User login
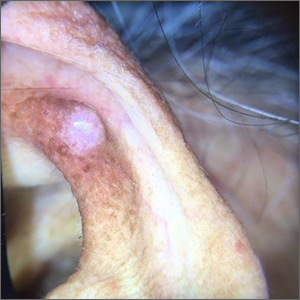
Ear growth
A shave biopsy of the lesion was performed and it confirmed the diagnosis of chondrodermatitis nodularis helicis (CNH).
CNH is an inflammatory process that most commonly occurs on the helix of the ear but can also occur on the antihelix and, rarely, on other areas of the ear. It generally manifests as a firm nodule with surrounding erythema that may be painful only when pressure is applied. Patients may describe bleeding, ulceration, and exudate. They will usually report discomfort from sleeping on the affected side.
The pathogenesis of CNH is poorly understood but is thought to be related to vasculitis and inflammation from prolonged pressure to the affected ear during sleep or from devices that are worn in or around the ear (eg, hearing aids, headphones). Other factors such as actinic damage or ear trauma have also been described. Histopathologic studies have identified arteriolar narrowing with ischemic changes and necrosis of cartilage causing localized inflammation.1
The differential diagnosis for this lesion includes nonmelanoma skin cancer, as well as tophaceous gout and seborrheic keratosis.
There are multiple conservative treatment options. One option is to relieve pressure by sleeping on the unaffected side or using commercially available pillows with a cutout or window where the affected ear can rest. Pharmacologic treatments include topical nitroglycerin1 and intralesional collagen or corticosteroid injections. If previous treatments are unsuccessful, consider surgical excision of the affected tissue and curettage of the underlying abnormal cartilage. Recurrence is possible with both conservative and surgical treatment.
This patient was counseled on the benign nature of her biopsy findings and treatment options were discussed. She elected to proceed with pressure-relieving measures when sleeping and planned to follow up if there was no improvement.
Image courtesy of Marion Cook, MD, First Choice Community Healthcare, Albuquerque, New Mexico. Text courtesy of Spenser Squire, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Nielsen LJ, Olsen CH, Lock-Anderson J. Therapeutic options of chondrodermatitis nodularis helicis. Plast Surg Int. 2016;2016:4340168. doi: 10.1155/2016/4340168

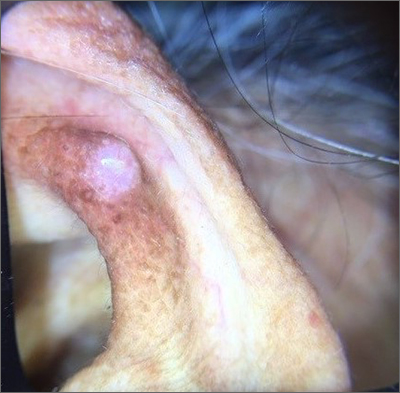
A shave biopsy of the lesion was performed and it confirmed the diagnosis of chondrodermatitis nodularis helicis (CNH).
CNH is an inflammatory process that most commonly occurs on the helix of the ear but can also occur on the antihelix and, rarely, on other areas of the ear. It generally manifests as a firm nodule with surrounding erythema that may be painful only when pressure is applied. Patients may describe bleeding, ulceration, and exudate. They will usually report discomfort from sleeping on the affected side.
The pathogenesis of CNH is poorly understood but is thought to be related to vasculitis and inflammation from prolonged pressure to the affected ear during sleep or from devices that are worn in or around the ear (eg, hearing aids, headphones). Other factors such as actinic damage or ear trauma have also been described. Histopathologic studies have identified arteriolar narrowing with ischemic changes and necrosis of cartilage causing localized inflammation.1
The differential diagnosis for this lesion includes nonmelanoma skin cancer, as well as tophaceous gout and seborrheic keratosis.
There are multiple conservative treatment options. One option is to relieve pressure by sleeping on the unaffected side or using commercially available pillows with a cutout or window where the affected ear can rest. Pharmacologic treatments include topical nitroglycerin1 and intralesional collagen or corticosteroid injections. If previous treatments are unsuccessful, consider surgical excision of the affected tissue and curettage of the underlying abnormal cartilage. Recurrence is possible with both conservative and surgical treatment.
This patient was counseled on the benign nature of her biopsy findings and treatment options were discussed. She elected to proceed with pressure-relieving measures when sleeping and planned to follow up if there was no improvement.
Image courtesy of Marion Cook, MD, First Choice Community Healthcare, Albuquerque, New Mexico. Text courtesy of Spenser Squire, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

A shave biopsy of the lesion was performed and it confirmed the diagnosis of chondrodermatitis nodularis helicis (CNH).
CNH is an inflammatory process that most commonly occurs on the helix of the ear but can also occur on the antihelix and, rarely, on other areas of the ear. It generally manifests as a firm nodule with surrounding erythema that may be painful only when pressure is applied. Patients may describe bleeding, ulceration, and exudate. They will usually report discomfort from sleeping on the affected side.
The pathogenesis of CNH is poorly understood but is thought to be related to vasculitis and inflammation from prolonged pressure to the affected ear during sleep or from devices that are worn in or around the ear (eg, hearing aids, headphones). Other factors such as actinic damage or ear trauma have also been described. Histopathologic studies have identified arteriolar narrowing with ischemic changes and necrosis of cartilage causing localized inflammation.1
The differential diagnosis for this lesion includes nonmelanoma skin cancer, as well as tophaceous gout and seborrheic keratosis.
There are multiple conservative treatment options. One option is to relieve pressure by sleeping on the unaffected side or using commercially available pillows with a cutout or window where the affected ear can rest. Pharmacologic treatments include topical nitroglycerin1 and intralesional collagen or corticosteroid injections. If previous treatments are unsuccessful, consider surgical excision of the affected tissue and curettage of the underlying abnormal cartilage. Recurrence is possible with both conservative and surgical treatment.
This patient was counseled on the benign nature of her biopsy findings and treatment options were discussed. She elected to proceed with pressure-relieving measures when sleeping and planned to follow up if there was no improvement.
Image courtesy of Marion Cook, MD, First Choice Community Healthcare, Albuquerque, New Mexico. Text courtesy of Spenser Squire, MD, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Nielsen LJ, Olsen CH, Lock-Anderson J. Therapeutic options of chondrodermatitis nodularis helicis. Plast Surg Int. 2016;2016:4340168. doi: 10.1155/2016/4340168
1. Nielsen LJ, Olsen CH, Lock-Anderson J. Therapeutic options of chondrodermatitis nodularis helicis. Plast Surg Int. 2016;2016:4340168. doi: 10.1155/2016/4340168
Cellulitis care costly from misdiagnosis, needless hospitalizations
BOSTON – The cost of care for the more than 14 million cases of cellulitis that occur each year in the United States is in the billions of dollars, but there are multiple opportunities, many involving dermatologists, to dramatically reduce these costs, according to an outline of strategies presented at the American Academy of Dermatology 2022 annual meeting in Boston.
“Cellulitis is misdiagnosed about one-third of the time, and that cost is very high,” reported Jennifer L. Adams, MD, assistant professor of dermatology, University of Nebraska, Omaha. She sees opportunities for dermatological consults to help weed through the many cellulitis mimickers, such as venous insufficiency or psoriasiform drug reactions, to prevent unnecessary admissions and ineffective therapy.
“There is a huge need for diagnostic accuracy as a means to deliver more cost-effective care,” Dr. Adams said.
Solving misdiagnosis is only part of the story. Costs of care are also ramped up by unnecessary hospitalizations. According to Dr. Adams, published criteria to triage emergency room patients with cellulitis to outpatient care are not always followed. In one review, 14% of admitted patients had met the criteria for outpatient treatment.
Cellulitis is a common skin infection that causes redness, swelling, and pain in the infected area, most often on the legs and feet.
Unnecessary hospitalizations for misdiagnosed cellulitis, which is associated with an average 4-day hospital stay, “range from $200 million to $500 million in avoidable direct healthcare costs,” Dr. Adams said.
Even for justifiable hospitalizations, there are still opportunities for cost savings. In one study, blood cultures were ordered in 73% of patients even though only 2% produced a finding relevant to care. According to Dr. Adams, most cellulitis cases are caused by the “usual suspects” – group A beta-hemolytic streptococcus, Streptococcus pneumoniae, and Staphylococcus aureus. The exceptions stand out by clinical criteria, such as known neutropenia, history of an animal bite, signs of Systemic Inflammatory Response Syndrome (SIRS), or a purulent appearance.
“Blood cultures are not cost-effective in uncomplicated cellulitis,” Dr. Adams said. She said there are numerous published algorithms to guide clinicians on decision-making in the management of soft tissue infections, including cellulitis, including a much-cited algorithm first published more than 15 years ago and updated in 2014.
Similarly, labs and imaging are commonly ordered with no strong likelihood that they will change management, she said. These types of decisions are also covered in published algorithms.
Strategies to prevent rehospitalization are another area where there is a large opportunity to reduce health care resources consumed by cellulitis. The rehospitalization rate at 30 days is approximately 10%, but many patients have recurrent episodes over years, according to Dr. Adams. The risk factors and the preventative measures have been well described.
“Scrupulous clinical care can reduce recurrence, and it is cost-effective,” said Dr. Adams, referring to control of edema, control of underlying conditions associated with increased risk, such as diabetes, and managing dry skin and erosions with topical agents or even moisturizers. Compression socks are a simple but effective tool, she added.
For patients with repeat episodes of cellulitis over years, Dr. Adams referred to a double-blind trial that associated a twice-daily dose of 250 mg penicillin with a 45% reduction in the risk of cellulitis recurrence over 1 year. At approximately $10 a month for this treatment, she said it is very cost-effective, although she acknowledged that recurrence rates of cellulitis climb back up when the penicillin is stopped.
“I think of this as a bridge while you work on addressing the venous insufficiency or other risk factors for cellulitis,” Dr. Adams said.
For reducing the costs of cellulitis, there is evidence that dermatologists can play a role. Dr. Adams cited a study that evaluated the impact of a dermatologist consultation for suspected cellulitis in the emergency room or within 24 hours of admission. Of 34 patients already prescribed antibiotics for presumed cellulitis, discontinuation was recommended in 82%. Of 39 admissions, pseudocellulitis was identified in 51%.
Extrapolating these data to national rates of cellulitis, there was an estimated savings of up to $200 million annually without any apparent increased risk of adverse outcomes, according to Dr. Adams.
When contacted about his experience, the senior investigator of that study, Arash Mostaghimi, MD, director of the Inpatient Dermatology Consult Service, Brigham and Women’s Hospital, Boston, largely agreed with the premise of Adam’s analysis. In particular, he said, avoiding misdiagnosis of cellulitis offers a major opportunity to lower costs while possibly improving care.
True of national practice and at the local level, “misdiagnosis of noninfectious inflammatory reactions such as cellulitis has substantial cost impacts,” Dr. Mostaghimi said in an interview. Based on evidence, the savings are derived directly from “unnecessary antibiotic exposure as well as inappropriate hospitalization.”
Following publication of his study, he became involved in addressing this issue at his institution.
“At Brigham and Women’s, we collaborated with colleagues in infectious disease and in the emergency department to create cellulitis protocols that identify patients at risk for misdiagnosis and facilitate early dermatology consultation for diagnostic confirmation,” he said.
Although there are algorithms to achieve this goal, he indicated that the expertise of dermatologists can quickly and efficiently differentiate inflammatory skin reactions and expedite appropriate care.
Dr. Adams and Dr. Mostaghimi have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
BOSTON – The cost of care for the more than 14 million cases of cellulitis that occur each year in the United States is in the billions of dollars, but there are multiple opportunities, many involving dermatologists, to dramatically reduce these costs, according to an outline of strategies presented at the American Academy of Dermatology 2022 annual meeting in Boston.
“Cellulitis is misdiagnosed about one-third of the time, and that cost is very high,” reported Jennifer L. Adams, MD, assistant professor of dermatology, University of Nebraska, Omaha. She sees opportunities for dermatological consults to help weed through the many cellulitis mimickers, such as venous insufficiency or psoriasiform drug reactions, to prevent unnecessary admissions and ineffective therapy.
“There is a huge need for diagnostic accuracy as a means to deliver more cost-effective care,” Dr. Adams said.
Solving misdiagnosis is only part of the story. Costs of care are also ramped up by unnecessary hospitalizations. According to Dr. Adams, published criteria to triage emergency room patients with cellulitis to outpatient care are not always followed. In one review, 14% of admitted patients had met the criteria for outpatient treatment.
Cellulitis is a common skin infection that causes redness, swelling, and pain in the infected area, most often on the legs and feet.
Unnecessary hospitalizations for misdiagnosed cellulitis, which is associated with an average 4-day hospital stay, “range from $200 million to $500 million in avoidable direct healthcare costs,” Dr. Adams said.
Even for justifiable hospitalizations, there are still opportunities for cost savings. In one study, blood cultures were ordered in 73% of patients even though only 2% produced a finding relevant to care. According to Dr. Adams, most cellulitis cases are caused by the “usual suspects” – group A beta-hemolytic streptococcus, Streptococcus pneumoniae, and Staphylococcus aureus. The exceptions stand out by clinical criteria, such as known neutropenia, history of an animal bite, signs of Systemic Inflammatory Response Syndrome (SIRS), or a purulent appearance.
“Blood cultures are not cost-effective in uncomplicated cellulitis,” Dr. Adams said. She said there are numerous published algorithms to guide clinicians on decision-making in the management of soft tissue infections, including cellulitis, including a much-cited algorithm first published more than 15 years ago and updated in 2014.
Similarly, labs and imaging are commonly ordered with no strong likelihood that they will change management, she said. These types of decisions are also covered in published algorithms.
Strategies to prevent rehospitalization are another area where there is a large opportunity to reduce health care resources consumed by cellulitis. The rehospitalization rate at 30 days is approximately 10%, but many patients have recurrent episodes over years, according to Dr. Adams. The risk factors and the preventative measures have been well described.
“Scrupulous clinical care can reduce recurrence, and it is cost-effective,” said Dr. Adams, referring to control of edema, control of underlying conditions associated with increased risk, such as diabetes, and managing dry skin and erosions with topical agents or even moisturizers. Compression socks are a simple but effective tool, she added.
For patients with repeat episodes of cellulitis over years, Dr. Adams referred to a double-blind trial that associated a twice-daily dose of 250 mg penicillin with a 45% reduction in the risk of cellulitis recurrence over 1 year. At approximately $10 a month for this treatment, she said it is very cost-effective, although she acknowledged that recurrence rates of cellulitis climb back up when the penicillin is stopped.
“I think of this as a bridge while you work on addressing the venous insufficiency or other risk factors for cellulitis,” Dr. Adams said.
For reducing the costs of cellulitis, there is evidence that dermatologists can play a role. Dr. Adams cited a study that evaluated the impact of a dermatologist consultation for suspected cellulitis in the emergency room or within 24 hours of admission. Of 34 patients already prescribed antibiotics for presumed cellulitis, discontinuation was recommended in 82%. Of 39 admissions, pseudocellulitis was identified in 51%.
Extrapolating these data to national rates of cellulitis, there was an estimated savings of up to $200 million annually without any apparent increased risk of adverse outcomes, according to Dr. Adams.
When contacted about his experience, the senior investigator of that study, Arash Mostaghimi, MD, director of the Inpatient Dermatology Consult Service, Brigham and Women’s Hospital, Boston, largely agreed with the premise of Adam’s analysis. In particular, he said, avoiding misdiagnosis of cellulitis offers a major opportunity to lower costs while possibly improving care.
True of national practice and at the local level, “misdiagnosis of noninfectious inflammatory reactions such as cellulitis has substantial cost impacts,” Dr. Mostaghimi said in an interview. Based on evidence, the savings are derived directly from “unnecessary antibiotic exposure as well as inappropriate hospitalization.”
Following publication of his study, he became involved in addressing this issue at his institution.
