User login
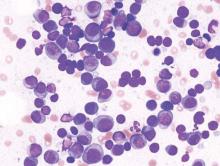
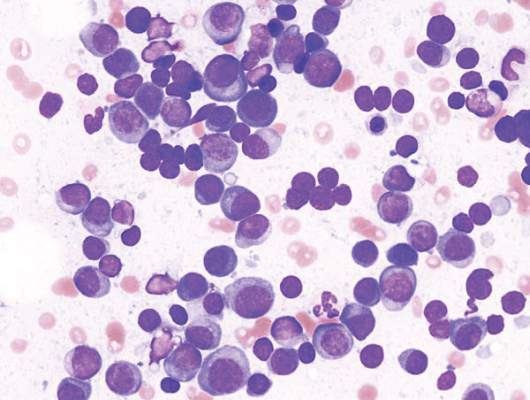
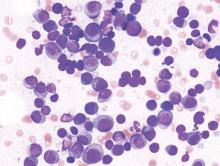
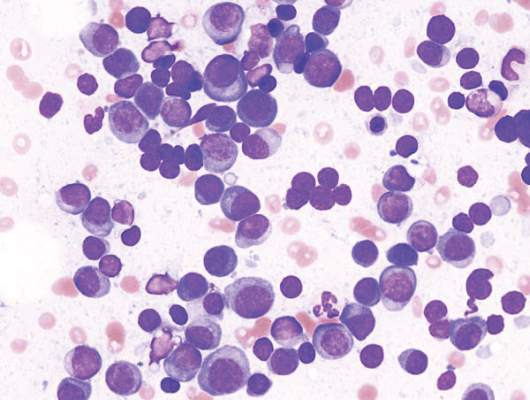
Lenalidomide-dexamethasone yields similar PFS as triplet regimens in elderly multiple myeloma patients
A comparison of lenalidomide-based treatments for multiple myeloma patients who were ineligible for stem cell transplantation showed similar progression-free survival (PFS) for two alkylator-containing triplet regimens and an alkylator-free doublet regimen but a higher risk of hematologic toxicity with a melphalan-prednisone-lenalidomide regimen.
For the triplet regimens, melphalan-prednisone-lenalidomide (MPR) and cyclophosphamide-prednisone-lenalidomide (CPR), the median PFS was 22 months, compared with 21 months for the doublet regimen lenalidomide plus low-dose dexamethasone (Rd). The hazard ratio (HR) was 0.906 (95% CI, 0.739-1.11; P = .344). The 4-year overall survival (OS) was 67% with triplet and 58% with doublet regimens (HR, 0.945; 95% CI, 0.700-1.274; P = .709) (Blood. 2016;127[9]:1102-8).
The major safety concern, according to the researchers, was the higher toxicity with MPR compared with CPR and Rd. The most frequent toxicities of grade 3 or more were hematologic, with at least one reported event in 68% of the MPR arm, 32% of CPR, and 29% or Rd patients (P less than .0001). In a post hoc analysis of safety according to patient fitness, the incidence of at least one hematologic adverse event ocurred in 75% of fit patients in the MPR arm occurred, 34% in CPR, and 29% in Rd; the incidence of at least one hematologic adverse event in intermediate fitness patients in the MPR arm was 61%, 33% in CPR, and 25% in Rd; in frail patients, 75% in MPR, 28% in CPR, and 3% in Rd (P = .001 for MPR vs. Rd and MPR vs. CPR). Nonhematologic adverse events were similar for the three groups and less than 10%.
Previous studies, such as the FIRST trial, showed the superiority of lenalidomide-containing regimens over standard treatments, but a question remained over the best drug to combine with lenalidomide – an alkylating agent or steroid. Separate analysis of the three arms further illustrated that the addition of an alkylating agent did not lead to better response or outcome. Median PFS for MPR, CPR, and Rd arms were 24, 20, and 21 months, respectively; 4-year OS rates were 65%, 68%, and 58%, respectively; overall response rates were 71%, 68%, and 74%, respectively.
Compared with the previous FIRST study, the less intense regimen in this study (Rd administered for only 9 months as induction treatment, followed by maintenance with lenalidomide at a lower dose) resulted in less hematological toxicity.
“This suggests that continuous treatment with Rd can be a valuable option for prolonging PFS and achieving a deeper response, and reducing the dose during maintenance can be a valuable strategy for improving tolerability,” wrote Dr. Valeria Magarotto of the myeloma unit in the division of hematology at the University of Torino (Italy), and colleagues. They added, “A more intensive induction treatment with Rd administered for a limited duration (9 months) followed by a less intensive continuous treatment with lenalidomide alone seems to be a sensible and effective choice.”
The phase III trial included 654 patients with newly diagnosed multiple myeloma who were ineligible for stem cell transplantation due to advanced age (65 years and older) or comorbidities. Patients were randomized to receive MPR (n = 217), CPR (n = 220), or Rd (n = 217).
A comparison of lenalidomide-based treatments for multiple myeloma patients who were ineligible for stem cell transplantation showed similar progression-free survival (PFS) for two alkylator-containing triplet regimens and an alkylator-free doublet regimen but a higher risk of hematologic toxicity with a melphalan-prednisone-lenalidomide regimen.
For the triplet regimens, melphalan-prednisone-lenalidomide (MPR) and cyclophosphamide-prednisone-lenalidomide (CPR), the median PFS was 22 months, compared with 21 months for the doublet regimen lenalidomide plus low-dose dexamethasone (Rd). The hazard ratio (HR) was 0.906 (95% CI, 0.739-1.11; P = .344). The 4-year overall survival (OS) was 67% with triplet and 58% with doublet regimens (HR, 0.945; 95% CI, 0.700-1.274; P = .709) (Blood. 2016;127[9]:1102-8).
The major safety concern, according to the researchers, was the higher toxicity with MPR compared with CPR and Rd. The most frequent toxicities of grade 3 or more were hematologic, with at least one reported event in 68% of the MPR arm, 32% of CPR, and 29% or Rd patients (P less than .0001). In a post hoc analysis of safety according to patient fitness, the incidence of at least one hematologic adverse event ocurred in 75% of fit patients in the MPR arm occurred, 34% in CPR, and 29% in Rd; the incidence of at least one hematologic adverse event in intermediate fitness patients in the MPR arm was 61%, 33% in CPR, and 25% in Rd; in frail patients, 75% in MPR, 28% in CPR, and 3% in Rd (P = .001 for MPR vs. Rd and MPR vs. CPR). Nonhematologic adverse events were similar for the three groups and less than 10%.
Previous studies, such as the FIRST trial, showed the superiority of lenalidomide-containing regimens over standard treatments, but a question remained over the best drug to combine with lenalidomide – an alkylating agent or steroid. Separate analysis of the three arms further illustrated that the addition of an alkylating agent did not lead to better response or outcome. Median PFS for MPR, CPR, and Rd arms were 24, 20, and 21 months, respectively; 4-year OS rates were 65%, 68%, and 58%, respectively; overall response rates were 71%, 68%, and 74%, respectively.
Compared with the previous FIRST study, the less intense regimen in this study (Rd administered for only 9 months as induction treatment, followed by maintenance with lenalidomide at a lower dose) resulted in less hematological toxicity.
“This suggests that continuous treatment with Rd can be a valuable option for prolonging PFS and achieving a deeper response, and reducing the dose during maintenance can be a valuable strategy for improving tolerability,” wrote Dr. Valeria Magarotto of the myeloma unit in the division of hematology at the University of Torino (Italy), and colleagues. They added, “A more intensive induction treatment with Rd administered for a limited duration (9 months) followed by a less intensive continuous treatment with lenalidomide alone seems to be a sensible and effective choice.”
The phase III trial included 654 patients with newly diagnosed multiple myeloma who were ineligible for stem cell transplantation due to advanced age (65 years and older) or comorbidities. Patients were randomized to receive MPR (n = 217), CPR (n = 220), or Rd (n = 217).
A comparison of lenalidomide-based treatments for multiple myeloma patients who were ineligible for stem cell transplantation showed similar progression-free survival (PFS) for two alkylator-containing triplet regimens and an alkylator-free doublet regimen but a higher risk of hematologic toxicity with a melphalan-prednisone-lenalidomide regimen.
For the triplet regimens, melphalan-prednisone-lenalidomide (MPR) and cyclophosphamide-prednisone-lenalidomide (CPR), the median PFS was 22 months, compared with 21 months for the doublet regimen lenalidomide plus low-dose dexamethasone (Rd). The hazard ratio (HR) was 0.906 (95% CI, 0.739-1.11; P = .344). The 4-year overall survival (OS) was 67% with triplet and 58% with doublet regimens (HR, 0.945; 95% CI, 0.700-1.274; P = .709) (Blood. 2016;127[9]:1102-8).
The major safety concern, according to the researchers, was the higher toxicity with MPR compared with CPR and Rd. The most frequent toxicities of grade 3 or more were hematologic, with at least one reported event in 68% of the MPR arm, 32% of CPR, and 29% or Rd patients (P less than .0001). In a post hoc analysis of safety according to patient fitness, the incidence of at least one hematologic adverse event ocurred in 75% of fit patients in the MPR arm occurred, 34% in CPR, and 29% in Rd; the incidence of at least one hematologic adverse event in intermediate fitness patients in the MPR arm was 61%, 33% in CPR, and 25% in Rd; in frail patients, 75% in MPR, 28% in CPR, and 3% in Rd (P = .001 for MPR vs. Rd and MPR vs. CPR). Nonhematologic adverse events were similar for the three groups and less than 10%.
Previous studies, such as the FIRST trial, showed the superiority of lenalidomide-containing regimens over standard treatments, but a question remained over the best drug to combine with lenalidomide – an alkylating agent or steroid. Separate analysis of the three arms further illustrated that the addition of an alkylating agent did not lead to better response or outcome. Median PFS for MPR, CPR, and Rd arms were 24, 20, and 21 months, respectively; 4-year OS rates were 65%, 68%, and 58%, respectively; overall response rates were 71%, 68%, and 74%, respectively.
Compared with the previous FIRST study, the less intense regimen in this study (Rd administered for only 9 months as induction treatment, followed by maintenance with lenalidomide at a lower dose) resulted in less hematological toxicity.
“This suggests that continuous treatment with Rd can be a valuable option for prolonging PFS and achieving a deeper response, and reducing the dose during maintenance can be a valuable strategy for improving tolerability,” wrote Dr. Valeria Magarotto of the myeloma unit in the division of hematology at the University of Torino (Italy), and colleagues. They added, “A more intensive induction treatment with Rd administered for a limited duration (9 months) followed by a less intensive continuous treatment with lenalidomide alone seems to be a sensible and effective choice.”
The phase III trial included 654 patients with newly diagnosed multiple myeloma who were ineligible for stem cell transplantation due to advanced age (65 years and older) or comorbidities. Patients were randomized to receive MPR (n = 217), CPR (n = 220), or Rd (n = 217).
FROM BLOOD
Key clinical point: In elderly patients with newly diagnosed multiple myeloma, progression-free survival was similar for alkylator-containing triplet regimens and an alkylator-free doublet regimen, but the doublet resulted in less hematologic toxicity.
Major finding: Median PFS for MPR, CPR and Rd arms were 24, 20, and 21 months, respectively; 4-year OS rates were 65%, 68%, and 58%, respectively.
Data sources: Phase III trial of 654 patients randomized to receive melphalan-prednisone-lenalidomide (n = 217), cyclophosphamide-prednisone-lenalidomide (n = 220), or lenalidomide plus low-dose dexamethasone (n = 217).
Disclosures: Dr. Magarotto reported having no disclosures. Several of her coauthors reported financial ties to industry sources.
Class of drugs could treat B-cell malignancies
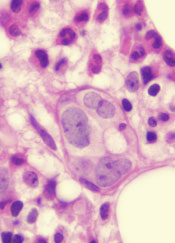
A class of drugs targeting a protein found in the endoplasmic reticulum could be effective against B-cell malignancies, according to a study published in Cancer Research.
The protein, STING, plays a critical role in producing type I interferons that help regulate the immune system.
Previous research suggested that STING agonists can improve immune responses when used in cancer immunotherapy or as vaccine adjuvants.
However, the way B cells respond to STING agonists was not well understood.
Chih-Chi Andrew Hu, PhD, of The Wistar Institute in Philadelphia, Pennsylvania, and his colleagues conducted a study to gain some insight.
The researchers found that normal B cells respond to STING agonists by undergoing mitochondria-mediated apoptosis, and STING agonists induce apoptosis in
malignant B cells through binding to STING.
STING agonists proved cytotoxic to B-cell leukemia, lymphoma, and multiple myeloma in vitro. But the drugs did not induce apoptosis in solid tumor malignancies or normal T cells.
The research also revealed that the IRE-1/XBP-1 stress response pathway is required for normal STING function. And B-cell leukemia, lymphoma, and myeloma require the IRE-1/XBP-1 pathway to be activated for survival.
Stimulation by STING agonists suppressed the IRE-1/XBP-1 pathway, which increased the level of apoptosis in malignant B cells.
The researchers confirmed these results in animal models, as treatment with STING agonists led to regression of chronic lymphocytic leukemia and multiple myeloma in mice.
“This specific cytotoxicity toward B cells strongly supports the use of STING agonists in the treatment of B-cell hematologic malignancies,” said Chih-Hang Anthony Tang, MD, PhD, of The Wistar Institute.
“We also believe that cytotoxicity in normal B cells can be managed with the administration of intravenous immunoglobulin that can help maintain normal levels of antibodies while treatment is being administered. This is something we plan on studying further.”
The Wistar Institute’s business development team is looking for a development partner for the advancement of novel STING agonists in treating B-cell hematologic malignancies. ![]()

