User login
Top 10 treatments for vitiligo
PARK CITY, UTAH – At the annual meeting of the Pacific Dermatologic Association, Dr. Sancy A. Leachman offered a top 10 list of new agents and technologies for the treatment of vitiligo.
No. 10: Ultraviolet A1 (UVA1) phototherapy
Dr. Harvey Lui at the University of British Columbia in Vancouver is leading a phase II trial to evaluate the potential for UVA1 to induce repigmentation within vitiligo patches and to assess the side effect profile of the treatment. “I think it might work,” said Dr. Leachman, professor and chair of dermatology at Oregon Health & Science University (OHSU), Portland.
No. 9: Ginkgo biloba
The use of ginko biloba 40-60 mg 2-3 times per day, 10 minutes before a meal, was mentioned in a Cochrane Review of vitiligo treatments published on Feb. 24, 2015. “I think I’m going to give this a try in people who have failed other treatments and see if I can get some response,” Dr. Leachman said.
No. 8: Red light
Dr. Lui is leading a randomized phase II trial of low-intensity and high-intensity red light versus no treatment for vitiligo patches. Treatments will be given twice weekly for 10 weeks, with follow-up assessments at 4, 8, and 12 weeks post treatment.
No. 7: Micrografting
A novel suction blister device known as the CelluTome epidermal harvesting system uses heat and slight vacuum pressure to harvest healthy epidermal skin tissue without damaging the donor site. Dr. Leachman characterized the technology as “semiautomating the process of suction graft transplantation.”
No. 6: The ReCell device
Manufactured by Avita Medical, this investigational autologous cell harvesting device is used after CO2 abrasion and enables clinicians to create regenerative epithelial suspension with a small sample of the patient’s skin. A phase IV trial in the Netherlands is underway to assess the efficacy and safety of autologous epidermal cell suspension grafting with the ReCell device after CO2 laser abrasion, compared with CO2 laser abrasion alone and no treatment, in patients with piebaldism and stable vitiligo.
No. 5: Topical Photocil
In a pilot study sponsored by Applied Biology, researchers are enrolling patients with vitiligo to assess the safety and efficacy of Photocil. The primary outcome measure is the Vitiligo Area Severity Index (VASI). “When this cream is activated by sunlight, it degrades into narrow-band and UVB light, so you can put a topical cream on that will administer narrow-band UVB only in that spot,” said Dr. Leachman, who is also director of OHSU’s Knight Melanoma Research Program. “That’s amazing to me.”
No. 4: Afamelanotide
This is an analogue of a melanocyte-stimulating hormone. A randomized study conducted at two academic medical centers found that the combination of afamelanotide implant and narrow-band UVB phototherapy resulted in statistically superior and faster repigmentation, compared with narrow-band UVB monotherapy (JAMA Dermatol. 2015 Jan;151(1):42-50).
No. 3: Abatacept (Orencia)
This is a soluble fusion protein consisting of human cytotoxic T-lymphocyte–associated antigen 4 (CTLA4), which prevents T-cell activation. A phase I trial is underway at Brigham and Women’s Hospital in Boston to determine if weekly self-injections of the agent lead to clinical improvements of vitiligo lesions. The primary outcome measure is change in repigmentation with abatacept therapy based on the VASI score.
No. 2. Simvastatin
The notion of its use is based on STAT1 inhibition reducing interferon-gamma–dependent activation of CD8-positive T cells, according to Dr. Leachman. The concept has been successful in a mouse model, and a study in humans was recently completed by Dr. John Harris at the University of Massachusetts, Worcester. “What we have is the ability to apply an existing drug (Simvastatin) to the process and see if it works,” she said. “Wouldn’t it be cool if we could give a statin and improve vitiligo?”
No 1: Tofacitinib
This is a Janus kinase inhibitor commonly used for rheumatoid arthritis. According to Dr. Leachman, Janus kinase inhibition prevents STAT activation, “which prevents [interferon]-gamma production, which reduces activation of CD8-positive T cells via CXCL10 binding to CXCR3,” she said. A case report demonstrating its efficacy in a 53-year-old patient was recently published in JAMA Dermatology by Dr. Brett A. King and Dr. Brittany Craiglow, dermatologists at Yale School of Medicine, New Haven, Conn. “I’m hopeful that this [agent] will be made into a topical cream because these drugs do have substantial side effects,” Dr. Leachman said.
Dr. Leachman disclosed that she is a member of the medical and scientific advisory board for Myriad Genetics Laboratory. She has also participated in an advisory board meeting for Castle Biosciences and has participated in the DecisionDx registry.
PARK CITY, UTAH – At the annual meeting of the Pacific Dermatologic Association, Dr. Sancy A. Leachman offered a top 10 list of new agents and technologies for the treatment of vitiligo.
No. 10: Ultraviolet A1 (UVA1) phototherapy
Dr. Harvey Lui at the University of British Columbia in Vancouver is leading a phase II trial to evaluate the potential for UVA1 to induce repigmentation within vitiligo patches and to assess the side effect profile of the treatment. “I think it might work,” said Dr. Leachman, professor and chair of dermatology at Oregon Health & Science University (OHSU), Portland.
No. 9: Ginkgo biloba
The use of ginko biloba 40-60 mg 2-3 times per day, 10 minutes before a meal, was mentioned in a Cochrane Review of vitiligo treatments published on Feb. 24, 2015. “I think I’m going to give this a try in people who have failed other treatments and see if I can get some response,” Dr. Leachman said.
No. 8: Red light
Dr. Lui is leading a randomized phase II trial of low-intensity and high-intensity red light versus no treatment for vitiligo patches. Treatments will be given twice weekly for 10 weeks, with follow-up assessments at 4, 8, and 12 weeks post treatment.
No. 7: Micrografting
A novel suction blister device known as the CelluTome epidermal harvesting system uses heat and slight vacuum pressure to harvest healthy epidermal skin tissue without damaging the donor site. Dr. Leachman characterized the technology as “semiautomating the process of suction graft transplantation.”
No. 6: The ReCell device
Manufactured by Avita Medical, this investigational autologous cell harvesting device is used after CO2 abrasion and enables clinicians to create regenerative epithelial suspension with a small sample of the patient’s skin. A phase IV trial in the Netherlands is underway to assess the efficacy and safety of autologous epidermal cell suspension grafting with the ReCell device after CO2 laser abrasion, compared with CO2 laser abrasion alone and no treatment, in patients with piebaldism and stable vitiligo.
No. 5: Topical Photocil
In a pilot study sponsored by Applied Biology, researchers are enrolling patients with vitiligo to assess the safety and efficacy of Photocil. The primary outcome measure is the Vitiligo Area Severity Index (VASI). “When this cream is activated by sunlight, it degrades into narrow-band and UVB light, so you can put a topical cream on that will administer narrow-band UVB only in that spot,” said Dr. Leachman, who is also director of OHSU’s Knight Melanoma Research Program. “That’s amazing to me.”
No. 4: Afamelanotide
This is an analogue of a melanocyte-stimulating hormone. A randomized study conducted at two academic medical centers found that the combination of afamelanotide implant and narrow-band UVB phototherapy resulted in statistically superior and faster repigmentation, compared with narrow-band UVB monotherapy (JAMA Dermatol. 2015 Jan;151(1):42-50).
No. 3: Abatacept (Orencia)
This is a soluble fusion protein consisting of human cytotoxic T-lymphocyte–associated antigen 4 (CTLA4), which prevents T-cell activation. A phase I trial is underway at Brigham and Women’s Hospital in Boston to determine if weekly self-injections of the agent lead to clinical improvements of vitiligo lesions. The primary outcome measure is change in repigmentation with abatacept therapy based on the VASI score.
No. 2. Simvastatin
The notion of its use is based on STAT1 inhibition reducing interferon-gamma–dependent activation of CD8-positive T cells, according to Dr. Leachman. The concept has been successful in a mouse model, and a study in humans was recently completed by Dr. John Harris at the University of Massachusetts, Worcester. “What we have is the ability to apply an existing drug (Simvastatin) to the process and see if it works,” she said. “Wouldn’t it be cool if we could give a statin and improve vitiligo?”
No 1: Tofacitinib
This is a Janus kinase inhibitor commonly used for rheumatoid arthritis. According to Dr. Leachman, Janus kinase inhibition prevents STAT activation, “which prevents [interferon]-gamma production, which reduces activation of CD8-positive T cells via CXCL10 binding to CXCR3,” she said. A case report demonstrating its efficacy in a 53-year-old patient was recently published in JAMA Dermatology by Dr. Brett A. King and Dr. Brittany Craiglow, dermatologists at Yale School of Medicine, New Haven, Conn. “I’m hopeful that this [agent] will be made into a topical cream because these drugs do have substantial side effects,” Dr. Leachman said.
Dr. Leachman disclosed that she is a member of the medical and scientific advisory board for Myriad Genetics Laboratory. She has also participated in an advisory board meeting for Castle Biosciences and has participated in the DecisionDx registry.
PARK CITY, UTAH – At the annual meeting of the Pacific Dermatologic Association, Dr. Sancy A. Leachman offered a top 10 list of new agents and technologies for the treatment of vitiligo.
No. 10: Ultraviolet A1 (UVA1) phototherapy
Dr. Harvey Lui at the University of British Columbia in Vancouver is leading a phase II trial to evaluate the potential for UVA1 to induce repigmentation within vitiligo patches and to assess the side effect profile of the treatment. “I think it might work,” said Dr. Leachman, professor and chair of dermatology at Oregon Health & Science University (OHSU), Portland.
No. 9: Ginkgo biloba
The use of ginko biloba 40-60 mg 2-3 times per day, 10 minutes before a meal, was mentioned in a Cochrane Review of vitiligo treatments published on Feb. 24, 2015. “I think I’m going to give this a try in people who have failed other treatments and see if I can get some response,” Dr. Leachman said.
No. 8: Red light
Dr. Lui is leading a randomized phase II trial of low-intensity and high-intensity red light versus no treatment for vitiligo patches. Treatments will be given twice weekly for 10 weeks, with follow-up assessments at 4, 8, and 12 weeks post treatment.
No. 7: Micrografting
A novel suction blister device known as the CelluTome epidermal harvesting system uses heat and slight vacuum pressure to harvest healthy epidermal skin tissue without damaging the donor site. Dr. Leachman characterized the technology as “semiautomating the process of suction graft transplantation.”
No. 6: The ReCell device
Manufactured by Avita Medical, this investigational autologous cell harvesting device is used after CO2 abrasion and enables clinicians to create regenerative epithelial suspension with a small sample of the patient’s skin. A phase IV trial in the Netherlands is underway to assess the efficacy and safety of autologous epidermal cell suspension grafting with the ReCell device after CO2 laser abrasion, compared with CO2 laser abrasion alone and no treatment, in patients with piebaldism and stable vitiligo.
No. 5: Topical Photocil
In a pilot study sponsored by Applied Biology, researchers are enrolling patients with vitiligo to assess the safety and efficacy of Photocil. The primary outcome measure is the Vitiligo Area Severity Index (VASI). “When this cream is activated by sunlight, it degrades into narrow-band and UVB light, so you can put a topical cream on that will administer narrow-band UVB only in that spot,” said Dr. Leachman, who is also director of OHSU’s Knight Melanoma Research Program. “That’s amazing to me.”
No. 4: Afamelanotide
This is an analogue of a melanocyte-stimulating hormone. A randomized study conducted at two academic medical centers found that the combination of afamelanotide implant and narrow-band UVB phototherapy resulted in statistically superior and faster repigmentation, compared with narrow-band UVB monotherapy (JAMA Dermatol. 2015 Jan;151(1):42-50).
No. 3: Abatacept (Orencia)
This is a soluble fusion protein consisting of human cytotoxic T-lymphocyte–associated antigen 4 (CTLA4), which prevents T-cell activation. A phase I trial is underway at Brigham and Women’s Hospital in Boston to determine if weekly self-injections of the agent lead to clinical improvements of vitiligo lesions. The primary outcome measure is change in repigmentation with abatacept therapy based on the VASI score.
No. 2. Simvastatin
The notion of its use is based on STAT1 inhibition reducing interferon-gamma–dependent activation of CD8-positive T cells, according to Dr. Leachman. The concept has been successful in a mouse model, and a study in humans was recently completed by Dr. John Harris at the University of Massachusetts, Worcester. “What we have is the ability to apply an existing drug (Simvastatin) to the process and see if it works,” she said. “Wouldn’t it be cool if we could give a statin and improve vitiligo?”
No 1: Tofacitinib
This is a Janus kinase inhibitor commonly used for rheumatoid arthritis. According to Dr. Leachman, Janus kinase inhibition prevents STAT activation, “which prevents [interferon]-gamma production, which reduces activation of CD8-positive T cells via CXCL10 binding to CXCR3,” she said. A case report demonstrating its efficacy in a 53-year-old patient was recently published in JAMA Dermatology by Dr. Brett A. King and Dr. Brittany Craiglow, dermatologists at Yale School of Medicine, New Haven, Conn. “I’m hopeful that this [agent] will be made into a topical cream because these drugs do have substantial side effects,” Dr. Leachman said.
Dr. Leachman disclosed that she is a member of the medical and scientific advisory board for Myriad Genetics Laboratory. She has also participated in an advisory board meeting for Castle Biosciences and has participated in the DecisionDx registry.
EXPERT ANALYSIS AT PDA 2015
SVELT uses small amounts of fat for breast augmentation
VANCOUVER, B.C. – Serial small-volume fat transfer via liposuction is a novel, safe, and economical breast-augmentation procedure with high patient satisfaction, Dr. Barry A.S. Lycka said at the World Congress of Dermatology.
SVELT, an acronym for Serial Small-Volume Elective Lipo Transfer, offers women who are interested in breast augmentation but leery of silicone implants or other foreign materials the appealing prospect of removing unwanted fat from donor sites on their thighs, abdomen, and flanks and transferring it to their breasts, according to Dr. Lycka, a cosmetic dermatologist in private practice and at the University of Alberta, Edmonton.
He developed the office-based procedure together with Dr. Daryl K. Hoffman, a plastic surgeon at Stanford (Calif.) University.
The traditional criticisms of fat augmentation of the breasts are that it can result in fat necrosis, resorption, and/or calcification and that it makes mammographic detection of breast cancer more difficult. These complications, however, typically result from transfer of large quantities of fat, trauma inflicted upon the harvested cells, and imprecise placement of the transferred material.
SVELT sidesteps these issues by harvesting small volumes, typically 100 cc or less, under sterile tumescent anesthesia. The material is purified and then carefully injected into the dermal plane above the breast tissue. Downtime is brief. After several sessions spaced weeks apart, the result is a natural-looking and -feeling breast that is typically one cup size larger than pretreatment, according to the dermatologist.
Dr. Lycka reported on 130 patients who underwent SVELT performed by him or Dr. Hoffman. Fat resorption was minimal. Patients gave the procedure and its outcomes high marks. Complications were limited to three patients who developed small nodules that showed evidence of fat necrosis upon biopsy.
To dermatologists interested in adopting SVELT, Dr. Lycka emphasized that patient selection is critical: This is not a procedure for women who want to dramatically increase their breast size. Adequate viable fat at the donor sites is a must. Careful placement of the transferred material in the dermal plane above the breast tissue is essential. And the patient must always get a mammogram before undergoing SVELT.
VANCOUVER, B.C. – Serial small-volume fat transfer via liposuction is a novel, safe, and economical breast-augmentation procedure with high patient satisfaction, Dr. Barry A.S. Lycka said at the World Congress of Dermatology.
SVELT, an acronym for Serial Small-Volume Elective Lipo Transfer, offers women who are interested in breast augmentation but leery of silicone implants or other foreign materials the appealing prospect of removing unwanted fat from donor sites on their thighs, abdomen, and flanks and transferring it to their breasts, according to Dr. Lycka, a cosmetic dermatologist in private practice and at the University of Alberta, Edmonton.
