant professor in the Department of Medicine, Division of Medical Oncology at Stanford University.
The associated costs, including the surgery that is needed to procure the TIL cells for expansion into the final drug product, will be different for each patient, she told this publication.
Patients’ costs for lifileucel will vary based on their insurance, explained Dr. Puzanov, chief of melanoma and professor of oncology at Roswell Park Comprehensive Cancer Center.
At Roswell Park, “we will work with our regionally-based payers on a case-by-case basis to seek approval for those patients we believe can most benefit from lifileucel,” he said in an interview. Preauthorization will be required, as is standard for many cancer treatments, he added.
Once payer approval is in place, Dr. Puzanov said, he did not anticipate significant delays in access for patients.
Certified centers such as the multidisciplinary team at Roswell Park are ready to treat patients now. Other centers are similarly prepared, especially those involved in the clinical trials of lifileucel, he said.
Logistics and Infrastructure
A position article and guidelines on the management of and best practices for TIL was published in the Journal for ImmunoTherapy of Cancer on February 29. The paper, of which both Dr. Betof Warner and Dr. Puzanov served as authors, noted that one of the barriers to the use of TIL cell therapy in clinical practice is the need for state-of-the art infrastructure at centers that want to offer the treatment. Scheduling, patient referrals, and surgery, as well as the production and infusion of TIL, must be organized and streamlined for successful treatment, the authors wrote.
The two supply chains involved in TIL — the transportation of the tumor tissue from the treatment center to the manufacturer and transport of the TIL infusion product back to the treatment center — must be timely and precise, they emphasized.
Docs Hope TIL Improves in Several Ways
Although the TIL technology is a breakthrough, “we hope to see even better efficacy and lower toxicity as further research looks at ways to improve on the current TIL standard,” Dr. Puzanov said.
More research and dose adjustments may impact patient costs and side effects, he noted. “I am looking to see TILs used in the front line, with or without checkpoint inhibitors.”
Research is needed to explore how to lower the chemotherapy doses and possibly the associated toxicity, he added. Finally, researchers must consider whether high-dose IL-2 therapy — given as part of the TIL cell therapy — could be replaced with other cytokines, or whether the number of doses could be lowered. Another avenue of exploration is engineering genes for cytokines into TILs, he said.
“The key is to think about TIL therapy before you need it — ideally, when the patient is still doing well on their frontline checkpoint inhibition immunotherapy,” Dr. Puzanov said in an interview. That is the time for evaluation, and specialty centers can provide an expert assessment, he said.
“We are constantly working to improve TIL therapy,” Dr. Betof Warner told this publication. More research is needed optimize the regimen to reduce side effects, which would not only make treatment easier for currently eligible patients, but might allow treatment for patients not currently eligible.
“For example, we are looking for ways to reduce the dose of preparative chemotherapy, which prepares the body for the cells to maximize their longevity and efficacy, and to reduce or eliminate the need to give IL-2 after the cell administration,” continued Dr. Betof Warner, who is also Director of Melanoma Medical Oncology, Director of Solid Tumor Cellular Therapy, and Codirector of the Pigmented Lesion and Melanoma Program at Stanford University. “We are also actively studying next-generation TIL therapies to try to increase the efficacy.”
“Lifileucel has about a 30% success rate for melanoma that has progressed after standard therapy; we are working hard to do better than that,” she noted.
In a press release, Iovance summarized the results of the trial that supported the FDA’s accelerated approval of lifileucel. In an open-label single-arm study, including multiple sites worldwide, 73 adults with unresectable or metastatic melanoma who had received at least one previous systemic therapy underwent a lymphodepleting regimen followed by treatments with fludarabine and aldesleukin. Patients then received lifileucel at a median dose of 21.1 x 109 viable cells; the recommended dose ranges from 7.5 x 109 to 72 x 109 cells.
The primary efficacy outcome was objective response rate (ORR). The ORR in the study was 31.5%, and the median time to initial lifileucel response was 1.5 months.
The clinical trials of lifileucel for which Dr. Betof Warner and Dr. Puzanov served as investigators were sponsored by Iovance.
Study Finds No Increased Cancer Risk With Spironolactone
TOPLINE:
than that of unexposed women.
METHODOLOGY:
- Spironolactone, used off-label for several skin conditions in women, carries a warning about an increased tumor risk associated with high doses in rat models, and its antiandrogen properties have prompted hypotheses about a possible increased risk for breast or gynecologic cancers.
- The researchers reviewed data on 420 women with a history of spironolactone use for acne, hair loss, and hirsutism and 3272 women with no spironolactone use at the authors› institution. Their mean age ranged from 42 to 63 years; the majority were White, and 38% were non-White.
- Median spironolactone doses ranged from 25 mg to 225 mg; chart reviews included 5-year follow-up data from the first spironolactone exposure to allow time for tumor development.
TAKEAWAY:
- A total of 37 of the 420 women exposed to spironolactone developed any tumors, as did 546 of the 3272 with no spironolactone exposure.
- After the researchers controlled for age and race, women exposed to spironolactone were no more likely to develop a malignant tumor than a benign tumor, compared with unexposed women (odds ratio [OR], 0.48, P = .2).
- The risk for breast or uterine cancer was not significantly different in the spironolactone and non-spironolactone groups (OR, 0.95, P > .9).
IN PRACTICE:
“Women taking spironolactone for acne, hair loss, and hirsutism and who are at low risk of breast or gynecologic cancers may be counseled to have regular gynecology follow-up, but no more frequently than the general population,” but more studies are needed to evaluate risk over longer periods of time, the researchers wrote.
SOURCE:
The lead author of the study was Rachel C. Hill, BS, a student at Weill Cornell Medical College, New York City, and Shari R. Lipner, MD, PhD, of the department of dermatology at Weill Cornell Medical College, was the corresponding author. The study was published online in The Journal of the American Academy of Dermatology.
LIMITATIONS:
The findings were limited by the retrospective design, as well as the small number of spironolactone patients analyzed, the short follow-up period, the lack of information about spironolactone courses, and the inability to control for family history of malignancy.
DISCLOSURES:
The study was supported by the National Center for Advancing Translational Sciences and a grant from the Clinical and Translational Science Center at Weill Cornell Medical College awarded to Ms. Hill. None of the authors had relevant disclosures; Dr. Lipner disclosed serving as a consultant for Ortho-Dermatologics, Eli Lilly, Moberg Pharmaceuticals, and BelleTorus Corporation.
A version of this article appeared on Medscape.com.
TOPLINE:
than that of unexposed women.
METHODOLOGY:
- Spironolactone, used off-label for several skin conditions in women, carries a warning about an increased tumor risk associated with high doses in rat models, and its antiandrogen properties have prompted hypotheses about a possible increased risk for breast or gynecologic cancers.
- The researchers reviewed data on 420 women with a history of spironolactone use for acne, hair loss, and hirsutism and 3272 women with no spironolactone use at the authors› institution. Their mean age ranged from 42 to 63 years; the majority were White, and 38% were non-White.
- Median spironolactone doses ranged from 25 mg to 225 mg; chart reviews included 5-year follow-up data from the first spironolactone exposure to allow time for tumor development.
TAKEAWAY:
- A total of 37 of the 420 women exposed to spironolactone developed any tumors, as did 546 of the 3272 with no spironolactone exposure.
- After the researchers controlled for age and race, women exposed to spironolactone were no more likely to develop a malignant tumor than a benign tumor, compared with unexposed women (odds ratio [OR], 0.48, P = .2).
- The risk for breast or uterine cancer was not significantly different in the spironolactone and non-spironolactone groups (OR, 0.95, P > .9).
IN PRACTICE:
“Women taking spironolactone for acne, hair loss, and hirsutism and who are at low risk of breast or gynecologic cancers may be counseled to have regular gynecology follow-up, but no more frequently than the general population,” but more studies are needed to evaluate risk over longer periods of time, the researchers wrote.
SOURCE:
The lead author of the study was Rachel C. Hill, BS, a student at Weill Cornell Medical College, New York City, and Shari R. Lipner, MD, PhD, of the department of dermatology at Weill Cornell Medical College, was the corresponding author. The study was published online in The Journal of the American Academy of Dermatology.
LIMITATIONS:
The findings were limited by the retrospective design, as well as the small number of spironolactone patients analyzed, the short follow-up period, the lack of information about spironolactone courses, and the inability to control for family history of malignancy.
DISCLOSURES:
The study was supported by the National Center for Advancing Translational Sciences and a grant from the Clinical and Translational Science Center at Weill Cornell Medical College awarded to Ms. Hill. None of the authors had relevant disclosures; Dr. Lipner disclosed serving as a consultant for Ortho-Dermatologics, Eli Lilly, Moberg Pharmaceuticals, and BelleTorus Corporation.
A version of this article appeared on Medscape.com.
TOPLINE:
than that of unexposed women.
METHODOLOGY:
- Spironolactone, used off-label for several skin conditions in women, carries a warning about an increased tumor risk associated with high doses in rat models, and its antiandrogen properties have prompted hypotheses about a possible increased risk for breast or gynecologic cancers.
- The researchers reviewed data on 420 women with a history of spironolactone use for acne, hair loss, and hirsutism and 3272 women with no spironolactone use at the authors› institution. Their mean age ranged from 42 to 63 years; the majority were White, and 38% were non-White.
- Median spironolactone doses ranged from 25 mg to 225 mg; chart reviews included 5-year follow-up data from the first spironolactone exposure to allow time for tumor development.
TAKEAWAY:
- A total of 37 of the 420 women exposed to spironolactone developed any tumors, as did 546 of the 3272 with no spironolactone exposure.
- After the researchers controlled for age and race, women exposed to spironolactone were no more likely to develop a malignant tumor than a benign tumor, compared with unexposed women (odds ratio [OR], 0.48, P = .2).
- The risk for breast or uterine cancer was not significantly different in the spironolactone and non-spironolactone groups (OR, 0.95, P > .9).
IN PRACTICE:
“Women taking spironolactone for acne, hair loss, and hirsutism and who are at low risk of breast or gynecologic cancers may be counseled to have regular gynecology follow-up, but no more frequently than the general population,” but more studies are needed to evaluate risk over longer periods of time, the researchers wrote.
SOURCE:
The lead author of the study was Rachel C. Hill, BS, a student at Weill Cornell Medical College, New York City, and Shari R. Lipner, MD, PhD, of the department of dermatology at Weill Cornell Medical College, was the corresponding author. The study was published online in The Journal of the American Academy of Dermatology.
LIMITATIONS:
The findings were limited by the retrospective design, as well as the small number of spironolactone patients analyzed, the short follow-up period, the lack of information about spironolactone courses, and the inability to control for family history of malignancy.
DISCLOSURES:
The study was supported by the National Center for Advancing Translational Sciences and a grant from the Clinical and Translational Science Center at Weill Cornell Medical College awarded to Ms. Hill. None of the authors had relevant disclosures; Dr. Lipner disclosed serving as a consultant for Ortho-Dermatologics, Eli Lilly, Moberg Pharmaceuticals, and BelleTorus Corporation.
A version of this article appeared on Medscape.com.
No Increase in Autoimmune Risk Seen With GLP-1 Receptor Agonists and SGLT2 Inhibitors
TOPLINE:
In patients with type 2 diabetes, there was no difference in risk of developing autoimmune disease if prescribed glucagon-like peptide 1 receptor agonists (GLP-1-RAs), sodium-glucose cotransporter-2 (SGLT2) inhibitors, or dipeptidyl peptidase-4 (DPP-4) inhibitors.
METHODOLOGY:
- The effect of GLP-1-RAs and SGLT2 inhibitors on autoimmune rheumatic disease (ARD) is understudied, though previous case reports and one study have hinted at increased risk.
- Researchers used administrative health data from 2014 to 2021 to identify 34,400 patients prescribed GLP-1-RAs and 83,500 patients prescribed SGLT2 inhibitors.
- They compared patients prescribed GLP-1-RAs or SGLT2 inhibitors with 68,400 patients prescribed DPP-4 inhibitors, which previous studies suggest do not increase ARD risk.
- Primary outcome was ARD incidence, defined by diagnostic codes.
TAKEAWAY:
- There were no significant differences in incident ARDs between the three groups.
- Mean follow-up time was 0.88-1.53 years.
- The hazard ratio (HR) for developing ARDs with GLP-1-RAs exposure was 0.93 (95% CI, 0.66-1.30) compared with DPP-4 inhibitors.
- The HR for developing ARDs with SGLT2 inhibitor exposure was 0.97 (95% CI, 0.76-1.24).
IN PRACTICE:
“Extended longitudinal data are needed to assess risk and benefit with longer-term exposure,” the authors wrote.
SOURCE:
First author Derin Karacabeyli, MD, of the University of British Columbia, Vancouver, Canada, presented the study in abstract form at the Canadian Rheumatology Association (CRA) 2024 Annual Meeting in Winnipeg on February 29.
LIMITATIONS:
The study was observational, which could have some residual or unmeasured confounding of data. The researchers relied on diagnostic codes and the average follow-up time was short.
DISCLOSURES:
The study was funded by the Canadian Institutes of Health Research. The authors had no disclosures.
A version of this article appeared on Medscape.com.
TOPLINE:
In patients with type 2 diabetes, there was no difference in risk of developing autoimmune disease if prescribed glucagon-like peptide 1 receptor agonists (GLP-1-RAs), sodium-glucose cotransporter-2 (SGLT2) inhibitors, or dipeptidyl peptidase-4 (DPP-4) inhibitors.
METHODOLOGY:
- The effect of GLP-1-RAs and SGLT2 inhibitors on autoimmune rheumatic disease (ARD) is understudied, though previous case reports and one study have hinted at increased risk.
- Researchers used administrative health data from 2014 to 2021 to identify 34,400 patients prescribed GLP-1-RAs and 83,500 patients prescribed SGLT2 inhibitors.
- They compared patients prescribed GLP-1-RAs or SGLT2 inhibitors with 68,400 patients prescribed DPP-4 inhibitors, which previous studies suggest do not increase ARD risk.
- Primary outcome was ARD incidence, defined by diagnostic codes.
TAKEAWAY:
- There were no significant differences in incident ARDs between the three groups.
- Mean follow-up time was 0.88-1.53 years.
- The hazard ratio (HR) for developing ARDs with GLP-1-RAs exposure was 0.93 (95% CI, 0.66-1.30) compared with DPP-4 inhibitors.
- The HR for developing ARDs with SGLT2 inhibitor exposure was 0.97 (95% CI, 0.76-1.24).
IN PRACTICE:
“Extended longitudinal data are needed to assess risk and benefit with longer-term exposure,” the authors wrote.
SOURCE:
First author Derin Karacabeyli, MD, of the University of British Columbia, Vancouver, Canada, presented the study in abstract form at the Canadian Rheumatology Association (CRA) 2024 Annual Meeting in Winnipeg on February 29.
LIMITATIONS:
The study was observational, which could have some residual or unmeasured confounding of data. The researchers relied on diagnostic codes and the average follow-up time was short.
DISCLOSURES:
The study was funded by the Canadian Institutes of Health Research. The authors had no disclosures.
A version of this article appeared on Medscape.com.
TOPLINE:
In patients with type 2 diabetes, there was no difference in risk of developing autoimmune disease if prescribed glucagon-like peptide 1 receptor agonists (GLP-1-RAs), sodium-glucose cotransporter-2 (SGLT2) inhibitors, or dipeptidyl peptidase-4 (DPP-4) inhibitors.
METHODOLOGY:
- The effect of GLP-1-RAs and SGLT2 inhibitors on autoimmune rheumatic disease (ARD) is understudied, though previous case reports and one study have hinted at increased risk.
- Researchers used administrative health data from 2014 to 2021 to identify 34,400 patients prescribed GLP-1-RAs and 83,500 patients prescribed SGLT2 inhibitors.
- They compared patients prescribed GLP-1-RAs or SGLT2 inhibitors with 68,400 patients prescribed DPP-4 inhibitors, which previous studies suggest do not increase ARD risk.
- Primary outcome was ARD incidence, defined by diagnostic codes.
TAKEAWAY:
- There were no significant differences in incident ARDs between the three groups.
- Mean follow-up time was 0.88-1.53 years.
- The hazard ratio (HR) for developing ARDs with GLP-1-RAs exposure was 0.93 (95% CI, 0.66-1.30) compared with DPP-4 inhibitors.
- The HR for developing ARDs with SGLT2 inhibitor exposure was 0.97 (95% CI, 0.76-1.24).
IN PRACTICE:
“Extended longitudinal data are needed to assess risk and benefit with longer-term exposure,” the authors wrote.
SOURCE:
First author Derin Karacabeyli, MD, of the University of British Columbia, Vancouver, Canada, presented the study in abstract form at the Canadian Rheumatology Association (CRA) 2024 Annual Meeting in Winnipeg on February 29.
LIMITATIONS:
The study was observational, which could have some residual or unmeasured confounding of data. The researchers relied on diagnostic codes and the average follow-up time was short.
DISCLOSURES:
The study was funded by the Canadian Institutes of Health Research. The authors had no disclosures.
A version of this article appeared on Medscape.com.
Vulvar Lichen Sclerosus: What’s New?
Vulvar lichen sclerosus (VLS) is an underserved area in medicine and dermatology. We discuss updates in VLS, which include the following: (1) development of core outcome domains to include in all future clinical trials, with current efforts focused on determining outcome measurements for each domain; (2) increased understanding of the impact VLS has on quality-of-life (QOL) outcomes; (3) expanded disease associations; (4) clinical and histologic variants, including vestibular sclerosis and nonsclerotic VLS; and (5) updates in management of VLS.
Core Outcomes Measures
The burden of VLS is challenging to quantify, with little agreement among experts.1 Recently there has been a focus on developing scoring scales to measure disease progression and treatment response. Simpson et al2 pioneered the development of a core outcome set to be included in all future clinical trials for genital lichen sclerosus (LS)—clinical (visible) signs, symptoms, and LS-specific QOL.
Although there is no standardized method for assessing disease severity, various scales have been proposed to measure clinical findings in VLS, such as the vulvar architecture severity scale3 as well as the clinical LS score,4 which is the only validated scale to incorporate the signs and architectural changes identified by a 2018 Delphi consensus group of the International Society for the Study of Vulvovaginal Disease.5 Work is ongoing to identify and evaluate outcome measurement instruments for each of the 3 core outcome domains.
Increased Understanding of QOL Impacts
Pain, pruritus, impairment of sexual function, genitourinary complications, architectural changes, and risk for squamous cell carcinoma (SCC) all have been well established as VLS sequelae.6,7 Recent studies have focused on the QOL impact and associations with psychiatric comorbidities. A matched case-control study found that LS was significantly associated with depression and anxiety among US women (P<.001), and individuals with LS had a more than 2-fold increased odds of receiving a diagnosis of depression or anxiety.8
A review evaluating QOL outcomes in LS found that overall QOL was impaired. Female patients reported worse QOL in the work-school domain of the dermatology life quality index compared with male counterparts.9
Finally, a study exploring the experiences of patients living with VLS highlighted the secrecy and stigma of the condition,10 which serves as a call to action to improve the general population’s knowledge about vulvar anatomy and create change in societal attitudes on vulvar conditions.
Although there are several instruments assessing vulvar-specific QOL, most are for patients with vulvar cancer and focus on sexual function. In 2020, Saunderson et al11 published the 15-item vulvar quality of life index (VQLI), which has broad implications for measuring vulvar disease burden and is an important tool for standardizing vulvar disease measurements and outcomes for clinical research.12 The VQLI, though not specific to VLS, consists of 4 domains to assess vulvar QOL including symptoms, anxiety, activities of daily living, and sexuality. Studies have evaluated this scoring system in patients with VLS, with 1 study finding that VQLI correlated with clinician-rated severity scores (P=.01) and overall patient itch/discomfort score (P<.001) in VLS.13,14
Expanded Disease Associations
Lichen sclerosus has a well-known association with vulvar SCC and other autoimmune conditions, including thyroid disease and bullous pemphigoid.15-17 Recent studies also have revealed an association between LS and psoriasis.18 A case-control study from a single center found VLS was associated with elevated body mass index, statin usage, and cholecystectomy.19 Gynecologic pain syndromes, interstitial cystitis, urinary incontinence, and some gastrointestinal tract disorders including celiac disease also have been found to be increased in patients with VLS.20 Finally, the incidence of cutaneous immune-related adverse events such as LS has increased as the use of immune checkpoint therapies as anticancer treatments has expanded.21 Clinicians should be aware of these potential disease associations when caring for patients with VLS.
The incidence of VLS is higher in lower estrogen states throughout the lifespan, and a recent case-control study evaluated the cutaneous hormonal and microbial landscapes in postmenopausal patients (6 patients with VLS; 12 controls).22 Levels of the following cutaneous hormones in the groin were found to be altered in patients with VLS compared with controls: estrone (lower; P=.006), progesterone (higher; P<.0001), and testosterone (lower; P=.02). The authors found that most hormone levels normalized following treatment with a topical steroid. Additionally, bacterial microbiome alterations were seen in patients with VLS compared with controls. Thus, cutaneous sex hormone and skin microbiome alterations may be associated with VLS.22
Updates in Clinical and Histologic Variants
Less-recognized variants of VLS have been characterized in recent years. Vestibular sclerosis is a variant of VLS with unique clinical and histopathologic features; it is characterized by involvement localized to the anterior vestibule and either an absent or sparse lymphocytic infiltrate on histopathology.23,24 Nonsclerotic VLS is a variant with clinical features consistent with VLS that does not exhibit dermal sclerosis on histopathology. Thus, a diagnosis of nonsclerotic VLS requires clinicopathologic correlation. Four nonsclerotic histopathologic subtypes are proposed: lichenoid, hypertrophic lichenoid, dermal fibrosis without acanthosis, and dermal fibrosis with acanthosis.25 Longitudinal studies that correlate duration, signs, and symptoms will be important to further understand these variants.
Management Updates
First-line treatment of VLS still consists of ultrapotent topical corticosteroids with chronic maintenance therapy (usually lifetime) to decrease the risk for SCC and architectural changes.26 However, a survey across social media platforms found steroid phobia is common in patients with VLS (N=865), with approximately 40% of respondents endorsing waiting as long as they could before using topical corticosteroids and stopping as soon as possible.27 Clinicians should be aware of possible patient perceptions in the use of chronic steroids when discussing this therapy.
Randomized controlled trials utilizing fractional CO2 devices for VLS have been performed with conflicting results and no consensus regarding outcome measurement.28,29 Additionally, long-term disease outcomes following laser use have not been investigated. Although there is evidence that both ablative and nonablative devices can improve symptoms and signs, there is no evidence that they offer a cure for a chronic inflammatory skin condition. Current evidence suggests that even for patients undergoing these procedures, maintenance therapy is still essential to prevent sequelae.30 Future studies incorporating standardized outcome measures will be important for assessing the benefits of laser therapy in VLS. Finally, the reasons why topical corticosteroids may fail in an individual patient are multifaceted and should be explored thoroughly when considering laser therapy for VLS.
Studies evaluating the role of systemic therapies for refractory cases of VLS have expanded. A systematic review of systemic therapies for both genital and extragenital LS found oral corticosteroids and methotrexate were the most-reported systemic treatment regimens.31 Use of biologics in LS has been reported, with cases utilizing adalimumab for VLS and dupilumab for extragenital LS. Use of Janus kinase inhibitors including abrocitinib and baricitinib also has been reported for LS.31 A clinical trial to evaluate the safety and efficacy of topical ruxolitinib in VLS was recently completed (ClinicalTrials.govidentifier NCT05593445). Future research studies likely will focus on the safety and efficacy of targeted and steroid-sparing therapies for patients with VLS.
Final Thoughts
Vulvar lichen sclerosus increasingly is becoming recognized as a chronic genital skin condition that impacts QOL and health outcomes, with a need to develop more effective and safe evidence-based therapies. Recent literature has focused on the importance of developing and standardizing disease outcomes; identifying disease associations including the role of cutaneous hormones and microbiome alterations; characterizing histologic and clinical variants; and staying up-to-date on management, including the need for understanding patient perceptions of chronic topical steroid therapy. Each of these are important updates for clinicians to consider when caring for patients with VLS. Future studies likely will focus on elucidating disease etiology and mechanisms to gain a better understanding of VLS pathogenesis and potential targets for therapies as well as implementation of clinical trials that incorporate standardized outcome domains to test efficacy and safety of additional therapies.