A class of drugs targeting a protein found in the endoplasmic reticulum could be effective against B-cell malignancies, according to a study published in Cancer Research.
The protein, STING, plays a critical role in producing type I interferons that help regulate the immune system.
Previous research suggested that STING agonists can improve immune responses when used in cancer immunotherapy or as vaccine adjuvants.
However, the way B cells respond to STING agonists was not well understood.
Chih-Chi Andrew Hu, PhD, of The Wistar Institute in Philadelphia, Pennsylvania, and his colleagues conducted a study to gain some insight.
The researchers found that normal B cells respond to STING agonists by undergoing mitochondria-mediated apoptosis, and STING agonists induce apoptosis in
malignant B cells through binding to STING.
STING agonists proved cytotoxic to B-cell leukemia, lymphoma, and multiple myeloma in vitro. But the drugs did not induce apoptosis in solid tumor malignancies or normal T cells.
The research also revealed that the IRE-1/XBP-1 stress response pathway is required for normal STING function. And B-cell leukemia, lymphoma, and myeloma require the IRE-1/XBP-1 pathway to be activated for survival.
Stimulation by STING agonists suppressed the IRE-1/XBP-1 pathway, which increased the level of apoptosis in malignant B cells.
The researchers confirmed these results in animal models, as treatment with STING agonists led to regression of chronic lymphocytic leukemia and multiple myeloma in mice.
“This specific cytotoxicity toward B cells strongly supports the use of STING agonists in the treatment of B-cell hematologic malignancies,” said Chih-Hang Anthony Tang, MD, PhD, of The Wistar Institute.
“We also believe that cytotoxicity in normal B cells can be managed with the administration of intravenous immunoglobulin that can help maintain normal levels of antibodies while treatment is being administered. This is something we plan on studying further.”
The Wistar Institute’s business development team is looking for a development partner for the advancement of novel STING agonists in treating B-cell hematologic malignancies. ![]()

A class of drugs targeting a protein found in the endoplasmic reticulum could be effective against B-cell malignancies, according to a study published in Cancer Research.
The protein, STING, plays a critical role in producing type I interferons that help regulate the immune system.
Previous research suggested that STING agonists can improve immune responses when used in cancer immunotherapy or as vaccine adjuvants.
However, the way B cells respond to STING agonists was not well understood.
Chih-Chi Andrew Hu, PhD, of The Wistar Institute in Philadelphia, Pennsylvania, and his colleagues conducted a study to gain some insight.
The researchers found that normal B cells respond to STING agonists by undergoing mitochondria-mediated apoptosis, and STING agonists induce apoptosis in
malignant B cells through binding to STING.
STING agonists proved cytotoxic to B-cell leukemia, lymphoma, and multiple myeloma in vitro. But the drugs did not induce apoptosis in solid tumor malignancies or normal T cells.
The research also revealed that the IRE-1/XBP-1 stress response pathway is required for normal STING function. And B-cell leukemia, lymphoma, and myeloma require the IRE-1/XBP-1 pathway to be activated for survival.
Stimulation by STING agonists suppressed the IRE-1/XBP-1 pathway, which increased the level of apoptosis in malignant B cells.
The researchers confirmed these results in animal models, as treatment with STING agonists led to regression of chronic lymphocytic leukemia and multiple myeloma in mice.
“This specific cytotoxicity toward B cells strongly supports the use of STING agonists in the treatment of B-cell hematologic malignancies,” said Chih-Hang Anthony Tang, MD, PhD, of The Wistar Institute.
“We also believe that cytotoxicity in normal B cells can be managed with the administration of intravenous immunoglobulin that can help maintain normal levels of antibodies while treatment is being administered. This is something we plan on studying further.”
The Wistar Institute’s business development team is looking for a development partner for the advancement of novel STING agonists in treating B-cell hematologic malignancies. ![]()
HIV drug may overcome proteasome-inhibitor resistance in multiple myeloma
The protease inhibitor nelfinavir was used successfully to resensitize patients with proteasome inhibitor–refractory multiple myeloma for proteasome-inhibitor treatment, Dr. Christoph Driessen of Kantonsspital St. Gallen (Switzerland) and colleagues reported in Haematologica.
The researchers had hypothesized that nelfinavir would induce the unfolded protein response and would overcome proteasome-inhibitor resistance. The researchers determined a nelfinavir dose of 2,500 mg twice daily based on a dose-finding study in 12 patients with advanced hematologic malignancies.
In an exploratory extension cohort trial that followed, six patients with relapsed, bortezomib-refractory, lenalidomide-resistant myeloma were treated with 2,500 mg of nelfinavir twice daily in combination with bortezomib; three reached a partial response, two had a minor response, and one had progressive disease. All began the investigational therapy less than 2 months after progressing under therapies with bortezomib/bendamustine/dexamethasone, bortezomib/bendamustine, or bortezomib monotherapy, respectively, the researchers said (Haematologica March 2016;101:346-55).
The study protocol did not allow dexamethasone co-administration until completion of cycle three, when patients who did not achieve a minor response were allowed to co-administer 8 mg dexamethasone with bortezomib. Higher doses were excluded because of potential interactions with nelfinavir through the Cyp3A4 system.
Future research should assess whether bortezomib may be replaced by next-generation drugs like carfilzomib to avoid drug interactions and improve activity and whether nelfinavir may likewise be used in combination with novel oral proteasome inhibitors to boost their low single agent activity. “This ultimately suggests exploring the addition of HIV protease inhibitors to established combinations of proteasome inhibitors with immunomodulatory drugs, for example, in the carfilzomib/lenalidomide/dexamethasone regimen, one of the most powerful and tolerable regimens available to date for advanced multiple myeloma,” they concluded.
On Twitter @maryjodales
The protease inhibitor nelfinavir was used successfully to resensitize patients with proteasome inhibitor–refractory multiple myeloma for proteasome-inhibitor treatment, Dr. Christoph Driessen of Kantonsspital St. Gallen (Switzerland) and colleagues reported in Haematologica.
The researchers had hypothesized that nelfinavir would induce the unfolded protein response and would overcome proteasome-inhibitor resistance. The researchers determined a nelfinavir dose of 2,500 mg twice daily based on a dose-finding study in 12 patients with advanced hematologic malignancies.
In an exploratory extension cohort trial that followed, six patients with relapsed, bortezomib-refractory, lenalidomide-resistant myeloma were treated with 2,500 mg of nelfinavir twice daily in combination with bortezomib; three reached a partial response, two had a minor response, and one had progressive disease. All began the investigational therapy less than 2 months after progressing under therapies with bortezomib/bendamustine/dexamethasone, bortezomib/bendamustine, or bortezomib monotherapy, respectively, the researchers said (Haematologica March 2016;101:346-55).
The study protocol did not allow dexamethasone co-administration until completion of cycle three, when patients who did not achieve a minor response were allowed to co-administer 8 mg dexamethasone with bortezomib. Higher doses were excluded because of potential interactions with nelfinavir through the Cyp3A4 system.
Future research should assess whether bortezomib may be replaced by next-generation drugs like carfilzomib to avoid drug interactions and improve activity and whether nelfinavir may likewise be used in combination with novel oral proteasome inhibitors to boost their low single agent activity. “This ultimately suggests exploring the addition of HIV protease inhibitors to established combinations of proteasome inhibitors with immunomodulatory drugs, for example, in the carfilzomib/lenalidomide/dexamethasone regimen, one of the most powerful and tolerable regimens available to date for advanced multiple myeloma,” they concluded.
On Twitter @maryjodales
The protease inhibitor nelfinavir was used successfully to resensitize patients with proteasome inhibitor–refractory multiple myeloma for proteasome-inhibitor treatment, Dr. Christoph Driessen of Kantonsspital St. Gallen (Switzerland) and colleagues reported in Haematologica.
The researchers had hypothesized that nelfinavir would induce the unfolded protein response and would overcome proteasome-inhibitor resistance. The researchers determined a nelfinavir dose of 2,500 mg twice daily based on a dose-finding study in 12 patients with advanced hematologic malignancies.
In an exploratory extension cohort trial that followed, six patients with relapsed, bortezomib-refractory, lenalidomide-resistant myeloma were treated with 2,500 mg of nelfinavir twice daily in combination with bortezomib; three reached a partial response, two had a minor response, and one had progressive disease. All began the investigational therapy less than 2 months after progressing under therapies with bortezomib/bendamustine/dexamethasone, bortezomib/bendamustine, or bortezomib monotherapy, respectively, the researchers said (Haematologica March 2016;101:346-55).
The study protocol did not allow dexamethasone co-administration until completion of cycle three, when patients who did not achieve a minor response were allowed to co-administer 8 mg dexamethasone with bortezomib. Higher doses were excluded because of potential interactions with nelfinavir through the Cyp3A4 system.
Future research should assess whether bortezomib may be replaced by next-generation drugs like carfilzomib to avoid drug interactions and improve activity and whether nelfinavir may likewise be used in combination with novel oral proteasome inhibitors to boost their low single agent activity. “This ultimately suggests exploring the addition of HIV protease inhibitors to established combinations of proteasome inhibitors with immunomodulatory drugs, for example, in the carfilzomib/lenalidomide/dexamethasone regimen, one of the most powerful and tolerable regimens available to date for advanced multiple myeloma,” they concluded.
On Twitter @maryjodales
FROM HAEMATOLOGIA
Key clinical point: Adding HIV protease inhibitors to established combinations of proteasome inhibitors may improve outcomes in patients with multiple myeloma.
Major finding: When six patients with relapsed, bortezomib-refractory, lenalidomide-resistant myeloma were treated with 2,500 mg of nelfinavir twice daily in combination with bortezomib, three reached a partial response, two had a minor response, and disease progressed in one.
Data source: A dose-finding study in 12 patients with advanced hematologic malignancies and an exploratory extension cohort trial that followed.
Disclosures: None of the authors had financial conflicts of interest in relation to the study.
Allele associated with poor outcome in CLL