He developed the office-based procedure together with Dr. Daryl K. Hoffman, a plastic surgeon at Stanford (Calif.) University.
The traditional criticisms of fat augmentation of the breasts are that it can result in fat necrosis, resorption, and/or calcification and that it makes mammographic detection of breast cancer more difficult. These complications, however, typically result from transfer of large quantities of fat, trauma inflicted upon the harvested cells, and imprecise placement of the transferred material.
SVELT sidesteps these issues by harvesting small volumes, typically 100 cc or less, under sterile tumescent anesthesia. The material is purified and then carefully injected into the dermal plane above the breast tissue. Downtime is brief. After several sessions spaced weeks apart, the result is a natural-looking and -feeling breast that is typically one cup size larger than pretreatment, according to the dermatologist.
Dr. Lycka reported on 130 patients who underwent SVELT performed by him or Dr. Hoffman. Fat resorption was minimal. Patients gave the procedure and its outcomes high marks. Complications were limited to three patients who developed small nodules that showed evidence of fat necrosis upon biopsy.
To dermatologists interested in adopting SVELT, Dr. Lycka emphasized that patient selection is critical: This is not a procedure for women who want to dramatically increase their breast size. Adequate viable fat at the donor sites is a must. Careful placement of the transferred material in the dermal plane above the breast tissue is essential. And the patient must always get a mammogram before undergoing SVELT.
VANCOUVER, B.C. – Serial small-volume fat transfer via liposuction is a novel, safe, and economical breast-augmentation procedure with high patient satisfaction, Dr. Barry A.S. Lycka said at the World Congress of Dermatology.
SVELT, an acronym for Serial Small-Volume Elective Lipo Transfer, offers women who are interested in breast augmentation but leery of silicone implants or other foreign materials the appealing prospect of removing unwanted fat from donor sites on their thighs, abdomen, and flanks and transferring it to their breasts, according to Dr. Lycka, a cosmetic dermatologist in private practice and at the University of Alberta, Edmonton.
He developed the office-based procedure together with Dr. Daryl K. Hoffman, a plastic surgeon at Stanford (Calif.) University.
The traditional criticisms of fat augmentation of the breasts are that it can result in fat necrosis, resorption, and/or calcification and that it makes mammographic detection of breast cancer more difficult. These complications, however, typically result from transfer of large quantities of fat, trauma inflicted upon the harvested cells, and imprecise placement of the transferred material.
SVELT sidesteps these issues by harvesting small volumes, typically 100 cc or less, under sterile tumescent anesthesia. The material is purified and then carefully injected into the dermal plane above the breast tissue. Downtime is brief. After several sessions spaced weeks apart, the result is a natural-looking and -feeling breast that is typically one cup size larger than pretreatment, according to the dermatologist.
Dr. Lycka reported on 130 patients who underwent SVELT performed by him or Dr. Hoffman. Fat resorption was minimal. Patients gave the procedure and its outcomes high marks. Complications were limited to three patients who developed small nodules that showed evidence of fat necrosis upon biopsy.
To dermatologists interested in adopting SVELT, Dr. Lycka emphasized that patient selection is critical: This is not a procedure for women who want to dramatically increase their breast size. Adequate viable fat at the donor sites is a must. Careful placement of the transferred material in the dermal plane above the breast tissue is essential. And the patient must always get a mammogram before undergoing SVELT.
EXPERT ANALYSIS FROM WCD 2015
Nail surgery: Top anesthesia tips
VANCOUVER – Achieving effective local anesthesia is the critical first step in successful nail surgery, Dr. Chris G. Adigun said at the World Congress of Dermatology.
“Always remember: Nail surgery hurts. Your patients will applaud you enthusiastically when they’re back home for your having used a long-acting anesthetic,” said Dr. Adigun, a dermatologist in group practice in Chapel Hill, N.C.
The three most widely used anesthetic agents in nail surgery are lidocaine (Xylocaine), bupivacaine (Marcaine), and ropivacaine (Naropin). Dr. Adigun said she strongly prefers ropivacaine. It combines the best features of the other two: lidocaine’s rapid onset along with a duration of action that’s even longer than bupivacaine’s, she noted. Ropivacaine’s duration of action is 8-12 hours – and it comes without bupivacaine’s potential for cardiotoxicity. Moreover, ropivacaine has a vasoconstrictive effect, which improves hemostasis and enhances visualization during the surgery.
She provided numerous additional tips on how to predictably achieve effective anesthesia for nail surgery:
• Buffer with sodium bicarbonate. The idea is to bring the anesthetic solution close to physiologic pH, which makes for a far less painful experience than injecting the acidic unbuffered solution.
• Warm it. Investigators have shown that warming anesthetic fluid reduces pain upon injection of both nonbuffered and buffered local anesthetics (Ann Emerg Med. 2011 Jul;58(1):86-98).
• Stick to a small-gauge needle. Dr. Adigan said she favors 30 gauge. It makes for a smaller, less painful puncture and limits the rate of flow of anesthetic fluid into the digital space.
• Inject in a perpendicular plane. This will disrupt fewer nerve endings than when going in at an angle.
“I think this is something that’s not frequently taught to residents in dermatology. I think we almost always go in at an angle, but if you go in at a perpendicular plane, you’re going to cause less pain,” according to Dr. Adigun.
• Inject just below the dermis. The dermis is nociceptor rich, and stretching those tissues by injecting a volume of fluid there will cause intense, continuous pain until the local anesthetic has time to take effect.
• Use distraction techniques liberally. Dr. Adigun said she likes to tell stories and jokes, which she calls “talkesthesia.” She also utilizes a battery-powered massager.
“Put the massager as close to your surgical field as you’re comfortable with. Under the gate theory of pain, you want to create as much sensory ‘noise’ as possible with your distraction techniques so that gate is filled with your sensory noise rather than pain,” the dermatologist explained.
There are three solid, time-tested completely acceptable techniques for getting the target digit numb: the wing block, the traditional digital block, and the transthecal digital block.
Dr. Adigun said she generally relies upon the wing block unless she is concerned that the associated blanching might cause her to lose her digital landmarks during surgery addressing a subtle abnormality. In that situation she turns mainly to the traditional digital block, which doesn’t interfere with digital landmarks and effectively anesthetizes both the paired digital and volar nerves.
The downside of the traditional digital block is it entails a 15- to 20-minute wait for the anesthetic to diffuse. So does the transthecal digital block, which has the additional shortcoming of achieving predictable results only when applied for surgery on the second, third, or fourth digits.
The wing block is an efficient infiltrative technique targeting the distal digit. It offers immediate anesthesia of the total nail unit. To achieve an excellent wing block, initially inject just 0.1-0.2 mL of anesthetic fluid subcutaneously into the proximal nail fold midway between the cuticle and the distal interphalangeal joint. Wait for a wheal to form; then wait an additional 45-60 seconds. At that point, inject obliquely along the lateral edge of the nail fold in the direction of the digital tip. The needle should be advanced while maintaining a gentle fluid bolus ahead of the needle tip in order to minimize the patient’s sensation of the moving needle. The process is then repeated on the opposite side of the digit.
“You want to keep that needle in the dermal plane and avoid filling the pulp with anesthetic fluid. If you do this correctly, only one prick is felt by the patient. I very rarely have to use a full cc of anesthetic fluid when I use a wing block,” Dr. Adigun said.
If any additional needle insertions are needed, make sure they’re placed into tissue that’s already been anesthetized, she added.
Dr. Adigun reported having no financial conflicts of interest.
VANCOUVER – Achieving effective local anesthesia is the critical first step in successful nail surgery, Dr. Chris G. Adigun said at the World Congress of Dermatology.
“Always remember: Nail surgery hurts. Your patients will applaud you enthusiastically when they’re back home for your having used a long-acting anesthetic,” said Dr. Adigun, a dermatologist in group practice in Chapel Hill, N.C.
The three most widely used anesthetic agents in nail surgery are lidocaine (Xylocaine), bupivacaine (Marcaine), and ropivacaine (Naropin). Dr. Adigun said she strongly prefers ropivacaine. It combines the best features of the other two: lidocaine’s rapid onset along with a duration of action that’s even longer than bupivacaine’s, she noted. Ropivacaine’s duration of action is 8-12 hours – and it comes without bupivacaine’s potential for cardiotoxicity. Moreover, ropivacaine has a vasoconstrictive effect, which improves hemostasis and enhances visualization during the surgery.
She provided numerous additional tips on how to predictably achieve effective anesthesia for nail surgery:
• Buffer with sodium bicarbonate. The idea is to bring the anesthetic solution close to physiologic pH, which makes for a far less painful experience than injecting the acidic unbuffered solution.
• Warm it. Investigators have shown that warming anesthetic fluid reduces pain upon injection of both nonbuffered and buffered local anesthetics (Ann Emerg Med. 2011 Jul;58(1):86-98).
• Stick to a small-gauge needle. Dr. Adigan said she favors 30 gauge. It makes for a smaller, less painful puncture and limits the rate of flow of anesthetic fluid into the digital space.
• Inject in a perpendicular plane. This will disrupt fewer nerve endings than when going in at an angle.
“I think this is something that’s not frequently taught to residents in dermatology. I think we almost always go in at an angle, but if you go in at a perpendicular plane, you’re going to cause less pain,” according to Dr. Adigun.
• Inject just below the dermis. The dermis is nociceptor rich, and stretching those tissues by injecting a volume of fluid there will cause intense, continuous pain until the local anesthetic has time to take effect.
• Use distraction techniques liberally. Dr. Adigun said she likes to tell stories and jokes, which she calls “talkesthesia.” She also utilizes a battery-powered massager.
“Put the massager as close to your surgical field as you’re comfortable with. Under the gate theory of pain, you want to create as much sensory ‘noise’ as possible with your distraction techniques so that gate is filled with your sensory noise rather than pain,” the dermatologist explained.
There are three solid, time-tested completely acceptable techniques for getting the target digit numb: the wing block, the traditional digital block, and the transthecal digital block.
Dr. Adigun said she generally relies upon the wing block unless she is concerned that the associated blanching might cause her to lose her digital landmarks during surgery addressing a subtle abnormality. In that situation she turns mainly to the traditional digital block, which doesn’t interfere with digital landmarks and effectively anesthetizes both the paired digital and volar nerves.
The downside of the traditional digital block is it entails a 15- to 20-minute wait for the anesthetic to diffuse. So does the transthecal digital block, which has the additional shortcoming of achieving predictable results only when applied for surgery on the second, third, or fourth digits.
The wing block is an efficient infiltrative technique targeting the distal digit. It offers immediate anesthesia of the total nail unit. To achieve an excellent wing block, initially inject just 0.1-0.2 mL of anesthetic fluid subcutaneously into the proximal nail fold midway between the cuticle and the distal interphalangeal joint. Wait for a wheal to form; then wait an additional 45-60 seconds. At that point, inject obliquely along the lateral edge of the nail fold in the direction of the digital tip. The needle should be advanced while maintaining a gentle fluid bolus ahead of the needle tip in order to minimize the patient’s sensation of the moving needle. The process is then repeated on the opposite side of the digit.
“You want to keep that needle in the dermal plane and avoid filling the pulp with anesthetic fluid. If you do this correctly, only one prick is felt by the patient. I very rarely have to use a full cc of anesthetic fluid when I use a wing block,” Dr. Adigun said.
If any additional needle insertions are needed, make sure they’re placed into tissue that’s already been anesthetized, she added.
Dr. Adigun reported having no financial conflicts of interest.
VANCOUVER – Achieving effective local anesthesia is the critical first step in successful nail surgery, Dr. Chris G. Adigun said at the World Congress of Dermatology.
“Always remember: Nail surgery hurts. Your patients will applaud you enthusiastically when they’re back home for your having used a long-acting anesthetic,” said Dr. Adigun, a dermatologist in group practice in Chapel Hill, N.C.
The three most widely used anesthetic agents in nail surgery are lidocaine (Xylocaine), bupivacaine (Marcaine), and ropivacaine (Naropin). Dr. Adigun said she strongly prefers ropivacaine. It combines the best features of the other two: lidocaine’s rapid onset along with a duration of action that’s even longer than bupivacaine’s, she noted. Ropivacaine’s duration of action is 8-12 hours – and it comes without bupivacaine’s potential for cardiotoxicity. Moreover, ropivacaine has a vasoconstrictive effect, which improves hemostasis and enhances visualization during the surgery.
She provided numerous additional tips on how to predictably achieve effective anesthesia for nail surgery:
• Buffer with sodium bicarbonate. The idea is to bring the anesthetic solution close to physiologic pH, which makes for a far less painful experience than injecting the acidic unbuffered solution.
• Warm it. Investigators have shown that warming anesthetic fluid reduces pain upon injection of both nonbuffered and buffered local anesthetics (Ann Emerg Med. 2011 Jul;58(1):86-98).
• Stick to a small-gauge needle. Dr. Adigan said she favors 30 gauge. It makes for a smaller, less painful puncture and limits the rate of flow of anesthetic fluid into the digital space.
• Inject in a perpendicular plane. This will disrupt fewer nerve endings than when going in at an angle.
“I think this is something that’s not frequently taught to residents in dermatology. I think we almost always go in at an angle, but if you go in at a perpendicular plane, you’re going to cause less pain,” according to Dr. Adigun.
• Inject just below the dermis. The dermis is nociceptor rich, and stretching those tissues by injecting a volume of fluid there will cause intense, continuous pain until the local anesthetic has time to take effect.
• Use distraction techniques liberally. Dr. Adigun said she likes to tell stories and jokes, which she calls “talkesthesia.” She also utilizes a battery-powered massager.
“Put the massager as close to your surgical field as you’re comfortable with. Under the gate theory of pain, you want to create as much sensory ‘noise’ as possible with your distraction techniques so that gate is filled with your sensory noise rather than pain,” the dermatologist explained.
There are three solid, time-tested completely acceptable techniques for getting the target digit numb: the wing block, the traditional digital block, and the transthecal digital block.
Dr. Adigun said she generally relies upon the wing block unless she is concerned that the associated blanching might cause her to lose her digital landmarks during surgery addressing a subtle abnormality. In that situation she turns mainly to the traditional digital block, which doesn’t interfere with digital landmarks and effectively anesthetizes both the paired digital and volar nerves.
The downside of the traditional digital block is it entails a 15- to 20-minute wait for the anesthetic to diffuse. So does the transthecal digital block, which has the additional shortcoming of achieving predictable results only when applied for surgery on the second, third, or fourth digits.
The wing block is an efficient infiltrative technique targeting the distal digit. It offers immediate anesthesia of the total nail unit. To achieve an excellent wing block, initially inject just 0.1-0.2 mL of anesthetic fluid subcutaneously into the proximal nail fold midway between the cuticle and the distal interphalangeal joint. Wait for a wheal to form; then wait an additional 45-60 seconds. At that point, inject obliquely along the lateral edge of the nail fold in the direction of the digital tip. The needle should be advanced while maintaining a gentle fluid bolus ahead of the needle tip in order to minimize the patient’s sensation of the moving needle. The process is then repeated on the opposite side of the digit.
“You want to keep that needle in the dermal plane and avoid filling the pulp with anesthetic fluid. If you do this correctly, only one prick is felt by the patient. I very rarely have to use a full cc of anesthetic fluid when I use a wing block,” Dr. Adigun said.
If any additional needle insertions are needed, make sure they’re placed into tissue that’s already been anesthetized, she added.
Dr. Adigun reported having no financial conflicts of interest.
