User login
For MD-IQ use only
Papulonodules on the Ankle in a Patient with Lung Cancer
Papulonodules on the Ankle in a Patient with Lung Cancer
THE DIAGNOSIS: Pembrolizumab-Induced Eruptive Squamous Proliferation
Histopathology showed a broad squamous proliferation with acanthosis of the epidermis. Large glassy keratinocytes were seen with scattered necrotic keratinocytes (Figure), and a dense lichenoid band of inflammation was present subjacent to the proliferation. Notably, no hypergranulosis, remarkable keratinocyte atypia, or increased mitotic figures were seen. Based on the patient’s medical history and biopsy results, a diagnosis of pembrolizumab-induced eruptive squamous proliferation was made. The diagnosis was supported by a growing body of evidence of this type of reaction in patients taking programmed death 1 (PD-1) inhibitors.1,2 Conservative treatment with clobetasol ointment 0.05% was initiated with complete resolution of the lesions at the 2-month follow-up appointment. Other common treatments include topical steroids, injected corticosteroids, or cryosurgery to locally control the inflammation and atypical proliferation of cells.3
Pembrolizumab is a humanized IgG4 monoclonal antibody targeting the PD-1 receptor that has been utilized for its antitumor activity against various cancers, including unresectable and metastatic melanoma, head and neck cancers, and non–small cell lung cancer (NSCLC).1,4,5 While this drug has extended the lives of many patients with cancer, there are adverse reactions associated with PD-1 inhibitors (eg, pembrolizumab, nivolumab). Skin toxicity to PD-1 inhibitors is the one of most common immune-mediated reactions worldwide, occurring in approximately 30% of patients.6,7 Reactions can occur while a patient is taking the inciting drug and can continue up to 2 months after treatment discontinuation.8 Skin reactions associated with PD-1 inhibitors vary from lichenoid reactions and vitiligolike patches to psoriasis or eczema flares and are organized into 4 categories: inflammatory, immunobullous, alteration of keratinocytes, and alteration of melanocytes.9 Our patient demonstrated alteration of keratinocytes, which is characterized by overlapping features of hypertrophic lichen planus and early keratoacanthoma.
The differential diagnoses for pembrolizumab-induced eruptive squamous proliferation include squamous cell carcinoma (SCC), psoriasis, hypertrophic lichen planus, and cutaneous metastasis of NSCLC. Hypertrophic lupus erythematosus also is a well-documented reaction to use of immune-checkpoint inhibitors.10 Direct immunofluorescence could have helped differentiate hypertrophic lupus erythematosus from an eruptive squamous proliferation in our patient; however, due to her response to treatment, no additional workup was done.
Squamous cell carcinoma, which is the most common type of skin cancer in Black patients in the United States,11 has been shown to manifest after a PD-1 inhibitor is taken.12 Although it typically has a more chronic persistent course, the clinical appearance of SCC can be similar to the findings seen in our patient. Histologically, SCC may demonstrate necrosis, but the atypical proliferations will invade the dermis—a feature not seen in our patient’s histopathology.13
Lichen planus (LP) is an eruptive immune reaction of violaceous polygonal papules and plaques commonly seen on the ankles14 that has been shown to be an adverse effect of pembrolizumab.15 There are several subtypes of LP including hypertrophic versions, which can appear clinically similar to the findings seen in our patient. On dermoscopy, the classic finding of white lines, known as Wickham striae, is seen in all subtypes and can help diagnose this pathologic process. Under the microscope, LP can manifest with hyperkeratosis without parakeratosis, irregular thickening of the stratum granulosum, sawtooth rete ridges, and destruction of the basal layer.14
Psoriasis also has been shown to be exacerbated by anti–PD-1 therapy, although the majority of patients diagnosed with PD-1–induced psoriasis have a personal or family history of the disease.6 Clinically, psoriasis can have a hyperpigmented or violaceous appearance in patients with skin of color.16 The histopathology of psoriasis typically reveals confluent parakeratosis, neutrophils in the stratum corneum, regular acanthosis, thinning of the suprapapillary plates, and vessels in the dermal papillae.17
Although cutaneous metastasis of NSCLC may appear clinically similar to the current case, it is one of the rarer organ sites of metastasis for lung cancer.18 In our patient, biopsy quickly ruled out this diagnosis. If it had been a site of metastasis, histopathology would have shown a dermal-based proliferation of dysplastic cells without epidermal connection.19
It is important for dermatologists to recognize eruptive squamous proliferations associated with pembrolizumab, as they often respond to conservative treatment and typically do not require dose reduction or discontinuation of the inciting drug.
- Freshwater T, Kondic A, Ahamadi M, et al. Evaluation of dosing strategy for pembrolizumab for oncology indications. J Immunother Cancer. 2017;5:43. doi:10.1186/s40425-017-0242-5
- Preti BTB, Pencz A, Cowger JJM, et al. Skin deep: a fascinating case report of immunotherapy-triggered, treatment-refractory autoimmune lichen planus and keratoacanthoma. Case Rep Oncol. 2021;14: 1189-1193. doi:10.1159/000518313
- Fradet M, Sibaud V, Tournier E, et al. Multiple keratoacanthoma-like lesions in a patient treated with pembrolizumab. Acta Derm Venereol. 2019;99:1301-1302. doi:10.2340/00015555-3301
- Flynn JP, Gerriets V. Pembrolizumab. StatPearls [Internet]. StatPearls Publishing; 2023. Updated June 26, 2023. Accessed April 2, 2025.
- Antonov NK, Nair KG, Halasz CL. Transient eruptive keratoacanthomas associated with Nivolumab. JAAD Case Rep. 2019;5:342-345. doi:10.1016/j.jdcr.2019.01.025
- Voudouri D, Nikolaou V, Laschos K, et al. Anti-Pd1/Pdl1 induced psoriasis. Curr Probl Cancer. 2017;41:407-412. doi:10.1016 /j.currproblcancer.2017.10.003
- Belum VR, Benhuri B, Postow MA, et al. Characterisation and management of dermatologic adverse events to agents targeting the PD-1 receptor. Eur J Cancer. 2016;60:12-25. doi:10.1016/j.ejca.2016.02.010
- Coscarart A, Martel J, Lee MP, et al. Pembrolizumab-induced pseudoepitheliomatous eruption consistent with hypertrophic lichen planus. J Cutan Pathol. 2020;47:275-279. doi:10.1111/cup.13587
- Curry JL, Tetzlaff MT, Nagarajan P, et al. Diverse types of dermatologic toxicities from immune checkpoint blockade therapy. J Cutan Pathol. 2017;44:158-176. doi:10.1111/cup.12858
- Vitzthum von Eckstaedt H, Singh A, Reid P, et al. Immune checkpoint inhibitors and lupus erythematosus. Pharmaceuticals (Basel). 2024;2:15;17. doi:10.3390/ph17020252
- Halder RM, Bridgeman-Shah S. Skin cancer in African Americans. Cancer. 1995;75:667-673.
- Vu M, Chapman S, Lenz B, et al. Squamous cell carcinoma or squamous proliferation associated with nivolumab treatment for metastatic melanoma. Dermatol Online J. 2022;6:28. doi:10.5070/d328357786
- Howell JY, Ramsey ML. Squamous cell skin cancer. StatPearls [Internet]. StatPearls Publishing; 2024. Updated July 2, 2024. Accessed April 2, 2025.
- Arnold DL, Krishnamurthy K. Lichen planus. StatPearls [Internet]. StatPearls Publishing; 2024. Updated October 29, 2024. Accessed April 2, 2025.
- Yamashita A, Akasaka E, Nakano H, et al. Pembrolizumab-induced lichen planus on the scalp of a patient with non-small-cell lung carcinoma. Case Rep Dermatol. 2021;13:487-491. doi:10.1159/000519486
- Alexis AF, Blackcloud P. Psoriasis in skin of color: epidemiology, genetics, clinical presentation, and treatment nuances. J Clin Aesthet Dermatol. 2014;7:16-24.
- Murphy M, Kerr P, Grant-Kels JM. The histopathologic spectrum of psoriasis. Clin Dermatol. 2007;25:524-528. doi:10.1016 /j.clindermatol.2007.08.005.
- Hidaka T, Ishii Y, Kitamura S. Clinical features of skin metastasis from lung cancer. Intern Med. 1996;35:459-462. doi:10.2169 /internalmedicine.35.459.
- Sariya D, Ruth K, Adams-McDonnell R, et al. Clinicopathologic correlation of cutaneous metastases: experience from a cancer center. Arch Dermatol. 2007;143:613–620. doi:10.1001/archderm.143.5.613
THE DIAGNOSIS: Pembrolizumab-Induced Eruptive Squamous Proliferation
Histopathology showed a broad squamous proliferation with acanthosis of the epidermis. Large glassy keratinocytes were seen with scattered necrotic keratinocytes (Figure), and a dense lichenoid band of inflammation was present subjacent to the proliferation. Notably, no hypergranulosis, remarkable keratinocyte atypia, or increased mitotic figures were seen. Based on the patient’s medical history and biopsy results, a diagnosis of pembrolizumab-induced eruptive squamous proliferation was made. The diagnosis was supported by a growing body of evidence of this type of reaction in patients taking programmed death 1 (PD-1) inhibitors.1,2 Conservative treatment with clobetasol ointment 0.05% was initiated with complete resolution of the lesions at the 2-month follow-up appointment. Other common treatments include topical steroids, injected corticosteroids, or cryosurgery to locally control the inflammation and atypical proliferation of cells.3

Pembrolizumab is a humanized IgG4 monoclonal antibody targeting the PD-1 receptor that has been utilized for its antitumor activity against various cancers, including unresectable and metastatic melanoma, head and neck cancers, and non–small cell lung cancer (NSCLC).1,4,5 While this drug has extended the lives of many patients with cancer, there are adverse reactions associated with PD-1 inhibitors (eg, pembrolizumab, nivolumab). Skin toxicity to PD-1 inhibitors is the one of most common immune-mediated reactions worldwide, occurring in approximately 30% of patients.6,7 Reactions can occur while a patient is taking the inciting drug and can continue up to 2 months after treatment discontinuation.8 Skin reactions associated with PD-1 inhibitors vary from lichenoid reactions and vitiligolike patches to psoriasis or eczema flares and are organized into 4 categories: inflammatory, immunobullous, alteration of keratinocytes, and alteration of melanocytes.9 Our patient demonstrated alteration of keratinocytes, which is characterized by overlapping features of hypertrophic lichen planus and early keratoacanthoma.
The differential diagnoses for pembrolizumab-induced eruptive squamous proliferation include squamous cell carcinoma (SCC), psoriasis, hypertrophic lichen planus, and cutaneous metastasis of NSCLC. Hypertrophic lupus erythematosus also is a well-documented reaction to use of immune-checkpoint inhibitors.10 Direct immunofluorescence could have helped differentiate hypertrophic lupus erythematosus from an eruptive squamous proliferation in our patient; however, due to her response to treatment, no additional workup was done.
Squamous cell carcinoma, which is the most common type of skin cancer in Black patients in the United States,11 has been shown to manifest after a PD-1 inhibitor is taken.12 Although it typically has a more chronic persistent course, the clinical appearance of SCC can be similar to the findings seen in our patient. Histologically, SCC may demonstrate necrosis, but the atypical proliferations will invade the dermis—a feature not seen in our patient’s histopathology.13
Lichen planus (LP) is an eruptive immune reaction of violaceous polygonal papules and plaques commonly seen on the ankles14 that has been shown to be an adverse effect of pembrolizumab.15 There are several subtypes of LP including hypertrophic versions, which can appear clinically similar to the findings seen in our patient. On dermoscopy, the classic finding of white lines, known as Wickham striae, is seen in all subtypes and can help diagnose this pathologic process. Under the microscope, LP can manifest with hyperkeratosis without parakeratosis, irregular thickening of the stratum granulosum, sawtooth rete ridges, and destruction of the basal layer.14
Psoriasis also has been shown to be exacerbated by anti–PD-1 therapy, although the majority of patients diagnosed with PD-1–induced psoriasis have a personal or family history of the disease.6 Clinically, psoriasis can have a hyperpigmented or violaceous appearance in patients with skin of color.16 The histopathology of psoriasis typically reveals confluent parakeratosis, neutrophils in the stratum corneum, regular acanthosis, thinning of the suprapapillary plates, and vessels in the dermal papillae.17
Although cutaneous metastasis of NSCLC may appear clinically similar to the current case, it is one of the rarer organ sites of metastasis for lung cancer.18 In our patient, biopsy quickly ruled out this diagnosis. If it had been a site of metastasis, histopathology would have shown a dermal-based proliferation of dysplastic cells without epidermal connection.19
It is important for dermatologists to recognize eruptive squamous proliferations associated with pembrolizumab, as they often respond to conservative treatment and typically do not require dose reduction or discontinuation of the inciting drug.
THE DIAGNOSIS: Pembrolizumab-Induced Eruptive Squamous Proliferation
Histopathology showed a broad squamous proliferation with acanthosis of the epidermis. Large glassy keratinocytes were seen with scattered necrotic keratinocytes (Figure), and a dense lichenoid band of inflammation was present subjacent to the proliferation. Notably, no hypergranulosis, remarkable keratinocyte atypia, or increased mitotic figures were seen. Based on the patient’s medical history and biopsy results, a diagnosis of pembrolizumab-induced eruptive squamous proliferation was made. The diagnosis was supported by a growing body of evidence of this type of reaction in patients taking programmed death 1 (PD-1) inhibitors.1,2 Conservative treatment with clobetasol ointment 0.05% was initiated with complete resolution of the lesions at the 2-month follow-up appointment. Other common treatments include topical steroids, injected corticosteroids, or cryosurgery to locally control the inflammation and atypical proliferation of cells.3

