User login
AD severity linked to S. aureus clonal complex types
A new study offers insight into the over time.
The research “suggests that different CC types might harbor different virulence factors and that the patient’s immune system needs to adjust to this,” concluded the authors of the study, published in the British Journal of Dermatology.
There is a strong association between disease severity and colonization with S. aureus in patients with AD, and as many as 90% are colonized with the microbe but, the authors pointed out, it’s not entirely clear how S. aureus affects the development of AD.
They added that there’s been little research into the possible effects of changes in S. aureus clonal types over time. Still, “new studies indicate that specific clonal types could be linked to specific host phenotypes, illustrating that host-microbe interactions might be important for colonization of AD skin,” they said.
The authors, led by Maja-Lisa Clausen, MD, of the department of dermatology at the University of Copenhagen, tracked 63 adult patients with AD at Denmark’s Bispebjerg Hospital from 2013-2015 to a 2016-2017 follow-up period. Their mean age was 36 years.
They analyzed bacterial swabs taken from the nose, lesional skin, and nonlesional skin. Of the 63 participants, 47 (75%) were colonized with S. aureus in at least one location when the study began, and 27 of those (57%) were still colonized at follow-up. Of the 16 patients not colonized at baseline, 7 patients (44%) had become colonized by follow-up.
Of the 27 patients who were colonized at both time points, 14 (52%) had no change in CC type.
Those who were colonized at follow-up in at least one of the three sites sampled had more severe disease, with a mean SCORAD – or disease severity score – of 37, compared with those who were not colonized at that time, with a mean SCORAD of 28 (P = .067).
There was a much bigger gap in mean SCORAD score between the 14 patients who had the same CC type at both baseline and follow-up (a mean score of 30), compared with the 11 patients with different CC types at follow-up (a mean score of 47), a statistically significant difference (P = .03). Mean severity scores went up in those who changed CC types and down in those whose CC types remained the same.
The findings “illustrate that colonization changes over time, and also probably reflect the relapsing course of this disease, as colonization likely occurs in relation to worsening of the eczema,” the study authors wrote. They cautioned that “other factors should be taken into considerations as these are known to influence AD severity, including change in treatment regimens, climate, or other disease.”
Novo Nordisk Foundation funded the study. No relevant disclosures were reported.
SOURCE: Clausen, ML et al. Br J Dermatol. 2018 Aug 2. doi: 10.1111/bjd.17033.
A new study offers insight into the over time.
The research “suggests that different CC types might harbor different virulence factors and that the patient’s immune system needs to adjust to this,” concluded the authors of the study, published in the British Journal of Dermatology.
There is a strong association between disease severity and colonization with S. aureus in patients with AD, and as many as 90% are colonized with the microbe but, the authors pointed out, it’s not entirely clear how S. aureus affects the development of AD.
They added that there’s been little research into the possible effects of changes in S. aureus clonal types over time. Still, “new studies indicate that specific clonal types could be linked to specific host phenotypes, illustrating that host-microbe interactions might be important for colonization of AD skin,” they said.
The authors, led by Maja-Lisa Clausen, MD, of the department of dermatology at the University of Copenhagen, tracked 63 adult patients with AD at Denmark’s Bispebjerg Hospital from 2013-2015 to a 2016-2017 follow-up period. Their mean age was 36 years.
They analyzed bacterial swabs taken from the nose, lesional skin, and nonlesional skin. Of the 63 participants, 47 (75%) were colonized with S. aureus in at least one location when the study began, and 27 of those (57%) were still colonized at follow-up. Of the 16 patients not colonized at baseline, 7 patients (44%) had become colonized by follow-up.
Of the 27 patients who were colonized at both time points, 14 (52%) had no change in CC type.
Those who were colonized at follow-up in at least one of the three sites sampled had more severe disease, with a mean SCORAD – or disease severity score – of 37, compared with those who were not colonized at that time, with a mean SCORAD of 28 (P = .067).
There was a much bigger gap in mean SCORAD score between the 14 patients who had the same CC type at both baseline and follow-up (a mean score of 30), compared with the 11 patients with different CC types at follow-up (a mean score of 47), a statistically significant difference (P = .03). Mean severity scores went up in those who changed CC types and down in those whose CC types remained the same.
The findings “illustrate that colonization changes over time, and also probably reflect the relapsing course of this disease, as colonization likely occurs in relation to worsening of the eczema,” the study authors wrote. They cautioned that “other factors should be taken into considerations as these are known to influence AD severity, including change in treatment regimens, climate, or other disease.”
Novo Nordisk Foundation funded the study. No relevant disclosures were reported.
SOURCE: Clausen, ML et al. Br J Dermatol. 2018 Aug 2. doi: 10.1111/bjd.17033.
A new study offers insight into the over time.
The research “suggests that different CC types might harbor different virulence factors and that the patient’s immune system needs to adjust to this,” concluded the authors of the study, published in the British Journal of Dermatology.
There is a strong association between disease severity and colonization with S. aureus in patients with AD, and as many as 90% are colonized with the microbe but, the authors pointed out, it’s not entirely clear how S. aureus affects the development of AD.
They added that there’s been little research into the possible effects of changes in S. aureus clonal types over time. Still, “new studies indicate that specific clonal types could be linked to specific host phenotypes, illustrating that host-microbe interactions might be important for colonization of AD skin,” they said.
The authors, led by Maja-Lisa Clausen, MD, of the department of dermatology at the University of Copenhagen, tracked 63 adult patients with AD at Denmark’s Bispebjerg Hospital from 2013-2015 to a 2016-2017 follow-up period. Their mean age was 36 years.
They analyzed bacterial swabs taken from the nose, lesional skin, and nonlesional skin. Of the 63 participants, 47 (75%) were colonized with S. aureus in at least one location when the study began, and 27 of those (57%) were still colonized at follow-up. Of the 16 patients not colonized at baseline, 7 patients (44%) had become colonized by follow-up.
Of the 27 patients who were colonized at both time points, 14 (52%) had no change in CC type.
Those who were colonized at follow-up in at least one of the three sites sampled had more severe disease, with a mean SCORAD – or disease severity score – of 37, compared with those who were not colonized at that time, with a mean SCORAD of 28 (P = .067).
There was a much bigger gap in mean SCORAD score between the 14 patients who had the same CC type at both baseline and follow-up (a mean score of 30), compared with the 11 patients with different CC types at follow-up (a mean score of 47), a statistically significant difference (P = .03). Mean severity scores went up in those who changed CC types and down in those whose CC types remained the same.
The findings “illustrate that colonization changes over time, and also probably reflect the relapsing course of this disease, as colonization likely occurs in relation to worsening of the eczema,” the study authors wrote. They cautioned that “other factors should be taken into considerations as these are known to influence AD severity, including change in treatment regimens, climate, or other disease.”
Novo Nordisk Foundation funded the study. No relevant disclosures were reported.
SOURCE: Clausen, ML et al. Br J Dermatol. 2018 Aug 2. doi: 10.1111/bjd.17033.
FROM BRITISH JOURNAL OF DERMATOLOGY
Key clinical point: Changes in skin colonization of Staphylococcus aureus clonal complex (CC) types in patients with atopic dermatitis (AD) over time may be related to relapses.
Major finding: Mean SCORAD among the 14 participants with the same CC type at baseline and follow-up was 30, vs. 47 in the 11 patients with different CC types at follow-up (P = .03).
Study details: The study of 63 adults with AD compared the association of disease severity and colonization with S. aureus, and changes in S. aureus clonal complex types over time.
Disclosures: Novo Nordisk Foundation funded the study. No relevant disclosures were reported.
Source: Clausen ML et al. Br J Dermatol. 2018 Aug 2. doi: 10.1111/bjd.17033.
Topical Corticosteroids for Treatment-Resistant Atopic Dermatitis
Atopic dermatitis (AD) is most often treated with mid-potency topical corticosteroids.1,2 Although this option is effective, not all patients respond to treatment, and those who do may lose efficacy over time, a phenomenon known as tachyphylaxis. The pathophysiology of tachyphylaxis to topical corticosteroids has been ascribed to loss of corticosteroid receptor function,3 but the evidence is weak.3,4 Patients with severe treatment-resistant AD improve when treated with mid-potency topical steroids in an inpatient setting; therefore, treatment resistance to topical corticosteroids may be largely due to poor adherence.5
Patients with treatment-resistant AD generally improve when treated with topical corticosteroids under conditions designed to promote treatment adherence, but this improvement often is reported for study groups, not individual patients. Focusing on group data may not give a clear picture of what is happening at the individual level. In this study, we evaluated changes at an individual level to determine how frequently AD patients who were previously treated with topical corticosteroids unsuccessfully would respond to desoximetasone spray 0.25% under conditions designed to promote good adherence over a 7-day period.
Methods
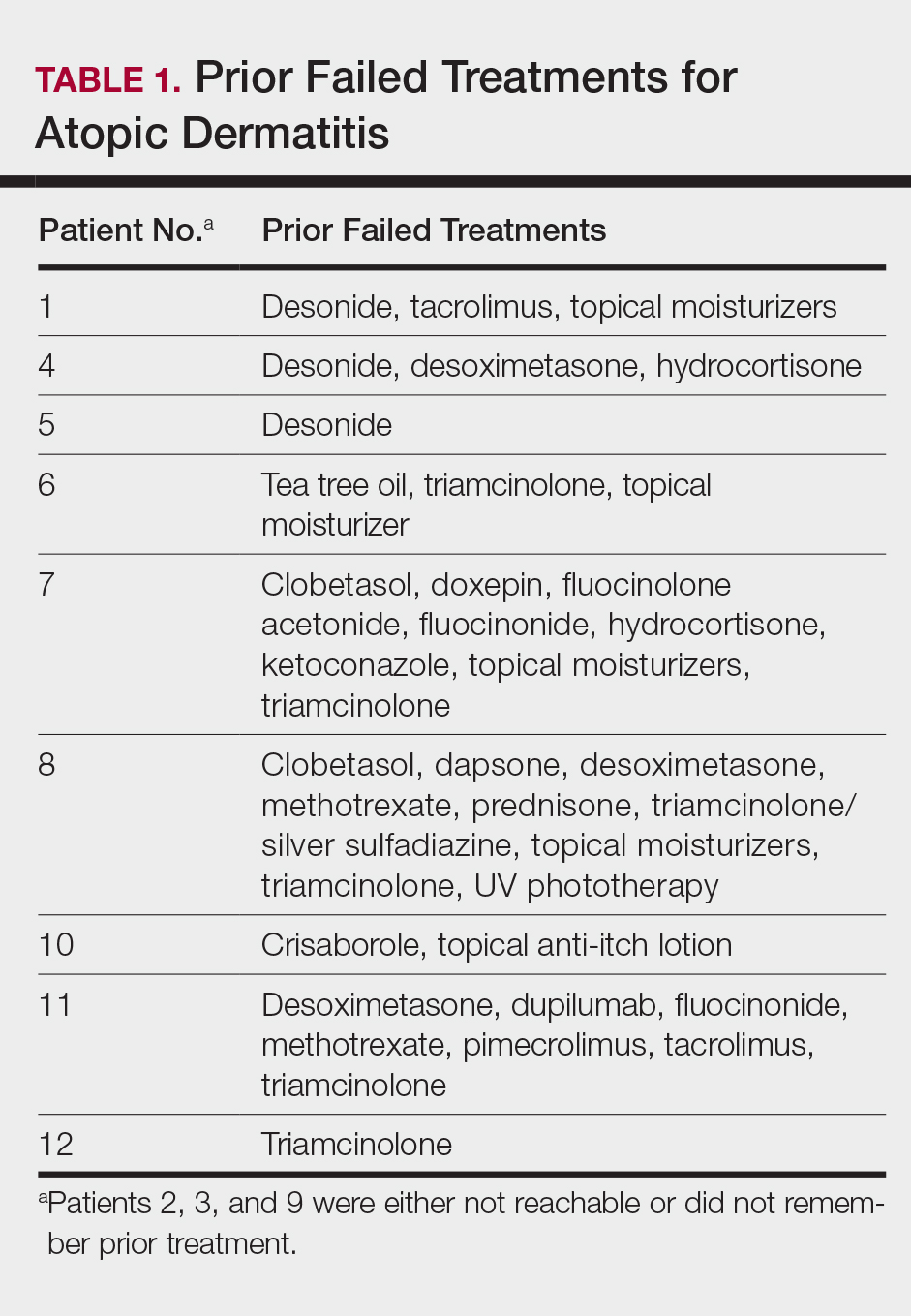
This open-label, randomized, single-center clinical study included 12 patients with AD who were previously unsuccessfully treated with topical corticosteroids in the Department of Dermatology at Wake Forest Baptist Medical Center (Winston-Salem, North Carolina)(Table 1). The study was approved by the local institutional review board.
Inclusion criteria included men and women 18 years or older at baseline who had AD that was considered amenable to therapy with topical corticosteroids by the clinician and were able to comply with the study protocol (Figure). Written informed consent also was obtained from each patient. Women who were pregnant, breastfeeding, or unwilling to practice birth control during participation in the study were excluded. Other exclusion criteria included presence of a condition that in the opinion of the investigator would compromise the safety of the patient or quality of data as well as patients with no access to a telephone throughout the day. Patients diagnosed with conditions affecting adherence to treatment (eg, dementia, Alzheimer disease), those with a history of allergy or sensitivity to corticosteroids, and those with a history of drug hypersensitivity were excluded from the study.
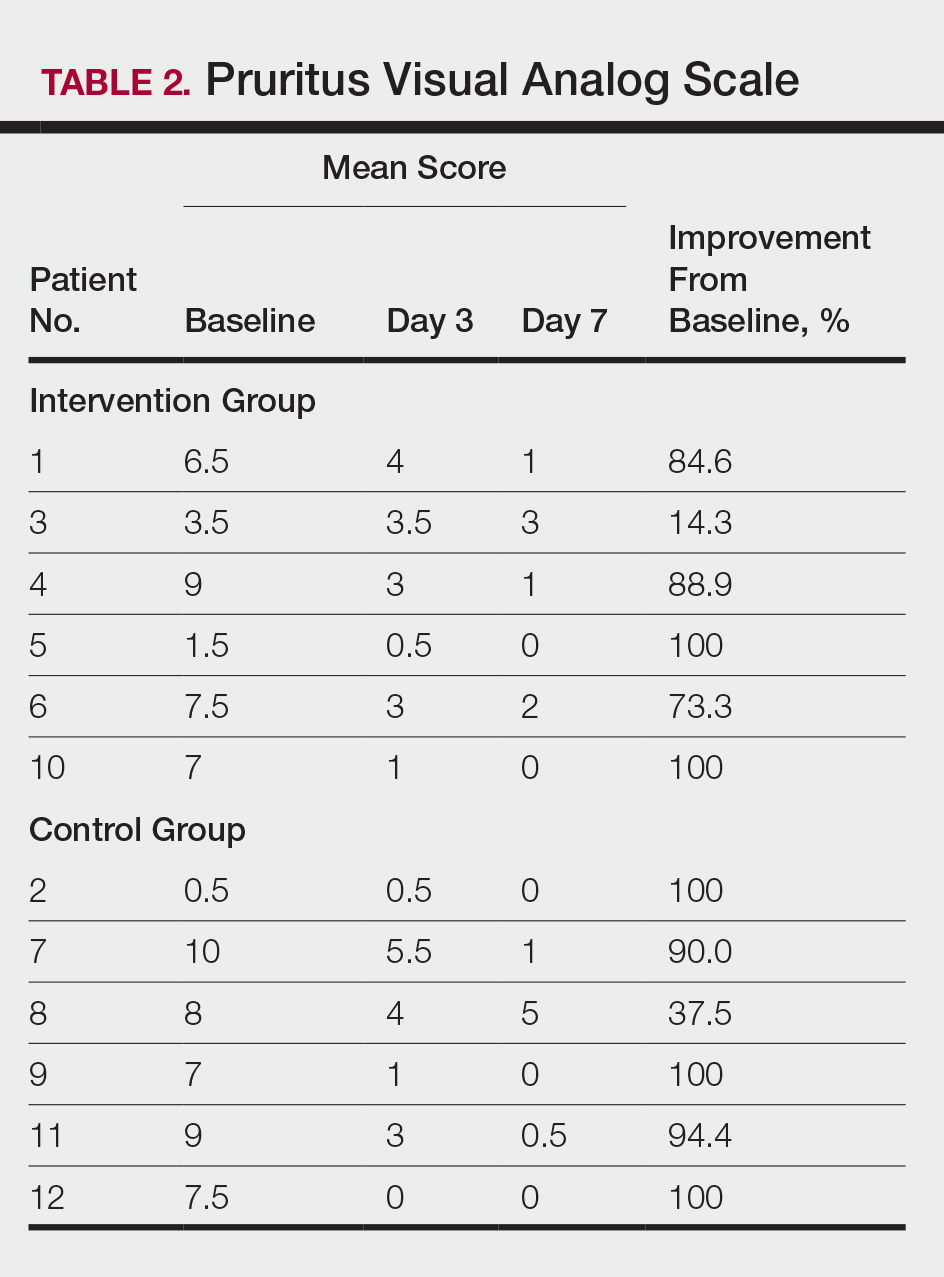
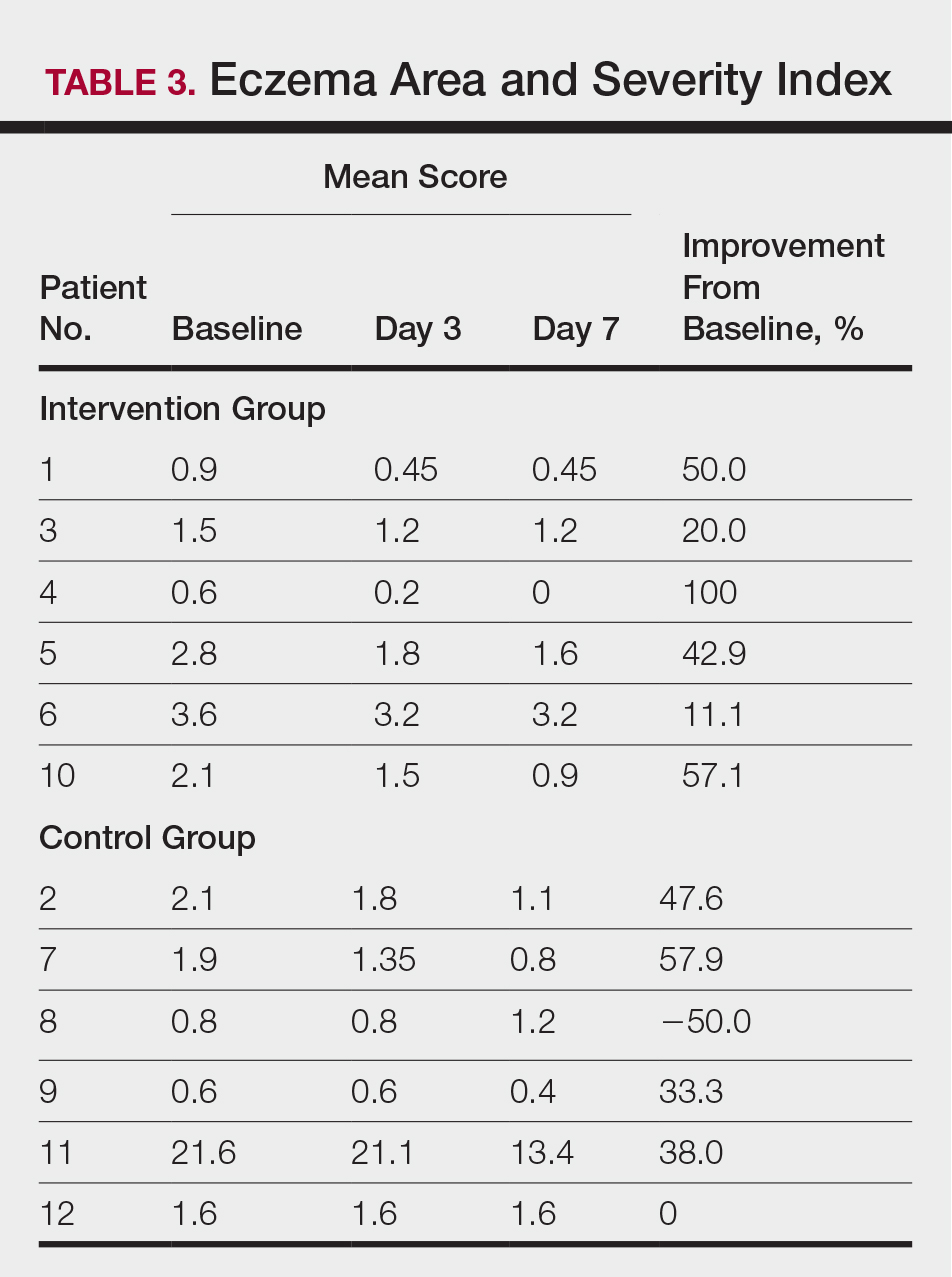
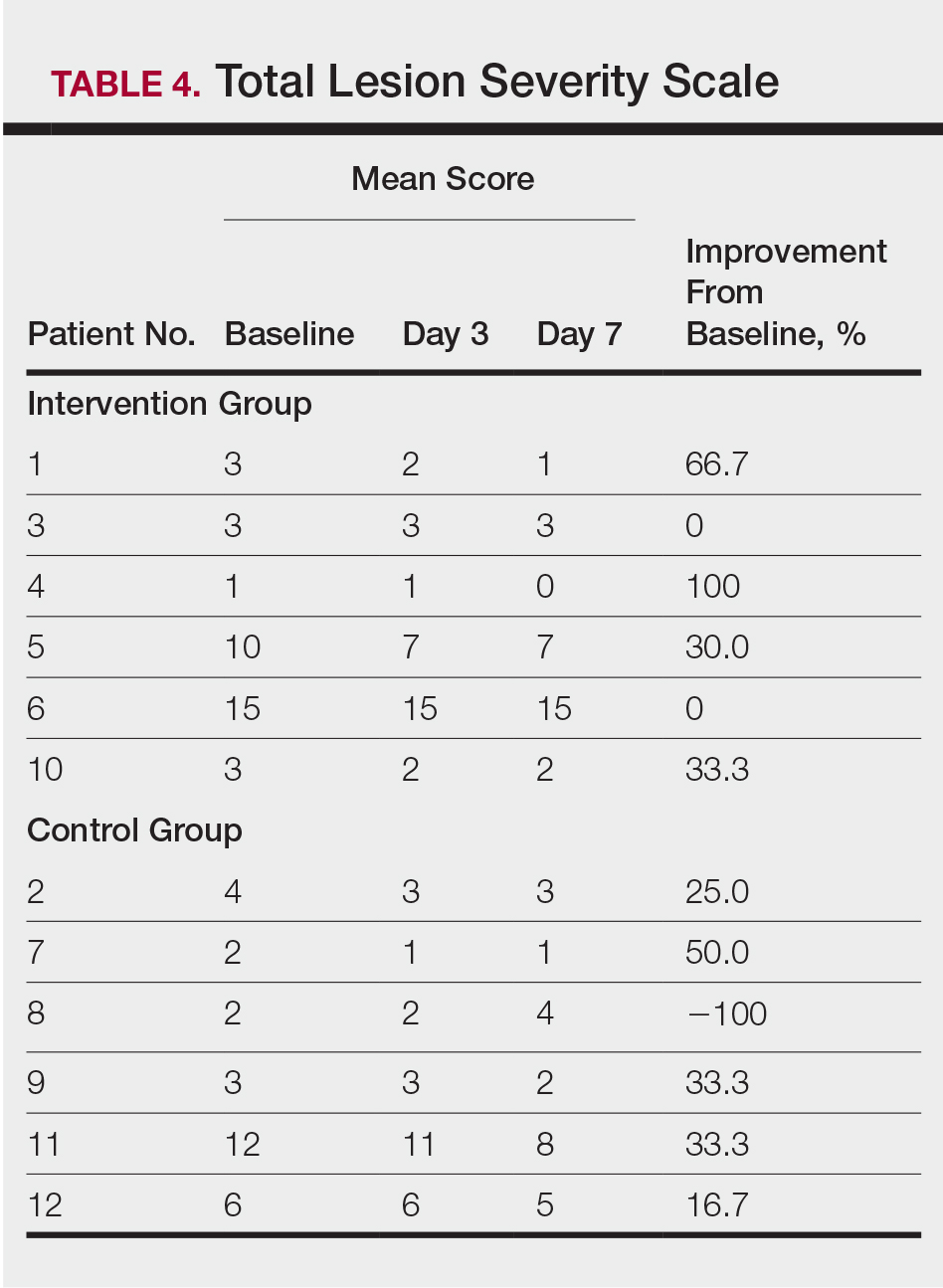
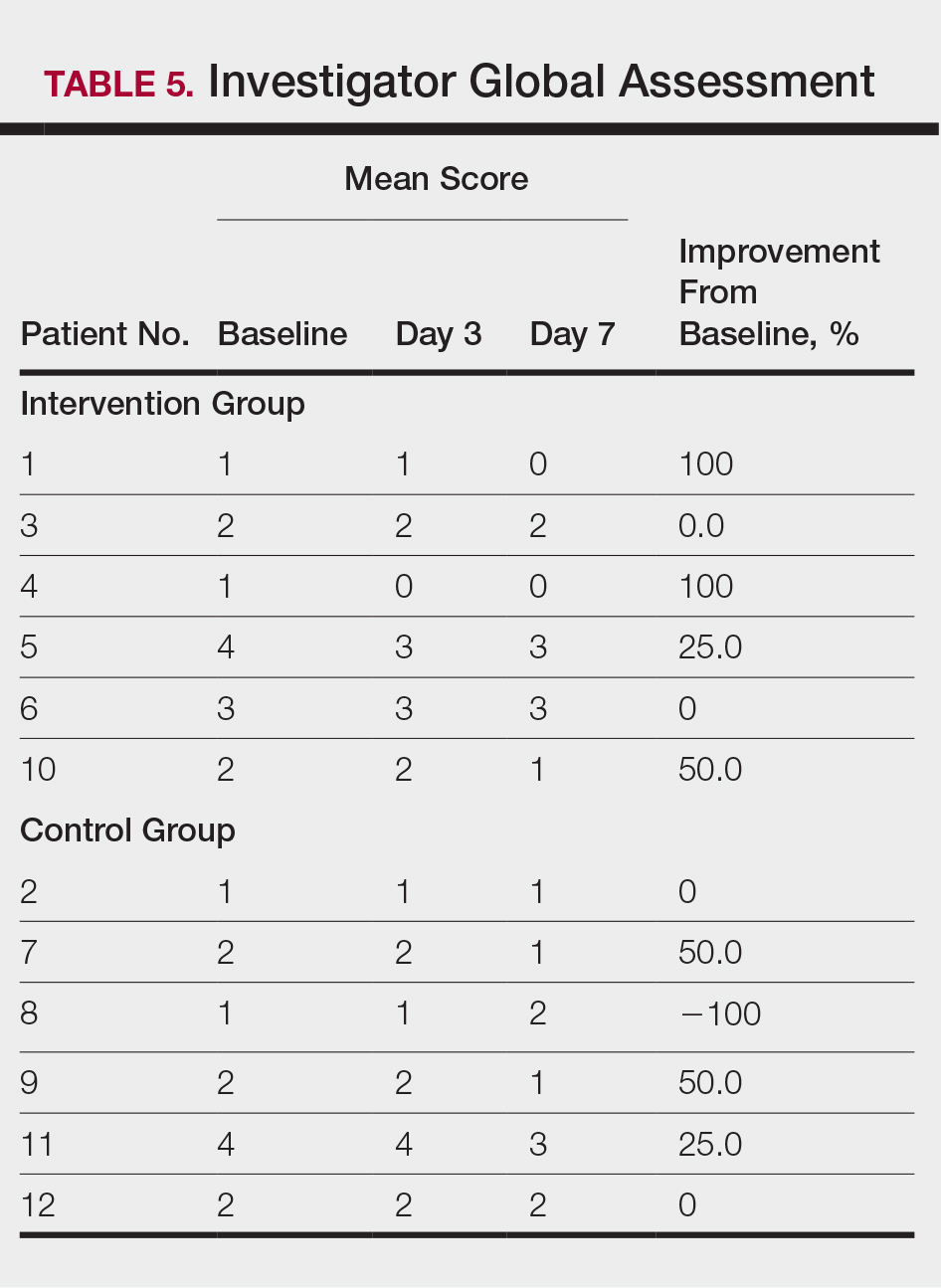
All 12 patients were treated with desoximetasone spray 0.25% for 7 days. Patients were instructed not to use other AD medications during the study period. At baseline, patients were randomized to receive either twice-daily telephone calls to discuss treatment adherence (intervention group) or no telephone calls (control) during the study period. Patients in both the intervention and control groups returned for evaluation on days 3 and 7. During these visits, disease severity was evaluated using the pruritus visual analog scale, Eczema Area and Severity Index (EASI), total lesion severity scale (TLSS), and investigator global assessment (IGA). Descriptive statistics were used to report the outcomes for each patient.
Results
Twelve AD patients who were previously unsuccessfully treated with topical corticosteroids were recruited for the study. Six patients were randomized to the intervention group and 6 were randomized to the control group. Fifty percent of patients were black, 50% were women, and the average age was 50.4 years. All 12 patients completed the study.
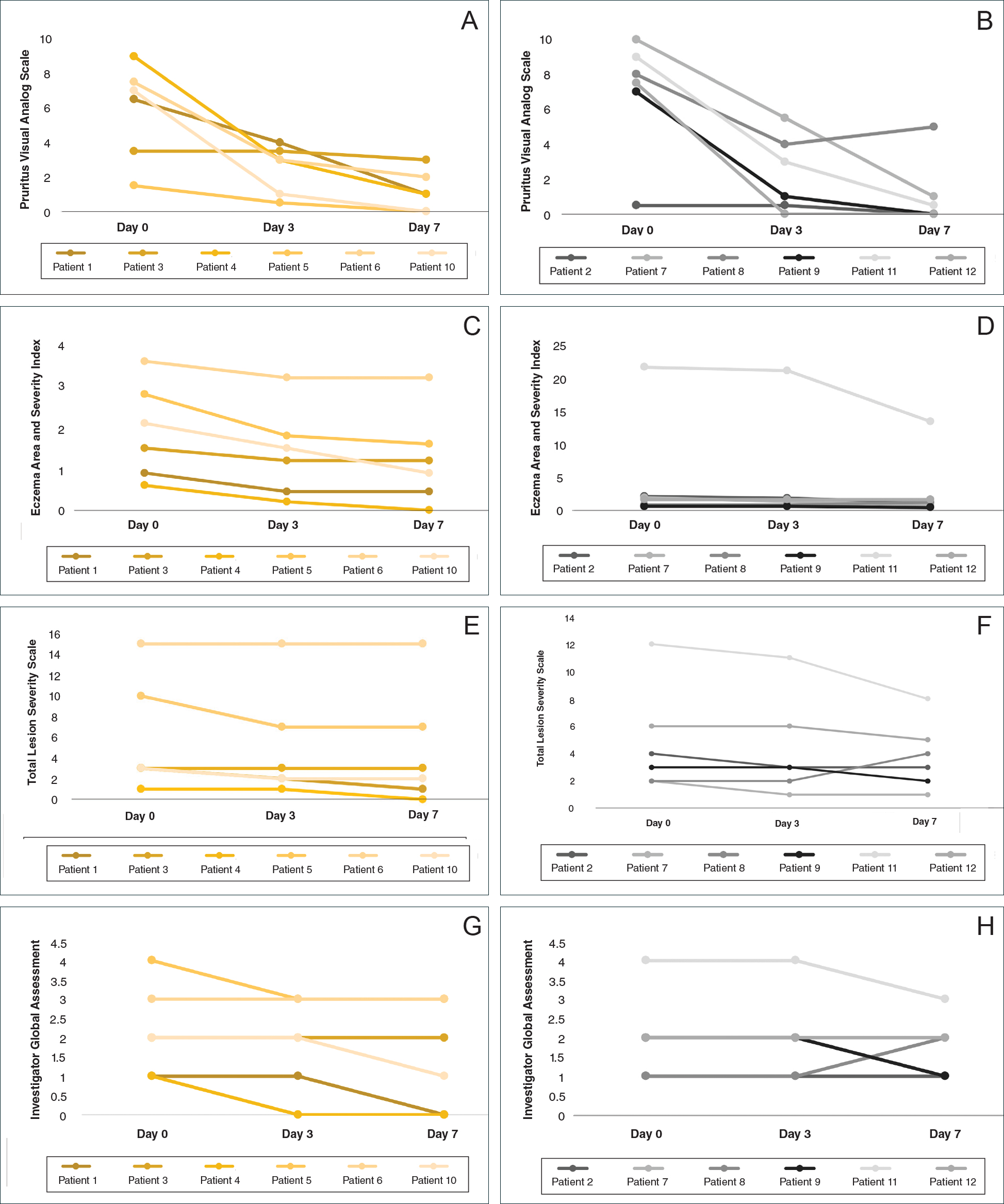
At the end of the study, most patients showed improvement in all evaluation parameters (eFigure). All 12 patients showed improvement in pruritus visual analog scores; 83.3% (10/12) showed improved EASI scores, 75.0% (9/12) showed improved TLSS scores, and 58.3% (7/12) showed improved IGA scores (Tables 2–5). Patients who received telephone calls in the intervention group showed greater improvement compared to those in the control group, except for pruritus; the mean reduction in pruritus was 76.9% in the intervention group versus 87.0% in the control group. The mean improvement in EASI score was 46.9% in the intervention group versus 21.1% in the control group. The mean improvement in TLSS score was 38.3% in the intervention group versus 9.7% in the control group. The mean improvement in IGA score was 45.8% in the intervention group versus 4.2% in the control group. Only one patient in the control group (patient 8) showed lower EASI, TLSS, and IGA scores at baseline.
Comment
Although topical corticosteroids are the mainstay for treatment of AD, many patients report treatment resistance after a period of a few doses or longer.6-9 There is strong evidence demonstrating rapid corticosteroid receptor downregulation in tissues after corticosteroid therapy, which is the accepted mechanism for tachyphylaxis, but the timing of this effect does not match up with clinical experiences. The physiologic significance of corticosteroid agonist-induced receptor downregulation is unknown and may not have any considerable effect on corticosteroid efficacy.3 A systematic review by Taheri et al3 on the development of resistance to topical corticosteroids proposed 2 theories for the underlying pathogenesis of tachyphylaxis: (1) long-term patient nonadherence, and (2) the initial maximal response during the first few weeks of therapy eventually plateaus. Because corticosteroids may plateau after a certain number of doses, natural disease flare-ups during this period may give the wrong impression of tachyphylaxis.10 The treatment “resistance” reported by the patients in our study may have been due to this plateau effect or to poor adherence.
Our finding that nearly all patients had rapid improvement of AD with the topical corticosteroid is not definitive proof but supports the notion that tachyphylaxis is largely mediated by poor adherence to treatment. Patients rapidly improved over the short study period. The short duration of treatment and multiple visits over the study period were designed to help ensure patient adherence. Rapid improvement in AD when topical corticosteroids are used should be expected, as AD patients have rapid improvement with application of topical corticosteroids in inpatient settings.11,12
Poor adherence to topical medication is common. In a Danish study, 99 of 322 patients (31%) did not redeem their AD prescriptions.13 In a single-center, 5-day, prospective study evaluating the use of fluocinonide cream 0.1% for treatment of children and adults with AD, the median percentage of prescribed doses taken was 40%, according to objective electronic monitors, even though patients reported 100% adherence in their medication diaries.Better adherence was seen on day 1 of treatment in which 66.6% (6/9) of patients adhered to their treatment strategy versus day 5 in which only 11.1% (1/9) of patients used their medication.1
Topical corticosteroids are safe and efficacious if used appropriately; however, patients commonly express fear and anxiety about using them. Topical corticosteroid phobia may stem from a misconception that these products carry the same adverse effects as their oral and systemic counterparts, which may be perpetuated by the media.1 Of 200 dermatology patients surveyed, 72.5% expressed concern about using topical corticosteroids on themselves or their children’s skin, and 24% of these patients stated they were noncompliant with their medication because of these worries. Almost 50% of patients requested prescriptions for corticosteroid-sparing medications such as tacrolimus.1 Patient education is important to help ensure treatment adherence. Other factors that can affect treatment adherence include forgetfulness; the chronic nature of AD; the need for ongoing application of topical treatments; prohibitive costs of some topical agents; and complexities in coordinating school, work, and family plans with the treatment regimen.2
We attempted to ensure good treatment adherence in our study by calling the patients in the intervention group twice daily. The mean improvement in EASI, TLSS, and IGA scores was higher in the intervention group versus the control group, which suggests that patient reminders have at least some benefit. Because AD treatment resistance appears more closely tied to nonadherence rather than loss of medication efficacy, it seems prudent to focus on interventions that would improve treatment adherence; however, such interventions generally are not well tested. Recommended interventions have included educating patients about the side effects of topical corticosteroids, avoiding use of medical jargon, and taking patient vehicle preference into account when prescribing treatments.8 Patients should be scheduled for a return visit within 1 to 2 weeks, as early return visits can augment treatment adherence.14 At the return visit, there can be a more detailed discussion of long-term management and side effects.8
Limitations of our study included a small sample size and brief treatment duration. Even though the patients had previously reported treatment failure with topical corticosteroids, all demonstrated improvement in only 1 week with a potent topical corticosteroid. The treatment resistance that initially was reported likely was due to poor adherence, but it is possible for AD patients to be resistant to treatment with topical corticosteroids due to allergic contact dermatitis. Patients could theoretically be allergic to components of the vehicle used in topical corticosteroids, which could aggravate their dermatitis; however, this effect seems unlikely in our patient population, as all the patients in our study showed improvement following treatment. Another study limitation was that adherence was not measured. The frequent follow-up visits were designed to encourage treatment adherence, but adherence was not specifically assessed. Although patients were encouraged to only use the desoximetasone spray during the study, it is not known whether patients used other products.
Conclusion
Some AD patients exhibit apparent decreased efficacy of topical corticosteroids over time, but this tachyphylaxis phenomenon is more likely due to poor treatment adherence than to loss of corticosteroid responsiveness. In our study, AD patients who reported treatment failure with topical corticosteroids improved rapidly with topical corticosteroids under conditions designed to promote good adherence to treatment. The majority of patients improved in all 4 parameters used for evaluating disease severity, with 100% of patients reporting improvement in pruritus. Intervention to improve treatment adherence may lead to better health outcomes. When AD appears resistant to topical corticosteroids, addressing adherence issues may be critical.
- Patel NU, D’Ambra V, Feldman SR. Increasing adherence with topical agents for atopic dermatitis. Am J Clin Dermatol. 2017;18:323-332.
- Mooney E, Rademaker M, Dailey R, et al. Adverse effects of topical corticosteroids in paediatric eczema: Australasian consensus statement. Australas J Dermatol. 2015;56:241-251.
- Taheri A, Cantrell J, Feldman SR. Tachyphylaxis to topical glucocorticoids; what is the evidence? Dermatol Online J. 2013;19:18954.
- Miller JJ, Roling D, Margolis D, et al. Failure to demonstrate therapeutic tachyphylaxis to topically applied steroids in patients with psoriasis. J Am Acad Dermatol. 1999;41:546-549.
- Smith SD, Harris V, Lee A, et al. General practitioners knowledge about use of topical corticosteroids in paediatric atopic dermatitis in Australia. Aust Fam Physician. 2017;46:335-340.
- Sathishkumar D, Moss C. Topical therapy in atopic dermatitis in children. Indian J Dermatol. 2016;61:656-661.
- Reitamo S, Remitz A. Topical agents for atopic dermatitis. In: Bieber T, ed. Advances in the Management of Atopic Dermatitis. London, United Kingdom: Future Medicine Ltd; 2013:62-72.
- Krejci-Manwaring J, Tusa MG, Carroll C, et al. Stealth monitoring of adherence to topical medication: adherence is very poor in children with atopic dermatitis. J Am Acad Dermatol. 2007;56:211-216.
- Fukaya M. Cortisol homeostasis in the epidermis is influenced by topical corticosteroids in patients with atopic dermatitis. Indian J Dermatol. 2017;62:440.
- Mehta AB, Nadkarni NJ, Patil SP, et al. Topical corticosteroids in dermatology. Indian J Dermatol Venereol Leprol. 2016;82:371-378.
- van der Schaft J, Keijzer WW, Sanders KJ, et al. Is there an additional value of inpatient treatment for patients with atopic dermatitis? Acta Derm Venereol. 2016;96:797-801.
- Dabade TS, Davis DM, Wetter DA, et al. Wet dressing therapy in conjunction with topical corticosteroids is effective for rapid control of severe pediatric atopic dermatitis: experience with 218 patients over 30 years at Mayo Clinic. J Am Acad Dermatol. 2011;67:100-106.
- Storm A, Andersen SE, Benfeldt E, et al. One in 3 prescriptions are never redeemed: primary nonadherence in an outpatient clinic. J Am Acad Dermatol. 2008;59:27-33.
- Sagransky MJ, Yentzer BA, Williams LL, et al. A randomized controlled pilot study of the effects of an extra office visit on adherence and outcomes in atopic dermatitis. Arch Dermatol. 2010;146:1428-1430.
Atopic dermatitis (AD) is most often treated with mid-potency topical corticosteroids.1,2 Although this option is effective, not all patients respond to treatment, and those who do may lose efficacy over time, a phenomenon known as tachyphylaxis. The pathophysiology of tachyphylaxis to topical corticosteroids has been ascribed to loss of corticosteroid receptor function,3 but the evidence is weak.3,4 Patients with severe treatment-resistant AD improve when treated with mid-potency topical steroids in an inpatient setting; therefore, treatment resistance to topical corticosteroids may be largely due to poor adherence.5
Patients with treatment-resistant AD generally improve when treated with topical corticosteroids under conditions designed to promote treatment adherence, but this improvement often is reported for study groups, not individual patients. Focusing on group data may not give a clear picture of what is happening at the individual level. In this study, we evaluated changes at an individual level to determine how frequently AD patients who were previously treated with topical corticosteroids unsuccessfully would respond to desoximetasone spray 0.25% under conditions designed to promote good adherence over a 7-day period.
Methods
This open-label, randomized, single-center clinical study included 12 patients with AD who were previously unsuccessfully treated with topical corticosteroids in the Department of Dermatology at Wake Forest Baptist Medical Center (Winston-Salem, North Carolina)(Table 1). The study was approved by the local institutional review board.

Inclusion criteria included men and women 18 years or older at baseline who had AD that was considered amenable to therapy with topical corticosteroids by the clinician and were able to comply with the study protocol (Figure). Written informed consent also was obtained from each patient. Women who were pregnant, breastfeeding, or unwilling to practice birth control during participation in the study were excluded. Other exclusion criteria included presence of a condition that in the opinion of the investigator would compromise the safety of the patient or quality of data as well as patients with no access to a telephone throughout the day. Patients diagnosed with conditions affecting adherence to treatment (eg, dementia, Alzheimer disease), those with a history of allergy or sensitivity to corticosteroids, and those with a history of drug hypersensitivity were excluded from the study.

All 12 patients were treated with desoximetasone spray 0.25% for 7 days. Patients were instructed not to use other AD medications during the study period. At baseline, patients were randomized to receive either twice-daily telephone calls to discuss treatment adherence (intervention group) or no telephone calls (control) during the study period. Patients in both the intervention and control groups returned for evaluation on days 3 and 7. During these visits, disease severity was evaluated using the pruritus visual analog scale, Eczema Area and Severity Index (EASI), total lesion severity scale (TLSS), and investigator global assessment (IGA). Descriptive statistics were used to report the outcomes for each patient.
Results
Twelve AD patients who were previously unsuccessfully treated with topical corticosteroids were recruited for the study. Six patients were randomized to the intervention group and 6 were randomized to the control group. Fifty percent of patients were black, 50% were women, and the average age was 50.4 years. All 12 patients completed the study.
At the end of the study, most patients showed improvement in all evaluation parameters (eFigure). All 12 patients showed improvement in pruritus visual analog scores; 83.3% (10/12) showed improved EASI scores, 75.0% (9/12) showed improved TLSS scores, and 58.3% (7/12) showed improved IGA scores (Tables 2–5). Patients who received telephone calls in the intervention group showed greater improvement compared to those in the control group, except for pruritus; the mean reduction in pruritus was 76.9% in the intervention group versus 87.0% in the control group. The mean improvement in EASI score was 46.9% in the intervention group versus 21.1% in the control group. The mean improvement in TLSS score was 38.3% in the intervention group versus 9.7% in the control group. The mean improvement in IGA score was 45.8% in the intervention group versus 4.2% in the control group. Only one patient in the control group (patient 8) showed lower EASI, TLSS, and IGA scores at baseline.





Comment
Although topical corticosteroids are the mainstay for treatment of AD, many patients report treatment resistance after a period of a few doses or longer.6-9 There is strong evidence demonstrating rapid corticosteroid receptor downregulation in tissues after corticosteroid therapy, which is the accepted mechanism for tachyphylaxis, but the timing of this effect does not match up with clinical experiences. The physiologic significance of corticosteroid agonist-induced receptor downregulation is unknown and may not have any considerable effect on corticosteroid efficacy.3 A systematic review by Taheri et al3 on the development of resistance to topical corticosteroids proposed 2 theories for the underlying pathogenesis of tachyphylaxis: (1) long-term patient nonadherence, and (2) the initial maximal response during the first few weeks of therapy eventually plateaus. Because corticosteroids may plateau after a certain number of doses, natural disease flare-ups during this period may give the wrong impression of tachyphylaxis.10 The treatment “resistance” reported by the patients in our study may have been due to this plateau effect or to poor adherence.
Our finding that nearly all patients had rapid improvement of AD with the topical corticosteroid is not definitive proof but supports the notion that tachyphylaxis is largely mediated by poor adherence to treatment. Patients rapidly improved over the short study period. The short duration of treatment and multiple visits over the study period were designed to help ensure patient adherence. Rapid improvement in AD when topical corticosteroids are used should be expected, as AD patients have rapid improvement with application of topical corticosteroids in inpatient settings.11,12
Poor adherence to topical medication is common. In a Danish study, 99 of 322 patients (31%) did not redeem their AD prescriptions.13 In a single-center, 5-day, prospective study evaluating the use of fluocinonide cream 0.1% for treatment of children and adults with AD, the median percentage of prescribed doses taken was 40%, according to objective electronic monitors, even though patients reported 100% adherence in their medication diaries.Better adherence was seen on day 1 of treatment in which 66.6% (6/9) of patients adhered to their treatment strategy versus day 5 in which only 11.1% (1/9) of patients used their medication.1
Topical corticosteroids are safe and efficacious if used appropriately; however, patients commonly express fear and anxiety about using them. Topical corticosteroid phobia may stem from a misconception that these products carry the same adverse effects as their oral and systemic counterparts, which may be perpetuated by the media.1 Of 200 dermatology patients surveyed, 72.5% expressed concern about using topical corticosteroids on themselves or their children’s skin, and 24% of these patients stated they were noncompliant with their medication because of these worries. Almost 50% of patients requested prescriptions for corticosteroid-sparing medications such as tacrolimus.1 Patient education is important to help ensure treatment adherence. Other factors that can affect treatment adherence include forgetfulness; the chronic nature of AD; the need for ongoing application of topical treatments; prohibitive costs of some topical agents; and complexities in coordinating school, work, and family plans with the treatment regimen.2
We attempted to ensure good treatment adherence in our study by calling the patients in the intervention group twice daily. The mean improvement in EASI, TLSS, and IGA scores was higher in the intervention group versus the control group, which suggests that patient reminders have at least some benefit. Because AD treatment resistance appears more closely tied to nonadherence rather than loss of medication efficacy, it seems prudent to focus on interventions that would improve treatment adherence; however, such interventions generally are not well tested. Recommended interventions have included educating patients about the side effects of topical corticosteroids, avoiding use of medical jargon, and taking patient vehicle preference into account when prescribing treatments.8 Patients should be scheduled for a return visit within 1 to 2 weeks, as early return visits can augment treatment adherence.14 At the return visit, there can be a more detailed discussion of long-term management and side effects.8
Limitations of our study included a small sample size and brief treatment duration. Even though the patients had previously reported treatment failure with topical corticosteroids, all demonstrated improvement in only 1 week with a potent topical corticosteroid. The treatment resistance that initially was reported likely was due to poor adherence, but it is possible for AD patients to be resistant to treatment with topical corticosteroids due to allergic contact dermatitis. Patients could theoretically be allergic to components of the vehicle used in topical corticosteroids, which could aggravate their dermatitis; however, this effect seems unlikely in our patient population, as all the patients in our study showed improvement following treatment. Another study limitation was that adherence was not measured. The frequent follow-up visits were designed to encourage treatment adherence, but adherence was not specifically assessed. Although patients were encouraged to only use the desoximetasone spray during the study, it is not known whether patients used other products.
Conclusion
Some AD patients exhibit apparent decreased efficacy of topical corticosteroids over time, but this tachyphylaxis phenomenon is more likely due to poor treatment adherence than to loss of corticosteroid responsiveness. In our study, AD patients who reported treatment failure with topical corticosteroids improved rapidly with topical corticosteroids under conditions designed to promote good adherence to treatment. The majority of patients improved in all 4 parameters used for evaluating disease severity, with 100% of patients reporting improvement in pruritus. Intervention to improve treatment adherence may lead to better health outcomes. When AD appears resistant to topical corticosteroids, addressing adherence issues may be critical.
Atopic dermatitis (AD) is most often treated with mid-potency topical corticosteroids.1,2 Although this option is effective, not all patients respond to treatment, and those who do may lose efficacy over time, a phenomenon known as tachyphylaxis. The pathophysiology of tachyphylaxis to topical corticosteroids has been ascribed to loss of corticosteroid receptor function,3 but the evidence is weak.3,4 Patients with severe treatment-resistant AD improve when treated with mid-potency topical steroids in an inpatient setting; therefore, treatment resistance to topical corticosteroids may be largely due to poor adherence.5
Patients with treatment-resistant AD generally improve when treated with topical corticosteroids under conditions designed to promote treatment adherence, but this improvement often is reported for study groups, not individual patients. Focusing on group data may not give a clear picture of what is happening at the individual level. In this study, we evaluated changes at an individual level to determine how frequently AD patients who were previously treated with topical corticosteroids unsuccessfully would respond to desoximetasone spray 0.25% under conditions designed to promote good adherence over a 7-day period.
Methods
This open-label, randomized, single-center clinical study included 12 patients with AD who were previously unsuccessfully treated with topical corticosteroids in the Department of Dermatology at Wake Forest Baptist Medical Center (Winston-Salem, North Carolina)(Table 1). The study was approved by the local institutional review board.

Inclusion criteria included men and women 18 years or older at baseline who had AD that was considered amenable to therapy with topical corticosteroids by the clinician and were able to comply with the study protocol (Figure). Written informed consent also was obtained from each patient. Women who were pregnant, breastfeeding, or unwilling to practice birth control during participation in the study were excluded. Other exclusion criteria included presence of a condition that in the opinion of the investigator would compromise the safety of the patient or quality of data as well as patients with no access to a telephone throughout the day. Patients diagnosed with conditions affecting adherence to treatment (eg, dementia, Alzheimer disease), those with a history of allergy or sensitivity to corticosteroids, and those with a history of drug hypersensitivity were excluded from the study.

All 12 patients were treated with desoximetasone spray 0.25% for 7 days. Patients were instructed not to use other AD medications during the study period. At baseline, patients were randomized to receive either twice-daily telephone calls to discuss treatment adherence (intervention group) or no telephone calls (control) during the study period. Patients in both the intervention and control groups returned for evaluation on days 3 and 7. During these visits, disease severity was evaluated using the pruritus visual analog scale, Eczema Area and Severity Index (EASI), total lesion severity scale (TLSS), and investigator global assessment (IGA). Descriptive statistics were used to report the outcomes for each patient.
Results
Twelve AD patients who were previously unsuccessfully treated with topical corticosteroids were recruited for the study. Six patients were randomized to the intervention group and 6 were randomized to the control group. Fifty percent of patients were black, 50% were women, and the average age was 50.4 years. All 12 patients completed the study.
At the end of the study, most patients showed improvement in all evaluation parameters (eFigure). All 12 patients showed improvement in pruritus visual analog scores; 83.3% (10/12) showed improved EASI scores, 75.0% (9/12) showed improved TLSS scores, and 58.3% (7/12) showed improved IGA scores (Tables 2–5). Patients who received telephone calls in the intervention group showed greater improvement compared to those in the control group, except for pruritus; the mean reduction in pruritus was 76.9% in the intervention group versus 87.0% in the control group. The mean improvement in EASI score was 46.9% in the intervention group versus 21.1% in the control group. The mean improvement in TLSS score was 38.3% in the intervention group versus 9.7% in the control group. The mean improvement in IGA score was 45.8% in the intervention group versus 4.2% in the control group. Only one patient in the control group (patient 8) showed lower EASI, TLSS, and IGA scores at baseline.





Comment
Although topical corticosteroids are the mainstay for treatment of AD, many patients report treatment resistance after a period of a few doses or longer.6-9 There is strong evidence demonstrating rapid corticosteroid receptor downregulation in tissues after corticosteroid therapy, which is the accepted mechanism for tachyphylaxis, but the timing of this effect does not match up with clinical experiences. The physiologic significance of corticosteroid agonist-induced receptor downregulation is unknown and may not have any considerable effect on corticosteroid efficacy.3 A systematic review by Taheri et al3 on the development of resistance to topical corticosteroids proposed 2 theories for the underlying pathogenesis of tachyphylaxis: (1) long-term patient nonadherence, and (2) the initial maximal response during the first few weeks of therapy eventually plateaus. Because corticosteroids may plateau after a certain number of doses, natural disease flare-ups during this period may give the wrong impression of tachyphylaxis.10 The treatment “resistance” reported by the patients in our study may have been due to this plateau effect or to poor adherence.
Our finding that nearly all patients had rapid improvement of AD with the topical corticosteroid is not definitive proof but supports the notion that tachyphylaxis is largely mediated by poor adherence to treatment. Patients rapidly improved over the short study period. The short duration of treatment and multiple visits over the study period were designed to help ensure patient adherence. Rapid improvement in AD when topical corticosteroids are used should be expected, as AD patients have rapid improvement with application of topical corticosteroids in inpatient settings.11,12
Poor adherence to topical medication is common. In a Danish study, 99 of 322 patients (31%) did not redeem their AD prescriptions.13 In a single-center, 5-day, prospective study evaluating the use of fluocinonide cream 0.1% for treatment of children and adults with AD, the median percentage of prescribed doses taken was 40%, according to objective electronic monitors, even though patients reported 100% adherence in their medication diaries.Better adherence was seen on day 1 of treatment in which 66.6% (6/9) of patients adhered to their treatment strategy versus day 5 in which only 11.1% (1/9) of patients used their medication.1
Topical corticosteroids are safe and efficacious if used appropriately; however, patients commonly express fear and anxiety about using them. Topical corticosteroid phobia may stem from a misconception that these products carry the same adverse effects as their oral and systemic counterparts, which may be perpetuated by the media.1 Of 200 dermatology patients surveyed, 72.5% expressed concern about using topical corticosteroids on themselves or their children’s skin, and 24% of these patients stated they were noncompliant with their medication because of these worries. Almost 50% of patients requested prescriptions for corticosteroid-sparing medications such as tacrolimus.1 Patient education is important to help ensure treatment adherence. Other factors that can affect treatment adherence include forgetfulness; the chronic nature of AD; the need for ongoing application of topical treatments; prohibitive costs of some topical agents; and complexities in coordinating school, work, and family plans with the treatment regimen.2
We attempted to ensure good treatment adherence in our study by calling the patients in the intervention group twice daily. The mean improvement in EASI, TLSS, and IGA scores was higher in the intervention group versus the control group, which suggests that patient reminders have at least some benefit. Because AD treatment resistance appears more closely tied to nonadherence rather than loss of medication efficacy, it seems prudent to focus on interventions that would improve treatment adherence; however, such interventions generally are not well tested. Recommended interventions have included educating patients about the side effects of topical corticosteroids, avoiding use of medical jargon, and taking patient vehicle preference into account when prescribing treatments.8 Patients should be scheduled for a return visit within 1 to 2 weeks, as early return visits can augment treatment adherence.14 At the return visit, there can be a more detailed discussion of long-term management and side effects.8
Limitations of our study included a small sample size and brief treatment duration. Even though the patients had previously reported treatment failure with topical corticosteroids, all demonstrated improvement in only 1 week with a potent topical corticosteroid. The treatment resistance that initially was reported likely was due to poor adherence, but it is possible for AD patients to be resistant to treatment with topical corticosteroids due to allergic contact dermatitis. Patients could theoretically be allergic to components of the vehicle used in topical corticosteroids, which could aggravate their dermatitis; however, this effect seems unlikely in our patient population, as all the patients in our study showed improvement following treatment. Another study limitation was that adherence was not measured. The frequent follow-up visits were designed to encourage treatment adherence, but adherence was not specifically assessed. Although patients were encouraged to only use the desoximetasone spray during the study, it is not known whether patients used other products.
Conclusion
Some AD patients exhibit apparent decreased efficacy of topical corticosteroids over time, but this tachyphylaxis phenomenon is more likely due to poor treatment adherence than to loss of corticosteroid responsiveness. In our study, AD patients who reported treatment failure with topical corticosteroids improved rapidly with topical corticosteroids under conditions designed to promote good adherence to treatment. The majority of patients improved in all 4 parameters used for evaluating disease severity, with 100% of patients reporting improvement in pruritus. Intervention to improve treatment adherence may lead to better health outcomes. When AD appears resistant to topical corticosteroids, addressing adherence issues may be critical.
- Patel NU, D’Ambra V, Feldman SR. Increasing adherence with topical agents for atopic dermatitis. Am J Clin Dermatol. 2017;18:323-332.
- Mooney E, Rademaker M, Dailey R, et al. Adverse effects of topical corticosteroids in paediatric eczema: Australasian consensus statement. Australas J Dermatol. 2015;56:241-251.
- Taheri A, Cantrell J, Feldman SR. Tachyphylaxis to topical glucocorticoids; what is the evidence? Dermatol Online J. 2013;19:18954.
- Miller JJ, Roling D, Margolis D, et al. Failure to demonstrate therapeutic tachyphylaxis to topically applied steroids in patients with psoriasis. J Am Acad Dermatol. 1999;41:546-549.
- Smith SD, Harris V, Lee A, et al. General practitioners knowledge about use of topical corticosteroids in paediatric atopic dermatitis in Australia. Aust Fam Physician. 2017;46:335-340.
- Sathishkumar D, Moss C. Topical therapy in atopic dermatitis in children. Indian J Dermatol. 2016;61:656-661.
- Reitamo S, Remitz A. Topical agents for atopic dermatitis. In: Bieber T, ed. Advances in the Management of Atopic Dermatitis. London, United Kingdom: Future Medicine Ltd; 2013:62-72.
- Krejci-Manwaring J, Tusa MG, Carroll C, et al. Stealth monitoring of adherence to topical medication: adherence is very poor in children with atopic dermatitis. J Am Acad Dermatol. 2007;56:211-216.
- Fukaya M. Cortisol homeostasis in the epidermis is influenced by topical corticosteroids in patients with atopic dermatitis. Indian J Dermatol. 2017;62:440.
- Mehta AB, Nadkarni NJ, Patil SP, et al. Topical corticosteroids in dermatology. Indian J Dermatol Venereol Leprol. 2016;82:371-378.
- van der Schaft J, Keijzer WW, Sanders KJ, et al. Is there an additional value of inpatient treatment for patients with atopic dermatitis? Acta Derm Venereol. 2016;96:797-801.
- Dabade TS, Davis DM, Wetter DA, et al. Wet dressing therapy in conjunction with topical corticosteroids is effective for rapid control of severe pediatric atopic dermatitis: experience with 218 patients over 30 years at Mayo Clinic. J Am Acad Dermatol. 2011;67:100-106.
- Storm A, Andersen SE, Benfeldt E, et al. One in 3 prescriptions are never redeemed: primary nonadherence in an outpatient clinic. J Am Acad Dermatol. 2008;59:27-33.
- Sagransky MJ, Yentzer BA, Williams LL, et al. A randomized controlled pilot study of the effects of an extra office visit on adherence and outcomes in atopic dermatitis. Arch Dermatol. 2010;146:1428-1430.
- Patel NU, D’Ambra V, Feldman SR. Increasing adherence with topical agents for atopic dermatitis. Am J Clin Dermatol. 2017;18:323-332.
- Mooney E, Rademaker M, Dailey R, et al. Adverse effects of topical corticosteroids in paediatric eczema: Australasian consensus statement. Australas J Dermatol. 2015;56:241-251.
- Taheri A, Cantrell J, Feldman SR. Tachyphylaxis to topical glucocorticoids; what is the evidence? Dermatol Online J. 2013;19:18954.
- Miller JJ, Roling D, Margolis D, et al. Failure to demonstrate therapeutic tachyphylaxis to topically applied steroids in patients with psoriasis. J Am Acad Dermatol. 1999;41:546-549.
- Smith SD, Harris V, Lee A, et al. General practitioners knowledge about use of topical corticosteroids in paediatric atopic dermatitis in Australia. Aust Fam Physician. 2017;46:335-340.
- Sathishkumar D, Moss C. Topical therapy in atopic dermatitis in children. Indian J Dermatol. 2016;61:656-661.
- Reitamo S, Remitz A. Topical agents for atopic dermatitis. In: Bieber T, ed. Advances in the Management of Atopic Dermatitis. London, United Kingdom: Future Medicine Ltd; 2013:62-72.
- Krejci-Manwaring J, Tusa MG, Carroll C, et al. Stealth monitoring of adherence to topical medication: adherence is very poor in children with atopic dermatitis. J Am Acad Dermatol. 2007;56:211-216.
- Fukaya M. Cortisol homeostasis in the epidermis is influenced by topical corticosteroids in patients with atopic dermatitis. Indian J Dermatol. 2017;62:440.
- Mehta AB, Nadkarni NJ, Patil SP, et al. Topical corticosteroids in dermatology. Indian J Dermatol Venereol Leprol. 2016;82:371-378.
- van der Schaft J, Keijzer WW, Sanders KJ, et al. Is there an additional value of inpatient treatment for patients with atopic dermatitis? Acta Derm Venereol. 2016;96:797-801.
- Dabade TS, Davis DM, Wetter DA, et al. Wet dressing therapy in conjunction with topical corticosteroids is effective for rapid control of severe pediatric atopic dermatitis: experience with 218 patients over 30 years at Mayo Clinic. J Am Acad Dermatol. 2011;67:100-106.
- Storm A, Andersen SE, Benfeldt E, et al. One in 3 prescriptions are never redeemed: primary nonadherence in an outpatient clinic. J Am Acad Dermatol. 2008;59:27-33.
- Sagransky MJ, Yentzer BA, Williams LL, et al. A randomized controlled pilot study of the effects of an extra office visit on adherence and outcomes in atopic dermatitis. Arch Dermatol. 2010;146:1428-1430.
Practice Points
- Mid-potency corticosteroids are the first-line treatment of atopic dermatitis (AD).
- Atopic dermatitis may fail to respond to topical corticosteroids initially or lose response over time, a phenomenon known as tachyphylaxis.
- Nonadherence to medication is the most likely cause of treatment resistance in patients with AD.
Dupilumab for Off-Label Treatment of Moderate to Severe Childhood Atopic Dermatitis
Case Report
A 7-year-old boy with a history of shellfish anaphylaxis, pollen allergy, asthma, rhinoconjunctivitis, frequent headaches and ear infections, sinusitis, bronchitis, vitiligo, warts, and cold sores presented to our dermatology clinic for evaluation of a widespread crusting, cracking, red rash that had been present since 6 months of age. The patient’s mother reported that he had many sleepless nights from uncontrolled itching. His medications included albuterol solution for nebulization, loratadine, and montelukast. Prior to the current presentation he had been treated with triamcinolone and betamethasone creams by the pediatrician. Despite compliance with topical therapy, his mother stated the itching persisted and lesions lingered with minimal improvement. He also was treated with oral corticosteroids for episodic sinusitis and bronchitis, which was beneficial to the skin lesions for only a short duration. The patient was adopted and therefore his family history was unavailable.
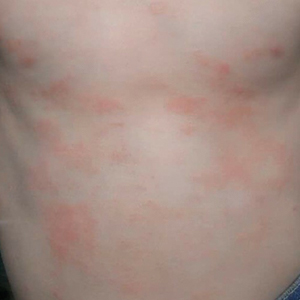
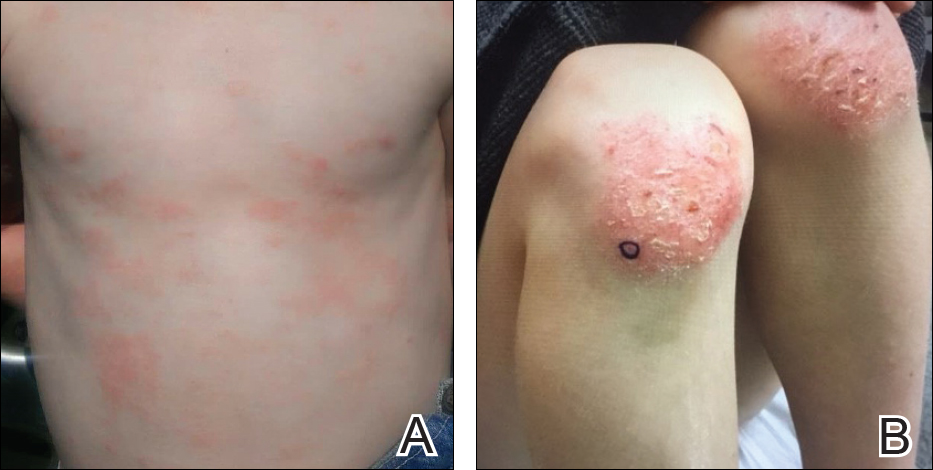
During physical examination, the patient was in the fetal position on the examination table and appeared uncomfortable, scratching himself. The patient admitted to severe widespread itching and burning. On skin examination, multiple thick, lichenified, highly pruritic plaques coalesced on the knees, ankles, arms, and wrists, and very discreet scaly patches were present on the scalp. Annular patches covered 50% of the patient’s body, with highly inflamed lesions concentrated in skin folds (Figure 1), leading to diagnosis of atopic dermatitis (AD).
Over the course of several months, a number of topical therapies were prescribed. The calcineurin inhibitor pimecrolimus cream 1% proffered minimal relief, and the patient experienced burning with crisaborole despite attempts to combine it with emollients and topical corticosteroids. The patient and his mother favored intermittent use of topical corticosteroids alone; however, he experienced frequent disease flares. Stabilized hypochlorous acid spray and mupirocin 2% antibiotic ointment were included in the treatment regimen as adjunctive topical therapies. Additionally, the patient underwent bleach and vinegar bath therapy without success.
Although UVA and UVB phototherapy has shown to be safe and effective in children, our patient had limited treatment options due to insurance restrictions. The patient had been taking oral corticosteroids on and off for years prior to presentation to our dermatology clinic.
Our patient weighed approximately 40 lb and was prescribed methotrexate 5 mg once weekly for 2 weeks along with oral folic acid 1 mg once daily, except when taking the methotrexate. Laboratory workup was ordered at 2- and then 4-week intervals. After 2 weeks of treatment, methotrexate was increased to 10 mg once weekly. His asthma was carefully monitored by the allergist, and his mother was instructed to stop the medication if he had worsening shortness of breath or exacerbation of asthma symptoms. He tolerated methotrexate at 10 mg once weekly well without clinical side effects for 6 months. His mother observed less frequent ear and sinus infections during methotrexate therapy; however, he developed anemia over time and the methotrexate was discontinued. Understanding the nature of off-label use in administering dupilumab, the patient’s mother consented to a scheduled dosage of 300 mg subcutaneous (SQ) injection every month in the absence of a loading dose with the assumption of future modifications pending his response to therapy.
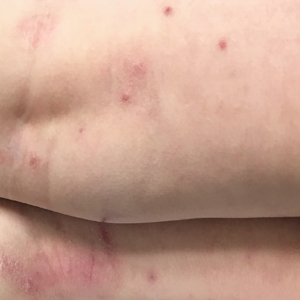
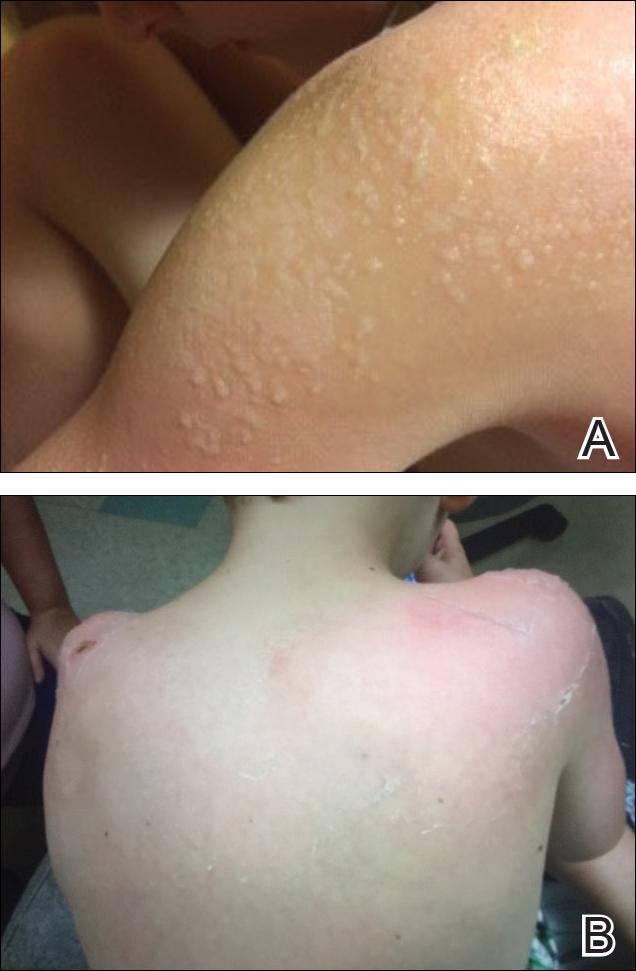
Five days after treatment with a 300-mg SQ dupilumab injection, the patient returned to clinic for evaluation of a vesicular rash with subsequent peeling confined to the shoulders (Figure 2). He and his mother denied any UV exposure, citing he had been completely out of the sun. He denied constitutional symptoms including fever, malaise, swelling, joint pain, headache, muscle pain, nausea, vomiting, diarrhea, enlarged lymph glands, difficulty urinating, breathing, or neurological disturbance. Upon physical examination, the rash was not considered to be a drug eruption. Had a mild drug reaction been suspected, a careful rechallenge, weighing the risks and benefits, would have been considered and was discussed with the mother and patient. New-onset or worsening eye symptoms should be reported; therefore, a referral to ophthalmology was prompted due to our patient’s history of rhinoconjunctivitis and persistent conjunctival injection observed early after initiating dupilumab therapy. Nothing remarkable was found.
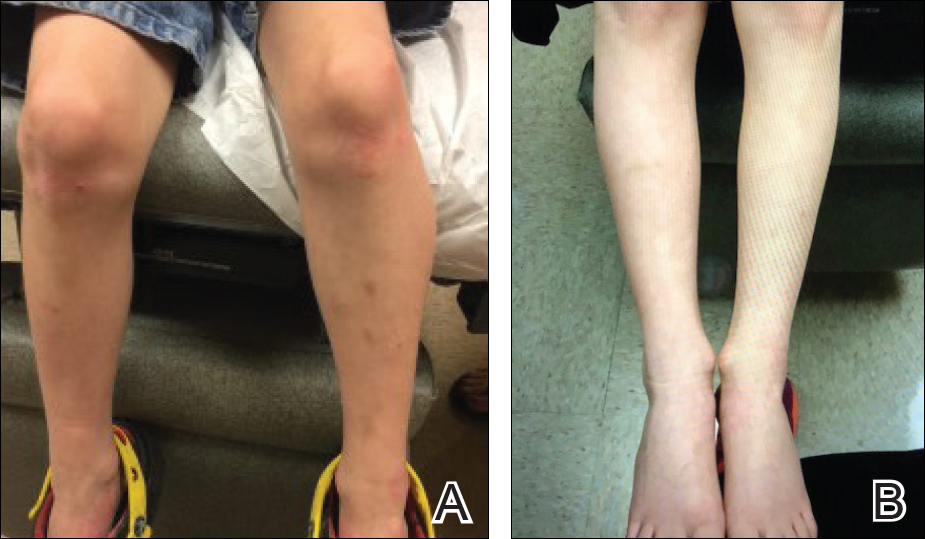
The patient was eager to continue dupilumab therapy due to considerable reduction of itching and elimination of lesions. His mother reported that the greatest benefit 1 month after starting dupilumab was almost no itching (Figure 3A). Additionally, he denied headache or nasopharyngitis at his 1-month office visit. After 2 months of dupilumab therapy, the patient reported persistent lesions on the feet and ankles despite concomitant treatment with topical corticosteroids. The decision to increase the dupilumab dose to 300-mg SQ injection once every 3 weeks for a total of 3 doses was made, which resulted in resolution of all lesions (Figure 3B).
Comment
Prevalence and Pathogenesis
Atopic dermatitis affects 31.6 million individuals in the United States, with 17.8 million experiencing moderate to severe lesions.1 The current prevalence of AD in the pediatric population ranges from 10% to 30% compared to 2% to 10% in adults. Fortunately, up to 70% of young children enter remission or improve by 12 years of age. Atopic diatheses may simultaneously occur, which includes asthma and rhinoconjunctivitis.2
Complications from AD include bacterial and viral infections and ocular disease. Furthermore, impaired growth in stature has been correlated with individuals who have extensive disease.2 Of interest, our 7-year-old patient gained 7 lb and grew almost 3 in within 6 months of being on immunosuppressant therapy. Children with AD have poorer sleep efficiency in contrast to children without AD.3 Eczema is associated with more frequent headaches in childhood, especially in those with sleep disturbances,4 as our patient had experienced prior to systemic therapy.
The pathogenesis of AD is complex, and one must take into consideration the multiple cellular activities including inflammatory mechanics in the absence of IgE-mediated sensitization, epidermal barrier changes, epicutaneous sensitization, dendritic cell roles, T-cell responses and cytokine orchestrations, actions of microbial colonization, and involvement of autoimmunity.5 Select patients with AD have IgE antibodies focused against self-proteins. Disease severity correlates with ubiquity of these antibodies. Moreover, certain autoallergens induce helper T cell (TH1) responses.5 Circulating TH2 cytokines and chemokines IL-4, IL4ra, and IL-13 also have been linked to AD pathogenesis. Additionally, nonlesional skin abnormalities have been observed.6 Most recently, researchers have identified a caspase recruitment domain family member 11 (CARD11) gene mutation possibly leading to AD.7 Clinically, our patient responded favorably to dupilumab, which inhibits TH2 cytokines IL-4 and IL13. He experienced a considerable decrease in itching and inflammation and reduced lesion count after 1 month of treatment with dupilumab. No skin lesions were identified on visual examination at week 17 and inevitably the patient discontinued messy topicals.
Treatment Options
Because AD is characterized by episodes of remission and relapse, management generally is comprised of trigger avoidance, including known allergens and irritants; a skin care regimen that promotes healthy epidermal barrier function; anti-inflammatory therapies to control both flares and subclinical inflammation; and adjunctive therapy for additional symptomatic control (eg, phototherapy, stabilized hypochlorous acid, topical antibiotic treatment) when needed. Avoidance of excessive washing or irritants, food provocation, and emotional stress, as well as toleration of body temperature fluctuations and humidity, is recommended to amend exacerbations.5
Current topical therapies include emollients; corticosteroids; calcineurin inhibitors; and crisaborole, a newer phosphodiesterase 4 inhibitor. There are a number of emollients and moisturizers available, and one over-the-counter preparation showed tolerability and improved skin hydration in AD patients and demonstrated less transepidermal water loss than the control group.8 Ointments such as petrolatum usually do not include ingredients such as preservatives, gelling agents, or humectants that can promote stinging or burning.9 Topical corticosteroids, which ameliorate inflammation by subduing proinflammatory cytokine expression, have been the mainstay of treatment for more than 60 years; however, caution should be used due to the potential for side effects, mainly but not limited to systemic absorption in children, development of striae, and skin atrophy. Calcineurin inhibitors prohibit T-cell activity, modify mast cell response, and decrease dendritic cells in the epidermis. Since 2000, calcineurin inhibitors have been utilized as steroid-sparing agents10; however, prior authorization is still necessary with some insurance providers. Crisaborole ointment 2%, the newest topical agent for AD treatment in the market, has shown improvement of erythema, exudation, and pruritus. Approved for patients aged 2 years and older, twice-daily application of topical crisaborole as a steroid-sparing agent has rendered AD symptom relief.11 It has been reported that 4% of patients encounter stinging or burning with topical crisaborole application, whereas up to 50% of calcineurin inhibitors induce these adverse effects.12 Stabilized hypochlorous acid spray or gel acts as an antipruritic and antimicrobial agent, relieving pain associated with skin irritations. Topical antimicrobial preparations such as mupirocin 2% antibiotic ointment can reduce Staphylococcus colonization when applied in the nasal passage as well as to affected skin lesions.2
In children, UVA and UVB phototherapy has proven safe and effective and can be utilized in AD when suitable.13 When patients inadequately respond to topical therapies and phototherapy, systemic immunomodulatory agents have been recommended as treatment options.A child’s developing immune system indeed may be sensitive to systemic therapies as the innate immune system fully matures in adolescence and his/her adaptive immune system is undergoing vigorous definition.14 Systemic immunomodulatory agents such as cyclosporine, azathioprine, mycophenolate mofetil, and methotrexate have been used off label for years and pose certain challenges in being identified as durable therapies due to potential side effects. Cyclosporine is effective for the treatment of AD; however, long-term administration should be dosed up to a 12-month period and then stopped to decrease cumulative exposure to the drug. Therefore, further treatment options must be considered. For children, cyclosporine should be administered in a dose of 3 to 6 mg/kg daily. Fluctuations in blood pressure and renal function should be monitored. The recommended pediatric dose for azathioprine is 1 to 4 mg/kg daily with laboratory monitoring, particularly of liver enzymes and complete blood cell count. Obtaining the patient’s thiopurine methyltransferase level may aid in dosing. Gastrointestinal tract symptoms such as nausea, vomiting, and diarrhea are common. Phototherapy is not advised in conjunction with azathioprine due to an increased risk of photocarcinogenicity.13 The literature supporting mycophenolate mofetil in children with AD is limited.
Biologic therapies targeting IgE, B-lymphocyte antigen CD20, IL-5, thymic stromal lymphopoietin, TH17 cells, IL-12, IL-23, interferon gamma, IL-6 receptors, tumor necrosis factor, phosphodiesterase 4, Janus kinase, chymase, and nuclear receptors expressed on adipocytes and immune cells have undergone investigation for treatment of AD.17 Additionally, biologic agents targeting IL-31, IL-13, and IL-22 also have been evaluated.1 Currently, there are no US Food and Drug Administration–approved biologic agents for moderate to severe childhood AD.
Dupilumab, an IL-4Rα and IL-13Rα antagonist, recently has been approved for treatment of moderate to severe AD in adults but not yet for children. Potential side effects include nasopharyngitis, headache, hypersensitivity reactions, and ocular symptoms,11 namely keratitis and conjunctivitis.18 Less than 1% of patients experienced keratitis in clinical trials, while conjunctivitis was reported in 4% of patients taking dupilumab with topical corticosteroids at 52 weeks.18 However, possible ocular findings on slit-lamp examination in AD patients include atopic keratoconjunctivitis, blepharitis, palpebral conjunctival scarring, papillary conjunctival reaction, Horner-Trantas dots, keratoconus, and atopic cataracts. Spontaneous retinal detachment is seen more commonly in individuals with AD than in the general population.19
In clinical trials, hypersensitivity reactions included urticaria and serum sickness or serum sickness–like reactions in less than 1% of patients taking dupilumab.18
Conclusion
Childhood AD can be debilitating, and affected individuals often lead a poorer quality of life if left untreated. Embarrassment and isolation are commonly experienced. Increased responsibility and work in tending for a child with eczema may result in parental exhaustion.21 As with psoriasis, AD can impair activity and productivity.22 Currently, dupilumab has proven to positively impact health-related quality of life for adults.23 Pending the outcome of ongoing pediatric clinical trials, dupilumab may become a benchmark therapy for children younger than 18 years.
- Samalonis L. What’s new in eczema and atopic dermatitis research. The Dermatologist. November 19, 2015. http://www.the-dermatologist.com/content/whats-new-eczema-and-atopic-dermatitis-research. Accessed July 19, 2018.
- Habif T. Atopic dermatitis. In: Bonnet C, Pinczewski A, Cook L, eds. Clinical Dermatology. 5th ed. Edinburgh, Scotland: Mosby Elsevier; 2010:160-180.
- Fishbein AB, Mueller K, Kruse L, et al. Sleep disturbance in children with moderate/severe atopic dermatitis: a case control study [published online October 28, 2017]. J Am Acad Dermatol. 2018;78:336-341.
- Silverberg J. Association between childhood eczema and headaches: an analysis of 19 US population-based studies [published online August 29, 2015]. J Allergy Clin Immunol. 2016;137:492-499.e5.
- Bieber T, Bussmann C. Atopic dermatitis. In: Bolognia JL, Jorizzo JL, Schaffer JV, eds. Dermatology. 3rd ed. China: Saunders Elsevier; 2012:203-216.
- Suarez-Farinas M, Tintle S, Shemer A, et al. Non-lesional atopic dermatitis (AD) skin is characterized by broad terminal differentiation defects and variable immune abnormalities. J Allergy Clin Immunol. 2011;127:954-964.
- Hilton L. AD gene mutation identified: discovery may lead to new therapeutic option for patients. Dermatol Times. 2017;38:30.
- Zeichner JA, Dryer L. Effect of CeraVe Healing Ointment on skin hydration and barrier function on normal and barrier-impaired skin. Poster presented at: Orlando Dermatology Aesthetic & Clinical Conference; January 15-16, 2016; Orlando, FL.
- Garg T, Rath G, Goyal AK. Comprehensive review on additives of topical dosage forms for drug delivery. Drug Delivery. 2015;22:969-987.
- Eichenfield LF, Tom WL, Berger TG, et al. Guidelines of care for the management of atopic dermatitis section 2. management and treatment of atopic dermatitis with topical therapies. J Am Acad Dermatol. 2014;71:116-132.
- Koutnik-Fotopoulous E. Update on the latest eczema treatments. The Dermatologist. February 17, 2016. http://www.the-dermatologist.com/content/update-latest-eczema-treatments. Accessed August 16, 2018.
- Paller AS, Tom WL, Lebwohl MG, et al. Efficacy and safety of crisaborole ointment, a novel phosphodiesterase 4 inhibitor for the topical treatment of AD in children and adults [published online July 11, 2016]. J Am Acad Dermatol. 2016;75:494-503.
- Sidbury R, Davis D, Cohen D, et al. Guidelines of care for the management of atopic dermatitis: section 3. management and treatment with phototherapy and systemic agents . J Am Acad Dermatol. 2014;71:327-349.
- van der Merwe R, Gianella-Borradori A. Industry perspective on the clinical development of systemic products for the treatment of atopic dermatitis in pediatric patients with inadequate response to topical prescription therapy. Presented at: FDA Dermatologic and Ophthalmic Drugs Advisory Committee Meeting; March 9, 2015; Silver Spring, MD.
- Heller M, Shin HT, Orlow SJ, et al. Mycophenolate mofetil for severe childhood atopic dermatitis: experience in 14 patients. Br J Dermatol. 2007;157:127-132.
- Callen JP, Kulp-Shorten CL. Methotrexate. In: Wolverton SE, ed. Comprehensive Dermatologic Drug Therapy. 3rd ed. China: Saunders Elsevier; 2013:169-181.
- Guttman-Yassky E, Dhingra N, Leung DY. New era of biological therapeutics in atopic dermatitis [published online January 16, 2013]. Expert Opin Biol Ther. 2013;13:549-561.
- Dupixent [package insert]. Tarrytown, NY: Regeneron Pharmaceuticals, Inc; 2017.
- Lowery RS. Ophthalmologic manifestations of atopic dermatitis clinical presentation. Medscape website. emedicine.medscape.com/article/1197636-clinical#b4. Updated September 7, 2016. Accessed July 19, 2018.
- Lenz HJ. Management and preparedness for infusion and hypersensitivity reactions. Oncologist. 2007;12:601-609.
- Lewis-Jones S. Quality of life and childhood atopic dermatitis: the misery of living with childhood eczema. Int J Clin Pract. 2006;60:984-992.
- Eckert L, Gupta S, Amand C, et al. Impact of atopic dermatitis on health-related quality of life and productivity in adults in the Unites States: an analysis using the National Health and Wellness Survey. J Am Acad Dermatol, 2017;77:274-279.
- Tsianakas A, Luger TA, Radin A. Dupilumab treatment improves quality of life in adult patients with moderate-to-severe atopic dermatitis: results from a randomized, placebo-controlled clinical trial [published online January 11, 2018]. Br J Dermatol. 2018;178:406-414.
Case Report
A 7-year-old boy with a history of shellfish anaphylaxis, pollen allergy, asthma, rhinoconjunctivitis, frequent headaches and ear infections, sinusitis, bronchitis, vitiligo, warts, and cold sores presented to our dermatology clinic for evaluation of a widespread crusting, cracking, red rash that had been present since 6 months of age. The patient’s mother reported that he had many sleepless nights from uncontrolled itching. His medications included albuterol solution for nebulization, loratadine, and montelukast. Prior to the current presentation he had been treated with triamcinolone and betamethasone creams by the pediatrician. Despite compliance with topical therapy, his mother stated the itching persisted and lesions lingered with minimal improvement. He also was treated with oral corticosteroids for episodic sinusitis and bronchitis, which was beneficial to the skin lesions for only a short duration. The patient was adopted and therefore his family history was unavailable.
During physical examination, the patient was in the fetal position on the examination table and appeared uncomfortable, scratching himself. The patient admitted to severe widespread itching and burning. On skin examination, multiple thick, lichenified, highly pruritic plaques coalesced on the knees, ankles, arms, and wrists, and very discreet scaly patches were present on the scalp. Annular patches covered 50% of the patient’s body, with highly inflamed lesions concentrated in skin folds (Figure 1), leading to diagnosis of atopic dermatitis (AD).

Over the course of several months, a number of topical therapies were prescribed. The calcineurin inhibitor pimecrolimus cream 1% proffered minimal relief, and the patient experienced burning with crisaborole despite attempts to combine it with emollients and topical corticosteroids. The patient and his mother favored intermittent use of topical corticosteroids alone; however, he experienced frequent disease flares. Stabilized hypochlorous acid spray and mupirocin 2% antibiotic ointment were included in the treatment regimen as adjunctive topical therapies. Additionally, the patient underwent bleach and vinegar bath therapy without success.
Although UVA and UVB phototherapy has shown to be safe and effective in children, our patient had limited treatment options due to insurance restrictions. The patient had been taking oral corticosteroids on and off for years prior to presentation to our dermatology clinic.
Our patient weighed approximately 40 lb and was prescribed methotrexate 5 mg once weekly for 2 weeks along with oral folic acid 1 mg once daily, except when taking the methotrexate. Laboratory workup was ordered at 2- and then 4-week intervals. After 2 weeks of treatment, methotrexate was increased to 10 mg once weekly. His asthma was carefully monitored by the allergist, and his mother was instructed to stop the medication if he had worsening shortness of breath or exacerbation of asthma symptoms. He tolerated methotrexate at 10 mg once weekly well without clinical side effects for 6 months. His mother observed less frequent ear and sinus infections during methotrexate therapy; however, he developed anemia over time and the methotrexate was discontinued. Understanding the nature of off-label use in administering dupilumab, the patient’s mother consented to a scheduled dosage of 300 mg subcutaneous (SQ) injection every month in the absence of a loading dose with the assumption of future modifications pending his response to therapy.
Five days after treatment with a 300-mg SQ dupilumab injection, the patient returned to clinic for evaluation of a vesicular rash with subsequent peeling confined to the shoulders (Figure 2). He and his mother denied any UV exposure, citing he had been completely out of the sun. He denied constitutional symptoms including fever, malaise, swelling, joint pain, headache, muscle pain, nausea, vomiting, diarrhea, enlarged lymph glands, difficulty urinating, breathing, or neurological disturbance. Upon physical examination, the rash was not considered to be a drug eruption. Had a mild drug reaction been suspected, a careful rechallenge, weighing the risks and benefits, would have been considered and was discussed with the mother and patient. New-onset or worsening eye symptoms should be reported; therefore, a referral to ophthalmology was prompted due to our patient’s history of rhinoconjunctivitis and persistent conjunctival injection observed early after initiating dupilumab therapy. Nothing remarkable was found.

The patient was eager to continue dupilumab therapy due to considerable reduction of itching and elimination of lesions. His mother reported that the greatest benefit 1 month after starting dupilumab was almost no itching (Figure 3A). Additionally, he denied headache or nasopharyngitis at his 1-month office visit. After 2 months of dupilumab therapy, the patient reported persistent lesions on the feet and ankles despite concomitant treatment with topical corticosteroids. The decision to increase the dupilumab dose to 300-mg SQ injection once every 3 weeks for a total of 3 doses was made, which resulted in resolution of all lesions (Figure 3B).

Comment
Prevalence and Pathogenesis
Atopic dermatitis affects 31.6 million individuals in the United States, with 17.8 million experiencing moderate to severe lesions.1 The current prevalence of AD in the pediatric population ranges from 10% to 30% compared to 2% to 10% in adults. Fortunately, up to 70% of young children enter remission or improve by 12 years of age. Atopic diatheses may simultaneously occur, which includes asthma and rhinoconjunctivitis.2
Complications from AD include bacterial and viral infections and ocular disease. Furthermore, impaired growth in stature has been correlated with individuals who have extensive disease.2 Of interest, our 7-year-old patient gained 7 lb and grew almost 3 in within 6 months of being on immunosuppressant therapy. Children with AD have poorer sleep efficiency in contrast to children without AD.3 Eczema is associated with more frequent headaches in childhood, especially in those with sleep disturbances,4 as our patient had experienced prior to systemic therapy.
The pathogenesis of AD is complex, and one must take into consideration the multiple cellular activities including inflammatory mechanics in the absence of IgE-mediated sensitization, epidermal barrier changes, epicutaneous sensitization, dendritic cell roles, T-cell responses and cytokine orchestrations, actions of microbial colonization, and involvement of autoimmunity.5 Select patients with AD have IgE antibodies focused against self-proteins. Disease severity correlates with ubiquity of these antibodies. Moreover, certain autoallergens induce helper T cell (TH1) responses.5 Circulating TH2 cytokines and chemokines IL-4, IL4ra, and IL-13 also have been linked to AD pathogenesis. Additionally, nonlesional skin abnormalities have been observed.6 Most recently, researchers have identified a caspase recruitment domain family member 11 (CARD11) gene mutation possibly leading to AD.7 Clinically, our patient responded favorably to dupilumab, which inhibits TH2 cytokines IL-4 and IL13. He experienced a considerable decrease in itching and inflammation and reduced lesion count after 1 month of treatment with dupilumab. No skin lesions were identified on visual examination at week 17 and inevitably the patient discontinued messy topicals.
Treatment Options
Because AD is characterized by episodes of remission and relapse, management generally is comprised of trigger avoidance, including known allergens and irritants; a skin care regimen that promotes healthy epidermal barrier function; anti-inflammatory therapies to control both flares and subclinical inflammation; and adjunctive therapy for additional symptomatic control (eg, phototherapy, stabilized hypochlorous acid, topical antibiotic treatment) when needed. Avoidance of excessive washing or irritants, food provocation, and emotional stress, as well as toleration of body temperature fluctuations and humidity, is recommended to amend exacerbations.5
Current topical therapies include emollients; corticosteroids; calcineurin inhibitors; and crisaborole, a newer phosphodiesterase 4 inhibitor. There are a number of emollients and moisturizers available, and one over-the-counter preparation showed tolerability and improved skin hydration in AD patients and demonstrated less transepidermal water loss than the control group.8 Ointments such as petrolatum usually do not include ingredients such as preservatives, gelling agents, or humectants that can promote stinging or burning.9 Topical corticosteroids, which ameliorate inflammation by subduing proinflammatory cytokine expression, have been the mainstay of treatment for more than 60 years; however, caution should be used due to the potential for side effects, mainly but not limited to systemic absorption in children, development of striae, and skin atrophy. Calcineurin inhibitors prohibit T-cell activity, modify mast cell response, and decrease dendritic cells in the epidermis. Since 2000, calcineurin inhibitors have been utilized as steroid-sparing agents10; however, prior authorization is still necessary with some insurance providers. Crisaborole ointment 2%, the newest topical agent for AD treatment in the market, has shown improvement of erythema, exudation, and pruritus. Approved for patients aged 2 years and older, twice-daily application of topical crisaborole as a steroid-sparing agent has rendered AD symptom relief.11 It has been reported that 4% of patients encounter stinging or burning with topical crisaborole application, whereas up to 50% of calcineurin inhibitors induce these adverse effects.12 Stabilized hypochlorous acid spray or gel acts as an antipruritic and antimicrobial agent, relieving pain associated with skin irritations. Topical antimicrobial preparations such as mupirocin 2% antibiotic ointment can reduce Staphylococcus colonization when applied in the nasal passage as well as to affected skin lesions.2
In children, UVA and UVB phototherapy has proven safe and effective and can be utilized in AD when suitable.13 When patients inadequately respond to topical therapies and phototherapy, systemic immunomodulatory agents have been recommended as treatment options.A child’s developing immune system indeed may be sensitive to systemic therapies as the innate immune system fully matures in adolescence and his/her adaptive immune system is undergoing vigorous definition.14 Systemic immunomodulatory agents such as cyclosporine, azathioprine, mycophenolate mofetil, and methotrexate have been used off label for years and pose certain challenges in being identified as durable therapies due to potential side effects. Cyclosporine is effective for the treatment of AD; however, long-term administration should be dosed up to a 12-month period and then stopped to decrease cumulative exposure to the drug. Therefore, further treatment options must be considered. For children, cyclosporine should be administered in a dose of 3 to 6 mg/kg daily. Fluctuations in blood pressure and renal function should be monitored. The recommended pediatric dose for azathioprine is 1 to 4 mg/kg daily with laboratory monitoring, particularly of liver enzymes and complete blood cell count. Obtaining the patient’s thiopurine methyltransferase level may aid in dosing. Gastrointestinal tract symptoms such as nausea, vomiting, and diarrhea are common. Phototherapy is not advised in conjunction with azathioprine due to an increased risk of photocarcinogenicity.13 The literature supporting mycophenolate mofetil in children with AD is limited.
Biologic therapies targeting IgE, B-lymphocyte antigen CD20, IL-5, thymic stromal lymphopoietin, TH17 cells, IL-12, IL-23, interferon gamma, IL-6 receptors, tumor necrosis factor, phosphodiesterase 4, Janus kinase, chymase, and nuclear receptors expressed on adipocytes and immune cells have undergone investigation for treatment of AD.17 Additionally, biologic agents targeting IL-31, IL-13, and IL-22 also have been evaluated.1 Currently, there are no US Food and Drug Administration–approved biologic agents for moderate to severe childhood AD.
Dupilumab, an IL-4Rα and IL-13Rα antagonist, recently has been approved for treatment of moderate to severe AD in adults but not yet for children. Potential side effects include nasopharyngitis, headache, hypersensitivity reactions, and ocular symptoms,11 namely keratitis and conjunctivitis.18 Less than 1% of patients experienced keratitis in clinical trials, while conjunctivitis was reported in 4% of patients taking dupilumab with topical corticosteroids at 52 weeks.18 However, possible ocular findings on slit-lamp examination in AD patients include atopic keratoconjunctivitis, blepharitis, palpebral conjunctival scarring, papillary conjunctival reaction, Horner-Trantas dots, keratoconus, and atopic cataracts. Spontaneous retinal detachment is seen more commonly in individuals with AD than in the general population.19
In clinical trials, hypersensitivity reactions included urticaria and serum sickness or serum sickness–like reactions in less than 1% of patients taking dupilumab.18
Conclusion
Childhood AD can be debilitating, and affected individuals often lead a poorer quality of life if left untreated. Embarrassment and isolation are commonly experienced. Increased responsibility and work in tending for a child with eczema may result in parental exhaustion.21 As with psoriasis, AD can impair activity and productivity.22 Currently, dupilumab has proven to positively impact health-related quality of life for adults.23 Pending the outcome of ongoing pediatric clinical trials, dupilumab may become a benchmark therapy for children younger than 18 years.
Case Report
A 7-year-old boy with a history of shellfish anaphylaxis, pollen allergy, asthma, rhinoconjunctivitis, frequent headaches and ear infections, sinusitis, bronchitis, vitiligo, warts, and cold sores presented to our dermatology clinic for evaluation of a widespread crusting, cracking, red rash that had been present since 6 months of age. The patient’s mother reported that he had many sleepless nights from uncontrolled itching. His medications included albuterol solution for nebulization, loratadine, and montelukast. Prior to the current presentation he had been treated with triamcinolone and betamethasone creams by the pediatrician. Despite compliance with topical therapy, his mother stated the itching persisted and lesions lingered with minimal improvement. He also was treated with oral corticosteroids for episodic sinusitis and bronchitis, which was beneficial to the skin lesions for only a short duration. The patient was adopted and therefore his family history was unavailable.
During physical examination, the patient was in the fetal position on the examination table and appeared uncomfortable, scratching himself. The patient admitted to severe widespread itching and burning. On skin examination, multiple thick, lichenified, highly pruritic plaques coalesced on the knees, ankles, arms, and wrists, and very discreet scaly patches were present on the scalp. Annular patches covered 50% of the patient’s body, with highly inflamed lesions concentrated in skin folds (Figure 1), leading to diagnosis of atopic dermatitis (AD).

Over the course of several months, a number of topical therapies were prescribed. The calcineurin inhibitor pimecrolimus cream 1% proffered minimal relief, and the patient experienced burning with crisaborole despite attempts to combine it with emollients and topical corticosteroids. The patient and his mother favored intermittent use of topical corticosteroids alone; however, he experienced frequent disease flares. Stabilized hypochlorous acid spray and mupirocin 2% antibiotic ointment were included in the treatment regimen as adjunctive topical therapies. Additionally, the patient underwent bleach and vinegar bath therapy without success.
Although UVA and UVB phototherapy has shown to be safe and effective in children, our patient had limited treatment options due to insurance restrictions. The patient had been taking oral corticosteroids on and off for years prior to presentation to our dermatology clinic.
Our patient weighed approximately 40 lb and was prescribed methotrexate 5 mg once weekly for 2 weeks along with oral folic acid 1 mg once daily, except when taking the methotrexate. Laboratory workup was ordered at 2- and then 4-week intervals. After 2 weeks of treatment, methotrexate was increased to 10 mg once weekly. His asthma was carefully monitored by the allergist, and his mother was instructed to stop the medication if he had worsening shortness of breath or exacerbation of asthma symptoms. He tolerated methotrexate at 10 mg once weekly well without clinical side effects for 6 months. His mother observed less frequent ear and sinus infections during methotrexate therapy; however, he developed anemia over time and the methotrexate was discontinued. Understanding the nature of off-label use in administering dupilumab, the patient’s mother consented to a scheduled dosage of 300 mg subcutaneous (SQ) injection every month in the absence of a loading dose with the assumption of future modifications pending his response to therapy.
Five days after treatment with a 300-mg SQ dupilumab injection, the patient returned to clinic for evaluation of a vesicular rash with subsequent peeling confined to the shoulders (Figure 2). He and his mother denied any UV exposure, citing he had been completely out of the sun. He denied constitutional symptoms including fever, malaise, swelling, joint pain, headache, muscle pain, nausea, vomiting, diarrhea, enlarged lymph glands, difficulty urinating, breathing, or neurological disturbance. Upon physical examination, the rash was not considered to be a drug eruption. Had a mild drug reaction been suspected, a careful rechallenge, weighing the risks and benefits, would have been considered and was discussed with the mother and patient. New-onset or worsening eye symptoms should be reported; therefore, a referral to ophthalmology was prompted due to our patient’s history of rhinoconjunctivitis and persistent conjunctival injection observed early after initiating dupilumab therapy. Nothing remarkable was found.

The patient was eager to continue dupilumab therapy due to considerable reduction of itching and elimination of lesions. His mother reported that the greatest benefit 1 month after starting dupilumab was almost no itching (Figure 3A). Additionally, he denied headache or nasopharyngitis at his 1-month office visit. After 2 months of dupilumab therapy, the patient reported persistent lesions on the feet and ankles despite concomitant treatment with topical corticosteroids. The decision to increase the dupilumab dose to 300-mg SQ injection once every 3 weeks for a total of 3 doses was made, which resulted in resolution of all lesions (Figure 3B).

Comment
Prevalence and Pathogenesis
Atopic dermatitis affects 31.6 million individuals in the United States, with 17.8 million experiencing moderate to severe lesions.1 The current prevalence of AD in the pediatric population ranges from 10% to 30% compared to 2% to 10% in adults. Fortunately, up to 70% of young children enter remission or improve by 12 years of age. Atopic diatheses may simultaneously occur, which includes asthma and rhinoconjunctivitis.2
Complications from AD include bacterial and viral infections and ocular disease. Furthermore, impaired growth in stature has been correlated with individuals who have extensive disease.2 Of interest, our 7-year-old patient gained 7 lb and grew almost 3 in within 6 months of being on immunosuppressant therapy. Children with AD have poorer sleep efficiency in contrast to children without AD.3 Eczema is associated with more frequent headaches in childhood, especially in those with sleep disturbances,4 as our patient had experienced prior to systemic therapy.
The pathogenesis of AD is complex, and one must take into consideration the multiple cellular activities including inflammatory mechanics in the absence of IgE-mediated sensitization, epidermal barrier changes, epicutaneous sensitization, dendritic cell roles, T-cell responses and cytokine orchestrations, actions of microbial colonization, and involvement of autoimmunity.5 Select patients with AD have IgE antibodies focused against self-proteins. Disease severity correlates with ubiquity of these antibodies. Moreover, certain autoallergens induce helper T cell (TH1) responses.5 Circulating TH2 cytokines and chemokines IL-4, IL4ra, and IL-13 also have been linked to AD pathogenesis. Additionally, nonlesional skin abnormalities have been observed.6 Most recently, researchers have identified a caspase recruitment domain family member 11 (CARD11) gene mutation possibly leading to AD.7 Clinically, our patient responded favorably to dupilumab, which inhibits TH2 cytokines IL-4 and IL13. He experienced a considerable decrease in itching and inflammation and reduced lesion count after 1 month of treatment with dupilumab. No skin lesions were identified on visual examination at week 17 and inevitably the patient discontinued messy topicals.
Treatment Options
Because AD is characterized by episodes of remission and relapse, management generally is comprised of trigger avoidance, including known allergens and irritants; a skin care regimen that promotes healthy epidermal barrier function; anti-inflammatory therapies to control both flares and subclinical inflammation; and adjunctive therapy for additional symptomatic control (eg, phototherapy, stabilized hypochlorous acid, topical antibiotic treatment) when needed. Avoidance of excessive washing or irritants, food provocation, and emotional stress, as well as toleration of body temperature fluctuations and humidity, is recommended to amend exacerbations.5
Current topical therapies include emollients; corticosteroids; calcineurin inhibitors; and crisaborole, a newer phosphodiesterase 4 inhibitor. There are a number of emollients and moisturizers available, and one over-the-counter preparation showed tolerability and improved skin hydration in AD patients and demonstrated less transepidermal water loss than the control group.8 Ointments such as petrolatum usually do not include ingredients such as preservatives, gelling agents, or humectants that can promote stinging or burning.9 Topical corticosteroids, which ameliorate inflammation by subduing proinflammatory cytokine expression, have been the mainstay of treatment for more than 60 years; however, caution should be used due to the potential for side effects, mainly but not limited to systemic absorption in children, development of striae, and skin atrophy. Calcineurin inhibitors prohibit T-cell activity, modify mast cell response, and decrease dendritic cells in the epidermis. Since 2000, calcineurin inhibitors have been utilized as steroid-sparing agents10; however, prior authorization is still necessary with some insurance providers. Crisaborole ointment 2%, the newest topical agent for AD treatment in the market, has shown improvement of erythema, exudation, and pruritus. Approved for patients aged 2 years and older, twice-daily application of topical crisaborole as a steroid-sparing agent has rendered AD symptom relief.11 It has been reported that 4% of patients encounter stinging or burning with topical crisaborole application, whereas up to 50% of calcineurin inhibitors induce these adverse effects.12 Stabilized hypochlorous acid spray or gel acts as an antipruritic and antimicrobial agent, relieving pain associated with skin irritations. Topical antimicrobial preparations such as mupirocin 2% antibiotic ointment can reduce Staphylococcus colonization when applied in the nasal passage as well as to affected skin lesions.2
In children, UVA and UVB phototherapy has proven safe and effective and can be utilized in AD when suitable.13 When patients inadequately respond to topical therapies and phototherapy, systemic immunomodulatory agents have been recommended as treatment options.A child’s developing immune system indeed may be sensitive to systemic therapies as the innate immune system fully matures in adolescence and his/her adaptive immune system is undergoing vigorous definition.14 Systemic immunomodulatory agents such as cyclosporine, azathioprine, mycophenolate mofetil, and methotrexate have been used off label for years and pose certain challenges in being identified as durable therapies due to potential side effects. Cyclosporine is effective for the treatment of AD; however, long-term administration should be dosed up to a 12-month period and then stopped to decrease cumulative exposure to the drug. Therefore, further treatment options must be considered. For children, cyclosporine should be administered in a dose of 3 to 6 mg/kg daily. Fluctuations in blood pressure and renal function should be monitored. The recommended pediatric dose for azathioprine is 1 to 4 mg/kg daily with laboratory monitoring, particularly of liver enzymes and complete blood cell count. Obtaining the patient’s thiopurine methyltransferase level may aid in dosing. Gastrointestinal tract symptoms such as nausea, vomiting, and diarrhea are common. Phototherapy is not advised in conjunction with azathioprine due to an increased risk of photocarcinogenicity.13 The literature supporting mycophenolate mofetil in children with AD is limited.
Biologic therapies targeting IgE, B-lymphocyte antigen CD20, IL-5, thymic stromal lymphopoietin, TH17 cells, IL-12, IL-23, interferon gamma, IL-6 receptors, tumor necrosis factor, phosphodiesterase 4, Janus kinase, chymase, and nuclear receptors expressed on adipocytes and immune cells have undergone investigation for treatment of AD.17 Additionally, biologic agents targeting IL-31, IL-13, and IL-22 also have been evaluated.1 Currently, there are no US Food and Drug Administration–approved biologic agents for moderate to severe childhood AD.
Dupilumab, an IL-4Rα and IL-13Rα antagonist, recently has been approved for treatment of moderate to severe AD in adults but not yet for children. Potential side effects include nasopharyngitis, headache, hypersensitivity reactions, and ocular symptoms,11 namely keratitis and conjunctivitis.18 Less than 1% of patients experienced keratitis in clinical trials, while conjunctivitis was reported in 4% of patients taking dupilumab with topical corticosteroids at 52 weeks.18 However, possible ocular findings on slit-lamp examination in AD patients include atopic keratoconjunctivitis, blepharitis, palpebral conjunctival scarring, papillary conjunctival reaction, Horner-Trantas dots, keratoconus, and atopic cataracts. Spontaneous retinal detachment is seen more commonly in individuals with AD than in the general population.19
In clinical trials, hypersensitivity reactions included urticaria and serum sickness or serum sickness–like reactions in less than 1% of patients taking dupilumab.18
Conclusion
Childhood AD can be debilitating, and affected individuals often lead a poorer quality of life if left untreated. Embarrassment and isolation are commonly experienced. Increased responsibility and work in tending for a child with eczema may result in parental exhaustion.21 As with psoriasis, AD can impair activity and productivity.22 Currently, dupilumab has proven to positively impact health-related quality of life for adults.23 Pending the outcome of ongoing pediatric clinical trials, dupilumab may become a benchmark therapy for children younger than 18 years.
- Samalonis L. What’s new in eczema and atopic dermatitis research. The Dermatologist. November 19, 2015. http://www.the-dermatologist.com/content/whats-new-eczema-and-atopic-dermatitis-research. Accessed July 19, 2018.
- Habif T. Atopic dermatitis. In: Bonnet C, Pinczewski A, Cook L, eds. Clinical Dermatology. 5th ed. Edinburgh, Scotland: Mosby Elsevier; 2010:160-180.
- Fishbein AB, Mueller K, Kruse L, et al. Sleep disturbance in children with moderate/severe atopic dermatitis: a case control study [published online October 28, 2017]. J Am Acad Dermatol. 2018;78:336-341.
- Silverberg J. Association between childhood eczema and headaches: an analysis of 19 US population-based studies [published online August 29, 2015]. J Allergy Clin Immunol. 2016;137:492-499.e5.
- Bieber T, Bussmann C. Atopic dermatitis. In: Bolognia JL, Jorizzo JL, Schaffer JV, eds. Dermatology. 3rd ed. China: Saunders Elsevier; 2012:203-216.
- Suarez-Farinas M, Tintle S, Shemer A, et al. Non-lesional atopic dermatitis (AD) skin is characterized by broad terminal differentiation defects and variable immune abnormalities. J Allergy Clin Immunol. 2011;127:954-964.
- Hilton L. AD gene mutation identified: discovery may lead to new therapeutic option for patients. Dermatol Times. 2017;38:30.
- Zeichner JA, Dryer L. Effect of CeraVe Healing Ointment on skin hydration and barrier function on normal and barrier-impaired skin. Poster presented at: Orlando Dermatology Aesthetic & Clinical Conference; January 15-16, 2016; Orlando, FL.
- Garg T, Rath G, Goyal AK. Comprehensive review on additives of topical dosage forms for drug delivery. Drug Delivery. 2015;22:969-987.
- Eichenfield LF, Tom WL, Berger TG, et al. Guidelines of care for the management of atopic dermatitis section 2. management and treatment of atopic dermatitis with topical therapies. J Am Acad Dermatol. 2014;71:116-132.
- Koutnik-Fotopoulous E. Update on the latest eczema treatments. The Dermatologist. February 17, 2016. http://www.the-dermatologist.com/content/update-latest-eczema-treatments. Accessed August 16, 2018.
- Paller AS, Tom WL, Lebwohl MG, et al. Efficacy and safety of crisaborole ointment, a novel phosphodiesterase 4 inhibitor for the topical treatment of AD in children and adults [published online July 11, 2016]. J Am Acad Dermatol. 2016;75:494-503.
- Sidbury R, Davis D, Cohen D, et al. Guidelines of care for the management of atopic dermatitis: section 3. management and treatment with phototherapy and systemic agents . J Am Acad Dermatol. 2014;71:327-349.
- van der Merwe R, Gianella-Borradori A. Industry perspective on the clinical development of systemic products for the treatment of atopic dermatitis in pediatric patients with inadequate response to topical prescription therapy. Presented at: FDA Dermatologic and Ophthalmic Drugs Advisory Committee Meeting; March 9, 2015; Silver Spring, MD.
- Heller M, Shin HT, Orlow SJ, et al. Mycophenolate mofetil for severe childhood atopic dermatitis: experience in 14 patients. Br J Dermatol. 2007;157:127-132.
- Callen JP, Kulp-Shorten CL. Methotrexate. In: Wolverton SE, ed. Comprehensive Dermatologic Drug Therapy. 3rd ed. China: Saunders Elsevier; 2013:169-181.
- Guttman-Yassky E, Dhingra N, Leung DY. New era of biological therapeutics in atopic dermatitis [published online January 16, 2013]. Expert Opin Biol Ther. 2013;13:549-561.
- Dupixent [package insert]. Tarrytown, NY: Regeneron Pharmaceuticals, Inc; 2017.
- Lowery RS. Ophthalmologic manifestations of atopic dermatitis clinical presentation. Medscape website. emedicine.medscape.com/article/1197636-clinical#b4. Updated September 7, 2016. Accessed July 19, 2018.
- Lenz HJ. Management and preparedness for infusion and hypersensitivity reactions. Oncologist. 2007;12:601-609.
- Lewis-Jones S. Quality of life and childhood atopic dermatitis: the misery of living with childhood eczema. Int J Clin Pract. 2006;60:984-992.
- Eckert L, Gupta S, Amand C, et al. Impact of atopic dermatitis on health-related quality of life and productivity in adults in the Unites States: an analysis using the National Health and Wellness Survey. J Am Acad Dermatol, 2017;77:274-279.
- Tsianakas A, Luger TA, Radin A. Dupilumab treatment improves quality of life in adult patients with moderate-to-severe atopic dermatitis: results from a randomized, placebo-controlled clinical trial [published online January 11, 2018]. Br J Dermatol. 2018;178:406-414.
- Samalonis L. What’s new in eczema and atopic dermatitis research. The Dermatologist. November 19, 2015. http://www.the-dermatologist.com/content/whats-new-eczema-and-atopic-dermatitis-research. Accessed July 19, 2018.
- Habif T. Atopic dermatitis. In: Bonnet C, Pinczewski A, Cook L, eds. Clinical Dermatology. 5th ed. Edinburgh, Scotland: Mosby Elsevier; 2010:160-180.
- Fishbein AB, Mueller K, Kruse L, et al. Sleep disturbance in children with moderate/severe atopic dermatitis: a case control study [published online October 28, 2017]. J Am Acad Dermatol. 2018;78:336-341.
- Silverberg J. Association between childhood eczema and headaches: an analysis of 19 US population-based studies [published online August 29, 2015]. J Allergy Clin Immunol. 2016;137:492-499.e5.
- Bieber T, Bussmann C. Atopic dermatitis. In: Bolognia JL, Jorizzo JL, Schaffer JV, eds. Dermatology. 3rd ed. China: Saunders Elsevier; 2012:203-216.
- Suarez-Farinas M, Tintle S, Shemer A, et al. Non-lesional atopic dermatitis (AD) skin is characterized by broad terminal differentiation defects and variable immune abnormalities. J Allergy Clin Immunol. 2011;127:954-964.
- Hilton L. AD gene mutation identified: discovery may lead to new therapeutic option for patients. Dermatol Times. 2017;38:30.
- Zeichner JA, Dryer L. Effect of CeraVe Healing Ointment on skin hydration and barrier function on normal and barrier-impaired skin. Poster presented at: Orlando Dermatology Aesthetic & Clinical Conference; January 15-16, 2016; Orlando, FL.
- Garg T, Rath G, Goyal AK. Comprehensive review on additives of topical dosage forms for drug delivery. Drug Delivery. 2015;22:969-987.
- Eichenfield LF, Tom WL, Berger TG, et al. Guidelines of care for the management of atopic dermatitis section 2. management and treatment of atopic dermatitis with topical therapies. J Am Acad Dermatol. 2014;71:116-132.
- Koutnik-Fotopoulous E. Update on the latest eczema treatments. The Dermatologist. February 17, 2016. http://www.the-dermatologist.com/content/update-latest-eczema-treatments. Accessed August 16, 2018.
- Paller AS, Tom WL, Lebwohl MG, et al. Efficacy and safety of crisaborole ointment, a novel phosphodiesterase 4 inhibitor for the topical treatment of AD in children and adults [published online July 11, 2016]. J Am Acad Dermatol. 2016;75:494-503.
- Sidbury R, Davis D, Cohen D, et al. Guidelines of care for the management of atopic dermatitis: section 3. management and treatment with phototherapy and systemic agents . J Am Acad Dermatol. 2014;71:327-349.
- van der Merwe R, Gianella-Borradori A. Industry perspective on the clinical development of systemic products for the treatment of atopic dermatitis in pediatric patients with inadequate response to topical prescription therapy. Presented at: FDA Dermatologic and Ophthalmic Drugs Advisory Committee Meeting; March 9, 2015; Silver Spring, MD.
- Heller M, Shin HT, Orlow SJ, et al. Mycophenolate mofetil for severe childhood atopic dermatitis: experience in 14 patients. Br J Dermatol. 2007;157:127-132.
- Callen JP, Kulp-Shorten CL. Methotrexate. In: Wolverton SE, ed. Comprehensive Dermatologic Drug Therapy. 3rd ed. China: Saunders Elsevier; 2013:169-181.
- Guttman-Yassky E, Dhingra N, Leung DY. New era of biological therapeutics in atopic dermatitis [published online January 16, 2013]. Expert Opin Biol Ther. 2013;13:549-561.
- Dupixent [package insert]. Tarrytown, NY: Regeneron Pharmaceuticals, Inc; 2017.
- Lowery RS. Ophthalmologic manifestations of atopic dermatitis clinical presentation. Medscape website. emedicine.medscape.com/article/1197636-clinical#b4. Updated September 7, 2016. Accessed July 19, 2018.
- Lenz HJ. Management and preparedness for infusion and hypersensitivity reactions. Oncologist. 2007;12:601-609.
- Lewis-Jones S. Quality of life and childhood atopic dermatitis: the misery of living with childhood eczema. Int J Clin Pract. 2006;60:984-992.
- Eckert L, Gupta S, Amand C, et al. Impact of atopic dermatitis on health-related quality of life and productivity in adults in the Unites States: an analysis using the National Health and Wellness Survey. J Am Acad Dermatol, 2017;77:274-279.
- Tsianakas A, Luger TA, Radin A. Dupilumab treatment improves quality of life in adult patients with moderate-to-severe atopic dermatitis: results from a randomized, placebo-controlled clinical trial [published online January 11, 2018]. Br J Dermatol. 2018;178:406-414.
Practice Points
- Childhood atopic dermatitis can be debilitating, and affected individuals often experience poorer quality of life if left untreated.
- Dupilumab may become a benchmark therapy for children younger than 18 years.
Molluscum Contagiosum Virus Infection Can Trigger Atopic Dermatitis Disease Onset or Flare
Molluscum contagiosum virus (MCV) is a common pediatric viral infection of the skin and/or mucous membranes.1 It has been noted in increasingly younger patient populations, ranging from congenital cases resulting from perinatal/vertical transmission to transmission from cobathing and pool usage.2,3
An association between MCV infection and atopic dermatitis (AD) has been reported to be caused by a predisposition to prolonged and severe cutaneous viral infections.4 However, the exact nature of the relationship between MCV and AD is unknown.
The purpose of this study was to identify pediatric patients with AD onset or flare of AD triggered by MCV infection as well as to characterize the setting under which MCV may trigger AD onset or flares in children.
Methods
Medical records for 50 children with prior or current MCV infection who presented sequentially to an outpatient pediatric dermatology practice over a 1-month period were identified. Institutional review board approval was obtained.
Results
The age range of the 50 patients with MCV infection was 1 to 13 years, with an average age of 3.6 years at the onset of infection (reported by parents/guardians) and 4.5 years at presentation to the pediatric dermatology office (Table 1).
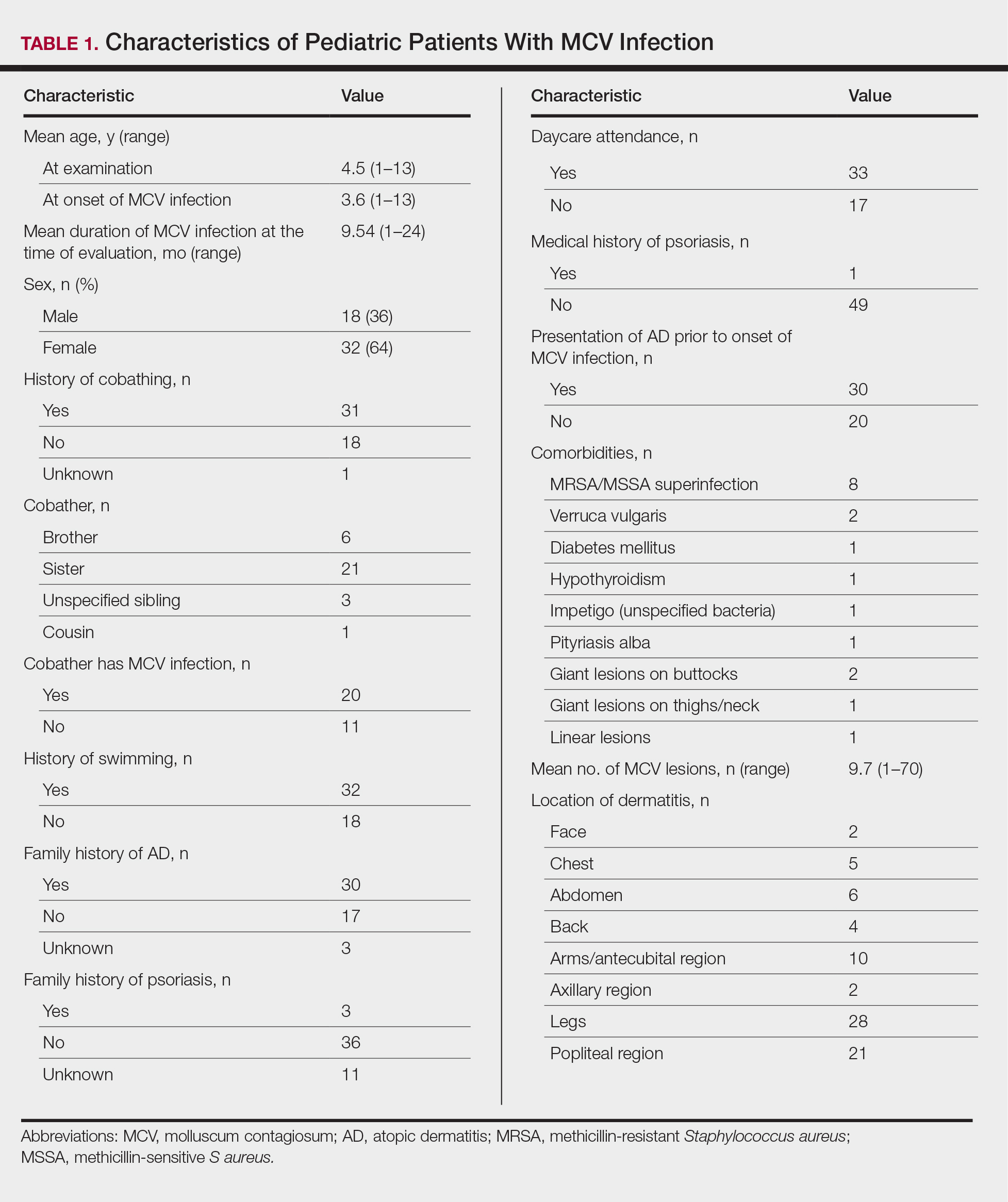
The role of cobathing is unknown; however, 62% (31/50) of patients previously or currently cobathed at home, suggesting it may be a risk factor for MCV infection. An association of MCV lesions in the popliteal region trended toward being more likely with cobathing, but the association was not statistically significant.
Children with AD onset triggered by MCV infection statistically were more likely to have flexural localization of MCV and AD lesions and were statistically more likely to have a family history of AD (P<.04)(Table 2). Children with AD flares triggered by MCV infection were more likely to have MCV and AD lesions of the popliteal region and legs (P<.05)(Figure) and family history of AD (P<.04)(Table 3). Location of MCV lesions on the upper and lower extremities, buttocks, and genitalia were more likely to be associated with presence of any dermatitis than facial and/or truncal lesions (P<.05). Treatment of the MCV infection did not appear to impact the course of AD when present, but prospective interventions would be needed to assess this issue.
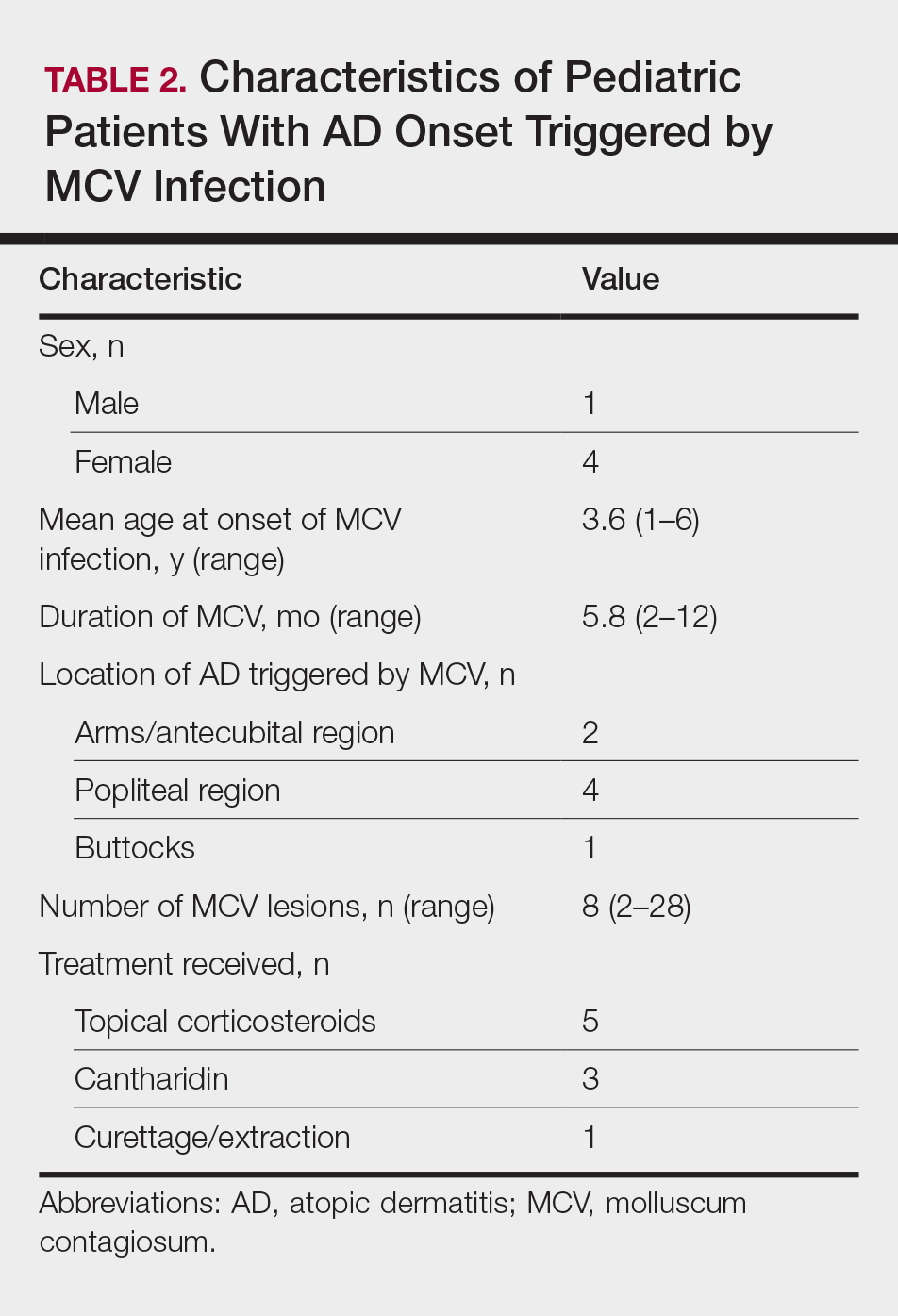
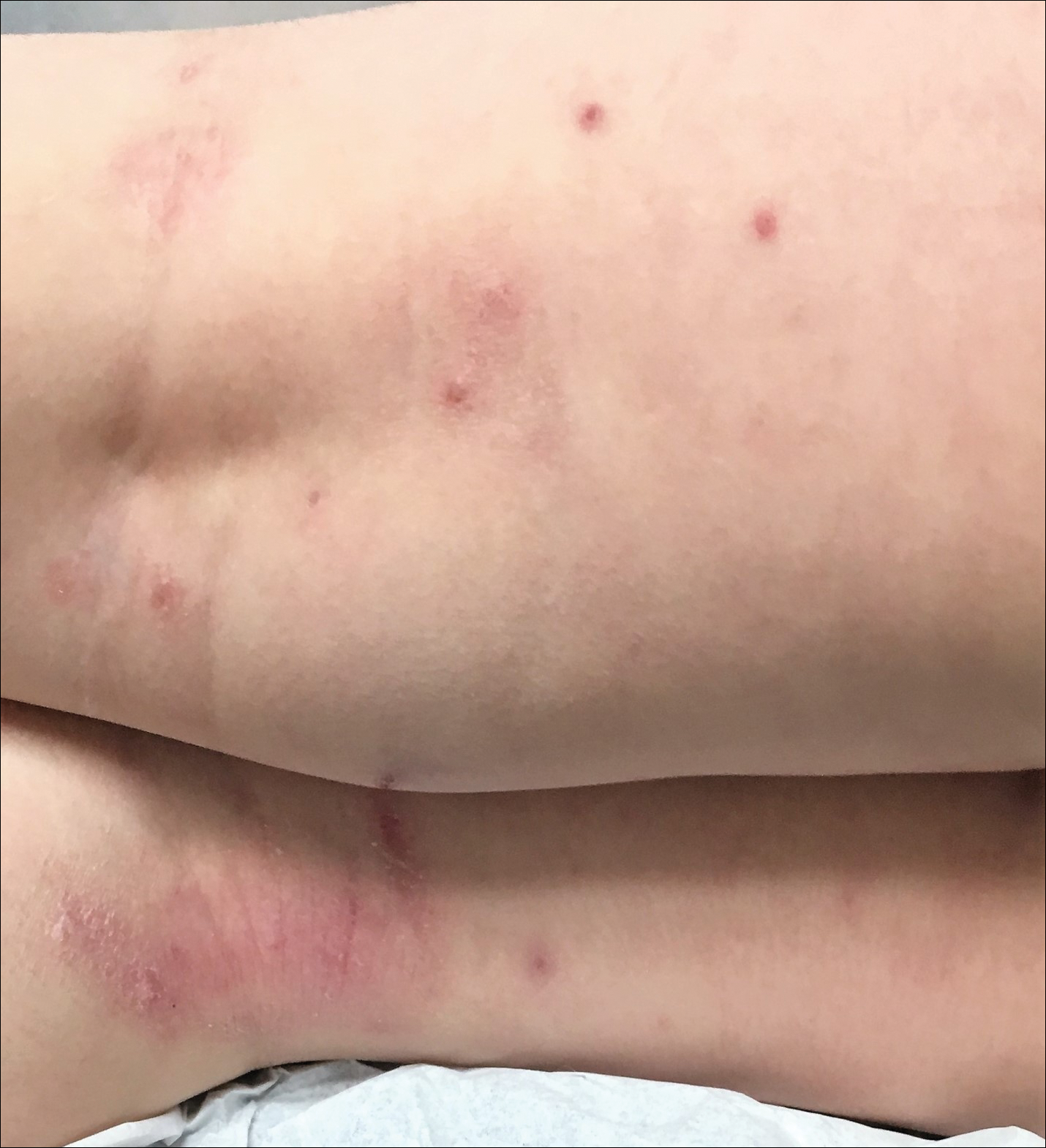
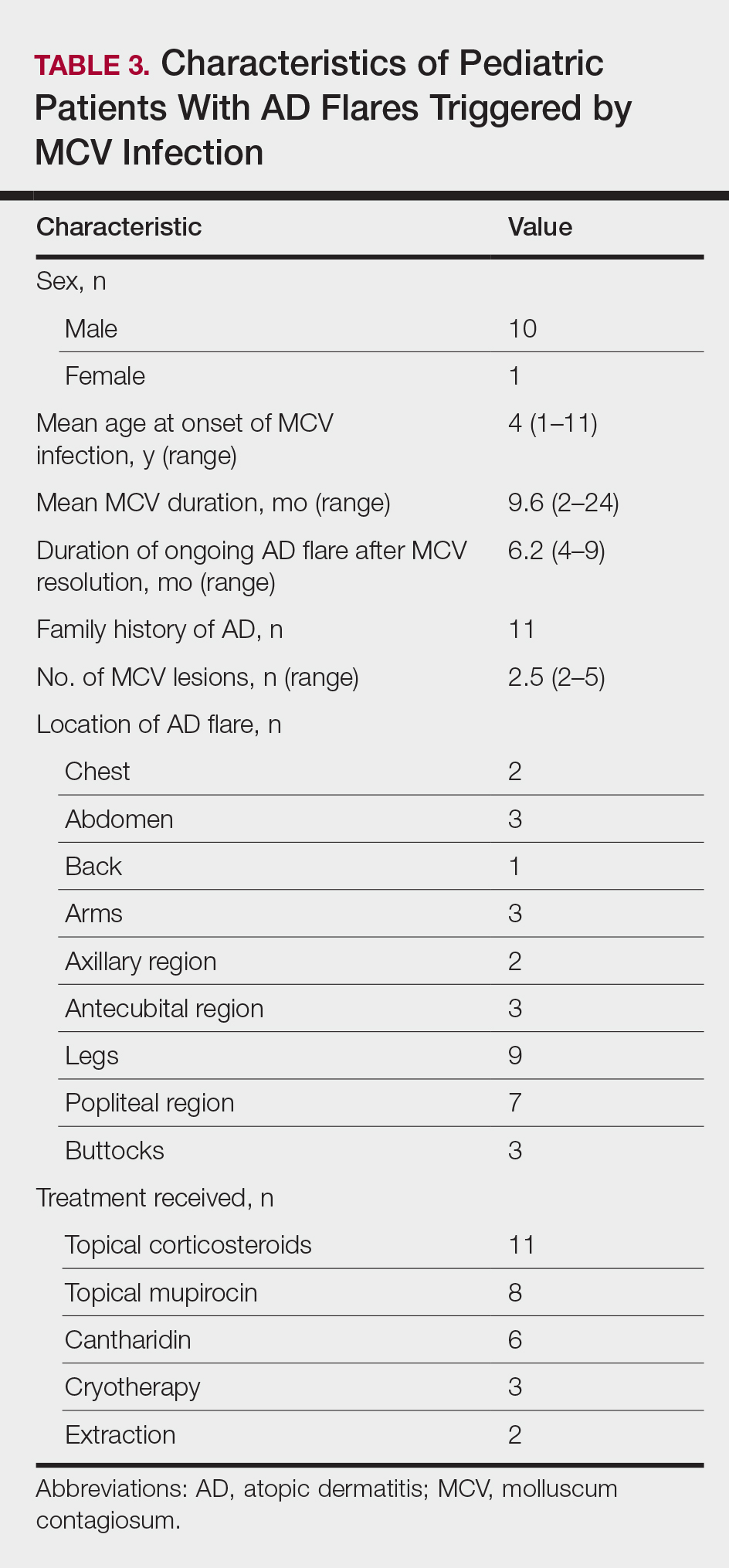
Superinfection with methicillin-resistant and methicillin-sensitive Staphylococcus aureus as well as atypical giant lesions of the intertriginous neck, inner thighs, and buttocks also were noted, but AD was uncommon in these cases. Given the limited number of cases, statistical significance could not be assessed.
Comment
Cutaneous infections with Malassezia have been postulated to trigger AD in infancy,1 while systemic viral infections such as varicella-zoster virus may be protective against AD when acquired in younger children.7 It appears that MCV infection in young children (eg, 3 years or younger) with specific localization to the flexural areas has the potential to trigger AD in susceptible hosts. Larger studies are needed to chart the long-term disease course of AD in these children. Due to the small size of this study, it is unclear if the rise of MCV infections since the 1980s has contributed to increased AD.8 Susceptible children appear to have a family history of AD and localization of MCV lesions on the legs, buttocks, and antecubital region. Atopic dermatitis risk appears to be highest when MCV lesions are localized to intertriginous or flexural locations.
In addition to triggering the onset of AD, MCV infection also can trigger persistent flaring of AD, especially in the popliteal region and legs. Atopic dermatitis flares can occur at any age, but they appear to cluster in preschoolers and typically are not prevented by AD or MCV treatments; however, randomized trials are needed to identify if early intervention of MCV has a preventive benefit on AD onset or flares, and longer-term observation is needed to identify true disease course modification. Reduction of the number of MCV lesions previously has been demonstrated with institution of topical corticosteroid therapy.6 Therefore, institution of atopic skin care generally is advisable in the setting of MCV infection. Future studies should address the potential use of interventions to prevent the triggering of AD onset or flares in the setting of MCV infection in children.5
- Brown J, Janniger CK, Schwartz RA, et al. Childhood molluscum contagiosum. Int J Dermatol. 2006;45:93-99.
- Connell CO, Oranje A, Van Gysel D, et al. Congenital molluscum contagiosum: report of four cases and review of the literature. Pediatr Dermatol. 2008;25:553-556.
- Luke JD, Silverberg NB. Vertically transmitted molluscum contagiosum infection. Pediatrics. 2010;125:E423-E425.
- Olsen JR, Piguet V, Gallacher J, et al. Molluscum contagiosum and associations with atopic eczema in children: a retrospective longitudinal study in primary care. Br J Gen Pract. 2016;66:E53-E58.
- Basdag H, Rainer BM, Cohen BA. Molluscum contagiosum: to treat or not to treat? experience with 170 children in an outpatient clinic setting in the northeastern United States. Pediatr Dermatol. 2015;32:353-357.
- Berger EM, Orlow SJ, Patel RR, et al. Experience with molluscum contagiosum and associated inflammatory reactions in a pediatric dermatology practice: the bump that rashes. Arch Dermatol. 2012;148:1257-1264.
- Silverberg JI, Norowitz KB, Kleiman E, et al. Association between varicella zoster virus infection and atopic dermatitis in early and late childhood: a case-control study. J Allergy Clin Immunol. 2010;126:300-305.
- Oriel JD. The increase in molluscum contagiosum. Br Med J (Clin Res Ed). 1987;294:74.
Molluscum contagiosum virus (MCV) is a common pediatric viral infection of the skin and/or mucous membranes.1 It has been noted in increasingly younger patient populations, ranging from congenital cases resulting from perinatal/vertical transmission to transmission from cobathing and pool usage.2,3
An association between MCV infection and atopic dermatitis (AD) has been reported to be caused by a predisposition to prolonged and severe cutaneous viral infections.4 However, the exact nature of the relationship between MCV and AD is unknown.
The purpose of this study was to identify pediatric patients with AD onset or flare of AD triggered by MCV infection as well as to characterize the setting under which MCV may trigger AD onset or flares in children.
Methods
Medical records for 50 children with prior or current MCV infection who presented sequentially to an outpatient pediatric dermatology practice over a 1-month period were identified. Institutional review board approval was obtained.
Results
The age range of the 50 patients with MCV infection was 1 to 13 years, with an average age of 3.6 years at the onset of infection (reported by parents/guardians) and 4.5 years at presentation to the pediatric dermatology office (Table 1).

The role of cobathing is unknown; however, 62% (31/50) of patients previously or currently cobathed at home, suggesting it may be a risk factor for MCV infection. An association of MCV lesions in the popliteal region trended toward being more likely with cobathing, but the association was not statistically significant.
Children with AD onset triggered by MCV infection statistically were more likely to have flexural localization of MCV and AD lesions and were statistically more likely to have a family history of AD (P<.04)(Table 2). Children with AD flares triggered by MCV infection were more likely to have MCV and AD lesions of the popliteal region and legs (P<.05)(Figure) and family history of AD (P<.04)(Table 3). Location of MCV lesions on the upper and lower extremities, buttocks, and genitalia were more likely to be associated with presence of any dermatitis than facial and/or truncal lesions (P<.05). Treatment of the MCV infection did not appear to impact the course of AD when present, but prospective interventions would be needed to assess this issue.



Superinfection with methicillin-resistant and methicillin-sensitive Staphylococcus aureus as well as atypical giant lesions of the intertriginous neck, inner thighs, and buttocks also were noted, but AD was uncommon in these cases. Given the limited number of cases, statistical significance could not be assessed.
Comment
Cutaneous infections with Malassezia have been postulated to trigger AD in infancy,1 while systemic viral infections such as varicella-zoster virus may be protective against AD when acquired in younger children.7 It appears that MCV infection in young children (eg, 3 years or younger) with specific localization to the flexural areas has the potential to trigger AD in susceptible hosts. Larger studies are needed to chart the long-term disease course of AD in these children. Due to the small size of this study, it is unclear if the rise of MCV infections since the 1980s has contributed to increased AD.8 Susceptible children appear to have a family history of AD and localization of MCV lesions on the legs, buttocks, and antecubital region. Atopic dermatitis risk appears to be highest when MCV lesions are localized to intertriginous or flexural locations.
In addition to triggering the onset of AD, MCV infection also can trigger persistent flaring of AD, especially in the popliteal region and legs. Atopic dermatitis flares can occur at any age, but they appear to cluster in preschoolers and typically are not prevented by AD or MCV treatments; however, randomized trials are needed to identify if early intervention of MCV has a preventive benefit on AD onset or flares, and longer-term observation is needed to identify true disease course modification. Reduction of the number of MCV lesions previously has been demonstrated with institution of topical corticosteroid therapy.6 Therefore, institution of atopic skin care generally is advisable in the setting of MCV infection. Future studies should address the potential use of interventions to prevent the triggering of AD onset or flares in the setting of MCV infection in children.5
Molluscum contagiosum virus (MCV) is a common pediatric viral infection of the skin and/or mucous membranes.1 It has been noted in increasingly younger patient populations, ranging from congenital cases resulting from perinatal/vertical transmission to transmission from cobathing and pool usage.2,3
An association between MCV infection and atopic dermatitis (AD) has been reported to be caused by a predisposition to prolonged and severe cutaneous viral infections.4 However, the exact nature of the relationship between MCV and AD is unknown.
The purpose of this study was to identify pediatric patients with AD onset or flare of AD triggered by MCV infection as well as to characterize the setting under which MCV may trigger AD onset or flares in children.
Methods
Medical records for 50 children with prior or current MCV infection who presented sequentially to an outpatient pediatric dermatology practice over a 1-month period were identified. Institutional review board approval was obtained.
Results
The age range of the 50 patients with MCV infection was 1 to 13 years, with an average age of 3.6 years at the onset of infection (reported by parents/guardians) and 4.5 years at presentation to the pediatric dermatology office (Table 1).

The role of cobathing is unknown; however, 62% (31/50) of patients previously or currently cobathed at home, suggesting it may be a risk factor for MCV infection. An association of MCV lesions in the popliteal region trended toward being more likely with cobathing, but the association was not statistically significant.
Children with AD onset triggered by MCV infection statistically were more likely to have flexural localization of MCV and AD lesions and were statistically more likely to have a family history of AD (P<.04)(Table 2). Children with AD flares triggered by MCV infection were more likely to have MCV and AD lesions of the popliteal region and legs (P<.05)(Figure) and family history of AD (P<.04)(Table 3). Location of MCV lesions on the upper and lower extremities, buttocks, and genitalia were more likely to be associated with presence of any dermatitis than facial and/or truncal lesions (P<.05). Treatment of the MCV infection did not appear to impact the course of AD when present, but prospective interventions would be needed to assess this issue.



Superinfection with methicillin-resistant and methicillin-sensitive Staphylococcus aureus as well as atypical giant lesions of the intertriginous neck, inner thighs, and buttocks also were noted, but AD was uncommon in these cases. Given the limited number of cases, statistical significance could not be assessed.
Comment
Cutaneous infections with Malassezia have been postulated to trigger AD in infancy,1 while systemic viral infections such as varicella-zoster virus may be protective against AD when acquired in younger children.7 It appears that MCV infection in young children (eg, 3 years or younger) with specific localization to the flexural areas has the potential to trigger AD in susceptible hosts. Larger studies are needed to chart the long-term disease course of AD in these children. Due to the small size of this study, it is unclear if the rise of MCV infections since the 1980s has contributed to increased AD.8 Susceptible children appear to have a family history of AD and localization of MCV lesions on the legs, buttocks, and antecubital region. Atopic dermatitis risk appears to be highest when MCV lesions are localized to intertriginous or flexural locations.
In addition to triggering the onset of AD, MCV infection also can trigger persistent flaring of AD, especially in the popliteal region and legs. Atopic dermatitis flares can occur at any age, but they appear to cluster in preschoolers and typically are not prevented by AD or MCV treatments; however, randomized trials are needed to identify if early intervention of MCV has a preventive benefit on AD onset or flares, and longer-term observation is needed to identify true disease course modification. Reduction of the number of MCV lesions previously has been demonstrated with institution of topical corticosteroid therapy.6 Therefore, institution of atopic skin care generally is advisable in the setting of MCV infection. Future studies should address the potential use of interventions to prevent the triggering of AD onset or flares in the setting of MCV infection in children.5
- Brown J, Janniger CK, Schwartz RA, et al. Childhood molluscum contagiosum. Int J Dermatol. 2006;45:93-99.
- Connell CO, Oranje A, Van Gysel D, et al. Congenital molluscum contagiosum: report of four cases and review of the literature. Pediatr Dermatol. 2008;25:553-556.
- Luke JD, Silverberg NB. Vertically transmitted molluscum contagiosum infection. Pediatrics. 2010;125:E423-E425.
- Olsen JR, Piguet V, Gallacher J, et al. Molluscum contagiosum and associations with atopic eczema in children: a retrospective longitudinal study in primary care. Br J Gen Pract. 2016;66:E53-E58.
- Basdag H, Rainer BM, Cohen BA. Molluscum contagiosum: to treat or not to treat? experience with 170 children in an outpatient clinic setting in the northeastern United States. Pediatr Dermatol. 2015;32:353-357.
- Berger EM, Orlow SJ, Patel RR, et al. Experience with molluscum contagiosum and associated inflammatory reactions in a pediatric dermatology practice: the bump that rashes. Arch Dermatol. 2012;148:1257-1264.
- Silverberg JI, Norowitz KB, Kleiman E, et al. Association between varicella zoster virus infection and atopic dermatitis in early and late childhood: a case-control study. J Allergy Clin Immunol. 2010;126:300-305.
- Oriel JD. The increase in molluscum contagiosum. Br Med J (Clin Res Ed). 1987;294:74.
- Brown J, Janniger CK, Schwartz RA, et al. Childhood molluscum contagiosum. Int J Dermatol. 2006;45:93-99.
- Connell CO, Oranje A, Van Gysel D, et al. Congenital molluscum contagiosum: report of four cases and review of the literature. Pediatr Dermatol. 2008;25:553-556.
- Luke JD, Silverberg NB. Vertically transmitted molluscum contagiosum infection. Pediatrics. 2010;125:E423-E425.
- Olsen JR, Piguet V, Gallacher J, et al. Molluscum contagiosum and associations with atopic eczema in children: a retrospective longitudinal study in primary care. Br J Gen Pract. 2016;66:E53-E58.
- Basdag H, Rainer BM, Cohen BA. Molluscum contagiosum: to treat or not to treat? experience with 170 children in an outpatient clinic setting in the northeastern United States. Pediatr Dermatol. 2015;32:353-357.
- Berger EM, Orlow SJ, Patel RR, et al. Experience with molluscum contagiosum and associated inflammatory reactions in a pediatric dermatology practice: the bump that rashes. Arch Dermatol. 2012;148:1257-1264.
- Silverberg JI, Norowitz KB, Kleiman E, et al. Association between varicella zoster virus infection and atopic dermatitis in early and late childhood: a case-control study. J Allergy Clin Immunol. 2010;126:300-305.
- Oriel JD. The increase in molluscum contagiosum. Br Med J (Clin Res Ed). 1987;294:74.
Practice Points
- Molluscum contagiosum virus (MCV) infection appears to aggravate atopic dermatitis (AD) symptoms in a subset of pediatric patients.
- In susceptible children, the first onset of AD symptoms can occur during the course of MCV infection.
Atopic Dermatitis Pipeline
Just when you might have thought dermatologic therapies were peaking, along came another banner year in atopic dermatitis (AD). Last year we saw the landmark launch of dupilumab, the first US Food and Drug Administration (FDA)–approved biologic therapy for AD. Dupilumab addresses a novel mechanism of AD in adults by blocking IL-4 and IL-13, which both play a central role in the type 2 helper T cell (TH2) axis on the dual development of barrier-impaired skin and aberrant immune response including IgE to cutaneous aggravating agents with resultant inflammation. Additional information has shown direct effects to reduce itch in AD.1 A 12-week study of dupilumab monotherapy showed that 85% (47/55) of treated patients had at least a 50% reduction in Eczema Area and Severity Index (EASI) score and 40% (22/55) were clear or almost clear on the investigator global assessment. With concomitant corticosteroid therapy, 100% of patients achieved EASI-50.2 Also notable, 2017 ushered in the appearance of a novel iteration of the 30-year-old concept of phosphodiesterase inhibition with the approval of the topical agent crisaborole for AD treatment in patients 2 years and older, which has been shown to be effective in both children and adults.3,4 However, despite these leaps of advancement in the care of AD, by no means has the condition been cured.
Atopic dermatitis has remained an incurable disease due to many factors: (1) variable immunologic and environmental triggers and patient disease course; (2) intolerance to therapeutic agents, including an enhanced sense of stinging and/or reactivity; (3) poor access to novel therapies among underserved patient populations; (4) lack of available data and information on variable treatment response by ethnicity and race; and (5) the absence of biologic treatments for severe childhood AD to modify long-term recurrence and progression of atopy, which is probably the most important issue, as the majority of AD cases start in children 5 years and younger.
Instituting a treatment today to provide children with disease-free skin for a lifetime truly is the Holy Grail in pediatric dermatology. To aid in the progress toward this goal, a deeper understanding of the manifestation of pediatric versus adult AD is now being investigated. It is clear that with adult chronicity, type 1 helper T cell (TH1) axis activity and prolonged defects are triggered in barrier maturation; however, recent data have started to demonstrate that the youngest patients have different issues in lipid maturation and lack TH1 activation. In particular, fatty acyl-CoA reductase 2 and fatty acid 2-hydroxylase is preferentially downregulated in children.5 It appears that the young immune system may be ripe for immune modification, which previously has been demonstrated with wild-type viral infections of varicella in children.6 However, future research will focus on what kind of tweaks to the immune system are required.
To encapsulate the AD pipeline, we will review drug trials that are in active recruitment as well as recently published data, which constitute an exciting group full of modifications of current therapies and agents with novel mechanisms of action.
Therapies targeting new mechanisms of action include Janus kinase (JAK) inhibitors, which have shown promising results for alopecia areata and vitiligo vulgaris. These agents may create selective modification of the immune system and are being tested topically and orally (Clinicaltrials.gov identifier NCT03011892).
Another mechanism that currently is being studied includes a topical IL-4 and IL-13 inhibitor, which would hopefully mimic the efficacy of dupilumab, antioxidant therapies, and antimicrobials (NCT03351777, NCT03381625, NCT02910011).
Data on the outcome of a phase 3 trial of dupilumab in adolescents has been released but not yet published by the manufacturer and shows promising results in children aged 12 to 17 years, both in reduction of EASI score and in achieving clear or almost clear skin.11 Interestingly, limited data available from a press release reported similar results with dupilumab injection every 2 weeks versus every 4 weeks, which may give alternative dosing regimens in this age group once approved11; however, publication has yet to occur for the latter data.
Other mechanistic agents include blockade of cytokines and interleukins, particularly those involved in type 2 helper T cell (TH2) activity, such as thymic stromal lymphopoietin (a cytokine), as well as targeted single inhibition of IL-4, IL-5, IL-13, and IL-31 and/or their receptors. Nemolizumab, an anti–IL-31 receptor A antibody, is showing promise in the control of AD-associated itch and reduction in EASI
The future of AD therapy is anyone’s guess. Having entered the biologic era with dupilumab, we have a high bar set for efficacy and safety of AD therapies, yet there remains a core group of AD patients who have not yet achieved clearance or refuse injectables; therefore, adjunctive or alternative therapeutics are still needed. Furthermore, we still have not identified who will best benefit long-term from systemic intervention and how to best effect long-term disease control with biologics or novel agents, and choosing the therapy based on patient disease characteristics or serotyping has not yet come of age. It is exciting to think about what next year will bring!
- Xu X, Zheng Y, Zhang X, et al. Efficacy and safety of dupilumab for the treatment of moderate-to-severe atopic dermatitis in adults. Oncotarget. 2017;8:108480-108491.
- Beck LA, Thaçi D, Hamilton JD, et al. Dupilumab treatment in adults with moderate-to-severe atopic dermatitis. N Engl J Med. 2014;371:130-139.
- Murrell D, Gebauer K, Spelman L, et al. Crisaborole topical ointment, 2% in adults with atopic dermatitis: a phase 2a, vehicle-controlled, proof-of-concept study. J Drugs Dermatol. 2015;14:1108-1112.
- Paller AS, Tom WL, Lebwohl MG, et al. Efficacy and safety of crisaborole ointment, a novel, nonsteroidal phosphodiesterase 4 (PDE4) inhibitor for the topical treatment of atopic dermatitis (AD) in children and adults. J Am Acad Dermatol. 2016;75:494-503.e6.
- Brunner PM, Israel A, Zhang N, et al. Early-onset pediatric atopic dermatitis is characterized by TH2/TH17/TH22-centered inflammation and lipid alterations. J Allergy Clin Immunol. 2018;141:2094-2106.
- Silverberg JI, Kleiman E, Silverberg NB, et al. Chickenpox in childhood is associated with decreased atopic disorders, IgE, allergic sensitization, and leukocyte subsets. Pediatr Allergy Immunol. 2012;23:50-58.
- Paller AS, Kabashima K, Bieber T. Therapeutic pipeline for atopic dermatitis: end of the drought? Allergy Clin Immunol. 2017;140:633-643.
- Renert-Yuval Y, Guttman-Yassky E. Systemic therapies in atopic dermatitis: the pipeline. Clin Dermatol. 2017;35:387-397.
- Bissonnette R, Papp KA, Poulin Y, et al. Topical tofacitinib for atopic dermatitis: a phase IIa randomized trial. Br J Dermatol. 2016;175:902-911.
- Guttman-Yassky E, Silverberg JI, Nemoto O, et al. Baricitinib in adult patients with moderate-to-severe atopic dermatitis: a phase 2 parallel, double-blinded, randomized placebo-controlled multiple-dose study [published online February 1, 2018]. J Am Acad Dermatol. doi: 10.1016/j.jaad.2018.01.018.
- Dupixent (dupilumab) showed positive phase 3 results in adolescents with inadequately controlled moderate-to-severe atopic dermatitis [press release]. Tarrytown, NY: Sanofi; May 16, 2018. https://www.prnewswire.com/news-releases/dupixent-dupilumab-showed-positive-phase-3-results-in-adolescents-with-inadequately-controlled-moderate-to-severe-atopic-dermatitis-300649146.html. Accessed July 11, 2018.
- Ruzicka T, Hanifin JM, Furue M, et al. Anti–interleukin-31 receptor A antibody for atopic dermatitis. N Engl J Med. 2017;376:826-835.
Just when you might have thought dermatologic therapies were peaking, along came another banner year in atopic dermatitis (AD). Last year we saw the landmark launch of dupilumab, the first US Food and Drug Administration (FDA)–approved biologic therapy for AD. Dupilumab addresses a novel mechanism of AD in adults by blocking IL-4 and IL-13, which both play a central role in the type 2 helper T cell (TH2) axis on the dual development of barrier-impaired skin and aberrant immune response including IgE to cutaneous aggravating agents with resultant inflammation. Additional information has shown direct effects to reduce itch in AD.1 A 12-week study of dupilumab monotherapy showed that 85% (47/55) of treated patients had at least a 50% reduction in Eczema Area and Severity Index (EASI) score and 40% (22/55) were clear or almost clear on the investigator global assessment. With concomitant corticosteroid therapy, 100% of patients achieved EASI-50.2 Also notable, 2017 ushered in the appearance of a novel iteration of the 30-year-old concept of phosphodiesterase inhibition with the approval of the topical agent crisaborole for AD treatment in patients 2 years and older, which has been shown to be effective in both children and adults.3,4 However, despite these leaps of advancement in the care of AD, by no means has the condition been cured.
Atopic dermatitis has remained an incurable disease due to many factors: (1) variable immunologic and environmental triggers and patient disease course; (2) intolerance to therapeutic agents, including an enhanced sense of stinging and/or reactivity; (3) poor access to novel therapies among underserved patient populations; (4) lack of available data and information on variable treatment response by ethnicity and race; and (5) the absence of biologic treatments for severe childhood AD to modify long-term recurrence and progression of atopy, which is probably the most important issue, as the majority of AD cases start in children 5 years and younger.
Instituting a treatment today to provide children with disease-free skin for a lifetime truly is the Holy Grail in pediatric dermatology. To aid in the progress toward this goal, a deeper understanding of the manifestation of pediatric versus adult AD is now being investigated. It is clear that with adult chronicity, type 1 helper T cell (TH1) axis activity and prolonged defects are triggered in barrier maturation; however, recent data have started to demonstrate that the youngest patients have different issues in lipid maturation and lack TH1 activation. In particular, fatty acyl-CoA reductase 2 and fatty acid 2-hydroxylase is preferentially downregulated in children.5 It appears that the young immune system may be ripe for immune modification, which previously has been demonstrated with wild-type viral infections of varicella in children.6 However, future research will focus on what kind of tweaks to the immune system are required.
To encapsulate the AD pipeline, we will review drug trials that are in active recruitment as well as recently published data, which constitute an exciting group full of modifications of current therapies and agents with novel mechanisms of action.
Therapies targeting new mechanisms of action include Janus kinase (JAK) inhibitors, which have shown promising results for alopecia areata and vitiligo vulgaris. These agents may create selective modification of the immune system and are being tested topically and orally (Clinicaltrials.gov identifier NCT03011892).
Another mechanism that currently is being studied includes a topical IL-4 and IL-13 inhibitor, which would hopefully mimic the efficacy of dupilumab, antioxidant therapies, and antimicrobials (NCT03351777, NCT03381625, NCT02910011).
Data on the outcome of a phase 3 trial of dupilumab in adolescents has been released but not yet published by the manufacturer and shows promising results in children aged 12 to 17 years, both in reduction of EASI score and in achieving clear or almost clear skin.11 Interestingly, limited data available from a press release reported similar results with dupilumab injection every 2 weeks versus every 4 weeks, which may give alternative dosing regimens in this age group once approved11; however, publication has yet to occur for the latter data.
Other mechanistic agents include blockade of cytokines and interleukins, particularly those involved in type 2 helper T cell (TH2) activity, such as thymic stromal lymphopoietin (a cytokine), as well as targeted single inhibition of IL-4, IL-5, IL-13, and IL-31 and/or their receptors. Nemolizumab, an anti–IL-31 receptor A antibody, is showing promise in the control of AD-associated itch and reduction in EASI
The future of AD therapy is anyone’s guess. Having entered the biologic era with dupilumab, we have a high bar set for efficacy and safety of AD therapies, yet there remains a core group of AD patients who have not yet achieved clearance or refuse injectables; therefore, adjunctive or alternative therapeutics are still needed. Furthermore, we still have not identified who will best benefit long-term from systemic intervention and how to best effect long-term disease control with biologics or novel agents, and choosing the therapy based on patient disease characteristics or serotyping has not yet come of age. It is exciting to think about what next year will bring!
Just when you might have thought dermatologic therapies were peaking, along came another banner year in atopic dermatitis (AD). Last year we saw the landmark launch of dupilumab, the first US Food and Drug Administration (FDA)–approved biologic therapy for AD. Dupilumab addresses a novel mechanism of AD in adults by blocking IL-4 and IL-13, which both play a central role in the type 2 helper T cell (TH2) axis on the dual development of barrier-impaired skin and aberrant immune response including IgE to cutaneous aggravating agents with resultant inflammation. Additional information has shown direct effects to reduce itch in AD.1 A 12-week study of dupilumab monotherapy showed that 85% (47/55) of treated patients had at least a 50% reduction in Eczema Area and Severity Index (EASI) score and 40% (22/55) were clear or almost clear on the investigator global assessment. With concomitant corticosteroid therapy, 100% of patients achieved EASI-50.2 Also notable, 2017 ushered in the appearance of a novel iteration of the 30-year-old concept of phosphodiesterase inhibition with the approval of the topical agent crisaborole for AD treatment in patients 2 years and older, which has been shown to be effective in both children and adults.3,4 However, despite these leaps of advancement in the care of AD, by no means has the condition been cured.
Atopic dermatitis has remained an incurable disease due to many factors: (1) variable immunologic and environmental triggers and patient disease course; (2) intolerance to therapeutic agents, including an enhanced sense of stinging and/or reactivity; (3) poor access to novel therapies among underserved patient populations; (4) lack of available data and information on variable treatment response by ethnicity and race; and (5) the absence of biologic treatments for severe childhood AD to modify long-term recurrence and progression of atopy, which is probably the most important issue, as the majority of AD cases start in children 5 years and younger.
Instituting a treatment today to provide children with disease-free skin for a lifetime truly is the Holy Grail in pediatric dermatology. To aid in the progress toward this goal, a deeper understanding of the manifestation of pediatric versus adult AD is now being investigated. It is clear that with adult chronicity, type 1 helper T cell (TH1) axis activity and prolonged defects are triggered in barrier maturation; however, recent data have started to demonstrate that the youngest patients have different issues in lipid maturation and lack TH1 activation. In particular, fatty acyl-CoA reductase 2 and fatty acid 2-hydroxylase is preferentially downregulated in children.5 It appears that the young immune system may be ripe for immune modification, which previously has been demonstrated with wild-type viral infections of varicella in children.6 However, future research will focus on what kind of tweaks to the immune system are required.
To encapsulate the AD pipeline, we will review drug trials that are in active recruitment as well as recently published data, which constitute an exciting group full of modifications of current therapies and agents with novel mechanisms of action.
Therapies targeting new mechanisms of action include Janus kinase (JAK) inhibitors, which have shown promising results for alopecia areata and vitiligo vulgaris. These agents may create selective modification of the immune system and are being tested topically and orally (Clinicaltrials.gov identifier NCT03011892).
Another mechanism that currently is being studied includes a topical IL-4 and IL-13 inhibitor, which would hopefully mimic the efficacy of dupilumab, antioxidant therapies, and antimicrobials (NCT03351777, NCT03381625, NCT02910011).
Data on the outcome of a phase 3 trial of dupilumab in adolescents has been released but not yet published by the manufacturer and shows promising results in children aged 12 to 17 years, both in reduction of EASI score and in achieving clear or almost clear skin.11 Interestingly, limited data available from a press release reported similar results with dupilumab injection every 2 weeks versus every 4 weeks, which may give alternative dosing regimens in this age group once approved11; however, publication has yet to occur for the latter data.
Other mechanistic agents include blockade of cytokines and interleukins, particularly those involved in type 2 helper T cell (TH2) activity, such as thymic stromal lymphopoietin (a cytokine), as well as targeted single inhibition of IL-4, IL-5, IL-13, and IL-31 and/or their receptors. Nemolizumab, an anti–IL-31 receptor A antibody, is showing promise in the control of AD-associated itch and reduction in EASI
The future of AD therapy is anyone’s guess. Having entered the biologic era with dupilumab, we have a high bar set for efficacy and safety of AD therapies, yet there remains a core group of AD patients who have not yet achieved clearance or refuse injectables; therefore, adjunctive or alternative therapeutics are still needed. Furthermore, we still have not identified who will best benefit long-term from systemic intervention and how to best effect long-term disease control with biologics or novel agents, and choosing the therapy based on patient disease characteristics or serotyping has not yet come of age. It is exciting to think about what next year will bring!
- Xu X, Zheng Y, Zhang X, et al. Efficacy and safety of dupilumab for the treatment of moderate-to-severe atopic dermatitis in adults. Oncotarget. 2017;8:108480-108491.
- Beck LA, Thaçi D, Hamilton JD, et al. Dupilumab treatment in adults with moderate-to-severe atopic dermatitis. N Engl J Med. 2014;371:130-139.
- Murrell D, Gebauer K, Spelman L, et al. Crisaborole topical ointment, 2% in adults with atopic dermatitis: a phase 2a, vehicle-controlled, proof-of-concept study. J Drugs Dermatol. 2015;14:1108-1112.
- Paller AS, Tom WL, Lebwohl MG, et al. Efficacy and safety of crisaborole ointment, a novel, nonsteroidal phosphodiesterase 4 (PDE4) inhibitor for the topical treatment of atopic dermatitis (AD) in children and adults. J Am Acad Dermatol. 2016;75:494-503.e6.
- Brunner PM, Israel A, Zhang N, et al. Early-onset pediatric atopic dermatitis is characterized by TH2/TH17/TH22-centered inflammation and lipid alterations. J Allergy Clin Immunol. 2018;141:2094-2106.
- Silverberg JI, Kleiman E, Silverberg NB, et al. Chickenpox in childhood is associated with decreased atopic disorders, IgE, allergic sensitization, and leukocyte subsets. Pediatr Allergy Immunol. 2012;23:50-58.
- Paller AS, Kabashima K, Bieber T. Therapeutic pipeline for atopic dermatitis: end of the drought? Allergy Clin Immunol. 2017;140:633-643.
- Renert-Yuval Y, Guttman-Yassky E. Systemic therapies in atopic dermatitis: the pipeline. Clin Dermatol. 2017;35:387-397.
- Bissonnette R, Papp KA, Poulin Y, et al. Topical tofacitinib for atopic dermatitis: a phase IIa randomized trial. Br J Dermatol. 2016;175:902-911.
- Guttman-Yassky E, Silverberg JI, Nemoto O, et al. Baricitinib in adult patients with moderate-to-severe atopic dermatitis: a phase 2 parallel, double-blinded, randomized placebo-controlled multiple-dose study [published online February 1, 2018]. J Am Acad Dermatol. doi: 10.1016/j.jaad.2018.01.018.
- Dupixent (dupilumab) showed positive phase 3 results in adolescents with inadequately controlled moderate-to-severe atopic dermatitis [press release]. Tarrytown, NY: Sanofi; May 16, 2018. https://www.prnewswire.com/news-releases/dupixent-dupilumab-showed-positive-phase-3-results-in-adolescents-with-inadequately-controlled-moderate-to-severe-atopic-dermatitis-300649146.html. Accessed July 11, 2018.
- Ruzicka T, Hanifin JM, Furue M, et al. Anti–interleukin-31 receptor A antibody for atopic dermatitis. N Engl J Med. 2017;376:826-835.
- Xu X, Zheng Y, Zhang X, et al. Efficacy and safety of dupilumab for the treatment of moderate-to-severe atopic dermatitis in adults. Oncotarget. 2017;8:108480-108491.
- Beck LA, Thaçi D, Hamilton JD, et al. Dupilumab treatment in adults with moderate-to-severe atopic dermatitis. N Engl J Med. 2014;371:130-139.
- Murrell D, Gebauer K, Spelman L, et al. Crisaborole topical ointment, 2% in adults with atopic dermatitis: a phase 2a, vehicle-controlled, proof-of-concept study. J Drugs Dermatol. 2015;14:1108-1112.
- Paller AS, Tom WL, Lebwohl MG, et al. Efficacy and safety of crisaborole ointment, a novel, nonsteroidal phosphodiesterase 4 (PDE4) inhibitor for the topical treatment of atopic dermatitis (AD) in children and adults. J Am Acad Dermatol. 2016;75:494-503.e6.
- Brunner PM, Israel A, Zhang N, et al. Early-onset pediatric atopic dermatitis is characterized by TH2/TH17/TH22-centered inflammation and lipid alterations. J Allergy Clin Immunol. 2018;141:2094-2106.
- Silverberg JI, Kleiman E, Silverberg NB, et al. Chickenpox in childhood is associated with decreased atopic disorders, IgE, allergic sensitization, and leukocyte subsets. Pediatr Allergy Immunol. 2012;23:50-58.
- Paller AS, Kabashima K, Bieber T. Therapeutic pipeline for atopic dermatitis: end of the drought? Allergy Clin Immunol. 2017;140:633-643.
- Renert-Yuval Y, Guttman-Yassky E. Systemic therapies in atopic dermatitis: the pipeline. Clin Dermatol. 2017;35:387-397.
- Bissonnette R, Papp KA, Poulin Y, et al. Topical tofacitinib for atopic dermatitis: a phase IIa randomized trial. Br J Dermatol. 2016;175:902-911.
- Guttman-Yassky E, Silverberg JI, Nemoto O, et al. Baricitinib in adult patients with moderate-to-severe atopic dermatitis: a phase 2 parallel, double-blinded, randomized placebo-controlled multiple-dose study [published online February 1, 2018]. J Am Acad Dermatol. doi: 10.1016/j.jaad.2018.01.018.
- Dupixent (dupilumab) showed positive phase 3 results in adolescents with inadequately controlled moderate-to-severe atopic dermatitis [press release]. Tarrytown, NY: Sanofi; May 16, 2018. https://www.prnewswire.com/news-releases/dupixent-dupilumab-showed-positive-phase-3-results-in-adolescents-with-inadequately-controlled-moderate-to-severe-atopic-dermatitis-300649146.html. Accessed July 11, 2018.
- Ruzicka T, Hanifin JM, Furue M, et al. Anti–interleukin-31 receptor A antibody for atopic dermatitis. N Engl J Med. 2017;376:826-835.
Climbing the therapeutic ladder in eczema-related itch
WASHINGTON – Currently available including antihistamines and an oral antiemetic approved for preventing chemotherapy-related nausea and vomiting, Peter Lio, MD, said at a symposium presented by the Coalition United for Better Eczema Care (CUBE-C).
There are four basic areas of treatment, which Dr. Lio, a dermatologist at Northwestern University, Chicago, referred to as the “itch therapeutic ladder.” In a video interview at the meeting, he reviewed the treatments, starting with topical therapies, which include camphor and menthol, strontium-containing topicals, as well as “dilute bleach-type products” that seem to have some anti-inflammatory and anti-itch effects.
The next levels: oral medications – antihistamines, followed by “more intense” options that may carry more risks, such as the antidepressant mirtazapine, and aprepitant, a neurokinin-1 receptor antagonist approved for the prevention of chemotherapy-induced and postoperative nausea and vomiting. Gabapentin and naltrexone can also be helpful for certain populations; all are used off-label, he pointed out.
Dr. Lio, formally trained in acupuncture, often uses alternative therapies as the fourth rung of the ladder. These include using a specific acupressure point, which he said “seems to give a little bit of relief.”
In the interview, he also discussed considerations in children with atopic dermatitis and exciting treatments in development, such as biologics that target “one of the master itch cytokines,” interleukin-31.
“Itch is such an important part of this disease because we know not only is it one of the key pieces that pushes the disease forward and keeps these cycles going, but also contributes a huge amount to the morbidity,” he said.
CUBE-C, established by the National Eczema Association (NEA), is a “network of cross-specialty leaders, patients and caregivers, constructing an educational curriculum based on standards of effective treatment and disease management,” according to the NEA.
The symposium was supported by an educational grant from Sanofi Genzyme, Regeneron Pharmaceuticals, and Pfizer. Dr. Lio reported serving as a speaker, consultant, and/or advisor for companies developing and marketing atopic dermatitis therapies and products.
WASHINGTON – Currently available including antihistamines and an oral antiemetic approved for preventing chemotherapy-related nausea and vomiting, Peter Lio, MD, said at a symposium presented by the Coalition United for Better Eczema Care (CUBE-C).
There are four basic areas of treatment, which Dr. Lio, a dermatologist at Northwestern University, Chicago, referred to as the “itch therapeutic ladder.” In a video interview at the meeting, he reviewed the treatments, starting with topical therapies, which include camphor and menthol, strontium-containing topicals, as well as “dilute bleach-type products” that seem to have some anti-inflammatory and anti-itch effects.
The next levels: oral medications – antihistamines, followed by “more intense” options that may carry more risks, such as the antidepressant mirtazapine, and aprepitant, a neurokinin-1 receptor antagonist approved for the prevention of chemotherapy-induced and postoperative nausea and vomiting. Gabapentin and naltrexone can also be helpful for certain populations; all are used off-label, he pointed out.
Dr. Lio, formally trained in acupuncture, often uses alternative therapies as the fourth rung of the ladder. These include using a specific acupressure point, which he said “seems to give a little bit of relief.”
In the interview, he also discussed considerations in children with atopic dermatitis and exciting treatments in development, such as biologics that target “one of the master itch cytokines,” interleukin-31.
“Itch is such an important part of this disease because we know not only is it one of the key pieces that pushes the disease forward and keeps these cycles going, but also contributes a huge amount to the morbidity,” he said.
CUBE-C, established by the National Eczema Association (NEA), is a “network of cross-specialty leaders, patients and caregivers, constructing an educational curriculum based on standards of effective treatment and disease management,” according to the NEA.
The symposium was supported by an educational grant from Sanofi Genzyme, Regeneron Pharmaceuticals, and Pfizer. Dr. Lio reported serving as a speaker, consultant, and/or advisor for companies developing and marketing atopic dermatitis therapies and products.
WASHINGTON – Currently available including antihistamines and an oral antiemetic approved for preventing chemotherapy-related nausea and vomiting, Peter Lio, MD, said at a symposium presented by the Coalition United for Better Eczema Care (CUBE-C).
There are four basic areas of treatment, which Dr. Lio, a dermatologist at Northwestern University, Chicago, referred to as the “itch therapeutic ladder.” In a video interview at the meeting, he reviewed the treatments, starting with topical therapies, which include camphor and menthol, strontium-containing topicals, as well as “dilute bleach-type products” that seem to have some anti-inflammatory and anti-itch effects.
The next levels: oral medications – antihistamines, followed by “more intense” options that may carry more risks, such as the antidepressant mirtazapine, and aprepitant, a neurokinin-1 receptor antagonist approved for the prevention of chemotherapy-induced and postoperative nausea and vomiting. Gabapentin and naltrexone can also be helpful for certain populations; all are used off-label, he pointed out.
Dr. Lio, formally trained in acupuncture, often uses alternative therapies as the fourth rung of the ladder. These include using a specific acupressure point, which he said “seems to give a little bit of relief.”
In the interview, he also discussed considerations in children with atopic dermatitis and exciting treatments in development, such as biologics that target “one of the master itch cytokines,” interleukin-31.
“Itch is such an important part of this disease because we know not only is it one of the key pieces that pushes the disease forward and keeps these cycles going, but also contributes a huge amount to the morbidity,” he said.
CUBE-C, established by the National Eczema Association (NEA), is a “network of cross-specialty leaders, patients and caregivers, constructing an educational curriculum based on standards of effective treatment and disease management,” according to the NEA.
The symposium was supported by an educational grant from Sanofi Genzyme, Regeneron Pharmaceuticals, and Pfizer. Dr. Lio reported serving as a speaker, consultant, and/or advisor for companies developing and marketing atopic dermatitis therapies and products.
CUBE-C initiative aims to educate about atopic dermatitis
WASHINGTON – The National Eczema Association (NEA) has established the Coalition United for Better Eczema Care (CUBE-C) to provide practitioners with a resource for “trustworthy, up-to-date, state of the art” information on atopic dermatitis, with the goal of improving health outcomes, according to Julie Block, president and chief executive officer of the NEA.
In an interview at a symposium presented by CUBE-C, Ms. Block provided more information on CUBE-C, including how and why it started and what it can offer to dermatologists, as well as primary care physicians, who care for patients with atopic dermatitis. She said that the NEA convened dermatologists, allergists, immunologists, psychologists, nurse practitioners, physician assistants, and patients “to design a curriculum that provided an entire picture of the patient experience, so that we could go out and educate not only on the basics of eczema and atopic dermatitis for a variety of practitioners ... but also for the specialists who are now going to be engaging in new innovations and new therapies for their patients.”
She was joined by Adam Friedman, MD, professor of dermatology and residency program director at George Washington University, Washington, where the symposium, a resident’s boot camp, was held. The boot camp was somewhat unique in that it was geared more towards trainees; typically, the CUBE-C program is a CME program for practitioners, but this reflects the flexibility of the program, which can be tailored to the audience, Dr. Friedman pointed out. “The hope is that programs like these pop up all over the place ... anywhere you have a critical mass of individuals who want to learn about this,” where planners can choose from a menu of topics provided by CUBE-C – which include therapeutics, infections, pathogenesis, and access to care – and “easily formulate a conference like we held here today for the right audience.”
Topics covered at the George Washington University symposium included the impact of climate on the prevalence of childhood eczema, the diagnosis and differential diagnosis in children, infections in atopic dermatitis patients, and itch treatment.
More information on CUBE-C is available on the NEA website.
The symposium was supported by an educational grant from Sanofi Genzyme, Regeneron Pharmaceuticals, and Pfizer. Dr. Friedman reported serving as a speaker for Regeneron, Pfizer, and other companies. He also consults and serves on the advisory board for Pfizer and multiple other companies developing and marketing atopic dermatitis therapies and products.
WASHINGTON – The National Eczema Association (NEA) has established the Coalition United for Better Eczema Care (CUBE-C) to provide practitioners with a resource for “trustworthy, up-to-date, state of the art” information on atopic dermatitis, with the goal of improving health outcomes, according to Julie Block, president and chief executive officer of the NEA.
In an interview at a symposium presented by CUBE-C, Ms. Block provided more information on CUBE-C, including how and why it started and what it can offer to dermatologists, as well as primary care physicians, who care for patients with atopic dermatitis. She said that the NEA convened dermatologists, allergists, immunologists, psychologists, nurse practitioners, physician assistants, and patients “to design a curriculum that provided an entire picture of the patient experience, so that we could go out and educate not only on the basics of eczema and atopic dermatitis for a variety of practitioners ... but also for the specialists who are now going to be engaging in new innovations and new therapies for their patients.”
She was joined by Adam Friedman, MD, professor of dermatology and residency program director at George Washington University, Washington, where the symposium, a resident’s boot camp, was held. The boot camp was somewhat unique in that it was geared more towards trainees; typically, the CUBE-C program is a CME program for practitioners, but this reflects the flexibility of the program, which can be tailored to the audience, Dr. Friedman pointed out. “The hope is that programs like these pop up all over the place ... anywhere you have a critical mass of individuals who want to learn about this,” where planners can choose from a menu of topics provided by CUBE-C – which include therapeutics, infections, pathogenesis, and access to care – and “easily formulate a conference like we held here today for the right audience.”
Topics covered at the George Washington University symposium included the impact of climate on the prevalence of childhood eczema, the diagnosis and differential diagnosis in children, infections in atopic dermatitis patients, and itch treatment.
More information on CUBE-C is available on the NEA website.
The symposium was supported by an educational grant from Sanofi Genzyme, Regeneron Pharmaceuticals, and Pfizer. Dr. Friedman reported serving as a speaker for Regeneron, Pfizer, and other companies. He also consults and serves on the advisory board for Pfizer and multiple other companies developing and marketing atopic dermatitis therapies and products.
WASHINGTON – The National Eczema Association (NEA) has established the Coalition United for Better Eczema Care (CUBE-C) to provide practitioners with a resource for “trustworthy, up-to-date, state of the art” information on atopic dermatitis, with the goal of improving health outcomes, according to Julie Block, president and chief executive officer of the NEA.
In an interview at a symposium presented by CUBE-C, Ms. Block provided more information on CUBE-C, including how and why it started and what it can offer to dermatologists, as well as primary care physicians, who care for patients with atopic dermatitis. She said that the NEA convened dermatologists, allergists, immunologists, psychologists, nurse practitioners, physician assistants, and patients “to design a curriculum that provided an entire picture of the patient experience, so that we could go out and educate not only on the basics of eczema and atopic dermatitis for a variety of practitioners ... but also for the specialists who are now going to be engaging in new innovations and new therapies for their patients.”
She was joined by Adam Friedman, MD, professor of dermatology and residency program director at George Washington University, Washington, where the symposium, a resident’s boot camp, was held. The boot camp was somewhat unique in that it was geared more towards trainees; typically, the CUBE-C program is a CME program for practitioners, but this reflects the flexibility of the program, which can be tailored to the audience, Dr. Friedman pointed out. “The hope is that programs like these pop up all over the place ... anywhere you have a critical mass of individuals who want to learn about this,” where planners can choose from a menu of topics provided by CUBE-C – which include therapeutics, infections, pathogenesis, and access to care – and “easily formulate a conference like we held here today for the right audience.”
Topics covered at the George Washington University symposium included the impact of climate on the prevalence of childhood eczema, the diagnosis and differential diagnosis in children, infections in atopic dermatitis patients, and itch treatment.
More information on CUBE-C is available on the NEA website.
The symposium was supported by an educational grant from Sanofi Genzyme, Regeneron Pharmaceuticals, and Pfizer. Dr. Friedman reported serving as a speaker for Regeneron, Pfizer, and other companies. He also consults and serves on the advisory board for Pfizer and multiple other companies developing and marketing atopic dermatitis therapies and products.
Immunosuppression often triggers skin side effects
LAKE TAHOE, CALIF. – , and side effects of vemurafenib treatment.
In a presentation on the cutaneous sequelae of different immunosuppressive regimens at the annual meeting of the Society for Pediatric Dermatology, Carrie C. Coughlin, MD, opened with a discussion of AD triggered by the tumor necrosis factor (TNF) blocker infliximab, especially in the setting of therapy for Crohn’s disease. “In this patient population you often think of psoriasis as a consequence of infliximab and other TNF therapies,” said Dr. Coughlin, a pediatric dermatologist at Washington University, St. Louis. “But you can also get true atopic dermatitis with infliximab as well. Who’s more at risk for this? Patients with Crohn’s disease seem to be. Most of the literature is in adults, but there are a few series of children. In a series of children looking at cutaneous sequelae of infliximab therapy, about 20% of cutaneous eruptions were atopic dermatitis. I think it’s a great opportunity for us in dermatology to do a more research in this area.”
Some researchers have proposed that atopic disease could be a marker of over-suppression of TNF-alpha in young Crohn’s disease patients on infliximab (Inflamm Bowel Dis. 2014; 20[8]:1309-15). “One question you could ask is, could these patients actually tolerate a dose reduction?” Dr. Coughlin said. She promoted the role of dermatologists in working at managing side effects to keep patients on medications helping their GI disease, but acknowledged this is not always possible.
Atopic disease can also occur after a solid organ transplant. In fact, the incidence of new-onset food allergies after a liver transplant is 6%-30% of cases, mainly in young patients (Pediatr Transplant. 2009;13[1]:63-9, Pediatr Transplant 2013;17[3]:251-5). “There are some mechanisms, including liver presentation of antigens, that spread through portal veins that could potentially put people at risk who have liver dysfunction,” Dr. Coughlin explained. “They could potentially have a higher risk for food allergies and AD. There is also some thought that tacrolimus potentially predisposes patients to having atopic dermatitis and atopy after their transplant. When you look at the mechanism of action of tacrolimus, you see increased levels of IL-5, IL-13, and skewing of IgE levels.”
Dr. Coughlin also discussed the possibilities of development of AD after transplant being a delayed presentation of an allergic sensitization that patients already had. Younger patients are at higher risk for AD post transplant. The renal transplant population, meanwhile, generally receives the transplant at a later age, “so they may not have that delay in terms of presentation; they may have already had their allergies and grown out of them by the time they’re getting their transplant,” she said. “I think there’s more for us to investigate.”
Solid organ transplant recipients also face an increased risk of skin cancer as a long-term side effect of immunosuppressive therapy. Risk factors include fair skin, sun exposure, and remote time from transplant. “Time from transplant is a risk factor,” Dr. Coughlin said. “Longer time on immunosuppression could predispose you to a risk for skin cancer. Our patients are living longer post transplant than they used to, so they have more potential years to develop their skin cancers.” She focused on the importance of educating transplant recipients and families early about photoprotection. “It’s interesting to think about how we can continue to intervene early on to continue to decrease risk.”
Young patients exposed to voriconazole also face an increased risk for skin cancer. “We know that longer-term dosing and higher cumulative dosing puts you at higher risk,” she said. Lung transplant recipients, who are often more likely to be treated with voriconazole, are thus at higher risk.
Dr. Coughlin ended her presentation by noting that side effects of the BRAF inhibitor vemurafenib (Zelboraf) used to treat advanced melanoma in children are similar to, but not the same as, those in adults. “We see BRAF mutations in multiple different tumor types: Langerhans cell histiocytosis, gliomas, and melanoma,” she said. “Trials of vemurafenib and dabrafenib are under way in the pediatric population. Vemurafenib can cause keratosis pilaris, panniculitis, alopecia, and granulomatous dermatitis.” In her experience, she has seen more alopecia in the older teenage population, but younger patients may not be asked about this side effect as frequently.
She counsels patients to expect keratosis pilaris–like eruptions and to take sun protection seriously. “It’s important to emphasize that each time they come in,” Dr. Coughlin said. She also discussed the potential for changing nevi and treatment options for vemurafenib-associated panniculitis.
Dr. Coughlin disclosed that she is the recipient of active pilot grants from the Pediatric Dermatology Research Alliance and the SPD.
LAKE TAHOE, CALIF. – , and side effects of vemurafenib treatment.
In a presentation on the cutaneous sequelae of different immunosuppressive regimens at the annual meeting of the Society for Pediatric Dermatology, Carrie C. Coughlin, MD, opened with a discussion of AD triggered by the tumor necrosis factor (TNF) blocker infliximab, especially in the setting of therapy for Crohn’s disease. “In this patient population you often think of psoriasis as a consequence of infliximab and other TNF therapies,” said Dr. Coughlin, a pediatric dermatologist at Washington University, St. Louis. “But you can also get true atopic dermatitis with infliximab as well. Who’s more at risk for this? Patients with Crohn’s disease seem to be. Most of the literature is in adults, but there are a few series of children. In a series of children looking at cutaneous sequelae of infliximab therapy, about 20% of cutaneous eruptions were atopic dermatitis. I think it’s a great opportunity for us in dermatology to do a more research in this area.”
Some researchers have proposed that atopic disease could be a marker of over-suppression of TNF-alpha in young Crohn’s disease patients on infliximab (Inflamm Bowel Dis. 2014; 20[8]:1309-15). “One question you could ask is, could these patients actually tolerate a dose reduction?” Dr. Coughlin said. She promoted the role of dermatologists in working at managing side effects to keep patients on medications helping their GI disease, but acknowledged this is not always possible.
Atopic disease can also occur after a solid organ transplant. In fact, the incidence of new-onset food allergies after a liver transplant is 6%-30% of cases, mainly in young patients (Pediatr Transplant. 2009;13[1]:63-9, Pediatr Transplant 2013;17[3]:251-5). “There are some mechanisms, including liver presentation of antigens, that spread through portal veins that could potentially put people at risk who have liver dysfunction,” Dr. Coughlin explained. “They could potentially have a higher risk for food allergies and AD. There is also some thought that tacrolimus potentially predisposes patients to having atopic dermatitis and atopy after their transplant. When you look at the mechanism of action of tacrolimus, you see increased levels of IL-5, IL-13, and skewing of IgE levels.”
Dr. Coughlin also discussed the possibilities of development of AD after transplant being a delayed presentation of an allergic sensitization that patients already had. Younger patients are at higher risk for AD post transplant. The renal transplant population, meanwhile, generally receives the transplant at a later age, “so they may not have that delay in terms of presentation; they may have already had their allergies and grown out of them by the time they’re getting their transplant,” she said. “I think there’s more for us to investigate.”
Solid organ transplant recipients also face an increased risk of skin cancer as a long-term side effect of immunosuppressive therapy. Risk factors include fair skin, sun exposure, and remote time from transplant. “Time from transplant is a risk factor,” Dr. Coughlin said. “Longer time on immunosuppression could predispose you to a risk for skin cancer. Our patients are living longer post transplant than they used to, so they have more potential years to develop their skin cancers.” She focused on the importance of educating transplant recipients and families early about photoprotection. “It’s interesting to think about how we can continue to intervene early on to continue to decrease risk.”
Young patients exposed to voriconazole also face an increased risk for skin cancer. “We know that longer-term dosing and higher cumulative dosing puts you at higher risk,” she said. Lung transplant recipients, who are often more likely to be treated with voriconazole, are thus at higher risk.
Dr. Coughlin ended her presentation by noting that side effects of the BRAF inhibitor vemurafenib (Zelboraf) used to treat advanced melanoma in children are similar to, but not the same as, those in adults. “We see BRAF mutations in multiple different tumor types: Langerhans cell histiocytosis, gliomas, and melanoma,” she said. “Trials of vemurafenib and dabrafenib are under way in the pediatric population. Vemurafenib can cause keratosis pilaris, panniculitis, alopecia, and granulomatous dermatitis.” In her experience, she has seen more alopecia in the older teenage population, but younger patients may not be asked about this side effect as frequently.
She counsels patients to expect keratosis pilaris–like eruptions and to take sun protection seriously. “It’s important to emphasize that each time they come in,” Dr. Coughlin said. She also discussed the potential for changing nevi and treatment options for vemurafenib-associated panniculitis.
Dr. Coughlin disclosed that she is the recipient of active pilot grants from the Pediatric Dermatology Research Alliance and the SPD.
LAKE TAHOE, CALIF. – , and side effects of vemurafenib treatment.
In a presentation on the cutaneous sequelae of different immunosuppressive regimens at the annual meeting of the Society for Pediatric Dermatology, Carrie C. Coughlin, MD, opened with a discussion of AD triggered by the tumor necrosis factor (TNF) blocker infliximab, especially in the setting of therapy for Crohn’s disease. “In this patient population you often think of psoriasis as a consequence of infliximab and other TNF therapies,” said Dr. Coughlin, a pediatric dermatologist at Washington University, St. Louis. “But you can also get true atopic dermatitis with infliximab as well. Who’s more at risk for this? Patients with Crohn’s disease seem to be. Most of the literature is in adults, but there are a few series of children. In a series of children looking at cutaneous sequelae of infliximab therapy, about 20% of cutaneous eruptions were atopic dermatitis. I think it’s a great opportunity for us in dermatology to do a more research in this area.”
Some researchers have proposed that atopic disease could be a marker of over-suppression of TNF-alpha in young Crohn’s disease patients on infliximab (Inflamm Bowel Dis. 2014; 20[8]:1309-15). “One question you could ask is, could these patients actually tolerate a dose reduction?” Dr. Coughlin said. She promoted the role of dermatologists in working at managing side effects to keep patients on medications helping their GI disease, but acknowledged this is not always possible.
Atopic disease can also occur after a solid organ transplant. In fact, the incidence of new-onset food allergies after a liver transplant is 6%-30% of cases, mainly in young patients (Pediatr Transplant. 2009;13[1]:63-9, Pediatr Transplant 2013;17[3]:251-5). “There are some mechanisms, including liver presentation of antigens, that spread through portal veins that could potentially put people at risk who have liver dysfunction,” Dr. Coughlin explained. “They could potentially have a higher risk for food allergies and AD. There is also some thought that tacrolimus potentially predisposes patients to having atopic dermatitis and atopy after their transplant. When you look at the mechanism of action of tacrolimus, you see increased levels of IL-5, IL-13, and skewing of IgE levels.”
Dr. Coughlin also discussed the possibilities of development of AD after transplant being a delayed presentation of an allergic sensitization that patients already had. Younger patients are at higher risk for AD post transplant. The renal transplant population, meanwhile, generally receives the transplant at a later age, “so they may not have that delay in terms of presentation; they may have already had their allergies and grown out of them by the time they’re getting their transplant,” she said. “I think there’s more for us to investigate.”
Solid organ transplant recipients also face an increased risk of skin cancer as a long-term side effect of immunosuppressive therapy. Risk factors include fair skin, sun exposure, and remote time from transplant. “Time from transplant is a risk factor,” Dr. Coughlin said. “Longer time on immunosuppression could predispose you to a risk for skin cancer. Our patients are living longer post transplant than they used to, so they have more potential years to develop their skin cancers.” She focused on the importance of educating transplant recipients and families early about photoprotection. “It’s interesting to think about how we can continue to intervene early on to continue to decrease risk.”
Young patients exposed to voriconazole also face an increased risk for skin cancer. “We know that longer-term dosing and higher cumulative dosing puts you at higher risk,” she said. Lung transplant recipients, who are often more likely to be treated with voriconazole, are thus at higher risk.
Dr. Coughlin ended her presentation by noting that side effects of the BRAF inhibitor vemurafenib (Zelboraf) used to treat advanced melanoma in children are similar to, but not the same as, those in adults. “We see BRAF mutations in multiple different tumor types: Langerhans cell histiocytosis, gliomas, and melanoma,” she said. “Trials of vemurafenib and dabrafenib are under way in the pediatric population. Vemurafenib can cause keratosis pilaris, panniculitis, alopecia, and granulomatous dermatitis.” In her experience, she has seen more alopecia in the older teenage population, but younger patients may not be asked about this side effect as frequently.
She counsels patients to expect keratosis pilaris–like eruptions and to take sun protection seriously. “It’s important to emphasize that each time they come in,” Dr. Coughlin said. She also discussed the potential for changing nevi and treatment options for vemurafenib-associated panniculitis.
Dr. Coughlin disclosed that she is the recipient of active pilot grants from the Pediatric Dermatology Research Alliance and the SPD.
REPORTING FROM SPD 2018
Advise parents on validity of AD-associated conditions
including allergic rhinitis and asthma, according to Douglas W. Kress, MD, of the department of dermatology at the University of Pittsburgh.
Recent studies suggest that atopic dermatitis (AD) affects 10%-17% of the U.S. population, and 80%-90% of patients are diagnosed by the age of 5 years, Dr. Kress said in a presentation at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
“There seem to be multiple pathways, which initiate and perpetuate the cutaneous inflammation of AD including exposure to allergens, irritants, and physical trauma, infection, stress, extremes in temperature and humidity,” Dr. Kress said. In addition, foods and airborne allergens may trigger AD.
Many parents may believe that certain factors are associated with AD, but most of these perceptions are not supported by evidence, said Dr. Kress, who is also chief of the division of pediatric dermatology at the Children’s Hospital of Pittsburgh. AD appears to be a disorder of T cell dysregulation dominated by Th2 lesions in acute cases and Th1 inflammation in patients with chronic lesions.
No associations have been proven between the development of AD in the first 18 months of life and any maternal dietary restrictions, according to a recent Cochrane review, nor is there evidence for an association between AD and the introduction of solid foods, exposure to fish oil, or exposure to animals at a young age, he said. In addition, a study published in 2016 showed a lack of evidence to support the use of specific allergen immunotherapy for AD.
However, evidence does support an association between the presence of AD in children and certain other conditions, Dr. Kress said. “Other associations include an increased incidence of alopecia areata, a threefold increase in autism spectrum disorders, and a twofold increase in ADHD in children with atopic dermatitis.”
The only known food allergy linked to AD severity is egg whites; reducing egg white exposure has been shown to improve AD in children with both conditions, he noted.
Although many patients with AD experience annoying but relatively mild symptoms, health care providers should be alert to the potential for infections, particularly with Staphylococcus aureus, and remember that an active egg white allergy has been associated with staphylococcal superantigen sensitization, said Dr. Kress. The increased risk for S. aureus in children with AD may stem from a tendency to underuse antibiotics in AD children, which results in a delayed treatment until the infection becomes overt. In addition, the increased pH in patients with AD might promote the development of pathogenic strains of staph. However, ceramide-based moisturizers could help inhibit these strains by increasing skin acidity.
For patients who have poor AD control with standard therapy, antibiotics may be used as adjunctive therapy. “Consider bleach baths and/or staph decolonization with mupirocin, both of which led to significant improvement in eczema severity compared to placebo,” Dr. Kress said. “Bleach may also have an anti-inflammatory effect.”
Dr. Kress disclosed relationships with Pfizer, Amgen, and Sanofi/Regeneron. SDEF and this news organization are owned by Frontline Medical Communications.
including allergic rhinitis and asthma, according to Douglas W. Kress, MD, of the department of dermatology at the University of Pittsburgh.
Recent studies suggest that atopic dermatitis (AD) affects 10%-17% of the U.S. population, and 80%-90% of patients are diagnosed by the age of 5 years, Dr. Kress said in a presentation at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
“There seem to be multiple pathways, which initiate and perpetuate the cutaneous inflammation of AD including exposure to allergens, irritants, and physical trauma, infection, stress, extremes in temperature and humidity,” Dr. Kress said. In addition, foods and airborne allergens may trigger AD.
Many parents may believe that certain factors are associated with AD, but most of these perceptions are not supported by evidence, said Dr. Kress, who is also chief of the division of pediatric dermatology at the Children’s Hospital of Pittsburgh. AD appears to be a disorder of T cell dysregulation dominated by Th2 lesions in acute cases and Th1 inflammation in patients with chronic lesions.
No associations have been proven between the development of AD in the first 18 months of life and any maternal dietary restrictions, according to a recent Cochrane review, nor is there evidence for an association between AD and the introduction of solid foods, exposure to fish oil, or exposure to animals at a young age, he said. In addition, a study published in 2016 showed a lack of evidence to support the use of specific allergen immunotherapy for AD.
However, evidence does support an association between the presence of AD in children and certain other conditions, Dr. Kress said. “Other associations include an increased incidence of alopecia areata, a threefold increase in autism spectrum disorders, and a twofold increase in ADHD in children with atopic dermatitis.”
The only known food allergy linked to AD severity is egg whites; reducing egg white exposure has been shown to improve AD in children with both conditions, he noted.
Although many patients with AD experience annoying but relatively mild symptoms, health care providers should be alert to the potential for infections, particularly with Staphylococcus aureus, and remember that an active egg white allergy has been associated with staphylococcal superantigen sensitization, said Dr. Kress. The increased risk for S. aureus in children with AD may stem from a tendency to underuse antibiotics in AD children, which results in a delayed treatment until the infection becomes overt. In addition, the increased pH in patients with AD might promote the development of pathogenic strains of staph. However, ceramide-based moisturizers could help inhibit these strains by increasing skin acidity.
For patients who have poor AD control with standard therapy, antibiotics may be used as adjunctive therapy. “Consider bleach baths and/or staph decolonization with mupirocin, both of which led to significant improvement in eczema severity compared to placebo,” Dr. Kress said. “Bleach may also have an anti-inflammatory effect.”
Dr. Kress disclosed relationships with Pfizer, Amgen, and Sanofi/Regeneron. SDEF and this news organization are owned by Frontline Medical Communications.
including allergic rhinitis and asthma, according to Douglas W. Kress, MD, of the department of dermatology at the University of Pittsburgh.
Recent studies suggest that atopic dermatitis (AD) affects 10%-17% of the U.S. population, and 80%-90% of patients are diagnosed by the age of 5 years, Dr. Kress said in a presentation at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
“There seem to be multiple pathways, which initiate and perpetuate the cutaneous inflammation of AD including exposure to allergens, irritants, and physical trauma, infection, stress, extremes in temperature and humidity,” Dr. Kress said. In addition, foods and airborne allergens may trigger AD.
Many parents may believe that certain factors are associated with AD, but most of these perceptions are not supported by evidence, said Dr. Kress, who is also chief of the division of pediatric dermatology at the Children’s Hospital of Pittsburgh. AD appears to be a disorder of T cell dysregulation dominated by Th2 lesions in acute cases and Th1 inflammation in patients with chronic lesions.
No associations have been proven between the development of AD in the first 18 months of life and any maternal dietary restrictions, according to a recent Cochrane review, nor is there evidence for an association between AD and the introduction of solid foods, exposure to fish oil, or exposure to animals at a young age, he said. In addition, a study published in 2016 showed a lack of evidence to support the use of specific allergen immunotherapy for AD.
However, evidence does support an association between the presence of AD in children and certain other conditions, Dr. Kress said. “Other associations include an increased incidence of alopecia areata, a threefold increase in autism spectrum disorders, and a twofold increase in ADHD in children with atopic dermatitis.”
The only known food allergy linked to AD severity is egg whites; reducing egg white exposure has been shown to improve AD in children with both conditions, he noted.
Although many patients with AD experience annoying but relatively mild symptoms, health care providers should be alert to the potential for infections, particularly with Staphylococcus aureus, and remember that an active egg white allergy has been associated with staphylococcal superantigen sensitization, said Dr. Kress. The increased risk for S. aureus in children with AD may stem from a tendency to underuse antibiotics in AD children, which results in a delayed treatment until the infection becomes overt. In addition, the increased pH in patients with AD might promote the development of pathogenic strains of staph. However, ceramide-based moisturizers could help inhibit these strains by increasing skin acidity.
For patients who have poor AD control with standard therapy, antibiotics may be used as adjunctive therapy. “Consider bleach baths and/or staph decolonization with mupirocin, both of which led to significant improvement in eczema severity compared to placebo,” Dr. Kress said. “Bleach may also have an anti-inflammatory effect.”
Dr. Kress disclosed relationships with Pfizer, Amgen, and Sanofi/Regeneron. SDEF and this news organization are owned by Frontline Medical Communications.
EXPERT ANALYSIS FROM SDEF WOMEN’S & PEDIATRIC DERMATOLOGY SEMINAR
Early-onset atopic dermatitis linked to elevated risk for seasonal allergies and asthma
LAKE TAHOE, CALIF. – results from a large, retrospective cohort study demonstrated.
“The atopic march is characterized by a progression from atopic dermatitis, usually early in childhood, to subsequent development of allergic rhinitis and asthma, lead study author Joy Wan, MD, said at the annual meeting of the Society for Pediatric Dermatology. “It is thought that the skin acts as the site of primary sensitization through a defective epithelial barrier, which then allows for allergic sensitization to occur in the airways. It is estimated that 30%-60% of AD patients go on to develop asthma and/or allergic rhinitis. However, not all patients complete the so-called atopic march, and this variation in the risk of asthma and allergic rhinitis among AD patients is not very well understood. Better ways to risk stratify these patients are needed.”
One possible explanation for this variation in the risk of atopy in AD patients could be the timing of their dermatitis onset. “We know that atopic dermatitis begins in infancy, but it can start at any age,” said Dr. Wan, who is a fellow in the section of pediatric dermatology at the Children’s Hospital of Philadelphia. “There has been a distinction between early-onset versus late-onset AD. Some past studies have also suggested that there is an increased risk of asthma and allergic rhinitis in children who have early-onset AD before the age of 1 or 2. This suggests that perhaps the model of the atopic march varies between early- and late-onset AD. However, past studies have had several limitations. They’ve often had short durations of follow-up, they’ve only examined narrow ranges of age of onset for AD, and most of them have been designed to primarily evaluate other exposures and outcomes, rather than looking at the timing of AD onset itself.”
For the current study, Dr. Wan and her associates set out to examine the risk of seasonal allergies and asthma among children with AD with respect to the age of AD onset. They used data from the Pediatric Eczema Elective Registry (PEER), an ongoing, prospective U.S. cohort of more than 7,700 children with physician-confirmed AD (JAMA Dermatol. 2014 Jun;150:593-600). All registry participants had used pimecrolimus cream in the past, but children with lymphoproliferative disease were excluded from the registry, as were those with malignancy or those who required the use of systemic immunosuppression.
The researchers evaluated 3,966 subjects in PEER with at least 3 years of follow-up. The exposure of interest was age of AD onset, and they divided patients into three broad age categories: early onset (age 2 years or younger), mid onset (3-7 years), and late onset (8-17 years). Primary outcomes were prevalent seasonal allergies and asthma at the time of registry enrollment, and incident seasonal allergies and asthma during follow-up, assessed via patient surveys every 3 years.
The study population included high proportions of white and black children, and there was a slight predominance of females. The median age at PEER enrollment increased with advancing age of AD onset (5.2 years in the early-onset group vs. 8.2 years in the mid-onset group and 13.1 years in the late-onset group), while the duration of follow-up was fairly similar across the three groups (a median of about 8.3 months). Family history of AD was common across all three groups, while patients in the late-onset group tended to have better control of their AD, compared with their younger counterparts.
At baseline, the prevalence of seasonal allergies was highest among the early-onset group at 74.6%, compared with 69.9% among the mid-onset group and 70.1% among the late-onset group. After adjusting for sex, race, and age at registry enrollment, the relative risk for prevalent seasonal allergies was 9% lower in the mid-onset group (0.91) and 18% lower in the late-onset group (0.82), compared with those in the early-onset group. Next, Dr. Wan and her associates calculated the incidence of seasonal allergies among 1,054 patients who did not have allergies at baseline. The cumulative incidence was highest among the early-onset group (56.1%), followed by the mid-onset group (46.8%), and the late-onset group (30.6%). On adjusted analysis, the relative risk for seasonal allergies among patients who had no allergies at baseline was 18% lower in the mid-onset group (0.82) and 36% lower in the late-onset group (0.64), compared with those in the early-onset group.
In the analysis of asthma risk by age of AD onset, prevalence was highest among patients in the early-onset group at 51.5%, compared with 44.7% among the mid-onset age group and 43% among the late-onset age group. On adjusted analysis, the relative risk for asthma was 15% lower in the mid-onset group (0.85) and 29% lower in the late-onset group (0.71), compared with those in the early-onset group. Meanwhile, the cumulative incidence of asthma among patients without asthma at baseline was also highest in the early-onset group (39.2%), compared with 31.9% in the mid-onset group and 29.9% in the late-onset group.
On adjusted analysis, the relative risk for asthma among this subset of patients was 4% lower in the mid-onset group (0.96) and 8% lower in the late-onset group (0.92), compared with those in the early-onset group, a difference that was not statistically significant. “One possible explanation for this is that asthma tends to develop soon after AD does, and the rates of developing asthma later on, as detected by our study, are nondifferential,” Dr. Wan said. “Another possibility is that the impact of early-onset versus late-onset AD is just different for asthma than it is for seasonal allergies.”
She acknowledged certain limitations of the study, including the risk of misclassification bias and limitations in recall with self-reported data, and the fact that the findings may not be generalizable to all patients with AD.
“Future studies with longer follow-up and studies of adult-onset AD will help extend our findings,” she concluded. “Nevertheless, our findings may inform how we risk stratify patients for AD treatment or atopic march prevention efforts in the future.”
PEER is funded by a grant from Valeant Pharmaceuticals, but Valeant had no role in this study. Dr. Wan reported having no financial disclosures. The study won an award at the meeting for best research presented by a dermatology resident or fellow.
LAKE TAHOE, CALIF. – results from a large, retrospective cohort study demonstrated.
“The atopic march is characterized by a progression from atopic dermatitis, usually early in childhood, to subsequent development of allergic rhinitis and asthma, lead study author Joy Wan, MD, said at the annual meeting of the Society for Pediatric Dermatology. “It is thought that the skin acts as the site of primary sensitization through a defective epithelial barrier, which then allows for allergic sensitization to occur in the airways. It is estimated that 30%-60% of AD patients go on to develop asthma and/or allergic rhinitis. However, not all patients complete the so-called atopic march, and this variation in the risk of asthma and allergic rhinitis among AD patients is not very well understood. Better ways to risk stratify these patients are needed.”
One possible explanation for this variation in the risk of atopy in AD patients could be the timing of their dermatitis onset. “We know that atopic dermatitis begins in infancy, but it can start at any age,” said Dr. Wan, who is a fellow in the section of pediatric dermatology at the Children’s Hospital of Philadelphia. “There has been a distinction between early-onset versus late-onset AD. Some past studies have also suggested that there is an increased risk of asthma and allergic rhinitis in children who have early-onset AD before the age of 1 or 2. This suggests that perhaps the model of the atopic march varies between early- and late-onset AD. However, past studies have had several limitations. They’ve often had short durations of follow-up, they’ve only examined narrow ranges of age of onset for AD, and most of them have been designed to primarily evaluate other exposures and outcomes, rather than looking at the timing of AD onset itself.”
For the current study, Dr. Wan and her associates set out to examine the risk of seasonal allergies and asthma among children with AD with respect to the age of AD onset. They used data from the Pediatric Eczema Elective Registry (PEER), an ongoing, prospective U.S. cohort of more than 7,700 children with physician-confirmed AD (JAMA Dermatol. 2014 Jun;150:593-600). All registry participants had used pimecrolimus cream in the past, but children with lymphoproliferative disease were excluded from the registry, as were those with malignancy or those who required the use of systemic immunosuppression.
The researchers evaluated 3,966 subjects in PEER with at least 3 years of follow-up. The exposure of interest was age of AD onset, and they divided patients into three broad age categories: early onset (age 2 years or younger), mid onset (3-7 years), and late onset (8-17 years). Primary outcomes were prevalent seasonal allergies and asthma at the time of registry enrollment, and incident seasonal allergies and asthma during follow-up, assessed via patient surveys every 3 years.
The study population included high proportions of white and black children, and there was a slight predominance of females. The median age at PEER enrollment increased with advancing age of AD onset (5.2 years in the early-onset group vs. 8.2 years in the mid-onset group and 13.1 years in the late-onset group), while the duration of follow-up was fairly similar across the three groups (a median of about 8.3 months). Family history of AD was common across all three groups, while patients in the late-onset group tended to have better control of their AD, compared with their younger counterparts.
At baseline, the prevalence of seasonal allergies was highest among the early-onset group at 74.6%, compared with 69.9% among the mid-onset group and 70.1% among the late-onset group. After adjusting for sex, race, and age at registry enrollment, the relative risk for prevalent seasonal allergies was 9% lower in the mid-onset group (0.91) and 18% lower in the late-onset group (0.82), compared with those in the early-onset group. Next, Dr. Wan and her associates calculated the incidence of seasonal allergies among 1,054 patients who did not have allergies at baseline. The cumulative incidence was highest among the early-onset group (56.1%), followed by the mid-onset group (46.8%), and the late-onset group (30.6%). On adjusted analysis, the relative risk for seasonal allergies among patients who had no allergies at baseline was 18% lower in the mid-onset group (0.82) and 36% lower in the late-onset group (0.64), compared with those in the early-onset group.
In the analysis of asthma risk by age of AD onset, prevalence was highest among patients in the early-onset group at 51.5%, compared with 44.7% among the mid-onset age group and 43% among the late-onset age group. On adjusted analysis, the relative risk for asthma was 15% lower in the mid-onset group (0.85) and 29% lower in the late-onset group (0.71), compared with those in the early-onset group. Meanwhile, the cumulative incidence of asthma among patients without asthma at baseline was also highest in the early-onset group (39.2%), compared with 31.9% in the mid-onset group and 29.9% in the late-onset group.
On adjusted analysis, the relative risk for asthma among this subset of patients was 4% lower in the mid-onset group (0.96) and 8% lower in the late-onset group (0.92), compared with those in the early-onset group, a difference that was not statistically significant. “One possible explanation for this is that asthma tends to develop soon after AD does, and the rates of developing asthma later on, as detected by our study, are nondifferential,” Dr. Wan said. “Another possibility is that the impact of early-onset versus late-onset AD is just different for asthma than it is for seasonal allergies.”
She acknowledged certain limitations of the study, including the risk of misclassification bias and limitations in recall with self-reported data, and the fact that the findings may not be generalizable to all patients with AD.
“Future studies with longer follow-up and studies of adult-onset AD will help extend our findings,” she concluded. “Nevertheless, our findings may inform how we risk stratify patients for AD treatment or atopic march prevention efforts in the future.”
PEER is funded by a grant from Valeant Pharmaceuticals, but Valeant had no role in this study. Dr. Wan reported having no financial disclosures. The study won an award at the meeting for best research presented by a dermatology resident or fellow.
LAKE TAHOE, CALIF. – results from a large, retrospective cohort study demonstrated.
“The atopic march is characterized by a progression from atopic dermatitis, usually early in childhood, to subsequent development of allergic rhinitis and asthma, lead study author Joy Wan, MD, said at the annual meeting of the Society for Pediatric Dermatology. “It is thought that the skin acts as the site of primary sensitization through a defective epithelial barrier, which then allows for allergic sensitization to occur in the airways. It is estimated that 30%-60% of AD patients go on to develop asthma and/or allergic rhinitis. However, not all patients complete the so-called atopic march, and this variation in the risk of asthma and allergic rhinitis among AD patients is not very well understood. Better ways to risk stratify these patients are needed.”
One possible explanation for this variation in the risk of atopy in AD patients could be the timing of their dermatitis onset. “We know that atopic dermatitis begins in infancy, but it can start at any age,” said Dr. Wan, who is a fellow in the section of pediatric dermatology at the Children’s Hospital of Philadelphia. “There has been a distinction between early-onset versus late-onset AD. Some past studies have also suggested that there is an increased risk of asthma and allergic rhinitis in children who have early-onset AD before the age of 1 or 2. This suggests that perhaps the model of the atopic march varies between early- and late-onset AD. However, past studies have had several limitations. They’ve often had short durations of follow-up, they’ve only examined narrow ranges of age of onset for AD, and most of them have been designed to primarily evaluate other exposures and outcomes, rather than looking at the timing of AD onset itself.”
For the current study, Dr. Wan and her associates set out to examine the risk of seasonal allergies and asthma among children with AD with respect to the age of AD onset. They used data from the Pediatric Eczema Elective Registry (PEER), an ongoing, prospective U.S. cohort of more than 7,700 children with physician-confirmed AD (JAMA Dermatol. 2014 Jun;150:593-600). All registry participants had used pimecrolimus cream in the past, but children with lymphoproliferative disease were excluded from the registry, as were those with malignancy or those who required the use of systemic immunosuppression.
The researchers evaluated 3,966 subjects in PEER with at least 3 years of follow-up. The exposure of interest was age of AD onset, and they divided patients into three broad age categories: early onset (age 2 years or younger), mid onset (3-7 years), and late onset (8-17 years). Primary outcomes were prevalent seasonal allergies and asthma at the time of registry enrollment, and incident seasonal allergies and asthma during follow-up, assessed via patient surveys every 3 years.
The study population included high proportions of white and black children, and there was a slight predominance of females. The median age at PEER enrollment increased with advancing age of AD onset (5.2 years in the early-onset group vs. 8.2 years in the mid-onset group and 13.1 years in the late-onset group), while the duration of follow-up was fairly similar across the three groups (a median of about 8.3 months). Family history of AD was common across all three groups, while patients in the late-onset group tended to have better control of their AD, compared with their younger counterparts.
At baseline, the prevalence of seasonal allergies was highest among the early-onset group at 74.6%, compared with 69.9% among the mid-onset group and 70.1% among the late-onset group. After adjusting for sex, race, and age at registry enrollment, the relative risk for prevalent seasonal allergies was 9% lower in the mid-onset group (0.91) and 18% lower in the late-onset group (0.82), compared with those in the early-onset group. Next, Dr. Wan and her associates calculated the incidence of seasonal allergies among 1,054 patients who did not have allergies at baseline. The cumulative incidence was highest among the early-onset group (56.1%), followed by the mid-onset group (46.8%), and the late-onset group (30.6%). On adjusted analysis, the relative risk for seasonal allergies among patients who had no allergies at baseline was 18% lower in the mid-onset group (0.82) and 36% lower in the late-onset group (0.64), compared with those in the early-onset group.
In the analysis of asthma risk by age of AD onset, prevalence was highest among patients in the early-onset group at 51.5%, compared with 44.7% among the mid-onset age group and 43% among the late-onset age group. On adjusted analysis, the relative risk for asthma was 15% lower in the mid-onset group (0.85) and 29% lower in the late-onset group (0.71), compared with those in the early-onset group. Meanwhile, the cumulative incidence of asthma among patients without asthma at baseline was also highest in the early-onset group (39.2%), compared with 31.9% in the mid-onset group and 29.9% in the late-onset group.
On adjusted analysis, the relative risk for asthma among this subset of patients was 4% lower in the mid-onset group (0.96) and 8% lower in the late-onset group (0.92), compared with those in the early-onset group, a difference that was not statistically significant. “One possible explanation for this is that asthma tends to develop soon after AD does, and the rates of developing asthma later on, as detected by our study, are nondifferential,” Dr. Wan said. “Another possibility is that the impact of early-onset versus late-onset AD is just different for asthma than it is for seasonal allergies.”
She acknowledged certain limitations of the study, including the risk of misclassification bias and limitations in recall with self-reported data, and the fact that the findings may not be generalizable to all patients with AD.
“Future studies with longer follow-up and studies of adult-onset AD will help extend our findings,” she concluded. “Nevertheless, our findings may inform how we risk stratify patients for AD treatment or atopic march prevention efforts in the future.”
PEER is funded by a grant from Valeant Pharmaceuticals, but Valeant had no role in this study. Dr. Wan reported having no financial disclosures. The study won an award at the meeting for best research presented by a dermatology resident or fellow.
AT SPD 2018