“At Brigham and Women’s, we collaborated with colleagues in infectious disease and in the emergency department to create cellulitis protocols that identify patients at risk for misdiagnosis and facilitate early dermatology consultation for diagnostic confirmation,” he said.
Although there are algorithms to achieve this goal, he indicated that the expertise of dermatologists can quickly and efficiently differentiate inflammatory skin reactions and expedite appropriate care.
Dr. Adams and Dr. Mostaghimi have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
BOSTON – The cost of care for the more than 14 million cases of cellulitis that occur each year in the United States is in the billions of dollars, but there are multiple opportunities, many involving dermatologists, to dramatically reduce these costs, according to an outline of strategies presented at the American Academy of Dermatology 2022 annual meeting in Boston.
“Cellulitis is misdiagnosed about one-third of the time, and that cost is very high,” reported Jennifer L. Adams, MD, assistant professor of dermatology, University of Nebraska, Omaha. She sees opportunities for dermatological consults to help weed through the many cellulitis mimickers, such as venous insufficiency or psoriasiform drug reactions, to prevent unnecessary admissions and ineffective therapy.
“There is a huge need for diagnostic accuracy as a means to deliver more cost-effective care,” Dr. Adams said.
Solving misdiagnosis is only part of the story. Costs of care are also ramped up by unnecessary hospitalizations. According to Dr. Adams, published criteria to triage emergency room patients with cellulitis to outpatient care are not always followed. In one review, 14% of admitted patients had met the criteria for outpatient treatment.
Cellulitis is a common skin infection that causes redness, swelling, and pain in the infected area, most often on the legs and feet.
Unnecessary hospitalizations for misdiagnosed cellulitis, which is associated with an average 4-day hospital stay, “range from $200 million to $500 million in avoidable direct healthcare costs,” Dr. Adams said.
Even for justifiable hospitalizations, there are still opportunities for cost savings. In one study, blood cultures were ordered in 73% of patients even though only 2% produced a finding relevant to care. According to Dr. Adams, most cellulitis cases are caused by the “usual suspects” – group A beta-hemolytic streptococcus, Streptococcus pneumoniae, and Staphylococcus aureus. The exceptions stand out by clinical criteria, such as known neutropenia, history of an animal bite, signs of Systemic Inflammatory Response Syndrome (SIRS), or a purulent appearance.
“Blood cultures are not cost-effective in uncomplicated cellulitis,” Dr. Adams said. She said there are numerous published algorithms to guide clinicians on decision-making in the management of soft tissue infections, including cellulitis, including a much-cited algorithm first published more than 15 years ago and updated in 2014.
Similarly, labs and imaging are commonly ordered with no strong likelihood that they will change management, she said. These types of decisions are also covered in published algorithms.
Strategies to prevent rehospitalization are another area where there is a large opportunity to reduce health care resources consumed by cellulitis. The rehospitalization rate at 30 days is approximately 10%, but many patients have recurrent episodes over years, according to Dr. Adams. The risk factors and the preventative measures have been well described.
“Scrupulous clinical care can reduce recurrence, and it is cost-effective,” said Dr. Adams, referring to control of edema, control of underlying conditions associated with increased risk, such as diabetes, and managing dry skin and erosions with topical agents or even moisturizers. Compression socks are a simple but effective tool, she added.
For patients with repeat episodes of cellulitis over years, Dr. Adams referred to a double-blind trial that associated a twice-daily dose of 250 mg penicillin with a 45% reduction in the risk of cellulitis recurrence over 1 year. At approximately $10 a month for this treatment, she said it is very cost-effective, although she acknowledged that recurrence rates of cellulitis climb back up when the penicillin is stopped.
“I think of this as a bridge while you work on addressing the venous insufficiency or other risk factors for cellulitis,” Dr. Adams said.
For reducing the costs of cellulitis, there is evidence that dermatologists can play a role. Dr. Adams cited a study that evaluated the impact of a dermatologist consultation for suspected cellulitis in the emergency room or within 24 hours of admission. Of 34 patients already prescribed antibiotics for presumed cellulitis, discontinuation was recommended in 82%. Of 39 admissions, pseudocellulitis was identified in 51%.
Extrapolating these data to national rates of cellulitis, there was an estimated savings of up to $200 million annually without any apparent increased risk of adverse outcomes, according to Dr. Adams.
When contacted about his experience, the senior investigator of that study, Arash Mostaghimi, MD, director of the Inpatient Dermatology Consult Service, Brigham and Women’s Hospital, Boston, largely agreed with the premise of Adam’s analysis. In particular, he said, avoiding misdiagnosis of cellulitis offers a major opportunity to lower costs while possibly improving care.
True of national practice and at the local level, “misdiagnosis of noninfectious inflammatory reactions such as cellulitis has substantial cost impacts,” Dr. Mostaghimi said in an interview. Based on evidence, the savings are derived directly from “unnecessary antibiotic exposure as well as inappropriate hospitalization.”
Following publication of his study, he became involved in addressing this issue at his institution.
“At Brigham and Women’s, we collaborated with colleagues in infectious disease and in the emergency department to create cellulitis protocols that identify patients at risk for misdiagnosis and facilitate early dermatology consultation for diagnostic confirmation,” he said.
Although there are algorithms to achieve this goal, he indicated that the expertise of dermatologists can quickly and efficiently differentiate inflammatory skin reactions and expedite appropriate care.
Dr. Adams and Dr. Mostaghimi have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT AAD 2022
Global registry tracks COVID-19 outcomes in atopic dermatitis patients
BOSTON – , results from a global registry demonstrated.
Moreover, combination systemic treatment, especially those that included systemic corticosteroids, was associated with the highest risk of COVID-19–related hospitalization.
“Patients with inflammatory skin diseases such as AD may be at higher risk of COVID-19,” Annelie H. Musters, MD, said during a late-breaking abstract session at the annual meeting of the American Academy of Dermatology. “Another factor to consider is that AD patients are often treated with systemic immunomodulatory therapy, including systemic corticosteroids and nonsteroidal immunosuppressants such as methotrexate, cyclosporin, biologics, and Janus kinase inhibitors. Different mechanisms of action and levels of immunosuppression may impart variable risks of serious infections.”
On the other hand, some degree of immunomodulation may have beneficial effects on the course of COVID-19 in AD patients, said Dr. Musters, of the department of dermatology at Academic Medical Center, University of Amsterdam. Targeting of specific immune pathways could reduce the development of a hyperinflammatory state in severe COVID-19. Dual blockade of interleukin (IL)-4 and IL-13 with dupilumab may have a protective effect in the context of COVID-19 infection, because expression of Th2 cytokines, including IL-4 and IL-13, may be increased during COVID-19.
“At the start of the pandemic, many of us were faced with important questions, like do systemic immunomodulatory treatments influence outcomes of COVID-19 in patients with AD?” she said. “Do patients on dupilumab or other novel systemics fare better than those on conventional systemic treatment?”
To answer these questions, she and her colleagues launched a web-based registry in April 2020 to investigate COVID-19 outcomes in patients with AD treated with or without systemic immunomodulatory treatments. For the registry, known as Surveillance Epidemiology of Coronavirus Under Research Exclusion for Atopic Dermatitis (SECURE-AD), clinicians in 27 countries used a web-based form to enter anonymized data after patients had fully recovered from COVID-19. Eligibility criteria included having proven or highly suspected COVID-19, and there were no restrictions on age nor the type of AD treatment they were receiving.
Dr. Musters reported results from 442 patients who were recruited between April 2, 2020, and Oct. 31, 2021. Their mean age was 35.6 years, their median body mass index was 23.7 kg/m2, and there was an even sex distribution. Most patients were White and were recruited from Italy. Of the 442 patients, 216 (48.8%) received dupilumab monotherapy, 131 (29.6%) received topical treatments, and 14 (3.16%) received combination systemic treatments, including systemic corticosteroids. About 12% presented to the emergency department and 6% were hospitalized. Of those hospitalized, 2% required intensive care and/or ventilation, and no deaths have occurred in the registry to date.
By treatment group, hospitalization rates were highest among those on combination treatments (35.7%), followed by systemic corticosteroids (14.3%), topical treatments only (9.9%), other conventional systemics (3.6%), methotrexate (3.3%), and dupilumab (2.3%).
To further explore the differences between hospitalization rates in treatment groups, the researchers performed a multivariable logistic regression analysis, adjusted for age, sex, ethnicity, and comorbidity score. Compared with those who received dupilumab, the adjusted odds ratios (ORs) for hospitalization were highest among those who received topical treatments (OR, 4.95), followed by those who received systemic corticosteroids (OR, 2.81), and those who received other conventional systemic treatments (OR, 2.36).
Dr. Musters and colleagues also found that compared with patients on nonsteroidal immunosuppressive therapy, patients on combination systemic therapy had a significantly higher odds of hospitalization, specifically an OR of 45.75 for those on combination treatment including corticosteroids, an OR of 37.57 for those on combination treatment not including steroids, and an OR of 1.87 for those on systemic corticosteroids as monotherapy.
“Overall, the risk of COVID-19 complications appears to be low in patients with AD, even when treated with systemic immunomodulatory agents,” Dr. Musters concluded. “Dupilumab monotherapy was associated with lower odds of hospitalizations compared with other therapies. Moreover, combination systemic treatment, especially combinations including systemic corticosteroids, was associated with the highest risk of severe COVID-19.”
She added that other population-based study designs are more suitable to answer other important questions, such as whether the overall risk of COVID-19 in patients with AD is higher or lower compared to healthy controls.
Amy S. Paller, MD, professor and chair of the department of dermatology at Northwestern University, Chicago, who was asked to comment on the study, characterized the results as reassuring. In this patient population, “we expected that dupilumab would not cause any problems,” she said. “We wouldn’t necessarily expect it to [confer] a benefit, but I think it’s because the patients who need a systemic medication are going on something that’s very targeted (dupilumab) rather than something that has a broader immunosuppressing function. It was interesting but not surprising that those on systemic steroids had more of a problem. Get them on something that’s very targeted if you can and don’t suppress the immune systems that might be handling COVID-19.”
Dr. Musters reported having no disclosures. Dr. Paller disclosed that she is consultant to and/or an investigator for many pharmaceutical companies.
BOSTON – , results from a global registry demonstrated.
Moreover, combination systemic treatment, especially those that included systemic corticosteroids, was associated with the highest risk of COVID-19–related hospitalization.
“Patients with inflammatory skin diseases such as AD may be at higher risk of COVID-19,” Annelie H. Musters, MD, said during a late-breaking abstract session at the annual meeting of the American Academy of Dermatology. “Another factor to consider is that AD patients are often treated with systemic immunomodulatory therapy, including systemic corticosteroids and nonsteroidal immunosuppressants such as methotrexate, cyclosporin, biologics, and Janus kinase inhibitors. Different mechanisms of action and levels of immunosuppression may impart variable risks of serious infections.”
On the other hand, some degree of immunomodulation may have beneficial effects on the course of COVID-19 in AD patients, said Dr. Musters, of the department of dermatology at Academic Medical Center, University of Amsterdam. Targeting of specific immune pathways could reduce the development of a hyperinflammatory state in severe COVID-19. Dual blockade of interleukin (IL)-4 and IL-13 with dupilumab may have a protective effect in the context of COVID-19 infection, because expression of Th2 cytokines, including IL-4 and IL-13, may be increased during COVID-19.
“At the start of the pandemic, many of us were faced with important questions, like do systemic immunomodulatory treatments influence outcomes of COVID-19 in patients with AD?” she said. “Do patients on dupilumab or other novel systemics fare better than those on conventional systemic treatment?”
To answer these questions, she and her colleagues launched a web-based registry in April 2020 to investigate COVID-19 outcomes in patients with AD treated with or without systemic immunomodulatory treatments. For the registry, known as Surveillance Epidemiology of Coronavirus Under Research Exclusion for Atopic Dermatitis (SECURE-AD), clinicians in 27 countries used a web-based form to enter anonymized data after patients had fully recovered from COVID-19. Eligibility criteria included having proven or highly suspected COVID-19, and there were no restrictions on age nor the type of AD treatment they were receiving.
Dr. Musters reported results from 442 patients who were recruited between April 2, 2020, and Oct. 31, 2021. Their mean age was 35.6 years, their median body mass index was 23.7 kg/m2, and there was an even sex distribution. Most patients were White and were recruited from Italy. Of the 442 patients, 216 (48.8%) received dupilumab monotherapy, 131 (29.6%) received topical treatments, and 14 (3.16%) received combination systemic treatments, including systemic corticosteroids. About 12% presented to the emergency department and 6% were hospitalized. Of those hospitalized, 2% required intensive care and/or ventilation, and no deaths have occurred in the registry to date.
By treatment group, hospitalization rates were highest among those on combination treatments (35.7%), followed by systemic corticosteroids (14.3%), topical treatments only (9.9%), other conventional systemics (3.6%), methotrexate (3.3%), and dupilumab (2.3%).
To further explore the differences between hospitalization rates in treatment groups, the researchers performed a multivariable logistic regression analysis, adjusted for age, sex, ethnicity, and comorbidity score. Compared with those who received dupilumab, the adjusted odds ratios (ORs) for hospitalization were highest among those who received topical treatments (OR, 4.95), followed by those who received systemic corticosteroids (OR, 2.81), and those who received other conventional systemic treatments (OR, 2.36).
Dr. Musters and colleagues also found that compared with patients on nonsteroidal immunosuppressive therapy, patients on combination systemic therapy had a significantly higher odds of hospitalization, specifically an OR of 45.75 for those on combination treatment including corticosteroids, an OR of 37.57 for those on combination treatment not including steroids, and an OR of 1.87 for those on systemic corticosteroids as monotherapy.
“Overall, the risk of COVID-19 complications appears to be low in patients with AD, even when treated with systemic immunomodulatory agents,” Dr. Musters concluded. “Dupilumab monotherapy was associated with lower odds of hospitalizations compared with other therapies. Moreover, combination systemic treatment, especially combinations including systemic corticosteroids, was associated with the highest risk of severe COVID-19.”
She added that other population-based study designs are more suitable to answer other important questions, such as whether the overall risk of COVID-19 in patients with AD is higher or lower compared to healthy controls.
Amy S. Paller, MD, professor and chair of the department of dermatology at Northwestern University, Chicago, who was asked to comment on the study, characterized the results as reassuring. In this patient population, “we expected that dupilumab would not cause any problems,” she said. “We wouldn’t necessarily expect it to [confer] a benefit, but I think it’s because the patients who need a systemic medication are going on something that’s very targeted (dupilumab) rather than something that has a broader immunosuppressing function. It was interesting but not surprising that those on systemic steroids had more of a problem. Get them on something that’s very targeted if you can and don’t suppress the immune systems that might be handling COVID-19.”
Dr. Musters reported having no disclosures. Dr. Paller disclosed that she is consultant to and/or an investigator for many pharmaceutical companies.
BOSTON – , results from a global registry demonstrated.
Moreover, combination systemic treatment, especially those that included systemic corticosteroids, was associated with the highest risk of COVID-19–related hospitalization.
“Patients with inflammatory skin diseases such as AD may be at higher risk of COVID-19,” Annelie H. Musters, MD, said during a late-breaking abstract session at the annual meeting of the American Academy of Dermatology. “Another factor to consider is that AD patients are often treated with systemic immunomodulatory therapy, including systemic corticosteroids and nonsteroidal immunosuppressants such as methotrexate, cyclosporin, biologics, and Janus kinase inhibitors. Different mechanisms of action and levels of immunosuppression may impart variable risks of serious infections.”
On the other hand, some degree of immunomodulation may have beneficial effects on the course of COVID-19 in AD patients, said Dr. Musters, of the department of dermatology at Academic Medical Center, University of Amsterdam. Targeting of specific immune pathways could reduce the development of a hyperinflammatory state in severe COVID-19. Dual blockade of interleukin (IL)-4 and IL-13 with dupilumab may have a protective effect in the context of COVID-19 infection, because expression of Th2 cytokines, including IL-4 and IL-13, may be increased during COVID-19.