Pembrolizumab is a humanized IgG4 monoclonal antibody targeting the PD-1 receptor that has been utilized for its antitumor activity against various cancers, including unresectable and metastatic melanoma, head and neck cancers, and non–small cell lung cancer (NSCLC).1,4,5 While this drug has extended the lives of many patients with cancer, there are adverse reactions associated with PD-1 inhibitors (eg, pembrolizumab, nivolumab). Skin toxicity to PD-1 inhibitors is the one of most common immune-mediated reactions worldwide, occurring in approximately 30% of patients.6,7 Reactions can occur while a patient is taking the inciting drug and can continue up to 2 months after treatment discontinuation.8 Skin reactions associated with PD-1 inhibitors vary from lichenoid reactions and vitiligolike patches to psoriasis or eczema flares and are organized into 4 categories: inflammatory, immunobullous, alteration of keratinocytes, and alteration of melanocytes.9 Our patient demonstrated alteration of keratinocytes, which is characterized by overlapping features of hypertrophic lichen planus and early keratoacanthoma.
The differential diagnoses for pembrolizumab-induced eruptive squamous proliferation include squamous cell carcinoma (SCC), psoriasis, hypertrophic lichen planus, and cutaneous metastasis of NSCLC. Hypertrophic lupus erythematosus also is a well-documented reaction to use of immune-checkpoint inhibitors.10 Direct immunofluorescence could have helped differentiate hypertrophic lupus erythematosus from an eruptive squamous proliferation in our patient; however, due to her response to treatment, no additional workup was done.
Squamous cell carcinoma, which is the most common type of skin cancer in Black patients in the United States,11 has been shown to manifest after a PD-1 inhibitor is taken.12 Although it typically has a more chronic persistent course, the clinical appearance of SCC can be similar to the findings seen in our patient. Histologically, SCC may demonstrate necrosis, but the atypical proliferations will invade the dermis—a feature not seen in our patient’s histopathology.13
Lichen planus (LP) is an eruptive immune reaction of violaceous polygonal papules and plaques commonly seen on the ankles14 that has been shown to be an adverse effect of pembrolizumab.15 There are several subtypes of LP including hypertrophic versions, which can appear clinically similar to the findings seen in our patient. On dermoscopy, the classic finding of white lines, known as Wickham striae, is seen in all subtypes and can help diagnose this pathologic process. Under the microscope, LP can manifest with hyperkeratosis without parakeratosis, irregular thickening of the stratum granulosum, sawtooth rete ridges, and destruction of the basal layer.14
Psoriasis also has been shown to be exacerbated by anti–PD-1 therapy, although the majority of patients diagnosed with PD-1–induced psoriasis have a personal or family history of the disease.6 Clinically, psoriasis can have a hyperpigmented or violaceous appearance in patients with skin of color.16 The histopathology of psoriasis typically reveals confluent parakeratosis, neutrophils in the stratum corneum, regular acanthosis, thinning of the suprapapillary plates, and vessels in the dermal papillae.17
Although cutaneous metastasis of NSCLC may appear clinically similar to the current case, it is one of the rarer organ sites of metastasis for lung cancer.18 In our patient, biopsy quickly ruled out this diagnosis. If it had been a site of metastasis, histopathology would have shown a dermal-based proliferation of dysplastic cells without epidermal connection.19
It is important for dermatologists to recognize eruptive squamous proliferations associated with pembrolizumab, as they often respond to conservative treatment and typically do not require dose reduction or discontinuation of the inciting drug.
- Freshwater T, Kondic A, Ahamadi M, et al. Evaluation of dosing strategy for pembrolizumab for oncology indications. J Immunother Cancer. 2017;5:43. doi:10.1186/s40425-017-0242-5
- Preti BTB, Pencz A, Cowger JJM, et al. Skin deep: a fascinating case report of immunotherapy-triggered, treatment-refractory autoimmune lichen planus and keratoacanthoma. Case Rep Oncol. 2021;14: 1189-1193. doi:10.1159/000518313
- Fradet M, Sibaud V, Tournier E, et al. Multiple keratoacanthoma-like lesions in a patient treated with pembrolizumab. Acta Derm Venereol. 2019;99:1301-1302. doi:10.2340/00015555-3301
- Flynn JP, Gerriets V. Pembrolizumab. StatPearls [Internet]. StatPearls Publishing; 2023. Updated June 26, 2023. Accessed April 2, 2025.
- Antonov NK, Nair KG, Halasz CL. Transient eruptive keratoacanthomas associated with Nivolumab. JAAD Case Rep. 2019;5:342-345. doi:10.1016/j.jdcr.2019.01.025
- Voudouri D, Nikolaou V, Laschos K, et al. Anti-Pd1/Pdl1 induced psoriasis. Curr Probl Cancer. 2017;41:407-412. doi:10.1016 /j.currproblcancer.2017.10.003
- Belum VR, Benhuri B, Postow MA, et al. Characterisation and management of dermatologic adverse events to agents targeting the PD-1 receptor. Eur J Cancer. 2016;60:12-25. doi:10.1016/j.ejca.2016.02.010
- Coscarart A, Martel J, Lee MP, et al. Pembrolizumab-induced pseudoepitheliomatous eruption consistent with hypertrophic lichen planus. J Cutan Pathol. 2020;47:275-279. doi:10.1111/cup.13587
- Curry JL, Tetzlaff MT, Nagarajan P, et al. Diverse types of dermatologic toxicities from immune checkpoint blockade therapy. J Cutan Pathol. 2017;44:158-176. doi:10.1111/cup.12858
- Vitzthum von Eckstaedt H, Singh A, Reid P, et al. Immune checkpoint inhibitors and lupus erythematosus. Pharmaceuticals (Basel). 2024;2:15;17. doi:10.3390/ph17020252
- Halder RM, Bridgeman-Shah S. Skin cancer in African Americans. Cancer. 1995;75:667-673.
- Vu M, Chapman S, Lenz B, et al. Squamous cell carcinoma or squamous proliferation associated with nivolumab treatment for metastatic melanoma. Dermatol Online J. 2022;6:28. doi:10.5070/d328357786
- Howell JY, Ramsey ML. Squamous cell skin cancer. StatPearls [Internet]. StatPearls Publishing; 2024. Updated July 2, 2024. Accessed April 2, 2025.
- Arnold DL, Krishnamurthy K. Lichen planus. StatPearls [Internet]. StatPearls Publishing; 2024. Updated October 29, 2024. Accessed April 2, 2025.
- Yamashita A, Akasaka E, Nakano H, et al. Pembrolizumab-induced lichen planus on the scalp of a patient with non-small-cell lung carcinoma. Case Rep Dermatol. 2021;13:487-491. doi:10.1159/000519486
- Alexis AF, Blackcloud P. Psoriasis in skin of color: epidemiology, genetics, clinical presentation, and treatment nuances. J Clin Aesthet Dermatol. 2014;7:16-24.
- Murphy M, Kerr P, Grant-Kels JM. The histopathologic spectrum of psoriasis. Clin Dermatol. 2007;25:524-528. doi:10.1016 /j.clindermatol.2007.08.005.
- Hidaka T, Ishii Y, Kitamura S. Clinical features of skin metastasis from lung cancer. Intern Med. 1996;35:459-462. doi:10.2169 /internalmedicine.35.459.
- Sariya D, Ruth K, Adams-McDonnell R, et al. Clinicopathologic correlation of cutaneous metastases: experience from a cancer center. Arch Dermatol. 2007;143:613–620. doi:10.1001/archderm.143.5.613
- Freshwater T, Kondic A, Ahamadi M, et al. Evaluation of dosing strategy for pembrolizumab for oncology indications. J Immunother Cancer. 2017;5:43. doi:10.1186/s40425-017-0242-5
- Preti BTB, Pencz A, Cowger JJM, et al. Skin deep: a fascinating case report of immunotherapy-triggered, treatment-refractory autoimmune lichen planus and keratoacanthoma. Case Rep Oncol. 2021;14: 1189-1193. doi:10.1159/000518313
- Fradet M, Sibaud V, Tournier E, et al. Multiple keratoacanthoma-like lesions in a patient treated with pembrolizumab. Acta Derm Venereol. 2019;99:1301-1302. doi:10.2340/00015555-3301
- Flynn JP, Gerriets V. Pembrolizumab. StatPearls [Internet]. StatPearls Publishing; 2023. Updated June 26, 2023. Accessed April 2, 2025.
- Antonov NK, Nair KG, Halasz CL. Transient eruptive keratoacanthomas associated with Nivolumab. JAAD Case Rep. 2019;5:342-345. doi:10.1016/j.jdcr.2019.01.025
- Voudouri D, Nikolaou V, Laschos K, et al. Anti-Pd1/Pdl1 induced psoriasis. Curr Probl Cancer. 2017;41:407-412. doi:10.1016 /j.currproblcancer.2017.10.003
- Belum VR, Benhuri B, Postow MA, et al. Characterisation and management of dermatologic adverse events to agents targeting the PD-1 receptor. Eur J Cancer. 2016;60:12-25. doi:10.1016/j.ejca.2016.02.010
- Coscarart A, Martel J, Lee MP, et al. Pembrolizumab-induced pseudoepitheliomatous eruption consistent with hypertrophic lichen planus. J Cutan Pathol. 2020;47:275-279. doi:10.1111/cup.13587
- Curry JL, Tetzlaff MT, Nagarajan P, et al. Diverse types of dermatologic toxicities from immune checkpoint blockade therapy. J Cutan Pathol. 2017;44:158-176. doi:10.1111/cup.12858
- Vitzthum von Eckstaedt H, Singh A, Reid P, et al. Immune checkpoint inhibitors and lupus erythematosus. Pharmaceuticals (Basel). 2024;2:15;17. doi:10.3390/ph17020252
- Halder RM, Bridgeman-Shah S. Skin cancer in African Americans. Cancer. 1995;75:667-673.
- Vu M, Chapman S, Lenz B, et al. Squamous cell carcinoma or squamous proliferation associated with nivolumab treatment for metastatic melanoma. Dermatol Online J. 2022;6:28. doi:10.5070/d328357786
- Howell JY, Ramsey ML. Squamous cell skin cancer. StatPearls [Internet]. StatPearls Publishing; 2024. Updated July 2, 2024. Accessed April 2, 2025.
- Arnold DL, Krishnamurthy K. Lichen planus. StatPearls [Internet]. StatPearls Publishing; 2024. Updated October 29, 2024. Accessed April 2, 2025.
- Yamashita A, Akasaka E, Nakano H, et al. Pembrolizumab-induced lichen planus on the scalp of a patient with non-small-cell lung carcinoma. Case Rep Dermatol. 2021;13:487-491. doi:10.1159/000519486
- Alexis AF, Blackcloud P. Psoriasis in skin of color: epidemiology, genetics, clinical presentation, and treatment nuances. J Clin Aesthet Dermatol. 2014;7:16-24.
- Murphy M, Kerr P, Grant-Kels JM. The histopathologic spectrum of psoriasis. Clin Dermatol. 2007;25:524-528. doi:10.1016 /j.clindermatol.2007.08.005.
- Hidaka T, Ishii Y, Kitamura S. Clinical features of skin metastasis from lung cancer. Intern Med. 1996;35:459-462. doi:10.2169 /internalmedicine.35.459.
- Sariya D, Ruth K, Adams-McDonnell R, et al. Clinicopathologic correlation of cutaneous metastases: experience from a cancer center. Arch Dermatol. 2007;143:613–620. doi:10.1001/archderm.143.5.613
Papulonodules on the Ankle in a Patient with Lung Cancer
Papulonodules on the Ankle in a Patient with Lung Cancer
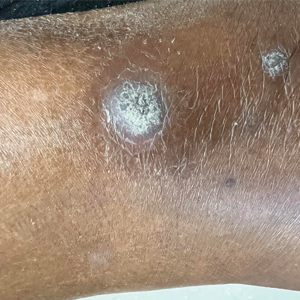
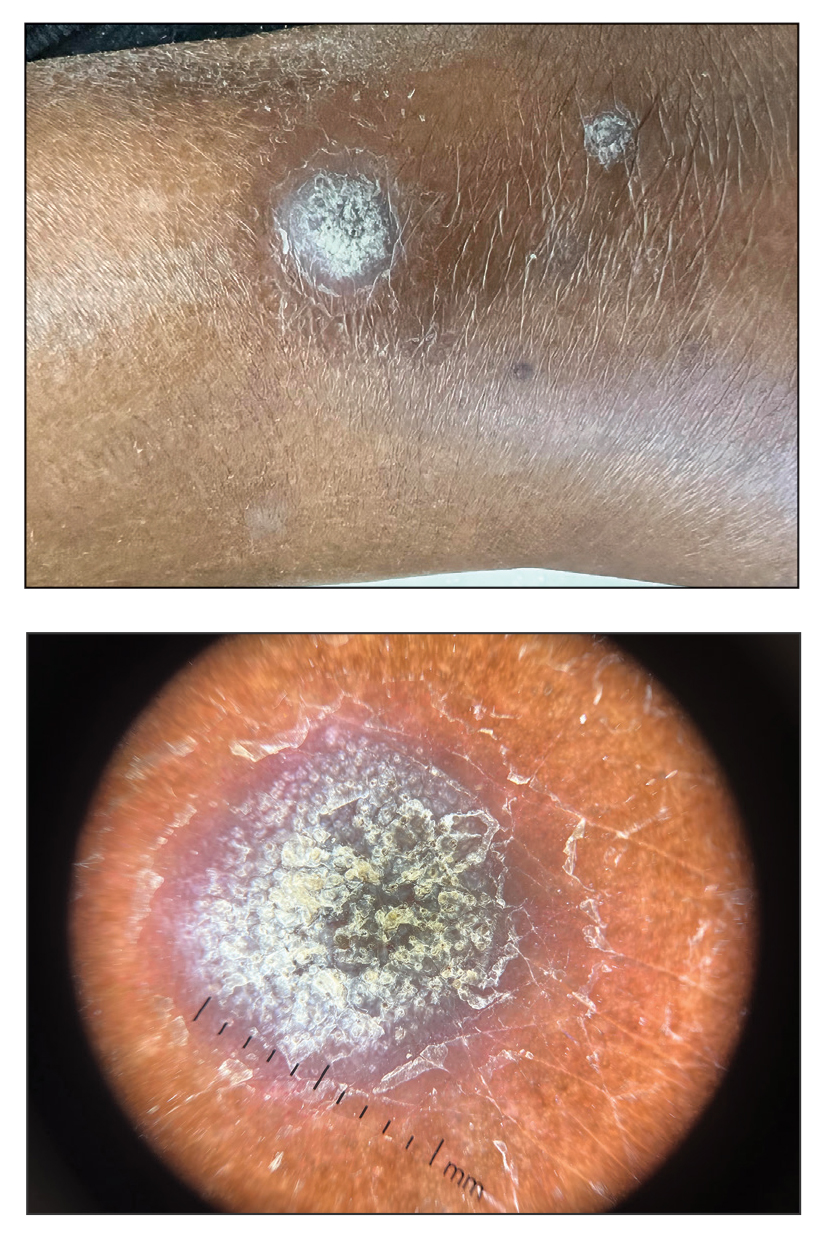
A 75-year-old woman presented to the dermatology department with well-circumscribed, round, hyperkeratotic papulonodules on the ankle of 3 months’ duration (top). The papulonodules also were evaluated by dermoscopy, which highlighted in greater detail the hyperkeratosis seen grossly (bottom). The patient had a history of chronic obstructive pulmonary disease and metastatic lung cancer and had been taking pembrolizumab for the past 2 years. The lesions initially appeared on the medial right foot and slowly spread proximally. Most of the lesions resolved spontaneously except for 2 on the right ankle. At the current presentation, one lesion was slightly tender to palpation, but both were otherwise asymptomatic. A lesion was biopsied and sent for dermatopathologic evaluation.
UK Funds AI Blood Test for Early Cancer Detection
A clinical trial of a promising blood test that could offer faster and more accurate diagnoses for common cancers has received funding from the Department of Health and Social Care (DHSC).
The miONCO-Dx test detects cancer at an early stage by analysing microRNA expression in blood.
It uses artificial intelligence to identify the presence and origin of the disease.
The test was developed by Xgenera, a University of Southampton spinout, in collaboration with the National Institute for Health and Care Research.
Initial analysis of data from more than 20,000 patients showed that the test detected 12 of the most common and lethal cancers at an early stage and with over 99% accuracy.
Bowel Cancer Among Key Targets
Bowel cancer, the fourth most common cancer in the United Kingdom, is a principal target for the test.
Around 44,000 people in the UK are diagnosed with bowel cancer each year. At stage 1, approximately 90% of people survive for 5 years or more, but this drops to around 10% at stage 4.
Wes Streeting, Secretary of State for Health and Social Care, said in a press release, “The key to surviving cancer is catching it as early as possible, so this government is taking the urgent action needed to make sure that happens.”
£2.4 Million Awarded for Clinical Trial
The DHSC has awarded Xgenera £2.4 million to advance development of the test, which has now been refined into a cheaper, faster, and more scalable version.
The funding will support a clinical trial involving 8000 patients. The DHSC described this as “a formal and significant step towards bringing the test closer to patients by ensuring it is fit for purpose in the NHS.”
The trial will be run by Cancer Research UK Southampton Clinical Trials Unit.
Potential for NHS Use
Dr Victoria Goss, head of early diagnosis and translational research at the trials unit, said in a press release, “A reliable test such as this could have the potential to see a major shift in cancer screening, making it easier and cheaper to provide on the NHS, cutting health inequalities, and ultimately reducing the number of people who die from the disease.”
Xgenera co-founder Dr Andy Shapanis, a research fellow at the University of Southampton, said that the new study would evaluate the useability, accuracy, and cost-effectiveness of the test for use within the NHS in future.
“The hope is that if the test is shown to be successful in the early diagnosis of the 12 cancers we have currently identified biomarkers for, then it could be expanded to look at over 50 other cancers in the future,” he said.
Comparison With Other Tests
The miONCO-Dx test follows other attempts at multicancer early detection, such as the Galleri test from Grail, which is already being trialled in the NHS.
Galleri screens for altered DNA methylation patterns in blood and claims to detect more than 50 types of cancer. It raised hopes for earlier diagnosis, less invasive treatment, and potential cost savings.
However, critics have raised concerns about low detection rates in early-stage cancers, high false-positive rates, imprecise cancer origin analysis, cost, and unproven mortality gains. Questions have also been expressed about possible political influence in its selection for NHS trials.
A Broader Screening Platform
Xgenera co-founder Professor Paul Skipp, director of the Centre for Proteomic Research at the University of Southampton, said earlier this year that the miONCO-Dx test was “a real game-changer.”
The test can detect lung, breast, prostate, pancreatic, colorectal, ovarian, liver, brain, oesophageal, bladder, and gastric cancer and bone and soft-tissue sarcoma. It works by identifying imbalances in microRNAs, a class of small noncoding RNAs with functions in posttranscriptional regulation of gene expression, influencing cellular activities including cell growth, differentiation, development, and apoptosis.
The presence of microRNA imbalances can be identified from just 10-15 drops of blood, across all stages of tumour growth.
In comparison, according to Skipp, screening is only available currently for three types of cancer in the UK, and each test targets a single type.
Xgenera has also received external investment from the innovation investment companies Qantx, Empirical Ventures, and Ascension Ventures to further develop the test.
Dr Sheena Meredith is an established medical writer, editor, and consultant in healthcare communications, with extensive experience writing for medical professionals and the general public. She is qualified in medicine and in law and medical ethics.
A version of this article first appeared on Medscape.com.
A clinical trial of a promising blood test that could offer faster and more accurate diagnoses for common cancers has received funding from the Department of Health and Social Care (DHSC).
The miONCO-Dx test detects cancer at an early stage by analysing microRNA expression in blood.
It uses artificial intelligence to identify the presence and origin of the disease.
The test was developed by Xgenera, a University of Southampton spinout, in collaboration with the National Institute for Health and Care Research.
Initial analysis of data from more than 20,000 patients showed that the test detected 12 of the most common and lethal cancers at an early stage and with over 99% accuracy.
Bowel Cancer Among Key Targets
Bowel cancer, the fourth most common cancer in the United Kingdom, is a principal target for the test.
Around 44,000 people in the UK are diagnosed with bowel cancer each year. At stage 1, approximately 90% of people survive for 5 years or more, but this drops to around 10% at stage 4.
Wes Streeting, Secretary of State for Health and Social Care, said in a press release, “The key to surviving cancer is catching it as early as possible, so this government is taking the urgent action needed to make sure that happens.”
£2.4 Million Awarded for Clinical Trial
The DHSC has awarded Xgenera £2.4 million to advance development of the test, which has now been refined into a cheaper, faster, and more scalable version.
The funding will support a clinical trial involving 8000 patients. The DHSC described this as “a formal and significant step towards bringing the test closer to patients by ensuring it is fit for purpose in the NHS.”
The trial will be run by Cancer Research UK Southampton Clinical Trials Unit.
Potential for NHS Use
Dr Victoria Goss, head of early diagnosis and translational research at the trials unit, said in a press release, “A reliable test such as this could have the potential to see a major shift in cancer screening, making it easier and cheaper to provide on the NHS, cutting health inequalities, and ultimately reducing the number of people who die from the disease.”
Xgenera co-founder Dr Andy Shapanis, a research fellow at the University of Southampton, said that the new study would evaluate the useability, accuracy, and cost-effectiveness of the test for use within the NHS in future.
“The hope is that if the test is shown to be successful in the early diagnosis of the 12 cancers we have currently identified biomarkers for, then it could be expanded to look at over 50 other cancers in the future,” he said.
Comparison With Other Tests
The miONCO-Dx test follows other attempts at multicancer early detection, such as the Galleri test from Grail, which is already being trialled in the NHS.
Galleri screens for altered DNA methylation patterns in blood and claims to detect more than 50 types of cancer. It raised hopes for earlier diagnosis, less invasive treatment, and potential cost savings.
However, critics have raised concerns about low detection rates in early-stage cancers, high false-positive rates, imprecise cancer origin analysis, cost, and unproven mortality gains. Questions have also been expressed about possible political influence in its selection for NHS trials.
A Broader Screening Platform
Xgenera co-founder Professor Paul Skipp, director of the Centre for Proteomic Research at the University of Southampton, said earlier this year that the miONCO-Dx test was “a real game-changer.”
The test can detect lung, breast, prostate, pancreatic, colorectal, ovarian, liver, brain, oesophageal, bladder, and gastric cancer and bone and soft-tissue sarcoma. It works by identifying imbalances in microRNAs, a class of small noncoding RNAs with functions in posttranscriptional regulation of gene expression, influencing cellular activities including cell growth, differentiation, development, and apoptosis.
The presence of microRNA imbalances can be identified from just 10-15 drops of blood, across all stages of tumour growth.
In comparison, according to Skipp, screening is only available currently for three types of cancer in the UK, and each test targets a single type.
Xgenera has also received external investment from the innovation investment companies Qantx, Empirical Ventures, and Ascension Ventures to further develop the test.
Dr Sheena Meredith is an established medical writer, editor, and consultant in healthcare communications, with extensive experience writing for medical professionals and the general public. She is qualified in medicine and in law and medical ethics.
A version of this article first appeared on Medscape.com.
A clinical trial of a promising blood test that could offer faster and more accurate diagnoses for common cancers has received funding from the Department of Health and Social Care (DHSC).
The miONCO-Dx test detects cancer at an early stage by analysing microRNA expression in blood.
It uses artificial intelligence to identify the presence and origin of the disease.
The test was developed by Xgenera, a University of Southampton spinout, in collaboration with the National Institute for Health and Care Research.
Initial analysis of data from more than 20,000 patients showed that the test detected 12 of the most common and lethal cancers at an early stage and with over 99% accuracy.
Bowel Cancer Among Key Targets
Bowel cancer, the fourth most common cancer in the United Kingdom, is a principal target for the test.
Around 44,000 people in the UK are diagnosed with bowel cancer each year. At stage 1, approximately 90% of people survive for 5 years or more, but this drops to around 10% at stage 4.
Wes Streeting, Secretary of State for Health and Social Care, said in a press release, “The key to surviving cancer is catching it as early as possible, so this government is taking the urgent action needed to make sure that happens.”
£2.4 Million Awarded for Clinical Trial
The DHSC has awarded Xgenera £2.4 million to advance development of the test, which has now been refined into a cheaper, faster, and more scalable version.
The funding will support a clinical trial involving 8000 patients. The DHSC described this as “a formal and significant step towards bringing the test closer to patients by ensuring it is fit for purpose in the NHS.”
The trial will be run by Cancer Research UK Southampton Clinical Trials Unit.
Potential for NHS Use
Dr Victoria Goss, head of early diagnosis and translational research at the trials unit, said in a press release, “A reliable test such as this could have the potential to see a major shift in cancer screening, making it easier and cheaper to provide on the NHS, cutting health inequalities, and ultimately reducing the number of people who die from the disease.”
Xgenera co-founder Dr Andy Shapanis, a research fellow at the University of Southampton, said that the new study would evaluate the useability, accuracy, and cost-effectiveness of the test for use within the NHS in future.
“The hope is that if the test is shown to be successful in the early diagnosis of the 12 cancers we have currently identified biomarkers for, then it could be expanded to look at over 50 other cancers in the future,” he said.
Comparison With Other Tests
The miONCO-Dx test follows other attempts at multicancer early detection, such as the Galleri test from Grail, which is already being trialled in the NHS.
Galleri screens for altered DNA methylation patterns in blood and claims to detect more than 50 types of cancer. It raised hopes for earlier diagnosis, less invasive treatment, and potential cost savings.
However, critics have raised concerns about low detection rates in early-stage cancers, high false-positive rates, imprecise cancer origin analysis, cost, and unproven mortality gains. Questions have also been expressed about possible political influence in its selection for NHS trials.
A Broader Screening Platform
Xgenera co-founder Professor Paul Skipp, director of the Centre for Proteomic Research at the University of Southampton, said earlier this year that the miONCO-Dx test was “a real game-changer.”
The test can detect lung, breast, prostate, pancreatic, colorectal, ovarian, liver, brain, oesophageal, bladder, and gastric cancer and bone and soft-tissue sarcoma. It works by identifying imbalances in microRNAs, a class of small noncoding RNAs with functions in posttranscriptional regulation of gene expression, influencing cellular activities including cell growth, differentiation, development, and apoptosis.
The presence of microRNA imbalances can be identified from just 10-15 drops of blood, across all stages of tumour growth.
In comparison, according to Skipp, screening is only available currently for three types of cancer in the UK, and each test targets a single type.
Xgenera has also received external investment from the innovation investment companies Qantx, Empirical Ventures, and Ascension Ventures to further develop the test.
Dr Sheena Meredith is an established medical writer, editor, and consultant in healthcare communications, with extensive experience writing for medical professionals and the general public. She is qualified in medicine and in law and medical ethics.
A version of this article first appeared on Medscape.com.
Can a Polygenic Risk Score Turn the Tide on Prostate Cancer Screening?
Incorporating a polygenic risk score into prostate cancer screening could enhance the detection of clinically significant prostate cancer that conventional screening may miss, according to results of the BARCODE 1 clinical trial conducted in the United Kingdom.
The study found that about 72% of participants with high polygenic risk scores were diagnosed with clinically significant prostate cancers, which would not have been detected with prostate-specific antigen (PSA) testing or MRI.
“With this test, it could be possible to turn the tide on prostate cancer,” study author Ros Eeles, PhD, professor of oncogenetics at The Institute of Cancer Research, London, England, said in a statement following the publication of the analysis in The New England Journal of Medicine.
Prostate cancer remains the second most commonly diagnosed cancer among men. As a screening tool, PSA testing has been criticized for leading to a high rate of false positive results and overdiagnosis — defined as a screen-detected cancer that would take longer to progress to clinical cancer than the patient’s lifetime. Both issues can result in overtreatment.
Given prostate cancer’s high heritability and the proliferation of genome-wide association studies identifying common genetic variants, there has been growing interest in using polygenic risk scores to improve risk stratification and guide screening.
“Building on decades of research into the genetic markers of prostate cancer, our study shows that the theory does work in practice — we can identify men at risk of aggressive cancers who need further tests and spare the men who are at lower risk from unnecessary treatments,” said Eeles.
An Adjunct to Screening?
The BARCODE 1 study, conducted in the United Kingdom, tested the clinical utility of a polygenic risk score as an adjunct to screening.
The researchers recruited men aged 55-69 years from primary care centers in the United Kingdom. Using germline DNA extracted from saliva, they derived polygenic risk scores from 130 genetic variants known to be associated with an increased risk for prostate cancer.
Among a total of 6393 men who had their scores calculated, 745 (12%) had a score in the top 10% of genetic risk (≥ 90th percentile) and were invited to undergo further screening.
Of these, 468 (63%) accepted the invite and underwent multiparametric MRI and transperineal prostate biopsy, irrespective of the PSA level. Overall, 187 (40%) were diagnosed with prostate cancer following biopsy. Of the 187 men with prostate cancer, 55% (n = 103) had disease classified as intermediate or high risk (Gleason score ≥ 7) per National Comprehensive Cancer Network criteria and therefore warranted further treatment.
Researchers then compared screening that incorporated polygenic risk scores with standard screening with PSA levels and MRI.
When participants’ risk was stratified by their polygenic risk score, 103 patients (55%) with prostate cancer could be classified as intermediate or higher risk, thus warranting treatment. Overall, 74 (71.8%) of those cancers would have been missed using the standard diagnostic pathway in the United Kingdom, which requires patients to have a high PSA level (> 3.0 μg/L) as well as a positive MRI result. These 74 patients either had PSA levels ≤ 3.0 μg/L or negative MRIs, which would mean these patients would typically fall below the action threshold for further testing.
Of the 103 participants warranting treatment, 40 of these men would have been classified as unfavorable intermediate, high, or very high risk, which would require radical treatment. Among this group, roughly 43% would have been missed using the UK diagnostic pathway.
However, the investigators estimated a rate of overdiagnosis with the use of polygenic risk scores of 16%-21%, similar to the overdiagnosis estimates in two prior PSA-based screening studies, signaling that the addition of polygenic risk scores does not necessarily reduce the risk for overdiagnosis.
Overall, “this study is the strongest evidence to date on the clinical utility of a polygenic score for prostate cancer screening,” commented Michael Inouye, professor of systems genomics & population health, University of Cambridge, Cambridge, England, in a statement from the UK nonprofit Science Media Centre (SMC).
“I suspect we will look back on this as a landmark study that really made the clinical case for polygenic scores as a new tool that moved health systems from disease management to early detection and prevention,” said Inouye, who was not involved in the study.
However, other experts were more cautious about the findings.
Dusko Ilic, MD, professor of stem cell sciences, King’s College London, London, England, said the results are “promising, especially in identifying significant cancers that would otherwise be missed,” but cautioned that “there is no direct evidence yet that using [polygenic risk scores] improves long-term outcomes such as mortality or quality-adjusted life years.”
“Modeling suggests benefit, but empirical confirmation is needed,” Ilic said in the SMC statement.
The hope is that the recently launched TRANSFORM trial will help answer some of these outstanding questions.
The current study suggests that polygenic risk scores for prostate cancer “would be a useful component of a multimodality screening program that assesses age, family history of prostate cancer, PSA, and MRI results as triage tools before biopsy is recommended,” David Hunter, MPH, ScD, with Harvard T. H. Chan School of Public Health, Boston, and University of Oxford, Oxford, England, wrote in an editorial accompanying the study.
“To make this integrated program a reality, however, changes to infrastructure would be needed to make running and analyzing a regulated genome array as easy as requesting a PSA level or ordering an MRI. Clearly, we are far from that future,” Hunter cautioned.
“A possible first step that would require less infrastructure could be to order a polygenic risk score only for men with a positive PSA result, then use the polygenic risk score to determine who should undergo an MRI, and then use all the information to determine whether biopsy is recommended,” Hunter said.
In his view, the current study is a “first step on a long road to evaluating new components of any disease screening pathway.”
The research received funding from the European Research Council, the Bob Willis Fund, Cancer Research UK, the Peacock Trust, and the National Institute for Health and Care Research Biomedical Research Centre at The Royal Marsden and The Institute of Cancer Research. Disclosures for authors and editorialists are available with the original article. Inouye and Ilic reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
Incorporating a polygenic risk score into prostate cancer screening could enhance the detection of clinically significant prostate cancer that conventional screening may miss, according to results of the BARCODE 1 clinical trial conducted in the United Kingdom.
The study found that about 72% of participants with high polygenic risk scores were diagnosed with clinically significant prostate cancers, which would not have been detected with prostate-specific antigen (PSA) testing or MRI.
“With this test, it could be possible to turn the tide on prostate cancer,” study author Ros Eeles, PhD, professor of oncogenetics at The Institute of Cancer Research, London, England, said in a statement following the publication of the analysis in The New England Journal of Medicine.
Prostate cancer remains the second most commonly diagnosed cancer among men. As a screening tool, PSA testing has been criticized for leading to a high rate of false positive results and overdiagnosis — defined as a screen-detected cancer that would take longer to progress to clinical cancer than the patient’s lifetime. Both issues can result in overtreatment.
Given prostate cancer’s high heritability and the proliferation of genome-wide association studies identifying common genetic variants, there has been growing interest in using polygenic risk scores to improve risk stratification and guide screening.
“Building on decades of research into the genetic markers of prostate cancer, our study shows that the theory does work in practice — we can identify men at risk of aggressive cancers who need further tests and spare the men who are at lower risk from unnecessary treatments,” said Eeles.
An Adjunct to Screening?
The BARCODE 1 study, conducted in the United Kingdom, tested the clinical utility of a polygenic risk score as an adjunct to screening.
The researchers recruited men aged 55-69 years from primary care centers in the United Kingdom. Using germline DNA extracted from saliva, they derived polygenic risk scores from 130 genetic variants known to be associated with an increased risk for prostate cancer.
Among a total of 6393 men who had their scores calculated, 745 (12%) had a score in the top 10% of genetic risk (≥ 90th percentile) and were invited to undergo further screening.
Of these, 468 (63%) accepted the invite and underwent multiparametric MRI and transperineal prostate biopsy, irrespective of the PSA level. Overall, 187 (40%) were diagnosed with prostate cancer following biopsy. Of the 187 men with prostate cancer, 55% (n = 103) had disease classified as intermediate or high risk (Gleason score ≥ 7) per National Comprehensive Cancer Network criteria and therefore warranted further treatment.
Researchers then compared screening that incorporated polygenic risk scores with standard screening with PSA levels and MRI.
When participants’ risk was stratified by their polygenic risk score, 103 patients (55%) with prostate cancer could be classified as intermediate or higher risk, thus warranting treatment. Overall, 74 (71.8%) of those cancers would have been missed using the standard diagnostic pathway in the United Kingdom, which requires patients to have a high PSA level (> 3.0 μg/L) as well as a positive MRI result. These 74 patients either had PSA levels ≤ 3.0 μg/L or negative MRIs, which would mean these patients would typically fall below the action threshold for further testing.
Of the 103 participants warranting treatment, 40 of these men would have been classified as unfavorable intermediate, high, or very high risk, which would require radical treatment. Among this group, roughly 43% would have been missed using the UK diagnostic pathway.
However, the investigators estimated a rate of overdiagnosis with the use of polygenic risk scores of 16%-21%, similar to the overdiagnosis estimates in two prior PSA-based screening studies, signaling that the addition of polygenic risk scores does not necessarily reduce the risk for overdiagnosis.
Overall, “this study is the strongest evidence to date on the clinical utility of a polygenic score for prostate cancer screening,” commented Michael Inouye, professor of systems genomics & population health, University of Cambridge, Cambridge, England, in a statement from the UK nonprofit Science Media Centre (SMC).
“I suspect we will look back on this as a landmark study that really made the clinical case for polygenic scores as a new tool that moved health systems from disease management to early detection and prevention,” said Inouye, who was not involved in the study.
However, other experts were more cautious about the findings.
Dusko Ilic, MD, professor of stem cell sciences, King’s College London, London, England, said the results are “promising, especially in identifying significant cancers that would otherwise be missed,” but cautioned that “there is no direct evidence yet that using [polygenic risk scores] improves long-term outcomes such as mortality or quality-adjusted life years.”
“Modeling suggests benefit, but empirical confirmation is needed,” Ilic said in the SMC statement.
The hope is that the recently launched TRANSFORM trial will help answer some of these outstanding questions.
The current study suggests that polygenic risk scores for prostate cancer “would be a useful component of a multimodality screening program that assesses age, family history of prostate cancer, PSA, and MRI results as triage tools before biopsy is recommended,” David Hunter, MPH, ScD, with Harvard T. H. Chan School of Public Health, Boston, and University of Oxford, Oxford, England, wrote in an editorial accompanying the study.
“To make this integrated program a reality, however, changes to infrastructure would be needed to make running and analyzing a regulated genome array as easy as requesting a PSA level or ordering an MRI. Clearly, we are far from that future,” Hunter cautioned.
“A possible first step that would require less infrastructure could be to order a polygenic risk score only for men with a positive PSA result, then use the polygenic risk score to determine who should undergo an MRI, and then use all the information to determine whether biopsy is recommended,” Hunter said.
In his view, the current study is a “first step on a long road to evaluating new components of any disease screening pathway.”
The research received funding from the European Research Council, the Bob Willis Fund, Cancer Research UK, the Peacock Trust, and the National Institute for Health and Care Research Biomedical Research Centre at The Royal Marsden and The Institute of Cancer Research. Disclosures for authors and editorialists are available with the original article. Inouye and Ilic reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
Incorporating a polygenic risk score into prostate cancer screening could enhance the detection of clinically significant prostate cancer that conventional screening may miss, according to results of the BARCODE 1 clinical trial conducted in the United Kingdom.
The study found that about 72% of participants with high polygenic risk scores were diagnosed with clinically significant prostate cancers, which would not have been detected with prostate-specific antigen (PSA) testing or MRI.
“With this test, it could be possible to turn the tide on prostate cancer,” study author Ros Eeles, PhD, professor of oncogenetics at The Institute of Cancer Research, London, England, said in a statement following the publication of the analysis in The New England Journal of Medicine.
Prostate cancer remains the second most commonly diagnosed cancer among men. As a screening tool, PSA testing has been criticized for leading to a high rate of false positive results and overdiagnosis — defined as a screen-detected cancer that would take longer to progress to clinical cancer than the patient’s lifetime. Both issues can result in overtreatment.
Given prostate cancer’s high heritability and the proliferation of genome-wide association studies identifying common genetic variants, there has been growing interest in using polygenic risk scores to improve risk stratification and guide screening.
“Building on decades of research into the genetic markers of prostate cancer, our study shows that the theory does work in practice — we can identify men at risk of aggressive cancers who need further tests and spare the men who are at lower risk from unnecessary treatments,” said Eeles.
An Adjunct to Screening?
The BARCODE 1 study, conducted in the United Kingdom, tested the clinical utility of a polygenic risk score as an adjunct to screening.
The researchers recruited men aged 55-69 years from primary care centers in the United Kingdom. Using germline DNA extracted from saliva, they derived polygenic risk scores from 130 genetic variants known to be associated with an increased risk for prostate cancer.
Among a total of 6393 men who had their scores calculated, 745 (12%) had a score in the top 10% of genetic risk (≥ 90th percentile) and were invited to undergo further screening.
Of these, 468 (63%) accepted the invite and underwent multiparametric MRI and transperineal prostate biopsy, irrespective of the PSA level. Overall, 187 (40%) were diagnosed with prostate cancer following biopsy. Of the 187 men with prostate cancer, 55% (n = 103) had disease classified as intermediate or high risk (Gleason score ≥ 7) per National Comprehensive Cancer Network criteria and therefore warranted further treatment.
Researchers then compared screening that incorporated polygenic risk scores with standard screening with PSA levels and MRI.
When participants’ risk was stratified by their polygenic risk score, 103 patients (55%) with prostate cancer could be classified as intermediate or higher risk, thus warranting treatment. Overall, 74 (71.8%) of those cancers would have been missed using the standard diagnostic pathway in the United Kingdom, which requires patients to have a high PSA level (> 3.0 μg/L) as well as a positive MRI result. These 74 patients either had PSA levels ≤ 3.0 μg/L or negative MRIs, which would mean these patients would typically fall below the action threshold for further testing.
Of the 103 participants warranting treatment, 40 of these men would have been classified as unfavorable intermediate, high, or very high risk, which would require radical treatment. Among this group, roughly 43% would have been missed using the UK diagnostic pathway.
However, the investigators estimated a rate of overdiagnosis with the use of polygenic risk scores of 16%-21%, similar to the overdiagnosis estimates in two prior PSA-based screening studies, signaling that the addition of polygenic risk scores does not necessarily reduce the risk for overdiagnosis.
Overall, “this study is the strongest evidence to date on the clinical utility of a polygenic score for prostate cancer screening,” commented Michael Inouye, professor of systems genomics & population health, University of Cambridge, Cambridge, England, in a statement from the UK nonprofit Science Media Centre (SMC).
“I suspect we will look back on this as a landmark study that really made the clinical case for polygenic scores as a new tool that moved health systems from disease management to early detection and prevention,” said Inouye, who was not involved in the study.
However, other experts were more cautious about the findings.
Dusko Ilic, MD, professor of stem cell sciences, King’s College London, London, England, said the results are “promising, especially in identifying significant cancers that would otherwise be missed,” but cautioned that “there is no direct evidence yet that using [polygenic risk scores] improves long-term outcomes such as mortality or quality-adjusted life years.”
“Modeling suggests benefit, but empirical confirmation is needed,” Ilic said in the SMC statement.
The hope is that the recently launched TRANSFORM trial will help answer some of these outstanding questions.
The current study suggests that polygenic risk scores for prostate cancer “would be a useful component of a multimodality screening program that assesses age, family history of prostate cancer, PSA, and MRI results as triage tools before biopsy is recommended,” David Hunter, MPH, ScD, with Harvard T. H. Chan School of Public Health, Boston, and University of Oxford, Oxford, England, wrote in an editorial accompanying the study.
“To make this integrated program a reality, however, changes to infrastructure would be needed to make running and analyzing a regulated genome array as easy as requesting a PSA level or ordering an MRI. Clearly, we are far from that future,” Hunter cautioned.
“A possible first step that would require less infrastructure could be to order a polygenic risk score only for men with a positive PSA result, then use the polygenic risk score to determine who should undergo an MRI, and then use all the information to determine whether biopsy is recommended,” Hunter said.
In his view, the current study is a “first step on a long road to evaluating new components of any disease screening pathway.”
The research received funding from the European Research Council, the Bob Willis Fund, Cancer Research UK, the Peacock Trust, and the National Institute for Health and Care Research Biomedical Research Centre at The Royal Marsden and The Institute of Cancer Research. Disclosures for authors and editorialists are available with the original article. Inouye and Ilic reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
Acral Erythema, Edema, and Scaly Plaques in a Patient With Polyneuropathy
Acral Erythema, Edema, and Scaly Plaques in a Patient With Polyneuropathy
THE DIAGNOSIS: Borderline-Borderline Leprosy With Type 1 Lepra Reaction
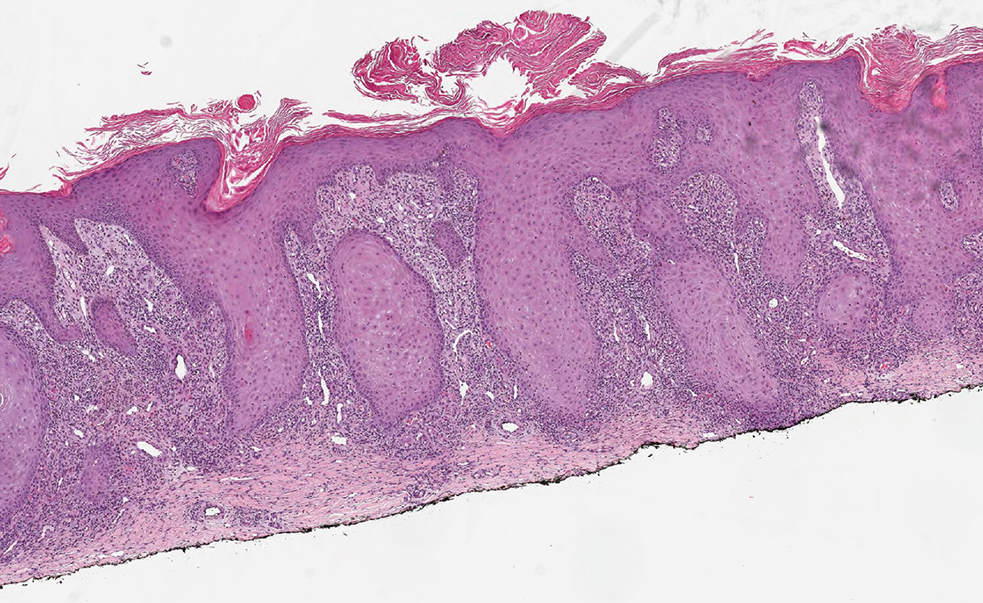
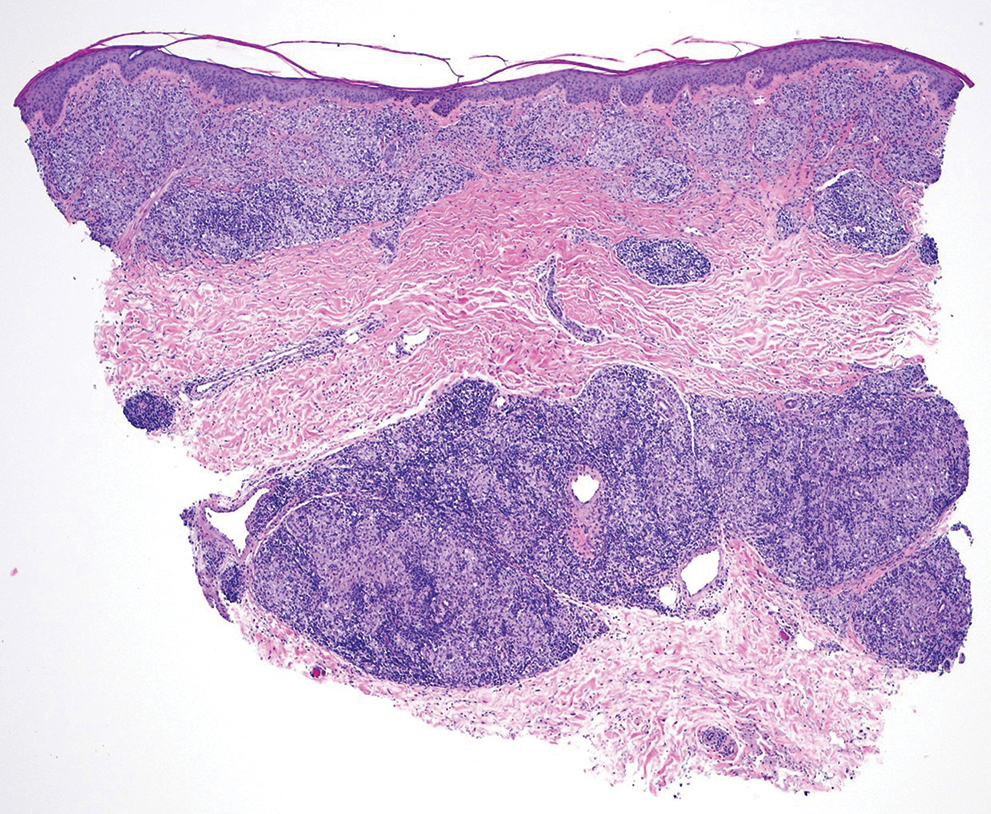
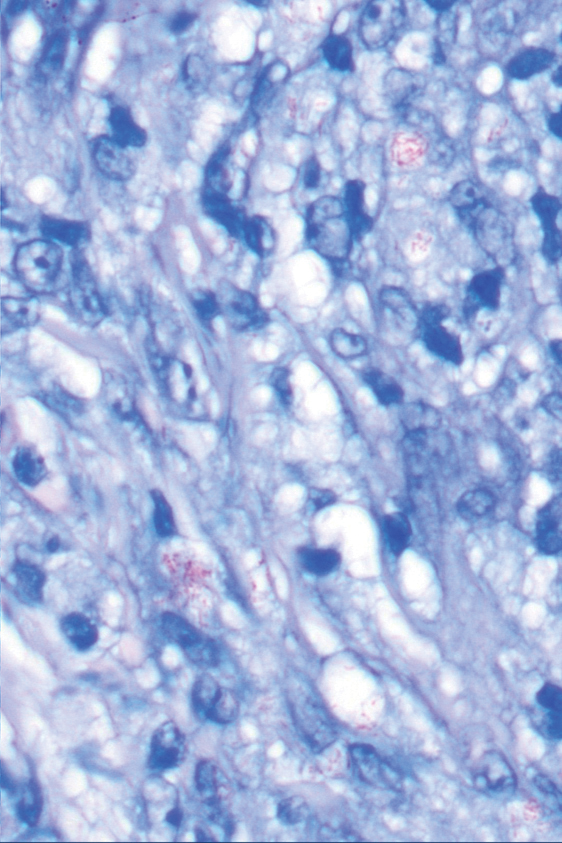
Punch biopsies from plaques on the right elbow and right shin revealed diffuse granulomatous dermatitis (Figure 1) with a narrow Grenz zone in the superficial dermis. The upper dermis contained a dense bandlike infiltrate of histiocytes with abundant foamy-gray cytoplasm and a moderate admixture of lymphocytes. The mid and deep dermis contained a nodular, perivascular, periadnexal, and perineural infiltrate of histiocytes and a dense admixture of lymphocytes. Periodic acid-Schiff and Gram stains were negative for microorganisms. Fite stain was positive for numerous organisms in histiocytes and small dermal nerves (Figure 2). These findings and the clinical examination confirmed a diagnosis of borderline-borderline leprosy with type 1 lepra reaction. The patient was started on dapsone 100 mg, rifampin 600 mg, and clofazimine 100 mg once daily and experienced clinical improvement within 6 months.
The World Health Organization reported more than 200,000 new leprosy cases globally in 2019, with most occurring in India, Brazil, and Indonesia.1 About 150 to 250 new cases are detected in the United States annually.1 The Ridley-Jopling classification of leprosy divides the condition into 5 categories: tuberculoid, borderline tuberculoid, borderline-borderline (BB), borderline lepromatous, and lepromatous. At one end of the spectrum, tuberculoid leprosy—a predominant Th1 immune response mediated by CD4 lymphocytes, interleukin (IL) 2, and interferon gamma2—is characterized by sharply demarcated erythematous and hypopigmented plaques with raised borders and an annular appearance.2,3 Lesions typically have atrophic and hypopigmented centers that often appear in an asymmetric distribution on the arms and legs.2,3 Histologic features include dermal tuberculoid granulomas with epithelioid cells—some located directly beneath the epidermis and others around deep vessels and nerves3—multinucleated Langerhans giant cells, thickened peripheral nerves with intraneural lymphocytic infiltrates, and granulomas with central necrosis. Fite-Faraco staining exhibits few bacteria.2
Lepromatous leprosy occurs in individuals with impaired T-cell immunity, leading to multiple red-brown nodular infiltrates in the skin and mucous membranes.2,3 Lesions typically are symmetric and favor the face and auricle of the ear.2,3 Histologically, there are bluish-gray foamy macrophages that form diffuse or nodular infiltrates with few lymphocytes,2 with a Grenz zone between the epidermis and dermis. Nerves may show lamination of the perineurium resembling an onion skin.2,3 Immunohistochemistry shows predominant CD8-positive infiltrates with a Th2 response and positive IL-4 and IL-10. Fite-Faraco stain shows numerous mycobacteria arranged in clusters and in histiocytes.2
Tuberculoid leprosy is treated with dapsone 100 mg and rifampin 600 mg once daily for 6 months,4 and lepromatous leprosy is treated with dapsone 100 mg, rifampin 600 mg, and clofazimine 50 mg once daily for 12 months.4 The prognosis for both is good with treatment; erythema and induration of skin lesions may improve within a few months, but residual nerve damage is common, especially in those with advanced disease prior to treatment.2 For direct contacts, a single dose of rifampin may be given.4
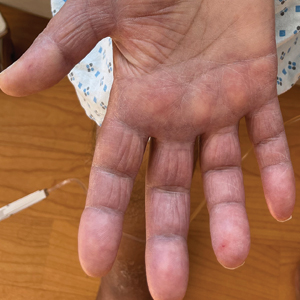
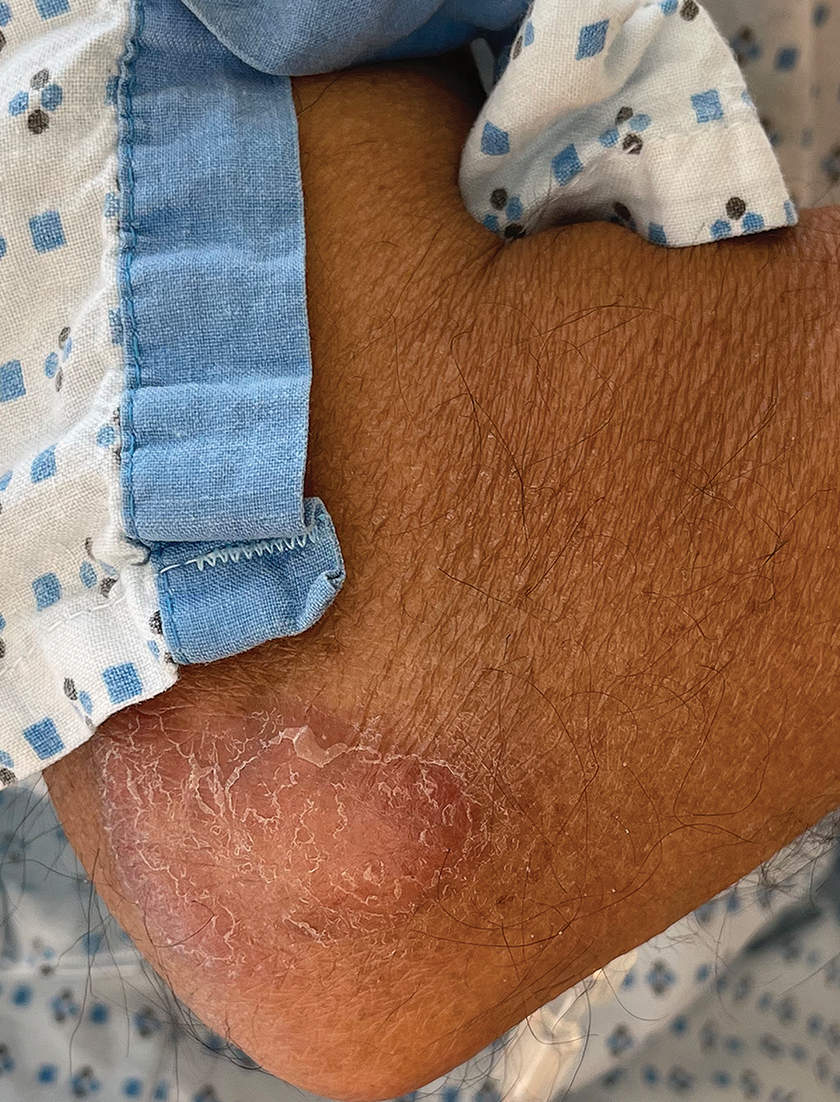
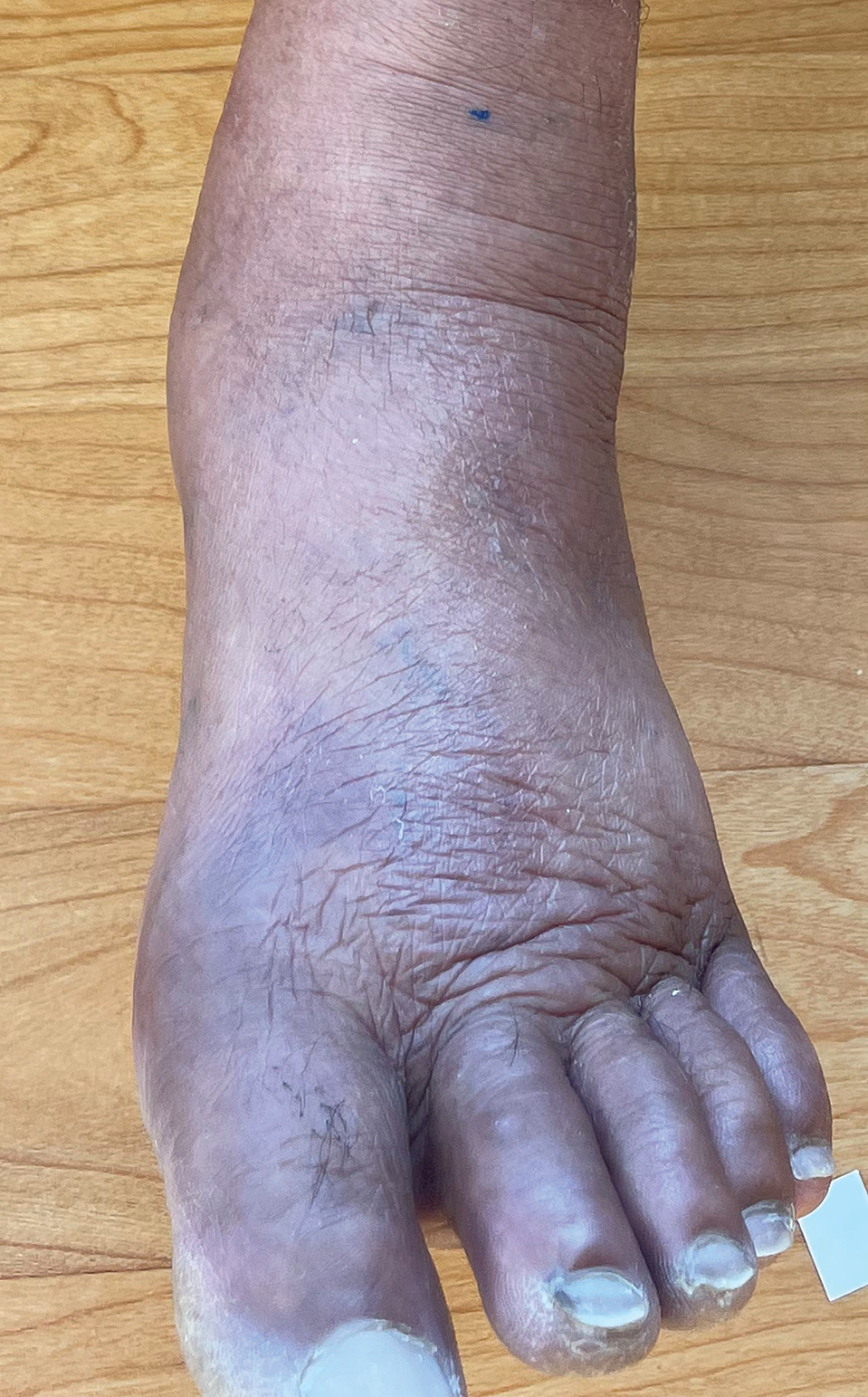
Borderline-borderline leprosy manifests with numerous asymmetric annular plaques, as seen in our patient (Figure 3). Histology findings can be variable and often overlap with other forms of leprosy. There can be epithelioid granulomas and only a few acid-fast bacilli (AFB) or diffuse histiocytic aggregates with foamy histiocytes containing large numbers of AFB.3 Nerve involvement is variable but can be severe in the setting of type 1 lepra reaction, which was present in our patient. Type 1 lepra reaction—a type IV cell-mediated allergic hypersensitivity reaction to Mycobacterium leprae antigens—manifests clinically with hyperesthesia, erythema, edema, and subsequent scaling.2 It occurs in up to 30% of patients with borderline leprosy, usually within 12 months of treatment initiation.2 Our patient had considerable edema and erythema of the hands and feet (Figure 4) along with extensive polyneuropathy prior to starting therapy.
Lucio phenomenon is a rare leprosy reaction found in patients with untreated lepromatous leprosy characterized by erythematous to violaceous macules that lead to ulceronecrotic lesions.5 Histologically, there are many AFB in the vascular endothelium, leukocytoclastic vasculitis, and ischemic epidermal necrosis.5 Our patient did not have ulcerative or necrotic lesions.
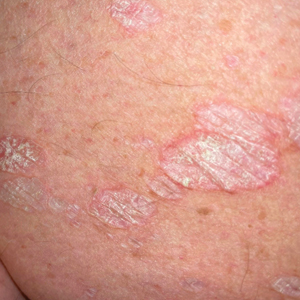
The classic skin lesions of psoriasis vulgaris can be described as well-demarcated pink plaques with white or silvery scales that usually are distributed symmetrically and often are found on extensor surfaces.6 Rapidly progressive lesions can be annular with normal skin in the center, mimicking the lesions seen in tuberculoid leprosy. Clinically, both psoriasis and tuberculoid forms of leprosy are sharply demarcated; however, psoriatic lesions often have micaceous overlying scale that is not present in leprosy. Characteristic histologic findings of psoriasis are hyperkeratosis, parakeratosis, and acanthosis of the epidermis with dilated blood vessels and a lymphocytic infiltrate, predominantly into the dermis.7 Psoriatic arthritis has a variable clinical course but tends to emerge 5 to 12 years after initial skin manifestation.8 Classic clinical symptoms include swelling, tenderness, stiffness, and pain in joints and surrounding tissues.8 Other than edema, our patient did not exhibit signs of psoriatic arthritis.
Sarcoidosis is a systemic autoimmune disease characterized by noncaseating epithelioid granulomas affecting various organs, with cutaneous manifestations present in approximately 30% of all cases. Cutaneous manifestations can be variable, including maculopapular lesions, plaques, and nodules.9 Differentiating between cutaneous sarcoidosis and tuberculoid leprosy can be challenging, as both are granulomatous processes; however, histology of sarcoidosis demonstrates noncaseating granulomas in the dermis and/or subcutaneous tissues without AFB9 compared to granulomas with necrotic centers in tuberculoid leprosy.
Cutaneous tuberculosis has variable morphologies. One subtype, lupus vulgaris, can manifest with violaceous, scaly, eroded plaques that could be confused for leprosy. Lupus vulgaris usually results from hematogenous or lymphatic seeding in individuals with high or moderate immunity to M tuberculosis.10
Histologically, the dermis has tuberculoid granulomas containing multinucleated giant cells,10 which can mimic those seen in BB leprosy. Tuberculin skin test results often are positive10; while this test was not performed in our patient, chest radiography was unremarkable, making this diagnosis less likely.
Mycobacterium leprae infections should be considered in a patient with a worsening rash and progressive polyneuropathy. Clinical diagnosis can be challenging due to similarities with other diseases; however, histopathologic findings can help differentiate M leprae from other conditions. This infection is treatable, and early detection can minimize long-term patient morbidity.
- CDC. Hansen’s disease (leprosy). Accessed April 23, 2025. https://www.cdc.gov/leprosy/about/index.html
- Fischer M. Leprosy—an overview of clinical features, diagnosis, and treatment. J Dtsch Dermatol Ges. 2017;15:801-827.
- Maymone MBC, Laughter M, Venkatesh S, et al. Leprosy: clinical aspects and diagnostic techniques. J Am Acad Dermatol. 2020; 83:1-14.
- World Health Organization. Guidelines for the diagnosis, treatment and prevention of leprosy. October 6, 2018. Accessed April 2, 2025. https://www.who.int/publications/i/item/9789290226383
- Frade MAC, Coltro PS, Filho FB, et al. Lucio’s phenomenon: a systematic literature review of definition, clinical features, histopathogenesis and management. Indian J Dermatol Venereol Leprol. 2022;88:464-477.
- Kimmel GW, Lebwohl M. Psoriasis: overview and diagnosis. In: Evidence-Based Psoriasis. Springer International Publishing; 2018:1-16.
- Griffiths CE, Barker JN. Pathogenesis and clinical features of psoriasis. Lancet. 2007;370:263-271.
- Menter A. Psoriasis and psoriatic arthritis overview. Am J Manag Care. 2016;22(8 suppl):S216-S224.
- Wu JH, Imadojemu S, Caplan AS. The evolving landscape of cutaneous sarcoidosis: pathogenic insight, clinical challenges, and new frontiers in therapy. Am J Clin Dermatol. 2022;23:499-514.
- Hill MK, Sanders CV. Cutaneous tuberculosis. Microbiol Spectr. 2017;5:1-6.
THE DIAGNOSIS: Borderline-Borderline Leprosy With Type 1 Lepra Reaction
Punch biopsies from plaques on the right elbow and right shin revealed diffuse granulomatous dermatitis (Figure 1) with a narrow Grenz zone in the superficial dermis. The upper dermis contained a dense bandlike infiltrate of histiocytes with abundant foamy-gray cytoplasm and a moderate admixture of lymphocytes. The mid and deep dermis contained a nodular, perivascular, periadnexal, and perineural infiltrate of histiocytes and a dense admixture of lymphocytes. Periodic acid-Schiff and Gram stains were negative for microorganisms. Fite stain was positive for numerous organisms in histiocytes and small dermal nerves (Figure 2). These findings and the clinical examination confirmed a diagnosis of borderline-borderline leprosy with type 1 lepra reaction. The patient was started on dapsone 100 mg, rifampin 600 mg, and clofazimine 100 mg once daily and experienced clinical improvement within 6 months.


