User login
Career Choices: Consultation-liaison psychiatry
Editor’s note: Career Choices features a psychiatry resident/fellow interviewing a psychiatrist about why he (she) has chosen a specific career path. The goal is to inform trainees about the various psychiatric career options, and to give them a feel for the pros and cons of the various paths.
In this Career Choices, Cornel Stanciu, MD, talked with Peter Ganpat, MD, a consultation-liaison (C-L) psychiatrist at Florida Hospital, where he provides guidance to various medical specialties on managing acute and chronic mental illness and substance use disorders. In addition, he also is the medical director for the repetitive transcranial magnetic stimulation service and staffs the inpatient unit.
Dr. Stanciu: What made you choose to become a C-L psychiatrist?
Dr. Ganpat: In my opinion, C-L is the most challenging area of psychiatry because not only are you thinking along the realms of a psychiatrist, but you’re also considering the viewpoint of the other subspecialties at the same time. For me, it brings together my medical background with my passion for psychiatry, and the patients I see daily allow for this incorporation.
Dr. Stanciu: How did your career path prepare you to become a C-L psychiatrist?
Dr. Ganpat: My career path was unique in that I completed a family medicine residency, and then immediately pursued training in psychiatry. Some may consider this as “overkill” for C-L, but as I’ve come to learn, this background grants me a level of understanding and confidence to step in when dealing with a complex case and lend a hand to the consulting physician beyond psychiatry. I do not feel a fellowship is required to practice C-L psychiatry. However, a psychosomatic fellowship will definitely provide the experience needed for this career path, and also will enable one to get a second American Board of Psychiatry and Neurology board certification.
Dr. Stanciu: What types of clinical conditions are you asked to provide input on managing, and how do you find working alongside other specialties?
Dr. Ganpat: I have been managing the full breadth of psychiatry, and in some cases I also provide medical management. Practicing in a metropolitan area with a high influx of tourists also brings in unique cultural cases. The level of respect that the other specialties give is impressive, because they have now seen what a C-L psychiatrist can do. Their performance scores also have improved as a result of my involvement. They greatly appreciate my efforts to shed light on cases or assist with the ever-challenging patient whose psychiatric complexity impedes care.
Dr. Stanciu: How would you describe a physician who is well-suited for such a setting?
Dr. Ganpat: The perfect candidate for this role should be capable of abstract as well as objective thinking. Having a good understanding of the other medical specialties and being able to solve problems is essential, because often it isn’t a clear-cut picture. It is imperative for the C-L psychiatrist to have sound teaching abilities and to be able to educate and communicate his (her) reasoning to the consulting team. It also is important to be well-versed in the psychiatric manifestations of various medical disorders and the psychiatric iatrogenesis of widely used prescription medications.
Dr. Stanciu: What challenges and surprises did you encounter when you first began to practice in this setting?
Dr. Ganpat: I think the largest challenge that I have encountered is the lack of resources. Substance abuse is a major problem here, especially opioids, and there are limited community resources for these patients, so they wind up in the hospital.
Dr. Stanciu: What are the disadvantages of C-L compared with other branches of psychiatry?
Dr. Ganpat: There isn’t much continuity of care with C-L psychiatry over the long run, but you do get to see patients improve during the duration of their hospitalization, which is very rewarding.
Dr. Stanciu: What is the typical reimbursement model for a C-L psychiatrist, and have you run into difficulties with insurance providers in this setting?
Dr. Ganpat: The reimbursement model varies from one system to the next. The common model is to bill just as any other hospital service would, based on the time or level of complexity. Obviously, the more consults you have, the more billing is generated. Most insurance carriers recognize this and so I haven’t had much of an issue with reimbursement, although some unexpected problems may arise.
Dr. Stanciu: What advice do you have for early career psychiatrists and trainees who are contemplating a C-L career?
Dr. Ganpat: If you enjoy working in the hospital and interfacing with other specialties, then consider C-L psychiatry. It is challenging but intellectually stimulating. Make sure you request a C-L rotation during your training, because the Accreditation Council for Graduate Medical Education requires it during a psychiatric residency.
Dr. Stanciu: What is the future outlook of C-L?
Dr. Ganpat: There is a shortage of C-L psychiatrists because >50% of practicing psychiatrists are in private practice in an outpatient setting. Because access to psychiatric care outside of a hospital setting is an issue, and much care is being driven to hospitals, there will be an increasing need for C-L psychiatrists.
Editor’s note: Career Choices features a psychiatry resident/fellow interviewing a psychiatrist about why he (she) has chosen a specific career path. The goal is to inform trainees about the various psychiatric career options, and to give them a feel for the pros and cons of the various paths.
In this Career Choices, Cornel Stanciu, MD, talked with Peter Ganpat, MD, a consultation-liaison (C-L) psychiatrist at Florida Hospital, where he provides guidance to various medical specialties on managing acute and chronic mental illness and substance use disorders. In addition, he also is the medical director for the repetitive transcranial magnetic stimulation service and staffs the inpatient unit.
Dr. Stanciu: What made you choose to become a C-L psychiatrist?
Dr. Ganpat: In my opinion, C-L is the most challenging area of psychiatry because not only are you thinking along the realms of a psychiatrist, but you’re also considering the viewpoint of the other subspecialties at the same time. For me, it brings together my medical background with my passion for psychiatry, and the patients I see daily allow for this incorporation.
Dr. Stanciu: How did your career path prepare you to become a C-L psychiatrist?
