User login
Evinacumab shows promise for HoFH in top line results
(HoFH), according to a release from the company developing the drug.
LDL cholesterol levels were 255 mg/dL on average for patients at the outset of the trial despite treatment with other lipid-lowering therapies; however, combining this drug with lipid-lowering therapies including maximally-tolerated statins, PCSK9 inhibitors, and LDL apheresis, reduced LDL cholesterol by an average of 49% by week 24 relative to treatment with lipid-lowering therapies alone (P less than .0001). Furthermore, 47% of patients taking evinacumab achieved LDL cholesterol levels under 100 mg/dL by that time point versus 23% of those taking lipid-lowering therapies only.
Treatment with evinacumab showed lowering effects as early as the first assessment at 2 weeks, and these effects were maintained.
HoFH is an inherited, rare, but serious condition estimated to affect 1,300 people in the United States; it can lead to early atherosclerotic disease, and even teenagers with this genetic disorder can suffer cardiac events. Further details and data from the trial, called ELIPSE HoFH, will be reported at a future medical meeting, and will be submitted to the Food and Drug Administration for consideration.
Evinacumab is an investigational, fully-human, monoclonal antibody that specifically binds to angiopoietin-like protein 3 (ANGPTL3), which acts as an inhibitor of lipoprotein lipase and endothelial lipase, and appears to play a central role in lipoprotein metabolism.
Evinacumab was granted breakthrough therapy designation for treatment of HoFH by the FDA in 2017, which entails an expedited review and development process for this drug because preliminary results have suggested it could have a substantial effect on a life-threatening or serious condition.
The company’s full release can be read on its website.
(HoFH), according to a release from the company developing the drug.
LDL cholesterol levels were 255 mg/dL on average for patients at the outset of the trial despite treatment with other lipid-lowering therapies; however, combining this drug with lipid-lowering therapies including maximally-tolerated statins, PCSK9 inhibitors, and LDL apheresis, reduced LDL cholesterol by an average of 49% by week 24 relative to treatment with lipid-lowering therapies alone (P less than .0001). Furthermore, 47% of patients taking evinacumab achieved LDL cholesterol levels under 100 mg/dL by that time point versus 23% of those taking lipid-lowering therapies only.
Treatment with evinacumab showed lowering effects as early as the first assessment at 2 weeks, and these effects were maintained.
HoFH is an inherited, rare, but serious condition estimated to affect 1,300 people in the United States; it can lead to early atherosclerotic disease, and even teenagers with this genetic disorder can suffer cardiac events. Further details and data from the trial, called ELIPSE HoFH, will be reported at a future medical meeting, and will be submitted to the Food and Drug Administration for consideration.
Evinacumab is an investigational, fully-human, monoclonal antibody that specifically binds to angiopoietin-like protein 3 (ANGPTL3), which acts as an inhibitor of lipoprotein lipase and endothelial lipase, and appears to play a central role in lipoprotein metabolism.
Evinacumab was granted breakthrough therapy designation for treatment of HoFH by the FDA in 2017, which entails an expedited review and development process for this drug because preliminary results have suggested it could have a substantial effect on a life-threatening or serious condition.
The company’s full release can be read on its website.
(HoFH), according to a release from the company developing the drug.
LDL cholesterol levels were 255 mg/dL on average for patients at the outset of the trial despite treatment with other lipid-lowering therapies; however, combining this drug with lipid-lowering therapies including maximally-tolerated statins, PCSK9 inhibitors, and LDL apheresis, reduced LDL cholesterol by an average of 49% by week 24 relative to treatment with lipid-lowering therapies alone (P less than .0001). Furthermore, 47% of patients taking evinacumab achieved LDL cholesterol levels under 100 mg/dL by that time point versus 23% of those taking lipid-lowering therapies only.
Treatment with evinacumab showed lowering effects as early as the first assessment at 2 weeks, and these effects were maintained.
HoFH is an inherited, rare, but serious condition estimated to affect 1,300 people in the United States; it can lead to early atherosclerotic disease, and even teenagers with this genetic disorder can suffer cardiac events. Further details and data from the trial, called ELIPSE HoFH, will be reported at a future medical meeting, and will be submitted to the Food and Drug Administration for consideration.
Evinacumab is an investigational, fully-human, monoclonal antibody that specifically binds to angiopoietin-like protein 3 (ANGPTL3), which acts as an inhibitor of lipoprotein lipase and endothelial lipase, and appears to play a central role in lipoprotein metabolism.
Evinacumab was granted breakthrough therapy designation for treatment of HoFH by the FDA in 2017, which entails an expedited review and development process for this drug because preliminary results have suggested it could have a substantial effect on a life-threatening or serious condition.
The company’s full release can be read on its website.
NSAIDs a significant mediator of cardiovascular risk in osteoarthritis
Writing in Arthritis & Rheumatology, researchers reported the outcomes of a longitudinal, population-based cohort study of 7,743 individuals with osteoarthritis patients and 23,229 age- and sex-matched controls without osteoarthritis.
“The prevailing hypothesis in the OA to CVD relationship has been that OA patients frequently take NSAIDs to control their pain and inflammation and that this may lead to them developing CVD,” wrote Mohammad Atiquzzaman, a PhD student at the University of British Columbia, Vancouver, and his coauthors. However they commented that no studies had so far examined this directly in patients with osteoarthritis.
Overall, people with osteoarthritis had a significant 23% higher risk of cardiovascular disease, compared with controls, after adjustment for factors such body mass index, hypertension, diabetes, hyperlipidemia, and socioeconomic status. They also had a 42% higher risk of congestive heart failure, 17% higher risk of ischemic heart disease, and 14% higher risk of stroke.
NSAID use was five times more common among people with osteoarthritis, and NSAIDs alone were associated with a greater than fourfold higher risk of cardiovascular disease, after adjusting for osteoarthritis and other potential confounders.
When the authors performed modeling to break down the effect of osteoarthritis on CVD risk into the direct effect of osteoarthritis itself and the indirect effect mediated by NSAID use, they concluded that 41% of the total effect of osteoarthritis on cardiovascular risk was mediated by NSAIDs. The effect of NSAIDs was particularly pronounced for stroke, in which cases they estimated that the drugs contributed to 64% of the increased in risk, and in ischemic heart disease, in which they contributed to 56% of the increased risk.
Subgroup analysis suggested that conventional NSAIDs were responsible for around 29% of the total increased risk of cardiovascular disease, while selective COX-2 inhibitors, or coxibs, such as celecoxib, lumiracoxib, rofecoxib, and valdecoxib mediated around 21%. For ischemic heart disease, conventional NSAIDs explained around 45% of the increased risk, while selective coxibs explained around 32% of the risk. Similarly, with congestive heart failure and stroke, the proportion of risk mediated by NSAIDs was higher for conventional NSAIDs, compared with coxibs.
The authors noted that while a number of previous studies have found osteoarthritis is an independent risk factor for cardiovascular disease, theirs was the first study to specifically examine the role that NSAIDs play in that increased risk.
However, they noted that their information on NSAID use was gleaned from prescription claims data, which did not include information on over-the-counter NSAID use. Their analysis was also unable to include information on family history of cardiovascular disease, smoking, and physical activity, which are important cardiovascular disease risk factors. They did observe that the rates of obesity were higher among the osteoarthritis group when compared with controls (29% vs. 20%), and hypertension and COPD were also more common among individuals with osteoarthritis.
There was no outside funding for the study, and the authors had no conflicts of interest to declare.
SOURCE: Atiquzzaman M et al. Arthritis Rheumatol. 2019 Aug 6. doi: 10.1002/art.41027
Writing in Arthritis & Rheumatology, researchers reported the outcomes of a longitudinal, population-based cohort study of 7,743 individuals with osteoarthritis patients and 23,229 age- and sex-matched controls without osteoarthritis.
“The prevailing hypothesis in the OA to CVD relationship has been that OA patients frequently take NSAIDs to control their pain and inflammation and that this may lead to them developing CVD,” wrote Mohammad Atiquzzaman, a PhD student at the University of British Columbia, Vancouver, and his coauthors. However they commented that no studies had so far examined this directly in patients with osteoarthritis.
Overall, people with osteoarthritis had a significant 23% higher risk of cardiovascular disease, compared with controls, after adjustment for factors such body mass index, hypertension, diabetes, hyperlipidemia, and socioeconomic status. They also had a 42% higher risk of congestive heart failure, 17% higher risk of ischemic heart disease, and 14% higher risk of stroke.
NSAID use was five times more common among people with osteoarthritis, and NSAIDs alone were associated with a greater than fourfold higher risk of cardiovascular disease, after adjusting for osteoarthritis and other potential confounders.
When the authors performed modeling to break down the effect of osteoarthritis on CVD risk into the direct effect of osteoarthritis itself and the indirect effect mediated by NSAID use, they concluded that 41% of the total effect of osteoarthritis on cardiovascular risk was mediated by NSAIDs. The effect of NSAIDs was particularly pronounced for stroke, in which cases they estimated that the drugs contributed to 64% of the increased in risk, and in ischemic heart disease, in which they contributed to 56% of the increased risk.
Subgroup analysis suggested that conventional NSAIDs were responsible for around 29% of the total increased risk of cardiovascular disease, while selective COX-2 inhibitors, or coxibs, such as celecoxib, lumiracoxib, rofecoxib, and valdecoxib mediated around 21%. For ischemic heart disease, conventional NSAIDs explained around 45% of the increased risk, while selective coxibs explained around 32% of the risk. Similarly, with congestive heart failure and stroke, the proportion of risk mediated by NSAIDs was higher for conventional NSAIDs, compared with coxibs.
The authors noted that while a number of previous studies have found osteoarthritis is an independent risk factor for cardiovascular disease, theirs was the first study to specifically examine the role that NSAIDs play in that increased risk.
However, they noted that their information on NSAID use was gleaned from prescription claims data, which did not include information on over-the-counter NSAID use. Their analysis was also unable to include information on family history of cardiovascular disease, smoking, and physical activity, which are important cardiovascular disease risk factors. They did observe that the rates of obesity were higher among the osteoarthritis group when compared with controls (29% vs. 20%), and hypertension and COPD were also more common among individuals with osteoarthritis.
There was no outside funding for the study, and the authors had no conflicts of interest to declare.
SOURCE: Atiquzzaman M et al. Arthritis Rheumatol. 2019 Aug 6. doi: 10.1002/art.41027
Writing in Arthritis & Rheumatology, researchers reported the outcomes of a longitudinal, population-based cohort study of 7,743 individuals with osteoarthritis patients and 23,229 age- and sex-matched controls without osteoarthritis.
“The prevailing hypothesis in the OA to CVD relationship has been that OA patients frequently take NSAIDs to control their pain and inflammation and that this may lead to them developing CVD,” wrote Mohammad Atiquzzaman, a PhD student at the University of British Columbia, Vancouver, and his coauthors. However they commented that no studies had so far examined this directly in patients with osteoarthritis.
Overall, people with osteoarthritis had a significant 23% higher risk of cardiovascular disease, compared with controls, after adjustment for factors such body mass index, hypertension, diabetes, hyperlipidemia, and socioeconomic status. They also had a 42% higher risk of congestive heart failure, 17% higher risk of ischemic heart disease, and 14% higher risk of stroke.
NSAID use was five times more common among people with osteoarthritis, and NSAIDs alone were associated with a greater than fourfold higher risk of cardiovascular disease, after adjusting for osteoarthritis and other potential confounders.
When the authors performed modeling to break down the effect of osteoarthritis on CVD risk into the direct effect of osteoarthritis itself and the indirect effect mediated by NSAID use, they concluded that 41% of the total effect of osteoarthritis on cardiovascular risk was mediated by NSAIDs. The effect of NSAIDs was particularly pronounced for stroke, in which cases they estimated that the drugs contributed to 64% of the increased in risk, and in ischemic heart disease, in which they contributed to 56% of the increased risk.
Subgroup analysis suggested that conventional NSAIDs were responsible for around 29% of the total increased risk of cardiovascular disease, while selective COX-2 inhibitors, or coxibs, such as celecoxib, lumiracoxib, rofecoxib, and valdecoxib mediated around 21%. For ischemic heart disease, conventional NSAIDs explained around 45% of the increased risk, while selective coxibs explained around 32% of the risk. Similarly, with congestive heart failure and stroke, the proportion of risk mediated by NSAIDs was higher for conventional NSAIDs, compared with coxibs.
The authors noted that while a number of previous studies have found osteoarthritis is an independent risk factor for cardiovascular disease, theirs was the first study to specifically examine the role that NSAIDs play in that increased risk.
However, they noted that their information on NSAID use was gleaned from prescription claims data, which did not include information on over-the-counter NSAID use. Their analysis was also unable to include information on family history of cardiovascular disease, smoking, and physical activity, which are important cardiovascular disease risk factors. They did observe that the rates of obesity were higher among the osteoarthritis group when compared with controls (29% vs. 20%), and hypertension and COPD were also more common among individuals with osteoarthritis.
There was no outside funding for the study, and the authors had no conflicts of interest to declare.
