User login
The Clinical Learning Environment Review as a Model for Impactful Self-directed Quality Control Initiatives in Clinical Practice
As part of its Next Accreditation System, the Accreditation Council for Graduate Medical Education (ACGME) has introduced the Clinical Learning Environment Review (CLER) program, designed to assess the learning environment of institutions that have ACGME residency and fellowship programs.1 The CLER program emphasizes the responsibility of these hospitals, multispecialty groups, and other organizations to focus on quality and safety in the health care environment of resident learning and patient care. The expectation is that emphasis on quality of care in a residency training program will influence these physicians’ approach to quality of care after graduation.2,3 The Department of Dermatology at the University of Mississippi Medical Center (UMMC)(Jackson, Mississippi) saw CLER as an opportunity to demonstrate leadership in the patient safety movement.
CLER Program at UMMC
As a model CLER program at our institution, our project at the outset concentrated resident efforts on the focus areas specified by the ACGME (Table 1). We also were aware that our ACGME committee would need to answer questions during CLER site visits (Table 2). Because the data generated would not be used for accreditation decisions, there was no concern that exposing errors would jeopardize our postgraduate training certification.

The first 15 minutes of monthly faculty meetings were devoted to the presentation of a resident project, called a QA/QI (quality assurance/quality improvement) moment, that addressed ACGME focus areas 1, 2, 3, or 6 (Table 1). (Transitions in care [focus area 4] and work hours and fatigue [focus area 5] generally are less important issues in a predominantly outpatient specialty such as dermatology.) The residents were encouraged to identify areas where patient harm could occur due to poorly designed systems and to report situations in which patients actually were harmed.

Each project had to be approved by the department chairperson based on the following 4 requirements: First, the initiative must have the potential to notably impact patient safety and reduce harm. Second, residents with faculty support had to design methods to assess the identified problem. Third, participants had to design (to the best of their abilities) cost-effective and achievable interventions in a manner that would not produce unintended consequences. Fourth, residents were asked to devise a system to close the loop, ensuring that the effort put into the process was not wasted.
Findings From the CLER Program
The CLER program generates data on program and institutional attributes that have a salutatory effect on quality and safety, specifically involving 6 focus areas highlighted in Table 1. Putting residents at the center of efforts to improve the quality of care in our department proved critical to improving patient safety.
Involving residents in a series of QA/QI initiatives was logical because they rotate with faculty members. They also are in a position to view inconsistencies and to work to establish consistent patterns of patient care. In addition, our busy faculty members are charged with a variety of other clinical, educational, and administrative duties complicated by requirements in the design of a new residency training program. Faculty and residents working together were able to find problem areas in our department and devise solutions to improve those problems.
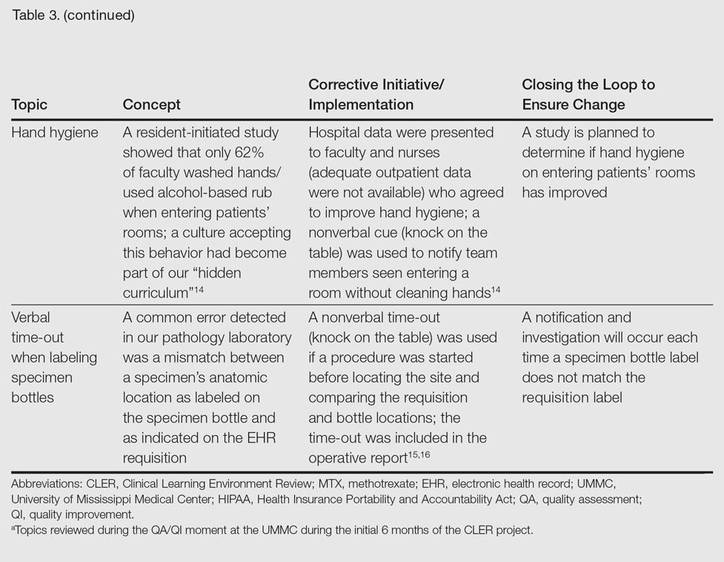
The CLER program involved a series of steps. Residents were charged with identifying errors (QA) and then devising a system to prevent similar errors from being repeated (QI)(Table 3). Efforts focused on preventing needless harm in our department. Initiatives developed by residents, who are closest to patients, have advantages over safety programs developed by the hospital’s administration. Residents became passionate about error prevention when they determined that their efforts could make a difference to patients.
Forward Thinking for Dermatology Practices
Perhaps there are lessons here that could apply to safety promotion in the practicing dermatologist’s office. The American Board of Dermatology, within the framework established by the American Board of Medical Specialties, requires physicians seeking recertification to participate in preapproved practice assessment QI exercises twice every 10 years.17 Six programs sponsored by the American Academy of Dermatology have now been approved in the areas of melanoma, biopsy follow-up measure, psoriasis, chronic urticaria, venous insufficiency, and laser- and light-based therapy for rejuvenation.18 An additional program has been approved for dermatopathologists through the American Society of Dermatopathology.19 None of these programs match the topics chosen by our residents in consultation with faculty to meet safety gaps identified in clinics at UMMC. Perhaps the next generation of performance improvement continuing medical education programs could include a pilot program for part 4 of Maintenance of Certification credit that is nonpunitive, patient focused, and allows dermatologists to design specific error-prevention solutions tailored to their individual practice in the same way residency programs are taking up this task.
- Nasca TJ, Philibert I, Brigham T, et al. The Next GME accreditation system—rationale and benefits. N Engl J Med. 2012;366:1051-1056.
- Philibert I, Gonzalez del Rey JA, Lannon C, et al. Quality improvement skills for pediatric residents: from lecture to implementation and sustainability. Acad Pediatr. 2014;14:40-46.
- Vidyarthi AR, Green AL, Rosenbluth G, et al. Engaging residents and fellows to improve institution-wide quality: the first six years of a novel financial incentive program. Acad Med. 2014;89:460-468.
- Brodell RT, Elewski B. Antifungal drug interactions. avoidance requires more than memorization. Postgrad Med. 2000;107:41-43.
- Kerr IG, Jolivet J, Collin JM, et al. Test dose for predicting high-dose methotrexate infusions. Clin Pharmacol Ther. 1983;33:44-51.
- Menter A, Korman NJ, Elmets CA, et al. Guidelines of care for the management of psoriasis and psoriatic arthritis: section 4. guidelines of care for the management and treatment of psoriasis with traditional systemic agents. J Am Acad Dermatol. 2009;61:451-485.
- Saporito FC, Menter MA. Methotrexate and psoriasis in the era of new biologic agents. J Am Acad Dermatol. 2004;50:301-309.
- Van Der Sijs H, Aarts J, Vulto A, et al. Overriding of drug safety alerts in computerized physician order entry. J Am Med Inform Assoc. 2006;13:138-147.
- Hunter KM. Implementation of an electronic medication administration record and bedside verification system. Online J Nurs Inform (OJNI). 2011;15:672.
- Nanji KC, Slight SP, Seger DL, et al. Overrides of medication-related clinical decision support alerts in outpatients. J Am Med Inform Assoc. 2014;21:487-491.
- Schedlbauer A, Prasad V, Mulvaney C, et al. What evidence supports the use of computerized alerts and prompts to improve clinicians’ prescribing behavior? J Am Med Inform Assoc. 2009;16:531-538.
- Lee EK, Mejia AF, Senior T, et al. Improving patient safety through medical alert management: an automated decision tool to reduce alert fatigue. AMIA Annu Symp Proc. 2010;2010:417-421.
- Brenner AB. Physician and nurse relationships, a key to patient safety. J Ky Med Assoc. 2007;105:165-169.
- Rush JL, Flowers RH, Casamiquela KM, et al. Research letter: the knock: an adjunct to education opening the door to improved outpatient hand hygiene. J Am Acad Dermatol. In press.
- Lee SL. The extended surgical time-out: does it improve quality and prevent wrong-site surgery? Perm J. 2010;14:19-23.
- Altpeter T, Luckhardt K, Lewis JN, et al. Expanded surgical time out: a key to real-time data collection and quality improvement. J Am Coll Surg. 2007;204:527-532.
- MOC requirements. American Board of Dermatology Web site. https://www.abderm.org/diplomates/fulfilling-moc-requirements/moc-requirements.aspx#PI. Accessed January 18, 2016.
- How AAD develops measures. American Academy of Dermatology Web site. https://www.aad.org/practice-tools/quality-care/quality-measures. Accessed January 20, 2016.
- Quality assurance programs. The American Society of Dermatopathology Web site. http://www.asdp.org/education/quality-assurance-programs. Accessed January 20, 2016.
As part of its Next Accreditation System, the Accreditation Council for Graduate Medical Education (ACGME) has introduced the Clinical Learning Environment Review (CLER) program, designed to assess the learning environment of institutions that have ACGME residency and fellowship programs.1 The CLER program emphasizes the responsibility of these hospitals, multispecialty groups, and other organizations to focus on quality and safety in the health care environment of resident learning and patient care. The expectation is that emphasis on quality of care in a residency training program will influence these physicians’ approach to quality of care after graduation.2,3 The Department of Dermatology at the University of Mississippi Medical Center (UMMC)(Jackson, Mississippi) saw CLER as an opportunity to demonstrate leadership in the patient safety movement.
CLER Program at UMMC
As a model CLER program at our institution, our project at the outset concentrated resident efforts on the focus areas specified by the ACGME (Table 1). We also were aware that our ACGME committee would need to answer questions during CLER site visits (Table 2). Because the data generated would not be used for accreditation decisions, there was no concern that exposing errors would jeopardize our postgraduate training certification.

The first 15 minutes of monthly faculty meetings were devoted to the presentation of a resident project, called a QA/QI (quality assurance/quality improvement) moment, that addressed ACGME focus areas 1, 2, 3, or 6 (Table 1). (Transitions in care [focus area 4] and work hours and fatigue [focus area 5] generally are less important issues in a predominantly outpatient specialty such as dermatology.) The residents were encouraged to identify areas where patient harm could occur due to poorly designed systems and to report situations in which patients actually were harmed.

Each project had to be approved by the department chairperson based on the following 4 requirements: First, the initiative must have the potential to notably impact patient safety and reduce harm. Second, residents with faculty support had to design methods to assess the identified problem. Third, participants had to design (to the best of their abilities) cost-effective and achievable interventions in a manner that would not produce unintended consequences. Fourth, residents were asked to devise a system to close the loop, ensuring that the effort put into the process was not wasted.
Findings From the CLER Program
The CLER program generates data on program and institutional attributes that have a salutatory effect on quality and safety, specifically involving 6 focus areas highlighted in Table 1. Putting residents at the center of efforts to improve the quality of care in our department proved critical to improving patient safety.
Involving residents in a series of QA/QI initiatives was logical because they rotate with faculty members. They also are in a position to view inconsistencies and to work to establish consistent patterns of patient care. In addition, our busy faculty members are charged with a variety of other clinical, educational, and administrative duties complicated by requirements in the design of a new residency training program. Faculty and residents working together were able to find problem areas in our department and devise solutions to improve those problems.
The CLER program involved a series of steps. Residents were charged with identifying errors (QA) and then devising a system to prevent similar errors from being repeated (QI)(Table 3). Efforts focused on preventing needless harm in our department. Initiatives developed by residents, who are closest to patients, have advantages over safety programs developed by the hospital’s administration. Residents became passionate about error prevention when they determined that their efforts could make a difference to patients.


Forward Thinking for Dermatology Practices
Perhaps there are lessons here that could apply to safety promotion in the practicing dermatologist’s office. The American Board of Dermatology, within the framework established by the American Board of Medical Specialties, requires physicians seeking recertification to participate in preapproved practice assessment QI exercises twice every 10 years.17 Six programs sponsored by the American Academy of Dermatology have now been approved in the areas of melanoma, biopsy follow-up measure, psoriasis, chronic urticaria, venous insufficiency, and laser- and light-based therapy for rejuvenation.18 An additional program has been approved for dermatopathologists through the American Society of Dermatopathology.19 None of these programs match the topics chosen by our residents in consultation with faculty to meet safety gaps identified in clinics at UMMC. Perhaps the next generation of performance improvement continuing medical education programs could include a pilot program for part 4 of Maintenance of Certification credit that is nonpunitive, patient focused, and allows dermatologists to design specific error-prevention solutions tailored to their individual practice in the same way residency programs are taking up this task.
As part of its Next Accreditation System, the Accreditation Council for Graduate Medical Education (ACGME) has introduced the Clinical Learning Environment Review (CLER) program, designed to assess the learning environment of institutions that have ACGME residency and fellowship programs.1 The CLER program emphasizes the responsibility of these hospitals, multispecialty groups, and other organizations to focus on quality and safety in the health care environment of resident learning and patient care. The expectation is that emphasis on quality of care in a residency training program will influence these physicians’ approach to quality of care after graduation.2,3 The Department of Dermatology at the University of Mississippi Medical Center (UMMC)(Jackson, Mississippi) saw CLER as an opportunity to demonstrate leadership in the patient safety movement.
CLER Program at UMMC
As a model CLER program at our institution, our project at the outset concentrated resident efforts on the focus areas specified by the ACGME (Table 1). We also were aware that our ACGME committee would need to answer questions during CLER site visits (Table 2). Because the data generated would not be used for accreditation decisions, there was no concern that exposing errors would jeopardize our postgraduate training certification.

The first 15 minutes of monthly faculty meetings were devoted to the presentation of a resident project, called a QA/QI (quality assurance/quality improvement) moment, that addressed ACGME focus areas 1, 2, 3, or 6 (Table 1). (Transitions in care [focus area 4] and work hours and fatigue [focus area 5] generally are less important issues in a predominantly outpatient specialty such as dermatology.) The residents were encouraged to identify areas where patient harm could occur due to poorly designed systems and to report situations in which patients actually were harmed.

Each project had to be approved by the department chairperson based on the following 4 requirements: First, the initiative must have the potential to notably impact patient safety and reduce harm. Second, residents with faculty support had to design methods to assess the identified problem. Third, participants had to design (to the best of their abilities) cost-effective and achievable interventions in a manner that would not produce unintended consequences. Fourth, residents were asked to devise a system to close the loop, ensuring that the effort put into the process was not wasted.
Findings From the CLER Program
The CLER program generates data on program and institutional attributes that have a salutatory effect on quality and safety, specifically involving 6 focus areas highlighted in Table 1. Putting residents at the center of efforts to improve the quality of care in our department proved critical to improving patient safety.
Involving residents in a series of QA/QI initiatives was logical because they rotate with faculty members. They also are in a position to view inconsistencies and to work to establish consistent patterns of patient care. In addition, our busy faculty members are charged with a variety of other clinical, educational, and administrative duties complicated by requirements in the design of a new residency training program. Faculty and residents working together were able to find problem areas in our department and devise solutions to improve those problems.
The CLER program involved a series of steps. Residents were charged with identifying errors (QA) and then devising a system to prevent similar errors from being repeated (QI)(Table 3). Efforts focused on preventing needless harm in our department. Initiatives developed by residents, who are closest to patients, have advantages over safety programs developed by the hospital’s administration. Residents became passionate about error prevention when they determined that their efforts could make a difference to patients.


Forward Thinking for Dermatology Practices
Perhaps there are lessons here that could apply to safety promotion in the practicing dermatologist’s office. The American Board of Dermatology, within the framework established by the American Board of Medical Specialties, requires physicians seeking recertification to participate in preapproved practice assessment QI exercises twice every 10 years.17 Six programs sponsored by the American Academy of Dermatology have now been approved in the areas of melanoma, biopsy follow-up measure, psoriasis, chronic urticaria, venous insufficiency, and laser- and light-based therapy for rejuvenation.18 An additional program has been approved for dermatopathologists through the American Society of Dermatopathology.19 None of these programs match the topics chosen by our residents in consultation with faculty to meet safety gaps identified in clinics at UMMC. Perhaps the next generation of performance improvement continuing medical education programs could include a pilot program for part 4 of Maintenance of Certification credit that is nonpunitive, patient focused, and allows dermatologists to design specific error-prevention solutions tailored to their individual practice in the same way residency programs are taking up this task.
- Nasca TJ, Philibert I, Brigham T, et al. The Next GME accreditation system—rationale and benefits. N Engl J Med. 2012;366:1051-1056.
- Philibert I, Gonzalez del Rey JA, Lannon C, et al. Quality improvement skills for pediatric residents: from lecture to implementation and sustainability. Acad Pediatr. 2014;14:40-46.
- Vidyarthi AR, Green AL, Rosenbluth G, et al. Engaging residents and fellows to improve institution-wide quality: the first six years of a novel financial incentive program. Acad Med. 2014;89:460-468.
- Brodell RT, Elewski B. Antifungal drug interactions. avoidance requires more than memorization. Postgrad Med. 2000;107:41-43.
- Kerr IG, Jolivet J, Collin JM, et al. Test dose for predicting high-dose methotrexate infusions. Clin Pharmacol Ther. 1983;33:44-51.
- Menter A, Korman NJ, Elmets CA, et al. Guidelines of care for the management of psoriasis and psoriatic arthritis: section 4. guidelines of care for the management and treatment of psoriasis with traditional systemic agents. J Am Acad Dermatol. 2009;61:451-485.
- Saporito FC, Menter MA. Methotrexate and psoriasis in the era of new biologic agents. J Am Acad Dermatol. 2004;50:301-309.
- Van Der Sijs H, Aarts J, Vulto A, et al. Overriding of drug safety alerts in computerized physician order entry. J Am Med Inform Assoc. 2006;13:138-147.
- Hunter KM. Implementation of an electronic medication administration record and bedside verification system. Online J Nurs Inform (OJNI). 2011;15:672.
- Nanji KC, Slight SP, Seger DL, et al. Overrides of medication-related clinical decision support alerts in outpatients. J Am Med Inform Assoc. 2014;21:487-491.
- Schedlbauer A, Prasad V, Mulvaney C, et al. What evidence supports the use of computerized alerts and prompts to improve clinicians’ prescribing behavior? J Am Med Inform Assoc. 2009;16:531-538.
- Lee EK, Mejia AF, Senior T, et al. Improving patient safety through medical alert management: an automated decision tool to reduce alert fatigue. AMIA Annu Symp Proc. 2010;2010:417-421.
- Brenner AB. Physician and nurse relationships, a key to patient safety. J Ky Med Assoc. 2007;105:165-169.
- Rush JL, Flowers RH, Casamiquela KM, et al. Research letter: the knock: an adjunct to education opening the door to improved outpatient hand hygiene. J Am Acad Dermatol. In press.
- Lee SL. The extended surgical time-out: does it improve quality and prevent wrong-site surgery? Perm J. 2010;14:19-23.
- Altpeter T, Luckhardt K, Lewis JN, et al. Expanded surgical time out: a key to real-time data collection and quality improvement. J Am Coll Surg. 2007;204:527-532.
- MOC requirements. American Board of Dermatology Web site. https://www.abderm.org/diplomates/fulfilling-moc-requirements/moc-requirements.aspx#PI. Accessed January 18, 2016.
- How AAD develops measures. American Academy of Dermatology Web site. https://www.aad.org/practice-tools/quality-care/quality-measures. Accessed January 20, 2016.
- Quality assurance programs. The American Society of Dermatopathology Web site. http://www.asdp.org/education/quality-assurance-programs. Accessed January 20, 2016.
- Nasca TJ, Philibert I, Brigham T, et al. The Next GME accreditation system—rationale and benefits. N Engl J Med. 2012;366:1051-1056.
- Philibert I, Gonzalez del Rey JA, Lannon C, et al. Quality improvement skills for pediatric residents: from lecture to implementation and sustainability. Acad Pediatr. 2014;14:40-46.
- Vidyarthi AR, Green AL, Rosenbluth G, et al. Engaging residents and fellows to improve institution-wide quality: the first six years of a novel financial incentive program. Acad Med. 2014;89:460-468.
- Brodell RT, Elewski B. Antifungal drug interactions. avoidance requires more than memorization. Postgrad Med. 2000;107:41-43.
- Kerr IG, Jolivet J, Collin JM, et al. Test dose for predicting high-dose methotrexate infusions. Clin Pharmacol Ther. 1983;33:44-51.
- Menter A, Korman NJ, Elmets CA, et al. Guidelines of care for the management of psoriasis and psoriatic arthritis: section 4. guidelines of care for the management and treatment of psoriasis with traditional systemic agents. J Am Acad Dermatol. 2009;61:451-485.
- Saporito FC, Menter MA. Methotrexate and psoriasis in the era of new biologic agents. J Am Acad Dermatol. 2004;50:301-309.
- Van Der Sijs H, Aarts J, Vulto A, et al. Overriding of drug safety alerts in computerized physician order entry. J Am Med Inform Assoc. 2006;13:138-147.
- Hunter KM. Implementation of an electronic medication administration record and bedside verification system. Online J Nurs Inform (OJNI). 2011;15:672.
- Nanji KC, Slight SP, Seger DL, et al. Overrides of medication-related clinical decision support alerts in outpatients. J Am Med Inform Assoc. 2014;21:487-491.
- Schedlbauer A, Prasad V, Mulvaney C, et al. What evidence supports the use of computerized alerts and prompts to improve clinicians’ prescribing behavior? J Am Med Inform Assoc. 2009;16:531-538.
- Lee EK, Mejia AF, Senior T, et al. Improving patient safety through medical alert management: an automated decision tool to reduce alert fatigue. AMIA Annu Symp Proc. 2010;2010:417-421.
- Brenner AB. Physician and nurse relationships, a key to patient safety. J Ky Med Assoc. 2007;105:165-169.
- Rush JL, Flowers RH, Casamiquela KM, et al. Research letter: the knock: an adjunct to education opening the door to improved outpatient hand hygiene. J Am Acad Dermatol. In press.
- Lee SL. The extended surgical time-out: does it improve quality and prevent wrong-site surgery? Perm J. 2010;14:19-23.
- Altpeter T, Luckhardt K, Lewis JN, et al. Expanded surgical time out: a key to real-time data collection and quality improvement. J Am Coll Surg. 2007;204:527-532.
- MOC requirements. American Board of Dermatology Web site. https://www.abderm.org/diplomates/fulfilling-moc-requirements/moc-requirements.aspx#PI. Accessed January 18, 2016.
- How AAD develops measures. American Academy of Dermatology Web site. https://www.aad.org/practice-tools/quality-care/quality-measures. Accessed January 20, 2016.
- Quality assurance programs. The American Society of Dermatopathology Web site. http://www.asdp.org/education/quality-assurance-programs. Accessed January 20, 2016.
Practice Points
- The Clinical Learning Environment Review mobilizes residency and fellowship training programs in the movement to improve the quality of patient care.
- Quality assessment/quality improvement (QA/QI) projects enhance communication between residents and faculty and promote systems that improve patient safety.
- Emphasis on resident-initiated QA/QI impacts quality of care in clinical practice long after graduation.
Postinflammatory Hyperpigmentation in Patients With Skin of Color
Postinflammatory hyperpigmentation (PIH) develops as darkly pigmented macules that occur after an inflammatory process of the skin such as acne, folliculitis, eczema, or shaving irritation. Patients with Fitzpatrick skin types III to VI usually are most commonly affected, and for many, the remnant pigmentation can be an even greater concern than the original inflammatory process.1,2 Reported treatments of PIH include tretinoin, hydroquinone, azelaic acid, and chemical peels. The ideal combination of therapy has yet to be delineated.
Tretinoin (Vitamin A Derivative)
Bulengo-Ransby et al3 performed one of the first clinical trials testing tretinoin cream 0.1% for PIH in patients with Fitzpatrick skin types IV to VI . The study included 54 patients (24 applied tretinoin and 30 applied a vehicle) with moderate to severe PIH on the face and arms. The patients were divided into therapy and placebo groups and were evaluated for 40 weeks. Changes were evaluated through colorimetry, light microscopy, histology, and photography, with significant clinical improvement in the tretinoin-treated group (P<.001).3 A double-blind, randomized study of 45 photoaged Chinese and Japanese patients using tretinoin cream 0.1% also was conducted for treatment of photoaging-associated hyperpigmented lesions of the face and hands. Assessment was done with clinical, colorimetric, and histological evaluation, with an overall statistical improvement noted in hyperpigmentation.4 Both of the above studies showed mild irritation (ie, retinoid dermatitis) with application of tretinoin, which creates a compliance issue in patients who are recommended to continue therapy with higher-strength tretinoin. This side-effect profile can be circumvented through gradual elevation in the strength of tretinoin.5
Combination Therapies
Combination therapies with tretinoin also have been used to improve PIH. Callender et al6 conducted a study evaluating the efficacy of clindamycin phosphate 1.2%–tretinoin 0.025% gel for the treatment of PIH secondary to mild to moderate acne in patients with Fitzpatrick skin types IV to VI. Thirty patients participated in the randomized, double-blinded, placebo-controlled study, with 15 patients in the clindamycin-tretinoin gel group and 15 in the placebo control group. Based on objective assessment using a chromameter and evaluator global acne severity scale score, clinical efficacy was demonstrated for treating acne and PIH as well as preventing further PIH.6
Hydroquinone Formulation (Tyrosine Inhibitor)
Hydroquinone bleaching cream has been the standard therapy for hyperpigmentation. It works by blocking the conversion of dihydroxyphenylalanine to melanin by inhibiting tyrosinase.7 Topical steroids directly inhibit the synthesis of melanin, and when combined with hydroquinone and tretinoin, they can be effective for short periods of time and may decrease the irritation of application.7,8 The most widely accepted formula consists of a topical steroid (triamcinolone cream 0.1%) in combination with hydroquinone 4% and tretinoin cream 0.05%.8 In a similar 12-week open-label study of 25 patients with darker skin types, Grimes9 used an alternative combination formula of hydroquinone 4% and retinol 0.15%. Overall improvement and tolerance was demonstrated through the use of colorimetry measurement. A combination of hydroquinone 4%, tretinoin 0.05%, and fluocinolone acetonide 0.01% also has been used effectively for the treatment of melasma.10 This formulation has been used more anecdotally for the treatment of PIH and has yet to have a randomized-controlled trial. The concern with repeated long-term use of hydroquinone remains. Permanent leukoderma, exogenous ochronosis, and hyperpigmentation of the surrounding normal skin (halo effect) can occur.
Azelaic Acid (Tyrosinase Inhibitor)
Azelaic acid is a dicarboxylic acid isolated from pityriasis versicolor that acts similar to a tyrosine inhibitor and has an antiproliferative effect toward abnormal melanocytes. Lowe et al11 conducted a randomized, double-blind, vehicle-controlled trial in patients with Fitzpatrick skin types IV through VI with facial hyperpigmentation using azelaic acid cream 20%. Over the course of 24 weeks, patients noted a decrease in overall pigment using both an investigator subjective scale and chromometer analysis.11
Kojic Acid (Tyrosinase Inhibitor)
Kojic acid is a tyrosinase inhibitor found in fungal metabolite species such as Acetobacter, Aspergillus, and Penicillium. It is commonly combined with other skin lightening agents such as hydroquinone or vitamin C to further enhance its efficacy. A randomized, 12-week, split-face study of Chinese women with melasma compared treatment with a glycolic acid 10%–hydroquinone 2% gel versus the combination plus kojic acid 2%. The results showed that 60% (24/40) of patients improved with the use of kojic acid as compared to those using the medication without kojic acid.12 Anecdotal data suggest kojic acid may be effective for PIH13; however, no studies specifically for PIH have been conducted.
Chemical Peels
Chemical peels have been used for a number of years, though their benefits in patients with skin of color is still being elucidated. The ideal chemical peels for Fitzpatrick skin types IV through VI are superficial to medium-depth peeling agents and techniques.14 Glycolic acid is a naturally occurring α-hydroxy acid that causes an increase in collagen synthesis, stimulates epidermolysis, and disperses basal layer melanin. Neutralization of glycolic acid peels can be done with the use of water, sodium bicarbonate, or sodium hydroxide to avoid unnecessary epidermal damage. Multiple clinical trials have been conducted to determine the response of glycolic acid peels in clearing PIH in patients with skin of color. Kessler et al15 compared glycolic acid 30% to salicylic acid 30% in 20 patients with mild to moderate acne and associated PIH. Chemical peels were performed every 2 weeks for 12 weeks. The study showed that salicylic acid was better tolerated than glycolic acid and both were equally effective after the second application (P<.05) for PIH.15 Finally, another study conducted for PIH in patients with Fitzpatrick skin types III and IV utilized glycolic acid peels with 20%, 35%, and 70% concentrations. The results showed overall improvement of PIH and acne from the use of all concentrations of glycolic peels, though faster efficacy was noted at higher concentrations.16
Other self-neutralizing peeling agents include salicylic acid and Jessner solution. Salicylic acid is a β-hydroxy acid that works through keratolysis and disrupting intercellular linkages. Jessner solution is a combination of resorcinol 14%, lactic acid 14%, and salicylic acid 14% in an alcohol base. Salicylic acid is well-tolerated in patients with Fitzpatrick skin types I through VI and has been helpful in treating acne, rosacea, melasma, hyperpigmentation, texturally rough skin, and mild photoaging. Jessner peeling solution has been used for a number of years and works as a keratolytic agent causing intercellular and intracellular edema, and due to its self-neutralizing agent, it is fairly superficial.17 Overall, superficial peeling agents should be used on patients with darker skin types to avoid the risk for worsening dyspigmentation, keloid formation, or deep scarring.18
Conclusion
These treatments are only some of the topical and chemical modalities for PIH in patients with skin of color. The patient history, evaluation, skin type, and underlying medical problems should be considered prior to using any topical or peeling agent. Lastly, photoprotection should be heavily emphasized with both sun protective gear and use of broad-spectrum sunscreens with a high sun protection factor, as UV radiation can cause darkening of PIH areas regardless of skin type and can reverse the progress made by a given therapy.18
- Savory SA, Agim NG, Mao R, et al. Reliability assessment and validation of the postacne hyperpigmentation index (PAHPI), a new instrument to measure postinflammatory hyperpigmentation from acne vulgaris. J Am Acad Dermatol. 2014;70:108-114.