Dr. Ganpat: My career path was unique in that I completed a family medicine residency, and then immediately pursued training in psychiatry. Some may consider this as “overkill” for C-L, but as I’ve come to learn, this background grants me a level of understanding and confidence to step in when dealing with a complex case and lend a hand to the consulting physician beyond psychiatry. I do not feel a fellowship is required to practice C-L psychiatry. However, a psychosomatic fellowship will definitely provide the experience needed for this career path, and also will enable one to get a second American Board of Psychiatry and Neurology board certification.
Dr. Stanciu: What types of clinical conditions are you asked to provide input on managing, and how do you find working alongside other specialties?
Dr. Ganpat: I have been managing the full breadth of psychiatry, and in some cases I also provide medical management. Practicing in a metropolitan area with a high influx of tourists also brings in unique cultural cases. The level of respect that the other specialties give is impressive, because they have now seen what a C-L psychiatrist can do. Their performance scores also have improved as a result of my involvement. They greatly appreciate my efforts to shed light on cases or assist with the ever-challenging patient whose psychiatric complexity impedes care.
Dr. Stanciu: How would you describe a physician who is well-suited for such a setting?
Dr. Ganpat: The perfect candidate for this role should be capable of abstract as well as objective thinking. Having a good understanding of the other medical specialties and being able to solve problems is essential, because often it isn’t a clear-cut picture. It is imperative for the C-L psychiatrist to have sound teaching abilities and to be able to educate and communicate his (her) reasoning to the consulting team. It also is important to be well-versed in the psychiatric manifestations of various medical disorders and the psychiatric iatrogenesis of widely used prescription medications.
Dr. Stanciu: What challenges and surprises did you encounter when you first began to practice in this setting?
Dr. Ganpat: I think the largest challenge that I have encountered is the lack of resources. Substance abuse is a major problem here, especially opioids, and there are limited community resources for these patients, so they wind up in the hospital.
Dr. Stanciu: What are the disadvantages of C-L compared with other branches of psychiatry?
Dr. Ganpat: There isn’t much continuity of care with C-L psychiatry over the long run, but you do get to see patients improve during the duration of their hospitalization, which is very rewarding.
Dr. Stanciu: What is the typical reimbursement model for a C-L psychiatrist, and have you run into difficulties with insurance providers in this setting?
Dr. Ganpat: The reimbursement model varies from one system to the next. The common model is to bill just as any other hospital service would, based on the time or level of complexity. Obviously, the more consults you have, the more billing is generated. Most insurance carriers recognize this and so I haven’t had much of an issue with reimbursement, although some unexpected problems may arise.
Dr. Stanciu: What advice do you have for early career psychiatrists and trainees who are contemplating a C-L career?
Dr. Ganpat: If you enjoy working in the hospital and interfacing with other specialties, then consider C-L psychiatry. It is challenging but intellectually stimulating. Make sure you request a C-L rotation during your training, because the Accreditation Council for Graduate Medical Education requires it during a psychiatric residency.
Dr. Stanciu: What is the future outlook of C-L?
Dr. Ganpat: There is a shortage of C-L psychiatrists because >50% of practicing psychiatrists are in private practice in an outpatient setting. Because access to psychiatric care outside of a hospital setting is an issue, and much care is being driven to hospitals, there will be an increasing need for C-L psychiatrists.
Editor’s note: Career Choices features a psychiatry resident/fellow interviewing a psychiatrist about why he (she) has chosen a specific career path. The goal is to inform trainees about the various psychiatric career options, and to give them a feel for the pros and cons of the various paths.
In this Career Choices, Cornel Stanciu, MD, talked with Peter Ganpat, MD, a consultation-liaison (C-L) psychiatrist at Florida Hospital, where he provides guidance to various medical specialties on managing acute and chronic mental illness and substance use disorders. In addition, he also is the medical director for the repetitive transcranial magnetic stimulation service and staffs the inpatient unit.
Dr. Stanciu: What made you choose to become a C-L psychiatrist?
Dr. Ganpat: In my opinion, C-L is the most challenging area of psychiatry because not only are you thinking along the realms of a psychiatrist, but you’re also considering the viewpoint of the other subspecialties at the same time. For me, it brings together my medical background with my passion for psychiatry, and the patients I see daily allow for this incorporation.
Dr. Stanciu: How did your career path prepare you to become a C-L psychiatrist?
Dr. Ganpat: My career path was unique in that I completed a family medicine residency, and then immediately pursued training in psychiatry. Some may consider this as “overkill” for C-L, but as I’ve come to learn, this background grants me a level of understanding and confidence to step in when dealing with a complex case and lend a hand to the consulting physician beyond psychiatry. I do not feel a fellowship is required to practice C-L psychiatry. However, a psychosomatic fellowship will definitely provide the experience needed for this career path, and also will enable one to get a second American Board of Psychiatry and Neurology board certification.
Dr. Stanciu: What types of clinical conditions are you asked to provide input on managing, and how do you find working alongside other specialties?
Dr. Ganpat: I have been managing the full breadth of psychiatry, and in some cases I also provide medical management. Practicing in a metropolitan area with a high influx of tourists also brings in unique cultural cases. The level of respect that the other specialties give is impressive, because they have now seen what a C-L psychiatrist can do. Their performance scores also have improved as a result of my involvement. They greatly appreciate my efforts to shed light on cases or assist with the ever-challenging patient whose psychiatric complexity impedes care.
Dr. Stanciu: How would you describe a physician who is well-suited for such a setting?