The World Health Organization reported more than 200,000 new leprosy cases globally in 2019, with most occurring in India, Brazil, and Indonesia.1 About 150 to 250 new cases are detected in the United States annually.1 The Ridley-Jopling classification of leprosy divides the condition into 5 categories: tuberculoid, borderline tuberculoid, borderline-borderline (BB), borderline lepromatous, and lepromatous. At one end of the spectrum, tuberculoid leprosy—a predominant Th1 immune response mediated by CD4 lymphocytes, interleukin (IL) 2, and interferon gamma2—is characterized by sharply demarcated erythematous and hypopigmented plaques with raised borders and an annular appearance.2,3 Lesions typically have atrophic and hypopigmented centers that often appear in an asymmetric distribution on the arms and legs.2,3 Histologic features include dermal tuberculoid granulomas with epithelioid cells—some located directly beneath the epidermis and others around deep vessels and nerves3—multinucleated Langerhans giant cells, thickened peripheral nerves with intraneural lymphocytic infiltrates, and granulomas with central necrosis. Fite-Faraco staining exhibits few bacteria.2
Lepromatous leprosy occurs in individuals with impaired T-cell immunity, leading to multiple red-brown nodular infiltrates in the skin and mucous membranes.2,3 Lesions typically are symmetric and favor the face and auricle of the ear.2,3 Histologically, there are bluish-gray foamy macrophages that form diffuse or nodular infiltrates with few lymphocytes,2 with a Grenz zone between the epidermis and dermis. Nerves may show lamination of the perineurium resembling an onion skin.2,3 Immunohistochemistry shows predominant CD8-positive infiltrates with a Th2 response and positive IL-4 and IL-10. Fite-Faraco stain shows numerous mycobacteria arranged in clusters and in histiocytes.2
Tuberculoid leprosy is treated with dapsone 100 mg and rifampin 600 mg once daily for 6 months,4 and lepromatous leprosy is treated with dapsone 100 mg, rifampin 600 mg, and clofazimine 50 mg once daily for 12 months.4 The prognosis for both is good with treatment; erythema and induration of skin lesions may improve within a few months, but residual nerve damage is common, especially in those with advanced disease prior to treatment.2 For direct contacts, a single dose of rifampin may be given.4
Borderline-borderline leprosy manifests with numerous asymmetric annular plaques, as seen in our patient (Figure 3). Histology findings can be variable and often overlap with other forms of leprosy. There can be epithelioid granulomas and only a few acid-fast bacilli (AFB) or diffuse histiocytic aggregates with foamy histiocytes containing large numbers of AFB.3 Nerve involvement is variable but can be severe in the setting of type 1 lepra reaction, which was present in our patient. Type 1 lepra reaction—a type IV cell-mediated allergic hypersensitivity reaction to Mycobacterium leprae antigens—manifests clinically with hyperesthesia, erythema, edema, and subsequent scaling.2 It occurs in up to 30% of patients with borderline leprosy, usually within 12 months of treatment initiation.2 Our patient had considerable edema and erythema of the hands and feet (Figure 4) along with extensive polyneuropathy prior to starting therapy.


Lucio phenomenon is a rare leprosy reaction found in patients with untreated lepromatous leprosy characterized by erythematous to violaceous macules that lead to ulceronecrotic lesions.5 Histologically, there are many AFB in the vascular endothelium, leukocytoclastic vasculitis, and ischemic epidermal necrosis.5 Our patient did not have ulcerative or necrotic lesions.
The classic skin lesions of psoriasis vulgaris can be described as well-demarcated pink plaques with white or silvery scales that usually are distributed symmetrically and often are found on extensor surfaces.6 Rapidly progressive lesions can be annular with normal skin in the center, mimicking the lesions seen in tuberculoid leprosy. Clinically, both psoriasis and tuberculoid forms of leprosy are sharply demarcated; however, psoriatic lesions often have micaceous overlying scale that is not present in leprosy. Characteristic histologic findings of psoriasis are hyperkeratosis, parakeratosis, and acanthosis of the epidermis with dilated blood vessels and a lymphocytic infiltrate, predominantly into the dermis.7 Psoriatic arthritis has a variable clinical course but tends to emerge 5 to 12 years after initial skin manifestation.8 Classic clinical symptoms include swelling, tenderness, stiffness, and pain in joints and surrounding tissues.8 Other than edema, our patient did not exhibit signs of psoriatic arthritis.
Sarcoidosis is a systemic autoimmune disease characterized by noncaseating epithelioid granulomas affecting various organs, with cutaneous manifestations present in approximately 30% of all cases. Cutaneous manifestations can be variable, including maculopapular lesions, plaques, and nodules.9 Differentiating between cutaneous sarcoidosis and tuberculoid leprosy can be challenging, as both are granulomatous processes; however, histology of sarcoidosis demonstrates noncaseating granulomas in the dermis and/or subcutaneous tissues without AFB9 compared to granulomas with necrotic centers in tuberculoid leprosy.
Cutaneous tuberculosis has variable morphologies. One subtype, lupus vulgaris, can manifest with violaceous, scaly, eroded plaques that could be confused for leprosy. Lupus vulgaris usually results from hematogenous or lymphatic seeding in individuals with high or moderate immunity to M tuberculosis.10
Histologically, the dermis has tuberculoid granulomas containing multinucleated giant cells,10 which can mimic those seen in BB leprosy. Tuberculin skin test results often are positive10; while this test was not performed in our patient, chest radiography was unremarkable, making this diagnosis less likely.
Mycobacterium leprae infections should be considered in a patient with a worsening rash and progressive polyneuropathy. Clinical diagnosis can be challenging due to similarities with other diseases; however, histopathologic findings can help differentiate M leprae from other conditions. This infection is treatable, and early detection can minimize long-term patient morbidity.
THE DIAGNOSIS: Borderline-Borderline Leprosy With Type 1 Lepra Reaction
Punch biopsies from plaques on the right elbow and right shin revealed diffuse granulomatous dermatitis (Figure 1) with a narrow Grenz zone in the superficial dermis. The upper dermis contained a dense bandlike infiltrate of histiocytes with abundant foamy-gray cytoplasm and a moderate admixture of lymphocytes. The mid and deep dermis contained a nodular, perivascular, periadnexal, and perineural infiltrate of histiocytes and a dense admixture of lymphocytes. Periodic acid-Schiff and Gram stains were negative for microorganisms. Fite stain was positive for numerous organisms in histiocytes and small dermal nerves (Figure 2). These findings and the clinical examination confirmed a diagnosis of borderline-borderline leprosy with type 1 lepra reaction. The patient was started on dapsone 100 mg, rifampin 600 mg, and clofazimine 100 mg once daily and experienced clinical improvement within 6 months.


The World Health Organization reported more than 200,000 new leprosy cases globally in 2019, with most occurring in India, Brazil, and Indonesia.1 About 150 to 250 new cases are detected in the United States annually.1 The Ridley-Jopling classification of leprosy divides the condition into 5 categories: tuberculoid, borderline tuberculoid, borderline-borderline (BB), borderline lepromatous, and lepromatous. At one end of the spectrum, tuberculoid leprosy—a predominant Th1 immune response mediated by CD4 lymphocytes, interleukin (IL) 2, and interferon gamma2—is characterized by sharply demarcated erythematous and hypopigmented plaques with raised borders and an annular appearance.2,3 Lesions typically have atrophic and hypopigmented centers that often appear in an asymmetric distribution on the arms and legs.2,3 Histologic features include dermal tuberculoid granulomas with epithelioid cells—some located directly beneath the epidermis and others around deep vessels and nerves3—multinucleated Langerhans giant cells, thickened peripheral nerves with intraneural lymphocytic infiltrates, and granulomas with central necrosis. Fite-Faraco staining exhibits few bacteria.2
Lepromatous leprosy occurs in individuals with impaired T-cell immunity, leading to multiple red-brown nodular infiltrates in the skin and mucous membranes.2,3 Lesions typically are symmetric and favor the face and auricle of the ear.2,3 Histologically, there are bluish-gray foamy macrophages that form diffuse or nodular infiltrates with few lymphocytes,2 with a Grenz zone between the epidermis and dermis. Nerves may show lamination of the perineurium resembling an onion skin.2,3 Immunohistochemistry shows predominant CD8-positive infiltrates with a Th2 response and positive IL-4 and IL-10. Fite-Faraco stain shows numerous mycobacteria arranged in clusters and in histiocytes.2
Tuberculoid leprosy is treated with dapsone 100 mg and rifampin 600 mg once daily for 6 months,4 and lepromatous leprosy is treated with dapsone 100 mg, rifampin 600 mg, and clofazimine 50 mg once daily for 12 months.4 The prognosis for both is good with treatment; erythema and induration of skin lesions may improve within a few months, but residual nerve damage is common, especially in those with advanced disease prior to treatment.2 For direct contacts, a single dose of rifampin may be given.4
Borderline-borderline leprosy manifests with numerous asymmetric annular plaques, as seen in our patient (Figure 3). Histology findings can be variable and often overlap with other forms of leprosy. There can be epithelioid granulomas and only a few acid-fast bacilli (AFB) or diffuse histiocytic aggregates with foamy histiocytes containing large numbers of AFB.3 Nerve involvement is variable but can be severe in the setting of type 1 lepra reaction, which was present in our patient. Type 1 lepra reaction—a type IV cell-mediated allergic hypersensitivity reaction to Mycobacterium leprae antigens—manifests clinically with hyperesthesia, erythema, edema, and subsequent scaling.2 It occurs in up to 30% of patients with borderline leprosy, usually within 12 months of treatment initiation.2 Our patient had considerable edema and erythema of the hands and feet (Figure 4) along with extensive polyneuropathy prior to starting therapy.


