User login
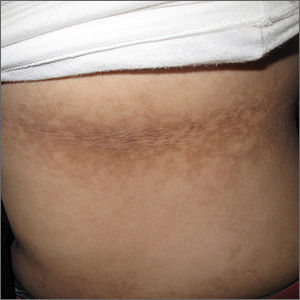
Dark patches around the trunk
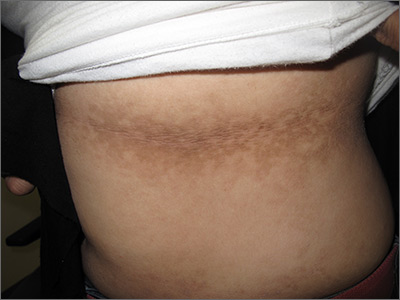
The FP noticed a lacy net-like or reticulate appearance and thin brown papules to warty plaques over the trunk and recognized this condition as confluent and reticulated papillomatosis (CARP). A potassium hydroxide (KOH) test of a skin scraping failed to reveal yeast forms or hyphae. The FP determined that a biopsy was not necessary for diagnosis due to the distinct clinical appearance and negative KOH test. However, a biopsy could have distinguished this presentation from similar appearing disorders, including acanthosis nigricans and pityriasis versicolor.
CARP is an uncommon disorder of keratinization that affects adolescents and young adults, and is more common in Caucasians. A classic presentation involves the neck, chest, and abdomen. The differential diagnosis includes acanthosis nigricans and pityriasis versicolor, as well as more rare disorders that include Darier disease and keratosis follicularis.
There appears to be an association between the disorder and weight (specifically, being overweight). In addition, some familial cases have been reported.
Most recently, Dietzia papillomatosis, a gram-positive actinomycete has been implicated as a likely cause, which supports antibiotic therapy as the first-line approach. Minocycline 50 mg bid for 6 weeks clears the papules and plaques for most patients. Azithromycin and clarithromycin are alternatives, with various dosing strategies lasting 6 to 12 weeks. Complete clearance may take months to more than a year. About 15% of patients will experience recurrence.
This patient was treated with minocycline 50 mg bid for 12 weeks, a more common strategy at the time she was diagnosed. This led to complete clearance at 3 months, and she remained clear a year after beginning treatment.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained).

The FP noticed a lacy net-like or reticulate appearance and thin brown papules to warty plaques over the trunk and recognized this condition as confluent and reticulated papillomatosis (CARP). A potassium hydroxide (KOH) test of a skin scraping failed to reveal yeast forms or hyphae. The FP determined that a biopsy was not necessary for diagnosis due to the distinct clinical appearance and negative KOH test. However, a biopsy could have distinguished this presentation from similar appearing disorders, including acanthosis nigricans and pityriasis versicolor.
CARP is an uncommon disorder of keratinization that affects adolescents and young adults, and is more common in Caucasians. A classic presentation involves the neck, chest, and abdomen. The differential diagnosis includes acanthosis nigricans and pityriasis versicolor, as well as more rare disorders that include Darier disease and keratosis follicularis.
There appears to be an association between the disorder and weight (specifically, being overweight). In addition, some familial cases have been reported.
Most recently, Dietzia papillomatosis, a gram-positive actinomycete has been implicated as a likely cause, which supports antibiotic therapy as the first-line approach. Minocycline 50 mg bid for 6 weeks clears the papules and plaques for most patients. Azithromycin and clarithromycin are alternatives, with various dosing strategies lasting 6 to 12 weeks. Complete clearance may take months to more than a year. About 15% of patients will experience recurrence.
This patient was treated with minocycline 50 mg bid for 12 weeks, a more common strategy at the time she was diagnosed. This led to complete clearance at 3 months, and she remained clear a year after beginning treatment.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained).

The FP noticed a lacy net-like or reticulate appearance and thin brown papules to warty plaques over the trunk and recognized this condition as confluent and reticulated papillomatosis (CARP). A potassium hydroxide (KOH) test of a skin scraping failed to reveal yeast forms or hyphae. The FP determined that a biopsy was not necessary for diagnosis due to the distinct clinical appearance and negative KOH test. However, a biopsy could have distinguished this presentation from similar appearing disorders, including acanthosis nigricans and pityriasis versicolor.
CARP is an uncommon disorder of keratinization that affects adolescents and young adults, and is more common in Caucasians. A classic presentation involves the neck, chest, and abdomen. The differential diagnosis includes acanthosis nigricans and pityriasis versicolor, as well as more rare disorders that include Darier disease and keratosis follicularis.
There appears to be an association between the disorder and weight (specifically, being overweight). In addition, some familial cases have been reported.
Most recently, Dietzia papillomatosis, a gram-positive actinomycete has been implicated as a likely cause, which supports antibiotic therapy as the first-line approach. Minocycline 50 mg bid for 6 weeks clears the papules and plaques for most patients. Azithromycin and clarithromycin are alternatives, with various dosing strategies lasting 6 to 12 weeks. Complete clearance may take months to more than a year. About 15% of patients will experience recurrence.
This patient was treated with minocycline 50 mg bid for 12 weeks, a more common strategy at the time she was diagnosed. This led to complete clearance at 3 months, and she remained clear a year after beginning treatment.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained).
Psoriasis risk rises with TNF inhibitor use in children with inflammatory disorders
, a retrospective cohort study has determined.
“The incidence rate and risk factors of psoriasis in children with IBD [inflammatory bowel disease], JIA [juvenile idiopathic arthritis], or CNO [chronic nonbacterial osteomyelitis] who are exposed to TNFi [tumor necrosis factor inhibitors] are unknown. Additionally, there is a well-established association between these inflammatory conditions and psoriasis development. Yet, as TNFi can both treat and trigger psoriasis, it is not clear how TNFi exposure affects this relationship,” wrote Lisa H. Buckley, MD, of Children’s Hospital at Vanderbilt, Nashville, Tenn., and colleagues. Their report is in Arthritis Care & Research.
The team examined the relationship in children who were treated for an inflammatory disorder at Children’s Hospital of Philadelphia during 2008-2018. IBD was most common at 74%, followed by JIA at 24% and CNO at 2%.
Among 4,111 children with those inflammatory disorders, the psoriasis incidence was 12.3 per 1,000 person-years in exposed children and 3.8 per 1,000 person-years in unexposed. This significant difference equated to a hazard ratio of 3.84 for developing psoriasis after TNFi exposure.
“These data reflect the established association between inflammatory conditions and psoriasis development and suggest that TNFi exposure further increases the risk of psoriasis,” Dr. Buckley and coauthors wrote.
The median duration of follow-up in this study was about 2.5 years for patients exposed to TNFi and 2 years for those unexposed. Among the entire cohort, 39% had been exposed to a TNFi, with 4,705 person-years of follow-up. Among the unexposed children (61%), there were 6,604 person-years of follow-up.
In all, 83 cases of psoriasis developed: 58 in the exposed group and 25 in the unexposed group. Psoriasis incidence varied by disorder. Exposed children with IBD had a higher incidence than did unexposed children (10.9 vs. 2.6 per 1,000 person-years; HR = 4.52). Exposed children with JIA also had a higher incidence than did unexposed children (14.7 vs. 5.5 per 1,000 person-years; HR = 2.90). Among those with CNO, incidences were similar for exposed and unexposed children (33.5 and 38.9 per 1,000 person-years).
A family history of psoriasis significantly increased the risk of psoriasis with a hazard ratio of 3.11, the authors noted. But none of the other covariates (age, sex, race, obesity, methotrexate exposure, and underlying diagnosis) exerted a significant additional risk.
The study had no outside funding source. The authors had no financial disclosures. Dr. Buckley conducted the research when she was a pediatric rheumatology fellow at Children’s Hospital of Philadelphia.
SOURCE: Buckley LH et al. Arthritis Care Res. 2019 Oct 23. doi: 10.1002/ACR.24100
, a retrospective cohort study has determined.
“The incidence rate and risk factors of psoriasis in children with IBD [inflammatory bowel disease], JIA [juvenile idiopathic arthritis], or CNO [chronic nonbacterial osteomyelitis] who are exposed to TNFi [tumor necrosis factor inhibitors] are unknown. Additionally, there is a well-established association between these inflammatory conditions and psoriasis development. Yet, as TNFi can both treat and trigger psoriasis, it is not clear how TNFi exposure affects this relationship,” wrote Lisa H. Buckley, MD, of Children’s Hospital at Vanderbilt, Nashville, Tenn., and colleagues. Their report is in Arthritis Care & Research.
The team examined the relationship in children who were treated for an inflammatory disorder at Children’s Hospital of Philadelphia during 2008-2018. IBD was most common at 74%, followed by JIA at 24% and CNO at 2%.
Among 4,111 children with those inflammatory disorders, the psoriasis incidence was 12.3 per 1,000 person-years in exposed children and 3.8 per 1,000 person-years in unexposed. This significant difference equated to a hazard ratio of 3.84 for developing psoriasis after TNFi exposure.
“These data reflect the established association between inflammatory conditions and psoriasis development and suggest that TNFi exposure further increases the risk of psoriasis,” Dr. Buckley and coauthors wrote.
The median duration of follow-up in this study was about 2.5 years for patients exposed to TNFi and 2 years for those unexposed. Among the entire cohort, 39% had been exposed to a TNFi, with 4,705 person-years of follow-up. Among the unexposed children (61%), there were 6,604 person-years of follow-up.
In all, 83 cases of psoriasis developed: 58 in the exposed group and 25 in the unexposed group. Psoriasis incidence varied by disorder. Exposed children with IBD had a higher incidence than did unexposed children (10.9 vs. 2.6 per 1,000 person-years; HR = 4.52). Exposed children with JIA also had a higher incidence than did unexposed children (14.7 vs. 5.5 per 1,000 person-years; HR = 2.90). Among those with CNO, incidences were similar for exposed and unexposed children (33.5 and 38.9 per 1,000 person-years).
A family history of psoriasis significantly increased the risk of psoriasis with a hazard ratio of 3.11, the authors noted. But none of the other covariates (age, sex, race, obesity, methotrexate exposure, and underlying diagnosis) exerted a significant additional risk.
The study had no outside funding source. The authors had no financial disclosures. Dr. Buckley conducted the research when she was a pediatric rheumatology fellow at Children’s Hospital of Philadelphia.
SOURCE: Buckley LH et al. Arthritis Care Res. 2019 Oct 23. doi: 10.1002/ACR.24100
, a retrospective cohort study has determined.
“The incidence rate and risk factors of psoriasis in children with IBD [inflammatory bowel disease], JIA [juvenile idiopathic arthritis], or CNO [chronic nonbacterial osteomyelitis] who are exposed to TNFi [tumor necrosis factor inhibitors] are unknown. Additionally, there is a well-established association between these inflammatory conditions and psoriasis development. Yet, as TNFi can both treat and trigger psoriasis, it is not clear how TNFi exposure affects this relationship,” wrote Lisa H. Buckley, MD, of Children’s Hospital at Vanderbilt, Nashville, Tenn., and colleagues. Their report is in Arthritis Care & Research.
The team examined the relationship in children who were treated for an inflammatory disorder at Children’s Hospital of Philadelphia during 2008-2018. IBD was most common at 74%, followed by JIA at 24% and CNO at 2%.
Among 4,111 children with those inflammatory disorders, the psoriasis incidence was 12.3 per 1,000 person-years in exposed children and 3.8 per 1,000 person-years in unexposed. This significant difference equated to a hazard ratio of 3.84 for developing psoriasis after TNFi exposure.
“These data reflect the established association between inflammatory conditions and psoriasis development and suggest that TNFi exposure further increases the risk of psoriasis,” Dr. Buckley and coauthors wrote.
The median duration of follow-up in this study was about 2.5 years for patients exposed to TNFi and 2 years for those unexposed. Among the entire cohort, 39% had been exposed to a TNFi, with 4,705 person-years of follow-up. Among the unexposed children (61%), there were 6,604 person-years of follow-up.
In all, 83 cases of psoriasis developed: 58 in the exposed group and 25 in the unexposed group. Psoriasis incidence varied by disorder. Exposed children with IBD had a higher incidence than did unexposed children (10.9 vs. 2.6 per 1,000 person-years; HR = 4.52). Exposed children with JIA also had a higher incidence than did unexposed children (14.7 vs. 5.5 per 1,000 person-years; HR = 2.90). Among those with CNO, incidences were similar for exposed and unexposed children (33.5 and 38.9 per 1,000 person-years).
A family history of psoriasis significantly increased the risk of psoriasis with a hazard ratio of 3.11, the authors noted. But none of the other covariates (age, sex, race, obesity, methotrexate exposure, and underlying diagnosis) exerted a significant additional risk.
The study had no outside funding source. The authors had no financial disclosures. Dr. Buckley conducted the research when she was a pediatric rheumatology fellow at Children’s Hospital of Philadelphia.
SOURCE: Buckley LH et al. Arthritis Care Res. 2019 Oct 23. doi: 10.1002/ACR.24100
FROM ARTHRITIS RESEARCH & CARE
No infection increase seen with biologics in older psoriasis patients
MADRID – Psoriasis patients aged 65 years and older are at more than twice the risk of serious bacterial and opportunistic infections, compared with younger patients, but that risk is not further elevated by being on biologic agents, Joseph F. Merola, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
He presented a large, The study implications, he said, are clear: When moderate to severe psoriasis warrants consideration of highly effective biologic therapies, that therapeutic option shouldn’t be taken off the table on the basis of a mistaken belief that biologics pose a greater infection risk just because the affected patient is over age 65 years.
“We really think that older patients should be offered treatments at the same level of disease control as all the rest of our psoriasis patients, in the context of shared decision making,” said Dr. Merola, a dermatologist and rheumatologist who is the director of the Center for Skin and Related Musculoskeletal Diseases at Brigham and Women’s Hospital, Boston.
The study utilized longitudinal claims data from a very large U.S. database covering the years 2003-2017. Among the 185 million covered lives were 1.1 million individuals with psoriasis, including 150,000 aged 65 years or older. After excluding older psoriasis patients with comorbid cancer or autoimmune disease, the investigators were left with 11,218 older psoriasis patients initiating systemic therapy for the first time and therefore eligible for propensity score matching using a highly accurate proprietary platform. The final study population consisted of 2,795 older psoriasis patients newly initiating biologic therapy, 2,795 others newly initiating nonbiologic systemic agents, and 2,529 seniors starting phototherapy. The matching was based upon factors including age, sex, prior infections, comorbid psoriatic arthritis, diabetes, and obesity.
The primary study endpoint was the rate of serious bacterial or opportunistic infections requiring hospitalization during the first 6 months of treatment. The bottom line: The rates were closely similar across all three groups, with the most common serious infections being pneumonia and cellulitis.
In contrast, among a population of 115,047 senior psoriasis patients who never used systemic therapy, the risk of serious infection was 12.2 events per 1,000 patients over 6 months, compared with 5.3 events in 120,174 matched controls without psoriasis. That translates to a 2.24-fold increased risk.
One audience member commented that a limitation of the study was that all biologics were lumped together. He would expect that the tumor necrosis factor inhibitors, for example, would be associated with a significantly higher serious infection risk than biologics with other targets.
Dr. Merola conceded the point, adding that the investigators are trying to reanalyze the data in a more granular way to address that shortcoming. Other study limitations included an inability to access the specific doses of systemic treatments used or to stratify patients by disease severity.
Another audience member noted that dermatologists often reassure surgeons that there’s no increased risk of infection associated with psoriasis when in fact there is increased risk in older psoriasis patients, according to these new data.
“We’re not trying to send a message to surgeons to withhold a knee transplant because of a psoriasis plaque over the knee,” Dr. Merola replied. “I think we’ve all been there; we’ve all fought that battle.” Based on the data, he said, he would advise that “our patients who need to be on systemics should remain appropriately on systemics as we see fit.”
The study was entirely funded by Brigham and Women’s Hospital. Dr. Merola reported serving as a consultant to and/or recipient of research grants from nearly two dozen pharmaceutical companies.
MADRID – Psoriasis patients aged 65 years and older are at more than twice the risk of serious bacterial and opportunistic infections, compared with younger patients, but that risk is not further elevated by being on biologic agents, Joseph F. Merola, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
He presented a large, The study implications, he said, are clear: When moderate to severe psoriasis warrants consideration of highly effective biologic therapies, that therapeutic option shouldn’t be taken off the table on the basis of a mistaken belief that biologics pose a greater infection risk just because the affected patient is over age 65 years.
“We really think that older patients should be offered treatments at the same level of disease control as all the rest of our psoriasis patients, in the context of shared decision making,” said Dr. Merola, a dermatologist and rheumatologist who is the director of the Center for Skin and Related Musculoskeletal Diseases at Brigham and Women’s Hospital, Boston.
The study utilized longitudinal claims data from a very large U.S. database covering the years 2003-2017. Among the 185 million covered lives were 1.1 million individuals with psoriasis, including 150,000 aged 65 years or older. After excluding older psoriasis patients with comorbid cancer or autoimmune disease, the investigators were left with 11,218 older psoriasis patients initiating systemic therapy for the first time and therefore eligible for propensity score matching using a highly accurate proprietary platform. The final study population consisted of 2,795 older psoriasis patients newly initiating biologic therapy, 2,795 others newly initiating nonbiologic systemic agents, and 2,529 seniors starting phototherapy. The matching was based upon factors including age, sex, prior infections, comorbid psoriatic arthritis, diabetes, and obesity.
The primary study endpoint was the rate of serious bacterial or opportunistic infections requiring hospitalization during the first 6 months of treatment. The bottom line: The rates were closely similar across all three groups, with the most common serious infections being pneumonia and cellulitis.
In contrast, among a population of 115,047 senior psoriasis patients who never used systemic therapy, the risk of serious infection was 12.2 events per 1,000 patients over 6 months, compared with 5.3 events in 120,174 matched controls without psoriasis. That translates to a 2.24-fold increased risk.
One audience member commented that a limitation of the study was that all biologics were lumped together. He would expect that the tumor necrosis factor inhibitors, for example, would be associated with a significantly higher serious infection risk than biologics with other targets.
Dr. Merola conceded the point, adding that the investigators are trying to reanalyze the data in a more granular way to address that shortcoming. Other study limitations included an inability to access the specific doses of systemic treatments used or to stratify patients by disease severity.
Another audience member noted that dermatologists often reassure surgeons that there’s no increased risk of infection associated with psoriasis when in fact there is increased risk in older psoriasis patients, according to these new data.
“We’re not trying to send a message to surgeons to withhold a knee transplant because of a psoriasis plaque over the knee,” Dr. Merola replied. “I think we’ve all been there; we’ve all fought that battle.” Based on the data, he said, he would advise that “our patients who need to be on systemics should remain appropriately on systemics as we see fit.”
The study was entirely funded by Brigham and Women’s Hospital. Dr. Merola reported serving as a consultant to and/or recipient of research grants from nearly two dozen pharmaceutical companies.
MADRID – Psoriasis patients aged 65 years and older are at more than twice the risk of serious bacterial and opportunistic infections, compared with younger patients, but that risk is not further elevated by being on biologic agents, Joseph F. Merola, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
He presented a large, The study implications, he said, are clear: When moderate to severe psoriasis warrants consideration of highly effective biologic therapies, that therapeutic option shouldn’t be taken off the table on the basis of a mistaken belief that biologics pose a greater infection risk just because the affected patient is over age 65 years.
“We really think that older patients should be offered treatments at the same level of disease control as all the rest of our psoriasis patients, in the context of shared decision making,” said Dr. Merola, a dermatologist and rheumatologist who is the director of the Center for Skin and Related Musculoskeletal Diseases at Brigham and Women’s Hospital, Boston.
The study utilized longitudinal claims data from a very large U.S. database covering the years 2003-2017. Among the 185 million covered lives were 1.1 million individuals with psoriasis, including 150,000 aged 65 years or older. After excluding older psoriasis patients with comorbid cancer or autoimmune disease, the investigators were left with 11,218 older psoriasis patients initiating systemic therapy for the first time and therefore eligible for propensity score matching using a highly accurate proprietary platform. The final study population consisted of 2,795 older psoriasis patients newly initiating biologic therapy, 2,795 others newly initiating nonbiologic systemic agents, and 2,529 seniors starting phototherapy. The matching was based upon factors including age, sex, prior infections, comorbid psoriatic arthritis, diabetes, and obesity.
The primary study endpoint was the rate of serious bacterial or opportunistic infections requiring hospitalization during the first 6 months of treatment. The bottom line: The rates were closely similar across all three groups, with the most common serious infections being pneumonia and cellulitis.
In contrast, among a population of 115,047 senior psoriasis patients who never used systemic therapy, the risk of serious infection was 12.2 events per 1,000 patients over 6 months, compared with 5.3 events in 120,174 matched controls without psoriasis. That translates to a 2.24-fold increased risk.
One audience member commented that a limitation of the study was that all biologics were lumped together. He would expect that the tumor necrosis factor inhibitors, for example, would be associated with a significantly higher serious infection risk than biologics with other targets.
Dr. Merola conceded the point, adding that the investigators are trying to reanalyze the data in a more granular way to address that shortcoming. Other study limitations included an inability to access the specific doses of systemic treatments used or to stratify patients by disease severity.
Another audience member noted that dermatologists often reassure surgeons that there’s no increased risk of infection associated with psoriasis when in fact there is increased risk in older psoriasis patients, according to these new data.
“We’re not trying to send a message to surgeons to withhold a knee transplant because of a psoriasis plaque over the knee,” Dr. Merola replied. “I think we’ve all been there; we’ve all fought that battle.” Based on the data, he said, he would advise that “our patients who need to be on systemics should remain appropriately on systemics as we see fit.”
The study was entirely funded by Brigham and Women’s Hospital. Dr. Merola reported serving as a consultant to and/or recipient of research grants from nearly two dozen pharmaceutical companies.
REPORTING FROM EADV 2019
PASI-75 with ixekizumab approaches 90% in pediatric psoriasis study
MADRID – The interleukin-17A inhibitor , Kim A. Papp, MD, PhD, reported at the annual congress of the European Academy of Dermatology and Venereology.
The results bode well for an underserved population.
“I think all of us know that there is still a vulnerable population that remains a high-risk population because of the limited number of therapies available for them, and that is children,” said Dr. Papp, a dermatologist and president of Probity Medical Research, Inc., of Waterloo, Ont.
At present, etanercept, one of the earliest biologics to become available, and a relatively less effective one, is the only biologic approved for treatment of pediatric psoriasis. However, Lilly, which sponsored the phase 3 ixekizumab study, has announced that based upon the highly positive findings the company plans to seek Food and Drug Administration approval for an expanded indication for the medication in pediatric psoriasis. The company now markets ixekizumab for the approved indications of treatment of adults with moderate to severe plaque psoriasis, active psoriatic arthritis, or active ankylosing spondylitis.
The 12-week, double-blind, multicenter phase 3 trial known as IXORA-PEDS included 115 pediatric psoriasis patients randomized to weight-based ixekizumab, 30 on weight-based etanercept, and 58 on placebo. At the 12-week mark, everyone was switched to open-label ixekizumab in a long-term extension study. Children weighing less than 25 kg received a 40-mg loading dose of ixekizumab, followed by a maintenance dose of 20 mg by subcutaneous injection every 4 weeks. Patients weighing 25-50 kg got a starting dose of 80 mg, then 40 mg for maintenance therapy. Those who weighed more than 50 kg got the usual adult dosing: a 160-mg loading dose followed by 80 mg every 4 weeks. Etanercept was dosed at 0.8 mg/kg once weekly.
The coprimary endpoints were the proportion of subjects achieving a static Physician’s Global Assessment (sPGA) of 0 or 1 – that is, clear or almost clear skin – at week 12, and the PASI 75 response rate.
An sPGA of 0 or 1 at week 12 was documented in 81% of the ixekizumab group, 11% on placebo, and 40% of etanercept-treated patients, who on average had more severe baseline disease than did the other two groups.
The PASI 75 rate was 89% with ixekizumab, 25% for placebo, and 63% on etanercept. But Dr. Papp indicated that’s too low a bar. “I don’t think PASI 75s are the standard any longer,” he said.
More revealing was the PASI 90 rate: 78% with the IL-17A inhibitor, 5% in placebo-treated controls, and 40% with etanercept.
And then there’s the PASI 100 response rate: 50% with ixekizumab, 2% for placebo, and 17% for etanercept.
“I think this is very telling. I’ll leave it as a tantalizing comment that if one looks at the slope of the curve, it doesn’t yet seem to have reached its plateau at week 12 – and this is very similar to the pattern that we see in the adult population. I don’t have the long-term extension efficacy data, but I am, like you, very interested in seeing where this PASI 100 response rate finally plateaus,” Dr. Papp said.
He did, however, have the combined safety data for the 12-week double-blind phase plus the open-label extension, which he described as essentially the same as the adult experience. Injection-site reactions occurred in 19% of pediatric patients on ixekizumab, but they were generally mild and there were few if any treatment discontinuations for that reason. There was a 2% incidence of Crohn’s disease. Candidiasis and other infections were rare.
Seventy-one percent of the ixekizumab group had at least a 4-point improvement in itch on a 10-point self-rated scale by week 12, as did 20% of placebo-treated controls. A Dermatologic Life Quality Index score of 0 or 1 at week 12, indicative of no or minimal impact of psoriasis on quality of life, was documented in 64% of the ixekizumab group and 23% of controls.
Dr. Papp reported serving as a consultant, investigator, and/or speaker for Lilly and more than three dozen other pharmaceutical companies.
SOURCE: Papp KA. EADV Late breaker.
MADRID – The interleukin-17A inhibitor , Kim A. Papp, MD, PhD, reported at the annual congress of the European Academy of Dermatology and Venereology.
The results bode well for an underserved population.
“I think all of us know that there is still a vulnerable population that remains a high-risk population because of the limited number of therapies available for them, and that is children,” said Dr. Papp, a dermatologist and president of Probity Medical Research, Inc., of Waterloo, Ont.
At present, etanercept, one of the earliest biologics to become available, and a relatively less effective one, is the only biologic approved for treatment of pediatric psoriasis. However, Lilly, which sponsored the phase 3 ixekizumab study, has announced that based upon the highly positive findings the company plans to seek Food and Drug Administration approval for an expanded indication for the medication in pediatric psoriasis. The company now markets ixekizumab for the approved indications of treatment of adults with moderate to severe plaque psoriasis, active psoriatic arthritis, or active ankylosing spondylitis.
The 12-week, double-blind, multicenter phase 3 trial known as IXORA-PEDS included 115 pediatric psoriasis patients randomized to weight-based ixekizumab, 30 on weight-based etanercept, and 58 on placebo. At the 12-week mark, everyone was switched to open-label ixekizumab in a long-term extension study. Children weighing less than 25 kg received a 40-mg loading dose of ixekizumab, followed by a maintenance dose of 20 mg by subcutaneous injection every 4 weeks. Patients weighing 25-50 kg got a starting dose of 80 mg, then 40 mg for maintenance therapy. Those who weighed more than 50 kg got the usual adult dosing: a 160-mg loading dose followed by 80 mg every 4 weeks. Etanercept was dosed at 0.8 mg/kg once weekly.
The coprimary endpoints were the proportion of subjects achieving a static Physician’s Global Assessment (sPGA) of 0 or 1 – that is, clear or almost clear skin – at week 12, and the PASI 75 response rate.
An sPGA of 0 or 1 at week 12 was documented in 81% of the ixekizumab group, 11% on placebo, and 40% of etanercept-treated patients, who on average had more severe baseline disease than did the other two groups.
The PASI 75 rate was 89% with ixekizumab, 25% for placebo, and 63% on etanercept. But Dr. Papp indicated that’s too low a bar. “I don’t think PASI 75s are the standard any longer,” he said.
More revealing was the PASI 90 rate: 78% with the IL-17A inhibitor, 5% in placebo-treated controls, and 40% with etanercept.
And then there’s the PASI 100 response rate: 50% with ixekizumab, 2% for placebo, and 17% for etanercept.
“I think this is very telling. I’ll leave it as a tantalizing comment that if one looks at the slope of the curve, it doesn’t yet seem to have reached its plateau at week 12 – and this is very similar to the pattern that we see in the adult population. I don’t have the long-term extension efficacy data, but I am, like you, very interested in seeing where this PASI 100 response rate finally plateaus,” Dr. Papp said.
He did, however, have the combined safety data for the 12-week double-blind phase plus the open-label extension, which he described as essentially the same as the adult experience. Injection-site reactions occurred in 19% of pediatric patients on ixekizumab, but they were generally mild and there were few if any treatment discontinuations for that reason. There was a 2% incidence of Crohn’s disease. Candidiasis and other infections were rare.
Seventy-one percent of the ixekizumab group had at least a 4-point improvement in itch on a 10-point self-rated scale by week 12, as did 20% of placebo-treated controls. A Dermatologic Life Quality Index score of 0 or 1 at week 12, indicative of no or minimal impact of psoriasis on quality of life, was documented in 64% of the ixekizumab group and 23% of controls.
Dr. Papp reported serving as a consultant, investigator, and/or speaker for Lilly and more than three dozen other pharmaceutical companies.
SOURCE: Papp KA. EADV Late breaker.
MADRID – The interleukin-17A inhibitor , Kim A. Papp, MD, PhD, reported at the annual congress of the European Academy of Dermatology and Venereology.
The results bode well for an underserved population.
“I think all of us know that there is still a vulnerable population that remains a high-risk population because of the limited number of therapies available for them, and that is children,” said Dr. Papp, a dermatologist and president of Probity Medical Research, Inc., of Waterloo, Ont.
At present, etanercept, one of the earliest biologics to become available, and a relatively less effective one, is the only biologic approved for treatment of pediatric psoriasis. However, Lilly, which sponsored the phase 3 ixekizumab study, has announced that based upon the highly positive findings the company plans to seek Food and Drug Administration approval for an expanded indication for the medication in pediatric psoriasis. The company now markets ixekizumab for the approved indications of treatment of adults with moderate to severe plaque psoriasis, active psoriatic arthritis, or active ankylosing spondylitis.
The 12-week, double-blind, multicenter phase 3 trial known as IXORA-PEDS included 115 pediatric psoriasis patients randomized to weight-based ixekizumab, 30 on weight-based etanercept, and 58 on placebo. At the 12-week mark, everyone was switched to open-label ixekizumab in a long-term extension study. Children weighing less than 25 kg received a 40-mg loading dose of ixekizumab, followed by a maintenance dose of 20 mg by subcutaneous injection every 4 weeks. Patients weighing 25-50 kg got a starting dose of 80 mg, then 40 mg for maintenance therapy. Those who weighed more than 50 kg got the usual adult dosing: a 160-mg loading dose followed by 80 mg every 4 weeks. Etanercept was dosed at 0.8 mg/kg once weekly.
The coprimary endpoints were the proportion of subjects achieving a static Physician’s Global Assessment (sPGA) of 0 or 1 – that is, clear or almost clear skin – at week 12, and the PASI 75 response rate.
An sPGA of 0 or 1 at week 12 was documented in 81% of the ixekizumab group, 11% on placebo, and 40% of etanercept-treated patients, who on average had more severe baseline disease than did the other two groups.
The PASI 75 rate was 89% with ixekizumab, 25% for placebo, and 63% on etanercept. But Dr. Papp indicated that’s too low a bar. “I don’t think PASI 75s are the standard any longer,” he said.
More revealing was the PASI 90 rate: 78% with the IL-17A inhibitor, 5% in placebo-treated controls, and 40% with etanercept.
And then there’s the PASI 100 response rate: 50% with ixekizumab, 2% for placebo, and 17% for etanercept.
“I think this is very telling. I’ll leave it as a tantalizing comment that if one looks at the slope of the curve, it doesn’t yet seem to have reached its plateau at week 12 – and this is very similar to the pattern that we see in the adult population. I don’t have the long-term extension efficacy data, but I am, like you, very interested in seeing where this PASI 100 response rate finally plateaus,” Dr. Papp said.
He did, however, have the combined safety data for the 12-week double-blind phase plus the open-label extension, which he described as essentially the same as the adult experience. Injection-site reactions occurred in 19% of pediatric patients on ixekizumab, but they were generally mild and there were few if any treatment discontinuations for that reason. There was a 2% incidence of Crohn’s disease. Candidiasis and other infections were rare.
Seventy-one percent of the ixekizumab group had at least a 4-point improvement in itch on a 10-point self-rated scale by week 12, as did 20% of placebo-treated controls. A Dermatologic Life Quality Index score of 0 or 1 at week 12, indicative of no or minimal impact of psoriasis on quality of life, was documented in 64% of the ixekizumab group and 23% of controls.
Dr. Papp reported serving as a consultant, investigator, and/or speaker for Lilly and more than three dozen other pharmaceutical companies.
SOURCE: Papp KA. EADV Late breaker.
REPORTING FROM THE EADV CONGRESS
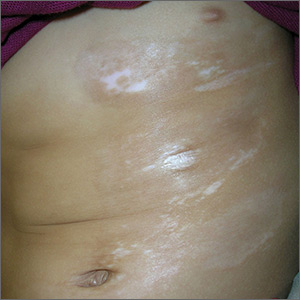
Skin changes on abdomen
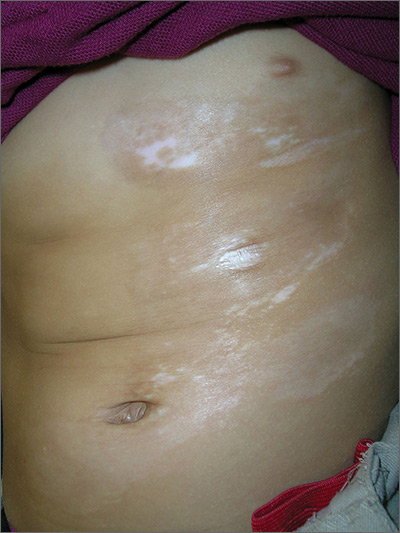
The FP suspected that the child had morphea because the skin was somewhat firm and thickened, and there was hypo- and hyperpigmentation.
Morphea is a localized type of scleroderma and may be seen in children. Fortunately, it does not involve the internal organs. There are no blood tests needed for the diagnosis and antinuclear antibodies should be normal. While a punch biopsy could be considered in less obvious cases in older children, it is probably unnecessary to put a 5-year-old child through the trauma of a biopsy.
The FP referred the child to a dermatologist to confirm the diagnosis and initiate treatment. Typical treatments include topical mid- to high-potency steroids and/or topical calcineurin inhibitors.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Mayeaux EJ, Usatine R. Scleroderma and morphea. In: Usatine R, Smith M, Mayeaux EJ, Chumley H. eds. Color Atlas and Synopsis of Family Medicine, 3rd Edition. New York, NY: McGraw-Hill; 2019:1204-1212.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the 3rd edition of the Color Atlas and Synopsis of Family Medicine as an app by clicking on this link: https://usatinemedia.com/app/color-atlas-of-family-medicine/

The FP suspected that the child had morphea because the skin was somewhat firm and thickened, and there was hypo- and hyperpigmentation.
Morphea is a localized type of scleroderma and may be seen in children. Fortunately, it does not involve the internal organs. There are no blood tests needed for the diagnosis and antinuclear antibodies should be normal. While a punch biopsy could be considered in less obvious cases in older children, it is probably unnecessary to put a 5-year-old child through the trauma of a biopsy.
The FP referred the child to a dermatologist to confirm the diagnosis and initiate treatment. Typical treatments include topical mid- to high-potency steroids and/or topical calcineurin inhibitors.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Mayeaux EJ, Usatine R. Scleroderma and morphea. In: Usatine R, Smith M, Mayeaux EJ, Chumley H. eds. Color Atlas and Synopsis of Family Medicine, 3rd Edition. New York, NY: McGraw-Hill; 2019:1204-1212.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the 3rd edition of the Color Atlas and Synopsis of Family Medicine as an app by clicking on this link: https://usatinemedia.com/app/color-atlas-of-family-medicine/

The FP suspected that the child had morphea because the skin was somewhat firm and thickened, and there was hypo- and hyperpigmentation.
Morphea is a localized type of scleroderma and may be seen in children. Fortunately, it does not involve the internal organs. There are no blood tests needed for the diagnosis and antinuclear antibodies should be normal. While a punch biopsy could be considered in less obvious cases in older children, it is probably unnecessary to put a 5-year-old child through the trauma of a biopsy.
The FP referred the child to a dermatologist to confirm the diagnosis and initiate treatment. Typical treatments include topical mid- to high-potency steroids and/or topical calcineurin inhibitors.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Mayeaux EJ, Usatine R. Scleroderma and morphea. In: Usatine R, Smith M, Mayeaux EJ, Chumley H. eds. Color Atlas and Synopsis of Family Medicine, 3rd Edition. New York, NY: McGraw-Hill; 2019:1204-1212.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the 3rd edition of the Color Atlas and Synopsis of Family Medicine as an app by clicking on this link: https://usatinemedia.com/app/color-atlas-of-family-medicine/
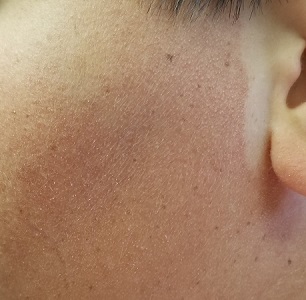
Boy, Is My Face Red!
A 17-year-old boy was born with rough skin on his face, arms, legs, thighs, and posterior shoulders. Over the years, his face, especially the posterior lateral aspects, has become progressively redder, while the roughness has increased. The redness is amplified with heat, exertion, anger, or embarrassment. Regarding the latter, mere mention of the condition by his siblings results in worsening of the erythema. Additionally, the skin in his eyebrows is now turning red and scaly.
The patient denies a history of dandruff. His parents, who have accompanied him to the clinic, report a family history of similar skin changes on triceps and thighs, but not on faces. There is no family history of cardiac anomalies or other congenital abnormalities. The boy’s health is otherwise excellent.
EXAMINATION
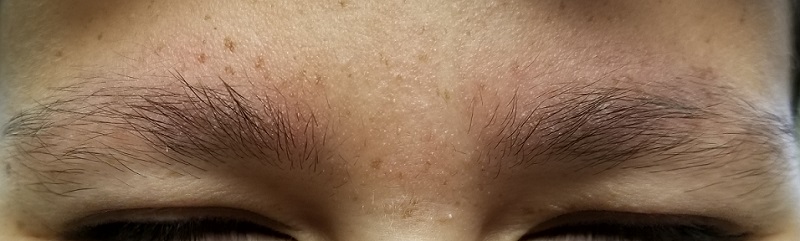
The patient’s bilateral triceps are covered with fine, rough, follicular papules, which create a faintly erythematous look. Similar lesions are visible on his posterior shoulders and anterior thighs. The skin beneath his eyebrows is faintly erythematous and scaly.
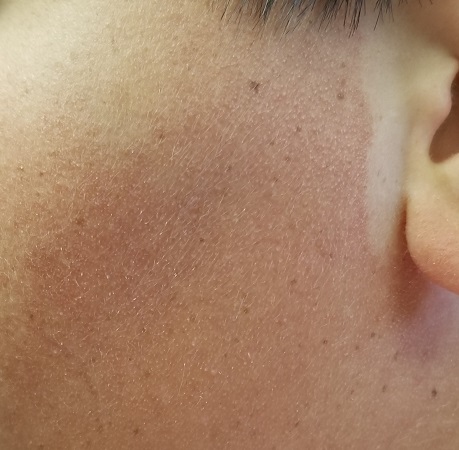
The posterior sides of his face are bright red and covered with the same type of papules. The erythema grows redder as it approaches the immediate preauricular areas, where it ends abruptly, creating a sharp demarcation with the white skin closer to the ears. The visual effect is almost clownish, as if bright red makeup had been applied.
What’s the diagnosis?
DISCUSSION
This young man has all the signs of an extremely rare variant of keratosis pilaris (KP) called ulerythema ophryogenes (UO). In the United States, about 40% of adults have ordinary KP, which usually manifests in childhood (with about 80% of cases occurring in adolescent girls). KP is inherited through autosomal dominance, with highly variable penetrance, though no specific gene has been identified. In this form, KP is considered by most experts to be a minor diagnostic criterion for atopic dermatitis.
However, UO is not merely a variant of KP. Over the decades, it has been connected with more serious conditions, such as cardiofaciocutaneous syndrome, Rubinstein-Taybi syndrome, and Cornelia de Lange disease. Although these conditions are not common, they should be considered when UO is seen.
For this patient, the main concern was his appearance, especially the pronounced erythema around the periphery of his face. This aspect of the problem was addressed with a referral to a provider who can, using an assortment of lasers, try to even out his skin color and hopefully erase the sharp border at the periphery of the affected area.
For the physical discomfort caused by UO, the patient was instructed either to use general moisturizers to reduce dryness or to consider using moisturizers containing salicylic acid, which should help to reduce the prominence of the papules. For the erythema in his brows, he is using 2.5% hydrocortisone ointment two to three times a week.
TAKE-HOME LEARNING POINTS
- Ulerythema ophryogenes is a rarely encountered variant of keratosis pilaris—a condition inherited by autosomal dominance with highly variable penetrance.
- Its main significance, beyond cosmetic concerns, is the possible connection with syndromes that involve heart and structural defects (eg, cardiofaciocutaneous syndrome).
- Treatment options include heavy emollients to soften the scaly papules and laser therapy to reduce the extreme redness seen on the periphery of the face.
A 17-year-old boy was born with rough skin on his face, arms, legs, thighs, and posterior shoulders. Over the years, his face, especially the posterior lateral aspects, has become progressively redder, while the roughness has increased. The redness is amplified with heat, exertion, anger, or embarrassment. Regarding the latter, mere mention of the condition by his siblings results in worsening of the erythema. Additionally, the skin in his eyebrows is now turning red and scaly.
The patient denies a history of dandruff. His parents, who have accompanied him to the clinic, report a family history of similar skin changes on triceps and thighs, but not on faces. There is no family history of cardiac anomalies or other congenital abnormalities. The boy’s health is otherwise excellent.

EXAMINATION
The patient’s bilateral triceps are covered with fine, rough, follicular papules, which create a faintly erythematous look. Similar lesions are visible on his posterior shoulders and anterior thighs. The skin beneath his eyebrows is faintly erythematous and scaly.
The posterior sides of his face are bright red and covered with the same type of papules. The erythema grows redder as it approaches the immediate preauricular areas, where it ends abruptly, creating a sharp demarcation with the white skin closer to the ears. The visual effect is almost clownish, as if bright red makeup had been applied.

What’s the diagnosis?
DISCUSSION
This young man has all the signs of an extremely rare variant of keratosis pilaris (KP) called ulerythema ophryogenes (UO). In the United States, about 40% of adults have ordinary KP, which usually manifests in childhood (with about 80% of cases occurring in adolescent girls). KP is inherited through autosomal dominance, with highly variable penetrance, though no specific gene has been identified. In this form, KP is considered by most experts to be a minor diagnostic criterion for atopic dermatitis.
However, UO is not merely a variant of KP. Over the decades, it has been connected with more serious conditions, such as cardiofaciocutaneous syndrome, Rubinstein-Taybi syndrome, and Cornelia de Lange disease. Although these conditions are not common, they should be considered when UO is seen.
For this patient, the main concern was his appearance, especially the pronounced erythema around the periphery of his face. This aspect of the problem was addressed with a referral to a provider who can, using an assortment of lasers, try to even out his skin color and hopefully erase the sharp border at the periphery of the affected area.
For the physical discomfort caused by UO, the patient was instructed either to use general moisturizers to reduce dryness or to consider using moisturizers containing salicylic acid, which should help to reduce the prominence of the papules. For the erythema in his brows, he is using 2.5% hydrocortisone ointment two to three times a week.
TAKE-HOME LEARNING POINTS
- Ulerythema ophryogenes is a rarely encountered variant of keratosis pilaris—a condition inherited by autosomal dominance with highly variable penetrance.
- Its main significance, beyond cosmetic concerns, is the possible connection with syndromes that involve heart and structural defects (eg, cardiofaciocutaneous syndrome).
- Treatment options include heavy emollients to soften the scaly papules and laser therapy to reduce the extreme redness seen on the periphery of the face.
A 17-year-old boy was born with rough skin on his face, arms, legs, thighs, and posterior shoulders. Over the years, his face, especially the posterior lateral aspects, has become progressively redder, while the roughness has increased. The redness is amplified with heat, exertion, anger, or embarrassment. Regarding the latter, mere mention of the condition by his siblings results in worsening of the erythema. Additionally, the skin in his eyebrows is now turning red and scaly.
The patient denies a history of dandruff. His parents, who have accompanied him to the clinic, report a family history of similar skin changes on triceps and thighs, but not on faces. There is no family history of cardiac anomalies or other congenital abnormalities. The boy’s health is otherwise excellent.

EXAMINATION
The patient’s bilateral triceps are covered with fine, rough, follicular papules, which create a faintly erythematous look. Similar lesions are visible on his posterior shoulders and anterior thighs. The skin beneath his eyebrows is faintly erythematous and scaly.
The posterior sides of his face are bright red and covered with the same type of papules. The erythema grows redder as it approaches the immediate preauricular areas, where it ends abruptly, creating a sharp demarcation with the white skin closer to the ears. The visual effect is almost clownish, as if bright red makeup had been applied.

What’s the diagnosis?
DISCUSSION
This young man has all the signs of an extremely rare variant of keratosis pilaris (KP) called ulerythema ophryogenes (UO). In the United States, about 40% of adults have ordinary KP, which usually manifests in childhood (with about 80% of cases occurring in adolescent girls). KP is inherited through autosomal dominance, with highly variable penetrance, though no specific gene has been identified. In this form, KP is considered by most experts to be a minor diagnostic criterion for atopic dermatitis.
However, UO is not merely a variant of KP. Over the decades, it has been connected with more serious conditions, such as cardiofaciocutaneous syndrome, Rubinstein-Taybi syndrome, and Cornelia de Lange disease. Although these conditions are not common, they should be considered when UO is seen.
For this patient, the main concern was his appearance, especially the pronounced erythema around the periphery of his face. This aspect of the problem was addressed with a referral to a provider who can, using an assortment of lasers, try to even out his skin color and hopefully erase the sharp border at the periphery of the affected area.
For the physical discomfort caused by UO, the patient was instructed either to use general moisturizers to reduce dryness or to consider using moisturizers containing salicylic acid, which should help to reduce the prominence of the papules. For the erythema in his brows, he is using 2.5% hydrocortisone ointment two to three times a week.
TAKE-HOME LEARNING POINTS
- Ulerythema ophryogenes is a rarely encountered variant of keratosis pilaris—a condition inherited by autosomal dominance with highly variable penetrance.
- Its main significance, beyond cosmetic concerns, is the possible connection with syndromes that involve heart and structural defects (eg, cardiofaciocutaneous syndrome).
- Treatment options include heavy emollients to soften the scaly papules and laser therapy to reduce the extreme redness seen on the periphery of the face.
No tacrolimus/cancer link in atopic dermatitis in 10-year study
MADRID – participating in the large, prospective, observational APPLES study, Regina Folster-Holst, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
With nearly 45,000 person-years of follow-up in APPLES (A Prospective Pediatric Longitudinal Evaluation Study), there were no lymphomas and just a single case of skin cancer. That’s highly reassuring, since those were the two types of malignancies singled out as being of particular concern in the boxed warnings for the topical calcineurin inhibitors tacrolimus and pimecrolimus mandated by U.S. and European regulatory agencies in 2005, noted Dr. Folster-Holst, professor of dermatology at Christian Albrechts University of Kiel (Germany).
APPLES included 7,954 children with moderate or severe AD who were a median of 6 years old at enrollment in the study, conducted at 314 sites in the United States, Canada, and seven European countries. This was a naturalistic study in which patients used the topical calcineurin inhibitor as needed, with no restrictions.
A total of six cancers were diagnosed in six individuals during 44,629 person-years of prospective follow-up: one case each of chronic myeloid leukemia, alveolar rhabdomyosarcoma, malignant paraganglioma, carcinoid tumor of the appendix, spinal cord neoplasm, and Spitzoid melanoma. None of those malignancies are classically associated with immunosuppressive therapy.
The primary outcome in APPLES was the standardized incidence ratio of observed cancers to the expected number based upon extrapolation from the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) database, as well as national cancer registries in the other countries where the study was carried out. The expected number of cancers was 5.95, yielding a standardized incidence ratio of 1.01.
Only 27% of patients completed the study. Investigators had anticipated a substantial attrition rate and recalculated their statistics based upon a range of hypothetically increased cancer rates among the dropouts. Even if the cancer rate was 2.5-fold higher in dropouts than in those who remained in the study – a far-fetched possibility – the standardized incidence ratio would not be significantly affected, according to Dr. Folster-Holst.
The new APPLES findings were preceded by a favorable report on long-term use of topical pimecrolimus from the Pediatric Eczema Elective Registry (PEER). The study included 7,457 pimecrolimus-using children with AD followed for 26,792 person-years. The standardized incidence ratio for all cancers was not significantly increased at 1.2. The investigators concluded “it seems unlikely” that topical pimecrolimus as generally used for treatment of AD is associated with an increased risk of malignancy (JAMA Dermatol. 2015 Jun;151[6]:594-9).
The boxed warnings for the topical calcineurin inhibitors have been the source of enormous frustration for dermatologists. The warnings were ordered because of regulatory concern about an increased risk of malignancy in organ transplant recipients on systemic calcineurin inhibitors for immunosuppression, even though the topical agents – unlike the systemic versions – are used intermittently, their systemic absorption is low to nil, and no plausible mechanism by which they could cause cancer has been put forth. Many physicians believe these drugs are probably safer than topical corticosteroids, so the first question put to Dr. Folster-Holst from the audience was, When will the boxed warnings be removed?
“That’s a good question,” she replied. “Patients and parents are afraid. But I think we have now a good argument to move forward with topically applied calcineurin inhibitors.”
Dr. Folster-Holst reported having no financial conflicts of interest regarding the APPLES study, funded by LEO Pharma.
MADRID – participating in the large, prospective, observational APPLES study, Regina Folster-Holst, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
With nearly 45,000 person-years of follow-up in APPLES (A Prospective Pediatric Longitudinal Evaluation Study), there were no lymphomas and just a single case of skin cancer. That’s highly reassuring, since those were the two types of malignancies singled out as being of particular concern in the boxed warnings for the topical calcineurin inhibitors tacrolimus and pimecrolimus mandated by U.S. and European regulatory agencies in 2005, noted Dr. Folster-Holst, professor of dermatology at Christian Albrechts University of Kiel (Germany).
APPLES included 7,954 children with moderate or severe AD who were a median of 6 years old at enrollment in the study, conducted at 314 sites in the United States, Canada, and seven European countries. This was a naturalistic study in which patients used the topical calcineurin inhibitor as needed, with no restrictions.
A total of six cancers were diagnosed in six individuals during 44,629 person-years of prospective follow-up: one case each of chronic myeloid leukemia, alveolar rhabdomyosarcoma, malignant paraganglioma, carcinoid tumor of the appendix, spinal cord neoplasm, and Spitzoid melanoma. None of those malignancies are classically associated with immunosuppressive therapy.
The primary outcome in APPLES was the standardized incidence ratio of observed cancers to the expected number based upon extrapolation from the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) database, as well as national cancer registries in the other countries where the study was carried out. The expected number of cancers was 5.95, yielding a standardized incidence ratio of 1.01.
Only 27% of patients completed the study. Investigators had anticipated a substantial attrition rate and recalculated their statistics based upon a range of hypothetically increased cancer rates among the dropouts. Even if the cancer rate was 2.5-fold higher in dropouts than in those who remained in the study – a far-fetched possibility – the standardized incidence ratio would not be significantly affected, according to Dr. Folster-Holst.
The new APPLES findings were preceded by a favorable report on long-term use of topical pimecrolimus from the Pediatric Eczema Elective Registry (PEER). The study included 7,457 pimecrolimus-using children with AD followed for 26,792 person-years. The standardized incidence ratio for all cancers was not significantly increased at 1.2. The investigators concluded “it seems unlikely” that topical pimecrolimus as generally used for treatment of AD is associated with an increased risk of malignancy (JAMA Dermatol. 2015 Jun;151[6]:594-9).
The boxed warnings for the topical calcineurin inhibitors have been the source of enormous frustration for dermatologists. The warnings were ordered because of regulatory concern about an increased risk of malignancy in organ transplant recipients on systemic calcineurin inhibitors for immunosuppression, even though the topical agents – unlike the systemic versions – are used intermittently, their systemic absorption is low to nil, and no plausible mechanism by which they could cause cancer has been put forth. Many physicians believe these drugs are probably safer than topical corticosteroids, so the first question put to Dr. Folster-Holst from the audience was, When will the boxed warnings be removed?
“That’s a good question,” she replied. “Patients and parents are afraid. But I think we have now a good argument to move forward with topically applied calcineurin inhibitors.”
Dr. Folster-Holst reported having no financial conflicts of interest regarding the APPLES study, funded by LEO Pharma.
MADRID – participating in the large, prospective, observational APPLES study, Regina Folster-Holst, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
With nearly 45,000 person-years of follow-up in APPLES (A Prospective Pediatric Longitudinal Evaluation Study), there were no lymphomas and just a single case of skin cancer. That’s highly reassuring, since those were the two types of malignancies singled out as being of particular concern in the boxed warnings for the topical calcineurin inhibitors tacrolimus and pimecrolimus mandated by U.S. and European regulatory agencies in 2005, noted Dr. Folster-Holst, professor of dermatology at Christian Albrechts University of Kiel (Germany).
APPLES included 7,954 children with moderate or severe AD who were a median of 6 years old at enrollment in the study, conducted at 314 sites in the United States, Canada, and seven European countries. This was a naturalistic study in which patients used the topical calcineurin inhibitor as needed, with no restrictions.
A total of six cancers were diagnosed in six individuals during 44,629 person-years of prospective follow-up: one case each of chronic myeloid leukemia, alveolar rhabdomyosarcoma, malignant paraganglioma, carcinoid tumor of the appendix, spinal cord neoplasm, and Spitzoid melanoma. None of those malignancies are classically associated with immunosuppressive therapy.
The primary outcome in APPLES was the standardized incidence ratio of observed cancers to the expected number based upon extrapolation from the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) database, as well as national cancer registries in the other countries where the study was carried out. The expected number of cancers was 5.95, yielding a standardized incidence ratio of 1.01.
Only 27% of patients completed the study. Investigators had anticipated a substantial attrition rate and recalculated their statistics based upon a range of hypothetically increased cancer rates among the dropouts. Even if the cancer rate was 2.5-fold higher in dropouts than in those who remained in the study – a far-fetched possibility – the standardized incidence ratio would not be significantly affected, according to Dr. Folster-Holst.
The new APPLES findings were preceded by a favorable report on long-term use of topical pimecrolimus from the Pediatric Eczema Elective Registry (PEER). The study included 7,457 pimecrolimus-using children with AD followed for 26,792 person-years. The standardized incidence ratio for all cancers was not significantly increased at 1.2. The investigators concluded “it seems unlikely” that topical pimecrolimus as generally used for treatment of AD is associated with an increased risk of malignancy (JAMA Dermatol. 2015 Jun;151[6]:594-9).
The boxed warnings for the topical calcineurin inhibitors have been the source of enormous frustration for dermatologists. The warnings were ordered because of regulatory concern about an increased risk of malignancy in organ transplant recipients on systemic calcineurin inhibitors for immunosuppression, even though the topical agents – unlike the systemic versions – are used intermittently, their systemic absorption is low to nil, and no plausible mechanism by which they could cause cancer has been put forth. Many physicians believe these drugs are probably safer than topical corticosteroids, so the first question put to Dr. Folster-Holst from the audience was, When will the boxed warnings be removed?
“That’s a good question,” she replied. “Patients and parents are afraid. But I think we have now a good argument to move forward with topically applied calcineurin inhibitors.”
Dr. Folster-Holst reported having no financial conflicts of interest regarding the APPLES study, funded by LEO Pharma.
REPORTING FROM THE EADV CONGRESS
Atopic Dermatitis
FDA approves minocycline foam for moderate, severe acne
The Food and Drug Administration has vulgaris in adults and pediatric patients aged at least 9 years.
FDA approval was based on three phase 3, 12-week, multicenter, randomized, double-blind, vehicle-controlled studies of patients with moderate to severe acne vulgaris who were treated once daily with minocycline 4% plus vehicle or vehicle alone, according to a press release from the manufacturer, Foamix Pharmaceuticals. In all three studies, patients receiving minocycline had a reduction in the number of inflammatory lesions, compared with vehicle alone; in two studies, patients in the minocycline groups had significantly improved Investigator’s Global Assessment scores.
The most commonly reported adverse event reported during the trials was headache. No serious adverse events were reported. The company says it is expected to be available in January 2020.
Find the prescribing information on the FDA website.
The Food and Drug Administration has vulgaris in adults and pediatric patients aged at least 9 years.
FDA approval was based on three phase 3, 12-week, multicenter, randomized, double-blind, vehicle-controlled studies of patients with moderate to severe acne vulgaris who were treated once daily with minocycline 4% plus vehicle or vehicle alone, according to a press release from the manufacturer, Foamix Pharmaceuticals. In all three studies, patients receiving minocycline had a reduction in the number of inflammatory lesions, compared with vehicle alone; in two studies, patients in the minocycline groups had significantly improved Investigator’s Global Assessment scores.
The most commonly reported adverse event reported during the trials was headache. No serious adverse events were reported. The company says it is expected to be available in January 2020.
Find the prescribing information on the FDA website.
The Food and Drug Administration has vulgaris in adults and pediatric patients aged at least 9 years.
FDA approval was based on three phase 3, 12-week, multicenter, randomized, double-blind, vehicle-controlled studies of patients with moderate to severe acne vulgaris who were treated once daily with minocycline 4% plus vehicle or vehicle alone, according to a press release from the manufacturer, Foamix Pharmaceuticals. In all three studies, patients receiving minocycline had a reduction in the number of inflammatory lesions, compared with vehicle alone; in two studies, patients in the minocycline groups had significantly improved Investigator’s Global Assessment scores.
The most commonly reported adverse event reported during the trials was headache. No serious adverse events were reported. The company says it is expected to be available in January 2020.
Find the prescribing information on the FDA website.
Acute Palmar and Plantar Rash in a 52-Year-Old Woman
A 52-year-old woman presented to a primary care clinic with a 3-week history of rash on her feet that had spread to her hands in the previous week. She described the rash as painful, burning, and itching with no drainage. She denied any recent illness, fever, chills, medication changes, or environmental exposures. Home treatments included Epsom salt baths and lotion with no improvement.
Past medical history included hypertension. She was a smoker with a 30-pack-year history and drank alcohol on a daily basis. Her medications included losartan and atorvastatin.
On examination, multiple papular and scabbed lesions were present with mild scaling. Additional review of systems and physical exam were benign. A KOH prep showed hyphae. The patient was diagnosed with tinea pedis and prescribed fluconazole (150-mg tablet once per week for 2 weeks).
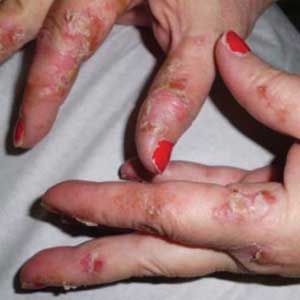
Two weeks later, after completing the antifungal therapy, the patient returned with pain limiting her ability to bear weight or grasp objects. Clinical examination showed well-demarcated erythematous scaly and hyperkeratotic plaques with scattered papular and pustular lesions on bilateral palmar and medial aspects of plantar surfaces (see Figures 1 and 2). A repeat KOH was not completed. The patient was diagnosed with palmoplantar pustulosis (PPP).
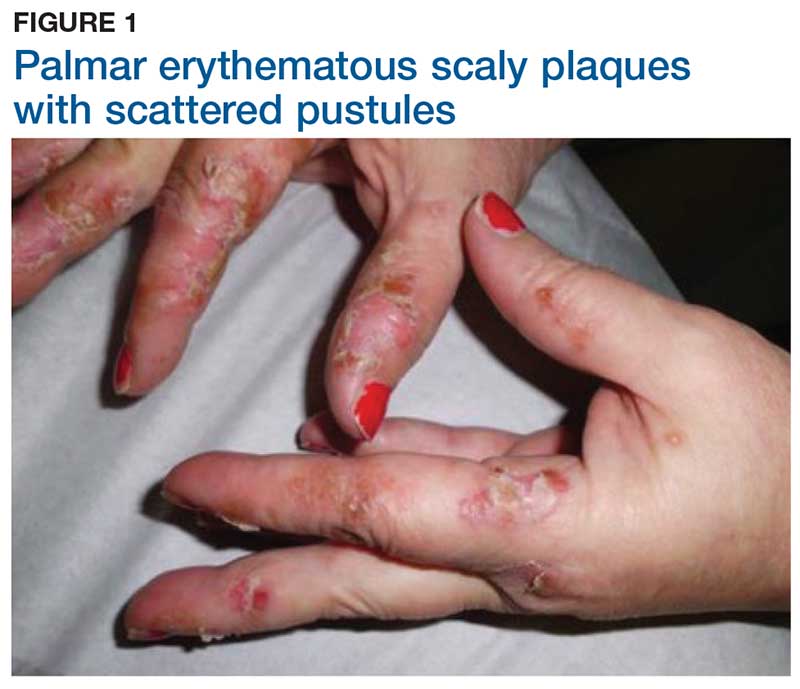
DISCUSSION
PPP is a chronic, relapsing, inflammatory skin condition that results in painful lesions on the palms and the soles.1,2 There is debate as to whether PPP is a variant of psoriasis or a separate condition; depending on physical manifestations, one can be diagnosed with palmoplantar plaque psoriasis, PPP, or a combination of the two.3,4
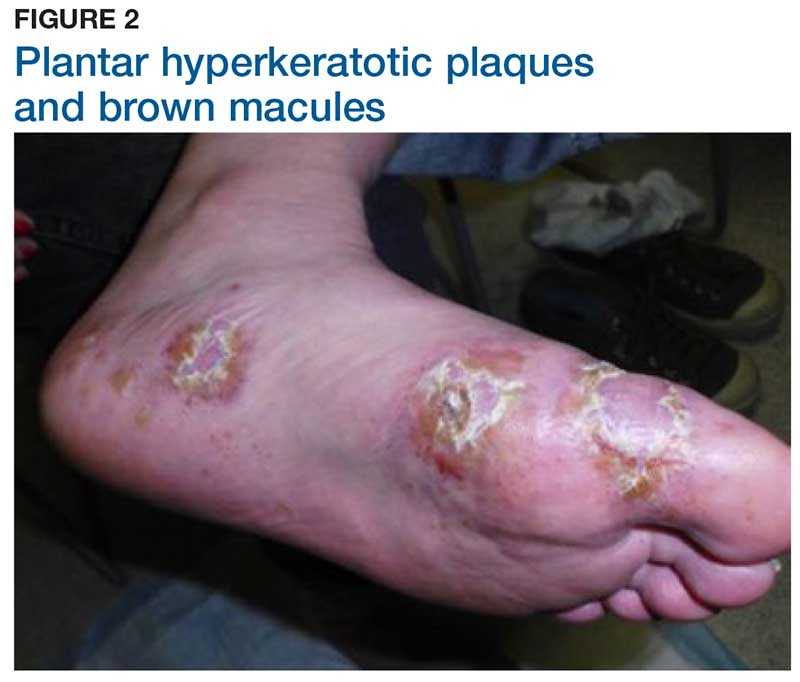
The exact cause of PPP is unknown; however, increased levels of inflammatory cytokines interleukin (IL)-17 and IL-22 may be involved in the pathogenesis of the disease.5 Additional genetic and environmental factors, most significantly smoking, play an important role in its development.2,6
Clinical presentation
Inflammation associated with PPP typically manifests in the classic features of pustules that coalesce and resolve over several days, resulting in brown macules, hyperkeratosis, fissures, and debilitating pain.4,7 Some patients may have co-occurring onycholysis resulting from nail dystrophy and destruction or plaque psoriasis elsewhere on their body.8 PPP often persists for years with periods of exacerbation and remission, and it significantly affects the patient’s ability to perform activities of daily living without pain.8,9 It is exceedingly rare and most commonly affects middle-aged women with a smoking history or current smoking status.7
Continue to: Laboratory diagnosis
Laboratory diagnosis
The diagnosis of PPP is based on clinical presentation and physical exam. Laboratory testing, such as KOH prep, may assist in ruling out dermatophyte infection; a complete blood count may assist in eliminating a bacterial infection as the cause. Skin biopsy is not necessary unless diagnosis is uncertain or prolonged treatment has not produced a response.
Differential diagnosis
The differential diagnosis of PPP includes skin conditions that involve the palms and/or the soles and may have fungal, allergic, or bacterial origins.
Fungal. Tinea manuum (palms) and tinea pedis (soles) result from dermatophyte infection and manifest with erythema and/or scaling and pruritis. A positive KOH examination can confirm diagnosis. On examination, fungal infections are commonly unilateral and asymmetric.8 Treatment with an antifungal agent should result in resolution of symptoms.
Allergic. Contact with an allergen can result in skin erythema, pruritis, and pain at the exposed area. Contact dermatitis can result from an inflammatory response to an allergen or irritant, and it is often localized and well demarcated. This is an acute condition that resolves over time with antihistamines and avoidance of irritants.
Dyshidrotic eczema results in small, pruritic blisters on the palms and the soles and can be recurrent and related to seasonal allergen exposure. Diagnosis is made from history and physical exam. Treatment often consists of emollients and occasionally topical steroids, depending on the severity.
Continue to: Bacterial
Bacterial. A primary bacterial cause of bilateral skin lesions on the palms and the soles is uncommon. However, any open skin lesion can result in secondary bacterial infection. The pustules of PPP are often sterile and do not require bacterial culture; however, additional symptoms of fever, purulence, warmth, and worsening of symptoms may prompt further evaluation for a bacterial origin or complication.
Management
Due to limited quality data on treatment recommendations, the treatment options for PPP vary greatly. Most studies recommend topical versus systemic therapy for initial management.1-2,8,10-11 Firstline therapy often consists of topical corticosteroids and occlusive dressings, followed by oral retinoids (acitretin, alitretinoin) or photochemotherapy.1,8 Third-line therapy can include immunosuppressants (ciclosporin, methotrexate) or biologics (secukinumab).1,12 Recent data have shown positive results with vitamin D3 analogs (maxacalcitol, betamethasone butyrate propionate) as monotherapy or in combination with corticosteroids.10-11 Duration of therapy ranges from 4 to 8 weeks throughout the literature, depending on severity; however, many patients see improvement in the first few weeks.
Conservative measures to maintain remission include smoking cessation, skin emollients, and avoidance of irritants. It is important to educate patients about the chronicity of the disease and early treatment to prevent secondary infection or significant impact on quality of life.
OUTCOME FOR THE CASE PATIENT
The patient was prescribed triamcinolone acetonide (0.5% ointment applied bid), to be used until symptoms improved. After 1 week of treatment, she confirmed (verbally) that symptoms had resolved. She declined a follow-up visit or referral to dermatology.
CONCLUSION
Although PPP is fairly uncommon, it is important for clinicians to consider this diagnosis in patients presenting with localized rash on their palms and soles. This debilitating condition greatly affects a patient’s quality of life and, although it is chronic in nature, available treatments described in the literature have shown success in both acute resolution and ongoing remission of the disease.
1. Sevrain M, Richard M-A, Barnetche T, et al. Treatment for palmoplantar pustular psoriasis: systematic literature review, evidence-based recommendations and expert opinion. J Eur Acad Dermatol Venereol. 2014;28(suppl 5):13-16.
2. Olazagasti JM, Ma JE, Wetter DA. Clinical features, etiological factors, associated disorders, and treatment of palmoplantar pustulosis: the Mayo Clinic experience, 1996-2013. Mayo Clin Proc. 2017;92(9):1351-1358.
3. Bissonnette R, Suárez-Fariñas M, Li X, et al. Based on molecular profiling of gene expression, palmoplantar pustulosis and palmoplantar pustular psoriasis are highly related diseases that appear to be distinct from psoriasis vulgaris. PLoS One. 2016;11(5):1-11.
4. Raposo I, Torres T. Palmoplantar psoriasis and palmoplantar pustulosis: current treatment and future prospects. Am J Clin Dermatol. 2016;17(4):349-358.
5. Bissonnette R, Fuentes-Duculan J, Mashiko S, et al. Palmoplantar pustular psoriasis (PPPP) is characterized by activation of the IL-17A pathway. J Dermatol Sci. 2017;85(1):20-26.
6. Misiak-Galazka M, Wolska H, Rudnicka L. What do we know about palmoplantar pustulosis? J Eur Acad Dermatol Venereol. 2017;31(1):38-44.
7. Brunasso AMG, Puntoni M, Aberer W, et al. Clinical and epidemiological comparison of patients affected by palmoplantar plaque psoriasis and palmoplantar pustulosis: a case series study. Br J Dermatol. 2013;168(6):1243-1251.
8. Engin B, As¸kın Ö, Tüzün Y. Palmoplantar psoriasis. Clin Dermatol. 2017; 35(1):19-27.
9. Chung J, Callas Duffin K, Takeshita J, et al. Palmoplantar psoriasis is associated with greater impairment of health-related quality of life compared to moderate-to-severe plaque psoriasis. J Am Acad Dermatol. 2014;71(4):623-632.
10. Umezawa Y, Nakagawa H, Tamaki K. Phase III clinical study of maxacalcitol ointment in patients with palmoplantar pustulosis: a randomized, double-blind, placebo-controlled trial. J Dermatol. 2016;43(3):288-293.
11. Muro M, Kawakami H, Matsumoto Y, et al. Topical combination therapy with vitamin D3 and corticosteroid ointment for palmoplantar pustulosis: a prospective, randomized, left-right comparison study. J Dermatolog Treat. 2016;27(1):51-53.
12. Gottlieb A, Sullivan J, van Doorn M, et al. Secukinumab shows significant efficacy in palmoplantar psoriasis: results from GESTURE, a randomized controlled trial. J Am Acad Dermatol. 2017;76(1):70-80.
A 52-year-old woman presented to a primary care clinic with a 3-week history of rash on her feet that had spread to her hands in the previous week. She described the rash as painful, burning, and itching with no drainage. She denied any recent illness, fever, chills, medication changes, or environmental exposures. Home treatments included Epsom salt baths and lotion with no improvement.
Past medical history included hypertension. She was a smoker with a 30-pack-year history and drank alcohol on a daily basis. Her medications included losartan and atorvastatin.
On examination, multiple papular and scabbed lesions were present with mild scaling. Additional review of systems and physical exam were benign. A KOH prep showed hyphae. The patient was diagnosed with tinea pedis and prescribed fluconazole (150-mg tablet once per week for 2 weeks).
Two weeks later, after completing the antifungal therapy, the patient returned with pain limiting her ability to bear weight or grasp objects. Clinical examination showed well-demarcated erythematous scaly and hyperkeratotic plaques with scattered papular and pustular lesions on bilateral palmar and medial aspects of plantar surfaces (see Figures 1 and 2). A repeat KOH was not completed. The patient was diagnosed with palmoplantar pustulosis (PPP).

DISCUSSION
PPP is a chronic, relapsing, inflammatory skin condition that results in painful lesions on the palms and the soles.1,2 There is debate as to whether PPP is a variant of psoriasis or a separate condition; depending on physical manifestations, one can be diagnosed with palmoplantar plaque psoriasis, PPP, or a combination of the two.3,4

The exact cause of PPP is unknown; however, increased levels of inflammatory cytokines interleukin (IL)-17 and IL-22 may be involved in the pathogenesis of the disease.5 Additional genetic and environmental factors, most significantly smoking, play an important role in its development.2,6
Clinical presentation
Inflammation associated with PPP typically manifests in the classic features of pustules that coalesce and resolve over several days, resulting in brown macules, hyperkeratosis, fissures, and debilitating pain.4,7 Some patients may have co-occurring onycholysis resulting from nail dystrophy and destruction or plaque psoriasis elsewhere on their body.8 PPP often persists for years with periods of exacerbation and remission, and it significantly affects the patient’s ability to perform activities of daily living without pain.8,9 It is exceedingly rare and most commonly affects middle-aged women with a smoking history or current smoking status.7
Continue to: Laboratory diagnosis
Laboratory diagnosis
The diagnosis of PPP is based on clinical presentation and physical exam. Laboratory testing, such as KOH prep, may assist in ruling out dermatophyte infection; a complete blood count may assist in eliminating a bacterial infection as the cause. Skin biopsy is not necessary unless diagnosis is uncertain or prolonged treatment has not produced a response.
Differential diagnosis
The differential diagnosis of PPP includes skin conditions that involve the palms and/or the soles and may have fungal, allergic, or bacterial origins.
Fungal. Tinea manuum (palms) and tinea pedis (soles) result from dermatophyte infection and manifest with erythema and/or scaling and pruritis. A positive KOH examination can confirm diagnosis. On examination, fungal infections are commonly unilateral and asymmetric.8 Treatment with an antifungal agent should result in resolution of symptoms.
Allergic. Contact with an allergen can result in skin erythema, pruritis, and pain at the exposed area. Contact dermatitis can result from an inflammatory response to an allergen or irritant, and it is often localized and well demarcated. This is an acute condition that resolves over time with antihistamines and avoidance of irritants.
Dyshidrotic eczema results in small, pruritic blisters on the palms and the soles and can be recurrent and related to seasonal allergen exposure. Diagnosis is made from history and physical exam. Treatment often consists of emollients and occasionally topical steroids, depending on the severity.
Continue to: Bacterial
Bacterial. A primary bacterial cause of bilateral skin lesions on the palms and the soles is uncommon. However, any open skin lesion can result in secondary bacterial infection. The pustules of PPP are often sterile and do not require bacterial culture; however, additional symptoms of fever, purulence, warmth, and worsening of symptoms may prompt further evaluation for a bacterial origin or complication.
Management
Due to limited quality data on treatment recommendations, the treatment options for PPP vary greatly. Most studies recommend topical versus systemic therapy for initial management.1-2,8,10-11 Firstline therapy often consists of topical corticosteroids and occlusive dressings, followed by oral retinoids (acitretin, alitretinoin) or photochemotherapy.1,8 Third-line therapy can include immunosuppressants (ciclosporin, methotrexate) or biologics (secukinumab).1,12 Recent data have shown positive results with vitamin D3 analogs (maxacalcitol, betamethasone butyrate propionate) as monotherapy or in combination with corticosteroids.10-11 Duration of therapy ranges from 4 to 8 weeks throughout the literature, depending on severity; however, many patients see improvement in the first few weeks.
Conservative measures to maintain remission include smoking cessation, skin emollients, and avoidance of irritants. It is important to educate patients about the chronicity of the disease and early treatment to prevent secondary infection or significant impact on quality of life.
OUTCOME FOR THE CASE PATIENT
The patient was prescribed triamcinolone acetonide (0.5% ointment applied bid), to be used until symptoms improved. After 1 week of treatment, she confirmed (verbally) that symptoms had resolved. She declined a follow-up visit or referral to dermatology.
CONCLUSION
Although PPP is fairly uncommon, it is important for clinicians to consider this diagnosis in patients presenting with localized rash on their palms and soles. This debilitating condition greatly affects a patient’s quality of life and, although it is chronic in nature, available treatments described in the literature have shown success in both acute resolution and ongoing remission of the disease.
A 52-year-old woman presented to a primary care clinic with a 3-week history of rash on her feet that had spread to her hands in the previous week. She described the rash as painful, burning, and itching with no drainage. She denied any recent illness, fever, chills, medication changes, or environmental exposures. Home treatments included Epsom salt baths and lotion with no improvement.
Past medical history included hypertension. She was a smoker with a 30-pack-year history and drank alcohol on a daily basis. Her medications included losartan and atorvastatin.
On examination, multiple papular and scabbed lesions were present with mild scaling. Additional review of systems and physical exam were benign. A KOH prep showed hyphae. The patient was diagnosed with tinea pedis and prescribed fluconazole (150-mg tablet once per week for 2 weeks).
Two weeks later, after completing the antifungal therapy, the patient returned with pain limiting her ability to bear weight or grasp objects. Clinical examination showed well-demarcated erythematous scaly and hyperkeratotic plaques with scattered papular and pustular lesions on bilateral palmar and medial aspects of plantar surfaces (see Figures 1 and 2). A repeat KOH was not completed. The patient was diagnosed with palmoplantar pustulosis (PPP).

DISCUSSION
PPP is a chronic, relapsing, inflammatory skin condition that results in painful lesions on the palms and the soles.1,2 There is debate as to whether PPP is a variant of psoriasis or a separate condition; depending on physical manifestations, one can be diagnosed with palmoplantar plaque psoriasis, PPP, or a combination of the two.3,4

The exact cause of PPP is unknown; however, increased levels of inflammatory cytokines interleukin (IL)-17 and IL-22 may be involved in the pathogenesis of the disease.5 Additional genetic and environmental factors, most significantly smoking, play an important role in its development.2,6
Clinical presentation
Inflammation associated with PPP typically manifests in the classic features of pustules that coalesce and resolve over several days, resulting in brown macules, hyperkeratosis, fissures, and debilitating pain.4,7 Some patients may have co-occurring onycholysis resulting from nail dystrophy and destruction or plaque psoriasis elsewhere on their body.8 PPP often persists for years with periods of exacerbation and remission, and it significantly affects the patient’s ability to perform activities of daily living without pain.8,9 It is exceedingly rare and most commonly affects middle-aged women with a smoking history or current smoking status.7
Continue to: Laboratory diagnosis
Laboratory diagnosis
The diagnosis of PPP is based on clinical presentation and physical exam. Laboratory testing, such as KOH prep, may assist in ruling out dermatophyte infection; a complete blood count may assist in eliminating a bacterial infection as the cause. Skin biopsy is not necessary unless diagnosis is uncertain or prolonged treatment has not produced a response.
Differential diagnosis
The differential diagnosis of PPP includes skin conditions that involve the palms and/or the soles and may have fungal, allergic, or bacterial origins.
Fungal. Tinea manuum (palms) and tinea pedis (soles) result from dermatophyte infection and manifest with erythema and/or scaling and pruritis. A positive KOH examination can confirm diagnosis. On examination, fungal infections are commonly unilateral and asymmetric.8 Treatment with an antifungal agent should result in resolution of symptoms.
Allergic. Contact with an allergen can result in skin erythema, pruritis, and pain at the exposed area. Contact dermatitis can result from an inflammatory response to an allergen or irritant, and it is often localized and well demarcated. This is an acute condition that resolves over time with antihistamines and avoidance of irritants.
Dyshidrotic eczema results in small, pruritic blisters on the palms and the soles and can be recurrent and related to seasonal allergen exposure. Diagnosis is made from history and physical exam. Treatment often consists of emollients and occasionally topical steroids, depending on the severity.
Continue to: Bacterial
Bacterial. A primary bacterial cause of bilateral skin lesions on the palms and the soles is uncommon. However, any open skin lesion can result in secondary bacterial infection. The pustules of PPP are often sterile and do not require bacterial culture; however, additional symptoms of fever, purulence, warmth, and worsening of symptoms may prompt further evaluation for a bacterial origin or complication.
Management
Due to limited quality data on treatment recommendations, the treatment options for PPP vary greatly. Most studies recommend topical versus systemic therapy for initial management.1-2,8,10-11 Firstline therapy often consists of topical corticosteroids and occlusive dressings, followed by oral retinoids (acitretin, alitretinoin) or photochemotherapy.1,8 Third-line therapy can include immunosuppressants (ciclosporin, methotrexate) or biologics (secukinumab).1,12 Recent data have shown positive results with vitamin D3 analogs (maxacalcitol, betamethasone butyrate propionate) as monotherapy or in combination with corticosteroids.10-11 Duration of therapy ranges from 4 to 8 weeks throughout the literature, depending on severity; however, many patients see improvement in the first few weeks.
Conservative measures to maintain remission include smoking cessation, skin emollients, and avoidance of irritants. It is important to educate patients about the chronicity of the disease and early treatment to prevent secondary infection or significant impact on quality of life.
OUTCOME FOR THE CASE PATIENT
The patient was prescribed triamcinolone acetonide (0.5% ointment applied bid), to be used until symptoms improved. After 1 week of treatment, she confirmed (verbally) that symptoms had resolved. She declined a follow-up visit or referral to dermatology.
CONCLUSION
Although PPP is fairly uncommon, it is important for clinicians to consider this diagnosis in patients presenting with localized rash on their palms and soles. This debilitating condition greatly affects a patient’s quality of life and, although it is chronic in nature, available treatments described in the literature have shown success in both acute resolution and ongoing remission of the disease.
1. Sevrain M, Richard M-A, Barnetche T, et al. Treatment for palmoplantar pustular psoriasis: systematic literature review, evidence-based recommendations and expert opinion. J Eur Acad Dermatol Venereol. 2014;28(suppl 5):13-16.
2. Olazagasti JM, Ma JE, Wetter DA. Clinical features, etiological factors, associated disorders, and treatment of palmoplantar pustulosis: the Mayo Clinic experience, 1996-2013. Mayo Clin Proc. 2017;92(9):1351-1358.
3. Bissonnette R, Suárez-Fariñas M, Li X, et al. Based on molecular profiling of gene expression, palmoplantar pustulosis and palmoplantar pustular psoriasis are highly related diseases that appear to be distinct from psoriasis vulgaris. PLoS One. 2016;11(5):1-11.
4. Raposo I, Torres T. Palmoplantar psoriasis and palmoplantar pustulosis: current treatment and future prospects. Am J Clin Dermatol. 2016;17(4):349-358.
5. Bissonnette R, Fuentes-Duculan J, Mashiko S, et al. Palmoplantar pustular psoriasis (PPPP) is characterized by activation of the IL-17A pathway. J Dermatol Sci. 2017;85(1):20-26.
6. Misiak-Galazka M, Wolska H, Rudnicka L. What do we know about palmoplantar pustulosis? J Eur Acad Dermatol Venereol. 2017;31(1):38-44.
7. Brunasso AMG, Puntoni M, Aberer W, et al. Clinical and epidemiological comparison of patients affected by palmoplantar plaque psoriasis and palmoplantar pustulosis: a case series study. Br J Dermatol. 2013;168(6):1243-1251.
8. Engin B, As¸kın Ö, Tüzün Y. Palmoplantar psoriasis. Clin Dermatol. 2017; 35(1):19-27.
9. Chung J, Callas Duffin K, Takeshita J, et al. Palmoplantar psoriasis is associated with greater impairment of health-related quality of life compared to moderate-to-severe plaque psoriasis. J Am Acad Dermatol. 2014;71(4):623-632.
10. Umezawa Y, Nakagawa H, Tamaki K. Phase III clinical study of maxacalcitol ointment in patients with palmoplantar pustulosis: a randomized, double-blind, placebo-controlled trial. J Dermatol. 2016;43(3):288-293.
11. Muro M, Kawakami H, Matsumoto Y, et al. Topical combination therapy with vitamin D3 and corticosteroid ointment for palmoplantar pustulosis: a prospective, randomized, left-right comparison study. J Dermatolog Treat. 2016;27(1):51-53.
12. Gottlieb A, Sullivan J, van Doorn M, et al. Secukinumab shows significant efficacy in palmoplantar psoriasis: results from GESTURE, a randomized controlled trial. J Am Acad Dermatol. 2017;76(1):70-80.
1. Sevrain M, Richard M-A, Barnetche T, et al. Treatment for palmoplantar pustular psoriasis: systematic literature review, evidence-based recommendations and expert opinion. J Eur Acad Dermatol Venereol. 2014;28(suppl 5):13-16.
2. Olazagasti JM, Ma JE, Wetter DA. Clinical features, etiological factors, associated disorders, and treatment of palmoplantar pustulosis: the Mayo Clinic experience, 1996-2013. Mayo Clin Proc. 2017;92(9):1351-1358.
3. Bissonnette R, Suárez-Fariñas M, Li X, et al. Based on molecular profiling of gene expression, palmoplantar pustulosis and palmoplantar pustular psoriasis are highly related diseases that appear to be distinct from psoriasis vulgaris. PLoS One. 2016;11(5):1-11.
4. Raposo I, Torres T. Palmoplantar psoriasis and palmoplantar pustulosis: current treatment and future prospects. Am J Clin Dermatol. 2016;17(4):349-358.
5. Bissonnette R, Fuentes-Duculan J, Mashiko S, et al. Palmoplantar pustular psoriasis (PPPP) is characterized by activation of the IL-17A pathway. J Dermatol Sci. 2017;85(1):20-26.
6. Misiak-Galazka M, Wolska H, Rudnicka L. What do we know about palmoplantar pustulosis? J Eur Acad Dermatol Venereol. 2017;31(1):38-44.
7. Brunasso AMG, Puntoni M, Aberer W, et al. Clinical and epidemiological comparison of patients affected by palmoplantar plaque psoriasis and palmoplantar pustulosis: a case series study. Br J Dermatol. 2013;168(6):1243-1251.
8. Engin B, As¸kın Ö, Tüzün Y. Palmoplantar psoriasis. Clin Dermatol. 2017; 35(1):19-27.
9. Chung J, Callas Duffin K, Takeshita J, et al. Palmoplantar psoriasis is associated with greater impairment of health-related quality of life compared to moderate-to-severe plaque psoriasis. J Am Acad Dermatol. 2014;71(4):623-632.
10. Umezawa Y, Nakagawa H, Tamaki K. Phase III clinical study of maxacalcitol ointment in patients with palmoplantar pustulosis: a randomized, double-blind, placebo-controlled trial. J Dermatol. 2016;43(3):288-293.
11. Muro M, Kawakami H, Matsumoto Y, et al. Topical combination therapy with vitamin D3 and corticosteroid ointment for palmoplantar pustulosis: a prospective, randomized, left-right comparison study. J Dermatolog Treat. 2016;27(1):51-53.
12. Gottlieb A, Sullivan J, van Doorn M, et al. Secukinumab shows significant efficacy in palmoplantar psoriasis: results from GESTURE, a randomized controlled trial. J Am Acad Dermatol. 2017;76(1):70-80.