“At the start of the pandemic, many of us were faced with important questions, like do systemic immunomodulatory treatments influence outcomes of COVID-19 in patients with AD?” she said. “Do patients on dupilumab or other novel systemics fare better than those on conventional systemic treatment?”
To answer these questions, she and her colleagues launched a web-based registry in April 2020 to investigate COVID-19 outcomes in patients with AD treated with or without systemic immunomodulatory treatments. For the registry, known as Surveillance Epidemiology of Coronavirus Under Research Exclusion for Atopic Dermatitis (SECURE-AD), clinicians in 27 countries used a web-based form to enter anonymized data after patients had fully recovered from COVID-19. Eligibility criteria included having proven or highly suspected COVID-19, and there were no restrictions on age nor the type of AD treatment they were receiving.
Dr. Musters reported results from 442 patients who were recruited between April 2, 2020, and Oct. 31, 2021. Their mean age was 35.6 years, their median body mass index was 23.7 kg/m2, and there was an even sex distribution. Most patients were White and were recruited from Italy. Of the 442 patients, 216 (48.8%) received dupilumab monotherapy, 131 (29.6%) received topical treatments, and 14 (3.16%) received combination systemic treatments, including systemic corticosteroids. About 12% presented to the emergency department and 6% were hospitalized. Of those hospitalized, 2% required intensive care and/or ventilation, and no deaths have occurred in the registry to date.
By treatment group, hospitalization rates were highest among those on combination treatments (35.7%), followed by systemic corticosteroids (14.3%), topical treatments only (9.9%), other conventional systemics (3.6%), methotrexate (3.3%), and dupilumab (2.3%).
To further explore the differences between hospitalization rates in treatment groups, the researchers performed a multivariable logistic regression analysis, adjusted for age, sex, ethnicity, and comorbidity score. Compared with those who received dupilumab, the adjusted odds ratios (ORs) for hospitalization were highest among those who received topical treatments (OR, 4.95), followed by those who received systemic corticosteroids (OR, 2.81), and those who received other conventional systemic treatments (OR, 2.36).
Dr. Musters and colleagues also found that compared with patients on nonsteroidal immunosuppressive therapy, patients on combination systemic therapy had a significantly higher odds of hospitalization, specifically an OR of 45.75 for those on combination treatment including corticosteroids, an OR of 37.57 for those on combination treatment not including steroids, and an OR of 1.87 for those on systemic corticosteroids as monotherapy.
“Overall, the risk of COVID-19 complications appears to be low in patients with AD, even when treated with systemic immunomodulatory agents,” Dr. Musters concluded. “Dupilumab monotherapy was associated with lower odds of hospitalizations compared with other therapies. Moreover, combination systemic treatment, especially combinations including systemic corticosteroids, was associated with the highest risk of severe COVID-19.”
She added that other population-based study designs are more suitable to answer other important questions, such as whether the overall risk of COVID-19 in patients with AD is higher or lower compared to healthy controls.
Amy S. Paller, MD, professor and chair of the department of dermatology at Northwestern University, Chicago, who was asked to comment on the study, characterized the results as reassuring. In this patient population, “we expected that dupilumab would not cause any problems,” she said. “We wouldn’t necessarily expect it to [confer] a benefit, but I think it’s because the patients who need a systemic medication are going on something that’s very targeted (dupilumab) rather than something that has a broader immunosuppressing function. It was interesting but not surprising that those on systemic steroids had more of a problem. Get them on something that’s very targeted if you can and don’t suppress the immune systems that might be handling COVID-19.”
Dr. Musters reported having no disclosures. Dr. Paller disclosed that she is consultant to and/or an investigator for many pharmaceutical companies.
AT AAD 22
Clinical clarity grows about toenail disorder, experts report
BOSTON – The main commonly leading to the wrong therapy and no resolution to the problem, according to an expert update at the annual meeting of the American Academy of Dermatology.
Misinterpretation of the yellow discoloration, a common feature of retronychia, means “many patients are maintained on antifungal therapy for years and years with no change in their condition,” reported Phoebe Rich, MD, director of the Nail Disorders Clinic, Oregon Health & Science University, Portland.
Infection is not commonly involved in retronychia, but importantly, antifungals and antibiotics “have no role in treating the underlying disorder,” Dr. Rich said.
The term retronychia and its description is only about 20 years old, according to Dr. Rich, who cited work by David A. de Berker, MBBS, PhD, a consultant dermatologist at University Hospitals in Bristol, England. His publication on this disorder appeared in 1999, with a more detailed description published about 10 years later.
Recently, the body of literature on this disorder has been growing, contributing to an increasing consensus about etiology, diagnosis, and treatments to consider in the context of causes and severity, Dr. Rich said.
Some but not all patients have abnormal formation of the nail bed, increasing susceptibility to retronychia, but trauma or microtrauma typically serve as a trigger in most cases. Dancing, high heels, steel-toed shoes, and other sources of trauma to the toes are implicated.
Whether or not patients have an inherent susceptibility, injury separates the existing nail from the matrix and nail bed so that newly forming nail begins to grow under the nail rather continuing to push out the old nail.
Susceptibility is increased substantially in individuals with a shortened nail bed, according to Dr. Rich. In severe cases, when there is simply inadequate nail bed for the nail growth to attach, recurrence is common or even inevitable. Even when the nail is removed and regrowth appears normal at the end of a year, those patients with very short nail beds cannot count on a cure.
“Due to the slow growth of nails, it might take 2 or 3 years for the problem to recur,” Dr. Rich cautioned. For this reason, cure rates reported for the various interventions at 1 year might not predict longer-term benefit.
Retronychia is usually a clinical diagnosis based on the presence of the increased bulk of the toenail when overlapping nails cannot be seen. This is not necessarily a single overgrowth. In some cases, multiple layers of nails are stacked one on top of the other. Xanthonychia (yellow nail) is usually present.
“The layering might not be visible without removing the nail,” said Dr. Rich, explaining one reason that the diagnosis is sometimes missed. Ultrasound is a noninvasive means to confirm the problem, although Rich warned that imaging is not necessarily reimbursed.
“There is no diagnosis by histopathology, so it cannot be confirmed with biopsy,” Dr. Rich said.
Treatments range from conservative strategies, particularly topical or intralesional steroids in mild cases, to more invasive procedures such as clipping of the nail plate or surgical avulsion. All can be effective when used appropriately, according to Dr. Rich.
“The more invasive procedures are the more effective, but the caveat is they are also associated with more complications,” said Dr. Rich, citing, for example, the risk of nail dystrophies. Because of the increasing number of studies, the relative benefits and risks of retronychia treatment have now been summarized in a recent review. Dr. Rich suggested the review is one of the most recent and detailed evaluations of the topic that “I encourage everyone to read.”
Despite progress in describing retronychia, Dr. Rich said that there might be more to learn about risk. In particular, she cited the work of Dana W. Stern, MD, a specialist in nail disorders who is in private practice in New York. Dr. Stern is pursuing a hypothesis that at least some cases are caused by potentially targetable biomechanical issues.
“I have observed that many of the younger patients in my practice with retronychia seem to have atypical foot anatomy,” Dr. Stern said in an interview. “I am collecting cases and hoping to explore this issue in more depth.”
She said that foot anatomy in relationship to retronychia has not been adequately evaluated.
“In my review of the literature, I could not find a single study that showed imagery of the feet,” she said. She is considering a collaboration with others, including Rich, to explore this as a factor in retronychia.
Asked about risk of misdiagnosis, Dr. Stern reiterated some of the points made by Dr. Rich. In particular, she agreed that discolored nails alone should not be a reason to initiate antimycotic therapy without considering the possibility of retronychia.
“So many providers are not familiar with the diagnosis, and only 50% of yellow thickened nails are in fact onychomycosis,” she said. “We end up seeing a plethora of patients [with retronychia] who are unfortunately misdiagnosed for years.”
Dr. Rich reported financial relationships with numerous pharmaceutical companies. Dr. Stern reported a financial relationship with Rare Beauty Brands. Neither Dr. Rich nor Dr. Stern said they had any disclosures related to this topic.
A version of this article first appeared on Medscape.com.
BOSTON – The main commonly leading to the wrong therapy and no resolution to the problem, according to an expert update at the annual meeting of the American Academy of Dermatology.
Misinterpretation of the yellow discoloration, a common feature of retronychia, means “many patients are maintained on antifungal therapy for years and years with no change in their condition,” reported Phoebe Rich, MD, director of the Nail Disorders Clinic, Oregon Health & Science University, Portland.
Infection is not commonly involved in retronychia, but importantly, antifungals and antibiotics “have no role in treating the underlying disorder,” Dr. Rich said.
The term retronychia and its description is only about 20 years old, according to Dr. Rich, who cited work by David A. de Berker, MBBS, PhD, a consultant dermatologist at University Hospitals in Bristol, England. His publication on this disorder appeared in 1999, with a more detailed description published about 10 years later.
Recently, the body of literature on this disorder has been growing, contributing to an increasing consensus about etiology, diagnosis, and treatments to consider in the context of causes and severity, Dr. Rich said.
Some but not all patients have abnormal formation of the nail bed, increasing susceptibility to retronychia, but trauma or microtrauma typically serve as a trigger in most cases. Dancing, high heels, steel-toed shoes, and other sources of trauma to the toes are implicated.
Whether or not patients have an inherent susceptibility, injury separates the existing nail from the matrix and nail bed so that newly forming nail begins to grow under the nail rather continuing to push out the old nail.
Susceptibility is increased substantially in individuals with a shortened nail bed, according to Dr. Rich. In severe cases, when there is simply inadequate nail bed for the nail growth to attach, recurrence is common or even inevitable. Even when the nail is removed and regrowth appears normal at the end of a year, those patients with very short nail beds cannot count on a cure.
“Due to the slow growth of nails, it might take 2 or 3 years for the problem to recur,” Dr. Rich cautioned. For this reason, cure rates reported for the various interventions at 1 year might not predict longer-term benefit.
Retronychia is usually a clinical diagnosis based on the presence of the increased bulk of the toenail when overlapping nails cannot be seen. This is not necessarily a single overgrowth. In some cases, multiple layers of nails are stacked one on top of the other. Xanthonychia (yellow nail) is usually present.
“The layering might not be visible without removing the nail,” said Dr. Rich, explaining one reason that the diagnosis is sometimes missed. Ultrasound is a noninvasive means to confirm the problem, although Rich warned that imaging is not necessarily reimbursed.
“There is no diagnosis by histopathology, so it cannot be confirmed with biopsy,” Dr. Rich said.
Treatments range from conservative strategies, particularly topical or intralesional steroids in mild cases, to more invasive procedures such as clipping of the nail plate or surgical avulsion. All can be effective when used appropriately, according to Dr. Rich.
“The more invasive procedures are the more effective, but the caveat is they are also associated with more complications,” said Dr. Rich, citing, for example, the risk of nail dystrophies. Because of the increasing number of studies, the relative benefits and risks of retronychia treatment have now been summarized in a recent review. Dr. Rich suggested the review is one of the most recent and detailed evaluations of the topic that “I encourage everyone to read.”
Despite progress in describing retronychia, Dr. Rich said that there might be more to learn about risk. In particular, she cited the work of Dana W. Stern, MD, a specialist in nail disorders who is in private practice in New York. Dr. Stern is pursuing a hypothesis that at least some cases are caused by potentially targetable biomechanical issues.
“I have observed that many of the younger patients in my practice with retronychia seem to have atypical foot anatomy,” Dr. Stern said in an interview. “I am collecting cases and hoping to explore this issue in more depth.”
She said that foot anatomy in relationship to retronychia has not been adequately evaluated.
“In my review of the literature, I could not find a single study that showed imagery of the feet,” she said. She is considering a collaboration with others, including Rich, to explore this as a factor in retronychia.
Asked about risk of misdiagnosis, Dr. Stern reiterated some of the points made by Dr. Rich. In particular, she agreed that discolored nails alone should not be a reason to initiate antimycotic therapy without considering the possibility of retronychia.
“So many providers are not familiar with the diagnosis, and only 50% of yellow thickened nails are in fact onychomycosis,” she said. “We end up seeing a plethora of patients [with retronychia] who are unfortunately misdiagnosed for years.”
Dr. Rich reported financial relationships with numerous pharmaceutical companies. Dr. Stern reported a financial relationship with Rare Beauty Brands. Neither Dr. Rich nor Dr. Stern said they had any disclosures related to this topic.
A version of this article first appeared on Medscape.com.
BOSTON – The main commonly leading to the wrong therapy and no resolution to the problem, according to an expert update at the annual meeting of the American Academy of Dermatology.
Misinterpretation of the yellow discoloration, a common feature of retronychia, means “many patients are maintained on antifungal therapy for years and years with no change in their condition,” reported Phoebe Rich, MD, director of the Nail Disorders Clinic, Oregon Health & Science University, Portland.
Infection is not commonly involved in retronychia, but importantly, antifungals and antibiotics “have no role in treating the underlying disorder,” Dr. Rich said.
The term retronychia and its description is only about 20 years old, according to Dr. Rich, who cited work by David A. de Berker, MBBS, PhD, a consultant dermatologist at University Hospitals in Bristol, England. His publication on this disorder appeared in 1999, with a more detailed description published about 10 years later.
Recently, the body of literature on this disorder has been growing, contributing to an increasing consensus about etiology, diagnosis, and treatments to consider in the context of causes and severity, Dr. Rich said.
Some but not all patients have abnormal formation of the nail bed, increasing susceptibility to retronychia, but trauma or microtrauma typically serve as a trigger in most cases. Dancing, high heels, steel-toed shoes, and other sources of trauma to the toes are implicated.
Whether or not patients have an inherent susceptibility, injury separates the existing nail from the matrix and nail bed so that newly forming nail begins to grow under the nail rather continuing to push out the old nail.
Susceptibility is increased substantially in individuals with a shortened nail bed, according to Dr. Rich. In severe cases, when there is simply inadequate nail bed for the nail growth to attach, recurrence is common or even inevitable. Even when the nail is removed and regrowth appears normal at the end of a year, those patients with very short nail beds cannot count on a cure.
“Due to the slow growth of nails, it might take 2 or 3 years for the problem to recur,” Dr. Rich cautioned. For this reason, cure rates reported for the various interventions at 1 year might not predict longer-term benefit.
Retronychia is usually a clinical diagnosis based on the presence of the increased bulk of the toenail when overlapping nails cannot be seen. This is not necessarily a single overgrowth. In some cases, multiple layers of nails are stacked one on top of the other. Xanthonychia (yellow nail) is usually present.
“The layering might not be visible without removing the nail,” said Dr. Rich, explaining one reason that the diagnosis is sometimes missed. Ultrasound is a noninvasive means to confirm the problem, although Rich warned that imaging is not necessarily reimbursed.
“There is no diagnosis by histopathology, so it cannot be confirmed with biopsy,” Dr. Rich said.
Treatments range from conservative strategies, particularly topical or intralesional steroids in mild cases, to more invasive procedures such as clipping of the nail plate or surgical avulsion. All can be effective when used appropriately, according to Dr. Rich.
“The more invasive procedures are the more effective, but the caveat is they are also associated with more complications,” said Dr. Rich, citing, for example, the risk of nail dystrophies. Because of the increasing number of studies, the relative benefits and risks of retronychia treatment have now been summarized in a recent review. Dr. Rich suggested the review is one of the most recent and detailed evaluations of the topic that “I encourage everyone to read.”
Despite progress in describing retronychia, Dr. Rich said that there might be more to learn about risk. In particular, she cited the work of Dana W. Stern, MD, a specialist in nail disorders who is in private practice in New York. Dr. Stern is pursuing a hypothesis that at least some cases are caused by potentially targetable biomechanical issues.
“I have observed that many of the younger patients in my practice with retronychia seem to have atypical foot anatomy,” Dr. Stern said in an interview. “I am collecting cases and hoping to explore this issue in more depth.”
She said that foot anatomy in relationship to retronychia has not been adequately evaluated.