EXPERT ANALYSIS FROM WCD 2015
Black tea
Camellia sinensis, an evergreen tree belonging to the Theaceae family and used by human beings for approximately 4,000 years, is the source of the beverage tea, which is popular throughout the world, especially in Asia.1 Of the four main true teas (that is, derived from the tea plant C. sinensis), green and white are unfermented, black tea is fermented, and oolong tea is semifermented.2,3
Polyphenols, many of which act as strong antioxidants, are a diverse family of thousands of chemical substances found in plants. Theaflavins are black tea polyphenols with well-documented tumor-suppressing activity.4 In fact, they are thought to be the primary constituents of black tea responsible for conferring chemoprotection against cancer.5 Black tea, through oral administration and topical application, has been shown in the laboratory setting to protect skin from UV-induced erythema, premature aging, and cancer.6
Halder et al. have found that theaflavins and thearubigins, another key class of black tea polyphenols, can suppress A431 (human epidermoid carcinoma) and A375 (human malignant melanoma) cell proliferation without adversely impacting normal human epidermal keratinocytes. The researchers concluded that theaflavins and thearubigins appear to impart chemopreventive activity via cell cycle arrest and promotion of apoptosis in human skin cancer cells through a mitochondrial death cascade.7
In a 2005 English-language literature review, Thornfeldt cited green and black tea, as well as pomegranate, as the only ingredients supported by clinical trial evidence for effectiveness in treating extrinsic aging.2
Oral administration findings in animals and humans
More than 2 decades ago, Wang et al. found that the effects of orally administered black tea were comparable to those of green tea in suppressing UVB-induced skin carcinogenesis in 7,12-dimethylbenz[a]anthracene (DMBA)-initiated SKH-1 mice.8
In 1997, Lu et al. found that orally administered black tea inhibited the proliferation of skin tumors and enhanced apoptosis in nonmalignant and malignant skin tumors in female CD-1 mice with tumors initiated by the application of DMBA and promoted with 12-O-tetradecanoylphorbol-13-acetate (TPA).9 Record et al. reported in 1998 that black tea may confer greater protection than green tea against simulated solar irradiation.10
Hakim and Harris conducted a population-based case-control study in 2001 to assess the effects of the consumption of citrus peel and black tea on squamous cell skin cancer. They found that participants who reported intake of hot black tea and citrus peel had a significant reduction in the risk of squamous cell carcinoma. Further, they concluded that hot black tea and citrus peel displayed independent potential protection against SCC.11
Two years earlier, Zhao et al. used cultured keratinocytes and mouse and human skin to evaluate the effect of both orally and topically administered standardized black tea extract and its two major polyphenolic subfractions against UVB-induced photodamage. Topical pretreatment with the extract on SKH-1 hairless mice significantly lowered the incidence and severity of erythema and diminished skinfold thickness, compared with UVB-exposed nontreated mice. The black tea extract was similarly effective in human subjects. UVB-induced inflammation in murine as well as human skin also was reduced when the standardized extract was administered 5 minutes after UVB exposure. The investigators suggested that their findings indicated that black tea extracts have the capacity to mitigate UVB-generated erythema in human and murine skin.12
In 2011, George et al. assessed the chemopreventive effects of topical resveratrol and oral black tea polyphenols in blocking skin carcinogenesis in a two-stage mouse model initiated and promoted by DMBA and TPA, respectively. The combined treatment was found to reduce tumor incidence by approximately 89% (resveratrol alone, approximately 67%; black tea polyphenols alone, approximately 75%). Tumor volume and number also were significantly diminished by the synergistic combination, which, histologically, was noted for suppressing cellular proliferation and inducing apoptosis. The investigators concluded that oral black tea polyphenols combined with topical resveratrol exert greater chemopreventive activity than either compound alone and warrant study in trials for treating skin and other cancers.13
Animal studies on topical application
In 1997, Katiyar et al. investigated the anti-inflammatory effects of topically applied black tea polyphenols, primarily theaflavin gallates and (-)-epigallocatechin-3-gallate (EGCG), against TPA-induced inflammatory responses in murine skin. Significant inhibition against TPA-promoted induction of epidermal edema, hyperplasia, leukocyte infiltration, and proinflammatory cytokine expression was rendered by the preapplication of black tea polyphenols prior to TPA exposure. The investigators concluded that black tea polyphenols may be effective against human cutaneous inflammatory responses.14
Just over a decade later, Patel et al. investigated the in vivo antitumor-promoting effects of the most plentiful polymeric black tea polyphenols (thearubigins) in mice exposed to tumor-initiating DMBA and tumor-promoting TPA over a 40-week period. Pretreatment with topical thearubigins resulted in antipromoting effects in terms of latency, multiplicity, and incidence of skin papillomas. The black tea polyphenols also were found to reduce TPA-induced cell proliferation and epidermal cell apoptosis. The researchers attributed the protective effects of these compounds to their inhibitory impact on TPA-induced cellular proliferation.15
In 2011, Choi and Kim assessed the whitening effect of black tea water extract topically applied twice daily (6 days a week for 4 weeks) to UVB-induced hyperpigmented spots on the backs of brown guinea pigs. Treatment was divided into control (UVB and saline), vehicle control (UVB, propylene glycol, ethanol, and water), positive control (UVB and 2% hydroquinone), and two experimental groups (UVB and 1% black tea; UVB and 2% black tea). The investigators observed that the hyperpigmented spots treated with hydroquinone and black tea were clearly lighter than those treated by the control or vehicle-control groups. Histologic examination revealed that melanin pigmentation, melanocyte proliferation, and melanin production were significantly diminished in the groups treated with hydroquinone and both concentrations of black tea. The authors concluded that black tea suppresses melanocyte proliferation and melanosome synthesis in vivo, thus displaying the capacity to whiten skin in brown guinea pigs.16
In 2013, Yeh et al. found in nude mouse skin in vitro that niosomes appear to be feasible as a delivery vehicle for the dermal administration of black tea extracts as a sunscreen agent.1
Topical studies in humans
Building on findings 3 years earlier18, Türkoglu et al., in 2010, assessed the photoprotective effects of dermal gels produced from green and black tea aqueous extracts tested in vivo in the forearms of six volunteers exposed to artificial UV light (200-400 nm). In addition to the green tea and black tea gels, a 0.3% caffeine gel, a carbomer gel base, and a control were tested. The investigators reported no eruptions of UV-induced erythema in any of the black and green tea gel sites, but erythema was present to varying degrees at the areas treated with caffeine gel, carbomer gel, and control. The investigators concluded that the black and green tea extracts exhibited potent UV absorbance and that the formulated gels were effective in protecting the skin against UV-induced erythema. Further, the investigators suggested that these agents have the potential to protect against other harm caused by UV radiation, including photoaging.19
Conclusion
Though not as widely investigated as green tea, the therapeutic potential of black tea is of great interest. Although an abundance of laboratory evidence has emerged, clinical evidence is sparse. Nevertheless, laboratory data suggest the potential uses of black tea in the dermatologic realm and justify more human trials.
References
1. Cancer Lett. 1997 Mar 19;114(1-2):315-7.
2. Dermatol. Surg. 2005;31(7 Pt 2):873-80.
3. Oxid Med Cell Longev. 2012:2012:560682.
4. J Environ Pathol Toxicol Oncol. 2010;29(1):55-68.
5. Mol Carcinog. 2000 Jul;28(3):148-55.
6. Am J Clin Dermatol. 2010;11(4):247-67.
7. Carcinogenesis. 2008 Jan;29(1):129-38.
8. Cancer Res. 1994 Jul 1;54(13):3428-35.
9. Carcinogenesis. 1997 Nov;18(11):2163-9.
10. Mutat Res. 1998 Nov 9;422(1):191-9.
12. Photochem Photobiol. 1999 Oct;70(4):637-44.
13. PLoS One. 2011;6(8):e23395.
14. Carcinogenesis. 1997 Oct;18(10):1911-6.
15. Cell Prolif. 2008 Jun;41(3):532-53.
16. Toxicol Res. 2011 Sep;27(3):153-60.
17. Int J Dermatol. 2013 Feb;52(2):239-45.
18. Int J Cosmet Sci. 2007 Dec;29(6):437-42.
19. Drug Discov Ther. 2010 Oct;4(5):362-7.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002) and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). She has contributed to the Cosmeceutical Critique column in Dermatology News since January 2001. Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever.
Camellia sinensis, an evergreen tree belonging to the Theaceae family and used by human beings for approximately 4,000 years, is the source of the beverage tea, which is popular throughout the world, especially in Asia.1 Of the four main true teas (that is, derived from the tea plant C. sinensis), green and white are unfermented, black tea is fermented, and oolong tea is semifermented.2,3
Polyphenols, many of which act as strong antioxidants, are a diverse family of thousands of chemical substances found in plants. Theaflavins are black tea polyphenols with well-documented tumor-suppressing activity.4 In fact, they are thought to be the primary constituents of black tea responsible for conferring chemoprotection against cancer.5 Black tea, through oral administration and topical application, has been shown in the laboratory setting to protect skin from UV-induced erythema, premature aging, and cancer.6
Halder et al. have found that theaflavins and thearubigins, another key class of black tea polyphenols, can suppress A431 (human epidermoid carcinoma) and A375 (human malignant melanoma) cell proliferation without adversely impacting normal human epidermal keratinocytes. The researchers concluded that theaflavins and thearubigins appear to impart chemopreventive activity via cell cycle arrest and promotion of apoptosis in human skin cancer cells through a mitochondrial death cascade.7
In a 2005 English-language literature review, Thornfeldt cited green and black tea, as well as pomegranate, as the only ingredients supported by clinical trial evidence for effectiveness in treating extrinsic aging.2
Oral administration findings in animals and humans
More than 2 decades ago, Wang et al. found that the effects of orally administered black tea were comparable to those of green tea in suppressing UVB-induced skin carcinogenesis in 7,12-dimethylbenz[a]anthracene (DMBA)-initiated SKH-1 mice.8
In 1997, Lu et al. found that orally administered black tea inhibited the proliferation of skin tumors and enhanced apoptosis in nonmalignant and malignant skin tumors in female CD-1 mice with tumors initiated by the application of DMBA and promoted with 12-O-tetradecanoylphorbol-13-acetate (TPA).9 Record et al. reported in 1998 that black tea may confer greater protection than green tea against simulated solar irradiation.10
Hakim and Harris conducted a population-based case-control study in 2001 to assess the effects of the consumption of citrus peel and black tea on squamous cell skin cancer. They found that participants who reported intake of hot black tea and citrus peel had a significant reduction in the risk of squamous cell carcinoma. Further, they concluded that hot black tea and citrus peel displayed independent potential protection against SCC.11
Two years earlier, Zhao et al. used cultured keratinocytes and mouse and human skin to evaluate the effect of both orally and topically administered standardized black tea extract and its two major polyphenolic subfractions against UVB-induced photodamage. Topical pretreatment with the extract on SKH-1 hairless mice significantly lowered the incidence and severity of erythema and diminished skinfold thickness, compared with UVB-exposed nontreated mice. The black tea extract was similarly effective in human subjects. UVB-induced inflammation in murine as well as human skin also was reduced when the standardized extract was administered 5 minutes after UVB exposure. The investigators suggested that their findings indicated that black tea extracts have the capacity to mitigate UVB-generated erythema in human and murine skin.12
In 2011, George et al. assessed the chemopreventive effects of topical resveratrol and oral black tea polyphenols in blocking skin carcinogenesis in a two-stage mouse model initiated and promoted by DMBA and TPA, respectively. The combined treatment was found to reduce tumor incidence by approximately 89% (resveratrol alone, approximately 67%; black tea polyphenols alone, approximately 75%). Tumor volume and number also were significantly diminished by the synergistic combination, which, histologically, was noted for suppressing cellular proliferation and inducing apoptosis. The investigators concluded that oral black tea polyphenols combined with topical resveratrol exert greater chemopreventive activity than either compound alone and warrant study in trials for treating skin and other cancers.13
Animal studies on topical application
In 1997, Katiyar et al. investigated the anti-inflammatory effects of topically applied black tea polyphenols, primarily theaflavin gallates and (-)-epigallocatechin-3-gallate (EGCG), against TPA-induced inflammatory responses in murine skin. Significant inhibition against TPA-promoted induction of epidermal edema, hyperplasia, leukocyte infiltration, and proinflammatory cytokine expression was rendered by the preapplication of black tea polyphenols prior to TPA exposure. The investigators concluded that black tea polyphenols may be effective against human cutaneous inflammatory responses.14
Just over a decade later, Patel et al. investigated the in vivo antitumor-promoting effects of the most plentiful polymeric black tea polyphenols (thearubigins) in mice exposed to tumor-initiating DMBA and tumor-promoting TPA over a 40-week period. Pretreatment with topical thearubigins resulted in antipromoting effects in terms of latency, multiplicity, and incidence of skin papillomas. The black tea polyphenols also were found to reduce TPA-induced cell proliferation and epidermal cell apoptosis. The researchers attributed the protective effects of these compounds to their inhibitory impact on TPA-induced cellular proliferation.15
In 2011, Choi and Kim assessed the whitening effect of black tea water extract topically applied twice daily (6 days a week for 4 weeks) to UVB-induced hyperpigmented spots on the backs of brown guinea pigs. Treatment was divided into control (UVB and saline), vehicle control (UVB, propylene glycol, ethanol, and water), positive control (UVB and 2% hydroquinone), and two experimental groups (UVB and 1% black tea; UVB and 2% black tea). The investigators observed that the hyperpigmented spots treated with hydroquinone and black tea were clearly lighter than those treated by the control or vehicle-control groups. Histologic examination revealed that melanin pigmentation, melanocyte proliferation, and melanin production were significantly diminished in the groups treated with hydroquinone and both concentrations of black tea. The authors concluded that black tea suppresses melanocyte proliferation and melanosome synthesis in vivo, thus displaying the capacity to whiten skin in brown guinea pigs.16
In 2013, Yeh et al. found in nude mouse skin in vitro that niosomes appear to be feasible as a delivery vehicle for the dermal administration of black tea extracts as a sunscreen agent.1
Topical studies in humans
Building on findings 3 years earlier18, Türkoglu et al., in 2010, assessed the photoprotective effects of dermal gels produced from green and black tea aqueous extracts tested in vivo in the forearms of six volunteers exposed to artificial UV light (200-400 nm). In addition to the green tea and black tea gels, a 0.3% caffeine gel, a carbomer gel base, and a control were tested. The investigators reported no eruptions of UV-induced erythema in any of the black and green tea gel sites, but erythema was present to varying degrees at the areas treated with caffeine gel, carbomer gel, and control. The investigators concluded that the black and green tea extracts exhibited potent UV absorbance and that the formulated gels were effective in protecting the skin against UV-induced erythema. Further, the investigators suggested that these agents have the potential to protect against other harm caused by UV radiation, including photoaging.19
Conclusion
Though not as widely investigated as green tea, the therapeutic potential of black tea is of great interest. Although an abundance of laboratory evidence has emerged, clinical evidence is sparse. Nevertheless, laboratory data suggest the potential uses of black tea in the dermatologic realm and justify more human trials.
References
1. Cancer Lett. 1997 Mar 19;114(1-2):315-7.
2. Dermatol. Surg. 2005;31(7 Pt 2):873-80.
3. Oxid Med Cell Longev. 2012:2012:560682.
4. J Environ Pathol Toxicol Oncol. 2010;29(1):55-68.
5. Mol Carcinog. 2000 Jul;28(3):148-55.
6. Am J Clin Dermatol. 2010;11(4):247-67.
7. Carcinogenesis. 2008 Jan;29(1):129-38.
8. Cancer Res. 1994 Jul 1;54(13):3428-35.
9. Carcinogenesis. 1997 Nov;18(11):2163-9.
10. Mutat Res. 1998 Nov 9;422(1):191-9.
12. Photochem Photobiol. 1999 Oct;70(4):637-44.
13. PLoS One. 2011;6(8):e23395.
14. Carcinogenesis. 1997 Oct;18(10):1911-6.
15. Cell Prolif. 2008 Jun;41(3):532-53.
16. Toxicol Res. 2011 Sep;27(3):153-60.
17. Int J Dermatol. 2013 Feb;52(2):239-45.
18. Int J Cosmet Sci. 2007 Dec;29(6):437-42.
19. Drug Discov Ther. 2010 Oct;4(5):362-7.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002) and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). She has contributed to the Cosmeceutical Critique column in Dermatology News since January 2001. Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever.
