User login
Drug shows promise for treating resistant AML, MCL

Preclinical research suggests the investigational anticancer drug ONC201 can be effective against mantle cell lymphoma (MCL) and acute myeloid leukemia (AML).
ONC201 induced p53-independent apoptosis in AML and MCL cell lines and in samples from patients with either disease.
Investigators noted that p53 dysfunction occurs in more than half of malignancies and can promote resistance to standard chemotherapy.
“The clinical challenge posed by p53 abnormalities in blood malignancies is that therapeutic strategies other than standard chemotherapies are required,” said Michael Andreeff, MD, PhD, of The University of Texas MD Anderson Cancer Center in Houston.
“We found that ONC201 caused p53-independent cell death and cell cycle arrest in cell lines and in lymphoma and acute leukemia patient samples.”
Dr Andreeff and his colleagues reported these findings in Science Signaling. Some of the investigators involved in this research are affiliated with Oncoceutics Inc., the company developing ONC201.
Dr Andreeff and his colleagues assessed the effects of ONC201 against AML and MCL, in both cultured cell lines and primary cells bearing either wild-type or mutant p53.
The patient samples included those that demonstrated genetic abnormalities linked to poor prognosis (FLT3 mutations, TP53 mutations) or resistance to ibrutinib. The team also tested ONC201 in a bortezomib-resistant myeloma cell line.
The experiments showed that ONC201 exerted anticancer activity regardless of p53 status, FLT3 mutations, or drug resistance. ONC201 proved active in the bortezomib-resistant myeloma cell line and in ibrutinib-resistant samples from MCL patients.
Experiments in mice showed that ONC201 caused cell death in AML and leukemia stem cells while sparing normal bone marrow cells.
And the investigators found that combining ONC201 with the BCL-2 antagonist venetoclax (ABT-199) synergistically increased apoptosis.
Further investigation revealed that ONC201 increased translation of the stress-induced protein ATF4 through stress signals similar to those caused by unfolded protein response (UPR) and integrated stress response (ISR).
“This increase in ATF4 in ONC201-treated hematopoietic cells promoted cell death,” Dr Andreeff explained. “However, unlike with UPR and ISR, the increase in ATF4 in ONC201-treated cells was not regulated by standard molecular signaling, indicating a novel mechanism of stressing cancer cells to death regardless of p53 status.”
The investigators noted that the mechanisms of ONC201 identified in solid tumors—namely, induction of TRAIL and DR5—were not operational in leukemia and lymphoma.
A study of ONC201 in solid tumors and multiple myeloma was published alongside this study in Science Signaling.
“There is clear evidence that ONC201 has clinical potential in hematological malignancies,” Dr Andreeff noted. “Clinical trials in leukemia and lymphoma patients have recently been initiated at MD Anderson.” ![]()

Preclinical research suggests the investigational anticancer drug ONC201 can be effective against mantle cell lymphoma (MCL) and acute myeloid leukemia (AML).
ONC201 induced p53-independent apoptosis in AML and MCL cell lines and in samples from patients with either disease.
Investigators noted that p53 dysfunction occurs in more than half of malignancies and can promote resistance to standard chemotherapy.
“The clinical challenge posed by p53 abnormalities in blood malignancies is that therapeutic strategies other than standard chemotherapies are required,” said Michael Andreeff, MD, PhD, of The University of Texas MD Anderson Cancer Center in Houston.
“We found that ONC201 caused p53-independent cell death and cell cycle arrest in cell lines and in lymphoma and acute leukemia patient samples.”
Dr Andreeff and his colleagues reported these findings in Science Signaling. Some of the investigators involved in this research are affiliated with Oncoceutics Inc., the company developing ONC201.
Dr Andreeff and his colleagues assessed the effects of ONC201 against AML and MCL, in both cultured cell lines and primary cells bearing either wild-type or mutant p53.
The patient samples included those that demonstrated genetic abnormalities linked to poor prognosis (FLT3 mutations, TP53 mutations) or resistance to ibrutinib. The team also tested ONC201 in a bortezomib-resistant myeloma cell line.
The experiments showed that ONC201 exerted anticancer activity regardless of p53 status, FLT3 mutations, or drug resistance. ONC201 proved active in the bortezomib-resistant myeloma cell line and in ibrutinib-resistant samples from MCL patients.
Experiments in mice showed that ONC201 caused cell death in AML and leukemia stem cells while sparing normal bone marrow cells.
And the investigators found that combining ONC201 with the BCL-2 antagonist venetoclax (ABT-199) synergistically increased apoptosis.
Further investigation revealed that ONC201 increased translation of the stress-induced protein ATF4 through stress signals similar to those caused by unfolded protein response (UPR) and integrated stress response (ISR).
“This increase in ATF4 in ONC201-treated hematopoietic cells promoted cell death,” Dr Andreeff explained. “However, unlike with UPR and ISR, the increase in ATF4 in ONC201-treated cells was not regulated by standard molecular signaling, indicating a novel mechanism of stressing cancer cells to death regardless of p53 status.”
The investigators noted that the mechanisms of ONC201 identified in solid tumors—namely, induction of TRAIL and DR5—were not operational in leukemia and lymphoma.
A study of ONC201 in solid tumors and multiple myeloma was published alongside this study in Science Signaling.
“There is clear evidence that ONC201 has clinical potential in hematological malignancies,” Dr Andreeff noted. “Clinical trials in leukemia and lymphoma patients have recently been initiated at MD Anderson.” ![]()

