User login
New mechanisms, therapies for acne considered
SEATTLE – It used to be thought that acne begins with microcomedones, which go on to develop either inflammatory lesions or noninflammatory lesions, but more recent evidence has changed that perception, according to Linda Stein Gold, MD, director of dermatology research at Henry Ford Hospital, Detroit.
Dr. Stein Gold said at the annual Coastal Dermatology Symposium. “All acne is inflammation acne,” and inflammation also persists, she added. Biopsies of scarred lesions, once considered postinflammatory, also have revealed persistent inflammation, she noted.
One study found that persistent scars can evolve from closed comedones, papules, and pustules, but the most common was a papule that turned into a postinflammatory lesion (J Drugs Dermatol 2017 Jun 1;16[6]:566-72). “So when patients come in and they have these red spots on their face, it’s not over. There’s still time to be aggressive because those inflammatory lesions are more likely to lead to scars than anything else,” Dr. Stein Gold said. “And we also know that papules that develop into scars do so because they’re there for a longer period of time. Those that develop scars are present about 10.5 days, compared with 6.6 days for those that don’t develop into scars.”
She went on to review some of the new treatments for acne that can be brought to bear in such cases. These include developments with topical retinoids that are aimed at improving delivery and reducing skin irritation.
A new topical retinoid, trifarotene cream, 0.005%, showed efficacy and tolerability for both the face and trunk in a recent phase 3 trial of patients with moderate facial and truncal acne and was recently approved for patients aged 9 years and older. In the study, about 30%-40% of people aged 9 years and older treated with once-daily trifarotene cream (Aklief) achieved clear or almost-clear status of the face at 12 weeks, vs. about 20% and 26%, of those on the vehicle cream (J Am Acad Dermatol. 2019 Jun;80[6]:1691-9).
The drug can also treat papules and pustules, nearly as well as it treats blackheads and whiteheads, according to Dr. Stein Gold. Like other retinoids, it produces some redness and scaling and rather than letting these adverse events discourage patients, she leans in. “I tell patients they’re going to have some sloughing of the skin the first 2 weeks. I tell them that people pay money for that. It’s called a chemical peel,” said Dr. Stein Gold, noting that patients respond well to this information.
If patients find the treatments too irritating, she advises them to avoid applying it to wet skin. They can also apply it every other night, or even less frequently, and then work up to more frequent use, she said at the meeting, jointly presented by the University of Louisville and Global Academy for Medical Education.
Tazarotene is another topical retinoid that can be very irritating. A new lotion formulation of tazarotene 0.045% contains a lower dose than the 0.1% typically used in creams, and has similar efficacy but reduced irritation, Dr. Stein Gold said. In August, the manufacturer submitted an application for approval with the Food and Drug Administration for treatment of acne.
Dr. Stein Gold also talked about using retinoids to minimize scarring, referring to a study of patients with moderate and severe facial acne, and atrophic acne scars, comparing adapalene 0.3% plus benzoyl peroxide 2.5% gel on one side of the face and vehicle on the other side of the face for 24 weeks, followed by application of the active treatment to both sides of the face for 24 weeks. Treatment was associated with a reduction of atrophic acne scars at 24 weeks, which was maintained for up to 48 weeks (Am J Clin Dermatol. 2019 Oct[5];20:725-32).
“We can now say to patients, ‘Not only can I help you with your acne, but I can potentially even improve your atrophic scarring,’ ” she said.
Finally, she discussed clascoterone, a novel androgen receptor antagonist, which inhibits sebum production and prevents colonization by Cutibacterium acnes (formerly called Propionibacterium acnes) and subsequent inflammation. “It does a lot of good things in terms of the pathogenesis of acne, but more importantly, it is one of the first drugs that topically has been shown to decrease the production of sebum,” Dr. Stein Gold said. A 1% cream formulation is being studied for acne.
Dr. Stein Gold is a consultant, investigator, and/or speaker for Galderma, Ortho Derm, Sol Gel, Foamix, Cassiopea, and Almirall.
This publication and Global Academy for Medical Education are owned by the same parent company.
SEATTLE – It used to be thought that acne begins with microcomedones, which go on to develop either inflammatory lesions or noninflammatory lesions, but more recent evidence has changed that perception, according to Linda Stein Gold, MD, director of dermatology research at Henry Ford Hospital, Detroit.
Dr. Stein Gold said at the annual Coastal Dermatology Symposium. “All acne is inflammation acne,” and inflammation also persists, she added. Biopsies of scarred lesions, once considered postinflammatory, also have revealed persistent inflammation, she noted.
One study found that persistent scars can evolve from closed comedones, papules, and pustules, but the most common was a papule that turned into a postinflammatory lesion (J Drugs Dermatol 2017 Jun 1;16[6]:566-72). “So when patients come in and they have these red spots on their face, it’s not over. There’s still time to be aggressive because those inflammatory lesions are more likely to lead to scars than anything else,” Dr. Stein Gold said. “And we also know that papules that develop into scars do so because they’re there for a longer period of time. Those that develop scars are present about 10.5 days, compared with 6.6 days for those that don’t develop into scars.”
She went on to review some of the new treatments for acne that can be brought to bear in such cases. These include developments with topical retinoids that are aimed at improving delivery and reducing skin irritation.
A new topical retinoid, trifarotene cream, 0.005%, showed efficacy and tolerability for both the face and trunk in a recent phase 3 trial of patients with moderate facial and truncal acne and was recently approved for patients aged 9 years and older. In the study, about 30%-40% of people aged 9 years and older treated with once-daily trifarotene cream (Aklief) achieved clear or almost-clear status of the face at 12 weeks, vs. about 20% and 26%, of those on the vehicle cream (J Am Acad Dermatol. 2019 Jun;80[6]:1691-9).
The drug can also treat papules and pustules, nearly as well as it treats blackheads and whiteheads, according to Dr. Stein Gold. Like other retinoids, it produces some redness and scaling and rather than letting these adverse events discourage patients, she leans in. “I tell patients they’re going to have some sloughing of the skin the first 2 weeks. I tell them that people pay money for that. It’s called a chemical peel,” said Dr. Stein Gold, noting that patients respond well to this information.
If patients find the treatments too irritating, she advises them to avoid applying it to wet skin. They can also apply it every other night, or even less frequently, and then work up to more frequent use, she said at the meeting, jointly presented by the University of Louisville and Global Academy for Medical Education.
Tazarotene is another topical retinoid that can be very irritating. A new lotion formulation of tazarotene 0.045% contains a lower dose than the 0.1% typically used in creams, and has similar efficacy but reduced irritation, Dr. Stein Gold said. In August, the manufacturer submitted an application for approval with the Food and Drug Administration for treatment of acne.
Dr. Stein Gold also talked about using retinoids to minimize scarring, referring to a study of patients with moderate and severe facial acne, and atrophic acne scars, comparing adapalene 0.3% plus benzoyl peroxide 2.5% gel on one side of the face and vehicle on the other side of the face for 24 weeks, followed by application of the active treatment to both sides of the face for 24 weeks. Treatment was associated with a reduction of atrophic acne scars at 24 weeks, which was maintained for up to 48 weeks (Am J Clin Dermatol. 2019 Oct[5];20:725-32).
“We can now say to patients, ‘Not only can I help you with your acne, but I can potentially even improve your atrophic scarring,’ ” she said.
Finally, she discussed clascoterone, a novel androgen receptor antagonist, which inhibits sebum production and prevents colonization by Cutibacterium acnes (formerly called Propionibacterium acnes) and subsequent inflammation. “It does a lot of good things in terms of the pathogenesis of acne, but more importantly, it is one of the first drugs that topically has been shown to decrease the production of sebum,” Dr. Stein Gold said. A 1% cream formulation is being studied for acne.
Dr. Stein Gold is a consultant, investigator, and/or speaker for Galderma, Ortho Derm, Sol Gel, Foamix, Cassiopea, and Almirall.
This publication and Global Academy for Medical Education are owned by the same parent company.
SEATTLE – It used to be thought that acne begins with microcomedones, which go on to develop either inflammatory lesions or noninflammatory lesions, but more recent evidence has changed that perception, according to Linda Stein Gold, MD, director of dermatology research at Henry Ford Hospital, Detroit.
Dr. Stein Gold said at the annual Coastal Dermatology Symposium. “All acne is inflammation acne,” and inflammation also persists, she added. Biopsies of scarred lesions, once considered postinflammatory, also have revealed persistent inflammation, she noted.
One study found that persistent scars can evolve from closed comedones, papules, and pustules, but the most common was a papule that turned into a postinflammatory lesion (J Drugs Dermatol 2017 Jun 1;16[6]:566-72). “So when patients come in and they have these red spots on their face, it’s not over. There’s still time to be aggressive because those inflammatory lesions are more likely to lead to scars than anything else,” Dr. Stein Gold said. “And we also know that papules that develop into scars do so because they’re there for a longer period of time. Those that develop scars are present about 10.5 days, compared with 6.6 days for those that don’t develop into scars.”
She went on to review some of the new treatments for acne that can be brought to bear in such cases. These include developments with topical retinoids that are aimed at improving delivery and reducing skin irritation.
A new topical retinoid, trifarotene cream, 0.005%, showed efficacy and tolerability for both the face and trunk in a recent phase 3 trial of patients with moderate facial and truncal acne and was recently approved for patients aged 9 years and older. In the study, about 30%-40% of people aged 9 years and older treated with once-daily trifarotene cream (Aklief) achieved clear or almost-clear status of the face at 12 weeks, vs. about 20% and 26%, of those on the vehicle cream (J Am Acad Dermatol. 2019 Jun;80[6]:1691-9).
The drug can also treat papules and pustules, nearly as well as it treats blackheads and whiteheads, according to Dr. Stein Gold. Like other retinoids, it produces some redness and scaling and rather than letting these adverse events discourage patients, she leans in. “I tell patients they’re going to have some sloughing of the skin the first 2 weeks. I tell them that people pay money for that. It’s called a chemical peel,” said Dr. Stein Gold, noting that patients respond well to this information.
If patients find the treatments too irritating, she advises them to avoid applying it to wet skin. They can also apply it every other night, or even less frequently, and then work up to more frequent use, she said at the meeting, jointly presented by the University of Louisville and Global Academy for Medical Education.
Tazarotene is another topical retinoid that can be very irritating. A new lotion formulation of tazarotene 0.045% contains a lower dose than the 0.1% typically used in creams, and has similar efficacy but reduced irritation, Dr. Stein Gold said. In August, the manufacturer submitted an application for approval with the Food and Drug Administration for treatment of acne.
Dr. Stein Gold also talked about using retinoids to minimize scarring, referring to a study of patients with moderate and severe facial acne, and atrophic acne scars, comparing adapalene 0.3% plus benzoyl peroxide 2.5% gel on one side of the face and vehicle on the other side of the face for 24 weeks, followed by application of the active treatment to both sides of the face for 24 weeks. Treatment was associated with a reduction of atrophic acne scars at 24 weeks, which was maintained for up to 48 weeks (Am J Clin Dermatol. 2019 Oct[5];20:725-32).
“We can now say to patients, ‘Not only can I help you with your acne, but I can potentially even improve your atrophic scarring,’ ” she said.
Finally, she discussed clascoterone, a novel androgen receptor antagonist, which inhibits sebum production and prevents colonization by Cutibacterium acnes (formerly called Propionibacterium acnes) and subsequent inflammation. “It does a lot of good things in terms of the pathogenesis of acne, but more importantly, it is one of the first drugs that topically has been shown to decrease the production of sebum,” Dr. Stein Gold said. A 1% cream formulation is being studied for acne.
Dr. Stein Gold is a consultant, investigator, and/or speaker for Galderma, Ortho Derm, Sol Gel, Foamix, Cassiopea, and Almirall.
This publication and Global Academy for Medical Education are owned by the same parent company.
EXPERT ANALYSIS FROM COASTAL DERM
Adjunctive therapy is among the roles for topical agents in psoriasis
SEATTLE – is not dead,” Linda Stein Gold, MD, said at the annual Coastal Dermatology Symposium.
“We have to remember when we think back to our practice, how many topical prescriptions do we write, compared to preventive prescriptions? Probably most are topical,” said Dr. Stein Gold, director of dermatology clinical research at the Henry Ford Hospital Center, Detroit.
Topical agents have a place when a patient is doing well on treatment with a biologic but is not responding completely, she noted. One open-label, single-arm study looked at adjunctive calcipotriene 0.005%/betamethasone dipropionate 0.064% (Enstilar) foam, applied once daily for 4 week, then twice a week on consecutive days for 12 weeks in 25 patients with psoriasis who had a mean body surface area (BSA) of less than 5% but significant remaining disease despite treatment with biologics.
At week 4, 76% achieved a BSA of 1% or less and Physician’s Global Assessment score of 1 or less at week 4, as did 68% at week 16. This was compared with 12% and 4%, respectively (J Drugs Dermatol. 2018 Aug 1;17[8]:845-50). “They found that a good potent topical on top of a biologic does really well. That can really kick up the last part of the efficacy to get the patients almost to clear,” she observed.
At the meeting, jointly presented by the University of Louisville and Global Academy for Medical Education, Dr. Stein Gold also discussed tazarotene, a topical retinoid approved by the Food and Drug Administration for treating psoriasis and is available as a 0.1% and 0.05% cream and gel. About 10%-30% of patients experience side effects with tazarotene, such as pruritus, stinging, and burning. Topical corticosteroids can help, which prompted development of a combined product, she noted.
She referred to a phase 2 study of patients with moderate to severe plaque psoriasis, which compared the fixed combination lotion formulation of tazarotene plus halobetasol propionate to the two components alone. The investigators found almost a 9% rate of treatment success with tazarotene alone, versus about 23% with halobetasol propionate alone and about 43% with the combined product. The combined individual effect of the two drugs was about 32%, so the 43% efficacy of the combined product had an absolute synergistic effect of about 11%, Dr. Stein Gold pointed out.
Two phase 3 trials of adults with moderate to severe psoriasis supported the phase 2 results of the combined lotion formulation (halobetasol 0.01% with tazarotene 0.045%), said Dr. Stein Gold, the first author (J Am Acad Dermatol. 2018 Aug;79[2]:287-93). Treatment success was defined as at least a 2-grade Investigator’s Global Assessment score and improvement from baseline and a score of “clear” or “almost clear.” In one of the studies, 36% of those on the combination versus 7% of those on the vehicle met this endpoint at week 8, as did 45% versus 13%, respectively, in the second study (P less than .001 for both studies).
