User login
Melanoma regression a positive prognostic indicator
Histologic regression in melanoma is associated with a significantly lower risk of sentinel lymph node positivity and may serve as a prognostic factor when deciding whether to perform a sentinel lymph node biopsy, according to a study published online Sept. 2 in JAMA Dermatology.
Researchers conducted a meta-analysis of 14 studies in 10,098 patients who showed histologic regression of a primary melanoma and whose sentinel lymph node status was available. In the 14 studies combined, the analysis showed patients with histologic regression had a 44% lower likelihood of having a positive sentinel lymph node than patients without regression, reported Dr. Simone Ribero of the University of Turin (Italy), and coauthors (JAMA Dermatol. 2015 Sep 2. doi: 10.1001/jamadermatol.2015.2235).
This figure was even lower for patients enrolled in high-quality studies, compared with lower-quality studies, they said. In the nine studies considered high quality, patients with regression had a 52% lower likelihood of having a positive sentinel lymph node, while those enrolled in the five studies considered low quality had a 27% lower likelihood of having a positive sentinel lymph node.
The prognostic significance of regression in primary melanoma has been confounded by the fact that disappearance of part of the tumor can result in an underestimation of the original Breslow thickness. Some previous studies have found an increase in sentinel lymph node positivity associated with regression, while other studies suggested that regression was in fact a protective factor against sentinel lymph node metastasis.
Although the studies used different definitions of histologic regression and all were observational (counted among the limitations of the studies), the authors concluded that regression was prognostically favorable. “The results of this meta-analysis may be useful when deciding to offer SLNB [sentinel lymph node biopsy] to patients with regressions of melanomas,” they wrote, adding: “It may help clinicians make a final selection of the most appropriate patients for this procedure.”
The authors had no conflicts of interest to declare.
The finding that patients with histologic regression have half the risk for micrometastases in sentinel lymph node biopsy, compared with patients without regression gives a clear answer to the question of whether patients with thin melanoma and regression should undergo SLNB, and that answer is “No.”

|
Dr. Claus Garbe |
These findings underline that fact that prognosis of patients with melanoma does not depend only on the characteristics of the tumor itself, such as tumor thickness, ulceration, and mitosis, but also on the immune response of the host.
Dr. Claus Garbe is from the Department of Dermatology, University Hospital Tübingen (Germany). These comments are taken from an accompanying editorial (JAMA Dermatol. 2015 Sep 2. doi: 10.1001/jamadermatol.2015.2235). No conflicts of interest were declared.
The finding that patients with histologic regression have half the risk for micrometastases in sentinel lymph node biopsy, compared with patients without regression gives a clear answer to the question of whether patients with thin melanoma and regression should undergo SLNB, and that answer is “No.”

|
Dr. Claus Garbe |
These findings underline that fact that prognosis of patients with melanoma does not depend only on the characteristics of the tumor itself, such as tumor thickness, ulceration, and mitosis, but also on the immune response of the host.
Dr. Claus Garbe is from the Department of Dermatology, University Hospital Tübingen (Germany). These comments are taken from an accompanying editorial (JAMA Dermatol. 2015 Sep 2. doi: 10.1001/jamadermatol.2015.2235). No conflicts of interest were declared.
The finding that patients with histologic regression have half the risk for micrometastases in sentinel lymph node biopsy, compared with patients without regression gives a clear answer to the question of whether patients with thin melanoma and regression should undergo SLNB, and that answer is “No.”

|
Dr. Claus Garbe |
These findings underline that fact that prognosis of patients with melanoma does not depend only on the characteristics of the tumor itself, such as tumor thickness, ulceration, and mitosis, but also on the immune response of the host.
Dr. Claus Garbe is from the Department of Dermatology, University Hospital Tübingen (Germany). These comments are taken from an accompanying editorial (JAMA Dermatol. 2015 Sep 2. doi: 10.1001/jamadermatol.2015.2235). No conflicts of interest were declared.
Histologic regression in melanoma is associated with a significantly lower risk of sentinel lymph node positivity and may serve as a prognostic factor when deciding whether to perform a sentinel lymph node biopsy, according to a study published online Sept. 2 in JAMA Dermatology.
Researchers conducted a meta-analysis of 14 studies in 10,098 patients who showed histologic regression of a primary melanoma and whose sentinel lymph node status was available. In the 14 studies combined, the analysis showed patients with histologic regression had a 44% lower likelihood of having a positive sentinel lymph node than patients without regression, reported Dr. Simone Ribero of the University of Turin (Italy), and coauthors (JAMA Dermatol. 2015 Sep 2. doi: 10.1001/jamadermatol.2015.2235).
This figure was even lower for patients enrolled in high-quality studies, compared with lower-quality studies, they said. In the nine studies considered high quality, patients with regression had a 52% lower likelihood of having a positive sentinel lymph node, while those enrolled in the five studies considered low quality had a 27% lower likelihood of having a positive sentinel lymph node.
The prognostic significance of regression in primary melanoma has been confounded by the fact that disappearance of part of the tumor can result in an underestimation of the original Breslow thickness. Some previous studies have found an increase in sentinel lymph node positivity associated with regression, while other studies suggested that regression was in fact a protective factor against sentinel lymph node metastasis.
Although the studies used different definitions of histologic regression and all were observational (counted among the limitations of the studies), the authors concluded that regression was prognostically favorable. “The results of this meta-analysis may be useful when deciding to offer SLNB [sentinel lymph node biopsy] to patients with regressions of melanomas,” they wrote, adding: “It may help clinicians make a final selection of the most appropriate patients for this procedure.”
The authors had no conflicts of interest to declare.
Histologic regression in melanoma is associated with a significantly lower risk of sentinel lymph node positivity and may serve as a prognostic factor when deciding whether to perform a sentinel lymph node biopsy, according to a study published online Sept. 2 in JAMA Dermatology.
Researchers conducted a meta-analysis of 14 studies in 10,098 patients who showed histologic regression of a primary melanoma and whose sentinel lymph node status was available. In the 14 studies combined, the analysis showed patients with histologic regression had a 44% lower likelihood of having a positive sentinel lymph node than patients without regression, reported Dr. Simone Ribero of the University of Turin (Italy), and coauthors (JAMA Dermatol. 2015 Sep 2. doi: 10.1001/jamadermatol.2015.2235).
This figure was even lower for patients enrolled in high-quality studies, compared with lower-quality studies, they said. In the nine studies considered high quality, patients with regression had a 52% lower likelihood of having a positive sentinel lymph node, while those enrolled in the five studies considered low quality had a 27% lower likelihood of having a positive sentinel lymph node.
The prognostic significance of regression in primary melanoma has been confounded by the fact that disappearance of part of the tumor can result in an underestimation of the original Breslow thickness. Some previous studies have found an increase in sentinel lymph node positivity associated with regression, while other studies suggested that regression was in fact a protective factor against sentinel lymph node metastasis.
Although the studies used different definitions of histologic regression and all were observational (counted among the limitations of the studies), the authors concluded that regression was prognostically favorable. “The results of this meta-analysis may be useful when deciding to offer SLNB [sentinel lymph node biopsy] to patients with regressions of melanomas,” they wrote, adding: “It may help clinicians make a final selection of the most appropriate patients for this procedure.”
The authors had no conflicts of interest to declare.
FROM JAMA DERMATOLOGY
Key clinical point: Histologic regression in melanoma is associated with a significantly lower risk of sentinel lymph node positivity.
Major finding: Individuals with histologic regression in melanoma had a 44% lower likelihood of having a positive sentinel lymph node than patients without regression.
Data source: A meta-analysis evaluated 14 studies in 10,098 patients.
Disclosures: None of the authors had conflicts of interest to disclose.
Characterization of Knuckle (Garrod) Pads Using Optical Coherence Tomography In Vivo
To the Editor:
Optical coherence tomography (OCT) is a noninvasive imaging technique that uses a low-power infrared laser light for cutaneous architecture visualization up to 2 mm in depth. Both malignant and nonmalignant lesions on OCT imaging have been correlated with histopathologic analysis.1 We describe the diagnostic features of knuckle pads on OCT.
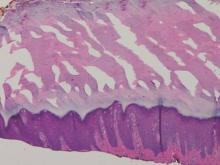
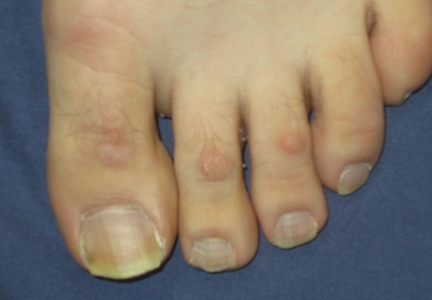
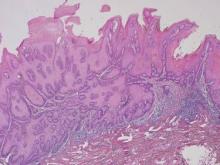
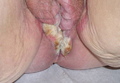
A 43-year-old-man presented with warts on the right thumb and bilateral feet of several months’ duration with noncontributory medical and social history. Physical examination revealed nontender, well-demarcated, flesh-colored, verrucous papules on the dorsal interphalangeal joints of the right thumb and several toes (Figure 1). Pinpoint vessels were absent on dermoscopy (Figure 2). Histopathologic analysis of a shave biopsy of the lesion on the left second toe revealed dense orthokeratosis with compact keratin, suggestive of reactive hyperkeratosis or a knuckle pad (Figure 3). In situ hybridization failed to demonstrate staining for human papillomavirus types 6, 11, and 16.

| 
| |
| Figure 1. Verrucous papules on the left foot.
| Figure 2. Corresponding dermoscopy of the left second toe revealed an absence of pinpoint vessels. |
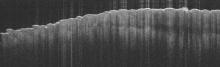
Optical coherence tomography demonstrated discrete thickening of the stratum corneum with distinctive granular and coarse textural appearance of the hyperkeratotic stratum corneum compared to normal adjacent skin. This textural difference was attributed to the alteration in collagen deposition of the knuckle pads, consistent with fibrous proliferation. Finally, OCT imaging provided further characterization of the lesion demonstrating the absence of any hair follicles and acrosyringium in areas resembling glabrous skin (Figure 4).
Knuckle pads, also known as Garrod pads, were first described by Garrod2 in 1893. They are benign, asymptomatic, fibrotic thickenings of the skin. Lesions are smooth, firm, flesh colored, and located on the dorsal aspect of the hands and feet along the metacarpophalangeal and interphalangeal joints. Knuckle pads are common, can develop at any age, and are observed more frequently in men than in women.3
Primary knuckle pads can be sporadic or associated with other conditions such as palmoplantar keratoderma, acrokeratoelastoidosis costa, fibrosing disorders, or Bart-Pumphrey syndrome.4 Secondary knuckle pads, which are more common, occur in sites of repetitive trauma or pressure. Certain occupations (eg, mechanics) or hobbies (eg, boxing) increase the risk for developing knuckle pads.3,4
The diagnosis of knuckle pads is usually made clinically, though several other conditions mimic knuckle pads, including scars, keloids, calluses, verruca vulgaris, fibromas, and rheumatoid nodules.3,5 We report a description of knuckle pads that was diagnosed with OCT imaging. Further characterization of both malignant and nonmalignant lesions on OCT imaging will contribute new insights to the role of OCT in the noninvasive diagnosis of skin diseases, pending future studies.
1. Forsea AM, Carstea EM, Ghervase L, et al. Clinical application of optical coherence tomography for the imaging of non-melanocytic cutaneous tumors: a pilot multi-modal study. J Med Life. 2010;3:381-389.
2. Garrod AE. On an unusual form of nodule upon joints of the fingers. St Bartholomew’s Hosp Rep. 1893;29:157-161.
3. Kodama BF, Gentry RH, Fitzpatrick JE. Papules and plaques over the joint spaces. knuckle pads (heloderma). Arch Dermatol. 1993;129:1044-1045, 1047.
4. Nenoff P, Woitek G. Images in clinical medicine. knuckle pads. N Engl J Med. 2011;364:2451.
5. Sehgal VN, Singh M, Saxena HM, et al. Primary knuckle pads. Clin Exp Dermatol. 1979;4:337-339.
To the Editor:
Optical coherence tomography (OCT) is a noninvasive imaging technique that uses a low-power infrared laser light for cutaneous architecture visualization up to 2 mm in depth. Both malignant and nonmalignant lesions on OCT imaging have been correlated with histopathologic analysis.1 We describe the diagnostic features of knuckle pads on OCT.
A 43-year-old-man presented with warts on the right thumb and bilateral feet of several months’ duration with noncontributory medical and social history. Physical examination revealed nontender, well-demarcated, flesh-colored, verrucous papules on the dorsal interphalangeal joints of the right thumb and several toes (Figure 1). Pinpoint vessels were absent on dermoscopy (Figure 2). Histopathologic analysis of a shave biopsy of the lesion on the left second toe revealed dense orthokeratosis with compact keratin, suggestive of reactive hyperkeratosis or a knuckle pad (Figure 3). In situ hybridization failed to demonstrate staining for human papillomavirus types 6, 11, and 16.

| 
| |
| Figure 1. Verrucous papules on the left foot.
| Figure 2. Corresponding dermoscopy of the left second toe revealed an absence of pinpoint vessels. |
Optical coherence tomography demonstrated discrete thickening of the stratum corneum with distinctive granular and coarse textural appearance of the hyperkeratotic stratum corneum compared to normal adjacent skin. This textural difference was attributed to the alteration in collagen deposition of the knuckle pads, consistent with fibrous proliferation. Finally, OCT imaging provided further characterization of the lesion demonstrating the absence of any hair follicles and acrosyringium in areas resembling glabrous skin (Figure 4).
Knuckle pads, also known as Garrod pads, were first described by Garrod2 in 1893. They are benign, asymptomatic, fibrotic thickenings of the skin. Lesions are smooth, firm, flesh colored, and located on the dorsal aspect of the hands and feet along the metacarpophalangeal and interphalangeal joints. Knuckle pads are common, can develop at any age, and are observed more frequently in men than in women.3
Primary knuckle pads can be sporadic or associated with other conditions such as palmoplantar keratoderma, acrokeratoelastoidosis costa, fibrosing disorders, or Bart-Pumphrey syndrome.4 Secondary knuckle pads, which are more common, occur in sites of repetitive trauma or pressure. Certain occupations (eg, mechanics) or hobbies (eg, boxing) increase the risk for developing knuckle pads.3,4
The diagnosis of knuckle pads is usually made clinically, though several other conditions mimic knuckle pads, including scars, keloids, calluses, verruca vulgaris, fibromas, and rheumatoid nodules.3,5 We report a description of knuckle pads that was diagnosed with OCT imaging. Further characterization of both malignant and nonmalignant lesions on OCT imaging will contribute new insights to the role of OCT in the noninvasive diagnosis of skin diseases, pending future studies.
To the Editor:
Optical coherence tomography (OCT) is a noninvasive imaging technique that uses a low-power infrared laser light for cutaneous architecture visualization up to 2 mm in depth. Both malignant and nonmalignant lesions on OCT imaging have been correlated with histopathologic analysis.1 We describe the diagnostic features of knuckle pads on OCT.
A 43-year-old-man presented with warts on the right thumb and bilateral feet of several months’ duration with noncontributory medical and social history. Physical examination revealed nontender, well-demarcated, flesh-colored, verrucous papules on the dorsal interphalangeal joints of the right thumb and several toes (Figure 1). Pinpoint vessels were absent on dermoscopy (Figure 2). Histopathologic analysis of a shave biopsy of the lesion on the left second toe revealed dense orthokeratosis with compact keratin, suggestive of reactive hyperkeratosis or a knuckle pad (Figure 3). In situ hybridization failed to demonstrate staining for human papillomavirus types 6, 11, and 16.

| 
| |
| Figure 1. Verrucous papules on the left foot.
| Figure 2. Corresponding dermoscopy of the left second toe revealed an absence of pinpoint vessels. |
Optical coherence tomography demonstrated discrete thickening of the stratum corneum with distinctive granular and coarse textural appearance of the hyperkeratotic stratum corneum compared to normal adjacent skin. This textural difference was attributed to the alteration in collagen deposition of the knuckle pads, consistent with fibrous proliferation. Finally, OCT imaging provided further characterization of the lesion demonstrating the absence of any hair follicles and acrosyringium in areas resembling glabrous skin (Figure 4).
Knuckle pads, also known as Garrod pads, were first described by Garrod2 in 1893. They are benign, asymptomatic, fibrotic thickenings of the skin. Lesions are smooth, firm, flesh colored, and located on the dorsal aspect of the hands and feet along the metacarpophalangeal and interphalangeal joints. Knuckle pads are common, can develop at any age, and are observed more frequently in men than in women.3
Primary knuckle pads can be sporadic or associated with other conditions such as palmoplantar keratoderma, acrokeratoelastoidosis costa, fibrosing disorders, or Bart-Pumphrey syndrome.4 Secondary knuckle pads, which are more common, occur in sites of repetitive trauma or pressure. Certain occupations (eg, mechanics) or hobbies (eg, boxing) increase the risk for developing knuckle pads.3,4
The diagnosis of knuckle pads is usually made clinically, though several other conditions mimic knuckle pads, including scars, keloids, calluses, verruca vulgaris, fibromas, and rheumatoid nodules.3,5 We report a description of knuckle pads that was diagnosed with OCT imaging. Further characterization of both malignant and nonmalignant lesions on OCT imaging will contribute new insights to the role of OCT in the noninvasive diagnosis of skin diseases, pending future studies.
1. Forsea AM, Carstea EM, Ghervase L, et al. Clinical application of optical coherence tomography for the imaging of non-melanocytic cutaneous tumors: a pilot multi-modal study. J Med Life. 2010;3:381-389.
2. Garrod AE. On an unusual form of nodule upon joints of the fingers. St Bartholomew’s Hosp Rep. 1893;29:157-161.
3. Kodama BF, Gentry RH, Fitzpatrick JE. Papules and plaques over the joint spaces. knuckle pads (heloderma). Arch Dermatol. 1993;129:1044-1045, 1047.
4. Nenoff P, Woitek G. Images in clinical medicine. knuckle pads. N Engl J Med. 2011;364:2451.
5. Sehgal VN, Singh M, Saxena HM, et al. Primary knuckle pads. Clin Exp Dermatol. 1979;4:337-339.
1. Forsea AM, Carstea EM, Ghervase L, et al. Clinical application of optical coherence tomography for the imaging of non-melanocytic cutaneous tumors: a pilot multi-modal study. J Med Life. 2010;3:381-389.
2. Garrod AE. On an unusual form of nodule upon joints of the fingers. St Bartholomew’s Hosp Rep. 1893;29:157-161.
3. Kodama BF, Gentry RH, Fitzpatrick JE. Papules and plaques over the joint spaces. knuckle pads (heloderma). Arch Dermatol. 1993;129:1044-1045, 1047.
4. Nenoff P, Woitek G. Images in clinical medicine. knuckle pads. N Engl J Med. 2011;364:2451.
5. Sehgal VN, Singh M, Saxena HM, et al. Primary knuckle pads. Clin Exp Dermatol. 1979;4:337-339.
Early intervention may forestall menopause-related skin aging
NEW YORK – Evidence is mounting that early intervention in the menopausal transition could help forestall some of the skin aging associated with estrogen decline.