SOURCE: Atiquzzaman M et al. Arthritis Rheumatol. 2019 Aug 6. doi: 10.1002/art.41027
FROM ARTHRITIS & RHEUMATOLOGY
Psoriasis patients on biologics show improved heart health
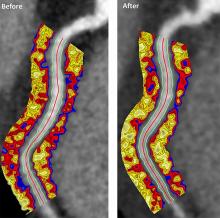
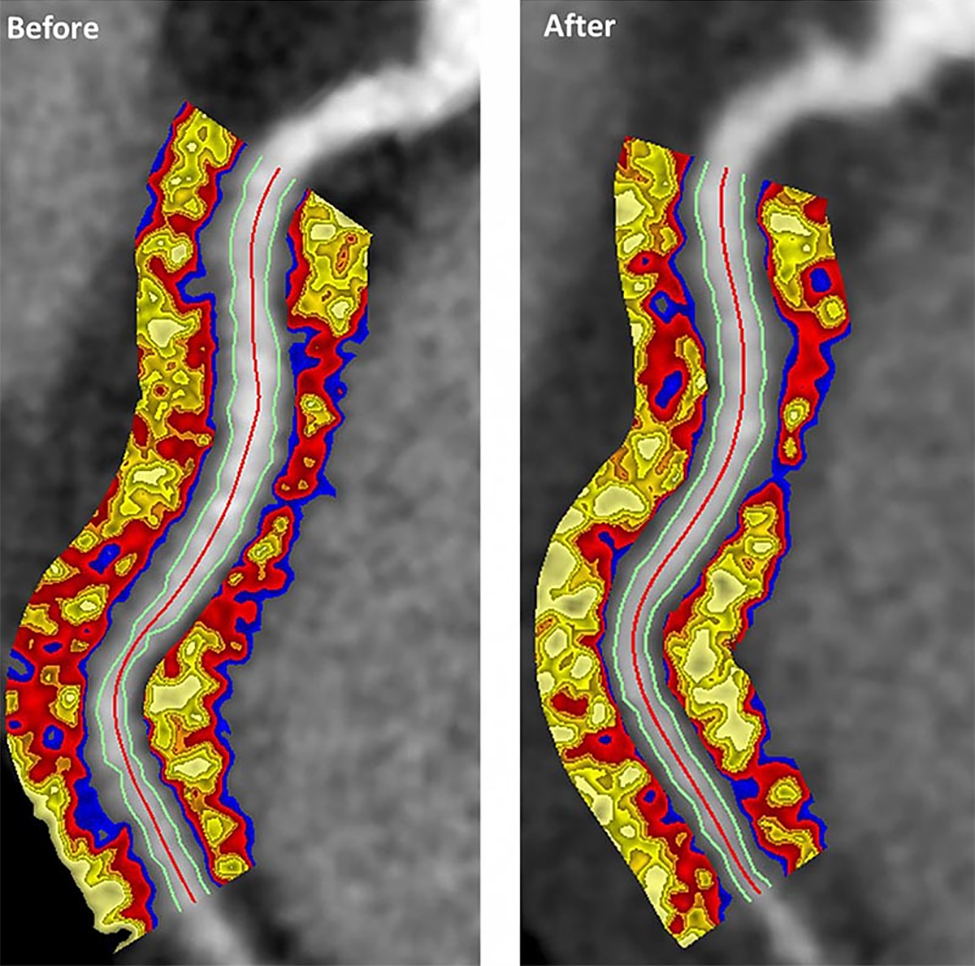
Biologics improved coronary inflammation as well as psoriasis symptoms, according to data from the perivascular fat attenuation index in 134 adults identified using coronary CT angiography.
“The perivascular fat attenuation index [FAI] is a [CT]-based, novel, noninvasive imaging technique that allows for direct visualization and quantification of coronary inflammation using differential mapping of attenuation gradients in pericoronary fat,” wrote Youssef A. Elnabawi, MD, of the National Heart, Lung, and Blood Institute and colleagues. Biologics have been associated with reduced noncalcified coronary plaques in psoriasis patients, which suggests possible reduction in coronary inflammation as well.
In a study published in JAMA Cardiology, the researchers analyzed data from 134 adults with moderate to severe psoriasis who received no biologic therapy for at least 3 months before starting the study. Of these, 52 chose not to receive biologics, and served as controls while being treated with topical or light therapies. The participants are part of the Psoriasis Atherosclerosis Cardiometabolic Initiative, an ongoing, prospective cohort study. The average age of the patients was 51 years, and 63% were male.
The 82 patients given biologics received anti–tumor necrosis factor–alpha, anti–interleukin-12/23, or anti-IL-17 for 1 year. Overall, patients on biologics showed a significant decrease in FAI from a median of –71.22 Hounsfield units (HU) at baseline to a median of –76.06 at 1 year. These patients also showed significant improvement in Psoriasis Area and Severity Index scores, from a median of 7.7 at baseline to a median of 3.2 at 1 year. The control patients not on biologics showed no significant changes in FAI, with a median of –71.98 HU at baseline and –72.66 HU at 1 year.
The changes were consistent among the various biologics used, and The median FAI for patients on anti–tumor necrosis factor–alpha changed from –71.25 at baseline to –75.49 at 1 year; median FAI for both IL-12/23 and anti-IL-17 treatment groups changed from –71.18 HU at baseline to –76.92 at 1 year.
In addition, only patients treated with biologics showed a significant reduction in median C-reactive protein levels from baseline (2.2 mg/L vs. 1.3 mg/L). The changes in FAI were not associated with the presence of coronary plaques, the researchers noted.
The study findings were limited by several factors, including the observational design, small size, and lack of data on cardiovascular endpoints. “Future studies will be needed to explore whether the residual CV risk detected by perivascular FAI can be attenuated using targeted anti-inflammatory interventions,” they wrote.
However, the results suggest that biologics impact coronary vasculature at the microenvironmental level, and that FAI can be a noninvasive, cost-effective way to stratify patients at increased risk for cardiovascular disease, the researchers noted.
“We believe that the strength of perivascular FAI in risk stratifying patients with increased coronary inflammation will allow for better identification of patients at increased risk of future myocardial events that are not captured by traditional CV risk factors,” they wrote.
The study was funded by the National Institutes of Health, several research foundations, Elsevier, Colgate-Palmolive, and Genentech. Dr. Elnabawi had no financial conflicts to disclose; several coauthors reported relationships with multiple companies. One coauthor disclosed a pending and licensed patent to a novel tool for cardiovascular risk stratification based on the CT attenuation of perivascular tissue (OxScore) and a pending and licensed patent to perivascular texture index.
SOURCE: Elnabawi YA et al. JAMA Cardiol. 2019 Jul 31. doi: 10.1001/jamacardio.2019.2589.
Biologics improved coronary inflammation as well as psoriasis symptoms, according to data from the perivascular fat attenuation index in 134 adults identified using coronary CT angiography.
“The perivascular fat attenuation index [FAI] is a [CT]-based, novel, noninvasive imaging technique that allows for direct visualization and quantification of coronary inflammation using differential mapping of attenuation gradients in pericoronary fat,” wrote Youssef A. Elnabawi, MD, of the National Heart, Lung, and Blood Institute and colleagues. Biologics have been associated with reduced noncalcified coronary plaques in psoriasis patients, which suggests possible reduction in coronary inflammation as well.
In a study published in JAMA Cardiology, the researchers analyzed data from 134 adults with moderate to severe psoriasis who received no biologic therapy for at least 3 months before starting the study. Of these, 52 chose not to receive biologics, and served as controls while being treated with topical or light therapies. The participants are part of the Psoriasis Atherosclerosis Cardiometabolic Initiative, an ongoing, prospective cohort study. The average age of the patients was 51 years, and 63% were male.
The 82 patients given biologics received anti–tumor necrosis factor–alpha, anti–interleukin-12/23, or anti-IL-17 for 1 year. Overall, patients on biologics showed a significant decrease in FAI from a median of –71.22 Hounsfield units (HU) at baseline to a median of –76.06 at 1 year. These patients also showed significant improvement in Psoriasis Area and Severity Index scores, from a median of 7.7 at baseline to a median of 3.2 at 1 year. The control patients not on biologics showed no significant changes in FAI, with a median of –71.98 HU at baseline and –72.66 HU at 1 year.
The changes were consistent among the various biologics used, and The median FAI for patients on anti–tumor necrosis factor–alpha changed from –71.25 at baseline to –75.49 at 1 year; median FAI for both IL-12/23 and anti-IL-17 treatment groups changed from –71.18 HU at baseline to –76.92 at 1 year.
In addition, only patients treated with biologics showed a significant reduction in median C-reactive protein levels from baseline (2.2 mg/L vs. 1.3 mg/L). The changes in FAI were not associated with the presence of coronary plaques, the researchers noted.
The study findings were limited by several factors, including the observational design, small size, and lack of data on cardiovascular endpoints. “Future studies will be needed to explore whether the residual CV risk detected by perivascular FAI can be attenuated using targeted anti-inflammatory interventions,” they wrote.
However, the results suggest that biologics impact coronary vasculature at the microenvironmental level, and that FAI can be a noninvasive, cost-effective way to stratify patients at increased risk for cardiovascular disease, the researchers noted.
“We believe that the strength of perivascular FAI in risk stratifying patients with increased coronary inflammation will allow for better identification of patients at increased risk of future myocardial events that are not captured by traditional CV risk factors,” they wrote.
The study was funded by the National Institutes of Health, several research foundations, Elsevier, Colgate-Palmolive, and Genentech. Dr. Elnabawi had no financial conflicts to disclose; several coauthors reported relationships with multiple companies. One coauthor disclosed a pending and licensed patent to a novel tool for cardiovascular risk stratification based on the CT attenuation of perivascular tissue (OxScore) and a pending and licensed patent to perivascular texture index.
SOURCE: Elnabawi YA et al. JAMA Cardiol. 2019 Jul 31. doi: 10.1001/jamacardio.2019.2589.
Biologics improved coronary inflammation as well as psoriasis symptoms, according to data from the perivascular fat attenuation index in 134 adults identified using coronary CT angiography.
“The perivascular fat attenuation index [FAI] is a [CT]-based, novel, noninvasive imaging technique that allows for direct visualization and quantification of coronary inflammation using differential mapping of attenuation gradients in pericoronary fat,” wrote Youssef A. Elnabawi, MD, of the National Heart, Lung, and Blood Institute and colleagues. Biologics have been associated with reduced noncalcified coronary plaques in psoriasis patients, which suggests possible reduction in coronary inflammation as well.
In a study published in JAMA Cardiology, the researchers analyzed data from 134 adults with moderate to severe psoriasis who received no biologic therapy for at least 3 months before starting the study. Of these, 52 chose not to receive biologics, and served as controls while being treated with topical or light therapies. The participants are part of the Psoriasis Atherosclerosis Cardiometabolic Initiative, an ongoing, prospective cohort study. The average age of the patients was 51 years, and 63% were male.
The 82 patients given biologics received anti–tumor necrosis factor–alpha, anti–interleukin-12/23, or anti-IL-17 for 1 year. Overall, patients on biologics showed a significant decrease in FAI from a median of –71.22 Hounsfield units (HU) at baseline to a median of –76.06 at 1 year. These patients also showed significant improvement in Psoriasis Area and Severity Index scores, from a median of 7.7 at baseline to a median of 3.2 at 1 year. The control patients not on biologics showed no significant changes in FAI, with a median of –71.98 HU at baseline and –72.66 HU at 1 year.
The changes were consistent among the various biologics used, and The median FAI for patients on anti–tumor necrosis factor–alpha changed from –71.25 at baseline to –75.49 at 1 year; median FAI for both IL-12/23 and anti-IL-17 treatment groups changed from –71.18 HU at baseline to –76.92 at 1 year.
In addition, only patients treated with biologics showed a significant reduction in median C-reactive protein levels from baseline (2.2 mg/L vs. 1.3 mg/L). The changes in FAI were not associated with the presence of coronary plaques, the researchers noted.
The study findings were limited by several factors, including the observational design, small size, and lack of data on cardiovascular endpoints. “Future studies will be needed to explore whether the residual CV risk detected by perivascular FAI can be attenuated using targeted anti-inflammatory interventions,” they wrote.
However, the results suggest that biologics impact coronary vasculature at the microenvironmental level, and that FAI can be a noninvasive, cost-effective way to stratify patients at increased risk for cardiovascular disease, the researchers noted.
“We believe that the strength of perivascular FAI in risk stratifying patients with increased coronary inflammation will allow for better identification of patients at increased risk of future myocardial events that are not captured by traditional CV risk factors,” they wrote.
The study was funded by the National Institutes of Health, several research foundations, Elsevier, Colgate-Palmolive, and Genentech. Dr. Elnabawi had no financial conflicts to disclose; several coauthors reported relationships with multiple companies. One coauthor disclosed a pending and licensed patent to a novel tool for cardiovascular risk stratification based on the CT attenuation of perivascular tissue (OxScore) and a pending and licensed patent to perivascular texture index.
SOURCE: Elnabawi YA et al. JAMA Cardiol. 2019 Jul 31. doi: 10.1001/jamacardio.2019.2589.
FROM JAMA CARDIOLOGY
Acupuncture cuts attacks in chronic angina
Adults with chronic stable angina who had acupuncture as an adjunct treatment had fewer angina attacks, compared with controls, based on data from a randomized trial of 398 patients in China.
“Acupuncture has been used as nonpharmacologic treatment for several decades, especially to relieve symptoms of myocardial ischemia, improve cardiac function, and prevent recurrence,” and several small studies have reported benefits in angina patients, wrote Ling Zhao, PhD, of Chengdu (China) University of Traditional Chinese Medicine and colleagues.
