User login
Don’t discount ultrapotent topical corticosteroids for vulvar lichen sclerosus
according to an expert speaking at the virtual conference on diseases of the vulva and vagina.
If needed, intralesional steroid injections or calcineurin inhibitors can be added to a topical corticosteroid regimen, Libby Edwards, MD, suggested at the meeting, hosted by the International Society for the Study of Vulvovaginal Disease. In addition, early reports indicate that newer interventions such as fractional CO2 laser treatments may help patients with refractory disease.
Still, “there is no question, there is no argument: First-, second- and third-line treatment for lichen sclerosus is an ultrapotent or superpotent topical corticosteroid,” she said. Steroids include halobetasol, clobetasol, or betamethasone dipropionate in augmented vehicle ointment once or twice per day. Patients should continue this regimen until the skin texture is normal or the disease is controlled as well as possible, which usually takes several months, said Dr. Edwards, of Southeast Vulvar Clinic in Charlotte, N.C.
Patients then should continue treatment, but less frequently or with a lower potency steroid.
Although corticosteroids are not Food and Drug Administration–approved for the treatment of lichen sclerosus, double-blind, placebo-controlled trials support their use, Dr. Edwards said.
Getting patients to use topical corticosteroids as directed can be a challenge, however, and patient education is crucial.
After about 10 days, many patients start to feel better and stop the medication prematurely, which may lead to recurrence.
“That is such an important counseling point,” Aruna Venkatesan, MD, chief of dermatology and director of the genital dermatology clinic at Santa Clara Valley Medical Center in San Jose, Calif., said during a panel discussion. “Tell them, listen, I may not see you back for a couple months, and you may start feeling better sooner. But I want you to keep using this at this frequency so that when you come back we can make a good decision about whether you’re ready” for a lower potency regimen.
To encourage daily use, Hope K. Haefner, MD, asks patients whether they brush their teeth every night. “When they say yes, I tell them to put the steroid ointment by their toothpaste and use it after brushing,” Dr. Haefner, the Harold A. Furlong Professor of Women’s Health at Michigan Medicine in Ann Arbor, said during the discussion. “But don’t mix up the tubes.”
Once lichen sclerosus is controlled, options include decreasing the superpotent steroid to once, three times per week or changing to a midpotency steroid such as triamcinolone ointment every day, Dr. Edwards said.
Evidence suggests that controlling lichen sclerosus may prevent squamous cell carcinoma and scarring. In a study of more than 500 patients, about 70% complied with treatment instructions, whereas about 30% were considered partially compliant (JAMA Dermatol. 2015 Oct;151[10]:1061-7.). Patients who adhered to their therapy were less likely to have cancer or ongoing scarring during an average of 4.7 years of follow-up.
Beyond topical steroids
“Almost always, topical steroids are all you need,” said Dr. Edwards. “Before I go beyond that, I think of other issues that may be causing symptoms,” such as atrophic vagina, steroid dermatitis, or vulvodynia.
For patients with refractory lichen sclerosus, other treatments “can add more oomph to your topical steroid, but they are not better,” she said.
Intralesional corticosteroid injections are one option.
Another option is adding a calcineurin inhibitor such as tacrolimus or pimecrolimus, although these medications can burn with application and irritate. In addition, they carry warnings about rare cases of cancer associated with their use.
Dr. Edwards also uses methotrexate, which is supported by case reports and an open-label study. In a recently published study that included 21 patients with vulvar lichen sclerosus and 24 patients with extragenital lichen sclerosus, about half improved after receiving methotrexate (Dermatol Ther. 2020 Apr 29;e13473.).
What about lasers?
Fractional CO2 laser treatments, which are pulsed to minimize damage from heat, have “a lot of providers very excited,” Dr. Edwards said. In one open-label study of 40 patients, most reported a decrease in symptoms. (J Low Genit Tract Dis. 2020 Apr;24[2]:225-8.)
“We’re awaiting blinded, controlled studies,” Dr. Edwards said. “We don’t have those available yet although they are in progress.”
Ten of Dr. Edwards’ patients who did not improve enough with medication have received laser treatments. One patient stopped laser therapy after one treatment. One did not improve. Two were completely cleared, and six had significant improvement.
If patients who improved stopped steroids against recommendations, lichen sclerosus recurred, Dr. Edwards said.
The ISSVD does not recommend laser for the routine treatment of lichen sclerosus because of a lack of adequate studies and long-term safety data and biologic implausibility, Dr. Edwards noted (J Low Genit Tract Dis. 2019 Apr;23[2]:151-60.) Laser treatments for lichen sclerosus should not be used outside of clinical trials or without special arrangements for clinical governance, consent, and audit, according to a consensus document from the society.
“I mostly agree with that,” Dr. Edwards said. “But I now think that this is a reasonable thing to use when other treatments have been exhausted.”
Dr. Edwards and Dr. Venkatesan had no conflicts of interest. Dr. Haefner is an author for UpToDate.
according to an expert speaking at the virtual conference on diseases of the vulva and vagina.
If needed, intralesional steroid injections or calcineurin inhibitors can be added to a topical corticosteroid regimen, Libby Edwards, MD, suggested at the meeting, hosted by the International Society for the Study of Vulvovaginal Disease. In addition, early reports indicate that newer interventions such as fractional CO2 laser treatments may help patients with refractory disease.
Still, “there is no question, there is no argument: First-, second- and third-line treatment for lichen sclerosus is an ultrapotent or superpotent topical corticosteroid,” she said. Steroids include halobetasol, clobetasol, or betamethasone dipropionate in augmented vehicle ointment once or twice per day. Patients should continue this regimen until the skin texture is normal or the disease is controlled as well as possible, which usually takes several months, said Dr. Edwards, of Southeast Vulvar Clinic in Charlotte, N.C.
Patients then should continue treatment, but less frequently or with a lower potency steroid.
Although corticosteroids are not Food and Drug Administration–approved for the treatment of lichen sclerosus, double-blind, placebo-controlled trials support their use, Dr. Edwards said.
Getting patients to use topical corticosteroids as directed can be a challenge, however, and patient education is crucial.
After about 10 days, many patients start to feel better and stop the medication prematurely, which may lead to recurrence.
“That is such an important counseling point,” Aruna Venkatesan, MD, chief of dermatology and director of the genital dermatology clinic at Santa Clara Valley Medical Center in San Jose, Calif., said during a panel discussion. “Tell them, listen, I may not see you back for a couple months, and you may start feeling better sooner. But I want you to keep using this at this frequency so that when you come back we can make a good decision about whether you’re ready” for a lower potency regimen.
To encourage daily use, Hope K. Haefner, MD, asks patients whether they brush their teeth every night. “When they say yes, I tell them to put the steroid ointment by their toothpaste and use it after brushing,” Dr. Haefner, the Harold A. Furlong Professor of Women’s Health at Michigan Medicine in Ann Arbor, said during the discussion. “But don’t mix up the tubes.”
Once lichen sclerosus is controlled, options include decreasing the superpotent steroid to once, three times per week or changing to a midpotency steroid such as triamcinolone ointment every day, Dr. Edwards said.
Evidence suggests that controlling lichen sclerosus may prevent squamous cell carcinoma and scarring. In a study of more than 500 patients, about 70% complied with treatment instructions, whereas about 30% were considered partially compliant (JAMA Dermatol. 2015 Oct;151[10]:1061-7.). Patients who adhered to their therapy were less likely to have cancer or ongoing scarring during an average of 4.7 years of follow-up.
Beyond topical steroids
“Almost always, topical steroids are all you need,” said Dr. Edwards. “Before I go beyond that, I think of other issues that may be causing symptoms,” such as atrophic vagina, steroid dermatitis, or vulvodynia.
For patients with refractory lichen sclerosus, other treatments “can add more oomph to your topical steroid, but they are not better,” she said.
Intralesional corticosteroid injections are one option.
Another option is adding a calcineurin inhibitor such as tacrolimus or pimecrolimus, although these medications can burn with application and irritate. In addition, they carry warnings about rare cases of cancer associated with their use.
Dr. Edwards also uses methotrexate, which is supported by case reports and an open-label study. In a recently published study that included 21 patients with vulvar lichen sclerosus and 24 patients with extragenital lichen sclerosus, about half improved after receiving methotrexate (Dermatol Ther. 2020 Apr 29;e13473.).
What about lasers?
Fractional CO2 laser treatments, which are pulsed to minimize damage from heat, have “a lot of providers very excited,” Dr. Edwards said. In one open-label study of 40 patients, most reported a decrease in symptoms. (J Low Genit Tract Dis. 2020 Apr;24[2]:225-8.)
“We’re awaiting blinded, controlled studies,” Dr. Edwards said. “We don’t have those available yet although they are in progress.”
Ten of Dr. Edwards’ patients who did not improve enough with medication have received laser treatments. One patient stopped laser therapy after one treatment. One did not improve. Two were completely cleared, and six had significant improvement.
If patients who improved stopped steroids against recommendations, lichen sclerosus recurred, Dr. Edwards said.
The ISSVD does not recommend laser for the routine treatment of lichen sclerosus because of a lack of adequate studies and long-term safety data and biologic implausibility, Dr. Edwards noted (J Low Genit Tract Dis. 2019 Apr;23[2]:151-60.) Laser treatments for lichen sclerosus should not be used outside of clinical trials or without special arrangements for clinical governance, consent, and audit, according to a consensus document from the society.
“I mostly agree with that,” Dr. Edwards said. “But I now think that this is a reasonable thing to use when other treatments have been exhausted.”
Dr. Edwards and Dr. Venkatesan had no conflicts of interest. Dr. Haefner is an author for UpToDate.
according to an expert speaking at the virtual conference on diseases of the vulva and vagina.
If needed, intralesional steroid injections or calcineurin inhibitors can be added to a topical corticosteroid regimen, Libby Edwards, MD, suggested at the meeting, hosted by the International Society for the Study of Vulvovaginal Disease. In addition, early reports indicate that newer interventions such as fractional CO2 laser treatments may help patients with refractory disease.
Still, “there is no question, there is no argument: First-, second- and third-line treatment for lichen sclerosus is an ultrapotent or superpotent topical corticosteroid,” she said. Steroids include halobetasol, clobetasol, or betamethasone dipropionate in augmented vehicle ointment once or twice per day. Patients should continue this regimen until the skin texture is normal or the disease is controlled as well as possible, which usually takes several months, said Dr. Edwards, of Southeast Vulvar Clinic in Charlotte, N.C.
Patients then should continue treatment, but less frequently or with a lower potency steroid.
Although corticosteroids are not Food and Drug Administration–approved for the treatment of lichen sclerosus, double-blind, placebo-controlled trials support their use, Dr. Edwards said.
Getting patients to use topical corticosteroids as directed can be a challenge, however, and patient education is crucial.
After about 10 days, many patients start to feel better and stop the medication prematurely, which may lead to recurrence.
“That is such an important counseling point,” Aruna Venkatesan, MD, chief of dermatology and director of the genital dermatology clinic at Santa Clara Valley Medical Center in San Jose, Calif., said during a panel discussion. “Tell them, listen, I may not see you back for a couple months, and you may start feeling better sooner. But I want you to keep using this at this frequency so that when you come back we can make a good decision about whether you’re ready” for a lower potency regimen.
To encourage daily use, Hope K. Haefner, MD, asks patients whether they brush their teeth every night. “When they say yes, I tell them to put the steroid ointment by their toothpaste and use it after brushing,” Dr. Haefner, the Harold A. Furlong Professor of Women’s Health at Michigan Medicine in Ann Arbor, said during the discussion. “But don’t mix up the tubes.”
Once lichen sclerosus is controlled, options include decreasing the superpotent steroid to once, three times per week or changing to a midpotency steroid such as triamcinolone ointment every day, Dr. Edwards said.
Evidence suggests that controlling lichen sclerosus may prevent squamous cell carcinoma and scarring. In a study of more than 500 patients, about 70% complied with treatment instructions, whereas about 30% were considered partially compliant (JAMA Dermatol. 2015 Oct;151[10]:1061-7.). Patients who adhered to their therapy were less likely to have cancer or ongoing scarring during an average of 4.7 years of follow-up.
Beyond topical steroids
“Almost always, topical steroids are all you need,” said Dr. Edwards. “Before I go beyond that, I think of other issues that may be causing symptoms,” such as atrophic vagina, steroid dermatitis, or vulvodynia.
For patients with refractory lichen sclerosus, other treatments “can add more oomph to your topical steroid, but they are not better,” she said.
Intralesional corticosteroid injections are one option.
Another option is adding a calcineurin inhibitor such as tacrolimus or pimecrolimus, although these medications can burn with application and irritate. In addition, they carry warnings about rare cases of cancer associated with their use.
Dr. Edwards also uses methotrexate, which is supported by case reports and an open-label study. In a recently published study that included 21 patients with vulvar lichen sclerosus and 24 patients with extragenital lichen sclerosus, about half improved after receiving methotrexate (Dermatol Ther. 2020 Apr 29;e13473.).
What about lasers?
Fractional CO2 laser treatments, which are pulsed to minimize damage from heat, have “a lot of providers very excited,” Dr. Edwards said. In one open-label study of 40 patients, most reported a decrease in symptoms. (J Low Genit Tract Dis. 2020 Apr;24[2]:225-8.)
“We’re awaiting blinded, controlled studies,” Dr. Edwards said. “We don’t have those available yet although they are in progress.”
Ten of Dr. Edwards’ patients who did not improve enough with medication have received laser treatments. One patient stopped laser therapy after one treatment. One did not improve. Two were completely cleared, and six had significant improvement.
If patients who improved stopped steroids against recommendations, lichen sclerosus recurred, Dr. Edwards said.
The ISSVD does not recommend laser for the routine treatment of lichen sclerosus because of a lack of adequate studies and long-term safety data and biologic implausibility, Dr. Edwards noted (J Low Genit Tract Dis. 2019 Apr;23[2]:151-60.) Laser treatments for lichen sclerosus should not be used outside of clinical trials or without special arrangements for clinical governance, consent, and audit, according to a consensus document from the society.
“I mostly agree with that,” Dr. Edwards said. “But I now think that this is a reasonable thing to use when other treatments have been exhausted.”