Estrogen supplementation and collagen stimulation both seem effective in preserving the integrity of a woman’s skin as levels of the hormone decrease, Dr. Diane Madfes said at the American Academy of Dermatology summer meeting.
Type 3 collagen decreases by up to 50% within a few years of menopause, said Dr. Madfes, a dermatologist in New York. This is directly related to a loss of estrogen receptor beta in the dermal matrix, which promotes collagen formation.
“There is a theory – the timing hypothesis – that we have a window of opportunity to intervene. If we can stimulate the collagen before the receptors go down, maybe we can have a beneficial effect on skin.”
Any method of collagen stimulation should work, she said: laser resurfacing, microneedling, or radiofrequency. “We are very good about being able to stimulate collagen. The method doesn’t matter as much as the timing. The important thing is to intervene early. If you see your patients starting to sag, see a loss of elasticity, that is the time to intervene. Get at the collagen while it’s still receptive.”
Estrogen exerts a plethora of antiaging, skin-preserving effects. “We know that a decrease in estrogen is related to telomere shortening. Estrogen protects against oxidative damage. It signals keratinocytes through IGF-1,” she said.
The hormone also protects skin’s water-binding qualities by promoting mucopolysaccharides, sebum production, barrier function, and hyaluronic acid. It may even play a role in protecting against ultraviolet light. Estrogen downregulation affects healing by inhibiting the proliferation of keratinocytes and the proliferation and migration of fibroblasts.
All these add up to rapid skin aging after estrogen levels drop.
“The visible effects of aging on women’s skin are not so much related to her chronological age as to the years after menopause,” Dr. Madfes said – a finding that is particularly illustrated in young women with surgical menopause and those with breast cancer who take tamoxifen. The observation seems to suggest that early intervention with estrogen might help prevent at least some of the signs of aging.
The ongoing KEEPS trial (Kronos Early Estrogen Prevention Study) may shed some light on the issue. KEEPS has randomized 729 women aged 42-58 years to oral or transdermal estrogen; the primary endpoint is rate of atherosclerosis. But an ancillary study is looking at the effect of estrogen on skin wrinkles and skin rigidity.
The substudy is based on positive findings of a 1996 study, which found evidence for facial application of topical estrogen designed for vulvar use. After 6 months, elasticity and firmness significantly improved. Skin moisture increased, as did type 3 collagen and collagen fibers.
Some women do use topical estrogens on their faces. “It seems to promote skin thickening and tightening,“ Dr. Madfes said, although a recent editorial suggested that using the product anywhere but on the genitals can cause estrogen-mediated side effects in both children and pets.
But recommending estrogen is fraught with controversy. Large studies have come to conflicting conclusions about its benefit and safety. And prescribing estrogen is not really within a dermatologist’s purview.
“It’s not for us to suggest that women go on hormone therapy. But we can explain these things and ask if she is taking it, or if she’s talked to her gynecologist about it.”
Dr. Madfes has no financial disclosures to report.
On Twitter @Alz_Gal
NEW YORK – Evidence is mounting that early intervention in the menopausal transition could help forestall some of the skin aging associated with estrogen decline.
Estrogen supplementation and collagen stimulation both seem effective in preserving the integrity of a woman’s skin as levels of the hormone decrease, Dr. Diane Madfes said at the American Academy of Dermatology summer meeting.
Type 3 collagen decreases by up to 50% within a few years of menopause, said Dr. Madfes, a dermatologist in New York. This is directly related to a loss of estrogen receptor beta in the dermal matrix, which promotes collagen formation.
“There is a theory – the timing hypothesis – that we have a window of opportunity to intervene. If we can stimulate the collagen before the receptors go down, maybe we can have a beneficial effect on skin.”
Any method of collagen stimulation should work, she said: laser resurfacing, microneedling, or radiofrequency. “We are very good about being able to stimulate collagen. The method doesn’t matter as much as the timing. The important thing is to intervene early. If you see your patients starting to sag, see a loss of elasticity, that is the time to intervene. Get at the collagen while it’s still receptive.”
Estrogen exerts a plethora of antiaging, skin-preserving effects. “We know that a decrease in estrogen is related to telomere shortening. Estrogen protects against oxidative damage. It signals keratinocytes through IGF-1,” she said.
The hormone also protects skin’s water-binding qualities by promoting mucopolysaccharides, sebum production, barrier function, and hyaluronic acid. It may even play a role in protecting against ultraviolet light. Estrogen downregulation affects healing by inhibiting the proliferation of keratinocytes and the proliferation and migration of fibroblasts.
All these add up to rapid skin aging after estrogen levels drop.
“The visible effects of aging on women’s skin are not so much related to her chronological age as to the years after menopause,” Dr. Madfes said – a finding that is particularly illustrated in young women with surgical menopause and those with breast cancer who take tamoxifen. The observation seems to suggest that early intervention with estrogen might help prevent at least some of the signs of aging.
The ongoing KEEPS trial (Kronos Early Estrogen Prevention Study) may shed some light on the issue. KEEPS has randomized 729 women aged 42-58 years to oral or transdermal estrogen; the primary endpoint is rate of atherosclerosis. But an ancillary study is looking at the effect of estrogen on skin wrinkles and skin rigidity.
The substudy is based on positive findings of a 1996 study, which found evidence for facial application of topical estrogen designed for vulvar use. After 6 months, elasticity and firmness significantly improved. Skin moisture increased, as did type 3 collagen and collagen fibers.
Some women do use topical estrogens on their faces. “It seems to promote skin thickening and tightening,“ Dr. Madfes said, although a recent editorial suggested that using the product anywhere but on the genitals can cause estrogen-mediated side effects in both children and pets.
But recommending estrogen is fraught with controversy. Large studies have come to conflicting conclusions about its benefit and safety. And prescribing estrogen is not really within a dermatologist’s purview.
“It’s not for us to suggest that women go on hormone therapy. But we can explain these things and ask if she is taking it, or if she’s talked to her gynecologist about it.”
Dr. Madfes has no financial disclosures to report.
On Twitter @Alz_Gal
NEW YORK – Evidence is mounting that early intervention in the menopausal transition could help forestall some of the skin aging associated with estrogen decline.
Estrogen supplementation and collagen stimulation both seem effective in preserving the integrity of a woman’s skin as levels of the hormone decrease, Dr. Diane Madfes said at the American Academy of Dermatology summer meeting.
Type 3 collagen decreases by up to 50% within a few years of menopause, said Dr. Madfes, a dermatologist in New York. This is directly related to a loss of estrogen receptor beta in the dermal matrix, which promotes collagen formation.
“There is a theory – the timing hypothesis – that we have a window of opportunity to intervene. If we can stimulate the collagen before the receptors go down, maybe we can have a beneficial effect on skin.”
Any method of collagen stimulation should work, she said: laser resurfacing, microneedling, or radiofrequency. “We are very good about being able to stimulate collagen. The method doesn’t matter as much as the timing. The important thing is to intervene early. If you see your patients starting to sag, see a loss of elasticity, that is the time to intervene. Get at the collagen while it’s still receptive.”
Estrogen exerts a plethora of antiaging, skin-preserving effects. “We know that a decrease in estrogen is related to telomere shortening. Estrogen protects against oxidative damage. It signals keratinocytes through IGF-1,” she said.
The hormone also protects skin’s water-binding qualities by promoting mucopolysaccharides, sebum production, barrier function, and hyaluronic acid. It may even play a role in protecting against ultraviolet light. Estrogen downregulation affects healing by inhibiting the proliferation of keratinocytes and the proliferation and migration of fibroblasts.
All these add up to rapid skin aging after estrogen levels drop.
“The visible effects of aging on women’s skin are not so much related to her chronological age as to the years after menopause,” Dr. Madfes said – a finding that is particularly illustrated in young women with surgical menopause and those with breast cancer who take tamoxifen. The observation seems to suggest that early intervention with estrogen might help prevent at least some of the signs of aging.
The ongoing KEEPS trial (Kronos Early Estrogen Prevention Study) may shed some light on the issue. KEEPS has randomized 729 women aged 42-58 years to oral or transdermal estrogen; the primary endpoint is rate of atherosclerosis. But an ancillary study is looking at the effect of estrogen on skin wrinkles and skin rigidity.
The substudy is based on positive findings of a 1996 study, which found evidence for facial application of topical estrogen designed for vulvar use. After 6 months, elasticity and firmness significantly improved. Skin moisture increased, as did type 3 collagen and collagen fibers.
Some women do use topical estrogens on their faces. “It seems to promote skin thickening and tightening,“ Dr. Madfes said, although a recent editorial suggested that using the product anywhere but on the genitals can cause estrogen-mediated side effects in both children and pets.
But recommending estrogen is fraught with controversy. Large studies have come to conflicting conclusions about its benefit and safety. And prescribing estrogen is not really within a dermatologist’s purview.
“It’s not for us to suggest that women go on hormone therapy. But we can explain these things and ask if she is taking it, or if she’s talked to her gynecologist about it.”
Dr. Madfes has no financial disclosures to report.
On Twitter @Alz_Gal
EXPERT ANALYSIS FROM THE AAD SUMMER ACADEMY 2015
Treatment options for lentigo maligna far from perfect
PARK CITY, UTAH – Dr. Glen M. Bowen has been working to improve the surgical treatment of lentigo maligna ever since he joined the Huntsman Cancer Institute at the University of Utah 16 years ago. A retrospective review from Memorial Sloan-Kettering Cancer Center found that on average, 7.1-mm margins are required to remove lentigo maligna (LM) (J. Am. Acad. Dermatol. 2008;58[1]:142-8).
“If you have an LM with a 10-mm diameter to begin with, 7.1-mm margins give you a final surgical diameter of 24.2 mm,” Dr. Bowen said at the annual meeting of the Pacific Dermatologic Association. “These are very morbid surgeries in cosmetically sensitive areas for a relatively low-risk tumor.”
The risk of LM progressing to an invasive melanoma is not known, but is estimated to range between 5% and 33%. Of 2,016 patients treated for LM at the Huntsman Cancer Institute, 522 have been treated with neo-adjuvant topical imiquimod 5% cream followed by a conservative staged excision with 2-mm margins with a recurrence rate of 2.3% during median follow-up of 5 years. Of their recurrences, about 20% recurred with invasion. All recurrences to date have been less than 1 mm in depth (stage IA), which has an estimated mortality risk of 5% at 5 years. “A 5% mortality rate of the 20% that recur with invasion of the 2.3% that recur after surgery yields a mortality risk of 0.023%,” Dr. Bowen said. “Due to the very low risk of actually dying from a recurrent LM, very large and morbid surgical defects strike me as a punishment that doesn’t fit the crime in terms of the cost-benefit ratio.”
He and his associates at the Huntsman Cancer Institute have observed some deaths in patients who presented with LM melanoma (invasive melanoma) but have not observed a single death in patients who presented with LM in situ that subsequently recurred. For these reasons, Dr. Bowen favors pretreating LM with imiquimod 5% cream followed by a conservative staged excision, a process that substantially decreases the size of the surgical defects.
His current treatment protocol involves a five-step process that begins with removing all of the visible lentigo maligna to rule out invasion, since 16% of LMs referred to him have harbored invasion when removed and are upgraded from stage 0 to IA. “I am not going to use a topical cream on an invasive melanoma,” he said. “After an excisional biopsy with minimal margins, I usually close the defect with a purse-string suture because it avoids removing standing cones and consequently enlarging the treatment area.”
Second, he traces a template of the LM border on transparent plastic and places a tiny tattoo in the center of the biopsy site to enable pinpoint placement of the template at the time of surgery.
Third, he treats the site with imiquimod 5% cream Monday through Friday for 2-3 months and sees the patient monthly for dosage adjustments when needed. The fourth step involves enabling the site to recover for 2-6 months to allow for resolution of the inflammatory infiltrate. The final step involves re-excising around the original template with 2-mm margins for confirmation with the use of a negative control taken from an equally sun-exposed site taken some distance away from the LM. “Caucasians will have atypical junctional melanocytic hyperplasia (AJMH), which must be subtracted out as background,” he said. “Otherwise, if you hold a non–sun-exposed site as your standard for a negative margin, you will never stop cutting.”
Dr. Bowen likes to use frozen radial sections with routine staining with H and E and immunostaining with MART-1 (Melan-A) and SOX-10. Processing takes 2 hours, he continued, “so I put in relaxing sutures, which will stretch out nicely over 2 hours so I can usually close the defects primarily.”
In Dr. Bowen’s opinion, topical imiquimod as monotherapy for LM is not safe, since about 30% of patients treated with imiquimod will still harbor residual LM. “In our dataset, about 70% have no residual LM, 20% have residual LM in the center but negative perimeter margins, and 10% have LM touching a perimeter margin and require a second stage,” he said. “Taken together, 90% of patients pretreated with imiquimod will be cleared in one stage of surgery with 2 mm margins.”
Making the distinction between LM and AJMH common to chronically sun-damaged skin is no easy task. Dr. Bowen cited a concordance study between dermatopathologists interpreting staged excisional margins on permanent sections for LM where the concordance was only moderate at best. In this study, the use of a negative control improved the concordance rate on “difficult” cases from 46% to 76%; P = .001 (Arch Dermatol. 2003 May;139(5):595-604). “What we really need is a molecular marker that will tell us if a melanocyte is malignant or not,” he said. “All we have now are immunostains that tell you if it’s melanocyte but nothing more.” He went on to say that in multivariate analysis in two studies of the histologic features of LM, the only feature that consistently predicted the difference between LM and AJMH was the melanocyte density and its ratio to the negative control (Dermatol Surg. 2011;37(5):657-63 and J. Plast. Reconstr. Aesthet. Surg. 2014;67(10):1322-32). “The MART-1 immunostain is extremely sensitive, but it makes the slide somewhat muddy, so it’s hard to do an accurate cell count,” he said. “For that reason, we also use a SOX-10 immunostain which is very specific but not as sensitive. I believe that the truth lies somewhere in between those two immunostains in light of a positive control from our lab and a negative control from the patient.”
He concluded that the neoadjuvant use of imiquimod followed by a conservative staged excision “allows me to clear 90% of LM with a 2 mm margin with a recurrence rate of 2.3% in patients with a mean follow-up of 5-years or greater.”
Dr. Bowen reported having no financial disclosures.
PARK CITY, UTAH – Dr. Glen M. Bowen has been working to improve the surgical treatment of lentigo maligna ever since he joined the Huntsman Cancer Institute at the University of Utah 16 years ago. A retrospective review from Memorial Sloan-Kettering Cancer Center found that on average, 7.1-mm margins are required to remove lentigo maligna (LM) (J. Am. Acad. Dermatol. 2008;58[1]:142-8).
“If you have an LM with a 10-mm diameter to begin with, 7.1-mm margins give you a final surgical diameter of 24.2 mm,” Dr. Bowen said at the annual meeting of the Pacific Dermatologic Association. “These are very morbid surgeries in cosmetically sensitive areas for a relatively low-risk tumor.”
The risk of LM progressing to an invasive melanoma is not known, but is estimated to range between 5% and 33%. Of 2,016 patients treated for LM at the Huntsman Cancer Institute, 522 have been treated with neo-adjuvant topical imiquimod 5% cream followed by a conservative staged excision with 2-mm margins with a recurrence rate of 2.3% during median follow-up of 5 years. Of their recurrences, about 20% recurred with invasion. All recurrences to date have been less than 1 mm in depth (stage IA), which has an estimated mortality risk of 5% at 5 years. “A 5% mortality rate of the 20% that recur with invasion of the 2.3% that recur after surgery yields a mortality risk of 0.023%,” Dr. Bowen said. “Due to the very low risk of actually dying from a recurrent LM, very large and morbid surgical defects strike me as a punishment that doesn’t fit the crime in terms of the cost-benefit ratio.”
He and his associates at the Huntsman Cancer Institute have observed some deaths in patients who presented with LM melanoma (invasive melanoma) but have not observed a single death in patients who presented with LM in situ that subsequently recurred. For these reasons, Dr. Bowen favors pretreating LM with imiquimod 5% cream followed by a conservative staged excision, a process that substantially decreases the size of the surgical defects.
His current treatment protocol involves a five-step process that begins with removing all of the visible lentigo maligna to rule out invasion, since 16% of LMs referred to him have harbored invasion when removed and are upgraded from stage 0 to IA. “I am not going to use a topical cream on an invasive melanoma,” he said. “After an excisional biopsy with minimal margins, I usually close the defect with a purse-string suture because it avoids removing standing cones and consequently enlarging the treatment area.”
Second, he traces a template of the LM border on transparent plastic and places a tiny tattoo in the center of the biopsy site to enable pinpoint placement of the template at the time of surgery.
Third, he treats the site with imiquimod 5% cream Monday through Friday for 2-3 months and sees the patient monthly for dosage adjustments when needed. The fourth step involves enabling the site to recover for 2-6 months to allow for resolution of the inflammatory infiltrate. The final step involves re-excising around the original template with 2-mm margins for confirmation with the use of a negative control taken from an equally sun-exposed site taken some distance away from the LM. “Caucasians will have atypical junctional melanocytic hyperplasia (AJMH), which must be subtracted out as background,” he said. “Otherwise, if you hold a non–sun-exposed site as your standard for a negative margin, you will never stop cutting.”
Dr. Bowen likes to use frozen radial sections with routine staining with H and E and immunostaining with MART-1 (Melan-A) and SOX-10. Processing takes 2 hours, he continued, “so I put in relaxing sutures, which will stretch out nicely over 2 hours so I can usually close the defects primarily.”
In Dr. Bowen’s opinion, topical imiquimod as monotherapy for LM is not safe, since about 30% of patients treated with imiquimod will still harbor residual LM. “In our dataset, about 70% have no residual LM, 20% have residual LM in the center but negative perimeter margins, and 10% have LM touching a perimeter margin and require a second stage,” he said. “Taken together, 90% of patients pretreated with imiquimod will be cleared in one stage of surgery with 2 mm margins.”
Making the distinction between LM and AJMH common to chronically sun-damaged skin is no easy task. Dr. Bowen cited a concordance study between dermatopathologists interpreting staged excisional margins on permanent sections for LM where the concordance was only moderate at best. In this study, the use of a negative control improved the concordance rate on “difficult” cases from 46% to 76%; P = .001 (Arch Dermatol. 2003 May;139(5):595-604). “What we really need is a molecular marker that will tell us if a melanocyte is malignant or not,” he said. “All we have now are immunostains that tell you if it’s melanocyte but nothing more.” He went on to say that in multivariate analysis in two studies of the histologic features of LM, the only feature that consistently predicted the difference between LM and AJMH was the melanocyte density and its ratio to the negative control (Dermatol Surg. 2011;37(5):657-63 and J. Plast. Reconstr. Aesthet. Surg. 2014;67(10):1322-32). “The MART-1 immunostain is extremely sensitive, but it makes the slide somewhat muddy, so it’s hard to do an accurate cell count,” he said. “For that reason, we also use a SOX-10 immunostain which is very specific but not as sensitive. I believe that the truth lies somewhere in between those two immunostains in light of a positive control from our lab and a negative control from the patient.”
He concluded that the neoadjuvant use of imiquimod followed by a conservative staged excision “allows me to clear 90% of LM with a 2 mm margin with a recurrence rate of 2.3% in patients with a mean follow-up of 5-years or greater.”
Dr. Bowen reported having no financial disclosures.