In a study published in JAMA Internal Medicine, the researchers randomized patients aged 35-80 years with chronic stable angina into four groups: treatment on the disease-affected meridian (DAM), treatment on the nonaffected meridian (NAM), sham acupuncture (SA), and no acupuncture (wait list, or WL).
Chronic stable angina is defined by the American College of Cardiology and the American Heart Association as angina at least twice a week. Patients with other serious conditions including a history of MI, severe heart failure, valvular heart disease, and poorly controlled blood pressure or diabetes were excluded.
Each treatment group received three acupuncture sessions for 30 minutes each week for 4 weeks. Patients kept diaries of angina attacks and were assessed every 4 weeks for 16 weeks. After 16 weeks, the DAM patients had a significantly greater reduction in angina attacks, compared with the NAM group (4.1 fewer attacks), the SA group (5.2 fewer attacks), and the WL group (5.6 fewer attacks).
Overall, 16 patients reported adverse events related to acupuncture, including 5 cases of subcutaneous hemorrhage at the insertion point, 3 reports of tingling at the insertion point, and 8 reports of sleeplessness during the study period, but no patients discontinued the study because of these events. One patient in the WL group died of an acute MI and received no acupuncture treatment.
“We found that acupuncture on the DAM had superior and clinically relevant benefits in reducing angina frequency and pain intensity to a greater degree than acupuncture on a NAM, SA, or no acupuncture,” the researchers wrote. They found improvements in DAM patients on most metrics, including the Seattle Angina Questionnaire, compared with the other groups.
In addition, “compared with SA and no acupuncture, acupuncture on the DAM resulted in better regulation of anxiety and depression within the 12 weeks after treatment than at the end of the treatment period,” they wrote. “Acupuncture on the DAM causes autonomic remodeling by improving the balance between the vagus nerve and sympathetic nervous system during treatment.”
The findings were limited by several factors including the small sample size, potential performance bias based on variation in the acupuncturists’ experience, lack of analysis of doses of rescue medication, and lack of subgroup analysis, the researchers noted. However, the results are consistent with previous studies and support acupuncture as a potential adjunct treatment for patients with mild to moderate chronic angina.
The researchers had no financial conflicts to disclose. The study was funded by the National Natural Science Foundation of China and the State Key Program for Basic Research of China.
SOURCE: Zhao L et al. JAMA Intern Med. 2019 Jul 29. doi: 10.1001/jamainternmed.2019.2407.
Adults with chronic stable angina who had acupuncture as an adjunct treatment had fewer angina attacks, compared with controls, based on data from a randomized trial of 398 patients in China.
“Acupuncture has been used as nonpharmacologic treatment for several decades, especially to relieve symptoms of myocardial ischemia, improve cardiac function, and prevent recurrence,” and several small studies have reported benefits in angina patients, wrote Ling Zhao, PhD, of Chengdu (China) University of Traditional Chinese Medicine and colleagues.
In a study published in JAMA Internal Medicine, the researchers randomized patients aged 35-80 years with chronic stable angina into four groups: treatment on the disease-affected meridian (DAM), treatment on the nonaffected meridian (NAM), sham acupuncture (SA), and no acupuncture (wait list, or WL).
Chronic stable angina is defined by the American College of Cardiology and the American Heart Association as angina at least twice a week. Patients with other serious conditions including a history of MI, severe heart failure, valvular heart disease, and poorly controlled blood pressure or diabetes were excluded.
Each treatment group received three acupuncture sessions for 30 minutes each week for 4 weeks. Patients kept diaries of angina attacks and were assessed every 4 weeks for 16 weeks. After 16 weeks, the DAM patients had a significantly greater reduction in angina attacks, compared with the NAM group (4.1 fewer attacks), the SA group (5.2 fewer attacks), and the WL group (5.6 fewer attacks).
Overall, 16 patients reported adverse events related to acupuncture, including 5 cases of subcutaneous hemorrhage at the insertion point, 3 reports of tingling at the insertion point, and 8 reports of sleeplessness during the study period, but no patients discontinued the study because of these events. One patient in the WL group died of an acute MI and received no acupuncture treatment.
“We found that acupuncture on the DAM had superior and clinically relevant benefits in reducing angina frequency and pain intensity to a greater degree than acupuncture on a NAM, SA, or no acupuncture,” the researchers wrote. They found improvements in DAM patients on most metrics, including the Seattle Angina Questionnaire, compared with the other groups.
In addition, “compared with SA and no acupuncture, acupuncture on the DAM resulted in better regulation of anxiety and depression within the 12 weeks after treatment than at the end of the treatment period,” they wrote. “Acupuncture on the DAM causes autonomic remodeling by improving the balance between the vagus nerve and sympathetic nervous system during treatment.”
The findings were limited by several factors including the small sample size, potential performance bias based on variation in the acupuncturists’ experience, lack of analysis of doses of rescue medication, and lack of subgroup analysis, the researchers noted. However, the results are consistent with previous studies and support acupuncture as a potential adjunct treatment for patients with mild to moderate chronic angina.
The researchers had no financial conflicts to disclose. The study was funded by the National Natural Science Foundation of China and the State Key Program for Basic Research of China.
SOURCE: Zhao L et al. JAMA Intern Med. 2019 Jul 29. doi: 10.1001/jamainternmed.2019.2407.
Adults with chronic stable angina who had acupuncture as an adjunct treatment had fewer angina attacks, compared with controls, based on data from a randomized trial of 398 patients in China.
“Acupuncture has been used as nonpharmacologic treatment for several decades, especially to relieve symptoms of myocardial ischemia, improve cardiac function, and prevent recurrence,” and several small studies have reported benefits in angina patients, wrote Ling Zhao, PhD, of Chengdu (China) University of Traditional Chinese Medicine and colleagues.
In a study published in JAMA Internal Medicine, the researchers randomized patients aged 35-80 years with chronic stable angina into four groups: treatment on the disease-affected meridian (DAM), treatment on the nonaffected meridian (NAM), sham acupuncture (SA), and no acupuncture (wait list, or WL).
Chronic stable angina is defined by the American College of Cardiology and the American Heart Association as angina at least twice a week. Patients with other serious conditions including a history of MI, severe heart failure, valvular heart disease, and poorly controlled blood pressure or diabetes were excluded.
Each treatment group received three acupuncture sessions for 30 minutes each week for 4 weeks. Patients kept diaries of angina attacks and were assessed every 4 weeks for 16 weeks. After 16 weeks, the DAM patients had a significantly greater reduction in angina attacks, compared with the NAM group (4.1 fewer attacks), the SA group (5.2 fewer attacks), and the WL group (5.6 fewer attacks).
Overall, 16 patients reported adverse events related to acupuncture, including 5 cases of subcutaneous hemorrhage at the insertion point, 3 reports of tingling at the insertion point, and 8 reports of sleeplessness during the study period, but no patients discontinued the study because of these events. One patient in the WL group died of an acute MI and received no acupuncture treatment.
“We found that acupuncture on the DAM had superior and clinically relevant benefits in reducing angina frequency and pain intensity to a greater degree than acupuncture on a NAM, SA, or no acupuncture,” the researchers wrote. They found improvements in DAM patients on most metrics, including the Seattle Angina Questionnaire, compared with the other groups.
In addition, “compared with SA and no acupuncture, acupuncture on the DAM resulted in better regulation of anxiety and depression within the 12 weeks after treatment than at the end of the treatment period,” they wrote. “Acupuncture on the DAM causes autonomic remodeling by improving the balance between the vagus nerve and sympathetic nervous system during treatment.”
The findings were limited by several factors including the small sample size, potential performance bias based on variation in the acupuncturists’ experience, lack of analysis of doses of rescue medication, and lack of subgroup analysis, the researchers noted. However, the results are consistent with previous studies and support acupuncture as a potential adjunct treatment for patients with mild to moderate chronic angina.
The researchers had no financial conflicts to disclose. The study was funded by the National Natural Science Foundation of China and the State Key Program for Basic Research of China.
SOURCE: Zhao L et al. JAMA Intern Med. 2019 Jul 29. doi: 10.1001/jamainternmed.2019.2407.
FROM JAMA INTERNAL MEDICINE
Key clinical point: Adults with chronic stable angina who were treated with acupuncture had significantly fewer attacks than either those treated with sham acupuncture or controls who received no acupuncture.
Major finding: The reduction in angina attacks was 4.07 in disease-affected meridian versus an unaffected meridian, 5.18 versus a sham group, and 5.63 versus untreated controls.
Study details: The data come from a randomized trial of 398 patients at five clinical centers in China.
Disclosures: The researchers had no financial conflicts to disclose. The study was funded by the National Natural Science Foundation of China and the State Key Program for Basic Research of China.
Source: Zhao L et al. JAMA Intern Med. 2019 Jul 29. doi: 10.1001/jamainternmed.2019.2407.
Atherosclerotic disease risk persists decades after smoking cessation
Adults who quit smoking reduced their risk for peripheral artery disease in the short term, but remained at increased risk for up to 30 years, compared with never-smokers, based on data from more than 13,000 adults in a community-based study.

Most reports on the impact of smoking cessation on cardiovascular disease have focused on coronary heart disease (CHD), and stroke, while data on the effects of smoking cessation on peripheral artery disease (PAD) are limited, wrote Ning Ding, MBBS, SCM, of the Johns Hopkins Bloomberg School of Public Health, Baltimore, Md., and colleagues.
To compare the impact of smoking on PAD, CHD, and stroke, the researchers used data from the Atherosclerosis Risk in Communities (ARIC) study, which included 15,792 adults aged 45-64 years in four communities. The findings were published in the Journal of the American College of Cardiology.
The study population of 13,355 individuals had no baseline history of PAD, CHD, or stroke. Over a median 26 years of follow-up, the researchers identified 492 cases of PAD, 1,798 cases of CHD, and 1,106 cases of stroke.
The risk of all three conditions began to decline within 5 years of smoking cessation, which could be encouraging to smokers who wish to quit, the researchers noted. In addition, the longer the duration of smoking cessation, the lower the risk for all three conditions (See central illustration).
However, a significantly elevated risk remained for PAD for up to 30 years after smoking cessation and for CHD for up to 20 years after smoking cessation, compared with never-smokers.
The researchers also found a roughly fourfold increased risk for PAD for smokers who smoked for 40 or more pack-years, compared with never-smokers, which was greater than the 2.1 hazard ratio for CHD and 1.8 HR for stroke. In addition, current smokers of at least one pack per day had a significantly greater risk of PAD, compared with never-smokers (HR, 5.36) that was higher than the risk for CHD or stroke (HR, 2.38 and HR, 1.88, respectively).
The study findings were limited by several factors including the reliance on self-reports, potential misclassification of data, and the potential exclusion of mild PAD cases that did not require hospitalization, the researchers noted. However, the results support the value of encouraging smokers to quit and support the need to include PAD risk in public health information, they said. “Although public statements about smoking and [cardiovascular disease] have been focusing on CHD and stroke, our results indicate the need to take account of PAD as well for comprehensively acknowledging the effect of smoking on overall cardiovascular health,” they added.
The ARIC study was funded by the National Heart, Lung, and Blood Institute, National Institutes of Health. Lead author Dr. Ding had no financial conflicts to disclose; coauthors disclosed relationships with Bristol-Myers Squibb and Fukuda Denshi.
SOURCE: Ding N et al. J Am Coll Cardiol. 2019 Jul 22;74:498-507. doi: 10.1016/j.jacc.2019.06.003.
Although the pathophysiology of smoking and cardiovascular disease has yet to be teased out, the current study findings support the public health message that any and all smokers can improve their health by quitting any time: “It is never too early or too late to benefit from quitting,” wrote Nancy A. Rigotti, MD, and Mary M. McDermott, MD, in an accompanying editorial. The editorialists questioned whether the findings were generalizable to patients with mild PAD or those who are not hospitalized. However, they found the data consistent with previous studies suggesting that atherosclerosis is not homogeneous. “Differences in shear stress and hemodynamic forces among the femoral, coronary, and carotid arterial beds may also explain variability in associations of smoking and smoking cessation with the incidence of PAD versus myocardial infarction or stroke,” they said.
The findings also support the need to emphasize PAD in public health messages and provide an opportunity to educate patients about the risks of limb loss and impaired mobility associated with PAD, they said.
Many clinicians put a low priority on smoking cessation, the editorialists wrote, but “long-term tobacco abstinence is achievable using a chronic disease management approach resembling the strategies used to manage other risk factors,” they said. They cited the American College of Cardiology’s recently released “Expert Consensus Decision Pathway on Tobacco Cessation Treatment.” The pathway outlines advice for clinicians, including how to provide a brief intervention and resources along with advice to quit smoking.
Dr. Rigotti is affiliated with Harvard Medical School, Boston. Dr. McDermott is affiliated with Northwestern University, Chicago. Dr. Rigotti disclosed royalties from UpToDate, serving as a consultant for Achieve Life Sciences, and travel expenses from Pfizer for unpaid consulting. Dr. McDermott disclosed research funding from Regeneron, the National Heart, Lung, and Blood Institute, the National Institute on Aging, and the American Heart Association, plus research support from Chromadex, ReserveAge, Hershey, and ViroMed.
