User login
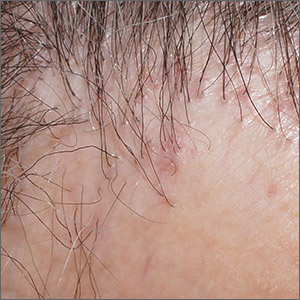
Hair loss and scalp papules
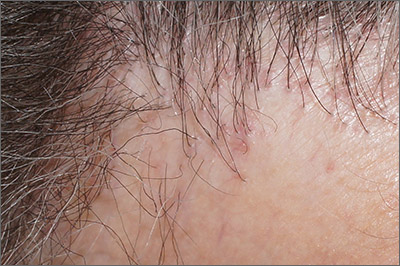
The punch biopsies were consistent with lichen planopilaris, an idiopathic, immune-mediated scarring alopecia that largely affects women between the ages of 40 and 70 years. In this variant of lichen planus, T cells target hair bulbs and cause destruction with scarring and permanent hair loss. Distribution may be patchy or may be more concentrated on the crown or involve the frontal scalp—a subtype called frontal fibrosing alopecia. Early recognition and intervention may save hair follicles and minimize disease severity.
The differential diagnosis includes traction alopecia, discoid lupus erythematosus, alopecia areata, centrifugal cicatricial alopecia, and folliculitis decalvans. The diagnosis may be confirmed with a scalp biopsy of actively inflamed follicles. Biopsy of scarred areas is likely to be nonspecific and unhelpful.
Treatment is targeted at slowing progression and symptom management. First-line therapy often includes potent corticosteroids (intralesional, topical, or systemic). Longer courses of steroid-sparing agents may be considered, including hydroxychloroquine, tacrolimus, ciclosporin, methotrexate, or acitretin. Hair styling and coloring, as well as hairpieces, often are used to conceal patches of hair loss. Hair transplantation is expensive but can be used to increase hair density in scarred areas once disease is controlled.
In this case, the patient was started on clobetasol solution 0.05% to be applied nightly to affected areas of the scalp. This treatment helped with the itching, but the inflammation and hair loss continued to worsen after 2 months. At that point, hydroxychloroquine 200 mg bid was added to the regimen, and hair loss and associated symptoms stopped. The patient remained on this therapy for 16 months. The hydroxychloroquine was then stopped, and the patient was advised to use the topical clobetasol, as needed.
Text courtesy of Tristan Reynolds, DO, Maine Dartmouth Family Medicine Residency, and Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
Errichetti E, Figini M, Croatto M, et al. Therapeutic management of classic lichen planopilaris: a systematic review. Clin Cosmet Investig Dermatol. 2018;11:91-102.

The punch biopsies were consistent with lichen planopilaris, an idiopathic, immune-mediated scarring alopecia that largely affects women between the ages of 40 and 70 years. In this variant of lichen planus, T cells target hair bulbs and cause destruction with scarring and permanent hair loss. Distribution may be patchy or may be more concentrated on the crown or involve the frontal scalp—a subtype called frontal fibrosing alopecia. Early recognition and intervention may save hair follicles and minimize disease severity.
The differential diagnosis includes traction alopecia, discoid lupus erythematosus, alopecia areata, centrifugal cicatricial alopecia, and folliculitis decalvans. The diagnosis may be confirmed with a scalp biopsy of actively inflamed follicles. Biopsy of scarred areas is likely to be nonspecific and unhelpful.
Treatment is targeted at slowing progression and symptom management. First-line therapy often includes potent corticosteroids (intralesional, topical, or systemic). Longer courses of steroid-sparing agents may be considered, including hydroxychloroquine, tacrolimus, ciclosporin, methotrexate, or acitretin. Hair styling and coloring, as well as hairpieces, often are used to conceal patches of hair loss. Hair transplantation is expensive but can be used to increase hair density in scarred areas once disease is controlled.
In this case, the patient was started on clobetasol solution 0.05% to be applied nightly to affected areas of the scalp. This treatment helped with the itching, but the inflammation and hair loss continued to worsen after 2 months. At that point, hydroxychloroquine 200 mg bid was added to the regimen, and hair loss and associated symptoms stopped. The patient remained on this therapy for 16 months. The hydroxychloroquine was then stopped, and the patient was advised to use the topical clobetasol, as needed.
Text courtesy of Tristan Reynolds, DO, Maine Dartmouth Family Medicine Residency, and Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).

The punch biopsies were consistent with lichen planopilaris, an idiopathic, immune-mediated scarring alopecia that largely affects women between the ages of 40 and 70 years. In this variant of lichen planus, T cells target hair bulbs and cause destruction with scarring and permanent hair loss. Distribution may be patchy or may be more concentrated on the crown or involve the frontal scalp—a subtype called frontal fibrosing alopecia. Early recognition and intervention may save hair follicles and minimize disease severity.
The differential diagnosis includes traction alopecia, discoid lupus erythematosus, alopecia areata, centrifugal cicatricial alopecia, and folliculitis decalvans. The diagnosis may be confirmed with a scalp biopsy of actively inflamed follicles. Biopsy of scarred areas is likely to be nonspecific and unhelpful.
Treatment is targeted at slowing progression and symptom management. First-line therapy often includes potent corticosteroids (intralesional, topical, or systemic). Longer courses of steroid-sparing agents may be considered, including hydroxychloroquine, tacrolimus, ciclosporin, methotrexate, or acitretin. Hair styling and coloring, as well as hairpieces, often are used to conceal patches of hair loss. Hair transplantation is expensive but can be used to increase hair density in scarred areas once disease is controlled.
In this case, the patient was started on clobetasol solution 0.05% to be applied nightly to affected areas of the scalp. This treatment helped with the itching, but the inflammation and hair loss continued to worsen after 2 months. At that point, hydroxychloroquine 200 mg bid was added to the regimen, and hair loss and associated symptoms stopped. The patient remained on this therapy for 16 months. The hydroxychloroquine was then stopped, and the patient was advised to use the topical clobetasol, as needed.
Text courtesy of Tristan Reynolds, DO, Maine Dartmouth Family Medicine Residency, and Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
Errichetti E, Figini M, Croatto M, et al. Therapeutic management of classic lichen planopilaris: a systematic review. Clin Cosmet Investig Dermatol. 2018;11:91-102.
Errichetti E, Figini M, Croatto M, et al. Therapeutic management of classic lichen planopilaris: a systematic review. Clin Cosmet Investig Dermatol. 2018;11:91-102.
EMA panel backs baricitinib for moderate to severe atopic dermatitis
The
Baricitinib (Olumiant) is already approved in the European Union and the United States to treat moderate to severe active rheumatoid arthritis.
If approved in Europe, it will be the first Janus kinase (JAK) inhibitor and first oral medication indicated to treat patients with AD.
The CHMP’s positive opinion on baricitinib for AD was based on three phase 3, randomized, double-blind, placebo-controlled studies where the JAK inhibitor was used alone or in combination with topical treatments in adults with moderate to severe AD for whom topical treatments were insufficient or not tolerated. In all three studies, baricitinib was shown to be more effective than placebo in achieving skin that is “clear” or “almost clear” at 16 weeks.
“Patients living with AD face difficulties on a daily basis, and this CHMP opinion marks an important milestone in providing adult AD patients with a new potential treatment option,” Thomas Bieber, MD, PhD, professor of dermatology and allergy, University of Bonn (Germany), said in a company news release.
The most common side effects with baricitinib in clinical trials include increased LDL cholesterol, upper respiratory tract infections, and headache.
Patients receiving baricitinib, particularly in combination with immunosuppressants, are at risk of developing serious infections that may lead to hospitalization or death. If a serious infection develops, baricitinib should be stopped until the infection is controlled.
The CHMP’s positive opinion will be sent to the European Commission, which will adopt a final decision regarding an European Union–wide marketing authorization. Once granted, each member state will make decisions about price and reimbursement, taking into account the potential role/use of baricitinib in the context of that country’s national health system.
A version of this story originally appeared on Medscape.com.
The
Baricitinib (Olumiant) is already approved in the European Union and the United States to treat moderate to severe active rheumatoid arthritis.
If approved in Europe, it will be the first Janus kinase (JAK) inhibitor and first oral medication indicated to treat patients with AD.
The CHMP’s positive opinion on baricitinib for AD was based on three phase 3, randomized, double-blind, placebo-controlled studies where the JAK inhibitor was used alone or in combination with topical treatments in adults with moderate to severe AD for whom topical treatments were insufficient or not tolerated. In all three studies, baricitinib was shown to be more effective than placebo in achieving skin that is “clear” or “almost clear” at 16 weeks.
“Patients living with AD face difficulties on a daily basis, and this CHMP opinion marks an important milestone in providing adult AD patients with a new potential treatment option,” Thomas Bieber, MD, PhD, professor of dermatology and allergy, University of Bonn (Germany), said in a company news release.
The most common side effects with baricitinib in clinical trials include increased LDL cholesterol, upper respiratory tract infections, and headache.
Patients receiving baricitinib, particularly in combination with immunosuppressants, are at risk of developing serious infections that may lead to hospitalization or death. If a serious infection develops, baricitinib should be stopped until the infection is controlled.
The CHMP’s positive opinion will be sent to the European Commission, which will adopt a final decision regarding an European Union–wide marketing authorization. Once granted, each member state will make decisions about price and reimbursement, taking into account the potential role/use of baricitinib in the context of that country’s national health system.
A version of this story originally appeared on Medscape.com.
The
Baricitinib (Olumiant) is already approved in the European Union and the United States to treat moderate to severe active rheumatoid arthritis.
If approved in Europe, it will be the first Janus kinase (JAK) inhibitor and first oral medication indicated to treat patients with AD.
The CHMP’s positive opinion on baricitinib for AD was based on three phase 3, randomized, double-blind, placebo-controlled studies where the JAK inhibitor was used alone or in combination with topical treatments in adults with moderate to severe AD for whom topical treatments were insufficient or not tolerated. In all three studies, baricitinib was shown to be more effective than placebo in achieving skin that is “clear” or “almost clear” at 16 weeks.
“Patients living with AD face difficulties on a daily basis, and this CHMP opinion marks an important milestone in providing adult AD patients with a new potential treatment option,” Thomas Bieber, MD, PhD, professor of dermatology and allergy, University of Bonn (Germany), said in a company news release.
The most common side effects with baricitinib in clinical trials include increased LDL cholesterol, upper respiratory tract infections, and headache.
Patients receiving baricitinib, particularly in combination with immunosuppressants, are at risk of developing serious infections that may lead to hospitalization or death. If a serious infection develops, baricitinib should be stopped until the infection is controlled.
The CHMP’s positive opinion will be sent to the European Commission, which will adopt a final decision regarding an European Union–wide marketing authorization. Once granted, each member state will make decisions about price and reimbursement, taking into account the potential role/use of baricitinib in the context of that country’s national health system.
A version of this story originally appeared on Medscape.com.
Three-step approach may help relieve one of the itchiest vulvar conditions
A three-step approach may help relieve itch in patients with lichen simplex chronicus, “one of the itchiest conditions that we ever see on the vulva,” an expert advised at the virtual conference on diseases of the vulva and vagina, hosted by the International Society for the Study of Vulvovaginal Disease.
For some patients, such as those with excessive sweating or underlying psoriasis, seeing a dermatologist may be beneficial, physicians at the meeting suggested.
Treatment should aim to optimize epithelial barrier function, reduce inflammation, and stop scratching, Lynette Margesson, MD, said in a lecture at the biennial meeting, which is held by the International Society for the Study of Vulvovaginal Disease (ISSVD). “With this condition, please look always for more than one problem.”
said Dr. Margesson, an obstetrician and gynecologist at Geisel School of Medicine at Dartmouth in Hanover, N.H. “It is because of chronic rubbing and scratching on top of something else.”
It may develop on top of atopic dermatitis, psoriasis, or contact dermatitis, as well as infection, lichen sclerosus, lichen planus, or neoplasia.