Preclinical research suggests the investigational anticancer drug ONC201 can be effective against mantle cell lymphoma (MCL) and acute myeloid leukemia (AML).
ONC201 induced p53-independent apoptosis in AML and MCL cell lines and in samples from patients with either disease.
Investigators noted that p53 dysfunction occurs in more than half of malignancies and can promote resistance to standard chemotherapy.
“The clinical challenge posed by p53 abnormalities in blood malignancies is that therapeutic strategies other than standard chemotherapies are required,” said Michael Andreeff, MD, PhD, of The University of Texas MD Anderson Cancer Center in Houston.
“We found that ONC201 caused p53-independent cell death and cell cycle arrest in cell lines and in lymphoma and acute leukemia patient samples.”
Dr Andreeff and his colleagues reported these findings in Science Signaling. Some of the investigators involved in this research are affiliated with Oncoceutics Inc., the company developing ONC201.
Dr Andreeff and his colleagues assessed the effects of ONC201 against AML and MCL, in both cultured cell lines and primary cells bearing either wild-type or mutant p53.
The patient samples included those that demonstrated genetic abnormalities linked to poor prognosis (FLT3 mutations, TP53 mutations) or resistance to ibrutinib. The team also tested ONC201 in a bortezomib-resistant myeloma cell line.
The experiments showed that ONC201 exerted anticancer activity regardless of p53 status, FLT3 mutations, or drug resistance. ONC201 proved active in the bortezomib-resistant myeloma cell line and in ibrutinib-resistant samples from MCL patients.
Experiments in mice showed that ONC201 caused cell death in AML and leukemia stem cells while sparing normal bone marrow cells.
And the investigators found that combining ONC201 with the BCL-2 antagonist venetoclax (ABT-199) synergistically increased apoptosis.
Further investigation revealed that ONC201 increased translation of the stress-induced protein ATF4 through stress signals similar to those caused by unfolded protein response (UPR) and integrated stress response (ISR).
“This increase in ATF4 in ONC201-treated hematopoietic cells promoted cell death,” Dr Andreeff explained. “However, unlike with UPR and ISR, the increase in ATF4 in ONC201-treated cells was not regulated by standard molecular signaling, indicating a novel mechanism of stressing cancer cells to death regardless of p53 status.”
The investigators noted that the mechanisms of ONC201 identified in solid tumors—namely, induction of TRAIL and DR5—were not operational in leukemia and lymphoma.
A study of ONC201 in solid tumors and multiple myeloma was published alongside this study in Science Signaling.
“There is clear evidence that ONC201 has clinical potential in hematological malignancies,” Dr Andreeff noted. “Clinical trials in leukemia and lymphoma patients have recently been initiated at MD Anderson.” ![]()
Nanoparticles deliver Aurora kinase inhibitor with increased safety and efficacy
Using nanoparticles to encapsulate an Aurora B kinase inhibitor improved the efficacy and tolerability of the drug and allowed less frequent dosing in preclinical models, according to researchers.
“The AZD2811 nanoparticles identified in this study have the potential to increase efficacy at tolerable doses using a more convenient dosing regimen, which may in turn extend the utility of Aurora B kinase inhibition to a broader range of hematological and solid tumor cancer indications,” wrote Susan Ashton of AstraZeneca, and her colleagues (Sci Transl Med. 2016 Feb 10. doi: 10.1126/scitranslmed.aad2355).
“The improved bone marrow profile observed with slow-releasing nanoparticles may enable efficacious combination treatments” with chemotherapy, radiotherapy, or poly(adenosine diphosphate–ribose) polymerase (PARP) inhibitors.
The study was undertaken because a free-drug version of the agent, known as AZD1152, had led to a significant improvement in the complete response rate of acute myeloid leukemia compared to standard of care in a phase II trial. Efficacy, however, was associated with major toxicities, including myelosuppression. Further, AZD1152 had to be administered as a 7-day continuous intravenous infusion.
By using the Accurin nanoparticle platform to vary drug release kinetics, the researchers devised a formulation to maximize the therapeutic effect of the kinase inhibitor while sparing healthy tissue. AZD1152 is a water-soluble prodrug of AZD2811, which the researchers used to develop their the nanoparticle formulation.
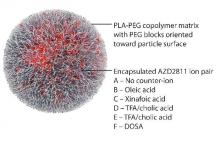
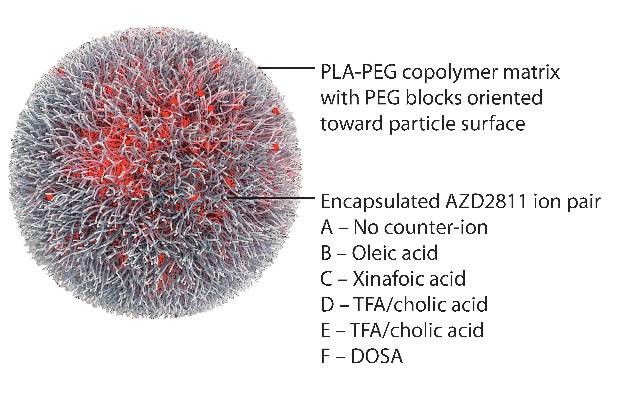
AZD2811 was encapsulated in polymeric nanoparticles termed Accurins, which are composed of block copolymers of poly-D,L-lactide (PLA) and poly(ethylene glycol) (PEG). Accurins accumulate in tumors, increasing the drug’s concentration and duration of exposure to the cancer cells. Organic acid counterions were used to increase encapsulation efficiency and decrease the release rate of AZD2811.
“We identified a formulation profile that could deliver active drug for more than 1 week, resulting in prolonged target inhibition in tumor tissue together with improved preclinical efficacy and therapeutic index over the AZD1152 prodrug in several animal models,” they wrote.
In nude rats bearing human colorectal adenocarcinoma SW620 xenografts, the nanoparticles inhibited kinase over a 96-hour time course, while the free drug resulted in complete enzyme recovery at 24 hours. Nanoparticles inhibited tumor growth by over 90%, compared with 58% for the free drug at twice the dose, and showed little toxicity as evidenced by stable body weight. Nanoparticles were retained in the tumor xenografts for up to 6 days, while the free drug was undetected in tumors 24 hours after administration.
“Although we selected a lead formulation using a tumor model (SW620) that supported the AZD1152 program – and, as such, we had extensive comparator data from which to benchmark the tolerability, PD, and efficacy of candidate nanoparticles – the model is subject to the known limitations of xenografted human tumor cell lines in assessing therapeutic candidates in oncology. Moreover, although rat bone marrow is commonly used to model myelotoxicity in humans, interrogation of the nanoparticle dose and schedule in patients may be required to achieve optimal clinical results,” they concluded.
AstraZeneca funded the study. Dr. Ashton and several coauthors are current or former employees and shareholders of AstraZeneca or BIND. The companies are developing the drug and technologies.
By encapsulating an Aurora B kinase inhibitor in Accurin particles, the researchers appear to have succeeded in enhancing the drug’s therapeutic activity and safety in mouse xenograft models. It remains to be seen how widely applicable this technology will be, and whether these results will be replicated in patients as the AZD1151-hQPA Accurin formulation heads toward first-in-human clinical trials. If the Accurin technology can solve the pharmacokinetic and toxicity issues for Aurora kinase inhibitors, it will likely also be applicable to other compounds that have encountered similar difficulties in clinical development and will add a formulation approach to address very meaningful and challenging issues that face many new molecules during clinical development.
David J. Bearss, Ph.D., is the chief executive officer of Tolero Pharmaceutical, Lehi, Utah. His remarks were part of an editorial accompanying the report in Science Translational Medicine (2016 Feb 10. doi: 10.1126/scitranslmed.aaf1417).
By encapsulating an Aurora B kinase inhibitor in Accurin particles, the researchers appear to have succeeded in enhancing the drug’s therapeutic activity and safety in mouse xenograft models. It remains to be seen how widely applicable this technology will be, and whether these results will be replicated in patients as the AZD1151-hQPA Accurin formulation heads toward first-in-human clinical trials. If the Accurin technology can solve the pharmacokinetic and toxicity issues for Aurora kinase inhibitors, it will likely also be applicable to other compounds that have encountered similar difficulties in clinical development and will add a formulation approach to address very meaningful and challenging issues that face many new molecules during clinical development.
David J. Bearss, Ph.D., is the chief executive officer of Tolero Pharmaceutical, Lehi, Utah. His remarks were part of an editorial accompanying the report in Science Translational Medicine (2016 Feb 10. doi: 10.1126/scitranslmed.aaf1417).
By encapsulating an Aurora B kinase inhibitor in Accurin particles, the researchers appear to have succeeded in enhancing the drug’s therapeutic activity and safety in mouse xenograft models. It remains to be seen how widely applicable this technology will be, and whether these results will be replicated in patients as the AZD1151-hQPA Accurin formulation heads toward first-in-human clinical trials. If the Accurin technology can solve the pharmacokinetic and toxicity issues for Aurora kinase inhibitors, it will likely also be applicable to other compounds that have encountered similar difficulties in clinical development and will add a formulation approach to address very meaningful and challenging issues that face many new molecules during clinical development.
David J. Bearss, Ph.D., is the chief executive officer of Tolero Pharmaceutical, Lehi, Utah. His remarks were part of an editorial accompanying the report in Science Translational Medicine (2016 Feb 10. doi: 10.1126/scitranslmed.aaf1417).
Using nanoparticles to encapsulate an Aurora B kinase inhibitor improved the efficacy and tolerability of the drug and allowed less frequent dosing in preclinical models, according to researchers.
“The AZD2811 nanoparticles identified in this study have the potential to increase efficacy at tolerable doses using a more convenient dosing regimen, which may in turn extend the utility of Aurora B kinase inhibition to a broader range of hematological and solid tumor cancer indications,” wrote Susan Ashton of AstraZeneca, and her colleagues (Sci Transl Med. 2016 Feb 10. doi: 10.1126/scitranslmed.aad2355).
“The improved bone marrow profile observed with slow-releasing nanoparticles may enable efficacious combination treatments” with chemotherapy, radiotherapy, or poly(adenosine diphosphate–ribose) polymerase (PARP) inhibitors.
The study was undertaken because a free-drug version of the agent, known as AZD1152, had led to a significant improvement in the complete response rate of acute myeloid leukemia compared to standard of care in a phase II trial. Efficacy, however, was associated with major toxicities, including myelosuppression. Further, AZD1152 had to be administered as a 7-day continuous intravenous infusion.
By using the Accurin nanoparticle platform to vary drug release kinetics, the researchers devised a formulation to maximize the therapeutic effect of the kinase inhibitor while sparing healthy tissue. AZD1152 is a water-soluble prodrug of AZD2811, which the researchers used to develop their the nanoparticle formulation.
AZD2811 was encapsulated in polymeric nanoparticles termed Accurins, which are composed of block copolymers of poly-D,L-lactide (PLA) and poly(ethylene glycol) (PEG). Accurins accumulate in tumors, increasing the drug’s concentration and duration of exposure to the cancer cells. Organic acid counterions were used to increase encapsulation efficiency and decrease the release rate of AZD2811.
“We identified a formulation profile that could deliver active drug for more than 1 week, resulting in prolonged target inhibition in tumor tissue together with improved preclinical efficacy and therapeutic index over the AZD1152 prodrug in several animal models,” they wrote.
In nude rats bearing human colorectal adenocarcinoma SW620 xenografts, the nanoparticles inhibited kinase over a 96-hour time course, while the free drug resulted in complete enzyme recovery at 24 hours. Nanoparticles inhibited tumor growth by over 90%, compared with 58% for the free drug at twice the dose, and showed little toxicity as evidenced by stable body weight. Nanoparticles were retained in the tumor xenografts for up to 6 days, while the free drug was undetected in tumors 24 hours after administration.
“Although we selected a lead formulation using a tumor model (SW620) that supported the AZD1152 program – and, as such, we had extensive comparator data from which to benchmark the tolerability, PD, and efficacy of candidate nanoparticles – the model is subject to the known limitations of xenografted human tumor cell lines in assessing therapeutic candidates in oncology. Moreover, although rat bone marrow is commonly used to model myelotoxicity in humans, interrogation of the nanoparticle dose and schedule in patients may be required to achieve optimal clinical results,” they concluded.
AstraZeneca funded the study. Dr. Ashton and several coauthors are current or former employees and shareholders of AstraZeneca or BIND. The companies are developing the drug and technologies.
Using nanoparticles to encapsulate an Aurora B kinase inhibitor improved the efficacy and tolerability of the drug and allowed less frequent dosing in preclinical models, according to researchers.
“The AZD2811 nanoparticles identified in this study have the potential to increase efficacy at tolerable doses using a more convenient dosing regimen, which may in turn extend the utility of Aurora B kinase inhibition to a broader range of hematological and solid tumor cancer indications,” wrote Susan Ashton of AstraZeneca, and her colleagues (Sci Transl Med. 2016 Feb 10. doi: 10.1126/scitranslmed.aad2355).
“The improved bone marrow profile observed with slow-releasing nanoparticles may enable efficacious combination treatments” with chemotherapy, radiotherapy, or poly(adenosine diphosphate–ribose) polymerase (PARP) inhibitors.
The study was undertaken because a free-drug version of the agent, known as AZD1152, had led to a significant improvement in the complete response rate of acute myeloid leukemia compared to standard of care in a phase II trial. Efficacy, however, was associated with major toxicities, including myelosuppression. Further, AZD1152 had to be administered as a 7-day continuous intravenous infusion.
By using the Accurin nanoparticle platform to vary drug release kinetics, the researchers devised a formulation to maximize the therapeutic effect of the kinase inhibitor while sparing healthy tissue. AZD1152 is a water-soluble prodrug of AZD2811, which the researchers used to develop their the nanoparticle formulation.
AZD2811 was encapsulated in polymeric nanoparticles termed Accurins, which are composed of block copolymers of poly-D,L-lactide (PLA) and poly(ethylene glycol) (PEG). Accurins accumulate in tumors, increasing the drug’s concentration and duration of exposure to the cancer cells. Organic acid counterions were used to increase encapsulation efficiency and decrease the release rate of AZD2811.
“We identified a formulation profile that could deliver active drug for more than 1 week, resulting in prolonged target inhibition in tumor tissue together with improved preclinical efficacy and therapeutic index over the AZD1152 prodrug in several animal models,” they wrote.
In nude rats bearing human colorectal adenocarcinoma SW620 xenografts, the nanoparticles inhibited kinase over a 96-hour time course, while the free drug resulted in complete enzyme recovery at 24 hours. Nanoparticles inhibited tumor growth by over 90%, compared with 58% for the free drug at twice the dose, and showed little toxicity as evidenced by stable body weight. Nanoparticles were retained in the tumor xenografts for up to 6 days, while the free drug was undetected in tumors 24 hours after administration.
“Although we selected a lead formulation using a tumor model (SW620) that supported the AZD1152 program – and, as such, we had extensive comparator data from which to benchmark the tolerability, PD, and efficacy of candidate nanoparticles – the model is subject to the known limitations of xenografted human tumor cell lines in assessing therapeutic candidates in oncology. Moreover, although rat bone marrow is commonly used to model myelotoxicity in humans, interrogation of the nanoparticle dose and schedule in patients may be required to achieve optimal clinical results,” they concluded.
AstraZeneca funded the study. Dr. Ashton and several coauthors are current or former employees and shareholders of AstraZeneca or BIND. The companies are developing the drug and technologies.
Key clinical point: Aurora B kinase inhibitor nanoparticles displayed accumulation and retention in tumors with improved efficacy and minimal bone marrow pathology in animal models.
Major finding: Nanoparticles inhibited tumor growth by over 90%, compared with 58% for the free drug at twice the dose, and showed little toxicity; the free drug was undetected in tumors 24 hours after administration, and nanoparticle-delivered drug was detectable up to 6 days.
Data sources: Nude rats and nude mice bearing human colorectal adenocarcinoma SW620 xenografts.
Disclosures: AstraZeneca funded the study. Dr. Ashton and several coauthors are current or former employees and shareholders of AstraZeneca or BIND. The companies are developing the drug and technologies.
Combo can produce durable remissions in PTCL

Photo by Larry Young
SAN FRANCISCO—A combination treatment regimen can produce durable remissions in patients newly diagnosed with peripheral T-cell lymphoma (PTCL), results of a phase 1 study suggest.
The patients received brentuximab vedotin plus cyclophosphamide, doxorubicin, and prednisolone (BV+CHP). In some cases, this was followed by BV monotherapy.
The estimated 3-year progression-free survival (PFS) for these patients was 52%, and the overall survival (OS) was 80%.
There was a high rate of peripheral neuropathy (73%), but most cases resolved or improved over time.
Steven Horwitz, MD, of Memorial Sloan Kettering Cancer Center in New York, New York, and his colleagues presented these data as a poster at the 8th Annual T-cell Lymphoma Forum. The study was supported by Seattle Genetics and Millennium Pharmaceuticals.
The researchers presented data on 26 patients newly diagnosed with PTCL. Nineteen patients had systemic anaplastic large-cell lymphoma (ALCL; 16 ALK- and 3 ALK+), 2 had PTCL not otherwise specified, 2 had angioimmunoblastic T-cell lymphoma, 2 had adult T-cell leukemia/lymphoma, and 1 had enteropathy-associated T-cell lymphoma.
The patients’ median age was 56 (range, 21-82). Sixty-nine percent of patients had an IPI score of 2 or higher, and 73% had stage III/IV disease.
Treatment
The patients received BV+CHP every 3 weeks for 6 cycles. Those who achieved at least a partial remission could go on to receive up to 10 additional cycles of single-agent BV at 1.8 mg/kg every 3 weeks.
Twenty-three patients (88%) completed all 6 cycles of BV+CHP, and 21 patients (81%) went on to receive BV monotherapy, 11 of whom (42%) received all 10 cycles.
Fifteen patients (58%) discontinued treatment, 3 due to progressive disease, 3 due to investigator decision, 6 due to adverse events, and 3 due to patient decision.
After a median observation period of 38.7 months (range, 4.6 to 44.3), 77% of patients (n=20) remained on study.
Toxicity
The most common adverse events (occurring in at least 30% of patients) were nausea (69%), peripheral sensory neuropathy (69%), diarrhea (62%), fatigue (58%), alopecia (54%), dyspnea (46%), constipation (35%), myalgia (31%), peripheral edema (31%), chills (31%), anemia (31%), insomnia (31%), and febrile neutropenia.
The most common grade 3 or higher adverse events (occurring in at least 10% of patients) were febrile neutropenia (31%), neutropenia (23%), anemia (15%), and pulmonary embolism (12%).
There were 6 adverse events resulting in treatment discontinuation—peripheral sensory neuropathy (n=3), abdominal pain (n=1), asthenia (n=1), and peripheral motor neuropathy (n=1).
Seventy-three percent of patients (19/26) experienced peripheral neuropathy. Thirty-seven percent (n=7) had a complete resolution of neuropathy, and 58% (n=11) had some improvement. The median time to resolution was 1.3 months. Twelve patients (63%) had ongoing neuropathy at last follow-up, but most had grade 1 (n=10).
Response and survival
The objective response rate was 100%, and the complete response rate was 88% (n=23). One patient who had a partial response at the end of combination therapy achieved a complete response after going on to single-agent BV.
Twenty-one of the 26 patients are still alive—15 with ALCL and 6 with other PTCLs. Sixteen patients have not progressed—9 with ALCL and 5 with other PTCLs.
After progression, 5 patients received BV again, and 3 received stem cell transplants (2 allogeneic and 1 autologous).
The estimated 3-year PFS was 52%, and the estimated OS was 80%. The researchers noted that these rates compare favorably with the historical 3-year PFS and OS rates of 30% and 40%, respectively.
Researchers are currently conducting a phase 3 trial comparing BV+CHP with CHOP as frontline treatment of CD30+ mature T-cell lymphomas (ECHELON-2, NCT01777152). ![]()