Camellia sinensis, an evergreen tree belonging to the Theaceae family and used by human beings for approximately 4,000 years, is the source of the beverage tea, which is popular throughout the world, especially in Asia.1 Of the four main true teas (that is, derived from the tea plant C. sinensis), green and white are unfermented, black tea is fermented, and oolong tea is semifermented.2,3
Polyphenols, many of which act as strong antioxidants, are a diverse family of thousands of chemical substances found in plants. Theaflavins are black tea polyphenols with well-documented tumor-suppressing activity.4 In fact, they are thought to be the primary constituents of black tea responsible for conferring chemoprotection against cancer.5 Black tea, through oral administration and topical application, has been shown in the laboratory setting to protect skin from UV-induced erythema, premature aging, and cancer.6
Halder et al. have found that theaflavins and thearubigins, another key class of black tea polyphenols, can suppress A431 (human epidermoid carcinoma) and A375 (human malignant melanoma) cell proliferation without adversely impacting normal human epidermal keratinocytes. The researchers concluded that theaflavins and thearubigins appear to impart chemopreventive activity via cell cycle arrest and promotion of apoptosis in human skin cancer cells through a mitochondrial death cascade.7
In a 2005 English-language literature review, Thornfeldt cited green and black tea, as well as pomegranate, as the only ingredients supported by clinical trial evidence for effectiveness in treating extrinsic aging.2
Oral administration findings in animals and humans
More than 2 decades ago, Wang et al. found that the effects of orally administered black tea were comparable to those of green tea in suppressing UVB-induced skin carcinogenesis in 7,12-dimethylbenz[a]anthracene (DMBA)-initiated SKH-1 mice.8
In 1997, Lu et al. found that orally administered black tea inhibited the proliferation of skin tumors and enhanced apoptosis in nonmalignant and malignant skin tumors in female CD-1 mice with tumors initiated by the application of DMBA and promoted with 12-O-tetradecanoylphorbol-13-acetate (TPA).9 Record et al. reported in 1998 that black tea may confer greater protection than green tea against simulated solar irradiation.10
Hakim and Harris conducted a population-based case-control study in 2001 to assess the effects of the consumption of citrus peel and black tea on squamous cell skin cancer. They found that participants who reported intake of hot black tea and citrus peel had a significant reduction in the risk of squamous cell carcinoma. Further, they concluded that hot black tea and citrus peel displayed independent potential protection against SCC.11
Two years earlier, Zhao et al. used cultured keratinocytes and mouse and human skin to evaluate the effect of both orally and topically administered standardized black tea extract and its two major polyphenolic subfractions against UVB-induced photodamage. Topical pretreatment with the extract on SKH-1 hairless mice significantly lowered the incidence and severity of erythema and diminished skinfold thickness, compared with UVB-exposed nontreated mice. The black tea extract was similarly effective in human subjects. UVB-induced inflammation in murine as well as human skin also was reduced when the standardized extract was administered 5 minutes after UVB exposure. The investigators suggested that their findings indicated that black tea extracts have the capacity to mitigate UVB-generated erythema in human and murine skin.12
In 2011, George et al. assessed the chemopreventive effects of topical resveratrol and oral black tea polyphenols in blocking skin carcinogenesis in a two-stage mouse model initiated and promoted by DMBA and TPA, respectively. The combined treatment was found to reduce tumor incidence by approximately 89% (resveratrol alone, approximately 67%; black tea polyphenols alone, approximately 75%). Tumor volume and number also were significantly diminished by the synergistic combination, which, histologically, was noted for suppressing cellular proliferation and inducing apoptosis. The investigators concluded that oral black tea polyphenols combined with topical resveratrol exert greater chemopreventive activity than either compound alone and warrant study in trials for treating skin and other cancers.13
Animal studies on topical application
In 1997, Katiyar et al. investigated the anti-inflammatory effects of topically applied black tea polyphenols, primarily theaflavin gallates and (-)-epigallocatechin-3-gallate (EGCG), against TPA-induced inflammatory responses in murine skin. Significant inhibition against TPA-promoted induction of epidermal edema, hyperplasia, leukocyte infiltration, and proinflammatory cytokine expression was rendered by the preapplication of black tea polyphenols prior to TPA exposure. The investigators concluded that black tea polyphenols may be effective against human cutaneous inflammatory responses.14
Just over a decade later, Patel et al. investigated the in vivo antitumor-promoting effects of the most plentiful polymeric black tea polyphenols (thearubigins) in mice exposed to tumor-initiating DMBA and tumor-promoting TPA over a 40-week period. Pretreatment with topical thearubigins resulted in antipromoting effects in terms of latency, multiplicity, and incidence of skin papillomas. The black tea polyphenols also were found to reduce TPA-induced cell proliferation and epidermal cell apoptosis. The researchers attributed the protective effects of these compounds to their inhibitory impact on TPA-induced cellular proliferation.15
In 2011, Choi and Kim assessed the whitening effect of black tea water extract topically applied twice daily (6 days a week for 4 weeks) to UVB-induced hyperpigmented spots on the backs of brown guinea pigs. Treatment was divided into control (UVB and saline), vehicle control (UVB, propylene glycol, ethanol, and water), positive control (UVB and 2% hydroquinone), and two experimental groups (UVB and 1% black tea; UVB and 2% black tea). The investigators observed that the hyperpigmented spots treated with hydroquinone and black tea were clearly lighter than those treated by the control or vehicle-control groups. Histologic examination revealed that melanin pigmentation, melanocyte proliferation, and melanin production were significantly diminished in the groups treated with hydroquinone and both concentrations of black tea. The authors concluded that black tea suppresses melanocyte proliferation and melanosome synthesis in vivo, thus displaying the capacity to whiten skin in brown guinea pigs.16
In 2013, Yeh et al. found in nude mouse skin in vitro that niosomes appear to be feasible as a delivery vehicle for the dermal administration of black tea extracts as a sunscreen agent.1
Topical studies in humans
Building on findings 3 years earlier18, Türkoglu et al., in 2010, assessed the photoprotective effects of dermal gels produced from green and black tea aqueous extracts tested in vivo in the forearms of six volunteers exposed to artificial UV light (200-400 nm). In addition to the green tea and black tea gels, a 0.3% caffeine gel, a carbomer gel base, and a control were tested. The investigators reported no eruptions of UV-induced erythema in any of the black and green tea gel sites, but erythema was present to varying degrees at the areas treated with caffeine gel, carbomer gel, and control. The investigators concluded that the black and green tea extracts exhibited potent UV absorbance and that the formulated gels were effective in protecting the skin against UV-induced erythema. Further, the investigators suggested that these agents have the potential to protect against other harm caused by UV radiation, including photoaging.19
Conclusion
Though not as widely investigated as green tea, the therapeutic potential of black tea is of great interest. Although an abundance of laboratory evidence has emerged, clinical evidence is sparse. Nevertheless, laboratory data suggest the potential uses of black tea in the dermatologic realm and justify more human trials.
References
1. Cancer Lett. 1997 Mar 19;114(1-2):315-7.
2. Dermatol. Surg. 2005;31(7 Pt 2):873-80.
3. Oxid Med Cell Longev. 2012:2012:560682.
4. J Environ Pathol Toxicol Oncol. 2010;29(1):55-68.
5. Mol Carcinog. 2000 Jul;28(3):148-55.
6. Am J Clin Dermatol. 2010;11(4):247-67.
7. Carcinogenesis. 2008 Jan;29(1):129-38.
8. Cancer Res. 1994 Jul 1;54(13):3428-35.
9. Carcinogenesis. 1997 Nov;18(11):2163-9.
10. Mutat Res. 1998 Nov 9;422(1):191-9.
12. Photochem Photobiol. 1999 Oct;70(4):637-44.
13. PLoS One. 2011;6(8):e23395.
14. Carcinogenesis. 1997 Oct;18(10):1911-6.
15. Cell Prolif. 2008 Jun;41(3):532-53.
16. Toxicol Res. 2011 Sep;27(3):153-60.
17. Int J Dermatol. 2013 Feb;52(2):239-45.
18. Int J Cosmet Sci. 2007 Dec;29(6):437-42.
19. Drug Discov Ther. 2010 Oct;4(5):362-7.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002) and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). She has contributed to the Cosmeceutical Critique column in Dermatology News since January 2001. Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever.
Cosmetic Corner: Dermatologists Weigh in on Products for Sensitive Skin
To improve patient care and outcomes, leading dermatologists offered their recommendations on top products for sensitive skin. Consideration must be given to:
- Aveeno Eczema Therapy Moisturizing Cream
- Cetaphil Restoraderm
- PRESCRIBEDsolutions Don’t Be So Sensitive Post-Procedure Cleanser
- Rosaliac AR Intense
- Vanicream
Cutis invites readers to send us their recommendations. Skin care products for babies, men’s shaving products, eye creams, and OTC dandruff treatments will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on top products for sensitive skin. Consideration must be given to:
- Aveeno Eczema Therapy Moisturizing Cream
- Cetaphil Restoraderm
- PRESCRIBEDsolutions Don’t Be So Sensitive Post-Procedure Cleanser
- Rosaliac AR Intense
- Vanicream
Cutis invites readers to send us their recommendations. Skin care products for babies, men’s shaving products, eye creams, and OTC dandruff treatments will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on top products for sensitive skin. Consideration must be given to:
- Aveeno Eczema Therapy Moisturizing Cream
- Cetaphil Restoraderm
- PRESCRIBEDsolutions Don’t Be So Sensitive Post-Procedure Cleanser
- Rosaliac AR Intense
- Vanicream
Cutis invites readers to send us their recommendations. Skin care products for babies, men’s shaving products, eye creams, and OTC dandruff treatments will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
High-Yield Biopsy Technique for Subepidermal Blisters
The traditional approach for confirming the diagnosis of subepidermal blistering diseases such as bullous pemphigoid (BP), epidermolysis bullosa acquisita (EBA), dermatitis herpetiformis (DH), linear IgA bullous dermatosis (LABD) requires 2 punch biopsies: one from perilesional skin for direct immunofluorescence (DIF) and one from lesional skin for light microscopy using hematoxylin and eosin (H&E) stain.1-4 These conditions are distinguished by a combination of features appreciated on H&E-stained sections, DIF, and indirect immunofluorescence for a subset of disorders. Additional information may be provided by DIF or indirect immunofluorescence utilizing the salt-split skin technique to differentiate BP (in which linear IgG deposition is identified by immunofluorescence on the roof of salt-split skin) from EBA and antiepiligrin cicatricial BP (in which linear IgG deposition is identified by immunofluorescence along the floor of the salt-split skin), which is more rare.4 One bisected punch biopsy of a subepidermal blister yields salt-split skin–like information through standard DIF and supersedes the need for the more cumbersome salt-split skin technique.
Serologic tests for the presence of circulating antibodies to BP180 and BP230 represent an emerging technology that can confirm the diagnosis of BP, but it has been difficult to identify clinically useful autoantibodies to confirm diagnoses of EBA and LABD.5-7 Serologic tests for tissue transglutaminase IgA antibodies may be useful in the diagnosis of DH.8 We present a cost-effective approach to biopsy in the diagnosis of subepidermal blistering diseases that provides the necessary diagnostic information to distinguish relevant disease processes.
Subepidermal Blistering Diseases
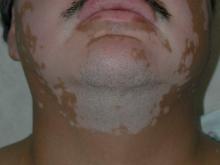
Bullous pemphigoid commonly presents with widespread tense bullae of varying sizes on an erythematous base or on otherwise normal skin.9 Some cases of BP present not with bullae but with pruritic, urticarial, plaquelike, or papular lesions. Bullous pemphigoid commonly involves flexural surfaces and the trunk but can appear anywhere on the skin. The induction of blisters by shearing with mechanical pressure on perilesional skin (Nikolsky sign) is not characteristically present in BP as it is in pemphigus vulgaris.10 Epidermolysis bullosa acquisita can mimic BP in the development of widespread tense bullae, but blisters typically appear on areas of the skin that are prone to trauma (eg, toes, knees, elbows, hands). Crusted erosions, scarring, and milia also are clinical manifestations of EBA.11 Dermatitis herpetiformis presents with grouped vesicles, papulovesicles, plaques, and excoriations that are symmetrically distributed on extensor surfaces of the skin but also can occur on the buttocks, scalp, and other areas of the skin.12,13 Although it may mimic both BP and DH, LABD frequently is less pruritic than these other conditions.14,15 Linear IgA bullous dermatosis also demonstrates the characteristic finding of multiple bullae that form concentrically around a crusted area of skin. This physical finding is known as a string of pearls. Linear IgA bullous dermatosis typically occurs in childhood and may resolve without treatment in months to years.16
Traditional Biopsy Approach
A review of several articles from the literature and multiple dermatology and dermatopathology textbooks revealed uniform recommendations for biopsy of subepidermal blistering conditions that manifest as tense blisters.1-4,9-23 A biopsy of early lesional skin or of a blister for light microscopy with H&E stain and biopsy of perilesional skin for DIF is recommended.1-4,9-23 Three review articles specifically suggested biopsy of “perilesional skin” for DIF.1-3 The majority of textbooks we reviewed also suggested that perilesional skin, or skin adjacent to a zone of erythema in the case of DH, should be sampled for DIF to assist in the diagnosis of BP, EBA, DH, and LABD.4,9-21 Biopsy of adjacent or nonlesional skin or skin around the lesion for DIF also was recommended by other textbooks for diagnosis of subepidermal blistering diseases.22,23 Perilesional skin is chosen because it is critical that the epidermis be included for adequate immunofluorescence studies.5,20 Biopsy of healed and crusted lesions should be avoided.24
Recommended Alternative Approach
A single punch biopsy produces the best possible specimen for H&E and DIF if it is obtained via one of 2 methods.
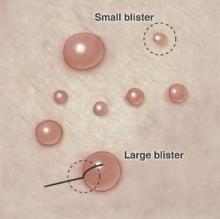
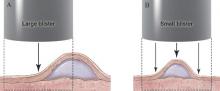
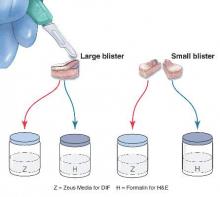
The first method involves choosing a small, 1- to 2-mm tense blister.25 Use an 8-mm punch centered on the blister that includes at least 3 mm of circumferential perilesional skin (Figures 1 and 2).20 Holding the specimen with forceps, use a no. 15 scalpel blade to bisect the blister with a sawing motion. Place half of the specimen in formalin for H&E staining and the other half in Zeus (or Michel) medium for DIF (Figure 3).
The second method is to choose any intact tense blister20,24 and, utilizing a surgical marker, draw a line from the roof of the blister onto the adjacent perilesional skin (Figure 1). After blotting with an alcohol pad so as not to remove the mark, anesthetize the site with lidocaine 1% with epinephrine,24 then take an 8-mm punch biopsy encompassing 75% perilesional skin and 25% of the blister centered on the line (Figure 2). After separating the punch specimen from the subcutaneous tissue with surgical scissors, hold the tissue with forceps and bisect the specimen with a no. 15 scalpel blade. Use a sawing motion along the line drawn in the prior steps (Figure 3). Submit half of the biopsy for H&E staining in formalin and the other half for DIF in Zeus medium.
Advantages
This approach offers several advantages. First, the technique requires only 1 invasive procedure, not 2 separate biopsies, so that the procedure can be done quickly and efficiently with the least morbidity and scarring. Secondly, because the patient is billed for 1 biopsy instead of 2, the single punch biopsy technique is more cost effective.