- Halder RM. The role of retinoids in the management of cutaneous conditions in blacks. J Am Acad Dermatol. 1998;39(2, pt 3):S98-S103.
- Bulengo-Ransby SM, Griffiths CE, Kimbrough-Green CK, et al. Topical tretinoin (retinoid acid) therapy for hyperpigmented lesions caused by inflammation of the skin in black patients. N Engl J Med. 1993;328:1438-1443.
- Griffiths CE, Goldfarb MT, Finkel LJ, et al. Topical tretinoin (retinoic acid) treatment of hyperpigmented lesions associated with photoaging in Chinese and Japanese patients: a vehicle-controlled trial. J Am Acad Dermatol. 1994;30:76-84.
- Callendar VD. Acne in ethnic skin: special considerations for therapy. Dermatol Ther. 2004;17:184-195.
- Callender VD, Young CM, Kindred C, et al. Efficacy and safety of clindamycin phosphate 1.2% and tretinoin 0.025% gel for the treatment of acne and acne-induced post-inflammatory hyperpigmentation in patients with skin of color. J Clin Aesthet Dermatol. 2012;5:25-32.
- Badreshia-Bansal S, Draelos ZD. Insight into skin lightening cosmeceuticals for women of color. J Drugs Dermatol. 2007;6:32-39.
- Kligman AM, Willis I. A new formula for depigmenting human skin. Arch Dermatol. 1975;111:40-48.
- Grimes PE. A microsponge formulation of hydroquinone 4% and retinol 0.15% in the treatment of melasma and postinflammatory hyperpigmentation. Cutis. 2004;74:326-328.
- Chan R, Park KC, Lee MH, et al. A randomized controlled trial of the efficacy and safety of a fixed triple combination (fluocinolone acetonide 0.01%, hydroquinone 4%, tretinoin 0.05%) compared with hydroquinone 4% cream in Asian patients with moderate to severe melasma. Br J Dermatol. 2008;159:697-703.
- Lowe NJ, Rizk D, Grimes P. Azelaic acid 20% cream in the treatment of facial hyperpigmentation in darker-skinned patients. Clin Ther. 1998;20:945-959.
- Lim JT. Treatment of melasma using kojic acid in a gel containing hydroquinone and glycolic acid. Dermatol Surg. 1999;25:282-284.
- Alexis AF, Blackcloud P. Natural ingredients for darker skin types: growing options for hyperpigmentation. J Drugs Dermatol. 2013;12:123-127.
- Roberts WE. Chemical peeling in ethnic/dark skin. Dermatol Ther. 2004;17:196-205.
- Kessler E, Flanagan K, Chia C, et al. Comparison of alpha- and beta-hydroxy acid chemical peels in the treatment of mild to moderately severe facial acne vulgaris [published online December 5, 2007]. Dermatol Surg. 2008;34:45-50, discussion 51.
- Erbağci Z, Akçali C. Biweekly serial glycolic acid peels vs. long-term daily use of topical low-strength glycolic acid in the treatment of atrophic acne scars. Int J Dermatol. 2000;39:789-794.
- Jackson A. Chemical peels [published online January 31, 2014]. Facial Plast Surg. 2014;30:26-34.
- Davis EC, Callender VD. Postinflammatory hyperpigmentation: a review of the epidemiology, clinical features, and treatment options in skin of color. J Clin Aesthet Dermatol. 2010;3:20-31.
Postinflammatory hyperpigmentation (PIH) develops as darkly pigmented macules that occur after an inflammatory process of the skin such as acne, folliculitis, eczema, or shaving irritation. Patients with Fitzpatrick skin types III to VI usually are most commonly affected, and for many, the remnant pigmentation can be an even greater concern than the original inflammatory process.1,2 Reported treatments of PIH include tretinoin, hydroquinone, azelaic acid, and chemical peels. The ideal combination of therapy has yet to be delineated.
Tretinoin (Vitamin A Derivative)
Bulengo-Ransby et al3 performed one of the first clinical trials testing tretinoin cream 0.1% for PIH in patients with Fitzpatrick skin types IV to VI . The study included 54 patients (24 applied tretinoin and 30 applied a vehicle) with moderate to severe PIH on the face and arms. The patients were divided into therapy and placebo groups and were evaluated for 40 weeks. Changes were evaluated through colorimetry, light microscopy, histology, and photography, with significant clinical improvement in the tretinoin-treated group (P<.001).3 A double-blind, randomized study of 45 photoaged Chinese and Japanese patients using tretinoin cream 0.1% also was conducted for treatment of photoaging-associated hyperpigmented lesions of the face and hands. Assessment was done with clinical, colorimetric, and histological evaluation, with an overall statistical improvement noted in hyperpigmentation.4 Both of the above studies showed mild irritation (ie, retinoid dermatitis) with application of tretinoin, which creates a compliance issue in patients who are recommended to continue therapy with higher-strength tretinoin. This side-effect profile can be circumvented through gradual elevation in the strength of tretinoin.5
Combination Therapies
Combination therapies with tretinoin also have been used to improve PIH. Callender et al6 conducted a study evaluating the efficacy of clindamycin phosphate 1.2%–tretinoin 0.025% gel for the treatment of PIH secondary to mild to moderate acne in patients with Fitzpatrick skin types IV to VI. Thirty patients participated in the randomized, double-blinded, placebo-controlled study, with 15 patients in the clindamycin-tretinoin gel group and 15 in the placebo control group. Based on objective assessment using a chromameter and evaluator global acne severity scale score, clinical efficacy was demonstrated for treating acne and PIH as well as preventing further PIH.6
Hydroquinone Formulation (Tyrosine Inhibitor)
Hydroquinone bleaching cream has been the standard therapy for hyperpigmentation. It works by blocking the conversion of dihydroxyphenylalanine to melanin by inhibiting tyrosinase.7 Topical steroids directly inhibit the synthesis of melanin, and when combined with hydroquinone and tretinoin, they can be effective for short periods of time and may decrease the irritation of application.7,8 The most widely accepted formula consists of a topical steroid (triamcinolone cream 0.1%) in combination with hydroquinone 4% and tretinoin cream 0.05%.8 In a similar 12-week open-label study of 25 patients with darker skin types, Grimes9 used an alternative combination formula of hydroquinone 4% and retinol 0.15%. Overall improvement and tolerance was demonstrated through the use of colorimetry measurement. A combination of hydroquinone 4%, tretinoin 0.05%, and fluocinolone acetonide 0.01% also has been used effectively for the treatment of melasma.10 This formulation has been used more anecdotally for the treatment of PIH and has yet to have a randomized-controlled trial. The concern with repeated long-term use of hydroquinone remains. Permanent leukoderma, exogenous ochronosis, and hyperpigmentation of the surrounding normal skin (halo effect) can occur.
Azelaic Acid (Tyrosinase Inhibitor)
Azelaic acid is a dicarboxylic acid isolated from pityriasis versicolor that acts similar to a tyrosine inhibitor and has an antiproliferative effect toward abnormal melanocytes. Lowe et al11 conducted a randomized, double-blind, vehicle-controlled trial in patients with Fitzpatrick skin types IV through VI with facial hyperpigmentation using azelaic acid cream 20%. Over the course of 24 weeks, patients noted a decrease in overall pigment using both an investigator subjective scale and chromometer analysis.11
Kojic Acid (Tyrosinase Inhibitor)
Kojic acid is a tyrosinase inhibitor found in fungal metabolite species such as Acetobacter, Aspergillus, and Penicillium. It is commonly combined with other skin lightening agents such as hydroquinone or vitamin C to further enhance its efficacy. A randomized, 12-week, split-face study of Chinese women with melasma compared treatment with a glycolic acid 10%–hydroquinone 2% gel versus the combination plus kojic acid 2%. The results showed that 60% (24/40) of patients improved with the use of kojic acid as compared to those using the medication without kojic acid.12 Anecdotal data suggest kojic acid may be effective for PIH13; however, no studies specifically for PIH have been conducted.
Chemical Peels
Chemical peels have been used for a number of years, though their benefits in patients with skin of color is still being elucidated. The ideal chemical peels for Fitzpatrick skin types IV through VI are superficial to medium-depth peeling agents and techniques.14 Glycolic acid is a naturally occurring α-hydroxy acid that causes an increase in collagen synthesis, stimulates epidermolysis, and disperses basal layer melanin. Neutralization of glycolic acid peels can be done with the use of water, sodium bicarbonate, or sodium hydroxide to avoid unnecessary epidermal damage. Multiple clinical trials have been conducted to determine the response of glycolic acid peels in clearing PIH in patients with skin of color. Kessler et al15 compared glycolic acid 30% to salicylic acid 30% in 20 patients with mild to moderate acne and associated PIH. Chemical peels were performed every 2 weeks for 12 weeks. The study showed that salicylic acid was better tolerated than glycolic acid and both were equally effective after the second application (P<.05) for PIH.15 Finally, another study conducted for PIH in patients with Fitzpatrick skin types III and IV utilized glycolic acid peels with 20%, 35%, and 70% concentrations. The results showed overall improvement of PIH and acne from the use of all concentrations of glycolic peels, though faster efficacy was noted at higher concentrations.16
Other self-neutralizing peeling agents include salicylic acid and Jessner solution. Salicylic acid is a β-hydroxy acid that works through keratolysis and disrupting intercellular linkages. Jessner solution is a combination of resorcinol 14%, lactic acid 14%, and salicylic acid 14% in an alcohol base. Salicylic acid is well-tolerated in patients with Fitzpatrick skin types I through VI and has been helpful in treating acne, rosacea, melasma, hyperpigmentation, texturally rough skin, and mild photoaging. Jessner peeling solution has been used for a number of years and works as a keratolytic agent causing intercellular and intracellular edema, and due to its self-neutralizing agent, it is fairly superficial.17 Overall, superficial peeling agents should be used on patients with darker skin types to avoid the risk for worsening dyspigmentation, keloid formation, or deep scarring.18
Conclusion
These treatments are only some of the topical and chemical modalities for PIH in patients with skin of color. The patient history, evaluation, skin type, and underlying medical problems should be considered prior to using any topical or peeling agent. Lastly, photoprotection should be heavily emphasized with both sun protective gear and use of broad-spectrum sunscreens with a high sun protection factor, as UV radiation can cause darkening of PIH areas regardless of skin type and can reverse the progress made by a given therapy.18
Postinflammatory hyperpigmentation (PIH) develops as darkly pigmented macules that occur after an inflammatory process of the skin such as acne, folliculitis, eczema, or shaving irritation. Patients with Fitzpatrick skin types III to VI usually are most commonly affected, and for many, the remnant pigmentation can be an even greater concern than the original inflammatory process.1,2 Reported treatments of PIH include tretinoin, hydroquinone, azelaic acid, and chemical peels. The ideal combination of therapy has yet to be delineated.
Tretinoin (Vitamin A Derivative)
Bulengo-Ransby et al3 performed one of the first clinical trials testing tretinoin cream 0.1% for PIH in patients with Fitzpatrick skin types IV to VI . The study included 54 patients (24 applied tretinoin and 30 applied a vehicle) with moderate to severe PIH on the face and arms. The patients were divided into therapy and placebo groups and were evaluated for 40 weeks. Changes were evaluated through colorimetry, light microscopy, histology, and photography, with significant clinical improvement in the tretinoin-treated group (P<.001).3 A double-blind, randomized study of 45 photoaged Chinese and Japanese patients using tretinoin cream 0.1% also was conducted for treatment of photoaging-associated hyperpigmented lesions of the face and hands. Assessment was done with clinical, colorimetric, and histological evaluation, with an overall statistical improvement noted in hyperpigmentation.4 Both of the above studies showed mild irritation (ie, retinoid dermatitis) with application of tretinoin, which creates a compliance issue in patients who are recommended to continue therapy with higher-strength tretinoin. This side-effect profile can be circumvented through gradual elevation in the strength of tretinoin.5
Combination Therapies
Combination therapies with tretinoin also have been used to improve PIH. Callender et al6 conducted a study evaluating the efficacy of clindamycin phosphate 1.2%–tretinoin 0.025% gel for the treatment of PIH secondary to mild to moderate acne in patients with Fitzpatrick skin types IV to VI. Thirty patients participated in the randomized, double-blinded, placebo-controlled study, with 15 patients in the clindamycin-tretinoin gel group and 15 in the placebo control group. Based on objective assessment using a chromameter and evaluator global acne severity scale score, clinical efficacy was demonstrated for treating acne and PIH as well as preventing further PIH.6
Hydroquinone Formulation (Tyrosine Inhibitor)
Hydroquinone bleaching cream has been the standard therapy for hyperpigmentation. It works by blocking the conversion of dihydroxyphenylalanine to melanin by inhibiting tyrosinase.7 Topical steroids directly inhibit the synthesis of melanin, and when combined with hydroquinone and tretinoin, they can be effective for short periods of time and may decrease the irritation of application.7,8 The most widely accepted formula consists of a topical steroid (triamcinolone cream 0.1%) in combination with hydroquinone 4% and tretinoin cream 0.05%.8 In a similar 12-week open-label study of 25 patients with darker skin types, Grimes9 used an alternative combination formula of hydroquinone 4% and retinol 0.15%. Overall improvement and tolerance was demonstrated through the use of colorimetry measurement. A combination of hydroquinone 4%, tretinoin 0.05%, and fluocinolone acetonide 0.01% also has been used effectively for the treatment of melasma.10 This formulation has been used more anecdotally for the treatment of PIH and has yet to have a randomized-controlled trial. The concern with repeated long-term use of hydroquinone remains. Permanent leukoderma, exogenous ochronosis, and hyperpigmentation of the surrounding normal skin (halo effect) can occur.
Azelaic Acid (Tyrosinase Inhibitor)
Azelaic acid is a dicarboxylic acid isolated from pityriasis versicolor that acts similar to a tyrosine inhibitor and has an antiproliferative effect toward abnormal melanocytes. Lowe et al11 conducted a randomized, double-blind, vehicle-controlled trial in patients with Fitzpatrick skin types IV through VI with facial hyperpigmentation using azelaic acid cream 20%. Over the course of 24 weeks, patients noted a decrease in overall pigment using both an investigator subjective scale and chromometer analysis.11
Kojic Acid (Tyrosinase Inhibitor)
Kojic acid is a tyrosinase inhibitor found in fungal metabolite species such as Acetobacter, Aspergillus, and Penicillium. It is commonly combined with other skin lightening agents such as hydroquinone or vitamin C to further enhance its efficacy. A randomized, 12-week, split-face study of Chinese women with melasma compared treatment with a glycolic acid 10%–hydroquinone 2% gel versus the combination plus kojic acid 2%. The results showed that 60% (24/40) of patients improved with the use of kojic acid as compared to those using the medication without kojic acid.12 Anecdotal data suggest kojic acid may be effective for PIH13; however, no studies specifically for PIH have been conducted.
Chemical Peels
Chemical peels have been used for a number of years, though their benefits in patients with skin of color is still being elucidated. The ideal chemical peels for Fitzpatrick skin types IV through VI are superficial to medium-depth peeling agents and techniques.14 Glycolic acid is a naturally occurring α-hydroxy acid that causes an increase in collagen synthesis, stimulates epidermolysis, and disperses basal layer melanin. Neutralization of glycolic acid peels can be done with the use of water, sodium bicarbonate, or sodium hydroxide to avoid unnecessary epidermal damage. Multiple clinical trials have been conducted to determine the response of glycolic acid peels in clearing PIH in patients with skin of color. Kessler et al15 compared glycolic acid 30% to salicylic acid 30% in 20 patients with mild to moderate acne and associated PIH. Chemical peels were performed every 2 weeks for 12 weeks. The study showed that salicylic acid was better tolerated than glycolic acid and both were equally effective after the second application (P<.05) for PIH.15 Finally, another study conducted for PIH in patients with Fitzpatrick skin types III and IV utilized glycolic acid peels with 20%, 35%, and 70% concentrations. The results showed overall improvement of PIH and acne from the use of all concentrations of glycolic peels, though faster efficacy was noted at higher concentrations.16
Other self-neutralizing peeling agents include salicylic acid and Jessner solution. Salicylic acid is a β-hydroxy acid that works through keratolysis and disrupting intercellular linkages. Jessner solution is a combination of resorcinol 14%, lactic acid 14%, and salicylic acid 14% in an alcohol base. Salicylic acid is well-tolerated in patients with Fitzpatrick skin types I through VI and has been helpful in treating acne, rosacea, melasma, hyperpigmentation, texturally rough skin, and mild photoaging. Jessner peeling solution has been used for a number of years and works as a keratolytic agent causing intercellular and intracellular edema, and due to its self-neutralizing agent, it is fairly superficial.17 Overall, superficial peeling agents should be used on patients with darker skin types to avoid the risk for worsening dyspigmentation, keloid formation, or deep scarring.18
Conclusion
These treatments are only some of the topical and chemical modalities for PIH in patients with skin of color. The patient history, evaluation, skin type, and underlying medical problems should be considered prior to using any topical or peeling agent. Lastly, photoprotection should be heavily emphasized with both sun protective gear and use of broad-spectrum sunscreens with a high sun protection factor, as UV radiation can cause darkening of PIH areas regardless of skin type and can reverse the progress made by a given therapy.18
- Savory SA, Agim NG, Mao R, et al. Reliability assessment and validation of the postacne hyperpigmentation index (PAHPI), a new instrument to measure postinflammatory hyperpigmentation from acne vulgaris. J Am Acad Dermatol. 2014;70:108-114.
- Halder RM. The role of retinoids in the management of cutaneous conditions in blacks. J Am Acad Dermatol. 1998;39(2, pt 3):S98-S103.
- Bulengo-Ransby SM, Griffiths CE, Kimbrough-Green CK, et al. Topical tretinoin (retinoid acid) therapy for hyperpigmented lesions caused by inflammation of the skin in black patients. N Engl J Med. 1993;328:1438-1443.
- Griffiths CE, Goldfarb MT, Finkel LJ, et al. Topical tretinoin (retinoic acid) treatment of hyperpigmented lesions associated with photoaging in Chinese and Japanese patients: a vehicle-controlled trial. J Am Acad Dermatol. 1994;30:76-84.
- Callendar VD. Acne in ethnic skin: special considerations for therapy. Dermatol Ther. 2004;17:184-195.
- Callender VD, Young CM, Kindred C, et al. Efficacy and safety of clindamycin phosphate 1.2% and tretinoin 0.025% gel for the treatment of acne and acne-induced post-inflammatory hyperpigmentation in patients with skin of color. J Clin Aesthet Dermatol. 2012;5:25-32.
- Badreshia-Bansal S, Draelos ZD. Insight into skin lightening cosmeceuticals for women of color. J Drugs Dermatol. 2007;6:32-39.
- Kligman AM, Willis I. A new formula for depigmenting human skin. Arch Dermatol. 1975;111:40-48.
- Grimes PE. A microsponge formulation of hydroquinone 4% and retinol 0.15% in the treatment of melasma and postinflammatory hyperpigmentation. Cutis. 2004;74:326-328.
- Chan R, Park KC, Lee MH, et al. A randomized controlled trial of the efficacy and safety of a fixed triple combination (fluocinolone acetonide 0.01%, hydroquinone 4%, tretinoin 0.05%) compared with hydroquinone 4% cream in Asian patients with moderate to severe melasma. Br J Dermatol. 2008;159:697-703.
- Lowe NJ, Rizk D, Grimes P. Azelaic acid 20% cream in the treatment of facial hyperpigmentation in darker-skinned patients. Clin Ther. 1998;20:945-959.
- Lim JT. Treatment of melasma using kojic acid in a gel containing hydroquinone and glycolic acid. Dermatol Surg. 1999;25:282-284.
- Alexis AF, Blackcloud P. Natural ingredients for darker skin types: growing options for hyperpigmentation. J Drugs Dermatol. 2013;12:123-127.
- Roberts WE. Chemical peeling in ethnic/dark skin. Dermatol Ther. 2004;17:196-205.
- Kessler E, Flanagan K, Chia C, et al. Comparison of alpha- and beta-hydroxy acid chemical peels in the treatment of mild to moderately severe facial acne vulgaris [published online December 5, 2007]. Dermatol Surg. 2008;34:45-50, discussion 51.
- Erbağci Z, Akçali C. Biweekly serial glycolic acid peels vs. long-term daily use of topical low-strength glycolic acid in the treatment of atrophic acne scars. Int J Dermatol. 2000;39:789-794.
- Jackson A. Chemical peels [published online January 31, 2014]. Facial Plast Surg. 2014;30:26-34.
- Davis EC, Callender VD. Postinflammatory hyperpigmentation: a review of the epidemiology, clinical features, and treatment options in skin of color. J Clin Aesthet Dermatol. 2010;3:20-31.
- Savory SA, Agim NG, Mao R, et al. Reliability assessment and validation of the postacne hyperpigmentation index (PAHPI), a new instrument to measure postinflammatory hyperpigmentation from acne vulgaris. J Am Acad Dermatol. 2014;70:108-114.
- Halder RM. The role of retinoids in the management of cutaneous conditions in blacks. J Am Acad Dermatol. 1998;39(2, pt 3):S98-S103.
- Bulengo-Ransby SM, Griffiths CE, Kimbrough-Green CK, et al. Topical tretinoin (retinoid acid) therapy for hyperpigmented lesions caused by inflammation of the skin in black patients. N Engl J Med. 1993;328:1438-1443.
- Griffiths CE, Goldfarb MT, Finkel LJ, et al. Topical tretinoin (retinoic acid) treatment of hyperpigmented lesions associated with photoaging in Chinese and Japanese patients: a vehicle-controlled trial. J Am Acad Dermatol. 1994;30:76-84.
- Callendar VD. Acne in ethnic skin: special considerations for therapy. Dermatol Ther. 2004;17:184-195.
- Callender VD, Young CM, Kindred C, et al. Efficacy and safety of clindamycin phosphate 1.2% and tretinoin 0.025% gel for the treatment of acne and acne-induced post-inflammatory hyperpigmentation in patients with skin of color. J Clin Aesthet Dermatol. 2012;5:25-32.
- Badreshia-Bansal S, Draelos ZD. Insight into skin lightening cosmeceuticals for women of color. J Drugs Dermatol. 2007;6:32-39.
- Kligman AM, Willis I. A new formula for depigmenting human skin. Arch Dermatol. 1975;111:40-48.
- Grimes PE. A microsponge formulation of hydroquinone 4% and retinol 0.15% in the treatment of melasma and postinflammatory hyperpigmentation. Cutis. 2004;74:326-328.
- Chan R, Park KC, Lee MH, et al. A randomized controlled trial of the efficacy and safety of a fixed triple combination (fluocinolone acetonide 0.01%, hydroquinone 4%, tretinoin 0.05%) compared with hydroquinone 4% cream in Asian patients with moderate to severe melasma. Br J Dermatol. 2008;159:697-703.
- Lowe NJ, Rizk D, Grimes P. Azelaic acid 20% cream in the treatment of facial hyperpigmentation in darker-skinned patients. Clin Ther. 1998;20:945-959.
- Lim JT. Treatment of melasma using kojic acid in a gel containing hydroquinone and glycolic acid. Dermatol Surg. 1999;25:282-284.
- Alexis AF, Blackcloud P. Natural ingredients for darker skin types: growing options for hyperpigmentation. J Drugs Dermatol. 2013;12:123-127.
- Roberts WE. Chemical peeling in ethnic/dark skin. Dermatol Ther. 2004;17:196-205.
- Kessler E, Flanagan K, Chia C, et al. Comparison of alpha- and beta-hydroxy acid chemical peels in the treatment of mild to moderately severe facial acne vulgaris [published online December 5, 2007]. Dermatol Surg. 2008;34:45-50, discussion 51.
- Erbağci Z, Akçali C. Biweekly serial glycolic acid peels vs. long-term daily use of topical low-strength glycolic acid in the treatment of atrophic acne scars. Int J Dermatol. 2000;39:789-794.
- Jackson A. Chemical peels [published online January 31, 2014]. Facial Plast Surg. 2014;30:26-34.
- Davis EC, Callender VD. Postinflammatory hyperpigmentation: a review of the epidemiology, clinical features, and treatment options in skin of color. J Clin Aesthet Dermatol. 2010;3:20-31.
The view from my office: How psychiatry residency programs have changed
As I approach my twentieth year as Residency Program Coordinator in the Department of Psychiatry at Saint Louis University School of Medicine, I’ve been reflecting on the many changes that have occurred: within our residency program; in the requirements that all residency programs must meet to continue as an Accreditation Council for Graduate Medical Education (ACGME)-accredited program; and in the overall scope of psychiatry residency training.
What has changed
During my time as Residency Program Coordinator, I have assisted 5 program directors and 3 associate program directors with day-to-day details of residency training. Our residency program has had couples, and a father and son; some residents even married each other while still in training.
The Electronic Residency Application System was not available until 2001; before that, applicants interested in being invited for an interview with a psychiatry residency program had to mail in their applications for review. This was a time-consuming, tedious process. In addition, residency programs today are required to use the American Board of Psychiatry and Neurology (ABPN) PreCERT credentialing program to verify training—instead of (as in the past) simply submitting a letter to ABPN that detailed the rotations and clinical skills examinations completed.
Residency programs have gone from evaluating residents by using the 6 competencies to the Milestones requirement from ACGME, which is the newest system of measuring residents’ competencies. Every month, the program faculty meets to discuss the progress of 1 of the classes of residents and the residents who are completing an individual self-assessment. Milestone scores for each resident are then reported to ACGME.
At one time, a resident’s files could be stored in a 2-inch binder; now, we need a 4-inch binder to accommodate required documentation! I am relieved—as, I am sure, many other residency program coordinators are—that residency programs are no longer required to prepare a Program Information Form but, instead, perform a self-study and, every 10 years, have a site visit. Last, every academic year, the Residency Program Coordinator is required to enter the incoming residents’ information into the graduate medical education track, ACGME, and PreCERT Web site systems.
Rewards of my position
As Residency Program Coordinator, I’ve had the rewarding experience of meeting physicians from all over the world without having to travel to other countries. Because I have a 3- or 4-year relationship with residents, I serve them in various roles: mentor, mother, confidante, motivator, and friend. As much as the job is rewarding, being the Residency Program Coordinator can, on some days, be overwhelming, particularly because I need to think “out of the box” to streamline decisions and thus avoid conflicts with program rotations and didactic schedules.
As I approach my twentieth year as Residency Program Coordinator in the Department of Psychiatry at Saint Louis University School of Medicine, I’ve been reflecting on the many changes that have occurred: within our residency program; in the requirements that all residency programs must meet to continue as an Accreditation Council for Graduate Medical Education (ACGME)-accredited program; and in the overall scope of psychiatry residency training.
What has changed
During my time as Residency Program Coordinator, I have assisted 5 program directors and 3 associate program directors with day-to-day details of residency training. Our residency program has had couples, and a father and son; some residents even married each other while still in training.
The Electronic Residency Application System was not available until 2001; before that, applicants interested in being invited for an interview with a psychiatry residency program had to mail in their applications for review. This was a time-consuming, tedious process. In addition, residency programs today are required to use the American Board of Psychiatry and Neurology (ABPN) PreCERT credentialing program to verify training—instead of (as in the past) simply submitting a letter to ABPN that detailed the rotations and clinical skills examinations completed.
Residency programs have gone from evaluating residents by using the 6 competencies to the Milestones requirement from ACGME, which is the newest system of measuring residents’ competencies. Every month, the program faculty meets to discuss the progress of 1 of the classes of residents and the residents who are completing an individual self-assessment. Milestone scores for each resident are then reported to ACGME.
At one time, a resident’s files could be stored in a 2-inch binder; now, we need a 4-inch binder to accommodate required documentation! I am relieved—as, I am sure, many other residency program coordinators are—that residency programs are no longer required to prepare a Program Information Form but, instead, perform a self-study and, every 10 years, have a site visit. Last, every academic year, the Residency Program Coordinator is required to enter the incoming residents’ information into the graduate medical education track, ACGME, and PreCERT Web site systems.
Rewards of my position
As Residency Program Coordinator, I’ve had the rewarding experience of meeting physicians from all over the world without having to travel to other countries. Because I have a 3- or 4-year relationship with residents, I serve them in various roles: mentor, mother, confidante, motivator, and friend. As much as the job is rewarding, being the Residency Program Coordinator can, on some days, be overwhelming, particularly because I need to think “out of the box” to streamline decisions and thus avoid conflicts with program rotations and didactic schedules.
As I approach my twentieth year as Residency Program Coordinator in the Department of Psychiatry at Saint Louis University School of Medicine, I’ve been reflecting on the many changes that have occurred: within our residency program; in the requirements that all residency programs must meet to continue as an Accreditation Council for Graduate Medical Education (ACGME)-accredited program; and in the overall scope of psychiatry residency training.
What has changed
During my time as Residency Program Coordinator, I have assisted 5 program directors and 3 associate program directors with day-to-day details of residency training. Our residency program has had couples, and a father and son; some residents even married each other while still in training.