Lucio phenomenon is a rare leprosy reaction found in patients with untreated lepromatous leprosy characterized by erythematous to violaceous macules that lead to ulceronecrotic lesions.5 Histologically, there are many AFB in the vascular endothelium, leukocytoclastic vasculitis, and ischemic epidermal necrosis.5 Our patient did not have ulcerative or necrotic lesions.
The classic skin lesions of psoriasis vulgaris can be described as well-demarcated pink plaques with white or silvery scales that usually are distributed symmetrically and often are found on extensor surfaces.6 Rapidly progressive lesions can be annular with normal skin in the center, mimicking the lesions seen in tuberculoid leprosy. Clinically, both psoriasis and tuberculoid forms of leprosy are sharply demarcated; however, psoriatic lesions often have micaceous overlying scale that is not present in leprosy. Characteristic histologic findings of psoriasis are hyperkeratosis, parakeratosis, and acanthosis of the epidermis with dilated blood vessels and a lymphocytic infiltrate, predominantly into the dermis.7 Psoriatic arthritis has a variable clinical course but tends to emerge 5 to 12 years after initial skin manifestation.8 Classic clinical symptoms include swelling, tenderness, stiffness, and pain in joints and surrounding tissues.8 Other than edema, our patient did not exhibit signs of psoriatic arthritis.
Sarcoidosis is a systemic autoimmune disease characterized by noncaseating epithelioid granulomas affecting various organs, with cutaneous manifestations present in approximately 30% of all cases. Cutaneous manifestations can be variable, including maculopapular lesions, plaques, and nodules.9 Differentiating between cutaneous sarcoidosis and tuberculoid leprosy can be challenging, as both are granulomatous processes; however, histology of sarcoidosis demonstrates noncaseating granulomas in the dermis and/or subcutaneous tissues without AFB9 compared to granulomas with necrotic centers in tuberculoid leprosy.
Cutaneous tuberculosis has variable morphologies. One subtype, lupus vulgaris, can manifest with violaceous, scaly, eroded plaques that could be confused for leprosy. Lupus vulgaris usually results from hematogenous or lymphatic seeding in individuals with high or moderate immunity to M tuberculosis.10
Histologically, the dermis has tuberculoid granulomas containing multinucleated giant cells,10 which can mimic those seen in BB leprosy. Tuberculin skin test results often are positive10; while this test was not performed in our patient, chest radiography was unremarkable, making this diagnosis less likely.
Mycobacterium leprae infections should be considered in a patient with a worsening rash and progressive polyneuropathy. Clinical diagnosis can be challenging due to similarities with other diseases; however, histopathologic findings can help differentiate M leprae from other conditions. This infection is treatable, and early detection can minimize long-term patient morbidity.
- CDC. Hansen’s disease (leprosy). Accessed April 23, 2025. https://www.cdc.gov/leprosy/about/index.html
- Fischer M. Leprosy—an overview of clinical features, diagnosis, and treatment. J Dtsch Dermatol Ges. 2017;15:801-827.
- Maymone MBC, Laughter M, Venkatesh S, et al. Leprosy: clinical aspects and diagnostic techniques. J Am Acad Dermatol. 2020; 83:1-14.
- World Health Organization. Guidelines for the diagnosis, treatment and prevention of leprosy. October 6, 2018. Accessed April 2, 2025. https://www.who.int/publications/i/item/9789290226383
- Frade MAC, Coltro PS, Filho FB, et al. Lucio’s phenomenon: a systematic literature review of definition, clinical features, histopathogenesis and management. Indian J Dermatol Venereol Leprol. 2022;88:464-477.
- Kimmel GW, Lebwohl M. Psoriasis: overview and diagnosis. In: Evidence-Based Psoriasis. Springer International Publishing; 2018:1-16.
- Griffiths CE, Barker JN. Pathogenesis and clinical features of psoriasis. Lancet. 2007;370:263-271.
- Menter A. Psoriasis and psoriatic arthritis overview. Am J Manag Care. 2016;22(8 suppl):S216-S224.
- Wu JH, Imadojemu S, Caplan AS. The evolving landscape of cutaneous sarcoidosis: pathogenic insight, clinical challenges, and new frontiers in therapy. Am J Clin Dermatol. 2022;23:499-514.
- Hill MK, Sanders CV. Cutaneous tuberculosis. Microbiol Spectr. 2017;5:1-6.
- CDC. Hansen’s disease (leprosy). Accessed April 23, 2025. https://www.cdc.gov/leprosy/about/index.html
- Fischer M. Leprosy—an overview of clinical features, diagnosis, and treatment. J Dtsch Dermatol Ges. 2017;15:801-827.
- Maymone MBC, Laughter M, Venkatesh S, et al. Leprosy: clinical aspects and diagnostic techniques. J Am Acad Dermatol. 2020; 83:1-14.
- World Health Organization. Guidelines for the diagnosis, treatment and prevention of leprosy. October 6, 2018. Accessed April 2, 2025. https://www.who.int/publications/i/item/9789290226383
- Frade MAC, Coltro PS, Filho FB, et al. Lucio’s phenomenon: a systematic literature review of definition, clinical features, histopathogenesis and management. Indian J Dermatol Venereol Leprol. 2022;88:464-477.
- Kimmel GW, Lebwohl M. Psoriasis: overview and diagnosis. In: Evidence-Based Psoriasis. Springer International Publishing; 2018:1-16.
- Griffiths CE, Barker JN. Pathogenesis and clinical features of psoriasis. Lancet. 2007;370:263-271.
- Menter A. Psoriasis and psoriatic arthritis overview. Am J Manag Care. 2016;22(8 suppl):S216-S224.
- Wu JH, Imadojemu S, Caplan AS. The evolving landscape of cutaneous sarcoidosis: pathogenic insight, clinical challenges, and new frontiers in therapy. Am J Clin Dermatol. 2022;23:499-514.
- Hill MK, Sanders CV. Cutaneous tuberculosis. Microbiol Spectr. 2017;5:1-6.
Acral Erythema, Edema, and Scaly Plaques in a Patient With Polyneuropathy
Acral Erythema, Edema, and Scaly Plaques in a Patient With Polyneuropathy
A 67-year-old man presented to his primary care physician with scaly plaques on the extensor surfaces along with distal neuropathy that had been slowly worsening over the past 6 months. The patient was prescribed triamcinolone cream 0.1% twice daily for presumed atopic dermatitis. Three months later, his symptoms rapidly worsened and he developed edema of the hands and feet. He was seen by neurology, and electromyography revealed severe distal sensorimotor neuropathy, prompting hospital admission for further evaluation of a potential rapidly progressive autoimmune disease. Laboratory workup and imaging were ordered, and the patient began an intravenous course of methylprednisolone. Minimal improvement in his symptoms was noted after 1 day, at which time dermatology was consulted.
Physical examination by dermatology revealed well-defined plaques with annular scale on extensor surfaces of the arms and legs, and edema on the hands and feet as well as distal sensorimotor neuropathy. The patient reported associated unspecified weight loss but denied any chest pain, shortness of breath, fevers, chills, cough, night sweats, exposure to chemicals, or recent travel. He reported that he had immigrated from India 37 years prior; his last visit to India was 6 years ago. He currently was taking famotidine for gastrointestinal reflux disease and losartan for hypertension. There was no personal or family history of autoimmune diseases. A complete workup for hematologic, thyroid, liver, and renal function was unremarkable. Initial autoimmune workup was negative for antinuclear antibodies and rheumatoid factor. Serum protein electrophoresis was normal. Results of testing for HIV, hepatitis B, and hepatitis C were negative. Chest radiography was unremarkable. Erythrocyte sedimentation rate and C-reactive protein level were elevated.
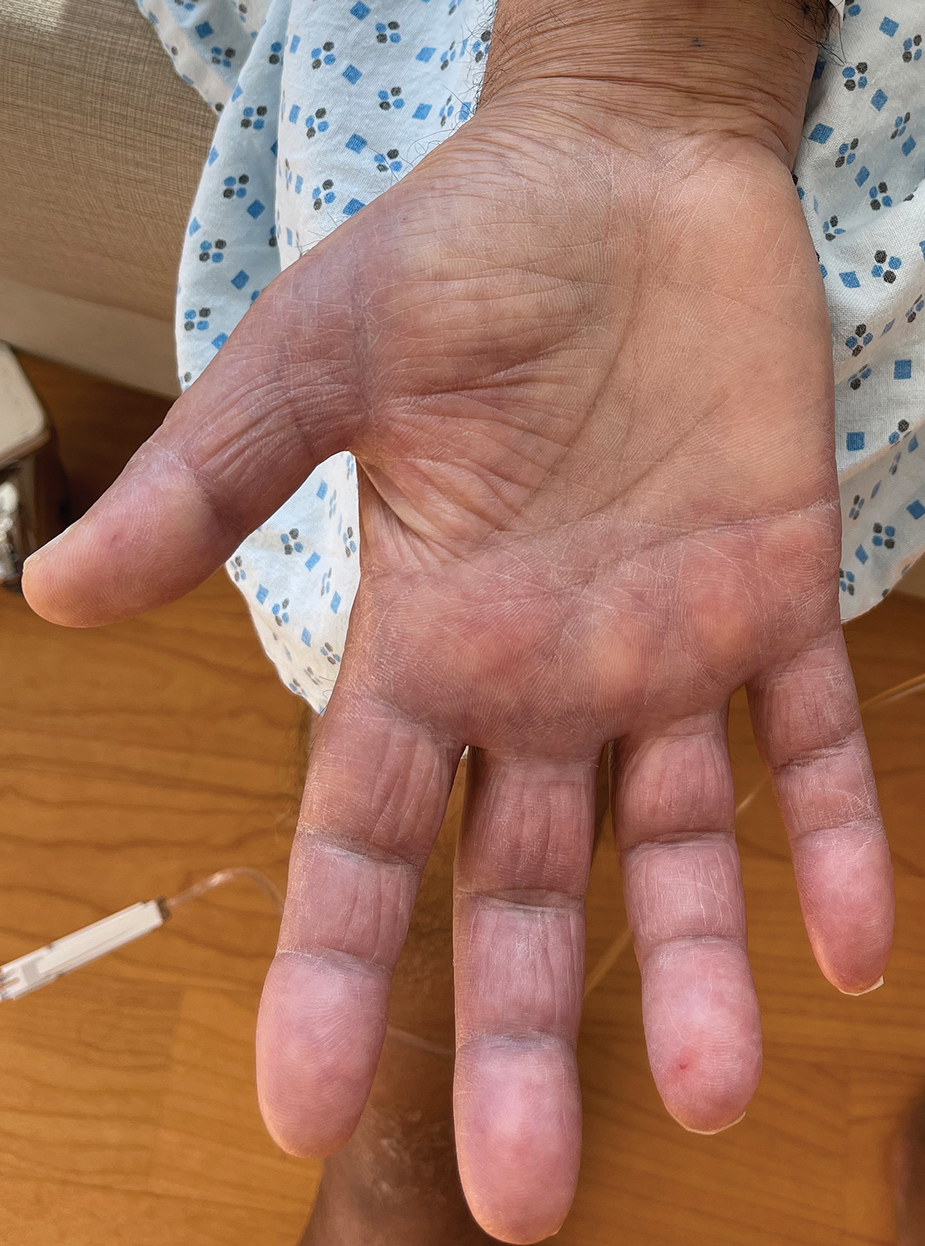
Dermatologists’ Perspectives Toward Disability Assessment: A Nationwide Survey Report
Dermatologists’ Perspectives Toward Disability Assessment: A Nationwide Survey Report
To the Editor:
Cutaneous medical conditions can have a substantial impact on patients’ functioning and quality of life. Many patients with severe skin disease are eligible to receive disability assistance that can provide them with essential income and health care. Previous research has highlighted disability assessment as one of the most important ways physicians can help mitigate the health consequences of poverty.1 Dermatologists can play an important role in the disability assessment process by documenting the facts associated with patients’ skin conditions.
Although skin conditions have a relatively high prevalence, they remain underrepresented in disability claims. Between 1997 and 2004, occupational skin diseases accounted for 12% to 17% of nonfatal work-related illnesses; however, during that same period, skin conditions comprised only 0.21% of disability claims in the United States.2,3 Historically, there has been hesitancy among dermatologists to complete disability paperwork; a 1976 survey of dermatologists cited extensive paperwork, “troublesome patients,” and fee schedule issues as reasons.4 The lack of training regarding disability assessment in medical school and residency also has been noted.5
To characterize modern attitudes toward disability assessments, we conducted a survey of dermatologists across the United States. Our study was reviewed and declared exempt by the institutional review board of the Lundquist Institute for Biomedical Innovation at Harbor-UCLA Medical Center (Torrance, California)(approval #18CR-32242-01). Using convenience sampling, we emailed dermatologists from the Association of Professors of Dermatology and dermatology state societies in all 50 states inviting them to participate in our voluntary and anonymous survey, which was administered using SurveyMonkey. The use of all society mailing lists was approved by the respective owners. The 15-question survey included multiple choice, Likert scale, and free response sections. Summary and descriptive statistics were used to describe respondent demographics and identify any patterns in responses.
For each Likert-based question, participants ranked their degree of agreement with a statement as: 1=strongly disagree, 2=somewhat disagree, 3=neither agree nor disagree/neutral, 4=somewhat agree, and 5=strongly agree. The mean response and standard deviation were reported for each Likert scale prompt. Preplanned 1-sample t testing was used to analyze Likert scale data, in which the mean response for each prompt was compared to a baseline response of 3 (neutral). A P value <.05 was considered statistically significant. Statistical analyses were performed using SPSS Statistics for MacOS, version 27 (IBM).
Seventy-eight dermatologists agreed to participate, and 70 completed the survey, for a response rate of 89.7% (Table 1). The dermatologists we surveyed practiced in a variety of clinical settings, including academic public hospitals (46.2% [36/78]), academic private hospitals (33.3% [26/78]), and private practices (32.1% [25/78]), and 60.3% (47/78) reported providing disability documentation at some point. Most of the respondents (64.3% [45/70]) did not perform assessments in an average month (Table 2). Medical assessment documentation was provided most frequently for workers’ compensation (50.0% [35/70]), private insurance (27.1% [19/70]), and Social Security Disability Insurance (25.7% [18/70]). Dermatologists overwhelmingly reported no formal training for disability assessment in medical school (94.3% [66/70]), residency (97.1% [68/70]), or clinical practice (81.4% [57/70]).


In the Likert scale prompts, respondents agreed that they were uncertain of their role in disability assessment (mean response, 3.6; P<.001). Moreover, they were uncomfortable providing assessments (mean response, 3.5; P<.001) and felt that they did not have sufficient time to perform them (mean response, 3.6; P<.001). Dermatologists disagreed that they received adequate compensation for performing assessments (mean response, 2.2; P<.001) and felt that they did not have enough time to participate in assessments (mean response, 3.6; P<.001). Respondents generally did not feel distrustful of patients seeking disability assessment (mean response, 2.8; P=.043). Dermatologists neither agreed nor disagreed when asked if they thought that physicians can determine disability status (mean response, 3.2; P=.118). The details of the Likert scale responses are described in Table 3. Respondents also were uncertain as to which dermatologic conditions were eligible for disability. When asked to select which conditions from a list of 10 were eligible per the Social Security Administration listing of disability impairments, only 15.4% (12/70) of respondents correctly identified that all the conditions qualified; these included ichthyosis, pemphigus vulgaris, allergic contact dermatitis, hidradenitis suppurativa, systemic lupus erythematosus, chromoblastomycosis, xeroderma pigmentosum, burns, malignant melanoma, and scleroderma.6

In the free-response prompts, respondents frequently described extensive paperwork, inadequate time, and lack of reimbursement as barriers to providing documentation. Often, dermatologists found that the forms were not well matched to the skin conditions they were evaluating and rather had a musculoskeletal focus. Multiple individuals commented on the challenge in assessing the percentage of disability and functional/psychosocial impairment in skin conditions. One respondent noted that workers’ compensation forms ask if the patient is “…permanent and stationary…for most conditions this has no meaning in dermatology.” Some felt hesitant to provide documentation because they had insufficient patient history, especially regarding employment, and opted to defer to primary care providers who might be more familiar with the full patient history.
A dermatologist described their perspective as follows:
“…As a specialist I feel that I don’t have a complete look into all the factors that could contribute to a patient[’]s need to go on disability, and I don’t have experience with filling out disability requests. That being said, if a patient[’]s request for disability was due to a skin disease that I know way more about than [a] primary care [physician] would, I would do the disability assessment.”
Another respondent noted the complexity in “establishing causality” for workers’ compensation. Another dermatologist reported,
“The most frequent challenging situation I encounter is being asked to evaluate for maximum medical improvement after patch testing. If the patient is not fully avoiding contact allergens either at home or at work, then I typically document that they are not at [maximum medical improvement]. The reality is that most frequently it is due to exposure to allergens at home so the line between what is a legitimate worker’s comp[ensation] issue and what is a home life choice is blurry.”
Nevertheless, respondents expressed interest in learning more about disability assessment procedures. Summary guides, lectures, and prefilled paperwork were the most popular initiatives that respondents agreed would be beneficial toward becoming educated regarding disability assessment (78.6%, 58.6%, and 58.6%, respectively)(Table 2). One respondent noted that “previous [internal medicine] history help[ed]” them in performing cutaneous disability assessments.
As with any survey, our study did have some inherent limitations. Only a relatively small sample size was willing to complete the survey. There was a predominance of respondents from California (34.6% [27/78]), as well as those practicing for less than 15 years (58.9% [46/78])(Figure). This could limit generalizability to the national population of dermatologists. In addition, there was potential for recall bias and errors in responding given the self-reported nature of the study. Different individuals may interpret the Likert scale options in various ways, which could skew results unintentionally. However, the survey was largely qualitative in nature, making it a legitimate tool for answering our research questions. Moreover, we were able to hear the perspectives of dermatologists across diverse practice settings, with free response prompts to increase the depth of the survey.
Almost 50 years later, our survey echoes common themes from Adams’ 1976 survey.4 Inadequate compensation, limited time, and burdensome paperwork all continue to hinder dermatologists’ ability to perform disability assessments. Our participants frequently commented that the current disability forms are not congruent with the nature of skin conditions, making it challenging to accurately document the facts.
Moreover, respondents felt uncertain in their role in disability assessment and occasionally noted distrust of patients or insufficient patient history as barriers to completing assessments. They also were unsure if physicians can grant disability status. This is a common misconception among physicians that leads to discomfort in helping with disability assessment.7 The role of physicians in disability assessment is to document the facts of a patient’s illness, not to determine whether they are eligible for benefits. We discovered uncertainty in our respondents’ ability to identify conditions eligible for disability, highlighting an area in need of greater education for physicians.
Despite these obstacles, respondents were interested in learning more about disability assessment and highlighted several practical approaches that could help them better perform this task. As skin specialists, dermatologists are the best-equipped physicians to assess cutaneous conditions and should play a greater role in performing disability assessments, which could be achieved through increased educational initiatives and individual physician motivation.7 We call for greater collaboration and reflection on the importance of disability assistance among dermatologists to increase participation in the disability-assessment process.
- O’Connell JJ, Zevin BD, Quick PD, et al. Documenting disability: simple strategies for medical providers. Health Care for the Homeless Clinicians’ Network. September 2007. Accessed March 31, 2025. https://nhchc.org/wp-content/uploads/2019/08/DocumentingDisability2007.pdf
- US Bureau of Labor Statistics. Injuries, illnesses, and fatalities. Accessed March 31, 2025. https://www.bls.gov/iif/
- Meseguer J. Outcome variation in the Social Security Disability Insurance Program: the role of primary diagnoses. Soc Secur Bull. 2013;73:39-75.
- Adams RM. Attitudes of California dermatologists toward Worker’s Compensation: results of a survey. West J Med. 1976;125:169-175.
- Talmage J, Melhorn J, Hyman M. AMA Guides to the Evaluation of Work Ability and Return to Work. 2nd ed. American Medical Association; 2011.
- Social Security Administration. Disability evaluation under Social Security. 8.00 skin disorders - adult. March 31, 2025. https://www.ssa.gov/disability/professionals/bluebook/8.00-Skin-Adult.htm
- Dawson J, Smogorzewski J. Demystifying disability assessments for dermatologists—a call to action. JAMA Dermatol. 2021;157:903-904. doi:10.1001/jamadermatol.2021.1767
To the Editor:
Cutaneous medical conditions can have a substantial impact on patients’ functioning and quality of life. Many patients with severe skin disease are eligible to receive disability assistance that can provide them with essential income and health care. Previous research has highlighted disability assessment as one of the most important ways physicians can help mitigate the health consequences of poverty.1 Dermatologists can play an important role in the disability assessment process by documenting the facts associated with patients’ skin conditions.
Although skin conditions have a relatively high prevalence, they remain underrepresented in disability claims. Between 1997 and 2004, occupational skin diseases accounted for 12% to 17% of nonfatal work-related illnesses; however, during that same period, skin conditions comprised only 0.21% of disability claims in the United States.2,3 Historically, there has been hesitancy among dermatologists to complete disability paperwork; a 1976 survey of dermatologists cited extensive paperwork, “troublesome patients,” and fee schedule issues as reasons.4 The lack of training regarding disability assessment in medical school and residency also has been noted.5
To characterize modern attitudes toward disability assessments, we conducted a survey of dermatologists across the United States. Our study was reviewed and declared exempt by the institutional review board of the Lundquist Institute for Biomedical Innovation at Harbor-UCLA Medical Center (Torrance, California)(approval #18CR-32242-01). Using convenience sampling, we emailed dermatologists from the Association of Professors of Dermatology and dermatology state societies in all 50 states inviting them to participate in our voluntary and anonymous survey, which was administered using SurveyMonkey. The use of all society mailing lists was approved by the respective owners. The 15-question survey included multiple choice, Likert scale, and free response sections. Summary and descriptive statistics were used to describe respondent demographics and identify any patterns in responses.
For each Likert-based question, participants ranked their degree of agreement with a statement as: 1=strongly disagree, 2=somewhat disagree, 3=neither agree nor disagree/neutral, 4=somewhat agree, and 5=strongly agree. The mean response and standard deviation were reported for each Likert scale prompt. Preplanned 1-sample t testing was used to analyze Likert scale data, in which the mean response for each prompt was compared to a baseline response of 3 (neutral). A P value <.05 was considered statistically significant. Statistical analyses were performed using SPSS Statistics for MacOS, version 27 (IBM).
Seventy-eight dermatologists agreed to participate, and 70 completed the survey, for a response rate of 89.7% (Table 1). The dermatologists we surveyed practiced in a variety of clinical settings, including academic public hospitals (46.2% [36/78]), academic private hospitals (33.3% [26/78]), and private practices (32.1% [25/78]), and 60.3% (47/78) reported providing disability documentation at some point. Most of the respondents (64.3% [45/70]) did not perform assessments in an average month (Table 2). Medical assessment documentation was provided most frequently for workers’ compensation (50.0% [35/70]), private insurance (27.1% [19/70]), and Social Security Disability Insurance (25.7% [18/70]). Dermatologists overwhelmingly reported no formal training for disability assessment in medical school (94.3% [66/70]), residency (97.1% [68/70]), or clinical practice (81.4% [57/70]).


In the Likert scale prompts, respondents agreed that they were uncertain of their role in disability assessment (mean response, 3.6; P<.001). Moreover, they were uncomfortable providing assessments (mean response, 3.5; P<.001) and felt that they did not have sufficient time to perform them (mean response, 3.6; P<.001). Dermatologists disagreed that they received adequate compensation for performing assessments (mean response, 2.2; P<.001) and felt that they did not have enough time to participate in assessments (mean response, 3.6; P<.001). Respondents generally did not feel distrustful of patients seeking disability assessment (mean response, 2.8; P=.043). Dermatologists neither agreed nor disagreed when asked if they thought that physicians can determine disability status (mean response, 3.2; P=.118). The details of the Likert scale responses are described in Table 3. Respondents also were uncertain as to which dermatologic conditions were eligible for disability. When asked to select which conditions from a list of 10 were eligible per the Social Security Administration listing of disability impairments, only 15.4% (12/70) of respondents correctly identified that all the conditions qualified; these included ichthyosis, pemphigus vulgaris, allergic contact dermatitis, hidradenitis suppurativa, systemic lupus erythematosus, chromoblastomycosis, xeroderma pigmentosum, burns, malignant melanoma, and scleroderma.6

In the free-response prompts, respondents frequently described extensive paperwork, inadequate time, and lack of reimbursement as barriers to providing documentation. Often, dermatologists found that the forms were not well matched to the skin conditions they were evaluating and rather had a musculoskeletal focus. Multiple individuals commented on the challenge in assessing the percentage of disability and functional/psychosocial impairment in skin conditions. One respondent noted that workers’ compensation forms ask if the patient is “…permanent and stationary…for most conditions this has no meaning in dermatology.” Some felt hesitant to provide documentation because they had insufficient patient history, especially regarding employment, and opted to defer to primary care providers who might be more familiar with the full patient history.
A dermatologist described their perspective as follows:
“…As a specialist I feel that I don’t have a complete look into all the factors that could contribute to a patient[’]s need to go on disability, and I don’t have experience with filling out disability requests. That being said, if a patient[’]s request for disability was due to a skin disease that I know way more about than [a] primary care [physician] would, I would do the disability assessment.”
Another respondent noted the complexity in “establishing causality” for workers’ compensation. Another dermatologist reported,
“The most frequent challenging situation I encounter is being asked to evaluate for maximum medical improvement after patch testing. If the patient is not fully avoiding contact allergens either at home or at work, then I typically document that they are not at [maximum medical improvement]. The reality is that most frequently it is due to exposure to allergens at home so the line between what is a legitimate worker’s comp[ensation] issue and what is a home life choice is blurry.”
Nevertheless, respondents expressed interest in learning more about disability assessment procedures. Summary guides, lectures, and prefilled paperwork were the most popular initiatives that respondents agreed would be beneficial toward becoming educated regarding disability assessment (78.6%, 58.6%, and 58.6%, respectively)(Table 2). One respondent noted that “previous [internal medicine] history help[ed]” them in performing cutaneous disability assessments.
As with any survey, our study did have some inherent limitations. Only a relatively small sample size was willing to complete the survey. There was a predominance of respondents from California (34.6% [27/78]), as well as those practicing for less than 15 years (58.9% [46/78])(Figure). This could limit generalizability to the national population of dermatologists. In addition, there was potential for recall bias and errors in responding given the self-reported nature of the study. Different individuals may interpret the Likert scale options in various ways, which could skew results unintentionally. However, the survey was largely qualitative in nature, making it a legitimate tool for answering our research questions. Moreover, we were able to hear the perspectives of dermatologists across diverse practice settings, with free response prompts to increase the depth of the survey.

Almost 50 years later, our survey echoes common themes from Adams’ 1976 survey.4 Inadequate compensation, limited time, and burdensome paperwork all continue to hinder dermatologists’ ability to perform disability assessments. Our participants frequently commented that the current disability forms are not congruent with the nature of skin conditions, making it challenging to accurately document the facts.
Moreover, respondents felt uncertain in their role in disability assessment and occasionally noted distrust of patients or insufficient patient history as barriers to completing assessments. They also were unsure if physicians can grant disability status. This is a common misconception among physicians that leads to discomfort in helping with disability assessment.7 The role of physicians in disability assessment is to document the facts of a patient’s illness, not to determine whether they are eligible for benefits. We discovered uncertainty in our respondents’ ability to identify conditions eligible for disability, highlighting an area in need of greater education for physicians.
Despite these obstacles, respondents were interested in learning more about disability assessment and highlighted several practical approaches that could help them better perform this task. As skin specialists, dermatologists are the best-equipped physicians to assess cutaneous conditions and should play a greater role in performing disability assessments, which could be achieved through increased educational initiatives and individual physician motivation.7 We call for greater collaboration and reflection on the importance of disability assistance among dermatologists to increase participation in the disability-assessment process.
To the Editor:
Cutaneous medical conditions can have a substantial impact on patients’ functioning and quality of life. Many patients with severe skin disease are eligible to receive disability assistance that can provide them with essential income and health care. Previous research has highlighted disability assessment as one of the most important ways physicians can help mitigate the health consequences of poverty.1 Dermatologists can play an important role in the disability assessment process by documenting the facts associated with patients’ skin conditions.
Although skin conditions have a relatively high prevalence, they remain underrepresented in disability claims. Between 1997 and 2004, occupational skin diseases accounted for 12% to 17% of nonfatal work-related illnesses; however, during that same period, skin conditions comprised only 0.21% of disability claims in the United States.2,3 Historically, there has been hesitancy among dermatologists to complete disability paperwork; a 1976 survey of dermatologists cited extensive paperwork, “troublesome patients,” and fee schedule issues as reasons.4 The lack of training regarding disability assessment in medical school and residency also has been noted.5
To characterize modern attitudes toward disability assessments, we conducted a survey of dermatologists across the United States. Our study was reviewed and declared exempt by the institutional review board of the Lundquist Institute for Biomedical Innovation at Harbor-UCLA Medical Center (Torrance, California)(approval #18CR-32242-01). Using convenience sampling, we emailed dermatologists from the Association of Professors of Dermatology and dermatology state societies in all 50 states inviting them to participate in our voluntary and anonymous survey, which was administered using SurveyMonkey. The use of all society mailing lists was approved by the respective owners. The 15-question survey included multiple choice, Likert scale, and free response sections. Summary and descriptive statistics were used to describe respondent demographics and identify any patterns in responses.
For each Likert-based question, participants ranked their degree of agreement with a statement as: 1=strongly disagree, 2=somewhat disagree, 3=neither agree nor disagree/neutral, 4=somewhat agree, and 5=strongly agree. The mean response and standard deviation were reported for each Likert scale prompt. Preplanned 1-sample t testing was used to analyze Likert scale data, in which the mean response for each prompt was compared to a baseline response of 3 (neutral). A P value <.05 was considered statistically significant. Statistical analyses were performed using SPSS Statistics for MacOS, version 27 (IBM).
Seventy-eight dermatologists agreed to participate, and 70 completed the survey, for a response rate of 89.7% (Table 1). The dermatologists we surveyed practiced in a variety of clinical settings, including academic public hospitals (46.2% [36/78]), academic private hospitals (33.3% [26/78]), and private practices (32.1% [25/78]), and 60.3% (47/78) reported providing disability documentation at some point. Most of the respondents (64.3% [45/70]) did not perform assessments in an average month (Table 2). Medical assessment documentation was provided most frequently for workers’ compensation (50.0% [35/70]), private insurance (27.1% [19/70]), and Social Security Disability Insurance (25.7% [18/70]). Dermatologists overwhelmingly reported no formal training for disability assessment in medical school (94.3% [66/70]), residency (97.1% [68/70]), or clinical practice (81.4% [57/70]).