A form of the CYP3A7 gene is associated with poor outcomes in chronic lymphocytic leukemia (CLL) and other cancers, according to a study published in Cancer Research.
Among patients with CLL, breast cancer, or lung cancer, those with the CYP3A7*1C allele were more likely than those without it to experience disease progression or death.
Researchers believe this may be related to how patients metabolize treatment.
“The CYP3A7 gene encodes an enzyme that breaks down all sorts of naturally occurring substances—such as sex steroids like estrogen and testosterone—as well as a wide range of drugs that are used in the treatment of cancer,” said Olivia Fletcher, PhD, of The Institute of Cancer Research in London, UK.
“The CYP3A7 gene is normally turned on in an embryo and then turned off shortly after a baby is born, but individuals who have 1 or more copies of the CYP3A7*1C form of the gene turn on their CYP3A7 gene in adult life.”
“We found that individuals with breast cancer, lung cancer, or CLL who carry 1 or more copies of the CYP3A7*1C allele tend to have worse outcomes. One possibility is that these patients break down the drugs that they are given to treat their cancer too fast. However, further independent studies that replicate our findings in larger numbers of patients and rule out biases are needed before we could recommend any changes to the treatment that cancer patients with the CYP3A7*1C allele receive.”
To assess the impact of the CYP3A7*1C allele on patient outcomes, Dr Fletcher and her colleagues analyzed DNA samples from 1008 breast cancer patients, 1142 patients with lung cancer, and 356 patients with CLL.
The team looked for the presence of the single nucleotide polymorphism (SNP) rs45446698. Dr Fletcher explained that rs45446698 is 1 of 7 SNPs that cluster together to form the CYP3A7*1C allele.
The researchers found that, among CLL patients, rs45446698 (and, therefore, the CYP3A7*1C allele) was associated with a 62% increased risk of disease progression (P=0.03).
Among breast cancer patients, rs45446698 was associated with a 74% increased risk of breast cancer mortality (P=0.03). And among the lung cancer patients, the SNP was associated with a 43% increased risk of death from any cause (P=0.009).
The researchers also found borderline evidence of a statistical interaction between the CYP3A7*1C allele, treatment of patients with a cytotoxic agent that is a CYP3A substrate, and clinical outcome (P=0.06).
“Even though we did not see a statistically significant difference when stratifying patients by treatment with a CYP3A7 substrate, the fact that we see the same effect in 3 very different cancer types suggests to me that it is more likely to be something to do with treatment than the disease itself,” Dr Fletcher said.
“However, we are looking at ways of replicating these results in additional cohorts of patients and types of cancer, as well as overcoming the limitations of this study.”
Dr Fletcher explained that the main limitation of this study is that the researchers used samples and clinical information collected for other studies. So they did not have the same clinical information for each patient, and the samples were collected at different time points and for patients treated with various drugs.
She also noted that the researchers were not able to determine how quickly the patients broke down their treatments.
This study was supported by Sanofi-Aventis, Breast Cancer Now, Bloodwise, Cancer Research UK, the Medical Research Council, the Cridlan Trust, and the Helen Rollason Cancer Charity. The authors’ institutions received funding from the National Health Service of the United Kingdom. ![]()

A form of the CYP3A7 gene is associated with poor outcomes in chronic lymphocytic leukemia (CLL) and other cancers, according to a study published in Cancer Research.
Among patients with CLL, breast cancer, or lung cancer, those with the CYP3A7*1C allele were more likely than those without it to experience disease progression or death.
Researchers believe this may be related to how patients metabolize treatment.
“The CYP3A7 gene encodes an enzyme that breaks down all sorts of naturally occurring substances—such as sex steroids like estrogen and testosterone—as well as a wide range of drugs that are used in the treatment of cancer,” said Olivia Fletcher, PhD, of The Institute of Cancer Research in London, UK.
“The CYP3A7 gene is normally turned on in an embryo and then turned off shortly after a baby is born, but individuals who have 1 or more copies of the CYP3A7*1C form of the gene turn on their CYP3A7 gene in adult life.”
“We found that individuals with breast cancer, lung cancer, or CLL who carry 1 or more copies of the CYP3A7*1C allele tend to have worse outcomes. One possibility is that these patients break down the drugs that they are given to treat their cancer too fast. However, further independent studies that replicate our findings in larger numbers of patients and rule out biases are needed before we could recommend any changes to the treatment that cancer patients with the CYP3A7*1C allele receive.”
To assess the impact of the CYP3A7*1C allele on patient outcomes, Dr Fletcher and her colleagues analyzed DNA samples from 1008 breast cancer patients, 1142 patients with lung cancer, and 356 patients with CLL.
The team looked for the presence of the single nucleotide polymorphism (SNP) rs45446698. Dr Fletcher explained that rs45446698 is 1 of 7 SNPs that cluster together to form the CYP3A7*1C allele.
The researchers found that, among CLL patients, rs45446698 (and, therefore, the CYP3A7*1C allele) was associated with a 62% increased risk of disease progression (P=0.03).
Among breast cancer patients, rs45446698 was associated with a 74% increased risk of breast cancer mortality (P=0.03). And among the lung cancer patients, the SNP was associated with a 43% increased risk of death from any cause (P=0.009).
The researchers also found borderline evidence of a statistical interaction between the CYP3A7*1C allele, treatment of patients with a cytotoxic agent that is a CYP3A substrate, and clinical outcome (P=0.06).
“Even though we did not see a statistically significant difference when stratifying patients by treatment with a CYP3A7 substrate, the fact that we see the same effect in 3 very different cancer types suggests to me that it is more likely to be something to do with treatment than the disease itself,” Dr Fletcher said.
“However, we are looking at ways of replicating these results in additional cohorts of patients and types of cancer, as well as overcoming the limitations of this study.”
Dr Fletcher explained that the main limitation of this study is that the researchers used samples and clinical information collected for other studies. So they did not have the same clinical information for each patient, and the samples were collected at different time points and for patients treated with various drugs.
She also noted that the researchers were not able to determine how quickly the patients broke down their treatments.
This study was supported by Sanofi-Aventis, Breast Cancer Now, Bloodwise, Cancer Research UK, the Medical Research Council, the Cridlan Trust, and the Helen Rollason Cancer Charity. The authors’ institutions received funding from the National Health Service of the United Kingdom. ![]()