The Electronic Residency Application System was not available until 2001; before that, applicants interested in being invited for an interview with a psychiatry residency program had to mail in their applications for review. This was a time-consuming, tedious process. In addition, residency programs today are required to use the American Board of Psychiatry and Neurology (ABPN) PreCERT credentialing program to verify training—instead of (as in the past) simply submitting a letter to ABPN that detailed the rotations and clinical skills examinations completed.
Residency programs have gone from evaluating residents by using the 6 competencies to the Milestones requirement from ACGME, which is the newest system of measuring residents’ competencies. Every month, the program faculty meets to discuss the progress of 1 of the classes of residents and the residents who are completing an individual self-assessment. Milestone scores for each resident are then reported to ACGME.
At one time, a resident’s files could be stored in a 2-inch binder; now, we need a 4-inch binder to accommodate required documentation! I am relieved—as, I am sure, many other residency program coordinators are—that residency programs are no longer required to prepare a Program Information Form but, instead, perform a self-study and, every 10 years, have a site visit. Last, every academic year, the Residency Program Coordinator is required to enter the incoming residents’ information into the graduate medical education track, ACGME, and PreCERT Web site systems.
Rewards of my position
As Residency Program Coordinator, I’ve had the rewarding experience of meeting physicians from all over the world without having to travel to other countries. Because I have a 3- or 4-year relationship with residents, I serve them in various roles: mentor, mother, confidante, motivator, and friend. As much as the job is rewarding, being the Residency Program Coordinator can, on some days, be overwhelming, particularly because I need to think “out of the box” to streamline decisions and thus avoid conflicts with program rotations and didactic schedules.
Reconstructive Shelf Arthroplasty as a Salvage Procedure for Complex Fifth Tarsometatarsal Joint Complex Injuries: A Case Review and Discussion
Fractures of the cuboid bone are uncommon, with an annual incidence of approximately 1.8 per 100,000.1 This is largely attributed to the inherent stability provided by its anatomy and position in the foot’s lateral column, where it functions as a link between the lateral column and transverse plantar arch.2 Regarding its anatomy, the cuboid is a pyramidal-shaped bone with 6 bony surfaces that provide tremendous stability—3 of these are articular, 3 nonarticular.
Although the cuboid bone is susceptible to low-energy avulsion injuries, injuries that occur in the setting of high-energy trauma are most concerning, as they often occur concurrently with other midfoot fractures and dislocations. These less common crush injuries are associated with comminution, articular disruption, and shortening of the lateral column.3-5 Avulsion injuries occur via a twisting mechanism, while the more complex nutcracker fracture evolves via longitudinal compression of the lateral column, with the foot in a position of forced plantarflexion.6 Other comminuted fractures occur from direct impact on the lateral aspect of the foot.
Management of cuboid fractures varies according to etiology, fracture displacement, and articular involvement. Conservative management is reserved solely for stable, nondisplaced fractures.7 Unstable fracture-dislocations and those with associated lateral column shortening necessitate operative treatment, which attempts to restore anatomy, stability, and length of the foot’s lateral column.7-9 However, with the exception of open injuries, fractures tenting the skin, and injuries with concomitant compartment syndrome, the high-energy nature of cuboid fractures often precludes early surgical intervention, as the foot’s soft-tissue envelope is too compromised. For this reason, operative intervention is often performed on a delayed basis only after recovery of the soft tissue.
In this case report and literature review, we describe a reconstructive shelf arthroplasty of the fifth tarsometatarsal (TMT) joint as a primary intervention for crush-type cuboid fractures with associated joint subsidence and lateral column shortening. The shelf arthroplasty, which was first credited to Konig in 1891, has historically been described as a remodeling operation using bone graft wedges for the treatment of nonconcentric acetabular dysplasia.10 Although bone grafting is recognized as an effective means of addressing osseous voids in the setting of comminuted cuboid fractures, its specific application in the form of a shelf arthroplasty has not been described.11 The patient provided written informed consent for print and electronic publication of this case report.
Case Report
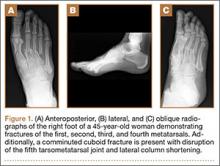
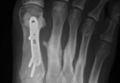
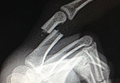
An otherwise healthy 45-year-old woman presented to our institution’s emergency department (ED) complaining of right foot pain after a motor vehicle accident. She was the restrained driver in a head-on collision. Primary survey revealed a swollen, ecchymotic, and tender right foot. Radiographs demonstrated fractures of her first, second, third, and fourth metatarsals, and a comminuted cuboid fracture with lateral column shortening and disruption of the fifth TMT joint (Figure 1).
Due to swelling, initial management consisted of soft-tissue management through the use of a well-padded splint. As this was her only injury, she was instructed to remain non-weight-bearing, ambulate with crutches, and return to our outpatient office for close follow-up. The need for delayed surgical intervention of her multiple foot injuries, due to her compromised soft-tissue envelope, was discussed prior to discharge.
Surgical intervention was performed 15 days after the injury, when the soft-tissue swelling had dissipated. The surgical plan included fixation of the multiple metatarsal fractures and lateral column reconstruction and stabilization. With regard to the lateral column, we obtained patient consent for several possible procedures, including fifth TMT joint closed reduction and percutaneous pinning, open reduction and internal fixation (ORIF), and TMT joint reconstruction with iliac crest bone graft (ICBG).
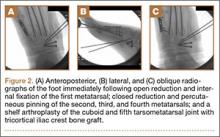
The metatarsals were addressed first via a dorsomedial incision, using a 5-hole 2.7-mm Limited Contact Dynamic Compression Plate (Synthes) to stabilize the first metatarsal and 2.0-mm Kirschner wires (K-wires) to maintain the length and alignment of the second, third, and fourth metatarsals (Figure 2). Closed reduction and percutaneous pinning of the fifth metatarsal was then attempted but abandoned because of persistent instability and subsidence of the cuboid in the proximal and plantar direction. ORIF was then attempted through a dorsolateral incision extending from just distal to the sinus tarsi to the base of the fourth metatarsal. However, the lateral cuboid was too comminuted to accommodate any fixation and prevent fifth TMT joint subluxation and lateral column shortening.
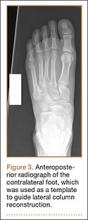
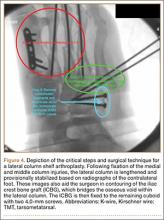
Autograft reconstruction of the lateral column was therefore performed, using radiographs of the patient’s uninjured, contralateral foot as a template for our lateral column shelf arthroplasty (Figure 3). Based on this template, the length and alignment of the lateral column were provisionally maintained with two 2.0-mm K-wires placed between the fifth metatarsal and intact cuboid (Figure 4). Tricortical ICBG was then harvested through an anterior approach to the iliac crest and contoured accordingly to fill the osseous void. To facilitate graft incorporation, comminuted fragments of cuboid bone were removed, with the remaining bone decorticated. The graft was then fixed to the remaining cuboid with two 4.0-mm partially threaded cannulated screws (Synthes; Figures 2, 4). This construct restored the length of the lateral column and effectively buttressed the fifth TMT joint, preventing subsidence and dislocation of the TMT joint.
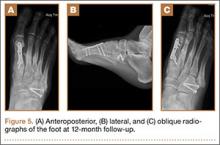
After a 2-day postoperative course in the hospital, the patient was discharged. She remained non-weight-bearing in a splint with Robert Jones cotton bandage. At her 2-week postoperative visit, all hardware was intact and there was no evidence of infection. Her sutures were removed and she was placed in a new splint. At the patient’s 5-week postoperative visit, all K-wires were removed. At this time she remained non-weight-bearing but was transitioned into a controlled ankle movement (CAM) boot and was allowed to begin active and passive ankle exercises. At her 10-week follow-up, radiographs revealed appropriate interval healing and callus formation. The patient began weight-bearing as tolerated in the CAM boot at that time. At 12 weeks, she was transitioned into a hard-soled shoe for comfort and was allowed to ambulate in the footwear of her choice as tolerated. Her activity levels were slowly advanced, and, at her 12-month follow-up, the patient had returned to playing tennis in her recreational league with no residual sequelae (Figure 5).
Discussion
Although rare, cuboid fractures are critical to identify and can result in significant disability, as they are frequently associated with additional foot trauma, as demonstrated in this case.1-4When isolated cuboid fractures are present, further imaging must be performed, including additional radiographic views and computed tomography, to search for other injuries, such as TMT joint complex disruption.
Only those cuboid fractures that are low-energy, stable, or nondisplaced can be effectively managed conservatively.12In the presence of instability, articular incongruity, or lateral column shortening, operative intervention is warranted. Arthritic degeneration, pain, and deformity result from residual incongruity at the calcaneocuboid or TMT joints, or when lateral column length is not restored.4-6,13 The latter leads to forefoot abduction and lateral subluxation of the lesser metatarsals, with ensuing posttraumatic pes planus or planovalgus deformity, which often necessitates secondary reconstructive procedures or arthrodesis.14,15 Stable reduction and restoration of lateral column length can be challenging, particularly in the setting of comminution and bone loss. Common methods of treatment involve lifting the dorsolateral cortex of the cuboid and buttressing the impacted articular surface with bone graft or bone graft substitutes. Fixation can be achieved with K-wires, small fragment plates and screws, and distraction external fixation.11 The latter is a particularly beneficial technique, as it can be used independent of or in conjunction with ORIF.
In a study by Weber and Locher,11 the short-term to midterm results of cuboid ORIF were assessed in 12 patients. Results were found to be good with respect to restoration of length, joint reconstruction, and overall return to function.11 Admittedly, these authors at times employed a similar but conceptually different approach to our patient. In their 7 patients with severe comminution and lateral column shortening, corticocancellous ICBG was used. However, Weber and Locher11did not describe this as a shelf arthroplasty, but instead as an adjunct to primary ORIF.
In our case, the tricortical ICBG shelf arthroplasty was used as it is in the hip, as a salvage procedure. Although little is known about outcomes following shelf arthroplasty for lateral column reconstruction in the foot, a 50% failure rate has been observed in the hip.16 As such, our preference was to perform an anatomic ORIF of the cuboid and lateral column, with the shelf arthroplasty only indicated if we were unable to achieve this. We believe that the need for tricortical ICBG in the treatment of cuboid fractures is indicative of a more severe injury and that it is a less optimal and more technically demanding intervention compared with primary ORIF. Furthermore, in other studies devoted to the treatment of cuboid fractures, patients requiring reconstruction with structural graft are not included in primary ORIF cohorts.17
As in the hip, suboptimal outcomes may occur when shelf arthroplasty is performed in the foot. There are additional considerations unique to the foot that surgeons must also contemplate when considering shelf arthroplasty. As demonstrated in the literature for adult-acquired flatfoot deformity, lateral column reconstruction is challenging and controversial and is associated with overload, pain, and the need to remove prominent hardware.18 These complications may also occur after shelf arthroplasty for cuboid fractures.
The work by Weber and Locher11 did not elucidate such considerations, and outcomes of ORIF and ICBG reconstruction were not compared. This is a limitation of their study, as differences in functional outcomes between the 2 procedures remain unknown. Given the degree of comminution that precludes ORIF and necessitates a graft reconstruction, we believe that the description of the shelf arthroplasty as a salvage procedure more accurately reflects the severity of injury. This may have implications regarding outcomes and patient expectations that the orthopedic surgeon must address. Future studies must further evaluate the outcomes of this technique, independent of and in comparison with ORIF.
Conclusion
In this case, we describe shelf arthroplasty for cuboid fractures. It is a reconstructive salvage procedure that is indicated when ORIF cannot be achieved. This useful approach to a complex injury must remain in the armamentarium of orthopedic surgeons. As we have demonstrated, it can effectively restore a damaged lateral column, providing length and, in our case, enabling the patient to return to her pre-injury level of activity.
1. Court-Brown C, Zinna S, Ekrol I. Classification and epidemiology of midfoot fractures. Foot. 2006;16(3):138-141.
2. Sarrafian SK. Osteology. In: Kelikian AS, ed. Sarrafian’s Anatomy of the Foot and Ankle. Philadelphia, PA: Lippincott; 1993:65-70.
3. Davis CA, Lubowitz J, Thordarson DB. Midtarsal fracture subluxation. Case report and review of the literature. Clin Orthop Relat Res. 1993;(292):264-268.
4. Dewar FP, Evans DC. Occult fracture-subluxation of the midtarsal joint. J Bone Joint Surg Br. 1968;50(2):386-388.
5. Sangeorzan BJ, Swiontkowski MF. Displaced fractures of the cuboid. J Bone Joint Surg Br. 1990;72(3):376-378.
6. Hermel MB, Gershon-Cohen J. The nutcracker fracture of the cuboid by indirect violence. Radiology. 1953;60(6):850-854.
7. Early J, Reid J. Fractures and dislocations of the midfoot and forefoot. In: Heckman JD, Bucholz RW, Court-Brown CM, Tornetta P, eds. Rockwood and Green’s Fractures in Adults. 7th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2009:2120-2126.
8. Richter M, Wippermann B, Krettek C, Schratt HE, Hufner T, Therman H. Fractures and fracture dislocations of the midfoot: occurrence, causes and long-term results. Foot Ankle Int. 2001;22(5):392-398.
9. Borrelli J Jr, De S, VanPelt M. Fracture of the cuboid. J Am Acad Orthop Surg. 2012;20(7):472-477.
10. Love BRT, Stevens PM, Williams PF. A long-term review of shelf arthroplasty. J Bone Joint Surg Br. 1980;62(3):321-325.
11. Weber M, Locher S. Reconstruction of the cuboid in compression fractures: short to midterm results in 12 patients. Foot Ankle Int. 2002;23(11):1008-1013.
12. Ebizie AO. Crush fractures of the cuboid from indirect violence. Injury. 1991;22(5):414-416.
13. Berlet GC, Hodges Davis W, Anderson RB. Tendon arthroplasty for basal fourth and fifth metatarsal arthritis. Foot Ankle Int. 2002;23(5):440-444.
14. Brunet JA, Wiley JJ. The late results of tarsometatarsal joint injuries. J Bone Joint Surg Br. 1987;69(3):437-440.
15. DeAsla R, Deland J. Anatomy and biomechanics of the foot and ankle. In: Thordarson DB, Tornetta P, Einhorn TA, eds. Orthopaedic Surgery Essentials: Foot & Ankle. Philadelphia, PA: Lippincott William & Wilkins; 2004:18-23.
16. Berton C, Bocquet D, Krantz N, Cotton A, Migaud H, Girard J. Shelf arthroplasties long-term outcome: influence of labral tears. A prospective study at a minimal 16 years’ follows up. Orthop Traumatol Surg Res. 2010;96(7):753-759.
17. van Raaij TM, Duffy PJ, Buckley RE. Displaced isolated cuboid fractures: results of four cases with operative treatment. Foot Ankle Int. 2010;31(3):242-246.
18. Grier KM, Walling AK. The use of tricortical autograft versus allograft in lateral column lengthening for adult acquired flatfoot deformity: an analysis of union rates and complications. Foot Ankle Int. 2010;31(9):760-769.
Fractures of the cuboid bone are uncommon, with an annual incidence of approximately 1.8 per 100,000.1 This is largely attributed to the inherent stability provided by its anatomy and position in the foot’s lateral column, where it functions as a link between the lateral column and transverse plantar arch.2 Regarding its anatomy, the cuboid is a pyramidal-shaped bone with 6 bony surfaces that provide tremendous stability—3 of these are articular, 3 nonarticular.
Although the cuboid bone is susceptible to low-energy avulsion injuries, injuries that occur in the setting of high-energy trauma are most concerning, as they often occur concurrently with other midfoot fractures and dislocations. These less common crush injuries are associated with comminution, articular disruption, and shortening of the lateral column.3-5 Avulsion injuries occur via a twisting mechanism, while the more complex nutcracker fracture evolves via longitudinal compression of the lateral column, with the foot in a position of forced plantarflexion.6 Other comminuted fractures occur from direct impact on the lateral aspect of the foot.
Management of cuboid fractures varies according to etiology, fracture displacement, and articular involvement. Conservative management is reserved solely for stable, nondisplaced fractures.7 Unstable fracture-dislocations and those with associated lateral column shortening necessitate operative treatment, which attempts to restore anatomy, stability, and length of the foot’s lateral column.7-9 However, with the exception of open injuries, fractures tenting the skin, and injuries with concomitant compartment syndrome, the high-energy nature of cuboid fractures often precludes early surgical intervention, as the foot’s soft-tissue envelope is too compromised. For this reason, operative intervention is often performed on a delayed basis only after recovery of the soft tissue.
In this case report and literature review, we describe a reconstructive shelf arthroplasty of the fifth tarsometatarsal (TMT) joint as a primary intervention for crush-type cuboid fractures with associated joint subsidence and lateral column shortening. The shelf arthroplasty, which was first credited to Konig in 1891, has historically been described as a remodeling operation using bone graft wedges for the treatment of nonconcentric acetabular dysplasia.10 Although bone grafting is recognized as an effective means of addressing osseous voids in the setting of comminuted cuboid fractures, its specific application in the form of a shelf arthroplasty has not been described.11 The patient provided written informed consent for print and electronic publication of this case report.
Case Report
An otherwise healthy 45-year-old woman presented to our institution’s emergency department (ED) complaining of right foot pain after a motor vehicle accident. She was the restrained driver in a head-on collision. Primary survey revealed a swollen, ecchymotic, and tender right foot. Radiographs demonstrated fractures of her first, second, third, and fourth metatarsals, and a comminuted cuboid fracture with lateral column shortening and disruption of the fifth TMT joint (Figure 1).
Due to swelling, initial management consisted of soft-tissue management through the use of a well-padded splint. As this was her only injury, she was instructed to remain non-weight-bearing, ambulate with crutches, and return to our outpatient office for close follow-up. The need for delayed surgical intervention of her multiple foot injuries, due to her compromised soft-tissue envelope, was discussed prior to discharge.
Surgical intervention was performed 15 days after the injury, when the soft-tissue swelling had dissipated. The surgical plan included fixation of the multiple metatarsal fractures and lateral column reconstruction and stabilization. With regard to the lateral column, we obtained patient consent for several possible procedures, including fifth TMT joint closed reduction and percutaneous pinning, open reduction and internal fixation (ORIF), and TMT joint reconstruction with iliac crest bone graft (ICBG).
The metatarsals were addressed first via a dorsomedial incision, using a 5-hole 2.7-mm Limited Contact Dynamic Compression Plate (Synthes) to stabilize the first metatarsal and 2.0-mm Kirschner wires (K-wires) to maintain the length and alignment of the second, third, and fourth metatarsals (Figure 2). Closed reduction and percutaneous pinning of the fifth metatarsal was then attempted but abandoned because of persistent instability and subsidence of the cuboid in the proximal and plantar direction. ORIF was then attempted through a dorsolateral incision extending from just distal to the sinus tarsi to the base of the fourth metatarsal. However, the lateral cuboid was too comminuted to accommodate any fixation and prevent fifth TMT joint subluxation and lateral column shortening.
Autograft reconstruction of the lateral column was therefore performed, using radiographs of the patient’s uninjured, contralateral foot as a template for our lateral column shelf arthroplasty (Figure 3). Based on this template, the length and alignment of the lateral column were provisionally maintained with two 2.0-mm K-wires placed between the fifth metatarsal and intact cuboid (Figure 4). Tricortical ICBG was then harvested through an anterior approach to the iliac crest and contoured accordingly to fill the osseous void. To facilitate graft incorporation, comminuted fragments of cuboid bone were removed, with the remaining bone decorticated. The graft was then fixed to the remaining cuboid with two 4.0-mm partially threaded cannulated screws (Synthes; Figures 2, 4). This construct restored the length of the lateral column and effectively buttressed the fifth TMT joint, preventing subsidence and dislocation of the TMT joint.
After a 2-day postoperative course in the hospital, the patient was discharged. She remained non-weight-bearing in a splint with Robert Jones cotton bandage. At her 2-week postoperative visit, all hardware was intact and there was no evidence of infection. Her sutures were removed and she was placed in a new splint. At the patient’s 5-week postoperative visit, all K-wires were removed. At this time she remained non-weight-bearing but was transitioned into a controlled ankle movement (CAM) boot and was allowed to begin active and passive ankle exercises. At her 10-week follow-up, radiographs revealed appropriate interval healing and callus formation. The patient began weight-bearing as tolerated in the CAM boot at that time. At 12 weeks, she was transitioned into a hard-soled shoe for comfort and was allowed to ambulate in the footwear of her choice as tolerated. Her activity levels were slowly advanced, and, at her 12-month follow-up, the patient had returned to playing tennis in her recreational league with no residual sequelae (Figure 5).
Discussion
Although rare, cuboid fractures are critical to identify and can result in significant disability, as they are frequently associated with additional foot trauma, as demonstrated in this case.1-4When isolated cuboid fractures are present, further imaging must be performed, including additional radiographic views and computed tomography, to search for other injuries, such as TMT joint complex disruption.
Only those cuboid fractures that are low-energy, stable, or nondisplaced can be effectively managed conservatively.12In the presence of instability, articular incongruity, or lateral column shortening, operative intervention is warranted. Arthritic degeneration, pain, and deformity result from residual incongruity at the calcaneocuboid or TMT joints, or when lateral column length is not restored.4-6,13 The latter leads to forefoot abduction and lateral subluxation of the lesser metatarsals, with ensuing posttraumatic pes planus or planovalgus deformity, which often necessitates secondary reconstructive procedures or arthrodesis.14,15 Stable reduction and restoration of lateral column length can be challenging, particularly in the setting of comminution and bone loss. Common methods of treatment involve lifting the dorsolateral cortex of the cuboid and buttressing the impacted articular surface with bone graft or bone graft substitutes. Fixation can be achieved with K-wires, small fragment plates and screws, and distraction external fixation.11 The latter is a particularly beneficial technique, as it can be used independent of or in conjunction with ORIF.
In a study by Weber and Locher,11 the short-term to midterm results of cuboid ORIF were assessed in 12 patients. Results were found to be good with respect to restoration of length, joint reconstruction, and overall return to function.11 Admittedly, these authors at times employed a similar but conceptually different approach to our patient. In their 7 patients with severe comminution and lateral column shortening, corticocancellous ICBG was used. However, Weber and Locher11did not describe this as a shelf arthroplasty, but instead as an adjunct to primary ORIF.
In our case, the tricortical ICBG shelf arthroplasty was used as it is in the hip, as a salvage procedure. Although little is known about outcomes following shelf arthroplasty for lateral column reconstruction in the foot, a 50% failure rate has been observed in the hip.16 As such, our preference was to perform an anatomic ORIF of the cuboid and lateral column, with the shelf arthroplasty only indicated if we were unable to achieve this. We believe that the need for tricortical ICBG in the treatment of cuboid fractures is indicative of a more severe injury and that it is a less optimal and more technically demanding intervention compared with primary ORIF. Furthermore, in other studies devoted to the treatment of cuboid fractures, patients requiring reconstruction with structural graft are not included in primary ORIF cohorts.17
As in the hip, suboptimal outcomes may occur when shelf arthroplasty is performed in the foot. There are additional considerations unique to the foot that surgeons must also contemplate when considering shelf arthroplasty. As demonstrated in the literature for adult-acquired flatfoot deformity, lateral column reconstruction is challenging and controversial and is associated with overload, pain, and the need to remove prominent hardware.18 These complications may also occur after shelf arthroplasty for cuboid fractures.
The work by Weber and Locher11 did not elucidate such considerations, and outcomes of ORIF and ICBG reconstruction were not compared. This is a limitation of their study, as differences in functional outcomes between the 2 procedures remain unknown. Given the degree of comminution that precludes ORIF and necessitates a graft reconstruction, we believe that the description of the shelf arthroplasty as a salvage procedure more accurately reflects the severity of injury. This may have implications regarding outcomes and patient expectations that the orthopedic surgeon must address. Future studies must further evaluate the outcomes of this technique, independent of and in comparison with ORIF.
Conclusion
In this case, we describe shelf arthroplasty for cuboid fractures. It is a reconstructive salvage procedure that is indicated when ORIF cannot be achieved. This useful approach to a complex injury must remain in the armamentarium of orthopedic surgeons. As we have demonstrated, it can effectively restore a damaged lateral column, providing length and, in our case, enabling the patient to return to her pre-injury level of activity.
Fractures of the cuboid bone are uncommon, with an annual incidence of approximately 1.8 per 100,000.1 This is largely attributed to the inherent stability provided by its anatomy and position in the foot’s lateral column, where it functions as a link between the lateral column and transverse plantar arch.2 Regarding its anatomy, the cuboid is a pyramidal-shaped bone with 6 bony surfaces that provide tremendous stability—3 of these are articular, 3 nonarticular.
Although the cuboid bone is susceptible to low-energy avulsion injuries, injuries that occur in the setting of high-energy trauma are most concerning, as they often occur concurrently with other midfoot fractures and dislocations. These less common crush injuries are associated with comminution, articular disruption, and shortening of the lateral column.3-5 Avulsion injuries occur via a twisting mechanism, while the more complex nutcracker fracture evolves via longitudinal compression of the lateral column, with the foot in a position of forced plantarflexion.6 Other comminuted fractures occur from direct impact on the lateral aspect of the foot.
Management of cuboid fractures varies according to etiology, fracture displacement, and articular involvement. Conservative management is reserved solely for stable, nondisplaced fractures.7 Unstable fracture-dislocations and those with associated lateral column shortening necessitate operative treatment, which attempts to restore anatomy, stability, and length of the foot’s lateral column.7-9 However, with the exception of open injuries, fractures tenting the skin, and injuries with concomitant compartment syndrome, the high-energy nature of cuboid fractures often precludes early surgical intervention, as the foot’s soft-tissue envelope is too compromised. For this reason, operative intervention is often performed on a delayed basis only after recovery of the soft tissue.
In this case report and literature review, we describe a reconstructive shelf arthroplasty of the fifth tarsometatarsal (TMT) joint as a primary intervention for crush-type cuboid fractures with associated joint subsidence and lateral column shortening. The shelf arthroplasty, which was first credited to Konig in 1891, has historically been described as a remodeling operation using bone graft wedges for the treatment of nonconcentric acetabular dysplasia.10 Although bone grafting is recognized as an effective means of addressing osseous voids in the setting of comminuted cuboid fractures, its specific application in the form of a shelf arthroplasty has not been described.11 The patient provided written informed consent for print and electronic publication of this case report.
Case Report
An otherwise healthy 45-year-old woman presented to our institution’s emergency department (ED) complaining of right foot pain after a motor vehicle accident. She was the restrained driver in a head-on collision. Primary survey revealed a swollen, ecchymotic, and tender right foot. Radiographs demonstrated fractures of her first, second, third, and fourth metatarsals, and a comminuted cuboid fracture with lateral column shortening and disruption of the fifth TMT joint (Figure 1).
Due to swelling, initial management consisted of soft-tissue management through the use of a well-padded splint. As this was her only injury, she was instructed to remain non-weight-bearing, ambulate with crutches, and return to our outpatient office for close follow-up. The need for delayed surgical intervention of her multiple foot injuries, due to her compromised soft-tissue envelope, was discussed prior to discharge.
Surgical intervention was performed 15 days after the injury, when the soft-tissue swelling had dissipated. The surgical plan included fixation of the multiple metatarsal fractures and lateral column reconstruction and stabilization. With regard to the lateral column, we obtained patient consent for several possible procedures, including fifth TMT joint closed reduction and percutaneous pinning, open reduction and internal fixation (ORIF), and TMT joint reconstruction with iliac crest bone graft (ICBG).
The metatarsals were addressed first via a dorsomedial incision, using a 5-hole 2.7-mm Limited Contact Dynamic Compression Plate (Synthes) to stabilize the first metatarsal and 2.0-mm Kirschner wires (K-wires) to maintain the length and alignment of the second, third, and fourth metatarsals (Figure 2). Closed reduction and percutaneous pinning of the fifth metatarsal was then attempted but abandoned because of persistent instability and subsidence of the cuboid in the proximal and plantar direction. ORIF was then attempted through a dorsolateral incision extending from just distal to the sinus tarsi to the base of the fourth metatarsal. However, the lateral cuboid was too comminuted to accommodate any fixation and prevent fifth TMT joint subluxation and lateral column shortening.
Autograft reconstruction of the lateral column was therefore performed, using radiographs of the patient’s uninjured, contralateral foot as a template for our lateral column shelf arthroplasty (Figure 3). Based on this template, the length and alignment of the lateral column were provisionally maintained with two 2.0-mm K-wires placed between the fifth metatarsal and intact cuboid (Figure 4). Tricortical ICBG was then harvested through an anterior approach to the iliac crest and contoured accordingly to fill the osseous void. To facilitate graft incorporation, comminuted fragments of cuboid bone were removed, with the remaining bone decorticated. The graft was then fixed to the remaining cuboid with two 4.0-mm partially threaded cannulated screws (Synthes; Figures 2, 4). This construct restored the length of the lateral column and effectively buttressed the fifth TMT joint, preventing subsidence and dislocation of the TMT joint.