- Sheinis M, Green N, Vieira-Baptista P, et al. Adult vulvar lichen sclerosus: can experts agree on the assessment of disease severity? J Low Genit Tract Dis. 2020;24:295-298. doi:10.1097/LGT.0000000000000534
- Simpson RC, Kirtschig G, Selk A, et al. Core outcome domains for lichen sclerosus: a CORALS initiative consensus statement. Br J Dermatol. 2023;188:628-635. doi:10.1093/bjd/ljac145
- Almadori A, Zenner N, Boyle D, et al. Development and validation of a clinical grading scale to assess the vulvar region: the Vulvar Architecture Severity Scale. Aesthet Surg J. 2020;40:1319-1326. doi:10.1093/asj/sjz342
- Erni B, Navarini AA, Huang D, et al. Proposition of a severity scale for lichen sclerosus: the “Clinical Lichen Sclerosus Score.” Dermatol Ther. 2021;34:E14773. doi:10.1111/dth.14773
- Sheinis M, Selk A. Development of the Adult Vulvar Lichen Sclerosus Severity Scale—a Delphi Consensus Exercise for Item Generation. J Low Genit Tract Dis. 2018;22:66-73. doi:10.1097/LGT.0000000000000361
- Mauskar MM, Marathe K, Venkatesan A, et al. Vulvar diseases. J Am Acad Dermatol. 2020;82:1287-1298. doi:10.1016/j.jaad.2019.10.077
- Wijaya M, Lee G, Fischer G. Why do some patients with vulval lichen sclerosus on long-term topical corticosteroid treatment experience ongoing poor quality of life? Australas J Dermatol. 2022;63:463-472. doi:10.1111/ajd.13926
- Fan R, Leasure AC, Maisha FI, et al. Depression and anxiety in patients with lichen sclerosus. JAMA Dermatol. 2022;158:953-954. doi:10.1001/jamadermatol.2022.1964
- Ranum A, Pearson DR. The impact of genital lichen sclerosus and lichen planus on quality of life: a review. Int J Womens Dermatol. 2022;8:E042. doi:10.1097/JW9.0000000000000042
- Arnold S, Fernando S, Rees S. Living with vulval lichen sclerosus: a qualitative interview study. Br J Dermatol. 2022;187:909-918. doi:10.1111/bjd.21777
- Saunderson RB, Harris V, Yeh R, et al. Vulvar quality of life index (VQLI)—a simple tool to measure quality of life in patients with vulvar disease. Australas J Dermatol. 2020;61:152-157. doi:10.1111/ajd.13235
- Pyle HJ, Evans JC, Vandergriff TW, et al. Vulvar lichen sclerosus clinical severity scales and histopathologic correlation: a case series. Am J Dermatopathol. 2023;45:588-592. doi:10.1097/DAD.0000000000002471
- Wijaya M, Lee G, Fischer G. Quality of life of women with untreated vulval lichen sclerosus assessed with vulval quality of life index (VQLI) [published online January 28, 2021]. Australas J Dermatol. 2021;62:177-182. doi:10.1111/ajd.13530
- Felmingham C, Chan L, Doyle LW, et al. The Vulval Disease Quality of Life Index in women with vulval lichen sclerosus correlates with clinician and symptom scores [published online November 14, 2019]. Australas J Dermatol. 2020;61:110-118. doi:10.1111/ajd.13197
- Walsh ML, Leonard N, Shawki H, et al. Lichen sclerosus and immunobullous disease. J Low Genit Tract Dis. 2012;16:468-470. doi:10.1097/LGT.0b013e31825e9b18
- Chin S, Scurry J, Bradford J, et al. Association of topical corticosteroids with reduced vulvar squamous cell carcinoma recurrence in patients with vulvar lichen sclerosus. JAMA Dermatol. 2020;156:813. doi:10.1001/jamadermatol.2020.1074
- Fan R, Leasure AC, Maisha FI, et al. Thyroid disorders associated with lichen sclerosus: a case–control study in the All of Us Research Program. Br J Dermatol. 2022;187:797-799. doi:10.1111/bjd.21702
- Fan R, Leasure AC, Little AJ, et al. Lichen sclerosus among women with psoriasis: a cross-sectional study in the All of Us research program. J Am Acad Dermatol. 2023;88:1175-1177. doi:10.1016/j.jaad.2022.12.012
- Luu Y, Cheng AL, Reisz C. Elevated body mass index, statin use, and cholecystectomy are associated with vulvar lichen sclerosus: a retrospective, case-control study. J Am Acad Dermatol. 2023;88:1376-1378. doi:10.1016/j.jaad.2023.01.023
- Söderlund JM, Hieta NK, Kurki SH, et al. Comorbidity of urogynecological and gastrointestinal disorders in female patients with lichen sclerosus. J Low Genit Tract Dis. 2023;2:156-160. doi:10.1097/LGT.0000000000000727
- Shin L, Smith J, Shiu J, et al. Association of lichen sclerosus and morphea with immune checkpoint therapy: a systematic review. Int J Womens Dermatol. 2023;9:E070. doi:10.1097/JW9.0000000000000070
- Pyle HJ, Evans JC, Artami M, et al. Assessment of the cutaneous hormone landscapes and microbiomes in vulvar lichen sclerosus [published online February 16, 2024]. J Invest Dermatol. 2024:S0022-202X(24)00111-8. doi:10.1016/j.jid.2024.01.027
- Day T, Burston K, Dennerstein G, et al. Vestibulovaginal sclerosis versus lichen sclerosus. Int J Gynecol Pathol. 2018;37:356-363. doi:10.1097/PGP.0000000000000441
- Croker BA, Scurry JP, Petry FM, et al. Vestibular sclerosis: is this a new, distinct clinicopathological entity? J Low Genit Tract Dis. 2018;22:260-263. doi:10.1097/LGT.0000000000000404
- Day T, Selim MA, Allbritton JI, et al. Nonsclerotic lichen sclerosus: definition of a concept and pathologic description. J Low Genit Tract Dis. 2023;27:358-364. doi:10.1097/LGT.0000000000000760
- Lee A, Bradford J, Fischer G. Long-term management of adult vulvar lichen sclerosus: a prospective cohort study of 507 women. JAMA Dermatol. 2015;151:1061. doi:10.1001/jamadermatol.2015.0643
- Delpero E, Sriharan A, Selk A. Steroid phobia in patients with vulvar lichen sclerosus. J Low Genit Tract Dis. 2023;27:286-290. doi:10.1097/LGT.0000000000000753
- Burkett LS, Siddique M, Zeymo A, et al. Clobetasol compared with fractionated carbon dioxide laser for lichen sclerosus: a randomized controlled trial. Obstet Gynecol. 2021;137:968-978. doi:10.1097/AOG.0000000000004332
- Mitchell L, Goldstein AT, Heller D, et al. Fractionated carbon dioxide laser for the treatment of vulvar lichen sclerosus: a randomized controlled trial. Obstet Gynecol. 2021;137:979-987. doi:10.1097/AOG.0000000000004409
- Li HOY, Bailey AMJ, Tan MG, Dover JS. Lasers as an adjuvant for vulvar lichen sclerosus: a systematic review and meta-analysis. J Am Acad Dermatol. 2022;86:694-696. doi:10.1016/j.jaad.2021.02.081
- Hargis A, Ngo M, Kraus CN, et al. Systemic therapy for lichen sclerosus: a systematic review [published online November 4, 2023]. J Low Genit Tract Dis. doi:10.1097/LGT.0000000000000775
Vulvar lichen sclerosus (VLS) is an underserved area in medicine and dermatology. We discuss updates in VLS, which include the following: (1) development of core outcome domains to include in all future clinical trials, with current efforts focused on determining outcome measurements for each domain; (2) increased understanding of the impact VLS has on quality-of-life (QOL) outcomes; (3) expanded disease associations; (4) clinical and histologic variants, including vestibular sclerosis and nonsclerotic VLS; and (5) updates in management of VLS.
Core Outcomes Measures
The burden of VLS is challenging to quantify, with little agreement among experts.1 Recently there has been a focus on developing scoring scales to measure disease progression and treatment response. Simpson et al2 pioneered the development of a core outcome set to be included in all future clinical trials for genital lichen sclerosus (LS)—clinical (visible) signs, symptoms, and LS-specific QOL.
Although there is no standardized method for assessing disease severity, various scales have been proposed to measure clinical findings in VLS, such as the vulvar architecture severity scale3 as well as the clinical LS score,4 which is the only validated scale to incorporate the signs and architectural changes identified by a 2018 Delphi consensus group of the International Society for the Study of Vulvovaginal Disease.5 Work is ongoing to identify and evaluate outcome measurement instruments for each of the 3 core outcome domains.
Increased Understanding of QOL Impacts
Pain, pruritus, impairment of sexual function, genitourinary complications, architectural changes, and risk for squamous cell carcinoma (SCC) all have been well established as VLS sequelae.6,7 Recent studies have focused on the QOL impact and associations with psychiatric comorbidities. A matched case-control study found that LS was significantly associated with depression and anxiety among US women (P<.001), and individuals with LS had a more than 2-fold increased odds of receiving a diagnosis of depression or anxiety.8
A review evaluating QOL outcomes in LS found that overall QOL was impaired. Female patients reported worse QOL in the work-school domain of the dermatology life quality index compared with male counterparts.9
Finally, a study exploring the experiences of patients living with VLS highlighted the secrecy and stigma of the condition,10 which serves as a call to action to improve the general population’s knowledge about vulvar anatomy and create change in societal attitudes on vulvar conditions.
Although there are several instruments assessing vulvar-specific QOL, most are for patients with vulvar cancer and focus on sexual function. In 2020, Saunderson et al11 published the 15-item vulvar quality of life index (VQLI), which has broad implications for measuring vulvar disease burden and is an important tool for standardizing vulvar disease measurements and outcomes for clinical research.12 The VQLI, though not specific to VLS, consists of 4 domains to assess vulvar QOL including symptoms, anxiety, activities of daily living, and sexuality. Studies have evaluated this scoring system in patients with VLS, with 1 study finding that VQLI correlated with clinician-rated severity scores (P=.01) and overall patient itch/discomfort score (P<.001) in VLS.13,14
Expanded Disease Associations
Lichen sclerosus has a well-known association with vulvar SCC and other autoimmune conditions, including thyroid disease and bullous pemphigoid.15-17 Recent studies also have revealed an association between LS and psoriasis.18 A case-control study from a single center found VLS was associated with elevated body mass index, statin usage, and cholecystectomy.19 Gynecologic pain syndromes, interstitial cystitis, urinary incontinence, and some gastrointestinal tract disorders including celiac disease also have been found to be increased in patients with VLS.20 Finally, the incidence of cutaneous immune-related adverse events such as LS has increased as the use of immune checkpoint therapies as anticancer treatments has expanded.21 Clinicians should be aware of these potential disease associations when caring for patients with VLS.
The incidence of VLS is higher in lower estrogen states throughout the lifespan, and a recent case-control study evaluated the cutaneous hormonal and microbial landscapes in postmenopausal patients (6 patients with VLS; 12 controls).22 Levels of the following cutaneous hormones in the groin were found to be altered in patients with VLS compared with controls: estrone (lower; P=.006), progesterone (higher; P<.0001), and testosterone (lower; P=.02). The authors found that most hormone levels normalized following treatment with a topical steroid. Additionally, bacterial microbiome alterations were seen in patients with VLS compared with controls. Thus, cutaneous sex hormone and skin microbiome alterations may be associated with VLS.22
Updates in Clinical and Histologic Variants
Less-recognized variants of VLS have been characterized in recent years. Vestibular sclerosis is a variant of VLS with unique clinical and histopathologic features; it is characterized by involvement localized to the anterior vestibule and either an absent or sparse lymphocytic infiltrate on histopathology.23,24 Nonsclerotic VLS is a variant with clinical features consistent with VLS that does not exhibit dermal sclerosis on histopathology. Thus, a diagnosis of nonsclerotic VLS requires clinicopathologic correlation. Four nonsclerotic histopathologic subtypes are proposed: lichenoid, hypertrophic lichenoid, dermal fibrosis without acanthosis, and dermal fibrosis with acanthosis.25 Longitudinal studies that correlate duration, signs, and symptoms will be important to further understand these variants.
Management Updates
First-line treatment of VLS still consists of ultrapotent topical corticosteroids with chronic maintenance therapy (usually lifetime) to decrease the risk for SCC and architectural changes.26 However, a survey across social media platforms found steroid phobia is common in patients with VLS (N=865), with approximately 40% of respondents endorsing waiting as long as they could before using topical corticosteroids and stopping as soon as possible.27 Clinicians should be aware of possible patient perceptions in the use of chronic steroids when discussing this therapy.
Randomized controlled trials utilizing fractional CO2 devices for VLS have been performed with conflicting results and no consensus regarding outcome measurement.28,29 Additionally, long-term disease outcomes following laser use have not been investigated. Although there is evidence that both ablative and nonablative devices can improve symptoms and signs, there is no evidence that they offer a cure for a chronic inflammatory skin condition. Current evidence suggests that even for patients undergoing these procedures, maintenance therapy is still essential to prevent sequelae.30 Future studies incorporating standardized outcome measures will be important for assessing the benefits of laser therapy in VLS. Finally, the reasons why topical corticosteroids may fail in an individual patient are multifaceted and should be explored thoroughly when considering laser therapy for VLS.
Studies evaluating the role of systemic therapies for refractory cases of VLS have expanded. A systematic review of systemic therapies for both genital and extragenital LS found oral corticosteroids and methotrexate were the most-reported systemic treatment regimens.31 Use of biologics in LS has been reported, with cases utilizing adalimumab for VLS and dupilumab for extragenital LS. Use of Janus kinase inhibitors including abrocitinib and baricitinib also has been reported for LS.31 A clinical trial to evaluate the safety and efficacy of topical ruxolitinib in VLS was recently completed (ClinicalTrials.govidentifier NCT05593445). Future research studies likely will focus on the safety and efficacy of targeted and steroid-sparing therapies for patients with VLS.
Final Thoughts
Vulvar lichen sclerosus increasingly is becoming recognized as a chronic genital skin condition that impacts QOL and health outcomes, with a need to develop more effective and safe evidence-based therapies. Recent literature has focused on the importance of developing and standardizing disease outcomes; identifying disease associations including the role of cutaneous hormones and microbiome alterations; characterizing histologic and clinical variants; and staying up-to-date on management, including the need for understanding patient perceptions of chronic topical steroid therapy. Each of these are important updates for clinicians to consider when caring for patients with VLS. Future studies likely will focus on elucidating disease etiology and mechanisms to gain a better understanding of VLS pathogenesis and potential targets for therapies as well as implementation of clinical trials that incorporate standardized outcome domains to test efficacy and safety of additional therapies.
Vulvar lichen sclerosus (VLS) is an underserved area in medicine and dermatology. We discuss updates in VLS, which include the following: (1) development of core outcome domains to include in all future clinical trials, with current efforts focused on determining outcome measurements for each domain; (2) increased understanding of the impact VLS has on quality-of-life (QOL) outcomes; (3) expanded disease associations; (4) clinical and histologic variants, including vestibular sclerosis and nonsclerotic VLS; and (5) updates in management of VLS.
Core Outcomes Measures
The burden of VLS is challenging to quantify, with little agreement among experts.1 Recently there has been a focus on developing scoring scales to measure disease progression and treatment response. Simpson et al2 pioneered the development of a core outcome set to be included in all future clinical trials for genital lichen sclerosus (LS)—clinical (visible) signs, symptoms, and LS-specific QOL.
Although there is no standardized method for assessing disease severity, various scales have been proposed to measure clinical findings in VLS, such as the vulvar architecture severity scale3 as well as the clinical LS score,4 which is the only validated scale to incorporate the signs and architectural changes identified by a 2018 Delphi consensus group of the International Society for the Study of Vulvovaginal Disease.5 Work is ongoing to identify and evaluate outcome measurement instruments for each of the 3 core outcome domains.
Increased Understanding of QOL Impacts
Pain, pruritus, impairment of sexual function, genitourinary complications, architectural changes, and risk for squamous cell carcinoma (SCC) all have been well established as VLS sequelae.6,7 Recent studies have focused on the QOL impact and associations with psychiatric comorbidities. A matched case-control study found that LS was significantly associated with depression and anxiety among US women (P<.001), and individuals with LS had a more than 2-fold increased odds of receiving a diagnosis of depression or anxiety.8
A review evaluating QOL outcomes in LS found that overall QOL was impaired. Female patients reported worse QOL in the work-school domain of the dermatology life quality index compared with male counterparts.9
Finally, a study exploring the experiences of patients living with VLS highlighted the secrecy and stigma of the condition,10 which serves as a call to action to improve the general population’s knowledge about vulvar anatomy and create change in societal attitudes on vulvar conditions.
Although there are several instruments assessing vulvar-specific QOL, most are for patients with vulvar cancer and focus on sexual function. In 2020, Saunderson et al11 published the 15-item vulvar quality of life index (VQLI), which has broad implications for measuring vulvar disease burden and is an important tool for standardizing vulvar disease measurements and outcomes for clinical research.12 The VQLI, though not specific to VLS, consists of 4 domains to assess vulvar QOL including symptoms, anxiety, activities of daily living, and sexuality. Studies have evaluated this scoring system in patients with VLS, with 1 study finding that VQLI correlated with clinician-rated severity scores (P=.01) and overall patient itch/discomfort score (P<.001) in VLS.13,14
Expanded Disease Associations
Lichen sclerosus has a well-known association with vulvar SCC and other autoimmune conditions, including thyroid disease and bullous pemphigoid.15-17 Recent studies also have revealed an association between LS and psoriasis.18 A case-control study from a single center found VLS was associated with elevated body mass index, statin usage, and cholecystectomy.19 Gynecologic pain syndromes, interstitial cystitis, urinary incontinence, and some gastrointestinal tract disorders including celiac disease also have been found to be increased in patients with VLS.20 Finally, the incidence of cutaneous immune-related adverse events such as LS has increased as the use of immune checkpoint therapies as anticancer treatments has expanded.21 Clinicians should be aware of these potential disease associations when caring for patients with VLS.
The incidence of VLS is higher in lower estrogen states throughout the lifespan, and a recent case-control study evaluated the cutaneous hormonal and microbial landscapes in postmenopausal patients (6 patients with VLS; 12 controls).22 Levels of the following cutaneous hormones in the groin were found to be altered in patients with VLS compared with controls: estrone (lower; P=.006), progesterone (higher; P<.0001), and testosterone (lower; P=.02). The authors found that most hormone levels normalized following treatment with a topical steroid. Additionally, bacterial microbiome alterations were seen in patients with VLS compared with controls. Thus, cutaneous sex hormone and skin microbiome alterations may be associated with VLS.22
Updates in Clinical and Histologic Variants
Less-recognized variants of VLS have been characterized in recent years. Vestibular sclerosis is a variant of VLS with unique clinical and histopathologic features; it is characterized by involvement localized to the anterior vestibule and either an absent or sparse lymphocytic infiltrate on histopathology.23,24 Nonsclerotic VLS is a variant with clinical features consistent with VLS that does not exhibit dermal sclerosis on histopathology. Thus, a diagnosis of nonsclerotic VLS requires clinicopathologic correlation. Four nonsclerotic histopathologic subtypes are proposed: lichenoid, hypertrophic lichenoid, dermal fibrosis without acanthosis, and dermal fibrosis with acanthosis.25 Longitudinal studies that correlate duration, signs, and symptoms will be important to further understand these variants.
Management Updates
First-line treatment of VLS still consists of ultrapotent topical corticosteroids with chronic maintenance therapy (usually lifetime) to decrease the risk for SCC and architectural changes.26 However, a survey across social media platforms found steroid phobia is common in patients with VLS (N=865), with approximately 40% of respondents endorsing waiting as long as they could before using topical corticosteroids and stopping as soon as possible.27 Clinicians should be aware of possible patient perceptions in the use of chronic steroids when discussing this therapy.
Randomized controlled trials utilizing fractional CO2 devices for VLS have been performed with conflicting results and no consensus regarding outcome measurement.28,29 Additionally, long-term disease outcomes following laser use have not been investigated. Although there is evidence that both ablative and nonablative devices can improve symptoms and signs, there is no evidence that they offer a cure for a chronic inflammatory skin condition. Current evidence suggests that even for patients undergoing these procedures, maintenance therapy is still essential to prevent sequelae.30 Future studies incorporating standardized outcome measures will be important for assessing the benefits of laser therapy in VLS. Finally, the reasons why topical corticosteroids may fail in an individual patient are multifaceted and should be explored thoroughly when considering laser therapy for VLS.
Studies evaluating the role of systemic therapies for refractory cases of VLS have expanded. A systematic review of systemic therapies for both genital and extragenital LS found oral corticosteroids and methotrexate were the most-reported systemic treatment regimens.31 Use of biologics in LS has been reported, with cases utilizing adalimumab for VLS and dupilumab for extragenital LS. Use of Janus kinase inhibitors including abrocitinib and baricitinib also has been reported for LS.31 A clinical trial to evaluate the safety and efficacy of topical ruxolitinib in VLS was recently completed (ClinicalTrials.govidentifier NCT05593445). Future research studies likely will focus on the safety and efficacy of targeted and steroid-sparing therapies for patients with VLS.
Final Thoughts
Vulvar lichen sclerosus increasingly is becoming recognized as a chronic genital skin condition that impacts QOL and health outcomes, with a need to develop more effective and safe evidence-based therapies. Recent literature has focused on the importance of developing and standardizing disease outcomes; identifying disease associations including the role of cutaneous hormones and microbiome alterations; characterizing histologic and clinical variants; and staying up-to-date on management, including the need for understanding patient perceptions of chronic topical steroid therapy. Each of these are important updates for clinicians to consider when caring for patients with VLS. Future studies likely will focus on elucidating disease etiology and mechanisms to gain a better understanding of VLS pathogenesis and potential targets for therapies as well as implementation of clinical trials that incorporate standardized outcome domains to test efficacy and safety of additional therapies.