Although the pathophysiology of smoking and cardiovascular disease has yet to be teased out, the current study findings support the public health message that any and all smokers can improve their health by quitting any time: “It is never too early or too late to benefit from quitting,” wrote Nancy A. Rigotti, MD, and Mary M. McDermott, MD, in an accompanying editorial. The editorialists questioned whether the findings were generalizable to patients with mild PAD or those who are not hospitalized. However, they found the data consistent with previous studies suggesting that atherosclerosis is not homogeneous. “Differences in shear stress and hemodynamic forces among the femoral, coronary, and carotid arterial beds may also explain variability in associations of smoking and smoking cessation with the incidence of PAD versus myocardial infarction or stroke,” they said.
The findings also support the need to emphasize PAD in public health messages and provide an opportunity to educate patients about the risks of limb loss and impaired mobility associated with PAD, they said.
Many clinicians put a low priority on smoking cessation, the editorialists wrote, but “long-term tobacco abstinence is achievable using a chronic disease management approach resembling the strategies used to manage other risk factors,” they said. They cited the American College of Cardiology’s recently released “Expert Consensus Decision Pathway on Tobacco Cessation Treatment.” The pathway outlines advice for clinicians, including how to provide a brief intervention and resources along with advice to quit smoking.
Dr. Rigotti is affiliated with Harvard Medical School, Boston. Dr. McDermott is affiliated with Northwestern University, Chicago. Dr. Rigotti disclosed royalties from UpToDate, serving as a consultant for Achieve Life Sciences, and travel expenses from Pfizer for unpaid consulting. Dr. McDermott disclosed research funding from Regeneron, the National Heart, Lung, and Blood Institute, the National Institute on Aging, and the American Heart Association, plus research support from Chromadex, ReserveAge, Hershey, and ViroMed.
Although the pathophysiology of smoking and cardiovascular disease has yet to be teased out, the current study findings support the public health message that any and all smokers can improve their health by quitting any time: “It is never too early or too late to benefit from quitting,” wrote Nancy A. Rigotti, MD, and Mary M. McDermott, MD, in an accompanying editorial. The editorialists questioned whether the findings were generalizable to patients with mild PAD or those who are not hospitalized. However, they found the data consistent with previous studies suggesting that atherosclerosis is not homogeneous. “Differences in shear stress and hemodynamic forces among the femoral, coronary, and carotid arterial beds may also explain variability in associations of smoking and smoking cessation with the incidence of PAD versus myocardial infarction or stroke,” they said.
The findings also support the need to emphasize PAD in public health messages and provide an opportunity to educate patients about the risks of limb loss and impaired mobility associated with PAD, they said.
Many clinicians put a low priority on smoking cessation, the editorialists wrote, but “long-term tobacco abstinence is achievable using a chronic disease management approach resembling the strategies used to manage other risk factors,” they said. They cited the American College of Cardiology’s recently released “Expert Consensus Decision Pathway on Tobacco Cessation Treatment.” The pathway outlines advice for clinicians, including how to provide a brief intervention and resources along with advice to quit smoking.
Dr. Rigotti is affiliated with Harvard Medical School, Boston. Dr. McDermott is affiliated with Northwestern University, Chicago. Dr. Rigotti disclosed royalties from UpToDate, serving as a consultant for Achieve Life Sciences, and travel expenses from Pfizer for unpaid consulting. Dr. McDermott disclosed research funding from Regeneron, the National Heart, Lung, and Blood Institute, the National Institute on Aging, and the American Heart Association, plus research support from Chromadex, ReserveAge, Hershey, and ViroMed.
Adults who quit smoking reduced their risk for peripheral artery disease in the short term, but remained at increased risk for up to 30 years, compared with never-smokers, based on data from more than 13,000 adults in a community-based study.

Most reports on the impact of smoking cessation on cardiovascular disease have focused on coronary heart disease (CHD), and stroke, while data on the effects of smoking cessation on peripheral artery disease (PAD) are limited, wrote Ning Ding, MBBS, SCM, of the Johns Hopkins Bloomberg School of Public Health, Baltimore, Md., and colleagues.
To compare the impact of smoking on PAD, CHD, and stroke, the researchers used data from the Atherosclerosis Risk in Communities (ARIC) study, which included 15,792 adults aged 45-64 years in four communities. The findings were published in the Journal of the American College of Cardiology.
The study population of 13,355 individuals had no baseline history of PAD, CHD, or stroke. Over a median 26 years of follow-up, the researchers identified 492 cases of PAD, 1,798 cases of CHD, and 1,106 cases of stroke.
The risk of all three conditions began to decline within 5 years of smoking cessation, which could be encouraging to smokers who wish to quit, the researchers noted. In addition, the longer the duration of smoking cessation, the lower the risk for all three conditions (See central illustration).
However, a significantly elevated risk remained for PAD for up to 30 years after smoking cessation and for CHD for up to 20 years after smoking cessation, compared with never-smokers.
The researchers also found a roughly fourfold increased risk for PAD for smokers who smoked for 40 or more pack-years, compared with never-smokers, which was greater than the 2.1 hazard ratio for CHD and 1.8 HR for stroke. In addition, current smokers of at least one pack per day had a significantly greater risk of PAD, compared with never-smokers (HR, 5.36) that was higher than the risk for CHD or stroke (HR, 2.38 and HR, 1.88, respectively).
The study findings were limited by several factors including the reliance on self-reports, potential misclassification of data, and the potential exclusion of mild PAD cases that did not require hospitalization, the researchers noted. However, the results support the value of encouraging smokers to quit and support the need to include PAD risk in public health information, they said. “Although public statements about smoking and [cardiovascular disease] have been focusing on CHD and stroke, our results indicate the need to take account of PAD as well for comprehensively acknowledging the effect of smoking on overall cardiovascular health,” they added.
The ARIC study was funded by the National Heart, Lung, and Blood Institute, National Institutes of Health. Lead author Dr. Ding had no financial conflicts to disclose; coauthors disclosed relationships with Bristol-Myers Squibb and Fukuda Denshi.
SOURCE: Ding N et al. J Am Coll Cardiol. 2019 Jul 22;74:498-507. doi: 10.1016/j.jacc.2019.06.003.
Adults who quit smoking reduced their risk for peripheral artery disease in the short term, but remained at increased risk for up to 30 years, compared with never-smokers, based on data from more than 13,000 adults in a community-based study.

Most reports on the impact of smoking cessation on cardiovascular disease have focused on coronary heart disease (CHD), and stroke, while data on the effects of smoking cessation on peripheral artery disease (PAD) are limited, wrote Ning Ding, MBBS, SCM, of the Johns Hopkins Bloomberg School of Public Health, Baltimore, Md., and colleagues.
To compare the impact of smoking on PAD, CHD, and stroke, the researchers used data from the Atherosclerosis Risk in Communities (ARIC) study, which included 15,792 adults aged 45-64 years in four communities. The findings were published in the Journal of the American College of Cardiology.
The study population of 13,355 individuals had no baseline history of PAD, CHD, or stroke. Over a median 26 years of follow-up, the researchers identified 492 cases of PAD, 1,798 cases of CHD, and 1,106 cases of stroke.
The risk of all three conditions began to decline within 5 years of smoking cessation, which could be encouraging to smokers who wish to quit, the researchers noted. In addition, the longer the duration of smoking cessation, the lower the risk for all three conditions (See central illustration).
However, a significantly elevated risk remained for PAD for up to 30 years after smoking cessation and for CHD for up to 20 years after smoking cessation, compared with never-smokers.
The researchers also found a roughly fourfold increased risk for PAD for smokers who smoked for 40 or more pack-years, compared with never-smokers, which was greater than the 2.1 hazard ratio for CHD and 1.8 HR for stroke. In addition, current smokers of at least one pack per day had a significantly greater risk of PAD, compared with never-smokers (HR, 5.36) that was higher than the risk for CHD or stroke (HR, 2.38 and HR, 1.88, respectively).
The study findings were limited by several factors including the reliance on self-reports, potential misclassification of data, and the potential exclusion of mild PAD cases that did not require hospitalization, the researchers noted. However, the results support the value of encouraging smokers to quit and support the need to include PAD risk in public health information, they said. “Although public statements about smoking and [cardiovascular disease] have been focusing on CHD and stroke, our results indicate the need to take account of PAD as well for comprehensively acknowledging the effect of smoking on overall cardiovascular health,” they added.
The ARIC study was funded by the National Heart, Lung, and Blood Institute, National Institutes of Health. Lead author Dr. Ding had no financial conflicts to disclose; coauthors disclosed relationships with Bristol-Myers Squibb and Fukuda Denshi.
SOURCE: Ding N et al. J Am Coll Cardiol. 2019 Jul 22;74:498-507. doi: 10.1016/j.jacc.2019.06.003.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Almost half of sudden cardiac deaths linked to prior silent MI
Almost half of individuals who died of sudden cardiac death (SCD) had a myocardial scar at autopsy, indicating a prior silent myocardial infarction (SMI), in a case-controlled study.
The research team led by Juha H. Vähätalo, MD, from the University of Oulu (Finland), compared autopsy findings, clinical characteristics, and ECG markers associated with SMI in 5,869 people in Northern Finland who had sudden cardiac deaths during 1998-2017.
Overall, 75% of the deaths were caused by coronary artery disease (CAD), and of them, 71% had no previous diagnosis of CAD. Of these latter individuals, 42% had a myocardial scar at autopsy (detected by macroscopic and microscopic evaluation of myocardium), a finding that the authors said indicated a previous, unrecognized MI.
The analysis showed that individuals with SMI were slightly older, at 69.9 years, than were those with no SMI, at 65.5 years, and were more likely to be male (83.4% vs. 75.5%).
The group with prior SMI also died during physical activity at a greater rate than did those without (18.2% vs. 12.4%), the study authors reported in their paper published in JAMA Cardiology.
The research team obtained 438 ECGs prior to SCD; 187 in individuals with SMI and 251 in the group previously diagnosed with CAD.
Of the premortem ECGs in the individuals who had had an SCD after an SMI, 67% were abnormal, the researchers reported.
The SMI group had more frequently inverted T waves (16.6% vs. 8.4%) and pathologic Q waves (12.8% vs. 6.8%), compared with the non-SMI group. Both differences were statistically significant.
Fragmented QRS was the most common marker of a scar in the SMI group, however the authors noted that the fQRS complex was “probably a sensitive marker of myocardial scarring, but its specificity is not very high”.
Overall, having at least one of the following ECG abnormalities – fQRS, Q wave, T-wave inversion, or QRS of at least 110 msec – was more common in the SMI group (66.8%) compared with the non-SMI group (55.4%).
“Among patients in whom SCD without a prior MI is the first sign of cardiac disease, a previous ECG result is likely to be normal. ... ECGs were available only in 187 individuals with SMI, so the data are not sufficient to draw definite conclusions. Rather, they support motivation for further studies on this question,” the study authors noted.
“In the future, other, more efficient methods might be useful for diagnosing SMI, in addition to standard ECGs,” such as cardiac magnetic resonance imaging, but the cost-effectiveness “is likely to be unreasonable. Therefore, screening high-risk populations with ECG to identify individuals for further examinations would probably be reasonable,” they wrote.
The research team noted some limitations of the study such as the autopsy data not revealing the size of the scar detected in the myocardium and not all individuals had an ECG recorded prior to death.
SOURCE: JAMA Cardiol. 2019 Jul 10; doi: 10.1001/jamacardio.2019.2210
Almost half of individuals who died of sudden cardiac death (SCD) had a myocardial scar at autopsy, indicating a prior silent myocardial infarction (SMI), in a case-controlled study.
The research team led by Juha H. Vähätalo, MD, from the University of Oulu (Finland), compared autopsy findings, clinical characteristics, and ECG markers associated with SMI in 5,869 people in Northern Finland who had sudden cardiac deaths during 1998-2017.
Overall, 75% of the deaths were caused by coronary artery disease (CAD), and of them, 71% had no previous diagnosis of CAD. Of these latter individuals, 42% had a myocardial scar at autopsy (detected by macroscopic and microscopic evaluation of myocardium), a finding that the authors said indicated a previous, unrecognized MI.
The analysis showed that individuals with SMI were slightly older, at 69.9 years, than were those with no SMI, at 65.5 years, and were more likely to be male (83.4% vs. 75.5%).
The group with prior SMI also died during physical activity at a greater rate than did those without (18.2% vs. 12.4%), the study authors reported in their paper published in JAMA Cardiology.
The research team obtained 438 ECGs prior to SCD; 187 in individuals with SMI and 251 in the group previously diagnosed with CAD.
Of the premortem ECGs in the individuals who had had an SCD after an SMI, 67% were abnormal, the researchers reported.
The SMI group had more frequently inverted T waves (16.6% vs. 8.4%) and pathologic Q waves (12.8% vs. 6.8%), compared with the non-SMI group. Both differences were statistically significant.
Fragmented QRS was the most common marker of a scar in the SMI group, however the authors noted that the fQRS complex was “probably a sensitive marker of myocardial scarring, but its specificity is not very high”.
Overall, having at least one of the following ECG abnormalities – fQRS, Q wave, T-wave inversion, or QRS of at least 110 msec – was more common in the SMI group (66.8%) compared with the non-SMI group (55.4%).
“Among patients in whom SCD without a prior MI is the first sign of cardiac disease, a previous ECG result is likely to be normal. ... ECGs were available only in 187 individuals with SMI, so the data are not sufficient to draw definite conclusions. Rather, they support motivation for further studies on this question,” the study authors noted.
