User login
Apremilast has neutral effect on vascular inflammation in psoriasis study
BOSTON – Treatment with , and glucose metabolism, in a study presented at the 2022 American Academy of Dermatology annual meeting.
In the phase 4, open-label, single arm trial, participants also lost subcutaneous and visceral fat after 16 weeks on the oral medication, a phosphodiesterase 4 (PDE4) inhibitor, and maintained that loss at 52 weeks.
People with psoriasis have an increased risk of obesity, type 2 diabetes, and cardiovascular events. Patients with more significant psoriasis “tend to die about 5 years younger than they should, based on their risk factors for mortality,” Joel Gelfand, MD, MSCE, professor of dermatology and epidemiology and vice chair of clinical research in dermatology at the University of Pennsylvania Perelman School of Medicine, Philadelphia, told this news organization.
He led the research and presented the findings at the AAD meeting March 26. “As a result, there has been a keen interest in understanding how psoriasis therapies impact cardiovascular risk, the idea being that by controlling inflammation, you may lower the risk of these patients developing cardiovascular disease over time,” he said.
Previous trials looking at the effect of psoriasis therapies on vascular inflammation “have been, for the most part, inconclusive,” Michael Garshick, MD, a cardiologist at NYU Langone Health, told this news organization. Dr. Garshick was not involved with the research. A 2021 systematic review of psoriasis clinicals trials reported that the tumor necrosis factor (TNF) blocker adalimumab (Humira) and phototherapy had the greatest effect on cardiometabolic markers, while ustekinumab (Stelara), an interleukin (IL)-12 and IL-23 antagonist, was the only treatment that improved vascular inflammation. These variable findings make this area “ripe for study,” noted Dr. Garshick.
To observe how apremilast, which is approved by the FDA for treating psoriasis and psoriatic arthritis, affected vascular inflammation, adiposity, and blood-based cardiometabolic markers, Dr. Gelfand organized an open-label study in adults with moderate-to-severe psoriasis. All participants were 18 years or older, had psoriasis for at least 6 months, and were candidates for systemic therapy. All patients underwent FDG PET/CT scans to assess aortic vascular inflammation and had blood work at baseline. Of the 70 patients originally enrolled in the study, 60 remained in the study at week 16, including 57 who underwent imaging for the second time. Thirty-nine participants remained in the study until week 52, and all except one had another scan.
The average age of participants was 47 years, and their mean BMI was 30. More than 80% of participants were White (83%) and 77% were male. The study population had lived with psoriasis for an average of 16 years and 8 patients also had psoriatic arthritis. At baseline, on average, participants had a Psoriasis Area and Severity Index (PASI) score of 18.62, a dermatology life quality index (DLQI) score of 11.60, and 22% of participants’ BSA (body surface area) were affected. The mean TBRmax, the marker for vascular inflammation, was 1.61.
Treatment responses were as expected for apremilast, with 35% of patients achieving PASI 75 and 65% of participants reporting DLQI scores of 5 or less by 16 weeks. At 52 weeks, 31% of the cohort had achieved PASI 75, and 67% reported DLQI score of 5 or higher. All psoriasis endpoints had improved since baseline (P = .001).
Throughout the study period, there was no significant change in TBRmax. However, in a sensitivity analysis, the 16 patients with a baseline TBRmax of 1.6 or higher had an absolute reduction of 0.21 in TBR by week 52. “That suggests that maybe a subset of people who have higher levels of aortic inflammation at baseline may experience some reduction that portend, potentially, some health benefits over time,” Dr. Gelfand said. “Ultimately, I wouldn’t hang my hat on the finding,” he said, noting that additional research comparing the treatment to placebo is necessary.
Both visceral and subcutaneous adipose tissue (VAT and SAT) decreased by week 16, and this reduction was maintained through week 52. In the first 16 weeks of the study, VAT decreased by 5.32% (P = .0009), and SAT decreased by 5.53% (P = .0005). From baseline to 52 weeks, VAT decreased by 5.52% (P = .0148), and SAT decreased by 5.50% (P = .0096). There were no significant differences between week 16 and week 52 in VAT or SAT.
Of the 68 blood biomarkers analyzed, there were significant decreases in the inflammatory markers ferritin (P = .015) and IL-beta (P = .006), the lipid metabolism biomarker HDL-cholesterol efflux (P = .008), and ketone bodies (P = .006). There were also increases in the inflammatory marker IL-8 (P = .003), the lipid metabolism marker ApoA (P = .05), and insulin (P = .05). Ferritin was the only biomarker that was reduced on both week 16 and week 52.
“If you want to be a purist, this was a negative trial,” said Dr. Garshick, because apremilast was not found to decrease vascular inflammation; however, he noted that the biomarker changes “were hopeful secondary endpoints.” It could be, he said, that another outcome measure may be better able to show changes in vascular inflammation compared with FDG. “It’s always hard to figure out what a good surrogate endpoint is in cardiovascular trials,” he noted, “so it may be that FDG/PET is too noisy or not reliable enough to see the outcome that we want to see.”
Dr. Gelfand reports consulting fees/grants from Amgen, AbbVie, BMS, Boehringer Ingelheim, Janssen Biologics, Novartis Corp, Pfizer, and UCB (DSMB). He serves as the Deputy Editor for the Journal of Investigative Dermatology and the Chief Medical Editor at Healio Psoriatic Disease and receives honoraria for both roles. Dr. Garshick has received consulting fees from AbbVie.
A version of this article first appeared on Medscape.com.
BOSTON – Treatment with , and glucose metabolism, in a study presented at the 2022 American Academy of Dermatology annual meeting.
In the phase 4, open-label, single arm trial, participants also lost subcutaneous and visceral fat after 16 weeks on the oral medication, a phosphodiesterase 4 (PDE4) inhibitor, and maintained that loss at 52 weeks.
People with psoriasis have an increased risk of obesity, type 2 diabetes, and cardiovascular events. Patients with more significant psoriasis “tend to die about 5 years younger than they should, based on their risk factors for mortality,” Joel Gelfand, MD, MSCE, professor of dermatology and epidemiology and vice chair of clinical research in dermatology at the University of Pennsylvania Perelman School of Medicine, Philadelphia, told this news organization.
He led the research and presented the findings at the AAD meeting March 26. “As a result, there has been a keen interest in understanding how psoriasis therapies impact cardiovascular risk, the idea being that by controlling inflammation, you may lower the risk of these patients developing cardiovascular disease over time,” he said.
Previous trials looking at the effect of psoriasis therapies on vascular inflammation “have been, for the most part, inconclusive,” Michael Garshick, MD, a cardiologist at NYU Langone Health, told this news organization. Dr. Garshick was not involved with the research. A 2021 systematic review of psoriasis clinicals trials reported that the tumor necrosis factor (TNF) blocker adalimumab (Humira) and phototherapy had the greatest effect on cardiometabolic markers, while ustekinumab (Stelara), an interleukin (IL)-12 and IL-23 antagonist, was the only treatment that improved vascular inflammation. These variable findings make this area “ripe for study,” noted Dr. Garshick.
To observe how apremilast, which is approved by the FDA for treating psoriasis and psoriatic arthritis, affected vascular inflammation, adiposity, and blood-based cardiometabolic markers, Dr. Gelfand organized an open-label study in adults with moderate-to-severe psoriasis. All participants were 18 years or older, had psoriasis for at least 6 months, and were candidates for systemic therapy. All patients underwent FDG PET/CT scans to assess aortic vascular inflammation and had blood work at baseline. Of the 70 patients originally enrolled in the study, 60 remained in the study at week 16, including 57 who underwent imaging for the second time. Thirty-nine participants remained in the study until week 52, and all except one had another scan.
The average age of participants was 47 years, and their mean BMI was 30. More than 80% of participants were White (83%) and 77% were male. The study population had lived with psoriasis for an average of 16 years and 8 patients also had psoriatic arthritis. At baseline, on average, participants had a Psoriasis Area and Severity Index (PASI) score of 18.62, a dermatology life quality index (DLQI) score of 11.60, and 22% of participants’ BSA (body surface area) were affected. The mean TBRmax, the marker for vascular inflammation, was 1.61.
Treatment responses were as expected for apremilast, with 35% of patients achieving PASI 75 and 65% of participants reporting DLQI scores of 5 or less by 16 weeks. At 52 weeks, 31% of the cohort had achieved PASI 75, and 67% reported DLQI score of 5 or higher. All psoriasis endpoints had improved since baseline (P = .001).
Throughout the study period, there was no significant change in TBRmax. However, in a sensitivity analysis, the 16 patients with a baseline TBRmax of 1.6 or higher had an absolute reduction of 0.21 in TBR by week 52. “That suggests that maybe a subset of people who have higher levels of aortic inflammation at baseline may experience some reduction that portend, potentially, some health benefits over time,” Dr. Gelfand said. “Ultimately, I wouldn’t hang my hat on the finding,” he said, noting that additional research comparing the treatment to placebo is necessary.
Both visceral and subcutaneous adipose tissue (VAT and SAT) decreased by week 16, and this reduction was maintained through week 52. In the first 16 weeks of the study, VAT decreased by 5.32% (P = .0009), and SAT decreased by 5.53% (P = .0005). From baseline to 52 weeks, VAT decreased by 5.52% (P = .0148), and SAT decreased by 5.50% (P = .0096). There were no significant differences between week 16 and week 52 in VAT or SAT.
Of the 68 blood biomarkers analyzed, there were significant decreases in the inflammatory markers ferritin (P = .015) and IL-beta (P = .006), the lipid metabolism biomarker HDL-cholesterol efflux (P = .008), and ketone bodies (P = .006). There were also increases in the inflammatory marker IL-8 (P = .003), the lipid metabolism marker ApoA (P = .05), and insulin (P = .05). Ferritin was the only biomarker that was reduced on both week 16 and week 52.
“If you want to be a purist, this was a negative trial,” said Dr. Garshick, because apremilast was not found to decrease vascular inflammation; however, he noted that the biomarker changes “were hopeful secondary endpoints.” It could be, he said, that another outcome measure may be better able to show changes in vascular inflammation compared with FDG. “It’s always hard to figure out what a good surrogate endpoint is in cardiovascular trials,” he noted, “so it may be that FDG/PET is too noisy or not reliable enough to see the outcome that we want to see.”
Dr. Gelfand reports consulting fees/grants from Amgen, AbbVie, BMS, Boehringer Ingelheim, Janssen Biologics, Novartis Corp, Pfizer, and UCB (DSMB). He serves as the Deputy Editor for the Journal of Investigative Dermatology and the Chief Medical Editor at Healio Psoriatic Disease and receives honoraria for both roles. Dr. Garshick has received consulting fees from AbbVie.
A version of this article first appeared on Medscape.com.
BOSTON – Treatment with , and glucose metabolism, in a study presented at the 2022 American Academy of Dermatology annual meeting.
In the phase 4, open-label, single arm trial, participants also lost subcutaneous and visceral fat after 16 weeks on the oral medication, a phosphodiesterase 4 (PDE4) inhibitor, and maintained that loss at 52 weeks.
People with psoriasis have an increased risk of obesity, type 2 diabetes, and cardiovascular events. Patients with more significant psoriasis “tend to die about 5 years younger than they should, based on their risk factors for mortality,” Joel Gelfand, MD, MSCE, professor of dermatology and epidemiology and vice chair of clinical research in dermatology at the University of Pennsylvania Perelman School of Medicine, Philadelphia, told this news organization.
He led the research and presented the findings at the AAD meeting March 26. “As a result, there has been a keen interest in understanding how psoriasis therapies impact cardiovascular risk, the idea being that by controlling inflammation, you may lower the risk of these patients developing cardiovascular disease over time,” he said.
Previous trials looking at the effect of psoriasis therapies on vascular inflammation “have been, for the most part, inconclusive,” Michael Garshick, MD, a cardiologist at NYU Langone Health, told this news organization. Dr. Garshick was not involved with the research. A 2021 systematic review of psoriasis clinicals trials reported that the tumor necrosis factor (TNF) blocker adalimumab (Humira) and phototherapy had the greatest effect on cardiometabolic markers, while ustekinumab (Stelara), an interleukin (IL)-12 and IL-23 antagonist, was the only treatment that improved vascular inflammation. These variable findings make this area “ripe for study,” noted Dr. Garshick.
To observe how apremilast, which is approved by the FDA for treating psoriasis and psoriatic arthritis, affected vascular inflammation, adiposity, and blood-based cardiometabolic markers, Dr. Gelfand organized an open-label study in adults with moderate-to-severe psoriasis. All participants were 18 years or older, had psoriasis for at least 6 months, and were candidates for systemic therapy. All patients underwent FDG PET/CT scans to assess aortic vascular inflammation and had blood work at baseline. Of the 70 patients originally enrolled in the study, 60 remained in the study at week 16, including 57 who underwent imaging for the second time. Thirty-nine participants remained in the study until week 52, and all except one had another scan.
The average age of participants was 47 years, and their mean BMI was 30. More than 80% of participants were White (83%) and 77% were male. The study population had lived with psoriasis for an average of 16 years and 8 patients also had psoriatic arthritis. At baseline, on average, participants had a Psoriasis Area and Severity Index (PASI) score of 18.62, a dermatology life quality index (DLQI) score of 11.60, and 22% of participants’ BSA (body surface area) were affected. The mean TBRmax, the marker for vascular inflammation, was 1.61.
Treatment responses were as expected for apremilast, with 35% of patients achieving PASI 75 and 65% of participants reporting DLQI scores of 5 or less by 16 weeks. At 52 weeks, 31% of the cohort had achieved PASI 75, and 67% reported DLQI score of 5 or higher. All psoriasis endpoints had improved since baseline (P = .001).
Throughout the study period, there was no significant change in TBRmax. However, in a sensitivity analysis, the 16 patients with a baseline TBRmax of 1.6 or higher had an absolute reduction of 0.21 in TBR by week 52. “That suggests that maybe a subset of people who have higher levels of aortic inflammation at baseline may experience some reduction that portend, potentially, some health benefits over time,” Dr. Gelfand said. “Ultimately, I wouldn’t hang my hat on the finding,” he said, noting that additional research comparing the treatment to placebo is necessary.
Both visceral and subcutaneous adipose tissue (VAT and SAT) decreased by week 16, and this reduction was maintained through week 52. In the first 16 weeks of the study, VAT decreased by 5.32% (P = .0009), and SAT decreased by 5.53% (P = .0005). From baseline to 52 weeks, VAT decreased by 5.52% (P = .0148), and SAT decreased by 5.50% (P = .0096). There were no significant differences between week 16 and week 52 in VAT or SAT.
Of the 68 blood biomarkers analyzed, there were significant decreases in the inflammatory markers ferritin (P = .015) and IL-beta (P = .006), the lipid metabolism biomarker HDL-cholesterol efflux (P = .008), and ketone bodies (P = .006). There were also increases in the inflammatory marker IL-8 (P = .003), the lipid metabolism marker ApoA (P = .05), and insulin (P = .05). Ferritin was the only biomarker that was reduced on both week 16 and week 52.
“If you want to be a purist, this was a negative trial,” said Dr. Garshick, because apremilast was not found to decrease vascular inflammation; however, he noted that the biomarker changes “were hopeful secondary endpoints.” It could be, he said, that another outcome measure may be better able to show changes in vascular inflammation compared with FDG. “It’s always hard to figure out what a good surrogate endpoint is in cardiovascular trials,” he noted, “so it may be that FDG/PET is too noisy or not reliable enough to see the outcome that we want to see.”
Dr. Gelfand reports consulting fees/grants from Amgen, AbbVie, BMS, Boehringer Ingelheim, Janssen Biologics, Novartis Corp, Pfizer, and UCB (DSMB). He serves as the Deputy Editor for the Journal of Investigative Dermatology and the Chief Medical Editor at Healio Psoriatic Disease and receives honoraria for both roles. Dr. Garshick has received consulting fees from AbbVie.
A version of this article first appeared on Medscape.com.
AT AAD 2022
Trichotillomania: What you should know about this common hair-pulling disorder
Trichotillomania is a chronic psychiatric disorder that causes people to repeatedly pull out their own hair. Not only does it result in alopecia with no other underlying causes but it can have significant psychosocial ramifications and rare, but serious, complications. Though the reported prevalence rates are up to approximately 2%, it’s probable that you’ll come upon a patient suffering with this disorder at your practice, if you haven’t already.
To find out more about the best methods for diagnosing and treating this disorder, we spoke with Jon E. Grant, JD, MD, MPH, a leading trichotillomania researcher and part of the department of psychiatry and behavioral neuroscience at the University of Chicago.
Defining trichotillomania
What were the earliest descriptions of trichotillomania in medical literature?
The first real discussion of it probably goes back to Hippocrates, but from a modern medical perspective, discussion began in the 19th century with reports from the French dermatologist François Hallopeau.
They didn’t really call them disorders then – it was long before the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) – but they described this in young men who kept pulling their hair for unclear reasons. These early case reports don’t provide a lot of psychological perspective, but they seem consistent with what we see now.
What are the diagnostic criteria for trichotillomania?
The current DSM-5 criteria are recurrent pulling out of hair, an inability to stop it, the pulling resulting in some noticeable thinning or hair loss, and that it causes some level of distress or some type of impairment in functioning.
At what age do most people experience an onset of symptoms?
Generally speaking, it’s in early adolescence, post puberty, around 12-15 years of age. Having said that, we do see children as young as 1-2 years who are pulling their hair, and we occasionally see somebody far older who is doing it for the first time, a sort of geriatric onset.
Overlap and differences with other disorders
You’ve written that although trichotillomania is grouped with obsessive-compulsive disorder (OCD) in the DSM-5, the thinking around that has recently shifted. Why is that?
At first, it was noticed that many of these people pulled their hair repetitively in an almost ritualized manner, perhaps every night before bed. That looked like a compulsion of OCD.
When DSM-5 came out in 2013, they grouped it with OCD. Yet people shifted to thinking that it’s kind of a cousin of OCD because it has this compulsive quality but doesn’t really have obsessive thinking that drives it. Many people just pull their hair. They’re not even always aware of it: sometimes yes, sometimes no.
We know that it has some links to OCD. You’ll see more OCD in folks with trichotillomania, but it clearly is not just the same as OCD. One of the biggest pieces of evidence for that is that our first-line treatment for OCD – a selective serotonin reuptake inhibitor antidepressant – does not really help hair pulling.
Having said that, if people are looking for help with trichotillomania, they often are best served by therapists and doctors who have a familiarity with OCD and have kept it on their radar over the past couple of decades.
How does trichotillomania overlap with skin picking disorder, which is another condition that you’ve closely researched?
It does have some overlap with skin picking in the sense that it often seems familial. For example, the mother may pull her hair and child picks their skin.
It also has a fair amount of comorbidity with skin picking. Many people who pull will pick a little bit or did at some point. Many people who pick pulled their hair at some point. It seems closely related to nail biting as well.
Studies have also shown that one of the things that runs in the histories of most families of people with trichotillomania might be substance abuse – alcohol or drug addiction.
All of this has led people to believe that there might be subtypes of trichotillomania: one that’s more like an OCD and one that’s more like an addiction. That’s similar to the debate with other mental health conditions, that there are probably multiple types of depression, multiple types of schizophrenia.
Is there a component of this that could be defined as self-harm?
That’s been its own debate. It doesn’t seem to have the same developmental trajectory that we see with self-harm, or even some of the personality features.
However, there may be a small segment of folks with trichotillomania that might more appropriately fit that category. For example, those with family histories of trauma, higher rates of posttraumatic stress disorder, or borderline personality. But it wouldn’t be the majority.
The problem is, if you look at some of the pediatrician data, they often group picking, pulling, and cutting. I think that’s far too all-inclusive.
A gap in clinician education
Are adolescent patients likely to self-report this behavior, or is it something that physicians need to suss out for themselves?
Clearly, if child psychologists, psychiatrists, or pediatricians see young people with patches of alopecia – eyebrows or eyelashes missing, head hair with spots – in addition to a dermatologic assessment, they should simply ask, “Do you pull your hair?”
But it’s interesting that with the internet, young people are much more likely to disclose and actually come forward and tell their parents that they think they have trichotillomania.
I also hear from a lot of the adolescents that they have to educate their doctors about trichotillomania because so often physicians don’t know much about it and will assume that it’s self-injury or just a symptom of anxiety. It’s a little bit of a flip from what we might have seen 20 years ago.
I’ve seen several patients who’ve said, basically, “I’m tired of no professionals seeming to know about this. I shouldn’t have to be educating my doctors about this.” I tell them that I completely agree. It’s a shame because if a doctor doesn’t know about it, then how can they get the appropriate care?
What are the complications that accompany trichotillomania?
A small percentage, maybe about 10%, will ingest their hair, much like people who bite and swallow their fingernails. The concern there is that because hair is nondigestible, it could create an intestinal plug that could rupture and be potentially life-threatening. That makes it all the more important to ask those who pull their hair what they do with the hair once they pull it.
However, with most people, the real problem is with self-esteem. Young people may not want to socialize, go on dates, or do other things they would normally do because of it. In adults, you may find that they’re far more educated than their job allows but don’t want to go to an interview because they don’t want to have somebody sit there and look at them and notice that perhaps they don’t have any eyebrows, or that they’re wearing a wig. Those psychosocial implications are huge for so many people.
Treatment options
In a 2021 study, you showed that nearly one-quarter of people with trichotillomania do naturally recover from it. What characteristics do they seem to have?
It’s interesting because we see natural recovery across many mental health problems: alcohol addition, gambling, OCD. The question then becomes why is that some people can seemingly just stop doing a behavior? Can we learn from those people?
We did see that those who naturally recovered were less likely to have some other mental health comorbidities. It seems like when you have other things such as skin picking or OCD plus trichotillomania, that it probably speaks to something that perhaps synergistically is keeping it going. But this is just a first study; learning how to harness and understand it is the next step.
What’s the goal of treating trichotillomania?
The desired goal is zero pulling. The realistic goal is more likely significantly reduced pulling that then leads to greater function in life, greater quality-of-life.
One doesn’t have to go from 100 to 0 in order to do that. I always tell people that maybe every now and then, every few months, when something is going on in life, you might find yourself pulling a hair or two. That’s okay. If you’re not pulling every day and it’s significantly reduced, we’ll call that a success. I think that setting reasonable goals at this point is really important.
And what would the treatment pathway look like for most patients?
The standard approach is probably some type of habit-reversal therapy, of which there have been many variants over the years. It involves doing something different with your hand, identifying the triggers that may set you off, and then doing something in response to those triggers that is not pulling and might neutralize whatever that anxious or stressed feeling is. That could be different with each person.
At this point, there is no drug approved by the U.S. Food and Drug Administration for trichotillomania. Our best approaches have included N-acetylcysteine, a glutamate modulator, which we’ve done research in.
That’s kind of a go-to option for people because its side-effect profile is generally innocuous. The data show that it could be beneficial in many people with very few, if any, side effects. That would be one “medication,” although it’s actually an over-the-counter vitamin. But we’re constantly looking for better and better treatments.
Do you have any final advice for clinicians or researchers?
Given how common it is, I don’t think clinicians should just see it as an innocuous little habit that people should be able to stop on their own. Clinicians should educate themselves about trichotillomania and know where the person should get the appropriate care.
From the research perspective, given the fact that we see this in animals of multiple species – that they overgroom – this seems to be deeply ingrained in us as animals. So when it comes to the underlying neuroscience, people should pay more attention because it probably has a lot to do with our understanding of habit and compulsive behaviors. It arguably can cut across a lot of different behaviors.
A version of this article first appeared on Medscape.com.
Trichotillomania is a chronic psychiatric disorder that causes people to repeatedly pull out their own hair. Not only does it result in alopecia with no other underlying causes but it can have significant psychosocial ramifications and rare, but serious, complications. Though the reported prevalence rates are up to approximately 2%, it’s probable that you’ll come upon a patient suffering with this disorder at your practice, if you haven’t already.
To find out more about the best methods for diagnosing and treating this disorder, we spoke with Jon E. Grant, JD, MD, MPH, a leading trichotillomania researcher and part of the department of psychiatry and behavioral neuroscience at the University of Chicago.
Defining trichotillomania
What were the earliest descriptions of trichotillomania in medical literature?
The first real discussion of it probably goes back to Hippocrates, but from a modern medical perspective, discussion began in the 19th century with reports from the French dermatologist François Hallopeau.
They didn’t really call them disorders then – it was long before the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) – but they described this in young men who kept pulling their hair for unclear reasons. These early case reports don’t provide a lot of psychological perspective, but they seem consistent with what we see now.
What are the diagnostic criteria for trichotillomania?
The current DSM-5 criteria are recurrent pulling out of hair, an inability to stop it, the pulling resulting in some noticeable thinning or hair loss, and that it causes some level of distress or some type of impairment in functioning.
At what age do most people experience an onset of symptoms?
Generally speaking, it’s in early adolescence, post puberty, around 12-15 years of age. Having said that, we do see children as young as 1-2 years who are pulling their hair, and we occasionally see somebody far older who is doing it for the first time, a sort of geriatric onset.
Overlap and differences with other disorders
You’ve written that although trichotillomania is grouped with obsessive-compulsive disorder (OCD) in the DSM-5, the thinking around that has recently shifted. Why is that?
At first, it was noticed that many of these people pulled their hair repetitively in an almost ritualized manner, perhaps every night before bed. That looked like a compulsion of OCD.
When DSM-5 came out in 2013, they grouped it with OCD. Yet people shifted to thinking that it’s kind of a cousin of OCD because it has this compulsive quality but doesn’t really have obsessive thinking that drives it. Many people just pull their hair. They’re not even always aware of it: sometimes yes, sometimes no.
We know that it has some links to OCD. You’ll see more OCD in folks with trichotillomania, but it clearly is not just the same as OCD. One of the biggest pieces of evidence for that is that our first-line treatment for OCD – a selective serotonin reuptake inhibitor antidepressant – does not really help hair pulling.
Having said that, if people are looking for help with trichotillomania, they often are best served by therapists and doctors who have a familiarity with OCD and have kept it on their radar over the past couple of decades.
How does trichotillomania overlap with skin picking disorder, which is another condition that you’ve closely researched?
It does have some overlap with skin picking in the sense that it often seems familial. For example, the mother may pull her hair and child picks their skin.
It also has a fair amount of comorbidity with skin picking. Many people who pull will pick a little bit or did at some point. Many people who pick pulled their hair at some point. It seems closely related to nail biting as well.
Studies have also shown that one of the things that runs in the histories of most families of people with trichotillomania might be substance abuse – alcohol or drug addiction.
All of this has led people to believe that there might be subtypes of trichotillomania: one that’s more like an OCD and one that’s more like an addiction. That’s similar to the debate with other mental health conditions, that there are probably multiple types of depression, multiple types of schizophrenia.
Is there a component of this that could be defined as self-harm?
That’s been its own debate. It doesn’t seem to have the same developmental trajectory that we see with self-harm, or even some of the personality features.
However, there may be a small segment of folks with trichotillomania that might more appropriately fit that category. For example, those with family histories of trauma, higher rates of posttraumatic stress disorder, or borderline personality. But it wouldn’t be the majority.
The problem is, if you look at some of the pediatrician data, they often group picking, pulling, and cutting. I think that’s far too all-inclusive.
A gap in clinician education
Are adolescent patients likely to self-report this behavior, or is it something that physicians need to suss out for themselves?
Clearly, if child psychologists, psychiatrists, or pediatricians see young people with patches of alopecia – eyebrows or eyelashes missing, head hair with spots – in addition to a dermatologic assessment, they should simply ask, “Do you pull your hair?”
But it’s interesting that with the internet, young people are much more likely to disclose and actually come forward and tell their parents that they think they have trichotillomania.
I also hear from a lot of the adolescents that they have to educate their doctors about trichotillomania because so often physicians don’t know much about it and will assume that it’s self-injury or just a symptom of anxiety. It’s a little bit of a flip from what we might have seen 20 years ago.
I’ve seen several patients who’ve said, basically, “I’m tired of no professionals seeming to know about this. I shouldn’t have to be educating my doctors about this.” I tell them that I completely agree. It’s a shame because if a doctor doesn’t know about it, then how can they get the appropriate care?
What are the complications that accompany trichotillomania?
A small percentage, maybe about 10%, will ingest their hair, much like people who bite and swallow their fingernails. The concern there is that because hair is nondigestible, it could create an intestinal plug that could rupture and be potentially life-threatening. That makes it all the more important to ask those who pull their hair what they do with the hair once they pull it.
However, with most people, the real problem is with self-esteem. Young people may not want to socialize, go on dates, or do other things they would normally do because of it. In adults, you may find that they’re far more educated than their job allows but don’t want to go to an interview because they don’t want to have somebody sit there and look at them and notice that perhaps they don’t have any eyebrows, or that they’re wearing a wig. Those psychosocial implications are huge for so many people.
Treatment options
In a 2021 study, you showed that nearly one-quarter of people with trichotillomania do naturally recover from it. What characteristics do they seem to have?
It’s interesting because we see natural recovery across many mental health problems: alcohol addition, gambling, OCD. The question then becomes why is that some people can seemingly just stop doing a behavior? Can we learn from those people?
We did see that those who naturally recovered were less likely to have some other mental health comorbidities. It seems like when you have other things such as skin picking or OCD plus trichotillomania, that it probably speaks to something that perhaps synergistically is keeping it going. But this is just a first study; learning how to harness and understand it is the next step.
What’s the goal of treating trichotillomania?
The desired goal is zero pulling. The realistic goal is more likely significantly reduced pulling that then leads to greater function in life, greater quality-of-life.
One doesn’t have to go from 100 to 0 in order to do that. I always tell people that maybe every now and then, every few months, when something is going on in life, you might find yourself pulling a hair or two. That’s okay. If you’re not pulling every day and it’s significantly reduced, we’ll call that a success. I think that setting reasonable goals at this point is really important.
And what would the treatment pathway look like for most patients?
The standard approach is probably some type of habit-reversal therapy, of which there have been many variants over the years. It involves doing something different with your hand, identifying the triggers that may set you off, and then doing something in response to those triggers that is not pulling and might neutralize whatever that anxious or stressed feeling is. That could be different with each person.
At this point, there is no drug approved by the U.S. Food and Drug Administration for trichotillomania. Our best approaches have included N-acetylcysteine, a glutamate modulator, which we’ve done research in.
That’s kind of a go-to option for people because its side-effect profile is generally innocuous. The data show that it could be beneficial in many people with very few, if any, side effects. That would be one “medication,” although it’s actually an over-the-counter vitamin. But we’re constantly looking for better and better treatments.
Do you have any final advice for clinicians or researchers?
Given how common it is, I don’t think clinicians should just see it as an innocuous little habit that people should be able to stop on their own. Clinicians should educate themselves about trichotillomania and know where the person should get the appropriate care.
From the research perspective, given the fact that we see this in animals of multiple species – that they overgroom – this seems to be deeply ingrained in us as animals. So when it comes to the underlying neuroscience, people should pay more attention because it probably has a lot to do with our understanding of habit and compulsive behaviors. It arguably can cut across a lot of different behaviors.
A version of this article first appeared on Medscape.com.
Trichotillomania is a chronic psychiatric disorder that causes people to repeatedly pull out their own hair. Not only does it result in alopecia with no other underlying causes but it can have significant psychosocial ramifications and rare, but serious, complications. Though the reported prevalence rates are up to approximately 2%, it’s probable that you’ll come upon a patient suffering with this disorder at your practice, if you haven’t already.
To find out more about the best methods for diagnosing and treating this disorder, we spoke with Jon E. Grant, JD, MD, MPH, a leading trichotillomania researcher and part of the department of psychiatry and behavioral neuroscience at the University of Chicago.
Defining trichotillomania
What were the earliest descriptions of trichotillomania in medical literature?
The first real discussion of it probably goes back to Hippocrates, but from a modern medical perspective, discussion began in the 19th century with reports from the French dermatologist François Hallopeau.
They didn’t really call them disorders then – it was long before the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) – but they described this in young men who kept pulling their hair for unclear reasons. These early case reports don’t provide a lot of psychological perspective, but they seem consistent with what we see now.
What are the diagnostic criteria for trichotillomania?
The current DSM-5 criteria are recurrent pulling out of hair, an inability to stop it, the pulling resulting in some noticeable thinning or hair loss, and that it causes some level of distress or some type of impairment in functioning.
At what age do most people experience an onset of symptoms?
Generally speaking, it’s in early adolescence, post puberty, around 12-15 years of age. Having said that, we do see children as young as 1-2 years who are pulling their hair, and we occasionally see somebody far older who is doing it for the first time, a sort of geriatric onset.
Overlap and differences with other disorders
You’ve written that although trichotillomania is grouped with obsessive-compulsive disorder (OCD) in the DSM-5, the thinking around that has recently shifted. Why is that?
At first, it was noticed that many of these people pulled their hair repetitively in an almost ritualized manner, perhaps every night before bed. That looked like a compulsion of OCD.
When DSM-5 came out in 2013, they grouped it with OCD. Yet people shifted to thinking that it’s kind of a cousin of OCD because it has this compulsive quality but doesn’t really have obsessive thinking that drives it. Many people just pull their hair. They’re not even always aware of it: sometimes yes, sometimes no.
We know that it has some links to OCD. You’ll see more OCD in folks with trichotillomania, but it clearly is not just the same as OCD. One of the biggest pieces of evidence for that is that our first-line treatment for OCD – a selective serotonin reuptake inhibitor antidepressant – does not really help hair pulling.
Having said that, if people are looking for help with trichotillomania, they often are best served by therapists and doctors who have a familiarity with OCD and have kept it on their radar over the past couple of decades.
How does trichotillomania overlap with skin picking disorder, which is another condition that you’ve closely researched?
It does have some overlap with skin picking in the sense that it often seems familial. For example, the mother may pull her hair and child picks their skin.
It also has a fair amount of comorbidity with skin picking. Many people who pull will pick a little bit or did at some point. Many people who pick pulled their hair at some point. It seems closely related to nail biting as well.
Studies have also shown that one of the things that runs in the histories of most families of people with trichotillomania might be substance abuse – alcohol or drug addiction.
All of this has led people to believe that there might be subtypes of trichotillomania: one that’s more like an OCD and one that’s more like an addiction. That’s similar to the debate with other mental health conditions, that there are probably multiple types of depression, multiple types of schizophrenia.
Is there a component of this that could be defined as self-harm?
That’s been its own debate. It doesn’t seem to have the same developmental trajectory that we see with self-harm, or even some of the personality features.
However, there may be a small segment of folks with trichotillomania that might more appropriately fit that category. For example, those with family histories of trauma, higher rates of posttraumatic stress disorder, or borderline personality. But it wouldn’t be the majority.
The problem is, if you look at some of the pediatrician data, they often group picking, pulling, and cutting. I think that’s far too all-inclusive.
A gap in clinician education
Are adolescent patients likely to self-report this behavior, or is it something that physicians need to suss out for themselves?
Clearly, if child psychologists, psychiatrists, or pediatricians see young people with patches of alopecia – eyebrows or eyelashes missing, head hair with spots – in addition to a dermatologic assessment, they should simply ask, “Do you pull your hair?”
But it’s interesting that with the internet, young people are much more likely to disclose and actually come forward and tell their parents that they think they have trichotillomania.
I also hear from a lot of the adolescents that they have to educate their doctors about trichotillomania because so often physicians don’t know much about it and will assume that it’s self-injury or just a symptom of anxiety. It’s a little bit of a flip from what we might have seen 20 years ago.
I’ve seen several patients who’ve said, basically, “I’m tired of no professionals seeming to know about this. I shouldn’t have to be educating my doctors about this.” I tell them that I completely agree. It’s a shame because if a doctor doesn’t know about it, then how can they get the appropriate care?
What are the complications that accompany trichotillomania?
A small percentage, maybe about 10%, will ingest their hair, much like people who bite and swallow their fingernails. The concern there is that because hair is nondigestible, it could create an intestinal plug that could rupture and be potentially life-threatening. That makes it all the more important to ask those who pull their hair what they do with the hair once they pull it.
However, with most people, the real problem is with self-esteem. Young people may not want to socialize, go on dates, or do other things they would normally do because of it. In adults, you may find that they’re far more educated than their job allows but don’t want to go to an interview because they don’t want to have somebody sit there and look at them and notice that perhaps they don’t have any eyebrows, or that they’re wearing a wig. Those psychosocial implications are huge for so many people.
Treatment options
In a 2021 study, you showed that nearly one-quarter of people with trichotillomania do naturally recover from it. What characteristics do they seem to have?
It’s interesting because we see natural recovery across many mental health problems: alcohol addition, gambling, OCD. The question then becomes why is that some people can seemingly just stop doing a behavior? Can we learn from those people?
We did see that those who naturally recovered were less likely to have some other mental health comorbidities. It seems like when you have other things such as skin picking or OCD plus trichotillomania, that it probably speaks to something that perhaps synergistically is keeping it going. But this is just a first study; learning how to harness and understand it is the next step.
What’s the goal of treating trichotillomania?
The desired goal is zero pulling. The realistic goal is more likely significantly reduced pulling that then leads to greater function in life, greater quality-of-life.
One doesn’t have to go from 100 to 0 in order to do that. I always tell people that maybe every now and then, every few months, when something is going on in life, you might find yourself pulling a hair or two. That’s okay. If you’re not pulling every day and it’s significantly reduced, we’ll call that a success. I think that setting reasonable goals at this point is really important.
And what would the treatment pathway look like for most patients?
The standard approach is probably some type of habit-reversal therapy, of which there have been many variants over the years. It involves doing something different with your hand, identifying the triggers that may set you off, and then doing something in response to those triggers that is not pulling and might neutralize whatever that anxious or stressed feeling is. That could be different with each person.
At this point, there is no drug approved by the U.S. Food and Drug Administration for trichotillomania. Our best approaches have included N-acetylcysteine, a glutamate modulator, which we’ve done research in.
That’s kind of a go-to option for people because its side-effect profile is generally innocuous. The data show that it could be beneficial in many people with very few, if any, side effects. That would be one “medication,” although it’s actually an over-the-counter vitamin. But we’re constantly looking for better and better treatments.
Do you have any final advice for clinicians or researchers?
Given how common it is, I don’t think clinicians should just see it as an innocuous little habit that people should be able to stop on their own. Clinicians should educate themselves about trichotillomania and know where the person should get the appropriate care.
From the research perspective, given the fact that we see this in animals of multiple species – that they overgroom – this seems to be deeply ingrained in us as animals. So when it comes to the underlying neuroscience, people should pay more attention because it probably has a lot to do with our understanding of habit and compulsive behaviors. It arguably can cut across a lot of different behaviors.
A version of this article first appeared on Medscape.com.
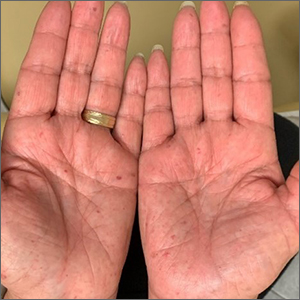
Bilateral palmar rash
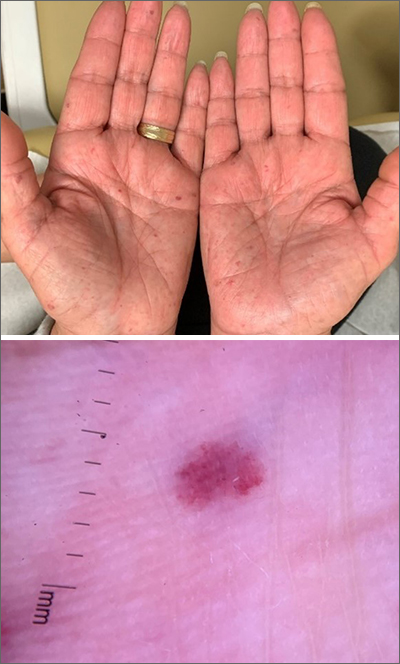
A biopsy was performed and the pathology report showed ectatic, thin-walled vessels consistent with telangiectasias. There were no other inflammatory, infectious, or malignant changes.
Telangiectasias are caused by permanent dilatation of subpapillary plexus end vessels. Unlike petechiae and angiomata, telangiectasias blanch with pressure. They usually manifest as small, bright red, nonpulsatile vascular lesions with a fine, netlike pattern on the surface of the skin. Telangiectasis can affect many organs (eg, intestines, bladder, brain, eyes) and may occur in patients with certain genetic disorders and environmental exposures (eg, radiation).1
Palmar telangiectasias are specifically associated with hereditary hemorrhagic telangiectasia, dermatomyositis, Grave disease, CREST syndrome, systemic lupus erythematosus, and smoking.2 Sun exposure and smoking are the main risk factors for the development of telangiectasias.1
This patient had no history of autoimmune disease or hyperthyroidism, and no one in her family had telangiectasis. Thus, the likely cause of her lesions was smoking. While the pathophysiology is not fully understood, it is likely related to the vasoconstrictive quality of nicotine, causing ischemia in the dermis. This chronic, low-grade ischemia may trigger the compensatory development of telangiectasias.2
This patient was informed that her telangiectasias were most likely caused by her smoking and that the lesions themselves did not require treatment. She was encouraged to continue her smoking cessation efforts with her primary care provider.
Photos courtesy of Daniel Stulberg, MD. Text courtesy of Mia MJ Coleman, BA, BS, University of New Mexico School of Medicine, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Schieving JH, Shoenaker MHD, Weemaes CM, et al. Telangiectasias: Small lesions referring to serious disorders. Eur J Paediatr Neurol. 2017;21:807-815. doi: 10.1016/j.ejpn.2017.07.016
2. Levi A, Shechter R, Lapidoth M, et al. Palmar telangiectasias: a cutaneous sign for smoking. Dermatology. 2017;233:390-395. doi: 10.1159/000481855

A biopsy was performed and the pathology report showed ectatic, thin-walled vessels consistent with telangiectasias. There were no other inflammatory, infectious, or malignant changes.
Telangiectasias are caused by permanent dilatation of subpapillary plexus end vessels. Unlike petechiae and angiomata, telangiectasias blanch with pressure. They usually manifest as small, bright red, nonpulsatile vascular lesions with a fine, netlike pattern on the surface of the skin. Telangiectasis can affect many organs (eg, intestines, bladder, brain, eyes) and may occur in patients with certain genetic disorders and environmental exposures (eg, radiation).1
Palmar telangiectasias are specifically associated with hereditary hemorrhagic telangiectasia, dermatomyositis, Grave disease, CREST syndrome, systemic lupus erythematosus, and smoking.2 Sun exposure and smoking are the main risk factors for the development of telangiectasias.1
This patient had no history of autoimmune disease or hyperthyroidism, and no one in her family had telangiectasis. Thus, the likely cause of her lesions was smoking. While the pathophysiology is not fully understood, it is likely related to the vasoconstrictive quality of nicotine, causing ischemia in the dermis. This chronic, low-grade ischemia may trigger the compensatory development of telangiectasias.2
This patient was informed that her telangiectasias were most likely caused by her smoking and that the lesions themselves did not require treatment. She was encouraged to continue her smoking cessation efforts with her primary care provider.
Photos courtesy of Daniel Stulberg, MD. Text courtesy of Mia MJ Coleman, BA, BS, University of New Mexico School of Medicine, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

A biopsy was performed and the pathology report showed ectatic, thin-walled vessels consistent with telangiectasias. There were no other inflammatory, infectious, or malignant changes.
Telangiectasias are caused by permanent dilatation of subpapillary plexus end vessels. Unlike petechiae and angiomata, telangiectasias blanch with pressure. They usually manifest as small, bright red, nonpulsatile vascular lesions with a fine, netlike pattern on the surface of the skin. Telangiectasis can affect many organs (eg, intestines, bladder, brain, eyes) and may occur in patients with certain genetic disorders and environmental exposures (eg, radiation).1
Palmar telangiectasias are specifically associated with hereditary hemorrhagic telangiectasia, dermatomyositis, Grave disease, CREST syndrome, systemic lupus erythematosus, and smoking.2 Sun exposure and smoking are the main risk factors for the development of telangiectasias.1
This patient had no history of autoimmune disease or hyperthyroidism, and no one in her family had telangiectasis. Thus, the likely cause of her lesions was smoking. While the pathophysiology is not fully understood, it is likely related to the vasoconstrictive quality of nicotine, causing ischemia in the dermis. This chronic, low-grade ischemia may trigger the compensatory development of telangiectasias.2
This patient was informed that her telangiectasias were most likely caused by her smoking and that the lesions themselves did not require treatment. She was encouraged to continue her smoking cessation efforts with her primary care provider.
Photos courtesy of Daniel Stulberg, MD. Text courtesy of Mia MJ Coleman, BA, BS, University of New Mexico School of Medicine, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Schieving JH, Shoenaker MHD, Weemaes CM, et al. Telangiectasias: Small lesions referring to serious disorders. Eur J Paediatr Neurol. 2017;21:807-815. doi: 10.1016/j.ejpn.2017.07.016
2. Levi A, Shechter R, Lapidoth M, et al. Palmar telangiectasias: a cutaneous sign for smoking. Dermatology. 2017;233:390-395. doi: 10.1159/000481855
1. Schieving JH, Shoenaker MHD, Weemaes CM, et al. Telangiectasias: Small lesions referring to serious disorders. Eur J Paediatr Neurol. 2017;21:807-815. doi: 10.1016/j.ejpn.2017.07.016
2. Levi A, Shechter R, Lapidoth M, et al. Palmar telangiectasias: a cutaneous sign for smoking. Dermatology. 2017;233:390-395. doi: 10.1159/000481855
Photoprotection strategies for melasma are increasing
BOSTON – Untinted chemical sunscreens on the market are not sufficient to protect the skin from the effects of visible light, complicating sun protection efforts for patients with melasma and other conditions aggravated by sun exposure, according to Henry W. Lim, MD.
A , Dr. Lim, former chair of the department of dermatology at Henry Ford Health, Detroit, said at the annual meeting of the American Academy of Dermatology. Tinted sunscreens contain iron oxides; some also contain pigmentary titanium dioxide.
“Black, red, and yellow iron oxide all reflect visible light,” he added, noting that currently, there are no regulations as to how tinted sunscreens are marketed, making it difficult for practicing clinicians to advise patients about what products to choose. However, he said, “unlike ‘SPF’ and ‘broad spectrum’ labeling, there is no specific guidance on tinted sunscreens. “ ‘Universal’ shade is a good start but might not be ideal for users with very fair or deep skin tones,” he noted.
In December 2021, a guide to tinted sunscreens, written by Dr. Lim and colleagues, was published, recommending that consumers choose a product that contains iron oxides, is labeled as broad spectrum, and has an SPF of at least 30.
A comprehensive list of 54 tinted sunscreens with an SPF of 30 or greater that contain iron oxide is also available . The authors of the guide contributed to this resource, which lists sunscreens by average price per ounce.
At the meeting, Dr. Lim highlighted tinted sunscreens that cost about $20 or less per ounce. They include Supergoop 100% Mineral CC Cream (SPF 50); Bare Republic Mineral Tinted Face Sunscreen Lotion (SPF 30); CeraVe Hydrating Sunscreen with Sheer Tint (SPF 30); Tizo Ultra Zinc Body & Face Sunscreen (SPF 40); Vichy Capital Soleil Tinted Face Mineral Sunscreen (SPF 60); EltaMD UV Elements Tinted (SPF 44); La Roche-Posay Anthelios Ultra-Light Tinted Mineral (SPF 50), SkinMedica Essential Defense Mineral Shield (SPF 32), ISDIN Eryfotona Ageless Ultralight Tinted Mineral Sunscreen (SPF 50), and SkinCeuticals Physical Fusion UV Defense (SPF 50).
Sunscreens with antioxidants
Sunscreens with biologically active antioxidants may be another option for patients with melasma. A proof-of-concept study that Dr. Lim and colleagues conducted in 20 patients found that application of a blend of topical antioxidants (2%) was associated with less erythema at the application sites among those with skin phototypes I-III and less pigmentation at the application sites among those with skin phototypes IV-VI after exposure to visible light and UVA-1, compared with controls.
Certain antioxidants have been added to sunscreens currently on the market, including niacinamide (vitamin B3), licochalcone A, carotenoids (beta-carotene), vitamin E, vitamin C, glycyrrhetinic acid, and diethylhexyl syringylidenemalonate.
A recently published paper on the role of antioxidants and free radical quenchers in protecting skin from visible light referred to unpublished data from Dr. Lim (the first author) and colleagues, which demonstrated a significant reduction in visual light–induced hyperpigmentation on skin with sunscreen that contained the antioxidants vitamin E, vitamin C, diethylhexyl syringylidenemalonate, licochalcone A, and a glycyrrhetinic acid, compared with sunscreen that had no antioxidants.
Novel filters
Another emerging option is sunscreen with new filters that cover UVA-1 and visible light. In a randomized, controlled trial of 19 patients, researchers evaluated the addition of methoxypropylamino cyclohexenylidene ethoxyethylcyanoacetate (MCE) absorber, a new UVA-1 filter known as Mexoryl 400, which has a peak absorption of 385 nm, to a sunscreen formulation.
“Currently, peak absorption in the U.S. is with avobenzone, which peaks at about 357 nm,” but MCE “covers a longer spectrum of UVA-1,” Dr. Lim said. The researchers found that the addition of MCE reduced UVA-1-induced dermal and epidermal alterations at cellular, biochemical, and molecular levels; and decreased UVA-1-induced pigmentation.
Another relatively new filter, phenylene bis-diphenyltriazine (also known as TriAsorB) not only protects against UVA but it extends into the blue light portion of visible light, according to a recently published paper. According to a press release from Pierre Fabre, which has developed the filter, studies have shown that TriAsorB is not toxic for three key species of marine biodiversity: a coral species, a phytoplankton species, and a zooplankton.
This filter and MCE are available in Europe but not in the United States.
Dr. Lim reported that he is an investigator for Incyte, L’Oréal, Pfizer, and the Patient-Centered Outcomes Research Institute.
BOSTON – Untinted chemical sunscreens on the market are not sufficient to protect the skin from the effects of visible light, complicating sun protection efforts for patients with melasma and other conditions aggravated by sun exposure, according to Henry W. Lim, MD.
A , Dr. Lim, former chair of the department of dermatology at Henry Ford Health, Detroit, said at the annual meeting of the American Academy of Dermatology. Tinted sunscreens contain iron oxides; some also contain pigmentary titanium dioxide.
“Black, red, and yellow iron oxide all reflect visible light,” he added, noting that currently, there are no regulations as to how tinted sunscreens are marketed, making it difficult for practicing clinicians to advise patients about what products to choose. However, he said, “unlike ‘SPF’ and ‘broad spectrum’ labeling, there is no specific guidance on tinted sunscreens. “ ‘Universal’ shade is a good start but might not be ideal for users with very fair or deep skin tones,” he noted.
In December 2021, a guide to tinted sunscreens, written by Dr. Lim and colleagues, was published, recommending that consumers choose a product that contains iron oxides, is labeled as broad spectrum, and has an SPF of at least 30.
A comprehensive list of 54 tinted sunscreens with an SPF of 30 or greater that contain iron oxide is also available . The authors of the guide contributed to this resource, which lists sunscreens by average price per ounce.
At the meeting, Dr. Lim highlighted tinted sunscreens that cost about $20 or less per ounce. They include Supergoop 100% Mineral CC Cream (SPF 50); Bare Republic Mineral Tinted Face Sunscreen Lotion (SPF 30); CeraVe Hydrating Sunscreen with Sheer Tint (SPF 30); Tizo Ultra Zinc Body & Face Sunscreen (SPF 40); Vichy Capital Soleil Tinted Face Mineral Sunscreen (SPF 60); EltaMD UV Elements Tinted (SPF 44); La Roche-Posay Anthelios Ultra-Light Tinted Mineral (SPF 50), SkinMedica Essential Defense Mineral Shield (SPF 32), ISDIN Eryfotona Ageless Ultralight Tinted Mineral Sunscreen (SPF 50), and SkinCeuticals Physical Fusion UV Defense (SPF 50).
Sunscreens with antioxidants
Sunscreens with biologically active antioxidants may be another option for patients with melasma. A proof-of-concept study that Dr. Lim and colleagues conducted in 20 patients found that application of a blend of topical antioxidants (2%) was associated with less erythema at the application sites among those with skin phototypes I-III and less pigmentation at the application sites among those with skin phototypes IV-VI after exposure to visible light and UVA-1, compared with controls.
Certain antioxidants have been added to sunscreens currently on the market, including niacinamide (vitamin B3), licochalcone A, carotenoids (beta-carotene), vitamin E, vitamin C, glycyrrhetinic acid, and diethylhexyl syringylidenemalonate.
A recently published paper on the role of antioxidants and free radical quenchers in protecting skin from visible light referred to unpublished data from Dr. Lim (the first author) and colleagues, which demonstrated a significant reduction in visual light–induced hyperpigmentation on skin with sunscreen that contained the antioxidants vitamin E, vitamin C, diethylhexyl syringylidenemalonate, licochalcone A, and a glycyrrhetinic acid, compared with sunscreen that had no antioxidants.
Novel filters
Another emerging option is sunscreen with new filters that cover UVA-1 and visible light. In a randomized, controlled trial of 19 patients, researchers evaluated the addition of methoxypropylamino cyclohexenylidene ethoxyethylcyanoacetate (MCE) absorber, a new UVA-1 filter known as Mexoryl 400, which has a peak absorption of 385 nm, to a sunscreen formulation.
“Currently, peak absorption in the U.S. is with avobenzone, which peaks at about 357 nm,” but MCE “covers a longer spectrum of UVA-1,” Dr. Lim said. The researchers found that the addition of MCE reduced UVA-1-induced dermal and epidermal alterations at cellular, biochemical, and molecular levels; and decreased UVA-1-induced pigmentation.
Another relatively new filter, phenylene bis-diphenyltriazine (also known as TriAsorB) not only protects against UVA but it extends into the blue light portion of visible light, according to a recently published paper. According to a press release from Pierre Fabre, which has developed the filter, studies have shown that TriAsorB is not toxic for three key species of marine biodiversity: a coral species, a phytoplankton species, and a zooplankton.
This filter and MCE are available in Europe but not in the United States.
Dr. Lim reported that he is an investigator for Incyte, L’Oréal, Pfizer, and the Patient-Centered Outcomes Research Institute.
BOSTON – Untinted chemical sunscreens on the market are not sufficient to protect the skin from the effects of visible light, complicating sun protection efforts for patients with melasma and other conditions aggravated by sun exposure, according to Henry W. Lim, MD.
A , Dr. Lim, former chair of the department of dermatology at Henry Ford Health, Detroit, said at the annual meeting of the American Academy of Dermatology. Tinted sunscreens contain iron oxides; some also contain pigmentary titanium dioxide.
“Black, red, and yellow iron oxide all reflect visible light,” he added, noting that currently, there are no regulations as to how tinted sunscreens are marketed, making it difficult for practicing clinicians to advise patients about what products to choose. However, he said, “unlike ‘SPF’ and ‘broad spectrum’ labeling, there is no specific guidance on tinted sunscreens. “ ‘Universal’ shade is a good start but might not be ideal for users with very fair or deep skin tones,” he noted.
In December 2021, a guide to tinted sunscreens, written by Dr. Lim and colleagues, was published, recommending that consumers choose a product that contains iron oxides, is labeled as broad spectrum, and has an SPF of at least 30.
A comprehensive list of 54 tinted sunscreens with an SPF of 30 or greater that contain iron oxide is also available . The authors of the guide contributed to this resource, which lists sunscreens by average price per ounce.
At the meeting, Dr. Lim highlighted tinted sunscreens that cost about $20 or less per ounce. They include Supergoop 100% Mineral CC Cream (SPF 50); Bare Republic Mineral Tinted Face Sunscreen Lotion (SPF 30); CeraVe Hydrating Sunscreen with Sheer Tint (SPF 30); Tizo Ultra Zinc Body & Face Sunscreen (SPF 40); Vichy Capital Soleil Tinted Face Mineral Sunscreen (SPF 60); EltaMD UV Elements Tinted (SPF 44); La Roche-Posay Anthelios Ultra-Light Tinted Mineral (SPF 50), SkinMedica Essential Defense Mineral Shield (SPF 32), ISDIN Eryfotona Ageless Ultralight Tinted Mineral Sunscreen (SPF 50), and SkinCeuticals Physical Fusion UV Defense (SPF 50).
Sunscreens with antioxidants
Sunscreens with biologically active antioxidants may be another option for patients with melasma. A proof-of-concept study that Dr. Lim and colleagues conducted in 20 patients found that application of a blend of topical antioxidants (2%) was associated with less erythema at the application sites among those with skin phototypes I-III and less pigmentation at the application sites among those with skin phototypes IV-VI after exposure to visible light and UVA-1, compared with controls.
Certain antioxidants have been added to sunscreens currently on the market, including niacinamide (vitamin B3), licochalcone A, carotenoids (beta-carotene), vitamin E, vitamin C, glycyrrhetinic acid, and diethylhexyl syringylidenemalonate.
A recently published paper on the role of antioxidants and free radical quenchers in protecting skin from visible light referred to unpublished data from Dr. Lim (the first author) and colleagues, which demonstrated a significant reduction in visual light–induced hyperpigmentation on skin with sunscreen that contained the antioxidants vitamin E, vitamin C, diethylhexyl syringylidenemalonate, licochalcone A, and a glycyrrhetinic acid, compared with sunscreen that had no antioxidants.
Novel filters
Another emerging option is sunscreen with new filters that cover UVA-1 and visible light. In a randomized, controlled trial of 19 patients, researchers evaluated the addition of methoxypropylamino cyclohexenylidene ethoxyethylcyanoacetate (MCE) absorber, a new UVA-1 filter known as Mexoryl 400, which has a peak absorption of 385 nm, to a sunscreen formulation.
“Currently, peak absorption in the U.S. is with avobenzone, which peaks at about 357 nm,” but MCE “covers a longer spectrum of UVA-1,” Dr. Lim said. The researchers found that the addition of MCE reduced UVA-1-induced dermal and epidermal alterations at cellular, biochemical, and molecular levels; and decreased UVA-1-induced pigmentation.
Another relatively new filter, phenylene bis-diphenyltriazine (also known as TriAsorB) not only protects against UVA but it extends into the blue light portion of visible light, according to a recently published paper. According to a press release from Pierre Fabre, which has developed the filter, studies have shown that TriAsorB is not toxic for three key species of marine biodiversity: a coral species, a phytoplankton species, and a zooplankton.
This filter and MCE are available in Europe but not in the United States.
Dr. Lim reported that he is an investigator for Incyte, L’Oréal, Pfizer, and the Patient-Centered Outcomes Research Institute.
AT AAD 22
Global melanoma incidence high and on the rise
Even by cautious calculations,
An estimated 325,000 people worldwide received a new diagnosis of cutaneous melanoma in 2020, and if present trends continue, the incidence of new cases is predicted to increase by about 50% in 2040, with melanoma deaths expected to rise by almost 70%, Melina Arnold, PhD, from the Cancer Surveillance Branch of the International Agency for Research on Cancer in Lyon, France, and colleagues reported.
“Melanoma is the most lethal form of skin cancer; this epidemiological assessment found a heavy public health and economic burden, and our projections suggest that it will remain so in the coming decades,” they wrote in a study published online in JAMA Dermatology.
In an accompanying editorial, Mavis Obeng-Kusi, MPharm and Ivo Abraham, PhD from the Center for Health Outcomes and PharmacoEconomic Research at the University of Arizona, Tucson, commented that the findings are “sobering,” but may substantially underestimate the gravity of the problem in low- and middle-income countries (LMIC).
“The study by Arnold et al. brings to the fore a public health concern that requires global attention and initiates conversations particularly related to LMIC settings, where the incidence and mortality of melanoma is thought to be minimal and for which preventive measures may be insufficient,” they wrote.
Down Under nations lead
Dr. Arnold and colleagues looked at data on age-standardized melanoma incidence and mortality rates per 100,000 person-years (PY) by country, each of 20 world regions as defined by the United Nations, and according to the UN’s four-tier Human Development Index, which stratifies countries into low-, medium-, high-, and very high–income categories.
As noted previously, the researchers estimated that there were 325,000 new melanoma cases worldwide in 2020 (174,000 cases in males and 151,000 in females). There were 57,000 estimated melanoma deaths the same year (32,000 in males and 25,000 in females.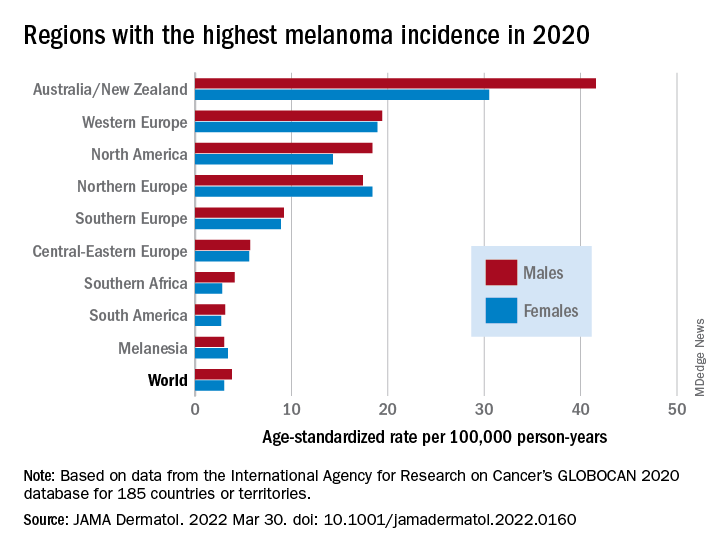
The highest incidence rates were seen in Australia and New Zealand, at 42 per 100,000 PY among males and 31 per 100,000 PY in females, followed by Western Europe with 19 per 100,000 PY in both males and females, North America with 18 and 14 cases per 100,000 PY in males and females respectively, and Northern Europe, with 17 per 100,000 PY in males, and 18 per 100,000 PY in females.
In contrast, in most African and Asian countries melanoma was rare, with rates commonly less than 1 per 100,000 PY, the investigators noted.
The melanoma mortality rate was highest in New Zealand, at 5 per 100,000 PY. Mortality rates worldwide varied less widely than incidence rates. In most other regions of the world, mortality rates were “much lower,” ranging between 0.2-1.0 per 100,000 PY, they wrote.
The authors estimated that, if 2020 rates remain stable, the global burden from melanoma in 2040 will increase to approximately 510,000 new cases and 96,000 deaths.
Public health efforts needed
In their editorial, Ms. Obeng-Kusi and Dr. Abraham pointed out that the study was hampered by the limited availability of cancer data from LMICs, leading the authors to estimate incidence and mortality rates based on proxy data, such as statistical modeling or averaged rates from neighboring countries.
They emphasized the need for going beyond the statistics: “Specific to cutaneous melanoma data, what is most important globally, knowing the exact numbers of cases and deaths or understanding the order of magnitude of the present and future epidemiology? No doubt the latter. Melanoma can be treated more easily if caught at earlier stages.”
Projections such as those provided by Dr. Arnold and colleagues could help to raise awareness of the importance of decreasing exposure to UV radiation, which accounts for three-fourths of all incident melanomas, the editorialists said.
The study was funded in part by a grant to coauthor Anna E. Cust, PhD, MPH. Dr. Cust reported receiving a fellowship from the Australian National Health and Medical Research Council outside the submitted work. Dr. Arnold had no conflicts of interested to disclose. Dr. Abraham reported financial relationships with various entities. Ms. Obeng-Kusi had no disclosures.
Even by cautious calculations,
An estimated 325,000 people worldwide received a new diagnosis of cutaneous melanoma in 2020, and if present trends continue, the incidence of new cases is predicted to increase by about 50% in 2040, with melanoma deaths expected to rise by almost 70%, Melina Arnold, PhD, from the Cancer Surveillance Branch of the International Agency for Research on Cancer in Lyon, France, and colleagues reported.
“Melanoma is the most lethal form of skin cancer; this epidemiological assessment found a heavy public health and economic burden, and our projections suggest that it will remain so in the coming decades,” they wrote in a study published online in JAMA Dermatology.
In an accompanying editorial, Mavis Obeng-Kusi, MPharm and Ivo Abraham, PhD from the Center for Health Outcomes and PharmacoEconomic Research at the University of Arizona, Tucson, commented that the findings are “sobering,” but may substantially underestimate the gravity of the problem in low- and middle-income countries (LMIC).
“The study by Arnold et al. brings to the fore a public health concern that requires global attention and initiates conversations particularly related to LMIC settings, where the incidence and mortality of melanoma is thought to be minimal and for which preventive measures may be insufficient,” they wrote.
Down Under nations lead
Dr. Arnold and colleagues looked at data on age-standardized melanoma incidence and mortality rates per 100,000 person-years (PY) by country, each of 20 world regions as defined by the United Nations, and according to the UN’s four-tier Human Development Index, which stratifies countries into low-, medium-, high-, and very high–income categories.
As noted previously, the researchers estimated that there were 325,000 new melanoma cases worldwide in 2020 (174,000 cases in males and 151,000 in females). There were 57,000 estimated melanoma deaths the same year (32,000 in males and 25,000 in females.
The highest incidence rates were seen in Australia and New Zealand, at 42 per 100,000 PY among males and 31 per 100,000 PY in females, followed by Western Europe with 19 per 100,000 PY in both males and females, North America with 18 and 14 cases per 100,000 PY in males and females respectively, and Northern Europe, with 17 per 100,000 PY in males, and 18 per 100,000 PY in females.
In contrast, in most African and Asian countries melanoma was rare, with rates commonly less than 1 per 100,000 PY, the investigators noted.
The melanoma mortality rate was highest in New Zealand, at 5 per 100,000 PY. Mortality rates worldwide varied less widely than incidence rates. In most other regions of the world, mortality rates were “much lower,” ranging between 0.2-1.0 per 100,000 PY, they wrote.
The authors estimated that, if 2020 rates remain stable, the global burden from melanoma in 2040 will increase to approximately 510,000 new cases and 96,000 deaths.
Public health efforts needed
In their editorial, Ms. Obeng-Kusi and Dr. Abraham pointed out that the study was hampered by the limited availability of cancer data from LMICs, leading the authors to estimate incidence and mortality rates based on proxy data, such as statistical modeling or averaged rates from neighboring countries.
They emphasized the need for going beyond the statistics: “Specific to cutaneous melanoma data, what is most important globally, knowing the exact numbers of cases and deaths or understanding the order of magnitude of the present and future epidemiology? No doubt the latter. Melanoma can be treated more easily if caught at earlier stages.”
Projections such as those provided by Dr. Arnold and colleagues could help to raise awareness of the importance of decreasing exposure to UV radiation, which accounts for three-fourths of all incident melanomas, the editorialists said.
The study was funded in part by a grant to coauthor Anna E. Cust, PhD, MPH. Dr. Cust reported receiving a fellowship from the Australian National Health and Medical Research Council outside the submitted work. Dr. Arnold had no conflicts of interested to disclose. Dr. Abraham reported financial relationships with various entities. Ms. Obeng-Kusi had no disclosures.
Even by cautious calculations,
An estimated 325,000 people worldwide received a new diagnosis of cutaneous melanoma in 2020, and if present trends continue, the incidence of new cases is predicted to increase by about 50% in 2040, with melanoma deaths expected to rise by almost 70%, Melina Arnold, PhD, from the Cancer Surveillance Branch of the International Agency for Research on Cancer in Lyon, France, and colleagues reported.
“Melanoma is the most lethal form of skin cancer; this epidemiological assessment found a heavy public health and economic burden, and our projections suggest that it will remain so in the coming decades,” they wrote in a study published online in JAMA Dermatology.
In an accompanying editorial, Mavis Obeng-Kusi, MPharm and Ivo Abraham, PhD from the Center for Health Outcomes and PharmacoEconomic Research at the University of Arizona, Tucson, commented that the findings are “sobering,” but may substantially underestimate the gravity of the problem in low- and middle-income countries (LMIC).
“The study by Arnold et al. brings to the fore a public health concern that requires global attention and initiates conversations particularly related to LMIC settings, where the incidence and mortality of melanoma is thought to be minimal and for which preventive measures may be insufficient,” they wrote.
Down Under nations lead
Dr. Arnold and colleagues looked at data on age-standardized melanoma incidence and mortality rates per 100,000 person-years (PY) by country, each of 20 world regions as defined by the United Nations, and according to the UN’s four-tier Human Development Index, which stratifies countries into low-, medium-, high-, and very high–income categories.
As noted previously, the researchers estimated that there were 325,000 new melanoma cases worldwide in 2020 (174,000 cases in males and 151,000 in females). There were 57,000 estimated melanoma deaths the same year (32,000 in males and 25,000 in females.
The highest incidence rates were seen in Australia and New Zealand, at 42 per 100,000 PY among males and 31 per 100,000 PY in females, followed by Western Europe with 19 per 100,000 PY in both males and females, North America with 18 and 14 cases per 100,000 PY in males and females respectively, and Northern Europe, with 17 per 100,000 PY in males, and 18 per 100,000 PY in females.
In contrast, in most African and Asian countries melanoma was rare, with rates commonly less than 1 per 100,000 PY, the investigators noted.
The melanoma mortality rate was highest in New Zealand, at 5 per 100,000 PY. Mortality rates worldwide varied less widely than incidence rates. In most other regions of the world, mortality rates were “much lower,” ranging between 0.2-1.0 per 100,000 PY, they wrote.
The authors estimated that, if 2020 rates remain stable, the global burden from melanoma in 2040 will increase to approximately 510,000 new cases and 96,000 deaths.
Public health efforts needed
In their editorial, Ms. Obeng-Kusi and Dr. Abraham pointed out that the study was hampered by the limited availability of cancer data from LMICs, leading the authors to estimate incidence and mortality rates based on proxy data, such as statistical modeling or averaged rates from neighboring countries.
They emphasized the need for going beyond the statistics: “Specific to cutaneous melanoma data, what is most important globally, knowing the exact numbers of cases and deaths or understanding the order of magnitude of the present and future epidemiology? No doubt the latter. Melanoma can be treated more easily if caught at earlier stages.”
Projections such as those provided by Dr. Arnold and colleagues could help to raise awareness of the importance of decreasing exposure to UV radiation, which accounts for three-fourths of all incident melanomas, the editorialists said.
The study was funded in part by a grant to coauthor Anna E. Cust, PhD, MPH. Dr. Cust reported receiving a fellowship from the Australian National Health and Medical Research Council outside the submitted work. Dr. Arnold had no conflicts of interested to disclose. Dr. Abraham reported financial relationships with various entities. Ms. Obeng-Kusi had no disclosures.
FROM JAMA DERMATOLOGY
IV gentamicin improves junctional epidermolysis bullosa in children
Intravenous (JEB) caused by nonsense variants.
The newly generated structural protein persisted during the 3-month randomized clinical trial and was associated with significant wound closure – with no signs of ototoxic effects, nephrotoxic effects, or anti–laminin 332 autoantibody induction, investigators recently reported in JAMA Dermatology.
JEB is a rare, autosomal recessive disorder caused mainly by nonsense variants (i.e., mutations) in the LAMA3, LAMB3, or LAMC2 genes that encode laminin, resulting in widespread blisters and erosions of the skin. Current treatment is limited to supportive management and palliative care, and children with its severe subtype are likely to die within the first year of life.
“With data indicating a robust response to short-term gentamicin treatment and the marked stability of laminin 332, we envision that gentamicin could be delivered as a short-term pulse therapy every 2-3 months for patients with JEB caused by nonsense variants,” the researchers wrote.
Of the five patients, ages 3 months to 10 years, three received 7.5 mg/kg IV gentamicin daily for 14 days, and two received 10 mg/kg daily for 24 days at the University of Southern California, Los Angeles.
All had confirmed nonsense variants in LAMA3 or LAMB3 in one or two alleles, and all had minimal laminin 332 expression at baseline as determined by immunofluorescence. After treatment, each of the children had increased, sustained expression of laminin 332.
The researchers monitored three open wounds in each patient. By 1 month, seven of nine wounds in those receiving the lower-dose therapy and all of the wounds in those receiving the higher-dose therapy showed at least 50% closure. By 3 months, eight of nine wounds in the lower-dose group, and all wounds in the higher-dose group showed greater than 85% closure.
In an interview, senior investigators Mei Chen, PhD, professor of dermatology, and David T. Woodley, MD, professor and chair of dermatology, both at USC, emphasized laminin’s long half-life.“Once these skin structural proteins are generated at the dermal-epidermal junction, they are long-lasting structures, which means the therapy can be pulsed rather than continuously delivered, which can obviate some of the known side effects of the medication,” Dr. Woodley said.
Gentamicin, an aminoglycoside, works as a “read-through therapy,” inducing ribosomal read-through of premature termination codons (PTCs) caused by nonsense mutations. The read-through allows translation to proceed and full-length proteins to be generated.
Gentamicin read-through therapy is also being investigated for recessive dystrophic epidermolysis bullosa (RDEB) attributable to nonsense mutations. The culprit mutations in this form of EB occur in a gene that encodes collagen type VII alpha 1, which, like laminin, is responsible for dermal-epidermal adherence. A clinical trial of intravenous gentamicin for RDEB is ongoing at USC, Dr. Chen said.
EBS-MD case report
It may also have a role in treating epidermolysis bullosa simplex with muscular dystrophy (EBS-MD), according to investigators in Madrid. Their case report, published in JAMA Dermatology, details how two 14-day courses of infused gentamicin therapy were followed by re-expression of plectin in the skin for 4-5 months and mild improvement in symptoms in one patient, a woman in her 30s, with a homozygous nonsense variant in PLEC1.
In an editorial accompanying the two reports, Anna L. Bruckner, MD, MSCS, professor of dermatology, University of Colorado at Denver, Aurora, and colleagues expressed cautious optimism and said that additional research on the feasibility, possible cumulative toxic effects, risk of microbial resistance, and overall clinical relevance is needed.
Still, the “investigators should be applauded for taking advantage of a readily available systemic treatment to target cutaneous and extracutaneous symptoms of patients who have very limited treatment options at this time,” they wrote. While all forms of EB are considered orphan disorders, JEB and EBS-MD have received less research attention than RDEB.
The JEB study evaluated patients with clinical assessments/quality of life surveys and with a validated clinical score that considers skin and mucosae – the Epidermolysis Bullosa Disease Activity and Scarring Index (EBDASI). There were small positive changes in EBDASI scores, but data were incomplete and therefore difficult to interpret.
A “noteworthy” finding, the authors wrote, were improvements in emotions and functioning in two of the children who were eligible given their older ages for assessment with the Skindex-16 quality-of-life survey. The improvements suggest “potential psychosocial benefits” of the gentamicin therapy.
The JEB study was supported in part by grants from the EB Research Partnership and EB Medical Research Foundation and an award from the Congressionally Directed Medical Research Program. In addition to the grants, Dr. Woodley and Dr. Chen reported receiving personal fees from Phoenix Tissue Repair outside of the submitted work. For the EBS-MD case report, the authors reported no disclosures. Dr. Bruckner, corresponding author of the editorial, reported grants from several companies outside the submitted work.
Intravenous (JEB) caused by nonsense variants.
The newly generated structural protein persisted during the 3-month randomized clinical trial and was associated with significant wound closure – with no signs of ototoxic effects, nephrotoxic effects, or anti–laminin 332 autoantibody induction, investigators recently reported in JAMA Dermatology.
JEB is a rare, autosomal recessive disorder caused mainly by nonsense variants (i.e., mutations) in the LAMA3, LAMB3, or LAMC2 genes that encode laminin, resulting in widespread blisters and erosions of the skin. Current treatment is limited to supportive management and palliative care, and children with its severe subtype are likely to die within the first year of life.
“With data indicating a robust response to short-term gentamicin treatment and the marked stability of laminin 332, we envision that gentamicin could be delivered as a short-term pulse therapy every 2-3 months for patients with JEB caused by nonsense variants,” the researchers wrote.
Of the five patients, ages 3 months to 10 years, three received 7.5 mg/kg IV gentamicin daily for 14 days, and two received 10 mg/kg daily for 24 days at the University of Southern California, Los Angeles.
All had confirmed nonsense variants in LAMA3 or LAMB3 in one or two alleles, and all had minimal laminin 332 expression at baseline as determined by immunofluorescence. After treatment, each of the children had increased, sustained expression of laminin 332.
The researchers monitored three open wounds in each patient. By 1 month, seven of nine wounds in those receiving the lower-dose therapy and all of the wounds in those receiving the higher-dose therapy showed at least 50% closure. By 3 months, eight of nine wounds in the lower-dose group, and all wounds in the higher-dose group showed greater than 85% closure.
In an interview, senior investigators Mei Chen, PhD, professor of dermatology, and David T. Woodley, MD, professor and chair of dermatology, both at USC, emphasized laminin’s long half-life.“Once these skin structural proteins are generated at the dermal-epidermal junction, they are long-lasting structures, which means the therapy can be pulsed rather than continuously delivered, which can obviate some of the known side effects of the medication,” Dr. Woodley said.
Gentamicin, an aminoglycoside, works as a “read-through therapy,” inducing ribosomal read-through of premature termination codons (PTCs) caused by nonsense mutations. The read-through allows translation to proceed and full-length proteins to be generated.
Gentamicin read-through therapy is also being investigated for recessive dystrophic epidermolysis bullosa (RDEB) attributable to nonsense mutations. The culprit mutations in this form of EB occur in a gene that encodes collagen type VII alpha 1, which, like laminin, is responsible for dermal-epidermal adherence. A clinical trial of intravenous gentamicin for RDEB is ongoing at USC, Dr. Chen said.
EBS-MD case report
It may also have a role in treating epidermolysis bullosa simplex with muscular dystrophy (EBS-MD), according to investigators in Madrid. Their case report, published in JAMA Dermatology, details how two 14-day courses of infused gentamicin therapy were followed by re-expression of plectin in the skin for 4-5 months and mild improvement in symptoms in one patient, a woman in her 30s, with a homozygous nonsense variant in PLEC1.
In an editorial accompanying the two reports, Anna L. Bruckner, MD, MSCS, professor of dermatology, University of Colorado at Denver, Aurora, and colleagues expressed cautious optimism and said that additional research on the feasibility, possible cumulative toxic effects, risk of microbial resistance, and overall clinical relevance is needed.
Still, the “investigators should be applauded for taking advantage of a readily available systemic treatment to target cutaneous and extracutaneous symptoms of patients who have very limited treatment options at this time,” they wrote. While all forms of EB are considered orphan disorders, JEB and EBS-MD have received less research attention than RDEB.
The JEB study evaluated patients with clinical assessments/quality of life surveys and with a validated clinical score that considers skin and mucosae – the Epidermolysis Bullosa Disease Activity and Scarring Index (EBDASI). There were small positive changes in EBDASI scores, but data were incomplete and therefore difficult to interpret.
A “noteworthy” finding, the authors wrote, were improvements in emotions and functioning in two of the children who were eligible given their older ages for assessment with the Skindex-16 quality-of-life survey. The improvements suggest “potential psychosocial benefits” of the gentamicin therapy.
The JEB study was supported in part by grants from the EB Research Partnership and EB Medical Research Foundation and an award from the Congressionally Directed Medical Research Program. In addition to the grants, Dr. Woodley and Dr. Chen reported receiving personal fees from Phoenix Tissue Repair outside of the submitted work. For the EBS-MD case report, the authors reported no disclosures. Dr. Bruckner, corresponding author of the editorial, reported grants from several companies outside the submitted work.
Intravenous (JEB) caused by nonsense variants.
The newly generated structural protein persisted during the 3-month randomized clinical trial and was associated with significant wound closure – with no signs of ototoxic effects, nephrotoxic effects, or anti–laminin 332 autoantibody induction, investigators recently reported in JAMA Dermatology.
JEB is a rare, autosomal recessive disorder caused mainly by nonsense variants (i.e., mutations) in the LAMA3, LAMB3, or LAMC2 genes that encode laminin, resulting in widespread blisters and erosions of the skin. Current treatment is limited to supportive management and palliative care, and children with its severe subtype are likely to die within the first year of life.
“With data indicating a robust response to short-term gentamicin treatment and the marked stability of laminin 332, we envision that gentamicin could be delivered as a short-term pulse therapy every 2-3 months for patients with JEB caused by nonsense variants,” the researchers wrote.
Of the five patients, ages 3 months to 10 years, three received 7.5 mg/kg IV gentamicin daily for 14 days, and two received 10 mg/kg daily for 24 days at the University of Southern California, Los Angeles.
All had confirmed nonsense variants in LAMA3 or LAMB3 in one or two alleles, and all had minimal laminin 332 expression at baseline as determined by immunofluorescence. After treatment, each of the children had increased, sustained expression of laminin 332.
The researchers monitored three open wounds in each patient. By 1 month, seven of nine wounds in those receiving the lower-dose therapy and all of the wounds in those receiving the higher-dose therapy showed at least 50% closure. By 3 months, eight of nine wounds in the lower-dose group, and all wounds in the higher-dose group showed greater than 85% closure.
In an interview, senior investigators Mei Chen, PhD, professor of dermatology, and David T. Woodley, MD, professor and chair of dermatology, both at USC, emphasized laminin’s long half-life.“Once these skin structural proteins are generated at the dermal-epidermal junction, they are long-lasting structures, which means the therapy can be pulsed rather than continuously delivered, which can obviate some of the known side effects of the medication,” Dr. Woodley said.
Gentamicin, an aminoglycoside, works as a “read-through therapy,” inducing ribosomal read-through of premature termination codons (PTCs) caused by nonsense mutations. The read-through allows translation to proceed and full-length proteins to be generated.
Gentamicin read-through therapy is also being investigated for recessive dystrophic epidermolysis bullosa (RDEB) attributable to nonsense mutations. The culprit mutations in this form of EB occur in a gene that encodes collagen type VII alpha 1, which, like laminin, is responsible for dermal-epidermal adherence. A clinical trial of intravenous gentamicin for RDEB is ongoing at USC, Dr. Chen said.
EBS-MD case report
It may also have a role in treating epidermolysis bullosa simplex with muscular dystrophy (EBS-MD), according to investigators in Madrid. Their case report, published in JAMA Dermatology, details how two 14-day courses of infused gentamicin therapy were followed by re-expression of plectin in the skin for 4-5 months and mild improvement in symptoms in one patient, a woman in her 30s, with a homozygous nonsense variant in PLEC1.
In an editorial accompanying the two reports, Anna L. Bruckner, MD, MSCS, professor of dermatology, University of Colorado at Denver, Aurora, and colleagues expressed cautious optimism and said that additional research on the feasibility, possible cumulative toxic effects, risk of microbial resistance, and overall clinical relevance is needed.
Still, the “investigators should be applauded for taking advantage of a readily available systemic treatment to target cutaneous and extracutaneous symptoms of patients who have very limited treatment options at this time,” they wrote. While all forms of EB are considered orphan disorders, JEB and EBS-MD have received less research attention than RDEB.
The JEB study evaluated patients with clinical assessments/quality of life surveys and with a validated clinical score that considers skin and mucosae – the Epidermolysis Bullosa Disease Activity and Scarring Index (EBDASI). There were small positive changes in EBDASI scores, but data were incomplete and therefore difficult to interpret.
A “noteworthy” finding, the authors wrote, were improvements in emotions and functioning in two of the children who were eligible given their older ages for assessment with the Skindex-16 quality-of-life survey. The improvements suggest “potential psychosocial benefits” of the gentamicin therapy.
The JEB study was supported in part by grants from the EB Research Partnership and EB Medical Research Foundation and an award from the Congressionally Directed Medical Research Program. In addition to the grants, Dr. Woodley and Dr. Chen reported receiving personal fees from Phoenix Tissue Repair outside of the submitted work. For the EBS-MD case report, the authors reported no disclosures. Dr. Bruckner, corresponding author of the editorial, reported grants from several companies outside the submitted work.
FROM JAMA DERMATOLOGY
For pemphigus, rituximab is first line, expert says
BOSTON – . This drug is more rapidly effective, more likely to provide sustained remission, better tolerated, and lowers health care costs, according to an expert summary at the annual meeting of the American Academy of Dermatology.
With rituximab “we are not only able to offer better efficacy, earlier and longer remissions, less side effects, less risk of relapse after a response, but it is actually cheaper,” reported Erin X. Wei, MD, director of the Bullous Diseases Clinic at Brigham and Women’s Hospital, Boston.
There are many treatments that reduce the inflammatory component of pemphigus. Corticosteroids, doxycycline, mycophenolate mofetil, azathioprine, and methotrexate are among those options commonly considered in the early control of this rare and potentially fatal autoimmune blistering disease of the skin, mouth, and other tissues.
Not all of these options have been compared directly in controlled trials, but Dr. Wei indicated that the preponderance of evidence is now on the side of rituximab as a first-line choice. For example, in the multicenter Ritux 3 trial, which compared a tapered regimen of prednisone alone to rituximab combined with a shorter and lower-dose prednisone taper in patients with pemphigus, complete response rates off therapy at 2 years were 89% in the rituximab group versus 34% in the group that received prednisone alone.
“This was quite a remarkable difference,” said Dr. Wei, who noted that remissions overall occurred faster in the rituximab group and were more durable once achieved.
No other treatment option has demonstrated this degree of relative benefit over corticosteroids, according to Dr. Wei. She said there is evidence that mycophenolate mofetil acts more rapidly, but it has not been shown to be superior for sustained complete response. Nor has azathioprine provided a clear advantage over steroids. There are no well-conducted comparisons of methotrexate and prednisone, according to Dr. Wei, assistant professor at Harvard Medical School, Boston.
Corticosteroids, doxycycline, and immunomodulators have been characterized as mainstays of early treatment in pemphigus, but Dr. Wei argued that the evidence supports starting with the most effective therapy first. There are many advantages to suppressing disease activity “as soon as possible” after diagnosis.
Early control “is associated with a more sustained remission, lower overall steroid use, and better quality of life,” said Dr. Wei, listing the hazards of starting with less effective therapy, and explaining why she has moved to rituximab as a first-line choice. According to her, there are data to support these advantages.
“Several studies have observed that rituximab, within the first 6 months of disease onset, is associated with a higher rate of complete response and a longer duration of complete response,” Dr. Wei said.
Intravenous immunoglobulin (IVIG) therapy is effective in many patients but less reliable, and it has other disadvantages relative to rituximab as a first-line therapy.
“IVIG in pemphigus works quickly when it works, but it is more expensive and it is more of an ongoing therapy relative to rituximab,” said Dr. Wei, referring to the lower likelihood of IVIG to provide sustained remissions.
The price of rituximab is high relative to prednisone or other immunomodulators, but management costs are ultimately reduced because of better disease control, according to Dr. Wei. She cited a Canadian study published several years ago in which health care costs in the 6 months prior to rituximab were compared to costs over 6 months after it was initiated.
In this cohort of 89 patients with pemphigus or pemphigoid, the average cost per patient for 6 months of care prior to starting rituximab was $42,231 in Canadian dollars. After treatment was started, the cost fell to $29,423, a 30% reduction, over the next 6 months.
“It takes rituximab up to 3 months or sometimes even longer to achieve its greatest benefit, making these results even more impressive,” Dr. Wei said.
The activity of rituximab to suppress autoreactive B-cells can be monitored with antidesmoglein autoantibody levels and by measuring CD20-positive cell percentages. Unlike severity of disease at baseline, which Dr. Wei said is not a reliable predictor of relapse risk, these can guide steroid tapering.
“If the patient is not making new autoantibodies, then tapering steroids can be considered safe,” Dr. Wei said.
One small case series cited by Dr. Wei has suggested that rituximab might be effectively employed as a maintenance therapy for pemphigus. The maintenance treatment, which initially consisted of 1 g of rituximab every 6 months, was evaluated in 11 patients with a history of severe and frequent relapses.
In this group, rituximab was first employed to achieve a complete response. The maintenance was initiated when patients were in remission. In some patients, the maintenance dose interval was extended to once every 12 months over time. During a mean follow-up of 4 years, all 11 patients remained in complete remission.
“This was a remarkable result,” said Dr. Wei, who noted that there were no serious adverse events associated with rituximab maintenance over this period. This cannot be considered a routine strategy without a large patient experience, according to Dr. Wei, but it does provide another piece of evidence that rituximab is effective and well tolerated.
There are no guidelines from a major organization that establish an evidence-based treatment algorithm for pemphigus, but Dr. Wei is not alone in considering early initiation of the most effective therapy as the best approach to sustained control.
“I agree that rituximab is a good first-line option for pemphigus patients,” said Kara Heelan, MBBCh, MD, a consultant dermatologist at the Royal Marsden and Lister Hospital, London. She was the first author of the cost-effectiveness study that Dr. Wei cited. The study was published when she was an associate in the division of dermatology at the University of Toronto.
By calling rituximab “a good” option rather than a potential standard, Dr. Heelan appeared to be more circumspect than Dr. Wei about its central role in the care of pemphigus, but she did agree in an interview that this agent “has been shown to be cost-effective.” In her study, this was an advantage attributed to relative efficacy and safety that reduced use of health care resources.
A version of this article first appeared on Medscape.com.
BOSTON – . This drug is more rapidly effective, more likely to provide sustained remission, better tolerated, and lowers health care costs, according to an expert summary at the annual meeting of the American Academy of Dermatology.
With rituximab “we are not only able to offer better efficacy, earlier and longer remissions, less side effects, less risk of relapse after a response, but it is actually cheaper,” reported Erin X. Wei, MD, director of the Bullous Diseases Clinic at Brigham and Women’s Hospital, Boston.
There are many treatments that reduce the inflammatory component of pemphigus. Corticosteroids, doxycycline, mycophenolate mofetil, azathioprine, and methotrexate are among those options commonly considered in the early control of this rare and potentially fatal autoimmune blistering disease of the skin, mouth, and other tissues.
Not all of these options have been compared directly in controlled trials, but Dr. Wei indicated that the preponderance of evidence is now on the side of rituximab as a first-line choice. For example, in the multicenter Ritux 3 trial, which compared a tapered regimen of prednisone alone to rituximab combined with a shorter and lower-dose prednisone taper in patients with pemphigus, complete response rates off therapy at 2 years were 89% in the rituximab group versus 34% in the group that received prednisone alone.
“This was quite a remarkable difference,” said Dr. Wei, who noted that remissions overall occurred faster in the rituximab group and were more durable once achieved.
No other treatment option has demonstrated this degree of relative benefit over corticosteroids, according to Dr. Wei. She said there is evidence that mycophenolate mofetil acts more rapidly, but it has not been shown to be superior for sustained complete response. Nor has azathioprine provided a clear advantage over steroids. There are no well-conducted comparisons of methotrexate and prednisone, according to Dr. Wei, assistant professor at Harvard Medical School, Boston.
Corticosteroids, doxycycline, and immunomodulators have been characterized as mainstays of early treatment in pemphigus, but Dr. Wei argued that the evidence supports starting with the most effective therapy first. There are many advantages to suppressing disease activity “as soon as possible” after diagnosis.
Early control “is associated with a more sustained remission, lower overall steroid use, and better quality of life,” said Dr. Wei, listing the hazards of starting with less effective therapy, and explaining why she has moved to rituximab as a first-line choice. According to her, there are data to support these advantages.
“Several studies have observed that rituximab, within the first 6 months of disease onset, is associated with a higher rate of complete response and a longer duration of complete response,” Dr. Wei said.
Intravenous immunoglobulin (IVIG) therapy is effective in many patients but less reliable, and it has other disadvantages relative to rituximab as a first-line therapy.
“IVIG in pemphigus works quickly when it works, but it is more expensive and it is more of an ongoing therapy relative to rituximab,” said Dr. Wei, referring to the lower likelihood of IVIG to provide sustained remissions.
The price of rituximab is high relative to prednisone or other immunomodulators, but management costs are ultimately reduced because of better disease control, according to Dr. Wei. She cited a Canadian study published several years ago in which health care costs in the 6 months prior to rituximab were compared to costs over 6 months after it was initiated.
In this cohort of 89 patients with pemphigus or pemphigoid, the average cost per patient for 6 months of care prior to starting rituximab was $42,231 in Canadian dollars. After treatment was started, the cost fell to $29,423, a 30% reduction, over the next 6 months.
“It takes rituximab up to 3 months or sometimes even longer to achieve its greatest benefit, making these results even more impressive,” Dr. Wei said.
The activity of rituximab to suppress autoreactive B-cells can be monitored with antidesmoglein autoantibody levels and by measuring CD20-positive cell percentages. Unlike severity of disease at baseline, which Dr. Wei said is not a reliable predictor of relapse risk, these can guide steroid tapering.
“If the patient is not making new autoantibodies, then tapering steroids can be considered safe,” Dr. Wei said.
One small case series cited by Dr. Wei has suggested that rituximab might be effectively employed as a maintenance therapy for pemphigus. The maintenance treatment, which initially consisted of 1 g of rituximab every 6 months, was evaluated in 11 patients with a history of severe and frequent relapses.
In this group, rituximab was first employed to achieve a complete response. The maintenance was initiated when patients were in remission. In some patients, the maintenance dose interval was extended to once every 12 months over time. During a mean follow-up of 4 years, all 11 patients remained in complete remission.
“This was a remarkable result,” said Dr. Wei, who noted that there were no serious adverse events associated with rituximab maintenance over this period. This cannot be considered a routine strategy without a large patient experience, according to Dr. Wei, but it does provide another piece of evidence that rituximab is effective and well tolerated.
There are no guidelines from a major organization that establish an evidence-based treatment algorithm for pemphigus, but Dr. Wei is not alone in considering early initiation of the most effective therapy as the best approach to sustained control.
“I agree that rituximab is a good first-line option for pemphigus patients,” said Kara Heelan, MBBCh, MD, a consultant dermatologist at the Royal Marsden and Lister Hospital, London. She was the first author of the cost-effectiveness study that Dr. Wei cited. The study was published when she was an associate in the division of dermatology at the University of Toronto.
By calling rituximab “a good” option rather than a potential standard, Dr. Heelan appeared to be more circumspect than Dr. Wei about its central role in the care of pemphigus, but she did agree in an interview that this agent “has been shown to be cost-effective.” In her study, this was an advantage attributed to relative efficacy and safety that reduced use of health care resources.
A version of this article first appeared on Medscape.com.
BOSTON – . This drug is more rapidly effective, more likely to provide sustained remission, better tolerated, and lowers health care costs, according to an expert summary at the annual meeting of the American Academy of Dermatology.
With rituximab “we are not only able to offer better efficacy, earlier and longer remissions, less side effects, less risk of relapse after a response, but it is actually cheaper,” reported Erin X. Wei, MD, director of the Bullous Diseases Clinic at Brigham and Women’s Hospital, Boston.
There are many treatments that reduce the inflammatory component of pemphigus. Corticosteroids, doxycycline, mycophenolate mofetil, azathioprine, and methotrexate are among those options commonly considered in the early control of this rare and potentially fatal autoimmune blistering disease of the skin, mouth, and other tissues.
Not all of these options have been compared directly in controlled trials, but Dr. Wei indicated that the preponderance of evidence is now on the side of rituximab as a first-line choice. For example, in the multicenter Ritux 3 trial, which compared a tapered regimen of prednisone alone to rituximab combined with a shorter and lower-dose prednisone taper in patients with pemphigus, complete response rates off therapy at 2 years were 89% in the rituximab group versus 34% in the group that received prednisone alone.
“This was quite a remarkable difference,” said Dr. Wei, who noted that remissions overall occurred faster in the rituximab group and were more durable once achieved.
No other treatment option has demonstrated this degree of relative benefit over corticosteroids, according to Dr. Wei. She said there is evidence that mycophenolate mofetil acts more rapidly, but it has not been shown to be superior for sustained complete response. Nor has azathioprine provided a clear advantage over steroids. There are no well-conducted comparisons of methotrexate and prednisone, according to Dr. Wei, assistant professor at Harvard Medical School, Boston.
Corticosteroids, doxycycline, and immunomodulators have been characterized as mainstays of early treatment in pemphigus, but Dr. Wei argued that the evidence supports starting with the most effective therapy first. There are many advantages to suppressing disease activity “as soon as possible” after diagnosis.
Early control “is associated with a more sustained remission, lower overall steroid use, and better quality of life,” said Dr. Wei, listing the hazards of starting with less effective therapy, and explaining why she has moved to rituximab as a first-line choice. According to her, there are data to support these advantages.
“Several studies have observed that rituximab, within the first 6 months of disease onset, is associated with a higher rate of complete response and a longer duration of complete response,” Dr. Wei said.
Intravenous immunoglobulin (IVIG) therapy is effective in many patients but less reliable, and it has other disadvantages relative to rituximab as a first-line therapy.
“IVIG in pemphigus works quickly when it works, but it is more expensive and it is more of an ongoing therapy relative to rituximab,” said Dr. Wei, referring to the lower likelihood of IVIG to provide sustained remissions.
The price of rituximab is high relative to prednisone or other immunomodulators, but management costs are ultimately reduced because of better disease control, according to Dr. Wei. She cited a Canadian study published several years ago in which health care costs in the 6 months prior to rituximab were compared to costs over 6 months after it was initiated.
In this cohort of 89 patients with pemphigus or pemphigoid, the average cost per patient for 6 months of care prior to starting rituximab was $42,231 in Canadian dollars. After treatment was started, the cost fell to $29,423, a 30% reduction, over the next 6 months.
“It takes rituximab up to 3 months or sometimes even longer to achieve its greatest benefit, making these results even more impressive,” Dr. Wei said.
The activity of rituximab to suppress autoreactive B-cells can be monitored with antidesmoglein autoantibody levels and by measuring CD20-positive cell percentages. Unlike severity of disease at baseline, which Dr. Wei said is not a reliable predictor of relapse risk, these can guide steroid tapering.
“If the patient is not making new autoantibodies, then tapering steroids can be considered safe,” Dr. Wei said.
One small case series cited by Dr. Wei has suggested that rituximab might be effectively employed as a maintenance therapy for pemphigus. The maintenance treatment, which initially consisted of 1 g of rituximab every 6 months, was evaluated in 11 patients with a history of severe and frequent relapses.
In this group, rituximab was first employed to achieve a complete response. The maintenance was initiated when patients were in remission. In some patients, the maintenance dose interval was extended to once every 12 months over time. During a mean follow-up of 4 years, all 11 patients remained in complete remission.
“This was a remarkable result,” said Dr. Wei, who noted that there were no serious adverse events associated with rituximab maintenance over this period. This cannot be considered a routine strategy without a large patient experience, according to Dr. Wei, but it does provide another piece of evidence that rituximab is effective and well tolerated.
There are no guidelines from a major organization that establish an evidence-based treatment algorithm for pemphigus, but Dr. Wei is not alone in considering early initiation of the most effective therapy as the best approach to sustained control.
“I agree that rituximab is a good first-line option for pemphigus patients,” said Kara Heelan, MBBCh, MD, a consultant dermatologist at the Royal Marsden and Lister Hospital, London. She was the first author of the cost-effectiveness study that Dr. Wei cited. The study was published when she was an associate in the division of dermatology at the University of Toronto.
By calling rituximab “a good” option rather than a potential standard, Dr. Heelan appeared to be more circumspect than Dr. Wei about its central role in the care of pemphigus, but she did agree in an interview that this agent “has been shown to be cost-effective.” In her study, this was an advantage attributed to relative efficacy and safety that reduced use of health care resources.
A version of this article first appeared on Medscape.com.
AT AAD 2022
Dupilumab treats itch and clears lesions in prurigo nodularis patients
BOSTON – of treatment, in a phase 3 trial presented at the American Academy of Dermatology 2022 Annual Meeting.
There are currently no Food and Drug Administration–approved systemic therapies for PN. Although several treatments for the disease are used off label for the condition, such as ultraviolet light therapy and immunosuppressive agents, moderate to severe PN is usually difficult to control, noted Gil Yosipovitch, MD, director of the Miami Itch Center at the University of Miami Miller School of Medicine, Florida. He led the research and presented the findings at the conference.
“Many dermatologists feel very uncomfortable dealing with these patients because they suffer from chronicity, they are miserable, and previously, the drugs didn’t work well,” Dr. Yosipovitch told this news organization. The results from this trial “are very promising,” he said. “It opens a new field of treatment for itchy conditions.”
The trial, named LIBERTY-PN PRIME2, enrolled patients aged 18-80 who had been living with PN for at least 3 months. Patients had at least 20 lesions at baseline as well as severe itch, defined as a score of 7 or greater on the Worst Itch Numerical Rating Scale (WI-NRS). The scale ranges from 0 (no itch) to 10 (worst itch imaginable). Participants also had a history of treatment failure with medium to super-potent topical corticosteroids (TCSs), or treatment with TCSs was not medically advisable for them.
The randomized, double-blinded study enrolled 160 adults with PN. Of those, 78 were assigned to the treatment arm and received a 600-mg loading dose of dupilumab, administered subcutaneously, followed by 300-mg doses every 2 weeks for 24 weeks; 82 patients were allocated to receive placebo.
During the study, 25 patients in the placebo arm discontinued treatment. In the treatment arm, one patient was not treated and two discontinued treatment due to lack of efficacy.
The primary endpoint of the study was a reduction of at least 4 points on the WI-NRS at 12 weeks. Secondary endpoints included at least a 4-point WI-NRS reduction at 24 weeks and clear to nearly clear skin, defined as having a score of 0 or 1 on the Investigator’s Global Assessment PN-Stage (IGN PN-S). The scale ranges from 0 (clear) to 4 (severe).
At 12 weeks, 37.2% of patients given dupilumab reported a reduction of at least 4 points in WI-NRS, compared with 22.0% of patients given placebo (P = .0216). By 24 weeks, 57.7% of adults who received dupilumab achieved a greater than or equal to 4-point reduction in WI-NRS, compared with 19.5% of those who received placebo (P < .0001). Additionally, 44.9% of participants in the treatment arm achieved a score of 0 or 1 on the IGA PN-S, compared with 15.9% of those in the placebo arm (P < .0001).
Forty-four participants who received dupilumab (57.1%) and 42 participants who received placebo (51.2%) reported at least one treatment-emergent adverse event (TEAE) during the study, though none of these events were serious. The most common TEAE in the study was headache, occurring in five patients taking placebo and four patients receiving dupilumab. In the dupilumab group, there were five cases of herpes virus infection, four non-herpes skin infections, and three cases of conjunctivitis. In the placebo group, seven non-herpes skin infections were reported.
Sanofi and Regeneron, who jointly developed dupilumab, plan to file for regulatory approval for dupilumab for PN “around the world” in the first half of this year, according to a press release.
“It’s great news and a step in the right direction,” Sarina Elmariah, MD, PhD, a dermatologist at Massachusetts General Hospital and instructor of dermatology at Harvard Medical School, both in Boston, told this news organization. She was not involved with the research.
“We’re finally starting to shed light on this condition and its pathogenesis,” she said. She noted that other potential therapeutics for PN are also in development. “It’s reflective of the fact that we are making strides in this area.”
Sanofi and Regeneron Pharmaceuticals sponsored the LIBERTY-PN PRIME2 trial. Dr. Yosipovitch has reported financial relationships with Bellus Health, Eli Lilly, Galderma, GSK, Kiniksa Pharmaceuticals, LEO Pharma, Novartis, Pfizer, Regeneron, Sanofi, and Trevi Therapeutics. Dr. Elmariah is on the advisory boards of Sanofi, Galderma, and Trevi Therapeutics.
A version of this article first appeared on Medscape.com.
BOSTON – of treatment, in a phase 3 trial presented at the American Academy of Dermatology 2022 Annual Meeting.
There are currently no Food and Drug Administration–approved systemic therapies for PN. Although several treatments for the disease are used off label for the condition, such as ultraviolet light therapy and immunosuppressive agents, moderate to severe PN is usually difficult to control, noted Gil Yosipovitch, MD, director of the Miami Itch Center at the University of Miami Miller School of Medicine, Florida. He led the research and presented the findings at the conference.
“Many dermatologists feel very uncomfortable dealing with these patients because they suffer from chronicity, they are miserable, and previously, the drugs didn’t work well,” Dr. Yosipovitch told this news organization. The results from this trial “are very promising,” he said. “It opens a new field of treatment for itchy conditions.”
The trial, named LIBERTY-PN PRIME2, enrolled patients aged 18-80 who had been living with PN for at least 3 months. Patients had at least 20 lesions at baseline as well as severe itch, defined as a score of 7 or greater on the Worst Itch Numerical Rating Scale (WI-NRS). The scale ranges from 0 (no itch) to 10 (worst itch imaginable). Participants also had a history of treatment failure with medium to super-potent topical corticosteroids (TCSs), or treatment with TCSs was not medically advisable for them.
The randomized, double-blinded study enrolled 160 adults with PN. Of those, 78 were assigned to the treatment arm and received a 600-mg loading dose of dupilumab, administered subcutaneously, followed by 300-mg doses every 2 weeks for 24 weeks; 82 patients were allocated to receive placebo.
During the study, 25 patients in the placebo arm discontinued treatment. In the treatment arm, one patient was not treated and two discontinued treatment due to lack of efficacy.
The primary endpoint of the study was a reduction of at least 4 points on the WI-NRS at 12 weeks. Secondary endpoints included at least a 4-point WI-NRS reduction at 24 weeks and clear to nearly clear skin, defined as having a score of 0 or 1 on the Investigator’s Global Assessment PN-Stage (IGN PN-S). The scale ranges from 0 (clear) to 4 (severe).
At 12 weeks, 37.2% of patients given dupilumab reported a reduction of at least 4 points in WI-NRS, compared with 22.0% of patients given placebo (P = .0216). By 24 weeks, 57.7% of adults who received dupilumab achieved a greater than or equal to 4-point reduction in WI-NRS, compared with 19.5% of those who received placebo (P < .0001). Additionally, 44.9% of participants in the treatment arm achieved a score of 0 or 1 on the IGA PN-S, compared with 15.9% of those in the placebo arm (P < .0001).
Forty-four participants who received dupilumab (57.1%) and 42 participants who received placebo (51.2%) reported at least one treatment-emergent adverse event (TEAE) during the study, though none of these events were serious. The most common TEAE in the study was headache, occurring in five patients taking placebo and four patients receiving dupilumab. In the dupilumab group, there were five cases of herpes virus infection, four non-herpes skin infections, and three cases of conjunctivitis. In the placebo group, seven non-herpes skin infections were reported.
Sanofi and Regeneron, who jointly developed dupilumab, plan to file for regulatory approval for dupilumab for PN “around the world” in the first half of this year, according to a press release.
“It’s great news and a step in the right direction,” Sarina Elmariah, MD, PhD, a dermatologist at Massachusetts General Hospital and instructor of dermatology at Harvard Medical School, both in Boston, told this news organization. She was not involved with the research.
“We’re finally starting to shed light on this condition and its pathogenesis,” she said. She noted that other potential therapeutics for PN are also in development. “It’s reflective of the fact that we are making strides in this area.”
Sanofi and Regeneron Pharmaceuticals sponsored the LIBERTY-PN PRIME2 trial. Dr. Yosipovitch has reported financial relationships with Bellus Health, Eli Lilly, Galderma, GSK, Kiniksa Pharmaceuticals, LEO Pharma, Novartis, Pfizer, Regeneron, Sanofi, and Trevi Therapeutics. Dr. Elmariah is on the advisory boards of Sanofi, Galderma, and Trevi Therapeutics.
A version of this article first appeared on Medscape.com.
BOSTON – of treatment, in a phase 3 trial presented at the American Academy of Dermatology 2022 Annual Meeting.
There are currently no Food and Drug Administration–approved systemic therapies for PN. Although several treatments for the disease are used off label for the condition, such as ultraviolet light therapy and immunosuppressive agents, moderate to severe PN is usually difficult to control, noted Gil Yosipovitch, MD, director of the Miami Itch Center at the University of Miami Miller School of Medicine, Florida. He led the research and presented the findings at the conference.
“Many dermatologists feel very uncomfortable dealing with these patients because they suffer from chronicity, they are miserable, and previously, the drugs didn’t work well,” Dr. Yosipovitch told this news organization. The results from this trial “are very promising,” he said. “It opens a new field of treatment for itchy conditions.”
The trial, named LIBERTY-PN PRIME2, enrolled patients aged 18-80 who had been living with PN for at least 3 months. Patients had at least 20 lesions at baseline as well as severe itch, defined as a score of 7 or greater on the Worst Itch Numerical Rating Scale (WI-NRS). The scale ranges from 0 (no itch) to 10 (worst itch imaginable). Participants also had a history of treatment failure with medium to super-potent topical corticosteroids (TCSs), or treatment with TCSs was not medically advisable for them.
The randomized, double-blinded study enrolled 160 adults with PN. Of those, 78 were assigned to the treatment arm and received a 600-mg loading dose of dupilumab, administered subcutaneously, followed by 300-mg doses every 2 weeks for 24 weeks; 82 patients were allocated to receive placebo.
During the study, 25 patients in the placebo arm discontinued treatment. In the treatment arm, one patient was not treated and two discontinued treatment due to lack of efficacy.
The primary endpoint of the study was a reduction of at least 4 points on the WI-NRS at 12 weeks. Secondary endpoints included at least a 4-point WI-NRS reduction at 24 weeks and clear to nearly clear skin, defined as having a score of 0 or 1 on the Investigator’s Global Assessment PN-Stage (IGN PN-S). The scale ranges from 0 (clear) to 4 (severe).
At 12 weeks, 37.2% of patients given dupilumab reported a reduction of at least 4 points in WI-NRS, compared with 22.0% of patients given placebo (P = .0216). By 24 weeks, 57.7% of adults who received dupilumab achieved a greater than or equal to 4-point reduction in WI-NRS, compared with 19.5% of those who received placebo (P < .0001). Additionally, 44.9% of participants in the treatment arm achieved a score of 0 or 1 on the IGA PN-S, compared with 15.9% of those in the placebo arm (P < .0001).
Forty-four participants who received dupilumab (57.1%) and 42 participants who received placebo (51.2%) reported at least one treatment-emergent adverse event (TEAE) during the study, though none of these events were serious. The most common TEAE in the study was headache, occurring in five patients taking placebo and four patients receiving dupilumab. In the dupilumab group, there were five cases of herpes virus infection, four non-herpes skin infections, and three cases of conjunctivitis. In the placebo group, seven non-herpes skin infections were reported.
Sanofi and Regeneron, who jointly developed dupilumab, plan to file for regulatory approval for dupilumab for PN “around the world” in the first half of this year, according to a press release.
“It’s great news and a step in the right direction,” Sarina Elmariah, MD, PhD, a dermatologist at Massachusetts General Hospital and instructor of dermatology at Harvard Medical School, both in Boston, told this news organization. She was not involved with the research.
“We’re finally starting to shed light on this condition and its pathogenesis,” she said. She noted that other potential therapeutics for PN are also in development. “It’s reflective of the fact that we are making strides in this area.”
Sanofi and Regeneron Pharmaceuticals sponsored the LIBERTY-PN PRIME2 trial. Dr. Yosipovitch has reported financial relationships with Bellus Health, Eli Lilly, Galderma, GSK, Kiniksa Pharmaceuticals, LEO Pharma, Novartis, Pfizer, Regeneron, Sanofi, and Trevi Therapeutics. Dr. Elmariah is on the advisory boards of Sanofi, Galderma, and Trevi Therapeutics.
A version of this article first appeared on Medscape.com.
AT AAD 2022
Skin reactions to first COVID-19 vaccine don’t justify forgoing second dose
BOSTON – Requests for a according to an analysis of several large sets of data presented at the annual meeting of the American Academy of Dermatology.
According to the data, “there are no serious adverse consequences from these cutaneous reactions,” said Esther Freeman, MD, PhD, director of Global Health Dermatology, Massachusetts General Hospital, Boston.
This is important because the risk of vaccine hesitancy goes up dramatically in patients who experience reactions to the first vaccine dose, according to follow-up of more than 50,000 employees vaccinated in the Mass General Brigham Healthcare System (MGBHS). According to Dr. Freeman, there was almost a fourfold increase in the rate of second-dose refusals for those with cutaneous reactions and a more than fourfold increase in those who developed angioedema.
Before the data were available, skin reactions were a source of concern among dermatologists and others involved in monitoring vaccine-related adverse events. Injection site reactions (ISRs) are associated with essentially every injectable vaccine, so these were expected, but a small proportion of patients developed large red plaques in the injection arm 7-8 days after the inoculation.
“These delayed reactions caused a lot of initial panic,” said Dr. Freeman, who counted herself among those alarmed about what the reactions might signify. “Was this cellulitis? Would the next dose cause anaphylaxis? We were concerned.”
This concern dissipated with the availability of more data. In a global registry that has so far captured more than 1,000 cutaneous reactions from 52 participating countries, it appears that about 2% of patients have a cutaneous reaction other than an ISR after the first dose. All resolve with minimal skin care or no treatment.
After the second dose, the proportion is lower. If there is a reaction, it typically occurs earlier and resolves more quickly.
“What we have learned is that fewer than half of patients who had a reaction to the first dose have a reaction to the second, and those who did have a reaction had a milder course,” said Dr. Freeman.
These data are “incredibly reassuring” on many levels, she explained. In addition, it allows clinicians to confidently explain to patients that there are no serious sequelae from the rashes, whether immediate or delayed, from the available COVID-19 vaccines.
“Every skin reaction I have seen is something we can treat through,” she added, noting that most reactions resolve with little or no supportive care. Following skin reactions, particularly the delayed lesions, it is not uncommon for patients to refuse a second shot. Some request a medical waiver to avoid further vaccine exposure. According to Dr. Freeman, this is unwarranted.
“I have granted exactly zero waivers,” she said. She explains to patients that these reactions have not been predictive of serious events, such as anaphylaxis. Although the trigger of the hypersensitivity reaction remains unknown, there is no evidence of serious consequences.
Delayed skin reactions are more commonly associated with the Moderna than the Pfizer vaccine. One notable difference between these vaccines is the greater content of mRNA in the Moderna formulation, but Freeman said that this is only one potential hypothesis for higher frequency of reactions to this version of the vaccine.
Patients with a history of allergic disease are more likely to develop a reaction but not significantly more likely to have a reaction that is more difficult to manage, according to Kimberly G. Blumenthal, MD, quality and safety officer for allergy, and codirector of the clinical epidemiology program in the division of rheumatology, allergy, and immunology at Mass General.
Anaphylaxis has been associated with COVD-19 vaccines just as it has with essentially every injectable vaccine, Dr. Blumenthal said during the same session. But the risk is very low, and it stays low even among those with a history of severe hypersensitivity reactions in the past.
Among the data collected from more than 52,000 vaccinated MGBHS employees, 0.9% had a history of severe allergic reaction to a prior vaccine. Of these, 11.6% had an allergic reaction to the COVID-19 vaccine. This was more than twice the 4.6% rate of allergic reactions among employees without a history of allergic reactions, but serious consequences were rare in both groups.
Of those with a reaction to the first dose, all but 2.4% took a subsequent dose. Again, serious reactions were exceedingly rare. These serious reactions did include anaphylaxis and hospitalization in 3% of patients, but there were no fatalities and all resolved.
The absence of serious sequelae from a reaction to a COVID-19 vaccine must be considered within the context of the benefit, which includes protection from death and hospitalization from the virus, according to Dr. Blumenthal. Citing the evidence that first-shot reactions are a source of vaccine hesitancy, she agreed that it is important to educate patients about relative risks.
“Even in our own cohort of MGBHS employees, we have people, including those who had been provaccine in the past, become hesitant,” commented Dr. Blumenthal, who said there are data from the Kaiser Permanente System showing similar vaccine reluctance following a first-shot reaction.
After more than 500 million doses of the Moderna and Pfizer vaccines had been administered worldwide, there was not a single reported death from anaphylaxis. Although Dr. Blumenthal said that an unconfirmed death of this type had been recently reported, she emphasized that this single death, if valid, is dwarfed by the lives saved with vaccination.
Asked about her strategy for counseling patients with vaccine hesitancy, Dr. Freeman said the body of safety data is large and compelling. There is overwhelming evidence of a favorable benefit-to-risk ratio overall and among those with a first-shot reaction.
“I can reassure them on the basis of the data,” Dr. Freeman said in an interview. “Less than half will have a reaction to the second shot and even if they do have a reaction, it is likely to be less severe.”
Although the main message is that vaccination is potentially lifesaving and far outweighs any risks, Freeman specifically gives this message to those hesitant to take a second shot after a first-shot reaction: “I can get you through it.”
Dr. Freeman encouraged health care professionals to report cases of COVID-19 vaccine–related dermatologic side effects to the American Academy of Dermatology / International League of Dermatologic Societies COVID-19 dermatology registry. Dermatologic manifestations of COVID-19 can also be reported to the registry.
Dr. Freeman disclosed receiving grants/research funding from the International League of Dermatologic Societies and from the National Institutes of Health. Dr. Blumenthal disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
BOSTON – Requests for a according to an analysis of several large sets of data presented at the annual meeting of the American Academy of Dermatology.
According to the data, “there are no serious adverse consequences from these cutaneous reactions,” said Esther Freeman, MD, PhD, director of Global Health Dermatology, Massachusetts General Hospital, Boston.
This is important because the risk of vaccine hesitancy goes up dramatically in patients who experience reactions to the first vaccine dose, according to follow-up of more than 50,000 employees vaccinated in the Mass General Brigham Healthcare System (MGBHS). According to Dr. Freeman, there was almost a fourfold increase in the rate of second-dose refusals for those with cutaneous reactions and a more than fourfold increase in those who developed angioedema.
Before the data were available, skin reactions were a source of concern among dermatologists and others involved in monitoring vaccine-related adverse events. Injection site reactions (ISRs) are associated with essentially every injectable vaccine, so these were expected, but a small proportion of patients developed large red plaques in the injection arm 7-8 days after the inoculation.
“These delayed reactions caused a lot of initial panic,” said Dr. Freeman, who counted herself among those alarmed about what the reactions might signify. “Was this cellulitis? Would the next dose cause anaphylaxis? We were concerned.”
This concern dissipated with the availability of more data. In a global registry that has so far captured more than 1,000 cutaneous reactions from 52 participating countries, it appears that about 2% of patients have a cutaneous reaction other than an ISR after the first dose. All resolve with minimal skin care or no treatment.
After the second dose, the proportion is lower. If there is a reaction, it typically occurs earlier and resolves more quickly.
“What we have learned is that fewer than half of patients who had a reaction to the first dose have a reaction to the second, and those who did have a reaction had a milder course,” said Dr. Freeman.
These data are “incredibly reassuring” on many levels, she explained. In addition, it allows clinicians to confidently explain to patients that there are no serious sequelae from the rashes, whether immediate or delayed, from the available COVID-19 vaccines.
“Every skin reaction I have seen is something we can treat through,” she added, noting that most reactions resolve with little or no supportive care. Following skin reactions, particularly the delayed lesions, it is not uncommon for patients to refuse a second shot. Some request a medical waiver to avoid further vaccine exposure. According to Dr. Freeman, this is unwarranted.
“I have granted exactly zero waivers,” she said. She explains to patients that these reactions have not been predictive of serious events, such as anaphylaxis. Although the trigger of the hypersensitivity reaction remains unknown, there is no evidence of serious consequences.
Delayed skin reactions are more commonly associated with the Moderna than the Pfizer vaccine. One notable difference between these vaccines is the greater content of mRNA in the Moderna formulation, but Freeman said that this is only one potential hypothesis for higher frequency of reactions to this version of the vaccine.
Patients with a history of allergic disease are more likely to develop a reaction but not significantly more likely to have a reaction that is more difficult to manage, according to Kimberly G. Blumenthal, MD, quality and safety officer for allergy, and codirector of the clinical epidemiology program in the division of rheumatology, allergy, and immunology at Mass General.
Anaphylaxis has been associated with COVD-19 vaccines just as it has with essentially every injectable vaccine, Dr. Blumenthal said during the same session. But the risk is very low, and it stays low even among those with a history of severe hypersensitivity reactions in the past.
Among the data collected from more than 52,000 vaccinated MGBHS employees, 0.9% had a history of severe allergic reaction to a prior vaccine. Of these, 11.6% had an allergic reaction to the COVID-19 vaccine. This was more than twice the 4.6% rate of allergic reactions among employees without a history of allergic reactions, but serious consequences were rare in both groups.
Of those with a reaction to the first dose, all but 2.4% took a subsequent dose. Again, serious reactions were exceedingly rare. These serious reactions did include anaphylaxis and hospitalization in 3% of patients, but there were no fatalities and all resolved.
The absence of serious sequelae from a reaction to a COVID-19 vaccine must be considered within the context of the benefit, which includes protection from death and hospitalization from the virus, according to Dr. Blumenthal. Citing the evidence that first-shot reactions are a source of vaccine hesitancy, she agreed that it is important to educate patients about relative risks.
“Even in our own cohort of MGBHS employees, we have people, including those who had been provaccine in the past, become hesitant,” commented Dr. Blumenthal, who said there are data from the Kaiser Permanente System showing similar vaccine reluctance following a first-shot reaction.
After more than 500 million doses of the Moderna and Pfizer vaccines had been administered worldwide, there was not a single reported death from anaphylaxis. Although Dr. Blumenthal said that an unconfirmed death of this type had been recently reported, she emphasized that this single death, if valid, is dwarfed by the lives saved with vaccination.
Asked about her strategy for counseling patients with vaccine hesitancy, Dr. Freeman said the body of safety data is large and compelling. There is overwhelming evidence of a favorable benefit-to-risk ratio overall and among those with a first-shot reaction.
“I can reassure them on the basis of the data,” Dr. Freeman said in an interview. “Less than half will have a reaction to the second shot and even if they do have a reaction, it is likely to be less severe.”
Although the main message is that vaccination is potentially lifesaving and far outweighs any risks, Freeman specifically gives this message to those hesitant to take a second shot after a first-shot reaction: “I can get you through it.”
Dr. Freeman encouraged health care professionals to report cases of COVID-19 vaccine–related dermatologic side effects to the American Academy of Dermatology / International League of Dermatologic Societies COVID-19 dermatology registry. Dermatologic manifestations of COVID-19 can also be reported to the registry.
Dr. Freeman disclosed receiving grants/research funding from the International League of Dermatologic Societies and from the National Institutes of Health. Dr. Blumenthal disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
BOSTON – Requests for a according to an analysis of several large sets of data presented at the annual meeting of the American Academy of Dermatology.
According to the data, “there are no serious adverse consequences from these cutaneous reactions,” said Esther Freeman, MD, PhD, director of Global Health Dermatology, Massachusetts General Hospital, Boston.
This is important because the risk of vaccine hesitancy goes up dramatically in patients who experience reactions to the first vaccine dose, according to follow-up of more than 50,000 employees vaccinated in the Mass General Brigham Healthcare System (MGBHS). According to Dr. Freeman, there was almost a fourfold increase in the rate of second-dose refusals for those with cutaneous reactions and a more than fourfold increase in those who developed angioedema.
Before the data were available, skin reactions were a source of concern among dermatologists and others involved in monitoring vaccine-related adverse events. Injection site reactions (ISRs) are associated with essentially every injectable vaccine, so these were expected, but a small proportion of patients developed large red plaques in the injection arm 7-8 days after the inoculation.
“These delayed reactions caused a lot of initial panic,” said Dr. Freeman, who counted herself among those alarmed about what the reactions might signify. “Was this cellulitis? Would the next dose cause anaphylaxis? We were concerned.”
This concern dissipated with the availability of more data. In a global registry that has so far captured more than 1,000 cutaneous reactions from 52 participating countries, it appears that about 2% of patients have a cutaneous reaction other than an ISR after the first dose. All resolve with minimal skin care or no treatment.
After the second dose, the proportion is lower. If there is a reaction, it typically occurs earlier and resolves more quickly.
“What we have learned is that fewer than half of patients who had a reaction to the first dose have a reaction to the second, and those who did have a reaction had a milder course,” said Dr. Freeman.
These data are “incredibly reassuring” on many levels, she explained. In addition, it allows clinicians to confidently explain to patients that there are no serious sequelae from the rashes, whether immediate or delayed, from the available COVID-19 vaccines.
“Every skin reaction I have seen is something we can treat through,” she added, noting that most reactions resolve with little or no supportive care. Following skin reactions, particularly the delayed lesions, it is not uncommon for patients to refuse a second shot. Some request a medical waiver to avoid further vaccine exposure. According to Dr. Freeman, this is unwarranted.
“I have granted exactly zero waivers,” she said. She explains to patients that these reactions have not been predictive of serious events, such as anaphylaxis. Although the trigger of the hypersensitivity reaction remains unknown, there is no evidence of serious consequences.
Delayed skin reactions are more commonly associated with the Moderna than the Pfizer vaccine. One notable difference between these vaccines is the greater content of mRNA in the Moderna formulation, but Freeman said that this is only one potential hypothesis for higher frequency of reactions to this version of the vaccine.
Patients with a history of allergic disease are more likely to develop a reaction but not significantly more likely to have a reaction that is more difficult to manage, according to Kimberly G. Blumenthal, MD, quality and safety officer for allergy, and codirector of the clinical epidemiology program in the division of rheumatology, allergy, and immunology at Mass General.
Anaphylaxis has been associated with COVD-19 vaccines just as it has with essentially every injectable vaccine, Dr. Blumenthal said during the same session. But the risk is very low, and it stays low even among those with a history of severe hypersensitivity reactions in the past.
Among the data collected from more than 52,000 vaccinated MGBHS employees, 0.9% had a history of severe allergic reaction to a prior vaccine. Of these, 11.6% had an allergic reaction to the COVID-19 vaccine. This was more than twice the 4.6% rate of allergic reactions among employees without a history of allergic reactions, but serious consequences were rare in both groups.
Of those with a reaction to the first dose, all but 2.4% took a subsequent dose. Again, serious reactions were exceedingly rare. These serious reactions did include anaphylaxis and hospitalization in 3% of patients, but there were no fatalities and all resolved.
The absence of serious sequelae from a reaction to a COVID-19 vaccine must be considered within the context of the benefit, which includes protection from death and hospitalization from the virus, according to Dr. Blumenthal. Citing the evidence that first-shot reactions are a source of vaccine hesitancy, she agreed that it is important to educate patients about relative risks.
“Even in our own cohort of MGBHS employees, we have people, including those who had been provaccine in the past, become hesitant,” commented Dr. Blumenthal, who said there are data from the Kaiser Permanente System showing similar vaccine reluctance following a first-shot reaction.
After more than 500 million doses of the Moderna and Pfizer vaccines had been administered worldwide, there was not a single reported death from anaphylaxis. Although Dr. Blumenthal said that an unconfirmed death of this type had been recently reported, she emphasized that this single death, if valid, is dwarfed by the lives saved with vaccination.
Asked about her strategy for counseling patients with vaccine hesitancy, Dr. Freeman said the body of safety data is large and compelling. There is overwhelming evidence of a favorable benefit-to-risk ratio overall and among those with a first-shot reaction.
“I can reassure them on the basis of the data,” Dr. Freeman said in an interview. “Less than half will have a reaction to the second shot and even if they do have a reaction, it is likely to be less severe.”
Although the main message is that vaccination is potentially lifesaving and far outweighs any risks, Freeman specifically gives this message to those hesitant to take a second shot after a first-shot reaction: “I can get you through it.”
Dr. Freeman encouraged health care professionals to report cases of COVID-19 vaccine–related dermatologic side effects to the American Academy of Dermatology / International League of Dermatologic Societies COVID-19 dermatology registry. Dermatologic manifestations of COVID-19 can also be reported to the registry.
Dr. Freeman disclosed receiving grants/research funding from the International League of Dermatologic Societies and from the National Institutes of Health. Dr. Blumenthal disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT AAD 2022
Melanoma increasing, but is this overdiagnosis?
Melanoma has been increasing in incidence in the United States over the last few decades, but is this a true increase? Or is this a case of overdiagnosis, fueled by screening?
A new study argues the case for overdiagnosis.
commented lead author lead author Adewole Adamson, MD, an assistant professor of internal medicine, division of dermatology, at the University of Texas at Austin.
He posted this conclusion on Twitter after the study was published in JAMA Dermatology.
“The discrepancies in incidence and mortality trends found in this cohort study suggest considerable overdiagnosis of melanoma occurring among White patients in the U.S.,” the authors concluded.
They estimated that an estimated 59% of White women and 60% of White men with melanoma were overdiagnosed in 2014.
These results are similar to those from a recent study from Australia, which used a different method of assessing overdiagnosis. Those findings estimated that 54%-58% of melanoma cases represented overdiagnosis in Australia, Dr. Adamson noted.
“Our estimates shed light on the HUGE scope of this problem in the United States that we need to address,” Dr. Adamson commented on Twitter. “Calls for screening for melanoma in the general public will only push these numbers higher, and make patients out of healthy people.”
“Screening the general population for melanoma has never been shown to save lives and likely is responsible for the increase in melanoma overdiagnosis,” Dr. Adamson said in an interview. “Screening average- and/or low-risk patients is of low value and the harms may outweigh the theoretical benefits.”
Screening programs should be directed to those who may derive the most benefit. “Screening should be limited to high-risk patients such as older White men, patients with a lot of atypical nevi, heavy sun exposure, fair skin, and red hair,” he said. “Just like for other cancers, such as breast, prostate, and colorectal, there should be clear guidelines as to which populations to screen, as well as when to start and when to stop screening.”
Overdiagnosis is defined as the diagnosis of cancer that would never have caused any symptoms or problems in a patient’s lifetime. But therein lies the problem, explained Dr. Adamson. “Because we do not know which early, screen-detected skin cancers would be destined to progress, we are obligated to treat all of them.” There is evidence to suggest that melanoma in situ is not an obligate precursor lesion to invasive melanoma, similar to the situation in which not all ductal carcinoma in situ leads to invasive breast cancer. “It is possible that less aggressive management strategies could be the subject of future studies,” he said.
Patients out of healthy people
For their study, Dr. Adamson and colleagues compared rates of melanoma among White and Black patients. Melanoma is much less common among Black individuals, and they are also less likely to be screened. Additionally, screening rates among Black patients have remained more or less the same over the last decades, whereas screening has increased in White patients.
The team used trends in mortality as a result of melanoma in Black patients as a marker for improvements in medical care. From this, they estimated the expected mortality trends in White patients if medical care had not improved. This served as a marker for the change in true cancer occurrence. Overdiagnosis was calculated as the difference between observed incidence and estimated true cancer occurrence.
The incidence of melanoma rose dramatically among White patients from 1975 to 2014, increasing about fourfold in White women (incidence rate ratio, 4.01) and sixfold in White men (IRR, 5.97).
At the same time, there was much smaller increase (of less than 25%) in the incidence of melanoma in both Black women and Black men.
In that time period, melanoma-related mortality decreased approximately 25% in Black women and men; it remained stable in White women, but increased almost 50% in White men.
Had medical care not improved, estimated mortality would have increased 60% in White women and more than doubled in White men, the authors assert.
Guidelines needed
“Recognizing and addressing overdiagnosis is important,” said Anthony J. Olszanski, MD, RPh, associate professor, department of hematology/oncology at Fox Chase Cancer Center, Philadelphia, who was approached for comment on the paper.
That said, Dr. Olszanski noted that this particular study has important limitations. “It is, by nature, a retrospective study using data from the [Surveillance, Epidemiology, and End Results] database registry, limited to patients only in the U.S., and uses a control group of Black patients to estimate overdiagnosis in White patients. These important factors can certainly influence their findings. However, the paper also notes that White men have realized a true increase in diagnosis, backed by a notable increase in mortality.”
The findings should and do raise a number of provocative questions, Dr. Olszanski emphasized. “Should we curtail public screening? Should we mandate revised guidelines for biopsies or pathologic diagnosis?
“As a medical oncologist,” he continued, “I treat patients who clearly do not have benign disease and so it is easy for me to be biased toward aggressive screening. However, it is my opinion that we should develop guidelines aimed at lessening this apparent overdiagnosis.”
These guidelines should be based on prospective studies and would better define which lesions are most suspect and should be biopsied, which are rational for ongoing surveillance, and what pathologic features are most consistent with melanoma, he noted. “We also need to continue to educate the public, as all too often I see the patient who ignored a lesion that was changing over time. A changing lesion requires medical attention. Importantly, we likewise need to improve our commitment in educating the public about the risks of excessive ultraviolet radiation exposure and how to avoid it, as prevention continues to be a most prudent course.”
Screening catches disease early
Another expert approached for comment emphasized that identifying melanomas early on may prevent the need for aggressive therapy. “Many primary melanomas in the U.S. are diagnosed now at an early stage and are cured with surgery, and that hardly constitutes overdiagnosis,” said Jeffrey S. Weber, MD, PhD, deputy director of the Perlmutter Cancer Center and codirector of the melanoma research program, New York University Langone Health.
“In addition, the death rate from melanoma is likely decreased due to the advent of more effective therapies for metastatic disease, and the increasing use of adjuvant immune and targeted therapies that are highly effective at preventing relapse and undoubtedly at prolonging survival, but they have been approved only since 2017-2018,” he added.
This study was supported in part by the Robert Wood Johnson Foundation. Dr. Adamson and Dr. Olszanski disclosed no relevant financial relationships. Dr. Weber disclosed relationships with numerous pharmaceutical companies and holds equity in CytoMx, Biond, Neximmune, and Immunimax.
A version of this article first appeared on Medscape.com.
Melanoma has been increasing in incidence in the United States over the last few decades, but is this a true increase? Or is this a case of overdiagnosis, fueled by screening?
A new study argues the case for overdiagnosis.
commented lead author lead author Adewole Adamson, MD, an assistant professor of internal medicine, division of dermatology, at the University of Texas at Austin.
He posted this conclusion on Twitter after the study was published in JAMA Dermatology.
“The discrepancies in incidence and mortality trends found in this cohort study suggest considerable overdiagnosis of melanoma occurring among White patients in the U.S.,” the authors concluded.
They estimated that an estimated 59% of White women and 60% of White men with melanoma were overdiagnosed in 2014.
These results are similar to those from a recent study from Australia, which used a different method of assessing overdiagnosis. Those findings estimated that 54%-58% of melanoma cases represented overdiagnosis in Australia, Dr. Adamson noted.
“Our estimates shed light on the HUGE scope of this problem in the United States that we need to address,” Dr. Adamson commented on Twitter. “Calls for screening for melanoma in the general public will only push these numbers higher, and make patients out of healthy people.”
“Screening the general population for melanoma has never been shown to save lives and likely is responsible for the increase in melanoma overdiagnosis,” Dr. Adamson said in an interview. “Screening average- and/or low-risk patients is of low value and the harms may outweigh the theoretical benefits.”
Screening programs should be directed to those who may derive the most benefit. “Screening should be limited to high-risk patients such as older White men, patients with a lot of atypical nevi, heavy sun exposure, fair skin, and red hair,” he said. “Just like for other cancers, such as breast, prostate, and colorectal, there should be clear guidelines as to which populations to screen, as well as when to start and when to stop screening.”
Overdiagnosis is defined as the diagnosis of cancer that would never have caused any symptoms or problems in a patient’s lifetime. But therein lies the problem, explained Dr. Adamson. “Because we do not know which early, screen-detected skin cancers would be destined to progress, we are obligated to treat all of them.” There is evidence to suggest that melanoma in situ is not an obligate precursor lesion to invasive melanoma, similar to the situation in which not all ductal carcinoma in situ leads to invasive breast cancer. “It is possible that less aggressive management strategies could be the subject of future studies,” he said.
Patients out of healthy people
For their study, Dr. Adamson and colleagues compared rates of melanoma among White and Black patients. Melanoma is much less common among Black individuals, and they are also less likely to be screened. Additionally, screening rates among Black patients have remained more or less the same over the last decades, whereas screening has increased in White patients.
The team used trends in mortality as a result of melanoma in Black patients as a marker for improvements in medical care. From this, they estimated the expected mortality trends in White patients if medical care had not improved. This served as a marker for the change in true cancer occurrence. Overdiagnosis was calculated as the difference between observed incidence and estimated true cancer occurrence.
The incidence of melanoma rose dramatically among White patients from 1975 to 2014, increasing about fourfold in White women (incidence rate ratio, 4.01) and sixfold in White men (IRR, 5.97).
At the same time, there was much smaller increase (of less than 25%) in the incidence of melanoma in both Black women and Black men.
In that time period, melanoma-related mortality decreased approximately 25% in Black women and men; it remained stable in White women, but increased almost 50% in White men.
Had medical care not improved, estimated mortality would have increased 60% in White women and more than doubled in White men, the authors assert.
Guidelines needed
“Recognizing and addressing overdiagnosis is important,” said Anthony J. Olszanski, MD, RPh, associate professor, department of hematology/oncology at Fox Chase Cancer Center, Philadelphia, who was approached for comment on the paper.
That said, Dr. Olszanski noted that this particular study has important limitations. “It is, by nature, a retrospective study using data from the [Surveillance, Epidemiology, and End Results] database registry, limited to patients only in the U.S., and uses a control group of Black patients to estimate overdiagnosis in White patients. These important factors can certainly influence their findings. However, the paper also notes that White men have realized a true increase in diagnosis, backed by a notable increase in mortality.”
The findings should and do raise a number of provocative questions, Dr. Olszanski emphasized. “Should we curtail public screening? Should we mandate revised guidelines for biopsies or pathologic diagnosis?
“As a medical oncologist,” he continued, “I treat patients who clearly do not have benign disease and so it is easy for me to be biased toward aggressive screening. However, it is my opinion that we should develop guidelines aimed at lessening this apparent overdiagnosis.”
These guidelines should be based on prospective studies and would better define which lesions are most suspect and should be biopsied, which are rational for ongoing surveillance, and what pathologic features are most consistent with melanoma, he noted. “We also need to continue to educate the public, as all too often I see the patient who ignored a lesion that was changing over time. A changing lesion requires medical attention. Importantly, we likewise need to improve our commitment in educating the public about the risks of excessive ultraviolet radiation exposure and how to avoid it, as prevention continues to be a most prudent course.”
Screening catches disease early
Another expert approached for comment emphasized that identifying melanomas early on may prevent the need for aggressive therapy. “Many primary melanomas in the U.S. are diagnosed now at an early stage and are cured with surgery, and that hardly constitutes overdiagnosis,” said Jeffrey S. Weber, MD, PhD, deputy director of the Perlmutter Cancer Center and codirector of the melanoma research program, New York University Langone Health.
“In addition, the death rate from melanoma is likely decreased due to the advent of more effective therapies for metastatic disease, and the increasing use of adjuvant immune and targeted therapies that are highly effective at preventing relapse and undoubtedly at prolonging survival, but they have been approved only since 2017-2018,” he added.
This study was supported in part by the Robert Wood Johnson Foundation. Dr. Adamson and Dr. Olszanski disclosed no relevant financial relationships. Dr. Weber disclosed relationships with numerous pharmaceutical companies and holds equity in CytoMx, Biond, Neximmune, and Immunimax.
A version of this article first appeared on Medscape.com.
Melanoma has been increasing in incidence in the United States over the last few decades, but is this a true increase? Or is this a case of overdiagnosis, fueled by screening?
A new study argues the case for overdiagnosis.
commented lead author lead author Adewole Adamson, MD, an assistant professor of internal medicine, division of dermatology, at the University of Texas at Austin.
He posted this conclusion on Twitter after the study was published in JAMA Dermatology.
“The discrepancies in incidence and mortality trends found in this cohort study suggest considerable overdiagnosis of melanoma occurring among White patients in the U.S.,” the authors concluded.
They estimated that an estimated 59% of White women and 60% of White men with melanoma were overdiagnosed in 2014.
These results are similar to those from a recent study from Australia, which used a different method of assessing overdiagnosis. Those findings estimated that 54%-58% of melanoma cases represented overdiagnosis in Australia, Dr. Adamson noted.
“Our estimates shed light on the HUGE scope of this problem in the United States that we need to address,” Dr. Adamson commented on Twitter. “Calls for screening for melanoma in the general public will only push these numbers higher, and make patients out of healthy people.”
“Screening the general population for melanoma has never been shown to save lives and likely is responsible for the increase in melanoma overdiagnosis,” Dr. Adamson said in an interview. “Screening average- and/or low-risk patients is of low value and the harms may outweigh the theoretical benefits.”
Screening programs should be directed to those who may derive the most benefit. “Screening should be limited to high-risk patients such as older White men, patients with a lot of atypical nevi, heavy sun exposure, fair skin, and red hair,” he said. “Just like for other cancers, such as breast, prostate, and colorectal, there should be clear guidelines as to which populations to screen, as well as when to start and when to stop screening.”
Overdiagnosis is defined as the diagnosis of cancer that would never have caused any symptoms or problems in a patient’s lifetime. But therein lies the problem, explained Dr. Adamson. “Because we do not know which early, screen-detected skin cancers would be destined to progress, we are obligated to treat all of them.” There is evidence to suggest that melanoma in situ is not an obligate precursor lesion to invasive melanoma, similar to the situation in which not all ductal carcinoma in situ leads to invasive breast cancer. “It is possible that less aggressive management strategies could be the subject of future studies,” he said.
Patients out of healthy people
For their study, Dr. Adamson and colleagues compared rates of melanoma among White and Black patients. Melanoma is much less common among Black individuals, and they are also less likely to be screened. Additionally, screening rates among Black patients have remained more or less the same over the last decades, whereas screening has increased in White patients.
The team used trends in mortality as a result of melanoma in Black patients as a marker for improvements in medical care. From this, they estimated the expected mortality trends in White patients if medical care had not improved. This served as a marker for the change in true cancer occurrence. Overdiagnosis was calculated as the difference between observed incidence and estimated true cancer occurrence.
The incidence of melanoma rose dramatically among White patients from 1975 to 2014, increasing about fourfold in White women (incidence rate ratio, 4.01) and sixfold in White men (IRR, 5.97).
At the same time, there was much smaller increase (of less than 25%) in the incidence of melanoma in both Black women and Black men.
In that time period, melanoma-related mortality decreased approximately 25% in Black women and men; it remained stable in White women, but increased almost 50% in White men.
Had medical care not improved, estimated mortality would have increased 60% in White women and more than doubled in White men, the authors assert.
Guidelines needed
“Recognizing and addressing overdiagnosis is important,” said Anthony J. Olszanski, MD, RPh, associate professor, department of hematology/oncology at Fox Chase Cancer Center, Philadelphia, who was approached for comment on the paper.
That said, Dr. Olszanski noted that this particular study has important limitations. “It is, by nature, a retrospective study using data from the [Surveillance, Epidemiology, and End Results] database registry, limited to patients only in the U.S., and uses a control group of Black patients to estimate overdiagnosis in White patients. These important factors can certainly influence their findings. However, the paper also notes that White men have realized a true increase in diagnosis, backed by a notable increase in mortality.”
The findings should and do raise a number of provocative questions, Dr. Olszanski emphasized. “Should we curtail public screening? Should we mandate revised guidelines for biopsies or pathologic diagnosis?
“As a medical oncologist,” he continued, “I treat patients who clearly do not have benign disease and so it is easy for me to be biased toward aggressive screening. However, it is my opinion that we should develop guidelines aimed at lessening this apparent overdiagnosis.”
These guidelines should be based on prospective studies and would better define which lesions are most suspect and should be biopsied, which are rational for ongoing surveillance, and what pathologic features are most consistent with melanoma, he noted. “We also need to continue to educate the public, as all too often I see the patient who ignored a lesion that was changing over time. A changing lesion requires medical attention. Importantly, we likewise need to improve our commitment in educating the public about the risks of excessive ultraviolet radiation exposure and how to avoid it, as prevention continues to be a most prudent course.”
Screening catches disease early
Another expert approached for comment emphasized that identifying melanomas early on may prevent the need for aggressive therapy. “Many primary melanomas in the U.S. are diagnosed now at an early stage and are cured with surgery, and that hardly constitutes overdiagnosis,” said Jeffrey S. Weber, MD, PhD, deputy director of the Perlmutter Cancer Center and codirector of the melanoma research program, New York University Langone Health.
“In addition, the death rate from melanoma is likely decreased due to the advent of more effective therapies for metastatic disease, and the increasing use of adjuvant immune and targeted therapies that are highly effective at preventing relapse and undoubtedly at prolonging survival, but they have been approved only since 2017-2018,” he added.
This study was supported in part by the Robert Wood Johnson Foundation. Dr. Adamson and Dr. Olszanski disclosed no relevant financial relationships. Dr. Weber disclosed relationships with numerous pharmaceutical companies and holds equity in CytoMx, Biond, Neximmune, and Immunimax.
A version of this article first appeared on Medscape.com.
FROM JAMA DERMATOLOGY