- Sheinis M, Green N, Vieira-Baptista P, et al. Adult vulvar lichen sclerosus: can experts agree on the assessment of disease severity? J Low Genit Tract Dis. 2020;24:295-298. doi:10.1097/LGT.0000000000000534
- Simpson RC, Kirtschig G, Selk A, et al. Core outcome domains for lichen sclerosus: a CORALS initiative consensus statement. Br J Dermatol. 2023;188:628-635. doi:10.1093/bjd/ljac145
- Almadori A, Zenner N, Boyle D, et al. Development and validation of a clinical grading scale to assess the vulvar region: the Vulvar Architecture Severity Scale. Aesthet Surg J. 2020;40:1319-1326. doi:10.1093/asj/sjz342
- Erni B, Navarini AA, Huang D, et al. Proposition of a severity scale for lichen sclerosus: the “Clinical Lichen Sclerosus Score.” Dermatol Ther. 2021;34:E14773. doi:10.1111/dth.14773
- Sheinis M, Selk A. Development of the Adult Vulvar Lichen Sclerosus Severity Scale—a Delphi Consensus Exercise for Item Generation. J Low Genit Tract Dis. 2018;22:66-73. doi:10.1097/LGT.0000000000000361
- Mauskar MM, Marathe K, Venkatesan A, et al. Vulvar diseases. J Am Acad Dermatol. 2020;82:1287-1298. doi:10.1016/j.jaad.2019.10.077
- Wijaya M, Lee G, Fischer G. Why do some patients with vulval lichen sclerosus on long-term topical corticosteroid treatment experience ongoing poor quality of life? Australas J Dermatol. 2022;63:463-472. doi:10.1111/ajd.13926
- Fan R, Leasure AC, Maisha FI, et al. Depression and anxiety in patients with lichen sclerosus. JAMA Dermatol. 2022;158:953-954. doi:10.1001/jamadermatol.2022.1964
- Ranum A, Pearson DR. The impact of genital lichen sclerosus and lichen planus on quality of life: a review. Int J Womens Dermatol. 2022;8:E042. doi:10.1097/JW9.0000000000000042
- Arnold S, Fernando S, Rees S. Living with vulval lichen sclerosus: a qualitative interview study. Br J Dermatol. 2022;187:909-918. doi:10.1111/bjd.21777
- Saunderson RB, Harris V, Yeh R, et al. Vulvar quality of life index (VQLI)—a simple tool to measure quality of life in patients with vulvar disease. Australas J Dermatol. 2020;61:152-157. doi:10.1111/ajd.13235
- Pyle HJ, Evans JC, Vandergriff TW, et al. Vulvar lichen sclerosus clinical severity scales and histopathologic correlation: a case series. Am J Dermatopathol. 2023;45:588-592. doi:10.1097/DAD.0000000000002471
- Wijaya M, Lee G, Fischer G. Quality of life of women with untreated vulval lichen sclerosus assessed with vulval quality of life index (VQLI) [published online January 28, 2021]. Australas J Dermatol. 2021;62:177-182. doi:10.1111/ajd.13530
- Felmingham C, Chan L, Doyle LW, et al. The Vulval Disease Quality of Life Index in women with vulval lichen sclerosus correlates with clinician and symptom scores [published online November 14, 2019]. Australas J Dermatol. 2020;61:110-118. doi:10.1111/ajd.13197
- Walsh ML, Leonard N, Shawki H, et al. Lichen sclerosus and immunobullous disease. J Low Genit Tract Dis. 2012;16:468-470. doi:10.1097/LGT.0b013e31825e9b18
- Chin S, Scurry J, Bradford J, et al. Association of topical corticosteroids with reduced vulvar squamous cell carcinoma recurrence in patients with vulvar lichen sclerosus. JAMA Dermatol. 2020;156:813. doi:10.1001/jamadermatol.2020.1074
- Fan R, Leasure AC, Maisha FI, et al. Thyroid disorders associated with lichen sclerosus: a case–control study in the All of Us Research Program. Br J Dermatol. 2022;187:797-799. doi:10.1111/bjd.21702
- Fan R, Leasure AC, Little AJ, et al. Lichen sclerosus among women with psoriasis: a cross-sectional study in the All of Us research program. J Am Acad Dermatol. 2023;88:1175-1177. doi:10.1016/j.jaad.2022.12.012
- Luu Y, Cheng AL, Reisz C. Elevated body mass index, statin use, and cholecystectomy are associated with vulvar lichen sclerosus: a retrospective, case-control study. J Am Acad Dermatol. 2023;88:1376-1378. doi:10.1016/j.jaad.2023.01.023
- Söderlund JM, Hieta NK, Kurki SH, et al. Comorbidity of urogynecological and gastrointestinal disorders in female patients with lichen sclerosus. J Low Genit Tract Dis. 2023;2:156-160. doi:10.1097/LGT.0000000000000727
- Shin L, Smith J, Shiu J, et al. Association of lichen sclerosus and morphea with immune checkpoint therapy: a systematic review. Int J Womens Dermatol. 2023;9:E070. doi:10.1097/JW9.0000000000000070
- Pyle HJ, Evans JC, Artami M, et al. Assessment of the cutaneous hormone landscapes and microbiomes in vulvar lichen sclerosus [published online February 16, 2024]. J Invest Dermatol. 2024:S0022-202X(24)00111-8. doi:10.1016/j.jid.2024.01.027
- Day T, Burston K, Dennerstein G, et al. Vestibulovaginal sclerosis versus lichen sclerosus. Int J Gynecol Pathol. 2018;37:356-363. doi:10.1097/PGP.0000000000000441
- Croker BA, Scurry JP, Petry FM, et al. Vestibular sclerosis: is this a new, distinct clinicopathological entity? J Low Genit Tract Dis. 2018;22:260-263. doi:10.1097/LGT.0000000000000404
- Day T, Selim MA, Allbritton JI, et al. Nonsclerotic lichen sclerosus: definition of a concept and pathologic description. J Low Genit Tract Dis. 2023;27:358-364. doi:10.1097/LGT.0000000000000760
- Lee A, Bradford J, Fischer G. Long-term management of adult vulvar lichen sclerosus: a prospective cohort study of 507 women. JAMA Dermatol. 2015;151:1061. doi:10.1001/jamadermatol.2015.0643
- Delpero E, Sriharan A, Selk A. Steroid phobia in patients with vulvar lichen sclerosus. J Low Genit Tract Dis. 2023;27:286-290. doi:10.1097/LGT.0000000000000753
- Burkett LS, Siddique M, Zeymo A, et al. Clobetasol compared with fractionated carbon dioxide laser for lichen sclerosus: a randomized controlled trial. Obstet Gynecol. 2021;137:968-978. doi:10.1097/AOG.0000000000004332
- Mitchell L, Goldstein AT, Heller D, et al. Fractionated carbon dioxide laser for the treatment of vulvar lichen sclerosus: a randomized controlled trial. Obstet Gynecol. 2021;137:979-987. doi:10.1097/AOG.0000000000004409
- Li HOY, Bailey AMJ, Tan MG, Dover JS. Lasers as an adjuvant for vulvar lichen sclerosus: a systematic review and meta-analysis. J Am Acad Dermatol. 2022;86:694-696. doi:10.1016/j.jaad.2021.02.081
- Hargis A, Ngo M, Kraus CN, et al. Systemic therapy for lichen sclerosus: a systematic review [published online November 4, 2023]. J Low Genit Tract Dis. doi:10.1097/LGT.0000000000000775
- Sheinis M, Green N, Vieira-Baptista P, et al. Adult vulvar lichen sclerosus: can experts agree on the assessment of disease severity? J Low Genit Tract Dis. 2020;24:295-298. doi:10.1097/LGT.0000000000000534
- Simpson RC, Kirtschig G, Selk A, et al. Core outcome domains for lichen sclerosus: a CORALS initiative consensus statement. Br J Dermatol. 2023;188:628-635. doi:10.1093/bjd/ljac145
- Almadori A, Zenner N, Boyle D, et al. Development and validation of a clinical grading scale to assess the vulvar region: the Vulvar Architecture Severity Scale. Aesthet Surg J. 2020;40:1319-1326. doi:10.1093/asj/sjz342
- Erni B, Navarini AA, Huang D, et al. Proposition of a severity scale for lichen sclerosus: the “Clinical Lichen Sclerosus Score.” Dermatol Ther. 2021;34:E14773. doi:10.1111/dth.14773
- Sheinis M, Selk A. Development of the Adult Vulvar Lichen Sclerosus Severity Scale—a Delphi Consensus Exercise for Item Generation. J Low Genit Tract Dis. 2018;22:66-73. doi:10.1097/LGT.0000000000000361
- Mauskar MM, Marathe K, Venkatesan A, et al. Vulvar diseases. J Am Acad Dermatol. 2020;82:1287-1298. doi:10.1016/j.jaad.2019.10.077
- Wijaya M, Lee G, Fischer G. Why do some patients with vulval lichen sclerosus on long-term topical corticosteroid treatment experience ongoing poor quality of life? Australas J Dermatol. 2022;63:463-472. doi:10.1111/ajd.13926
- Fan R, Leasure AC, Maisha FI, et al. Depression and anxiety in patients with lichen sclerosus. JAMA Dermatol. 2022;158:953-954. doi:10.1001/jamadermatol.2022.1964
- Ranum A, Pearson DR. The impact of genital lichen sclerosus and lichen planus on quality of life: a review. Int J Womens Dermatol. 2022;8:E042. doi:10.1097/JW9.0000000000000042
- Arnold S, Fernando S, Rees S. Living with vulval lichen sclerosus: a qualitative interview study. Br J Dermatol. 2022;187:909-918. doi:10.1111/bjd.21777
- Saunderson RB, Harris V, Yeh R, et al. Vulvar quality of life index (VQLI)—a simple tool to measure quality of life in patients with vulvar disease. Australas J Dermatol. 2020;61:152-157. doi:10.1111/ajd.13235
- Pyle HJ, Evans JC, Vandergriff TW, et al. Vulvar lichen sclerosus clinical severity scales and histopathologic correlation: a case series. Am J Dermatopathol. 2023;45:588-592. doi:10.1097/DAD.0000000000002471
- Wijaya M, Lee G, Fischer G. Quality of life of women with untreated vulval lichen sclerosus assessed with vulval quality of life index (VQLI) [published online January 28, 2021]. Australas J Dermatol. 2021;62:177-182. doi:10.1111/ajd.13530
- Felmingham C, Chan L, Doyle LW, et al. The Vulval Disease Quality of Life Index in women with vulval lichen sclerosus correlates with clinician and symptom scores [published online November 14, 2019]. Australas J Dermatol. 2020;61:110-118. doi:10.1111/ajd.13197
- Walsh ML, Leonard N, Shawki H, et al. Lichen sclerosus and immunobullous disease. J Low Genit Tract Dis. 2012;16:468-470. doi:10.1097/LGT.0b013e31825e9b18
- Chin S, Scurry J, Bradford J, et al. Association of topical corticosteroids with reduced vulvar squamous cell carcinoma recurrence in patients with vulvar lichen sclerosus. JAMA Dermatol. 2020;156:813. doi:10.1001/jamadermatol.2020.1074
- Fan R, Leasure AC, Maisha FI, et al. Thyroid disorders associated with lichen sclerosus: a case–control study in the All of Us Research Program. Br J Dermatol. 2022;187:797-799. doi:10.1111/bjd.21702
- Fan R, Leasure AC, Little AJ, et al. Lichen sclerosus among women with psoriasis: a cross-sectional study in the All of Us research program. J Am Acad Dermatol. 2023;88:1175-1177. doi:10.1016/j.jaad.2022.12.012
- Luu Y, Cheng AL, Reisz C. Elevated body mass index, statin use, and cholecystectomy are associated with vulvar lichen sclerosus: a retrospective, case-control study. J Am Acad Dermatol. 2023;88:1376-1378. doi:10.1016/j.jaad.2023.01.023
- Söderlund JM, Hieta NK, Kurki SH, et al. Comorbidity of urogynecological and gastrointestinal disorders in female patients with lichen sclerosus. J Low Genit Tract Dis. 2023;2:156-160. doi:10.1097/LGT.0000000000000727
- Shin L, Smith J, Shiu J, et al. Association of lichen sclerosus and morphea with immune checkpoint therapy: a systematic review. Int J Womens Dermatol. 2023;9:E070. doi:10.1097/JW9.0000000000000070
- Pyle HJ, Evans JC, Artami M, et al. Assessment of the cutaneous hormone landscapes and microbiomes in vulvar lichen sclerosus [published online February 16, 2024]. J Invest Dermatol. 2024:S0022-202X(24)00111-8. doi:10.1016/j.jid.2024.01.027
- Day T, Burston K, Dennerstein G, et al. Vestibulovaginal sclerosis versus lichen sclerosus. Int J Gynecol Pathol. 2018;37:356-363. doi:10.1097/PGP.0000000000000441
- Croker BA, Scurry JP, Petry FM, et al. Vestibular sclerosis: is this a new, distinct clinicopathological entity? J Low Genit Tract Dis. 2018;22:260-263. doi:10.1097/LGT.0000000000000404
- Day T, Selim MA, Allbritton JI, et al. Nonsclerotic lichen sclerosus: definition of a concept and pathologic description. J Low Genit Tract Dis. 2023;27:358-364. doi:10.1097/LGT.0000000000000760
- Lee A, Bradford J, Fischer G. Long-term management of adult vulvar lichen sclerosus: a prospective cohort study of 507 women. JAMA Dermatol. 2015;151:1061. doi:10.1001/jamadermatol.2015.0643
- Delpero E, Sriharan A, Selk A. Steroid phobia in patients with vulvar lichen sclerosus. J Low Genit Tract Dis. 2023;27:286-290. doi:10.1097/LGT.0000000000000753
- Burkett LS, Siddique M, Zeymo A, et al. Clobetasol compared with fractionated carbon dioxide laser for lichen sclerosus: a randomized controlled trial. Obstet Gynecol. 2021;137:968-978. doi:10.1097/AOG.0000000000004332
- Mitchell L, Goldstein AT, Heller D, et al. Fractionated carbon dioxide laser for the treatment of vulvar lichen sclerosus: a randomized controlled trial. Obstet Gynecol. 2021;137:979-987. doi:10.1097/AOG.0000000000004409
- Li HOY, Bailey AMJ, Tan MG, Dover JS. Lasers as an adjuvant for vulvar lichen sclerosus: a systematic review and meta-analysis. J Am Acad Dermatol. 2022;86:694-696. doi:10.1016/j.jaad.2021.02.081
- Hargis A, Ngo M, Kraus CN, et al. Systemic therapy for lichen sclerosus: a systematic review [published online November 4, 2023]. J Low Genit Tract Dis. doi:10.1097/LGT.0000000000000775
Brown Plaque in the Axilla Following Immobilization of the Arm
The Diagnosis: Granular Parakeratosis
Histopathology demonstrated diffuse parakeratosis with retention of keratohyalin granules throughout the stratum corneum consistent with a diagnosis of granular parakeratosis (Figure), a rare benign cutaneous condition that is thought to occur due to a defect in epidermal differentiation. The lesion resolved without additional treatment.
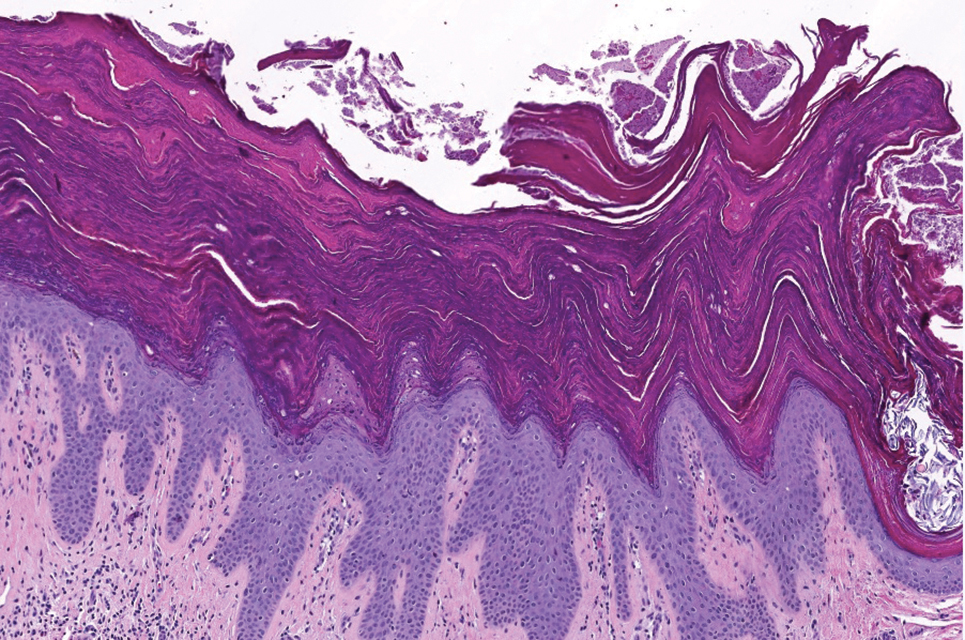
The pathogenesis of granular parakeratosis is unclear, but a reactive process in which locoregional irritation or occlusion prompts increased cell turnover and prevention of profilaggrin breakdown has been proposed.1,2 The diagnosis is linked to various precipitating agents, most commonly topical products (eg, zinc oxide, antiperspirants) and products with benzalkonium chloride (eg, laundry rinses). These agents are thought to cause retention of keratohyalin granules in the stratum corneum during epidermal differentiation.1,2
Most affected patients are middle-aged women (mean age at diagnosis, 37.8 years).2 Patients present with eruptions of erythematous, brown, hyperkeratotic patches and papules that coalesce into plaques.1,2 These lesions can be pruritic and painful or asymptomatic. They often manifest bilaterally in intertriginous sites, most commonly the axillae, groin, or inguinal folds.1,2
Treatment involves identification and removal of potential triggers including changing antiperspirants, limiting use of irritating agents (eg, topical products with strong fragrances), and reducing heat and moisture in the affected areas. If the lesion persists, stepwise treatment can be initiated with topical agents (eg, corticosteroids, vitamin D analogues, retinoids, keratolytics, calcineurin inhibitors) followed by systemic medications (eg, antibiotics, isotretinoin, antifungals, dexamethasone) and procedures (eg, botulinum toxin injections, surgery, laser, cryotherapy).1,2
Unilateral granular parakeratosis, as seen in our patient, is an uncommon manifestation. Our case supports the theory that occlusion is a precipitating factor for this condition, given persistent axillary exposure to heat, sweat, and friction in the setting of limb immobilization.3
Granular parakeratosis is a challenge to diagnose due to clinical overlap with several other cutaneous conditions; histopathologic confirmation is required. Fox- Fordyce disease is a rare condition that is thought to result from keratin buildup or occlusion of apocrine or apoeccrine sweat ducts leading to duct rupture and surrounding inflammation.4 Common triggers include laser hair removal, hormonal changes, and living conditions that promote hot and humid environments.5 It can manifest similarly to granular parakeratosis, with eruptions of multiple red-violet papules that appear bilaterally in aprocine gland–rich areas, including the axillae and less commonly the genital, periareolar, thoracic, abdominal, and facial areas.4,5 However, most patients with Fox-Fordyce disease tend to be younger females (aged 13–35 years) with severely pruritic lesions,4,5 unlike our patient. In addition, histopathology shows hyperkeratosis, hair follicle plugging, and sweat gland and duct dilation.4
Seborrheic keratoses are common benign epidermal tumors caused by an overproliferation of immature keratinocytes.6,7 Similar to granular parakeratosis, they commonly manifest in older adults as hyperpigmented, well-demarcated, verrucous plaques with a hyperkeratotic surface.6 However, they are more common on the face, neck, trunk, and extremities, and they tend to be asymptomatic, differentiating them from granular parakerosis.6 Histopathology demonstrates a papillomatous epidermal surface, large capillaries in the dermal papillae, and intraepidermal and pseudohorn epidermal cysts.7
Inverse lichen planus, a variant of lichen planus, is a rare inflammatory condition that involves the lysis of basal keratinocytes by CD8+ lymphocytes.8 Similar to granular parakeratosis, lichen planus commonly affects middle-aged women (aged 30–60 years), and this particular variant manifests with asymptomatic or mildly pruritic, hyperpigmented patches and plaques in intertriginous areas. Although it also shows hyperkeratosis on histopathology, it can be differentiated from granular parakeratosis by the additional findings of epidermal hypergranulosis, sawtooth acanthosis of rete ridges, apoptotic keratinocytes in the dermoepidermal junction, and lymphocytic infiltrate in the upper dermis.8
Hailey-Hailey disease (also known as familial benign pemphigus) is a rare condition caused by an autosomaldominant mutation affecting intracellular calcium signaling that impairs keratinocyte adhesion.9 Similar to granular parakeratosis, it is most common in middle-aged adults (aged 30–40 years) and manifests as pruritic and burning lesions in symmetric intertriginous areas that also can be triggered by heat and sweating. However, patients present with recurrent blistering and vesicular lesions that may lead to erosions and secondary infections, which reduced clinical suspicion for this diagnosis in our patient. Histopathology shows suprabasilar and intraepidermal clefts, full-thickness acantholysis, protruding dermal papillae, and a perivascular lymphocytic infiltrate in the superficial dermis.9
- Ding CY, Liu H, Khachemoune A. Granular parakeratosis: a comprehensive review and a critical reappraisal. Am J Clin Dermatol. 2015;16:495-500. doi:10.1007/s40257-015-0148-2
- Ip KH, Li A. Clinical features, histology, and treatment outcomes of granular parakeratosis: a systematic review. Int J Dermatol. 2022;61:973-978. doi:10.1111/ijd.16107
- Mehregan DA, Thomas JE, Mehregan DR. Intertriginous granular parakeratosis. J Am Acad Dermatol. 1998;39:495-496. doi:10.1016/s0190-9622(98)70333-0
- Kamada A, Saga K, Jimbow K. Apoeccrine sweat duct obstruction as a cause for Fox-Fordyce disease. J Am Acad Dermatol. 2003;48:453-455. doi:10.1067/mjd.2003.93
- Salloum A, Bouferraa Y, Bazzi N, et al. Pathophysiology, clinical findings, and management of Fox-Fordyce disease: a systematic review. J Cosmet Dermatol. 2022;21:482-500. doi:10.1111/jocd.14135
- Sun MD, Halpern AC. Advances in the etiology, detection, and clinical management of seborrheic keratoses. Dermatology. 2022;238:205-217. doi:10.1159/000517070
- Minagawa A. Dermoscopy-pathology relationship in seborrheic keratosis. J Dermatol. 2017;44:518-524. doi:10.1111/1346-8138.13657
- Weston G, Payette M. Update on lichen planus and its clinical variants [published online September 16, 2015]. Int J Womens Dermatol. 2015;1:140-149. doi:10.1016/j.ijwd.2015.04.001
- Ben Lagha I, Ashack K, Khachemoune A. Hailey-Hailey disease: an update review with a focus on treatment data. Am J Clin Dermatol. 2020;21:49-68. doi:10.1007/s40257-019-00477-z
The Diagnosis: Granular Parakeratosis
Histopathology demonstrated diffuse parakeratosis with retention of keratohyalin granules throughout the stratum corneum consistent with a diagnosis of granular parakeratosis (Figure), a rare benign cutaneous condition that is thought to occur due to a defect in epidermal differentiation. The lesion resolved without additional treatment.

The pathogenesis of granular parakeratosis is unclear, but a reactive process in which locoregional irritation or occlusion prompts increased cell turnover and prevention of profilaggrin breakdown has been proposed.1,2 The diagnosis is linked to various precipitating agents, most commonly topical products (eg, zinc oxide, antiperspirants) and products with benzalkonium chloride (eg, laundry rinses). These agents are thought to cause retention of keratohyalin granules in the stratum corneum during epidermal differentiation.1,2
Most affected patients are middle-aged women (mean age at diagnosis, 37.8 years).2 Patients present with eruptions of erythematous, brown, hyperkeratotic patches and papules that coalesce into plaques.1,2 These lesions can be pruritic and painful or asymptomatic. They often manifest bilaterally in intertriginous sites, most commonly the axillae, groin, or inguinal folds.1,2
Treatment involves identification and removal of potential triggers including changing antiperspirants, limiting use of irritating agents (eg, topical products with strong fragrances), and reducing heat and moisture in the affected areas. If the lesion persists, stepwise treatment can be initiated with topical agents (eg, corticosteroids, vitamin D analogues, retinoids, keratolytics, calcineurin inhibitors) followed by systemic medications (eg, antibiotics, isotretinoin, antifungals, dexamethasone) and procedures (eg, botulinum toxin injections, surgery, laser, cryotherapy).1,2
Unilateral granular parakeratosis, as seen in our patient, is an uncommon manifestation. Our case supports the theory that occlusion is a precipitating factor for this condition, given persistent axillary exposure to heat, sweat, and friction in the setting of limb immobilization.3
Granular parakeratosis is a challenge to diagnose due to clinical overlap with several other cutaneous conditions; histopathologic confirmation is required. Fox- Fordyce disease is a rare condition that is thought to result from keratin buildup or occlusion of apocrine or apoeccrine sweat ducts leading to duct rupture and surrounding inflammation.4 Common triggers include laser hair removal, hormonal changes, and living conditions that promote hot and humid environments.5 It can manifest similarly to granular parakeratosis, with eruptions of multiple red-violet papules that appear bilaterally in aprocine gland–rich areas, including the axillae and less commonly the genital, periareolar, thoracic, abdominal, and facial areas.4,5 However, most patients with Fox-Fordyce disease tend to be younger females (aged 13–35 years) with severely pruritic lesions,4,5 unlike our patient. In addition, histopathology shows hyperkeratosis, hair follicle plugging, and sweat gland and duct dilation.4
Seborrheic keratoses are common benign epidermal tumors caused by an overproliferation of immature keratinocytes.6,7 Similar to granular parakeratosis, they commonly manifest in older adults as hyperpigmented, well-demarcated, verrucous plaques with a hyperkeratotic surface.6 However, they are more common on the face, neck, trunk, and extremities, and they tend to be asymptomatic, differentiating them from granular parakerosis.6 Histopathology demonstrates a papillomatous epidermal surface, large capillaries in the dermal papillae, and intraepidermal and pseudohorn epidermal cysts.7
Inverse lichen planus, a variant of lichen planus, is a rare inflammatory condition that involves the lysis of basal keratinocytes by CD8+ lymphocytes.8 Similar to granular parakeratosis, lichen planus commonly affects middle-aged women (aged 30–60 years), and this particular variant manifests with asymptomatic or mildly pruritic, hyperpigmented patches and plaques in intertriginous areas. Although it also shows hyperkeratosis on histopathology, it can be differentiated from granular parakeratosis by the additional findings of epidermal hypergranulosis, sawtooth acanthosis of rete ridges, apoptotic keratinocytes in the dermoepidermal junction, and lymphocytic infiltrate in the upper dermis.8
Hailey-Hailey disease (also known as familial benign pemphigus) is a rare condition caused by an autosomaldominant mutation affecting intracellular calcium signaling that impairs keratinocyte adhesion.9 Similar to granular parakeratosis, it is most common in middle-aged adults (aged 30–40 years) and manifests as pruritic and burning lesions in symmetric intertriginous areas that also can be triggered by heat and sweating. However, patients present with recurrent blistering and vesicular lesions that may lead to erosions and secondary infections, which reduced clinical suspicion for this diagnosis in our patient. Histopathology shows suprabasilar and intraepidermal clefts, full-thickness acantholysis, protruding dermal papillae, and a perivascular lymphocytic infiltrate in the superficial dermis.9
The Diagnosis: Granular Parakeratosis
Histopathology demonstrated diffuse parakeratosis with retention of keratohyalin granules throughout the stratum corneum consistent with a diagnosis of granular parakeratosis (Figure), a rare benign cutaneous condition that is thought to occur due to a defect in epidermal differentiation. The lesion resolved without additional treatment.