Dr. Edwards and Dr. Venkatesan had no conflicts of interest. Dr. Haefner is an author for UpToDate.
EXPERT ANALYSIS FROM THE ISSVD BIENNIAL CONFERENCE
New lupus classification criteria perform well in children, young adults
, according to results from a single-center, retrospective study.
However, the 2019 criteria, which were developed using cohorts of adult patients with SLE, were statistically no better than the 1997 ACR criteria at identifying those without the disease, first author Najla Aljaberi, MBBS, of the Cincinnati Children’s Hospital Medical Center, and colleagues reported in Arthritis Care & Research.
The 2019 criteria were especially good at correctly classifying SLE in non-White youths, but the two sets of criteria performed equally well among male and female youths with SLE and across age groups.
“Our study confirms superior sensitivity of the new criteria over the 1997-ACR criteria in youths with SLE. The difference in sensitivity estimates between the two criteria sets (2019-EULAR/ACR vs. 1997-ACR) may be explained by a higher weight being assigned to immunologic criteria, less strict hematologic criteria (not requiring >2 occurrences), and the inclusion of subjective features of arthritis. Notably, our estimates of the sensitivity of the 2019-EULAR/ACR criteria were similar to those reported from a Brazilian pediatric study by Fonseca et al. (87.7%) that also used physician diagnosis as reference standard,” the researchers wrote.
Dr. Aljaberi and colleagues reviewed electronic medical records of 112 patients with SLE aged 2-21 years and 105 controls aged 1-19 years at Cincinnati Children’s Hospital Medical Center during 2008-2019. Patients identified in the records at the center were considered to have SLE based on ICD-10 codes assigned by experienced pediatric rheumatologists. The control patients included 69 (66%) with juvenile dermatomyositis and 36 with juvenile scleroderma/systemic sclerosis, based on corresponding ICD-10 codes.
Among the SLE cases, 57% were White and 81% were female, while Whites represented 83% and females 71% of control patients. Young adults aged 18-21 years represented a minority of SLE cases (18%) and controls (7%).
The 2019 criteria had significantly higher sensitivity than did the 1997 criteria (85% vs. 72%, respectively; P = .023) but similar specificity (83% vs. 87%; P = .456). A total of 17 out of the 112 SLE cases failed to meet the 2019 criteria, 13 (76%) of whom were White. Overall, 31 SLE cases did not meet the 1997 criteria, but 15 of those fulfilled the 2019 criteria. While there was no statistically significant difference in the sensitivity of the 2019 criteria between non-White and White cases (92% vs. 80%, respectively; P = .08), the difference in sensitivity was significant with the 1997 criteria (83% vs. 64%; P < .02).
The 2019 criteria had similar sensitivity in males and females (86% vs. 81%, respectively), as well as specificity (81% vs. 87%). The 1997 criteria also provided similar sensitivity between males and females (71% vs. 76%) as well as specificity (85% vs. 90%).
In only four instances did SLE cases meet 2019 criteria before ICD-10 diagnosis of SLE, whereas in the other 108 cases the ICD-10 diagnosis coincided with reaching the threshold for meeting 2019 criteria.
There was no funding secured for the study, and the authors had no conflicts of interest to disclose.
SOURCE: Aljaberi N et al. Arthritis Care Res. 2020 Aug 25. doi: 10.1002/acr.24430.
, according to results from a single-center, retrospective study.
However, the 2019 criteria, which were developed using cohorts of adult patients with SLE, were statistically no better than the 1997 ACR criteria at identifying those without the disease, first author Najla Aljaberi, MBBS, of the Cincinnati Children’s Hospital Medical Center, and colleagues reported in Arthritis Care & Research.
The 2019 criteria were especially good at correctly classifying SLE in non-White youths, but the two sets of criteria performed equally well among male and female youths with SLE and across age groups.
“Our study confirms superior sensitivity of the new criteria over the 1997-ACR criteria in youths with SLE. The difference in sensitivity estimates between the two criteria sets (2019-EULAR/ACR vs. 1997-ACR) may be explained by a higher weight being assigned to immunologic criteria, less strict hematologic criteria (not requiring >2 occurrences), and the inclusion of subjective features of arthritis. Notably, our estimates of the sensitivity of the 2019-EULAR/ACR criteria were similar to those reported from a Brazilian pediatric study by Fonseca et al. (87.7%) that also used physician diagnosis as reference standard,” the researchers wrote.
Dr. Aljaberi and colleagues reviewed electronic medical records of 112 patients with SLE aged 2-21 years and 105 controls aged 1-19 years at Cincinnati Children’s Hospital Medical Center during 2008-2019. Patients identified in the records at the center were considered to have SLE based on ICD-10 codes assigned by experienced pediatric rheumatologists. The control patients included 69 (66%) with juvenile dermatomyositis and 36 with juvenile scleroderma/systemic sclerosis, based on corresponding ICD-10 codes.
Among the SLE cases, 57% were White and 81% were female, while Whites represented 83% and females 71% of control patients. Young adults aged 18-21 years represented a minority of SLE cases (18%) and controls (7%).
The 2019 criteria had significantly higher sensitivity than did the 1997 criteria (85% vs. 72%, respectively; P = .023) but similar specificity (83% vs. 87%; P = .456). A total of 17 out of the 112 SLE cases failed to meet the 2019 criteria, 13 (76%) of whom were White. Overall, 31 SLE cases did not meet the 1997 criteria, but 15 of those fulfilled the 2019 criteria. While there was no statistically significant difference in the sensitivity of the 2019 criteria between non-White and White cases (92% vs. 80%, respectively; P = .08), the difference in sensitivity was significant with the 1997 criteria (83% vs. 64%; P < .02).
The 2019 criteria had similar sensitivity in males and females (86% vs. 81%, respectively), as well as specificity (81% vs. 87%). The 1997 criteria also provided similar sensitivity between males and females (71% vs. 76%) as well as specificity (85% vs. 90%).
In only four instances did SLE cases meet 2019 criteria before ICD-10 diagnosis of SLE, whereas in the other 108 cases the ICD-10 diagnosis coincided with reaching the threshold for meeting 2019 criteria.
There was no funding secured for the study, and the authors had no conflicts of interest to disclose.
SOURCE: Aljaberi N et al. Arthritis Care Res. 2020 Aug 25. doi: 10.1002/acr.24430.
, according to results from a single-center, retrospective study.
However, the 2019 criteria, which were developed using cohorts of adult patients with SLE, were statistically no better than the 1997 ACR criteria at identifying those without the disease, first author Najla Aljaberi, MBBS, of the Cincinnati Children’s Hospital Medical Center, and colleagues reported in Arthritis Care & Research.
The 2019 criteria were especially good at correctly classifying SLE in non-White youths, but the two sets of criteria performed equally well among male and female youths with SLE and across age groups.
“Our study confirms superior sensitivity of the new criteria over the 1997-ACR criteria in youths with SLE. The difference in sensitivity estimates between the two criteria sets (2019-EULAR/ACR vs. 1997-ACR) may be explained by a higher weight being assigned to immunologic criteria, less strict hematologic criteria (not requiring >2 occurrences), and the inclusion of subjective features of arthritis. Notably, our estimates of the sensitivity of the 2019-EULAR/ACR criteria were similar to those reported from a Brazilian pediatric study by Fonseca et al. (87.7%) that also used physician diagnosis as reference standard,” the researchers wrote.
Dr. Aljaberi and colleagues reviewed electronic medical records of 112 patients with SLE aged 2-21 years and 105 controls aged 1-19 years at Cincinnati Children’s Hospital Medical Center during 2008-2019. Patients identified in the records at the center were considered to have SLE based on ICD-10 codes assigned by experienced pediatric rheumatologists. The control patients included 69 (66%) with juvenile dermatomyositis and 36 with juvenile scleroderma/systemic sclerosis, based on corresponding ICD-10 codes.
Among the SLE cases, 57% were White and 81% were female, while Whites represented 83% and females 71% of control patients. Young adults aged 18-21 years represented a minority of SLE cases (18%) and controls (7%).
The 2019 criteria had significantly higher sensitivity than did the 1997 criteria (85% vs. 72%, respectively; P = .023) but similar specificity (83% vs. 87%; P = .456). A total of 17 out of the 112 SLE cases failed to meet the 2019 criteria, 13 (76%) of whom were White. Overall, 31 SLE cases did not meet the 1997 criteria, but 15 of those fulfilled the 2019 criteria. While there was no statistically significant difference in the sensitivity of the 2019 criteria between non-White and White cases (92% vs. 80%, respectively; P = .08), the difference in sensitivity was significant with the 1997 criteria (83% vs. 64%; P < .02).
The 2019 criteria had similar sensitivity in males and females (86% vs. 81%, respectively), as well as specificity (81% vs. 87%). The 1997 criteria also provided similar sensitivity between males and females (71% vs. 76%) as well as specificity (85% vs. 90%).
In only four instances did SLE cases meet 2019 criteria before ICD-10 diagnosis of SLE, whereas in the other 108 cases the ICD-10 diagnosis coincided with reaching the threshold for meeting 2019 criteria.
There was no funding secured for the study, and the authors had no conflicts of interest to disclose.
SOURCE: Aljaberi N et al. Arthritis Care Res. 2020 Aug 25. doi: 10.1002/acr.24430.
FROM ARTHRITIS CARE & RESEARCH
Hidradenitis suppurativa therapy options should be patient guided
of their most challenging symptoms, according to an expert summary presented at the Skin of Color Update 2020.
“If your patient is only focused on the appearance of the lesions or the presence of sinus tracts, they might not think your treatment is working,” said Ginette A. Okoye, MD, professor and chair, department of dermatology, Howard University, Washington.
Instead, she advised working with patients to define priorities, allowing them to measure and appreciate improvement. The most difficult symptoms for one patient, such as pain or persistent abscess drainage, might not be the same for another.
There is a large array of treatment options for HS. These were once typically employed in stepwise manner, moving from steroids to hormonal therapies, antibiotics, and on to biologics and lasers, but Dr. Okoye reported that she layers on treatments, guided by patient priorities and responses. “Most of my patients are not on just one treatment at a time,” she said.
In addition to patient goals, her treatment choices are also influenced by the presence of comorbidities such as metabolic syndrome, polycystic ovarian syndrome (PCOS), or inflammatory bowel disease (IBD). For example, she reported she is more likely to include metformin among treatment options in patients with central obesity or insulin resistance, whereas she moves more quickly to a biologic for those with another systemic inflammatory disease such as IBD.
Although multiple factors appear to contribute to the symptoms of HS, the pathophysiology remains incompletely understood, but follicular occlusion is often “a primary inciting event,” Dr. Okoye said.
For this reason, laser hair removal can provide substantial benefit, she noted. Not only does it eliminate the occlusion, but the heat generated by the laser eliminates some of the pathogens, such as Porphyromonas gingivalis, associated with HS.
“Lasers work well for preventing new lesions from forming but also in making active lesions go away faster,” said Dr. Okoye, who relies on the Nd:YAG laser when treating this disease in darker skin. She has found lasers to be particularly effective in mild to moderate disease.
When using lasers, one challenge is third-party insurance, according to Dr. Okoye, who reported that she has tried repeatedly to convince payers that this treatment is medically indicated for HS, but claims have been routinely denied. As a result, she has had to significantly discount the cost of laser at her center in order to provide access to “a modality that actually works.”
Incision and drainage of inflamed painful lesions is a common intervention in HS, but Dr. Okoye discourages this approach. Because of the high recurrence rates, the benefits are temporary. Instead, she recommends an intralesional injection of triamcinolone acetonide diluted with equal amounts of lidocaine.
With this injection, “there is immediate pain relief followed by significant resolution of the inflammation,” she said. Because of the likelihood that patients seeking care in the emergency department for acutely inflamed lesions will receive surgical treatment, Dr. Okoye recommends offering patients urgent appointments for steroid injections when painful and inflamed lesions need immediate attention.
In contrast, marsupialization of abscesses or sinus tracts, often called deroofing, is associated with a relatively low risk of recurrence, can be done under local anesthesia in an office, and can lead to resolution of persistent nodules in patients with mild disease.
“This is an easy procedure that takes relatively little time,” advised Dr. Okoye, who provided CPT codes (10060 and 10061) that will provide reimbursement as long as procedural notes describe the rationale.
Metformin is an attractive adjunctive therapy for HS in patients with type 2 diabetes or features that suggest metabolic disturbances, such as central obesity, hypercholesterolemia, hypertension, or hypertriglyceridemia. It should also be considered in patients with PCOS because metformin decreases ovarian androgen production, she said.
When prescribing metformin in HS, which is an off-label indication, “I prefer the extended release formulation. It has a better profile in regard to gastrointestinal side effects and it can be taken once-daily,” Dr. Okoye said.
Citing a study that suggests patients with HS have even worse quality of life scores than do patients with diabetes, Dr. Okoye also emphasized the importance of psychosocial support and lifestyle modification as part of a holistic approach. With multiple manifestations of varying severity, individualizing therapy to control symptoms that the patient finds most bothersome is essential for optimizing patient well being.
Tien Viet Nguyen, MD, who practices dermatology and conducts clinical research in Bellevue, Wash., agrees that a comprehensive treatment program is needed. First author of a recent review article on HS, Dr. Nguyen agreed that common comorbidities like IBD, PCOS, and diabetes are accompanied frequently by a host of mental health and behavioral issues that contribute to impaired quality of life, such as depression, low self-esteem, sexual dysfunction, impaired sleep, and substance use disorders.
“Therefore, addressing these important comorbidities and quality of life issues with other health care professionals as a team is the best approach to improving health outcomes,” he said in an interview.
Dr. Nguyen also recently authored a chapter on quality of life issues associated with HS in the soon-to-be-published Comprehensive Guide to Hidradenitis Suppurativa (1st Edition, Dermatology Clinics). He agreed that optimal outcomes are achieved by an interdisciplinary team of health care providers who can address the sometimes independent but often interrelated comorbidities associated with this disorder.
Dr. Okoye has financial relationships with Pfizer and Unilver, but neither is relevant to this topic.
of their most challenging symptoms, according to an expert summary presented at the Skin of Color Update 2020.