Patients also had less itching, drying, and stinging than typically seen with tazarotene alone, Dr. Stein Gold said. In the studies, contact dermatitis was the most common side effect associated with treatment, reported in 6.3%
Dr. Stein Gold has received research support from Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan, and Foamix. She has been a consultant for Sol-gel, Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan, Foamix, Promis, Anacor, and Medimetriks. She has been on the speakers bureau of Galderma, Leo, Valeant, Novartis, Celgene, and Allergan. She has been a member of scientific advisory boards for Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan, Foamix, and Promius.
This publication and Global Academy for Medical Education are owned by the same parent company.
SEATTLE – is not dead,” Linda Stein Gold, MD, said at the annual Coastal Dermatology Symposium.
“We have to remember when we think back to our practice, how many topical prescriptions do we write, compared to preventive prescriptions? Probably most are topical,” said Dr. Stein Gold, director of dermatology clinical research at the Henry Ford Hospital Center, Detroit.
Topical agents have a place when a patient is doing well on treatment with a biologic but is not responding completely, she noted. One open-label, single-arm study looked at adjunctive calcipotriene 0.005%/betamethasone dipropionate 0.064% (Enstilar) foam, applied once daily for 4 week, then twice a week on consecutive days for 12 weeks in 25 patients with psoriasis who had a mean body surface area (BSA) of less than 5% but significant remaining disease despite treatment with biologics.
At week 4, 76% achieved a BSA of 1% or less and Physician’s Global Assessment score of 1 or less at week 4, as did 68% at week 16. This was compared with 12% and 4%, respectively (J Drugs Dermatol. 2018 Aug 1;17[8]:845-50). “They found that a good potent topical on top of a biologic does really well. That can really kick up the last part of the efficacy to get the patients almost to clear,” she observed.
At the meeting, jointly presented by the University of Louisville and Global Academy for Medical Education, Dr. Stein Gold also discussed tazarotene, a topical retinoid approved by the Food and Drug Administration for treating psoriasis and is available as a 0.1% and 0.05% cream and gel. About 10%-30% of patients experience side effects with tazarotene, such as pruritus, stinging, and burning. Topical corticosteroids can help, which prompted development of a combined product, she noted.
She referred to a phase 2 study of patients with moderate to severe plaque psoriasis, which compared the fixed combination lotion formulation of tazarotene plus halobetasol propionate to the two components alone. The investigators found almost a 9% rate of treatment success with tazarotene alone, versus about 23% with halobetasol propionate alone and about 43% with the combined product. The combined individual effect of the two drugs was about 32%, so the 43% efficacy of the combined product had an absolute synergistic effect of about 11%, Dr. Stein Gold pointed out.
Two phase 3 trials of adults with moderate to severe psoriasis supported the phase 2 results of the combined lotion formulation (halobetasol 0.01% with tazarotene 0.045%), said Dr. Stein Gold, the first author (J Am Acad Dermatol. 2018 Aug;79[2]:287-93). Treatment success was defined as at least a 2-grade Investigator’s Global Assessment score and improvement from baseline and a score of “clear” or “almost clear.” In one of the studies, 36% of those on the combination versus 7% of those on the vehicle met this endpoint at week 8, as did 45% versus 13%, respectively, in the second study (P less than .001 for both studies).
Patients also had less itching, drying, and stinging than typically seen with tazarotene alone, Dr. Stein Gold said. In the studies, contact dermatitis was the most common side effect associated with treatment, reported in 6.3%
Dr. Stein Gold has received research support from Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan, and Foamix. She has been a consultant for Sol-gel, Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan, Foamix, Promis, Anacor, and Medimetriks. She has been on the speakers bureau of Galderma, Leo, Valeant, Novartis, Celgene, and Allergan. She has been a member of scientific advisory boards for Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan, Foamix, and Promius.
This publication and Global Academy for Medical Education are owned by the same parent company.
SEATTLE – is not dead,” Linda Stein Gold, MD, said at the annual Coastal Dermatology Symposium.
“We have to remember when we think back to our practice, how many topical prescriptions do we write, compared to preventive prescriptions? Probably most are topical,” said Dr. Stein Gold, director of dermatology clinical research at the Henry Ford Hospital Center, Detroit.
Topical agents have a place when a patient is doing well on treatment with a biologic but is not responding completely, she noted. One open-label, single-arm study looked at adjunctive calcipotriene 0.005%/betamethasone dipropionate 0.064% (Enstilar) foam, applied once daily for 4 week, then twice a week on consecutive days for 12 weeks in 25 patients with psoriasis who had a mean body surface area (BSA) of less than 5% but significant remaining disease despite treatment with biologics.
At week 4, 76% achieved a BSA of 1% or less and Physician’s Global Assessment score of 1 or less at week 4, as did 68% at week 16. This was compared with 12% and 4%, respectively (J Drugs Dermatol. 2018 Aug 1;17[8]:845-50). “They found that a good potent topical on top of a biologic does really well. That can really kick up the last part of the efficacy to get the patients almost to clear,” she observed.
At the meeting, jointly presented by the University of Louisville and Global Academy for Medical Education, Dr. Stein Gold also discussed tazarotene, a topical retinoid approved by the Food and Drug Administration for treating psoriasis and is available as a 0.1% and 0.05% cream and gel. About 10%-30% of patients experience side effects with tazarotene, such as pruritus, stinging, and burning. Topical corticosteroids can help, which prompted development of a combined product, she noted.
She referred to a phase 2 study of patients with moderate to severe plaque psoriasis, which compared the fixed combination lotion formulation of tazarotene plus halobetasol propionate to the two components alone. The investigators found almost a 9% rate of treatment success with tazarotene alone, versus about 23% with halobetasol propionate alone and about 43% with the combined product. The combined individual effect of the two drugs was about 32%, so the 43% efficacy of the combined product had an absolute synergistic effect of about 11%, Dr. Stein Gold pointed out.
Two phase 3 trials of adults with moderate to severe psoriasis supported the phase 2 results of the combined lotion formulation (halobetasol 0.01% with tazarotene 0.045%), said Dr. Stein Gold, the first author (J Am Acad Dermatol. 2018 Aug;79[2]:287-93). Treatment success was defined as at least a 2-grade Investigator’s Global Assessment score and improvement from baseline and a score of “clear” or “almost clear.” In one of the studies, 36% of those on the combination versus 7% of those on the vehicle met this endpoint at week 8, as did 45% versus 13%, respectively, in the second study (P less than .001 for both studies).
Patients also had less itching, drying, and stinging than typically seen with tazarotene alone, Dr. Stein Gold said. In the studies, contact dermatitis was the most common side effect associated with treatment, reported in 6.3%
Dr. Stein Gold has received research support from Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan, and Foamix. She has been a consultant for Sol-gel, Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan, Foamix, Promis, Anacor, and Medimetriks. She has been on the speakers bureau of Galderma, Leo, Valeant, Novartis, Celgene, and Allergan. She has been a member of scientific advisory boards for Galderma, Leo, Novan, Valeant, Dermira, Novartis, Celgene, Allergan, Foamix, and Promius.
This publication and Global Academy for Medical Education are owned by the same parent company.
EXPERT ANALYSIS FROM COASTAL DERM
Once-daily oral JAK inhibitor for atopic dermatitis effective in phase 3 study
MADRID – (AD) on the strength of its knockout performance in the pivotal phase 3 JADE MONO-1 trial, presented at the annual congress of the European Academy of Dermatology and Venereology.
“The IGA [Investigator Global Assessment] and EASI [Eczema Area and Severity Index]-75 responses for both doses of abrocitinib were significantly greater than placebo as early as week 2 and continued to increase until week 12 with no plateau. This study stopped at week 12. I would have liked to see what happened at 16 weeks or further. I don’t know where this is going to end up maxing out,” commented principal investigator Eric Simpson, MD, professor of dermatology at Oregon Health & Science University, Portland.
JADE MONO-1 (JAK1 Atopic Dermatitis Efficacy and Safety Monotherapy–1) was a 12-week, double-blind, multicenter study which included 387 adolescents and adults with moderate or severe AD randomized 2:2:1 to abrocitinib at 200 mg or 100 mg once daily, or to placebo. Overall, this was a fairly severely affected population, with a mean baseline EASI (Eczema Area and Severity Index) score of about 30, a peak pruritus numeric rating scale score of 7 out of 10, and a mean Dermatology Quality Life Index score greater than 14. Nonetheless, this was a rigorous abrocitinib monotherapy trial, as participants were not allowed to take even a single dose of topical corticosteroids.
The coprimary endpoints in JADE MONO-1 were achievement of an IGA score of 0 or 1 – that is, clear or almost clear – at week 12 plus at least a 2-point improvement, compared with baseline on the 0-4 scale, and an EASI-75 response as defined by a 75% or greater improvement from baseline.
A clear-cut dose response was evident: the IGA response rate was 43.8% in the group on abrocitinib at 200 mg/day, 23.7% with 100 mg, and 7.9% with placebo. The EASI-75 response rates were 62.7%, 39.7%, and 11.8%, respectively, with a statistically significant separation from placebo by week 2.
Key secondary endpoints included the rigorous EASI-90 response rate: 38.6% with the higher dose of abrocitinib, 18.6% at 100 mg per day, and 5.3% with placebo. And again, that’s without resort to topical steroids, Dr. Simpson noted.
Another important secondary endpoint was the proportion of patients who achieved at least a 4-point improvement in itch on the pruritus self-rating scale by week 12: 57.2%, 37.7%, and 15.3%. The rapidity of the improvement was notable: By week 2, this endpoint was achieved in 45.6% of patients on abrocitinib on the 200-mg dose, 20.4% on the 100-mg dose, and 2.7% of placebo-treated controls.
“By day 2, 1 day after taking the first pill, you can see really nice reductions in itch at both doses, compared with placebo,” the dermatologist said.
Study dropout rates were low despite the inability to utilize topical steroids: 11% in the higher-dose abrocitinib group and 13.5% with abrocitinib at 100 mg/day, both of which were lower than in controls.
Turning to safety results, Dr. Simpson noted that the 9.1% rate of discontinuations because of adverse events in the placebo group was significantly higher than the 5.8% rates in each of the active treatment arms. The serious adverse event rate was 3.2% in each of the abrocitinib groups and 1.9% with placebo. Among the serious adverse events in abrocitinib-treated patients Dr. Simpson considered worthy of special mention were a single case of inflammatory bowel disease, which resolved after halting treatment; one case of peritonsillitis; and a case of pancreatitis in an alcoholic patient. No deaths, major adverse cardiovascular events, or malignancies occurred in this brief 12-week trial. Nor were there any cases of deep vein thrombosis or pulmonary embolism, which has been an issue with some other JAK inhibitors.
“We need long-term safety data, of course,” he said.
Laboratory findings were generally unremarkable, with no clinically significant changes. Platelet counts dropped to a nadir at about 4 weeks while staying within normal range, then came back up. Mean LDL cholesterol levels rose by about 10%, an unwelcome event that was counterbalanced by a favorable 20% rise in HDL cholesterol.
Asked about the efficacy rates in the 20%-plus adolescent study participants, compared with the adults, Dr. Simpson replied that a detailed analysis is planned, adding: “I can say that things are looking pretty similar.”
Abrocitinib is selective for inhibition of JAK1, with resultant modulation of interleukins-4, -13, and -31, as well as interferon-gamma, all of which are cytokines involved in the pathophysiology of AD.
Of note, Pfizer, the drug’s developer, has announced positive results from the sister pivotal phase 3 trial, JADE MONO-2, with full details forthcoming in early 2020. Results of a phase 2b study of abrocitinib were also recently published (JAMA Dermatol. 2019 Oct 2. doi: 10.1001/jamadermatol.2019.2855).
Dr. Simpson reported receiving research grants from and serving as a consultant to Pfizer, the study sponsor. He has similar financial relationships with close to a dozen other pharmaceutical companies.
MADRID – (AD) on the strength of its knockout performance in the pivotal phase 3 JADE MONO-1 trial, presented at the annual congress of the European Academy of Dermatology and Venereology.
“The IGA [Investigator Global Assessment] and EASI [Eczema Area and Severity Index]-75 responses for both doses of abrocitinib were significantly greater than placebo as early as week 2 and continued to increase until week 12 with no plateau. This study stopped at week 12. I would have liked to see what happened at 16 weeks or further. I don’t know where this is going to end up maxing out,” commented principal investigator Eric Simpson, MD, professor of dermatology at Oregon Health & Science University, Portland.
JADE MONO-1 (JAK1 Atopic Dermatitis Efficacy and Safety Monotherapy–1) was a 12-week, double-blind, multicenter study which included 387 adolescents and adults with moderate or severe AD randomized 2:2:1 to abrocitinib at 200 mg or 100 mg once daily, or to placebo. Overall, this was a fairly severely affected population, with a mean baseline EASI (Eczema Area and Severity Index) score of about 30, a peak pruritus numeric rating scale score of 7 out of 10, and a mean Dermatology Quality Life Index score greater than 14. Nonetheless, this was a rigorous abrocitinib monotherapy trial, as participants were not allowed to take even a single dose of topical corticosteroids.
The coprimary endpoints in JADE MONO-1 were achievement of an IGA score of 0 or 1 – that is, clear or almost clear – at week 12 plus at least a 2-point improvement, compared with baseline on the 0-4 scale, and an EASI-75 response as defined by a 75% or greater improvement from baseline.
A clear-cut dose response was evident: the IGA response rate was 43.8% in the group on abrocitinib at 200 mg/day, 23.7% with 100 mg, and 7.9% with placebo. The EASI-75 response rates were 62.7%, 39.7%, and 11.8%, respectively, with a statistically significant separation from placebo by week 2.
Key secondary endpoints included the rigorous EASI-90 response rate: 38.6% with the higher dose of abrocitinib, 18.6% at 100 mg per day, and 5.3% with placebo. And again, that’s without resort to topical steroids, Dr. Simpson noted.