After a 2-day postoperative course in the hospital, the patient was discharged. She remained non-weight-bearing in a splint with Robert Jones cotton bandage. At her 2-week postoperative visit, all hardware was intact and there was no evidence of infection. Her sutures were removed and she was placed in a new splint. At the patient’s 5-week postoperative visit, all K-wires were removed. At this time she remained non-weight-bearing but was transitioned into a controlled ankle movement (CAM) boot and was allowed to begin active and passive ankle exercises. At her 10-week follow-up, radiographs revealed appropriate interval healing and callus formation. The patient began weight-bearing as tolerated in the CAM boot at that time. At 12 weeks, she was transitioned into a hard-soled shoe for comfort and was allowed to ambulate in the footwear of her choice as tolerated. Her activity levels were slowly advanced, and, at her 12-month follow-up, the patient had returned to playing tennis in her recreational league with no residual sequelae (Figure 5).
Discussion
Although rare, cuboid fractures are critical to identify and can result in significant disability, as they are frequently associated with additional foot trauma, as demonstrated in this case.1-4When isolated cuboid fractures are present, further imaging must be performed, including additional radiographic views and computed tomography, to search for other injuries, such as TMT joint complex disruption.
Only those cuboid fractures that are low-energy, stable, or nondisplaced can be effectively managed conservatively.12In the presence of instability, articular incongruity, or lateral column shortening, operative intervention is warranted. Arthritic degeneration, pain, and deformity result from residual incongruity at the calcaneocuboid or TMT joints, or when lateral column length is not restored.4-6,13 The latter leads to forefoot abduction and lateral subluxation of the lesser metatarsals, with ensuing posttraumatic pes planus or planovalgus deformity, which often necessitates secondary reconstructive procedures or arthrodesis.14,15 Stable reduction and restoration of lateral column length can be challenging, particularly in the setting of comminution and bone loss. Common methods of treatment involve lifting the dorsolateral cortex of the cuboid and buttressing the impacted articular surface with bone graft or bone graft substitutes. Fixation can be achieved with K-wires, small fragment plates and screws, and distraction external fixation.11 The latter is a particularly beneficial technique, as it can be used independent of or in conjunction with ORIF.
In a study by Weber and Locher,11 the short-term to midterm results of cuboid ORIF were assessed in 12 patients. Results were found to be good with respect to restoration of length, joint reconstruction, and overall return to function.11 Admittedly, these authors at times employed a similar but conceptually different approach to our patient. In their 7 patients with severe comminution and lateral column shortening, corticocancellous ICBG was used. However, Weber and Locher11did not describe this as a shelf arthroplasty, but instead as an adjunct to primary ORIF.
In our case, the tricortical ICBG shelf arthroplasty was used as it is in the hip, as a salvage procedure. Although little is known about outcomes following shelf arthroplasty for lateral column reconstruction in the foot, a 50% failure rate has been observed in the hip.16 As such, our preference was to perform an anatomic ORIF of the cuboid and lateral column, with the shelf arthroplasty only indicated if we were unable to achieve this. We believe that the need for tricortical ICBG in the treatment of cuboid fractures is indicative of a more severe injury and that it is a less optimal and more technically demanding intervention compared with primary ORIF. Furthermore, in other studies devoted to the treatment of cuboid fractures, patients requiring reconstruction with structural graft are not included in primary ORIF cohorts.17
As in the hip, suboptimal outcomes may occur when shelf arthroplasty is performed in the foot. There are additional considerations unique to the foot that surgeons must also contemplate when considering shelf arthroplasty. As demonstrated in the literature for adult-acquired flatfoot deformity, lateral column reconstruction is challenging and controversial and is associated with overload, pain, and the need to remove prominent hardware.18 These complications may also occur after shelf arthroplasty for cuboid fractures.
The work by Weber and Locher11 did not elucidate such considerations, and outcomes of ORIF and ICBG reconstruction were not compared. This is a limitation of their study, as differences in functional outcomes between the 2 procedures remain unknown. Given the degree of comminution that precludes ORIF and necessitates a graft reconstruction, we believe that the description of the shelf arthroplasty as a salvage procedure more accurately reflects the severity of injury. This may have implications regarding outcomes and patient expectations that the orthopedic surgeon must address. Future studies must further evaluate the outcomes of this technique, independent of and in comparison with ORIF.
Conclusion
In this case, we describe shelf arthroplasty for cuboid fractures. It is a reconstructive salvage procedure that is indicated when ORIF cannot be achieved. This useful approach to a complex injury must remain in the armamentarium of orthopedic surgeons. As we have demonstrated, it can effectively restore a damaged lateral column, providing length and, in our case, enabling the patient to return to her pre-injury level of activity.
1. Court-Brown C, Zinna S, Ekrol I. Classification and epidemiology of midfoot fractures. Foot. 2006;16(3):138-141.
2. Sarrafian SK. Osteology. In: Kelikian AS, ed. Sarrafian’s Anatomy of the Foot and Ankle. Philadelphia, PA: Lippincott; 1993:65-70.
3. Davis CA, Lubowitz J, Thordarson DB. Midtarsal fracture subluxation. Case report and review of the literature. Clin Orthop Relat Res. 1993;(292):264-268.
4. Dewar FP, Evans DC. Occult fracture-subluxation of the midtarsal joint. J Bone Joint Surg Br. 1968;50(2):386-388.
5. Sangeorzan BJ, Swiontkowski MF. Displaced fractures of the cuboid. J Bone Joint Surg Br. 1990;72(3):376-378.
6. Hermel MB, Gershon-Cohen J. The nutcracker fracture of the cuboid by indirect violence. Radiology. 1953;60(6):850-854.
7. Early J, Reid J. Fractures and dislocations of the midfoot and forefoot. In: Heckman JD, Bucholz RW, Court-Brown CM, Tornetta P, eds. Rockwood and Green’s Fractures in Adults. 7th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2009:2120-2126.
8. Richter M, Wippermann B, Krettek C, Schratt HE, Hufner T, Therman H. Fractures and fracture dislocations of the midfoot: occurrence, causes and long-term results. Foot Ankle Int. 2001;22(5):392-398.
9. Borrelli J Jr, De S, VanPelt M. Fracture of the cuboid. J Am Acad Orthop Surg. 2012;20(7):472-477.
10. Love BRT, Stevens PM, Williams PF. A long-term review of shelf arthroplasty. J Bone Joint Surg Br. 1980;62(3):321-325.
11. Weber M, Locher S. Reconstruction of the cuboid in compression fractures: short to midterm results in 12 patients. Foot Ankle Int. 2002;23(11):1008-1013.
12. Ebizie AO. Crush fractures of the cuboid from indirect violence. Injury. 1991;22(5):414-416.
13. Berlet GC, Hodges Davis W, Anderson RB. Tendon arthroplasty for basal fourth and fifth metatarsal arthritis. Foot Ankle Int. 2002;23(5):440-444.
14. Brunet JA, Wiley JJ. The late results of tarsometatarsal joint injuries. J Bone Joint Surg Br. 1987;69(3):437-440.
15. DeAsla R, Deland J. Anatomy and biomechanics of the foot and ankle. In: Thordarson DB, Tornetta P, Einhorn TA, eds. Orthopaedic Surgery Essentials: Foot & Ankle. Philadelphia, PA: Lippincott William & Wilkins; 2004:18-23.
16. Berton C, Bocquet D, Krantz N, Cotton A, Migaud H, Girard J. Shelf arthroplasties long-term outcome: influence of labral tears. A prospective study at a minimal 16 years’ follows up. Orthop Traumatol Surg Res. 2010;96(7):753-759.
17. van Raaij TM, Duffy PJ, Buckley RE. Displaced isolated cuboid fractures: results of four cases with operative treatment. Foot Ankle Int. 2010;31(3):242-246.
18. Grier KM, Walling AK. The use of tricortical autograft versus allograft in lateral column lengthening for adult acquired flatfoot deformity: an analysis of union rates and complications. Foot Ankle Int. 2010;31(9):760-769.
1. Court-Brown C, Zinna S, Ekrol I. Classification and epidemiology of midfoot fractures. Foot. 2006;16(3):138-141.
2. Sarrafian SK. Osteology. In: Kelikian AS, ed. Sarrafian’s Anatomy of the Foot and Ankle. Philadelphia, PA: Lippincott; 1993:65-70.
3. Davis CA, Lubowitz J, Thordarson DB. Midtarsal fracture subluxation. Case report and review of the literature. Clin Orthop Relat Res. 1993;(292):264-268.
4. Dewar FP, Evans DC. Occult fracture-subluxation of the midtarsal joint. J Bone Joint Surg Br. 1968;50(2):386-388.
5. Sangeorzan BJ, Swiontkowski MF. Displaced fractures of the cuboid. J Bone Joint Surg Br. 1990;72(3):376-378.
6. Hermel MB, Gershon-Cohen J. The nutcracker fracture of the cuboid by indirect violence. Radiology. 1953;60(6):850-854.
7. Early J, Reid J. Fractures and dislocations of the midfoot and forefoot. In: Heckman JD, Bucholz RW, Court-Brown CM, Tornetta P, eds. Rockwood and Green’s Fractures in Adults. 7th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2009:2120-2126.
8. Richter M, Wippermann B, Krettek C, Schratt HE, Hufner T, Therman H. Fractures and fracture dislocations of the midfoot: occurrence, causes and long-term results. Foot Ankle Int. 2001;22(5):392-398.
9. Borrelli J Jr, De S, VanPelt M. Fracture of the cuboid. J Am Acad Orthop Surg. 2012;20(7):472-477.
10. Love BRT, Stevens PM, Williams PF. A long-term review of shelf arthroplasty. J Bone Joint Surg Br. 1980;62(3):321-325.
11. Weber M, Locher S. Reconstruction of the cuboid in compression fractures: short to midterm results in 12 patients. Foot Ankle Int. 2002;23(11):1008-1013.
12. Ebizie AO. Crush fractures of the cuboid from indirect violence. Injury. 1991;22(5):414-416.
13. Berlet GC, Hodges Davis W, Anderson RB. Tendon arthroplasty for basal fourth and fifth metatarsal arthritis. Foot Ankle Int. 2002;23(5):440-444.
14. Brunet JA, Wiley JJ. The late results of tarsometatarsal joint injuries. J Bone Joint Surg Br. 1987;69(3):437-440.
15. DeAsla R, Deland J. Anatomy and biomechanics of the foot and ankle. In: Thordarson DB, Tornetta P, Einhorn TA, eds. Orthopaedic Surgery Essentials: Foot & Ankle. Philadelphia, PA: Lippincott William & Wilkins; 2004:18-23.
16. Berton C, Bocquet D, Krantz N, Cotton A, Migaud H, Girard J. Shelf arthroplasties long-term outcome: influence of labral tears. A prospective study at a minimal 16 years’ follows up. Orthop Traumatol Surg Res. 2010;96(7):753-759.
17. van Raaij TM, Duffy PJ, Buckley RE. Displaced isolated cuboid fractures: results of four cases with operative treatment. Foot Ankle Int. 2010;31(3):242-246.
18. Grier KM, Walling AK. The use of tricortical autograft versus allograft in lateral column lengthening for adult acquired flatfoot deformity: an analysis of union rates and complications. Foot Ankle Int. 2010;31(9):760-769.
Definitive Fixation of Hand and Wrist Fractures in the Emergency Department
A mentor—now in his 60s—related his experiences as a resident. On call as a second-year resident, he would often be alone at a busy trauma center with no backup. When a case came in, he would quickly read about it in the library, then manage it in the emergency department (ED) if possible, or, if necessary, take the patient to the operating room (OR).
In the era of improved patient care, increased supervision, and decreased autonomy, this is not the reality anymore.1 In theory, more reliable patient care is the result; however, the pendulum may have swung too far.
There are a number of injuries that are amenable to definitive fixation in the ED, but not as limited an array of injuries as we have perhaps grown accustomed to. Hand injuries are among the most common orthopedic injuries seen in the ED, with fractures of the metacarpals and phalanges constituting nearly one-half of all hand injuries.2 The authors recently attended an excellent instructional course lecture on “The Lost and Found Art of Percutaneous Pinning in the Hand and Wrist” at the annual conference of the American Academy of Orthopaedic Surgeons.3 The presenters itemized a comprehensive list of fractures and simple dislocations of the hand, which could be simply, safely, effectively, and definitively managed through percutaneous pinning techniques. A significant number of unstable fractures of the phalanges and metacarpals can be treated in the ED under mini–C-arm fluoroscopy without an admission and trip to the OR.3,4 Most phalangeal and metacarpal fractures are nondisplaced or minimally displaced and stable, and can often be handled with a combination of closed reduction, buddy-taping, and splinting.5 The indications for percutaneous versus internal fixation depend on a number of factors, including bone quality, degree of comminution, quality of the soft-tissue envelope, articular involvement, acuity of presentation, and goals for motion.6,7
Many simple injury patterns involving unstable fractures or dislocations may be definitively managed in the ED with percutaneous pinning (eg, injuries that are unstable with closed reduction alone but that do not necessitate soft-tissue dissection). These include but are not limited to bony mallet injuries, unstable transverse or oblique fractures or fracture-dislocations of the phalanges and metacarpals, carpometacarpal fracture- dislocations, and underlying fractures that need protection of nail-bed repairs, soft-tissue flaps, or extensor tendon injuries (Figures 1, 2).7,8 The techniques for specific fracture types are beyond the scope of this article but are readily available.5,6
There are certain situations that undoubtedly warrant surgery in the OR, such as neurovascular injury necessitating microvascular repair, flexor tendon laceration, severely comminuted or segmental fractures, irreducible dislocations, and fractures with severe soft-tissue injury or contamination not amenable to primary irrigation, débridement, and closure at bedside.4,7,8
You might ask, “Why would one treat an operative injury in the ED and not formally in the OR?,” and we submit that there are a number of reasons.
First, and most important, with increasing health care costs and decreasing reimbursements, physicians are faced with providing safe but economical care. Percutaneous Kirschner wire (K-wire) fixation is dramatically more cost-effective when performed in the ED than in the OR. The cost of a procedure performed in either setting is similarly dependent on a variety of factors, generally including complexity of the patient or procedure, costs of supplies and pharmacologic agents, fixed versus variable overhead costs, and the professional fees of providers and ancillary personnel.9,10
While the patient is not charged per hour in the ED, it is estimated that ORs in the United States cost, on average, $62 per minute, ranging from as low as $22 to as high as $133 per minute.9 Additionally, the number of personnel involved in running an OR exceeds those for a similar procedure performed in the ED, considering (at a minimum) the orthopedic surgeon, anesthesiologist, scrub and radiology technicians, and nursing personnel required before, during, and after an operation.
While analgesia and procedural sedation can be performed similarly in either setting, it is our experience that patients are managed much more often in the ED with local anesthesia under direct care of only the orthopedic provider, whereas intravenous sedation and general anesthesia are far more commonly implemented in the OR. There are exceptions for pediatric patients or those who are unable to tolerate the procedure under only local anesthesia. Local anesthesia or even intravenous conscious sedation entails less risk as well as lower associated drug costs.11
The difference in risk is especially true for sicker patients undergoing minimally invasive procedures.11 Although administration of adequate procedural analgesia grows increasingly difficult the more proximal the injury, the hand and the fingers are easily and reliably anesthetized with well-placed wrist or digital blocks, with infrequent complications.12 Application of a lidocaine/bupivacaine mixture provides up to 6 to 8 hours of analgesia. A small tourniquet alternative, such as the finger of a sterile glove or phlebotomy tourniquet, applied to the base of the finger or the wrist additionally provides a relatively bloodless field and effectively acts as a Bier block.
Percutaneous pins are much more forgiving than rigid internal fixation. If the initial placement of a pin is unsatisfactory, the pin can be reinserted at little cost.12 Conversely, it may not be possible to reposition a misplaced screw or screw with inadequate purchase and still maintain adequate fixation. While percutaneous pin fixation is not as rigid as screw fixation, the degree of stability provided is adequate for the small forces affecting the hand in most cases. Accordingly, there is a very low incidence of fibrous union or nonunion.13,14 With an increasing appreciation of soft-tissue handling over the past few decades, another significant advantage of K-wire fixation is the obviation of soft-tissue dissection, preserving the biology to maximize healing and minimize adverse sequelae.12 Percutaneous fixation has been shown to achieve functional outcomes comparable to open reduction with internal fixation of operative phalangeal and metacarpal fractures, without soft-tissue disruption, scarring, or implant irritation, and with minimal risk of infection.3,13,15,16 Ultimate range of motion after percutaneous fixation is comparable, if not superior, to that of internal fixation, despite the initial advantage of rigid internal fixation secondary to decreased scarring and lack of indwelling hardware.16,17
While the risk of infection, perhaps the primary concern with percutaneous fixation, has been cited as high as 7%, osteomyelitis is exceedingly rare (<0.5%).3,13,14 Furthermore, pins are often left in place for 3 to 6 weeks, and infection has been found to occur most often at a mean of 10 weeks.7,13 Infection can also be mitigated by intelligent pin placement, relief of residual tension, and splint immobilization.4,15 Pin loosening has similarly been reported in up to 4% of cases in large retrospective studies, occurring at an average of 8 weeks, by which time most pins would have been extricated.13 Other complications related to impaling adjacent neurovascular or tendinous structures have also been cited but are rare.13 A 12-month prospective study of 75 patients specifically evaluating the outcomes after closed reduction with percutaneous fixation of unstable hand fractures in the ED reported only 6 complications at final follow-up.4 Complications were all minor, with no cases of nonunion, delayed union, malunion, pin-tract infection, pyarthrosis, or cellulitis, even in the setting of open fractures. Three patients required revision in the OR for pin migration, initial malreduction, and bone loss in the setting of comminution, respectively. The authors credited their low complication rate to supplementary immobilization.
In conclusion, many unstable simple fractures and dislocations of the hand and wrist can be safely and effectively treated in the ED. While it may seem daunting for a junior resident who is unfamiliar with percutaneous techniques, the authors advocate learning from a more senior mentor. The only additional training required is an understanding of how to apply this skill set in a different setting.
1. Levine WN, Spang RC 3rd. ACGME duty hour requirements: perceptions and impact on resident training and patient care. J Am Acad Orthop Surg. 2014;22(9):535-544.
2. Chung KC, Spilson SV. The frequency and epidemiology of hand and forearm fractures in the United States. J Hand Surg Am. 2001;26(5):908-915.
3. Catalano LW 3rd, Glickel SZ, Strauch RJ, Barron AO. The lost and found art of percutaneous pinning in the hand and wrist. Instructional Course Lectures. Annual Meeting of the American Academy of Orthopaedic Surgeons; March 24, 2015; Las Vegas, NV.
4. Starker I, Eaton RG. Kirschner wire placement in the emergency room. Is there a risk? J Hand Surg Br. 1995;20(4):535-538.
5. Meals C, Meals R. Hand fractures: a review of current treatment strategies. J Hand Surg Am. 2013;38(5):1021-1031.
6. Henry MH. Fractures of the proximal phalanx and metacarpals in the hand: preferred methods of stabilization. J Am Acad Orthop Surg. 2008;16(10):586-595.
7. Klein DM, Belsole RJ. Percutaneous treatment of carpal, metacarpal, and phalangeal injuries. Clin Orthop Relat Res. 2000;(375):116-125.
8. Bernstein ML, Chung KC. Hand fractures and their management: an international view. Injury. 2006;37(11):1043-1048.
9. Macario A. What does one minute of operating room time cost? J Clin Anesth. 2010;22(4):233-236.
10. Williams RM. The costs of visits to emergency departments. N Engl J Med. 1996;334(10):642-646.
11. Bodenham AR, Howell SJ. General anesthesia vs local anaesthesia: an ongoing story. Br J Anaesth. 2009;103(6):785-789.
12. Stern PJ. Management of fractures of the hand over the last 25 years. J Hand Surg Am. 2000;25(5):817-823.
13. Botte MJ, Davis JL, Rose BA, et al. Complications of smooth pin fixation of fractures and dislocations in the hand and wrist. Clin Orthop Relat Res. 1992;(276):194-201.
14. Wray RC Jr, Glunk R. Treatment of delayed union, nonunion, and malunion of the phalanges of the hand. Ann Plast Surg. 1989;22(1):14-18.
15. Hsu LP, Schwartz EG, Kalainov DM, Chen F, Makowiec RL. Complications of K-wire fixation in procedures involving the hand and wrist. J Hand Surg Am. 2011;36(4):610-616.
16. Stem PJ, Wieser MJ, Reilly DG. Complications of plate fixation in the hand skeleton. Clin Orthop Relat Res. 1987;(214):59-65.
17. Page SM, Stern PJ. Complications and range of motion following plate fixation of metacarpal and phalangeal fractures. J Hand Surg Am. 1998;23(5):827-832.
A mentor—now in his 60s—related his experiences as a resident. On call as a second-year resident, he would often be alone at a busy trauma center with no backup. When a case came in, he would quickly read about it in the library, then manage it in the emergency department (ED) if possible, or, if necessary, take the patient to the operating room (OR).
In the era of improved patient care, increased supervision, and decreased autonomy, this is not the reality anymore.1 In theory, more reliable patient care is the result; however, the pendulum may have swung too far.
There are a number of injuries that are amenable to definitive fixation in the ED, but not as limited an array of injuries as we have perhaps grown accustomed to. Hand injuries are among the most common orthopedic injuries seen in the ED, with fractures of the metacarpals and phalanges constituting nearly one-half of all hand injuries.2 The authors recently attended an excellent instructional course lecture on “The Lost and Found Art of Percutaneous Pinning in the Hand and Wrist” at the annual conference of the American Academy of Orthopaedic Surgeons.3 The presenters itemized a comprehensive list of fractures and simple dislocations of the hand, which could be simply, safely, effectively, and definitively managed through percutaneous pinning techniques. A significant number of unstable fractures of the phalanges and metacarpals can be treated in the ED under mini–C-arm fluoroscopy without an admission and trip to the OR.3,4 Most phalangeal and metacarpal fractures are nondisplaced or minimally displaced and stable, and can often be handled with a combination of closed reduction, buddy-taping, and splinting.5 The indications for percutaneous versus internal fixation depend on a number of factors, including bone quality, degree of comminution, quality of the soft-tissue envelope, articular involvement, acuity of presentation, and goals for motion.6,7
Many simple injury patterns involving unstable fractures or dislocations may be definitively managed in the ED with percutaneous pinning (eg, injuries that are unstable with closed reduction alone but that do not necessitate soft-tissue dissection). These include but are not limited to bony mallet injuries, unstable transverse or oblique fractures or fracture-dislocations of the phalanges and metacarpals, carpometacarpal fracture- dislocations, and underlying fractures that need protection of nail-bed repairs, soft-tissue flaps, or extensor tendon injuries (Figures 1, 2).7,8 The techniques for specific fracture types are beyond the scope of this article but are readily available.5,6
There are certain situations that undoubtedly warrant surgery in the OR, such as neurovascular injury necessitating microvascular repair, flexor tendon laceration, severely comminuted or segmental fractures, irreducible dislocations, and fractures with severe soft-tissue injury or contamination not amenable to primary irrigation, débridement, and closure at bedside.4,7,8
You might ask, “Why would one treat an operative injury in the ED and not formally in the OR?,” and we submit that there are a number of reasons.
First, and most important, with increasing health care costs and decreasing reimbursements, physicians are faced with providing safe but economical care. Percutaneous Kirschner wire (K-wire) fixation is dramatically more cost-effective when performed in the ED than in the OR. The cost of a procedure performed in either setting is similarly dependent on a variety of factors, generally including complexity of the patient or procedure, costs of supplies and pharmacologic agents, fixed versus variable overhead costs, and the professional fees of providers and ancillary personnel.9,10
While the patient is not charged per hour in the ED, it is estimated that ORs in the United States cost, on average, $62 per minute, ranging from as low as $22 to as high as $133 per minute.9 Additionally, the number of personnel involved in running an OR exceeds those for a similar procedure performed in the ED, considering (at a minimum) the orthopedic surgeon, anesthesiologist, scrub and radiology technicians, and nursing personnel required before, during, and after an operation.
While analgesia and procedural sedation can be performed similarly in either setting, it is our experience that patients are managed much more often in the ED with local anesthesia under direct care of only the orthopedic provider, whereas intravenous sedation and general anesthesia are far more commonly implemented in the OR. There are exceptions for pediatric patients or those who are unable to tolerate the procedure under only local anesthesia. Local anesthesia or even intravenous conscious sedation entails less risk as well as lower associated drug costs.11
The difference in risk is especially true for sicker patients undergoing minimally invasive procedures.11 Although administration of adequate procedural analgesia grows increasingly difficult the more proximal the injury, the hand and the fingers are easily and reliably anesthetized with well-placed wrist or digital blocks, with infrequent complications.12 Application of a lidocaine/bupivacaine mixture provides up to 6 to 8 hours of analgesia. A small tourniquet alternative, such as the finger of a sterile glove or phlebotomy tourniquet, applied to the base of the finger or the wrist additionally provides a relatively bloodless field and effectively acts as a Bier block.
Percutaneous pins are much more forgiving than rigid internal fixation. If the initial placement of a pin is unsatisfactory, the pin can be reinserted at little cost.12 Conversely, it may not be possible to reposition a misplaced screw or screw with inadequate purchase and still maintain adequate fixation. While percutaneous pin fixation is not as rigid as screw fixation, the degree of stability provided is adequate for the small forces affecting the hand in most cases. Accordingly, there is a very low incidence of fibrous union or nonunion.13,14 With an increasing appreciation of soft-tissue handling over the past few decades, another significant advantage of K-wire fixation is the obviation of soft-tissue dissection, preserving the biology to maximize healing and minimize adverse sequelae.12 Percutaneous fixation has been shown to achieve functional outcomes comparable to open reduction with internal fixation of operative phalangeal and metacarpal fractures, without soft-tissue disruption, scarring, or implant irritation, and with minimal risk of infection.3,13,15,16 Ultimate range of motion after percutaneous fixation is comparable, if not superior, to that of internal fixation, despite the initial advantage of rigid internal fixation secondary to decreased scarring and lack of indwelling hardware.16,17
While the risk of infection, perhaps the primary concern with percutaneous fixation, has been cited as high as 7%, osteomyelitis is exceedingly rare (<0.5%).3,13,14 Furthermore, pins are often left in place for 3 to 6 weeks, and infection has been found to occur most often at a mean of 10 weeks.7,13 Infection can also be mitigated by intelligent pin placement, relief of residual tension, and splint immobilization.4,15 Pin loosening has similarly been reported in up to 4% of cases in large retrospective studies, occurring at an average of 8 weeks, by which time most pins would have been extricated.13 Other complications related to impaling adjacent neurovascular or tendinous structures have also been cited but are rare.13 A 12-month prospective study of 75 patients specifically evaluating the outcomes after closed reduction with percutaneous fixation of unstable hand fractures in the ED reported only 6 complications at final follow-up.4 Complications were all minor, with no cases of nonunion, delayed union, malunion, pin-tract infection, pyarthrosis, or cellulitis, even in the setting of open fractures. Three patients required revision in the OR for pin migration, initial malreduction, and bone loss in the setting of comminution, respectively. The authors credited their low complication rate to supplementary immobilization.
In conclusion, many unstable simple fractures and dislocations of the hand and wrist can be safely and effectively treated in the ED. While it may seem daunting for a junior resident who is unfamiliar with percutaneous techniques, the authors advocate learning from a more senior mentor. The only additional training required is an understanding of how to apply this skill set in a different setting.
A mentor—now in his 60s—related his experiences as a resident. On call as a second-year resident, he would often be alone at a busy trauma center with no backup. When a case came in, he would quickly read about it in the library, then manage it in the emergency department (ED) if possible, or, if necessary, take the patient to the operating room (OR).
In the era of improved patient care, increased supervision, and decreased autonomy, this is not the reality anymore.1 In theory, more reliable patient care is the result; however, the pendulum may have swung too far.
There are a number of injuries that are amenable to definitive fixation in the ED, but not as limited an array of injuries as we have perhaps grown accustomed to. Hand injuries are among the most common orthopedic injuries seen in the ED, with fractures of the metacarpals and phalanges constituting nearly one-half of all hand injuries.2 The authors recently attended an excellent instructional course lecture on “The Lost and Found Art of Percutaneous Pinning in the Hand and Wrist” at the annual conference of the American Academy of Orthopaedic Surgeons.3 The presenters itemized a comprehensive list of fractures and simple dislocations of the hand, which could be simply, safely, effectively, and definitively managed through percutaneous pinning techniques. A significant number of unstable fractures of the phalanges and metacarpals can be treated in the ED under mini–C-arm fluoroscopy without an admission and trip to the OR.3,4 Most phalangeal and metacarpal fractures are nondisplaced or minimally displaced and stable, and can often be handled with a combination of closed reduction, buddy-taping, and splinting.5 The indications for percutaneous versus internal fixation depend on a number of factors, including bone quality, degree of comminution, quality of the soft-tissue envelope, articular involvement, acuity of presentation, and goals for motion.6,7
Many simple injury patterns involving unstable fractures or dislocations may be definitively managed in the ED with percutaneous pinning (eg, injuries that are unstable with closed reduction alone but that do not necessitate soft-tissue dissection). These include but are not limited to bony mallet injuries, unstable transverse or oblique fractures or fracture-dislocations of the phalanges and metacarpals, carpometacarpal fracture- dislocations, and underlying fractures that need protection of nail-bed repairs, soft-tissue flaps, or extensor tendon injuries (Figures 1, 2).7,8 The techniques for specific fracture types are beyond the scope of this article but are readily available.5,6
There are certain situations that undoubtedly warrant surgery in the OR, such as neurovascular injury necessitating microvascular repair, flexor tendon laceration, severely comminuted or segmental fractures, irreducible dislocations, and fractures with severe soft-tissue injury or contamination not amenable to primary irrigation, débridement, and closure at bedside.4,7,8
You might ask, “Why would one treat an operative injury in the ED and not formally in the OR?,” and we submit that there are a number of reasons.
First, and most important, with increasing health care costs and decreasing reimbursements, physicians are faced with providing safe but economical care. Percutaneous Kirschner wire (K-wire) fixation is dramatically more cost-effective when performed in the ED than in the OR. The cost of a procedure performed in either setting is similarly dependent on a variety of factors, generally including complexity of the patient or procedure, costs of supplies and pharmacologic agents, fixed versus variable overhead costs, and the professional fees of providers and ancillary personnel.9,10
While the patient is not charged per hour in the ED, it is estimated that ORs in the United States cost, on average, $62 per minute, ranging from as low as $22 to as high as $133 per minute.9 Additionally, the number of personnel involved in running an OR exceeds those for a similar procedure performed in the ED, considering (at a minimum) the orthopedic surgeon, anesthesiologist, scrub and radiology technicians, and nursing personnel required before, during, and after an operation.
While analgesia and procedural sedation can be performed similarly in either setting, it is our experience that patients are managed much more often in the ED with local anesthesia under direct care of only the orthopedic provider, whereas intravenous sedation and general anesthesia are far more commonly implemented in the OR. There are exceptions for pediatric patients or those who are unable to tolerate the procedure under only local anesthesia. Local anesthesia or even intravenous conscious sedation entails less risk as well as lower associated drug costs.11
The difference in risk is especially true for sicker patients undergoing minimally invasive procedures.11 Although administration of adequate procedural analgesia grows increasingly difficult the more proximal the injury, the hand and the fingers are easily and reliably anesthetized with well-placed wrist or digital blocks, with infrequent complications.12 Application of a lidocaine/bupivacaine mixture provides up to 6 to 8 hours of analgesia. A small tourniquet alternative, such as the finger of a sterile glove or phlebotomy tourniquet, applied to the base of the finger or the wrist additionally provides a relatively bloodless field and effectively acts as a Bier block.
Percutaneous pins are much more forgiving than rigid internal fixation. If the initial placement of a pin is unsatisfactory, the pin can be reinserted at little cost.12 Conversely, it may not be possible to reposition a misplaced screw or screw with inadequate purchase and still maintain adequate fixation. While percutaneous pin fixation is not as rigid as screw fixation, the degree of stability provided is adequate for the small forces affecting the hand in most cases. Accordingly, there is a very low incidence of fibrous union or nonunion.13,14 With an increasing appreciation of soft-tissue handling over the past few decades, another significant advantage of K-wire fixation is the obviation of soft-tissue dissection, preserving the biology to maximize healing and minimize adverse sequelae.12 Percutaneous fixation has been shown to achieve functional outcomes comparable to open reduction with internal fixation of operative phalangeal and metacarpal fractures, without soft-tissue disruption, scarring, or implant irritation, and with minimal risk of infection.3,13,15,16 Ultimate range of motion after percutaneous fixation is comparable, if not superior, to that of internal fixation, despite the initial advantage of rigid internal fixation secondary to decreased scarring and lack of indwelling hardware.16,17
While the risk of infection, perhaps the primary concern with percutaneous fixation, has been cited as high as 7%, osteomyelitis is exceedingly rare (<0.5%).3,13,14 Furthermore, pins are often left in place for 3 to 6 weeks, and infection has been found to occur most often at a mean of 10 weeks.7,13 Infection can also be mitigated by intelligent pin placement, relief of residual tension, and splint immobilization.4,15 Pin loosening has similarly been reported in up to 4% of cases in large retrospective studies, occurring at an average of 8 weeks, by which time most pins would have been extricated.13 Other complications related to impaling adjacent neurovascular or tendinous structures have also been cited but are rare.13 A 12-month prospective study of 75 patients specifically evaluating the outcomes after closed reduction with percutaneous fixation of unstable hand fractures in the ED reported only 6 complications at final follow-up.4 Complications were all minor, with no cases of nonunion, delayed union, malunion, pin-tract infection, pyarthrosis, or cellulitis, even in the setting of open fractures. Three patients required revision in the OR for pin migration, initial malreduction, and bone loss in the setting of comminution, respectively. The authors credited their low complication rate to supplementary immobilization.
In conclusion, many unstable simple fractures and dislocations of the hand and wrist can be safely and effectively treated in the ED. While it may seem daunting for a junior resident who is unfamiliar with percutaneous techniques, the authors advocate learning from a more senior mentor. The only additional training required is an understanding of how to apply this skill set in a different setting.
1. Levine WN, Spang RC 3rd. ACGME duty hour requirements: perceptions and impact on resident training and patient care. J Am Acad Orthop Surg. 2014;22(9):535-544.
2. Chung KC, Spilson SV. The frequency and epidemiology of hand and forearm fractures in the United States. J Hand Surg Am. 2001;26(5):908-915.
3. Catalano LW 3rd, Glickel SZ, Strauch RJ, Barron AO. The lost and found art of percutaneous pinning in the hand and wrist. Instructional Course Lectures. Annual Meeting of the American Academy of Orthopaedic Surgeons; March 24, 2015; Las Vegas, NV.
4. Starker I, Eaton RG. Kirschner wire placement in the emergency room. Is there a risk? J Hand Surg Br. 1995;20(4):535-538.
5. Meals C, Meals R. Hand fractures: a review of current treatment strategies. J Hand Surg Am. 2013;38(5):1021-1031.
6. Henry MH. Fractures of the proximal phalanx and metacarpals in the hand: preferred methods of stabilization. J Am Acad Orthop Surg. 2008;16(10):586-595.
7. Klein DM, Belsole RJ. Percutaneous treatment of carpal, metacarpal, and phalangeal injuries. Clin Orthop Relat Res. 2000;(375):116-125.
8. Bernstein ML, Chung KC. Hand fractures and their management: an international view. Injury. 2006;37(11):1043-1048.
9. Macario A. What does one minute of operating room time cost? J Clin Anesth. 2010;22(4):233-236.
10. Williams RM. The costs of visits to emergency departments. N Engl J Med. 1996;334(10):642-646.
11. Bodenham AR, Howell SJ. General anesthesia vs local anaesthesia: an ongoing story. Br J Anaesth. 2009;103(6):785-789.
12. Stern PJ. Management of fractures of the hand over the last 25 years. J Hand Surg Am. 2000;25(5):817-823.
13. Botte MJ, Davis JL, Rose BA, et al. Complications of smooth pin fixation of fractures and dislocations in the hand and wrist. Clin Orthop Relat Res. 1992;(276):194-201.
14. Wray RC Jr, Glunk R. Treatment of delayed union, nonunion, and malunion of the phalanges of the hand. Ann Plast Surg. 1989;22(1):14-18.
15. Hsu LP, Schwartz EG, Kalainov DM, Chen F, Makowiec RL. Complications of K-wire fixation in procedures involving the hand and wrist. J Hand Surg Am. 2011;36(4):610-616.
16. Stem PJ, Wieser MJ, Reilly DG. Complications of plate fixation in the hand skeleton. Clin Orthop Relat Res. 1987;(214):59-65.
17. Page SM, Stern PJ. Complications and range of motion following plate fixation of metacarpal and phalangeal fractures. J Hand Surg Am. 1998;23(5):827-832.
1. Levine WN, Spang RC 3rd. ACGME duty hour requirements: perceptions and impact on resident training and patient care. J Am Acad Orthop Surg. 2014;22(9):535-544.
2. Chung KC, Spilson SV. The frequency and epidemiology of hand and forearm fractures in the United States. J Hand Surg Am. 2001;26(5):908-915.
3. Catalano LW 3rd, Glickel SZ, Strauch RJ, Barron AO. The lost and found art of percutaneous pinning in the hand and wrist. Instructional Course Lectures. Annual Meeting of the American Academy of Orthopaedic Surgeons; March 24, 2015; Las Vegas, NV.
4. Starker I, Eaton RG. Kirschner wire placement in the emergency room. Is there a risk? J Hand Surg Br. 1995;20(4):535-538.
5. Meals C, Meals R. Hand fractures: a review of current treatment strategies. J Hand Surg Am. 2013;38(5):1021-1031.
6. Henry MH. Fractures of the proximal phalanx and metacarpals in the hand: preferred methods of stabilization. J Am Acad Orthop Surg. 2008;16(10):586-595.
7. Klein DM, Belsole RJ. Percutaneous treatment of carpal, metacarpal, and phalangeal injuries. Clin Orthop Relat Res. 2000;(375):116-125.
8. Bernstein ML, Chung KC. Hand fractures and their management: an international view. Injury. 2006;37(11):1043-1048.
9. Macario A. What does one minute of operating room time cost? J Clin Anesth. 2010;22(4):233-236.
10. Williams RM. The costs of visits to emergency departments. N Engl J Med. 1996;334(10):642-646.
11. Bodenham AR, Howell SJ. General anesthesia vs local anaesthesia: an ongoing story. Br J Anaesth. 2009;103(6):785-789.
12. Stern PJ. Management of fractures of the hand over the last 25 years. J Hand Surg Am. 2000;25(5):817-823.
13. Botte MJ, Davis JL, Rose BA, et al. Complications of smooth pin fixation of fractures and dislocations in the hand and wrist. Clin Orthop Relat Res. 1992;(276):194-201.
14. Wray RC Jr, Glunk R. Treatment of delayed union, nonunion, and malunion of the phalanges of the hand. Ann Plast Surg. 1989;22(1):14-18.
15. Hsu LP, Schwartz EG, Kalainov DM, Chen F, Makowiec RL. Complications of K-wire fixation in procedures involving the hand and wrist. J Hand Surg Am. 2011;36(4):610-616.
16. Stem PJ, Wieser MJ, Reilly DG. Complications of plate fixation in the hand skeleton. Clin Orthop Relat Res. 1987;(214):59-65.
17. Page SM, Stern PJ. Complications and range of motion following plate fixation of metacarpal and phalangeal fractures. J Hand Surg Am. 1998;23(5):827-832.
Resident Guide to Advocacy in Dermatology
It is never too early (or too late!) to get involved in dermatology advocacy. Residency is an ideal time to start learning about advocating on behalf of the specialty of dermatology as well as on behalf of our patients. Many opportunities are available for residents to gain experience and become advocates on national and grassroots levels. As residents, participating in these efforts can help set a solid foundation for future involvement in advocacy, regardless of our ultimate career goals.
American Medical Association
The mission of the American Medical Association (AMA) is “to promote the art and science of medicine and the betterment of public health.”1 Joining the AMA costs $45 for 1 year of resident membership (with a discounted rate for multiyear memberships). As a member, you are given the opportunity to cast a ballot for the national medical specialty society that best represents you in the House of Delegates, the AMA’s principle policy-making body.2 The more votes a particular society receives, the more delegates from that society are added to the House of Delegates, meaning more representation for that specialty organization. It is advised that members choose the society that best represents them: for dermatologists, this most likely would be the American Academy of Dermatology (AAD), among other dermatology organizations that are candidates (ie, the American College of Mohs Surgery, the American Society for Dermatologic Surgery, and the Society for Investigative Dermatology). This representation is key for a specialty like dermatology, which has a relatively smaller number of physicians compared to other larger specialties and therefore has less representation in the House of Delegates.
Additionally, AMA membership grants you access to the entire Journal of the American Medical Association network including a subscription to the specialty journal of your choice.
Patient Advocacy
Patient advocacy groups generally have 3 main goals: education (for patients, patient support networks, and the layperson), research, and lobbying for issues that are in the interest of patients and treatment of dermatologic conditions (eg, funding support, regulation of medical devices, etc).3 In dermatology, the number of patient advocacy groups is growing to represent a myriad of dermatologic conditions, from common conditions like psoriasis to rare genodermatoses (Table). As dermatologists in training, it is key for residents to be involved in patient advocacy and to be aware of the resources that exist for patients to access educational information and support for their respective conditions. These educational materials can help provide more comprehensive care for patients and give patients more autonomy in choosing a physician or hospital to manage their care, help patients become more knowledgeable about available treatment options, and arm patients with more information to address questions that may arise from laypeople regarding their condition.
In terms of patient education, the resources available to patients include informational websites, access to educational materials like pamphlets and multimedia (eg, videos), and special events; for example, the National Psoriasis Foundation hosts walks for patients and their friends and family to raise money for the organization as well as to promote psoriasis awareness and give patients an opportunity to build a support network. Patient advocacy groups also help raise funding for research and have shown to be influential in research initiatives that are granted funding.3 Often, these groups also play a political role and take part in lobbying efforts by patients and support groups by working with politicians to raise awareness or request financial support for particular skin diseases.
The Society for Investigative Dermatology sponsors an application for mobile devices that can assist residents in referring patients to support and advocacy groups (http://www.skinadvocateapp.com).
Grassroots Advocacy
Grassroots advocacy in dermatology means that an individual or group of individuals (in this case, a resident or group of residents) is motivated to take action by contacting legislators and other government officials about gaps in funding and regulation for particular dermatology issues. These efforts often are noticed and taken into consideration by politicians because it is in their best interest to listen to their constituents rather than risk losing support.
The American Academy of Dermatology Association, the advocacy entity of the AAD, hosts the Dermatology Advocacy Network (www.aad-dan.com/default.aspx), which is dedicated to helping dermatologists become advocates. The DAN website helps residents easily identify and contact their local, state, and national legislators to discuss issues or concerns related to the dermatology specialty and medicine as a whole. For example, tanning bed regulation currently is a priority among dermatologists, and the DAN website provides customizable form letters that can be sent electronically to legislators for review.
Furthermore, the AAD offers helpful resources and suggestions for dermatologists and dermatology residents who want to get involved with grassroots advocacy efforts. The website (www.aad.org/advocacy) details current AAD advocacy priorities as well as specific topics such as Medicare physician payment, skin cancer and indoor tanning, drug pricing and availability, state policy, and network adequacy, as these are high-priority issues identified by the AAD that would benefit from action by its members.
Final Thoughts
Many opportunities exist for dermatology residents to get involved in advocacy, from opportunities on the national level with the AMA to patient advocacy and grassroots efforts. It is important for dermatology residents to get involved in advocacy efforts during their training so they may continue to be involved in these efforts as their careers develop. Advocacy helps keep the dermatology specialty relevant and maintain its voice in the national medical arena. It also enhances the dermatology resident’s ability to provide comprehensive quality care for patients by addressing some of their educational and supportive needs that perhaps cannot be addressed in a clinic visit alone. Advocacy also gives residents the opportunity to network and meet colleagues and other individuals with similar goals and interests, which may be beneficial for their future careers. Thus, early involvement in advocacy may be a productive and interesting part of dermatology residency for trainees to be further involved in the specialty.
- AMA mission & guiding principles. American Medical Association Web site. http://www.ama-assn.org/ama/pub/about-ama.page?. Accessed December 22, 2015.
- Specialty Society Representation Ballot. American Medical Association Web site. http://www.ama-assn.org/ama/pub/about-ama/our-people/the-federation-medicine/specialty-society-ballot.page. Accessed December 22, 2015.
- Nijsten T, Bergstresser PR. Patient advocacy groups: let’s stick together. J Invest Dermatol. 2010;130:1757-1759.
It is never too early (or too late!) to get involved in dermatology advocacy. Residency is an ideal time to start learning about advocating on behalf of the specialty of dermatology as well as on behalf of our patients. Many opportunities are available for residents to gain experience and become advocates on national and grassroots levels. As residents, participating in these efforts can help set a solid foundation for future involvement in advocacy, regardless of our ultimate career goals.
American Medical Association
The mission of the American Medical Association (AMA) is “to promote the art and science of medicine and the betterment of public health.”1 Joining the AMA costs $45 for 1 year of resident membership (with a discounted rate for multiyear memberships). As a member, you are given the opportunity to cast a ballot for the national medical specialty society that best represents you in the House of Delegates, the AMA’s principle policy-making body.2 The more votes a particular society receives, the more delegates from that society are added to the House of Delegates, meaning more representation for that specialty organization. It is advised that members choose the society that best represents them: for dermatologists, this most likely would be the American Academy of Dermatology (AAD), among other dermatology organizations that are candidates (ie, the American College of Mohs Surgery, the American Society for Dermatologic Surgery, and the Society for Investigative Dermatology). This representation is key for a specialty like dermatology, which has a relatively smaller number of physicians compared to other larger specialties and therefore has less representation in the House of Delegates.
Additionally, AMA membership grants you access to the entire Journal of the American Medical Association network including a subscription to the specialty journal of your choice.
Patient Advocacy
Patient advocacy groups generally have 3 main goals: education (for patients, patient support networks, and the layperson), research, and lobbying for issues that are in the interest of patients and treatment of dermatologic conditions (eg, funding support, regulation of medical devices, etc).3 In dermatology, the number of patient advocacy groups is growing to represent a myriad of dermatologic conditions, from common conditions like psoriasis to rare genodermatoses (Table). As dermatologists in training, it is key for residents to be involved in patient advocacy and to be aware of the resources that exist for patients to access educational information and support for their respective conditions. These educational materials can help provide more comprehensive care for patients and give patients more autonomy in choosing a physician or hospital to manage their care, help patients become more knowledgeable about available treatment options, and arm patients with more information to address questions that may arise from laypeople regarding their condition.

In terms of patient education, the resources available to patients include informational websites, access to educational materials like pamphlets and multimedia (eg, videos), and special events; for example, the National Psoriasis Foundation hosts walks for patients and their friends and family to raise money for the organization as well as to promote psoriasis awareness and give patients an opportunity to build a support network. Patient advocacy groups also help raise funding for research and have shown to be influential in research initiatives that are granted funding.3 Often, these groups also play a political role and take part in lobbying efforts by patients and support groups by working with politicians to raise awareness or request financial support for particular skin diseases.
The Society for Investigative Dermatology sponsors an application for mobile devices that can assist residents in referring patients to support and advocacy groups (http://www.skinadvocateapp.com).
Grassroots Advocacy
Grassroots advocacy in dermatology means that an individual or group of individuals (in this case, a resident or group of residents) is motivated to take action by contacting legislators and other government officials about gaps in funding and regulation for particular dermatology issues. These efforts often are noticed and taken into consideration by politicians because it is in their best interest to listen to their constituents rather than risk losing support.
The American Academy of Dermatology Association, the advocacy entity of the AAD, hosts the Dermatology Advocacy Network (www.aad-dan.com/default.aspx), which is dedicated to helping dermatologists become advocates. The DAN website helps residents easily identify and contact their local, state, and national legislators to discuss issues or concerns related to the dermatology specialty and medicine as a whole. For example, tanning bed regulation currently is a priority among dermatologists, and the DAN website provides customizable form letters that can be sent electronically to legislators for review.
Furthermore, the AAD offers helpful resources and suggestions for dermatologists and dermatology residents who want to get involved with grassroots advocacy efforts. The website (www.aad.org/advocacy) details current AAD advocacy priorities as well as specific topics such as Medicare physician payment, skin cancer and indoor tanning, drug pricing and availability, state policy, and network adequacy, as these are high-priority issues identified by the AAD that would benefit from action by its members.
Final Thoughts
Many opportunities exist for dermatology residents to get involved in advocacy, from opportunities on the national level with the AMA to patient advocacy and grassroots efforts. It is important for dermatology residents to get involved in advocacy efforts during their training so they may continue to be involved in these efforts as their careers develop. Advocacy helps keep the dermatology specialty relevant and maintain its voice in the national medical arena. It also enhances the dermatology resident’s ability to provide comprehensive quality care for patients by addressing some of their educational and supportive needs that perhaps cannot be addressed in a clinic visit alone. Advocacy also gives residents the opportunity to network and meet colleagues and other individuals with similar goals and interests, which may be beneficial for their future careers. Thus, early involvement in advocacy may be a productive and interesting part of dermatology residency for trainees to be further involved in the specialty.
It is never too early (or too late!) to get involved in dermatology advocacy. Residency is an ideal time to start learning about advocating on behalf of the specialty of dermatology as well as on behalf of our patients. Many opportunities are available for residents to gain experience and become advocates on national and grassroots levels. As residents, participating in these efforts can help set a solid foundation for future involvement in advocacy, regardless of our ultimate career goals.
American Medical Association
The mission of the American Medical Association (AMA) is “to promote the art and science of medicine and the betterment of public health.”1 Joining the AMA costs $45 for 1 year of resident membership (with a discounted rate for multiyear memberships). As a member, you are given the opportunity to cast a ballot for the national medical specialty society that best represents you in the House of Delegates, the AMA’s principle policy-making body.2 The more votes a particular society receives, the more delegates from that society are added to the House of Delegates, meaning more representation for that specialty organization. It is advised that members choose the society that best represents them: for dermatologists, this most likely would be the American Academy of Dermatology (AAD), among other dermatology organizations that are candidates (ie, the American College of Mohs Surgery, the American Society for Dermatologic Surgery, and the Society for Investigative Dermatology). This representation is key for a specialty like dermatology, which has a relatively smaller number of physicians compared to other larger specialties and therefore has less representation in the House of Delegates.
Additionally, AMA membership grants you access to the entire Journal of the American Medical Association network including a subscription to the specialty journal of your choice.
Patient Advocacy
Patient advocacy groups generally have 3 main goals: education (for patients, patient support networks, and the layperson), research, and lobbying for issues that are in the interest of patients and treatment of dermatologic conditions (eg, funding support, regulation of medical devices, etc).3 In dermatology, the number of patient advocacy groups is growing to represent a myriad of dermatologic conditions, from common conditions like psoriasis to rare genodermatoses (Table). As dermatologists in training, it is key for residents to be involved in patient advocacy and to be aware of the resources that exist for patients to access educational information and support for their respective conditions. These educational materials can help provide more comprehensive care for patients and give patients more autonomy in choosing a physician or hospital to manage their care, help patients become more knowledgeable about available treatment options, and arm patients with more information to address questions that may arise from laypeople regarding their condition.

In terms of patient education, the resources available to patients include informational websites, access to educational materials like pamphlets and multimedia (eg, videos), and special events; for example, the National Psoriasis Foundation hosts walks for patients and their friends and family to raise money for the organization as well as to promote psoriasis awareness and give patients an opportunity to build a support network. Patient advocacy groups also help raise funding for research and have shown to be influential in research initiatives that are granted funding.3 Often, these groups also play a political role and take part in lobbying efforts by patients and support groups by working with politicians to raise awareness or request financial support for particular skin diseases.
The Society for Investigative Dermatology sponsors an application for mobile devices that can assist residents in referring patients to support and advocacy groups (http://www.skinadvocateapp.com).
Grassroots Advocacy
Grassroots advocacy in dermatology means that an individual or group of individuals (in this case, a resident or group of residents) is motivated to take action by contacting legislators and other government officials about gaps in funding and regulation for particular dermatology issues. These efforts often are noticed and taken into consideration by politicians because it is in their best interest to listen to their constituents rather than risk losing support.
The American Academy of Dermatology Association, the advocacy entity of the AAD, hosts the Dermatology Advocacy Network (www.aad-dan.com/default.aspx), which is dedicated to helping dermatologists become advocates. The DAN website helps residents easily identify and contact their local, state, and national legislators to discuss issues or concerns related to the dermatology specialty and medicine as a whole. For example, tanning bed regulation currently is a priority among dermatologists, and the DAN website provides customizable form letters that can be sent electronically to legislators for review.
Furthermore, the AAD offers helpful resources and suggestions for dermatologists and dermatology residents who want to get involved with grassroots advocacy efforts. The website (www.aad.org/advocacy) details current AAD advocacy priorities as well as specific topics such as Medicare physician payment, skin cancer and indoor tanning, drug pricing and availability, state policy, and network adequacy, as these are high-priority issues identified by the AAD that would benefit from action by its members.
Final Thoughts
Many opportunities exist for dermatology residents to get involved in advocacy, from opportunities on the national level with the AMA to patient advocacy and grassroots efforts. It is important for dermatology residents to get involved in advocacy efforts during their training so they may continue to be involved in these efforts as their careers develop. Advocacy helps keep the dermatology specialty relevant and maintain its voice in the national medical arena. It also enhances the dermatology resident’s ability to provide comprehensive quality care for patients by addressing some of their educational and supportive needs that perhaps cannot be addressed in a clinic visit alone. Advocacy also gives residents the opportunity to network and meet colleagues and other individuals with similar goals and interests, which may be beneficial for their future careers. Thus, early involvement in advocacy may be a productive and interesting part of dermatology residency for trainees to be further involved in the specialty.
- AMA mission & guiding principles. American Medical Association Web site. http://www.ama-assn.org/ama/pub/about-ama.page?. Accessed December 22, 2015.
- Specialty Society Representation Ballot. American Medical Association Web site. http://www.ama-assn.org/ama/pub/about-ama/our-people/the-federation-medicine/specialty-society-ballot.page. Accessed December 22, 2015.
- Nijsten T, Bergstresser PR. Patient advocacy groups: let’s stick together. J Invest Dermatol. 2010;130:1757-1759.
- AMA mission & guiding principles. American Medical Association Web site. http://www.ama-assn.org/ama/pub/about-ama.page?. Accessed December 22, 2015.
- Specialty Society Representation Ballot. American Medical Association Web site. http://www.ama-assn.org/ama/pub/about-ama/our-people/the-federation-medicine/specialty-society-ballot.page. Accessed December 22, 2015.
- Nijsten T, Bergstresser PR. Patient advocacy groups: let’s stick together. J Invest Dermatol. 2010;130:1757-1759.
Changing trends in diet pill use, from weight loss agent to recreational drug
The prevalence of obesity and obesity-related conditions in the United States is increasing. Many weight-loss products and dietary supplements are used in an attempt to combat this epidemic, but little evidence exists of their efficacy and safety.
We present a case report of a middle-age woman who developed severe psychotic symptoms while taking phentermine hydrochloride (HCl), a psychostimulant similar to amphetamine that is used as a weight-loss agent and for recreational purposes. Phentermine has been associated with mood and psychotic symptoms and has a tendency to cause psychological dependence and tolerance.
To investigate the risks and potential effects of using this drug, we searched OVID and PubMed databases using the search string “phentermine + psychosis.” We conclude that there is a need for awareness about early detection and treatment of reversible psychotic and mood symptoms caused by what might appear to be harmless weight-loss and energy pills.
Obesity epidemic, wide-ranging weight-loss effortsThere has been a dramatic increase in obesity in the United States in the past 20 years: More than one-third of adults and approximately 17% of children and adolescents are obese. Obesity-related conditions, such as heart disease, stroke, and type 2 diabetes mellitus, are leading causes of preventable death.1 Weight monitoring, a healthy lifestyle, surgical intervention, traditional herbs, and diet-pill supplements are some of the modalities used to address this epidemic.
Most so-called supplements for weight loss are exempt from FDA regulation. They do not undergo rigorous testing for safety. Furthermore, many contain controlled substances; some supplements are anti-seizure medications or other prescription drugs; and some are drugs not approved in the United States.2 Since the 1930s, such drugs as dinitrophenol, ephedrine, amphetamine, fenfluramine, and phentermine have flooded the market with the promise of quick weight loss.3,4
Phentermine, a contraction of “phenyltertiary-butylamine,” and its various types (Table) is a psychostimulant of the phenethylamine class, with a pharmacologic profile similar to that of amphetamine. It is known to yield false-positive immunoassay screening results for amphetamines.
CASE REPORT Acute psychotic break
Ms. B, age 37, with a history of postpartum depression, arrives at the emergency room reporting auditory hallucinations of her son and boyfriend; vivid visual hallucinations; and persecutory ideas toward her boyfriend, whom she believes had kidnapped her son. She also complains of insomnia and intermittent confusion for the past week.
Speech is pressured, fast, and difficult to comprehend at times; affect is labile and irritable. Ms. B denies suicidal ideation and is oriented to time, place, and person.
A urine drug screen is positive for amphetamine.
Pre-admission medications include alprazolam, 1 mg as needed, and zolpidem, 10 mg at bedtime, prescribed by Ms. B’s primary care physician for anxiety and insomnia. She discontinued these medications 3 weeks ago because of increased drowsiness at work. She denies other substance use and is unable to account for the positive urine drug screen.
Her medical history, physical examination, and a CT scan of the head are unremarkable. The components of a comprehensive metabolic panel and complete blood count are within normal limits.
After admission, in-depth assessment reveals that Ms. B has been taking phentermine, 37.5 mg (under the brand name Adipex-P), once daily since age 16 for weight loss. She recently discontinued the drug, abruptly, for 1 month, then resumed taking it at an unspecified higher dosage 1 week before she came to the emergency room, for what she said was recreational use and to meet the demands of her job, which required shift work and long hours.
Over the next few days in the hospital, Ms. B’s symptoms resolve as the drug is eliminated from her body. Speech becomes comprehensible and sleep improves. Affective distress diminishes considerably after admission; slight mood lability persists. She no longer reports perceptual disturbances or distress secondary to intrusive thoughts.
Ms. B is discharged 1 week after admission, with instructions to follow up at a dual-diagnosis outpatient program.
Pharmacologic profilePhentermine acts through sympathomimetic pathways by increasing brain noradrenaline and dopamine. The drug has no effect on serotonin.4,5 Phentermine can lead to elevated blood pressure and heart rate, palpitations, restlessness, and insomnia, and can suppress appetite. Increased sympathomimetic activity has been implicated in the ability of phentermine to induce psychotic symptoms.
The literature. Our PubMed search of “phentermine + psychosis” produced 13 results, including 6 case reports of phentermine use. Five citations were more than 4 decades old5-12; only 1 could be considered recent (2011).13
Patients in these reports developed psychotic or manic features after chronic or acute phentermine use, mainly for weight reduction. The most recent article13 mentioned 4 patients who were abusing diet pills recreationally (including “for lethargy”). As with Ms. B, in all 4 of those patients, phentermine precipitated the primary pathology (mania in bipolar disorder; depression in postpartum depression and substance abuse) or revealed underlying illness.
Changing landscape of use and abuseThere has been a trend observed in the pattern of diet pill use: Initially marketed as an appetite suppressant, these pills are now being abused across ethnic, racial, and socioeconomic groups, by males and females.14 There is also a scarcity of useful guidance for clinicians.
Not only are diet pills used by people with an eating disorder; their recreational use is an emerging problem. If reports12,13 continue to reveal that phentermine is a substance of abuse and has catastrophic effects on the user’s psyche, the need for stronger warnings and guidelines might be warranted to allow consumers to make an informed choice about using the drug.
Call for awarenessThe case we presented here exemplifies the importance of tighter regulation of both over-the-counter and prescription stimulant analogs. There is a need for awareness among practitioners about early detection and treatment of reversible psychotic and mood symptoms secondary to what might be promoted as, or appear to be, “harmless” weight loss and energy pills.
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
This unfunded study was presented as a case report poster at the Annual Meeting of the Academy of Psychosomatic Medicine, November 2013, Tucson, Arizona, and at the Colloquium of Scholars of the Philadelphia Psychiatric Society, March 2014, Philadelphia, Pennsylvania.
Drug Brand Names
Alprazolam • Xanax
Fenfluramine • Pondimin
Phentermine HCl • Adipex-P, Fen-Phen, Qsymia, Suprenza
Topiramate • Topamax, Trokendi XR
Zolpidem • Ambien
1. Center for Disease Control. Division of nutrition, physical activity, and obesity. http://www.cdc.gov/obesity/data/adult. html. Updated September 14, 2015. Accessed October 27, 2015.
2. Retamero C, Rivera T, Murphy K. “Ephedra-free” diet pill-induced psychosis. Psychosomatics. 2011;52(6):579-582.
3. Cohen PA, Goday A, Swann JP. The return of rainbow diet pills. Am J Public Health. 2012;102(9):1676-1686.
4. Wellman PJ. Overview of adrenergic anorectic agents. Am J Clin Nutr. 1992;55(suppl 1):193S-198S.
5. Devan GS. Phentermine and psychosis. Br J Psychiatry. 1990;156:442-443.
6. Hoffman BF. Diet pill psychosis. Can Med Assoc J. 1977;116(4):351-355.
7. Hoffman BF. Diet pill psychosis: follow-up after 6 years. Can Med Assoc J. 1983;129(10):1077-1078.
8. Rubin RT. Acute psychotic reaction following ingestion of phentermine. Am J Psychiatry. 1964;120:1124-1125.
9. Schaffer CB, Pauli MW. Psychotic reaction caused by proprietary oral diet agent. Am J Psychiatry. 1980;137(10):1256-12567.
10. Lee SH, Liu CY, Yang YY. Schizophreniform-like psychotic disorder induced by phentermine: a case report. Zhonghua Yi Xue Za Zhi (Taipei). 1998;61(1):44-47.
11. Zimmer JE, Gregory RJ. Bipolar depression associated with fenfluramine and phentermine. J Clin Psychiatry. 1998;59(7):383-384.
12. Bagri S, Reddy G. Delirium with manic symptoms induced by diet pills. J Clin Psychiatry. 1998;59(2):83.
13. Alexander J, Cheng Y, Choudhary J, et al. Phentermine (Duromine) precipitated psychosis. Aust N Z J Psychiatry. 2011;45(8):684-685.
14. Pomeranz JL, Taylor LM, Austin SB. Over-the-counter and out-of-control: legal strategies to protect youths from abusing products for weight control. Am J Public Health. 2013;103(2):222-2253.
The prevalence of obesity and obesity-related conditions in the United States is increasing. Many weight-loss products and dietary supplements are used in an attempt to combat this epidemic, but little evidence exists of their efficacy and safety.
We present a case report of a middle-age woman who developed severe psychotic symptoms while taking phentermine hydrochloride (HCl), a psychostimulant similar to amphetamine that is used as a weight-loss agent and for recreational purposes. Phentermine has been associated with mood and psychotic symptoms and has a tendency to cause psychological dependence and tolerance.
To investigate the risks and potential effects of using this drug, we searched OVID and PubMed databases using the search string “phentermine + psychosis.” We conclude that there is a need for awareness about early detection and treatment of reversible psychotic and mood symptoms caused by what might appear to be harmless weight-loss and energy pills.
Obesity epidemic, wide-ranging weight-loss effortsThere has been a dramatic increase in obesity in the United States in the past 20 years: More than one-third of adults and approximately 17% of children and adolescents are obese. Obesity-related conditions, such as heart disease, stroke, and type 2 diabetes mellitus, are leading causes of preventable death.1 Weight monitoring, a healthy lifestyle, surgical intervention, traditional herbs, and diet-pill supplements are some of the modalities used to address this epidemic.
Most so-called supplements for weight loss are exempt from FDA regulation. They do not undergo rigorous testing for safety. Furthermore, many contain controlled substances; some supplements are anti-seizure medications or other prescription drugs; and some are drugs not approved in the United States.2 Since the 1930s, such drugs as dinitrophenol, ephedrine, amphetamine, fenfluramine, and phentermine have flooded the market with the promise of quick weight loss.3,4
Phentermine, a contraction of “phenyltertiary-butylamine,” and its various types (Table) is a psychostimulant of the phenethylamine class, with a pharmacologic profile similar to that of amphetamine. It is known to yield false-positive immunoassay screening results for amphetamines.
CASE REPORT Acute psychotic break
Ms. B, age 37, with a history of postpartum depression, arrives at the emergency room reporting auditory hallucinations of her son and boyfriend; vivid visual hallucinations; and persecutory ideas toward her boyfriend, whom she believes had kidnapped her son. She also complains of insomnia and intermittent confusion for the past week.
Speech is pressured, fast, and difficult to comprehend at times; affect is labile and irritable. Ms. B denies suicidal ideation and is oriented to time, place, and person.
A urine drug screen is positive for amphetamine.
Pre-admission medications include alprazolam, 1 mg as needed, and zolpidem, 10 mg at bedtime, prescribed by Ms. B’s primary care physician for anxiety and insomnia. She discontinued these medications 3 weeks ago because of increased drowsiness at work. She denies other substance use and is unable to account for the positive urine drug screen.
Her medical history, physical examination, and a CT scan of the head are unremarkable. The components of a comprehensive metabolic panel and complete blood count are within normal limits.
After admission, in-depth assessment reveals that Ms. B has been taking phentermine, 37.5 mg (under the brand name Adipex-P), once daily since age 16 for weight loss. She recently discontinued the drug, abruptly, for 1 month, then resumed taking it at an unspecified higher dosage 1 week before she came to the emergency room, for what she said was recreational use and to meet the demands of her job, which required shift work and long hours.
Over the next few days in the hospital, Ms. B’s symptoms resolve as the drug is eliminated from her body. Speech becomes comprehensible and sleep improves. Affective distress diminishes considerably after admission; slight mood lability persists. She no longer reports perceptual disturbances or distress secondary to intrusive thoughts.
Ms. B is discharged 1 week after admission, with instructions to follow up at a dual-diagnosis outpatient program.
Pharmacologic profilePhentermine acts through sympathomimetic pathways by increasing brain noradrenaline and dopamine. The drug has no effect on serotonin.4,5 Phentermine can lead to elevated blood pressure and heart rate, palpitations, restlessness, and insomnia, and can suppress appetite. Increased sympathomimetic activity has been implicated in the ability of phentermine to induce psychotic symptoms.
The literature. Our PubMed search of “phentermine + psychosis” produced 13 results, including 6 case reports of phentermine use. Five citations were more than 4 decades old5-12; only 1 could be considered recent (2011).13
Patients in these reports developed psychotic or manic features after chronic or acute phentermine use, mainly for weight reduction. The most recent article13 mentioned 4 patients who were abusing diet pills recreationally (including “for lethargy”). As with Ms. B, in all 4 of those patients, phentermine precipitated the primary pathology (mania in bipolar disorder; depression in postpartum depression and substance abuse) or revealed underlying illness.
Changing landscape of use and abuseThere has been a trend observed in the pattern of diet pill use: Initially marketed as an appetite suppressant, these pills are now being abused across ethnic, racial, and socioeconomic groups, by males and females.14 There is also a scarcity of useful guidance for clinicians.
Not only are diet pills used by people with an eating disorder; their recreational use is an emerging problem. If reports12,13 continue to reveal that phentermine is a substance of abuse and has catastrophic effects on the user’s psyche, the need for stronger warnings and guidelines might be warranted to allow consumers to make an informed choice about using the drug.
Call for awarenessThe case we presented here exemplifies the importance of tighter regulation of both over-the-counter and prescription stimulant analogs. There is a need for awareness among practitioners about early detection and treatment of reversible psychotic and mood symptoms secondary to what might be promoted as, or appear to be, “harmless” weight loss and energy pills.
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
This unfunded study was presented as a case report poster at the Annual Meeting of the Academy of Psychosomatic Medicine, November 2013, Tucson, Arizona, and at the Colloquium of Scholars of the Philadelphia Psychiatric Society, March 2014, Philadelphia, Pennsylvania.
Drug Brand Names
Alprazolam • Xanax
Fenfluramine • Pondimin
Phentermine HCl • Adipex-P, Fen-Phen, Qsymia, Suprenza
Topiramate • Topamax, Trokendi XR
Zolpidem • Ambien
The prevalence of obesity and obesity-related conditions in the United States is increasing. Many weight-loss products and dietary supplements are used in an attempt to combat this epidemic, but little evidence exists of their efficacy and safety.
We present a case report of a middle-age woman who developed severe psychotic symptoms while taking phentermine hydrochloride (HCl), a psychostimulant similar to amphetamine that is used as a weight-loss agent and for recreational purposes. Phentermine has been associated with mood and psychotic symptoms and has a tendency to cause psychological dependence and tolerance.
To investigate the risks and potential effects of using this drug, we searched OVID and PubMed databases using the search string “phentermine + psychosis.” We conclude that there is a need for awareness about early detection and treatment of reversible psychotic and mood symptoms caused by what might appear to be harmless weight-loss and energy pills.
Obesity epidemic, wide-ranging weight-loss effortsThere has been a dramatic increase in obesity in the United States in the past 20 years: More than one-third of adults and approximately 17% of children and adolescents are obese. Obesity-related conditions, such as heart disease, stroke, and type 2 diabetes mellitus, are leading causes of preventable death.1 Weight monitoring, a healthy lifestyle, surgical intervention, traditional herbs, and diet-pill supplements are some of the modalities used to address this epidemic.
Most so-called supplements for weight loss are exempt from FDA regulation. They do not undergo rigorous testing for safety. Furthermore, many contain controlled substances; some supplements are anti-seizure medications or other prescription drugs; and some are drugs not approved in the United States.2 Since the 1930s, such drugs as dinitrophenol, ephedrine, amphetamine, fenfluramine, and phentermine have flooded the market with the promise of quick weight loss.3,4
Phentermine, a contraction of “phenyltertiary-butylamine,” and its various types (Table) is a psychostimulant of the phenethylamine class, with a pharmacologic profile similar to that of amphetamine. It is known to yield false-positive immunoassay screening results for amphetamines.
CASE REPORT Acute psychotic break
Ms. B, age 37, with a history of postpartum depression, arrives at the emergency room reporting auditory hallucinations of her son and boyfriend; vivid visual hallucinations; and persecutory ideas toward her boyfriend, whom she believes had kidnapped her son. She also complains of insomnia and intermittent confusion for the past week.
Speech is pressured, fast, and difficult to comprehend at times; affect is labile and irritable. Ms. B denies suicidal ideation and is oriented to time, place, and person.
A urine drug screen is positive for amphetamine.
Pre-admission medications include alprazolam, 1 mg as needed, and zolpidem, 10 mg at bedtime, prescribed by Ms. B’s primary care physician for anxiety and insomnia. She discontinued these medications 3 weeks ago because of increased drowsiness at work. She denies other substance use and is unable to account for the positive urine drug screen.
Her medical history, physical examination, and a CT scan of the head are unremarkable. The components of a comprehensive metabolic panel and complete blood count are within normal limits.
After admission, in-depth assessment reveals that Ms. B has been taking phentermine, 37.5 mg (under the brand name Adipex-P), once daily since age 16 for weight loss. She recently discontinued the drug, abruptly, for 1 month, then resumed taking it at an unspecified higher dosage 1 week before she came to the emergency room, for what she said was recreational use and to meet the demands of her job, which required shift work and long hours.
Over the next few days in the hospital, Ms. B’s symptoms resolve as the drug is eliminated from her body. Speech becomes comprehensible and sleep improves. Affective distress diminishes considerably after admission; slight mood lability persists. She no longer reports perceptual disturbances or distress secondary to intrusive thoughts.
Ms. B is discharged 1 week after admission, with instructions to follow up at a dual-diagnosis outpatient program.
Pharmacologic profilePhentermine acts through sympathomimetic pathways by increasing brain noradrenaline and dopamine. The drug has no effect on serotonin.4,5 Phentermine can lead to elevated blood pressure and heart rate, palpitations, restlessness, and insomnia, and can suppress appetite. Increased sympathomimetic activity has been implicated in the ability of phentermine to induce psychotic symptoms.
The literature. Our PubMed search of “phentermine + psychosis” produced 13 results, including 6 case reports of phentermine use. Five citations were more than 4 decades old5-12; only 1 could be considered recent (2011).13
Patients in these reports developed psychotic or manic features after chronic or acute phentermine use, mainly for weight reduction. The most recent article13 mentioned 4 patients who were abusing diet pills recreationally (including “for lethargy”). As with Ms. B, in all 4 of those patients, phentermine precipitated the primary pathology (mania in bipolar disorder; depression in postpartum depression and substance abuse) or revealed underlying illness.
Changing landscape of use and abuseThere has been a trend observed in the pattern of diet pill use: Initially marketed as an appetite suppressant, these pills are now being abused across ethnic, racial, and socioeconomic groups, by males and females.14 There is also a scarcity of useful guidance for clinicians.
Not only are diet pills used by people with an eating disorder; their recreational use is an emerging problem. If reports12,13 continue to reveal that phentermine is a substance of abuse and has catastrophic effects on the user’s psyche, the need for stronger warnings and guidelines might be warranted to allow consumers to make an informed choice about using the drug.
Call for awarenessThe case we presented here exemplifies the importance of tighter regulation of both over-the-counter and prescription stimulant analogs. There is a need for awareness among practitioners about early detection and treatment of reversible psychotic and mood symptoms secondary to what might be promoted as, or appear to be, “harmless” weight loss and energy pills.
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
This unfunded study was presented as a case report poster at the Annual Meeting of the Academy of Psychosomatic Medicine, November 2013, Tucson, Arizona, and at the Colloquium of Scholars of the Philadelphia Psychiatric Society, March 2014, Philadelphia, Pennsylvania.
Drug Brand Names
Alprazolam • Xanax
Fenfluramine • Pondimin
Phentermine HCl • Adipex-P, Fen-Phen, Qsymia, Suprenza
Topiramate • Topamax, Trokendi XR
Zolpidem • Ambien
1. Center for Disease Control. Division of nutrition, physical activity, and obesity. http://www.cdc.gov/obesity/data/adult. html. Updated September 14, 2015. Accessed October 27, 2015.
2. Retamero C, Rivera T, Murphy K. “Ephedra-free” diet pill-induced psychosis. Psychosomatics. 2011;52(6):579-582.
3. Cohen PA, Goday A, Swann JP. The return of rainbow diet pills. Am J Public Health. 2012;102(9):1676-1686.
4. Wellman PJ. Overview of adrenergic anorectic agents. Am J Clin Nutr. 1992;55(suppl 1):193S-198S.
5. Devan GS. Phentermine and psychosis. Br J Psychiatry. 1990;156:442-443.
6. Hoffman BF. Diet pill psychosis. Can Med Assoc J. 1977;116(4):351-355.
7. Hoffman BF. Diet pill psychosis: follow-up after 6 years. Can Med Assoc J. 1983;129(10):1077-1078.
8. Rubin RT. Acute psychotic reaction following ingestion of phentermine. Am J Psychiatry. 1964;120:1124-1125.
9. Schaffer CB, Pauli MW. Psychotic reaction caused by proprietary oral diet agent. Am J Psychiatry. 1980;137(10):1256-12567.
10. Lee SH, Liu CY, Yang YY. Schizophreniform-like psychotic disorder induced by phentermine: a case report. Zhonghua Yi Xue Za Zhi (Taipei). 1998;61(1):44-47.
11. Zimmer JE, Gregory RJ. Bipolar depression associated with fenfluramine and phentermine. J Clin Psychiatry. 1998;59(7):383-384.
12. Bagri S, Reddy G. Delirium with manic symptoms induced by diet pills. J Clin Psychiatry. 1998;59(2):83.
13. Alexander J, Cheng Y, Choudhary J, et al. Phentermine (Duromine) precipitated psychosis. Aust N Z J Psychiatry. 2011;45(8):684-685.
14. Pomeranz JL, Taylor LM, Austin SB. Over-the-counter and out-of-control: legal strategies to protect youths from abusing products for weight control. Am J Public Health. 2013;103(2):222-2253.
1. Center for Disease Control. Division of nutrition, physical activity, and obesity. http://www.cdc.gov/obesity/data/adult. html. Updated September 14, 2015. Accessed October 27, 2015.
2. Retamero C, Rivera T, Murphy K. “Ephedra-free” diet pill-induced psychosis. Psychosomatics. 2011;52(6):579-582.
3. Cohen PA, Goday A, Swann JP. The return of rainbow diet pills. Am J Public Health. 2012;102(9):1676-1686.
4. Wellman PJ. Overview of adrenergic anorectic agents. Am J Clin Nutr. 1992;55(suppl 1):193S-198S.
5. Devan GS. Phentermine and psychosis. Br J Psychiatry. 1990;156:442-443.
6. Hoffman BF. Diet pill psychosis. Can Med Assoc J. 1977;116(4):351-355.
7. Hoffman BF. Diet pill psychosis: follow-up after 6 years. Can Med Assoc J. 1983;129(10):1077-1078.
8. Rubin RT. Acute psychotic reaction following ingestion of phentermine. Am J Psychiatry. 1964;120:1124-1125.
9. Schaffer CB, Pauli MW. Psychotic reaction caused by proprietary oral diet agent. Am J Psychiatry. 1980;137(10):1256-12567.
10. Lee SH, Liu CY, Yang YY. Schizophreniform-like psychotic disorder induced by phentermine: a case report. Zhonghua Yi Xue Za Zhi (Taipei). 1998;61(1):44-47.
11. Zimmer JE, Gregory RJ. Bipolar depression associated with fenfluramine and phentermine. J Clin Psychiatry. 1998;59(7):383-384.
12. Bagri S, Reddy G. Delirium with manic symptoms induced by diet pills. J Clin Psychiatry. 1998;59(2):83.
13. Alexander J, Cheng Y, Choudhary J, et al. Phentermine (Duromine) precipitated psychosis. Aust N Z J Psychiatry. 2011;45(8):684-685.
14. Pomeranz JL, Taylor LM, Austin SB. Over-the-counter and out-of-control: legal strategies to protect youths from abusing products for weight control. Am J Public Health. 2013;103(2):222-2253.
The Changing Landscape of Orthopedic Practice: Challenges and Opportunities
Orthopedic surgery is going through a time of remarkable change. Health care reform, heightened public scrutiny, shifting population demographics, increased reliance on the Internet for information, ongoing metamorphosis of our profession into a business, and lack of consistent high-quality clinical evidence have created a new frontier of challenges and opportunities. At heart are the needs to deliver high-quality education that is in line with new technological media, to reclaim our ability to guide musculoskeletal care at the policymaking level, to fortify our long-held tradition of ethical responsibility, to invest in research and the training of physician-scientists, to maintain unity among the different subspecialties, and to increase female and minority representation. Never before has understanding and applying the key tenets of our philosophy as orthopedic surgeons been more crucial.
The changing landscape of orthopedic practice has been an unsettling topic in many of the American Academy of Orthopaedic Surgeons (AAOS) presidential addresses in recent years.1-11 What are the challenges and what can we learn moving forward? In this article, we seek to answer these questions by drawing insights from the combined experience and wisdom of past AAOS presidents since the turn of the 21st century.
Education
Education is the cornerstone of providing quality musculoskeletal care12 and staying up to date with technological advances.13 The modes of education delivery, however, have changed. No longer is orthopedic education confined to tangible textbooks and journal articles, nor is it limited to those of us in the profession. Instead, orthopedic education has shifted toward online learning14 and is available to patients and nonorthopedic providers.12 With more patients gaining access to rapidly growing online resources, a unique challenge has arisen: an abundance of data with variable quality of evidence influencing the decision-making process. This has created what Richard Kyle15 described as the “trap of the new technology war,” where patient misinformation and direct-to-consumer marketing can lead to dangerous musculoskeletal care delivery, including unrealistic patient expectations.3 To compound the problem, our ability to provide orthopedic education in formats compatible with the new learning mediums has not been up to the demand, with issues of cost, accessibility, and efficacy plaguing the current process.3,5 Also, we have yet to unlock the benefits of surgical simulation, which has the potential to provide more effective training at no risk to the patient.4,13 By adapting to the new learning formats, we can provide numerous new opportunities for keeping up to date on evolving practice management principles, which, with added accessibility, will be used more often by orthopedic surgeons and the public.13
Research
Research is vital for quality improvement and the continuation of excellence.5 It is only with research that we can provide groundbreaking advances and measure the outcomes of our interventions.2 Unfortunately, orthopedic research funding continues to be disproportionately low, especially given that musculoskeletal ailments are the leading cause of both physician visits and chronic impairment in the United States.2 For example, the National Institute of Arthritis and Musculoskeletal and Skin Diseases receives only 10% of what our country spends on cancer research and 15% of what is spent on heart- and lung-disease research.2 To compound the problem of limited funding, the number of physician-scientists has been dropping at an alarming rate.2 As a result, we must not only refocus our research efforts so that they are efficient and effective, but we must also invest in the training of orthopedic physician-scientists to ensure a continuous stream of groundbreaking discoveries. We owe it to our patients to provide them with proven, effective, and high-quality care.
Industry Relationships
Local and national attention will continue to focus on our relationships with industry. The challenge is twofold: mitigating the negative portrayal of industry relationships and navigating the changes applied to industry funding for research and education.9 Our collaboration with industry is important for the development and advancement of orthopedics,15 but it must be guided by the professional and ethical guidelines established by the AAOS, ensuring that the best interest of patients remains a top priority.8,15 We must maintain the public’s trust by using every opportunity to convey our lone goal in collaborating with industry, ie, improving patient care.9 According to James Beaty,7 any relationship with industry should be “so ethical that it could be printed on the front page of the newspaper and we could face our neighbors with our heads held high.”
Gender and Minority Representation
The racial and ethnic makeup of the United States is undergoing a rapid change. Over the next 4 decades, the white population is projected to become the minority, while women will continue to outnumber men.16 Despite the rapidly changing demographics of the United States, health care disparities persist. As of 2011, minorities and women made up only 22.55% and 14.52%, respectively, of all orthopedic surgery residents.17 This limited diversity in orthopedic training programs is alarming and may lead to suboptimal physician–patient relationships, because patients tend to be more comfortable with and respond better to the care provided by physicians of similar background.3 In addition, if we do not integrate women into orthopedics, the number of female medical students applying to orthopedic residency programs might decline.3
Equating excellent medical care with diversity and cultural competence requires that we bridge the gap that has prevented patients from obtaining high-quality care.8 To achieve this goal, we need to continue recruiting orthopedic surgeons from all segments of our population. Ultimately, health care disparities can be effectively reduced through the delivery of culturally competent care.8
Physician–Patient Relationship
Medical liability has resulted in the development of damaging attitudes among physicians, with many viewing patients as potential adversaries and even avoiding high-risk procedures altogether.6 This deterioration of the physician–patient relationship has been another troubling consequence of managed care that emphasizes quantity and speed.1 As a result, we are perceived by the public as impersonal, poor listeners, and difficult to see on short notice.1
The poor perception of orthopedic surgeons by the general public is not acceptable for a field that places such a high value on excellence. Patient-centered care is at the core of quality improvement, and improving patient relationships starts and ends with us and with each patient we treat.6 In a health care environment in which the average orthopedic surgeon cares for thousands of patients each year, we must make certain to use each opportunity to engage our patients and enhance our relationships with them.6 The basic necessities of patient-centered care include empowerment of the patient through education, better communication, and transparency; providing accurate and evidence-based information; and cooperation among physicians.3,6 The benefits of improving personal relationships with patients are multifold and could have lasting positive effects: increased physician and patient satisfaction, better patient compliance, greater practice efficiency, and fewer malpractice lawsuits.1 We can also benefit from mobilizing a greater constituency to advocate alongside us.6
Unity
Despite accounting for less than 3% of all physicians, orthopedic surgeons have assumed an influential voice in the field of medicine.13 This is attributed not only to the success of our interventions but, more importantly, to the fact that we have “stuck together.”13 The concept of “sticking together” may seem a cliché and facile but will certainly be a pressing need as we move ahead. We draw strength from the breadth and diversity of our subspecialties, but this strength may become a weakness if we do not join in promoting the betterment of our profession as a whole.14 To avoid duplications and bring synergy to all our efforts, we should continue to develop new partnerships in our specialty societies6 and speak with one voice to our patients and to the public.15 Joshua Jacobs11 reminds us of the warning Benjamin Franklin imparted to the signers of the Declaration of Independence, “We must hang together, or most assuredly, we will all hang separately.” To ensure the continued strength of the house of orthopedics, we must live by this tenet.
Advocacy
The federal government has become increasingly involved in regulating the practice of medicine.9 Orthopedic surgery has been hit especially hard, because the cost of implants and continued innovation has fueled the belief that our profession is a major contributor to unsustainable health care costs.11 We now face multiple legislative regulations related to physician reimbursement, ownership, self-referral, medical liability, and mandates of the Affordable Care Act.9 As a result, there has been a decreasing role for orthopedic surgeons as independent practitioners, with more orthopedists forgoing physician-owned practices for large hospital corporations. We are also in increasing competition for limited resources.10 This is compounded by the fact that those regulating health care, paying for health care, and allocating research funding fail to comprehend the high societal needs for treatment of musculoskeletal diseases and injuries,6 which will only increase in the coming decades.14
The aforementioned challenges make our involvement at all levels of the political process more necessary than ever before.5,9 E. Anthony Rankin8 reminds us, “As physicians, we cannot in good conscience allow our patients’ access to quality orthopedic care to be determined solely by the government, the insurance companies, the trial lawyers, or others…. Either we will have a voice in defining the future of health care, or it will be defined by others for us.” Our advocacy approach, however, should be very careful. Joshua Jacobs11 cautions that “we will be most effective if our advocacy message is presented as a potential solution to the current health care crisis, not just as a demand for fair reimbursement.” Instead, we can achieve this goal with what Richard Gelberman2 summarized as “doing what we do best: accumulating knowledge, positioning ourselves as the authorities that we are, and using what we learn to advocate for better patient care and research.”
Value Medicine
Orthopedic surgeons are now expected to provide not just high-quality care but low-cost care. In line with the emerging emphasis on value, sharp focus has been placed on the assessment of physician performance and treatment outcomes as quality-of-care measures.6 But how have we measured the quality of the care we provide? We have not, or, at least, we have not had valid or reliable means of doing so.6 Gone are the days of telling the world of the excellence of our profession in the treatment of musculoskeletal disease. We now must prove to our patients, the government, and payers that what we do works.12,13 If we fail to communicate the cost effectiveness of our interventions, our new knowledge and technologies will not be accepted or funded.10 We should, however, not be discouraged by the new “value equation,” but use it as an incentive to provide evidence-based care and to improve the efficiency of resource utilization.14 Today, we are urged to be leaders in quality improvement, both in our individual orthopedic practices and as a profession.10,12,13
Conclusion
Meeting increasingly higher demands for musculoskeletal care in an evolving medical landscape will bring a new set of challenges that will be more frequent and more intense than those in the past.14 Today, we are tasked with providing fiscally efficient, culturally competent, high-quality, evidence-based, and compassionate care. We are also tasked with reclaiming our ability to shape the future of our profession at the policymaking level. In doing so, the need for unity, advocacy, commitment to education and research, women and minority representation, and open communication with the public has never been more relevant. As we continue to advance as a profession, we must resist the temptation to look back in defiance of change but move forward, confident in our ability to evolve. ◾
1. Canale ST. The orthopaedic forum. Falling in love again. J Bone Joint Surg Am. 2000;82(5):739-742.
2. Gelberman RH. The Academy on the edge: taking charge of our future. J Bone Joint Surg Am. 2001;83(6):946-950.
3. Tolo VT. The challenges of change: is orthopaedics ready? J Bone Joint Surg Am. 2002;84(9):1707-1713.
4. Herndon JH. One more turn of the wrench. J Bone Joint Surg Am. 2003;85(10):2036-2048.
5. Bucholz RW. Knowledge is our business. J Bone Joint Surg Am. 2004;86(7):1575-1578.
6. Weinstein SL. Nothing about you...without you. J Bone Joint Surg Am. 2005;87(7):1648-1652.
7. Beaty JH. Presidential address: “Building the best . . . Lifelong learning”. J Am Acad Orthop Surg. 2007;15(9):515-518.
8. Rankin EA. Presidential Address: advocacy now... for our patients and our profession. J Am Acad Orthop Surg. 2008;16(6):303-305.
9. Zuckerman JD. Silk purses, sows’ ears, and heap ash—turning challenges into opportunities. J Am Acad Orthop Surg. 2009;17(5):271-275.
10. Tongue JR. Strong on vision, flexible on details. J Am Acad Orthop Surg. 2012;20(4):187-189.
11. Jacobs JJ. Moving forward: from curses to blessings. J Am Acad Orthop Surg. 2013;21(5):261-265.
12. Callaghan JJ. Quality of care: getting from good to great. J Am Acad Orthop Surg. 2010;8(9):516-519.
13. Berry DJ. Informed by our past, building our future. J Am Acad Orthop Surg. 2011;19(4):187-190.
14. Azar FM. Building a bigger box. J Am Acad Orthop Surg. 2014;22(6):341-345.
15. Kyle RF. Presidential Address: Together we are one. J Am Acad Orthop Surg. 2006;14(5):261-264.
16. Vincent GK, Velkoff VA. The Next Four Decades: The Older Population in the United States: 2010 to 2050. Washington, DC: Economics and Statistics Administration, US Census Bureau, US Dept of Commerce; 2010.
17. American Academy of Orthopaedic Surgeons Department of Research and Scientific Affairs. 1998-2011 Resident Diversity Survey Report. American Academy of Orthopaedic Surgeons website. http://www3.aaos.org/about/diversity/pdfs/resident_trend.pdf. Published March 9, 2012. Accessed October 26, 2015.
Orthopedic surgery is going through a time of remarkable change. Health care reform, heightened public scrutiny, shifting population demographics, increased reliance on the Internet for information, ongoing metamorphosis of our profession into a business, and lack of consistent high-quality clinical evidence have created a new frontier of challenges and opportunities. At heart are the needs to deliver high-quality education that is in line with new technological media, to reclaim our ability to guide musculoskeletal care at the policymaking level, to fortify our long-held tradition of ethical responsibility, to invest in research and the training of physician-scientists, to maintain unity among the different subspecialties, and to increase female and minority representation. Never before has understanding and applying the key tenets of our philosophy as orthopedic surgeons been more crucial.
The changing landscape of orthopedic practice has been an unsettling topic in many of the American Academy of Orthopaedic Surgeons (AAOS) presidential addresses in recent years.1-11 What are the challenges and what can we learn moving forward? In this article, we seek to answer these questions by drawing insights from the combined experience and wisdom of past AAOS presidents since the turn of the 21st century.
Education
Education is the cornerstone of providing quality musculoskeletal care12 and staying up to date with technological advances.13 The modes of education delivery, however, have changed. No longer is orthopedic education confined to tangible textbooks and journal articles, nor is it limited to those of us in the profession. Instead, orthopedic education has shifted toward online learning14 and is available to patients and nonorthopedic providers.12 With more patients gaining access to rapidly growing online resources, a unique challenge has arisen: an abundance of data with variable quality of evidence influencing the decision-making process. This has created what Richard Kyle15 described as the “trap of the new technology war,” where patient misinformation and direct-to-consumer marketing can lead to dangerous musculoskeletal care delivery, including unrealistic patient expectations.3 To compound the problem, our ability to provide orthopedic education in formats compatible with the new learning mediums has not been up to the demand, with issues of cost, accessibility, and efficacy plaguing the current process.3,5 Also, we have yet to unlock the benefits of surgical simulation, which has the potential to provide more effective training at no risk to the patient.4,13 By adapting to the new learning formats, we can provide numerous new opportunities for keeping up to date on evolving practice management principles, which, with added accessibility, will be used more often by orthopedic surgeons and the public.13
Research
Research is vital for quality improvement and the continuation of excellence.5 It is only with research that we can provide groundbreaking advances and measure the outcomes of our interventions.2 Unfortunately, orthopedic research funding continues to be disproportionately low, especially given that musculoskeletal ailments are the leading cause of both physician visits and chronic impairment in the United States.2 For example, the National Institute of Arthritis and Musculoskeletal and Skin Diseases receives only 10% of what our country spends on cancer research and 15% of what is spent on heart- and lung-disease research.2 To compound the problem of limited funding, the number of physician-scientists has been dropping at an alarming rate.2 As a result, we must not only refocus our research efforts so that they are efficient and effective, but we must also invest in the training of orthopedic physician-scientists to ensure a continuous stream of groundbreaking discoveries. We owe it to our patients to provide them with proven, effective, and high-quality care.
Industry Relationships
Local and national attention will continue to focus on our relationships with industry. The challenge is twofold: mitigating the negative portrayal of industry relationships and navigating the changes applied to industry funding for research and education.9 Our collaboration with industry is important for the development and advancement of orthopedics,15 but it must be guided by the professional and ethical guidelines established by the AAOS, ensuring that the best interest of patients remains a top priority.8,15 We must maintain the public’s trust by using every opportunity to convey our lone goal in collaborating with industry, ie, improving patient care.9 According to James Beaty,7 any relationship with industry should be “so ethical that it could be printed on the front page of the newspaper and we could face our neighbors with our heads held high.”
Gender and Minority Representation
The racial and ethnic makeup of the United States is undergoing a rapid change. Over the next 4 decades, the white population is projected to become the minority, while women will continue to outnumber men.16 Despite the rapidly changing demographics of the United States, health care disparities persist. As of 2011, minorities and women made up only 22.55% and 14.52%, respectively, of all orthopedic surgery residents.17 This limited diversity in orthopedic training programs is alarming and may lead to suboptimal physician–patient relationships, because patients tend to be more comfortable with and respond better to the care provided by physicians of similar background.3 In addition, if we do not integrate women into orthopedics, the number of female medical students applying to orthopedic residency programs might decline.3
Equating excellent medical care with diversity and cultural competence requires that we bridge the gap that has prevented patients from obtaining high-quality care.8 To achieve this goal, we need to continue recruiting orthopedic surgeons from all segments of our population. Ultimately, health care disparities can be effectively reduced through the delivery of culturally competent care.8
Physician–Patient Relationship
Medical liability has resulted in the development of damaging attitudes among physicians, with many viewing patients as potential adversaries and even avoiding high-risk procedures altogether.6 This deterioration of the physician–patient relationship has been another troubling consequence of managed care that emphasizes quantity and speed.1 As a result, we are perceived by the public as impersonal, poor listeners, and difficult to see on short notice.1
The poor perception of orthopedic surgeons by the general public is not acceptable for a field that places such a high value on excellence. Patient-centered care is at the core of quality improvement, and improving patient relationships starts and ends with us and with each patient we treat.6 In a health care environment in which the average orthopedic surgeon cares for thousands of patients each year, we must make certain to use each opportunity to engage our patients and enhance our relationships with them.6 The basic necessities of patient-centered care include empowerment of the patient through education, better communication, and transparency; providing accurate and evidence-based information; and cooperation among physicians.3,6 The benefits of improving personal relationships with patients are multifold and could have lasting positive effects: increased physician and patient satisfaction, better patient compliance, greater practice efficiency, and fewer malpractice lawsuits.1 We can also benefit from mobilizing a greater constituency to advocate alongside us.6
Unity
Despite accounting for less than 3% of all physicians, orthopedic surgeons have assumed an influential voice in the field of medicine.13 This is attributed not only to the success of our interventions but, more importantly, to the fact that we have “stuck together.”13 The concept of “sticking together” may seem a cliché and facile but will certainly be a pressing need as we move ahead. We draw strength from the breadth and diversity of our subspecialties, but this strength may become a weakness if we do not join in promoting the betterment of our profession as a whole.14 To avoid duplications and bring synergy to all our efforts, we should continue to develop new partnerships in our specialty societies6 and speak with one voice to our patients and to the public.15 Joshua Jacobs11 reminds us of the warning Benjamin Franklin imparted to the signers of the Declaration of Independence, “We must hang together, or most assuredly, we will all hang separately.” To ensure the continued strength of the house of orthopedics, we must live by this tenet.
Advocacy
The federal government has become increasingly involved in regulating the practice of medicine.9 Orthopedic surgery has been hit especially hard, because the cost of implants and continued innovation has fueled the belief that our profession is a major contributor to unsustainable health care costs.11 We now face multiple legislative regulations related to physician reimbursement, ownership, self-referral, medical liability, and mandates of the Affordable Care Act.9 As a result, there has been a decreasing role for orthopedic surgeons as independent practitioners, with more orthopedists forgoing physician-owned practices for large hospital corporations. We are also in increasing competition for limited resources.10 This is compounded by the fact that those regulating health care, paying for health care, and allocating research funding fail to comprehend the high societal needs for treatment of musculoskeletal diseases and injuries,6 which will only increase in the coming decades.14
The aforementioned challenges make our involvement at all levels of the political process more necessary than ever before.5,9 E. Anthony Rankin8 reminds us, “As physicians, we cannot in good conscience allow our patients’ access to quality orthopedic care to be determined solely by the government, the insurance companies, the trial lawyers, or others…. Either we will have a voice in defining the future of health care, or it will be defined by others for us.” Our advocacy approach, however, should be very careful. Joshua Jacobs11 cautions that “we will be most effective if our advocacy message is presented as a potential solution to the current health care crisis, not just as a demand for fair reimbursement.” Instead, we can achieve this goal with what Richard Gelberman2 summarized as “doing what we do best: accumulating knowledge, positioning ourselves as the authorities that we are, and using what we learn to advocate for better patient care and research.”
Value Medicine
Orthopedic surgeons are now expected to provide not just high-quality care but low-cost care. In line with the emerging emphasis on value, sharp focus has been placed on the assessment of physician performance and treatment outcomes as quality-of-care measures.6 But how have we measured the quality of the care we provide? We have not, or, at least, we have not had valid or reliable means of doing so.6 Gone are the days of telling the world of the excellence of our profession in the treatment of musculoskeletal disease. We now must prove to our patients, the government, and payers that what we do works.12,13 If we fail to communicate the cost effectiveness of our interventions, our new knowledge and technologies will not be accepted or funded.10 We should, however, not be discouraged by the new “value equation,” but use it as an incentive to provide evidence-based care and to improve the efficiency of resource utilization.14 Today, we are urged to be leaders in quality improvement, both in our individual orthopedic practices and as a profession.10,12,13
Conclusion
Meeting increasingly higher demands for musculoskeletal care in an evolving medical landscape will bring a new set of challenges that will be more frequent and more intense than those in the past.14 Today, we are tasked with providing fiscally efficient, culturally competent, high-quality, evidence-based, and compassionate care. We are also tasked with reclaiming our ability to shape the future of our profession at the policymaking level. In doing so, the need for unity, advocacy, commitment to education and research, women and minority representation, and open communication with the public has never been more relevant. As we continue to advance as a profession, we must resist the temptation to look back in defiance of change but move forward, confident in our ability to evolve. ◾
Orthopedic surgery is going through a time of remarkable change. Health care reform, heightened public scrutiny, shifting population demographics, increased reliance on the Internet for information, ongoing metamorphosis of our profession into a business, and lack of consistent high-quality clinical evidence have created a new frontier of challenges and opportunities. At heart are the needs to deliver high-quality education that is in line with new technological media, to reclaim our ability to guide musculoskeletal care at the policymaking level, to fortify our long-held tradition of ethical responsibility, to invest in research and the training of physician-scientists, to maintain unity among the different subspecialties, and to increase female and minority representation. Never before has understanding and applying the key tenets of our philosophy as orthopedic surgeons been more crucial.
The changing landscape of orthopedic practice has been an unsettling topic in many of the American Academy of Orthopaedic Surgeons (AAOS) presidential addresses in recent years.1-11 What are the challenges and what can we learn moving forward? In this article, we seek to answer these questions by drawing insights from the combined experience and wisdom of past AAOS presidents since the turn of the 21st century.
Education
Education is the cornerstone of providing quality musculoskeletal care12 and staying up to date with technological advances.13 The modes of education delivery, however, have changed. No longer is orthopedic education confined to tangible textbooks and journal articles, nor is it limited to those of us in the profession. Instead, orthopedic education has shifted toward online learning14 and is available to patients and nonorthopedic providers.12 With more patients gaining access to rapidly growing online resources, a unique challenge has arisen: an abundance of data with variable quality of evidence influencing the decision-making process. This has created what Richard Kyle15 described as the “trap of the new technology war,” where patient misinformation and direct-to-consumer marketing can lead to dangerous musculoskeletal care delivery, including unrealistic patient expectations.3 To compound the problem, our ability to provide orthopedic education in formats compatible with the new learning mediums has not been up to the demand, with issues of cost, accessibility, and efficacy plaguing the current process.3,5 Also, we have yet to unlock the benefits of surgical simulation, which has the potential to provide more effective training at no risk to the patient.4,13 By adapting to the new learning formats, we can provide numerous new opportunities for keeping up to date on evolving practice management principles, which, with added accessibility, will be used more often by orthopedic surgeons and the public.13
Research
Research is vital for quality improvement and the continuation of excellence.5 It is only with research that we can provide groundbreaking advances and measure the outcomes of our interventions.2 Unfortunately, orthopedic research funding continues to be disproportionately low, especially given that musculoskeletal ailments are the leading cause of both physician visits and chronic impairment in the United States.2 For example, the National Institute of Arthritis and Musculoskeletal and Skin Diseases receives only 10% of what our country spends on cancer research and 15% of what is spent on heart- and lung-disease research.2 To compound the problem of limited funding, the number of physician-scientists has been dropping at an alarming rate.2 As a result, we must not only refocus our research efforts so that they are efficient and effective, but we must also invest in the training of orthopedic physician-scientists to ensure a continuous stream of groundbreaking discoveries. We owe it to our patients to provide them with proven, effective, and high-quality care.
Industry Relationships
Local and national attention will continue to focus on our relationships with industry. The challenge is twofold: mitigating the negative portrayal of industry relationships and navigating the changes applied to industry funding for research and education.9 Our collaboration with industry is important for the development and advancement of orthopedics,15 but it must be guided by the professional and ethical guidelines established by the AAOS, ensuring that the best interest of patients remains a top priority.8,15 We must maintain the public’s trust by using every opportunity to convey our lone goal in collaborating with industry, ie, improving patient care.9 According to James Beaty,7 any relationship with industry should be “so ethical that it could be printed on the front page of the newspaper and we could face our neighbors with our heads held high.”
Gender and Minority Representation
The racial and ethnic makeup of the United States is undergoing a rapid change. Over the next 4 decades, the white population is projected to become the minority, while women will continue to outnumber men.16 Despite the rapidly changing demographics of the United States, health care disparities persist. As of 2011, minorities and women made up only 22.55% and 14.52%, respectively, of all orthopedic surgery residents.17 This limited diversity in orthopedic training programs is alarming and may lead to suboptimal physician–patient relationships, because patients tend to be more comfortable with and respond better to the care provided by physicians of similar background.3 In addition, if we do not integrate women into orthopedics, the number of female medical students applying to orthopedic residency programs might decline.3
Equating excellent medical care with diversity and cultural competence requires that we bridge the gap that has prevented patients from obtaining high-quality care.8 To achieve this goal, we need to continue recruiting orthopedic surgeons from all segments of our population. Ultimately, health care disparities can be effectively reduced through the delivery of culturally competent care.8
Physician–Patient Relationship
Medical liability has resulted in the development of damaging attitudes among physicians, with many viewing patients as potential adversaries and even avoiding high-risk procedures altogether.6 This deterioration of the physician–patient relationship has been another troubling consequence of managed care that emphasizes quantity and speed.1 As a result, we are perceived by the public as impersonal, poor listeners, and difficult to see on short notice.1
The poor perception of orthopedic surgeons by the general public is not acceptable for a field that places such a high value on excellence. Patient-centered care is at the core of quality improvement, and improving patient relationships starts and ends with us and with each patient we treat.6 In a health care environment in which the average orthopedic surgeon cares for thousands of patients each year, we must make certain to use each opportunity to engage our patients and enhance our relationships with them.6 The basic necessities of patient-centered care include empowerment of the patient through education, better communication, and transparency; providing accurate and evidence-based information; and cooperation among physicians.3,6 The benefits of improving personal relationships with patients are multifold and could have lasting positive effects: increased physician and patient satisfaction, better patient compliance, greater practice efficiency, and fewer malpractice lawsuits.1 We can also benefit from mobilizing a greater constituency to advocate alongside us.6
Unity
Despite accounting for less than 3% of all physicians, orthopedic surgeons have assumed an influential voice in the field of medicine.13 This is attributed not only to the success of our interventions but, more importantly, to the fact that we have “stuck together.”13 The concept of “sticking together” may seem a cliché and facile but will certainly be a pressing need as we move ahead. We draw strength from the breadth and diversity of our subspecialties, but this strength may become a weakness if we do not join in promoting the betterment of our profession as a whole.14 To avoid duplications and bring synergy to all our efforts, we should continue to develop new partnerships in our specialty societies6 and speak with one voice to our patients and to the public.15 Joshua Jacobs11 reminds us of the warning Benjamin Franklin imparted to the signers of the Declaration of Independence, “We must hang together, or most assuredly, we will all hang separately.” To ensure the continued strength of the house of orthopedics, we must live by this tenet.
Advocacy
The federal government has become increasingly involved in regulating the practice of medicine.9 Orthopedic surgery has been hit especially hard, because the cost of implants and continued innovation has fueled the belief that our profession is a major contributor to unsustainable health care costs.11 We now face multiple legislative regulations related to physician reimbursement, ownership, self-referral, medical liability, and mandates of the Affordable Care Act.9 As a result, there has been a decreasing role for orthopedic surgeons as independent practitioners, with more orthopedists forgoing physician-owned practices for large hospital corporations. We are also in increasing competition for limited resources.10 This is compounded by the fact that those regulating health care, paying for health care, and allocating research funding fail to comprehend the high societal needs for treatment of musculoskeletal diseases and injuries,6 which will only increase in the coming decades.14
The aforementioned challenges make our involvement at all levels of the political process more necessary than ever before.5,9 E. Anthony Rankin8 reminds us, “As physicians, we cannot in good conscience allow our patients’ access to quality orthopedic care to be determined solely by the government, the insurance companies, the trial lawyers, or others…. Either we will have a voice in defining the future of health care, or it will be defined by others for us.” Our advocacy approach, however, should be very careful. Joshua Jacobs11 cautions that “we will be most effective if our advocacy message is presented as a potential solution to the current health care crisis, not just as a demand for fair reimbursement.” Instead, we can achieve this goal with what Richard Gelberman2 summarized as “doing what we do best: accumulating knowledge, positioning ourselves as the authorities that we are, and using what we learn to advocate for better patient care and research.”
Value Medicine
Orthopedic surgeons are now expected to provide not just high-quality care but low-cost care. In line with the emerging emphasis on value, sharp focus has been placed on the assessment of physician performance and treatment outcomes as quality-of-care measures.6 But how have we measured the quality of the care we provide? We have not, or, at least, we have not had valid or reliable means of doing so.6 Gone are the days of telling the world of the excellence of our profession in the treatment of musculoskeletal disease. We now must prove to our patients, the government, and payers that what we do works.12,13 If we fail to communicate the cost effectiveness of our interventions, our new knowledge and technologies will not be accepted or funded.10 We should, however, not be discouraged by the new “value equation,” but use it as an incentive to provide evidence-based care and to improve the efficiency of resource utilization.14 Today, we are urged to be leaders in quality improvement, both in our individual orthopedic practices and as a profession.10,12,13
Conclusion
Meeting increasingly higher demands for musculoskeletal care in an evolving medical landscape will bring a new set of challenges that will be more frequent and more intense than those in the past.14 Today, we are tasked with providing fiscally efficient, culturally competent, high-quality, evidence-based, and compassionate care. We are also tasked with reclaiming our ability to shape the future of our profession at the policymaking level. In doing so, the need for unity, advocacy, commitment to education and research, women and minority representation, and open communication with the public has never been more relevant. As we continue to advance as a profession, we must resist the temptation to look back in defiance of change but move forward, confident in our ability to evolve. ◾
1. Canale ST. The orthopaedic forum. Falling in love again. J Bone Joint Surg Am. 2000;82(5):739-742.
2. Gelberman RH. The Academy on the edge: taking charge of our future. J Bone Joint Surg Am. 2001;83(6):946-950.
3. Tolo VT. The challenges of change: is orthopaedics ready? J Bone Joint Surg Am. 2002;84(9):1707-1713.
4. Herndon JH. One more turn of the wrench. J Bone Joint Surg Am. 2003;85(10):2036-2048.
5. Bucholz RW. Knowledge is our business. J Bone Joint Surg Am. 2004;86(7):1575-1578.
6. Weinstein SL. Nothing about you...without you. J Bone Joint Surg Am. 2005;87(7):1648-1652.
7. Beaty JH. Presidential address: “Building the best . . . Lifelong learning”. J Am Acad Orthop Surg. 2007;15(9):515-518.
8. Rankin EA. Presidential Address: advocacy now... for our patients and our profession. J Am Acad Orthop Surg. 2008;16(6):303-305.
9. Zuckerman JD. Silk purses, sows’ ears, and heap ash—turning challenges into opportunities. J Am Acad Orthop Surg. 2009;17(5):271-275.
10. Tongue JR. Strong on vision, flexible on details. J Am Acad Orthop Surg. 2012;20(4):187-189.
11. Jacobs JJ. Moving forward: from curses to blessings. J Am Acad Orthop Surg. 2013;21(5):261-265.
12. Callaghan JJ. Quality of care: getting from good to great. J Am Acad Orthop Surg. 2010;8(9):516-519.
13. Berry DJ. Informed by our past, building our future. J Am Acad Orthop Surg. 2011;19(4):187-190.
14. Azar FM. Building a bigger box. J Am Acad Orthop Surg. 2014;22(6):341-345.
15. Kyle RF. Presidential Address: Together we are one. J Am Acad Orthop Surg. 2006;14(5):261-264.
16. Vincent GK, Velkoff VA. The Next Four Decades: The Older Population in the United States: 2010 to 2050. Washington, DC: Economics and Statistics Administration, US Census Bureau, US Dept of Commerce; 2010.
17. American Academy of Orthopaedic Surgeons Department of Research and Scientific Affairs. 1998-2011 Resident Diversity Survey Report. American Academy of Orthopaedic Surgeons website. http://www3.aaos.org/about/diversity/pdfs/resident_trend.pdf. Published March 9, 2012. Accessed October 26, 2015.
1. Canale ST. The orthopaedic forum. Falling in love again. J Bone Joint Surg Am. 2000;82(5):739-742.
2. Gelberman RH. The Academy on the edge: taking charge of our future. J Bone Joint Surg Am. 2001;83(6):946-950.
3. Tolo VT. The challenges of change: is orthopaedics ready? J Bone Joint Surg Am. 2002;84(9):1707-1713.
4. Herndon JH. One more turn of the wrench. J Bone Joint Surg Am. 2003;85(10):2036-2048.
5. Bucholz RW. Knowledge is our business. J Bone Joint Surg Am. 2004;86(7):1575-1578.
6. Weinstein SL. Nothing about you...without you. J Bone Joint Surg Am. 2005;87(7):1648-1652.
7. Beaty JH. Presidential address: “Building the best . . . Lifelong learning”. J Am Acad Orthop Surg. 2007;15(9):515-518.
8. Rankin EA. Presidential Address: advocacy now... for our patients and our profession. J Am Acad Orthop Surg. 2008;16(6):303-305.
9. Zuckerman JD. Silk purses, sows’ ears, and heap ash—turning challenges into opportunities. J Am Acad Orthop Surg. 2009;17(5):271-275.
10. Tongue JR. Strong on vision, flexible on details. J Am Acad Orthop Surg. 2012;20(4):187-189.
11. Jacobs JJ. Moving forward: from curses to blessings. J Am Acad Orthop Surg. 2013;21(5):261-265.
12. Callaghan JJ. Quality of care: getting from good to great. J Am Acad Orthop Surg. 2010;8(9):516-519.
13. Berry DJ. Informed by our past, building our future. J Am Acad Orthop Surg. 2011;19(4):187-190.
14. Azar FM. Building a bigger box. J Am Acad Orthop Surg. 2014;22(6):341-345.
15. Kyle RF. Presidential Address: Together we are one. J Am Acad Orthop Surg. 2006;14(5):261-264.
16. Vincent GK, Velkoff VA. The Next Four Decades: The Older Population in the United States: 2010 to 2050. Washington, DC: Economics and Statistics Administration, US Census Bureau, US Dept of Commerce; 2010.
17. American Academy of Orthopaedic Surgeons Department of Research and Scientific Affairs. 1998-2011 Resident Diversity Survey Report. American Academy of Orthopaedic Surgeons website. http://www3.aaos.org/about/diversity/pdfs/resident_trend.pdf. Published March 9, 2012. Accessed October 26, 2015.
Fillers in Dermatology: From Past to Present
Fillers are products that are injected into soft tissue and are classified as either resorbable or nonresorbable (permanent). Several dermal and subcutaneous fillers for soft tissue augmentation currently are available. This article provides a brief review of the history of filler agents currently available for soft tissue augmentation. Cadaveric-derived fillers and implants will not be discussed, as these materials are expensive, are not all approved by the US Food and Drug Administration (FDA), and are more commonly used in burn victims than in cosmetic patients.
History of Fillers in Dermatology
The first known injectable agent was paraffin, but its use as a dermal filler was abandoned after complications including embolization, migration (ie, movement into surrounding tissue), and granuloma formation were reported.1 Silicone also was banned by the FDA for use as a soft tissue filler due to similar complications. Bovine collagen was the first agent to be approved by the FDA for cosmetic injection, and since then many filler agents have revolutionized a new era of cosmetic enhancement.1 Injectable soft tissue fillers now can be classified based on several characteristics, including site of placement (ie, dermal or subcutaneous), animal versus nonanimal derivation (eg, autologous, xenograft, semisynthetic, synthetic), and duration of effects (temporary [<6 months], long-lasting [6 months– 2 years], semipermanent [2–5 years], permanent [>5 years]).2
In the 19th century, early instances of soft tissue augmentation using dermal fillers included autologous fat harvested from the arms for correction of depressed facial defects and scars in a patient with tuberculous osteitis as well as injection of paraffin into the scrotum as a testicular prosthesis in a patient with advanced tuberculosis.1 Later, a technique using a syringe to transfer autologous fat from the extremities was used for facial soft tissue augmentation and contouring, and permanent facial soft tissue augmentation using liquid silicone also was performed. In 1981, purified bovine dermal collagen was first approved by the FDA as a xenogenic agent for dermal injection.3
In 2003, the FDA began to approve new fillers for temporary soft tissue augmentation.3 Approval of injectable purified human collagen derived from fibroblasts was followed by another class of new agents: hyaluronic acid fillers.1 A biodegradable non–animal based stabilized hyaluronic acid was followed by more agents derived from rooster combs. Further investigations and research have continued, and more long-lasting synthetic fillers have become available, including calcium hydroxylapatite and poly-L-lactic acid. A renewed interest in permanent agents and longer lasting products such as silicone oil and polymethyl methacrylate also has emerged.4
The Future of Fillers
While earlier filler materials were limited, those used today are composed of a wide range of substances including collagen, hyaluronic acid, calcium hydroxylapatite, poly-L-lactic acid, and synthetic or manmade polymers (Table). The FDA has approved approximately 21 filler products for dermatologic indications, each with unique properties, advantages, and disadvantages.3 For example, there are several fillers that not only provide soft tissue augmentation but also stimulate collagen production. However, complications can occur, often many years after the initial treatment. Side effects such as swelling, erythema, and nodules may occur and in rare instances foreign body granulomas may develop and may be difficult to eradicate.5 Except for autologous fat, all fillers are foreign bodies and therefore can cause foreign body granulomatous reactions ranging from common (eg, with paraffin) to rare (eg, with hyaluronic acid) in occurrence.6 The clinical presentation of these reactions is variable, ranging from single to multiple nodules at the injection site to diffuse, hard swelling of the face accompanied by reddening of the skin.
With the increasing desire for a youthful appearance among the aging population, the pharmaceutical industry has responded by increasing the number of available treatment options to meet the demands of cosmetic patients, one of the fastest growing subpopulations in the field of dermatology. Fillers also have provided new options for patients who are unable to afford plastic surgery or those who are poor surgical candidates. The ideal filler material is nonallergenic, noncarcinogenic, and nonteratogenic. It should be stable, affordable, malleable, reversible, and durable with results that can be reproduced and should cause minimal inflammation, migration, and detectable changes. The ideal filler also should have predictable and consistent results, feel natural, take little time to administer, require minimal preparation, cause no patient downtime, and have a low risk for complications. Ideal administration should be painless, user-friendly, and conducted in an outpatient setting with minimal recovery and easy storage. Although no single filler is ideal for all patients, indications, and situations, residents should be aware of the properties and characteristics that make each product unique in order to optimize treatment in all patients.
As demands for cosmetic procedures increase, it is important to incorporate knowledge of cosmetic procedures (eg, fillers for soft tissue augmentation) in resident education and training. Although cosmetic dermatology has been featured prominently in dermatology residency, surveys have shown that residents desire more training in this area.7 Although lectures on soft tissue augmentation are popular topics in dermatology, hands-on experience performing these procedures varies widely depending on different training programs. My institution offers several lectures on cosmetic dermatology, and residents are able to perform procedures for soft tissue augmentation as the first assistant or first surgeon during our cosmetic clinic sessions twice weekly.
Final Thoughts
There are a variety of fillers on the horizon to improve aging and volume loss and the science behind cosmetic injections is evolving. Regardless of the filler material chosen, optimal results are yielded by the combination of patient expectations, physician judgment based on clinical experience, and injection technique.
1. Kontis TC, Rivkin A. The history of injectable facial fillers. Facial Plast Surg. 2009;25:67-72.
2. Ashinoff R. Overview: soft tissue augmentation. Clin Plast Surg. 2000;27:479-487.
3. Soft tissue fillers approved by the Center for Devices and Radiological Health. US Food and Drug Administration Web site. http://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/CosmeticDevices/WrinkleFillers/ucm227749.htm. Updated July 27, 2015. Accessed November 16, 2015.
4. Rohrich RJ, Ghavami A, Crosby MA. The role of hyaluronic acid fillers (restylane) in facial cosmetic surgery: review and technical considerations. Plast Reconstr Surg. 2007;120(suppl 6):S41-S54.
5. Braun M, Braun S. Nodule formation following lip augmentation using porcine collagen-derived filler. J Drugs Dermatol. 2008;7:579-581.
6. Lee JM, Kim YJ. Foreign body granulomas after the use of dermal fillers: pathophysiology, clinical appearance, histologic features, and treatment. Arch Plast Surg. 2015;42:232-239.
7. Kirby JS, Adgerson CN, Anderson BE. A survey of dermatology resident education in cosmetic procedures. J Am Acad Dermatol 2013;68:e23-e28.
Fillers are products that are injected into soft tissue and are classified as either resorbable or nonresorbable (permanent). Several dermal and subcutaneous fillers for soft tissue augmentation currently are available. This article provides a brief review of the history of filler agents currently available for soft tissue augmentation. Cadaveric-derived fillers and implants will not be discussed, as these materials are expensive, are not all approved by the US Food and Drug Administration (FDA), and are more commonly used in burn victims than in cosmetic patients.
History of Fillers in Dermatology
The first known injectable agent was paraffin, but its use as a dermal filler was abandoned after complications including embolization, migration (ie, movement into surrounding tissue), and granuloma formation were reported.1 Silicone also was banned by the FDA for use as a soft tissue filler due to similar complications. Bovine collagen was the first agent to be approved by the FDA for cosmetic injection, and since then many filler agents have revolutionized a new era of cosmetic enhancement.1 Injectable soft tissue fillers now can be classified based on several characteristics, including site of placement (ie, dermal or subcutaneous), animal versus nonanimal derivation (eg, autologous, xenograft, semisynthetic, synthetic), and duration of effects (temporary [<6 months], long-lasting [6 months– 2 years], semipermanent [2–5 years], permanent [>5 years]).2
In the 19th century, early instances of soft tissue augmentation using dermal fillers included autologous fat harvested from the arms for correction of depressed facial defects and scars in a patient with tuberculous osteitis as well as injection of paraffin into the scrotum as a testicular prosthesis in a patient with advanced tuberculosis.1 Later, a technique using a syringe to transfer autologous fat from the extremities was used for facial soft tissue augmentation and contouring, and permanent facial soft tissue augmentation using liquid silicone also was performed. In 1981, purified bovine dermal collagen was first approved by the FDA as a xenogenic agent for dermal injection.3
In 2003, the FDA began to approve new fillers for temporary soft tissue augmentation.3 Approval of injectable purified human collagen derived from fibroblasts was followed by another class of new agents: hyaluronic acid fillers.1 A biodegradable non–animal based stabilized hyaluronic acid was followed by more agents derived from rooster combs. Further investigations and research have continued, and more long-lasting synthetic fillers have become available, including calcium hydroxylapatite and poly-L-lactic acid. A renewed interest in permanent agents and longer lasting products such as silicone oil and polymethyl methacrylate also has emerged.4
The Future of Fillers
While earlier filler materials were limited, those used today are composed of a wide range of substances including collagen, hyaluronic acid, calcium hydroxylapatite, poly-L-lactic acid, and synthetic or manmade polymers (Table). The FDA has approved approximately 21 filler products for dermatologic indications, each with unique properties, advantages, and disadvantages.3 For example, there are several fillers that not only provide soft tissue augmentation but also stimulate collagen production. However, complications can occur, often many years after the initial treatment. Side effects such as swelling, erythema, and nodules may occur and in rare instances foreign body granulomas may develop and may be difficult to eradicate.5 Except for autologous fat, all fillers are foreign bodies and therefore can cause foreign body granulomatous reactions ranging from common (eg, with paraffin) to rare (eg, with hyaluronic acid) in occurrence.6 The clinical presentation of these reactions is variable, ranging from single to multiple nodules at the injection site to diffuse, hard swelling of the face accompanied by reddening of the skin.
With the increasing desire for a youthful appearance among the aging population, the pharmaceutical industry has responded by increasing the number of available treatment options to meet the demands of cosmetic patients, one of the fastest growing subpopulations in the field of dermatology. Fillers also have provided new options for patients who are unable to afford plastic surgery or those who are poor surgical candidates. The ideal filler material is nonallergenic, noncarcinogenic, and nonteratogenic. It should be stable, affordable, malleable, reversible, and durable with results that can be reproduced and should cause minimal inflammation, migration, and detectable changes. The ideal filler also should have predictable and consistent results, feel natural, take little time to administer, require minimal preparation, cause no patient downtime, and have a low risk for complications. Ideal administration should be painless, user-friendly, and conducted in an outpatient setting with minimal recovery and easy storage. Although no single filler is ideal for all patients, indications, and situations, residents should be aware of the properties and characteristics that make each product unique in order to optimize treatment in all patients.
As demands for cosmetic procedures increase, it is important to incorporate knowledge of cosmetic procedures (eg, fillers for soft tissue augmentation) in resident education and training. Although cosmetic dermatology has been featured prominently in dermatology residency, surveys have shown that residents desire more training in this area.7 Although lectures on soft tissue augmentation are popular topics in dermatology, hands-on experience performing these procedures varies widely depending on different training programs. My institution offers several lectures on cosmetic dermatology, and residents are able to perform procedures for soft tissue augmentation as the first assistant or first surgeon during our cosmetic clinic sessions twice weekly.
Final Thoughts
There are a variety of fillers on the horizon to improve aging and volume loss and the science behind cosmetic injections is evolving. Regardless of the filler material chosen, optimal results are yielded by the combination of patient expectations, physician judgment based on clinical experience, and injection technique.
Fillers are products that are injected into soft tissue and are classified as either resorbable or nonresorbable (permanent). Several dermal and subcutaneous fillers for soft tissue augmentation currently are available. This article provides a brief review of the history of filler agents currently available for soft tissue augmentation. Cadaveric-derived fillers and implants will not be discussed, as these materials are expensive, are not all approved by the US Food and Drug Administration (FDA), and are more commonly used in burn victims than in cosmetic patients.
History of Fillers in Dermatology
The first known injectable agent was paraffin, but its use as a dermal filler was abandoned after complications including embolization, migration (ie, movement into surrounding tissue), and granuloma formation were reported.1 Silicone also was banned by the FDA for use as a soft tissue filler due to similar complications. Bovine collagen was the first agent to be approved by the FDA for cosmetic injection, and since then many filler agents have revolutionized a new era of cosmetic enhancement.1 Injectable soft tissue fillers now can be classified based on several characteristics, including site of placement (ie, dermal or subcutaneous), animal versus nonanimal derivation (eg, autologous, xenograft, semisynthetic, synthetic), and duration of effects (temporary [<6 months], long-lasting [6 months– 2 years], semipermanent [2–5 years], permanent [>5 years]).2
In the 19th century, early instances of soft tissue augmentation using dermal fillers included autologous fat harvested from the arms for correction of depressed facial defects and scars in a patient with tuberculous osteitis as well as injection of paraffin into the scrotum as a testicular prosthesis in a patient with advanced tuberculosis.1 Later, a technique using a syringe to transfer autologous fat from the extremities was used for facial soft tissue augmentation and contouring, and permanent facial soft tissue augmentation using liquid silicone also was performed. In 1981, purified bovine dermal collagen was first approved by the FDA as a xenogenic agent for dermal injection.3
In 2003, the FDA began to approve new fillers for temporary soft tissue augmentation.3 Approval of injectable purified human collagen derived from fibroblasts was followed by another class of new agents: hyaluronic acid fillers.1 A biodegradable non–animal based stabilized hyaluronic acid was followed by more agents derived from rooster combs. Further investigations and research have continued, and more long-lasting synthetic fillers have become available, including calcium hydroxylapatite and poly-L-lactic acid. A renewed interest in permanent agents and longer lasting products such as silicone oil and polymethyl methacrylate also has emerged.4
The Future of Fillers
While earlier filler materials were limited, those used today are composed of a wide range of substances including collagen, hyaluronic acid, calcium hydroxylapatite, poly-L-lactic acid, and synthetic or manmade polymers (Table). The FDA has approved approximately 21 filler products for dermatologic indications, each with unique properties, advantages, and disadvantages.3 For example, there are several fillers that not only provide soft tissue augmentation but also stimulate collagen production. However, complications can occur, often many years after the initial treatment. Side effects such as swelling, erythema, and nodules may occur and in rare instances foreign body granulomas may develop and may be difficult to eradicate.5 Except for autologous fat, all fillers are foreign bodies and therefore can cause foreign body granulomatous reactions ranging from common (eg, with paraffin) to rare (eg, with hyaluronic acid) in occurrence.6 The clinical presentation of these reactions is variable, ranging from single to multiple nodules at the injection site to diffuse, hard swelling of the face accompanied by reddening of the skin.
With the increasing desire for a youthful appearance among the aging population, the pharmaceutical industry has responded by increasing the number of available treatment options to meet the demands of cosmetic patients, one of the fastest growing subpopulations in the field of dermatology. Fillers also have provided new options for patients who are unable to afford plastic surgery or those who are poor surgical candidates. The ideal filler material is nonallergenic, noncarcinogenic, and nonteratogenic. It should be stable, affordable, malleable, reversible, and durable with results that can be reproduced and should cause minimal inflammation, migration, and detectable changes. The ideal filler also should have predictable and consistent results, feel natural, take little time to administer, require minimal preparation, cause no patient downtime, and have a low risk for complications. Ideal administration should be painless, user-friendly, and conducted in an outpatient setting with minimal recovery and easy storage. Although no single filler is ideal for all patients, indications, and situations, residents should be aware of the properties and characteristics that make each product unique in order to optimize treatment in all patients.
As demands for cosmetic procedures increase, it is important to incorporate knowledge of cosmetic procedures (eg, fillers for soft tissue augmentation) in resident education and training. Although cosmetic dermatology has been featured prominently in dermatology residency, surveys have shown that residents desire more training in this area.7 Although lectures on soft tissue augmentation are popular topics in dermatology, hands-on experience performing these procedures varies widely depending on different training programs. My institution offers several lectures on cosmetic dermatology, and residents are able to perform procedures for soft tissue augmentation as the first assistant or first surgeon during our cosmetic clinic sessions twice weekly.
Final Thoughts
There are a variety of fillers on the horizon to improve aging and volume loss and the science behind cosmetic injections is evolving. Regardless of the filler material chosen, optimal results are yielded by the combination of patient expectations, physician judgment based on clinical experience, and injection technique.
1. Kontis TC, Rivkin A. The history of injectable facial fillers. Facial Plast Surg. 2009;25:67-72.
2. Ashinoff R. Overview: soft tissue augmentation. Clin Plast Surg. 2000;27:479-487.
3. Soft tissue fillers approved by the Center for Devices and Radiological Health. US Food and Drug Administration Web site. http://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/CosmeticDevices/WrinkleFillers/ucm227749.htm. Updated July 27, 2015. Accessed November 16, 2015.
4. Rohrich RJ, Ghavami A, Crosby MA. The role of hyaluronic acid fillers (restylane) in facial cosmetic surgery: review and technical considerations. Plast Reconstr Surg. 2007;120(suppl 6):S41-S54.
5. Braun M, Braun S. Nodule formation following lip augmentation using porcine collagen-derived filler. J Drugs Dermatol. 2008;7:579-581.
6. Lee JM, Kim YJ. Foreign body granulomas after the use of dermal fillers: pathophysiology, clinical appearance, histologic features, and treatment. Arch Plast Surg. 2015;42:232-239.
7. Kirby JS, Adgerson CN, Anderson BE. A survey of dermatology resident education in cosmetic procedures. J Am Acad Dermatol 2013;68:e23-e28.
1. Kontis TC, Rivkin A. The history of injectable facial fillers. Facial Plast Surg. 2009;25:67-72.
2. Ashinoff R. Overview: soft tissue augmentation. Clin Plast Surg. 2000;27:479-487.
3. Soft tissue fillers approved by the Center for Devices and Radiological Health. US Food and Drug Administration Web site. http://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/CosmeticDevices/WrinkleFillers/ucm227749.htm. Updated July 27, 2015. Accessed November 16, 2015.
4. Rohrich RJ, Ghavami A, Crosby MA. The role of hyaluronic acid fillers (restylane) in facial cosmetic surgery: review and technical considerations. Plast Reconstr Surg. 2007;120(suppl 6):S41-S54.
5. Braun M, Braun S. Nodule formation following lip augmentation using porcine collagen-derived filler. J Drugs Dermatol. 2008;7:579-581.
6. Lee JM, Kim YJ. Foreign body granulomas after the use of dermal fillers: pathophysiology, clinical appearance, histologic features, and treatment. Arch Plast Surg. 2015;42:232-239.
7. Kirby JS, Adgerson CN, Anderson BE. A survey of dermatology resident education in cosmetic procedures. J Am Acad Dermatol 2013;68:e23-e28.
Resident Involvement in Policy-Making
As dermatology residents, we hear rumblings of the political aspects of medicine on the news, at conferences, or from our attending physicians. Most of us conveniently ignore them until after we graduate; however, once we start practicing, we may be shocked to discover just how much politics affect the practice of medicine. In this article, the role of the American Academy of Dermatology Association (AADA) in policy-making is discussed as well as some ways residents can be involved in the process and emphasize just how vital our participation is, even this early on in our careers.
Role of the AADA in Policy-Making
The AADA advocates on behalf of its members and dermatology patients with the US Congress, state legislatures, and regulatory agencies on issues of concern to the specialty and offers members several opportunities to become involved in advocacy at the state, national, and grassroots levels.1 The AAD provides several sources of information on the Web, including the Dermatology Advocacy Network (http://www.aad-dan.com/), which includes information on how to contact your member of Congress through the AADA; updates on activities and topics discussed at the AADA Legislative Conference (https://www.aad.org/meetings/legislative -conference) and information on how to participate in future conferences; resources on getting involved in advocacy at the grassroots level (https://www.aad.org /members/practice-and-advocacy/get-involved /grassroots-advocacy); and information on how to become involved with SkinPAC (https://www.skinpac.org/), the AADA’s political action committee (PAC). A PAC is organized for the purpose of advancing a particular legislative agenda, whatever the issue may be. SkinPAC ensures that the concerns of dermatologists and their patients are heard on Capitol Hill.
Opportunities for Resident Involvement
There are several ways residents can be involved in policy-making, including (1) attending the annual AADA Legislative Conference, which offers participants a unique opportunity to attend advocacy training sessions taught by health policy experts, discuss dermatology issues with colleagues, and become trusted and influential voices that members of Congress can rely on,1 (2) sending a letter to your state or federal officials through the AADA Dermatology Advocacy Network, (3) becoming a state advocacy leader, especially through your state’s PAC, and (4) reading the AAD’s bi-weekly Dermatology Advocate e-newsletter (https://www.aad.org /members/publications/dermatology-advocate), which includes information on congressional actions, federal agency and administration activities, state-level legislative and regulatory news, actions by private payers, and the AADA’s active engagement in these arenas. Also featured in the Dermatology Advocate e-newsletter is news and information about how members can get involved in advocacy efforts by the AADA and SkinPAC.
AADA Legislative Conference
The 2015 AADA Legislative Conference took place in September in Washington, DC. This conference offers a unique opportunity for residents to learn how federal legislation will impact the future of dermatology. The AADA awards several scholarships to residents who commit to one year of involvement in AADA grassroots advocacy.2 The AADA covers all expenses to attend the conference for each scholarship recipient, and residents are not required to have any political knowledge or experience in order to attend the conference or receive a scholarship. Advocacy training, which is offered by a panel of health policy experts, covers all aspects of the legislative process as well as information about the legislators themselves.
In addition to the opportunities to get involved on a national level through the AADA, most states also have their own PACs with which physicians can work on grassroots-level issues, such as advocating for state laws prohibiting minors under 18 years of age from using indoor tanning beds, or larger issues including the Medicare sustainable growth rate or global period codes. Additionally, some subspecialties also have their own advocacy groups, including the American Society for Dermatologic Surgery Association’s State-based Advocacy Network for Dermatology Surgery (http://asdsa.asds.net/ResidentStateAdvocacy.aspx).
The Importance and Influence of Involvement in Policy-Making
It has been shown that many residents are in fact interested in joining PACs that are relevant to their specialty after learning about the roles these committees play in policy-making but that time constraints and obligations of residency often interfere with their participation.3 As residents, involvement by our attending physicians plays a huge role. A recent study showed more resident involvement when faculty members set an example of civic involvement and PAC support.3
I was inspired to write this column by my residency program’s impressive involvement with SkinPAC last year as well as my own personal experience being an active advocate. During medical school, I helped testify for the Texas State Senate Committee on Health and Human Services in Austin about the risks of indoor tanning and witnessed the eventual ban on indoor tanning use by minors in the state of Texas, which came as a direct result of our advocacy and push for change. I used the University of Texas Medical Branch Dermatology Interest blog (of which I was the Editor during medical school) to help educate others on this issue, lay down the facts to be discussed in an organized and powerful way, and provide contact information for state legislators (http://digutmb.blogspot.com/2013/05 /act-now-tell-governor-perry-to-support.html).
Final Thoughts
As dermatology residents, a substantial amount of what we do in the routine care of our patients is influenced by policy decisions made by legislators at the state and federal levels, who often do not understand the impact their decisions have on our ability to effectively practice medicine. Learning about these issues now and becoming involved in efforts to affect change is something every resident can do, so get active!
Acknowledgement—I would like to thank the faculty members of the Department of Dermatology and Cutaneous Surgery at the University of South Florida, Tampa, for their active involvement in SkinPAC and the AADA Legislative Conference.
1. Get involved: AADA advocacy. American Academy of Dermatology Web site. https://www.aad.org /members/practice-and-advocacy//get-involved. Accessed September 21, 2015.
2. Resident scholarship to legislative conference. American Academy of Dermatology Web site. https://www.aad.org /education/awards-grants-and-scholarships/resident -scholarship-to-legislative-conference. Accessed October 19, 2015.
3. Shah RP, Froelich, JM, Weinstein SL, et al. Factors influencing resident participation in the AAOS Political Action Committee. Orthopedics. 2013;36:826-830.
As dermatology residents, we hear rumblings of the political aspects of medicine on the news, at conferences, or from our attending physicians. Most of us conveniently ignore them until after we graduate; however, once we start practicing, we may be shocked to discover just how much politics affect the practice of medicine. In this article, the role of the American Academy of Dermatology Association (AADA) in policy-making is discussed as well as some ways residents can be involved in the process and emphasize just how vital our participation is, even this early on in our careers.
Role of the AADA in Policy-Making
The AADA advocates on behalf of its members and dermatology patients with the US Congress, state legislatures, and regulatory agencies on issues of concern to the specialty and offers members several opportunities to become involved in advocacy at the state, national, and grassroots levels.1 The AAD provides several sources of information on the Web, including the Dermatology Advocacy Network (http://www.aad-dan.com/), which includes information on how to contact your member of Congress through the AADA; updates on activities and topics discussed at the AADA Legislative Conference (https://www.aad.org/meetings/legislative -conference) and information on how to participate in future conferences; resources on getting involved in advocacy at the grassroots level (https://www.aad.org /members/practice-and-advocacy/get-involved /grassroots-advocacy); and information on how to become involved with SkinPAC (https://www.skinpac.org/), the AADA’s political action committee (PAC). A PAC is organized for the purpose of advancing a particular legislative agenda, whatever the issue may be. SkinPAC ensures that the concerns of dermatologists and their patients are heard on Capitol Hill.
Opportunities for Resident Involvement
There are several ways residents can be involved in policy-making, including (1) attending the annual AADA Legislative Conference, which offers participants a unique opportunity to attend advocacy training sessions taught by health policy experts, discuss dermatology issues with colleagues, and become trusted and influential voices that members of Congress can rely on,1 (2) sending a letter to your state or federal officials through the AADA Dermatology Advocacy Network, (3) becoming a state advocacy leader, especially through your state’s PAC, and (4) reading the AAD’s bi-weekly Dermatology Advocate e-newsletter (https://www.aad.org /members/publications/dermatology-advocate), which includes information on congressional actions, federal agency and administration activities, state-level legislative and regulatory news, actions by private payers, and the AADA’s active engagement in these arenas. Also featured in the Dermatology Advocate e-newsletter is news and information about how members can get involved in advocacy efforts by the AADA and SkinPAC.
AADA Legislative Conference
The 2015 AADA Legislative Conference took place in September in Washington, DC. This conference offers a unique opportunity for residents to learn how federal legislation will impact the future of dermatology. The AADA awards several scholarships to residents who commit to one year of involvement in AADA grassroots advocacy.2 The AADA covers all expenses to attend the conference for each scholarship recipient, and residents are not required to have any political knowledge or experience in order to attend the conference or receive a scholarship. Advocacy training, which is offered by a panel of health policy experts, covers all aspects of the legislative process as well as information about the legislators themselves.
In addition to the opportunities to get involved on a national level through the AADA, most states also have their own PACs with which physicians can work on grassroots-level issues, such as advocating for state laws prohibiting minors under 18 years of age from using indoor tanning beds, or larger issues including the Medicare sustainable growth rate or global period codes. Additionally, some subspecialties also have their own advocacy groups, including the American Society for Dermatologic Surgery Association’s State-based Advocacy Network for Dermatology Surgery (http://asdsa.asds.net/ResidentStateAdvocacy.aspx).
The Importance and Influence of Involvement in Policy-Making
It has been shown that many residents are in fact interested in joining PACs that are relevant to their specialty after learning about the roles these committees play in policy-making but that time constraints and obligations of residency often interfere with their participation.3 As residents, involvement by our attending physicians plays a huge role. A recent study showed more resident involvement when faculty members set an example of civic involvement and PAC support.3
I was inspired to write this column by my residency program’s impressive involvement with SkinPAC last year as well as my own personal experience being an active advocate. During medical school, I helped testify for the Texas State Senate Committee on Health and Human Services in Austin about the risks of indoor tanning and witnessed the eventual ban on indoor tanning use by minors in the state of Texas, which came as a direct result of our advocacy and push for change. I used the University of Texas Medical Branch Dermatology Interest blog (of which I was the Editor during medical school) to help educate others on this issue, lay down the facts to be discussed in an organized and powerful way, and provide contact information for state legislators (http://digutmb.blogspot.com/2013/05 /act-now-tell-governor-perry-to-support.html).
Final Thoughts
As dermatology residents, a substantial amount of what we do in the routine care of our patients is influenced by policy decisions made by legislators at the state and federal levels, who often do not understand the impact their decisions have on our ability to effectively practice medicine. Learning about these issues now and becoming involved in efforts to affect change is something every resident can do, so get active!
Acknowledgement—I would like to thank the faculty members of the Department of Dermatology and Cutaneous Surgery at the University of South Florida, Tampa, for their active involvement in SkinPAC and the AADA Legislative Conference.
As dermatology residents, we hear rumblings of the political aspects of medicine on the news, at conferences, or from our attending physicians. Most of us conveniently ignore them until after we graduate; however, once we start practicing, we may be shocked to discover just how much politics affect the practice of medicine. In this article, the role of the American Academy of Dermatology Association (AADA) in policy-making is discussed as well as some ways residents can be involved in the process and emphasize just how vital our participation is, even this early on in our careers.
Role of the AADA in Policy-Making
The AADA advocates on behalf of its members and dermatology patients with the US Congress, state legislatures, and regulatory agencies on issues of concern to the specialty and offers members several opportunities to become involved in advocacy at the state, national, and grassroots levels.1 The AAD provides several sources of information on the Web, including the Dermatology Advocacy Network (http://www.aad-dan.com/), which includes information on how to contact your member of Congress through the AADA; updates on activities and topics discussed at the AADA Legislative Conference (https://www.aad.org/meetings/legislative -conference) and information on how to participate in future conferences; resources on getting involved in advocacy at the grassroots level (https://www.aad.org /members/practice-and-advocacy/get-involved /grassroots-advocacy); and information on how to become involved with SkinPAC (https://www.skinpac.org/), the AADA’s political action committee (PAC). A PAC is organized for the purpose of advancing a particular legislative agenda, whatever the issue may be. SkinPAC ensures that the concerns of dermatologists and their patients are heard on Capitol Hill.
Opportunities for Resident Involvement
There are several ways residents can be involved in policy-making, including (1) attending the annual AADA Legislative Conference, which offers participants a unique opportunity to attend advocacy training sessions taught by health policy experts, discuss dermatology issues with colleagues, and become trusted and influential voices that members of Congress can rely on,1 (2) sending a letter to your state or federal officials through the AADA Dermatology Advocacy Network, (3) becoming a state advocacy leader, especially through your state’s PAC, and (4) reading the AAD’s bi-weekly Dermatology Advocate e-newsletter (https://www.aad.org /members/publications/dermatology-advocate), which includes information on congressional actions, federal agency and administration activities, state-level legislative and regulatory news, actions by private payers, and the AADA’s active engagement in these arenas. Also featured in the Dermatology Advocate e-newsletter is news and information about how members can get involved in advocacy efforts by the AADA and SkinPAC.
AADA Legislative Conference
The 2015 AADA Legislative Conference took place in September in Washington, DC. This conference offers a unique opportunity for residents to learn how federal legislation will impact the future of dermatology. The AADA awards several scholarships to residents who commit to one year of involvement in AADA grassroots advocacy.2 The AADA covers all expenses to attend the conference for each scholarship recipient, and residents are not required to have any political knowledge or experience in order to attend the conference or receive a scholarship. Advocacy training, which is offered by a panel of health policy experts, covers all aspects of the legislative process as well as information about the legislators themselves.
In addition to the opportunities to get involved on a national level through the AADA, most states also have their own PACs with which physicians can work on grassroots-level issues, such as advocating for state laws prohibiting minors under 18 years of age from using indoor tanning beds, or larger issues including the Medicare sustainable growth rate or global period codes. Additionally, some subspecialties also have their own advocacy groups, including the American Society for Dermatologic Surgery Association’s State-based Advocacy Network for Dermatology Surgery (http://asdsa.asds.net/ResidentStateAdvocacy.aspx).
The Importance and Influence of Involvement in Policy-Making
It has been shown that many residents are in fact interested in joining PACs that are relevant to their specialty after learning about the roles these committees play in policy-making but that time constraints and obligations of residency often interfere with their participation.3 As residents, involvement by our attending physicians plays a huge role. A recent study showed more resident involvement when faculty members set an example of civic involvement and PAC support.3
I was inspired to write this column by my residency program’s impressive involvement with SkinPAC last year as well as my own personal experience being an active advocate. During medical school, I helped testify for the Texas State Senate Committee on Health and Human Services in Austin about the risks of indoor tanning and witnessed the eventual ban on indoor tanning use by minors in the state of Texas, which came as a direct result of our advocacy and push for change. I used the University of Texas Medical Branch Dermatology Interest blog (of which I was the Editor during medical school) to help educate others on this issue, lay down the facts to be discussed in an organized and powerful way, and provide contact information for state legislators (http://digutmb.blogspot.com/2013/05 /act-now-tell-governor-perry-to-support.html).
Final Thoughts
As dermatology residents, a substantial amount of what we do in the routine care of our patients is influenced by policy decisions made by legislators at the state and federal levels, who often do not understand the impact their decisions have on our ability to effectively practice medicine. Learning about these issues now and becoming involved in efforts to affect change is something every resident can do, so get active!
Acknowledgement—I would like to thank the faculty members of the Department of Dermatology and Cutaneous Surgery at the University of South Florida, Tampa, for their active involvement in SkinPAC and the AADA Legislative Conference.
1. Get involved: AADA advocacy. American Academy of Dermatology Web site. https://www.aad.org /members/practice-and-advocacy//get-involved. Accessed September 21, 2015.
2. Resident scholarship to legislative conference. American Academy of Dermatology Web site. https://www.aad.org /education/awards-grants-and-scholarships/resident -scholarship-to-legislative-conference. Accessed October 19, 2015.
3. Shah RP, Froelich, JM, Weinstein SL, et al. Factors influencing resident participation in the AAOS Political Action Committee. Orthopedics. 2013;36:826-830.
1. Get involved: AADA advocacy. American Academy of Dermatology Web site. https://www.aad.org /members/practice-and-advocacy//get-involved. Accessed September 21, 2015.
2. Resident scholarship to legislative conference. American Academy of Dermatology Web site. https://www.aad.org /education/awards-grants-and-scholarships/resident -scholarship-to-legislative-conference. Accessed October 19, 2015.
3. Shah RP, Froelich, JM, Weinstein SL, et al. Factors influencing resident participation in the AAOS Political Action Committee. Orthopedics. 2013;36:826-830.