Lichen simplex chronicus is characterized by years of relentless itching, and patients may wake up at night scratching. The skin looks and feels leathery, and the condition can be localized or around the entire vulva. Heat, humidity, stress, and irritants may exacerbate the condition.
Patients often try to wash the rash away with scrubbers and cleansers, which only makes it worse, Dr. Margesson said.
To get patients better, improve barrier function, such as by controlling infections, reducing sweating, avoiding irritants, and stopping excessive hygiene. Immediate therapy may include soaks, cool compresses, and ointments.
A superpotent steroid taper (e.g., clobetasol 0.05% ointment), a prednisone taper, or intramuscular triamcinolone may reduce inflammation. Dr. Margesson usually uses clobetasol, although this treatment or halobetasol can burn if patients have open skin. In such cases, she uses prednisone or intramuscular triamcinolone.
Sedating medications may help patients stop scratching, especially at night. Hydroxyzine, doxepin, or amitriptyline 2-3 hours before bedtime can help. Scratching can be a form of obsessive-compulsive disorder, and a small dose of citalopram may help during the day. Patients with significant psychological factors can be difficult to manage and tend to relapse easily, Dr. Margesson said.
If lichen simplex chronicus recurs, test for infections and allergies. “Maybe they need a mild corticosteroid all the time, like 2.5% hydrocortisone to alternate with your superpotent steroid so you can use it longer without thinning the skin,” she suggested.
Although Dr. Margesson does not often treat hyperhidrosis, addressing excessive sweating can make a big difference for patients, she said.
If a gynecologist identifies a patient who may benefit from treatment of hyperhidrosis but has limited experience with medications for this condition, it might make sense to work with a dermatologist, Aruna Venkatesan, MD, chief of dermatology and director of the genital dermatology clinic at Santa Clara Valley Medical Center in San Jose, Calif., suggested during a panel discussion. Most dermatologists treat hyperhidrosis regularly, she said.
Dermatologists also may help treat patients with psoriasis who need systemic medication, Dr. Margesson said.
“In terms of ... doing the lab monitoring and knowing what side effects to look out for, your colleagues who use these medicines more are going to be more comfortable with that,” Dr. Venkatesan said. They also may have more experience navigating insurance denials to obtain a therapy. “Don’t think you are passing the buck to someone else. Sometimes that is the right thing to do, to get that help from someone else.”
Dr. Margesson is an author for UpToDate. Dr. Venkatesan had no conflicts of interest.
A three-step approach may help relieve itch in patients with lichen simplex chronicus, “one of the itchiest conditions that we ever see on the vulva,” an expert advised at the virtual conference on diseases of the vulva and vagina, hosted by the International Society for the Study of Vulvovaginal Disease.
For some patients, such as those with excessive sweating or underlying psoriasis, seeing a dermatologist may be beneficial, physicians at the meeting suggested.
Treatment should aim to optimize epithelial barrier function, reduce inflammation, and stop scratching, Lynette Margesson, MD, said in a lecture at the biennial meeting, which is held by the International Society for the Study of Vulvovaginal Disease (ISSVD). “With this condition, please look always for more than one problem.”
said Dr. Margesson, an obstetrician and gynecologist at Geisel School of Medicine at Dartmouth in Hanover, N.H. “It is because of chronic rubbing and scratching on top of something else.”
It may develop on top of atopic dermatitis, psoriasis, or contact dermatitis, as well as infection, lichen sclerosus, lichen planus, or neoplasia.
Lichen simplex chronicus is characterized by years of relentless itching, and patients may wake up at night scratching. The skin looks and feels leathery, and the condition can be localized or around the entire vulva. Heat, humidity, stress, and irritants may exacerbate the condition.
Patients often try to wash the rash away with scrubbers and cleansers, which only makes it worse, Dr. Margesson said.
To get patients better, improve barrier function, such as by controlling infections, reducing sweating, avoiding irritants, and stopping excessive hygiene. Immediate therapy may include soaks, cool compresses, and ointments.
A superpotent steroid taper (e.g., clobetasol 0.05% ointment), a prednisone taper, or intramuscular triamcinolone may reduce inflammation. Dr. Margesson usually uses clobetasol, although this treatment or halobetasol can burn if patients have open skin. In such cases, she uses prednisone or intramuscular triamcinolone.
Sedating medications may help patients stop scratching, especially at night. Hydroxyzine, doxepin, or amitriptyline 2-3 hours before bedtime can help. Scratching can be a form of obsessive-compulsive disorder, and a small dose of citalopram may help during the day. Patients with significant psychological factors can be difficult to manage and tend to relapse easily, Dr. Margesson said.
If lichen simplex chronicus recurs, test for infections and allergies. “Maybe they need a mild corticosteroid all the time, like 2.5% hydrocortisone to alternate with your superpotent steroid so you can use it longer without thinning the skin,” she suggested.
Although Dr. Margesson does not often treat hyperhidrosis, addressing excessive sweating can make a big difference for patients, she said.
If a gynecologist identifies a patient who may benefit from treatment of hyperhidrosis but has limited experience with medications for this condition, it might make sense to work with a dermatologist, Aruna Venkatesan, MD, chief of dermatology and director of the genital dermatology clinic at Santa Clara Valley Medical Center in San Jose, Calif., suggested during a panel discussion. Most dermatologists treat hyperhidrosis regularly, she said.
Dermatologists also may help treat patients with psoriasis who need systemic medication, Dr. Margesson said.
“In terms of ... doing the lab monitoring and knowing what side effects to look out for, your colleagues who use these medicines more are going to be more comfortable with that,” Dr. Venkatesan said. They also may have more experience navigating insurance denials to obtain a therapy. “Don’t think you are passing the buck to someone else. Sometimes that is the right thing to do, to get that help from someone else.”
Dr. Margesson is an author for UpToDate. Dr. Venkatesan had no conflicts of interest.
A three-step approach may help relieve itch in patients with lichen simplex chronicus, “one of the itchiest conditions that we ever see on the vulva,” an expert advised at the virtual conference on diseases of the vulva and vagina, hosted by the International Society for the Study of Vulvovaginal Disease.
For some patients, such as those with excessive sweating or underlying psoriasis, seeing a dermatologist may be beneficial, physicians at the meeting suggested.
Treatment should aim to optimize epithelial barrier function, reduce inflammation, and stop scratching, Lynette Margesson, MD, said in a lecture at the biennial meeting, which is held by the International Society for the Study of Vulvovaginal Disease (ISSVD). “With this condition, please look always for more than one problem.”
said Dr. Margesson, an obstetrician and gynecologist at Geisel School of Medicine at Dartmouth in Hanover, N.H. “It is because of chronic rubbing and scratching on top of something else.”
It may develop on top of atopic dermatitis, psoriasis, or contact dermatitis, as well as infection, lichen sclerosus, lichen planus, or neoplasia.
Lichen simplex chronicus is characterized by years of relentless itching, and patients may wake up at night scratching. The skin looks and feels leathery, and the condition can be localized or around the entire vulva. Heat, humidity, stress, and irritants may exacerbate the condition.
Patients often try to wash the rash away with scrubbers and cleansers, which only makes it worse, Dr. Margesson said.
To get patients better, improve barrier function, such as by controlling infections, reducing sweating, avoiding irritants, and stopping excessive hygiene. Immediate therapy may include soaks, cool compresses, and ointments.
A superpotent steroid taper (e.g., clobetasol 0.05% ointment), a prednisone taper, or intramuscular triamcinolone may reduce inflammation. Dr. Margesson usually uses clobetasol, although this treatment or halobetasol can burn if patients have open skin. In such cases, she uses prednisone or intramuscular triamcinolone.
Sedating medications may help patients stop scratching, especially at night. Hydroxyzine, doxepin, or amitriptyline 2-3 hours before bedtime can help. Scratching can be a form of obsessive-compulsive disorder, and a small dose of citalopram may help during the day. Patients with significant psychological factors can be difficult to manage and tend to relapse easily, Dr. Margesson said.
If lichen simplex chronicus recurs, test for infections and allergies. “Maybe they need a mild corticosteroid all the time, like 2.5% hydrocortisone to alternate with your superpotent steroid so you can use it longer without thinning the skin,” she suggested.
Although Dr. Margesson does not often treat hyperhidrosis, addressing excessive sweating can make a big difference for patients, she said.
If a gynecologist identifies a patient who may benefit from treatment of hyperhidrosis but has limited experience with medications for this condition, it might make sense to work with a dermatologist, Aruna Venkatesan, MD, chief of dermatology and director of the genital dermatology clinic at Santa Clara Valley Medical Center in San Jose, Calif., suggested during a panel discussion. Most dermatologists treat hyperhidrosis regularly, she said.
Dermatologists also may help treat patients with psoriasis who need systemic medication, Dr. Margesson said.
“In terms of ... doing the lab monitoring and knowing what side effects to look out for, your colleagues who use these medicines more are going to be more comfortable with that,” Dr. Venkatesan said. They also may have more experience navigating insurance denials to obtain a therapy. “Don’t think you are passing the buck to someone else. Sometimes that is the right thing to do, to get that help from someone else.”
Dr. Margesson is an author for UpToDate. Dr. Venkatesan had no conflicts of interest.
FROM ISSVD BIENNIAL CONFERENCE
What a Slow Boil Got Him
ANSWER
The correct answer is hidradenitis suppurativa (HS; choice “c”).
DISCUSSION
HS (also known as acne inversa) is a relatively common condition first described in 1839 by French physician Frederick Velpeau, who thought it was probably a form of acne. Because HS only affects intertriginous areas (groin, axillae, inframammary, intergluteal, and abdominal folds rich in apocrine glands), during the next 130 years, it was presumed that this disease was solely caused by malfunctioning apocrine glands.
Though experts disagree about the true etiology of HS, most agree that it is caused by several environmental and genetic factors leading to plugged or malfunctioning hair follicles and apocrine glands that become inflamed, swollen, painful, and purulent. In severe cases, tracts develop between lesions, resulting in the formation of ropy, linear hypertrophic scarring. Early on and in milder cases, comedones can manifest—often in multiples—along with widely scattered cysts. It appears that a case can be made for a genetic predisposition to develop HS because at least one-third of affected patients report a positive family history.
Females (usually postpubescent) are affected at 3 times the rate of males. Hormones, especially androgens, appear to play a role. This assertion is bolstered not only by the age of onset, but also because HS has been observed to improve after pregnancy and after menopause.
Though not causative, smoking and obesity have a strong statistical correlation with HS as exacerbating factors. Many patients with HS find movement from exercise too painful, which may contribute to obesity.
Because HS has a wide range of clinical presentations, the Hurley Staging scale can help clinicians distinguish the condition’s severity by stages I, II, or III. Stage I manifests with scant signs of disease, including comedones and sparsely separated small cysts. Stage II includes numerous painful cysts with tract formation between lesions. Stage III involves symptoms described in the other stages as well as multiple large, painful, draining cysts; widespread erythema; and extensive hypertrophic scarring.
Regarding the differential, the culture would have identified community-acquired methicillin-resistant Staphylococcus aureus (choice “a”) or flesh-eating bacterial infection (choice “d”), but neither would be as chronic as the patient’s disease. Acne conglobata (choice “b”) manifests with chronic dense acne largely confined to the trunk. Interestingly, acne conglobata, HS, dissecting cellulitis of the scalp, and pilonidal cyst are collectively known as the follicular occlusion tetrad.
TREATMENT
Treatment for HS is limited. Adalimumab is the most recent and promising biologic, though its use comes at a high cost ($60,000/y) and with an adverse effect profile that should give any prescriber pause. Also, the best one could hope for in using adalimumab is about a 30% improvement in the patient.