“If your patient is only focused on the appearance of the lesions or the presence of sinus tracts, they might not think your treatment is working,” said Ginette A. Okoye, MD, professor and chair, department of dermatology, Howard University, Washington.
Instead, she advised working with patients to define priorities, allowing them to measure and appreciate improvement. The most difficult symptoms for one patient, such as pain or persistent abscess drainage, might not be the same for another.
There is a large array of treatment options for HS. These were once typically employed in stepwise manner, moving from steroids to hormonal therapies, antibiotics, and on to biologics and lasers, but Dr. Okoye reported that she layers on treatments, guided by patient priorities and responses. “Most of my patients are not on just one treatment at a time,” she said.
In addition to patient goals, her treatment choices are also influenced by the presence of comorbidities such as metabolic syndrome, polycystic ovarian syndrome (PCOS), or inflammatory bowel disease (IBD). For example, she reported she is more likely to include metformin among treatment options in patients with central obesity or insulin resistance, whereas she moves more quickly to a biologic for those with another systemic inflammatory disease such as IBD.
Although multiple factors appear to contribute to the symptoms of HS, the pathophysiology remains incompletely understood, but follicular occlusion is often “a primary inciting event,” Dr. Okoye said.
For this reason, laser hair removal can provide substantial benefit, she noted. Not only does it eliminate the occlusion, but the heat generated by the laser eliminates some of the pathogens, such as Porphyromonas gingivalis, associated with HS.
“Lasers work well for preventing new lesions from forming but also in making active lesions go away faster,” said Dr. Okoye, who relies on the Nd:YAG laser when treating this disease in darker skin. She has found lasers to be particularly effective in mild to moderate disease.
When using lasers, one challenge is third-party insurance, according to Dr. Okoye, who reported that she has tried repeatedly to convince payers that this treatment is medically indicated for HS, but claims have been routinely denied. As a result, she has had to significantly discount the cost of laser at her center in order to provide access to “a modality that actually works.”
Incision and drainage of inflamed painful lesions is a common intervention in HS, but Dr. Okoye discourages this approach. Because of the high recurrence rates, the benefits are temporary. Instead, she recommends an intralesional injection of triamcinolone acetonide diluted with equal amounts of lidocaine.
With this injection, “there is immediate pain relief followed by significant resolution of the inflammation,” she said. Because of the likelihood that patients seeking care in the emergency department for acutely inflamed lesions will receive surgical treatment, Dr. Okoye recommends offering patients urgent appointments for steroid injections when painful and inflamed lesions need immediate attention.
In contrast, marsupialization of abscesses or sinus tracts, often called deroofing, is associated with a relatively low risk of recurrence, can be done under local anesthesia in an office, and can lead to resolution of persistent nodules in patients with mild disease.
“This is an easy procedure that takes relatively little time,” advised Dr. Okoye, who provided CPT codes (10060 and 10061) that will provide reimbursement as long as procedural notes describe the rationale.
Metformin is an attractive adjunctive therapy for HS in patients with type 2 diabetes or features that suggest metabolic disturbances, such as central obesity, hypercholesterolemia, hypertension, or hypertriglyceridemia. It should also be considered in patients with PCOS because metformin decreases ovarian androgen production, she said.
When prescribing metformin in HS, which is an off-label indication, “I prefer the extended release formulation. It has a better profile in regard to gastrointestinal side effects and it can be taken once-daily,” Dr. Okoye said.
Citing a study that suggests patients with HS have even worse quality of life scores than do patients with diabetes, Dr. Okoye also emphasized the importance of psychosocial support and lifestyle modification as part of a holistic approach. With multiple manifestations of varying severity, individualizing therapy to control symptoms that the patient finds most bothersome is essential for optimizing patient well being.
Tien Viet Nguyen, MD, who practices dermatology and conducts clinical research in Bellevue, Wash., agrees that a comprehensive treatment program is needed. First author of a recent review article on HS, Dr. Nguyen agreed that common comorbidities like IBD, PCOS, and diabetes are accompanied frequently by a host of mental health and behavioral issues that contribute to impaired quality of life, such as depression, low self-esteem, sexual dysfunction, impaired sleep, and substance use disorders.
“Therefore, addressing these important comorbidities and quality of life issues with other health care professionals as a team is the best approach to improving health outcomes,” he said in an interview.
Dr. Nguyen also recently authored a chapter on quality of life issues associated with HS in the soon-to-be-published Comprehensive Guide to Hidradenitis Suppurativa (1st Edition, Dermatology Clinics). He agreed that optimal outcomes are achieved by an interdisciplinary team of health care providers who can address the sometimes independent but often interrelated comorbidities associated with this disorder.
Dr. Okoye has financial relationships with Pfizer and Unilver, but neither is relevant to this topic.
of their most challenging symptoms, according to an expert summary presented at the Skin of Color Update 2020.
“If your patient is only focused on the appearance of the lesions or the presence of sinus tracts, they might not think your treatment is working,” said Ginette A. Okoye, MD, professor and chair, department of dermatology, Howard University, Washington.
Instead, she advised working with patients to define priorities, allowing them to measure and appreciate improvement. The most difficult symptoms for one patient, such as pain or persistent abscess drainage, might not be the same for another.
There is a large array of treatment options for HS. These were once typically employed in stepwise manner, moving from steroids to hormonal therapies, antibiotics, and on to biologics and lasers, but Dr. Okoye reported that she layers on treatments, guided by patient priorities and responses. “Most of my patients are not on just one treatment at a time,” she said.
In addition to patient goals, her treatment choices are also influenced by the presence of comorbidities such as metabolic syndrome, polycystic ovarian syndrome (PCOS), or inflammatory bowel disease (IBD). For example, she reported she is more likely to include metformin among treatment options in patients with central obesity or insulin resistance, whereas she moves more quickly to a biologic for those with another systemic inflammatory disease such as IBD.
Although multiple factors appear to contribute to the symptoms of HS, the pathophysiology remains incompletely understood, but follicular occlusion is often “a primary inciting event,” Dr. Okoye said.
For this reason, laser hair removal can provide substantial benefit, she noted. Not only does it eliminate the occlusion, but the heat generated by the laser eliminates some of the pathogens, such as Porphyromonas gingivalis, associated with HS.
“Lasers work well for preventing new lesions from forming but also in making active lesions go away faster,” said Dr. Okoye, who relies on the Nd:YAG laser when treating this disease in darker skin. She has found lasers to be particularly effective in mild to moderate disease.
When using lasers, one challenge is third-party insurance, according to Dr. Okoye, who reported that she has tried repeatedly to convince payers that this treatment is medically indicated for HS, but claims have been routinely denied. As a result, she has had to significantly discount the cost of laser at her center in order to provide access to “a modality that actually works.”
Incision and drainage of inflamed painful lesions is a common intervention in HS, but Dr. Okoye discourages this approach. Because of the high recurrence rates, the benefits are temporary. Instead, she recommends an intralesional injection of triamcinolone acetonide diluted with equal amounts of lidocaine.
With this injection, “there is immediate pain relief followed by significant resolution of the inflammation,” she said. Because of the likelihood that patients seeking care in the emergency department for acutely inflamed lesions will receive surgical treatment, Dr. Okoye recommends offering patients urgent appointments for steroid injections when painful and inflamed lesions need immediate attention.
In contrast, marsupialization of abscesses or sinus tracts, often called deroofing, is associated with a relatively low risk of recurrence, can be done under local anesthesia in an office, and can lead to resolution of persistent nodules in patients with mild disease.
“This is an easy procedure that takes relatively little time,” advised Dr. Okoye, who provided CPT codes (10060 and 10061) that will provide reimbursement as long as procedural notes describe the rationale.
Metformin is an attractive adjunctive therapy for HS in patients with type 2 diabetes or features that suggest metabolic disturbances, such as central obesity, hypercholesterolemia, hypertension, or hypertriglyceridemia. It should also be considered in patients with PCOS because metformin decreases ovarian androgen production, she said.
When prescribing metformin in HS, which is an off-label indication, “I prefer the extended release formulation. It has a better profile in regard to gastrointestinal side effects and it can be taken once-daily,” Dr. Okoye said.
Citing a study that suggests patients with HS have even worse quality of life scores than do patients with diabetes, Dr. Okoye also emphasized the importance of psychosocial support and lifestyle modification as part of a holistic approach. With multiple manifestations of varying severity, individualizing therapy to control symptoms that the patient finds most bothersome is essential for optimizing patient well being.
Tien Viet Nguyen, MD, who practices dermatology and conducts clinical research in Bellevue, Wash., agrees that a comprehensive treatment program is needed. First author of a recent review article on HS, Dr. Nguyen agreed that common comorbidities like IBD, PCOS, and diabetes are accompanied frequently by a host of mental health and behavioral issues that contribute to impaired quality of life, such as depression, low self-esteem, sexual dysfunction, impaired sleep, and substance use disorders.
“Therefore, addressing these important comorbidities and quality of life issues with other health care professionals as a team is the best approach to improving health outcomes,” he said in an interview.
Dr. Nguyen also recently authored a chapter on quality of life issues associated with HS in the soon-to-be-published Comprehensive Guide to Hidradenitis Suppurativa (1st Edition, Dermatology Clinics). He agreed that optimal outcomes are achieved by an interdisciplinary team of health care providers who can address the sometimes independent but often interrelated comorbidities associated with this disorder.
Dr. Okoye has financial relationships with Pfizer and Unilver, but neither is relevant to this topic.
FROM SOC 2020
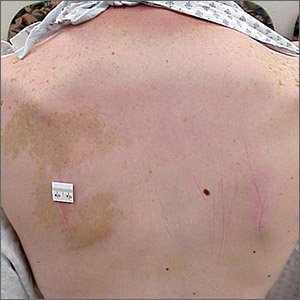
Hyperpigmented patch on the back
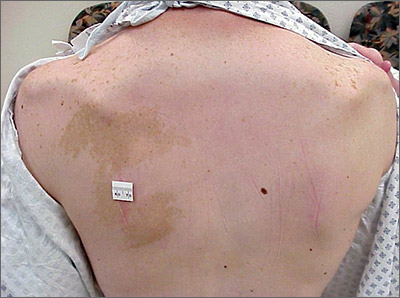
Large unilateral hyperpigmented patches on the trunk with onset around puberty are the hallmark of a Becker nevus, also more descriptively called pigmented hairy epidermal nevus.
Becker nevi are a form of epidermal nevus that usually occur on the upper back or chest. They most commonly develop during puberty when there are increasing circulating levels of androgens. (Becker nevus cells are androgen sensitive.) This is consistent with this patient’s history of the lesion developing in her teens when the lesions become hyperpigmented and noticeable. The localized androgen sensitivity also can lead to unilateral hypoplastic breast growth when it occurs on the chest in young women.
The lesions are more common in males than females and often have associated hypertrichosis. The etiology is not certain but is thought to be due to regional loss of heterozygosity during embryogenesis leading to the abnormally elevated levels of androgen receptors and increased androgen sensitivity in the basal keratinocytes and dermal fibroblasts.
Laser is the most effective therapy for the hyperpigmentation and for hypertrichosis when present. If a young woman with a Becker nevus has breast hypoplasia, spironolactone (an antiandrogen) has been helpful in restoring breast growth. For this patient, the hyperpigmented patch was asymptomatic and not troublesome, so she opted not to treat it.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Patel P, Malik K, Khachemoune A. Sebaceus and Becker’s nevus: overview of their presentation, pathogenesis, associations, and treatment. Am J Clin Dermatol. 2015;16:197-204.

Large unilateral hyperpigmented patches on the trunk with onset around puberty are the hallmark of a Becker nevus, also more descriptively called pigmented hairy epidermal nevus.
Becker nevi are a form of epidermal nevus that usually occur on the upper back or chest. They most commonly develop during puberty when there are increasing circulating levels of androgens. (Becker nevus cells are androgen sensitive.) This is consistent with this patient’s history of the lesion developing in her teens when the lesions become hyperpigmented and noticeable. The localized androgen sensitivity also can lead to unilateral hypoplastic breast growth when it occurs on the chest in young women.
The lesions are more common in males than females and often have associated hypertrichosis. The etiology is not certain but is thought to be due to regional loss of heterozygosity during embryogenesis leading to the abnormally elevated levels of androgen receptors and increased androgen sensitivity in the basal keratinocytes and dermal fibroblasts.
Laser is the most effective therapy for the hyperpigmentation and for hypertrichosis when present. If a young woman with a Becker nevus has breast hypoplasia, spironolactone (an antiandrogen) has been helpful in restoring breast growth. For this patient, the hyperpigmented patch was asymptomatic and not troublesome, so she opted not to treat it.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

Large unilateral hyperpigmented patches on the trunk with onset around puberty are the hallmark of a Becker nevus, also more descriptively called pigmented hairy epidermal nevus.
Becker nevi are a form of epidermal nevus that usually occur on the upper back or chest. They most commonly develop during puberty when there are increasing circulating levels of androgens. (Becker nevus cells are androgen sensitive.) This is consistent with this patient’s history of the lesion developing in her teens when the lesions become hyperpigmented and noticeable. The localized androgen sensitivity also can lead to unilateral hypoplastic breast growth when it occurs on the chest in young women.
The lesions are more common in males than females and often have associated hypertrichosis. The etiology is not certain but is thought to be due to regional loss of heterozygosity during embryogenesis leading to the abnormally elevated levels of androgen receptors and increased androgen sensitivity in the basal keratinocytes and dermal fibroblasts.
Laser is the most effective therapy for the hyperpigmentation and for hypertrichosis when present. If a young woman with a Becker nevus has breast hypoplasia, spironolactone (an antiandrogen) has been helpful in restoring breast growth. For this patient, the hyperpigmented patch was asymptomatic and not troublesome, so she opted not to treat it.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Patel P, Malik K, Khachemoune A. Sebaceus and Becker’s nevus: overview of their presentation, pathogenesis, associations, and treatment. Am J Clin Dermatol. 2015;16:197-204.
Patel P, Malik K, Khachemoune A. Sebaceus and Becker’s nevus: overview of their presentation, pathogenesis, associations, and treatment. Am J Clin Dermatol. 2015;16:197-204.
