User login
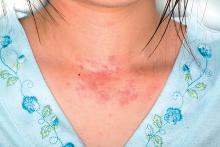
Atopic eczema linked to cardiovascular disease risk
according to a case-control study published online May 23 in BMJ.
In a population-based cohort study, researchers compared the electronic health records of 387,439 adults with eczema and 1,528,477 patients without eczema in the United Kingddom, matched according to age, sex, general practice, and calendar time, during 1998-2015. Patients were followed up for a median of 5.1 years
With the exception of cardiovascular death, atopic eczema was associated with all cardiovascular outcomes (MI, unstable angina, heart failure, atrial fibrillation, and stroke). The associations were stronger for severe atopic eczema, with significantly higher risks of MI, unstable angina, atrial fibrillation, stroke, cardiovascular death, and coronary revascularization among individuals with severe atopic eczema, compared with controls.
After adjustment for potential mediators such as body mass index, smoking, hyperlipidemia, diabetes, and severe alcohol use, individuals with severe eczema had a significant 37% increased risk of MI, 67% greater risk of heart failure, 35% greater risk of atrial fibrillation, 30% greater risk of cardiovascular death, and 36% greater risk of coronary revascularization, compared with controls with no eczema.
Increased cardiovascular risks also were seen in individuals whose atopic eczema was active more than half the time at follow-up. This group had a 37% greater risk of heart failure, 36% greater risk of unstable angina, and 19% greater risk of stroke, as well as significantly elevated risks of MI, atrial fibrillation, cardiovascular death, and coronary revascularization, compared with those without eczema.
Overall, atopic eczema contributed around 2.4% of the population-attributable risk for unstable angina, and 1.9% for heart failure (the highest population attributable risks). Ethnicity or high-dose corticosteroid use did not significantly impact outcomes.
Richard J. Silverwood, PhD, from the London School of Hygiene and Tropical Medicine, and his coauthors wrote that previous work examining the relationship between atopic eczema and cardiovascular disease had shown inconsistent outcomes, with some studies even pointing to a possible protective effect of mild atopic eczema.
“Mechanistic work suggests that atopic eczema may be associated with increased platelet activation and decreased fibrinolysis, which may increase the risk of clotting, though a recent study found no association with metabolite levels,” the authors wrote.
They noted that the strengths of their study were that it was largest to examine the association between atopic eczema and cardiovascular risk, and that they had access to data on body mass index, smoking, and severe alcohol use for most of the study population, which enabled them to adjust for these potential mediators.
“Consideration should be given to developing prevention strategies to reduce the risk of cardiovascular disease among patients with severe or predominantly active atopic eczema, including awareness of and screening for conventional cardiovascular risk factors by those providing clinical care,” they wrote.
The study was supported by the Wellcome Trust, and no relevant conflicts of interest were declared.
SOURCE: Silverwood R et al. BMJ 2018 May 23;361:k1786. doi: 10.1136/bmj.k1786.
The evidence in favor of a link between chronic inflammatory conditions, such as rheumatoid arthritis, and cardiovascular disease is growing. However, there are conflicting data and some of the uncertainty may be a result of a dose-response effect, where the increased cardiovascular risk is seen only in people with more severe disease.
This study and its finding of increased cardiovascular risk in patients with severe or more active eczema supports the case for targeted screening of this group for standard cardiovascular disease risk factors. It also could prompt incorporation of severe eczema as an independent cardiovascular disease risk factor in calculation of thresholds for primary prevention interventions.
The findings also may have implications for health care resources allocated to treatment of eczema, as prevention of cardiovascular disease could contribute to the argument in favor of the more expensive next-generation biologic treatments for eczema that are becoming available.
John R. Ingram, MD, is senior lecturer and consultant dermatologist, dermatology and academic wound healing in the division of infection and immunity at Cardiff (U.K.) University. These comments are taken from an accompanying editorial No conflicts of interest were declared. (BMJ. 2018 May 23. doi: 10.1136/bmj.k2064).
The evidence in favor of a link between chronic inflammatory conditions, such as rheumatoid arthritis, and cardiovascular disease is growing. However, there are conflicting data and some of the uncertainty may be a result of a dose-response effect, where the increased cardiovascular risk is seen only in people with more severe disease.
This study and its finding of increased cardiovascular risk in patients with severe or more active eczema supports the case for targeted screening of this group for standard cardiovascular disease risk factors. It also could prompt incorporation of severe eczema as an independent cardiovascular disease risk factor in calculation of thresholds for primary prevention interventions.
The findings also may have implications for health care resources allocated to treatment of eczema, as prevention of cardiovascular disease could contribute to the argument in favor of the more expensive next-generation biologic treatments for eczema that are becoming available.
John R. Ingram, MD, is senior lecturer and consultant dermatologist, dermatology and academic wound healing in the division of infection and immunity at Cardiff (U.K.) University. These comments are taken from an accompanying editorial No conflicts of interest were declared. (BMJ. 2018 May 23. doi: 10.1136/bmj.k2064).
The evidence in favor of a link between chronic inflammatory conditions, such as rheumatoid arthritis, and cardiovascular disease is growing. However, there are conflicting data and some of the uncertainty may be a result of a dose-response effect, where the increased cardiovascular risk is seen only in people with more severe disease.
This study and its finding of increased cardiovascular risk in patients with severe or more active eczema supports the case for targeted screening of this group for standard cardiovascular disease risk factors. It also could prompt incorporation of severe eczema as an independent cardiovascular disease risk factor in calculation of thresholds for primary prevention interventions.
The findings also may have implications for health care resources allocated to treatment of eczema, as prevention of cardiovascular disease could contribute to the argument in favor of the more expensive next-generation biologic treatments for eczema that are becoming available.
John R. Ingram, MD, is senior lecturer and consultant dermatologist, dermatology and academic wound healing in the division of infection and immunity at Cardiff (U.K.) University. These comments are taken from an accompanying editorial No conflicts of interest were declared. (BMJ. 2018 May 23. doi: 10.1136/bmj.k2064).
according to a case-control study published online May 23 in BMJ.
In a population-based cohort study, researchers compared the electronic health records of 387,439 adults with eczema and 1,528,477 patients without eczema in the United Kingddom, matched according to age, sex, general practice, and calendar time, during 1998-2015. Patients were followed up for a median of 5.1 years
With the exception of cardiovascular death, atopic eczema was associated with all cardiovascular outcomes (MI, unstable angina, heart failure, atrial fibrillation, and stroke). The associations were stronger for severe atopic eczema, with significantly higher risks of MI, unstable angina, atrial fibrillation, stroke, cardiovascular death, and coronary revascularization among individuals with severe atopic eczema, compared with controls.
After adjustment for potential mediators such as body mass index, smoking, hyperlipidemia, diabetes, and severe alcohol use, individuals with severe eczema had a significant 37% increased risk of MI, 67% greater risk of heart failure, 35% greater risk of atrial fibrillation, 30% greater risk of cardiovascular death, and 36% greater risk of coronary revascularization, compared with controls with no eczema.
Increased cardiovascular risks also were seen in individuals whose atopic eczema was active more than half the time at follow-up. This group had a 37% greater risk of heart failure, 36% greater risk of unstable angina, and 19% greater risk of stroke, as well as significantly elevated risks of MI, atrial fibrillation, cardiovascular death, and coronary revascularization, compared with those without eczema.
Overall, atopic eczema contributed around 2.4% of the population-attributable risk for unstable angina, and 1.9% for heart failure (the highest population attributable risks). Ethnicity or high-dose corticosteroid use did not significantly impact outcomes.
Richard J. Silverwood, PhD, from the London School of Hygiene and Tropical Medicine, and his coauthors wrote that previous work examining the relationship between atopic eczema and cardiovascular disease had shown inconsistent outcomes, with some studies even pointing to a possible protective effect of mild atopic eczema.
“Mechanistic work suggests that atopic eczema may be associated with increased platelet activation and decreased fibrinolysis, which may increase the risk of clotting, though a recent study found no association with metabolite levels,” the authors wrote.
They noted that the strengths of their study were that it was largest to examine the association between atopic eczema and cardiovascular risk, and that they had access to data on body mass index, smoking, and severe alcohol use for most of the study population, which enabled them to adjust for these potential mediators.
“Consideration should be given to developing prevention strategies to reduce the risk of cardiovascular disease among patients with severe or predominantly active atopic eczema, including awareness of and screening for conventional cardiovascular risk factors by those providing clinical care,” they wrote.
The study was supported by the Wellcome Trust, and no relevant conflicts of interest were declared.
SOURCE: Silverwood R et al. BMJ 2018 May 23;361:k1786. doi: 10.1136/bmj.k1786.
according to a case-control study published online May 23 in BMJ.
In a population-based cohort study, researchers compared the electronic health records of 387,439 adults with eczema and 1,528,477 patients without eczema in the United Kingddom, matched according to age, sex, general practice, and calendar time, during 1998-2015. Patients were followed up for a median of 5.1 years
With the exception of cardiovascular death, atopic eczema was associated with all cardiovascular outcomes (MI, unstable angina, heart failure, atrial fibrillation, and stroke). The associations were stronger for severe atopic eczema, with significantly higher risks of MI, unstable angina, atrial fibrillation, stroke, cardiovascular death, and coronary revascularization among individuals with severe atopic eczema, compared with controls.
After adjustment for potential mediators such as body mass index, smoking, hyperlipidemia, diabetes, and severe alcohol use, individuals with severe eczema had a significant 37% increased risk of MI, 67% greater risk of heart failure, 35% greater risk of atrial fibrillation, 30% greater risk of cardiovascular death, and 36% greater risk of coronary revascularization, compared with controls with no eczema.
Increased cardiovascular risks also were seen in individuals whose atopic eczema was active more than half the time at follow-up. This group had a 37% greater risk of heart failure, 36% greater risk of unstable angina, and 19% greater risk of stroke, as well as significantly elevated risks of MI, atrial fibrillation, cardiovascular death, and coronary revascularization, compared with those without eczema.
Overall, atopic eczema contributed around 2.4% of the population-attributable risk for unstable angina, and 1.9% for heart failure (the highest population attributable risks). Ethnicity or high-dose corticosteroid use did not significantly impact outcomes.
Richard J. Silverwood, PhD, from the London School of Hygiene and Tropical Medicine, and his coauthors wrote that previous work examining the relationship between atopic eczema and cardiovascular disease had shown inconsistent outcomes, with some studies even pointing to a possible protective effect of mild atopic eczema.
“Mechanistic work suggests that atopic eczema may be associated with increased platelet activation and decreased fibrinolysis, which may increase the risk of clotting, though a recent study found no association with metabolite levels,” the authors wrote.
They noted that the strengths of their study were that it was largest to examine the association between atopic eczema and cardiovascular risk, and that they had access to data on body mass index, smoking, and severe alcohol use for most of the study population, which enabled them to adjust for these potential mediators.
“Consideration should be given to developing prevention strategies to reduce the risk of cardiovascular disease among patients with severe or predominantly active atopic eczema, including awareness of and screening for conventional cardiovascular risk factors by those providing clinical care,” they wrote.
The study was supported by the Wellcome Trust, and no relevant conflicts of interest were declared.
SOURCE: Silverwood R et al. BMJ 2018 May 23;361:k1786. doi: 10.1136/bmj.k1786.
FROM THE BMJ
Key clinical point: Severe atopic eczema may be associated with a significantly increased risk of cardiovascular disease.
Major finding: Individuals with severe atopic eczema were at increased for cardiovascular disease, including a 67% greater risk of heart failure.
Study details: A population-based case-control cohort study in 387,439 patients with atopic eczema, compared with more than 1 million controls.
Disclosures: The study was supported by the Wellcome Trust, and no relevant conflicts of interest were declared.
Source: Silverwood R et al. BMJ. 2018 May 23;361:k1786.
Atopic dermatitis severity reduced by topical microbiome treatment
The Beginning Assessment of Cutaneous Treatment Efficacy of Roseomonas in Atopic Dermatitis trial; BACTERiAD I/II study, an open-label phase I/II trial, first looked at the therapeutic use of R. mucosa in 10 adults aged 18 years or older. Three sucrose mixtures with increasing doses of live R. mucosa bacteria were topically applied to two body areas – the antecubital fossae and a body surface of their choice – twice per week for 2 weeks per dose. At 6 weeks, the patients stopped using the mixtures and followed a 4-week washout phase.
Treatment was found to reduce mean antecubital SCORAD (SCORing Atopic Dermatitis) scores by 59.8%. Reduction in pruritus was even more pronounced, with a mean decrease of 78.5%. Treating the hands did not improve disease severity, even in patients whose symptoms improved in other body areas. One explanation may be the increased exposure of the hands to topical antimicrobials and environmental exposures, the researchers noted.
With the success in the adult cohort, the researchers enrolled five children aged 7-17 years in the study. These patients were treated twice weekly for 16 weeks. The pediatric patients experienced a mean decrease of 70.3% in their SCORAD scores. The mean decrease in pruritis was 78.8%.
All adults who responded continued to report improved symptoms after the washout period. The pediatric patients are now being evaluated in a washout period.
Four patients did not respond; three of them had a family history of AD persisting into adulthood. “The association between these complex medical histories and the lack of clinical response suggests that differences in heritable host and/or microbial factors may impact treatment responses,” wrote Ian A. Myles, MD, and his colleagues.
“Overall, our findings suggest the safety of topical R. mucosa therapy and justify continuation of our ongoing trial to assess safety and activity in a pediatric cohort of patients with AD. These studies will additionally assess changes in host serum markers, skin metabolomics, and the skin microbiota by culture and genomic methods.”
The researchers noted that expanding to the pediatric population will deepen understanding of topical microbiome transplantation and lay the foundation for placebo-controlled trials to assess efficacy.
This work was supported by the Intramural Research Program of the National Institutes of Allergy and Infectious Diseases and the National Institutes of Health. The researchers had no disclosures.
SOURCE: Myles IA et al. JCI Insight. 2018 May 3. doi: 10.1172/jci.insight.120608.
The Beginning Assessment of Cutaneous Treatment Efficacy of Roseomonas in Atopic Dermatitis trial; BACTERiAD I/II study, an open-label phase I/II trial, first looked at the therapeutic use of R. mucosa in 10 adults aged 18 years or older. Three sucrose mixtures with increasing doses of live R. mucosa bacteria were topically applied to two body areas – the antecubital fossae and a body surface of their choice – twice per week for 2 weeks per dose. At 6 weeks, the patients stopped using the mixtures and followed a 4-week washout phase.
Treatment was found to reduce mean antecubital SCORAD (SCORing Atopic Dermatitis) scores by 59.8%. Reduction in pruritus was even more pronounced, with a mean decrease of 78.5%. Treating the hands did not improve disease severity, even in patients whose symptoms improved in other body areas. One explanation may be the increased exposure of the hands to topical antimicrobials and environmental exposures, the researchers noted.
With the success in the adult cohort, the researchers enrolled five children aged 7-17 years in the study. These patients were treated twice weekly for 16 weeks. The pediatric patients experienced a mean decrease of 70.3% in their SCORAD scores. The mean decrease in pruritis was 78.8%.
All adults who responded continued to report improved symptoms after the washout period. The pediatric patients are now being evaluated in a washout period.
Four patients did not respond; three of them had a family history of AD persisting into adulthood. “The association between these complex medical histories and the lack of clinical response suggests that differences in heritable host and/or microbial factors may impact treatment responses,” wrote Ian A. Myles, MD, and his colleagues.
“Overall, our findings suggest the safety of topical R. mucosa therapy and justify continuation of our ongoing trial to assess safety and activity in a pediatric cohort of patients with AD. These studies will additionally assess changes in host serum markers, skin metabolomics, and the skin microbiota by culture and genomic methods.”
The researchers noted that expanding to the pediatric population will deepen understanding of topical microbiome transplantation and lay the foundation for placebo-controlled trials to assess efficacy.
This work was supported by the Intramural Research Program of the National Institutes of Allergy and Infectious Diseases and the National Institutes of Health. The researchers had no disclosures.
SOURCE: Myles IA et al. JCI Insight. 2018 May 3. doi: 10.1172/jci.insight.120608.
The Beginning Assessment of Cutaneous Treatment Efficacy of Roseomonas in Atopic Dermatitis trial; BACTERiAD I/II study, an open-label phase I/II trial, first looked at the therapeutic use of R. mucosa in 10 adults aged 18 years or older. Three sucrose mixtures with increasing doses of live R. mucosa bacteria were topically applied to two body areas – the antecubital fossae and a body surface of their choice – twice per week for 2 weeks per dose. At 6 weeks, the patients stopped using the mixtures and followed a 4-week washout phase.
Treatment was found to reduce mean antecubital SCORAD (SCORing Atopic Dermatitis) scores by 59.8%. Reduction in pruritus was even more pronounced, with a mean decrease of 78.5%. Treating the hands did not improve disease severity, even in patients whose symptoms improved in other body areas. One explanation may be the increased exposure of the hands to topical antimicrobials and environmental exposures, the researchers noted.
With the success in the adult cohort, the researchers enrolled five children aged 7-17 years in the study. These patients were treated twice weekly for 16 weeks. The pediatric patients experienced a mean decrease of 70.3% in their SCORAD scores. The mean decrease in pruritis was 78.8%.
All adults who responded continued to report improved symptoms after the washout period. The pediatric patients are now being evaluated in a washout period.
Four patients did not respond; three of them had a family history of AD persisting into adulthood. “The association between these complex medical histories and the lack of clinical response suggests that differences in heritable host and/or microbial factors may impact treatment responses,” wrote Ian A. Myles, MD, and his colleagues.
“Overall, our findings suggest the safety of topical R. mucosa therapy and justify continuation of our ongoing trial to assess safety and activity in a pediatric cohort of patients with AD. These studies will additionally assess changes in host serum markers, skin metabolomics, and the skin microbiota by culture and genomic methods.”
The researchers noted that expanding to the pediatric population will deepen understanding of topical microbiome transplantation and lay the foundation for placebo-controlled trials to assess efficacy.
This work was supported by the Intramural Research Program of the National Institutes of Allergy and Infectious Diseases and the National Institutes of Health. The researchers had no disclosures.
SOURCE: Myles IA et al. JCI Insight. 2018 May 3. doi: 10.1172/jci.insight.120608.
FROM JCI INSIGHT
Key clinical point: Roseomonas mucosa reduces disease severity.
Major finding: There were reductions in SCORAD scores of 78.5% and 70.3% in the adult and pediatric cohorts, respectively.
Study details: Case study of 10 adult and 5 pediatric patients with atopic dermatitis.
Disclosures: No relevant financial disclosures were reported.
Source: Myles IA et al. JCI Insight. 2018 May 3. doi: 10.1172/jci.insight.120608.
MDedge Daily News: How to handle opioid constipation
Bath emollients are a washout for childhood eczema. Does warfarin cause acute kidney injury? And there may be a new option for postpartum depression.
Listen to the MDedge Daily News podcast for all the details on today’s top news.
Bath emollients are a washout for childhood eczema. Does warfarin cause acute kidney injury? And there may be a new option for postpartum depression.
Listen to the MDedge Daily News podcast for all the details on today’s top news.
Bath emollients are a washout for childhood eczema. Does warfarin cause acute kidney injury? And there may be a new option for postpartum depression.
Listen to the MDedge Daily News podcast for all the details on today’s top news.
No benefits from bath emollients for childhood eczema
In a trial of 482 children with atopic dermatitis who were randomized to either use of prescribed emollient bath additives regularly for 12 months or no additives, in addition to usual care, there was no significant difference between the two groups in the patient-oriented eczema measure (POEM) – a score of symptoms in the previous week – for the first 16 weeks of treatment, reported Miriam Santer, MD, of the University of Southampton, England, and her coauthors. The mean POEM score was 7.5 in the bath additives group and 8.4 in the no bath additives group.
There also was no significant effect of emollient bath additives on the secondary outcomes, which included POEM over 52 weeks, dermatitis family impact, generic quality of life, number of eczema exacerbations, or topical corticosteroid or topical calcineurin inhibitor use.
The authors noted that there was the suggestion of benefit in children who bathed five or more times a week, and in children aged under 5 years, but in both cases, the numbers were small.
“Although there is evidence for the need for leave-on emollients and widespread clinical consensus around soap substitutes, there is less agreement about the potential additional benefits of bath additives and a dearth of evidence on their effectiveness,” wrote Dr. Santer and her associates. “Bath additives are, however, widely prescribed at a cost of more than 23 million pounds ($33 million US; 26 million euros) annually to the National Health Service in England.”
The bath emollients used in the study included Aveeno bath oil, and Oilatum bath additive and Balneum bath oil – products available largely in the United Kingdom – along with others. Adherence was reasonable, with 93% of participants in the emollient group using bath additives more than half or all the time, and 92% of those in the no bath additives group using them less than half the time or never.
The authors stressed that there was strong evidence that supported regular use of leave-on emollients, and clinical consensus around the use of emollients as soap substitutes. “These findings are timely for clinicians and prescribing advisers, as prescribing guidelines vary widely in their advice on the use of bath additives, and pressure on budgets has led to formularies becoming increasingly restrictive.”
The study was supported by the National Institute for Health Research. No conflicts of interest were declared.
SOURCE: Santer M et al. BMJ 2018 May 2. doi: 10.1136/bmj.k1332
The National Institute for Health and Care Excellence in the United Kingdom currently recommends regular use of “emollient wash products” for children with eczema, despite the fact that little good evidence of their benefit exists. It is therefore heartening that this study has largely addressed that evidence gap: There was no statistically significant difference between those children prescribed one of three bath additives in addition to standard care and those who received standard care alone.
However, there is still the question of whether younger children – particularly those who are bathed daily – might still benefit from bath emollients. Likewise, children who experience recurrent skin infections may derive some additional benefit from emollient bath additives with antiseptic properties.
Carsten Flohr, MD, is with the St. John’s Institute of Dermatology at Kings College London, and Amina Ahmed is a patient panel member at the Centre of Evidence-Based Dermatology in Nottingham, England. These comments are taken from an editorial accompanying the article by Santer et al. (BMJ. 2018 May 2. doi: 10.1136/bmj.k1791.) No conflicts of interest were declared.
The National Institute for Health and Care Excellence in the United Kingdom currently recommends regular use of “emollient wash products” for children with eczema, despite the fact that little good evidence of their benefit exists. It is therefore heartening that this study has largely addressed that evidence gap: There was no statistically significant difference between those children prescribed one of three bath additives in addition to standard care and those who received standard care alone.
However, there is still the question of whether younger children – particularly those who are bathed daily – might still benefit from bath emollients. Likewise, children who experience recurrent skin infections may derive some additional benefit from emollient bath additives with antiseptic properties.
Carsten Flohr, MD, is with the St. John’s Institute of Dermatology at Kings College London, and Amina Ahmed is a patient panel member at the Centre of Evidence-Based Dermatology in Nottingham, England. These comments are taken from an editorial accompanying the article by Santer et al. (BMJ. 2018 May 2. doi: 10.1136/bmj.k1791.) No conflicts of interest were declared.
The National Institute for Health and Care Excellence in the United Kingdom currently recommends regular use of “emollient wash products” for children with eczema, despite the fact that little good evidence of their benefit exists. It is therefore heartening that this study has largely addressed that evidence gap: There was no statistically significant difference between those children prescribed one of three bath additives in addition to standard care and those who received standard care alone.
However, there is still the question of whether younger children – particularly those who are bathed daily – might still benefit from bath emollients. Likewise, children who experience recurrent skin infections may derive some additional benefit from emollient bath additives with antiseptic properties.
Carsten Flohr, MD, is with the St. John’s Institute of Dermatology at Kings College London, and Amina Ahmed is a patient panel member at the Centre of Evidence-Based Dermatology in Nottingham, England. These comments are taken from an editorial accompanying the article by Santer et al. (BMJ. 2018 May 2. doi: 10.1136/bmj.k1791.) No conflicts of interest were declared.
In a trial of 482 children with atopic dermatitis who were randomized to either use of prescribed emollient bath additives regularly for 12 months or no additives, in addition to usual care, there was no significant difference between the two groups in the patient-oriented eczema measure (POEM) – a score of symptoms in the previous week – for the first 16 weeks of treatment, reported Miriam Santer, MD, of the University of Southampton, England, and her coauthors. The mean POEM score was 7.5 in the bath additives group and 8.4 in the no bath additives group.
There also was no significant effect of emollient bath additives on the secondary outcomes, which included POEM over 52 weeks, dermatitis family impact, generic quality of life, number of eczema exacerbations, or topical corticosteroid or topical calcineurin inhibitor use.
The authors noted that there was the suggestion of benefit in children who bathed five or more times a week, and in children aged under 5 years, but in both cases, the numbers were small.
“Although there is evidence for the need for leave-on emollients and widespread clinical consensus around soap substitutes, there is less agreement about the potential additional benefits of bath additives and a dearth of evidence on their effectiveness,” wrote Dr. Santer and her associates. “Bath additives are, however, widely prescribed at a cost of more than 23 million pounds ($33 million US; 26 million euros) annually to the National Health Service in England.”
The bath emollients used in the study included Aveeno bath oil, and Oilatum bath additive and Balneum bath oil – products available largely in the United Kingdom – along with others. Adherence was reasonable, with 93% of participants in the emollient group using bath additives more than half or all the time, and 92% of those in the no bath additives group using them less than half the time or never.
The authors stressed that there was strong evidence that supported regular use of leave-on emollients, and clinical consensus around the use of emollients as soap substitutes. “These findings are timely for clinicians and prescribing advisers, as prescribing guidelines vary widely in their advice on the use of bath additives, and pressure on budgets has led to formularies becoming increasingly restrictive.”
The study was supported by the National Institute for Health Research. No conflicts of interest were declared.
SOURCE: Santer M et al. BMJ 2018 May 2. doi: 10.1136/bmj.k1332
In a trial of 482 children with atopic dermatitis who were randomized to either use of prescribed emollient bath additives regularly for 12 months or no additives, in addition to usual care, there was no significant difference between the two groups in the patient-oriented eczema measure (POEM) – a score of symptoms in the previous week – for the first 16 weeks of treatment, reported Miriam Santer, MD, of the University of Southampton, England, and her coauthors. The mean POEM score was 7.5 in the bath additives group and 8.4 in the no bath additives group.
There also was no significant effect of emollient bath additives on the secondary outcomes, which included POEM over 52 weeks, dermatitis family impact, generic quality of life, number of eczema exacerbations, or topical corticosteroid or topical calcineurin inhibitor use.
The authors noted that there was the suggestion of benefit in children who bathed five or more times a week, and in children aged under 5 years, but in both cases, the numbers were small.
“Although there is evidence for the need for leave-on emollients and widespread clinical consensus around soap substitutes, there is less agreement about the potential additional benefits of bath additives and a dearth of evidence on their effectiveness,” wrote Dr. Santer and her associates. “Bath additives are, however, widely prescribed at a cost of more than 23 million pounds ($33 million US; 26 million euros) annually to the National Health Service in England.”
The bath emollients used in the study included Aveeno bath oil, and Oilatum bath additive and Balneum bath oil – products available largely in the United Kingdom – along with others. Adherence was reasonable, with 93% of participants in the emollient group using bath additives more than half or all the time, and 92% of those in the no bath additives group using them less than half the time or never.
The authors stressed that there was strong evidence that supported regular use of leave-on emollients, and clinical consensus around the use of emollients as soap substitutes. “These findings are timely for clinicians and prescribing advisers, as prescribing guidelines vary widely in their advice on the use of bath additives, and pressure on budgets has led to formularies becoming increasingly restrictive.”
The study was supported by the National Institute for Health Research. No conflicts of interest were declared.
SOURCE: Santer M et al. BMJ 2018 May 2. doi: 10.1136/bmj.k1332
FROM THE BMJ
Key clinical point: Emollient bath additives do not improve eczema scores in children.
Major finding: There was no significant difference in eczema scores between bath emollients or no bath emollients: the mean POEM score was 7.5 in the bath additives group and 8.4 in the no bath additives group.
Study details: An open-label randomized trial in 482 children with atopic dermatitis.
Disclosures: The study was supported by the National Institute for Health Research. No conflicts of interest were declared.
Source: Santer M et al. BMJ 2018 May 2. doi: 10.1136/bmj.k1332
Allergy, eczema common after pediatric solid organ transplantation
A total of 34% of children who underwent solid organ transplantation subsequently developed eczema, food allergy, rhinitis, eosinophilic gastrointestinal disease, or asthma, according to the results of a single-center retrospective cohort study.
Another 6.6% of patients developed autoimmunity, usually autoimmune cytopenia, inflammatory bowel disease, or vasculitis, wrote Nufar Marcus, MD, of the University of Toronto, and her associates.
Posttransplant allergy, autoimmunity, and immune-mediated disorders (PTAA) likely share a common pathogenesis “and may represent a unique state of post-transplant immune-dysregulation,” they wrote. The report was published in the Journal of Pediatrics.