“In my review of the literature, I could not find a single study that showed imagery of the feet,” she said. She is considering a collaboration with others, including Rich, to explore this as a factor in retronychia.
Asked about risk of misdiagnosis, Dr. Stern reiterated some of the points made by Dr. Rich. In particular, she agreed that discolored nails alone should not be a reason to initiate antimycotic therapy without considering the possibility of retronychia.
“So many providers are not familiar with the diagnosis, and only 50% of yellow thickened nails are in fact onychomycosis,” she said. “We end up seeing a plethora of patients [with retronychia] who are unfortunately misdiagnosed for years.”
Dr. Rich reported financial relationships with numerous pharmaceutical companies. Dr. Stern reported a financial relationship with Rare Beauty Brands. Neither Dr. Rich nor Dr. Stern said they had any disclosures related to this topic.
A version of this article first appeared on Medscape.com.
AT AAD 2022
New trial data show hair growth in more alopecia areata patients
BOSTON – according to updated results from two phase 3 trials presented at the annual meeting of the American Academy of Dermatology.
The results indicate improved response rates and hair growth among trial participants, said Brett King, MD, PhD, an associate professor of dermatology at Yale University, New Haven, Conn. He is the lead author of the analyses and presented the research.
Dr. King presented 36-week results from the clinical trials at the 2021 annual meeting of the European Academy of Dermatology and Venereology. The same results were also published March 26, 2022, in the New England Journal of Medicine.
“Every bit of data we’ve had is hugely important,” Dr. King said in an interview. “Every time we add 16 weeks of data across hundreds of patients, we are making a huge step forward toward the goal of [Food and Drug Administration approval for a medication for alopecia areata.”
All patients enrolled in the two trials, called BRAVE-AA1 and BRAVE-AA2, had severe alopecia areata, defined as a Severity of Alopecia Tool (SALT) score of at least 50, meaning 50% or less scalp coverage. The score ranges from 0 (no hair loss) to 100 (complete hair loss). The primary endpoint was a SALT score of 20 or less (80% scalp hair coverage).
The researchers pooled data from both clinical trials, with a combined enrollment of 1,200, for the 52-week results presented at the meeting. The placebo group stopped at 36 weeks, and these patients were randomly reassigned to either the 4-mg or 2-mg once-daily baricitinib treatment groups.
At baseline, patients enrolled in the trial had a mean SALT score of 85.5. After 52 weeks, 39.0% of patients who received 4 mg of baricitinib had at least 80% scalp coverage. Of this group, nearly three out of four (74.1%) had at least 90% scalp coverage, or a SALT score of 10 or less.
In patients who received 2 mg of baricitinib, 22.6% had a SALT score of 20 or less 20 (at least 80% scalp hair coverage) at 52 weeks, and two-thirds of that group (67.5%) had at least 90% scalp hair coverage at 52 weeks.
Comparatively, at 36 weeks, 35.2% of participants in BRAVE-AA1 and 32.5% of participants in BRAVE-AA2 receiving 4 mg of baricitinib had at least 80% scalp coverage. In the group taking the lower dose, 21.7% and 17.3% of patients in the BRAVE-AA1 and BRAVE-AA2 trials, respectively, had achieved at least 80% scalp coverage at 36 weeks. (These percentages differ slightly from the NEJM article because of a different analysis of missing data, Dr. King said. For comparison of both 36- and 52-week results, the percentages from the EADV are used above.)
The results indicate that 5% more patients reached the primary endpoint in the additional 16 weeks of the trial, Dr. King said.
Alopecia areata is an autoimmune condition where immune cells attack hair follicles, causing the hair to fall out, and is associated with emotional and psychological distress. Any hair follicle can be attacked, but they are rarely destroyed, so hair can regrow.
"Many underestimate the impact of this autoimmune hair loss condition," Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, told this news organization. He was not involved with the trial. "The burden of the disease, which certainly is an emotional but also a physical one, definitely needs to be addressed with indicated FDA-approved drugs," he noted, which is the goal of these trials.
The BRAVE-AA1 and BRAVE-AA2 trials focused on scalp hair regrowth.
Eyebrow and eyelash growth, secondary outcomes, also improved between 36 and 52 weeks in both groups, calculated using the proportion of participants who had achieved full regrowth or regrowth with minimal gaps. At 36 weeks, about 31%-35% of patients who received 4 mg of baricitinib regrew eyebrow and eyelash hair. By 52 weeks, more than two out of five patients regrew eyebrow (44.1%) and eyelash (45.3%) hair.
“It’s a fantastic achievement and a major step forward in alopecia areata, especially for patients with the most severe and refractory cases,” said Arash Mostaghimi, MD, MPH, the director of inpatient dermatology at Brigham and Women’s Hospital in Boston, Massachusetts. Dr. Mostaghimi is on the advisory board for Eli Lilly, which manufactures baricitinib, and Brigham and Women’s was one of the clinical sites of the trial.
While dermatologists have been aware of how JAK inhibitors can affect hair regrowth in alopecia patients, they have been using these drugs off label, Dr. Friedman said. Therefore, these drugs are expensive and more difficult to access. These trials provide "data that proves the efficacy and safety of [baricitinib] under the umbrella of the FDA portal," he added, which will hopefully lead to an approved indication for alopecia areata, so it can be more accessible to patients.
Adverse events at 52 weeks were consistent with data from 36 weeks, which found that none of these adverse events occurred in more than 10% of participants. The most common adverse events were headache, acne, and increases in muscle-related blood markers. The most common infections reported were pneumonia, herpes zoster, and urinary tract infection.
In February 2022, the FDA granted priority review for baricitinib for the treatment of severe alopecia areata. Lilly expects a regulatory decision by the end of 2022, they said in a press release.
Lilly provided funding for the BRAVE-AA1 and BRAVE-AA2 trials. Dr. King reported financial relationships with Aclaris, Arena Pharmaceuticals, Bristol-Myers Squibb, Concert Pharmaceutics, Dermavant, Lilly, Pfizer, Regeneron, Sanofi Genzyme, and Viela Bio. Dr. Mostaghimi has reported serving on an advisory board for Lilly. Dr. Friedman reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
*This article was updated on 3/28/2022 to include Dr. Friedman's comments, and on 3/31/2022 to correct the statement regarding adverse events reported in the study
BOSTON – according to updated results from two phase 3 trials presented at the annual meeting of the American Academy of Dermatology.
The results indicate improved response rates and hair growth among trial participants, said Brett King, MD, PhD, an associate professor of dermatology at Yale University, New Haven, Conn. He is the lead author of the analyses and presented the research.
Dr. King presented 36-week results from the clinical trials at the 2021 annual meeting of the European Academy of Dermatology and Venereology. The same results were also published March 26, 2022, in the New England Journal of Medicine.
“Every bit of data we’ve had is hugely important,” Dr. King said in an interview. “Every time we add 16 weeks of data across hundreds of patients, we are making a huge step forward toward the goal of [Food and Drug Administration approval for a medication for alopecia areata.”
All patients enrolled in the two trials, called BRAVE-AA1 and BRAVE-AA2, had severe alopecia areata, defined as a Severity of Alopecia Tool (SALT) score of at least 50, meaning 50% or less scalp coverage. The score ranges from 0 (no hair loss) to 100 (complete hair loss). The primary endpoint was a SALT score of 20 or less (80% scalp hair coverage).
The researchers pooled data from both clinical trials, with a combined enrollment of 1,200, for the 52-week results presented at the meeting. The placebo group stopped at 36 weeks, and these patients were randomly reassigned to either the 4-mg or 2-mg once-daily baricitinib treatment groups.
At baseline, patients enrolled in the trial had a mean SALT score of 85.5. After 52 weeks, 39.0% of patients who received 4 mg of baricitinib had at least 80% scalp coverage. Of this group, nearly three out of four (74.1%) had at least 90% scalp coverage, or a SALT score of 10 or less.
In patients who received 2 mg of baricitinib, 22.6% had a SALT score of 20 or less 20 (at least 80% scalp hair coverage) at 52 weeks, and two-thirds of that group (67.5%) had at least 90% scalp hair coverage at 52 weeks.
Comparatively, at 36 weeks, 35.2% of participants in BRAVE-AA1 and 32.5% of participants in BRAVE-AA2 receiving 4 mg of baricitinib had at least 80% scalp coverage. In the group taking the lower dose, 21.7% and 17.3% of patients in the BRAVE-AA1 and BRAVE-AA2 trials, respectively, had achieved at least 80% scalp coverage at 36 weeks. (These percentages differ slightly from the NEJM article because of a different analysis of missing data, Dr. King said. For comparison of both 36- and 52-week results, the percentages from the EADV are used above.)
The results indicate that 5% more patients reached the primary endpoint in the additional 16 weeks of the trial, Dr. King said.
Alopecia areata is an autoimmune condition where immune cells attack hair follicles, causing the hair to fall out, and is associated with emotional and psychological distress. Any hair follicle can be attacked, but they are rarely destroyed, so hair can regrow.
"Many underestimate the impact of this autoimmune hair loss condition," Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, told this news organization. He was not involved with the trial. "The burden of the disease, which certainly is an emotional but also a physical one, definitely needs to be addressed with indicated FDA-approved drugs," he noted, which is the goal of these trials.
The BRAVE-AA1 and BRAVE-AA2 trials focused on scalp hair regrowth.
Eyebrow and eyelash growth, secondary outcomes, also improved between 36 and 52 weeks in both groups, calculated using the proportion of participants who had achieved full regrowth or regrowth with minimal gaps. At 36 weeks, about 31%-35% of patients who received 4 mg of baricitinib regrew eyebrow and eyelash hair. By 52 weeks, more than two out of five patients regrew eyebrow (44.1%) and eyelash (45.3%) hair.
“It’s a fantastic achievement and a major step forward in alopecia areata, especially for patients with the most severe and refractory cases,” said Arash Mostaghimi, MD, MPH, the director of inpatient dermatology at Brigham and Women’s Hospital in Boston, Massachusetts. Dr. Mostaghimi is on the advisory board for Eli Lilly, which manufactures baricitinib, and Brigham and Women’s was one of the clinical sites of the trial.
While dermatologists have been aware of how JAK inhibitors can affect hair regrowth in alopecia patients, they have been using these drugs off label, Dr. Friedman said. Therefore, these drugs are expensive and more difficult to access. These trials provide "data that proves the efficacy and safety of [baricitinib] under the umbrella of the FDA portal," he added, which will hopefully lead to an approved indication for alopecia areata, so it can be more accessible to patients.
Adverse events at 52 weeks were consistent with data from 36 weeks, which found that none of these adverse events occurred in more than 10% of participants. The most common adverse events were headache, acne, and increases in muscle-related blood markers. The most common infections reported were pneumonia, herpes zoster, and urinary tract infection.
In February 2022, the FDA granted priority review for baricitinib for the treatment of severe alopecia areata. Lilly expects a regulatory decision by the end of 2022, they said in a press release.
Lilly provided funding for the BRAVE-AA1 and BRAVE-AA2 trials. Dr. King reported financial relationships with Aclaris, Arena Pharmaceuticals, Bristol-Myers Squibb, Concert Pharmaceutics, Dermavant, Lilly, Pfizer, Regeneron, Sanofi Genzyme, and Viela Bio. Dr. Mostaghimi has reported serving on an advisory board for Lilly. Dr. Friedman reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
*This article was updated on 3/28/2022 to include Dr. Friedman's comments, and on 3/31/2022 to correct the statement regarding adverse events reported in the study
BOSTON – according to updated results from two phase 3 trials presented at the annual meeting of the American Academy of Dermatology.
The results indicate improved response rates and hair growth among trial participants, said Brett King, MD, PhD, an associate professor of dermatology at Yale University, New Haven, Conn. He is the lead author of the analyses and presented the research.
Dr. King presented 36-week results from the clinical trials at the 2021 annual meeting of the European Academy of Dermatology and Venereology. The same results were also published March 26, 2022, in the New England Journal of Medicine.
“Every bit of data we’ve had is hugely important,” Dr. King said in an interview. “Every time we add 16 weeks of data across hundreds of patients, we are making a huge step forward toward the goal of [Food and Drug Administration approval for a medication for alopecia areata.”
All patients enrolled in the two trials, called BRAVE-AA1 and BRAVE-AA2, had severe alopecia areata, defined as a Severity of Alopecia Tool (SALT) score of at least 50, meaning 50% or less scalp coverage. The score ranges from 0 (no hair loss) to 100 (complete hair loss). The primary endpoint was a SALT score of 20 or less (80% scalp hair coverage).
The researchers pooled data from both clinical trials, with a combined enrollment of 1,200, for the 52-week results presented at the meeting. The placebo group stopped at 36 weeks, and these patients were randomly reassigned to either the 4-mg or 2-mg once-daily baricitinib treatment groups.
At baseline, patients enrolled in the trial had a mean SALT score of 85.5. After 52 weeks, 39.0% of patients who received 4 mg of baricitinib had at least 80% scalp coverage. Of this group, nearly three out of four (74.1%) had at least 90% scalp coverage, or a SALT score of 10 or less.
In patients who received 2 mg of baricitinib, 22.6% had a SALT score of 20 or less 20 (at least 80% scalp hair coverage) at 52 weeks, and two-thirds of that group (67.5%) had at least 90% scalp hair coverage at 52 weeks.
Comparatively, at 36 weeks, 35.2% of participants in BRAVE-AA1 and 32.5% of participants in BRAVE-AA2 receiving 4 mg of baricitinib had at least 80% scalp coverage. In the group taking the lower dose, 21.7% and 17.3% of patients in the BRAVE-AA1 and BRAVE-AA2 trials, respectively, had achieved at least 80% scalp coverage at 36 weeks. (These percentages differ slightly from the NEJM article because of a different analysis of missing data, Dr. King said. For comparison of both 36- and 52-week results, the percentages from the EADV are used above.)
The results indicate that 5% more patients reached the primary endpoint in the additional 16 weeks of the trial, Dr. King said.
Alopecia areata is an autoimmune condition where immune cells attack hair follicles, causing the hair to fall out, and is associated with emotional and psychological distress. Any hair follicle can be attacked, but they are rarely destroyed, so hair can regrow.
"Many underestimate the impact of this autoimmune hair loss condition," Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, told this news organization. He was not involved with the trial. "The burden of the disease, which certainly is an emotional but also a physical one, definitely needs to be addressed with indicated FDA-approved drugs," he noted, which is the goal of these trials.
The BRAVE-AA1 and BRAVE-AA2 trials focused on scalp hair regrowth.
Eyebrow and eyelash growth, secondary outcomes, also improved between 36 and 52 weeks in both groups, calculated using the proportion of participants who had achieved full regrowth or regrowth with minimal gaps. At 36 weeks, about 31%-35% of patients who received 4 mg of baricitinib regrew eyebrow and eyelash hair. By 52 weeks, more than two out of five patients regrew eyebrow (44.1%) and eyelash (45.3%) hair.
“It’s a fantastic achievement and a major step forward in alopecia areata, especially for patients with the most severe and refractory cases,” said Arash Mostaghimi, MD, MPH, the director of inpatient dermatology at Brigham and Women’s Hospital in Boston, Massachusetts. Dr. Mostaghimi is on the advisory board for Eli Lilly, which manufactures baricitinib, and Brigham and Women’s was one of the clinical sites of the trial.
While dermatologists have been aware of how JAK inhibitors can affect hair regrowth in alopecia patients, they have been using these drugs off label, Dr. Friedman said. Therefore, these drugs are expensive and more difficult to access. These trials provide "data that proves the efficacy and safety of [baricitinib] under the umbrella of the FDA portal," he added, which will hopefully lead to an approved indication for alopecia areata, so it can be more accessible to patients.
Adverse events at 52 weeks were consistent with data from 36 weeks, which found that none of these adverse events occurred in more than 10% of participants. The most common adverse events were headache, acne, and increases in muscle-related blood markers. The most common infections reported were pneumonia, herpes zoster, and urinary tract infection.
In February 2022, the FDA granted priority review for baricitinib for the treatment of severe alopecia areata. Lilly expects a regulatory decision by the end of 2022, they said in a press release.
Lilly provided funding for the BRAVE-AA1 and BRAVE-AA2 trials. Dr. King reported financial relationships with Aclaris, Arena Pharmaceuticals, Bristol-Myers Squibb, Concert Pharmaceutics, Dermavant, Lilly, Pfizer, Regeneron, Sanofi Genzyme, and Viela Bio. Dr. Mostaghimi has reported serving on an advisory board for Lilly. Dr. Friedman reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
*This article was updated on 3/28/2022 to include Dr. Friedman's comments, and on 3/31/2022 to correct the statement regarding adverse events reported in the study
AT AAD 2022
Aluminum named allergen of the year
BOSTON – The . Aluminum salts, which are the major cause of allergic reactions, are “ubiquitous,” Donald Belsito, MD, professor of dermatology at Columbia University, New York, said at the annual meeting of the American Contact Dermatitis Society.
These salts can be found in sunscreen, cosmetics, dental restorations, and food, to name a few, though the most commonly identified reactions are from aluminum hydroxide, which can be found in some vaccines or preparations for allergen-specific immunotherapy. “It’s the aluminum hydroxide that seems to be more allergenic than other aluminum salts,” Dr. Belsito said in an interview.
“It’s not a dangerous allergy; It’s not a threat,” he said, “but it’s something that dermatologists need to be aware of.”
These reactions normally present as itchy nodules that can last for months and even years, like some reactions from patch testing. “We’re not talking about a vaccine allergy in such a way where people are getting anaphylaxis,” JiaDe Yu, MD, a pediatric dermatologist specializing in allergic contact dermatitis at Massachusetts General Hospital, Boston, said in an interview. “An itchy rash is what we tend to see.”
There have also been occasional reports of atopic dermatitis from aluminum in antiperspirants, astringents, as well as from the metallic aluminum.
Dr. Yu noted that aluminum allergies are not thought to be very common, but the overall prevalence is not known. Studies do suggest, however, that the allergy may be more prevalent in children. In one recent study in Sweden, 5% of children and 0.9% of adults who underwent patch testing had an aluminum contact allergy.
Recommendations for testing
Aluminum is not included in baseline patch testing in the United States, though a recent report about the allergen in the journal Dermatitis argued for its inclusion for pediatric patch testing. Both Dr. Belsito and Dr. Yu agreed that the best approach is to do targeted testing. “If there is a suspicion for it, absolutely test for it,” Dr. Yu said, but if a patient comes in with something like eyelid dermatitis or a rash after a hair care appointment, an aluminum allergy is not very likely.
Because aluminum is also present in Finn Chambers for patch testing, Dr. Belsito advised using plastic chambers in people suspected of having an aluminum allergy. He now uses only plastic chambers in children, he said, as some patients have had reactions to the Finn Chambers even if they have no history of reactions to vaccines or other aluminum-containing products.
While aluminum chloride hexahydrate (ACH) 2% in petrolatum is the commercially available preparation in patch testing, a preparation with ACH 10% is more sensitive, Dr. Belsito said. If a physician strongly suspects an aluminum allergy in a patient but the test with the ACH 2% is negative, he or she should then try a 10% solution, he noted, adding that 7-day readings are also necessary to maximize accuracy.
Vaccine safety
One of the concerns about naming aluminum as the allergen of the year is the potential to cause anxiety around vaccines. “We want to make sure that we’re not giving more fuel to people who have an excuse not to get a vaccine,” Dr. Yu said. “We certainly want to reinforce that fact that it is safe.” Dr. Belsito noted that COVID-19 vaccines do not contain aluminum.
Even on the rare chance that a patient does have a reaction to an aluminum-containing vaccine, these subcutaneous nodules resolve over time, Dr. Belsito said. In his own clinical experience, “99.99% of the time they resolve and there is no residual.” He did add that overreacting to the rash by prescribing injectable steroids can lead to steroid atrophy. In these cases, a topical steroid may be more appropriate.
All unexpected or clinically significant vaccine reactions should be reported to the Vaccine Adverse Event Reporting System, cosponsored by the Centers for Disease Control and Prevention and the Food and Drug Administration. The Clinical Immunization Project Safety Assessment Project, from the CDC, also can provide expertise and advice on aluminum-free alternatives for some vaccines.
Dr. Belsito and Dr. Yu have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
BOSTON – The . Aluminum salts, which are the major cause of allergic reactions, are “ubiquitous,” Donald Belsito, MD, professor of dermatology at Columbia University, New York, said at the annual meeting of the American Contact Dermatitis Society.
These salts can be found in sunscreen, cosmetics, dental restorations, and food, to name a few, though the most commonly identified reactions are from aluminum hydroxide, which can be found in some vaccines or preparations for allergen-specific immunotherapy. “It’s the aluminum hydroxide that seems to be more allergenic than other aluminum salts,” Dr. Belsito said in an interview.
“It’s not a dangerous allergy; It’s not a threat,” he said, “but it’s something that dermatologists need to be aware of.”
These reactions normally present as itchy nodules that can last for months and even years, like some reactions from patch testing. “We’re not talking about a vaccine allergy in such a way where people are getting anaphylaxis,” JiaDe Yu, MD, a pediatric dermatologist specializing in allergic contact dermatitis at Massachusetts General Hospital, Boston, said in an interview. “An itchy rash is what we tend to see.”
There have also been occasional reports of atopic dermatitis from aluminum in antiperspirants, astringents, as well as from the metallic aluminum.
Dr. Yu noted that aluminum allergies are not thought to be very common, but the overall prevalence is not known. Studies do suggest, however, that the allergy may be more prevalent in children. In one recent study in Sweden, 5% of children and 0.9% of adults who underwent patch testing had an aluminum contact allergy.
Recommendations for testing
Aluminum is not included in baseline patch testing in the United States, though a recent report about the allergen in the journal Dermatitis argued for its inclusion for pediatric patch testing. Both Dr. Belsito and Dr. Yu agreed that the best approach is to do targeted testing. “If there is a suspicion for it, absolutely test for it,” Dr. Yu said, but if a patient comes in with something like eyelid dermatitis or a rash after a hair care appointment, an aluminum allergy is not very likely.
Because aluminum is also present in Finn Chambers for patch testing, Dr. Belsito advised using plastic chambers in people suspected of having an aluminum allergy. He now uses only plastic chambers in children, he said, as some patients have had reactions to the Finn Chambers even if they have no history of reactions to vaccines or other aluminum-containing products.
While aluminum chloride hexahydrate (ACH) 2% in petrolatum is the commercially available preparation in patch testing, a preparation with ACH 10% is more sensitive, Dr. Belsito said. If a physician strongly suspects an aluminum allergy in a patient but the test with the ACH 2% is negative, he or she should then try a 10% solution, he noted, adding that 7-day readings are also necessary to maximize accuracy.
Vaccine safety
One of the concerns about naming aluminum as the allergen of the year is the potential to cause anxiety around vaccines. “We want to make sure that we’re not giving more fuel to people who have an excuse not to get a vaccine,” Dr. Yu said. “We certainly want to reinforce that fact that it is safe.” Dr. Belsito noted that COVID-19 vaccines do not contain aluminum.
Even on the rare chance that a patient does have a reaction to an aluminum-containing vaccine, these subcutaneous nodules resolve over time, Dr. Belsito said. In his own clinical experience, “99.99% of the time they resolve and there is no residual.” He did add that overreacting to the rash by prescribing injectable steroids can lead to steroid atrophy. In these cases, a topical steroid may be more appropriate.
All unexpected or clinically significant vaccine reactions should be reported to the Vaccine Adverse Event Reporting System, cosponsored by the Centers for Disease Control and Prevention and the Food and Drug Administration. The Clinical Immunization Project Safety Assessment Project, from the CDC, also can provide expertise and advice on aluminum-free alternatives for some vaccines.
Dr. Belsito and Dr. Yu have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
BOSTON – The . Aluminum salts, which are the major cause of allergic reactions, are “ubiquitous,” Donald Belsito, MD, professor of dermatology at Columbia University, New York, said at the annual meeting of the American Contact Dermatitis Society.
These salts can be found in sunscreen, cosmetics, dental restorations, and food, to name a few, though the most commonly identified reactions are from aluminum hydroxide, which can be found in some vaccines or preparations for allergen-specific immunotherapy. “It’s the aluminum hydroxide that seems to be more allergenic than other aluminum salts,” Dr. Belsito said in an interview.
“It’s not a dangerous allergy; It’s not a threat,” he said, “but it’s something that dermatologists need to be aware of.”
These reactions normally present as itchy nodules that can last for months and even years, like some reactions from patch testing. “We’re not talking about a vaccine allergy in such a way where people are getting anaphylaxis,” JiaDe Yu, MD, a pediatric dermatologist specializing in allergic contact dermatitis at Massachusetts General Hospital, Boston, said in an interview. “An itchy rash is what we tend to see.”
There have also been occasional reports of atopic dermatitis from aluminum in antiperspirants, astringents, as well as from the metallic aluminum.
Dr. Yu noted that aluminum allergies are not thought to be very common, but the overall prevalence is not known. Studies do suggest, however, that the allergy may be more prevalent in children. In one recent study in Sweden, 5% of children and 0.9% of adults who underwent patch testing had an aluminum contact allergy.
Recommendations for testing
Aluminum is not included in baseline patch testing in the United States, though a recent report about the allergen in the journal Dermatitis argued for its inclusion for pediatric patch testing. Both Dr. Belsito and Dr. Yu agreed that the best approach is to do targeted testing. “If there is a suspicion for it, absolutely test for it,” Dr. Yu said, but if a patient comes in with something like eyelid dermatitis or a rash after a hair care appointment, an aluminum allergy is not very likely.
Because aluminum is also present in Finn Chambers for patch testing, Dr. Belsito advised using plastic chambers in people suspected of having an aluminum allergy. He now uses only plastic chambers in children, he said, as some patients have had reactions to the Finn Chambers even if they have no history of reactions to vaccines or other aluminum-containing products.
While aluminum chloride hexahydrate (ACH) 2% in petrolatum is the commercially available preparation in patch testing, a preparation with ACH 10% is more sensitive, Dr. Belsito said. If a physician strongly suspects an aluminum allergy in a patient but the test with the ACH 2% is negative, he or she should then try a 10% solution, he noted, adding that 7-day readings are also necessary to maximize accuracy.
Vaccine safety
One of the concerns about naming aluminum as the allergen of the year is the potential to cause anxiety around vaccines. “We want to make sure that we’re not giving more fuel to people who have an excuse not to get a vaccine,” Dr. Yu said. “We certainly want to reinforce that fact that it is safe.” Dr. Belsito noted that COVID-19 vaccines do not contain aluminum.
Even on the rare chance that a patient does have a reaction to an aluminum-containing vaccine, these subcutaneous nodules resolve over time, Dr. Belsito said. In his own clinical experience, “99.99% of the time they resolve and there is no residual.” He did add that overreacting to the rash by prescribing injectable steroids can lead to steroid atrophy. In these cases, a topical steroid may be more appropriate.
All unexpected or clinically significant vaccine reactions should be reported to the Vaccine Adverse Event Reporting System, cosponsored by the Centers for Disease Control and Prevention and the Food and Drug Administration. The Clinical Immunization Project Safety Assessment Project, from the CDC, also can provide expertise and advice on aluminum-free alternatives for some vaccines.
Dr. Belsito and Dr. Yu have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT ACDS 2022
COVID-19 infection linked to risk of cutaneous autoimmune and vascular diseases
BOSTON – . This predominately favored systemic disease states with cutaneous involvement, rather than skin-limited processes.
The findings come from a large multicenter analysis that Zachary Holcomb, MD, presented during a late-breaking abstract session at the annual meeting of the American Academy of Dermatology.
“Viral triggers have been implicated in the pathogenesis of rheumatologic disease, but information regarding development of autoimmune disease following SARS-CoV-2 infection is limited,” said Dr. Holcomb, chief resident in the Harvard Combined Internal Medicine–Dermatology Residency, Boston. “Given its proposed thromboinflammatory pathobiology, we hypothesized that SARS-CoV-2 infection increases the risk of development of autoimmune disease with cutaneous manifestations and sought to define incidence rates of newly-diagnosed autoimmune diseases following SARS-CoV-2 infection.”
The researchers drew from the TriNetX Dataworks platform, an online cloud-based system that contains aggregated and deidentified patient information from about 75 million patients across 48 health care organizations. The infected cohort was defined as having a positive lab test for severe SARS-CoV-2 within the study window using Logical Observation Identifiers Names and Codes (LOINCs). Healthy controls consisted of a documented health care contact (inpatient or outpatient visit) during the study window without a positive SARS-CoV-2 lab test. Each cohort included patients aged 18-65 at the time of the study, and patients with previously diagnosed cutaneous autoimmune or vascular diseases were excluded from the analysis.
After propensity matching, the COVID-19 infected cohort and the healthy cohort included 1,904,864 patients each, with no baseline differences in age at index event, ethnicity, race, or sex. The study window was between April 1, 2020, and Oct. 1, 2020. The index event was a COVID-19 infection for the infected group and first documented health care contact in the healthy control group. The researchers looked at a window of 60 days following this index event for new incidence of cutaneous or vascular disease.
In the realm of connective tissue and related diseases, they found the incidence was increased among the COVID-19 infected group compared with controls for dermatomyositis (risk ratio, 2.273; P = .0196), scleroderma (RR, 1.959; P = .0001), and systemic lupus erythematosus (RR, 1.401; P < .0001). They also noted a significant decrease in the new incidence of alopecia areata in the COVID-19 infected group compared with controls (RR, 0.527; P < .0001).
No significant differences in the incidence of bullous and papulosquamous diseases were observed between the two groups. However, sarcoidosis was significantly more common in the COVID-19–infected group compared with controls (RR, 2.086; P < .001). “When taking all of these autoinflammatory diseases as a whole, there was an increased incidence in the COVID-19 infected group overall with a RR of 1.168 (P < .0001),” Dr. Holcomb said.
In the realm of vascular skin diseases, there was an increased incidence in the COVID-19 infected group in acrocyanosis (RR, 2.825; P < .001), Raynaud’s phenomenon (RR, 1.462; P < .0001), cutaneous small vessel vasculitis (RR, 1.714; P < .0001), granulomatosis with polyangiitis (RR, 2.667; P = .0002), and temporal arteritis (RR, 1.900; P = .0038).
“Interestingly, despite the academic and lay press reports of COVID toes, we did not see that in our data related to the COVID-infected group,” he said.
Dr. Holcomb acknowledged certain limitations of the study, including a narrow study window with a relatively short follow-up. “We were able to propensity match based on baseline demographics but not necessarily so based on health status and prior autoimmune disease,” he said. In addition, since the study was limited to those aged 18-65, the results may not be generalizable to pediatric and elderly patients, he said.
He described the study findings as “somewhat hypothesis-generating.” For instance, “why would we have more of a systemic process [at play?]. Our theory is that the severe inflammatory nature of COVID-19 leads to a lot of internal organ damage and exposure of autoantigens in that process, with relative skin sparing.”
One of the session moderators, Robert Paul Dellavalle, MD, PhD, professor of dermatology at the University of Colorado, Aurora, characterized the findings as “intriguing” but preliminary. “It would be interesting to look at more recent cohorts and see how vaccination for COVID-19 would impact the incidence rates of some of these diseases,” he said.
When asked for comment, Jeffrey A. Sparks, MD, MMSc, a rheumatologist at Brigham and Women's Hospital and assistant professor of medicine at Harvard Medical School, both in Boston, said, "This is an interesting study that should be followed up. Viral triggers have been known to precede autoimmune diseases so it will be very important to understand whether COVID-19 also impacts systemic autoimmune rheumatic diseases. I would be interested in differences in surveillance between the infection and control groups early in the pandemic. Many patients were avoiding interaction with the health care system at that point."
Dr. Holcomb reported having no financial disclosures. Dr. Dellavalle disclosed that he is a consultant for Altus Labs and ParaPRO LLC. He has received grants and research funding from Pfizer.
* This story was updated on 3/29/22.
BOSTON – . This predominately favored systemic disease states with cutaneous involvement, rather than skin-limited processes.
The findings come from a large multicenter analysis that Zachary Holcomb, MD, presented during a late-breaking abstract session at the annual meeting of the American Academy of Dermatology.
“Viral triggers have been implicated in the pathogenesis of rheumatologic disease, but information regarding development of autoimmune disease following SARS-CoV-2 infection is limited,” said Dr. Holcomb, chief resident in the Harvard Combined Internal Medicine–Dermatology Residency, Boston. “Given its proposed thromboinflammatory pathobiology, we hypothesized that SARS-CoV-2 infection increases the risk of development of autoimmune disease with cutaneous manifestations and sought to define incidence rates of newly-diagnosed autoimmune diseases following SARS-CoV-2 infection.”
The researchers drew from the TriNetX Dataworks platform, an online cloud-based system that contains aggregated and deidentified patient information from about 75 million patients across 48 health care organizations. The infected cohort was defined as having a positive lab test for severe SARS-CoV-2 within the study window using Logical Observation Identifiers Names and Codes (LOINCs). Healthy controls consisted of a documented health care contact (inpatient or outpatient visit) during the study window without a positive SARS-CoV-2 lab test. Each cohort included patients aged 18-65 at the time of the study, and patients with previously diagnosed cutaneous autoimmune or vascular diseases were excluded from the analysis.
After propensity matching, the COVID-19 infected cohort and the healthy cohort included 1,904,864 patients each, with no baseline differences in age at index event, ethnicity, race, or sex. The study window was between April 1, 2020, and Oct. 1, 2020. The index event was a COVID-19 infection for the infected group and first documented health care contact in the healthy control group. The researchers looked at a window of 60 days following this index event for new incidence of cutaneous or vascular disease.
In the realm of connective tissue and related diseases, they found the incidence was increased among the COVID-19 infected group compared with controls for dermatomyositis (risk ratio, 2.273; P = .0196), scleroderma (RR, 1.959; P = .0001), and systemic lupus erythematosus (RR, 1.401; P < .0001). They also noted a significant decrease in the new incidence of alopecia areata in the COVID-19 infected group compared with controls (RR, 0.527; P < .0001).
No significant differences in the incidence of bullous and papulosquamous diseases were observed between the two groups. However, sarcoidosis was significantly more common in the COVID-19–infected group compared with controls (RR, 2.086; P < .001). “When taking all of these autoinflammatory diseases as a whole, there was an increased incidence in the COVID-19 infected group overall with a RR of 1.168 (P < .0001),” Dr. Holcomb said.
In the realm of vascular skin diseases, there was an increased incidence in the COVID-19 infected group in acrocyanosis (RR, 2.825; P < .001), Raynaud’s phenomenon (RR, 1.462; P < .0001), cutaneous small vessel vasculitis (RR, 1.714; P < .0001), granulomatosis with polyangiitis (RR, 2.667; P = .0002), and temporal arteritis (RR, 1.900; P = .0038).
“Interestingly, despite the academic and lay press reports of COVID toes, we did not see that in our data related to the COVID-infected group,” he said.
Dr. Holcomb acknowledged certain limitations of the study, including a narrow study window with a relatively short follow-up. “We were able to propensity match based on baseline demographics but not necessarily so based on health status and prior autoimmune disease,” he said. In addition, since the study was limited to those aged 18-65, the results may not be generalizable to pediatric and elderly patients, he said.
He described the study findings as “somewhat hypothesis-generating.” For instance, “why would we have more of a systemic process [at play?]. Our theory is that the severe inflammatory nature of COVID-19 leads to a lot of internal organ damage and exposure of autoantigens in that process, with relative skin sparing.”
One of the session moderators, Robert Paul Dellavalle, MD, PhD, professor of dermatology at the University of Colorado, Aurora, characterized the findings as “intriguing” but preliminary. “It would be interesting to look at more recent cohorts and see how vaccination for COVID-19 would impact the incidence rates of some of these diseases,” he said.
When asked for comment, Jeffrey A. Sparks, MD, MMSc, a rheumatologist at Brigham and Women's Hospital and assistant professor of medicine at Harvard Medical School, both in Boston, said, "This is an interesting study that should be followed up. Viral triggers have been known to precede autoimmune diseases so it will be very important to understand whether COVID-19 also impacts systemic autoimmune rheumatic diseases. I would be interested in differences in surveillance between the infection and control groups early in the pandemic. Many patients were avoiding interaction with the health care system at that point."
Dr. Holcomb reported having no financial disclosures. Dr. Dellavalle disclosed that he is a consultant for Altus Labs and ParaPRO LLC. He has received grants and research funding from Pfizer.
* This story was updated on 3/29/22.
BOSTON – . This predominately favored systemic disease states with cutaneous involvement, rather than skin-limited processes.
The findings come from a large multicenter analysis that Zachary Holcomb, MD, presented during a late-breaking abstract session at the annual meeting of the American Academy of Dermatology.
“Viral triggers have been implicated in the pathogenesis of rheumatologic disease, but information regarding development of autoimmune disease following SARS-CoV-2 infection is limited,” said Dr. Holcomb, chief resident in the Harvard Combined Internal Medicine–Dermatology Residency, Boston. “Given its proposed thromboinflammatory pathobiology, we hypothesized that SARS-CoV-2 infection increases the risk of development of autoimmune disease with cutaneous manifestations and sought to define incidence rates of newly-diagnosed autoimmune diseases following SARS-CoV-2 infection.”
The researchers drew from the TriNetX Dataworks platform, an online cloud-based system that contains aggregated and deidentified patient information from about 75 million patients across 48 health care organizations. The infected cohort was defined as having a positive lab test for severe SARS-CoV-2 within the study window using Logical Observation Identifiers Names and Codes (LOINCs). Healthy controls consisted of a documented health care contact (inpatient or outpatient visit) during the study window without a positive SARS-CoV-2 lab test. Each cohort included patients aged 18-65 at the time of the study, and patients with previously diagnosed cutaneous autoimmune or vascular diseases were excluded from the analysis.
After propensity matching, the COVID-19 infected cohort and the healthy cohort included 1,904,864 patients each, with no baseline differences in age at index event, ethnicity, race, or sex. The study window was between April 1, 2020, and Oct. 1, 2020. The index event was a COVID-19 infection for the infected group and first documented health care contact in the healthy control group. The researchers looked at a window of 60 days following this index event for new incidence of cutaneous or vascular disease.
In the realm of connective tissue and related diseases, they found the incidence was increased among the COVID-19 infected group compared with controls for dermatomyositis (risk ratio, 2.273; P = .0196), scleroderma (RR, 1.959; P = .0001), and systemic lupus erythematosus (RR, 1.401; P < .0001). They also noted a significant decrease in the new incidence of alopecia areata in the COVID-19 infected group compared with controls (RR, 0.527; P < .0001).
No significant differences in the incidence of bullous and papulosquamous diseases were observed between the two groups. However, sarcoidosis was significantly more common in the COVID-19–infected group compared with controls (RR, 2.086; P < .001). “When taking all of these autoinflammatory diseases as a whole, there was an increased incidence in the COVID-19 infected group overall with a RR of 1.168 (P < .0001),” Dr. Holcomb said.
In the realm of vascular skin diseases, there was an increased incidence in the COVID-19 infected group in acrocyanosis (RR, 2.825; P < .001), Raynaud’s phenomenon (RR, 1.462; P < .0001), cutaneous small vessel vasculitis (RR, 1.714; P < .0001), granulomatosis with polyangiitis (RR, 2.667; P = .0002), and temporal arteritis (RR, 1.900; P = .0038).
“Interestingly, despite the academic and lay press reports of COVID toes, we did not see that in our data related to the COVID-infected group,” he said.
Dr. Holcomb acknowledged certain limitations of the study, including a narrow study window with a relatively short follow-up. “We were able to propensity match based on baseline demographics but not necessarily so based on health status and prior autoimmune disease,” he said. In addition, since the study was limited to those aged 18-65, the results may not be generalizable to pediatric and elderly patients, he said.
He described the study findings as “somewhat hypothesis-generating.” For instance, “why would we have more of a systemic process [at play?]. Our theory is that the severe inflammatory nature of COVID-19 leads to a lot of internal organ damage and exposure of autoantigens in that process, with relative skin sparing.”
One of the session moderators, Robert Paul Dellavalle, MD, PhD, professor of dermatology at the University of Colorado, Aurora, characterized the findings as “intriguing” but preliminary. “It would be interesting to look at more recent cohorts and see how vaccination for COVID-19 would impact the incidence rates of some of these diseases,” he said.
When asked for comment, Jeffrey A. Sparks, MD, MMSc, a rheumatologist at Brigham and Women's Hospital and assistant professor of medicine at Harvard Medical School, both in Boston, said, "This is an interesting study that should be followed up. Viral triggers have been known to precede autoimmune diseases so it will be very important to understand whether COVID-19 also impacts systemic autoimmune rheumatic diseases. I would be interested in differences in surveillance between the infection and control groups early in the pandemic. Many patients were avoiding interaction with the health care system at that point."
Dr. Holcomb reported having no financial disclosures. Dr. Dellavalle disclosed that he is a consultant for Altus Labs and ParaPRO LLC. He has received grants and research funding from Pfizer.
* This story was updated on 3/29/22.
AT AAD 2022
Abdominal rash
Despite his insistence that he was not scratching his abdomen, the lack of primary lesions and the appearance of horizontally oriented excoriations over the abdomen in multiple stages of healing were consistent with neurotic excoriations.
Neurotic excoriation is frequently associated with psychiatric disease, especially obsessive-compulsive disorder and depression.1 Stimulant-use, either by prescription or illicit, can lead to increased self-grooming behaviors, motor tics, and scratching. High doses of stimulants can trigger paranoia and tactile hallucinations.
In this case, the preponderance of skin lesions occurring on the left side of the patient’s abdomen fit with a right-handed individual, which the patient was. On his anterior lower legs, there were linear excoriations oriented vertically. Close observation of the patient during history taking revealed unconscious skin-picking behavior, and dead skin and debris could be noted under his fingernails. Two punch biopsies of active lesions were consistent with excoriations and excluded inflammatory causes of itching. (Careful evaluation for scabies, eczema, urticaria, and contact dermatitis was also performed.)
In this case, the patient’s psychiatrist reduced his dosage of lisdexamfetamine to a starting dose of 30 mg daily, which led to decreased skin scratching behavior. While the patient continued to have limited insight into the nature of his skin changes, progress was measured by a reduction in the number of active lesions.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Gupta MA, Vujcic B, Pur DR, et al. Use of antipsychotic drugs in dermatology. Clin Dermatol. 2018;36:765-773. doi: 10.1016/j.clindermatol.2018.08.006

Despite his insistence that he was not scratching his abdomen, the lack of primary lesions and the appearance of horizontally oriented excoriations over the abdomen in multiple stages of healing were consistent with neurotic excoriations.
Neurotic excoriation is frequently associated with psychiatric disease, especially obsessive-compulsive disorder and depression.1 Stimulant-use, either by prescription or illicit, can lead to increased self-grooming behaviors, motor tics, and scratching. High doses of stimulants can trigger paranoia and tactile hallucinations.
In this case, the preponderance of skin lesions occurring on the left side of the patient’s abdomen fit with a right-handed individual, which the patient was. On his anterior lower legs, there were linear excoriations oriented vertically. Close observation of the patient during history taking revealed unconscious skin-picking behavior, and dead skin and debris could be noted under his fingernails. Two punch biopsies of active lesions were consistent with excoriations and excluded inflammatory causes of itching. (Careful evaluation for scabies, eczema, urticaria, and contact dermatitis was also performed.)
In this case, the patient’s psychiatrist reduced his dosage of lisdexamfetamine to a starting dose of 30 mg daily, which led to decreased skin scratching behavior. While the patient continued to have limited insight into the nature of his skin changes, progress was measured by a reduction in the number of active lesions.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).

Despite his insistence that he was not scratching his abdomen, the lack of primary lesions and the appearance of horizontally oriented excoriations over the abdomen in multiple stages of healing were consistent with neurotic excoriations.
Neurotic excoriation is frequently associated with psychiatric disease, especially obsessive-compulsive disorder and depression.1 Stimulant-use, either by prescription or illicit, can lead to increased self-grooming behaviors, motor tics, and scratching. High doses of stimulants can trigger paranoia and tactile hallucinations.
In this case, the preponderance of skin lesions occurring on the left side of the patient’s abdomen fit with a right-handed individual, which the patient was. On his anterior lower legs, there were linear excoriations oriented vertically. Close observation of the patient during history taking revealed unconscious skin-picking behavior, and dead skin and debris could be noted under his fingernails. Two punch biopsies of active lesions were consistent with excoriations and excluded inflammatory causes of itching. (Careful evaluation for scabies, eczema, urticaria, and contact dermatitis was also performed.)
In this case, the patient’s psychiatrist reduced his dosage of lisdexamfetamine to a starting dose of 30 mg daily, which led to decreased skin scratching behavior. While the patient continued to have limited insight into the nature of his skin changes, progress was measured by a reduction in the number of active lesions.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Gupta MA, Vujcic B, Pur DR, et al. Use of antipsychotic drugs in dermatology. Clin Dermatol. 2018;36:765-773. doi: 10.1016/j.clindermatol.2018.08.006
1. Gupta MA, Vujcic B, Pur DR, et al. Use of antipsychotic drugs in dermatology. Clin Dermatol. 2018;36:765-773. doi: 10.1016/j.clindermatol.2018.08.006
Trial gives new guidance for choosing initial PsA treatment
For patients with psoriatic arthritis (PsA) whose condition doesn’t respond adequately to methotrexate, addition of the tumor necrosis factor (TNF) inhibitor adalimumab increased the likelihood of achieving minimum disease activity (MDA), compared with escalation of MTX dose, according to results from a phase 4, open-label study.
The new study is one of only a few to compare treatment protocols in a field that has seen new therapeutic options become available in recent years. That lack of evidence can leave patients and physicians uncertain about the next step if the initial results of treatment are disappointing.
“There are some gaps in our database and our understanding of psoriatic arthritis, compared to rheumatoid arthritis, where we have had many more studies over the years,” Arthur Kavanaugh, MD, told this news organization when asked to comment on the study.
The trial provides one answer, at least. “There was a clear-cut signal that it made more sense to add adalimumab at that early juncture where a person is not quite doing well enough on methotrexate to satisfy our goal of getting the patient to low disease activity. It gives us as clinicians some ammunition to speak to our insurance formulary people on this side of the Atlantic, or [for] people in the U.K. to go to their local regulatory board that approves medicines and be able to show them some actual practically derived evidence about this very common question that comes up in practice,” senior and corresponding author Philip Mease, MD, said in an interview. The study was published online in The Lancet Rheumatology.
“When a clinician and patient are making the decision to move on from methotrexate monotherapy, either because of lack of efficacy or safety issues, tolerability issues, it makes most sense to add on a biologic medication such as a TNF inhibitor at that juncture, rather than intensifying methotrexate therapy,” said Dr. Mease, who is director of rheumatology research at Swedish Medical Center/Providence St. Joseph Health and a clinical professor at the University of Washington, both in Seattle.
Physicians may be tempted to bump up the dose for patients who can tolerate MTX and who may be showing some improvement, but the new study should prompt a different strategy if MDA isn’t achieved, according to Oliver FitzGerald, MD, a professor at the Conway Institute for Biomolecular Research at University College Dublin, who was asked to comment on the study. “This study clearly shows that the early addition of adalimumab is the better choice, and it would change practice. That being said, there are clearly some patients who do respond sufficiently to increasing methotrexate, and it would be useful to be able to predict which patients might do that.” He added that the study focused on adalimumab and that the results might not apply to other biologics.