Before using a biologic, other options should at least be considered. Though not entirely satisfactory, there are oral antibiotics (minocycline, trimethoprim/sulfa, or a combination of rifampin and clindamycin) or intralesional steroids. Incision and drainage of individual lesions and isotretinoin therapy can temporarily relieve pain, but they are not effective as long-term therapies. Large-scale surgical extirpation of the affected tissue can be considered in extreme cases, but there can be recurrences and associated morbidity (eg, chronic lymphedema in the affected arm, extreme scarring, and postoperative pain).
ANSWER
The correct answer is hidradenitis suppurativa (HS; choice “c”).
DISCUSSION
HS (also known as acne inversa) is a relatively common condition first described in 1839 by French physician Frederick Velpeau, who thought it was probably a form of acne. Because HS only affects intertriginous areas (groin, axillae, inframammary, intergluteal, and abdominal folds rich in apocrine glands), during the next 130 years, it was presumed that this disease was solely caused by malfunctioning apocrine glands.
Though experts disagree about the true etiology of HS, most agree that it is caused by several environmental and genetic factors leading to plugged or malfunctioning hair follicles and apocrine glands that become inflamed, swollen, painful, and purulent. In severe cases, tracts develop between lesions, resulting in the formation of ropy, linear hypertrophic scarring. Early on and in milder cases, comedones can manifest—often in multiples—along with widely scattered cysts. It appears that a case can be made for a genetic predisposition to develop HS because at least one-third of affected patients report a positive family history.
Females (usually postpubescent) are affected at 3 times the rate of males. Hormones, especially androgens, appear to play a role. This assertion is bolstered not only by the age of onset, but also because HS has been observed to improve after pregnancy and after menopause.
Though not causative, smoking and obesity have a strong statistical correlation with HS as exacerbating factors. Many patients with HS find movement from exercise too painful, which may contribute to obesity.
Because HS has a wide range of clinical presentations, the Hurley Staging scale can help clinicians distinguish the condition’s severity by stages I, II, or III. Stage I manifests with scant signs of disease, including comedones and sparsely separated small cysts. Stage II includes numerous painful cysts with tract formation between lesions. Stage III involves symptoms described in the other stages as well as multiple large, painful, draining cysts; widespread erythema; and extensive hypertrophic scarring.
Regarding the differential, the culture would have identified community-acquired methicillin-resistant Staphylococcus aureus (choice “a”) or flesh-eating bacterial infection (choice “d”), but neither would be as chronic as the patient’s disease. Acne conglobata (choice “b”) manifests with chronic dense acne largely confined to the trunk. Interestingly, acne conglobata, HS, dissecting cellulitis of the scalp, and pilonidal cyst are collectively known as the follicular occlusion tetrad.
TREATMENT
Treatment for HS is limited. Adalimumab is the most recent and promising biologic, though its use comes at a high cost ($60,000/y) and with an adverse effect profile that should give any prescriber pause. Also, the best one could hope for in using adalimumab is about a 30% improvement in the patient.
Before using a biologic, other options should at least be considered. Though not entirely satisfactory, there are oral antibiotics (minocycline, trimethoprim/sulfa, or a combination of rifampin and clindamycin) or intralesional steroids. Incision and drainage of individual lesions and isotretinoin therapy can temporarily relieve pain, but they are not effective as long-term therapies. Large-scale surgical extirpation of the affected tissue can be considered in extreme cases, but there can be recurrences and associated morbidity (eg, chronic lymphedema in the affected arm, extreme scarring, and postoperative pain).
ANSWER
The correct answer is hidradenitis suppurativa (HS; choice “c”).
DISCUSSION
HS (also known as acne inversa) is a relatively common condition first described in 1839 by French physician Frederick Velpeau, who thought it was probably a form of acne. Because HS only affects intertriginous areas (groin, axillae, inframammary, intergluteal, and abdominal folds rich in apocrine glands), during the next 130 years, it was presumed that this disease was solely caused by malfunctioning apocrine glands.
Though experts disagree about the true etiology of HS, most agree that it is caused by several environmental and genetic factors leading to plugged or malfunctioning hair follicles and apocrine glands that become inflamed, swollen, painful, and purulent. In severe cases, tracts develop between lesions, resulting in the formation of ropy, linear hypertrophic scarring. Early on and in milder cases, comedones can manifest—often in multiples—along with widely scattered cysts. It appears that a case can be made for a genetic predisposition to develop HS because at least one-third of affected patients report a positive family history.
Females (usually postpubescent) are affected at 3 times the rate of males. Hormones, especially androgens, appear to play a role. This assertion is bolstered not only by the age of onset, but also because HS has been observed to improve after pregnancy and after menopause.
Though not causative, smoking and obesity have a strong statistical correlation with HS as exacerbating factors. Many patients with HS find movement from exercise too painful, which may contribute to obesity.
Because HS has a wide range of clinical presentations, the Hurley Staging scale can help clinicians distinguish the condition’s severity by stages I, II, or III. Stage I manifests with scant signs of disease, including comedones and sparsely separated small cysts. Stage II includes numerous painful cysts with tract formation between lesions. Stage III involves symptoms described in the other stages as well as multiple large, painful, draining cysts; widespread erythema; and extensive hypertrophic scarring.
Regarding the differential, the culture would have identified community-acquired methicillin-resistant Staphylococcus aureus (choice “a”) or flesh-eating bacterial infection (choice “d”), but neither would be as chronic as the patient’s disease. Acne conglobata (choice “b”) manifests with chronic dense acne largely confined to the trunk. Interestingly, acne conglobata, HS, dissecting cellulitis of the scalp, and pilonidal cyst are collectively known as the follicular occlusion tetrad.
TREATMENT
Treatment for HS is limited. Adalimumab is the most recent and promising biologic, though its use comes at a high cost ($60,000/y) and with an adverse effect profile that should give any prescriber pause. Also, the best one could hope for in using adalimumab is about a 30% improvement in the patient.
Before using a biologic, other options should at least be considered. Though not entirely satisfactory, there are oral antibiotics (minocycline, trimethoprim/sulfa, or a combination of rifampin and clindamycin) or intralesional steroids. Incision and drainage of individual lesions and isotretinoin therapy can temporarily relieve pain, but they are not effective as long-term therapies. Large-scale surgical extirpation of the affected tissue can be considered in extreme cases, but there can be recurrences and associated morbidity (eg, chronic lymphedema in the affected arm, extreme scarring, and postoperative pain).
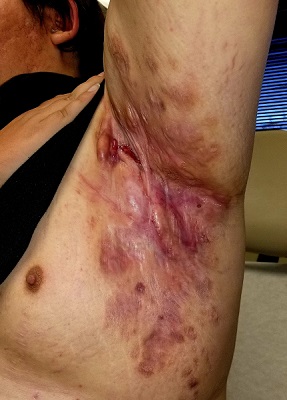
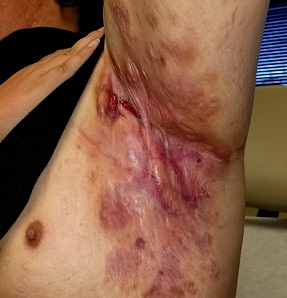
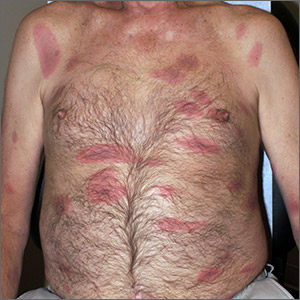
Boils first manifested on this 25-year-old man’s axillae when he was 13 (at the onset of puberty). As he grew older, they appeared in other intertriginous areas, including the groin and between the buttocks. As he became increasingly overweight, the folds under his breasts and abdomen became affected.
Over the years, several primary care providers consistently diagnosed him with either boils or staph infection. They also would reliably admonish him regarding his weight and lack of cleanliness. Treatment included various oral antibiotics that would only calm the condition for a short time. At no point was he referred to dermatology. Family history revealed similar skin problems in his mother and 1 sister.
Recently, his lesions have grown more numerous and painful. They drain pustular fluid, which makes it difficult for him to function in classrooms or at work. Even walking is excruciatingly painful. As a result, he is more sedentary. He also has started smoking.
On examination, the patient is 5 ft 8 in and grossly obese (weight, 300 lb). His affect is decidedly morose. His gait is ponderous and halting. His type 4 skin is swarthy, sweaty, and oily.
All intertriginous areas are similarly affected with large, ropy keloidal and hypertrophic scars, as well as diffuse bright red erythema and edema. There are dozens of single and multiple comedones, along with multiple draining sinuses.
A bacterial culture is collected from the fluid draining from the lesions. The results show a mixed, normal, cutaneous flora—including staph epidermidis—with no predominant organism.
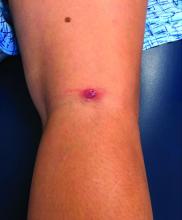
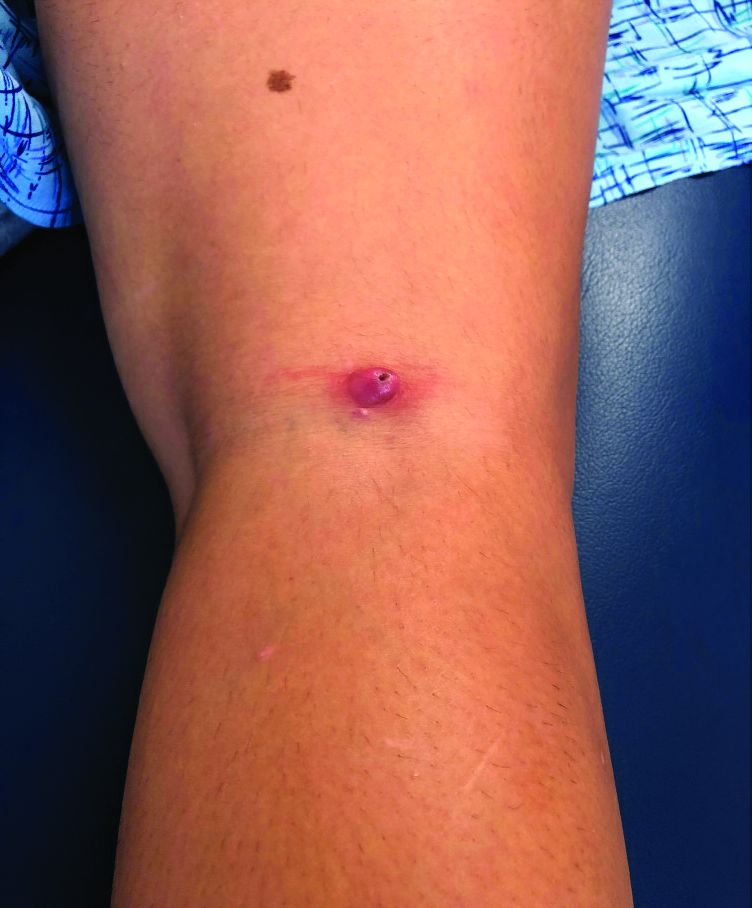
A teen girl presents with a pinkish-red bump on her right leg
This atypical lesion might warrant a biopsy. However, upon closer examination, you can appreciate a small papule with a whitish center, at the inferior margin of the tumor (6 o’clock), and another flat-topped papule with a white center several centimeters inferior-lateral to the lesion, both consistent with molluscum lesions. Therefore, the tumor is consistent with a giant molluscum contagiosum.
Molluscum contagiosum is a cutaneous viral infection caused by the poxvirus, which commonly affects children. It can spread easily by direct physical contact, fomites, and autoinoculation.1 It usually presents with skin-colored or pink pearly dome-shaped papules with central umbilication that can occur anywhere on the face or body. The skin lesions can be asymptomatic or pruritic. When the size of the molluscum is 0.5 cm or more in diameter, it is considered a giant molluscum. Atypical size and appearance may be seen in patients with altered or impaired immunity such as those with HIV.2,3 Giant molluscum has been reported in immunocompetent patients as well.4,5
The diagnosis of molluscum contagiosum usually is made clinically. Our patient had typically appearing molluscum lesions approximate to the larger lesion of concern. She was overall healthy without any history of impaired immunity so no further work-up was pursued. However, a biopsy of the skin lesion may be considered if the diagnosis is unclear.