Dr. Ganpat: The perfect candidate for this role should be capable of abstract as well as objective thinking. Having a good understanding of the other medical specialties and being able to solve problems is essential, because often it isn’t a clear-cut picture. It is imperative for the C-L psychiatrist to have sound teaching abilities and to be able to educate and communicate his (her) reasoning to the consulting team. It also is important to be well-versed in the psychiatric manifestations of various medical disorders and the psychiatric iatrogenesis of widely used prescription medications.
Dr. Stanciu: What challenges and surprises did you encounter when you first began to practice in this setting?
Dr. Ganpat: I think the largest challenge that I have encountered is the lack of resources. Substance abuse is a major problem here, especially opioids, and there are limited community resources for these patients, so they wind up in the hospital.
Dr. Stanciu: What are the disadvantages of C-L compared with other branches of psychiatry?
Dr. Ganpat: There isn’t much continuity of care with C-L psychiatry over the long run, but you do get to see patients improve during the duration of their hospitalization, which is very rewarding.
Dr. Stanciu: What is the typical reimbursement model for a C-L psychiatrist, and have you run into difficulties with insurance providers in this setting?
Dr. Ganpat: The reimbursement model varies from one system to the next. The common model is to bill just as any other hospital service would, based on the time or level of complexity. Obviously, the more consults you have, the more billing is generated. Most insurance carriers recognize this and so I haven’t had much of an issue with reimbursement, although some unexpected problems may arise.
Dr. Stanciu: What advice do you have for early career psychiatrists and trainees who are contemplating a C-L career?
Dr. Ganpat: If you enjoy working in the hospital and interfacing with other specialties, then consider C-L psychiatry. It is challenging but intellectually stimulating. Make sure you request a C-L rotation during your training, because the Accreditation Council for Graduate Medical Education requires it during a psychiatric residency.
Dr. Stanciu: What is the future outlook of C-L?
Dr. Ganpat: There is a shortage of C-L psychiatrists because >50% of practicing psychiatrists are in private practice in an outpatient setting. Because access to psychiatric care outside of a hospital setting is an issue, and much care is being driven to hospitals, there will be an increasing need for C-L psychiatrists.
Disfiguring Ulcerative Neutrophilic Dermatosis Secondary to Doxycycline and Isotretinoin in an Adolescent Boy With Acne Conglobata
Acne fulminans is an uncommon and debilitating disease that presents as an acute eruption of nodular and ulcerative acne lesions with associated systemic symptoms.1,2 Although its underlying pathophysiology is not well understood, it occurs commonly during treatment of severe acne (eg, acne conglobata) with isotretinoin in young adolescent males.3 Zaba et al4 indicated that an underlying genetic disorder, increase in serum androgen levels, or presence of autoimmune disorders may contribute to the development of acne fulminans.
Isotretinoin and doxycycline also can potentially induce development of neutrophilic dermatoses including Sweet syndrome and pyoderma gangrenosum in patients with severe acne lesions, which can be clinically similar to an acne fulminans eruption. The neutrophilic dermatosis is characterized by the acute appearance of painful ulcerative papulonodules accompanied by systemic symptoms including fever and leukocytosis.
Case Report
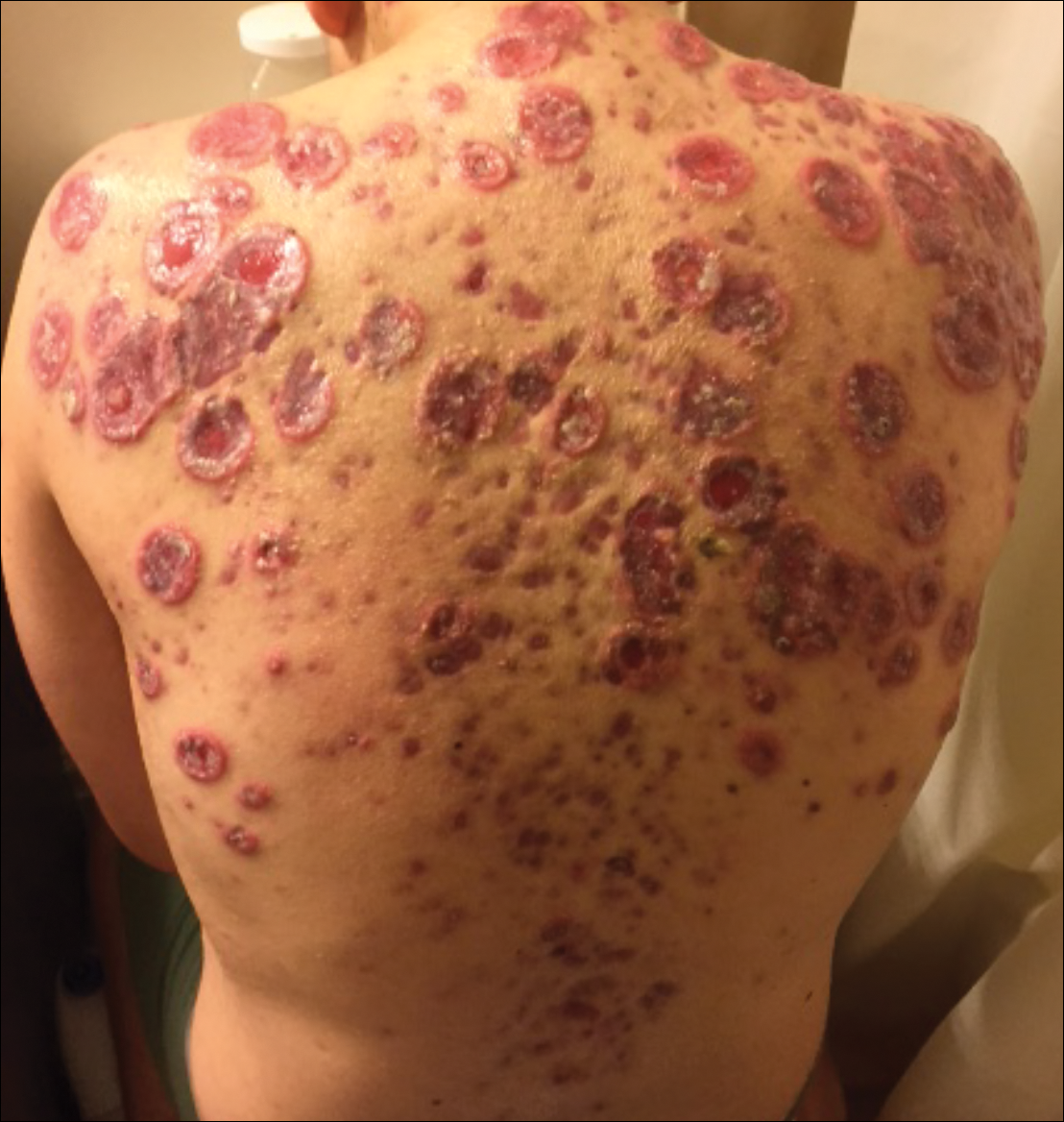
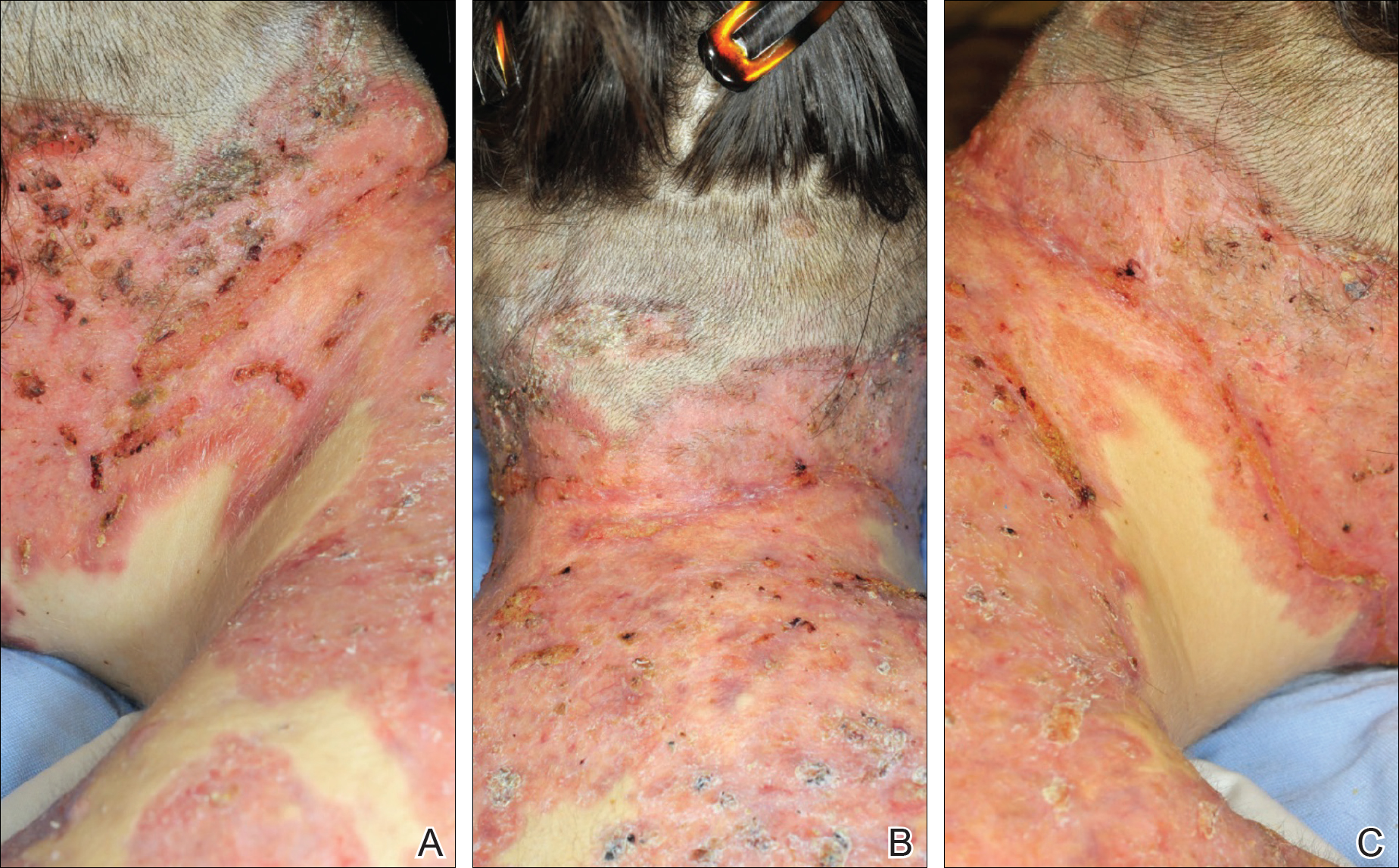
A 13-year-old adolescent boy was initially assessed by his family physician 2 months prior and started on oral doxycycline 100 mg twice daily for acne conglobata on the back. Unfortunately, the acne lesions, especially those on the upper back (Figure 1), started getting worse after 1 month of treatment with doxycycline; thus, he subsequently was switched to oral isotretinoin 0.5 mg/kg once daily. Less than 2 weeks later, the acne lesions worsened, and the patient also developed severe generalized arthralgia, myalgia, and fever (>38.3°C). He acutely developed hundreds of ulcerative plaques covering the entire trunk, upper extremities, face, and neck.
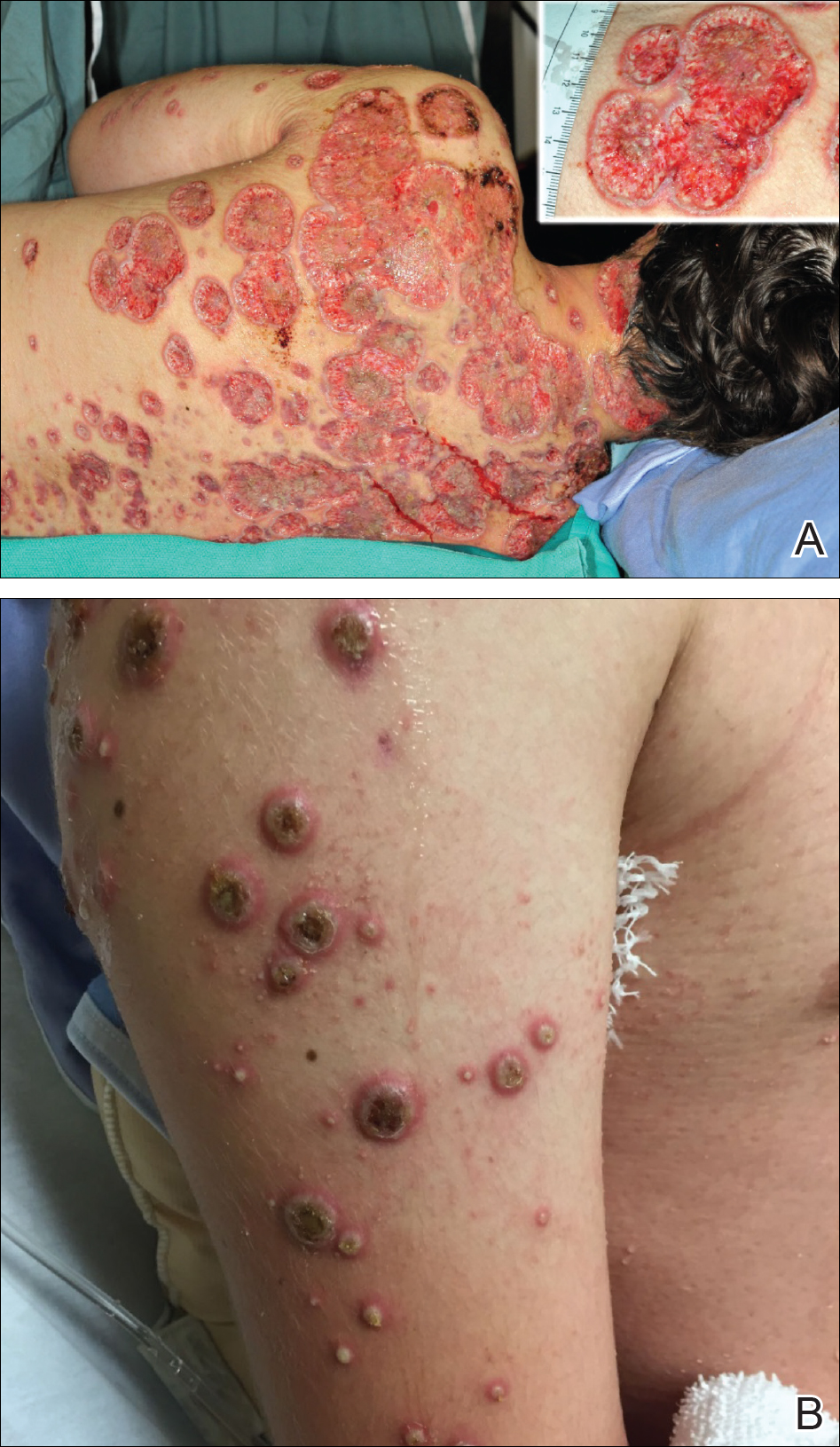
He was admitted to the Stollery Children’s Hospital (Edmonton, Alberta, Canada) and was assessed by the dermatology, rheumatology, and general pediatric teams (Figure 2). He initially was investigated for the potential presence of autoinflammatory disorders, such as PAPA syndrome (pyogenic arthritis, pyoderma gangrenosum, acne) and SAPHO syndrome (synovitis, acne, pustulosis, hyperostosis, osteitis).
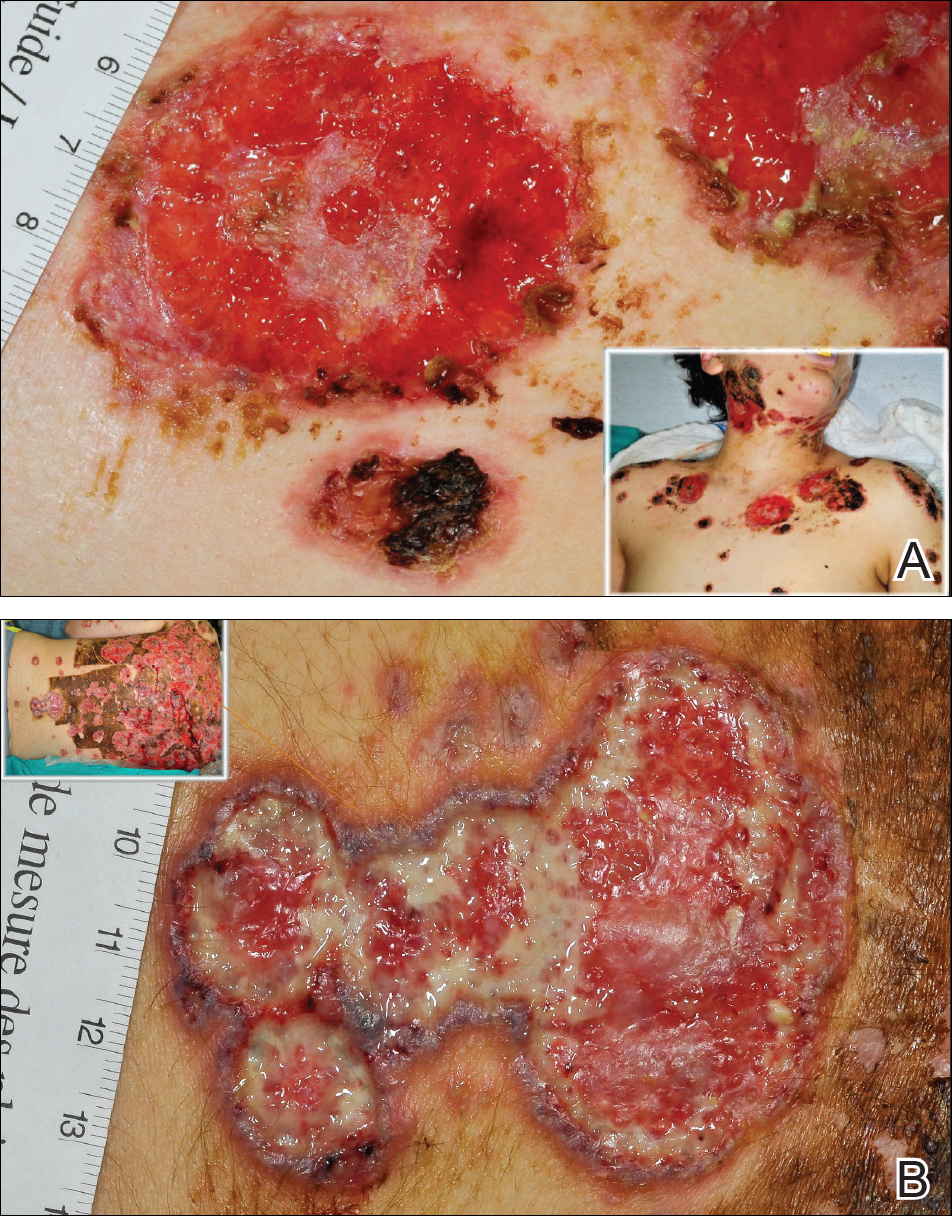
The patient initially was treated with prednisone 30 mg once daily for 3 weeks; dapsone 50 mg once daily and colchicine 0.6 mg twice daily were added while attempting to slowly wean off the prednisone (starting at 30 mg daily and reducing by 5 mg every other week). An attempt to discontinue the prednisone after 2 months was followed by immediate recurrence of the lesions (Figure 3), and the prednisone was restarted for another month. He was subsequently switched to oral cyclosporine 5 mg/kg once daily and achieved considerable improvement in his skin condition (Figure 4).
Comment
Thomson and Cunliffe5 reported a small case series of 11 young male patients with a mean age of 17 years who presented with severe worsening of their acne eruptions after taking isotretinoin, and they all responded well to an oral steroid. In another study, Bottomley and Cunliffe6 indicated that young male patients with notable acne on the trunk who are receiving a minimum dose of 0.5 mg/kg once daily of isotretinoin are at considerable risk for severe worsening of their skin condition.
Although severe worsening of acne lesions leading to acne fulminans or neutrophilic dermatosis secondary to isotretinoin or even doxycycline use is a rare entity, precautionary steps should be taken prior to treating acne conglobata patients with these agents. A review of PubMed articles indexed for MEDLINE using the terms acne, acne conglobata, and doxycycline revealed 2 prior cases of worsening acne in patients treated with doxycycline.7,8 Therefore, any patient presenting with acute worsening of an acne eruption while being treated with isotretinoin or doxycycline needs to be assessed for potential diagnosis of drug-induced acne fulminans or neutrophilic dermatosis.
It has been clearly documented in the literature that both doxycycline and isotretinoin can induce or exacerbate neutrophilic dermatoses in patients with severe underlying acne.6-8 The presentation may be mistaken for worsening acne, leading to inappropriate initiation or increase in the dose of isotretinoin therapy and worsening of the disease with potentially devastating disfiguring consequences. These patients tend to respond well to high-dose oral steroids alone or in combination with dapsone. A slow steroid taper over several months is recommended due to a high tendency for recurrence.
- Grando LR, Leite OG, Cestari TF. Pseudo-acne fulminans associated with oral isotretinoin. An Bras Dermatol. 2014;89:657-659.
- Burns RE, Colville JM. Acne conglobata with septicemia. Arch Dermatol. 1959;79:361-363.
- Karvonen SL. Acne fulminans: report of clinical findings and treatment of twenty-four patients. J Am Acad Dermatol. 1993;28:572-579.
- Zaba R, Schwartz R, Jarmuda S, et al. Acne fulminans: explosive systemic form of acne. J Eur Acad Dermatol Venereol. 2011;25:501-507.
- Thomson KF, Cunliffe WJ. Acne fulminans ‘sine fulminans.’ Clin Exp Dermatol. 2000;25:299-301.
- Bottomley WW, Cunliffe WJ. Severe flares of acne following isotretinoin: large closed comedones (macrocomedones) are a risk factor. Acta Derm Venereol. 1993;73:74.
- Weinstein M, Laxer R, Debosz J, et al. Doxycycline-induced cutaneous inflammation with systemic symptoms in a patient with acne vulgaris. J Cutan Med Surg. 2013;17:283-286.
- Yeo PM, Koh WL, Ang CC, et al. Paradoxical worsening of truncal acne with doxycycline. Ann Acad Med Singapore. 2016;45:430-431.
Acne fulminans is an uncommon and debilitating disease that presents as an acute eruption of nodular and ulcerative acne lesions with associated systemic symptoms.1,2 Although its underlying pathophysiology is not well understood, it occurs commonly during treatment of severe acne (eg, acne conglobata) with isotretinoin in young adolescent males.3 Zaba et al4 indicated that an underlying genetic disorder, increase in serum androgen levels, or presence of autoimmune disorders may contribute to the development of acne fulminans.
Isotretinoin and doxycycline also can potentially induce development of neutrophilic dermatoses including Sweet syndrome and pyoderma gangrenosum in patients with severe acne lesions, which can be clinically similar to an acne fulminans eruption. The neutrophilic dermatosis is characterized by the acute appearance of painful ulcerative papulonodules accompanied by systemic symptoms including fever and leukocytosis.
Case Report
A 13-year-old adolescent boy was initially assessed by his family physician 2 months prior and started on oral doxycycline 100 mg twice daily for acne conglobata on the back. Unfortunately, the acne lesions, especially those on the upper back (Figure 1), started getting worse after 1 month of treatment with doxycycline; thus, he subsequently was switched to oral isotretinoin 0.5 mg/kg once daily. Less than 2 weeks later, the acne lesions worsened, and the patient also developed severe generalized arthralgia, myalgia, and fever (>38.3°C). He acutely developed hundreds of ulcerative plaques covering the entire trunk, upper extremities, face, and neck.

He was admitted to the Stollery Children’s Hospital (Edmonton, Alberta, Canada) and was assessed by the dermatology, rheumatology, and general pediatric teams (Figure 2). He initially was investigated for the potential presence of autoinflammatory disorders, such as PAPA syndrome (pyogenic arthritis, pyoderma gangrenosum, acne) and SAPHO syndrome (synovitis, acne, pustulosis, hyperostosis, osteitis).

The patient initially was treated with prednisone 30 mg once daily for 3 weeks; dapsone 50 mg once daily and colchicine 0.6 mg twice daily were added while attempting to slowly wean off the prednisone (starting at 30 mg daily and reducing by 5 mg every other week). An attempt to discontinue the prednisone after 2 months was followed by immediate recurrence of the lesions (Figure 3), and the prednisone was restarted for another month. He was subsequently switched to oral cyclosporine 5 mg/kg once daily and achieved considerable improvement in his skin condition (Figure 4).


Comment
Thomson and Cunliffe5 reported a small case series of 11 young male patients with a mean age of 17 years who presented with severe worsening of their acne eruptions after taking isotretinoin, and they all responded well to an oral steroid. In another study, Bottomley and Cunliffe6 indicated that young male patients with notable acne on the trunk who are receiving a minimum dose of 0.5 mg/kg once daily of isotretinoin are at considerable risk for severe worsening of their skin condition.
Although severe worsening of acne lesions leading to acne fulminans or neutrophilic dermatosis secondary to isotretinoin or even doxycycline use is a rare entity, precautionary steps should be taken prior to treating acne conglobata patients with these agents. A review of PubMed articles indexed for MEDLINE using the terms acne, acne conglobata, and doxycycline revealed 2 prior cases of worsening acne in patients treated with doxycycline.7,8 Therefore, any patient presenting with acute worsening of an acne eruption while being treated with isotretinoin or doxycycline needs to be assessed for potential diagnosis of drug-induced acne fulminans or neutrophilic dermatosis.
It has been clearly documented in the literature that both doxycycline and isotretinoin can induce or exacerbate neutrophilic dermatoses in patients with severe underlying acne.6-8 The presentation may be mistaken for worsening acne, leading to inappropriate initiation or increase in the dose of isotretinoin therapy and worsening of the disease with potentially devastating disfiguring consequences. These patients tend to respond well to high-dose oral steroids alone or in combination with dapsone. A slow steroid taper over several months is recommended due to a high tendency for recurrence.
Acne fulminans is an uncommon and debilitating disease that presents as an acute eruption of nodular and ulcerative acne lesions with associated systemic symptoms.1,2 Although its underlying pathophysiology is not well understood, it occurs commonly during treatment of severe acne (eg, acne conglobata) with isotretinoin in young adolescent males.3 Zaba et al4 indicated that an underlying genetic disorder, increase in serum androgen levels, or presence of autoimmune disorders may contribute to the development of acne fulminans.
Isotretinoin and doxycycline also can potentially induce development of neutrophilic dermatoses including Sweet syndrome and pyoderma gangrenosum in patients with severe acne lesions, which can be clinically similar to an acne fulminans eruption. The neutrophilic dermatosis is characterized by the acute appearance of painful ulcerative papulonodules accompanied by systemic symptoms including fever and leukocytosis.