“In the future, other, more efficient methods might be useful for diagnosing SMI, in addition to standard ECGs,” such as cardiac magnetic resonance imaging, but the cost-effectiveness “is likely to be unreasonable. Therefore, screening high-risk populations with ECG to identify individuals for further examinations would probably be reasonable,” they wrote.
The research team noted some limitations of the study such as the autopsy data not revealing the size of the scar detected in the myocardium and not all individuals had an ECG recorded prior to death.
SOURCE: JAMA Cardiol. 2019 Jul 10; doi: 10.1001/jamacardio.2019.2210
Almost half of individuals who died of sudden cardiac death (SCD) had a myocardial scar at autopsy, indicating a prior silent myocardial infarction (SMI), in a case-controlled study.
The research team led by Juha H. Vähätalo, MD, from the University of Oulu (Finland), compared autopsy findings, clinical characteristics, and ECG markers associated with SMI in 5,869 people in Northern Finland who had sudden cardiac deaths during 1998-2017.
Overall, 75% of the deaths were caused by coronary artery disease (CAD), and of them, 71% had no previous diagnosis of CAD. Of these latter individuals, 42% had a myocardial scar at autopsy (detected by macroscopic and microscopic evaluation of myocardium), a finding that the authors said indicated a previous, unrecognized MI.
The analysis showed that individuals with SMI were slightly older, at 69.9 years, than were those with no SMI, at 65.5 years, and were more likely to be male (83.4% vs. 75.5%).
The group with prior SMI also died during physical activity at a greater rate than did those without (18.2% vs. 12.4%), the study authors reported in their paper published in JAMA Cardiology.
The research team obtained 438 ECGs prior to SCD; 187 in individuals with SMI and 251 in the group previously diagnosed with CAD.
Of the premortem ECGs in the individuals who had had an SCD after an SMI, 67% were abnormal, the researchers reported.
The SMI group had more frequently inverted T waves (16.6% vs. 8.4%) and pathologic Q waves (12.8% vs. 6.8%), compared with the non-SMI group. Both differences were statistically significant.
Fragmented QRS was the most common marker of a scar in the SMI group, however the authors noted that the fQRS complex was “probably a sensitive marker of myocardial scarring, but its specificity is not very high”.
Overall, having at least one of the following ECG abnormalities – fQRS, Q wave, T-wave inversion, or QRS of at least 110 msec – was more common in the SMI group (66.8%) compared with the non-SMI group (55.4%).
“Among patients in whom SCD without a prior MI is the first sign of cardiac disease, a previous ECG result is likely to be normal. ... ECGs were available only in 187 individuals with SMI, so the data are not sufficient to draw definite conclusions. Rather, they support motivation for further studies on this question,” the study authors noted.
“In the future, other, more efficient methods might be useful for diagnosing SMI, in addition to standard ECGs,” such as cardiac magnetic resonance imaging, but the cost-effectiveness “is likely to be unreasonable. Therefore, screening high-risk populations with ECG to identify individuals for further examinations would probably be reasonable,” they wrote.
The research team noted some limitations of the study such as the autopsy data not revealing the size of the scar detected in the myocardium and not all individuals had an ECG recorded prior to death.
SOURCE: JAMA Cardiol. 2019 Jul 10; doi: 10.1001/jamacardio.2019.2210
FROM JAMA CARDIOLOGY
Could some elderly skip statins based on negative risk markers?
Older individuals with certain negative risk markers have a very low risk of atherosclerotic cardiovascular disease, raising the possibility that some could forgo preventive treatment even if it’s indicated by current standards of risk assessment.
Low levels of coronary artery calcification (CAC), low galectin-3 levels, and absence of carotid plaque were all linked to a lower likelihood of disease than might be expected based on traditional risk assessment, according to the authors of analysis of a large, contemporary cohort of elderly individuals published in the Journal of the American College of Cardiology.
“Our results hold the potential to markedly improve statin allocation in elderly individuals by de-escalating or even withholding preventive therapy in elderly individuals at truly low atherosclerotic cardiovascular disease risk despite advancing age,“ wrote Martin Bødtker Mortensen, MD, PhD, of Aarhus (Denmark) University Hospital.
Most elderly individuals now qualify for lifelong preventive statin treatment, based on the broader indication for treatment in recent guidelines, and the substantial impact that age has when risk is being calculated, Dr. Mortensen and coauthors said in their report.
“Because frailty, comorbidity, and polypharmacy are increasing concerns in elderly individuals and have been proposed to increase the risk for adverse effects, the appropriateness of treating almost all elderly individuals is questionable,” they said in the report.
In their study, Dr. Mortensen and colleagues evaluated a set of 13 biomarkers or imaging tests that they though had potential to “downgrade” risk of coronary heart disease (CHD) and cardiovascular disease (CVD). They based their analysis on 5,805 patients in the BioImage Study, a prospective cohort study of elderly men and women with no atherosclerotic cardiovascular disease at the time of enrollment in 2008 and 2009. The mean age at the time of enrollment was 69 years, and the mean follow-up in this analysis was 2.7 years.
The overall rate of CHD was 6.1 per 1,000 person-years, though looking at negative risk markers, the event rate was just 0.9 for individuals with CAC of 0 and also 0.9 for those with a CAC of 10 or less, followed by 1.7 for absence of carotid plaque, and 2.6 for galectin-3 in the bottom 25th percentile, according to Dr. Mortensen and coinvestigators. Similarly, the rate of CVD was 9.2 per 1,000 person-years overall, and just 3.2 for a CAC of 0, 2.8 for a CAC of 10 or lower, 4.4 for no carotid plaque, and 4.0 for low galectin-3.
Results were less impressive for other negative risk markers, including normal ankle-brachial index (ABI) test, lack of family history, and low levels of circulating biomarkers such as high-sensitivity C-reactive protein and lipoprotein (a).
Investigators also calculated diagnostic likelihood ratios (DLR), a measure they said assesses the value of performing a diagnostic test, with values lower than 1 indicating a specific marker has value for downgrading risk.
Zero or low CAC exerted the greatest downward change in pre- to post-test risk, according to the investigators, with a multivariable-adjusted DLR of 0.20 for CHD, translating into an 80% relative risk reduction. Similarly, the adjusted DLRs for zero or low CAC for CVD were 0.48 and 0.41, respectively, translating into a 59% risk reduction.
Low galectin-3 also resulted in significant downward change in that pre- to post-test risk, investigators added.
The BioImage Study was funded by Abbott, AstraZeneca, Merck, Philips, and Takeda. Dr. Mortensen had no disclosures related to the present analysis. Coauthors provided disclosures related to G3 Pharmaceuticals, Abbott Laboratories, AstraZeneca, Bayer, Bristol-Myers Squibb, CSL Behring, Eli Lilly/DSI, Medtronic, Novartis Pharmaceuticals, OrbusNeich, and PLC/Renal Guard, among others.
SOURCE: Mortensen MB et al. J Am Coll Cardiol. 2019 Jul 1. doi: 10.1016/j.jacc.2019.04.049.
This study suggests that atherosclerosis imaging tests are the strongest negative risk factors identified to date in cardiovascular medicine. The results are clinically actionable and should shape our approach to these tests in clinical practice.
Atherosclerosis imaging tests could be important in preventing overuse of pharmacotherapy for primary prevention in older adults, nearly all of whom would be considered at elevated risk by current standards.
Results for other candidate markers in the study were generally unimpressive, though the finding that low galectin-3 levels predicted low cardiovascular risk is novel, highly interesting, and deserving of further study.
Meanwhile, guidelines are already taking notice of an emerging consensus on the value of imaging studies as a negative risk factor.
The 2018 prevention guidelines from the American College of Cardiology/American Heart Association recommend coronary artery calcium (CAC) testing to guide individualized patient decision-making in certain adults between the ages of 40 and 75 years who are at borderline to intermediate risk, and that using CAC results to reclassify risk is reasonable in those aged 76-80 years, they said.
For the first time, the ACC/AHA guidelines devoted a section to negative risk factors, specifically highlighting the value of CAC = 0 and stating that intensive statin therapy is of less value in such patients and can potentially be avoided.
Likewise, 2017 guidelines from the Society of Cardiovascular Computed Tomography say that aspirin for primary prevention can almost always be forgone in patients with a CAC of 0.
“The bar for preventive therapy has justifiably fallen, reaching a point where many patients will qualify based on their age alone,” the authors said.
In light of that development, negative risk factors might meaningfully downgrade risk and help identify individuals who can safely focus on lifestyle therapies and defer preventive medication.
Michael J. Blaha, MD, MPH, and Khurram Nasir, MD, MPH are with the Johns Hopkins Ciccarone Center for the Prevention of Cardiovascular Disease in Baltimore; Dr. Nasir also is affiliated with the Center for Outcomes Research and Evaluation, Yale University, New Haven, Conn. Ron Blankstein, MD, is with the cardiovascular division, department of medicine, Brigham and Women’s Hospital, Boston. These comments are adapted from their editorial (J Am Coll Cardiol. 2019 Jul 1. doi: 10.1016/j.jacc.2019.05.032 ). Dr. Blankstein reported research funding/grant support from Amgen and Astellas; Dr. Blaha and Dr. Nasir said they had no disclosures to report.
This study suggests that atherosclerosis imaging tests are the strongest negative risk factors identified to date in cardiovascular medicine. The results are clinically actionable and should shape our approach to these tests in clinical practice.
Atherosclerosis imaging tests could be important in preventing overuse of pharmacotherapy for primary prevention in older adults, nearly all of whom would be considered at elevated risk by current standards.
Results for other candidate markers in the study were generally unimpressive, though the finding that low galectin-3 levels predicted low cardiovascular risk is novel, highly interesting, and deserving of further study.
Meanwhile, guidelines are already taking notice of an emerging consensus on the value of imaging studies as a negative risk factor.
The 2018 prevention guidelines from the American College of Cardiology/American Heart Association recommend coronary artery calcium (CAC) testing to guide individualized patient decision-making in certain adults between the ages of 40 and 75 years who are at borderline to intermediate risk, and that using CAC results to reclassify risk is reasonable in those aged 76-80 years, they said.
For the first time, the ACC/AHA guidelines devoted a section to negative risk factors, specifically highlighting the value of CAC = 0 and stating that intensive statin therapy is of less value in such patients and can potentially be avoided.
Likewise, 2017 guidelines from the Society of Cardiovascular Computed Tomography say that aspirin for primary prevention can almost always be forgone in patients with a CAC of 0.
“The bar for preventive therapy has justifiably fallen, reaching a point where many patients will qualify based on their age alone,” the authors said.
In light of that development, negative risk factors might meaningfully downgrade risk and help identify individuals who can safely focus on lifestyle therapies and defer preventive medication.
Michael J. Blaha, MD, MPH, and Khurram Nasir, MD, MPH are with the Johns Hopkins Ciccarone Center for the Prevention of Cardiovascular Disease in Baltimore; Dr. Nasir also is affiliated with the Center for Outcomes Research and Evaluation, Yale University, New Haven, Conn. Ron Blankstein, MD, is with the cardiovascular division, department of medicine, Brigham and Women’s Hospital, Boston. These comments are adapted from their editorial (J Am Coll Cardiol. 2019 Jul 1. doi: 10.1016/j.jacc.2019.05.032 ). Dr. Blankstein reported research funding/grant support from Amgen and Astellas; Dr. Blaha and Dr. Nasir said they had no disclosures to report.
This study suggests that atherosclerosis imaging tests are the strongest negative risk factors identified to date in cardiovascular medicine. The results are clinically actionable and should shape our approach to these tests in clinical practice.
Atherosclerosis imaging tests could be important in preventing overuse of pharmacotherapy for primary prevention in older adults, nearly all of whom would be considered at elevated risk by current standards.
Results for other candidate markers in the study were generally unimpressive, though the finding that low galectin-3 levels predicted low cardiovascular risk is novel, highly interesting, and deserving of further study.
Meanwhile, guidelines are already taking notice of an emerging consensus on the value of imaging studies as a negative risk factor.
The 2018 prevention guidelines from the American College of Cardiology/American Heart Association recommend coronary artery calcium (CAC) testing to guide individualized patient decision-making in certain adults between the ages of 40 and 75 years who are at borderline to intermediate risk, and that using CAC results to reclassify risk is reasonable in those aged 76-80 years, they said.
For the first time, the ACC/AHA guidelines devoted a section to negative risk factors, specifically highlighting the value of CAC = 0 and stating that intensive statin therapy is of less value in such patients and can potentially be avoided.
Likewise, 2017 guidelines from the Society of Cardiovascular Computed Tomography say that aspirin for primary prevention can almost always be forgone in patients with a CAC of 0.
“The bar for preventive therapy has justifiably fallen, reaching a point where many patients will qualify based on their age alone,” the authors said.
In light of that development, negative risk factors might meaningfully downgrade risk and help identify individuals who can safely focus on lifestyle therapies and defer preventive medication.
Michael J. Blaha, MD, MPH, and Khurram Nasir, MD, MPH are with the Johns Hopkins Ciccarone Center for the Prevention of Cardiovascular Disease in Baltimore; Dr. Nasir also is affiliated with the Center for Outcomes Research and Evaluation, Yale University, New Haven, Conn. Ron Blankstein, MD, is with the cardiovascular division, department of medicine, Brigham and Women’s Hospital, Boston. These comments are adapted from their editorial (J Am Coll Cardiol. 2019 Jul 1. doi: 10.1016/j.jacc.2019.05.032 ). Dr. Blankstein reported research funding/grant support from Amgen and Astellas; Dr. Blaha and Dr. Nasir said they had no disclosures to report.