Photo by Larry Young
SAN FRANCISCO—A combination treatment regimen can produce durable remissions in patients newly diagnosed with peripheral T-cell lymphoma (PTCL), results of a phase 1 study suggest.
The patients received brentuximab vedotin plus cyclophosphamide, doxorubicin, and prednisolone (BV+CHP). In some cases, this was followed by BV monotherapy.
The estimated 3-year progression-free survival (PFS) for these patients was 52%, and the overall survival (OS) was 80%.
There was a high rate of peripheral neuropathy (73%), but most cases resolved or improved over time.
Steven Horwitz, MD, of Memorial Sloan Kettering Cancer Center in New York, New York, and his colleagues presented these data as a poster at the 8th Annual T-cell Lymphoma Forum. The study was supported by Seattle Genetics and Millennium Pharmaceuticals.
The researchers presented data on 26 patients newly diagnosed with PTCL. Nineteen patients had systemic anaplastic large-cell lymphoma (ALCL; 16 ALK- and 3 ALK+), 2 had PTCL not otherwise specified, 2 had angioimmunoblastic T-cell lymphoma, 2 had adult T-cell leukemia/lymphoma, and 1 had enteropathy-associated T-cell lymphoma.
The patients’ median age was 56 (range, 21-82). Sixty-nine percent of patients had an IPI score of 2 or higher, and 73% had stage III/IV disease.
Treatment
The patients received BV+CHP every 3 weeks for 6 cycles. Those who achieved at least a partial remission could go on to receive up to 10 additional cycles of single-agent BV at 1.8 mg/kg every 3 weeks.
Twenty-three patients (88%) completed all 6 cycles of BV+CHP, and 21 patients (81%) went on to receive BV monotherapy, 11 of whom (42%) received all 10 cycles.
Fifteen patients (58%) discontinued treatment, 3 due to progressive disease, 3 due to investigator decision, 6 due to adverse events, and 3 due to patient decision.
After a median observation period of 38.7 months (range, 4.6 to 44.3), 77% of patients (n=20) remained on study.
Toxicity
The most common adverse events (occurring in at least 30% of patients) were nausea (69%), peripheral sensory neuropathy (69%), diarrhea (62%), fatigue (58%), alopecia (54%), dyspnea (46%), constipation (35%), myalgia (31%), peripheral edema (31%), chills (31%), anemia (31%), insomnia (31%), and febrile neutropenia.
The most common grade 3 or higher adverse events (occurring in at least 10% of patients) were febrile neutropenia (31%), neutropenia (23%), anemia (15%), and pulmonary embolism (12%).
There were 6 adverse events resulting in treatment discontinuation—peripheral sensory neuropathy (n=3), abdominal pain (n=1), asthenia (n=1), and peripheral motor neuropathy (n=1).
Seventy-three percent of patients (19/26) experienced peripheral neuropathy. Thirty-seven percent (n=7) had a complete resolution of neuropathy, and 58% (n=11) had some improvement. The median time to resolution was 1.3 months. Twelve patients (63%) had ongoing neuropathy at last follow-up, but most had grade 1 (n=10).
Response and survival
The objective response rate was 100%, and the complete response rate was 88% (n=23). One patient who had a partial response at the end of combination therapy achieved a complete response after going on to single-agent BV.
Twenty-one of the 26 patients are still alive—15 with ALCL and 6 with other PTCLs. Sixteen patients have not progressed—9 with ALCL and 5 with other PTCLs.
After progression, 5 patients received BV again, and 3 received stem cell transplants (2 allogeneic and 1 autologous).
The estimated 3-year PFS was 52%, and the estimated OS was 80%. The researchers noted that these rates compare favorably with the historical 3-year PFS and OS rates of 30% and 40%, respectively.
Researchers are currently conducting a phase 3 trial comparing BV+CHP with CHOP as frontline treatment of CD30+ mature T-cell lymphomas (ECHELON-2, NCT01777152). ![]()

Photo by Larry Young
SAN FRANCISCO—A combination treatment regimen can produce durable remissions in patients newly diagnosed with peripheral T-cell lymphoma (PTCL), results of a phase 1 study suggest.
The patients received brentuximab vedotin plus cyclophosphamide, doxorubicin, and prednisolone (BV+CHP). In some cases, this was followed by BV monotherapy.
The estimated 3-year progression-free survival (PFS) for these patients was 52%, and the overall survival (OS) was 80%.
There was a high rate of peripheral neuropathy (73%), but most cases resolved or improved over time.
Steven Horwitz, MD, of Memorial Sloan Kettering Cancer Center in New York, New York, and his colleagues presented these data as a poster at the 8th Annual T-cell Lymphoma Forum. The study was supported by Seattle Genetics and Millennium Pharmaceuticals.
The researchers presented data on 26 patients newly diagnosed with PTCL. Nineteen patients had systemic anaplastic large-cell lymphoma (ALCL; 16 ALK- and 3 ALK+), 2 had PTCL not otherwise specified, 2 had angioimmunoblastic T-cell lymphoma, 2 had adult T-cell leukemia/lymphoma, and 1 had enteropathy-associated T-cell lymphoma.
The patients’ median age was 56 (range, 21-82). Sixty-nine percent of patients had an IPI score of 2 or higher, and 73% had stage III/IV disease.
Treatment
The patients received BV+CHP every 3 weeks for 6 cycles. Those who achieved at least a partial remission could go on to receive up to 10 additional cycles of single-agent BV at 1.8 mg/kg every 3 weeks.
Twenty-three patients (88%) completed all 6 cycles of BV+CHP, and 21 patients (81%) went on to receive BV monotherapy, 11 of whom (42%) received all 10 cycles.
Fifteen patients (58%) discontinued treatment, 3 due to progressive disease, 3 due to investigator decision, 6 due to adverse events, and 3 due to patient decision.
After a median observation period of 38.7 months (range, 4.6 to 44.3), 77% of patients (n=20) remained on study.
Toxicity
The most common adverse events (occurring in at least 30% of patients) were nausea (69%), peripheral sensory neuropathy (69%), diarrhea (62%), fatigue (58%), alopecia (54%), dyspnea (46%), constipation (35%), myalgia (31%), peripheral edema (31%), chills (31%), anemia (31%), insomnia (31%), and febrile neutropenia.
The most common grade 3 or higher adverse events (occurring in at least 10% of patients) were febrile neutropenia (31%), neutropenia (23%), anemia (15%), and pulmonary embolism (12%).
There were 6 adverse events resulting in treatment discontinuation—peripheral sensory neuropathy (n=3), abdominal pain (n=1), asthenia (n=1), and peripheral motor neuropathy (n=1).
Seventy-three percent of patients (19/26) experienced peripheral neuropathy. Thirty-seven percent (n=7) had a complete resolution of neuropathy, and 58% (n=11) had some improvement. The median time to resolution was 1.3 months. Twelve patients (63%) had ongoing neuropathy at last follow-up, but most had grade 1 (n=10).
Response and survival
The objective response rate was 100%, and the complete response rate was 88% (n=23). One patient who had a partial response at the end of combination therapy achieved a complete response after going on to single-agent BV.
Twenty-one of the 26 patients are still alive—15 with ALCL and 6 with other PTCLs. Sixteen patients have not progressed—9 with ALCL and 5 with other PTCLs.
After progression, 5 patients received BV again, and 3 received stem cell transplants (2 allogeneic and 1 autologous).
The estimated 3-year PFS was 52%, and the estimated OS was 80%. The researchers noted that these rates compare favorably with the historical 3-year PFS and OS rates of 30% and 40%, respectively.
Researchers are currently conducting a phase 3 trial comparing BV+CHP with CHOP as frontline treatment of CD30+ mature T-cell lymphomas (ECHELON-2, NCT01777152). ![]()
Method may reduce toxicity of anticancer agents

Researchers believe they have found a way to make certain anticancer agents safer without compromising their efficacy.
The team noted that Aurora B kinase inhibitors and other agents targeting the cell cycle have proven effective but highly toxic in clinical trials.
In an attempt to solve this problem, the researchers turned to nanotechnology. They encapsulated the Aurora B kinase inhibitor AZD2811 in polymeric nanoparticles called Accurins.
Susan Ashton, of AstraZeneca in Macclesfield, Cheshire, UK, and her colleagues developed the Accurins and described the work in Science Translational Medicine. The work was funded by AstraZeneca.
The Accurins consist of block copolymers of poly-D,L-lactide and poly(ethylene glycol). The researchers used an ion pairing approach to efficiently encapsulate AZD2811 and control release of the drug.
They found the Accurins could release AZD2811 continuously for more than 1 week in vitro. The nanoparticles also reduced tumor phosphorylated histone H3 levels in vivo for up to 96 hours after a single administration.
The researchers tested the AZD2811 Accurins in mice with diffuse large B-cell lymphoma and rats with colorectal tumors. The nanoparticles accumulated specifically in tumors, where they slowly released AZD2811 to cancer cells.
Compared to AZD1152 (a water-soluble prodrug of AZD2811), the AZD2811 Accurins blocked tumor growth more effectively at one-half the drug dose and caused fewer side effects in the rodents.
Based on these results, the researchers said Accurins could provide efficacy and tolerability using a more convenient dosing regimen, which may extend the utility of Aurora B kinase inhibition to a broader range of hematologic and solid tumor malignancies.
A phase 1 study (NCT02579226) testing AZD2811 Accurins in advanced solid tumors is currently recruiting patients.
A related Focus article published in Science Translational Medicine offers more insights on how Accurin nanoparticles may help enhance the safety and antitumor activity of Aurora kinase inhibitors and other molecularly targeted drugs. ![]()

Researchers believe they have found a way to make certain anticancer agents safer without compromising their efficacy.
The team noted that Aurora B kinase inhibitors and other agents targeting the cell cycle have proven effective but highly toxic in clinical trials.
In an attempt to solve this problem, the researchers turned to nanotechnology. They encapsulated the Aurora B kinase inhibitor AZD2811 in polymeric nanoparticles called Accurins.
Susan Ashton, of AstraZeneca in Macclesfield, Cheshire, UK, and her colleagues developed the Accurins and described the work in Science Translational Medicine. The work was funded by AstraZeneca.
The Accurins consist of block copolymers of poly-D,L-lactide and poly(ethylene glycol). The researchers used an ion pairing approach to efficiently encapsulate AZD2811 and control release of the drug.
They found the Accurins could release AZD2811 continuously for more than 1 week in vitro. The nanoparticles also reduced tumor phosphorylated histone H3 levels in vivo for up to 96 hours after a single administration.
The researchers tested the AZD2811 Accurins in mice with diffuse large B-cell lymphoma and rats with colorectal tumors. The nanoparticles accumulated specifically in tumors, where they slowly released AZD2811 to cancer cells.
Compared to AZD1152 (a water-soluble prodrug of AZD2811), the AZD2811 Accurins blocked tumor growth more effectively at one-half the drug dose and caused fewer side effects in the rodents.
Based on these results, the researchers said Accurins could provide efficacy and tolerability using a more convenient dosing regimen, which may extend the utility of Aurora B kinase inhibition to a broader range of hematologic and solid tumor malignancies.
A phase 1 study (NCT02579226) testing AZD2811 Accurins in advanced solid tumors is currently recruiting patients.
A related Focus article published in Science Translational Medicine offers more insights on how Accurin nanoparticles may help enhance the safety and antitumor activity of Aurora kinase inhibitors and other molecularly targeted drugs. ![]()

Researchers believe they have found a way to make certain anticancer agents safer without compromising their efficacy.
The team noted that Aurora B kinase inhibitors and other agents targeting the cell cycle have proven effective but highly toxic in clinical trials.
In an attempt to solve this problem, the researchers turned to nanotechnology. They encapsulated the Aurora B kinase inhibitor AZD2811 in polymeric nanoparticles called Accurins.
Susan Ashton, of AstraZeneca in Macclesfield, Cheshire, UK, and her colleagues developed the Accurins and described the work in Science Translational Medicine. The work was funded by AstraZeneca.
The Accurins consist of block copolymers of poly-D,L-lactide and poly(ethylene glycol). The researchers used an ion pairing approach to efficiently encapsulate AZD2811 and control release of the drug.
They found the Accurins could release AZD2811 continuously for more than 1 week in vitro. The nanoparticles also reduced tumor phosphorylated histone H3 levels in vivo for up to 96 hours after a single administration.
The researchers tested the AZD2811 Accurins in mice with diffuse large B-cell lymphoma and rats with colorectal tumors. The nanoparticles accumulated specifically in tumors, where they slowly released AZD2811 to cancer cells.
Compared to AZD1152 (a water-soluble prodrug of AZD2811), the AZD2811 Accurins blocked tumor growth more effectively at one-half the drug dose and caused fewer side effects in the rodents.
Based on these results, the researchers said Accurins could provide efficacy and tolerability using a more convenient dosing regimen, which may extend the utility of Aurora B kinase inhibition to a broader range of hematologic and solid tumor malignancies.
A phase 1 study (NCT02579226) testing AZD2811 Accurins in advanced solid tumors is currently recruiting patients.
A related Focus article published in Science Translational Medicine offers more insights on how Accurin nanoparticles may help enhance the safety and antitumor activity of Aurora kinase inhibitors and other molecularly targeted drugs. ![]()
Chemo regimen can be ‘highly effective’ against ENKTL

