User login
Debunking Atopic Dermatitis Myths: Can Adults Develop Eczema?
Myth: Atopic Dermatitis Does Not Start in Adulthood
Atopic dermatitis (AD) typically first appears in childhood and tends to disappear before puberty begins; however, some patients experience AD that persists into adulthood or occurs de novo. Bannister and Freeman coined the term adult-onset atopic dermatitis after reviewing 2604 cases of AD and noting that 243 patients (9%) were first diagnosed with AD at 20 years of age or older. Adult-onset AD may be its own subset of AD or childhood AD that was simply not diagnosed until adulthood or was forgotten by the patient.
Characteristically, AD presents in adults as inflammatory eczema with areas of lichenification. It could occur after a change in residence to a cold dry climate or exposure to central heating, as patients who grew up in warm, sunny, humid climates might not have had diagnosable AD in childhood or adolescence. The more common forms of adult-onset AD are hand and neck dermatitis, hand eczema, nummular eczema, or prurigo, while childhood AD often manifests in a flexural distribution. Because it is difficult to detect, adult-onset AD is diagnosed after ruling out other diseases. Diagnostic procedures, such as patch tests, skin prick tests, biopsies, or blood screenings, usually are necessary to rule out other diseases or types of eczema. Contact eczema is the first diagnostic sign of AD in adults.
Maintaining AD in the differential diagnosis for patients with clinical symptoms of pruritus and eczema is essential due to the quality of life impact of the condition. Sleep disturbance is common in adults with severe AD and treatment may help to improve sleep quality.
Hanifin suggested the following when assessing adults for AD:
- Verify diagnosis (not allergic contact dermatitis or psoriasis)
- Determine patient's history of allergies (eg, food allergy) or childhood eczema
- Obtain family history of eczema/allergies
- Evaluate if patient's occupation may impact condition (eg, contact with irritants or known contact allergens)
- Inquire about patient's childhood residence (eg, tropical climate)
Adult-onset AD is a recalcitrant condition that can be difficult to treat, and appropriately labeling/diagnosing the condition will lead to better management.
Expert Commentary
Whereas once it was the rite of the pediatric AD patient to outgrow disease, it has now become clear that resolution is not as hard a stop in atopic disease as expected. Adult-onset and persistent disease in AD is clearly a significant problem, especially in developed nations and carries a host of comorbidites. Time and enhanced research will hopefully identify interventions to reverse the trend towards persistence into adulthood.
—Nanette B. Silverberg, MD (New York, New York)
Bannister MJ, Freeman S. Adult-onset atopic dermatitis. Australas J Dermatol. 2000;41:225-228.
Hanifin JM. Adult-onset atopic dermatitis: fact or fancy? Dermatol Clin. 2017;35:299-302.
Silvestre Salvador JF, Romero-Pérez D, Encabo-Durán B. Atopic dermatitis in adults: a diagnostic challenge. J Investig Allergol Clin Immunol. 2017;27:78-88.
Myth: Atopic Dermatitis Does Not Start in Adulthood
Atopic dermatitis (AD) typically first appears in childhood and tends to disappear before puberty begins; however, some patients experience AD that persists into adulthood or occurs de novo. Bannister and Freeman coined the term adult-onset atopic dermatitis after reviewing 2604 cases of AD and noting that 243 patients (9%) were first diagnosed with AD at 20 years of age or older. Adult-onset AD may be its own subset of AD or childhood AD that was simply not diagnosed until adulthood or was forgotten by the patient.
Characteristically, AD presents in adults as inflammatory eczema with areas of lichenification. It could occur after a change in residence to a cold dry climate or exposure to central heating, as patients who grew up in warm, sunny, humid climates might not have had diagnosable AD in childhood or adolescence. The more common forms of adult-onset AD are hand and neck dermatitis, hand eczema, nummular eczema, or prurigo, while childhood AD often manifests in a flexural distribution. Because it is difficult to detect, adult-onset AD is diagnosed after ruling out other diseases. Diagnostic procedures, such as patch tests, skin prick tests, biopsies, or blood screenings, usually are necessary to rule out other diseases or types of eczema. Contact eczema is the first diagnostic sign of AD in adults.
Maintaining AD in the differential diagnosis for patients with clinical symptoms of pruritus and eczema is essential due to the quality of life impact of the condition. Sleep disturbance is common in adults with severe AD and treatment may help to improve sleep quality.
Hanifin suggested the following when assessing adults for AD:
- Verify diagnosis (not allergic contact dermatitis or psoriasis)
- Determine patient's history of allergies (eg, food allergy) or childhood eczema
- Obtain family history of eczema/allergies
- Evaluate if patient's occupation may impact condition (eg, contact with irritants or known contact allergens)
- Inquire about patient's childhood residence (eg, tropical climate)
Adult-onset AD is a recalcitrant condition that can be difficult to treat, and appropriately labeling/diagnosing the condition will lead to better management.
Expert Commentary
Whereas once it was the rite of the pediatric AD patient to outgrow disease, it has now become clear that resolution is not as hard a stop in atopic disease as expected. Adult-onset and persistent disease in AD is clearly a significant problem, especially in developed nations and carries a host of comorbidites. Time and enhanced research will hopefully identify interventions to reverse the trend towards persistence into adulthood.
—Nanette B. Silverberg, MD (New York, New York)
Myth: Atopic Dermatitis Does Not Start in Adulthood
Atopic dermatitis (AD) typically first appears in childhood and tends to disappear before puberty begins; however, some patients experience AD that persists into adulthood or occurs de novo. Bannister and Freeman coined the term adult-onset atopic dermatitis after reviewing 2604 cases of AD and noting that 243 patients (9%) were first diagnosed with AD at 20 years of age or older. Adult-onset AD may be its own subset of AD or childhood AD that was simply not diagnosed until adulthood or was forgotten by the patient.
Characteristically, AD presents in adults as inflammatory eczema with areas of lichenification. It could occur after a change in residence to a cold dry climate or exposure to central heating, as patients who grew up in warm, sunny, humid climates might not have had diagnosable AD in childhood or adolescence. The more common forms of adult-onset AD are hand and neck dermatitis, hand eczema, nummular eczema, or prurigo, while childhood AD often manifests in a flexural distribution. Because it is difficult to detect, adult-onset AD is diagnosed after ruling out other diseases. Diagnostic procedures, such as patch tests, skin prick tests, biopsies, or blood screenings, usually are necessary to rule out other diseases or types of eczema. Contact eczema is the first diagnostic sign of AD in adults.
Maintaining AD in the differential diagnosis for patients with clinical symptoms of pruritus and eczema is essential due to the quality of life impact of the condition. Sleep disturbance is common in adults with severe AD and treatment may help to improve sleep quality.
Hanifin suggested the following when assessing adults for AD:
- Verify diagnosis (not allergic contact dermatitis or psoriasis)
- Determine patient's history of allergies (eg, food allergy) or childhood eczema
- Obtain family history of eczema/allergies
- Evaluate if patient's occupation may impact condition (eg, contact with irritants or known contact allergens)
- Inquire about patient's childhood residence (eg, tropical climate)
Adult-onset AD is a recalcitrant condition that can be difficult to treat, and appropriately labeling/diagnosing the condition will lead to better management.
Expert Commentary
Whereas once it was the rite of the pediatric AD patient to outgrow disease, it has now become clear that resolution is not as hard a stop in atopic disease as expected. Adult-onset and persistent disease in AD is clearly a significant problem, especially in developed nations and carries a host of comorbidites. Time and enhanced research will hopefully identify interventions to reverse the trend towards persistence into adulthood.
—Nanette B. Silverberg, MD (New York, New York)
Bannister MJ, Freeman S. Adult-onset atopic dermatitis. Australas J Dermatol. 2000;41:225-228.
Hanifin JM. Adult-onset atopic dermatitis: fact or fancy? Dermatol Clin. 2017;35:299-302.
Silvestre Salvador JF, Romero-Pérez D, Encabo-Durán B. Atopic dermatitis in adults: a diagnostic challenge. J Investig Allergol Clin Immunol. 2017;27:78-88.
Bannister MJ, Freeman S. Adult-onset atopic dermatitis. Australas J Dermatol. 2000;41:225-228.
Hanifin JM. Adult-onset atopic dermatitis: fact or fancy? Dermatol Clin. 2017;35:299-302.
Silvestre Salvador JF, Romero-Pérez D, Encabo-Durán B. Atopic dermatitis in adults: a diagnostic challenge. J Investig Allergol Clin Immunol. 2017;27:78-88.
Diet and Dermatology: Google Search Results for Acne, Psoriasis, and Eczema
Researching medical information currently is the third most common use of the Internet in the United States,1 with the majority of adults using the Web as their first source for health information before seeing a physician.2 When assessing health-related information online, resources can be grouped into 4 categories: (1) those attributed to self-proclaimed experts, (2) promotional, (3) social media, and (4) educational.3 Access to such a wide range of sources may give readers the opportunity to share personal anecdotes and opinions, thereby serving as a forum for information that essentially cannot be validated. Although such websites may include useful information and cite current literature, in other instances health-related information may be misleading or fabricated.3
In a study evaluating 291 skin conditions and related Google trends, acne, psoriasis, and eczema were among the most burdensome diseases, with acne yielding the highest number of search results.4 Results of the study indicated a positive correlation between disease burden and online search interest.4 The impact of these online searches and the validity of Google search results are topics worth considering, as more dermatology patients are relying on holistic and nonpharmaceutical approaches to treatment and disease management.5 The purpose of this study was to evaluate content on diet and dermatology available on the Internet for acne, psoriasis, and eczema.
Methods
Google searches were performed in December 2017 using the terms diet and acne, diet and psoriasis, and diet and eczema. The first 10 results for each respective search were reviewed for recommendations about which foods to incorporate in the diet and which to avoid. They also were classified according to the following 4 website categories: (1) those attributed to self-proclaimed experts, (2) promotional, (3) social media, and (4) educational. The recommendations gathered from the 30 websites were then compared to the current literature assessing the impact of diet on these respective conditions by conducting PubMed searches of articles indexed for MEDLINE using the same terms.
Results
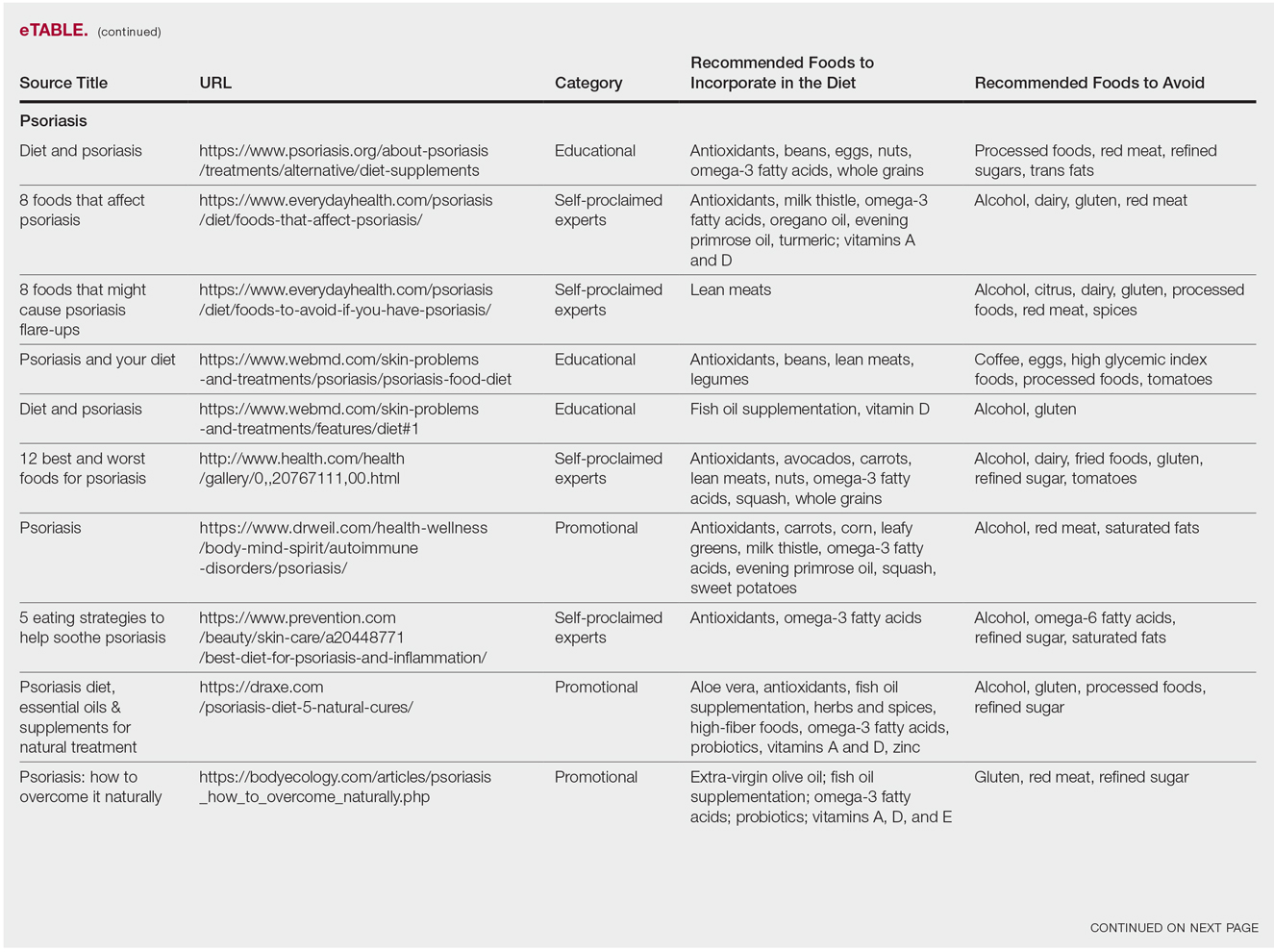
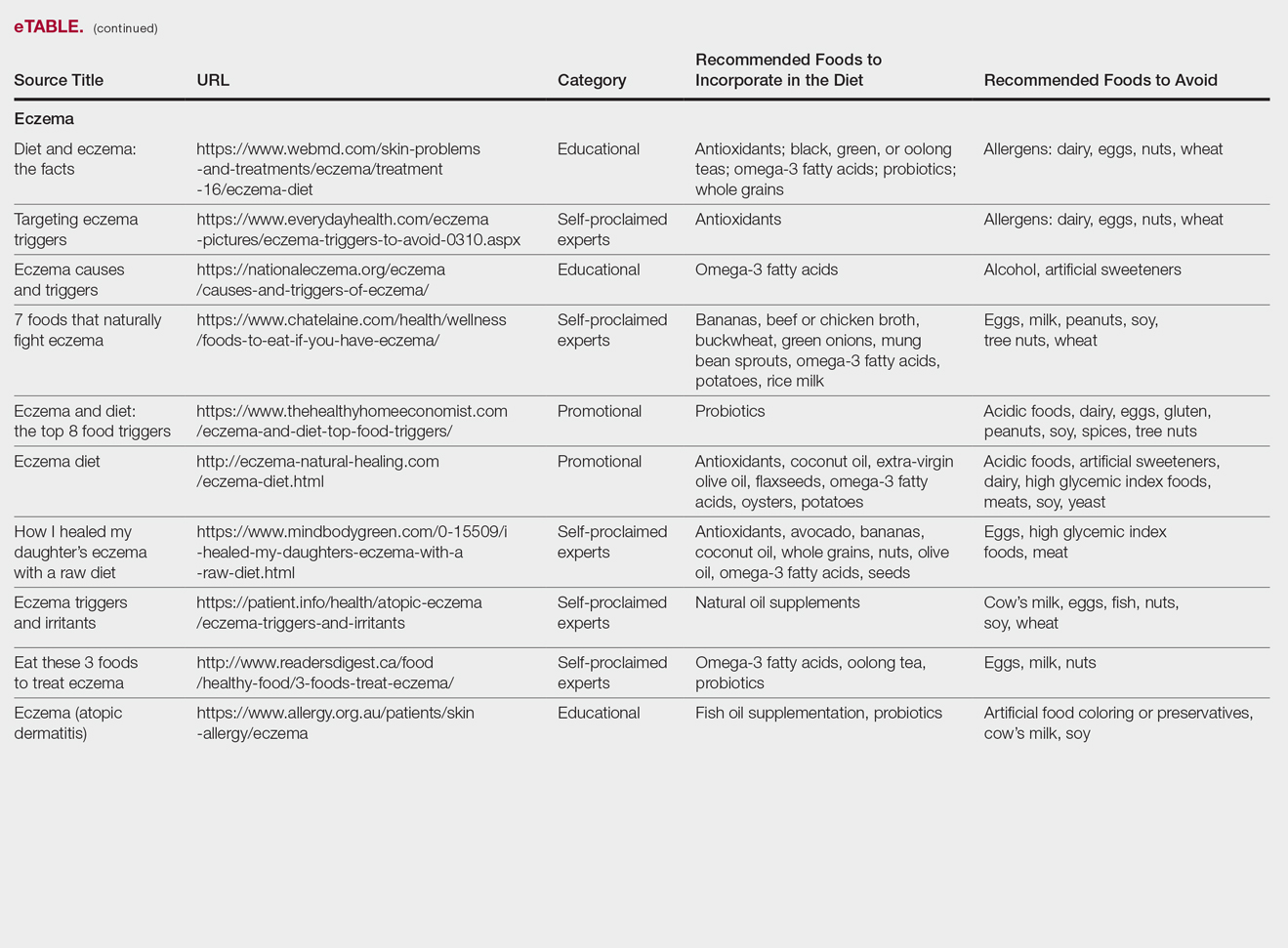
The results of this study are outlined in the eTable.

Acne
Our Google search using the term diet and acne produced 17,500,000 results. Of the first 10 search results, 40% (4/10) were websites attributed to self-proclaimed experts, 40% (4/10) were educational resources, and 20% (2/10) were promotional websites. Most of the websites advised acne patients to avoid high glycemic index foods (90% [9/10]) and dairy products (90% [9/10]). When discussing which foods to include in the diet, 70% (7/10) of websites recommended that patients incorporate omega-3 fatty acids and antioxidants in the diet.
Research has shown that a low glycemic index diet can lead to a decrease in patients’ acne lesion counts in some instances.6,7 In a case-controlled study of 2258 patients on a popular weight loss diet that emphasized low glycemic index foods, 87% of participants reported a reduction in acne and 91% reported a decrease in their dosage or number of acne medications.7 Still, the exact correlation between acne development and consumption of glycemic index foods has not been confirmed. However, high glycemic index diets have been linked to hyperinsulinemia, indicating that insulin levels may play a role in acne formation.8 The majority of other currently available studies evaluated the potential link between dairy consumption and acne. A retrospective analysis of 47,355 women spanning 12 weeks showed a positive link between increased dairy consumption, specifically skim milk, and acne formation. Despite the positive trend, limitations such as recall bias made it difficult to draw a conclusion based on these findings.9 However, results of a longitudinal questionnaire-based population study evaluating the impact of dairy consumption on acne in 2489 adolescent patients confirmed a positive correlation.10 Studies conducted in 2009 and 2011 concluded that milk consumption results in elevated insulinlike growth factor 1 levels, which were linked to comedogenesis.8,11
Currently, there are well-described mechanisms to explain the association of dairy consumption and glycemic index with acne. Confirming a correlation between acne development and dairy consumption suggests that a dairy-free diet may benefit acne patients.5 Other trials indicate that low glycemic index diets are beneficial in treating acne.6,7 Therefore, some of the recommendations made in our search results may be of merit; however, there is minimal evidence proving the benefits of the other dietary recommendations made in the websites we evaluated.
Psoriasis
Our Google search using the term diet and psoriasis yielded a total of 9,420,000 results. Of the first 10 search results, 40% (4/10) were websites attributed to self-proclaimed experts, 30% (3/10) were promotional, and 30% (3/10) were educational. Seventy percent (7/10) of websites recommended avoiding alcohol and 60% (6/10) recommended avoiding gluten, with others discouraging consumption of red meat. Most of the websites encouraged patients to consume omega-3 fatty acids and antioxidants, while a few also recommended vitamins A, D, and E, as well as evening primrose oil supplements.
Although current research indicates a positive correlation between excessive alcohol use and psoriasis severity, it is still unclear whether alcohol consumption can be directly linked to the disease.12-14 Likewise, despite belief that increased oxidative stress likely contributes to inflammation in psoriasis, there is little evidence linking antioxidants to improvement in psoriasis symptoms.12 However, the current literature is inconsistent regarding the effects of fish oil supplementation on psoriasis.12 In a randomized double-blind study of 145 patients, there was no significant difference in psoriasis area and severity index scores between a control group and a treatment group receiving fish oil supplementation.15 In another RCT of 45 participants, those given daily very long-chain omega-3 fatty acid supplements saw no difference in psoriasis symptoms.15 Despite debate, literature assessing the impact of gluten-free diets has described improvement in psoriasis lesions in patients with celiac-specific antibodies.16 Although some observational studies described vitamin D supplementation to be beneficial in the treatment of psoriatic lesions, a more recent RCT found no significant difference between control and treatment groups.17-19
Studies also have revealed that certain eating patterns, such as those associated with the Mediterranean diet that is rich in fruits, vegetables, whole grains, and omega-3 fatty acids may be linked to improved endothelial function scores and reduced C-reactive protein and IL-18levels.20,21 In a double-blind RCT of 75 patients with plaque psoriasis, mean (SD) psoriasis area and severity index scores decreased by 11.2 (9.8) in a group treated with omega-3 fatty acids compared to 7.5 (8.8) with omega-6 fatty acids (P=.048).22
Although excessive alcohol use may be linked to psoriasis, there is no conclusive evidence indicating causation, thereby discrediting online claims.12-14 Research has revealed that gluten-free diets in psoriasis patients with celiac disease may improve psoriasis treatment16; however, sufficient evidence is lacking for diets low in gluten and high in polyunsaturated fatty acids or antioxidant supplementation. Of the dietary supplements recommended in the search results we reviewed, fish oil appears to be the most promising, but no recommendations can be made based on the current research.
Eczema
Our Google search using the term diet and eczema yielded 1,160,000 results, with 50% (5/10) of websites attributed to self-proclaimed experts, 30% (3/10) to educational websites, and 20% (2/10) to promotional sites. Of the first 10 results, 80% (8/10) recommended that patients with eczema avoid milk/dairy and 50% (5/10) advised to avoid soy and wheat/gluten. Other websites indicated to avoid eggs, nuts, and artificial sweeteners. Patients were encouraged to incorporate omega-3 fatty acids in their diets, and a few sites recommended bananas, coconut oil, olive oil, and various teas.
In a review of 11 studies with a total of 596 participants, supplementation with vitamins D and E, fish oil, olive oil, and linoleic acid was evaluated for the treatment of eczema.23 Although results indicated modest improvement of eczema severity with supplementation of fish oil, evidence favoring this treatment is limited and unconvincing. Furthermore, some evidence indicates that elimination diets are only appropriate for patients with food allergies.24 In a study evaluating an egg-free and dairy-free diet for eczema patients, only participants with positive egg-specific serum IgE levels saw improvement in disease severity.23 Even though IgE-mediated food allergies have been reported in 40% of children with moderate eczema, the contribution of these allergies to eczema is questionable.25
There is little evidence in the literature to indicate a definitive correlation between the foods mentioned in the search results we evaluated and the development of eczema; however, for patients with food allergies and eczema, elimination diets may decrease disease severity.25,26 There is insufficient evidence to suggest a benefit from evening primrose oil or fish oil supplementation, thereby debunking claims found online.
Comment
Although our Google search results included a wide range of sources and information regarding diet and dermatologic conditions such as acne, psoriasis, and eczema, most of the information we found was either unfounded or misleading. Study limitations in the current literature include small sample size, potential recall bias, lack of appropriate controls, incomplete reported results, and the failure to clearly define skin changes.
When considering the accuracy and type of information regarding skin conditions that is available on the Internet, it is important to note that most of the results we reviewed were webpages attributed to self-proclaimed experts. Although educational websites also were included in the search results, whether or not patients prefer or understand the content of such websites is still unknown; therefore, health organizations should consider revising online patient education materials to allow universal comprehension.27
Furthermore, it is important to consider the impact that widespread Internet access may have on the physician-patient relationship. Having access to health-related information online and being able to potentially self-diagnose could delay or deter patients from seeking professional advice or care.3 A study evaluating the impact of online searches on the physician-patient relationship among 175 patients determined that 36.5% of patients gathered information online prior to their consultation with a physician, while 67.3% chose to complement the information given to them by their physician with online resources.28 Based on these statistics, it is important that physicians be up-to-date with Internet discourse to discredit unfounded recommendations. Ultimately, when it comes to diet and dermatology, patients ought to be skeptical of the information currently available on the Internet, given that most of it is unsubstantiated by medical research.
- Fox S. Online health search 2006. Pew Research Center website. http://www.pewinternet.org/2006/10/29/online-health-search-2006/. Published October 29, 2006. Accessed May 3, 2018.
- Prestin A, Vieux SN, Chou WY. Is online health activity alive and well or flatlining? findings from 10 years of the health information national trends survey. J Health Commun. 2015;20:790-798.
- Zeichner JA, Del Rosso JQ. Acne and the internet. Dermatol Clin. 2016;34:129-132.
- Whitsitt J, Karimkhani C, Boyers LN, et al. Comparing burden of dermatologic disease to search interest on Google trends. Dermatol Online J. 2015;21. pii:13030/qt5xg811qp.
- Shokeen D. Influence of diet in acne vulgaris and atopic dermatitis. Cutis. 2016;98:E28-E29.
- Veith WB, Silverberg NB. The association of acne vulgaris with diet. Cutis. 2011;88:84-91.
- Rouhani P. Acne improves with a popular, low glycemic diet from South Beach. J Am Acad Dermatol. 2009;60(3, suppl 1):P706.
- Melnick BC. Evidence for acne-promoting effect of milk and other insulinotropic dairy products. Nestle Nutr Worksop Ser Pediatr Program. 2011;67:131-145.
- Adebamowo CA, Spiegelman D, Berkey CS, et al. High school dietary diary intake and teenage acne. J Am Acad Dermatol. 2005;52:207-214.
- Ulvestad M, Bjertness E, Dalgard F, et al. Acne and dairy products in adolescence: results from a Norwegian longitudinal study [published online July 16, 2016]. J Eur Acad Dermatol Venereol. 2017;31:530-535.
- Melnick BC, Schmitz G. Role of insulin, insulin like growth factor 1, hyperglycemic food and milk consumption in the pathogenesis of acne vulgaris. Exp Dermatol. 2009;18:833-841.
- Murzaku EC, Bronsnick T, Rao BK. Diet in dermatology: part II. melanoma, chronic urticaria, and psoriasis. J Am Acad Dermatol. 2014;71:1053.E1-1053.E16.
- Tobin AM, Higgins EM, Norris S, et al. Prevalence of psoriasis in patients with alcoholic liver disease. Clin Exp Dermatol. 2009;34:698-701.
- Kirby B, Richards HL, Mason DL, et al. Alcohol consumption and psychological distress in patients with psoriasis. Br J Dermatol. 2008;158:138-140.
- Søyland E, Funk J, Rajika G, et al. Effect of dietary supplementation with very long-chain n-3 fatty acids in patients with psoriasis. N Engl J Med. 1993;328:1812-1816.
- Michaëlsson G, Gerdén B, Hagforsen E, et al. Psoriasis patients with antibodies to gliadin can be improved by a gluten-free diet. Br J Dermatol. 2000;142:44-51.
- Morimoto S, Yoshikawa K. Psoriasis and vitamin D3. a review of our experience. Arch Dermatol. 1989;125:231-234.
- Smith EL, Pincus SH, Donovan L, et al. A novel approach for the evaluation and treatment of psoriasis. oral or topical use of 1,25-dihydroxyvitamin D3 can be a safe and effective therapy for psoriasis. J Am Acad Dermatol. 1988;19:516-528.
- Siddiqui MA, Al-Khawajah MM. Vitamin D3 and psoriasis: a randomized double-blind placebo-controlled study. J Dermatol Treat. 1990;1:243-245.
- Wang Y, Gao H, Loyd CM, et al. Chronic skin-specific inflammation promotes vascular inflammation and thrombosis. J Invest Dermatol. 2012;132:2067-2075.
- Barrea L, Nappi F, Di Somma C, et al. Environmental risk factors in psoriasis: the point of view of the nutritionist. Int J Environ Res Public Health. 2016;13. pii:E743. doi:10.3390/ijerph13070743.
- Mayser P, Mrowietz U, Arenberger P, et al. Omega-3 fatty acid-based lipid infusion in patients with chronic plaque psoriasis: results of a double-blind, randomized, placebo-controlled, multicenter trial. J Am Acad Dermatol. 1998;38:539-547.
- Bath-Hextall FJ, Jenkinson C, Humphreys R, et al. Dietary supplements for established atopic eczema. Cochrane Database Syst Rev. 2012;2:CD005205.
- Bronsnick T, Murzaku EC, Rao BK. Diet in dermatology: part I. atopic dermatitis, acne, and nonmelanoma skin cancer [published online November 15, 2014]. J Am Acad Dermatol. 2014;71:1039.E1-1039.E12.
- Campbell DE. The role of food allergy in childhood atopic dermatitis. J Paediatr Child Health. 2012;48:1058-1064.
- Werfel T, Erdmann S, Fuchs T, et al. Approach to suspected food allergy in atopic dermatitis. guideline of the Task Force on Food Allergy of the German Society of Allergology and Clinical Immunology (DGAKI) and the Medical Association of German Allergologists (ADA) and the German Society of Pediatric Allergology (GPA). J Dtsch Dermatol Ges. 2009;3:265-271.
- John AM, John ES, Hansberry DR, et al. Assessment of online patient education materials from major dermatologic associations. J Clin Aesthet Dermatol. 2016;9:23-28.
- Orgaz-Molina J, Cotugno M, Girón-Prieto MS, et al. A study of internet searches for medical information in dermatology patients: the patient-physician relationship. Actas Dermosifiliogr. 2015;106:493-499.
Researching medical information currently is the third most common use of the Internet in the United States,1 with the majority of adults using the Web as their first source for health information before seeing a physician.2 When assessing health-related information online, resources can be grouped into 4 categories: (1) those attributed to self-proclaimed experts, (2) promotional, (3) social media, and (4) educational.3 Access to such a wide range of sources may give readers the opportunity to share personal anecdotes and opinions, thereby serving as a forum for information that essentially cannot be validated. Although such websites may include useful information and cite current literature, in other instances health-related information may be misleading or fabricated.3
In a study evaluating 291 skin conditions and related Google trends, acne, psoriasis, and eczema were among the most burdensome diseases, with acne yielding the highest number of search results.4 Results of the study indicated a positive correlation between disease burden and online search interest.4 The impact of these online searches and the validity of Google search results are topics worth considering, as more dermatology patients are relying on holistic and nonpharmaceutical approaches to treatment and disease management.5 The purpose of this study was to evaluate content on diet and dermatology available on the Internet for acne, psoriasis, and eczema.
Methods
Google searches were performed in December 2017 using the terms diet and acne, diet and psoriasis, and diet and eczema. The first 10 results for each respective search were reviewed for recommendations about which foods to incorporate in the diet and which to avoid. They also were classified according to the following 4 website categories: (1) those attributed to self-proclaimed experts, (2) promotional, (3) social media, and (4) educational. The recommendations gathered from the 30 websites were then compared to the current literature assessing the impact of diet on these respective conditions by conducting PubMed searches of articles indexed for MEDLINE using the same terms.
Results
The results of this study are outlined in the eTable.



Acne
Our Google search using the term diet and acne produced 17,500,000 results. Of the first 10 search results, 40% (4/10) were websites attributed to self-proclaimed experts, 40% (4/10) were educational resources, and 20% (2/10) were promotional websites. Most of the websites advised acne patients to avoid high glycemic index foods (90% [9/10]) and dairy products (90% [9/10]). When discussing which foods to include in the diet, 70% (7/10) of websites recommended that patients incorporate omega-3 fatty acids and antioxidants in the diet.
Research has shown that a low glycemic index diet can lead to a decrease in patients’ acne lesion counts in some instances.6,7 In a case-controlled study of 2258 patients on a popular weight loss diet that emphasized low glycemic index foods, 87% of participants reported a reduction in acne and 91% reported a decrease in their dosage or number of acne medications.7 Still, the exact correlation between acne development and consumption of glycemic index foods has not been confirmed. However, high glycemic index diets have been linked to hyperinsulinemia, indicating that insulin levels may play a role in acne formation.8 The majority of other currently available studies evaluated the potential link between dairy consumption and acne. A retrospective analysis of 47,355 women spanning 12 weeks showed a positive link between increased dairy consumption, specifically skim milk, and acne formation. Despite the positive trend, limitations such as recall bias made it difficult to draw a conclusion based on these findings.9 However, results of a longitudinal questionnaire-based population study evaluating the impact of dairy consumption on acne in 2489 adolescent patients confirmed a positive correlation.10 Studies conducted in 2009 and 2011 concluded that milk consumption results in elevated insulinlike growth factor 1 levels, which were linked to comedogenesis.8,11
Currently, there are well-described mechanisms to explain the association of dairy consumption and glycemic index with acne. Confirming a correlation between acne development and dairy consumption suggests that a dairy-free diet may benefit acne patients.5 Other trials indicate that low glycemic index diets are beneficial in treating acne.6,7 Therefore, some of the recommendations made in our search results may be of merit; however, there is minimal evidence proving the benefits of the other dietary recommendations made in the websites we evaluated.
Psoriasis
Our Google search using the term diet and psoriasis yielded a total of 9,420,000 results. Of the first 10 search results, 40% (4/10) were websites attributed to self-proclaimed experts, 30% (3/10) were promotional, and 30% (3/10) were educational. Seventy percent (7/10) of websites recommended avoiding alcohol and 60% (6/10) recommended avoiding gluten, with others discouraging consumption of red meat. Most of the websites encouraged patients to consume omega-3 fatty acids and antioxidants, while a few also recommended vitamins A, D, and E, as well as evening primrose oil supplements.
Although current research indicates a positive correlation between excessive alcohol use and psoriasis severity, it is still unclear whether alcohol consumption can be directly linked to the disease.12-14 Likewise, despite belief that increased oxidative stress likely contributes to inflammation in psoriasis, there is little evidence linking antioxidants to improvement in psoriasis symptoms.12 However, the current literature is inconsistent regarding the effects of fish oil supplementation on psoriasis.12 In a randomized double-blind study of 145 patients, there was no significant difference in psoriasis area and severity index scores between a control group and a treatment group receiving fish oil supplementation.15 In another RCT of 45 participants, those given daily very long-chain omega-3 fatty acid supplements saw no difference in psoriasis symptoms.15 Despite debate, literature assessing the impact of gluten-free diets has described improvement in psoriasis lesions in patients with celiac-specific antibodies.16 Although some observational studies described vitamin D supplementation to be beneficial in the treatment of psoriatic lesions, a more recent RCT found no significant difference between control and treatment groups.17-19
Studies also have revealed that certain eating patterns, such as those associated with the Mediterranean diet that is rich in fruits, vegetables, whole grains, and omega-3 fatty acids may be linked to improved endothelial function scores and reduced C-reactive protein and IL-18levels.20,21 In a double-blind RCT of 75 patients with plaque psoriasis, mean (SD) psoriasis area and severity index scores decreased by 11.2 (9.8) in a group treated with omega-3 fatty acids compared to 7.5 (8.8) with omega-6 fatty acids (P=.048).22
Although excessive alcohol use may be linked to psoriasis, there is no conclusive evidence indicating causation, thereby discrediting online claims.12-14 Research has revealed that gluten-free diets in psoriasis patients with celiac disease may improve psoriasis treatment16; however, sufficient evidence is lacking for diets low in gluten and high in polyunsaturated fatty acids or antioxidant supplementation. Of the dietary supplements recommended in the search results we reviewed, fish oil appears to be the most promising, but no recommendations can be made based on the current research.
Eczema
Our Google search using the term diet and eczema yielded 1,160,000 results, with 50% (5/10) of websites attributed to self-proclaimed experts, 30% (3/10) to educational websites, and 20% (2/10) to promotional sites. Of the first 10 results, 80% (8/10) recommended that patients with eczema avoid milk/dairy and 50% (5/10) advised to avoid soy and wheat/gluten. Other websites indicated to avoid eggs, nuts, and artificial sweeteners. Patients were encouraged to incorporate omega-3 fatty acids in their diets, and a few sites recommended bananas, coconut oil, olive oil, and various teas.
In a review of 11 studies with a total of 596 participants, supplementation with vitamins D and E, fish oil, olive oil, and linoleic acid was evaluated for the treatment of eczema.23 Although results indicated modest improvement of eczema severity with supplementation of fish oil, evidence favoring this treatment is limited and unconvincing. Furthermore, some evidence indicates that elimination diets are only appropriate for patients with food allergies.24 In a study evaluating an egg-free and dairy-free diet for eczema patients, only participants with positive egg-specific serum IgE levels saw improvement in disease severity.23 Even though IgE-mediated food allergies have been reported in 40% of children with moderate eczema, the contribution of these allergies to eczema is questionable.25
There is little evidence in the literature to indicate a definitive correlation between the foods mentioned in the search results we evaluated and the development of eczema; however, for patients with food allergies and eczema, elimination diets may decrease disease severity.25,26 There is insufficient evidence to suggest a benefit from evening primrose oil or fish oil supplementation, thereby debunking claims found online.
Comment
Although our Google search results included a wide range of sources and information regarding diet and dermatologic conditions such as acne, psoriasis, and eczema, most of the information we found was either unfounded or misleading. Study limitations in the current literature include small sample size, potential recall bias, lack of appropriate controls, incomplete reported results, and the failure to clearly define skin changes.
When considering the accuracy and type of information regarding skin conditions that is available on the Internet, it is important to note that most of the results we reviewed were webpages attributed to self-proclaimed experts. Although educational websites also were included in the search results, whether or not patients prefer or understand the content of such websites is still unknown; therefore, health organizations should consider revising online patient education materials to allow universal comprehension.27
Furthermore, it is important to consider the impact that widespread Internet access may have on the physician-patient relationship. Having access to health-related information online and being able to potentially self-diagnose could delay or deter patients from seeking professional advice or care.3 A study evaluating the impact of online searches on the physician-patient relationship among 175 patients determined that 36.5% of patients gathered information online prior to their consultation with a physician, while 67.3% chose to complement the information given to them by their physician with online resources.28 Based on these statistics, it is important that physicians be up-to-date with Internet discourse to discredit unfounded recommendations. Ultimately, when it comes to diet and dermatology, patients ought to be skeptical of the information currently available on the Internet, given that most of it is unsubstantiated by medical research.
Researching medical information currently is the third most common use of the Internet in the United States,1 with the majority of adults using the Web as their first source for health information before seeing a physician.2 When assessing health-related information online, resources can be grouped into 4 categories: (1) those attributed to self-proclaimed experts, (2) promotional, (3) social media, and (4) educational.3 Access to such a wide range of sources may give readers the opportunity to share personal anecdotes and opinions, thereby serving as a forum for information that essentially cannot be validated. Although such websites may include useful information and cite current literature, in other instances health-related information may be misleading or fabricated.3
In a study evaluating 291 skin conditions and related Google trends, acne, psoriasis, and eczema were among the most burdensome diseases, with acne yielding the highest number of search results.4 Results of the study indicated a positive correlation between disease burden and online search interest.4 The impact of these online searches and the validity of Google search results are topics worth considering, as more dermatology patients are relying on holistic and nonpharmaceutical approaches to treatment and disease management.5 The purpose of this study was to evaluate content on diet and dermatology available on the Internet for acne, psoriasis, and eczema.
Methods
Google searches were performed in December 2017 using the terms diet and acne, diet and psoriasis, and diet and eczema. The first 10 results for each respective search were reviewed for recommendations about which foods to incorporate in the diet and which to avoid. They also were classified according to the following 4 website categories: (1) those attributed to self-proclaimed experts, (2) promotional, (3) social media, and (4) educational. The recommendations gathered from the 30 websites were then compared to the current literature assessing the impact of diet on these respective conditions by conducting PubMed searches of articles indexed for MEDLINE using the same terms.
Results
The results of this study are outlined in the eTable.



Acne
Our Google search using the term diet and acne produced 17,500,000 results. Of the first 10 search results, 40% (4/10) were websites attributed to self-proclaimed experts, 40% (4/10) were educational resources, and 20% (2/10) were promotional websites. Most of the websites advised acne patients to avoid high glycemic index foods (90% [9/10]) and dairy products (90% [9/10]). When discussing which foods to include in the diet, 70% (7/10) of websites recommended that patients incorporate omega-3 fatty acids and antioxidants in the diet.
Research has shown that a low glycemic index diet can lead to a decrease in patients’ acne lesion counts in some instances.6,7 In a case-controlled study of 2258 patients on a popular weight loss diet that emphasized low glycemic index foods, 87% of participants reported a reduction in acne and 91% reported a decrease in their dosage or number of acne medications.7 Still, the exact correlation between acne development and consumption of glycemic index foods has not been confirmed. However, high glycemic index diets have been linked to hyperinsulinemia, indicating that insulin levels may play a role in acne formation.8 The majority of other currently available studies evaluated the potential link between dairy consumption and acne. A retrospective analysis of 47,355 women spanning 12 weeks showed a positive link between increased dairy consumption, specifically skim milk, and acne formation. Despite the positive trend, limitations such as recall bias made it difficult to draw a conclusion based on these findings.9 However, results of a longitudinal questionnaire-based population study evaluating the impact of dairy consumption on acne in 2489 adolescent patients confirmed a positive correlation.10 Studies conducted in 2009 and 2011 concluded that milk consumption results in elevated insulinlike growth factor 1 levels, which were linked to comedogenesis.8,11
Currently, there are well-described mechanisms to explain the association of dairy consumption and glycemic index with acne. Confirming a correlation between acne development and dairy consumption suggests that a dairy-free diet may benefit acne patients.5 Other trials indicate that low glycemic index diets are beneficial in treating acne.6,7 Therefore, some of the recommendations made in our search results may be of merit; however, there is minimal evidence proving the benefits of the other dietary recommendations made in the websites we evaluated.
Psoriasis
Our Google search using the term diet and psoriasis yielded a total of 9,420,000 results. Of the first 10 search results, 40% (4/10) were websites attributed to self-proclaimed experts, 30% (3/10) were promotional, and 30% (3/10) were educational. Seventy percent (7/10) of websites recommended avoiding alcohol and 60% (6/10) recommended avoiding gluten, with others discouraging consumption of red meat. Most of the websites encouraged patients to consume omega-3 fatty acids and antioxidants, while a few also recommended vitamins A, D, and E, as well as evening primrose oil supplements.
Although current research indicates a positive correlation between excessive alcohol use and psoriasis severity, it is still unclear whether alcohol consumption can be directly linked to the disease.12-14 Likewise, despite belief that increased oxidative stress likely contributes to inflammation in psoriasis, there is little evidence linking antioxidants to improvement in psoriasis symptoms.12 However, the current literature is inconsistent regarding the effects of fish oil supplementation on psoriasis.12 In a randomized double-blind study of 145 patients, there was no significant difference in psoriasis area and severity index scores between a control group and a treatment group receiving fish oil supplementation.15 In another RCT of 45 participants, those given daily very long-chain omega-3 fatty acid supplements saw no difference in psoriasis symptoms.15 Despite debate, literature assessing the impact of gluten-free diets has described improvement in psoriasis lesions in patients with celiac-specific antibodies.16 Although some observational studies described vitamin D supplementation to be beneficial in the treatment of psoriatic lesions, a more recent RCT found no significant difference between control and treatment groups.17-19
Studies also have revealed that certain eating patterns, such as those associated with the Mediterranean diet that is rich in fruits, vegetables, whole grains, and omega-3 fatty acids may be linked to improved endothelial function scores and reduced C-reactive protein and IL-18levels.20,21 In a double-blind RCT of 75 patients with plaque psoriasis, mean (SD) psoriasis area and severity index scores decreased by 11.2 (9.8) in a group treated with omega-3 fatty acids compared to 7.5 (8.8) with omega-6 fatty acids (P=.048).22
Although excessive alcohol use may be linked to psoriasis, there is no conclusive evidence indicating causation, thereby discrediting online claims.12-14 Research has revealed that gluten-free diets in psoriasis patients with celiac disease may improve psoriasis treatment16; however, sufficient evidence is lacking for diets low in gluten and high in polyunsaturated fatty acids or antioxidant supplementation. Of the dietary supplements recommended in the search results we reviewed, fish oil appears to be the most promising, but no recommendations can be made based on the current research.
Eczema
Our Google search using the term diet and eczema yielded 1,160,000 results, with 50% (5/10) of websites attributed to self-proclaimed experts, 30% (3/10) to educational websites, and 20% (2/10) to promotional sites. Of the first 10 results, 80% (8/10) recommended that patients with eczema avoid milk/dairy and 50% (5/10) advised to avoid soy and wheat/gluten. Other websites indicated to avoid eggs, nuts, and artificial sweeteners. Patients were encouraged to incorporate omega-3 fatty acids in their diets, and a few sites recommended bananas, coconut oil, olive oil, and various teas.
In a review of 11 studies with a total of 596 participants, supplementation with vitamins D and E, fish oil, olive oil, and linoleic acid was evaluated for the treatment of eczema.23 Although results indicated modest improvement of eczema severity with supplementation of fish oil, evidence favoring this treatment is limited and unconvincing. Furthermore, some evidence indicates that elimination diets are only appropriate for patients with food allergies.24 In a study evaluating an egg-free and dairy-free diet for eczema patients, only participants with positive egg-specific serum IgE levels saw improvement in disease severity.23 Even though IgE-mediated food allergies have been reported in 40% of children with moderate eczema, the contribution of these allergies to eczema is questionable.25
There is little evidence in the literature to indicate a definitive correlation between the foods mentioned in the search results we evaluated and the development of eczema; however, for patients with food allergies and eczema, elimination diets may decrease disease severity.25,26 There is insufficient evidence to suggest a benefit from evening primrose oil or fish oil supplementation, thereby debunking claims found online.
Comment
Although our Google search results included a wide range of sources and information regarding diet and dermatologic conditions such as acne, psoriasis, and eczema, most of the information we found was either unfounded or misleading. Study limitations in the current literature include small sample size, potential recall bias, lack of appropriate controls, incomplete reported results, and the failure to clearly define skin changes.
When considering the accuracy and type of information regarding skin conditions that is available on the Internet, it is important to note that most of the results we reviewed were webpages attributed to self-proclaimed experts. Although educational websites also were included in the search results, whether or not patients prefer or understand the content of such websites is still unknown; therefore, health organizations should consider revising online patient education materials to allow universal comprehension.27
Furthermore, it is important to consider the impact that widespread Internet access may have on the physician-patient relationship. Having access to health-related information online and being able to potentially self-diagnose could delay or deter patients from seeking professional advice or care.3 A study evaluating the impact of online searches on the physician-patient relationship among 175 patients determined that 36.5% of patients gathered information online prior to their consultation with a physician, while 67.3% chose to complement the information given to them by their physician with online resources.28 Based on these statistics, it is important that physicians be up-to-date with Internet discourse to discredit unfounded recommendations. Ultimately, when it comes to diet and dermatology, patients ought to be skeptical of the information currently available on the Internet, given that most of it is unsubstantiated by medical research.
- Fox S. Online health search 2006. Pew Research Center website. http://www.pewinternet.org/2006/10/29/online-health-search-2006/. Published October 29, 2006. Accessed May 3, 2018.
- Prestin A, Vieux SN, Chou WY. Is online health activity alive and well or flatlining? findings from 10 years of the health information national trends survey. J Health Commun. 2015;20:790-798.
- Zeichner JA, Del Rosso JQ. Acne and the internet. Dermatol Clin. 2016;34:129-132.
- Whitsitt J, Karimkhani C, Boyers LN, et al. Comparing burden of dermatologic disease to search interest on Google trends. Dermatol Online J. 2015;21. pii:13030/qt5xg811qp.
- Shokeen D. Influence of diet in acne vulgaris and atopic dermatitis. Cutis. 2016;98:E28-E29.
- Veith WB, Silverberg NB. The association of acne vulgaris with diet. Cutis. 2011;88:84-91.
- Rouhani P. Acne improves with a popular, low glycemic diet from South Beach. J Am Acad Dermatol. 2009;60(3, suppl 1):P706.
- Melnick BC. Evidence for acne-promoting effect of milk and other insulinotropic dairy products. Nestle Nutr Worksop Ser Pediatr Program. 2011;67:131-145.
- Adebamowo CA, Spiegelman D, Berkey CS, et al. High school dietary diary intake and teenage acne. J Am Acad Dermatol. 2005;52:207-214.
- Ulvestad M, Bjertness E, Dalgard F, et al. Acne and dairy products in adolescence: results from a Norwegian longitudinal study [published online July 16, 2016]. J Eur Acad Dermatol Venereol. 2017;31:530-535.
- Melnick BC, Schmitz G. Role of insulin, insulin like growth factor 1, hyperglycemic food and milk consumption in the pathogenesis of acne vulgaris. Exp Dermatol. 2009;18:833-841.
- Murzaku EC, Bronsnick T, Rao BK. Diet in dermatology: part II. melanoma, chronic urticaria, and psoriasis. J Am Acad Dermatol. 2014;71:1053.E1-1053.E16.
- Tobin AM, Higgins EM, Norris S, et al. Prevalence of psoriasis in patients with alcoholic liver disease. Clin Exp Dermatol. 2009;34:698-701.
- Kirby B, Richards HL, Mason DL, et al. Alcohol consumption and psychological distress in patients with psoriasis. Br J Dermatol. 2008;158:138-140.
- Søyland E, Funk J, Rajika G, et al. Effect of dietary supplementation with very long-chain n-3 fatty acids in patients with psoriasis. N Engl J Med. 1993;328:1812-1816.
- Michaëlsson G, Gerdén B, Hagforsen E, et al. Psoriasis patients with antibodies to gliadin can be improved by a gluten-free diet. Br J Dermatol. 2000;142:44-51.
- Morimoto S, Yoshikawa K. Psoriasis and vitamin D3. a review of our experience. Arch Dermatol. 1989;125:231-234.
- Smith EL, Pincus SH, Donovan L, et al. A novel approach for the evaluation and treatment of psoriasis. oral or topical use of 1,25-dihydroxyvitamin D3 can be a safe and effective therapy for psoriasis. J Am Acad Dermatol. 1988;19:516-528.
- Siddiqui MA, Al-Khawajah MM. Vitamin D3 and psoriasis: a randomized double-blind placebo-controlled study. J Dermatol Treat. 1990;1:243-245.
- Wang Y, Gao H, Loyd CM, et al. Chronic skin-specific inflammation promotes vascular inflammation and thrombosis. J Invest Dermatol. 2012;132:2067-2075.
- Barrea L, Nappi F, Di Somma C, et al. Environmental risk factors in psoriasis: the point of view of the nutritionist. Int J Environ Res Public Health. 2016;13. pii:E743. doi:10.3390/ijerph13070743.
- Mayser P, Mrowietz U, Arenberger P, et al. Omega-3 fatty acid-based lipid infusion in patients with chronic plaque psoriasis: results of a double-blind, randomized, placebo-controlled, multicenter trial. J Am Acad Dermatol. 1998;38:539-547.
- Bath-Hextall FJ, Jenkinson C, Humphreys R, et al. Dietary supplements for established atopic eczema. Cochrane Database Syst Rev. 2012;2:CD005205.
- Bronsnick T, Murzaku EC, Rao BK. Diet in dermatology: part I. atopic dermatitis, acne, and nonmelanoma skin cancer [published online November 15, 2014]. J Am Acad Dermatol. 2014;71:1039.E1-1039.E12.
- Campbell DE. The role of food allergy in childhood atopic dermatitis. J Paediatr Child Health. 2012;48:1058-1064.
- Werfel T, Erdmann S, Fuchs T, et al. Approach to suspected food allergy in atopic dermatitis. guideline of the Task Force on Food Allergy of the German Society of Allergology and Clinical Immunology (DGAKI) and the Medical Association of German Allergologists (ADA) and the German Society of Pediatric Allergology (GPA). J Dtsch Dermatol Ges. 2009;3:265-271.
- John AM, John ES, Hansberry DR, et al. Assessment of online patient education materials from major dermatologic associations. J Clin Aesthet Dermatol. 2016;9:23-28.
- Orgaz-Molina J, Cotugno M, Girón-Prieto MS, et al. A study of internet searches for medical information in dermatology patients: the patient-physician relationship. Actas Dermosifiliogr. 2015;106:493-499.
- Fox S. Online health search 2006. Pew Research Center website. http://www.pewinternet.org/2006/10/29/online-health-search-2006/. Published October 29, 2006. Accessed May 3, 2018.
- Prestin A, Vieux SN, Chou WY. Is online health activity alive and well or flatlining? findings from 10 years of the health information national trends survey. J Health Commun. 2015;20:790-798.
- Zeichner JA, Del Rosso JQ. Acne and the internet. Dermatol Clin. 2016;34:129-132.
- Whitsitt J, Karimkhani C, Boyers LN, et al. Comparing burden of dermatologic disease to search interest on Google trends. Dermatol Online J. 2015;21. pii:13030/qt5xg811qp.
- Shokeen D. Influence of diet in acne vulgaris and atopic dermatitis. Cutis. 2016;98:E28-E29.
- Veith WB, Silverberg NB. The association of acne vulgaris with diet. Cutis. 2011;88:84-91.
- Rouhani P. Acne improves with a popular, low glycemic diet from South Beach. J Am Acad Dermatol. 2009;60(3, suppl 1):P706.
- Melnick BC. Evidence for acne-promoting effect of milk and other insulinotropic dairy products. Nestle Nutr Worksop Ser Pediatr Program. 2011;67:131-145.
- Adebamowo CA, Spiegelman D, Berkey CS, et al. High school dietary diary intake and teenage acne. J Am Acad Dermatol. 2005;52:207-214.
- Ulvestad M, Bjertness E, Dalgard F, et al. Acne and dairy products in adolescence: results from a Norwegian longitudinal study [published online July 16, 2016]. J Eur Acad Dermatol Venereol. 2017;31:530-535.
- Melnick BC, Schmitz G. Role of insulin, insulin like growth factor 1, hyperglycemic food and milk consumption in the pathogenesis of acne vulgaris. Exp Dermatol. 2009;18:833-841.
- Murzaku EC, Bronsnick T, Rao BK. Diet in dermatology: part II. melanoma, chronic urticaria, and psoriasis. J Am Acad Dermatol. 2014;71:1053.E1-1053.E16.
- Tobin AM, Higgins EM, Norris S, et al. Prevalence of psoriasis in patients with alcoholic liver disease. Clin Exp Dermatol. 2009;34:698-701.
- Kirby B, Richards HL, Mason DL, et al. Alcohol consumption and psychological distress in patients with psoriasis. Br J Dermatol. 2008;158:138-140.
- Søyland E, Funk J, Rajika G, et al. Effect of dietary supplementation with very long-chain n-3 fatty acids in patients with psoriasis. N Engl J Med. 1993;328:1812-1816.
- Michaëlsson G, Gerdén B, Hagforsen E, et al. Psoriasis patients with antibodies to gliadin can be improved by a gluten-free diet. Br J Dermatol. 2000;142:44-51.
- Morimoto S, Yoshikawa K. Psoriasis and vitamin D3. a review of our experience. Arch Dermatol. 1989;125:231-234.
- Smith EL, Pincus SH, Donovan L, et al. A novel approach for the evaluation and treatment of psoriasis. oral or topical use of 1,25-dihydroxyvitamin D3 can be a safe and effective therapy for psoriasis. J Am Acad Dermatol. 1988;19:516-528.
- Siddiqui MA, Al-Khawajah MM. Vitamin D3 and psoriasis: a randomized double-blind placebo-controlled study. J Dermatol Treat. 1990;1:243-245.
- Wang Y, Gao H, Loyd CM, et al. Chronic skin-specific inflammation promotes vascular inflammation and thrombosis. J Invest Dermatol. 2012;132:2067-2075.
- Barrea L, Nappi F, Di Somma C, et al. Environmental risk factors in psoriasis: the point of view of the nutritionist. Int J Environ Res Public Health. 2016;13. pii:E743. doi:10.3390/ijerph13070743.
- Mayser P, Mrowietz U, Arenberger P, et al. Omega-3 fatty acid-based lipid infusion in patients with chronic plaque psoriasis: results of a double-blind, randomized, placebo-controlled, multicenter trial. J Am Acad Dermatol. 1998;38:539-547.
- Bath-Hextall FJ, Jenkinson C, Humphreys R, et al. Dietary supplements for established atopic eczema. Cochrane Database Syst Rev. 2012;2:CD005205.
- Bronsnick T, Murzaku EC, Rao BK. Diet in dermatology: part I. atopic dermatitis, acne, and nonmelanoma skin cancer [published online November 15, 2014]. J Am Acad Dermatol. 2014;71:1039.E1-1039.E12.
- Campbell DE. The role of food allergy in childhood atopic dermatitis. J Paediatr Child Health. 2012;48:1058-1064.
- Werfel T, Erdmann S, Fuchs T, et al. Approach to suspected food allergy in atopic dermatitis. guideline of the Task Force on Food Allergy of the German Society of Allergology and Clinical Immunology (DGAKI) and the Medical Association of German Allergologists (ADA) and the German Society of Pediatric Allergology (GPA). J Dtsch Dermatol Ges. 2009;3:265-271.
- John AM, John ES, Hansberry DR, et al. Assessment of online patient education materials from major dermatologic associations. J Clin Aesthet Dermatol. 2016;9:23-28.
- Orgaz-Molina J, Cotugno M, Girón-Prieto MS, et al. A study of internet searches for medical information in dermatology patients: the patient-physician relationship. Actas Dermosifiliogr. 2015;106:493-499.
Practice Points
- It is important physicians be well-informed regarding Internet discourse to discredit unfounded recommendations.
- It is likely that patients seeking medical advice regarding their dermatologic condition and treatment will have done prior research on the Internet.
- Oftentimes, the information on educational health websites can be confusing to patients.
- Because of widespread Internet access to health-related information, patients may opt to self-diagnose and therefore delay seeking professional care.
Patient survey results highlight disease burden in atopic dermatitis
More than half of the patients with moderate to severe atopic dermatitis (AD) had inadequately controlled disease, which was associated with a higher patient-reported disease burden compared with those who had adequately controlled disease, in a cross-sectional study of adults with AD.
Disease control aside, patient-reported burden was generally higher in those with moderate to severe AD versus patients with mild AD, according to Eric L. Simpson, MD, professor of dermatology, Oregon Health & Science University, Portland, and his coauthors.
“These results highlight beyond using measures of disease activity,” the researchers wrote. The study, published in JAMA Dermatology, was conducted before the introduction of dupilumab (Dupixent), the first biologic approved by the Food and Drug Administration for treatment of moderate to severe AD, the authors noted. (The study was supported by the manufacturers of dupilumab.)
The patients were in the Adults With Atopic Dermatitis Reporting on Their Experience (AD-AWARE) study, a cross-sectional analysis of burden of illness in adults with AD in clinical practices at six U.S. academic medical centers. The 1,519 patients completed a self-administered, Internet-based questionnaire during 2013-2014. Among these patients, 830 (54.6%) had moderate to severe AD.
A total of 185 patients with moderate to severe disease received systemic immunomodulators or phototherapy, and of those, more than half (103, or 55.7%) reported inadequate disease control, according to the survey results.
Regardless of disease control, the patients with moderate to severe AD had a greater burden of disease compared with patients with mild AD, according to the investigators. Those burdens included more severe pain and itching, sleep effects, anxiety and depression, and impairment of health-related quality of life, they reported.
Those with moderate to severe disease had a mean of 5.7 days per week with itchy skin, and 22.8% reported itch lasting for more than half a day, compared with a mean of 2.7 days and 2.9%, respectively, for those with mild disease, all significant differences.
Those with moderate to severe disease also reported more trouble sleeping, along with more frequent sleep disturbances, longer time transitioning into sleep, and more use of nonprescription sleep medications than those with mild disease.
Among those with moderate to severe disease, those who were inadequately controlled had a higher level of itch intensity and more frequent itching (a mean of 6.3 days per week), compared with those who were controlled (a mean of 5.7 days per week).
In a previous study looking at patient burden in a phase 2b clinical trial of dupilumab, Dr. Simpson and his coinvestigators found that adults with moderate to severe AD reported a “multidimensional burden” of disease that included disease activity, patient-reported symptoms, quality-of-life impact, and comorbidities (J Am Acad Dermatol. 2016 Mar;74[3]:491-8).
The current analysis based on the AD-AWARE study was supported by dupilumab manufacturers Regeneron Pharmaceuticals and Sanofi. Dr. Simpson reported disclosures related to Amgen, Anacor, Asubio, Celgene, Chugai, Galderma, Genentech, Medicis, Merck, and Regeneron; five of the 15 authors were employees of Sanofi or Regeneron. Other authors reported disclosures related to these and other companies.
SOURCE: Simpson EL et al. JAMA Dermatol. 2018 Jul 3. doi: 10.1001/jamadermatol.2018.1572.
More than half of the patients with moderate to severe atopic dermatitis (AD) had inadequately controlled disease, which was associated with a higher patient-reported disease burden compared with those who had adequately controlled disease, in a cross-sectional study of adults with AD.
Disease control aside, patient-reported burden was generally higher in those with moderate to severe AD versus patients with mild AD, according to Eric L. Simpson, MD, professor of dermatology, Oregon Health & Science University, Portland, and his coauthors.
“These results highlight beyond using measures of disease activity,” the researchers wrote. The study, published in JAMA Dermatology, was conducted before the introduction of dupilumab (Dupixent), the first biologic approved by the Food and Drug Administration for treatment of moderate to severe AD, the authors noted. (The study was supported by the manufacturers of dupilumab.)
The patients were in the Adults With Atopic Dermatitis Reporting on Their Experience (AD-AWARE) study, a cross-sectional analysis of burden of illness in adults with AD in clinical practices at six U.S. academic medical centers. The 1,519 patients completed a self-administered, Internet-based questionnaire during 2013-2014. Among these patients, 830 (54.6%) had moderate to severe AD.
A total of 185 patients with moderate to severe disease received systemic immunomodulators or phototherapy, and of those, more than half (103, or 55.7%) reported inadequate disease control, according to the survey results.
Regardless of disease control, the patients with moderate to severe AD had a greater burden of disease compared with patients with mild AD, according to the investigators. Those burdens included more severe pain and itching, sleep effects, anxiety and depression, and impairment of health-related quality of life, they reported.
Those with moderate to severe disease had a mean of 5.7 days per week with itchy skin, and 22.8% reported itch lasting for more than half a day, compared with a mean of 2.7 days and 2.9%, respectively, for those with mild disease, all significant differences.
Those with moderate to severe disease also reported more trouble sleeping, along with more frequent sleep disturbances, longer time transitioning into sleep, and more use of nonprescription sleep medications than those with mild disease.
Among those with moderate to severe disease, those who were inadequately controlled had a higher level of itch intensity and more frequent itching (a mean of 6.3 days per week), compared with those who were controlled (a mean of 5.7 days per week).
In a previous study looking at patient burden in a phase 2b clinical trial of dupilumab, Dr. Simpson and his coinvestigators found that adults with moderate to severe AD reported a “multidimensional burden” of disease that included disease activity, patient-reported symptoms, quality-of-life impact, and comorbidities (J Am Acad Dermatol. 2016 Mar;74[3]:491-8).
The current analysis based on the AD-AWARE study was supported by dupilumab manufacturers Regeneron Pharmaceuticals and Sanofi. Dr. Simpson reported disclosures related to Amgen, Anacor, Asubio, Celgene, Chugai, Galderma, Genentech, Medicis, Merck, and Regeneron; five of the 15 authors were employees of Sanofi or Regeneron. Other authors reported disclosures related to these and other companies.
SOURCE: Simpson EL et al. JAMA Dermatol. 2018 Jul 3. doi: 10.1001/jamadermatol.2018.1572.
More than half of the patients with moderate to severe atopic dermatitis (AD) had inadequately controlled disease, which was associated with a higher patient-reported disease burden compared with those who had adequately controlled disease, in a cross-sectional study of adults with AD.
Disease control aside, patient-reported burden was generally higher in those with moderate to severe AD versus patients with mild AD, according to Eric L. Simpson, MD, professor of dermatology, Oregon Health & Science University, Portland, and his coauthors.
“These results highlight beyond using measures of disease activity,” the researchers wrote. The study, published in JAMA Dermatology, was conducted before the introduction of dupilumab (Dupixent), the first biologic approved by the Food and Drug Administration for treatment of moderate to severe AD, the authors noted. (The study was supported by the manufacturers of dupilumab.)
The patients were in the Adults With Atopic Dermatitis Reporting on Their Experience (AD-AWARE) study, a cross-sectional analysis of burden of illness in adults with AD in clinical practices at six U.S. academic medical centers. The 1,519 patients completed a self-administered, Internet-based questionnaire during 2013-2014. Among these patients, 830 (54.6%) had moderate to severe AD.
A total of 185 patients with moderate to severe disease received systemic immunomodulators or phototherapy, and of those, more than half (103, or 55.7%) reported inadequate disease control, according to the survey results.
Regardless of disease control, the patients with moderate to severe AD had a greater burden of disease compared with patients with mild AD, according to the investigators. Those burdens included more severe pain and itching, sleep effects, anxiety and depression, and impairment of health-related quality of life, they reported.
Those with moderate to severe disease had a mean of 5.7 days per week with itchy skin, and 22.8% reported itch lasting for more than half a day, compared with a mean of 2.7 days and 2.9%, respectively, for those with mild disease, all significant differences.
Those with moderate to severe disease also reported more trouble sleeping, along with more frequent sleep disturbances, longer time transitioning into sleep, and more use of nonprescription sleep medications than those with mild disease.
Among those with moderate to severe disease, those who were inadequately controlled had a higher level of itch intensity and more frequent itching (a mean of 6.3 days per week), compared with those who were controlled (a mean of 5.7 days per week).
In a previous study looking at patient burden in a phase 2b clinical trial of dupilumab, Dr. Simpson and his coinvestigators found that adults with moderate to severe AD reported a “multidimensional burden” of disease that included disease activity, patient-reported symptoms, quality-of-life impact, and comorbidities (J Am Acad Dermatol. 2016 Mar;74[3]:491-8).
The current analysis based on the AD-AWARE study was supported by dupilumab manufacturers Regeneron Pharmaceuticals and Sanofi. Dr. Simpson reported disclosures related to Amgen, Anacor, Asubio, Celgene, Chugai, Galderma, Genentech, Medicis, Merck, and Regeneron; five of the 15 authors were employees of Sanofi or Regeneron. Other authors reported disclosures related to these and other companies.
SOURCE: Simpson EL et al. JAMA Dermatol. 2018 Jul 3. doi: 10.1001/jamadermatol.2018.1572.
FROM JAMA DERMATOLOGY
Key clinical point: Consider the burden of disease in patients, in addition to severity, when evaluating patients with atopic dermatitis.
Major finding: Patients with moderate to severe AD experienced itchy skin a mean of 5.7 days per week, with 22.8% reporting itch lasting for more than half a day, vs. a mean of 2.7 days and 2.9%, respectively, among those with mild disease (P less than .001 for both measures).
Study details: A cross-sectional study of 1,519 adult patients with AD, who answered a questionnaire related to disease burden.
Disclosures: Dr. Simpson reported disclosures related to Amgen, Anacor, Asubio, Celgene, Chugai, Galderma, Genentech, Medicis, Merck, and Regeneron. Five of the 15 authors were employees of Sanofi or Regeneron. Other authors reported disclosures related to these and/or other companies.
Source: Simpson EL et al. JAMA Dermatol. 2018 Jul 3. doi: 10.1001/jamadermatol.2018.1572.
Consider potty seats when you see contact dermatitis on toddler bottoms
In such cases, be on the alert for contact dermatitis, reported Claire O. Dorfman, DO, of Lehigh Valley Health Network, Allentown, Pa., and her associates at Hershey (Pa.) Medical Center.
A 3-year-old white boy with a 6-month history of a pruritic rash on his buttocks and bilateral posterior thighs was treated without improvement at the pediatric dermatology clinic with low-potency topical corticosteroids, as well as topical antibiotic and antifungal agents.
Only mild improvement was seen once disposable paper toilet seat covers were added to treatment regimen. Following the purchase of a new potty seat through an online retailer, the child’s mother discovered a number of consumer product reviews also detailing similar complaints about the manufacturer, Prince Lionheart WeePOD Basix, by more than 30 other consumers. Photos highlighting identical rash presentation in other toddlers confirmed that the toilet seat was responsible for the allergic reaction. A warning had been posted by the manufacturer but this warning was not provided by the online retailer.
Use of the seat was immediately discontinued, and complete resolution of lesions was achieved within 1 month; subsequently, a report to the Consumer Product Safety Commission was made.
Allergic contact dermatitis to toilet seats is becoming increasingly common, the authors noted. Although the source of allergies is varied, wood historically has been identified as the most common material associated with the condition. Polypropylene and polyurethane foam also have been found to cause irritation. However, in the case reported by Dr. Dorfman and her associates, the precise irritant could not be identified because of the atypical pattern of the lesions and their irregular presentation on the buttocks and thighs. They speculated that this irregularity could be attributed to “the small, round shape of the seat and the squirmy behavior of a toddler,” because the typical arciform distribution was not present. Relief was not achieved with the paper liners because they did not completely cover the seat.
Because the rash resolved when the seat was replaced, parents declined patch testing. As a result, it was not possible to identify the specific allergenic component of the polyurethane. The polyurethanes used to make the seats are synthetic polymers that contain isocyanates, and frequently diaminodiphenylmethane, a curing agent. Possible allergy to the dyes used during manufacture also was considered but the presenting rash was reported in all four of the available colors made.
Although it was speculated that exposure to cleansers could be to blame for possible irritant dermatitis given reports of cracking of the potty seat, the mother and several online reviews indicated only soap and water were used, not harsh cleaning agents.
The clinicians had no relevant financial disclosures.
SOURCE: Dorfman CO et al. Pediatr Dermatol. 2018 May 29. doi: 10.1111/pde.13534.
In such cases, be on the alert for contact dermatitis, reported Claire O. Dorfman, DO, of Lehigh Valley Health Network, Allentown, Pa., and her associates at Hershey (Pa.) Medical Center.
A 3-year-old white boy with a 6-month history of a pruritic rash on his buttocks and bilateral posterior thighs was treated without improvement at the pediatric dermatology clinic with low-potency topical corticosteroids, as well as topical antibiotic and antifungal agents.
Only mild improvement was seen once disposable paper toilet seat covers were added to treatment regimen. Following the purchase of a new potty seat through an online retailer, the child’s mother discovered a number of consumer product reviews also detailing similar complaints about the manufacturer, Prince Lionheart WeePOD Basix, by more than 30 other consumers. Photos highlighting identical rash presentation in other toddlers confirmed that the toilet seat was responsible for the allergic reaction. A warning had been posted by the manufacturer but this warning was not provided by the online retailer.
Use of the seat was immediately discontinued, and complete resolution of lesions was achieved within 1 month; subsequently, a report to the Consumer Product Safety Commission was made.
Allergic contact dermatitis to toilet seats is becoming increasingly common, the authors noted. Although the source of allergies is varied, wood historically has been identified as the most common material associated with the condition. Polypropylene and polyurethane foam also have been found to cause irritation. However, in the case reported by Dr. Dorfman and her associates, the precise irritant could not be identified because of the atypical pattern of the lesions and their irregular presentation on the buttocks and thighs. They speculated that this irregularity could be attributed to “the small, round shape of the seat and the squirmy behavior of a toddler,” because the typical arciform distribution was not present. Relief was not achieved with the paper liners because they did not completely cover the seat.
Because the rash resolved when the seat was replaced, parents declined patch testing. As a result, it was not possible to identify the specific allergenic component of the polyurethane. The polyurethanes used to make the seats are synthetic polymers that contain isocyanates, and frequently diaminodiphenylmethane, a curing agent. Possible allergy to the dyes used during manufacture also was considered but the presenting rash was reported in all four of the available colors made.
Although it was speculated that exposure to cleansers could be to blame for possible irritant dermatitis given reports of cracking of the potty seat, the mother and several online reviews indicated only soap and water were used, not harsh cleaning agents.
The clinicians had no relevant financial disclosures.
SOURCE: Dorfman CO et al. Pediatr Dermatol. 2018 May 29. doi: 10.1111/pde.13534.
In such cases, be on the alert for contact dermatitis, reported Claire O. Dorfman, DO, of Lehigh Valley Health Network, Allentown, Pa., and her associates at Hershey (Pa.) Medical Center.
A 3-year-old white boy with a 6-month history of a pruritic rash on his buttocks and bilateral posterior thighs was treated without improvement at the pediatric dermatology clinic with low-potency topical corticosteroids, as well as topical antibiotic and antifungal agents.
Only mild improvement was seen once disposable paper toilet seat covers were added to treatment regimen. Following the purchase of a new potty seat through an online retailer, the child’s mother discovered a number of consumer product reviews also detailing similar complaints about the manufacturer, Prince Lionheart WeePOD Basix, by more than 30 other consumers. Photos highlighting identical rash presentation in other toddlers confirmed that the toilet seat was responsible for the allergic reaction. A warning had been posted by the manufacturer but this warning was not provided by the online retailer.
Use of the seat was immediately discontinued, and complete resolution of lesions was achieved within 1 month; subsequently, a report to the Consumer Product Safety Commission was made.
Allergic contact dermatitis to toilet seats is becoming increasingly common, the authors noted. Although the source of allergies is varied, wood historically has been identified as the most common material associated with the condition. Polypropylene and polyurethane foam also have been found to cause irritation. However, in the case reported by Dr. Dorfman and her associates, the precise irritant could not be identified because of the atypical pattern of the lesions and their irregular presentation on the buttocks and thighs. They speculated that this irregularity could be attributed to “the small, round shape of the seat and the squirmy behavior of a toddler,” because the typical arciform distribution was not present. Relief was not achieved with the paper liners because they did not completely cover the seat.
Because the rash resolved when the seat was replaced, parents declined patch testing. As a result, it was not possible to identify the specific allergenic component of the polyurethane. The polyurethanes used to make the seats are synthetic polymers that contain isocyanates, and frequently diaminodiphenylmethane, a curing agent. Possible allergy to the dyes used during manufacture also was considered but the presenting rash was reported in all four of the available colors made.
Although it was speculated that exposure to cleansers could be to blame for possible irritant dermatitis given reports of cracking of the potty seat, the mother and several online reviews indicated only soap and water were used, not harsh cleaning agents.
The clinicians had no relevant financial disclosures.
SOURCE: Dorfman CO et al. Pediatr Dermatol. 2018 May 29. doi: 10.1111/pde.13534.
FROM THE JOURNAL OF PEDIATRIC DERMATOLOGY
Key clinical point: Suspect contact dermatitis in cases of unexplained pruritic rash.
Major finding: Allergic contact dermatitis to toilet seats is becoming increasingly common.
Study details: A case study.
Disclosures: The authors had no relevant financial disclosures.
Source: Dorfman CO et al. Pediatr Dermatol. 2018 May 29. doi: 10.1111/pde.13534.
Tralokinumab appears safe and effective for atopic dermatitis, in phase 2b study
Treatment with symptoms in patients with moderate to severe atopic dermatitis (AD), in a recent phase 2b study published in the Journal of Allergy and Clinical Immunology.
The randomized, double-blind, placebo-controlled, dose-ranging study assigned 204 patients to receive placebo or 45 mg, 150 mg, or 300 mg of tralokinumab administered subcutaneously every second week for 12 weeks. The groups had similar demographics and disease characteristics. The patients were aged 15-75 years and had Eczema Area and Severity Index (EASI) scores of 12 or more and an Investigator Global Assessment (IGA) score of 3 or higher. The coprimary endpoints were change in EASI from baseline to week 12 and the percentage of patients with either 0 (clear) or 1 (almost clear) on the IGA scale.
The higher dosages of tralokinumab showed the greatest adjusted mean differences in EASI scores: reductions of 4.36 for the 150-mg group (P = .03) and 4.94 for the 300-mg group (P = .01), compared with placebo. The changes in the 300-mg group were apparent as early as 4 weeks into treatment and were maintained beyond the 12-week mark. The greatest differences, compared with placebo, in IGA were seen in the 300-mg group as well.
Furthermore, patients who had high levels of biomarkers associated with IL-13 showed greater improvements than those seen in the intention-to-treat population at large. By week 12, patient-reported pruritus was also improved, and there were improvements in Dermatology Quality of Life Index (which did not persist past 12 weeks).
Most treatment-emergent adverse events were considered only mild or moderate, and the few more serious events were deemed unrelated to the study drug. The most common adverse events were upper respiratory infections and headaches.
“Participants entering the study had not achieved an adequate response to stable topical glucocorticoids during the 2-week run-in period and, therefore, represent a population with moderate to severe AD and major unmet treatment needs,” the investigators wrote. “The clinically meaningful benefits observed by combining tralokinumab treatment with topical glucocorticoids suggests that tralokinumab could demonstrate improvements in participants whose symptoms cannot be effectively controlled by topical glucocorticoids alone.”
The study was funded by MedImmune, a member of the AstraZeneca Group. Five authors were or are employees of the company; the three remaining authors had disclosures related to numerous pharmaceutical companies, including two with disclosures that included MedImmune.
SOURCE: Wollenberg A et al. J Allergy Clin Immunol. 2018 Jun 12. doi: 10.1016/j.jaci.2018.05.029.
Treatment with symptoms in patients with moderate to severe atopic dermatitis (AD), in a recent phase 2b study published in the Journal of Allergy and Clinical Immunology.
The randomized, double-blind, placebo-controlled, dose-ranging study assigned 204 patients to receive placebo or 45 mg, 150 mg, or 300 mg of tralokinumab administered subcutaneously every second week for 12 weeks. The groups had similar demographics and disease characteristics. The patients were aged 15-75 years and had Eczema Area and Severity Index (EASI) scores of 12 or more and an Investigator Global Assessment (IGA) score of 3 or higher. The coprimary endpoints were change in EASI from baseline to week 12 and the percentage of patients with either 0 (clear) or 1 (almost clear) on the IGA scale.
The higher dosages of tralokinumab showed the greatest adjusted mean differences in EASI scores: reductions of 4.36 for the 150-mg group (P = .03) and 4.94 for the 300-mg group (P = .01), compared with placebo. The changes in the 300-mg group were apparent as early as 4 weeks into treatment and were maintained beyond the 12-week mark. The greatest differences, compared with placebo, in IGA were seen in the 300-mg group as well.
Furthermore, patients who had high levels of biomarkers associated with IL-13 showed greater improvements than those seen in the intention-to-treat population at large. By week 12, patient-reported pruritus was also improved, and there were improvements in Dermatology Quality of Life Index (which did not persist past 12 weeks).
Most treatment-emergent adverse events were considered only mild or moderate, and the few more serious events were deemed unrelated to the study drug. The most common adverse events were upper respiratory infections and headaches.
“Participants entering the study had not achieved an adequate response to stable topical glucocorticoids during the 2-week run-in period and, therefore, represent a population with moderate to severe AD and major unmet treatment needs,” the investigators wrote. “The clinically meaningful benefits observed by combining tralokinumab treatment with topical glucocorticoids suggests that tralokinumab could demonstrate improvements in participants whose symptoms cannot be effectively controlled by topical glucocorticoids alone.”
The study was funded by MedImmune, a member of the AstraZeneca Group. Five authors were or are employees of the company; the three remaining authors had disclosures related to numerous pharmaceutical companies, including two with disclosures that included MedImmune.
SOURCE: Wollenberg A et al. J Allergy Clin Immunol. 2018 Jun 12. doi: 10.1016/j.jaci.2018.05.029.
Treatment with symptoms in patients with moderate to severe atopic dermatitis (AD), in a recent phase 2b study published in the Journal of Allergy and Clinical Immunology.
The randomized, double-blind, placebo-controlled, dose-ranging study assigned 204 patients to receive placebo or 45 mg, 150 mg, or 300 mg of tralokinumab administered subcutaneously every second week for 12 weeks. The groups had similar demographics and disease characteristics. The patients were aged 15-75 years and had Eczema Area and Severity Index (EASI) scores of 12 or more and an Investigator Global Assessment (IGA) score of 3 or higher. The coprimary endpoints were change in EASI from baseline to week 12 and the percentage of patients with either 0 (clear) or 1 (almost clear) on the IGA scale.
The higher dosages of tralokinumab showed the greatest adjusted mean differences in EASI scores: reductions of 4.36 for the 150-mg group (P = .03) and 4.94 for the 300-mg group (P = .01), compared with placebo. The changes in the 300-mg group were apparent as early as 4 weeks into treatment and were maintained beyond the 12-week mark. The greatest differences, compared with placebo, in IGA were seen in the 300-mg group as well.
Furthermore, patients who had high levels of biomarkers associated with IL-13 showed greater improvements than those seen in the intention-to-treat population at large. By week 12, patient-reported pruritus was also improved, and there were improvements in Dermatology Quality of Life Index (which did not persist past 12 weeks).
Most treatment-emergent adverse events were considered only mild or moderate, and the few more serious events were deemed unrelated to the study drug. The most common adverse events were upper respiratory infections and headaches.
“Participants entering the study had not achieved an adequate response to stable topical glucocorticoids during the 2-week run-in period and, therefore, represent a population with moderate to severe AD and major unmet treatment needs,” the investigators wrote. “The clinically meaningful benefits observed by combining tralokinumab treatment with topical glucocorticoids suggests that tralokinumab could demonstrate improvements in participants whose symptoms cannot be effectively controlled by topical glucocorticoids alone.”
The study was funded by MedImmune, a member of the AstraZeneca Group. Five authors were or are employees of the company; the three remaining authors had disclosures related to numerous pharmaceutical companies, including two with disclosures that included MedImmune.
SOURCE: Wollenberg A et al. J Allergy Clin Immunol. 2018 Jun 12. doi: 10.1016/j.jaci.2018.05.029.
FROM THE JOURNAL OF ALLERGY AND CLINICAL IMMUNOLOGY
Debunking Atopic Dermatitis Myths: Should You Use Systemic Therapy?
Myth: Because atopic dermatitis is skin-deep, systemic therapy is unnecessary.
Although atopic dermatitis (AD) primarily is known as a skin condition, recent research has indicated that it may be the start of the “atopic march” leading to the development of 1 or more other atopic conditions with multiorgan involvement. In infancy AD can progress to asthma and allergic rhinitis. Adult AD can be accompanied by systemic diseases such as inflammatory bowel disease, nephritic syndrome, and others. There also is a link between impairment of epidermal barrier function and disturbed skin microbiome in patients with AD. Therefore, systemic therapy may be warranted; the question is when should you use systemic therapy?
Most AD patients have mild to moderate disease that responds well to emollients and avoidance of disease triggers and other skin irritants. However, many AD patients experience a more severe disease course that does not respond adequately to topical therapy. For these patients, systemic therapy is a viable treatment option to improve quality of life (QOL), prevent flares, and control skin inflammation and other AD symptoms.
In 2017 an expert panel of the International Eczema Council proposed an algorithm to be used to determine if systemic therapy is warranted in patients with AD. Dermatologists must consider disease severity, impact on QOL, and risks and benefits of systemic therapies. Before starting systemic therapy, the panel recommends the following:
- Consider alternate or concomitant diagnoses
- Avoid triggers
- Optimize topical therapy
- Ensure adequate patient/caregiver education
- Treat coexistent infection
- Assess QOL
- Consider phototherapy
The American Academy of Dermatology established Guidelines of Care for the Management of AD in 2014, which provide recommendations for the most efficacious systemic agents.
Armed with these guidelines, dermatologists can work with patients to determine the most appropriate treatment course for this condition that is more than skin-deep.
Expert Commentary
Atopic dermatitis is a skin barrier abnormality that causes inflammatory skin disease and an inflammatory disorder triggering abnormal barrier. Whether we choose the outside-in or inside-out approach, it is clear that there is a systemic inflammation associated with skin disease. It is true that children respond well to barrier repair and topical therapy in many settings, as do many adults. However, chronic skin inflammation is not in isolation, triggering mucosal barrier changes allowing for more sensitization to foods and respiratory allergens as well as systemic inflammation in adults. Despite the utility of systemic steroids, the side effects generally outweigh benefit. On the other hand, phototherapy and systemic agents can clear skin and induce remissions and improved QOL. The AAD guidelines were reported before US Food and Drug Administration approval of newer agents such as dupilumab, leaving it up to the dermatologist to find the niche for this first biologic agent for AD.
—Nanette B. Silverberg, MD
Suggested Readings
Darlenski R, Kazandjieva J, Hristakieva E, et al. Atopic dermatitis as a systemic disease [published online November 22, 2013]. Clin Dermatol. 2014;32:409-413.
Sidbury R, Davis DM, Cohen DE, et al; American Academy of Dermatology. Guidelines of care for the management of atopic dermatitis: section 3. management and treatment with phototherapy and systemic agents [published online May 9, 2014]. J Am Acad Dermatol. 2014;71:327-349.
Simpson EL, Bruin-Weller M, Flohr C, et al. When does atopic dermatitis warrant systemic therapy? recommendations from an expert panel of the International Eczema Council [published online August 10, 2017]. J Am Acad Dermatol. 2017;77:623-633.
Thomas CL, Fernández-Peñas P. The microbiome and atopic eczema: more than skin deep [published online January 28, 2016]. Australas J Dermatol. 2017;58:18-24.
Myth: Because atopic dermatitis is skin-deep, systemic therapy is unnecessary.
Although atopic dermatitis (AD) primarily is known as a skin condition, recent research has indicated that it may be the start of the “atopic march” leading to the development of 1 or more other atopic conditions with multiorgan involvement. In infancy AD can progress to asthma and allergic rhinitis. Adult AD can be accompanied by systemic diseases such as inflammatory bowel disease, nephritic syndrome, and others. There also is a link between impairment of epidermal barrier function and disturbed skin microbiome in patients with AD. Therefore, systemic therapy may be warranted; the question is when should you use systemic therapy?
Most AD patients have mild to moderate disease that responds well to emollients and avoidance of disease triggers and other skin irritants. However, many AD patients experience a more severe disease course that does not respond adequately to topical therapy. For these patients, systemic therapy is a viable treatment option to improve quality of life (QOL), prevent flares, and control skin inflammation and other AD symptoms.
In 2017 an expert panel of the International Eczema Council proposed an algorithm to be used to determine if systemic therapy is warranted in patients with AD. Dermatologists must consider disease severity, impact on QOL, and risks and benefits of systemic therapies. Before starting systemic therapy, the panel recommends the following:
- Consider alternate or concomitant diagnoses
- Avoid triggers
- Optimize topical therapy
- Ensure adequate patient/caregiver education
- Treat coexistent infection
- Assess QOL
- Consider phototherapy
The American Academy of Dermatology established Guidelines of Care for the Management of AD in 2014, which provide recommendations for the most efficacious systemic agents.
Armed with these guidelines, dermatologists can work with patients to determine the most appropriate treatment course for this condition that is more than skin-deep.
Expert Commentary
Atopic dermatitis is a skin barrier abnormality that causes inflammatory skin disease and an inflammatory disorder triggering abnormal barrier. Whether we choose the outside-in or inside-out approach, it is clear that there is a systemic inflammation associated with skin disease. It is true that children respond well to barrier repair and topical therapy in many settings, as do many adults. However, chronic skin inflammation is not in isolation, triggering mucosal barrier changes allowing for more sensitization to foods and respiratory allergens as well as systemic inflammation in adults. Despite the utility of systemic steroids, the side effects generally outweigh benefit. On the other hand, phototherapy and systemic agents can clear skin and induce remissions and improved QOL. The AAD guidelines were reported before US Food and Drug Administration approval of newer agents such as dupilumab, leaving it up to the dermatologist to find the niche for this first biologic agent for AD.
—Nanette B. Silverberg, MD
Suggested Readings
Darlenski R, Kazandjieva J, Hristakieva E, et al. Atopic dermatitis as a systemic disease [published online November 22, 2013]. Clin Dermatol. 2014;32:409-413.
Sidbury R, Davis DM, Cohen DE, et al; American Academy of Dermatology. Guidelines of care for the management of atopic dermatitis: section 3. management and treatment with phototherapy and systemic agents [published online May 9, 2014]. J Am Acad Dermatol. 2014;71:327-349.
Simpson EL, Bruin-Weller M, Flohr C, et al. When does atopic dermatitis warrant systemic therapy? recommendations from an expert panel of the International Eczema Council [published online August 10, 2017]. J Am Acad Dermatol. 2017;77:623-633.
Thomas CL, Fernández-Peñas P. The microbiome and atopic eczema: more than skin deep [published online January 28, 2016]. Australas J Dermatol. 2017;58:18-24.
Myth: Because atopic dermatitis is skin-deep, systemic therapy is unnecessary.
Although atopic dermatitis (AD) primarily is known as a skin condition, recent research has indicated that it may be the start of the “atopic march” leading to the development of 1 or more other atopic conditions with multiorgan involvement. In infancy AD can progress to asthma and allergic rhinitis. Adult AD can be accompanied by systemic diseases such as inflammatory bowel disease, nephritic syndrome, and others. There also is a link between impairment of epidermal barrier function and disturbed skin microbiome in patients with AD. Therefore, systemic therapy may be warranted; the question is when should you use systemic therapy?
Most AD patients have mild to moderate disease that responds well to emollients and avoidance of disease triggers and other skin irritants. However, many AD patients experience a more severe disease course that does not respond adequately to topical therapy. For these patients, systemic therapy is a viable treatment option to improve quality of life (QOL), prevent flares, and control skin inflammation and other AD symptoms.
In 2017 an expert panel of the International Eczema Council proposed an algorithm to be used to determine if systemic therapy is warranted in patients with AD. Dermatologists must consider disease severity, impact on QOL, and risks and benefits of systemic therapies. Before starting systemic therapy, the panel recommends the following:
- Consider alternate or concomitant diagnoses
- Avoid triggers
- Optimize topical therapy
- Ensure adequate patient/caregiver education
- Treat coexistent infection
- Assess QOL
- Consider phototherapy
The American Academy of Dermatology established Guidelines of Care for the Management of AD in 2014, which provide recommendations for the most efficacious systemic agents.
Armed with these guidelines, dermatologists can work with patients to determine the most appropriate treatment course for this condition that is more than skin-deep.
Expert Commentary
Atopic dermatitis is a skin barrier abnormality that causes inflammatory skin disease and an inflammatory disorder triggering abnormal barrier. Whether we choose the outside-in or inside-out approach, it is clear that there is a systemic inflammation associated with skin disease. It is true that children respond well to barrier repair and topical therapy in many settings, as do many adults. However, chronic skin inflammation is not in isolation, triggering mucosal barrier changes allowing for more sensitization to foods and respiratory allergens as well as systemic inflammation in adults. Despite the utility of systemic steroids, the side effects generally outweigh benefit. On the other hand, phototherapy and systemic agents can clear skin and induce remissions and improved QOL. The AAD guidelines were reported before US Food and Drug Administration approval of newer agents such as dupilumab, leaving it up to the dermatologist to find the niche for this first biologic agent for AD.
—Nanette B. Silverberg, MD
Suggested Readings
Darlenski R, Kazandjieva J, Hristakieva E, et al. Atopic dermatitis as a systemic disease [published online November 22, 2013]. Clin Dermatol. 2014;32:409-413.
Sidbury R, Davis DM, Cohen DE, et al; American Academy of Dermatology. Guidelines of care for the management of atopic dermatitis: section 3. management and treatment with phototherapy and systemic agents [published online May 9, 2014]. J Am Acad Dermatol. 2014;71:327-349.
Simpson EL, Bruin-Weller M, Flohr C, et al. When does atopic dermatitis warrant systemic therapy? recommendations from an expert panel of the International Eczema Council [published online August 10, 2017]. J Am Acad Dermatol. 2017;77:623-633.
Thomas CL, Fernández-Peñas P. The microbiome and atopic eczema: more than skin deep [published online January 28, 2016]. Australas J Dermatol. 2017;58:18-24.
Eosinophilic Pustular Folliculitis With Underlying Mantle Cell Lymphoma
Eosinophilic pustular folliculitis (EPF) was originally described in 1965 and has since evolved into 3 distinct subtypes: classic, immunosuppressed (IS), and infantile types. Immunosuppressed EPF can be further subdivided into human immunodeficiency virus (HIV) associated (IS-HIV) and non-HIV associated. Human immunodeficiency virus–seronegative cases have been associated with underlying malignancies (IS-heme) or chronic immunosuppression, such as that seen in transplant patients.
Case Report
A 52-year-old man with a medical history limited to prostate adenocarcinoma treated with a robotic prostatectomy presented with a pruritic red rash on the face, neck, shoulders, and chest of 1 month’s duration. The patient previously completed a course of azithromycin 250 mg, intramuscular triamcinolone, and oral prednisone with only minor improvement. Physical examination demonstrated multiple pink folliculocentric papules and pustules scattered on the head (Figure 1A), neck, and chest (Figure 1B), as well as edematous pink papules and plaques on the forehead (Figures 1C and 1D). The palms, soles, and oral mucosa were clear.
Initial biopsy of the right side of the chest was nonspecific and most consistent with a reaction to an arthropod bite. The patient was started on oral doxycycline 100 mg twice daily for 2 weeks. With no improvement seen, additional biopsies were obtained from the left side of the chest and forehead. The biopsy of the chest showed ruptured folliculitis with evidence of acute and chronic inflammation. The biopsy of the forehead demonstrated eosinophilic follicular spongiosis with intrafollicular Langerhans cell microgranulomas along with abundant eosinophils adjacent to follicles, consistent with EPF (Figure 2). Serum HIV testing was negative. Serum white blood cell count was normal at 6400/µL (reference range, 4500–11,000/µL) with mild elevation of eosinophils (8%). The remaining complete blood cell count and comprehensive metabolic panel were within reference range. The patient was subsequently started on oral indomethacin 25 mg twice daily and triamcinolone cream 0.1%. Within a few days he experienced initial improvement in his symptoms of pruritus and diminution in the number of inflammatory follicular papules.
Approximately 1 month after presentation, he began to experience symptoms of dysphagia and fatigue. In addition, tonsillar hypertrophy and palpable neck and axillary lymphadenopathy were present. Computed tomography of the neck, chest, and abdomen showed diffuse lymphadenopathy. Full-body positron emission tomography–computed tomography demonstrated extensive metabolically active lymphoma in multiple nodal groups above and below the diaphragm. There also was lymphomatous involvement of the spleen. An axillary lymph node biopsy was diagnostic for mantle cell lymphoma (CD4:CD8, 1:1; CD45 negative; CD20 positive; CD5 positive). He
Comment
Subtypes of EPF
Eosinophilic pustular folliculitis was first described in a Japanese female presenting with folliculocentric pustules distributed on the face, torso, and arms.1 This noninfectious eosinophilic infiltration of hair follicles predominantly seen in the Japanese population is now regarded as the classic form. Three distinct subtypes of EPF now exist, including the originally described classic variant (Ofuji disease), an IS variant, and a rare infantile form.1
All 3 subtypes of EPF are more commonly seen in men than women. The classic form has a peak incidence between the third and fourth decades of life. It presents as chronic annular papules and sterile pustules exhibiting peripheral extension, with individual lesions lasting for approximately 7 to 10 days with frequent relapses. The face is the most common area of involvement, followed by the trunk, extremities, and more rarely the palmoplantar surfaces. Concomitant leukocytosis with eosinophilia is seen in up to 35% of patients.1 The infantile type represents the rarest EPF form. The average age of onset is 5 months, with most cases resolving by 14 months of age.1
Clinically, EPF is characterized by recurrent papules and pustules predominantly on the scalp without annular or polycyclic ring formation, as seen in the classic type. The palms and soles may be involved, which can clinically mimic infantile acropustulosis and scabies infection. Most patients exhibit a concomitant peripheral eosinophilia.1,2
In the late 1980s, the IS variant of EPF was recognized in HIV-positive (IS-HIV) and HIV-negative malignancy-associated (IS-heme) populations.1,3 This newly characterized form differs morphologically and biologically from the classic and infantile subtypes. The IS subtype has a unique presentation including intensely pruritic, discrete, erythematous, follicular papules with palmoplantar sparing and infrequent annular or circinate plaque forms.1 Frequently, with the IS-HIV form, CD4+ T-cell counts are below 300 cells/mL, and 25% to 50% of patients have lymphopenia with eosinophilia.3 Highly active antiretroviral therapy has been associated with EPF resolution in HIV-positive individuals; however, it also has been shown to induce transient EPF during the first 3 to 6 months of initiation.1,3,4
Unlike the IS-HIV form, the IS-heme form has occurred solely in males and is predominantly associated with hematologic malignancies (eg, non-Hodgkin lymphoma, acute lymphoblastic leukemia, acute myeloid leukemia, myelodysplastic syndrome) 30 to 90 days following bone marrow transplant, peripheral blood stem cell transplant, or chemotherapy treatment.5,6 Unlike the chronic and persistent IS-HIV form, prior cases of IS-heme EPF have been predominantly self-limited. Interestingly, only 2 reported cases of EPF have occurred prior to the diagnosis of malignancy including B-cell leukemia and myelodysplastic syndrome.5
Histopathology
All 3 identified forms of EPF histopathologically show acute and chronic lymphoeosinophilic infiltrate concentrated at the follicular isthmus, which can lead to follicular destruction. Scattered mononuclear cells, eosinophils, and neutrophils are found within the pilar outer root sheath, sebaceous glands, and ducts. Approximately 40% of cases demonstrate follicular mucinosis.1 Histopathology of lesional palmar skin in classic-type EPF demonstrates intraepidermal pustule formation with abundant eosinophils and neutrophils adjacent to the acrosyringium.7,8
Pathogenesis
Although the pathophysiology of EPF is largely unknown, it is thought to represent a helper T cell (TH2) response involving IL-4, IL-5, and IL-13 cytokines.9 Chemoattractant receptor homologous molecule 2, which is expressed on eosinophils and lymphocytes, is believed to play a role in the pruritus, edema, and inflammatory response seen adjacent to pilosebaceous units in EPF.10 Moreover, immunohistochemical and flow cytometry analysis has revealed a prevalence of prostaglandin D2 within the perisebocyte infiltrate in EPF.9 Prostaglandin D2 induces eotaxin-3 production within sebocytes via peroxisome proliferator-activated receptor γ, which enhances chemoattraction of eosinophils. This pathogenesis represents a prostaglandin-based mechanism and potentially explains the efficacy of indomethacin treatment of EPF through its cyclooxygenase inhibition and reduction of chemoattractant receptor homologous molecule 2 expression.9-11
Treatment
Multiple therapeutic modalities have been reported for the treatment of EPF. For all 3 subtypes, moderate- to high-potency topical corticosteroids are considered first-line therapy. UVB phototherapy 2 to 3 times weekly remains the gold standard, given its consistent efficacy.1,12 Indomethacin (50–75 mg daily) remains first-line treatment of classic EPF.4,12 Previously reported cases of classic EPF and IS-EPF have responded well to oral prednisone (1 mg/kg daily).12,13 In a retrospective review of EPF treatment data, the following treatments also have been reported to be successful: psoralen plus UVA, oral cetirizine (20–40 mg daily, particularly for IS-EPF cases), metronidazole (250 mg 3 times daily), minocycline (150 mg daily), itraconazole (200–400 mg daily, dapsone (50–200 mg daily), systemic retinoids, tacrolimus ointment 0.1%, and permethrin cream.4,12
Malignancy
Although the entity of IS-heme EPF is rare, the morphology and treatment are unique and can potentially unmask an underlying hematologic malignancy. In patients with EPF and associated malignancy, such as our patient, a differential diagnosis to consider is eosinophilic dermatosis of hematologic malignancy (EDHM). Eosinophilic dermatosis of hematologic malignancy is most commonly associated with chronic lymphocytic leukemia and can be differentiated from EPF clinically, histopathologically, and by treatment response. Eosinophilic dermatosis of hematologic malignancy clinically presents with nonspecific papules, pustules, and/or vesicles on the head, trunk, and extremities. On histopathology, EDHM shows a superficial and deep perivascular and interstitial lymphoeosinophilic infiltration. Furthermore, EDHM patients typically exhibit a poor treatment response to oral indomethacin.14
Conclusion
Eosinophilic pustular folliculitis is a noninfectious folliculocentric process comprised of 3 distinct types. The histopathology shows follicular spongiosis with increased eosinophils. The pathogenesis is most likely related to a multifactorial immune system dysregulation involving TH2 T cells, prostaglandin D2, and eotaxin-3. The treatment of EPF may involve topical corticosteroids, UVB phototherapy, or most notably oral indomethacin. In patients with EPF and malignancy, EDHM is a differential diagnosis to consider. Our case serves as a reminder that rare eosinophilic dermatoses may represent manifestations of underlying hematopoietic malignancy and, when investigated early, can lead to appropriate life-saving treatment.
- Nervi J, Stephen. Eosinophilic pustular folliculitis: a 40 year retrospect. J Am Acad Dermatol. 2006;55:285-289.
- Hernández-Martín Á, Nuño-González A, Colmenero I, et al. Eosinophilic pustular folliculitis of infancy: a series of 15 cases and review of the literature [published online July 21, 2012]. J Am Acad Dermatol. 2013;68:150-155.
- Soepr
ono F, Schinella R. Eosinophilic pustular folliculitis in patients with acquired immunodeficiency syndrome. report of three cases. J Am Acad Dermatol. 1986;14:1020-1022. - Katoh
M, Nomura T, Miyachi Y, et al. Eosinophilic pustular folliculitis: a review of the Japanese published works. J Dermatol. 2013;40:15-20. - Keida
T, Hayashi N, Kawashima M. Eosinophilic pustular folliculitis following autologous peripheral blood stem-cell transplant. J Dermatol. 2004;31:21-26. - Goiriz R, Gul-Millán G, Peñas PF, et al. Eosinophilic folliculitis following allogeneic peripheral blood stem cell transplantation: case report and review. J Cutan Pathol. 2007;34(suppl 1):33-36.
- Satoh T, Ikeda H, Yokozeki H. Acrosyringeal involvement of palmoplantar lesions of eosinophilic pustular folliculitis. Acta Derm Venereol. 2013;93:99.
- Tsuboi H, Wakita K, Fujimura T, et al. Acral variant of eosinophilic pustular folliculitis (Ofuji’s disease). Clin Exp Dermatol. 2003;28:321-324.
- Nakahig
ashi K, Doi H, Otsuka A, et al. PGD2 induces eotaxin-3 via PPARgamma from sebocytes: a possible pathogenesis of eosinophilic pustular folliculitis. J Allergy Clin Immunol. 2012;129:536-543. - Satoh
T, Shimura C, Miyagishi C, et al. Indomethacin-induced reduction in CRTH2 in eosinophilic pustular folliculitis (Ofuji’s disease): a proposed mechanism of action. Acta Derm Venereol. 2010;90:18-22. - Hagiwara A, Fujimura T, Furudate S, et al. Induction of CD163(+)M2 macrophages in the lesional skin of eosinophilic pustular folliculitis. Acta Derm Venereol. 2014;94:104-106.
- Ellis
E, Scheinfeld N. Eosinophilic pustular folliculitis: a comprehensive review of treatment options. Am J Clin Dermatol. 2004;5:189-197. - Bull R
H, Harland CA, Fallowfield ME, et al. Eosinophilic folliculitis: a self-limiting illness in patients being treated for haematological malignancy. Br J Dermatol. 1993;129:178-182. - Farber M, Forgia S, Sahu J, et al. Eosinophilic dermatosis of hematologic malignancy. J Cutan Pathol. 2012;39:690-695.
Eosinophilic pustular folliculitis (EPF) was originally described in 1965 and has since evolved into 3 distinct subtypes: classic, immunosuppressed (IS), and infantile types. Immunosuppressed EPF can be further subdivided into human immunodeficiency virus (HIV) associated (IS-HIV) and non-HIV associated. Human immunodeficiency virus–seronegative cases have been associated with underlying malignancies (IS-heme) or chronic immunosuppression, such as that seen in transplant patients.
Case Report
A 52-year-old man with a medical history limited to prostate adenocarcinoma treated with a robotic prostatectomy presented with a pruritic red rash on the face, neck, shoulders, and chest of 1 month’s duration. The patient previously completed a course of azithromycin 250 mg, intramuscular triamcinolone, and oral prednisone with only minor improvement. Physical examination demonstrated multiple pink folliculocentric papules and pustules scattered on the head (Figure 1A), neck, and chest (Figure 1B), as well as edematous pink papules and plaques on the forehead (Figures 1C and 1D). The palms, soles, and oral mucosa were clear.

Initial biopsy of the right side of the chest was nonspecific and most consistent with a reaction to an arthropod bite. The patient was started on oral doxycycline 100 mg twice daily for 2 weeks. With no improvement seen, additional biopsies were obtained from the left side of the chest and forehead. The biopsy of the chest showed ruptured folliculitis with evidence of acute and chronic inflammation. The biopsy of the forehead demonstrated eosinophilic follicular spongiosis with intrafollicular Langerhans cell microgranulomas along with abundant eosinophils adjacent to follicles, consistent with EPF (Figure 2). Serum HIV testing was negative. Serum white blood cell count was normal at 6400/µL (reference range, 4500–11,000/µL) with mild elevation of eosinophils (8%). The remaining complete blood cell count and comprehensive metabolic panel were within reference range. The patient was subsequently started on oral indomethacin 25 mg twice daily and triamcinolone cream 0.1%. Within a few days he experienced initial improvement in his symptoms of pruritus and diminution in the number of inflammatory follicular papules.

Approximately 1 month after presentation, he began to experience symptoms of dysphagia and fatigue. In addition, tonsillar hypertrophy and palpable neck and axillary lymphadenopathy were present. Computed tomography of the neck, chest, and abdomen showed diffuse lymphadenopathy. Full-body positron emission tomography–computed tomography demonstrated extensive metabolically active lymphoma in multiple nodal groups above and below the diaphragm. There also was lymphomatous involvement of the spleen. An axillary lymph node biopsy was diagnostic for mantle cell lymphoma (CD4:CD8, 1:1; CD45 negative; CD20 positive; CD5 positive). He
Comment
Subtypes of EPF
Eosinophilic pustular folliculitis was first described in a Japanese female presenting with folliculocentric pustules distributed on the face, torso, and arms.1 This noninfectious eosinophilic infiltration of hair follicles predominantly seen in the Japanese population is now regarded as the classic form. Three distinct subtypes of EPF now exist, including the originally described classic variant (Ofuji disease), an IS variant, and a rare infantile form.1
All 3 subtypes of EPF are more commonly seen in men than women. The classic form has a peak incidence between the third and fourth decades of life. It presents as chronic annular papules and sterile pustules exhibiting peripheral extension, with individual lesions lasting for approximately 7 to 10 days with frequent relapses. The face is the most common area of involvement, followed by the trunk, extremities, and more rarely the palmoplantar surfaces. Concomitant leukocytosis with eosinophilia is seen in up to 35% of patients.1 The infantile type represents the rarest EPF form. The average age of onset is 5 months, with most cases resolving by 14 months of age.1
Clinically, EPF is characterized by recurrent papules and pustules predominantly on the scalp without annular or polycyclic ring formation, as seen in the classic type. The palms and soles may be involved, which can clinically mimic infantile acropustulosis and scabies infection. Most patients exhibit a concomitant peripheral eosinophilia.1,2
In the late 1980s, the IS variant of EPF was recognized in HIV-positive (IS-HIV) and HIV-negative malignancy-associated (IS-heme) populations.1,3 This newly characterized form differs morphologically and biologically from the classic and infantile subtypes. The IS subtype has a unique presentation including intensely pruritic, discrete, erythematous, follicular papules with palmoplantar sparing and infrequent annular or circinate plaque forms.1 Frequently, with the IS-HIV form, CD4+ T-cell counts are below 300 cells/mL, and 25% to 50% of patients have lymphopenia with eosinophilia.3 Highly active antiretroviral therapy has been associated with EPF resolution in HIV-positive individuals; however, it also has been shown to induce transient EPF during the first 3 to 6 months of initiation.1,3,4
Unlike the IS-HIV form, the IS-heme form has occurred solely in males and is predominantly associated with hematologic malignancies (eg, non-Hodgkin lymphoma, acute lymphoblastic leukemia, acute myeloid leukemia, myelodysplastic syndrome) 30 to 90 days following bone marrow transplant, peripheral blood stem cell transplant, or chemotherapy treatment.5,6 Unlike the chronic and persistent IS-HIV form, prior cases of IS-heme EPF have been predominantly self-limited. Interestingly, only 2 reported cases of EPF have occurred prior to the diagnosis of malignancy including B-cell leukemia and myelodysplastic syndrome.5
Histopathology
All 3 identified forms of EPF histopathologically show acute and chronic lymphoeosinophilic infiltrate concentrated at the follicular isthmus, which can lead to follicular destruction. Scattered mononuclear cells, eosinophils, and neutrophils are found within the pilar outer root sheath, sebaceous glands, and ducts. Approximately 40% of cases demonstrate follicular mucinosis.1 Histopathology of lesional palmar skin in classic-type EPF demonstrates intraepidermal pustule formation with abundant eosinophils and neutrophils adjacent to the acrosyringium.7,8
Pathogenesis
Although the pathophysiology of EPF is largely unknown, it is thought to represent a helper T cell (TH2) response involving IL-4, IL-5, and IL-13 cytokines.9 Chemoattractant receptor homologous molecule 2, which is expressed on eosinophils and lymphocytes, is believed to play a role in the pruritus, edema, and inflammatory response seen adjacent to pilosebaceous units in EPF.10 Moreover, immunohistochemical and flow cytometry analysis has revealed a prevalence of prostaglandin D2 within the perisebocyte infiltrate in EPF.9 Prostaglandin D2 induces eotaxin-3 production within sebocytes via peroxisome proliferator-activated receptor γ, which enhances chemoattraction of eosinophils. This pathogenesis represents a prostaglandin-based mechanism and potentially explains the efficacy of indomethacin treatment of EPF through its cyclooxygenase inhibition and reduction of chemoattractant receptor homologous molecule 2 expression.9-11
Treatment
Multiple therapeutic modalities have been reported for the treatment of EPF. For all 3 subtypes, moderate- to high-potency topical corticosteroids are considered first-line therapy. UVB phototherapy 2 to 3 times weekly remains the gold standard, given its consistent efficacy.1,12 Indomethacin (50–75 mg daily) remains first-line treatment of classic EPF.4,12 Previously reported cases of classic EPF and IS-EPF have responded well to oral prednisone (1 mg/kg daily).12,13 In a retrospective review of EPF treatment data, the following treatments also have been reported to be successful: psoralen plus UVA, oral cetirizine (20–40 mg daily, particularly for IS-EPF cases), metronidazole (250 mg 3 times daily), minocycline (150 mg daily), itraconazole (200–400 mg daily, dapsone (50–200 mg daily), systemic retinoids, tacrolimus ointment 0.1%, and permethrin cream.4,12
Malignancy
Although the entity of IS-heme EPF is rare, the morphology and treatment are unique and can potentially unmask an underlying hematologic malignancy. In patients with EPF and associated malignancy, such as our patient, a differential diagnosis to consider is eosinophilic dermatosis of hematologic malignancy (EDHM). Eosinophilic dermatosis of hematologic malignancy is most commonly associated with chronic lymphocytic leukemia and can be differentiated from EPF clinically, histopathologically, and by treatment response. Eosinophilic dermatosis of hematologic malignancy clinically presents with nonspecific papules, pustules, and/or vesicles on the head, trunk, and extremities. On histopathology, EDHM shows a superficial and deep perivascular and interstitial lymphoeosinophilic infiltration. Furthermore, EDHM patients typically exhibit a poor treatment response to oral indomethacin.14
Conclusion
Eosinophilic pustular folliculitis is a noninfectious folliculocentric process comprised of 3 distinct types. The histopathology shows follicular spongiosis with increased eosinophils. The pathogenesis is most likely related to a multifactorial immune system dysregulation involving TH2 T cells, prostaglandin D2, and eotaxin-3. The treatment of EPF may involve topical corticosteroids, UVB phototherapy, or most notably oral indomethacin. In patients with EPF and malignancy, EDHM is a differential diagnosis to consider. Our case serves as a reminder that rare eosinophilic dermatoses may represent manifestations of underlying hematopoietic malignancy and, when investigated early, can lead to appropriate life-saving treatment.
Eosinophilic pustular folliculitis (EPF) was originally described in 1965 and has since evolved into 3 distinct subtypes: classic, immunosuppressed (IS), and infantile types. Immunosuppressed EPF can be further subdivided into human immunodeficiency virus (HIV) associated (IS-HIV) and non-HIV associated. Human immunodeficiency virus–seronegative cases have been associated with underlying malignancies (IS-heme) or chronic immunosuppression, such as that seen in transplant patients.
Case Report
A 52-year-old man with a medical history limited to prostate adenocarcinoma treated with a robotic prostatectomy presented with a pruritic red rash on the face, neck, shoulders, and chest of 1 month’s duration. The patient previously completed a course of azithromycin 250 mg, intramuscular triamcinolone, and oral prednisone with only minor improvement. Physical examination demonstrated multiple pink folliculocentric papules and pustules scattered on the head (Figure 1A), neck, and chest (Figure 1B), as well as edematous pink papules and plaques on the forehead (Figures 1C and 1D). The palms, soles, and oral mucosa were clear.

Initial biopsy of the right side of the chest was nonspecific and most consistent with a reaction to an arthropod bite. The patient was started on oral doxycycline 100 mg twice daily for 2 weeks. With no improvement seen, additional biopsies were obtained from the left side of the chest and forehead. The biopsy of the chest showed ruptured folliculitis with evidence of acute and chronic inflammation. The biopsy of the forehead demonstrated eosinophilic follicular spongiosis with intrafollicular Langerhans cell microgranulomas along with abundant eosinophils adjacent to follicles, consistent with EPF (Figure 2). Serum HIV testing was negative. Serum white blood cell count was normal at 6400/µL (reference range, 4500–11,000/µL) with mild elevation of eosinophils (8%). The remaining complete blood cell count and comprehensive metabolic panel were within reference range. The patient was subsequently started on oral indomethacin 25 mg twice daily and triamcinolone cream 0.1%. Within a few days he experienced initial improvement in his symptoms of pruritus and diminution in the number of inflammatory follicular papules.

Approximately 1 month after presentation, he began to experience symptoms of dysphagia and fatigue. In addition, tonsillar hypertrophy and palpable neck and axillary lymphadenopathy were present. Computed tomography of the neck, chest, and abdomen showed diffuse lymphadenopathy. Full-body positron emission tomography–computed tomography demonstrated extensive metabolically active lymphoma in multiple nodal groups above and below the diaphragm. There also was lymphomatous involvement of the spleen. An axillary lymph node biopsy was diagnostic for mantle cell lymphoma (CD4:CD8, 1:1; CD45 negative; CD20 positive; CD5 positive). He
Comment
Subtypes of EPF
Eosinophilic pustular folliculitis was first described in a Japanese female presenting with folliculocentric pustules distributed on the face, torso, and arms.1 This noninfectious eosinophilic infiltration of hair follicles predominantly seen in the Japanese population is now regarded as the classic form. Three distinct subtypes of EPF now exist, including the originally described classic variant (Ofuji disease), an IS variant, and a rare infantile form.1
All 3 subtypes of EPF are more commonly seen in men than women. The classic form has a peak incidence between the third and fourth decades of life. It presents as chronic annular papules and sterile pustules exhibiting peripheral extension, with individual lesions lasting for approximately 7 to 10 days with frequent relapses. The face is the most common area of involvement, followed by the trunk, extremities, and more rarely the palmoplantar surfaces. Concomitant leukocytosis with eosinophilia is seen in up to 35% of patients.1 The infantile type represents the rarest EPF form. The average age of onset is 5 months, with most cases resolving by 14 months of age.1
Clinically, EPF is characterized by recurrent papules and pustules predominantly on the scalp without annular or polycyclic ring formation, as seen in the classic type. The palms and soles may be involved, which can clinically mimic infantile acropustulosis and scabies infection. Most patients exhibit a concomitant peripheral eosinophilia.1,2
In the late 1980s, the IS variant of EPF was recognized in HIV-positive (IS-HIV) and HIV-negative malignancy-associated (IS-heme) populations.1,3 This newly characterized form differs morphologically and biologically from the classic and infantile subtypes. The IS subtype has a unique presentation including intensely pruritic, discrete, erythematous, follicular papules with palmoplantar sparing and infrequent annular or circinate plaque forms.1 Frequently, with the IS-HIV form, CD4+ T-cell counts are below 300 cells/mL, and 25% to 50% of patients have lymphopenia with eosinophilia.3 Highly active antiretroviral therapy has been associated with EPF resolution in HIV-positive individuals; however, it also has been shown to induce transient EPF during the first 3 to 6 months of initiation.1,3,4
Unlike the IS-HIV form, the IS-heme form has occurred solely in males and is predominantly associated with hematologic malignancies (eg, non-Hodgkin lymphoma, acute lymphoblastic leukemia, acute myeloid leukemia, myelodysplastic syndrome) 30 to 90 days following bone marrow transplant, peripheral blood stem cell transplant, or chemotherapy treatment.5,6 Unlike the chronic and persistent IS-HIV form, prior cases of IS-heme EPF have been predominantly self-limited. Interestingly, only 2 reported cases of EPF have occurred prior to the diagnosis of malignancy including B-cell leukemia and myelodysplastic syndrome.5
Histopathology
All 3 identified forms of EPF histopathologically show acute and chronic lymphoeosinophilic infiltrate concentrated at the follicular isthmus, which can lead to follicular destruction. Scattered mononuclear cells, eosinophils, and neutrophils are found within the pilar outer root sheath, sebaceous glands, and ducts. Approximately 40% of cases demonstrate follicular mucinosis.1 Histopathology of lesional palmar skin in classic-type EPF demonstrates intraepidermal pustule formation with abundant eosinophils and neutrophils adjacent to the acrosyringium.7,8
Pathogenesis
Although the pathophysiology of EPF is largely unknown, it is thought to represent a helper T cell (TH2) response involving IL-4, IL-5, and IL-13 cytokines.9 Chemoattractant receptor homologous molecule 2, which is expressed on eosinophils and lymphocytes, is believed to play a role in the pruritus, edema, and inflammatory response seen adjacent to pilosebaceous units in EPF.10 Moreover, immunohistochemical and flow cytometry analysis has revealed a prevalence of prostaglandin D2 within the perisebocyte infiltrate in EPF.9 Prostaglandin D2 induces eotaxin-3 production within sebocytes via peroxisome proliferator-activated receptor γ, which enhances chemoattraction of eosinophils. This pathogenesis represents a prostaglandin-based mechanism and potentially explains the efficacy of indomethacin treatment of EPF through its cyclooxygenase inhibition and reduction of chemoattractant receptor homologous molecule 2 expression.9-11
Treatment
Multiple therapeutic modalities have been reported for the treatment of EPF. For all 3 subtypes, moderate- to high-potency topical corticosteroids are considered first-line therapy. UVB phototherapy 2 to 3 times weekly remains the gold standard, given its consistent efficacy.1,12 Indomethacin (50–75 mg daily) remains first-line treatment of classic EPF.4,12 Previously reported cases of classic EPF and IS-EPF have responded well to oral prednisone (1 mg/kg daily).12,13 In a retrospective review of EPF treatment data, the following treatments also have been reported to be successful: psoralen plus UVA, oral cetirizine (20–40 mg daily, particularly for IS-EPF cases), metronidazole (250 mg 3 times daily), minocycline (150 mg daily), itraconazole (200–400 mg daily, dapsone (50–200 mg daily), systemic retinoids, tacrolimus ointment 0.1%, and permethrin cream.4,12
Malignancy
Although the entity of IS-heme EPF is rare, the morphology and treatment are unique and can potentially unmask an underlying hematologic malignancy. In patients with EPF and associated malignancy, such as our patient, a differential diagnosis to consider is eosinophilic dermatosis of hematologic malignancy (EDHM). Eosinophilic dermatosis of hematologic malignancy is most commonly associated with chronic lymphocytic leukemia and can be differentiated from EPF clinically, histopathologically, and by treatment response. Eosinophilic dermatosis of hematologic malignancy clinically presents with nonspecific papules, pustules, and/or vesicles on the head, trunk, and extremities. On histopathology, EDHM shows a superficial and deep perivascular and interstitial lymphoeosinophilic infiltration. Furthermore, EDHM patients typically exhibit a poor treatment response to oral indomethacin.14
Conclusion
Eosinophilic pustular folliculitis is a noninfectious folliculocentric process comprised of 3 distinct types. The histopathology shows follicular spongiosis with increased eosinophils. The pathogenesis is most likely related to a multifactorial immune system dysregulation involving TH2 T cells, prostaglandin D2, and eotaxin-3. The treatment of EPF may involve topical corticosteroids, UVB phototherapy, or most notably oral indomethacin. In patients with EPF and malignancy, EDHM is a differential diagnosis to consider. Our case serves as a reminder that rare eosinophilic dermatoses may represent manifestations of underlying hematopoietic malignancy and, when investigated early, can lead to appropriate life-saving treatment.
- Nervi J, Stephen. Eosinophilic pustular folliculitis: a 40 year retrospect. J Am Acad Dermatol. 2006;55:285-289.
- Hernández-Martín Á, Nuño-González A, Colmenero I, et al. Eosinophilic pustular folliculitis of infancy: a series of 15 cases and review of the literature [published online July 21, 2012]. J Am Acad Dermatol. 2013;68:150-155.
- Soepr
ono F, Schinella R. Eosinophilic pustular folliculitis in patients with acquired immunodeficiency syndrome. report of three cases. J Am Acad Dermatol. 1986;14:1020-1022. - Katoh
M, Nomura T, Miyachi Y, et al. Eosinophilic pustular folliculitis: a review of the Japanese published works. J Dermatol. 2013;40:15-20. - Keida
T, Hayashi N, Kawashima M. Eosinophilic pustular folliculitis following autologous peripheral blood stem-cell transplant. J Dermatol. 2004;31:21-26. - Goiriz R, Gul-Millán G, Peñas PF, et al. Eosinophilic folliculitis following allogeneic peripheral blood stem cell transplantation: case report and review. J Cutan Pathol. 2007;34(suppl 1):33-36.
- Satoh T, Ikeda H, Yokozeki H. Acrosyringeal involvement of palmoplantar lesions of eosinophilic pustular folliculitis. Acta Derm Venereol. 2013;93:99.
- Tsuboi H, Wakita K, Fujimura T, et al. Acral variant of eosinophilic pustular folliculitis (Ofuji’s disease). Clin Exp Dermatol. 2003;28:321-324.
- Nakahig
ashi K, Doi H, Otsuka A, et al. PGD2 induces eotaxin-3 via PPARgamma from sebocytes: a possible pathogenesis of eosinophilic pustular folliculitis. J Allergy Clin Immunol. 2012;129:536-543. - Satoh
T, Shimura C, Miyagishi C, et al. Indomethacin-induced reduction in CRTH2 in eosinophilic pustular folliculitis (Ofuji’s disease): a proposed mechanism of action. Acta Derm Venereol. 2010;90:18-22. - Hagiwara A, Fujimura T, Furudate S, et al. Induction of CD163(+)M2 macrophages in the lesional skin of eosinophilic pustular folliculitis. Acta Derm Venereol. 2014;94:104-106.
- Ellis
E, Scheinfeld N. Eosinophilic pustular folliculitis: a comprehensive review of treatment options. Am J Clin Dermatol. 2004;5:189-197. - Bull R
H, Harland CA, Fallowfield ME, et al. Eosinophilic folliculitis: a self-limiting illness in patients being treated for haematological malignancy. Br J Dermatol. 1993;129:178-182. - Farber M, Forgia S, Sahu J, et al. Eosinophilic dermatosis of hematologic malignancy. J Cutan Pathol. 2012;39:690-695.
- Nervi J, Stephen. Eosinophilic pustular folliculitis: a 40 year retrospect. J Am Acad Dermatol. 2006;55:285-289.
- Hernández-Martín Á, Nuño-González A, Colmenero I, et al. Eosinophilic pustular folliculitis of infancy: a series of 15 cases and review of the literature [published online July 21, 2012]. J Am Acad Dermatol. 2013;68:150-155.
- Soepr
ono F, Schinella R. Eosinophilic pustular folliculitis in patients with acquired immunodeficiency syndrome. report of three cases. J Am Acad Dermatol. 1986;14:1020-1022. - Katoh
M, Nomura T, Miyachi Y, et al. Eosinophilic pustular folliculitis: a review of the Japanese published works. J Dermatol. 2013;40:15-20. - Keida
T, Hayashi N, Kawashima M. Eosinophilic pustular folliculitis following autologous peripheral blood stem-cell transplant. J Dermatol. 2004;31:21-26. - Goiriz R, Gul-Millán G, Peñas PF, et al. Eosinophilic folliculitis following allogeneic peripheral blood stem cell transplantation: case report and review. J Cutan Pathol. 2007;34(suppl 1):33-36.
- Satoh T, Ikeda H, Yokozeki H. Acrosyringeal involvement of palmoplantar lesions of eosinophilic pustular folliculitis. Acta Derm Venereol. 2013;93:99.
- Tsuboi H, Wakita K, Fujimura T, et al. Acral variant of eosinophilic pustular folliculitis (Ofuji’s disease). Clin Exp Dermatol. 2003;28:321-324.
- Nakahig
ashi K, Doi H, Otsuka A, et al. PGD2 induces eotaxin-3 via PPARgamma from sebocytes: a possible pathogenesis of eosinophilic pustular folliculitis. J Allergy Clin Immunol. 2012;129:536-543. - Satoh
T, Shimura C, Miyagishi C, et al. Indomethacin-induced reduction in CRTH2 in eosinophilic pustular folliculitis (Ofuji’s disease): a proposed mechanism of action. Acta Derm Venereol. 2010;90:18-22. - Hagiwara A, Fujimura T, Furudate S, et al. Induction of CD163(+)M2 macrophages in the lesional skin of eosinophilic pustular folliculitis. Acta Derm Venereol. 2014;94:104-106.
- Ellis
E, Scheinfeld N. Eosinophilic pustular folliculitis: a comprehensive review of treatment options. Am J Clin Dermatol. 2004;5:189-197. - Bull R
H, Harland CA, Fallowfield ME, et al. Eosinophilic folliculitis: a self-limiting illness in patients being treated for haematological malignancy. Br J Dermatol. 1993;129:178-182. - Farber M, Forgia S, Sahu J, et al. Eosinophilic dermatosis of hematologic malignancy. J Cutan Pathol. 2012;39:690-695.
Practice Points
- Recalcitrant folliculocentric papules and pustules involving the head, trunk, arms, and legs should raise suspicion of possible eosinophilic pustular folliculitis (EPF).
- Underlying hematopoietic malignancy may be associated with cases of EPF.
Acrodermatitis Enteropathica From Zinc-Deficient Total Parenteral Nutrition
Case Report
A 54-year-old woman presented with a pruritic and slightly painful skin eruption that began perinasally and progressed over 1 week to involve the labial commissures, finger webs, dorsal surfaces of the feet, heels, and bilateral gluteal folds. In addition, the eruption involved the left thigh at the donor site of a prior skin graft. She received no relief after an intramuscular steroid injection and hydrocortisone cream 1% prescribed by a primary care physician who diagnosed the rash as poison ivy contact dermatitis despite no exposure to plants. Review of systems was negative and she denied any new medication use. Her medical history was notable for extensive mesenteric injury secondary to a motor vehicle accident. She subsequently had multiple enterocutaneous fistulas that resulted in a complete small bowel enterectomy 10 months prior to presentation, which caused her to become dependent on total parenteral nutrition (TPN).
Physical examination revealed sharply demarcated, erythematous, scaly plaques perinasally, periorally, and on the bilateral gluteal folds (Figure 1). There were sharply demarcated, erythematous, scaly plaques on the right and left finger webs, dorsal surface of the right foot, and left upper thigh. Hemorrhagic bullae were appreciated on the left finger webs. Large flaccid bullae were present on the bilateral heels and dorsum of the right foot (Figure 2).
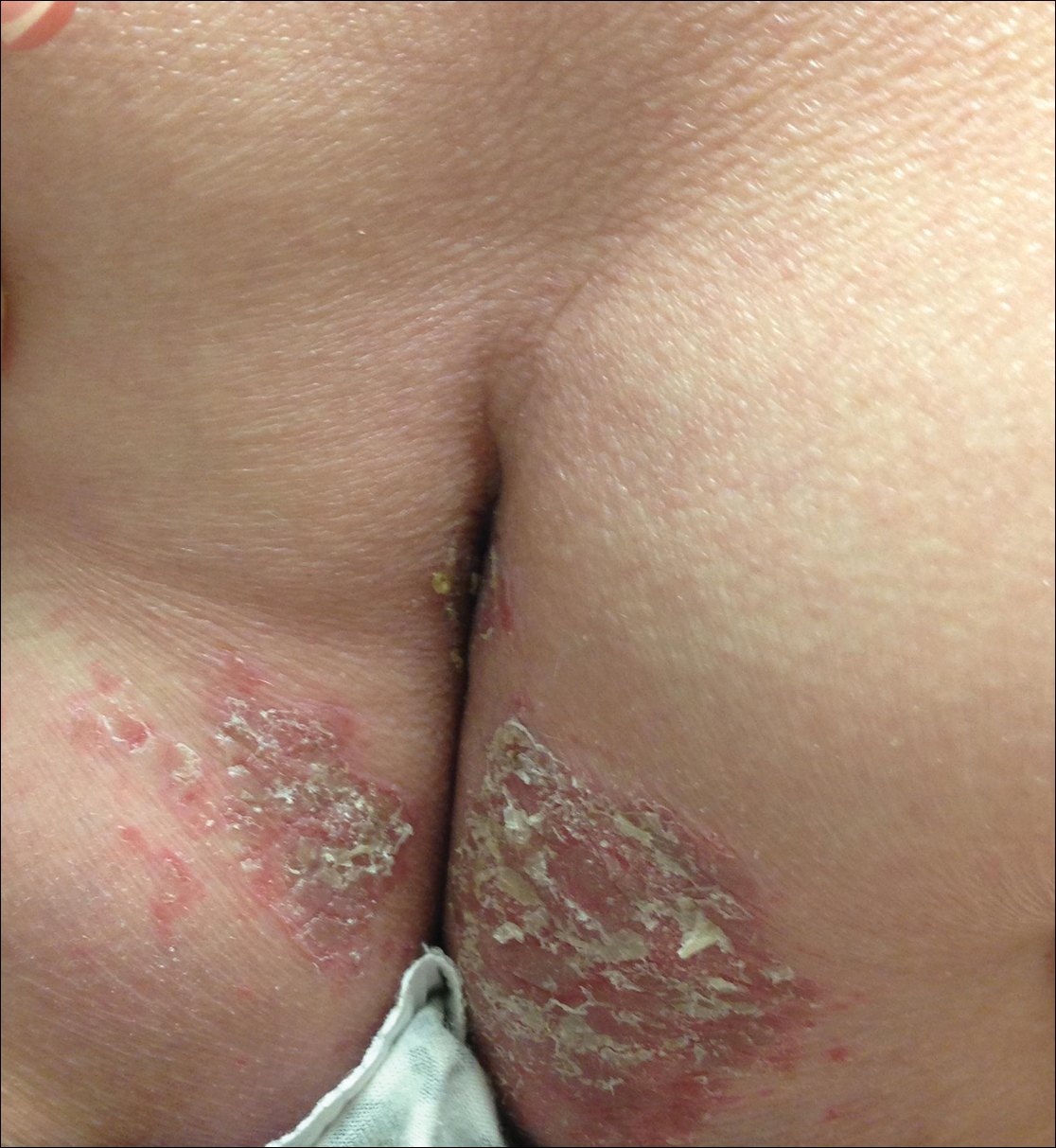
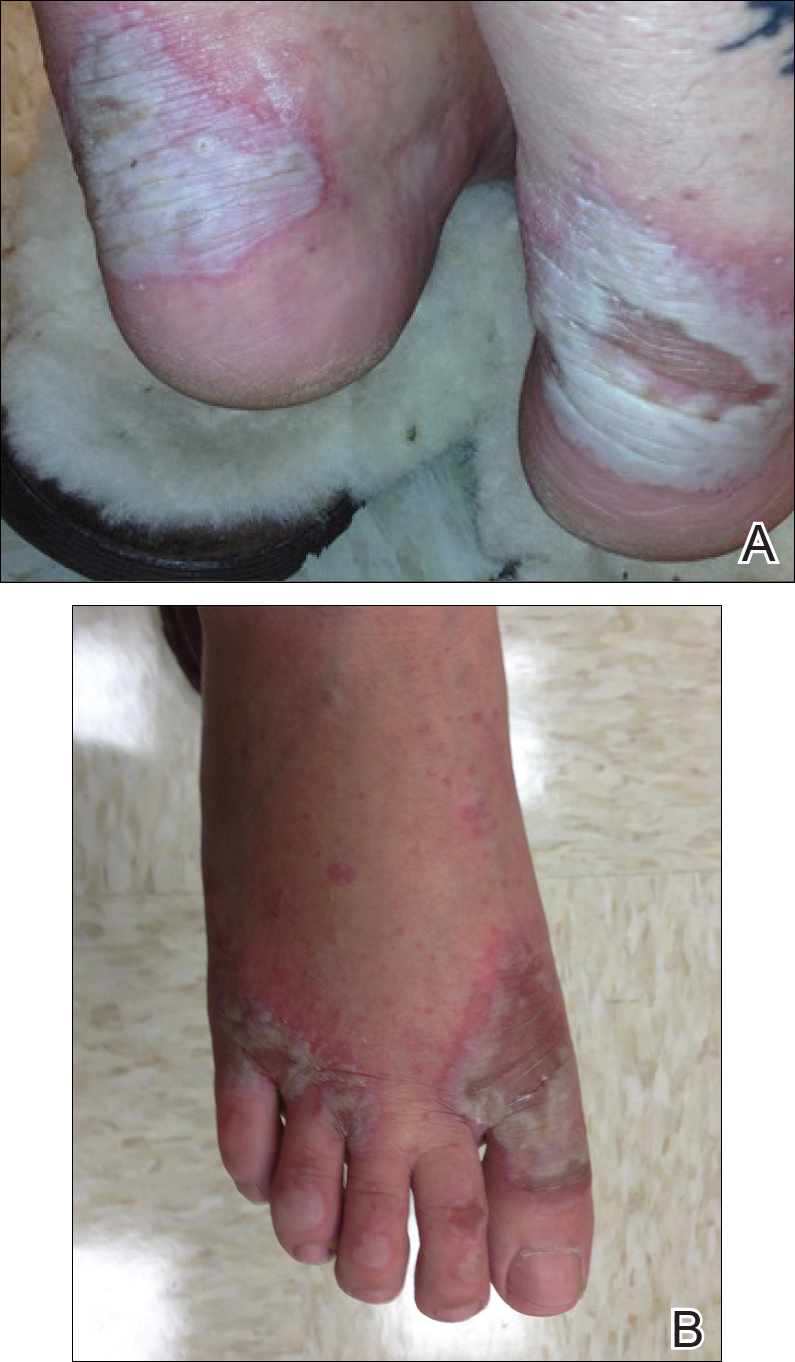
Suspecting a diagnosis of acrodermatitis enteropathica (AE), laboratory testing included a serum zinc level, which was 42 µg/dL (reference range, 70–130 µg/dL). The copper and selenium levels also were low with values of 71 µg/dL (reference range, 80–155 µg/dL) and 31 µg/dL (reference range, 79–326 µg/dL), respectively. No additional vitamin or mineral deficiencies were discovered. A complete blood cell count and comprehensive metabolic panel were performed and showed no abnormalities other than a mildly elevated sodium level of 147 mEq/L (reference range, 136–142 mEq/L).
A punch biopsy was performed. Histopathology revealed subcorneal neutrophils and neutrophilic crust, mild spongiosis, and a dense upper dermal mixed neutrophilic and lymphohistiocytic infiltrate. The specimen also exhibited mild intercellular edema and prominent capillaries (Figure 3).

After further investigation, the company providing the patient’s TPN confirmed that zinc had been removed several weeks prior to the onset of symptoms due to a critical national shortage of trace element additives. Zinc was supplemented at 15 mg daily to the TPN solution. Three days later a skin examination revealed dramatic changes with notable improvement of the finger web plaques and complete resolution of the facial lesions. The plaques and bullae on the lower extremities also had resolved (Figure 4).
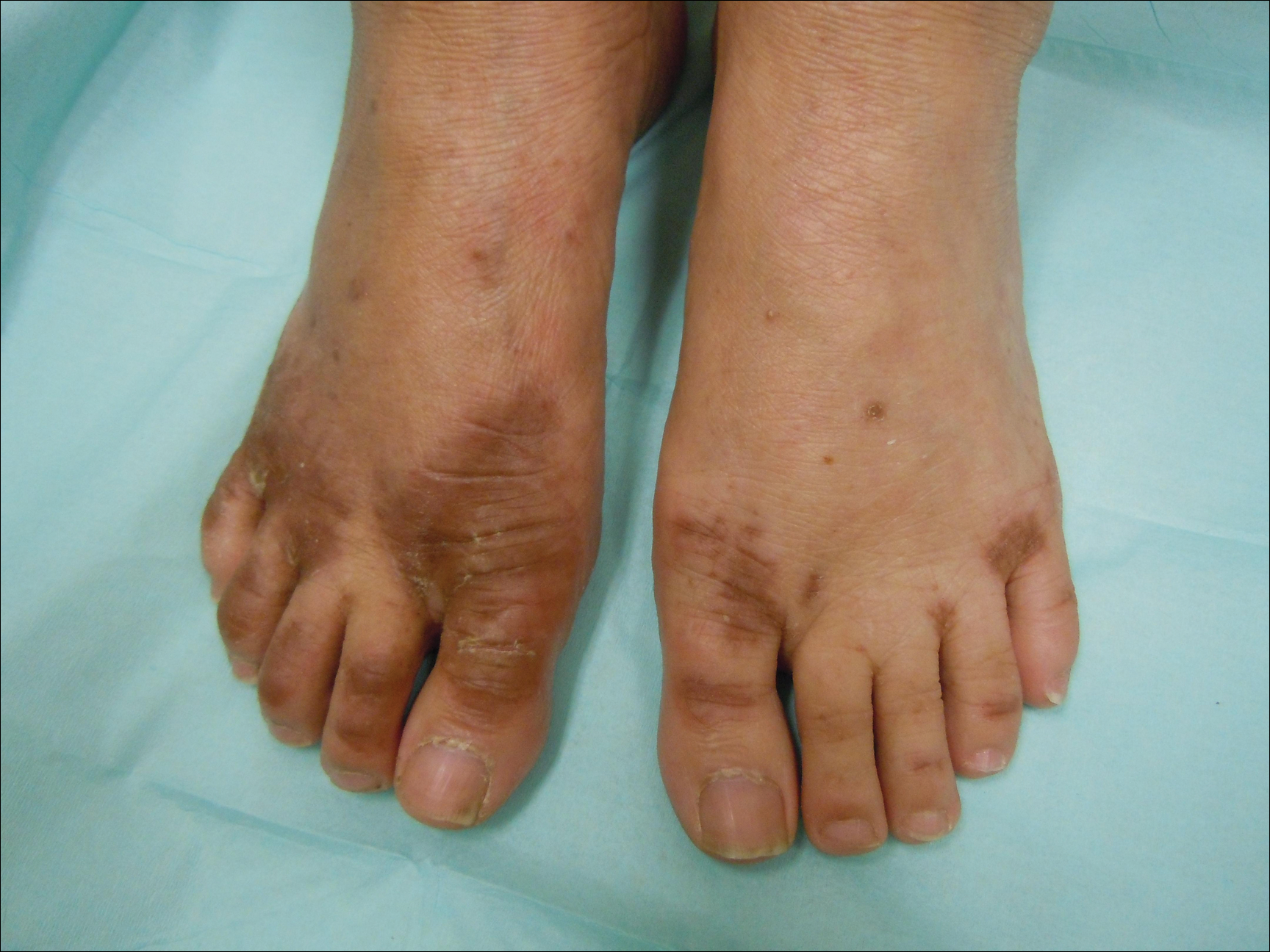
Comment
Background
Acrodermatitis enteropathica is a rare autosomal-recessive disorder of zinc metabolism characterized by skin lesions predominantly distributed in acral and periorificial sites as well as alopecia and diarrhea. Acrodermatitis enteropathica was first described by Brandt1 in 1936 and later characterized by Danbolt and Closs2 in 1942 as a unique and often fatal disease of unknown etiology. More than 30 years later, the link between zinc deficiency and AE was illustrated by Moynahan3 who demonstrated clinical improvement with zinc supplementation. It was not until 2002 that the molecular pathogenesis of hypozincemia in patients with inherited AE was described. Küry et al4 identified a mutation in the SLC39A4 gene responsible for encoding the Zip4 protein, a zinc transporter found on enterocytes, particularly in the proximal small intestine.5,6 Classically, patients with inherited AE are children who present within days of birth or days to weeks after being weaned from breast milk to cow’s milk. The zinc in bovine milk is less bioavailable than breast milk, though both have similar total zinc concentrations, which results in the decreased plasma zinc levels seen in children with inherited AE.5-8 Occasionally, children present before weaning due to decreased maternal mammary zinc secretion (lactogenic AE).9,10
Clinical Presentation
Similar clinical findings are seen in patients with noninherited forms of zinc deficiency known as acquired AE. Acquired zinc deficiency may be broadly categorized as being from inadequate intake, deficient absorption, excess demand, or overexcretion.8 Such disturbances of zinc balance are most frequently seen in patients with restrictive diets, anorexia nervosa, intestinal bypass procedures, Crohn disease, pancreatic insufficiency, alcoholism, human immunodeficiency virus, and extensive cutaneous burns. Premature infants, mothers who are breastfeeding, and those dependent on TPN are at risk for developing acquired zinc deficiency.7-9,11
Differentiating Characteristics
Both acquired and inherited AE present as erythematous or pink eczematous scaly plaques with the variable presence of vesicular or bullous lesions involving periorificial, acral, and anogenital regions. Early manifestations of AE may include angular cheilitis and paronychia. Alopecia and diarrhea are characteristics of later disease. In fact, the complete triad of dermatitis, alopecia, and diarrhea is seen in only 20% of cases.7 Without treatment, patients may develop blepharitis, conjunctivitis, photophobia, irritability, anorexia, apathy, growth retardation, hypogonadism, hypogeusia, and mental slowing. Skin lesions frequently become secondarily infected with Candida albicans and/or bacteria.5,7,11
Histopathology
Histopathologic examination of skin biopsy specimens from AE lesions demonstrates nonspecific findings similar to other deficiency dermatoses, such as pellagra and glucagonoma-associated necrolytic migratory erythema. Histology typically reveals cytoplasmic pallor with vacuolization and ballooning degeneration of keratinocytes, followed by confluent keratinocyte necrosis within the stratum granulosum and stratum spinosum of the epidermis.5 Confluent parakeratosis with hypogranulosis variably associated with neutrophil crust also is seen. Scattered dyskeratotic keratinocytes may be found within all levels of the epidermis. In resolving or chronic AE lesions, psoriasiform hyperplasia is prevalent, though necrolysis may be minimal or absent.5,11
Diagnosis
Evaluation includes measurement of plasma zinc levels. Zinc levels less than 50 µg/dL are suggestive but not diagnostic of AE.5 Although plasma zinc measurement is the most useful indicator of zinc status, its utility in assessing the true total body store of zinc is limited. Plasma zinc is tightly regulated and only represents 0.1% of body stores.5,6 Additionally, zinc levels may decrease in proinflammatory states.12 Beyond zinc measurement, evaluation of alkaline phosphatase, a zinc-dependent enzyme, can provide useful diagnostic information.5,6
Zinc and TPN
Patients on TPN are at a unique risk for developing zinc and other nutritional deficiencies. Because the daily recommended dietary allowance for zinc is low (8 mg daily for adult women and 11 mg daily for adult men)5 and the element is found in a wide variety of foods, maintaining adequate zinc levels is easily achieved in healthy individuals with normal diets. Kay et al13 described 4 patients on parenteral nutrition who developed hypozincemia and an AE-like syndrome within weeks of TPN induction. The authors described rapid and drastic clinical improvement after initiating zinc supplementation, accentuating the importance of including zinc as a component of TPN.13,14 Brazin et al15 also reported a case of an AE-like syndrome from zinc-deficient hyperalimentation in a patient receiving TPN for short bowel syndrome. Chun et al16 described another case of acquired AE in a patient on TPN for acute pancreatitis. Both cases demonstrated prompt improvement of skin lesions after treatment with zinc supplementation. Other nutrient deficiencies may reveal themselves through similar dermatologic manifestations. For example, cases of scaly dermatitis secondary to the development of essential fatty acid deficiency from TPN formulations lacking adequate quantities of linoleic acid have been reported.Similar to our case, the resolution of skin lesions was seen after TPN was supplemented with the deficient nutrient.17 These cases exemplify the importance in considering deficiency dermatoses in the TPN-dependent patient population.
Conclusion
In our case, the development of skin lesions directly coincided with a recent removal of zinc from the patient’s TPN, which provided us with a unique opportunity to observe the causal relationship between decreased zinc intake and the development of clinical signs of acquired AE. This association was further elucidated by laboratory confirmation of low serum zinc levels and rapid improvement in all skin lesions after zinc supplementation was initiated.
- Brandt T. Dermatitis in children with disturbances of general condition and absorption of food. Acta Derm Venereol. 1936;17:513-537.
- Danbolt N, Closs K. Acrodermatitis enteropathica. Acta Derm Venereol. 1942;23:127-169.
- Moynahan E. Acrodermatitis enteropathica: a lethal inherited human zinc deficiency disorder. Lancet. 1974;2:299-400.
- Küry S, Dréno B, Bézieau S, et al. Identification of SLC39A4, a gene involved in acrodermatitis enteropathica. Nat Genet. 2002;31:238-240.
- Maverakis E, Fung MA, Lynch PJ, et al. Acrodermatitis enteropathica and an overview of zinc metabolism. J Am Acad Dermatol. 2007;56:116-124.
- Thrash B, Patel M, Shah KR, et al. Cutaneous manifestations of gastrointestinal disease: part II. J Am Acad Dermatol. 2013;68:211.e1-211.e33; quiz 244-246.
- Perafán-Riveros C, França LF, Alves AC, et al. Acrodermatitis enteropathica: case report and review of the literature. Pediatr Dermatol. 2002;19:426-431.
- Kumar P, Ranjan NR, Mondal AK. Zinc and skin: a brief summary. Dermatol Online J. 2012;18:1.
- Saritha M, Gupta D, Chandrashekar L, et al. Acquired zinc deficiency in an adult female. Indian J Dermatol. 2012;57:492-494.
- Neldner K, Hambidge K, Walravens P. Acrodermatitis enteropathica.Int J Dermatol. 1978;17:380-387.
- Gehrig K, Dinulos J. Acrodermatitis due to nutritional deficiency. Curr Opin Pediatr. 2010;22:107-112.
- Liuzzi JP, Lichten LA, Rivera S, et al. Interleukin-6 regulates the zinc transporter Zip14 in liver and contributes to hypozincemia of the acute-phase response. Proct Natl Acad Sci U S A. 2005;102:6843-6848.
- Kay RG, Tasman-Jones C, Pybus J, et al. A syndrome of acute zinc deficiency during total parenteral nutrition in man. Ann Surg. 1976;183:331-340.
- Jeejeebhoy K. Zinc: an essential trace element for parenteral nutrition. Gastroenterology. 2009;137(5 suppl):S7-S12.
- Brazin SA, Johnson WT, Abramson LJ. The acrodermatitis enteropathica-like syndrome. Arch Dermatol. 1979;115:597-599.
- Chun JH, Baek JH, Chung NG. Development of bullous acrodermatitis enteropathica during the course of chemotherapy for acute lymphocytic leukemia. Ann Dermatol. 2011;23(suppl 3):S326-S328.
- Roongpisuthipong W, Phanachet P, Roongpisuthipong C, et al. Essential fatty acid deficiency while a patient receiving fat regimen total parenteral nutrition [published June 14, 2012]. BMJ Case Rep. doi:10.1136/bcr.07.2011.4475.
Case Report
A 54-year-old woman presented with a pruritic and slightly painful skin eruption that began perinasally and progressed over 1 week to involve the labial commissures, finger webs, dorsal surfaces of the feet, heels, and bilateral gluteal folds. In addition, the eruption involved the left thigh at the donor site of a prior skin graft. She received no relief after an intramuscular steroid injection and hydrocortisone cream 1% prescribed by a primary care physician who diagnosed the rash as poison ivy contact dermatitis despite no exposure to plants. Review of systems was negative and she denied any new medication use. Her medical history was notable for extensive mesenteric injury secondary to a motor vehicle accident. She subsequently had multiple enterocutaneous fistulas that resulted in a complete small bowel enterectomy 10 months prior to presentation, which caused her to become dependent on total parenteral nutrition (TPN).
Physical examination revealed sharply demarcated, erythematous, scaly plaques perinasally, periorally, and on the bilateral gluteal folds (Figure 1). There were sharply demarcated, erythematous, scaly plaques on the right and left finger webs, dorsal surface of the right foot, and left upper thigh. Hemorrhagic bullae were appreciated on the left finger webs. Large flaccid bullae were present on the bilateral heels and dorsum of the right foot (Figure 2).


Suspecting a diagnosis of acrodermatitis enteropathica (AE), laboratory testing included a serum zinc level, which was 42 µg/dL (reference range, 70–130 µg/dL). The copper and selenium levels also were low with values of 71 µg/dL (reference range, 80–155 µg/dL) and 31 µg/dL (reference range, 79–326 µg/dL), respectively. No additional vitamin or mineral deficiencies were discovered. A complete blood cell count and comprehensive metabolic panel were performed and showed no abnormalities other than a mildly elevated sodium level of 147 mEq/L (reference range, 136–142 mEq/L).
A punch biopsy was performed. Histopathology revealed subcorneal neutrophils and neutrophilic crust, mild spongiosis, and a dense upper dermal mixed neutrophilic and lymphohistiocytic infiltrate. The specimen also exhibited mild intercellular edema and prominent capillaries (Figure 3).

After further investigation, the company providing the patient’s TPN confirmed that zinc had been removed several weeks prior to the onset of symptoms due to a critical national shortage of trace element additives. Zinc was supplemented at 15 mg daily to the TPN solution. Three days later a skin examination revealed dramatic changes with notable improvement of the finger web plaques and complete resolution of the facial lesions. The plaques and bullae on the lower extremities also had resolved (Figure 4).

Comment
Background
Acrodermatitis enteropathica is a rare autosomal-recessive disorder of zinc metabolism characterized by skin lesions predominantly distributed in acral and periorificial sites as well as alopecia and diarrhea. Acrodermatitis enteropathica was first described by Brandt1 in 1936 and later characterized by Danbolt and Closs2 in 1942 as a unique and often fatal disease of unknown etiology. More than 30 years later, the link between zinc deficiency and AE was illustrated by Moynahan3 who demonstrated clinical improvement with zinc supplementation. It was not until 2002 that the molecular pathogenesis of hypozincemia in patients with inherited AE was described. Küry et al4 identified a mutation in the SLC39A4 gene responsible for encoding the Zip4 protein, a zinc transporter found on enterocytes, particularly in the proximal small intestine.5,6 Classically, patients with inherited AE are children who present within days of birth or days to weeks after being weaned from breast milk to cow’s milk. The zinc in bovine milk is less bioavailable than breast milk, though both have similar total zinc concentrations, which results in the decreased plasma zinc levels seen in children with inherited AE.5-8 Occasionally, children present before weaning due to decreased maternal mammary zinc secretion (lactogenic AE).9,10
Clinical Presentation
Similar clinical findings are seen in patients with noninherited forms of zinc deficiency known as acquired AE. Acquired zinc deficiency may be broadly categorized as being from inadequate intake, deficient absorption, excess demand, or overexcretion.8 Such disturbances of zinc balance are most frequently seen in patients with restrictive diets, anorexia nervosa, intestinal bypass procedures, Crohn disease, pancreatic insufficiency, alcoholism, human immunodeficiency virus, and extensive cutaneous burns. Premature infants, mothers who are breastfeeding, and those dependent on TPN are at risk for developing acquired zinc deficiency.7-9,11
Differentiating Characteristics
Both acquired and inherited AE present as erythematous or pink eczematous scaly plaques with the variable presence of vesicular or bullous lesions involving periorificial, acral, and anogenital regions. Early manifestations of AE may include angular cheilitis and paronychia. Alopecia and diarrhea are characteristics of later disease. In fact, the complete triad of dermatitis, alopecia, and diarrhea is seen in only 20% of cases.7 Without treatment, patients may develop blepharitis, conjunctivitis, photophobia, irritability, anorexia, apathy, growth retardation, hypogonadism, hypogeusia, and mental slowing. Skin lesions frequently become secondarily infected with Candida albicans and/or bacteria.5,7,11
Histopathology
Histopathologic examination of skin biopsy specimens from AE lesions demonstrates nonspecific findings similar to other deficiency dermatoses, such as pellagra and glucagonoma-associated necrolytic migratory erythema. Histology typically reveals cytoplasmic pallor with vacuolization and ballooning degeneration of keratinocytes, followed by confluent keratinocyte necrosis within the stratum granulosum and stratum spinosum of the epidermis.5 Confluent parakeratosis with hypogranulosis variably associated with neutrophil crust also is seen. Scattered dyskeratotic keratinocytes may be found within all levels of the epidermis. In resolving or chronic AE lesions, psoriasiform hyperplasia is prevalent, though necrolysis may be minimal or absent.5,11
Diagnosis
Evaluation includes measurement of plasma zinc levels. Zinc levels less than 50 µg/dL are suggestive but not diagnostic of AE.5 Although plasma zinc measurement is the most useful indicator of zinc status, its utility in assessing the true total body store of zinc is limited. Plasma zinc is tightly regulated and only represents 0.1% of body stores.5,6 Additionally, zinc levels may decrease in proinflammatory states.12 Beyond zinc measurement, evaluation of alkaline phosphatase, a zinc-dependent enzyme, can provide useful diagnostic information.5,6
Zinc and TPN
Patients on TPN are at a unique risk for developing zinc and other nutritional deficiencies. Because the daily recommended dietary allowance for zinc is low (8 mg daily for adult women and 11 mg daily for adult men)5 and the element is found in a wide variety of foods, maintaining adequate zinc levels is easily achieved in healthy individuals with normal diets. Kay et al13 described 4 patients on parenteral nutrition who developed hypozincemia and an AE-like syndrome within weeks of TPN induction. The authors described rapid and drastic clinical improvement after initiating zinc supplementation, accentuating the importance of including zinc as a component of TPN.13,14 Brazin et al15 also reported a case of an AE-like syndrome from zinc-deficient hyperalimentation in a patient receiving TPN for short bowel syndrome. Chun et al16 described another case of acquired AE in a patient on TPN for acute pancreatitis. Both cases demonstrated prompt improvement of skin lesions after treatment with zinc supplementation. Other nutrient deficiencies may reveal themselves through similar dermatologic manifestations. For example, cases of scaly dermatitis secondary to the development of essential fatty acid deficiency from TPN formulations lacking adequate quantities of linoleic acid have been reported.Similar to our case, the resolution of skin lesions was seen after TPN was supplemented with the deficient nutrient.17 These cases exemplify the importance in considering deficiency dermatoses in the TPN-dependent patient population.
Conclusion
In our case, the development of skin lesions directly coincided with a recent removal of zinc from the patient’s TPN, which provided us with a unique opportunity to observe the causal relationship between decreased zinc intake and the development of clinical signs of acquired AE. This association was further elucidated by laboratory confirmation of low serum zinc levels and rapid improvement in all skin lesions after zinc supplementation was initiated.
Case Report
A 54-year-old woman presented with a pruritic and slightly painful skin eruption that began perinasally and progressed over 1 week to involve the labial commissures, finger webs, dorsal surfaces of the feet, heels, and bilateral gluteal folds. In addition, the eruption involved the left thigh at the donor site of a prior skin graft. She received no relief after an intramuscular steroid injection and hydrocortisone cream 1% prescribed by a primary care physician who diagnosed the rash as poison ivy contact dermatitis despite no exposure to plants. Review of systems was negative and she denied any new medication use. Her medical history was notable for extensive mesenteric injury secondary to a motor vehicle accident. She subsequently had multiple enterocutaneous fistulas that resulted in a complete small bowel enterectomy 10 months prior to presentation, which caused her to become dependent on total parenteral nutrition (TPN).
Physical examination revealed sharply demarcated, erythematous, scaly plaques perinasally, periorally, and on the bilateral gluteal folds (Figure 1). There were sharply demarcated, erythematous, scaly plaques on the right and left finger webs, dorsal surface of the right foot, and left upper thigh. Hemorrhagic bullae were appreciated on the left finger webs. Large flaccid bullae were present on the bilateral heels and dorsum of the right foot (Figure 2).


Suspecting a diagnosis of acrodermatitis enteropathica (AE), laboratory testing included a serum zinc level, which was 42 µg/dL (reference range, 70–130 µg/dL). The copper and selenium levels also were low with values of 71 µg/dL (reference range, 80–155 µg/dL) and 31 µg/dL (reference range, 79–326 µg/dL), respectively. No additional vitamin or mineral deficiencies were discovered. A complete blood cell count and comprehensive metabolic panel were performed and showed no abnormalities other than a mildly elevated sodium level of 147 mEq/L (reference range, 136–142 mEq/L).
A punch biopsy was performed. Histopathology revealed subcorneal neutrophils and neutrophilic crust, mild spongiosis, and a dense upper dermal mixed neutrophilic and lymphohistiocytic infiltrate. The specimen also exhibited mild intercellular edema and prominent capillaries (Figure 3).

After further investigation, the company providing the patient’s TPN confirmed that zinc had been removed several weeks prior to the onset of symptoms due to a critical national shortage of trace element additives. Zinc was supplemented at 15 mg daily to the TPN solution. Three days later a skin examination revealed dramatic changes with notable improvement of the finger web plaques and complete resolution of the facial lesions. The plaques and bullae on the lower extremities also had resolved (Figure 4).

Comment
Background
Acrodermatitis enteropathica is a rare autosomal-recessive disorder of zinc metabolism characterized by skin lesions predominantly distributed in acral and periorificial sites as well as alopecia and diarrhea. Acrodermatitis enteropathica was first described by Brandt1 in 1936 and later characterized by Danbolt and Closs2 in 1942 as a unique and often fatal disease of unknown etiology. More than 30 years later, the link between zinc deficiency and AE was illustrated by Moynahan3 who demonstrated clinical improvement with zinc supplementation. It was not until 2002 that the molecular pathogenesis of hypozincemia in patients with inherited AE was described. Küry et al4 identified a mutation in the SLC39A4 gene responsible for encoding the Zip4 protein, a zinc transporter found on enterocytes, particularly in the proximal small intestine.5,6 Classically, patients with inherited AE are children who present within days of birth or days to weeks after being weaned from breast milk to cow’s milk. The zinc in bovine milk is less bioavailable than breast milk, though both have similar total zinc concentrations, which results in the decreased plasma zinc levels seen in children with inherited AE.5-8 Occasionally, children present before weaning due to decreased maternal mammary zinc secretion (lactogenic AE).9,10
Clinical Presentation
Similar clinical findings are seen in patients with noninherited forms of zinc deficiency known as acquired AE. Acquired zinc deficiency may be broadly categorized as being from inadequate intake, deficient absorption, excess demand, or overexcretion.8 Such disturbances of zinc balance are most frequently seen in patients with restrictive diets, anorexia nervosa, intestinal bypass procedures, Crohn disease, pancreatic insufficiency, alcoholism, human immunodeficiency virus, and extensive cutaneous burns. Premature infants, mothers who are breastfeeding, and those dependent on TPN are at risk for developing acquired zinc deficiency.7-9,11
Differentiating Characteristics
Both acquired and inherited AE present as erythematous or pink eczematous scaly plaques with the variable presence of vesicular or bullous lesions involving periorificial, acral, and anogenital regions. Early manifestations of AE may include angular cheilitis and paronychia. Alopecia and diarrhea are characteristics of later disease. In fact, the complete triad of dermatitis, alopecia, and diarrhea is seen in only 20% of cases.7 Without treatment, patients may develop blepharitis, conjunctivitis, photophobia, irritability, anorexia, apathy, growth retardation, hypogonadism, hypogeusia, and mental slowing. Skin lesions frequently become secondarily infected with Candida albicans and/or bacteria.5,7,11
Histopathology
Histopathologic examination of skin biopsy specimens from AE lesions demonstrates nonspecific findings similar to other deficiency dermatoses, such as pellagra and glucagonoma-associated necrolytic migratory erythema. Histology typically reveals cytoplasmic pallor with vacuolization and ballooning degeneration of keratinocytes, followed by confluent keratinocyte necrosis within the stratum granulosum and stratum spinosum of the epidermis.5 Confluent parakeratosis with hypogranulosis variably associated with neutrophil crust also is seen. Scattered dyskeratotic keratinocytes may be found within all levels of the epidermis. In resolving or chronic AE lesions, psoriasiform hyperplasia is prevalent, though necrolysis may be minimal or absent.5,11
Diagnosis
Evaluation includes measurement of plasma zinc levels. Zinc levels less than 50 µg/dL are suggestive but not diagnostic of AE.5 Although plasma zinc measurement is the most useful indicator of zinc status, its utility in assessing the true total body store of zinc is limited. Plasma zinc is tightly regulated and only represents 0.1% of body stores.5,6 Additionally, zinc levels may decrease in proinflammatory states.12 Beyond zinc measurement, evaluation of alkaline phosphatase, a zinc-dependent enzyme, can provide useful diagnostic information.5,6
Zinc and TPN
Patients on TPN are at a unique risk for developing zinc and other nutritional deficiencies. Because the daily recommended dietary allowance for zinc is low (8 mg daily for adult women and 11 mg daily for adult men)5 and the element is found in a wide variety of foods, maintaining adequate zinc levels is easily achieved in healthy individuals with normal diets. Kay et al13 described 4 patients on parenteral nutrition who developed hypozincemia and an AE-like syndrome within weeks of TPN induction. The authors described rapid and drastic clinical improvement after initiating zinc supplementation, accentuating the importance of including zinc as a component of TPN.13,14 Brazin et al15 also reported a case of an AE-like syndrome from zinc-deficient hyperalimentation in a patient receiving TPN for short bowel syndrome. Chun et al16 described another case of acquired AE in a patient on TPN for acute pancreatitis. Both cases demonstrated prompt improvement of skin lesions after treatment with zinc supplementation. Other nutrient deficiencies may reveal themselves through similar dermatologic manifestations. For example, cases of scaly dermatitis secondary to the development of essential fatty acid deficiency from TPN formulations lacking adequate quantities of linoleic acid have been reported.Similar to our case, the resolution of skin lesions was seen after TPN was supplemented with the deficient nutrient.17 These cases exemplify the importance in considering deficiency dermatoses in the TPN-dependent patient population.
Conclusion
In our case, the development of skin lesions directly coincided with a recent removal of zinc from the patient’s TPN, which provided us with a unique opportunity to observe the causal relationship between decreased zinc intake and the development of clinical signs of acquired AE. This association was further elucidated by laboratory confirmation of low serum zinc levels and rapid improvement in all skin lesions after zinc supplementation was initiated.
- Brandt T. Dermatitis in children with disturbances of general condition and absorption of food. Acta Derm Venereol. 1936;17:513-537.
- Danbolt N, Closs K. Acrodermatitis enteropathica. Acta Derm Venereol. 1942;23:127-169.
- Moynahan E. Acrodermatitis enteropathica: a lethal inherited human zinc deficiency disorder. Lancet. 1974;2:299-400.
- Küry S, Dréno B, Bézieau S, et al. Identification of SLC39A4, a gene involved in acrodermatitis enteropathica. Nat Genet. 2002;31:238-240.
- Maverakis E, Fung MA, Lynch PJ, et al. Acrodermatitis enteropathica and an overview of zinc metabolism. J Am Acad Dermatol. 2007;56:116-124.
- Thrash B, Patel M, Shah KR, et al. Cutaneous manifestations of gastrointestinal disease: part II. J Am Acad Dermatol. 2013;68:211.e1-211.e33; quiz 244-246.
- Perafán-Riveros C, França LF, Alves AC, et al. Acrodermatitis enteropathica: case report and review of the literature. Pediatr Dermatol. 2002;19:426-431.
- Kumar P, Ranjan NR, Mondal AK. Zinc and skin: a brief summary. Dermatol Online J. 2012;18:1.
- Saritha M, Gupta D, Chandrashekar L, et al. Acquired zinc deficiency in an adult female. Indian J Dermatol. 2012;57:492-494.
- Neldner K, Hambidge K, Walravens P. Acrodermatitis enteropathica.Int J Dermatol. 1978;17:380-387.
- Gehrig K, Dinulos J. Acrodermatitis due to nutritional deficiency. Curr Opin Pediatr. 2010;22:107-112.
- Liuzzi JP, Lichten LA, Rivera S, et al. Interleukin-6 regulates the zinc transporter Zip14 in liver and contributes to hypozincemia of the acute-phase response. Proct Natl Acad Sci U S A. 2005;102:6843-6848.
- Kay RG, Tasman-Jones C, Pybus J, et al. A syndrome of acute zinc deficiency during total parenteral nutrition in man. Ann Surg. 1976;183:331-340.
- Jeejeebhoy K. Zinc: an essential trace element for parenteral nutrition. Gastroenterology. 2009;137(5 suppl):S7-S12.
- Brazin SA, Johnson WT, Abramson LJ. The acrodermatitis enteropathica-like syndrome. Arch Dermatol. 1979;115:597-599.
- Chun JH, Baek JH, Chung NG. Development of bullous acrodermatitis enteropathica during the course of chemotherapy for acute lymphocytic leukemia. Ann Dermatol. 2011;23(suppl 3):S326-S328.
- Roongpisuthipong W, Phanachet P, Roongpisuthipong C, et al. Essential fatty acid deficiency while a patient receiving fat regimen total parenteral nutrition [published June 14, 2012]. BMJ Case Rep. doi:10.1136/bcr.07.2011.4475.
- Brandt T. Dermatitis in children with disturbances of general condition and absorption of food. Acta Derm Venereol. 1936;17:513-537.
- Danbolt N, Closs K. Acrodermatitis enteropathica. Acta Derm Venereol. 1942;23:127-169.
- Moynahan E. Acrodermatitis enteropathica: a lethal inherited human zinc deficiency disorder. Lancet. 1974;2:299-400.
- Küry S, Dréno B, Bézieau S, et al. Identification of SLC39A4, a gene involved in acrodermatitis enteropathica. Nat Genet. 2002;31:238-240.
- Maverakis E, Fung MA, Lynch PJ, et al. Acrodermatitis enteropathica and an overview of zinc metabolism. J Am Acad Dermatol. 2007;56:116-124.
- Thrash B, Patel M, Shah KR, et al. Cutaneous manifestations of gastrointestinal disease: part II. J Am Acad Dermatol. 2013;68:211.e1-211.e33; quiz 244-246.
- Perafán-Riveros C, França LF, Alves AC, et al. Acrodermatitis enteropathica: case report and review of the literature. Pediatr Dermatol. 2002;19:426-431.
- Kumar P, Ranjan NR, Mondal AK. Zinc and skin: a brief summary. Dermatol Online J. 2012;18:1.
- Saritha M, Gupta D, Chandrashekar L, et al. Acquired zinc deficiency in an adult female. Indian J Dermatol. 2012;57:492-494.
- Neldner K, Hambidge K, Walravens P. Acrodermatitis enteropathica.Int J Dermatol. 1978;17:380-387.
- Gehrig K, Dinulos J. Acrodermatitis due to nutritional deficiency. Curr Opin Pediatr. 2010;22:107-112.
- Liuzzi JP, Lichten LA, Rivera S, et al. Interleukin-6 regulates the zinc transporter Zip14 in liver and contributes to hypozincemia of the acute-phase response. Proct Natl Acad Sci U S A. 2005;102:6843-6848.
- Kay RG, Tasman-Jones C, Pybus J, et al. A syndrome of acute zinc deficiency during total parenteral nutrition in man. Ann Surg. 1976;183:331-340.
- Jeejeebhoy K. Zinc: an essential trace element for parenteral nutrition. Gastroenterology. 2009;137(5 suppl):S7-S12.
- Brazin SA, Johnson WT, Abramson LJ. The acrodermatitis enteropathica-like syndrome. Arch Dermatol. 1979;115:597-599.
- Chun JH, Baek JH, Chung NG. Development of bullous acrodermatitis enteropathica during the course of chemotherapy for acute lymphocytic leukemia. Ann Dermatol. 2011;23(suppl 3):S326-S328.
- Roongpisuthipong W, Phanachet P, Roongpisuthipong C, et al. Essential fatty acid deficiency while a patient receiving fat regimen total parenteral nutrition [published June 14, 2012]. BMJ Case Rep. doi:10.1136/bcr.07.2011.4475.
Practice Points
- Acrodermatitis enteropathica (AE) may be acquired or due to a rare autosomal-recessive disorder of zinc absorption.
- Hereditary AE typically becomes symptomatic during infancy, while acquired AE may develop during hypozincemia in patients of any age.
- Both acquired and hereditary AE improve with zinc supplementation.
Atopic Dermatitis and Peanut Allergy Prevention: New Guidelines
Updated Guidelines on Peanut Allergy Prevention in Infants With Atopic Dermatitis
It has been said that “extraordinary claims require extraordinary evidence.”1 In the pursuit of evidence-based medicine, we are encouraged to follow a similar standard, with an emphasis on waiting for multiple studies with good-quality data and high levels of agreement before changing any aspect of our clinical practice. The ostensible purpose is that studies can be flawed, conclusions can be incorrect, or biases can be overlooked. In such cases, acting on questionable results could imperil patients. It is for this reason that so many review articles sometimes frustratingly seem to conclude that further evidence is needed.2
Based on this standard, recently published addendum guidelines from the National Institute of Allergy and Infectious Diseases for prevention of peanut allergy in the United States3 are somewhat striking in that they make fairly bold recommendations based on results from the 2015 Learning Early about Peanut Allergy (LEAP) study,4 a randomized trial evaluating early peanut introduction as a preventive strategy for peanut allergy. Of note, this study was not placebo controlled, was conducted at only 1 site in the United Kingdom, and only included 640 children, though the number of participants was admittedly large for this type of study.4 Arguably, the LEAP study alone does not provide enough evidence upon which to base what essentially amounts to an about-face in the official recommendations for prevention of peanut and other food allergies, which emphasized delayed introduction of high-risk foods, especially in high-risk individuals.5-7 To better understand this shift, we need to briefly explore the context of the addendum guidelines.
As many as one-third of pediatric patients with atopic dermatitis (AD) have food allergies, thus diet often is invoked by patients and providers alike as an underlying cause of the disease.8 Many patients in my practice are so focused on potential food allergies that actual treatment of the affected skin is marginalized and often dismissed as a stopgap that does not address the root of the problem. A 2004 study of 100 children with AD found that diet was manipulated by the parents in 75% of patients in an attempt to manage the disease.9
Patients are not the only ones who consider food allergies to be a driving force in AD. The medical literature indicates that this theory has existed for centuries; for instance, with regard to the relationship between diet and AD, the author of an article from 1830 quipped, “There is probably no subject in which more deeply rooted convictions have been held . . . than the connection between diet and disease, both as regards the causation and treatment of the latter . . .”10 More apropos perhaps is a statement from the 2010 National Institute of Allergy and Infectious Diseases guidelines on food allergy management, which noted that while the expert group “does not mean to imply that AD results from [food allergies], the role of [food allergies] in the pathogenesis and severity of this condition remains controversial.”11
Prior to the LEAP study, food allergy recommendations for clinical practice in the United Kingdom in 199812 and the United States in 200013 recommended excluding allergenic foods (eg, peanuts, tree nuts, soy, milk, eggs) from the diet in infants with a family history of atopy until 3 years of age. However, those recommendations did not seem to be working, when in fact just the opposite was happening. From 1997 until the LEAP study was conducted in 2015, the prevalence of peanut allergy more than quadrupled and became the leading cause of anaphylaxis and death related to food allergy.14 Additionally, study after study concluded that elimination diets did not seem to help most patients with AD.15 As is required in good scientific thinking, when a hypothesis is proven false, other approaches must be considered.