Older individuals with certain negative risk markers have a very low risk of atherosclerotic cardiovascular disease, raising the possibility that some could forgo preventive treatment even if it’s indicated by current standards of risk assessment.
Low levels of coronary artery calcification (CAC), low galectin-3 levels, and absence of carotid plaque were all linked to a lower likelihood of disease than might be expected based on traditional risk assessment, according to the authors of analysis of a large, contemporary cohort of elderly individuals published in the Journal of the American College of Cardiology.
“Our results hold the potential to markedly improve statin allocation in elderly individuals by de-escalating or even withholding preventive therapy in elderly individuals at truly low atherosclerotic cardiovascular disease risk despite advancing age,“ wrote Martin Bødtker Mortensen, MD, PhD, of Aarhus (Denmark) University Hospital.
Most elderly individuals now qualify for lifelong preventive statin treatment, based on the broader indication for treatment in recent guidelines, and the substantial impact that age has when risk is being calculated, Dr. Mortensen and coauthors said in their report.
“Because frailty, comorbidity, and polypharmacy are increasing concerns in elderly individuals and have been proposed to increase the risk for adverse effects, the appropriateness of treating almost all elderly individuals is questionable,” they said in the report.
In their study, Dr. Mortensen and colleagues evaluated a set of 13 biomarkers or imaging tests that they though had potential to “downgrade” risk of coronary heart disease (CHD) and cardiovascular disease (CVD). They based their analysis on 5,805 patients in the BioImage Study, a prospective cohort study of elderly men and women with no atherosclerotic cardiovascular disease at the time of enrollment in 2008 and 2009. The mean age at the time of enrollment was 69 years, and the mean follow-up in this analysis was 2.7 years.
The overall rate of CHD was 6.1 per 1,000 person-years, though looking at negative risk markers, the event rate was just 0.9 for individuals with CAC of 0 and also 0.9 for those with a CAC of 10 or less, followed by 1.7 for absence of carotid plaque, and 2.6 for galectin-3 in the bottom 25th percentile, according to Dr. Mortensen and coinvestigators. Similarly, the rate of CVD was 9.2 per 1,000 person-years overall, and just 3.2 for a CAC of 0, 2.8 for a CAC of 10 or lower, 4.4 for no carotid plaque, and 4.0 for low galectin-3.
Results were less impressive for other negative risk markers, including normal ankle-brachial index (ABI) test, lack of family history, and low levels of circulating biomarkers such as high-sensitivity C-reactive protein and lipoprotein (a).
Investigators also calculated diagnostic likelihood ratios (DLR), a measure they said assesses the value of performing a diagnostic test, with values lower than 1 indicating a specific marker has value for downgrading risk.
Zero or low CAC exerted the greatest downward change in pre- to post-test risk, according to the investigators, with a multivariable-adjusted DLR of 0.20 for CHD, translating into an 80% relative risk reduction. Similarly, the adjusted DLRs for zero or low CAC for CVD were 0.48 and 0.41, respectively, translating into a 59% risk reduction.
Low galectin-3 also resulted in significant downward change in that pre- to post-test risk, investigators added.
The BioImage Study was funded by Abbott, AstraZeneca, Merck, Philips, and Takeda. Dr. Mortensen had no disclosures related to the present analysis. Coauthors provided disclosures related to G3 Pharmaceuticals, Abbott Laboratories, AstraZeneca, Bayer, Bristol-Myers Squibb, CSL Behring, Eli Lilly/DSI, Medtronic, Novartis Pharmaceuticals, OrbusNeich, and PLC/Renal Guard, among others.
SOURCE: Mortensen MB et al. J Am Coll Cardiol. 2019 Jul 1. doi: 10.1016/j.jacc.2019.04.049.
Older individuals with certain negative risk markers have a very low risk of atherosclerotic cardiovascular disease, raising the possibility that some could forgo preventive treatment even if it’s indicated by current standards of risk assessment.
Low levels of coronary artery calcification (CAC), low galectin-3 levels, and absence of carotid plaque were all linked to a lower likelihood of disease than might be expected based on traditional risk assessment, according to the authors of analysis of a large, contemporary cohort of elderly individuals published in the Journal of the American College of Cardiology.
“Our results hold the potential to markedly improve statin allocation in elderly individuals by de-escalating or even withholding preventive therapy in elderly individuals at truly low atherosclerotic cardiovascular disease risk despite advancing age,“ wrote Martin Bødtker Mortensen, MD, PhD, of Aarhus (Denmark) University Hospital.
Most elderly individuals now qualify for lifelong preventive statin treatment, based on the broader indication for treatment in recent guidelines, and the substantial impact that age has when risk is being calculated, Dr. Mortensen and coauthors said in their report.
“Because frailty, comorbidity, and polypharmacy are increasing concerns in elderly individuals and have been proposed to increase the risk for adverse effects, the appropriateness of treating almost all elderly individuals is questionable,” they said in the report.
In their study, Dr. Mortensen and colleagues evaluated a set of 13 biomarkers or imaging tests that they though had potential to “downgrade” risk of coronary heart disease (CHD) and cardiovascular disease (CVD). They based their analysis on 5,805 patients in the BioImage Study, a prospective cohort study of elderly men and women with no atherosclerotic cardiovascular disease at the time of enrollment in 2008 and 2009. The mean age at the time of enrollment was 69 years, and the mean follow-up in this analysis was 2.7 years.
The overall rate of CHD was 6.1 per 1,000 person-years, though looking at negative risk markers, the event rate was just 0.9 for individuals with CAC of 0 and also 0.9 for those with a CAC of 10 or less, followed by 1.7 for absence of carotid plaque, and 2.6 for galectin-3 in the bottom 25th percentile, according to Dr. Mortensen and coinvestigators. Similarly, the rate of CVD was 9.2 per 1,000 person-years overall, and just 3.2 for a CAC of 0, 2.8 for a CAC of 10 or lower, 4.4 for no carotid plaque, and 4.0 for low galectin-3.
Results were less impressive for other negative risk markers, including normal ankle-brachial index (ABI) test, lack of family history, and low levels of circulating biomarkers such as high-sensitivity C-reactive protein and lipoprotein (a).
Investigators also calculated diagnostic likelihood ratios (DLR), a measure they said assesses the value of performing a diagnostic test, with values lower than 1 indicating a specific marker has value for downgrading risk.
Zero or low CAC exerted the greatest downward change in pre- to post-test risk, according to the investigators, with a multivariable-adjusted DLR of 0.20 for CHD, translating into an 80% relative risk reduction. Similarly, the adjusted DLRs for zero or low CAC for CVD were 0.48 and 0.41, respectively, translating into a 59% risk reduction.
Low galectin-3 also resulted in significant downward change in that pre- to post-test risk, investigators added.
The BioImage Study was funded by Abbott, AstraZeneca, Merck, Philips, and Takeda. Dr. Mortensen had no disclosures related to the present analysis. Coauthors provided disclosures related to G3 Pharmaceuticals, Abbott Laboratories, AstraZeneca, Bayer, Bristol-Myers Squibb, CSL Behring, Eli Lilly/DSI, Medtronic, Novartis Pharmaceuticals, OrbusNeich, and PLC/Renal Guard, among others.
SOURCE: Mortensen MB et al. J Am Coll Cardiol. 2019 Jul 1. doi: 10.1016/j.jacc.2019.04.049.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Key clinical point: Low or no coronary artery calcification (CAC), low galectin-3 levels, and absence of carotid plaque were all linked to a lower likelihood of atherosclerotic cardiovascular disease in elderly patients, suggesting pharmacotherapy might be avoidable in a certain proportion of them.
Major finding: The diagnostic likelihood ratios (DLR) were low for those negative risk markers, e.g., 0.20 for no or low CAC, indicating a relative risk reduction of about 80%.
Study details: Analysis of data from the BioImage Study, a prospective cohort study of 5,805 older men and women who had no atherosclerotic cardiovascular disease at the time of enrollment.
Disclosures: The BioImage Study was funded by Abbott, AstraZeneca, Merck, Philips, and Takeda. Authors of the present analysis provided disclosures related to G3 Pharmaceuticals, Abbott Laboratories, AstraZeneca, Bayer, Bristol-Myers Squibb, CSL Behring, Eli Lilly/DSI, Medtronic, Novartis Pharmaceuticals, OrbusNeich, and PLC/Renal Guard, among others.
Source: Mortensen MB et al. J Am Coll Cardiol. 2019 Jul 1. doi: j.jacc.2019.04.049.
MRI-guided revascularization noninferior to FFR
Myocardial perfusion cardiovascular MRI is as good as invasive angiography and measurement of fractional flow reserve to guide revascularization in patients with angina, research suggests.
In the New England Journal of Medicine, researchers present the outcomes of an unblinded, multicenter, clinical effectiveness trial comparing the two revascularization strategies in 918 patients who had typical angina and either two or more cardiovascular risk factors or a positive exercise treadmill test.
In the fractional flow reserve method, revascularization was recommended in all vessels with an FFR of 0.8 or less. In the MRI-guided method, all patients underwent myocardial perfusion cardiovascular MRI, and patients with clinically significant inducible ischemia then underwent invasive angiography, and revascularization if required.
Significantly fewer patients in the cardiovascular MRI group underwent index revascularization, compared with the fractional flow reserve group (36% vs. 45% respectively; P = .005), and only 48% in the cardiovascular MRI group underwent invasive angiography, compared with 97% of patients in the fractional flow reserve arm.
However, there was no significant difference between the two groups in the incidence of major cardiac adverse events after 1 year, signifying that the MRI approach met the criteria for noninferiority.
There was also no significant difference between the two groups in the percentage of patients who were free from angina after 12 months (49.2% in the MRI group and 43.8% in the FFR group).
“Current guidelines on the management of the care of patients with suspected coronary artery disease separate diagnostic strategies from therapeutic strategies owing to a lack of evidence comparing combined diagnostic and therapeutic pathways,” wrote Eike Nagel, MD, of the Goethe University Frankfurt Institute for Experimental and Translational Cardiovascular Imaging and coauthors. “The MR-INFORM trial closes this knowledge gap by comparing two frequently used, well-defined, standardized, and validated clinical management strategies.”
However, they pointed out that one limitation of their study was the lack of a third group of patients who received medical therapy without planned revascularization. They also noted that the incidence of the primary outcome of major adverse cardiac events was lower than expected at 1 year.
The study was supported by the Guy’s and St. Thomas’ Biomedical Research Centre of the National Institute for Health Research. Three authors declared support from study supporters related to the study, three declared grants, personal fees, and other support from the private sector unrelated to the study. No other conflicts of interest were declared.
SOURCE: Nagel E et al. New Engl J Med. 2019;380:2418-28. doi: 10.1056/NEJMoa1716734.
Myocardial perfusion cardiovascular MRI is as good as invasive angiography and measurement of fractional flow reserve to guide revascularization in patients with angina, research suggests.
In the New England Journal of Medicine, researchers present the outcomes of an unblinded, multicenter, clinical effectiveness trial comparing the two revascularization strategies in 918 patients who had typical angina and either two or more cardiovascular risk factors or a positive exercise treadmill test.
In the fractional flow reserve method, revascularization was recommended in all vessels with an FFR of 0.8 or less. In the MRI-guided method, all patients underwent myocardial perfusion cardiovascular MRI, and patients with clinically significant inducible ischemia then underwent invasive angiography, and revascularization if required.
Significantly fewer patients in the cardiovascular MRI group underwent index revascularization, compared with the fractional flow reserve group (36% vs. 45% respectively; P = .005), and only 48% in the cardiovascular MRI group underwent invasive angiography, compared with 97% of patients in the fractional flow reserve arm.
However, there was no significant difference between the two groups in the incidence of major cardiac adverse events after 1 year, signifying that the MRI approach met the criteria for noninferiority.
There was also no significant difference between the two groups in the percentage of patients who were free from angina after 12 months (49.2% in the MRI group and 43.8% in the FFR group).
“Current guidelines on the management of the care of patients with suspected coronary artery disease separate diagnostic strategies from therapeutic strategies owing to a lack of evidence comparing combined diagnostic and therapeutic pathways,” wrote Eike Nagel, MD, of the Goethe University Frankfurt Institute for Experimental and Translational Cardiovascular Imaging and coauthors. “The MR-INFORM trial closes this knowledge gap by comparing two frequently used, well-defined, standardized, and validated clinical management strategies.”
However, they pointed out that one limitation of their study was the lack of a third group of patients who received medical therapy without planned revascularization. They also noted that the incidence of the primary outcome of major adverse cardiac events was lower than expected at 1 year.
The study was supported by the Guy’s and St. Thomas’ Biomedical Research Centre of the National Institute for Health Research. Three authors declared support from study supporters related to the study, three declared grants, personal fees, and other support from the private sector unrelated to the study. No other conflicts of interest were declared.
SOURCE: Nagel E et al. New Engl J Med. 2019;380:2418-28. doi: 10.1056/NEJMoa1716734.
Myocardial perfusion cardiovascular MRI is as good as invasive angiography and measurement of fractional flow reserve to guide revascularization in patients with angina, research suggests.
In the New England Journal of Medicine, researchers present the outcomes of an unblinded, multicenter, clinical effectiveness trial comparing the two revascularization strategies in 918 patients who had typical angina and either two or more cardiovascular risk factors or a positive exercise treadmill test.