The bisected specimen resulting from complete excision of a small blister or from biopsy of a larger blister that includes 75% perilesional skin and 25% from the blister cavity also provides the best tissue specimen for interpretation of the subepidermal blistering processes via H&E staining.4,20,24 When traditional unmarked punch specimens of a blister margin are sent to the laboratory in formalin for H&E staining, the technician that grosses the specimen may or may not bisect the specimen showing the “take-off” point of the blister.
Finally, when the DIF specimen is prepared using either of these 2 approaches, the immunoprecipitants can be seen at the dermoepidermal junction or in the papillary dermis in the perilesional portion of the specimen.2,4 Additionally, the immunoprecipitant may be identified on the roof or floor of the blister. Although this approach has not been studied in a systematic fashion, we believe this technique provides “bonus” information to help differentiate BP and EBA correlating with salt-split skin blisters produced for indirect immunofluorescence.5,6
Limitations
It is critical for the pathologist or technician grossing these specimens to understand the techniques that are being employed and to ensure that the submitted half punch specimens are embedded so that the flat surface is cut so that the edge of the blister is properly sectioned for both H&E and DIF specimens. Additionally, with either recommended technique, if the portion of perilesional skin is not sufficient and the epidermis completely separates from the dermis, interpretation of both the H&E staining and DIF sections is substantially compromised.20 Therefore, an 8-mm disposable punch is recommended to avoid mangling the specimens when they are bisected and to insure that the epithelium is not lost. This technique is less suitable for blistering processes with a positive Nikolsky sign, such as pemphigus and toxic epidermal necrolysis, because the small area of perilesional skin adjacent to the blister may detach completely, requiring the epidermis and dermis to be evaluated separately or, in the worst-case scenario, the epidermis may be lost in processing.
Conclusion
Bisecting a single punch biopsy on subepidermal blisters provides the best specimen for H&E staining and DIF. The single punch biopsy technique also differentiates BP and EBA without utilizing salt-split skin immunofluorescence studies. This technique is more efficient and cost effective than the traditional approach of multiple biopsies on subepidermal blisters.
The traditional approach for confirming the diagnosis of subepidermal blistering diseases such as bullous pemphigoid (BP), epidermolysis bullosa acquisita (EBA), dermatitis herpetiformis (DH), linear IgA bullous dermatosis (LABD) requires 2 punch biopsies: one from perilesional skin for direct immunofluorescence (DIF) and one from lesional skin for light microscopy using hematoxylin and eosin (H&E) stain.1-4 These conditions are distinguished by a combination of features appreciated on H&E-stained sections, DIF, and indirect immunofluorescence for a subset of disorders. Additional information may be provided by DIF or indirect immunofluorescence utilizing the salt-split skin technique to differentiate BP (in which linear IgG deposition is identified by immunofluorescence on the roof of salt-split skin) from EBA and antiepiligrin cicatricial BP (in which linear IgG deposition is identified by immunofluorescence along the floor of the salt-split skin), which is more rare.4 One bisected punch biopsy of a subepidermal blister yields salt-split skin–like information through standard DIF and supersedes the need for the more cumbersome salt-split skin technique.
Serologic tests for the presence of circulating antibodies to BP180 and BP230 represent an emerging technology that can confirm the diagnosis of BP, but it has been difficult to identify clinically useful autoantibodies to confirm diagnoses of EBA and LABD.5-7 Serologic tests for tissue transglutaminase IgA antibodies may be useful in the diagnosis of DH.8 We present a cost-effective approach to biopsy in the diagnosis of subepidermal blistering diseases that provides the necessary diagnostic information to distinguish relevant disease processes.
Subepidermal Blistering Diseases
Bullous pemphigoid commonly presents with widespread tense bullae of varying sizes on an erythematous base or on otherwise normal skin.9 Some cases of BP present not with bullae but with pruritic, urticarial, plaquelike, or papular lesions. Bullous pemphigoid commonly involves flexural surfaces and the trunk but can appear anywhere on the skin. The induction of blisters by shearing with mechanical pressure on perilesional skin (Nikolsky sign) is not characteristically present in BP as it is in pemphigus vulgaris.10 Epidermolysis bullosa acquisita can mimic BP in the development of widespread tense bullae, but blisters typically appear on areas of the skin that are prone to trauma (eg, toes, knees, elbows, hands). Crusted erosions, scarring, and milia also are clinical manifestations of EBA.11 Dermatitis herpetiformis presents with grouped vesicles, papulovesicles, plaques, and excoriations that are symmetrically distributed on extensor surfaces of the skin but also can occur on the buttocks, scalp, and other areas of the skin.12,13 Although it may mimic both BP and DH, LABD frequently is less pruritic than these other conditions.14,15 Linear IgA bullous dermatosis also demonstrates the characteristic finding of multiple bullae that form concentrically around a crusted area of skin. This physical finding is known as a string of pearls. Linear IgA bullous dermatosis typically occurs in childhood and may resolve without treatment in months to years.16
Traditional Biopsy Approach
A review of several articles from the literature and multiple dermatology and dermatopathology textbooks revealed uniform recommendations for biopsy of subepidermal blistering conditions that manifest as tense blisters.1-4,9-23 A biopsy of early lesional skin or of a blister for light microscopy with H&E stain and biopsy of perilesional skin for DIF is recommended.1-4,9-23 Three review articles specifically suggested biopsy of “perilesional skin” for DIF.1-3 The majority of textbooks we reviewed also suggested that perilesional skin, or skin adjacent to a zone of erythema in the case of DH, should be sampled for DIF to assist in the diagnosis of BP, EBA, DH, and LABD.4,9-21 Biopsy of adjacent or nonlesional skin or skin around the lesion for DIF also was recommended by other textbooks for diagnosis of subepidermal blistering diseases.22,23 Perilesional skin is chosen because it is critical that the epidermis be included for adequate immunofluorescence studies.5,20 Biopsy of healed and crusted lesions should be avoided.24
Recommended Alternative Approach
A single punch biopsy produces the best possible specimen for H&E and DIF if it is obtained via one of 2 methods.
The first method involves choosing a small, 1- to 2-mm tense blister.25 Use an 8-mm punch centered on the blister that includes at least 3 mm of circumferential perilesional skin (Figures 1 and 2).20 Holding the specimen with forceps, use a no. 15 scalpel blade to bisect the blister with a sawing motion. Place half of the specimen in formalin for H&E staining and the other half in Zeus (or Michel) medium for DIF (Figure 3).
The second method is to choose any intact tense blister20,24 and, utilizing a surgical marker, draw a line from the roof of the blister onto the adjacent perilesional skin (Figure 1). After blotting with an alcohol pad so as not to remove the mark, anesthetize the site with lidocaine 1% with epinephrine,24 then take an 8-mm punch biopsy encompassing 75% perilesional skin and 25% of the blister centered on the line (Figure 2). After separating the punch specimen from the subcutaneous tissue with surgical scissors, hold the tissue with forceps and bisect the specimen with a no. 15 scalpel blade. Use a sawing motion along the line drawn in the prior steps (Figure 3). Submit half of the biopsy for H&E staining in formalin and the other half for DIF in Zeus medium.
Advantages
This approach offers several advantages. First, the technique requires only 1 invasive procedure, not 2 separate biopsies, so that the procedure can be done quickly and efficiently with the least morbidity and scarring. Secondly, because the patient is billed for 1 biopsy instead of 2, the single punch biopsy technique is more cost effective.
The bisected specimen resulting from complete excision of a small blister or from biopsy of a larger blister that includes 75% perilesional skin and 25% from the blister cavity also provides the best tissue specimen for interpretation of the subepidermal blistering processes via H&E staining.4,20,24 When traditional unmarked punch specimens of a blister margin are sent to the laboratory in formalin for H&E staining, the technician that grosses the specimen may or may not bisect the specimen showing the “take-off” point of the blister.
Finally, when the DIF specimen is prepared using either of these 2 approaches, the immunoprecipitants can be seen at the dermoepidermal junction or in the papillary dermis in the perilesional portion of the specimen.2,4 Additionally, the immunoprecipitant may be identified on the roof or floor of the blister. Although this approach has not been studied in a systematic fashion, we believe this technique provides “bonus” information to help differentiate BP and EBA correlating with salt-split skin blisters produced for indirect immunofluorescence.5,6
Limitations
It is critical for the pathologist or technician grossing these specimens to understand the techniques that are being employed and to ensure that the submitted half punch specimens are embedded so that the flat surface is cut so that the edge of the blister is properly sectioned for both H&E and DIF specimens. Additionally, with either recommended technique, if the portion of perilesional skin is not sufficient and the epidermis completely separates from the dermis, interpretation of both the H&E staining and DIF sections is substantially compromised.20 Therefore, an 8-mm disposable punch is recommended to avoid mangling the specimens when they are bisected and to insure that the epithelium is not lost. This technique is less suitable for blistering processes with a positive Nikolsky sign, such as pemphigus and toxic epidermal necrolysis, because the small area of perilesional skin adjacent to the blister may detach completely, requiring the epidermis and dermis to be evaluated separately or, in the worst-case scenario, the epidermis may be lost in processing.
Conclusion
Bisecting a single punch biopsy on subepidermal blisters provides the best specimen for H&E staining and DIF. The single punch biopsy technique also differentiates BP and EBA without utilizing salt-split skin immunofluorescence studies. This technique is more efficient and cost effective than the traditional approach of multiple biopsies on subepidermal blisters.
The traditional approach for confirming the diagnosis of subepidermal blistering diseases such as bullous pemphigoid (BP), epidermolysis bullosa acquisita (EBA), dermatitis herpetiformis (DH), linear IgA bullous dermatosis (LABD) requires 2 punch biopsies: one from perilesional skin for direct immunofluorescence (DIF) and one from lesional skin for light microscopy using hematoxylin and eosin (H&E) stain.1-4 These conditions are distinguished by a combination of features appreciated on H&E-stained sections, DIF, and indirect immunofluorescence for a subset of disorders. Additional information may be provided by DIF or indirect immunofluorescence utilizing the salt-split skin technique to differentiate BP (in which linear IgG deposition is identified by immunofluorescence on the roof of salt-split skin) from EBA and antiepiligrin cicatricial BP (in which linear IgG deposition is identified by immunofluorescence along the floor of the salt-split skin), which is more rare.4 One bisected punch biopsy of a subepidermal blister yields salt-split skin–like information through standard DIF and supersedes the need for the more cumbersome salt-split skin technique.
Serologic tests for the presence of circulating antibodies to BP180 and BP230 represent an emerging technology that can confirm the diagnosis of BP, but it has been difficult to identify clinically useful autoantibodies to confirm diagnoses of EBA and LABD.5-7 Serologic tests for tissue transglutaminase IgA antibodies may be useful in the diagnosis of DH.8 We present a cost-effective approach to biopsy in the diagnosis of subepidermal blistering diseases that provides the necessary diagnostic information to distinguish relevant disease processes.
Subepidermal Blistering Diseases
Bullous pemphigoid commonly presents with widespread tense bullae of varying sizes on an erythematous base or on otherwise normal skin.9 Some cases of BP present not with bullae but with pruritic, urticarial, plaquelike, or papular lesions. Bullous pemphigoid commonly involves flexural surfaces and the trunk but can appear anywhere on the skin. The induction of blisters by shearing with mechanical pressure on perilesional skin (Nikolsky sign) is not characteristically present in BP as it is in pemphigus vulgaris.10 Epidermolysis bullosa acquisita can mimic BP in the development of widespread tense bullae, but blisters typically appear on areas of the skin that are prone to trauma (eg, toes, knees, elbows, hands). Crusted erosions, scarring, and milia also are clinical manifestations of EBA.11 Dermatitis herpetiformis presents with grouped vesicles, papulovesicles, plaques, and excoriations that are symmetrically distributed on extensor surfaces of the skin but also can occur on the buttocks, scalp, and other areas of the skin.12,13 Although it may mimic both BP and DH, LABD frequently is less pruritic than these other conditions.14,15 Linear IgA bullous dermatosis also demonstrates the characteristic finding of multiple bullae that form concentrically around a crusted area of skin. This physical finding is known as a string of pearls. Linear IgA bullous dermatosis typically occurs in childhood and may resolve without treatment in months to years.16
Traditional Biopsy Approach
A review of several articles from the literature and multiple dermatology and dermatopathology textbooks revealed uniform recommendations for biopsy of subepidermal blistering conditions that manifest as tense blisters.1-4,9-23 A biopsy of early lesional skin or of a blister for light microscopy with H&E stain and biopsy of perilesional skin for DIF is recommended.1-4,9-23 Three review articles specifically suggested biopsy of “perilesional skin” for DIF.1-3 The majority of textbooks we reviewed also suggested that perilesional skin, or skin adjacent to a zone of erythema in the case of DH, should be sampled for DIF to assist in the diagnosis of BP, EBA, DH, and LABD.4,9-21 Biopsy of adjacent or nonlesional skin or skin around the lesion for DIF also was recommended by other textbooks for diagnosis of subepidermal blistering diseases.22,23 Perilesional skin is chosen because it is critical that the epidermis be included for adequate immunofluorescence studies.5,20 Biopsy of healed and crusted lesions should be avoided.24
Recommended Alternative Approach
A single punch biopsy produces the best possible specimen for H&E and DIF if it is obtained via one of 2 methods.
The first method involves choosing a small, 1- to 2-mm tense blister.25 Use an 8-mm punch centered on the blister that includes at least 3 mm of circumferential perilesional skin (Figures 1 and 2).20 Holding the specimen with forceps, use a no. 15 scalpel blade to bisect the blister with a sawing motion. Place half of the specimen in formalin for H&E staining and the other half in Zeus (or Michel) medium for DIF (Figure 3).
The second method is to choose any intact tense blister20,24 and, utilizing a surgical marker, draw a line from the roof of the blister onto the adjacent perilesional skin (Figure 1). After blotting with an alcohol pad so as not to remove the mark, anesthetize the site with lidocaine 1% with epinephrine,24 then take an 8-mm punch biopsy encompassing 75% perilesional skin and 25% of the blister centered on the line (Figure 2). After separating the punch specimen from the subcutaneous tissue with surgical scissors, hold the tissue with forceps and bisect the specimen with a no. 15 scalpel blade. Use a sawing motion along the line drawn in the prior steps (Figure 3). Submit half of the biopsy for H&E staining in formalin and the other half for DIF in Zeus medium.
Advantages
This approach offers several advantages. First, the technique requires only 1 invasive procedure, not 2 separate biopsies, so that the procedure can be done quickly and efficiently with the least morbidity and scarring. Secondly, because the patient is billed for 1 biopsy instead of 2, the single punch biopsy technique is more cost effective.
The bisected specimen resulting from complete excision of a small blister or from biopsy of a larger blister that includes 75% perilesional skin and 25% from the blister cavity also provides the best tissue specimen for interpretation of the subepidermal blistering processes via H&E staining.4,20,24 When traditional unmarked punch specimens of a blister margin are sent to the laboratory in formalin for H&E staining, the technician that grosses the specimen may or may not bisect the specimen showing the “take-off” point of the blister.
Finally, when the DIF specimen is prepared using either of these 2 approaches, the immunoprecipitants can be seen at the dermoepidermal junction or in the papillary dermis in the perilesional portion of the specimen.2,4 Additionally, the immunoprecipitant may be identified on the roof or floor of the blister. Although this approach has not been studied in a systematic fashion, we believe this technique provides “bonus” information to help differentiate BP and EBA correlating with salt-split skin blisters produced for indirect immunofluorescence.5,6
Limitations
It is critical for the pathologist or technician grossing these specimens to understand the techniques that are being employed and to ensure that the submitted half punch specimens are embedded so that the flat surface is cut so that the edge of the blister is properly sectioned for both H&E and DIF specimens. Additionally, with either recommended technique, if the portion of perilesional skin is not sufficient and the epidermis completely separates from the dermis, interpretation of both the H&E staining and DIF sections is substantially compromised.20 Therefore, an 8-mm disposable punch is recommended to avoid mangling the specimens when they are bisected and to insure that the epithelium is not lost. This technique is less suitable for blistering processes with a positive Nikolsky sign, such as pemphigus and toxic epidermal necrolysis, because the small area of perilesional skin adjacent to the blister may detach completely, requiring the epidermis and dermis to be evaluated separately or, in the worst-case scenario, the epidermis may be lost in processing.
Conclusion
Bisecting a single punch biopsy on subepidermal blisters provides the best specimen for H&E staining and DIF. The single punch biopsy technique also differentiates BP and EBA without utilizing salt-split skin immunofluorescence studies. This technique is more efficient and cost effective than the traditional approach of multiple biopsies on subepidermal blisters.
This is purlsCopyright text added for testing purposes.
WCD: Six new technologies will change dermatology practice
VANCOUVER – Six developments – daylight photodynamic therapy, body sculpting, better laser treatments for tattoo removal, marketing of home-use fractionated lasers for skin rejuvenation, novel lasers for vascular malformations, and light-based multiphoton microscopy as an alternative to conventional histopathology – will have significant impact upon dermatologic practice, Dr. Christopher B. Zachary predicted in a keynote address at the World Congress of Dermatology.
Savvy patients will increasingly be asking their dermatologists about them, added Dr. Zachary, professor and chair of the department of dermatology at the University of California, Irvine.
Several of these advances fall within cosmetic dermatology, a field he vigorously defended. Showing before-and-after facial photos of a middle-aged woman who appeared 10-15 years younger following skin resurfacing with an ablative laser, Dr. Zachary asserted, “You can dramatically change people’s lives with this therapy.
“When this lady’s at work, she’s surrounded by younger women who want her job. So, this therapy has social and economic benefits,” he said. “I know that some of you in the audience might think that aesthetics is sort of a Cinderella sister to mainstream dermatology, but it’s important. We’re going to live longer, hopefully, and we want to look good – because in this culture, that’s important.”
• Daylight photodynamic therapy. Developed in Scandinavia as a field therapy for photodamaged skin and precancerous skin conditions, daylight PDT delivers continuous, low-level activation of protoporphyrin IX rather than the faster activation achieved with pulsed dye laser or other artificial light sources.
The big advantage of daylight PDT is that it’s essentially pain free. In contrast, most patients find traditional PDT so unpleasant they are reluctant to return for a second session. Moreover, natural daylight is actually a better light source, in Dr. Zachary’s view, because daylight includes all the relevant absorption wavelengths, including those in the red spectrum that allow for deeper penetration into the skin.
Daylight PDT is quite popular throughout Europe, and it’s sure to catch on here, according to Dr. Zachary, who has published research confirming its safety and efficacy (Lasers Surg. Med. 2015;47:168-72). Because patients receive their daylight therapy at home, it saves time for the patient, physician, and clinic. Plus, physicians avoid the need for expensive equipment, he noted.
Dr. Zachary offered a tip based upon his daylight PDT experience: Because aminolevulinic acid (ALA) is poorly absorbed on the arms and legs, he often pretreats those target areas with a superficial fractional ablative laser to a depth of 20 microns.
“It enhances ALA uptake tremendously,” the dermatologist said.
He also finds daylight PDT to be an excellent therapy for patients with severe, drug-resistant acne. The only difference is they get 4 hours of daylight exposure ALA incubation instead of the 2.5 hours used in field therapy for photodamaged skin. The result is a robust acute PDT reaction, reliably followed by a significant reduction in inflammatory and comedonal acne lesions. Dr. Zachary typically treats at 2- to 3-week intervals and finds fewer and fewer new lesions appear between consecutive treatments.
“This is, to me, a great therapy. But I’ll tell you, these patients come in after the first 2 weeks and say, ‘I’m never going to have this procedure again, ever.’ Then 2 weeks later they call and ask, ‘When can I have my next treatment session?’ Because they actually get phenomenal results,” according to Dr. Zachary.
• Body sculpting. Physicians should expect hard scientific evidence of benefit from the device companies before investing large sums in the purchase of new noninvasive body-sculpting devices. Cryolipolysis is the leading technology in this regard, and for good reason. Careful studies have confirmed that bulk cooling of the fat can provide safe, effective, and long-lasting fat removal. The treatment is relatively painless, and the results are predictable, Dr. Zachary noted.
New studies have demonstrated encouraging outcomes with bulk heating devices, including 1,060-nm infrared laser systems.
“I will tell you that whether you cool or heat fat for a long period actually doesn’t make much difference,” he explained. “In either case, you’re inducing a delayed-onset panniculitis. There are no immediate histologic changes. The low-level panniculitic process is evident histologically at day 14 or so post treatment; with an apoptotic response and gradual fat reduction over the next 2 months.”
Other recent useful developments in body sculpting, albeit for treatment of smaller areas, include the Food and Drug Administration’s approval of ATX-101 for reduction of preplatysmal excess submental fat, or the double chin, and the use of botulinum toxin injections for facial contouring. An example of the latter is botulinum toxin injections to reduce the size of large masseter muscles in order to change the facial shape and reduce the common problem of nighttime teeth grinding known as bruxism, he continued.
• Novel laser therapies for tattoo removal. The new picosecond pulse lasers are what everyone in laser medicine is talking about. “I think this is definitely the pico epoch,” Dr. Zachary declared.
Twenty years in development, these picosecond lasers produce faster results and more complete clearing of tattoos than obtainable with Q-switched nanosecond lasers. The results are particularly impressive when addressing the traditionally challenging green and blue pigments.
Other noteworthy developments in laser tattoo removal include the so-called R20 technique developed by Dr. Dora Kossida and Dr. R. Rox Anderson of Harvard Medical School, Boston, and coworkers (J. Am. Acad. Dermatol. 2012;66:271-7) and the use of topical perfluorodecalin. The R20 technique allows for four treatment passes per session rather than just one pass. However, it’s impractically time consuming, because the passes have to be spaced 20 minutes apart.
Physicians at the Laser and Skin Surgery Center of New York have refined the R20 technique by showing that applying perfluorodecalin speeds resolution of the laser-induced immediate whitening reaction. This effectively turns the R20 technique into R0 (Lasers Surg. Med. 2013;45:76-80).
• Home-use fractionated lasers for skin rejuvenation. The skin rejuvenation lasers that dermatologists use in their offices are being miniaturized and made available for home self-treatment. Do they work?
“Some do,” according to Dr. Zachary. “If you use them frequently – once or twice a week for an entire year – they’ll probably give you the same benefit as with the traditional in-office treatments. Histologically, they cause a similar tissue injury.”
A host of handheld ultrasound devices for home-use skin rejuvenation are coming soon as well, he added.
• Laser treatment of vascular malformations. There is a growing sense among experts that the 595-nm pulsed dye laser, the traditional workhorse in treating port wine stains, is not necessarily the best tool for the job. Utilization of alternative wavelengths may be a better way to prevent lesion recurrences. The long-pulsed Nd:YAG 1,065-nm laser and 755-nm alexandrite laser appear advantageous in this regard. But so too could those wavelengths that target deoxygenated hemoglobin.
• Multiphoton microscopy. At the University of California, Irvine’s Beckman Laser Institute, Dr. Zachary and coworkers are using laser-scanning microscopy to achieve submicron-resolution three-dimensional in vivo images of skin lesions. They have found it compares favorably to standard histopathology of processed biopsy specimens for basal cell carcinoma (JAMA Dermatol. April 24, 2015 [doi:10.1001/jamadermatol.2015.0453]).
In other studies, they have compared in vivo multiphoton microscopy to standard histopathology for melanocytic nevi and melanomas. “In some ways, there is better detail than with H&E staining,” according to the dermatologist.
Will this novel in vivo imaging method replace conventional histopathology? Quite possibly, but not for many years to come, Dr. Zachary cautioned.
”I think what’s going to happen is it will take a whole generation of people to understand this. We’ll have to miniaturize these devices, which currently are very large, and put them into clinics so we can look at tens of thousands of lesions before we feel confident about replacing conventional histopathology with noninvasive imaging,” he predicted.
Dr. Zachary reported serving as a consultant to and receiving honoraria and/or equipment loans from Amway, Solta, Valeant, Cynosure, Sciton, and Zeltiq.
VANCOUVER – Six developments – daylight photodynamic therapy, body sculpting, better laser treatments for tattoo removal, marketing of home-use fractionated lasers for skin rejuvenation, novel lasers for vascular malformations, and light-based multiphoton microscopy as an alternative to conventional histopathology – will have significant impact upon dermatologic practice, Dr. Christopher B. Zachary predicted in a keynote address at the World Congress of Dermatology.
Savvy patients will increasingly be asking their dermatologists about them, added Dr. Zachary, professor and chair of the department of dermatology at the University of California, Irvine.
Several of these advances fall within cosmetic dermatology, a field he vigorously defended. Showing before-and-after facial photos of a middle-aged woman who appeared 10-15 years younger following skin resurfacing with an ablative laser, Dr. Zachary asserted, “You can dramatically change people’s lives with this therapy.
“When this lady’s at work, she’s surrounded by younger women who want her job. So, this therapy has social and economic benefits,” he said. “I know that some of you in the audience might think that aesthetics is sort of a Cinderella sister to mainstream dermatology, but it’s important. We’re going to live longer, hopefully, and we want to look good – because in this culture, that’s important.”
• Daylight photodynamic therapy. Developed in Scandinavia as a field therapy for photodamaged skin and precancerous skin conditions, daylight PDT delivers continuous, low-level activation of protoporphyrin IX rather than the faster activation achieved with pulsed dye laser or other artificial light sources.
The big advantage of daylight PDT is that it’s essentially pain free. In contrast, most patients find traditional PDT so unpleasant they are reluctant to return for a second session. Moreover, natural daylight is actually a better light source, in Dr. Zachary’s view, because daylight includes all the relevant absorption wavelengths, including those in the red spectrum that allow for deeper penetration into the skin.
Daylight PDT is quite popular throughout Europe, and it’s sure to catch on here, according to Dr. Zachary, who has published research confirming its safety and efficacy (Lasers Surg. Med. 2015;47:168-72). Because patients receive their daylight therapy at home, it saves time for the patient, physician, and clinic. Plus, physicians avoid the need for expensive equipment, he noted.
Dr. Zachary offered a tip based upon his daylight PDT experience: Because aminolevulinic acid (ALA) is poorly absorbed on the arms and legs, he often pretreats those target areas with a superficial fractional ablative laser to a depth of 20 microns.
“It enhances ALA uptake tremendously,” the dermatologist said.
He also finds daylight PDT to be an excellent therapy for patients with severe, drug-resistant acne. The only difference is they get 4 hours of daylight exposure ALA incubation instead of the 2.5 hours used in field therapy for photodamaged skin. The result is a robust acute PDT reaction, reliably followed by a significant reduction in inflammatory and comedonal acne lesions. Dr. Zachary typically treats at 2- to 3-week intervals and finds fewer and fewer new lesions appear between consecutive treatments.
“This is, to me, a great therapy. But I’ll tell you, these patients come in after the first 2 weeks and say, ‘I’m never going to have this procedure again, ever.’ Then 2 weeks later they call and ask, ‘When can I have my next treatment session?’ Because they actually get phenomenal results,” according to Dr. Zachary.
• Body sculpting. Physicians should expect hard scientific evidence of benefit from the device companies before investing large sums in the purchase of new noninvasive body-sculpting devices. Cryolipolysis is the leading technology in this regard, and for good reason. Careful studies have confirmed that bulk cooling of the fat can provide safe, effective, and long-lasting fat removal. The treatment is relatively painless, and the results are predictable, Dr. Zachary noted.
New studies have demonstrated encouraging outcomes with bulk heating devices, including 1,060-nm infrared laser systems.
“I will tell you that whether you cool or heat fat for a long period actually doesn’t make much difference,” he explained. “In either case, you’re inducing a delayed-onset panniculitis. There are no immediate histologic changes. The low-level panniculitic process is evident histologically at day 14 or so post treatment; with an apoptotic response and gradual fat reduction over the next 2 months.”
Other recent useful developments in body sculpting, albeit for treatment of smaller areas, include the Food and Drug Administration’s approval of ATX-101 for reduction of preplatysmal excess submental fat, or the double chin, and the use of botulinum toxin injections for facial contouring. An example of the latter is botulinum toxin injections to reduce the size of large masseter muscles in order to change the facial shape and reduce the common problem of nighttime teeth grinding known as bruxism, he continued.
• Novel laser therapies for tattoo removal. The new picosecond pulse lasers are what everyone in laser medicine is talking about. “I think this is definitely the pico epoch,” Dr. Zachary declared.
Twenty years in development, these picosecond lasers produce faster results and more complete clearing of tattoos than obtainable with Q-switched nanosecond lasers. The results are particularly impressive when addressing the traditionally challenging green and blue pigments.
Other noteworthy developments in laser tattoo removal include the so-called R20 technique developed by Dr. Dora Kossida and Dr. R. Rox Anderson of Harvard Medical School, Boston, and coworkers (J. Am. Acad. Dermatol. 2012;66:271-7) and the use of topical perfluorodecalin. The R20 technique allows for four treatment passes per session rather than just one pass. However, it’s impractically time consuming, because the passes have to be spaced 20 minutes apart.
Physicians at the Laser and Skin Surgery Center of New York have refined the R20 technique by showing that applying perfluorodecalin speeds resolution of the laser-induced immediate whitening reaction. This effectively turns the R20 technique into R0 (Lasers Surg. Med. 2013;45:76-80).
• Home-use fractionated lasers for skin rejuvenation. The skin rejuvenation lasers that dermatologists use in their offices are being miniaturized and made available for home self-treatment. Do they work?
“Some do,” according to Dr. Zachary. “If you use them frequently – once or twice a week for an entire year – they’ll probably give you the same benefit as with the traditional in-office treatments. Histologically, they cause a similar tissue injury.”
A host of handheld ultrasound devices for home-use skin rejuvenation are coming soon as well, he added.
• Laser treatment of vascular malformations. There is a growing sense among experts that the 595-nm pulsed dye laser, the traditional workhorse in treating port wine stains, is not necessarily the best tool for the job. Utilization of alternative wavelengths may be a better way to prevent lesion recurrences. The long-pulsed Nd:YAG 1,065-nm laser and 755-nm alexandrite laser appear advantageous in this regard. But so too could those wavelengths that target deoxygenated hemoglobin.
• Multiphoton microscopy. At the University of California, Irvine’s Beckman Laser Institute, Dr. Zachary and coworkers are using laser-scanning microscopy to achieve submicron-resolution three-dimensional in vivo images of skin lesions. They have found it compares favorably to standard histopathology of processed biopsy specimens for basal cell carcinoma (JAMA Dermatol. April 24, 2015 [doi:10.1001/jamadermatol.2015.0453]).
In other studies, they have compared in vivo multiphoton microscopy to standard histopathology for melanocytic nevi and melanomas. “In some ways, there is better detail than with H&E staining,” according to the dermatologist.
Will this novel in vivo imaging method replace conventional histopathology? Quite possibly, but not for many years to come, Dr. Zachary cautioned.
”I think what’s going to happen is it will take a whole generation of people to understand this. We’ll have to miniaturize these devices, which currently are very large, and put them into clinics so we can look at tens of thousands of lesions before we feel confident about replacing conventional histopathology with noninvasive imaging,” he predicted.
Dr. Zachary reported serving as a consultant to and receiving honoraria and/or equipment loans from Amway, Solta, Valeant, Cynosure, Sciton, and Zeltiq.
VANCOUVER – Six developments – daylight photodynamic therapy, body sculpting, better laser treatments for tattoo removal, marketing of home-use fractionated lasers for skin rejuvenation, novel lasers for vascular malformations, and light-based multiphoton microscopy as an alternative to conventional histopathology – will have significant impact upon dermatologic practice, Dr. Christopher B. Zachary predicted in a keynote address at the World Congress of Dermatology.