In the Likert scale prompts, respondents agreed that they were uncertain of their role in disability assessment (mean response, 3.6; P<.001). Moreover, they were uncomfortable providing assessments (mean response, 3.5; P<.001) and felt that they did not have sufficient time to perform them (mean response, 3.6; P<.001). Dermatologists disagreed that they received adequate compensation for performing assessments (mean response, 2.2; P<.001) and felt that they did not have enough time to participate in assessments (mean response, 3.6; P<.001). Respondents generally did not feel distrustful of patients seeking disability assessment (mean response, 2.8; P=.043). Dermatologists neither agreed nor disagreed when asked if they thought that physicians can determine disability status (mean response, 3.2; P=.118). The details of the Likert scale responses are described in Table 3. Respondents also were uncertain as to which dermatologic conditions were eligible for disability. When asked to select which conditions from a list of 10 were eligible per the Social Security Administration listing of disability impairments, only 15.4% (12/70) of respondents correctly identified that all the conditions qualified; these included ichthyosis, pemphigus vulgaris, allergic contact dermatitis, hidradenitis suppurativa, systemic lupus erythematosus, chromoblastomycosis, xeroderma pigmentosum, burns, malignant melanoma, and scleroderma.6

In the free-response prompts, respondents frequently described extensive paperwork, inadequate time, and lack of reimbursement as barriers to providing documentation. Often, dermatologists found that the forms were not well matched to the skin conditions they were evaluating and rather had a musculoskeletal focus. Multiple individuals commented on the challenge in assessing the percentage of disability and functional/psychosocial impairment in skin conditions. One respondent noted that workers’ compensation forms ask if the patient is “…permanent and stationary…for most conditions this has no meaning in dermatology.” Some felt hesitant to provide documentation because they had insufficient patient history, especially regarding employment, and opted to defer to primary care providers who might be more familiar with the full patient history.
A dermatologist described their perspective as follows:
“…As a specialist I feel that I don’t have a complete look into all the factors that could contribute to a patient[’]s need to go on disability, and I don’t have experience with filling out disability requests. That being said, if a patient[’]s request for disability was due to a skin disease that I know way more about than [a] primary care [physician] would, I would do the disability assessment.”
Another respondent noted the complexity in “establishing causality” for workers’ compensation. Another dermatologist reported,
“The most frequent challenging situation I encounter is being asked to evaluate for maximum medical improvement after patch testing. If the patient is not fully avoiding contact allergens either at home or at work, then I typically document that they are not at [maximum medical improvement]. The reality is that most frequently it is due to exposure to allergens at home so the line between what is a legitimate worker’s comp[ensation] issue and what is a home life choice is blurry.”
Nevertheless, respondents expressed interest in learning more about disability assessment procedures. Summary guides, lectures, and prefilled paperwork were the most popular initiatives that respondents agreed would be beneficial toward becoming educated regarding disability assessment (78.6%, 58.6%, and 58.6%, respectively)(Table 2). One respondent noted that “previous [internal medicine] history help[ed]” them in performing cutaneous disability assessments.
As with any survey, our study did have some inherent limitations. Only a relatively small sample size was willing to complete the survey. There was a predominance of respondents from California (34.6% [27/78]), as well as those practicing for less than 15 years (58.9% [46/78])(Figure). This could limit generalizability to the national population of dermatologists. In addition, there was potential for recall bias and errors in responding given the self-reported nature of the study. Different individuals may interpret the Likert scale options in various ways, which could skew results unintentionally. However, the survey was largely qualitative in nature, making it a legitimate tool for answering our research questions. Moreover, we were able to hear the perspectives of dermatologists across diverse practice settings, with free response prompts to increase the depth of the survey.

Almost 50 years later, our survey echoes common themes from Adams’ 1976 survey.4 Inadequate compensation, limited time, and burdensome paperwork all continue to hinder dermatologists’ ability to perform disability assessments. Our participants frequently commented that the current disability forms are not congruent with the nature of skin conditions, making it challenging to accurately document the facts.
Moreover, respondents felt uncertain in their role in disability assessment and occasionally noted distrust of patients or insufficient patient history as barriers to completing assessments. They also were unsure if physicians can grant disability status. This is a common misconception among physicians that leads to discomfort in helping with disability assessment.7 The role of physicians in disability assessment is to document the facts of a patient’s illness, not to determine whether they are eligible for benefits. We discovered uncertainty in our respondents’ ability to identify conditions eligible for disability, highlighting an area in need of greater education for physicians.
Despite these obstacles, respondents were interested in learning more about disability assessment and highlighted several practical approaches that could help them better perform this task. As skin specialists, dermatologists are the best-equipped physicians to assess cutaneous conditions and should play a greater role in performing disability assessments, which could be achieved through increased educational initiatives and individual physician motivation.7 We call for greater collaboration and reflection on the importance of disability assistance among dermatologists to increase participation in the disability-assessment process.
- O’Connell JJ, Zevin BD, Quick PD, et al. Documenting disability: simple strategies for medical providers. Health Care for the Homeless Clinicians’ Network. September 2007. Accessed March 31, 2025. https://nhchc.org/wp-content/uploads/2019/08/DocumentingDisability2007.pdf
- US Bureau of Labor Statistics. Injuries, illnesses, and fatalities. Accessed March 31, 2025. https://www.bls.gov/iif/
- Meseguer J. Outcome variation in the Social Security Disability Insurance Program: the role of primary diagnoses. Soc Secur Bull. 2013;73:39-75.
- Adams RM. Attitudes of California dermatologists toward Worker’s Compensation: results of a survey. West J Med. 1976;125:169-175.
- Talmage J, Melhorn J, Hyman M. AMA Guides to the Evaluation of Work Ability and Return to Work. 2nd ed. American Medical Association; 2011.
- Social Security Administration. Disability evaluation under Social Security. 8.00 skin disorders - adult. March 31, 2025. https://www.ssa.gov/disability/professionals/bluebook/8.00-Skin-Adult.htm
- Dawson J, Smogorzewski J. Demystifying disability assessments for dermatologists—a call to action. JAMA Dermatol. 2021;157:903-904. doi:10.1001/jamadermatol.2021.1767
- O’Connell JJ, Zevin BD, Quick PD, et al. Documenting disability: simple strategies for medical providers. Health Care for the Homeless Clinicians’ Network. September 2007. Accessed March 31, 2025. https://nhchc.org/wp-content/uploads/2019/08/DocumentingDisability2007.pdf
- US Bureau of Labor Statistics. Injuries, illnesses, and fatalities. Accessed March 31, 2025. https://www.bls.gov/iif/
- Meseguer J. Outcome variation in the Social Security Disability Insurance Program: the role of primary diagnoses. Soc Secur Bull. 2013;73:39-75.
- Adams RM. Attitudes of California dermatologists toward Worker’s Compensation: results of a survey. West J Med. 1976;125:169-175.
- Talmage J, Melhorn J, Hyman M. AMA Guides to the Evaluation of Work Ability and Return to Work. 2nd ed. American Medical Association; 2011.
- Social Security Administration. Disability evaluation under Social Security. 8.00 skin disorders - adult. March 31, 2025. https://www.ssa.gov/disability/professionals/bluebook/8.00-Skin-Adult.htm
- Dawson J, Smogorzewski J. Demystifying disability assessments for dermatologists—a call to action. JAMA Dermatol. 2021;157:903-904. doi:10.1001/jamadermatol.2021.1767
Dermatologists’ Perspectives Toward Disability Assessment: A Nationwide Survey Report
Dermatologists’ Perspectives Toward Disability Assessment: A Nationwide Survey Report
PRACTICE POINTS
- As experts in skin conditions, dermatologists are most qualified to assist with disability assessment for dermatologic concerns.
- There are several barriers to dermatologists participating in the disability assessment process, including lack of time, compensation, and education on the subject.
- Many dermatologists may be interested in learning more about disability assessment, and education could be provided in the form of summary guides, lectures, and prefilled paperwork.
Analysis of Errors in the Management of Cutaneous Disorders
Analysis of Errors in the Management of Cutaneous Disorders
Humans are inherently prone to errors. The extent and consequences of medical errors were documented in the 2000 publication of To Err is Human: Building a Safer Health System.1 Published research on medical errors in dermatology has emphasized the heuristic issues involved in diagnosis,2-6 essentially approaching the “why?” and “how?” of such errors. By contrast, the current study aimed to elucidate the “what?”—what are the dermatologic conditions most prone to diagnostic and/or management errors? One study published in 1987 approached this question by analyzing patterns of errors for dermatologic conditions in patients referred for specialty care by primary care physicians.7 The current study aimed to update and expand on the findings of this 1987 report by comparing more recent data on the errors made by providers and patients regarding skin conditions.
Methods
Data were collected prospectively from March 18, 2021, through July 25, 2023. Prospective data were obtained by recording the nature of errors noted for all patients seen by a board-certified dermatologist (R.J.P.) during routine outpatient practice in Norfolk, Virginia. This practice is limited to medical dermatology and accepts patients of any age from any referral source, with or without medical insurance. Retrospective data were obtained by review of electronic medical records for all patients seen by the same board-certified dermatologist from June 5, 2020, through March 12, 2021, who previously had been seen by an outside provider or were self-referred. In this study, the term diagnosis is used to describe providers’ explicit or imputed conclusions as to the nature of a dermatosis, and the term interpretation is used to describe patients' conclusions about their own condition. For this study, the patients’ self-made interpretations of their dermatoses were deemed to be correct when they agreed with those made by the dermatologist using standard clinicopathologic criteria supplemented by rapid bedside diagnostic techniques, as detailed in the 1987 study.7
Cases in which diagnostic or therapeutic errors were noted were entered into a spreadsheet that excluded patients’ names or other identifiers. For each noted case of diagnostic or therapeutic error, the following data were entered: patient’s age and sex; the name of the incorrect diagnosis, interpretation, or treatment; and the name of the correct (missed) diagnosis, along with the source of the error (provider or patient). Provider diagnoses were determined from medical records or patient statements or were imputed from the generally accepted indications for prescribed treatments. A provider was deemed to be any practitioner with prescriptive authority. Patients’ interpretations of their conditions were determined by patient statements or were imputed based on the indications for treatments being used. A treatment error was recorded when a diagnosis or interpretation was deemed to be correct, but treatment was deemed to be inappropriate. The same dermatologist (R.J.P) made all determinations as to the nature of the errors and their source.
Diagnostic errors were determined in several situations: (1) if the interpretation made by the patient of their dermatosis differed from the correct diagnosis in the absence of any additional diagnostic documentation, the correct diagnosis was scored as a missed diagnosis and the incorrect interpretation was scored as such; (2) if the provider’s diagnosis in the patient’s medical record differed from the correct diagnosis, both the correct (missed) and incorrect diagnoses were recorded; and (3) if the indication(s) of the medication(s) prescribed by the provider or used by the patient for their condition differed from the correct diagnosis, an imputed diagnosis based on this indication was scored as the incorrect diagnosis and the correct (missed) diagnosis was recorded; for example, an error would be entered into the spreadsheet for a patient using terbinafine cream for what was actually psoriasis. For a medication with multiple active agents, an error would be entered into the spreadsheet only if none of its indications matched the correct diagnosis; for example, if the patient had been prescribed a betamethasone/clotrimazole product, no error would be scored if the correct diagnosis was a steroid-responsive dermatosis, dermatophytosis, candidiasis, or tinea versicolor. For a single medication with multiple indications, no error would be recorded if the correct diagnosis was any of these indications; for example, in a patient who had been prescribed topical ketoconazole, no error would be scored if the correct diagnosis was dermatophytosis, candidiasis, tinea versicolor, or seborrheic dermatitis. Additionally, no error would be recorded if the correct diagnosis was uncertain at the time of initial patient evaluation or during chart review.
Standard spreadsheet functions and the pandas package8 from the Python programming language9 were used to extract relevant data from the spreadsheet (Tables 1-4).




Results
A total of 446 patient visits (182 males, 264 females) were included in the study, in which a total of 486 errors were found in the combined prospective and retrospective portions of the study. These errors involved 1.4% of all patient visits for the study period—specifically, all in routine practice as well as all patient records retrospectively reviewed. The age of the patients ranged from 4 to 95 years; the mean age was 51.5 years for males and 50.8 years for females.
The study results are outlined in Tables 1 through 4. To minimize the amount of data provided with no appreciable effect on the results, cases in which an incorrect or missed diagnosis/interpretation occurred only once (ie, unique case errors) were excluded from the tables. Tables 1 and 2 indicate the numbers and types of incorrect and missed diagnoses.
In the combined patient and provider cases, there were 434 instances in which provider diagnoses and patient interpretations were incorrect, 320 (73.7%) of which involved infectious disorders. By contrast, of the 413 instances of provider and patient missed diagnoses 289 (70.0%) were inflammatory dermatoses. The pattern was similar for patients’ incorrect interpretations compared to the incorrect diagnoses of the medical providers. Patients incorrectly interpreted their dermatoses as infectious in 79.5% (101/127) of cases. Similarly, providers incorrectly diagnosed their patients’ dermatoses as infectious in 75.4% (211/280) of cases (Table 3). For patients’ missed diagnoses, 70.7% (82/116) involved inflammatory dermatoses. For providers’ missed diagnoses, 63.9% (179/280) involved inflammatory dermatoses (Table 4).
Treatment errors in the context of correct diagnoses were uncommon. Fifteen (3.4%) such cases were noted in the 446 error-containing patient visits. In 4 (26.7%) of the 15 cases, potent topical corticosteroids were used long term on inappropriate cutaneous sites (eg, genital, facial, or intertriginous areas). Another 4 (26.7%) cases involved fungal infections: nystatin used for tinea versicolor in 1 case and for dermatophytosis in another, widespread dermatophytosis treated topically, and use of a nonindicated topical antifungal for onychomycosis. Other examples involved inadequate dosing of systemic corticosteroids for extensive acute contact dermatitis, psoriasis treated with systemic corticosteroids, inadequate dosing of medication for seborrheic dermatitis, and treatment with valacyclovir based solely on serologic testing.
Comment
The results of our study indicate that errors in management of cutaneous disorders are overwhelmingly diagnostic in nature, while treatment errors appear to be unusual when the correct diagnosis is made. Both the current study and the 1987 study indicated a notable tendency of providers to incorrectly diagnose infectious disorders and to miss the diagnosis of inflammatory dermatoses.7 The current study extends this finding to include patients’ interpretive errors.
It is notable that many of the incorrect and missed diagnoses can be confirmed or ruled out by rapid bedside techniques, namely potassium hydroxide (KOH) preparation for dermatophytes, candidiasis, and tinea versicolor; wet preparation for scabies and pediculosis; Tzanck preparation for herpes simplex and herpes zoster; and crush preparation for molluscum contagiosum. Notably, 57.8% (281/486) of cases in which error was noted involved disorders for which the use of one of these bedside diagnostic tests could have correctly established a diagnosis or ruled out an incorrect one; thus in an ideal world in which these tests were performed perfectly in all appropriate cases, more than half of the errors detected in this study could have been avoided. Dermatophytosis was involved in 35.8% (174/486) of the error-containing patient encounters in this study; therefore, if only the KOH preparation is considered, more than one-third of all errors documented in this study could have been avoided. Unfortunately, surveys have suggested that among dermatologists in the United States and some other countries, KOH preparations are used infrequently.10-12
Certain limitations were inherent to this study. The data were derived from a single dermatology practice by one physician in one geographic region over a short period of time. These factors may limit the generalizability of the results. Although the goal was to identify all errors made for the patients seen, some errors likely were missed due to incomplete patient history or inaccurate medication listings. There is no absolute way to determine if the diagnoses or the treatments deemed correct by the dermatologist were, in fact, correct. For cases in which a patient’s interpretation or a provider’s diagnosis was imputed from the indication(s) associated with the medication(s) being used, one cannot exclude the possibility that a medication was used appropriately for a nonlabeled or nonstandard indication. The designation of treatment errors may be subject to different interpretations by different clinicians. Despite these limitations, it is likely that the results of this study can be extrapolated to reasonably similar dermatology practices. The apparently persistent and consistent tendency of clinicians to incorrectly diagnose infectious dermatoses and to miss inflammatory conditions has implications for teaching of medical dermatology in the academic and clinical settings. In particular, given that dermatophytosis is the diagnosis involved in the highest number of errors, special emphasis should be placed on this infection in clinician education.
Acknowledgement—The authors would like to acknowledge the essential contributions to this study by Urvi Jain (Virginia Beach, Virginia), particularly for analysis and interpretation of data and for suggestions to improve the manuscript.
- Institute of Medicine (US) Committee on Quality of Health Care in America. To Err is Human: Building a Safer Health System. Kohn LT, Corrigan JM, Donaldson MS, eds. National Academies Press; 2000.
- Lowenstein EJ, Sidlow R, Ko CJ. Visual perception, cognition, and error in dermatologic diagnosis: diagnosis and error. J Am Acad Dermatol. 2019;81:1237-1245.
- Ko CJ, Braverman I, Sidlow R, et al. Visual perception, cognition, and error in dermatologic diagnosis: key cognitive principles. J Am Acad Dermatol. 2019;81:1227-1234.
- Lowenstein EJ. Dermatology and its unique diagnostic heuristics. J Am Acad Dermatol. 2018;78:1239-1240.
- Elston DM. Cognitive bias and medical errors. J Am Acad Dermatol. 2019;81:1249.
- Costa Filho GB, Moura AS, Brandão PR, et al. Effects of deliberate reflection on diagnostic accuracy, confidence and diagnostic calibration in dermatology. Perspect Med Educ. 2019;8:230-236.
- Pariser RJ, Pariser DM. Primary physicians’ errors in handling cutaneous disorders. J Am Acad Dermatol. 1987;17:239-245.
- van Rossum G, Drake FL Jr. Python Reference Manual. Centrum voor Wiskunde en Informatica; 1995.
- The pandas development team. pandas-dev/pandas: Pandas. Zenodo. February 2020. doi:10.5281/zenodo.3509134
- Murphy EC, Friedman AJ. Use of in-office preparations by dermatologists for the diagnosis of cutaneous fungal infections. J Drugs Dermatol. 2019;18:798-802.
- Dhafiri MA, Alhamed AS, Aljughayman MA. Use of potassium hydroxide in dermatology daily practice: a local study from Saudi Arabia. Cureus. 2022;14:E30612. doi:10.7759/cureus .30612.eCollection
- Chandler JD, Yamamoto R, Hay RJ. Use of direct microscopy to diagnose superficial mycoses: a survey of UK dermatology practice. Br J Dermatol. 2023;189:480-481.
Humans are inherently prone to errors. The extent and consequences of medical errors were documented in the 2000 publication of To Err is Human: Building a Safer Health System.1 Published research on medical errors in dermatology has emphasized the heuristic issues involved in diagnosis,2-6 essentially approaching the “why?” and “how?” of such errors. By contrast, the current study aimed to elucidate the “what?”—what are the dermatologic conditions most prone to diagnostic and/or management errors? One study published in 1987 approached this question by analyzing patterns of errors for dermatologic conditions in patients referred for specialty care by primary care physicians.7 The current study aimed to update and expand on the findings of this 1987 report by comparing more recent data on the errors made by providers and patients regarding skin conditions.
Methods
Data were collected prospectively from March 18, 2021, through July 25, 2023. Prospective data were obtained by recording the nature of errors noted for all patients seen by a board-certified dermatologist (R.J.P.) during routine outpatient practice in Norfolk, Virginia. This practice is limited to medical dermatology and accepts patients of any age from any referral source, with or without medical insurance. Retrospective data were obtained by review of electronic medical records for all patients seen by the same board-certified dermatologist from June 5, 2020, through March 12, 2021, who previously had been seen by an outside provider or were self-referred. In this study, the term diagnosis is used to describe providers’ explicit or imputed conclusions as to the nature of a dermatosis, and the term interpretation is used to describe patients' conclusions about their own condition. For this study, the patients’ self-made interpretations of their dermatoses were deemed to be correct when they agreed with those made by the dermatologist using standard clinicopathologic criteria supplemented by rapid bedside diagnostic techniques, as detailed in the 1987 study.7
Cases in which diagnostic or therapeutic errors were noted were entered into a spreadsheet that excluded patients’ names or other identifiers. For each noted case of diagnostic or therapeutic error, the following data were entered: patient’s age and sex; the name of the incorrect diagnosis, interpretation, or treatment; and the name of the correct (missed) diagnosis, along with the source of the error (provider or patient). Provider diagnoses were determined from medical records or patient statements or were imputed from the generally accepted indications for prescribed treatments. A provider was deemed to be any practitioner with prescriptive authority. Patients’ interpretations of their conditions were determined by patient statements or were imputed based on the indications for treatments being used. A treatment error was recorded when a diagnosis or interpretation was deemed to be correct, but treatment was deemed to be inappropriate. The same dermatologist (R.J.P) made all determinations as to the nature of the errors and their source.
Diagnostic errors were determined in several situations: (1) if the interpretation made by the patient of their dermatosis differed from the correct diagnosis in the absence of any additional diagnostic documentation, the correct diagnosis was scored as a missed diagnosis and the incorrect interpretation was scored as such; (2) if the provider’s diagnosis in the patient’s medical record differed from the correct diagnosis, both the correct (missed) and incorrect diagnoses were recorded; and (3) if the indication(s) of the medication(s) prescribed by the provider or used by the patient for their condition differed from the correct diagnosis, an imputed diagnosis based on this indication was scored as the incorrect diagnosis and the correct (missed) diagnosis was recorded; for example, an error would be entered into the spreadsheet for a patient using terbinafine cream for what was actually psoriasis. For a medication with multiple active agents, an error would be entered into the spreadsheet only if none of its indications matched the correct diagnosis; for example, if the patient had been prescribed a betamethasone/clotrimazole product, no error would be scored if the correct diagnosis was a steroid-responsive dermatosis, dermatophytosis, candidiasis, or tinea versicolor. For a single medication with multiple indications, no error would be recorded if the correct diagnosis was any of these indications; for example, in a patient who had been prescribed topical ketoconazole, no error would be scored if the correct diagnosis was dermatophytosis, candidiasis, tinea versicolor, or seborrheic dermatitis. Additionally, no error would be recorded if the correct diagnosis was uncertain at the time of initial patient evaluation or during chart review.
Standard spreadsheet functions and the pandas package8 from the Python programming language9 were used to extract relevant data from the spreadsheet (Tables 1-4).




Results
A total of 446 patient visits (182 males, 264 females) were included in the study, in which a total of 486 errors were found in the combined prospective and retrospective portions of the study. These errors involved 1.4% of all patient visits for the study period—specifically, all in routine practice as well as all patient records retrospectively reviewed. The age of the patients ranged from 4 to 95 years; the mean age was 51.5 years for males and 50.8 years for females.
The study results are outlined in Tables 1 through 4. To minimize the amount of data provided with no appreciable effect on the results, cases in which an incorrect or missed diagnosis/interpretation occurred only once (ie, unique case errors) were excluded from the tables. Tables 1 and 2 indicate the numbers and types of incorrect and missed diagnoses.
In the combined patient and provider cases, there were 434 instances in which provider diagnoses and patient interpretations were incorrect, 320 (73.7%) of which involved infectious disorders. By contrast, of the 413 instances of provider and patient missed diagnoses 289 (70.0%) were inflammatory dermatoses. The pattern was similar for patients’ incorrect interpretations compared to the incorrect diagnoses of the medical providers. Patients incorrectly interpreted their dermatoses as infectious in 79.5% (101/127) of cases. Similarly, providers incorrectly diagnosed their patients’ dermatoses as infectious in 75.4% (211/280) of cases (Table 3). For patients’ missed diagnoses, 70.7% (82/116) involved inflammatory dermatoses. For providers’ missed diagnoses, 63.9% (179/280) involved inflammatory dermatoses (Table 4).
Treatment errors in the context of correct diagnoses were uncommon. Fifteen (3.4%) such cases were noted in the 446 error-containing patient visits. In 4 (26.7%) of the 15 cases, potent topical corticosteroids were used long term on inappropriate cutaneous sites (eg, genital, facial, or intertriginous areas). Another 4 (26.7%) cases involved fungal infections: nystatin used for tinea versicolor in 1 case and for dermatophytosis in another, widespread dermatophytosis treated topically, and use of a nonindicated topical antifungal for onychomycosis. Other examples involved inadequate dosing of systemic corticosteroids for extensive acute contact dermatitis, psoriasis treated with systemic corticosteroids, inadequate dosing of medication for seborrheic dermatitis, and treatment with valacyclovir based solely on serologic testing.
Comment
The results of our study indicate that errors in management of cutaneous disorders are overwhelmingly diagnostic in nature, while treatment errors appear to be unusual when the correct diagnosis is made. Both the current study and the 1987 study indicated a notable tendency of providers to incorrectly diagnose infectious disorders and to miss the diagnosis of inflammatory dermatoses.7 The current study extends this finding to include patients’ interpretive errors.
It is notable that many of the incorrect and missed diagnoses can be confirmed or ruled out by rapid bedside techniques, namely potassium hydroxide (KOH) preparation for dermatophytes, candidiasis, and tinea versicolor; wet preparation for scabies and pediculosis; Tzanck preparation for herpes simplex and herpes zoster; and crush preparation for molluscum contagiosum. Notably, 57.8% (281/486) of cases in which error was noted involved disorders for which the use of one of these bedside diagnostic tests could have correctly established a diagnosis or ruled out an incorrect one; thus in an ideal world in which these tests were performed perfectly in all appropriate cases, more than half of the errors detected in this study could have been avoided. Dermatophytosis was involved in 35.8% (174/486) of the error-containing patient encounters in this study; therefore, if only the KOH preparation is considered, more than one-third of all errors documented in this study could have been avoided. Unfortunately, surveys have suggested that among dermatologists in the United States and some other countries, KOH preparations are used infrequently.10-12
Certain limitations were inherent to this study. The data were derived from a single dermatology practice by one physician in one geographic region over a short period of time. These factors may limit the generalizability of the results. Although the goal was to identify all errors made for the patients seen, some errors likely were missed due to incomplete patient history or inaccurate medication listings. There is no absolute way to determine if the diagnoses or the treatments deemed correct by the dermatologist were, in fact, correct. For cases in which a patient’s interpretation or a provider’s diagnosis was imputed from the indication(s) associated with the medication(s) being used, one cannot exclude the possibility that a medication was used appropriately for a nonlabeled or nonstandard indication. The designation of treatment errors may be subject to different interpretations by different clinicians. Despite these limitations, it is likely that the results of this study can be extrapolated to reasonably similar dermatology practices. The apparently persistent and consistent tendency of clinicians to incorrectly diagnose infectious dermatoses and to miss inflammatory conditions has implications for teaching of medical dermatology in the academic and clinical settings. In particular, given that dermatophytosis is the diagnosis involved in the highest number of errors, special emphasis should be placed on this infection in clinician education.
Acknowledgement—The authors would like to acknowledge the essential contributions to this study by Urvi Jain (Virginia Beach, Virginia), particularly for analysis and interpretation of data and for suggestions to improve the manuscript.
Humans are inherently prone to errors. The extent and consequences of medical errors were documented in the 2000 publication of To Err is Human: Building a Safer Health System.1 Published research on medical errors in dermatology has emphasized the heuristic issues involved in diagnosis,2-6 essentially approaching the “why?” and “how?” of such errors. By contrast, the current study aimed to elucidate the “what?”—what are the dermatologic conditions most prone to diagnostic and/or management errors? One study published in 1987 approached this question by analyzing patterns of errors for dermatologic conditions in patients referred for specialty care by primary care physicians.7 The current study aimed to update and expand on the findings of this 1987 report by comparing more recent data on the errors made by providers and patients regarding skin conditions.
Methods
Data were collected prospectively from March 18, 2021, through July 25, 2023. Prospective data were obtained by recording the nature of errors noted for all patients seen by a board-certified dermatologist (R.J.P.) during routine outpatient practice in Norfolk, Virginia. This practice is limited to medical dermatology and accepts patients of any age from any referral source, with or without medical insurance. Retrospective data were obtained by review of electronic medical records for all patients seen by the same board-certified dermatologist from June 5, 2020, through March 12, 2021, who previously had been seen by an outside provider or were self-referred. In this study, the term diagnosis is used to describe providers’ explicit or imputed conclusions as to the nature of a dermatosis, and the term interpretation is used to describe patients' conclusions about their own condition. For this study, the patients’ self-made interpretations of their dermatoses were deemed to be correct when they agreed with those made by the dermatologist using standard clinicopathologic criteria supplemented by rapid bedside diagnostic techniques, as detailed in the 1987 study.7
Cases in which diagnostic or therapeutic errors were noted were entered into a spreadsheet that excluded patients’ names or other identifiers. For each noted case of diagnostic or therapeutic error, the following data were entered: patient’s age and sex; the name of the incorrect diagnosis, interpretation, or treatment; and the name of the correct (missed) diagnosis, along with the source of the error (provider or patient). Provider diagnoses were determined from medical records or patient statements or were imputed from the generally accepted indications for prescribed treatments. A provider was deemed to be any practitioner with prescriptive authority. Patients’ interpretations of their conditions were determined by patient statements or were imputed based on the indications for treatments being used. A treatment error was recorded when a diagnosis or interpretation was deemed to be correct, but treatment was deemed to be inappropriate. The same dermatologist (R.J.P) made all determinations as to the nature of the errors and their source.
Diagnostic errors were determined in several situations: (1) if the interpretation made by the patient of their dermatosis differed from the correct diagnosis in the absence of any additional diagnostic documentation, the correct diagnosis was scored as a missed diagnosis and the incorrect interpretation was scored as such; (2) if the provider’s diagnosis in the patient’s medical record differed from the correct diagnosis, both the correct (missed) and incorrect diagnoses were recorded; and (3) if the indication(s) of the medication(s) prescribed by the provider or used by the patient for their condition differed from the correct diagnosis, an imputed diagnosis based on this indication was scored as the incorrect diagnosis and the correct (missed) diagnosis was recorded; for example, an error would be entered into the spreadsheet for a patient using terbinafine cream for what was actually psoriasis. For a medication with multiple active agents, an error would be entered into the spreadsheet only if none of its indications matched the correct diagnosis; for example, if the patient had been prescribed a betamethasone/clotrimazole product, no error would be scored if the correct diagnosis was a steroid-responsive dermatosis, dermatophytosis, candidiasis, or tinea versicolor. For a single medication with multiple indications, no error would be recorded if the correct diagnosis was any of these indications; for example, in a patient who had been prescribed topical ketoconazole, no error would be scored if the correct diagnosis was dermatophytosis, candidiasis, tinea versicolor, or seborrheic dermatitis. Additionally, no error would be recorded if the correct diagnosis was uncertain at the time of initial patient evaluation or during chart review.
Standard spreadsheet functions and the pandas package8 from the Python programming language9 were used to extract relevant data from the spreadsheet (Tables 1-4).




Results
A total of 446 patient visits (182 males, 264 females) were included in the study, in which a total of 486 errors were found in the combined prospective and retrospective portions of the study. These errors involved 1.4% of all patient visits for the study period—specifically, all in routine practice as well as all patient records retrospectively reviewed. The age of the patients ranged from 4 to 95 years; the mean age was 51.5 years for males and 50.8 years for females.
The study results are outlined in Tables 1 through 4. To minimize the amount of data provided with no appreciable effect on the results, cases in which an incorrect or missed diagnosis/interpretation occurred only once (ie, unique case errors) were excluded from the tables. Tables 1 and 2 indicate the numbers and types of incorrect and missed diagnoses.
In the combined patient and provider cases, there were 434 instances in which provider diagnoses and patient interpretations were incorrect, 320 (73.7%) of which involved infectious disorders. By contrast, of the 413 instances of provider and patient missed diagnoses 289 (70.0%) were inflammatory dermatoses. The pattern was similar for patients’ incorrect interpretations compared to the incorrect diagnoses of the medical providers. Patients incorrectly interpreted their dermatoses as infectious in 79.5% (101/127) of cases. Similarly, providers incorrectly diagnosed their patients’ dermatoses as infectious in 75.4% (211/280) of cases (Table 3). For patients’ missed diagnoses, 70.7% (82/116) involved inflammatory dermatoses. For providers’ missed diagnoses, 63.9% (179/280) involved inflammatory dermatoses (Table 4).
Treatment errors in the context of correct diagnoses were uncommon. Fifteen (3.4%) such cases were noted in the 446 error-containing patient visits. In 4 (26.7%) of the 15 cases, potent topical corticosteroids were used long term on inappropriate cutaneous sites (eg, genital, facial, or intertriginous areas). Another 4 (26.7%) cases involved fungal infections: nystatin used for tinea versicolor in 1 case and for dermatophytosis in another, widespread dermatophytosis treated topically, and use of a nonindicated topical antifungal for onychomycosis. Other examples involved inadequate dosing of systemic corticosteroids for extensive acute contact dermatitis, psoriasis treated with systemic corticosteroids, inadequate dosing of medication for seborrheic dermatitis, and treatment with valacyclovir based solely on serologic testing.
Comment
The results of our study indicate that errors in management of cutaneous disorders are overwhelmingly diagnostic in nature, while treatment errors appear to be unusual when the correct diagnosis is made. Both the current study and the 1987 study indicated a notable tendency of providers to incorrectly diagnose infectious disorders and to miss the diagnosis of inflammatory dermatoses.7 The current study extends this finding to include patients’ interpretive errors.
It is notable that many of the incorrect and missed diagnoses can be confirmed or ruled out by rapid bedside techniques, namely potassium hydroxide (KOH) preparation for dermatophytes, candidiasis, and tinea versicolor; wet preparation for scabies and pediculosis; Tzanck preparation for herpes simplex and herpes zoster; and crush preparation for molluscum contagiosum. Notably, 57.8% (281/486) of cases in which error was noted involved disorders for which the use of one of these bedside diagnostic tests could have correctly established a diagnosis or ruled out an incorrect one; thus in an ideal world in which these tests were performed perfectly in all appropriate cases, more than half of the errors detected in this study could have been avoided. Dermatophytosis was involved in 35.8% (174/486) of the error-containing patient encounters in this study; therefore, if only the KOH preparation is considered, more than one-third of all errors documented in this study could have been avoided. Unfortunately, surveys have suggested that among dermatologists in the United States and some other countries, KOH preparations are used infrequently.10-12
Certain limitations were inherent to this study. The data were derived from a single dermatology practice by one physician in one geographic region over a short period of time. These factors may limit the generalizability of the results. Although the goal was to identify all errors made for the patients seen, some errors likely were missed due to incomplete patient history or inaccurate medication listings. There is no absolute way to determine if the diagnoses or the treatments deemed correct by the dermatologist were, in fact, correct. For cases in which a patient’s interpretation or a provider’s diagnosis was imputed from the indication(s) associated with the medication(s) being used, one cannot exclude the possibility that a medication was used appropriately for a nonlabeled or nonstandard indication. The designation of treatment errors may be subject to different interpretations by different clinicians. Despite these limitations, it is likely that the results of this study can be extrapolated to reasonably similar dermatology practices. The apparently persistent and consistent tendency of clinicians to incorrectly diagnose infectious dermatoses and to miss inflammatory conditions has implications for teaching of medical dermatology in the academic and clinical settings. In particular, given that dermatophytosis is the diagnosis involved in the highest number of errors, special emphasis should be placed on this infection in clinician education.
Acknowledgement—The authors would like to acknowledge the essential contributions to this study by Urvi Jain (Virginia Beach, Virginia), particularly for analysis and interpretation of data and for suggestions to improve the manuscript.
- Institute of Medicine (US) Committee on Quality of Health Care in America. To Err is Human: Building a Safer Health System. Kohn LT, Corrigan JM, Donaldson MS, eds. National Academies Press; 2000.
- Lowenstein EJ, Sidlow R, Ko CJ. Visual perception, cognition, and error in dermatologic diagnosis: diagnosis and error. J Am Acad Dermatol. 2019;81:1237-1245.
- Ko CJ, Braverman I, Sidlow R, et al. Visual perception, cognition, and error in dermatologic diagnosis: key cognitive principles. J Am Acad Dermatol. 2019;81:1227-1234.
- Lowenstein EJ. Dermatology and its unique diagnostic heuristics. J Am Acad Dermatol. 2018;78:1239-1240.
- Elston DM. Cognitive bias and medical errors. J Am Acad Dermatol. 2019;81:1249.
- Costa Filho GB, Moura AS, Brandão PR, et al. Effects of deliberate reflection on diagnostic accuracy, confidence and diagnostic calibration in dermatology. Perspect Med Educ. 2019;8:230-236.
- Pariser RJ, Pariser DM. Primary physicians’ errors in handling cutaneous disorders. J Am Acad Dermatol. 1987;17:239-245.
- van Rossum G, Drake FL Jr. Python Reference Manual. Centrum voor Wiskunde en Informatica; 1995.
- The pandas development team. pandas-dev/pandas: Pandas. Zenodo. February 2020. doi:10.5281/zenodo.3509134
- Murphy EC, Friedman AJ. Use of in-office preparations by dermatologists for the diagnosis of cutaneous fungal infections. J Drugs Dermatol. 2019;18:798-802.
- Dhafiri MA, Alhamed AS, Aljughayman MA. Use of potassium hydroxide in dermatology daily practice: a local study from Saudi Arabia. Cureus. 2022;14:E30612. doi:10.7759/cureus .30612.eCollection
- Chandler JD, Yamamoto R, Hay RJ. Use of direct microscopy to diagnose superficial mycoses: a survey of UK dermatology practice. Br J Dermatol. 2023;189:480-481.
- Institute of Medicine (US) Committee on Quality of Health Care in America. To Err is Human: Building a Safer Health System. Kohn LT, Corrigan JM, Donaldson MS, eds. National Academies Press; 2000.
- Lowenstein EJ, Sidlow R, Ko CJ. Visual perception, cognition, and error in dermatologic diagnosis: diagnosis and error. J Am Acad Dermatol. 2019;81:1237-1245.
- Ko CJ, Braverman I, Sidlow R, et al. Visual perception, cognition, and error in dermatologic diagnosis: key cognitive principles. J Am Acad Dermatol. 2019;81:1227-1234.
- Lowenstein EJ. Dermatology and its unique diagnostic heuristics. J Am Acad Dermatol. 2018;78:1239-1240.
- Elston DM. Cognitive bias and medical errors. J Am Acad Dermatol. 2019;81:1249.
- Costa Filho GB, Moura AS, Brandão PR, et al. Effects of deliberate reflection on diagnostic accuracy, confidence and diagnostic calibration in dermatology. Perspect Med Educ. 2019;8:230-236.
- Pariser RJ, Pariser DM. Primary physicians’ errors in handling cutaneous disorders. J Am Acad Dermatol. 1987;17:239-245.
- van Rossum G, Drake FL Jr. Python Reference Manual. Centrum voor Wiskunde en Informatica; 1995.
- The pandas development team. pandas-dev/pandas: Pandas. Zenodo. February 2020. doi:10.5281/zenodo.3509134
- Murphy EC, Friedman AJ. Use of in-office preparations by dermatologists for the diagnosis of cutaneous fungal infections. J Drugs Dermatol. 2019;18:798-802.
- Dhafiri MA, Alhamed AS, Aljughayman MA. Use of potassium hydroxide in dermatology daily practice: a local study from Saudi Arabia. Cureus. 2022;14:E30612. doi:10.7759/cureus .30612.eCollection
- Chandler JD, Yamamoto R, Hay RJ. Use of direct microscopy to diagnose superficial mycoses: a survey of UK dermatology practice. Br J Dermatol. 2023;189:480-481.
Analysis of Errors in the Management of Cutaneous Disorders
Analysis of Errors in the Management of Cutaneous Disorders
PRACTICE POINTS
- Errors in the management of cutaneous disorders predominantly are due to misdiagnosis rather than treatment oversights.
- There is a tendency among medical providers to incorrectly diagnose dermatoses as infectious disorders and to miss the diagnosis of inflammatory dermatoses.
- A similar pattern of errors occurs for patients’ interpretations of their own skin conditions.
- Use of available rapid bedside diagnostic techniques can reduce the likelihood of errors made by medical providers.
White Atrophic Plaques on the Thighs
White Atrophic Plaques on the Thighs
THE DIAGNOSIS: Lichen Sclerosus
Given the clinical appearance of white atrophic plaques with characteristic wrinkling of the skin, a diagnosis of lichen sclerosus was strongly suspected. At the initial office visit, the patient was prescribed clobetasol 0.05% ointment twice daily for 6 weeks. Histopathology revealed hyperkeratosis, follicular plugging, papillary dermal pallor, and adjacent lymphocytic inflammation, confirming the clinical diagnosis of lichen sclerosus (Figure). The patient then was lost to follow-up.
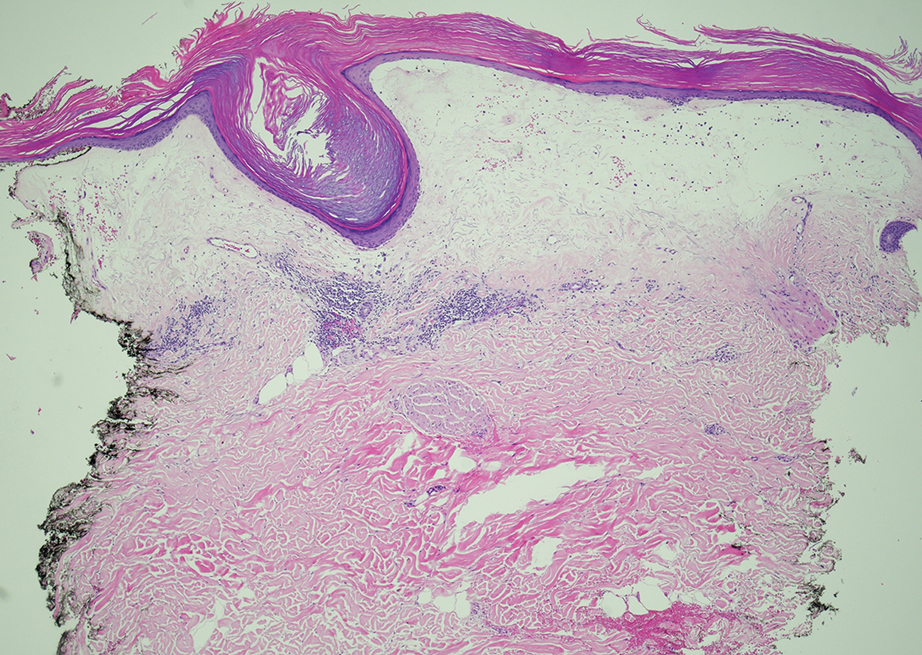
Lichen sclerosus is a chronic benign dermatologic condition of unknown etiology that is characterized by epidermal atrophy and inflammation and is common in postmenopausal women. It features pale, ivory-colored lesions with partially atrophic skin and a wrinkled cigarette paper appearance.1 The differential for lichen sclerosus is broad, and definitive diagnosis is made via biopsy to rule out potential malignancy and other inflammatory skin diseases.1 Lichen sclerosus is an immune-mediated disorder driven by type 1 T helper cells and regulated by miR-155. There has been an association with extracellular matrix protein 1, a glycoprotein that is found in the dermal-epidermal basement membrane zone, which provides structural integrity to the skin. Autoantibodies against extracellular matrix protein 1 and other antigens in the basement membrane generally are found in anogenital lichen sclerosus; however, their precise roles in the pathogenesis of lichen sclerosus remains unclear.1
The differential diagnoses for lichen sclerosus include psoriasis, tinea corporis, lichen simplex chronicus, and atopic dermatitis. Psoriasis typically manifests as pink plaques with silver scales on the elbows, knees, and scalp in adult patients.2 Our patient’s white plaques may have suggested psoriasis, but the partially atrophic skin with a wrinkled cigarette paper appearance was not compatible with that diagnosis.
Tinea corporis, a superficial fungal infection of the skin, manifests as circular or ovoid lesions with raised erythematous scaly borders, often with central clearing resembling a ring, that can occur anywhere on the body other than the feet, groin, face, scalp, or beard area.3 The fact that our patient previously had tried topical antifungal medications with no relief and that the skin lesions were atrophic rather than ring shaped made the diagnosis of tinea corporis unlikely.
Lichen simplex chronicus is a chronic condition caused by friction or scratching that is characterized by dry, patchy, scaly, and thickened areas of the skin. Typically affecting the head, arms, neck, scalp, and genital region, lichen simplex chronicus manifests with violaceous or hyperpigmented lesions.4 The nonpruritic atrophic plaques on the inner thighs and the presence of white patches on the vaginal area were not indicative of lichen simplex chronicus in our patient.
Atopic dermatitis manifests as pruritic erythematous scaly papules and plaques with secondary excoriation and possible lichenification. In adults, atopic dermatitis commonly appears on flexural surfaces.2 Atopic dermatitis does not manifest with atrophy and skin wrinkling as seen in our patient.
In the management of lichen sclerosus, the standard treatment is potent topical corticosteroids. Alternatively, topical calcineurin inhibitors can be employed; however, due to the unknown nature of the condition’s underlying cause, targeted treatment is challenging. Our case underscores how lichen sclerosus can be misdiagnosed, highlighting the need for more frequent reporting in the literature to enhance early recognition and reduce delays in patient treatment.
- De Luca DA, Papara C, Vorobyev A, et al. Lichen sclerosus: the 2023 update. Front Med (Lausanne). 2023;10:1106318. doi:10.3389 /fmed.2023.1106318
- Chovatiya R, Silverberg JI. Pathophysiology of atopic dermatitis and psoriasis: implications for management in children. Children (Basel). 2019;6:108. doi:10.3390/children6100108
- Trayes KP, Savage K, Studdiford JS. Annular lesions: diagnosis and treatment. Am Fam Physician. 2018;98:283-291.
- Ju T, Vander Does A, Mohsin N, et al. Lichen simplex chronicus itch: an update. Acta Derm Venereol. 2022;102:adv00796. doi:10.2340 /actadv.v102.4367
THE DIAGNOSIS: Lichen Sclerosus
Given the clinical appearance of white atrophic plaques with characteristic wrinkling of the skin, a diagnosis of lichen sclerosus was strongly suspected. At the initial office visit, the patient was prescribed clobetasol 0.05% ointment twice daily for 6 weeks. Histopathology revealed hyperkeratosis, follicular plugging, papillary dermal pallor, and adjacent lymphocytic inflammation, confirming the clinical diagnosis of lichen sclerosus (Figure). The patient then was lost to follow-up.

Lichen sclerosus is a chronic benign dermatologic condition of unknown etiology that is characterized by epidermal atrophy and inflammation and is common in postmenopausal women. It features pale, ivory-colored lesions with partially atrophic skin and a wrinkled cigarette paper appearance.1 The differential for lichen sclerosus is broad, and definitive diagnosis is made via biopsy to rule out potential malignancy and other inflammatory skin diseases.1 Lichen sclerosus is an immune-mediated disorder driven by type 1 T helper cells and regulated by miR-155. There has been an association with extracellular matrix protein 1, a glycoprotein that is found in the dermal-epidermal basement membrane zone, which provides structural integrity to the skin. Autoantibodies against extracellular matrix protein 1 and other antigens in the basement membrane generally are found in anogenital lichen sclerosus; however, their precise roles in the pathogenesis of lichen sclerosus remains unclear.1
The differential diagnoses for lichen sclerosus include psoriasis, tinea corporis, lichen simplex chronicus, and atopic dermatitis. Psoriasis typically manifests as pink plaques with silver scales on the elbows, knees, and scalp in adult patients.2 Our patient’s white plaques may have suggested psoriasis, but the partially atrophic skin with a wrinkled cigarette paper appearance was not compatible with that diagnosis.
Tinea corporis, a superficial fungal infection of the skin, manifests as circular or ovoid lesions with raised erythematous scaly borders, often with central clearing resembling a ring, that can occur anywhere on the body other than the feet, groin, face, scalp, or beard area.3 The fact that our patient previously had tried topical antifungal medications with no relief and that the skin lesions were atrophic rather than ring shaped made the diagnosis of tinea corporis unlikely.
Lichen simplex chronicus is a chronic condition caused by friction or scratching that is characterized by dry, patchy, scaly, and thickened areas of the skin. Typically affecting the head, arms, neck, scalp, and genital region, lichen simplex chronicus manifests with violaceous or hyperpigmented lesions.4 The nonpruritic atrophic plaques on the inner thighs and the presence of white patches on the vaginal area were not indicative of lichen simplex chronicus in our patient.
Atopic dermatitis manifests as pruritic erythematous scaly papules and plaques with secondary excoriation and possible lichenification. In adults, atopic dermatitis commonly appears on flexural surfaces.2 Atopic dermatitis does not manifest with atrophy and skin wrinkling as seen in our patient.
In the management of lichen sclerosus, the standard treatment is potent topical corticosteroids. Alternatively, topical calcineurin inhibitors can be employed; however, due to the unknown nature of the condition’s underlying cause, targeted treatment is challenging. Our case underscores how lichen sclerosus can be misdiagnosed, highlighting the need for more frequent reporting in the literature to enhance early recognition and reduce delays in patient treatment.
THE DIAGNOSIS: Lichen Sclerosus
Given the clinical appearance of white atrophic plaques with characteristic wrinkling of the skin, a diagnosis of lichen sclerosus was strongly suspected. At the initial office visit, the patient was prescribed clobetasol 0.05% ointment twice daily for 6 weeks. Histopathology revealed hyperkeratosis, follicular plugging, papillary dermal pallor, and adjacent lymphocytic inflammation, confirming the clinical diagnosis of lichen sclerosus (Figure). The patient then was lost to follow-up.

Lichen sclerosus is a chronic benign dermatologic condition of unknown etiology that is characterized by epidermal atrophy and inflammation and is common in postmenopausal women. It features pale, ivory-colored lesions with partially atrophic skin and a wrinkled cigarette paper appearance.1 The differential for lichen sclerosus is broad, and definitive diagnosis is made via biopsy to rule out potential malignancy and other inflammatory skin diseases.1 Lichen sclerosus is an immune-mediated disorder driven by type 1 T helper cells and regulated by miR-155. There has been an association with extracellular matrix protein 1, a glycoprotein that is found in the dermal-epidermal basement membrane zone, which provides structural integrity to the skin. Autoantibodies against extracellular matrix protein 1 and other antigens in the basement membrane generally are found in anogenital lichen sclerosus; however, their precise roles in the pathogenesis of lichen sclerosus remains unclear.1
The differential diagnoses for lichen sclerosus include psoriasis, tinea corporis, lichen simplex chronicus, and atopic dermatitis. Psoriasis typically manifests as pink plaques with silver scales on the elbows, knees, and scalp in adult patients.2 Our patient’s white plaques may have suggested psoriasis, but the partially atrophic skin with a wrinkled cigarette paper appearance was not compatible with that diagnosis.
Tinea corporis, a superficial fungal infection of the skin, manifests as circular or ovoid lesions with raised erythematous scaly borders, often with central clearing resembling a ring, that can occur anywhere on the body other than the feet, groin, face, scalp, or beard area.3 The fact that our patient previously had tried topical antifungal medications with no relief and that the skin lesions were atrophic rather than ring shaped made the diagnosis of tinea corporis unlikely.
Lichen simplex chronicus is a chronic condition caused by friction or scratching that is characterized by dry, patchy, scaly, and thickened areas of the skin. Typically affecting the head, arms, neck, scalp, and genital region, lichen simplex chronicus manifests with violaceous or hyperpigmented lesions.4 The nonpruritic atrophic plaques on the inner thighs and the presence of white patches on the vaginal area were not indicative of lichen simplex chronicus in our patient.
Atopic dermatitis manifests as pruritic erythematous scaly papules and plaques with secondary excoriation and possible lichenification. In adults, atopic dermatitis commonly appears on flexural surfaces.2 Atopic dermatitis does not manifest with atrophy and skin wrinkling as seen in our patient.
In the management of lichen sclerosus, the standard treatment is potent topical corticosteroids. Alternatively, topical calcineurin inhibitors can be employed; however, due to the unknown nature of the condition’s underlying cause, targeted treatment is challenging. Our case underscores how lichen sclerosus can be misdiagnosed, highlighting the need for more frequent reporting in the literature to enhance early recognition and reduce delays in patient treatment.
- De Luca DA, Papara C, Vorobyev A, et al. Lichen sclerosus: the 2023 update. Front Med (Lausanne). 2023;10:1106318. doi:10.3389 /fmed.2023.1106318
- Chovatiya R, Silverberg JI. Pathophysiology of atopic dermatitis and psoriasis: implications for management in children. Children (Basel). 2019;6:108. doi:10.3390/children6100108
- Trayes KP, Savage K, Studdiford JS. Annular lesions: diagnosis and treatment. Am Fam Physician. 2018;98:283-291.
- Ju T, Vander Does A, Mohsin N, et al. Lichen simplex chronicus itch: an update. Acta Derm Venereol. 2022;102:adv00796. doi:10.2340 /actadv.v102.4367
- De Luca DA, Papara C, Vorobyev A, et al. Lichen sclerosus: the 2023 update. Front Med (Lausanne). 2023;10:1106318. doi:10.3389 /fmed.2023.1106318
- Chovatiya R, Silverberg JI. Pathophysiology of atopic dermatitis and psoriasis: implications for management in children. Children (Basel). 2019;6:108. doi:10.3390/children6100108
- Trayes KP, Savage K, Studdiford JS. Annular lesions: diagnosis and treatment. Am Fam Physician. 2018;98:283-291.
- Ju T, Vander Does A, Mohsin N, et al. Lichen simplex chronicus itch: an update. Acta Derm Venereol. 2022;102:adv00796. doi:10.2340 /actadv.v102.4367
White Atrophic Plaques on the Thighs
White Atrophic Plaques on the Thighs
A 71-year-old woman presented to the dermatology clinic for evaluation of intense pruritus of the vaginal region and a nonpruritic rash on the inner thighs of 7 months’ duration. Physical examination revealed white atrophic plaques with scaling and a wrinkled appearance on the inner thighs. White atrophic patches also were noted on the vulva. The patient reported that she had tried over-the-counter antifungals with no improvement. A punch biopsy was performed.
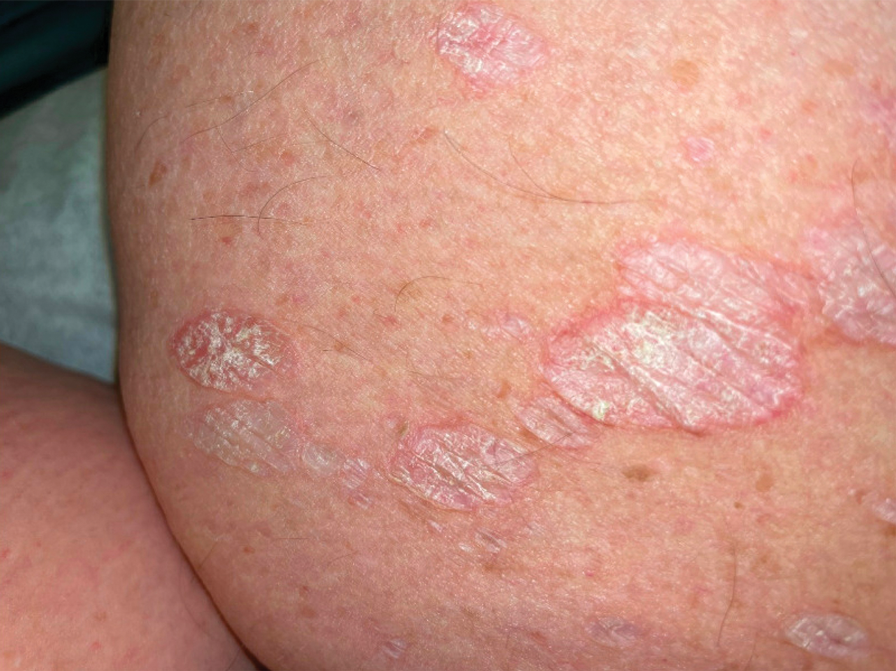
Gastroenterology Knows No Country
The United States boasts one of the premier health care systems for medical education in the world. Indeed, institutions such as Johns Hopkins, Harvard, and the Mayo Clinic have storied reputations and are recognized names the world over. The United States also stands as a country of remarkable discovery in medicine with an abundance of enormously talented and productive medical scientists. This reputation draws physicians from every corner of the world who dream of studying medicine in our country.
Unfortunately, many US medical institutions, particularly the most prestigious medical centers, lean heavily toward preferential acceptance of US medical school graduates as an indicator of the highest-quality trainees. This historical bias is being further compounded by our current government’s pejorative view of immigrants in general. Will this affect the pool of tomorrow’s stars who will change the course of American medicine?
A glance at the list of recent AGA Presidents may yield some insight; over the past 10 years, three of our presidents trained internationally at universities in Malta, Libya, and Germany. This is a small snapshot of the multitude of international graduates in gastroenterology and hepatology who have served as division chiefs, AGA award winners, and journal editors, all now US citizens. This is not to mention the influence of varied insights and talents native to international study and culture that enhance our practice of medicine and biomedical research.
We live in time when “immigrant” has been assigned a negative and almost subhuman connotation, and diversity has become something to be demonized rather than celebrated. Yet, intuitively, should a top US medical graduate be any more intelligent or driven than a top graduate from the United Kingdom, India, China, or Syria? As American medical physicians, we place the utmost value on our traditions and high standards. We boast an unmatched depth of medical talent spread across our GI divisions and practices and take pride in the way we teach medicine, like no other nation. American medicine benefits from their talent and they inspire us to remember and care for diseases in our field that affect the world’s population, not just ours.
Over 100 years ago, Dr. William Mayo stated “American practice is too broad to be national. It had the scientific spirit, and science knows no country.” Dr. Mayo also said, “Democracy is safe only so long as culture is in the ascendancy.” These lessons apply more than ever today.
David Katzka, MD
Associate Editor
The United States boasts one of the premier health care systems for medical education in the world. Indeed, institutions such as Johns Hopkins, Harvard, and the Mayo Clinic have storied reputations and are recognized names the world over. The United States also stands as a country of remarkable discovery in medicine with an abundance of enormously talented and productive medical scientists. This reputation draws physicians from every corner of the world who dream of studying medicine in our country.
Unfortunately, many US medical institutions, particularly the most prestigious medical centers, lean heavily toward preferential acceptance of US medical school graduates as an indicator of the highest-quality trainees. This historical bias is being further compounded by our current government’s pejorative view of immigrants in general. Will this affect the pool of tomorrow’s stars who will change the course of American medicine?
A glance at the list of recent AGA Presidents may yield some insight; over the past 10 years, three of our presidents trained internationally at universities in Malta, Libya, and Germany. This is a small snapshot of the multitude of international graduates in gastroenterology and hepatology who have served as division chiefs, AGA award winners, and journal editors, all now US citizens. This is not to mention the influence of varied insights and talents native to international study and culture that enhance our practice of medicine and biomedical research.
We live in time when “immigrant” has been assigned a negative and almost subhuman connotation, and diversity has become something to be demonized rather than celebrated. Yet, intuitively, should a top US medical graduate be any more intelligent or driven than a top graduate from the United Kingdom, India, China, or Syria? As American medical physicians, we place the utmost value on our traditions and high standards. We boast an unmatched depth of medical talent spread across our GI divisions and practices and take pride in the way we teach medicine, like no other nation. American medicine benefits from their talent and they inspire us to remember and care for diseases in our field that affect the world’s population, not just ours.
Over 100 years ago, Dr. William Mayo stated “American practice is too broad to be national. It had the scientific spirit, and science knows no country.” Dr. Mayo also said, “Democracy is safe only so long as culture is in the ascendancy.” These lessons apply more than ever today.
David Katzka, MD
Associate Editor
The United States boasts one of the premier health care systems for medical education in the world. Indeed, institutions such as Johns Hopkins, Harvard, and the Mayo Clinic have storied reputations and are recognized names the world over. The United States also stands as a country of remarkable discovery in medicine with an abundance of enormously talented and productive medical scientists. This reputation draws physicians from every corner of the world who dream of studying medicine in our country.
Unfortunately, many US medical institutions, particularly the most prestigious medical centers, lean heavily toward preferential acceptance of US medical school graduates as an indicator of the highest-quality trainees. This historical bias is being further compounded by our current government’s pejorative view of immigrants in general. Will this affect the pool of tomorrow’s stars who will change the course of American medicine?
A glance at the list of recent AGA Presidents may yield some insight; over the past 10 years, three of our presidents trained internationally at universities in Malta, Libya, and Germany. This is a small snapshot of the multitude of international graduates in gastroenterology and hepatology who have served as division chiefs, AGA award winners, and journal editors, all now US citizens. This is not to mention the influence of varied insights and talents native to international study and culture that enhance our practice of medicine and biomedical research.
We live in time when “immigrant” has been assigned a negative and almost subhuman connotation, and diversity has become something to be demonized rather than celebrated. Yet, intuitively, should a top US medical graduate be any more intelligent or driven than a top graduate from the United Kingdom, India, China, or Syria? As American medical physicians, we place the utmost value on our traditions and high standards. We boast an unmatched depth of medical talent spread across our GI divisions and practices and take pride in the way we teach medicine, like no other nation. American medicine benefits from their talent and they inspire us to remember and care for diseases in our field that affect the world’s population, not just ours.
Over 100 years ago, Dr. William Mayo stated “American practice is too broad to be national. It had the scientific spirit, and science knows no country.” Dr. Mayo also said, “Democracy is safe only so long as culture is in the ascendancy.” These lessons apply more than ever today.
David Katzka, MD
Associate Editor
How Doctors Use Travel to Heal Themselves
Whatever’s ailing you, a vacation might just be the cure. Yes, getting away can improve your health, according to research published in in 2023. It might help combat symptoms of aging, suggested a 2024 study in Journal of Travel Research. But it could also have even more powerful psychological and physical benefits, transforming your life before you pack a bag and long after you return home.
This news organization spoke with two healthcare professionals who believe in the healing power of travel. They shared which personal “diagnoses” they have successfully treated with faraway places and how this therapy might work for you.
Stacey Funt, MD, NBC-HWC, a radiologist at Northwell Health in Long Island, New York, started the boutique wellness adventure travel company, LH Adventure Travel, in 2023. Funt curates and leads small groups to destinations like Peru, Guatemala, Morocco, and Italy. Each tour incorporates tenets of lifestyle medicine, including healthy eating, movement, stress management, and community building.
Kiya Thompson, RN, a surgical trauma nurse for 20 years, was similarly inspired to share her passion for travel. She is now a certified family travel coach who helps parents plan meaningful trips through her company, LuckyBucky, LLC.
Dx: Self-Esteem Deficiency / Rx: Vivaldi in Venice
In June 2015, Thompson found herself at an all-time low. As a nurse, she felt confident that she was “built for the adrenaline rush and could take on anything.” But outside the trauma center, Thompson felt inadequate, her self-esteem eroded by years of abusive relationships. “The daily hardships of my personal life, combined with the mental fortitude it took to endure the demands of caring for the sickest of the sick, were incredibly weighty,” she recalled.
To escape, Thompson booked her first solo trip: 3 weeks in Italy. But days after she arrived, she felt the need to “escape her escape.” On a bus in Naples, she was pick-pocketed. The man she had been dating before her trip stopped responding to her messages. In her hotel room in Venice, she felt “lost, alone, and helpless.”
One evening, Thompson attended a small orchestral performance of Vivaldi’s “The Four Seasons” in a centuries-old church. The music triggered memories of her Italian grandparents at whose home she’d listened to the same piece.
“A switch flipped, and I changed my whole outlook,” she remembers.
During the concert, she reflected on strangers who had shown her kindness and care. A Canadian man who gave her €50 after her wallet was stolen. A friend-of-a-friend who showed her around Rome. The clerk at her Venice hotel who had offered her a hug.
“In the wake of experiencing the worst of people, I’d experienced so much more of the best of people; strangers who were willing to go above and beyond to help me,” Thompson said.
When Thompson returned home, she brought her new mindset along. “ My ability to problem-solve my way through a solo trip that presented unexpected hardships empowered me,” she explained. “I learned I was much more capable than I’d thought.”
Dx: Wilderness Phobia / Rx: A Safari in Tanzania
On an evening in the mid-1990s, Funt was alone in a tent on a budget camping safari in Tanzania. Animals roared threateningly outside the thin walls. Earlier that day, a vulture had ripped a sandwich out of her hands. Funt was frightened to the core. Worrying that she’d be the next meal for the local wildlife, she started to sob. “This was as raw as I had ever gotten at that point in my life,” she said.
Suddenly, Funt said her brain shifted into problem-solving mode. She made one small decision: To switch to a different Jeep for the next day’s excursion. Having made a seemingly insignificant choice, she felt calmer and no longer like a victim. It brought control. Instead of worrying, she began looking forward to the wildlife she would see.
In the morning, in the new Jeep, she befriended a nurse from Canada. Together, they visited the Maasai Mara tribe and nearby pubs, meeting members of the community.
“It was the most exciting experience of my life,” Funt said. “And it had started with me crying.”
Dx: Parenting-itis / Rx: A Mountain Getaway
As Thompson pointed out, sometimes the destination is secondary to the intension behind a trip. And the quality of the time away matters more than how long you can stay. After becoming parents 4 years ago, Thompson and her husband hadn’t traveled alone together. Like many parents of young children, they were short on time to relax and reconnect as a couple.
So Thompson planned a weekend trip to an isolated cabin in the Massanutten Mountain Range within the George Washington National Forest, about a 2-hour drive from their Washington, DC, area home.
“We put our devices away and focused on being completely present with one another,” said Thompson. The couple took a walk in the woods, where “all we could hear were drops of water from the snowmelt, the crunch of the snow beneath our feet, and the occasional bird looking for food,” she recalled. “There were no cars, no other people. It was quiet, calm, and incredibly peaceful.”
Whether sitting by the fire, soaking in the outdoor hot tub, or playing card games, “our conversation didn’t surround what we’d have for dinner or who would do baths and bedtime with whom,” Thompson said. “We didn’t talk about work, upcoming commitments, or items on our to-do lists.” The getaway was so refreshing, the couple intend to repeat the trip each year.
Dx: Persistent Grief / Rx: Hiking and Hinduism in Nepal
Nearly 3 years ago, Funt experienced a 2-month period where both of her kids left for college and both her father and father-in-law passed away. Besieged by grief, she found herself questioning whether her best years were behind her. She was also grappling with her mortality, because she was then approaching 59, the age at which her own mother had died. So Funt decided to go trekking in Nepal. “I am a traveler — it’s what I do,” she said.
Having the trip to prepare for changed Funt’s whole outlook, she remembers. Throwing herself into the planning helped her transcend her grief. But being in Nepal was even more impactful. She and her husband spent hours trekking through majestic mountain ranges, which “touched their souls.” At a crematorium, they learned about Hindu beliefs on death, which helped them with the grieving process.
The trip “lifted me so high up on so many levels and brought me back to my authentic self,” Funt said. On her flight home from Kathmandu, she decided to start her travel business.
“I needed something else [in addition to radiology] to put my passion, heart, and creativity into, and it would be another way of doing service,” she explained.
Dx: Couch Potato Syndrome / Rx: Planning an Adventure
Like all of us, Funt knows exercise is important for health. But that knowledge alone doesn’t motivate her to move, she admitted. What does get her off the couch is scheduling an active trip — and then training for it. “When I have a goal tied to my values of adventure, connection, and community, fear will set in if I don’t start to move,” she said. It was after booking her Nepal trip (which included an 8-mile, 3000-foot trek) that Funt started getting in shape.
Travel has motivated Funt’s clients in similar ways. Last year, 8 months before one of her Morocco trips, Funt spoke over Zoom with a woman who’d just enrolled. This woman told her she’d signed up in order to commit to her health.
By the time Funt saw her again, on day 1 of the trip, the woman had lost 50 pounds. “It was the greatest transformation,” Funt recalled. “On the trip, she was the first one up the mountain and beamed the whole time. It was beautiful to watch her reclaim her power, body, and life.”
Getting Lost — Finding Inspiration
Since Thompson’s trip to Italy, she has traveled extensively, visiting nearly 25 countries. “Traveling inspired me to continue exploring the world and myself,” she said.
Since leading her first trip to Morocco in 2023, Funt said she’s received more letters of appreciation from her clients than her patients. The results from this type of travel therapy can be dramatic.
After a trip with Funt, one burned-out physician decided that she needed to find a job with a better work-life balance. An empty nester realized the “feeling of belonging and community” on the trip was what had been missing in her “regular” life. After returning home, she began rekindling relationships with old friends.
To many, a vacation is a treat. But, as Funt and Thompson have learned firsthand, it can also be a prescription — for ennui, sadness, loneliness, and all the physical issues that come with them. Sometimes, going far away helps you come home to yourself.
A version of this article first appeared on Medscape.com.
Whatever’s ailing you, a vacation might just be the cure. Yes, getting away can improve your health, according to research published in in 2023. It might help combat symptoms of aging, suggested a 2024 study in Journal of Travel Research. But it could also have even more powerful psychological and physical benefits, transforming your life before you pack a bag and long after you return home.
This news organization spoke with two healthcare professionals who believe in the healing power of travel. They shared which personal “diagnoses” they have successfully treated with faraway places and how this therapy might work for you.
Stacey Funt, MD, NBC-HWC, a radiologist at Northwell Health in Long Island, New York, started the boutique wellness adventure travel company, LH Adventure Travel, in 2023. Funt curates and leads small groups to destinations like Peru, Guatemala, Morocco, and Italy. Each tour incorporates tenets of lifestyle medicine, including healthy eating, movement, stress management, and community building.
Kiya Thompson, RN, a surgical trauma nurse for 20 years, was similarly inspired to share her passion for travel. She is now a certified family travel coach who helps parents plan meaningful trips through her company, LuckyBucky, LLC.
Dx: Self-Esteem Deficiency / Rx: Vivaldi in Venice
In June 2015, Thompson found herself at an all-time low. As a nurse, she felt confident that she was “built for the adrenaline rush and could take on anything.” But outside the trauma center, Thompson felt inadequate, her self-esteem eroded by years of abusive relationships. “The daily hardships of my personal life, combined with the mental fortitude it took to endure the demands of caring for the sickest of the sick, were incredibly weighty,” she recalled.
To escape, Thompson booked her first solo trip: 3 weeks in Italy. But days after she arrived, she felt the need to “escape her escape.” On a bus in Naples, she was pick-pocketed. The man she had been dating before her trip stopped responding to her messages. In her hotel room in Venice, she felt “lost, alone, and helpless.”
One evening, Thompson attended a small orchestral performance of Vivaldi’s “The Four Seasons” in a centuries-old church. The music triggered memories of her Italian grandparents at whose home she’d listened to the same piece.
“A switch flipped, and I changed my whole outlook,” she remembers.
During the concert, she reflected on strangers who had shown her kindness and care. A Canadian man who gave her €50 after her wallet was stolen. A friend-of-a-friend who showed her around Rome. The clerk at her Venice hotel who had offered her a hug.
“In the wake of experiencing the worst of people, I’d experienced so much more of the best of people; strangers who were willing to go above and beyond to help me,” Thompson said.
When Thompson returned home, she brought her new mindset along. “ My ability to problem-solve my way through a solo trip that presented unexpected hardships empowered me,” she explained. “I learned I was much more capable than I’d thought.”
Dx: Wilderness Phobia / Rx: A Safari in Tanzania
On an evening in the mid-1990s, Funt was alone in a tent on a budget camping safari in Tanzania. Animals roared threateningly outside the thin walls. Earlier that day, a vulture had ripped a sandwich out of her hands. Funt was frightened to the core. Worrying that she’d be the next meal for the local wildlife, she started to sob. “This was as raw as I had ever gotten at that point in my life,” she said.
Suddenly, Funt said her brain shifted into problem-solving mode. She made one small decision: To switch to a different Jeep for the next day’s excursion. Having made a seemingly insignificant choice, she felt calmer and no longer like a victim. It brought control. Instead of worrying, she began looking forward to the wildlife she would see.
In the morning, in the new Jeep, she befriended a nurse from Canada. Together, they visited the Maasai Mara tribe and nearby pubs, meeting members of the community.
“It was the most exciting experience of my life,” Funt said. “And it had started with me crying.”
Dx: Parenting-itis / Rx: A Mountain Getaway
As Thompson pointed out, sometimes the destination is secondary to the intension behind a trip. And the quality of the time away matters more than how long you can stay. After becoming parents 4 years ago, Thompson and her husband hadn’t traveled alone together. Like many parents of young children, they were short on time to relax and reconnect as a couple.
So Thompson planned a weekend trip to an isolated cabin in the Massanutten Mountain Range within the George Washington National Forest, about a 2-hour drive from their Washington, DC, area home.
“We put our devices away and focused on being completely present with one another,” said Thompson. The couple took a walk in the woods, where “all we could hear were drops of water from the snowmelt, the crunch of the snow beneath our feet, and the occasional bird looking for food,” she recalled. “There were no cars, no other people. It was quiet, calm, and incredibly peaceful.”
Whether sitting by the fire, soaking in the outdoor hot tub, or playing card games, “our conversation didn’t surround what we’d have for dinner or who would do baths and bedtime with whom,” Thompson said. “We didn’t talk about work, upcoming commitments, or items on our to-do lists.” The getaway was so refreshing, the couple intend to repeat the trip each year.
Dx: Persistent Grief / Rx: Hiking and Hinduism in Nepal
Nearly 3 years ago, Funt experienced a 2-month period where both of her kids left for college and both her father and father-in-law passed away. Besieged by grief, she found herself questioning whether her best years were behind her. She was also grappling with her mortality, because she was then approaching 59, the age at which her own mother had died. So Funt decided to go trekking in Nepal. “I am a traveler — it’s what I do,” she said.
Having the trip to prepare for changed Funt’s whole outlook, she remembers. Throwing herself into the planning helped her transcend her grief. But being in Nepal was even more impactful. She and her husband spent hours trekking through majestic mountain ranges, which “touched their souls.” At a crematorium, they learned about Hindu beliefs on death, which helped them with the grieving process.
The trip “lifted me so high up on so many levels and brought me back to my authentic self,” Funt said. On her flight home from Kathmandu, she decided to start her travel business.
“I needed something else [in addition to radiology] to put my passion, heart, and creativity into, and it would be another way of doing service,” she explained.
Dx: Couch Potato Syndrome / Rx: Planning an Adventure
Like all of us, Funt knows exercise is important for health. But that knowledge alone doesn’t motivate her to move, she admitted. What does get her off the couch is scheduling an active trip — and then training for it. “When I have a goal tied to my values of adventure, connection, and community, fear will set in if I don’t start to move,” she said. It was after booking her Nepal trip (which included an 8-mile, 3000-foot trek) that Funt started getting in shape.
Travel has motivated Funt’s clients in similar ways. Last year, 8 months before one of her Morocco trips, Funt spoke over Zoom with a woman who’d just enrolled. This woman told her she’d signed up in order to commit to her health.
By the time Funt saw her again, on day 1 of the trip, the woman had lost 50 pounds. “It was the greatest transformation,” Funt recalled. “On the trip, she was the first one up the mountain and beamed the whole time. It was beautiful to watch her reclaim her power, body, and life.”
Getting Lost — Finding Inspiration
Since Thompson’s trip to Italy, she has traveled extensively, visiting nearly 25 countries. “Traveling inspired me to continue exploring the world and myself,” she said.
Since leading her first trip to Morocco in 2023, Funt said she’s received more letters of appreciation from her clients than her patients. The results from this type of travel therapy can be dramatic.
After a trip with Funt, one burned-out physician decided that she needed to find a job with a better work-life balance. An empty nester realized the “feeling of belonging and community” on the trip was what had been missing in her “regular” life. After returning home, she began rekindling relationships with old friends.
To many, a vacation is a treat. But, as Funt and Thompson have learned firsthand, it can also be a prescription — for ennui, sadness, loneliness, and all the physical issues that come with them. Sometimes, going far away helps you come home to yourself.
A version of this article first appeared on Medscape.com.
Whatever’s ailing you, a vacation might just be the cure. Yes, getting away can improve your health, according to research published in in 2023. It might help combat symptoms of aging, suggested a 2024 study in Journal of Travel Research. But it could also have even more powerful psychological and physical benefits, transforming your life before you pack a bag and long after you return home.
This news organization spoke with two healthcare professionals who believe in the healing power of travel. They shared which personal “diagnoses” they have successfully treated with faraway places and how this therapy might work for you.
Stacey Funt, MD, NBC-HWC, a radiologist at Northwell Health in Long Island, New York, started the boutique wellness adventure travel company, LH Adventure Travel, in 2023. Funt curates and leads small groups to destinations like Peru, Guatemala, Morocco, and Italy. Each tour incorporates tenets of lifestyle medicine, including healthy eating, movement, stress management, and community building.
Kiya Thompson, RN, a surgical trauma nurse for 20 years, was similarly inspired to share her passion for travel. She is now a certified family travel coach who helps parents plan meaningful trips through her company, LuckyBucky, LLC.
Dx: Self-Esteem Deficiency / Rx: Vivaldi in Venice
In June 2015, Thompson found herself at an all-time low. As a nurse, she felt confident that she was “built for the adrenaline rush and could take on anything.” But outside the trauma center, Thompson felt inadequate, her self-esteem eroded by years of abusive relationships. “The daily hardships of my personal life, combined with the mental fortitude it took to endure the demands of caring for the sickest of the sick, were incredibly weighty,” she recalled.
To escape, Thompson booked her first solo trip: 3 weeks in Italy. But days after she arrived, she felt the need to “escape her escape.” On a bus in Naples, she was pick-pocketed. The man she had been dating before her trip stopped responding to her messages. In her hotel room in Venice, she felt “lost, alone, and helpless.”
One evening, Thompson attended a small orchestral performance of Vivaldi’s “The Four Seasons” in a centuries-old church. The music triggered memories of her Italian grandparents at whose home she’d listened to the same piece.
“A switch flipped, and I changed my whole outlook,” she remembers.
During the concert, she reflected on strangers who had shown her kindness and care. A Canadian man who gave her €50 after her wallet was stolen. A friend-of-a-friend who showed her around Rome. The clerk at her Venice hotel who had offered her a hug.
“In the wake of experiencing the worst of people, I’d experienced so much more of the best of people; strangers who were willing to go above and beyond to help me,” Thompson said.
When Thompson returned home, she brought her new mindset along. “ My ability to problem-solve my way through a solo trip that presented unexpected hardships empowered me,” she explained. “I learned I was much more capable than I’d thought.”
Dx: Wilderness Phobia / Rx: A Safari in Tanzania
On an evening in the mid-1990s, Funt was alone in a tent on a budget camping safari in Tanzania. Animals roared threateningly outside the thin walls. Earlier that day, a vulture had ripped a sandwich out of her hands. Funt was frightened to the core. Worrying that she’d be the next meal for the local wildlife, she started to sob. “This was as raw as I had ever gotten at that point in my life,” she said.
Suddenly, Funt said her brain shifted into problem-solving mode. She made one small decision: To switch to a different Jeep for the next day’s excursion. Having made a seemingly insignificant choice, she felt calmer and no longer like a victim. It brought control. Instead of worrying, she began looking forward to the wildlife she would see.
In the morning, in the new Jeep, she befriended a nurse from Canada. Together, they visited the Maasai Mara tribe and nearby pubs, meeting members of the community.
“It was the most exciting experience of my life,” Funt said. “And it had started with me crying.”
Dx: Parenting-itis / Rx: A Mountain Getaway
As Thompson pointed out, sometimes the destination is secondary to the intension behind a trip. And the quality of the time away matters more than how long you can stay. After becoming parents 4 years ago, Thompson and her husband hadn’t traveled alone together. Like many parents of young children, they were short on time to relax and reconnect as a couple.
So Thompson planned a weekend trip to an isolated cabin in the Massanutten Mountain Range within the George Washington National Forest, about a 2-hour drive from their Washington, DC, area home.
“We put our devices away and focused on being completely present with one another,” said Thompson. The couple took a walk in the woods, where “all we could hear were drops of water from the snowmelt, the crunch of the snow beneath our feet, and the occasional bird looking for food,” she recalled. “There were no cars, no other people. It was quiet, calm, and incredibly peaceful.”
Whether sitting by the fire, soaking in the outdoor hot tub, or playing card games, “our conversation didn’t surround what we’d have for dinner or who would do baths and bedtime with whom,” Thompson said. “We didn’t talk about work, upcoming commitments, or items on our to-do lists.” The getaway was so refreshing, the couple intend to repeat the trip each year.
Dx: Persistent Grief / Rx: Hiking and Hinduism in Nepal
Nearly 3 years ago, Funt experienced a 2-month period where both of her kids left for college and both her father and father-in-law passed away. Besieged by grief, she found herself questioning whether her best years were behind her. She was also grappling with her mortality, because she was then approaching 59, the age at which her own mother had died. So Funt decided to go trekking in Nepal. “I am a traveler — it’s what I do,” she said.
Having the trip to prepare for changed Funt’s whole outlook, she remembers. Throwing herself into the planning helped her transcend her grief. But being in Nepal was even more impactful. She and her husband spent hours trekking through majestic mountain ranges, which “touched their souls.” At a crematorium, they learned about Hindu beliefs on death, which helped them with the grieving process.
The trip “lifted me so high up on so many levels and brought me back to my authentic self,” Funt said. On her flight home from Kathmandu, she decided to start her travel business.
“I needed something else [in addition to radiology] to put my passion, heart, and creativity into, and it would be another way of doing service,” she explained.
Dx: Couch Potato Syndrome / Rx: Planning an Adventure
Like all of us, Funt knows exercise is important for health. But that knowledge alone doesn’t motivate her to move, she admitted. What does get her off the couch is scheduling an active trip — and then training for it. “When I have a goal tied to my values of adventure, connection, and community, fear will set in if I don’t start to move,” she said. It was after booking her Nepal trip (which included an 8-mile, 3000-foot trek) that Funt started getting in shape.
Travel has motivated Funt’s clients in similar ways. Last year, 8 months before one of her Morocco trips, Funt spoke over Zoom with a woman who’d just enrolled. This woman told her she’d signed up in order to commit to her health.
By the time Funt saw her again, on day 1 of the trip, the woman had lost 50 pounds. “It was the greatest transformation,” Funt recalled. “On the trip, she was the first one up the mountain and beamed the whole time. It was beautiful to watch her reclaim her power, body, and life.”
Getting Lost — Finding Inspiration
Since Thompson’s trip to Italy, she has traveled extensively, visiting nearly 25 countries. “Traveling inspired me to continue exploring the world and myself,” she said.
Since leading her first trip to Morocco in 2023, Funt said she’s received more letters of appreciation from her clients than her patients. The results from this type of travel therapy can be dramatic.
After a trip with Funt, one burned-out physician decided that she needed to find a job with a better work-life balance. An empty nester realized the “feeling of belonging and community” on the trip was what had been missing in her “regular” life. After returning home, she began rekindling relationships with old friends.
To many, a vacation is a treat. But, as Funt and Thompson have learned firsthand, it can also be a prescription — for ennui, sadness, loneliness, and all the physical issues that come with them. Sometimes, going far away helps you come home to yourself.
A version of this article first appeared on Medscape.com.
Scholarly Activity Among VA Podiatrists: A Cross-Sectional Study
Scholarly Activity Among VA Podiatrists: A Cross-Sectional Study
The US Department of Veterans Affairs (VA) delivers care to > 9 million veterans, including primary and specialty care.1 While clinical duties remain important across the health system, proposed productivity models have included clinician research activity, given that many hold roles in academia.2 Within this framework, research plays a pivotal role in advancing clinical practices and outcomes. Studies have found that physicians who participated in research report higher job satisfaction.3
As a specialty within the VA, podiatrists diagnose, treat, and prevent foot and ankle disorders. In addition to clinical practice, various scholarly activities are shared among these physicians.4 Reasons for scholarly pursuits among podiatrists vary, including participation in research for academic promotion or to establish expertise in a given area.4-7 Although research remains a component associated with promotion within the VA, little is known about the scholarly activity of VA podiatrists. Specifically, there remains a paucity of data concerning their expertise, as evidenced through peer-reviewed publications, among these physicians and surgeons. To date, no analysis of scholarly activity among VA podiatrists has been conducted.
The primary aim of this investigation was to describe the scholarly productivity among podiatrists employed by the VA through an analysis of the number of peer-reviewed publications and the respective h-index of each physician. The secondary aim of this investigation was to assess the effect of academic productivity on compensation. This study describes research activities pursued by VA physicians and provides the veteran patient population with the confidence that their foot health care remains in the hands of experts within the field.
MATERIALS AND METHODS
The Feds Data Center (www.fedsdatacenter.com) online database of employees was used to identify VA podiatrists on June 17, 2024. All GS-15 physicians and their respective salaries in fiscal year 2023 were recorded. Administratively determined employees, including residents, were excluded. The h-index and number of published documents from any point during a physician’s training or career were reported for each podiatrist using Scopus; podiatrists without an h-index or publication were excluded. 8 Among podiatrists with scholarly activity, this analysis collected academic appointment, sex, and region of practice.
Statistical Analysis
Descriptive statistics, presented as counts and frequencies, were used. The median and IQR were used to describe the number of publications and h-index due to their nonnormal distribution. A Kruskal-Wallis test was used to compare median publication counts and h-index values among for junior faculty (JF), which includes instructors and assistant professors; senior faculty (SF), which includes associate professors and professors; and those with no academic affiliation (NF). Salary was reported as mean (SD) as it remained normally distributed and was compared using analysis of variance with posthoc Tukey test to increase statistical power. Additionally, this analysis used linear regression to investigate the relationship between scholarly activity and salary. The threshold for statistical significance was set at P < .05.
RESULTS
Among 819 VA podiatrists, 150 were administratively determined and excluded, and 512 were excluded for no history of publications, leaving 157 eligible for analysis (Table). A statistically significant difference was found in median (IQR) publication count by faculty appointment. JF had 6.0 (9.5), SF had 12.5 (22.3), and NF had 1.0 (2.0) publication(s) (P < .001) (Figure 1A). There was a statistically significant difference in h-index by faculty appointment. The median (IQR) h-index for JF was 2.0 (3.5), for SF was 5.5 (4.25), and for NF was 1.0 (2.0) (P = .002) (Figure 1B). Salary was not significantly associated with publication count (P = .20) or h-index (P = .62) (Figure 2). No statistically significant difference was found between academic appointment and mean (SD) salary. JF had a median (IQR) salary of $224,063 (27,989), SF of $234,260 (42,963), and NF of $219,811 (P = .35).
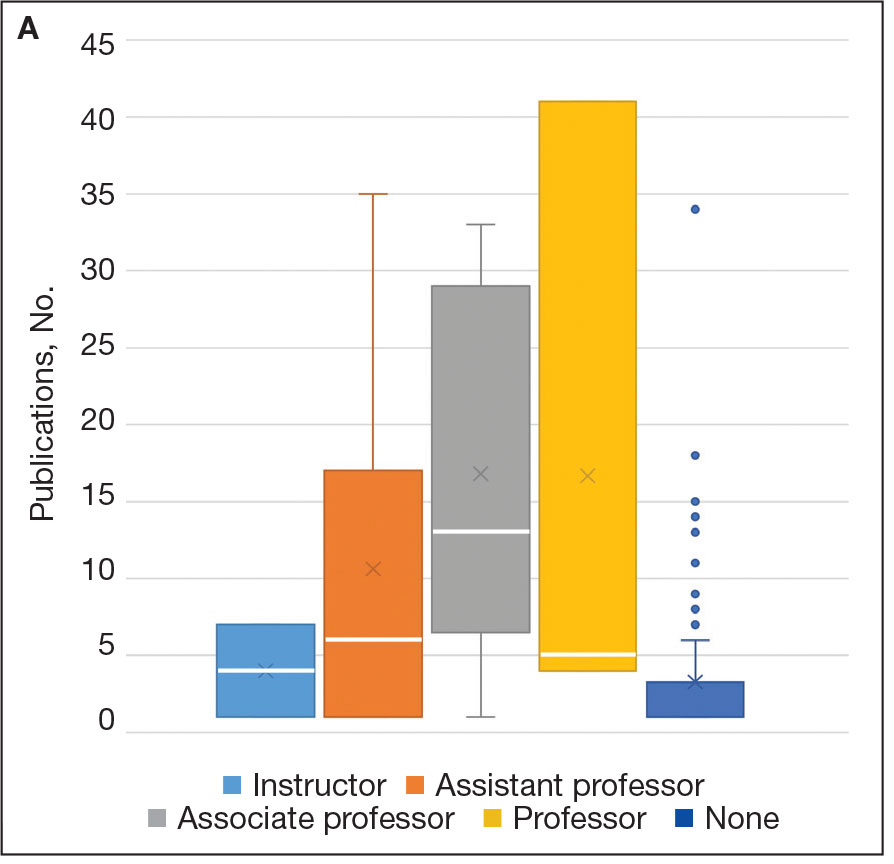
(B) h-index.a
aBox sizes indicate IQR (bottom, IQR 1; top, IQR 3); whiskers indicate minimum and maximum within 1.5 x IQR; Xs indicate means; white
lines indicate medians; and dots indicate outliers.
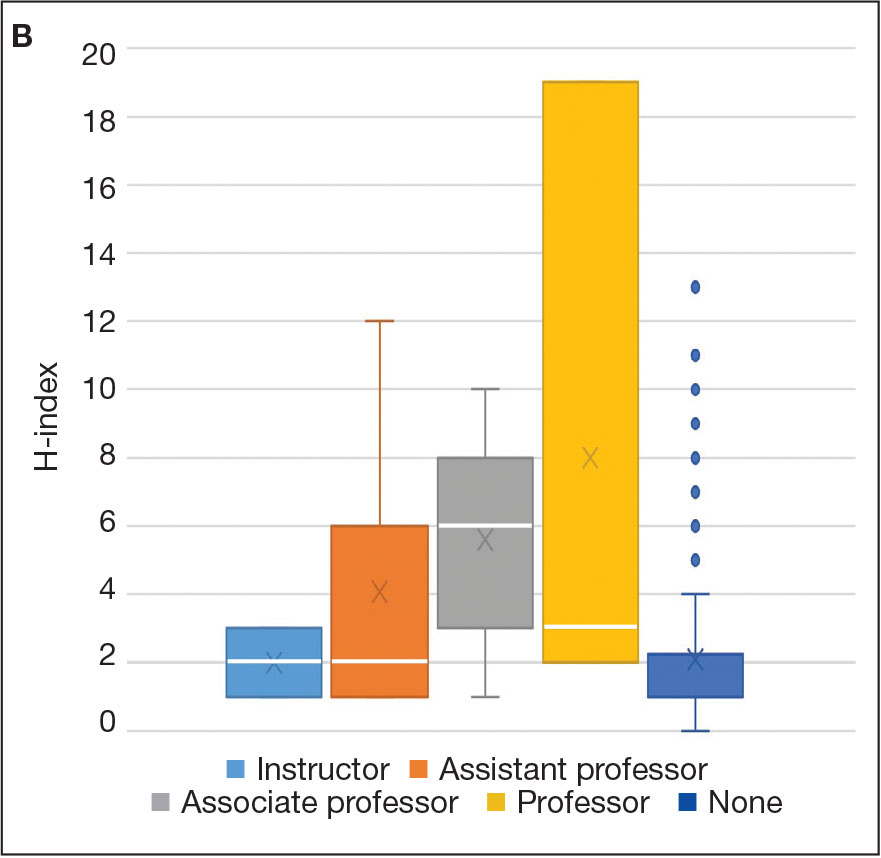
(B) h-index.a
aBox sizes indicate IQR (bottom, IQR 1; top, IQR 3); whiskers indicate minimum and maximum within 1.5 x IQR; Xs indicate means; white
lines indicate medians; and dots indicate outliers.
DISCUSSION
Focused on providing high-quality care, VA physicians use their expertise to practice comprehensive and specialized care.9,10 A cornerstone to this expertise is scholarly activity that contributes to the body of knowledge and, ultimately, the evidence-based medicine by which these physicians practice.11 With veterans considering VA care, it is important to highlight the commitment and dedication to the science and the practice of medicine. This analysis describes the scholarly activity of VA podiatrists and underscores the expertise veterans will receive for the diagnosis and treatment of their foot and ankle pathology.
were not part of an academic facility, a finding that may encourage further action to increase academic productivity in this specialty. For example, collaboration through academic affiliations has been seen throughout VA medical and surgical specialties and provides many benefits. Beginning with graduate medical education, the VA serves as a tremendous resource for resident training.12 Additionally, veterans who sought emergency care at the VA had a lower risk of death than those treated at non-VA hospitals.13 In podiatric medicine and surgery, scholarly activity has been linked to improved outcomes, particularly in the study of ulceration development and its role in either prolonging or preventing amputation.14
Beyond improving clinical outcomes and patient care, engagement in research and inquiry offers other benefits. A cross-sectional study of 7734 physicians within the VA found that research involvement was associated with more favorable job characteristics and job satisfaction perceptions. 3 While this analysis found that about 19% of podiatrists have published once in their career, it remains likely that more may continue to engage in research during their VA tenure. Although this finding shows that an appreciable number of VA podiatrists have published in their field of study, it also encourages departments to provide resources to engage in research. Similar to previous research among foot and ankle surgeons, this analysis also found an increase in publications and h-index as tenure increased.4 Unlike previous research, which found h-index and academic appointment to be contributors to VA dermatologists’ salaries, no significant difference in salary was found in this study associated with publications, h-index, or academic role.15 Although the increase was not statistically significant, salary tended to rise as these variables increased.
Limitations
This analysis was confined to the most recent year of available data, which may not fully capture the longitudinal academic contributions and trends of individual podiatrists. Academic productivity can fluctuate significantly over time due to various factors, including changes in research focus and administrative responsibilities. The study also relied on Scopus to identify and quantify academic productivity. This database may not include all publications relevant to podiatrists, particularly those in niche or nonindexed journals. Additionally, name variations and potential misspellings could lead to missing data for individual podiatrists’ publications. Furthermore, this study did not account for other significant contributors to salary and career advancement within the federal system. Factors such as clinical performance, administrative duties, patient satisfaction, and contributions to teaching and mentoring are critical elements that also influence career progression and compensation but were not captured in this analysis. The retrospective design of this study inherently limits the ability to establish causal relationships. While associations between academic productivity and certain outcomes may be identified, it is not possible to definitively determine the direction or causality of these relationships. Future research may examine how scholarly activity continues once a clinician is part of VA.
CONCLUSIONS
This study highlights the significant academic contributions of VA podiatrists to research and the medical literature. By fostering an active research environment, the VA can ensure veterans receive the highest quality of care from knowledgeable and expert clinicians. Future research should aim to provide a more comprehensive analysis, capturing long-term trends and considering all factors influencing career advancement in VA.
- Rosland AM, Nelson K, Sun H, et al. The patient-centered medical home in the Veterans Health Administration. Am J Manag Care. 2013;19(7):e263-e272.
- Coleman DL, Moran E, Serfilippi D, et al. Measuring physicians’ productivity in a Veterans’ Affairs Medical Center. Acad Med. 2003;78(7):682-689. doi:10.1097/00001888-200307000-00007
- Mohr DC, Burgess JF Jr. Job characteristics and job satisfaction among physicians involved with research in the Veterans Health Administration. Acad Med. 2011;86(8):938-945. doi:10.1097/ACM.0b013e3182223b76
- Casciato DJ, Cravey KS, Barron IM. Scholarly productivity among academic foot and ankle surgeons affiliated with US podiatric medicine and surgery residency and fellowship training programs. J Foot Ankle Surg. 2021;60(6):1222-1226. doi:10.1053/j.jfas.2021.04.017
- Hyer CF, Casciato DJ, Rushing CJ, Schuberth JM. Incidence of scholarly publication by selected content experts presenting at national society foot and ankle meetings from 2016 to 2020. J Foot Ankle Surg. 2022;61(6):1317-1320. doi:10.1053/j.jfas.2022.04.011
- Casciato DJ, Thompson J, Yancovitz S, Chandra A, Prissel MA, Hyer CF. Research activity among foot and ankle surgery fellows: a systematic review. J Foot Ankle Surg. 2021;60(6):1227-1231. doi:10.1053/j.jfas.2021.04.018
- Casciato DJ, Thompson J, Hyer CF. Post-fellowship foot and ankle surgeon research productivity: a systematic review. J Foot Ankle Surg. 2022;61(4):896-899. doi:10.1053/j.jfas.2021.12.028
- Hirsch JE. An index to quantify an individual’s scientific research output. Proc Natl Acad Sci USA. 2005;102(46):16569-16572. doi:10.1073/pnas.0507655102
- US Department of Veterans Affairs. Veterans Health Administration. About VHA. Updated January 20, 2025. Accessed February 17, 2025. https://www.va.gov/health/aboutvha.asp
- US Department of Veterans Affairs. VHA National Center for Patient Safety. About Us. Updated November 29, 2023. Accessed February 17, 2025. https://www.patientsafety.va.gov/
- US Department of Veterans Affairs. VA/DoD Clinical Practice Guidelines. Updated February 7, 2025. Accessed February 17, 2025. https://www.healthquality.va.gov
- Ravin AG, Gottlieb NB, Wang HT, et al. Effect of the Veterans Affairs Medical System on plastic surgery residency training. Plast Reconstr Surg. 2006;117(2):656-660. doi:10.1097/01.prs.0000197216.95544.f7
- Chan DC, Danesh K, Costantini S, Card D, Taylor L, Studdert DM. Mortality among US veterans after emergency visits to Veterans Affairs and other hospitals: retrospective cohort study. BMJ. 2022;376:e068099. doi:10.1136/bmj-2021-068099
- Gibson LW, Abbas A. Limb salvage for veterans with diabetes: to care for him who has borne the battle. Crit Care Nurs Clin North Am. 2013;25(1):131-134. doi:10.1016/j.ccell.2012.11.004
- Do MH, Lipner SR. Contribution of gender on compensation of Veterans Affairs-affiliated dermatologists: a cross-sectional study. Int J Womens Dermatol. 2020;6(5):414-418. doi:10.1016/j.ijwd.2020.09.009
The US Department of Veterans Affairs (VA) delivers care to > 9 million veterans, including primary and specialty care.1 While clinical duties remain important across the health system, proposed productivity models have included clinician research activity, given that many hold roles in academia.2 Within this framework, research plays a pivotal role in advancing clinical practices and outcomes. Studies have found that physicians who participated in research report higher job satisfaction.3
As a specialty within the VA, podiatrists diagnose, treat, and prevent foot and ankle disorders. In addition to clinical practice, various scholarly activities are shared among these physicians.4 Reasons for scholarly pursuits among podiatrists vary, including participation in research for academic promotion or to establish expertise in a given area.4-7 Although research remains a component associated with promotion within the VA, little is known about the scholarly activity of VA podiatrists. Specifically, there remains a paucity of data concerning their expertise, as evidenced through peer-reviewed publications, among these physicians and surgeons. To date, no analysis of scholarly activity among VA podiatrists has been conducted.
The primary aim of this investigation was to describe the scholarly productivity among podiatrists employed by the VA through an analysis of the number of peer-reviewed publications and the respective h-index of each physician. The secondary aim of this investigation was to assess the effect of academic productivity on compensation. This study describes research activities pursued by VA physicians and provides the veteran patient population with the confidence that their foot health care remains in the hands of experts within the field.
MATERIALS AND METHODS
The Feds Data Center (www.fedsdatacenter.com) online database of employees was used to identify VA podiatrists on June 17, 2024. All GS-15 physicians and their respective salaries in fiscal year 2023 were recorded. Administratively determined employees, including residents, were excluded. The h-index and number of published documents from any point during a physician’s training or career were reported for each podiatrist using Scopus; podiatrists without an h-index or publication were excluded. 8 Among podiatrists with scholarly activity, this analysis collected academic appointment, sex, and region of practice.
Statistical Analysis
Descriptive statistics, presented as counts and frequencies, were used. The median and IQR were used to describe the number of publications and h-index due to their nonnormal distribution. A Kruskal-Wallis test was used to compare median publication counts and h-index values among for junior faculty (JF), which includes instructors and assistant professors; senior faculty (SF), which includes associate professors and professors; and those with no academic affiliation (NF). Salary was reported as mean (SD) as it remained normally distributed and was compared using analysis of variance with posthoc Tukey test to increase statistical power. Additionally, this analysis used linear regression to investigate the relationship between scholarly activity and salary. The threshold for statistical significance was set at P < .05.
RESULTS
Among 819 VA podiatrists, 150 were administratively determined and excluded, and 512 were excluded for no history of publications, leaving 157 eligible for analysis (Table). A statistically significant difference was found in median (IQR) publication count by faculty appointment. JF had 6.0 (9.5), SF had 12.5 (22.3), and NF had 1.0 (2.0) publication(s) (P < .001) (Figure 1A). There was a statistically significant difference in h-index by faculty appointment. The median (IQR) h-index for JF was 2.0 (3.5), for SF was 5.5 (4.25), and for NF was 1.0 (2.0) (P = .002) (Figure 1B). Salary was not significantly associated with publication count (P = .20) or h-index (P = .62) (Figure 2). No statistically significant difference was found between academic appointment and mean (SD) salary. JF had a median (IQR) salary of $224,063 (27,989), SF of $234,260 (42,963), and NF of $219,811 (P = .35).

(B) h-index.a
aBox sizes indicate IQR (bottom, IQR 1; top, IQR 3); whiskers indicate minimum and maximum within 1.5 x IQR; Xs indicate means; white
lines indicate medians; and dots indicate outliers.

(B) h-index.a
aBox sizes indicate IQR (bottom, IQR 1; top, IQR 3); whiskers indicate minimum and maximum within 1.5 x IQR; Xs indicate means; white
lines indicate medians; and dots indicate outliers.


DISCUSSION
Focused on providing high-quality care, VA physicians use their expertise to practice comprehensive and specialized care.9,10 A cornerstone to this expertise is scholarly activity that contributes to the body of knowledge and, ultimately, the evidence-based medicine by which these physicians practice.11 With veterans considering VA care, it is important to highlight the commitment and dedication to the science and the practice of medicine. This analysis describes the scholarly activity of VA podiatrists and underscores the expertise veterans will receive for the diagnosis and treatment of their foot and ankle pathology.
were not part of an academic facility, a finding that may encourage further action to increase academic productivity in this specialty. For example, collaboration through academic affiliations has been seen throughout VA medical and surgical specialties and provides many benefits. Beginning with graduate medical education, the VA serves as a tremendous resource for resident training.12 Additionally, veterans who sought emergency care at the VA had a lower risk of death than those treated at non-VA hospitals.13 In podiatric medicine and surgery, scholarly activity has been linked to improved outcomes, particularly in the study of ulceration development and its role in either prolonging or preventing amputation.14
Beyond improving clinical outcomes and patient care, engagement in research and inquiry offers other benefits. A cross-sectional study of 7734 physicians within the VA found that research involvement was associated with more favorable job characteristics and job satisfaction perceptions. 3 While this analysis found that about 19% of podiatrists have published once in their career, it remains likely that more may continue to engage in research during their VA tenure. Although this finding shows that an appreciable number of VA podiatrists have published in their field of study, it also encourages departments to provide resources to engage in research. Similar to previous research among foot and ankle surgeons, this analysis also found an increase in publications and h-index as tenure increased.4 Unlike previous research, which found h-index and academic appointment to be contributors to VA dermatologists’ salaries, no significant difference in salary was found in this study associated with publications, h-index, or academic role.15 Although the increase was not statistically significant, salary tended to rise as these variables increased.
Limitations
This analysis was confined to the most recent year of available data, which may not fully capture the longitudinal academic contributions and trends of individual podiatrists. Academic productivity can fluctuate significantly over time due to various factors, including changes in research focus and administrative responsibilities. The study also relied on Scopus to identify and quantify academic productivity. This database may not include all publications relevant to podiatrists, particularly those in niche or nonindexed journals. Additionally, name variations and potential misspellings could lead to missing data for individual podiatrists’ publications. Furthermore, this study did not account for other significant contributors to salary and career advancement within the federal system. Factors such as clinical performance, administrative duties, patient satisfaction, and contributions to teaching and mentoring are critical elements that also influence career progression and compensation but were not captured in this analysis. The retrospective design of this study inherently limits the ability to establish causal relationships. While associations between academic productivity and certain outcomes may be identified, it is not possible to definitively determine the direction or causality of these relationships. Future research may examine how scholarly activity continues once a clinician is part of VA.
CONCLUSIONS
This study highlights the significant academic contributions of VA podiatrists to research and the medical literature. By fostering an active research environment, the VA can ensure veterans receive the highest quality of care from knowledgeable and expert clinicians. Future research should aim to provide a more comprehensive analysis, capturing long-term trends and considering all factors influencing career advancement in VA.
The US Department of Veterans Affairs (VA) delivers care to > 9 million veterans, including primary and specialty care.1 While clinical duties remain important across the health system, proposed productivity models have included clinician research activity, given that many hold roles in academia.2 Within this framework, research plays a pivotal role in advancing clinical practices and outcomes. Studies have found that physicians who participated in research report higher job satisfaction.3
As a specialty within the VA, podiatrists diagnose, treat, and prevent foot and ankle disorders. In addition to clinical practice, various scholarly activities are shared among these physicians.4 Reasons for scholarly pursuits among podiatrists vary, including participation in research for academic promotion or to establish expertise in a given area.4-7 Although research remains a component associated with promotion within the VA, little is known about the scholarly activity of VA podiatrists. Specifically, there remains a paucity of data concerning their expertise, as evidenced through peer-reviewed publications, among these physicians and surgeons. To date, no analysis of scholarly activity among VA podiatrists has been conducted.
The primary aim of this investigation was to describe the scholarly productivity among podiatrists employed by the VA through an analysis of the number of peer-reviewed publications and the respective h-index of each physician. The secondary aim of this investigation was to assess the effect of academic productivity on compensation. This study describes research activities pursued by VA physicians and provides the veteran patient population with the confidence that their foot health care remains in the hands of experts within the field.
MATERIALS AND METHODS
The Feds Data Center (www.fedsdatacenter.com) online database of employees was used to identify VA podiatrists on June 17, 2024. All GS-15 physicians and their respective salaries in fiscal year 2023 were recorded. Administratively determined employees, including residents, were excluded. The h-index and number of published documents from any point during a physician’s training or career were reported for each podiatrist using Scopus; podiatrists without an h-index or publication were excluded. 8 Among podiatrists with scholarly activity, this analysis collected academic appointment, sex, and region of practice.
Statistical Analysis
Descriptive statistics, presented as counts and frequencies, were used. The median and IQR were used to describe the number of publications and h-index due to their nonnormal distribution. A Kruskal-Wallis test was used to compare median publication counts and h-index values among for junior faculty (JF), which includes instructors and assistant professors; senior faculty (SF), which includes associate professors and professors; and those with no academic affiliation (NF). Salary was reported as mean (SD) as it remained normally distributed and was compared using analysis of variance with posthoc Tukey test to increase statistical power. Additionally, this analysis used linear regression to investigate the relationship between scholarly activity and salary. The threshold for statistical significance was set at P < .05.
RESULTS
Among 819 VA podiatrists, 150 were administratively determined and excluded, and 512 were excluded for no history of publications, leaving 157 eligible for analysis (Table). A statistically significant difference was found in median (IQR) publication count by faculty appointment. JF had 6.0 (9.5), SF had 12.5 (22.3), and NF had 1.0 (2.0) publication(s) (P < .001) (Figure 1A). There was a statistically significant difference in h-index by faculty appointment. The median (IQR) h-index for JF was 2.0 (3.5), for SF was 5.5 (4.25), and for NF was 1.0 (2.0) (P = .002) (Figure 1B). Salary was not significantly associated with publication count (P = .20) or h-index (P = .62) (Figure 2). No statistically significant difference was found between academic appointment and mean (SD) salary. JF had a median (IQR) salary of $224,063 (27,989), SF of $234,260 (42,963), and NF of $219,811 (P = .35).

(B) h-index.a
aBox sizes indicate IQR (bottom, IQR 1; top, IQR 3); whiskers indicate minimum and maximum within 1.5 x IQR; Xs indicate means; white
lines indicate medians; and dots indicate outliers.

(B) h-index.a
aBox sizes indicate IQR (bottom, IQR 1; top, IQR 3); whiskers indicate minimum and maximum within 1.5 x IQR; Xs indicate means; white
lines indicate medians; and dots indicate outliers.


DISCUSSION
Focused on providing high-quality care, VA physicians use their expertise to practice comprehensive and specialized care.9,10 A cornerstone to this expertise is scholarly activity that contributes to the body of knowledge and, ultimately, the evidence-based medicine by which these physicians practice.11 With veterans considering VA care, it is important to highlight the commitment and dedication to the science and the practice of medicine. This analysis describes the scholarly activity of VA podiatrists and underscores the expertise veterans will receive for the diagnosis and treatment of their foot and ankle pathology.
were not part of an academic facility, a finding that may encourage further action to increase academic productivity in this specialty. For example, collaboration through academic affiliations has been seen throughout VA medical and surgical specialties and provides many benefits. Beginning with graduate medical education, the VA serves as a tremendous resource for resident training.12 Additionally, veterans who sought emergency care at the VA had a lower risk of death than those treated at non-VA hospitals.13 In podiatric medicine and surgery, scholarly activity has been linked to improved outcomes, particularly in the study of ulceration development and its role in either prolonging or preventing amputation.14
Beyond improving clinical outcomes and patient care, engagement in research and inquiry offers other benefits. A cross-sectional study of 7734 physicians within the VA found that research involvement was associated with more favorable job characteristics and job satisfaction perceptions. 3 While this analysis found that about 19% of podiatrists have published once in their career, it remains likely that more may continue to engage in research during their VA tenure. Although this finding shows that an appreciable number of VA podiatrists have published in their field of study, it also encourages departments to provide resources to engage in research. Similar to previous research among foot and ankle surgeons, this analysis also found an increase in publications and h-index as tenure increased.4 Unlike previous research, which found h-index and academic appointment to be contributors to VA dermatologists’ salaries, no significant difference in salary was found in this study associated with publications, h-index, or academic role.15 Although the increase was not statistically significant, salary tended to rise as these variables increased.
Limitations
This analysis was confined to the most recent year of available data, which may not fully capture the longitudinal academic contributions and trends of individual podiatrists. Academic productivity can fluctuate significantly over time due to various factors, including changes in research focus and administrative responsibilities. The study also relied on Scopus to identify and quantify academic productivity. This database may not include all publications relevant to podiatrists, particularly those in niche or nonindexed journals. Additionally, name variations and potential misspellings could lead to missing data for individual podiatrists’ publications. Furthermore, this study did not account for other significant contributors to salary and career advancement within the federal system. Factors such as clinical performance, administrative duties, patient satisfaction, and contributions to teaching and mentoring are critical elements that also influence career progression and compensation but were not captured in this analysis. The retrospective design of this study inherently limits the ability to establish causal relationships. While associations between academic productivity and certain outcomes may be identified, it is not possible to definitively determine the direction or causality of these relationships. Future research may examine how scholarly activity continues once a clinician is part of VA.
CONCLUSIONS
This study highlights the significant academic contributions of VA podiatrists to research and the medical literature. By fostering an active research environment, the VA can ensure veterans receive the highest quality of care from knowledgeable and expert clinicians. Future research should aim to provide a more comprehensive analysis, capturing long-term trends and considering all factors influencing career advancement in VA.
- Rosland AM, Nelson K, Sun H, et al. The patient-centered medical home in the Veterans Health Administration. Am J Manag Care. 2013;19(7):e263-e272.
- Coleman DL, Moran E, Serfilippi D, et al. Measuring physicians’ productivity in a Veterans’ Affairs Medical Center. Acad Med. 2003;78(7):682-689. doi:10.1097/00001888-200307000-00007
- Mohr DC, Burgess JF Jr. Job characteristics and job satisfaction among physicians involved with research in the Veterans Health Administration. Acad Med. 2011;86(8):938-945. doi:10.1097/ACM.0b013e3182223b76
- Casciato DJ, Cravey KS, Barron IM. Scholarly productivity among academic foot and ankle surgeons affiliated with US podiatric medicine and surgery residency and fellowship training programs. J Foot Ankle Surg. 2021;60(6):1222-1226. doi:10.1053/j.jfas.2021.04.017
- Hyer CF, Casciato DJ, Rushing CJ, Schuberth JM. Incidence of scholarly publication by selected content experts presenting at national society foot and ankle meetings from 2016 to 2020. J Foot Ankle Surg. 2022;61(6):1317-1320. doi:10.1053/j.jfas.2022.04.011
- Casciato DJ, Thompson J, Yancovitz S, Chandra A, Prissel MA, Hyer CF. Research activity among foot and ankle surgery fellows: a systematic review. J Foot Ankle Surg. 2021;60(6):1227-1231. doi:10.1053/j.jfas.2021.04.018
- Casciato DJ, Thompson J, Hyer CF. Post-fellowship foot and ankle surgeon research productivity: a systematic review. J Foot Ankle Surg. 2022;61(4):896-899. doi:10.1053/j.jfas.2021.12.028
- Hirsch JE. An index to quantify an individual’s scientific research output. Proc Natl Acad Sci USA. 2005;102(46):16569-16572. doi:10.1073/pnas.0507655102
- US Department of Veterans Affairs. Veterans Health Administration. About VHA. Updated January 20, 2025. Accessed February 17, 2025. https://www.va.gov/health/aboutvha.asp
- US Department of Veterans Affairs. VHA National Center for Patient Safety. About Us. Updated November 29, 2023. Accessed February 17, 2025. https://www.patientsafety.va.gov/
- US Department of Veterans Affairs. VA/DoD Clinical Practice Guidelines. Updated February 7, 2025. Accessed February 17, 2025. https://www.healthquality.va.gov
- Ravin AG, Gottlieb NB, Wang HT, et al. Effect of the Veterans Affairs Medical System on plastic surgery residency training. Plast Reconstr Surg. 2006;117(2):656-660. doi:10.1097/01.prs.0000197216.95544.f7
- Chan DC, Danesh K, Costantini S, Card D, Taylor L, Studdert DM. Mortality among US veterans after emergency visits to Veterans Affairs and other hospitals: retrospective cohort study. BMJ. 2022;376:e068099. doi:10.1136/bmj-2021-068099
- Gibson LW, Abbas A. Limb salvage for veterans with diabetes: to care for him who has borne the battle. Crit Care Nurs Clin North Am. 2013;25(1):131-134. doi:10.1016/j.ccell.2012.11.004
- Do MH, Lipner SR. Contribution of gender on compensation of Veterans Affairs-affiliated dermatologists: a cross-sectional study. Int J Womens Dermatol. 2020;6(5):414-418. doi:10.1016/j.ijwd.2020.09.009
- Rosland AM, Nelson K, Sun H, et al. The patient-centered medical home in the Veterans Health Administration. Am J Manag Care. 2013;19(7):e263-e272.
- Coleman DL, Moran E, Serfilippi D, et al. Measuring physicians’ productivity in a Veterans’ Affairs Medical Center. Acad Med. 2003;78(7):682-689. doi:10.1097/00001888-200307000-00007
- Mohr DC, Burgess JF Jr. Job characteristics and job satisfaction among physicians involved with research in the Veterans Health Administration. Acad Med. 2011;86(8):938-945. doi:10.1097/ACM.0b013e3182223b76
- Casciato DJ, Cravey KS, Barron IM. Scholarly productivity among academic foot and ankle surgeons affiliated with US podiatric medicine and surgery residency and fellowship training programs. J Foot Ankle Surg. 2021;60(6):1222-1226. doi:10.1053/j.jfas.2021.04.017
- Hyer CF, Casciato DJ, Rushing CJ, Schuberth JM. Incidence of scholarly publication by selected content experts presenting at national society foot and ankle meetings from 2016 to 2020. J Foot Ankle Surg. 2022;61(6):1317-1320. doi:10.1053/j.jfas.2022.04.011
- Casciato DJ, Thompson J, Yancovitz S, Chandra A, Prissel MA, Hyer CF. Research activity among foot and ankle surgery fellows: a systematic review. J Foot Ankle Surg. 2021;60(6):1227-1231. doi:10.1053/j.jfas.2021.04.018
- Casciato DJ, Thompson J, Hyer CF. Post-fellowship foot and ankle surgeon research productivity: a systematic review. J Foot Ankle Surg. 2022;61(4):896-899. doi:10.1053/j.jfas.2021.12.028
- Hirsch JE. An index to quantify an individual’s scientific research output. Proc Natl Acad Sci USA. 2005;102(46):16569-16572. doi:10.1073/pnas.0507655102
- US Department of Veterans Affairs. Veterans Health Administration. About VHA. Updated January 20, 2025. Accessed February 17, 2025. https://www.va.gov/health/aboutvha.asp
- US Department of Veterans Affairs. VHA National Center for Patient Safety. About Us. Updated November 29, 2023. Accessed February 17, 2025. https://www.patientsafety.va.gov/
- US Department of Veterans Affairs. VA/DoD Clinical Practice Guidelines. Updated February 7, 2025. Accessed February 17, 2025. https://www.healthquality.va.gov
- Ravin AG, Gottlieb NB, Wang HT, et al. Effect of the Veterans Affairs Medical System on plastic surgery residency training. Plast Reconstr Surg. 2006;117(2):656-660. doi:10.1097/01.prs.0000197216.95544.f7
- Chan DC, Danesh K, Costantini S, Card D, Taylor L, Studdert DM. Mortality among US veterans after emergency visits to Veterans Affairs and other hospitals: retrospective cohort study. BMJ. 2022;376:e068099. doi:10.1136/bmj-2021-068099
- Gibson LW, Abbas A. Limb salvage for veterans with diabetes: to care for him who has borne the battle. Crit Care Nurs Clin North Am. 2013;25(1):131-134. doi:10.1016/j.ccell.2012.11.004
- Do MH, Lipner SR. Contribution of gender on compensation of Veterans Affairs-affiliated dermatologists: a cross-sectional study. Int J Womens Dermatol. 2020;6(5):414-418. doi:10.1016/j.ijwd.2020.09.009
Scholarly Activity Among VA Podiatrists: A Cross-Sectional Study
Scholarly Activity Among VA Podiatrists: A Cross-Sectional Study