Expert spotlights recent advances in the medical treatment of acne
During the virtual annual Masters of Aesthetics Symposium, he highlighted the following new acne treatment options:
- Trifarotene cream 0.005% (Aklief). This marks the first new retinoid indicated for acne in several decades. It is indicated for the topical treatment of acne vulgaris in patients 9 years of age and older and has been studied in acne of the face, chest, and back. “It’s nice to have in our armamentarium,” he said.
- Tazarotene lotion 0.045% (Arazlo). The 0.1% formulation of tazarotene is commonly used for acne, but it can cause skin irritation, dryness, and erythema. The new 0.045% formulation was developed in a three-dimensional mesh matrix, with ingredients from an oil-in-water emulsion. “This allows for graduated dosing on the skin without as much irritation,” said Dr. Eichenfield, who is chief of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego.
- Minocycline 4% topical foam (Amzeeq). This marks the first and only topical minocycline prescription treatment for acne. “Its hydrophobic composition allows for stable and efficient delivery of inherently unstable pharmaceutical ingredients,” he said. “There is no evidence of photosensitivity as you’d expect from a minocycline-based product, and there are low systemic levels compared with oral minocycline.”
- Clascoterone cream 1% (Winlevi). This first-in-class topical androgen receptor inhibitor has been approved for the treatment of acne in patients 12 years and older. It competes with dihydrotestosterone and selectively targets androgen receptors in sebocytes and hair papilla cells. “It has been studied on the face and trunk and has been shown to inhibit sebum production, reduce secretion of inflammatory cytokines, and inhibit inflammatory pathways,” said Dr. Eichenfield, who is also professor of dermatology and pediatrics at the University of California, San Diego.
- From a systemic standpoint, sarecycline, a new tetracycline class antibiotic, has been approved for the treatment of inflammatory lesions of nonnodular moderate to severe acne vulgaris in patients 9 years and older. The once-daily drug can be taken with or without food in a weight-based dose. “This medicine appears to have a narrow spectrum of antibacterial activity compared with other tetracyclines,” he said. “It may have less of a negative effect on gut microbiome than traditional oral antibiotics.”
As for integrating these new options into existing clinical practice, Dr. Eichenfield predicts that the general approach to acne treatment will remain the same. “We’ll have to wait to see where the topical androgens fit into the treatment algorithms,” he said. “Our goal is to minimize scarring, minimize disease, and to modulate the disease course.”
Dr. Eichenfield disclosed that he has been an investigator and/or consultant for Almirall, Cassiopea, Dermata, Foamix, Galderma, L’Oreal, and Ortho Dermatologics.
During the virtual annual Masters of Aesthetics Symposium, he highlighted the following new acne treatment options:
- Trifarotene cream 0.005% (Aklief). This marks the first new retinoid indicated for acne in several decades. It is indicated for the topical treatment of acne vulgaris in patients 9 years of age and older and has been studied in acne of the face, chest, and back. “It’s nice to have in our armamentarium,” he said.
- Tazarotene lotion 0.045% (Arazlo). The 0.1% formulation of tazarotene is commonly used for acne, but it can cause skin irritation, dryness, and erythema. The new 0.045% formulation was developed in a three-dimensional mesh matrix, with ingredients from an oil-in-water emulsion. “This allows for graduated dosing on the skin without as much irritation,” said Dr. Eichenfield, who is chief of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego.
- Minocycline 4% topical foam (Amzeeq). This marks the first and only topical minocycline prescription treatment for acne. “Its hydrophobic composition allows for stable and efficient delivery of inherently unstable pharmaceutical ingredients,” he said. “There is no evidence of photosensitivity as you’d expect from a minocycline-based product, and there are low systemic levels compared with oral minocycline.”
- Clascoterone cream 1% (Winlevi). This first-in-class topical androgen receptor inhibitor has been approved for the treatment of acne in patients 12 years and older. It competes with dihydrotestosterone and selectively targets androgen receptors in sebocytes and hair papilla cells. “It has been studied on the face and trunk and has been shown to inhibit sebum production, reduce secretion of inflammatory cytokines, and inhibit inflammatory pathways,” said Dr. Eichenfield, who is also professor of dermatology and pediatrics at the University of California, San Diego.
- From a systemic standpoint, sarecycline, a new tetracycline class antibiotic, has been approved for the treatment of inflammatory lesions of nonnodular moderate to severe acne vulgaris in patients 9 years and older. The once-daily drug can be taken with or without food in a weight-based dose. “This medicine appears to have a narrow spectrum of antibacterial activity compared with other tetracyclines,” he said. “It may have less of a negative effect on gut microbiome than traditional oral antibiotics.”
As for integrating these new options into existing clinical practice, Dr. Eichenfield predicts that the general approach to acne treatment will remain the same. “We’ll have to wait to see where the topical androgens fit into the treatment algorithms,” he said. “Our goal is to minimize scarring, minimize disease, and to modulate the disease course.”
Dr. Eichenfield disclosed that he has been an investigator and/or consultant for Almirall, Cassiopea, Dermata, Foamix, Galderma, L’Oreal, and Ortho Dermatologics.
During the virtual annual Masters of Aesthetics Symposium, he highlighted the following new acne treatment options:
- Trifarotene cream 0.005% (Aklief). This marks the first new retinoid indicated for acne in several decades. It is indicated for the topical treatment of acne vulgaris in patients 9 years of age and older and has been studied in acne of the face, chest, and back. “It’s nice to have in our armamentarium,” he said.
- Tazarotene lotion 0.045% (Arazlo). The 0.1% formulation of tazarotene is commonly used for acne, but it can cause skin irritation, dryness, and erythema. The new 0.045% formulation was developed in a three-dimensional mesh matrix, with ingredients from an oil-in-water emulsion. “This allows for graduated dosing on the skin without as much irritation,” said Dr. Eichenfield, who is chief of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego.
- Minocycline 4% topical foam (Amzeeq). This marks the first and only topical minocycline prescription treatment for acne. “Its hydrophobic composition allows for stable and efficient delivery of inherently unstable pharmaceutical ingredients,” he said. “There is no evidence of photosensitivity as you’d expect from a minocycline-based product, and there are low systemic levels compared with oral minocycline.”
- Clascoterone cream 1% (Winlevi). This first-in-class topical androgen receptor inhibitor has been approved for the treatment of acne in patients 12 years and older. It competes with dihydrotestosterone and selectively targets androgen receptors in sebocytes and hair papilla cells. “It has been studied on the face and trunk and has been shown to inhibit sebum production, reduce secretion of inflammatory cytokines, and inhibit inflammatory pathways,” said Dr. Eichenfield, who is also professor of dermatology and pediatrics at the University of California, San Diego.
- From a systemic standpoint, sarecycline, a new tetracycline class antibiotic, has been approved for the treatment of inflammatory lesions of nonnodular moderate to severe acne vulgaris in patients 9 years and older. The once-daily drug can be taken with or without food in a weight-based dose. “This medicine appears to have a narrow spectrum of antibacterial activity compared with other tetracyclines,” he said. “It may have less of a negative effect on gut microbiome than traditional oral antibiotics.”
As for integrating these new options into existing clinical practice, Dr. Eichenfield predicts that the general approach to acne treatment will remain the same. “We’ll have to wait to see where the topical androgens fit into the treatment algorithms,” he said. “Our goal is to minimize scarring, minimize disease, and to modulate the disease course.”
Dr. Eichenfield disclosed that he has been an investigator and/or consultant for Almirall, Cassiopea, Dermata, Foamix, Galderma, L’Oreal, and Ortho Dermatologics.
FROM MOA 2020
Study highlights differences between White and Latino patients with psoriasis
in the same studies, according to new data presented at the virtual Skin of Color Update 2020.
“Our findings demonstrate that, though White psoriasis patients may have higher severity in certain body regions such as the trunk, axilla, and groin areas, Latino psoriasis patients have a greater distribution of involvement, particularly in their upper limbs,” reported Alyssa G. Ashbaugh, a third-year medical student at the University of California, Irvine.
The study also found that psoriasis had a greater adverse impact on well-being, as measured with the Dermatology Life Quality Index (DLQI). At entry into the trials from which these patients were drawn, the higher DLQI score, significantly lower quality of life, was nearly two times higher (13.78 vs. 7.31; P = .01) among the Latino patients, compared with White patients.
This is not the first study to show a greater negative impact from psoriasis on Latinos than Whites, according to Ms. Ashbaugh. For example, Latinos had the worse quality of life at baseline by DLQI score than White, Asians, or Black participants in a trial of etanercept that enrolled more than 2000 patients.
In this retrospective chart review, patient characteristics were evaluated in all 21 Latino patients enrolled in psoriasis clinical trials at the University of California, Irvine, in a recent period. They were matched by age and gender to an equal number of White patients participating in the same trials.
The mean age at diagnosis of psoriasis was older in the Latino group than in the White population (42.4 vs. 35.6 years; P = .20), but the difference did not reach statistical significance. The proportion of patients with severe disease on investigator global assessment was also greater but not significantly different in the Latino group, compared with the White group, respectively (42.9% vs. 28.6%; P = .10).
However, differences in the patterns of disease did reach significance. This included a lower mean Psoriasis Assessment Severity Index score of the trunk, axilla, and groin in Latinos (4.74 vs. 9.73; P = .02). But compared with White participants, Latinos had a higher mean percentage of body surface area involvement in the upper limbs (4.78 vs. 1.85; P = .004) and a higher percentage of total body surface area involvement (20.50 vs. 10.03; P = .02).
“While White patients were found to have lived many more years with psoriasis, it is important for future studies to examine whether this is due to earlier onset or delayed diagnosis, given the fact that minorities are less likely to have access to a dermatologist,” reported Ms. Ashbaugh, who performed this work under the guidance of the senior author, Natasha Mesinkovska, MD, PhD, with the department of dermatology, University of California, Irvine.
Overall, the study suggested that body surface coverage and severity is not similarly distributed in Latinos relative to Whites. Although Ms. Ashbaugh conceded that the small sample size and retrospective design of this study are important limitations, she believes that her study, along with previously published studies that suggest psoriasis characteristics may differ meaningfully by race or ethnicity, raises issues that should be explored in future studies designed to confirm differences and whether those differences should affect management.
Other studies have suggested “there are notable differences in the presentation of psoriasis between racial and ethnic groups with the Latino population often presenting to physicians with more severe psoriasis and increased body surface area involvement,” Ms. Ashbaugh noted. Although this appears to be one of the first studies to examine psoriasis characteristics in Latinos relative to Whites, she believes this is an area ripe for further analysis.
Psoriasis “is not a rare occurrence” in non-White populations even if U.S. data suggest that the prevalence in “people of color is lower than that of psoriasis in the U.S. white population,” Amy McMichael, MD, chair of the department of dermatology, Wake Forest Baptist Medical Center, Winston-Salem, N.C., commented in an interview after the meeting. She agreed that it cannot be assumed that psoriasis in skin of color has the same manifestations or responds to treatment in the same way as in White patients.
“Studies have suggested that lesion thickness and, often, extent of disease can be worse in patients of color. Few studies to date have examined the efficacy of treatments and impact of disease in these populations,” she said.
One exception was a study Dr. McMichael and colleagues published last year on the efficacy and safety of the interleukin-17 receptor A antagonist brodalumab for psoriasis in patients of color. The study showed that Black, Latino, and Asian patients participating in the AMAGINE-2 and AMAGINE-3 trials achieved similar outcomes as White participants.
“We published this study because this is one of the first, if not the first, to have enough patients of color to actually draw conclusions about the efficacy of the biologic as well as the patient-reported outcomes,” she explained.
Like the author of the evaluation of Latino patients undertaken at the University of California, Irvine, Dr. McMichael said studies of psoriasis specific to patients of color are needed.
“We cannot assume all patients of color will have the same outcomes as their Caucasian counterparts. It is imperative to include those of color in future psoriasis treatment trials in order to determine the efficacy of new medications,” she added, specifically calling for collection of data on patient-reported outcomes.
Ms. Ashbaugh has no relevant financial relationships to disclose. Dr. McMichael’s disclosures included serving as an investigator and/or consultant for companies that included Allergan, Procter & Gamble, Johnson & Johnson, and Aclaris.
in the same studies, according to new data presented at the virtual Skin of Color Update 2020.
“Our findings demonstrate that, though White psoriasis patients may have higher severity in certain body regions such as the trunk, axilla, and groin areas, Latino psoriasis patients have a greater distribution of involvement, particularly in their upper limbs,” reported Alyssa G. Ashbaugh, a third-year medical student at the University of California, Irvine.
The study also found that psoriasis had a greater adverse impact on well-being, as measured with the Dermatology Life Quality Index (DLQI). At entry into the trials from which these patients were drawn, the higher DLQI score, significantly lower quality of life, was nearly two times higher (13.78 vs. 7.31; P = .01) among the Latino patients, compared with White patients.
This is not the first study to show a greater negative impact from psoriasis on Latinos than Whites, according to Ms. Ashbaugh. For example, Latinos had the worse quality of life at baseline by DLQI score than White, Asians, or Black participants in a trial of etanercept that enrolled more than 2000 patients.
In this retrospective chart review, patient characteristics were evaluated in all 21 Latino patients enrolled in psoriasis clinical trials at the University of California, Irvine, in a recent period. They were matched by age and gender to an equal number of White patients participating in the same trials.
The mean age at diagnosis of psoriasis was older in the Latino group than in the White population (42.4 vs. 35.6 years; P = .20), but the difference did not reach statistical significance. The proportion of patients with severe disease on investigator global assessment was also greater but not significantly different in the Latino group, compared with the White group, respectively (42.9% vs. 28.6%; P = .10).
However, differences in the patterns of disease did reach significance. This included a lower mean Psoriasis Assessment Severity Index score of the trunk, axilla, and groin in Latinos (4.74 vs. 9.73; P = .02). But compared with White participants, Latinos had a higher mean percentage of body surface area involvement in the upper limbs (4.78 vs. 1.85; P = .004) and a higher percentage of total body surface area involvement (20.50 vs. 10.03; P = .02).