In the fractional flow reserve method, revascularization was recommended in all vessels with an FFR of 0.8 or less. In the MRI-guided method, all patients underwent myocardial perfusion cardiovascular MRI, and patients with clinically significant inducible ischemia then underwent invasive angiography, and revascularization if required.
Significantly fewer patients in the cardiovascular MRI group underwent index revascularization, compared with the fractional flow reserve group (36% vs. 45% respectively; P = .005), and only 48% in the cardiovascular MRI group underwent invasive angiography, compared with 97% of patients in the fractional flow reserve arm.
However, there was no significant difference between the two groups in the incidence of major cardiac adverse events after 1 year, signifying that the MRI approach met the criteria for noninferiority.
There was also no significant difference between the two groups in the percentage of patients who were free from angina after 12 months (49.2% in the MRI group and 43.8% in the FFR group).
“Current guidelines on the management of the care of patients with suspected coronary artery disease separate diagnostic strategies from therapeutic strategies owing to a lack of evidence comparing combined diagnostic and therapeutic pathways,” wrote Eike Nagel, MD, of the Goethe University Frankfurt Institute for Experimental and Translational Cardiovascular Imaging and coauthors. “The MR-INFORM trial closes this knowledge gap by comparing two frequently used, well-defined, standardized, and validated clinical management strategies.”
However, they pointed out that one limitation of their study was the lack of a third group of patients who received medical therapy without planned revascularization. They also noted that the incidence of the primary outcome of major adverse cardiac events was lower than expected at 1 year.
The study was supported by the Guy’s and St. Thomas’ Biomedical Research Centre of the National Institute for Health Research. Three authors declared support from study supporters related to the study, three declared grants, personal fees, and other support from the private sector unrelated to the study. No other conflicts of interest were declared.
SOURCE: Nagel E et al. New Engl J Med. 2019;380:2418-28. doi: 10.1056/NEJMoa1716734.
FROM NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: .
Major finding: The incidence of major cardiac adverse events similar at 1 year with cardiovascular MRI and invasive angiography.
Study details: MR-INFORM, an unblinded, multicenter, clinical effectiveness trial in 918 patients with angina.
Disclosures: The study was supported by the Guy’s and St. Thomas’ Biomedical Research Centre of the National Institute for Health Research. Three authors declared support from study supporters related to the study, three declared grants, personal fees and other support from the private sector unrelated to the study. No other conflicts of interest were declared.
Source: Nagel E et al. New Engl J Med. 2019;380:2418-28. doi: 10.1056/NEJMoa1716734.
Aggressive lowering of LDL cholesterol: Is it a good idea?
SAN FRANCISCO – Powerful drugs now make it possible to lower LDL cholesterol levels to dramatically low levels. But is this a good idea? There are risks, and a cardiologist urged diabetes professionals to not overdo cholesterol reduction. But a colleague argued in favor of aggressively targeting “bad” cholesterol.
“We used to say you can’t be too rich or too thin. We now say you can’t be too rich or too thin or have a too-low LDL cholesterol,” said cardiologist Steven E. Nissen, MD, chairman of cardiovascular medicine at the Cleveland Clinic Foundation, who spoke at the annual scientific sessions of the American Diabetes Association about the wisdom of extreme LDL cholesterol lowering.
Dr. Nissen faced off in a debate with cardiologist Sanket Dhruva, MD, of the University of California, San Francisco, who doesn’t support aggressive LDL cholesterol lowering.
It is fine, Dr. Dhruva said, to treat patients so their LDL cholesterol levels drop below 100 mg/dL. “I don’t think there’s any argument there.”
But Dr. Dhruva questioned whether it’s a good idea to generally decrease LDL cholesterol well below 70 mg/dL, as is now possible with the use of proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors.
He pointed to a 2010 study that found aggressively lowering LDL cholesterol led to a mean net gain of 4.1 quality-adjusted life-years in high-risk patients, but less than 1 quality-adjusted life-year in low-risk patients. According to him, the study also found that the biggest benefits in both high- and low-risk patients came from the initial lower statin dose (Arch Intern Med. 2010 Jun 28;170[12]:1037-44).
“It’s really the statin initiation that provides the most benefit to our patients with diabetes,” Dr. Dhruva said.
Also, he added, a 2016 study questioned the value of aggressively lowering LDL cholesterol. It found that, although patients on statins with LDL cholesterol levels of 70-100 mg/dL had a lower risk of adverse cardiac outcomes than did those with levels between 100 and 130 mg/dL, no additional benefit was gained by achieving an LDL cholesterol level below 70 mg/dL (JAMA Intern Med. 2016 Aug 1;176[8]:1105-13)
As for risks, Dr. Dhruva highlighted a 2016 pooled analysis of 14 trials that linked the PCSK9 inhibitor alirocumab (Praluent) and LDL cholesterol levels below 25 mg/dL to significantly higher levels of cataracts, compared with levels of at least 25 mg/dL (hazard ratio, 3.4).
There are other reasons to be cautious of aggressive LDL cholesterol lowering. For one, many patients are not on statins when they’re prescribed PCSK-9 inhibitors. “We’re sometimes missing the building blocks before getting to expensive medications,” he said.
He added that PCSK-9 inhibitors are pricey, and some patients can’t get access to them. “Lipid control is incredibly important, but what about the stress or anxiety of our patients who are told this medication will reduce their cardiac risk but they can’t afford it? That’s not good for their cardiovascular risk.”
For his part, Dr. Nissen challenged Dr. Dhruva’s concerns about the cost of the drugs. “It’s not like they’re way out of line in terms of expense,” he said, noting that their cost – several thousand dollars a year – is similar to the cost of diabetes drugs known as glucagonlike peptide–1 receptor agonists and sodium-glucose transporter 2 inhibitors.
According to Dr. Nissen, multiple studies have supported aggressive LDL cholesterol lowering. “You’re going to see this over and over again in clinical trials: Every time we lower LDL by more, we get more reductions in morbidity and mortality.”
For example, he said, the FOURIER trial of the PCSK9 inhibitor evolocumab (Repatha) found that it lowered LDL cholesterol levels to a median 30 mg/dL “and reduced the risk of cardiovascular events. These findings show that patients with atherosclerotic cardiovascular disease benefit from lowering of LDL cholesterol levels below current targets [N Engl J Med 2017;376:1713-22].”
Dr. Nissen pointed to another study, this one also from 2017, that reported “in individuals with 5-year risk of major vascular events lower than 10%, each 1 mmol/L reduction in LDL cholesterol produced an absolute reduction in major vascular events of about 11 per 1,000 over 5 years. This benefit greatly exceeds any known hazards of statin therapy.”
In regard to adverse effects, he said, research has hinted at a slight uptick in blood sugar levels “that does not take away the major cardiovascular benefits of the drugs.”
Overall, he said, “compelling evidence from trials in hundreds of thousands of patients demonstrates that reducing LDL cholesterol to very low levels reduces cardiovascular events in broad populations and is extremely safe.”
Dr. Nissen reported consulting for many pharmaceutical companies and performing clinical trials for Amgen, AbbVie, AstraZeneca, Cerenis Therapeutics, Esperion Therapeutics, Lilly, Novartis, Novo Nordisk, the Medicines Company, Orexigen Therapeutics, Takeda, and Pfizer. He does not receive income for honoraria, speaking fees, or consulting fees as they are paid directly to charity.
SAN FRANCISCO – Powerful drugs now make it possible to lower LDL cholesterol levels to dramatically low levels. But is this a good idea? There are risks, and a cardiologist urged diabetes professionals to not overdo cholesterol reduction. But a colleague argued in favor of aggressively targeting “bad” cholesterol.
“We used to say you can’t be too rich or too thin. We now say you can’t be too rich or too thin or have a too-low LDL cholesterol,” said cardiologist Steven E. Nissen, MD, chairman of cardiovascular medicine at the Cleveland Clinic Foundation, who spoke at the annual scientific sessions of the American Diabetes Association about the wisdom of extreme LDL cholesterol lowering.
Dr. Nissen faced off in a debate with cardiologist Sanket Dhruva, MD, of the University of California, San Francisco, who doesn’t support aggressive LDL cholesterol lowering.
It is fine, Dr. Dhruva said, to treat patients so their LDL cholesterol levels drop below 100 mg/dL. “I don’t think there’s any argument there.”
But Dr. Dhruva questioned whether it’s a good idea to generally decrease LDL cholesterol well below 70 mg/dL, as is now possible with the use of proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors.
He pointed to a 2010 study that found aggressively lowering LDL cholesterol led to a mean net gain of 4.1 quality-adjusted life-years in high-risk patients, but less than 1 quality-adjusted life-year in low-risk patients. According to him, the study also found that the biggest benefits in both high- and low-risk patients came from the initial lower statin dose (Arch Intern Med. 2010 Jun 28;170[12]:1037-44).
“It’s really the statin initiation that provides the most benefit to our patients with diabetes,” Dr. Dhruva said.
Also, he added, a 2016 study questioned the value of aggressively lowering LDL cholesterol. It found that, although patients on statins with LDL cholesterol levels of 70-100 mg/dL had a lower risk of adverse cardiac outcomes than did those with levels between 100 and 130 mg/dL, no additional benefit was gained by achieving an LDL cholesterol level below 70 mg/dL (JAMA Intern Med. 2016 Aug 1;176[8]:1105-13)
As for risks, Dr. Dhruva highlighted a 2016 pooled analysis of 14 trials that linked the PCSK9 inhibitor alirocumab (Praluent) and LDL cholesterol levels below 25 mg/dL to significantly higher levels of cataracts, compared with levels of at least 25 mg/dL (hazard ratio, 3.4).
There are other reasons to be cautious of aggressive LDL cholesterol lowering. For one, many patients are not on statins when they’re prescribed PCSK-9 inhibitors. “We’re sometimes missing the building blocks before getting to expensive medications,” he said.
He added that PCSK-9 inhibitors are pricey, and some patients can’t get access to them. “Lipid control is incredibly important, but what about the stress or anxiety of our patients who are told this medication will reduce their cardiac risk but they can’t afford it? That’s not good for their cardiovascular risk.”
For his part, Dr. Nissen challenged Dr. Dhruva’s concerns about the cost of the drugs. “It’s not like they’re way out of line in terms of expense,” he said, noting that their cost – several thousand dollars a year – is similar to the cost of diabetes drugs known as glucagonlike peptide–1 receptor agonists and sodium-glucose transporter 2 inhibitors.
According to Dr. Nissen, multiple studies have supported aggressive LDL cholesterol lowering. “You’re going to see this over and over again in clinical trials: Every time we lower LDL by more, we get more reductions in morbidity and mortality.”
For example, he said, the FOURIER trial of the PCSK9 inhibitor evolocumab (Repatha) found that it lowered LDL cholesterol levels to a median 30 mg/dL “and reduced the risk of cardiovascular events. These findings show that patients with atherosclerotic cardiovascular disease benefit from lowering of LDL cholesterol levels below current targets [N Engl J Med 2017;376:1713-22].”
Dr. Nissen pointed to another study, this one also from 2017, that reported “in individuals with 5-year risk of major vascular events lower than 10%, each 1 mmol/L reduction in LDL cholesterol produced an absolute reduction in major vascular events of about 11 per 1,000 over 5 years. This benefit greatly exceeds any known hazards of statin therapy.”
In regard to adverse effects, he said, research has hinted at a slight uptick in blood sugar levels “that does not take away the major cardiovascular benefits of the drugs.”
Overall, he said, “compelling evidence from trials in hundreds of thousands of patients demonstrates that reducing LDL cholesterol to very low levels reduces cardiovascular events in broad populations and is extremely safe.”
Dr. Nissen reported consulting for many pharmaceutical companies and performing clinical trials for Amgen, AbbVie, AstraZeneca, Cerenis Therapeutics, Esperion Therapeutics, Lilly, Novartis, Novo Nordisk, the Medicines Company, Orexigen Therapeutics, Takeda, and Pfizer. He does not receive income for honoraria, speaking fees, or consulting fees as they are paid directly to charity.
SAN FRANCISCO – Powerful drugs now make it possible to lower LDL cholesterol levels to dramatically low levels. But is this a good idea? There are risks, and a cardiologist urged diabetes professionals to not overdo cholesterol reduction. But a colleague argued in favor of aggressively targeting “bad” cholesterol.
“We used to say you can’t be too rich or too thin. We now say you can’t be too rich or too thin or have a too-low LDL cholesterol,” said cardiologist Steven E. Nissen, MD, chairman of cardiovascular medicine at the Cleveland Clinic Foundation, who spoke at the annual scientific sessions of the American Diabetes Association about the wisdom of extreme LDL cholesterol lowering.
Dr. Nissen faced off in a debate with cardiologist Sanket Dhruva, MD, of the University of California, San Francisco, who doesn’t support aggressive LDL cholesterol lowering.
It is fine, Dr. Dhruva said, to treat patients so their LDL cholesterol levels drop below 100 mg/dL. “I don’t think there’s any argument there.”
But Dr. Dhruva questioned whether it’s a good idea to generally decrease LDL cholesterol well below 70 mg/dL, as is now possible with the use of proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors.
He pointed to a 2010 study that found aggressively lowering LDL cholesterol led to a mean net gain of 4.1 quality-adjusted life-years in high-risk patients, but less than 1 quality-adjusted life-year in low-risk patients. According to him, the study also found that the biggest benefits in both high- and low-risk patients came from the initial lower statin dose (Arch Intern Med. 2010 Jun 28;170[12]:1037-44).