The pathogenesis of granular parakeratosis is unclear, but a reactive process in which locoregional irritation or occlusion prompts increased cell turnover and prevention of profilaggrin breakdown has been proposed.1,2 The diagnosis is linked to various precipitating agents, most commonly topical products (eg, zinc oxide, antiperspirants) and products with benzalkonium chloride (eg, laundry rinses). These agents are thought to cause retention of keratohyalin granules in the stratum corneum during epidermal differentiation.1,2
Most affected patients are middle-aged women (mean age at diagnosis, 37.8 years).2 Patients present with eruptions of erythematous, brown, hyperkeratotic patches and papules that coalesce into plaques.1,2 These lesions can be pruritic and painful or asymptomatic. They often manifest bilaterally in intertriginous sites, most commonly the axillae, groin, or inguinal folds.1,2
Treatment involves identification and removal of potential triggers including changing antiperspirants, limiting use of irritating agents (eg, topical products with strong fragrances), and reducing heat and moisture in the affected areas. If the lesion persists, stepwise treatment can be initiated with topical agents (eg, corticosteroids, vitamin D analogues, retinoids, keratolytics, calcineurin inhibitors) followed by systemic medications (eg, antibiotics, isotretinoin, antifungals, dexamethasone) and procedures (eg, botulinum toxin injections, surgery, laser, cryotherapy).1,2
Unilateral granular parakeratosis, as seen in our patient, is an uncommon manifestation. Our case supports the theory that occlusion is a precipitating factor for this condition, given persistent axillary exposure to heat, sweat, and friction in the setting of limb immobilization.3
Granular parakeratosis is a challenge to diagnose due to clinical overlap with several other cutaneous conditions; histopathologic confirmation is required. Fox- Fordyce disease is a rare condition that is thought to result from keratin buildup or occlusion of apocrine or apoeccrine sweat ducts leading to duct rupture and surrounding inflammation.4 Common triggers include laser hair removal, hormonal changes, and living conditions that promote hot and humid environments.5 It can manifest similarly to granular parakeratosis, with eruptions of multiple red-violet papules that appear bilaterally in aprocine gland–rich areas, including the axillae and less commonly the genital, periareolar, thoracic, abdominal, and facial areas.4,5 However, most patients with Fox-Fordyce disease tend to be younger females (aged 13–35 years) with severely pruritic lesions,4,5 unlike our patient. In addition, histopathology shows hyperkeratosis, hair follicle plugging, and sweat gland and duct dilation.4
Seborrheic keratoses are common benign epidermal tumors caused by an overproliferation of immature keratinocytes.6,7 Similar to granular parakeratosis, they commonly manifest in older adults as hyperpigmented, well-demarcated, verrucous plaques with a hyperkeratotic surface.6 However, they are more common on the face, neck, trunk, and extremities, and they tend to be asymptomatic, differentiating them from granular parakerosis.6 Histopathology demonstrates a papillomatous epidermal surface, large capillaries in the dermal papillae, and intraepidermal and pseudohorn epidermal cysts.7
Inverse lichen planus, a variant of lichen planus, is a rare inflammatory condition that involves the lysis of basal keratinocytes by CD8+ lymphocytes.8 Similar to granular parakeratosis, lichen planus commonly affects middle-aged women (aged 30–60 years), and this particular variant manifests with asymptomatic or mildly pruritic, hyperpigmented patches and plaques in intertriginous areas. Although it also shows hyperkeratosis on histopathology, it can be differentiated from granular parakeratosis by the additional findings of epidermal hypergranulosis, sawtooth acanthosis of rete ridges, apoptotic keratinocytes in the dermoepidermal junction, and lymphocytic infiltrate in the upper dermis.8
Hailey-Hailey disease (also known as familial benign pemphigus) is a rare condition caused by an autosomaldominant mutation affecting intracellular calcium signaling that impairs keratinocyte adhesion.9 Similar to granular parakeratosis, it is most common in middle-aged adults (aged 30–40 years) and manifests as pruritic and burning lesions in symmetric intertriginous areas that also can be triggered by heat and sweating. However, patients present with recurrent blistering and vesicular lesions that may lead to erosions and secondary infections, which reduced clinical suspicion for this diagnosis in our patient. Histopathology shows suprabasilar and intraepidermal clefts, full-thickness acantholysis, protruding dermal papillae, and a perivascular lymphocytic infiltrate in the superficial dermis.9
- Ding CY, Liu H, Khachemoune A. Granular parakeratosis: a comprehensive review and a critical reappraisal. Am J Clin Dermatol. 2015;16:495-500. doi:10.1007/s40257-015-0148-2
- Ip KH, Li A. Clinical features, histology, and treatment outcomes of granular parakeratosis: a systematic review. Int J Dermatol. 2022;61:973-978. doi:10.1111/ijd.16107
- Mehregan DA, Thomas JE, Mehregan DR. Intertriginous granular parakeratosis. J Am Acad Dermatol. 1998;39:495-496. doi:10.1016/s0190-9622(98)70333-0
- Kamada A, Saga K, Jimbow K. Apoeccrine sweat duct obstruction as a cause for Fox-Fordyce disease. J Am Acad Dermatol. 2003;48:453-455. doi:10.1067/mjd.2003.93
- Salloum A, Bouferraa Y, Bazzi N, et al. Pathophysiology, clinical findings, and management of Fox-Fordyce disease: a systematic review. J Cosmet Dermatol. 2022;21:482-500. doi:10.1111/jocd.14135
- Sun MD, Halpern AC. Advances in the etiology, detection, and clinical management of seborrheic keratoses. Dermatology. 2022;238:205-217. doi:10.1159/000517070
- Minagawa A. Dermoscopy-pathology relationship in seborrheic keratosis. J Dermatol. 2017;44:518-524. doi:10.1111/1346-8138.13657
- Weston G, Payette M. Update on lichen planus and its clinical variants [published online September 16, 2015]. Int J Womens Dermatol. 2015;1:140-149. doi:10.1016/j.ijwd.2015.04.001
- Ben Lagha I, Ashack K, Khachemoune A. Hailey-Hailey disease: an update review with a focus on treatment data. Am J Clin Dermatol. 2020;21:49-68. doi:10.1007/s40257-019-00477-z
- Ding CY, Liu H, Khachemoune A. Granular parakeratosis: a comprehensive review and a critical reappraisal. Am J Clin Dermatol. 2015;16:495-500. doi:10.1007/s40257-015-0148-2
- Ip KH, Li A. Clinical features, histology, and treatment outcomes of granular parakeratosis: a systematic review. Int J Dermatol. 2022;61:973-978. doi:10.1111/ijd.16107
- Mehregan DA, Thomas JE, Mehregan DR. Intertriginous granular parakeratosis. J Am Acad Dermatol. 1998;39:495-496. doi:10.1016/s0190-9622(98)70333-0
- Kamada A, Saga K, Jimbow K. Apoeccrine sweat duct obstruction as a cause for Fox-Fordyce disease. J Am Acad Dermatol. 2003;48:453-455. doi:10.1067/mjd.2003.93
- Salloum A, Bouferraa Y, Bazzi N, et al. Pathophysiology, clinical findings, and management of Fox-Fordyce disease: a systematic review. J Cosmet Dermatol. 2022;21:482-500. doi:10.1111/jocd.14135
- Sun MD, Halpern AC. Advances in the etiology, detection, and clinical management of seborrheic keratoses. Dermatology. 2022;238:205-217. doi:10.1159/000517070
- Minagawa A. Dermoscopy-pathology relationship in seborrheic keratosis. J Dermatol. 2017;44:518-524. doi:10.1111/1346-8138.13657
- Weston G, Payette M. Update on lichen planus and its clinical variants [published online September 16, 2015]. Int J Womens Dermatol. 2015;1:140-149. doi:10.1016/j.ijwd.2015.04.001
- Ben Lagha I, Ashack K, Khachemoune A. Hailey-Hailey disease: an update review with a focus on treatment data. Am J Clin Dermatol. 2020;21:49-68. doi:10.1007/s40257-019-00477-z
A 62-year-old woman presented to our clinic for evaluation of a brown plaque in the left axilla of 2 weeks’ duration. She had a history of a rotator cuff injury and adhesive capsulitis several months prior that required immobilization of the left arm in a shoulder orthosis for several months. After the sling was removed, she noticed the lesion and reported mild cutaneous pain. Physical examination revealed a 1.5-cm, verrucous, red-brown plaque in the left axillary vault. A shave biopsy of the plaque was performed.
A Pharmacist-Led Process to Monitor Discrepant Urine Drug Screen Results
Urine drug screen (UDS) monitoring is a common risk-mitigation strategy tool for prescribing controlled substances.1-3 Not only is UDS monitoring highlighted by clinical practice guidelines for opioid prescribing for chronic pain,1,2 it has also been suggested as best practice for benzodiazepines3 and a consideration for other controlled substances. Monitoring UDSs helps confirm adherence to the prescribed treatment regimen while also screening for substance use that may increase patient risk.
UDS results can be complex and have profound implications for the patient’s treatment plan. Drug metabolites for opioids are particularly complicated; for example, synthetic and semisynthetic opioids are not detected on routine opiate immunoassays.4 This may lead a clinician to falsely assume the patient is not taking their fentanyl or tramadol medication as directed—or potentially even diverting—in the face of a negative opiate result.5 Routine UDSs are also subject to the pitfall of false-positive results due to coprescribed medications; for example, bupropion can lead to a false-positive amphetamine result, whereas sertraline can lead to a false-positive benzodiazepine result.6 Retrospective reviews of clinician behavior surrounding UDS interpretation have demonstrated knowledge gaps and inconsistent communication practices with patients.7,8
Given the complexity of UDS interpretation and its close relationship with medications, pharmacists are positioned to play an important role in the process. Pharmacists are embedded in pain-management teams and involved in prescription drug monitoring programs (PDMPs) for many health systems. The Veterans Health Administration (VHA) has supported the hiring of pain management, opioid safety, and PDMP coordinators (PMOP) at its facilities to provide clinical pain-management guidance, support national initiatives, and uphold legislative requirements.9 In many facilities, a pharmacist is hired specifically for these positions.
Clinical dashboards have been used by pharmacists in a variety of settings.10-13 They allow clinicians at a broad level to target interventions needed across a patient population, then produce a list of actionable patients to facilitate delivery of that intervention on an individual level.13 Between 2021 and 2022, a clinical dashboard to review potentially discrepant UDS results was made available for use at US Department of Veterans Affairs (VA) medical centers. Evidence exists in primary and specialty care settings that implementation of an opioid-prescribing clinical dashboard improves completion rates of risk-mitigation strategies such as UDS and opioid treatment agreements.14,15 To our knowledge there is no published research on the use and outcomes of a clinical dashboard that allows users to efficiently review discrepant UDS results when compared to a list of currently prescribed medications.
Given the availability of the UDS dashboard at the VA Black Hills Health Care System (VABHHCS) in South Dakota and the hiring of a PMOP coordinator pharmacist, the aim of this quality improvement project was 2-fold: to implement a pharmacist-led process to monitor the UDS dashboard for potentially discrepant results and to describe the quantity and types of interventions made by the clinical pharmacist leading this process.
Quality Improvement Project
A clinical UDS dashboard was created by the VA Northwest Health Network and made available for use at VHA sites between 2021 and 2022. The UDS dashboard is housed on a secure, Power BI Report Server (Microsoft), with access restricted to only those with patient health data privileges. The dashboard identifies all local patients with a UDS that returned with a potential discrepancy, defined as an unexpected positive result (eg, a detected substance not recently prescribed or documented on the patient’s medication list) and/or an unexpected negative result (eg, a prescribed substance not detected). The UDS dashboard identifies these discrepancies by comparing the patient’s current medication list (both VHA and non-VHA) to their UDS results.
The UDS dashboard displays a summary of UDSs performed, unexpected negative results, unexpected positive results, and potential discrepancies. The user may also specify the laboratory type and time frame of interest to limit displayed results. The user can then view patient-specific data for any category. Among the data are the patient’s UDS results and the completion date, detected (or nondetected) substance(s), ordering clinician, associated medication(s) with last fill date and days’ supply, and whether a confirmatory test has been performed in the past year.
VABHHCS uses an extended UDS immunoassay (PROFILE-V, MEDTOX Diagnostics) that reports on 11 substances: opiates, oxycodone, buprenorphine, methadone, amphetamines, methamphetamine, barbiturates, benzodiazepines, cocaine metabolites, cannabinoids (tetrahydrocannabinol [THC]), and phencyclidine. These substances appear on the UDS dashboard. The project protocol initially included monitoring for tramadol but that was later removed because it was not available with this UDS immunoassay.
Pharmacist Process
Either the PMOP coordinator or pharmacy resident monitored the UDS dashboard weekly. Any patients identified as having a potential discrepancy were reviewed. If the discrepancy was determined to be significant, the PMOP coordinator or pharmacy resident would review the patient electronic health record. If warranted, the patient was contacted and asked about newly prescribed medications, missed and recent medication doses, and illicit substance use. Potential interventions during in-depth review included: (1) discussing future actions with the primary care clinician and/or prescriber of the controlled substance; (2) ordering a confirmatory test on the original urine sample; (3) evaluating for sources of potential false-positive results; (4) completing an updated PDMP if not performed within the past year; (5) referring patients for substance use disorder treatment or counseling; or (6) consulting the local narcotics review committee. A progress note was entered into the electronic health record with the findings and any actions taken, and an alert for the primary care clinician and/or prescriber of the controlled substance.
Implementation and Analysis
This quality improvement project spanned 16 weeks from June 2022 through September 2022. Any patient with a UDS that returned with a significant discrepancy was reviewed. The primary outcome was interventions made by the PMOP coordinator or pharmacy resident, as well as time taken to perform the in-depth review of each patient. Patient demographics were also collected. The protocol for this project was approved by the VABHHCS pharmacy and therapeutics committee and was determined to meet guidelines for a nonresearch quality improvement project.
Results
From June 2022 through September 2022, 700 UDSs were performed at VABHHCS with 278 (39.7%) patients identified as having a potential discrepancy based on UDS results. Sixty patients (8.6%) had significant discrepancies that warranted in-depth review. The most common reasons for determining whether a potential discrepancy was not significant included unexpected negatives due to documented non-VA medications no longer being prescribed, unexpected positives due to recent expiration of a controlled substance prescription the patient was still taking, or unexpected positives due to the detection of a substance for which the clinician was already aware. During the 16-week study period, the mean number of patients warranting in-depth review was 4 per week.
The patients were predominantly male with a mean age of 61 years, and most (87%) were prescribed at least 1 controlled substance (mean, 1.1), primarily opioids for pain management (Table 1). Most patients had recent substance risk mitigation with UDS (56%) and PDMP (65%) checks within the past year. Of the 60 patients reviewed with significant UDS discrepancies, 50% had a history of discrepant UDS results. Of the 60 UDS discrepancies, there were 37 unexpected positive results (62%), 17 unexpected negative results (28%), and 10 patients with both positive and negative results (17%). THC was the most frequently detected substance, followed by opiates, benzodiazepines, and amphetamines (Table 2).
Each in-depth review with interventions by the PMOP coordinator or pharmacy resident lasted a mean of 14 minutes (Table 3). Five patients were successfully contacted for an interview and 7 patients could not be contacted. The ordering clinician of the UDS sometimes had contacted these patients prior to the PMOP coordinator or pharmacy resident reviewing the UDS dashboard, eliminating the need for additional follow-up.
The most common pharmacist intervention was discussing future actions with the primary care clinician and/or prescriber of the controlled substance (n = 39; 65%). These conversations resulted in actions such as ordering a repeat UDS with confirmatory testing at a future date or agreeing that the clinician would discuss the results and subsequent actions with the patient at an upcoming visit. Pharmacist interventions also included 25 PDMP queries (42%) and 9 orders of confirmatory UDS on the original urine sample (15%). Only 1 patient was evaluated by the narcotics review committee, which resulted in a controlled substance flag being placed on their profile. No patients were referred to substance use disorder treatment or counseling. It was offered to and declined by 1 patient, and 3 patients were already engaged in these services.
Medication therapies that could contribute to false-positive results were also evaluated. Fourteen patients who tested positive for THC had a prescription for a nonsteroidal anti-inflammatory drug or proton-pump inhibitor, which could have created a false-positive result.6 One patient who tested positive for amphetamines had a prescription for phentermine.16 No other potential false-positive results were identified.
Discussion
Findings of this project illustrate that the use of a clinical pharmacist to monitor a dashboard of discrepant UDS results created opportunities for collaboration with clinicians and impacted confirmatory testing and PDMP monitoring practices.
At the local level, the process had numerous benefits. First, it was a reasonable amount of workload to generate pharmacist interventions: the PMOP coordinator conducted an average of 4 in-depth reviews weekly, each lasting about 14 minutes. Thus, the UDS dashboard allowed the PMOP coordinator to actively surveil all incoming UDS results for potential discrepancies in about 1 hour each week. Pairing the automation of the UDS dashboard with the clinical judgment of the PMOP coordinator seemed to maximize efficiency. VABHHCS provides primary and secondary medical and surgical care to a rural population of approximately 20,000 patients across 5 states; the time required at facilities that serve a higher volume of patients may be greater.
Second, the project served as an opportunity for the PMOP coordinator to provide case-specific clinician education on UDS monitoring. As medication experts, pharmacists can apply their medication-related knowledge to UDS interpretation. This includes understanding drug metabolism and classification and how they apply to UDS results, as well as recognizing medication therapies that could contribute to false-positive UDS results. Research suggests that clinicians may have gaps in their knowledge and may welcome pharmacist assistance in interpreting UDS results.7,8
Third, the project helped improve rates of confirmatory testing for those with unexpected positive UDS results. Confirmatory testing should be strongly considered if positive results would have significant implications on the future course of treatment.4 The PMOP coordinator ordered a confirmatory test on 9 patients using the same urine sample used to conduct the initial UDS, minimizing the burden on the patient and laboratory staff. Confirmatory testing was limited by the laboratory’s sample retention period; if the need for confirmatory testing was not recognized soon enough, the sample would no longer be available for retesting. Health systems may consider the use of reflexive confirmatory testing with UDS as an alternative approach, although this may come at an additional cost and may not be warranted in many cases (eg, only 39.7% of all potential discrepancies were deemed as significant within our project).
There were notable incidental findings in our quality improvement project. Among patients with a significant discrepancy on UDS, 50% had a history of ≥ 1 discrepant UDS result. This further emphasizes the importance of appropriate use and interpretation of UDS monitoring for all clinicians, as this may prevent prolonged and potentially inappropriate treatment regimens. Secondly, rates of mental health diagnoses among those with a significant UDS discrepancy seemed relatively high compared to population-level data. For example, among veterans, the overall lifetime prevalence of posttraumatic stress disorder is estimated to be 8.0%; in our project, 35% of patients with a significant UDS discrepancy had a posttraumatic stress disorder diagnosis.17 This relationship may be an area of further study.
Lastly, it was surprising that the overall rates of UDS and PDMP checks within the past year were 56% and 65%, respectively. VABHHCS requires veterans on controlled substances to have these risk-mitigation strategies performed annually, so our suspicion is that many were falling out due to having been most recently evaluated 12 to 16 months prior. This may represent a limitation of our data-collection method, which reviewed only the previous 12 months.
Limitations
This project was carried out over a period of only 4 months. As a result, only 60 patients received an in-depth review from the PMOP coordinator. Second, the timeliness of the intervention seemed crucial, as delayed in-depth reviews resulted in fewer opportunities to order confirmatory tests or collaborate with clinicians prior to devising an updated plan. Additionally, our process called for UDS dashboard monitoring once a week. Given that the laboratory held samples for only 48 hours, twice- or thrice-weekly review of the UDS dashboard would have allowed for more confirmatory testing, along with more immediate clinician collaboration. Most importantly, the outcomes of this project are only presented via descriptive statistics and without the results of any comparison group, making it impossible to draw firm conclusions about this approach compared to standard-care processes.
Conclusions
This quality improvement project has proven to be valuable at VABHHCS and we intend to continue this pharmacist-led process to monitor the UDS dashboard. VABHHCS leadership are also discussing UDS practices more broadly to further enhance patient management. Within the VA, the PMOP coordinator—charged with being the local coordinator of appropriate pain management and opioid safety practices—is well positioned to assume these responsibilities. Outside of the VA, a pain-management clinical pharmacist or any pharmacist embedded within primary care could similarly perform these duties. Previous literature regarding the implementation of clinical dashboards suggests that with the appropriate software engineering teams and infrastructure, this tool could also be feasibly developed and implemented at other health systems relatively quickly.14
Overall, a pharmacist-led process to efficiently monitor a dashboard of discrepant UDS results led to opportunities for collaboration with clinicians and positively impacted confirmatory testing and PDMP monitoring at a rural VA health system.
Acknowledgments
The authors express their gratitude to Patrick Spoutz, PharmD, BCPS, VISN 20 Pharmacist Executive, for introducing and sharing the UDS dashboard with our team.
1. Dowell D, Ragan KR, Jones CM, Baldwin GT, Chou R. CDC Clinical Practice Guideline for Prescribing Opioids for Pain - United States, 2022. MMWR Recomm Rep. 2022;71(3):1-95. doi:10.15585/mmwr.rr7103a1
2. US Department of Defense, US Department of Veterans Affairs. VA/DoD clinical practice guidelines for the use of opioids in the management of chronic pain. Version 4.0. Published 2002. Accessed January 22, 2024. https://www.healthquality.va.gov/guidelines/Pain/cot/VADoDOpioidsCPG.pdf
3. Champion C, Kameg BN. Best practices in benzodiazepine prescribing and management in primary care. Nurse Pract. 2021;46(3):30-36.doi:10.1097/01.NPR.0000733684.24949.19
4. Kale N. Urine drug tests: ordering and interpretation. Am Fam Physician. 2019;99(1):33-39.
5. Gillespie E, Cunningham JM, Indovina KA. Interpretation of the urine drug screen. The Hospitalist. May 2, 2022. Accessed January 19, 2024. https://www.the-hospitalist.org/hospitalist/article/32085/interpreting-diagnostic-tests/interpretation-of-the-urine-drug-screen/
6. Schwebach A, Ball J. Urine drug screening: minimizing false-positives and false-negatives to optimize patient care. US Pharm. 2016;41(8):26-30.
7. Starrels JL, Fox AD, Kunins HV, Cunningham CO. They don’t know what they don’t know: internal medicine residents’ knowledge and confidence in urine drug test interpretation for patients with chronic pain. J Gen Intern Med. 2012;27(11):1521-1527. doi:10.1007/s11606-012-2165-7
8. Chua I, Petrides AK, Schiff GD, et al. Provider misinterpretation, documentation, and follow-up of definitive urine drug testing results. J Gen Intern Med. 2020;35(1):283-290. doi:10.1007/s11606-019-05514-5
9. US Department of Veterans Affairs, Veterans Health Administration. VHA Pain Management, Opioid Safety, and Prescription Drug Monitoring Program (PMOP) National Program Field Roles and Responsibilities Manual. October 2021 (V1).[Source not verified]
10. Dorsch MP, Chen CS, Allen AL, et al. Nationwide implementation of a population management dashboard for monitoring direct oral anticoagulants: insights from the Veterans Affairs Health System. Circ Cardiovasc Qual Outcomes. 2023;16(2):e009256. doi:10.1161/CIRCOUTCOMES.122.009256
11. Hu AM, Pepin MJ, Hashem MG, et al. Development of a specialty medication clinical dashboard to improve tumor necrosis factor-α inhibitor safety and adherence monitoring. Am J Health Syst Pharm. 2022;79(8):683-688. doi:10.1093/ajhp/zxab454
12. Homsted FAE, Magee CE, Nesin N. Population health management in a small health system: impact of controlled substance stewardship in a patient-centered medical home. Am J Health Syst Pharm. 2017;74(18):1468-1475. doi:10.2146/ajhp161032
13. US Department of Veterans Affairs, Veterans Health Administration, Pharmacy Benefits (PBM) Services, Clinical Pharmacy Practice Office. Fact Sheet: CPS Role in Population Health Management. 2019. [Source not verified]
14. Anderson D, Zlateva I, Khatri K, Ciaburri N. Using health information technology to improve adherence to opioid prescribing guidelines in primary care. Clin J Pain. 2015;31(6):573-579. doi:10.1097/AJP.0000000000000177
15. Wang EJ, Helgesen R, Johr CR, Lacko HS, Ashburn MA, Merkel PA. Targeted program in an academic rheumatology practice to improve compliance with opioid prescribing guidelines for the treatment of chronic pain. Arthritis Care Res (Hoboken). 2021;73(10):1425-1429. doi:10.1002/acr.24354
16. Moeller KE, Kissack JC, Atayee RS, Lee KC. Clinical interpretation of urine drug tests: what clinicians need to know about urine drug screens. Mayo Clin Proc. 2017;92(5):774-796. doi:10.1016/j.mayocp.2016.12.007
17. Wisco BE, Marx BP, Wolf EJ, Miller MW, Southwick SM, Pietrzak RH. Posttraumatic stress disorder in the US veteran population: results from the National Health and Resilience in Veterans Study. J Clin Psychiatry. 2014;75(12):1338-46. doi:10.4088/JCP.14m09328
Urine drug screen (UDS) monitoring is a common risk-mitigation strategy tool for prescribing controlled substances.1-3 Not only is UDS monitoring highlighted by clinical practice guidelines for opioid prescribing for chronic pain,1,2 it has also been suggested as best practice for benzodiazepines3 and a consideration for other controlled substances. Monitoring UDSs helps confirm adherence to the prescribed treatment regimen while also screening for substance use that may increase patient risk.
UDS results can be complex and have profound implications for the patient’s treatment plan. Drug metabolites for opioids are particularly complicated; for example, synthetic and semisynthetic opioids are not detected on routine opiate immunoassays.4 This may lead a clinician to falsely assume the patient is not taking their fentanyl or tramadol medication as directed—or potentially even diverting—in the face of a negative opiate result.5 Routine UDSs are also subject to the pitfall of false-positive results due to coprescribed medications; for example, bupropion can lead to a false-positive amphetamine result, whereas sertraline can lead to a false-positive benzodiazepine result.6 Retrospective reviews of clinician behavior surrounding UDS interpretation have demonstrated knowledge gaps and inconsistent communication practices with patients.7,8
Given the complexity of UDS interpretation and its close relationship with medications, pharmacists are positioned to play an important role in the process. Pharmacists are embedded in pain-management teams and involved in prescription drug monitoring programs (PDMPs) for many health systems. The Veterans Health Administration (VHA) has supported the hiring of pain management, opioid safety, and PDMP coordinators (PMOP) at its facilities to provide clinical pain-management guidance, support national initiatives, and uphold legislative requirements.9 In many facilities, a pharmacist is hired specifically for these positions.
Clinical dashboards have been used by pharmacists in a variety of settings.10-13 They allow clinicians at a broad level to target interventions needed across a patient population, then produce a list of actionable patients to facilitate delivery of that intervention on an individual level.13 Between 2021 and 2022, a clinical dashboard to review potentially discrepant UDS results was made available for use at US Department of Veterans Affairs (VA) medical centers. Evidence exists in primary and specialty care settings that implementation of an opioid-prescribing clinical dashboard improves completion rates of risk-mitigation strategies such as UDS and opioid treatment agreements.14,15 To our knowledge there is no published research on the use and outcomes of a clinical dashboard that allows users to efficiently review discrepant UDS results when compared to a list of currently prescribed medications.
Given the availability of the UDS dashboard at the VA Black Hills Health Care System (VABHHCS) in South Dakota and the hiring of a PMOP coordinator pharmacist, the aim of this quality improvement project was 2-fold: to implement a pharmacist-led process to monitor the UDS dashboard for potentially discrepant results and to describe the quantity and types of interventions made by the clinical pharmacist leading this process.
Quality Improvement Project
A clinical UDS dashboard was created by the VA Northwest Health Network and made available for use at VHA sites between 2021 and 2022. The UDS dashboard is housed on a secure, Power BI Report Server (Microsoft), with access restricted to only those with patient health data privileges. The dashboard identifies all local patients with a UDS that returned with a potential discrepancy, defined as an unexpected positive result (eg, a detected substance not recently prescribed or documented on the patient’s medication list) and/or an unexpected negative result (eg, a prescribed substance not detected). The UDS dashboard identifies these discrepancies by comparing the patient’s current medication list (both VHA and non-VHA) to their UDS results.
The UDS dashboard displays a summary of UDSs performed, unexpected negative results, unexpected positive results, and potential discrepancies. The user may also specify the laboratory type and time frame of interest to limit displayed results. The user can then view patient-specific data for any category. Among the data are the patient’s UDS results and the completion date, detected (or nondetected) substance(s), ordering clinician, associated medication(s) with last fill date and days’ supply, and whether a confirmatory test has been performed in the past year.