“While White patients were found to have lived many more years with psoriasis, it is important for future studies to examine whether this is due to earlier onset or delayed diagnosis, given the fact that minorities are less likely to have access to a dermatologist,” reported Ms. Ashbaugh, who performed this work under the guidance of the senior author, Natasha Mesinkovska, MD, PhD, with the department of dermatology, University of California, Irvine.
Overall, the study suggested that body surface coverage and severity is not similarly distributed in Latinos relative to Whites. Although Ms. Ashbaugh conceded that the small sample size and retrospective design of this study are important limitations, she believes that her study, along with previously published studies that suggest psoriasis characteristics may differ meaningfully by race or ethnicity, raises issues that should be explored in future studies designed to confirm differences and whether those differences should affect management.
Other studies have suggested “there are notable differences in the presentation of psoriasis between racial and ethnic groups with the Latino population often presenting to physicians with more severe psoriasis and increased body surface area involvement,” Ms. Ashbaugh noted. Although this appears to be one of the first studies to examine psoriasis characteristics in Latinos relative to Whites, she believes this is an area ripe for further analysis.
Psoriasis “is not a rare occurrence” in non-White populations even if U.S. data suggest that the prevalence in “people of color is lower than that of psoriasis in the U.S. white population,” Amy McMichael, MD, chair of the department of dermatology, Wake Forest Baptist Medical Center, Winston-Salem, N.C., commented in an interview after the meeting. She agreed that it cannot be assumed that psoriasis in skin of color has the same manifestations or responds to treatment in the same way as in White patients.
“Studies have suggested that lesion thickness and, often, extent of disease can be worse in patients of color. Few studies to date have examined the efficacy of treatments and impact of disease in these populations,” she said.
One exception was a study Dr. McMichael and colleagues published last year on the efficacy and safety of the interleukin-17 receptor A antagonist brodalumab for psoriasis in patients of color. The study showed that Black, Latino, and Asian patients participating in the AMAGINE-2 and AMAGINE-3 trials achieved similar outcomes as White participants.
“We published this study because this is one of the first, if not the first, to have enough patients of color to actually draw conclusions about the efficacy of the biologic as well as the patient-reported outcomes,” she explained.
Like the author of the evaluation of Latino patients undertaken at the University of California, Irvine, Dr. McMichael said studies of psoriasis specific to patients of color are needed.
“We cannot assume all patients of color will have the same outcomes as their Caucasian counterparts. It is imperative to include those of color in future psoriasis treatment trials in order to determine the efficacy of new medications,” she added, specifically calling for collection of data on patient-reported outcomes.
Ms. Ashbaugh has no relevant financial relationships to disclose. Dr. McMichael’s disclosures included serving as an investigator and/or consultant for companies that included Allergan, Procter & Gamble, Johnson & Johnson, and Aclaris.
in the same studies, according to new data presented at the virtual Skin of Color Update 2020.
“Our findings demonstrate that, though White psoriasis patients may have higher severity in certain body regions such as the trunk, axilla, and groin areas, Latino psoriasis patients have a greater distribution of involvement, particularly in their upper limbs,” reported Alyssa G. Ashbaugh, a third-year medical student at the University of California, Irvine.
The study also found that psoriasis had a greater adverse impact on well-being, as measured with the Dermatology Life Quality Index (DLQI). At entry into the trials from which these patients were drawn, the higher DLQI score, significantly lower quality of life, was nearly two times higher (13.78 vs. 7.31; P = .01) among the Latino patients, compared with White patients.
This is not the first study to show a greater negative impact from psoriasis on Latinos than Whites, according to Ms. Ashbaugh. For example, Latinos had the worse quality of life at baseline by DLQI score than White, Asians, or Black participants in a trial of etanercept that enrolled more than 2000 patients.
In this retrospective chart review, patient characteristics were evaluated in all 21 Latino patients enrolled in psoriasis clinical trials at the University of California, Irvine, in a recent period. They were matched by age and gender to an equal number of White patients participating in the same trials.
The mean age at diagnosis of psoriasis was older in the Latino group than in the White population (42.4 vs. 35.6 years; P = .20), but the difference did not reach statistical significance. The proportion of patients with severe disease on investigator global assessment was also greater but not significantly different in the Latino group, compared with the White group, respectively (42.9% vs. 28.6%; P = .10).
However, differences in the patterns of disease did reach significance. This included a lower mean Psoriasis Assessment Severity Index score of the trunk, axilla, and groin in Latinos (4.74 vs. 9.73; P = .02). But compared with White participants, Latinos had a higher mean percentage of body surface area involvement in the upper limbs (4.78 vs. 1.85; P = .004) and a higher percentage of total body surface area involvement (20.50 vs. 10.03; P = .02).
“While White patients were found to have lived many more years with psoriasis, it is important for future studies to examine whether this is due to earlier onset or delayed diagnosis, given the fact that minorities are less likely to have access to a dermatologist,” reported Ms. Ashbaugh, who performed this work under the guidance of the senior author, Natasha Mesinkovska, MD, PhD, with the department of dermatology, University of California, Irvine.
Overall, the study suggested that body surface coverage and severity is not similarly distributed in Latinos relative to Whites. Although Ms. Ashbaugh conceded that the small sample size and retrospective design of this study are important limitations, she believes that her study, along with previously published studies that suggest psoriasis characteristics may differ meaningfully by race or ethnicity, raises issues that should be explored in future studies designed to confirm differences and whether those differences should affect management.
Other studies have suggested “there are notable differences in the presentation of psoriasis between racial and ethnic groups with the Latino population often presenting to physicians with more severe psoriasis and increased body surface area involvement,” Ms. Ashbaugh noted. Although this appears to be one of the first studies to examine psoriasis characteristics in Latinos relative to Whites, she believes this is an area ripe for further analysis.
Psoriasis “is not a rare occurrence” in non-White populations even if U.S. data suggest that the prevalence in “people of color is lower than that of psoriasis in the U.S. white population,” Amy McMichael, MD, chair of the department of dermatology, Wake Forest Baptist Medical Center, Winston-Salem, N.C., commented in an interview after the meeting. She agreed that it cannot be assumed that psoriasis in skin of color has the same manifestations or responds to treatment in the same way as in White patients.
“Studies have suggested that lesion thickness and, often, extent of disease can be worse in patients of color. Few studies to date have examined the efficacy of treatments and impact of disease in these populations,” she said.
One exception was a study Dr. McMichael and colleagues published last year on the efficacy and safety of the interleukin-17 receptor A antagonist brodalumab for psoriasis in patients of color. The study showed that Black, Latino, and Asian patients participating in the AMAGINE-2 and AMAGINE-3 trials achieved similar outcomes as White participants.
“We published this study because this is one of the first, if not the first, to have enough patients of color to actually draw conclusions about the efficacy of the biologic as well as the patient-reported outcomes,” she explained.
Like the author of the evaluation of Latino patients undertaken at the University of California, Irvine, Dr. McMichael said studies of psoriasis specific to patients of color are needed.
“We cannot assume all patients of color will have the same outcomes as their Caucasian counterparts. It is imperative to include those of color in future psoriasis treatment trials in order to determine the efficacy of new medications,” she added, specifically calling for collection of data on patient-reported outcomes.
Ms. Ashbaugh has no relevant financial relationships to disclose. Dr. McMichael’s disclosures included serving as an investigator and/or consultant for companies that included Allergan, Procter & Gamble, Johnson & Johnson, and Aclaris.
FROM SOC 2020
Shingrix effective in older adults with preexisting immune-mediated disorders
The adjuvanted recombinant zoster vaccine Shingrix appears to be effective in older adults with autoimmune diseases who are not receiving treatment regimens that suppress the immune system, according to a post hoc analysis of patients in two clinical trials.
A two-dose regimen of Shingrix was effective in 90.5% of a subset of patients in two phase 3 clinical trials of adults who were aged at least 50 years, according to Alemnew F. Dagnew, MD, of GlaxoSmithKline and colleagues. The lowest rates of effectiveness with Shingrix, for patients aged between 70-79 years, was 84.4%, the researchers reported in Rheumatology.
The CDC recommends adults aged at least 50 years receive two doses of Shingrix to help prevent reoccurrence of herpes zoster, or Zostavax (zoster vaccine live) if adults are allergic to components of the Shingrix vaccine or have tested negative for varicella zoster virus immunity.
Dr. Dagnew and colleagues evaluated Shingrix in 983 patients who received two doses of Shingrix and 960 patients who received placebo from the ZOE-50 and ZOE-70 trials, where each dose was administered at least 2 months apart. The mean age of patients in both groups was 68.8 years in the Shingrix group and 69.4 years in the placebo group, and more than half of patients in both Shingrix (59.9%) and placebo groups (60.8%) were women. About 7% of the patients in two clinical trial had a pIMD.
At enrollment, the most common preexisting immune-mediated disorders (pIMDs) were psoriasis (215 patients taking Shingrix vs. 239 patients on placebo), spondyloarthropathy (109 patients taking Shingrix vs. 89 patients on placebo), rheumatoid arthritis (96 patients taking Shingrix vs. 94 patients on placebo), and celiac disease (41 patients taking Shingrix vs. 34 patients on placebo). Dr. Dagnew and colleagues examined the subgroup of patients with pIMDs for safety and vaccine efficacy, which was defined as not developing herpes zoster before the second dose.
Overall, the efficacy of Shingrix was 90.5% across all age groups (95% confidence interval, 73.5%-97.5%), with the group aged between 70-79 years having the lowest rate of effectiveness (95% CI, 30.8%-98.3%). The rate of severe adverse events was 14.6% in the Shingrix group and 11.7% in the placebo group between the first Shingrix dose and for up to 1 year after the second dose. The most common adverse events were infections and infestations as well as cardiac disorders. “Our data show a balance between study groups in the frequency and nature of SAEs, confirming the favorable safety profile of [Shingrix] in populations with pIMDs,” Dr. Dagnew and colleagues wrote.
The researchers acknowledged that the ZOE-50/70 studies were underpowered to detect the efficacy and safety of Shingrix in individuals with pIMDs but said that the large number of participants in the studies let them estimate efficacy and adverse events for this subgroup. They also noted there was no randomization of pIMDs at enrollment, even though pIMDs occurred at similar rates between Shingrix and placebo groups.
This study was funded by GlaxoSmithKline; the company helped with conducting and analyzing the study and also provided the costs associated with publishing it. Five authors reported being an employee of GlaxoSmithKline during the time the work was conducted, and four of the five own stock in the company. One author is now an employee of UCB. One author reported having served on the advisory boards for Merck Sharp & Dohme, GlaxoSmithKline, and Curevo.
SOURCE: Dagnew AF et al. Rheumatology. 2020 Sep 10. doi: 10.1093/rheumatology/keaa424.
The adjuvanted recombinant zoster vaccine Shingrix appears to be effective in older adults with autoimmune diseases who are not receiving treatment regimens that suppress the immune system, according to a post hoc analysis of patients in two clinical trials.
A two-dose regimen of Shingrix was effective in 90.5% of a subset of patients in two phase 3 clinical trials of adults who were aged at least 50 years, according to Alemnew F. Dagnew, MD, of GlaxoSmithKline and colleagues. The lowest rates of effectiveness with Shingrix, for patients aged between 70-79 years, was 84.4%, the researchers reported in Rheumatology.
The CDC recommends adults aged at least 50 years receive two doses of Shingrix to help prevent reoccurrence of herpes zoster, or Zostavax (zoster vaccine live) if adults are allergic to components of the Shingrix vaccine or have tested negative for varicella zoster virus immunity.
Dr. Dagnew and colleagues evaluated Shingrix in 983 patients who received two doses of Shingrix and 960 patients who received placebo from the ZOE-50 and ZOE-70 trials, where each dose was administered at least 2 months apart. The mean age of patients in both groups was 68.8 years in the Shingrix group and 69.4 years in the placebo group, and more than half of patients in both Shingrix (59.9%) and placebo groups (60.8%) were women. About 7% of the patients in two clinical trial had a pIMD.
At enrollment, the most common preexisting immune-mediated disorders (pIMDs) were psoriasis (215 patients taking Shingrix vs. 239 patients on placebo), spondyloarthropathy (109 patients taking Shingrix vs. 89 patients on placebo), rheumatoid arthritis (96 patients taking Shingrix vs. 94 patients on placebo), and celiac disease (41 patients taking Shingrix vs. 34 patients on placebo). Dr. Dagnew and colleagues examined the subgroup of patients with pIMDs for safety and vaccine efficacy, which was defined as not developing herpes zoster before the second dose.
Overall, the efficacy of Shingrix was 90.5% across all age groups (95% confidence interval, 73.5%-97.5%), with the group aged between 70-79 years having the lowest rate of effectiveness (95% CI, 30.8%-98.3%). The rate of severe adverse events was 14.6% in the Shingrix group and 11.7% in the placebo group between the first Shingrix dose and for up to 1 year after the second dose. The most common adverse events were infections and infestations as well as cardiac disorders. “Our data show a balance between study groups in the frequency and nature of SAEs, confirming the favorable safety profile of [Shingrix] in populations with pIMDs,” Dr. Dagnew and colleagues wrote.
The researchers acknowledged that the ZOE-50/70 studies were underpowered to detect the efficacy and safety of Shingrix in individuals with pIMDs but said that the large number of participants in the studies let them estimate efficacy and adverse events for this subgroup. They also noted there was no randomization of pIMDs at enrollment, even though pIMDs occurred at similar rates between Shingrix and placebo groups.
This study was funded by GlaxoSmithKline; the company helped with conducting and analyzing the study and also provided the costs associated with publishing it. Five authors reported being an employee of GlaxoSmithKline during the time the work was conducted, and four of the five own stock in the company. One author is now an employee of UCB. One author reported having served on the advisory boards for Merck Sharp & Dohme, GlaxoSmithKline, and Curevo.
SOURCE: Dagnew AF et al. Rheumatology. 2020 Sep 10. doi: 10.1093/rheumatology/keaa424.
The adjuvanted recombinant zoster vaccine Shingrix appears to be effective in older adults with autoimmune diseases who are not receiving treatment regimens that suppress the immune system, according to a post hoc analysis of patients in two clinical trials.