The study included 273 children who underwent solid organ transplantation and were followed for a median 3.6 years (range, 1.7-6.3 years). None had immune-mediated conditions or allergies diagnosed at baseline. Posttransplantation allergies most commonly included eczema (51%), asthma (32%), food allergy (25%, including 5% with associated anaphylaxis), rhinitis (17%), and eosinophilic esophagitis, gastritis, or enteritis (13%).
Although only 31% of patients had information available on family history of allergy, those with a positive family history of allergy had a fivefold greater odds of posttransplantation PTAA, compared with other patients. Other risk factors for PTAA included female sex, young age at transplantation, eosinophilia, and a positive test for Epstein-Barr virus after transplantation, Dr. Marcus and associates said.
“The association of blood eosinophilia and PTAA reached statistical significance only when the transplant recipient was at least 6 months of age, demonstrating the nonspecific nature of abnormally high eosinophil counts during the first months of life,” they noted. The longer patients had eosinophilia after transplantation, the more likely they were to develop PTAA, “suggest[ing] a potential detrimental effect of prolonged activation of the eosinophilic-associated immune arms.”
Factors that appeared unlinked with PTAA included acute organ rejection, duration of posttransplantation steroidal treatment, organ type (living versus cadaveric), donor/recipient blood type and compatibility, infections besides Epstein-Barr virus, and posttransplant lymphoproliferative disease. “The specific type of post-transplantation immunosuppression regimen was neither associated nor protective of PTAA,” the investigators wrote. “However, a significant limitation was our inability to assess the effect of tacrolimus, as nearly all the cohort (97.8%) was treated with this medication.”
Ashley’s Angels fund provided support. The researchers reported having no conflicts of interest.
SOURCE: Marcus N et al. J Pediatr. 2018;196:154-60.
The study is one of several to highlight the occurrence of atopy and allergy following solid organ transplantation in children, Helen M. Evans, MBChB, wrote in an editorial accompanying the report by Marcus et al.
This report differed because it studied the differences in rates of atopy and allergy between transplanted solid organ groups. These occurred in 41% and 40% of liver and heart recipients, respectively, but in only 4% of kidney recipients. Atopy or allergy developed in 57% of multivisceral transplant patients, but the number of patients was very small (n = 7). The majority of the conditions developed within 1 year of transplantation.
The recent spike in these reports could signify better recognition of the problem or “the widespread switch of primary immunosuppression from cyclosporine to tacrolimus over the last few decades,” wrote Dr. Evans.
Most of these reports have been single-center retrospective studies, which are subject to inconsistent case definitions and recall bias, she noted. “The time is right for well-conducted multicenter prospective studies to better inform the true extent of these conditions after solid organ transplantation.”
In the meantime, transplantation centers should routinely track de novo eczema, allergy, and eosinophilic gastrointestinal disease in children being assessed for solid organ transplantation, and should take “rigorous” personal and family histories, said Dr. Evans. Ultimately, this work will help “minimize the risk of children developing these conditions” and “effectively treat them in the setting of immunosuppression after transplantation.”
Dr. Evans is a pediatric gastroenterologist at Starship Child Health in Aukland, New Zealand. She reported having no conflicts of interest. These comments summarize her editorial ( J Pediatr. 2018;196:10-11 ).
The study is one of several to highlight the occurrence of atopy and allergy following solid organ transplantation in children, Helen M. Evans, MBChB, wrote in an editorial accompanying the report by Marcus et al.
This report differed because it studied the differences in rates of atopy and allergy between transplanted solid organ groups. These occurred in 41% and 40% of liver and heart recipients, respectively, but in only 4% of kidney recipients. Atopy or allergy developed in 57% of multivisceral transplant patients, but the number of patients was very small (n = 7). The majority of the conditions developed within 1 year of transplantation.
The recent spike in these reports could signify better recognition of the problem or “the widespread switch of primary immunosuppression from cyclosporine to tacrolimus over the last few decades,” wrote Dr. Evans.
Most of these reports have been single-center retrospective studies, which are subject to inconsistent case definitions and recall bias, she noted. “The time is right for well-conducted multicenter prospective studies to better inform the true extent of these conditions after solid organ transplantation.”
In the meantime, transplantation centers should routinely track de novo eczema, allergy, and eosinophilic gastrointestinal disease in children being assessed for solid organ transplantation, and should take “rigorous” personal and family histories, said Dr. Evans. Ultimately, this work will help “minimize the risk of children developing these conditions” and “effectively treat them in the setting of immunosuppression after transplantation.”
Dr. Evans is a pediatric gastroenterologist at Starship Child Health in Aukland, New Zealand. She reported having no conflicts of interest. These comments summarize her editorial ( J Pediatr. 2018;196:10-11 ).
The study is one of several to highlight the occurrence of atopy and allergy following solid organ transplantation in children, Helen M. Evans, MBChB, wrote in an editorial accompanying the report by Marcus et al.
This report differed because it studied the differences in rates of atopy and allergy between transplanted solid organ groups. These occurred in 41% and 40% of liver and heart recipients, respectively, but in only 4% of kidney recipients. Atopy or allergy developed in 57% of multivisceral transplant patients, but the number of patients was very small (n = 7). The majority of the conditions developed within 1 year of transplantation.
The recent spike in these reports could signify better recognition of the problem or “the widespread switch of primary immunosuppression from cyclosporine to tacrolimus over the last few decades,” wrote Dr. Evans.
Most of these reports have been single-center retrospective studies, which are subject to inconsistent case definitions and recall bias, she noted. “The time is right for well-conducted multicenter prospective studies to better inform the true extent of these conditions after solid organ transplantation.”
In the meantime, transplantation centers should routinely track de novo eczema, allergy, and eosinophilic gastrointestinal disease in children being assessed for solid organ transplantation, and should take “rigorous” personal and family histories, said Dr. Evans. Ultimately, this work will help “minimize the risk of children developing these conditions” and “effectively treat them in the setting of immunosuppression after transplantation.”
Dr. Evans is a pediatric gastroenterologist at Starship Child Health in Aukland, New Zealand. She reported having no conflicts of interest. These comments summarize her editorial ( J Pediatr. 2018;196:10-11 ).
A total of 34% of children who underwent solid organ transplantation subsequently developed eczema, food allergy, rhinitis, eosinophilic gastrointestinal disease, or asthma, according to the results of a single-center retrospective cohort study.
Another 6.6% of patients developed autoimmunity, usually autoimmune cytopenia, inflammatory bowel disease, or vasculitis, wrote Nufar Marcus, MD, of the University of Toronto, and her associates.
Posttransplant allergy, autoimmunity, and immune-mediated disorders (PTAA) likely share a common pathogenesis “and may represent a unique state of post-transplant immune-dysregulation,” they wrote. The report was published in the Journal of Pediatrics.
The study included 273 children who underwent solid organ transplantation and were followed for a median 3.6 years (range, 1.7-6.3 years). None had immune-mediated conditions or allergies diagnosed at baseline. Posttransplantation allergies most commonly included eczema (51%), asthma (32%), food allergy (25%, including 5% with associated anaphylaxis), rhinitis (17%), and eosinophilic esophagitis, gastritis, or enteritis (13%).
Although only 31% of patients had information available on family history of allergy, those with a positive family history of allergy had a fivefold greater odds of posttransplantation PTAA, compared with other patients. Other risk factors for PTAA included female sex, young age at transplantation, eosinophilia, and a positive test for Epstein-Barr virus after transplantation, Dr. Marcus and associates said.
“The association of blood eosinophilia and PTAA reached statistical significance only when the transplant recipient was at least 6 months of age, demonstrating the nonspecific nature of abnormally high eosinophil counts during the first months of life,” they noted. The longer patients had eosinophilia after transplantation, the more likely they were to develop PTAA, “suggest[ing] a potential detrimental effect of prolonged activation of the eosinophilic-associated immune arms.”
Factors that appeared unlinked with PTAA included acute organ rejection, duration of posttransplantation steroidal treatment, organ type (living versus cadaveric), donor/recipient blood type and compatibility, infections besides Epstein-Barr virus, and posttransplant lymphoproliferative disease. “The specific type of post-transplantation immunosuppression regimen was neither associated nor protective of PTAA,” the investigators wrote. “However, a significant limitation was our inability to assess the effect of tacrolimus, as nearly all the cohort (97.8%) was treated with this medication.”
Ashley’s Angels fund provided support. The researchers reported having no conflicts of interest.
SOURCE: Marcus N et al. J Pediatr. 2018;196:154-60.
A total of 34% of children who underwent solid organ transplantation subsequently developed eczema, food allergy, rhinitis, eosinophilic gastrointestinal disease, or asthma, according to the results of a single-center retrospective cohort study.
Another 6.6% of patients developed autoimmunity, usually autoimmune cytopenia, inflammatory bowel disease, or vasculitis, wrote Nufar Marcus, MD, of the University of Toronto, and her associates.
Posttransplant allergy, autoimmunity, and immune-mediated disorders (PTAA) likely share a common pathogenesis “and may represent a unique state of post-transplant immune-dysregulation,” they wrote. The report was published in the Journal of Pediatrics.
The study included 273 children who underwent solid organ transplantation and were followed for a median 3.6 years (range, 1.7-6.3 years). None had immune-mediated conditions or allergies diagnosed at baseline. Posttransplantation allergies most commonly included eczema (51%), asthma (32%), food allergy (25%, including 5% with associated anaphylaxis), rhinitis (17%), and eosinophilic esophagitis, gastritis, or enteritis (13%).
Although only 31% of patients had information available on family history of allergy, those with a positive family history of allergy had a fivefold greater odds of posttransplantation PTAA, compared with other patients. Other risk factors for PTAA included female sex, young age at transplantation, eosinophilia, and a positive test for Epstein-Barr virus after transplantation, Dr. Marcus and associates said.
