User login
Neck Rejuvenation [editorial]
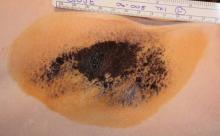
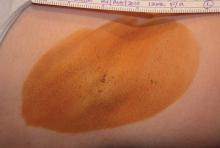
How We Do It: Biopsy of a Suspicious Pigmented Lesion and How to Reduce Scarring and Improve Cosmetic Outcome
FDA Panel Backs Approval of Restylane for Lip Augmentation
Dermatologists may not have to use Restylane off-label for lip augmentation any longer. A Food and Drug Administration panel voted 6-0 with 1 abstention that benefits outweigh risks for using the filler as a submucosal injection for lip augmentation on April 27.
The panel also voted 6-0 with 1 abstention that the filler is safe and effective for the expanded indication.
Restylane (Medicis Aesthetics) is a hyaluronic acid gel generated by Streptococcus bacteria, chemically crosslinked with 1,4 butanediol diglycidyl ether. The filler was first approved in 2005 for mid-to-deep dermal implantation for the correction of moderate-to-severe facial wrinkles and folds, such as nasolabial folds.
Medicis recently conducted a clinical study (MA-1399-15) to evaluate the safety and effectiveness of the filler in the augmentation of soft tissue fullness of the lips. The study included 135 patients, who received lip augmentation with Restylane, and 45 patients with no treatment. The mean volume of filler was 2.9 cc per patient, with a range of 0.6 -5.6 cc per patient. At 8 weeks, 92% of patients who received Restylane were considered responders.
Adverse events occurred in 99% of patients. Expected treatment-emergent adverse events included bruising, redness, swelling, pain, tenderness, itching, and skin exfoliation. Of note, herpes simplex virus 1 outbreaks occurred in 4% of patients. The outbreaks were determined to be associated with injection of the filler in 7 or 10 cases.
Forty percent of patients who received the filler had adverse outcomes that they felt affected their daily activity or were disabling; 15% of Restylane patients experienced adverse events (typically swelling and tenderness) that lasted more than 15 days.
The panel was particularly concerned about the implications of the treatment in younger patients who may still be growing and are likely to receive repeat treatments over time.
"My only problem with voting completely yes … is that I don't think that the question was worded in such a way that I felt totally comfortable giving my final approval for safety, efficacy, and risk/benefit ratio to all populations," said panel member Dr. Delora L. Mount, who is an associate professor of surgery and pediatrics at the University of Wisconsin in Madison.
The FDA panel was also concerned about the lack of men and individuals with dark skin in the trial. Only one man was included and 38 individuals were Fitzpatrick skin type IV, 3 individuals were type V, and none were type VI.
The FDA usually follows its panels' advice, but is not obligated to do so.
Dermatologists may not have to use Restylane off-label for lip augmentation any longer. A Food and Drug Administration panel voted 6-0 with 1 abstention that benefits outweigh risks for using the filler as a submucosal injection for lip augmentation on April 27.
The panel also voted 6-0 with 1 abstention that the filler is safe and effective for the expanded indication.
Restylane (Medicis Aesthetics) is a hyaluronic acid gel generated by Streptococcus bacteria, chemically crosslinked with 1,4 butanediol diglycidyl ether. The filler was first approved in 2005 for mid-to-deep dermal implantation for the correction of moderate-to-severe facial wrinkles and folds, such as nasolabial folds.
Medicis recently conducted a clinical study (MA-1399-15) to evaluate the safety and effectiveness of the filler in the augmentation of soft tissue fullness of the lips. The study included 135 patients, who received lip augmentation with Restylane, and 45 patients with no treatment. The mean volume of filler was 2.9 cc per patient, with a range of 0.6 -5.6 cc per patient. At 8 weeks, 92% of patients who received Restylane were considered responders.
Adverse events occurred in 99% of patients. Expected treatment-emergent adverse events included bruising, redness, swelling, pain, tenderness, itching, and skin exfoliation. Of note, herpes simplex virus 1 outbreaks occurred in 4% of patients. The outbreaks were determined to be associated with injection of the filler in 7 or 10 cases.
Forty percent of patients who received the filler had adverse outcomes that they felt affected their daily activity or were disabling; 15% of Restylane patients experienced adverse events (typically swelling and tenderness) that lasted more than 15 days.
The panel was particularly concerned about the implications of the treatment in younger patients who may still be growing and are likely to receive repeat treatments over time.
"My only problem with voting completely yes … is that I don't think that the question was worded in such a way that I felt totally comfortable giving my final approval for safety, efficacy, and risk/benefit ratio to all populations," said panel member Dr. Delora L. Mount, who is an associate professor of surgery and pediatrics at the University of Wisconsin in Madison.
The FDA panel was also concerned about the lack of men and individuals with dark skin in the trial. Only one man was included and 38 individuals were Fitzpatrick skin type IV, 3 individuals were type V, and none were type VI.
The FDA usually follows its panels' advice, but is not obligated to do so.
Dermatologists may not have to use Restylane off-label for lip augmentation any longer. A Food and Drug Administration panel voted 6-0 with 1 abstention that benefits outweigh risks for using the filler as a submucosal injection for lip augmentation on April 27.
The panel also voted 6-0 with 1 abstention that the filler is safe and effective for the expanded indication.
Restylane (Medicis Aesthetics) is a hyaluronic acid gel generated by Streptococcus bacteria, chemically crosslinked with 1,4 butanediol diglycidyl ether. The filler was first approved in 2005 for mid-to-deep dermal implantation for the correction of moderate-to-severe facial wrinkles and folds, such as nasolabial folds.
Medicis recently conducted a clinical study (MA-1399-15) to evaluate the safety and effectiveness of the filler in the augmentation of soft tissue fullness of the lips. The study included 135 patients, who received lip augmentation with Restylane, and 45 patients with no treatment. The mean volume of filler was 2.9 cc per patient, with a range of 0.6 -5.6 cc per patient. At 8 weeks, 92% of patients who received Restylane were considered responders.
Adverse events occurred in 99% of patients. Expected treatment-emergent adverse events included bruising, redness, swelling, pain, tenderness, itching, and skin exfoliation. Of note, herpes simplex virus 1 outbreaks occurred in 4% of patients. The outbreaks were determined to be associated with injection of the filler in 7 or 10 cases.
Forty percent of patients who received the filler had adverse outcomes that they felt affected their daily activity or were disabling; 15% of Restylane patients experienced adverse events (typically swelling and tenderness) that lasted more than 15 days.
The panel was particularly concerned about the implications of the treatment in younger patients who may still be growing and are likely to receive repeat treatments over time.
"My only problem with voting completely yes … is that I don't think that the question was worded in such a way that I felt totally comfortable giving my final approval for safety, efficacy, and risk/benefit ratio to all populations," said panel member Dr. Delora L. Mount, who is an associate professor of surgery and pediatrics at the University of Wisconsin in Madison.
The FDA panel was also concerned about the lack of men and individuals with dark skin in the trial. Only one man was included and 38 individuals were Fitzpatrick skin type IV, 3 individuals were type V, and none were type VI.
The FDA usually follows its panels' advice, but is not obligated to do so.
Microwave Device Offers Long-Term Treatment for Hyperhidrosis
GRAPEVINE, TEX. – A novel microwave device significantly reduced underarm sweating in two studies involving a total of 151 patients with axillary hyperhidrosis.
The condition affects millions of people, yet current treatments are limited by either duration of effect or efficacy. The microwave device has the potential for a longer-term and possibly even permanent effect via eradication of eccrine sweat glands, Dr. Suzanne L. Kilmer and Dr. Mark Lupin reported in separate presentations at the annual meeting of the American Society for Laser Medicine and Surgery.
Miramar Labs' miraDry system focuses microwave energy to selectively heat the interface between the skin and underlying fat, where the sweat glands reside. The system comprises a console, handpiece, and disposable tip. The in-office procedure takes approximately 40 minutes. The system was cleared for licensure in the United States by the Food and Drug Administration in January 2011, and will be marketed later this year, according to a company spokesperson.
Dr. Kilmer, of the Laser and Skin Surgery Center of Northern California, Sacramento, presented data on a second-generation version of the device that was FDA approved but never marketed. In a multicenter, sham-controlled study, 120 adults with hyperhidrosis were randomized to miraDry (81) or sham treatment (39) for one to three sessions. The patients had to have a Hyperhidrosis Disease Severity Scale (HDSS) score of 3 or 4 ("my underarm sweating is barely tolerable and frequently/always interferes with my daily activities") at baseline, and gravimetric readings greater than 50 mg at each axilla (in 5 minutes).
Follow-up was 6 months for the sham group and 12 months for the treatment group. Responders were defined as subjects reporting a reduction to an HDSS score of 1 ("My underarm sweating is never noticeable and never interferes with daily activities") or 2 ("My underarm sweating is tolerable but sometimes interferes with daily activities"). The patients had a mean age of 33 years, 58% were female, and 84% were white.
Efficacy for the treatment group was 89% at 30 days, 74% at 3 months, 67% at 6 months, and 69% at the 9- and 12-month visits. In the sham group, efficacy was 54% at 30 days and 44% for the 3- and 6-month visits. At all time points, the differences between the treatment and sham groups were significant, Dr. Kilmer reported.
Treatment-related adverse events were generally mild, and all but one resolved. The most common adverse events in the treatment group were transient patches of altered sensation in the treatment limb, occurring in 8 patients (10%), and axillary pain requiring prescription medication in 5 patients (6%). Most subjects experienced transient post-treatment local sequelae in the axilla such as edema, tenderness, and bruising.
Follow-up of the treated subjects showed stable efficacy through 12 months, she said.
The second study, involving 31 patients, investigated the third-generation, optimized version of the device. As in the earlier study, all patients had to have primary axillary hyperhidrosis with an HDSS score of 3 or 4. The patients also were required to have a gravimetric sweat assessment of at least 50 mg in each axilla (in 5 minutes). None of the patients had surgery for axillary hyperhidrosis or botulinum toxin injections in the axillae in the prior 12 months, said Dr. Lupin of Cosmedica Laser Centre, Victoria, B.C.
The patients had a mean age of 33 years (range 18-65 years), and three-quarters were female. They had a mean BMI of 24.8 kg/m2. Of 28 patients seen at 30 days, all but one (96%) had HDSS scores of 1 or 2. Of 25 seen at 3 months, all had HDSS scores of 1 or 2 (100% efficacy). Gravimetric assessments 3 months after treatment showed that 94% of patients had at least a 50% reduction in axillary sweat compared with baseline, with an average sweat reduction of 82%.
Nineteen of the 31 initially enrolled patients (61%) experienced at least one treatment-related adverse event, of which most (88%) were rated as mild. The most common were numbness or tingling in an area of the treated limb (12 patients, 39%), and edema in the chest or treatment limb lasting about a week (9 patients, 29%). Most of the patients also experienced acute post-treatment transient effects in the treatment area such as localized edema, tenderness, or erythema. Follow-up of safety is ongoing, Dr. Lupin noted.
