User login
Birch bark derivative gel found effective for EB, in phase 3 study
A . The results come from the largest double-blind, randomized trial performed in this patient population.
More than 41% of EB target wounds that were treated with Oleogel-S10 healed within 45 days, compared with about 29% of target wounds treated with placebo, in the EASE phase 3 trial, conducted at 58 sites in 28 countries.
A group of rare genetic disorders, EB “is described as the worst disease you’ve never heard of,” explained lead investigator Dedee Murrell, MD, director of dermatology, St. George Hospital at the University of New South Wales, Sydney. “It starts in children and is like having burns that heal with scars, and no treatment has been approved for it” by the Food and Drug Administration.
“This is the first large clinical trial with placebo of a topical treatment that’s worked for this terrible disease,” Dr. Murrell said in an interview. She noted that standard EB treatment currently consists of applying nonstick dressings to wounds to protect skin from trauma and infection.
Dr. Murrell, who has focused her work on EB patients since 1990, presented the findings at the virtual annual Congress of the European Academy of Dermatology and Venereology.
The trial enrolled 223 patients (average age, 12 years, but ages ranged to 81 years) with three types of EB, including dystrophic and junctional EB and Kindler syndrome. For each participant, a target wound was selected for use as the primary efficacy endpoint. Those wounds had a partial thickness of between 10 cm2 and 50 cm2 and lasted between 21 days and 9 months. Patients were stratified into groups depending on type of EB and size of target wound.
Participants were randomly assigned to receive either Oleogel-S10 (n = 109) or placebo (n = 114). All applied the blinded-study gel to all their wounds at least every 4 days at the time dressings were changed.
The primary endpoint was the percentage of patients whose target wounds completely closed within 45 days. Key secondary endpoints included time to wound healing and percentage of target wounds that healed within 90 days of treatment; incidence and severity of target wound infection; change in total body wound burden, as measured by the Epidermolysis Bullosa Disease Activity and Scarring Index skin activity subscore; change in itching, as measured by the Itch Man Scale and the Leuven Itch Scale; and adverse events.
Nearly 92% of patients who were treated with Oleogel-S10 completed the double-blind phase of the trial, compared with nearly 87% who received placebo. As noted, the primary endpoint was met, with 41.3% of Oleogel-S10 patients achieving target wound closure within 45 days, compared with 28.9% of the patients who received placebo (P = .013).
But the difference in time to wound healing by day 90 between the two patient groups was not statistically significant (P = .302), with 50.5% of Oleogel-S10 patients achieving wound closure vs. 43.9% of control patients.
Target-wound infection occurred in eight participants, including three who used Oleogel-S10 and five who received placebo; all moderate or severe infections occurred in patients who received placebo. Total wound burden was reduced to a greater extent among Oleogel-S10 patients by day 60, but there was no apparent difference at day 90.
Both treatment groups reported qualitative improvements in itch, with no significant differences between groups. The prevalence of adverse events was also similar between groups (Oleogel-S10, 81.7%; placebo, 80.7%). The most frequently reported adverse events among Oleogel-S10 patients, compared with patients who received placebo, were wound complications, pyrexia, wound infection, pruritus, and anemia; only 4.5% of adverse events were deemed severe.
Dr. Murrell said that, on the basis of the trial results, she expects the FDA to fast-track approval of Oleogel-S10, which contains triterpene extract and sunflower oil.
The gel is “a treatment patients will be able to put under their dressings, added to normal treatment, which will accelerate their wound healing, with no significant increase in any side effects,” she added.
Jemima Mellerio, MD, of St. Thomas’ Hospital in London who sees about 400 EB patients each year, agreed with Dr. Murrell that the results are “very exciting.” Dr. Mellerio was not involved in the study.
“Practicing dermatologists seeing people with EB will have something to offer that appears to speed up wound healing in chronic wounds,” Dr. Mellerio said in an interview. “It’s a positive option rather than just supportive treatment, something that makes a difference to the natural history of wounds.”
She said the trial’s biggest strength was including “such a large cohort of patients.
“It’s extremely difficult to do that kind of study, especially with a placebo-controlled arm and especially in a rare disease,” Dr. Mellerio said. “If you think about the product itself, it’s easy to apply, so it’s not particularly onerous for people to add to their daily regimen of dressings.”
The study was funded by Amryt Pharma. Dr. Murrell is an advisory board member for Amryt Pharma. Dr. Mellerio is a consultant for Amryt Pharma.
A version of this article originally appeared on Medscape.com.
A . The results come from the largest double-blind, randomized trial performed in this patient population.
More than 41% of EB target wounds that were treated with Oleogel-S10 healed within 45 days, compared with about 29% of target wounds treated with placebo, in the EASE phase 3 trial, conducted at 58 sites in 28 countries.
A group of rare genetic disorders, EB “is described as the worst disease you’ve never heard of,” explained lead investigator Dedee Murrell, MD, director of dermatology, St. George Hospital at the University of New South Wales, Sydney. “It starts in children and is like having burns that heal with scars, and no treatment has been approved for it” by the Food and Drug Administration.
“This is the first large clinical trial with placebo of a topical treatment that’s worked for this terrible disease,” Dr. Murrell said in an interview. She noted that standard EB treatment currently consists of applying nonstick dressings to wounds to protect skin from trauma and infection.
Dr. Murrell, who has focused her work on EB patients since 1990, presented the findings at the virtual annual Congress of the European Academy of Dermatology and Venereology.
The trial enrolled 223 patients (average age, 12 years, but ages ranged to 81 years) with three types of EB, including dystrophic and junctional EB and Kindler syndrome. For each participant, a target wound was selected for use as the primary efficacy endpoint. Those wounds had a partial thickness of between 10 cm2 and 50 cm2 and lasted between 21 days and 9 months. Patients were stratified into groups depending on type of EB and size of target wound.
Participants were randomly assigned to receive either Oleogel-S10 (n = 109) or placebo (n = 114). All applied the blinded-study gel to all their wounds at least every 4 days at the time dressings were changed.
The primary endpoint was the percentage of patients whose target wounds completely closed within 45 days. Key secondary endpoints included time to wound healing and percentage of target wounds that healed within 90 days of treatment; incidence and severity of target wound infection; change in total body wound burden, as measured by the Epidermolysis Bullosa Disease Activity and Scarring Index skin activity subscore; change in itching, as measured by the Itch Man Scale and the Leuven Itch Scale; and adverse events.
Nearly 92% of patients who were treated with Oleogel-S10 completed the double-blind phase of the trial, compared with nearly 87% who received placebo. As noted, the primary endpoint was met, with 41.3% of Oleogel-S10 patients achieving target wound closure within 45 days, compared with 28.9% of the patients who received placebo (P = .013).
But the difference in time to wound healing by day 90 between the two patient groups was not statistically significant (P = .302), with 50.5% of Oleogel-S10 patients achieving wound closure vs. 43.9% of control patients.
Target-wound infection occurred in eight participants, including three who used Oleogel-S10 and five who received placebo; all moderate or severe infections occurred in patients who received placebo. Total wound burden was reduced to a greater extent among Oleogel-S10 patients by day 60, but there was no apparent difference at day 90.
Both treatment groups reported qualitative improvements in itch, with no significant differences between groups. The prevalence of adverse events was also similar between groups (Oleogel-S10, 81.7%; placebo, 80.7%). The most frequently reported adverse events among Oleogel-S10 patients, compared with patients who received placebo, were wound complications, pyrexia, wound infection, pruritus, and anemia; only 4.5% of adverse events were deemed severe.
Dr. Murrell said that, on the basis of the trial results, she expects the FDA to fast-track approval of Oleogel-S10, which contains triterpene extract and sunflower oil.
The gel is “a treatment patients will be able to put under their dressings, added to normal treatment, which will accelerate their wound healing, with no significant increase in any side effects,” she added.
Jemima Mellerio, MD, of St. Thomas’ Hospital in London who sees about 400 EB patients each year, agreed with Dr. Murrell that the results are “very exciting.” Dr. Mellerio was not involved in the study.
“Practicing dermatologists seeing people with EB will have something to offer that appears to speed up wound healing in chronic wounds,” Dr. Mellerio said in an interview. “It’s a positive option rather than just supportive treatment, something that makes a difference to the natural history of wounds.”
She said the trial’s biggest strength was including “such a large cohort of patients.
“It’s extremely difficult to do that kind of study, especially with a placebo-controlled arm and especially in a rare disease,” Dr. Mellerio said. “If you think about the product itself, it’s easy to apply, so it’s not particularly onerous for people to add to their daily regimen of dressings.”
The study was funded by Amryt Pharma. Dr. Murrell is an advisory board member for Amryt Pharma. Dr. Mellerio is a consultant for Amryt Pharma.
A version of this article originally appeared on Medscape.com.
A . The results come from the largest double-blind, randomized trial performed in this patient population.
More than 41% of EB target wounds that were treated with Oleogel-S10 healed within 45 days, compared with about 29% of target wounds treated with placebo, in the EASE phase 3 trial, conducted at 58 sites in 28 countries.
A group of rare genetic disorders, EB “is described as the worst disease you’ve never heard of,” explained lead investigator Dedee Murrell, MD, director of dermatology, St. George Hospital at the University of New South Wales, Sydney. “It starts in children and is like having burns that heal with scars, and no treatment has been approved for it” by the Food and Drug Administration.
“This is the first large clinical trial with placebo of a topical treatment that’s worked for this terrible disease,” Dr. Murrell said in an interview. She noted that standard EB treatment currently consists of applying nonstick dressings to wounds to protect skin from trauma and infection.
Dr. Murrell, who has focused her work on EB patients since 1990, presented the findings at the virtual annual Congress of the European Academy of Dermatology and Venereology.
The trial enrolled 223 patients (average age, 12 years, but ages ranged to 81 years) with three types of EB, including dystrophic and junctional EB and Kindler syndrome. For each participant, a target wound was selected for use as the primary efficacy endpoint. Those wounds had a partial thickness of between 10 cm2 and 50 cm2 and lasted between 21 days and 9 months. Patients were stratified into groups depending on type of EB and size of target wound.
Participants were randomly assigned to receive either Oleogel-S10 (n = 109) or placebo (n = 114). All applied the blinded-study gel to all their wounds at least every 4 days at the time dressings were changed.
The primary endpoint was the percentage of patients whose target wounds completely closed within 45 days. Key secondary endpoints included time to wound healing and percentage of target wounds that healed within 90 days of treatment; incidence and severity of target wound infection; change in total body wound burden, as measured by the Epidermolysis Bullosa Disease Activity and Scarring Index skin activity subscore; change in itching, as measured by the Itch Man Scale and the Leuven Itch Scale; and adverse events.
Nearly 92% of patients who were treated with Oleogel-S10 completed the double-blind phase of the trial, compared with nearly 87% who received placebo. As noted, the primary endpoint was met, with 41.3% of Oleogel-S10 patients achieving target wound closure within 45 days, compared with 28.9% of the patients who received placebo (P = .013).
But the difference in time to wound healing by day 90 between the two patient groups was not statistically significant (P = .302), with 50.5% of Oleogel-S10 patients achieving wound closure vs. 43.9% of control patients.
Target-wound infection occurred in eight participants, including three who used Oleogel-S10 and five who received placebo; all moderate or severe infections occurred in patients who received placebo. Total wound burden was reduced to a greater extent among Oleogel-S10 patients by day 60, but there was no apparent difference at day 90.
Both treatment groups reported qualitative improvements in itch, with no significant differences between groups. The prevalence of adverse events was also similar between groups (Oleogel-S10, 81.7%; placebo, 80.7%). The most frequently reported adverse events among Oleogel-S10 patients, compared with patients who received placebo, were wound complications, pyrexia, wound infection, pruritus, and anemia; only 4.5% of adverse events were deemed severe.
Dr. Murrell said that, on the basis of the trial results, she expects the FDA to fast-track approval of Oleogel-S10, which contains triterpene extract and sunflower oil.
The gel is “a treatment patients will be able to put under their dressings, added to normal treatment, which will accelerate their wound healing, with no significant increase in any side effects,” she added.
Jemima Mellerio, MD, of St. Thomas’ Hospital in London who sees about 400 EB patients each year, agreed with Dr. Murrell that the results are “very exciting.” Dr. Mellerio was not involved in the study.
“Practicing dermatologists seeing people with EB will have something to offer that appears to speed up wound healing in chronic wounds,” Dr. Mellerio said in an interview. “It’s a positive option rather than just supportive treatment, something that makes a difference to the natural history of wounds.”
She said the trial’s biggest strength was including “such a large cohort of patients.
“It’s extremely difficult to do that kind of study, especially with a placebo-controlled arm and especially in a rare disease,” Dr. Mellerio said. “If you think about the product itself, it’s easy to apply, so it’s not particularly onerous for people to add to their daily regimen of dressings.”
The study was funded by Amryt Pharma. Dr. Murrell is an advisory board member for Amryt Pharma. Dr. Mellerio is a consultant for Amryt Pharma.
A version of this article originally appeared on Medscape.com.
Biologics may protect psoriasis patients against severe COVID-19
presented at the virtual annual congress of the European Academy of Dermatology and Venereology.
“Biologics seem to be very protective against severe, poor-prognosis COVID-19, but they do not prevent infection with the virus,” reported Giovanni Damiani, MD, a dermatologist at the University of Milan.
This apparent protective effect of biologic agents against severe and even fatal COVID-19 is all the more impressive because the psoriasis patients included in the Italian study – as is true of those elsewhere throughout the world – had relatively high rates of obesity, smoking, and chronic obstructive pulmonary disease, known risk factors for severe COVID-19, he added.
He presented a case-control study including 1,193 adult psoriasis patients on biologics or apremilast (Otezla) at Milan’s San Donato Hospital during the period from Feb. 21 to April 9, 2020. The control group comprised more than 10 million individuals, the entire adult population of the Lombardy region, of which Milan is the capital. This was the hardest-hit area in all of Italy during the first wave of COVID-19.
Twenty-two of the 1,193 psoriasis patients experienced confirmed COVID-19 during the study period. Seventeen were quarantined at home because their disease was mild. Five were hospitalized. But no psoriasis patients were placed in intensive care, and none died.
Psoriasis patients on biologics were significantly more likely than the general Lombardian population to test positive for COVID-19, with an unadjusted odds ratio of 3.43. They were at 9.05-fold increased risk of home quarantine for mild disease, and at 3.59-fold greater risk than controls for hospitalization for COVID-19. However, they were not at significantly increased risk of ICU admission. And while they actually had a 59% relative risk reduction for death, this didn’t achieve statistical significance.
Forty-five percent of the psoriasis patients were on an interleukin-17 (IL-17) inhibitor, 22% were on a tumor necrosis factor–alpha inhibitor, and 20% were taking an IL-12/23 inhibitor. Of note, none of 77 patients on apremilast developed COVID-19, even though it is widely considered a less potent psoriasis therapy than the injectable monoclonal antibody biologics.
The French experience
Anne-Claire Fougerousse, MD, and her French coinvestigators conducted a study designed to address a different question: Is it safe to start psoriasis patients on biologics or older conventional systemic agents such as methotrexate during the pandemic?
She presented a French national cross-sectional study of 1,418 adult psoriasis patients on a biologic or standard systemic therapy during a snapshot in time near the peak of the first wave of the pandemic in France: the period from April 27 to May 7, 2020. The group included 1,188 psoriasis patients on maintenance therapy and 230 who had initiated systemic treatment within the past 4 months. More than one-third of the patients had at least one risk factor for severe COVID-19.
Although testing wasn’t available to confirm all cases, 54 patients developed probable COVID-19 during the study period. Only five required hospitalization. None died. The two hospitalized psoriasis patients admitted to an ICU had obesity as a risk factor for severe COVID-19, as did another of the five hospitalized patients, reported Dr. Fougerousse, a dermatologist at the Bégin Military Teaching Hospital in Saint-Mandé, France. Hospitalization for COVID-19 was required in 0.43% of the French treatment initiators, not significantly different from the 0.34% rate in patients on maintenance systemic therapy. A study limitation was the lack of a control group.
Nonetheless, the data did answer the investigators’ main question: “This is the first data showing no increased incidence of severe COVID-19 in psoriasis patients receiving systemic therapy in the treatment initiation period compared to those on maintenance therapy. This may now allow physicians to initiate conventional systemic or biologic therapy in patients with severe psoriasis on a case-by-case basis in the context of the persistent COVID-19 pandemic,” Dr. Fougerousse concluded.
Proposed mechanism of benefit
The Italian study findings that biologics boost the risk of infection with the SARS-CoV-2 virus in psoriasis patients while potentially protecting them against ICU admission and death are backed by a biologically plausible albeit as yet unproven mechanism of action, Dr. Damiani asserted.
He elaborated: A vast body of high-quality clinical trials data demonstrates that these targeted immunosuppressive agents are associated with modestly increased risk of viral infections, including both skin and respiratory tract infections. So there is no reason to suppose these agents would offer protection against the first phase of COVID-19, involving SARS-CoV-2 infection, nor protect against the second (pulmonary phase), whose hallmarks are dyspnea with or without hypoxia. But progression to the third phase, involving hyperinflammation and hypercoagulation – dubbed the cytokine storm – could be a different matter.
“Of particular interest was that our patients on IL-17 inhibitors displayed a really great outcome. Interleukin-17 has procoagulant and prothrombotic effects, organizes bronchoalveolar remodeling, has a profibrotic effect, induces mitochondrial dysfunction, and encourages dendritic cell migration in peribronchial lymph nodes. Therefore, by antagonizing this interleukin, we may have a better prognosis, although further studies are needed to be certain,” Dr. Damiani commented.
Publication of his preliminary findings drew the attention of a group of highly respected thought leaders in psoriasis, including James G. Krueger, MD, head of the laboratory for investigative dermatology and codirector of the center for clinical and investigative science at Rockefeller University, New York.
The Italian report prompted them to analyze data from the phase 4, double-blind, randomized ObePso-S study investigating the effects of the IL-17 inhibitor secukinumab (Cosentyx) on systemic inflammatory markers and gene expression in psoriasis patients. The investigators demonstrated that IL-17–mediated inflammation in psoriasis patients was associated with increased expression of the angiotensin-converting enzyme 2 (ACE2) receptor in lesional skin, and that treatment with secukinumab dropped ACE2 expression to levels seen in nonlesional skin. Given that ACE2 is the chief portal of entry for SARS-CoV-2 and that IL-17 exerts systemic proinflammatory effects, it’s plausible that inhibition of IL-17–mediated inflammation via dampening of ACE2 expression in noncutaneous epithelia “could prove to be advantageous in patients with psoriasis who are at risk for SARS-CoV-2 infection,” according to Dr. Krueger and his coinvestigators in the Journal of Allergy and Clinical Immunology.
Dr. Damiani and Dr. Fougerousse reported having no financial conflicts regarding their studies. The secukinumab/ACE2 receptor study was funded by Novartis.
presented at the virtual annual congress of the European Academy of Dermatology and Venereology.
“Biologics seem to be very protective against severe, poor-prognosis COVID-19, but they do not prevent infection with the virus,” reported Giovanni Damiani, MD, a dermatologist at the University of Milan.
This apparent protective effect of biologic agents against severe and even fatal COVID-19 is all the more impressive because the psoriasis patients included in the Italian study – as is true of those elsewhere throughout the world – had relatively high rates of obesity, smoking, and chronic obstructive pulmonary disease, known risk factors for severe COVID-19, he added.
He presented a case-control study including 1,193 adult psoriasis patients on biologics or apremilast (Otezla) at Milan’s San Donato Hospital during the period from Feb. 21 to April 9, 2020. The control group comprised more than 10 million individuals, the entire adult population of the Lombardy region, of which Milan is the capital. This was the hardest-hit area in all of Italy during the first wave of COVID-19.
Twenty-two of the 1,193 psoriasis patients experienced confirmed COVID-19 during the study period. Seventeen were quarantined at home because their disease was mild. Five were hospitalized. But no psoriasis patients were placed in intensive care, and none died.
Psoriasis patients on biologics were significantly more likely than the general Lombardian population to test positive for COVID-19, with an unadjusted odds ratio of 3.43. They were at 9.05-fold increased risk of home quarantine for mild disease, and at 3.59-fold greater risk than controls for hospitalization for COVID-19. However, they were not at significantly increased risk of ICU admission. And while they actually had a 59% relative risk reduction for death, this didn’t achieve statistical significance.
Forty-five percent of the psoriasis patients were on an interleukin-17 (IL-17) inhibitor, 22% were on a tumor necrosis factor–alpha inhibitor, and 20% were taking an IL-12/23 inhibitor. Of note, none of 77 patients on apremilast developed COVID-19, even though it is widely considered a less potent psoriasis therapy than the injectable monoclonal antibody biologics.
The French experience
Anne-Claire Fougerousse, MD, and her French coinvestigators conducted a study designed to address a different question: Is it safe to start psoriasis patients on biologics or older conventional systemic agents such as methotrexate during the pandemic?
She presented a French national cross-sectional study of 1,418 adult psoriasis patients on a biologic or standard systemic therapy during a snapshot in time near the peak of the first wave of the pandemic in France: the period from April 27 to May 7, 2020. The group included 1,188 psoriasis patients on maintenance therapy and 230 who had initiated systemic treatment within the past 4 months. More than one-third of the patients had at least one risk factor for severe COVID-19.
Although testing wasn’t available to confirm all cases, 54 patients developed probable COVID-19 during the study period. Only five required hospitalization. None died. The two hospitalized psoriasis patients admitted to an ICU had obesity as a risk factor for severe COVID-19, as did another of the five hospitalized patients, reported Dr. Fougerousse, a dermatologist at the Bégin Military Teaching Hospital in Saint-Mandé, France. Hospitalization for COVID-19 was required in 0.43% of the French treatment initiators, not significantly different from the 0.34% rate in patients on maintenance systemic therapy. A study limitation was the lack of a control group.
Nonetheless, the data did answer the investigators’ main question: “This is the first data showing no increased incidence of severe COVID-19 in psoriasis patients receiving systemic therapy in the treatment initiation period compared to those on maintenance therapy. This may now allow physicians to initiate conventional systemic or biologic therapy in patients with severe psoriasis on a case-by-case basis in the context of the persistent COVID-19 pandemic,” Dr. Fougerousse concluded.
Proposed mechanism of benefit
The Italian study findings that biologics boost the risk of infection with the SARS-CoV-2 virus in psoriasis patients while potentially protecting them against ICU admission and death are backed by a biologically plausible albeit as yet unproven mechanism of action, Dr. Damiani asserted.
He elaborated: A vast body of high-quality clinical trials data demonstrates that these targeted immunosuppressive agents are associated with modestly increased risk of viral infections, including both skin and respiratory tract infections. So there is no reason to suppose these agents would offer protection against the first phase of COVID-19, involving SARS-CoV-2 infection, nor protect against the second (pulmonary phase), whose hallmarks are dyspnea with or without hypoxia. But progression to the third phase, involving hyperinflammation and hypercoagulation – dubbed the cytokine storm – could be a different matter.
“Of particular interest was that our patients on IL-17 inhibitors displayed a really great outcome. Interleukin-17 has procoagulant and prothrombotic effects, organizes bronchoalveolar remodeling, has a profibrotic effect, induces mitochondrial dysfunction, and encourages dendritic cell migration in peribronchial lymph nodes. Therefore, by antagonizing this interleukin, we may have a better prognosis, although further studies are needed to be certain,” Dr. Damiani commented.
Publication of his preliminary findings drew the attention of a group of highly respected thought leaders in psoriasis, including James G. Krueger, MD, head of the laboratory for investigative dermatology and codirector of the center for clinical and investigative science at Rockefeller University, New York.
The Italian report prompted them to analyze data from the phase 4, double-blind, randomized ObePso-S study investigating the effects of the IL-17 inhibitor secukinumab (Cosentyx) on systemic inflammatory markers and gene expression in psoriasis patients. The investigators demonstrated that IL-17–mediated inflammation in psoriasis patients was associated with increased expression of the angiotensin-converting enzyme 2 (ACE2) receptor in lesional skin, and that treatment with secukinumab dropped ACE2 expression to levels seen in nonlesional skin. Given that ACE2 is the chief portal of entry for SARS-CoV-2 and that IL-17 exerts systemic proinflammatory effects, it’s plausible that inhibition of IL-17–mediated inflammation via dampening of ACE2 expression in noncutaneous epithelia “could prove to be advantageous in patients with psoriasis who are at risk for SARS-CoV-2 infection,” according to Dr. Krueger and his coinvestigators in the Journal of Allergy and Clinical Immunology.
Dr. Damiani and Dr. Fougerousse reported having no financial conflicts regarding their studies. The secukinumab/ACE2 receptor study was funded by Novartis.
presented at the virtual annual congress of the European Academy of Dermatology and Venereology.
“Biologics seem to be very protective against severe, poor-prognosis COVID-19, but they do not prevent infection with the virus,” reported Giovanni Damiani, MD, a dermatologist at the University of Milan.
This apparent protective effect of biologic agents against severe and even fatal COVID-19 is all the more impressive because the psoriasis patients included in the Italian study – as is true of those elsewhere throughout the world – had relatively high rates of obesity, smoking, and chronic obstructive pulmonary disease, known risk factors for severe COVID-19, he added.
He presented a case-control study including 1,193 adult psoriasis patients on biologics or apremilast (Otezla) at Milan’s San Donato Hospital during the period from Feb. 21 to April 9, 2020. The control group comprised more than 10 million individuals, the entire adult population of the Lombardy region, of which Milan is the capital. This was the hardest-hit area in all of Italy during the first wave of COVID-19.
Twenty-two of the 1,193 psoriasis patients experienced confirmed COVID-19 during the study period. Seventeen were quarantined at home because their disease was mild. Five were hospitalized. But no psoriasis patients were placed in intensive care, and none died.
Psoriasis patients on biologics were significantly more likely than the general Lombardian population to test positive for COVID-19, with an unadjusted odds ratio of 3.43. They were at 9.05-fold increased risk of home quarantine for mild disease, and at 3.59-fold greater risk than controls for hospitalization for COVID-19. However, they were not at significantly increased risk of ICU admission. And while they actually had a 59% relative risk reduction for death, this didn’t achieve statistical significance.
Forty-five percent of the psoriasis patients were on an interleukin-17 (IL-17) inhibitor, 22% were on a tumor necrosis factor–alpha inhibitor, and 20% were taking an IL-12/23 inhibitor. Of note, none of 77 patients on apremilast developed COVID-19, even though it is widely considered a less potent psoriasis therapy than the injectable monoclonal antibody biologics.
The French experience
Anne-Claire Fougerousse, MD, and her French coinvestigators conducted a study designed to address a different question: Is it safe to start psoriasis patients on biologics or older conventional systemic agents such as methotrexate during the pandemic?
She presented a French national cross-sectional study of 1,418 adult psoriasis patients on a biologic or standard systemic therapy during a snapshot in time near the peak of the first wave of the pandemic in France: the period from April 27 to May 7, 2020. The group included 1,188 psoriasis patients on maintenance therapy and 230 who had initiated systemic treatment within the past 4 months. More than one-third of the patients had at least one risk factor for severe COVID-19.
Although testing wasn’t available to confirm all cases, 54 patients developed probable COVID-19 during the study period. Only five required hospitalization. None died. The two hospitalized psoriasis patients admitted to an ICU had obesity as a risk factor for severe COVID-19, as did another of the five hospitalized patients, reported Dr. Fougerousse, a dermatologist at the Bégin Military Teaching Hospital in Saint-Mandé, France. Hospitalization for COVID-19 was required in 0.43% of the French treatment initiators, not significantly different from the 0.34% rate in patients on maintenance systemic therapy. A study limitation was the lack of a control group.
Nonetheless, the data did answer the investigators’ main question: “This is the first data showing no increased incidence of severe COVID-19 in psoriasis patients receiving systemic therapy in the treatment initiation period compared to those on maintenance therapy. This may now allow physicians to initiate conventional systemic or biologic therapy in patients with severe psoriasis on a case-by-case basis in the context of the persistent COVID-19 pandemic,” Dr. Fougerousse concluded.
Proposed mechanism of benefit
The Italian study findings that biologics boost the risk of infection with the SARS-CoV-2 virus in psoriasis patients while potentially protecting them against ICU admission and death are backed by a biologically plausible albeit as yet unproven mechanism of action, Dr. Damiani asserted.
He elaborated: A vast body of high-quality clinical trials data demonstrates that these targeted immunosuppressive agents are associated with modestly increased risk of viral infections, including both skin and respiratory tract infections. So there is no reason to suppose these agents would offer protection against the first phase of COVID-19, involving SARS-CoV-2 infection, nor protect against the second (pulmonary phase), whose hallmarks are dyspnea with or without hypoxia. But progression to the third phase, involving hyperinflammation and hypercoagulation – dubbed the cytokine storm – could be a different matter.
“Of particular interest was that our patients on IL-17 inhibitors displayed a really great outcome. Interleukin-17 has procoagulant and prothrombotic effects, organizes bronchoalveolar remodeling, has a profibrotic effect, induces mitochondrial dysfunction, and encourages dendritic cell migration in peribronchial lymph nodes. Therefore, by antagonizing this interleukin, we may have a better prognosis, although further studies are needed to be certain,” Dr. Damiani commented.
Publication of his preliminary findings drew the attention of a group of highly respected thought leaders in psoriasis, including James G. Krueger, MD, head of the laboratory for investigative dermatology and codirector of the center for clinical and investigative science at Rockefeller University, New York.
The Italian report prompted them to analyze data from the phase 4, double-blind, randomized ObePso-S study investigating the effects of the IL-17 inhibitor secukinumab (Cosentyx) on systemic inflammatory markers and gene expression in psoriasis patients. The investigators demonstrated that IL-17–mediated inflammation in psoriasis patients was associated with increased expression of the angiotensin-converting enzyme 2 (ACE2) receptor in lesional skin, and that treatment with secukinumab dropped ACE2 expression to levels seen in nonlesional skin. Given that ACE2 is the chief portal of entry for SARS-CoV-2 and that IL-17 exerts systemic proinflammatory effects, it’s plausible that inhibition of IL-17–mediated inflammation via dampening of ACE2 expression in noncutaneous epithelia “could prove to be advantageous in patients with psoriasis who are at risk for SARS-CoV-2 infection,” according to Dr. Krueger and his coinvestigators in the Journal of Allergy and Clinical Immunology.
Dr. Damiani and Dr. Fougerousse reported having no financial conflicts regarding their studies. The secukinumab/ACE2 receptor study was funded by Novartis.
FROM THE EADV CONGRESS
Hand eczema: Pan-JAK inhibitor delgocitinib shows dose-dependent response in phase 2b trial
a new international phase 2b research suggests.
An investigational pan–Janus kinase inhibitor that blocks all four members of the JAK family, twice-daily delgocitinib doses of 8 mg/g and 20 mg/g demonstrated the highest efficacy in adults with mild to severe chronic hand eczema. By week 16, nearly 40% of patients receiving either dose were clear or almost clear of symptoms.
“By mode of action, we think delgocitinib is more selective in the way of acting,” said lead investigator Margitta Worm, MD, PhD, of the department of dermatology, venereology, and allergology at Charité University Hospital in Berlin, during a presentation of the results at the virtual annual congress of the European Academy of Dermatology and Venereology.
“We do know that JAKs play an important role in chronic inflammation and interfering with the JAK pathway can have anti-inflammatory effects,” Dr. Worm said in an interview. “Whenever it’s possible to use a molecule topically or locally, it’s advantageous for patients because it’s only acting where you apply it and there are no systemic side effects.”
Defined as lasting more than 3 months or relapsing twice or more within a year, chronic hand eczema is a particularly problematic form of atopic dermatitis because “we need our hands every day for almost every activity, so having eczema on your hands has a huge impact on quality of life,” Dr. Worm said.
Many people whose hands are integral to their occupations also have trouble working because of the disorder, she explained. But current topical treatments are limited to emollients, corticosteroids, and calcineurin inhibitors.
“Topical corticosteroids are efficacious, but can cause skin atrophy,” she said. “Their long-term side-effect profile limits their use.”
The number of patients in each treatment group was too small to focus on different subtypes of chronic hand eczema, “but this is something that will probably be looked at in the future,” Dr. Worm said. “At the moment it’s nice to see a dose-dependent clinical efficacy and good tolerability, and now we have to wait for phase 3 data in the future.”
Dr. Worm and colleagues aimed to establish the dose-response relationship of twice-daily applications of delgocitinib cream in doses of 1, 3, 8, and 20 mg/g and a delgocitinib cream vehicle for 16 weeks. The 258 participants (61% women; average age, 46 years) were randomly assigned in equal groups to each dose of delgocitinib cream or the vehicle cream twice daily at centers in Denmark, Germany, and the United States.
The primary endpoint for the double-blind, 26-center trial was the proportion of patients who achieved an Investigator’s Global Assessment score of 0 (“clear”) or 1 (“almost clear”), with a 2-point or higher improvement from baseline over the study period. A key secondary endpoint was a change in the Hand Eczema Severity Index (HECSI) from baseline to week 16.
At week 16, a statistically significant dose response was established for both primary and secondary endpoints (P < .025). More patients in the delgocitinib 8-mg/g and 20-mg/g groups met the primary endpoint (36.5% and 37.7%, respectively) than patients in the 1-mg/g and 3-mg/g groups (21.2% and 7.8%, respectively) and vehicle group (8%, P = .0004).
This primary skin clearance effect at week 16 was demonstrated from week 4 in the 8-mg/g group and week 6 in the 20-mg/g group. But all active doses achieved a statistically significant greater jump in HECSI from baseline to week 16 than the vehicle cream (P < .05).
“The strength of the trial is that there were different concentrations of the substance used,” Dr. Worm said. “When you look to the results, you can demonstrate a dose-dependent clinical efficacy. This is of great value to really compare the efficacy of single doses.”
Most adverse events reported were not considered treatment related and were mild or moderate. The most frequently reported side effects were nasopharyngitis, eczema, and headache.
Commenting on the results, Asli Bilgic, MD, from Akdeniz University in Antalya, Turkey, who was not involved with the study, said that phase 3 studies of delgocitinib should probe further into the effects of the 8-mg/g dosage in this patient group since it appears to show similar efficacy and safety to 20 mg/g.
It’s important for research to focus on hand eczema “because it’s a very common disease, and treatment options are really sparse,” Dr. Bilgic said in an interview.
“Especially in the COVID era, many health care professionals, along with cleaning, catering, and mechanical jobs” are essential workers affected by the condition, she said. “It affects people’s self-esteem and their ability to do their job.”
The study was funded by LEO Pharma. Dr. Worm received lecture honoraria from LEO Pharma. Dr. Bilgic disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
a new international phase 2b research suggests.
An investigational pan–Janus kinase inhibitor that blocks all four members of the JAK family, twice-daily delgocitinib doses of 8 mg/g and 20 mg/g demonstrated the highest efficacy in adults with mild to severe chronic hand eczema. By week 16, nearly 40% of patients receiving either dose were clear or almost clear of symptoms.
“By mode of action, we think delgocitinib is more selective in the way of acting,” said lead investigator Margitta Worm, MD, PhD, of the department of dermatology, venereology, and allergology at Charité University Hospital in Berlin, during a presentation of the results at the virtual annual congress of the European Academy of Dermatology and Venereology.
“We do know that JAKs play an important role in chronic inflammation and interfering with the JAK pathway can have anti-inflammatory effects,” Dr. Worm said in an interview. “Whenever it’s possible to use a molecule topically or locally, it’s advantageous for patients because it’s only acting where you apply it and there are no systemic side effects.”
Defined as lasting more than 3 months or relapsing twice or more within a year, chronic hand eczema is a particularly problematic form of atopic dermatitis because “we need our hands every day for almost every activity, so having eczema on your hands has a huge impact on quality of life,” Dr. Worm said.
Many people whose hands are integral to their occupations also have trouble working because of the disorder, she explained. But current topical treatments are limited to emollients, corticosteroids, and calcineurin inhibitors.
“Topical corticosteroids are efficacious, but can cause skin atrophy,” she said. “Their long-term side-effect profile limits their use.”
The number of patients in each treatment group was too small to focus on different subtypes of chronic hand eczema, “but this is something that will probably be looked at in the future,” Dr. Worm said. “At the moment it’s nice to see a dose-dependent clinical efficacy and good tolerability, and now we have to wait for phase 3 data in the future.”
Dr. Worm and colleagues aimed to establish the dose-response relationship of twice-daily applications of delgocitinib cream in doses of 1, 3, 8, and 20 mg/g and a delgocitinib cream vehicle for 16 weeks. The 258 participants (61% women; average age, 46 years) were randomly assigned in equal groups to each dose of delgocitinib cream or the vehicle cream twice daily at centers in Denmark, Germany, and the United States.
The primary endpoint for the double-blind, 26-center trial was the proportion of patients who achieved an Investigator’s Global Assessment score of 0 (“clear”) or 1 (“almost clear”), with a 2-point or higher improvement from baseline over the study period. A key secondary endpoint was a change in the Hand Eczema Severity Index (HECSI) from baseline to week 16.
At week 16, a statistically significant dose response was established for both primary and secondary endpoints (P < .025). More patients in the delgocitinib 8-mg/g and 20-mg/g groups met the primary endpoint (36.5% and 37.7%, respectively) than patients in the 1-mg/g and 3-mg/g groups (21.2% and 7.8%, respectively) and vehicle group (8%, P = .0004).
This primary skin clearance effect at week 16 was demonstrated from week 4 in the 8-mg/g group and week 6 in the 20-mg/g group. But all active doses achieved a statistically significant greater jump in HECSI from baseline to week 16 than the vehicle cream (P < .05).
“The strength of the trial is that there were different concentrations of the substance used,” Dr. Worm said. “When you look to the results, you can demonstrate a dose-dependent clinical efficacy. This is of great value to really compare the efficacy of single doses.”
Most adverse events reported were not considered treatment related and were mild or moderate. The most frequently reported side effects were nasopharyngitis, eczema, and headache.
Commenting on the results, Asli Bilgic, MD, from Akdeniz University in Antalya, Turkey, who was not involved with the study, said that phase 3 studies of delgocitinib should probe further into the effects of the 8-mg/g dosage in this patient group since it appears to show similar efficacy and safety to 20 mg/g.
It’s important for research to focus on hand eczema “because it’s a very common disease, and treatment options are really sparse,” Dr. Bilgic said in an interview.
“Especially in the COVID era, many health care professionals, along with cleaning, catering, and mechanical jobs” are essential workers affected by the condition, she said. “It affects people’s self-esteem and their ability to do their job.”
The study was funded by LEO Pharma. Dr. Worm received lecture honoraria from LEO Pharma. Dr. Bilgic disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
a new international phase 2b research suggests.
An investigational pan–Janus kinase inhibitor that blocks all four members of the JAK family, twice-daily delgocitinib doses of 8 mg/g and 20 mg/g demonstrated the highest efficacy in adults with mild to severe chronic hand eczema. By week 16, nearly 40% of patients receiving either dose were clear or almost clear of symptoms.
“By mode of action, we think delgocitinib is more selective in the way of acting,” said lead investigator Margitta Worm, MD, PhD, of the department of dermatology, venereology, and allergology at Charité University Hospital in Berlin, during a presentation of the results at the virtual annual congress of the European Academy of Dermatology and Venereology.
“We do know that JAKs play an important role in chronic inflammation and interfering with the JAK pathway can have anti-inflammatory effects,” Dr. Worm said in an interview. “Whenever it’s possible to use a molecule topically or locally, it’s advantageous for patients because it’s only acting where you apply it and there are no systemic side effects.”
Defined as lasting more than 3 months or relapsing twice or more within a year, chronic hand eczema is a particularly problematic form of atopic dermatitis because “we need our hands every day for almost every activity, so having eczema on your hands has a huge impact on quality of life,” Dr. Worm said.
Many people whose hands are integral to their occupations also have trouble working because of the disorder, she explained. But current topical treatments are limited to emollients, corticosteroids, and calcineurin inhibitors.
“Topical corticosteroids are efficacious, but can cause skin atrophy,” she said. “Their long-term side-effect profile limits their use.”
The number of patients in each treatment group was too small to focus on different subtypes of chronic hand eczema, “but this is something that will probably be looked at in the future,” Dr. Worm said. “At the moment it’s nice to see a dose-dependent clinical efficacy and good tolerability, and now we have to wait for phase 3 data in the future.”
Dr. Worm and colleagues aimed to establish the dose-response relationship of twice-daily applications of delgocitinib cream in doses of 1, 3, 8, and 20 mg/g and a delgocitinib cream vehicle for 16 weeks. The 258 participants (61% women; average age, 46 years) were randomly assigned in equal groups to each dose of delgocitinib cream or the vehicle cream twice daily at centers in Denmark, Germany, and the United States.
The primary endpoint for the double-blind, 26-center trial was the proportion of patients who achieved an Investigator’s Global Assessment score of 0 (“clear”) or 1 (“almost clear”), with a 2-point or higher improvement from baseline over the study period. A key secondary endpoint was a change in the Hand Eczema Severity Index (HECSI) from baseline to week 16.
At week 16, a statistically significant dose response was established for both primary and secondary endpoints (P < .025). More patients in the delgocitinib 8-mg/g and 20-mg/g groups met the primary endpoint (36.5% and 37.7%, respectively) than patients in the 1-mg/g and 3-mg/g groups (21.2% and 7.8%, respectively) and vehicle group (8%, P = .0004).
This primary skin clearance effect at week 16 was demonstrated from week 4 in the 8-mg/g group and week 6 in the 20-mg/g group. But all active doses achieved a statistically significant greater jump in HECSI from baseline to week 16 than the vehicle cream (P < .05).
“The strength of the trial is that there were different concentrations of the substance used,” Dr. Worm said. “When you look to the results, you can demonstrate a dose-dependent clinical efficacy. This is of great value to really compare the efficacy of single doses.”
Most adverse events reported were not considered treatment related and were mild or moderate. The most frequently reported side effects were nasopharyngitis, eczema, and headache.
Commenting on the results, Asli Bilgic, MD, from Akdeniz University in Antalya, Turkey, who was not involved with the study, said that phase 3 studies of delgocitinib should probe further into the effects of the 8-mg/g dosage in this patient group since it appears to show similar efficacy and safety to 20 mg/g.
It’s important for research to focus on hand eczema “because it’s a very common disease, and treatment options are really sparse,” Dr. Bilgic said in an interview.
“Especially in the COVID era, many health care professionals, along with cleaning, catering, and mechanical jobs” are essential workers affected by the condition, she said. “It affects people’s self-esteem and their ability to do their job.”
The study was funded by LEO Pharma. Dr. Worm received lecture honoraria from LEO Pharma. Dr. Bilgic disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM THE EADV CONGRESS
No lab monitoring needed in adolescents on dupilumab
, Michael J. Cork, MBBS, PhD, reported at the virtual annual congress of the European Academy of Dermatology and Venereology.
These reassuring results from the ongoing LIBERTY AD PED-OLE study confirm that, as previously established in adults, no blood monitoring is required in adolescents on the monoclonal antibody, which inhibits signaling of interleukins-4 and -13, said Dr. Cork, professor of dermatology and head of Sheffield Dermatology Research at the University of Sheffield (England).
“The practical importance of this finding is that there are no other systemic drugs available that don’t require blood samples. Cyclosporine, methotrexate, and the others used for atopic dermatitis require a lot of blood monitoring, and they’re off-license anyway for use in children and adolescents,” he said in an interview.
Many pediatric patients are afraid of needles and have an intense dislike of blood draws. And in a pandemic, no one wants to come into the office for blood draws if they don’t need to.
“Blood draws are very different from the injection for dupilumab. Taking a blood sample is much more painful for children. The needle in the autoinjector is really, really tiny; you can hardly feel it, and with the autoinjector you can’t even see it,” noted Dr. Cork, who is both a pediatric and adult dermatologist.
This report from the ongoing LIBERTY AD PED-OLE study included 105 patients aged 12-17 years who completed 52 weeks on dupilumab (Dupixent) with assessments of hematologic and serum chemistry parameters at baseline and weeks 16 and 52.
“The results were anticipated, but we want to know the drug is safe in every age group. The immune system is different in different age groups, so we have to be really careful,” Dr. Cork said.
The clinical side-effect profile was the same as in adults, consisting mainly of mild conjunctivitis and injection-site reactions. It’s a much less problematic side effect picture than with the older drugs.
“We’re finding the conjunctivitis to be slightly less severe than in adults, maybe because we’ve learned from the first trials in adults and from clinical experience to use prophylactic therapy. There would be no child going on dupilumab now – and no adult – that I wouldn’t put on prophylactic eye drops with replacement tears. I start them 2 weeks before I start dupilumab,” the dermatologist explained.
He and others with extensive experience using the biologic agent also work closely with an ophthalmologist.
“If we see an eye problem before going on dupilumab we get an assessment and then ophthalmologic monitoring during treatment,” Dr. Cork said.
As a dermatologist specializing in atopic dermatitis, he confessed to feeling deprived over the years as he watched the multitude of targeted biologic agents being developed for psoriasis. When he became involved in the first pediatric clinical trials of dupilumab, he had a realization: “It’s a miraculous treatment.”
“The first child I put on dupilumab spent 70 days in the hospital for IV antibiotics in the prior year. Seventy days! He almost died from MRSA septicemia. His serum IgE was 155,000 kU/L. And his IgE just went down and down and down as the dupilumab took effect. It was just incredible,” he recalled.
Dr. Cork reported receiving research funding from and serving as a consultant to Sanofi and Regeneron, which fund the LIBERTY AD PED-OLE study, as well as numerous other pharmaceutical companies.
SOURCE: Cork MJ. EADV 2020, Abstract 1772.
, Michael J. Cork, MBBS, PhD, reported at the virtual annual congress of the European Academy of Dermatology and Venereology.
These reassuring results from the ongoing LIBERTY AD PED-OLE study confirm that, as previously established in adults, no blood monitoring is required in adolescents on the monoclonal antibody, which inhibits signaling of interleukins-4 and -13, said Dr. Cork, professor of dermatology and head of Sheffield Dermatology Research at the University of Sheffield (England).
“The practical importance of this finding is that there are no other systemic drugs available that don’t require blood samples. Cyclosporine, methotrexate, and the others used for atopic dermatitis require a lot of blood monitoring, and they’re off-license anyway for use in children and adolescents,” he said in an interview.
Many pediatric patients are afraid of needles and have an intense dislike of blood draws. And in a pandemic, no one wants to come into the office for blood draws if they don’t need to.
“Blood draws are very different from the injection for dupilumab. Taking a blood sample is much more painful for children. The needle in the autoinjector is really, really tiny; you can hardly feel it, and with the autoinjector you can’t even see it,” noted Dr. Cork, who is both a pediatric and adult dermatologist.
This report from the ongoing LIBERTY AD PED-OLE study included 105 patients aged 12-17 years who completed 52 weeks on dupilumab (Dupixent) with assessments of hematologic and serum chemistry parameters at baseline and weeks 16 and 52.
“The results were anticipated, but we want to know the drug is safe in every age group. The immune system is different in different age groups, so we have to be really careful,” Dr. Cork said.
The clinical side-effect profile was the same as in adults, consisting mainly of mild conjunctivitis and injection-site reactions. It’s a much less problematic side effect picture than with the older drugs.
“We’re finding the conjunctivitis to be slightly less severe than in adults, maybe because we’ve learned from the first trials in adults and from clinical experience to use prophylactic therapy. There would be no child going on dupilumab now – and no adult – that I wouldn’t put on prophylactic eye drops with replacement tears. I start them 2 weeks before I start dupilumab,” the dermatologist explained.
He and others with extensive experience using the biologic agent also work closely with an ophthalmologist.
“If we see an eye problem before going on dupilumab we get an assessment and then ophthalmologic monitoring during treatment,” Dr. Cork said.
As a dermatologist specializing in atopic dermatitis, he confessed to feeling deprived over the years as he watched the multitude of targeted biologic agents being developed for psoriasis. When he became involved in the first pediatric clinical trials of dupilumab, he had a realization: “It’s a miraculous treatment.”
“The first child I put on dupilumab spent 70 days in the hospital for IV antibiotics in the prior year. Seventy days! He almost died from MRSA septicemia. His serum IgE was 155,000 kU/L. And his IgE just went down and down and down as the dupilumab took effect. It was just incredible,” he recalled.
Dr. Cork reported receiving research funding from and serving as a consultant to Sanofi and Regeneron, which fund the LIBERTY AD PED-OLE study, as well as numerous other pharmaceutical companies.
SOURCE: Cork MJ. EADV 2020, Abstract 1772.
, Michael J. Cork, MBBS, PhD, reported at the virtual annual congress of the European Academy of Dermatology and Venereology.
These reassuring results from the ongoing LIBERTY AD PED-OLE study confirm that, as previously established in adults, no blood monitoring is required in adolescents on the monoclonal antibody, which inhibits signaling of interleukins-4 and -13, said Dr. Cork, professor of dermatology and head of Sheffield Dermatology Research at the University of Sheffield (England).
“The practical importance of this finding is that there are no other systemic drugs available that don’t require blood samples. Cyclosporine, methotrexate, and the others used for atopic dermatitis require a lot of blood monitoring, and they’re off-license anyway for use in children and adolescents,” he said in an interview.
Many pediatric patients are afraid of needles and have an intense dislike of blood draws. And in a pandemic, no one wants to come into the office for blood draws if they don’t need to.
“Blood draws are very different from the injection for dupilumab. Taking a blood sample is much more painful for children. The needle in the autoinjector is really, really tiny; you can hardly feel it, and with the autoinjector you can’t even see it,” noted Dr. Cork, who is both a pediatric and adult dermatologist.
This report from the ongoing LIBERTY AD PED-OLE study included 105 patients aged 12-17 years who completed 52 weeks on dupilumab (Dupixent) with assessments of hematologic and serum chemistry parameters at baseline and weeks 16 and 52.
“The results were anticipated, but we want to know the drug is safe in every age group. The immune system is different in different age groups, so we have to be really careful,” Dr. Cork said.
The clinical side-effect profile was the same as in adults, consisting mainly of mild conjunctivitis and injection-site reactions. It’s a much less problematic side effect picture than with the older drugs.
“We’re finding the conjunctivitis to be slightly less severe than in adults, maybe because we’ve learned from the first trials in adults and from clinical experience to use prophylactic therapy. There would be no child going on dupilumab now – and no adult – that I wouldn’t put on prophylactic eye drops with replacement tears. I start them 2 weeks before I start dupilumab,” the dermatologist explained.
He and others with extensive experience using the biologic agent also work closely with an ophthalmologist.
“If we see an eye problem before going on dupilumab we get an assessment and then ophthalmologic monitoring during treatment,” Dr. Cork said.
As a dermatologist specializing in atopic dermatitis, he confessed to feeling deprived over the years as he watched the multitude of targeted biologic agents being developed for psoriasis. When he became involved in the first pediatric clinical trials of dupilumab, he had a realization: “It’s a miraculous treatment.”
“The first child I put on dupilumab spent 70 days in the hospital for IV antibiotics in the prior year. Seventy days! He almost died from MRSA septicemia. His serum IgE was 155,000 kU/L. And his IgE just went down and down and down as the dupilumab took effect. It was just incredible,” he recalled.
Dr. Cork reported receiving research funding from and serving as a consultant to Sanofi and Regeneron, which fund the LIBERTY AD PED-OLE study, as well as numerous other pharmaceutical companies.
SOURCE: Cork MJ. EADV 2020, Abstract 1772.
FROM THE EADV CONGRESS
OTC topical ivermectin lotion earns FDA approval for head lice
in patients aged 6 months and older.
Ivermectin was approved as a prescription treatment for head lice in February 2012, according to an FDA press release, and is now approved as an over-the-counter treatment through an “Rx-to-OTC” switch process. The approval was granted to Arbor Pharmaceuticals.
The expanded approval for ivermectin increases access to effective care for head lice, which is estimated to affect between 6 million and 12 million children each year in the United States, according to the Centers for Disease Control and Prevention.
“The Rx-to-OTC switch process aims to promote public health by increasing consumer access to drugs that would otherwise only be available by prescription,” Theresa Michele, MD, acting director of the Office of Nonprescription Drugs in the FDA’s Center for Drug Evaluation and Research, said in the press release.
The FDA also noted in the press release that “Sklice, and its active ingredient ivermectin, have not been shown to be safe or effective for the treatment or prevention of COVID-19 and they are not FDA-approved for this use.”
The drug is approved only for treating head lice, and should be used on the scalp and dry hair, according to the labeling. In the wake of the approval, ivermectin will no longer be available as a prescription drug, according to the FDA, and patients currently using prescription versions should contact their health care providers.
An Rx-to-OTC switch is contingent on the manufacturer’s data showing that the drug is safe and effective when used as directed. In addition, “the manufacturer must show that consumers can understand how to use the drug safely and effectively without the supervision of a health care professional,” according to the FDA.
in patients aged 6 months and older.
Ivermectin was approved as a prescription treatment for head lice in February 2012, according to an FDA press release, and is now approved as an over-the-counter treatment through an “Rx-to-OTC” switch process. The approval was granted to Arbor Pharmaceuticals.
The expanded approval for ivermectin increases access to effective care for head lice, which is estimated to affect between 6 million and 12 million children each year in the United States, according to the Centers for Disease Control and Prevention.
“The Rx-to-OTC switch process aims to promote public health by increasing consumer access to drugs that would otherwise only be available by prescription,” Theresa Michele, MD, acting director of the Office of Nonprescription Drugs in the FDA’s Center for Drug Evaluation and Research, said in the press release.
The FDA also noted in the press release that “Sklice, and its active ingredient ivermectin, have not been shown to be safe or effective for the treatment or prevention of COVID-19 and they are not FDA-approved for this use.”
The drug is approved only for treating head lice, and should be used on the scalp and dry hair, according to the labeling. In the wake of the approval, ivermectin will no longer be available as a prescription drug, according to the FDA, and patients currently using prescription versions should contact their health care providers.
An Rx-to-OTC switch is contingent on the manufacturer’s data showing that the drug is safe and effective when used as directed. In addition, “the manufacturer must show that consumers can understand how to use the drug safely and effectively without the supervision of a health care professional,” according to the FDA.
in patients aged 6 months and older.
Ivermectin was approved as a prescription treatment for head lice in February 2012, according to an FDA press release, and is now approved as an over-the-counter treatment through an “Rx-to-OTC” switch process. The approval was granted to Arbor Pharmaceuticals.
The expanded approval for ivermectin increases access to effective care for head lice, which is estimated to affect between 6 million and 12 million children each year in the United States, according to the Centers for Disease Control and Prevention.
“The Rx-to-OTC switch process aims to promote public health by increasing consumer access to drugs that would otherwise only be available by prescription,” Theresa Michele, MD, acting director of the Office of Nonprescription Drugs in the FDA’s Center for Drug Evaluation and Research, said in the press release.
The FDA also noted in the press release that “Sklice, and its active ingredient ivermectin, have not been shown to be safe or effective for the treatment or prevention of COVID-19 and they are not FDA-approved for this use.”
The drug is approved only for treating head lice, and should be used on the scalp and dry hair, according to the labeling. In the wake of the approval, ivermectin will no longer be available as a prescription drug, according to the FDA, and patients currently using prescription versions should contact their health care providers.
An Rx-to-OTC switch is contingent on the manufacturer’s data showing that the drug is safe and effective when used as directed. In addition, “the manufacturer must show that consumers can understand how to use the drug safely and effectively without the supervision of a health care professional,” according to the FDA.
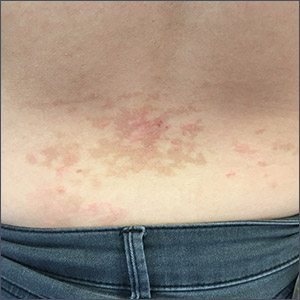
Pruritic rash on flank, back, and chest
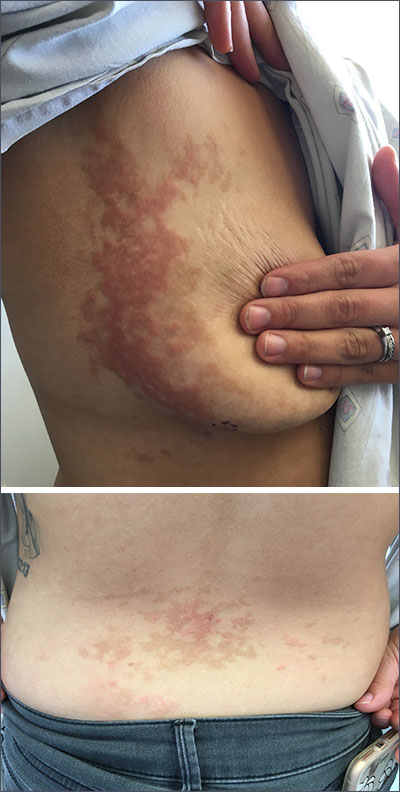
The patient was given a diagnosis of prurigo pigmentosa based on the characteristic pruritic rash that had developed after the patient started a strict ketogenic diet.
Prurigo pigmentosa is a benign, pruritic rash that most commonly presents with erythematous or hyperpigmented, symmetrically distributed urticarial papules and plaques on the chest and back. Females represent approximately 70% of cases with a predominant age range of 11 to 30. It more commonly is seen in people of Asian descent.
While the pathophysiology remains unknown, the rash most commonly occurs in association with diabetes, ketosis, and more recently with ketogenic diets. Despite occurring in only a fraction of patients on the ketogenic diet, the characteristic presentation has led to the alternative name of the “keto rash” in online nutritional forums and blogs.
The diagnosis is made clinically, so the appearance of a symmetric pruritic, hyperpigmented rash on the chest and back should prompt the physician to ask about any recent changes in diet. Laboratory analysis is unnecessary, as a complete blood count, basic metabolic panel, and liver function panel are almost always normal.
Other conditions can mimic prurigo pigmentosa such as urticaria, irritant contact dermatitis, confluent and reticulated papillomatosis, and pityriasis rosea.
Primary treatment includes resumption of a normal diet. This often leads to rapid resolution of pruritis. Residual hyperpigmentation may take months to fade. If additional treatment is required, minocycline 100 to 200 mg/d has been reported most effective, likely due to its anti-inflammatory properties. Topical corticosteroids and oral antihistamines provide symptomatic relief in some patients.
This patient had resolution of the pruritis and urticarial lesions within 2 days of resuming a normal diet; however, residual asymptomatic hyperpigmentation persisted. A retrial of the ketogenic diet initiated a flare of the rash in the same distribution. It rapidly resolved with carbohydrate intake.
This case was adapted from: Croom D, Barlow T, Landers JT. Pruritic rash on chest and back. J Fam Pract. 2019;68:113-114,116

The patient was given a diagnosis of prurigo pigmentosa based on the characteristic pruritic rash that had developed after the patient started a strict ketogenic diet.
Prurigo pigmentosa is a benign, pruritic rash that most commonly presents with erythematous or hyperpigmented, symmetrically distributed urticarial papules and plaques on the chest and back. Females represent approximately 70% of cases with a predominant age range of 11 to 30. It more commonly is seen in people of Asian descent.
While the pathophysiology remains unknown, the rash most commonly occurs in association with diabetes, ketosis, and more recently with ketogenic diets. Despite occurring in only a fraction of patients on the ketogenic diet, the characteristic presentation has led to the alternative name of the “keto rash” in online nutritional forums and blogs.
The diagnosis is made clinically, so the appearance of a symmetric pruritic, hyperpigmented rash on the chest and back should prompt the physician to ask about any recent changes in diet. Laboratory analysis is unnecessary, as a complete blood count, basic metabolic panel, and liver function panel are almost always normal.
Other conditions can mimic prurigo pigmentosa such as urticaria, irritant contact dermatitis, confluent and reticulated papillomatosis, and pityriasis rosea.
Primary treatment includes resumption of a normal diet. This often leads to rapid resolution of pruritis. Residual hyperpigmentation may take months to fade. If additional treatment is required, minocycline 100 to 200 mg/d has been reported most effective, likely due to its anti-inflammatory properties. Topical corticosteroids and oral antihistamines provide symptomatic relief in some patients.
This patient had resolution of the pruritis and urticarial lesions within 2 days of resuming a normal diet; however, residual asymptomatic hyperpigmentation persisted. A retrial of the ketogenic diet initiated a flare of the rash in the same distribution. It rapidly resolved with carbohydrate intake.
This case was adapted from: Croom D, Barlow T, Landers JT. Pruritic rash on chest and back. J Fam Pract. 2019;68:113-114,116

The patient was given a diagnosis of prurigo pigmentosa based on the characteristic pruritic rash that had developed after the patient started a strict ketogenic diet.
Prurigo pigmentosa is a benign, pruritic rash that most commonly presents with erythematous or hyperpigmented, symmetrically distributed urticarial papules and plaques on the chest and back. Females represent approximately 70% of cases with a predominant age range of 11 to 30. It more commonly is seen in people of Asian descent.
While the pathophysiology remains unknown, the rash most commonly occurs in association with diabetes, ketosis, and more recently with ketogenic diets. Despite occurring in only a fraction of patients on the ketogenic diet, the characteristic presentation has led to the alternative name of the “keto rash” in online nutritional forums and blogs.
The diagnosis is made clinically, so the appearance of a symmetric pruritic, hyperpigmented rash on the chest and back should prompt the physician to ask about any recent changes in diet. Laboratory analysis is unnecessary, as a complete blood count, basic metabolic panel, and liver function panel are almost always normal.
Other conditions can mimic prurigo pigmentosa such as urticaria, irritant contact dermatitis, confluent and reticulated papillomatosis, and pityriasis rosea.
Primary treatment includes resumption of a normal diet. This often leads to rapid resolution of pruritis. Residual hyperpigmentation may take months to fade. If additional treatment is required, minocycline 100 to 200 mg/d has been reported most effective, likely due to its anti-inflammatory properties. Topical corticosteroids and oral antihistamines provide symptomatic relief in some patients.
This patient had resolution of the pruritis and urticarial lesions within 2 days of resuming a normal diet; however, residual asymptomatic hyperpigmentation persisted. A retrial of the ketogenic diet initiated a flare of the rash in the same distribution. It rapidly resolved with carbohydrate intake.
This case was adapted from: Croom D, Barlow T, Landers JT. Pruritic rash on chest and back. J Fam Pract. 2019;68:113-114,116
How to assess erythema in children with skin of color
When assessing inflammatory dermatoses in children with skin of color, it may be necessary to train the eye to recognize subtle changes and colors other than red, a doctor suggested at the virtual American Academy of Pediatrics annual meeting.
First, doctors should see whether they can detect any erythema, said Latanya T. Benjamin, MD, associate professor of pediatric dermatology at Florida Atlantic University, Boca Raton. “If the answer is no because of the background competing chromophore, then shift your focus off of the erythema and perhaps onto other colors that the skin can demonstrate,” such as red-brown, violaceous, or grayish hues.
Comparing involved areas with normal skin also may help. “Sometimes you can pick up subtleties in colors that way,” Dr. Benjamin said.
Finally, look for other changes that could relate to the patient’s condition. For example, when diagnosing acne, Dr. Benjamin looks for pigmentary sequelae like hyperpigmentation. “If a patient has atopic dermatitis, is there hypopigmentation on other areas of the face?”
Consider cutaneous T-cell lymphoma in the differential diagnosis of generalized hypopigmented patches and plaques in patients with darker skin types, Dr. Benjamin noted. Other diagnoses that may result in hypopigmentation include pityriasis alba, vitiligo, tinea versicolor, ash-leaf macules, Hansen’s disease, postinflammatory hypopigmentation secondary to atopic dermatitis, and tinea corporis.
Be sensitive to the fact that changes in skin color can be “very annoying or devastating to the family,” even with medically benign conditions such as pityriasis alba, Dr. Benjamin added.
Detecting redness in brown skin tones can take practice, Candrice R. Heath, MD, a member of the board of directors for the Skin of Color Society, commented in an interview.
Furthermore, presentations vary. For instance, depictions of atopic dermatitis in educational materials may focus on red patches and plaques but “miss that there are several presentations in those with darker skin tones, including follicular prominence, hyperpigmented plaques, and coin-shaped lesions,” said Dr. Heath, assistant professor of dermatology at Temple University, Philadelphia.
“The skin of color population is growing,” noted Dr. Heath. “By 2023, there will be more children with skin of color than without in the United States.”
While Dr. Heath has lectured about skin of color as it relates to pediatric patients for years, “now with the nation’s renewed interest in disparities in health care, it is the perfect time to highlight conditions that present more commonly in skin of color and present differently in those with skin of color.”
Dr. Benjamin had no conflicts of interest. Dr. Heath serves as associate editor of Cutis, which is owned by the same company as this publication.
When assessing inflammatory dermatoses in children with skin of color, it may be necessary to train the eye to recognize subtle changes and colors other than red, a doctor suggested at the virtual American Academy of Pediatrics annual meeting.
First, doctors should see whether they can detect any erythema, said Latanya T. Benjamin, MD, associate professor of pediatric dermatology at Florida Atlantic University, Boca Raton. “If the answer is no because of the background competing chromophore, then shift your focus off of the erythema and perhaps onto other colors that the skin can demonstrate,” such as red-brown, violaceous, or grayish hues.
Comparing involved areas with normal skin also may help. “Sometimes you can pick up subtleties in colors that way,” Dr. Benjamin said.
Finally, look for other changes that could relate to the patient’s condition. For example, when diagnosing acne, Dr. Benjamin looks for pigmentary sequelae like hyperpigmentation. “If a patient has atopic dermatitis, is there hypopigmentation on other areas of the face?”
Consider cutaneous T-cell lymphoma in the differential diagnosis of generalized hypopigmented patches and plaques in patients with darker skin types, Dr. Benjamin noted. Other diagnoses that may result in hypopigmentation include pityriasis alba, vitiligo, tinea versicolor, ash-leaf macules, Hansen’s disease, postinflammatory hypopigmentation secondary to atopic dermatitis, and tinea corporis.
Be sensitive to the fact that changes in skin color can be “very annoying or devastating to the family,” even with medically benign conditions such as pityriasis alba, Dr. Benjamin added.
Detecting redness in brown skin tones can take practice, Candrice R. Heath, MD, a member of the board of directors for the Skin of Color Society, commented in an interview.
Furthermore, presentations vary. For instance, depictions of atopic dermatitis in educational materials may focus on red patches and plaques but “miss that there are several presentations in those with darker skin tones, including follicular prominence, hyperpigmented plaques, and coin-shaped lesions,” said Dr. Heath, assistant professor of dermatology at Temple University, Philadelphia.
“The skin of color population is growing,” noted Dr. Heath. “By 2023, there will be more children with skin of color than without in the United States.”
While Dr. Heath has lectured about skin of color as it relates to pediatric patients for years, “now with the nation’s renewed interest in disparities in health care, it is the perfect time to highlight conditions that present more commonly in skin of color and present differently in those with skin of color.”
Dr. Benjamin had no conflicts of interest. Dr. Heath serves as associate editor of Cutis, which is owned by the same company as this publication.
When assessing inflammatory dermatoses in children with skin of color, it may be necessary to train the eye to recognize subtle changes and colors other than red, a doctor suggested at the virtual American Academy of Pediatrics annual meeting.
First, doctors should see whether they can detect any erythema, said Latanya T. Benjamin, MD, associate professor of pediatric dermatology at Florida Atlantic University, Boca Raton. “If the answer is no because of the background competing chromophore, then shift your focus off of the erythema and perhaps onto other colors that the skin can demonstrate,” such as red-brown, violaceous, or grayish hues.
Comparing involved areas with normal skin also may help. “Sometimes you can pick up subtleties in colors that way,” Dr. Benjamin said.
Finally, look for other changes that could relate to the patient’s condition. For example, when diagnosing acne, Dr. Benjamin looks for pigmentary sequelae like hyperpigmentation. “If a patient has atopic dermatitis, is there hypopigmentation on other areas of the face?”
Consider cutaneous T-cell lymphoma in the differential diagnosis of generalized hypopigmented patches and plaques in patients with darker skin types, Dr. Benjamin noted. Other diagnoses that may result in hypopigmentation include pityriasis alba, vitiligo, tinea versicolor, ash-leaf macules, Hansen’s disease, postinflammatory hypopigmentation secondary to atopic dermatitis, and tinea corporis.
Be sensitive to the fact that changes in skin color can be “very annoying or devastating to the family,” even with medically benign conditions such as pityriasis alba, Dr. Benjamin added.
Detecting redness in brown skin tones can take practice, Candrice R. Heath, MD, a member of the board of directors for the Skin of Color Society, commented in an interview.
Furthermore, presentations vary. For instance, depictions of atopic dermatitis in educational materials may focus on red patches and plaques but “miss that there are several presentations in those with darker skin tones, including follicular prominence, hyperpigmented plaques, and coin-shaped lesions,” said Dr. Heath, assistant professor of dermatology at Temple University, Philadelphia.
“The skin of color population is growing,” noted Dr. Heath. “By 2023, there will be more children with skin of color than without in the United States.”
While Dr. Heath has lectured about skin of color as it relates to pediatric patients for years, “now with the nation’s renewed interest in disparities in health care, it is the perfect time to highlight conditions that present more commonly in skin of color and present differently in those with skin of color.”
Dr. Benjamin had no conflicts of interest. Dr. Heath serves as associate editor of Cutis, which is owned by the same company as this publication.
FROM AAP 2020
Red hair in women linked to elevated CRP levels in Nurses’ Health Study
Red-haired women were significantly more likely than were women with nonred hair to have elevated levels of C-reactive protein that may increase risk for cardiovascular conditions, according to data from nearly 9,000 women participating in the Nurses’ Health Study.
“Positive associations between red hair and cardiovascular disease and cancer in women, but not men, have been reported,” wrote Rebecca I. Hartman, MD, of Brigham and Women’s Hospital, Harvard Medical School, Boston, and colleagues.
In a study published in the Journal of Investigative Dermatology, they reviewed data from the Nurses’ Health Study, a 1976 cohort study of 121,700 women registered nurses in the United States. They analyzed blood specimens from 8,994 women that were collected between 1989 and 1990. Participants’ natural hair color was determined by asking them their natural hair color at age 21 years, with choices of red, blonde, light brown, dark brown, or black. Overall, dark brown/black hair was the most common color (45%) and 390 of the women (4.3%) had red hair.
The average CRP levels were significantly higher for women with red hair (3.7 mg/L), compared with those with blonde (3.3 mg/L), light brown (3.0 mg/mL), or dark brown/black (3.2 mg/L).
Using the CRP levels for red-haired women as a reference, women with blond, light brown, and dark brown/black hair averaged significantly lower CRP levels than those of red-haired women in an age-adjusted model (–15.2%, –18/1%, and –14.2%, respectively) and in a multivariate analysis (–12.7%, –14.1%, and –10.9%, respectively).
Non-red-haired women had significantly lower odds of high CRP levels compared with red-haired women, with odds ratios of 0.62, 0.60, and 0.67 for women with blonde, light brown, and dark brown/black hair, respectively, in multivariate analysis, the researchers found.
The study was limited by several factors including the use of self-reports for hair color and the relative homogeneity of the Nurses’ Health Study, which has a population of mostly white, female health professionals, the researchers noted.
However, the findings of significantly increased CRP levels “could potentially explain a prior report of increased risks of cardiovascular disease and cancer in red-haired women,” they said. “Although, we observed similar associations in the NHS between red hair and cardiovascular disease and cancer, they were not statistically significant,” they added.
Additional studies are needed to validate and examine the clinical significance of the results, they concluded.
“Elevated CRP levels, a marker of inflammation, have been associated with increased risk for several diseases, including colon cancer and heart disease,” lead author Dr. Hartman said in an interview. “Another study suggested red-haired women have elevated risks of cardiovascular disease and cancer. We wanted to see if different levels of inflammation in red-haired women could possibly explain these findings.”
She said she was not surprised by the findings, “as they were in line with our hypothesis.” In addition, “animal studies suggest that the gene most responsible for red hair, MC1R, may be linked to inflammation,” she said.
While red-haired women were found to have higher CRP levels in the study, “the underlying mechanism and clinical significance remain unknown,” and more research is needed, Dr. Hartman emphasized. “First, our findings need to be validated in women and also examined in men. If our findings are validated, future studies should examine the mechanism of CRP elevation in red-haired women, and whether these women have elevated risks of colon cancer and heart disease,” she said.
“If red-haired women do have increased levels of inflammation, and as a result have elevated risks of colon cancer and heart disease, then future interventions can focus on enhanced screening and possibly chemoprevention in this population,” she added.
The study was supported by the National Institutes of Health. Lead author Dr. Hartman was supported by an American Skin Association Research Grant.
SOURCE: Hartman RI et al. J Invest Dermatol. 2020 Oct 12. doi: 10.1016/j.jid.2020.09.015.
Red-haired women were significantly more likely than were women with nonred hair to have elevated levels of C-reactive protein that may increase risk for cardiovascular conditions, according to data from nearly 9,000 women participating in the Nurses’ Health Study.
“Positive associations between red hair and cardiovascular disease and cancer in women, but not men, have been reported,” wrote Rebecca I. Hartman, MD, of Brigham and Women’s Hospital, Harvard Medical School, Boston, and colleagues.
In a study published in the Journal of Investigative Dermatology, they reviewed data from the Nurses’ Health Study, a 1976 cohort study of 121,700 women registered nurses in the United States. They analyzed blood specimens from 8,994 women that were collected between 1989 and 1990. Participants’ natural hair color was determined by asking them their natural hair color at age 21 years, with choices of red, blonde, light brown, dark brown, or black. Overall, dark brown/black hair was the most common color (45%) and 390 of the women (4.3%) had red hair.
The average CRP levels were significantly higher for women with red hair (3.7 mg/L), compared with those with blonde (3.3 mg/L), light brown (3.0 mg/mL), or dark brown/black (3.2 mg/L).
Using the CRP levels for red-haired women as a reference, women with blond, light brown, and dark brown/black hair averaged significantly lower CRP levels than those of red-haired women in an age-adjusted model (–15.2%, –18/1%, and –14.2%, respectively) and in a multivariate analysis (–12.7%, –14.1%, and –10.9%, respectively).
Non-red-haired women had significantly lower odds of high CRP levels compared with red-haired women, with odds ratios of 0.62, 0.60, and 0.67 for women with blonde, light brown, and dark brown/black hair, respectively, in multivariate analysis, the researchers found.
The study was limited by several factors including the use of self-reports for hair color and the relative homogeneity of the Nurses’ Health Study, which has a population of mostly white, female health professionals, the researchers noted.
However, the findings of significantly increased CRP levels “could potentially explain a prior report of increased risks of cardiovascular disease and cancer in red-haired women,” they said. “Although, we observed similar associations in the NHS between red hair and cardiovascular disease and cancer, they were not statistically significant,” they added.
Additional studies are needed to validate and examine the clinical significance of the results, they concluded.
“Elevated CRP levels, a marker of inflammation, have been associated with increased risk for several diseases, including colon cancer and heart disease,” lead author Dr. Hartman said in an interview. “Another study suggested red-haired women have elevated risks of cardiovascular disease and cancer. We wanted to see if different levels of inflammation in red-haired women could possibly explain these findings.”
She said she was not surprised by the findings, “as they were in line with our hypothesis.” In addition, “animal studies suggest that the gene most responsible for red hair, MC1R, may be linked to inflammation,” she said.
While red-haired women were found to have higher CRP levels in the study, “the underlying mechanism and clinical significance remain unknown,” and more research is needed, Dr. Hartman emphasized. “First, our findings need to be validated in women and also examined in men. If our findings are validated, future studies should examine the mechanism of CRP elevation in red-haired women, and whether these women have elevated risks of colon cancer and heart disease,” she said.
“If red-haired women do have increased levels of inflammation, and as a result have elevated risks of colon cancer and heart disease, then future interventions can focus on enhanced screening and possibly chemoprevention in this population,” she added.
The study was supported by the National Institutes of Health. Lead author Dr. Hartman was supported by an American Skin Association Research Grant.
SOURCE: Hartman RI et al. J Invest Dermatol. 2020 Oct 12. doi: 10.1016/j.jid.2020.09.015.
Red-haired women were significantly more likely than were women with nonred hair to have elevated levels of C-reactive protein that may increase risk for cardiovascular conditions, according to data from nearly 9,000 women participating in the Nurses’ Health Study.
“Positive associations between red hair and cardiovascular disease and cancer in women, but not men, have been reported,” wrote Rebecca I. Hartman, MD, of Brigham and Women’s Hospital, Harvard Medical School, Boston, and colleagues.
In a study published in the Journal of Investigative Dermatology, they reviewed data from the Nurses’ Health Study, a 1976 cohort study of 121,700 women registered nurses in the United States. They analyzed blood specimens from 8,994 women that were collected between 1989 and 1990. Participants’ natural hair color was determined by asking them their natural hair color at age 21 years, with choices of red, blonde, light brown, dark brown, or black. Overall, dark brown/black hair was the most common color (45%) and 390 of the women (4.3%) had red hair.
The average CRP levels were significantly higher for women with red hair (3.7 mg/L), compared with those with blonde (3.3 mg/L), light brown (3.0 mg/mL), or dark brown/black (3.2 mg/L).
Using the CRP levels for red-haired women as a reference, women with blond, light brown, and dark brown/black hair averaged significantly lower CRP levels than those of red-haired women in an age-adjusted model (–15.2%, –18/1%, and –14.2%, respectively) and in a multivariate analysis (–12.7%, –14.1%, and –10.9%, respectively).
Non-red-haired women had significantly lower odds of high CRP levels compared with red-haired women, with odds ratios of 0.62, 0.60, and 0.67 for women with blonde, light brown, and dark brown/black hair, respectively, in multivariate analysis, the researchers found.
The study was limited by several factors including the use of self-reports for hair color and the relative homogeneity of the Nurses’ Health Study, which has a population of mostly white, female health professionals, the researchers noted.
However, the findings of significantly increased CRP levels “could potentially explain a prior report of increased risks of cardiovascular disease and cancer in red-haired women,” they said. “Although, we observed similar associations in the NHS between red hair and cardiovascular disease and cancer, they were not statistically significant,” they added.
Additional studies are needed to validate and examine the clinical significance of the results, they concluded.
“Elevated CRP levels, a marker of inflammation, have been associated with increased risk for several diseases, including colon cancer and heart disease,” lead author Dr. Hartman said in an interview. “Another study suggested red-haired women have elevated risks of cardiovascular disease and cancer. We wanted to see if different levels of inflammation in red-haired women could possibly explain these findings.”
She said she was not surprised by the findings, “as they were in line with our hypothesis.” In addition, “animal studies suggest that the gene most responsible for red hair, MC1R, may be linked to inflammation,” she said.
While red-haired women were found to have higher CRP levels in the study, “the underlying mechanism and clinical significance remain unknown,” and more research is needed, Dr. Hartman emphasized. “First, our findings need to be validated in women and also examined in men. If our findings are validated, future studies should examine the mechanism of CRP elevation in red-haired women, and whether these women have elevated risks of colon cancer and heart disease,” she said.
“If red-haired women do have increased levels of inflammation, and as a result have elevated risks of colon cancer and heart disease, then future interventions can focus on enhanced screening and possibly chemoprevention in this population,” she added.
The study was supported by the National Institutes of Health. Lead author Dr. Hartman was supported by an American Skin Association Research Grant.
SOURCE: Hartman RI et al. J Invest Dermatol. 2020 Oct 12. doi: 10.1016/j.jid.2020.09.015.
FROM THE JOURNAL OF INVESTIGATIVE DERMATOLOGY
Eyebrow hair loss
Although it appeared that the hair loss was preceded by some dry skin, the patch of hair loss was smooth and nonscarring, consistent with a diagnosis of alopecia areata (AA).
AA typically is found on the scalp in solitary areas but can affect the whole body, including the eyelashes and eyebrows. Affected hair typically is narrower at the proximal end, resembling an exclamation point, as the hair fails to grow and falls out. Sometimes, patches of alopecia may coalesce into a larger area. Nail changes may be noted, as well. Nails may become brittle, with pitting and/or longitudinal ridges. Patients are usually asymptomatic but may complain of pruritus.
AA is believed to be an autoimmune disorder. It affects males and females of all ages but is more common in children and young adults. AA is believed to result from a T-cell–mediated immune response that transitions the hair follicles from the growth phase to the resting phase. This leads to sudden hair loss and inhibition of regrowth of the hair. However, the hair follicle is not permanently destroyed as in other processes of alopecia. There is also an association between AA and other autoimmune disorders such as vitiligo, thyroid disease, and lupus.
The diagnosis usually is made clinically, as in this patient, but a definitive diagnosis can be made by biopsy and pathology. Other differential diagnoses to consider are trichotillomania, tinea, traumatic alopecia, and lupus.
In almost half of cases, AA is self-resolving; therefore, in first episodes of localized disease, watchful waiting is appropriate. Treatment is tailored to the individual patient and may be difficult. Intralesional corticosteroids often are used for mild cases. Typically, 10 mg/mL of a glucocorticoid is injected into the mid-dermis every 4 to 6 weeks; however, this treatment carries a risk of transient or even permanent atrophy of the injection site. Potent topical corticosteroids often are used, especially in children who do not tolerate intralesional injections, and success can be variable. Other topical treatments to consider are photochemotherapy (psoralen plus UVA), or an irritant agent such as anthralin and a vasodilator such as minoxidil. Regrowth can be expected in a few months to a year, but recurrence is common.
Image courtesy of Stacy Nguy, MD, and text courtesy of Stacy Nguy, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Papadopoulos AJ, Schwartz RA, Janniger C. Alopecia areata: pathogenesis, diagnosis, and therapy. Am J Clin Dermatol. 2000;1:101-105

Although it appeared that the hair loss was preceded by some dry skin, the patch of hair loss was smooth and nonscarring, consistent with a diagnosis of alopecia areata (AA).
AA typically is found on the scalp in solitary areas but can affect the whole body, including the eyelashes and eyebrows. Affected hair typically is narrower at the proximal end, resembling an exclamation point, as the hair fails to grow and falls out. Sometimes, patches of alopecia may coalesce into a larger area. Nail changes may be noted, as well. Nails may become brittle, with pitting and/or longitudinal ridges. Patients are usually asymptomatic but may complain of pruritus.
AA is believed to be an autoimmune disorder. It affects males and females of all ages but is more common in children and young adults. AA is believed to result from a T-cell–mediated immune response that transitions the hair follicles from the growth phase to the resting phase. This leads to sudden hair loss and inhibition of regrowth of the hair. However, the hair follicle is not permanently destroyed as in other processes of alopecia. There is also an association between AA and other autoimmune disorders such as vitiligo, thyroid disease, and lupus.
The diagnosis usually is made clinically, as in this patient, but a definitive diagnosis can be made by biopsy and pathology. Other differential diagnoses to consider are trichotillomania, tinea, traumatic alopecia, and lupus.
In almost half of cases, AA is self-resolving; therefore, in first episodes of localized disease, watchful waiting is appropriate. Treatment is tailored to the individual patient and may be difficult. Intralesional corticosteroids often are used for mild cases. Typically, 10 mg/mL of a glucocorticoid is injected into the mid-dermis every 4 to 6 weeks; however, this treatment carries a risk of transient or even permanent atrophy of the injection site. Potent topical corticosteroids often are used, especially in children who do not tolerate intralesional injections, and success can be variable. Other topical treatments to consider are photochemotherapy (psoralen plus UVA), or an irritant agent such as anthralin and a vasodilator such as minoxidil. Regrowth can be expected in a few months to a year, but recurrence is common.
Image courtesy of Stacy Nguy, MD, and text courtesy of Stacy Nguy, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

Although it appeared that the hair loss was preceded by some dry skin, the patch of hair loss was smooth and nonscarring, consistent with a diagnosis of alopecia areata (AA).
AA typically is found on the scalp in solitary areas but can affect the whole body, including the eyelashes and eyebrows. Affected hair typically is narrower at the proximal end, resembling an exclamation point, as the hair fails to grow and falls out. Sometimes, patches of alopecia may coalesce into a larger area. Nail changes may be noted, as well. Nails may become brittle, with pitting and/or longitudinal ridges. Patients are usually asymptomatic but may complain of pruritus.
AA is believed to be an autoimmune disorder. It affects males and females of all ages but is more common in children and young adults. AA is believed to result from a T-cell–mediated immune response that transitions the hair follicles from the growth phase to the resting phase. This leads to sudden hair loss and inhibition of regrowth of the hair. However, the hair follicle is not permanently destroyed as in other processes of alopecia. There is also an association between AA and other autoimmune disorders such as vitiligo, thyroid disease, and lupus.
The diagnosis usually is made clinically, as in this patient, but a definitive diagnosis can be made by biopsy and pathology. Other differential diagnoses to consider are trichotillomania, tinea, traumatic alopecia, and lupus.
In almost half of cases, AA is self-resolving; therefore, in first episodes of localized disease, watchful waiting is appropriate. Treatment is tailored to the individual patient and may be difficult. Intralesional corticosteroids often are used for mild cases. Typically, 10 mg/mL of a glucocorticoid is injected into the mid-dermis every 4 to 6 weeks; however, this treatment carries a risk of transient or even permanent atrophy of the injection site. Potent topical corticosteroids often are used, especially in children who do not tolerate intralesional injections, and success can be variable. Other topical treatments to consider are photochemotherapy (psoralen plus UVA), or an irritant agent such as anthralin and a vasodilator such as minoxidil. Regrowth can be expected in a few months to a year, but recurrence is common.
Image courtesy of Stacy Nguy, MD, and text courtesy of Stacy Nguy, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Papadopoulos AJ, Schwartz RA, Janniger C. Alopecia areata: pathogenesis, diagnosis, and therapy. Am J Clin Dermatol. 2000;1:101-105
Papadopoulos AJ, Schwartz RA, Janniger C. Alopecia areata: pathogenesis, diagnosis, and therapy. Am J Clin Dermatol. 2000;1:101-105
Systemic sclerosis patients share their perspectives and needs in treatment trials
Patients with systemic sclerosis have variable disease progression but often experience debilitating fatigue, pain, and digestive issues – and they’re extremely concerned about progressive organ damage, according to those who spoke at and provided input at a public meeting on patient-focused drug development for the disease.
The virtual meeting was part of the Food and Drug Administration’s Patient-Focused Drug Development (PFDD) initiative, which began in 2012 and aims to provide a systematic way for patients’ experiences, needs, and priorities to be “captured and meaningfully incorporated” into drug development and evaluation.
Patients rate their most impactful symptoms
Dinesh Khanna, MBBS, MSc, a rheumatologist who directs a scleroderma research program at the University of Michigan, Ann Arbor, attended the meeting after giving an opening presentation on the disease to FDA officials, patients, and other participants. In a later interview, he said that patients’ ratings of their most impactful symptoms was especially striking.
Raynaud’s phenomenon, digestive symptoms, and fatigue were the top three answers to a poll question that asked patients what symptom had the most significant impact on daily life, he noted, “and none of these are being [strongly] addressed right now [in clinical trials] apart from Raynaud’s phenomenon, for which there are some trials ongoing.”
He and other researchers are “struggling with what outcomes measures to use [in their studies],” said Dr. Khanna, the Frederick G.L. Huetwell Professor of Rheumatology at the University. “My takeaway from the meeting as a clinical trialist is that we should be paying close attention to the symptoms that patients tell us are the most important. We should be including these in our trial designs as secondary endpoints, if not primary endpoints. We have not done that [thus far], really.”
Approximately 200,000 patients in the United States have scleroderma, and approximately 75,000-80,000 of these patients have systemic scleroderma, or systemic sclerosis, Dr. Khanna said in his opening presentation. Each year, he estimates, about 6,000 new diagnoses of systemic sclerosis are made.
More than 200 people – patients, FDA officials, and others – participated in the PFDD meeting. Patients participated in one of two panels – one focused on health effects and daily impacts, and the other on treatments – or submitted input electronically. All were invited to answer poll questions.
Raj Nair, MD, one of eight FDA leaders attending the meeting, noted in closing remarks that the pain experienced by patients with systemic sclerosis includes severe pain from Raynaud’s phenomenon and pain caused by digital ulcers and by calcinosis. “We heard about how paralyzing the pain from calcinosis is, and that there are very few options for alleviating this pain,” said Dr. Nair, of the division of rheumatology and transplant medicine.
Another takeaway, he said, is that the “fatigue can be severe and debilitating, leading to days where it is impossible to get out of bed,” and that digestive symptoms can also be severe. “Reflux,” he noted, “requires significant medical intervention.”
Patients describe their experiences
Rosemary Lyons, diagnosed with scleroderma 35 years ago, explained that while her skin is no longer hardened, she is overly sensitive to fabrics and skin care products and has difficulty with sleeping and eating. She moved away from family in the Northeast to live in the South where the climate is warmer, but even on a 90-degree night she needs a blanket and two comforters to curb the cold and attempt to sleep.
Impaired gastrointestinal motility has made food her “biggest problem” for the past 10 years, and because of GI symptoms, she can eat only one meal a day. She also experiences fainting, brain fog, and severe fatigue. On a good day, Ms. Lyons noted, she sometimes opts to do some house chores “knowing that I’ll have 1-3 days of recovery.”
Another patient, Amy Harding, said that 22 years after her scleroderma diagnosis, “the calcinosis I get in my fingers, elbows, toes, and ears tops all the prior symptoms.” The skin tightening and digital ulcers that she experienced in the first 10 years have tapered off, and while Raynaud’s symptoms and heartburn have worsened, they are at least partly manageable with medications, unlike the pain from calcinosis.
Treating symptoms vs. disease may be key in risk-benefit analysis
In questions after patient presentations, FDA officials probed for more perspective on issues such as how fatigue should be assessed, the differences between fatigue and brain fog, the impact of calcinosis on functioning, and how much risk patients would be willing to assume from treatments that have side effects and that may or may not modulate the disease and slow disease progression.
Most patients said in response to an FDA poll question that they definitely (almost 40%) or possibly (almost 50%) would be willing to try a hypothetical new self-injectable medication if it were shown to reduce their most impactful symptoms but had side effects.
“I think what [we’ve been hearing] today is that whether we’re working on the symptoms or the disease itself is [the key]” to patients’ risk-benefit analysis, said meeting moderator Capt. Robyn Bent, RN, MS, of the U.S. Public Health Service, and director of the PFDD.
Anita Devine, diagnosed 13 years ago with systemic sclerosis, was one of several panel members who said she would accept more bothersome treatment side effects and risks “if the gain was control of disease progression and overall quality of life ... and organ preservation.” Ms. Devine, who has needed kidney dialysis and multiple hand surgeries, noted that she previously took anti-neoplastic and anti-inflammatory agents “to try to stem the course of my disease, but unfortunately the disease did not abate.”
Treatments for systemic sclerosis include vasodilators, immunosuppressive medications, antifibrotic therapies, and stem cell transplants, Dr. Khanna said in his opening remarks.
Trials of drugs for scleroderma have focused on early disease that may be amenable to treatment, with the exception of trials for pulmonary arterial hypertension, which affects some patients with systemic sclerosis. There are multiple FDA-approved drugs for pulmonary arterial hypertension and more trials are underway.
Outcomes such as pain and fatigue are included in many of the trials currently underway, but they tend to be lower-level secondary outcomes measures that cannot be incorporated into drug labeling or are more “exploratory in nature,” Dr. Khanna said in the interview.
Dr. Khanna disclosed that he is the chief medical officer (an equity position) for CiVi Biopharma/Eicos Sciences Inc., which is developing a drug for Raynaud’s, and serves as a consultant and grant recipient for numerous companies that make or are developing drugs for systemic sclerosis.
The FDA will accept patient comments until Dec. 15, 2020, at which time comments will be compiled into a summary report, Ms. Bent said.
Patients with systemic sclerosis have variable disease progression but often experience debilitating fatigue, pain, and digestive issues – and they’re extremely concerned about progressive organ damage, according to those who spoke at and provided input at a public meeting on patient-focused drug development for the disease.
The virtual meeting was part of the Food and Drug Administration’s Patient-Focused Drug Development (PFDD) initiative, which began in 2012 and aims to provide a systematic way for patients’ experiences, needs, and priorities to be “captured and meaningfully incorporated” into drug development and evaluation.
Patients rate their most impactful symptoms
Dinesh Khanna, MBBS, MSc, a rheumatologist who directs a scleroderma research program at the University of Michigan, Ann Arbor, attended the meeting after giving an opening presentation on the disease to FDA officials, patients, and other participants. In a later interview, he said that patients’ ratings of their most impactful symptoms was especially striking.
Raynaud’s phenomenon, digestive symptoms, and fatigue were the top three answers to a poll question that asked patients what symptom had the most significant impact on daily life, he noted, “and none of these are being [strongly] addressed right now [in clinical trials] apart from Raynaud’s phenomenon, for which there are some trials ongoing.”
He and other researchers are “struggling with what outcomes measures to use [in their studies],” said Dr. Khanna, the Frederick G.L. Huetwell Professor of Rheumatology at the University. “My takeaway from the meeting as a clinical trialist is that we should be paying close attention to the symptoms that patients tell us are the most important. We should be including these in our trial designs as secondary endpoints, if not primary endpoints. We have not done that [thus far], really.”
Approximately 200,000 patients in the United States have scleroderma, and approximately 75,000-80,000 of these patients have systemic scleroderma, or systemic sclerosis, Dr. Khanna said in his opening presentation. Each year, he estimates, about 6,000 new diagnoses of systemic sclerosis are made.
More than 200 people – patients, FDA officials, and others – participated in the PFDD meeting. Patients participated in one of two panels – one focused on health effects and daily impacts, and the other on treatments – or submitted input electronically. All were invited to answer poll questions.
Raj Nair, MD, one of eight FDA leaders attending the meeting, noted in closing remarks that the pain experienced by patients with systemic sclerosis includes severe pain from Raynaud’s phenomenon and pain caused by digital ulcers and by calcinosis. “We heard about how paralyzing the pain from calcinosis is, and that there are very few options for alleviating this pain,” said Dr. Nair, of the division of rheumatology and transplant medicine.
Another takeaway, he said, is that the “fatigue can be severe and debilitating, leading to days where it is impossible to get out of bed,” and that digestive symptoms can also be severe. “Reflux,” he noted, “requires significant medical intervention.”
Patients describe their experiences
Rosemary Lyons, diagnosed with scleroderma 35 years ago, explained that while her skin is no longer hardened, she is overly sensitive to fabrics and skin care products and has difficulty with sleeping and eating. She moved away from family in the Northeast to live in the South where the climate is warmer, but even on a 90-degree night she needs a blanket and two comforters to curb the cold and attempt to sleep.
Impaired gastrointestinal motility has made food her “biggest problem” for the past 10 years, and because of GI symptoms, she can eat only one meal a day. She also experiences fainting, brain fog, and severe fatigue. On a good day, Ms. Lyons noted, she sometimes opts to do some house chores “knowing that I’ll have 1-3 days of recovery.”
Another patient, Amy Harding, said that 22 years after her scleroderma diagnosis, “the calcinosis I get in my fingers, elbows, toes, and ears tops all the prior symptoms.” The skin tightening and digital ulcers that she experienced in the first 10 years have tapered off, and while Raynaud’s symptoms and heartburn have worsened, they are at least partly manageable with medications, unlike the pain from calcinosis.
Treating symptoms vs. disease may be key in risk-benefit analysis
In questions after patient presentations, FDA officials probed for more perspective on issues such as how fatigue should be assessed, the differences between fatigue and brain fog, the impact of calcinosis on functioning, and how much risk patients would be willing to assume from treatments that have side effects and that may or may not modulate the disease and slow disease progression.
Most patients said in response to an FDA poll question that they definitely (almost 40%) or possibly (almost 50%) would be willing to try a hypothetical new self-injectable medication if it were shown to reduce their most impactful symptoms but had side effects.
“I think what [we’ve been hearing] today is that whether we’re working on the symptoms or the disease itself is [the key]” to patients’ risk-benefit analysis, said meeting moderator Capt. Robyn Bent, RN, MS, of the U.S. Public Health Service, and director of the PFDD.
Anita Devine, diagnosed 13 years ago with systemic sclerosis, was one of several panel members who said she would accept more bothersome treatment side effects and risks “if the gain was control of disease progression and overall quality of life ... and organ preservation.” Ms. Devine, who has needed kidney dialysis and multiple hand surgeries, noted that she previously took anti-neoplastic and anti-inflammatory agents “to try to stem the course of my disease, but unfortunately the disease did not abate.”
Treatments for systemic sclerosis include vasodilators, immunosuppressive medications, antifibrotic therapies, and stem cell transplants, Dr. Khanna said in his opening remarks.
Trials of drugs for scleroderma have focused on early disease that may be amenable to treatment, with the exception of trials for pulmonary arterial hypertension, which affects some patients with systemic sclerosis. There are multiple FDA-approved drugs for pulmonary arterial hypertension and more trials are underway.
Outcomes such as pain and fatigue are included in many of the trials currently underway, but they tend to be lower-level secondary outcomes measures that cannot be incorporated into drug labeling or are more “exploratory in nature,” Dr. Khanna said in the interview.
Dr. Khanna disclosed that he is the chief medical officer (an equity position) for CiVi Biopharma/Eicos Sciences Inc., which is developing a drug for Raynaud’s, and serves as a consultant and grant recipient for numerous companies that make or are developing drugs for systemic sclerosis.
The FDA will accept patient comments until Dec. 15, 2020, at which time comments will be compiled into a summary report, Ms. Bent said.
Patients with systemic sclerosis have variable disease progression but often experience debilitating fatigue, pain, and digestive issues – and they’re extremely concerned about progressive organ damage, according to those who spoke at and provided input at a public meeting on patient-focused drug development for the disease.
The virtual meeting was part of the Food and Drug Administration’s Patient-Focused Drug Development (PFDD) initiative, which began in 2012 and aims to provide a systematic way for patients’ experiences, needs, and priorities to be “captured and meaningfully incorporated” into drug development and evaluation.
Patients rate their most impactful symptoms
Dinesh Khanna, MBBS, MSc, a rheumatologist who directs a scleroderma research program at the University of Michigan, Ann Arbor, attended the meeting after giving an opening presentation on the disease to FDA officials, patients, and other participants. In a later interview, he said that patients’ ratings of their most impactful symptoms was especially striking.
Raynaud’s phenomenon, digestive symptoms, and fatigue were the top three answers to a poll question that asked patients what symptom had the most significant impact on daily life, he noted, “and none of these are being [strongly] addressed right now [in clinical trials] apart from Raynaud’s phenomenon, for which there are some trials ongoing.”
He and other researchers are “struggling with what outcomes measures to use [in their studies],” said Dr. Khanna, the Frederick G.L. Huetwell Professor of Rheumatology at the University. “My takeaway from the meeting as a clinical trialist is that we should be paying close attention to the symptoms that patients tell us are the most important. We should be including these in our trial designs as secondary endpoints, if not primary endpoints. We have not done that [thus far], really.”
Approximately 200,000 patients in the United States have scleroderma, and approximately 75,000-80,000 of these patients have systemic scleroderma, or systemic sclerosis, Dr. Khanna said in his opening presentation. Each year, he estimates, about 6,000 new diagnoses of systemic sclerosis are made.
More than 200 people – patients, FDA officials, and others – participated in the PFDD meeting. Patients participated in one of two panels – one focused on health effects and daily impacts, and the other on treatments – or submitted input electronically. All were invited to answer poll questions.
Raj Nair, MD, one of eight FDA leaders attending the meeting, noted in closing remarks that the pain experienced by patients with systemic sclerosis includes severe pain from Raynaud’s phenomenon and pain caused by digital ulcers and by calcinosis. “We heard about how paralyzing the pain from calcinosis is, and that there are very few options for alleviating this pain,” said Dr. Nair, of the division of rheumatology and transplant medicine.
Another takeaway, he said, is that the “fatigue can be severe and debilitating, leading to days where it is impossible to get out of bed,” and that digestive symptoms can also be severe. “Reflux,” he noted, “requires significant medical intervention.”
Patients describe their experiences
Rosemary Lyons, diagnosed with scleroderma 35 years ago, explained that while her skin is no longer hardened, she is overly sensitive to fabrics and skin care products and has difficulty with sleeping and eating. She moved away from family in the Northeast to live in the South where the climate is warmer, but even on a 90-degree night she needs a blanket and two comforters to curb the cold and attempt to sleep.
Impaired gastrointestinal motility has made food her “biggest problem” for the past 10 years, and because of GI symptoms, she can eat only one meal a day. She also experiences fainting, brain fog, and severe fatigue. On a good day, Ms. Lyons noted, she sometimes opts to do some house chores “knowing that I’ll have 1-3 days of recovery.”
Another patient, Amy Harding, said that 22 years after her scleroderma diagnosis, “the calcinosis I get in my fingers, elbows, toes, and ears tops all the prior symptoms.” The skin tightening and digital ulcers that she experienced in the first 10 years have tapered off, and while Raynaud’s symptoms and heartburn have worsened, they are at least partly manageable with medications, unlike the pain from calcinosis.
Treating symptoms vs. disease may be key in risk-benefit analysis
In questions after patient presentations, FDA officials probed for more perspective on issues such as how fatigue should be assessed, the differences between fatigue and brain fog, the impact of calcinosis on functioning, and how much risk patients would be willing to assume from treatments that have side effects and that may or may not modulate the disease and slow disease progression.
Most patients said in response to an FDA poll question that they definitely (almost 40%) or possibly (almost 50%) would be willing to try a hypothetical new self-injectable medication if it were shown to reduce their most impactful symptoms but had side effects.
“I think what [we’ve been hearing] today is that whether we’re working on the symptoms or the disease itself is [the key]” to patients’ risk-benefit analysis, said meeting moderator Capt. Robyn Bent, RN, MS, of the U.S. Public Health Service, and director of the PFDD.
Anita Devine, diagnosed 13 years ago with systemic sclerosis, was one of several panel members who said she would accept more bothersome treatment side effects and risks “if the gain was control of disease progression and overall quality of life ... and organ preservation.” Ms. Devine, who has needed kidney dialysis and multiple hand surgeries, noted that she previously took anti-neoplastic and anti-inflammatory agents “to try to stem the course of my disease, but unfortunately the disease did not abate.”
Treatments for systemic sclerosis include vasodilators, immunosuppressive medications, antifibrotic therapies, and stem cell transplants, Dr. Khanna said in his opening remarks.
Trials of drugs for scleroderma have focused on early disease that may be amenable to treatment, with the exception of trials for pulmonary arterial hypertension, which affects some patients with systemic sclerosis. There are multiple FDA-approved drugs for pulmonary arterial hypertension and more trials are underway.
Outcomes such as pain and fatigue are included in many of the trials currently underway, but they tend to be lower-level secondary outcomes measures that cannot be incorporated into drug labeling or are more “exploratory in nature,” Dr. Khanna said in the interview.
Dr. Khanna disclosed that he is the chief medical officer (an equity position) for CiVi Biopharma/Eicos Sciences Inc., which is developing a drug for Raynaud’s, and serves as a consultant and grant recipient for numerous companies that make or are developing drugs for systemic sclerosis.
The FDA will accept patient comments until Dec. 15, 2020, at which time comments will be compiled into a summary report, Ms. Bent said.
FROM AN FDA PATIENT-FOCUSED DRUG DEVELOPMENT MEETING