What’s the treatment plan?
Treatment may not be necessary for molluscum contagiosum because it is often self-limited in immunocompetent children, although it can take many months to years to resolve. Treatment may be considered to reduce autoinoculation or risk of transmission because of close contact to others, to alleviate discomfort, including itching, to reduce cosmetic concerns and to prevent secondary infection.6
The most common treatments for molluscum contagiosum are cantharidin or cryotherapy. Other treatment available include topical retinoids, immunomodulators such as cimetidine, or antivirals such as cidofovir.1 Lesions with or without treatment may exhibit the BOTE (beginning of the end) sign, which is an apparent worsening associated with the body’s immune response to the molluscum virus and generally indicates imminent resolution.
What’s the differential diagnosis?
The differential diagnosis for giant molluscum contagiosum includes epidermal inclusion cyst, skin tag, pilomatrixoma, and amelanotic melanoma.
Epidermal inclusion cyst typically presents as a firm, mobile nodule under the skin with central punctum, which can enlarge and become inflamed. It can be painful, especially when infected. Definitive treatment is surgical excision because it rarely resolves spontaneously.
Skin tags, also known as acrochordons, are benign skin-colored papules most often found in the skin folds. People with obesity and type 2 diabetes are at higher risk for skin tags. Skin tags may be treated with cryotherapy, surgical excision, or ligation.
Pilomatrixoma is a benign skin tumor derived from hair matrix cells. It is usually a nontender, firm, skin-colored or red-purple subcutaneous nodule that may have calcifications. Treatment is surgical excision.
Amelanotic melanoma is a melanoma with little or no pigment and can present as a skin- or red-colored nodule. While these are quite uncommon, recognition that many pediatric melanomas present as amelanotic lesions makes it important to consider this in the differential diagnosis of growing papules and nodules.7 Treatment and prognosis is similar to that of pigmented melanoma, but as it is often clinically challenging to diagnose because of atypical features, it may be detected in more advanced stages.
Our patient underwent cryotherapy with liquid nitrogen to the nodule given the large size of the lesion, with resolution without recurrence.
Dr. Lee is a pediatric dermatology research fellow in the division of pediatric and adolescent dermatology at the University of California, San Diego and Rady Children’s Hospital–San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Neither Dr. Lee nor Dr. Eichenfield had any relevant financial disclosures. Email them at pdnews@mdedge.com.
References
1. Recent Pat Inflamm Allergy Drug Discov. 2017. doi: 10.2174/1872213X11666170518114456.
2. J Epidemiol Glob Health. 2013 Dec. doi: 10.1016/j.jegh.2013.06.002.
3. Trop Doct. 2015 Apr. doi: 10.1177/0049475514568133.
4. J Pak Med Assoc. 2013 Jun;63(6):778-9.
5. Dermatol Pract Concept. 2016 Jul. doi: 10.5826/dpc.0603a15.
6 Molluscum Contagiosum, in “Red Book: 2018 Report of the Committee on Infectious Diseases,” 31st ed. (Itasca, Ill.: American Academy of Pediatrics, 2018, pp. 565-66).
7. J Am Acad Dermatol. 2013 Jun. doi: 10.1016/j.jaad.2012.12.953.
This atypical lesion might warrant a biopsy. However, upon closer examination, you can appreciate a small papule with a whitish center, at the inferior margin of the tumor (6 o’clock), and another flat-topped papule with a white center several centimeters inferior-lateral to the lesion, both consistent with molluscum lesions. Therefore, the tumor is consistent with a giant molluscum contagiosum.
Molluscum contagiosum is a cutaneous viral infection caused by the poxvirus, which commonly affects children. It can spread easily by direct physical contact, fomites, and autoinoculation.1 It usually presents with skin-colored or pink pearly dome-shaped papules with central umbilication that can occur anywhere on the face or body. The skin lesions can be asymptomatic or pruritic. When the size of the molluscum is 0.5 cm or more in diameter, it is considered a giant molluscum. Atypical size and appearance may be seen in patients with altered or impaired immunity such as those with HIV.2,3 Giant molluscum has been reported in immunocompetent patients as well.4,5
The diagnosis of molluscum contagiosum usually is made clinically. Our patient had typically appearing molluscum lesions approximate to the larger lesion of concern. She was overall healthy without any history of impaired immunity so no further work-up was pursued. However, a biopsy of the skin lesion may be considered if the diagnosis is unclear.
What’s the treatment plan?
Treatment may not be necessary for molluscum contagiosum because it is often self-limited in immunocompetent children, although it can take many months to years to resolve. Treatment may be considered to reduce autoinoculation or risk of transmission because of close contact to others, to alleviate discomfort, including itching, to reduce cosmetic concerns and to prevent secondary infection.6
The most common treatments for molluscum contagiosum are cantharidin or cryotherapy. Other treatment available include topical retinoids, immunomodulators such as cimetidine, or antivirals such as cidofovir.1 Lesions with or without treatment may exhibit the BOTE (beginning of the end) sign, which is an apparent worsening associated with the body’s immune response to the molluscum virus and generally indicates imminent resolution.
What’s the differential diagnosis?
The differential diagnosis for giant molluscum contagiosum includes epidermal inclusion cyst, skin tag, pilomatrixoma, and amelanotic melanoma.
Epidermal inclusion cyst typically presents as a firm, mobile nodule under the skin with central punctum, which can enlarge and become inflamed. It can be painful, especially when infected. Definitive treatment is surgical excision because it rarely resolves spontaneously.
Skin tags, also known as acrochordons, are benign skin-colored papules most often found in the skin folds. People with obesity and type 2 diabetes are at higher risk for skin tags. Skin tags may be treated with cryotherapy, surgical excision, or ligation.
Pilomatrixoma is a benign skin tumor derived from hair matrix cells. It is usually a nontender, firm, skin-colored or red-purple subcutaneous nodule that may have calcifications. Treatment is surgical excision.
Amelanotic melanoma is a melanoma with little or no pigment and can present as a skin- or red-colored nodule. While these are quite uncommon, recognition that many pediatric melanomas present as amelanotic lesions makes it important to consider this in the differential diagnosis of growing papules and nodules.7 Treatment and prognosis is similar to that of pigmented melanoma, but as it is often clinically challenging to diagnose because of atypical features, it may be detected in more advanced stages.
Our patient underwent cryotherapy with liquid nitrogen to the nodule given the large size of the lesion, with resolution without recurrence.
Dr. Lee is a pediatric dermatology research fellow in the division of pediatric and adolescent dermatology at the University of California, San Diego and Rady Children’s Hospital–San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Neither Dr. Lee nor Dr. Eichenfield had any relevant financial disclosures. Email them at pdnews@mdedge.com.
References
1. Recent Pat Inflamm Allergy Drug Discov. 2017. doi: 10.2174/1872213X11666170518114456.
2. J Epidemiol Glob Health. 2013 Dec. doi: 10.1016/j.jegh.2013.06.002.
3. Trop Doct. 2015 Apr. doi: 10.1177/0049475514568133.
4. J Pak Med Assoc. 2013 Jun;63(6):778-9.
5. Dermatol Pract Concept. 2016 Jul. doi: 10.5826/dpc.0603a15.
6 Molluscum Contagiosum, in “Red Book: 2018 Report of the Committee on Infectious Diseases,” 31st ed. (Itasca, Ill.: American Academy of Pediatrics, 2018, pp. 565-66).
7. J Am Acad Dermatol. 2013 Jun. doi: 10.1016/j.jaad.2012.12.953.
This atypical lesion might warrant a biopsy. However, upon closer examination, you can appreciate a small papule with a whitish center, at the inferior margin of the tumor (6 o’clock), and another flat-topped papule with a white center several centimeters inferior-lateral to the lesion, both consistent with molluscum lesions. Therefore, the tumor is consistent with a giant molluscum contagiosum.
Molluscum contagiosum is a cutaneous viral infection caused by the poxvirus, which commonly affects children. It can spread easily by direct physical contact, fomites, and autoinoculation.1 It usually presents with skin-colored or pink pearly dome-shaped papules with central umbilication that can occur anywhere on the face or body. The skin lesions can be asymptomatic or pruritic. When the size of the molluscum is 0.5 cm or more in diameter, it is considered a giant molluscum. Atypical size and appearance may be seen in patients with altered or impaired immunity such as those with HIV.2,3 Giant molluscum has been reported in immunocompetent patients as well.4,5
The diagnosis of molluscum contagiosum usually is made clinically. Our patient had typically appearing molluscum lesions approximate to the larger lesion of concern. She was overall healthy without any history of impaired immunity so no further work-up was pursued. However, a biopsy of the skin lesion may be considered if the diagnosis is unclear.
What’s the treatment plan?
Treatment may not be necessary for molluscum contagiosum because it is often self-limited in immunocompetent children, although it can take many months to years to resolve. Treatment may be considered to reduce autoinoculation or risk of transmission because of close contact to others, to alleviate discomfort, including itching, to reduce cosmetic concerns and to prevent secondary infection.6
The most common treatments for molluscum contagiosum are cantharidin or cryotherapy. Other treatment available include topical retinoids, immunomodulators such as cimetidine, or antivirals such as cidofovir.1 Lesions with or without treatment may exhibit the BOTE (beginning of the end) sign, which is an apparent worsening associated with the body’s immune response to the molluscum virus and generally indicates imminent resolution.
What’s the differential diagnosis?
The differential diagnosis for giant molluscum contagiosum includes epidermal inclusion cyst, skin tag, pilomatrixoma, and amelanotic melanoma.
Epidermal inclusion cyst typically presents as a firm, mobile nodule under the skin with central punctum, which can enlarge and become inflamed. It can be painful, especially when infected. Definitive treatment is surgical excision because it rarely resolves spontaneously.
Skin tags, also known as acrochordons, are benign skin-colored papules most often found in the skin folds. People with obesity and type 2 diabetes are at higher risk for skin tags. Skin tags may be treated with cryotherapy, surgical excision, or ligation.
Pilomatrixoma is a benign skin tumor derived from hair matrix cells. It is usually a nontender, firm, skin-colored or red-purple subcutaneous nodule that may have calcifications. Treatment is surgical excision.
Amelanotic melanoma is a melanoma with little or no pigment and can present as a skin- or red-colored nodule. While these are quite uncommon, recognition that many pediatric melanomas present as amelanotic lesions makes it important to consider this in the differential diagnosis of growing papules and nodules.7 Treatment and prognosis is similar to that of pigmented melanoma, but as it is often clinically challenging to diagnose because of atypical features, it may be detected in more advanced stages.
Our patient underwent cryotherapy with liquid nitrogen to the nodule given the large size of the lesion, with resolution without recurrence.
Dr. Lee is a pediatric dermatology research fellow in the division of pediatric and adolescent dermatology at the University of California, San Diego and Rady Children’s Hospital–San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Neither Dr. Lee nor Dr. Eichenfield had any relevant financial disclosures. Email them at pdnews@mdedge.com.
References
1. Recent Pat Inflamm Allergy Drug Discov. 2017. doi: 10.2174/1872213X11666170518114456.
2. J Epidemiol Glob Health. 2013 Dec. doi: 10.1016/j.jegh.2013.06.002.
3. Trop Doct. 2015 Apr. doi: 10.1177/0049475514568133.
4. J Pak Med Assoc. 2013 Jun;63(6):778-9.
5. Dermatol Pract Concept. 2016 Jul. doi: 10.5826/dpc.0603a15.
6 Molluscum Contagiosum, in “Red Book: 2018 Report of the Committee on Infectious Diseases,” 31st ed. (Itasca, Ill.: American Academy of Pediatrics, 2018, pp. 565-66).
7. J Am Acad Dermatol. 2013 Jun. doi: 10.1016/j.jaad.2012.12.953.
Biologics for psoriasis may also reduce coronary plaque
Biologics used as treatment for psoriasis may also help reduce lipid-rich necrotic core (LRNC), a high-risk plaque associated with cardiovascular events, recent research from a prospective, observational study suggests.
Cardiac CT scans performed on patients with psoriasis 1 year after starting biologic therapy revealed a reduction in LRNC, compared with patients who were not receiving biologics, according to Harry Choi, MD, of the National Heart, Lung, and Blood Institute at the National Institutes of Health and colleagues. The association with reduction in LRNC and biologic therapy remained significant when adjusted for type of biologic. “These findings demonstrate that LRNC may be modulated by the control of systemic inflammation,” the researchers wrote in their study, published Sept. 15 in Circulation: Cardiovascular Imaging.
Dr. Choi and colleagues evaluated 289 patients with psoriasis within the Psoriasis Atherosclerosis and Cardiometabolic Disease Initiative cohort. The patients had a mean age of 50 years and a mean body mass index of 29.4 kg/m2, as well as a mean Psoriasis Area and Severity Index (PASI) score of 6.0. At baseline, 29% of patients had hypertension, 41% had hyperlipidemia, their mean Framingham risk score was 1.9, and a three-quarters (212 of 289) had mild to moderate psoriasis.
Changes in LRNC were observed at 1 year, compared with baseline prior to and after receiving biologic therapy (124 patients) in comparison with patients who did not undergo biologic therapy (85 patients). Biologic therapies were grouped by type, which included anti–tumor necrosis factor (anti-TNF), anti–interleukin (IL)–12/23, and anti–IL-17 biologics.
There were a significant associations between LRNC and Framingham risk score (standardized beta coefficient, 0.12; 95% confidence interval, 0.00-0.15; P = .045) and severity of psoriasis (beta, 0.13; 95% CI, 0.01-0.26; P = .029) at baseline.
Key findings
The researchers found a significant reduction in LRNC 1 year after patients began biologic therapy (median, 2.97 mm2; interquartile range, 1.99-4.66), compared with baseline (median, 3.12 mm2; IQR, 1.84-4.35) (P = .028), while patients who did not receive biologic therapy had nonsignificantly higher LRNC after 1 year (median, 3.12 mm2; IQR, 1.82-4.60), compared with baseline measurements (median, 3.34 mm2; IQR, 2.04–4.74) (P = .06).
The results remained significant after the researchers adjusted for psoriasis severity, Framingham risk score, BMI, use of statins (beta, −0.09; 95% CI, −0.01 to −0.18; P = .033). Significant reductions in LRNC also remained when analyzing patients receiving anti-TNF, anti–IL-12/23, and anti–IL-17 biologics independently, and there were no significant between-group differences in reduction of LRNC.
The potential of biologics for improving vascular health
Discussing the study results in a press release from the American Heart Association, senior author Nehal N. Mehta, MD, MSCE, FAHA, chief of the Lab of Inflammation and Cardiometabolic Diseases at the NHLBI at NIH, compared the effect biologic therapy had on coronary plaque reduction with that of statins.
“There is approximately 6%-8% reduction in coronary plaque following therapy with statins. Similarly, our treatment with biologic therapy reduced coronary plaque by the same amount after one year. These findings suggest that biologic therapy to treat psoriasis may be just as beneficial as statin therapy on heart arteries,” Dr. Mehta said in the release.
In an interview, Nieca Goldberg, MD, medical director of NYU Women’s Heart Program at NYU Langone Health, echoed Dr. Mehta’s commments and said psoriasis carries the “potential to treat two conditions with the same drug.”
“We know conditions such as psoriatic arthritis and rheumatoid arthritis cause chronic inflammation. Chronic inflammation causes injury to blood vessels and high-risk coronary plaque. Individuals with these inflammatory conditions are at high risk for heart attack,” she said. “This study shows that biologic treatment for psoriatic arthritis can reduce the presence of high-risk plaque. It shows the potential to treat chronic inflammation and high-risk coronary plaque.”
While the results show an association between use of biologics and LRNC reduction, the study design was observational and patients had a short follow-up period. Dr. Goldberg noted more studies are needed to evaluate the effect of biologics on reducing cardiovascular events such as a myocardial infarction.
“We have never before been able to show healing of an inflamed plaque like this in humans. Biologic therapy reduces systemic inflammation and immune activation, and it has a favorable impact on improving overall vascular health,” Dr. Mehta said in the press release. “Imagine if we can treat both psoriasis and coronary heart disease with one therapy – that is the question to be asked in future studies.”
This study was funded with support from the NHLBI Intramural Research Program and the NIH Medical Research Scholars Program at the National Institutes of Health. One investigator reports financial relationships with numerous pharmaceutical companies. The other authors report no relevant conflicts of interest. Dr. Mehta also reports numerous such relationships. Dr. Goldberg reports no relevant conflicts of interest.
SOURCE: Choi H et al. Circ Cardiovasc Imaging. 2020 Sep;13(9):e011199.
Biologics used as treatment for psoriasis may also help reduce lipid-rich necrotic core (LRNC), a high-risk plaque associated with cardiovascular events, recent research from a prospective, observational study suggests.
Cardiac CT scans performed on patients with psoriasis 1 year after starting biologic therapy revealed a reduction in LRNC, compared with patients who were not receiving biologics, according to Harry Choi, MD, of the National Heart, Lung, and Blood Institute at the National Institutes of Health and colleagues. The association with reduction in LRNC and biologic therapy remained significant when adjusted for type of biologic. “These findings demonstrate that LRNC may be modulated by the control of systemic inflammation,” the researchers wrote in their study, published Sept. 15 in Circulation: Cardiovascular Imaging.
Dr. Choi and colleagues evaluated 289 patients with psoriasis within the Psoriasis Atherosclerosis and Cardiometabolic Disease Initiative cohort. The patients had a mean age of 50 years and a mean body mass index of 29.4 kg/m2, as well as a mean Psoriasis Area and Severity Index (PASI) score of 6.0. At baseline, 29% of patients had hypertension, 41% had hyperlipidemia, their mean Framingham risk score was 1.9, and a three-quarters (212 of 289) had mild to moderate psoriasis.
Changes in LRNC were observed at 1 year, compared with baseline prior to and after receiving biologic therapy (124 patients) in comparison with patients who did not undergo biologic therapy (85 patients). Biologic therapies were grouped by type, which included anti–tumor necrosis factor (anti-TNF), anti–interleukin (IL)–12/23, and anti–IL-17 biologics.
There were a significant associations between LRNC and Framingham risk score (standardized beta coefficient, 0.12; 95% confidence interval, 0.00-0.15; P = .045) and severity of psoriasis (beta, 0.13; 95% CI, 0.01-0.26; P = .029) at baseline.
Key findings
The researchers found a significant reduction in LRNC 1 year after patients began biologic therapy (median, 2.97 mm2; interquartile range, 1.99-4.66), compared with baseline (median, 3.12 mm2; IQR, 1.84-4.35) (P = .028), while patients who did not receive biologic therapy had nonsignificantly higher LRNC after 1 year (median, 3.12 mm2; IQR, 1.82-4.60), compared with baseline measurements (median, 3.34 mm2; IQR, 2.04–4.74) (P = .06).
The results remained significant after the researchers adjusted for psoriasis severity, Framingham risk score, BMI, use of statins (beta, −0.09; 95% CI, −0.01 to −0.18; P = .033). Significant reductions in LRNC also remained when analyzing patients receiving anti-TNF, anti–IL-12/23, and anti–IL-17 biologics independently, and there were no significant between-group differences in reduction of LRNC.
The potential of biologics for improving vascular health
Discussing the study results in a press release from the American Heart Association, senior author Nehal N. Mehta, MD, MSCE, FAHA, chief of the Lab of Inflammation and Cardiometabolic Diseases at the NHLBI at NIH, compared the effect biologic therapy had on coronary plaque reduction with that of statins.
“There is approximately 6%-8% reduction in coronary plaque following therapy with statins. Similarly, our treatment with biologic therapy reduced coronary plaque by the same amount after one year. These findings suggest that biologic therapy to treat psoriasis may be just as beneficial as statin therapy on heart arteries,” Dr. Mehta said in the release.
In an interview, Nieca Goldberg, MD, medical director of NYU Women’s Heart Program at NYU Langone Health, echoed Dr. Mehta’s commments and said psoriasis carries the “potential to treat two conditions with the same drug.”
“We know conditions such as psoriatic arthritis and rheumatoid arthritis cause chronic inflammation. Chronic inflammation causes injury to blood vessels and high-risk coronary plaque. Individuals with these inflammatory conditions are at high risk for heart attack,” she said. “This study shows that biologic treatment for psoriatic arthritis can reduce the presence of high-risk plaque. It shows the potential to treat chronic inflammation and high-risk coronary plaque.”
While the results show an association between use of biologics and LRNC reduction, the study design was observational and patients had a short follow-up period. Dr. Goldberg noted more studies are needed to evaluate the effect of biologics on reducing cardiovascular events such as a myocardial infarction.
“We have never before been able to show healing of an inflamed plaque like this in humans. Biologic therapy reduces systemic inflammation and immune activation, and it has a favorable impact on improving overall vascular health,” Dr. Mehta said in the press release. “Imagine if we can treat both psoriasis and coronary heart disease with one therapy – that is the question to be asked in future studies.”
This study was funded with support from the NHLBI Intramural Research Program and the NIH Medical Research Scholars Program at the National Institutes of Health. One investigator reports financial relationships with numerous pharmaceutical companies. The other authors report no relevant conflicts of interest. Dr. Mehta also reports numerous such relationships. Dr. Goldberg reports no relevant conflicts of interest.
SOURCE: Choi H et al. Circ Cardiovasc Imaging. 2020 Sep;13(9):e011199.
Biologics used as treatment for psoriasis may also help reduce lipid-rich necrotic core (LRNC), a high-risk plaque associated with cardiovascular events, recent research from a prospective, observational study suggests.
Cardiac CT scans performed on patients with psoriasis 1 year after starting biologic therapy revealed a reduction in LRNC, compared with patients who were not receiving biologics, according to Harry Choi, MD, of the National Heart, Lung, and Blood Institute at the National Institutes of Health and colleagues. The association with reduction in LRNC and biologic therapy remained significant when adjusted for type of biologic. “These findings demonstrate that LRNC may be modulated by the control of systemic inflammation,” the researchers wrote in their study, published Sept. 15 in Circulation: Cardiovascular Imaging.
Dr. Choi and colleagues evaluated 289 patients with psoriasis within the Psoriasis Atherosclerosis and Cardiometabolic Disease Initiative cohort. The patients had a mean age of 50 years and a mean body mass index of 29.4 kg/m2, as well as a mean Psoriasis Area and Severity Index (PASI) score of 6.0. At baseline, 29% of patients had hypertension, 41% had hyperlipidemia, their mean Framingham risk score was 1.9, and a three-quarters (212 of 289) had mild to moderate psoriasis.
Changes in LRNC were observed at 1 year, compared with baseline prior to and after receiving biologic therapy (124 patients) in comparison with patients who did not undergo biologic therapy (85 patients). Biologic therapies were grouped by type, which included anti–tumor necrosis factor (anti-TNF), anti–interleukin (IL)–12/23, and anti–IL-17 biologics.
There were a significant associations between LRNC and Framingham risk score (standardized beta coefficient, 0.12; 95% confidence interval, 0.00-0.15; P = .045) and severity of psoriasis (beta, 0.13; 95% CI, 0.01-0.26; P = .029) at baseline.
Key findings
The researchers found a significant reduction in LRNC 1 year after patients began biologic therapy (median, 2.97 mm2; interquartile range, 1.99-4.66), compared with baseline (median, 3.12 mm2; IQR, 1.84-4.35) (P = .028), while patients who did not receive biologic therapy had nonsignificantly higher LRNC after 1 year (median, 3.12 mm2; IQR, 1.82-4.60), compared with baseline measurements (median, 3.34 mm2; IQR, 2.04–4.74) (P = .06).