"The study is continuing out to 12 months, and at 6 months the preliminary data so far are showing sustained positive improvements in all measures of quality of life, reduction of sweat, and impact on daily living," Dr. Lupin said in an interview. Many patients also reported reduction of axillary hair, and a few patients noted improvement in odor, he said.
Overall, patient satisfaction, as measured by the Dermatology Life Quality Index (DLQI), was 96% at 3 months.
When Dr. Lupin was asked whether miraDry had any disadvantages compared with botulinum toxin type A, he said that there are several short-term advantages to botulinum toxin type A: It is a quicker and easier procedure (about 5-10 minutes) with sweat reduction occurring in just a few days, versus 1 hour for the miraDry procedure, which requires anesthesia and can take a week or longer to produce results. Moreover, botulinum toxin type A is a single treatment and its benefit lasts about 6-8 months, whereas miraDry takes 1-3 sessions for a benefit of at least a year and possibly longer.
Indeed, Dr. Kilmer said in an interview, duration of benefit is an advantage of miraDry over just about every current hyperhidrosis treatment other than sympathectomy, a procedure that is rarely done. So far, the longest miraDry has been studied is 12 months, so "we can’t say for sure, but it didn’t drop off much during that time, so we expect it will last much longer," she said.
Dr. Mathew Avram, director of the Massachusetts General Hospital Dermatology Laser and Cosmetic Center, Boston, said, "This is innovative and interesting technology to address an issue that is very problematic for patients. We need to learn a little more about the duration of these benefits and what if any side effects may be created with repeated treatments over time."
The studies were sponsored by Miramar Labs. Dr. Lupin disclosed that he received a research grant for the study and travel expenses from Miramar, and also received honoraria and travel expenses from Allergan. Dr. Kilmer disclosed that she received research support from Miramar. Dr. Avram is a stockholder in Zeltiq.
GRAPEVINE, TEX. – A novel microwave device significantly reduced underarm sweating in two studies involving a total of 151 patients with axillary hyperhidrosis.
The condition affects millions of people, yet current treatments are limited by either duration of effect or efficacy. The microwave device has the potential for a longer-term and possibly even permanent effect via eradication of eccrine sweat glands, Dr. Suzanne L. Kilmer and Dr. Mark Lupin reported in separate presentations at the annual meeting of the American Society for Laser Medicine and Surgery.
Miramar Labs' miraDry system focuses microwave energy to selectively heat the interface between the skin and underlying fat, where the sweat glands reside. The system comprises a console, handpiece, and disposable tip. The in-office procedure takes approximately 40 minutes. The system was cleared for licensure in the United States by the Food and Drug Administration in January 2011, and will be marketed later this year, according to a company spokesperson.
Dr. Kilmer, of the Laser and Skin Surgery Center of Northern California, Sacramento, presented data on a second-generation version of the device that was FDA approved but never marketed. In a multicenter, sham-controlled study, 120 adults with hyperhidrosis were randomized to miraDry (81) or sham treatment (39) for one to three sessions. The patients had to have a Hyperhidrosis Disease Severity Scale (HDSS) score of 3 or 4 ("my underarm sweating is barely tolerable and frequently/always interferes with my daily activities") at baseline, and gravimetric readings greater than 50 mg at each axilla (in 5 minutes).
Follow-up was 6 months for the sham group and 12 months for the treatment group. Responders were defined as subjects reporting a reduction to an HDSS score of 1 ("My underarm sweating is never noticeable and never interferes with daily activities") or 2 ("My underarm sweating is tolerable but sometimes interferes with daily activities"). The patients had a mean age of 33 years, 58% were female, and 84% were white.
Efficacy for the treatment group was 89% at 30 days, 74% at 3 months, 67% at 6 months, and 69% at the 9- and 12-month visits. In the sham group, efficacy was 54% at 30 days and 44% for the 3- and 6-month visits. At all time points, the differences between the treatment and sham groups were significant, Dr. Kilmer reported.
Treatment-related adverse events were generally mild, and all but one resolved. The most common adverse events in the treatment group were transient patches of altered sensation in the treatment limb, occurring in 8 patients (10%), and axillary pain requiring prescription medication in 5 patients (6%). Most subjects experienced transient post-treatment local sequelae in the axilla such as edema, tenderness, and bruising.
Follow-up of the treated subjects showed stable efficacy through 12 months, she said.
The second study, involving 31 patients, investigated the third-generation, optimized version of the device. As in the earlier study, all patients had to have primary axillary hyperhidrosis with an HDSS score of 3 or 4. The patients also were required to have a gravimetric sweat assessment of at least 50 mg in each axilla (in 5 minutes). None of the patients had surgery for axillary hyperhidrosis or botulinum toxin injections in the axillae in the prior 12 months, said Dr. Lupin of Cosmedica Laser Centre, Victoria, B.C.
The patients had a mean age of 33 years (range 18-65 years), and three-quarters were female. They had a mean BMI of 24.8 kg/m2. Of 28 patients seen at 30 days, all but one (96%) had HDSS scores of 1 or 2. Of 25 seen at 3 months, all had HDSS scores of 1 or 2 (100% efficacy). Gravimetric assessments 3 months after treatment showed that 94% of patients had at least a 50% reduction in axillary sweat compared with baseline, with an average sweat reduction of 82%.
Nineteen of the 31 initially enrolled patients (61%) experienced at least one treatment-related adverse event, of which most (88%) were rated as mild. The most common were numbness or tingling in an area of the treated limb (12 patients, 39%), and edema in the chest or treatment limb lasting about a week (9 patients, 29%). Most of the patients also experienced acute post-treatment transient effects in the treatment area such as localized edema, tenderness, or erythema. Follow-up of safety is ongoing, Dr. Lupin noted.
"The study is continuing out to 12 months, and at 6 months the preliminary data so far are showing sustained positive improvements in all measures of quality of life, reduction of sweat, and impact on daily living," Dr. Lupin said in an interview. Many patients also reported reduction of axillary hair, and a few patients noted improvement in odor, he said.
Overall, patient satisfaction, as measured by the Dermatology Life Quality Index (DLQI), was 96% at 3 months.
When Dr. Lupin was asked whether miraDry had any disadvantages compared with botulinum toxin type A, he said that there are several short-term advantages to botulinum toxin type A: It is a quicker and easier procedure (about 5-10 minutes) with sweat reduction occurring in just a few days, versus 1 hour for the miraDry procedure, which requires anesthesia and can take a week or longer to produce results. Moreover, botulinum toxin type A is a single treatment and its benefit lasts about 6-8 months, whereas miraDry takes 1-3 sessions for a benefit of at least a year and possibly longer.
Indeed, Dr. Kilmer said in an interview, duration of benefit is an advantage of miraDry over just about every current hyperhidrosis treatment other than sympathectomy, a procedure that is rarely done. So far, the longest miraDry has been studied is 12 months, so "we can’t say for sure, but it didn’t drop off much during that time, so we expect it will last much longer," she said.
Dr. Mathew Avram, director of the Massachusetts General Hospital Dermatology Laser and Cosmetic Center, Boston, said, "This is innovative and interesting technology to address an issue that is very problematic for patients. We need to learn a little more about the duration of these benefits and what if any side effects may be created with repeated treatments over time."
The studies were sponsored by Miramar Labs. Dr. Lupin disclosed that he received a research grant for the study and travel expenses from Miramar, and also received honoraria and travel expenses from Allergan. Dr. Kilmer disclosed that she received research support from Miramar. Dr. Avram is a stockholder in Zeltiq.
GRAPEVINE, TEX. – A novel microwave device significantly reduced underarm sweating in two studies involving a total of 151 patients with axillary hyperhidrosis.
The condition affects millions of people, yet current treatments are limited by either duration of effect or efficacy. The microwave device has the potential for a longer-term and possibly even permanent effect via eradication of eccrine sweat glands, Dr. Suzanne L. Kilmer and Dr. Mark Lupin reported in separate presentations at the annual meeting of the American Society for Laser Medicine and Surgery.
Miramar Labs' miraDry system focuses microwave energy to selectively heat the interface between the skin and underlying fat, where the sweat glands reside. The system comprises a console, handpiece, and disposable tip. The in-office procedure takes approximately 40 minutes. The system was cleared for licensure in the United States by the Food and Drug Administration in January 2011, and will be marketed later this year, according to a company spokesperson.
Dr. Kilmer, of the Laser and Skin Surgery Center of Northern California, Sacramento, presented data on a second-generation version of the device that was FDA approved but never marketed. In a multicenter, sham-controlled study, 120 adults with hyperhidrosis were randomized to miraDry (81) or sham treatment (39) for one to three sessions. The patients had to have a Hyperhidrosis Disease Severity Scale (HDSS) score of 3 or 4 ("my underarm sweating is barely tolerable and frequently/always interferes with my daily activities") at baseline, and gravimetric readings greater than 50 mg at each axilla (in 5 minutes).
Follow-up was 6 months for the sham group and 12 months for the treatment group. Responders were defined as subjects reporting a reduction to an HDSS score of 1 ("My underarm sweating is never noticeable and never interferes with daily activities") or 2 ("My underarm sweating is tolerable but sometimes interferes with daily activities"). The patients had a mean age of 33 years, 58% were female, and 84% were white.
Efficacy for the treatment group was 89% at 30 days, 74% at 3 months, 67% at 6 months, and 69% at the 9- and 12-month visits. In the sham group, efficacy was 54% at 30 days and 44% for the 3- and 6-month visits. At all time points, the differences between the treatment and sham groups were significant, Dr. Kilmer reported.
Treatment-related adverse events were generally mild, and all but one resolved. The most common adverse events in the treatment group were transient patches of altered sensation in the treatment limb, occurring in 8 patients (10%), and axillary pain requiring prescription medication in 5 patients (6%). Most subjects experienced transient post-treatment local sequelae in the axilla such as edema, tenderness, and bruising.
Follow-up of the treated subjects showed stable efficacy through 12 months, she said.
The second study, involving 31 patients, investigated the third-generation, optimized version of the device. As in the earlier study, all patients had to have primary axillary hyperhidrosis with an HDSS score of 3 or 4. The patients also were required to have a gravimetric sweat assessment of at least 50 mg in each axilla (in 5 minutes). None of the patients had surgery for axillary hyperhidrosis or botulinum toxin injections in the axillae in the prior 12 months, said Dr. Lupin of Cosmedica Laser Centre, Victoria, B.C.
The patients had a mean age of 33 years (range 18-65 years), and three-quarters were female. They had a mean BMI of 24.8 kg/m2. Of 28 patients seen at 30 days, all but one (96%) had HDSS scores of 1 or 2. Of 25 seen at 3 months, all had HDSS scores of 1 or 2 (100% efficacy). Gravimetric assessments 3 months after treatment showed that 94% of patients had at least a 50% reduction in axillary sweat compared with baseline, with an average sweat reduction of 82%.
Nineteen of the 31 initially enrolled patients (61%) experienced at least one treatment-related adverse event, of which most (88%) were rated as mild. The most common were numbness or tingling in an area of the treated limb (12 patients, 39%), and edema in the chest or treatment limb lasting about a week (9 patients, 29%). Most of the patients also experienced acute post-treatment transient effects in the treatment area such as localized edema, tenderness, or erythema. Follow-up of safety is ongoing, Dr. Lupin noted.
"The study is continuing out to 12 months, and at 6 months the preliminary data so far are showing sustained positive improvements in all measures of quality of life, reduction of sweat, and impact on daily living," Dr. Lupin said in an interview. Many patients also reported reduction of axillary hair, and a few patients noted improvement in odor, he said.
Overall, patient satisfaction, as measured by the Dermatology Life Quality Index (DLQI), was 96% at 3 months.
When Dr. Lupin was asked whether miraDry had any disadvantages compared with botulinum toxin type A, he said that there are several short-term advantages to botulinum toxin type A: It is a quicker and easier procedure (about 5-10 minutes) with sweat reduction occurring in just a few days, versus 1 hour for the miraDry procedure, which requires anesthesia and can take a week or longer to produce results. Moreover, botulinum toxin type A is a single treatment and its benefit lasts about 6-8 months, whereas miraDry takes 1-3 sessions for a benefit of at least a year and possibly longer.
Indeed, Dr. Kilmer said in an interview, duration of benefit is an advantage of miraDry over just about every current hyperhidrosis treatment other than sympathectomy, a procedure that is rarely done. So far, the longest miraDry has been studied is 12 months, so "we can’t say for sure, but it didn’t drop off much during that time, so we expect it will last much longer," she said.
Dr. Mathew Avram, director of the Massachusetts General Hospital Dermatology Laser and Cosmetic Center, Boston, said, "This is innovative and interesting technology to address an issue that is very problematic for patients. We need to learn a little more about the duration of these benefits and what if any side effects may be created with repeated treatments over time."
The studies were sponsored by Miramar Labs. Dr. Lupin disclosed that he received a research grant for the study and travel expenses from Miramar, and also received honoraria and travel expenses from Allergan. Dr. Kilmer disclosed that she received research support from Miramar. Dr. Avram is a stockholder in Zeltiq.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY FOR LASER MEDICINE AND SURGERY
Major Finding: First study: Efficacy of treatment in 81 patients as determined by HDSS scores was 89% at 30 days and 67% at 6 months. Second study: Of 25 patients seen at 3 months, all had HDSS scores of 1 or 2 (100% efficacy).
Data Source: Two studies of 120 and 31 patients, respectively, with axillary hyperhidrosis
Disclosures: The studies were sponsored by Miramar Labs. Dr. Lupin disclosed that he received a research grant for the study and travel expenses from Miramar, and also received honoraria and travel expenses from Allergan. Dr. Kilmer disclosed that she received research support from Miramar. Dr. Avram is a stockholder in Zeltiq.
Lignin Peroxidase
Lignin peroxidase, a novel skin-lightening active agent that is derived from a fungus, is being studied with some interest and is being developed as an ingredient in products to treat pigmentation disorders.
Melanin, the dark pigment in the skin, is produced in the basal layer of the epidermis by melanocytes. Melanocytes make melanin, which is packaged into melanosomes and then transferred to the epidermal cells (keratinocytes). Accumulation of melanin in the epidermis is the main cause of pigmentation disorders, which are observed in all demographic groups but most commonly in people with darker skin types.
Excessive sun exposure in dark and light skin types can lead to unwanted accumulation of pigment (known as solar lentigo) in the skin. Pigmentation disorders are notoriously difficult to treat. Melanin is a very durable compound, and researchers have been largely unsuccessful in finding ways to break down melanin to reduce unwanted skin pigment. The existing topical treatments for skin lightening focus on the prevention of melanin formation by blocking tyrosinase and inhibiting its biosynthesis; by preventing the stimulation of melanocytes by UVA: or by blocking the transfer of melanosomes to keratinocytes via the PAR-2 receptor.
Alternative to Hydroquinone
Historically, the most effective treatments for skin lightening have contained hydroquinone. However, hydroquinone has become controversial, and related safety concerns have prompted research into alternative agents to treat skin pigmentation disorders. In addition, the skin develops tachyphylaxis to hydroquinone requiring 1-month "holidays" in order to maintain effectiveness, and a subset of people may develop contact allergy to hydroquinone. Many other compounds have been studied for the treatment of pigmentation disorders, including retinoids, mequinol, azelaic acid, arbutin, kojic acid, aloesin, licorice extract, ascorbic acid, soy proteins, N-acetyl glucosamine, and most recently, lignin peroxidase.
The enzyme lignin peroxidase (LIP) was first identified in 1984 (Arch. Biochem. Biophys. 1984;234:353-62), and has been researched for many years as a potential agent to break down lignin to whiten wood pulp in paper production. It was later found to break down eumelanin, which has a chemical structure similar to lignin. The development of lignin peroxidase as a skin-lightening agent resulted from these discoveries (U.S. Patent and Trademark Office Patent Application 20060051305). This novel skin-lightening active ingredient is produced extracellularly during submerged fermentation of the fungus Phanerochaete chrysosporium 3 (Biotechnol. Bioproc. E. 2004;9:153-68) and then purified from the fermented liquid medium (Lonza of Switzerland).The LIP enzyme (trademarked as Melanozyme) identifies eumelanin in the epidermis and specifically breaks down the pigment without affecting melanin biosynthesis or blocking tyrosinase. Although there are other types of lignin peroxidase enzymes, at this point, Melanozyme is the only one that has been developed and proved to be effective for skin lightening. Melanozyme is a glycoprotein active at pH 2-4.5 and inactive above that pH level. (The normal pH of skin is around 5.5, with slight variations between 5.0 and 6.5.)
Product Based on Lignin Peroxidase
Melanozyme is currently proprietary and is available only in a new skin-lightening product known on the market as Elure The Elure products are presented in a two-sided dispenser with one side containing the Melanozyme component and the other side an activator. Melanozyme alone has little ability to lighten skin, and first needs to be oxidized by hydrogen peroxide (0.012% in the activator) to enter an "activated state." The activator, which contains a small amount of hydrogen peroxide, is applied to the surface of the skin after the Melanozyme.
When applied to skin, the products that contain the Melanozyme and the activator have to be slightly acidic and buffered in order for the enzyme to perform. In addition, the enzyme is required to be first oxidized by H2O2, and then reduced by a substrate molecule (for example, veratryl alcohol) before the melanin is oxidized. After application of Elure lotion or cream, the skin pH is temporarily reduced to 3.5 but subsequently increases to its normal level of around 5.5. As the skin surface returns to the normal pH level, the enzyme is inactivated. It becomes a simple glycoprotein and is hydrolyzed in the skin by the naturally present proteases and other glycosidases into amino acids.
The safety of lignin peroxidase as a skin-lightening active ingredient has been demonstrated in preclinical studies (data on file at Rakuto Bio Technologies Ltd., 5 Carmel Street, P.O. Box 528, New Industrial Park, Yokneam 20692 Israel) with doses that are 17,000 times the recommended dose without prompting any side effects. LIP is nonmutagenic and nonirritating to eyes. The potential for skin irritation is very low, and in studies of 50 subjects each, there were no reports of skin irritation during acute sensitivity or cumulative sensitivity, or when used in sensitized skin.
Conclusion
Three open-label clinical trials and one double-blind, split-face controlled study (Rakuto Bio Technologies) in subjects with Fitzpatrick skin types II-IV have confirmed the tolerability of Elure. In all clinical studies conducted with Elure, significant improvement in tone, evenness, and dyspigmentation were achieved in most subjects within 1 month of use. Elure has been shown to be better tolerated and more effective than 2% hydroquinone. However, more studies are needed to compare the product against stronger concentrations of hydroquinone and other existing treatments, as well as to demonstrate its effectiveness in the treatment of other pigmentary conditions in a broader range of patients. The use of Elure in a combination skin care regimen with hydroquinone and glycolic acid has not been studied, but there is no reason to believe that these products would be incompatible. In fact, a glycolic cleanser that lowers the pH of the skin prior to application could theoretically enhance the efficacy of the product.
Dr. Baumann is on the advisory board of Syneron, the manufacturer of Elure.
Lignin peroxidase, a novel skin-lightening active agent that is derived from a fungus, is being studied with some interest and is being developed as an ingredient in products to treat pigmentation disorders.
Melanin, the dark pigment in the skin, is produced in the basal layer of the epidermis by melanocytes. Melanocytes make melanin, which is packaged into melanosomes and then transferred to the epidermal cells (keratinocytes). Accumulation of melanin in the epidermis is the main cause of pigmentation disorders, which are observed in all demographic groups but most commonly in people with darker skin types.
Excessive sun exposure in dark and light skin types can lead to unwanted accumulation of pigment (known as solar lentigo) in the skin. Pigmentation disorders are notoriously difficult to treat. Melanin is a very durable compound, and researchers have been largely unsuccessful in finding ways to break down melanin to reduce unwanted skin pigment. The existing topical treatments for skin lightening focus on the prevention of melanin formation by blocking tyrosinase and inhibiting its biosynthesis; by preventing the stimulation of melanocytes by UVA: or by blocking the transfer of melanosomes to keratinocytes via the PAR-2 receptor.
Alternative to Hydroquinone
Historically, the most effective treatments for skin lightening have contained hydroquinone. However, hydroquinone has become controversial, and related safety concerns have prompted research into alternative agents to treat skin pigmentation disorders. In addition, the skin develops tachyphylaxis to hydroquinone requiring 1-month "holidays" in order to maintain effectiveness, and a subset of people may develop contact allergy to hydroquinone. Many other compounds have been studied for the treatment of pigmentation disorders, including retinoids, mequinol, azelaic acid, arbutin, kojic acid, aloesin, licorice extract, ascorbic acid, soy proteins, N-acetyl glucosamine, and most recently, lignin peroxidase.
The enzyme lignin peroxidase (LIP) was first identified in 1984 (Arch. Biochem. Biophys. 1984;234:353-62), and has been researched for many years as a potential agent to break down lignin to whiten wood pulp in paper production. It was later found to break down eumelanin, which has a chemical structure similar to lignin. The development of lignin peroxidase as a skin-lightening agent resulted from these discoveries (U.S. Patent and Trademark Office Patent Application 20060051305). This novel skin-lightening active ingredient is produced extracellularly during submerged fermentation of the fungus Phanerochaete chrysosporium 3 (Biotechnol. Bioproc. E. 2004;9:153-68) and then purified from the fermented liquid medium (Lonza of Switzerland).The LIP enzyme (trademarked as Melanozyme) identifies eumelanin in the epidermis and specifically breaks down the pigment without affecting melanin biosynthesis or blocking tyrosinase. Although there are other types of lignin peroxidase enzymes, at this point, Melanozyme is the only one that has been developed and proved to be effective for skin lightening. Melanozyme is a glycoprotein active at pH 2-4.5 and inactive above that pH level. (The normal pH of skin is around 5.5, with slight variations between 5.0 and 6.5.)
Product Based on Lignin Peroxidase
Melanozyme is currently proprietary and is available only in a new skin-lightening product known on the market as Elure The Elure products are presented in a two-sided dispenser with one side containing the Melanozyme component and the other side an activator. Melanozyme alone has little ability to lighten skin, and first needs to be oxidized by hydrogen peroxide (0.012% in the activator) to enter an "activated state." The activator, which contains a small amount of hydrogen peroxide, is applied to the surface of the skin after the Melanozyme.
When applied to skin, the products that contain the Melanozyme and the activator have to be slightly acidic and buffered in order for the enzyme to perform. In addition, the enzyme is required to be first oxidized by H2O2, and then reduced by a substrate molecule (for example, veratryl alcohol) before the melanin is oxidized. After application of Elure lotion or cream, the skin pH is temporarily reduced to 3.5 but subsequently increases to its normal level of around 5.5. As the skin surface returns to the normal pH level, the enzyme is inactivated. It becomes a simple glycoprotein and is hydrolyzed in the skin by the naturally present proteases and other glycosidases into amino acids.
The safety of lignin peroxidase as a skin-lightening active ingredient has been demonstrated in preclinical studies (data on file at Rakuto Bio Technologies Ltd., 5 Carmel Street, P.O. Box 528, New Industrial Park, Yokneam 20692 Israel) with doses that are 17,000 times the recommended dose without prompting any side effects. LIP is nonmutagenic and nonirritating to eyes. The potential for skin irritation is very low, and in studies of 50 subjects each, there were no reports of skin irritation during acute sensitivity or cumulative sensitivity, or when used in sensitized skin.
Conclusion
Three open-label clinical trials and one double-blind, split-face controlled study (Rakuto Bio Technologies) in subjects with Fitzpatrick skin types II-IV have confirmed the tolerability of Elure. In all clinical studies conducted with Elure, significant improvement in tone, evenness, and dyspigmentation were achieved in most subjects within 1 month of use. Elure has been shown to be better tolerated and more effective than 2% hydroquinone. However, more studies are needed to compare the product against stronger concentrations of hydroquinone and other existing treatments, as well as to demonstrate its effectiveness in the treatment of other pigmentary conditions in a broader range of patients. The use of Elure in a combination skin care regimen with hydroquinone and glycolic acid has not been studied, but there is no reason to believe that these products would be incompatible. In fact, a glycolic cleanser that lowers the pH of the skin prior to application could theoretically enhance the efficacy of the product.
Dr. Baumann is on the advisory board of Syneron, the manufacturer of Elure.
Lignin peroxidase, a novel skin-lightening active agent that is derived from a fungus, is being studied with some interest and is being developed as an ingredient in products to treat pigmentation disorders.
Melanin, the dark pigment in the skin, is produced in the basal layer of the epidermis by melanocytes. Melanocytes make melanin, which is packaged into melanosomes and then transferred to the epidermal cells (keratinocytes). Accumulation of melanin in the epidermis is the main cause of pigmentation disorders, which are observed in all demographic groups but most commonly in people with darker skin types.
Excessive sun exposure in dark and light skin types can lead to unwanted accumulation of pigment (known as solar lentigo) in the skin. Pigmentation disorders are notoriously difficult to treat. Melanin is a very durable compound, and researchers have been largely unsuccessful in finding ways to break down melanin to reduce unwanted skin pigment. The existing topical treatments for skin lightening focus on the prevention of melanin formation by blocking tyrosinase and inhibiting its biosynthesis; by preventing the stimulation of melanocytes by UVA: or by blocking the transfer of melanosomes to keratinocytes via the PAR-2 receptor.
Alternative to Hydroquinone
Historically, the most effective treatments for skin lightening have contained hydroquinone. However, hydroquinone has become controversial, and related safety concerns have prompted research into alternative agents to treat skin pigmentation disorders. In addition, the skin develops tachyphylaxis to hydroquinone requiring 1-month "holidays" in order to maintain effectiveness, and a subset of people may develop contact allergy to hydroquinone. Many other compounds have been studied for the treatment of pigmentation disorders, including retinoids, mequinol, azelaic acid, arbutin, kojic acid, aloesin, licorice extract, ascorbic acid, soy proteins, N-acetyl glucosamine, and most recently, lignin peroxidase.
The enzyme lignin peroxidase (LIP) was first identified in 1984 (Arch. Biochem. Biophys. 1984;234:353-62), and has been researched for many years as a potential agent to break down lignin to whiten wood pulp in paper production. It was later found to break down eumelanin, which has a chemical structure similar to lignin. The development of lignin peroxidase as a skin-lightening agent resulted from these discoveries (U.S. Patent and Trademark Office Patent Application 20060051305). This novel skin-lightening active ingredient is produced extracellularly during submerged fermentation of the fungus Phanerochaete chrysosporium 3 (Biotechnol. Bioproc. E. 2004;9:153-68) and then purified from the fermented liquid medium (Lonza of Switzerland).The LIP enzyme (trademarked as Melanozyme) identifies eumelanin in the epidermis and specifically breaks down the pigment without affecting melanin biosynthesis or blocking tyrosinase. Although there are other types of lignin peroxidase enzymes, at this point, Melanozyme is the only one that has been developed and proved to be effective for skin lightening. Melanozyme is a glycoprotein active at pH 2-4.5 and inactive above that pH level. (The normal pH of skin is around 5.5, with slight variations between 5.0 and 6.5.)
Product Based on Lignin Peroxidase
Melanozyme is currently proprietary and is available only in a new skin-lightening product known on the market as Elure The Elure products are presented in a two-sided dispenser with one side containing the Melanozyme component and the other side an activator. Melanozyme alone has little ability to lighten skin, and first needs to be oxidized by hydrogen peroxide (0.012% in the activator) to enter an "activated state." The activator, which contains a small amount of hydrogen peroxide, is applied to the surface of the skin after the Melanozyme.
When applied to skin, the products that contain the Melanozyme and the activator have to be slightly acidic and buffered in order for the enzyme to perform. In addition, the enzyme is required to be first oxidized by H2O2, and then reduced by a substrate molecule (for example, veratryl alcohol) before the melanin is oxidized. After application of Elure lotion or cream, the skin pH is temporarily reduced to 3.5 but subsequently increases to its normal level of around 5.5. As the skin surface returns to the normal pH level, the enzyme is inactivated. It becomes a simple glycoprotein and is hydrolyzed in the skin by the naturally present proteases and other glycosidases into amino acids.
The safety of lignin peroxidase as a skin-lightening active ingredient has been demonstrated in preclinical studies (data on file at Rakuto Bio Technologies Ltd., 5 Carmel Street, P.O. Box 528, New Industrial Park, Yokneam 20692 Israel) with doses that are 17,000 times the recommended dose without prompting any side effects. LIP is nonmutagenic and nonirritating to eyes. The potential for skin irritation is very low, and in studies of 50 subjects each, there were no reports of skin irritation during acute sensitivity or cumulative sensitivity, or when used in sensitized skin.
Conclusion
Three open-label clinical trials and one double-blind, split-face controlled study (Rakuto Bio Technologies) in subjects with Fitzpatrick skin types II-IV have confirmed the tolerability of Elure. In all clinical studies conducted with Elure, significant improvement in tone, evenness, and dyspigmentation were achieved in most subjects within 1 month of use. Elure has been shown to be better tolerated and more effective than 2% hydroquinone. However, more studies are needed to compare the product against stronger concentrations of hydroquinone and other existing treatments, as well as to demonstrate its effectiveness in the treatment of other pigmentary conditions in a broader range of patients. The use of Elure in a combination skin care regimen with hydroquinone and glycolic acid has not been studied, but there is no reason to believe that these products would be incompatible. In fact, a glycolic cleanser that lowers the pH of the skin prior to application could theoretically enhance the efficacy of the product.
Dr. Baumann is on the advisory board of Syneron, the manufacturer of Elure.
Thin vs. Thick Melanomas: Both Carry Same SLN Risk
SAN ANTONIO – Patients with thin melanomas and positive deep margins on initial biopsy had the same incidence of sentinel lymph node metastasis as those with thicker melanomas, according to the results of a retrospective analysis of 260 patients with melanoma.
At least one positive sentinel lymph node was detected in 6 of 73 patients (8%) with a melanoma Breslow thickness of less than 0.8 mm and positive deep margins vs. 17 of 187 patients (9%) with a melanoma Breslow thickness of 0.8-2.0 mm, regardless of margin status (P = .82).
Immunohistochemistry was the most common method of identifying positive sentinel nodes in both the thin and thick melanoma groups (5 cases vs. 10 cases, respectively), Dr. Victor Koshenkov said at a symposium sponsored by the Society of Surgical Oncology.
The decision to perform sentinel node biopsy is largely driven by tumor thickness. When the initial biopsy of a thin melanoma shows positive deep margins, many clinicians will treat these cases as potentially thicker melanomas and perform sentinel lymph node (SLN) biopsy. There are few data on the impact of positive deep margins on surgical decision making, prognosis, and outcome, even though positive deep margins are the most common cause of incompletely measured or indeterminate tumor thickness, said Dr. Koshenkov of the department of surgery at Atlantic Health Memorial Hospital in Morristown, N.J.
He presented data from a retrospective analysis of 260 adult patients who underwent wide excision plus SLN biopsy for cutaneous melanoma from January 2004 to May 2010.
Demographics were not statistically different between the two groups, except for tumor site and Clark’s level, he said. In 53% of patients in the thicker melanoma group, the extremities were the primary tumor site vs. 38% in the thin melanoma group (P = .042), while 40% had Clark’s level IV-V vs. 22% in the thin melanoma group (P less than .001).
Multivariate regression analysis revealed that only female gender (P = .046; odds ratio, 2.68) and Clark’s level IV-V (P = .024; OR, 3.54) were significantly associated with an increased risk of positive SLNs. Belonging to the thin melanoma group versus the thicker melanoma group was not significant (P = .66; OR, 1.29) Dr. Koshenkov said.
The presence of residual disease approached, but did not reach, statistical significance (P = .062; OR, 2.60). Residual disease was found in about 20% of both groups. Only 4 of the 73 patients (5.5%) with positive SLNs in the thin melanoma group required further reexcision with wide margins.