Another important secondary endpoint was the proportion of patients who achieved at least a 4-point improvement in itch on the pruritus self-rating scale by week 12: 57.2%, 37.7%, and 15.3%. The rapidity of the improvement was notable: By week 2, this endpoint was achieved in 45.6% of patients on abrocitinib on the 200-mg dose, 20.4% on the 100-mg dose, and 2.7% of placebo-treated controls.
“By day 2, 1 day after taking the first pill, you can see really nice reductions in itch at both doses, compared with placebo,” the dermatologist said.
Study dropout rates were low despite the inability to utilize topical steroids: 11% in the higher-dose abrocitinib group and 13.5% with abrocitinib at 100 mg/day, both of which were lower than in controls.
Turning to safety results, Dr. Simpson noted that the 9.1% rate of discontinuations because of adverse events in the placebo group was significantly higher than the 5.8% rates in each of the active treatment arms. The serious adverse event rate was 3.2% in each of the abrocitinib groups and 1.9% with placebo. Among the serious adverse events in abrocitinib-treated patients Dr. Simpson considered worthy of special mention were a single case of inflammatory bowel disease, which resolved after halting treatment; one case of peritonsillitis; and a case of pancreatitis in an alcoholic patient. No deaths, major adverse cardiovascular events, or malignancies occurred in this brief 12-week trial. Nor were there any cases of deep vein thrombosis or pulmonary embolism, which has been an issue with some other JAK inhibitors.
“We need long-term safety data, of course,” he said.
Laboratory findings were generally unremarkable, with no clinically significant changes. Platelet counts dropped to a nadir at about 4 weeks while staying within normal range, then came back up. Mean LDL cholesterol levels rose by about 10%, an unwelcome event that was counterbalanced by a favorable 20% rise in HDL cholesterol.
Asked about the efficacy rates in the 20%-plus adolescent study participants, compared with the adults, Dr. Simpson replied that a detailed analysis is planned, adding: “I can say that things are looking pretty similar.”
Abrocitinib is selective for inhibition of JAK1, with resultant modulation of interleukins-4, -13, and -31, as well as interferon-gamma, all of which are cytokines involved in the pathophysiology of AD.
Of note, Pfizer, the drug’s developer, has announced positive results from the sister pivotal phase 3 trial, JADE MONO-2, with full details forthcoming in early 2020. Results of a phase 2b study of abrocitinib were also recently published (JAMA Dermatol. 2019 Oct 2. doi: 10.1001/jamadermatol.2019.2855).
Dr. Simpson reported receiving research grants from and serving as a consultant to Pfizer, the study sponsor. He has similar financial relationships with close to a dozen other pharmaceutical companies.
MADRID – (AD) on the strength of its knockout performance in the pivotal phase 3 JADE MONO-1 trial, presented at the annual congress of the European Academy of Dermatology and Venereology.
“The IGA [Investigator Global Assessment] and EASI [Eczema Area and Severity Index]-75 responses for both doses of abrocitinib were significantly greater than placebo as early as week 2 and continued to increase until week 12 with no plateau. This study stopped at week 12. I would have liked to see what happened at 16 weeks or further. I don’t know where this is going to end up maxing out,” commented principal investigator Eric Simpson, MD, professor of dermatology at Oregon Health & Science University, Portland.
JADE MONO-1 (JAK1 Atopic Dermatitis Efficacy and Safety Monotherapy–1) was a 12-week, double-blind, multicenter study which included 387 adolescents and adults with moderate or severe AD randomized 2:2:1 to abrocitinib at 200 mg or 100 mg once daily, or to placebo. Overall, this was a fairly severely affected population, with a mean baseline EASI (Eczema Area and Severity Index) score of about 30, a peak pruritus numeric rating scale score of 7 out of 10, and a mean Dermatology Quality Life Index score greater than 14. Nonetheless, this was a rigorous abrocitinib monotherapy trial, as participants were not allowed to take even a single dose of topical corticosteroids.
The coprimary endpoints in JADE MONO-1 were achievement of an IGA score of 0 or 1 – that is, clear or almost clear – at week 12 plus at least a 2-point improvement, compared with baseline on the 0-4 scale, and an EASI-75 response as defined by a 75% or greater improvement from baseline.
A clear-cut dose response was evident: the IGA response rate was 43.8% in the group on abrocitinib at 200 mg/day, 23.7% with 100 mg, and 7.9% with placebo. The EASI-75 response rates were 62.7%, 39.7%, and 11.8%, respectively, with a statistically significant separation from placebo by week 2.
Key secondary endpoints included the rigorous EASI-90 response rate: 38.6% with the higher dose of abrocitinib, 18.6% at 100 mg per day, and 5.3% with placebo. And again, that’s without resort to topical steroids, Dr. Simpson noted.
Another important secondary endpoint was the proportion of patients who achieved at least a 4-point improvement in itch on the pruritus self-rating scale by week 12: 57.2%, 37.7%, and 15.3%. The rapidity of the improvement was notable: By week 2, this endpoint was achieved in 45.6% of patients on abrocitinib on the 200-mg dose, 20.4% on the 100-mg dose, and 2.7% of placebo-treated controls.
“By day 2, 1 day after taking the first pill, you can see really nice reductions in itch at both doses, compared with placebo,” the dermatologist said.
Study dropout rates were low despite the inability to utilize topical steroids: 11% in the higher-dose abrocitinib group and 13.5% with abrocitinib at 100 mg/day, both of which were lower than in controls.
Turning to safety results, Dr. Simpson noted that the 9.1% rate of discontinuations because of adverse events in the placebo group was significantly higher than the 5.8% rates in each of the active treatment arms. The serious adverse event rate was 3.2% in each of the abrocitinib groups and 1.9% with placebo. Among the serious adverse events in abrocitinib-treated patients Dr. Simpson considered worthy of special mention were a single case of inflammatory bowel disease, which resolved after halting treatment; one case of peritonsillitis; and a case of pancreatitis in an alcoholic patient. No deaths, major adverse cardiovascular events, or malignancies occurred in this brief 12-week trial. Nor were there any cases of deep vein thrombosis or pulmonary embolism, which has been an issue with some other JAK inhibitors.
“We need long-term safety data, of course,” he said.
Laboratory findings were generally unremarkable, with no clinically significant changes. Platelet counts dropped to a nadir at about 4 weeks while staying within normal range, then came back up. Mean LDL cholesterol levels rose by about 10%, an unwelcome event that was counterbalanced by a favorable 20% rise in HDL cholesterol.
Asked about the efficacy rates in the 20%-plus adolescent study participants, compared with the adults, Dr. Simpson replied that a detailed analysis is planned, adding: “I can say that things are looking pretty similar.”
Abrocitinib is selective for inhibition of JAK1, with resultant modulation of interleukins-4, -13, and -31, as well as interferon-gamma, all of which are cytokines involved in the pathophysiology of AD.
Of note, Pfizer, the drug’s developer, has announced positive results from the sister pivotal phase 3 trial, JADE MONO-2, with full details forthcoming in early 2020. Results of a phase 2b study of abrocitinib were also recently published (JAMA Dermatol. 2019 Oct 2. doi: 10.1001/jamadermatol.2019.2855).
Dr. Simpson reported receiving research grants from and serving as a consultant to Pfizer, the study sponsor. He has similar financial relationships with close to a dozen other pharmaceutical companies.
REPORTING FROM THE EADV CONGRESS
Rituximab bests mycophenolate in pemphigus vulgaris
MADRID – Pascal Joly, MD, PhD, reported at the annual congress of the European Academy of Dermatology and Venereology.
Not only did rituximab prove superior in terms of efficacy in the PEMPHIX trial, with a five times greater likelihood of achieving a complete remission lasting for at least 16 weeks while off oral corticosteroids than with mycophenolate mofetil in the 52-week study, but the total number of disease flares in the mycophenolate mofetil group was five times higher. Moreover, rituximab-treated patients received a markedly lower cumulative dose of prednisone as well.
“Rituximab has a superior overall benefit-risk profile, compared to mycophenolate mofetil, in patients with moderate to severe pemphigus vulgaris,” concluded Dr. Joly, professor of dermatology at the University of Rouen (France) and president of the French Society of Dermatology.
The study was undertaken because mycophenolate mofetil is commonly used as a corticosteroid-sparing drug in patients with pemphigus vulgaris, even though its efficacy for the treatment of this rare, severe autoimmune blistering disease is unproven, he explained.
In contrast, rituximab was approved by the Food and Drug Administration and European regulators for treatment of pemphigus vulgaris on the strength of the pivotal phase 3 Ritux 3 trial – also led by Dr. Joly – which demonstrated the superiority of this intravenously administered anti-CD20 monoclonal antibody plus short-term prednisone over high-dose corticosteroid monotherapy, which for decades had been the standard treatment despite its considerable toxicity burden (Lancet. 2017 May 20;389[10083]:2031-40).
An independently conducted analysis of Ritux 3 recently concluded that rituximab plus short-term prednisone was more effective than prednisone alone, with a lower risk of life-threatening, corticosteroid-related adverse events and less cumulative corticosteroid exposure (Br J Dermatol. 2019 Sep 5. doi: 10.1111/bjd.18482).
Also, an international panel of 93 pemphigus experts has declared that rituximab should be considered an evidence-based first-line therapy for moderate to-severe pemphigus (J Am Acad Dermatol. 2018 Feb 10. doi: 10.1016/j.jaad.2018.02.021).
The phase 3, placebo-controlled PEMPHIX trial randomized 135 patients with moderate or severe pemphigus at 49 academic medical centers in the United States and nine other countries to double-blind rituximab or mycophenolate mofetil on top of background oral prednisone at 1.0-1.5 mg/kg per day, with the steroid to be tapered and discontinued within 4-6 months.
The primary endpoint of the study was the proportion of patients in each study arm at week 52 who had achieved a sustained complete remission lasting for at least 16 weeks while off prednisone. The rate was 40.3% in the rituximab group and 9.5% with mycophenolate mofetil, for a 383% increased likelihood of sustained complete remission in the rituximab group.
In addition, all of the study’s secondary endpoints significantly favored rituximab. The median cumulative dose of corticosteroid was 2.7 g through 52 weeks in the rituximab arm, compared with 4 g with mycophenolate mofetil. The total number of disease flares over 52 weeks was 6 in the rituximab group and 44 in the mycophenolate arm. Five rituximab-treated patients experienced disease flares, as did 26 on mycophenolate. Thus, the likelihood of a flare was seven times lower with rituximab.
Scores on the Dermatology Life Quality Index improved by an average of 8.87 points from baseline to week 52 in the rituximab group versus 6 points with mycophenolate. And 62.7% of rituximab-treated patients had a week-52 Dermatology Life Quality Index score of 0, meaning no impact of the disease on their quality of life, compared with 25% of the mycophenolate group.
Dr. Joly characterized the safety profile of rituximab as “manageable, with acceptable tolerability.” About 9% of the rituximab group and 7.4% of mycophenolate-treated patients had one or more treatment-related adverse events, a nonsignificant difference. The rate of treatment-related serious infections was 3.0% with rituximab and 2.9% with mycophenolate. Serious infusion reactions leading to study withdrawal occurred in three patients on rituximab and one on mycophenolate. The rate of grade 3 or worse corticosteroid-related adverse events was 1.5% with rituximab and significantly greater at 7.4% with mycophenolate.
An additional 48-week safety assessment beyond the 52-week primary outcome is ongoing.
Asked what future role he sees for mycophenolate in pemphigus vulgaris, Dr. Joly replied that the only study in the literature that shows the drug outperforms placebo was seriously flawed. “In the future, it’s very likely that the indications for use of mycophenolate in pemphigus vulgaris will be fewer and fewer,” the dermatologist added.
In reply to a question about the merits of routine antibiotic prophylaxis against pneumocystis carinii pneumonia in patients taking rituximab for pemphigus vulgaris, Dr. Joly said the incidence isn’t sufficiently high to justify such practice. After all, he noted, there were no cases of pneumocystis carinii pneumonia in rituximab-treated patients in PEMPHIX and only one in Ritux 3.
EADV Scientific Programming Committee Chair Brigitte Dreno, MD, PhD, professor and head of dermatology at University Hospital in Nantes, France, inquired as to whether there’s a role for maintenance therapy in a potent rituximab-based treatment strategy such as utilized in PEMPHIX.
Definitely, Dr. Joly replied. However, further study is required to work out the best maintenance program.
“There are many arguments for maintenance therapy in these patients. For one, the frequency of relapses increases with the length of follow-up. Also, anti–desmoglein-specific T cells can still be detected after rituximab therapy, even in patients in complete remission. So there is a need for maintenance therapy, perhaps at months 6, 12, and 18, but the optimal regimen isn’t determined yet,” according to Dr. Joly.
PEMPHIX was sponsored by F. Hoffmann-La Roche. Dr. Joly reported serving as a consultant to Roche, Amgen, Principia Biopharma, and Argenx.
MADRID – Pascal Joly, MD, PhD, reported at the annual congress of the European Academy of Dermatology and Venereology.
Not only did rituximab prove superior in terms of efficacy in the PEMPHIX trial, with a five times greater likelihood of achieving a complete remission lasting for at least 16 weeks while off oral corticosteroids than with mycophenolate mofetil in the 52-week study, but the total number of disease flares in the mycophenolate mofetil group was five times higher. Moreover, rituximab-treated patients received a markedly lower cumulative dose of prednisone as well.
“Rituximab has a superior overall benefit-risk profile, compared to mycophenolate mofetil, in patients with moderate to severe pemphigus vulgaris,” concluded Dr. Joly, professor of dermatology at the University of Rouen (France) and president of the French Society of Dermatology.
The study was undertaken because mycophenolate mofetil is commonly used as a corticosteroid-sparing drug in patients with pemphigus vulgaris, even though its efficacy for the treatment of this rare, severe autoimmune blistering disease is unproven, he explained.
In contrast, rituximab was approved by the Food and Drug Administration and European regulators for treatment of pemphigus vulgaris on the strength of the pivotal phase 3 Ritux 3 trial – also led by Dr. Joly – which demonstrated the superiority of this intravenously administered anti-CD20 monoclonal antibody plus short-term prednisone over high-dose corticosteroid monotherapy, which for decades had been the standard treatment despite its considerable toxicity burden (Lancet. 2017 May 20;389[10083]:2031-40).
An independently conducted analysis of Ritux 3 recently concluded that rituximab plus short-term prednisone was more effective than prednisone alone, with a lower risk of life-threatening, corticosteroid-related adverse events and less cumulative corticosteroid exposure (Br J Dermatol. 2019 Sep 5. doi: 10.1111/bjd.18482).
Also, an international panel of 93 pemphigus experts has declared that rituximab should be considered an evidence-based first-line therapy for moderate to-severe pemphigus (J Am Acad Dermatol. 2018 Feb 10. doi: 10.1016/j.jaad.2018.02.021).
The phase 3, placebo-controlled PEMPHIX trial randomized 135 patients with moderate or severe pemphigus at 49 academic medical centers in the United States and nine other countries to double-blind rituximab or mycophenolate mofetil on top of background oral prednisone at 1.0-1.5 mg/kg per day, with the steroid to be tapered and discontinued within 4-6 months.
The primary endpoint of the study was the proportion of patients in each study arm at week 52 who had achieved a sustained complete remission lasting for at least 16 weeks while off prednisone. The rate was 40.3% in the rituximab group and 9.5% with mycophenolate mofetil, for a 383% increased likelihood of sustained complete remission in the rituximab group.
In addition, all of the study’s secondary endpoints significantly favored rituximab. The median cumulative dose of corticosteroid was 2.7 g through 52 weeks in the rituximab arm, compared with 4 g with mycophenolate mofetil. The total number of disease flares over 52 weeks was 6 in the rituximab group and 44 in the mycophenolate arm. Five rituximab-treated patients experienced disease flares, as did 26 on mycophenolate. Thus, the likelihood of a flare was seven times lower with rituximab.
Scores on the Dermatology Life Quality Index improved by an average of 8.87 points from baseline to week 52 in the rituximab group versus 6 points with mycophenolate. And 62.7% of rituximab-treated patients had a week-52 Dermatology Life Quality Index score of 0, meaning no impact of the disease on their quality of life, compared with 25% of the mycophenolate group.
Dr. Joly characterized the safety profile of rituximab as “manageable, with acceptable tolerability.” About 9% of the rituximab group and 7.4% of mycophenolate-treated patients had one or more treatment-related adverse events, a nonsignificant difference. The rate of treatment-related serious infections was 3.0% with rituximab and 2.9% with mycophenolate. Serious infusion reactions leading to study withdrawal occurred in three patients on rituximab and one on mycophenolate. The rate of grade 3 or worse corticosteroid-related adverse events was 1.5% with rituximab and significantly greater at 7.4% with mycophenolate.
An additional 48-week safety assessment beyond the 52-week primary outcome is ongoing.
Asked what future role he sees for mycophenolate in pemphigus vulgaris, Dr. Joly replied that the only study in the literature that shows the drug outperforms placebo was seriously flawed. “In the future, it’s very likely that the indications for use of mycophenolate in pemphigus vulgaris will be fewer and fewer,” the dermatologist added.
In reply to a question about the merits of routine antibiotic prophylaxis against pneumocystis carinii pneumonia in patients taking rituximab for pemphigus vulgaris, Dr. Joly said the incidence isn’t sufficiently high to justify such practice. After all, he noted, there were no cases of pneumocystis carinii pneumonia in rituximab-treated patients in PEMPHIX and only one in Ritux 3.
EADV Scientific Programming Committee Chair Brigitte Dreno, MD, PhD, professor and head of dermatology at University Hospital in Nantes, France, inquired as to whether there’s a role for maintenance therapy in a potent rituximab-based treatment strategy such as utilized in PEMPHIX.
Definitely, Dr. Joly replied. However, further study is required to work out the best maintenance program.
“There are many arguments for maintenance therapy in these patients. For one, the frequency of relapses increases with the length of follow-up. Also, anti–desmoglein-specific T cells can still be detected after rituximab therapy, even in patients in complete remission. So there is a need for maintenance therapy, perhaps at months 6, 12, and 18, but the optimal regimen isn’t determined yet,” according to Dr. Joly.
PEMPHIX was sponsored by F. Hoffmann-La Roche. Dr. Joly reported serving as a consultant to Roche, Amgen, Principia Biopharma, and Argenx.
MADRID – Pascal Joly, MD, PhD, reported at the annual congress of the European Academy of Dermatology and Venereology.
Not only did rituximab prove superior in terms of efficacy in the PEMPHIX trial, with a five times greater likelihood of achieving a complete remission lasting for at least 16 weeks while off oral corticosteroids than with mycophenolate mofetil in the 52-week study, but the total number of disease flares in the mycophenolate mofetil group was five times higher. Moreover, rituximab-treated patients received a markedly lower cumulative dose of prednisone as well.
“Rituximab has a superior overall benefit-risk profile, compared to mycophenolate mofetil, in patients with moderate to severe pemphigus vulgaris,” concluded Dr. Joly, professor of dermatology at the University of Rouen (France) and president of the French Society of Dermatology.
The study was undertaken because mycophenolate mofetil is commonly used as a corticosteroid-sparing drug in patients with pemphigus vulgaris, even though its efficacy for the treatment of this rare, severe autoimmune blistering disease is unproven, he explained.
In contrast, rituximab was approved by the Food and Drug Administration and European regulators for treatment of pemphigus vulgaris on the strength of the pivotal phase 3 Ritux 3 trial – also led by Dr. Joly – which demonstrated the superiority of this intravenously administered anti-CD20 monoclonal antibody plus short-term prednisone over high-dose corticosteroid monotherapy, which for decades had been the standard treatment despite its considerable toxicity burden (Lancet. 2017 May 20;389[10083]:2031-40).
An independently conducted analysis of Ritux 3 recently concluded that rituximab plus short-term prednisone was more effective than prednisone alone, with a lower risk of life-threatening, corticosteroid-related adverse events and less cumulative corticosteroid exposure (Br J Dermatol. 2019 Sep 5. doi: 10.1111/bjd.18482).
Also, an international panel of 93 pemphigus experts has declared that rituximab should be considered an evidence-based first-line therapy for moderate to-severe pemphigus (J Am Acad Dermatol. 2018 Feb 10. doi: 10.1016/j.jaad.2018.02.021).
The phase 3, placebo-controlled PEMPHIX trial randomized 135 patients with moderate or severe pemphigus at 49 academic medical centers in the United States and nine other countries to double-blind rituximab or mycophenolate mofetil on top of background oral prednisone at 1.0-1.5 mg/kg per day, with the steroid to be tapered and discontinued within 4-6 months.
The primary endpoint of the study was the proportion of patients in each study arm at week 52 who had achieved a sustained complete remission lasting for at least 16 weeks while off prednisone. The rate was 40.3% in the rituximab group and 9.5% with mycophenolate mofetil, for a 383% increased likelihood of sustained complete remission in the rituximab group.
In addition, all of the study’s secondary endpoints significantly favored rituximab. The median cumulative dose of corticosteroid was 2.7 g through 52 weeks in the rituximab arm, compared with 4 g with mycophenolate mofetil. The total number of disease flares over 52 weeks was 6 in the rituximab group and 44 in the mycophenolate arm. Five rituximab-treated patients experienced disease flares, as did 26 on mycophenolate. Thus, the likelihood of a flare was seven times lower with rituximab.
Scores on the Dermatology Life Quality Index improved by an average of 8.87 points from baseline to week 52 in the rituximab group versus 6 points with mycophenolate. And 62.7% of rituximab-treated patients had a week-52 Dermatology Life Quality Index score of 0, meaning no impact of the disease on their quality of life, compared with 25% of the mycophenolate group.
Dr. Joly characterized the safety profile of rituximab as “manageable, with acceptable tolerability.” About 9% of the rituximab group and 7.4% of mycophenolate-treated patients had one or more treatment-related adverse events, a nonsignificant difference. The rate of treatment-related serious infections was 3.0% with rituximab and 2.9% with mycophenolate. Serious infusion reactions leading to study withdrawal occurred in three patients on rituximab and one on mycophenolate. The rate of grade 3 or worse corticosteroid-related adverse events was 1.5% with rituximab and significantly greater at 7.4% with mycophenolate.
An additional 48-week safety assessment beyond the 52-week primary outcome is ongoing.
Asked what future role he sees for mycophenolate in pemphigus vulgaris, Dr. Joly replied that the only study in the literature that shows the drug outperforms placebo was seriously flawed. “In the future, it’s very likely that the indications for use of mycophenolate in pemphigus vulgaris will be fewer and fewer,” the dermatologist added.
In reply to a question about the merits of routine antibiotic prophylaxis against pneumocystis carinii pneumonia in patients taking rituximab for pemphigus vulgaris, Dr. Joly said the incidence isn’t sufficiently high to justify such practice. After all, he noted, there were no cases of pneumocystis carinii pneumonia in rituximab-treated patients in PEMPHIX and only one in Ritux 3.
EADV Scientific Programming Committee Chair Brigitte Dreno, MD, PhD, professor and head of dermatology at University Hospital in Nantes, France, inquired as to whether there’s a role for maintenance therapy in a potent rituximab-based treatment strategy such as utilized in PEMPHIX.
Definitely, Dr. Joly replied. However, further study is required to work out the best maintenance program.
“There are many arguments for maintenance therapy in these patients. For one, the frequency of relapses increases with the length of follow-up. Also, anti–desmoglein-specific T cells can still be detected after rituximab therapy, even in patients in complete remission. So there is a need for maintenance therapy, perhaps at months 6, 12, and 18, but the optimal regimen isn’t determined yet,” according to Dr. Joly.
PEMPHIX was sponsored by F. Hoffmann-La Roche. Dr. Joly reported serving as a consultant to Roche, Amgen, Principia Biopharma, and Argenx.
REPORTING FROM THE EADV CONGRESS
Telangiectasias with tight skin
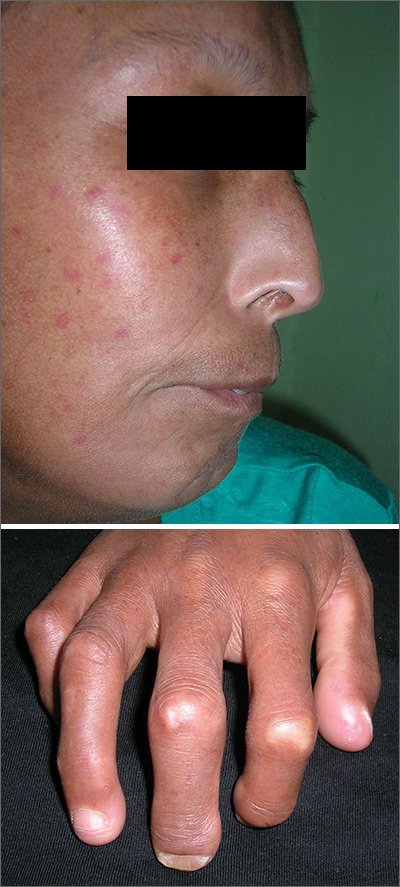
The FP suspected that the patient had CREST (calcinosis, Raynaud’s phenomenon, esophageal dysmotility, sclerodactyly, telangiectasias) syndrome also known as limited cutaneous systemic sclerosis (LcSSc). He examined her arms and found that she had firm nodules around the elbows consistent with calcinosis. Further history revealed Raynaud's phenomenon. This clinched the diagnosis of CREST syndrome. The FP ordered blood tests, a chest x-ray (CXR), and pulmonary function tests to determine if there was pulmonary involvement and to learn more about possible systemic effects of her disease.
Systemic sclerosis (scleroderma) is characterized by skin induration and thickening accompanied by variable tissue fibrosis and inflammatory infiltration in numerous visceral organs. Systemic sclerosis can be diffuse or limited to the skin and adjacent tissues (LcSSc). Patients with LcSSc usually have skin sclerosis that is restricted to the hands and, to a lesser extent, the face and neck.
The patient’s antinuclear antibody test was positive with a speckled nucleolar staining pattern, which is a common finding in systemic sclerosis. Her CXR showed evidence of interstitial lung disease with a ground glass pattern. Her pulmonary function test showed a diminished diffusion capacity. Pulmonary disease is seen in all types of systemic sclerosis and not diagnostic for one type. The patient was referred to Rheumatology for further diagnosis and management.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Mayeaux EJ, Usatine R. Scleroderma and morphea. In: Usatine R, Smith M, Mayeaux EJ, et al. eds. Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019:1204-1212.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the 3rd edition of the Color Atlas and Synopsis of Family Medicine as an app by clicking on this link: https://usatinemedia.com/app/color-atlas-of-family-medicine/

The FP suspected that the patient had CREST (calcinosis, Raynaud’s phenomenon, esophageal dysmotility, sclerodactyly, telangiectasias) syndrome also known as limited cutaneous systemic sclerosis (LcSSc). He examined her arms and found that she had firm nodules around the elbows consistent with calcinosis. Further history revealed Raynaud's phenomenon. This clinched the diagnosis of CREST syndrome. The FP ordered blood tests, a chest x-ray (CXR), and pulmonary function tests to determine if there was pulmonary involvement and to learn more about possible systemic effects of her disease.
Systemic sclerosis (scleroderma) is characterized by skin induration and thickening accompanied by variable tissue fibrosis and inflammatory infiltration in numerous visceral organs. Systemic sclerosis can be diffuse or limited to the skin and adjacent tissues (LcSSc). Patients with LcSSc usually have skin sclerosis that is restricted to the hands and, to a lesser extent, the face and neck.
The patient’s antinuclear antibody test was positive with a speckled nucleolar staining pattern, which is a common finding in systemic sclerosis. Her CXR showed evidence of interstitial lung disease with a ground glass pattern. Her pulmonary function test showed a diminished diffusion capacity. Pulmonary disease is seen in all types of systemic sclerosis and not diagnostic for one type. The patient was referred to Rheumatology for further diagnosis and management.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Mayeaux EJ, Usatine R. Scleroderma and morphea. In: Usatine R, Smith M, Mayeaux EJ, et al. eds. Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019:1204-1212.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the 3rd edition of the Color Atlas and Synopsis of Family Medicine as an app by clicking on this link: https://usatinemedia.com/app/color-atlas-of-family-medicine/

The FP suspected that the patient had CREST (calcinosis, Raynaud’s phenomenon, esophageal dysmotility, sclerodactyly, telangiectasias) syndrome also known as limited cutaneous systemic sclerosis (LcSSc). He examined her arms and found that she had firm nodules around the elbows consistent with calcinosis. Further history revealed Raynaud's phenomenon. This clinched the diagnosis of CREST syndrome. The FP ordered blood tests, a chest x-ray (CXR), and pulmonary function tests to determine if there was pulmonary involvement and to learn more about possible systemic effects of her disease.
Systemic sclerosis (scleroderma) is characterized by skin induration and thickening accompanied by variable tissue fibrosis and inflammatory infiltration in numerous visceral organs. Systemic sclerosis can be diffuse or limited to the skin and adjacent tissues (LcSSc). Patients with LcSSc usually have skin sclerosis that is restricted to the hands and, to a lesser extent, the face and neck.
The patient’s antinuclear antibody test was positive with a speckled nucleolar staining pattern, which is a common finding in systemic sclerosis. Her CXR showed evidence of interstitial lung disease with a ground glass pattern. Her pulmonary function test showed a diminished diffusion capacity. Pulmonary disease is seen in all types of systemic sclerosis and not diagnostic for one type. The patient was referred to Rheumatology for further diagnosis and management.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Mayeaux EJ, Usatine R. Scleroderma and morphea. In: Usatine R, Smith M, Mayeaux EJ, et al. eds. Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019:1204-1212.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the 3rd edition of the Color Atlas and Synopsis of Family Medicine as an app by clicking on this link: https://usatinemedia.com/app/color-atlas-of-family-medicine/
Hyperhidrosis treatment options update
SEATTLE – , David Pariser, MD, said at the annual Coastal Dermatology Symposium.
Hyperhidrosis is among the dermatological conditions that have the greatest impact on quality of life, and it can be particularly concerning to teens, said Dr. Pariser, professor of dermatology at Eastern Virginia Medical School, Norfolk. He referred to some new developments for an old, often misunderstood, standby: antiperspirants. “I am amazed that many people do not know the difference between an antiperspirant and a deodorant,” he said, pointing out that antiperspirants contain active ingredients – aluminum and zirconium salts – that block sweat glands, while deodorants contain a masking fragrance.
There are new-generation topical antiperspirants available over the counter, with descriptions that include “clinical strength” or “clinical protection” on the labels; they come in a box and cost about twice as much as standard products. “But they are better, and they work just as well as some of the commercial preparations,” Dr. Pariser said at the meeting, jointly presented by the University of Louisville and Global Academy for Medical Education.
One issue he highlighted was that antiperspirants are often misapplied. They shouldn’t be applied on wet skin because they react with water to create hydrochloric acid, which can irritate the skin, he said. The best time to apply an antiperspirant is right before bedtime, since it gives the salts time to clog sweat pores before sweat or water can interfere. “The plugs last for a couple of days,” so there’s no need to worry about rinsing the product off during a morning shower, he noted.
Additional therapeutic options include agents like oral glycopyrrolate, starting at a low dose (1 mg twice per day), increasing by 1 mg/day weekly until efficacy is achieved or limited by adverse events. There is also a glycopyrrolate oral solution 1mg/5ml (Cuvposa) that can be used in children.
A topical version of the anticholinergic glycopyrronium tosylate, applied using an infused cloth, was approved for treating axillary hyperhidrosis in June2018 and offers the potential for an enhanced local anticholinergic effect. Dr. Pariser, one of the authors, discussed the recently published results of two pivotal studies that found good improvement in a specially-designed quality of life endpoint (J Am Acad Derm. 2019; Jan;80[1]:128-138.e2).
Efficacy in a subanalysis of 44 pediatric subjects (ages 9-16 years) was similar as those reported in adults, and the rate of those reporting dry mouth (24% in both age groups) was similar. Of concern was a 16% rate of mydriasis in the pediatric group, compared with 6% in the older group. One patient even wound up in the emergency room for a stroke work-up as a result, said Dr. Pariser, who is confident that the problem was caused by inadvertent exposure to the eye during application. He advises patients to avoid contact with the eyes.
Other approaches to treatment of hyperhidrosis include oxybutynin, iontophoresis, an microwave thermolysis (which may also reduce odor and hair). Endoscopic thoracic sympathectomy is effective but is the most invasive option; botulinum toxin is a minimally invasive alternative to surgery.
For those who sweat when they experience anxiety, propranolol 5-10 mg taken about 1 hour before an event that could cause hyperhydrosis can be effective, said Dr. Pariser, who recommends a test dose. “I don’t normally tell patients to try something at home. But they should try this at home” before using it prior to an important event, he added.
Dr. Pariser is a consultant and/or investigator for Dermira, Brickell Biotech, TheraVida, Atacama, TDI Surgitech, Dermavant, and Revance Therapeutics. He has not done commercial speaking, has not been on speaker’s bureaus, and has no stock or options in any company.
This publication and Global Academy for Medical Education are owned by the same parent company.
SEATTLE – , David Pariser, MD, said at the annual Coastal Dermatology Symposium.
Hyperhidrosis is among the dermatological conditions that have the greatest impact on quality of life, and it can be particularly concerning to teens, said Dr. Pariser, professor of dermatology at Eastern Virginia Medical School, Norfolk. He referred to some new developments for an old, often misunderstood, standby: antiperspirants. “I am amazed that many people do not know the difference between an antiperspirant and a deodorant,” he said, pointing out that antiperspirants contain active ingredients – aluminum and zirconium salts – that block sweat glands, while deodorants contain a masking fragrance.
There are new-generation topical antiperspirants available over the counter, with descriptions that include “clinical strength” or “clinical protection” on the labels; they come in a box and cost about twice as much as standard products. “But they are better, and they work just as well as some of the commercial preparations,” Dr. Pariser said at the meeting, jointly presented by the University of Louisville and Global Academy for Medical Education.
One issue he highlighted was that antiperspirants are often misapplied. They shouldn’t be applied on wet skin because they react with water to create hydrochloric acid, which can irritate the skin, he said. The best time to apply an antiperspirant is right before bedtime, since it gives the salts time to clog sweat pores before sweat or water can interfere. “The plugs last for a couple of days,” so there’s no need to worry about rinsing the product off during a morning shower, he noted.
Additional therapeutic options include agents like oral glycopyrrolate, starting at a low dose (1 mg twice per day), increasing by 1 mg/day weekly until efficacy is achieved or limited by adverse events. There is also a glycopyrrolate oral solution 1mg/5ml (Cuvposa) that can be used in children.
A topical version of the anticholinergic glycopyrronium tosylate, applied using an infused cloth, was approved for treating axillary hyperhidrosis in June2018 and offers the potential for an enhanced local anticholinergic effect. Dr. Pariser, one of the authors, discussed the recently published results of two pivotal studies that found good improvement in a specially-designed quality of life endpoint (J Am Acad Derm. 2019; Jan;80[1]:128-138.e2).
Efficacy in a subanalysis of 44 pediatric subjects (ages 9-16 years) was similar as those reported in adults, and the rate of those reporting dry mouth (24% in both age groups) was similar. Of concern was a 16% rate of mydriasis in the pediatric group, compared with 6% in the older group. One patient even wound up in the emergency room for a stroke work-up as a result, said Dr. Pariser, who is confident that the problem was caused by inadvertent exposure to the eye during application. He advises patients to avoid contact with the eyes.
Other approaches to treatment of hyperhidrosis include oxybutynin, iontophoresis, an microwave thermolysis (which may also reduce odor and hair). Endoscopic thoracic sympathectomy is effective but is the most invasive option; botulinum toxin is a minimally invasive alternative to surgery.
For those who sweat when they experience anxiety, propranolol 5-10 mg taken about 1 hour before an event that could cause hyperhydrosis can be effective, said Dr. Pariser, who recommends a test dose. “I don’t normally tell patients to try something at home. But they should try this at home” before using it prior to an important event, he added.
Dr. Pariser is a consultant and/or investigator for Dermira, Brickell Biotech, TheraVida, Atacama, TDI Surgitech, Dermavant, and Revance Therapeutics. He has not done commercial speaking, has not been on speaker’s bureaus, and has no stock or options in any company.
This publication and Global Academy for Medical Education are owned by the same parent company.
SEATTLE – , David Pariser, MD, said at the annual Coastal Dermatology Symposium.
Hyperhidrosis is among the dermatological conditions that have the greatest impact on quality of life, and it can be particularly concerning to teens, said Dr. Pariser, professor of dermatology at Eastern Virginia Medical School, Norfolk. He referred to some new developments for an old, often misunderstood, standby: antiperspirants. “I am amazed that many people do not know the difference between an antiperspirant and a deodorant,” he said, pointing out that antiperspirants contain active ingredients – aluminum and zirconium salts – that block sweat glands, while deodorants contain a masking fragrance.
There are new-generation topical antiperspirants available over the counter, with descriptions that include “clinical strength” or “clinical protection” on the labels; they come in a box and cost about twice as much as standard products. “But they are better, and they work just as well as some of the commercial preparations,” Dr. Pariser said at the meeting, jointly presented by the University of Louisville and Global Academy for Medical Education.
One issue he highlighted was that antiperspirants are often misapplied. They shouldn’t be applied on wet skin because they react with water to create hydrochloric acid, which can irritate the skin, he said. The best time to apply an antiperspirant is right before bedtime, since it gives the salts time to clog sweat pores before sweat or water can interfere. “The plugs last for a couple of days,” so there’s no need to worry about rinsing the product off during a morning shower, he noted.
Additional therapeutic options include agents like oral glycopyrrolate, starting at a low dose (1 mg twice per day), increasing by 1 mg/day weekly until efficacy is achieved or limited by adverse events. There is also a glycopyrrolate oral solution 1mg/5ml (Cuvposa) that can be used in children.
A topical version of the anticholinergic glycopyrronium tosylate, applied using an infused cloth, was approved for treating axillary hyperhidrosis in June2018 and offers the potential for an enhanced local anticholinergic effect. Dr. Pariser, one of the authors, discussed the recently published results of two pivotal studies that found good improvement in a specially-designed quality of life endpoint (J Am Acad Derm. 2019; Jan;80[1]:128-138.e2).
Efficacy in a subanalysis of 44 pediatric subjects (ages 9-16 years) was similar as those reported in adults, and the rate of those reporting dry mouth (24% in both age groups) was similar. Of concern was a 16% rate of mydriasis in the pediatric group, compared with 6% in the older group. One patient even wound up in the emergency room for a stroke work-up as a result, said Dr. Pariser, who is confident that the problem was caused by inadvertent exposure to the eye during application. He advises patients to avoid contact with the eyes.
Other approaches to treatment of hyperhidrosis include oxybutynin, iontophoresis, an microwave thermolysis (which may also reduce odor and hair). Endoscopic thoracic sympathectomy is effective but is the most invasive option; botulinum toxin is a minimally invasive alternative to surgery.
For those who sweat when they experience anxiety, propranolol 5-10 mg taken about 1 hour before an event that could cause hyperhydrosis can be effective, said Dr. Pariser, who recommends a test dose. “I don’t normally tell patients to try something at home. But they should try this at home” before using it prior to an important event, he added.
Dr. Pariser is a consultant and/or investigator for Dermira, Brickell Biotech, TheraVida, Atacama, TDI Surgitech, Dermavant, and Revance Therapeutics. He has not done commercial speaking, has not been on speaker’s bureaus, and has no stock or options in any company.
This publication and Global Academy for Medical Education are owned by the same parent company.
EXPERT ANALYSIS FROM COASTAL DERM
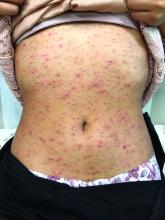
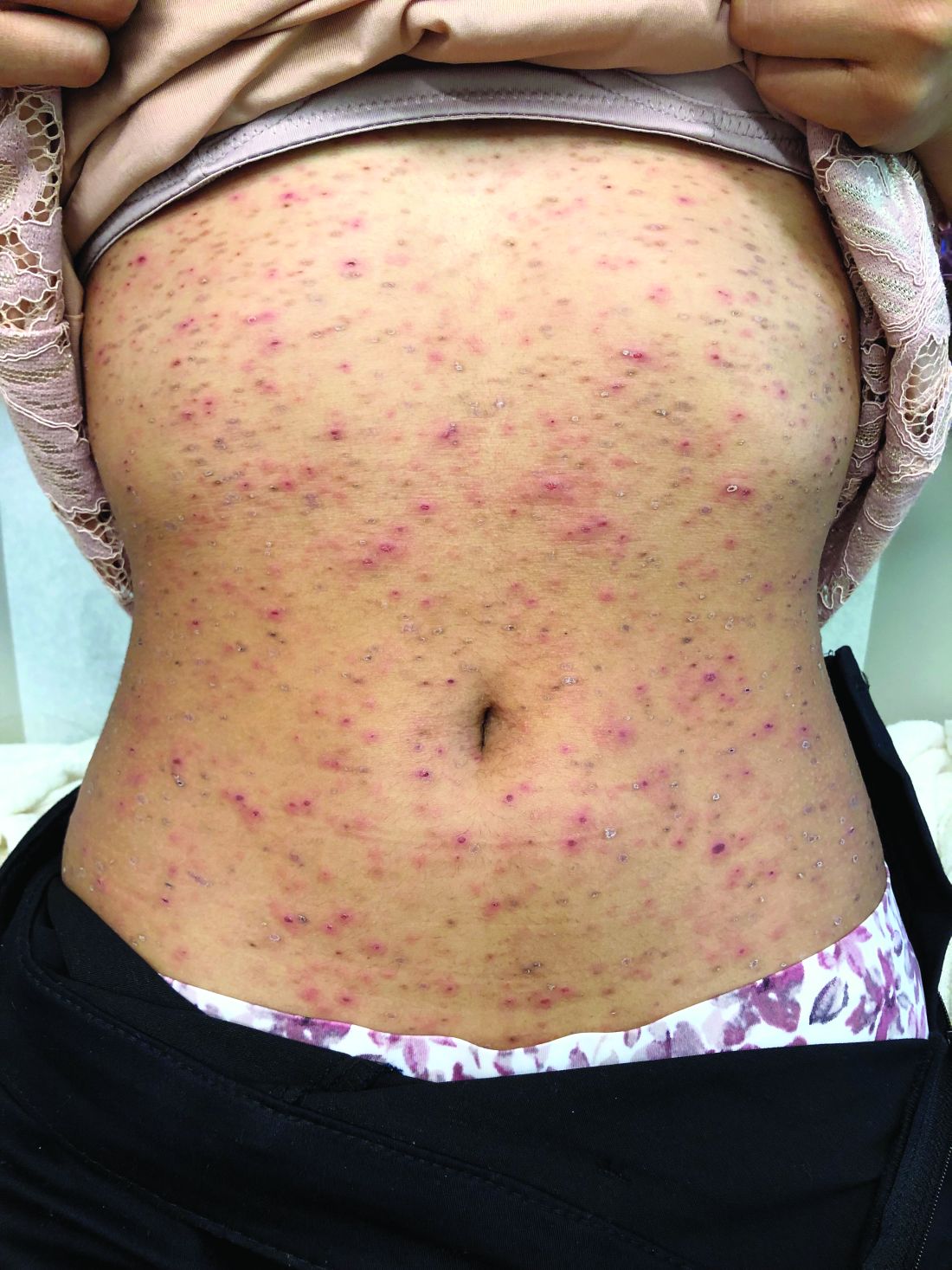
Pruritic, pink to violaceous, scaly papules
A skin biopsy of one of the lesions was consistent with pityriasis lichenoides et varioliformis acuta (PLEVA).