A two-dose regimen of Shingrix was effective in 90.5% of a subset of patients in two phase 3 clinical trials of adults who were aged at least 50 years, according to Alemnew F. Dagnew, MD, of GlaxoSmithKline and colleagues. The lowest rates of effectiveness with Shingrix, for patients aged between 70-79 years, was 84.4%, the researchers reported in Rheumatology.
The CDC recommends adults aged at least 50 years receive two doses of Shingrix to help prevent reoccurrence of herpes zoster, or Zostavax (zoster vaccine live) if adults are allergic to components of the Shingrix vaccine or have tested negative for varicella zoster virus immunity.
Dr. Dagnew and colleagues evaluated Shingrix in 983 patients who received two doses of Shingrix and 960 patients who received placebo from the ZOE-50 and ZOE-70 trials, where each dose was administered at least 2 months apart. The mean age of patients in both groups was 68.8 years in the Shingrix group and 69.4 years in the placebo group, and more than half of patients in both Shingrix (59.9%) and placebo groups (60.8%) were women. About 7% of the patients in two clinical trial had a pIMD.
At enrollment, the most common preexisting immune-mediated disorders (pIMDs) were psoriasis (215 patients taking Shingrix vs. 239 patients on placebo), spondyloarthropathy (109 patients taking Shingrix vs. 89 patients on placebo), rheumatoid arthritis (96 patients taking Shingrix vs. 94 patients on placebo), and celiac disease (41 patients taking Shingrix vs. 34 patients on placebo). Dr. Dagnew and colleagues examined the subgroup of patients with pIMDs for safety and vaccine efficacy, which was defined as not developing herpes zoster before the second dose.
Overall, the efficacy of Shingrix was 90.5% across all age groups (95% confidence interval, 73.5%-97.5%), with the group aged between 70-79 years having the lowest rate of effectiveness (95% CI, 30.8%-98.3%). The rate of severe adverse events was 14.6% in the Shingrix group and 11.7% in the placebo group between the first Shingrix dose and for up to 1 year after the second dose. The most common adverse events were infections and infestations as well as cardiac disorders. “Our data show a balance between study groups in the frequency and nature of SAEs, confirming the favorable safety profile of [Shingrix] in populations with pIMDs,” Dr. Dagnew and colleagues wrote.
The researchers acknowledged that the ZOE-50/70 studies were underpowered to detect the efficacy and safety of Shingrix in individuals with pIMDs but said that the large number of participants in the studies let them estimate efficacy and adverse events for this subgroup. They also noted there was no randomization of pIMDs at enrollment, even though pIMDs occurred at similar rates between Shingrix and placebo groups.
This study was funded by GlaxoSmithKline; the company helped with conducting and analyzing the study and also provided the costs associated with publishing it. Five authors reported being an employee of GlaxoSmithKline during the time the work was conducted, and four of the five own stock in the company. One author is now an employee of UCB. One author reported having served on the advisory boards for Merck Sharp & Dohme, GlaxoSmithKline, and Curevo.
SOURCE: Dagnew AF et al. Rheumatology. 2020 Sep 10. doi: 10.1093/rheumatology/keaa424.
FROM RHEUMATOLOGY
Pregnancy studies on psoriasis, PsA medications pick up
Christina Chambers, PhD, MPH, who runs the MotherToBaby Pregnancy Studies research center at the University of California, San Diego, has found most pregnant women to be “entirely altruistic” about sharing their experiences with drug treatment during pregnancy.
– but dermatologists, rheumatologists, and their female patients with psoriasis and psoriatic arthritis (PsA) want much more.
And women’s participation in the MotherToBaby studies conducted by the nonprofit Organization of Teratology Information Specialists (OTIS) is key, say physicians who are treating women of reproductive age. OTIS is now listed in drug labeling as the “pregnancy registry” contact for many of the medications they may be discussing with patients.
Dr. Chambers said that most women appreciate “that participating in a study may not help her with her pregnancy, but it can help her sister or her friend or someone else who has these same questions in planning a pregnancy of ‘Can I stay on my treatment?’ or, in the case of an unplanned pregnancy, ‘Should I be concerned?’ ”
OTIS has enrolled women with psoriasis and/or PsA in studies of nine medications, most of them biologics (both TNF-alpha blockers and newer anti-interleukin agents).
Four of the studies – those evaluating etanercept (Enbrel), adalimumab (Humira), abatacept (Orencia), and ustekinumab (Stelara) – are now closed to enrollment with analyses either underway or completed. The other five are currently enrolling patients and involve treatment with certolizumab pegol (Cimzia), tildrakizumab (Ilumya), apremilast (Otezla), guselkumab (Tremfya), and tofacitinib (Xeljanz).
Lisa R. Sammaritano, MD, a rheumatologist at the Hospital for Special Surgery, New York, who led the development of the American College of Rheumatology’s first guideline for the management of reproductive health in rheumatic and musculoskeletal diseases, recommends to some of her patients that they contact OTIS. “Their pregnancy registry studies have added important information to the field over the years,” she said.
Most recently, a study of the anti–TNF-alpha medication adalimumab that began in 2004 in pregnant patients with RA and Crohn’s disease culminated in a 2019 PLOS ONE paper reporting no associations between exposure to the medication and an increased risk of adverse outcomes. The outcomes studied were major structural birth defects, minor defects, spontaneous abortion, preterm delivery, prenatal and postnatal growth deficiency, serious or opportunistic infections, and malignancies.
An analysis is underway of adalimumab exposure in women with PsA – a patient subset that was added after the study started. But in the meantime, Dr. Chambers said, the 2019 research article is relevant to questions of drug safety across indications.
OTIS’s MothertoBaby studies are structured as prospective cohort studies. Dr. Chambers, a perinatal epidemiologist, is president of OTIS, which recruits women who have an exposure to the medication under study – at least one dose, for any length of time. And in most cases, it also recruits women with the underlying condition but no exposure and healthy women without the condition to represent the general population.
It’s the disease-matched comparison group that makes OTIS’s studies different from traditional pregnancy registries involving “a simple exposure series and outcomes that are described in the context of what you’d expect in the general population,” said Dr. Chambers, professor in the department of pediatrics, as well as family and preventative medicine, at UCSD and codirector of the Center for Better Beginnings at that university. “Many maternal conditions themselves [or their comorbidities] carry some risk of adverse outcomes in pregnancy.”
The OTIS studies typically involve at least 100 exposed pregnancies and a similar number of unexposed pregnancies; some have cohorts of 200-300.
The recently published study of adalimumab, for instance, included 257 women with exposure to the drug and 120 women in a disease comparison group with no exposure. In addition to finding no associations between drug exposure and adverse outcomes, the study found that women with RA or Crohn’s were at increased risk of preterm delivery, irrespective of adalimumab exposure.
“There’s insufficient [power with any of these numbers] to come to the conclusion that a drug is safe,” she said. “But what we have been able to say [through our studies] is that we’ve looked carefully at the whole array of outcomes ... and we don’t see anything unusual. That early view can be reassuring” until large population-based studies or claims analyses become possible.
Dr. Sammaritano, also with Weill Cornell Medicine, New York, said that she does not recommend registry participation for patients who stop biologics at the diagnosis of pregnancy. Since “the start of IgG antibody transfer during pregnancy is about 16 weeks,” she worries that including these patients might lead to falsely reassuring findings. “We are most interested in [knowing the outcomes of] patients who must continue the drugs through pregnancy,” she said.
Dr. Chambers, however, said that in her view, placental transfer is not a requirement for a medication to have some effect on the outcome of pregnancy. “The outcome could be influenced by an effect of the medication that doesn’t require placental transfer or require placental transfer in large amounts,” she said. “So it’s relevant to examine exposures that have occurred only in the first trimester, and this is especially true for the outcome of major birth defects, most of which are initiated in the first trimester.”
The MotherToBaby studies typically include both early, short exposures and longer exposures, she said. “And certainly, duration of use is a factor that we do consider in looking at specific outcomes such as growth, preterm delivery, and risk of serious or opportunistic infections.”
(In the published study of adalimumab, 65.3% of women in the medication-exposed cohort used the medication in all three trimesters, 10.5% in the first and second trimesters, and 22.4% in the first trimester only.)
Women participating in the MotherToBaby studies complete two to four interviews during pregnancy and may be interviewed again after delivery. They are asked for their permission to share a copy of their medical records – and their baby’s medical records – and their babies receive a follow-up pediatric exam by a pediatrician with expertise in dysmorphology/genetics (who is blinded to exposure status), most commonly in the participant’s home. Providers are not asked to enter any data.
Eliza Chakravarty, MD, a rheumatologist with the Oklahoma Medical Research Foundation in Oklahoma City who treats patients with PsA who are pregnant or considering pregnancy, said that her referrals for research participation “have been mostly to MothertoBaby.”
“Most drug companies [in the autoimmune space] are now contracting with them [for their pregnancy exposure research],” she said. “I really like that it’s become so centralized.”
She tells patients that many questions can be answered through research, that their experience matters, and that “there are benefits” to the extra pediatric examination. “I give them the information and let them decide whether or not they want to call [MotherToBaby],” she said. “I don’t want to impose. I want to make them aware.”
Dr. Chambers emphasizes to patients and physicians that the studies are strictly observational and do not require any changes in personal or medical regimens. “When people hear the word ‘research’ they think of clinical trials. We’re saying, you and your provider do everything you normally would do, just let us observe what happens during your pregnancy.”
Physicians should assure patients, moreover, that “just because the drug is being studied doesn’t mean there’s a known risk or even a suspected risk,” she said.
The MotherToBaby studies receive funding from the pharmaceutical companies, which are required by the Food and Drug Administration to conduct pregnancy exposure registries for medications used during pregnancy or in women of reproductive age. OTIS has an independent advisory board, however, and independently analyzes and publishes its findings. Progress reports are shared with the pharmaceutical companies, and in turn, the FDA, Dr. Chambers said.
To refer patients for MotherToBaby studies, physicians can use an online referral form found on the MothertoBaby web site, a service of OTIS, or call the pregnancy studies team at 877-311-8972 to provide them with the patient’s name or number. Patients may also be given the number and advised to consider calling. MotherToBaby offers medication fact sheets that answer questions about exposures during pregnancy and breastfeeding, and runs a free and confidential teratogen counseling service: 866-626-6847.
Christina Chambers, PhD, MPH, who runs the MotherToBaby Pregnancy Studies research center at the University of California, San Diego, has found most pregnant women to be “entirely altruistic” about sharing their experiences with drug treatment during pregnancy.
– but dermatologists, rheumatologists, and their female patients with psoriasis and psoriatic arthritis (PsA) want much more.
And women’s participation in the MotherToBaby studies conducted by the nonprofit Organization of Teratology Information Specialists (OTIS) is key, say physicians who are treating women of reproductive age. OTIS is now listed in drug labeling as the “pregnancy registry” contact for many of the medications they may be discussing with patients.
Dr. Chambers said that most women appreciate “that participating in a study may not help her with her pregnancy, but it can help her sister or her friend or someone else who has these same questions in planning a pregnancy of ‘Can I stay on my treatment?’ or, in the case of an unplanned pregnancy, ‘Should I be concerned?’ ”
OTIS has enrolled women with psoriasis and/or PsA in studies of nine medications, most of them biologics (both TNF-alpha blockers and newer anti-interleukin agents).
Four of the studies – those evaluating etanercept (Enbrel), adalimumab (Humira), abatacept (Orencia), and ustekinumab (Stelara) – are now closed to enrollment with analyses either underway or completed. The other five are currently enrolling patients and involve treatment with certolizumab pegol (Cimzia), tildrakizumab (Ilumya), apremilast (Otezla), guselkumab (Tremfya), and tofacitinib (Xeljanz).
Lisa R. Sammaritano, MD, a rheumatologist at the Hospital for Special Surgery, New York, who led the development of the American College of Rheumatology’s first guideline for the management of reproductive health in rheumatic and musculoskeletal diseases, recommends to some of her patients that they contact OTIS. “Their pregnancy registry studies have added important information to the field over the years,” she said.
Most recently, a study of the anti–TNF-alpha medication adalimumab that began in 2004 in pregnant patients with RA and Crohn’s disease culminated in a 2019 PLOS ONE paper reporting no associations between exposure to the medication and an increased risk of adverse outcomes. The outcomes studied were major structural birth defects, minor defects, spontaneous abortion, preterm delivery, prenatal and postnatal growth deficiency, serious or opportunistic infections, and malignancies.
An analysis is underway of adalimumab exposure in women with PsA – a patient subset that was added after the study started. But in the meantime, Dr. Chambers said, the 2019 research article is relevant to questions of drug safety across indications.
OTIS’s MothertoBaby studies are structured as prospective cohort studies. Dr. Chambers, a perinatal epidemiologist, is president of OTIS, which recruits women who have an exposure to the medication under study – at least one dose, for any length of time. And in most cases, it also recruits women with the underlying condition but no exposure and healthy women without the condition to represent the general population.
It’s the disease-matched comparison group that makes OTIS’s studies different from traditional pregnancy registries involving “a simple exposure series and outcomes that are described in the context of what you’d expect in the general population,” said Dr. Chambers, professor in the department of pediatrics, as well as family and preventative medicine, at UCSD and codirector of the Center for Better Beginnings at that university. “Many maternal conditions themselves [or their comorbidities] carry some risk of adverse outcomes in pregnancy.”
The OTIS studies typically involve at least 100 exposed pregnancies and a similar number of unexposed pregnancies; some have cohorts of 200-300.
The recently published study of adalimumab, for instance, included 257 women with exposure to the drug and 120 women in a disease comparison group with no exposure. In addition to finding no associations between drug exposure and adverse outcomes, the study found that women with RA or Crohn’s were at increased risk of preterm delivery, irrespective of adalimumab exposure.
“There’s insufficient [power with any of these numbers] to come to the conclusion that a drug is safe,” she said. “But what we have been able to say [through our studies] is that we’ve looked carefully at the whole array of outcomes ... and we don’t see anything unusual. That early view can be reassuring” until large population-based studies or claims analyses become possible.