“It’s really the statin initiation that provides the most benefit to our patients with diabetes,” Dr. Dhruva said.
Also, he added, a 2016 study questioned the value of aggressively lowering LDL cholesterol. It found that, although patients on statins with LDL cholesterol levels of 70-100 mg/dL had a lower risk of adverse cardiac outcomes than did those with levels between 100 and 130 mg/dL, no additional benefit was gained by achieving an LDL cholesterol level below 70 mg/dL (JAMA Intern Med. 2016 Aug 1;176[8]:1105-13)
As for risks, Dr. Dhruva highlighted a 2016 pooled analysis of 14 trials that linked the PCSK9 inhibitor alirocumab (Praluent) and LDL cholesterol levels below 25 mg/dL to significantly higher levels of cataracts, compared with levels of at least 25 mg/dL (hazard ratio, 3.4).
There are other reasons to be cautious of aggressive LDL cholesterol lowering. For one, many patients are not on statins when they’re prescribed PCSK-9 inhibitors. “We’re sometimes missing the building blocks before getting to expensive medications,” he said.
He added that PCSK-9 inhibitors are pricey, and some patients can’t get access to them. “Lipid control is incredibly important, but what about the stress or anxiety of our patients who are told this medication will reduce their cardiac risk but they can’t afford it? That’s not good for their cardiovascular risk.”
For his part, Dr. Nissen challenged Dr. Dhruva’s concerns about the cost of the drugs. “It’s not like they’re way out of line in terms of expense,” he said, noting that their cost – several thousand dollars a year – is similar to the cost of diabetes drugs known as glucagonlike peptide–1 receptor agonists and sodium-glucose transporter 2 inhibitors.
According to Dr. Nissen, multiple studies have supported aggressive LDL cholesterol lowering. “You’re going to see this over and over again in clinical trials: Every time we lower LDL by more, we get more reductions in morbidity and mortality.”
For example, he said, the FOURIER trial of the PCSK9 inhibitor evolocumab (Repatha) found that it lowered LDL cholesterol levels to a median 30 mg/dL “and reduced the risk of cardiovascular events. These findings show that patients with atherosclerotic cardiovascular disease benefit from lowering of LDL cholesterol levels below current targets [N Engl J Med 2017;376:1713-22].”
Dr. Nissen pointed to another study, this one also from 2017, that reported “in individuals with 5-year risk of major vascular events lower than 10%, each 1 mmol/L reduction in LDL cholesterol produced an absolute reduction in major vascular events of about 11 per 1,000 over 5 years. This benefit greatly exceeds any known hazards of statin therapy.”
In regard to adverse effects, he said, research has hinted at a slight uptick in blood sugar levels “that does not take away the major cardiovascular benefits of the drugs.”
Overall, he said, “compelling evidence from trials in hundreds of thousands of patients demonstrates that reducing LDL cholesterol to very low levels reduces cardiovascular events in broad populations and is extremely safe.”
Dr. Nissen reported consulting for many pharmaceutical companies and performing clinical trials for Amgen, AbbVie, AstraZeneca, Cerenis Therapeutics, Esperion Therapeutics, Lilly, Novartis, Novo Nordisk, the Medicines Company, Orexigen Therapeutics, Takeda, and Pfizer. He does not receive income for honoraria, speaking fees, or consulting fees as they are paid directly to charity.
EXPERT ANALYSIS FROM ADA 2019
Cognitive decline sped up after CHD
according to the results of a large prospective study with a median of 12 years of follow-up.
“We found that incident CHD was significantly associated with faster post–CHD-diagnosis cognitive decline, but not pre–CHD-diagnosis or short-term cognitive decline after the event,” Wuxiang Xie, PhD, of Peking University Health Science Center, Beijing, and associates wrote in the Journal of the American College of Cardiology. Linear mixed models showed that cognitive decline sped up during the year after incident CHD.
Past research had suggested a link between accelerated cognitive decline and CHD, but the temporal pattern of the relationship was unclear. For the study, Dr. Xie and associates followed 7,888 adults from the English Longitudinal Study of Aging who were an average of 62 years old and had no history of stroke, MI, angina, or dementia (Alzheimer’s disease or otherwise). All participants underwent a baseline cognitive assessment for verbal memory, semantic fluency, and temporal orientation, plus a median of six follow-up assessments.
In all, 480 (6%) participants developed CHD during follow-up. Their rate of cognitive decline remained constant before and immediately after their CHD diagnosis, but in subsequent years, they experienced significant accelerations in loss of global cognitive function, verbal memory, and temporal orientation even after accounting for time and many demographic and clinical variables. For example, the slope representing temporal change in global cognitive score decreased by a mean of 0.039 per year, compared with the pre-CHD slope (slope difference, –0.039; 95% confidence interval, –0.063 to –0.015; P =. 002). Semantic fluency also declined faster after CHD, but the difference, compared with before CHD, did not reach statistical significance (P = .11).
Individuals without CHD showed no such accelerations in cognitive decline throughout follow-up in adjusted models, the researchers wrote. “Based on repeated cognitive measurements over a long follow-up period, this study revealed a reliable and robust trajectory of cognitive decline [after CHD]. Future studies are warranted to determine the precise mechanisms linking incident CHD to cognitive decline.”
Funders included the National Natural Science Foundation of China, the Beijing Natural Science Foundation, and the Newton International Fellowship from the Academy of Medical Sciences. The researchers reported having no relevant financial disclosures.
SOURCE: Xie W et al. J Amer Coll Cardiol. 2019 Jun 17. doi: 10.1016/j.jacc.2019.04.019.
The findings “highlight the role of cardiovascular risk factors and cardiovascular health as crucial determinants of cognitive trajectories in later life,” wrote Suvi P. Rovio, PhD; Katja Pahkala, PhD; and Olli T. Raitakari, MD, PhD. For example, accelerated declines in verbal memory might indicate a specific vulnerability to vascular changes within the medial temporal lobe and hippocampus.
The fact that cognitive decline did not accelerate immediately after coronary heart disease suggests that CHD itself does not acutely alter the brain, such as by causing microinfarcts, they commented. Instead, CHD might induce longer-term shifts in cerebral vascular function by affecting the blood-brain barrier or perfusion and oxidation in the brain. While these complex relationships need further untangling, the study suggests interventions that cut CHD risk also might help prevent cognitive decline itself and slow the rate of cognitive decline if it occurs.
Dr. Rovio, Dr. Pahkala, and Dr. Raitakari are at the University of Turku (Finland) and Turku University Hospital. These comments are adapted from an editorial accompanying the article by Xie et al. (J Amer Coll Cardiol. 2019 Jun 17. doi: 10.1016/j.jacc.2019.04.020). They reported having no relevant financial disclosures.
The findings “highlight the role of cardiovascular risk factors and cardiovascular health as crucial determinants of cognitive trajectories in later life,” wrote Suvi P. Rovio, PhD; Katja Pahkala, PhD; and Olli T. Raitakari, MD, PhD. For example, accelerated declines in verbal memory might indicate a specific vulnerability to vascular changes within the medial temporal lobe and hippocampus.
The fact that cognitive decline did not accelerate immediately after coronary heart disease suggests that CHD itself does not acutely alter the brain, such as by causing microinfarcts, they commented. Instead, CHD might induce longer-term shifts in cerebral vascular function by affecting the blood-brain barrier or perfusion and oxidation in the brain. While these complex relationships need further untangling, the study suggests interventions that cut CHD risk also might help prevent cognitive decline itself and slow the rate of cognitive decline if it occurs.
Dr. Rovio, Dr. Pahkala, and Dr. Raitakari are at the University of Turku (Finland) and Turku University Hospital. These comments are adapted from an editorial accompanying the article by Xie et al. (J Amer Coll Cardiol. 2019 Jun 17. doi: 10.1016/j.jacc.2019.04.020). They reported having no relevant financial disclosures.
The findings “highlight the role of cardiovascular risk factors and cardiovascular health as crucial determinants of cognitive trajectories in later life,” wrote Suvi P. Rovio, PhD; Katja Pahkala, PhD; and Olli T. Raitakari, MD, PhD. For example, accelerated declines in verbal memory might indicate a specific vulnerability to vascular changes within the medial temporal lobe and hippocampus.
The fact that cognitive decline did not accelerate immediately after coronary heart disease suggests that CHD itself does not acutely alter the brain, such as by causing microinfarcts, they commented. Instead, CHD might induce longer-term shifts in cerebral vascular function by affecting the blood-brain barrier or perfusion and oxidation in the brain. While these complex relationships need further untangling, the study suggests interventions that cut CHD risk also might help prevent cognitive decline itself and slow the rate of cognitive decline if it occurs.
Dr. Rovio, Dr. Pahkala, and Dr. Raitakari are at the University of Turku (Finland) and Turku University Hospital. These comments are adapted from an editorial accompanying the article by Xie et al. (J Amer Coll Cardiol. 2019 Jun 17. doi: 10.1016/j.jacc.2019.04.020). They reported having no relevant financial disclosures.
according to the results of a large prospective study with a median of 12 years of follow-up.
“We found that incident CHD was significantly associated with faster post–CHD-diagnosis cognitive decline, but not pre–CHD-diagnosis or short-term cognitive decline after the event,” Wuxiang Xie, PhD, of Peking University Health Science Center, Beijing, and associates wrote in the Journal of the American College of Cardiology. Linear mixed models showed that cognitive decline sped up during the year after incident CHD.
Past research had suggested a link between accelerated cognitive decline and CHD, but the temporal pattern of the relationship was unclear. For the study, Dr. Xie and associates followed 7,888 adults from the English Longitudinal Study of Aging who were an average of 62 years old and had no history of stroke, MI, angina, or dementia (Alzheimer’s disease or otherwise). All participants underwent a baseline cognitive assessment for verbal memory, semantic fluency, and temporal orientation, plus a median of six follow-up assessments.
In all, 480 (6%) participants developed CHD during follow-up. Their rate of cognitive decline remained constant before and immediately after their CHD diagnosis, but in subsequent years, they experienced significant accelerations in loss of global cognitive function, verbal memory, and temporal orientation even after accounting for time and many demographic and clinical variables. For example, the slope representing temporal change in global cognitive score decreased by a mean of 0.039 per year, compared with the pre-CHD slope (slope difference, –0.039; 95% confidence interval, –0.063 to –0.015; P =. 002). Semantic fluency also declined faster after CHD, but the difference, compared with before CHD, did not reach statistical significance (P = .11).
Individuals without CHD showed no such accelerations in cognitive decline throughout follow-up in adjusted models, the researchers wrote. “Based on repeated cognitive measurements over a long follow-up period, this study revealed a reliable and robust trajectory of cognitive decline [after CHD]. Future studies are warranted to determine the precise mechanisms linking incident CHD to cognitive decline.”
Funders included the National Natural Science Foundation of China, the Beijing Natural Science Foundation, and the Newton International Fellowship from the Academy of Medical Sciences. The researchers reported having no relevant financial disclosures.
SOURCE: Xie W et al. J Amer Coll Cardiol. 2019 Jun 17. doi: 10.1016/j.jacc.2019.04.019.
according to the results of a large prospective study with a median of 12 years of follow-up.
“We found that incident CHD was significantly associated with faster post–CHD-diagnosis cognitive decline, but not pre–CHD-diagnosis or short-term cognitive decline after the event,” Wuxiang Xie, PhD, of Peking University Health Science Center, Beijing, and associates wrote in the Journal of the American College of Cardiology. Linear mixed models showed that cognitive decline sped up during the year after incident CHD.
Past research had suggested a link between accelerated cognitive decline and CHD, but the temporal pattern of the relationship was unclear. For the study, Dr. Xie and associates followed 7,888 adults from the English Longitudinal Study of Aging who were an average of 62 years old and had no history of stroke, MI, angina, or dementia (Alzheimer’s disease or otherwise). All participants underwent a baseline cognitive assessment for verbal memory, semantic fluency, and temporal orientation, plus a median of six follow-up assessments.
In all, 480 (6%) participants developed CHD during follow-up. Their rate of cognitive decline remained constant before and immediately after their CHD diagnosis, but in subsequent years, they experienced significant accelerations in loss of global cognitive function, verbal memory, and temporal orientation even after accounting for time and many demographic and clinical variables. For example, the slope representing temporal change in global cognitive score decreased by a mean of 0.039 per year, compared with the pre-CHD slope (slope difference, –0.039; 95% confidence interval, –0.063 to –0.015; P =. 002). Semantic fluency also declined faster after CHD, but the difference, compared with before CHD, did not reach statistical significance (P = .11).
Individuals without CHD showed no such accelerations in cognitive decline throughout follow-up in adjusted models, the researchers wrote. “Based on repeated cognitive measurements over a long follow-up period, this study revealed a reliable and robust trajectory of cognitive decline [after CHD]. Future studies are warranted to determine the precise mechanisms linking incident CHD to cognitive decline.”
Funders included the National Natural Science Foundation of China, the Beijing Natural Science Foundation, and the Newton International Fellowship from the Academy of Medical Sciences. The researchers reported having no relevant financial disclosures.
SOURCE: Xie W et al. J Amer Coll Cardiol. 2019 Jun 17. doi: 10.1016/j.jacc.2019.04.019.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY