User login
Picosecond-domain laser removes multicolor tattoos
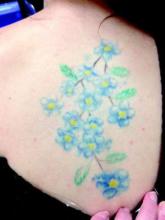
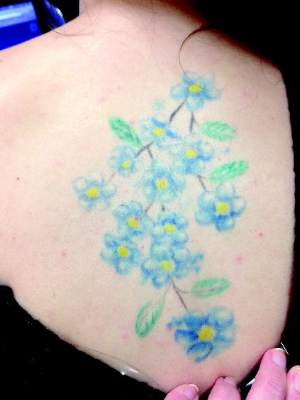
A prototype picosecond-domain Nd:YAG laser was safe and effective at removing multicolor decorative tattoos in a preliminary study, achieving 79% removal in an average of 6.5 treatments, according to a report published in Lasers in Surgery and Medicine.
Nanosecond-domain Q-switched lasers have been the standard tools for tattoo removal for decades, but a new class of the device that generates picosecond-domain pulses was developed to remove tattoos more efficiently. In this prospective study, a prototype device (PicoWay, Syneron-Candela Corporation) was used to remove 31 multicolor tattoos on 21 patients aged 19-55 years (average age, 32 years), according to Dr. Eric F. Bernstein of Main Line Center for Laser Surgery, Ardmore, Pa., and his associates.
All the tattoos were previously untreated and measured no more than 10 cm by 10 cm in area. All were treated through a hydrogel dressing to protect the epidermis and minimize scarring. Treatment sessions were done at 6- to 10-week intervals until the tattoos were cleared or demonstrated a lack of further improvement, for a maximum of 7 treatments.
Three physicians blinded to treatment conditions assessed digital photographs of the treated areas taken at two fixed focal lengths before each treatment session, at 6-10 weeks following each treatment, and at 12 weeks after the final treatment session. This panel of physicians assessed the photographs, which were presented in a randomized order, grading them on percentage improvement for overall clearance and for clearance of each color contained within a given tattoo. The photographs had been taken with a cross-polarized flash, which enhances the view of the tattoo beyond what is normally seen by the naked eye.
Overall, the panel judged that the new device produced 79% clearance after an average of 6.5 treatments. Clearance scores were 92% for black ink, 85% for yellow ink, 80% for red ink, 78% for purple ink, 65% for green ink, and 43% for blue ink. Black and red inks were removed the most effectively, as expected; green and blue inks were more difficult to remove, also as expected. However, the 85% clearance of yellow ink, usually the most difficult color to remove, in an average of four sessions was “surprising and encouraging,” Dr. Bernstein and his associates said.
“It was hoped that picosecond-domain lasers would be ‘color blind’ and remove all colors equally; however, we found this not to be the case,” they noted (Lasers Surg Med. 2015;47[7]:542-8).
Regarding the safety of the new device, purpura was noted immediately after one treatment in one patient but completely resolved by the next session. Mild pinpoint bleeding developed immediately after 14% of sessions, almost always with attendant edema and erythema; all of these effects resolved with time. No immediate blistering was observed in any patient. At 3-month follow-up, no scarring and no moderate to severe pigmentary alterations were noted. Mild hypopigmentation occurred in red or yellow portions of two tattoos, and mild hyperpigmentation occurred in black areas of five tattoos.
“The true benefits of picosecond-domain devices for tattoo removal and other applications should become more apparent as these devices are used more frequently in clinical practice,” the investigators added.
This study was funded by Syneron-Candela Corporation, maker of the prototype picosecond-domain laser tested here. Dr. Bernstein reported serving as a consultant for Syneron-Candela, and two of his associates are employees of the company.
A prototype picosecond-domain Nd:YAG laser was safe and effective at removing multicolor decorative tattoos in a preliminary study, achieving 79% removal in an average of 6.5 treatments, according to a report published in Lasers in Surgery and Medicine.
Nanosecond-domain Q-switched lasers have been the standard tools for tattoo removal for decades, but a new class of the device that generates picosecond-domain pulses was developed to remove tattoos more efficiently. In this prospective study, a prototype device (PicoWay, Syneron-Candela Corporation) was used to remove 31 multicolor tattoos on 21 patients aged 19-55 years (average age, 32 years), according to Dr. Eric F. Bernstein of Main Line Center for Laser Surgery, Ardmore, Pa., and his associates.
All the tattoos were previously untreated and measured no more than 10 cm by 10 cm in area. All were treated through a hydrogel dressing to protect the epidermis and minimize scarring. Treatment sessions were done at 6- to 10-week intervals until the tattoos were cleared or demonstrated a lack of further improvement, for a maximum of 7 treatments.
Three physicians blinded to treatment conditions assessed digital photographs of the treated areas taken at two fixed focal lengths before each treatment session, at 6-10 weeks following each treatment, and at 12 weeks after the final treatment session. This panel of physicians assessed the photographs, which were presented in a randomized order, grading them on percentage improvement for overall clearance and for clearance of each color contained within a given tattoo. The photographs had been taken with a cross-polarized flash, which enhances the view of the tattoo beyond what is normally seen by the naked eye.
Overall, the panel judged that the new device produced 79% clearance after an average of 6.5 treatments. Clearance scores were 92% for black ink, 85% for yellow ink, 80% for red ink, 78% for purple ink, 65% for green ink, and 43% for blue ink. Black and red inks were removed the most effectively, as expected; green and blue inks were more difficult to remove, also as expected. However, the 85% clearance of yellow ink, usually the most difficult color to remove, in an average of four sessions was “surprising and encouraging,” Dr. Bernstein and his associates said.
“It was hoped that picosecond-domain lasers would be ‘color blind’ and remove all colors equally; however, we found this not to be the case,” they noted (Lasers Surg Med. 2015;47[7]:542-8).
Regarding the safety of the new device, purpura was noted immediately after one treatment in one patient but completely resolved by the next session. Mild pinpoint bleeding developed immediately after 14% of sessions, almost always with attendant edema and erythema; all of these effects resolved with time. No immediate blistering was observed in any patient. At 3-month follow-up, no scarring and no moderate to severe pigmentary alterations were noted. Mild hypopigmentation occurred in red or yellow portions of two tattoos, and mild hyperpigmentation occurred in black areas of five tattoos.
“The true benefits of picosecond-domain devices for tattoo removal and other applications should become more apparent as these devices are used more frequently in clinical practice,” the investigators added.
This study was funded by Syneron-Candela Corporation, maker of the prototype picosecond-domain laser tested here. Dr. Bernstein reported serving as a consultant for Syneron-Candela, and two of his associates are employees of the company.
A prototype picosecond-domain Nd:YAG laser was safe and effective at removing multicolor decorative tattoos in a preliminary study, achieving 79% removal in an average of 6.5 treatments, according to a report published in Lasers in Surgery and Medicine.
Nanosecond-domain Q-switched lasers have been the standard tools for tattoo removal for decades, but a new class of the device that generates picosecond-domain pulses was developed to remove tattoos more efficiently. In this prospective study, a prototype device (PicoWay, Syneron-Candela Corporation) was used to remove 31 multicolor tattoos on 21 patients aged 19-55 years (average age, 32 years), according to Dr. Eric F. Bernstein of Main Line Center for Laser Surgery, Ardmore, Pa., and his associates.
All the tattoos were previously untreated and measured no more than 10 cm by 10 cm in area. All were treated through a hydrogel dressing to protect the epidermis and minimize scarring. Treatment sessions were done at 6- to 10-week intervals until the tattoos were cleared or demonstrated a lack of further improvement, for a maximum of 7 treatments.
Three physicians blinded to treatment conditions assessed digital photographs of the treated areas taken at two fixed focal lengths before each treatment session, at 6-10 weeks following each treatment, and at 12 weeks after the final treatment session. This panel of physicians assessed the photographs, which were presented in a randomized order, grading them on percentage improvement for overall clearance and for clearance of each color contained within a given tattoo. The photographs had been taken with a cross-polarized flash, which enhances the view of the tattoo beyond what is normally seen by the naked eye.
Overall, the panel judged that the new device produced 79% clearance after an average of 6.5 treatments. Clearance scores were 92% for black ink, 85% for yellow ink, 80% for red ink, 78% for purple ink, 65% for green ink, and 43% for blue ink. Black and red inks were removed the most effectively, as expected; green and blue inks were more difficult to remove, also as expected. However, the 85% clearance of yellow ink, usually the most difficult color to remove, in an average of four sessions was “surprising and encouraging,” Dr. Bernstein and his associates said.
“It was hoped that picosecond-domain lasers would be ‘color blind’ and remove all colors equally; however, we found this not to be the case,” they noted (Lasers Surg Med. 2015;47[7]:542-8).
Regarding the safety of the new device, purpura was noted immediately after one treatment in one patient but completely resolved by the next session. Mild pinpoint bleeding developed immediately after 14% of sessions, almost always with attendant edema and erythema; all of these effects resolved with time. No immediate blistering was observed in any patient. At 3-month follow-up, no scarring and no moderate to severe pigmentary alterations were noted. Mild hypopigmentation occurred in red or yellow portions of two tattoos, and mild hyperpigmentation occurred in black areas of five tattoos.
“The true benefits of picosecond-domain devices for tattoo removal and other applications should become more apparent as these devices are used more frequently in clinical practice,” the investigators added.
This study was funded by Syneron-Candela Corporation, maker of the prototype picosecond-domain laser tested here. Dr. Bernstein reported serving as a consultant for Syneron-Candela, and two of his associates are employees of the company.
FROM LASERS IN SURGERY AND MEDICINE
Key clinical point: A prototype picosecond-domain laser achieved 79% removal of multicolor tattoos in an average of 6.5 treatments.
Major finding: Removal scores were 92% for black ink, 85% for yellow ink, 80% for red ink, 78% for purple ink, 65% for green ink, and 43% for blue ink.
Data source: A preliminary prospective study of the efficacy and safety of a picosecond-domain laser for removing 36 tattoos on 26 patients.
Disclosures: This study was funded by Syneron-Candela Corporation, maker of the prototype picosecond-domain laser tested here. Dr. Bernstein reported serving as a consultant for Syneron-Candela, and two of his associates are employees of the company.
White tea
White tea, like green tea, is derived from the plant Camellia sinensis, a member of the Theaceae family and the source of all the globally popular “true tea” beverages.
Of the four main true teas, green and white are unfermented (white is the least processed), black tea is fermented, and oolong tea is semifermented.1,2,3 White tea actually comes from the tips of the green tea leaves or leaves that have not yet fully opened, with buds covered by fine white hair. As a commodity, white tea is more expensive than green tea because it is more difficult to obtain. EGCG [(-)epigallocatechin-3-O-gallate], the most abundant and biologically active polyphenolic catechin found in green tea, is also the constituent in white tea that accounts for its antioxidant properties.4,5 Indeed, white tea is included in topical products for its antioxidant as well as antiseptic activity, and is considered a more potent antioxidant additive medium than green tea.6,1
As an ingredient in a combination formula
White tea is included in the dietary supplement Imedeen Prime Renewal, along with fish protein polysaccharides, vitamins C and E, zinc, and extracts from soy, grape seed, chamomile, and tomato.
In 2006, Skovgaard et al. conducted a 6-month, double-blind, placebo-controlled randomized study on 80 healthy postmenopausal women (38 in the treatment group, 42 in the placebo group completed the study) to determine antiaging effects on the skin. Subjects took 2 tablets of the supplement or placebo twice daily. Clinical, photo, and ultrasound evaluations showed significantly greater improvements in the treatment group, compared with the placebo group, in the face (forehead, periocular, and perioral wrinkles; mottled pigmentation, laxity, sagging, dark circles under the eyes; and overall appearance), hands, and décolletage.7
Antioxidant and antiaging activity
In 2009, Thring et al. studied the antiaging and antioxidant characteristics of 23 plant extracts (from 21 species) by considering antielastase and anticollagenase activities. White tea was found to exhibit the greatest inhibitory activity against both elastase and collagenase, greater than burdock root and angelica in terms of antielastase activity, and greater than green tea, rose tincture, and lavender in relation to anticollagenase activity. The Trolox equivalent antioxidant capacity assay also showed that white tea displayed the highest antioxidant activity. The investigators noted the very high phenolic content of white tea in characterizing its potent inhibitory activity against enzymes that accelerate cutaneous aging.6
Earlier in 2009, Camouse et al. examined skin samples from volunteers or skin explants treated with topical white or green tea after ultraviolet exposure to ascertain that the antioxidant could prevent simulated solar radiation–induced damage to DNA and Langerhans cells. They noted that each product displayed a sun protection factor of 1, suggesting that the photoprotection conferred was not due to direct UV absorption. Both forms of topically applied tea extracts were equally effective and judged by the researchers to be potential photoprotective agents when used along with other substantiated approaches to skin protection. These findings provided the first reported evidence of topically applied white tea preventing UV-induced immunosuppression. The researchers further suggested that the color of white tea might render it more cosmetically desirable than green tea.8
It should be noted that a systematic review performed by Hunt et al. in 2010 of MEDLINE, Embase, CINAHL (Cumulative Index to Nursing and Allied Health Literature), CENTRAL (Cochrane Central Register of Controlled Trials), and AMED (Allied and Complementary Medicine Database) databases up to 2009 identified 11 randomized clinical or controlled clinical trials evaluating the effectiveness of botanical extracts for diminishing wrinkling and other signs of cutaneous aging. No significant reductions in wrinkling were associated with the use of green tea or Vitaphenol (a combination of green and white teas, mangosteen, and pomegranate extract). The authors noted, however, that all of the trials that they identified were characterized by poor methodologic quality.9
Thring et al. conducted an in vitro study in 2011 to evaluate the antioxidant and anti-inflammatory activity of white tea, rose, and witch hazel extracts in primary human skin fibroblasts. The investigators measured significant anticollagenase, antielastase, and antioxidant activities for the white tea extracts, which also spurred a significant reduction in the interleukin-8 amount synthesized by fibroblasts, compared with controls. They concluded that white tea (as well as the other extracts) yielded a protective effect on fibroblasts against damage induced by hydrogen peroxide exposure.10
In 2014, Azman et al. used the spin trap method and electron paramagnetic resonance (EPR) spectroscopy to show that among white tea constituents, EGCG and epicatechin-3-gallate (ECG) exhibit the greatest antiradical activity against the methoxy radical.1
Conclusion
Tea is one of the most popular beverages in the world and is touted for its antioxidant and anticancer properties. While the ingredients of green tea polyphenols have inspired a spate of recent research, much is yet to be learned about the potential health benefits of white tea, which is even less processed. Some evidence appears to suggest that white tea may be shown to be more effective overall, and in the dermatologic realm, than green tea. I look forward to seeing more research.
References
1. J Agric Food Chem. 2014;62(1):5743-8.
2. Dermatol Surg. 2005;31(7 Pt 2):873-80.
3. Oxid Med Cell Longev. 2012:2012:560682.
4. Mol Cell Biochem. 2000;206(1-2):125-32.
5. Free Radic Biol Med. 1999;26(11-12):1427-35.
6. BMC Complement Altern Med. 2009;9:27.
7. Eur J Clin Nutr. 2006;60(10):1201-6.
8. Exp Dermatol. 2009;18(6):522-6.
9. Drugs Aging. 2010;27(12):973-85.
10. J Inflamm (Lond). 2011;8(1):27).
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook, “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). She has contributed to the Cosmeceutical Critique column in Dermatology News since January 2001. Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever.
White tea, like green tea, is derived from the plant Camellia sinensis, a member of the Theaceae family and the source of all the globally popular “true tea” beverages.
Of the four main true teas, green and white are unfermented (white is the least processed), black tea is fermented, and oolong tea is semifermented.1,2,3 White tea actually comes from the tips of the green tea leaves or leaves that have not yet fully opened, with buds covered by fine white hair. As a commodity, white tea is more expensive than green tea because it is more difficult to obtain. EGCG [(-)epigallocatechin-3-O-gallate], the most abundant and biologically active polyphenolic catechin found in green tea, is also the constituent in white tea that accounts for its antioxidant properties.4,5 Indeed, white tea is included in topical products for its antioxidant as well as antiseptic activity, and is considered a more potent antioxidant additive medium than green tea.6,1
As an ingredient in a combination formula
White tea is included in the dietary supplement Imedeen Prime Renewal, along with fish protein polysaccharides, vitamins C and E, zinc, and extracts from soy, grape seed, chamomile, and tomato.
In 2006, Skovgaard et al. conducted a 6-month, double-blind, placebo-controlled randomized study on 80 healthy postmenopausal women (38 in the treatment group, 42 in the placebo group completed the study) to determine antiaging effects on the skin. Subjects took 2 tablets of the supplement or placebo twice daily. Clinical, photo, and ultrasound evaluations showed significantly greater improvements in the treatment group, compared with the placebo group, in the face (forehead, periocular, and perioral wrinkles; mottled pigmentation, laxity, sagging, dark circles under the eyes; and overall appearance), hands, and décolletage.7
Antioxidant and antiaging activity
In 2009, Thring et al. studied the antiaging and antioxidant characteristics of 23 plant extracts (from 21 species) by considering antielastase and anticollagenase activities. White tea was found to exhibit the greatest inhibitory activity against both elastase and collagenase, greater than burdock root and angelica in terms of antielastase activity, and greater than green tea, rose tincture, and lavender in relation to anticollagenase activity. The Trolox equivalent antioxidant capacity assay also showed that white tea displayed the highest antioxidant activity. The investigators noted the very high phenolic content of white tea in characterizing its potent inhibitory activity against enzymes that accelerate cutaneous aging.6
Earlier in 2009, Camouse et al. examined skin samples from volunteers or skin explants treated with topical white or green tea after ultraviolet exposure to ascertain that the antioxidant could prevent simulated solar radiation–induced damage to DNA and Langerhans cells. They noted that each product displayed a sun protection factor of 1, suggesting that the photoprotection conferred was not due to direct UV absorption. Both forms of topically applied tea extracts were equally effective and judged by the researchers to be potential photoprotective agents when used along with other substantiated approaches to skin protection. These findings provided the first reported evidence of topically applied white tea preventing UV-induced immunosuppression. The researchers further suggested that the color of white tea might render it more cosmetically desirable than green tea.8
It should be noted that a systematic review performed by Hunt et al. in 2010 of MEDLINE, Embase, CINAHL (Cumulative Index to Nursing and Allied Health Literature), CENTRAL (Cochrane Central Register of Controlled Trials), and AMED (Allied and Complementary Medicine Database) databases up to 2009 identified 11 randomized clinical or controlled clinical trials evaluating the effectiveness of botanical extracts for diminishing wrinkling and other signs of cutaneous aging. No significant reductions in wrinkling were associated with the use of green tea or Vitaphenol (a combination of green and white teas, mangosteen, and pomegranate extract). The authors noted, however, that all of the trials that they identified were characterized by poor methodologic quality.9
Thring et al. conducted an in vitro study in 2011 to evaluate the antioxidant and anti-inflammatory activity of white tea, rose, and witch hazel extracts in primary human skin fibroblasts. The investigators measured significant anticollagenase, antielastase, and antioxidant activities for the white tea extracts, which also spurred a significant reduction in the interleukin-8 amount synthesized by fibroblasts, compared with controls. They concluded that white tea (as well as the other extracts) yielded a protective effect on fibroblasts against damage induced by hydrogen peroxide exposure.10
In 2014, Azman et al. used the spin trap method and electron paramagnetic resonance (EPR) spectroscopy to show that among white tea constituents, EGCG and epicatechin-3-gallate (ECG) exhibit the greatest antiradical activity against the methoxy radical.1
Conclusion
Tea is one of the most popular beverages in the world and is touted for its antioxidant and anticancer properties. While the ingredients of green tea polyphenols have inspired a spate of recent research, much is yet to be learned about the potential health benefits of white tea, which is even less processed. Some evidence appears to suggest that white tea may be shown to be more effective overall, and in the dermatologic realm, than green tea. I look forward to seeing more research.
References
1. J Agric Food Chem. 2014;62(1):5743-8.
2. Dermatol Surg. 2005;31(7 Pt 2):873-80.
3. Oxid Med Cell Longev. 2012:2012:560682.
4. Mol Cell Biochem. 2000;206(1-2):125-32.
5. Free Radic Biol Med. 1999;26(11-12):1427-35.
6. BMC Complement Altern Med. 2009;9:27.
7. Eur J Clin Nutr. 2006;60(10):1201-6.
8. Exp Dermatol. 2009;18(6):522-6.
9. Drugs Aging. 2010;27(12):973-85.
10. J Inflamm (Lond). 2011;8(1):27).
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook, “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). She has contributed to the Cosmeceutical Critique column in Dermatology News since January 2001. Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever.
White tea, like green tea, is derived from the plant Camellia sinensis, a member of the Theaceae family and the source of all the globally popular “true tea” beverages.
Of the four main true teas, green and white are unfermented (white is the least processed), black tea is fermented, and oolong tea is semifermented.1,2,3 White tea actually comes from the tips of the green tea leaves or leaves that have not yet fully opened, with buds covered by fine white hair. As a commodity, white tea is more expensive than green tea because it is more difficult to obtain. EGCG [(-)epigallocatechin-3-O-gallate], the most abundant and biologically active polyphenolic catechin found in green tea, is also the constituent in white tea that accounts for its antioxidant properties.4,5 Indeed, white tea is included in topical products for its antioxidant as well as antiseptic activity, and is considered a more potent antioxidant additive medium than green tea.6,1
As an ingredient in a combination formula
White tea is included in the dietary supplement Imedeen Prime Renewal, along with fish protein polysaccharides, vitamins C and E, zinc, and extracts from soy, grape seed, chamomile, and tomato.
In 2006, Skovgaard et al. conducted a 6-month, double-blind, placebo-controlled randomized study on 80 healthy postmenopausal women (38 in the treatment group, 42 in the placebo group completed the study) to determine antiaging effects on the skin. Subjects took 2 tablets of the supplement or placebo twice daily. Clinical, photo, and ultrasound evaluations showed significantly greater improvements in the treatment group, compared with the placebo group, in the face (forehead, periocular, and perioral wrinkles; mottled pigmentation, laxity, sagging, dark circles under the eyes; and overall appearance), hands, and décolletage.7
Antioxidant and antiaging activity
In 2009, Thring et al. studied the antiaging and antioxidant characteristics of 23 plant extracts (from 21 species) by considering antielastase and anticollagenase activities. White tea was found to exhibit the greatest inhibitory activity against both elastase and collagenase, greater than burdock root and angelica in terms of antielastase activity, and greater than green tea, rose tincture, and lavender in relation to anticollagenase activity. The Trolox equivalent antioxidant capacity assay also showed that white tea displayed the highest antioxidant activity. The investigators noted the very high phenolic content of white tea in characterizing its potent inhibitory activity against enzymes that accelerate cutaneous aging.6
Earlier in 2009, Camouse et al. examined skin samples from volunteers or skin explants treated with topical white or green tea after ultraviolet exposure to ascertain that the antioxidant could prevent simulated solar radiation–induced damage to DNA and Langerhans cells. They noted that each product displayed a sun protection factor of 1, suggesting that the photoprotection conferred was not due to direct UV absorption. Both forms of topically applied tea extracts were equally effective and judged by the researchers to be potential photoprotective agents when used along with other substantiated approaches to skin protection. These findings provided the first reported evidence of topically applied white tea preventing UV-induced immunosuppression. The researchers further suggested that the color of white tea might render it more cosmetically desirable than green tea.8
It should be noted that a systematic review performed by Hunt et al. in 2010 of MEDLINE, Embase, CINAHL (Cumulative Index to Nursing and Allied Health Literature), CENTRAL (Cochrane Central Register of Controlled Trials), and AMED (Allied and Complementary Medicine Database) databases up to 2009 identified 11 randomized clinical or controlled clinical trials evaluating the effectiveness of botanical extracts for diminishing wrinkling and other signs of cutaneous aging. No significant reductions in wrinkling were associated with the use of green tea or Vitaphenol (a combination of green and white teas, mangosteen, and pomegranate extract). The authors noted, however, that all of the trials that they identified were characterized by poor methodologic quality.9
Thring et al. conducted an in vitro study in 2011 to evaluate the antioxidant and anti-inflammatory activity of white tea, rose, and witch hazel extracts in primary human skin fibroblasts. The investigators measured significant anticollagenase, antielastase, and antioxidant activities for the white tea extracts, which also spurred a significant reduction in the interleukin-8 amount synthesized by fibroblasts, compared with controls. They concluded that white tea (as well as the other extracts) yielded a protective effect on fibroblasts against damage induced by hydrogen peroxide exposure.10
In 2014, Azman et al. used the spin trap method and electron paramagnetic resonance (EPR) spectroscopy to show that among white tea constituents, EGCG and epicatechin-3-gallate (ECG) exhibit the greatest antiradical activity against the methoxy radical.1
Conclusion
Tea is one of the most popular beverages in the world and is touted for its antioxidant and anticancer properties. While the ingredients of green tea polyphenols have inspired a spate of recent research, much is yet to be learned about the potential health benefits of white tea, which is even less processed. Some evidence appears to suggest that white tea may be shown to be more effective overall, and in the dermatologic realm, than green tea. I look forward to seeing more research.
References
1. J Agric Food Chem. 2014;62(1):5743-8.
2. Dermatol Surg. 2005;31(7 Pt 2):873-80.
3. Oxid Med Cell Longev. 2012:2012:560682.
4. Mol Cell Biochem. 2000;206(1-2):125-32.
5. Free Radic Biol Med. 1999;26(11-12):1427-35.
6. BMC Complement Altern Med. 2009;9:27.
7. Eur J Clin Nutr. 2006;60(10):1201-6.
8. Exp Dermatol. 2009;18(6):522-6.
9. Drugs Aging. 2010;27(12):973-85.
10. J Inflamm (Lond). 2011;8(1):27).
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook, “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). She has contributed to the Cosmeceutical Critique column in Dermatology News since January 2001. Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever.
A Novel Method of Skin Closure for Aging or Fragile Skin
Patients who have been on steroids, aspirin, or anticoagulants or who are elderly may have a fragile outer skin layer that is similar to parchment paper, which may be challenging for surgeons. In these patients, the epidermal layer is thin and translucent; when a surgeon cuts through this thin layer, the tissue beneath shows minimal dermis and poor-quality fat with weakened tissue support. When undergoing excisional surgery, there is no strong tissue to help the closure sutures remain intact. Surgeons may struggle with skin tears around the sutures and dehiscence on suture removal.
This article describes a novel approach to skin closure in patients with aging or thin skin using a polyethylene film with an acrylate adhesive in the excision area to aid in maintaining skin integrity throughout the healing process following surgery.
Closure Technique
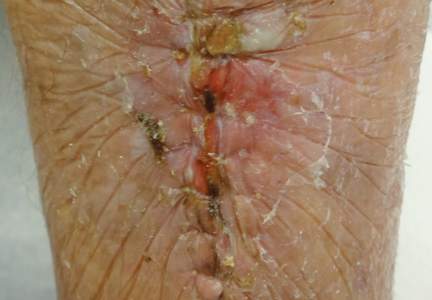
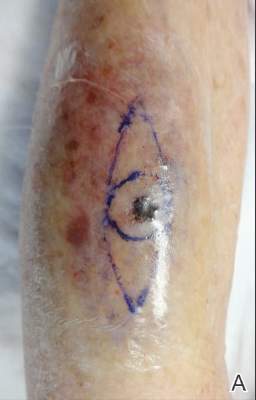
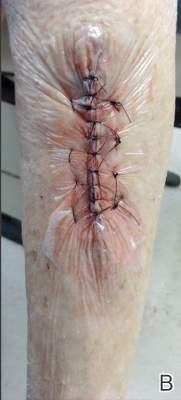
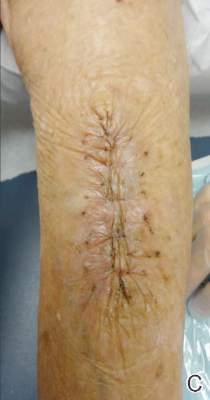
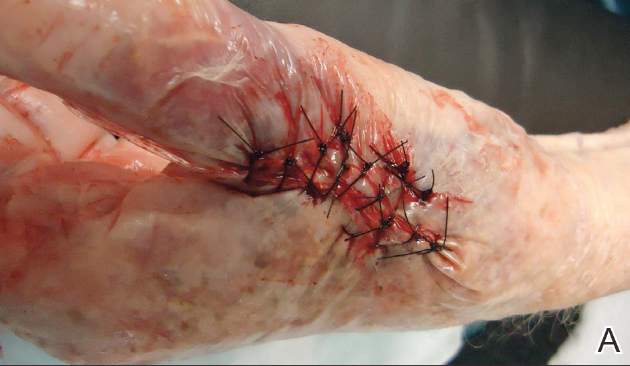
First, the skin area is cleansed with a sterilizing soap preparation. A sterile marking pen then is used to outline the excision area. A 10×12-cm layer of polyethylene film is then attached to the excision site. Excision of the tumor is performed by cutting through the film in the marked area (Figure 1A), and closure is performed by suturing the wound edges through the polyethylene film while the area is still covered with the film (Figure 1B). The sutures can be left in for 2 weeks or longer if necessary. The patient should be instructed not to remove the film or perform any extensive cleansing of the treatment area. Antibiotics should be administered, as the polyethylene film maintains its sterile integrity for 7 days only. Because sutures are on the surface of the film, they are easily accessed for removal. Figure 1C shows the excision site after removal of the sutures and polyethylene film on the left tibia of a 95-year-old woman. Adhesive butterfly closures can be applied to strengthen the excision area after suture removal and prevent dehiscence.
|
Figure 1. The excision site was marked after polyethylene adhesive film was applied to a squamous cell carcinoma on the left tibia of 95-year-old woman (A). Closure was performed by suturing the wound edges through the polyethylene film (B). The excision site appeared to have no dehiscence or signs of infection after removal of the sutures and polyethylene film (C). |
Case Reports
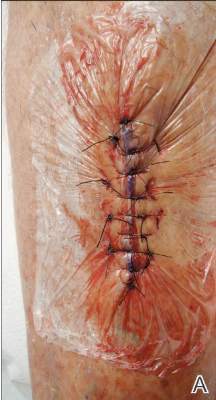
Twelve procedures for skin cancer excision were conducted in 10 patients using polyethylene adhesive film as a surgical aid due to extremely poor quality of the epidermis. The tumors were all squamous cell carcinomas and were located on the arms and legs. Patients were aged 73 to 95 years. Figure 2 demonstrates an example of excision of a squamous cell carcinoma on the left tibia of an 82-year-old man with prior dehiscence and infection after leg surgeries. Good results were achieved using the closure technique described here, along with prophylactic antibiotics.
|
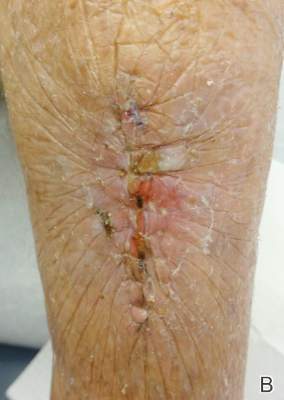
Figure 2. A squamous cell carcinoma excision site on the left tibia of an 82-year-old man that had been covered with polyethylene adhesive film prior to excision (A) and 17 days following removal of the sutures and film (B). |
One patient had complications from a Staphylococcus infection because antibiotics were not administered. The patient had prior infections with other surgeries. Antibiotics were given 4 days after surgery. The infection was cleared and the polyethylene film was retained for a total of 12 days.
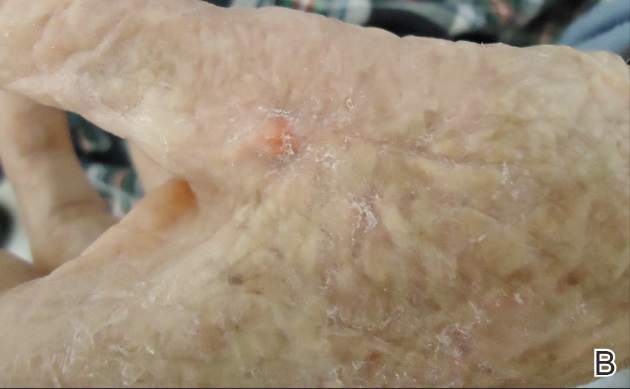
Sutures were removed after 14 days for excision sites on the arms and 17 days for excision sites on the legs. All excision sites healed without dehiscence with a cosmetically acceptable scar. Figure 3A shows a completed excision on the left hand of a 92-year-old man, and Figure 3B is the result 5 weeks after excision.
|
Figure 3. A squamous cell carcinoma excision site on the left thumb of a 92-year-old man that had been covered with polyethylene adhesive film prior to exci- sion (A). No visible scarring or dehiscence was noted 5 weeks after excision, following removal of the sutures and film (B). |
None of the patients reported discomfort from the polyethylene film remaining on the skin following surgery, though postoperative care required extra caution when dressing so as not to disturb or compromise the film. Patients were advised about postoperative care and were instructed not to remove the dressing. They were all given antibiotics as a necessary adjunct to maintain a lessened bacteria burden imposed by an impervious layer of acrylate adhesive. Complications resulted from failure to immediately provide antibiotics to 1 patient. The polyethylene film did not hinder healing or postoperative results.
Comment
Various techniques for handling fragile skin during surgery have been described in the literature. Fomon et al1 discussed aging skin as it relates to plastic surgery. Foster and Chan2 described a skin support technique for closing elliptical incisions in patients with fragile skin. Mazzurco and Krach3 discussed the use of a hydrocolloid dressing to aid in the closure of surgical wounds in patients with fragile skin.
The closure method described here was found to be particularly helpful when used as an adjunct to surgery in patients with fragile skin that lacked a suitable dermis. The polyethylene adhesive film helped to hold the sutures more securely. This method is cost-effective and is associated with a high level of patient satisfaction. For the surgeon, this technique may aid in dealing with difficult surgical situations and helps prevent wound complications in elderly patients or those with fragile skin.
1. Fomon S, Bell JW, Schattner A. Aging skin, a surgical challenge. AMA Arch Otolaryngol. 1955;61:554-562.
2. Foster RS, Chan J. The Fixomull skin support method for wound closure in patients with fragile skin. Australas J Dermatol. 2011;52:209-211.
3. Mazzurco JD, Krach KJ. Use of a hydrocolloid dressing to aid in the closure of surgical wounds in patients with fragile skin. J Am Acad Dermatol. 2012;66:335-336.
Patients who have been on steroids, aspirin, or anticoagulants or who are elderly may have a fragile outer skin layer that is similar to parchment paper, which may be challenging for surgeons. In these patients, the epidermal layer is thin and translucent; when a surgeon cuts through this thin layer, the tissue beneath shows minimal dermis and poor-quality fat with weakened tissue support. When undergoing excisional surgery, there is no strong tissue to help the closure sutures remain intact. Surgeons may struggle with skin tears around the sutures and dehiscence on suture removal.
This article describes a novel approach to skin closure in patients with aging or thin skin using a polyethylene film with an acrylate adhesive in the excision area to aid in maintaining skin integrity throughout the healing process following surgery.
Closure Technique
First, the skin area is cleansed with a sterilizing soap preparation. A sterile marking pen then is used to outline the excision area. A 10×12-cm layer of polyethylene film is then attached to the excision site. Excision of the tumor is performed by cutting through the film in the marked area (Figure 1A), and closure is performed by suturing the wound edges through the polyethylene film while the area is still covered with the film (Figure 1B). The sutures can be left in for 2 weeks or longer if necessary. The patient should be instructed not to remove the film or perform any extensive cleansing of the treatment area. Antibiotics should be administered, as the polyethylene film maintains its sterile integrity for 7 days only. Because sutures are on the surface of the film, they are easily accessed for removal. Figure 1C shows the excision site after removal of the sutures and polyethylene film on the left tibia of a 95-year-old woman. Adhesive butterfly closures can be applied to strengthen the excision area after suture removal and prevent dehiscence.



|
Figure 1. The excision site was marked after polyethylene adhesive film was applied to a squamous cell carcinoma on the left tibia of 95-year-old woman (A). Closure was performed by suturing the wound edges through the polyethylene film (B). The excision site appeared to have no dehiscence or signs of infection after removal of the sutures and polyethylene film (C). |
Case Reports
Twelve procedures for skin cancer excision were conducted in 10 patients using polyethylene adhesive film as a surgical aid due to extremely poor quality of the epidermis. The tumors were all squamous cell carcinomas and were located on the arms and legs. Patients were aged 73 to 95 years. Figure 2 demonstrates an example of excision of a squamous cell carcinoma on the left tibia of an 82-year-old man with prior dehiscence and infection after leg surgeries. Good results were achieved using the closure technique described here, along with prophylactic antibiotics.


|
Figure 2. A squamous cell carcinoma excision site on the left tibia of an 82-year-old man that had been covered with polyethylene adhesive film prior to excision (A) and 17 days following removal of the sutures and film (B). |
One patient had complications from a Staphylococcus infection because antibiotics were not administered. The patient had prior infections with other surgeries. Antibiotics were given 4 days after surgery. The infection was cleared and the polyethylene film was retained for a total of 12 days.
Sutures were removed after 14 days for excision sites on the arms and 17 days for excision sites on the legs. All excision sites healed without dehiscence with a cosmetically acceptable scar. Figure 3A shows a completed excision on the left hand of a 92-year-old man, and Figure 3B is the result 5 weeks after excision.


|
Figure 3. A squamous cell carcinoma excision site on the left thumb of a 92-year-old man that had been covered with polyethylene adhesive film prior to exci- sion (A). No visible scarring or dehiscence was noted 5 weeks after excision, following removal of the sutures and film (B). |
None of the patients reported discomfort from the polyethylene film remaining on the skin following surgery, though postoperative care required extra caution when dressing so as not to disturb or compromise the film. Patients were advised about postoperative care and were instructed not to remove the dressing. They were all given antibiotics as a necessary adjunct to maintain a lessened bacteria burden imposed by an impervious layer of acrylate adhesive. Complications resulted from failure to immediately provide antibiotics to 1 patient. The polyethylene film did not hinder healing or postoperative results.
Comment
Various techniques for handling fragile skin during surgery have been described in the literature. Fomon et al1 discussed aging skin as it relates to plastic surgery. Foster and Chan2 described a skin support technique for closing elliptical incisions in patients with fragile skin. Mazzurco and Krach3 discussed the use of a hydrocolloid dressing to aid in the closure of surgical wounds in patients with fragile skin.
The closure method described here was found to be particularly helpful when used as an adjunct to surgery in patients with fragile skin that lacked a suitable dermis. The polyethylene adhesive film helped to hold the sutures more securely. This method is cost-effective and is associated with a high level of patient satisfaction. For the surgeon, this technique may aid in dealing with difficult surgical situations and helps prevent wound complications in elderly patients or those with fragile skin.
Patients who have been on steroids, aspirin, or anticoagulants or who are elderly may have a fragile outer skin layer that is similar to parchment paper, which may be challenging for surgeons. In these patients, the epidermal layer is thin and translucent; when a surgeon cuts through this thin layer, the tissue beneath shows minimal dermis and poor-quality fat with weakened tissue support. When undergoing excisional surgery, there is no strong tissue to help the closure sutures remain intact. Surgeons may struggle with skin tears around the sutures and dehiscence on suture removal.
This article describes a novel approach to skin closure in patients with aging or thin skin using a polyethylene film with an acrylate adhesive in the excision area to aid in maintaining skin integrity throughout the healing process following surgery.
Closure Technique
First, the skin area is cleansed with a sterilizing soap preparation. A sterile marking pen then is used to outline the excision area. A 10×12-cm layer of polyethylene film is then attached to the excision site. Excision of the tumor is performed by cutting through the film in the marked area (Figure 1A), and closure is performed by suturing the wound edges through the polyethylene film while the area is still covered with the film (Figure 1B). The sutures can be left in for 2 weeks or longer if necessary. The patient should be instructed not to remove the film or perform any extensive cleansing of the treatment area. Antibiotics should be administered, as the polyethylene film maintains its sterile integrity for 7 days only. Because sutures are on the surface of the film, they are easily accessed for removal. Figure 1C shows the excision site after removal of the sutures and polyethylene film on the left tibia of a 95-year-old woman. Adhesive butterfly closures can be applied to strengthen the excision area after suture removal and prevent dehiscence.



|
Figure 1. The excision site was marked after polyethylene adhesive film was applied to a squamous cell carcinoma on the left tibia of 95-year-old woman (A). Closure was performed by suturing the wound edges through the polyethylene film (B). The excision site appeared to have no dehiscence or signs of infection after removal of the sutures and polyethylene film (C). |
Case Reports
Twelve procedures for skin cancer excision were conducted in 10 patients using polyethylene adhesive film as a surgical aid due to extremely poor quality of the epidermis. The tumors were all squamous cell carcinomas and were located on the arms and legs. Patients were aged 73 to 95 years. Figure 2 demonstrates an example of excision of a squamous cell carcinoma on the left tibia of an 82-year-old man with prior dehiscence and infection after leg surgeries. Good results were achieved using the closure technique described here, along with prophylactic antibiotics.


|
Figure 2. A squamous cell carcinoma excision site on the left tibia of an 82-year-old man that had been covered with polyethylene adhesive film prior to excision (A) and 17 days following removal of the sutures and film (B). |
One patient had complications from a Staphylococcus infection because antibiotics were not administered. The patient had prior infections with other surgeries. Antibiotics were given 4 days after surgery. The infection was cleared and the polyethylene film was retained for a total of 12 days.
Sutures were removed after 14 days for excision sites on the arms and 17 days for excision sites on the legs. All excision sites healed without dehiscence with a cosmetically acceptable scar. Figure 3A shows a completed excision on the left hand of a 92-year-old man, and Figure 3B is the result 5 weeks after excision.


|
Figure 3. A squamous cell carcinoma excision site on the left thumb of a 92-year-old man that had been covered with polyethylene adhesive film prior to exci- sion (A). No visible scarring or dehiscence was noted 5 weeks after excision, following removal of the sutures and film (B). |
None of the patients reported discomfort from the polyethylene film remaining on the skin following surgery, though postoperative care required extra caution when dressing so as not to disturb or compromise the film. Patients were advised about postoperative care and were instructed not to remove the dressing. They were all given antibiotics as a necessary adjunct to maintain a lessened bacteria burden imposed by an impervious layer of acrylate adhesive. Complications resulted from failure to immediately provide antibiotics to 1 patient. The polyethylene film did not hinder healing or postoperative results.
Comment
Various techniques for handling fragile skin during surgery have been described in the literature. Fomon et al1 discussed aging skin as it relates to plastic surgery. Foster and Chan2 described a skin support technique for closing elliptical incisions in patients with fragile skin. Mazzurco and Krach3 discussed the use of a hydrocolloid dressing to aid in the closure of surgical wounds in patients with fragile skin.
The closure method described here was found to be particularly helpful when used as an adjunct to surgery in patients with fragile skin that lacked a suitable dermis. The polyethylene adhesive film helped to hold the sutures more securely. This method is cost-effective and is associated with a high level of patient satisfaction. For the surgeon, this technique may aid in dealing with difficult surgical situations and helps prevent wound complications in elderly patients or those with fragile skin.
1. Fomon S, Bell JW, Schattner A. Aging skin, a surgical challenge. AMA Arch Otolaryngol. 1955;61:554-562.
2. Foster RS, Chan J. The Fixomull skin support method for wound closure in patients with fragile skin. Australas J Dermatol. 2011;52:209-211.
3. Mazzurco JD, Krach KJ. Use of a hydrocolloid dressing to aid in the closure of surgical wounds in patients with fragile skin. J Am Acad Dermatol. 2012;66:335-336.