The results remained significant after the researchers adjusted for psoriasis severity, Framingham risk score, BMI, use of statins (beta, −0.09; 95% CI, −0.01 to −0.18; P = .033). Significant reductions in LRNC also remained when analyzing patients receiving anti-TNF, anti–IL-12/23, and anti–IL-17 biologics independently, and there were no significant between-group differences in reduction of LRNC.
The potential of biologics for improving vascular health
Discussing the study results in a press release from the American Heart Association, senior author Nehal N. Mehta, MD, MSCE, FAHA, chief of the Lab of Inflammation and Cardiometabolic Diseases at the NHLBI at NIH, compared the effect biologic therapy had on coronary plaque reduction with that of statins.
“There is approximately 6%-8% reduction in coronary plaque following therapy with statins. Similarly, our treatment with biologic therapy reduced coronary plaque by the same amount after one year. These findings suggest that biologic therapy to treat psoriasis may be just as beneficial as statin therapy on heart arteries,” Dr. Mehta said in the release.
In an interview, Nieca Goldberg, MD, medical director of NYU Women’s Heart Program at NYU Langone Health, echoed Dr. Mehta’s commments and said psoriasis carries the “potential to treat two conditions with the same drug.”
“We know conditions such as psoriatic arthritis and rheumatoid arthritis cause chronic inflammation. Chronic inflammation causes injury to blood vessels and high-risk coronary plaque. Individuals with these inflammatory conditions are at high risk for heart attack,” she said. “This study shows that biologic treatment for psoriatic arthritis can reduce the presence of high-risk plaque. It shows the potential to treat chronic inflammation and high-risk coronary plaque.”
While the results show an association between use of biologics and LRNC reduction, the study design was observational and patients had a short follow-up period. Dr. Goldberg noted more studies are needed to evaluate the effect of biologics on reducing cardiovascular events such as a myocardial infarction.
“We have never before been able to show healing of an inflamed plaque like this in humans. Biologic therapy reduces systemic inflammation and immune activation, and it has a favorable impact on improving overall vascular health,” Dr. Mehta said in the press release. “Imagine if we can treat both psoriasis and coronary heart disease with one therapy – that is the question to be asked in future studies.”
This study was funded with support from the NHLBI Intramural Research Program and the NIH Medical Research Scholars Program at the National Institutes of Health. One investigator reports financial relationships with numerous pharmaceutical companies. The other authors report no relevant conflicts of interest. Dr. Mehta also reports numerous such relationships. Dr. Goldberg reports no relevant conflicts of interest.
SOURCE: Choi H et al. Circ Cardiovasc Imaging. 2020 Sep;13(9):e011199.
FROM CIRCULATION: CARDIOVASCULAR IMAGING
Painful periocular rash
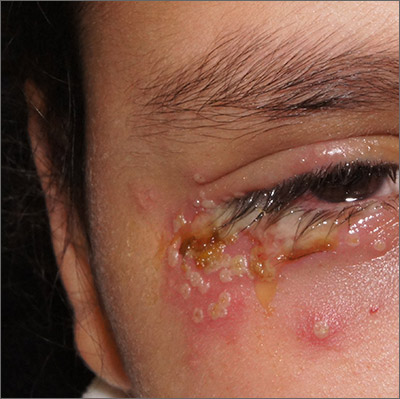
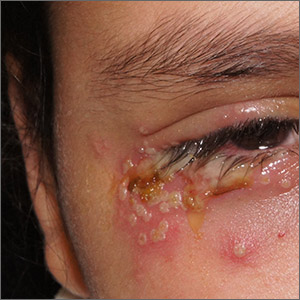
This patient was given a diagnosis of primary herpes simplex virus (HSV) based on the appearance of her eyelid. Swabs were performed for bacterial culture, and polymerase chain reaction (PCR) testing was done for HSV and varicella, but results were pending prior to her transfer to the Emergency Department (ED).
The patient was given a single dose of 800 mg oral acyclovir (200 mg/5mL) and 500 mg of oral cephalexin (250 mg/5mL) and referred to the ED for a more detailed eye exam and to exclude orbital erosions.
HSV classically causes clustered vesicles on an erythematous base. Superinfection with skin flora can cause pustules instead of vesicles. Severe complications of HSV can include widespread skin involvement, eczema herpeticum, local destruction, central nervous system involvement, throat infections (affecting airway and oral intake), and dissemination in immunocompromised hosts. Ocular or periorbital infections increase the risk of keratitis, corneal ulcers, and loss of sight. Viral involvement of the cornea is best seen with fluorescein staining.
In cases like this one, PCR is the preferred method of testing over viral cultures or serology, given its speed, accuracy, and temporal relevance. Ophthalmology referral is warranted, although it should not delay treatment. Topical and oral antivirals are both effective when treating corneal disease; patient preference should be considered.
Most cases of HSV may resolve without treatment; however, treatment started while vesicles are present and within 72 hours of infection may shorten the time of viral replication and prevent progression to stromal involvement.
After a 12-hour wait in the ED, this patient was seen by an ophthalmology resident who did not observe orbital erosions but did note umbilication and misdiagnosed molluscum contagiosum. Umbilication is not pathognomonic for molluscum; few experienced in diagnosing molluscum contagiosum would make this error.
The patient was instructed to stop the acyclovir. Two days later when the PCR came back positive for HSV-1 and the bacterial culture confirmed growth of superimposed Staphylococcus aureus, the patient had been lost to follow-up. A better approach would have been for the ophthalmology resident to continue the acyclovir until PCR excluded herpetic disease.
Text courtesy of Tristan Reynolds, DO, Maine Dartmouth Family Medicine Residency, and Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
Barker NH. Ocular herpes simplex. BMJ Clin Evid. 2008;2008:0707.

This patient was given a diagnosis of primary herpes simplex virus (HSV) based on the appearance of her eyelid. Swabs were performed for bacterial culture, and polymerase chain reaction (PCR) testing was done for HSV and varicella, but results were pending prior to her transfer to the Emergency Department (ED).
The patient was given a single dose of 800 mg oral acyclovir (200 mg/5mL) and 500 mg of oral cephalexin (250 mg/5mL) and referred to the ED for a more detailed eye exam and to exclude orbital erosions.
HSV classically causes clustered vesicles on an erythematous base. Superinfection with skin flora can cause pustules instead of vesicles. Severe complications of HSV can include widespread skin involvement, eczema herpeticum, local destruction, central nervous system involvement, throat infections (affecting airway and oral intake), and dissemination in immunocompromised hosts. Ocular or periorbital infections increase the risk of keratitis, corneal ulcers, and loss of sight. Viral involvement of the cornea is best seen with fluorescein staining.
In cases like this one, PCR is the preferred method of testing over viral cultures or serology, given its speed, accuracy, and temporal relevance. Ophthalmology referral is warranted, although it should not delay treatment. Topical and oral antivirals are both effective when treating corneal disease; patient preference should be considered.
Most cases of HSV may resolve without treatment; however, treatment started while vesicles are present and within 72 hours of infection may shorten the time of viral replication and prevent progression to stromal involvement.
After a 12-hour wait in the ED, this patient was seen by an ophthalmology resident who did not observe orbital erosions but did note umbilication and misdiagnosed molluscum contagiosum. Umbilication is not pathognomonic for molluscum; few experienced in diagnosing molluscum contagiosum would make this error.
The patient was instructed to stop the acyclovir. Two days later when the PCR came back positive for HSV-1 and the bacterial culture confirmed growth of superimposed Staphylococcus aureus, the patient had been lost to follow-up. A better approach would have been for the ophthalmology resident to continue the acyclovir until PCR excluded herpetic disease.
Text courtesy of Tristan Reynolds, DO, Maine Dartmouth Family Medicine Residency, and Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).

This patient was given a diagnosis of primary herpes simplex virus (HSV) based on the appearance of her eyelid. Swabs were performed for bacterial culture, and polymerase chain reaction (PCR) testing was done for HSV and varicella, but results were pending prior to her transfer to the Emergency Department (ED).
The patient was given a single dose of 800 mg oral acyclovir (200 mg/5mL) and 500 mg of oral cephalexin (250 mg/5mL) and referred to the ED for a more detailed eye exam and to exclude orbital erosions.
HSV classically causes clustered vesicles on an erythematous base. Superinfection with skin flora can cause pustules instead of vesicles. Severe complications of HSV can include widespread skin involvement, eczema herpeticum, local destruction, central nervous system involvement, throat infections (affecting airway and oral intake), and dissemination in immunocompromised hosts. Ocular or periorbital infections increase the risk of keratitis, corneal ulcers, and loss of sight. Viral involvement of the cornea is best seen with fluorescein staining.
In cases like this one, PCR is the preferred method of testing over viral cultures or serology, given its speed, accuracy, and temporal relevance. Ophthalmology referral is warranted, although it should not delay treatment. Topical and oral antivirals are both effective when treating corneal disease; patient preference should be considered.
Most cases of HSV may resolve without treatment; however, treatment started while vesicles are present and within 72 hours of infection may shorten the time of viral replication and prevent progression to stromal involvement.
After a 12-hour wait in the ED, this patient was seen by an ophthalmology resident who did not observe orbital erosions but did note umbilication and misdiagnosed molluscum contagiosum. Umbilication is not pathognomonic for molluscum; few experienced in diagnosing molluscum contagiosum would make this error.
The patient was instructed to stop the acyclovir. Two days later when the PCR came back positive for HSV-1 and the bacterial culture confirmed growth of superimposed Staphylococcus aureus, the patient had been lost to follow-up. A better approach would have been for the ophthalmology resident to continue the acyclovir until PCR excluded herpetic disease.
Text courtesy of Tristan Reynolds, DO, Maine Dartmouth Family Medicine Residency, and Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
Barker NH. Ocular herpes simplex. BMJ Clin Evid. 2008;2008:0707.
Barker NH. Ocular herpes simplex. BMJ Clin Evid. 2008;2008:0707.
COVID-19 outcomes no worse in patients on TNF inhibitors or methotrexate
Continued use of tumor necrosis factor inhibitors or methotrexate is acceptable in most patients who acquire COVID-19, results of a recent cohort study suggest.
Among patients on tumor necrosis factor inhibitors (TNFi) or methotrexate who developed COVID-19, death and hospitalization rates were similar to matched COVID-19 patients not on those medications, according to authors of the multicenter research network study.
Reassuringly, likelihood of hospitalization and mortality were not significantly different between 214 patients with COVID-19 taking TNFi or methotrexate and 31,862 matched COVID-19 patients not on those medications, according to the investigators, whose findings were published recently in the Journal of the American Academy of Dermatology.
Zachary Zinn, MD, corresponding author on the study, said in an interview that the findings suggest these medicines can be safely continued in the majority of patients taking them during the COVID-19 pandemic.
“If you’re a prescribing physician who’s giving patients TNF inhibitors or methotrexate or both, I think you can comfortably tell your patients there is good data that these do not lead to worse outcomes if you get COVID-19,” said Dr. Zinn, associate professor in the department of dermatology at West Virginia University, Morgantown.
The findings from these researchers corroborate a growing body of evidence suggesting that immunosuppressive treatments can be continued in patients with dermatologic and rheumatic conditions.
In recent guidance from the National Psoriasis Foundation, released Sept. 4, an expert consensus panel cited 15 studies that they said suggested that treatments for psoriasis or psoriatic arthritis “do not meaningfully alter the risk of acquiring SARS-CoV-2 infection or having worse COVID-19 outcomes.”
That said, the data to date are mainly from small case series and registry studies based on spontaneously reported COVID-19 cases, which suggests a continued need for shared decision making. In addition, chronic systemic corticosteroids should be avoided for management of psoriatic arthritis, the guidance states, based on rheumatology and gastroenterology literature suggesting this treatment is linked to worse COVID-19 outcomes.
