User login
For MD-IQ use only
Scurvy in Hospitalized Patients
Scurvy in Hospitalized Patients
Scurvy, caused by vitamin C or ascorbic acid deficiency, historically has been associated primarily with developing nations and famine; however, specific populations in industrialized nations remain at an increased risk, particularly individuals with a history of smoking, alcohol use, restrictive diet, poor oral intake, psychiatric disorders, dementia, bone marrow transplantation, gastroesophageal reflux disease, end-stage renal disease, and hospitalization.1 Micronutrient deficiency– associated dermatoses have been linked to poor clinical outcomes in hospitalized patients.2 In this case series, we report 4 hospitalized patients with scurvy, each presenting with unique comorbidities and risk factors for vitamin C deficiency (eTable).
Case Reports
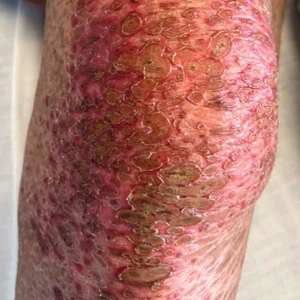
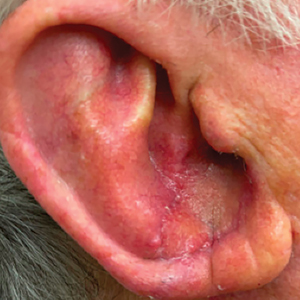
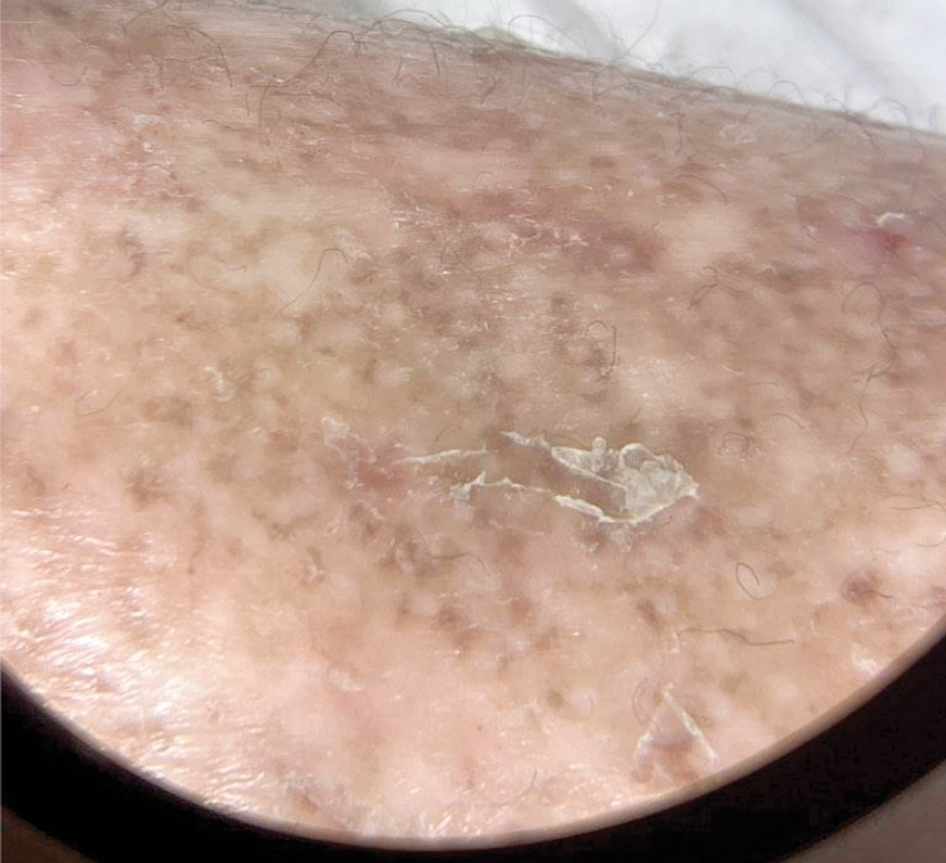
Patient 1—A 50-year-old man with a 6-month history of eczema and restrictive dietary intake was admitted to the hospital for septic shock attributed to a left foot infection of 5-days’ duration. The patient had experienced unintentional weight loss with severe protein-calorie malnutrition. His dietary history was notable for selective eating behaviors, intermittent meal skipping, and vegetarianism. Mucocutaneous examination by the dermatology consult team showed exfoliative dermatitis with angular cheilitis, corkscrew hairs on the legs (eFigure 1), and scattered purpura throughout the body. The differential diagnosis included eczema exacerbation, cutaneous T-cell lymphoma/Sézary syndrome, and malnutrition-related dermatosis. Punch biopsies of the left medial knee and right lateral arm revealed impetiginized, spongiotic, psoriasiform dermatitis with papillary dermal edema. The histologic changes were consistent with malnutrition-related dermatosis. Laboratory results included low vitamin C levels (0.1 mg/dL [reference range, 0.2-2.1 mg/dL]), undetectable zinc levels (<10 μg/dL [reference range ,60-130 μg/dL]), a low platelet count (21 kμ/L [reference range, 150-400 k/μL]),low albumin levels (0.9 mg/dL (13.0 g/dL [reference range, 14.0-17.4 g/dL]). The final diagnosis was exfoliative dermatitis due to eczema and multiple nutrient deficiencies (vitamin C and zinc). The patient was treated with vitamin C 500 mg/d and was started on mirtazapine to improve his appetite. Following a 3-month hospitalization, the patient was lost to follow-up after discharge.
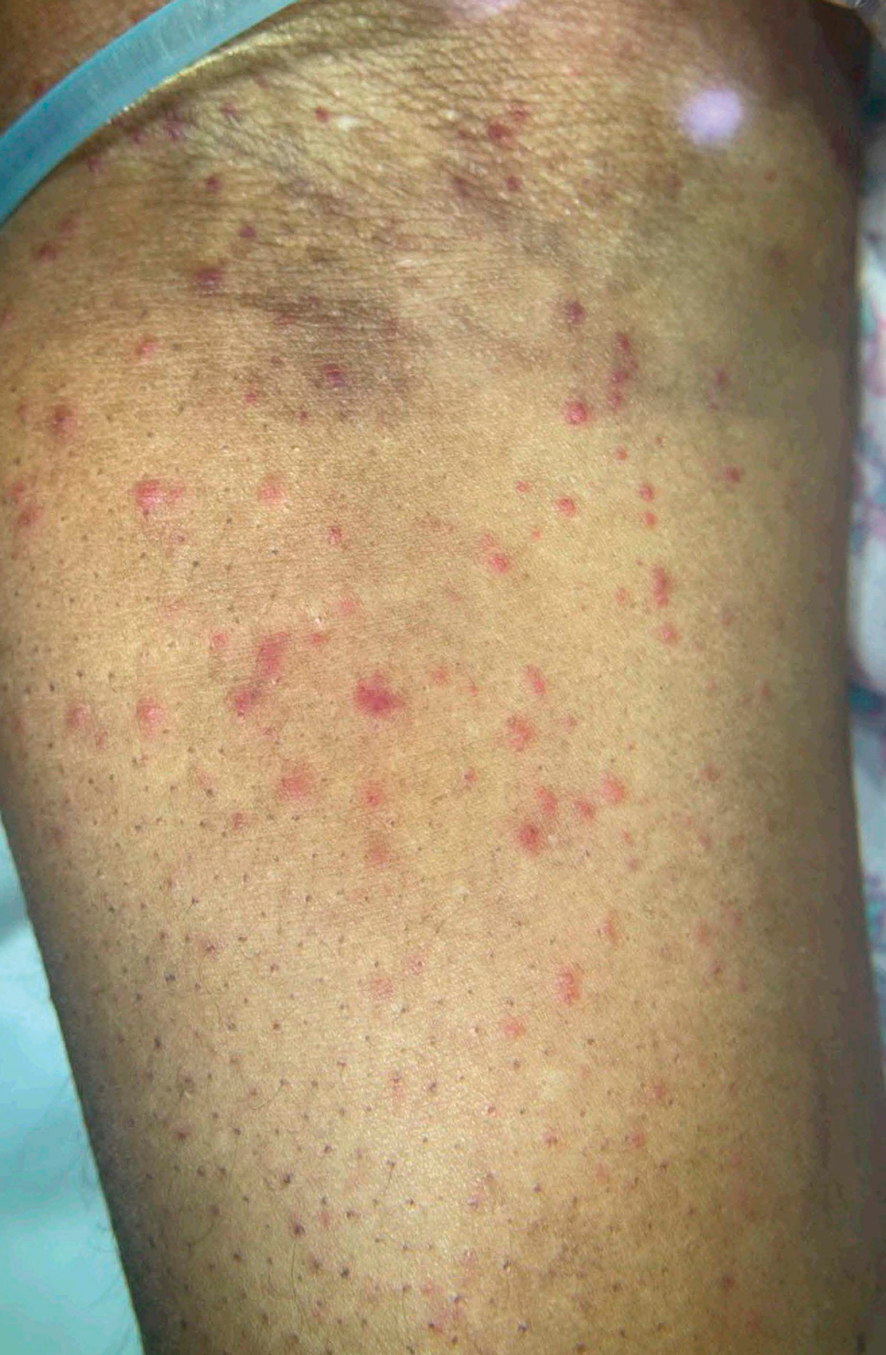
Patient 2—A 55-year-old woman with a history of multiple psychiatric disorders presented to the dermatology consult service with an asymptomatic purpuric eruption on the right antecubital fossa of 2 days’ duration that spread to the proximal thighs. Five days prior to presentation, she had received an allogeneic bone marrow transplant complicated by mucositis. She also reported a 4-month history of decreased appetite. At the current presentation, numerous acral, follicular based, purpuric macules and papules without associated corkscrew hairs were observed (eFigure 2). The differential diagnosis included a purpuric drug reaction, viral exanthem, acute graft-vs-host disease, neutrophilic dermatoses, and vitamin C deficiency–related dermatosis. Laboratory results revealed undetectable vitamin C levels (<0.1 mg/dL [reference range, 0.3-2.7 mg/dL]), a low platelet count (8 k/μL [reference range, 150-400 k/μL]), normal albumin levels (3.7 g/dL [reference range, 3.5-5.0 g/dL]), and low hemoglobin (7.8 g/dL [reference range, 14.0-17.4 g/dL]). Based on the histopathologic finding of subtle interface dermatitis with purpura from a punch biopsy of the right forearm, the eruption was attributed to scurvy. Although dermatology recommended supplementation with vitamin C 1000 mg/d, the decision was deferred by the primary team and the purpura improved without it—suggesting the purpura was only partly attributable to low vitamin C.
Patient 3—A 77-year-old woman with a history of low oral intake, a low body mass index (18.15 kg/m2 [reference range, 18.5-24.9]), vegetarianism, multiple psychiatric disorders, dementia, recent Clostridioides difficile colitis treated with meropenem, and recurrent idiopathic pancytopenia presented to the hospital with recurrent oral erosions and purpura of the legs for an unknown period. Physical examination by the dermatology consult team revealed superficial lip desquamation; erosions of the buccal mucosa with no involvement of the inner lip or gingiva; mild gingival hyperplasia (eFigure 3); and scaly, purpuric, follicular macules and papules on the legs. The arms and legs were devoid of hair. Laboratory results were notable for low vitamin C levels (0.1 mg/dL [reference range, 0.3-2.7 mg/dL]), a low platelet count (28 k/μL [reference range, 150-500 k/μL]), low albumin levels (2.9 g/dL [reference range, 3.5-5.0 g/dL]), and low hemoglobin (8.8 g/dL [reference range, 12.0-16.0 g/ dL]). A punch biopsy from the left thigh revealed pauci-inflammatory interface dermatitis with purpura. Based on the clinical and histologic findings, a final diagnosis of purpuric drug eruption (from the meropenem) and scurvy was made. Nutritional support included supplementation with vitamin C 1000 mg/d. The patient’s oral erosions and purpura gradually resolved with treatment throughout her 1.5-month hospitalization.
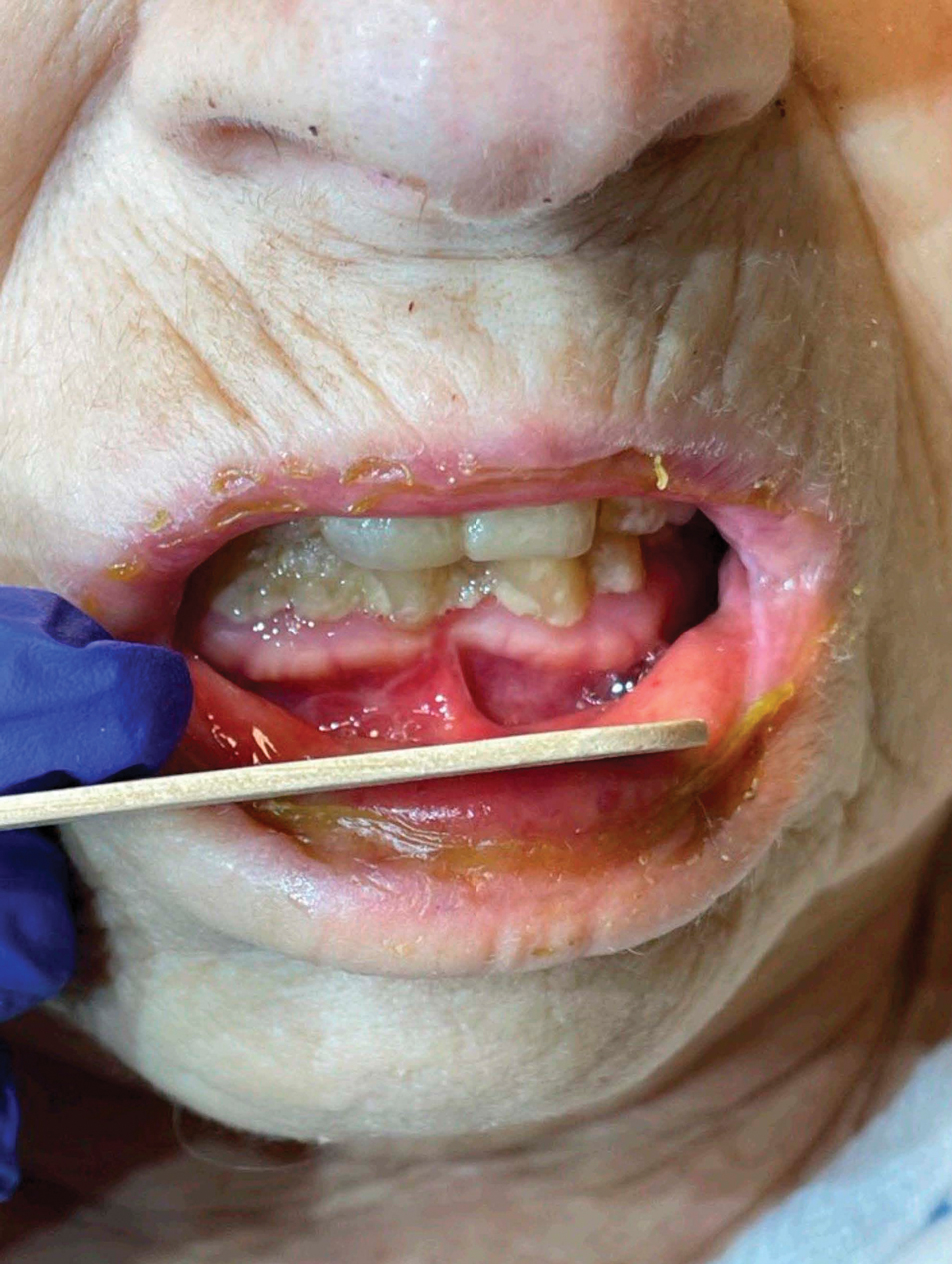
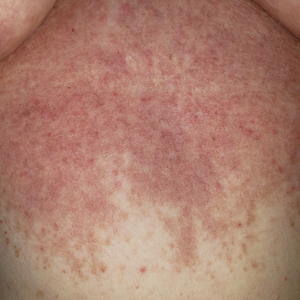
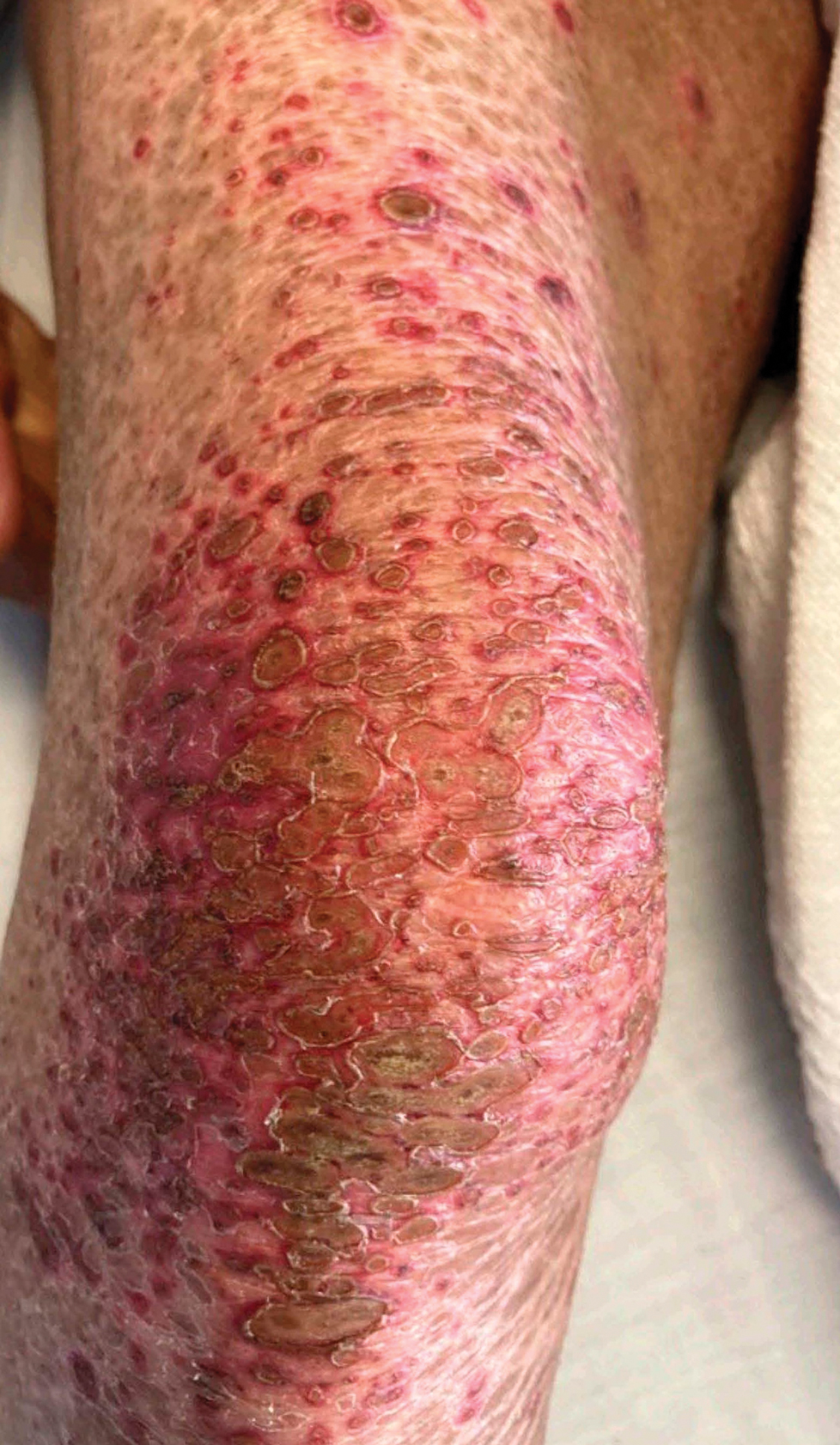
Patient 4—A 67-year-old woman with a history of extensive cardiovascular disease, gastroesophageal reflux disease without esophagitis, end-stage renal disease not requiring hemodialysis, and loss of appetite presented with a painful pruritic eruption on the legs with groin involvement of 2 months’ duration. The patient was admitted to the hospital for worsening mental status and weakness accompanied by dark stools, hematuria, and a productive cough with red-tinged sputum. Physical examination by the dermatology consult team showed a scaly, follicular, purpuric eruption affecting the acral and intertriginous sites (eFigure 4). The patient had sparse leg hair, making it difficult to assess for hair tortuosity. A punch biopsy of the left posterior knee revealed purpuric psoriasiform dermatitis, which was consistent with nutritional deficiency– associated dermatosis. Laboratory results included low vitamin C (<0.1 mg/dL [reference range, 0.3-2.7 mg/dL]), zinc, (58 μg/dL [reference range, 60-130 μg/dL]), and albumin levels (3.3 g/dL [reference range, 3.5-5.0 g/dL]) and a low platelet count (67 k/μL [reference range, 150- 500 k/μL]). The patient was started on supplementation with vitamin C 1000 mg/d with improvement of the purpura.
Comment
Micronutrient deficiencies may be common in hospitalized patients due to an increased prevalence of predisposing risk factors including infection, malnutrition, malabsorptive conditions, psychiatric diseases, and chronic illnesses.3 Acute-phase response in hospitalized patients also has been strongly associated with decreased plasma vitamin C levels.4 This phenomenon is postulated to be due to the increase in ascorbic acid uptake by circulating granulocytes in acute disease5; however because low vitamin C levels during the acute-phase response may not always accurately reflect total body stores, other clinical features should be assessed. Previously reported social history risk factors include smoking, alcohol consumption, marijuana use, restrictive diets, vegetarianism, and living alone.6,7
The unifying clinical clues for scurvy in our 4 patients were a history of poor oral intake and purpura. While purpura is nonspecific and can appear after traumatic injury to the skin in elderly patients with photodamage and coagulation disorders, it also is associated with vitamin C deficiency, even with a normal platelet count, circulating von Willebrand factor levels, and prothrombin time/partial thromboplastin time.8 This is because vitamin C is vital in forming the collagen and extracellular matrix. Specifically, it is a cofactor for lysine and proline hydroxylase enzymes needed for the á-helix crosslinks in collagen, which are essential for its structural integrity.9 Collagen is a structural protein that maintains the blood vessel walls, skin, and the basement membrane. A deficiency in vitamin C leads to impairment in collagen synthesis, and insufficient collagen results in compromised connective tissue, blood vessels, and hair strength, which may lead to purpura. All of our patients had thrombocytopenia, and similarly, consideration for scurvy in hospitalized patients with risk factors for micronutrient deficiency is a must. Additional findings such as a follicular-based pattern of the purpura, hair tortuosity, restrictive dietary history, histopathology reports consistent with nutritional dermatoses, serum vitamin C levels, and improvement with vitamin C supplementation are more specific for scurvy. All of these factors can assist the clinician in detecting and confirming these micronutrient deficiencies.
Although there are no established therapeutic guidelines for scurvy, the mainstay of treatment is vitamin C repletion, either orally or parenterally. In hospitalized patients, one suggested regimen is 1000 mg of intravenous ascorbic acid daily for 3 days, followed by further supplementation with a dose of 250 to 500 mg twice daily for 1 month as needed after discharge.10 Symptom improvement occurs about 72 hours after vitamin replacement.8 We recommended 500 to 1000 mg of daily vitamin C supplementation for our patients.
Final Thoughts
This case series highlights the importance of maintaining a high index of suspicion for scurvy in hospitalized patients presenting with purpura, especially in a follicular-based pattern, who have multiple medical comorbidities and risk factors for vitamin C deficiency. The manifestations of scurvy are heterogeneous, necessitating a comprehensive mucocutaneous examination. The diagnosis of scurvy requires correlation of the findings from the patient history, clinical examination, laboratory results, and histopathology.
- Hirschmann JV, Raugi GJ. Adult scurvy. J Am Acad Dermatol. 1999; 41:895-910.
- Marsh RL, Trinidad J, Shearer S, et al. Association between micronutrient deficiency dermatoses and clinical outcomes in hospitalized patients. J Am Acad Dermatol. 2020;82:1226-1228.
- Hoffman M, Micheletti RG, Shields BE. Nutritional dermatoses in the hospitalized patient. Cutis. 2020;105:296-302, 308, E1-E5.
- Fain O, Pariés J, Jacquart B, et al. Hypovitaminosis C in hospitalized patients. Eur J Intern Med. 2003;14:419-425.
- Moser U, Weber F. Uptake of ascorbic acid by human granulocytes. Int J Vitam Nutr Res. 1984;54:47-53.
- Swanson AM, Hughey LC. Acute inpatient presentation of scurvy. Cutis. 2010;86:205-207.
- Christopher KL, Menachof KK, Fathi R. Scurvy masquerading as reactive arthritis. Cutis. 2019;103:E21-E23.
- Antonelli M, Burzo ML, Pecorini G, et al. Scurvy as cause of purpura in the XXI century: a review on this “ancient” disease. Eur Rev Med Pharmacol Sci. 2018;22:4355-4358.
- Maxfield L, Daley SF, Crane JS. Vitamin C deficiency. StatPearls [Internet]. Updated November 12, 2023. Accessed September 6, 2024. https://www.ncbi.nlm.nih.gov/books/NBK493187/
- Gandhi M, Elfeky O, Ertugrul H, et al. Scurvy: rediscovering a forgotten disease. Diseases. 2023;11:78.
Scurvy, caused by vitamin C or ascorbic acid deficiency, historically has been associated primarily with developing nations and famine; however, specific populations in industrialized nations remain at an increased risk, particularly individuals with a history of smoking, alcohol use, restrictive diet, poor oral intake, psychiatric disorders, dementia, bone marrow transplantation, gastroesophageal reflux disease, end-stage renal disease, and hospitalization.1 Micronutrient deficiency– associated dermatoses have been linked to poor clinical outcomes in hospitalized patients.2 In this case series, we report 4 hospitalized patients with scurvy, each presenting with unique comorbidities and risk factors for vitamin C deficiency (eTable).

Case Reports
Patient 1—A 50-year-old man with a 6-month history of eczema and restrictive dietary intake was admitted to the hospital for septic shock attributed to a left foot infection of 5-days’ duration. The patient had experienced unintentional weight loss with severe protein-calorie malnutrition. His dietary history was notable for selective eating behaviors, intermittent meal skipping, and vegetarianism. Mucocutaneous examination by the dermatology consult team showed exfoliative dermatitis with angular cheilitis, corkscrew hairs on the legs (eFigure 1), and scattered purpura throughout the body. The differential diagnosis included eczema exacerbation, cutaneous T-cell lymphoma/Sézary syndrome, and malnutrition-related dermatosis. Punch biopsies of the left medial knee and right lateral arm revealed impetiginized, spongiotic, psoriasiform dermatitis with papillary dermal edema. The histologic changes were consistent with malnutrition-related dermatosis. Laboratory results included low vitamin C levels (0.1 mg/dL [reference range, 0.2-2.1 mg/dL]), undetectable zinc levels (<10 μg/dL [reference range ,60-130 μg/dL]), a low platelet count (21 kμ/L [reference range, 150-400 k/μL]),low albumin levels (0.9 mg/dL (13.0 g/dL [reference range, 14.0-17.4 g/dL]). The final diagnosis was exfoliative dermatitis due to eczema and multiple nutrient deficiencies (vitamin C and zinc). The patient was treated with vitamin C 500 mg/d and was started on mirtazapine to improve his appetite. Following a 3-month hospitalization, the patient was lost to follow-up after discharge.

Patient 2—A 55-year-old woman with a history of multiple psychiatric disorders presented to the dermatology consult service with an asymptomatic purpuric eruption on the right antecubital fossa of 2 days’ duration that spread to the proximal thighs. Five days prior to presentation, she had received an allogeneic bone marrow transplant complicated by mucositis. She also reported a 4-month history of decreased appetite. At the current presentation, numerous acral, follicular based, purpuric macules and papules without associated corkscrew hairs were observed (eFigure 2). The differential diagnosis included a purpuric drug reaction, viral exanthem, acute graft-vs-host disease, neutrophilic dermatoses, and vitamin C deficiency–related dermatosis. Laboratory results revealed undetectable vitamin C levels (<0.1 mg/dL [reference range, 0.3-2.7 mg/dL]), a low platelet count (8 k/μL [reference range, 150-400 k/μL]), normal albumin levels (3.7 g/dL [reference range, 3.5-5.0 g/dL]), and low hemoglobin (7.8 g/dL [reference range, 14.0-17.4 g/dL]). Based on the histopathologic finding of subtle interface dermatitis with purpura from a punch biopsy of the right forearm, the eruption was attributed to scurvy. Although dermatology recommended supplementation with vitamin C 1000 mg/d, the decision was deferred by the primary team and the purpura improved without it—suggesting the purpura was only partly attributable to low vitamin C.

Patient 3—A 77-year-old woman with a history of low oral intake, a low body mass index (18.15 kg/m2 [reference range, 18.5-24.9]), vegetarianism, multiple psychiatric disorders, dementia, recent Clostridioides difficile colitis treated with meropenem, and recurrent idiopathic pancytopenia presented to the hospital with recurrent oral erosions and purpura of the legs for an unknown period. Physical examination by the dermatology consult team revealed superficial lip desquamation; erosions of the buccal mucosa with no involvement of the inner lip or gingiva; mild gingival hyperplasia (eFigure 3); and scaly, purpuric, follicular macules and papules on the legs. The arms and legs were devoid of hair. Laboratory results were notable for low vitamin C levels (0.1 mg/dL [reference range, 0.3-2.7 mg/dL]), a low platelet count (28 k/μL [reference range, 150-500 k/μL]), low albumin levels (2.9 g/dL [reference range, 3.5-5.0 g/dL]), and low hemoglobin (8.8 g/dL [reference range, 12.0-16.0 g/ dL]). A punch biopsy from the left thigh revealed pauci-inflammatory interface dermatitis with purpura. Based on the clinical and histologic findings, a final diagnosis of purpuric drug eruption (from the meropenem) and scurvy was made. Nutritional support included supplementation with vitamin C 1000 mg/d. The patient’s oral erosions and purpura gradually resolved with treatment throughout her 1.5-month hospitalization.

Patient 4—A 67-year-old woman with a history of extensive cardiovascular disease, gastroesophageal reflux disease without esophagitis, end-stage renal disease not requiring hemodialysis, and loss of appetite presented with a painful pruritic eruption on the legs with groin involvement of 2 months’ duration. The patient was admitted to the hospital for worsening mental status and weakness accompanied by dark stools, hematuria, and a productive cough with red-tinged sputum. Physical examination by the dermatology consult team showed a scaly, follicular, purpuric eruption affecting the acral and intertriginous sites (eFigure 4). The patient had sparse leg hair, making it difficult to assess for hair tortuosity. A punch biopsy of the left posterior knee revealed purpuric psoriasiform dermatitis, which was consistent with nutritional deficiency– associated dermatosis. Laboratory results included low vitamin C (<0.1 mg/dL [reference range, 0.3-2.7 mg/dL]), zinc, (58 μg/dL [reference range, 60-130 μg/dL]), and albumin levels (3.3 g/dL [reference range, 3.5-5.0 g/dL]) and a low platelet count (67 k/μL [reference range, 150- 500 k/μL]). The patient was started on supplementation with vitamin C 1000 mg/d with improvement of the purpura.