“The association of blood eosinophilia and PTAA reached statistical significance only when the transplant recipient was at least 6 months of age, demonstrating the nonspecific nature of abnormally high eosinophil counts during the first months of life,” they noted. The longer patients had eosinophilia after transplantation, the more likely they were to develop PTAA, “suggest[ing] a potential detrimental effect of prolonged activation of the eosinophilic-associated immune arms.”
Factors that appeared unlinked with PTAA included acute organ rejection, duration of posttransplantation steroidal treatment, organ type (living versus cadaveric), donor/recipient blood type and compatibility, infections besides Epstein-Barr virus, and posttransplant lymphoproliferative disease. “The specific type of post-transplantation immunosuppression regimen was neither associated nor protective of PTAA,” the investigators wrote. “However, a significant limitation was our inability to assess the effect of tacrolimus, as nearly all the cohort (97.8%) was treated with this medication.”
Ashley’s Angels fund provided support. The researchers reported having no conflicts of interest.
SOURCE: Marcus N et al. J Pediatr. 2018;196:154-60.
FROM JOURNAL OF PEDIATRICS
Key clinical point: Children undergoing solid organ transplantation often developed allergy or autoimmunity.
Major finding: Study details: Single-center retrospective cross-sectional study of 273 patients aged 18 and under who underwent solid organ transplantation followed for a median 3.6 years.
Disclosures: Ashley’s Angels fund provided support. The researchers reported having no conflicts of interest.
Source: Marcus N et al. J Pediatr. 2018;196:154-60.
Absurdity
Absurdity is everywhere you look. Or don’t look.
As the old comedian Henny Youngman might have said:
Take my prior authorizations. Please!
Prior authorizations
1. Marissa had been taking isotretinoin for 2 months. She learned that three 20-mg capsules would cost her less than the two 30-mg capsules she’d been on.
My secretary called them. The insurance representative (Pharmacist? Clerk? Gal who stopped by to read the gas meter?) couldn’t help. “When I input your information, it issues a denial.” (Who is “it”? Watson’s evil twin Jensen?)
I got on the phone.
“Forgive me,” I said, gently, “but Marissa has been taking isotretinoin for 2 months, 60-mg per day. She was taking two 30’s. I want her to have three 20’s. They both add up to 60 mg. It’s the same dose. Why do you need to authorize it again?”
“Let me input that,” she replied. “Oh, now it’s accepting it. Your patient can have up to four pills a day.”
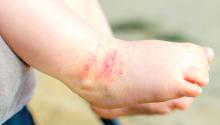
I was going to say she only needs three, but kept my mouth shut. Maybe Jensen only authorizes even numbers. 2. Danny has the worst atopic dermatitis I’ve ever seen. It’s all over him and never lets up. Topical steroids don’t touch it. Even prednisone – he’s had plenty over the years – barely makes a dent. I put in a Prior Authorization request for dupilumab.
“Your request for dupilumab has been denied,” read the insurer’s reply. “You have not shown failure with tacrolimus ointment or crisaborole.”
Say what? Prednisone didn’t help, and they expect tacrolimus or crisaborole to do the job?
I prescribed tacrolimus (which doesn’t come in a big enough tube to cover Daniel’s affected area anyway). It failed. Amazing.
“Your request for dupilumab has been denied,” said the insurer. “You have not also shown failure with crisaborole.”
Really? OK, I prescribed crisaborole. They denied coverage for it.
Now I was really getting into this. I wrote them. “I prescribed crisaborole.” I observed, “because you asked me to.”
They approved crisaborole. It failed. I reapplied for dupilumab. No response.
I called the insurer’s medical director, on a mobile with a Missouri exchange. After some telephone tag, he called back. “I cannot discuss this case with you,” he said, “because I have already made my determination.” Then he hung up without telling me what his determination was.
Further phone calls went unanswered. I thought Missouri was the Show-Me state.
The patient remained miserable. I decided to try one last time and wrote a long, sarcastic letter, detailing the whole episode. My secretary sent it off.
They approved dupilumab within the hour.
Malice? Nah, that’s giving them too much credit.
Now all Daniel has to do is improve.
Patient privacy!
Some news from abroad: what we know as HIPAA is called the “Data Protection Act” in the United Kingdom.
“We are obliged to conform to the demands of the Data Protection Act, and this specifically applies to the rabbi publicly mentioning the names of individuals who are unwell. The rabbi can only mention specific individuals with their permission or that of a relative designated by the sick person to do so. Anyone wishing for the rabbi to say a public prayer on their behalf must contact him directly by phone, text, or e-mail. To do anything else is breaking the law.”
If someone breaks this law, perhaps the rabbi can assist with atonement.
In any event, henceforth all entreaties to the Almighty must be encrypted. At least in the U.K.
What???!!!
Marina showed me her sunscreen. The label read, “Protects against UVA and UVB rays.”
“What’s the problem?” I asked.
She showed me our American Academy of Dermatology-produced sunscreen handout, which recommends “a broad-spectrum sunscreen that protects against both UVA and UVB rays, both of which cause cancer.”
“Does this mean my sunscreen causes cancer?” asked Marina.
“Not to worry,” I assured her.
I sighed and wafted a small prayer heavenward. Encrypted, of course.
Lo, the answer from above may tarry, but He will never forget His password.
Dr. Rockoff practices dermatology in Brookline, Mass., and is a longtime contributor to Dermatology News. He serves on the clinical faculty at Tufts University, Boston, and has taught senior medical students and other trainees for 30 years. His second book, “Act Like a Doctor, Think Like a Patient,” is available at amazon.com and barnesandnoble.com. Write to him at dermnews@mdedge.com.
Absurdity is everywhere you look. Or don’t look.
As the old comedian Henny Youngman might have said:
Take my prior authorizations. Please!
Prior authorizations
1. Marissa had been taking isotretinoin for 2 months. She learned that three 20-mg capsules would cost her less than the two 30-mg capsules she’d been on.
My secretary called them. The insurance representative (Pharmacist? Clerk? Gal who stopped by to read the gas meter?) couldn’t help. “When I input your information, it issues a denial.” (Who is “it”? Watson’s evil twin Jensen?)
I got on the phone.
“Forgive me,” I said, gently, “but Marissa has been taking isotretinoin for 2 months, 60-mg per day. She was taking two 30’s. I want her to have three 20’s. They both add up to 60 mg. It’s the same dose. Why do you need to authorize it again?”
“Let me input that,” she replied. “Oh, now it’s accepting it. Your patient can have up to four pills a day.”
I was going to say she only needs three, but kept my mouth shut. Maybe Jensen only authorizes even numbers. 2. Danny has the worst atopic dermatitis I’ve ever seen. It’s all over him and never lets up. Topical steroids don’t touch it. Even prednisone – he’s had plenty over the years – barely makes a dent. I put in a Prior Authorization request for dupilumab.
“Your request for dupilumab has been denied,” read the insurer’s reply. “You have not shown failure with tacrolimus ointment or crisaborole.”
Say what? Prednisone didn’t help, and they expect tacrolimus or crisaborole to do the job?
I prescribed tacrolimus (which doesn’t come in a big enough tube to cover Daniel’s affected area anyway). It failed. Amazing.
“Your request for dupilumab has been denied,” said the insurer. “You have not also shown failure with crisaborole.”
Really? OK, I prescribed crisaborole. They denied coverage for it.
Now I was really getting into this. I wrote them. “I prescribed crisaborole.” I observed, “because you asked me to.”
They approved crisaborole. It failed. I reapplied for dupilumab. No response.
I called the insurer’s medical director, on a mobile with a Missouri exchange. After some telephone tag, he called back. “I cannot discuss this case with you,” he said, “because I have already made my determination.” Then he hung up without telling me what his determination was.
Further phone calls went unanswered. I thought Missouri was the Show-Me state.
The patient remained miserable. I decided to try one last time and wrote a long, sarcastic letter, detailing the whole episode. My secretary sent it off.
They approved dupilumab within the hour.
Malice? Nah, that’s giving them too much credit.
Now all Daniel has to do is improve.
Patient privacy!
Some news from abroad: what we know as HIPAA is called the “Data Protection Act” in the United Kingdom.
“We are obliged to conform to the demands of the Data Protection Act, and this specifically applies to the rabbi publicly mentioning the names of individuals who are unwell. The rabbi can only mention specific individuals with their permission or that of a relative designated by the sick person to do so. Anyone wishing for the rabbi to say a public prayer on their behalf must contact him directly by phone, text, or e-mail. To do anything else is breaking the law.”
If someone breaks this law, perhaps the rabbi can assist with atonement.
In any event, henceforth all entreaties to the Almighty must be encrypted. At least in the U.K.
What???!!!
Marina showed me her sunscreen. The label read, “Protects against UVA and UVB rays.”
“What’s the problem?” I asked.
She showed me our American Academy of Dermatology-produced sunscreen handout, which recommends “a broad-spectrum sunscreen that protects against both UVA and UVB rays, both of which cause cancer.”
“Does this mean my sunscreen causes cancer?” asked Marina.
“Not to worry,” I assured her.
I sighed and wafted a small prayer heavenward. Encrypted, of course.
Lo, the answer from above may tarry, but He will never forget His password.
Dr. Rockoff practices dermatology in Brookline, Mass., and is a longtime contributor to Dermatology News. He serves on the clinical faculty at Tufts University, Boston, and has taught senior medical students and other trainees for 30 years. His second book, “Act Like a Doctor, Think Like a Patient,” is available at amazon.com and barnesandnoble.com. Write to him at dermnews@mdedge.com.
Absurdity is everywhere you look. Or don’t look.
As the old comedian Henny Youngman might have said:
Take my prior authorizations. Please!
Prior authorizations
1. Marissa had been taking isotretinoin for 2 months. She learned that three 20-mg capsules would cost her less than the two 30-mg capsules she’d been on.
My secretary called them. The insurance representative (Pharmacist? Clerk? Gal who stopped by to read the gas meter?) couldn’t help. “When I input your information, it issues a denial.” (Who is “it”? Watson’s evil twin Jensen?)
I got on the phone.
“Forgive me,” I said, gently, “but Marissa has been taking isotretinoin for 2 months, 60-mg per day. She was taking two 30’s. I want her to have three 20’s. They both add up to 60 mg. It’s the same dose. Why do you need to authorize it again?”
“Let me input that,” she replied. “Oh, now it’s accepting it. Your patient can have up to four pills a day.”
I was going to say she only needs three, but kept my mouth shut. Maybe Jensen only authorizes even numbers. 2. Danny has the worst atopic dermatitis I’ve ever seen. It’s all over him and never lets up. Topical steroids don’t touch it. Even prednisone – he’s had plenty over the years – barely makes a dent. I put in a Prior Authorization request for dupilumab.
“Your request for dupilumab has been denied,” read the insurer’s reply. “You have not shown failure with tacrolimus ointment or crisaborole.”
Say what? Prednisone didn’t help, and they expect tacrolimus or crisaborole to do the job?
I prescribed tacrolimus (which doesn’t come in a big enough tube to cover Daniel’s affected area anyway). It failed. Amazing.
“Your request for dupilumab has been denied,” said the insurer. “You have not also shown failure with crisaborole.”
Really? OK, I prescribed crisaborole. They denied coverage for it.
Now I was really getting into this. I wrote them. “I prescribed crisaborole.” I observed, “because you asked me to.”
They approved crisaborole. It failed. I reapplied for dupilumab. No response.
I called the insurer’s medical director, on a mobile with a Missouri exchange. After some telephone tag, he called back. “I cannot discuss this case with you,” he said, “because I have already made my determination.” Then he hung up without telling me what his determination was.
Further phone calls went unanswered. I thought Missouri was the Show-Me state.
The patient remained miserable. I decided to try one last time and wrote a long, sarcastic letter, detailing the whole episode. My secretary sent it off.
They approved dupilumab within the hour.
Malice? Nah, that’s giving them too much credit.
Now all Daniel has to do is improve.
Patient privacy!
Some news from abroad: what we know as HIPAA is called the “Data Protection Act” in the United Kingdom.
“We are obliged to conform to the demands of the Data Protection Act, and this specifically applies to the rabbi publicly mentioning the names of individuals who are unwell. The rabbi can only mention specific individuals with their permission or that of a relative designated by the sick person to do so. Anyone wishing for the rabbi to say a public prayer on their behalf must contact him directly by phone, text, or e-mail. To do anything else is breaking the law.”
If someone breaks this law, perhaps the rabbi can assist with atonement.
In any event, henceforth all entreaties to the Almighty must be encrypted. At least in the U.K.
What???!!!
Marina showed me her sunscreen. The label read, “Protects against UVA and UVB rays.”
“What’s the problem?” I asked.
She showed me our American Academy of Dermatology-produced sunscreen handout, which recommends “a broad-spectrum sunscreen that protects against both UVA and UVB rays, both of which cause cancer.”
“Does this mean my sunscreen causes cancer?” asked Marina.
“Not to worry,” I assured her.
I sighed and wafted a small prayer heavenward. Encrypted, of course.
Lo, the answer from above may tarry, but He will never forget His password.
Dr. Rockoff practices dermatology in Brookline, Mass., and is a longtime contributor to Dermatology News. He serves on the clinical faculty at Tufts University, Boston, and has taught senior medical students and other trainees for 30 years. His second book, “Act Like a Doctor, Think Like a Patient,” is available at amazon.com and barnesandnoble.com. Write to him at dermnews@mdedge.com.
IL-22 blocker investigated in phase 2a atopic dermatitis study
Fezakinumab, an interleukin-22 monoclonal antibody, “resulted in consistent improvements in clinical and molecular disease scores as compared with placebo” in a phase 2a study of adults with moderate to severe atopic dermatitis (AD), according to Emma Guttman-Yassky, MD, of Icahn School of Medicine at Mount Sinai, New York, and her associates.
In the double-blind, placebo-controlled trial, 60 patients were randomized to intravenous fezakinumab every 2 weeks for 10 weeks (40 patients) or placebo (20). Beginning at week 4, those who received fezakinumab “showed a consistently stronger and more significant mean SCORAD decline from baseline” compared with those on placebo. This became statistically significant at weeks 6-10 (P less than .05). “Differences between drug and placebo extended beyond the last dose” at week 10, they noted.*
The primary endpoint, the change in the SCORAD score from baseline at 12 weeks, was not statistically significant, however.
In addition, progressive reductions were seen during weeks 14-20, with a significant difference between the drug and placebo arms (P = .049) observed at week 20.
The mean decline in body surface area was “consistently stronger” among those on the biologic, “and was significantly different from the placebo group starting from week 8 until the end of study,” which included the 12th week (P = .009), the researchers noted.
In addition, among those on fezakinumab, mean improvements in Investigator Global Assessment scores compared with baseline were stronger and appeared earlier and were significantly different compared with those on placebo at week 16 (P less than .001).
There were two serious adverse events among those in the treatment group: facial cellulitis after a dental procedure and a pregnancy with elective termination, which were considered “most likely unrelated” to treatment. In the fezakinumab group, four patients had upper respiratory tract infections, the most common adverse event.
“This is the first clinical trial investigating IL-22 blockade in patients with AD, and the first to suggest a pathogenic role of IL-22 in any human disease,” the authors concluded.
SOURCE: Guttman-Yassky et al. J Am Acad Dermatol. 78(5);872-81.
Correction, 4/27/17: An earlier version of this article misstated the statistical significance of the primary endpoint.
Fezakinumab, an interleukin-22 monoclonal antibody, “resulted in consistent improvements in clinical and molecular disease scores as compared with placebo” in a phase 2a study of adults with moderate to severe atopic dermatitis (AD), according to Emma Guttman-Yassky, MD, of Icahn School of Medicine at Mount Sinai, New York, and her associates.
In the double-blind, placebo-controlled trial, 60 patients were randomized to intravenous fezakinumab every 2 weeks for 10 weeks (40 patients) or placebo (20). Beginning at week 4, those who received fezakinumab “showed a consistently stronger and more significant mean SCORAD decline from baseline” compared with those on placebo. This became statistically significant at weeks 6-10 (P less than .05). “Differences between drug and placebo extended beyond the last dose” at week 10, they noted.*
The primary endpoint, the change in the SCORAD score from baseline at 12 weeks, was not statistically significant, however.
In addition, progressive reductions were seen during weeks 14-20, with a significant difference between the drug and placebo arms (P = .049) observed at week 20.
The mean decline in body surface area was “consistently stronger” among those on the biologic, “and was significantly different from the placebo group starting from week 8 until the end of study,” which included the 12th week (P = .009), the researchers noted.
In addition, among those on fezakinumab, mean improvements in Investigator Global Assessment scores compared with baseline were stronger and appeared earlier and were significantly different compared with those on placebo at week 16 (P less than .001).
There were two serious adverse events among those in the treatment group: facial cellulitis after a dental procedure and a pregnancy with elective termination, which were considered “most likely unrelated” to treatment. In the fezakinumab group, four patients had upper respiratory tract infections, the most common adverse event.
“This is the first clinical trial investigating IL-22 blockade in patients with AD, and the first to suggest a pathogenic role of IL-22 in any human disease,” the authors concluded.
SOURCE: Guttman-Yassky et al. J Am Acad Dermatol. 78(5);872-81.
Correction, 4/27/17: An earlier version of this article misstated the statistical significance of the primary endpoint.
Fezakinumab, an interleukin-22 monoclonal antibody, “resulted in consistent improvements in clinical and molecular disease scores as compared with placebo” in a phase 2a study of adults with moderate to severe atopic dermatitis (AD), according to Emma Guttman-Yassky, MD, of Icahn School of Medicine at Mount Sinai, New York, and her associates.
In the double-blind, placebo-controlled trial, 60 patients were randomized to intravenous fezakinumab every 2 weeks for 10 weeks (40 patients) or placebo (20). Beginning at week 4, those who received fezakinumab “showed a consistently stronger and more significant mean SCORAD decline from baseline” compared with those on placebo. This became statistically significant at weeks 6-10 (P less than .05). “Differences between drug and placebo extended beyond the last dose” at week 10, they noted.*
The primary endpoint, the change in the SCORAD score from baseline at 12 weeks, was not statistically significant, however.
In addition, progressive reductions were seen during weeks 14-20, with a significant difference between the drug and placebo arms (P = .049) observed at week 20.
The mean decline in body surface area was “consistently stronger” among those on the biologic, “and was significantly different from the placebo group starting from week 8 until the end of study,” which included the 12th week (P = .009), the researchers noted.
In addition, among those on fezakinumab, mean improvements in Investigator Global Assessment scores compared with baseline were stronger and appeared earlier and were significantly different compared with those on placebo at week 16 (P less than .001).
There were two serious adverse events among those in the treatment group: facial cellulitis after a dental procedure and a pregnancy with elective termination, which were considered “most likely unrelated” to treatment. In the fezakinumab group, four patients had upper respiratory tract infections, the most common adverse event.
“This is the first clinical trial investigating IL-22 blockade in patients with AD, and the first to suggest a pathogenic role of IL-22 in any human disease,” the authors concluded.
SOURCE: Guttman-Yassky et al. J Am Acad Dermatol. 78(5);872-81.
Correction, 4/27/17: An earlier version of this article misstated the statistical significance of the primary endpoint.
FROM THE JOURNAL OF the AMERICAN ACADEMY OF DERMATOLOGY
Peanut exposure in at-risk infants does not reduce eczema, other allergic diseases
and does not hasten the resolution of eczema, according to George du Toit, MB, and his associates.
In a study published in The Journal of Allergy and Clinical Immunology, they reported on the results of a 12-month extension of the Learning Early About Peanut Allergy (LEAP) study, which found that early introduction of peanuts into the diets of infants at high risk of peanut allergy markedly reduced their risk of developing peanut allergy at age 5, compared with those who did not consume peanuts. As infants, the majority had severe eczema, which, with egg allergy, was used to identify those at high risk.
In the extension study, the rates of eczema decreased across study time points to 72 months of age in both groups, to 39% in the peanut avoidance group and 37% in the peanut consumption group. Eczema severity decreased across study time points, and there were no significant differences in severity between the two groups at any time point.
There were also no differences between the two groups in the rates of asthma, seasonal rhinoconjunctivitis, and perennial rhinoconjunctivitis at 30, 60, and 72 months of age.
“The underlying immune mechanisms associated with tolerance to peanut do not alter the natural history of allergic disease,” the researchers concluded. “Different prevention strategies or strategies that include multiple dietary interventions need to be tested to assess whether the reduction in peanut allergy observed in the LEAP consumption group can be extended to other common food allergens and allergic diseases.”
SOURCE: Du Toit G et al. J Allergy Clin Immunol. 2018 Apr;141[4]1343-53.
and does not hasten the resolution of eczema, according to George du Toit, MB, and his associates.
In a study published in The Journal of Allergy and Clinical Immunology, they reported on the results of a 12-month extension of the Learning Early About Peanut Allergy (LEAP) study, which found that early introduction of peanuts into the diets of infants at high risk of peanut allergy markedly reduced their risk of developing peanut allergy at age 5, compared with those who did not consume peanuts. As infants, the majority had severe eczema, which, with egg allergy, was used to identify those at high risk.
In the extension study, the rates of eczema decreased across study time points to 72 months of age in both groups, to 39% in the peanut avoidance group and 37% in the peanut consumption group. Eczema severity decreased across study time points, and there were no significant differences in severity between the two groups at any time point.
There were also no differences between the two groups in the rates of asthma, seasonal rhinoconjunctivitis, and perennial rhinoconjunctivitis at 30, 60, and 72 months of age.
“The underlying immune mechanisms associated with tolerance to peanut do not alter the natural history of allergic disease,” the researchers concluded. “Different prevention strategies or strategies that include multiple dietary interventions need to be tested to assess whether the reduction in peanut allergy observed in the LEAP consumption group can be extended to other common food allergens and allergic diseases.”
SOURCE: Du Toit G et al. J Allergy Clin Immunol. 2018 Apr;141[4]1343-53.
and does not hasten the resolution of eczema, according to George du Toit, MB, and his associates.
In a study published in The Journal of Allergy and Clinical Immunology, they reported on the results of a 12-month extension of the Learning Early About Peanut Allergy (LEAP) study, which found that early introduction of peanuts into the diets of infants at high risk of peanut allergy markedly reduced their risk of developing peanut allergy at age 5, compared with those who did not consume peanuts. As infants, the majority had severe eczema, which, with egg allergy, was used to identify those at high risk.
In the extension study, the rates of eczema decreased across study time points to 72 months of age in both groups, to 39% in the peanut avoidance group and 37% in the peanut consumption group. Eczema severity decreased across study time points, and there were no significant differences in severity between the two groups at any time point.
There were also no differences between the two groups in the rates of asthma, seasonal rhinoconjunctivitis, and perennial rhinoconjunctivitis at 30, 60, and 72 months of age.
“The underlying immune mechanisms associated with tolerance to peanut do not alter the natural history of allergic disease,” the researchers concluded. “Different prevention strategies or strategies that include multiple dietary interventions need to be tested to assess whether the reduction in peanut allergy observed in the LEAP consumption group can be extended to other common food allergens and allergic diseases.”
SOURCE: Du Toit G et al. J Allergy Clin Immunol. 2018 Apr;141[4]1343-53.
FROM THE JOURNAL OF ALLERGY AND CLINICAL IMMUNOLOGY
Debunking Atopic Dermatitis Myths: Do Most Children Outgrow Atopic Dermatitis?
Myth: Children eventually outgrow atopic dermatitis and therefore do not need treatment
The negative impact of atopic dermatitis (AD) on quality of life in the pediatric population often prompts parents/guardians to inquire about whether a child with AD will ever outgrow their disease. If remission is expected as the child gets older, many may question if it is necessary to pursue treatment or just let the disease run its course. Although AD often is reported to resolve soon after the first decade of life, symptoms can persist well into the second decade and beyond, suggesting that AD may be a lifelong disease with periods of waxing and waning symptoms that require persistent treatment throughout the patient’s life.
A 2014 study included 7157 children with AD (mean age of disease onset, 1.7 years) who were enrolled in the Pediatric Eczema Elective Registry (PEER) program between the ages of 2 and 17 years with measurement of disease activity at regular 6-month intervals for up to 5 years. The study results indicated that more than 80% of patients at every age (age range, 2–26 years) had symptoms of AD and/or were using medication to treat their disease, and the majority (64%) of patients had never reported a 6-month period during which they achieved clearance of symptoms without medication. At the age of 20 years, 50% of patients reported at least 1 lifetime 6-month period during which they were both symptom and treatment free. In another study of adolescents with AD who also had AD in childhood (N=82), 48% of patients remained in the same AD severity grades and 13% deteriorated from childhood to adolescence; only 39% of patients showed improvement in disease severity from childhood to adolescence. The findings of these reports are contradictory to conventional clinical teaching, which indicates that AD generally resolves by age 12 in 50% to 70% of children.
Even though some children with AD may experience periods of disease clearance, these findings often do not persist and should not be confused with a permanent remission. Most patients require continued treatment with medications to achieve relief of symptoms. Therefore, physicians should not assure parents/guardians that a child can outgrow AD; rather, they should educate pediatric patients and their caregivers about the potentially lifelong disease course and encourage early intervention to mitigate symptoms and manage comorbidities as the patient ages.
Hon KL, Tsang YCK, Poon TCW, et al. Predicating eczema severity beyond childhood. World J Pediatr. 2016;12:44-48.
Margolis JS, Abuabara K, Bilker W, et al. Persistence of mild to moderate atopic dermatitis [published online April 2, 2014]. JAMA Dermatol. 2014;150:593-600.
Myth: Children eventually outgrow atopic dermatitis and therefore do not need treatment
The negative impact of atopic dermatitis (AD) on quality of life in the pediatric population often prompts parents/guardians to inquire about whether a child with AD will ever outgrow their disease. If remission is expected as the child gets older, many may question if it is necessary to pursue treatment or just let the disease run its course. Although AD often is reported to resolve soon after the first decade of life, symptoms can persist well into the second decade and beyond, suggesting that AD may be a lifelong disease with periods of waxing and waning symptoms that require persistent treatment throughout the patient’s life.
A 2014 study included 7157 children with AD (mean age of disease onset, 1.7 years) who were enrolled in the Pediatric Eczema Elective Registry (PEER) program between the ages of 2 and 17 years with measurement of disease activity at regular 6-month intervals for up to 5 years. The study results indicated that more than 80% of patients at every age (age range, 2–26 years) had symptoms of AD and/or were using medication to treat their disease, and the majority (64%) of patients had never reported a 6-month period during which they achieved clearance of symptoms without medication. At the age of 20 years, 50% of patients reported at least 1 lifetime 6-month period during which they were both symptom and treatment free. In another study of adolescents with AD who also had AD in childhood (N=82), 48% of patients remained in the same AD severity grades and 13% deteriorated from childhood to adolescence; only 39% of patients showed improvement in disease severity from childhood to adolescence. The findings of these reports are contradictory to conventional clinical teaching, which indicates that AD generally resolves by age 12 in 50% to 70% of children.
Even though some children with AD may experience periods of disease clearance, these findings often do not persist and should not be confused with a permanent remission. Most patients require continued treatment with medications to achieve relief of symptoms. Therefore, physicians should not assure parents/guardians that a child can outgrow AD; rather, they should educate pediatric patients and their caregivers about the potentially lifelong disease course and encourage early intervention to mitigate symptoms and manage comorbidities as the patient ages.
Myth: Children eventually outgrow atopic dermatitis and therefore do not need treatment
The negative impact of atopic dermatitis (AD) on quality of life in the pediatric population often prompts parents/guardians to inquire about whether a child with AD will ever outgrow their disease. If remission is expected as the child gets older, many may question if it is necessary to pursue treatment or just let the disease run its course. Although AD often is reported to resolve soon after the first decade of life, symptoms can persist well into the second decade and beyond, suggesting that AD may be a lifelong disease with periods of waxing and waning symptoms that require persistent treatment throughout the patient’s life.
A 2014 study included 7157 children with AD (mean age of disease onset, 1.7 years) who were enrolled in the Pediatric Eczema Elective Registry (PEER) program between the ages of 2 and 17 years with measurement of disease activity at regular 6-month intervals for up to 5 years. The study results indicated that more than 80% of patients at every age (age range, 2–26 years) had symptoms of AD and/or were using medication to treat their disease, and the majority (64%) of patients had never reported a 6-month period during which they achieved clearance of symptoms without medication. At the age of 20 years, 50% of patients reported at least 1 lifetime 6-month period during which they were both symptom and treatment free. In another study of adolescents with AD who also had AD in childhood (N=82), 48% of patients remained in the same AD severity grades and 13% deteriorated from childhood to adolescence; only 39% of patients showed improvement in disease severity from childhood to adolescence. The findings of these reports are contradictory to conventional clinical teaching, which indicates that AD generally resolves by age 12 in 50% to 70% of children.
Even though some children with AD may experience periods of disease clearance, these findings often do not persist and should not be confused with a permanent remission. Most patients require continued treatment with medications to achieve relief of symptoms. Therefore, physicians should not assure parents/guardians that a child can outgrow AD; rather, they should educate pediatric patients and their caregivers about the potentially lifelong disease course and encourage early intervention to mitigate symptoms and manage comorbidities as the patient ages.
Hon KL, Tsang YCK, Poon TCW, et al. Predicating eczema severity beyond childhood. World J Pediatr. 2016;12:44-48.
Margolis JS, Abuabara K, Bilker W, et al. Persistence of mild to moderate atopic dermatitis [published online April 2, 2014]. JAMA Dermatol. 2014;150:593-600.
Hon KL, Tsang YCK, Poon TCW, et al. Predicating eczema severity beyond childhood. World J Pediatr. 2016;12:44-48.
Margolis JS, Abuabara K, Bilker W, et al. Persistence of mild to moderate atopic dermatitis [published online April 2, 2014]. JAMA Dermatol. 2014;150:593-600.
Swamp coolers not linked to dust mite sensitization in atopic children
ORLANDO – Swamp coolers – a low-cost alternative to air-conditioning in dry regions – weren’t found to increase sensitization to house dust mites or mold in atopic pediatric patients, researchers reported.
Neema Izadi, MD, and his associates say the findings, seen in a pediatric Colorado population in a study evaluating data over 10 years, could mean that not everyone at risk of dust mite and mold sensitization needs to avoid these cooling systems.
“Evaporative coolers have been shown to raise relative humidity by about 10%,” said Dr. Izadi, a pediatric allergy and immunology fellow at National Jewish Health, Denver, presenting at the joint congress of the American Academy of Asthma, Allergy and Immunology and the World Asthma Organization. “They work best in environments where the air is very warm and dry.”
House dust mites and mold thrive in higher humidity. Small studies performed in Colorado, Utah, and other locations have shown that the swamp coolers increase house dust mite allergen content, but there have been very few studies that have looked at actual sensitization. One smaller study in Nevada did find that the coolers increased sensitization to dust mites and mold.
In this study – thought to be the largest ever to look at this question – Dr. Izadi and his colleagues assessed data on patients aged 21 years and younger who were seen at National Jewish Health during 2008-2017 and who had at least one positive environmental skin-prick test. The average age was about 9 years. The cohort included 8,503 patients with sensitization to house dust mites and 9,286 with sensitization to mold. Researchers examined data on swamp coolers in their homes.
The researchers found that 29% of those with swamp coolers were dust-mite positive on skin testing, and 28% of those without one were positive. This was not a significant difference (P = .85). They found that 45% of those with the coolers were positive for sensitization to any mold, compared with 44% without one – also not a significant difference (P = .43).
They also found no difference according to age group, sex, or individually for atopic dermatitis, asthma, or allergic rhinitis.
He acknowledged that the study had no way to reliably account for patients who were transplants to Colorado, having moved there from somewhere else. The study also didn’t examine the age of homes, whether it had carpeting, or other factors.
He noted that the amount of time the coolers were run in the home was not examined and that “it might matter how much it is on.” This, he said, might account for differences in these results, compared with the Nevada study that did find a sensitization increase cause by the coolers.
“Evaporative coolers or swamp coolers are a great low-cost alternative in semiarid and arid environments – they can cut costs from 15%-35%,” Dr. Izadi said. “These data may indicate that it may be unnecessary to recommend that patients remove their swamp cooler, at least from a dust-mite and mold-sensitization standpoint.”
Dr. Izadi had no relevant financial disclosures.
SOURCE: Izadi N et al. AAAAI/WAO Joint Congress, Abstract 586
Susan Millard, MD, FCCP, comments: Swamp coolers are used in semi-arid and arid climates like Arizona, where I did my fellowship training but they didn't work well to keep apartments and homes cool enough if over about 100°F outside! The system is cheaper than air conditioning. So it is great to know that this type of cooling system does not cause more mold and dust mite allergies.
Susan Millard, MD, FCCP, comments: Swamp coolers are used in semi-arid and arid climates like Arizona, where I did my fellowship training but they didn't work well to keep apartments and homes cool enough if over about 100°F outside! The system is cheaper than air conditioning. So it is great to know that this type of cooling system does not cause more mold and dust mite allergies.
Susan Millard, MD, FCCP, comments: Swamp coolers are used in semi-arid and arid climates like Arizona, where I did my fellowship training but they didn't work well to keep apartments and homes cool enough if over about 100°F outside! The system is cheaper than air conditioning. So it is great to know that this type of cooling system does not cause more mold and dust mite allergies.
ORLANDO – Swamp coolers – a low-cost alternative to air-conditioning in dry regions – weren’t found to increase sensitization to house dust mites or mold in atopic pediatric patients, researchers reported.
Neema Izadi, MD, and his associates say the findings, seen in a pediatric Colorado population in a study evaluating data over 10 years, could mean that not everyone at risk of dust mite and mold sensitization needs to avoid these cooling systems.
“Evaporative coolers have been shown to raise relative humidity by about 10%,” said Dr. Izadi, a pediatric allergy and immunology fellow at National Jewish Health, Denver, presenting at the joint congress of the American Academy of Asthma, Allergy and Immunology and the World Asthma Organization. “They work best in environments where the air is very warm and dry.”
House dust mites and mold thrive in higher humidity. Small studies performed in Colorado, Utah, and other locations have shown that the swamp coolers increase house dust mite allergen content, but there have been very few studies that have looked at actual sensitization. One smaller study in Nevada did find that the coolers increased sensitization to dust mites and mold.
In this study – thought to be the largest ever to look at this question – Dr. Izadi and his colleagues assessed data on patients aged 21 years and younger who were seen at National Jewish Health during 2008-2017 and who had at least one positive environmental skin-prick test. The average age was about 9 years. The cohort included 8,503 patients with sensitization to house dust mites and 9,286 with sensitization to mold. Researchers examined data on swamp coolers in their homes.
The researchers found that 29% of those with swamp coolers were dust-mite positive on skin testing, and 28% of those without one were positive. This was not a significant difference (P = .85). They found that 45% of those with the coolers were positive for sensitization to any mold, compared with 44% without one – also not a significant difference (P = .43).
They also found no difference according to age group, sex, or individually for atopic dermatitis, asthma, or allergic rhinitis.
He acknowledged that the study had no way to reliably account for patients who were transplants to Colorado, having moved there from somewhere else. The study also didn’t examine the age of homes, whether it had carpeting, or other factors.
He noted that the amount of time the coolers were run in the home was not examined and that “it might matter how much it is on.” This, he said, might account for differences in these results, compared with the Nevada study that did find a sensitization increase cause by the coolers.
“Evaporative coolers or swamp coolers are a great low-cost alternative in semiarid and arid environments – they can cut costs from 15%-35%,” Dr. Izadi said. “These data may indicate that it may be unnecessary to recommend that patients remove their swamp cooler, at least from a dust-mite and mold-sensitization standpoint.”
Dr. Izadi had no relevant financial disclosures.
SOURCE: Izadi N et al. AAAAI/WAO Joint Congress, Abstract 586
ORLANDO – Swamp coolers – a low-cost alternative to air-conditioning in dry regions – weren’t found to increase sensitization to house dust mites or mold in atopic pediatric patients, researchers reported.
Neema Izadi, MD, and his associates say the findings, seen in a pediatric Colorado population in a study evaluating data over 10 years, could mean that not everyone at risk of dust mite and mold sensitization needs to avoid these cooling systems.
“Evaporative coolers have been shown to raise relative humidity by about 10%,” said Dr. Izadi, a pediatric allergy and immunology fellow at National Jewish Health, Denver, presenting at the joint congress of the American Academy of Asthma, Allergy and Immunology and the World Asthma Organization. “They work best in environments where the air is very warm and dry.”
House dust mites and mold thrive in higher humidity. Small studies performed in Colorado, Utah, and other locations have shown that the swamp coolers increase house dust mite allergen content, but there have been very few studies that have looked at actual sensitization. One smaller study in Nevada did find that the coolers increased sensitization to dust mites and mold.
In this study – thought to be the largest ever to look at this question – Dr. Izadi and his colleagues assessed data on patients aged 21 years and younger who were seen at National Jewish Health during 2008-2017 and who had at least one positive environmental skin-prick test. The average age was about 9 years. The cohort included 8,503 patients with sensitization to house dust mites and 9,286 with sensitization to mold. Researchers examined data on swamp coolers in their homes.
The researchers found that 29% of those with swamp coolers were dust-mite positive on skin testing, and 28% of those without one were positive. This was not a significant difference (P = .85). They found that 45% of those with the coolers were positive for sensitization to any mold, compared with 44% without one – also not a significant difference (P = .43).
They also found no difference according to age group, sex, or individually for atopic dermatitis, asthma, or allergic rhinitis.
He acknowledged that the study had no way to reliably account for patients who were transplants to Colorado, having moved there from somewhere else. The study also didn’t examine the age of homes, whether it had carpeting, or other factors.
He noted that the amount of time the coolers were run in the home was not examined and that “it might matter how much it is on.” This, he said, might account for differences in these results, compared with the Nevada study that did find a sensitization increase cause by the coolers.
“Evaporative coolers or swamp coolers are a great low-cost alternative in semiarid and arid environments – they can cut costs from 15%-35%,” Dr. Izadi said. “These data may indicate that it may be unnecessary to recommend that patients remove their swamp cooler, at least from a dust-mite and mold-sensitization standpoint.”
Dr. Izadi had no relevant financial disclosures.
SOURCE: Izadi N et al. AAAAI/WAO Joint Congress, Abstract 586
REPORTING FROM AAAAI/WAO JOINT CONGRESS
Key clinical point:
Major finding: Researchers found that 29% of those with swamp coolers were dust-mite positive on skin testing, and 28% of those without one were as well. This was not a significant difference (P = .85).
Study details: A retrospective review of more than 17,000 cases of atopic children aged 21 years and younger who were seen at National Jewish Health and had a positive environmental skin prick test.
Disclosures: Dr. Izadi had no relevant financial disclosures.
Source: Izadi N et al. AAAAI/WAO Joint Congress, Abstract 586