In the interview, Dr. Zinn noted that some previous studies of immunosuppressive treatments in patients who acquire COVID-19 have aggregated data on numerous classes of biologic medications, lessening the strength of data for each specific medication.
“By focusing specifically on TNF inhibitors and methotrexate, this study gives better guidance to prescribers of these medications,” he said.
To see whether TNFi or methotrexate increased risk of worsened COVID-19 outcomes, Dr. Zinn and coinvestigators evaluated data from TriNetX, a research network that includes approximately 53 million unique patient records, predominantly in the United States.
They identified 32,076 adult patients with COVID-19, of whom 214 had recent exposure to TNFi or methotrexate. The patients in the TNFi/methotrexate group were similar in age to those without exposure to those drugs, at 55.1 versus 53.2 years, respectively. However, patients in the drug exposure group were more frequently White, female, and had substantially more comorbidities, including diabetes and obesity, according to the investigators.
Nevertheless, the likelihood of hospitalization was not statistically different in the TNFi/methotrexate group versus the non-TNFi/methotrexate group, with a risk ratio of 0.91 (95% confidence interval, 0.68-1.22; P = .5260).
Likewise, the likelihood of death was not different between groups, with a RR of 0.87 (95% CI, 0.42-1.78; P = .6958). Looking at subgroups of patients exposed to TNFi or methotrexate only didn’t change the results, the investigators added.
Taken together, the findings argue against interruption of these treatments because of the fear of the possibly worse COVID-19 outcomes, the investigators concluded, although they emphasized the need for more research.
“Because the COVID-19 pandemic is ongoing, there is a desperate need for evidence-based data on biologic and immunomodulator exposure in the setting of COVID-19 infection,” they wrote.
Dr. Zinn and coauthors reported no conflicts of interest and no funding sources related to the study.
SOURCE: Zinn Z et al. J Am Acad Dermatol. 2020 Sep 11. doi: 10.1016/j.jaad.2020.09.009.
Continued use of tumor necrosis factor inhibitors or methotrexate is acceptable in most patients who acquire COVID-19, results of a recent cohort study suggest.
Among patients on tumor necrosis factor inhibitors (TNFi) or methotrexate who developed COVID-19, death and hospitalization rates were similar to matched COVID-19 patients not on those medications, according to authors of the multicenter research network study.
Reassuringly, likelihood of hospitalization and mortality were not significantly different between 214 patients with COVID-19 taking TNFi or methotrexate and 31,862 matched COVID-19 patients not on those medications, according to the investigators, whose findings were published recently in the Journal of the American Academy of Dermatology.
Zachary Zinn, MD, corresponding author on the study, said in an interview that the findings suggest these medicines can be safely continued in the majority of patients taking them during the COVID-19 pandemic.
“If you’re a prescribing physician who’s giving patients TNF inhibitors or methotrexate or both, I think you can comfortably tell your patients there is good data that these do not lead to worse outcomes if you get COVID-19,” said Dr. Zinn, associate professor in the department of dermatology at West Virginia University, Morgantown.
The findings from these researchers corroborate a growing body of evidence suggesting that immunosuppressive treatments can be continued in patients with dermatologic and rheumatic conditions.
In recent guidance from the National Psoriasis Foundation, released Sept. 4, an expert consensus panel cited 15 studies that they said suggested that treatments for psoriasis or psoriatic arthritis “do not meaningfully alter the risk of acquiring SARS-CoV-2 infection or having worse COVID-19 outcomes.”
That said, the data to date are mainly from small case series and registry studies based on spontaneously reported COVID-19 cases, which suggests a continued need for shared decision making. In addition, chronic systemic corticosteroids should be avoided for management of psoriatic arthritis, the guidance states, based on rheumatology and gastroenterology literature suggesting this treatment is linked to worse COVID-19 outcomes.
In the interview, Dr. Zinn noted that some previous studies of immunosuppressive treatments in patients who acquire COVID-19 have aggregated data on numerous classes of biologic medications, lessening the strength of data for each specific medication.
“By focusing specifically on TNF inhibitors and methotrexate, this study gives better guidance to prescribers of these medications,” he said.
To see whether TNFi or methotrexate increased risk of worsened COVID-19 outcomes, Dr. Zinn and coinvestigators evaluated data from TriNetX, a research network that includes approximately 53 million unique patient records, predominantly in the United States.
They identified 32,076 adult patients with COVID-19, of whom 214 had recent exposure to TNFi or methotrexate. The patients in the TNFi/methotrexate group were similar in age to those without exposure to those drugs, at 55.1 versus 53.2 years, respectively. However, patients in the drug exposure group were more frequently White, female, and had substantially more comorbidities, including diabetes and obesity, according to the investigators.
Nevertheless, the likelihood of hospitalization was not statistically different in the TNFi/methotrexate group versus the non-TNFi/methotrexate group, with a risk ratio of 0.91 (95% confidence interval, 0.68-1.22; P = .5260).
Likewise, the likelihood of death was not different between groups, with a RR of 0.87 (95% CI, 0.42-1.78; P = .6958). Looking at subgroups of patients exposed to TNFi or methotrexate only didn’t change the results, the investigators added.
Taken together, the findings argue against interruption of these treatments because of the fear of the possibly worse COVID-19 outcomes, the investigators concluded, although they emphasized the need for more research.
“Because the COVID-19 pandemic is ongoing, there is a desperate need for evidence-based data on biologic and immunomodulator exposure in the setting of COVID-19 infection,” they wrote.
Dr. Zinn and coauthors reported no conflicts of interest and no funding sources related to the study.
SOURCE: Zinn Z et al. J Am Acad Dermatol. 2020 Sep 11. doi: 10.1016/j.jaad.2020.09.009.
Continued use of tumor necrosis factor inhibitors or methotrexate is acceptable in most patients who acquire COVID-19, results of a recent cohort study suggest.
Among patients on tumor necrosis factor inhibitors (TNFi) or methotrexate who developed COVID-19, death and hospitalization rates were similar to matched COVID-19 patients not on those medications, according to authors of the multicenter research network study.
Reassuringly, likelihood of hospitalization and mortality were not significantly different between 214 patients with COVID-19 taking TNFi or methotrexate and 31,862 matched COVID-19 patients not on those medications, according to the investigators, whose findings were published recently in the Journal of the American Academy of Dermatology.
Zachary Zinn, MD, corresponding author on the study, said in an interview that the findings suggest these medicines can be safely continued in the majority of patients taking them during the COVID-19 pandemic.
“If you’re a prescribing physician who’s giving patients TNF inhibitors or methotrexate or both, I think you can comfortably tell your patients there is good data that these do not lead to worse outcomes if you get COVID-19,” said Dr. Zinn, associate professor in the department of dermatology at West Virginia University, Morgantown.
The findings from these researchers corroborate a growing body of evidence suggesting that immunosuppressive treatments can be continued in patients with dermatologic and rheumatic conditions.
In recent guidance from the National Psoriasis Foundation, released Sept. 4, an expert consensus panel cited 15 studies that they said suggested that treatments for psoriasis or psoriatic arthritis “do not meaningfully alter the risk of acquiring SARS-CoV-2 infection or having worse COVID-19 outcomes.”
That said, the data to date are mainly from small case series and registry studies based on spontaneously reported COVID-19 cases, which suggests a continued need for shared decision making. In addition, chronic systemic corticosteroids should be avoided for management of psoriatic arthritis, the guidance states, based on rheumatology and gastroenterology literature suggesting this treatment is linked to worse COVID-19 outcomes.
In the interview, Dr. Zinn noted that some previous studies of immunosuppressive treatments in patients who acquire COVID-19 have aggregated data on numerous classes of biologic medications, lessening the strength of data for each specific medication.
“By focusing specifically on TNF inhibitors and methotrexate, this study gives better guidance to prescribers of these medications,” he said.
To see whether TNFi or methotrexate increased risk of worsened COVID-19 outcomes, Dr. Zinn and coinvestigators evaluated data from TriNetX, a research network that includes approximately 53 million unique patient records, predominantly in the United States.
They identified 32,076 adult patients with COVID-19, of whom 214 had recent exposure to TNFi or methotrexate. The patients in the TNFi/methotrexate group were similar in age to those without exposure to those drugs, at 55.1 versus 53.2 years, respectively. However, patients in the drug exposure group were more frequently White, female, and had substantially more comorbidities, including diabetes and obesity, according to the investigators.
Nevertheless, the likelihood of hospitalization was not statistically different in the TNFi/methotrexate group versus the non-TNFi/methotrexate group, with a risk ratio of 0.91 (95% confidence interval, 0.68-1.22; P = .5260).
Likewise, the likelihood of death was not different between groups, with a RR of 0.87 (95% CI, 0.42-1.78; P = .6958). Looking at subgroups of patients exposed to TNFi or methotrexate only didn’t change the results, the investigators added.
Taken together, the findings argue against interruption of these treatments because of the fear of the possibly worse COVID-19 outcomes, the investigators concluded, although they emphasized the need for more research.
“Because the COVID-19 pandemic is ongoing, there is a desperate need for evidence-based data on biologic and immunomodulator exposure in the setting of COVID-19 infection,” they wrote.
Dr. Zinn and coauthors reported no conflicts of interest and no funding sources related to the study.
SOURCE: Zinn Z et al. J Am Acad Dermatol. 2020 Sep 11. doi: 10.1016/j.jaad.2020.09.009.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
A woman with an asymptomatic eruption on her palms after exposure to water
This eruption can be accompanied by a mild burning or tingling sensation, which will subside with the rest of the symptoms in minutes to hours after drying.1
AWP is most frequently associated with cystic fibrosis (CF).2 It can be observed in up to 80% of CF patients and is considered a clinical sign of the disease. AWP can be present in CF carriers to a lesser extent,2,4 and has also been associated with focal hyperhidrosis, atopic dermatitis, Raynaud phenomenon, and COX-2 inhibitor use.5
While a definitive cause is unknown, it is thought that AWP is caused by dysregulation of sweat glands in the palms through increased expression of aquaporin, a protein crucial in the transport of water between cells.3
AWP is quite rare and benign in nature. However, because of its strong association with CF, genetic screening should be considered in asymptomatic patients. Our patient had been screened in the past and is not a CF carrier. Often, the itching or burning associated with CF is mild and easily controlled. The patient was placed on low dose isotretinoin for treatment of her acne. Interestingly, the patient claimed her eruption no longer appeared after starting isotretinoin therapy. To our knowledge, this is the first reported case of AWP resolving with isotretinoin use.
This case and photo were submitted by Mr. Birk, University of Texas, Austin, Texas; and Dr. Mamelak, Sanova Dermatology, in Austin. Donna Bilu Martin, MD, edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at MDedge.com/Dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
1. Katz M, Ramot Y. CMAJ. 2015 Dec 8;187(18):E515.
2. Tolland JP et al. Dermatology. 2010;221(4):326-30.
3. Kabashima K et al. J Am Acad Dermatol. 2008 Aug;59(2 Suppl 1):S28-32.
4. Gild R et al. Br J Dermatol. 2010 Nov;163(5):1082-4.
5. Glatz M and Muellegger RR. BMJ Case Rep. 2014. doi: 10.1136/bcr-2014-203929.
This eruption can be accompanied by a mild burning or tingling sensation, which will subside with the rest of the symptoms in minutes to hours after drying.1
AWP is most frequently associated with cystic fibrosis (CF).2 It can be observed in up to 80% of CF patients and is considered a clinical sign of the disease. AWP can be present in CF carriers to a lesser extent,2,4 and has also been associated with focal hyperhidrosis, atopic dermatitis, Raynaud phenomenon, and COX-2 inhibitor use.5
While a definitive cause is unknown, it is thought that AWP is caused by dysregulation of sweat glands in the palms through increased expression of aquaporin, a protein crucial in the transport of water between cells.3
AWP is quite rare and benign in nature. However, because of its strong association with CF, genetic screening should be considered in asymptomatic patients. Our patient had been screened in the past and is not a CF carrier. Often, the itching or burning associated with CF is mild and easily controlled. The patient was placed on low dose isotretinoin for treatment of her acne. Interestingly, the patient claimed her eruption no longer appeared after starting isotretinoin therapy. To our knowledge, this is the first reported case of AWP resolving with isotretinoin use.