Only 1 of the 23 sentinel node–positive patients went on to have additional positive nodes on completion of lymph node dissection, he said.
"Patients with thin melanomas and positive deep margins on initial biopsy have an incidence of SLN metastasis statistically no different than patients with thicker melanomas," Dr. Koshenkov concluded. "Thus, we believe that thin melanomas with positive deep margins should be treated with wide excision and a sentinel lymph node biopsy. Of course, these findings should be tested and verified in larger, multi-institutional databases."
During a discussion of the study, the audience questioned the ability to make almost a practice-changing conclusion based on the small number of patients and the low incidence of positive SLNs in the thicker melanoma group. Dr. Koshenkov replied that the reason the rate of sentinel node positivity was lower than predicted in this group was that a larger proportion of patients had melanomas 0.8-1 mm in depth, rather than 1-2 mm in depth.
Another attendee remarked that before concluding that every patient with a positive deep margin on initial biopsy needs to undergo SLN biopsy, it is important to know how many patients with positive sentinel nodes had a positive deep margin as their only indication or whether factors such as mitotic rate or ulceration played a role. Dr. Koshenkov said mitotic rate was not analyzed because it was not regularly included in the pathology report at the time of the review, and that ulceration and Clark's level IV were factored into the multivariate analysis.
The authors said they had no relevant financial disclosures.
SAN ANTONIO – Patients with thin melanomas and positive deep margins on initial biopsy had the same incidence of sentinel lymph node metastasis as those with thicker melanomas, according to the results of a retrospective analysis of 260 patients with melanoma.
At least one positive sentinel lymph node was detected in 6 of 73 patients (8%) with a melanoma Breslow thickness of less than 0.8 mm and positive deep margins vs. 17 of 187 patients (9%) with a melanoma Breslow thickness of 0.8-2.0 mm, regardless of margin status (P = .82).
Immunohistochemistry was the most common method of identifying positive sentinel nodes in both the thin and thick melanoma groups (5 cases vs. 10 cases, respectively), Dr. Victor Koshenkov said at a symposium sponsored by the Society of Surgical Oncology.
The decision to perform sentinel node biopsy is largely driven by tumor thickness. When the initial biopsy of a thin melanoma shows positive deep margins, many clinicians will treat these cases as potentially thicker melanomas and perform sentinel lymph node (SLN) biopsy. There are few data on the impact of positive deep margins on surgical decision making, prognosis, and outcome, even though positive deep margins are the most common cause of incompletely measured or indeterminate tumor thickness, said Dr. Koshenkov of the department of surgery at Atlantic Health Memorial Hospital in Morristown, N.J.
He presented data from a retrospective analysis of 260 adult patients who underwent wide excision plus SLN biopsy for cutaneous melanoma from January 2004 to May 2010.
Demographics were not statistically different between the two groups, except for tumor site and Clark’s level, he said. In 53% of patients in the thicker melanoma group, the extremities were the primary tumor site vs. 38% in the thin melanoma group (P = .042), while 40% had Clark’s level IV-V vs. 22% in the thin melanoma group (P less than .001).
Multivariate regression analysis revealed that only female gender (P = .046; odds ratio, 2.68) and Clark’s level IV-V (P = .024; OR, 3.54) were significantly associated with an increased risk of positive SLNs. Belonging to the thin melanoma group versus the thicker melanoma group was not significant (P = .66; OR, 1.29) Dr. Koshenkov said.
The presence of residual disease approached, but did not reach, statistical significance (P = .062; OR, 2.60). Residual disease was found in about 20% of both groups. Only 4 of the 73 patients (5.5%) with positive SLNs in the thin melanoma group required further reexcision with wide margins.
Only 1 of the 23 sentinel node–positive patients went on to have additional positive nodes on completion of lymph node dissection, he said.
"Patients with thin melanomas and positive deep margins on initial biopsy have an incidence of SLN metastasis statistically no different than patients with thicker melanomas," Dr. Koshenkov concluded. "Thus, we believe that thin melanomas with positive deep margins should be treated with wide excision and a sentinel lymph node biopsy. Of course, these findings should be tested and verified in larger, multi-institutional databases."
During a discussion of the study, the audience questioned the ability to make almost a practice-changing conclusion based on the small number of patients and the low incidence of positive SLNs in the thicker melanoma group. Dr. Koshenkov replied that the reason the rate of sentinel node positivity was lower than predicted in this group was that a larger proportion of patients had melanomas 0.8-1 mm in depth, rather than 1-2 mm in depth.
Another attendee remarked that before concluding that every patient with a positive deep margin on initial biopsy needs to undergo SLN biopsy, it is important to know how many patients with positive sentinel nodes had a positive deep margin as their only indication or whether factors such as mitotic rate or ulceration played a role. Dr. Koshenkov said mitotic rate was not analyzed because it was not regularly included in the pathology report at the time of the review, and that ulceration and Clark's level IV were factored into the multivariate analysis.
The authors said they had no relevant financial disclosures.
SAN ANTONIO – Patients with thin melanomas and positive deep margins on initial biopsy had the same incidence of sentinel lymph node metastasis as those with thicker melanomas, according to the results of a retrospective analysis of 260 patients with melanoma.
At least one positive sentinel lymph node was detected in 6 of 73 patients (8%) with a melanoma Breslow thickness of less than 0.8 mm and positive deep margins vs. 17 of 187 patients (9%) with a melanoma Breslow thickness of 0.8-2.0 mm, regardless of margin status (P = .82).
Immunohistochemistry was the most common method of identifying positive sentinel nodes in both the thin and thick melanoma groups (5 cases vs. 10 cases, respectively), Dr. Victor Koshenkov said at a symposium sponsored by the Society of Surgical Oncology.
The decision to perform sentinel node biopsy is largely driven by tumor thickness. When the initial biopsy of a thin melanoma shows positive deep margins, many clinicians will treat these cases as potentially thicker melanomas and perform sentinel lymph node (SLN) biopsy. There are few data on the impact of positive deep margins on surgical decision making, prognosis, and outcome, even though positive deep margins are the most common cause of incompletely measured or indeterminate tumor thickness, said Dr. Koshenkov of the department of surgery at Atlantic Health Memorial Hospital in Morristown, N.J.
He presented data from a retrospective analysis of 260 adult patients who underwent wide excision plus SLN biopsy for cutaneous melanoma from January 2004 to May 2010.
Demographics were not statistically different between the two groups, except for tumor site and Clark’s level, he said. In 53% of patients in the thicker melanoma group, the extremities were the primary tumor site vs. 38% in the thin melanoma group (P = .042), while 40% had Clark’s level IV-V vs. 22% in the thin melanoma group (P less than .001).
Multivariate regression analysis revealed that only female gender (P = .046; odds ratio, 2.68) and Clark’s level IV-V (P = .024; OR, 3.54) were significantly associated with an increased risk of positive SLNs. Belonging to the thin melanoma group versus the thicker melanoma group was not significant (P = .66; OR, 1.29) Dr. Koshenkov said.
The presence of residual disease approached, but did not reach, statistical significance (P = .062; OR, 2.60). Residual disease was found in about 20% of both groups. Only 4 of the 73 patients (5.5%) with positive SLNs in the thin melanoma group required further reexcision with wide margins.
Only 1 of the 23 sentinel node–positive patients went on to have additional positive nodes on completion of lymph node dissection, he said.
"Patients with thin melanomas and positive deep margins on initial biopsy have an incidence of SLN metastasis statistically no different than patients with thicker melanomas," Dr. Koshenkov concluded. "Thus, we believe that thin melanomas with positive deep margins should be treated with wide excision and a sentinel lymph node biopsy. Of course, these findings should be tested and verified in larger, multi-institutional databases."
During a discussion of the study, the audience questioned the ability to make almost a practice-changing conclusion based on the small number of patients and the low incidence of positive SLNs in the thicker melanoma group. Dr. Koshenkov replied that the reason the rate of sentinel node positivity was lower than predicted in this group was that a larger proportion of patients had melanomas 0.8-1 mm in depth, rather than 1-2 mm in depth.
Another attendee remarked that before concluding that every patient with a positive deep margin on initial biopsy needs to undergo SLN biopsy, it is important to know how many patients with positive sentinel nodes had a positive deep margin as their only indication or whether factors such as mitotic rate or ulceration played a role. Dr. Koshenkov said mitotic rate was not analyzed because it was not regularly included in the pathology report at the time of the review, and that ulceration and Clark's level IV were factored into the multivariate analysis.
The authors said they had no relevant financial disclosures.
FROM A SYMPOSIUM SPONSORED BY THE SOCIETY OF SURGICAL ONCOLOGY
Major Finding: A positive sentinel lymph node was identified in 8.2% of patients with thin melanomas and positive deep margins vs. 9% of those with thicker margins, regardless of margin status.
Data Source: Retrospective analysis of 260 patients with cutaneous melanoma.
Disclosures: The authors reported no relevant financial disclosures.
Nanotechnology Vehicle Speeds Numbing of Topical Lidocaine
MIAMI BEACH – A 4% topical lidocaine cream with a nanotechnology-based vehicle in development provided superior pain relief before facial filler injections, compared with the most commonly-used topical anesthetic cream, according to patient and physician ratings in two split-face studies.
"This really has potential as the next topical anesthetic," Dr. Glynis Ablon said at the South Beach Symposium.
Although most dermatologists and pediatricians apply topical anesthetic products (such as commonly used LMX4 cream) approximately 20 minutes before needle-based procedures, the efficacy of NTL4 (Cutiecaine Cream) was seen in as little as 5 minutes, Dr. Ablon said.
NTL4 is formulated with a novel nanoparticle vehicle (INParT Drug Delivery System) that passively transports active compounds deeper into the skin. "It's a great new way to deliver lidocaine into the skin faster," said Dr. Ablon, a dermatologist in private practice in Manhattan Beach, Calif. The technology is also being assessed for new topical hyaluronic acid and topical botulinum toxins.
Dr. Ablon and her colleague Dr. Mark Nestor, who is in private practice in Aventura, Fla., conducted an initial study of 30 patients with NTL4 applied to one side of their face and over-the-counter LMX4 to the contralateral side, followed by a 20-second massage. After 20 minutes, Restylane was injected for facial rejuvenation. Participants and blinded raters assessed pain relief.
"The study was started to see if we could get that [numbing effect] in 20 minutes, and obviously faster numbing times could be a great thing for dermatologists as well as pediatricians," Dr. Ablon said.
Twenty patients (67%) reported minimal or no pain when injected on the NTL4 side of their face, compared with 12 patients (40%) who reported minimal or no pain on the LMX4 treated side. "Overall, patients significantly preferred treatment with NTL4 over LMX4," she said. The mean visual analog score (VAS) for pain was 1.99 in the NTL4 group vs. 3.08 in the LMX4 group immediately after injection; 0.21 vs. 0.74 at 1 hour; and 0.07 and 0.31 at 3 hours postinjection.
Blinded investigators also rated significantly less pain with NTL4, Dr. Ablon said.
Adverse events were minor and included some tenderness and bruising. Also, there was "some edema we believe was due to the actual injection of the Restylane."
In a second study, Dr. Ablon and Dr. Nestor assessed 5-, 10-, and 15-minute application times. The 20 participants had a 30-second massage of the topical anesthetic and subsequent Restylane injections of their nasolabial folds.
Sixteen patients (80%) treated with NTL4 reported minimal or no pain, compared with one patient (5%) treated with LMX4, according to pooled data for subjective pain ratings after 5-, 10-, and 15-minute application times.
"We did show that they had significantly less pain on injection with the NTL4 at the 5- and 15-minute incubation, with a trend favoring it at 10 minutes," Dr. Ablon said.
Mean VAS pain scores were significantly lower for the NTL4 group when injected 5 minutes after application: 1.72 for NTL4 vs. 4.20 for LMX4. Patients also reported a significant difference in pain when injected 15 minutes after injection: 1.92 with NTL4 vs. 4.67 with LMX4. There was a nonsignificant trend favoring lower VAS scores with NTL4 after a 10-minute application time(1.08 for NTL4 and 3.00 for LMX4).
All patients preferred the NTL4 side versus the LMX4 side for pain relief. Blinded investigators’ evaluation of pain was also statistically significant in favor of the NTL4, Dr. Ablon said.
Adverse events were minor in this study as well. "We did have one patient with edema that we think might have been an actual reaction to lidocaine," she noted.
"There is a lot going on with nanotechnology, and [it] is here to stay," Dr. Ablon said. This technology can be found in suntan lotions, age-defying makeup, and even toothpaste that coats damaged enamel.
The studies were funded through an unrestricted educational grant from Innovatech. Dr. Ablon and Dr. Nestor are consultants and investigators for Transdermal Corp.
MIAMI BEACH – A 4% topical lidocaine cream with a nanotechnology-based vehicle in development provided superior pain relief before facial filler injections, compared with the most commonly-used topical anesthetic cream, according to patient and physician ratings in two split-face studies.
"This really has potential as the next topical anesthetic," Dr. Glynis Ablon said at the South Beach Symposium.
Although most dermatologists and pediatricians apply topical anesthetic products (such as commonly used LMX4 cream) approximately 20 minutes before needle-based procedures, the efficacy of NTL4 (Cutiecaine Cream) was seen in as little as 5 minutes, Dr. Ablon said.
NTL4 is formulated with a novel nanoparticle vehicle (INParT Drug Delivery System) that passively transports active compounds deeper into the skin. "It's a great new way to deliver lidocaine into the skin faster," said Dr. Ablon, a dermatologist in private practice in Manhattan Beach, Calif. The technology is also being assessed for new topical hyaluronic acid and topical botulinum toxins.
Dr. Ablon and her colleague Dr. Mark Nestor, who is in private practice in Aventura, Fla., conducted an initial study of 30 patients with NTL4 applied to one side of their face and over-the-counter LMX4 to the contralateral side, followed by a 20-second massage. After 20 minutes, Restylane was injected for facial rejuvenation. Participants and blinded raters assessed pain relief.
"The study was started to see if we could get that [numbing effect] in 20 minutes, and obviously faster numbing times could be a great thing for dermatologists as well as pediatricians," Dr. Ablon said.
Twenty patients (67%) reported minimal or no pain when injected on the NTL4 side of their face, compared with 12 patients (40%) who reported minimal or no pain on the LMX4 treated side. "Overall, patients significantly preferred treatment with NTL4 over LMX4," she said. The mean visual analog score (VAS) for pain was 1.99 in the NTL4 group vs. 3.08 in the LMX4 group immediately after injection; 0.21 vs. 0.74 at 1 hour; and 0.07 and 0.31 at 3 hours postinjection.
Blinded investigators also rated significantly less pain with NTL4, Dr. Ablon said.
Adverse events were minor and included some tenderness and bruising. Also, there was "some edema we believe was due to the actual injection of the Restylane."
In a second study, Dr. Ablon and Dr. Nestor assessed 5-, 10-, and 15-minute application times. The 20 participants had a 30-second massage of the topical anesthetic and subsequent Restylane injections of their nasolabial folds.
Sixteen patients (80%) treated with NTL4 reported minimal or no pain, compared with one patient (5%) treated with LMX4, according to pooled data for subjective pain ratings after 5-, 10-, and 15-minute application times.
"We did show that they had significantly less pain on injection with the NTL4 at the 5- and 15-minute incubation, with a trend favoring it at 10 minutes," Dr. Ablon said.
Mean VAS pain scores were significantly lower for the NTL4 group when injected 5 minutes after application: 1.72 for NTL4 vs. 4.20 for LMX4. Patients also reported a significant difference in pain when injected 15 minutes after injection: 1.92 with NTL4 vs. 4.67 with LMX4. There was a nonsignificant trend favoring lower VAS scores with NTL4 after a 10-minute application time(1.08 for NTL4 and 3.00 for LMX4).
All patients preferred the NTL4 side versus the LMX4 side for pain relief. Blinded investigators’ evaluation of pain was also statistically significant in favor of the NTL4, Dr. Ablon said.
Adverse events were minor in this study as well. "We did have one patient with edema that we think might have been an actual reaction to lidocaine," she noted.
"There is a lot going on with nanotechnology, and [it] is here to stay," Dr. Ablon said. This technology can be found in suntan lotions, age-defying makeup, and even toothpaste that coats damaged enamel.
The studies were funded through an unrestricted educational grant from Innovatech. Dr. Ablon and Dr. Nestor are consultants and investigators for Transdermal Corp.
MIAMI BEACH – A 4% topical lidocaine cream with a nanotechnology-based vehicle in development provided superior pain relief before facial filler injections, compared with the most commonly-used topical anesthetic cream, according to patient and physician ratings in two split-face studies.
"This really has potential as the next topical anesthetic," Dr. Glynis Ablon said at the South Beach Symposium.
Although most dermatologists and pediatricians apply topical anesthetic products (such as commonly used LMX4 cream) approximately 20 minutes before needle-based procedures, the efficacy of NTL4 (Cutiecaine Cream) was seen in as little as 5 minutes, Dr. Ablon said.
NTL4 is formulated with a novel nanoparticle vehicle (INParT Drug Delivery System) that passively transports active compounds deeper into the skin. "It's a great new way to deliver lidocaine into the skin faster," said Dr. Ablon, a dermatologist in private practice in Manhattan Beach, Calif. The technology is also being assessed for new topical hyaluronic acid and topical botulinum toxins.
Dr. Ablon and her colleague Dr. Mark Nestor, who is in private practice in Aventura, Fla., conducted an initial study of 30 patients with NTL4 applied to one side of their face and over-the-counter LMX4 to the contralateral side, followed by a 20-second massage. After 20 minutes, Restylane was injected for facial rejuvenation. Participants and blinded raters assessed pain relief.
"The study was started to see if we could get that [numbing effect] in 20 minutes, and obviously faster numbing times could be a great thing for dermatologists as well as pediatricians," Dr. Ablon said.
Twenty patients (67%) reported minimal or no pain when injected on the NTL4 side of their face, compared with 12 patients (40%) who reported minimal or no pain on the LMX4 treated side. "Overall, patients significantly preferred treatment with NTL4 over LMX4," she said. The mean visual analog score (VAS) for pain was 1.99 in the NTL4 group vs. 3.08 in the LMX4 group immediately after injection; 0.21 vs. 0.74 at 1 hour; and 0.07 and 0.31 at 3 hours postinjection.
Blinded investigators also rated significantly less pain with NTL4, Dr. Ablon said.
Adverse events were minor and included some tenderness and bruising. Also, there was "some edema we believe was due to the actual injection of the Restylane."
In a second study, Dr. Ablon and Dr. Nestor assessed 5-, 10-, and 15-minute application times. The 20 participants had a 30-second massage of the topical anesthetic and subsequent Restylane injections of their nasolabial folds.
Sixteen patients (80%) treated with NTL4 reported minimal or no pain, compared with one patient (5%) treated with LMX4, according to pooled data for subjective pain ratings after 5-, 10-, and 15-minute application times.
"We did show that they had significantly less pain on injection with the NTL4 at the 5- and 15-minute incubation, with a trend favoring it at 10 minutes," Dr. Ablon said.
Mean VAS pain scores were significantly lower for the NTL4 group when injected 5 minutes after application: 1.72 for NTL4 vs. 4.20 for LMX4. Patients also reported a significant difference in pain when injected 15 minutes after injection: 1.92 with NTL4 vs. 4.67 with LMX4. There was a nonsignificant trend favoring lower VAS scores with NTL4 after a 10-minute application time(1.08 for NTL4 and 3.00 for LMX4).
All patients preferred the NTL4 side versus the LMX4 side for pain relief. Blinded investigators’ evaluation of pain was also statistically significant in favor of the NTL4, Dr. Ablon said.
Adverse events were minor in this study as well. "We did have one patient with edema that we think might have been an actual reaction to lidocaine," she noted.
"There is a lot going on with nanotechnology, and [it] is here to stay," Dr. Ablon said. This technology can be found in suntan lotions, age-defying makeup, and even toothpaste that coats damaged enamel.
The studies were funded through an unrestricted educational grant from Innovatech. Dr. Ablon and Dr. Nestor are consultants and investigators for Transdermal Corp.
FROM THE SOUTH BEACH SYMPOSIUM
Major Finding: Sixteen patients (80%) treated with NTL4 reported minimal or no pain, compared with one patient (5%) treated with LMX4. Mean visual analog scale scores were significantly lower for injection pain with a 5-minute application: 1.72 in the NTL4 group vs. 4.20 in the LMX4 group.
Data Source: Split-face comparison of 20 patients applying NTL4 or LMX4 for 5, 10, or 15 minutes prior to facial filler injections.
Disclosures: Dr. Ablon and Dr. Nestor are consultants and investigators for Transdermal Corp.
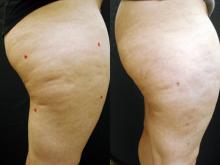
Investigational Nd:YAG Laser Plus 3-D Optical Fiber Targets Cellulite
GRAPEVINE, TEX. – An investigational sidelight 3-D optical fiber and 1440-nm Nd:YAG laser produced significant improvement of cellulite with one treatment at 6 months in a study of 15 healthy women.
Cynosure Inc.'s Cellulaze Cellulite Laser Workstation's 1440 wavelength is well absorbed by adipose tissue and water. The side-firing SideLight 3-D optical fiber thermally subcises subcutaneous septa, deplanes fat cells, and heats dermal tissue to promote skin thickening and tightening, said Dr. Bruce E. Katz of the department of dermatology at Mount Sinai School of Medicine, New York.
The 15 women (aged 20-55 years) all had cellulite on their lateral or posterior thighs or buttocks, body mass indexes less than 30 kg/m2, and skin types I-V. Following local anesthesia, two 1.5-mm incisions were made, and the probe was inserted. Subcutaneous temperature was kept at lower than 47° C and surface temperature at lower than 40° C. The Nd:YAG laser delivered 1,000 J per 5- x 5-cm square.
Digital photographic evaluation by two independent observers at 6 months found that 68% of the women had excellent improvement in cellulite; Vectra 3-D imaging demonstrated significant improvement in 65% of them. By physician evaluation, 76% had good or excellent results (69% by patient evaluation). Vectra analysis of skin contour demonstrated an average 47% reduction in depth and 32% reduction in height of skin bulges, Dr. Katz reported at the annual meeting of the American Society for Laser Medicine and Surgery.
Histologically, there was an increase in coarser collagen and elastic fibers in the dermis, noted Dr. Katz, who is also director of the cosmetic surgery and laser clinic at Mount Sinai Medical Center.
Three patients experienced mild ecchymoses, and four had edema lasting less than a week. There were no other adverse events.
"This may be a game changer for the treatment of cellulite," Dr. Katz said.
Cynosure received CE marking certification for Cellulaze in the European Union in February. The U.S. Food and Drug Administration approved an Investigational Device Exemption (IDE) for the device, and a clinical IDE study is currently underway. Regulatory action on a 510(k) submission, filed in late 2010, is currently expected in the first half of 2011, according to a company statement.
Dr. Katz disclosed that he is a Cynosure stockholder.
GRAPEVINE, TEX. – An investigational sidelight 3-D optical fiber and 1440-nm Nd:YAG laser produced significant improvement of cellulite with one treatment at 6 months in a study of 15 healthy women.
Cynosure Inc.'s Cellulaze Cellulite Laser Workstation's 1440 wavelength is well absorbed by adipose tissue and water. The side-firing SideLight 3-D optical fiber thermally subcises subcutaneous septa, deplanes fat cells, and heats dermal tissue to promote skin thickening and tightening, said Dr. Bruce E. Katz of the department of dermatology at Mount Sinai School of Medicine, New York.
The 15 women (aged 20-55 years) all had cellulite on their lateral or posterior thighs or buttocks, body mass indexes less than 30 kg/m2, and skin types I-V. Following local anesthesia, two 1.5-mm incisions were made, and the probe was inserted. Subcutaneous temperature was kept at lower than 47° C and surface temperature at lower than 40° C. The Nd:YAG laser delivered 1,000 J per 5- x 5-cm square.
Digital photographic evaluation by two independent observers at 6 months found that 68% of the women had excellent improvement in cellulite; Vectra 3-D imaging demonstrated significant improvement in 65% of them. By physician evaluation, 76% had good or excellent results (69% by patient evaluation). Vectra analysis of skin contour demonstrated an average 47% reduction in depth and 32% reduction in height of skin bulges, Dr. Katz reported at the annual meeting of the American Society for Laser Medicine and Surgery.
Histologically, there was an increase in coarser collagen and elastic fibers in the dermis, noted Dr. Katz, who is also director of the cosmetic surgery and laser clinic at Mount Sinai Medical Center.
Three patients experienced mild ecchymoses, and four had edema lasting less than a week. There were no other adverse events.
"This may be a game changer for the treatment of cellulite," Dr. Katz said.
Cynosure received CE marking certification for Cellulaze in the European Union in February. The U.S. Food and Drug Administration approved an Investigational Device Exemption (IDE) for the device, and a clinical IDE study is currently underway. Regulatory action on a 510(k) submission, filed in late 2010, is currently expected in the first half of 2011, according to a company statement.
Dr. Katz disclosed that he is a Cynosure stockholder.
GRAPEVINE, TEX. – An investigational sidelight 3-D optical fiber and 1440-nm Nd:YAG laser produced significant improvement of cellulite with one treatment at 6 months in a study of 15 healthy women.
Cynosure Inc.'s Cellulaze Cellulite Laser Workstation's 1440 wavelength is well absorbed by adipose tissue and water. The side-firing SideLight 3-D optical fiber thermally subcises subcutaneous septa, deplanes fat cells, and heats dermal tissue to promote skin thickening and tightening, said Dr. Bruce E. Katz of the department of dermatology at Mount Sinai School of Medicine, New York.
The 15 women (aged 20-55 years) all had cellulite on their lateral or posterior thighs or buttocks, body mass indexes less than 30 kg/m2, and skin types I-V. Following local anesthesia, two 1.5-mm incisions were made, and the probe was inserted. Subcutaneous temperature was kept at lower than 47° C and surface temperature at lower than 40° C. The Nd:YAG laser delivered 1,000 J per 5- x 5-cm square.
Digital photographic evaluation by two independent observers at 6 months found that 68% of the women had excellent improvement in cellulite; Vectra 3-D imaging demonstrated significant improvement in 65% of them. By physician evaluation, 76% had good or excellent results (69% by patient evaluation). Vectra analysis of skin contour demonstrated an average 47% reduction in depth and 32% reduction in height of skin bulges, Dr. Katz reported at the annual meeting of the American Society for Laser Medicine and Surgery.
Histologically, there was an increase in coarser collagen and elastic fibers in the dermis, noted Dr. Katz, who is also director of the cosmetic surgery and laser clinic at Mount Sinai Medical Center.
Three patients experienced mild ecchymoses, and four had edema lasting less than a week. There were no other adverse events.
"This may be a game changer for the treatment of cellulite," Dr. Katz said.
Cynosure received CE marking certification for Cellulaze in the European Union in February. The U.S. Food and Drug Administration approved an Investigational Device Exemption (IDE) for the device, and a clinical IDE study is currently underway. Regulatory action on a 510(k) submission, filed in late 2010, is currently expected in the first half of 2011, according to a company statement.
Dr. Katz disclosed that he is a Cynosure stockholder.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY FOR LASER MEDICINE AND SURGERY
Major Finding: Digital photographic evaluation by two independent observers at 6 months found that 68% had excellent improvement in cellulite, and Vectra 3-D imaging demonstrated significant improvement in 65%. By physician evaluation, 76% had good or excellent results (69% by patient evaluation).
Data Source: A 6-month study of 15 women aged 20-55 years with cellulite on their lateral or posterior thighs or buttocks.
Disclosures: The study was funded by Cynosure, of which Dr. Katz is a stockholder.
Lipolysis With Sodium Deoxycholate Reduces Submental Fat
MIAMI BEACH – Lipolysis with proprietary sodium deoxycholate injections significantly decreased the volume of submental fat in a randomized, vehicle-control, phase IIb study.
Investigators at 10 sites in the United States randomized 129 patients with moderate-to-severe "submental convexity" to low-dose or high-dose sodium deoxycholate (ATX-101, Kythera Biopharmaceuticals) or placebo. Administration was once a month for up to 5 months, Dr. Patricia S. Walker said at the South Beach Symposium.
Patients and clinicians rated changes at 16 weeks and 3 months after the final treatment, compared with baseline. At the same time, investigators also compared MRI volume measurements as an objective measure of fat changes.
MRI revealed significant reductions in submental fat at week 16 and week 32 from baseline, compared with placebo, for patients randomized to 2 mg/cm2 sodium deoxycholate (the high-dose group).
Seven dermatologists and three plastic surgeons at the 10 sites treated the participants and performed global assessments. The clinicians rated changes using a 5-point Clinician-Reported Submental Fat Impact Scale.
Again, the 2-mg/cm2 dose was more efficacious than placebo, with statistically significant reductions rated by doctors at week 16 and week 32. Although the effect on patients who received the 1-mg/cm2 dose (low-dose group) did not reach significance, this dose was also associated with improvement compared with placebo, suggesting a dose response, Dr. Walker said.
Patients also rated their own changes in submental fat using a 5-point Patient-Reported Submental Fat Impact Scale at baseline, week 16, and week 32.
Interestingly, all patients reported subjective improvements compared with baseline – even those assigned to vehicle-only injections. "Patients have a perception of self-improvement," Dr. Walker said. "There was very little change on MRI in the vehicle group, so it’s a true placebo effect – patients want to see a response."
Sodium deoxycholate selectively targets adipocytes. Histology showed infiltration of macrophages by day 3, with destruction and clearance of fat over time. "Twenty-eight days is about the right time for these injections to clear fat," said Dr. Walker, who is chief medical officer for Kythera Biopharmaceuticals.
Sodium deoxycholate is an endogenous, secondary bile acid. This is "how we break down fat in our diet," Dr. Walker said.
A total 114 patients completed the study. Fitzpatrick skin types ranged from I to VI, average age was 46 years (range 24-65 years), and the mean body mass index was 30.6 kg/m2 (range 19.5-48.8). "We found chin fat is not necessarily related to [body] size. It can be familial," Dr. Walker said.
Most adverse events were mild, transient, and limited to the chin area. "If you don’t have a safe product, you don’t have an aesthetic product," she said.
Treatment-related swelling, pain, and numbness were the most common adverse events. "There also was erythema in the vehicle group, so it was probably injection-technique related."
ATX-101 is a safe and effective treatment for submental fat reduction, Dr. Walker said. "It is nice to have an aesthetic product that everyone sees works."
Phase III trials are planned this year, according to Kythera Biopharmaceuticals.
Dr. Walker is an employee and shareholder of the company.
MIAMI BEACH – Lipolysis with proprietary sodium deoxycholate injections significantly decreased the volume of submental fat in a randomized, vehicle-control, phase IIb study.
Investigators at 10 sites in the United States randomized 129 patients with moderate-to-severe "submental convexity" to low-dose or high-dose sodium deoxycholate (ATX-101, Kythera Biopharmaceuticals) or placebo. Administration was once a month for up to 5 months, Dr. Patricia S. Walker said at the South Beach Symposium.
Patients and clinicians rated changes at 16 weeks and 3 months after the final treatment, compared with baseline. At the same time, investigators also compared MRI volume measurements as an objective measure of fat changes.
MRI revealed significant reductions in submental fat at week 16 and week 32 from baseline, compared with placebo, for patients randomized to 2 mg/cm2 sodium deoxycholate (the high-dose group).
Seven dermatologists and three plastic surgeons at the 10 sites treated the participants and performed global assessments. The clinicians rated changes using a 5-point Clinician-Reported Submental Fat Impact Scale.
Again, the 2-mg/cm2 dose was more efficacious than placebo, with statistically significant reductions rated by doctors at week 16 and week 32. Although the effect on patients who received the 1-mg/cm2 dose (low-dose group) did not reach significance, this dose was also associated with improvement compared with placebo, suggesting a dose response, Dr. Walker said.
Patients also rated their own changes in submental fat using a 5-point Patient-Reported Submental Fat Impact Scale at baseline, week 16, and week 32.
Interestingly, all patients reported subjective improvements compared with baseline – even those assigned to vehicle-only injections. "Patients have a perception of self-improvement," Dr. Walker said. "There was very little change on MRI in the vehicle group, so it’s a true placebo effect – patients want to see a response."
Sodium deoxycholate selectively targets adipocytes. Histology showed infiltration of macrophages by day 3, with destruction and clearance of fat over time. "Twenty-eight days is about the right time for these injections to clear fat," said Dr. Walker, who is chief medical officer for Kythera Biopharmaceuticals.
Sodium deoxycholate is an endogenous, secondary bile acid. This is "how we break down fat in our diet," Dr. Walker said.
A total 114 patients completed the study. Fitzpatrick skin types ranged from I to VI, average age was 46 years (range 24-65 years), and the mean body mass index was 30.6 kg/m2 (range 19.5-48.8). "We found chin fat is not necessarily related to [body] size. It can be familial," Dr. Walker said.
Most adverse events were mild, transient, and limited to the chin area. "If you don’t have a safe product, you don’t have an aesthetic product," she said.
Treatment-related swelling, pain, and numbness were the most common adverse events. "There also was erythema in the vehicle group, so it was probably injection-technique related."
ATX-101 is a safe and effective treatment for submental fat reduction, Dr. Walker said. "It is nice to have an aesthetic product that everyone sees works."
Phase III trials are planned this year, according to Kythera Biopharmaceuticals.
Dr. Walker is an employee and shareholder of the company.
MIAMI BEACH – Lipolysis with proprietary sodium deoxycholate injections significantly decreased the volume of submental fat in a randomized, vehicle-control, phase IIb study.
Investigators at 10 sites in the United States randomized 129 patients with moderate-to-severe "submental convexity" to low-dose or high-dose sodium deoxycholate (ATX-101, Kythera Biopharmaceuticals) or placebo. Administration was once a month for up to 5 months, Dr. Patricia S. Walker said at the South Beach Symposium.
Patients and clinicians rated changes at 16 weeks and 3 months after the final treatment, compared with baseline. At the same time, investigators also compared MRI volume measurements as an objective measure of fat changes.
MRI revealed significant reductions in submental fat at week 16 and week 32 from baseline, compared with placebo, for patients randomized to 2 mg/cm2 sodium deoxycholate (the high-dose group).
Seven dermatologists and three plastic surgeons at the 10 sites treated the participants and performed global assessments. The clinicians rated changes using a 5-point Clinician-Reported Submental Fat Impact Scale.
Again, the 2-mg/cm2 dose was more efficacious than placebo, with statistically significant reductions rated by doctors at week 16 and week 32. Although the effect on patients who received the 1-mg/cm2 dose (low-dose group) did not reach significance, this dose was also associated with improvement compared with placebo, suggesting a dose response, Dr. Walker said.
Patients also rated their own changes in submental fat using a 5-point Patient-Reported Submental Fat Impact Scale at baseline, week 16, and week 32.
Interestingly, all patients reported subjective improvements compared with baseline – even those assigned to vehicle-only injections. "Patients have a perception of self-improvement," Dr. Walker said. "There was very little change on MRI in the vehicle group, so it’s a true placebo effect – patients want to see a response."
Sodium deoxycholate selectively targets adipocytes. Histology showed infiltration of macrophages by day 3, with destruction and clearance of fat over time. "Twenty-eight days is about the right time for these injections to clear fat," said Dr. Walker, who is chief medical officer for Kythera Biopharmaceuticals.
Sodium deoxycholate is an endogenous, secondary bile acid. This is "how we break down fat in our diet," Dr. Walker said.
A total 114 patients completed the study. Fitzpatrick skin types ranged from I to VI, average age was 46 years (range 24-65 years), and the mean body mass index was 30.6 kg/m2 (range 19.5-48.8). "We found chin fat is not necessarily related to [body] size. It can be familial," Dr. Walker said.
Most adverse events were mild, transient, and limited to the chin area. "If you don’t have a safe product, you don’t have an aesthetic product," she said.
Treatment-related swelling, pain, and numbness were the most common adverse events. "There also was erythema in the vehicle group, so it was probably injection-technique related."
ATX-101 is a safe and effective treatment for submental fat reduction, Dr. Walker said. "It is nice to have an aesthetic product that everyone sees works."
Phase III trials are planned this year, according to Kythera Biopharmaceuticals.
Dr. Walker is an employee and shareholder of the company.
FROM THE SOUTH BEACH SYMPOSIUM
Major Finding: MRI revealed significant reductions in submental fat at week 16 and week 32 from baseline, compared with placebo, for patients randomized to 2 mg/cm2 sodium deoxycholate (the high-dose group).
Data Source: Investigators at 10 sites in the United States randomized 129 patients with moderate-to-severe "submental convexity" to low-dose or high-dose sodium deoxycholate or placebo.
Disclosures: Dr. Walker is an employee and shareholder of Kythera.
Skin of Color: Dangers of Black Market Bleaching Agents
Black market skin bleaching agents with potentially unsafe ingredients are a growing concern in some countries, according to an article published yesterday, April 11, by the Associated Press, "Skin Bleaching a Growing Problem in Jamaican Slums."
The use of these agents stems from societal and cultural beliefs that lighter skin could lead to a better way of life, particularly for those that are impoverished.
In Jamaica, for example, a "23-year-old resident of a Kingston ghetto hopes to transform her dark complexion to a cafe-au-lait-color common among Jamaica's elite and favored by many men in her neighborhood. She believes a fairer skin could be her ticket to a better life. So she spends her meager savings on cheap black-market concoctions that promise to lighten her pigment," (excerpt from AP story).
This phenomenon is not exclusive to Jamaica, but is inherent to many nations around the world. It has been documented in India, the Americas, the Middle East, the Philippines, and in some parts of Africa and Asia.
Lightening agents, such as hydroquinone, have been banned in over the counter preparations in Japan, the European Union, and Australia. OTC hydroquinone is still available in the United States in percentages of up to 2%. While hydroquinone use may lead to adverse events such as ochronosis, it is a rare event typically seen with use of much higher concentrations over longer periods of time. In my opinion, it is not low concentrations of hydroquinone, but other potentially harmful ingredients that may be found in some of the black market products that are of major concern.
"Lightening creams are not effectively regulated in Jamaica, where even roadside vendors sell tubes and plastic bags of powders and ointments from cardboard boxes stacked along sidewalks in market districts," according to the AP story. "Many of the tubes are unlabeled as to their actual ingredients," said Dr. Richard Desnoes, president of the Dermatology Association of Jamaica.
"Hardcore bleachers use illegal ointments smuggled into the Caribbean country that contain toxins like mercury, a metal that blocks production of melanin, which give skin its color, but can also be toxic," the story continued.
In addition, potent topical corticosteroids have been found in some of these products, leading to reports of atrophy, striae, and even symptoms of Cushing’s syndrome.
Black market skin bleaching agents with potentially unsafe ingredients are a growing concern in some countries, according to an article published yesterday, April 11, by the Associated Press, "Skin Bleaching a Growing Problem in Jamaican Slums."
The use of these agents stems from societal and cultural beliefs that lighter skin could lead to a better way of life, particularly for those that are impoverished.
In Jamaica, for example, a "23-year-old resident of a Kingston ghetto hopes to transform her dark complexion to a cafe-au-lait-color common among Jamaica's elite and favored by many men in her neighborhood. She believes a fairer skin could be her ticket to a better life. So she spends her meager savings on cheap black-market concoctions that promise to lighten her pigment," (excerpt from AP story).
This phenomenon is not exclusive to Jamaica, but is inherent to many nations around the world. It has been documented in India, the Americas, the Middle East, the Philippines, and in some parts of Africa and Asia.
Lightening agents, such as hydroquinone, have been banned in over the counter preparations in Japan, the European Union, and Australia. OTC hydroquinone is still available in the United States in percentages of up to 2%. While hydroquinone use may lead to adverse events such as ochronosis, it is a rare event typically seen with use of much higher concentrations over longer periods of time. In my opinion, it is not low concentrations of hydroquinone, but other potentially harmful ingredients that may be found in some of the black market products that are of major concern.
"Lightening creams are not effectively regulated in Jamaica, where even roadside vendors sell tubes and plastic bags of powders and ointments from cardboard boxes stacked along sidewalks in market districts," according to the AP story. "Many of the tubes are unlabeled as to their actual ingredients," said Dr. Richard Desnoes, president of the Dermatology Association of Jamaica.
"Hardcore bleachers use illegal ointments smuggled into the Caribbean country that contain toxins like mercury, a metal that blocks production of melanin, which give skin its color, but can also be toxic," the story continued.
In addition, potent topical corticosteroids have been found in some of these products, leading to reports of atrophy, striae, and even symptoms of Cushing’s syndrome.
Black market skin bleaching agents with potentially unsafe ingredients are a growing concern in some countries, according to an article published yesterday, April 11, by the Associated Press, "Skin Bleaching a Growing Problem in Jamaican Slums."
The use of these agents stems from societal and cultural beliefs that lighter skin could lead to a better way of life, particularly for those that are impoverished.
In Jamaica, for example, a "23-year-old resident of a Kingston ghetto hopes to transform her dark complexion to a cafe-au-lait-color common among Jamaica's elite and favored by many men in her neighborhood. She believes a fairer skin could be her ticket to a better life. So she spends her meager savings on cheap black-market concoctions that promise to lighten her pigment," (excerpt from AP story).
This phenomenon is not exclusive to Jamaica, but is inherent to many nations around the world. It has been documented in India, the Americas, the Middle East, the Philippines, and in some parts of Africa and Asia.
Lightening agents, such as hydroquinone, have been banned in over the counter preparations in Japan, the European Union, and Australia. OTC hydroquinone is still available in the United States in percentages of up to 2%. While hydroquinone use may lead to adverse events such as ochronosis, it is a rare event typically seen with use of much higher concentrations over longer periods of time. In my opinion, it is not low concentrations of hydroquinone, but other potentially harmful ingredients that may be found in some of the black market products that are of major concern.
"Lightening creams are not effectively regulated in Jamaica, where even roadside vendors sell tubes and plastic bags of powders and ointments from cardboard boxes stacked along sidewalks in market districts," according to the AP story. "Many of the tubes are unlabeled as to their actual ingredients," said Dr. Richard Desnoes, president of the Dermatology Association of Jamaica.
"Hardcore bleachers use illegal ointments smuggled into the Caribbean country that contain toxins like mercury, a metal that blocks production of melanin, which give skin its color, but can also be toxic," the story continued.
In addition, potent topical corticosteroids have been found in some of these products, leading to reports of atrophy, striae, and even symptoms of Cushing’s syndrome.