Photo by Larry Young
SAN FRANCISCO—A 3-agent chemotherapy regimen can be “highly effective” in patients with extranodal natural killer/T-cell lymphoma (ENKTL), according to researchers.
In a single-center study, this regimen—pegaspargase, gemcitabine, and oxaliplatin (P-GEMOX)—followed by extensive involved-field radiotherapy (EIFRT) produced high rates of long-term overall survival (OS) and progression-free survival (PFS) in newly diagnosed patients with stage I/II ENKTL.
P-GEMOX also proved effective—though to a much lesser degree—in advanced, relapsed, or refractory ENKTL, and these patients appeared to benefit from autologous stem cell transplant (auto-SCT) as consolidation.
Toxicity associated with P-GEMOX was mild to moderate and tolerable, according to Hui-Qiang Huang, MD, PhD, of State Key Laboratory of Oncology in Southern China, Guangzhou, China.
Dr Huang presented these results at the 8th Annual T-cell Lymphoma Forum.
Newly diagnosed patients
Dr Huang and his colleagues studied 56 patients newly diagnosed with stage I/II, nasal-type ENKTL. Most patients were younger than 60 years of age (80.4%, n=45).
About 79% (n=44) had an ECOG status of 0, and 21.4% (n=12) had a status of 1. About 61% (n=34) had stage I disease, and 39.3% (n=22) had stage II.
All patients received P-GEMOX—gemcitabine at 1000 mg/m2 on days 1 and 8, oxaliplatin at 150 mg/m2 on day 1, and pegaspargase at 2000 U/m2 on day 1. Doses could be adjusted in the event of toxicity.
The regimen was repeated every 3 weeks for a maximum of 4 cycles. Patients then underwent EIFRT—56 Gy in 28 fractions over 4 weeks.
The overall response rate (ORR) after P-GEMOX was 89.3% (50/56). Thirty-five patients achieved a complete response (CR), 15 had a partial response (PR), and 4 had stable disease (SD).
After EIFRT, the ORR increased to 94.6% (53/56). Fifty patients had a CR, 3 had a PR, and 1 had SD.
The median follow-up was 35.2 months (range, 10.6-51.4). Six patients relapsed, and the median time to relapse was 6.2 months.
Five patients died of disease progression. The median time to death was 10.9 months after the completion of EIFRT.
The 4-year OS rate was 90.7±4.0%, and the 4-year PFS rate was 89.1±4.2%.
OS and PFS were superior in patients with stage I disease as compared to stage II (P=0.056 and 0.023, respectively). And OS and PFS were superior in patients who responded to P-GEMOX (P=0.004 and 0.001, respectively).
There were no treatment-related deaths. The most common toxicities (occurring in more than 50% of patients) after P-GEMOX were neutropenia (80.3%), thrombocytopenia (55.3%), and hypoproteinemia (75.0%).
The most common grade 3/4 toxicities (occurring in more than 10% of patients) were granulocytosis (23.2%), thrombocytopenia (19.6%), and hypoproteinemia (10.7%).
Advanced & relapsed/refractory patients
Dr Huang and his colleagues also studied 60 patients with newly diagnosed, stage III/IV ENKTL (25%, n=15), relapsed ENKTL (21.7%, n=19), or refractory disease (43.3%, n=26). Seventy percent of these patients (n=42) had nasal-type ENKTL.
Most patients were younger than 60 years of age (91.7%, n=55). About 73% (n=44) had an ECOG status of 0-1, and 26.7% (n=16) had a status of 2. Fifteen percent of patients (n=9) had stage I disease, 16.7% (n=10) had stage II, 35% (n=21) had stage III, and 33.3% (n=20) had stage IV.
The patients received the same P-GEMOX regimen as the newly diagnosed, stage I/II patients, but they did not receive EIFRT, and responders could undergo auto-SCT.
For the whole cohort, the ORR after P-GEMOX was 70% (42/60). Twenty-one patients had a CR, 21 had a PR, and 9 had SD.
In the newly diagnosed patients, the ORR was 80% (12/15). Four patients had a CR, 8 had a PR, and 2 had SD. In the relapsed/refractory patients, the ORR was 66.7% (30/45). Seventeen patients had a CR, 13 had a PR, and 7 had SD.
The 4-year OS was 43.0±7.3%, and the 4-year PFS was 36.5±6.9%.
There was no significant difference in OS or PFS between the newly diagnosed and relapsed/refractory patients (P=0.653 and 0.825, respectively). However, there was a significant difference in PFS and OS between responders and non-responders (P<0.001 for both).
There was a difference in 3-year OS between patients who went on to auto-SCT and those did not, although it did not reach statistical significance (P=0.08). Eleven patients who achieved a CR went on to auto-SCT.
There were no treatment-related deaths. The most common toxicities (occurring in more than 50% of patients) after P-GEMOX were neutropenia (85%), hypoproteinemia (88.3%), anemia (71.6%), fibrinogen decrease (68.3%), and anorexia (53.3%).
The most common grade 3/4 toxicities (occurring in more than 10% of patients) were neutropenia (31.6%), hypoproteinemia (13.3%), and thrombocytopenia (11.7%).
Dr Huang said this research suggests P-GEMOX can be effective for patients with newly diagnosed or previously treated ENKTL. The next step is to investigate which novel agents could be added to the regimen to improve its efficacy. ![]()

Photo by Larry Young
SAN FRANCISCO—A 3-agent chemotherapy regimen can be “highly effective” in patients with extranodal natural killer/T-cell lymphoma (ENKTL), according to researchers.
In a single-center study, this regimen—pegaspargase, gemcitabine, and oxaliplatin (P-GEMOX)—followed by extensive involved-field radiotherapy (EIFRT) produced high rates of long-term overall survival (OS) and progression-free survival (PFS) in newly diagnosed patients with stage I/II ENKTL.
P-GEMOX also proved effective—though to a much lesser degree—in advanced, relapsed, or refractory ENKTL, and these patients appeared to benefit from autologous stem cell transplant (auto-SCT) as consolidation.
Toxicity associated with P-GEMOX was mild to moderate and tolerable, according to Hui-Qiang Huang, MD, PhD, of State Key Laboratory of Oncology in Southern China, Guangzhou, China.
Dr Huang presented these results at the 8th Annual T-cell Lymphoma Forum.
Newly diagnosed patients
Dr Huang and his colleagues studied 56 patients newly diagnosed with stage I/II, nasal-type ENKTL. Most patients were younger than 60 years of age (80.4%, n=45).
About 79% (n=44) had an ECOG status of 0, and 21.4% (n=12) had a status of 1. About 61% (n=34) had stage I disease, and 39.3% (n=22) had stage II.
All patients received P-GEMOX—gemcitabine at 1000 mg/m2 on days 1 and 8, oxaliplatin at 150 mg/m2 on day 1, and pegaspargase at 2000 U/m2 on day 1. Doses could be adjusted in the event of toxicity.
The regimen was repeated every 3 weeks for a maximum of 4 cycles. Patients then underwent EIFRT—56 Gy in 28 fractions over 4 weeks.
The overall response rate (ORR) after P-GEMOX was 89.3% (50/56). Thirty-five patients achieved a complete response (CR), 15 had a partial response (PR), and 4 had stable disease (SD).
After EIFRT, the ORR increased to 94.6% (53/56). Fifty patients had a CR, 3 had a PR, and 1 had SD.
The median follow-up was 35.2 months (range, 10.6-51.4). Six patients relapsed, and the median time to relapse was 6.2 months.
Five patients died of disease progression. The median time to death was 10.9 months after the completion of EIFRT.
The 4-year OS rate was 90.7±4.0%, and the 4-year PFS rate was 89.1±4.2%.
OS and PFS were superior in patients with stage I disease as compared to stage II (P=0.056 and 0.023, respectively). And OS and PFS were superior in patients who responded to P-GEMOX (P=0.004 and 0.001, respectively).
There were no treatment-related deaths. The most common toxicities (occurring in more than 50% of patients) after P-GEMOX were neutropenia (80.3%), thrombocytopenia (55.3%), and hypoproteinemia (75.0%).
The most common grade 3/4 toxicities (occurring in more than 10% of patients) were granulocytosis (23.2%), thrombocytopenia (19.6%), and hypoproteinemia (10.7%).
Advanced & relapsed/refractory patients
Dr Huang and his colleagues also studied 60 patients with newly diagnosed, stage III/IV ENKTL (25%, n=15), relapsed ENKTL (21.7%, n=19), or refractory disease (43.3%, n=26). Seventy percent of these patients (n=42) had nasal-type ENKTL.
Most patients were younger than 60 years of age (91.7%, n=55). About 73% (n=44) had an ECOG status of 0-1, and 26.7% (n=16) had a status of 2. Fifteen percent of patients (n=9) had stage I disease, 16.7% (n=10) had stage II, 35% (n=21) had stage III, and 33.3% (n=20) had stage IV.
The patients received the same P-GEMOX regimen as the newly diagnosed, stage I/II patients, but they did not receive EIFRT, and responders could undergo auto-SCT.
For the whole cohort, the ORR after P-GEMOX was 70% (42/60). Twenty-one patients had a CR, 21 had a PR, and 9 had SD.
In the newly diagnosed patients, the ORR was 80% (12/15). Four patients had a CR, 8 had a PR, and 2 had SD. In the relapsed/refractory patients, the ORR was 66.7% (30/45). Seventeen patients had a CR, 13 had a PR, and 7 had SD.
The 4-year OS was 43.0±7.3%, and the 4-year PFS was 36.5±6.9%.
There was no significant difference in OS or PFS between the newly diagnosed and relapsed/refractory patients (P=0.653 and 0.825, respectively). However, there was a significant difference in PFS and OS between responders and non-responders (P<0.001 for both).
There was a difference in 3-year OS between patients who went on to auto-SCT and those did not, although it did not reach statistical significance (P=0.08). Eleven patients who achieved a CR went on to auto-SCT.
There were no treatment-related deaths. The most common toxicities (occurring in more than 50% of patients) after P-GEMOX were neutropenia (85%), hypoproteinemia (88.3%), anemia (71.6%), fibrinogen decrease (68.3%), and anorexia (53.3%).
The most common grade 3/4 toxicities (occurring in more than 10% of patients) were neutropenia (31.6%), hypoproteinemia (13.3%), and thrombocytopenia (11.7%).
Dr Huang said this research suggests P-GEMOX can be effective for patients with newly diagnosed or previously treated ENKTL. The next step is to investigate which novel agents could be added to the regimen to improve its efficacy. ![]()

Photo by Larry Young
SAN FRANCISCO—A 3-agent chemotherapy regimen can be “highly effective” in patients with extranodal natural killer/T-cell lymphoma (ENKTL), according to researchers.
In a single-center study, this regimen—pegaspargase, gemcitabine, and oxaliplatin (P-GEMOX)—followed by extensive involved-field radiotherapy (EIFRT) produced high rates of long-term overall survival (OS) and progression-free survival (PFS) in newly diagnosed patients with stage I/II ENKTL.
P-GEMOX also proved effective—though to a much lesser degree—in advanced, relapsed, or refractory ENKTL, and these patients appeared to benefit from autologous stem cell transplant (auto-SCT) as consolidation.
Toxicity associated with P-GEMOX was mild to moderate and tolerable, according to Hui-Qiang Huang, MD, PhD, of State Key Laboratory of Oncology in Southern China, Guangzhou, China.
Dr Huang presented these results at the 8th Annual T-cell Lymphoma Forum.
Newly diagnosed patients
Dr Huang and his colleagues studied 56 patients newly diagnosed with stage I/II, nasal-type ENKTL. Most patients were younger than 60 years of age (80.4%, n=45).
About 79% (n=44) had an ECOG status of 0, and 21.4% (n=12) had a status of 1. About 61% (n=34) had stage I disease, and 39.3% (n=22) had stage II.
All patients received P-GEMOX—gemcitabine at 1000 mg/m2 on days 1 and 8, oxaliplatin at 150 mg/m2 on day 1, and pegaspargase at 2000 U/m2 on day 1. Doses could be adjusted in the event of toxicity.
The regimen was repeated every 3 weeks for a maximum of 4 cycles. Patients then underwent EIFRT—56 Gy in 28 fractions over 4 weeks.
The overall response rate (ORR) after P-GEMOX was 89.3% (50/56). Thirty-five patients achieved a complete response (CR), 15 had a partial response (PR), and 4 had stable disease (SD).
After EIFRT, the ORR increased to 94.6% (53/56). Fifty patients had a CR, 3 had a PR, and 1 had SD.
The median follow-up was 35.2 months (range, 10.6-51.4). Six patients relapsed, and the median time to relapse was 6.2 months.
Five patients died of disease progression. The median time to death was 10.9 months after the completion of EIFRT.
The 4-year OS rate was 90.7±4.0%, and the 4-year PFS rate was 89.1±4.2%.
OS and PFS were superior in patients with stage I disease as compared to stage II (P=0.056 and 0.023, respectively). And OS and PFS were superior in patients who responded to P-GEMOX (P=0.004 and 0.001, respectively).
There were no treatment-related deaths. The most common toxicities (occurring in more than 50% of patients) after P-GEMOX were neutropenia (80.3%), thrombocytopenia (55.3%), and hypoproteinemia (75.0%).
The most common grade 3/4 toxicities (occurring in more than 10% of patients) were granulocytosis (23.2%), thrombocytopenia (19.6%), and hypoproteinemia (10.7%).
Advanced & relapsed/refractory patients
Dr Huang and his colleagues also studied 60 patients with newly diagnosed, stage III/IV ENKTL (25%, n=15), relapsed ENKTL (21.7%, n=19), or refractory disease (43.3%, n=26). Seventy percent of these patients (n=42) had nasal-type ENKTL.
Most patients were younger than 60 years of age (91.7%, n=55). About 73% (n=44) had an ECOG status of 0-1, and 26.7% (n=16) had a status of 2. Fifteen percent of patients (n=9) had stage I disease, 16.7% (n=10) had stage II, 35% (n=21) had stage III, and 33.3% (n=20) had stage IV.
The patients received the same P-GEMOX regimen as the newly diagnosed, stage I/II patients, but they did not receive EIFRT, and responders could undergo auto-SCT.
For the whole cohort, the ORR after P-GEMOX was 70% (42/60). Twenty-one patients had a CR, 21 had a PR, and 9 had SD.
In the newly diagnosed patients, the ORR was 80% (12/15). Four patients had a CR, 8 had a PR, and 2 had SD. In the relapsed/refractory patients, the ORR was 66.7% (30/45). Seventeen patients had a CR, 13 had a PR, and 7 had SD.
The 4-year OS was 43.0±7.3%, and the 4-year PFS was 36.5±6.9%.
There was no significant difference in OS or PFS between the newly diagnosed and relapsed/refractory patients (P=0.653 and 0.825, respectively). However, there was a significant difference in PFS and OS between responders and non-responders (P<0.001 for both).
There was a difference in 3-year OS between patients who went on to auto-SCT and those did not, although it did not reach statistical significance (P=0.08). Eleven patients who achieved a CR went on to auto-SCT.
There were no treatment-related deaths. The most common toxicities (occurring in more than 50% of patients) after P-GEMOX were neutropenia (85%), hypoproteinemia (88.3%), anemia (71.6%), fibrinogen decrease (68.3%), and anorexia (53.3%).
The most common grade 3/4 toxicities (occurring in more than 10% of patients) were neutropenia (31.6%), hypoproteinemia (13.3%), and thrombocytopenia (11.7%).
Dr Huang said this research suggests P-GEMOX can be effective for patients with newly diagnosed or previously treated ENKTL. The next step is to investigate which novel agents could be added to the regimen to improve its efficacy. ![]()
Dual inhibitor could treat ATLL