PARK CITY, UTAH – Dr. Glen M. Bowen has been working to improve the surgical treatment of lentigo maligna ever since he joined the Huntsman Cancer Institute at the University of Utah 16 years ago. A retrospective review from Memorial Sloan-Kettering Cancer Center found that on average, 7.1-mm margins are required to remove lentigo maligna (LM) (J. Am. Acad. Dermatol. 2008;58[1]:142-8).
“If you have an LM with a 10-mm diameter to begin with, 7.1-mm margins give you a final surgical diameter of 24.2 mm,” Dr. Bowen said at the annual meeting of the Pacific Dermatologic Association. “These are very morbid surgeries in cosmetically sensitive areas for a relatively low-risk tumor.”
The risk of LM progressing to an invasive melanoma is not known, but is estimated to range between 5% and 33%. Of 2,016 patients treated for LM at the Huntsman Cancer Institute, 522 have been treated with neo-adjuvant topical imiquimod 5% cream followed by a conservative staged excision with 2-mm margins with a recurrence rate of 2.3% during median follow-up of 5 years. Of their recurrences, about 20% recurred with invasion. All recurrences to date have been less than 1 mm in depth (stage IA), which has an estimated mortality risk of 5% at 5 years. “A 5% mortality rate of the 20% that recur with invasion of the 2.3% that recur after surgery yields a mortality risk of 0.023%,” Dr. Bowen said. “Due to the very low risk of actually dying from a recurrent LM, very large and morbid surgical defects strike me as a punishment that doesn’t fit the crime in terms of the cost-benefit ratio.”
He and his associates at the Huntsman Cancer Institute have observed some deaths in patients who presented with LM melanoma (invasive melanoma) but have not observed a single death in patients who presented with LM in situ that subsequently recurred. For these reasons, Dr. Bowen favors pretreating LM with imiquimod 5% cream followed by a conservative staged excision, a process that substantially decreases the size of the surgical defects.
His current treatment protocol involves a five-step process that begins with removing all of the visible lentigo maligna to rule out invasion, since 16% of LMs referred to him have harbored invasion when removed and are upgraded from stage 0 to IA. “I am not going to use a topical cream on an invasive melanoma,” he said. “After an excisional biopsy with minimal margins, I usually close the defect with a purse-string suture because it avoids removing standing cones and consequently enlarging the treatment area.”
Second, he traces a template of the LM border on transparent plastic and places a tiny tattoo in the center of the biopsy site to enable pinpoint placement of the template at the time of surgery.
Third, he treats the site with imiquimod 5% cream Monday through Friday for 2-3 months and sees the patient monthly for dosage adjustments when needed. The fourth step involves enabling the site to recover for 2-6 months to allow for resolution of the inflammatory infiltrate. The final step involves re-excising around the original template with 2-mm margins for confirmation with the use of a negative control taken from an equally sun-exposed site taken some distance away from the LM. “Caucasians will have atypical junctional melanocytic hyperplasia (AJMH), which must be subtracted out as background,” he said. “Otherwise, if you hold a non–sun-exposed site as your standard for a negative margin, you will never stop cutting.”
Dr. Bowen likes to use frozen radial sections with routine staining with H and E and immunostaining with MART-1 (Melan-A) and SOX-10. Processing takes 2 hours, he continued, “so I put in relaxing sutures, which will stretch out nicely over 2 hours so I can usually close the defects primarily.”
In Dr. Bowen’s opinion, topical imiquimod as monotherapy for LM is not safe, since about 30% of patients treated with imiquimod will still harbor residual LM. “In our dataset, about 70% have no residual LM, 20% have residual LM in the center but negative perimeter margins, and 10% have LM touching a perimeter margin and require a second stage,” he said. “Taken together, 90% of patients pretreated with imiquimod will be cleared in one stage of surgery with 2 mm margins.”
Making the distinction between LM and AJMH common to chronically sun-damaged skin is no easy task. Dr. Bowen cited a concordance study between dermatopathologists interpreting staged excisional margins on permanent sections for LM where the concordance was only moderate at best. In this study, the use of a negative control improved the concordance rate on “difficult” cases from 46% to 76%; P = .001 (Arch Dermatol. 2003 May;139(5):595-604). “What we really need is a molecular marker that will tell us if a melanocyte is malignant or not,” he said. “All we have now are immunostains that tell you if it’s melanocyte but nothing more.” He went on to say that in multivariate analysis in two studies of the histologic features of LM, the only feature that consistently predicted the difference between LM and AJMH was the melanocyte density and its ratio to the negative control (Dermatol Surg. 2011;37(5):657-63 and J. Plast. Reconstr. Aesthet. Surg. 2014;67(10):1322-32). “The MART-1 immunostain is extremely sensitive, but it makes the slide somewhat muddy, so it’s hard to do an accurate cell count,” he said. “For that reason, we also use a SOX-10 immunostain which is very specific but not as sensitive. I believe that the truth lies somewhere in between those two immunostains in light of a positive control from our lab and a negative control from the patient.”
He concluded that the neoadjuvant use of imiquimod followed by a conservative staged excision “allows me to clear 90% of LM with a 2 mm margin with a recurrence rate of 2.3% in patients with a mean follow-up of 5-years or greater.”
Dr. Bowen reported having no financial disclosures.
EXPERT ANALYSIS from PDA 2015
Survey identifies top cosmetic concerns
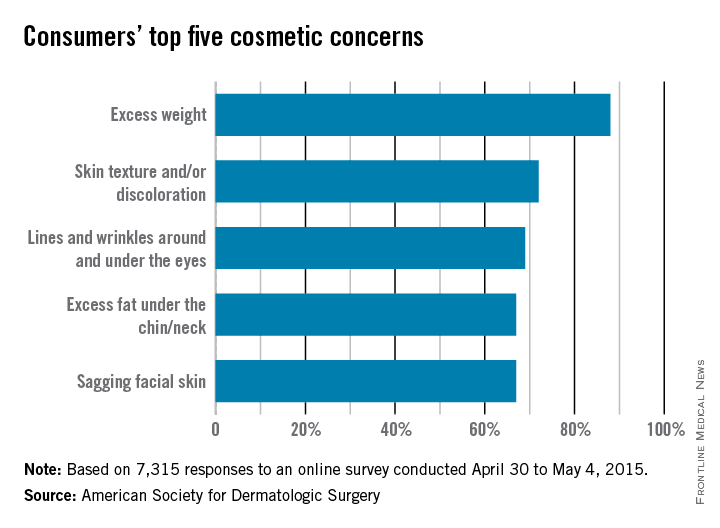
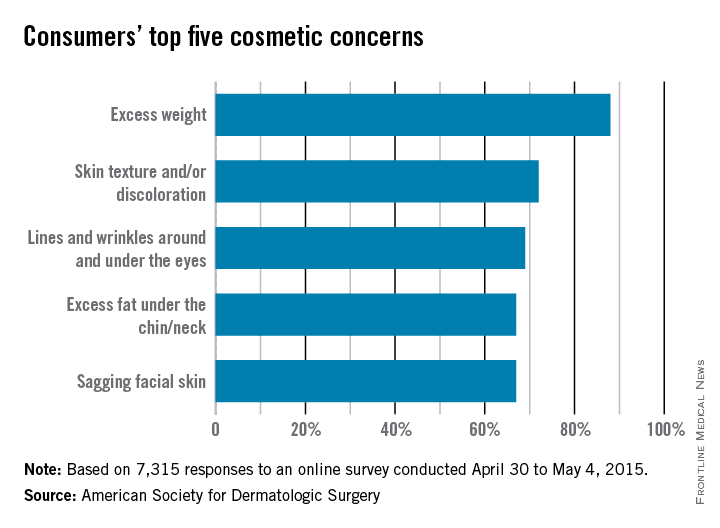
The American Society for Dermatologic Surgery took a poll this spring, and the winner was fat.
The ASDS asked people to identify their biggest cosmetic concerns, and 88% said that they were somewhat to extremely bothered by excess weight. The next most-concerning condition was “skin texture and/or discoloration” (72%), followed by “lines and wrinkles around and under the eyes” (69%), “excess fat [there it is again] under the chin/neck” (67%), and “sagging facial skin” (67%).
More than 207,000 body-sculpting procedures were performed by ASDS members in 2014, with cryolipolysis the most-commonly done procedure.
The 7,315 survey respondents were less-concerned about “lines and wrinkles between my eyebrows” (55%), “hair loss” (43%), and “acne scars” (27%), according to the ASDS.
About half of the respondents were considering a cosmetic procedure, with “ultrasound, laser, light, and radiofrequency treatments for skin tightening or wrinkles” the top choice at 66% and “laser hair removal” next at 55%, the ASDS reported.
The American Society for Dermatologic Surgery took a poll this spring, and the winner was fat.
The ASDS asked people to identify their biggest cosmetic concerns, and 88% said that they were somewhat to extremely bothered by excess weight. The next most-concerning condition was “skin texture and/or discoloration” (72%), followed by “lines and wrinkles around and under the eyes” (69%), “excess fat [there it is again] under the chin/neck” (67%), and “sagging facial skin” (67%).
More than 207,000 body-sculpting procedures were performed by ASDS members in 2014, with cryolipolysis the most-commonly done procedure.
The 7,315 survey respondents were less-concerned about “lines and wrinkles between my eyebrows” (55%), “hair loss” (43%), and “acne scars” (27%), according to the ASDS.
About half of the respondents were considering a cosmetic procedure, with “ultrasound, laser, light, and radiofrequency treatments for skin tightening or wrinkles” the top choice at 66% and “laser hair removal” next at 55%, the ASDS reported.

The American Society for Dermatologic Surgery took a poll this spring, and the winner was fat.
The ASDS asked people to identify their biggest cosmetic concerns, and 88% said that they were somewhat to extremely bothered by excess weight. The next most-concerning condition was “skin texture and/or discoloration” (72%), followed by “lines and wrinkles around and under the eyes” (69%), “excess fat [there it is again] under the chin/neck” (67%), and “sagging facial skin” (67%).
More than 207,000 body-sculpting procedures were performed by ASDS members in 2014, with cryolipolysis the most-commonly done procedure.
The 7,315 survey respondents were less-concerned about “lines and wrinkles between my eyebrows” (55%), “hair loss” (43%), and “acne scars” (27%), according to the ASDS.
About half of the respondents were considering a cosmetic procedure, with “ultrasound, laser, light, and radiofrequency treatments for skin tightening or wrinkles” the top choice at 66% and “laser hair removal” next at 55%, the ASDS reported.

Indoor Tanning Is More Harmful Than Americans Believe
The Surgeon General has called on partners in prevention from various sectors to address skin cancer as a major public health problem. One of the main goals outlined in The Surgeon General’s Call to Action to Prevent Skin Cancer is to reduce harm from indoor tanning, which has been linked to increased risk for skin cancer, including melanoma, basal cell carcinoma, and squamous cell carcinoma.
Based on reports from the American Cancer Society, Centers for Disease Control and Prevention, Federal Trade Commission, Mayo Clinic, and US Food and Drug Administration, the following common myths about indoor tanning should be communicated to dermatology patients.
Myth: Indoor tanning will not increase your risk for skin cancer.
Fact: As many as 90% of melanomas are caused by UV exposure. Indoor tanning exposure to UVA and UVB radiation damages the skin and may lead to cancer. Melanoma is linked to severe sunburns, especially at a young age.
Myth: Indoor tanning is safer than tanning outdoors because it is a controlled dose of UV radiation.
Fact: Both indoor tanning and tanning outside are dangerous. Tanning beds may be more dangerous than the sun because they can be used at the same high intensity every day of the year, regardless of time of day, season, or cloud cover. Furthermore, the Surgeon General and US Food and Drug Administration report that an estimated 3000 Americans each year go to emergency departments with injuries caused by indoor tanning, including burns, eye injuries, immune suppression, and allergic reactions. Indoor tanning also causes premature skin aging.
Myth: A “base tan” protects your skin from sunburn.
Fact: Although many patients believe that a few sessions of indoor tanning will prevent them from burning in the sun, a tan does little to protect the skin from future UV exposure. In fact, the Centers for Disease Control and Prevention notes that people who tan indoors are more likely to report getting sunburned. The best way to protect the skin from sunburn is by using sun protection and avoiding indoor tanning.
Myth: Indoor tanning is a safe way to increase vitamin D levels.
Fact: It is important to get enough vitamin D; however, the safest way is through what you eat. Although UVB radiation helps the body produce vitamin D, patients do not need a tan to get that benefit. Ten to 15 minutes of unprotected natural sun exposure on the face and hands 2 to 3 times a week during the summer allows for a healthy dose of vitamin D. Dietary sources, such as low-fat milk, salmon, tuna, and fortified orange juice, are the safest way to get enough vitamin D.
Myth: Indoor tanning is approved by the government.
Fact: According to the Federal Trade Commission, no US government agency recommends the use of indoor tanning equipment. Tanning bed use by minors has been banned in many states, and efforts are ongoing to protect consumers younger than 18 years on local, state, and federal levels. In July 2009, the International Agency for Research on Cancer, part of the World Health Organization, moved tanning devices that emit UV radiation into the highest cancer risk category—carcinogenic to humans—concluding that they are more dangerous than previously thought.
Studies have consistently shown that indoor tanning increases a person’s risk of getting skin cancer and indoor tanning at a young age appears to be more strongly related to lifetime skin cancer risk. Patients should be reminded that every time they tan, they increase their risk of melanoma as well as premature skin aging and other skin cancers. Dermatologists should counsel patients on using sun protection and avoiding indoor tanning.
The Surgeon General has called on partners in prevention from various sectors to address skin cancer as a major public health problem. One of the main goals outlined in The Surgeon General’s Call to Action to Prevent Skin Cancer is to reduce harm from indoor tanning, which has been linked to increased risk for skin cancer, including melanoma, basal cell carcinoma, and squamous cell carcinoma.
Based on reports from the American Cancer Society, Centers for Disease Control and Prevention, Federal Trade Commission, Mayo Clinic, and US Food and Drug Administration, the following common myths about indoor tanning should be communicated to dermatology patients.
Myth: Indoor tanning will not increase your risk for skin cancer.
Fact: As many as 90% of melanomas are caused by UV exposure. Indoor tanning exposure to UVA and UVB radiation damages the skin and may lead to cancer. Melanoma is linked to severe sunburns, especially at a young age.
Myth: Indoor tanning is safer than tanning outdoors because it is a controlled dose of UV radiation.
Fact: Both indoor tanning and tanning outside are dangerous. Tanning beds may be more dangerous than the sun because they can be used at the same high intensity every day of the year, regardless of time of day, season, or cloud cover. Furthermore, the Surgeon General and US Food and Drug Administration report that an estimated 3000 Americans each year go to emergency departments with injuries caused by indoor tanning, including burns, eye injuries, immune suppression, and allergic reactions. Indoor tanning also causes premature skin aging.
Myth: A “base tan” protects your skin from sunburn.
Fact: Although many patients believe that a few sessions of indoor tanning will prevent them from burning in the sun, a tan does little to protect the skin from future UV exposure. In fact, the Centers for Disease Control and Prevention notes that people who tan indoors are more likely to report getting sunburned. The best way to protect the skin from sunburn is by using sun protection and avoiding indoor tanning.
Myth: Indoor tanning is a safe way to increase vitamin D levels.
Fact: It is important to get enough vitamin D; however, the safest way is through what you eat. Although UVB radiation helps the body produce vitamin D, patients do not need a tan to get that benefit. Ten to 15 minutes of unprotected natural sun exposure on the face and hands 2 to 3 times a week during the summer allows for a healthy dose of vitamin D. Dietary sources, such as low-fat milk, salmon, tuna, and fortified orange juice, are the safest way to get enough vitamin D.
Myth: Indoor tanning is approved by the government.
Fact: According to the Federal Trade Commission, no US government agency recommends the use of indoor tanning equipment. Tanning bed use by minors has been banned in many states, and efforts are ongoing to protect consumers younger than 18 years on local, state, and federal levels. In July 2009, the International Agency for Research on Cancer, part of the World Health Organization, moved tanning devices that emit UV radiation into the highest cancer risk category—carcinogenic to humans—concluding that they are more dangerous than previously thought.
Studies have consistently shown that indoor tanning increases a person’s risk of getting skin cancer and indoor tanning at a young age appears to be more strongly related to lifetime skin cancer risk. Patients should be reminded that every time they tan, they increase their risk of melanoma as well as premature skin aging and other skin cancers. Dermatologists should counsel patients on using sun protection and avoiding indoor tanning.
The Surgeon General has called on partners in prevention from various sectors to address skin cancer as a major public health problem. One of the main goals outlined in The Surgeon General’s Call to Action to Prevent Skin Cancer is to reduce harm from indoor tanning, which has been linked to increased risk for skin cancer, including melanoma, basal cell carcinoma, and squamous cell carcinoma.
Based on reports from the American Cancer Society, Centers for Disease Control and Prevention, Federal Trade Commission, Mayo Clinic, and US Food and Drug Administration, the following common myths about indoor tanning should be communicated to dermatology patients.
Myth: Indoor tanning will not increase your risk for skin cancer.
Fact: As many as 90% of melanomas are caused by UV exposure. Indoor tanning exposure to UVA and UVB radiation damages the skin and may lead to cancer. Melanoma is linked to severe sunburns, especially at a young age.
Myth: Indoor tanning is safer than tanning outdoors because it is a controlled dose of UV radiation.
Fact: Both indoor tanning and tanning outside are dangerous. Tanning beds may be more dangerous than the sun because they can be used at the same high intensity every day of the year, regardless of time of day, season, or cloud cover. Furthermore, the Surgeon General and US Food and Drug Administration report that an estimated 3000 Americans each year go to emergency departments with injuries caused by indoor tanning, including burns, eye injuries, immune suppression, and allergic reactions. Indoor tanning also causes premature skin aging.
Myth: A “base tan” protects your skin from sunburn.
Fact: Although many patients believe that a few sessions of indoor tanning will prevent them from burning in the sun, a tan does little to protect the skin from future UV exposure. In fact, the Centers for Disease Control and Prevention notes that people who tan indoors are more likely to report getting sunburned. The best way to protect the skin from sunburn is by using sun protection and avoiding indoor tanning.
Myth: Indoor tanning is a safe way to increase vitamin D levels.
Fact: It is important to get enough vitamin D; however, the safest way is through what you eat. Although UVB radiation helps the body produce vitamin D, patients do not need a tan to get that benefit. Ten to 15 minutes of unprotected natural sun exposure on the face and hands 2 to 3 times a week during the summer allows for a healthy dose of vitamin D. Dietary sources, such as low-fat milk, salmon, tuna, and fortified orange juice, are the safest way to get enough vitamin D.
Myth: Indoor tanning is approved by the government.
Fact: According to the Federal Trade Commission, no US government agency recommends the use of indoor tanning equipment. Tanning bed use by minors has been banned in many states, and efforts are ongoing to protect consumers younger than 18 years on local, state, and federal levels. In July 2009, the International Agency for Research on Cancer, part of the World Health Organization, moved tanning devices that emit UV radiation into the highest cancer risk category—carcinogenic to humans—concluding that they are more dangerous than previously thought.