The study should encourage use of a quantitative treat-to-target measure like MDA, which is a composite measure of patient perspectives, Dr. Mease said. The American College of Rheumatology and National Psoriasis Foundation and Group for Research and Assessment of Psoriasis and Psoriatic Arthritis have recommended the use of MDA as a treat-to-target measure for PsA. The ACR and NPF recommend TNF inhibitors as first-line treatment, and GRAPPA includes it as a first-line option, whereas the European Alliance of Associations for Rheumatology recommends MTX only in the first line.
The study also suggests that there is value to using adalimumab on a weekly basis if an every-other-week schedule doesn’t produce the desired results. This strategy hasn’t been examined in PsA or even RA, according to Dr. Kavanaugh, who is a professor of medicine at the University of California, San Diego. “It did look like raising the dose might be an option for patients who are on every other week and are not doing quite as well as we would have hoped.”
The CONTROL study was a phase 4, two-part, open-label study. It included 245 patients in 14 countries who did not have MDA with MTX. In the first part of the study, patients were randomly assigned to receive weekly 15 mg MTX along with 40 mg adalimumab every other week, or escalation of MTX dose to 20-25 mg/week. MTX could be administered orally or intravenously. After 16 weeks (part 1), for patients who achieved MDA, current therapy was maintained or modified; for patients who did not achieve MDA, therapy was escalated over the following 16 weeks by giving adalimumab every week in the combination group or by adding adalimumab every other week in the MTX escalation arm.
Overall, 95% of the MTX plus adalimumab group completed part 1, as did 90% of the MTX escalation group. A total of 41% of the adalimumab group achieved MDA at 16 weeks versus 13% of the MTX group (P < .0001). The result held after accounting for sex and the interaction between sex and treatment (odds ratio, 4.6; 95% confidence interval, 2.4-8.9).
Among patients who achieved MDA at 16 weeks, 80% in the adalimumab group continued to have MDA at 32 weeks even after MTX had been withdrawn. Of those in the MTX escalation group, 67% continued to have MDA at 32 weeks with continued escalation of MTX.
Of the patients in the MTX escalation group who did not respond, 55% reached MDA following introduction of adalimumab every other week. Of those who did not respond to adalimumab, 30% reached MDA after switching to weekly adalimumab doses.
The study was open label, and patients who received adalimumab may have expected some improvement; that could have skewed the findings, Dr. Kavanaugh said. “I think that’s an important consideration as we interpret the data. The people who got the MTX arm probably had less of an expectation that they were going to do much better than those who switched to the adalimumab, as did the doctors taking care of them.”
The CONTROL study was funded by AbbVie. Dr. Mease has received research grants, consulted for, or received speaker honoraria from AbbVie, Amgen, Bristol-Myers Squibb, Boehringer Ingelheim, Celgene, Galapagos, Gilead, Janssen, Lilly, Novartis, Pfizer, Sun Pharma, and UCB. Dr. FitzGerald has received grant support and honoraria from AbbVie. Dr. Kavanaugh has received research support from or consulted for AbbVie, Janssen, Pfizer, Lilly, Novartis, and UCB.
A version of this article first appeared on Medscape.com.
For patients with psoriatic arthritis (PsA) whose condition doesn’t respond adequately to methotrexate, addition of the tumor necrosis factor (TNF) inhibitor adalimumab increased the likelihood of achieving minimum disease activity (MDA), compared with escalation of MTX dose, according to results from a phase 4, open-label study.
The new study is one of only a few to compare treatment protocols in a field that has seen new therapeutic options become available in recent years. That lack of evidence can leave patients and physicians uncertain about the next step if the initial results of treatment are disappointing.
“There are some gaps in our database and our understanding of psoriatic arthritis, compared to rheumatoid arthritis, where we have had many more studies over the years,” Arthur Kavanaugh, MD, told this news organization when asked to comment on the study.
The trial provides one answer, at least. “There was a clear-cut signal that it made more sense to add adalimumab at that early juncture where a person is not quite doing well enough on methotrexate to satisfy our goal of getting the patient to low disease activity. It gives us as clinicians some ammunition to speak to our insurance formulary people on this side of the Atlantic, or [for] people in the U.K. to go to their local regulatory board that approves medicines and be able to show them some actual practically derived evidence about this very common question that comes up in practice,” senior and corresponding author Philip Mease, MD, said in an interview. The study was published online in The Lancet Rheumatology.
“When a clinician and patient are making the decision to move on from methotrexate monotherapy, either because of lack of efficacy or safety issues, tolerability issues, it makes most sense to add on a biologic medication such as a TNF inhibitor at that juncture, rather than intensifying methotrexate therapy,” said Dr. Mease, who is director of rheumatology research at Swedish Medical Center/Providence St. Joseph Health and a clinical professor at the University of Washington, both in Seattle.
Physicians may be tempted to bump up the dose for patients who can tolerate MTX and who may be showing some improvement, but the new study should prompt a different strategy if MDA isn’t achieved, according to Oliver FitzGerald, MD, a professor at the Conway Institute for Biomolecular Research at University College Dublin, who was asked to comment on the study. “This study clearly shows that the early addition of adalimumab is the better choice, and it would change practice. That being said, there are clearly some patients who do respond sufficiently to increasing methotrexate, and it would be useful to be able to predict which patients might do that.” He added that the study focused on adalimumab and that the results might not apply to other biologics.
The study should encourage use of a quantitative treat-to-target measure like MDA, which is a composite measure of patient perspectives, Dr. Mease said. The American College of Rheumatology and National Psoriasis Foundation and Group for Research and Assessment of Psoriasis and Psoriatic Arthritis have recommended the use of MDA as a treat-to-target measure for PsA. The ACR and NPF recommend TNF inhibitors as first-line treatment, and GRAPPA includes it as a first-line option, whereas the European Alliance of Associations for Rheumatology recommends MTX only in the first line.
The study also suggests that there is value to using adalimumab on a weekly basis if an every-other-week schedule doesn’t produce the desired results. This strategy hasn’t been examined in PsA or even RA, according to Dr. Kavanaugh, who is a professor of medicine at the University of California, San Diego. “It did look like raising the dose might be an option for patients who are on every other week and are not doing quite as well as we would have hoped.”
The CONTROL study was a phase 4, two-part, open-label study. It included 245 patients in 14 countries who did not have MDA with MTX. In the first part of the study, patients were randomly assigned to receive weekly 15 mg MTX along with 40 mg adalimumab every other week, or escalation of MTX dose to 20-25 mg/week. MTX could be administered orally or intravenously. After 16 weeks (part 1), for patients who achieved MDA, current therapy was maintained or modified; for patients who did not achieve MDA, therapy was escalated over the following 16 weeks by giving adalimumab every week in the combination group or by adding adalimumab every other week in the MTX escalation arm.
Overall, 95% of the MTX plus adalimumab group completed part 1, as did 90% of the MTX escalation group. A total of 41% of the adalimumab group achieved MDA at 16 weeks versus 13% of the MTX group (P < .0001). The result held after accounting for sex and the interaction between sex and treatment (odds ratio, 4.6; 95% confidence interval, 2.4-8.9).
Among patients who achieved MDA at 16 weeks, 80% in the adalimumab group continued to have MDA at 32 weeks even after MTX had been withdrawn. Of those in the MTX escalation group, 67% continued to have MDA at 32 weeks with continued escalation of MTX.
Of the patients in the MTX escalation group who did not respond, 55% reached MDA following introduction of adalimumab every other week. Of those who did not respond to adalimumab, 30% reached MDA after switching to weekly adalimumab doses.
The study was open label, and patients who received adalimumab may have expected some improvement; that could have skewed the findings, Dr. Kavanaugh said. “I think that’s an important consideration as we interpret the data. The people who got the MTX arm probably had less of an expectation that they were going to do much better than those who switched to the adalimumab, as did the doctors taking care of them.”
The CONTROL study was funded by AbbVie. Dr. Mease has received research grants, consulted for, or received speaker honoraria from AbbVie, Amgen, Bristol-Myers Squibb, Boehringer Ingelheim, Celgene, Galapagos, Gilead, Janssen, Lilly, Novartis, Pfizer, Sun Pharma, and UCB. Dr. FitzGerald has received grant support and honoraria from AbbVie. Dr. Kavanaugh has received research support from or consulted for AbbVie, Janssen, Pfizer, Lilly, Novartis, and UCB.
A version of this article first appeared on Medscape.com.
For patients with psoriatic arthritis (PsA) whose condition doesn’t respond adequately to methotrexate, addition of the tumor necrosis factor (TNF) inhibitor adalimumab increased the likelihood of achieving minimum disease activity (MDA), compared with escalation of MTX dose, according to results from a phase 4, open-label study.
The new study is one of only a few to compare treatment protocols in a field that has seen new therapeutic options become available in recent years. That lack of evidence can leave patients and physicians uncertain about the next step if the initial results of treatment are disappointing.
“There are some gaps in our database and our understanding of psoriatic arthritis, compared to rheumatoid arthritis, where we have had many more studies over the years,” Arthur Kavanaugh, MD, told this news organization when asked to comment on the study.
The trial provides one answer, at least. “There was a clear-cut signal that it made more sense to add adalimumab at that early juncture where a person is not quite doing well enough on methotrexate to satisfy our goal of getting the patient to low disease activity. It gives us as clinicians some ammunition to speak to our insurance formulary people on this side of the Atlantic, or [for] people in the U.K. to go to their local regulatory board that approves medicines and be able to show them some actual practically derived evidence about this very common question that comes up in practice,” senior and corresponding author Philip Mease, MD, said in an interview. The study was published online in The Lancet Rheumatology.
“When a clinician and patient are making the decision to move on from methotrexate monotherapy, either because of lack of efficacy or safety issues, tolerability issues, it makes most sense to add on a biologic medication such as a TNF inhibitor at that juncture, rather than intensifying methotrexate therapy,” said Dr. Mease, who is director of rheumatology research at Swedish Medical Center/Providence St. Joseph Health and a clinical professor at the University of Washington, both in Seattle.
Physicians may be tempted to bump up the dose for patients who can tolerate MTX and who may be showing some improvement, but the new study should prompt a different strategy if MDA isn’t achieved, according to Oliver FitzGerald, MD, a professor at the Conway Institute for Biomolecular Research at University College Dublin, who was asked to comment on the study. “This study clearly shows that the early addition of adalimumab is the better choice, and it would change practice. That being said, there are clearly some patients who do respond sufficiently to increasing methotrexate, and it would be useful to be able to predict which patients might do that.” He added that the study focused on adalimumab and that the results might not apply to other biologics.
The study should encourage use of a quantitative treat-to-target measure like MDA, which is a composite measure of patient perspectives, Dr. Mease said. The American College of Rheumatology and National Psoriasis Foundation and Group for Research and Assessment of Psoriasis and Psoriatic Arthritis have recommended the use of MDA as a treat-to-target measure for PsA. The ACR and NPF recommend TNF inhibitors as first-line treatment, and GRAPPA includes it as a first-line option, whereas the European Alliance of Associations for Rheumatology recommends MTX only in the first line.
The study also suggests that there is value to using adalimumab on a weekly basis if an every-other-week schedule doesn’t produce the desired results. This strategy hasn’t been examined in PsA or even RA, according to Dr. Kavanaugh, who is a professor of medicine at the University of California, San Diego. “It did look like raising the dose might be an option for patients who are on every other week and are not doing quite as well as we would have hoped.”
The CONTROL study was a phase 4, two-part, open-label study. It included 245 patients in 14 countries who did not have MDA with MTX. In the first part of the study, patients were randomly assigned to receive weekly 15 mg MTX along with 40 mg adalimumab every other week, or escalation of MTX dose to 20-25 mg/week. MTX could be administered orally or intravenously. After 16 weeks (part 1), for patients who achieved MDA, current therapy was maintained or modified; for patients who did not achieve MDA, therapy was escalated over the following 16 weeks by giving adalimumab every week in the combination group or by adding adalimumab every other week in the MTX escalation arm.
Overall, 95% of the MTX plus adalimumab group completed part 1, as did 90% of the MTX escalation group. A total of 41% of the adalimumab group achieved MDA at 16 weeks versus 13% of the MTX group (P < .0001). The result held after accounting for sex and the interaction between sex and treatment (odds ratio, 4.6; 95% confidence interval, 2.4-8.9).
Among patients who achieved MDA at 16 weeks, 80% in the adalimumab group continued to have MDA at 32 weeks even after MTX had been withdrawn. Of those in the MTX escalation group, 67% continued to have MDA at 32 weeks with continued escalation of MTX.
Of the patients in the MTX escalation group who did not respond, 55% reached MDA following introduction of adalimumab every other week. Of those who did not respond to adalimumab, 30% reached MDA after switching to weekly adalimumab doses.
The study was open label, and patients who received adalimumab may have expected some improvement; that could have skewed the findings, Dr. Kavanaugh said. “I think that’s an important consideration as we interpret the data. The people who got the MTX arm probably had less of an expectation that they were going to do much better than those who switched to the adalimumab, as did the doctors taking care of them.”
The CONTROL study was funded by AbbVie. Dr. Mease has received research grants, consulted for, or received speaker honoraria from AbbVie, Amgen, Bristol-Myers Squibb, Boehringer Ingelheim, Celgene, Galapagos, Gilead, Janssen, Lilly, Novartis, Pfizer, Sun Pharma, and UCB. Dr. FitzGerald has received grant support and honoraria from AbbVie. Dr. Kavanaugh has received research support from or consulted for AbbVie, Janssen, Pfizer, Lilly, Novartis, and UCB.
A version of this article first appeared on Medscape.com.
FROM THE LANCET RHEUMATOLOGY
Adverse skin effects of cancer immunotherapy reviewed
Immune checkpoint inhibitors (ICIs) have unquestionably revolutionized the care of patients with malignant melanoma, non-small cell lung cancer, and other types of cancer.
, according to members of a European Academy of Dermatology and Venereology (EADV) task force.
“The desirable, immune-mediated oncologic response is often achieved at the cost of immune-related adverse events (irAEs) that may potentially affect any organ system,” they write in a position statement on the management of ICI-derived dermatologic adverse events.
Recommendations from the EADV “Dermatology for Cancer Patients” task force have been published in the Journal of the European Academy of Dermatology and Venereology.
Task force members developed the recommendations based on clinical experience from published data and came up with specific recommendations for treating cutaneous toxicities associated with dermatologic immune-related adverse events (dirAEs) that occur in patients receiving immunotherapy with an ICI.
ICIs include the cytotoxic T-lymphocyte–associated antigen 4 (CTLA-4) inhibitor ipilimumab (Yervoy, Bristol Myers Squibb), and inhibitors of programmed death protein 1 (PD-1) and its ligand (PD-L1), including nivolumab (Opdivo, Bristol Myers Squibb), pembrolizumab (Keytruda, Merck), and other agents.
“The basic principle of management is that the interventions should be tailored to serve the equilibrium between patients’ relief from the symptoms and signs of skin toxicity and the preservation of an unimpeded oncologic treatment,” they write.
The recommendations are in line with those included in a 2021 update of the American Society of Clinical Oncology (ASCO) guidelines on the management of irAEs in patients treated with ICIs across the whole range of organ systems, said Milan J. Anadkat, MD, professor of dermatology and director of dermatology clinical trials at Washington University School of Medicine, St. Louis. Dr. Anadkat was a coauthor of the ASCO guideline update.
Although the European recommendations focus only on dermatologic side effects of ICIs in patients with cancer, “that doesn’t diminish their importance. They do a good job of summarizing how to approach and how to manage it depending on the severity of the toxicities and the various types of toxicities,” he told this news organization.
Having a paper focused exclusively on the dermatologic side effects of ICIs allows the inclusion of photographs that can help clinicians identify specific conditions that may require referral to a dermatologist, he said.
Both Dr. Anadkat and the authors of the European recommendations noted that dermatologic irAEs are more common with CTLA-4 inhibition than with PD-1/PD-L1 inhibition.
“It has to do with where the target is,” Dr. Anadkat said. “CTLA-4 inhibition works on a central aspect of the immune system, so it’s a much less specific site, whereas PD-1 affects an interaction at the site of the tumor cell itself, so it’s a little more specific.”
Pruritus
ICI-induced pruritus can occur without apparent skin changes, they write, noting that in a recent study of patients with dirAEs, about one-third had isolated pruritus.
The task force members cite a meta-analysis indicating a pruritus incidence of 13.2% for patients treated with nivolumab and 20.2% for patients treated with pembrolizumab but respective grade 3 pruritus rates of only 0.5% and 2.3%. The reported incidence of pruritus with ipilimumab was 47% in a different study.
Recommended treatments include topical moisturizers with or without medium-to-high potency corticosteroids for grade 1 reactions, non-sedating histamines and/or GABA agonists such as pregabalin, or gabapentin for grade 2 pruritus, and suspension of ICIs until pruritus improves in patients with grade 3 pruritus.
Maculopapular rash
Maculopapular or eczema-like rashes may occur in up to 68% of patients who receive a CTLA-4 inhibitor and up to 20% of those who receive a PD1/PD-L1 inhibitor, the authors note. Rashes commonly appear within 3-6 weeks of initiating therapy.
“The clinical presentation is nonspecific and consists of a rapid onset of multiple minimally scaly, erythematous macules and papules, congregating into plaques. Lesions are mostly located on trunk and extensor surfaces of the extremities and the face is generally spared,” they write.
Maculopapular rashes are typically accompanied by itching but could be asymptomatic, they noted.
Mild (grade 1) rashes may respond to moisturizers and topical potent or super-potent corticosteroids. Patients with grade 2 rash should also receive oral antihistamines. Systemic corticosteroids may be considered for patients with grade 3 rashes but only after other dirAEs that may require specific management, such as psoriasis, are ruled out.
Psoriasis-like rash
The most common form of psoriasis seen in patients treated with ICIs is psoriasis vulgaris with plaques, but other clinical variants are also seen, the authors note.
“Topical agents (corticosteroids, Vitamin D analogues) are prescribed in Grades 1/2 and supplementary” to systemic treatment for patients with grade 3 or recalcitrant lesions, they write. “If skin-directed therapies fail to provide symptomatic control,” systemic treatment and narrow band UVB phototherapy “should be considered,” they add.
Evidence regarding the use of systemic therapies to treat psoriasis-like rash associated with ICIs is sparse. Acitretin can be safely used in patients with cancer. Low-dose methotrexate is also safe to use except in patients with non-melanoma skin cancers. Cyclosporine, however, should be avoided because of the potential for tumor-promoting effects, they emphasized.
The recommendations also cover treatment of lichen planus-like and vitiligo-like rashes, as well as hair and nail changes, autoimmune bullous disorders, and oral mucosal dirAEs.
In addition, the recommendations cover severe cutaneous adverse reactions as well as serious, potentially life-threatening dirAEs, including Stevens-Johnson syndrome/TEN, acute generalized exanthematous pustulosis (AGEP), and drug reaction with eosinophilia and systemic symptoms/drug-induced hypersensitivity syndrome (DRESS/DIHS).
“The dose of corticosteroids may be adapted to the severity of DRESS. The therapeutic benefit of systemic corticosteroids in the management of SJS/TEN remains controversial, and some authors favor treatment with cyclosporine. However, the use of corticosteroids in this context of ICI treatment appears reasonable and should be proposed. Short courses of steroids seem also effective in AGEP,” the task force members write.
The recommendations did not have outside funding. Of the 19 authors, 6 disclosed relationships with various pharmaceutical companies, including AbbVie, Leo Pharma, Boehringer Ingelheim, Bristol Myers Squibb, and/or Janssen. Dr. Anadkat disclosed previous relationships with Merck, Bristol Myers Squibb, and current relationships with others.
A version of this article first appeared on Medscape.com.
Immune checkpoint inhibitors (ICIs) have unquestionably revolutionized the care of patients with malignant melanoma, non-small cell lung cancer, and other types of cancer.
, according to members of a European Academy of Dermatology and Venereology (EADV) task force.
“The desirable, immune-mediated oncologic response is often achieved at the cost of immune-related adverse events (irAEs) that may potentially affect any organ system,” they write in a position statement on the management of ICI-derived dermatologic adverse events.
Recommendations from the EADV “Dermatology for Cancer Patients” task force have been published in the Journal of the European Academy of Dermatology and Venereology.
Task force members developed the recommendations based on clinical experience from published data and came up with specific recommendations for treating cutaneous toxicities associated with dermatologic immune-related adverse events (dirAEs) that occur in patients receiving immunotherapy with an ICI.
ICIs include the cytotoxic T-lymphocyte–associated antigen 4 (CTLA-4) inhibitor ipilimumab (Yervoy, Bristol Myers Squibb), and inhibitors of programmed death protein 1 (PD-1) and its ligand (PD-L1), including nivolumab (Opdivo, Bristol Myers Squibb), pembrolizumab (Keytruda, Merck), and other agents.
“The basic principle of management is that the interventions should be tailored to serve the equilibrium between patients’ relief from the symptoms and signs of skin toxicity and the preservation of an unimpeded oncologic treatment,” they write.
The recommendations are in line with those included in a 2021 update of the American Society of Clinical Oncology (ASCO) guidelines on the management of irAEs in patients treated with ICIs across the whole range of organ systems, said Milan J. Anadkat, MD, professor of dermatology and director of dermatology clinical trials at Washington University School of Medicine, St. Louis. Dr. Anadkat was a coauthor of the ASCO guideline update.
Although the European recommendations focus only on dermatologic side effects of ICIs in patients with cancer, “that doesn’t diminish their importance. They do a good job of summarizing how to approach and how to manage it depending on the severity of the toxicities and the various types of toxicities,” he told this news organization.
Having a paper focused exclusively on the dermatologic side effects of ICIs allows the inclusion of photographs that can help clinicians identify specific conditions that may require referral to a dermatologist, he said.
Both Dr. Anadkat and the authors of the European recommendations noted that dermatologic irAEs are more common with CTLA-4 inhibition than with PD-1/PD-L1 inhibition.
“It has to do with where the target is,” Dr. Anadkat said. “CTLA-4 inhibition works on a central aspect of the immune system, so it’s a much less specific site, whereas PD-1 affects an interaction at the site of the tumor cell itself, so it’s a little more specific.”
Pruritus
ICI-induced pruritus can occur without apparent skin changes, they write, noting that in a recent study of patients with dirAEs, about one-third had isolated pruritus.
The task force members cite a meta-analysis indicating a pruritus incidence of 13.2% for patients treated with nivolumab and 20.2% for patients treated with pembrolizumab but respective grade 3 pruritus rates of only 0.5% and 2.3%. The reported incidence of pruritus with ipilimumab was 47% in a different study.
Recommended treatments include topical moisturizers with or without medium-to-high potency corticosteroids for grade 1 reactions, non-sedating histamines and/or GABA agonists such as pregabalin, or gabapentin for grade 2 pruritus, and suspension of ICIs until pruritus improves in patients with grade 3 pruritus.
Maculopapular rash
Maculopapular or eczema-like rashes may occur in up to 68% of patients who receive a CTLA-4 inhibitor and up to 20% of those who receive a PD1/PD-L1 inhibitor, the authors note. Rashes commonly appear within 3-6 weeks of initiating therapy.
“The clinical presentation is nonspecific and consists of a rapid onset of multiple minimally scaly, erythematous macules and papules, congregating into plaques. Lesions are mostly located on trunk and extensor surfaces of the extremities and the face is generally spared,” they write.
Maculopapular rashes are typically accompanied by itching but could be asymptomatic, they noted.
Mild (grade 1) rashes may respond to moisturizers and topical potent or super-potent corticosteroids. Patients with grade 2 rash should also receive oral antihistamines. Systemic corticosteroids may be considered for patients with grade 3 rashes but only after other dirAEs that may require specific management, such as psoriasis, are ruled out.
Psoriasis-like rash
The most common form of psoriasis seen in patients treated with ICIs is psoriasis vulgaris with plaques, but other clinical variants are also seen, the authors note.
“Topical agents (corticosteroids, Vitamin D analogues) are prescribed in Grades 1/2 and supplementary” to systemic treatment for patients with grade 3 or recalcitrant lesions, they write. “If skin-directed therapies fail to provide symptomatic control,” systemic treatment and narrow band UVB phototherapy “should be considered,” they add.
Evidence regarding the use of systemic therapies to treat psoriasis-like rash associated with ICIs is sparse. Acitretin can be safely used in patients with cancer. Low-dose methotrexate is also safe to use except in patients with non-melanoma skin cancers. Cyclosporine, however, should be avoided because of the potential for tumor-promoting effects, they emphasized.
The recommendations also cover treatment of lichen planus-like and vitiligo-like rashes, as well as hair and nail changes, autoimmune bullous disorders, and oral mucosal dirAEs.
In addition, the recommendations cover severe cutaneous adverse reactions as well as serious, potentially life-threatening dirAEs, including Stevens-Johnson syndrome/TEN, acute generalized exanthematous pustulosis (AGEP), and drug reaction with eosinophilia and systemic symptoms/drug-induced hypersensitivity syndrome (DRESS/DIHS).
“The dose of corticosteroids may be adapted to the severity of DRESS. The therapeutic benefit of systemic corticosteroids in the management of SJS/TEN remains controversial, and some authors favor treatment with cyclosporine. However, the use of corticosteroids in this context of ICI treatment appears reasonable and should be proposed. Short courses of steroids seem also effective in AGEP,” the task force members write.
The recommendations did not have outside funding. Of the 19 authors, 6 disclosed relationships with various pharmaceutical companies, including AbbVie, Leo Pharma, Boehringer Ingelheim, Bristol Myers Squibb, and/or Janssen. Dr. Anadkat disclosed previous relationships with Merck, Bristol Myers Squibb, and current relationships with others.
A version of this article first appeared on Medscape.com.
Immune checkpoint inhibitors (ICIs) have unquestionably revolutionized the care of patients with malignant melanoma, non-small cell lung cancer, and other types of cancer.
, according to members of a European Academy of Dermatology and Venereology (EADV) task force.
“The desirable, immune-mediated oncologic response is often achieved at the cost of immune-related adverse events (irAEs) that may potentially affect any organ system,” they write in a position statement on the management of ICI-derived dermatologic adverse events.
Recommendations from the EADV “Dermatology for Cancer Patients” task force have been published in the Journal of the European Academy of Dermatology and Venereology.
Task force members developed the recommendations based on clinical experience from published data and came up with specific recommendations for treating cutaneous toxicities associated with dermatologic immune-related adverse events (dirAEs) that occur in patients receiving immunotherapy with an ICI.
ICIs include the cytotoxic T-lymphocyte–associated antigen 4 (CTLA-4) inhibitor ipilimumab (Yervoy, Bristol Myers Squibb), and inhibitors of programmed death protein 1 (PD-1) and its ligand (PD-L1), including nivolumab (Opdivo, Bristol Myers Squibb), pembrolizumab (Keytruda, Merck), and other agents.
“The basic principle of management is that the interventions should be tailored to serve the equilibrium between patients’ relief from the symptoms and signs of skin toxicity and the preservation of an unimpeded oncologic treatment,” they write.
The recommendations are in line with those included in a 2021 update of the American Society of Clinical Oncology (ASCO) guidelines on the management of irAEs in patients treated with ICIs across the whole range of organ systems, said Milan J. Anadkat, MD, professor of dermatology and director of dermatology clinical trials at Washington University School of Medicine, St. Louis. Dr. Anadkat was a coauthor of the ASCO guideline update.
Although the European recommendations focus only on dermatologic side effects of ICIs in patients with cancer, “that doesn’t diminish their importance. They do a good job of summarizing how to approach and how to manage it depending on the severity of the toxicities and the various types of toxicities,” he told this news organization.
Having a paper focused exclusively on the dermatologic side effects of ICIs allows the inclusion of photographs that can help clinicians identify specific conditions that may require referral to a dermatologist, he said.
Both Dr. Anadkat and the authors of the European recommendations noted that dermatologic irAEs are more common with CTLA-4 inhibition than with PD-1/PD-L1 inhibition.
“It has to do with where the target is,” Dr. Anadkat said. “CTLA-4 inhibition works on a central aspect of the immune system, so it’s a much less specific site, whereas PD-1 affects an interaction at the site of the tumor cell itself, so it’s a little more specific.”
Pruritus
ICI-induced pruritus can occur without apparent skin changes, they write, noting that in a recent study of patients with dirAEs, about one-third had isolated pruritus.
The task force members cite a meta-analysis indicating a pruritus incidence of 13.2% for patients treated with nivolumab and 20.2% for patients treated with pembrolizumab but respective grade 3 pruritus rates of only 0.5% and 2.3%. The reported incidence of pruritus with ipilimumab was 47% in a different study.
Recommended treatments include topical moisturizers with or without medium-to-high potency corticosteroids for grade 1 reactions, non-sedating histamines and/or GABA agonists such as pregabalin, or gabapentin for grade 2 pruritus, and suspension of ICIs until pruritus improves in patients with grade 3 pruritus.
Maculopapular rash
Maculopapular or eczema-like rashes may occur in up to 68% of patients who receive a CTLA-4 inhibitor and up to 20% of those who receive a PD1/PD-L1 inhibitor, the authors note. Rashes commonly appear within 3-6 weeks of initiating therapy.
“The clinical presentation is nonspecific and consists of a rapid onset of multiple minimally scaly, erythematous macules and papules, congregating into plaques. Lesions are mostly located on trunk and extensor surfaces of the extremities and the face is generally spared,” they write.
Maculopapular rashes are typically accompanied by itching but could be asymptomatic, they noted.
Mild (grade 1) rashes may respond to moisturizers and topical potent or super-potent corticosteroids. Patients with grade 2 rash should also receive oral antihistamines. Systemic corticosteroids may be considered for patients with grade 3 rashes but only after other dirAEs that may require specific management, such as psoriasis, are ruled out.
Psoriasis-like rash
The most common form of psoriasis seen in patients treated with ICIs is psoriasis vulgaris with plaques, but other clinical variants are also seen, the authors note.
“Topical agents (corticosteroids, Vitamin D analogues) are prescribed in Grades 1/2 and supplementary” to systemic treatment for patients with grade 3 or recalcitrant lesions, they write. “If skin-directed therapies fail to provide symptomatic control,” systemic treatment and narrow band UVB phototherapy “should be considered,” they add.
Evidence regarding the use of systemic therapies to treat psoriasis-like rash associated with ICIs is sparse. Acitretin can be safely used in patients with cancer. Low-dose methotrexate is also safe to use except in patients with non-melanoma skin cancers. Cyclosporine, however, should be avoided because of the potential for tumor-promoting effects, they emphasized.
The recommendations also cover treatment of lichen planus-like and vitiligo-like rashes, as well as hair and nail changes, autoimmune bullous disorders, and oral mucosal dirAEs.
In addition, the recommendations cover severe cutaneous adverse reactions as well as serious, potentially life-threatening dirAEs, including Stevens-Johnson syndrome/TEN, acute generalized exanthematous pustulosis (AGEP), and drug reaction with eosinophilia and systemic symptoms/drug-induced hypersensitivity syndrome (DRESS/DIHS).
“The dose of corticosteroids may be adapted to the severity of DRESS. The therapeutic benefit of systemic corticosteroids in the management of SJS/TEN remains controversial, and some authors favor treatment with cyclosporine. However, the use of corticosteroids in this context of ICI treatment appears reasonable and should be proposed. Short courses of steroids seem also effective in AGEP,” the task force members write.
The recommendations did not have outside funding. Of the 19 authors, 6 disclosed relationships with various pharmaceutical companies, including AbbVie, Leo Pharma, Boehringer Ingelheim, Bristol Myers Squibb, and/or Janssen. Dr. Anadkat disclosed previous relationships with Merck, Bristol Myers Squibb, and current relationships with others.
A version of this article first appeared on Medscape.com.