Photo by Larry Young
SAN FRANCISCO—Preclinical research suggests a compound that inhibits both EZH1 and EZH2 could be effective against adult T-cell leukemia/lymphoma (ATLL).
The compound, known as OR-S1, has demonstrated activity against ATLL in vitro and in vivo.
Researchers said OR-S1 reversed epigenetic disruption in ATLL cells, selectively eliminated both ATLL cells and cells infected with human T-cell leukemia virus type I (HTLV-1), and inhibited tumor growth in mouse models of ATLL.
Based on these results, the researchers are planning a phase 1 study of the compound.
Makoto Yamagishi, PhD, of The University of Tokyo in Japan, described the preclinical research with OR-S1 and discussed the rationale for developing the compound at the 8th Annual T-cell Lymphoma Forum. The work was carried out in collaboration with Daiichi Sankyo Co., Ltd.
“We do not precisely understand the molecular mechanism of ATLL development, including genetic and epigenetic abnormalities,” Dr Yamagishi noted.
To gain some insight, he and his colleagues performed microRNA profiling, gene expression profiling, and histone methylation/epigenetic factor profiling on cells from ATLL patients and CD4+ T cells from healthy donors.
The team found that PRC2 factors were significantly upregulated in ATLL. EZH2 was the most upregulated histone methyltransferase, but ATLL cells did not have active mutations in the EZH2 gene. Dr Yamagishi said this suggests EZH2 upregulation is critical for the ATLL-specific epigenome.
“At long last, we determined the epigenetic pattern of ATLL,” he said. “ATLL cells showed specific and significant reprogramming of the epigenome, especially H3K27me3 gain. We found abnormal H3K27me3 change in half of genes, and gain was dominant.”
“But, interestingly, the methylated genes are specific in ATLL and do not overlap with other EZH2-dependent cell types, such as embryonic stem cells and diffuse large B-cell lymphoma cells. So ATLL has a very unique epigenome.”
Further investigation revealed that both EZH1 and EZH2 contribute to ATLL-specific epigenetic deregulation. More than 80% of H3K27me3 accumulated genes are occupied by EZH1 and/or EZH2.
So the researchers decided to examine the effects of knocking down EZH1 and EZH2 in ATLL cells.
Compared with knockdown of either gene alone, double knockdown synergistically influenced target gene expression. It led to complete dysfunction of the Polycomb family and had a significant impact on ATLL cell survival.
The researchers also found that EZH1 depletion enhanced ATLL cells’ sensitivity to the EZH2 inhibitor GSK126.
So the team decided to develop a dual EZH1/EZH2 inhibitor. They created OR-S1, which showed “strong activity” against EZH1 and EZH2 but none of the other histone methyltransferases tested.
In in vitro experiments, OR-S1 completely removed H3K27me3 and significantly reduced cell growth in the ATLL-derived cell line TL-Om1.
The drug also reduced cell viability in primary ATLL cells. All 15 samples tested proved sensitive to OR-S1. In addition, OR-S1 treatment selectively removed HTLV-1-infected cells from samples taken from 16 asymptomatic carriers.
Finally, OR-S1 proved active in mice. The drug prevented engraftment of ATLL cells in immunocompromised mice. All 6 OR-S1-treated mice were alive and tumor-free at 49 days, whereas 5 of 6 control mice had died (P=0.0041).
In mice treated after ATLL cell engraftment, OR-S1 reduced tumor growth without causing notable weight loss.
“Synthetic lethality by targeting EZH1 and EZH2 is promising [for ATLL],” Dr Yamagishi said. “Toxicity tests suggest the EZH1/2 dual inhibitor may be sufficient for clinical use, so we are now planning a phase 1 study.” ![]()

Photo by Larry Young
SAN FRANCISCO—Preclinical research suggests a compound that inhibits both EZH1 and EZH2 could be effective against adult T-cell leukemia/lymphoma (ATLL).
The compound, known as OR-S1, has demonstrated activity against ATLL in vitro and in vivo.
Researchers said OR-S1 reversed epigenetic disruption in ATLL cells, selectively eliminated both ATLL cells and cells infected with human T-cell leukemia virus type I (HTLV-1), and inhibited tumor growth in mouse models of ATLL.
Based on these results, the researchers are planning a phase 1 study of the compound.
Makoto Yamagishi, PhD, of The University of Tokyo in Japan, described the preclinical research with OR-S1 and discussed the rationale for developing the compound at the 8th Annual T-cell Lymphoma Forum. The work was carried out in collaboration with Daiichi Sankyo Co., Ltd.
“We do not precisely understand the molecular mechanism of ATLL development, including genetic and epigenetic abnormalities,” Dr Yamagishi noted.
To gain some insight, he and his colleagues performed microRNA profiling, gene expression profiling, and histone methylation/epigenetic factor profiling on cells from ATLL patients and CD4+ T cells from healthy donors.
The team found that PRC2 factors were significantly upregulated in ATLL. EZH2 was the most upregulated histone methyltransferase, but ATLL cells did not have active mutations in the EZH2 gene. Dr Yamagishi said this suggests EZH2 upregulation is critical for the ATLL-specific epigenome.
“At long last, we determined the epigenetic pattern of ATLL,” he said. “ATLL cells showed specific and significant reprogramming of the epigenome, especially H3K27me3 gain. We found abnormal H3K27me3 change in half of genes, and gain was dominant.”
“But, interestingly, the methylated genes are specific in ATLL and do not overlap with other EZH2-dependent cell types, such as embryonic stem cells and diffuse large B-cell lymphoma cells. So ATLL has a very unique epigenome.”
Further investigation revealed that both EZH1 and EZH2 contribute to ATLL-specific epigenetic deregulation. More than 80% of H3K27me3 accumulated genes are occupied by EZH1 and/or EZH2.
So the researchers decided to examine the effects of knocking down EZH1 and EZH2 in ATLL cells.
Compared with knockdown of either gene alone, double knockdown synergistically influenced target gene expression. It led to complete dysfunction of the Polycomb family and had a significant impact on ATLL cell survival.
The researchers also found that EZH1 depletion enhanced ATLL cells’ sensitivity to the EZH2 inhibitor GSK126.
So the team decided to develop a dual EZH1/EZH2 inhibitor. They created OR-S1, which showed “strong activity” against EZH1 and EZH2 but none of the other histone methyltransferases tested.
In in vitro experiments, OR-S1 completely removed H3K27me3 and significantly reduced cell growth in the ATLL-derived cell line TL-Om1.
The drug also reduced cell viability in primary ATLL cells. All 15 samples tested proved sensitive to OR-S1. In addition, OR-S1 treatment selectively removed HTLV-1-infected cells from samples taken from 16 asymptomatic carriers.
Finally, OR-S1 proved active in mice. The drug prevented engraftment of ATLL cells in immunocompromised mice. All 6 OR-S1-treated mice were alive and tumor-free at 49 days, whereas 5 of 6 control mice had died (P=0.0041).
In mice treated after ATLL cell engraftment, OR-S1 reduced tumor growth without causing notable weight loss.
“Synthetic lethality by targeting EZH1 and EZH2 is promising [for ATLL],” Dr Yamagishi said. “Toxicity tests suggest the EZH1/2 dual inhibitor may be sufficient for clinical use, so we are now planning a phase 1 study.” ![]()

Photo by Larry Young
SAN FRANCISCO—Preclinical research suggests a compound that inhibits both EZH1 and EZH2 could be effective against adult T-cell leukemia/lymphoma (ATLL).
The compound, known as OR-S1, has demonstrated activity against ATLL in vitro and in vivo.
Researchers said OR-S1 reversed epigenetic disruption in ATLL cells, selectively eliminated both ATLL cells and cells infected with human T-cell leukemia virus type I (HTLV-1), and inhibited tumor growth in mouse models of ATLL.
Based on these results, the researchers are planning a phase 1 study of the compound.
Makoto Yamagishi, PhD, of The University of Tokyo in Japan, described the preclinical research with OR-S1 and discussed the rationale for developing the compound at the 8th Annual T-cell Lymphoma Forum. The work was carried out in collaboration with Daiichi Sankyo Co., Ltd.
“We do not precisely understand the molecular mechanism of ATLL development, including genetic and epigenetic abnormalities,” Dr Yamagishi noted.
To gain some insight, he and his colleagues performed microRNA profiling, gene expression profiling, and histone methylation/epigenetic factor profiling on cells from ATLL patients and CD4+ T cells from healthy donors.
The team found that PRC2 factors were significantly upregulated in ATLL. EZH2 was the most upregulated histone methyltransferase, but ATLL cells did not have active mutations in the EZH2 gene. Dr Yamagishi said this suggests EZH2 upregulation is critical for the ATLL-specific epigenome.
“At long last, we determined the epigenetic pattern of ATLL,” he said. “ATLL cells showed specific and significant reprogramming of the epigenome, especially H3K27me3 gain. We found abnormal H3K27me3 change in half of genes, and gain was dominant.”
“But, interestingly, the methylated genes are specific in ATLL and do not overlap with other EZH2-dependent cell types, such as embryonic stem cells and diffuse large B-cell lymphoma cells. So ATLL has a very unique epigenome.”
Further investigation revealed that both EZH1 and EZH2 contribute to ATLL-specific epigenetic deregulation. More than 80% of H3K27me3 accumulated genes are occupied by EZH1 and/or EZH2.
So the researchers decided to examine the effects of knocking down EZH1 and EZH2 in ATLL cells.
Compared with knockdown of either gene alone, double knockdown synergistically influenced target gene expression. It led to complete dysfunction of the Polycomb family and had a significant impact on ATLL cell survival.
The researchers also found that EZH1 depletion enhanced ATLL cells’ sensitivity to the EZH2 inhibitor GSK126.
So the team decided to develop a dual EZH1/EZH2 inhibitor. They created OR-S1, which showed “strong activity” against EZH1 and EZH2 but none of the other histone methyltransferases tested.
In in vitro experiments, OR-S1 completely removed H3K27me3 and significantly reduced cell growth in the ATLL-derived cell line TL-Om1.
The drug also reduced cell viability in primary ATLL cells. All 15 samples tested proved sensitive to OR-S1. In addition, OR-S1 treatment selectively removed HTLV-1-infected cells from samples taken from 16 asymptomatic carriers.
Finally, OR-S1 proved active in mice. The drug prevented engraftment of ATLL cells in immunocompromised mice. All 6 OR-S1-treated mice were alive and tumor-free at 49 days, whereas 5 of 6 control mice had died (P=0.0041).
In mice treated after ATLL cell engraftment, OR-S1 reduced tumor growth without causing notable weight loss.
“Synthetic lethality by targeting EZH1 and EZH2 is promising [for ATLL],” Dr Yamagishi said. “Toxicity tests suggest the EZH1/2 dual inhibitor may be sufficient for clinical use, so we are now planning a phase 1 study.” ![]()
Immunotherapy proves active against MF, SS