Studies have consistently shown that indoor tanning increases a person’s risk of getting skin cancer and indoor tanning at a young age appears to be more strongly related to lifetime skin cancer risk. Patients should be reminded that every time they tan, they increase their risk of melanoma as well as premature skin aging and other skin cancers. Dermatologists should counsel patients on using sun protection and avoiding indoor tanning.
How to Teach the Potassium Hydroxide Preparation: A Disappearing Clinical Art Form
Potassium hydroxide (KOH) preparations remain an important bedside test for prompt and accurate diagnosis of superficial fungal infections known as dermatophytoses. This tool has been used for at least 100 years, with early terminology referring to it as potash; for the last century, it has largely been a technique passed down as a skill from master technician to learning apprentice. The original pioneer of the KOH preparation remains a mystery.1
Variations on techniques for performing the KOH preparation exist, and tips and tricks on the use of this test are a hot topic among dermatologists.2 Although primary care and dermatology-specific publications espouse the importance of the KOH preparation,3,4 it has unfortunately been identified and labeled as one of the forgotten diagnostic tools.5
It is incumbent on dermatologists to educate medical students and residents using a simple and specific method to ensure that this simple and effective technique, with sensitivity reported between 87% and 91% depending on the expertise of the examiner,6 remains part of the clinical armamentarium. One concern in the instruction of large groups of students and clinicians is the ready accessibility or availability of viable skin samples. This article describes a method of collecting and storing skin samples that will allow educators to train large groups of students on performing KOH preparations without having to repeatedly seek skin samples or patients with superficial skin infections. A detailed description of the pedagogy used to teach the preparation and interpretation of KOH slides to a large group of students also is reviewed.
Specimen Collection
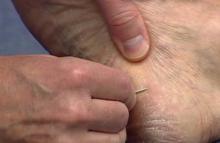
The first step in teaching the KOH preparation to a large group is the collection of a suitable number of skin scrapings from patients with a superficial fungal skin infection (eg, tinea corporis, tinea versicolor). A common technique for obtaining skin samples is to use a no. 15 scalpel blade (Figure 1) to scrape the scale of the lesion at its scaly border once the area is moistened with an alcohol pad or soap and water.7 The moisture from the alcohol pad allows the scale to stick to the no. 15 blade, facilitating collection. Once a suitable amount of scale is collected, it is placed on a glass microscope slide by smearing the scale from the blade onto the slide. This process has been modified to facilitate a larger quantity of specimen as follows: dermatophyte-infected plaques with scale are rubbed with the no. 15 blade and the free scale drops into a standard urine specimen cup. This process is repeated multiple times from different sites to capture the displaced scale with the dermatophyte. We have found that as long as the specimen cups are sealed tightly and stored in a relatively dry and cool environment (room temperature), the samples can be used to construct KOH teaching slides for at least 3 years. We have not used them beyond 3 years but suspect that they would continue to be viable after this time.
Preparation of Slides
Given that time for teaching often is limited, it is beneficial to fix many skin scrapings on a large number of glass slides prior to the session, which enables students to simply add KOH to the slides on the teaching day. To prepare the slides in advance, it is necessary to gather the following materials: a specimen cup with skin samples, glass slides, pickups or tweezers, a small pipette, a cup of water, protective gloves, and a pencil. After donning protective gloves, the pickups or tweezers are used to retrieve a few flakes of scale from the specimen cup and place them on the center of a glass slide. Using the pipette, 1 or 2 drops of water are added to the scale, and the slide is then allowed to dry. The slides are marked with the pencil to indicate the “up” side to prevent the students from applying KOH solution to the wrong side of the slide. The skin scale is fixed in place on the slide as the water evaporates and may be stored until needed for use in a standard slide box or folder.
Performing the KOH Preparation
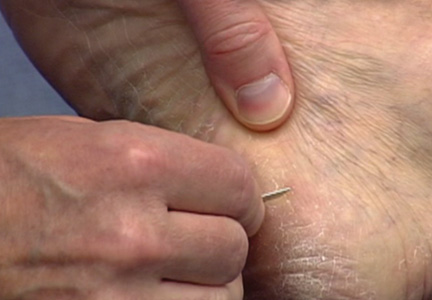
On the day of teaching, it is helpful to engage the entire group of students with an introductory lecture on the purpose and use of the KOH preparation. Upon completion, students move to a workstation with all of the materials needed to prepare the slide. Additional items needed at this time are 10% KOH solution, coverslips, and a heating device (eg, lighter, Bunsen burner, match)(optional). Students are instructed to place 1 or 2 skin scales onto a glass slide or retrieve a slide with skin scales already fixed, and then add 1 drop of 10% KOH solution directly to the sample (Figure 2). Next, they should place a slide coverslip onto the KOH drop and skin sample using a side-to-side technique that will move the scale into a thin layer within the KOH solution and push away any excess solution to the periphery (Figure 3). Large amounts of excess KOH solution should be cleared away with a paper towel, lens paper, or tissue. The heat source can be used to gently heat the underside of the glass slide (Figure 4), but it often is sufficient to simply wait 3 to 5 minutes for the KOH solution to take effect. The heat accelerates the maceration of the scale and makes it easier to see the hyphae among the keratinocytes. Some physicians advocate the use of dimethyl sulfoxide in lieu of heating,8 but this solution may not be available in all primary care settings.

| 
|
Microscopic Examination
Prior to examining the slides under the microscope, students may complete a self-guided tutorial (eg, digital or paper slide show) on the various features seen through the microscope that are indicative of dermatophytes, including branching hyphae and yeast buds. They also should be educated about the common appearance of artifacts that may resemble hyphae. Once the students have completed the tutorial, they may proceed to microscopic examination.
While the students are viewing their slides under the microscope, we find it helpful to have at least 1 experienced faculty member for every group of 10 students. This instructor should encourage the students to lower the microscope condenser all the way to facilitate better observation. Students should start with low power (×4 or red band) and scan for areas that are rich in skin scale. Once a collection of scale is found, the student can switch to higher power (×10 or yellow band) and start scanning for hyphae. Students should be reminded to search for filamentous and branching tubes that are refractile. The term refractile may be confusing to some students, so we explain that shifting the focus up or down will show the hyphae to change in brightness and may reveal a greenish tint. Another helpful indicator to point out is the feature that hyphae will cross the border of epidermal skin cells, whereas artifacts will not (Figure 5). Once the students have identified evidence of a dermatophyte infection, they must call the instructor to their station to verify the presence of hyphae or yeast buds, which helps confirm their understanding of the procedure. Once the student accurately identifies these items, the session is complete.
Comment
The use of a KOH preparation is a fast, simple, accurate, and cost-effective way to diagnose superficial fungal infections; however, because of insufficient familiarity with this tool, the technique often is replaced by initiation of empiric antifungal therapy in patients with suspected dermatophytosis. This empiric treatment has the potential to delay appropriate diagnosis and treatment (eg, in a patient with nummular dermatitis, which can clinically mimic tinea corporis). One way to encourage the use of the KOH preparation in the primary care and dermatologic setting is to educate large groups of next-generation physicians while in medical training. This article describes a teaching technique that allows for long-term storage of positive skin samples and a detailed description of the pedagogy used to train and educate a large group of students in a relatively short period of time.
All KOH preparations fall under the US federal government’s Clinical Laboratory Improvement Amendments and require proficiency testing.9 Although the teaching method presented here is designed for teaching medical students, it may be utilized to educate or refamiliarize experienced physicians with the procedure in an effort to improve proficiency in point-of-care testing programs used in many health care systems to comply with the Clinical Laboratories Improvement Amendments. Future analyses could assess whether the method described here improves provider performance on such proficiency measures and whether it ultimately helps ensure quality patient care.
1. Dasgupta T, Sahu J. Origins of the KOH technique. Clin Dermatol. 2012;2:238-242.
2. Stone S. Editor’s commentary. Clin Dermatol. 2012;2:241-242.
3. Monroe JR. The diagnostic value of a KOH. JAAPA. 2001;4:50-51.
4. Hainer BL. Dermatophyte infections. Am Fam Physician. 2003;1:101-109.
5. Ponka D, Baddar F. Microscopic potassium hydroxide preparation. Can Fam Physician. 2014;60:57.
6. Lilly KK, Koshnick RL, Grill JP, et al. Cost-effectiveness of diagnostic tests for toenail onychomycosis: a repeated-measure, single-blinded, cross-sectional evaluation of 7 diagnostic tests. J Am Acad Dermatol. 2006;4:620-626.
7. Bolognia JL, Jorizzo JL, Schaffer JV. Dermatology. 3rd ed. New York, NY: Elsevier Saunders; 2012.
8. James WD, Berger T, Elston D. Andrew’s Diseases of the Skin: Clinical Dermatology. 11th ed. New York, NY: Elsevier Saunders; 2011.
9. Clinical Laboratory Improvement Amendments (CLIA). Centers for Medicare & Medicaid Services Web site. https://www.cms.gov/Regulations-and-Guidance/Legislation/CLIA/index.html?redirect=/clia/. Updated June 6, 2015. Accessed July 21, 2015.
Potassium hydroxide (KOH) preparations remain an important bedside test for prompt and accurate diagnosis of superficial fungal infections known as dermatophytoses. This tool has been used for at least 100 years, with early terminology referring to it as potash; for the last century, it has largely been a technique passed down as a skill from master technician to learning apprentice. The original pioneer of the KOH preparation remains a mystery.1
Variations on techniques for performing the KOH preparation exist, and tips and tricks on the use of this test are a hot topic among dermatologists.2 Although primary care and dermatology-specific publications espouse the importance of the KOH preparation,3,4 it has unfortunately been identified and labeled as one of the forgotten diagnostic tools.5
It is incumbent on dermatologists to educate medical students and residents using a simple and specific method to ensure that this simple and effective technique, with sensitivity reported between 87% and 91% depending on the expertise of the examiner,6 remains part of the clinical armamentarium. One concern in the instruction of large groups of students and clinicians is the ready accessibility or availability of viable skin samples. This article describes a method of collecting and storing skin samples that will allow educators to train large groups of students on performing KOH preparations without having to repeatedly seek skin samples or patients with superficial skin infections. A detailed description of the pedagogy used to teach the preparation and interpretation of KOH slides to a large group of students also is reviewed.
Specimen Collection
The first step in teaching the KOH preparation to a large group is the collection of a suitable number of skin scrapings from patients with a superficial fungal skin infection (eg, tinea corporis, tinea versicolor). A common technique for obtaining skin samples is to use a no. 15 scalpel blade (Figure 1) to scrape the scale of the lesion at its scaly border once the area is moistened with an alcohol pad or soap and water.7 The moisture from the alcohol pad allows the scale to stick to the no. 15 blade, facilitating collection. Once a suitable amount of scale is collected, it is placed on a glass microscope slide by smearing the scale from the blade onto the slide. This process has been modified to facilitate a larger quantity of specimen as follows: dermatophyte-infected plaques with scale are rubbed with the no. 15 blade and the free scale drops into a standard urine specimen cup. This process is repeated multiple times from different sites to capture the displaced scale with the dermatophyte. We have found that as long as the specimen cups are sealed tightly and stored in a relatively dry and cool environment (room temperature), the samples can be used to construct KOH teaching slides for at least 3 years. We have not used them beyond 3 years but suspect that they would continue to be viable after this time.
Preparation of Slides
Given that time for teaching often is limited, it is beneficial to fix many skin scrapings on a large number of glass slides prior to the session, which enables students to simply add KOH to the slides on the teaching day. To prepare the slides in advance, it is necessary to gather the following materials: a specimen cup with skin samples, glass slides, pickups or tweezers, a small pipette, a cup of water, protective gloves, and a pencil. After donning protective gloves, the pickups or tweezers are used to retrieve a few flakes of scale from the specimen cup and place them on the center of a glass slide. Using the pipette, 1 or 2 drops of water are added to the scale, and the slide is then allowed to dry. The slides are marked with the pencil to indicate the “up” side to prevent the students from applying KOH solution to the wrong side of the slide. The skin scale is fixed in place on the slide as the water evaporates and may be stored until needed for use in a standard slide box or folder.
Performing the KOH Preparation
On the day of teaching, it is helpful to engage the entire group of students with an introductory lecture on the purpose and use of the KOH preparation. Upon completion, students move to a workstation with all of the materials needed to prepare the slide. Additional items needed at this time are 10% KOH solution, coverslips, and a heating device (eg, lighter, Bunsen burner, match)(optional). Students are instructed to place 1 or 2 skin scales onto a glass slide or retrieve a slide with skin scales already fixed, and then add 1 drop of 10% KOH solution directly to the sample (Figure 2). Next, they should place a slide coverslip onto the KOH drop and skin sample using a side-to-side technique that will move the scale into a thin layer within the KOH solution and push away any excess solution to the periphery (Figure 3). Large amounts of excess KOH solution should be cleared away with a paper towel, lens paper, or tissue. The heat source can be used to gently heat the underside of the glass slide (Figure 4), but it often is sufficient to simply wait 3 to 5 minutes for the KOH solution to take effect. The heat accelerates the maceration of the scale and makes it easier to see the hyphae among the keratinocytes. Some physicians advocate the use of dimethyl sulfoxide in lieu of heating,8 but this solution may not be available in all primary care settings.

| 
|
Microscopic Examination
Prior to examining the slides under the microscope, students may complete a self-guided tutorial (eg, digital or paper slide show) on the various features seen through the microscope that are indicative of dermatophytes, including branching hyphae and yeast buds. They also should be educated about the common appearance of artifacts that may resemble hyphae. Once the students have completed the tutorial, they may proceed to microscopic examination.
While the students are viewing their slides under the microscope, we find it helpful to have at least 1 experienced faculty member for every group of 10 students. This instructor should encourage the students to lower the microscope condenser all the way to facilitate better observation. Students should start with low power (×4 or red band) and scan for areas that are rich in skin scale. Once a collection of scale is found, the student can switch to higher power (×10 or yellow band) and start scanning for hyphae. Students should be reminded to search for filamentous and branching tubes that are refractile. The term refractile may be confusing to some students, so we explain that shifting the focus up or down will show the hyphae to change in brightness and may reveal a greenish tint. Another helpful indicator to point out is the feature that hyphae will cross the border of epidermal skin cells, whereas artifacts will not (Figure 5). Once the students have identified evidence of a dermatophyte infection, they must call the instructor to their station to verify the presence of hyphae or yeast buds, which helps confirm their understanding of the procedure. Once the student accurately identifies these items, the session is complete.
Comment
The use of a KOH preparation is a fast, simple, accurate, and cost-effective way to diagnose superficial fungal infections; however, because of insufficient familiarity with this tool, the technique often is replaced by initiation of empiric antifungal therapy in patients with suspected dermatophytosis. This empiric treatment has the potential to delay appropriate diagnosis and treatment (eg, in a patient with nummular dermatitis, which can clinically mimic tinea corporis). One way to encourage the use of the KOH preparation in the primary care and dermatologic setting is to educate large groups of next-generation physicians while in medical training. This article describes a teaching technique that allows for long-term storage of positive skin samples and a detailed description of the pedagogy used to train and educate a large group of students in a relatively short period of time.
All KOH preparations fall under the US federal government’s Clinical Laboratory Improvement Amendments and require proficiency testing.9 Although the teaching method presented here is designed for teaching medical students, it may be utilized to educate or refamiliarize experienced physicians with the procedure in an effort to improve proficiency in point-of-care testing programs used in many health care systems to comply with the Clinical Laboratories Improvement Amendments. Future analyses could assess whether the method described here improves provider performance on such proficiency measures and whether it ultimately helps ensure quality patient care.
Potassium hydroxide (KOH) preparations remain an important bedside test for prompt and accurate diagnosis of superficial fungal infections known as dermatophytoses. This tool has been used for at least 100 years, with early terminology referring to it as potash; for the last century, it has largely been a technique passed down as a skill from master technician to learning apprentice. The original pioneer of the KOH preparation remains a mystery.1
Variations on techniques for performing the KOH preparation exist, and tips and tricks on the use of this test are a hot topic among dermatologists.2 Although primary care and dermatology-specific publications espouse the importance of the KOH preparation,3,4 it has unfortunately been identified and labeled as one of the forgotten diagnostic tools.5
It is incumbent on dermatologists to educate medical students and residents using a simple and specific method to ensure that this simple and effective technique, with sensitivity reported between 87% and 91% depending on the expertise of the examiner,6 remains part of the clinical armamentarium. One concern in the instruction of large groups of students and clinicians is the ready accessibility or availability of viable skin samples. This article describes a method of collecting and storing skin samples that will allow educators to train large groups of students on performing KOH preparations without having to repeatedly seek skin samples or patients with superficial skin infections. A detailed description of the pedagogy used to teach the preparation and interpretation of KOH slides to a large group of students also is reviewed.
Specimen Collection
The first step in teaching the KOH preparation to a large group is the collection of a suitable number of skin scrapings from patients with a superficial fungal skin infection (eg, tinea corporis, tinea versicolor). A common technique for obtaining skin samples is to use a no. 15 scalpel blade (Figure 1) to scrape the scale of the lesion at its scaly border once the area is moistened with an alcohol pad or soap and water.7 The moisture from the alcohol pad allows the scale to stick to the no. 15 blade, facilitating collection. Once a suitable amount of scale is collected, it is placed on a glass microscope slide by smearing the scale from the blade onto the slide. This process has been modified to facilitate a larger quantity of specimen as follows: dermatophyte-infected plaques with scale are rubbed with the no. 15 blade and the free scale drops into a standard urine specimen cup. This process is repeated multiple times from different sites to capture the displaced scale with the dermatophyte. We have found that as long as the specimen cups are sealed tightly and stored in a relatively dry and cool environment (room temperature), the samples can be used to construct KOH teaching slides for at least 3 years. We have not used them beyond 3 years but suspect that they would continue to be viable after this time.
Preparation of Slides
Given that time for teaching often is limited, it is beneficial to fix many skin scrapings on a large number of glass slides prior to the session, which enables students to simply add KOH to the slides on the teaching day. To prepare the slides in advance, it is necessary to gather the following materials: a specimen cup with skin samples, glass slides, pickups or tweezers, a small pipette, a cup of water, protective gloves, and a pencil. After donning protective gloves, the pickups or tweezers are used to retrieve a few flakes of scale from the specimen cup and place them on the center of a glass slide. Using the pipette, 1 or 2 drops of water are added to the scale, and the slide is then allowed to dry. The slides are marked with the pencil to indicate the “up” side to prevent the students from applying KOH solution to the wrong side of the slide. The skin scale is fixed in place on the slide as the water evaporates and may be stored until needed for use in a standard slide box or folder.
Performing the KOH Preparation
On the day of teaching, it is helpful to engage the entire group of students with an introductory lecture on the purpose and use of the KOH preparation. Upon completion, students move to a workstation with all of the materials needed to prepare the slide. Additional items needed at this time are 10% KOH solution, coverslips, and a heating device (eg, lighter, Bunsen burner, match)(optional). Students are instructed to place 1 or 2 skin scales onto a glass slide or retrieve a slide with skin scales already fixed, and then add 1 drop of 10% KOH solution directly to the sample (Figure 2). Next, they should place a slide coverslip onto the KOH drop and skin sample using a side-to-side technique that will move the scale into a thin layer within the KOH solution and push away any excess solution to the periphery (Figure 3). Large amounts of excess KOH solution should be cleared away with a paper towel, lens paper, or tissue. The heat source can be used to gently heat the underside of the glass slide (Figure 4), but it often is sufficient to simply wait 3 to 5 minutes for the KOH solution to take effect. The heat accelerates the maceration of the scale and makes it easier to see the hyphae among the keratinocytes. Some physicians advocate the use of dimethyl sulfoxide in lieu of heating,8 but this solution may not be available in all primary care settings.

| 
|
Microscopic Examination
Prior to examining the slides under the microscope, students may complete a self-guided tutorial (eg, digital or paper slide show) on the various features seen through the microscope that are indicative of dermatophytes, including branching hyphae and yeast buds. They also should be educated about the common appearance of artifacts that may resemble hyphae. Once the students have completed the tutorial, they may proceed to microscopic examination.
While the students are viewing their slides under the microscope, we find it helpful to have at least 1 experienced faculty member for every group of 10 students. This instructor should encourage the students to lower the microscope condenser all the way to facilitate better observation. Students should start with low power (×4 or red band) and scan for areas that are rich in skin scale. Once a collection of scale is found, the student can switch to higher power (×10 or yellow band) and start scanning for hyphae. Students should be reminded to search for filamentous and branching tubes that are refractile. The term refractile may be confusing to some students, so we explain that shifting the focus up or down will show the hyphae to change in brightness and may reveal a greenish tint. Another helpful indicator to point out is the feature that hyphae will cross the border of epidermal skin cells, whereas artifacts will not (Figure 5). Once the students have identified evidence of a dermatophyte infection, they must call the instructor to their station to verify the presence of hyphae or yeast buds, which helps confirm their understanding of the procedure. Once the student accurately identifies these items, the session is complete.
Comment
The use of a KOH preparation is a fast, simple, accurate, and cost-effective way to diagnose superficial fungal infections; however, because of insufficient familiarity with this tool, the technique often is replaced by initiation of empiric antifungal therapy in patients with suspected dermatophytosis. This empiric treatment has the potential to delay appropriate diagnosis and treatment (eg, in a patient with nummular dermatitis, which can clinically mimic tinea corporis). One way to encourage the use of the KOH preparation in the primary care and dermatologic setting is to educate large groups of next-generation physicians while in medical training. This article describes a teaching technique that allows for long-term storage of positive skin samples and a detailed description of the pedagogy used to train and educate a large group of students in a relatively short period of time.
All KOH preparations fall under the US federal government’s Clinical Laboratory Improvement Amendments and require proficiency testing.9 Although the teaching method presented here is designed for teaching medical students, it may be utilized to educate or refamiliarize experienced physicians with the procedure in an effort to improve proficiency in point-of-care testing programs used in many health care systems to comply with the Clinical Laboratories Improvement Amendments. Future analyses could assess whether the method described here improves provider performance on such proficiency measures and whether it ultimately helps ensure quality patient care.
1. Dasgupta T, Sahu J. Origins of the KOH technique. Clin Dermatol. 2012;2:238-242.
2. Stone S. Editor’s commentary. Clin Dermatol. 2012;2:241-242.
3. Monroe JR. The diagnostic value of a KOH. JAAPA. 2001;4:50-51.
4. Hainer BL. Dermatophyte infections. Am Fam Physician. 2003;1:101-109.
5. Ponka D, Baddar F. Microscopic potassium hydroxide preparation. Can Fam Physician. 2014;60:57.
6. Lilly KK, Koshnick RL, Grill JP, et al. Cost-effectiveness of diagnostic tests for toenail onychomycosis: a repeated-measure, single-blinded, cross-sectional evaluation of 7 diagnostic tests. J Am Acad Dermatol. 2006;4:620-626.
7. Bolognia JL, Jorizzo JL, Schaffer JV. Dermatology. 3rd ed. New York, NY: Elsevier Saunders; 2012.
8. James WD, Berger T, Elston D. Andrew’s Diseases of the Skin: Clinical Dermatology. 11th ed. New York, NY: Elsevier Saunders; 2011.
9. Clinical Laboratory Improvement Amendments (CLIA). Centers for Medicare & Medicaid Services Web site. https://www.cms.gov/Regulations-and-Guidance/Legislation/CLIA/index.html?redirect=/clia/. Updated June 6, 2015. Accessed July 21, 2015.
1. Dasgupta T, Sahu J. Origins of the KOH technique. Clin Dermatol. 2012;2:238-242.
2. Stone S. Editor’s commentary. Clin Dermatol. 2012;2:241-242.
3. Monroe JR. The diagnostic value of a KOH. JAAPA. 2001;4:50-51.
4. Hainer BL. Dermatophyte infections. Am Fam Physician. 2003;1:101-109.
5. Ponka D, Baddar F. Microscopic potassium hydroxide preparation. Can Fam Physician. 2014;60:57.
6. Lilly KK, Koshnick RL, Grill JP, et al. Cost-effectiveness of diagnostic tests for toenail onychomycosis: a repeated-measure, single-blinded, cross-sectional evaluation of 7 diagnostic tests. J Am Acad Dermatol. 2006;4:620-626.
7. Bolognia JL, Jorizzo JL, Schaffer JV. Dermatology. 3rd ed. New York, NY: Elsevier Saunders; 2012.
8. James WD, Berger T, Elston D. Andrew’s Diseases of the Skin: Clinical Dermatology. 11th ed. New York, NY: Elsevier Saunders; 2011.
9. Clinical Laboratory Improvement Amendments (CLIA). Centers for Medicare & Medicaid Services Web site. https://www.cms.gov/Regulations-and-Guidance/Legislation/CLIA/index.html?redirect=/clia/. Updated June 6, 2015. Accessed July 21, 2015.
Practice Points
- Potassium hydroxide (KOH) preparations can lead to diagnostic confidence and direct appropriate therapy.
- Refreshing the basics of this simple technique can lead to better patient outcomes in the primary care setting and in the dermatology specialty clinic.
- Teaching the KOH preparation to the next generation of physicians will ensure its longevity and assure future benefit to patients.
Evaluating Endoleaks in the Dermatology Office
Endoleaks are common complications following endovascular aneurysm repairs (EVARs) that may occur any time after surgery. There are 5 types of endoleaks with various etiologies. A type V endoleak (also known as endotension) is not considered a true endoleak but instead is characterized by continued aneurysm expansion without a leak, which is demonstrated via imaging tests.1 Type V endoleaks typically require open aneurysm repair.2 We report the case of a 69-year-old woman who presented to our dermatology office for treatment of a suspected lipoma overlying the right mid sternum that was confirmed to be a type V endoleak via computed tomography angiography.
Case Report
A 69-year-old woman was referred to our dermatology office by her primary care physician for evaluation of a subcutaneous mass overlying the right mid sternum, which was a suspected lipoma. The patient reported that the mass had been present for approximately 2 weeks and was enlarging but otherwise asymptomatic. Her medical history was remarkable for hypertension, an ascending aortic aneurysm, and a subsequent aortic valve replacement approximately 2.5 years prior. Her current medications included amlodipine, lisinopril, nebivolol, ibuprofen, and aspirin. She denied use of alcohol, tobacco, or illicit drugs. A review of systems was noncontributory.
Physical examination revealed a single 3.5×4.5-cm, soft, nonmobile subcutaneous mass located at the site of the thoracotomy scar (Figure 1). The mass appeared to have a central attachment to the sternum. No erythema, swelling, or exudate was noted, and the patient denied tenderness on palpation. The diagnosis of lipoma was questioned, and the patient was referred for ultrasonography and computed tomography angiography. Ultrasonography showed a nonspecific chest wall mass with internal blood flow, and computed tomography angiography showed a large, low-attenuation collection of blood around the entire circumference of the ascending aorta, extending from the aortic root to the arch of the aorta. There was extension of the collection of blood through either the sternocostal junction or the sternotomy defect into the subcutaneous tissue anterior to the sternum (Figure 2). Findings were most consistent with a type V endoleak, and the patient was referred to a cardiothoracic surgeon for treatment. We later learned that our patient died during surgery attempting to repair the aneurysm approximately 2 weeks after her presentation to our office.
Comment
An endoleak is a common complication following an EVAR that is characterized by persistent blood flow within the aneurysm sac. Endoleaks have been described as the Achilles’ heel of EVARs.1 The goal of an EVAR is to create a complete seal so that the flow of blood completely excludes the aneurysm, thus ultimately preventing an aneurysm rupture. An endoleak results when there is failure to obtain a complete seal due to a variety of different mechanisms. White et al3 first described and classified endoleaks in 1997. The initial terminology used to classify endoleaks was based on timing (primary or secondary/late) and location (graft related/perigraft or non–graft related/retrograde). Today, endoleaks are classified into 5 types, 3 of which are considered true endoleaks and 2 of which are not.4 Type I endoleaks result from a failure to create an adequate seal at one of the attachments of the graft to the vessel wall. Type II endoleaks are due to retrograde flow through collateral vessels into the aneurysm sac. They are much more common than type I, occurring in 10% to 25% of abdominal endograft cases. The last true endoleak, type III, occurs due to device failure in the form of disjunction of the components of the graft system (type IIIa) or a defect in the graft fabric (type IIIb). Type IV and type V endoleaks are not considered to be true endoleaks. Type IV endoleaks are due to the porosity of the graft material and have virtually been eliminated by changes in graft materials to decrease porosity. Type V endoleaks are characterized by continued blood flow into the aneurysm without any evidence of a leak on any imaging modality. Type V endoleaks are poorly understood but are believed to be due to pulsation of the graft wall, which is transmitted through the perivascular space to the aneurysm wall.4

|
Treatment of type V endoleaks is controversial. It is important to characterize the endoleak by various imaging modalities, and if a type V endoleak is confirmed, an open aneurysm repair often is required.2 A case of nonsurgical management of a type V endoleak has been described but is rare.5 In this case, the patient was referred to our dermatology office by her primary care physician for what appeared to be a benign lipoma, but it proved to be a type V endoleak on further examination. It is imperative that dermatologists are aware of endoleaks as common complications of EVARs, as they can be life threatening and usually require surgical intervention.
Conclusion
Endoleaks are common complications of EVARs. Dermatologists may encounter endoleaks that have been misdiagnosed as benign subcutaneous masses such as lipomas. It is imperative that dermatologists are aware of endoleaks, and patients who present with subcutaneous thoracic masses with a history of aneurysm repair require imaging, including computed tomography angiography, and referral to a cardiothoracic surgeon if appropriate.
1. Rosen RJ, Green RM. Endoleak management following endovascular aneurysm repair. J Vasc Interv Radiol. 2008;19(suppl 6):S37-S43.
2. Stavropoulos SW, Charagundla SR. Imaging techniques for detection and management of endoleaks after endovascular aortic aneurysm repair. Radiology. 2007;243:641-655.
3. White GH, Yu W, May J, et al. Endoleak as a complication of endoluminal grafting of abdominal aortic aneurysms: classification, incidence, diagnosis and management. J Endovascular Surg. 1997;4:152-168.
4. Veith FJ, Baum BA, Ohki T, et al. Nature and significance of endoleaks and endotension: summary of opinions expressed at an international conference. J Vasc Surg. 2002;35:1029-1035.
5. Mennander A, Pimenoff G, Heikkinen M, et al. Nonoperative approach to endotension. J Vasc Surg. 2005;42:194-198.
Endoleaks are common complications following endovascular aneurysm repairs (EVARs) that may occur any time after surgery. There are 5 types of endoleaks with various etiologies. A type V endoleak (also known as endotension) is not considered a true endoleak but instead is characterized by continued aneurysm expansion without a leak, which is demonstrated via imaging tests.1 Type V endoleaks typically require open aneurysm repair.2 We report the case of a 69-year-old woman who presented to our dermatology office for treatment of a suspected lipoma overlying the right mid sternum that was confirmed to be a type V endoleak via computed tomography angiography.
Case Report
A 69-year-old woman was referred to our dermatology office by her primary care physician for evaluation of a subcutaneous mass overlying the right mid sternum, which was a suspected lipoma. The patient reported that the mass had been present for approximately 2 weeks and was enlarging but otherwise asymptomatic. Her medical history was remarkable for hypertension, an ascending aortic aneurysm, and a subsequent aortic valve replacement approximately 2.5 years prior. Her current medications included amlodipine, lisinopril, nebivolol, ibuprofen, and aspirin. She denied use of alcohol, tobacco, or illicit drugs. A review of systems was noncontributory.
Physical examination revealed a single 3.5×4.5-cm, soft, nonmobile subcutaneous mass located at the site of the thoracotomy scar (Figure 1). The mass appeared to have a central attachment to the sternum. No erythema, swelling, or exudate was noted, and the patient denied tenderness on palpation. The diagnosis of lipoma was questioned, and the patient was referred for ultrasonography and computed tomography angiography. Ultrasonography showed a nonspecific chest wall mass with internal blood flow, and computed tomography angiography showed a large, low-attenuation collection of blood around the entire circumference of the ascending aorta, extending from the aortic root to the arch of the aorta. There was extension of the collection of blood through either the sternocostal junction or the sternotomy defect into the subcutaneous tissue anterior to the sternum (Figure 2). Findings were most consistent with a type V endoleak, and the patient was referred to a cardiothoracic surgeon for treatment. We later learned that our patient died during surgery attempting to repair the aneurysm approximately 2 weeks after her presentation to our office.
Comment
An endoleak is a common complication following an EVAR that is characterized by persistent blood flow within the aneurysm sac. Endoleaks have been described as the Achilles’ heel of EVARs.1 The goal of an EVAR is to create a complete seal so that the flow of blood completely excludes the aneurysm, thus ultimately preventing an aneurysm rupture. An endoleak results when there is failure to obtain a complete seal due to a variety of different mechanisms. White et al3 first described and classified endoleaks in 1997. The initial terminology used to classify endoleaks was based on timing (primary or secondary/late) and location (graft related/perigraft or non–graft related/retrograde). Today, endoleaks are classified into 5 types, 3 of which are considered true endoleaks and 2 of which are not.4 Type I endoleaks result from a failure to create an adequate seal at one of the attachments of the graft to the vessel wall. Type II endoleaks are due to retrograde flow through collateral vessels into the aneurysm sac. They are much more common than type I, occurring in 10% to 25% of abdominal endograft cases. The last true endoleak, type III, occurs due to device failure in the form of disjunction of the components of the graft system (type IIIa) or a defect in the graft fabric (type IIIb). Type IV and type V endoleaks are not considered to be true endoleaks. Type IV endoleaks are due to the porosity of the graft material and have virtually been eliminated by changes in graft materials to decrease porosity. Type V endoleaks are characterized by continued blood flow into the aneurysm without any evidence of a leak on any imaging modality. Type V endoleaks are poorly understood but are believed to be due to pulsation of the graft wall, which is transmitted through the perivascular space to the aneurysm wall.4


|
Treatment of type V endoleaks is controversial. It is important to characterize the endoleak by various imaging modalities, and if a type V endoleak is confirmed, an open aneurysm repair often is required.2 A case of nonsurgical management of a type V endoleak has been described but is rare.5 In this case, the patient was referred to our dermatology office by her primary care physician for what appeared to be a benign lipoma, but it proved to be a type V endoleak on further examination. It is imperative that dermatologists are aware of endoleaks as common complications of EVARs, as they can be life threatening and usually require surgical intervention.
Conclusion
Endoleaks are common complications of EVARs. Dermatologists may encounter endoleaks that have been misdiagnosed as benign subcutaneous masses such as lipomas. It is imperative that dermatologists are aware of endoleaks, and patients who present with subcutaneous thoracic masses with a history of aneurysm repair require imaging, including computed tomography angiography, and referral to a cardiothoracic surgeon if appropriate.
Endoleaks are common complications following endovascular aneurysm repairs (EVARs) that may occur any time after surgery. There are 5 types of endoleaks with various etiologies. A type V endoleak (also known as endotension) is not considered a true endoleak but instead is characterized by continued aneurysm expansion without a leak, which is demonstrated via imaging tests.1 Type V endoleaks typically require open aneurysm repair.2 We report the case of a 69-year-old woman who presented to our dermatology office for treatment of a suspected lipoma overlying the right mid sternum that was confirmed to be a type V endoleak via computed tomography angiography.
Case Report
A 69-year-old woman was referred to our dermatology office by her primary care physician for evaluation of a subcutaneous mass overlying the right mid sternum, which was a suspected lipoma. The patient reported that the mass had been present for approximately 2 weeks and was enlarging but otherwise asymptomatic. Her medical history was remarkable for hypertension, an ascending aortic aneurysm, and a subsequent aortic valve replacement approximately 2.5 years prior. Her current medications included amlodipine, lisinopril, nebivolol, ibuprofen, and aspirin. She denied use of alcohol, tobacco, or illicit drugs. A review of systems was noncontributory.
Physical examination revealed a single 3.5×4.5-cm, soft, nonmobile subcutaneous mass located at the site of the thoracotomy scar (Figure 1). The mass appeared to have a central attachment to the sternum. No erythema, swelling, or exudate was noted, and the patient denied tenderness on palpation. The diagnosis of lipoma was questioned, and the patient was referred for ultrasonography and computed tomography angiography. Ultrasonography showed a nonspecific chest wall mass with internal blood flow, and computed tomography angiography showed a large, low-attenuation collection of blood around the entire circumference of the ascending aorta, extending from the aortic root to the arch of the aorta. There was extension of the collection of blood through either the sternocostal junction or the sternotomy defect into the subcutaneous tissue anterior to the sternum (Figure 2). Findings were most consistent with a type V endoleak, and the patient was referred to a cardiothoracic surgeon for treatment. We later learned that our patient died during surgery attempting to repair the aneurysm approximately 2 weeks after her presentation to our office.
Comment
An endoleak is a common complication following an EVAR that is characterized by persistent blood flow within the aneurysm sac. Endoleaks have been described as the Achilles’ heel of EVARs.1 The goal of an EVAR is to create a complete seal so that the flow of blood completely excludes the aneurysm, thus ultimately preventing an aneurysm rupture. An endoleak results when there is failure to obtain a complete seal due to a variety of different mechanisms. White et al3 first described and classified endoleaks in 1997. The initial terminology used to classify endoleaks was based on timing (primary or secondary/late) and location (graft related/perigraft or non–graft related/retrograde). Today, endoleaks are classified into 5 types, 3 of which are considered true endoleaks and 2 of which are not.4 Type I endoleaks result from a failure to create an adequate seal at one of the attachments of the graft to the vessel wall. Type II endoleaks are due to retrograde flow through collateral vessels into the aneurysm sac. They are much more common than type I, occurring in 10% to 25% of abdominal endograft cases. The last true endoleak, type III, occurs due to device failure in the form of disjunction of the components of the graft system (type IIIa) or a defect in the graft fabric (type IIIb). Type IV and type V endoleaks are not considered to be true endoleaks. Type IV endoleaks are due to the porosity of the graft material and have virtually been eliminated by changes in graft materials to decrease porosity. Type V endoleaks are characterized by continued blood flow into the aneurysm without any evidence of a leak on any imaging modality. Type V endoleaks are poorly understood but are believed to be due to pulsation of the graft wall, which is transmitted through the perivascular space to the aneurysm wall.4


|
Treatment of type V endoleaks is controversial. It is important to characterize the endoleak by various imaging modalities, and if a type V endoleak is confirmed, an open aneurysm repair often is required.2 A case of nonsurgical management of a type V endoleak has been described but is rare.5 In this case, the patient was referred to our dermatology office by her primary care physician for what appeared to be a benign lipoma, but it proved to be a type V endoleak on further examination. It is imperative that dermatologists are aware of endoleaks as common complications of EVARs, as they can be life threatening and usually require surgical intervention.
Conclusion
Endoleaks are common complications of EVARs. Dermatologists may encounter endoleaks that have been misdiagnosed as benign subcutaneous masses such as lipomas. It is imperative that dermatologists are aware of endoleaks, and patients who present with subcutaneous thoracic masses with a history of aneurysm repair require imaging, including computed tomography angiography, and referral to a cardiothoracic surgeon if appropriate.
1. Rosen RJ, Green RM. Endoleak management following endovascular aneurysm repair. J Vasc Interv Radiol. 2008;19(suppl 6):S37-S43.
2. Stavropoulos SW, Charagundla SR. Imaging techniques for detection and management of endoleaks after endovascular aortic aneurysm repair. Radiology. 2007;243:641-655.
3. White GH, Yu W, May J, et al. Endoleak as a complication of endoluminal grafting of abdominal aortic aneurysms: classification, incidence, diagnosis and management. J Endovascular Surg. 1997;4:152-168.
4. Veith FJ, Baum BA, Ohki T, et al. Nature and significance of endoleaks and endotension: summary of opinions expressed at an international conference. J Vasc Surg. 2002;35:1029-1035.
5. Mennander A, Pimenoff G, Heikkinen M, et al. Nonoperative approach to endotension. J Vasc Surg. 2005;42:194-198.
1. Rosen RJ, Green RM. Endoleak management following endovascular aneurysm repair. J Vasc Interv Radiol. 2008;19(suppl 6):S37-S43.
2. Stavropoulos SW, Charagundla SR. Imaging techniques for detection and management of endoleaks after endovascular aortic aneurysm repair. Radiology. 2007;243:641-655.
3. White GH, Yu W, May J, et al. Endoleak as a complication of endoluminal grafting of abdominal aortic aneurysms: classification, incidence, diagnosis and management. J Endovascular Surg. 1997;4:152-168.
4. Veith FJ, Baum BA, Ohki T, et al. Nature and significance of endoleaks and endotension: summary of opinions expressed at an international conference. J Vasc Surg. 2002;35:1029-1035.
5. Mennander A, Pimenoff G, Heikkinen M, et al. Nonoperative approach to endotension. J Vasc Surg. 2005;42:194-198.
Practice Points
- An endoleak should be considered in any patient with a thoracic subcutaneous mass and history of aneurysm repair.
- Order imaging when an endoleak is suspected, including computed tomography angiography. Endoleaks can result in substantial morbidity and mortality if they are not recognized and treated appropriately.
- Dermatologists should be familiar with and able to recognize endoleaks, as patients may present to a dermatologist for evaluation of a subcutaneous mass that proves to be an endoleak.
Update on Hyaluronic Acid Fillers for Facial Rejuvenation
Facial rejuvenation has become increasingly popular, with nonsurgical and noninvasive procedures comprising a large part of aesthetic practice. According to the American Society for Aesthetic Plastic Surgery, Americans spent $12 billion on cosmetic procedures in 2014, with more than 10 million surgical and nonsurgical procedures performed. The top 5 nonsurgical procedures for both men and women combined were botulinum toxin, hyaluronic acid (HA), hair removal, chemical peel, and microdermabrasion.1
The first dermal filler used was bovine collagen, which was approved by the US Food and Drug Administration (FDA) in 1981. Despite its efficacy in the correction of facial rhytides, bovine collagen required allergy testing prior to use and was discontinued in 2010. Dermal fillers have evolved over the years, and newer products that are superior to earlier fillers with regard to longevity, safety, and tolerability and that do not require allergy testing have become available; however, advances in the use of dermal fillers are not only related to the development of newer products but also to evolving injection techniques. Initially, the aim of treatment with dermal fillers was to correct lines and wrinkles, but an increased understanding of the complex changes that occur with aging have changed our approach to one of volume replacement, with an emphasis on volume restoration in the midface. This approach requires an in-depth understanding of facial anatomy as well as the interactions of the skin, soft tissue, muscle, and bone. Furthermore, placement of filler in specific fat compartments can provide a more natural appearance and an all-around youthful face.2 In this article, we discuss HA fillers that have gained FDA approval within the last 5 years (Table).
Overview of HA Fillers
Hyaluronic acid is a naturally occurring linear glycosaminoglycan with a disaccharide unit, which repeats several thousand times.3 Hyaluronic acid is an essential part of the extracellular matrix of many tissues including the dermis and plays an important role in tissue growth, development, and wound healing. Hyaluronic acid is hygroscopic and absorbs water extensively, thus creating volume.4 Treatment with HA fillers is popular, as they are biocompatible and have a low potential for allergic reactions. They also are easy to use and reversible.4 The first HA filler to gain FDA approval was Restylane (Galderma Laboratories, LP). Currently, several HA fillers are approved in the United States, and each product differs from the others in polymer chain length, degree of HA concentration, particle size, gel consistency, gel hardness, gel viscosity, and degree of water solubility, as well as amount and degree of cross-linking. Cross-linking is essential to avoid enzymatic degradation by endogenous hyaluronidase when injected into the skin and thus to prolong the product’s half-life.5 Cross-linkers used to manufacture HA fillers include 1,4-butanediol diglycidyl ether and divinyl sulfone. More concentrated products with a greater degree of cross-linking provide increased longevity, but they are associated with a higher risk for inflammation and nodule formation. The elastic modulus (G′) is a measure of the firmness of dermal fillers, describing their resistance to deformation. Materials with a higher G′ are stiffer and are meant for deeper injections. Hyaluronic acid fillers can be further classified as biphasic or monophasic. Biphasic fillers (eg, Restylane, Perlane [Galderma Laboratories, LP]) contain a range of microsphere sizes, while monophasic fillers (eg, Juvéderm [Allergan, Inc], Belotero Balance [Merz North America, Inc]) contain homogeneous microspheres. Although randomized clinical trials have reported comparable efficacy and durability of biphasic and monophasic fillers when used to treat the nasolabial folds,6-8 monophasic HA fillers are more cohesive and may not migrate as much following injection.
Restylane Family
Restylane was the first FDA-approved HA filler, gaining its approval in 2003. Restylane is a nonanimal stabilized HA (NASHA) that is produced from the fermentation of equine streptococci. It is cross-linked with 1,4-butanediol diglycidyl ether with a 1% degree of cross-linking. Restylane has an HA concentration of 20 mg/mL. The particle size range of Restylane and Restylane-L is 330 to 430 mm. Restylane (and also Perlane) get passed through sizing screens via sieves and are quantified by their size. The longevity of HA fillers is approximately 6 months; however, various factors affect the product’s longevity, such as the degree of cross-linking, treatment area, and the patient’s metabolism. Restylane-L, which was FDA approved in 2012, is a newer product with 0.3% lidocaine incorporated into the syringe itself. It was the first product from the Restylane range to be approved for lip augmentation. The addition of lidocaine (designated by the L in the product name) does not affect the longevity of a filler.9
The newest FDA-approved HA filler was Restylane Silk (approved in 2014), which has been specifically designed for lip augmentation and correction of perioral rhytides. To avoid postprocedural swelling, it generally is recommended that Restylane Silk be injected slowly. If required, a short course of oral prednisone may be administered after the procedure to treat any edema. Restylane Silk is less viscous than Restylane and requires less pressure to inject. Therefore, it is more suited for treatment of fine perioral lines, as it flows more easily. Because it contains 0.3% lidocaine, discomfort usually is minimal, with treatment lasting approximately 30 to 60 minutes. In the author’s experience (G.G.), Restylane Silk provides a softer correction, though one has to be careful to inject slowly to avoid postinjection swelling. Restylane Silk also may be suitable for neck rejuvenation (off label), but several treatment sessions usually are required. Patients should be warned that they are likely to experience ecchymoses. In our experience, the effects of Restylane Silk injections last approximately 6 to 9 months.
Juvéderm Family
The first Juvéderm product was approved by the FDA in 2006. Juvéderm is a bacterium-derived NASHA. Injectable gel formulations of Juvéderm, including Juvéderm Ultra and Juvéderm Ultra Plus, are FDA approved for the correction of moderate to severe facial wrinkles and folds. The first products in the Juvéderm line were produced using a technology called Hylacross technology, with cohesive molecules of cross-linked HA. In contrast to the sizing technology used by Restylane and Perlane, the Hylacross technology does not break up the cross-linked HA by passing the product through sizing screens via sieves, but instead produces monophasic gels. These Juvéderm products have a high concentration of cross-linked HAs, which accounts for their longevity, and they are soft and easy to use.
Juvéderm Voluma XC was FDA approved in 2013. It is a 20-mg/mL, smooth, highly cohesive, viscous HA, gel that is manufactured using Vycross technology, a combination of low- and high-molecular-weight HA, and it is the only HA filler that is indicated for deep injection for cheek augmentation,10 as it creates a lift due to its higher G′ and a low swelling capacity, with results lasting up to 2 years. The mean volume administered over the initial 4-week period of one study was 5.1 mL.11
Belotero Balance
Belotero Balance was approved by the FDA in 2011 for the correction of moderate to severe facial wrinkles and folds (eg, nasolabial folds).12 Belotero Balance is an HA filler with a cohesive polydensified matrix technology and low elasticity and viscosity. It has the lowest G′ of the currently available dermal fillers12 and therefore is associated with increased injection precision. Belotero Balance is ideal for superficial injections,13 such as forehead lines, vermilion border, tear trough, atrophic scars, and neck lines (off label). Some clinicians reconstitute Belotero Balance with lidocaine (off label) to provide a more pain-free procedure. In our experience, results typically last at least 6 to 8 months.
Complications
Hyaluronic acid fillers share the same adverse events across the product lines. The most common reactions include erythema, swelling, and bruising, which often are unavoidable and may be considered expected effects. Less-frequent events include contour irregularities; product migration; bluish discoloration known as the Tyndall effect, which is more likely to occur with superficial injections; nodules; infection at the injection site; scarring; and vascular occlusion, potentially leading to blindness.14 These more severe complications often can be avoided. Appropriate skin preparation and a sterile technique are critical in preventing infections, while deep placement of filler material reduces the risk for Tyndall effect, nodules, and scarring. Skin necrosis occurs by external compression of the blood supply by the product or occlusion via direct injection into a vessel. Aspirating prior to injection, administering lower volumes, and tenting the skin to inject more superficially can reduce the risk for skin necrosis. Every clinician needs to be able to rapidly recognize the signs of necrosis and to administer urgent therapy, such as the application of warm gauze and nitroglycerin paste, tapping the area to facilitate vasodilatation, and injecting hyaluronidase when required.
On the Horizon
Other dermal fillers that may gain FDA approval in the next few years include Teosyal (Laboratories Teoxane Geneva),6 a new range of monophasic NASHA products that provide high viscosity and elasticity with results lasting 6 to 9 months, and potentially Juvéderm Volbella, a smooth, nonparticle, viscous HA gel developed specifically for the lip area with results lasting up to 1 year.15
Conclusion
Over the last decade, the popularity of dermal fillers has steadily increased, and fillers have become a cornerstone of aesthetic medicine. The increased number of available products necessitates thorough knowledge by the treating physician to ensure optimal outcomes. There is no universal filler that can achieve ideal outcomes in all anatomic sites or in all patients. Often a combination approach may be ideal, such as the use of a product with a higher G′ for re-volumization, followed by a filler with a lower G′ for superficial injection. Some patients may also benefit from a combination of both dermal fillers and neurotoxin injections, either on the same day or at separate visits, which may increase the longevity of the filler.
1. The American Society for Aesthetic Plastic Surgery reports Americans spent more than 12 billion in 2014; procedures for men up 43% over five year period [news release]. New York, NY: American Society for Aesthetic Plastic Surgery; March 11, 2015. http://www.surgery.org/media/news-releases/the-american-society-for-aesthetic-plastic-surgery-reports-americans-spent-more-than-12-billion-in-2014--pro. Accessed July 7, 2015.
2. Fitzgerald R, Rubin AG. Filler placement and the fat compartments. Dermatol Clin. 2014;32:37-50.
3. Cowman MK, Matsuoka S. Experimental approaches to hyaluronan structure. Carbohydr Res. 2005;340:791-809.
4. Lee A, Grummer SE, Kriegel D, et al. Hyaluronidase. Dermatol Surg. 2010;36:1071-1077.
5. Kablik J, Monheit GD, Yu L, et al. Comparative physical properties of hyaluronic acid dermal fillers. Dermatol Surg. 2009;35(suppl 1):302-312.
6. Nast A, Reytan N, Hartmann V, et al. Efficacy and durability of two hyaluronic acid-based fillers in the correction of nasolabial folds: results of a prospective, randomized, double-blind, actively controlled clinical pilot study. Dermatol Surg. 2011;37:768-775.
7. Ascher B, Bayerl C, Brun P, et al. Efficacy and safety of a new hyaluronic acid dermal filler in the treatment of severe nasolabial lines: 6-month interim results of a randomized, evaluator-blinded, intra-individual comparison study. J Cosmet Dermatol. 2011;10:94-98.
8. Rzany B, Bayerl C, Bodokh I, et al. Efficacy and safety of a new hyaluronic acid dermal filler in the treatment of moderate nasolabial folds: 6-month interim results of a randomized, evaluator-blinded, intra-individual comparison study. J Cosmet Laser Ther. 2011;13:107-112.
9. Lupo MP, Swetman G, Waller W. The effect of lidocaine when mixed with large gel particle hyaluronic acid filler tolerability and longevity: a six-month trial. J Drugs Dermatol. 2010;9:1097-1100.
10. Ho D, Jagdeo J. Biological properties of a new volumizing hyaluronic acid filler: a systematic review. J Drugs Dermatol. 2015;14:50-54.
11. Callan P, Goodman GJ, Carlisle I, et al. Efficacy and safety of a hyaluronic acid filler in subjects treated for correction of midface volume deficiency: a 24 month study. Clin Cosmet Investig Dermatol. 2013;6:81-89.
12. Hevia O, Cohen BH, Howell DJ. Safety and efficacy of a cohesive polydensified matrix hyaluronic acid for the correction of infraorbital hollow: an observational study with results at 40 weeks. J Drugs Dermatol. 2014;13:1030-1036.
13. Lorenc ZP, Fagien S, Flynn TC, et al. Clinical application and assessment of Belotero: a roundtable discussion. Plast Reconstr Surg. 2013;132(4, suppl 2):69S-76S.
14. Carruthers JD, Fagien S, Rohrich RJ, et al. Blindness caused by cosmetic filler injection: a review of cause and therapy. Plast Reconstr Surg. 2014;134:1197-1201.
15. Eccleston D, Murphy DK. Juvéderm(®) Volbella™ in the perioral area: a 12-month prospective, multicenter, open-label study. Clin Cosmet Investig Dermatol. 2012;5:167-172.
Facial rejuvenation has become increasingly popular, with nonsurgical and noninvasive procedures comprising a large part of aesthetic practice. According to the American Society for Aesthetic Plastic Surgery, Americans spent $12 billion on cosmetic procedures in 2014, with more than 10 million surgical and nonsurgical procedures performed. The top 5 nonsurgical procedures for both men and women combined were botulinum toxin, hyaluronic acid (HA), hair removal, chemical peel, and microdermabrasion.1
The first dermal filler used was bovine collagen, which was approved by the US Food and Drug Administration (FDA) in 1981. Despite its efficacy in the correction of facial rhytides, bovine collagen required allergy testing prior to use and was discontinued in 2010. Dermal fillers have evolved over the years, and newer products that are superior to earlier fillers with regard to longevity, safety, and tolerability and that do not require allergy testing have become available; however, advances in the use of dermal fillers are not only related to the development of newer products but also to evolving injection techniques. Initially, the aim of treatment with dermal fillers was to correct lines and wrinkles, but an increased understanding of the complex changes that occur with aging have changed our approach to one of volume replacement, with an emphasis on volume restoration in the midface. This approach requires an in-depth understanding of facial anatomy as well as the interactions of the skin, soft tissue, muscle, and bone. Furthermore, placement of filler in specific fat compartments can provide a more natural appearance and an all-around youthful face.2 In this article, we discuss HA fillers that have gained FDA approval within the last 5 years (Table).
Overview of HA Fillers
Hyaluronic acid is a naturally occurring linear glycosaminoglycan with a disaccharide unit, which repeats several thousand times.3 Hyaluronic acid is an essential part of the extracellular matrix of many tissues including the dermis and plays an important role in tissue growth, development, and wound healing. Hyaluronic acid is hygroscopic and absorbs water extensively, thus creating volume.4 Treatment with HA fillers is popular, as they are biocompatible and have a low potential for allergic reactions. They also are easy to use and reversible.4 The first HA filler to gain FDA approval was Restylane (Galderma Laboratories, LP). Currently, several HA fillers are approved in the United States, and each product differs from the others in polymer chain length, degree of HA concentration, particle size, gel consistency, gel hardness, gel viscosity, and degree of water solubility, as well as amount and degree of cross-linking. Cross-linking is essential to avoid enzymatic degradation by endogenous hyaluronidase when injected into the skin and thus to prolong the product’s half-life.5 Cross-linkers used to manufacture HA fillers include 1,4-butanediol diglycidyl ether and divinyl sulfone. More concentrated products with a greater degree of cross-linking provide increased longevity, but they are associated with a higher risk for inflammation and nodule formation. The elastic modulus (G′) is a measure of the firmness of dermal fillers, describing their resistance to deformation. Materials with a higher G′ are stiffer and are meant for deeper injections. Hyaluronic acid fillers can be further classified as biphasic or monophasic. Biphasic fillers (eg, Restylane, Perlane [Galderma Laboratories, LP]) contain a range of microsphere sizes, while monophasic fillers (eg, Juvéderm [Allergan, Inc], Belotero Balance [Merz North America, Inc]) contain homogeneous microspheres. Although randomized clinical trials have reported comparable efficacy and durability of biphasic and monophasic fillers when used to treat the nasolabial folds,6-8 monophasic HA fillers are more cohesive and may not migrate as much following injection.
Restylane Family
Restylane was the first FDA-approved HA filler, gaining its approval in 2003. Restylane is a nonanimal stabilized HA (NASHA) that is produced from the fermentation of equine streptococci. It is cross-linked with 1,4-butanediol diglycidyl ether with a 1% degree of cross-linking. Restylane has an HA concentration of 20 mg/mL. The particle size range of Restylane and Restylane-L is 330 to 430 mm. Restylane (and also Perlane) get passed through sizing screens via sieves and are quantified by their size. The longevity of HA fillers is approximately 6 months; however, various factors affect the product’s longevity, such as the degree of cross-linking, treatment area, and the patient’s metabolism. Restylane-L, which was FDA approved in 2012, is a newer product with 0.3% lidocaine incorporated into the syringe itself. It was the first product from the Restylane range to be approved for lip augmentation. The addition of lidocaine (designated by the L in the product name) does not affect the longevity of a filler.9
The newest FDA-approved HA filler was Restylane Silk (approved in 2014), which has been specifically designed for lip augmentation and correction of perioral rhytides. To avoid postprocedural swelling, it generally is recommended that Restylane Silk be injected slowly. If required, a short course of oral prednisone may be administered after the procedure to treat any edema. Restylane Silk is less viscous than Restylane and requires less pressure to inject. Therefore, it is more suited for treatment of fine perioral lines, as it flows more easily. Because it contains 0.3% lidocaine, discomfort usually is minimal, with treatment lasting approximately 30 to 60 minutes. In the author’s experience (G.G.), Restylane Silk provides a softer correction, though one has to be careful to inject slowly to avoid postinjection swelling. Restylane Silk also may be suitable for neck rejuvenation (off label), but several treatment sessions usually are required. Patients should be warned that they are likely to experience ecchymoses. In our experience, the effects of Restylane Silk injections last approximately 6 to 9 months.
Juvéderm Family
The first Juvéderm product was approved by the FDA in 2006. Juvéderm is a bacterium-derived NASHA. Injectable gel formulations of Juvéderm, including Juvéderm Ultra and Juvéderm Ultra Plus, are FDA approved for the correction of moderate to severe facial wrinkles and folds. The first products in the Juvéderm line were produced using a technology called Hylacross technology, with cohesive molecules of cross-linked HA. In contrast to the sizing technology used by Restylane and Perlane, the Hylacross technology does not break up the cross-linked HA by passing the product through sizing screens via sieves, but instead produces monophasic gels. These Juvéderm products have a high concentration of cross-linked HAs, which accounts for their longevity, and they are soft and easy to use.
Juvéderm Voluma XC was FDA approved in 2013. It is a 20-mg/mL, smooth, highly cohesive, viscous HA, gel that is manufactured using Vycross technology, a combination of low- and high-molecular-weight HA, and it is the only HA filler that is indicated for deep injection for cheek augmentation,10 as it creates a lift due to its higher G′ and a low swelling capacity, with results lasting up to 2 years. The mean volume administered over the initial 4-week period of one study was 5.1 mL.11
Belotero Balance
Belotero Balance was approved by the FDA in 2011 for the correction of moderate to severe facial wrinkles and folds (eg, nasolabial folds).12 Belotero Balance is an HA filler with a cohesive polydensified matrix technology and low elasticity and viscosity. It has the lowest G′ of the currently available dermal fillers12 and therefore is associated with increased injection precision. Belotero Balance is ideal for superficial injections,13 such as forehead lines, vermilion border, tear trough, atrophic scars, and neck lines (off label). Some clinicians reconstitute Belotero Balance with lidocaine (off label) to provide a more pain-free procedure. In our experience, results typically last at least 6 to 8 months.
Complications
Hyaluronic acid fillers share the same adverse events across the product lines. The most common reactions include erythema, swelling, and bruising, which often are unavoidable and may be considered expected effects. Less-frequent events include contour irregularities; product migration; bluish discoloration known as the Tyndall effect, which is more likely to occur with superficial injections; nodules; infection at the injection site; scarring; and vascular occlusion, potentially leading to blindness.14 These more severe complications often can be avoided. Appropriate skin preparation and a sterile technique are critical in preventing infections, while deep placement of filler material reduces the risk for Tyndall effect, nodules, and scarring. Skin necrosis occurs by external compression of the blood supply by the product or occlusion via direct injection into a vessel. Aspirating prior to injection, administering lower volumes, and tenting the skin to inject more superficially can reduce the risk for skin necrosis. Every clinician needs to be able to rapidly recognize the signs of necrosis and to administer urgent therapy, such as the application of warm gauze and nitroglycerin paste, tapping the area to facilitate vasodilatation, and injecting hyaluronidase when required.
On the Horizon
Other dermal fillers that may gain FDA approval in the next few years include Teosyal (Laboratories Teoxane Geneva),6 a new range of monophasic NASHA products that provide high viscosity and elasticity with results lasting 6 to 9 months, and potentially Juvéderm Volbella, a smooth, nonparticle, viscous HA gel developed specifically for the lip area with results lasting up to 1 year.15
Conclusion
Over the last decade, the popularity of dermal fillers has steadily increased, and fillers have become a cornerstone of aesthetic medicine. The increased number of available products necessitates thorough knowledge by the treating physician to ensure optimal outcomes. There is no universal filler that can achieve ideal outcomes in all anatomic sites or in all patients. Often a combination approach may be ideal, such as the use of a product with a higher G′ for re-volumization, followed by a filler with a lower G′ for superficial injection. Some patients may also benefit from a combination of both dermal fillers and neurotoxin injections, either on the same day or at separate visits, which may increase the longevity of the filler.
Facial rejuvenation has become increasingly popular, with nonsurgical and noninvasive procedures comprising a large part of aesthetic practice. According to the American Society for Aesthetic Plastic Surgery, Americans spent $12 billion on cosmetic procedures in 2014, with more than 10 million surgical and nonsurgical procedures performed. The top 5 nonsurgical procedures for both men and women combined were botulinum toxin, hyaluronic acid (HA), hair removal, chemical peel, and microdermabrasion.1
The first dermal filler used was bovine collagen, which was approved by the US Food and Drug Administration (FDA) in 1981. Despite its efficacy in the correction of facial rhytides, bovine collagen required allergy testing prior to use and was discontinued in 2010. Dermal fillers have evolved over the years, and newer products that are superior to earlier fillers with regard to longevity, safety, and tolerability and that do not require allergy testing have become available; however, advances in the use of dermal fillers are not only related to the development of newer products but also to evolving injection techniques. Initially, the aim of treatment with dermal fillers was to correct lines and wrinkles, but an increased understanding of the complex changes that occur with aging have changed our approach to one of volume replacement, with an emphasis on volume restoration in the midface. This approach requires an in-depth understanding of facial anatomy as well as the interactions of the skin, soft tissue, muscle, and bone. Furthermore, placement of filler in specific fat compartments can provide a more natural appearance and an all-around youthful face.2 In this article, we discuss HA fillers that have gained FDA approval within the last 5 years (Table).
Overview of HA Fillers
Hyaluronic acid is a naturally occurring linear glycosaminoglycan with a disaccharide unit, which repeats several thousand times.3 Hyaluronic acid is an essential part of the extracellular matrix of many tissues including the dermis and plays an important role in tissue growth, development, and wound healing. Hyaluronic acid is hygroscopic and absorbs water extensively, thus creating volume.4 Treatment with HA fillers is popular, as they are biocompatible and have a low potential for allergic reactions. They also are easy to use and reversible.4 The first HA filler to gain FDA approval was Restylane (Galderma Laboratories, LP). Currently, several HA fillers are approved in the United States, and each product differs from the others in polymer chain length, degree of HA concentration, particle size, gel consistency, gel hardness, gel viscosity, and degree of water solubility, as well as amount and degree of cross-linking. Cross-linking is essential to avoid enzymatic degradation by endogenous hyaluronidase when injected into the skin and thus to prolong the product’s half-life.5 Cross-linkers used to manufacture HA fillers include 1,4-butanediol diglycidyl ether and divinyl sulfone. More concentrated products with a greater degree of cross-linking provide increased longevity, but they are associated with a higher risk for inflammation and nodule formation. The elastic modulus (G′) is a measure of the firmness of dermal fillers, describing their resistance to deformation. Materials with a higher G′ are stiffer and are meant for deeper injections. Hyaluronic acid fillers can be further classified as biphasic or monophasic. Biphasic fillers (eg, Restylane, Perlane [Galderma Laboratories, LP]) contain a range of microsphere sizes, while monophasic fillers (eg, Juvéderm [Allergan, Inc], Belotero Balance [Merz North America, Inc]) contain homogeneous microspheres. Although randomized clinical trials have reported comparable efficacy and durability of biphasic and monophasic fillers when used to treat the nasolabial folds,6-8 monophasic HA fillers are more cohesive and may not migrate as much following injection.
Restylane Family
Restylane was the first FDA-approved HA filler, gaining its approval in 2003. Restylane is a nonanimal stabilized HA (NASHA) that is produced from the fermentation of equine streptococci. It is cross-linked with 1,4-butanediol diglycidyl ether with a 1% degree of cross-linking. Restylane has an HA concentration of 20 mg/mL. The particle size range of Restylane and Restylane-L is 330 to 430 mm. Restylane (and also Perlane) get passed through sizing screens via sieves and are quantified by their size. The longevity of HA fillers is approximately 6 months; however, various factors affect the product’s longevity, such as the degree of cross-linking, treatment area, and the patient’s metabolism. Restylane-L, which was FDA approved in 2012, is a newer product with 0.3% lidocaine incorporated into the syringe itself. It was the first product from the Restylane range to be approved for lip augmentation. The addition of lidocaine (designated by the L in the product name) does not affect the longevity of a filler.9
The newest FDA-approved HA filler was Restylane Silk (approved in 2014), which has been specifically designed for lip augmentation and correction of perioral rhytides. To avoid postprocedural swelling, it generally is recommended that Restylane Silk be injected slowly. If required, a short course of oral prednisone may be administered after the procedure to treat any edema. Restylane Silk is less viscous than Restylane and requires less pressure to inject. Therefore, it is more suited for treatment of fine perioral lines, as it flows more easily. Because it contains 0.3% lidocaine, discomfort usually is minimal, with treatment lasting approximately 30 to 60 minutes. In the author’s experience (G.G.), Restylane Silk provides a softer correction, though one has to be careful to inject slowly to avoid postinjection swelling. Restylane Silk also may be suitable for neck rejuvenation (off label), but several treatment sessions usually are required. Patients should be warned that they are likely to experience ecchymoses. In our experience, the effects of Restylane Silk injections last approximately 6 to 9 months.
Juvéderm Family
The first Juvéderm product was approved by the FDA in 2006. Juvéderm is a bacterium-derived NASHA. Injectable gel formulations of Juvéderm, including Juvéderm Ultra and Juvéderm Ultra Plus, are FDA approved for the correction of moderate to severe facial wrinkles and folds. The first products in the Juvéderm line were produced using a technology called Hylacross technology, with cohesive molecules of cross-linked HA. In contrast to the sizing technology used by Restylane and Perlane, the Hylacross technology does not break up the cross-linked HA by passing the product through sizing screens via sieves, but instead produces monophasic gels. These Juvéderm products have a high concentration of cross-linked HAs, which accounts for their longevity, and they are soft and easy to use.
Juvéderm Voluma XC was FDA approved in 2013. It is a 20-mg/mL, smooth, highly cohesive, viscous HA, gel that is manufactured using Vycross technology, a combination of low- and high-molecular-weight HA, and it is the only HA filler that is indicated for deep injection for cheek augmentation,10 as it creates a lift due to its higher G′ and a low swelling capacity, with results lasting up to 2 years. The mean volume administered over the initial 4-week period of one study was 5.1 mL.11
Belotero Balance
Belotero Balance was approved by the FDA in 2011 for the correction of moderate to severe facial wrinkles and folds (eg, nasolabial folds).12 Belotero Balance is an HA filler with a cohesive polydensified matrix technology and low elasticity and viscosity. It has the lowest G′ of the currently available dermal fillers12 and therefore is associated with increased injection precision. Belotero Balance is ideal for superficial injections,13 such as forehead lines, vermilion border, tear trough, atrophic scars, and neck lines (off label). Some clinicians reconstitute Belotero Balance with lidocaine (off label) to provide a more pain-free procedure. In our experience, results typically last at least 6 to 8 months.
Complications
Hyaluronic acid fillers share the same adverse events across the product lines. The most common reactions include erythema, swelling, and bruising, which often are unavoidable and may be considered expected effects. Less-frequent events include contour irregularities; product migration; bluish discoloration known as the Tyndall effect, which is more likely to occur with superficial injections; nodules; infection at the injection site; scarring; and vascular occlusion, potentially leading to blindness.14 These more severe complications often can be avoided. Appropriate skin preparation and a sterile technique are critical in preventing infections, while deep placement of filler material reduces the risk for Tyndall effect, nodules, and scarring. Skin necrosis occurs by external compression of the blood supply by the product or occlusion via direct injection into a vessel. Aspirating prior to injection, administering lower volumes, and tenting the skin to inject more superficially can reduce the risk for skin necrosis. Every clinician needs to be able to rapidly recognize the signs of necrosis and to administer urgent therapy, such as the application of warm gauze and nitroglycerin paste, tapping the area to facilitate vasodilatation, and injecting hyaluronidase when required.
On the Horizon
Other dermal fillers that may gain FDA approval in the next few years include Teosyal (Laboratories Teoxane Geneva),6 a new range of monophasic NASHA products that provide high viscosity and elasticity with results lasting 6 to 9 months, and potentially Juvéderm Volbella, a smooth, nonparticle, viscous HA gel developed specifically for the lip area with results lasting up to 1 year.15
Conclusion
Over the last decade, the popularity of dermal fillers has steadily increased, and fillers have become a cornerstone of aesthetic medicine. The increased number of available products necessitates thorough knowledge by the treating physician to ensure optimal outcomes. There is no universal filler that can achieve ideal outcomes in all anatomic sites or in all patients. Often a combination approach may be ideal, such as the use of a product with a higher G′ for re-volumization, followed by a filler with a lower G′ for superficial injection. Some patients may also benefit from a combination of both dermal fillers and neurotoxin injections, either on the same day or at separate visits, which may increase the longevity of the filler.
1. The American Society for Aesthetic Plastic Surgery reports Americans spent more than 12 billion in 2014; procedures for men up 43% over five year period [news release]. New York, NY: American Society for Aesthetic Plastic Surgery; March 11, 2015. http://www.surgery.org/media/news-releases/the-american-society-for-aesthetic-plastic-surgery-reports-americans-spent-more-than-12-billion-in-2014--pro. Accessed July 7, 2015.
2. Fitzgerald R, Rubin AG. Filler placement and the fat compartments. Dermatol Clin. 2014;32:37-50.
3. Cowman MK, Matsuoka S. Experimental approaches to hyaluronan structure. Carbohydr Res. 2005;340:791-809.
4. Lee A, Grummer SE, Kriegel D, et al. Hyaluronidase. Dermatol Surg. 2010;36:1071-1077.
5. Kablik J, Monheit GD, Yu L, et al. Comparative physical properties of hyaluronic acid dermal fillers. Dermatol Surg. 2009;35(suppl 1):302-312.
6. Nast A, Reytan N, Hartmann V, et al. Efficacy and durability of two hyaluronic acid-based fillers in the correction of nasolabial folds: results of a prospective, randomized, double-blind, actively controlled clinical pilot study. Dermatol Surg. 2011;37:768-775.
7. Ascher B, Bayerl C, Brun P, et al. Efficacy and safety of a new hyaluronic acid dermal filler in the treatment of severe nasolabial lines: 6-month interim results of a randomized, evaluator-blinded, intra-individual comparison study. J Cosmet Dermatol. 2011;10:94-98.
8. Rzany B, Bayerl C, Bodokh I, et al. Efficacy and safety of a new hyaluronic acid dermal filler in the treatment of moderate nasolabial folds: 6-month interim results of a randomized, evaluator-blinded, intra-individual comparison study. J Cosmet Laser Ther. 2011;13:107-112.
9. Lupo MP, Swetman G, Waller W. The effect of lidocaine when mixed with large gel particle hyaluronic acid filler tolerability and longevity: a six-month trial. J Drugs Dermatol. 2010;9:1097-1100.
10. Ho D, Jagdeo J. Biological properties of a new volumizing hyaluronic acid filler: a systematic review. J Drugs Dermatol. 2015;14:50-54.
11. Callan P, Goodman GJ, Carlisle I, et al. Efficacy and safety of a hyaluronic acid filler in subjects treated for correction of midface volume deficiency: a 24 month study. Clin Cosmet Investig Dermatol. 2013;6:81-89.
12. Hevia O, Cohen BH, Howell DJ. Safety and efficacy of a cohesive polydensified matrix hyaluronic acid for the correction of infraorbital hollow: an observational study with results at 40 weeks. J Drugs Dermatol. 2014;13:1030-1036.
13. Lorenc ZP, Fagien S, Flynn TC, et al. Clinical application and assessment of Belotero: a roundtable discussion. Plast Reconstr Surg. 2013;132(4, suppl 2):69S-76S.
14. Carruthers JD, Fagien S, Rohrich RJ, et al. Blindness caused by cosmetic filler injection: a review of cause and therapy. Plast Reconstr Surg. 2014;134:1197-1201.
15. Eccleston D, Murphy DK. Juvéderm(®) Volbella™ in the perioral area: a 12-month prospective, multicenter, open-label study. Clin Cosmet Investig Dermatol. 2012;5:167-172.
1. The American Society for Aesthetic Plastic Surgery reports Americans spent more than 12 billion in 2014; procedures for men up 43% over five year period [news release]. New York, NY: American Society for Aesthetic Plastic Surgery; March 11, 2015. http://www.surgery.org/media/news-releases/the-american-society-for-aesthetic-plastic-surgery-reports-americans-spent-more-than-12-billion-in-2014--pro. Accessed July 7, 2015.
2. Fitzgerald R, Rubin AG. Filler placement and the fat compartments. Dermatol Clin. 2014;32:37-50.
3. Cowman MK, Matsuoka S. Experimental approaches to hyaluronan structure. Carbohydr Res. 2005;340:791-809.
4. Lee A, Grummer SE, Kriegel D, et al. Hyaluronidase. Dermatol Surg. 2010;36:1071-1077.
5. Kablik J, Monheit GD, Yu L, et al. Comparative physical properties of hyaluronic acid dermal fillers. Dermatol Surg. 2009;35(suppl 1):302-312.
6. Nast A, Reytan N, Hartmann V, et al. Efficacy and durability of two hyaluronic acid-based fillers in the correction of nasolabial folds: results of a prospective, randomized, double-blind, actively controlled clinical pilot study. Dermatol Surg. 2011;37:768-775.
7. Ascher B, Bayerl C, Brun P, et al. Efficacy and safety of a new hyaluronic acid dermal filler in the treatment of severe nasolabial lines: 6-month interim results of a randomized, evaluator-blinded, intra-individual comparison study. J Cosmet Dermatol. 2011;10:94-98.
8. Rzany B, Bayerl C, Bodokh I, et al. Efficacy and safety of a new hyaluronic acid dermal filler in the treatment of moderate nasolabial folds: 6-month interim results of a randomized, evaluator-blinded, intra-individual comparison study. J Cosmet Laser Ther. 2011;13:107-112.
9. Lupo MP, Swetman G, Waller W. The effect of lidocaine when mixed with large gel particle hyaluronic acid filler tolerability and longevity: a six-month trial. J Drugs Dermatol. 2010;9:1097-1100.
10. Ho D, Jagdeo J. Biological properties of a new volumizing hyaluronic acid filler: a systematic review. J Drugs Dermatol. 2015;14:50-54.
11. Callan P, Goodman GJ, Carlisle I, et al. Efficacy and safety of a hyaluronic acid filler in subjects treated for correction of midface volume deficiency: a 24 month study. Clin Cosmet Investig Dermatol. 2013;6:81-89.
12. Hevia O, Cohen BH, Howell DJ. Safety and efficacy of a cohesive polydensified matrix hyaluronic acid for the correction of infraorbital hollow: an observational study with results at 40 weeks. J Drugs Dermatol. 2014;13:1030-1036.
13. Lorenc ZP, Fagien S, Flynn TC, et al. Clinical application and assessment of Belotero: a roundtable discussion. Plast Reconstr Surg. 2013;132(4, suppl 2):69S-76S.
14. Carruthers JD, Fagien S, Rohrich RJ, et al. Blindness caused by cosmetic filler injection: a review of cause and therapy. Plast Reconstr Surg. 2014;134:1197-1201.
15. Eccleston D, Murphy DK. Juvéderm(®) Volbella™ in the perioral area: a 12-month prospective, multicenter, open-label study. Clin Cosmet Investig Dermatol. 2012;5:167-172.
Practice Points
- Restylane Silk is useful for the treatment of fine perioral lines.
- Juvéderm Voluma XC is a newer product in the Juvéderm range and is indicated for cheek augmentation.
- Belotero Balance has the lowest G′ of the currently available dermal fillers and allows greater precision.
What Is Your Diagnosis? Verrucous Carcinoma
An 81-year-old woman presented for evaluation of a nodule on the right labia majora that had been present for 1 year. She had a history of intertriginous psoriasis, and several biopsies were performed at an outside facility over the last 5 years that revealed psoriasis but were otherwise noncontributory. Physical examination revealed erythema and scaling on the buttocks with maceration in the intertriginous area (top) and the perineum associated with a verrucous nodule (bottom).
The Diagnosis: Verrucous Carcinoma
Biopsies of early lesions often may be difficult to interpret without clinicopathological correlation. Our patient’s tumor was associated with intertriginous psoriasis, which was the only abnormality previously noted on superficial biopsies performed at an outside facility. The patient was scheduled for an excisional biopsy due to the large tumor size and clinical suspicion that the prior biopsies were inadequate and failed to demonstrate the primary underlying pathology. Excisional biopsy of the verrucous tumor revealed epithelium composed of keratinocytes with glassy cytoplasm. Papillomatosis was noted along with an endophytic component of well-differentiated epithelial cells extending into the dermis in a bulbous pattern consistent with the verrucous carcinoma variant of squamous cell carcinoma (SCC)(Figure). Verrucous carcinoma often requires correlation with both the clinical and histopathologic findings for definitive diagnosis, as keratinocytes often appear to be well differentiated.1
Verrucous carcinoma may begin as an innocuous papule that slowly grows into a large fungating tumor. Verrucous carcinomas typically are slow growing, exophytic, and low grade. The etiology of verrucous carcinoma is not clear, and the role of human papillomavirus (HPV) infection is controversial.2 Best classified as a well-differentiated SCC, verrucous carcinoma rarely metastasizes but may invade adjacent tissues.
Differential diagnoses include a giant inflamed seborrheic keratosis, condyloma acuminatum, rupioid psoriasis, and inflammatory linear verrucous epidermal nevus (ILVEN). Although large and inflamed seborrheic keratoses may have squamous eddies that mimic SCC, seborrheic keratoses do not invade the dermis and typically have a well-circumscribed stuck-on appearance. Abnormal mitotic figures are not identified. Condylomas are genital warts caused by HPV infection that often are clustered, well circumscribed, and exophytic. Large lesions can be difficult to distinguish from verrucous carcinomas, and biopsy generally reveals koilocytes identified by perinuclear clearing and raisinlike nuclei. Immunohistochemical staining and in situ hybridization studies can be of value in diagnosis and in identifying those lesions that are at high risk for malignant transformation. High-risk condylomas are associated with HPV-16, HPV-18, HPV-31, HPV-33, HPV-35, and HPV-39, as well as other types, whereas low-risk condylomas are associated with HPV-6, HPV-11, HPV-42, and others.2 Differentiating squamous cell hyperplasia from squamous cell carcinoma in situ also can be aided by immunohistochemistry. Squamous cell hyperplasia is usually negative for INK4 p16Ink4A and p53 and exhibits variable Ki-67 staining. Differentiated squamous cell carcinoma in situ exhibits a profile that is p16Ink4A negative, Ki-67 positive, and exhibits variable p53 staining.3 Basaloid and warty intraepithelial neoplasia is consistently p16Ink4A positive, Ki-67 positive, and variably positive for p53.3 Therefore, p16 staining of high-grade areas is a useful biomarker that can help establish diagnosis of associated squamous cell carcinoma.4 The role of papillomaviruses in the development of nonmelanoma skin cancer is an area of active study, and research suggests that papillomaviruses may have a much greater role than previously suspected.5
At times, psoriasis may be markedly hyperkeratotic, clinically mimicking a verrucous neoplasm. This hyperkeratotic type of psoriasis is known as rupioid psoriasis. However, these psoriatic lesions are exophytic, are associated with spongiform pustules, and lack the atypia and endophytic pattern typically seen with verrucous carcinoma. An ILVEN also lacks atypia and an endophytic pattern and usually presents in childhood as a persistent linear plaque, rather than the verrucous plaque noted in our patient. Squamous cell carcinoma has been reported to arise in the setting of verrucoid ILVEN but is exceptionally uncommon.6
Successful treatment of verrucous carcinoma is best achieved by complete excision. Oral retinoids and immunomodulators such as imiquimod also may be of value.7 Our patient’s tumor qualifies as T2N0M0 because it was greater than 2 cm in size.8 A Breslow thickness of 2 mm or greater and Clark level IV are high-risk features associated with a worse prognosis, but clinical evaluation of our patient’s lymph nodes was unremarkable and no distant metastases were identified. Our patient continues to do well with no evidence of recurrence.
1. Bambao C, Nofech-Mozes S, Shier M. Giant condyloma versus verrucous carcinoma: a case report. J Low Genit Tract Dis. 2010;14:230-233.
2. Asiaf A, Ahmad ST, Mohannad SO, et al. Review of the current knowledge on the epidemiology, pathogenesis, and prevention of human papillomavirus infection. Eur J Cancer Prev. 2014;23:206-224.
3. Chaux A, Pfannl R, Rodríguez IM, et al. Distinctive immunohistochemical profile of penile intraepithelial lesions: a study of 74 cases. Am J Surg Pathol. 2011;35:553-562.
4. Darragh TM, Colgan TJ, Cox JT, et al. The lower anogenital squamous terminology standardization project for HPV-associated lesions: background and consensus recommendations from the College of American Pathologists and the American Society for Colposcopy and Cervical Pathology. Arch Pathol Lab Med. 2012;136:1266-1297.
5. Aldabagh B, Angeles J, Cardones AR, et al. Cutaneous squamous cell carcinoma and human papillomavirus: is there an association? Dermatol Surg. 2013;39:1-23.
6. Turk BG, Ertam I, Urkmez A, et al. Development of squamous cell carcinoma on an inflammatory linear verrucous epidermal nevus in the genital area. Cutis. 2012;89:273-275.
7. Erkek E, Basar H, Bozdogan O, et al. Giant condyloma acuminata of Buschke-Löwenstein: successful treatment with a combination of surgical excision, oral acitretin and topical imiquimod. Clin Exp Dermatol. 2009;34:366-368.
8. Cutaneous squamous cell carcinoma and other cutaneous carcinomas. In: Edge SB, Byrd DR, Compton CC, et al, eds. AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer; 2010:301-314.
An 81-year-old woman presented for evaluation of a nodule on the right labia majora that had been present for 1 year. She had a history of intertriginous psoriasis, and several biopsies were performed at an outside facility over the last 5 years that revealed psoriasis but were otherwise noncontributory. Physical examination revealed erythema and scaling on the buttocks with maceration in the intertriginous area (top) and the perineum associated with a verrucous nodule (bottom).
The Diagnosis: Verrucous Carcinoma
Biopsies of early lesions often may be difficult to interpret without clinicopathological correlation. Our patient’s tumor was associated with intertriginous psoriasis, which was the only abnormality previously noted on superficial biopsies performed at an outside facility. The patient was scheduled for an excisional biopsy due to the large tumor size and clinical suspicion that the prior biopsies were inadequate and failed to demonstrate the primary underlying pathology. Excisional biopsy of the verrucous tumor revealed epithelium composed of keratinocytes with glassy cytoplasm. Papillomatosis was noted along with an endophytic component of well-differentiated epithelial cells extending into the dermis in a bulbous pattern consistent with the verrucous carcinoma variant of squamous cell carcinoma (SCC)(Figure). Verrucous carcinoma often requires correlation with both the clinical and histopathologic findings for definitive diagnosis, as keratinocytes often appear to be well differentiated.1
Verrucous carcinoma may begin as an innocuous papule that slowly grows into a large fungating tumor. Verrucous carcinomas typically are slow growing, exophytic, and low grade. The etiology of verrucous carcinoma is not clear, and the role of human papillomavirus (HPV) infection is controversial.2 Best classified as a well-differentiated SCC, verrucous carcinoma rarely metastasizes but may invade adjacent tissues.
Differential diagnoses include a giant inflamed seborrheic keratosis, condyloma acuminatum, rupioid psoriasis, and inflammatory linear verrucous epidermal nevus (ILVEN). Although large and inflamed seborrheic keratoses may have squamous eddies that mimic SCC, seborrheic keratoses do not invade the dermis and typically have a well-circumscribed stuck-on appearance. Abnormal mitotic figures are not identified. Condylomas are genital warts caused by HPV infection that often are clustered, well circumscribed, and exophytic. Large lesions can be difficult to distinguish from verrucous carcinomas, and biopsy generally reveals koilocytes identified by perinuclear clearing and raisinlike nuclei. Immunohistochemical staining and in situ hybridization studies can be of value in diagnosis and in identifying those lesions that are at high risk for malignant transformation. High-risk condylomas are associated with HPV-16, HPV-18, HPV-31, HPV-33, HPV-35, and HPV-39, as well as other types, whereas low-risk condylomas are associated with HPV-6, HPV-11, HPV-42, and others.2 Differentiating squamous cell hyperplasia from squamous cell carcinoma in situ also can be aided by immunohistochemistry. Squamous cell hyperplasia is usually negative for INK4 p16Ink4A and p53 and exhibits variable Ki-67 staining. Differentiated squamous cell carcinoma in situ exhibits a profile that is p16Ink4A negative, Ki-67 positive, and exhibits variable p53 staining.3 Basaloid and warty intraepithelial neoplasia is consistently p16Ink4A positive, Ki-67 positive, and variably positive for p53.3 Therefore, p16 staining of high-grade areas is a useful biomarker that can help establish diagnosis of associated squamous cell carcinoma.4 The role of papillomaviruses in the development of nonmelanoma skin cancer is an area of active study, and research suggests that papillomaviruses may have a much greater role than previously suspected.5
At times, psoriasis may be markedly hyperkeratotic, clinically mimicking a verrucous neoplasm. This hyperkeratotic type of psoriasis is known as rupioid psoriasis. However, these psoriatic lesions are exophytic, are associated with spongiform pustules, and lack the atypia and endophytic pattern typically seen with verrucous carcinoma. An ILVEN also lacks atypia and an endophytic pattern and usually presents in childhood as a persistent linear plaque, rather than the verrucous plaque noted in our patient. Squamous cell carcinoma has been reported to arise in the setting of verrucoid ILVEN but is exceptionally uncommon.6
Successful treatment of verrucous carcinoma is best achieved by complete excision. Oral retinoids and immunomodulators such as imiquimod also may be of value.7 Our patient’s tumor qualifies as T2N0M0 because it was greater than 2 cm in size.8 A Breslow thickness of 2 mm or greater and Clark level IV are high-risk features associated with a worse prognosis, but clinical evaluation of our patient’s lymph nodes was unremarkable and no distant metastases were identified. Our patient continues to do well with no evidence of recurrence.
An 81-year-old woman presented for evaluation of a nodule on the right labia majora that had been present for 1 year. She had a history of intertriginous psoriasis, and several biopsies were performed at an outside facility over the last 5 years that revealed psoriasis but were otherwise noncontributory. Physical examination revealed erythema and scaling on the buttocks with maceration in the intertriginous area (top) and the perineum associated with a verrucous nodule (bottom).
The Diagnosis: Verrucous Carcinoma
Biopsies of early lesions often may be difficult to interpret without clinicopathological correlation. Our patient’s tumor was associated with intertriginous psoriasis, which was the only abnormality previously noted on superficial biopsies performed at an outside facility. The patient was scheduled for an excisional biopsy due to the large tumor size and clinical suspicion that the prior biopsies were inadequate and failed to demonstrate the primary underlying pathology. Excisional biopsy of the verrucous tumor revealed epithelium composed of keratinocytes with glassy cytoplasm. Papillomatosis was noted along with an endophytic component of well-differentiated epithelial cells extending into the dermis in a bulbous pattern consistent with the verrucous carcinoma variant of squamous cell carcinoma (SCC)(Figure). Verrucous carcinoma often requires correlation with both the clinical and histopathologic findings for definitive diagnosis, as keratinocytes often appear to be well differentiated.1
Verrucous carcinoma may begin as an innocuous papule that slowly grows into a large fungating tumor. Verrucous carcinomas typically are slow growing, exophytic, and low grade. The etiology of verrucous carcinoma is not clear, and the role of human papillomavirus (HPV) infection is controversial.2 Best classified as a well-differentiated SCC, verrucous carcinoma rarely metastasizes but may invade adjacent tissues.
Differential diagnoses include a giant inflamed seborrheic keratosis, condyloma acuminatum, rupioid psoriasis, and inflammatory linear verrucous epidermal nevus (ILVEN). Although large and inflamed seborrheic keratoses may have squamous eddies that mimic SCC, seborrheic keratoses do not invade the dermis and typically have a well-circumscribed stuck-on appearance. Abnormal mitotic figures are not identified. Condylomas are genital warts caused by HPV infection that often are clustered, well circumscribed, and exophytic. Large lesions can be difficult to distinguish from verrucous carcinomas, and biopsy generally reveals koilocytes identified by perinuclear clearing and raisinlike nuclei. Immunohistochemical staining and in situ hybridization studies can be of value in diagnosis and in identifying those lesions that are at high risk for malignant transformation. High-risk condylomas are associated with HPV-16, HPV-18, HPV-31, HPV-33, HPV-35, and HPV-39, as well as other types, whereas low-risk condylomas are associated with HPV-6, HPV-11, HPV-42, and others.2 Differentiating squamous cell hyperplasia from squamous cell carcinoma in situ also can be aided by immunohistochemistry. Squamous cell hyperplasia is usually negative for INK4 p16Ink4A and p53 and exhibits variable Ki-67 staining. Differentiated squamous cell carcinoma in situ exhibits a profile that is p16Ink4A negative, Ki-67 positive, and exhibits variable p53 staining.3 Basaloid and warty intraepithelial neoplasia is consistently p16Ink4A positive, Ki-67 positive, and variably positive for p53.3 Therefore, p16 staining of high-grade areas is a useful biomarker that can help establish diagnosis of associated squamous cell carcinoma.4 The role of papillomaviruses in the development of nonmelanoma skin cancer is an area of active study, and research suggests that papillomaviruses may have a much greater role than previously suspected.5
At times, psoriasis may be markedly hyperkeratotic, clinically mimicking a verrucous neoplasm. This hyperkeratotic type of psoriasis is known as rupioid psoriasis. However, these psoriatic lesions are exophytic, are associated with spongiform pustules, and lack the atypia and endophytic pattern typically seen with verrucous carcinoma. An ILVEN also lacks atypia and an endophytic pattern and usually presents in childhood as a persistent linear plaque, rather than the verrucous plaque noted in our patient. Squamous cell carcinoma has been reported to arise in the setting of verrucoid ILVEN but is exceptionally uncommon.6
Successful treatment of verrucous carcinoma is best achieved by complete excision. Oral retinoids and immunomodulators such as imiquimod also may be of value.7 Our patient’s tumor qualifies as T2N0M0 because it was greater than 2 cm in size.8 A Breslow thickness of 2 mm or greater and Clark level IV are high-risk features associated with a worse prognosis, but clinical evaluation of our patient’s lymph nodes was unremarkable and no distant metastases were identified. Our patient continues to do well with no evidence of recurrence.
1. Bambao C, Nofech-Mozes S, Shier M. Giant condyloma versus verrucous carcinoma: a case report. J Low Genit Tract Dis. 2010;14:230-233.
2. Asiaf A, Ahmad ST, Mohannad SO, et al. Review of the current knowledge on the epidemiology, pathogenesis, and prevention of human papillomavirus infection. Eur J Cancer Prev. 2014;23:206-224.
3. Chaux A, Pfannl R, Rodríguez IM, et al. Distinctive immunohistochemical profile of penile intraepithelial lesions: a study of 74 cases. Am J Surg Pathol. 2011;35:553-562.
4. Darragh TM, Colgan TJ, Cox JT, et al. The lower anogenital squamous terminology standardization project for HPV-associated lesions: background and consensus recommendations from the College of American Pathologists and the American Society for Colposcopy and Cervical Pathology. Arch Pathol Lab Med. 2012;136:1266-1297.
5. Aldabagh B, Angeles J, Cardones AR, et al. Cutaneous squamous cell carcinoma and human papillomavirus: is there an association? Dermatol Surg. 2013;39:1-23.
6. Turk BG, Ertam I, Urkmez A, et al. Development of squamous cell carcinoma on an inflammatory linear verrucous epidermal nevus in the genital area. Cutis. 2012;89:273-275.
7. Erkek E, Basar H, Bozdogan O, et al. Giant condyloma acuminata of Buschke-Löwenstein: successful treatment with a combination of surgical excision, oral acitretin and topical imiquimod. Clin Exp Dermatol. 2009;34:366-368.
8. Cutaneous squamous cell carcinoma and other cutaneous carcinomas. In: Edge SB, Byrd DR, Compton CC, et al, eds. AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer; 2010:301-314.
1. Bambao C, Nofech-Mozes S, Shier M. Giant condyloma versus verrucous carcinoma: a case report. J Low Genit Tract Dis. 2010;14:230-233.
2. Asiaf A, Ahmad ST, Mohannad SO, et al. Review of the current knowledge on the epidemiology, pathogenesis, and prevention of human papillomavirus infection. Eur J Cancer Prev. 2014;23:206-224.
3. Chaux A, Pfannl R, Rodríguez IM, et al. Distinctive immunohistochemical profile of penile intraepithelial lesions: a study of 74 cases. Am J Surg Pathol. 2011;35:553-562.
4. Darragh TM, Colgan TJ, Cox JT, et al. The lower anogenital squamous terminology standardization project for HPV-associated lesions: background and consensus recommendations from the College of American Pathologists and the American Society for Colposcopy and Cervical Pathology. Arch Pathol Lab Med. 2012;136:1266-1297.
5. Aldabagh B, Angeles J, Cardones AR, et al. Cutaneous squamous cell carcinoma and human papillomavirus: is there an association? Dermatol Surg. 2013;39:1-23.
6. Turk BG, Ertam I, Urkmez A, et al. Development of squamous cell carcinoma on an inflammatory linear verrucous epidermal nevus in the genital area. Cutis. 2012;89:273-275.
7. Erkek E, Basar H, Bozdogan O, et al. Giant condyloma acuminata of Buschke-Löwenstein: successful treatment with a combination of surgical excision, oral acitretin and topical imiquimod. Clin Exp Dermatol. 2009;34:366-368.
8. Cutaneous squamous cell carcinoma and other cutaneous carcinomas. In: Edge SB, Byrd DR, Compton CC, et al, eds. AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer; 2010:301-314.