Savvy patients will increasingly be asking their dermatologists about them, added Dr. Zachary, professor and chair of the department of dermatology at the University of California, Irvine.
Several of these advances fall within cosmetic dermatology, a field he vigorously defended. Showing before-and-after facial photos of a middle-aged woman who appeared 10-15 years younger following skin resurfacing with an ablative laser, Dr. Zachary asserted, “You can dramatically change people’s lives with this therapy.
“When this lady’s at work, she’s surrounded by younger women who want her job. So, this therapy has social and economic benefits,” he said. “I know that some of you in the audience might think that aesthetics is sort of a Cinderella sister to mainstream dermatology, but it’s important. We’re going to live longer, hopefully, and we want to look good – because in this culture, that’s important.”
• Daylight photodynamic therapy. Developed in Scandinavia as a field therapy for photodamaged skin and precancerous skin conditions, daylight PDT delivers continuous, low-level activation of protoporphyrin IX rather than the faster activation achieved with pulsed dye laser or other artificial light sources.
The big advantage of daylight PDT is that it’s essentially pain free. In contrast, most patients find traditional PDT so unpleasant they are reluctant to return for a second session. Moreover, natural daylight is actually a better light source, in Dr. Zachary’s view, because daylight includes all the relevant absorption wavelengths, including those in the red spectrum that allow for deeper penetration into the skin.
Daylight PDT is quite popular throughout Europe, and it’s sure to catch on here, according to Dr. Zachary, who has published research confirming its safety and efficacy (Lasers Surg. Med. 2015;47:168-72). Because patients receive their daylight therapy at home, it saves time for the patient, physician, and clinic. Plus, physicians avoid the need for expensive equipment, he noted.
Dr. Zachary offered a tip based upon his daylight PDT experience: Because aminolevulinic acid (ALA) is poorly absorbed on the arms and legs, he often pretreats those target areas with a superficial fractional ablative laser to a depth of 20 microns.
“It enhances ALA uptake tremendously,” the dermatologist said.
He also finds daylight PDT to be an excellent therapy for patients with severe, drug-resistant acne. The only difference is they get 4 hours of daylight exposure ALA incubation instead of the 2.5 hours used in field therapy for photodamaged skin. The result is a robust acute PDT reaction, reliably followed by a significant reduction in inflammatory and comedonal acne lesions. Dr. Zachary typically treats at 2- to 3-week intervals and finds fewer and fewer new lesions appear between consecutive treatments.
“This is, to me, a great therapy. But I’ll tell you, these patients come in after the first 2 weeks and say, ‘I’m never going to have this procedure again, ever.’ Then 2 weeks later they call and ask, ‘When can I have my next treatment session?’ Because they actually get phenomenal results,” according to Dr. Zachary.
• Body sculpting. Physicians should expect hard scientific evidence of benefit from the device companies before investing large sums in the purchase of new noninvasive body-sculpting devices. Cryolipolysis is the leading technology in this regard, and for good reason. Careful studies have confirmed that bulk cooling of the fat can provide safe, effective, and long-lasting fat removal. The treatment is relatively painless, and the results are predictable, Dr. Zachary noted.
New studies have demonstrated encouraging outcomes with bulk heating devices, including 1,060-nm infrared laser systems.
“I will tell you that whether you cool or heat fat for a long period actually doesn’t make much difference,” he explained. “In either case, you’re inducing a delayed-onset panniculitis. There are no immediate histologic changes. The low-level panniculitic process is evident histologically at day 14 or so post treatment; with an apoptotic response and gradual fat reduction over the next 2 months.”
Other recent useful developments in body sculpting, albeit for treatment of smaller areas, include the Food and Drug Administration’s approval of ATX-101 for reduction of preplatysmal excess submental fat, or the double chin, and the use of botulinum toxin injections for facial contouring. An example of the latter is botulinum toxin injections to reduce the size of large masseter muscles in order to change the facial shape and reduce the common problem of nighttime teeth grinding known as bruxism, he continued.
• Novel laser therapies for tattoo removal. The new picosecond pulse lasers are what everyone in laser medicine is talking about. “I think this is definitely the pico epoch,” Dr. Zachary declared.
Twenty years in development, these picosecond lasers produce faster results and more complete clearing of tattoos than obtainable with Q-switched nanosecond lasers. The results are particularly impressive when addressing the traditionally challenging green and blue pigments.
Other noteworthy developments in laser tattoo removal include the so-called R20 technique developed by Dr. Dora Kossida and Dr. R. Rox Anderson of Harvard Medical School, Boston, and coworkers (J. Am. Acad. Dermatol. 2012;66:271-7) and the use of topical perfluorodecalin. The R20 technique allows for four treatment passes per session rather than just one pass. However, it’s impractically time consuming, because the passes have to be spaced 20 minutes apart.
Physicians at the Laser and Skin Surgery Center of New York have refined the R20 technique by showing that applying perfluorodecalin speeds resolution of the laser-induced immediate whitening reaction. This effectively turns the R20 technique into R0 (Lasers Surg. Med. 2013;45:76-80).
• Home-use fractionated lasers for skin rejuvenation. The skin rejuvenation lasers that dermatologists use in their offices are being miniaturized and made available for home self-treatment. Do they work?
“Some do,” according to Dr. Zachary. “If you use them frequently – once or twice a week for an entire year – they’ll probably give you the same benefit as with the traditional in-office treatments. Histologically, they cause a similar tissue injury.”
A host of handheld ultrasound devices for home-use skin rejuvenation are coming soon as well, he added.
• Laser treatment of vascular malformations. There is a growing sense among experts that the 595-nm pulsed dye laser, the traditional workhorse in treating port wine stains, is not necessarily the best tool for the job. Utilization of alternative wavelengths may be a better way to prevent lesion recurrences. The long-pulsed Nd:YAG 1,065-nm laser and 755-nm alexandrite laser appear advantageous in this regard. But so too could those wavelengths that target deoxygenated hemoglobin.
• Multiphoton microscopy. At the University of California, Irvine’s Beckman Laser Institute, Dr. Zachary and coworkers are using laser-scanning microscopy to achieve submicron-resolution three-dimensional in vivo images of skin lesions. They have found it compares favorably to standard histopathology of processed biopsy specimens for basal cell carcinoma (JAMA Dermatol. April 24, 2015 [doi:10.1001/jamadermatol.2015.0453]).
In other studies, they have compared in vivo multiphoton microscopy to standard histopathology for melanocytic nevi and melanomas. “In some ways, there is better detail than with H&E staining,” according to the dermatologist.
Will this novel in vivo imaging method replace conventional histopathology? Quite possibly, but not for many years to come, Dr. Zachary cautioned.
”I think what’s going to happen is it will take a whole generation of people to understand this. We’ll have to miniaturize these devices, which currently are very large, and put them into clinics so we can look at tens of thousands of lesions before we feel confident about replacing conventional histopathology with noninvasive imaging,” he predicted.
Dr. Zachary reported serving as a consultant to and receiving honoraria and/or equipment loans from Amway, Solta, Valeant, Cynosure, Sciton, and Zeltiq.
EXPERT ANALYSIS FROM WCD 2015
Chemical Peels
After, test your knowledge by answering the 5 practice questions.
Practice Questions
1. Which one of the following peels produces “frosting” after application?
a. citric acid
b. glycolic acid
c. mandelic acid
d. salicylic acid
e. trichloroacetic acid
2. Which one of the following peels is lipophilic?
a. citric acid
b. glycolic acid
c. mandelic acid
d. salicylic acid
e. trichloroacetic acid
3. A Jessner solution peel contains which of the following 4 components?
a. lactic acid, resorcinol, salicylic acid, ethanol
b. lactic acid, resorcinol, salicylic acid, methanol
c. lactic acid, resorcinol, salicylic acid, retinoic acid
d. retinoic acid, resorcinol, phenol, ethanol
e. retinoic acid, resorcinol, glycolic acid, methanol
4. What is the most serious risk associated with phenol peels?
a. cardiac dysrhythmia
b. hearing loss
c. scarring
d. seizure
e. tinnitus
5. Which one of the following peels self-neutralizes?
a. citric acid
b. glycolic acid
c. lactic acid
d. mandelic acid
e. salicylic acid
The answers appear on the next page.
Practice Question Answers
1. Which one of the following peels produces “frosting” after application?
a. citric acid
b. glycolic acid
c. mandelic acid
d. salicylic acid
e. trichloroacetic acid
2. Which one of the following peels is lipophilic?
a. citric acid
b. glycolic acid
c. mandelic acid
d. salicylic acid
e. trichloroacetic acid
3. A Jessner solution peel contains which of the following 4 components?
a. lactic acid, resorcinol, salicylic acid, ethanol
b. lactic acid, resorcinol, salicylic acid, methanol
c. lactic acid, resorcinol, salicylic acid, retinoic acid
d. retinoic acid, resorcinol, phenol, ethanol
e. retinoic acid, resorcinol, glycolic acid, methanol
4. What is the most serious risk associated with phenol peels?
a. cardiac dysrhythmia
b. hearing loss
c. scarring
d. seizure
e. tinnitus
5. Which one of the following peels self-neutralizes?
a. citric acid
b. glycolic acid
c. lactic acid
d. mandelic acid
e. salicylic acid
After, test your knowledge by answering the 5 practice questions.
Practice Questions
1. Which one of the following peels produces “frosting” after application?
a. citric acid
b. glycolic acid
c. mandelic acid
d. salicylic acid
e. trichloroacetic acid
2. Which one of the following peels is lipophilic?
a. citric acid
b. glycolic acid
c. mandelic acid
d. salicylic acid
e. trichloroacetic acid
3. A Jessner solution peel contains which of the following 4 components?
a. lactic acid, resorcinol, salicylic acid, ethanol
b. lactic acid, resorcinol, salicylic acid, methanol
c. lactic acid, resorcinol, salicylic acid, retinoic acid
d. retinoic acid, resorcinol, phenol, ethanol
e. retinoic acid, resorcinol, glycolic acid, methanol
4. What is the most serious risk associated with phenol peels?
a. cardiac dysrhythmia
b. hearing loss
c. scarring
d. seizure
e. tinnitus
5. Which one of the following peels self-neutralizes?
a. citric acid
b. glycolic acid
c. lactic acid
d. mandelic acid
e. salicylic acid
The answers appear on the next page.
Practice Question Answers
1. Which one of the following peels produces “frosting” after application?
a. citric acid
b. glycolic acid
c. mandelic acid
d. salicylic acid
e. trichloroacetic acid
2. Which one of the following peels is lipophilic?
a. citric acid
b. glycolic acid
c. mandelic acid
d. salicylic acid
e. trichloroacetic acid
3. A Jessner solution peel contains which of the following 4 components?
a. lactic acid, resorcinol, salicylic acid, ethanol
b. lactic acid, resorcinol, salicylic acid, methanol
c. lactic acid, resorcinol, salicylic acid, retinoic acid
d. retinoic acid, resorcinol, phenol, ethanol
e. retinoic acid, resorcinol, glycolic acid, methanol
4. What is the most serious risk associated with phenol peels?
a. cardiac dysrhythmia
b. hearing loss
c. scarring
d. seizure
e. tinnitus
5. Which one of the following peels self-neutralizes?
a. citric acid
b. glycolic acid
c. lactic acid
d. mandelic acid
e. salicylic acid
After, test your knowledge by answering the 5 practice questions.
Practice Questions
1. Which one of the following peels produces “frosting” after application?
a. citric acid
b. glycolic acid
c. mandelic acid
d. salicylic acid
e. trichloroacetic acid
2. Which one of the following peels is lipophilic?
a. citric acid
b. glycolic acid
c. mandelic acid
d. salicylic acid
e. trichloroacetic acid
3. A Jessner solution peel contains which of the following 4 components?
a. lactic acid, resorcinol, salicylic acid, ethanol
b. lactic acid, resorcinol, salicylic acid, methanol
c. lactic acid, resorcinol, salicylic acid, retinoic acid
d. retinoic acid, resorcinol, phenol, ethanol
e. retinoic acid, resorcinol, glycolic acid, methanol
4. What is the most serious risk associated with phenol peels?
a. cardiac dysrhythmia
b. hearing loss
c. scarring
d. seizure
e. tinnitus
5. Which one of the following peels self-neutralizes?
a. citric acid
b. glycolic acid
c. lactic acid
d. mandelic acid
e. salicylic acid
The answers appear on the next page.
Practice Question Answers
1. Which one of the following peels produces “frosting” after application?
a. citric acid
b. glycolic acid
c. mandelic acid
d. salicylic acid
e. trichloroacetic acid
2. Which one of the following peels is lipophilic?
a. citric acid
b. glycolic acid
c. mandelic acid
d. salicylic acid
e. trichloroacetic acid
3. A Jessner solution peel contains which of the following 4 components?
a. lactic acid, resorcinol, salicylic acid, ethanol
b. lactic acid, resorcinol, salicylic acid, methanol
c. lactic acid, resorcinol, salicylic acid, retinoic acid
d. retinoic acid, resorcinol, phenol, ethanol
e. retinoic acid, resorcinol, glycolic acid, methanol
4. What is the most serious risk associated with phenol peels?
a. cardiac dysrhythmia
b. hearing loss
c. scarring
d. seizure
e. tinnitus
5. Which one of the following peels self-neutralizes?
a. citric acid
b. glycolic acid
c. lactic acid
d. mandelic acid
e. salicylic acid
WCD: EDTA wash eliminates toluidine blue staining artifacts in Mohs surgery
VANCOUVER – Investigators at the Memorial Sloan Kettering Skin Cancer Center in Hauppauge, N.Y. have found a simple solution for a vexing problem in Mohs surgery, histology sections that don’t fully stain with toluidine blue.
The problem, it turns out, is that aluminum chloride and Monsel’s solution (ferric subsulfate) – common topical hemostatics used during the procedure – block the stain from binding tissue. Washing sections in ethylenediaminetetraacetic acid (EDTA) before staining completely eliminates the problem.
“I think this will be very useful for Mohs surgeons. It’s a very simple step, a simple modification of routine staining protocol that [eradicates] this type of artifact. When technicians collect the specimen onto the slide, they wash it with EDTA solution for about 10 seconds.” Toluidine blue staining afterward “will be perfect. There won’t be any artifact,” said senior investigator Dr. Chih-Shan Jason Chen, director of dermatologic surgery at the Hauppauge center.
It’s an important finding (and one not yet published in the medical literature, according to Dr. Chen) because staining artifacts make it impossible to read toluidine blue sections with confidence, so Mohs surgeons have to go back and take more sections, creating a deeper wound.
Over a period of the past 2 years, the EDTA wash “has made a significant difference in my practice. I never see this type of artifact anymore. I can be very comfortable taking a very thin layer of tissue and don’t need to take extra stages. We don’t need to do fancy reconstructive surgery” for the wound to heal. “This process fits with the spirit of Mohs surgery, the concept of tissue conservation,” Dr. Chen said at the World Congress of Dermatology.
Perhaps about 20% of Mohs surgeons rely heavily on toluidine blue, especially to stain for basal cell carcinomas. “The nests of cancer cells are very small, so if you have an artifact, you are going to miss them,” he noted.
The first clue that hemostatic agents were to blame was that the artifacts didn’t occur with the first Mohs cut, but only in second and subsequent sections after hemostatic agents had been applied, and especially in thinner sections more permeable to those agents. Dr. Chen tried the EDTA wash, and it worked.
His research assistant, Curtis Chen, an incoming medical student at the University of Miami, figured out the chemistry. Aluminum and iron cations from the agents bind the tissue sample, inhibiting adherence of toluidine blue. EDTA, a chelating agent, scavenges the ions, releasing the tissue for staining.
Dr. Chen and Mr. Chen had no relevant financial disclosures, and there was no outside funding for the work.
VANCOUVER – Investigators at the Memorial Sloan Kettering Skin Cancer Center in Hauppauge, N.Y. have found a simple solution for a vexing problem in Mohs surgery, histology sections that don’t fully stain with toluidine blue.
The problem, it turns out, is that aluminum chloride and Monsel’s solution (ferric subsulfate) – common topical hemostatics used during the procedure – block the stain from binding tissue. Washing sections in ethylenediaminetetraacetic acid (EDTA) before staining completely eliminates the problem.
“I think this will be very useful for Mohs surgeons. It’s a very simple step, a simple modification of routine staining protocol that [eradicates] this type of artifact. When technicians collect the specimen onto the slide, they wash it with EDTA solution for about 10 seconds.” Toluidine blue staining afterward “will be perfect. There won’t be any artifact,” said senior investigator Dr. Chih-Shan Jason Chen, director of dermatologic surgery at the Hauppauge center.
It’s an important finding (and one not yet published in the medical literature, according to Dr. Chen) because staining artifacts make it impossible to read toluidine blue sections with confidence, so Mohs surgeons have to go back and take more sections, creating a deeper wound.
Over a period of the past 2 years, the EDTA wash “has made a significant difference in my practice. I never see this type of artifact anymore. I can be very comfortable taking a very thin layer of tissue and don’t need to take extra stages. We don’t need to do fancy reconstructive surgery” for the wound to heal. “This process fits with the spirit of Mohs surgery, the concept of tissue conservation,” Dr. Chen said at the World Congress of Dermatology.
Perhaps about 20% of Mohs surgeons rely heavily on toluidine blue, especially to stain for basal cell carcinomas. “The nests of cancer cells are very small, so if you have an artifact, you are going to miss them,” he noted.
The first clue that hemostatic agents were to blame was that the artifacts didn’t occur with the first Mohs cut, but only in second and subsequent sections after hemostatic agents had been applied, and especially in thinner sections more permeable to those agents. Dr. Chen tried the EDTA wash, and it worked.
His research assistant, Curtis Chen, an incoming medical student at the University of Miami, figured out the chemistry. Aluminum and iron cations from the agents bind the tissue sample, inhibiting adherence of toluidine blue. EDTA, a chelating agent, scavenges the ions, releasing the tissue for staining.
Dr. Chen and Mr. Chen had no relevant financial disclosures, and there was no outside funding for the work.
VANCOUVER – Investigators at the Memorial Sloan Kettering Skin Cancer Center in Hauppauge, N.Y. have found a simple solution for a vexing problem in Mohs surgery, histology sections that don’t fully stain with toluidine blue.
The problem, it turns out, is that aluminum chloride and Monsel’s solution (ferric subsulfate) – common topical hemostatics used during the procedure – block the stain from binding tissue. Washing sections in ethylenediaminetetraacetic acid (EDTA) before staining completely eliminates the problem.
“I think this will be very useful for Mohs surgeons. It’s a very simple step, a simple modification of routine staining protocol that [eradicates] this type of artifact. When technicians collect the specimen onto the slide, they wash it with EDTA solution for about 10 seconds.” Toluidine blue staining afterward “will be perfect. There won’t be any artifact,” said senior investigator Dr. Chih-Shan Jason Chen, director of dermatologic surgery at the Hauppauge center.
It’s an important finding (and one not yet published in the medical literature, according to Dr. Chen) because staining artifacts make it impossible to read toluidine blue sections with confidence, so Mohs surgeons have to go back and take more sections, creating a deeper wound.
Over a period of the past 2 years, the EDTA wash “has made a significant difference in my practice. I never see this type of artifact anymore. I can be very comfortable taking a very thin layer of tissue and don’t need to take extra stages. We don’t need to do fancy reconstructive surgery” for the wound to heal. “This process fits with the spirit of Mohs surgery, the concept of tissue conservation,” Dr. Chen said at the World Congress of Dermatology.
Perhaps about 20% of Mohs surgeons rely heavily on toluidine blue, especially to stain for basal cell carcinomas. “The nests of cancer cells are very small, so if you have an artifact, you are going to miss them,” he noted.
The first clue that hemostatic agents were to blame was that the artifacts didn’t occur with the first Mohs cut, but only in second and subsequent sections after hemostatic agents had been applied, and especially in thinner sections more permeable to those agents. Dr. Chen tried the EDTA wash, and it worked.
His research assistant, Curtis Chen, an incoming medical student at the University of Miami, figured out the chemistry. Aluminum and iron cations from the agents bind the tissue sample, inhibiting adherence of toluidine blue. EDTA, a chelating agent, scavenges the ions, releasing the tissue for staining.
Dr. Chen and Mr. Chen had no relevant financial disclosures, and there was no outside funding for the work.
EXPERT ANALYSIS FROM WCD 2015
Deoxycholic acid (Kybella) for treatment of submental fullness
We are so lucky to be part of a field of medicine where advances in patient treatment options continue to occur. Having been involved in the Kybella clinical trials, it is exciting and satisfactory to see a new successful aesthetic treatment come to fruition. Kybella is the first and only Food and Drug Administration–approved injectable drug to reduce the appearance of “double chin” (submental fullness associated with submental fat) in adult patients. It is a synthetic form of naturally occurring deoxycholic acid (DCA), which lyses adipocytes when properly injected into subcutaneous fat. The safe and effective use of Kybella for the treatment of subcutaneous fat outside of the submental region has not been established and is not recommended.
The drug received unanimous support from an FDA advisory panel in March based on two placebo-controlled phase III trials involving more than 1,000 adults. In over 1,600 patients treated, 79% saw great improvement. In the studies, safety and efficacy were demonstrated with treatment of up to 50 injections of 0.2 mL each of the 1% DCA solution administered in a single treatment. Up to six treatments were administered at least 1 month apart.
Serious side effects associated with injection of DCA may include injury to the marginal mandibular nerve and dysphagia, but the most common side effects are swelling, bruising, pain, numbness, redness, and areas of hardness in the treatment area. Other potential side effects include: tingling, nodule, itching, skin tightness, headache, alopecia, and skin ulceration. In the studies, all cases of marginal mandibular nerve injury, manifesting as an asymmetric smile or facial muscle weakness, resolved spontaneously (range 1-298 days, median 44 days). Dysphagia occurred in the clinical trials as a result of administration site reactions (for example, pain, swelling, and induration in the submental area). Cases of dysphagia resolved spontaneously (range 1-81 days, median 3 days).
Caution should be taken in patients with a history of medical conditions in the neck area, difficulty swallowing, bleeding problems, or who take blood thinners. Likewise, caution should be used in patients who are or plan to become pregnant or breastfeed as Kybella has not been studied in pregnant or breastfeeding patients. Injection is contraindicated in the presence of infection at injection sites. Kybella only should be administered by a trained health care professional.
In addition to assessing whether not the patient is an ideal candidate, setting realistic expectations, and counseling about potential side effects, consultation also should include preprocedure photographs in the Frankfurt plane. Patients with moderate to severe convexity or fullness of the submental area are ideal candidates for the procedure. Those with little submental fat and excessive skin laxity may not be good candidates for this procedure and should consider a neck lift surgery as an alternative. Patients with prominent platysmal bands prior to procedure still may notice these bands after the procedure and may consider botulinum toxin injections or platysmal banding to treat these. While the active ingredient targets fat, some beneficial skin tightening may occur as a result of inflammation and fibrosis.
After photographs are taken, it is highly recommended to mark out specific anatomic landmarks on the patient, to avoid injury to the marginal mandibular nerve, salivary glands, lymph nodes, and the subhyoid region.
The procedure takes about 15-20 minutes with a short preparation time involved. Antihistamines and anti-inflammatory medications such as loratadine and ibuprofen may be given before the procedure to help reduce risk of discomfort and edema often experienced after injection. Preprocedure injection with local anesthetic also is recommended.
Once the treatment area is demarcated with a grid placed on the patient’s skin, injections of 0.2 mL of DCA are performed with a 30-gauge ½ inch needle. The product is supplied in a box with four 2-mL vials (10 mg/mL). No refrigeration is required. Once a vial is opened, it should only be used on one patient. A maximum of up to 10 mL may be injected in one patient in one session. Ice may applied after treatment. Postprocedure swelling and throbbing can be expected for several days and may rarely last up to 1 month. Patients may require two to six treatments spaced at least 1 month apart.
Dr. Wesley and Dr. Talakoub are cocontributors to a monthly Aesthetic Dermatology column in Dermatology News. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Wesley. Dr. Wesley was an investigator in the phase III Kybella clinical trials. E-mail her at dermnews@frontlinemedcom.com.
We are so lucky to be part of a field of medicine where advances in patient treatment options continue to occur. Having been involved in the Kybella clinical trials, it is exciting and satisfactory to see a new successful aesthetic treatment come to fruition. Kybella is the first and only Food and Drug Administration–approved injectable drug to reduce the appearance of “double chin” (submental fullness associated with submental fat) in adult patients. It is a synthetic form of naturally occurring deoxycholic acid (DCA), which lyses adipocytes when properly injected into subcutaneous fat. The safe and effective use of Kybella for the treatment of subcutaneous fat outside of the submental region has not been established and is not recommended.
The drug received unanimous support from an FDA advisory panel in March based on two placebo-controlled phase III trials involving more than 1,000 adults. In over 1,600 patients treated, 79% saw great improvement. In the studies, safety and efficacy were demonstrated with treatment of up to 50 injections of 0.2 mL each of the 1% DCA solution administered in a single treatment. Up to six treatments were administered at least 1 month apart.
Serious side effects associated with injection of DCA may include injury to the marginal mandibular nerve and dysphagia, but the most common side effects are swelling, bruising, pain, numbness, redness, and areas of hardness in the treatment area. Other potential side effects include: tingling, nodule, itching, skin tightness, headache, alopecia, and skin ulceration. In the studies, all cases of marginal mandibular nerve injury, manifesting as an asymmetric smile or facial muscle weakness, resolved spontaneously (range 1-298 days, median 44 days). Dysphagia occurred in the clinical trials as a result of administration site reactions (for example, pain, swelling, and induration in the submental area). Cases of dysphagia resolved spontaneously (range 1-81 days, median 3 days).
Caution should be taken in patients with a history of medical conditions in the neck area, difficulty swallowing, bleeding problems, or who take blood thinners. Likewise, caution should be used in patients who are or plan to become pregnant or breastfeed as Kybella has not been studied in pregnant or breastfeeding patients. Injection is contraindicated in the presence of infection at injection sites. Kybella only should be administered by a trained health care professional.
In addition to assessing whether not the patient is an ideal candidate, setting realistic expectations, and counseling about potential side effects, consultation also should include preprocedure photographs in the Frankfurt plane. Patients with moderate to severe convexity or fullness of the submental area are ideal candidates for the procedure. Those with little submental fat and excessive skin laxity may not be good candidates for this procedure and should consider a neck lift surgery as an alternative. Patients with prominent platysmal bands prior to procedure still may notice these bands after the procedure and may consider botulinum toxin injections or platysmal banding to treat these. While the active ingredient targets fat, some beneficial skin tightening may occur as a result of inflammation and fibrosis.
After photographs are taken, it is highly recommended to mark out specific anatomic landmarks on the patient, to avoid injury to the marginal mandibular nerve, salivary glands, lymph nodes, and the subhyoid region.
The procedure takes about 15-20 minutes with a short preparation time involved. Antihistamines and anti-inflammatory medications such as loratadine and ibuprofen may be given before the procedure to help reduce risk of discomfort and edema often experienced after injection. Preprocedure injection with local anesthetic also is recommended.
Once the treatment area is demarcated with a grid placed on the patient’s skin, injections of 0.2 mL of DCA are performed with a 30-gauge ½ inch needle. The product is supplied in a box with four 2-mL vials (10 mg/mL). No refrigeration is required. Once a vial is opened, it should only be used on one patient. A maximum of up to 10 mL may be injected in one patient in one session. Ice may applied after treatment. Postprocedure swelling and throbbing can be expected for several days and may rarely last up to 1 month. Patients may require two to six treatments spaced at least 1 month apart.
Dr. Wesley and Dr. Talakoub are cocontributors to a monthly Aesthetic Dermatology column in Dermatology News. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Wesley. Dr. Wesley was an investigator in the phase III Kybella clinical trials. E-mail her at dermnews@frontlinemedcom.com.
We are so lucky to be part of a field of medicine where advances in patient treatment options continue to occur. Having been involved in the Kybella clinical trials, it is exciting and satisfactory to see a new successful aesthetic treatment come to fruition. Kybella is the first and only Food and Drug Administration–approved injectable drug to reduce the appearance of “double chin” (submental fullness associated with submental fat) in adult patients. It is a synthetic form of naturally occurring deoxycholic acid (DCA), which lyses adipocytes when properly injected into subcutaneous fat. The safe and effective use of Kybella for the treatment of subcutaneous fat outside of the submental region has not been established and is not recommended.
The drug received unanimous support from an FDA advisory panel in March based on two placebo-controlled phase III trials involving more than 1,000 adults. In over 1,600 patients treated, 79% saw great improvement. In the studies, safety and efficacy were demonstrated with treatment of up to 50 injections of 0.2 mL each of the 1% DCA solution administered in a single treatment. Up to six treatments were administered at least 1 month apart.
Serious side effects associated with injection of DCA may include injury to the marginal mandibular nerve and dysphagia, but the most common side effects are swelling, bruising, pain, numbness, redness, and areas of hardness in the treatment area. Other potential side effects include: tingling, nodule, itching, skin tightness, headache, alopecia, and skin ulceration. In the studies, all cases of marginal mandibular nerve injury, manifesting as an asymmetric smile or facial muscle weakness, resolved spontaneously (range 1-298 days, median 44 days). Dysphagia occurred in the clinical trials as a result of administration site reactions (for example, pain, swelling, and induration in the submental area). Cases of dysphagia resolved spontaneously (range 1-81 days, median 3 days).
Caution should be taken in patients with a history of medical conditions in the neck area, difficulty swallowing, bleeding problems, or who take blood thinners. Likewise, caution should be used in patients who are or plan to become pregnant or breastfeed as Kybella has not been studied in pregnant or breastfeeding patients. Injection is contraindicated in the presence of infection at injection sites. Kybella only should be administered by a trained health care professional.
In addition to assessing whether not the patient is an ideal candidate, setting realistic expectations, and counseling about potential side effects, consultation also should include preprocedure photographs in the Frankfurt plane. Patients with moderate to severe convexity or fullness of the submental area are ideal candidates for the procedure. Those with little submental fat and excessive skin laxity may not be good candidates for this procedure and should consider a neck lift surgery as an alternative. Patients with prominent platysmal bands prior to procedure still may notice these bands after the procedure and may consider botulinum toxin injections or platysmal banding to treat these. While the active ingredient targets fat, some beneficial skin tightening may occur as a result of inflammation and fibrosis.
After photographs are taken, it is highly recommended to mark out specific anatomic landmarks on the patient, to avoid injury to the marginal mandibular nerve, salivary glands, lymph nodes, and the subhyoid region.
The procedure takes about 15-20 minutes with a short preparation time involved. Antihistamines and anti-inflammatory medications such as loratadine and ibuprofen may be given before the procedure to help reduce risk of discomfort and edema often experienced after injection. Preprocedure injection with local anesthetic also is recommended.
Once the treatment area is demarcated with a grid placed on the patient’s skin, injections of 0.2 mL of DCA are performed with a 30-gauge ½ inch needle. The product is supplied in a box with four 2-mL vials (10 mg/mL). No refrigeration is required. Once a vial is opened, it should only be used on one patient. A maximum of up to 10 mL may be injected in one patient in one session. Ice may applied after treatment. Postprocedure swelling and throbbing can be expected for several days and may rarely last up to 1 month. Patients may require two to six treatments spaced at least 1 month apart.
Dr. Wesley and Dr. Talakoub are cocontributors to a monthly Aesthetic Dermatology column in Dermatology News. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Wesley. Dr. Wesley was an investigator in the phase III Kybella clinical trials. E-mail her at dermnews@frontlinemedcom.com.