Photo by Larry Young
SAN FRANCISCO—The PD-1-blocking antibody pembrolizumab can produce “significant objective clinical responses” in patients with relapsed or refractory cutaneous T-cell lymphoma, according to researchers.
The drug elicited partial responses in 33% of patients enrolled in a phase 2 study. Half of the responders had mycosis fungoides (MF), and half had Sézary syndrome (SS).
All responses are ongoing, and a few patients with stable disease remain on treatment, so they may convert to partial responses, according to Youn Kim, MD, of Stanford University School of Medicine in California.
Dr Kim presented this research at the 8th Annual T-cell Lymphoma Forum.
The study was conducted by the Cancer Immunotherapy Trials Network (CITN) and supported by the National Cancer Institute and Merck, the company developing pembrolizumab.
“There’s good rationale for immune checkpoint blockade in [MF and SS],” Dr Kim said. “There’s systemic and local immune impairment in MF and Sézary, and there’s mounting evidence that T-cell immunity is critical for meaningful antitumor response.”
“[T]umor-infiltrating CD8+ T cells [have been] associated with improved survival, and therapies which augment T-cell function are effective in [MF and SS]. PD-1 and PD-L1 are very well expressed in the tissue and blood, [and] there’s good genomic evidence of immune evasion in [MF and SS].”
With all this in mind, Dr Kim and her colleagues conducted their phase 2 trial of pembrolizumab in 24 patients with relapsed or refractory MF/SS. Patients were excluded if they had central nervous system disease, autoimmune disease, immunodeficiency, or had received immunosuppressive therapy within 7 days.
The patients’ median age was 67 (range, 44-85), and most were male (75%). Thirty-eight percent of patients (n=9) had MF, and 62% (n=15) had SS. Twelve percent (n=3) had large-cell transformation.
Most patients had Stage IVA disease (62%, n=15), followed by IIIB (13%, n=3), IIIA (13%, n=3), IIB (8%, n=2), and IB (4%, n=1). The median number of prior systemic therapies was 4 (range, 1-10).
Treatment and response
Patients received pembrolizumab at 2 mg/kg intravenously every 3 weeks and were allowed to continue therapy for up to 2 years. Dr Kim noted that patients could continue treatment even after the initial documentation of progressive disease (PD) due to the possibility of immune-mediated flare reactions.
“So it’s the investigator’s decision to allow treatment beyond the initial PD,” she said. “However, if there’s confirmation of PD, those people will be removed.”
The median follow-up was 21 weeks (range, 7-39). Eight patients responded to treatment (according to global response criteria), all of which were partial responses. Four of the responders had MF, and 4 had SS. Responses occurred across all disease stages except IB.
“The range of prior therapies varied in the responders,” Dr Kim noted. “People think [patients tend to respond to] immunotherapy [if they are only] mildly [pre-]treated, but that was not the case. Heavily treated patients had great responses to pembrolizumab.”
All responses are ongoing, with a median duration of 13+ weeks (range, 3+ to 30+). The median time to response was 11 weeks (range, 8-22).
Dr Kim noted that 1 responder discontinued treatment because of a severe adverse event, but this patient remains in response without having received subsequent treatment.
The median best mSWAT (modified Skin Weighted Assessment Tool) reduction was 16%. Two patients had near-complete responses in the skin, and 2 patients with stable disease in the skin continue to improve.
Eleven of the 15 SS patients had measurable Sézary burden pre-treatment. And 3 of these patients had a greater than 50% reduction in Sézary count after treatment.
Four patients with stable disease are still on treatment. And at 20 weeks, 75% of patients are progression-free, according to Kaplan-Meier estimates.
Adverse events
Drug-related adverse events occurring at least twice included skin eruptions (21%, n=5), anemia (13%, n=3), decrease in white blood cell count (8%, n=2), elevated liver tests (8%, n=2), diarrhea (8%, n=2), fever (8%, n=2), and face edema (8%, n=2).
Grade 3/4 adverse events included skin eruptions (8%, n=2), anemia (8%, n=2), elevated liver tests (4%, n=1), and face edema (4%, n=1). Skin eruptions (of all grades) included exfoliative dermatitis (n=2), immune-mediated skin flare (n=2), and excessive peeling/edema (n=1).
There were no drug-related serious adverse events. The cause of the aforementioned serious adverse event (which prompted the responding patient to discontinue treatment) could not be determined.
There were 4 patients who did not report any adverse events, regardless of attribution.
In closing, Dr Kim said it is important to conduct biomarker correlative studies to understand the tumor escape mechanisms and enrich the response population.
She and her colleagues at CITN are now exploring the use of pembrolizumab in combination therapy. They are considering combining the drug with interferon-gamma, interleukin-12, low-dose total skin radiation, intratumoral ipilimumab, or Toll-like receptor agonists. ![]()

Photo by Larry Young
SAN FRANCISCO—The PD-1-blocking antibody pembrolizumab can produce “significant objective clinical responses” in patients with relapsed or refractory cutaneous T-cell lymphoma, according to researchers.
The drug elicited partial responses in 33% of patients enrolled in a phase 2 study. Half of the responders had mycosis fungoides (MF), and half had Sézary syndrome (SS).
All responses are ongoing, and a few patients with stable disease remain on treatment, so they may convert to partial responses, according to Youn Kim, MD, of Stanford University School of Medicine in California.
Dr Kim presented this research at the 8th Annual T-cell Lymphoma Forum.
The study was conducted by the Cancer Immunotherapy Trials Network (CITN) and supported by the National Cancer Institute and Merck, the company developing pembrolizumab.
“There’s good rationale for immune checkpoint blockade in [MF and SS],” Dr Kim said. “There’s systemic and local immune impairment in MF and Sézary, and there’s mounting evidence that T-cell immunity is critical for meaningful antitumor response.”
“[T]umor-infiltrating CD8+ T cells [have been] associated with improved survival, and therapies which augment T-cell function are effective in [MF and SS]. PD-1 and PD-L1 are very well expressed in the tissue and blood, [and] there’s good genomic evidence of immune evasion in [MF and SS].”
With all this in mind, Dr Kim and her colleagues conducted their phase 2 trial of pembrolizumab in 24 patients with relapsed or refractory MF/SS. Patients were excluded if they had central nervous system disease, autoimmune disease, immunodeficiency, or had received immunosuppressive therapy within 7 days.
The patients’ median age was 67 (range, 44-85), and most were male (75%). Thirty-eight percent of patients (n=9) had MF, and 62% (n=15) had SS. Twelve percent (n=3) had large-cell transformation.
Most patients had Stage IVA disease (62%, n=15), followed by IIIB (13%, n=3), IIIA (13%, n=3), IIB (8%, n=2), and IB (4%, n=1). The median number of prior systemic therapies was 4 (range, 1-10).
Treatment and response
Patients received pembrolizumab at 2 mg/kg intravenously every 3 weeks and were allowed to continue therapy for up to 2 years. Dr Kim noted that patients could continue treatment even after the initial documentation of progressive disease (PD) due to the possibility of immune-mediated flare reactions.
“So it’s the investigator’s decision to allow treatment beyond the initial PD,” she said. “However, if there’s confirmation of PD, those people will be removed.”
The median follow-up was 21 weeks (range, 7-39). Eight patients responded to treatment (according to global response criteria), all of which were partial responses. Four of the responders had MF, and 4 had SS. Responses occurred across all disease stages except IB.
“The range of prior therapies varied in the responders,” Dr Kim noted. “People think [patients tend to respond to] immunotherapy [if they are only] mildly [pre-]treated, but that was not the case. Heavily treated patients had great responses to pembrolizumab.”
All responses are ongoing, with a median duration of 13+ weeks (range, 3+ to 30+). The median time to response was 11 weeks (range, 8-22).
Dr Kim noted that 1 responder discontinued treatment because of a severe adverse event, but this patient remains in response without having received subsequent treatment.
The median best mSWAT (modified Skin Weighted Assessment Tool) reduction was 16%. Two patients had near-complete responses in the skin, and 2 patients with stable disease in the skin continue to improve.
Eleven of the 15 SS patients had measurable Sézary burden pre-treatment. And 3 of these patients had a greater than 50% reduction in Sézary count after treatment.
Four patients with stable disease are still on treatment. And at 20 weeks, 75% of patients are progression-free, according to Kaplan-Meier estimates.
Adverse events
Drug-related adverse events occurring at least twice included skin eruptions (21%, n=5), anemia (13%, n=3), decrease in white blood cell count (8%, n=2), elevated liver tests (8%, n=2), diarrhea (8%, n=2), fever (8%, n=2), and face edema (8%, n=2).
Grade 3/4 adverse events included skin eruptions (8%, n=2), anemia (8%, n=2), elevated liver tests (4%, n=1), and face edema (4%, n=1). Skin eruptions (of all grades) included exfoliative dermatitis (n=2), immune-mediated skin flare (n=2), and excessive peeling/edema (n=1).
There were no drug-related serious adverse events. The cause of the aforementioned serious adverse event (which prompted the responding patient to discontinue treatment) could not be determined.
There were 4 patients who did not report any adverse events, regardless of attribution.
In closing, Dr Kim said it is important to conduct biomarker correlative studies to understand the tumor escape mechanisms and enrich the response population.
She and her colleagues at CITN are now exploring the use of pembrolizumab in combination therapy. They are considering combining the drug with interferon-gamma, interleukin-12, low-dose total skin radiation, intratumoral ipilimumab, or Toll-like receptor agonists. ![]()

Photo by Larry Young
SAN FRANCISCO—The PD-1-blocking antibody pembrolizumab can produce “significant objective clinical responses” in patients with relapsed or refractory cutaneous T-cell lymphoma, according to researchers.
The drug elicited partial responses in 33% of patients enrolled in a phase 2 study. Half of the responders had mycosis fungoides (MF), and half had Sézary syndrome (SS).
All responses are ongoing, and a few patients with stable disease remain on treatment, so they may convert to partial responses, according to Youn Kim, MD, of Stanford University School of Medicine in California.
Dr Kim presented this research at the 8th Annual T-cell Lymphoma Forum.
The study was conducted by the Cancer Immunotherapy Trials Network (CITN) and supported by the National Cancer Institute and Merck, the company developing pembrolizumab.
“There’s good rationale for immune checkpoint blockade in [MF and SS],” Dr Kim said. “There’s systemic and local immune impairment in MF and Sézary, and there’s mounting evidence that T-cell immunity is critical for meaningful antitumor response.”
“[T]umor-infiltrating CD8+ T cells [have been] associated with improved survival, and therapies which augment T-cell function are effective in [MF and SS]. PD-1 and PD-L1 are very well expressed in the tissue and blood, [and] there’s good genomic evidence of immune evasion in [MF and SS].”
With all this in mind, Dr Kim and her colleagues conducted their phase 2 trial of pembrolizumab in 24 patients with relapsed or refractory MF/SS. Patients were excluded if they had central nervous system disease, autoimmune disease, immunodeficiency, or had received immunosuppressive therapy within 7 days.
The patients’ median age was 67 (range, 44-85), and most were male (75%). Thirty-eight percent of patients (n=9) had MF, and 62% (n=15) had SS. Twelve percent (n=3) had large-cell transformation.
Most patients had Stage IVA disease (62%, n=15), followed by IIIB (13%, n=3), IIIA (13%, n=3), IIB (8%, n=2), and IB (4%, n=1). The median number of prior systemic therapies was 4 (range, 1-10).
Treatment and response
Patients received pembrolizumab at 2 mg/kg intravenously every 3 weeks and were allowed to continue therapy for up to 2 years. Dr Kim noted that patients could continue treatment even after the initial documentation of progressive disease (PD) due to the possibility of immune-mediated flare reactions.
“So it’s the investigator’s decision to allow treatment beyond the initial PD,” she said. “However, if there’s confirmation of PD, those people will be removed.”
The median follow-up was 21 weeks (range, 7-39). Eight patients responded to treatment (according to global response criteria), all of which were partial responses. Four of the responders had MF, and 4 had SS. Responses occurred across all disease stages except IB.
“The range of prior therapies varied in the responders,” Dr Kim noted. “People think [patients tend to respond to] immunotherapy [if they are only] mildly [pre-]treated, but that was not the case. Heavily treated patients had great responses to pembrolizumab.”
All responses are ongoing, with a median duration of 13+ weeks (range, 3+ to 30+). The median time to response was 11 weeks (range, 8-22).
Dr Kim noted that 1 responder discontinued treatment because of a severe adverse event, but this patient remains in response without having received subsequent treatment.
The median best mSWAT (modified Skin Weighted Assessment Tool) reduction was 16%. Two patients had near-complete responses in the skin, and 2 patients with stable disease in the skin continue to improve.
Eleven of the 15 SS patients had measurable Sézary burden pre-treatment. And 3 of these patients had a greater than 50% reduction in Sézary count after treatment.
Four patients with stable disease are still on treatment. And at 20 weeks, 75% of patients are progression-free, according to Kaplan-Meier estimates.
Adverse events
Drug-related adverse events occurring at least twice included skin eruptions (21%, n=5), anemia (13%, n=3), decrease in white blood cell count (8%, n=2), elevated liver tests (8%, n=2), diarrhea (8%, n=2), fever (8%, n=2), and face edema (8%, n=2).
Grade 3/4 adverse events included skin eruptions (8%, n=2), anemia (8%, n=2), elevated liver tests (4%, n=1), and face edema (4%, n=1). Skin eruptions (of all grades) included exfoliative dermatitis (n=2), immune-mediated skin flare (n=2), and excessive peeling/edema (n=1).
There were no drug-related serious adverse events. The cause of the aforementioned serious adverse event (which prompted the responding patient to discontinue treatment) could not be determined.
There were 4 patients who did not report any adverse events, regardless of attribution.
In closing, Dr Kim said it is important to conduct biomarker correlative studies to understand the tumor escape mechanisms and enrich the response population.
She and her colleagues at CITN are now exploring the use of pembrolizumab in combination therapy. They are considering combining the drug with interferon-gamma, interleukin-12, low-dose total skin radiation, intratumoral ipilimumab, or Toll-like receptor agonists. ![]()
FDA approves drug for patients receiving MEC