Comment
Micronutrient deficiencies may be common in hospitalized patients due to an increased prevalence of predisposing risk factors including infection, malnutrition, malabsorptive conditions, psychiatric diseases, and chronic illnesses.3 Acute-phase response in hospitalized patients also has been strongly associated with decreased plasma vitamin C levels.4 This phenomenon is postulated to be due to the increase in ascorbic acid uptake by circulating granulocytes in acute disease5; however because low vitamin C levels during the acute-phase response may not always accurately reflect total body stores, other clinical features should be assessed. Previously reported social history risk factors include smoking, alcohol consumption, marijuana use, restrictive diets, vegetarianism, and living alone.6,7
The unifying clinical clues for scurvy in our 4 patients were a history of poor oral intake and purpura. While purpura is nonspecific and can appear after traumatic injury to the skin in elderly patients with photodamage and coagulation disorders, it also is associated with vitamin C deficiency, even with a normal platelet count, circulating von Willebrand factor levels, and prothrombin time/partial thromboplastin time.8 This is because vitamin C is vital in forming the collagen and extracellular matrix. Specifically, it is a cofactor for lysine and proline hydroxylase enzymes needed for the á-helix crosslinks in collagen, which are essential for its structural integrity.9 Collagen is a structural protein that maintains the blood vessel walls, skin, and the basement membrane. A deficiency in vitamin C leads to impairment in collagen synthesis, and insufficient collagen results in compromised connective tissue, blood vessels, and hair strength, which may lead to purpura. All of our patients had thrombocytopenia, and similarly, consideration for scurvy in hospitalized patients with risk factors for micronutrient deficiency is a must. Additional findings such as a follicular-based pattern of the purpura, hair tortuosity, restrictive dietary history, histopathology reports consistent with nutritional dermatoses, serum vitamin C levels, and improvement with vitamin C supplementation are more specific for scurvy. All of these factors can assist the clinician in detecting and confirming these micronutrient deficiencies.
Although there are no established therapeutic guidelines for scurvy, the mainstay of treatment is vitamin C repletion, either orally or parenterally. In hospitalized patients, one suggested regimen is 1000 mg of intravenous ascorbic acid daily for 3 days, followed by further supplementation with a dose of 250 to 500 mg twice daily for 1 month as needed after discharge.10 Symptom improvement occurs about 72 hours after vitamin replacement.8 We recommended 500 to 1000 mg of daily vitamin C supplementation for our patients.
Final Thoughts
This case series highlights the importance of maintaining a high index of suspicion for scurvy in hospitalized patients presenting with purpura, especially in a follicular-based pattern, who have multiple medical comorbidities and risk factors for vitamin C deficiency. The manifestations of scurvy are heterogeneous, necessitating a comprehensive mucocutaneous examination. The diagnosis of scurvy requires correlation of the findings from the patient history, clinical examination, laboratory results, and histopathology.
Scurvy, caused by vitamin C or ascorbic acid deficiency, historically has been associated primarily with developing nations and famine; however, specific populations in industrialized nations remain at an increased risk, particularly individuals with a history of smoking, alcohol use, restrictive diet, poor oral intake, psychiatric disorders, dementia, bone marrow transplantation, gastroesophageal reflux disease, end-stage renal disease, and hospitalization.1 Micronutrient deficiency– associated dermatoses have been linked to poor clinical outcomes in hospitalized patients.2 In this case series, we report 4 hospitalized patients with scurvy, each presenting with unique comorbidities and risk factors for vitamin C deficiency (eTable).

Case Reports
Patient 1—A 50-year-old man with a 6-month history of eczema and restrictive dietary intake was admitted to the hospital for septic shock attributed to a left foot infection of 5-days’ duration. The patient had experienced unintentional weight loss with severe protein-calorie malnutrition. His dietary history was notable for selective eating behaviors, intermittent meal skipping, and vegetarianism. Mucocutaneous examination by the dermatology consult team showed exfoliative dermatitis with angular cheilitis, corkscrew hairs on the legs (eFigure 1), and scattered purpura throughout the body. The differential diagnosis included eczema exacerbation, cutaneous T-cell lymphoma/Sézary syndrome, and malnutrition-related dermatosis. Punch biopsies of the left medial knee and right lateral arm revealed impetiginized, spongiotic, psoriasiform dermatitis with papillary dermal edema. The histologic changes were consistent with malnutrition-related dermatosis. Laboratory results included low vitamin C levels (0.1 mg/dL [reference range, 0.2-2.1 mg/dL]), undetectable zinc levels (<10 μg/dL [reference range ,60-130 μg/dL]), a low platelet count (21 kμ/L [reference range, 150-400 k/μL]),low albumin levels (0.9 mg/dL (13.0 g/dL [reference range, 14.0-17.4 g/dL]). The final diagnosis was exfoliative dermatitis due to eczema and multiple nutrient deficiencies (vitamin C and zinc). The patient was treated with vitamin C 500 mg/d and was started on mirtazapine to improve his appetite. Following a 3-month hospitalization, the patient was lost to follow-up after discharge.

Patient 2—A 55-year-old woman with a history of multiple psychiatric disorders presented to the dermatology consult service with an asymptomatic purpuric eruption on the right antecubital fossa of 2 days’ duration that spread to the proximal thighs. Five days prior to presentation, she had received an allogeneic bone marrow transplant complicated by mucositis. She also reported a 4-month history of decreased appetite. At the current presentation, numerous acral, follicular based, purpuric macules and papules without associated corkscrew hairs were observed (eFigure 2). The differential diagnosis included a purpuric drug reaction, viral exanthem, acute graft-vs-host disease, neutrophilic dermatoses, and vitamin C deficiency–related dermatosis. Laboratory results revealed undetectable vitamin C levels (<0.1 mg/dL [reference range, 0.3-2.7 mg/dL]), a low platelet count (8 k/μL [reference range, 150-400 k/μL]), normal albumin levels (3.7 g/dL [reference range, 3.5-5.0 g/dL]), and low hemoglobin (7.8 g/dL [reference range, 14.0-17.4 g/dL]). Based on the histopathologic finding of subtle interface dermatitis with purpura from a punch biopsy of the right forearm, the eruption was attributed to scurvy. Although dermatology recommended supplementation with vitamin C 1000 mg/d, the decision was deferred by the primary team and the purpura improved without it—suggesting the purpura was only partly attributable to low vitamin C.

Patient 3—A 77-year-old woman with a history of low oral intake, a low body mass index (18.15 kg/m2 [reference range, 18.5-24.9]), vegetarianism, multiple psychiatric disorders, dementia, recent Clostridioides difficile colitis treated with meropenem, and recurrent idiopathic pancytopenia presented to the hospital with recurrent oral erosions and purpura of the legs for an unknown period. Physical examination by the dermatology consult team revealed superficial lip desquamation; erosions of the buccal mucosa with no involvement of the inner lip or gingiva; mild gingival hyperplasia (eFigure 3); and scaly, purpuric, follicular macules and papules on the legs. The arms and legs were devoid of hair. Laboratory results were notable for low vitamin C levels (0.1 mg/dL [reference range, 0.3-2.7 mg/dL]), a low platelet count (28 k/μL [reference range, 150-500 k/μL]), low albumin levels (2.9 g/dL [reference range, 3.5-5.0 g/dL]), and low hemoglobin (8.8 g/dL [reference range, 12.0-16.0 g/ dL]). A punch biopsy from the left thigh revealed pauci-inflammatory interface dermatitis with purpura. Based on the clinical and histologic findings, a final diagnosis of purpuric drug eruption (from the meropenem) and scurvy was made. Nutritional support included supplementation with vitamin C 1000 mg/d. The patient’s oral erosions and purpura gradually resolved with treatment throughout her 1.5-month hospitalization.

Patient 4—A 67-year-old woman with a history of extensive cardiovascular disease, gastroesophageal reflux disease without esophagitis, end-stage renal disease not requiring hemodialysis, and loss of appetite presented with a painful pruritic eruption on the legs with groin involvement of 2 months’ duration. The patient was admitted to the hospital for worsening mental status and weakness accompanied by dark stools, hematuria, and a productive cough with red-tinged sputum. Physical examination by the dermatology consult team showed a scaly, follicular, purpuric eruption affecting the acral and intertriginous sites (eFigure 4). The patient had sparse leg hair, making it difficult to assess for hair tortuosity. A punch biopsy of the left posterior knee revealed purpuric psoriasiform dermatitis, which was consistent with nutritional deficiency– associated dermatosis. Laboratory results included low vitamin C (<0.1 mg/dL [reference range, 0.3-2.7 mg/dL]), zinc, (58 μg/dL [reference range, 60-130 μg/dL]), and albumin levels (3.3 g/dL [reference range, 3.5-5.0 g/dL]) and a low platelet count (67 k/μL [reference range, 150- 500 k/μL]). The patient was started on supplementation with vitamin C 1000 mg/d with improvement of the purpura.

Comment
Micronutrient deficiencies may be common in hospitalized patients due to an increased prevalence of predisposing risk factors including infection, malnutrition, malabsorptive conditions, psychiatric diseases, and chronic illnesses.3 Acute-phase response in hospitalized patients also has been strongly associated with decreased plasma vitamin C levels.4 This phenomenon is postulated to be due to the increase in ascorbic acid uptake by circulating granulocytes in acute disease5; however because low vitamin C levels during the acute-phase response may not always accurately reflect total body stores, other clinical features should be assessed. Previously reported social history risk factors include smoking, alcohol consumption, marijuana use, restrictive diets, vegetarianism, and living alone.6,7
The unifying clinical clues for scurvy in our 4 patients were a history of poor oral intake and purpura. While purpura is nonspecific and can appear after traumatic injury to the skin in elderly patients with photodamage and coagulation disorders, it also is associated with vitamin C deficiency, even with a normal platelet count, circulating von Willebrand factor levels, and prothrombin time/partial thromboplastin time.8 This is because vitamin C is vital in forming the collagen and extracellular matrix. Specifically, it is a cofactor for lysine and proline hydroxylase enzymes needed for the á-helix crosslinks in collagen, which are essential for its structural integrity.9 Collagen is a structural protein that maintains the blood vessel walls, skin, and the basement membrane. A deficiency in vitamin C leads to impairment in collagen synthesis, and insufficient collagen results in compromised connective tissue, blood vessels, and hair strength, which may lead to purpura. All of our patients had thrombocytopenia, and similarly, consideration for scurvy in hospitalized patients with risk factors for micronutrient deficiency is a must. Additional findings such as a follicular-based pattern of the purpura, hair tortuosity, restrictive dietary history, histopathology reports consistent with nutritional dermatoses, serum vitamin C levels, and improvement with vitamin C supplementation are more specific for scurvy. All of these factors can assist the clinician in detecting and confirming these micronutrient deficiencies.
Although there are no established therapeutic guidelines for scurvy, the mainstay of treatment is vitamin C repletion, either orally or parenterally. In hospitalized patients, one suggested regimen is 1000 mg of intravenous ascorbic acid daily for 3 days, followed by further supplementation with a dose of 250 to 500 mg twice daily for 1 month as needed after discharge.10 Symptom improvement occurs about 72 hours after vitamin replacement.8 We recommended 500 to 1000 mg of daily vitamin C supplementation for our patients.
Final Thoughts
This case series highlights the importance of maintaining a high index of suspicion for scurvy in hospitalized patients presenting with purpura, especially in a follicular-based pattern, who have multiple medical comorbidities and risk factors for vitamin C deficiency. The manifestations of scurvy are heterogeneous, necessitating a comprehensive mucocutaneous examination. The diagnosis of scurvy requires correlation of the findings from the patient history, clinical examination, laboratory results, and histopathology.
- Hirschmann JV, Raugi GJ. Adult scurvy. J Am Acad Dermatol. 1999; 41:895-910.
- Marsh RL, Trinidad J, Shearer S, et al. Association between micronutrient deficiency dermatoses and clinical outcomes in hospitalized patients. J Am Acad Dermatol. 2020;82:1226-1228.
- Hoffman M, Micheletti RG, Shields BE. Nutritional dermatoses in the hospitalized patient. Cutis. 2020;105:296-302, 308, E1-E5.
- Fain O, Pariés J, Jacquart B, et al. Hypovitaminosis C in hospitalized patients. Eur J Intern Med. 2003;14:419-425.
- Moser U, Weber F. Uptake of ascorbic acid by human granulocytes. Int J Vitam Nutr Res. 1984;54:47-53.
- Swanson AM, Hughey LC. Acute inpatient presentation of scurvy. Cutis. 2010;86:205-207.
- Christopher KL, Menachof KK, Fathi R. Scurvy masquerading as reactive arthritis. Cutis. 2019;103:E21-E23.
- Antonelli M, Burzo ML, Pecorini G, et al. Scurvy as cause of purpura in the XXI century: a review on this “ancient” disease. Eur Rev Med Pharmacol Sci. 2018;22:4355-4358.
- Maxfield L, Daley SF, Crane JS. Vitamin C deficiency. StatPearls [Internet]. Updated November 12, 2023. Accessed September 6, 2024. https://www.ncbi.nlm.nih.gov/books/NBK493187/
- Gandhi M, Elfeky O, Ertugrul H, et al. Scurvy: rediscovering a forgotten disease. Diseases. 2023;11:78.
- Hirschmann JV, Raugi GJ. Adult scurvy. J Am Acad Dermatol. 1999; 41:895-910.
- Marsh RL, Trinidad J, Shearer S, et al. Association between micronutrient deficiency dermatoses and clinical outcomes in hospitalized patients. J Am Acad Dermatol. 2020;82:1226-1228.
- Hoffman M, Micheletti RG, Shields BE. Nutritional dermatoses in the hospitalized patient. Cutis. 2020;105:296-302, 308, E1-E5.
- Fain O, Pariés J, Jacquart B, et al. Hypovitaminosis C in hospitalized patients. Eur J Intern Med. 2003;14:419-425.
- Moser U, Weber F. Uptake of ascorbic acid by human granulocytes. Int J Vitam Nutr Res. 1984;54:47-53.
- Swanson AM, Hughey LC. Acute inpatient presentation of scurvy. Cutis. 2010;86:205-207.
- Christopher KL, Menachof KK, Fathi R. Scurvy masquerading as reactive arthritis. Cutis. 2019;103:E21-E23.
- Antonelli M, Burzo ML, Pecorini G, et al. Scurvy as cause of purpura in the XXI century: a review on this “ancient” disease. Eur Rev Med Pharmacol Sci. 2018;22:4355-4358.
- Maxfield L, Daley SF, Crane JS. Vitamin C deficiency. StatPearls [Internet]. Updated November 12, 2023. Accessed September 6, 2024. https://www.ncbi.nlm.nih.gov/books/NBK493187/
- Gandhi M, Elfeky O, Ertugrul H, et al. Scurvy: rediscovering a forgotten disease. Diseases. 2023;11:78.
Scurvy in Hospitalized Patients
Scurvy in Hospitalized Patients
PRACTICE POINTS
- Clinicians should maintain a high index of suspicion for vitamin C deficiency/scurvy in hospitalized patients with purpura who have multiple medical comorbidities and risk factors.
- A low platelet count may mask underlying vitamin C deficiency, and patients may have concurrent deficiencies in other nutrients such as zinc.
Comparing the Quality of Patient Guidance on Dermatologic Care Generated by ChatGPT vs Reddit
Comparing the Quality of Patient Guidance on Dermatologic Care Generated by ChatGPT vs Reddit
To the Editor:
Online resources that are convenient and affordable play a crucial role in mitigating health inequality and improving patient access to health care information; however, the benefits are limited by the quality of information available, as medical misinformation can lead to patients engaging in harmful practices, making dangerous decisions, and even avoiding safe and effective treatments. In this study, we aimed to assess and compare the quality of patient guidance on dermatologic care generated by ChatGPT vs Reddit based on accuracy, appropriateness, and safety. It is essential to assess the quality and reliability of online health information to support patients in making informed decisions about their health.
The emergence and advancement of artificial intelligence and large language models such as ChatGPT present a new method for patients to access health care advice. ChatGPT can engage in conversation by accessing information from existing publicly available data on the internet, including books and websites, up to the year 2023 and providing humanlike responses with context.1 ChatGPT’s access to a breadth of online evidence-based literature ensures the dissemination of quality information that is quick and without inherent bias, offering the potential to more closely align with health care professionals. ChatGPT’s use in dermatology by patients has shown efficacy, with a 98.87% approval rate by dermatologists scoring its ability to recommend appropriate medication for common dermatologic conditions.2 However, ChatGPT has limitations when providing health care advice and has been observed to misunderstand health care standards, lack personalization, and offer incorrect references; currently, the latest publicly available version (ChatGPT 3.5) also is unable to analyze clinical images.3,4
Reddit is an online social media forum that allows users to post questions and photographs to which anyone can reply and offer advice. Patients may find comfort in online communities where they can connect with others facing similar challenges related to their diagnosis. Within these communities, the responses often share users’ own lived experiences and offer support based on what has and has not worked for them. Prior research found that users intentionally seeking health information via Reddit are likely to implement the advice they receive even without verification of its credibility, suggesting a trust and receptibility to ideas offered on the platform.5 Furthermore, a study analyzing the dermatologic content of 17 dermatology related subreddits that had 1000 or more subscribers found that 70.6% of posts fell under the category of “seeking health/cosmetic advice.”6 Reddit users thus are vulnerable to receiving advice based on personal bias and exposing their health information to the public.
We hypothesized that ChatGPT would provide users with guidance that was more closely aligned with typical dermatologists’ advice due to its thorough analysis and compilation of diverse sources and recommendations available on the internet. We expected Reddit to yield recommendations of lesser quality and a diminished safety score, primarily due to the absence of credibility-vetting mechanisms and the influence of personal biases within the advice shared.
User-submitted posts to large dermatologic community Reddit forums representing a few of the most common skin conditions (r/eczema, r/acne, r/Folliculitis, r/SebDerm, r/Hidradenitis, r/keratosis, and r/Psoriasis) were retrospectively reviewed from January 2024 to March 2024. The most popular posts that did not include photographs were included in our study. Posts with photographs were excluded, as clinical images were not able to be uploaded to the publicly available ChatGPT 3.5. We collected real user questions about common skin conditions from Reddit forums and then asked ChatGPT to answer those same questions. We compared ChatGPT’s responses to the most upvoted Reddit comments to see how they matched up (eTable).

Each ChatGPT response and the top-rated Reddit comment were independently evaluated by a board certified dermatologist (S.A.) and a dermatology resident (A.H.K.). The quality of the ChatGPT and Reddit responses were determined by scoring the accuracy, appropriateness, safety consideration, and specificity on a 5-point Likert scale (1=low, 5=high). The 2 evaluators’ mean scores for each of the 4 categories were calculated based on adequate interrater reliability, which was tested using Cohen’s κ coefficient. Related-samples sign tests were used to compare ChatGPT and Reddit responses for each of the 4 categories. Analysis was completed using SPSS statistics software version 29.0 (IBM). The evaluators also were asked to provide qualitative feedback on the strengths and weaknesses of each response.
Our retrospective review yielded 20 total questions: 5 (25%) on atopic dermatitis, 4 (20%) on acne, 4 (20%) on hidradenitis suppurativa, 4 (20%) on psoriasis, 1 (5%) on folliculitis, 1 (5%) on keratosis pilaris, and 1 (5%) on seborrheic dermatitis. The number of posts was limited to 20 due to the extensive time required for grading each response. These 20 questions were selected from a larger pool of eligible posts based on factors such as clarity and relevance to common skin conditions. With regard to the types of questions that were asked, 6 (30%) were related to general management of a diagnosis, 5 (25%) were on treatment recommendations for symptom relief, 3 (15%) were on optimal utilization of current treatment regimens, 2 (10%) were on prescription side effects, 2 (10%) were on diagnosis presentation, 1 (5%) was on potential triggers of the diagnosis, and 1 (5%) was on natural treatment recommendations.
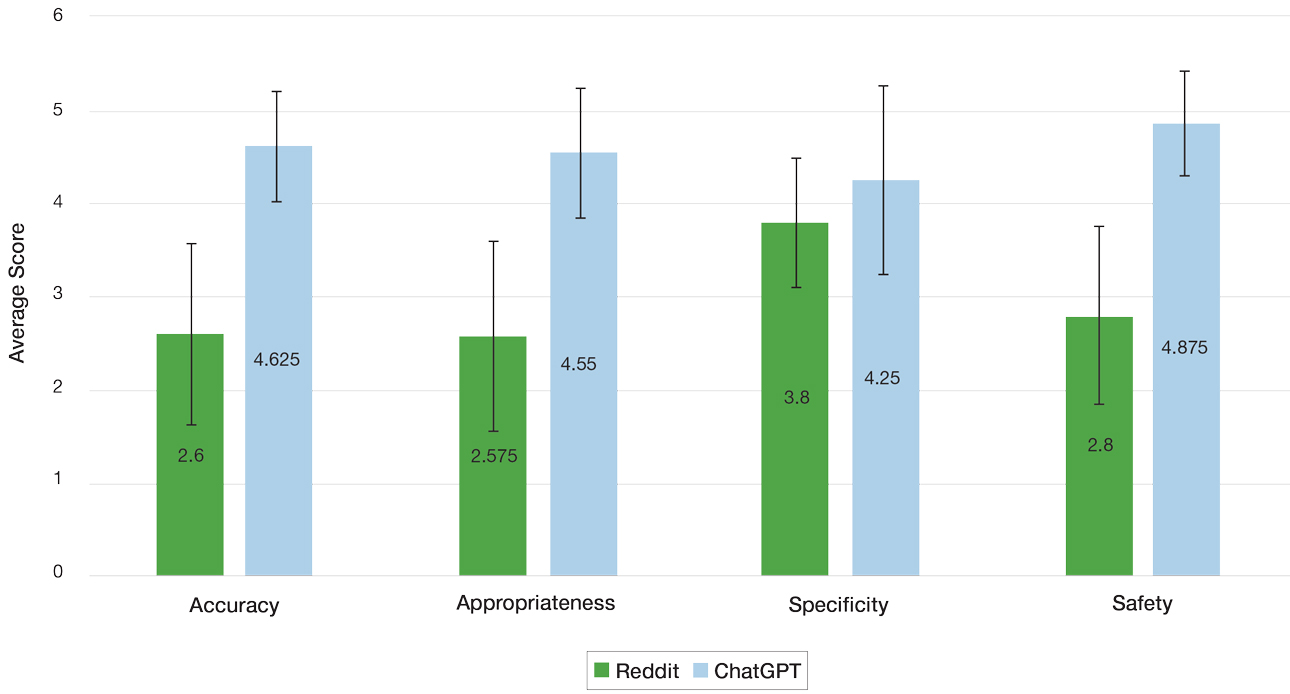
Mean (SD) evaluator scores for accuracy were significantly higher among ChatGPT responses compared with Reddit (4.63 [0.60] vs 2.60 [0.98])(P<.001). ChatGPT responses also were significantly higher for appropriateness compared with Reddit (4.55 [0.71] vs 2.58 [1.02])(P<.001) and safety consideration (4.88 [0.56] vs 2.80[0.97])(P <.001). There was no significant difference in mean specificity scores between ChatGPT and Reddit (4.25[1.02] vs 3.80 [0.70])(P=.096)(Figure).
For the Reddit responses, the weighted Cohen’s κ coefficient between the 2 evaluators was 0.200 (95% CI, –.089 to .489) for accuracy, 0.255 (95% CI, .014-.497) for appropriateness, 0.385 (95% CI, .176-.594) for safety consideration, and –0.024 (95% CI, –.177 to .129) for specificity. For the ChatGPT responses, the weighted Cohen’s κ coefficient between the 2 evaluators was 0.426 (95% CI, .122-.730) for accuracy, 0.571 (95% CI, .294-.849) for appropriateness, 0.655 (95% CI, .632-.678) for safety consideration, and 0.313 (95% CI, .043-.584) for specificity.
The strengths and weaknesses of the responses also were qualitatively analyzed. One commonly observed strength was ChatGPT’s frequent and appropriate recommendation for users to consult a dermatologist. In the case of atopic dermatitis—one of the more frequently asked about conditions—ChatGPT consistently emphasized evidence-based strategies such as gentle skin care and moisturization, reflecting alignment with clinical guidelines. Additionally, a common weakness of both ChatGPT and Reddit responses generally was the lack of personalized guidance and comprehensive discussion of the risks and benefits of specific treatments. It also was noted that neither platform consistently explored differential diagnoses—for example, distinguishing atopic dermatitis from conditions such as allergic contact dermatitis—limiting the diagnostic depth of the responses.
ChatGPT and Reddit can provide patients with quick and accessible health information for various dermatologic concerns. The results of our study demonstrated a significantly higher level of accuracy, appropriateness, and safety of responses generated by ChatGPT compared with human-generated responses on Reddit (P<.001). Both platforms offered similarly specific responses to user inquiries, demonstrating ChatGPT’s ability to comprehend user questions and draw from publicly available texts and Reddit users’ contributing insights based on their own first-hand experiences.
Reddit’s dermatologic forums often feature personal anecdotes and unique treatments described by individual users. Although specific to particular dermatologic concerns, such advice lacks an evidence-based standard of care. With the noted inherent trust of patients seeking guidance within Reddit communities, patients may follow unhelpful or potentially dangerous medical advice.5 A study examining 300 user-submitted posts on popular Reddit dermatology forums during the COVID-19 pandemic found that the mean scores for top-rated comments’ potential to be misleading or dangerous was 2.33 out of 5 on a Likert scale (95% CI, 2.18- 2.48).7 Dermatologists should be aware of the potential risks associated with dermatologic advice offered on Reddit and should caution patients against relying solely on this information without consulting a qualified dermatologist first.
Reddit’s open-forum design provides licensed dermatologists with the opportunity to disseminate evidence based information regarding dermatologic conditions. Currently, there is a subreddit (r/AskDocs) that allows users to post medical questions that can be answered by moderator-verified physicians. Participation from dermatologists in online communities such as this can improve the quality of dermatologic information shared online, combat misinformation, and promote safe skin care practices.
ChatGPT offers more accurate, appropriate, and safe information compared to Reddit responses, but its answers lack personalization. In a clinical setting, a personalized treatment plan from a physician can be tailored with a comprehensive discussion of the risks and benefits. Further, clinical settings allow for diagnosis and confirmation via biopsy and meticulous history taking to ensure that the diagnosis and treatment plan are accurate. While ChatGPT may be an option for seeking basic advice on dermatologic conditions, a licensed dermatologist should always be consulted for proper medical advice. Services such as telehealth may be another option to for patients with limited access to care.
Since ChatGPT 3.5 does not support the ability to upload images, our study acknowledges a limitation regarding the inclusion of Reddit posts containing photographs. Images can improve the response quality from both Reddit users and ChatGPT. While the updated ChatGPT 4o is capable of processing images, it requires a monthly subscription fee. The free version was chosen for use in this study, as this may reflect the most likely version that patients of low socioeconomic status would utilize to access dermatologic care; however, there is potential for growth and improvement of ChatGPT’s capability in providing medical advice.
This study compared the strengths and limitations of ChatGPT’s and Reddit’s responses to common dermatologic inquiries. ChatGPT and Reddit both show potential to be helpful sources of dermatologic health information; however, their current versions have many limitations and require caution and careful examination by patients of the guidance provided. Clinicians should be aware of these limitations when advising patients and emphasize the importance of consulting a licensed dermatologist for personalized, evidence-based care. For the best medical advice, it is always advisable to consult with a licensed dermatologist.
- Roumeliotis KI, Tselikas ND. ChatGPT and open-AI models: a preliminary review. Future Internet. 2023;15:192. doi:10.3390/fi15060192
- Iqbal U, Lee LTJ, Rahmanti AR, et al. Can large language models provide secondary reliable opinion on treatment options for dermatological diseases? J Am Med Inform Assoc. 2024;31:1341-1347. doi:10.1093/jamia/ocae067
- Whiles BB, Bird VG, Canales BK, et al. Caution! AI bot has entered the patient chat: ChatGPT has limitations in providing accurate urologic healthcare advice. Urology. 2023;180:278-284. doi:10.1016/j.urology.2023.07.010
- Nastasi AJ, Courtright KR, Halpern SD, et al. A vignette-based evaluation of ChatGPT’s ability to provide appropriate and equitable medical advice across care contexts. Sci Rep. 2023;13:17885. doi:10.1038/s41598-023-45223-y
- Record RA, Silberman WR, Santiago JE, et al. I sought it, I Reddit: examining health information engagement behaviors among Reddit users. J Health Commun. 2018;23:470-476. doi:10.1080/1081073 0.2018.1465493
- Buntinx-Krieg T, Caravaglio J, Domozych R, et al. Dermatology on Reddit: elucidating trends in dermatologic communications on the world wide web. Dermatol Online J. 2017;23:13030/qt9dr1f7x6.
- Aboul-Fettouh N, Lee KP, Kash N, et al. Social media and dermatology during the COVID-19 pandemic: analyzing usersubmitted posts seeking dermatologic advice on Reddit. Cureus. 2023;15:E33720. doi:10.7759/cureus.33720
To the Editor:
Online resources that are convenient and affordable play a crucial role in mitigating health inequality and improving patient access to health care information; however, the benefits are limited by the quality of information available, as medical misinformation can lead to patients engaging in harmful practices, making dangerous decisions, and even avoiding safe and effective treatments. In this study, we aimed to assess and compare the quality of patient guidance on dermatologic care generated by ChatGPT vs Reddit based on accuracy, appropriateness, and safety. It is essential to assess the quality and reliability of online health information to support patients in making informed decisions about their health.
The emergence and advancement of artificial intelligence and large language models such as ChatGPT present a new method for patients to access health care advice. ChatGPT can engage in conversation by accessing information from existing publicly available data on the internet, including books and websites, up to the year 2023 and providing humanlike responses with context.1 ChatGPT’s access to a breadth of online evidence-based literature ensures the dissemination of quality information that is quick and without inherent bias, offering the potential to more closely align with health care professionals. ChatGPT’s use in dermatology by patients has shown efficacy, with a 98.87% approval rate by dermatologists scoring its ability to recommend appropriate medication for common dermatologic conditions.2 However, ChatGPT has limitations when providing health care advice and has been observed to misunderstand health care standards, lack personalization, and offer incorrect references; currently, the latest publicly available version (ChatGPT 3.5) also is unable to analyze clinical images.3,4
Reddit is an online social media forum that allows users to post questions and photographs to which anyone can reply and offer advice. Patients may find comfort in online communities where they can connect with others facing similar challenges related to their diagnosis. Within these communities, the responses often share users’ own lived experiences and offer support based on what has and has not worked for them. Prior research found that users intentionally seeking health information via Reddit are likely to implement the advice they receive even without verification of its credibility, suggesting a trust and receptibility to ideas offered on the platform.5 Furthermore, a study analyzing the dermatologic content of 17 dermatology related subreddits that had 1000 or more subscribers found that 70.6% of posts fell under the category of “seeking health/cosmetic advice.”6 Reddit users thus are vulnerable to receiving advice based on personal bias and exposing their health information to the public.
We hypothesized that ChatGPT would provide users with guidance that was more closely aligned with typical dermatologists’ advice due to its thorough analysis and compilation of diverse sources and recommendations available on the internet. We expected Reddit to yield recommendations of lesser quality and a diminished safety score, primarily due to the absence of credibility-vetting mechanisms and the influence of personal biases within the advice shared.
User-submitted posts to large dermatologic community Reddit forums representing a few of the most common skin conditions (r/eczema, r/acne, r/Folliculitis, r/SebDerm, r/Hidradenitis, r/keratosis, and r/Psoriasis) were retrospectively reviewed from January 2024 to March 2024. The most popular posts that did not include photographs were included in our study. Posts with photographs were excluded, as clinical images were not able to be uploaded to the publicly available ChatGPT 3.5. We collected real user questions about common skin conditions from Reddit forums and then asked ChatGPT to answer those same questions. We compared ChatGPT’s responses to the most upvoted Reddit comments to see how they matched up (eTable).

Each ChatGPT response and the top-rated Reddit comment were independently evaluated by a board certified dermatologist (S.A.) and a dermatology resident (A.H.K.). The quality of the ChatGPT and Reddit responses were determined by scoring the accuracy, appropriateness, safety consideration, and specificity on a 5-point Likert scale (1=low, 5=high). The 2 evaluators’ mean scores for each of the 4 categories were calculated based on adequate interrater reliability, which was tested using Cohen’s κ coefficient. Related-samples sign tests were used to compare ChatGPT and Reddit responses for each of the 4 categories. Analysis was completed using SPSS statistics software version 29.0 (IBM). The evaluators also were asked to provide qualitative feedback on the strengths and weaknesses of each response.
Our retrospective review yielded 20 total questions: 5 (25%) on atopic dermatitis, 4 (20%) on acne, 4 (20%) on hidradenitis suppurativa, 4 (20%) on psoriasis, 1 (5%) on folliculitis, 1 (5%) on keratosis pilaris, and 1 (5%) on seborrheic dermatitis. The number of posts was limited to 20 due to the extensive time required for grading each response. These 20 questions were selected from a larger pool of eligible posts based on factors such as clarity and relevance to common skin conditions. With regard to the types of questions that were asked, 6 (30%) were related to general management of a diagnosis, 5 (25%) were on treatment recommendations for symptom relief, 3 (15%) were on optimal utilization of current treatment regimens, 2 (10%) were on prescription side effects, 2 (10%) were on diagnosis presentation, 1 (5%) was on potential triggers of the diagnosis, and 1 (5%) was on natural treatment recommendations.
Mean (SD) evaluator scores for accuracy were significantly higher among ChatGPT responses compared with Reddit (4.63 [0.60] vs 2.60 [0.98])(P<.001). ChatGPT responses also were significantly higher for appropriateness compared with Reddit (4.55 [0.71] vs 2.58 [1.02])(P<.001) and safety consideration (4.88 [0.56] vs 2.80[0.97])(P <.001). There was no significant difference in mean specificity scores between ChatGPT and Reddit (4.25[1.02] vs 3.80 [0.70])(P=.096)(Figure).

For the Reddit responses, the weighted Cohen’s κ coefficient between the 2 evaluators was 0.200 (95% CI, –.089 to .489) for accuracy, 0.255 (95% CI, .014-.497) for appropriateness, 0.385 (95% CI, .176-.594) for safety consideration, and –0.024 (95% CI, –.177 to .129) for specificity. For the ChatGPT responses, the weighted Cohen’s κ coefficient between the 2 evaluators was 0.426 (95% CI, .122-.730) for accuracy, 0.571 (95% CI, .294-.849) for appropriateness, 0.655 (95% CI, .632-.678) for safety consideration, and 0.313 (95% CI, .043-.584) for specificity.
The strengths and weaknesses of the responses also were qualitatively analyzed. One commonly observed strength was ChatGPT’s frequent and appropriate recommendation for users to consult a dermatologist. In the case of atopic dermatitis—one of the more frequently asked about conditions—ChatGPT consistently emphasized evidence-based strategies such as gentle skin care and moisturization, reflecting alignment with clinical guidelines. Additionally, a common weakness of both ChatGPT and Reddit responses generally was the lack of personalized guidance and comprehensive discussion of the risks and benefits of specific treatments. It also was noted that neither platform consistently explored differential diagnoses—for example, distinguishing atopic dermatitis from conditions such as allergic contact dermatitis—limiting the diagnostic depth of the responses.
ChatGPT and Reddit can provide patients with quick and accessible health information for various dermatologic concerns. The results of our study demonstrated a significantly higher level of accuracy, appropriateness, and safety of responses generated by ChatGPT compared with human-generated responses on Reddit (P<.001). Both platforms offered similarly specific responses to user inquiries, demonstrating ChatGPT’s ability to comprehend user questions and draw from publicly available texts and Reddit users’ contributing insights based on their own first-hand experiences.
Reddit’s dermatologic forums often feature personal anecdotes and unique treatments described by individual users. Although specific to particular dermatologic concerns, such advice lacks an evidence-based standard of care. With the noted inherent trust of patients seeking guidance within Reddit communities, patients may follow unhelpful or potentially dangerous medical advice.5 A study examining 300 user-submitted posts on popular Reddit dermatology forums during the COVID-19 pandemic found that the mean scores for top-rated comments’ potential to be misleading or dangerous was 2.33 out of 5 on a Likert scale (95% CI, 2.18- 2.48).7 Dermatologists should be aware of the potential risks associated with dermatologic advice offered on Reddit and should caution patients against relying solely on this information without consulting a qualified dermatologist first.
Reddit’s open-forum design provides licensed dermatologists with the opportunity to disseminate evidence based information regarding dermatologic conditions. Currently, there is a subreddit (r/AskDocs) that allows users to post medical questions that can be answered by moderator-verified physicians. Participation from dermatologists in online communities such as this can improve the quality of dermatologic information shared online, combat misinformation, and promote safe skin care practices.
ChatGPT offers more accurate, appropriate, and safe information compared to Reddit responses, but its answers lack personalization. In a clinical setting, a personalized treatment plan from a physician can be tailored with a comprehensive discussion of the risks and benefits. Further, clinical settings allow for diagnosis and confirmation via biopsy and meticulous history taking to ensure that the diagnosis and treatment plan are accurate. While ChatGPT may be an option for seeking basic advice on dermatologic conditions, a licensed dermatologist should always be consulted for proper medical advice. Services such as telehealth may be another option to for patients with limited access to care.
Since ChatGPT 3.5 does not support the ability to upload images, our study acknowledges a limitation regarding the inclusion of Reddit posts containing photographs. Images can improve the response quality from both Reddit users and ChatGPT. While the updated ChatGPT 4o is capable of processing images, it requires a monthly subscription fee. The free version was chosen for use in this study, as this may reflect the most likely version that patients of low socioeconomic status would utilize to access dermatologic care; however, there is potential for growth and improvement of ChatGPT’s capability in providing medical advice.
This study compared the strengths and limitations of ChatGPT’s and Reddit’s responses to common dermatologic inquiries. ChatGPT and Reddit both show potential to be helpful sources of dermatologic health information; however, their current versions have many limitations and require caution and careful examination by patients of the guidance provided. Clinicians should be aware of these limitations when advising patients and emphasize the importance of consulting a licensed dermatologist for personalized, evidence-based care. For the best medical advice, it is always advisable to consult with a licensed dermatologist.
To the Editor:
Online resources that are convenient and affordable play a crucial role in mitigating health inequality and improving patient access to health care information; however, the benefits are limited by the quality of information available, as medical misinformation can lead to patients engaging in harmful practices, making dangerous decisions, and even avoiding safe and effective treatments. In this study, we aimed to assess and compare the quality of patient guidance on dermatologic care generated by ChatGPT vs Reddit based on accuracy, appropriateness, and safety. It is essential to assess the quality and reliability of online health information to support patients in making informed decisions about their health.
The emergence and advancement of artificial intelligence and large language models such as ChatGPT present a new method for patients to access health care advice. ChatGPT can engage in conversation by accessing information from existing publicly available data on the internet, including books and websites, up to the year 2023 and providing humanlike responses with context.1 ChatGPT’s access to a breadth of online evidence-based literature ensures the dissemination of quality information that is quick and without inherent bias, offering the potential to more closely align with health care professionals. ChatGPT’s use in dermatology by patients has shown efficacy, with a 98.87% approval rate by dermatologists scoring its ability to recommend appropriate medication for common dermatologic conditions.2 However, ChatGPT has limitations when providing health care advice and has been observed to misunderstand health care standards, lack personalization, and offer incorrect references; currently, the latest publicly available version (ChatGPT 3.5) also is unable to analyze clinical images.3,4
Reddit is an online social media forum that allows users to post questions and photographs to which anyone can reply and offer advice. Patients may find comfort in online communities where they can connect with others facing similar challenges related to their diagnosis. Within these communities, the responses often share users’ own lived experiences and offer support based on what has and has not worked for them. Prior research found that users intentionally seeking health information via Reddit are likely to implement the advice they receive even without verification of its credibility, suggesting a trust and receptibility to ideas offered on the platform.5 Furthermore, a study analyzing the dermatologic content of 17 dermatology related subreddits that had 1000 or more subscribers found that 70.6% of posts fell under the category of “seeking health/cosmetic advice.”6 Reddit users thus are vulnerable to receiving advice based on personal bias and exposing their health information to the public.
We hypothesized that ChatGPT would provide users with guidance that was more closely aligned with typical dermatologists’ advice due to its thorough analysis and compilation of diverse sources and recommendations available on the internet. We expected Reddit to yield recommendations of lesser quality and a diminished safety score, primarily due to the absence of credibility-vetting mechanisms and the influence of personal biases within the advice shared.
User-submitted posts to large dermatologic community Reddit forums representing a few of the most common skin conditions (r/eczema, r/acne, r/Folliculitis, r/SebDerm, r/Hidradenitis, r/keratosis, and r/Psoriasis) were retrospectively reviewed from January 2024 to March 2024. The most popular posts that did not include photographs were included in our study. Posts with photographs were excluded, as clinical images were not able to be uploaded to the publicly available ChatGPT 3.5. We collected real user questions about common skin conditions from Reddit forums and then asked ChatGPT to answer those same questions. We compared ChatGPT’s responses to the most upvoted Reddit comments to see how they matched up (eTable).

Each ChatGPT response and the top-rated Reddit comment were independently evaluated by a board certified dermatologist (S.A.) and a dermatology resident (A.H.K.). The quality of the ChatGPT and Reddit responses were determined by scoring the accuracy, appropriateness, safety consideration, and specificity on a 5-point Likert scale (1=low, 5=high). The 2 evaluators’ mean scores for each of the 4 categories were calculated based on adequate interrater reliability, which was tested using Cohen’s κ coefficient. Related-samples sign tests were used to compare ChatGPT and Reddit responses for each of the 4 categories. Analysis was completed using SPSS statistics software version 29.0 (IBM). The evaluators also were asked to provide qualitative feedback on the strengths and weaknesses of each response.
Our retrospective review yielded 20 total questions: 5 (25%) on atopic dermatitis, 4 (20%) on acne, 4 (20%) on hidradenitis suppurativa, 4 (20%) on psoriasis, 1 (5%) on folliculitis, 1 (5%) on keratosis pilaris, and 1 (5%) on seborrheic dermatitis. The number of posts was limited to 20 due to the extensive time required for grading each response. These 20 questions were selected from a larger pool of eligible posts based on factors such as clarity and relevance to common skin conditions. With regard to the types of questions that were asked, 6 (30%) were related to general management of a diagnosis, 5 (25%) were on treatment recommendations for symptom relief, 3 (15%) were on optimal utilization of current treatment regimens, 2 (10%) were on prescription side effects, 2 (10%) were on diagnosis presentation, 1 (5%) was on potential triggers of the diagnosis, and 1 (5%) was on natural treatment recommendations.
Mean (SD) evaluator scores for accuracy were significantly higher among ChatGPT responses compared with Reddit (4.63 [0.60] vs 2.60 [0.98])(P<.001). ChatGPT responses also were significantly higher for appropriateness compared with Reddit (4.55 [0.71] vs 2.58 [1.02])(P<.001) and safety consideration (4.88 [0.56] vs 2.80[0.97])(P <.001). There was no significant difference in mean specificity scores between ChatGPT and Reddit (4.25[1.02] vs 3.80 [0.70])(P=.096)(Figure).

For the Reddit responses, the weighted Cohen’s κ coefficient between the 2 evaluators was 0.200 (95% CI, –.089 to .489) for accuracy, 0.255 (95% CI, .014-.497) for appropriateness, 0.385 (95% CI, .176-.594) for safety consideration, and –0.024 (95% CI, –.177 to .129) for specificity. For the ChatGPT responses, the weighted Cohen’s κ coefficient between the 2 evaluators was 0.426 (95% CI, .122-.730) for accuracy, 0.571 (95% CI, .294-.849) for appropriateness, 0.655 (95% CI, .632-.678) for safety consideration, and 0.313 (95% CI, .043-.584) for specificity.
The strengths and weaknesses of the responses also were qualitatively analyzed. One commonly observed strength was ChatGPT’s frequent and appropriate recommendation for users to consult a dermatologist. In the case of atopic dermatitis—one of the more frequently asked about conditions—ChatGPT consistently emphasized evidence-based strategies such as gentle skin care and moisturization, reflecting alignment with clinical guidelines. Additionally, a common weakness of both ChatGPT and Reddit responses generally was the lack of personalized guidance and comprehensive discussion of the risks and benefits of specific treatments. It also was noted that neither platform consistently explored differential diagnoses—for example, distinguishing atopic dermatitis from conditions such as allergic contact dermatitis—limiting the diagnostic depth of the responses.
ChatGPT and Reddit can provide patients with quick and accessible health information for various dermatologic concerns. The results of our study demonstrated a significantly higher level of accuracy, appropriateness, and safety of responses generated by ChatGPT compared with human-generated responses on Reddit (P<.001). Both platforms offered similarly specific responses to user inquiries, demonstrating ChatGPT’s ability to comprehend user questions and draw from publicly available texts and Reddit users’ contributing insights based on their own first-hand experiences.
Reddit’s dermatologic forums often feature personal anecdotes and unique treatments described by individual users. Although specific to particular dermatologic concerns, such advice lacks an evidence-based standard of care. With the noted inherent trust of patients seeking guidance within Reddit communities, patients may follow unhelpful or potentially dangerous medical advice.5 A study examining 300 user-submitted posts on popular Reddit dermatology forums during the COVID-19 pandemic found that the mean scores for top-rated comments’ potential to be misleading or dangerous was 2.33 out of 5 on a Likert scale (95% CI, 2.18- 2.48).7 Dermatologists should be aware of the potential risks associated with dermatologic advice offered on Reddit and should caution patients against relying solely on this information without consulting a qualified dermatologist first.
Reddit’s open-forum design provides licensed dermatologists with the opportunity to disseminate evidence based information regarding dermatologic conditions. Currently, there is a subreddit (r/AskDocs) that allows users to post medical questions that can be answered by moderator-verified physicians. Participation from dermatologists in online communities such as this can improve the quality of dermatologic information shared online, combat misinformation, and promote safe skin care practices.
ChatGPT offers more accurate, appropriate, and safe information compared to Reddit responses, but its answers lack personalization. In a clinical setting, a personalized treatment plan from a physician can be tailored with a comprehensive discussion of the risks and benefits. Further, clinical settings allow for diagnosis and confirmation via biopsy and meticulous history taking to ensure that the diagnosis and treatment plan are accurate. While ChatGPT may be an option for seeking basic advice on dermatologic conditions, a licensed dermatologist should always be consulted for proper medical advice. Services such as telehealth may be another option to for patients with limited access to care.
Since ChatGPT 3.5 does not support the ability to upload images, our study acknowledges a limitation regarding the inclusion of Reddit posts containing photographs. Images can improve the response quality from both Reddit users and ChatGPT. While the updated ChatGPT 4o is capable of processing images, it requires a monthly subscription fee. The free version was chosen for use in this study, as this may reflect the most likely version that patients of low socioeconomic status would utilize to access dermatologic care; however, there is potential for growth and improvement of ChatGPT’s capability in providing medical advice.
This study compared the strengths and limitations of ChatGPT’s and Reddit’s responses to common dermatologic inquiries. ChatGPT and Reddit both show potential to be helpful sources of dermatologic health information; however, their current versions have many limitations and require caution and careful examination by patients of the guidance provided. Clinicians should be aware of these limitations when advising patients and emphasize the importance of consulting a licensed dermatologist for personalized, evidence-based care. For the best medical advice, it is always advisable to consult with a licensed dermatologist.
- Roumeliotis KI, Tselikas ND. ChatGPT and open-AI models: a preliminary review. Future Internet. 2023;15:192. doi:10.3390/fi15060192
- Iqbal U, Lee LTJ, Rahmanti AR, et al. Can large language models provide secondary reliable opinion on treatment options for dermatological diseases? J Am Med Inform Assoc. 2024;31:1341-1347. doi:10.1093/jamia/ocae067
- Whiles BB, Bird VG, Canales BK, et al. Caution! AI bot has entered the patient chat: ChatGPT has limitations in providing accurate urologic healthcare advice. Urology. 2023;180:278-284. doi:10.1016/j.urology.2023.07.010
- Nastasi AJ, Courtright KR, Halpern SD, et al. A vignette-based evaluation of ChatGPT’s ability to provide appropriate and equitable medical advice across care contexts. Sci Rep. 2023;13:17885. doi:10.1038/s41598-023-45223-y
- Record RA, Silberman WR, Santiago JE, et al. I sought it, I Reddit: examining health information engagement behaviors among Reddit users. J Health Commun. 2018;23:470-476. doi:10.1080/1081073 0.2018.1465493
- Buntinx-Krieg T, Caravaglio J, Domozych R, et al. Dermatology on Reddit: elucidating trends in dermatologic communications on the world wide web. Dermatol Online J. 2017;23:13030/qt9dr1f7x6.
- Aboul-Fettouh N, Lee KP, Kash N, et al. Social media and dermatology during the COVID-19 pandemic: analyzing usersubmitted posts seeking dermatologic advice on Reddit. Cureus. 2023;15:E33720. doi:10.7759/cureus.33720
- Roumeliotis KI, Tselikas ND. ChatGPT and open-AI models: a preliminary review. Future Internet. 2023;15:192. doi:10.3390/fi15060192
- Iqbal U, Lee LTJ, Rahmanti AR, et al. Can large language models provide secondary reliable opinion on treatment options for dermatological diseases? J Am Med Inform Assoc. 2024;31:1341-1347. doi:10.1093/jamia/ocae067
- Whiles BB, Bird VG, Canales BK, et al. Caution! AI bot has entered the patient chat: ChatGPT has limitations in providing accurate urologic healthcare advice. Urology. 2023;180:278-284. doi:10.1016/j.urology.2023.07.010
- Nastasi AJ, Courtright KR, Halpern SD, et al. A vignette-based evaluation of ChatGPT’s ability to provide appropriate and equitable medical advice across care contexts. Sci Rep. 2023;13:17885. doi:10.1038/s41598-023-45223-y
- Record RA, Silberman WR, Santiago JE, et al. I sought it, I Reddit: examining health information engagement behaviors among Reddit users. J Health Commun. 2018;23:470-476. doi:10.1080/1081073 0.2018.1465493
- Buntinx-Krieg T, Caravaglio J, Domozych R, et al. Dermatology on Reddit: elucidating trends in dermatologic communications on the world wide web. Dermatol Online J. 2017;23:13030/qt9dr1f7x6.
- Aboul-Fettouh N, Lee KP, Kash N, et al. Social media and dermatology during the COVID-19 pandemic: analyzing usersubmitted posts seeking dermatologic advice on Reddit. Cureus. 2023;15:E33720. doi:10.7759/cureus.33720
Comparing the Quality of Patient Guidance on Dermatologic Care Generated by ChatGPT vs Reddit
Comparing the Quality of Patient Guidance on Dermatologic Care Generated by ChatGPT vs Reddit
PRACTICE POINTS
- ChatGPT and Reddit are free, convenient, and accessible online resources that patients may use for guidance on dermatologic care.
- Dermatologists should be aware of the potential risks associated with obtaining medical guidance from ChatGPT and Reddit and caution patients on them.
- An increasing presence of dermatologists on online public forums can increase the dissemination of reliable health care information.
Large Bullae on the Legs in a Hospitalized Patient Following a Gunshot Wound
Large Bullae on the Legs in a Hospitalized Patient Following a Gunshot Wound
THE DIAGNOSIS: Bullous Hemorrhagic Dermatosis
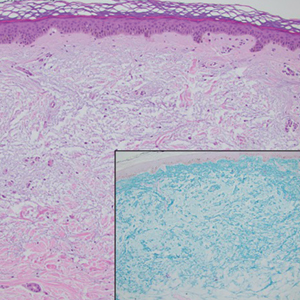
Biopsy results showed an intraepidermal blister with a floor composed of maturing epidermis. The roof of the blister was composed of necrotic keratinocytes with overlying orthokeratosis, and the cavity was filled with a moderate amount of fibrin and dead cells with neutrophils. Direct immunofluorescence (DIF) using specific antihuman IgG, IgM, IgA, C3, and fibrin was negative. Aerobic, anaerobic, and fungal cultures also were negative. With these histopathologic findings, medication exposure, and timing of bullae onset, our patient was diagnosed with bullous hemorrhagic dermatosis (BHD) secondary to enoxaparin administration. Enoxaparin was continued due to increased risk for coagulopathy, and there was complete resolution of the bullae after 5 weeks with no residual symptoms.
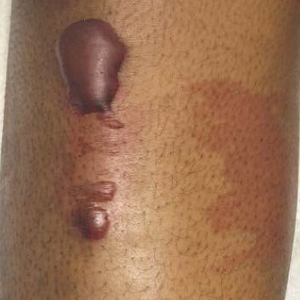
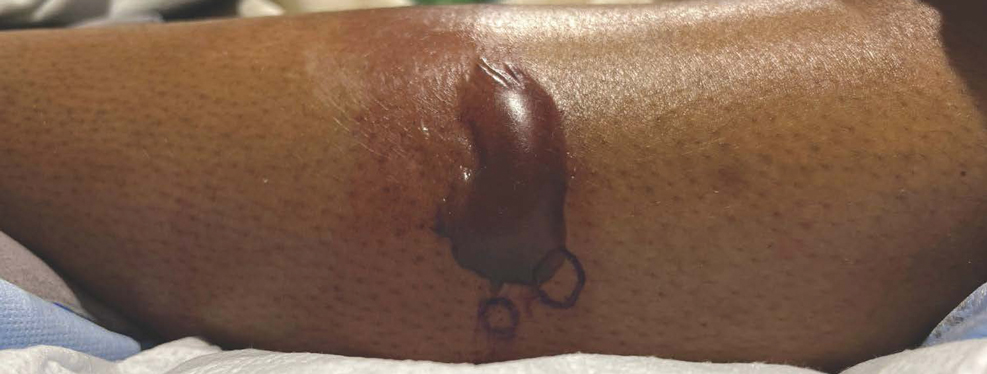
Bullous hemorrhagic dermatosis is a rare eruption that can occur after administration of heparin and low-molecular-weight heparin, with enoxaparin being the most commonly implicated drug.1 The lesions typically are seen in elderly men in the seventh decade of life and appear within a median of 7 days after drug exposure. The time course for the postexposure eruption can vary from 2 to 21 days, with reports of skin lesions appearing up to 4 months after exposure.1,2 hemorrhagic bullae (Figure) typically on the arms and legs, though lesions also can develop on the trunk. The lesions can occur in distant areas from the injection site, suggesting BHD may be a systemic reaction, although the etiology is poorly understood.1
Another heparin reaction that can manifest similarly to BHD is heparin-induced skin necrosis.3 Patients with this condition also may have associated heparin-induced thrombocytopenia upon laboratory investigation and have a more aggressive clinical course than BHD. Biopsy can help differentiate BHD and early heparin-induced skin necrosis if the clinical manifestation is unclear. Histopathologically, BHD typically has intraepidermal bullae filled with blood, whereas heparin-induced skin necrosis has dermal thrombi.1,4 Treatment of both conditions differs in whether to discontinue anticoagulants: heparin-induced skin necrosis requires discontinuation of the medication, while BHD does not.2,3
In patients with BHD, the lesions are self-resolving, and treatment is supportive, although whether enoxaparin is discontinued varies among physicians.2 Lesions typically resolve within 2 weeks of onset, although it is unclear whether continuing anticoagulants delays resolution.1 Discontinuing anticoagulants in certain patients can be life-threatening due to complex comorbidities (eg, risk for venous thromboembolism or pulmonary embolism from prolonged hospitalization or severe trauma) and is not necessary for the resolution of BHD.
In addition to BHD and heparin-induced skin necrosis, our differential diagnosis included bullous pemphigoid, coma blisters, and Vibrio vulnificus infection. Although bullous pemphigoid can manifest with tense bullae that are pauci-inflammatory on histology, DIF would show linear IgG and C3 deposition at the dermal-epidermal junction. In our patient, DIF was negative and favored another etiology for the lesions. Coma blisters can occur in areas of sustained pressure and typically develop in patients with a prolonged hospitalization or those who are sedentary for long periods of time. The distribution of bullae on our patient’s bilateral pretibial shins made this diagnosis unlikely. Vibrio vulnificus infection can manifest as hemorrhagic bullae, though typically after a break in the skin exposed to brackish water. Vibrio vulnificus infection can be life-threatening, resulting in septicemia and increased mortality, and a thorough patient history is important for diagnosis.5
- Russo A, Curtis S, Balbuena-Merle R, et al. Bullous hemorrhagic dermatosis is an under-recognized side effect of full dose lowmolecular weight heparin: a case report and review of the literature. Exp Hematol Oncol. 2018;7:15. doi:10.1186/s40164-018-0108-7
- Dhattarwal N, Gurjar R. Bullous hemorrhagic dermatosis: a rare cutaneous reaction of heparin. J Postgrad Med. 2023;69:97-98. doi:10.4103/jpgm.jpgm_282_22
- Maldonado Cid P, Alonso de Celada RM, Noguera Morel L, et al. Cutaneous adverse events associated with heparin. Clin Exp Dermatol. 2012;37:707-711. doi:10.1111/j.1365-2230.2012.04395.x
- Handschin AE, Trentz O, Kock HJ, et al. Low molecular weight heparininduced skin necrosis-a systematic review. Langenbecks Arch Surg. 2005;390:249-254. doi:10.1007/s00423-004-0522-7
- Jones MK, Oliver JD. Vibrio vulnificus: disease and pathogenesis. Infect Immun. 2009;77:1723-1733. doi:10.1128/IAI.01046-08
THE DIAGNOSIS: Bullous Hemorrhagic Dermatosis
Biopsy results showed an intraepidermal blister with a floor composed of maturing epidermis. The roof of the blister was composed of necrotic keratinocytes with overlying orthokeratosis, and the cavity was filled with a moderate amount of fibrin and dead cells with neutrophils. Direct immunofluorescence (DIF) using specific antihuman IgG, IgM, IgA, C3, and fibrin was negative. Aerobic, anaerobic, and fungal cultures also were negative. With these histopathologic findings, medication exposure, and timing of bullae onset, our patient was diagnosed with bullous hemorrhagic dermatosis (BHD) secondary to enoxaparin administration. Enoxaparin was continued due to increased risk for coagulopathy, and there was complete resolution of the bullae after 5 weeks with no residual symptoms.
Bullous hemorrhagic dermatosis is a rare eruption that can occur after administration of heparin and low-molecular-weight heparin, with enoxaparin being the most commonly implicated drug.1 The lesions typically are seen in elderly men in the seventh decade of life and appear within a median of 7 days after drug exposure. The time course for the postexposure eruption can vary from 2 to 21 days, with reports of skin lesions appearing up to 4 months after exposure.1,2 hemorrhagic bullae (Figure) typically on the arms and legs, though lesions also can develop on the trunk. The lesions can occur in distant areas from the injection site, suggesting BHD may be a systemic reaction, although the etiology is poorly understood.1

Another heparin reaction that can manifest similarly to BHD is heparin-induced skin necrosis.3 Patients with this condition also may have associated heparin-induced thrombocytopenia upon laboratory investigation and have a more aggressive clinical course than BHD. Biopsy can help differentiate BHD and early heparin-induced skin necrosis if the clinical manifestation is unclear. Histopathologically, BHD typically has intraepidermal bullae filled with blood, whereas heparin-induced skin necrosis has dermal thrombi.1,4 Treatment of both conditions differs in whether to discontinue anticoagulants: heparin-induced skin necrosis requires discontinuation of the medication, while BHD does not.2,3
In patients with BHD, the lesions are self-resolving, and treatment is supportive, although whether enoxaparin is discontinued varies among physicians.2 Lesions typically resolve within 2 weeks of onset, although it is unclear whether continuing anticoagulants delays resolution.1 Discontinuing anticoagulants in certain patients can be life-threatening due to complex comorbidities (eg, risk for venous thromboembolism or pulmonary embolism from prolonged hospitalization or severe trauma) and is not necessary for the resolution of BHD.
In addition to BHD and heparin-induced skin necrosis, our differential diagnosis included bullous pemphigoid, coma blisters, and Vibrio vulnificus infection. Although bullous pemphigoid can manifest with tense bullae that are pauci-inflammatory on histology, DIF would show linear IgG and C3 deposition at the dermal-epidermal junction. In our patient, DIF was negative and favored another etiology for the lesions. Coma blisters can occur in areas of sustained pressure and typically develop in patients with a prolonged hospitalization or those who are sedentary for long periods of time. The distribution of bullae on our patient’s bilateral pretibial shins made this diagnosis unlikely. Vibrio vulnificus infection can manifest as hemorrhagic bullae, though typically after a break in the skin exposed to brackish water. Vibrio vulnificus infection can be life-threatening, resulting in septicemia and increased mortality, and a thorough patient history is important for diagnosis.5
THE DIAGNOSIS: Bullous Hemorrhagic Dermatosis
Biopsy results showed an intraepidermal blister with a floor composed of maturing epidermis. The roof of the blister was composed of necrotic keratinocytes with overlying orthokeratosis, and the cavity was filled with a moderate amount of fibrin and dead cells with neutrophils. Direct immunofluorescence (DIF) using specific antihuman IgG, IgM, IgA, C3, and fibrin was negative. Aerobic, anaerobic, and fungal cultures also were negative. With these histopathologic findings, medication exposure, and timing of bullae onset, our patient was diagnosed with bullous hemorrhagic dermatosis (BHD) secondary to enoxaparin administration. Enoxaparin was continued due to increased risk for coagulopathy, and there was complete resolution of the bullae after 5 weeks with no residual symptoms.
Bullous hemorrhagic dermatosis is a rare eruption that can occur after administration of heparin and low-molecular-weight heparin, with enoxaparin being the most commonly implicated drug.1 The lesions typically are seen in elderly men in the seventh decade of life and appear within a median of 7 days after drug exposure. The time course for the postexposure eruption can vary from 2 to 21 days, with reports of skin lesions appearing up to 4 months after exposure.1,2 hemorrhagic bullae (Figure) typically on the arms and legs, though lesions also can develop on the trunk. The lesions can occur in distant areas from the injection site, suggesting BHD may be a systemic reaction, although the etiology is poorly understood.1

Another heparin reaction that can manifest similarly to BHD is heparin-induced skin necrosis.3 Patients with this condition also may have associated heparin-induced thrombocytopenia upon laboratory investigation and have a more aggressive clinical course than BHD. Biopsy can help differentiate BHD and early heparin-induced skin necrosis if the clinical manifestation is unclear. Histopathologically, BHD typically has intraepidermal bullae filled with blood, whereas heparin-induced skin necrosis has dermal thrombi.1,4 Treatment of both conditions differs in whether to discontinue anticoagulants: heparin-induced skin necrosis requires discontinuation of the medication, while BHD does not.2,3
In patients with BHD, the lesions are self-resolving, and treatment is supportive, although whether enoxaparin is discontinued varies among physicians.2 Lesions typically resolve within 2 weeks of onset, although it is unclear whether continuing anticoagulants delays resolution.1 Discontinuing anticoagulants in certain patients can be life-threatening due to complex comorbidities (eg, risk for venous thromboembolism or pulmonary embolism from prolonged hospitalization or severe trauma) and is not necessary for the resolution of BHD.
In addition to BHD and heparin-induced skin necrosis, our differential diagnosis included bullous pemphigoid, coma blisters, and Vibrio vulnificus infection. Although bullous pemphigoid can manifest with tense bullae that are pauci-inflammatory on histology, DIF would show linear IgG and C3 deposition at the dermal-epidermal junction. In our patient, DIF was negative and favored another etiology for the lesions. Coma blisters can occur in areas of sustained pressure and typically develop in patients with a prolonged hospitalization or those who are sedentary for long periods of time. The distribution of bullae on our patient’s bilateral pretibial shins made this diagnosis unlikely. Vibrio vulnificus infection can manifest as hemorrhagic bullae, though typically after a break in the skin exposed to brackish water. Vibrio vulnificus infection can be life-threatening, resulting in septicemia and increased mortality, and a thorough patient history is important for diagnosis.5
- Russo A, Curtis S, Balbuena-Merle R, et al. Bullous hemorrhagic dermatosis is an under-recognized side effect of full dose lowmolecular weight heparin: a case report and review of the literature. Exp Hematol Oncol. 2018;7:15. doi:10.1186/s40164-018-0108-7
- Dhattarwal N, Gurjar R. Bullous hemorrhagic dermatosis: a rare cutaneous reaction of heparin. J Postgrad Med. 2023;69:97-98. doi:10.4103/jpgm.jpgm_282_22
- Maldonado Cid P, Alonso de Celada RM, Noguera Morel L, et al. Cutaneous adverse events associated with heparin. Clin Exp Dermatol. 2012;37:707-711. doi:10.1111/j.1365-2230.2012.04395.x
- Handschin AE, Trentz O, Kock HJ, et al. Low molecular weight heparininduced skin necrosis-a systematic review. Langenbecks Arch Surg. 2005;390:249-254. doi:10.1007/s00423-004-0522-7
- Jones MK, Oliver JD. Vibrio vulnificus: disease and pathogenesis. Infect Immun. 2009;77:1723-1733. doi:10.1128/IAI.01046-08
- Russo A, Curtis S, Balbuena-Merle R, et al. Bullous hemorrhagic dermatosis is an under-recognized side effect of full dose lowmolecular weight heparin: a case report and review of the literature. Exp Hematol Oncol. 2018;7:15. doi:10.1186/s40164-018-0108-7
- Dhattarwal N, Gurjar R. Bullous hemorrhagic dermatosis: a rare cutaneous reaction of heparin. J Postgrad Med. 2023;69:97-98. doi:10.4103/jpgm.jpgm_282_22
- Maldonado Cid P, Alonso de Celada RM, Noguera Morel L, et al. Cutaneous adverse events associated with heparin. Clin Exp Dermatol. 2012;37:707-711. doi:10.1111/j.1365-2230.2012.04395.x
- Handschin AE, Trentz O, Kock HJ, et al. Low molecular weight heparininduced skin necrosis-a systematic review. Langenbecks Arch Surg. 2005;390:249-254. doi:10.1007/s00423-004-0522-7
- Jones MK, Oliver JD. Vibrio vulnificus: disease and pathogenesis. Infect Immun. 2009;77:1723-1733. doi:10.1128/IAI.01046-08
Large Bullae on the Legs in a Hospitalized Patient Following a Gunshot Wound
Large Bullae on the Legs in a Hospitalized Patient Following a Gunshot Wound
A 19-year-old man developed fluid-filled blisters on both legs within 1 month of a prolonged hospitalization following a gunshot wound that resulted in complete paralysis of the legs. His medical history was otherwise unremarkable. Medications started during hospitalization included moxifloxacin, levetiracetam, and prophylactic subcutaneous enoxaparin. Physical examination by dermatology revealed tense blood-filled bullae measuring several centimeters with well-demarcated, pink to red, irregularly shaped patches on both legs. A biopsy of a blister was taken.
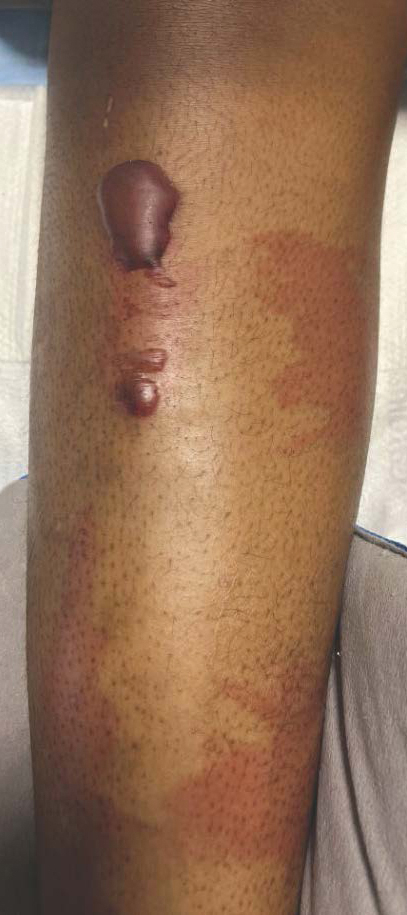
Low-Dose Oral Naltrexone for Darier Disease
To the Editor:
A 34-year-old Brazilian woman presented to the dermatology department with pruritic lesions on the neck and chest that had been present since adolescence. She reported a family history of Darier disease in her father. Physical examination revealed erythematous follicular papules on the neck, inframammary region, and abdomen (Figure 1A), as well as longitudinal bandlike leukonychia and distal nail splits on the fingernails (Figure 1B). Histopathology of a lesion on the back revealed compact hyperkeratosis and parakeratosis above an acantholytic cleft accompanied by dyskeratotic keratinocytes, including some corps ronds and grains, which supported the clinical impression of Darier disease (Figure 2). The typical clinical presentation along with the family history and histopathology confirmed the diagnosis. After therapeutic failure with topical corticosteroids and oral antibiotics for 3 months, low-dose oral naltrexone (4.5 mg/d) as monotherapy noticeably improved the lesions and pruritus within 2 months, with near-complete regression at 6 months, achieving disease stability (Figures 1C and 1D). The patient remained stable with no recurrence after 1 year of follow-up.
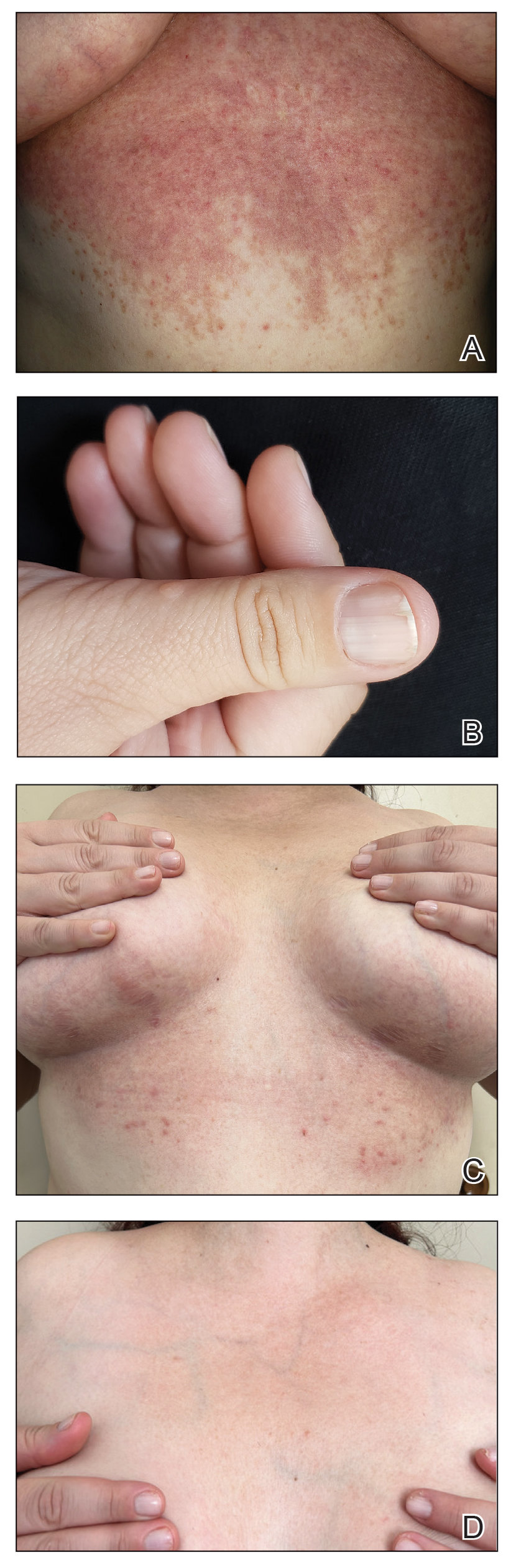
Darier disease is an autosomal-dominant genodermatosis caused by a mutation in the ATP2A2 gene, which encodes the sarco/endoplasmic reticulum calcium ATPase, leading to defective intracellular calcium signaling and alterations in epidermal adhesion and keratinization.1 Darier disease typically begins in adolescence and is aggravated by exposure to heat and friction. It is characterized by seborrheic distribution of painful and pruritic red-brown keratotic papules. Nail manifestations include longitudinal ridges—erythronychia and/or leukonychia—and grooves that end in a V-shaped notch. The differential diagnosis includes Hailey-Hailey disease, psoriasis, and pityriasis rubra pilaris.1,2 The diagnosis is clinical and is confirmed by histopathology, which reveals suprabasal cleavage, acantholytic dyskeratosis, corps ronds, and grains. Treatment options are limited and include corticosteroids, oral and/or topical antibiotics, and systemic retinoids.2
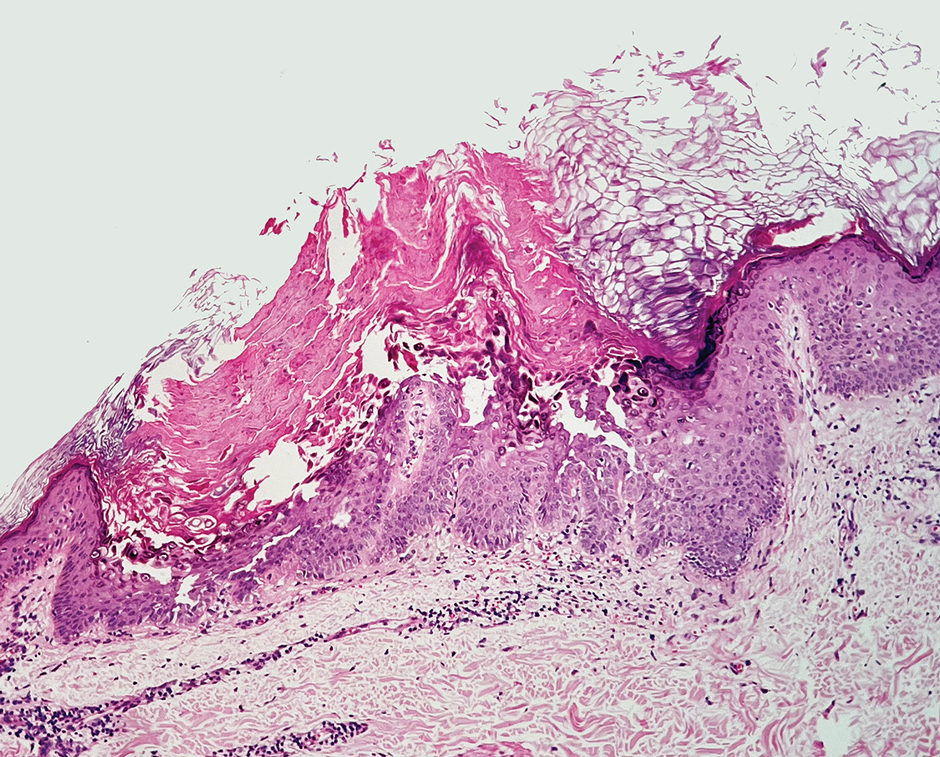
Oral naltrexone has been used in Darier disease based on its observed effectiveness in Hailey-Hailey disease, considering the histopathologic similarities and alterations in calcium homeostasis in both conditions. Low-dose oral naltrexone (1-5 mg/d) increases the expression of opioid receptors (δ, μ, κ), enhancing its immunomodulatory and antinociceptive effects. The δ opioid receptor regulates the expression of desmoglein, improving epidermal differentiation and wound healing.3 Activation of the δ and μ receptors increases intracellular calcium through the inositol phosphate pathway, which contributes to calcium homeostasis.4 Naltrexone blocks the nonopioid toll-like receptor 4 found in keratinocytes and macrophages, exerting an anti-inflammatory effect by reducing proinflammatory cytokines.3 Adverse events associated with low-dose naltrexone are minimal, mostly mild, and often related to sleep disorders3,5; however, patients should undergo screening for prior opioid dependence, recent opioid usage, and signs of opioid withdrawal before initiating naltrexone treatment.5
Boehmer et al6 used naltrexone (4.5 mg/d) and oral magnesium (200 mg/d) in 6 patients with inconsistent results, except for 1 case that concurrently used acitretin (25 mg/d) with satisfactory improvement. Pessoa et al7 added naltrexone (4.5 mg/d) to oral isotretinoin (0.5 mg/kg/d) in 1 patient, resulting in notable improvement of lesions within 3 months.
In our patient with Darier disease, low-dose naltrexone demonstrated a substantial response as monotherapy after 2 months of treatment and nearly complete regression of lesions within 6 months, with no reported side effects after 1 year of follow-up. The use of low-dose naltrexone could be a promising and safe treatment option as monotherapy or in combination with conventional therapy for Darier disease; however, further studies are needed.
Sakuntabhai A, Ruiz-Perez V, Carter S, et al. Mutations in ATP2A2, encoding a Ca2+ pump, cause Darier disease. Nat Genet. 1999;21:271-277. doi:10.1038/6784
Burge SM, Wilkinson JD. Darier-White disease: a review of the clinical features in 163 patients. J Am Acad Dermatol. 1992;27:40-50. doi:10.1016/0190-9622(92)70154-8
Lee B, Elston DM. The uses of naltrexone in dermatologic conditions. Am Acad Dermatol. 2019;80:1746-1752. doi:10.1016/j.jaad.2018.12.031
Samways DSK, Henderson G. Opioid elevation of intracellular free calcium: possible mechanisms and physiological relevance. Cell Signal. 2006;18:151-161. doi:10.1016/j.cellsig.2005.08.005
Ekelem C, Juhasz M, Khera P, et al. Utility of naltrexone treatment for chronic inflammatory dermatologic conditions: a systematic review. JAMA Dermatol. 2019;155:229-236. doi:10.1001/jamadermatol.2018.4093
Boehmer D, Eyerich K, Darsow U, et al. Variable response to low‐dose naltrexone in patients with Darier disease: a case series. J Eur Acad Dermatol Venereol. 2019;33:950-953. doi:10.1111/jdv.15457
Pessoa T, Rebelo C, Gabriela Marques Pinto, et al. Combination of naltrexone and isotretinoin for the treatment of Darier disease. Cureus. 2023;15:E33321. doi:10.7759/cureus.33321
To the Editor:
A 34-year-old Brazilian woman presented to the dermatology department with pruritic lesions on the neck and chest that had been present since adolescence. She reported a family history of Darier disease in her father. Physical examination revealed erythematous follicular papules on the neck, inframammary region, and abdomen (Figure 1A), as well as longitudinal bandlike leukonychia and distal nail splits on the fingernails (Figure 1B). Histopathology of a lesion on the back revealed compact hyperkeratosis and parakeratosis above an acantholytic cleft accompanied by dyskeratotic keratinocytes, including some corps ronds and grains, which supported the clinical impression of Darier disease (Figure 2). The typical clinical presentation along with the family history and histopathology confirmed the diagnosis. After therapeutic failure with topical corticosteroids and oral antibiotics for 3 months, low-dose oral naltrexone (4.5 mg/d) as monotherapy noticeably improved the lesions and pruritus within 2 months, with near-complete regression at 6 months, achieving disease stability (Figures 1C and 1D). The patient remained stable with no recurrence after 1 year of follow-up.

Darier disease is an autosomal-dominant genodermatosis caused by a mutation in the ATP2A2 gene, which encodes the sarco/endoplasmic reticulum calcium ATPase, leading to defective intracellular calcium signaling and alterations in epidermal adhesion and keratinization.1 Darier disease typically begins in adolescence and is aggravated by exposure to heat and friction. It is characterized by seborrheic distribution of painful and pruritic red-brown keratotic papules. Nail manifestations include longitudinal ridges—erythronychia and/or leukonychia—and grooves that end in a V-shaped notch. The differential diagnosis includes Hailey-Hailey disease, psoriasis, and pityriasis rubra pilaris.1,2 The diagnosis is clinical and is confirmed by histopathology, which reveals suprabasal cleavage, acantholytic dyskeratosis, corps ronds, and grains. Treatment options are limited and include corticosteroids, oral and/or topical antibiotics, and systemic retinoids.2

Oral naltrexone has been used in Darier disease based on its observed effectiveness in Hailey-Hailey disease, considering the histopathologic similarities and alterations in calcium homeostasis in both conditions. Low-dose oral naltrexone (1-5 mg/d) increases the expression of opioid receptors (δ, μ, κ), enhancing its immunomodulatory and antinociceptive effects. The δ opioid receptor regulates the expression of desmoglein, improving epidermal differentiation and wound healing.3 Activation of the δ and μ receptors increases intracellular calcium through the inositol phosphate pathway, which contributes to calcium homeostasis.4 Naltrexone blocks the nonopioid toll-like receptor 4 found in keratinocytes and macrophages, exerting an anti-inflammatory effect by reducing proinflammatory cytokines.3 Adverse events associated with low-dose naltrexone are minimal, mostly mild, and often related to sleep disorders3,5; however, patients should undergo screening for prior opioid dependence, recent opioid usage, and signs of opioid withdrawal before initiating naltrexone treatment.5
Boehmer et al6 used naltrexone (4.5 mg/d) and oral magnesium (200 mg/d) in 6 patients with inconsistent results, except for 1 case that concurrently used acitretin (25 mg/d) with satisfactory improvement. Pessoa et al7 added naltrexone (4.5 mg/d) to oral isotretinoin (0.5 mg/kg/d) in 1 patient, resulting in notable improvement of lesions within 3 months.
In our patient with Darier disease, low-dose naltrexone demonstrated a substantial response as monotherapy after 2 months of treatment and nearly complete regression of lesions within 6 months, with no reported side effects after 1 year of follow-up. The use of low-dose naltrexone could be a promising and safe treatment option as monotherapy or in combination with conventional therapy for Darier disease; however, further studies are needed.
To the Editor:
A 34-year-old Brazilian woman presented to the dermatology department with pruritic lesions on the neck and chest that had been present since adolescence. She reported a family history of Darier disease in her father. Physical examination revealed erythematous follicular papules on the neck, inframammary region, and abdomen (Figure 1A), as well as longitudinal bandlike leukonychia and distal nail splits on the fingernails (Figure 1B). Histopathology of a lesion on the back revealed compact hyperkeratosis and parakeratosis above an acantholytic cleft accompanied by dyskeratotic keratinocytes, including some corps ronds and grains, which supported the clinical impression of Darier disease (Figure 2). The typical clinical presentation along with the family history and histopathology confirmed the diagnosis. After therapeutic failure with topical corticosteroids and oral antibiotics for 3 months, low-dose oral naltrexone (4.5 mg/d) as monotherapy noticeably improved the lesions and pruritus within 2 months, with near-complete regression at 6 months, achieving disease stability (Figures 1C and 1D). The patient remained stable with no recurrence after 1 year of follow-up.

Darier disease is an autosomal-dominant genodermatosis caused by a mutation in the ATP2A2 gene, which encodes the sarco/endoplasmic reticulum calcium ATPase, leading to defective intracellular calcium signaling and alterations in epidermal adhesion and keratinization.1 Darier disease typically begins in adolescence and is aggravated by exposure to heat and friction. It is characterized by seborrheic distribution of painful and pruritic red-brown keratotic papules. Nail manifestations include longitudinal ridges—erythronychia and/or leukonychia—and grooves that end in a V-shaped notch. The differential diagnosis includes Hailey-Hailey disease, psoriasis, and pityriasis rubra pilaris.1,2 The diagnosis is clinical and is confirmed by histopathology, which reveals suprabasal cleavage, acantholytic dyskeratosis, corps ronds, and grains. Treatment options are limited and include corticosteroids, oral and/or topical antibiotics, and systemic retinoids.2

Oral naltrexone has been used in Darier disease based on its observed effectiveness in Hailey-Hailey disease, considering the histopathologic similarities and alterations in calcium homeostasis in both conditions. Low-dose oral naltrexone (1-5 mg/d) increases the expression of opioid receptors (δ, μ, κ), enhancing its immunomodulatory and antinociceptive effects. The δ opioid receptor regulates the expression of desmoglein, improving epidermal differentiation and wound healing.3 Activation of the δ and μ receptors increases intracellular calcium through the inositol phosphate pathway, which contributes to calcium homeostasis.4 Naltrexone blocks the nonopioid toll-like receptor 4 found in keratinocytes and macrophages, exerting an anti-inflammatory effect by reducing proinflammatory cytokines.3 Adverse events associated with low-dose naltrexone are minimal, mostly mild, and often related to sleep disorders3,5; however, patients should undergo screening for prior opioid dependence, recent opioid usage, and signs of opioid withdrawal before initiating naltrexone treatment.5
Boehmer et al6 used naltrexone (4.5 mg/d) and oral magnesium (200 mg/d) in 6 patients with inconsistent results, except for 1 case that concurrently used acitretin (25 mg/d) with satisfactory improvement. Pessoa et al7 added naltrexone (4.5 mg/d) to oral isotretinoin (0.5 mg/kg/d) in 1 patient, resulting in notable improvement of lesions within 3 months.
In our patient with Darier disease, low-dose naltrexone demonstrated a substantial response as monotherapy after 2 months of treatment and nearly complete regression of lesions within 6 months, with no reported side effects after 1 year of follow-up. The use of low-dose naltrexone could be a promising and safe treatment option as monotherapy or in combination with conventional therapy for Darier disease; however, further studies are needed.
Sakuntabhai A, Ruiz-Perez V, Carter S, et al. Mutations in ATP2A2, encoding a Ca2+ pump, cause Darier disease. Nat Genet. 1999;21:271-277. doi:10.1038/6784
Burge SM, Wilkinson JD. Darier-White disease: a review of the clinical features in 163 patients. J Am Acad Dermatol. 1992;27:40-50. doi:10.1016/0190-9622(92)70154-8
Lee B, Elston DM. The uses of naltrexone in dermatologic conditions. Am Acad Dermatol. 2019;80:1746-1752. doi:10.1016/j.jaad.2018.12.031
Samways DSK, Henderson G. Opioid elevation of intracellular free calcium: possible mechanisms and physiological relevance. Cell Signal. 2006;18:151-161. doi:10.1016/j.cellsig.2005.08.005
Ekelem C, Juhasz M, Khera P, et al. Utility of naltrexone treatment for chronic inflammatory dermatologic conditions: a systematic review. JAMA Dermatol. 2019;155:229-236. doi:10.1001/jamadermatol.2018.4093
Boehmer D, Eyerich K, Darsow U, et al. Variable response to low‐dose naltrexone in patients with Darier disease: a case series. J Eur Acad Dermatol Venereol. 2019;33:950-953. doi:10.1111/jdv.15457
Pessoa T, Rebelo C, Gabriela Marques Pinto, et al. Combination of naltrexone and isotretinoin for the treatment of Darier disease. Cureus. 2023;15:E33321. doi:10.7759/cureus.33321
Sakuntabhai A, Ruiz-Perez V, Carter S, et al. Mutations in ATP2A2, encoding a Ca2+ pump, cause Darier disease. Nat Genet. 1999;21:271-277. doi:10.1038/6784
Burge SM, Wilkinson JD. Darier-White disease: a review of the clinical features in 163 patients. J Am Acad Dermatol. 1992;27:40-50. doi:10.1016/0190-9622(92)70154-8
Lee B, Elston DM. The uses of naltrexone in dermatologic conditions. Am Acad Dermatol. 2019;80:1746-1752. doi:10.1016/j.jaad.2018.12.031
Samways DSK, Henderson G. Opioid elevation of intracellular free calcium: possible mechanisms and physiological relevance. Cell Signal. 2006;18:151-161. doi:10.1016/j.cellsig.2005.08.005
Ekelem C, Juhasz M, Khera P, et al. Utility of naltrexone treatment for chronic inflammatory dermatologic conditions: a systematic review. JAMA Dermatol. 2019;155:229-236. doi:10.1001/jamadermatol.2018.4093
Boehmer D, Eyerich K, Darsow U, et al. Variable response to low‐dose naltrexone in patients with Darier disease: a case series. J Eur Acad Dermatol Venereol. 2019;33:950-953. doi:10.1111/jdv.15457
Pessoa T, Rebelo C, Gabriela Marques Pinto, et al. Combination of naltrexone and isotretinoin for the treatment of Darier disease. Cureus. 2023;15:E33321. doi:10.7759/cureus.33321
Practice Points
- Consider low-dose naltrexone as a potential treatment option for patients with Darier disease, as it regulates opioid receptors and has shown benefits in enhancing epidermal differentiation, wound healing, and anti-inflammatory effects.
- Further research is needed to validate the efficacy and safety of low-dose naltrexone in treating Darier disease considering its observed clinical improvement in this single patient case.
An Uncertain Future for No-Cost Preventive Care
Later this month, the U.S. Supreme Court is anticipated to announce its decision in Kennedy vs. Braidwood Management, a case that could significantly impact the no-cost coverage of preventive healthcare services under the Patient Protection and Affordable Care Act (ACA). At the center of the case is whether the structure of the U.S. Preventive Services Task Force (USPSTF) – an independent body convened by the federal government that makes recommendations for preventive services that nearly all private insurances must cover without cost sharing under provisions of the ACA (specifically, Grade A and B recommendations) – violates the Appointments Clause of the U.S. Constitution. This clause states that “officers of the United States” may only be appointed by the president with the Senate’s approval.
The case, initiated in 2022 by a self-insured, Christian-owned business, specifically targeted the coverage of pre-exposure prophylaxis (PrEP) for preventing HIV in high-risk individuals. However, the decision could broadly affect the coverage of other preventive services, including colorectal cancer screening tests. In June 2024, the 5th Circuit Court of Appeals upheld a district court’s ruling that the ACA’s requirement to cover without cost-sharing services recommended by USPSTF is unconstitutional, paving the way for the current Supreme Court showdown.
The consequences of this ruling could be significant. This would likely reverse the progress we have made in increasing colorectal cancer screening rates by reducing financial barriers to care. Interestingly, despite a new administration, the federal government continues to advocate for upholding the law, asserting that USPSTF members are “inferior officers” such that the Secretary of Health and Human Services can dismiss individual members and oversee or veto the Task Force’s recommendations at will, potentially threatening scientific independence. Though it’s often challenging to predict the Supreme Court’s final decision, the tone of questioning during oral arguments in April hinted at a possible win for the ACA and preventive care. Stay tuned, as the decision to be released later this month has seismic clinical implications.
Megan A. Adams, MD, JD, MSc
Editor in Chief
Later this month, the U.S. Supreme Court is anticipated to announce its decision in Kennedy vs. Braidwood Management, a case that could significantly impact the no-cost coverage of preventive healthcare services under the Patient Protection and Affordable Care Act (ACA). At the center of the case is whether the structure of the U.S. Preventive Services Task Force (USPSTF) – an independent body convened by the federal government that makes recommendations for preventive services that nearly all private insurances must cover without cost sharing under provisions of the ACA (specifically, Grade A and B recommendations) – violates the Appointments Clause of the U.S. Constitution. This clause states that “officers of the United States” may only be appointed by the president with the Senate’s approval.
The case, initiated in 2022 by a self-insured, Christian-owned business, specifically targeted the coverage of pre-exposure prophylaxis (PrEP) for preventing HIV in high-risk individuals. However, the decision could broadly affect the coverage of other preventive services, including colorectal cancer screening tests. In June 2024, the 5th Circuit Court of Appeals upheld a district court’s ruling that the ACA’s requirement to cover without cost-sharing services recommended by USPSTF is unconstitutional, paving the way for the current Supreme Court showdown.
The consequences of this ruling could be significant. This would likely reverse the progress we have made in increasing colorectal cancer screening rates by reducing financial barriers to care. Interestingly, despite a new administration, the federal government continues to advocate for upholding the law, asserting that USPSTF members are “inferior officers” such that the Secretary of Health and Human Services can dismiss individual members and oversee or veto the Task Force’s recommendations at will, potentially threatening scientific independence. Though it’s often challenging to predict the Supreme Court’s final decision, the tone of questioning during oral arguments in April hinted at a possible win for the ACA and preventive care. Stay tuned, as the decision to be released later this month has seismic clinical implications.
Megan A. Adams, MD, JD, MSc
Editor in Chief
Later this month, the U.S. Supreme Court is anticipated to announce its decision in Kennedy vs. Braidwood Management, a case that could significantly impact the no-cost coverage of preventive healthcare services under the Patient Protection and Affordable Care Act (ACA). At the center of the case is whether the structure of the U.S. Preventive Services Task Force (USPSTF) – an independent body convened by the federal government that makes recommendations for preventive services that nearly all private insurances must cover without cost sharing under provisions of the ACA (specifically, Grade A and B recommendations) – violates the Appointments Clause of the U.S. Constitution. This clause states that “officers of the United States” may only be appointed by the president with the Senate’s approval.
The case, initiated in 2022 by a self-insured, Christian-owned business, specifically targeted the coverage of pre-exposure prophylaxis (PrEP) for preventing HIV in high-risk individuals. However, the decision could broadly affect the coverage of other preventive services, including colorectal cancer screening tests. In June 2024, the 5th Circuit Court of Appeals upheld a district court’s ruling that the ACA’s requirement to cover without cost-sharing services recommended by USPSTF is unconstitutional, paving the way for the current Supreme Court showdown.
The consequences of this ruling could be significant. This would likely reverse the progress we have made in increasing colorectal cancer screening rates by reducing financial barriers to care. Interestingly, despite a new administration, the federal government continues to advocate for upholding the law, asserting that USPSTF members are “inferior officers” such that the Secretary of Health and Human Services can dismiss individual members and oversee or veto the Task Force’s recommendations at will, potentially threatening scientific independence. Though it’s often challenging to predict the Supreme Court’s final decision, the tone of questioning during oral arguments in April hinted at a possible win for the ACA and preventive care. Stay tuned, as the decision to be released later this month has seismic clinical implications.
Megan A. Adams, MD, JD, MSc
Editor in Chief
Should I Stay or Should I Go? Federal Health Care Professional Retirement Dilemmas
Should I Stay or Should I Go? Federal Health Care Professional Retirement Dilemmas
The uselessness of men above sixty years of age and the incalculable benefit it would be in commercial, in political, and in professional life, if as a matter of course, men stopped working at this age.
Sir William Osler1
The first time I remember hearing the word retirement was when I was 5 or 6 years old. My mother told me that my father had been given new orders: either be promoted to general and move to oversee a hospital somewhere far away, or retire from the Army. He was a scholar, teacher, and physician with no interest or aptitude for military politics and health care administration. Reluctantly, he resigned himself to retirement before he had planned. I recall being angry with him, because in my solipsistic child mind he was depriving me of the opportunity to live in a big house across from the parade field, where the generals lived or having a reserved parking spot in front of the post exchange. As a psychiatrist, I suspect that the anger was a primitive defense against the fear of leaving the only home I had ever known on an Army base.
I recently finished reading Michael Bliss’s seminal biography of Sir William Osler (1848-1919), the great Anglo-American physician and medical educator.2 Bliss found few blemishes on Osler’s character or missteps in his stellar career, but one of the few may be his views on retirement. The epigraph is from an address Osler gave before leaving Johns Hopkins for semiretirement in Oxford, England. The farewell speech caused a media controversy with his comments reflecting attitudes that seem ageist today, when many people are active, productive, and happy long past the age of 60 years.3 I do not endorse Osler’s philosophy of aging, nor his exclusion of women (if I did, I would not be around to write this editorial). Not even Osler himself followed his advice: he was active in medicine almost until his death at 70 years old.2
Yet like many of my fellow federal health care practitioners (HCPs), I have been thinking about and planning for retirement earlier than expected, given the memos and directives about voluntary early retirement, deferred resignation, and reductions in force.4,5 The COVID-19 pandemic sadly compelled many burned-out and traumatized HCPs to cross the retirement Rubicon far sooner than they imagined.6
A Google search for information about HCP retirement, particularly among physicians, produces a cascade of advisory articles. They primarily focus on finances, with many pushing their own commercial agenda for retirement planning.7 Although money is a necessary piece of the retirement puzzle, for HCPs it may not be sufficient to ensure a healthy and satisfying retirement. Two other considerations may be even more important to weigh in making the retirement decision, namely timing and meaning.8
For earlier generations of HCPs, work was almost their sole identity. Although younger practitioners are more likely to embrace a better work-life balance, it is still a driving factor for many in the decision to retire.9 It is not just about the cliché of being a workaholic, rather many clinicians continue to enjoy lifelong learning, the rewards of helping people in need, and professional satisfaction. HCPs also spend a longer time training than many other professions; perhaps since we waited so long to practice, we want to stay a little longer.10 For those whose motivation for federal practice was a commitment to service, these may be even more powerful incentives to continue working.
When a nurse, physician, pharmacist, or social worker no longer finds the same gratification and stimulation in their work, whether due to unwelcome changes in the clinical setting or the profession at large, declining health or emotional exhaustion, or the very human need to move onto another phase of life (what Osler likely really meant), then that may be a signal to think hard about retiring. Of course, there have always been—and will continue to be—professionals of all stripes who, even in the most agreeable situation, just cannot wait to retire. Simply because there are so many other ways they want to spend their remaining energy and time: travel, grandchildren, hobbies, even a second career. Because none of us knows how far out our life extends, it is prudent to periodically ask what is the optimal path that combines both purpose and well-being.
All of us as HCPs, and even more as human beings with desires and duties far beyond our respective professions, face a dilemma: a choice between 2 goods that cannot both be fulfilled simultaneously. This is likely why HCPs frequently do what is technically called a phased retirement, a fancy name for working part-time, or retiring from 1 position and taking up another. This temporizes the decision and tempers the bittersweet emotional experience of leaving the profession in one way, and in another, it delays the inevitable.
Over the last few years, I have learned 2 important lessons while watching many of my closest friends retire. First, for those who are still working and those who are retired may seem to inhabit a separate country; hence, special efforts must be made to both appreciate them while they are in our immediate circle of concern and to make efforts to stay in contact once they are emeriti. It is almost as if after being a daily integral aspect of the workplace they have passed into a different dimension of existence. In terms of priorities and mindsets, many of them have. Second, what makes retirement a reality with peace and growth rather than regret and stagnation is owning the decision to retire. There are always constraints: financial, medical, and familial. However, those who retire on their own terms and not primarily in response to fear or uncertainty appear to fare better than those feeling the same pressures who give away their power.11 Having read about retirement in the last months, the best advice I have seen is from Harry Emerson Fosdick, a Protestant minister in the early 20th century: “Don’t simply retire from something; have something to retire to.”12
I have not yet decided about my retirement. Whatever decision you make, remember it is solely yours. After a lifetime of caring for others, retirement is all about caring for yourself.
- Osler W. The Fixed Period. In: Osler W, ed. Aequanimitas With Other Addresses to Medical Students, Nurses and Practitioners of Medicine. 3rd ed. The Blakiston Company; 1932:373-393.
- Bliss M. William Osler: A Life in Medicine. Oxford University Press; 1999.
- Anderson M, Scofield RH. The “Fixed period,” the wildfire news, and an unpublished manuscript: Osler’s farewell speech revisited in geographical breadth and emotional depth. Am J Med Sci. Published online February 11, 2025. doi:10.1016/j.amjms.2025.02.005
- Obis A. What federal workers should consider before accepting deferred resignation. Federal News Network. April 8, 2025. Accessed April 25, 2025. https://federalnewsnetwork.com/workforce/2025/04/what-federal-workers-should-consider-before-accepting-deferred-resignation/
- Dyer J. VA exempts clinical staff from OPM deferred resignation program. Federal Practitioner. February 11, 2025. Accessed April 28, 2025. https://www.mdedge.com/content/va-exempts-clinical-staff-opm-deferred-resignation-program
- Shyrock T. Retirement planning secrets for physicians. Medical Economics. 2024;101(8). Accessed April 28, 2025. https:// www.medicaleconomics.com/view/retirement-planningsecrets-for-physicians
- Sinsky CA, Brown RL, Stillman MJ, Linzer M. COVID-related stress and work intentions in a sample of US health care workers. Mayo Clin Proc Innov Qual Outcomes. 2021;5(6):1165-1173. doi:10.1016/j.mayocpiqo.2021.08.007
- Tabloski PA. Life after retirement. American Nurse. March 3, 2022. Accessed April 25, 2025. https://www.myamericannurse.com/life-after-retirement/
- Chen T-P. Young doctors want work-life balance. Older doctors say that’s not the job. The Wall Street Journal. November 3, 2024. Accessed April 25, 2025. https://www.wsj.com/lifestyle/careers/young-doctors-want-work-life-balance-older-doctors-say-thats-not-the-job-6cb37d48
- Sweeny JF. Physician retirement: Why it’s hard for doctors to retire. Medical Economics. 2019;96(4). Accessed April 25, 2025. https://www.medicaleconomics.com/view/physician-retirement-why-its-hard-doctors-retire
- Nelson J. Wisdom for Our Time. W.W. Norton; 1961.
- Silver MP, Hamilton AD, Biswas A, Williams SA. Life after medicine: a systematic review of studies physician’s adjustment to retirement. Arch Community Med Public Health. 2016;2(1):001-007. doi:10.17352/2455-5479.000006
The uselessness of men above sixty years of age and the incalculable benefit it would be in commercial, in political, and in professional life, if as a matter of course, men stopped working at this age.
Sir William Osler1
The first time I remember hearing the word retirement was when I was 5 or 6 years old. My mother told me that my father had been given new orders: either be promoted to general and move to oversee a hospital somewhere far away, or retire from the Army. He was a scholar, teacher, and physician with no interest or aptitude for military politics and health care administration. Reluctantly, he resigned himself to retirement before he had planned. I recall being angry with him, because in my solipsistic child mind he was depriving me of the opportunity to live in a big house across from the parade field, where the generals lived or having a reserved parking spot in front of the post exchange. As a psychiatrist, I suspect that the anger was a primitive defense against the fear of leaving the only home I had ever known on an Army base.
I recently finished reading Michael Bliss’s seminal biography of Sir William Osler (1848-1919), the great Anglo-American physician and medical educator.2 Bliss found few blemishes on Osler’s character or missteps in his stellar career, but one of the few may be his views on retirement. The epigraph is from an address Osler gave before leaving Johns Hopkins for semiretirement in Oxford, England. The farewell speech caused a media controversy with his comments reflecting attitudes that seem ageist today, when many people are active, productive, and happy long past the age of 60 years.3 I do not endorse Osler’s philosophy of aging, nor his exclusion of women (if I did, I would not be around to write this editorial). Not even Osler himself followed his advice: he was active in medicine almost until his death at 70 years old.2
Yet like many of my fellow federal health care practitioners (HCPs), I have been thinking about and planning for retirement earlier than expected, given the memos and directives about voluntary early retirement, deferred resignation, and reductions in force.4,5 The COVID-19 pandemic sadly compelled many burned-out and traumatized HCPs to cross the retirement Rubicon far sooner than they imagined.6
A Google search for information about HCP retirement, particularly among physicians, produces a cascade of advisory articles. They primarily focus on finances, with many pushing their own commercial agenda for retirement planning.7 Although money is a necessary piece of the retirement puzzle, for HCPs it may not be sufficient to ensure a healthy and satisfying retirement. Two other considerations may be even more important to weigh in making the retirement decision, namely timing and meaning.8
For earlier generations of HCPs, work was almost their sole identity. Although younger practitioners are more likely to embrace a better work-life balance, it is still a driving factor for many in the decision to retire.9 It is not just about the cliché of being a workaholic, rather many clinicians continue to enjoy lifelong learning, the rewards of helping people in need, and professional satisfaction. HCPs also spend a longer time training than many other professions; perhaps since we waited so long to practice, we want to stay a little longer.10 For those whose motivation for federal practice was a commitment to service, these may be even more powerful incentives to continue working.
When a nurse, physician, pharmacist, or social worker no longer finds the same gratification and stimulation in their work, whether due to unwelcome changes in the clinical setting or the profession at large, declining health or emotional exhaustion, or the very human need to move onto another phase of life (what Osler likely really meant), then that may be a signal to think hard about retiring. Of course, there have always been—and will continue to be—professionals of all stripes who, even in the most agreeable situation, just cannot wait to retire. Simply because there are so many other ways they want to spend their remaining energy and time: travel, grandchildren, hobbies, even a second career. Because none of us knows how far out our life extends, it is prudent to periodically ask what is the optimal path that combines both purpose and well-being.
All of us as HCPs, and even more as human beings with desires and duties far beyond our respective professions, face a dilemma: a choice between 2 goods that cannot both be fulfilled simultaneously. This is likely why HCPs frequently do what is technically called a phased retirement, a fancy name for working part-time, or retiring from 1 position and taking up another. This temporizes the decision and tempers the bittersweet emotional experience of leaving the profession in one way, and in another, it delays the inevitable.
Over the last few years, I have learned 2 important lessons while watching many of my closest friends retire. First, for those who are still working and those who are retired may seem to inhabit a separate country; hence, special efforts must be made to both appreciate them while they are in our immediate circle of concern and to make efforts to stay in contact once they are emeriti. It is almost as if after being a daily integral aspect of the workplace they have passed into a different dimension of existence. In terms of priorities and mindsets, many of them have. Second, what makes retirement a reality with peace and growth rather than regret and stagnation is owning the decision to retire. There are always constraints: financial, medical, and familial. However, those who retire on their own terms and not primarily in response to fear or uncertainty appear to fare better than those feeling the same pressures who give away their power.11 Having read about retirement in the last months, the best advice I have seen is from Harry Emerson Fosdick, a Protestant minister in the early 20th century: “Don’t simply retire from something; have something to retire to.”12
I have not yet decided about my retirement. Whatever decision you make, remember it is solely yours. After a lifetime of caring for others, retirement is all about caring for yourself.
The uselessness of men above sixty years of age and the incalculable benefit it would be in commercial, in political, and in professional life, if as a matter of course, men stopped working at this age.
Sir William Osler1
The first time I remember hearing the word retirement was when I was 5 or 6 years old. My mother told me that my father had been given new orders: either be promoted to general and move to oversee a hospital somewhere far away, or retire from the Army. He was a scholar, teacher, and physician with no interest or aptitude for military politics and health care administration. Reluctantly, he resigned himself to retirement before he had planned. I recall being angry with him, because in my solipsistic child mind he was depriving me of the opportunity to live in a big house across from the parade field, where the generals lived or having a reserved parking spot in front of the post exchange. As a psychiatrist, I suspect that the anger was a primitive defense against the fear of leaving the only home I had ever known on an Army base.
I recently finished reading Michael Bliss’s seminal biography of Sir William Osler (1848-1919), the great Anglo-American physician and medical educator.2 Bliss found few blemishes on Osler’s character or missteps in his stellar career, but one of the few may be his views on retirement. The epigraph is from an address Osler gave before leaving Johns Hopkins for semiretirement in Oxford, England. The farewell speech caused a media controversy with his comments reflecting attitudes that seem ageist today, when many people are active, productive, and happy long past the age of 60 years.3 I do not endorse Osler’s philosophy of aging, nor his exclusion of women (if I did, I would not be around to write this editorial). Not even Osler himself followed his advice: he was active in medicine almost until his death at 70 years old.2
Yet like many of my fellow federal health care practitioners (HCPs), I have been thinking about and planning for retirement earlier than expected, given the memos and directives about voluntary early retirement, deferred resignation, and reductions in force.4,5 The COVID-19 pandemic sadly compelled many burned-out and traumatized HCPs to cross the retirement Rubicon far sooner than they imagined.6
A Google search for information about HCP retirement, particularly among physicians, produces a cascade of advisory articles. They primarily focus on finances, with many pushing their own commercial agenda for retirement planning.7 Although money is a necessary piece of the retirement puzzle, for HCPs it may not be sufficient to ensure a healthy and satisfying retirement. Two other considerations may be even more important to weigh in making the retirement decision, namely timing and meaning.8
For earlier generations of HCPs, work was almost their sole identity. Although younger practitioners are more likely to embrace a better work-life balance, it is still a driving factor for many in the decision to retire.9 It is not just about the cliché of being a workaholic, rather many clinicians continue to enjoy lifelong learning, the rewards of helping people in need, and professional satisfaction. HCPs also spend a longer time training than many other professions; perhaps since we waited so long to practice, we want to stay a little longer.10 For those whose motivation for federal practice was a commitment to service, these may be even more powerful incentives to continue working.
When a nurse, physician, pharmacist, or social worker no longer finds the same gratification and stimulation in their work, whether due to unwelcome changes in the clinical setting or the profession at large, declining health or emotional exhaustion, or the very human need to move onto another phase of life (what Osler likely really meant), then that may be a signal to think hard about retiring. Of course, there have always been—and will continue to be—professionals of all stripes who, even in the most agreeable situation, just cannot wait to retire. Simply because there are so many other ways they want to spend their remaining energy and time: travel, grandchildren, hobbies, even a second career. Because none of us knows how far out our life extends, it is prudent to periodically ask what is the optimal path that combines both purpose and well-being.
All of us as HCPs, and even more as human beings with desires and duties far beyond our respective professions, face a dilemma: a choice between 2 goods that cannot both be fulfilled simultaneously. This is likely why HCPs frequently do what is technically called a phased retirement, a fancy name for working part-time, or retiring from 1 position and taking up another. This temporizes the decision and tempers the bittersweet emotional experience of leaving the profession in one way, and in another, it delays the inevitable.
Over the last few years, I have learned 2 important lessons while watching many of my closest friends retire. First, for those who are still working and those who are retired may seem to inhabit a separate country; hence, special efforts must be made to both appreciate them while they are in our immediate circle of concern and to make efforts to stay in contact once they are emeriti. It is almost as if after being a daily integral aspect of the workplace they have passed into a different dimension of existence. In terms of priorities and mindsets, many of them have. Second, what makes retirement a reality with peace and growth rather than regret and stagnation is owning the decision to retire. There are always constraints: financial, medical, and familial. However, those who retire on their own terms and not primarily in response to fear or uncertainty appear to fare better than those feeling the same pressures who give away their power.11 Having read about retirement in the last months, the best advice I have seen is from Harry Emerson Fosdick, a Protestant minister in the early 20th century: “Don’t simply retire from something; have something to retire to.”12
I have not yet decided about my retirement. Whatever decision you make, remember it is solely yours. After a lifetime of caring for others, retirement is all about caring for yourself.
- Osler W. The Fixed Period. In: Osler W, ed. Aequanimitas With Other Addresses to Medical Students, Nurses and Practitioners of Medicine. 3rd ed. The Blakiston Company; 1932:373-393.
- Bliss M. William Osler: A Life in Medicine. Oxford University Press; 1999.
- Anderson M, Scofield RH. The “Fixed period,” the wildfire news, and an unpublished manuscript: Osler’s farewell speech revisited in geographical breadth and emotional depth. Am J Med Sci. Published online February 11, 2025. doi:10.1016/j.amjms.2025.02.005
- Obis A. What federal workers should consider before accepting deferred resignation. Federal News Network. April 8, 2025. Accessed April 25, 2025. https://federalnewsnetwork.com/workforce/2025/04/what-federal-workers-should-consider-before-accepting-deferred-resignation/
- Dyer J. VA exempts clinical staff from OPM deferred resignation program. Federal Practitioner. February 11, 2025. Accessed April 28, 2025. https://www.mdedge.com/content/va-exempts-clinical-staff-opm-deferred-resignation-program
- Shyrock T. Retirement planning secrets for physicians. Medical Economics. 2024;101(8). Accessed April 28, 2025. https:// www.medicaleconomics.com/view/retirement-planningsecrets-for-physicians
- Sinsky CA, Brown RL, Stillman MJ, Linzer M. COVID-related stress and work intentions in a sample of US health care workers. Mayo Clin Proc Innov Qual Outcomes. 2021;5(6):1165-1173. doi:10.1016/j.mayocpiqo.2021.08.007
- Tabloski PA. Life after retirement. American Nurse. March 3, 2022. Accessed April 25, 2025. https://www.myamericannurse.com/life-after-retirement/
- Chen T-P. Young doctors want work-life balance. Older doctors say that’s not the job. The Wall Street Journal. November 3, 2024. Accessed April 25, 2025. https://www.wsj.com/lifestyle/careers/young-doctors-want-work-life-balance-older-doctors-say-thats-not-the-job-6cb37d48
- Sweeny JF. Physician retirement: Why it’s hard for doctors to retire. Medical Economics. 2019;96(4). Accessed April 25, 2025. https://www.medicaleconomics.com/view/physician-retirement-why-its-hard-doctors-retire
- Nelson J. Wisdom for Our Time. W.W. Norton; 1961.
- Silver MP, Hamilton AD, Biswas A, Williams SA. Life after medicine: a systematic review of studies physician’s adjustment to retirement. Arch Community Med Public Health. 2016;2(1):001-007. doi:10.17352/2455-5479.000006
- Osler W. The Fixed Period. In: Osler W, ed. Aequanimitas With Other Addresses to Medical Students, Nurses and Practitioners of Medicine. 3rd ed. The Blakiston Company; 1932:373-393.
- Bliss M. William Osler: A Life in Medicine. Oxford University Press; 1999.
- Anderson M, Scofield RH. The “Fixed period,” the wildfire news, and an unpublished manuscript: Osler’s farewell speech revisited in geographical breadth and emotional depth. Am J Med Sci. Published online February 11, 2025. doi:10.1016/j.amjms.2025.02.005
- Obis A. What federal workers should consider before accepting deferred resignation. Federal News Network. April 8, 2025. Accessed April 25, 2025. https://federalnewsnetwork.com/workforce/2025/04/what-federal-workers-should-consider-before-accepting-deferred-resignation/
- Dyer J. VA exempts clinical staff from OPM deferred resignation program. Federal Practitioner. February 11, 2025. Accessed April 28, 2025. https://www.mdedge.com/content/va-exempts-clinical-staff-opm-deferred-resignation-program
- Shyrock T. Retirement planning secrets for physicians. Medical Economics. 2024;101(8). Accessed April 28, 2025. https:// www.medicaleconomics.com/view/retirement-planningsecrets-for-physicians
- Sinsky CA, Brown RL, Stillman MJ, Linzer M. COVID-related stress and work intentions in a sample of US health care workers. Mayo Clin Proc Innov Qual Outcomes. 2021;5(6):1165-1173. doi:10.1016/j.mayocpiqo.2021.08.007
- Tabloski PA. Life after retirement. American Nurse. March 3, 2022. Accessed April 25, 2025. https://www.myamericannurse.com/life-after-retirement/
- Chen T-P. Young doctors want work-life balance. Older doctors say that’s not the job. The Wall Street Journal. November 3, 2024. Accessed April 25, 2025. https://www.wsj.com/lifestyle/careers/young-doctors-want-work-life-balance-older-doctors-say-thats-not-the-job-6cb37d48
- Sweeny JF. Physician retirement: Why it’s hard for doctors to retire. Medical Economics. 2019;96(4). Accessed April 25, 2025. https://www.medicaleconomics.com/view/physician-retirement-why-its-hard-doctors-retire
- Nelson J. Wisdom for Our Time. W.W. Norton; 1961.
- Silver MP, Hamilton AD, Biswas A, Williams SA. Life after medicine: a systematic review of studies physician’s adjustment to retirement. Arch Community Med Public Health. 2016;2(1):001-007. doi:10.17352/2455-5479.000006
Should I Stay or Should I Go? Federal Health Care Professional Retirement Dilemmas
Should I Stay or Should I Go? Federal Health Care Professional Retirement Dilemmas
Multiple Firm Papules on the Wrists and Forearms
Multiple Firm Papules on the Wrists and Forearms
THE DIAGNOSIS: Acral Persistent Papular Mucinosis
Histopathologic analysis revealed conspicuous interstitial mucin deposition throughout the upper to mid reticular dermis in the absence of a cellular infiltrate or fibroplasia. Colloidal iron staining confirmed the presence of mucin. In correlation with the clinical presentation, a diagnosis of acral persistent papular mucinosis (APPM) was made. The patient was counseled on the benign disease course and lack of associated comorbidities, and additional treatment was not pursued.
Acral persistent papular mucinosis is a rare distinct subtype of cutaneous mucinosis that initially was described by Rongioletti et al1 in 1986. As a localized form of lichen myxedematosus, APPM is characterized by mucin deposition in the dermis with no systemic involvement. The precise pathogenesis remains unclear, although some investigators have suggested that cytokine-mediated stimulation of glycosaminoglycan production may contribute to increased mucin accumulation in the dermis.2 Acral persistent papular mucinosis predominantly affects middle-aged women with a 5:1 female-to-male predominance.3 Clinically, patients present with discrete, nonfollicular, waxy papules that typically measure 2 to 5 mm and are distributed symmetrically on the extensor surfaces of the wrists and forearms. While the lesions generally are asymptomatic, some patients may report mild pruritus. The condition is chronic, with lesions seldom resolving and often increasing in number over time.3
Histologically, APPM is characterized by focal deposits of mucin in the upper reticular dermis with no evidence of increased fibroblast proliferation or fibrosis.4 This feature is pivotal in differentiating APPM from other subtypes of localized lichen myxedematosus and similar dermatoses. Diagnosis of APPM requires exclusion of systemic involvement, including thyroid abnormalities and monoclonal gammopathy, aligning with its classification as a purely cutaneous condition.5 Management of APPM is unclear due to its rarity. Reassurance for patients of its benign nature as well as clinical observation are recommended, though some reports cite benefits of treatment with topical corticosteroids or calcineurin inhibitors.6,7 The long-term prognosis for patients with APPM is favorable, although the persistence of and potential increase in lesions over time can be a cosmetic concern.
The differential diagnoses for APPM include scleromyxedema, scleredema, and other cutaneous eruptions that manifest as smooth flesh-colored papules, such as granuloma annulare and lichen nitidus.3 Scleromyxedema is a systemic cutaneous mucinosis that is part of the same disease spectrum as lichen myxedematosus. The papular eruption of scleromyxedema is much more widespread, and coalescing of the lesions may lead to characteristic skin thickening, creating leonine facies and deep furrowing over the trunk.8 Extracutaneous manifestations are frequent in scleromyxedema, and up to 90% of patients exhibit evidence of an underlying plasma cell dyscrasia.2 Histopathologically, scleromyxedema shows extensive fibroblast proliferation and fibrosis, in contrast to the findings of APPM (Figure 1).
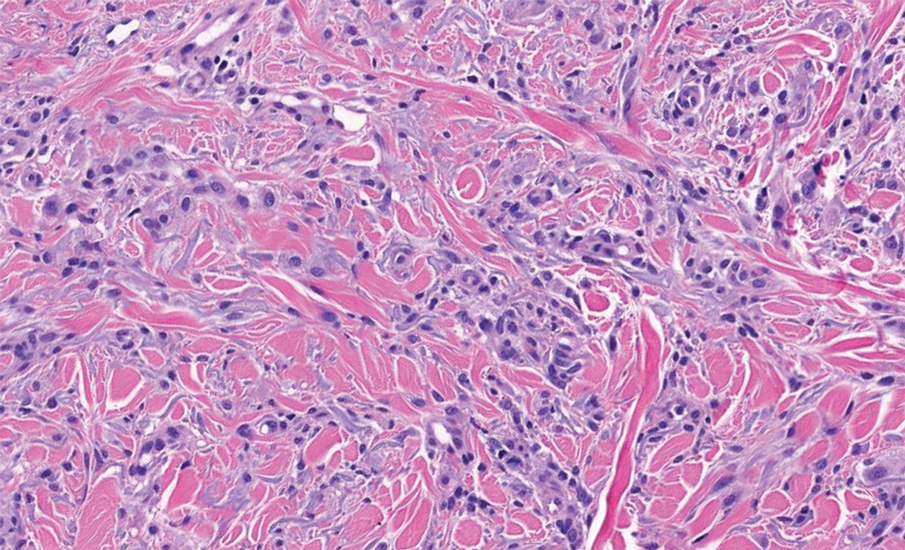
The histopathology of APPM is most similar to scleredema, a rare fibromucinous disorder of the skin associated with diabetes, infection (especially poststreptococcal), or monoclonal gammopathy.9 Biopsy evaluation of scleredema reveals a normal epidermis with mucin deposition between collagen bundles predominantly in the deep reticular dermis as well as absent fibroblast proliferation (Figure 2). Unlike APPM, scleredema manifests with diffuse woody induration with erythema and hyperpigmentation on the posterior neck and upper back.9 On physical examination, the distinct clinical features of scleredema distinguish this condition from APPM and scleromyxedema.
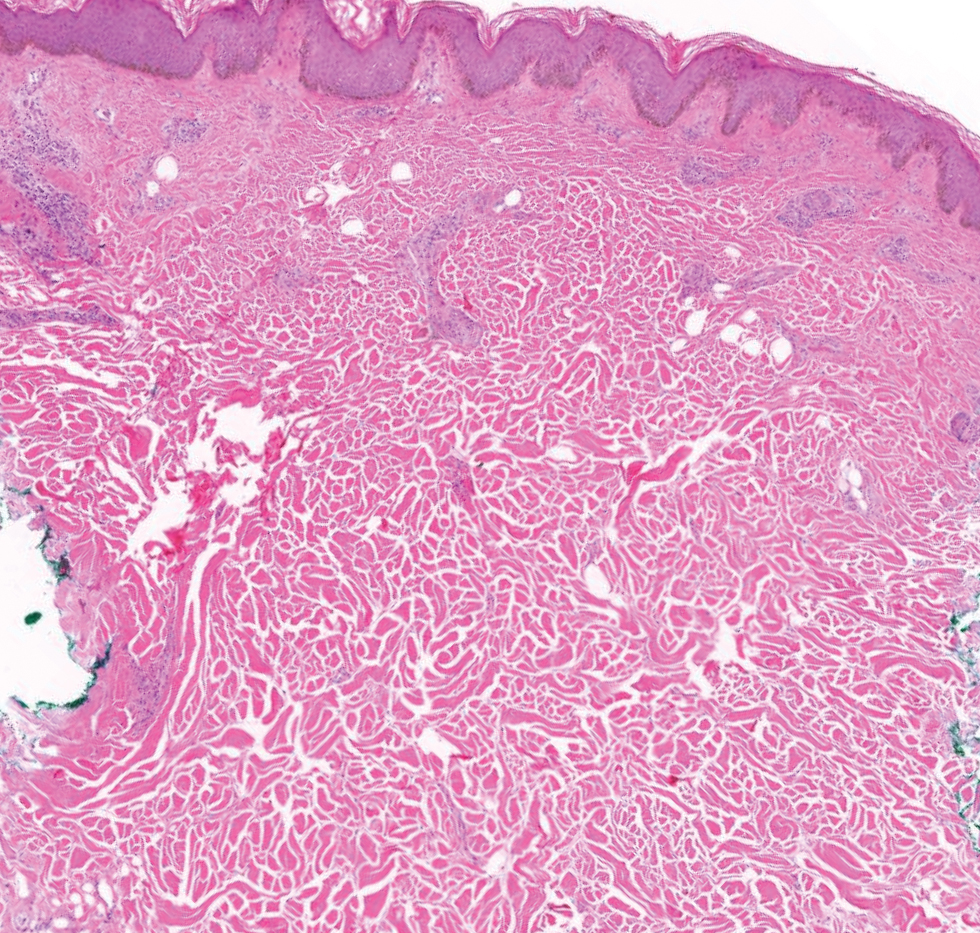
Papular granuloma annulare also was considered in our patient due to the presence of small flesh-colored papules. Histologically, granuloma annulare is characterized by palisading granulomas and mucin deposition in the dermis.10 However, the pattern of mucin deposition differs from that seen in APPM. In granuloma annulare, mucin is observed around foci of degenerated collagen (Figure 3), which was not observed in our patient.10 Additionally, the absence of an inflammatory infiltrate in our patient further ruled out this diagnosis.
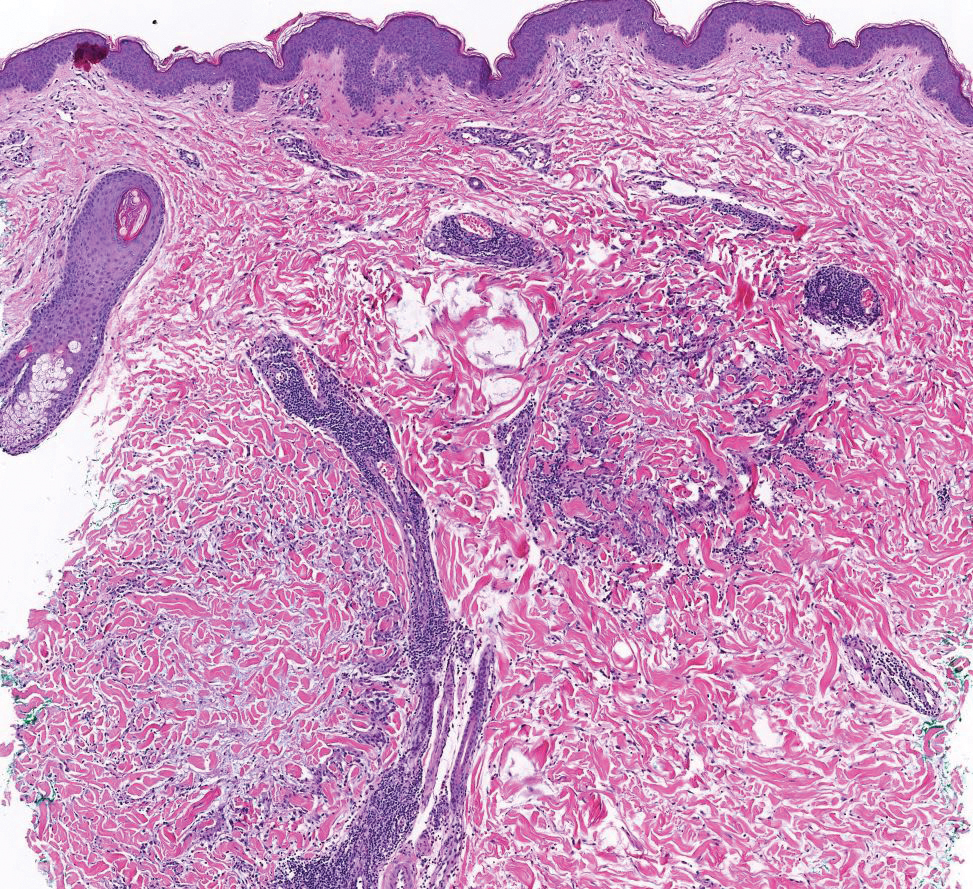
Lichen nitidus also could be considered in the differential diagnosis for ACCM. It typically manifests with minute, clustered, monomorphous papules with a predilection for the chest, abdomen, flexural forearms, and genitalia. The histology of lichen nitidus is distinct, showing a well-circumscribed lymphohistiocytic infiltrate in the papillary dermis bordered by epidermal ridges, resembling a ball and clutch appearance (Figure 4).11
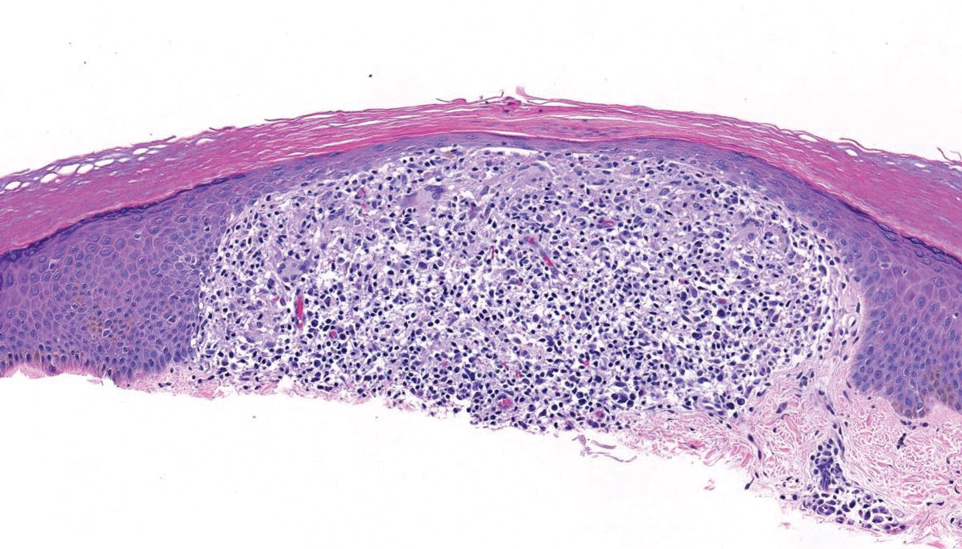
Although the clinical differential diagnosis in our patient was broad, histopathologic evaluation played a crucial role in confirming the diagnosis of APPM. This benign condition could be overlooked by patients and physicians; thorough clinical evaluation is necessary to rule out systemic mucinoses, which are associated with higher risks of morbidity and mortality.
- Rongioletti F, Rebora A. Acral persistent papular mucinosis: a new entity. Arch Dermatol. 1986;122:1237-1239. doi:10.1001 /archderm.1986.01660230027002
- Christman MP, Sukhdeo K, Kim RH, et al. Papular mucinosis, or localized lichen myxedematosus (LM)(discrete papular type). Dermatol Online J. 2017;23:13030/qt3xp109qd.
- Rongioletti F, Ferreli C, Atzori L. Acral persistent papular mucinosis. Clin Dermatol. 2021;39:211-214. doi:10.1016/j.clindermatol.2020.10.001
- Rongioletti F, Rebora A. Cutaneous mucinoses: microscopic criteria for diagnosis. Am J Dermatopathol. 2001;23:257-267. doi:10.1097/00000372- 200106000-00022
- Rongioletti F. Lichen myxedematosus (papular mucinosis): new concepts and perspectives for an old disease. Semin Cutan Med Surg. 2006;25:100-104. doi:10.1016/j.sder.2006.04.001
- Jun JY, Oh SH, Shim JH, et al. Acral persistent papular mucinosis with partial response to tacrolimus ointment. Ann Dermatol. 2016;28:517-519. doi:10.5021/ad.2016.28.4.517
- Rongioletti F, Zaccaria E, Cozzani E, et al. Treatment of localized lichen myxedematosus of discrete type with tacrolimus ointment. J Am Acad Dermatol. 2008;58:530-532. doi:10.1016/j.jaad.2006.10.021
- Rongioletti F, Merlo G, Cinotti E, et al. Scleromyxedema: a multicenter study of characteristics, comorbidities, course, and therapy in 30 patients. J Am Acad Dermatol. 2013;69:66-72. doi:10.1016 /j.jaad.2013.01.007
- Rongioletti F, Kaiser F, Cinotti E, et al. Scleredema. a multicentre study of characteristics, comorbidities, course and therapy in 44 patients. J Eur Acad Dermatol Venereol. 2015;29:2399-2404. doi:10.1111/jdv.13272
- Piette EW, Rosenbach M. Granuloma annulare: clinical and histologic variants, epidemiology, and genetics. J Am Acad Dermatol. 2016;75:457-465. doi:10.1016/j.jaad.2015.03.054
- Al-Mutairi N, Hassanein A, Nour-Eldin O, et al. Generalized lichen nitidus. Pediatr Dermatol. 2005;22:158-160. doi:10.1111 /j.1525-1470.2005.22215.x
THE DIAGNOSIS: Acral Persistent Papular Mucinosis
Histopathologic analysis revealed conspicuous interstitial mucin deposition throughout the upper to mid reticular dermis in the absence of a cellular infiltrate or fibroplasia. Colloidal iron staining confirmed the presence of mucin. In correlation with the clinical presentation, a diagnosis of acral persistent papular mucinosis (APPM) was made. The patient was counseled on the benign disease course and lack of associated comorbidities, and additional treatment was not pursued.
Acral persistent papular mucinosis is a rare distinct subtype of cutaneous mucinosis that initially was described by Rongioletti et al1 in 1986. As a localized form of lichen myxedematosus, APPM is characterized by mucin deposition in the dermis with no systemic involvement. The precise pathogenesis remains unclear, although some investigators have suggested that cytokine-mediated stimulation of glycosaminoglycan production may contribute to increased mucin accumulation in the dermis.2 Acral persistent papular mucinosis predominantly affects middle-aged women with a 5:1 female-to-male predominance.3 Clinically, patients present with discrete, nonfollicular, waxy papules that typically measure 2 to 5 mm and are distributed symmetrically on the extensor surfaces of the wrists and forearms. While the lesions generally are asymptomatic, some patients may report mild pruritus. The condition is chronic, with lesions seldom resolving and often increasing in number over time.3
Histologically, APPM is characterized by focal deposits of mucin in the upper reticular dermis with no evidence of increased fibroblast proliferation or fibrosis.4 This feature is pivotal in differentiating APPM from other subtypes of localized lichen myxedematosus and similar dermatoses. Diagnosis of APPM requires exclusion of systemic involvement, including thyroid abnormalities and monoclonal gammopathy, aligning with its classification as a purely cutaneous condition.5 Management of APPM is unclear due to its rarity. Reassurance for patients of its benign nature as well as clinical observation are recommended, though some reports cite benefits of treatment with topical corticosteroids or calcineurin inhibitors.6,7 The long-term prognosis for patients with APPM is favorable, although the persistence of and potential increase in lesions over time can be a cosmetic concern.
The differential diagnoses for APPM include scleromyxedema, scleredema, and other cutaneous eruptions that manifest as smooth flesh-colored papules, such as granuloma annulare and lichen nitidus.3 Scleromyxedema is a systemic cutaneous mucinosis that is part of the same disease spectrum as lichen myxedematosus. The papular eruption of scleromyxedema is much more widespread, and coalescing of the lesions may lead to characteristic skin thickening, creating leonine facies and deep furrowing over the trunk.8 Extracutaneous manifestations are frequent in scleromyxedema, and up to 90% of patients exhibit evidence of an underlying plasma cell dyscrasia.2 Histopathologically, scleromyxedema shows extensive fibroblast proliferation and fibrosis, in contrast to the findings of APPM (Figure 1).

The histopathology of APPM is most similar to scleredema, a rare fibromucinous disorder of the skin associated with diabetes, infection (especially poststreptococcal), or monoclonal gammopathy.9 Biopsy evaluation of scleredema reveals a normal epidermis with mucin deposition between collagen bundles predominantly in the deep reticular dermis as well as absent fibroblast proliferation (Figure 2). Unlike APPM, scleredema manifests with diffuse woody induration with erythema and hyperpigmentation on the posterior neck and upper back.9 On physical examination, the distinct clinical features of scleredema distinguish this condition from APPM and scleromyxedema.

Papular granuloma annulare also was considered in our patient due to the presence of small flesh-colored papules. Histologically, granuloma annulare is characterized by palisading granulomas and mucin deposition in the dermis.10 However, the pattern of mucin deposition differs from that seen in APPM. In granuloma annulare, mucin is observed around foci of degenerated collagen (Figure 3), which was not observed in our patient.10 Additionally, the absence of an inflammatory infiltrate in our patient further ruled out this diagnosis.

Lichen nitidus also could be considered in the differential diagnosis for ACCM. It typically manifests with minute, clustered, monomorphous papules with a predilection for the chest, abdomen, flexural forearms, and genitalia. The histology of lichen nitidus is distinct, showing a well-circumscribed lymphohistiocytic infiltrate in the papillary dermis bordered by epidermal ridges, resembling a ball and clutch appearance (Figure 4).11

Although the clinical differential diagnosis in our patient was broad, histopathologic evaluation played a crucial role in confirming the diagnosis of APPM. This benign condition could be overlooked by patients and physicians; thorough clinical evaluation is necessary to rule out systemic mucinoses, which are associated with higher risks of morbidity and mortality.
THE DIAGNOSIS: Acral Persistent Papular Mucinosis
Histopathologic analysis revealed conspicuous interstitial mucin deposition throughout the upper to mid reticular dermis in the absence of a cellular infiltrate or fibroplasia. Colloidal iron staining confirmed the presence of mucin. In correlation with the clinical presentation, a diagnosis of acral persistent papular mucinosis (APPM) was made. The patient was counseled on the benign disease course and lack of associated comorbidities, and additional treatment was not pursued.
Acral persistent papular mucinosis is a rare distinct subtype of cutaneous mucinosis that initially was described by Rongioletti et al1 in 1986. As a localized form of lichen myxedematosus, APPM is characterized by mucin deposition in the dermis with no systemic involvement. The precise pathogenesis remains unclear, although some investigators have suggested that cytokine-mediated stimulation of glycosaminoglycan production may contribute to increased mucin accumulation in the dermis.2 Acral persistent papular mucinosis predominantly affects middle-aged women with a 5:1 female-to-male predominance.3 Clinically, patients present with discrete, nonfollicular, waxy papules that typically measure 2 to 5 mm and are distributed symmetrically on the extensor surfaces of the wrists and forearms. While the lesions generally are asymptomatic, some patients may report mild pruritus. The condition is chronic, with lesions seldom resolving and often increasing in number over time.3
Histologically, APPM is characterized by focal deposits of mucin in the upper reticular dermis with no evidence of increased fibroblast proliferation or fibrosis.4 This feature is pivotal in differentiating APPM from other subtypes of localized lichen myxedematosus and similar dermatoses. Diagnosis of APPM requires exclusion of systemic involvement, including thyroid abnormalities and monoclonal gammopathy, aligning with its classification as a purely cutaneous condition.5 Management of APPM is unclear due to its rarity. Reassurance for patients of its benign nature as well as clinical observation are recommended, though some reports cite benefits of treatment with topical corticosteroids or calcineurin inhibitors.6,7 The long-term prognosis for patients with APPM is favorable, although the persistence of and potential increase in lesions over time can be a cosmetic concern.
The differential diagnoses for APPM include scleromyxedema, scleredema, and other cutaneous eruptions that manifest as smooth flesh-colored papules, such as granuloma annulare and lichen nitidus.3 Scleromyxedema is a systemic cutaneous mucinosis that is part of the same disease spectrum as lichen myxedematosus. The papular eruption of scleromyxedema is much more widespread, and coalescing of the lesions may lead to characteristic skin thickening, creating leonine facies and deep furrowing over the trunk.8 Extracutaneous manifestations are frequent in scleromyxedema, and up to 90% of patients exhibit evidence of an underlying plasma cell dyscrasia.2 Histopathologically, scleromyxedema shows extensive fibroblast proliferation and fibrosis, in contrast to the findings of APPM (Figure 1).

The histopathology of APPM is most similar to scleredema, a rare fibromucinous disorder of the skin associated with diabetes, infection (especially poststreptococcal), or monoclonal gammopathy.9 Biopsy evaluation of scleredema reveals a normal epidermis with mucin deposition between collagen bundles predominantly in the deep reticular dermis as well as absent fibroblast proliferation (Figure 2). Unlike APPM, scleredema manifests with diffuse woody induration with erythema and hyperpigmentation on the posterior neck and upper back.9 On physical examination, the distinct clinical features of scleredema distinguish this condition from APPM and scleromyxedema.

Papular granuloma annulare also was considered in our patient due to the presence of small flesh-colored papules. Histologically, granuloma annulare is characterized by palisading granulomas and mucin deposition in the dermis.10 However, the pattern of mucin deposition differs from that seen in APPM. In granuloma annulare, mucin is observed around foci of degenerated collagen (Figure 3), which was not observed in our patient.10 Additionally, the absence of an inflammatory infiltrate in our patient further ruled out this diagnosis.

Lichen nitidus also could be considered in the differential diagnosis for ACCM. It typically manifests with minute, clustered, monomorphous papules with a predilection for the chest, abdomen, flexural forearms, and genitalia. The histology of lichen nitidus is distinct, showing a well-circumscribed lymphohistiocytic infiltrate in the papillary dermis bordered by epidermal ridges, resembling a ball and clutch appearance (Figure 4).11

Although the clinical differential diagnosis in our patient was broad, histopathologic evaluation played a crucial role in confirming the diagnosis of APPM. This benign condition could be overlooked by patients and physicians; thorough clinical evaluation is necessary to rule out systemic mucinoses, which are associated with higher risks of morbidity and mortality.
- Rongioletti F, Rebora A. Acral persistent papular mucinosis: a new entity. Arch Dermatol. 1986;122:1237-1239. doi:10.1001 /archderm.1986.01660230027002
- Christman MP, Sukhdeo K, Kim RH, et al. Papular mucinosis, or localized lichen myxedematosus (LM)(discrete papular type). Dermatol Online J. 2017;23:13030/qt3xp109qd.
- Rongioletti F, Ferreli C, Atzori L. Acral persistent papular mucinosis. Clin Dermatol. 2021;39:211-214. doi:10.1016/j.clindermatol.2020.10.001
- Rongioletti F, Rebora A. Cutaneous mucinoses: microscopic criteria for diagnosis. Am J Dermatopathol. 2001;23:257-267. doi:10.1097/00000372- 200106000-00022
- Rongioletti F. Lichen myxedematosus (papular mucinosis): new concepts and perspectives for an old disease. Semin Cutan Med Surg. 2006;25:100-104. doi:10.1016/j.sder.2006.04.001
- Jun JY, Oh SH, Shim JH, et al. Acral persistent papular mucinosis with partial response to tacrolimus ointment. Ann Dermatol. 2016;28:517-519. doi:10.5021/ad.2016.28.4.517
- Rongioletti F, Zaccaria E, Cozzani E, et al. Treatment of localized lichen myxedematosus of discrete type with tacrolimus ointment. J Am Acad Dermatol. 2008;58:530-532. doi:10.1016/j.jaad.2006.10.021
- Rongioletti F, Merlo G, Cinotti E, et al. Scleromyxedema: a multicenter study of characteristics, comorbidities, course, and therapy in 30 patients. J Am Acad Dermatol. 2013;69:66-72. doi:10.1016 /j.jaad.2013.01.007
- Rongioletti F, Kaiser F, Cinotti E, et al. Scleredema. a multicentre study of characteristics, comorbidities, course and therapy in 44 patients. J Eur Acad Dermatol Venereol. 2015;29:2399-2404. doi:10.1111/jdv.13272
- Piette EW, Rosenbach M. Granuloma annulare: clinical and histologic variants, epidemiology, and genetics. J Am Acad Dermatol. 2016;75:457-465. doi:10.1016/j.jaad.2015.03.054
- Al-Mutairi N, Hassanein A, Nour-Eldin O, et al. Generalized lichen nitidus. Pediatr Dermatol. 2005;22:158-160. doi:10.1111 /j.1525-1470.2005.22215.x
- Rongioletti F, Rebora A. Acral persistent papular mucinosis: a new entity. Arch Dermatol. 1986;122:1237-1239. doi:10.1001 /archderm.1986.01660230027002
- Christman MP, Sukhdeo K, Kim RH, et al. Papular mucinosis, or localized lichen myxedematosus (LM)(discrete papular type). Dermatol Online J. 2017;23:13030/qt3xp109qd.
- Rongioletti F, Ferreli C, Atzori L. Acral persistent papular mucinosis. Clin Dermatol. 2021;39:211-214. doi:10.1016/j.clindermatol.2020.10.001
- Rongioletti F, Rebora A. Cutaneous mucinoses: microscopic criteria for diagnosis. Am J Dermatopathol. 2001;23:257-267. doi:10.1097/00000372- 200106000-00022
- Rongioletti F. Lichen myxedematosus (papular mucinosis): new concepts and perspectives for an old disease. Semin Cutan Med Surg. 2006;25:100-104. doi:10.1016/j.sder.2006.04.001
- Jun JY, Oh SH, Shim JH, et al. Acral persistent papular mucinosis with partial response to tacrolimus ointment. Ann Dermatol. 2016;28:517-519. doi:10.5021/ad.2016.28.4.517
- Rongioletti F, Zaccaria E, Cozzani E, et al. Treatment of localized lichen myxedematosus of discrete type with tacrolimus ointment. J Am Acad Dermatol. 2008;58:530-532. doi:10.1016/j.jaad.2006.10.021
- Rongioletti F, Merlo G, Cinotti E, et al. Scleromyxedema: a multicenter study of characteristics, comorbidities, course, and therapy in 30 patients. J Am Acad Dermatol. 2013;69:66-72. doi:10.1016 /j.jaad.2013.01.007
- Rongioletti F, Kaiser F, Cinotti E, et al. Scleredema. a multicentre study of characteristics, comorbidities, course and therapy in 44 patients. J Eur Acad Dermatol Venereol. 2015;29:2399-2404. doi:10.1111/jdv.13272
- Piette EW, Rosenbach M. Granuloma annulare: clinical and histologic variants, epidemiology, and genetics. J Am Acad Dermatol. 2016;75:457-465. doi:10.1016/j.jaad.2015.03.054
- Al-Mutairi N, Hassanein A, Nour-Eldin O, et al. Generalized lichen nitidus. Pediatr Dermatol. 2005;22:158-160. doi:10.1111 /j.1525-1470.2005.22215.x
Multiple Firm Papules on the Wrists and Forearms
Multiple Firm Papules on the Wrists and Forearms
A 69-year-old woman presented to the dermatology department with persistent asymptomatic skin lesions on the wrists and forearms of several months’ duration. The lesions had slowly grown in number over the past few months with no identifiable triggers. The patient reported no known history of injury or trauma to the affected sites and was not taking any prescription medications other than daily vitamins. She denied any family history of similar lesions and was otherwise healthy. Physical examination revealed multiple waxy, firm, hypopigmented, 3- to 5-mm papules located exclusively on the dorsal wrists and forearms. No extracutaneous involvement was observed. A 4-mm punch biopsy from the forearm was obtained.
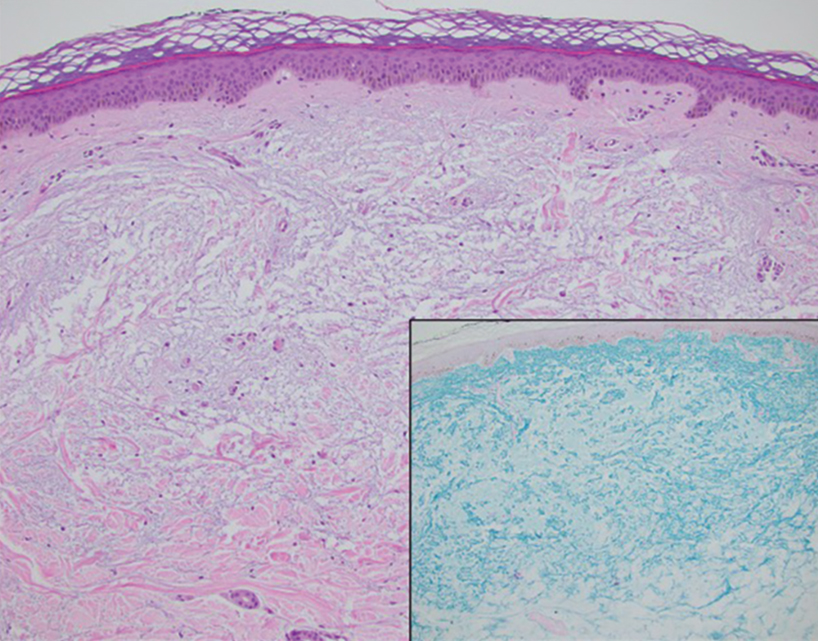
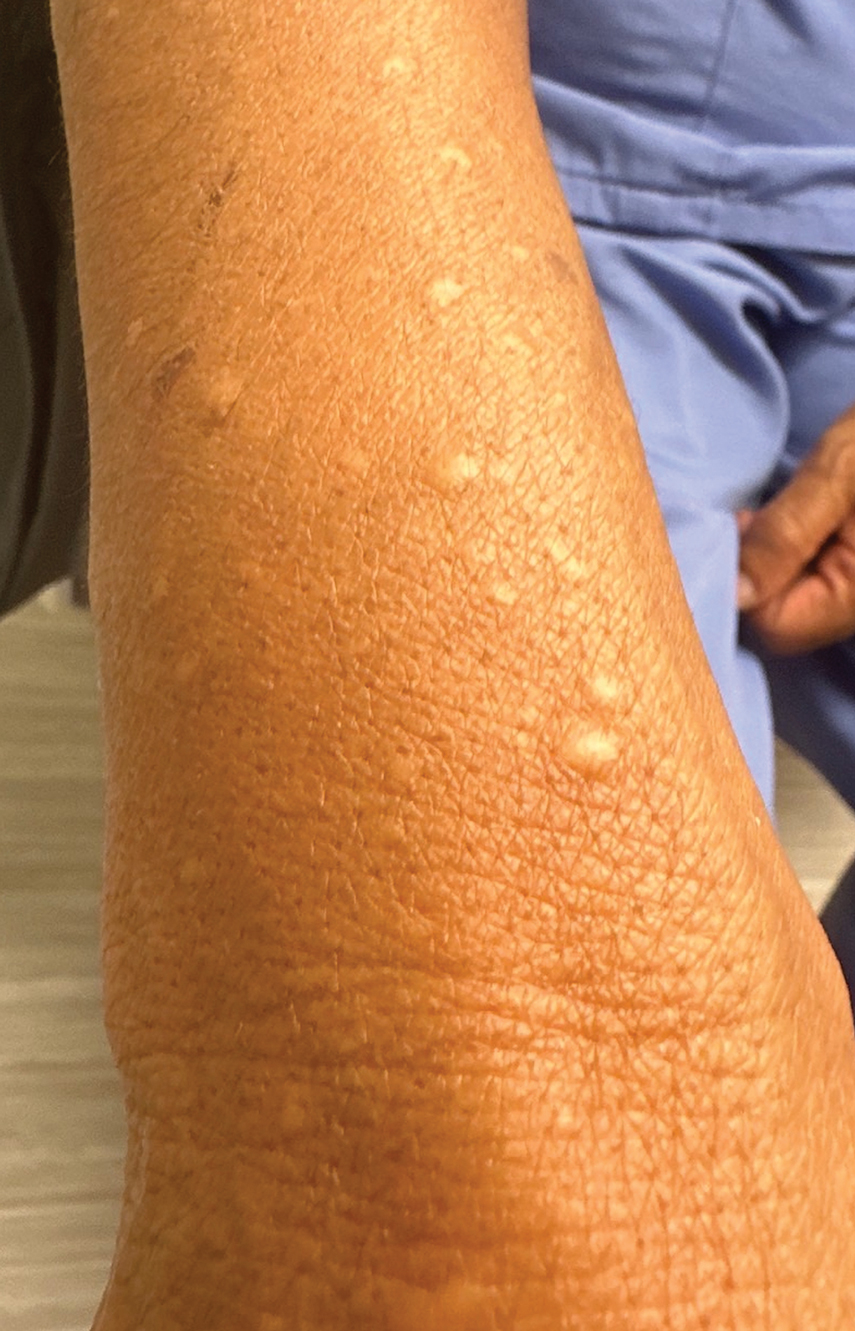
Bridging the Knowledge-Action Gap in Skin Cancer Prevention Among US Military Personnel
Bridging the Knowledge-Action Gap in Skin Cancer Prevention Among US Military Personnel
Skin cancer is a major health concern for military service members, who experience notably higher incidence rates than the general population.1 Active-duty military personnel are particularly vulnerable to prolonged sun exposure due to deployments, specialized training, and everyday outdoor duties.1 Despite skin cancer being the most commonly diagnosed malignancy in active-duty service members,2 tracking and documenting the quantity and diversity of these risk factors remain limited. This knowledge gap comes at high cost, simultaneously impairing military medicine preventive measures while burdening the military health care system with substantial expenditures.3 These findings underscore the critical need for targeted surveillance, early-detection programs, and policy-driven interventions to mitigate these medical and economic concerns.
Skin cancer has been recognized as a major health risk to the military population for decades, yet incidence and prevalence remain high. This phenomenon is closely linked to the inherent responsibilities and expectations of active-duty military members, including outdoor physical training, field exercises, standing in formation, and outdoor working environments—all of which can occur during peak sunlight hours. These risks are further elevated at duty stations in geographic regions with high levels of UV exposure, such as those in tropical and arid regions of the world. Certain military occupational specialties and missions may further introduce unique risk factors; for instance, pilots with frequent high-altitude missions experience heightened UV exposure and melanoma risk.4 Secondary to compounding determinants, the aviation, diving, and nuclear subgroups of the military community are particularly vulnerable to skin cancer.5
Despite well-documented risks, considerable gaps remain in quantifying and analyzing variations in UV exposure across military occupations, duty locations, and operational roles. Factors such as the existence of over 150 distinct military occupational specialties, frequent geographic relocations, and routine work in austere environments contribute to a wide range of UV exposure profiles that remain insufficiently characterized. This lack of comprehensive exposure data hinders the development of large-scale, targeted skin cancer prevention strategies. Initial approaches to addressing these challenges include enhanced surveillance, education, and policy initiatives. The Table presents practical recommendations for military leadership to consider in implementing preventive measures for skin cancer. Herein, we outline broader systemic strategies to bridge knowledge gaps and address underrecognized occupational risk factors for skin cancer in military service members; these elements include proposed modifications to the electronic Periodic Health Assessment (ePHA) and the development of standardized, military-specific screening and prevention guidelines to support early detection and resource optimization.
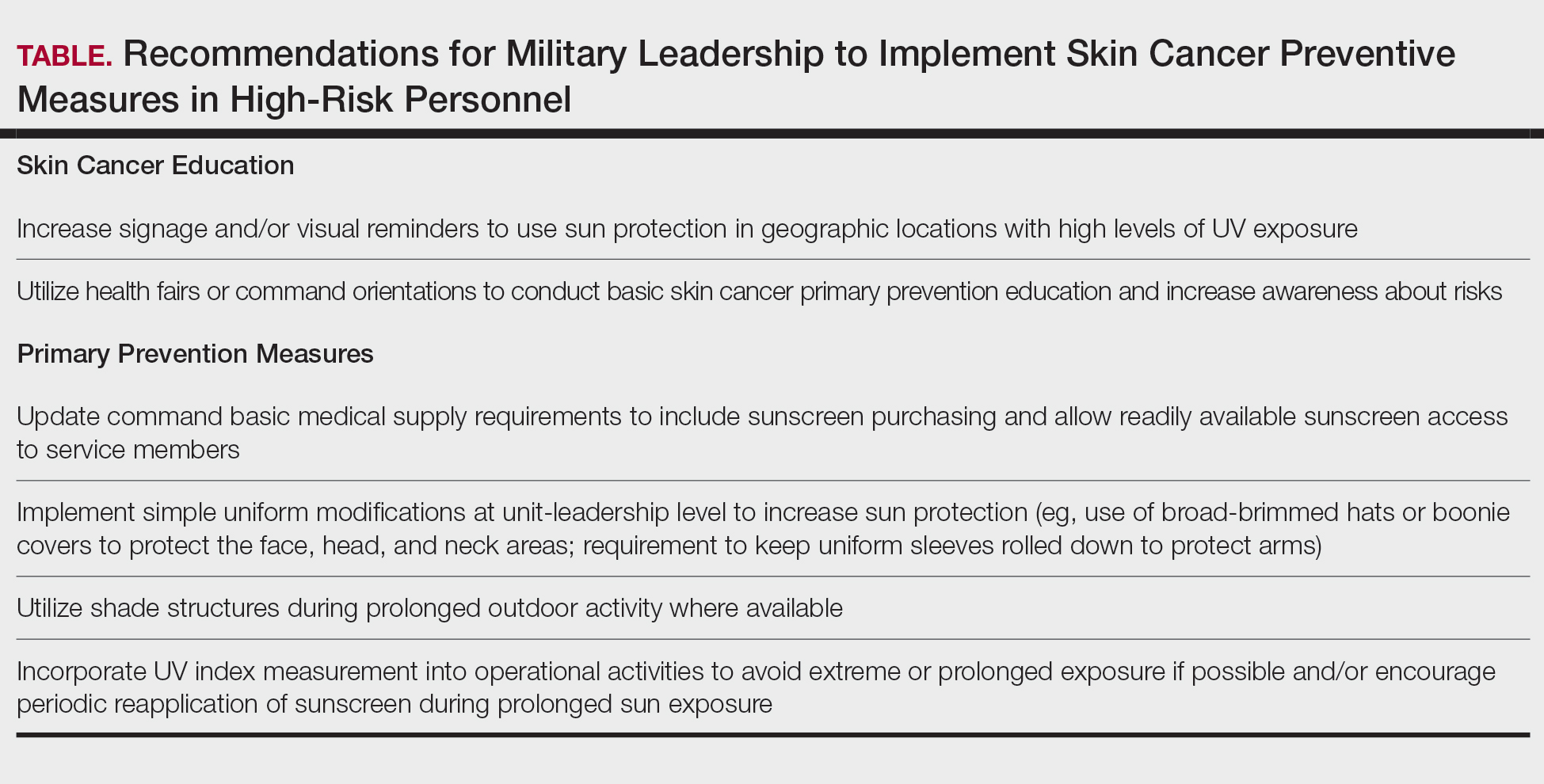
Skin Cancer Education for Service Members
Sunscreen and Signage—Diligent primary prevention offers a promising avenue for mitigating skin cancer incidence in military service members. Basic education and precautionary messaging on photoprotection can be widely implemented to simultaneously educate service members on the dangers of sun exposure while reinforcing healthy behaviors in real time. Simple low-cost initiatives such as strategically placed visual signage reminding service members to apply sunscreen in high UV environments can support consistent sun-safe practices. Educational efforts also should emphasize proper sunscreen use, including application on high-risk anatomic sites (eg, the face, neck, scalp, dorsal hands, and ears) and the essentiality of using sufficient quantities of broad-spectrum sunscreen for effective protection. Incorporating this guidance into training materials, briefings, and visual reminders allow seamless integration of photoprotection into service members’ daily routines without compromising operational efficiency.6 Younger service members, who may be less likely to prioritize preventive behaviors, may be particularly responsive to sun safety reminders in training areas, bases, and deployment zones.7 Health fairs and orientation briefs in high-UV regions also offer potential opportunities for targeted education.
Resources for Sun Protection in the Military
Sunscreen—Although sunscreen is critical in minimizing the risk for UV-induced skin cancer, its widespread use in the military is hindered by practical challenges related to accessibility and the need for consistent reapplication; for instance, providing free sunscreen dispensers at institutions for staff working under intense or prolonged UV exposure may improve sunscreen accessibility and use.8 Including sunscreen in standard-issue gear offers another logical way to embed its use into operational readiness as part of the routine protective measures.
Uniform Modifications—Adapting military uniforms and practices to improve sun protection plays a critical role in reducing skin cancer risk. Targeted protective gear for commonly sun-exposed areas can help mitigate UV exposure. One practical option is the use of wide-brimmed headgear (eg, boonie hats), which provide more face and neck coverage than standard-issue military caps, or covers. The wide-brimmed headgear currently is only selectively authorized during specific scenarios, such as field operations and training exercises, or at the discretion of unit-level leadership. Wide-brimmed headgear, already used by many service members, has been associated with up to a 17% reduction in UV exposure to inadequately protected areas, potentially lowering skin cancer risk.9,10 Similarly, a “sleeves-down” policy—requiring sleeves to remain unrolled and covering the forearms during outdoor activities—offers a simple way to minimize sun exposure without necessitating additional gear. Other specialized clothing items, including UV-blocking neck gaiters, photoprotective clothing, and lightweight gloves, also may be appropriate for high-risk groups and can be implemented in a relatively straightforward manner.
Shade Structures and UV Index Monitoring—Aside from uniform adaptation, physical barrier intervention can further complement skin cancer prevention efforts in the military. Shade structures offer a straightforward way to reduce UV exposure during prolonged outdoor activities. Incorporating daily UV index monitoring into operational guidance can help inform adjustments to training schedules and guide the implementation of additional sun protection measures, such as mandatory sunscreen application, use of wide-brimmed hats, or increased access to shaded rest areas during heavy sunlight hours. Currently, outdoor physical training is restricted during periods of high heat index, measured via Wet Bulb Globe Temperature, to reduce heat-related injuries. We argue that avoidance of nonoperational outdoor activity during peak UV index hours also should be incorporated into standardized policies. This intervention is of particular benefit to service members stationed in regions with a high UV index year-round, such as those stationed in the Middle East, Guam, Okinawa, and southern coastal United States bases.
Policy Changes to Support Photoprotective Measures
Annual Risk Factor Screening‐Screening—Effective secondary prevention efforts by military dermatologists remain an important measure in reducing the burden of skin cancer among military personnel; however, these efforts have become increasingly challenging due to 2 main factors—the diversity of military occupational specialties and their associated unique occupational risks as well as the limited availability of military dermatologists across all branches (approximately 100 active-duty dermatologists for nearly 3 million service members).11 Therefore, targeted interventions that enhance risk assessment, refined screening protocols, and leveraging of existing military health networks can improve early skin cancer detection while optimizing resource allocation.
The ePHA is an online screening tool used annually by all service members to evaluate their overall health. Presently, the ePHA lacks specific questions to assess sun exposure and skin cancer risks. Integrating annual skin cancer risk factor assessments into the ePHA would offer a practical and straightforward approach to identifying at-risk individuals, as suggested by Newnam et al12 in 2022. Skin cancer risk factor assessments allow for targeted data collection related to sun exposure history, family history, and personal risk factors, which can be used to determine individualized risk stratification to assess the need for early secondary prevention measures and specialist referral. These ePHA data can also support population-based analyses to inform preventive strategies and address knowledge gaps related to high-risk exposures, such as extended field exercises or assignments in high-UV regions, that may impede effective skin cancer prevention.
Development of Military-Specific Screening Guidelines—Given the limited number of military dermatologists, a standardized risk-assessment tool could enhance early detection of skin cancer and streamline the referral process. We propose a military-specific skin cancer screening algorithm or risk nomogram that could help to consolidate risk factors into a clear and actionable framework for more efficient triage and appropriate allocation of dermatologic resources and manpower. This nomogram could be developed by military dermatologists and then implemented on a command level, affording primary care providers a useful tool to expedite evaluation of individuals at higher risk for skin cancer while simultaneously promoting judicious use of limited dermatology resources.
Although the United States Preventive Services Task Force does not universally recommend routine skin cancer screenings for asymptomatic adults, military service members are exposed to higher occupational risks than the general population, as previously mentioned. Currently, there is no standardized screening guideline across all military services due to the unique nature and exposure risks for each branch of service and their varied occupations; however, we propose the development of basic standardized screening guidelines by adapting the framework of the United States Preventive Services Task Force and adjusting for military-specific UV exposure and occupational risks to improve early detection of skin cancer. These guidelines could be updated and tailored appropriately when additional population-based data are collected and analyzed through ePHA.
Critiques and Limitations of Implementation
Several challenges and limitations must be considered when attempting to integrate large-scale preventive measures for skin cancer within the US military. A primary concern is the extent to which military resources should be allocated to prevention when off-duty sun exposure remains largely beyond institutional control. Although military health initiatives can address workplace risk through education and policy, individual decisions during both work and leisure time remain a major variable that cannot be feasibly controlled. Cultural and operational barriers also pose challenges; for instance, the US Marine Corps maintains a strong cultural identity tied to uniform appearance, making it difficult to implement widespread changes to clothing-based sun-protection measures. Institutional changes, particularly those involving uniforms, likely will face substantial administrative resistance and potential operational limitations. When broad uniform modifications are unattainable, a more feasible approach may be to encourage unit-level leadership to authorize and promote the frequent use of nonuniform protective measures.
Furthermore, integrating additional skin cancer risk questions into the already extensive ePHA means extra time required to complete the assessment; this adds to service members’ administrative burden, potentially leading to reduced timely compliance, rushed responses, and survey fatigue, which threaten data quality. If new items are to be included, they should be carefully selected for efficiency and clinical relevance. Existing validated questionnaires such as those from the study by Lyford et al7 published in 2021 can serve as a foundation.
Another critical limitation is access to dermatologic care for active-duty service members. Raising awareness of skin cancer risk without ensuring adequate resources may create ethical concerns, particularly in high-risk environments such as the Middle East and Indo-Pacific. Additionally, because skin cancer often develops years or decades after exposure, securing early buy-in from service members and their leaders can be challenging. These concerns make it clear that, while skin cancer prevention is important, implementing widespread measures is not straightforward and requires a practical and balanced approach.
Final Thoughts
Implementing prevention strategies for skin cancer in the military requires balancing evidence-based recommendations with the practical realities of military culture, resource limitations, and operational demands. Challenges remain for dermatologists in providing targeted recommendations due to the multifaceted nature of military roles, including over 150 Navy Military Occupational Specialties, limited familiarity with the unique UV exposure risks associated with each occupation, and variability in local and regional policies on uniform wear, physical training requirements, and other operational practices. Although targeted prevention measures are difficult to establish in the setting of these knowledge gaps, leveraging unit-level leadership to align with existing screening guidelines and optimizing primary prevention measures can be meaningful steps toward reducing skin cancer risk for military service members while maintaining mission readiness.
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancerincidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Lee T, Taubman SB, Williams VF. Incident diagnoses of non-melanoma skin cancer, active component, U.S. Armed Forces, 2005-2014. MSMR. 2016;23:2-6.
- Krivda KR, Watson NL, Lyford WH, et al. The burden of skin cancer in the military health system, 2017-2022. Cutis. 2024;113:200-215. doi:10.12788/cutis.1015
- Sanlorenzo M, Wehner MR, Linos E, et al. The risk of melanoma in airline pilots and cabin crew: a meta-analysis. JAMA Dermatol. 2015;151:51-58. doi:10.1001/jamadermatol.2014.1077
- Brundage JF, Williams VF, Stahlman S, et al. Incidence rates of malignant melanoma in relation to years of military service, overall and in selected military occupational groups, active component, U.S. Armed Forces, 2001-2015. MMSR. 2017;24:8-14.
- Subramaniam P, Olsen CM, Thompson BS, et al, for the QSkin Sun and Health Study Investigators. Anatomical distributions of basal cell carcinoma and squamous cell carcinoma in a population-based study in Queensland, Australia. JAMA Dermatol. 2017;153:175-182. doi:10.1001/jamadermatol.2016.4070
- Lyford WH, Crotty A, Logemann NF. Sun exposure prevention practices within U.S. naval aviation. Mil Med. 2021;186:1169-1175. doi:10.1093/milmed/usab099
- Wood M, Raisanen T, Polcari I. Observational study of free public sunscreen dispenser use at a major US outdoor event. J Am Acad Dermatol. 2017;77:164-166.
- Schissel D. Operation shadow warrior: a quantitative analysis of the ultraviolet radiation protection demonstrated by various headgear. Mil Med. 2001;166:783-785.
- Milch JM, Logemann NF. Photoprotection prevents skin cancer: let’s make it fashionable to wear sun-protective clothing. Cutis. 2017;99:89-92.
- Association of Military Dermatologists. (n.d.). Military dermatology. https://militaryderm.org/military-dermatology/
- Newnam R, Le-Jenkins U, Rutledge C, et al. The association of skin cancer prevention knowledge, sun-protective attitudes, and sunprotective behaviors in a Navy population. Mil Med. 2024;189:1-7. doi:10.1093/milmed/usac285
Skin cancer is a major health concern for military service members, who experience notably higher incidence rates than the general population.1 Active-duty military personnel are particularly vulnerable to prolonged sun exposure due to deployments, specialized training, and everyday outdoor duties.1 Despite skin cancer being the most commonly diagnosed malignancy in active-duty service members,2 tracking and documenting the quantity and diversity of these risk factors remain limited. This knowledge gap comes at high cost, simultaneously impairing military medicine preventive measures while burdening the military health care system with substantial expenditures.3 These findings underscore the critical need for targeted surveillance, early-detection programs, and policy-driven interventions to mitigate these medical and economic concerns.
Skin cancer has been recognized as a major health risk to the military population for decades, yet incidence and prevalence remain high. This phenomenon is closely linked to the inherent responsibilities and expectations of active-duty military members, including outdoor physical training, field exercises, standing in formation, and outdoor working environments—all of which can occur during peak sunlight hours. These risks are further elevated at duty stations in geographic regions with high levels of UV exposure, such as those in tropical and arid regions of the world. Certain military occupational specialties and missions may further introduce unique risk factors; for instance, pilots with frequent high-altitude missions experience heightened UV exposure and melanoma risk.4 Secondary to compounding determinants, the aviation, diving, and nuclear subgroups of the military community are particularly vulnerable to skin cancer.5
Despite well-documented risks, considerable gaps remain in quantifying and analyzing variations in UV exposure across military occupations, duty locations, and operational roles. Factors such as the existence of over 150 distinct military occupational specialties, frequent geographic relocations, and routine work in austere environments contribute to a wide range of UV exposure profiles that remain insufficiently characterized. This lack of comprehensive exposure data hinders the development of large-scale, targeted skin cancer prevention strategies. Initial approaches to addressing these challenges include enhanced surveillance, education, and policy initiatives. The Table presents practical recommendations for military leadership to consider in implementing preventive measures for skin cancer. Herein, we outline broader systemic strategies to bridge knowledge gaps and address underrecognized occupational risk factors for skin cancer in military service members; these elements include proposed modifications to the electronic Periodic Health Assessment (ePHA) and the development of standardized, military-specific screening and prevention guidelines to support early detection and resource optimization.

Skin Cancer Education for Service Members
Sunscreen and Signage—Diligent primary prevention offers a promising avenue for mitigating skin cancer incidence in military service members. Basic education and precautionary messaging on photoprotection can be widely implemented to simultaneously educate service members on the dangers of sun exposure while reinforcing healthy behaviors in real time. Simple low-cost initiatives such as strategically placed visual signage reminding service members to apply sunscreen in high UV environments can support consistent sun-safe practices. Educational efforts also should emphasize proper sunscreen use, including application on high-risk anatomic sites (eg, the face, neck, scalp, dorsal hands, and ears) and the essentiality of using sufficient quantities of broad-spectrum sunscreen for effective protection. Incorporating this guidance into training materials, briefings, and visual reminders allow seamless integration of photoprotection into service members’ daily routines without compromising operational efficiency.6 Younger service members, who may be less likely to prioritize preventive behaviors, may be particularly responsive to sun safety reminders in training areas, bases, and deployment zones.7 Health fairs and orientation briefs in high-UV regions also offer potential opportunities for targeted education.
Resources for Sun Protection in the Military
Sunscreen—Although sunscreen is critical in minimizing the risk for UV-induced skin cancer, its widespread use in the military is hindered by practical challenges related to accessibility and the need for consistent reapplication; for instance, providing free sunscreen dispensers at institutions for staff working under intense or prolonged UV exposure may improve sunscreen accessibility and use.8 Including sunscreen in standard-issue gear offers another logical way to embed its use into operational readiness as part of the routine protective measures.
Uniform Modifications—Adapting military uniforms and practices to improve sun protection plays a critical role in reducing skin cancer risk. Targeted protective gear for commonly sun-exposed areas can help mitigate UV exposure. One practical option is the use of wide-brimmed headgear (eg, boonie hats), which provide more face and neck coverage than standard-issue military caps, or covers. The wide-brimmed headgear currently is only selectively authorized during specific scenarios, such as field operations and training exercises, or at the discretion of unit-level leadership. Wide-brimmed headgear, already used by many service members, has been associated with up to a 17% reduction in UV exposure to inadequately protected areas, potentially lowering skin cancer risk.9,10 Similarly, a “sleeves-down” policy—requiring sleeves to remain unrolled and covering the forearms during outdoor activities—offers a simple way to minimize sun exposure without necessitating additional gear. Other specialized clothing items, including UV-blocking neck gaiters, photoprotective clothing, and lightweight gloves, also may be appropriate for high-risk groups and can be implemented in a relatively straightforward manner.
Shade Structures and UV Index Monitoring—Aside from uniform adaptation, physical barrier intervention can further complement skin cancer prevention efforts in the military. Shade structures offer a straightforward way to reduce UV exposure during prolonged outdoor activities. Incorporating daily UV index monitoring into operational guidance can help inform adjustments to training schedules and guide the implementation of additional sun protection measures, such as mandatory sunscreen application, use of wide-brimmed hats, or increased access to shaded rest areas during heavy sunlight hours. Currently, outdoor physical training is restricted during periods of high heat index, measured via Wet Bulb Globe Temperature, to reduce heat-related injuries. We argue that avoidance of nonoperational outdoor activity during peak UV index hours also should be incorporated into standardized policies. This intervention is of particular benefit to service members stationed in regions with a high UV index year-round, such as those stationed in the Middle East, Guam, Okinawa, and southern coastal United States bases.
Policy Changes to Support Photoprotective Measures
Annual Risk Factor Screening‐Screening—Effective secondary prevention efforts by military dermatologists remain an important measure in reducing the burden of skin cancer among military personnel; however, these efforts have become increasingly challenging due to 2 main factors—the diversity of military occupational specialties and their associated unique occupational risks as well as the limited availability of military dermatologists across all branches (approximately 100 active-duty dermatologists for nearly 3 million service members).11 Therefore, targeted interventions that enhance risk assessment, refined screening protocols, and leveraging of existing military health networks can improve early skin cancer detection while optimizing resource allocation.
The ePHA is an online screening tool used annually by all service members to evaluate their overall health. Presently, the ePHA lacks specific questions to assess sun exposure and skin cancer risks. Integrating annual skin cancer risk factor assessments into the ePHA would offer a practical and straightforward approach to identifying at-risk individuals, as suggested by Newnam et al12 in 2022. Skin cancer risk factor assessments allow for targeted data collection related to sun exposure history, family history, and personal risk factors, which can be used to determine individualized risk stratification to assess the need for early secondary prevention measures and specialist referral. These ePHA data can also support population-based analyses to inform preventive strategies and address knowledge gaps related to high-risk exposures, such as extended field exercises or assignments in high-UV regions, that may impede effective skin cancer prevention.
Development of Military-Specific Screening Guidelines—Given the limited number of military dermatologists, a standardized risk-assessment tool could enhance early detection of skin cancer and streamline the referral process. We propose a military-specific skin cancer screening algorithm or risk nomogram that could help to consolidate risk factors into a clear and actionable framework for more efficient triage and appropriate allocation of dermatologic resources and manpower. This nomogram could be developed by military dermatologists and then implemented on a command level, affording primary care providers a useful tool to expedite evaluation of individuals at higher risk for skin cancer while simultaneously promoting judicious use of limited dermatology resources.
Although the United States Preventive Services Task Force does not universally recommend routine skin cancer screenings for asymptomatic adults, military service members are exposed to higher occupational risks than the general population, as previously mentioned. Currently, there is no standardized screening guideline across all military services due to the unique nature and exposure risks for each branch of service and their varied occupations; however, we propose the development of basic standardized screening guidelines by adapting the framework of the United States Preventive Services Task Force and adjusting for military-specific UV exposure and occupational risks to improve early detection of skin cancer. These guidelines could be updated and tailored appropriately when additional population-based data are collected and analyzed through ePHA.
Critiques and Limitations of Implementation
Several challenges and limitations must be considered when attempting to integrate large-scale preventive measures for skin cancer within the US military. A primary concern is the extent to which military resources should be allocated to prevention when off-duty sun exposure remains largely beyond institutional control. Although military health initiatives can address workplace risk through education and policy, individual decisions during both work and leisure time remain a major variable that cannot be feasibly controlled. Cultural and operational barriers also pose challenges; for instance, the US Marine Corps maintains a strong cultural identity tied to uniform appearance, making it difficult to implement widespread changes to clothing-based sun-protection measures. Institutional changes, particularly those involving uniforms, likely will face substantial administrative resistance and potential operational limitations. When broad uniform modifications are unattainable, a more feasible approach may be to encourage unit-level leadership to authorize and promote the frequent use of nonuniform protective measures.
Furthermore, integrating additional skin cancer risk questions into the already extensive ePHA means extra time required to complete the assessment; this adds to service members’ administrative burden, potentially leading to reduced timely compliance, rushed responses, and survey fatigue, which threaten data quality. If new items are to be included, they should be carefully selected for efficiency and clinical relevance. Existing validated questionnaires such as those from the study by Lyford et al7 published in 2021 can serve as a foundation.
Another critical limitation is access to dermatologic care for active-duty service members. Raising awareness of skin cancer risk without ensuring adequate resources may create ethical concerns, particularly in high-risk environments such as the Middle East and Indo-Pacific. Additionally, because skin cancer often develops years or decades after exposure, securing early buy-in from service members and their leaders can be challenging. These concerns make it clear that, while skin cancer prevention is important, implementing widespread measures is not straightforward and requires a practical and balanced approach.
Final Thoughts
Implementing prevention strategies for skin cancer in the military requires balancing evidence-based recommendations with the practical realities of military culture, resource limitations, and operational demands. Challenges remain for dermatologists in providing targeted recommendations due to the multifaceted nature of military roles, including over 150 Navy Military Occupational Specialties, limited familiarity with the unique UV exposure risks associated with each occupation, and variability in local and regional policies on uniform wear, physical training requirements, and other operational practices. Although targeted prevention measures are difficult to establish in the setting of these knowledge gaps, leveraging unit-level leadership to align with existing screening guidelines and optimizing primary prevention measures can be meaningful steps toward reducing skin cancer risk for military service members while maintaining mission readiness.
Skin cancer is a major health concern for military service members, who experience notably higher incidence rates than the general population.1 Active-duty military personnel are particularly vulnerable to prolonged sun exposure due to deployments, specialized training, and everyday outdoor duties.1 Despite skin cancer being the most commonly diagnosed malignancy in active-duty service members,2 tracking and documenting the quantity and diversity of these risk factors remain limited. This knowledge gap comes at high cost, simultaneously impairing military medicine preventive measures while burdening the military health care system with substantial expenditures.3 These findings underscore the critical need for targeted surveillance, early-detection programs, and policy-driven interventions to mitigate these medical and economic concerns.
Skin cancer has been recognized as a major health risk to the military population for decades, yet incidence and prevalence remain high. This phenomenon is closely linked to the inherent responsibilities and expectations of active-duty military members, including outdoor physical training, field exercises, standing in formation, and outdoor working environments—all of which can occur during peak sunlight hours. These risks are further elevated at duty stations in geographic regions with high levels of UV exposure, such as those in tropical and arid regions of the world. Certain military occupational specialties and missions may further introduce unique risk factors; for instance, pilots with frequent high-altitude missions experience heightened UV exposure and melanoma risk.4 Secondary to compounding determinants, the aviation, diving, and nuclear subgroups of the military community are particularly vulnerable to skin cancer.5
Despite well-documented risks, considerable gaps remain in quantifying and analyzing variations in UV exposure across military occupations, duty locations, and operational roles. Factors such as the existence of over 150 distinct military occupational specialties, frequent geographic relocations, and routine work in austere environments contribute to a wide range of UV exposure profiles that remain insufficiently characterized. This lack of comprehensive exposure data hinders the development of large-scale, targeted skin cancer prevention strategies. Initial approaches to addressing these challenges include enhanced surveillance, education, and policy initiatives. The Table presents practical recommendations for military leadership to consider in implementing preventive measures for skin cancer. Herein, we outline broader systemic strategies to bridge knowledge gaps and address underrecognized occupational risk factors for skin cancer in military service members; these elements include proposed modifications to the electronic Periodic Health Assessment (ePHA) and the development of standardized, military-specific screening and prevention guidelines to support early detection and resource optimization.

Skin Cancer Education for Service Members
Sunscreen and Signage—Diligent primary prevention offers a promising avenue for mitigating skin cancer incidence in military service members. Basic education and precautionary messaging on photoprotection can be widely implemented to simultaneously educate service members on the dangers of sun exposure while reinforcing healthy behaviors in real time. Simple low-cost initiatives such as strategically placed visual signage reminding service members to apply sunscreen in high UV environments can support consistent sun-safe practices. Educational efforts also should emphasize proper sunscreen use, including application on high-risk anatomic sites (eg, the face, neck, scalp, dorsal hands, and ears) and the essentiality of using sufficient quantities of broad-spectrum sunscreen for effective protection. Incorporating this guidance into training materials, briefings, and visual reminders allow seamless integration of photoprotection into service members’ daily routines without compromising operational efficiency.6 Younger service members, who may be less likely to prioritize preventive behaviors, may be particularly responsive to sun safety reminders in training areas, bases, and deployment zones.7 Health fairs and orientation briefs in high-UV regions also offer potential opportunities for targeted education.
Resources for Sun Protection in the Military
Sunscreen—Although sunscreen is critical in minimizing the risk for UV-induced skin cancer, its widespread use in the military is hindered by practical challenges related to accessibility and the need for consistent reapplication; for instance, providing free sunscreen dispensers at institutions for staff working under intense or prolonged UV exposure may improve sunscreen accessibility and use.8 Including sunscreen in standard-issue gear offers another logical way to embed its use into operational readiness as part of the routine protective measures.
Uniform Modifications—Adapting military uniforms and practices to improve sun protection plays a critical role in reducing skin cancer risk. Targeted protective gear for commonly sun-exposed areas can help mitigate UV exposure. One practical option is the use of wide-brimmed headgear (eg, boonie hats), which provide more face and neck coverage than standard-issue military caps, or covers. The wide-brimmed headgear currently is only selectively authorized during specific scenarios, such as field operations and training exercises, or at the discretion of unit-level leadership. Wide-brimmed headgear, already used by many service members, has been associated with up to a 17% reduction in UV exposure to inadequately protected areas, potentially lowering skin cancer risk.9,10 Similarly, a “sleeves-down” policy—requiring sleeves to remain unrolled and covering the forearms during outdoor activities—offers a simple way to minimize sun exposure without necessitating additional gear. Other specialized clothing items, including UV-blocking neck gaiters, photoprotective clothing, and lightweight gloves, also may be appropriate for high-risk groups and can be implemented in a relatively straightforward manner.
Shade Structures and UV Index Monitoring—Aside from uniform adaptation, physical barrier intervention can further complement skin cancer prevention efforts in the military. Shade structures offer a straightforward way to reduce UV exposure during prolonged outdoor activities. Incorporating daily UV index monitoring into operational guidance can help inform adjustments to training schedules and guide the implementation of additional sun protection measures, such as mandatory sunscreen application, use of wide-brimmed hats, or increased access to shaded rest areas during heavy sunlight hours. Currently, outdoor physical training is restricted during periods of high heat index, measured via Wet Bulb Globe Temperature, to reduce heat-related injuries. We argue that avoidance of nonoperational outdoor activity during peak UV index hours also should be incorporated into standardized policies. This intervention is of particular benefit to service members stationed in regions with a high UV index year-round, such as those stationed in the Middle East, Guam, Okinawa, and southern coastal United States bases.
Policy Changes to Support Photoprotective Measures
Annual Risk Factor Screening‐Screening—Effective secondary prevention efforts by military dermatologists remain an important measure in reducing the burden of skin cancer among military personnel; however, these efforts have become increasingly challenging due to 2 main factors—the diversity of military occupational specialties and their associated unique occupational risks as well as the limited availability of military dermatologists across all branches (approximately 100 active-duty dermatologists for nearly 3 million service members).11 Therefore, targeted interventions that enhance risk assessment, refined screening protocols, and leveraging of existing military health networks can improve early skin cancer detection while optimizing resource allocation.
The ePHA is an online screening tool used annually by all service members to evaluate their overall health. Presently, the ePHA lacks specific questions to assess sun exposure and skin cancer risks. Integrating annual skin cancer risk factor assessments into the ePHA would offer a practical and straightforward approach to identifying at-risk individuals, as suggested by Newnam et al12 in 2022. Skin cancer risk factor assessments allow for targeted data collection related to sun exposure history, family history, and personal risk factors, which can be used to determine individualized risk stratification to assess the need for early secondary prevention measures and specialist referral. These ePHA data can also support population-based analyses to inform preventive strategies and address knowledge gaps related to high-risk exposures, such as extended field exercises or assignments in high-UV regions, that may impede effective skin cancer prevention.
Development of Military-Specific Screening Guidelines—Given the limited number of military dermatologists, a standardized risk-assessment tool could enhance early detection of skin cancer and streamline the referral process. We propose a military-specific skin cancer screening algorithm or risk nomogram that could help to consolidate risk factors into a clear and actionable framework for more efficient triage and appropriate allocation of dermatologic resources and manpower. This nomogram could be developed by military dermatologists and then implemented on a command level, affording primary care providers a useful tool to expedite evaluation of individuals at higher risk for skin cancer while simultaneously promoting judicious use of limited dermatology resources.
Although the United States Preventive Services Task Force does not universally recommend routine skin cancer screenings for asymptomatic adults, military service members are exposed to higher occupational risks than the general population, as previously mentioned. Currently, there is no standardized screening guideline across all military services due to the unique nature and exposure risks for each branch of service and their varied occupations; however, we propose the development of basic standardized screening guidelines by adapting the framework of the United States Preventive Services Task Force and adjusting for military-specific UV exposure and occupational risks to improve early detection of skin cancer. These guidelines could be updated and tailored appropriately when additional population-based data are collected and analyzed through ePHA.
Critiques and Limitations of Implementation
Several challenges and limitations must be considered when attempting to integrate large-scale preventive measures for skin cancer within the US military. A primary concern is the extent to which military resources should be allocated to prevention when off-duty sun exposure remains largely beyond institutional control. Although military health initiatives can address workplace risk through education and policy, individual decisions during both work and leisure time remain a major variable that cannot be feasibly controlled. Cultural and operational barriers also pose challenges; for instance, the US Marine Corps maintains a strong cultural identity tied to uniform appearance, making it difficult to implement widespread changes to clothing-based sun-protection measures. Institutional changes, particularly those involving uniforms, likely will face substantial administrative resistance and potential operational limitations. When broad uniform modifications are unattainable, a more feasible approach may be to encourage unit-level leadership to authorize and promote the frequent use of nonuniform protective measures.
Furthermore, integrating additional skin cancer risk questions into the already extensive ePHA means extra time required to complete the assessment; this adds to service members’ administrative burden, potentially leading to reduced timely compliance, rushed responses, and survey fatigue, which threaten data quality. If new items are to be included, they should be carefully selected for efficiency and clinical relevance. Existing validated questionnaires such as those from the study by Lyford et al7 published in 2021 can serve as a foundation.
Another critical limitation is access to dermatologic care for active-duty service members. Raising awareness of skin cancer risk without ensuring adequate resources may create ethical concerns, particularly in high-risk environments such as the Middle East and Indo-Pacific. Additionally, because skin cancer often develops years or decades after exposure, securing early buy-in from service members and their leaders can be challenging. These concerns make it clear that, while skin cancer prevention is important, implementing widespread measures is not straightforward and requires a practical and balanced approach.
Final Thoughts
Implementing prevention strategies for skin cancer in the military requires balancing evidence-based recommendations with the practical realities of military culture, resource limitations, and operational demands. Challenges remain for dermatologists in providing targeted recommendations due to the multifaceted nature of military roles, including over 150 Navy Military Occupational Specialties, limited familiarity with the unique UV exposure risks associated with each occupation, and variability in local and regional policies on uniform wear, physical training requirements, and other operational practices. Although targeted prevention measures are difficult to establish in the setting of these knowledge gaps, leveraging unit-level leadership to align with existing screening guidelines and optimizing primary prevention measures can be meaningful steps toward reducing skin cancer risk for military service members while maintaining mission readiness.
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancerincidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Lee T, Taubman SB, Williams VF. Incident diagnoses of non-melanoma skin cancer, active component, U.S. Armed Forces, 2005-2014. MSMR. 2016;23:2-6.
- Krivda KR, Watson NL, Lyford WH, et al. The burden of skin cancer in the military health system, 2017-2022. Cutis. 2024;113:200-215. doi:10.12788/cutis.1015
- Sanlorenzo M, Wehner MR, Linos E, et al. The risk of melanoma in airline pilots and cabin crew: a meta-analysis. JAMA Dermatol. 2015;151:51-58. doi:10.1001/jamadermatol.2014.1077
- Brundage JF, Williams VF, Stahlman S, et al. Incidence rates of malignant melanoma in relation to years of military service, overall and in selected military occupational groups, active component, U.S. Armed Forces, 2001-2015. MMSR. 2017;24:8-14.
- Subramaniam P, Olsen CM, Thompson BS, et al, for the QSkin Sun and Health Study Investigators. Anatomical distributions of basal cell carcinoma and squamous cell carcinoma in a population-based study in Queensland, Australia. JAMA Dermatol. 2017;153:175-182. doi:10.1001/jamadermatol.2016.4070
- Lyford WH, Crotty A, Logemann NF. Sun exposure prevention practices within U.S. naval aviation. Mil Med. 2021;186:1169-1175. doi:10.1093/milmed/usab099
- Wood M, Raisanen T, Polcari I. Observational study of free public sunscreen dispenser use at a major US outdoor event. J Am Acad Dermatol. 2017;77:164-166.
- Schissel D. Operation shadow warrior: a quantitative analysis of the ultraviolet radiation protection demonstrated by various headgear. Mil Med. 2001;166:783-785.
- Milch JM, Logemann NF. Photoprotection prevents skin cancer: let’s make it fashionable to wear sun-protective clothing. Cutis. 2017;99:89-92.
- Association of Military Dermatologists. (n.d.). Military dermatology. https://militaryderm.org/military-dermatology/
- Newnam R, Le-Jenkins U, Rutledge C, et al. The association of skin cancer prevention knowledge, sun-protective attitudes, and sunprotective behaviors in a Navy population. Mil Med. 2024;189:1-7. doi:10.1093/milmed/usac285
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancerincidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Lee T, Taubman SB, Williams VF. Incident diagnoses of non-melanoma skin cancer, active component, U.S. Armed Forces, 2005-2014. MSMR. 2016;23:2-6.
- Krivda KR, Watson NL, Lyford WH, et al. The burden of skin cancer in the military health system, 2017-2022. Cutis. 2024;113:200-215. doi:10.12788/cutis.1015
- Sanlorenzo M, Wehner MR, Linos E, et al. The risk of melanoma in airline pilots and cabin crew: a meta-analysis. JAMA Dermatol. 2015;151:51-58. doi:10.1001/jamadermatol.2014.1077
- Brundage JF, Williams VF, Stahlman S, et al. Incidence rates of malignant melanoma in relation to years of military service, overall and in selected military occupational groups, active component, U.S. Armed Forces, 2001-2015. MMSR. 2017;24:8-14.
- Subramaniam P, Olsen CM, Thompson BS, et al, for the QSkin Sun and Health Study Investigators. Anatomical distributions of basal cell carcinoma and squamous cell carcinoma in a population-based study in Queensland, Australia. JAMA Dermatol. 2017;153:175-182. doi:10.1001/jamadermatol.2016.4070
- Lyford WH, Crotty A, Logemann NF. Sun exposure prevention practices within U.S. naval aviation. Mil Med. 2021;186:1169-1175. doi:10.1093/milmed/usab099
- Wood M, Raisanen T, Polcari I. Observational study of free public sunscreen dispenser use at a major US outdoor event. J Am Acad Dermatol. 2017;77:164-166.
- Schissel D. Operation shadow warrior: a quantitative analysis of the ultraviolet radiation protection demonstrated by various headgear. Mil Med. 2001;166:783-785.
- Milch JM, Logemann NF. Photoprotection prevents skin cancer: let’s make it fashionable to wear sun-protective clothing. Cutis. 2017;99:89-92.
- Association of Military Dermatologists. (n.d.). Military dermatology. https://militaryderm.org/military-dermatology/
- Newnam R, Le-Jenkins U, Rutledge C, et al. The association of skin cancer prevention knowledge, sun-protective attitudes, and sunprotective behaviors in a Navy population. Mil Med. 2024;189:1-7. doi:10.1093/milmed/usac285
Bridging the Knowledge-Action Gap in Skin Cancer Prevention Among US Military Personnel
Bridging the Knowledge-Action Gap in Skin Cancer Prevention Among US Military Personnel
PRACTICE POINTS
- Military personnel face elevated skin cancer risks due to prolonged occupational UV exposure.
- Medical providers can partner with unit-level leadership to implement low-cost interventions such as shade structures and uniform modifications.
- Annual sun exposure risk assessments should be integrated into the military Electronic Periodic Health Assessment for targeted screening and early intervention of risk factors.
- Photoprotective gear and signage in high—UV index areas can improve service member awareness and adherence to preventive measures.
Gastroenterology Data Trends 2025
Gastroenterology Data Trends 2025
GI & Hepatology News and the American Gastroenterological Association (AGA) present Gastroenterology Data Trends 2025, a special report on hot topics in GI told through original infographics and visual storytelling.
In this issue:
The Role of Bedside Intestinal Ultrasound in IBD Management
Bincy Abraham, MD, MS
Obesity Management in the Era of GLP-1: The Role of GLP-1 RAs
Michael Camilleri, MD, MPhil, DSc
Ergonomics in Endoscopy
Amandeep K. Shergill, MD, MS
Optimizing the Delivery of GI Care in Transgender and Gender-Diverse Communities
Kira Newman, MD, PhD
New Therapeutic Frontiers in the Treatment of Eosinophilic Esophagitis
Evan S. Dellon, MD, MPH
New and Emerging Treatments for MASLD/MASH
Naim Alkhouri, MD
Advances in Screening for Barrett’s Esophagus and Esophageal Adenocarcinoma
Joel Rubenstein, MD, MS
Alagille Syndrome: Epidemiology and Management of a Rare Genetic Disease
Alisha Mavis, MD
IBS: Mental Health Factors and Comorbidities
Lin Chang, MD, and Laurie A. Keefer, PhD
GI & Hepatology News and the American Gastroenterological Association (AGA) present Gastroenterology Data Trends 2025, a special report on hot topics in GI told through original infographics and visual storytelling.
In this issue:
The Role of Bedside Intestinal Ultrasound in IBD Management
Bincy Abraham, MD, MS
Obesity Management in the Era of GLP-1: The Role of GLP-1 RAs
Michael Camilleri, MD, MPhil, DSc
Ergonomics in Endoscopy
Amandeep K. Shergill, MD, MS
Optimizing the Delivery of GI Care in Transgender and Gender-Diverse Communities
Kira Newman, MD, PhD
New Therapeutic Frontiers in the Treatment of Eosinophilic Esophagitis
Evan S. Dellon, MD, MPH
New and Emerging Treatments for MASLD/MASH
Naim Alkhouri, MD
Advances in Screening for Barrett’s Esophagus and Esophageal Adenocarcinoma
Joel Rubenstein, MD, MS
Alagille Syndrome: Epidemiology and Management of a Rare Genetic Disease
Alisha Mavis, MD
IBS: Mental Health Factors and Comorbidities
Lin Chang, MD, and Laurie A. Keefer, PhD
GI & Hepatology News and the American Gastroenterological Association (AGA) present Gastroenterology Data Trends 2025, a special report on hot topics in GI told through original infographics and visual storytelling.
In this issue:
The Role of Bedside Intestinal Ultrasound in IBD Management
Bincy Abraham, MD, MS
Obesity Management in the Era of GLP-1: The Role of GLP-1 RAs
Michael Camilleri, MD, MPhil, DSc
Ergonomics in Endoscopy
Amandeep K. Shergill, MD, MS
Optimizing the Delivery of GI Care in Transgender and Gender-Diverse Communities
Kira Newman, MD, PhD
New Therapeutic Frontiers in the Treatment of Eosinophilic Esophagitis
Evan S. Dellon, MD, MPH
New and Emerging Treatments for MASLD/MASH
Naim Alkhouri, MD
Advances in Screening for Barrett’s Esophagus and Esophageal Adenocarcinoma
Joel Rubenstein, MD, MS
Alagille Syndrome: Epidemiology and Management of a Rare Genetic Disease
Alisha Mavis, MD
IBS: Mental Health Factors and Comorbidities
Lin Chang, MD, and Laurie A. Keefer, PhD
Gastroenterology Data Trends 2025
Gastroenterology Data Trends 2025
Repair of a Large Full-Thickness Conchal Bowl Defect
Repair of a Large Full-Thickness Conchal Bowl Defect
Practice Gap
Large full-thickness conchal bowl defects often pose a reconstructive challenge. Maintaining the shape and structural integrity of the concha is fundamental for optimal cosmetic and functional outcomes. Prior reports have suggested wedge excisions, composite grafts, interpolation flaps with or without cartilage struts, and hinge flaps as possible options for reconstruction.1-3 However, patients with large defects who prefer single-stage reconstruction procedures present a unique challenge. Herein, we describe a single-stage full-thickness hinge flap technique for a large conchal bowl defect.
The Technique
A 77-year-old man was referred to our dermatology clinic by an outside dermatologist for Mohs micrographic surgery of a biopsy-proven cutaneous squamous cell carcinoma on the right conchal bowl measuring 1.1×2.1 cm and extending to the edge of the external auditory canal (EAC). The excision was performed that same day and was completed in 2 stages, achieving negative margins and resulting in a full-thickness defect measuring 2.0×3.6 cm that included the posterior auricular sulcus, cavum, antitragus, and proximal EAC (Figure 1). The patient requested a single-stage procedure but emphasized that his main priority was an optimal cosmetic outcome.
To repair this large defect, a full-thickness hinge flap with Burow graft was performed. The hinge-type flap was designed in a triangular fashion emanating at the posterior auricular sulcus adjacent to the posterior aspect of the defect and extending down the lateral neck (Figure 2). The flap was incised and the surrounding tissue was undermined, maintaining a robust pedicle in the center of its body on the superolateral neck. The flap was passed through the posterior aspect of the full-thickness defect and was secured in place with 4-0 polyglactin sutures in a buried interrupted fashion, thereby recreating the anterior portion of the defect. The superficial skin edges were reapproximated using 4-0 and 5-0 polypropylene sutures in a running interrupted fashion. The distal Burow triangle created from closure of the flap’s secondary defect was aggressively thinned and was utilized as a full-thickness graft for the residual postauricular groove defect (Figure 3). At 2 weeks’ follow-up, the patient was healing well with no postoperative issues and the sutures were removed (Figure 4).
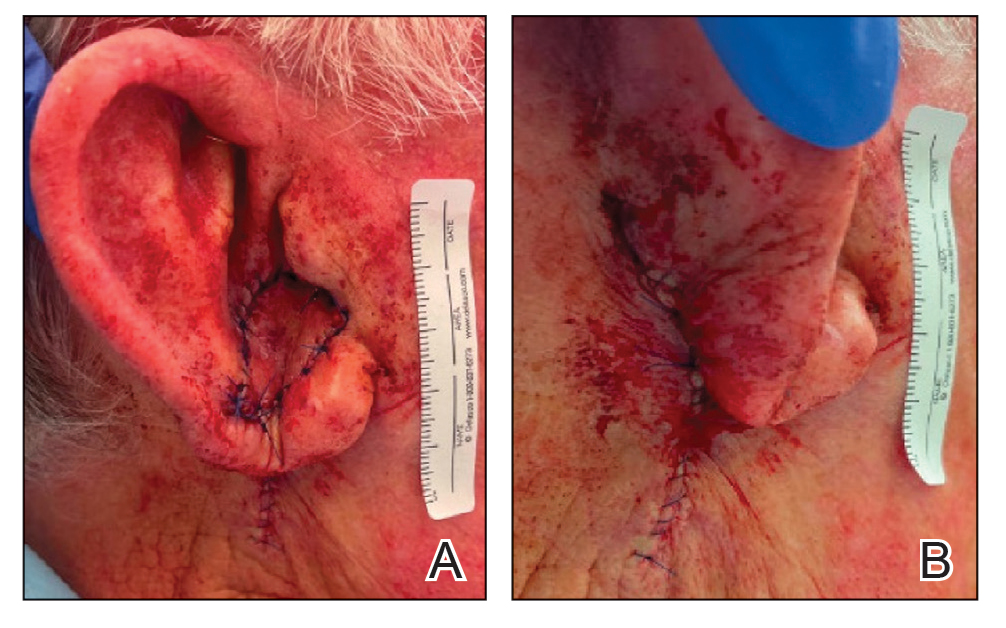
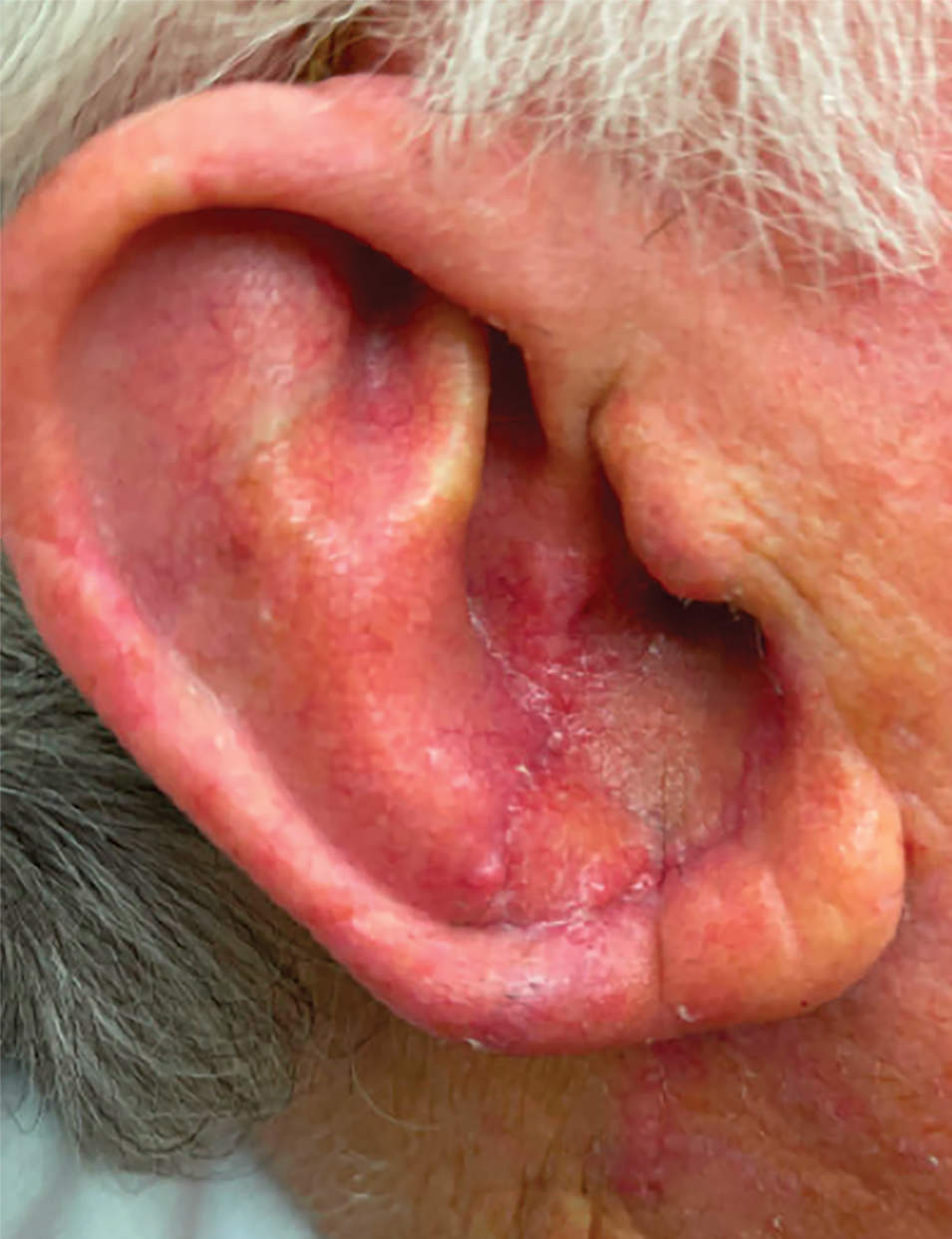
Practice Implications
There are many different reconstructive options for conchal bowl defects, including primary repair, wedge excision, composite graft and interpolation flaps with or without cartilage struts, and hinge flaps. Structural support, EAC patency, auricle symmetry, overall auricle size, and re-creation of natural contours were considered when designing the reconstruction of the defect in our patient; however, his main priority was achieving the greatest cosmetic outcome in a single-stage procedure, therefore limiting our reconstruction options.
Wedge excision, in which the residual lobule and inferior helical rim are removed, could have been considered in our patient but would have drastically altered the symmetry of the size of the ears. A folded postauricular flap, as described in the otolaryngology literature, is an interpolation flap based on the posterior auricular artery that was designed for full-thickness defects of the auricle to prevent any posterior pinning.1 This technique may have worked well in our case, but the patient preferred to avoid a multistage procedure. Additionally, the positional symmetry of the ears was maintained despite utilizing a hinge flap, which does not involve takedown of the pedicle. A composite graft from the contralateral ear could be considered for smaller conchal bowl defects but likely would have resulted in graft failure in our patient’s large defect due to its need for rich blood supply to heal and dependence on lateral wound edges. Cartilage struts in conjunction with a flap could have been considered in this scenario for greater structural support, but in our patient’s case, by maintaining the robust pedicle of our flap and having residual superior cartilage, further structural support was not necessary.
A prior case report described a partial and full-thickness defect in a similar location that was repaired with a retroauricular hinge flap, in which a portion of the flap was extensively de-epithelialized to address the varied thicknesses of the surgical defect.2 In our patient, the defect abutted the skin reservoir on the superolateral neck, and therefore no de-epithelialization was required as the entire epithelialized portion was utilized to recreate the anterior aspect of the defect. Postauricular hinge-type flaps are a reliable, single-stage surgical alternative to the 2-stage folded postauricular interpolation flap when reconstructing large conchal bowl defects. For small full-thickness defects of the ear, a composite graft may be considered; however, blood supply and other nutritional requirements limit this option for large full-thickness defects.
- Roche AM, Griffin M, Shelton R, et al. The folded postauricular flap: a novel approach to reconstruction of large full thickness defects of the conchal bowl. Am J Otolaryngol. 2017;38:706-709. doi:10.1016 /j.amjoto.2017.09.006
- Klein JC, Nijhawan RI. Retroauricular hinge flaps for full-thickness conchal bowl defects. J Am Acad Dermatol. 2024;90:E71-E72. doi:10.1016/j.jaad.2022.10.056
- Pickrell BB, Hughes CD, Maricevich RS. Partial ear defects. Semin Plast Surg. 2017 Aug;31:134-140. doi:10.1055/s-0037-1603968.
Practice Gap
Large full-thickness conchal bowl defects often pose a reconstructive challenge. Maintaining the shape and structural integrity of the concha is fundamental for optimal cosmetic and functional outcomes. Prior reports have suggested wedge excisions, composite grafts, interpolation flaps with or without cartilage struts, and hinge flaps as possible options for reconstruction.1-3 However, patients with large defects who prefer single-stage reconstruction procedures present a unique challenge. Herein, we describe a single-stage full-thickness hinge flap technique for a large conchal bowl defect.
The Technique
A 77-year-old man was referred to our dermatology clinic by an outside dermatologist for Mohs micrographic surgery of a biopsy-proven cutaneous squamous cell carcinoma on the right conchal bowl measuring 1.1×2.1 cm and extending to the edge of the external auditory canal (EAC). The excision was performed that same day and was completed in 2 stages, achieving negative margins and resulting in a full-thickness defect measuring 2.0×3.6 cm that included the posterior auricular sulcus, cavum, antitragus, and proximal EAC (Figure 1). The patient requested a single-stage procedure but emphasized that his main priority was an optimal cosmetic outcome.

To repair this large defect, a full-thickness hinge flap with Burow graft was performed. The hinge-type flap was designed in a triangular fashion emanating at the posterior auricular sulcus adjacent to the posterior aspect of the defect and extending down the lateral neck (Figure 2). The flap was incised and the surrounding tissue was undermined, maintaining a robust pedicle in the center of its body on the superolateral neck. The flap was passed through the posterior aspect of the full-thickness defect and was secured in place with 4-0 polyglactin sutures in a buried interrupted fashion, thereby recreating the anterior portion of the defect. The superficial skin edges were reapproximated using 4-0 and 5-0 polypropylene sutures in a running interrupted fashion. The distal Burow triangle created from closure of the flap’s secondary defect was aggressively thinned and was utilized as a full-thickness graft for the residual postauricular groove defect (Figure 3). At 2 weeks’ follow-up, the patient was healing well with no postoperative issues and the sutures were removed (Figure 4).



Practice Implications
There are many different reconstructive options for conchal bowl defects, including primary repair, wedge excision, composite graft and interpolation flaps with or without cartilage struts, and hinge flaps. Structural support, EAC patency, auricle symmetry, overall auricle size, and re-creation of natural contours were considered when designing the reconstruction of the defect in our patient; however, his main priority was achieving the greatest cosmetic outcome in a single-stage procedure, therefore limiting our reconstruction options.
Wedge excision, in which the residual lobule and inferior helical rim are removed, could have been considered in our patient but would have drastically altered the symmetry of the size of the ears. A folded postauricular flap, as described in the otolaryngology literature, is an interpolation flap based on the posterior auricular artery that was designed for full-thickness defects of the auricle to prevent any posterior pinning.1 This technique may have worked well in our case, but the patient preferred to avoid a multistage procedure. Additionally, the positional symmetry of the ears was maintained despite utilizing a hinge flap, which does not involve takedown of the pedicle. A composite graft from the contralateral ear could be considered for smaller conchal bowl defects but likely would have resulted in graft failure in our patient’s large defect due to its need for rich blood supply to heal and dependence on lateral wound edges. Cartilage struts in conjunction with a flap could have been considered in this scenario for greater structural support, but in our patient’s case, by maintaining the robust pedicle of our flap and having residual superior cartilage, further structural support was not necessary.
A prior case report described a partial and full-thickness defect in a similar location that was repaired with a retroauricular hinge flap, in which a portion of the flap was extensively de-epithelialized to address the varied thicknesses of the surgical defect.2 In our patient, the defect abutted the skin reservoir on the superolateral neck, and therefore no de-epithelialization was required as the entire epithelialized portion was utilized to recreate the anterior aspect of the defect. Postauricular hinge-type flaps are a reliable, single-stage surgical alternative to the 2-stage folded postauricular interpolation flap when reconstructing large conchal bowl defects. For small full-thickness defects of the ear, a composite graft may be considered; however, blood supply and other nutritional requirements limit this option for large full-thickness defects.
Practice Gap
Large full-thickness conchal bowl defects often pose a reconstructive challenge. Maintaining the shape and structural integrity of the concha is fundamental for optimal cosmetic and functional outcomes. Prior reports have suggested wedge excisions, composite grafts, interpolation flaps with or without cartilage struts, and hinge flaps as possible options for reconstruction.1-3 However, patients with large defects who prefer single-stage reconstruction procedures present a unique challenge. Herein, we describe a single-stage full-thickness hinge flap technique for a large conchal bowl defect.
The Technique
A 77-year-old man was referred to our dermatology clinic by an outside dermatologist for Mohs micrographic surgery of a biopsy-proven cutaneous squamous cell carcinoma on the right conchal bowl measuring 1.1×2.1 cm and extending to the edge of the external auditory canal (EAC). The excision was performed that same day and was completed in 2 stages, achieving negative margins and resulting in a full-thickness defect measuring 2.0×3.6 cm that included the posterior auricular sulcus, cavum, antitragus, and proximal EAC (Figure 1). The patient requested a single-stage procedure but emphasized that his main priority was an optimal cosmetic outcome.

To repair this large defect, a full-thickness hinge flap with Burow graft was performed. The hinge-type flap was designed in a triangular fashion emanating at the posterior auricular sulcus adjacent to the posterior aspect of the defect and extending down the lateral neck (Figure 2). The flap was incised and the surrounding tissue was undermined, maintaining a robust pedicle in the center of its body on the superolateral neck. The flap was passed through the posterior aspect of the full-thickness defect and was secured in place with 4-0 polyglactin sutures in a buried interrupted fashion, thereby recreating the anterior portion of the defect. The superficial skin edges were reapproximated using 4-0 and 5-0 polypropylene sutures in a running interrupted fashion. The distal Burow triangle created from closure of the flap’s secondary defect was aggressively thinned and was utilized as a full-thickness graft for the residual postauricular groove defect (Figure 3). At 2 weeks’ follow-up, the patient was healing well with no postoperative issues and the sutures were removed (Figure 4).



Practice Implications
There are many different reconstructive options for conchal bowl defects, including primary repair, wedge excision, composite graft and interpolation flaps with or without cartilage struts, and hinge flaps. Structural support, EAC patency, auricle symmetry, overall auricle size, and re-creation of natural contours were considered when designing the reconstruction of the defect in our patient; however, his main priority was achieving the greatest cosmetic outcome in a single-stage procedure, therefore limiting our reconstruction options.
Wedge excision, in which the residual lobule and inferior helical rim are removed, could have been considered in our patient but would have drastically altered the symmetry of the size of the ears. A folded postauricular flap, as described in the otolaryngology literature, is an interpolation flap based on the posterior auricular artery that was designed for full-thickness defects of the auricle to prevent any posterior pinning.1 This technique may have worked well in our case, but the patient preferred to avoid a multistage procedure. Additionally, the positional symmetry of the ears was maintained despite utilizing a hinge flap, which does not involve takedown of the pedicle. A composite graft from the contralateral ear could be considered for smaller conchal bowl defects but likely would have resulted in graft failure in our patient’s large defect due to its need for rich blood supply to heal and dependence on lateral wound edges. Cartilage struts in conjunction with a flap could have been considered in this scenario for greater structural support, but in our patient’s case, by maintaining the robust pedicle of our flap and having residual superior cartilage, further structural support was not necessary.
A prior case report described a partial and full-thickness defect in a similar location that was repaired with a retroauricular hinge flap, in which a portion of the flap was extensively de-epithelialized to address the varied thicknesses of the surgical defect.2 In our patient, the defect abutted the skin reservoir on the superolateral neck, and therefore no de-epithelialization was required as the entire epithelialized portion was utilized to recreate the anterior aspect of the defect. Postauricular hinge-type flaps are a reliable, single-stage surgical alternative to the 2-stage folded postauricular interpolation flap when reconstructing large conchal bowl defects. For small full-thickness defects of the ear, a composite graft may be considered; however, blood supply and other nutritional requirements limit this option for large full-thickness defects.
- Roche AM, Griffin M, Shelton R, et al. The folded postauricular flap: a novel approach to reconstruction of large full thickness defects of the conchal bowl. Am J Otolaryngol. 2017;38:706-709. doi:10.1016 /j.amjoto.2017.09.006
- Klein JC, Nijhawan RI. Retroauricular hinge flaps for full-thickness conchal bowl defects. J Am Acad Dermatol. 2024;90:E71-E72. doi:10.1016/j.jaad.2022.10.056
- Pickrell BB, Hughes CD, Maricevich RS. Partial ear defects. Semin Plast Surg. 2017 Aug;31:134-140. doi:10.1055/s-0037-1603968.
- Roche AM, Griffin M, Shelton R, et al. The folded postauricular flap: a novel approach to reconstruction of large full thickness defects of the conchal bowl. Am J Otolaryngol. 2017;38:706-709. doi:10.1016 /j.amjoto.2017.09.006
- Klein JC, Nijhawan RI. Retroauricular hinge flaps for full-thickness conchal bowl defects. J Am Acad Dermatol. 2024;90:E71-E72. doi:10.1016/j.jaad.2022.10.056
- Pickrell BB, Hughes CD, Maricevich RS. Partial ear defects. Semin Plast Surg. 2017 Aug;31:134-140. doi:10.1055/s-0037-1603968.
Repair of a Large Full-Thickness Conchal Bowl Defect
Repair of a Large Full-Thickness Conchal Bowl Defect