Dr. Sammaritano, also with Weill Cornell Medicine, New York, said that she does not recommend registry participation for patients who stop biologics at the diagnosis of pregnancy. Since “the start of IgG antibody transfer during pregnancy is about 16 weeks,” she worries that including these patients might lead to falsely reassuring findings. “We are most interested in [knowing the outcomes of] patients who must continue the drugs through pregnancy,” she said.
Dr. Chambers, however, said that in her view, placental transfer is not a requirement for a medication to have some effect on the outcome of pregnancy. “The outcome could be influenced by an effect of the medication that doesn’t require placental transfer or require placental transfer in large amounts,” she said. “So it’s relevant to examine exposures that have occurred only in the first trimester, and this is especially true for the outcome of major birth defects, most of which are initiated in the first trimester.”
The MotherToBaby studies typically include both early, short exposures and longer exposures, she said. “And certainly, duration of use is a factor that we do consider in looking at specific outcomes such as growth, preterm delivery, and risk of serious or opportunistic infections.”
(In the published study of adalimumab, 65.3% of women in the medication-exposed cohort used the medication in all three trimesters, 10.5% in the first and second trimesters, and 22.4% in the first trimester only.)
Women participating in the MotherToBaby studies complete two to four interviews during pregnancy and may be interviewed again after delivery. They are asked for their permission to share a copy of their medical records – and their baby’s medical records – and their babies receive a follow-up pediatric exam by a pediatrician with expertise in dysmorphology/genetics (who is blinded to exposure status), most commonly in the participant’s home. Providers are not asked to enter any data.
Eliza Chakravarty, MD, a rheumatologist with the Oklahoma Medical Research Foundation in Oklahoma City who treats patients with PsA who are pregnant or considering pregnancy, said that her referrals for research participation “have been mostly to MothertoBaby.”
“Most drug companies [in the autoimmune space] are now contracting with them [for their pregnancy exposure research],” she said. “I really like that it’s become so centralized.”
She tells patients that many questions can be answered through research, that their experience matters, and that “there are benefits” to the extra pediatric examination. “I give them the information and let them decide whether or not they want to call [MotherToBaby],” she said. “I don’t want to impose. I want to make them aware.”
Dr. Chambers emphasizes to patients and physicians that the studies are strictly observational and do not require any changes in personal or medical regimens. “When people hear the word ‘research’ they think of clinical trials. We’re saying, you and your provider do everything you normally would do, just let us observe what happens during your pregnancy.”
Physicians should assure patients, moreover, that “just because the drug is being studied doesn’t mean there’s a known risk or even a suspected risk,” she said.
The MotherToBaby studies receive funding from the pharmaceutical companies, which are required by the Food and Drug Administration to conduct pregnancy exposure registries for medications used during pregnancy or in women of reproductive age. OTIS has an independent advisory board, however, and independently analyzes and publishes its findings. Progress reports are shared with the pharmaceutical companies, and in turn, the FDA, Dr. Chambers said.
To refer patients for MotherToBaby studies, physicians can use an online referral form found on the MothertoBaby web site, a service of OTIS, or call the pregnancy studies team at 877-311-8972 to provide them with the patient’s name or number. Patients may also be given the number and advised to consider calling. MotherToBaby offers medication fact sheets that answer questions about exposures during pregnancy and breastfeeding, and runs a free and confidential teratogen counseling service: 866-626-6847.
Christina Chambers, PhD, MPH, who runs the MotherToBaby Pregnancy Studies research center at the University of California, San Diego, has found most pregnant women to be “entirely altruistic” about sharing their experiences with drug treatment during pregnancy.
– but dermatologists, rheumatologists, and their female patients with psoriasis and psoriatic arthritis (PsA) want much more.
And women’s participation in the MotherToBaby studies conducted by the nonprofit Organization of Teratology Information Specialists (OTIS) is key, say physicians who are treating women of reproductive age. OTIS is now listed in drug labeling as the “pregnancy registry” contact for many of the medications they may be discussing with patients.
Dr. Chambers said that most women appreciate “that participating in a study may not help her with her pregnancy, but it can help her sister or her friend or someone else who has these same questions in planning a pregnancy of ‘Can I stay on my treatment?’ or, in the case of an unplanned pregnancy, ‘Should I be concerned?’ ”
OTIS has enrolled women with psoriasis and/or PsA in studies of nine medications, most of them biologics (both TNF-alpha blockers and newer anti-interleukin agents).
Four of the studies – those evaluating etanercept (Enbrel), adalimumab (Humira), abatacept (Orencia), and ustekinumab (Stelara) – are now closed to enrollment with analyses either underway or completed. The other five are currently enrolling patients and involve treatment with certolizumab pegol (Cimzia), tildrakizumab (Ilumya), apremilast (Otezla), guselkumab (Tremfya), and tofacitinib (Xeljanz).
Lisa R. Sammaritano, MD, a rheumatologist at the Hospital for Special Surgery, New York, who led the development of the American College of Rheumatology’s first guideline for the management of reproductive health in rheumatic and musculoskeletal diseases, recommends to some of her patients that they contact OTIS. “Their pregnancy registry studies have added important information to the field over the years,” she said.
Most recently, a study of the anti–TNF-alpha medication adalimumab that began in 2004 in pregnant patients with RA and Crohn’s disease culminated in a 2019 PLOS ONE paper reporting no associations between exposure to the medication and an increased risk of adverse outcomes. The outcomes studied were major structural birth defects, minor defects, spontaneous abortion, preterm delivery, prenatal and postnatal growth deficiency, serious or opportunistic infections, and malignancies.
An analysis is underway of adalimumab exposure in women with PsA – a patient subset that was added after the study started. But in the meantime, Dr. Chambers said, the 2019 research article is relevant to questions of drug safety across indications.
OTIS’s MothertoBaby studies are structured as prospective cohort studies. Dr. Chambers, a perinatal epidemiologist, is president of OTIS, which recruits women who have an exposure to the medication under study – at least one dose, for any length of time. And in most cases, it also recruits women with the underlying condition but no exposure and healthy women without the condition to represent the general population.
It’s the disease-matched comparison group that makes OTIS’s studies different from traditional pregnancy registries involving “a simple exposure series and outcomes that are described in the context of what you’d expect in the general population,” said Dr. Chambers, professor in the department of pediatrics, as well as family and preventative medicine, at UCSD and codirector of the Center for Better Beginnings at that university. “Many maternal conditions themselves [or their comorbidities] carry some risk of adverse outcomes in pregnancy.”
The OTIS studies typically involve at least 100 exposed pregnancies and a similar number of unexposed pregnancies; some have cohorts of 200-300.
The recently published study of adalimumab, for instance, included 257 women with exposure to the drug and 120 women in a disease comparison group with no exposure. In addition to finding no associations between drug exposure and adverse outcomes, the study found that women with RA or Crohn’s were at increased risk of preterm delivery, irrespective of adalimumab exposure.
“There’s insufficient [power with any of these numbers] to come to the conclusion that a drug is safe,” she said. “But what we have been able to say [through our studies] is that we’ve looked carefully at the whole array of outcomes ... and we don’t see anything unusual. That early view can be reassuring” until large population-based studies or claims analyses become possible.
Dr. Sammaritano, also with Weill Cornell Medicine, New York, said that she does not recommend registry participation for patients who stop biologics at the diagnosis of pregnancy. Since “the start of IgG antibody transfer during pregnancy is about 16 weeks,” she worries that including these patients might lead to falsely reassuring findings. “We are most interested in [knowing the outcomes of] patients who must continue the drugs through pregnancy,” she said.
Dr. Chambers, however, said that in her view, placental transfer is not a requirement for a medication to have some effect on the outcome of pregnancy. “The outcome could be influenced by an effect of the medication that doesn’t require placental transfer or require placental transfer in large amounts,” she said. “So it’s relevant to examine exposures that have occurred only in the first trimester, and this is especially true for the outcome of major birth defects, most of which are initiated in the first trimester.”
The MotherToBaby studies typically include both early, short exposures and longer exposures, she said. “And certainly, duration of use is a factor that we do consider in looking at specific outcomes such as growth, preterm delivery, and risk of serious or opportunistic infections.”
(In the published study of adalimumab, 65.3% of women in the medication-exposed cohort used the medication in all three trimesters, 10.5% in the first and second trimesters, and 22.4% in the first trimester only.)
Women participating in the MotherToBaby studies complete two to four interviews during pregnancy and may be interviewed again after delivery. They are asked for their permission to share a copy of their medical records – and their baby’s medical records – and their babies receive a follow-up pediatric exam by a pediatrician with expertise in dysmorphology/genetics (who is blinded to exposure status), most commonly in the participant’s home. Providers are not asked to enter any data.
Eliza Chakravarty, MD, a rheumatologist with the Oklahoma Medical Research Foundation in Oklahoma City who treats patients with PsA who are pregnant or considering pregnancy, said that her referrals for research participation “have been mostly to MothertoBaby.”
“Most drug companies [in the autoimmune space] are now contracting with them [for their pregnancy exposure research],” she said. “I really like that it’s become so centralized.”
She tells patients that many questions can be answered through research, that their experience matters, and that “there are benefits” to the extra pediatric examination. “I give them the information and let them decide whether or not they want to call [MotherToBaby],” she said. “I don’t want to impose. I want to make them aware.”
Dr. Chambers emphasizes to patients and physicians that the studies are strictly observational and do not require any changes in personal or medical regimens. “When people hear the word ‘research’ they think of clinical trials. We’re saying, you and your provider do everything you normally would do, just let us observe what happens during your pregnancy.”
Physicians should assure patients, moreover, that “just because the drug is being studied doesn’t mean there’s a known risk or even a suspected risk,” she said.
The MotherToBaby studies receive funding from the pharmaceutical companies, which are required by the Food and Drug Administration to conduct pregnancy exposure registries for medications used during pregnancy or in women of reproductive age. OTIS has an independent advisory board, however, and independently analyzes and publishes its findings. Progress reports are shared with the pharmaceutical companies, and in turn, the FDA, Dr. Chambers said.
To refer patients for MotherToBaby studies, physicians can use an online referral form found on the MothertoBaby web site, a service of OTIS, or call the pregnancy studies team at 877-311-8972 to provide them with the patient’s name or number. Patients may also be given the number and advised to consider calling. MotherToBaby offers medication fact sheets that answer questions about exposures during pregnancy and breastfeeding, and runs a free and confidential teratogen counseling service: 866-626-6847.
Recurrent leg lesions
Tender erythematous nodules or plaques on the extensor surfaces—usually on the legs and occasionally on the arms—are the hallmarks for erythema nodosum, which was diagnosed in this case. It typically occurs in young women, ages 15 to 30, and the nodules or plaques are often accompanied by prodromal fever and malaise. The lesions often are painful and tender to pressure or palpation; they are thought to be caused by a reaction to a stimulus, leading to inflammation of the septa in the subcutaneous fat. While the trigger is often unknown, in some cases, an underlying infection, particularly Streptococcus or tuberculosis (TB), is identified. Sarcoidosis, malignancy, or an increase in estrogen (exogenous or endogenous) also can provoke the disorder.
Due to the risk of underlying disease or triggers, it is prudent to perform radiography of the chest, as well as obtain a complete blood count, sedimentation rate or C reactive protein, and an antistreptolysin O titer when you suspect erythema nodosum. TB testing is also advised. Biopsy typically is not performed because the diagnosis usually is made clinically. If the diagnosis is in doubt, a biopsy can offer confirmation or lead to a different diagnosis such as vasculitis—especially if the lesions are eroded. Since erythema nodosum is an inflammation of the subcutaneous fat, it is important to sample skin lesions deeper than the usual punch biopsy; an incisional biopsy may be required to get an adequate sample.
Erythema nodosum typically resolves spontaneously over a period of weeks, even if there is underlying disease. Therefore, it may be possible to defer treatment if minimal symptoms are present. Otherwise, first-line treatment for the pain and malaise is a nonsteroidal anti-inflammatory drug (NSAID). Oral potassium iodide (360-900 mg/d) is considered second-line treatment and systemic corticosteroids are a third-line option.
For this patient, biopsy was deferred and diagnostic tests were all negative. She had notable pain and a history of good resolution of symptoms with prednisone (5 mg/d), so this drug was prescribed for a 7-day course. She was counseled to avoid taking the NSAIDs and prednisone together due to increased risk of gastritis and ulceration. Recurrent disease can be treated with dapsone (100 mg/d) or hydroxychloroquine (200 mg bid).
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Blake T, Manahan M, Rodins K. Erythema nodosum - a review of an uncommon panniculitis. Dermatol Online J. 2014;20:22376.

Tender erythematous nodules or plaques on the extensor surfaces—usually on the legs and occasionally on the arms—are the hallmarks for erythema nodosum, which was diagnosed in this case. It typically occurs in young women, ages 15 to 30, and the nodules or plaques are often accompanied by prodromal fever and malaise. The lesions often are painful and tender to pressure or palpation; they are thought to be caused by a reaction to a stimulus, leading to inflammation of the septa in the subcutaneous fat. While the trigger is often unknown, in some cases, an underlying infection, particularly Streptococcus or tuberculosis (TB), is identified. Sarcoidosis, malignancy, or an increase in estrogen (exogenous or endogenous) also can provoke the disorder.
Due to the risk of underlying disease or triggers, it is prudent to perform radiography of the chest, as well as obtain a complete blood count, sedimentation rate or C reactive protein, and an antistreptolysin O titer when you suspect erythema nodosum. TB testing is also advised. Biopsy typically is not performed because the diagnosis usually is made clinically. If the diagnosis is in doubt, a biopsy can offer confirmation or lead to a different diagnosis such as vasculitis—especially if the lesions are eroded. Since erythema nodosum is an inflammation of the subcutaneous fat, it is important to sample skin lesions deeper than the usual punch biopsy; an incisional biopsy may be required to get an adequate sample.
Erythema nodosum typically resolves spontaneously over a period of weeks, even if there is underlying disease. Therefore, it may be possible to defer treatment if minimal symptoms are present. Otherwise, first-line treatment for the pain and malaise is a nonsteroidal anti-inflammatory drug (NSAID). Oral potassium iodide (360-900 mg/d) is considered second-line treatment and systemic corticosteroids are a third-line option.