The patient was diagnosed with PLEVA, also known as Mucha-Habermann disease. The true incidence of the condition is not known.
The typical presentation is an abrupt onset of pink to violaceous, scaly papules and plaques that later develop violaceous or necrotic centers, like the ones seen in our patient. The lesions more typically occur on the trunk and proximal extremities, but they may present in any other part of the body, rarely in the mucosa.1 Some patients can develop the febrile, more severe form of PLEVA called febrile, ulceronecrotic Mucha-Habermann disease (FUMHD), which potentially can be life threatening.
Patients with PLEVA can complain of pruritus or a burning sensation, and in some cases can have associated arthralgia and edema. The more severe form FUMHD is characterized by persistent high fevers with associated internal organ involvement such as cardiomyopathy, small vessel vasculitis, abdominal pain, arthritis, pneumonitis, and hematologic abnormalities.2 Mucosal involvement is a common finding in patients with FUMHD.
The pathogenesis of PLEVA is not very well understood. Some theories include a T-cell dyscrasia and an atypical immune response to an infection or vaccination.3,4
The differential diagnosis of PLEVA includes varicella, pityriasis lichenoides chronica (PLC), lymphomatoid papulosis (LyP), disseminated herpes simplex infection, Gianotti-Crosti syndrome, and Langerhans cell histiocytosis.
Patients with varicella also present with lesions in different stages, similar to PLEVA, but the classic lesions are usually vesicular and described as dewdrops on a rose petal. The course of varicella is 1-2 weeks, compared with PLEVA where the lesions can be present for months to years.
Patients with PLC can have similar lesions to PLEVA, but the lesions rarely are necrotic. Some consider these two entities a spectrum of the same condition.5
LyP is a rare condition in children, and it is characterized by crops of pink papules and nodules that resolve within weeks. A skin biopsy may help distinguish between the two conditions because LyP lesions are characterized by atypical lymphocytes that are CD30 positive.
Children with Gianotti-Crosti syndrome present with papules and papulovesicles on the face, arms, buttocks, and legs, after an upper respiratory or GI infection. Sometimes the lesions may be hemorrhagic. Lesions resolve within weeks to months.
Hemorrhagic-crusted papules on a seborrheic and intertriginous distribution characterize Langerhans cell histiocytosis. These patients may present hepatosplenomegaly and lymphadenopathy – neither of which were present on our patient.
Children with mild PLEVA disease and who are not symptomatic may be followed without intervention. In those with more severe disease and who are symptomatic can be treated with tetracyclines such as minocycline or doxycycline or erythromycin for about 3 months.6,7 Phototherapy also is recommended as a first-line therapy. In cases that do not respond to oral antibiotics and light therapy, methotrexate can be an alternative.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Email her at pdnews@mdedge.com.
References
1. J Drugs Dermatol. 2019 Jul 1;18(7):690-1.
2. Pediatr Dermatol. 1991 Mar;8(1):51-7.
3. Arch Dermatol. 2000 Dec;136(12):1483-6.
4. Actas Dermosifiliogr. 2018 Sep;109(7):e6-10.
5. Pediatr Dermatol. 2018 Mar;35(2):213-9.
6. Pediatr Dermatol. 2012 Nov-Dec;29(6):719-24.
7. J Eur Acad Dermatol Venereol. 2019 Jul 18. doi: 10.1111/jdv.15813.
A skin biopsy of one of the lesions was consistent with pityriasis lichenoides et varioliformis acuta (PLEVA).
The patient was diagnosed with PLEVA, also known as Mucha-Habermann disease. The true incidence of the condition is not known.
The typical presentation is an abrupt onset of pink to violaceous, scaly papules and plaques that later develop violaceous or necrotic centers, like the ones seen in our patient. The lesions more typically occur on the trunk and proximal extremities, but they may present in any other part of the body, rarely in the mucosa.1 Some patients can develop the febrile, more severe form of PLEVA called febrile, ulceronecrotic Mucha-Habermann disease (FUMHD), which potentially can be life threatening.
Patients with PLEVA can complain of pruritus or a burning sensation, and in some cases can have associated arthralgia and edema. The more severe form FUMHD is characterized by persistent high fevers with associated internal organ involvement such as cardiomyopathy, small vessel vasculitis, abdominal pain, arthritis, pneumonitis, and hematologic abnormalities.2 Mucosal involvement is a common finding in patients with FUMHD.
The pathogenesis of PLEVA is not very well understood. Some theories include a T-cell dyscrasia and an atypical immune response to an infection or vaccination.3,4
The differential diagnosis of PLEVA includes varicella, pityriasis lichenoides chronica (PLC), lymphomatoid papulosis (LyP), disseminated herpes simplex infection, Gianotti-Crosti syndrome, and Langerhans cell histiocytosis.
Patients with varicella also present with lesions in different stages, similar to PLEVA, but the classic lesions are usually vesicular and described as dewdrops on a rose petal. The course of varicella is 1-2 weeks, compared with PLEVA where the lesions can be present for months to years.
Patients with PLC can have similar lesions to PLEVA, but the lesions rarely are necrotic. Some consider these two entities a spectrum of the same condition.5
LyP is a rare condition in children, and it is characterized by crops of pink papules and nodules that resolve within weeks. A skin biopsy may help distinguish between the two conditions because LyP lesions are characterized by atypical lymphocytes that are CD30 positive.
Children with Gianotti-Crosti syndrome present with papules and papulovesicles on the face, arms, buttocks, and legs, after an upper respiratory or GI infection. Sometimes the lesions may be hemorrhagic. Lesions resolve within weeks to months.
Hemorrhagic-crusted papules on a seborrheic and intertriginous distribution characterize Langerhans cell histiocytosis. These patients may present hepatosplenomegaly and lymphadenopathy – neither of which were present on our patient.
Children with mild PLEVA disease and who are not symptomatic may be followed without intervention. In those with more severe disease and who are symptomatic can be treated with tetracyclines such as minocycline or doxycycline or erythromycin for about 3 months.6,7 Phototherapy also is recommended as a first-line therapy. In cases that do not respond to oral antibiotics and light therapy, methotrexate can be an alternative.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Email her at pdnews@mdedge.com.
References
1. J Drugs Dermatol. 2019 Jul 1;18(7):690-1.
2. Pediatr Dermatol. 1991 Mar;8(1):51-7.
3. Arch Dermatol. 2000 Dec;136(12):1483-6.
4. Actas Dermosifiliogr. 2018 Sep;109(7):e6-10.
5. Pediatr Dermatol. 2018 Mar;35(2):213-9.
6. Pediatr Dermatol. 2012 Nov-Dec;29(6):719-24.
7. J Eur Acad Dermatol Venereol. 2019 Jul 18. doi: 10.1111/jdv.15813.
A skin biopsy of one of the lesions was consistent with pityriasis lichenoides et varioliformis acuta (PLEVA).
The patient was diagnosed with PLEVA, also known as Mucha-Habermann disease. The true incidence of the condition is not known.
The typical presentation is an abrupt onset of pink to violaceous, scaly papules and plaques that later develop violaceous or necrotic centers, like the ones seen in our patient. The lesions more typically occur on the trunk and proximal extremities, but they may present in any other part of the body, rarely in the mucosa.1 Some patients can develop the febrile, more severe form of PLEVA called febrile, ulceronecrotic Mucha-Habermann disease (FUMHD), which potentially can be life threatening.
Patients with PLEVA can complain of pruritus or a burning sensation, and in some cases can have associated arthralgia and edema. The more severe form FUMHD is characterized by persistent high fevers with associated internal organ involvement such as cardiomyopathy, small vessel vasculitis, abdominal pain, arthritis, pneumonitis, and hematologic abnormalities.2 Mucosal involvement is a common finding in patients with FUMHD.
The pathogenesis of PLEVA is not very well understood. Some theories include a T-cell dyscrasia and an atypical immune response to an infection or vaccination.3,4
The differential diagnosis of PLEVA includes varicella, pityriasis lichenoides chronica (PLC), lymphomatoid papulosis (LyP), disseminated herpes simplex infection, Gianotti-Crosti syndrome, and Langerhans cell histiocytosis.
Patients with varicella also present with lesions in different stages, similar to PLEVA, but the classic lesions are usually vesicular and described as dewdrops on a rose petal. The course of varicella is 1-2 weeks, compared with PLEVA where the lesions can be present for months to years.
Patients with PLC can have similar lesions to PLEVA, but the lesions rarely are necrotic. Some consider these two entities a spectrum of the same condition.5
LyP is a rare condition in children, and it is characterized by crops of pink papules and nodules that resolve within weeks. A skin biopsy may help distinguish between the two conditions because LyP lesions are characterized by atypical lymphocytes that are CD30 positive.
Children with Gianotti-Crosti syndrome present with papules and papulovesicles on the face, arms, buttocks, and legs, after an upper respiratory or GI infection. Sometimes the lesions may be hemorrhagic. Lesions resolve within weeks to months.
Hemorrhagic-crusted papules on a seborrheic and intertriginous distribution characterize Langerhans cell histiocytosis. These patients may present hepatosplenomegaly and lymphadenopathy – neither of which were present on our patient.
Children with mild PLEVA disease and who are not symptomatic may be followed without intervention. In those with more severe disease and who are symptomatic can be treated with tetracyclines such as minocycline or doxycycline or erythromycin for about 3 months.6,7 Phototherapy also is recommended as a first-line therapy. In cases that do not respond to oral antibiotics and light therapy, methotrexate can be an alternative.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Email her at pdnews@mdedge.com.
References
1. J Drugs Dermatol. 2019 Jul 1;18(7):690-1.
2. Pediatr Dermatol. 1991 Mar;8(1):51-7.
3. Arch Dermatol. 2000 Dec;136(12):1483-6.
4. Actas Dermosifiliogr. 2018 Sep;109(7):e6-10.
5. Pediatr Dermatol. 2018 Mar;35(2):213-9.
6. Pediatr Dermatol. 2012 Nov-Dec;29(6):719-24.
7. J Eur Acad Dermatol Venereol. 2019 Jul 18. doi: 10.1111/jdv.15813.
A healthy 14-year-old female was referred urgently by her pediatrician to our pediatric dermatology clinic for evaluation of a rash. The rash had been present for 4 weeks on her torso and proximal extremities, and had been spreading. She had been very itchy. She denied any fevers, chills, joint pain, oral or genital lesions.
She was visiting some family members in Washington State during the summer. The rash started 1 month after this visit.
The adolescent had been treated with acyclovir, trimethoprim/sulfamethoxazole, and intramuscular triamcinolone without improvement. She had been taking children's multivitamins occasionally. Her vaccinations were up-to-date. She denied any history of varicella or herpes infection. Her mom has a history of cold sores. The teen is not sexually active.
On physical examination, the girl was not in acute distress. Her vital signs were stable. She was not febrile. She had pink, scaly, and hyperpigmented papules and plaques, some of which were crusted with violaceous centers on the trunk and proximal extremities. There were no lesions on the mouth, palms, or soles. She had no lymphadenopathy or hepatosplenomegaly.
Short-term statin use linked to risk of skin and soft tissue infections
according to a sequence symmetry analysis of prescription claims over a 10-year period reported in the British Journal of Clinical Pharmacology.
In the study, statin use for as little as 91 days was linked with elevated risks of SSTIs and diabetes. However, the increased risk of infection was seen in individuals who did and did not develop diabetes, wrote Humphrey Ko, of the school of pharmacy and biomedical sciences, Curtin University, Perth, Australia, and colleagues.
The current literature on the impact of statins on SSTIs is conflicted, they noted. Previous research shows that statins “may reduce the risk of community-acquired [Staphylococcus aureus] bacteremia and exert antibacterial effects against S. aureus,” and therefore may have potential for reducing SSTI risk “or evolve into promising novel treatments for SSTIs,” the researchers said; they noted, however, that other data show that statins may induce new-onset diabetes.
They examined prescription claims (for statins, antidiabetic medications, and antistaphylococcal antibiotics) from 2001 to 2011 from the Australian Department of Veterans’ Affairs that included more than 228,000 veterans, war widows, and widowers. Prescriptions for antistaphylococcal antibiotics were used as a marker of SSTIs.
Overall, statins were significantly associated with an increased risk of SSTIs at 91 days (adjusted sequence ratio, 1.40). The risk of SSTIs from statin use was similar at 182 (ASR, 1.41) and 365 days (ASR, 1.40). In this case, the ASRs represent the incidence rate ratios of prescribing antibiotics in statin-exposed versus statin-nonexposed person-time.
Statins were associated with a significantly increased risk of new onset diabetes, but the SSTI risk was not significantly different between statin users with and without diabetes. Statin users who did not have diabetes had significant SSTI risks at 91, 182, and 365 days (ASR, 1.39, 1.41, and 1.37, respectively) and statin users with diabetes had similarly significant risks of SSTIs (ASR,1.43, 1.42, and 1.49, respectively).
In addition, socioeconomic status appeared to have no significant effect on the relationship between statin use, SSTIs, and diabetes, the researchers noted.
The findings were limited by several factors including the inability to account for patient compliance in taking the medications, a lack of dosage data to determine the impact of dosage on outcomes, and potential confounding by the presence of diabetes, they said. However, the results suggest that “it would seem prudent for clinicians to monitor blood glucose levels of statin users who are predisposed to diabetes, and be mindful of possible increased SSTI risks in such patients,” they concluded. Statins, they added, “may increase SSTI risk via direct or indirect mechanisms.”
More clinical trials are needed to confirm the mechanisms, and “to ascertain the effect of statins on gut dysbiosis, impaired bile acid metabolism, vitamin D levels, and cholesterol inhibition on skin function,” they wrote.
The study was supported in part by the Australian Government Research Training Program Scholarship, the Curtin Health Innovation Research Institute Biosciences Research Precinct Core Facility, and the School of Pharmacy and Biomedical Sciences (Curtin University). The researchers had no financial conflicts to disclose.
SOURCE: Ko H et al. Br J Clin Pharmacol. 2019 Oct 9. doi: 10.1111/bcp.14077.
according to a sequence symmetry analysis of prescription claims over a 10-year period reported in the British Journal of Clinical Pharmacology.
In the study, statin use for as little as 91 days was linked with elevated risks of SSTIs and diabetes. However, the increased risk of infection was seen in individuals who did and did not develop diabetes, wrote Humphrey Ko, of the school of pharmacy and biomedical sciences, Curtin University, Perth, Australia, and colleagues.
The current literature on the impact of statins on SSTIs is conflicted, they noted. Previous research shows that statins “may reduce the risk of community-acquired [Staphylococcus aureus] bacteremia and exert antibacterial effects against S. aureus,” and therefore may have potential for reducing SSTI risk “or evolve into promising novel treatments for SSTIs,” the researchers said; they noted, however, that other data show that statins may induce new-onset diabetes.
They examined prescription claims (for statins, antidiabetic medications, and antistaphylococcal antibiotics) from 2001 to 2011 from the Australian Department of Veterans’ Affairs that included more than 228,000 veterans, war widows, and widowers. Prescriptions for antistaphylococcal antibiotics were used as a marker of SSTIs.
Overall, statins were significantly associated with an increased risk of SSTIs at 91 days (adjusted sequence ratio, 1.40). The risk of SSTIs from statin use was similar at 182 (ASR, 1.41) and 365 days (ASR, 1.40). In this case, the ASRs represent the incidence rate ratios of prescribing antibiotics in statin-exposed versus statin-nonexposed person-time.
Statins were associated with a significantly increased risk of new onset diabetes, but the SSTI risk was not significantly different between statin users with and without diabetes. Statin users who did not have diabetes had significant SSTI risks at 91, 182, and 365 days (ASR, 1.39, 1.41, and 1.37, respectively) and statin users with diabetes had similarly significant risks of SSTIs (ASR,1.43, 1.42, and 1.49, respectively).
In addition, socioeconomic status appeared to have no significant effect on the relationship between statin use, SSTIs, and diabetes, the researchers noted.
The findings were limited by several factors including the inability to account for patient compliance in taking the medications, a lack of dosage data to determine the impact of dosage on outcomes, and potential confounding by the presence of diabetes, they said. However, the results suggest that “it would seem prudent for clinicians to monitor blood glucose levels of statin users who are predisposed to diabetes, and be mindful of possible increased SSTI risks in such patients,” they concluded. Statins, they added, “may increase SSTI risk via direct or indirect mechanisms.”
More clinical trials are needed to confirm the mechanisms, and “to ascertain the effect of statins on gut dysbiosis, impaired bile acid metabolism, vitamin D levels, and cholesterol inhibition on skin function,” they wrote.
The study was supported in part by the Australian Government Research Training Program Scholarship, the Curtin Health Innovation Research Institute Biosciences Research Precinct Core Facility, and the School of Pharmacy and Biomedical Sciences (Curtin University). The researchers had no financial conflicts to disclose.
SOURCE: Ko H et al. Br J Clin Pharmacol. 2019 Oct 9. doi: 10.1111/bcp.14077.
according to a sequence symmetry analysis of prescription claims over a 10-year period reported in the British Journal of Clinical Pharmacology.
In the study, statin use for as little as 91 days was linked with elevated risks of SSTIs and diabetes. However, the increased risk of infection was seen in individuals who did and did not develop diabetes, wrote Humphrey Ko, of the school of pharmacy and biomedical sciences, Curtin University, Perth, Australia, and colleagues.
The current literature on the impact of statins on SSTIs is conflicted, they noted. Previous research shows that statins “may reduce the risk of community-acquired [Staphylococcus aureus] bacteremia and exert antibacterial effects against S. aureus,” and therefore may have potential for reducing SSTI risk “or evolve into promising novel treatments for SSTIs,” the researchers said; they noted, however, that other data show that statins may induce new-onset diabetes.
They examined prescription claims (for statins, antidiabetic medications, and antistaphylococcal antibiotics) from 2001 to 2011 from the Australian Department of Veterans’ Affairs that included more than 228,000 veterans, war widows, and widowers. Prescriptions for antistaphylococcal antibiotics were used as a marker of SSTIs.
Overall, statins were significantly associated with an increased risk of SSTIs at 91 days (adjusted sequence ratio, 1.40). The risk of SSTIs from statin use was similar at 182 (ASR, 1.41) and 365 days (ASR, 1.40). In this case, the ASRs represent the incidence rate ratios of prescribing antibiotics in statin-exposed versus statin-nonexposed person-time.
Statins were associated with a significantly increased risk of new onset diabetes, but the SSTI risk was not significantly different between statin users with and without diabetes. Statin users who did not have diabetes had significant SSTI risks at 91, 182, and 365 days (ASR, 1.39, 1.41, and 1.37, respectively) and statin users with diabetes had similarly significant risks of SSTIs (ASR,1.43, 1.42, and 1.49, respectively).
In addition, socioeconomic status appeared to have no significant effect on the relationship between statin use, SSTIs, and diabetes, the researchers noted.
The findings were limited by several factors including the inability to account for patient compliance in taking the medications, a lack of dosage data to determine the impact of dosage on outcomes, and potential confounding by the presence of diabetes, they said. However, the results suggest that “it would seem prudent for clinicians to monitor blood glucose levels of statin users who are predisposed to diabetes, and be mindful of possible increased SSTI risks in such patients,” they concluded. Statins, they added, “may increase SSTI risk via direct or indirect mechanisms.”
More clinical trials are needed to confirm the mechanisms, and “to ascertain the effect of statins on gut dysbiosis, impaired bile acid metabolism, vitamin D levels, and cholesterol inhibition on skin function,” they wrote.
The study was supported in part by the Australian Government Research Training Program Scholarship, the Curtin Health Innovation Research Institute Biosciences Research Precinct Core Facility, and the School of Pharmacy and Biomedical Sciences (Curtin University). The researchers had no financial conflicts to disclose.
SOURCE: Ko H et al. Br J Clin Pharmacol. 2019 Oct 9. doi: 10.1111/bcp.14077.
FROM THE BRITISH JOURNAL OF CLINICAL PHARMACOLOGY
Tape strips useful to identify biomarkers in skin of young children with atopic dermatitis
Adhesive according to a study published online on October 9 in JAMA Dermatology.
“Minimally invasive approaches that accurately capture key immune and barrier biomarkers in the skin of patients with early-onset pediatric AD are needed,” wrote Emma Guttman-Yassky, MD, professor of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and coauthors. “Because tissue biopsies are considered the criterion standard for evaluating dysregulation in AD lesional and nonlesional skin, it is crucial to understand whether tape-strip profiling can accurately yield key AD-related biomarkers.”
In their cross-sectional study, researchers used large D-Squame tape strips to collect skin samples from 51 children under the age of 5 years (mean, 1.7-1.8 years), including 21 with moderate to severe AD and 30 controls who did not have AD. Samples were collected from lesional skin inside the crook of the elbow and nonlesional skin, on the same arm, then subjected to gene- and protein-expression analysis to identify skin biomarkers of disease.
The participants tolerated the tape stripping well, and there were no clinical effects of the procedure. The authors were able to detect mRNA in 70 of 71 samples.
They then analyzed a panel of 15 cellular markers that assessed markers of monocytes and macrophages, T cells, activated TH2 cells, dendritic cells and dendritic-cell subsets, and Langerhans cells. They found that most showed significant differences between lesional AD skin and normal skin.
They also found that levels of OX40 ligand receptor, a marker associated with atopic dendritic cells, the inducible T-cell costimulatory activation marker, CD209, CD123, and langerin protein, were also significantly higher in nonlesional AD skin.
When comparing lesional and nonlesional skin samples in the AD patients, the authors saw significant differences only in levels of colony-stimulating factor 1 and 2.
The authors noted that some of the mediators detected from the tape-strip samples had not been detected or evaluated in previous studies of the use of tape strips in AD. These included measures of cellular infiltrates, atopic dendritic cells, and key inflammatory markers.
“The novel epidermal cytokines IL [interleukin]–33 and IL-17C, which are currently targeted in clinical trials of patients with AD, were also highlighted as novel tape-strip biomarkers and demonstrated significant correlations with AD severity,” they wrote.
“Because tape stripping is painless, nonscarring, and allows repeated sampling, it may be associated with benefits for longitudinal pediatric studies and clinical trials, in which serial measures are needed to identify predictors of response, course, and comorbidities,” the authors concluded.
The study was supported by the Northwestern University Skin Disease Research Center and the Northwestern University Clinical and Translational Sciences Institute, and partly by a grant to two authors from Regeneron and Sanofi. Dr. Guttman-Yassky reported receiving grants from Regeneron during the study, and had other disclosures related to multiple pharmaceutical companies. Another author also received grants from Regeneron during the study, and another author had disclosures related to various manufacturers; no disclosures were reported for the remaining authors.
SOURCE: Guttman-Yassky E et al. JAMA Dermatol. 2019 Oct 9. doi: 10.1001/jamadermatol.2019.2983.
Skin biomarkers of atopic dermatitis (AD) are not well studied in children despite the fact that the disease largely affects this age group. Part of the challenge is the difficulty obtaining samples from children because phlebotomy and skin biopsies can cause trauma and anxiety both in children and their guardians. Better, noninvasive sampling techniques are needed.
This and another recent study show that tape stripping achieves skin samples that can provide clinically relevant AD DNA-expression levels and biomarkers that have been shown in multiple other studies – including some AD biomarkers not previously reported. Importantly, these biomarkers distinguish between children with AD and those without, and even between lesional and nonlesional skin.
While it remains to be seen if these biomarkers can predict disease outcomes or response to medication, this study shows that tape stripping in children with AD is a viable and useful method for future studies.
Leslie Castelo-Soccio, MD, PhD, is with the department of dermatology at the Children’s Hospital of Philadelphia. These comments are adapted from an accompanying editorial (JAMA Dermatol. 2019 Oct 9. doi: 10.1001/jamadermatol.2019.2792). No conflicts of interest were reported.
Skin biomarkers of atopic dermatitis (AD) are not well studied in children despite the fact that the disease largely affects this age group. Part of the challenge is the difficulty obtaining samples from children because phlebotomy and skin biopsies can cause trauma and anxiety both in children and their guardians. Better, noninvasive sampling techniques are needed.
This and another recent study show that tape stripping achieves skin samples that can provide clinically relevant AD DNA-expression levels and biomarkers that have been shown in multiple other studies – including some AD biomarkers not previously reported. Importantly, these biomarkers distinguish between children with AD and those without, and even between lesional and nonlesional skin.
While it remains to be seen if these biomarkers can predict disease outcomes or response to medication, this study shows that tape stripping in children with AD is a viable and useful method for future studies.
Leslie Castelo-Soccio, MD, PhD, is with the department of dermatology at the Children’s Hospital of Philadelphia. These comments are adapted from an accompanying editorial (JAMA Dermatol. 2019 Oct 9. doi: 10.1001/jamadermatol.2019.2792). No conflicts of interest were reported.
Skin biomarkers of atopic dermatitis (AD) are not well studied in children despite the fact that the disease largely affects this age group. Part of the challenge is the difficulty obtaining samples from children because phlebotomy and skin biopsies can cause trauma and anxiety both in children and their guardians. Better, noninvasive sampling techniques are needed.
This and another recent study show that tape stripping achieves skin samples that can provide clinically relevant AD DNA-expression levels and biomarkers that have been shown in multiple other studies – including some AD biomarkers not previously reported. Importantly, these biomarkers distinguish between children with AD and those without, and even between lesional and nonlesional skin.
While it remains to be seen if these biomarkers can predict disease outcomes or response to medication, this study shows that tape stripping in children with AD is a viable and useful method for future studies.
Leslie Castelo-Soccio, MD, PhD, is with the department of dermatology at the Children’s Hospital of Philadelphia. These comments are adapted from an accompanying editorial (JAMA Dermatol. 2019 Oct 9. doi: 10.1001/jamadermatol.2019.2792). No conflicts of interest were reported.
Adhesive according to a study published online on October 9 in JAMA Dermatology.
“Minimally invasive approaches that accurately capture key immune and barrier biomarkers in the skin of patients with early-onset pediatric AD are needed,” wrote Emma Guttman-Yassky, MD, professor of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and coauthors. “Because tissue biopsies are considered the criterion standard for evaluating dysregulation in AD lesional and nonlesional skin, it is crucial to understand whether tape-strip profiling can accurately yield key AD-related biomarkers.”
In their cross-sectional study, researchers used large D-Squame tape strips to collect skin samples from 51 children under the age of 5 years (mean, 1.7-1.8 years), including 21 with moderate to severe AD and 30 controls who did not have AD. Samples were collected from lesional skin inside the crook of the elbow and nonlesional skin, on the same arm, then subjected to gene- and protein-expression analysis to identify skin biomarkers of disease.
The participants tolerated the tape stripping well, and there were no clinical effects of the procedure. The authors were able to detect mRNA in 70 of 71 samples.
They then analyzed a panel of 15 cellular markers that assessed markers of monocytes and macrophages, T cells, activated TH2 cells, dendritic cells and dendritic-cell subsets, and Langerhans cells. They found that most showed significant differences between lesional AD skin and normal skin.
They also found that levels of OX40 ligand receptor, a marker associated with atopic dendritic cells, the inducible T-cell costimulatory activation marker, CD209, CD123, and langerin protein, were also significantly higher in nonlesional AD skin.
When comparing lesional and nonlesional skin samples in the AD patients, the authors saw significant differences only in levels of colony-stimulating factor 1 and 2.
The authors noted that some of the mediators detected from the tape-strip samples had not been detected or evaluated in previous studies of the use of tape strips in AD. These included measures of cellular infiltrates, atopic dendritic cells, and key inflammatory markers.
“The novel epidermal cytokines IL [interleukin]–33 and IL-17C, which are currently targeted in clinical trials of patients with AD, were also highlighted as novel tape-strip biomarkers and demonstrated significant correlations with AD severity,” they wrote.
“Because tape stripping is painless, nonscarring, and allows repeated sampling, it may be associated with benefits for longitudinal pediatric studies and clinical trials, in which serial measures are needed to identify predictors of response, course, and comorbidities,” the authors concluded.
The study was supported by the Northwestern University Skin Disease Research Center and the Northwestern University Clinical and Translational Sciences Institute, and partly by a grant to two authors from Regeneron and Sanofi. Dr. Guttman-Yassky reported receiving grants from Regeneron during the study, and had other disclosures related to multiple pharmaceutical companies. Another author also received grants from Regeneron during the study, and another author had disclosures related to various manufacturers; no disclosures were reported for the remaining authors.
SOURCE: Guttman-Yassky E et al. JAMA Dermatol. 2019 Oct 9. doi: 10.1001/jamadermatol.2019.2983.
Adhesive according to a study published online on October 9 in JAMA Dermatology.
“Minimally invasive approaches that accurately capture key immune and barrier biomarkers in the skin of patients with early-onset pediatric AD are needed,” wrote Emma Guttman-Yassky, MD, professor of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and coauthors. “Because tissue biopsies are considered the criterion standard for evaluating dysregulation in AD lesional and nonlesional skin, it is crucial to understand whether tape-strip profiling can accurately yield key AD-related biomarkers.”
In their cross-sectional study, researchers used large D-Squame tape strips to collect skin samples from 51 children under the age of 5 years (mean, 1.7-1.8 years), including 21 with moderate to severe AD and 30 controls who did not have AD. Samples were collected from lesional skin inside the crook of the elbow and nonlesional skin, on the same arm, then subjected to gene- and protein-expression analysis to identify skin biomarkers of disease.
The participants tolerated the tape stripping well, and there were no clinical effects of the procedure. The authors were able to detect mRNA in 70 of 71 samples.
They then analyzed a panel of 15 cellular markers that assessed markers of monocytes and macrophages, T cells, activated TH2 cells, dendritic cells and dendritic-cell subsets, and Langerhans cells. They found that most showed significant differences between lesional AD skin and normal skin.
They also found that levels of OX40 ligand receptor, a marker associated with atopic dendritic cells, the inducible T-cell costimulatory activation marker, CD209, CD123, and langerin protein, were also significantly higher in nonlesional AD skin.
When comparing lesional and nonlesional skin samples in the AD patients, the authors saw significant differences only in levels of colony-stimulating factor 1 and 2.
The authors noted that some of the mediators detected from the tape-strip samples had not been detected or evaluated in previous studies of the use of tape strips in AD. These included measures of cellular infiltrates, atopic dendritic cells, and key inflammatory markers.
“The novel epidermal cytokines IL [interleukin]–33 and IL-17C, which are currently targeted in clinical trials of patients with AD, were also highlighted as novel tape-strip biomarkers and demonstrated significant correlations with AD severity,” they wrote.
“Because tape stripping is painless, nonscarring, and allows repeated sampling, it may be associated with benefits for longitudinal pediatric studies and clinical trials, in which serial measures are needed to identify predictors of response, course, and comorbidities,” the authors concluded.
The study was supported by the Northwestern University Skin Disease Research Center and the Northwestern University Clinical and Translational Sciences Institute, and partly by a grant to two authors from Regeneron and Sanofi. Dr. Guttman-Yassky reported receiving grants from Regeneron during the study, and had other disclosures related to multiple pharmaceutical companies. Another author also received grants from Regeneron during the study, and another author had disclosures related to various manufacturers; no disclosures were reported for the remaining authors.
SOURCE: Guttman-Yassky E et al. JAMA Dermatol. 2019 Oct 9. doi: 10.1001/jamadermatol.2019.2983.
FROM JAMA DERMATOLOGY
Start From Scratch
A 50-year-old man presents with complaints of a rash on his right leg that manifested 20 years ago. Although the rash is worrisome, he says the associated pruritus is worse. During the workday, he is able to ignore the itching—but the minute he gets home, he begins to scratch.
He knows the scratching is counterproductive in the long run, but the urge to do it is quite compelling. Sometimes he uses a wet washcloth; other times, he will actually use a hair brush to satiate the itching. The relief is intensely satisfying albeit short lived.
The rash has persisted despite multiple treatment attempts. Tried products include OTC moisturizers and antifungal creams, as well as prescription antifungal creams. None has had an effect.
The patient denies any other skin problems. He does recall having eczema as a child. Although that has long since resolved, he remains quite allergy prone and is particularly sensitive to airborne allergens—a trait that runs strongly in his family.
EXAMINATION
A pink, oval rash covers most of the patient’s right lateral calf. It has a thickened, faintly scaly surface that is uniform and sharply circumscribed. There is no increased warmth or tenderness on palpation. No lymph nodes can be felt in the right groin. A check of the patient’s knees, elbows, scalp, nails, and trunk show no sign of rash or other changes.
What’s the diagnosis?
DISCUSSION
Lichen simplex chronicus (LSC), previously known as neurodermatitis, is quite common but frequently misdiagnosed. Patients often report that their condition started with a bug bite or poison ivy—a provocation that gets the patient in the habit of scratching, which continues long after the initial outbreak has subsided. Thus, LSC is often associated with significant chronicity, as typified by this case.
What patients seldom understand is their own role in the perpetuation of their condition. The urge to scratch is so unbearable that few can resist it. Over time, the scratching creates more nerves that have a lower threshold for itching, and thus the itch-scratch-itch cycle is born. Many LSC patients are atopic, which predisposes them to itching in general and to xerosis especially.
The literature asserts that LSC affects the genders equally, but this ignores the fact that it can present significantly differently in men and women. This patient’s area of involvement is quite typical for men, most of whom never moisturize and for whom the lateral calf is readily accessible. In the author’s experience, the most common location for LSC in women is the nuchal scalp, where heat and sweat appear to play a role, along with ready accessibility to fingernails or the sharp end of a pencil. Other common areas of involvement include the dorsal forearms and the scrotum or vulvae.
Biopsy is seldom necessary, but if performed, it will show a marked thickening of the epidermis, orthokeratosis (normal keratinocytes about to shed), and compacted, elongated rete ridges. These and other changes effectively rule out other items in the differential (eg, psoriasis, simple eczema, fungal infection).
Stopping the itch-scratch-itch cycle with mid-strength topical steroid creams or foams is the first step in treating LSC. But then the patient must be convinced of his contribution to the treatment: leaving the affected sites alone. Truth be told, after 20 years of scratching, the best this patient can look forward to is some relief—not only from the itching, but also from concern about all the terrible things he now knows he doesn’t have.
TAKE-HOME LEARNING POINTS
- Lichen simplex chronicus is quite common in both genders and is typified by longstanding severe itching, usually confined to one area.
- Atopy, xerosis, and stress all appear to contribute to the problem.
- Stopping the itch-scratch-itch cycle with topical steroids is a key component of treatment.
- Patient education—on the nature of the problem and the patient’s role in controlling it—is just as important as any prescribed medication.
A 50-year-old man presents with complaints of a rash on his right leg that manifested 20 years ago. Although the rash is worrisome, he says the associated pruritus is worse. During the workday, he is able to ignore the itching—but the minute he gets home, he begins to scratch.
He knows the scratching is counterproductive in the long run, but the urge to do it is quite compelling. Sometimes he uses a wet washcloth; other times, he will actually use a hair brush to satiate the itching. The relief is intensely satisfying albeit short lived.
The rash has persisted despite multiple treatment attempts. Tried products include OTC moisturizers and antifungal creams, as well as prescription antifungal creams. None has had an effect.
The patient denies any other skin problems. He does recall having eczema as a child. Although that has long since resolved, he remains quite allergy prone and is particularly sensitive to airborne allergens—a trait that runs strongly in his family.

EXAMINATION
A pink, oval rash covers most of the patient’s right lateral calf. It has a thickened, faintly scaly surface that is uniform and sharply circumscribed. There is no increased warmth or tenderness on palpation. No lymph nodes can be felt in the right groin. A check of the patient’s knees, elbows, scalp, nails, and trunk show no sign of rash or other changes.
What’s the diagnosis?
DISCUSSION
Lichen simplex chronicus (LSC), previously known as neurodermatitis, is quite common but frequently misdiagnosed. Patients often report that their condition started with a bug bite or poison ivy—a provocation that gets the patient in the habit of scratching, which continues long after the initial outbreak has subsided. Thus, LSC is often associated with significant chronicity, as typified by this case.
What patients seldom understand is their own role in the perpetuation of their condition. The urge to scratch is so unbearable that few can resist it. Over time, the scratching creates more nerves that have a lower threshold for itching, and thus the itch-scratch-itch cycle is born. Many LSC patients are atopic, which predisposes them to itching in general and to xerosis especially.
The literature asserts that LSC affects the genders equally, but this ignores the fact that it can present significantly differently in men and women. This patient’s area of involvement is quite typical for men, most of whom never moisturize and for whom the lateral calf is readily accessible. In the author’s experience, the most common location for LSC in women is the nuchal scalp, where heat and sweat appear to play a role, along with ready accessibility to fingernails or the sharp end of a pencil. Other common areas of involvement include the dorsal forearms and the scrotum or vulvae.
Biopsy is seldom necessary, but if performed, it will show a marked thickening of the epidermis, orthokeratosis (normal keratinocytes about to shed), and compacted, elongated rete ridges. These and other changes effectively rule out other items in the differential (eg, psoriasis, simple eczema, fungal infection).
Stopping the itch-scratch-itch cycle with mid-strength topical steroid creams or foams is the first step in treating LSC. But then the patient must be convinced of his contribution to the treatment: leaving the affected sites alone. Truth be told, after 20 years of scratching, the best this patient can look forward to is some relief—not only from the itching, but also from concern about all the terrible things he now knows he doesn’t have.
TAKE-HOME LEARNING POINTS
- Lichen simplex chronicus is quite common in both genders and is typified by longstanding severe itching, usually confined to one area.
- Atopy, xerosis, and stress all appear to contribute to the problem.
- Stopping the itch-scratch-itch cycle with topical steroids is a key component of treatment.
- Patient education—on the nature of the problem and the patient’s role in controlling it—is just as important as any prescribed medication.
A 50-year-old man presents with complaints of a rash on his right leg that manifested 20 years ago. Although the rash is worrisome, he says the associated pruritus is worse. During the workday, he is able to ignore the itching—but the minute he gets home, he begins to scratch.
He knows the scratching is counterproductive in the long run, but the urge to do it is quite compelling. Sometimes he uses a wet washcloth; other times, he will actually use a hair brush to satiate the itching. The relief is intensely satisfying albeit short lived.
The rash has persisted despite multiple treatment attempts. Tried products include OTC moisturizers and antifungal creams, as well as prescription antifungal creams. None has had an effect.
The patient denies any other skin problems. He does recall having eczema as a child. Although that has long since resolved, he remains quite allergy prone and is particularly sensitive to airborne allergens—a trait that runs strongly in his family.

EXAMINATION
A pink, oval rash covers most of the patient’s right lateral calf. It has a thickened, faintly scaly surface that is uniform and sharply circumscribed. There is no increased warmth or tenderness on palpation. No lymph nodes can be felt in the right groin. A check of the patient’s knees, elbows, scalp, nails, and trunk show no sign of rash or other changes.
What’s the diagnosis?
DISCUSSION
Lichen simplex chronicus (LSC), previously known as neurodermatitis, is quite common but frequently misdiagnosed. Patients often report that their condition started with a bug bite or poison ivy—a provocation that gets the patient in the habit of scratching, which continues long after the initial outbreak has subsided. Thus, LSC is often associated with significant chronicity, as typified by this case.
What patients seldom understand is their own role in the perpetuation of their condition. The urge to scratch is so unbearable that few can resist it. Over time, the scratching creates more nerves that have a lower threshold for itching, and thus the itch-scratch-itch cycle is born. Many LSC patients are atopic, which predisposes them to itching in general and to xerosis especially.
The literature asserts that LSC affects the genders equally, but this ignores the fact that it can present significantly differently in men and women. This patient’s area of involvement is quite typical for men, most of whom never moisturize and for whom the lateral calf is readily accessible. In the author’s experience, the most common location for LSC in women is the nuchal scalp, where heat and sweat appear to play a role, along with ready accessibility to fingernails or the sharp end of a pencil. Other common areas of involvement include the dorsal forearms and the scrotum or vulvae.
Biopsy is seldom necessary, but if performed, it will show a marked thickening of the epidermis, orthokeratosis (normal keratinocytes about to shed), and compacted, elongated rete ridges. These and other changes effectively rule out other items in the differential (eg, psoriasis, simple eczema, fungal infection).
Stopping the itch-scratch-itch cycle with mid-strength topical steroid creams or foams is the first step in treating LSC. But then the patient must be convinced of his contribution to the treatment: leaving the affected sites alone. Truth be told, after 20 years of scratching, the best this patient can look forward to is some relief—not only from the itching, but also from concern about all the terrible things he now knows he doesn’t have.
TAKE-HOME LEARNING POINTS
- Lichen simplex chronicus is quite common in both genders and is typified by longstanding severe itching, usually confined to one area.
- Atopy, xerosis, and stress all appear to contribute to the problem.
- Stopping the itch-scratch-itch cycle with topical steroids is a key component of treatment.
- Patient education—on the nature of the problem and the patient’s role in controlling it—is just as important as any prescribed medication.