The idea arose that perhaps delaying introduction of allergenic foods was the opposite of the answer.4 The LEAP study tested the notion that peanut allergies are rare in countries where peanuts are introduced early and if telling families to delay introduction of peanuts in infants might actually be causing development of a peanut allergy, and the tests bore fruit. It was found that giving infants peanut-containing foods resulted in a more than 80% reduction in peanut allergy at 5 years of age (P<.001).4 What was perhaps even more interesting was the connection between AD and peanut allergy. An important idea articulated in the LEAP study is in some ways revolutionary: Rather than foods causing AD, it could be that “early environmental exposure (through the skin) to peanut may account for early sensitization, whereas early oral exposure may lead to immune tolerance.”4 This concept—that impaired eczematous skin may actually lead to the development of food allergies—turns the whole thing upside down.
What do these updated guidelines actually suggest? The first guideline focuses on infants with severe AD, egg allergy, or both, who therefore are thought to be at the highest risk for developing peanut allergy.3 Because of the higher baseline risk in this subgroup, measurement of the peanut-specific IgE (peanut sIgE) level, skin prick testing (SPT), or both is strongly recommended before introducing peanut protein into the diet. This testing can be performed by qualified providers as a screening measure, but if positive (≥0.35 kUA/L for peanut sIgE or >2 mm on the peanut SPT), referral to an allergy specialist is warranted. If these studies are negative, it is thought the likelihood of peanut allergy is low, and it is recommended that caregivers introduce age-appropriate peanut-containing foods (eg, peanut butter snack puffs, diluted peanut butter) as early as 4 to 6 months of age. The second guideline recommends that peanut-containing foods should be introduced into the diets of infants with mild or moderate AD at approximately 6 months of age without the need for prior screening via peanut sIgE or SPT. Lastly, the third guideline recommends that caregivers freely introduce peanut-containing foods together with other solid foods in infants without AD or food allergies in accordance with family preference.3
The results of the LEAP study are certainly exciting, and although the theoretical basis makes good scientific sense and the updated guidelines truly address an important and growing problem, there are several issues with this update that are worth considering. Given the constraints of the LEAP study, it certainly seems possible that the results will not be applicable to all populations or foods. More research is needed to ensure that this robust finding applies to other children and to explore the introduction of other allergenic foods, which the LEAP study investigators also emphasized.4
In fairness, the updated guidelines clearly state the quality of evidence of their recommendations and make it clear that expert opinion is playing a large role.3 For the first guideline regarding recommendations for those with severe AD and/or egg allergy, the quality of evidence is deemed moderate, while the contribution of expert opinion is listed as significant. For the second and third guidelines regarding recommendations for mild to moderate AD and those without AD, respectively, the quality of evidence is low and expert opinion is again listed as significant.3
Importantly, delineating severe AD from moderate disease—which is necessary because only severe AD warrants evaluation with peanut sIgE and/or SPT—can be difficult, as the distinction relies on a degree of subjectivity that may vary between specialists. Indeed, 2 publications suggest extending the definition of severe AD to include infants with early-onset AD (<3 months of age) and those with moderate AD not responding to treatment.16,17
Despite these reservations, the updated guidelines represent a breakthrough in understanding in an area truly in need of advancement. Although the evidence may not be exactly extraordinary, the context for these developments and our deeper understanding suggest that we do indeed live in extraordinary times.
- Encyclopaedia Galactica [television transcript]. Cosmos: A Personal Voyage. Public Broadcasting Service. December 14, 1980.
- Ezzo J, Bausell B, Moerman DE, et al. Reviewing the reviews: how strong is the evidence? how clear are the conclusions? Int J Technol Assess Health Care. 2001;17:457-466.
- Togias A, Cooper SF, Acebal ML, et al. Addendum guidelines for the prevention of peanut allergy in the United States: report of the National Institute of Allergy and Infectious Diseases–sponsored expert panel.J Allergy Clin Immunol. 2017;139:29-44.
- Du Toit G, Roberts G, Sayre PH, et al. Randomized trial of peanut consumption in infants at risk for peanut allergy. N Engl J Med. 2015;372:803-813.
- Høst A, Koletzko B, Dreborg S, et al. Dietary products used in infants for treatment and prevention of food allergy. joint statement of the European Society for Paediatric Allergology and Clinical Immunology (ESPACI) Committee on Hypoallergenic Formulas and the European Society for Paediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN) Committee on Nutrition. Arch Dis Child. 1999;81:80-84.
- American Academy of Pediatrics. Committee on Nutrition. hypoallergenic infant formulas. Pediatrics. 2000;106(2, pt 1):346-349.
- Fiocchi A, Assa’ad A, Bahna S; Adverse Reactions to Foods Committee; American College of Allergy, Asthma and Immunology. Food allergy and the introduction of solid foods to infants: a consensus document. Ann Allergy Asthma Immunol. 2006;97:10-20.
- Thompson MM, Hanifin JM. Effective therapy of childhood atopic dermatitis allays food allergy concerns. J Am Acad Dermatol. 2005;53(2 suppl 2):S214-S219.
- Johnston GA, Bilbao RM, Graham-Brown RA. The use of dietary manipulation by parents of children with atopic dermatitis. Br J Dermatol. 2004;150:1186-1189.
- Mackenzie S. The inaugural address on the advantages to be derived from the study of dermatology. BMJ. 1830;1:193-197.
- Boyce JA, Assa’ad A, Burks AW, et al; NIAID-Sponsored Expert Panel. Guidelines for the diagnosis and management of food allergy in the United States: report of the NIAID-sponsored expert panel. J Allergy Clin Immunol. 2010;126(6 suppl):S1-S58.
- Committee on Toxicity of Chemicals in Food, Consumer Products and the Environment. Peanut Allergy. London, England: Department of Health; 1998.
- American Academy of Pediatrics Committee on Nutrition. Hypoallergenic infant formulas. Pediatrics. 2000;106(2, pt 1):346-349.
- Gruchalla RS, Sampson HA. Preventing peanut allergy through early consumption—ready for prime time? N Engl J Med. 2015;372:875-877.
- Lim NR, Lohman ME, Lio PA. The role of elimination diets in atopic dermatitis: a comprehensive review. Pediatr Dermatol. 2017;34:516-527.
- Wong CC, Allen KJ, Orchard D. Changes to infant feeding guidelines: relevance to dermatologists. Australas J Dermatol. 2017;58:e171-e175.
- Martin PE, Eckert JK, Koplin JJ, et al. Which infants with eczema are at risk of food allergy? results from a population-based cohort. Clin Exp Allergy. 2015;45:255-264.
It has been said that “extraordinary claims require extraordinary evidence.”1 In the pursuit of evidence-based medicine, we are encouraged to follow a similar standard, with an emphasis on waiting for multiple studies with good-quality data and high levels of agreement before changing any aspect of our clinical practice. The ostensible purpose is that studies can be flawed, conclusions can be incorrect, or biases can be overlooked. In such cases, acting on questionable results could imperil patients. It is for this reason that so many review articles sometimes frustratingly seem to conclude that further evidence is needed.2
Based on this standard, recently published addendum guidelines from the National Institute of Allergy and Infectious Diseases for prevention of peanut allergy in the United States3 are somewhat striking in that they make fairly bold recommendations based on results from the 2015 Learning Early about Peanut Allergy (LEAP) study,4 a randomized trial evaluating early peanut introduction as a preventive strategy for peanut allergy. Of note, this study was not placebo controlled, was conducted at only 1 site in the United Kingdom, and only included 640 children, though the number of participants was admittedly large for this type of study.4 Arguably, the LEAP study alone does not provide enough evidence upon which to base what essentially amounts to an about-face in the official recommendations for prevention of peanut and other food allergies, which emphasized delayed introduction of high-risk foods, especially in high-risk individuals.5-7 To better understand this shift, we need to briefly explore the context of the addendum guidelines.
As many as one-third of pediatric patients with atopic dermatitis (AD) have food allergies, thus diet often is invoked by patients and providers alike as an underlying cause of the disease.8 Many patients in my practice are so focused on potential food allergies that actual treatment of the affected skin is marginalized and often dismissed as a stopgap that does not address the root of the problem. A 2004 study of 100 children with AD found that diet was manipulated by the parents in 75% of patients in an attempt to manage the disease.9
Patients are not the only ones who consider food allergies to be a driving force in AD. The medical literature indicates that this theory has existed for centuries; for instance, with regard to the relationship between diet and AD, the author of an article from 1830 quipped, “There is probably no subject in which more deeply rooted convictions have been held . . . than the connection between diet and disease, both as regards the causation and treatment of the latter . . .”10 More apropos perhaps is a statement from the 2010 National Institute of Allergy and Infectious Diseases guidelines on food allergy management, which noted that while the expert group “does not mean to imply that AD results from [food allergies], the role of [food allergies] in the pathogenesis and severity of this condition remains controversial.”11
Prior to the LEAP study, food allergy recommendations for clinical practice in the United Kingdom in 199812 and the United States in 200013 recommended excluding allergenic foods (eg, peanuts, tree nuts, soy, milk, eggs) from the diet in infants with a family history of atopy until 3 years of age. However, those recommendations did not seem to be working, when in fact just the opposite was happening. From 1997 until the LEAP study was conducted in 2015, the prevalence of peanut allergy more than quadrupled and became the leading cause of anaphylaxis and death related to food allergy.14 Additionally, study after study concluded that elimination diets did not seem to help most patients with AD.15 As is required in good scientific thinking, when a hypothesis is proven false, other approaches must be considered.
The idea arose that perhaps delaying introduction of allergenic foods was the opposite of the answer.4 The LEAP study tested the notion that peanut allergies are rare in countries where peanuts are introduced early and if telling families to delay introduction of peanuts in infants might actually be causing development of a peanut allergy, and the tests bore fruit. It was found that giving infants peanut-containing foods resulted in a more than 80% reduction in peanut allergy at 5 years of age (P<.001).4 What was perhaps even more interesting was the connection between AD and peanut allergy. An important idea articulated in the LEAP study is in some ways revolutionary: Rather than foods causing AD, it could be that “early environmental exposure (through the skin) to peanut may account for early sensitization, whereas early oral exposure may lead to immune tolerance.”4 This concept—that impaired eczematous skin may actually lead to the development of food allergies—turns the whole thing upside down.
What do these updated guidelines actually suggest? The first guideline focuses on infants with severe AD, egg allergy, or both, who therefore are thought to be at the highest risk for developing peanut allergy.3 Because of the higher baseline risk in this subgroup, measurement of the peanut-specific IgE (peanut sIgE) level, skin prick testing (SPT), or both is strongly recommended before introducing peanut protein into the diet. This testing can be performed by qualified providers as a screening measure, but if positive (≥0.35 kUA/L for peanut sIgE or >2 mm on the peanut SPT), referral to an allergy specialist is warranted. If these studies are negative, it is thought the likelihood of peanut allergy is low, and it is recommended that caregivers introduce age-appropriate peanut-containing foods (eg, peanut butter snack puffs, diluted peanut butter) as early as 4 to 6 months of age. The second guideline recommends that peanut-containing foods should be introduced into the diets of infants with mild or moderate AD at approximately 6 months of age without the need for prior screening via peanut sIgE or SPT. Lastly, the third guideline recommends that caregivers freely introduce peanut-containing foods together with other solid foods in infants without AD or food allergies in accordance with family preference.3
The results of the LEAP study are certainly exciting, and although the theoretical basis makes good scientific sense and the updated guidelines truly address an important and growing problem, there are several issues with this update that are worth considering. Given the constraints of the LEAP study, it certainly seems possible that the results will not be applicable to all populations or foods. More research is needed to ensure that this robust finding applies to other children and to explore the introduction of other allergenic foods, which the LEAP study investigators also emphasized.4
In fairness, the updated guidelines clearly state the quality of evidence of their recommendations and make it clear that expert opinion is playing a large role.3 For the first guideline regarding recommendations for those with severe AD and/or egg allergy, the quality of evidence is deemed moderate, while the contribution of expert opinion is listed as significant. For the second and third guidelines regarding recommendations for mild to moderate AD and those without AD, respectively, the quality of evidence is low and expert opinion is again listed as significant.3
Importantly, delineating severe AD from moderate disease—which is necessary because only severe AD warrants evaluation with peanut sIgE and/or SPT—can be difficult, as the distinction relies on a degree of subjectivity that may vary between specialists. Indeed, 2 publications suggest extending the definition of severe AD to include infants with early-onset AD (<3 months of age) and those with moderate AD not responding to treatment.16,17
Despite these reservations, the updated guidelines represent a breakthrough in understanding in an area truly in need of advancement. Although the evidence may not be exactly extraordinary, the context for these developments and our deeper understanding suggest that we do indeed live in extraordinary times.
It has been said that “extraordinary claims require extraordinary evidence.”1 In the pursuit of evidence-based medicine, we are encouraged to follow a similar standard, with an emphasis on waiting for multiple studies with good-quality data and high levels of agreement before changing any aspect of our clinical practice. The ostensible purpose is that studies can be flawed, conclusions can be incorrect, or biases can be overlooked. In such cases, acting on questionable results could imperil patients. It is for this reason that so many review articles sometimes frustratingly seem to conclude that further evidence is needed.2
Based on this standard, recently published addendum guidelines from the National Institute of Allergy and Infectious Diseases for prevention of peanut allergy in the United States3 are somewhat striking in that they make fairly bold recommendations based on results from the 2015 Learning Early about Peanut Allergy (LEAP) study,4 a randomized trial evaluating early peanut introduction as a preventive strategy for peanut allergy. Of note, this study was not placebo controlled, was conducted at only 1 site in the United Kingdom, and only included 640 children, though the number of participants was admittedly large for this type of study.4 Arguably, the LEAP study alone does not provide enough evidence upon which to base what essentially amounts to an about-face in the official recommendations for prevention of peanut and other food allergies, which emphasized delayed introduction of high-risk foods, especially in high-risk individuals.5-7 To better understand this shift, we need to briefly explore the context of the addendum guidelines.
As many as one-third of pediatric patients with atopic dermatitis (AD) have food allergies, thus diet often is invoked by patients and providers alike as an underlying cause of the disease.8 Many patients in my practice are so focused on potential food allergies that actual treatment of the affected skin is marginalized and often dismissed as a stopgap that does not address the root of the problem. A 2004 study of 100 children with AD found that diet was manipulated by the parents in 75% of patients in an attempt to manage the disease.9
Patients are not the only ones who consider food allergies to be a driving force in AD. The medical literature indicates that this theory has existed for centuries; for instance, with regard to the relationship between diet and AD, the author of an article from 1830 quipped, “There is probably no subject in which more deeply rooted convictions have been held . . . than the connection between diet and disease, both as regards the causation and treatment of the latter . . .”10 More apropos perhaps is a statement from the 2010 National Institute of Allergy and Infectious Diseases guidelines on food allergy management, which noted that while the expert group “does not mean to imply that AD results from [food allergies], the role of [food allergies] in the pathogenesis and severity of this condition remains controversial.”11
Prior to the LEAP study, food allergy recommendations for clinical practice in the United Kingdom in 199812 and the United States in 200013 recommended excluding allergenic foods (eg, peanuts, tree nuts, soy, milk, eggs) from the diet in infants with a family history of atopy until 3 years of age. However, those recommendations did not seem to be working, when in fact just the opposite was happening. From 1997 until the LEAP study was conducted in 2015, the prevalence of peanut allergy more than quadrupled and became the leading cause of anaphylaxis and death related to food allergy.14 Additionally, study after study concluded that elimination diets did not seem to help most patients with AD.15 As is required in good scientific thinking, when a hypothesis is proven false, other approaches must be considered.
The idea arose that perhaps delaying introduction of allergenic foods was the opposite of the answer.4 The LEAP study tested the notion that peanut allergies are rare in countries where peanuts are introduced early and if telling families to delay introduction of peanuts in infants might actually be causing development of a peanut allergy, and the tests bore fruit. It was found that giving infants peanut-containing foods resulted in a more than 80% reduction in peanut allergy at 5 years of age (P<.001).4 What was perhaps even more interesting was the connection between AD and peanut allergy. An important idea articulated in the LEAP study is in some ways revolutionary: Rather than foods causing AD, it could be that “early environmental exposure (through the skin) to peanut may account for early sensitization, whereas early oral exposure may lead to immune tolerance.”4 This concept—that impaired eczematous skin may actually lead to the development of food allergies—turns the whole thing upside down.
What do these updated guidelines actually suggest? The first guideline focuses on infants with severe AD, egg allergy, or both, who therefore are thought to be at the highest risk for developing peanut allergy.3 Because of the higher baseline risk in this subgroup, measurement of the peanut-specific IgE (peanut sIgE) level, skin prick testing (SPT), or both is strongly recommended before introducing peanut protein into the diet. This testing can be performed by qualified providers as a screening measure, but if positive (≥0.35 kUA/L for peanut sIgE or >2 mm on the peanut SPT), referral to an allergy specialist is warranted. If these studies are negative, it is thought the likelihood of peanut allergy is low, and it is recommended that caregivers introduce age-appropriate peanut-containing foods (eg, peanut butter snack puffs, diluted peanut butter) as early as 4 to 6 months of age. The second guideline recommends that peanut-containing foods should be introduced into the diets of infants with mild or moderate AD at approximately 6 months of age without the need for prior screening via peanut sIgE or SPT. Lastly, the third guideline recommends that caregivers freely introduce peanut-containing foods together with other solid foods in infants without AD or food allergies in accordance with family preference.3
The results of the LEAP study are certainly exciting, and although the theoretical basis makes good scientific sense and the updated guidelines truly address an important and growing problem, there are several issues with this update that are worth considering. Given the constraints of the LEAP study, it certainly seems possible that the results will not be applicable to all populations or foods. More research is needed to ensure that this robust finding applies to other children and to explore the introduction of other allergenic foods, which the LEAP study investigators also emphasized.4
In fairness, the updated guidelines clearly state the quality of evidence of their recommendations and make it clear that expert opinion is playing a large role.3 For the first guideline regarding recommendations for those with severe AD and/or egg allergy, the quality of evidence is deemed moderate, while the contribution of expert opinion is listed as significant. For the second and third guidelines regarding recommendations for mild to moderate AD and those without AD, respectively, the quality of evidence is low and expert opinion is again listed as significant.3
Importantly, delineating severe AD from moderate disease—which is necessary because only severe AD warrants evaluation with peanut sIgE and/or SPT—can be difficult, as the distinction relies on a degree of subjectivity that may vary between specialists. Indeed, 2 publications suggest extending the definition of severe AD to include infants with early-onset AD (<3 months of age) and those with moderate AD not responding to treatment.16,17
Despite these reservations, the updated guidelines represent a breakthrough in understanding in an area truly in need of advancement. Although the evidence may not be exactly extraordinary, the context for these developments and our deeper understanding suggest that we do indeed live in extraordinary times.
- Encyclopaedia Galactica [television transcript]. Cosmos: A Personal Voyage. Public Broadcasting Service. December 14, 1980.
- Ezzo J, Bausell B, Moerman DE, et al. Reviewing the reviews: how strong is the evidence? how clear are the conclusions? Int J Technol Assess Health Care. 2001;17:457-466.
- Togias A, Cooper SF, Acebal ML, et al. Addendum guidelines for the prevention of peanut allergy in the United States: report of the National Institute of Allergy and Infectious Diseases–sponsored expert panel.J Allergy Clin Immunol. 2017;139:29-44.
- Du Toit G, Roberts G, Sayre PH, et al. Randomized trial of peanut consumption in infants at risk for peanut allergy. N Engl J Med. 2015;372:803-813.
- Høst A, Koletzko B, Dreborg S, et al. Dietary products used in infants for treatment and prevention of food allergy. joint statement of the European Society for Paediatric Allergology and Clinical Immunology (ESPACI) Committee on Hypoallergenic Formulas and the European Society for Paediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN) Committee on Nutrition. Arch Dis Child. 1999;81:80-84.
- American Academy of Pediatrics. Committee on Nutrition. hypoallergenic infant formulas. Pediatrics. 2000;106(2, pt 1):346-349.
- Fiocchi A, Assa’ad A, Bahna S; Adverse Reactions to Foods Committee; American College of Allergy, Asthma and Immunology. Food allergy and the introduction of solid foods to infants: a consensus document. Ann Allergy Asthma Immunol. 2006;97:10-20.
- Thompson MM, Hanifin JM. Effective therapy of childhood atopic dermatitis allays food allergy concerns. J Am Acad Dermatol. 2005;53(2 suppl 2):S214-S219.
- Johnston GA, Bilbao RM, Graham-Brown RA. The use of dietary manipulation by parents of children with atopic dermatitis. Br J Dermatol. 2004;150:1186-1189.
- Mackenzie S. The inaugural address on the advantages to be derived from the study of dermatology. BMJ. 1830;1:193-197.
- Boyce JA, Assa’ad A, Burks AW, et al; NIAID-Sponsored Expert Panel. Guidelines for the diagnosis and management of food allergy in the United States: report of the NIAID-sponsored expert panel. J Allergy Clin Immunol. 2010;126(6 suppl):S1-S58.
- Committee on Toxicity of Chemicals in Food, Consumer Products and the Environment. Peanut Allergy. London, England: Department of Health; 1998.
- American Academy of Pediatrics Committee on Nutrition. Hypoallergenic infant formulas. Pediatrics. 2000;106(2, pt 1):346-349.
- Gruchalla RS, Sampson HA. Preventing peanut allergy through early consumption—ready for prime time? N Engl J Med. 2015;372:875-877.
- Lim NR, Lohman ME, Lio PA. The role of elimination diets in atopic dermatitis: a comprehensive review. Pediatr Dermatol. 2017;34:516-527.
- Wong CC, Allen KJ, Orchard D. Changes to infant feeding guidelines: relevance to dermatologists. Australas J Dermatol. 2017;58:e171-e175.
- Martin PE, Eckert JK, Koplin JJ, et al. Which infants with eczema are at risk of food allergy? results from a population-based cohort. Clin Exp Allergy. 2015;45:255-264.
- Encyclopaedia Galactica [television transcript]. Cosmos: A Personal Voyage. Public Broadcasting Service. December 14, 1980.
- Ezzo J, Bausell B, Moerman DE, et al. Reviewing the reviews: how strong is the evidence? how clear are the conclusions? Int J Technol Assess Health Care. 2001;17:457-466.
- Togias A, Cooper SF, Acebal ML, et al. Addendum guidelines for the prevention of peanut allergy in the United States: report of the National Institute of Allergy and Infectious Diseases–sponsored expert panel.J Allergy Clin Immunol. 2017;139:29-44.
- Du Toit G, Roberts G, Sayre PH, et al. Randomized trial of peanut consumption in infants at risk for peanut allergy. N Engl J Med. 2015;372:803-813.
- Høst A, Koletzko B, Dreborg S, et al. Dietary products used in infants for treatment and prevention of food allergy. joint statement of the European Society for Paediatric Allergology and Clinical Immunology (ESPACI) Committee on Hypoallergenic Formulas and the European Society for Paediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN) Committee on Nutrition. Arch Dis Child. 1999;81:80-84.
- American Academy of Pediatrics. Committee on Nutrition. hypoallergenic infant formulas. Pediatrics. 2000;106(2, pt 1):346-349.
- Fiocchi A, Assa’ad A, Bahna S; Adverse Reactions to Foods Committee; American College of Allergy, Asthma and Immunology. Food allergy and the introduction of solid foods to infants: a consensus document. Ann Allergy Asthma Immunol. 2006;97:10-20.
- Thompson MM, Hanifin JM. Effective therapy of childhood atopic dermatitis allays food allergy concerns. J Am Acad Dermatol. 2005;53(2 suppl 2):S214-S219.
- Johnston GA, Bilbao RM, Graham-Brown RA. The use of dietary manipulation by parents of children with atopic dermatitis. Br J Dermatol. 2004;150:1186-1189.
- Mackenzie S. The inaugural address on the advantages to be derived from the study of dermatology. BMJ. 1830;1:193-197.
- Boyce JA, Assa’ad A, Burks AW, et al; NIAID-Sponsored Expert Panel. Guidelines for the diagnosis and management of food allergy in the United States: report of the NIAID-sponsored expert panel. J Allergy Clin Immunol. 2010;126(6 suppl):S1-S58.
- Committee on Toxicity of Chemicals in Food, Consumer Products and the Environment. Peanut Allergy. London, England: Department of Health; 1998.
- American Academy of Pediatrics Committee on Nutrition. Hypoallergenic infant formulas. Pediatrics. 2000;106(2, pt 1):346-349.
- Gruchalla RS, Sampson HA. Preventing peanut allergy through early consumption—ready for prime time? N Engl J Med. 2015;372:875-877.
- Lim NR, Lohman ME, Lio PA. The role of elimination diets in atopic dermatitis: a comprehensive review. Pediatr Dermatol. 2017;34:516-527.
- Wong CC, Allen KJ, Orchard D. Changes to infant feeding guidelines: relevance to dermatologists. Australas J Dermatol. 2017;58:e171-e175.
- Martin PE, Eckert JK, Koplin JJ, et al. Which infants with eczema are at risk of food allergy? results from a population-based cohort. Clin Exp Allergy. 2015;45:255-264.