For this patient, biopsy was deferred and diagnostic tests were all negative. She had notable pain and a history of good resolution of symptoms with prednisone (5 mg/d), so this drug was prescribed for a 7-day course. She was counseled to avoid taking the NSAIDs and prednisone together due to increased risk of gastritis and ulceration. Recurrent disease can be treated with dapsone (100 mg/d) or hydroxychloroquine (200 mg bid).
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

Tender erythematous nodules or plaques on the extensor surfaces—usually on the legs and occasionally on the arms—are the hallmarks for erythema nodosum, which was diagnosed in this case. It typically occurs in young women, ages 15 to 30, and the nodules or plaques are often accompanied by prodromal fever and malaise. The lesions often are painful and tender to pressure or palpation; they are thought to be caused by a reaction to a stimulus, leading to inflammation of the septa in the subcutaneous fat. While the trigger is often unknown, in some cases, an underlying infection, particularly Streptococcus or tuberculosis (TB), is identified. Sarcoidosis, malignancy, or an increase in estrogen (exogenous or endogenous) also can provoke the disorder.
Due to the risk of underlying disease or triggers, it is prudent to perform radiography of the chest, as well as obtain a complete blood count, sedimentation rate or C reactive protein, and an antistreptolysin O titer when you suspect erythema nodosum. TB testing is also advised. Biopsy typically is not performed because the diagnosis usually is made clinically. If the diagnosis is in doubt, a biopsy can offer confirmation or lead to a different diagnosis such as vasculitis—especially if the lesions are eroded. Since erythema nodosum is an inflammation of the subcutaneous fat, it is important to sample skin lesions deeper than the usual punch biopsy; an incisional biopsy may be required to get an adequate sample.
Erythema nodosum typically resolves spontaneously over a period of weeks, even if there is underlying disease. Therefore, it may be possible to defer treatment if minimal symptoms are present. Otherwise, first-line treatment for the pain and malaise is a nonsteroidal anti-inflammatory drug (NSAID). Oral potassium iodide (360-900 mg/d) is considered second-line treatment and systemic corticosteroids are a third-line option.
For this patient, biopsy was deferred and diagnostic tests were all negative. She had notable pain and a history of good resolution of symptoms with prednisone (5 mg/d), so this drug was prescribed for a 7-day course. She was counseled to avoid taking the NSAIDs and prednisone together due to increased risk of gastritis and ulceration. Recurrent disease can be treated with dapsone (100 mg/d) or hydroxychloroquine (200 mg bid).
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Blake T, Manahan M, Rodins K. Erythema nodosum - a review of an uncommon panniculitis. Dermatol Online J. 2014;20:22376.
Blake T, Manahan M, Rodins K. Erythema nodosum - a review of an uncommon panniculitis. Dermatol Online J. 2014;20:22376.
Review finds evidence for beta-blockers for some rosacea symptoms
, while at the same time underscoring the paucity of evidence supporting their use, investigators reported.
“The evidence was highest for carvedilol and propranolol, two nonselective beta-blockers,” wrote the authors of the review, Jade G.M. Logger, MD, of the department of dermatology, Radboud University Medical Center in Nijmegen, the Netherlands, and coauthors. Their review is in the Journal of the American Academy of Dermatology.
The systematic review included a case control study of 53,927 patients and an equal number of controls that evaluated beta-blockers in general, but the remaining studies and case reports included only 106 patients in total. The largest was a prospective cohort study of propranolol in 63 patients. Other studies included a 15-patient randomized clinical trial of nadolol published 31 years ago and three single-patient case reports.
The studies included patients with a history of failed therapies; only a small number of beta-blockers were evaluated. Outcomes reported in the studies varied widely, which ruled out doing a meta-analysis. “Erythema and flushing were assessed by using a wide spectrum of mostly subjective clinical and patient-based scores, and method standardization was often missing,” the researchers stated.
“Most studies showed improved erythema and flushing after initiation of oral beta-blockers,” Dr. Logger and colleagues wrote. Treatment of facial erythema and flushing remains a clinical challenge despite approved therapies, for which poor response and reactivation are common. “Diminishing erythema and flushing in rosacea is challenging because it hardly responds to conventional anti-inflammatory treatment,” they noted.
“The study adds no new evidence to support the use of beta-blockers,” Diane M. Thiboutot, MD, professor of dermatology at Penn State University, Hershey, said in an interview. “As the authors point out, the nine studies reviewed were of low quality with a variety of outcome measures that precluded generation of a meta-analysis, which would have represented new information.”
Dr. Thiboutot is lead author of a 2019 update of management options for rosacea published by the National Rosacea Society Expert Committee last year.. Beta blockers are among the drugs that are sometimes prescribed off label to help rosacea-associated flushing, along with nonsteroidal anti-inflammatory drugs, antihistamines, and clonidine, according to the update.
Dr. Logger and coauthors noted that beta-blockers come with risks, and can aggravate asthma and psoriasis and are contraindicated in patients with heart failure, cardiogenic shock, and other cardiovascular diseases, along with hyperactive airway and Raynaud’s disease. “It is important to monitor patients for adverse effects, especially blood pressure and heart rate,” they stated. Carvedilol and propranolol may have more antioxidant and anti-inflammatory properties than other nonselective beta-blockers that may curtail rosacea manifestations, they wrote.
They called for large, prospective clinical trials to more accurately assess the efficacy of beta-blockers in rosacea patients. “Researchers should further focus on the determination of the optimal dosage, treatment duration, and long-term therapeutic effects for adequate treatment of erythema and flushing in rosacea,” they said.
Getting those trials is challenging, Dr. Thiboutot said. “Objective and even subjective measurement of transient and persistent facial erythema is extremely challenging, particularly in the setting of a prospective clinical trial.” The trials would have to control for a number of variables, including room conditions, patient diet, and timing of medication, and large trials require multiple sites,” which could add to the variability of the data,” she said in the interview. Funding such trials would be difficult because adding an indication for rosacea-related symptoms would have limited commercial potential, she added.
Nonetheless, the studies would be welcome, Dr. Thiboutot said. “If standardized outcome measures for facial erythema were to be developed, a study would be more feasible.”
Dr. Logger disclosed financial relationships with Galderma, AbbVie, Novartis, Janssen, and LEO Pharma; one author disclosed conducting clinical trials for AbbVie and Novartis; the third author disclosed relationships with Galderma, Cutanea Life Sciences, AbbVie, Novartis, and Janssen, with fees paid to his institution. Dr. Thiboutot disclosed a financial relationship with Galderma.
SOURCE: Logger JGM et al. J Am Acad Dermatol. 2020 Oct;83(4):1088-97.
, while at the same time underscoring the paucity of evidence supporting their use, investigators reported.
“The evidence was highest for carvedilol and propranolol, two nonselective beta-blockers,” wrote the authors of the review, Jade G.M. Logger, MD, of the department of dermatology, Radboud University Medical Center in Nijmegen, the Netherlands, and coauthors. Their review is in the Journal of the American Academy of Dermatology.
The systematic review included a case control study of 53,927 patients and an equal number of controls that evaluated beta-blockers in general, but the remaining studies and case reports included only 106 patients in total. The largest was a prospective cohort study of propranolol in 63 patients. Other studies included a 15-patient randomized clinical trial of nadolol published 31 years ago and three single-patient case reports.
The studies included patients with a history of failed therapies; only a small number of beta-blockers were evaluated. Outcomes reported in the studies varied widely, which ruled out doing a meta-analysis. “Erythema and flushing were assessed by using a wide spectrum of mostly subjective clinical and patient-based scores, and method standardization was often missing,” the researchers stated.
“Most studies showed improved erythema and flushing after initiation of oral beta-blockers,” Dr. Logger and colleagues wrote. Treatment of facial erythema and flushing remains a clinical challenge despite approved therapies, for which poor response and reactivation are common. “Diminishing erythema and flushing in rosacea is challenging because it hardly responds to conventional anti-inflammatory treatment,” they noted.
“The study adds no new evidence to support the use of beta-blockers,” Diane M. Thiboutot, MD, professor of dermatology at Penn State University, Hershey, said in an interview. “As the authors point out, the nine studies reviewed were of low quality with a variety of outcome measures that precluded generation of a meta-analysis, which would have represented new information.”
Dr. Thiboutot is lead author of a 2019 update of management options for rosacea published by the National Rosacea Society Expert Committee last year.. Beta blockers are among the drugs that are sometimes prescribed off label to help rosacea-associated flushing, along with nonsteroidal anti-inflammatory drugs, antihistamines, and clonidine, according to the update.
Dr. Logger and coauthors noted that beta-blockers come with risks, and can aggravate asthma and psoriasis and are contraindicated in patients with heart failure, cardiogenic shock, and other cardiovascular diseases, along with hyperactive airway and Raynaud’s disease. “It is important to monitor patients for adverse effects, especially blood pressure and heart rate,” they stated. Carvedilol and propranolol may have more antioxidant and anti-inflammatory properties than other nonselective beta-blockers that may curtail rosacea manifestations, they wrote.
They called for large, prospective clinical trials to more accurately assess the efficacy of beta-blockers in rosacea patients. “Researchers should further focus on the determination of the optimal dosage, treatment duration, and long-term therapeutic effects for adequate treatment of erythema and flushing in rosacea,” they said.
Getting those trials is challenging, Dr. Thiboutot said. “Objective and even subjective measurement of transient and persistent facial erythema is extremely challenging, particularly in the setting of a prospective clinical trial.” The trials would have to control for a number of variables, including room conditions, patient diet, and timing of medication, and large trials require multiple sites,” which could add to the variability of the data,” she said in the interview. Funding such trials would be difficult because adding an indication for rosacea-related symptoms would have limited commercial potential, she added.
Nonetheless, the studies would be welcome, Dr. Thiboutot said. “If standardized outcome measures for facial erythema were to be developed, a study would be more feasible.”
Dr. Logger disclosed financial relationships with Galderma, AbbVie, Novartis, Janssen, and LEO Pharma; one author disclosed conducting clinical trials for AbbVie and Novartis; the third author disclosed relationships with Galderma, Cutanea Life Sciences, AbbVie, Novartis, and Janssen, with fees paid to his institution. Dr. Thiboutot disclosed a financial relationship with Galderma.
SOURCE: Logger JGM et al. J Am Acad Dermatol. 2020 Oct;83(4):1088-97.
, while at the same time underscoring the paucity of evidence supporting their use, investigators reported.
“The evidence was highest for carvedilol and propranolol, two nonselective beta-blockers,” wrote the authors of the review, Jade G.M. Logger, MD, of the department of dermatology, Radboud University Medical Center in Nijmegen, the Netherlands, and coauthors. Their review is in the Journal of the American Academy of Dermatology.
The systematic review included a case control study of 53,927 patients and an equal number of controls that evaluated beta-blockers in general, but the remaining studies and case reports included only 106 patients in total. The largest was a prospective cohort study of propranolol in 63 patients. Other studies included a 15-patient randomized clinical trial of nadolol published 31 years ago and three single-patient case reports.
The studies included patients with a history of failed therapies; only a small number of beta-blockers were evaluated. Outcomes reported in the studies varied widely, which ruled out doing a meta-analysis. “Erythema and flushing were assessed by using a wide spectrum of mostly subjective clinical and patient-based scores, and method standardization was often missing,” the researchers stated.
“Most studies showed improved erythema and flushing after initiation of oral beta-blockers,” Dr. Logger and colleagues wrote. Treatment of facial erythema and flushing remains a clinical challenge despite approved therapies, for which poor response and reactivation are common. “Diminishing erythema and flushing in rosacea is challenging because it hardly responds to conventional anti-inflammatory treatment,” they noted.
“The study adds no new evidence to support the use of beta-blockers,” Diane M. Thiboutot, MD, professor of dermatology at Penn State University, Hershey, said in an interview. “As the authors point out, the nine studies reviewed were of low quality with a variety of outcome measures that precluded generation of a meta-analysis, which would have represented new information.”
Dr. Thiboutot is lead author of a 2019 update of management options for rosacea published by the National Rosacea Society Expert Committee last year.. Beta blockers are among the drugs that are sometimes prescribed off label to help rosacea-associated flushing, along with nonsteroidal anti-inflammatory drugs, antihistamines, and clonidine, according to the update.
Dr. Logger and coauthors noted that beta-blockers come with risks, and can aggravate asthma and psoriasis and are contraindicated in patients with heart failure, cardiogenic shock, and other cardiovascular diseases, along with hyperactive airway and Raynaud’s disease. “It is important to monitor patients for adverse effects, especially blood pressure and heart rate,” they stated. Carvedilol and propranolol may have more antioxidant and anti-inflammatory properties than other nonselective beta-blockers that may curtail rosacea manifestations, they wrote.
They called for large, prospective clinical trials to more accurately assess the efficacy of beta-blockers in rosacea patients. “Researchers should further focus on the determination of the optimal dosage, treatment duration, and long-term therapeutic effects for adequate treatment of erythema and flushing in rosacea,” they said.
Getting those trials is challenging, Dr. Thiboutot said. “Objective and even subjective measurement of transient and persistent facial erythema is extremely challenging, particularly in the setting of a prospective clinical trial.” The trials would have to control for a number of variables, including room conditions, patient diet, and timing of medication, and large trials require multiple sites,” which could add to the variability of the data,” she said in the interview. Funding such trials would be difficult because adding an indication for rosacea-related symptoms would have limited commercial potential, she added.
Nonetheless, the studies would be welcome, Dr. Thiboutot said. “If standardized outcome measures for facial erythema were to be developed, a study would be more feasible.”
Dr. Logger disclosed financial relationships with Galderma, AbbVie, Novartis, Janssen, and LEO Pharma; one author disclosed conducting clinical trials for AbbVie and Novartis; the third author disclosed relationships with Galderma, Cutanea Life Sciences, AbbVie, Novartis, and Janssen, with fees paid to his institution. Dr. Thiboutot disclosed a financial relationship with Galderma.
SOURCE: Logger JGM et al. J Am Acad Dermatol. 2020 Oct;83(4):1088-97.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY