User login
Ibrutinib bodes well for relapsed mantle-cell lymphoma
Progression-free survival was significantly better when patients with relapsed or refractory mantle-cell lymphoma were treated with oral ibrutinib than with intravenous temsirolimus, based on results from 280 patients in an international, randomized, open-label phase III trial.
Study subjects had undergone one or more previous rituximab-containing chemotherapy regimens to receive intravenous temsirolimus or oral ibrutinib at a daily dose of 560 mg.
Compared with temsirolimus, ibrutinib resulted in a 57% reduction in the risk of disease progression or death at a median follow-up of 20 months. Median progression-free survival – the trial’s primary endpoint – was 14.6 months for the ibrutinib group and 6.2 months for the temsirolimus group.
Ibrutinib was also better tolerated, with 68% of patients having grade 3 or higher treatment-emergent adverse events as compared to 87% of patients in the temsirolimus group, despite a median 4-fold longer treatment duration for the ibrutinib group than the temsirolimus group. Additionally, 6% of patients discontinued ibrutinib because of adverse events versus 26% in the temsirolimus group, reported Dr. Martin Dreyling of Klinikum der Universität in Munich, Germany, and his associates.
Based on results of the Functional Assessment of Cancer Therapy-Lymphoma (FACT-Lym) questionnaire, ibrutinib was associated with greater and more rapid improvements, and also with less worsening in lymphoma symptoms, as measured by the lymphoma subscale of the FACT-Lym (Lancet. 2016;387:770-78).
Ibrutinib, a first-in-class oral inhibitor of Bruton’s tyrosine kinase, is approved in the United States and the European Union at a dose of 560 mg per day for patients with mantle cell lymphoma who have received at least one previous line of therapy.
The mammalian target of rapamycin (mTOR) inhibitor temsirolimus is approved in the European Union for relapsed or refractory mantle-cell lymphoma, but does not have FDA approval for this indication.
The study, funded by Janssen, is ongoing. Future research, the investigators say, should examine ibrutinib-based combination approaches for patients with relapsed or refractory mantle-cell lymphoma and in front-line therapy.
Dr. Dreyling reported grants and personal fees from Janssen and Pfizer outside of the study. Several other authors reported grants from Janssen during the study and financial ties to the company.
The findings from this phase III trial clearly establish ibrutinib as a new standard for treatment of relapsed mantle-cell lymphoma. Within the next 2 years, many expect the agent will find its way into the frontline setting for treatment of mantle cell lymphoma in combination with standard chemotherapy, based on results of another already completed phase III trial (the SHINE trial).
Despite this remarkable progress, however, mantle-cell lymphoma remains incurable. Roughly 30%-40% of people with the disease will not respond to ibrutinib, and even among responders relapse seems inevitable.
Mantle-cell lymphoma has been a model for accelerated development of novel drugs. Ibrutinib was developed with tremendous speed, and the FDA’s approval of the agent in 2013 based on findings from a non-pivotal phase II trial was surprising to everyone other than the participating patients and physicians. Hopefully the resources mobilized to bring ibrutinib so far, so fast, will continue to be available to help us learn how best to use the drug.
Dr. Peter Martin is with the department of medicine at Weill Cornell Medical College in New York. His comments are excerpted from an editorial that accompanied the study in The Lancet. Dr. Martin reported that he is a consultant for Janssen and has received honoraria from the company for speaking.
The findings from this phase III trial clearly establish ibrutinib as a new standard for treatment of relapsed mantle-cell lymphoma. Within the next 2 years, many expect the agent will find its way into the frontline setting for treatment of mantle cell lymphoma in combination with standard chemotherapy, based on results of another already completed phase III trial (the SHINE trial).
Despite this remarkable progress, however, mantle-cell lymphoma remains incurable. Roughly 30%-40% of people with the disease will not respond to ibrutinib, and even among responders relapse seems inevitable.
Mantle-cell lymphoma has been a model for accelerated development of novel drugs. Ibrutinib was developed with tremendous speed, and the FDA’s approval of the agent in 2013 based on findings from a non-pivotal phase II trial was surprising to everyone other than the participating patients and physicians. Hopefully the resources mobilized to bring ibrutinib so far, so fast, will continue to be available to help us learn how best to use the drug.
Dr. Peter Martin is with the department of medicine at Weill Cornell Medical College in New York. His comments are excerpted from an editorial that accompanied the study in The Lancet. Dr. Martin reported that he is a consultant for Janssen and has received honoraria from the company for speaking.
The findings from this phase III trial clearly establish ibrutinib as a new standard for treatment of relapsed mantle-cell lymphoma. Within the next 2 years, many expect the agent will find its way into the frontline setting for treatment of mantle cell lymphoma in combination with standard chemotherapy, based on results of another already completed phase III trial (the SHINE trial).
Despite this remarkable progress, however, mantle-cell lymphoma remains incurable. Roughly 30%-40% of people with the disease will not respond to ibrutinib, and even among responders relapse seems inevitable.
Mantle-cell lymphoma has been a model for accelerated development of novel drugs. Ibrutinib was developed with tremendous speed, and the FDA’s approval of the agent in 2013 based on findings from a non-pivotal phase II trial was surprising to everyone other than the participating patients and physicians. Hopefully the resources mobilized to bring ibrutinib so far, so fast, will continue to be available to help us learn how best to use the drug.
Dr. Peter Martin is with the department of medicine at Weill Cornell Medical College in New York. His comments are excerpted from an editorial that accompanied the study in The Lancet. Dr. Martin reported that he is a consultant for Janssen and has received honoraria from the company for speaking.
Progression-free survival was significantly better when patients with relapsed or refractory mantle-cell lymphoma were treated with oral ibrutinib than with intravenous temsirolimus, based on results from 280 patients in an international, randomized, open-label phase III trial.
Study subjects had undergone one or more previous rituximab-containing chemotherapy regimens to receive intravenous temsirolimus or oral ibrutinib at a daily dose of 560 mg.
Compared with temsirolimus, ibrutinib resulted in a 57% reduction in the risk of disease progression or death at a median follow-up of 20 months. Median progression-free survival – the trial’s primary endpoint – was 14.6 months for the ibrutinib group and 6.2 months for the temsirolimus group.
Ibrutinib was also better tolerated, with 68% of patients having grade 3 or higher treatment-emergent adverse events as compared to 87% of patients in the temsirolimus group, despite a median 4-fold longer treatment duration for the ibrutinib group than the temsirolimus group. Additionally, 6% of patients discontinued ibrutinib because of adverse events versus 26% in the temsirolimus group, reported Dr. Martin Dreyling of Klinikum der Universität in Munich, Germany, and his associates.
Based on results of the Functional Assessment of Cancer Therapy-Lymphoma (FACT-Lym) questionnaire, ibrutinib was associated with greater and more rapid improvements, and also with less worsening in lymphoma symptoms, as measured by the lymphoma subscale of the FACT-Lym (Lancet. 2016;387:770-78).
Ibrutinib, a first-in-class oral inhibitor of Bruton’s tyrosine kinase, is approved in the United States and the European Union at a dose of 560 mg per day for patients with mantle cell lymphoma who have received at least one previous line of therapy.
The mammalian target of rapamycin (mTOR) inhibitor temsirolimus is approved in the European Union for relapsed or refractory mantle-cell lymphoma, but does not have FDA approval for this indication.
The study, funded by Janssen, is ongoing. Future research, the investigators say, should examine ibrutinib-based combination approaches for patients with relapsed or refractory mantle-cell lymphoma and in front-line therapy.
Dr. Dreyling reported grants and personal fees from Janssen and Pfizer outside of the study. Several other authors reported grants from Janssen during the study and financial ties to the company.
Progression-free survival was significantly better when patients with relapsed or refractory mantle-cell lymphoma were treated with oral ibrutinib than with intravenous temsirolimus, based on results from 280 patients in an international, randomized, open-label phase III trial.
Study subjects had undergone one or more previous rituximab-containing chemotherapy regimens to receive intravenous temsirolimus or oral ibrutinib at a daily dose of 560 mg.
Compared with temsirolimus, ibrutinib resulted in a 57% reduction in the risk of disease progression or death at a median follow-up of 20 months. Median progression-free survival – the trial’s primary endpoint – was 14.6 months for the ibrutinib group and 6.2 months for the temsirolimus group.
Ibrutinib was also better tolerated, with 68% of patients having grade 3 or higher treatment-emergent adverse events as compared to 87% of patients in the temsirolimus group, despite a median 4-fold longer treatment duration for the ibrutinib group than the temsirolimus group. Additionally, 6% of patients discontinued ibrutinib because of adverse events versus 26% in the temsirolimus group, reported Dr. Martin Dreyling of Klinikum der Universität in Munich, Germany, and his associates.
Based on results of the Functional Assessment of Cancer Therapy-Lymphoma (FACT-Lym) questionnaire, ibrutinib was associated with greater and more rapid improvements, and also with less worsening in lymphoma symptoms, as measured by the lymphoma subscale of the FACT-Lym (Lancet. 2016;387:770-78).
Ibrutinib, a first-in-class oral inhibitor of Bruton’s tyrosine kinase, is approved in the United States and the European Union at a dose of 560 mg per day for patients with mantle cell lymphoma who have received at least one previous line of therapy.
The mammalian target of rapamycin (mTOR) inhibitor temsirolimus is approved in the European Union for relapsed or refractory mantle-cell lymphoma, but does not have FDA approval for this indication.
The study, funded by Janssen, is ongoing. Future research, the investigators say, should examine ibrutinib-based combination approaches for patients with relapsed or refractory mantle-cell lymphoma and in front-line therapy.
Dr. Dreyling reported grants and personal fees from Janssen and Pfizer outside of the study. Several other authors reported grants from Janssen during the study and financial ties to the company.
FROM THE LANCET
Key clinical point: Ibrutinib significantly improved progression-free survival, compared with temsirolimus in patients with relapsed or refractory mantle-cell lymphoma.
Major finding: Median progression-free survival was 14.6 months with ibrutinib and 6.2 months with temsirolimus.
Data source: A randomized open-label phase III trial (ongoing) that randomized 280 patients to each treatment group.
Disclosures: The study was funded by Janssen. Dr. Dreyling reported grants and personal fees from Janssen and Pfizer outside of the study, and other authors reported grants from Janssen during the study and financial ties to the company.
Children’s cancer survival steadily increasing
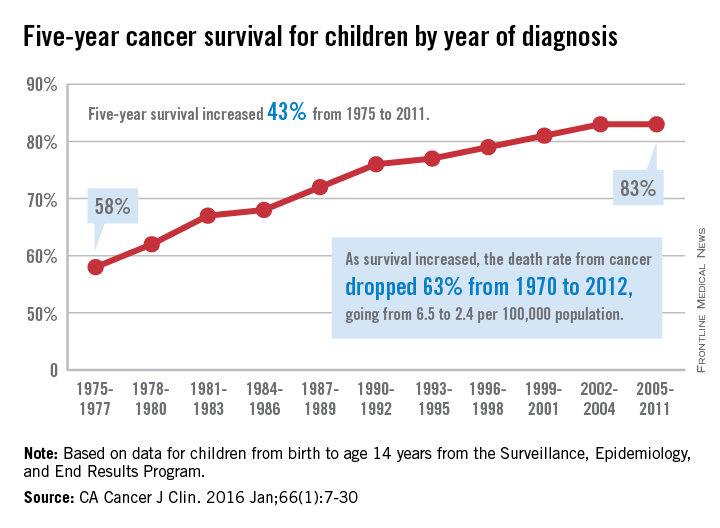
The 5-year cancer survival rate for children younger than 15 years old is up by 43% since 1975, according to investigators from the American Cancer Society.
The 5-year survival rate for all cancers showed a statistically significant rise from 58% in 1975 to 83% in 2011, said Rebecca L. Siegel and her associates at the ACS (CA Cancer J Clin. 2016 Jan;66[1]:7-30).
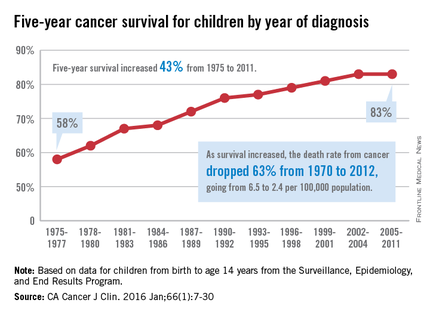
“The substantial progress for all of the major childhood cancers reflects both improvements in treatment and high levels of participation in clinical trials,” they wrote.
Survival for cancers of the brain and nervous system – now the leading cause of cancer death for those younger than 20 years old – increased from 57% in 1975 to 74% in 2011. The next-most-common cause of cancer death in children and adolescents is leukemia, and 5-year survival for acute myeloid leukemia went from 19% in 1975 to 67% in 2011, while 5-year survival for acute lymphocytic leukemia rose from 57% to 91% over that time period, the investigators reported.
The authors reported no conflicts of interest.
The 5-year cancer survival rate for children younger than 15 years old is up by 43% since 1975, according to investigators from the American Cancer Society.
The 5-year survival rate for all cancers showed a statistically significant rise from 58% in 1975 to 83% in 2011, said Rebecca L. Siegel and her associates at the ACS (CA Cancer J Clin. 2016 Jan;66[1]:7-30).

“The substantial progress for all of the major childhood cancers reflects both improvements in treatment and high levels of participation in clinical trials,” they wrote.
Survival for cancers of the brain and nervous system – now the leading cause of cancer death for those younger than 20 years old – increased from 57% in 1975 to 74% in 2011. The next-most-common cause of cancer death in children and adolescents is leukemia, and 5-year survival for acute myeloid leukemia went from 19% in 1975 to 67% in 2011, while 5-year survival for acute lymphocytic leukemia rose from 57% to 91% over that time period, the investigators reported.
The authors reported no conflicts of interest.
The 5-year cancer survival rate for children younger than 15 years old is up by 43% since 1975, according to investigators from the American Cancer Society.
The 5-year survival rate for all cancers showed a statistically significant rise from 58% in 1975 to 83% in 2011, said Rebecca L. Siegel and her associates at the ACS (CA Cancer J Clin. 2016 Jan;66[1]:7-30).

“The substantial progress for all of the major childhood cancers reflects both improvements in treatment and high levels of participation in clinical trials,” they wrote.
Survival for cancers of the brain and nervous system – now the leading cause of cancer death for those younger than 20 years old – increased from 57% in 1975 to 74% in 2011. The next-most-common cause of cancer death in children and adolescents is leukemia, and 5-year survival for acute myeloid leukemia went from 19% in 1975 to 67% in 2011, while 5-year survival for acute lymphocytic leukemia rose from 57% to 91% over that time period, the investigators reported.
The authors reported no conflicts of interest.
FROM CA: A CANCER JOURNAL FOR CLINICIANS
Ki-67 bests cytology, growth pattern as prognostic factor for MCL
Evaluating routinely available histopathological prognostic features from more than 500 MCL patients in prospective trials, researchers found that the Ki-67 index is a better prognostic factor than are cytology and growth pattern in mantle-cell lymphoma (MCL). In addition, the combination of the Ki-67 index with the Mantle Cell Lymphoma International Prognostic Index [MIPI] defined four prognostic groups with better discrimination than did MIPI or the two-category biologic MIPI (MIPI-b) alone.
Higher Ki-67 index was associated with poorer overall survival (OS) (hazard ratio [HR], 1.24 per 10% increase; P less than .001) and progression-free survival (PFS) (HR, 1.17; P less than .001). Consistent with an earlier, population-based study, results showed prognostic value for a 30% cutoff of the Ki-67 index. Quantitative levels below 30% provided no additional prognostic information.
“The Ki-67 index remains the only routinely available independent prognostic factor in addition to MIPI. In contrast to cytology and growth pattern, the Ki-67 evaluation has been standardized for routine application,” wrote Dr. Eva Hoster of University Hospital Munich, and colleagues. “The modified combination of Ki-67 index and MIPI integrates the most important clinical and biologic markers currently available in clinical routine and was shown to allow a simple and powerful risk stratification superior to MIPI and MIPI-b in our evaluation,” they added (J Clin Oncol. 2016 Feb. 29. doi: 10.1200/jco.63.8387).
Blastoid cytology was associated with inferior 5-year OS compared with nonblastoid cytology (35% vs. 68%; HR, 2.35; P less than .001) and PFS (29% vs. 44%; HR, 1.58; P = .007), but the effect was largely accounted for by a generally higher Ki-67 index in blastoid MCL. Diffuse growth pattern was associated slightly worse 5-year OS (61% vs. 72%; HR, 1.38; P = .048) and PFS (38% vs. 49%; HR, 1.25; P = .087), but the effect was largely explained by MIPI score.
Combining dichotomized Ki-67 (above or below 30%) with MIPI risk groups defined four prognostic groups by the sum of weights (total 0 to 3): Ki-67 of 30% or more (weight 1), intermediate-risk MIPI (weight 1), and high-risk MIPI (weight 2). The 5-year OS rates for the four groups ranged from 17% to 85%, with OS hazard ratios greater than 2 between adjacent risk groups.
The study analyzed pooled data from two randomized trials initiated in 2004 by the European Mantle Cell Lymphoma Network, MCL Younger and MCL Elderly. In total, 508 patients of median age 62 years were included. The proportion of low-risk, intermediate-risk, and high-risk MIPI were 41%, 35%, and 24%, respectively.
Research was supported in part by Roche. Dr. Hoster reported receiving funding from Roche Pharma AG and Celgene. Several of her coauthors reported ties to industry.
Evaluating routinely available histopathological prognostic features from more than 500 MCL patients in prospective trials, researchers found that the Ki-67 index is a better prognostic factor than are cytology and growth pattern in mantle-cell lymphoma (MCL). In addition, the combination of the Ki-67 index with the Mantle Cell Lymphoma International Prognostic Index [MIPI] defined four prognostic groups with better discrimination than did MIPI or the two-category biologic MIPI (MIPI-b) alone.
Higher Ki-67 index was associated with poorer overall survival (OS) (hazard ratio [HR], 1.24 per 10% increase; P less than .001) and progression-free survival (PFS) (HR, 1.17; P less than .001). Consistent with an earlier, population-based study, results showed prognostic value for a 30% cutoff of the Ki-67 index. Quantitative levels below 30% provided no additional prognostic information.
“The Ki-67 index remains the only routinely available independent prognostic factor in addition to MIPI. In contrast to cytology and growth pattern, the Ki-67 evaluation has been standardized for routine application,” wrote Dr. Eva Hoster of University Hospital Munich, and colleagues. “The modified combination of Ki-67 index and MIPI integrates the most important clinical and biologic markers currently available in clinical routine and was shown to allow a simple and powerful risk stratification superior to MIPI and MIPI-b in our evaluation,” they added (J Clin Oncol. 2016 Feb. 29. doi: 10.1200/jco.63.8387).
Blastoid cytology was associated with inferior 5-year OS compared with nonblastoid cytology (35% vs. 68%; HR, 2.35; P less than .001) and PFS (29% vs. 44%; HR, 1.58; P = .007), but the effect was largely accounted for by a generally higher Ki-67 index in blastoid MCL. Diffuse growth pattern was associated slightly worse 5-year OS (61% vs. 72%; HR, 1.38; P = .048) and PFS (38% vs. 49%; HR, 1.25; P = .087), but the effect was largely explained by MIPI score.
Combining dichotomized Ki-67 (above or below 30%) with MIPI risk groups defined four prognostic groups by the sum of weights (total 0 to 3): Ki-67 of 30% or more (weight 1), intermediate-risk MIPI (weight 1), and high-risk MIPI (weight 2). The 5-year OS rates for the four groups ranged from 17% to 85%, with OS hazard ratios greater than 2 between adjacent risk groups.
The study analyzed pooled data from two randomized trials initiated in 2004 by the European Mantle Cell Lymphoma Network, MCL Younger and MCL Elderly. In total, 508 patients of median age 62 years were included. The proportion of low-risk, intermediate-risk, and high-risk MIPI were 41%, 35%, and 24%, respectively.
Research was supported in part by Roche. Dr. Hoster reported receiving funding from Roche Pharma AG and Celgene. Several of her coauthors reported ties to industry.
Evaluating routinely available histopathological prognostic features from more than 500 MCL patients in prospective trials, researchers found that the Ki-67 index is a better prognostic factor than are cytology and growth pattern in mantle-cell lymphoma (MCL). In addition, the combination of the Ki-67 index with the Mantle Cell Lymphoma International Prognostic Index [MIPI] defined four prognostic groups with better discrimination than did MIPI or the two-category biologic MIPI (MIPI-b) alone.
Higher Ki-67 index was associated with poorer overall survival (OS) (hazard ratio [HR], 1.24 per 10% increase; P less than .001) and progression-free survival (PFS) (HR, 1.17; P less than .001). Consistent with an earlier, population-based study, results showed prognostic value for a 30% cutoff of the Ki-67 index. Quantitative levels below 30% provided no additional prognostic information.
“The Ki-67 index remains the only routinely available independent prognostic factor in addition to MIPI. In contrast to cytology and growth pattern, the Ki-67 evaluation has been standardized for routine application,” wrote Dr. Eva Hoster of University Hospital Munich, and colleagues. “The modified combination of Ki-67 index and MIPI integrates the most important clinical and biologic markers currently available in clinical routine and was shown to allow a simple and powerful risk stratification superior to MIPI and MIPI-b in our evaluation,” they added (J Clin Oncol. 2016 Feb. 29. doi: 10.1200/jco.63.8387).
Blastoid cytology was associated with inferior 5-year OS compared with nonblastoid cytology (35% vs. 68%; HR, 2.35; P less than .001) and PFS (29% vs. 44%; HR, 1.58; P = .007), but the effect was largely accounted for by a generally higher Ki-67 index in blastoid MCL. Diffuse growth pattern was associated slightly worse 5-year OS (61% vs. 72%; HR, 1.38; P = .048) and PFS (38% vs. 49%; HR, 1.25; P = .087), but the effect was largely explained by MIPI score.
Combining dichotomized Ki-67 (above or below 30%) with MIPI risk groups defined four prognostic groups by the sum of weights (total 0 to 3): Ki-67 of 30% or more (weight 1), intermediate-risk MIPI (weight 1), and high-risk MIPI (weight 2). The 5-year OS rates for the four groups ranged from 17% to 85%, with OS hazard ratios greater than 2 between adjacent risk groups.
The study analyzed pooled data from two randomized trials initiated in 2004 by the European Mantle Cell Lymphoma Network, MCL Younger and MCL Elderly. In total, 508 patients of median age 62 years were included. The proportion of low-risk, intermediate-risk, and high-risk MIPI were 41%, 35%, and 24%, respectively.
Research was supported in part by Roche. Dr. Hoster reported receiving funding from Roche Pharma AG and Celgene. Several of her coauthors reported ties to industry.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: The Ki-67 index was superior to cytology and growth pattern as a prognostic factor in mantle-cell lymphoma (MCL).
Major finding: Higher Ki-67 index was associated with poorer overall survival (hazard ratio [HR], 1.24 per 10% increase; P less than .001) and progression-free survival (HR, 1.17; P less than .001).
Data source: Pooled data from two randomized trials initiated in 2004 by the European Mantle Cell Lymphoma Network, MCL Younger and MCL Elderly, included 508 patients.
Disclosures: Research was supported in part by Roche. Dr. Hoster reported receiving funding from Roche Pharma AG and Celgene. Several of her coauthors reported ties to industry.
FDA approves obinutuzumab for FL

The US Food and Drug Administration (FDA) has approved obinutuzumab (Gazyva) for certain patients with previously treated follicular lymphoma (FL).
Obinutuzumab is a glycoengineered, humanized, monoclonal antibody that selectively binds to the extracellular domain of the CD20 antigen on B cells.
The drug was previously approved by the FDA for use in combination with chlorambucil to treat patients with previously untreated chronic lymphocytic leukemia.
Now, obinutuzumab is approved for use in combination with bendamustine, followed by obinutuzumab alone, to treat patients with FL who did not respond to a rituximab-containing regimen or whose FL returned after such treatment.
The recommended dose and schedule for the regimen is:
- Obinutuzumab at 1000 mg by intravenous infusion on days 1, 8, and 15 of cycle 1; on day 1 of cycles 2-6 (28-day cycles); then every 2 months for 2 years.
- Bendamustine at 90 mg/m2 by intravenous infusion on days 1 and 2 of cycles 1-6.
Full prescribing information for obinutuzumab is available on the FDA website or at www.Gazyva.com.
Phase 3 study
The approval for obinutuzumab in FL is based on results from the phase 3 GADOLIN study. The trial included 413 patients with rituximab-refractory non-Hodgkin lymphoma, including 321 patients with FL, 46 with marginal zone lymphoma, and 28 with small lymphocytic lymphoma.
The patients were randomized to receive bendamustine alone (control arm) or a combination of bendamustine and obinutuzumab followed by obinutuzumab maintenance (every 2 months for 2 years or until progression).
The primary endpoint of the study was progression-free survival (PFS), as assessed by an independent review committee (IRC). The secondary endpoints were PFS as assessed by investigator review, best overall response, complete response (CR), partial response (PR), duration of response, overall survival, and safety profile.
Among patients with FL, the obinutuzumab regimen improved PFS compared to bendamustine alone, as assessed by IRC (hazard ratio [HR]=0.48, P<0.0001). The median PFS was not reached in patients receiving the obinutuzumab regimen but was 13.8 months in those receiving bendamustine alone.
Investigator-assessed PFS was consistent with IRC-assessed PFS. Investigators said the median PFS with the obinutuzumab regimen was more than double that with bendamustine alone—29.2 months vs 13.7 months (HR=0.48, P<0.0001).
Best overall response for patients receiving the obinutuzumab regimen was 78.7% (15.5% CR, 63.2% PR), compared to 74.7% for those receiving bendamustine alone (18.7% CR, 56% PR), as assessed by the IRC.
The median duration of response was not reached for patients receiving the obinutuzumab regimen and was 11.6 months for those receiving bendamustine alone.
The median overall survival has not yet been reached in either study arm.
The most common grade 3/4 adverse events observed in patients receiving the obinutuzumab regimen were neutropenia (33%), infusion reactions (11%), and thrombocytopenia (10%).
The most common adverse events of any grade were infusion reactions (69%), neutropenia (35%), nausea (54%), fatigue (39%), cough (26%), diarrhea (27%), constipation (19%), fever (18%), thrombocytopenia (15%), vomiting (22%), upper respiratory tract infection (13%), decreased appetite (18%), joint or muscle pain (12%), sinusitis (12%), anemia (12%), general weakness (11%), and urinary tract infection (10%).
About obinutuzumab
Obinutuzumab is being studied in a large clinical program, including the phase 3 GOYA and GALLIUM studies.
In GOYA, researchers are comparing obinutuzumab head-to-head with rituximab plus CHOP chemotherapy in first-line diffuse large B-cell lymphoma. In GALLIUM, researchers are comparing obinutuzumab plus chemotherapy head-to-head with rituximab plus chemotherapy in first-line indolent non-Hodgkin lymphoma.
Additional combination studies investigating obinutuzumab with other approved or investigational medicines, including cancer immunotherapies and small-molecule inhibitors, are planned or underway across a range of blood cancers.
Obinutuzumab was discovered by Roche Glycart AG, a wholly owned, independent research unit of Roche. In the US, obinutuzumab is part of a collaboration between Genentech and Biogen.
Genentech has a patient assistance program, Genentech Access Solutions, that can help qualifying patients access obinutuzumab and other Genentech medications.
The program is designed to help people navigate the access and reimbursement process and provide assistance to eligible patients in the US who are uninsured or cannot afford the out-of-pocket costs for their medicine. For more information, visit www.Genentech-Access.com. ![]()

The US Food and Drug Administration (FDA) has approved obinutuzumab (Gazyva) for certain patients with previously treated follicular lymphoma (FL).
Obinutuzumab is a glycoengineered, humanized, monoclonal antibody that selectively binds to the extracellular domain of the CD20 antigen on B cells.
The drug was previously approved by the FDA for use in combination with chlorambucil to treat patients with previously untreated chronic lymphocytic leukemia.
Now, obinutuzumab is approved for use in combination with bendamustine, followed by obinutuzumab alone, to treat patients with FL who did not respond to a rituximab-containing regimen or whose FL returned after such treatment.
The recommended dose and schedule for the regimen is:
- Obinutuzumab at 1000 mg by intravenous infusion on days 1, 8, and 15 of cycle 1; on day 1 of cycles 2-6 (28-day cycles); then every 2 months for 2 years.
- Bendamustine at 90 mg/m2 by intravenous infusion on days 1 and 2 of cycles 1-6.
Full prescribing information for obinutuzumab is available on the FDA website or at www.Gazyva.com.
Phase 3 study
The approval for obinutuzumab in FL is based on results from the phase 3 GADOLIN study. The trial included 413 patients with rituximab-refractory non-Hodgkin lymphoma, including 321 patients with FL, 46 with marginal zone lymphoma, and 28 with small lymphocytic lymphoma.
The patients were randomized to receive bendamustine alone (control arm) or a combination of bendamustine and obinutuzumab followed by obinutuzumab maintenance (every 2 months for 2 years or until progression).
The primary endpoint of the study was progression-free survival (PFS), as assessed by an independent review committee (IRC). The secondary endpoints were PFS as assessed by investigator review, best overall response, complete response (CR), partial response (PR), duration of response, overall survival, and safety profile.
Among patients with FL, the obinutuzumab regimen improved PFS compared to bendamustine alone, as assessed by IRC (hazard ratio [HR]=0.48, P<0.0001). The median PFS was not reached in patients receiving the obinutuzumab regimen but was 13.8 months in those receiving bendamustine alone.
Investigator-assessed PFS was consistent with IRC-assessed PFS. Investigators said the median PFS with the obinutuzumab regimen was more than double that with bendamustine alone—29.2 months vs 13.7 months (HR=0.48, P<0.0001).
Best overall response for patients receiving the obinutuzumab regimen was 78.7% (15.5% CR, 63.2% PR), compared to 74.7% for those receiving bendamustine alone (18.7% CR, 56% PR), as assessed by the IRC.
The median duration of response was not reached for patients receiving the obinutuzumab regimen and was 11.6 months for those receiving bendamustine alone.
The median overall survival has not yet been reached in either study arm.
The most common grade 3/4 adverse events observed in patients receiving the obinutuzumab regimen were neutropenia (33%), infusion reactions (11%), and thrombocytopenia (10%).
The most common adverse events of any grade were infusion reactions (69%), neutropenia (35%), nausea (54%), fatigue (39%), cough (26%), diarrhea (27%), constipation (19%), fever (18%), thrombocytopenia (15%), vomiting (22%), upper respiratory tract infection (13%), decreased appetite (18%), joint or muscle pain (12%), sinusitis (12%), anemia (12%), general weakness (11%), and urinary tract infection (10%).
About obinutuzumab
Obinutuzumab is being studied in a large clinical program, including the phase 3 GOYA and GALLIUM studies.
In GOYA, researchers are comparing obinutuzumab head-to-head with rituximab plus CHOP chemotherapy in first-line diffuse large B-cell lymphoma. In GALLIUM, researchers are comparing obinutuzumab plus chemotherapy head-to-head with rituximab plus chemotherapy in first-line indolent non-Hodgkin lymphoma.
Additional combination studies investigating obinutuzumab with other approved or investigational medicines, including cancer immunotherapies and small-molecule inhibitors, are planned or underway across a range of blood cancers.
Obinutuzumab was discovered by Roche Glycart AG, a wholly owned, independent research unit of Roche. In the US, obinutuzumab is part of a collaboration between Genentech and Biogen.
Genentech has a patient assistance program, Genentech Access Solutions, that can help qualifying patients access obinutuzumab and other Genentech medications.
The program is designed to help people navigate the access and reimbursement process and provide assistance to eligible patients in the US who are uninsured or cannot afford the out-of-pocket costs for their medicine. For more information, visit www.Genentech-Access.com. ![]()

The US Food and Drug Administration (FDA) has approved obinutuzumab (Gazyva) for certain patients with previously treated follicular lymphoma (FL).
Obinutuzumab is a glycoengineered, humanized, monoclonal antibody that selectively binds to the extracellular domain of the CD20 antigen on B cells.
The drug was previously approved by the FDA for use in combination with chlorambucil to treat patients with previously untreated chronic lymphocytic leukemia.
Now, obinutuzumab is approved for use in combination with bendamustine, followed by obinutuzumab alone, to treat patients with FL who did not respond to a rituximab-containing regimen or whose FL returned after such treatment.
The recommended dose and schedule for the regimen is:
- Obinutuzumab at 1000 mg by intravenous infusion on days 1, 8, and 15 of cycle 1; on day 1 of cycles 2-6 (28-day cycles); then every 2 months for 2 years.
- Bendamustine at 90 mg/m2 by intravenous infusion on days 1 and 2 of cycles 1-6.
Full prescribing information for obinutuzumab is available on the FDA website or at www.Gazyva.com.
Phase 3 study
The approval for obinutuzumab in FL is based on results from the phase 3 GADOLIN study. The trial included 413 patients with rituximab-refractory non-Hodgkin lymphoma, including 321 patients with FL, 46 with marginal zone lymphoma, and 28 with small lymphocytic lymphoma.
The patients were randomized to receive bendamustine alone (control arm) or a combination of bendamustine and obinutuzumab followed by obinutuzumab maintenance (every 2 months for 2 years or until progression).
The primary endpoint of the study was progression-free survival (PFS), as assessed by an independent review committee (IRC). The secondary endpoints were PFS as assessed by investigator review, best overall response, complete response (CR), partial response (PR), duration of response, overall survival, and safety profile.
Among patients with FL, the obinutuzumab regimen improved PFS compared to bendamustine alone, as assessed by IRC (hazard ratio [HR]=0.48, P<0.0001). The median PFS was not reached in patients receiving the obinutuzumab regimen but was 13.8 months in those receiving bendamustine alone.
Investigator-assessed PFS was consistent with IRC-assessed PFS. Investigators said the median PFS with the obinutuzumab regimen was more than double that with bendamustine alone—29.2 months vs 13.7 months (HR=0.48, P<0.0001).
Best overall response for patients receiving the obinutuzumab regimen was 78.7% (15.5% CR, 63.2% PR), compared to 74.7% for those receiving bendamustine alone (18.7% CR, 56% PR), as assessed by the IRC.
The median duration of response was not reached for patients receiving the obinutuzumab regimen and was 11.6 months for those receiving bendamustine alone.
The median overall survival has not yet been reached in either study arm.
The most common grade 3/4 adverse events observed in patients receiving the obinutuzumab regimen were neutropenia (33%), infusion reactions (11%), and thrombocytopenia (10%).
The most common adverse events of any grade were infusion reactions (69%), neutropenia (35%), nausea (54%), fatigue (39%), cough (26%), diarrhea (27%), constipation (19%), fever (18%), thrombocytopenia (15%), vomiting (22%), upper respiratory tract infection (13%), decreased appetite (18%), joint or muscle pain (12%), sinusitis (12%), anemia (12%), general weakness (11%), and urinary tract infection (10%).
About obinutuzumab
Obinutuzumab is being studied in a large clinical program, including the phase 3 GOYA and GALLIUM studies.
In GOYA, researchers are comparing obinutuzumab head-to-head with rituximab plus CHOP chemotherapy in first-line diffuse large B-cell lymphoma. In GALLIUM, researchers are comparing obinutuzumab plus chemotherapy head-to-head with rituximab plus chemotherapy in first-line indolent non-Hodgkin lymphoma.
Additional combination studies investigating obinutuzumab with other approved or investigational medicines, including cancer immunotherapies and small-molecule inhibitors, are planned or underway across a range of blood cancers.
Obinutuzumab was discovered by Roche Glycart AG, a wholly owned, independent research unit of Roche. In the US, obinutuzumab is part of a collaboration between Genentech and Biogen.
Genentech has a patient assistance program, Genentech Access Solutions, that can help qualifying patients access obinutuzumab and other Genentech medications.
The program is designed to help people navigate the access and reimbursement process and provide assistance to eligible patients in the US who are uninsured or cannot afford the out-of-pocket costs for their medicine. For more information, visit www.Genentech-Access.com. ![]()
AAs have lower rate of most blood cancers than NHWs

receiving treatment
Photo by Rhoda Baer
A new report suggests African Americans (AAs) have significantly lower rates of most hematologic malignancies than non-Hispanic white (NHW) individuals in the US.
AAs of both sexes had significantly lower rates of leukemia, Hodgkin lymphoma (HL), and non-Hodgkin lymphoma (NHL) than NHWs, but the rate of myeloma was significantly higher among AAs.
The death rates for these malignancies followed the same patterns, with the exception of HL. There was no significant difference in HL mortality between AAs and NHWs of either sex.
These findings can be found in the report, “Cancer Statistics for African Americans, 2016,” appearing in CA: A Cancer Journal for Clinicians.
To compile this report, the researchers used data from the Surveillance, Epidemiology, and End Results program and the Centers for Disease Control and Prevention’s National Program of Cancer Registries.
Incidence
For part of the report, the researchers compared the incidence of cancers between AAs and NHWs (divided by gender) for the period from 2008 to 2012.
Among females, the incidence of leukemia was 8.6 per 100,000 in AAs and 10.7 per 100,000 in NHWs (P<0.05). Among males, the incidence was 13.2 per 100,000 in AAs and 17.7 per 100,000 in NHWs (P<0.05).
The incidence of HL in females was 2.4 per 100,000 in AAs and 2.7 per 100,000 in NHWs (P<0.05). The incidence of HL in males was 3.2 per 100,000 in AAs and 3.4 per 100,000 in NHWs (P<0.05).
The incidence of NHL in females was 12.0 per 100,000 in AAs and 16.6 per 100,000 in NHWs (P<0.05). The incidence of NHL in males was 17.2 per 100,000 in AAs and 24.1 per 100,000 in NHWs (P<0.05).
The incidence of myeloma in females was 11.1 per 100,000 in AAs and 4.3 per 100,000 in NHWs (P<0.05). The incidence of myeloma in males was 14.8 per 100,000 in AAs and 7.0 per 100,000 in NHWs (P<0.05).
Mortality
The researchers also compared cancer mortality between AAs and NHWs (divided by gender) for the period from 2008 to 2012.
The death rate for female leukemia patients was 4.8 per 100,000 in AAs and 5.4 per 100,000 in NHWs (P<0.05). The death rate for male leukemia patients was 8.1 per 100,000 in AAs and 9.9 per 100,000 in NHWs (P<0.05).
The death rate for female HL patients was 0.3 per 100,000 for both AAs and NHWs. The death rate for male HL patients was 0.4 per 100,000 for AAs and 0.5 per 100,000 in NHWs (not significant).
The death rate for female NHL patients was 3.6 per 100,000 in AAs and 5.0 per 100,000 in NHWs (P<0.05). The death rate for male NHL patients was 5.9 per 100,000 in AAs and 8.3 per 100,000 in NHWs (P<0.05).
The death rate for female myeloma patients was 5.4 per 100,000 in AAs and 2.4 per 100,000 in NHWs (P<0.05). The death rate for male myeloma patients was 7.8 per 100,000 in AAs and 4.0 per 100,000 in NHWs (P<0.05).
The researchers noted that the reasons for the higher rates of myeloma and myeloma death among AAs are, at present, unknown. ![]()

receiving treatment
Photo by Rhoda Baer
A new report suggests African Americans (AAs) have significantly lower rates of most hematologic malignancies than non-Hispanic white (NHW) individuals in the US.
AAs of both sexes had significantly lower rates of leukemia, Hodgkin lymphoma (HL), and non-Hodgkin lymphoma (NHL) than NHWs, but the rate of myeloma was significantly higher among AAs.
The death rates for these malignancies followed the same patterns, with the exception of HL. There was no significant difference in HL mortality between AAs and NHWs of either sex.
These findings can be found in the report, “Cancer Statistics for African Americans, 2016,” appearing in CA: A Cancer Journal for Clinicians.
To compile this report, the researchers used data from the Surveillance, Epidemiology, and End Results program and the Centers for Disease Control and Prevention’s National Program of Cancer Registries.
Incidence
For part of the report, the researchers compared the incidence of cancers between AAs and NHWs (divided by gender) for the period from 2008 to 2012.
Among females, the incidence of leukemia was 8.6 per 100,000 in AAs and 10.7 per 100,000 in NHWs (P<0.05). Among males, the incidence was 13.2 per 100,000 in AAs and 17.7 per 100,000 in NHWs (P<0.05).
The incidence of HL in females was 2.4 per 100,000 in AAs and 2.7 per 100,000 in NHWs (P<0.05). The incidence of HL in males was 3.2 per 100,000 in AAs and 3.4 per 100,000 in NHWs (P<0.05).
The incidence of NHL in females was 12.0 per 100,000 in AAs and 16.6 per 100,000 in NHWs (P<0.05). The incidence of NHL in males was 17.2 per 100,000 in AAs and 24.1 per 100,000 in NHWs (P<0.05).
The incidence of myeloma in females was 11.1 per 100,000 in AAs and 4.3 per 100,000 in NHWs (P<0.05). The incidence of myeloma in males was 14.8 per 100,000 in AAs and 7.0 per 100,000 in NHWs (P<0.05).
Mortality
The researchers also compared cancer mortality between AAs and NHWs (divided by gender) for the period from 2008 to 2012.
The death rate for female leukemia patients was 4.8 per 100,000 in AAs and 5.4 per 100,000 in NHWs (P<0.05). The death rate for male leukemia patients was 8.1 per 100,000 in AAs and 9.9 per 100,000 in NHWs (P<0.05).
The death rate for female HL patients was 0.3 per 100,000 for both AAs and NHWs. The death rate for male HL patients was 0.4 per 100,000 for AAs and 0.5 per 100,000 in NHWs (not significant).
The death rate for female NHL patients was 3.6 per 100,000 in AAs and 5.0 per 100,000 in NHWs (P<0.05). The death rate for male NHL patients was 5.9 per 100,000 in AAs and 8.3 per 100,000 in NHWs (P<0.05).
The death rate for female myeloma patients was 5.4 per 100,000 in AAs and 2.4 per 100,000 in NHWs (P<0.05). The death rate for male myeloma patients was 7.8 per 100,000 in AAs and 4.0 per 100,000 in NHWs (P<0.05).
The researchers noted that the reasons for the higher rates of myeloma and myeloma death among AAs are, at present, unknown. ![]()

receiving treatment
Photo by Rhoda Baer
A new report suggests African Americans (AAs) have significantly lower rates of most hematologic malignancies than non-Hispanic white (NHW) individuals in the US.
AAs of both sexes had significantly lower rates of leukemia, Hodgkin lymphoma (HL), and non-Hodgkin lymphoma (NHL) than NHWs, but the rate of myeloma was significantly higher among AAs.
The death rates for these malignancies followed the same patterns, with the exception of HL. There was no significant difference in HL mortality between AAs and NHWs of either sex.
These findings can be found in the report, “Cancer Statistics for African Americans, 2016,” appearing in CA: A Cancer Journal for Clinicians.
To compile this report, the researchers used data from the Surveillance, Epidemiology, and End Results program and the Centers for Disease Control and Prevention’s National Program of Cancer Registries.
Incidence
For part of the report, the researchers compared the incidence of cancers between AAs and NHWs (divided by gender) for the period from 2008 to 2012.
Among females, the incidence of leukemia was 8.6 per 100,000 in AAs and 10.7 per 100,000 in NHWs (P<0.05). Among males, the incidence was 13.2 per 100,000 in AAs and 17.7 per 100,000 in NHWs (P<0.05).
The incidence of HL in females was 2.4 per 100,000 in AAs and 2.7 per 100,000 in NHWs (P<0.05). The incidence of HL in males was 3.2 per 100,000 in AAs and 3.4 per 100,000 in NHWs (P<0.05).
The incidence of NHL in females was 12.0 per 100,000 in AAs and 16.6 per 100,000 in NHWs (P<0.05). The incidence of NHL in males was 17.2 per 100,000 in AAs and 24.1 per 100,000 in NHWs (P<0.05).
The incidence of myeloma in females was 11.1 per 100,000 in AAs and 4.3 per 100,000 in NHWs (P<0.05). The incidence of myeloma in males was 14.8 per 100,000 in AAs and 7.0 per 100,000 in NHWs (P<0.05).
Mortality
The researchers also compared cancer mortality between AAs and NHWs (divided by gender) for the period from 2008 to 2012.
The death rate for female leukemia patients was 4.8 per 100,000 in AAs and 5.4 per 100,000 in NHWs (P<0.05). The death rate for male leukemia patients was 8.1 per 100,000 in AAs and 9.9 per 100,000 in NHWs (P<0.05).
The death rate for female HL patients was 0.3 per 100,000 for both AAs and NHWs. The death rate for male HL patients was 0.4 per 100,000 for AAs and 0.5 per 100,000 in NHWs (not significant).
The death rate for female NHL patients was 3.6 per 100,000 in AAs and 5.0 per 100,000 in NHWs (P<0.05). The death rate for male NHL patients was 5.9 per 100,000 in AAs and 8.3 per 100,000 in NHWs (P<0.05).
The death rate for female myeloma patients was 5.4 per 100,000 in AAs and 2.4 per 100,000 in NHWs (P<0.05). The death rate for male myeloma patients was 7.8 per 100,000 in AAs and 4.0 per 100,000 in NHWs (P<0.05).
The researchers noted that the reasons for the higher rates of myeloma and myeloma death among AAs are, at present, unknown. ![]()
Orphan designation recommended for BTK inhibitor

The European Medicines Agency’s Committee for Orphan Medicinal Products (COMP) is recommending orphan designation for the second-generation BTK inhibitor acalabrutinib (ACP-196) for 3 indications.
The COMP is recommending the drug receive orphan designation as a treatment for chronic lymphocytic leukemia (CLL)/small lymphocytic lymphoma (SLL), mantle cell lymphoma, and Waldenström’s macroglobulinemia.
The COMP adopts an opinion on orphan drug designation, and that opinion is submitted to the European Commission (EC) for endorsement.
To be granted orphan designation by the EC, a medicine must be intended for the treatment, prevention, or diagnosis of a disease that is life-threatening and has a prevalence of up to 5 in 10,000 in the European Union. Additionally, the medicine must aim to provide significant benefit to those affected by the condition.
Orphan designation provides companies with development and market exclusivity incentives for designated compounds and medicines.
About acalabrutinib
Acalabrutinib is under development by AstraZeneca and Acerta Pharma BV. The drug is currently being evaluated in trials of patients with CLL/SLL, mantle cell lymphoma, Waldentröm’s macroglobulinemia, and a range of other hematologic malignancies and solid tumor cancers.
Data from a phase 1/2 trial of acalabrutinib in CLL were presented at the 2015 ASH Annual Meeting and simultaneously published in NEJM.
The researchers reported on 61 patients with relapsed CLL who had a median age of 62 (range, 44-84) and a median of 3 prior therapies (range, 1-13).
Patients enrolled in the phase 1 portion of the study received escalating doses of acalabrutinib, with a maximum dose of 400 mg once daily. Patients involved in the phase 2 portion of the study were treated with a 100 mg dose twice daily.
At a median follow-up of 14.3 months (range, 0.5 to 20), 53 patients were still receiving treatment.
The most common adverse events of all grades (occurring in at least 20% of patients) were headache (43%), diarrhea (39%), increased weight (26%), pyrexia (23%), upper respiratory tract infection (23%), fatigue (21%), peripheral edema (21%), hypertension (20%), and nausea (20%).
Grade 3/4 adverse events included diarrhea (2%), increased weight (2%), pyrexia (3%), fatigue (3%), hypertension (7%), and arthralgia (2%).
The overall response rate among the 60 evaluable patients was 95%. This included partial responses in 85% of patients and partial responses with lymphocytosis in 10%. The rate of stable disease was 5%.
The researchers noted that responses occurred in all dosing cohorts, and the response rate increased over time. ![]()

The European Medicines Agency’s Committee for Orphan Medicinal Products (COMP) is recommending orphan designation for the second-generation BTK inhibitor acalabrutinib (ACP-196) for 3 indications.
The COMP is recommending the drug receive orphan designation as a treatment for chronic lymphocytic leukemia (CLL)/small lymphocytic lymphoma (SLL), mantle cell lymphoma, and Waldenström’s macroglobulinemia.
The COMP adopts an opinion on orphan drug designation, and that opinion is submitted to the European Commission (EC) for endorsement.
To be granted orphan designation by the EC, a medicine must be intended for the treatment, prevention, or diagnosis of a disease that is life-threatening and has a prevalence of up to 5 in 10,000 in the European Union. Additionally, the medicine must aim to provide significant benefit to those affected by the condition.
Orphan designation provides companies with development and market exclusivity incentives for designated compounds and medicines.
About acalabrutinib
Acalabrutinib is under development by AstraZeneca and Acerta Pharma BV. The drug is currently being evaluated in trials of patients with CLL/SLL, mantle cell lymphoma, Waldentröm’s macroglobulinemia, and a range of other hematologic malignancies and solid tumor cancers.
Data from a phase 1/2 trial of acalabrutinib in CLL were presented at the 2015 ASH Annual Meeting and simultaneously published in NEJM.
The researchers reported on 61 patients with relapsed CLL who had a median age of 62 (range, 44-84) and a median of 3 prior therapies (range, 1-13).
Patients enrolled in the phase 1 portion of the study received escalating doses of acalabrutinib, with a maximum dose of 400 mg once daily. Patients involved in the phase 2 portion of the study were treated with a 100 mg dose twice daily.
At a median follow-up of 14.3 months (range, 0.5 to 20), 53 patients were still receiving treatment.
The most common adverse events of all grades (occurring in at least 20% of patients) were headache (43%), diarrhea (39%), increased weight (26%), pyrexia (23%), upper respiratory tract infection (23%), fatigue (21%), peripheral edema (21%), hypertension (20%), and nausea (20%).
Grade 3/4 adverse events included diarrhea (2%), increased weight (2%), pyrexia (3%), fatigue (3%), hypertension (7%), and arthralgia (2%).
The overall response rate among the 60 evaluable patients was 95%. This included partial responses in 85% of patients and partial responses with lymphocytosis in 10%. The rate of stable disease was 5%.
The researchers noted that responses occurred in all dosing cohorts, and the response rate increased over time. ![]()

The European Medicines Agency’s Committee for Orphan Medicinal Products (COMP) is recommending orphan designation for the second-generation BTK inhibitor acalabrutinib (ACP-196) for 3 indications.
The COMP is recommending the drug receive orphan designation as a treatment for chronic lymphocytic leukemia (CLL)/small lymphocytic lymphoma (SLL), mantle cell lymphoma, and Waldenström’s macroglobulinemia.
The COMP adopts an opinion on orphan drug designation, and that opinion is submitted to the European Commission (EC) for endorsement.
To be granted orphan designation by the EC, a medicine must be intended for the treatment, prevention, or diagnosis of a disease that is life-threatening and has a prevalence of up to 5 in 10,000 in the European Union. Additionally, the medicine must aim to provide significant benefit to those affected by the condition.
Orphan designation provides companies with development and market exclusivity incentives for designated compounds and medicines.
About acalabrutinib
Acalabrutinib is under development by AstraZeneca and Acerta Pharma BV. The drug is currently being evaluated in trials of patients with CLL/SLL, mantle cell lymphoma, Waldentröm’s macroglobulinemia, and a range of other hematologic malignancies and solid tumor cancers.
Data from a phase 1/2 trial of acalabrutinib in CLL were presented at the 2015 ASH Annual Meeting and simultaneously published in NEJM.
The researchers reported on 61 patients with relapsed CLL who had a median age of 62 (range, 44-84) and a median of 3 prior therapies (range, 1-13).
Patients enrolled in the phase 1 portion of the study received escalating doses of acalabrutinib, with a maximum dose of 400 mg once daily. Patients involved in the phase 2 portion of the study were treated with a 100 mg dose twice daily.
At a median follow-up of 14.3 months (range, 0.5 to 20), 53 patients were still receiving treatment.
The most common adverse events of all grades (occurring in at least 20% of patients) were headache (43%), diarrhea (39%), increased weight (26%), pyrexia (23%), upper respiratory tract infection (23%), fatigue (21%), peripheral edema (21%), hypertension (20%), and nausea (20%).
Grade 3/4 adverse events included diarrhea (2%), increased weight (2%), pyrexia (3%), fatigue (3%), hypertension (7%), and arthralgia (2%).
The overall response rate among the 60 evaluable patients was 95%. This included partial responses in 85% of patients and partial responses with lymphocytosis in 10%. The rate of stable disease was 5%.
The researchers noted that responses occurred in all dosing cohorts, and the response rate increased over time. ![]()
How an anticancer drug fights lymphoid malignancies

Photo by Cameron Wells,
Walter and Eliza Hall
Institute of Medical Research
Research published in Cell Reports helps explain how the anticancer agent Nutlin3a fights lymphoma and other hematologic malignancies.
Nutlin3a is known to activate the tumor suppressor p53, but it hasn’t been clear exactly which p53 target genes are essential for the drug’s therapeutic activity.
The new research revealed that PUMA-mediated apoptosis—not p21-mediated cell-cycle arrest or senescence—is responsible for Nutlin3a’s therapeutic activity in lymphoid malignancies.
“By understanding how nutlins are killing cancer cells, we can begin to formulate their best possible use, including choosing the best partner drugs to combine the nutlins with,” said study author Andreas Strasser, PhD, of the Walter and Eliza Hall Institute of Medical Research in Parkville, Victoria, Australia.
With this study, Dr Strasser and his colleagues first found that Nutlin3a activates p53 target gene expression and causes cell-cycle arrest and apoptosis in non-transformed mouse lymphoid cells in vitro.
The team then showed that Nutlin3a-mediated killing of these cells requires PUMA but not p21. In vivo, loss of PUMA protected non-transformed mouse lymphoid cells against Nutlin3a-induced killing. Loss of p21 did not provide the same protection.
Next, the researchers found that malignant Eµ-Myc lymphoma cells were much more sensitive to Nutlin3a than were non-transformed lymphoid cells. In vitro experiments with Eµ-Myc lymphoma cells showed that Nutlin3a promotes p53 accumulation and downstream effector pathway activation.
As in previous experiments, PUMA (not p21) proved critical for Nutlin3a-induced killing of Eµ-Myc lymphoma cells in vitro. And loss of PUMA (but not p21) impaired the regression of Eµ-Myc lymphomas induced by Nutlin3a in vivo.
Finally, the researchers found that PUMA contributed to Nutlin3a-induced apoptosis in myeloid leukemia, multiple myeloma, and Burkitt lymphoma cell lines.
The team noted that, because PUMA, a pro-apoptotic BH3-only protein, is critical for the therapeutic impact of Nutlin3a, it may be possible to boost the drug’s efficacy by combining it with BH3 mimetic drugs such as navitoclax or venetoclax. ![]()

Photo by Cameron Wells,
Walter and Eliza Hall
Institute of Medical Research
Research published in Cell Reports helps explain how the anticancer agent Nutlin3a fights lymphoma and other hematologic malignancies.
Nutlin3a is known to activate the tumor suppressor p53, but it hasn’t been clear exactly which p53 target genes are essential for the drug’s therapeutic activity.
The new research revealed that PUMA-mediated apoptosis—not p21-mediated cell-cycle arrest or senescence—is responsible for Nutlin3a’s therapeutic activity in lymphoid malignancies.
“By understanding how nutlins are killing cancer cells, we can begin to formulate their best possible use, including choosing the best partner drugs to combine the nutlins with,” said study author Andreas Strasser, PhD, of the Walter and Eliza Hall Institute of Medical Research in Parkville, Victoria, Australia.
With this study, Dr Strasser and his colleagues first found that Nutlin3a activates p53 target gene expression and causes cell-cycle arrest and apoptosis in non-transformed mouse lymphoid cells in vitro.
The team then showed that Nutlin3a-mediated killing of these cells requires PUMA but not p21. In vivo, loss of PUMA protected non-transformed mouse lymphoid cells against Nutlin3a-induced killing. Loss of p21 did not provide the same protection.
Next, the researchers found that malignant Eµ-Myc lymphoma cells were much more sensitive to Nutlin3a than were non-transformed lymphoid cells. In vitro experiments with Eµ-Myc lymphoma cells showed that Nutlin3a promotes p53 accumulation and downstream effector pathway activation.
As in previous experiments, PUMA (not p21) proved critical for Nutlin3a-induced killing of Eµ-Myc lymphoma cells in vitro. And loss of PUMA (but not p21) impaired the regression of Eµ-Myc lymphomas induced by Nutlin3a in vivo.
Finally, the researchers found that PUMA contributed to Nutlin3a-induced apoptosis in myeloid leukemia, multiple myeloma, and Burkitt lymphoma cell lines.
The team noted that, because PUMA, a pro-apoptotic BH3-only protein, is critical for the therapeutic impact of Nutlin3a, it may be possible to boost the drug’s efficacy by combining it with BH3 mimetic drugs such as navitoclax or venetoclax. ![]()

Photo by Cameron Wells,
Walter and Eliza Hall
Institute of Medical Research
Research published in Cell Reports helps explain how the anticancer agent Nutlin3a fights lymphoma and other hematologic malignancies.
Nutlin3a is known to activate the tumor suppressor p53, but it hasn’t been clear exactly which p53 target genes are essential for the drug’s therapeutic activity.
The new research revealed that PUMA-mediated apoptosis—not p21-mediated cell-cycle arrest or senescence—is responsible for Nutlin3a’s therapeutic activity in lymphoid malignancies.
“By understanding how nutlins are killing cancer cells, we can begin to formulate their best possible use, including choosing the best partner drugs to combine the nutlins with,” said study author Andreas Strasser, PhD, of the Walter and Eliza Hall Institute of Medical Research in Parkville, Victoria, Australia.
With this study, Dr Strasser and his colleagues first found that Nutlin3a activates p53 target gene expression and causes cell-cycle arrest and apoptosis in non-transformed mouse lymphoid cells in vitro.
The team then showed that Nutlin3a-mediated killing of these cells requires PUMA but not p21. In vivo, loss of PUMA protected non-transformed mouse lymphoid cells against Nutlin3a-induced killing. Loss of p21 did not provide the same protection.
Next, the researchers found that malignant Eµ-Myc lymphoma cells were much more sensitive to Nutlin3a than were non-transformed lymphoid cells. In vitro experiments with Eµ-Myc lymphoma cells showed that Nutlin3a promotes p53 accumulation and downstream effector pathway activation.
As in previous experiments, PUMA (not p21) proved critical for Nutlin3a-induced killing of Eµ-Myc lymphoma cells in vitro. And loss of PUMA (but not p21) impaired the regression of Eµ-Myc lymphomas induced by Nutlin3a in vivo.
Finally, the researchers found that PUMA contributed to Nutlin3a-induced apoptosis in myeloid leukemia, multiple myeloma, and Burkitt lymphoma cell lines.
The team noted that, because PUMA, a pro-apoptotic BH3-only protein, is critical for the therapeutic impact of Nutlin3a, it may be possible to boost the drug’s efficacy by combining it with BH3 mimetic drugs such as navitoclax or venetoclax. ![]()
Teens’ weight, height linked to risk of NHL

A new analysis indicates that having a higher body weight and taller stature during adolescence may increase the risk of developing non-Hodgkin lymphoma (NHL).
Global rates of NHL have been on the rise in recent years, and research suggests that rising rates of obesity may be contributing to this trend.
With this in mind, investigators examined whether adolescent weight and height might be associated with the risk of developing NHL later in life.
They reported their results in Cancer.
The study included 2,352,988 subjects, ages 16 to 19, who were examined between 1967 and 2011. Their information was linked to the Israel National Cancer Registry, which included 4021 cases of NHL from 1967 through 2012.
The data showed that being overweight or obese in adolescence was associated with an increased risk of NHL later in life. When compared to adolescents of normal weight, the hazard ratio (HR) was 1.25 for subjects who were overweight or obese. The HR for underweight individuals was 0.98.
Being overweight or obese in adolescence was a significant predictor for marginal zone lymphoma (HR=1.70), primary cutaneous lymphoma (PCL, HR=1.44), and diffuse large B-cell lymphoma (DLBCL, HR=1.31). Excess weight was a borderline predictor for follicular lymphoma (HR=1.28).
“It is important to be aware that overweight and obesity are not risk factors only for diabetes and cardiovascular disease but also for lymphomas,” said study author Merav Leiba, MD, of the Sheba Medical Center in Israel.
Dr Leiba and her colleagues also observed an increased risk of NHL corresponding with increases in subjects’ height. When compared with the mid-range height category, shorter individuals had an HR of 1.25, and the tallest individuals had an HR of 1.28.
The strongest associations between taller height and NHL were observed for primary cutaneous lymphoma and diffuse large B-cell lymphoma. The HRs for the tallest group, compared to the shortest group, were 3.19 for PCL and 2.21 for DLBCL.
The investigators said additional research is needed to help explain the links between height, weight, and NHL. ![]()

A new analysis indicates that having a higher body weight and taller stature during adolescence may increase the risk of developing non-Hodgkin lymphoma (NHL).
Global rates of NHL have been on the rise in recent years, and research suggests that rising rates of obesity may be contributing to this trend.
With this in mind, investigators examined whether adolescent weight and height might be associated with the risk of developing NHL later in life.
They reported their results in Cancer.
The study included 2,352,988 subjects, ages 16 to 19, who were examined between 1967 and 2011. Their information was linked to the Israel National Cancer Registry, which included 4021 cases of NHL from 1967 through 2012.
The data showed that being overweight or obese in adolescence was associated with an increased risk of NHL later in life. When compared to adolescents of normal weight, the hazard ratio (HR) was 1.25 for subjects who were overweight or obese. The HR for underweight individuals was 0.98.
Being overweight or obese in adolescence was a significant predictor for marginal zone lymphoma (HR=1.70), primary cutaneous lymphoma (PCL, HR=1.44), and diffuse large B-cell lymphoma (DLBCL, HR=1.31). Excess weight was a borderline predictor for follicular lymphoma (HR=1.28).
“It is important to be aware that overweight and obesity are not risk factors only for diabetes and cardiovascular disease but also for lymphomas,” said study author Merav Leiba, MD, of the Sheba Medical Center in Israel.
Dr Leiba and her colleagues also observed an increased risk of NHL corresponding with increases in subjects’ height. When compared with the mid-range height category, shorter individuals had an HR of 1.25, and the tallest individuals had an HR of 1.28.
The strongest associations between taller height and NHL were observed for primary cutaneous lymphoma and diffuse large B-cell lymphoma. The HRs for the tallest group, compared to the shortest group, were 3.19 for PCL and 2.21 for DLBCL.
The investigators said additional research is needed to help explain the links between height, weight, and NHL. ![]()

A new analysis indicates that having a higher body weight and taller stature during adolescence may increase the risk of developing non-Hodgkin lymphoma (NHL).
Global rates of NHL have been on the rise in recent years, and research suggests that rising rates of obesity may be contributing to this trend.
With this in mind, investigators examined whether adolescent weight and height might be associated with the risk of developing NHL later in life.
They reported their results in Cancer.
The study included 2,352,988 subjects, ages 16 to 19, who were examined between 1967 and 2011. Their information was linked to the Israel National Cancer Registry, which included 4021 cases of NHL from 1967 through 2012.
The data showed that being overweight or obese in adolescence was associated with an increased risk of NHL later in life. When compared to adolescents of normal weight, the hazard ratio (HR) was 1.25 for subjects who were overweight or obese. The HR for underweight individuals was 0.98.
Being overweight or obese in adolescence was a significant predictor for marginal zone lymphoma (HR=1.70), primary cutaneous lymphoma (PCL, HR=1.44), and diffuse large B-cell lymphoma (DLBCL, HR=1.31). Excess weight was a borderline predictor for follicular lymphoma (HR=1.28).
“It is important to be aware that overweight and obesity are not risk factors only for diabetes and cardiovascular disease but also for lymphomas,” said study author Merav Leiba, MD, of the Sheba Medical Center in Israel.
Dr Leiba and her colleagues also observed an increased risk of NHL corresponding with increases in subjects’ height. When compared with the mid-range height category, shorter individuals had an HR of 1.25, and the tallest individuals had an HR of 1.28.
The strongest associations between taller height and NHL were observed for primary cutaneous lymphoma and diffuse large B-cell lymphoma. The HRs for the tallest group, compared to the shortest group, were 3.19 for PCL and 2.21 for DLBCL.
The investigators said additional research is needed to help explain the links between height, weight, and NHL. ![]()
Panobinostat plus bortezomib and dexamethasone improved outcomes in previously treated multiple myeloma
The addition of panobinostat to bortezomib and dexamethasone (PAN-BTZ-Dex) improved outcomes in patients with multiple myeloma who had received prior treatment with immunomodulatory drugs (IMiDs), prior bortezomib plus an IMiD, and two or more prior regimens including bortezomib and an IMiD.
Subgroup analysis of the PANORAMA phase III trial of patients with relapsed or relapsed and refractory multiple myeloma (MM) reported median progression-free survival (PFS) with PAN-BTZ-Dex versus placebo-BTZ-Dex (Pbo-BTZ-Dex) in groups defined by prior treatment: prior IMiD (12.3 vs. 7.4 months; hazard ratio, 0.54; 95% confidence interval, 0.43-0.68), prior bortezomib plus IMiD (10.6 vs. 5.8 months; HR, 0.52; 95% CI, 0.36-0.76), and two or more prior regimens including bortezomib and an IMiD (12.5 vs. 4.7 months; HR, 0.47; 95% CI, 0.31-0.72) (Blood. 2016 Feb 11. doi: 10.1182/blood-2015-09-665018).
The greatest difference in median PFS between the panobinostat and placebo arms (7.8 months) was observed among patients who had received two or more prior regimens including bortezomib and an IMiD, a population with a poorer prognosis and an urgent unmet need, according to investigators.
“Panobinostat represents a novel addition to the MM treatment armamentarium by introducing an agent with a novel mechanism of action. Novel agents are needed to address the ongoing unmet need in patients who progress on bortezomib and IMiDs as a strategy to overcome therapeutic resistance,” wrote Dr. Paul G. Richardson of the Dana Farber Cancer Institute, Boston, and his colleagues, adding that since the deacetylase inhibitor “acts on distinct epigenetic and protein metabolism pathways, it is uniquely suited to provide benefit in patients previously treated with proteasome inhibitors and/or IMiDs.”
The analysis examined treatment outcomes from the PANORAMA trial, including 485 patients who had received prior IMiD (63% of total population), 193 who received prior bortezomib plus IMiD (25% of total population), and 147 who received two or more prior regimens including bortezomib and an IMiD (19% of total population).
Common adverse events (AEs) by treatment subgroups were similar to those of the overall trial population, which were increased in the PAN-BTZ-Dex compared with the placebo arm. The most common nonhematologic AE was diarrhea and the most common hematologic abnormality was thrombocytopenia.
The PANORAMA 1 study was funded by Novartis Pharmaceuticals. Dr. Richardson reported having no disclosures. Several of his coauthors reported ties to industry.
The addition of panobinostat to bortezomib and dexamethasone (PAN-BTZ-Dex) improved outcomes in patients with multiple myeloma who had received prior treatment with immunomodulatory drugs (IMiDs), prior bortezomib plus an IMiD, and two or more prior regimens including bortezomib and an IMiD.
Subgroup analysis of the PANORAMA phase III trial of patients with relapsed or relapsed and refractory multiple myeloma (MM) reported median progression-free survival (PFS) with PAN-BTZ-Dex versus placebo-BTZ-Dex (Pbo-BTZ-Dex) in groups defined by prior treatment: prior IMiD (12.3 vs. 7.4 months; hazard ratio, 0.54; 95% confidence interval, 0.43-0.68), prior bortezomib plus IMiD (10.6 vs. 5.8 months; HR, 0.52; 95% CI, 0.36-0.76), and two or more prior regimens including bortezomib and an IMiD (12.5 vs. 4.7 months; HR, 0.47; 95% CI, 0.31-0.72) (Blood. 2016 Feb 11. doi: 10.1182/blood-2015-09-665018).
The greatest difference in median PFS between the panobinostat and placebo arms (7.8 months) was observed among patients who had received two or more prior regimens including bortezomib and an IMiD, a population with a poorer prognosis and an urgent unmet need, according to investigators.
“Panobinostat represents a novel addition to the MM treatment armamentarium by introducing an agent with a novel mechanism of action. Novel agents are needed to address the ongoing unmet need in patients who progress on bortezomib and IMiDs as a strategy to overcome therapeutic resistance,” wrote Dr. Paul G. Richardson of the Dana Farber Cancer Institute, Boston, and his colleagues, adding that since the deacetylase inhibitor “acts on distinct epigenetic and protein metabolism pathways, it is uniquely suited to provide benefit in patients previously treated with proteasome inhibitors and/or IMiDs.”
The analysis examined treatment outcomes from the PANORAMA trial, including 485 patients who had received prior IMiD (63% of total population), 193 who received prior bortezomib plus IMiD (25% of total population), and 147 who received two or more prior regimens including bortezomib and an IMiD (19% of total population).
Common adverse events (AEs) by treatment subgroups were similar to those of the overall trial population, which were increased in the PAN-BTZ-Dex compared with the placebo arm. The most common nonhematologic AE was diarrhea and the most common hematologic abnormality was thrombocytopenia.
The PANORAMA 1 study was funded by Novartis Pharmaceuticals. Dr. Richardson reported having no disclosures. Several of his coauthors reported ties to industry.
The addition of panobinostat to bortezomib and dexamethasone (PAN-BTZ-Dex) improved outcomes in patients with multiple myeloma who had received prior treatment with immunomodulatory drugs (IMiDs), prior bortezomib plus an IMiD, and two or more prior regimens including bortezomib and an IMiD.
Subgroup analysis of the PANORAMA phase III trial of patients with relapsed or relapsed and refractory multiple myeloma (MM) reported median progression-free survival (PFS) with PAN-BTZ-Dex versus placebo-BTZ-Dex (Pbo-BTZ-Dex) in groups defined by prior treatment: prior IMiD (12.3 vs. 7.4 months; hazard ratio, 0.54; 95% confidence interval, 0.43-0.68), prior bortezomib plus IMiD (10.6 vs. 5.8 months; HR, 0.52; 95% CI, 0.36-0.76), and two or more prior regimens including bortezomib and an IMiD (12.5 vs. 4.7 months; HR, 0.47; 95% CI, 0.31-0.72) (Blood. 2016 Feb 11. doi: 10.1182/blood-2015-09-665018).
The greatest difference in median PFS between the panobinostat and placebo arms (7.8 months) was observed among patients who had received two or more prior regimens including bortezomib and an IMiD, a population with a poorer prognosis and an urgent unmet need, according to investigators.
“Panobinostat represents a novel addition to the MM treatment armamentarium by introducing an agent with a novel mechanism of action. Novel agents are needed to address the ongoing unmet need in patients who progress on bortezomib and IMiDs as a strategy to overcome therapeutic resistance,” wrote Dr. Paul G. Richardson of the Dana Farber Cancer Institute, Boston, and his colleagues, adding that since the deacetylase inhibitor “acts on distinct epigenetic and protein metabolism pathways, it is uniquely suited to provide benefit in patients previously treated with proteasome inhibitors and/or IMiDs.”
The analysis examined treatment outcomes from the PANORAMA trial, including 485 patients who had received prior IMiD (63% of total population), 193 who received prior bortezomib plus IMiD (25% of total population), and 147 who received two or more prior regimens including bortezomib and an IMiD (19% of total population).
Common adverse events (AEs) by treatment subgroups were similar to those of the overall trial population, which were increased in the PAN-BTZ-Dex compared with the placebo arm. The most common nonhematologic AE was diarrhea and the most common hematologic abnormality was thrombocytopenia.
The PANORAMA 1 study was funded by Novartis Pharmaceuticals. Dr. Richardson reported having no disclosures. Several of his coauthors reported ties to industry.
FROM BLOOD
Key clinical point: Panobinostat plus bortezomib and dexamethasone improved outcomes in patients with previously treated multiple myeloma, particularly those with two or more prior regimens including immunomodulatory drugs (IMiDs) and bortezomib.
Major finding: In patients with two or more prior regimens including IMiDs and bortezomib, progression-free survival for the panobinostat plus bortezomib and dexamethasone arm, compared with placebo plus bortezomib and dexamethasone arm, was 12.5 months (95% CI, 7.3-14.0) vs. 4.7 months (95% CI, 3.7-6.1); hazard ratio, 0.47 (95% CI, 0.31-0.72).
Data source: Subgroup analysis of the phase III PANORAMA trial evaluating panobinostat plus bortezomib and dexamethasone versus placebo plus bortezomib and dexamethasone in patients with relapsed or relapsed and refractory multiple myeloma.
Disclosures: The PANORAMA 1 study was funded by Novartis Pharmaceuticals. Dr. Richardson reported having no disclosures. Several of his coauthors reported ties to industry.
Risk of reproductive problems in male cancer survivors

A study of Norwegian men has revealed several factors that may help predict reproductive problems among males diagnosed with cancer before age 25.
Cancer type, age at diagnosis, and time period of diagnosis were all associated with the likelihood of paternity.
And although cancer survivors were less likely to reproduce and more likely to use assisted reproductive technology, their first offspring were no less healthy than the offspring of control subjects.
This research was published in the British Journal of Cancer.
The study began with all Norwegian men born between 1965 and 1985 (n=626,495). The researchers excluded men who emigrated or died before reaching fertile age, which left 2687 men who were diagnosed with cancer before age 25 and 607,668 cancer-free controls.
The most common cancers were testicular cancer (27%), CNS tumors (18%), lymphoma (15%), and leukemia (13%). Thirty percent of the cancer cases were diagnosed in childhood (0–14 years of age), 26% in adolescence (15–19 years), and 43% in young adulthood (20–24 years).
Nine percent (n=247) of cancer cases were diagnosed from 1965 through 1979, 50% (n=1346) from 1980 through 1994, and 41% (n=1094) from 1995 through 2007.
The cancer survivors were less likely to have children than controls, with a hazard ratio (HR) of 0.72.
The reduction in paternity was significant for survivors of leukemia (HR=0.78), lymphoma (HR=0.78), testicular cancer (HR=0.77), CNS tumors (HR=0.45), bone tumors (HR=0.69), sympathetic nervous system tumors (HR=0.50), and retinoblastoma (HR=0.52).
The reduction in paternity was also more pronounced for cancer patients diagnosed before 1995. The HR was 0.61 for those diagnosed from 1965 through 1979 and 0.66 for those diagnosed from 1980 through 1994.
Patients who were diagnosed before age 15 were less likely to reproduce as well, with an HR of 0.59.
“These finds are important for male cancer survivors, seeing as we can identify groups at risk of having reproduction problems,” said study author Maria Winther Gunnes, a PhD candidate at the University of Bergen in Norway.
Another finding was that male cancer survivors were more likely than controls to have pregnancies resulting from assisted reproductive technology. The relative risk was 3.32.
When assessed by cancer type, the relative risk was 2.29 for leukemia, 3.79 for lymphoma, 2.41 for CNS tumors, 5.71 for sympathetic nervous system tumors, 2.20 for renal tumors, 4.77 for bone tumors, 1.32 for soft tissue sarcomas, 3.70 for testicular cancer, 4.36 for thyroid carcinoma, and 0.45 for malignant melanoma.
There was no increased risk among the first offspring of cancer survivors for perinatal death, congenital malformations, being small for gestational age, low birth weight, or preterm birth.
“It is important to be able to assure young male cancer survivors that their illness and treatment will not have a negative impact on their own children,” Gunnes said. ![]()

A study of Norwegian men has revealed several factors that may help predict reproductive problems among males diagnosed with cancer before age 25.
Cancer type, age at diagnosis, and time period of diagnosis were all associated with the likelihood of paternity.
And although cancer survivors were less likely to reproduce and more likely to use assisted reproductive technology, their first offspring were no less healthy than the offspring of control subjects.
This research was published in the British Journal of Cancer.
The study began with all Norwegian men born between 1965 and 1985 (n=626,495). The researchers excluded men who emigrated or died before reaching fertile age, which left 2687 men who were diagnosed with cancer before age 25 and 607,668 cancer-free controls.
The most common cancers were testicular cancer (27%), CNS tumors (18%), lymphoma (15%), and leukemia (13%). Thirty percent of the cancer cases were diagnosed in childhood (0–14 years of age), 26% in adolescence (15–19 years), and 43% in young adulthood (20–24 years).
Nine percent (n=247) of cancer cases were diagnosed from 1965 through 1979, 50% (n=1346) from 1980 through 1994, and 41% (n=1094) from 1995 through 2007.
The cancer survivors were less likely to have children than controls, with a hazard ratio (HR) of 0.72.
The reduction in paternity was significant for survivors of leukemia (HR=0.78), lymphoma (HR=0.78), testicular cancer (HR=0.77), CNS tumors (HR=0.45), bone tumors (HR=0.69), sympathetic nervous system tumors (HR=0.50), and retinoblastoma (HR=0.52).
The reduction in paternity was also more pronounced for cancer patients diagnosed before 1995. The HR was 0.61 for those diagnosed from 1965 through 1979 and 0.66 for those diagnosed from 1980 through 1994.
Patients who were diagnosed before age 15 were less likely to reproduce as well, with an HR of 0.59.
“These finds are important for male cancer survivors, seeing as we can identify groups at risk of having reproduction problems,” said study author Maria Winther Gunnes, a PhD candidate at the University of Bergen in Norway.
Another finding was that male cancer survivors were more likely than controls to have pregnancies resulting from assisted reproductive technology. The relative risk was 3.32.
When assessed by cancer type, the relative risk was 2.29 for leukemia, 3.79 for lymphoma, 2.41 for CNS tumors, 5.71 for sympathetic nervous system tumors, 2.20 for renal tumors, 4.77 for bone tumors, 1.32 for soft tissue sarcomas, 3.70 for testicular cancer, 4.36 for thyroid carcinoma, and 0.45 for malignant melanoma.
There was no increased risk among the first offspring of cancer survivors for perinatal death, congenital malformations, being small for gestational age, low birth weight, or preterm birth.
“It is important to be able to assure young male cancer survivors that their illness and treatment will not have a negative impact on their own children,” Gunnes said. ![]()

A study of Norwegian men has revealed several factors that may help predict reproductive problems among males diagnosed with cancer before age 25.
Cancer type, age at diagnosis, and time period of diagnosis were all associated with the likelihood of paternity.
And although cancer survivors were less likely to reproduce and more likely to use assisted reproductive technology, their first offspring were no less healthy than the offspring of control subjects.
This research was published in the British Journal of Cancer.
The study began with all Norwegian men born between 1965 and 1985 (n=626,495). The researchers excluded men who emigrated or died before reaching fertile age, which left 2687 men who were diagnosed with cancer before age 25 and 607,668 cancer-free controls.
The most common cancers were testicular cancer (27%), CNS tumors (18%), lymphoma (15%), and leukemia (13%). Thirty percent of the cancer cases were diagnosed in childhood (0–14 years of age), 26% in adolescence (15–19 years), and 43% in young adulthood (20–24 years).
Nine percent (n=247) of cancer cases were diagnosed from 1965 through 1979, 50% (n=1346) from 1980 through 1994, and 41% (n=1094) from 1995 through 2007.
The cancer survivors were less likely to have children than controls, with a hazard ratio (HR) of 0.72.
The reduction in paternity was significant for survivors of leukemia (HR=0.78), lymphoma (HR=0.78), testicular cancer (HR=0.77), CNS tumors (HR=0.45), bone tumors (HR=0.69), sympathetic nervous system tumors (HR=0.50), and retinoblastoma (HR=0.52).
The reduction in paternity was also more pronounced for cancer patients diagnosed before 1995. The HR was 0.61 for those diagnosed from 1965 through 1979 and 0.66 for those diagnosed from 1980 through 1994.
Patients who were diagnosed before age 15 were less likely to reproduce as well, with an HR of 0.59.
“These finds are important for male cancer survivors, seeing as we can identify groups at risk of having reproduction problems,” said study author Maria Winther Gunnes, a PhD candidate at the University of Bergen in Norway.
Another finding was that male cancer survivors were more likely than controls to have pregnancies resulting from assisted reproductive technology. The relative risk was 3.32.
When assessed by cancer type, the relative risk was 2.29 for leukemia, 3.79 for lymphoma, 2.41 for CNS tumors, 5.71 for sympathetic nervous system tumors, 2.20 for renal tumors, 4.77 for bone tumors, 1.32 for soft tissue sarcomas, 3.70 for testicular cancer, 4.36 for thyroid carcinoma, and 0.45 for malignant melanoma.
There was no increased risk among the first offspring of cancer survivors for perinatal death, congenital malformations, being small for gestational age, low birth weight, or preterm birth.
“It is important to be able to assure young male cancer survivors that their illness and treatment will not have a negative impact on their own children,” Gunnes said. ![]()