VABHHCS uses an extended UDS immunoassay (PROFILE-V, MEDTOX Diagnostics) that reports on 11 substances: opiates, oxycodone, buprenorphine, methadone, amphetamines, methamphetamine, barbiturates, benzodiazepines, cocaine metabolites, cannabinoids (tetrahydrocannabinol [THC]), and phencyclidine. These substances appear on the UDS dashboard. The project protocol initially included monitoring for tramadol but that was later removed because it was not available with this UDS immunoassay.
Pharmacist Process
Either the PMOP coordinator or pharmacy resident monitored the UDS dashboard weekly. Any patients identified as having a potential discrepancy were reviewed. If the discrepancy was determined to be significant, the PMOP coordinator or pharmacy resident would review the patient electronic health record. If warranted, the patient was contacted and asked about newly prescribed medications, missed and recent medication doses, and illicit substance use. Potential interventions during in-depth review included: (1) discussing future actions with the primary care clinician and/or prescriber of the controlled substance; (2) ordering a confirmatory test on the original urine sample; (3) evaluating for sources of potential false-positive results; (4) completing an updated PDMP if not performed within the past year; (5) referring patients for substance use disorder treatment or counseling; or (6) consulting the local narcotics review committee. A progress note was entered into the electronic health record with the findings and any actions taken, and an alert for the primary care clinician and/or prescriber of the controlled substance.
Implementation and Analysis
This quality improvement project spanned 16 weeks from June 2022 through September 2022. Any patient with a UDS that returned with a significant discrepancy was reviewed. The primary outcome was interventions made by the PMOP coordinator or pharmacy resident, as well as time taken to perform the in-depth review of each patient. Patient demographics were also collected. The protocol for this project was approved by the VABHHCS pharmacy and therapeutics committee and was determined to meet guidelines for a nonresearch quality improvement project.
Results
From June 2022 through September 2022, 700 UDSs were performed at VABHHCS with 278 (39.7%) patients identified as having a potential discrepancy based on UDS results. Sixty patients (8.6%) had significant discrepancies that warranted in-depth review. The most common reasons for determining whether a potential discrepancy was not significant included unexpected negatives due to documented non-VA medications no longer being prescribed, unexpected positives due to recent expiration of a controlled substance prescription the patient was still taking, or unexpected positives due to the detection of a substance for which the clinician was already aware. During the 16-week study period, the mean number of patients warranting in-depth review was 4 per week.
The patients were predominantly male with a mean age of 61 years, and most (87%) were prescribed at least 1 controlled substance (mean, 1.1), primarily opioids for pain management (Table 1). Most patients had recent substance risk mitigation with UDS (56%) and PDMP (65%) checks within the past year. Of the 60 patients reviewed with significant UDS discrepancies, 50% had a history of discrepant UDS results. Of the 60 UDS discrepancies, there were 37 unexpected positive results (62%), 17 unexpected negative results (28%), and 10 patients with both positive and negative results (17%). THC was the most frequently detected substance, followed by opiates, benzodiazepines, and amphetamines (Table 2).
Each in-depth review with interventions by the PMOP coordinator or pharmacy resident lasted a mean of 14 minutes (Table 3). Five patients were successfully contacted for an interview and 7 patients could not be contacted. The ordering clinician of the UDS sometimes had contacted these patients prior to the PMOP coordinator or pharmacy resident reviewing the UDS dashboard, eliminating the need for additional follow-up.
The most common pharmacist intervention was discussing future actions with the primary care clinician and/or prescriber of the controlled substance (n = 39; 65%). These conversations resulted in actions such as ordering a repeat UDS with confirmatory testing at a future date or agreeing that the clinician would discuss the results and subsequent actions with the patient at an upcoming visit. Pharmacist interventions also included 25 PDMP queries (42%) and 9 orders of confirmatory UDS on the original urine sample (15%). Only 1 patient was evaluated by the narcotics review committee, which resulted in a controlled substance flag being placed on their profile. No patients were referred to substance use disorder treatment or counseling. It was offered to and declined by 1 patient, and 3 patients were already engaged in these services.
Medication therapies that could contribute to false-positive results were also evaluated. Fourteen patients who tested positive for THC had a prescription for a nonsteroidal anti-inflammatory drug or proton-pump inhibitor, which could have created a false-positive result.6 One patient who tested positive for amphetamines had a prescription for phentermine.16 No other potential false-positive results were identified.
Discussion
Findings of this project illustrate that the use of a clinical pharmacist to monitor a dashboard of discrepant UDS results created opportunities for collaboration with clinicians and impacted confirmatory testing and PDMP monitoring practices.
At the local level, the process had numerous benefits. First, it was a reasonable amount of workload to generate pharmacist interventions: the PMOP coordinator conducted an average of 4 in-depth reviews weekly, each lasting about 14 minutes. Thus, the UDS dashboard allowed the PMOP coordinator to actively surveil all incoming UDS results for potential discrepancies in about 1 hour each week. Pairing the automation of the UDS dashboard with the clinical judgment of the PMOP coordinator seemed to maximize efficiency. VABHHCS provides primary and secondary medical and surgical care to a rural population of approximately 20,000 patients across 5 states; the time required at facilities that serve a higher volume of patients may be greater.
Second, the project served as an opportunity for the PMOP coordinator to provide case-specific clinician education on UDS monitoring. As medication experts, pharmacists can apply their medication-related knowledge to UDS interpretation. This includes understanding drug metabolism and classification and how they apply to UDS results, as well as recognizing medication therapies that could contribute to false-positive UDS results. Research suggests that clinicians may have gaps in their knowledge and may welcome pharmacist assistance in interpreting UDS results.7,8
Third, the project helped improve rates of confirmatory testing for those with unexpected positive UDS results. Confirmatory testing should be strongly considered if positive results would have significant implications on the future course of treatment.4 The PMOP coordinator ordered a confirmatory test on 9 patients using the same urine sample used to conduct the initial UDS, minimizing the burden on the patient and laboratory staff. Confirmatory testing was limited by the laboratory’s sample retention period; if the need for confirmatory testing was not recognized soon enough, the sample would no longer be available for retesting. Health systems may consider the use of reflexive confirmatory testing with UDS as an alternative approach, although this may come at an additional cost and may not be warranted in many cases (eg, only 39.7% of all potential discrepancies were deemed as significant within our project).
There were notable incidental findings in our quality improvement project. Among patients with a significant discrepancy on UDS, 50% had a history of ≥ 1 discrepant UDS result. This further emphasizes the importance of appropriate use and interpretation of UDS monitoring for all clinicians, as this may prevent prolonged and potentially inappropriate treatment regimens. Secondly, rates of mental health diagnoses among those with a significant UDS discrepancy seemed relatively high compared to population-level data. For example, among veterans, the overall lifetime prevalence of posttraumatic stress disorder is estimated to be 8.0%; in our project, 35% of patients with a significant UDS discrepancy had a posttraumatic stress disorder diagnosis.17 This relationship may be an area of further study.
Lastly, it was surprising that the overall rates of UDS and PDMP checks within the past year were 56% and 65%, respectively. VABHHCS requires veterans on controlled substances to have these risk-mitigation strategies performed annually, so our suspicion is that many were falling out due to having been most recently evaluated 12 to 16 months prior. This may represent a limitation of our data-collection method, which reviewed only the previous 12 months.
Limitations
This project was carried out over a period of only 4 months. As a result, only 60 patients received an in-depth review from the PMOP coordinator. Second, the timeliness of the intervention seemed crucial, as delayed in-depth reviews resulted in fewer opportunities to order confirmatory tests or collaborate with clinicians prior to devising an updated plan. Additionally, our process called for UDS dashboard monitoring once a week. Given that the laboratory held samples for only 48 hours, twice- or thrice-weekly review of the UDS dashboard would have allowed for more confirmatory testing, along with more immediate clinician collaboration. Most importantly, the outcomes of this project are only presented via descriptive statistics and without the results of any comparison group, making it impossible to draw firm conclusions about this approach compared to standard-care processes.
Conclusions
This quality improvement project has proven to be valuable at VABHHCS and we intend to continue this pharmacist-led process to monitor the UDS dashboard. VABHHCS leadership are also discussing UDS practices more broadly to further enhance patient management. Within the VA, the PMOP coordinator—charged with being the local coordinator of appropriate pain management and opioid safety practices—is well positioned to assume these responsibilities. Outside of the VA, a pain-management clinical pharmacist or any pharmacist embedded within primary care could similarly perform these duties. Previous literature regarding the implementation of clinical dashboards suggests that with the appropriate software engineering teams and infrastructure, this tool could also be feasibly developed and implemented at other health systems relatively quickly.14
Overall, a pharmacist-led process to efficiently monitor a dashboard of discrepant UDS results led to opportunities for collaboration with clinicians and positively impacted confirmatory testing and PDMP monitoring at a rural VA health system.
Acknowledgments
The authors express their gratitude to Patrick Spoutz, PharmD, BCPS, VISN 20 Pharmacist Executive, for introducing and sharing the UDS dashboard with our team.
Urine drug screen (UDS) monitoring is a common risk-mitigation strategy tool for prescribing controlled substances.1-3 Not only is UDS monitoring highlighted by clinical practice guidelines for opioid prescribing for chronic pain,1,2 it has also been suggested as best practice for benzodiazepines3 and a consideration for other controlled substances. Monitoring UDSs helps confirm adherence to the prescribed treatment regimen while also screening for substance use that may increase patient risk.
UDS results can be complex and have profound implications for the patient’s treatment plan. Drug metabolites for opioids are particularly complicated; for example, synthetic and semisynthetic opioids are not detected on routine opiate immunoassays.4 This may lead a clinician to falsely assume the patient is not taking their fentanyl or tramadol medication as directed—or potentially even diverting—in the face of a negative opiate result.5 Routine UDSs are also subject to the pitfall of false-positive results due to coprescribed medications; for example, bupropion can lead to a false-positive amphetamine result, whereas sertraline can lead to a false-positive benzodiazepine result.6 Retrospective reviews of clinician behavior surrounding UDS interpretation have demonstrated knowledge gaps and inconsistent communication practices with patients.7,8
Given the complexity of UDS interpretation and its close relationship with medications, pharmacists are positioned to play an important role in the process. Pharmacists are embedded in pain-management teams and involved in prescription drug monitoring programs (PDMPs) for many health systems. The Veterans Health Administration (VHA) has supported the hiring of pain management, opioid safety, and PDMP coordinators (PMOP) at its facilities to provide clinical pain-management guidance, support national initiatives, and uphold legislative requirements.9 In many facilities, a pharmacist is hired specifically for these positions.
Clinical dashboards have been used by pharmacists in a variety of settings.10-13 They allow clinicians at a broad level to target interventions needed across a patient population, then produce a list of actionable patients to facilitate delivery of that intervention on an individual level.13 Between 2021 and 2022, a clinical dashboard to review potentially discrepant UDS results was made available for use at US Department of Veterans Affairs (VA) medical centers. Evidence exists in primary and specialty care settings that implementation of an opioid-prescribing clinical dashboard improves completion rates of risk-mitigation strategies such as UDS and opioid treatment agreements.14,15 To our knowledge there is no published research on the use and outcomes of a clinical dashboard that allows users to efficiently review discrepant UDS results when compared to a list of currently prescribed medications.
Given the availability of the UDS dashboard at the VA Black Hills Health Care System (VABHHCS) in South Dakota and the hiring of a PMOP coordinator pharmacist, the aim of this quality improvement project was 2-fold: to implement a pharmacist-led process to monitor the UDS dashboard for potentially discrepant results and to describe the quantity and types of interventions made by the clinical pharmacist leading this process.
Quality Improvement Project
A clinical UDS dashboard was created by the VA Northwest Health Network and made available for use at VHA sites between 2021 and 2022. The UDS dashboard is housed on a secure, Power BI Report Server (Microsoft), with access restricted to only those with patient health data privileges. The dashboard identifies all local patients with a UDS that returned with a potential discrepancy, defined as an unexpected positive result (eg, a detected substance not recently prescribed or documented on the patient’s medication list) and/or an unexpected negative result (eg, a prescribed substance not detected). The UDS dashboard identifies these discrepancies by comparing the patient’s current medication list (both VHA and non-VHA) to their UDS results.
The UDS dashboard displays a summary of UDSs performed, unexpected negative results, unexpected positive results, and potential discrepancies. The user may also specify the laboratory type and time frame of interest to limit displayed results. The user can then view patient-specific data for any category. Among the data are the patient’s UDS results and the completion date, detected (or nondetected) substance(s), ordering clinician, associated medication(s) with last fill date and days’ supply, and whether a confirmatory test has been performed in the past year.
VABHHCS uses an extended UDS immunoassay (PROFILE-V, MEDTOX Diagnostics) that reports on 11 substances: opiates, oxycodone, buprenorphine, methadone, amphetamines, methamphetamine, barbiturates, benzodiazepines, cocaine metabolites, cannabinoids (tetrahydrocannabinol [THC]), and phencyclidine. These substances appear on the UDS dashboard. The project protocol initially included monitoring for tramadol but that was later removed because it was not available with this UDS immunoassay.
Pharmacist Process
Either the PMOP coordinator or pharmacy resident monitored the UDS dashboard weekly. Any patients identified as having a potential discrepancy were reviewed. If the discrepancy was determined to be significant, the PMOP coordinator or pharmacy resident would review the patient electronic health record. If warranted, the patient was contacted and asked about newly prescribed medications, missed and recent medication doses, and illicit substance use. Potential interventions during in-depth review included: (1) discussing future actions with the primary care clinician and/or prescriber of the controlled substance; (2) ordering a confirmatory test on the original urine sample; (3) evaluating for sources of potential false-positive results; (4) completing an updated PDMP if not performed within the past year; (5) referring patients for substance use disorder treatment or counseling; or (6) consulting the local narcotics review committee. A progress note was entered into the electronic health record with the findings and any actions taken, and an alert for the primary care clinician and/or prescriber of the controlled substance.
Implementation and Analysis
This quality improvement project spanned 16 weeks from June 2022 through September 2022. Any patient with a UDS that returned with a significant discrepancy was reviewed. The primary outcome was interventions made by the PMOP coordinator or pharmacy resident, as well as time taken to perform the in-depth review of each patient. Patient demographics were also collected. The protocol for this project was approved by the VABHHCS pharmacy and therapeutics committee and was determined to meet guidelines for a nonresearch quality improvement project.
Results
From June 2022 through September 2022, 700 UDSs were performed at VABHHCS with 278 (39.7%) patients identified as having a potential discrepancy based on UDS results. Sixty patients (8.6%) had significant discrepancies that warranted in-depth review. The most common reasons for determining whether a potential discrepancy was not significant included unexpected negatives due to documented non-VA medications no longer being prescribed, unexpected positives due to recent expiration of a controlled substance prescription the patient was still taking, or unexpected positives due to the detection of a substance for which the clinician was already aware. During the 16-week study period, the mean number of patients warranting in-depth review was 4 per week.
The patients were predominantly male with a mean age of 61 years, and most (87%) were prescribed at least 1 controlled substance (mean, 1.1), primarily opioids for pain management (Table 1). Most patients had recent substance risk mitigation with UDS (56%) and PDMP (65%) checks within the past year. Of the 60 patients reviewed with significant UDS discrepancies, 50% had a history of discrepant UDS results. Of the 60 UDS discrepancies, there were 37 unexpected positive results (62%), 17 unexpected negative results (28%), and 10 patients with both positive and negative results (17%). THC was the most frequently detected substance, followed by opiates, benzodiazepines, and amphetamines (Table 2).
Each in-depth review with interventions by the PMOP coordinator or pharmacy resident lasted a mean of 14 minutes (Table 3). Five patients were successfully contacted for an interview and 7 patients could not be contacted. The ordering clinician of the UDS sometimes had contacted these patients prior to the PMOP coordinator or pharmacy resident reviewing the UDS dashboard, eliminating the need for additional follow-up.
The most common pharmacist intervention was discussing future actions with the primary care clinician and/or prescriber of the controlled substance (n = 39; 65%). These conversations resulted in actions such as ordering a repeat UDS with confirmatory testing at a future date or agreeing that the clinician would discuss the results and subsequent actions with the patient at an upcoming visit. Pharmacist interventions also included 25 PDMP queries (42%) and 9 orders of confirmatory UDS on the original urine sample (15%). Only 1 patient was evaluated by the narcotics review committee, which resulted in a controlled substance flag being placed on their profile. No patients were referred to substance use disorder treatment or counseling. It was offered to and declined by 1 patient, and 3 patients were already engaged in these services.
Medication therapies that could contribute to false-positive results were also evaluated. Fourteen patients who tested positive for THC had a prescription for a nonsteroidal anti-inflammatory drug or proton-pump inhibitor, which could have created a false-positive result.6 One patient who tested positive for amphetamines had a prescription for phentermine.16 No other potential false-positive results were identified.
Discussion
Findings of this project illustrate that the use of a clinical pharmacist to monitor a dashboard of discrepant UDS results created opportunities for collaboration with clinicians and impacted confirmatory testing and PDMP monitoring practices.
At the local level, the process had numerous benefits. First, it was a reasonable amount of workload to generate pharmacist interventions: the PMOP coordinator conducted an average of 4 in-depth reviews weekly, each lasting about 14 minutes. Thus, the UDS dashboard allowed the PMOP coordinator to actively surveil all incoming UDS results for potential discrepancies in about 1 hour each week. Pairing the automation of the UDS dashboard with the clinical judgment of the PMOP coordinator seemed to maximize efficiency. VABHHCS provides primary and secondary medical and surgical care to a rural population of approximately 20,000 patients across 5 states; the time required at facilities that serve a higher volume of patients may be greater.
Second, the project served as an opportunity for the PMOP coordinator to provide case-specific clinician education on UDS monitoring. As medication experts, pharmacists can apply their medication-related knowledge to UDS interpretation. This includes understanding drug metabolism and classification and how they apply to UDS results, as well as recognizing medication therapies that could contribute to false-positive UDS results. Research suggests that clinicians may have gaps in their knowledge and may welcome pharmacist assistance in interpreting UDS results.7,8
Third, the project helped improve rates of confirmatory testing for those with unexpected positive UDS results. Confirmatory testing should be strongly considered if positive results would have significant implications on the future course of treatment.4 The PMOP coordinator ordered a confirmatory test on 9 patients using the same urine sample used to conduct the initial UDS, minimizing the burden on the patient and laboratory staff. Confirmatory testing was limited by the laboratory’s sample retention period; if the need for confirmatory testing was not recognized soon enough, the sample would no longer be available for retesting. Health systems may consider the use of reflexive confirmatory testing with UDS as an alternative approach, although this may come at an additional cost and may not be warranted in many cases (eg, only 39.7% of all potential discrepancies were deemed as significant within our project).
There were notable incidental findings in our quality improvement project. Among patients with a significant discrepancy on UDS, 50% had a history of ≥ 1 discrepant UDS result. This further emphasizes the importance of appropriate use and interpretation of UDS monitoring for all clinicians, as this may prevent prolonged and potentially inappropriate treatment regimens. Secondly, rates of mental health diagnoses among those with a significant UDS discrepancy seemed relatively high compared to population-level data. For example, among veterans, the overall lifetime prevalence of posttraumatic stress disorder is estimated to be 8.0%; in our project, 35% of patients with a significant UDS discrepancy had a posttraumatic stress disorder diagnosis.17 This relationship may be an area of further study.
Lastly, it was surprising that the overall rates of UDS and PDMP checks within the past year were 56% and 65%, respectively. VABHHCS requires veterans on controlled substances to have these risk-mitigation strategies performed annually, so our suspicion is that many were falling out due to having been most recently evaluated 12 to 16 months prior. This may represent a limitation of our data-collection method, which reviewed only the previous 12 months.
Limitations
This project was carried out over a period of only 4 months. As a result, only 60 patients received an in-depth review from the PMOP coordinator. Second, the timeliness of the intervention seemed crucial, as delayed in-depth reviews resulted in fewer opportunities to order confirmatory tests or collaborate with clinicians prior to devising an updated plan. Additionally, our process called for UDS dashboard monitoring once a week. Given that the laboratory held samples for only 48 hours, twice- or thrice-weekly review of the UDS dashboard would have allowed for more confirmatory testing, along with more immediate clinician collaboration. Most importantly, the outcomes of this project are only presented via descriptive statistics and without the results of any comparison group, making it impossible to draw firm conclusions about this approach compared to standard-care processes.
Conclusions
This quality improvement project has proven to be valuable at VABHHCS and we intend to continue this pharmacist-led process to monitor the UDS dashboard. VABHHCS leadership are also discussing UDS practices more broadly to further enhance patient management. Within the VA, the PMOP coordinator—charged with being the local coordinator of appropriate pain management and opioid safety practices—is well positioned to assume these responsibilities. Outside of the VA, a pain-management clinical pharmacist or any pharmacist embedded within primary care could similarly perform these duties. Previous literature regarding the implementation of clinical dashboards suggests that with the appropriate software engineering teams and infrastructure, this tool could also be feasibly developed and implemented at other health systems relatively quickly.14
Overall, a pharmacist-led process to efficiently monitor a dashboard of discrepant UDS results led to opportunities for collaboration with clinicians and positively impacted confirmatory testing and PDMP monitoring at a rural VA health system.
Acknowledgments
The authors express their gratitude to Patrick Spoutz, PharmD, BCPS, VISN 20 Pharmacist Executive, for introducing and sharing the UDS dashboard with our team.
1. Dowell D, Ragan KR, Jones CM, Baldwin GT, Chou R. CDC Clinical Practice Guideline for Prescribing Opioids for Pain - United States, 2022. MMWR Recomm Rep. 2022;71(3):1-95. doi:10.15585/mmwr.rr7103a1
2. US Department of Defense, US Department of Veterans Affairs. VA/DoD clinical practice guidelines for the use of opioids in the management of chronic pain. Version 4.0. Published 2002. Accessed January 22, 2024. https://www.healthquality.va.gov/guidelines/Pain/cot/VADoDOpioidsCPG.pdf
3. Champion C, Kameg BN. Best practices in benzodiazepine prescribing and management in primary care. Nurse Pract. 2021;46(3):30-36.doi:10.1097/01.NPR.0000733684.24949.19
4. Kale N. Urine drug tests: ordering and interpretation. Am Fam Physician. 2019;99(1):33-39.
5. Gillespie E, Cunningham JM, Indovina KA. Interpretation of the urine drug screen. The Hospitalist. May 2, 2022. Accessed January 19, 2024. https://www.the-hospitalist.org/hospitalist/article/32085/interpreting-diagnostic-tests/interpretation-of-the-urine-drug-screen/
6. Schwebach A, Ball J. Urine drug screening: minimizing false-positives and false-negatives to optimize patient care. US Pharm. 2016;41(8):26-30.
7. Starrels JL, Fox AD, Kunins HV, Cunningham CO. They don’t know what they don’t know: internal medicine residents’ knowledge and confidence in urine drug test interpretation for patients with chronic pain. J Gen Intern Med. 2012;27(11):1521-1527. doi:10.1007/s11606-012-2165-7
8. Chua I, Petrides AK, Schiff GD, et al. Provider misinterpretation, documentation, and follow-up of definitive urine drug testing results. J Gen Intern Med. 2020;35(1):283-290. doi:10.1007/s11606-019-05514-5
9. US Department of Veterans Affairs, Veterans Health Administration. VHA Pain Management, Opioid Safety, and Prescription Drug Monitoring Program (PMOP) National Program Field Roles and Responsibilities Manual. October 2021 (V1).[Source not verified]
10. Dorsch MP, Chen CS, Allen AL, et al. Nationwide implementation of a population management dashboard for monitoring direct oral anticoagulants: insights from the Veterans Affairs Health System. Circ Cardiovasc Qual Outcomes. 2023;16(2):e009256. doi:10.1161/CIRCOUTCOMES.122.009256
11. Hu AM, Pepin MJ, Hashem MG, et al. Development of a specialty medication clinical dashboard to improve tumor necrosis factor-α inhibitor safety and adherence monitoring. Am J Health Syst Pharm. 2022;79(8):683-688. doi:10.1093/ajhp/zxab454
12. Homsted FAE, Magee CE, Nesin N. Population health management in a small health system: impact of controlled substance stewardship in a patient-centered medical home. Am J Health Syst Pharm. 2017;74(18):1468-1475. doi:10.2146/ajhp161032
13. US Department of Veterans Affairs, Veterans Health Administration, Pharmacy Benefits (PBM) Services, Clinical Pharmacy Practice Office. Fact Sheet: CPS Role in Population Health Management. 2019. [Source not verified]
14. Anderson D, Zlateva I, Khatri K, Ciaburri N. Using health information technology to improve adherence to opioid prescribing guidelines in primary care. Clin J Pain. 2015;31(6):573-579. doi:10.1097/AJP.0000000000000177
15. Wang EJ, Helgesen R, Johr CR, Lacko HS, Ashburn MA, Merkel PA. Targeted program in an academic rheumatology practice to improve compliance with opioid prescribing guidelines for the treatment of chronic pain. Arthritis Care Res (Hoboken). 2021;73(10):1425-1429. doi:10.1002/acr.24354
16. Moeller KE, Kissack JC, Atayee RS, Lee KC. Clinical interpretation of urine drug tests: what clinicians need to know about urine drug screens. Mayo Clin Proc. 2017;92(5):774-796. doi:10.1016/j.mayocp.2016.12.007
17. Wisco BE, Marx BP, Wolf EJ, Miller MW, Southwick SM, Pietrzak RH. Posttraumatic stress disorder in the US veteran population: results from the National Health and Resilience in Veterans Study. J Clin Psychiatry. 2014;75(12):1338-46. doi:10.4088/JCP.14m09328
1. Dowell D, Ragan KR, Jones CM, Baldwin GT, Chou R. CDC Clinical Practice Guideline for Prescribing Opioids for Pain - United States, 2022. MMWR Recomm Rep. 2022;71(3):1-95. doi:10.15585/mmwr.rr7103a1
2. US Department of Defense, US Department of Veterans Affairs. VA/DoD clinical practice guidelines for the use of opioids in the management of chronic pain. Version 4.0. Published 2002. Accessed January 22, 2024. https://www.healthquality.va.gov/guidelines/Pain/cot/VADoDOpioidsCPG.pdf
3. Champion C, Kameg BN. Best practices in benzodiazepine prescribing and management in primary care. Nurse Pract. 2021;46(3):30-36.doi:10.1097/01.NPR.0000733684.24949.19
4. Kale N. Urine drug tests: ordering and interpretation. Am Fam Physician. 2019;99(1):33-39.
5. Gillespie E, Cunningham JM, Indovina KA. Interpretation of the urine drug screen. The Hospitalist. May 2, 2022. Accessed January 19, 2024. https://www.the-hospitalist.org/hospitalist/article/32085/interpreting-diagnostic-tests/interpretation-of-the-urine-drug-screen/
6. Schwebach A, Ball J. Urine drug screening: minimizing false-positives and false-negatives to optimize patient care. US Pharm. 2016;41(8):26-30.
7. Starrels JL, Fox AD, Kunins HV, Cunningham CO. They don’t know what they don’t know: internal medicine residents’ knowledge and confidence in urine drug test interpretation for patients with chronic pain. J Gen Intern Med. 2012;27(11):1521-1527. doi:10.1007/s11606-012-2165-7
8. Chua I, Petrides AK, Schiff GD, et al. Provider misinterpretation, documentation, and follow-up of definitive urine drug testing results. J Gen Intern Med. 2020;35(1):283-290. doi:10.1007/s11606-019-05514-5
9. US Department of Veterans Affairs, Veterans Health Administration. VHA Pain Management, Opioid Safety, and Prescription Drug Monitoring Program (PMOP) National Program Field Roles and Responsibilities Manual. October 2021 (V1).[Source not verified]
10. Dorsch MP, Chen CS, Allen AL, et al. Nationwide implementation of a population management dashboard for monitoring direct oral anticoagulants: insights from the Veterans Affairs Health System. Circ Cardiovasc Qual Outcomes. 2023;16(2):e009256. doi:10.1161/CIRCOUTCOMES.122.009256
11. Hu AM, Pepin MJ, Hashem MG, et al. Development of a specialty medication clinical dashboard to improve tumor necrosis factor-α inhibitor safety and adherence monitoring. Am J Health Syst Pharm. 2022;79(8):683-688. doi:10.1093/ajhp/zxab454
12. Homsted FAE, Magee CE, Nesin N. Population health management in a small health system: impact of controlled substance stewardship in a patient-centered medical home. Am J Health Syst Pharm. 2017;74(18):1468-1475. doi:10.2146/ajhp161032
13. US Department of Veterans Affairs, Veterans Health Administration, Pharmacy Benefits (PBM) Services, Clinical Pharmacy Practice Office. Fact Sheet: CPS Role in Population Health Management. 2019. [Source not verified]
14. Anderson D, Zlateva I, Khatri K, Ciaburri N. Using health information technology to improve adherence to opioid prescribing guidelines in primary care. Clin J Pain. 2015;31(6):573-579. doi:10.1097/AJP.0000000000000177
15. Wang EJ, Helgesen R, Johr CR, Lacko HS, Ashburn MA, Merkel PA. Targeted program in an academic rheumatology practice to improve compliance with opioid prescribing guidelines for the treatment of chronic pain. Arthritis Care Res (Hoboken). 2021;73(10):1425-1429. doi:10.1002/acr.24354
16. Moeller KE, Kissack JC, Atayee RS, Lee KC. Clinical interpretation of urine drug tests: what clinicians need to know about urine drug screens. Mayo Clin Proc. 2017;92(5):774-796. doi:10.1016/j.mayocp.2016.12.007
17. Wisco BE, Marx BP, Wolf EJ, Miller MW, Southwick SM, Pietrzak RH. Posttraumatic stress disorder in the US veteran population: results from the National Health and Resilience in Veterans Study. J Clin Psychiatry. 2014;75(12):1338-46. doi:10.4088/JCP.14m09328
Medication-Nonadherent Hypothyroidism Requiring Frequent Primary Care Visits to Achieve Euthyroidism
Nonadherence to medications is an issue across health care. In endocrinology, hypothyroidism, a deficiency of thyroid hormones, is most often treated with levothyroxine and if left untreated can lead to myxedema coma, which can lead to death due to multiorgan dysfunction.1 Therefore, adherence to levothyroxine is very important in preventing fatal complications.
We present the case of a patient with persistent primary hypothyroidism who was suspected to be nonadherent to levothyroxine, although the patient consistently claimed adherence. The patient’s plasma thyrotropin (TSH) level improved to reference range after 6 weeks of weekly primary care clinic visits. After stopping the visits, his plasma TSH level increased again, so 9 more weeks of visits resumed, which again helped bring down his plasma TSH levels.
Case Presentation
A male patient aged 67 years presented to the Dayton Veterans Affairs Medical Center (VAMC) endocrinology clinic for evaluation of thyroid nodules. The patient reported no history of neck irradiation and a physical examination was unremarkable. At that time, laboratory results showed a slightly elevated plasma TSH level of 4.35 uIU/mL (reference range, 0.35-4.00 uIU/mL) and normal free thyroxine (T4) of 1.00 ng/dL (reference range, 0.74-1.46 ng/dL). Later that year, the patient underwent a total thyroidectomy at the Cincinnati VAMC for Hurthle cell variant papillary thyroid carcinoma that was noted on biopsy at the Dayton VAMC. After surgical pathology results were available, the patient started levothyroxine 200 mcg daily, although 224 mcg would have been more appropriate based on his 142 kg weight. Due to a history of arrhythmia, the goal plasma TSH level was 0.10 to 0.50 uIU/mL. The patient subsequently underwent radioactive iodine ablation. After levothyroxine dose adjustments, the patient’s plasma TSH level was noted to be within his target range at 0.28 uIU/mL 3 months postablation.
Over the next 5 years the patient had regular laboratory tests during which his plasma TSH level rose and were typically high despite adjusting levothyroxine doses between 200 mcg and 325 mcg. The patient received counseling on taking the medication in the morning on an empty stomach and waiting at least 1 hour before consuming anything, and he went to many follow-up visits at the Dayton VAMC endocrinology clinic. He reported no vomiting or diarrhea but endorsed weight gain once. The patient also had high free T4 at times and did not take extra levothyroxine before undergoing laboratory tests.
Nonadherence to levothyroxine was suspected, but the patient insisted he was adherent. He received the medication in the mail regularly, generally had 90-day refills unless a dose change was made, used a pill box, and had social support from his son, but he did not use a phone alarm to remind him to take it. A home care nurse made weekly visits to make sure the remaining levothyroxine pill counts were correct; however, the patient continued to have difficulty maintaining daily adherence at home as indicated by the nurse’s pill counts not aligning with the number of pills which should have been left if the patient was talking the pills daily.
The patient was asked to visit a local community-based outpatient clinic (CBOC) weekly (to avoid patient travel time to Dayton VAMC > 1 hour) to check pill counts and assess adherence. The patient went to the CBOC clinic for these visits, during which pill counts indicated much better but not 100% adherence. After 6 weeks of clinic visits, his plasma TSH decreased to 1.01 uIU/mL, which was within the reference range, and the patient stopped coming to the weekly clinic visits (Table). Four months later, the patient's plasma TSH levels increased to 80.72 uIU/mL. Nonadherence to levothyroxine was suspected again. He was asked to resume weekly clinic visits, and the life-threatening effects of hypothyroidism and not taking levothyroxine were discussed with the patient and his son. The patient made CBOC clinic visits for 9 weeks, after which his plasma TSH level was low at 0.23 uIU/mL.
Discussion
There are multiple important causes to consider in patients with persistent hypothyroidism. One is medication nonadherence, which was most likely seen in the patient in this case. Missing even 1 day of levothyroxine can affect TSH and thyroid hormone levels for several days due to the long half-life of the medication.2 Hepp and colleagues found that patients with hypothyroidism were significantly more likely to be nonadherent to levothyroxine if they had comorbid conditions such as type 2 diabetes or were obese.3 Another study of levothyroxine adherence found that the most common reason for missing doses was forgetfulness.4 However, memory and cognition impairments can also be symptoms of hypothyroidism itself; Haskard-Zolnierek and colleagues found a significant association between nonadherence to levothyroxine and self-reported brain fog in patients with hypothyroidism.5
Another cause of persistent hypothyroidism is malabsorption. Absorption of levothyroxine can be affected by intestinal malabsorption due to inflammatory bowel disease, lactose intolerance, or gastrointestinal infection, as well as several foods, drinks (eg, coffee), medications, vitamins, and supplements (eg, proton-pump inhibitors and calcium).2,6 Levothyroxine is absorbed mainly at the jejunum and upper ileum, so any pathologies or ingested items that would directly or indirectly affect absorption at those sites can affect levothyroxine absorption.2
A liquid levothyroxine formulation can help with malabsorption.2 Alternatively, weight gain may lead to a need for increasing the dosage of levothyroxine.2,6 Other factors that can affect TSH levels include Addison disease, dysregulation of the hypothalamic-pituitary-thyroid axis, and TSH heterophile antibodies.2
Research describes methods that have effectively treated hypothyroidism in patients struggling with levothyroxine adherence. Two case reports describe weekly visits for levothyroxine administration successfully treating uncontrolled hypothyroidism.7,8 A meta-analysis found that while weekly levothyroxine tablets led to a higher mean TSH level than daily use, weekly use still led to reference-range TSH levels, suggesting that weekly levothyroxine may be a helpful alternative for nonadherent patients.9 Alternatively, patients taking levothyroxine tablets have been shown to forget to take their medication more frequently compared to those taking the liquid formulation.10,11 Additionally, a study by El Helou and colleagues found that adherence to levothyroxine was significantly improved when patients had endocrinology visits once a month and when the endocrinologist provided information about hypothyroidism.12
Another method that may improve adherence to levothyroxine is telehealth visits. This would be especially helpful for patients who live far from the clinic or do not have the time, transportation, or financial means to visit the clinic for weekly visits to assess medication adherence. Additionally, patients may be afraid of admitting to a health care professional that they are nonadherent. Clinicians must be tactful when asking about adherence to make the patient feel comfortable with admitting to nonadherence if their cognition is not impaired. Then, a patient-led conversation can occur regarding realistic ways the patient feels they can work toward adherence.
To our knowledge, the patient in this case report had no symptoms of intestinal malabsorption, and weight gain was not thought to be the issue, as levothyroxine dosage was adjusted multiple times. His plasma TSH levels returned to reference range after weekly pill count visits for 6 weeks and after weekly pill count visits for 9 weeks. Therefore, nonadherence to levothyroxine was suspected to be the cause of frequently elevated plasma TSH levels despite the patient’s insistence on adherence. While the patient did not report memory issues, cognitive impairments due to hypothyroidism may have been contributing to his probable nonadherence. Additionally, he had comorbidities, such as type 2 diabetes mellitus and obesity, which may have made adherence more difficult.
Levothyroxine was also only prescribed in daily tablet form, so the frequency and formulation may have also contributed to nonadherence. While the home nurse was originally sent to assess the patient’s adherence, the care team could have had the nurse start giving the patient weekly levothyroxine once nonadherence was determined to be a likely issue. The patient’s adherence only improved when he went to the clinic for pill counts but not when the home nurse came to his house weekly; this could be because the patient knew he had to invest the time to physically go to clinic visits for pill checks, motivating him to increase adherence.
Conclusions
This case reports a patient with frequently high plasma TSH levels achieving normalization of plasma TSH levels after weekly medication adherence checks at a primary care clinic. Weekly visits to a clinic seem impractical compared to weekly dosing with a visiting nurse; however, after review of the literature, this may be an approach to consider in the future. This strategy may especially help in cases of persistent abnormal plasma TSH levels in which no etiology can be found other than suspected medication nonadherence. Knowing their medication use will be checked at weekly clinic visits may motivate patients to be adherent.
1. Chaker L, Bianco AC, Jonklaas J, Peeters RP. Hypothyroidism. Lancet. 2017;390(10101):1550-1562. doi:10.1016/S0140-6736(17)30703-1
2. Centanni M, Benvenga S, Sachmechi I. Diagnosis and management of treatment-refractory hypothyroidism: an expert consensus report. J Endocrinol Invest. 2017;40(12):1289-1301. doi:10.1007/s40618-017-0706-y
3. Hepp Z, Lage MJ, Espaillat R, Gossain VV. The association between adherence to levothyroxine and economic and clinical outcomes in patients with hypothyroidism in the US. J Med Econ. 2018;21(9):912-919. doi:10.1080/13696998.2018.1484749
4. Shakya Shrestha S, Risal K, Shrestha R, Bhatta RD. Medication Adherence to Levothyroxine Therapy among Hypothyroid Patients and their Clinical Outcomes with Special Reference to Thyroid Function Parameters. Kathmandu Univ Med J (KUMJ). 2018;16(62):129-137.
5. Haskard-Zolnierek K, Wilson C, Pruin J, Deason R, Howard K. The Relationship Between Brain Fog and Medication Adherence for Individuals With Hypothyroidism. Clin Nurs Res. 2022;31(3):445-452. doi:10.1177/10547738211038127
6. McNally LJ, Ofiaeli CI, Oyibo SO. Treatment-refractory hypothyroidism. BMJ. 2019;364:l579. Published 2019 Feb 25. doi:10.1136/bmj.l579
7. Nakano Y, Hashimoto K, Ohkiba N, et al. A Case of Refractory Hypothyroidism due to Poor Compliance Treated with the Weekly Intravenous and Oral Levothyroxine Administration. Case Rep Endocrinol. 2019;2019:5986014. Published 2019 Feb 5. doi:10.1155/2019/5986014
8. Kiran Z, Shaikh KS, Fatima N, Tariq N, Baloch AA. Levothyroxine absorption test followed by directly observed treatment on an outpatient basis to address long-term high TSH levels in a hypothyroid patient: a case report. J Med Case Rep. 2023;17(1):24. Published 2023 Jan 25. doi:10.1186/s13256-023-03760-0
9. Chiu HH, Larrazabal R Jr, Uy AB, Jimeno C. Weekly Versus Daily Levothyroxine Tablet Replacement in Adults with Hypothyroidism: A Meta-Analysis. J ASEAN Fed Endocr Soc. 2021;36(2):156-160. doi:10.15605/jafes.036.02.07
10. Cappelli C, Castello R, Marini F, et al. Adherence to Levothyroxine Treatment Among Patients With Hypothyroidism: A Northeastern Italian Survey. Front Endocrinol (Lausanne). 2018;9:699. Published 2018 Nov 23. doi:10.3389/fendo.2018.00699
11. Bocale R, Desideri G, Barini A, et al. Long-Term Adherence to Levothyroxine Replacement Therapy in Thyroidectomized Patients. J Clin Med. 2022;11(15):4296. Published 2022 Jul 24. doi:10.3390/jcm11154296
12. El Helou S, Hallit S, Awada S, et al. Adherence to levothyroxine among patients with hypothyroidism in Lebanon. East Mediterr Health J. 2019;25(3):149-159. Published 2019 Apr 25. doi:10.26719/emhj.18.022
Nonadherence to medications is an issue across health care. In endocrinology, hypothyroidism, a deficiency of thyroid hormones, is most often treated with levothyroxine and if left untreated can lead to myxedema coma, which can lead to death due to multiorgan dysfunction.1 Therefore, adherence to levothyroxine is very important in preventing fatal complications.
We present the case of a patient with persistent primary hypothyroidism who was suspected to be nonadherent to levothyroxine, although the patient consistently claimed adherence. The patient’s plasma thyrotropin (TSH) level improved to reference range after 6 weeks of weekly primary care clinic visits. After stopping the visits, his plasma TSH level increased again, so 9 more weeks of visits resumed, which again helped bring down his plasma TSH levels.
Case Presentation
A male patient aged 67 years presented to the Dayton Veterans Affairs Medical Center (VAMC) endocrinology clinic for evaluation of thyroid nodules. The patient reported no history of neck irradiation and a physical examination was unremarkable. At that time, laboratory results showed a slightly elevated plasma TSH level of 4.35 uIU/mL (reference range, 0.35-4.00 uIU/mL) and normal free thyroxine (T4) of 1.00 ng/dL (reference range, 0.74-1.46 ng/dL). Later that year, the patient underwent a total thyroidectomy at the Cincinnati VAMC for Hurthle cell variant papillary thyroid carcinoma that was noted on biopsy at the Dayton VAMC. After surgical pathology results were available, the patient started levothyroxine 200 mcg daily, although 224 mcg would have been more appropriate based on his 142 kg weight. Due to a history of arrhythmia, the goal plasma TSH level was 0.10 to 0.50 uIU/mL. The patient subsequently underwent radioactive iodine ablation. After levothyroxine dose adjustments, the patient’s plasma TSH level was noted to be within his target range at 0.28 uIU/mL 3 months postablation.
Over the next 5 years the patient had regular laboratory tests during which his plasma TSH level rose and were typically high despite adjusting levothyroxine doses between 200 mcg and 325 mcg. The patient received counseling on taking the medication in the morning on an empty stomach and waiting at least 1 hour before consuming anything, and he went to many follow-up visits at the Dayton VAMC endocrinology clinic. He reported no vomiting or diarrhea but endorsed weight gain once. The patient also had high free T4 at times and did not take extra levothyroxine before undergoing laboratory tests.
Nonadherence to levothyroxine was suspected, but the patient insisted he was adherent. He received the medication in the mail regularly, generally had 90-day refills unless a dose change was made, used a pill box, and had social support from his son, but he did not use a phone alarm to remind him to take it. A home care nurse made weekly visits to make sure the remaining levothyroxine pill counts were correct; however, the patient continued to have difficulty maintaining daily adherence at home as indicated by the nurse’s pill counts not aligning with the number of pills which should have been left if the patient was talking the pills daily.
The patient was asked to visit a local community-based outpatient clinic (CBOC) weekly (to avoid patient travel time to Dayton VAMC > 1 hour) to check pill counts and assess adherence. The patient went to the CBOC clinic for these visits, during which pill counts indicated much better but not 100% adherence. After 6 weeks of clinic visits, his plasma TSH decreased to 1.01 uIU/mL, which was within the reference range, and the patient stopped coming to the weekly clinic visits (Table). Four months later, the patient's plasma TSH levels increased to 80.72 uIU/mL. Nonadherence to levothyroxine was suspected again. He was asked to resume weekly clinic visits, and the life-threatening effects of hypothyroidism and not taking levothyroxine were discussed with the patient and his son. The patient made CBOC clinic visits for 9 weeks, after which his plasma TSH level was low at 0.23 uIU/mL.
Discussion
There are multiple important causes to consider in patients with persistent hypothyroidism. One is medication nonadherence, which was most likely seen in the patient in this case. Missing even 1 day of levothyroxine can affect TSH and thyroid hormone levels for several days due to the long half-life of the medication.2 Hepp and colleagues found that patients with hypothyroidism were significantly more likely to be nonadherent to levothyroxine if they had comorbid conditions such as type 2 diabetes or were obese.3 Another study of levothyroxine adherence found that the most common reason for missing doses was forgetfulness.4 However, memory and cognition impairments can also be symptoms of hypothyroidism itself; Haskard-Zolnierek and colleagues found a significant association between nonadherence to levothyroxine and self-reported brain fog in patients with hypothyroidism.5
Another cause of persistent hypothyroidism is malabsorption. Absorption of levothyroxine can be affected by intestinal malabsorption due to inflammatory bowel disease, lactose intolerance, or gastrointestinal infection, as well as several foods, drinks (eg, coffee), medications, vitamins, and supplements (eg, proton-pump inhibitors and calcium).2,6 Levothyroxine is absorbed mainly at the jejunum and upper ileum, so any pathologies or ingested items that would directly or indirectly affect absorption at those sites can affect levothyroxine absorption.2
A liquid levothyroxine formulation can help with malabsorption.2 Alternatively, weight gain may lead to a need for increasing the dosage of levothyroxine.2,6 Other factors that can affect TSH levels include Addison disease, dysregulation of the hypothalamic-pituitary-thyroid axis, and TSH heterophile antibodies.2
Research describes methods that have effectively treated hypothyroidism in patients struggling with levothyroxine adherence. Two case reports describe weekly visits for levothyroxine administration successfully treating uncontrolled hypothyroidism.7,8 A meta-analysis found that while weekly levothyroxine tablets led to a higher mean TSH level than daily use, weekly use still led to reference-range TSH levels, suggesting that weekly levothyroxine may be a helpful alternative for nonadherent patients.9 Alternatively, patients taking levothyroxine tablets have been shown to forget to take their medication more frequently compared to those taking the liquid formulation.10,11 Additionally, a study by El Helou and colleagues found that adherence to levothyroxine was significantly improved when patients had endocrinology visits once a month and when the endocrinologist provided information about hypothyroidism.12
Another method that may improve adherence to levothyroxine is telehealth visits. This would be especially helpful for patients who live far from the clinic or do not have the time, transportation, or financial means to visit the clinic for weekly visits to assess medication adherence. Additionally, patients may be afraid of admitting to a health care professional that they are nonadherent. Clinicians must be tactful when asking about adherence to make the patient feel comfortable with admitting to nonadherence if their cognition is not impaired. Then, a patient-led conversation can occur regarding realistic ways the patient feels they can work toward adherence.
To our knowledge, the patient in this case report had no symptoms of intestinal malabsorption, and weight gain was not thought to be the issue, as levothyroxine dosage was adjusted multiple times. His plasma TSH levels returned to reference range after weekly pill count visits for 6 weeks and after weekly pill count visits for 9 weeks. Therefore, nonadherence to levothyroxine was suspected to be the cause of frequently elevated plasma TSH levels despite the patient’s insistence on adherence. While the patient did not report memory issues, cognitive impairments due to hypothyroidism may have been contributing to his probable nonadherence. Additionally, he had comorbidities, such as type 2 diabetes mellitus and obesity, which may have made adherence more difficult.
Levothyroxine was also only prescribed in daily tablet form, so the frequency and formulation may have also contributed to nonadherence. While the home nurse was originally sent to assess the patient’s adherence, the care team could have had the nurse start giving the patient weekly levothyroxine once nonadherence was determined to be a likely issue. The patient’s adherence only improved when he went to the clinic for pill counts but not when the home nurse came to his house weekly; this could be because the patient knew he had to invest the time to physically go to clinic visits for pill checks, motivating him to increase adherence.
Conclusions
This case reports a patient with frequently high plasma TSH levels achieving normalization of plasma TSH levels after weekly medication adherence checks at a primary care clinic. Weekly visits to a clinic seem impractical compared to weekly dosing with a visiting nurse; however, after review of the literature, this may be an approach to consider in the future. This strategy may especially help in cases of persistent abnormal plasma TSH levels in which no etiology can be found other than suspected medication nonadherence. Knowing their medication use will be checked at weekly clinic visits may motivate patients to be adherent.
Nonadherence to medications is an issue across health care. In endocrinology, hypothyroidism, a deficiency of thyroid hormones, is most often treated with levothyroxine and if left untreated can lead to myxedema coma, which can lead to death due to multiorgan dysfunction.1 Therefore, adherence to levothyroxine is very important in preventing fatal complications.
We present the case of a patient with persistent primary hypothyroidism who was suspected to be nonadherent to levothyroxine, although the patient consistently claimed adherence. The patient’s plasma thyrotropin (TSH) level improved to reference range after 6 weeks of weekly primary care clinic visits. After stopping the visits, his plasma TSH level increased again, so 9 more weeks of visits resumed, which again helped bring down his plasma TSH levels.
Case Presentation
A male patient aged 67 years presented to the Dayton Veterans Affairs Medical Center (VAMC) endocrinology clinic for evaluation of thyroid nodules. The patient reported no history of neck irradiation and a physical examination was unremarkable. At that time, laboratory results showed a slightly elevated plasma TSH level of 4.35 uIU/mL (reference range, 0.35-4.00 uIU/mL) and normal free thyroxine (T4) of 1.00 ng/dL (reference range, 0.74-1.46 ng/dL). Later that year, the patient underwent a total thyroidectomy at the Cincinnati VAMC for Hurthle cell variant papillary thyroid carcinoma that was noted on biopsy at the Dayton VAMC. After surgical pathology results were available, the patient started levothyroxine 200 mcg daily, although 224 mcg would have been more appropriate based on his 142 kg weight. Due to a history of arrhythmia, the goal plasma TSH level was 0.10 to 0.50 uIU/mL. The patient subsequently underwent radioactive iodine ablation. After levothyroxine dose adjustments, the patient’s plasma TSH level was noted to be within his target range at 0.28 uIU/mL 3 months postablation.
Over the next 5 years the patient had regular laboratory tests during which his plasma TSH level rose and were typically high despite adjusting levothyroxine doses between 200 mcg and 325 mcg. The patient received counseling on taking the medication in the morning on an empty stomach and waiting at least 1 hour before consuming anything, and he went to many follow-up visits at the Dayton VAMC endocrinology clinic. He reported no vomiting or diarrhea but endorsed weight gain once. The patient also had high free T4 at times and did not take extra levothyroxine before undergoing laboratory tests.
Nonadherence to levothyroxine was suspected, but the patient insisted he was adherent. He received the medication in the mail regularly, generally had 90-day refills unless a dose change was made, used a pill box, and had social support from his son, but he did not use a phone alarm to remind him to take it. A home care nurse made weekly visits to make sure the remaining levothyroxine pill counts were correct; however, the patient continued to have difficulty maintaining daily adherence at home as indicated by the nurse’s pill counts not aligning with the number of pills which should have been left if the patient was talking the pills daily.
The patient was asked to visit a local community-based outpatient clinic (CBOC) weekly (to avoid patient travel time to Dayton VAMC > 1 hour) to check pill counts and assess adherence. The patient went to the CBOC clinic for these visits, during which pill counts indicated much better but not 100% adherence. After 6 weeks of clinic visits, his plasma TSH decreased to 1.01 uIU/mL, which was within the reference range, and the patient stopped coming to the weekly clinic visits (Table). Four months later, the patient's plasma TSH levels increased to 80.72 uIU/mL. Nonadherence to levothyroxine was suspected again. He was asked to resume weekly clinic visits, and the life-threatening effects of hypothyroidism and not taking levothyroxine were discussed with the patient and his son. The patient made CBOC clinic visits for 9 weeks, after which his plasma TSH level was low at 0.23 uIU/mL.
Discussion
There are multiple important causes to consider in patients with persistent hypothyroidism. One is medication nonadherence, which was most likely seen in the patient in this case. Missing even 1 day of levothyroxine can affect TSH and thyroid hormone levels for several days due to the long half-life of the medication.2 Hepp and colleagues found that patients with hypothyroidism were significantly more likely to be nonadherent to levothyroxine if they had comorbid conditions such as type 2 diabetes or were obese.3 Another study of levothyroxine adherence found that the most common reason for missing doses was forgetfulness.4 However, memory and cognition impairments can also be symptoms of hypothyroidism itself; Haskard-Zolnierek and colleagues found a significant association between nonadherence to levothyroxine and self-reported brain fog in patients with hypothyroidism.5
Another cause of persistent hypothyroidism is malabsorption. Absorption of levothyroxine can be affected by intestinal malabsorption due to inflammatory bowel disease, lactose intolerance, or gastrointestinal infection, as well as several foods, drinks (eg, coffee), medications, vitamins, and supplements (eg, proton-pump inhibitors and calcium).2,6 Levothyroxine is absorbed mainly at the jejunum and upper ileum, so any pathologies or ingested items that would directly or indirectly affect absorption at those sites can affect levothyroxine absorption.2
A liquid levothyroxine formulation can help with malabsorption.2 Alternatively, weight gain may lead to a need for increasing the dosage of levothyroxine.2,6 Other factors that can affect TSH levels include Addison disease, dysregulation of the hypothalamic-pituitary-thyroid axis, and TSH heterophile antibodies.2
Research describes methods that have effectively treated hypothyroidism in patients struggling with levothyroxine adherence. Two case reports describe weekly visits for levothyroxine administration successfully treating uncontrolled hypothyroidism.7,8 A meta-analysis found that while weekly levothyroxine tablets led to a higher mean TSH level than daily use, weekly use still led to reference-range TSH levels, suggesting that weekly levothyroxine may be a helpful alternative for nonadherent patients.9 Alternatively, patients taking levothyroxine tablets have been shown to forget to take their medication more frequently compared to those taking the liquid formulation.10,11 Additionally, a study by El Helou and colleagues found that adherence to levothyroxine was significantly improved when patients had endocrinology visits once a month and when the endocrinologist provided information about hypothyroidism.12
Another method that may improve adherence to levothyroxine is telehealth visits. This would be especially helpful for patients who live far from the clinic or do not have the time, transportation, or financial means to visit the clinic for weekly visits to assess medication adherence. Additionally, patients may be afraid of admitting to a health care professional that they are nonadherent. Clinicians must be tactful when asking about adherence to make the patient feel comfortable with admitting to nonadherence if their cognition is not impaired. Then, a patient-led conversation can occur regarding realistic ways the patient feels they can work toward adherence.
To our knowledge, the patient in this case report had no symptoms of intestinal malabsorption, and weight gain was not thought to be the issue, as levothyroxine dosage was adjusted multiple times. His plasma TSH levels returned to reference range after weekly pill count visits for 6 weeks and after weekly pill count visits for 9 weeks. Therefore, nonadherence to levothyroxine was suspected to be the cause of frequently elevated plasma TSH levels despite the patient’s insistence on adherence. While the patient did not report memory issues, cognitive impairments due to hypothyroidism may have been contributing to his probable nonadherence. Additionally, he had comorbidities, such as type 2 diabetes mellitus and obesity, which may have made adherence more difficult.
Levothyroxine was also only prescribed in daily tablet form, so the frequency and formulation may have also contributed to nonadherence. While the home nurse was originally sent to assess the patient’s adherence, the care team could have had the nurse start giving the patient weekly levothyroxine once nonadherence was determined to be a likely issue. The patient’s adherence only improved when he went to the clinic for pill counts but not when the home nurse came to his house weekly; this could be because the patient knew he had to invest the time to physically go to clinic visits for pill checks, motivating him to increase adherence.
Conclusions
This case reports a patient with frequently high plasma TSH levels achieving normalization of plasma TSH levels after weekly medication adherence checks at a primary care clinic. Weekly visits to a clinic seem impractical compared to weekly dosing with a visiting nurse; however, after review of the literature, this may be an approach to consider in the future. This strategy may especially help in cases of persistent abnormal plasma TSH levels in which no etiology can be found other than suspected medication nonadherence. Knowing their medication use will be checked at weekly clinic visits may motivate patients to be adherent.
1. Chaker L, Bianco AC, Jonklaas J, Peeters RP. Hypothyroidism. Lancet. 2017;390(10101):1550-1562. doi:10.1016/S0140-6736(17)30703-1
2. Centanni M, Benvenga S, Sachmechi I. Diagnosis and management of treatment-refractory hypothyroidism: an expert consensus report. J Endocrinol Invest. 2017;40(12):1289-1301. doi:10.1007/s40618-017-0706-y
3. Hepp Z, Lage MJ, Espaillat R, Gossain VV. The association between adherence to levothyroxine and economic and clinical outcomes in patients with hypothyroidism in the US. J Med Econ. 2018;21(9):912-919. doi:10.1080/13696998.2018.1484749
4. Shakya Shrestha S, Risal K, Shrestha R, Bhatta RD. Medication Adherence to Levothyroxine Therapy among Hypothyroid Patients and their Clinical Outcomes with Special Reference to Thyroid Function Parameters. Kathmandu Univ Med J (KUMJ). 2018;16(62):129-137.
5. Haskard-Zolnierek K, Wilson C, Pruin J, Deason R, Howard K. The Relationship Between Brain Fog and Medication Adherence for Individuals With Hypothyroidism. Clin Nurs Res. 2022;31(3):445-452. doi:10.1177/10547738211038127
6. McNally LJ, Ofiaeli CI, Oyibo SO. Treatment-refractory hypothyroidism. BMJ. 2019;364:l579. Published 2019 Feb 25. doi:10.1136/bmj.l579
7. Nakano Y, Hashimoto K, Ohkiba N, et al. A Case of Refractory Hypothyroidism due to Poor Compliance Treated with the Weekly Intravenous and Oral Levothyroxine Administration. Case Rep Endocrinol. 2019;2019:5986014. Published 2019 Feb 5. doi:10.1155/2019/5986014
8. Kiran Z, Shaikh KS, Fatima N, Tariq N, Baloch AA. Levothyroxine absorption test followed by directly observed treatment on an outpatient basis to address long-term high TSH levels in a hypothyroid patient: a case report. J Med Case Rep. 2023;17(1):24. Published 2023 Jan 25. doi:10.1186/s13256-023-03760-0
9. Chiu HH, Larrazabal R Jr, Uy AB, Jimeno C. Weekly Versus Daily Levothyroxine Tablet Replacement in Adults with Hypothyroidism: A Meta-Analysis. J ASEAN Fed Endocr Soc. 2021;36(2):156-160. doi:10.15605/jafes.036.02.07
10. Cappelli C, Castello R, Marini F, et al. Adherence to Levothyroxine Treatment Among Patients With Hypothyroidism: A Northeastern Italian Survey. Front Endocrinol (Lausanne). 2018;9:699. Published 2018 Nov 23. doi:10.3389/fendo.2018.00699
11. Bocale R, Desideri G, Barini A, et al. Long-Term Adherence to Levothyroxine Replacement Therapy in Thyroidectomized Patients. J Clin Med. 2022;11(15):4296. Published 2022 Jul 24. doi:10.3390/jcm11154296
12. El Helou S, Hallit S, Awada S, et al. Adherence to levothyroxine among patients with hypothyroidism in Lebanon. East Mediterr Health J. 2019;25(3):149-159. Published 2019 Apr 25. doi:10.26719/emhj.18.022
1. Chaker L, Bianco AC, Jonklaas J, Peeters RP. Hypothyroidism. Lancet. 2017;390(10101):1550-1562. doi:10.1016/S0140-6736(17)30703-1
2. Centanni M, Benvenga S, Sachmechi I. Diagnosis and management of treatment-refractory hypothyroidism: an expert consensus report. J Endocrinol Invest. 2017;40(12):1289-1301. doi:10.1007/s40618-017-0706-y
3. Hepp Z, Lage MJ, Espaillat R, Gossain VV. The association between adherence to levothyroxine and economic and clinical outcomes in patients with hypothyroidism in the US. J Med Econ. 2018;21(9):912-919. doi:10.1080/13696998.2018.1484749
4. Shakya Shrestha S, Risal K, Shrestha R, Bhatta RD. Medication Adherence to Levothyroxine Therapy among Hypothyroid Patients and their Clinical Outcomes with Special Reference to Thyroid Function Parameters. Kathmandu Univ Med J (KUMJ). 2018;16(62):129-137.
5. Haskard-Zolnierek K, Wilson C, Pruin J, Deason R, Howard K. The Relationship Between Brain Fog and Medication Adherence for Individuals With Hypothyroidism. Clin Nurs Res. 2022;31(3):445-452. doi:10.1177/10547738211038127
6. McNally LJ, Ofiaeli CI, Oyibo SO. Treatment-refractory hypothyroidism. BMJ. 2019;364:l579. Published 2019 Feb 25. doi:10.1136/bmj.l579
7. Nakano Y, Hashimoto K, Ohkiba N, et al. A Case of Refractory Hypothyroidism due to Poor Compliance Treated with the Weekly Intravenous and Oral Levothyroxine Administration. Case Rep Endocrinol. 2019;2019:5986014. Published 2019 Feb 5. doi:10.1155/2019/5986014
8. Kiran Z, Shaikh KS, Fatima N, Tariq N, Baloch AA. Levothyroxine absorption test followed by directly observed treatment on an outpatient basis to address long-term high TSH levels in a hypothyroid patient: a case report. J Med Case Rep. 2023;17(1):24. Published 2023 Jan 25. doi:10.1186/s13256-023-03760-0
9. Chiu HH, Larrazabal R Jr, Uy AB, Jimeno C. Weekly Versus Daily Levothyroxine Tablet Replacement in Adults with Hypothyroidism: A Meta-Analysis. J ASEAN Fed Endocr Soc. 2021;36(2):156-160. doi:10.15605/jafes.036.02.07
10. Cappelli C, Castello R, Marini F, et al. Adherence to Levothyroxine Treatment Among Patients With Hypothyroidism: A Northeastern Italian Survey. Front Endocrinol (Lausanne). 2018;9:699. Published 2018 Nov 23. doi:10.3389/fendo.2018.00699
11. Bocale R, Desideri G, Barini A, et al. Long-Term Adherence to Levothyroxine Replacement Therapy in Thyroidectomized Patients. J Clin Med. 2022;11(15):4296. Published 2022 Jul 24. doi:10.3390/jcm11154296
12. El Helou S, Hallit S, Awada S, et al. Adherence to levothyroxine among patients with hypothyroidism in Lebanon. East Mediterr Health J. 2019;25(3):149-159. Published 2019 Apr 25. doi:10.26719/emhj.18.022
Patient-Reported Outcomes Predict Mortality in Cutaneous Chronic GVHD
. Independent of potential confounders, these PROs moreover predicted non-relapse mortality for all three disease subtypes, making PROs potentially useful adjuncts for risk stratification and treatment decisions, the study authors said.
“These two findings highlight the importance of patient-reported outcomes in measuring this disease,” lead author Emily Baumrin, MD, MSCE, assistant professor of dermatology and medicine at the University of Pennsylvania, Philadelphia, told this news organization. The study was published online February 28 in JAMA Dermatology.
Symptoms and QOL
The investigators monitored 436 patients from the Chronic GVHD Consortium until December 2020. The Lee Symptom Scale (LSS) skin subscale was used to evaluate symptom burden and the Functional Assessment of Cancer Therapy–Bone Marrow Transplantation (FACT-BMT) was used to measure quality of life.
Patients with sclerotic GVHD and combination disease at diagnosis had significantly worse median LSS scores than did those with epidermal disease (25, 35, and 20 points, respectively; P = .01). Patients with sclerotic disease had worse median FACT-BMT scores versus those with epidermal involvement (104 versus 109 points, respectively; P = .08).
Although these scores improved with all skin subtypes, LSS skin subscale and FACT-BMT scores remained significantly worse (by 9.0 points and 6.1 points, respectively) for patients with combination and sclerotic disease versus those with epidermal disease after adjusting for potential confounders.
Regarding mortality, every 7-point worsening (clinically meaningful difference) in FACT-BMT score at diagnosis of skin chronic GVHD conferred 9.1% increases in odds of both all-cause mortality and non-relapse mortality, after adjustment for factors such as age and sex. Likewise, for every 11 points worsening (clinically meaningful difference) in LSS skin subscale scores at diagnosis, researchers observed odds increases of 10% in all-cause mortality and 16.4% in non-relapse mortality.
Because patients with combination disease had only slightly more epidermal body surface area (BSA) involvement but significantly higher symptom burden than the other subtypes, the authors added, combination disease may represent a distinct phenotype. “Since we’ve also shown that the severity of patient-reported outcomes is associated with mortality,” Dr. Baumrin said in the interview, “perhaps these patients are at the highest risk of mortality as well.”
A growing population
Although many might think of chronic GVHD as rare, she noted, the number of allogeneic hematopoietic cell transplant (HCT) survivors living in the United States is growing. In a modeling study published in October of 2013 in Biology of Blood and Marrow Transplantation, authors predicted that by 2030, this figure will reach 502,000 — about half of whom will develop chronic GVHD, she said.
With more HCTs being performed each year and ongoing improvements in supportive care, patients are living longer post transplant. “Therefore, many transplant survivors are being taken care of in the community outside of transplant centers.”
Accordingly, Dr. Baumrin said, study findings are relevant to dermatologists in academic and transplant centers and the community who provide skin cancer screenings or other dermatologic care for transplant recipients. “Upon diagnosis of chronic GVHD, the evaluation of disease burden by patient-reported outcome measures may assist in assessing disease severity and response to treatments over time — and to stratify patients at higher risk for mortality and communicate that back to transplant physicians.”
Incorporating PROs into clinical practice might prove especially helpful for patients with sclerotic chronic cutaneous GVHD. Currently, clinicians assess cutaneous GVHD clinically, using parameters including skin thickness. The National Institutes of Health (NIH) Skin Score, used in clinical trials, also measures BSA.
“The issue with sclerosis is, it’s hard to determine clinical severity based on physical examination alone,” Dr. Baumrin said. It can be difficult to quantify skin thickness and changes over time. “So it’s hard to detect improvements, which are often slow. Patient-reported outcome measures may be a more sensitive way to detect response to treatment than our clinical assessments, which are often crude for sclerotic disease.”
In a secondary analysis of the phase 2 clinical trial of belumosudil, a treatment for chronic GVHD, published in October 2022 in Transplantation and Cellular Therapy, response rate was around 30% measured by NIH Skin Score and 77% by PROs. “Our clinical examination in sclerotic type disease falls short in terms of determining therapeutic benefit. PROs might complement those clinical measures,” she said.
Future research will involve determining and validating which PROs matter most clinically and to patients, added Dr. Baumrin. Although widely used in evaluating transplant patients, LSS skin subscale and FACT-BMT scores may not represent patients’ experience of living with cutaneous chronic GVHD as effectively as might other tools such as the Dermatology Life Quality Index (DLQI) or Patient-Reported Outcomes Measurement Information System (PROMIS) measures, she explained.
Study strengths included authors’ use of well-validated PROs rather than novel unvalidated measures, Sandra A. Mitchell, PhD, CRNP, of the National Cancer Institute, Rockville, Maryland, and Edward W. Cowen, MD, MHSc, of the Dermatology Branch at the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS), Bethesda, Maryland, wrote in an accompanying editorial in JAMA Dermatology. However, they added, incorporating causes of death might have revealed that the excess mortality associated with sclerotic disease stemmed at least partly from adverse effects of prolonged immunosuppression, particularly infection.
If future studies establish this to be the case, said Dr. Baumrin, reducing immunosuppression might be warranted for these patients. “And if death is primarily due to chronic GVHD itself, maybe we should treat more aggressively. PROs can help guide this decision.”
The study was supported by the NIH/NIAMS and the University of Pennsylvania. Dr. Baumrin and three coauthors report no relevant financial relationships; other authors had disclosures related to several pharmaceutical companies. Dr. Mitchell and Dr. Cowen had no disclosures.
. Independent of potential confounders, these PROs moreover predicted non-relapse mortality for all three disease subtypes, making PROs potentially useful adjuncts for risk stratification and treatment decisions, the study authors said.
“These two findings highlight the importance of patient-reported outcomes in measuring this disease,” lead author Emily Baumrin, MD, MSCE, assistant professor of dermatology and medicine at the University of Pennsylvania, Philadelphia, told this news organization. The study was published online February 28 in JAMA Dermatology.
Symptoms and QOL
The investigators monitored 436 patients from the Chronic GVHD Consortium until December 2020. The Lee Symptom Scale (LSS) skin subscale was used to evaluate symptom burden and the Functional Assessment of Cancer Therapy–Bone Marrow Transplantation (FACT-BMT) was used to measure quality of life.
Patients with sclerotic GVHD and combination disease at diagnosis had significantly worse median LSS scores than did those with epidermal disease (25, 35, and 20 points, respectively; P = .01). Patients with sclerotic disease had worse median FACT-BMT scores versus those with epidermal involvement (104 versus 109 points, respectively; P = .08).
Although these scores improved with all skin subtypes, LSS skin subscale and FACT-BMT scores remained significantly worse (by 9.0 points and 6.1 points, respectively) for patients with combination and sclerotic disease versus those with epidermal disease after adjusting for potential confounders.
Regarding mortality, every 7-point worsening (clinically meaningful difference) in FACT-BMT score at diagnosis of skin chronic GVHD conferred 9.1% increases in odds of both all-cause mortality and non-relapse mortality, after adjustment for factors such as age and sex. Likewise, for every 11 points worsening (clinically meaningful difference) in LSS skin subscale scores at diagnosis, researchers observed odds increases of 10% in all-cause mortality and 16.4% in non-relapse mortality.
Because patients with combination disease had only slightly more epidermal body surface area (BSA) involvement but significantly higher symptom burden than the other subtypes, the authors added, combination disease may represent a distinct phenotype. “Since we’ve also shown that the severity of patient-reported outcomes is associated with mortality,” Dr. Baumrin said in the interview, “perhaps these patients are at the highest risk of mortality as well.”
A growing population
Although many might think of chronic GVHD as rare, she noted, the number of allogeneic hematopoietic cell transplant (HCT) survivors living in the United States is growing. In a modeling study published in October of 2013 in Biology of Blood and Marrow Transplantation, authors predicted that by 2030, this figure will reach 502,000 — about half of whom will develop chronic GVHD, she said.
With more HCTs being performed each year and ongoing improvements in supportive care, patients are living longer post transplant. “Therefore, many transplant survivors are being taken care of in the community outside of transplant centers.”
Accordingly, Dr. Baumrin said, study findings are relevant to dermatologists in academic and transplant centers and the community who provide skin cancer screenings or other dermatologic care for transplant recipients. “Upon diagnosis of chronic GVHD, the evaluation of disease burden by patient-reported outcome measures may assist in assessing disease severity and response to treatments over time — and to stratify patients at higher risk for mortality and communicate that back to transplant physicians.”
Incorporating PROs into clinical practice might prove especially helpful for patients with sclerotic chronic cutaneous GVHD. Currently, clinicians assess cutaneous GVHD clinically, using parameters including skin thickness. The National Institutes of Health (NIH) Skin Score, used in clinical trials, also measures BSA.
“The issue with sclerosis is, it’s hard to determine clinical severity based on physical examination alone,” Dr. Baumrin said. It can be difficult to quantify skin thickness and changes over time. “So it’s hard to detect improvements, which are often slow. Patient-reported outcome measures may be a more sensitive way to detect response to treatment than our clinical assessments, which are often crude for sclerotic disease.”
In a secondary analysis of the phase 2 clinical trial of belumosudil, a treatment for chronic GVHD, published in October 2022 in Transplantation and Cellular Therapy, response rate was around 30% measured by NIH Skin Score and 77% by PROs. “Our clinical examination in sclerotic type disease falls short in terms of determining therapeutic benefit. PROs might complement those clinical measures,” she said.
Future research will involve determining and validating which PROs matter most clinically and to patients, added Dr. Baumrin. Although widely used in evaluating transplant patients, LSS skin subscale and FACT-BMT scores may not represent patients’ experience of living with cutaneous chronic GVHD as effectively as might other tools such as the Dermatology Life Quality Index (DLQI) or Patient-Reported Outcomes Measurement Information System (PROMIS) measures, she explained.
Study strengths included authors’ use of well-validated PROs rather than novel unvalidated measures, Sandra A. Mitchell, PhD, CRNP, of the National Cancer Institute, Rockville, Maryland, and Edward W. Cowen, MD, MHSc, of the Dermatology Branch at the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS), Bethesda, Maryland, wrote in an accompanying editorial in JAMA Dermatology. However, they added, incorporating causes of death might have revealed that the excess mortality associated with sclerotic disease stemmed at least partly from adverse effects of prolonged immunosuppression, particularly infection.
If future studies establish this to be the case, said Dr. Baumrin, reducing immunosuppression might be warranted for these patients. “And if death is primarily due to chronic GVHD itself, maybe we should treat more aggressively. PROs can help guide this decision.”
The study was supported by the NIH/NIAMS and the University of Pennsylvania. Dr. Baumrin and three coauthors report no relevant financial relationships; other authors had disclosures related to several pharmaceutical companies. Dr. Mitchell and Dr. Cowen had no disclosures.
. Independent of potential confounders, these PROs moreover predicted non-relapse mortality for all three disease subtypes, making PROs potentially useful adjuncts for risk stratification and treatment decisions, the study authors said.
“These two findings highlight the importance of patient-reported outcomes in measuring this disease,” lead author Emily Baumrin, MD, MSCE, assistant professor of dermatology and medicine at the University of Pennsylvania, Philadelphia, told this news organization. The study was published online February 28 in JAMA Dermatology.
Symptoms and QOL
The investigators monitored 436 patients from the Chronic GVHD Consortium until December 2020. The Lee Symptom Scale (LSS) skin subscale was used to evaluate symptom burden and the Functional Assessment of Cancer Therapy–Bone Marrow Transplantation (FACT-BMT) was used to measure quality of life.
Patients with sclerotic GVHD and combination disease at diagnosis had significantly worse median LSS scores than did those with epidermal disease (25, 35, and 20 points, respectively; P = .01). Patients with sclerotic disease had worse median FACT-BMT scores versus those with epidermal involvement (104 versus 109 points, respectively; P = .08).
Although these scores improved with all skin subtypes, LSS skin subscale and FACT-BMT scores remained significantly worse (by 9.0 points and 6.1 points, respectively) for patients with combination and sclerotic disease versus those with epidermal disease after adjusting for potential confounders.
Regarding mortality, every 7-point worsening (clinically meaningful difference) in FACT-BMT score at diagnosis of skin chronic GVHD conferred 9.1% increases in odds of both all-cause mortality and non-relapse mortality, after adjustment for factors such as age and sex. Likewise, for every 11 points worsening (clinically meaningful difference) in LSS skin subscale scores at diagnosis, researchers observed odds increases of 10% in all-cause mortality and 16.4% in non-relapse mortality.
Because patients with combination disease had only slightly more epidermal body surface area (BSA) involvement but significantly higher symptom burden than the other subtypes, the authors added, combination disease may represent a distinct phenotype. “Since we’ve also shown that the severity of patient-reported outcomes is associated with mortality,” Dr. Baumrin said in the interview, “perhaps these patients are at the highest risk of mortality as well.”
A growing population
Although many might think of chronic GVHD as rare, she noted, the number of allogeneic hematopoietic cell transplant (HCT) survivors living in the United States is growing. In a modeling study published in October of 2013 in Biology of Blood and Marrow Transplantation, authors predicted that by 2030, this figure will reach 502,000 — about half of whom will develop chronic GVHD, she said.
With more HCTs being performed each year and ongoing improvements in supportive care, patients are living longer post transplant. “Therefore, many transplant survivors are being taken care of in the community outside of transplant centers.”
Accordingly, Dr. Baumrin said, study findings are relevant to dermatologists in academic and transplant centers and the community who provide skin cancer screenings or other dermatologic care for transplant recipients. “Upon diagnosis of chronic GVHD, the evaluation of disease burden by patient-reported outcome measures may assist in assessing disease severity and response to treatments over time — and to stratify patients at higher risk for mortality and communicate that back to transplant physicians.”
Incorporating PROs into clinical practice might prove especially helpful for patients with sclerotic chronic cutaneous GVHD. Currently, clinicians assess cutaneous GVHD clinically, using parameters including skin thickness. The National Institutes of Health (NIH) Skin Score, used in clinical trials, also measures BSA.
“The issue with sclerosis is, it’s hard to determine clinical severity based on physical examination alone,” Dr. Baumrin said. It can be difficult to quantify skin thickness and changes over time. “So it’s hard to detect improvements, which are often slow. Patient-reported outcome measures may be a more sensitive way to detect response to treatment than our clinical assessments, which are often crude for sclerotic disease.”