1. Fomon S, Bell JW, Schattner A. Aging skin, a surgical challenge. AMA Arch Otolaryngol. 1955;61:554-562.
2. Foster RS, Chan J. The Fixomull skin support method for wound closure in patients with fragile skin. Australas J Dermatol. 2011;52:209-211.
3. Mazzurco JD, Krach KJ. Use of a hydrocolloid dressing to aid in the closure of surgical wounds in patients with fragile skin. J Am Acad Dermatol. 2012;66:335-336.
Practice Points
- A novel method of skin closure using a polyethylene film with an acrylate adhesive can aid in strengthening suture integrity and preventing skin tears.
- Dehiscence of excision sites in patients with aging or fragile skin can be prevented.
- This closure technique promotes healing and efficient scar formation.
Small study: Physicians sued for failing to supervise soft-tissue filler procedures
In medical malpractice claims involving soft-tissue fillers, dermatologists and other doctors were frequently named as defendants for allegedly failing to supervise nonphysicians who did the procedures, in a study published in the October issue of the Journal of the American Academy of Dermatology.
In a study of 24 legal and administrative actions, physicians were sued in 13 of the cases, although physician extenders injected the fillers in half of those actions. The results were reported in a research letter (J Am Acad Dermatol. 2015 Oct;73[4]:702-4).
Dr. Navid Ezra, a private practice dermatologist and former resident at Indiana University, Indianapolis, and colleagues analyzed public legal documents from 1995 to 2013 using the national legal research service WestlawNext. They searched three database categories: cases, trial court orders, and administrative guidance and decisions. The categories included opinions by state and federal trial, appellate, and supreme courts, as well as regulatory agencies such as medical boards. A total of 24 legal documents involving soft-tissue fillers were identified – 19 legal cases and 5 disciplinary actions.
Dermatologists and plastic surgeons were sued most often, each accounting for 17% of cases respectively, according to the study. Family practice physicians were named as defendants in 8% of cases. Radiologists, pediatricians, internists, oculoplastic surgeons, and physical medicine/rehabilitation doctors were each named as defendants in 4% of the cases, respectively. Nonphysicians were sued in 25% of cases. Many of the lawsuits named multiple defendants.
The most common injury that resulted in litigation was granuloma formation or other autoimmune reaction. Zyderm was the soft-tissue filler most commonly associated with litigation. However, the authors noted that Zyderm was the first soft-tissue filler approved and temporal bias likely explains the higher number of associated cases.
Of the five disciplinary actions, the majority of doctors were reprimanded for not being present while a nonphysician employee injected patients with soft-tissue fillers. In three of the five reprimands, physicians were functioning as medical directors of medical spas.
The authors concluded that the medico-legal culture is such that physicians are responsible for actions by their physician extenders. They called for further research to evaluate whether the presence of a physician during soft-tissue filler procedures affects the rate of developing complications. They noted that the study’s total number of cases and disciplinary actions underreports the true incidence of legal events related to soft-tissue fillers. The sample size did not include settlements or cases that did not involve an appeal.
On Twitter @legal_med
In medical malpractice claims involving soft-tissue fillers, dermatologists and other doctors were frequently named as defendants for allegedly failing to supervise nonphysicians who did the procedures, in a study published in the October issue of the Journal of the American Academy of Dermatology.
In a study of 24 legal and administrative actions, physicians were sued in 13 of the cases, although physician extenders injected the fillers in half of those actions. The results were reported in a research letter (J Am Acad Dermatol. 2015 Oct;73[4]:702-4).
Dr. Navid Ezra, a private practice dermatologist and former resident at Indiana University, Indianapolis, and colleagues analyzed public legal documents from 1995 to 2013 using the national legal research service WestlawNext. They searched three database categories: cases, trial court orders, and administrative guidance and decisions. The categories included opinions by state and federal trial, appellate, and supreme courts, as well as regulatory agencies such as medical boards. A total of 24 legal documents involving soft-tissue fillers were identified – 19 legal cases and 5 disciplinary actions.
Dermatologists and plastic surgeons were sued most often, each accounting for 17% of cases respectively, according to the study. Family practice physicians were named as defendants in 8% of cases. Radiologists, pediatricians, internists, oculoplastic surgeons, and physical medicine/rehabilitation doctors were each named as defendants in 4% of the cases, respectively. Nonphysicians were sued in 25% of cases. Many of the lawsuits named multiple defendants.
The most common injury that resulted in litigation was granuloma formation or other autoimmune reaction. Zyderm was the soft-tissue filler most commonly associated with litigation. However, the authors noted that Zyderm was the first soft-tissue filler approved and temporal bias likely explains the higher number of associated cases.
Of the five disciplinary actions, the majority of doctors were reprimanded for not being present while a nonphysician employee injected patients with soft-tissue fillers. In three of the five reprimands, physicians were functioning as medical directors of medical spas.
The authors concluded that the medico-legal culture is such that physicians are responsible for actions by their physician extenders. They called for further research to evaluate whether the presence of a physician during soft-tissue filler procedures affects the rate of developing complications. They noted that the study’s total number of cases and disciplinary actions underreports the true incidence of legal events related to soft-tissue fillers. The sample size did not include settlements or cases that did not involve an appeal.
On Twitter @legal_med
In medical malpractice claims involving soft-tissue fillers, dermatologists and other doctors were frequently named as defendants for allegedly failing to supervise nonphysicians who did the procedures, in a study published in the October issue of the Journal of the American Academy of Dermatology.
In a study of 24 legal and administrative actions, physicians were sued in 13 of the cases, although physician extenders injected the fillers in half of those actions. The results were reported in a research letter (J Am Acad Dermatol. 2015 Oct;73[4]:702-4).
Dr. Navid Ezra, a private practice dermatologist and former resident at Indiana University, Indianapolis, and colleagues analyzed public legal documents from 1995 to 2013 using the national legal research service WestlawNext. They searched three database categories: cases, trial court orders, and administrative guidance and decisions. The categories included opinions by state and federal trial, appellate, and supreme courts, as well as regulatory agencies such as medical boards. A total of 24 legal documents involving soft-tissue fillers were identified – 19 legal cases and 5 disciplinary actions.
Dermatologists and plastic surgeons were sued most often, each accounting for 17% of cases respectively, according to the study. Family practice physicians were named as defendants in 8% of cases. Radiologists, pediatricians, internists, oculoplastic surgeons, and physical medicine/rehabilitation doctors were each named as defendants in 4% of the cases, respectively. Nonphysicians were sued in 25% of cases. Many of the lawsuits named multiple defendants.
The most common injury that resulted in litigation was granuloma formation or other autoimmune reaction. Zyderm was the soft-tissue filler most commonly associated with litigation. However, the authors noted that Zyderm was the first soft-tissue filler approved and temporal bias likely explains the higher number of associated cases.
Of the five disciplinary actions, the majority of doctors were reprimanded for not being present while a nonphysician employee injected patients with soft-tissue fillers. In three of the five reprimands, physicians were functioning as medical directors of medical spas.
The authors concluded that the medico-legal culture is such that physicians are responsible for actions by their physician extenders. They called for further research to evaluate whether the presence of a physician during soft-tissue filler procedures affects the rate of developing complications. They noted that the study’s total number of cases and disciplinary actions underreports the true incidence of legal events related to soft-tissue fillers. The sample size did not include settlements or cases that did not involve an appeal.
On Twitter @legal_med
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Key clinical point: A study shows that some medical malpractice claims involving soft-tissue fillers result from procedures performed by nonphysicians, but dermatologists and plastic surgeons are frequently named as defendants for allegedly failing to supervise.
Major finding: In a study of 24 legal and administrative actions, physicians were sued in 13 of the cases, although physician extenders injected the fillers in half of those actions.
Data source: The cases were identified in a search for relevant public legal documents in a national legal database from 1995 to 2013.
Disclosures: The authors had no relevant disclosures; and there was no funding source.
Cosmetic Corner: Dermatologists Weigh in on Eye Creams
To improve patient care and outcomes, leading dermatologists offered their recommendations on the top eye creams. Consideration must be given to:
- CeraVe Eye Repair Cream
Valeant Pharmaceuticals North America LLC
—Recommended by Gary Goldenberg, MD, New York, New York
- No7 Protect & Perfect Intense ADVANCED Eye Cream
The Boots Company PLC
“Contains the new ingredient Matrixyl 3000, which [the manufacturer] showed improved fine lines in [its] clinical studies.”—Anthony M. Rossi, MD, New York, New York Powerful Wrinkle Reducing Eye Cream
- Powerful Wrinkle Reducing Eye Cream
Kiehl's
—Recommended by Gary Goldenberg, MD, New York, New York
- Tensage Radiance Eye Cream
Biopelle, Inc.
“I like that fact that it is a growth factor product that goes on elegantly and has a tint to help camouflage under-eye circles.” —Joel L. Cohen, MD, Englewood, Colorado.
Cutis invites readers to send us their recommendations. Cuticle creams, men’s shaving products, antiaging products, and products for babies will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to cutis@frontlinemedcom.com.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on the top eye creams. Consideration must be given to:
- CeraVe Eye Repair Cream
Valeant Pharmaceuticals North America LLC
—Recommended by Gary Goldenberg, MD, New York, New York
- No7 Protect & Perfect Intense ADVANCED Eye Cream
The Boots Company PLC
“Contains the new ingredient Matrixyl 3000, which [the manufacturer] showed improved fine lines in [its] clinical studies.”—Anthony M. Rossi, MD, New York, New York Powerful Wrinkle Reducing Eye Cream
- Powerful Wrinkle Reducing Eye Cream
Kiehl's
—Recommended by Gary Goldenberg, MD, New York, New York
- Tensage Radiance Eye Cream
Biopelle, Inc.
“I like that fact that it is a growth factor product that goes on elegantly and has a tint to help camouflage under-eye circles.” —Joel L. Cohen, MD, Englewood, Colorado.
Cutis invites readers to send us their recommendations. Cuticle creams, men’s shaving products, antiaging products, and products for babies will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to cutis@frontlinemedcom.com.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on the top eye creams. Consideration must be given to:
- CeraVe Eye Repair Cream
Valeant Pharmaceuticals North America LLC
—Recommended by Gary Goldenberg, MD, New York, New York
- No7 Protect & Perfect Intense ADVANCED Eye Cream
The Boots Company PLC
“Contains the new ingredient Matrixyl 3000, which [the manufacturer] showed improved fine lines in [its] clinical studies.”—Anthony M. Rossi, MD, New York, New York Powerful Wrinkle Reducing Eye Cream
- Powerful Wrinkle Reducing Eye Cream
Kiehl's
—Recommended by Gary Goldenberg, MD, New York, New York
- Tensage Radiance Eye Cream
Biopelle, Inc.
“I like that fact that it is a growth factor product that goes on elegantly and has a tint to help camouflage under-eye circles.” —Joel L. Cohen, MD, Englewood, Colorado.
Cutis invites readers to send us their recommendations. Cuticle creams, men’s shaving products, antiaging products, and products for babies will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to cutis@frontlinemedcom.com.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
Filler approved for lip augmentation
The Food and Drug Administration has approved a Juvederm product for lip augmentation, according to the manufacturer.
In an Oct. 1 press release, Allergan announced that Juvederm Ultra XC had been approved for “injection into the lips and perioral area for lip augmentation in adults over the age of 21.”
The product is described as a smooth gel formulation made up of a modified form of hyaluronic acid; it also contains lidocaine. In clinical studies, 79% of participants “showed a meaningful improvement in lip fullness” 3 months after treatment, the company said, and more than 78% “reported an improvement in their overall satisfaction with the look and feel of their lips” 1 year after treatment.
The Food and Drug Administration has approved a Juvederm product for lip augmentation, according to the manufacturer.
In an Oct. 1 press release, Allergan announced that Juvederm Ultra XC had been approved for “injection into the lips and perioral area for lip augmentation in adults over the age of 21.”
The product is described as a smooth gel formulation made up of a modified form of hyaluronic acid; it also contains lidocaine. In clinical studies, 79% of participants “showed a meaningful improvement in lip fullness” 3 months after treatment, the company said, and more than 78% “reported an improvement in their overall satisfaction with the look and feel of their lips” 1 year after treatment.
The Food and Drug Administration has approved a Juvederm product for lip augmentation, according to the manufacturer.
In an Oct. 1 press release, Allergan announced that Juvederm Ultra XC had been approved for “injection into the lips and perioral area for lip augmentation in adults over the age of 21.”
The product is described as a smooth gel formulation made up of a modified form of hyaluronic acid; it also contains lidocaine. In clinical studies, 79% of participants “showed a meaningful improvement in lip fullness” 3 months after treatment, the company said, and more than 78% “reported an improvement in their overall satisfaction with the look and feel of their lips” 1 year after treatment.
Filler approved for lip augmentation
The Food and Drug Administration has approved a Juvederm product for lip augmentation, according to the manufacturer.
In an Oct. 1 press release, Allergan announced that Juvederm Ultra XC had been approved for “injection into the lips and perioral area for lip augmentation in adults over the age of 21.”
The product is described as a smooth gel formulation made up of a modified form of hyaluronic acid; it also contains lidocaine. In clinical studies, 79% of participants “showed a meaningful improvement in lip fullness” 3 months after treatment, the company said, and more than 78% “reported an improvement in their overall satisfaction with the look and feel of their lips” 1 year after treatment.
The Food and Drug Administration has approved a Juvederm product for lip augmentation, according to the manufacturer.
In an Oct. 1 press release, Allergan announced that Juvederm Ultra XC had been approved for “injection into the lips and perioral area for lip augmentation in adults over the age of 21.”
The product is described as a smooth gel formulation made up of a modified form of hyaluronic acid; it also contains lidocaine. In clinical studies, 79% of participants “showed a meaningful improvement in lip fullness” 3 months after treatment, the company said, and more than 78% “reported an improvement in their overall satisfaction with the look and feel of their lips” 1 year after treatment.
The Food and Drug Administration has approved a Juvederm product for lip augmentation, according to the manufacturer.
In an Oct. 1 press release, Allergan announced that Juvederm Ultra XC had been approved for “injection into the lips and perioral area for lip augmentation in adults over the age of 21.”
The product is described as a smooth gel formulation made up of a modified form of hyaluronic acid; it also contains lidocaine. In clinical studies, 79% of participants “showed a meaningful improvement in lip fullness” 3 months after treatment, the company said, and more than 78% “reported an improvement in their overall satisfaction with the look and feel of their lips” 1 year after treatment.
Nail care safety
I recently went to a local salon for a manicure, and when I asked the manicurist not to cut my cuticles, she looked at me as though I was offending her. Shortly thereafter, I took a phone call that swayed my attention, and she secretly dove in and quickly started cutting my cuticles thinking I would not notice. Why is cuticle-cutting a necessary part of nail care ... and almost a rampant ritual?
The cuticle is the protective barrier surrounding the nail plate and nail folds. Biting, pulling, or improper cutting of the cuticle over time can cause long-term damage to the nail plate, such as ridging of the nail, median nail dystrophy, or permanent destruction of the nail plate. Trimming the cuticles can also break the seal that protects the surrounding skin and nails. Not only can the removal of the cuticle introduce infection, but it can also cause deformities in the nail plate itself. Infections to consider around the nail include acute or chronic paronychia, herpetic whitlow, onychomycosis, and warts. These infections can be the direct result of entry from the removal of the cuticle barrier or improperly cleaned and sterilized instruments.
Tools used to remove cuticles can transfer infections. In addition to skin infections, viruses that cause systemic infections, such as hepatitis C, can live in dry blood for up to 3 days and can be transferred on tools that have not been cleaned properly. Sterilized tools must first be cleaned and submerged in antiseptic solutions, then sterilized in an autoclave or a Food and Drug Administration–registered dry-heat sterilizer, not a UV box. UV boxes are commonly used and do not actually sterilize tools; they keep tools clean only if they have been previously sterilized.
The best way to ensure proper sterilization is to check the indicator tape or indicator color on the packaging. Autoclave tape and dry heat sterilizer strips work by changing colors when exposed to a certain temperature (and pressure for the autoclave tape) for a certain amount of time. I routinely check the sterilizing packets and immediately look up the indicator color on the Internet to ensure the color change was correct. I ask about what sterilization techniques the salon uses, and I often require salons to use my own nail care tools (which should be cleaned after every use).
Trimming or cutting cuticles is a bad habit and can be a dangerous salon ritual. Many states, such as New York and Massachusetts, do not allow manicurists to cut the cuticles given blood-borne pathogen risks and improper sanitation; however, this regulation is often loosely enforced. It also creates an endless cycle of cuticle trimming as the growing cuticle can often look frayed – and thus creates the need for them to be cut over and over again. Pushing the cuticle back may be a better option for those who prefer the cosmetic appearance of trimmed cuticles, but it still poses a portal of entry for pathogens.
Let’s educate our patients, the salons, and the regulatory boards to prevent the spread of infection and ensure safe nail care techniques.
Dr. Wesley and Dr. Talakoub are co-contributors to a monthly Aesthetic Dermatology column in Dermatology News. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub.
I recently went to a local salon for a manicure, and when I asked the manicurist not to cut my cuticles, she looked at me as though I was offending her. Shortly thereafter, I took a phone call that swayed my attention, and she secretly dove in and quickly started cutting my cuticles thinking I would not notice. Why is cuticle-cutting a necessary part of nail care ... and almost a rampant ritual?
The cuticle is the protective barrier surrounding the nail plate and nail folds. Biting, pulling, or improper cutting of the cuticle over time can cause long-term damage to the nail plate, such as ridging of the nail, median nail dystrophy, or permanent destruction of the nail plate. Trimming the cuticles can also break the seal that protects the surrounding skin and nails. Not only can the removal of the cuticle introduce infection, but it can also cause deformities in the nail plate itself. Infections to consider around the nail include acute or chronic paronychia, herpetic whitlow, onychomycosis, and warts. These infections can be the direct result of entry from the removal of the cuticle barrier or improperly cleaned and sterilized instruments.
Tools used to remove cuticles can transfer infections. In addition to skin infections, viruses that cause systemic infections, such as hepatitis C, can live in dry blood for up to 3 days and can be transferred on tools that have not been cleaned properly. Sterilized tools must first be cleaned and submerged in antiseptic solutions, then sterilized in an autoclave or a Food and Drug Administration–registered dry-heat sterilizer, not a UV box. UV boxes are commonly used and do not actually sterilize tools; they keep tools clean only if they have been previously sterilized.
The best way to ensure proper sterilization is to check the indicator tape or indicator color on the packaging. Autoclave tape and dry heat sterilizer strips work by changing colors when exposed to a certain temperature (and pressure for the autoclave tape) for a certain amount of time. I routinely check the sterilizing packets and immediately look up the indicator color on the Internet to ensure the color change was correct. I ask about what sterilization techniques the salon uses, and I often require salons to use my own nail care tools (which should be cleaned after every use).
Trimming or cutting cuticles is a bad habit and can be a dangerous salon ritual. Many states, such as New York and Massachusetts, do not allow manicurists to cut the cuticles given blood-borne pathogen risks and improper sanitation; however, this regulation is often loosely enforced. It also creates an endless cycle of cuticle trimming as the growing cuticle can often look frayed – and thus creates the need for them to be cut over and over again. Pushing the cuticle back may be a better option for those who prefer the cosmetic appearance of trimmed cuticles, but it still poses a portal of entry for pathogens.
Let’s educate our patients, the salons, and the regulatory boards to prevent the spread of infection and ensure safe nail care techniques.
Dr. Wesley and Dr. Talakoub are co-contributors to a monthly Aesthetic Dermatology column in Dermatology News. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub.
I recently went to a local salon for a manicure, and when I asked the manicurist not to cut my cuticles, she looked at me as though I was offending her. Shortly thereafter, I took a phone call that swayed my attention, and she secretly dove in and quickly started cutting my cuticles thinking I would not notice. Why is cuticle-cutting a necessary part of nail care ... and almost a rampant ritual?
The cuticle is the protective barrier surrounding the nail plate and nail folds. Biting, pulling, or improper cutting of the cuticle over time can cause long-term damage to the nail plate, such as ridging of the nail, median nail dystrophy, or permanent destruction of the nail plate. Trimming the cuticles can also break the seal that protects the surrounding skin and nails. Not only can the removal of the cuticle introduce infection, but it can also cause deformities in the nail plate itself. Infections to consider around the nail include acute or chronic paronychia, herpetic whitlow, onychomycosis, and warts. These infections can be the direct result of entry from the removal of the cuticle barrier or improperly cleaned and sterilized instruments.
Tools used to remove cuticles can transfer infections. In addition to skin infections, viruses that cause systemic infections, such as hepatitis C, can live in dry blood for up to 3 days and can be transferred on tools that have not been cleaned properly. Sterilized tools must first be cleaned and submerged in antiseptic solutions, then sterilized in an autoclave or a Food and Drug Administration–registered dry-heat sterilizer, not a UV box. UV boxes are commonly used and do not actually sterilize tools; they keep tools clean only if they have been previously sterilized.
The best way to ensure proper sterilization is to check the indicator tape or indicator color on the packaging. Autoclave tape and dry heat sterilizer strips work by changing colors when exposed to a certain temperature (and pressure for the autoclave tape) for a certain amount of time. I routinely check the sterilizing packets and immediately look up the indicator color on the Internet to ensure the color change was correct. I ask about what sterilization techniques the salon uses, and I often require salons to use my own nail care tools (which should be cleaned after every use).
Trimming or cutting cuticles is a bad habit and can be a dangerous salon ritual. Many states, such as New York and Massachusetts, do not allow manicurists to cut the cuticles given blood-borne pathogen risks and improper sanitation; however, this regulation is often loosely enforced. It also creates an endless cycle of cuticle trimming as the growing cuticle can often look frayed – and thus creates the need for them to be cut over and over again. Pushing the cuticle back may be a better option for those who prefer the cosmetic appearance of trimmed cuticles, but it still poses a portal of entry for pathogens.
Let’s educate our patients, the salons, and the regulatory boards to prevent the spread of infection and ensure safe nail care techniques.
Dr. Wesley and Dr. Talakoub are co-contributors to a monthly Aesthetic Dermatology column in Dermatology News. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub.
Noninvasive Cosmetic Procedures: Delegation Among Dermatologists
Noninvasive cosmetic surgery procedures continue to increase in number, according to the American Society of Plastic Surgeons. Furthermore, these procedures are being performed in the offices of a growing number of physician specialties. As demand increases and offices become busy, there is increased delegation of these procedures to nurses, physician assistants, aestheticians, and medical assistants.
Austin et al (Dermatol Surg. 2015;41:827-832.) performed an Internet-based survey of physician members of the American Society for Dermatologic Surgery, the American Society for Laser Medicine and Surgery, and the American Society for Aesthetic Plastic Surgery to evaluate the delegation practices among the various specialties. The survey asked for physician training, age, gender, residency, and geographic location, as well as a breakdown of how much of their practice involved cosmetic procedures. Respondents were asked if they delegate procedures, to whom do they delegate (eg, registered nurse, physician assistant, MAs), and which procedures they delegate. A point system was used to give a score based on the perceived level of education and/or training of the person delegated to perform a procedure: do not delegate (1); physician assistant or nurse practitioner (2); registered nurse (3); and MA, aesthetician, or other (4).
Respondents included 823 physicians: 521 dermatologists and 302 nondermatologists. Of the respondents, 291 of dermatologists (55.9%) and 223 of nondermatologist physicians (73.8%) delegated cosmetic procedures. Procedures most often delegated by dermatologists compared to nondermatologists included chemical peels, neuromodulator and filler injections, laser hair removal, pulsed dye laser, tattoo removal, intense pulsed light, nonablative factional laser, and sclerotherapy.
The outcome of this survey shows that delegating certain cosmetic procedures is common, with more than half of all survey respondents delegating at least 1 cosmetic procedure. Dermatologists, as a whole, delegated less frequently than nondermatologist physicians, and when they did, dermatologists delegated to higher-level providers compared to nondermatologists. Medical practices in which the focus is primarily cosmetic and in those in which the physician is older also were more likely to delegate procedures.
What’s the issue?
There is tremendous variability between states about who is competent to perform many of the noninvasive cosmetic procedures. Some state medical boards specify the type of provider and the procedures allowed, while other states have no policy. However, this rule is changing, and there are more regulations being brought before the state medical boards with the goal of ensuring patient safety. As the demand for noninvasive procedures continues to grow, there will be an increase in the number of nonphysician providers performing these procedures. Patient safety should be at the forefront of all legislation, but at the same time there should be training courses and oversight boards to allow for safe delegation. Do you see yourself delegating more of these noninvasive procedures in the future?
Noninvasive cosmetic surgery procedures continue to increase in number, according to the American Society of Plastic Surgeons. Furthermore, these procedures are being performed in the offices of a growing number of physician specialties. As demand increases and offices become busy, there is increased delegation of these procedures to nurses, physician assistants, aestheticians, and medical assistants.
Austin et al (Dermatol Surg. 2015;41:827-832.) performed an Internet-based survey of physician members of the American Society for Dermatologic Surgery, the American Society for Laser Medicine and Surgery, and the American Society for Aesthetic Plastic Surgery to evaluate the delegation practices among the various specialties. The survey asked for physician training, age, gender, residency, and geographic location, as well as a breakdown of how much of their practice involved cosmetic procedures. Respondents were asked if they delegate procedures, to whom do they delegate (eg, registered nurse, physician assistant, MAs), and which procedures they delegate. A point system was used to give a score based on the perceived level of education and/or training of the person delegated to perform a procedure: do not delegate (1); physician assistant or nurse practitioner (2); registered nurse (3); and MA, aesthetician, or other (4).
Respondents included 823 physicians: 521 dermatologists and 302 nondermatologists. Of the respondents, 291 of dermatologists (55.9%) and 223 of nondermatologist physicians (73.8%) delegated cosmetic procedures. Procedures most often delegated by dermatologists compared to nondermatologists included chemical peels, neuromodulator and filler injections, laser hair removal, pulsed dye laser, tattoo removal, intense pulsed light, nonablative factional laser, and sclerotherapy.
The outcome of this survey shows that delegating certain cosmetic procedures is common, with more than half of all survey respondents delegating at least 1 cosmetic procedure. Dermatologists, as a whole, delegated less frequently than nondermatologist physicians, and when they did, dermatologists delegated to higher-level providers compared to nondermatologists. Medical practices in which the focus is primarily cosmetic and in those in which the physician is older also were more likely to delegate procedures.
What’s the issue?
There is tremendous variability between states about who is competent to perform many of the noninvasive cosmetic procedures. Some state medical boards specify the type of provider and the procedures allowed, while other states have no policy. However, this rule is changing, and there are more regulations being brought before the state medical boards with the goal of ensuring patient safety. As the demand for noninvasive procedures continues to grow, there will be an increase in the number of nonphysician providers performing these procedures. Patient safety should be at the forefront of all legislation, but at the same time there should be training courses and oversight boards to allow for safe delegation. Do you see yourself delegating more of these noninvasive procedures in the future?
Noninvasive cosmetic surgery procedures continue to increase in number, according to the American Society of Plastic Surgeons. Furthermore, these procedures are being performed in the offices of a growing number of physician specialties. As demand increases and offices become busy, there is increased delegation of these procedures to nurses, physician assistants, aestheticians, and medical assistants.
Austin et al (Dermatol Surg. 2015;41:827-832.) performed an Internet-based survey of physician members of the American Society for Dermatologic Surgery, the American Society for Laser Medicine and Surgery, and the American Society for Aesthetic Plastic Surgery to evaluate the delegation practices among the various specialties. The survey asked for physician training, age, gender, residency, and geographic location, as well as a breakdown of how much of their practice involved cosmetic procedures. Respondents were asked if they delegate procedures, to whom do they delegate (eg, registered nurse, physician assistant, MAs), and which procedures they delegate. A point system was used to give a score based on the perceived level of education and/or training of the person delegated to perform a procedure: do not delegate (1); physician assistant or nurse practitioner (2); registered nurse (3); and MA, aesthetician, or other (4).
Respondents included 823 physicians: 521 dermatologists and 302 nondermatologists. Of the respondents, 291 of dermatologists (55.9%) and 223 of nondermatologist physicians (73.8%) delegated cosmetic procedures. Procedures most often delegated by dermatologists compared to nondermatologists included chemical peels, neuromodulator and filler injections, laser hair removal, pulsed dye laser, tattoo removal, intense pulsed light, nonablative factional laser, and sclerotherapy.
The outcome of this survey shows that delegating certain cosmetic procedures is common, with more than half of all survey respondents delegating at least 1 cosmetic procedure. Dermatologists, as a whole, delegated less frequently than nondermatologist physicians, and when they did, dermatologists delegated to higher-level providers compared to nondermatologists. Medical practices in which the focus is primarily cosmetic and in those in which the physician is older also were more likely to delegate procedures.
What’s the issue?
There is tremendous variability between states about who is competent to perform many of the noninvasive cosmetic procedures. Some state medical boards specify the type of provider and the procedures allowed, while other states have no policy. However, this rule is changing, and there are more regulations being brought before the state medical boards with the goal of ensuring patient safety. As the demand for noninvasive procedures continues to grow, there will be an increase in the number of nonphysician providers performing these procedures. Patient safety should be at the forefront of all legislation, but at the same time there should be training courses and oversight boards to allow for safe delegation. Do you see yourself delegating more of these noninvasive procedures in the future?
Vegetative Sacral Plaque in a Patient With Human Immunodeficiency Virus
The Diagnosis: Herpes Simplex Vegetans
Histopathologic examination using hematoxylin and eosin stain demonstrated marked pseudoepitheliomatous hyperplasia with granulation tissue, ulceration, and abundant exudate joined by a dense mixed inflammatory cell infiltrate that included a myriad of eosinophils (Figure, A). At higher power (Figure, B), many single and multinucleate acantholytic keratinocytes showed ground-glass nuclei and peripheral margination of chromatin within zones of ulceration and crust. Viral culture and direct fluorescent antibody assay identified herpes simplex virus (HSV) type 2. Based on the clinical and histopathologic findings, the patient was diagnosed with herpes simplex vegetans. He was initially treated with oral acyclovir and then oral famciclovir but showed minimal improvement. Eventually, he was referred to surgery and the mass was totally excised with clear margins and no evidence of underlying malignancy.

|
| Histopathology revealed marked pseudoepitheliomatous hyperplasia, ulceration, and a dense mixed inflammatory cell infiltrate (A)(H&E, original magnification ×20). Many multinucleate acantholytic keratinocytes with ground-glass nuclei and peripheral margination of chromatin were shown (B)(H&E, original magnification ×400). |
Herpes simplex virus is one of the most common sexually transmitted infections, with a notably increased incidence and prevalence among individuals with human immunodeficiency virus (HIV) infection.1 Although typical HSV manifestation in immunocompetent patients includes clustered vesicles and/or ulcerations, immunocompromised patients may have unusual presentations, such as persistent and extensive ulcerations or nodular hyperkeratotic lesions.2,3 Herpes vegetans, a term used to describe these atypical exophytic lesions, rarely has been reported in literature, but its presence should raise suspicion for possible underlying immunocompromise. The pathogenesis behind the hypertrophic nature of these lesions is not well understood, but it is postulated that the immune dysregulation from concomitant HIV and HSV infection plays a role.2 Overproduction of tumor necrosis factor and IL-6 by HIV-infected dermal dendritic cells causes an increase in antiapoptotic factors within the epidermis, resulting in enhanced keratinocyte proliferation and clinical hyperkeratosis.2,4
The differential diagnosis for herpes vegetans is somewhat broad, owing to the verrucous and often eroded appearance of the lesions. Biopsy and cultures can be obtained to differentiate from condyloma acuminatum, condyloma latum (secondary syphilis), pyoderma vegetans, pemphigus vegetans, granuloma inguinale, extraintestinal Crohn disease, deep fungal infections, cutaneous tuberculosis, and malignancy.2-4 Histopathology shows epithelial hyperplasia and ulceration with scattered multinucleate keratinocytes, usually at the periphery of the ulcer, and intranuclear inclusions typical of HSV. In addition, a dense dermal infiltrate of lymphocytes, histiocytes, plasma cells, and eosinophils is usually present beneath the base of the ulcer.2,4
Treatment options for herpes vegetans are limited due to the high prevalence of acyclovir-resistant (ACV-R) HSV-2 strains in HIV patients. Valacyclovir and penciclovir have been largely ineffective against ACV-R HSV due to their dependence on the same enzyme—thymidine kinase—involved in the mechanism of acyclovir resistance. Intravenous foscarnet and cidofovir have shown efficacy against ACV-R virus, but concerns of nephrotoxicity have limited their use over prolonged intervals.5 Castelo-Soccio et al6 reported promising results with intralesional cidofovir. This route of administration provides the advantage of increased bioavailability with reduced risk for nephrotoxicity.6 Finally, surgical resection may be considered for refractory lesions to circumvent the toxicity from systemically administered drugs.3
- Severson JL, Tyring SK. Relation between herpes simplex viruses and human immunodeficiency virus infections. Arch Dermatol. 1999;135:1393-1397.
- Patel AB, Rosen T. Herpes vegetans as a sign of HIV infection. Dermatol Online J. 2008;14:6.
- Chung VQ, Parker DC, Parker SR. Surgical excision for vegetative herpes simplex virus infection. Dermatol Surg. 2007;33:1374-1379.
- Beasley KL, Cooley GE, Kao GF, et al. Herpes simplex vegetans: atypical genital herpes infection in a patient with common variable immunodeficiency. J Am Acad Dermatol. 1997;37(5, pt 2):860-863.
- Chilukuri S, Rosen T. Management of acyclovir-resistant herpes simplex virus. Dermatol Clin. 2003;21:311-320.
- Castelo-Soccio L, Bernardin R, Stern J, et al. Successful treatment of acyclovir-resistant herpes simplex virus with intralesional cidofovir. Arch Dermatol. 2010;146:124-126.
The Diagnosis: Herpes Simplex Vegetans
Histopathologic examination using hematoxylin and eosin stain demonstrated marked pseudoepitheliomatous hyperplasia with granulation tissue, ulceration, and abundant exudate joined by a dense mixed inflammatory cell infiltrate that included a myriad of eosinophils (Figure, A). At higher power (Figure, B), many single and multinucleate acantholytic keratinocytes showed ground-glass nuclei and peripheral margination of chromatin within zones of ulceration and crust. Viral culture and direct fluorescent antibody assay identified herpes simplex virus (HSV) type 2. Based on the clinical and histopathologic findings, the patient was diagnosed with herpes simplex vegetans. He was initially treated with oral acyclovir and then oral famciclovir but showed minimal improvement. Eventually, he was referred to surgery and the mass was totally excised with clear margins and no evidence of underlying malignancy.


|
| Histopathology revealed marked pseudoepitheliomatous hyperplasia, ulceration, and a dense mixed inflammatory cell infiltrate (A)(H&E, original magnification ×20). Many multinucleate acantholytic keratinocytes with ground-glass nuclei and peripheral margination of chromatin were shown (B)(H&E, original magnification ×400). |
Herpes simplex virus is one of the most common sexually transmitted infections, with a notably increased incidence and prevalence among individuals with human immunodeficiency virus (HIV) infection.1 Although typical HSV manifestation in immunocompetent patients includes clustered vesicles and/or ulcerations, immunocompromised patients may have unusual presentations, such as persistent and extensive ulcerations or nodular hyperkeratotic lesions.2,3 Herpes vegetans, a term used to describe these atypical exophytic lesions, rarely has been reported in literature, but its presence should raise suspicion for possible underlying immunocompromise. The pathogenesis behind the hypertrophic nature of these lesions is not well understood, but it is postulated that the immune dysregulation from concomitant HIV and HSV infection plays a role.2 Overproduction of tumor necrosis factor and IL-6 by HIV-infected dermal dendritic cells causes an increase in antiapoptotic factors within the epidermis, resulting in enhanced keratinocyte proliferation and clinical hyperkeratosis.2,4
The differential diagnosis for herpes vegetans is somewhat broad, owing to the verrucous and often eroded appearance of the lesions. Biopsy and cultures can be obtained to differentiate from condyloma acuminatum, condyloma latum (secondary syphilis), pyoderma vegetans, pemphigus vegetans, granuloma inguinale, extraintestinal Crohn disease, deep fungal infections, cutaneous tuberculosis, and malignancy.2-4 Histopathology shows epithelial hyperplasia and ulceration with scattered multinucleate keratinocytes, usually at the periphery of the ulcer, and intranuclear inclusions typical of HSV. In addition, a dense dermal infiltrate of lymphocytes, histiocytes, plasma cells, and eosinophils is usually present beneath the base of the ulcer.2,4
Treatment options for herpes vegetans are limited due to the high prevalence of acyclovir-resistant (ACV-R) HSV-2 strains in HIV patients. Valacyclovir and penciclovir have been largely ineffective against ACV-R HSV due to their dependence on the same enzyme—thymidine kinase—involved in the mechanism of acyclovir resistance. Intravenous foscarnet and cidofovir have shown efficacy against ACV-R virus, but concerns of nephrotoxicity have limited their use over prolonged intervals.5 Castelo-Soccio et al6 reported promising results with intralesional cidofovir. This route of administration provides the advantage of increased bioavailability with reduced risk for nephrotoxicity.6 Finally, surgical resection may be considered for refractory lesions to circumvent the toxicity from systemically administered drugs.3
The Diagnosis: Herpes Simplex Vegetans
Histopathologic examination using hematoxylin and eosin stain demonstrated marked pseudoepitheliomatous hyperplasia with granulation tissue, ulceration, and abundant exudate joined by a dense mixed inflammatory cell infiltrate that included a myriad of eosinophils (Figure, A). At higher power (Figure, B), many single and multinucleate acantholytic keratinocytes showed ground-glass nuclei and peripheral margination of chromatin within zones of ulceration and crust. Viral culture and direct fluorescent antibody assay identified herpes simplex virus (HSV) type 2. Based on the clinical and histopathologic findings, the patient was diagnosed with herpes simplex vegetans. He was initially treated with oral acyclovir and then oral famciclovir but showed minimal improvement. Eventually, he was referred to surgery and the mass was totally excised with clear margins and no evidence of underlying malignancy.


|
| Histopathology revealed marked pseudoepitheliomatous hyperplasia, ulceration, and a dense mixed inflammatory cell infiltrate (A)(H&E, original magnification ×20). Many multinucleate acantholytic keratinocytes with ground-glass nuclei and peripheral margination of chromatin were shown (B)(H&E, original magnification ×400). |
Herpes simplex virus is one of the most common sexually transmitted infections, with a notably increased incidence and prevalence among individuals with human immunodeficiency virus (HIV) infection.1 Although typical HSV manifestation in immunocompetent patients includes clustered vesicles and/or ulcerations, immunocompromised patients may have unusual presentations, such as persistent and extensive ulcerations or nodular hyperkeratotic lesions.2,3 Herpes vegetans, a term used to describe these atypical exophytic lesions, rarely has been reported in literature, but its presence should raise suspicion for possible underlying immunocompromise. The pathogenesis behind the hypertrophic nature of these lesions is not well understood, but it is postulated that the immune dysregulation from concomitant HIV and HSV infection plays a role.2 Overproduction of tumor necrosis factor and IL-6 by HIV-infected dermal dendritic cells causes an increase in antiapoptotic factors within the epidermis, resulting in enhanced keratinocyte proliferation and clinical hyperkeratosis.2,4
The differential diagnosis for herpes vegetans is somewhat broad, owing to the verrucous and often eroded appearance of the lesions. Biopsy and cultures can be obtained to differentiate from condyloma acuminatum, condyloma latum (secondary syphilis), pyoderma vegetans, pemphigus vegetans, granuloma inguinale, extraintestinal Crohn disease, deep fungal infections, cutaneous tuberculosis, and malignancy.2-4 Histopathology shows epithelial hyperplasia and ulceration with scattered multinucleate keratinocytes, usually at the periphery of the ulcer, and intranuclear inclusions typical of HSV. In addition, a dense dermal infiltrate of lymphocytes, histiocytes, plasma cells, and eosinophils is usually present beneath the base of the ulcer.2,4
Treatment options for herpes vegetans are limited due to the high prevalence of acyclovir-resistant (ACV-R) HSV-2 strains in HIV patients. Valacyclovir and penciclovir have been largely ineffective against ACV-R HSV due to their dependence on the same enzyme—thymidine kinase—involved in the mechanism of acyclovir resistance. Intravenous foscarnet and cidofovir have shown efficacy against ACV-R virus, but concerns of nephrotoxicity have limited their use over prolonged intervals.5 Castelo-Soccio et al6 reported promising results with intralesional cidofovir. This route of administration provides the advantage of increased bioavailability with reduced risk for nephrotoxicity.6 Finally, surgical resection may be considered for refractory lesions to circumvent the toxicity from systemically administered drugs.3
- Severson JL, Tyring SK. Relation between herpes simplex viruses and human immunodeficiency virus infections. Arch Dermatol. 1999;135:1393-1397.
- Patel AB, Rosen T. Herpes vegetans as a sign of HIV infection. Dermatol Online J. 2008;14:6.
- Chung VQ, Parker DC, Parker SR. Surgical excision for vegetative herpes simplex virus infection. Dermatol Surg. 2007;33:1374-1379.
- Beasley KL, Cooley GE, Kao GF, et al. Herpes simplex vegetans: atypical genital herpes infection in a patient with common variable immunodeficiency. J Am Acad Dermatol. 1997;37(5, pt 2):860-863.
- Chilukuri S, Rosen T. Management of acyclovir-resistant herpes simplex virus. Dermatol Clin. 2003;21:311-320.
- Castelo-Soccio L, Bernardin R, Stern J, et al. Successful treatment of acyclovir-resistant herpes simplex virus with intralesional cidofovir. Arch Dermatol. 2010;146:124-126.
- Severson JL, Tyring SK. Relation between herpes simplex viruses and human immunodeficiency virus infections. Arch Dermatol. 1999;135:1393-1397.
- Patel AB, Rosen T. Herpes vegetans as a sign of HIV infection. Dermatol Online J. 2008;14:6.
- Chung VQ, Parker DC, Parker SR. Surgical excision for vegetative herpes simplex virus infection. Dermatol Surg. 2007;33:1374-1379.
- Beasley KL, Cooley GE, Kao GF, et al. Herpes simplex vegetans: atypical genital herpes infection in a patient with common variable immunodeficiency. J Am Acad Dermatol. 1997;37(5, pt 2):860-863.
- Chilukuri S, Rosen T. Management of acyclovir-resistant herpes simplex virus. Dermatol Clin. 2003;21:311-320.
- Castelo-Soccio L, Bernardin R, Stern J, et al. Successful treatment of acyclovir-resistant herpes simplex virus with intralesional cidofovir. Arch Dermatol. 2010;146:124-126.
A 53-year-old man presented to our clinic with a sacral mass that had progressively enlarged over 2 years. The patient reported occasional oozing from the mass as well as pain when laying flat but denied fever or other symptoms. His medical history was remarkable for human immunodeficiency virus infection with variable adherence to a highly active antiretroviral therapy regimen. At the time of presentation, the patient had a CD4 lymphocyte count of 78 cells/mm3 (reference range, 500–1400 cells/mm3) and a viral load of 290 copies/mL (reference range, 0 copies/mL). Physical examination revealed a 10-cm discrete, moist and pink, exophytic plaque on the sacrum with superficial erosions. The plaque was nontender and without associated lymphadenopathy. The skin and mucous membranes were otherwise clear. A cutaneous biopsy specimen was obtained from the tumor and sent for histopathologic analysis.