A form of the CYP3A7 gene is associated with poor outcomes in chronic lymphocytic leukemia (CLL) and other cancers, according to a study published in Cancer Research.
Among patients with CLL, breast cancer, or lung cancer, those with the CYP3A7*1C allele were more likely than those without it to experience disease progression or death.
Researchers believe this may be related to how patients metabolize treatment.
“The CYP3A7 gene encodes an enzyme that breaks down all sorts of naturally occurring substances—such as sex steroids like estrogen and testosterone—as well as a wide range of drugs that are used in the treatment of cancer,” said Olivia Fletcher, PhD, of The Institute of Cancer Research in London, UK.
“The CYP3A7 gene is normally turned on in an embryo and then turned off shortly after a baby is born, but individuals who have 1 or more copies of the CYP3A7*1C form of the gene turn on their CYP3A7 gene in adult life.”
“We found that individuals with breast cancer, lung cancer, or CLL who carry 1 or more copies of the CYP3A7*1C allele tend to have worse outcomes. One possibility is that these patients break down the drugs that they are given to treat their cancer too fast. However, further independent studies that replicate our findings in larger numbers of patients and rule out biases are needed before we could recommend any changes to the treatment that cancer patients with the CYP3A7*1C allele receive.”
To assess the impact of the CYP3A7*1C allele on patient outcomes, Dr Fletcher and her colleagues analyzed DNA samples from 1008 breast cancer patients, 1142 patients with lung cancer, and 356 patients with CLL.
The team looked for the presence of the single nucleotide polymorphism (SNP) rs45446698. Dr Fletcher explained that rs45446698 is 1 of 7 SNPs that cluster together to form the CYP3A7*1C allele.
The researchers found that, among CLL patients, rs45446698 (and, therefore, the CYP3A7*1C allele) was associated with a 62% increased risk of disease progression (P=0.03).
Among breast cancer patients, rs45446698 was associated with a 74% increased risk of breast cancer mortality (P=0.03). And among the lung cancer patients, the SNP was associated with a 43% increased risk of death from any cause (P=0.009).
The researchers also found borderline evidence of a statistical interaction between the CYP3A7*1C allele, treatment of patients with a cytotoxic agent that is a CYP3A substrate, and clinical outcome (P=0.06).
“Even though we did not see a statistically significant difference when stratifying patients by treatment with a CYP3A7 substrate, the fact that we see the same effect in 3 very different cancer types suggests to me that it is more likely to be something to do with treatment than the disease itself,” Dr Fletcher said.
“However, we are looking at ways of replicating these results in additional cohorts of patients and types of cancer, as well as overcoming the limitations of this study.”
Dr Fletcher explained that the main limitation of this study is that the researchers used samples and clinical information collected for other studies. So they did not have the same clinical information for each patient, and the samples were collected at different time points and for patients treated with various drugs.
She also noted that the researchers were not able to determine how quickly the patients broke down their treatments.
This study was supported by Sanofi-Aventis, Breast Cancer Now, Bloodwise, Cancer Research UK, the Medical Research Council, the Cridlan Trust, and the Helen Rollason Cancer Charity. The authors’ institutions received funding from the National Health Service of the United Kingdom. ![]()
Follicular lymphoma: Quantitative PET/CT measures for detecting bone marrow involvement
Quantifying bone marrow uptake of FDG (18fluorodeoxyglucose) improved the diagnostic accuracy of PET/CT for predicting bone marrow involvement in patients with follicular lymphoma, based on the results of a retrospective study.
Visual evidence of focal increased uptake on PET/CT indicates marrow involvement in follicular lymphoma; however, diffuse uptake is a nonspecific finding. Measuring the mean bone marrow standardized uptake value (BM SUV mean) improves PET/CT diagnostic accuracy, Dr. Chava Perry and his colleagues at Tel Aviv Sourasky Medical Center reported in Medicine [(Baltimore). 2016 Mar;95(9):e2910].
The researchers evaluated 68 consecutive patients with follicular lymphoma; 16 had bone marrow involvement – 13 had biopsy-proven involvement and 3 had a negative biopsy with increased medullary uptake that normalized after treatment. BM FDG uptake was diffuse in 8 of them and focal in the other 8.
While focal increased uptake is indicative of bone marrow involvement, diffuse uptake can be associated with false-positive results, as it was in the case of 17 patients (32.7% of those with diffuse uptake). Overall, visual assessment of scan results had a negative predictive value of 100% and a positive predictive value (PPV) of 48.5%.
On a quantitative assessment, however, BM SUV mean was significantly higher in patients with bone marrow involvement (SUV mean of 3.7 [1.7-6] vs. 1.4 [0.4-2.65]; P less than .001). On the receiver operator curve (ROC) analysis, a BM SUV mean exceeding 2.7 had a positive predictive value of 100% for bone marrow involvement (sensitivity of 68%). A BM SUV mean less than 1.7 had an negative predictive value of 100% (specificity of 73%).
A mean standardized uptake value (BM SUV mean) below 1.7 may spare the need for bone marrow biopsy while a BM SUV mean above 2.7 is compatible with bone marrow involvement, although biopsy may still be recommended to exclude large cell transformation, the researchers concluded.
On Twitter @maryjodales
Quantifying bone marrow uptake of FDG (18fluorodeoxyglucose) improved the diagnostic accuracy of PET/CT for predicting bone marrow involvement in patients with follicular lymphoma, based on the results of a retrospective study.
Visual evidence of focal increased uptake on PET/CT indicates marrow involvement in follicular lymphoma; however, diffuse uptake is a nonspecific finding. Measuring the mean bone marrow standardized uptake value (BM SUV mean) improves PET/CT diagnostic accuracy, Dr. Chava Perry and his colleagues at Tel Aviv Sourasky Medical Center reported in Medicine [(Baltimore). 2016 Mar;95(9):e2910].
The researchers evaluated 68 consecutive patients with follicular lymphoma; 16 had bone marrow involvement – 13 had biopsy-proven involvement and 3 had a negative biopsy with increased medullary uptake that normalized after treatment. BM FDG uptake was diffuse in 8 of them and focal in the other 8.
While focal increased uptake is indicative of bone marrow involvement, diffuse uptake can be associated with false-positive results, as it was in the case of 17 patients (32.7% of those with diffuse uptake). Overall, visual assessment of scan results had a negative predictive value of 100% and a positive predictive value (PPV) of 48.5%.
On a quantitative assessment, however, BM SUV mean was significantly higher in patients with bone marrow involvement (SUV mean of 3.7 [1.7-6] vs. 1.4 [0.4-2.65]; P less than .001). On the receiver operator curve (ROC) analysis, a BM SUV mean exceeding 2.7 had a positive predictive value of 100% for bone marrow involvement (sensitivity of 68%). A BM SUV mean less than 1.7 had an negative predictive value of 100% (specificity of 73%).
A mean standardized uptake value (BM SUV mean) below 1.7 may spare the need for bone marrow biopsy while a BM SUV mean above 2.7 is compatible with bone marrow involvement, although biopsy may still be recommended to exclude large cell transformation, the researchers concluded.
On Twitter @maryjodales
Quantifying bone marrow uptake of FDG (18fluorodeoxyglucose) improved the diagnostic accuracy of PET/CT for predicting bone marrow involvement in patients with follicular lymphoma, based on the results of a retrospective study.
Visual evidence of focal increased uptake on PET/CT indicates marrow involvement in follicular lymphoma; however, diffuse uptake is a nonspecific finding. Measuring the mean bone marrow standardized uptake value (BM SUV mean) improves PET/CT diagnostic accuracy, Dr. Chava Perry and his colleagues at Tel Aviv Sourasky Medical Center reported in Medicine [(Baltimore). 2016 Mar;95(9):e2910].
The researchers evaluated 68 consecutive patients with follicular lymphoma; 16 had bone marrow involvement – 13 had biopsy-proven involvement and 3 had a negative biopsy with increased medullary uptake that normalized after treatment. BM FDG uptake was diffuse in 8 of them and focal in the other 8.
While focal increased uptake is indicative of bone marrow involvement, diffuse uptake can be associated with false-positive results, as it was in the case of 17 patients (32.7% of those with diffuse uptake). Overall, visual assessment of scan results had a negative predictive value of 100% and a positive predictive value (PPV) of 48.5%.
On a quantitative assessment, however, BM SUV mean was significantly higher in patients with bone marrow involvement (SUV mean of 3.7 [1.7-6] vs. 1.4 [0.4-2.65]; P less than .001). On the receiver operator curve (ROC) analysis, a BM SUV mean exceeding 2.7 had a positive predictive value of 100% for bone marrow involvement (sensitivity of 68%). A BM SUV mean less than 1.7 had an negative predictive value of 100% (specificity of 73%).
A mean standardized uptake value (BM SUV mean) below 1.7 may spare the need for bone marrow biopsy while a BM SUV mean above 2.7 is compatible with bone marrow involvement, although biopsy may still be recommended to exclude large cell transformation, the researchers concluded.
On Twitter @maryjodales
FROM MEDICINE
Key clinical point: Measuring the mean standardized uptake value of 18fluorodeoxyglucose in the bone marrow of patients with follicular lymphoma improves the diagnostic accuracy of PET/CT.
Major finding: In this study, diffuse uptake was associated with 17 (32.7%) false positive cases.
Data source: Retrospective study of 68 consecutive patients with follicular lymphoma.
Disclosures: The authors had no funding and conflicts of interest to disclose.
Germline mutations linked to hematologic malignancies