This case and photo were submitted by Mr. Birk, University of Texas, Austin, Texas; and Dr. Mamelak, Sanova Dermatology, in Austin. Donna Bilu Martin, MD, edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at MDedge.com/Dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
1. Katz M, Ramot Y. CMAJ. 2015 Dec 8;187(18):E515.
2. Tolland JP et al. Dermatology. 2010;221(4):326-30.
3. Kabashima K et al. J Am Acad Dermatol. 2008 Aug;59(2 Suppl 1):S28-32.
4. Gild R et al. Br J Dermatol. 2010 Nov;163(5):1082-4.
5. Glatz M and Muellegger RR. BMJ Case Rep. 2014. doi: 10.1136/bcr-2014-203929.
This eruption can be accompanied by a mild burning or tingling sensation, which will subside with the rest of the symptoms in minutes to hours after drying.1
AWP is most frequently associated with cystic fibrosis (CF).2 It can be observed in up to 80% of CF patients and is considered a clinical sign of the disease. AWP can be present in CF carriers to a lesser extent,2,4 and has also been associated with focal hyperhidrosis, atopic dermatitis, Raynaud phenomenon, and COX-2 inhibitor use.5
While a definitive cause is unknown, it is thought that AWP is caused by dysregulation of sweat glands in the palms through increased expression of aquaporin, a protein crucial in the transport of water between cells.3
AWP is quite rare and benign in nature. However, because of its strong association with CF, genetic screening should be considered in asymptomatic patients. Our patient had been screened in the past and is not a CF carrier. Often, the itching or burning associated with CF is mild and easily controlled. The patient was placed on low dose isotretinoin for treatment of her acne. Interestingly, the patient claimed her eruption no longer appeared after starting isotretinoin therapy. To our knowledge, this is the first reported case of AWP resolving with isotretinoin use.
This case and photo were submitted by Mr. Birk, University of Texas, Austin, Texas; and Dr. Mamelak, Sanova Dermatology, in Austin. Donna Bilu Martin, MD, edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at MDedge.com/Dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
1. Katz M, Ramot Y. CMAJ. 2015 Dec 8;187(18):E515.
2. Tolland JP et al. Dermatology. 2010;221(4):326-30.
3. Kabashima K et al. J Am Acad Dermatol. 2008 Aug;59(2 Suppl 1):S28-32.
4. Gild R et al. Br J Dermatol. 2010 Nov;163(5):1082-4.
5. Glatz M and Muellegger RR. BMJ Case Rep. 2014. doi: 10.1136/bcr-2014-203929.
Rash, muscle weakness, and confusion
The constellation of symptoms was suggestive of Lyme disease, although connective tissue disease and syphilis were also considered. Two punch biopsies were performed in the office, and erythrocyte sedimentation rate (ESR), complete blood cell count (CBC), international normalized ratio (INR), comprehensive metabolic panel (CMP), Lyme enzyme-linked immunosorbent assay (ELISA) antibody panel, and rapid plasma reagin (RPR) laboratory tests were ordered.
Immediately available laboratory results included ESR, CBC, INR, and CMP. Findings were notable for elevated INR, as well as elevated alanine aminotransferase and aspartate transaminase. The transaminitis suggested myopathy and was consistent with clinical muscle weakness. RPR testing was negative.
Because of the confusion, severity of muscle weakness, and plausibility of early encephalopathy with Lyme disease, the patient was admitted to the hospital for further work-up. Lumbar puncture was delayed until his INR was reduced, but subsequently was found to be normal. He received intravenous (IV) ceftriaxone (2 g/d) empirically for possible early disseminated disease with neurologic complications. His confusion, muscle weakness, and transaminitis rapidly improved.
His Lyme antibody panel was positive for IgM after his third day of hospitalization. A reflexive confirmatory western blot for IgG was not positive on the initial set of labs but was positive when redrawn 4 weeks after this hospitalization, confirming Lyme disease.
Lyme disease is a vector-borne disease caused by the Borrelia genus of spirochete bacteria, most commonly Borrelia burgdorferi in North America. Transmission occurs through prolonged (typically 36-48 hours) attachment of a blacklegged tick.
The disease can be divided into 3 stages:
- localized (3-30 days): erythema migrans rash and flulike illness
- early disseminated (days to weeks; seen in this patient): multiple erythema migrans rashes, early neuroborreliosis, arthritis, carditis, and rarely hepatitis and uveitis
- late disseminated (months to years): chronic Lyme arthritis, chronic neurological disorders (eg, encephalopathy, radicular pain, and chronic neuropathy).
The initial erythema migrans rash is classically red and targetoid; it expands from the site of attachment. Early disseminated patches tend to be smaller and can occur on any body part. The rash is rarely itchy or painful but may be warm to the touch or sensitive. The rash resolves spontaneously within 3 to 4 weeks of onset.
Treatment of all early and early disseminated Lyme disease typically involves a 14- to 28-day course of doxycycline (100 mg bid for adults, 2.2 mg/kg bid [maximum 100 mg bid] for children). Patients with acute neurologic disease often can be treated with doxycycline, but patients who cannot tolerate doxycycline and those with parenchymal disease such as encephalitis should receive IV therapy with ceftriaxone 2 g/d.
In this case, the patient was discharged home on a 3-week course of doxycycline 100 mg bid and cleared without further symptoms.
Text courtesy of Tristan Reynolds, DO, Maine Dartmouth Family Medicine Residency, and Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
Lyme disease. Centers for Disease Control and Prevention Web site. https://www.cdc.gov/lyme/healthcare/index.html. Accessed September 1, 2020.

The constellation of symptoms was suggestive of Lyme disease, although connective tissue disease and syphilis were also considered. Two punch biopsies were performed in the office, and erythrocyte sedimentation rate (ESR), complete blood cell count (CBC), international normalized ratio (INR), comprehensive metabolic panel (CMP), Lyme enzyme-linked immunosorbent assay (ELISA) antibody panel, and rapid plasma reagin (RPR) laboratory tests were ordered.
Immediately available laboratory results included ESR, CBC, INR, and CMP. Findings were notable for elevated INR, as well as elevated alanine aminotransferase and aspartate transaminase. The transaminitis suggested myopathy and was consistent with clinical muscle weakness. RPR testing was negative.
Because of the confusion, severity of muscle weakness, and plausibility of early encephalopathy with Lyme disease, the patient was admitted to the hospital for further work-up. Lumbar puncture was delayed until his INR was reduced, but subsequently was found to be normal. He received intravenous (IV) ceftriaxone (2 g/d) empirically for possible early disseminated disease with neurologic complications. His confusion, muscle weakness, and transaminitis rapidly improved.
His Lyme antibody panel was positive for IgM after his third day of hospitalization. A reflexive confirmatory western blot for IgG was not positive on the initial set of labs but was positive when redrawn 4 weeks after this hospitalization, confirming Lyme disease.
Lyme disease is a vector-borne disease caused by the Borrelia genus of spirochete bacteria, most commonly Borrelia burgdorferi in North America. Transmission occurs through prolonged (typically 36-48 hours) attachment of a blacklegged tick.
The disease can be divided into 3 stages:
- localized (3-30 days): erythema migrans rash and flulike illness
- early disseminated (days to weeks; seen in this patient): multiple erythema migrans rashes, early neuroborreliosis, arthritis, carditis, and rarely hepatitis and uveitis
- late disseminated (months to years): chronic Lyme arthritis, chronic neurological disorders (eg, encephalopathy, radicular pain, and chronic neuropathy).
The initial erythema migrans rash is classically red and targetoid; it expands from the site of attachment. Early disseminated patches tend to be smaller and can occur on any body part. The rash is rarely itchy or painful but may be warm to the touch or sensitive. The rash resolves spontaneously within 3 to 4 weeks of onset.
Treatment of all early and early disseminated Lyme disease typically involves a 14- to 28-day course of doxycycline (100 mg bid for adults, 2.2 mg/kg bid [maximum 100 mg bid] for children). Patients with acute neurologic disease often can be treated with doxycycline, but patients who cannot tolerate doxycycline and those with parenchymal disease such as encephalitis should receive IV therapy with ceftriaxone 2 g/d.
In this case, the patient was discharged home on a 3-week course of doxycycline 100 mg bid and cleared without further symptoms.
Text courtesy of Tristan Reynolds, DO, Maine Dartmouth Family Medicine Residency, and Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).

The constellation of symptoms was suggestive of Lyme disease, although connective tissue disease and syphilis were also considered. Two punch biopsies were performed in the office, and erythrocyte sedimentation rate (ESR), complete blood cell count (CBC), international normalized ratio (INR), comprehensive metabolic panel (CMP), Lyme enzyme-linked immunosorbent assay (ELISA) antibody panel, and rapid plasma reagin (RPR) laboratory tests were ordered.
Immediately available laboratory results included ESR, CBC, INR, and CMP. Findings were notable for elevated INR, as well as elevated alanine aminotransferase and aspartate transaminase. The transaminitis suggested myopathy and was consistent with clinical muscle weakness. RPR testing was negative.
Because of the confusion, severity of muscle weakness, and plausibility of early encephalopathy with Lyme disease, the patient was admitted to the hospital for further work-up. Lumbar puncture was delayed until his INR was reduced, but subsequently was found to be normal. He received intravenous (IV) ceftriaxone (2 g/d) empirically for possible early disseminated disease with neurologic complications. His confusion, muscle weakness, and transaminitis rapidly improved.
His Lyme antibody panel was positive for IgM after his third day of hospitalization. A reflexive confirmatory western blot for IgG was not positive on the initial set of labs but was positive when redrawn 4 weeks after this hospitalization, confirming Lyme disease.
Lyme disease is a vector-borne disease caused by the Borrelia genus of spirochete bacteria, most commonly Borrelia burgdorferi in North America. Transmission occurs through prolonged (typically 36-48 hours) attachment of a blacklegged tick.
The disease can be divided into 3 stages:
- localized (3-30 days): erythema migrans rash and flulike illness
- early disseminated (days to weeks; seen in this patient): multiple erythema migrans rashes, early neuroborreliosis, arthritis, carditis, and rarely hepatitis and uveitis
- late disseminated (months to years): chronic Lyme arthritis, chronic neurological disorders (eg, encephalopathy, radicular pain, and chronic neuropathy).
The initial erythema migrans rash is classically red and targetoid; it expands from the site of attachment. Early disseminated patches tend to be smaller and can occur on any body part. The rash is rarely itchy or painful but may be warm to the touch or sensitive. The rash resolves spontaneously within 3 to 4 weeks of onset.
Treatment of all early and early disseminated Lyme disease typically involves a 14- to 28-day course of doxycycline (100 mg bid for adults, 2.2 mg/kg bid [maximum 100 mg bid] for children). Patients with acute neurologic disease often can be treated with doxycycline, but patients who cannot tolerate doxycycline and those with parenchymal disease such as encephalitis should receive IV therapy with ceftriaxone 2 g/d.
In this case, the patient was discharged home on a 3-week course of doxycycline 100 mg bid and cleared without further symptoms.
Text courtesy of Tristan Reynolds, DO, Maine Dartmouth Family Medicine Residency, and Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
Lyme disease. Centers for Disease Control and Prevention Web site. https://www.cdc.gov/lyme/healthcare/index.html. Accessed September 1, 2020.
Lyme disease. Centers for Disease Control and Prevention Web site. https://www.cdc.gov/lyme/healthcare/index.html. Accessed September 1, 2020.