Photo by Rhoda Baer
The US Food and Drug Administration (FDA) has approved a supplemental new drug application for single-dose fosaprepitant dimeglumine (Emend) for injection.
The agency approved the substance P/neurokinin-1 (NK1) receptor antagonist for use in combination with other anti-emetic medicines to prevent delayed nausea and vomiting in adults receiving initial and repeat courses of moderately emetogenic chemotherapy (MEC).
This makes fosaprepitant dimeglumine the first intravenous NK1 receptor antagonist approved in the US for patients receiving either highly emetogenic chemotherapy or MEC.
Fosaprepitant dimeglumine has not been studied for the treatment of established nausea and vomiting.
The FDA’s latest approval of fosaprepitant dimeglumine is supported by data from a phase 3 study published in the Annals of Oncology.
Patients receiving MEC were given ondansetron and dexamethasone (n=498) or ondansetron and dexamethasone plus a single intravenous infusion of fosaprepitant dimeglumine (n=502).
The primary endpoint was complete response (CR)—defined as no vomiting and no use of rescue therapy—in the delayed phase of chemotherapy-induced nausea and vomiting, which is 25 to 120 hours after the initiation of chemotherapy.
Secondary endpoints included CR in the overall and acute phases—0 to 120 and 0 to 24 hours after MEC initiation, respectively—and no vomiting in the overall phase.
The fosaprepitant regimen improved CR significantly in the delayed and overall phases but not in the acute phase.
In the delayed phase, the CR rate was 78.9% with the fosaprepitant regimen and 68.5% with the control regimen (P<0.001). In the acute phase, the CR rate was 93.2% and 91.0%, respectively (P=0.184). Overall, the CR rate was 77.1% and 66.9%, respectively (P<0.001).
In the overall phase, the proportion of subjects with no vomiting was 82.7% with the fosaprepitant regimen and 72.9% with the control regimen (P<0.001). The proportion of patients with no significant nausea was 83.2% and 77.9%, respectively (P=0.030).
The most common adverse events reported in the fosaprepitant and control arms, respectively, were fatigue (15% vs 13%), diarrhea (13% vs 11%), neutropenia (8% vs 7%), asthenia (4% vs 3%), anemia (3% vs 2%), peripheral neuropathy (3% vs 2%), leukopenia (2% vs 1%), dyspepsia (2% vs 1%), urinary tract infection (2% vs 1%), and pain in extremity (2% vs 1%).
Fosaprepitant dimeglumine is a product of Merck. For more details on the drug, see the prescribing information. ![]()

Photo by Rhoda Baer
The US Food and Drug Administration (FDA) has approved a supplemental new drug application for single-dose fosaprepitant dimeglumine (Emend) for injection.
The agency approved the substance P/neurokinin-1 (NK1) receptor antagonist for use in combination with other anti-emetic medicines to prevent delayed nausea and vomiting in adults receiving initial and repeat courses of moderately emetogenic chemotherapy (MEC).
This makes fosaprepitant dimeglumine the first intravenous NK1 receptor antagonist approved in the US for patients receiving either highly emetogenic chemotherapy or MEC.
Fosaprepitant dimeglumine has not been studied for the treatment of established nausea and vomiting.
The FDA’s latest approval of fosaprepitant dimeglumine is supported by data from a phase 3 study published in the Annals of Oncology.
Patients receiving MEC were given ondansetron and dexamethasone (n=498) or ondansetron and dexamethasone plus a single intravenous infusion of fosaprepitant dimeglumine (n=502).
The primary endpoint was complete response (CR)—defined as no vomiting and no use of rescue therapy—in the delayed phase of chemotherapy-induced nausea and vomiting, which is 25 to 120 hours after the initiation of chemotherapy.
Secondary endpoints included CR in the overall and acute phases—0 to 120 and 0 to 24 hours after MEC initiation, respectively—and no vomiting in the overall phase.
The fosaprepitant regimen improved CR significantly in the delayed and overall phases but not in the acute phase.
In the delayed phase, the CR rate was 78.9% with the fosaprepitant regimen and 68.5% with the control regimen (P<0.001). In the acute phase, the CR rate was 93.2% and 91.0%, respectively (P=0.184). Overall, the CR rate was 77.1% and 66.9%, respectively (P<0.001).
In the overall phase, the proportion of subjects with no vomiting was 82.7% with the fosaprepitant regimen and 72.9% with the control regimen (P<0.001). The proportion of patients with no significant nausea was 83.2% and 77.9%, respectively (P=0.030).
The most common adverse events reported in the fosaprepitant and control arms, respectively, were fatigue (15% vs 13%), diarrhea (13% vs 11%), neutropenia (8% vs 7%), asthenia (4% vs 3%), anemia (3% vs 2%), peripheral neuropathy (3% vs 2%), leukopenia (2% vs 1%), dyspepsia (2% vs 1%), urinary tract infection (2% vs 1%), and pain in extremity (2% vs 1%).
Fosaprepitant dimeglumine is a product of Merck. For more details on the drug, see the prescribing information. ![]()

Photo by Rhoda Baer
The US Food and Drug Administration (FDA) has approved a supplemental new drug application for single-dose fosaprepitant dimeglumine (Emend) for injection.
The agency approved the substance P/neurokinin-1 (NK1) receptor antagonist for use in combination with other anti-emetic medicines to prevent delayed nausea and vomiting in adults receiving initial and repeat courses of moderately emetogenic chemotherapy (MEC).
This makes fosaprepitant dimeglumine the first intravenous NK1 receptor antagonist approved in the US for patients receiving either highly emetogenic chemotherapy or MEC.
Fosaprepitant dimeglumine has not been studied for the treatment of established nausea and vomiting.
The FDA’s latest approval of fosaprepitant dimeglumine is supported by data from a phase 3 study published in the Annals of Oncology.
Patients receiving MEC were given ondansetron and dexamethasone (n=498) or ondansetron and dexamethasone plus a single intravenous infusion of fosaprepitant dimeglumine (n=502).
The primary endpoint was complete response (CR)—defined as no vomiting and no use of rescue therapy—in the delayed phase of chemotherapy-induced nausea and vomiting, which is 25 to 120 hours after the initiation of chemotherapy.
Secondary endpoints included CR in the overall and acute phases—0 to 120 and 0 to 24 hours after MEC initiation, respectively—and no vomiting in the overall phase.
The fosaprepitant regimen improved CR significantly in the delayed and overall phases but not in the acute phase.
In the delayed phase, the CR rate was 78.9% with the fosaprepitant regimen and 68.5% with the control regimen (P<0.001). In the acute phase, the CR rate was 93.2% and 91.0%, respectively (P=0.184). Overall, the CR rate was 77.1% and 66.9%, respectively (P<0.001).
In the overall phase, the proportion of subjects with no vomiting was 82.7% with the fosaprepitant regimen and 72.9% with the control regimen (P<0.001). The proportion of patients with no significant nausea was 83.2% and 77.9%, respectively (P=0.030).
The most common adverse events reported in the fosaprepitant and control arms, respectively, were fatigue (15% vs 13%), diarrhea (13% vs 11%), neutropenia (8% vs 7%), asthenia (4% vs 3%), anemia (3% vs 2%), peripheral neuropathy (3% vs 2%), leukopenia (2% vs 1%), dyspepsia (2% vs 1%), urinary tract infection (2% vs 1%), and pain in extremity (2% vs 1%).
Fosaprepitant dimeglumine is a product of Merck. For more details on the drug, see the prescribing information.
Mutations may impact response to HDACis in PTCL-NOS

Photo by Larry Young
SAN FRANCISCO—Preclinical research has revealed mutations that may affect the performance of histone deacetylase inhibitors (HDACis) in patients with peripheral T-cell lymphoma-not otherwise specified (PTCL-NOS).
The researchers identified histone-modifying gene mutations in patients with PTCL-NOS and found evidence to suggest these mutations confer shorter survival.
The team also conducted experiments in Jurkat cells showing that certain HDACis could counteract loss-of-function mutations, while others could not.
Meng-Meng Ji, MD, PhD, of the Shanghai Institute of Hematology in China, presented this research at the 8th Annual T-cell Lymphoma Forum.
Dr Ji and her colleagues first performed targeted sequencing in tumor samples from 105 patients newly diagnosed with PTCL-NOS.
The team discovered 62 mutations of “important” lymphoma-associated histone-modifying genes in 31 patients. They found mutations in MLL2 (n=21), TET2 (n=17), EP300 (n=8), CREBBP (n=8), and SETD2 (n=6).
Clinical data revealed a significant difference in overall survival between patients who had these mutations and those who did not (P=0.0038).
Because most of the mutations they identified are loss-of-function mutations, Dr Ji and her colleagues wanted to determine whether HDACis could restore the expression of the mutated genes. So they tested 4 HDACis—valproic acid, vorinostat, romidepsin, and chidamide—in Jurkat cells.
All 4 HDACis upregulated expression of EP300 and CREBBP. However, only romidepsin and chidamide upregulated MLL2, and only valproic acid and vorinostat upregulated SETD2. Dr Ji said there was no obvious change in TET2 expression with any of the HDACis.
The researchers then took a closer look at the EP300, MLL2, and SETD2 mutations. They found that most EP300 mutations were located on the HAT domain. MLL2 mutations could be found in a variety of locations, but some were located on the SET domain. And most SETD2 mutations were located on the SET domain.
Based on the crystal structure of each gene, the team found that EP300 mutations on the HAT domain and both MLL2 mutations and SETD2 mutations located on the SET domain induce loss of function.
So the researchers constructed a mutant for EP300 (p.H1377R), MLL2 (p.V5389M), and SETD2 (p.R1598_) and transfected Jurkat cells with each mutant.
The mutants reduced gene expression significantly when compared to wild-type cells.
Like in the previous experiments, all 4 HDACis could restore the expression of EP300. But only romidepsin and chidamide could restore MLL2 expression, and only valproic acid and vorinostat could restore SETD2 expression.
In addition, all 4 HDACis restored H3K18 hypoacetylation, which was inhibited in Jurkat cells transfected with the EP300 mutant.
Romidepsin and chidamide restored H3K4me3 expression, which was inhibited by the MLL2 mutant. And valproic acid and vorinostat restored H3K36me3 expression, which was inhibited by the SETD2 mutant.
“HDACis targeted differently histone H3 acetylation or methylation modulated by the mutations, suggesting their distinct therapeutic efficiency in PTCL-NOS,” Dr Ji noted.
She said she and her colleagues are continuing this research in samples from patients with PTCL-NOS.

Photo by Larry Young
SAN FRANCISCO—Preclinical research has revealed mutations that may affect the performance of histone deacetylase inhibitors (HDACis) in patients with peripheral T-cell lymphoma-not otherwise specified (PTCL-NOS).
The researchers identified histone-modifying gene mutations in patients with PTCL-NOS and found evidence to suggest these mutations confer shorter survival.
The team also conducted experiments in Jurkat cells showing that certain HDACis could counteract loss-of-function mutations, while others could not.
Meng-Meng Ji, MD, PhD, of the Shanghai Institute of Hematology in China, presented this research at the 8th Annual T-cell Lymphoma Forum.
Dr Ji and her colleagues first performed targeted sequencing in tumor samples from 105 patients newly diagnosed with PTCL-NOS.
The team discovered 62 mutations of “important” lymphoma-associated histone-modifying genes in 31 patients. They found mutations in MLL2 (n=21), TET2 (n=17), EP300 (n=8), CREBBP (n=8), and SETD2 (n=6).
Clinical data revealed a significant difference in overall survival between patients who had these mutations and those who did not (P=0.0038).
Because most of the mutations they identified are loss-of-function mutations, Dr Ji and her colleagues wanted to determine whether HDACis could restore the expression of the mutated genes. So they tested 4 HDACis—valproic acid, vorinostat, romidepsin, and chidamide—in Jurkat cells.
All 4 HDACis upregulated expression of EP300 and CREBBP. However, only romidepsin and chidamide upregulated MLL2, and only valproic acid and vorinostat upregulated SETD2. Dr Ji said there was no obvious change in TET2 expression with any of the HDACis.
The researchers then took a closer look at the EP300, MLL2, and SETD2 mutations. They found that most EP300 mutations were located on the HAT domain. MLL2 mutations could be found in a variety of locations, but some were located on the SET domain. And most SETD2 mutations were located on the SET domain.
Based on the crystal structure of each gene, the team found that EP300 mutations on the HAT domain and both MLL2 mutations and SETD2 mutations located on the SET domain induce loss of function.
So the researchers constructed a mutant for EP300 (p.H1377R), MLL2 (p.V5389M), and SETD2 (p.R1598_) and transfected Jurkat cells with each mutant.
The mutants reduced gene expression significantly when compared to wild-type cells.
Like in the previous experiments, all 4 HDACis could restore the expression of EP300. But only romidepsin and chidamide could restore MLL2 expression, and only valproic acid and vorinostat could restore SETD2 expression.
In addition, all 4 HDACis restored H3K18 hypoacetylation, which was inhibited in Jurkat cells transfected with the EP300 mutant.
Romidepsin and chidamide restored H3K4me3 expression, which was inhibited by the MLL2 mutant. And valproic acid and vorinostat restored H3K36me3 expression, which was inhibited by the SETD2 mutant.
“HDACis targeted differently histone H3 acetylation or methylation modulated by the mutations, suggesting their distinct therapeutic efficiency in PTCL-NOS,” Dr Ji noted.
She said she and her colleagues are continuing this research in samples from patients with PTCL-NOS.

Photo by Larry Young
SAN FRANCISCO—Preclinical research has revealed mutations that may affect the performance of histone deacetylase inhibitors (HDACis) in patients with peripheral T-cell lymphoma-not otherwise specified (PTCL-NOS).
The researchers identified histone-modifying gene mutations in patients with PTCL-NOS and found evidence to suggest these mutations confer shorter survival.
The team also conducted experiments in Jurkat cells showing that certain HDACis could counteract loss-of-function mutations, while others could not.
Meng-Meng Ji, MD, PhD, of the Shanghai Institute of Hematology in China, presented this research at the 8th Annual T-cell Lymphoma Forum.
Dr Ji and her colleagues first performed targeted sequencing in tumor samples from 105 patients newly diagnosed with PTCL-NOS.
The team discovered 62 mutations of “important” lymphoma-associated histone-modifying genes in 31 patients. They found mutations in MLL2 (n=21), TET2 (n=17), EP300 (n=8), CREBBP (n=8), and SETD2 (n=6).
Clinical data revealed a significant difference in overall survival between patients who had these mutations and those who did not (P=0.0038).
Because most of the mutations they identified are loss-of-function mutations, Dr Ji and her colleagues wanted to determine whether HDACis could restore the expression of the mutated genes. So they tested 4 HDACis—valproic acid, vorinostat, romidepsin, and chidamide—in Jurkat cells.
All 4 HDACis upregulated expression of EP300 and CREBBP. However, only romidepsin and chidamide upregulated MLL2, and only valproic acid and vorinostat upregulated SETD2. Dr Ji said there was no obvious change in TET2 expression with any of the HDACis.
The researchers then took a closer look at the EP300, MLL2, and SETD2 mutations. They found that most EP300 mutations were located on the HAT domain. MLL2 mutations could be found in a variety of locations, but some were located on the SET domain. And most SETD2 mutations were located on the SET domain.
Based on the crystal structure of each gene, the team found that EP300 mutations on the HAT domain and both MLL2 mutations and SETD2 mutations located on the SET domain induce loss of function.
So the researchers constructed a mutant for EP300 (p.H1377R), MLL2 (p.V5389M), and SETD2 (p.R1598_) and transfected Jurkat cells with each mutant.
The mutants reduced gene expression significantly when compared to wild-type cells.
Like in the previous experiments, all 4 HDACis could restore the expression of EP300. But only romidepsin and chidamide could restore MLL2 expression, and only valproic acid and vorinostat could restore SETD2 expression.
In addition, all 4 HDACis restored H3K18 hypoacetylation, which was inhibited in Jurkat cells transfected with the EP300 mutant.
Romidepsin and chidamide restored H3K4me3 expression, which was inhibited by the MLL2 mutant. And valproic acid and vorinostat restored H3K36me3 expression, which was inhibited by the SETD2 mutant.
“HDACis targeted differently histone H3 acetylation or methylation modulated by the mutations, suggesting their distinct therapeutic efficiency in PTCL-NOS,” Dr Ji noted.
She said she and her colleagues are continuing this research in samples from patients with PTCL-NOS.
Lenalidomide shows promise for treating ATLL

Photo by Larry Young
SAN FRANCISCO—Results of a phase 2 trial suggest lenalidomide may be a treatment option for patients with relapsed adult T-cell leukemia-lymphoma (ATLL).
Lenalidomide produced a 42% overall response rate (ORR) in this trial, and patients had a “favorable” median overall survival, according to Kisato Nosaka, MD, PhD, of the National Center for Global Health and Medicine in Tokyo, Japan.
He noted, however, that the survival data are still immature and may have been confounded by subsequent therapies.
Dr Nosaka presented these results at the 8th Annual T-cell Lymphoma Forum. The study was sponsored by Celgene K.K.
The trial included 26 Japanese patients with relapsed ATLL. Fifty-eight percent of patients had the acute subtype, 27% had the lymphoma subtype, and 15% had the unfavorable chronic subtype.
The patients’ median age was 68.5 (range, 53-81), and 65% of patients were 65 or older. Fifty percent of patients had an ECOG performance status of 0, 35% had a status of 1, and 15% had a status of 2.
Most patients had a low-risk simplified ATL-PI score (65%), but 35% had an intermediate-risk score. Fifteen percent had bone marrow involvement.
The median number of prior treatment regimens was 2 (range, 1-4). Forty-two percent of patients had prior mogamulizumab, and 35% had received LSG15 or modified LSG15.
Patients received lenalidomide at 25 mg per day, given continuously until disease progression or intolerability.
Safety
The median duration of treatment was 3.7 months (range, 0.4 to 18.3 months). There were no deaths during treatment or for 28 days after.
Nine patients (35%) experienced serious adverse events (AEs), but only 1 serious AE occurred in more than 1 patient. Two patients had serious thrombocytopenia.
The most frequent AEs were thrombocytopenia (77%), neutropenia (73%), lymphopenia (69%), and increased C-reactive protein (42%). The most frequent grade 3 or higher AEs were neutropenia (65%), leukopenia (38%), and lymphopenia (38%).
Response and survival
The median follow-up was 3.9 months. The ORR was 42%, including 5 complete responses/unconfirmed complete responses and 6 partial responses. Eight patients (31%) had stable disease, and 7 (27%) progressed.
Dr Nosaka noted that responses occurred in all disease subtypes and at all disease sites. The ORR was 33% (5/15) for the acute subtype, 50% (2/4) for the unfavorable chronic subtype, and 57% (4/7) for the lymphoma subtype.
The ORR was 31% (5/16) at the target lesion, 60% (6/10) in the peripheral blood, and 75% (6/8) for PGA (Physician’s Global Assessment of Clinical Condition, used to assess skin lesions).
Dr Nosaka also pointed out that the ORR was higher in patients who did not receive prior mogamulizumab (60%) than in patients who did (18%). However, he said the number of patients was too small for a definitive conclusion to be reached.
Similarly, the ORR was higher for patients in the low-risk simplified ATL-PI risk group than for those in the intermediate-risk group—53% and 22%, respectively.
Among the 11 responders, the median duration of response was not reached (range, 0.5 months to not reached). The mean duration of response was 5.2 months (range, 0 to 16.6 months).
The median progression-free survival was 3.8 months (range, 1.9 months to not reached). The median overall survival was 20.3 months (range, 9.1 months to not reached).
The overall survival was longer for patients in the low-risk ATL-PI group than the intermediate-risk group—not reached and 10.1 months, respectively (P=0.03).
In closing, Dr Nosaka said these results support lenalidomide as a possible treatment option for patients with relapsed/recurrent ATLL.

Photo by Larry Young
SAN FRANCISCO—Results of a phase 2 trial suggest lenalidomide may be a treatment option for patients with relapsed adult T-cell leukemia-lymphoma (ATLL).
Lenalidomide produced a 42% overall response rate (ORR) in this trial, and patients had a “favorable” median overall survival, according to Kisato Nosaka, MD, PhD, of the National Center for Global Health and Medicine in Tokyo, Japan.
He noted, however, that the survival data are still immature and may have been confounded by subsequent therapies.
Dr Nosaka presented these results at the 8th Annual T-cell Lymphoma Forum. The study was sponsored by Celgene K.K.
The trial included 26 Japanese patients with relapsed ATLL. Fifty-eight percent of patients had the acute subtype, 27% had the lymphoma subtype, and 15% had the unfavorable chronic subtype.
The patients’ median age was 68.5 (range, 53-81), and 65% of patients were 65 or older. Fifty percent of patients had an ECOG performance status of 0, 35% had a status of 1, and 15% had a status of 2.
Most patients had a low-risk simplified ATL-PI score (65%), but 35% had an intermediate-risk score. Fifteen percent had bone marrow involvement.
The median number of prior treatment regimens was 2 (range, 1-4). Forty-two percent of patients had prior mogamulizumab, and 35% had received LSG15 or modified LSG15.
Patients received lenalidomide at 25 mg per day, given continuously until disease progression or intolerability.
Safety
The median duration of treatment was 3.7 months (range, 0.4 to 18.3 months). There were no deaths during treatment or for 28 days after.
Nine patients (35%) experienced serious adverse events (AEs), but only 1 serious AE occurred in more than 1 patient. Two patients had serious thrombocytopenia.
The most frequent AEs were thrombocytopenia (77%), neutropenia (73%), lymphopenia (69%), and increased C-reactive protein (42%). The most frequent grade 3 or higher AEs were neutropenia (65%), leukopenia (38%), and lymphopenia (38%).
Response and survival
The median follow-up was 3.9 months. The ORR was 42%, including 5 complete responses/unconfirmed complete responses and 6 partial responses. Eight patients (31%) had stable disease, and 7 (27%) progressed.
Dr Nosaka noted that responses occurred in all disease subtypes and at all disease sites. The ORR was 33% (5/15) for the acute subtype, 50% (2/4) for the unfavorable chronic subtype, and 57% (4/7) for the lymphoma subtype.
The ORR was 31% (5/16) at the target lesion, 60% (6/10) in the peripheral blood, and 75% (6/8) for PGA (Physician’s Global Assessment of Clinical Condition, used to assess skin lesions).
Dr Nosaka also pointed out that the ORR was higher in patients who did not receive prior mogamulizumab (60%) than in patients who did (18%). However, he said the number of patients was too small for a definitive conclusion to be reached.
Similarly, the ORR was higher for patients in the low-risk simplified ATL-PI risk group than for those in the intermediate-risk group—53% and 22%, respectively.
Among the 11 responders, the median duration of response was not reached (range, 0.5 months to not reached). The mean duration of response was 5.2 months (range, 0 to 16.6 months).
The median progression-free survival was 3.8 months (range, 1.9 months to not reached). The median overall survival was 20.3 months (range, 9.1 months to not reached).
The overall survival was longer for patients in the low-risk ATL-PI group than the intermediate-risk group—not reached and 10.1 months, respectively (P=0.03).
In closing, Dr Nosaka said these results support lenalidomide as a possible treatment option for patients with relapsed/recurrent ATLL.

Photo by Larry Young
SAN FRANCISCO—Results of a phase 2 trial suggest lenalidomide may be a treatment option for patients with relapsed adult T-cell leukemia-lymphoma (ATLL).
Lenalidomide produced a 42% overall response rate (ORR) in this trial, and patients had a “favorable” median overall survival, according to Kisato Nosaka, MD, PhD, of the National Center for Global Health and Medicine in Tokyo, Japan.
He noted, however, that the survival data are still immature and may have been confounded by subsequent therapies.
Dr Nosaka presented these results at the 8th Annual T-cell Lymphoma Forum. The study was sponsored by Celgene K.K.
The trial included 26 Japanese patients with relapsed ATLL. Fifty-eight percent of patients had the acute subtype, 27% had the lymphoma subtype, and 15% had the unfavorable chronic subtype.
The patients’ median age was 68.5 (range, 53-81), and 65% of patients were 65 or older. Fifty percent of patients had an ECOG performance status of 0, 35% had a status of 1, and 15% had a status of 2.
Most patients had a low-risk simplified ATL-PI score (65%), but 35% had an intermediate-risk score. Fifteen percent had bone marrow involvement.
The median number of prior treatment regimens was 2 (range, 1-4). Forty-two percent of patients had prior mogamulizumab, and 35% had received LSG15 or modified LSG15.
Patients received lenalidomide at 25 mg per day, given continuously until disease progression or intolerability.
Safety
The median duration of treatment was 3.7 months (range, 0.4 to 18.3 months). There were no deaths during treatment or for 28 days after.
Nine patients (35%) experienced serious adverse events (AEs), but only 1 serious AE occurred in more than 1 patient. Two patients had serious thrombocytopenia.
The most frequent AEs were thrombocytopenia (77%), neutropenia (73%), lymphopenia (69%), and increased C-reactive protein (42%). The most frequent grade 3 or higher AEs were neutropenia (65%), leukopenia (38%), and lymphopenia (38%).
Response and survival
The median follow-up was 3.9 months. The ORR was 42%, including 5 complete responses/unconfirmed complete responses and 6 partial responses. Eight patients (31%) had stable disease, and 7 (27%) progressed.
Dr Nosaka noted that responses occurred in all disease subtypes and at all disease sites. The ORR was 33% (5/15) for the acute subtype, 50% (2/4) for the unfavorable chronic subtype, and 57% (4/7) for the lymphoma subtype.
The ORR was 31% (5/16) at the target lesion, 60% (6/10) in the peripheral blood, and 75% (6/8) for PGA (Physician’s Global Assessment of Clinical Condition, used to assess skin lesions).
Dr Nosaka also pointed out that the ORR was higher in patients who did not receive prior mogamulizumab (60%) than in patients who did (18%). However, he said the number of patients was too small for a definitive conclusion to be reached.
Similarly, the ORR was higher for patients in the low-risk simplified ATL-PI risk group than for those in the intermediate-risk group—53% and 22%, respectively.
Among the 11 responders, the median duration of response was not reached (range, 0.5 months to not reached). The mean duration of response was 5.2 months (range, 0 to 16.6 months).
The median progression-free survival was 3.8 months (range, 1.9 months to not reached). The median overall survival was 20.3 months (range, 9.1 months to not reached).
The overall survival was longer for patients in the low-risk ATL-PI group than the intermediate-risk group—not reached and 10.1 months, respectively (P=0.03).
In closing, Dr Nosaka said these results support lenalidomide as a possible treatment option for patients with relapsed/recurrent ATLL.