A new study suggests mutations in the gene DDX41 occur in families where hematologic malignancies are common.
Previous research showed that both germline and acquired DDX41 mutations occur in families with multiple cases of late-onset myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML).
The new study, published in Blood, has linked germline mutations in DDX41 to chronic myeloid leukemia and lymphomas as well.
“This is the first gene identified in families with lymphoma and represents a major breakthrough for the field,” said study author Hamish Scott, PhD, of the University of Adelaide in South Australia.
“Researchers are recognizing now that genetic predisposition to blood cancer is more common than previously thought, and our study shows the importance of taking a thorough family history at diagnosis.”
To conduct this study, Dr Scott and his colleagues screened 2 cohorts of families with a range of hematologic disorders (malignant and non-malignant). One cohort included 240 individuals from 93 families in Australia. The other included 246 individuals from 198 families in the US.
In all, 9 of the families (3%) had germline DDX41 mutations.
Three families carried the recurrent p.D140Gfs*2 mutation, which was linked to AML.
One family carried a germline mutation—p.R525H, c.1574G.A—that was previously described only as a somatic mutation at the time of progression to MDS or AML. In the current study, the mutation was again linked to MDS and AML.
Five families carried novel DDX41 mutations.
One of these mutations was a germline substitution—c.435-2_435-1delAGinsCA—that was linked to MDS in 1 family.
Two families had a missense start-loss substitution—c.3G.A, p.M1I—that was linked to MDS, AML, chronic myeloid leukemia, and non-Hodgkin lymphoma.
One family had a DDX41 missense variant—c.490C.T, p.R164W. This was linked to Hodgkin and non-Hodgkin lymphoma (including 3 cases of follicular lymphoma). There was a possible link to multiple myeloma as well, but the diagnosis could not be confirmed.
And 1 family had a missense mutation in the helicase domain—p.G530D—that was linked to AML.
“DDX41 is a new type of cancer predisposition gene, and we are still investigating its function,” Dr Scott noted.
“But it appears to have dual roles in regulating the correct expression of genes in the cell and also enabling the immune system to respond to threats such as bacteria and viruses, as well as the development of cancer cells. Immunotherapy is a promising approach for cancer treatment, and our research to understand the function of DDX41 will help design better therapies.” ![]()

A new study suggests mutations in the gene DDX41 occur in families where hematologic malignancies are common.
Previous research showed that both germline and acquired DDX41 mutations occur in families with multiple cases of late-onset myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML).
The new study, published in Blood, has linked germline mutations in DDX41 to chronic myeloid leukemia and lymphomas as well.
“This is the first gene identified in families with lymphoma and represents a major breakthrough for the field,” said study author Hamish Scott, PhD, of the University of Adelaide in South Australia.
“Researchers are recognizing now that genetic predisposition to blood cancer is more common than previously thought, and our study shows the importance of taking a thorough family history at diagnosis.”
To conduct this study, Dr Scott and his colleagues screened 2 cohorts of families with a range of hematologic disorders (malignant and non-malignant). One cohort included 240 individuals from 93 families in Australia. The other included 246 individuals from 198 families in the US.
In all, 9 of the families (3%) had germline DDX41 mutations.
Three families carried the recurrent p.D140Gfs*2 mutation, which was linked to AML.
One family carried a germline mutation—p.R525H, c.1574G.A—that was previously described only as a somatic mutation at the time of progression to MDS or AML. In the current study, the mutation was again linked to MDS and AML.
Five families carried novel DDX41 mutations.
One of these mutations was a germline substitution—c.435-2_435-1delAGinsCA—that was linked to MDS in 1 family.
Two families had a missense start-loss substitution—c.3G.A, p.M1I—that was linked to MDS, AML, chronic myeloid leukemia, and non-Hodgkin lymphoma.
One family had a DDX41 missense variant—c.490C.T, p.R164W. This was linked to Hodgkin and non-Hodgkin lymphoma (including 3 cases of follicular lymphoma). There was a possible link to multiple myeloma as well, but the diagnosis could not be confirmed.
And 1 family had a missense mutation in the helicase domain—p.G530D—that was linked to AML.
“DDX41 is a new type of cancer predisposition gene, and we are still investigating its function,” Dr Scott noted.
“But it appears to have dual roles in regulating the correct expression of genes in the cell and also enabling the immune system to respond to threats such as bacteria and viruses, as well as the development of cancer cells. Immunotherapy is a promising approach for cancer treatment, and our research to understand the function of DDX41 will help design better therapies.” ![]()

A new study suggests mutations in the gene DDX41 occur in families where hematologic malignancies are common.
Previous research showed that both germline and acquired DDX41 mutations occur in families with multiple cases of late-onset myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML).
The new study, published in Blood, has linked germline mutations in DDX41 to chronic myeloid leukemia and lymphomas as well.
“This is the first gene identified in families with lymphoma and represents a major breakthrough for the field,” said study author Hamish Scott, PhD, of the University of Adelaide in South Australia.
“Researchers are recognizing now that genetic predisposition to blood cancer is more common than previously thought, and our study shows the importance of taking a thorough family history at diagnosis.”
To conduct this study, Dr Scott and his colleagues screened 2 cohorts of families with a range of hematologic disorders (malignant and non-malignant). One cohort included 240 individuals from 93 families in Australia. The other included 246 individuals from 198 families in the US.
In all, 9 of the families (3%) had germline DDX41 mutations.
Three families carried the recurrent p.D140Gfs*2 mutation, which was linked to AML.
One family carried a germline mutation—p.R525H, c.1574G.A—that was previously described only as a somatic mutation at the time of progression to MDS or AML. In the current study, the mutation was again linked to MDS and AML.
Five families carried novel DDX41 mutations.
One of these mutations was a germline substitution—c.435-2_435-1delAGinsCA—that was linked to MDS in 1 family.
Two families had a missense start-loss substitution—c.3G.A, p.M1I—that was linked to MDS, AML, chronic myeloid leukemia, and non-Hodgkin lymphoma.
One family had a DDX41 missense variant—c.490C.T, p.R164W. This was linked to Hodgkin and non-Hodgkin lymphoma (including 3 cases of follicular lymphoma). There was a possible link to multiple myeloma as well, but the diagnosis could not be confirmed.
And 1 family had a missense mutation in the helicase domain—p.G530D—that was linked to AML.
“DDX41 is a new type of cancer predisposition gene, and we are still investigating its function,” Dr Scott noted.
“But it appears to have dual roles in regulating the correct expression of genes in the cell and also enabling the immune system to respond to threats such as bacteria and viruses, as well as the development of cancer cells. Immunotherapy is a promising approach for cancer treatment, and our research to understand the function of DDX41 will help design better therapies.” ![]()
FDA lifts partial clinical hold on pidilizumab

The US Food and Drug Administration (FDA) has lifted the partial clinical hold on the investigational new drug (IND) application for pidilizumab (MDV9300) in hematologic malignancies.
This means the phase 2 trial of pidilizumab in patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL), as well as other studies that cross-reference the IND for the drug, may now proceed.
The partial clinical hold on pidilizumab was not related to any safety concerns.
The FDA placed the hold because the company developing pidilizumab, Medivation Inc., determined that the drug is not an inhibitor of PD-1, as researchers previously thought.
The phase 2 trial of pidilizumab in DLBCL was launched in late 2015 but had not enrolled any patients before the FDA placed the partial clinical hold.
Patients who were receiving pidilizumab through investigator-sponsored trials have continued to receive treatment despite the hold, and those investigators have been told to update their protocols and informed consent documents to reflect that pidilizumab is not an anti-PD-1 antibody.
Medivation has likewise revised the investigator brochure, protocols, and informed consent documents related to the phase 2 trial of DLBCL patients.
The company said it is still trying to determine pidilizumab’s mechanism of action.
“We are delighted that the FDA has lifted the partial clinical hold and that we may proceed with our potentially pivotal trial in this area of high unmet medical need,” said David Hung, MD, founder, president, and chief executive officer of Medivation.
“As we move forward, we also are working to determine the compound’s exact binding mechanism which, we believe, modulates the body’s innate immune response and differentiates it from the heavily crowded immuno-oncology space that targets the adaptive side of immunity.”
Medivation said it intends to submit an amendment to the Chemistry, Manufacturing, and Controls section of the IND for pidilizumab to provide for larger manufacturing lot sizes to better support the current and planned clinical activities for the drug.
The company also said it plans to resume the phase 2 trial of pidilizumab in DLBLCL in the second half of this year.
The trial is expected to enroll approximately 180 patients who had an incomplete response to salvage therapy or autologous stem cell transplant for relapsed or refractory CD20+ DLBCL, transformed indolent lymphoma, or primary mediastinal B-cell lymphoma.
The patients will be assessed in 2 parallel cohorts of approximately 90 patients each. One cohort will enroll patients who have received an autologous stem cell transplant, and the other will enroll patients who have received salvage chemotherapy but are ineligible for transplant.
Pidilizumab will be given at a dose of 200 mg by intravenous infusion. The primary endpoint of the trial is best overall response rate. ![]()

The US Food and Drug Administration (FDA) has lifted the partial clinical hold on the investigational new drug (IND) application for pidilizumab (MDV9300) in hematologic malignancies.
This means the phase 2 trial of pidilizumab in patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL), as well as other studies that cross-reference the IND for the drug, may now proceed.
The partial clinical hold on pidilizumab was not related to any safety concerns.
The FDA placed the hold because the company developing pidilizumab, Medivation Inc., determined that the drug is not an inhibitor of PD-1, as researchers previously thought.
The phase 2 trial of pidilizumab in DLBCL was launched in late 2015 but had not enrolled any patients before the FDA placed the partial clinical hold.
Patients who were receiving pidilizumab through investigator-sponsored trials have continued to receive treatment despite the hold, and those investigators have been told to update their protocols and informed consent documents to reflect that pidilizumab is not an anti-PD-1 antibody.
Medivation has likewise revised the investigator brochure, protocols, and informed consent documents related to the phase 2 trial of DLBCL patients.
The company said it is still trying to determine pidilizumab’s mechanism of action.
“We are delighted that the FDA has lifted the partial clinical hold and that we may proceed with our potentially pivotal trial in this area of high unmet medical need,” said David Hung, MD, founder, president, and chief executive officer of Medivation.
“As we move forward, we also are working to determine the compound’s exact binding mechanism which, we believe, modulates the body’s innate immune response and differentiates it from the heavily crowded immuno-oncology space that targets the adaptive side of immunity.”
Medivation said it intends to submit an amendment to the Chemistry, Manufacturing, and Controls section of the IND for pidilizumab to provide for larger manufacturing lot sizes to better support the current and planned clinical activities for the drug.
The company also said it plans to resume the phase 2 trial of pidilizumab in DLBLCL in the second half of this year.
The trial is expected to enroll approximately 180 patients who had an incomplete response to salvage therapy or autologous stem cell transplant for relapsed or refractory CD20+ DLBCL, transformed indolent lymphoma, or primary mediastinal B-cell lymphoma.
The patients will be assessed in 2 parallel cohorts of approximately 90 patients each. One cohort will enroll patients who have received an autologous stem cell transplant, and the other will enroll patients who have received salvage chemotherapy but are ineligible for transplant.
Pidilizumab will be given at a dose of 200 mg by intravenous infusion. The primary endpoint of the trial is best overall response rate. ![]()

The US Food and Drug Administration (FDA) has lifted the partial clinical hold on the investigational new drug (IND) application for pidilizumab (MDV9300) in hematologic malignancies.
This means the phase 2 trial of pidilizumab in patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL), as well as other studies that cross-reference the IND for the drug, may now proceed.
The partial clinical hold on pidilizumab was not related to any safety concerns.
The FDA placed the hold because the company developing pidilizumab, Medivation Inc., determined that the drug is not an inhibitor of PD-1, as researchers previously thought.
The phase 2 trial of pidilizumab in DLBCL was launched in late 2015 but had not enrolled any patients before the FDA placed the partial clinical hold.
Patients who were receiving pidilizumab through investigator-sponsored trials have continued to receive treatment despite the hold, and those investigators have been told to update their protocols and informed consent documents to reflect that pidilizumab is not an anti-PD-1 antibody.
Medivation has likewise revised the investigator brochure, protocols, and informed consent documents related to the phase 2 trial of DLBCL patients.
The company said it is still trying to determine pidilizumab’s mechanism of action.
“We are delighted that the FDA has lifted the partial clinical hold and that we may proceed with our potentially pivotal trial in this area of high unmet medical need,” said David Hung, MD, founder, president, and chief executive officer of Medivation.
“As we move forward, we also are working to determine the compound’s exact binding mechanism which, we believe, modulates the body’s innate immune response and differentiates it from the heavily crowded immuno-oncology space that targets the adaptive side of immunity.”
Medivation said it intends to submit an amendment to the Chemistry, Manufacturing, and Controls section of the IND for pidilizumab to provide for larger manufacturing lot sizes to better support the current and planned clinical activities for the drug.
The company also said it plans to resume the phase 2 trial of pidilizumab in DLBLCL in the second half of this year.
The trial is expected to enroll approximately 180 patients who had an incomplete response to salvage therapy or autologous stem cell transplant for relapsed or refractory CD20+ DLBCL, transformed indolent lymphoma, or primary mediastinal B-cell lymphoma.
The patients will be assessed in 2 parallel cohorts of approximately 90 patients each. One cohort will enroll patients who have received an autologous stem cell transplant, and the other will enroll patients who have received salvage chemotherapy but are ineligible for transplant.
Pidilizumab will be given at a dose of 200 mg by intravenous infusion. The primary endpoint of the trial is best overall response rate. ![]()
Targeting a protein to prevent malignancy

Photo by Aaron Logan
New research suggests hematologic malignancies driven by MYC might be prevented by lowering levels of another protein, MCL-1.
“Our colleagues had previously discovered that reducing the activity of MCL-1 is a promising strategy to treat malignant MYC-driven cancers,” said Stephanie Grabow, PhD, of the Walter and Eliza Hall Institute of Medical Research in Parkville, Victoria, Australia.
“We have now shown that the same approach might be able to prevent those cancers from forming in the first place.”
Dr Grabow and her colleagues described this work in Cell Reports.
Previous research indicated that expression from both MCL-1 alleles is essential for the survival of hematopoietic stem and progenitor cells during stress-induced repopulation of the hematopoietic system.
So, with this study, Dr Grabow and her colleagues set out to determine whether reducing MCL-1 protein levels might hinder the development of hematologic malignancies.
In experiments with mice, the investigators found that loss of one MCL-1 allele significantly delayed the development of MYC-driven lymphoma and reduced MYC-driven accumulation of pre-leukemic cancer-initiating cells.
However, loss of one p53 allele accelerated MYC-driven lymphomagenesis even when one MCL-1 allele was deleted. Loss of PUMA accelerated lymphoma development as well, though to a much lesser extent.
Loss of BIM substantially accelerated lymphomagenesis when one MCL-1 allele was deleted, restoring lymphoma-initiating cells and the rate of tumor development.
And loss of one BIM allele overrode the survival defect observed in pre-leukemic Eμ-Myc B-cell progenitors when one MCL-1 allele was deleted.
The investigators noted that loss of one MCL-1 allele did not noticeably impair the survival of normal B lymphoid cells even though it greatly diminished the survival of MYC-overexpressing B-cell progenitors.
“No one had realized just how vulnerable cells undergoing cancerous changes are to a relatively minor reduction in the levels of MCL-1,” Dr Grabow said.
“We found that MCL-1 is critical for keeping developing cancer cells alive through the stressful events that cause the transformation of a healthy cell into a cancerous cell. This result is particularly exciting because MCL-1 inhibitors are already in development as anticancer drugs.”
Study investigator Brandon Aubrey, MBBS, also of the Walter and Eliza Hall Institute, said this research could inform future strategies to prevent cancer.
“Early treatment or even cancer prevention are likely to be a more effective way to fight cancer than treating an established cancer after it has already formed and made a person sick,” he said. ”Our research has suggested that dependency on MCL-1 could be a key vulnerability of many developing cancers.”
“In the future, MCL-1 inhibitors might have potential benefit for treating the very early stages of MYC-driven cancers, or we may even be able use these agents to prevent people from getting cancer in the first place.” ![]()

Photo by Aaron Logan
New research suggests hematologic malignancies driven by MYC might be prevented by lowering levels of another protein, MCL-1.
“Our colleagues had previously discovered that reducing the activity of MCL-1 is a promising strategy to treat malignant MYC-driven cancers,” said Stephanie Grabow, PhD, of the Walter and Eliza Hall Institute of Medical Research in Parkville, Victoria, Australia.
“We have now shown that the same approach might be able to prevent those cancers from forming in the first place.”
Dr Grabow and her colleagues described this work in Cell Reports.
Previous research indicated that expression from both MCL-1 alleles is essential for the survival of hematopoietic stem and progenitor cells during stress-induced repopulation of the hematopoietic system.
So, with this study, Dr Grabow and her colleagues set out to determine whether reducing MCL-1 protein levels might hinder the development of hematologic malignancies.
In experiments with mice, the investigators found that loss of one MCL-1 allele significantly delayed the development of MYC-driven lymphoma and reduced MYC-driven accumulation of pre-leukemic cancer-initiating cells.
However, loss of one p53 allele accelerated MYC-driven lymphomagenesis even when one MCL-1 allele was deleted. Loss of PUMA accelerated lymphoma development as well, though to a much lesser extent.
Loss of BIM substantially accelerated lymphomagenesis when one MCL-1 allele was deleted, restoring lymphoma-initiating cells and the rate of tumor development.
And loss of one BIM allele overrode the survival defect observed in pre-leukemic Eμ-Myc B-cell progenitors when one MCL-1 allele was deleted.
The investigators noted that loss of one MCL-1 allele did not noticeably impair the survival of normal B lymphoid cells even though it greatly diminished the survival of MYC-overexpressing B-cell progenitors.
“No one had realized just how vulnerable cells undergoing cancerous changes are to a relatively minor reduction in the levels of MCL-1,” Dr Grabow said.
“We found that MCL-1 is critical for keeping developing cancer cells alive through the stressful events that cause the transformation of a healthy cell into a cancerous cell. This result is particularly exciting because MCL-1 inhibitors are already in development as anticancer drugs.”
Study investigator Brandon Aubrey, MBBS, also of the Walter and Eliza Hall Institute, said this research could inform future strategies to prevent cancer.
“Early treatment or even cancer prevention are likely to be a more effective way to fight cancer than treating an established cancer after it has already formed and made a person sick,” he said. ”Our research has suggested that dependency on MCL-1 could be a key vulnerability of many developing cancers.”
“In the future, MCL-1 inhibitors might have potential benefit for treating the very early stages of MYC-driven cancers, or we may even be able use these agents to prevent people from getting cancer in the first place.” ![]()

Photo by Aaron Logan
New research suggests hematologic malignancies driven by MYC might be prevented by lowering levels of another protein, MCL-1.
“Our colleagues had previously discovered that reducing the activity of MCL-1 is a promising strategy to treat malignant MYC-driven cancers,” said Stephanie Grabow, PhD, of the Walter and Eliza Hall Institute of Medical Research in Parkville, Victoria, Australia.
“We have now shown that the same approach might be able to prevent those cancers from forming in the first place.”
Dr Grabow and her colleagues described this work in Cell Reports.
Previous research indicated that expression from both MCL-1 alleles is essential for the survival of hematopoietic stem and progenitor cells during stress-induced repopulation of the hematopoietic system.
So, with this study, Dr Grabow and her colleagues set out to determine whether reducing MCL-1 protein levels might hinder the development of hematologic malignancies.
In experiments with mice, the investigators found that loss of one MCL-1 allele significantly delayed the development of MYC-driven lymphoma and reduced MYC-driven accumulation of pre-leukemic cancer-initiating cells.
However, loss of one p53 allele accelerated MYC-driven lymphomagenesis even when one MCL-1 allele was deleted. Loss of PUMA accelerated lymphoma development as well, though to a much lesser extent.
Loss of BIM substantially accelerated lymphomagenesis when one MCL-1 allele was deleted, restoring lymphoma-initiating cells and the rate of tumor development.
And loss of one BIM allele overrode the survival defect observed in pre-leukemic Eμ-Myc B-cell progenitors when one MCL-1 allele was deleted.
The investigators noted that loss of one MCL-1 allele did not noticeably impair the survival of normal B lymphoid cells even though it greatly diminished the survival of MYC-overexpressing B-cell progenitors.
“No one had realized just how vulnerable cells undergoing cancerous changes are to a relatively minor reduction in the levels of MCL-1,” Dr Grabow said.
“We found that MCL-1 is critical for keeping developing cancer cells alive through the stressful events that cause the transformation of a healthy cell into a cancerous cell. This result is particularly exciting because MCL-1 inhibitors are already in development as anticancer drugs.”
Study investigator Brandon Aubrey, MBBS, also of the Walter and Eliza Hall Institute, said this research could inform future strategies to prevent cancer.
“Early treatment or even cancer prevention are likely to be a more effective way to fight cancer than treating an established cancer after it has already formed and made a person sick,” he said. ”Our research has suggested that dependency on MCL-1 could be a key vulnerability of many developing cancers.”
“In the future, MCL-1 inhibitors might have potential benefit for treating the very early stages of MYC-driven cancers, or we may even be able use these agents to prevent people from getting cancer in the first place.” ![]()
FDA approves ibrutinib as first-line CLL therapy

Photo courtesy of Janssen
The US Food and Drug Administration (FDA) has approved the BTK inhibitor ibrutinib (Imbruvica) as a first-line treatment for patients with chronic lymphocytic leukemia (CLL).
This means ibrutinib is now FDA-approved to treat CLL patients regardless of their treatment history, including patients with 17p deletion.
Ibrutinib is also FDA-approved to treat Waldenström’s macroglobulinemia, and the drug was granted accelerated approval to treat patients with mantle cell lymphoma who have received at least 1 prior therapy.
Ibrutinib is jointly developed and commercialized by Pharmacyclics LLC, an AbbVie company, and Janssen Biotech, Inc. For more details on the drug, see the full prescribing information, available at imbruvica.com.
RESONATE-2 trial
The latest FDA approval for ibrutinib is based on results from the phase 3 RESONATE-2 trial (PCYC-1115), which were presented at the 2015 ASH Annual Meeting and simultaneously published in NEJM.
RESONATE-2 enrolled 269 treatment-naïve patients with CLL or small lymphocytic lymphoma who were 65 or older.
Patients were randomized to receive ibrutinib (n=136) at 420 mg once a day until progression or unacceptable toxicity, or chlorambucil (n=133) on days 1 and 15 of each 28-day cycle for up to 12 cycles. The starting dose for chlorambucil in cycle 1 was 0.5 mg/kg and was increased based on tolerability in cycle 2 by increments of 0.1 mg/kg to a maximum of 0.8 mg/kg.
The primary endpoint of the study was progression-free survival (PFS), as assessed by an independent review committee (IRC) according to the International Workshop on Chronic Lymphocytic Leukemia (iWCLL) 2008 criteria, with modification for treatment-related lymphocytosis.
Key secondary endpoints included overall response rate (based on the same iWCLL criteria), overall survival (OS), and safety.
Ibrutinib significantly prolonged PFS, as determined by the IRC, reducing the risk of progression or death by 84% compared to chlorambucil. The hazard ratio was 0.16 (P<0.001). The median PFS was not reached in the ibrutinib arm but was 18.9 months for the chlorambucil arm.
Ibrutinib significantly prolonged OS as well, although the median OS was not reached in either treatment arm. The OS rate at 24 months was 98% with ibrutinib and 85% with chlorambucil. The relative risk of death with ibrutinib was 84% lower than that with chlorambucil. The hazard ratio was 0.16 (P=0.001).
Ibrutinib was associated with a significantly higher IRC-assessed overall response rate compared to chlorambucil—82% and 35%, respectively (P<0.0001). Five patients (4%) in the ibrutinib arm achieved a complete response, as did 2 patients (2%) in the chlorambucil arm.
The median duration of treatment was 17.4 months in the ibrutinib arm and 7.1 months in the chlorambucil arm.
The most common adverse events of any grade—in the ibrutinib and chlorambucil arms, respectively—were diarrhea (42% and 17%), fatigue (30% and 38%), cough (22% and 15%), nausea (22% and 39%), peripheral edema (19% and 9%), dry eye (17% and 5%), arthralgia (16% and 7%), neutropenia (16% and 23%), and vomiting (13% and 20%).
Adverse events of grade 3 or higher—in the ibrutinib and chlorambucil arms, respectively—were neutropenia (10% and 18%), anemia (6% and 8%), hypertension (4% and 0%), pneumonia (4% and 2%), diarrhea (4% and 0%), maculopapular rash (3% and 2%), decreased platelet count (3% and 1%), abdominal pain (3% and 1%), hyponatremia (3% and 0%), thrombocytopenia (2% and 6%), febrile neutropenia (2% and 2%), upper respiratory tract infection (2% and 2%), pleural effusion (2% and 1%), cellulitis (2% and 0%), fatigue (1% and 5%), syncope (1% and 2%), and hemolytic anemia (0% and 2%). ![]()

Photo courtesy of Janssen
The US Food and Drug Administration (FDA) has approved the BTK inhibitor ibrutinib (Imbruvica) as a first-line treatment for patients with chronic lymphocytic leukemia (CLL).
This means ibrutinib is now FDA-approved to treat CLL patients regardless of their treatment history, including patients with 17p deletion.
Ibrutinib is also FDA-approved to treat Waldenström’s macroglobulinemia, and the drug was granted accelerated approval to treat patients with mantle cell lymphoma who have received at least 1 prior therapy.
Ibrutinib is jointly developed and commercialized by Pharmacyclics LLC, an AbbVie company, and Janssen Biotech, Inc. For more details on the drug, see the full prescribing information, available at imbruvica.com.
RESONATE-2 trial
The latest FDA approval for ibrutinib is based on results from the phase 3 RESONATE-2 trial (PCYC-1115), which were presented at the 2015 ASH Annual Meeting and simultaneously published in NEJM.
RESONATE-2 enrolled 269 treatment-naïve patients with CLL or small lymphocytic lymphoma who were 65 or older.
Patients were randomized to receive ibrutinib (n=136) at 420 mg once a day until progression or unacceptable toxicity, or chlorambucil (n=133) on days 1 and 15 of each 28-day cycle for up to 12 cycles. The starting dose for chlorambucil in cycle 1 was 0.5 mg/kg and was increased based on tolerability in cycle 2 by increments of 0.1 mg/kg to a maximum of 0.8 mg/kg.
The primary endpoint of the study was progression-free survival (PFS), as assessed by an independent review committee (IRC) according to the International Workshop on Chronic Lymphocytic Leukemia (iWCLL) 2008 criteria, with modification for treatment-related lymphocytosis.
Key secondary endpoints included overall response rate (based on the same iWCLL criteria), overall survival (OS), and safety.
Ibrutinib significantly prolonged PFS, as determined by the IRC, reducing the risk of progression or death by 84% compared to chlorambucil. The hazard ratio was 0.16 (P<0.001). The median PFS was not reached in the ibrutinib arm but was 18.9 months for the chlorambucil arm.
Ibrutinib significantly prolonged OS as well, although the median OS was not reached in either treatment arm. The OS rate at 24 months was 98% with ibrutinib and 85% with chlorambucil. The relative risk of death with ibrutinib was 84% lower than that with chlorambucil. The hazard ratio was 0.16 (P=0.001).
Ibrutinib was associated with a significantly higher IRC-assessed overall response rate compared to chlorambucil—82% and 35%, respectively (P<0.0001). Five patients (4%) in the ibrutinib arm achieved a complete response, as did 2 patients (2%) in the chlorambucil arm.
The median duration of treatment was 17.4 months in the ibrutinib arm and 7.1 months in the chlorambucil arm.
The most common adverse events of any grade—in the ibrutinib and chlorambucil arms, respectively—were diarrhea (42% and 17%), fatigue (30% and 38%), cough (22% and 15%), nausea (22% and 39%), peripheral edema (19% and 9%), dry eye (17% and 5%), arthralgia (16% and 7%), neutropenia (16% and 23%), and vomiting (13% and 20%).
Adverse events of grade 3 or higher—in the ibrutinib and chlorambucil arms, respectively—were neutropenia (10% and 18%), anemia (6% and 8%), hypertension (4% and 0%), pneumonia (4% and 2%), diarrhea (4% and 0%), maculopapular rash (3% and 2%), decreased platelet count (3% and 1%), abdominal pain (3% and 1%), hyponatremia (3% and 0%), thrombocytopenia (2% and 6%), febrile neutropenia (2% and 2%), upper respiratory tract infection (2% and 2%), pleural effusion (2% and 1%), cellulitis (2% and 0%), fatigue (1% and 5%), syncope (1% and 2%), and hemolytic anemia (0% and 2%). ![]()

Photo courtesy of Janssen
The US Food and Drug Administration (FDA) has approved the BTK inhibitor ibrutinib (Imbruvica) as a first-line treatment for patients with chronic lymphocytic leukemia (CLL).
This means ibrutinib is now FDA-approved to treat CLL patients regardless of their treatment history, including patients with 17p deletion.
Ibrutinib is also FDA-approved to treat Waldenström’s macroglobulinemia, and the drug was granted accelerated approval to treat patients with mantle cell lymphoma who have received at least 1 prior therapy.
Ibrutinib is jointly developed and commercialized by Pharmacyclics LLC, an AbbVie company, and Janssen Biotech, Inc. For more details on the drug, see the full prescribing information, available at imbruvica.com.
RESONATE-2 trial
The latest FDA approval for ibrutinib is based on results from the phase 3 RESONATE-2 trial (PCYC-1115), which were presented at the 2015 ASH Annual Meeting and simultaneously published in NEJM.
RESONATE-2 enrolled 269 treatment-naïve patients with CLL or small lymphocytic lymphoma who were 65 or older.
Patients were randomized to receive ibrutinib (n=136) at 420 mg once a day until progression or unacceptable toxicity, or chlorambucil (n=133) on days 1 and 15 of each 28-day cycle for up to 12 cycles. The starting dose for chlorambucil in cycle 1 was 0.5 mg/kg and was increased based on tolerability in cycle 2 by increments of 0.1 mg/kg to a maximum of 0.8 mg/kg.
The primary endpoint of the study was progression-free survival (PFS), as assessed by an independent review committee (IRC) according to the International Workshop on Chronic Lymphocytic Leukemia (iWCLL) 2008 criteria, with modification for treatment-related lymphocytosis.
Key secondary endpoints included overall response rate (based on the same iWCLL criteria), overall survival (OS), and safety.
Ibrutinib significantly prolonged PFS, as determined by the IRC, reducing the risk of progression or death by 84% compared to chlorambucil. The hazard ratio was 0.16 (P<0.001). The median PFS was not reached in the ibrutinib arm but was 18.9 months for the chlorambucil arm.
Ibrutinib significantly prolonged OS as well, although the median OS was not reached in either treatment arm. The OS rate at 24 months was 98% with ibrutinib and 85% with chlorambucil. The relative risk of death with ibrutinib was 84% lower than that with chlorambucil. The hazard ratio was 0.16 (P=0.001).
Ibrutinib was associated with a significantly higher IRC-assessed overall response rate compared to chlorambucil—82% and 35%, respectively (P<0.0001). Five patients (4%) in the ibrutinib arm achieved a complete response, as did 2 patients (2%) in the chlorambucil arm.
The median duration of treatment was 17.4 months in the ibrutinib arm and 7.1 months in the chlorambucil arm.
The most common adverse events of any grade—in the ibrutinib and chlorambucil arms, respectively—were diarrhea (42% and 17%), fatigue (30% and 38%), cough (22% and 15%), nausea (22% and 39%), peripheral edema (19% and 9%), dry eye (17% and 5%), arthralgia (16% and 7%), neutropenia (16% and 23%), and vomiting (13% and 20%).
Adverse events of grade 3 or higher—in the ibrutinib and chlorambucil arms, respectively—were neutropenia (10% and 18%), anemia (6% and 8%), hypertension (4% and 0%), pneumonia (4% and 2%), diarrhea (4% and 0%), maculopapular rash (3% and 2%), decreased platelet count (3% and 1%), abdominal pain (3% and 1%), hyponatremia (3% and 0%), thrombocytopenia (2% and 6%), febrile neutropenia (2% and 2%), upper respiratory tract infection (2% and 2%), pleural effusion (2% and 1%), cellulitis (2% and 0%), fatigue (1% and 5%), syncope (1% and 2%), and hemolytic anemia (0% and 2%). ![]()
Study results support screening of childhood HL survivors
A new study indicates that early screening can reduce the risk of death from breast cancer among female survivors of childhood Hodgkin lymphoma (HL) who received chest radiation.
Researchers found evidence to suggest that starting mammograms at age 25 can reduce the risk of breast cancer death among these patients, but using MRI can reduce the risk further.
Unfortunately, both methods come with a risk of false-positive results.
David Hodgson, MD, of the University of Toronto in Canada, and his colleagues reported these findings in the Journal of the National Cancer Institute.
Dr Hodgson estimates there are thousands of HL survivors in North America treated throughout the 1990s and later who received chest radiation and are unaware they are at risk of breast cancer and eligible for early screening.
“Many of these are women who received radiotherapy to more normal tissue or at higher doses than are used currently,” he said. “But even for more recently treated patients, screening should reduce the risk of breast cancer death.”
For this study, Dr Hodgson and his colleagues gathered published information from dozens of studies about the risk of developing breast cancer in childhood HL survivors, the accuracy of different forms of breast cancer screening, and the rates at which women agree to be screened when asked.
Using mathematical models, the researchers used these data to quantify the effectiveness of starting screening early—at age 25.
The team found that, using mammography, about 260 survivors of childhood HL would need to be invited to have early breast cancer screening to prevent 1 breast cancer death.
However, the use of MRI for screening improved the effectiveness considerably. It reduced the number of women needing screening to prevent 1 breast cancer death to less than 80.
For HL survivors treated at age 15, the absolute risk of breast cancer mortality by age 75 was predicted to decrease from 16.65% with no early screening to 16.28% with annual mammography, 15.40% with annual MRI, 15.38% with same-day annual mammography and MRI, and 15.37% with alternating mammography and MRI every 6 months.
Dr Hodgson cautioned that there is a risk of false-positive screening results, particuarly with MRI, given that the method can detect many changes in breast tissue, most of which are not cancer.
The data suggested that, from age 25 to 75, at least one false-positive result would occur in 48% of women screened with mammography, 74% screened with MRI alone, and 79% screened with both methods.
The number of false-positives per 1000 screens would be 29.98 with mammography, 71.71 with MRI, and 99.52 with either same-day mammography and MRI or alternating mammography and MRI.
“So this is important for patients to know and for physicians to counsel patients about because it’s stressful for a patient to be called back about suspicious findings,” Dr Hodgson said. ![]()

A new study indicates that early screening can reduce the risk of death from breast cancer among female survivors of childhood Hodgkin lymphoma (HL) who received chest radiation.
Researchers found evidence to suggest that starting mammograms at age 25 can reduce the risk of breast cancer death among these patients, but using MRI can reduce the risk further.
Unfortunately, both methods come with a risk of false-positive results.
David Hodgson, MD, of the University of Toronto in Canada, and his colleagues reported these findings in the Journal of the National Cancer Institute.
Dr Hodgson estimates there are thousands of HL survivors in North America treated throughout the 1990s and later who received chest radiation and are unaware they are at risk of breast cancer and eligible for early screening.
“Many of these are women who received radiotherapy to more normal tissue or at higher doses than are used currently,” he said. “But even for more recently treated patients, screening should reduce the risk of breast cancer death.”
For this study, Dr Hodgson and his colleagues gathered published information from dozens of studies about the risk of developing breast cancer in childhood HL survivors, the accuracy of different forms of breast cancer screening, and the rates at which women agree to be screened when asked.
Using mathematical models, the researchers used these data to quantify the effectiveness of starting screening early—at age 25.
The team found that, using mammography, about 260 survivors of childhood HL would need to be invited to have early breast cancer screening to prevent 1 breast cancer death.
However, the use of MRI for screening improved the effectiveness considerably. It reduced the number of women needing screening to prevent 1 breast cancer death to less than 80.
For HL survivors treated at age 15, the absolute risk of breast cancer mortality by age 75 was predicted to decrease from 16.65% with no early screening to 16.28% with annual mammography, 15.40% with annual MRI, 15.38% with same-day annual mammography and MRI, and 15.37% with alternating mammography and MRI every 6 months.
Dr Hodgson cautioned that there is a risk of false-positive screening results, particuarly with MRI, given that the method can detect many changes in breast tissue, most of which are not cancer.
The data suggested that, from age 25 to 75, at least one false-positive result would occur in 48% of women screened with mammography, 74% screened with MRI alone, and 79% screened with both methods.
The number of false-positives per 1000 screens would be 29.98 with mammography, 71.71 with MRI, and 99.52 with either same-day mammography and MRI or alternating mammography and MRI.
“So this is important for patients to know and for physicians to counsel patients about because it’s stressful for a patient to be called back about suspicious findings,” Dr Hodgson said. ![]()

A new study indicates that early screening can reduce the risk of death from breast cancer among female survivors of childhood Hodgkin lymphoma (HL) who received chest radiation.
Researchers found evidence to suggest that starting mammograms at age 25 can reduce the risk of breast cancer death among these patients, but using MRI can reduce the risk further.
Unfortunately, both methods come with a risk of false-positive results.
David Hodgson, MD, of the University of Toronto in Canada, and his colleagues reported these findings in the Journal of the National Cancer Institute.
Dr Hodgson estimates there are thousands of HL survivors in North America treated throughout the 1990s and later who received chest radiation and are unaware they are at risk of breast cancer and eligible for early screening.
“Many of these are women who received radiotherapy to more normal tissue or at higher doses than are used currently,” he said. “But even for more recently treated patients, screening should reduce the risk of breast cancer death.”
For this study, Dr Hodgson and his colleagues gathered published information from dozens of studies about the risk of developing breast cancer in childhood HL survivors, the accuracy of different forms of breast cancer screening, and the rates at which women agree to be screened when asked.
Using mathematical models, the researchers used these data to quantify the effectiveness of starting screening early—at age 25.
The team found that, using mammography, about 260 survivors of childhood HL would need to be invited to have early breast cancer screening to prevent 1 breast cancer death.
However, the use of MRI for screening improved the effectiveness considerably. It reduced the number of women needing screening to prevent 1 breast cancer death to less than 80.
For HL survivors treated at age 15, the absolute risk of breast cancer mortality by age 75 was predicted to decrease from 16.65% with no early screening to 16.28% with annual mammography, 15.40% with annual MRI, 15.38% with same-day annual mammography and MRI, and 15.37% with alternating mammography and MRI every 6 months.
Dr Hodgson cautioned that there is a risk of false-positive screening results, particuarly with MRI, given that the method can detect many changes in breast tissue, most of which are not cancer.
The data suggested that, from age 25 to 75, at least one false-positive result would occur in 48% of women screened with mammography, 74% screened with MRI alone, and 79% screened with both methods.
The number of false-positives per 1000 screens would be 29.98 with mammography, 71.71 with MRI, and 99.52 with either same-day mammography and MRI or alternating mammography and MRI.
“So this is important for patients to know and for physicians to counsel patients about because it’s stressful for a patient to be called back about suspicious findings,” Dr Hodgson said.