In a secondary analysis of the phase 2 clinical trial of belumosudil, a treatment for chronic GVHD, published in October 2022 in Transplantation and Cellular Therapy, response rate was around 30% measured by NIH Skin Score and 77% by PROs. “Our clinical examination in sclerotic type disease falls short in terms of determining therapeutic benefit. PROs might complement those clinical measures,” she said.
Future research will involve determining and validating which PROs matter most clinically and to patients, added Dr. Baumrin. Although widely used in evaluating transplant patients, LSS skin subscale and FACT-BMT scores may not represent patients’ experience of living with cutaneous chronic GVHD as effectively as might other tools such as the Dermatology Life Quality Index (DLQI) or Patient-Reported Outcomes Measurement Information System (PROMIS) measures, she explained.
Study strengths included authors’ use of well-validated PROs rather than novel unvalidated measures, Sandra A. Mitchell, PhD, CRNP, of the National Cancer Institute, Rockville, Maryland, and Edward W. Cowen, MD, MHSc, of the Dermatology Branch at the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS), Bethesda, Maryland, wrote in an accompanying editorial in JAMA Dermatology. However, they added, incorporating causes of death might have revealed that the excess mortality associated with sclerotic disease stemmed at least partly from adverse effects of prolonged immunosuppression, particularly infection.
If future studies establish this to be the case, said Dr. Baumrin, reducing immunosuppression might be warranted for these patients. “And if death is primarily due to chronic GVHD itself, maybe we should treat more aggressively. PROs can help guide this decision.”
The study was supported by the NIH/NIAMS and the University of Pennsylvania. Dr. Baumrin and three coauthors report no relevant financial relationships; other authors had disclosures related to several pharmaceutical companies. Dr. Mitchell and Dr. Cowen had no disclosures.
FROM JAMA DERMATOLOGY
Bigfoot, Bermuda Triangle, ‘No Lido With Epi’?
“Fingers, toes, ears, and nose are places where epinephrine never goes,” Thomas Ehlers, DPM, wrote in Podiatry Today. “That is an adage I heard during podiatry school, my clerkships, and from various attendings throughout my training.”
But as Dr. Ehlers added, epinephrine gets a bad rap. The catchy admonition “has been proven a myth time and time again.”
So Although medical trainees across multiple disciplines are taught to fear the practice, citing the potential for gangrene, its reputation for harm is not supported by the evidence.
Lack of Feeling Doesn’t Care About Your Facts
The debate surfaced anew in response to a recent column by Kenny Lin, MD, MPH, family physician and associate director of the Lancaster General Hospital Family Medicine Residency, in Lancaster, Pennsylvania, about the rather pedestrian topic of why he no longer performs surgery to correct ingrown toenails. Dr. Lin’s admission that he used to do the procedure with a combination of epinephrine and lidocaine turned into a major focus of the comments — many of them harshly critical of the practice:
“Epinephrine is not an appropriate drug to use for podiatry or use in any peripheral area. Gangrene?” one commenter posted.
“Leave epi out of lidocaine to fingers, toes, nose, and ear lobes,” another wrote.
“No lido with epi, whether or not it is contraindicated, because: If there’s any adverse outcome, a lawyer will find plenty of references saying it was contraindicated,” a reader chimed in.
Other commenters disagreed, with one saying, “Please, folks, don’t show that you trained 50 years ago and haven’t changed practice since…”
For Dr. Lin, the response was surprising given what he believes to be the lack of evidence supporting the purported dangers.
“When I think about this, it’s something that was taught to me during residency — that they should not be used on certain areas,” Dr. Lin said. “But since then, studies have been published looking at thousands of cases of people using epinephrine with lidocaine and haven’t found any cases of necrosis.”
Many doctors, like Dr. Lin, say they were cautioned against this in their training. Others don’t remember exactly where they’ve heard it but recognize the idea has a nebulous hold on practice.
Combining epinephrine with lidocaine helps make the numbing last longer, stops bleeding, and reduces the use of lidocaine required, all of which improve the chances of an effective and comfortable intervention for the patient, Dr. Lin said. The approach also reduces the use of tourniquets, which come with their own risks including nerve injury.
However, in areas with limited circulation, this vasoconstrictive effect may be more pronounced, potentially leading to complications for patients with complicating factors.
Clinicians who regularly use the combination of epinephrine and lidocaine for surgery do concede that it can pose certain hazards and considerations in areas without robust blood flow.
But the literature largely points to its safety.
In 2001, California-based plastic and reconstructive surgeon Keith Denkler, MD, published a deep dive on the topic starting in the 19th century, including a review of Index Medicus from 1880 to 1966, a computer review of the National Library of Medicine database from 1966 to 2000, and major textbooks from 1900 to 2000.
He found a total of 48 cases of digital gangrene — but most involved the use of cocaine or procaine. Of the 48 cases, 21 involved the use of epinephrine, and 17 used an unknown concentration based on manual dilution.
“Multiple other concurrent conditions (hot soaks, tight tourniquets, and infection) existed in these case reports, making it difficult to determine the exact cause of the tissue insult,” Dr. Denkler wrote.
In a 2010 retrospective review in the Journal of the American Society of Plastic Surgeons, authors examined 1111 cases involving digital and hand surgery. Of the 611 patients who received injections of 1% lidocaine with epinephrine, none experienced digital necrosis.
Another review from 2003 touted the combination’s safety, in hopes to “help dispel the myth that epinephrine has no place in podiatric anesthesia.” But authors noted limitations of use, including “known sensitivity, thyrotoxicosis, and use of either tricyclic antidepressants or monoamine oxidase inhibitors.”
James Christina, DPM, executive director and CEO of the American Podiatric Medical Association, echoed that sentiment. He said he regularly used the combination to correct bunions, hammer toes, and ingrown toenails over his 20 years of practicing but acknowledged the technique is not appropriate for all such patients.
“There’s always been caution when using epinephrine with local anesthetic,” Dr. Christina told this news organization. “You need a healthy patient with normal circulation and no other complications; someone without vascular compromise.”
Marie Hanna, MD, MEHP, chief of regional anesthesia and acute pain management at Johns Hopkins University, Baltimore, counts herself among the cautious. Citing Principles of Office Anesthesia: Part I. Infiltrative Anesthesia, Dr. Hanna said epinephrine should never be used in digital and penile blocks or in skin flaps with marginal viability.
“It is perfectly fine in certain areas, like the wrist or the arm,” Dr. Hanna said. “But specifically for use in end organs like nose, fingers, ears, toes — all of these with tenuous blood supply — it is not good practice.”
The divide among doctors comes down to theoretical concern, rather than empirical basis, said Rebecca Johnson, MD, chair of the American Society of Anesthesiologists committee on Regional Anesthesia and Acute Pain Medicine and a faculty member at Mayo Clinic, in Rochester, Minnesota.
“It’s just one of those myths we have in practice,” she said.
And legally, Dr. Johnson noted, the mere existence of a myth can be enough of a deterrent for medical practitioners: “Like anything, when you’re trying to do the right thing, if a complication would occur for another reason, you’d want to make sure a jury of your peers didn’t bring up that myth.”
The sources in this story reported no relevant financial conflicts of interest.
A version of this article appeared on Medscape.com.
“Fingers, toes, ears, and nose are places where epinephrine never goes,” Thomas Ehlers, DPM, wrote in Podiatry Today. “That is an adage I heard during podiatry school, my clerkships, and from various attendings throughout my training.”
But as Dr. Ehlers added, epinephrine gets a bad rap. The catchy admonition “has been proven a myth time and time again.”
So Although medical trainees across multiple disciplines are taught to fear the practice, citing the potential for gangrene, its reputation for harm is not supported by the evidence.
Lack of Feeling Doesn’t Care About Your Facts
The debate surfaced anew in response to a recent column by Kenny Lin, MD, MPH, family physician and associate director of the Lancaster General Hospital Family Medicine Residency, in Lancaster, Pennsylvania, about the rather pedestrian topic of why he no longer performs surgery to correct ingrown toenails. Dr. Lin’s admission that he used to do the procedure with a combination of epinephrine and lidocaine turned into a major focus of the comments — many of them harshly critical of the practice:
“Epinephrine is not an appropriate drug to use for podiatry or use in any peripheral area. Gangrene?” one commenter posted.
“Leave epi out of lidocaine to fingers, toes, nose, and ear lobes,” another wrote.
“No lido with epi, whether or not it is contraindicated, because: If there’s any adverse outcome, a lawyer will find plenty of references saying it was contraindicated,” a reader chimed in.
Other commenters disagreed, with one saying, “Please, folks, don’t show that you trained 50 years ago and haven’t changed practice since…”
For Dr. Lin, the response was surprising given what he believes to be the lack of evidence supporting the purported dangers.
“When I think about this, it’s something that was taught to me during residency — that they should not be used on certain areas,” Dr. Lin said. “But since then, studies have been published looking at thousands of cases of people using epinephrine with lidocaine and haven’t found any cases of necrosis.”
Many doctors, like Dr. Lin, say they were cautioned against this in their training. Others don’t remember exactly where they’ve heard it but recognize the idea has a nebulous hold on practice.
Combining epinephrine with lidocaine helps make the numbing last longer, stops bleeding, and reduces the use of lidocaine required, all of which improve the chances of an effective and comfortable intervention for the patient, Dr. Lin said. The approach also reduces the use of tourniquets, which come with their own risks including nerve injury.
However, in areas with limited circulation, this vasoconstrictive effect may be more pronounced, potentially leading to complications for patients with complicating factors.
Clinicians who regularly use the combination of epinephrine and lidocaine for surgery do concede that it can pose certain hazards and considerations in areas without robust blood flow.
But the literature largely points to its safety.
In 2001, California-based plastic and reconstructive surgeon Keith Denkler, MD, published a deep dive on the topic starting in the 19th century, including a review of Index Medicus from 1880 to 1966, a computer review of the National Library of Medicine database from 1966 to 2000, and major textbooks from 1900 to 2000.
He found a total of 48 cases of digital gangrene — but most involved the use of cocaine or procaine. Of the 48 cases, 21 involved the use of epinephrine, and 17 used an unknown concentration based on manual dilution.
“Multiple other concurrent conditions (hot soaks, tight tourniquets, and infection) existed in these case reports, making it difficult to determine the exact cause of the tissue insult,” Dr. Denkler wrote.
In a 2010 retrospective review in the Journal of the American Society of Plastic Surgeons, authors examined 1111 cases involving digital and hand surgery. Of the 611 patients who received injections of 1% lidocaine with epinephrine, none experienced digital necrosis.
Another review from 2003 touted the combination’s safety, in hopes to “help dispel the myth that epinephrine has no place in podiatric anesthesia.” But authors noted limitations of use, including “known sensitivity, thyrotoxicosis, and use of either tricyclic antidepressants or monoamine oxidase inhibitors.”
James Christina, DPM, executive director and CEO of the American Podiatric Medical Association, echoed that sentiment. He said he regularly used the combination to correct bunions, hammer toes, and ingrown toenails over his 20 years of practicing but acknowledged the technique is not appropriate for all such patients.
“There’s always been caution when using epinephrine with local anesthetic,” Dr. Christina told this news organization. “You need a healthy patient with normal circulation and no other complications; someone without vascular compromise.”
Marie Hanna, MD, MEHP, chief of regional anesthesia and acute pain management at Johns Hopkins University, Baltimore, counts herself among the cautious. Citing Principles of Office Anesthesia: Part I. Infiltrative Anesthesia, Dr. Hanna said epinephrine should never be used in digital and penile blocks or in skin flaps with marginal viability.
“It is perfectly fine in certain areas, like the wrist or the arm,” Dr. Hanna said. “But specifically for use in end organs like nose, fingers, ears, toes — all of these with tenuous blood supply — it is not good practice.”
The divide among doctors comes down to theoretical concern, rather than empirical basis, said Rebecca Johnson, MD, chair of the American Society of Anesthesiologists committee on Regional Anesthesia and Acute Pain Medicine and a faculty member at Mayo Clinic, in Rochester, Minnesota.
“It’s just one of those myths we have in practice,” she said.
And legally, Dr. Johnson noted, the mere existence of a myth can be enough of a deterrent for medical practitioners: “Like anything, when you’re trying to do the right thing, if a complication would occur for another reason, you’d want to make sure a jury of your peers didn’t bring up that myth.”
The sources in this story reported no relevant financial conflicts of interest.
A version of this article appeared on Medscape.com.
“Fingers, toes, ears, and nose are places where epinephrine never goes,” Thomas Ehlers, DPM, wrote in Podiatry Today. “That is an adage I heard during podiatry school, my clerkships, and from various attendings throughout my training.”
But as Dr. Ehlers added, epinephrine gets a bad rap. The catchy admonition “has been proven a myth time and time again.”
So Although medical trainees across multiple disciplines are taught to fear the practice, citing the potential for gangrene, its reputation for harm is not supported by the evidence.
Lack of Feeling Doesn’t Care About Your Facts
The debate surfaced anew in response to a recent column by Kenny Lin, MD, MPH, family physician and associate director of the Lancaster General Hospital Family Medicine Residency, in Lancaster, Pennsylvania, about the rather pedestrian topic of why he no longer performs surgery to correct ingrown toenails. Dr. Lin’s admission that he used to do the procedure with a combination of epinephrine and lidocaine turned into a major focus of the comments — many of them harshly critical of the practice:
“Epinephrine is not an appropriate drug to use for podiatry or use in any peripheral area. Gangrene?” one commenter posted.
“Leave epi out of lidocaine to fingers, toes, nose, and ear lobes,” another wrote.
“No lido with epi, whether or not it is contraindicated, because: If there’s any adverse outcome, a lawyer will find plenty of references saying it was contraindicated,” a reader chimed in.
Other commenters disagreed, with one saying, “Please, folks, don’t show that you trained 50 years ago and haven’t changed practice since…”
For Dr. Lin, the response was surprising given what he believes to be the lack of evidence supporting the purported dangers.
“When I think about this, it’s something that was taught to me during residency — that they should not be used on certain areas,” Dr. Lin said. “But since then, studies have been published looking at thousands of cases of people using epinephrine with lidocaine and haven’t found any cases of necrosis.”
Many doctors, like Dr. Lin, say they were cautioned against this in their training. Others don’t remember exactly where they’ve heard it but recognize the idea has a nebulous hold on practice.
Combining epinephrine with lidocaine helps make the numbing last longer, stops bleeding, and reduces the use of lidocaine required, all of which improve the chances of an effective and comfortable intervention for the patient, Dr. Lin said. The approach also reduces the use of tourniquets, which come with their own risks including nerve injury.
However, in areas with limited circulation, this vasoconstrictive effect may be more pronounced, potentially leading to complications for patients with complicating factors.
Clinicians who regularly use the combination of epinephrine and lidocaine for surgery do concede that it can pose certain hazards and considerations in areas without robust blood flow.
But the literature largely points to its safety.
In 2001, California-based plastic and reconstructive surgeon Keith Denkler, MD, published a deep dive on the topic starting in the 19th century, including a review of Index Medicus from 1880 to 1966, a computer review of the National Library of Medicine database from 1966 to 2000, and major textbooks from 1900 to 2000.
He found a total of 48 cases of digital gangrene — but most involved the use of cocaine or procaine. Of the 48 cases, 21 involved the use of epinephrine, and 17 used an unknown concentration based on manual dilution.
“Multiple other concurrent conditions (hot soaks, tight tourniquets, and infection) existed in these case reports, making it difficult to determine the exact cause of the tissue insult,” Dr. Denkler wrote.
In a 2010 retrospective review in the Journal of the American Society of Plastic Surgeons, authors examined 1111 cases involving digital and hand surgery. Of the 611 patients who received injections of 1% lidocaine with epinephrine, none experienced digital necrosis.
Another review from 2003 touted the combination’s safety, in hopes to “help dispel the myth that epinephrine has no place in podiatric anesthesia.” But authors noted limitations of use, including “known sensitivity, thyrotoxicosis, and use of either tricyclic antidepressants or monoamine oxidase inhibitors.”
James Christina, DPM, executive director and CEO of the American Podiatric Medical Association, echoed that sentiment. He said he regularly used the combination to correct bunions, hammer toes, and ingrown toenails over his 20 years of practicing but acknowledged the technique is not appropriate for all such patients.
“There’s always been caution when using epinephrine with local anesthetic,” Dr. Christina told this news organization. “You need a healthy patient with normal circulation and no other complications; someone without vascular compromise.”
Marie Hanna, MD, MEHP, chief of regional anesthesia and acute pain management at Johns Hopkins University, Baltimore, counts herself among the cautious. Citing Principles of Office Anesthesia: Part I. Infiltrative Anesthesia, Dr. Hanna said epinephrine should never be used in digital and penile blocks or in skin flaps with marginal viability.
“It is perfectly fine in certain areas, like the wrist or the arm,” Dr. Hanna said. “But specifically for use in end organs like nose, fingers, ears, toes — all of these with tenuous blood supply — it is not good practice.”
The divide among doctors comes down to theoretical concern, rather than empirical basis, said Rebecca Johnson, MD, chair of the American Society of Anesthesiologists committee on Regional Anesthesia and Acute Pain Medicine and a faculty member at Mayo Clinic, in Rochester, Minnesota.
“It’s just one of those myths we have in practice,” she said.
And legally, Dr. Johnson noted, the mere existence of a myth can be enough of a deterrent for medical practitioners: “Like anything, when you’re trying to do the right thing, if a complication would occur for another reason, you’d want to make sure a jury of your peers didn’t bring up that myth.”
The sources in this story reported no relevant financial conflicts of interest.
A version of this article appeared on Medscape.com.
Oral Herpes Tied to Double Dementia Risk in Older Adults
TOPLINE:
A history of herpes simplex virus (HSV) is associated with a more than doubling of the risk for dementia in older people, results of a prospective epidemiological study showed.
METHODOLOGY:
- The study included 1002 dementia-free 70-year-olds from the Prospective Investigation of Vasculature in Uppsala Seniors cohort who were assessed at baseline and at age 75 and 80 years and followed through medical records at age 85 years.
- Researchers collected and analyzed blood samples to detect anti-HSV and anti-HSV-1 immunoglobulin (Ig) G, anti-cytomegalovirus (CMV) IgG, anti-HSV IgM, and anti-HSV and anti-CMV IgG levels and apolipoprotein epsilon 4 (APOE 4) status of participants.
- Investigators collected information on anti-herpesvirus drug treatment and reviewed dementia diagnoses obtained from medical records to classify as established or probable dementia or Alzheimer’s disease (AD).
TAKEAWAY:
- 82% of participants were anti-HSV IgG carriers, of which 6% had received drug treatment for herpes virus, and 7% of participants developed all-cause dementia and 4% AD during the median 15-year follow up.
- In HSV and HSV-1 subsamples, treatment for herpes virus was not significantly associated with lower risks for AD (HR, 1.46, P = .532 and HR, 1.64; P = .419, respectively) or dementia (HR 1.70; P = .222 and HR, 1.60; P = .320, respectively).
- There was no significant interaction between anti-HSV IgG seroprevalence and APOE 4 with regard to dementia risk, likely due to underpowering, and there were no associations between anti-CMV IgG positivity or anti-HSV IgM positivity and AD or dementia.
IN PRACTICE:
“What’s special about this particular study is that the participants are roughly the same age, which makes the results even more reliable since age differences, which are otherwise linked to the development of dementia, cannot confuse the results,” lead author Erika Vestin, a medical student in the Department of Public Health and Caring Sciences, Clinical Geriatrics, Uppsala University, Sweden, said in a press release. Findings may drive dementia research further towards treating the illness at an early stage using common anti-herpes virus drugs, Ms. Vestin added.
SOURCE:
The study, with Ms. Vestin as lead author, was published online on February 14, 2024, in the Journal of Alzheimer’s Disease.
LIMITATIONS:
The study underrepresented people with diabetes, heart failure, and stroke and lacked information on treatment compliance, dosage, and length and number of prescriptions, which prevented analysis of dose dependency. Since dementia data collection relied on medical records, dementia cases may be underreported. Some cases of AD could have been misclassified as vascular dementia or other dementia.
DISCLOSURES:
The study was supported by the Gun and Bertil Stohne’s Foundation, Swedish Dementia Association, Swedish Society of Medicine, Märta Lundqvist Foundation, Thureus Foundation, Region Uppsala, Gamla Tjänarinnor Foundation, and Swedish Brain Foundation. The authors had no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
A history of herpes simplex virus (HSV) is associated with a more than doubling of the risk for dementia in older people, results of a prospective epidemiological study showed.
METHODOLOGY:
- The study included 1002 dementia-free 70-year-olds from the Prospective Investigation of Vasculature in Uppsala Seniors cohort who were assessed at baseline and at age 75 and 80 years and followed through medical records at age 85 years.
- Researchers collected and analyzed blood samples to detect anti-HSV and anti-HSV-1 immunoglobulin (Ig) G, anti-cytomegalovirus (CMV) IgG, anti-HSV IgM, and anti-HSV and anti-CMV IgG levels and apolipoprotein epsilon 4 (APOE 4) status of participants.
- Investigators collected information on anti-herpesvirus drug treatment and reviewed dementia diagnoses obtained from medical records to classify as established or probable dementia or Alzheimer’s disease (AD).
TAKEAWAY:
- 82% of participants were anti-HSV IgG carriers, of which 6% had received drug treatment for herpes virus, and 7% of participants developed all-cause dementia and 4% AD during the median 15-year follow up.
- In HSV and HSV-1 subsamples, treatment for herpes virus was not significantly associated with lower risks for AD (HR, 1.46, P = .532 and HR, 1.64; P = .419, respectively) or dementia (HR 1.70; P = .222 and HR, 1.60; P = .320, respectively).
- There was no significant interaction between anti-HSV IgG seroprevalence and APOE 4 with regard to dementia risk, likely due to underpowering, and there were no associations between anti-CMV IgG positivity or anti-HSV IgM positivity and AD or dementia.
IN PRACTICE:
“What’s special about this particular study is that the participants are roughly the same age, which makes the results even more reliable since age differences, which are otherwise linked to the development of dementia, cannot confuse the results,” lead author Erika Vestin, a medical student in the Department of Public Health and Caring Sciences, Clinical Geriatrics, Uppsala University, Sweden, said in a press release. Findings may drive dementia research further towards treating the illness at an early stage using common anti-herpes virus drugs, Ms. Vestin added.
SOURCE:
The study, with Ms. Vestin as lead author, was published online on February 14, 2024, in the Journal of Alzheimer’s Disease.
LIMITATIONS:
The study underrepresented people with diabetes, heart failure, and stroke and lacked information on treatment compliance, dosage, and length and number of prescriptions, which prevented analysis of dose dependency. Since dementia data collection relied on medical records, dementia cases may be underreported. Some cases of AD could have been misclassified as vascular dementia or other dementia.
DISCLOSURES:
The study was supported by the Gun and Bertil Stohne’s Foundation, Swedish Dementia Association, Swedish Society of Medicine, Märta Lundqvist Foundation, Thureus Foundation, Region Uppsala, Gamla Tjänarinnor Foundation, and Swedish Brain Foundation. The authors had no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
A history of herpes simplex virus (HSV) is associated with a more than doubling of the risk for dementia in older people, results of a prospective epidemiological study showed.
METHODOLOGY:
- The study included 1002 dementia-free 70-year-olds from the Prospective Investigation of Vasculature in Uppsala Seniors cohort who were assessed at baseline and at age 75 and 80 years and followed through medical records at age 85 years.
- Researchers collected and analyzed blood samples to detect anti-HSV and anti-HSV-1 immunoglobulin (Ig) G, anti-cytomegalovirus (CMV) IgG, anti-HSV IgM, and anti-HSV and anti-CMV IgG levels and apolipoprotein epsilon 4 (APOE 4) status of participants.
- Investigators collected information on anti-herpesvirus drug treatment and reviewed dementia diagnoses obtained from medical records to classify as established or probable dementia or Alzheimer’s disease (AD).
TAKEAWAY:
- 82% of participants were anti-HSV IgG carriers, of which 6% had received drug treatment for herpes virus, and 7% of participants developed all-cause dementia and 4% AD during the median 15-year follow up.
- In HSV and HSV-1 subsamples, treatment for herpes virus was not significantly associated with lower risks for AD (HR, 1.46, P = .532 and HR, 1.64; P = .419, respectively) or dementia (HR 1.70; P = .222 and HR, 1.60; P = .320, respectively).
- There was no significant interaction between anti-HSV IgG seroprevalence and APOE 4 with regard to dementia risk, likely due to underpowering, and there were no associations between anti-CMV IgG positivity or anti-HSV IgM positivity and AD or dementia.
IN PRACTICE:
“What’s special about this particular study is that the participants are roughly the same age, which makes the results even more reliable since age differences, which are otherwise linked to the development of dementia, cannot confuse the results,” lead author Erika Vestin, a medical student in the Department of Public Health and Caring Sciences, Clinical Geriatrics, Uppsala University, Sweden, said in a press release. Findings may drive dementia research further towards treating the illness at an early stage using common anti-herpes virus drugs, Ms. Vestin added.
SOURCE:
The study, with Ms. Vestin as lead author, was published online on February 14, 2024, in the Journal of Alzheimer’s Disease.
LIMITATIONS:
The study underrepresented people with diabetes, heart failure, and stroke and lacked information on treatment compliance, dosage, and length and number of prescriptions, which prevented analysis of dose dependency. Since dementia data collection relied on medical records, dementia cases may be underreported. Some cases of AD could have been misclassified as vascular dementia or other dementia.
DISCLOSURES:
The study was supported by the Gun and Bertil Stohne’s Foundation, Swedish Dementia Association, Swedish Society of Medicine, Märta Lundqvist Foundation, Thureus Foundation, Region Uppsala, Gamla Tjänarinnor Foundation, and Swedish Brain Foundation. The authors had no relevant conflicts of interest.
A version of this article appeared on Medscape.com.