Case Report
A 13-year-old adolescent boy was initially assessed by his family physician 2 months prior and started on oral doxycycline 100 mg twice daily for acne conglobata on the back. Unfortunately, the acne lesions, especially those on the upper back (Figure 1), started getting worse after 1 month of treatment with doxycycline; thus, he subsequently was switched to oral isotretinoin 0.5 mg/kg once daily. Less than 2 weeks later, the acne lesions worsened, and the patient also developed severe generalized arthralgia, myalgia, and fever (>38.3°C). He acutely developed hundreds of ulcerative plaques covering the entire trunk, upper extremities, face, and neck.

He was admitted to the Stollery Children’s Hospital (Edmonton, Alberta, Canada) and was assessed by the dermatology, rheumatology, and general pediatric teams (Figure 2). He initially was investigated for the potential presence of autoinflammatory disorders, such as PAPA syndrome (pyogenic arthritis, pyoderma gangrenosum, acne) and SAPHO syndrome (synovitis, acne, pustulosis, hyperostosis, osteitis).

The patient initially was treated with prednisone 30 mg once daily for 3 weeks; dapsone 50 mg once daily and colchicine 0.6 mg twice daily were added while attempting to slowly wean off the prednisone (starting at 30 mg daily and reducing by 5 mg every other week). An attempt to discontinue the prednisone after 2 months was followed by immediate recurrence of the lesions (Figure 3), and the prednisone was restarted for another month. He was subsequently switched to oral cyclosporine 5 mg/kg once daily and achieved considerable improvement in his skin condition (Figure 4).


Comment
Thomson and Cunliffe5 reported a small case series of 11 young male patients with a mean age of 17 years who presented with severe worsening of their acne eruptions after taking isotretinoin, and they all responded well to an oral steroid. In another study, Bottomley and Cunliffe6 indicated that young male patients with notable acne on the trunk who are receiving a minimum dose of 0.5 mg/kg once daily of isotretinoin are at considerable risk for severe worsening of their skin condition.
Although severe worsening of acne lesions leading to acne fulminans or neutrophilic dermatosis secondary to isotretinoin or even doxycycline use is a rare entity, precautionary steps should be taken prior to treating acne conglobata patients with these agents. A review of PubMed articles indexed for MEDLINE using the terms acne, acne conglobata, and doxycycline revealed 2 prior cases of worsening acne in patients treated with doxycycline.7,8 Therefore, any patient presenting with acute worsening of an acne eruption while being treated with isotretinoin or doxycycline needs to be assessed for potential diagnosis of drug-induced acne fulminans or neutrophilic dermatosis.
It has been clearly documented in the literature that both doxycycline and isotretinoin can induce or exacerbate neutrophilic dermatoses in patients with severe underlying acne.6-8 The presentation may be mistaken for worsening acne, leading to inappropriate initiation or increase in the dose of isotretinoin therapy and worsening of the disease with potentially devastating disfiguring consequences. These patients tend to respond well to high-dose oral steroids alone or in combination with dapsone. A slow steroid taper over several months is recommended due to a high tendency for recurrence.
- Grando LR, Leite OG, Cestari TF. Pseudo-acne fulminans associated with oral isotretinoin. An Bras Dermatol. 2014;89:657-659.
- Burns RE, Colville JM. Acne conglobata with septicemia. Arch Dermatol. 1959;79:361-363.
- Karvonen SL. Acne fulminans: report of clinical findings and treatment of twenty-four patients. J Am Acad Dermatol. 1993;28:572-579.
- Zaba R, Schwartz R, Jarmuda S, et al. Acne fulminans: explosive systemic form of acne. J Eur Acad Dermatol Venereol. 2011;25:501-507.
- Thomson KF, Cunliffe WJ. Acne fulminans ‘sine fulminans.’ Clin Exp Dermatol. 2000;25:299-301.
- Bottomley WW, Cunliffe WJ. Severe flares of acne following isotretinoin: large closed comedones (macrocomedones) are a risk factor. Acta Derm Venereol. 1993;73:74.
- Weinstein M, Laxer R, Debosz J, et al. Doxycycline-induced cutaneous inflammation with systemic symptoms in a patient with acne vulgaris. J Cutan Med Surg. 2013;17:283-286.
- Yeo PM, Koh WL, Ang CC, et al. Paradoxical worsening of truncal acne with doxycycline. Ann Acad Med Singapore. 2016;45:430-431.
- Grando LR, Leite OG, Cestari TF. Pseudo-acne fulminans associated with oral isotretinoin. An Bras Dermatol. 2014;89:657-659.
- Burns RE, Colville JM. Acne conglobata with septicemia. Arch Dermatol. 1959;79:361-363.
- Karvonen SL. Acne fulminans: report of clinical findings and treatment of twenty-four patients. J Am Acad Dermatol. 1993;28:572-579.
- Zaba R, Schwartz R, Jarmuda S, et al. Acne fulminans: explosive systemic form of acne. J Eur Acad Dermatol Venereol. 2011;25:501-507.
- Thomson KF, Cunliffe WJ. Acne fulminans ‘sine fulminans.’ Clin Exp Dermatol. 2000;25:299-301.
- Bottomley WW, Cunliffe WJ. Severe flares of acne following isotretinoin: large closed comedones (macrocomedones) are a risk factor. Acta Derm Venereol. 1993;73:74.
- Weinstein M, Laxer R, Debosz J, et al. Doxycycline-induced cutaneous inflammation with systemic symptoms in a patient with acne vulgaris. J Cutan Med Surg. 2013;17:283-286.
- Yeo PM, Koh WL, Ang CC, et al. Paradoxical worsening of truncal acne with doxycycline. Ann Acad Med Singapore. 2016;45:430-431.
Resident Pearl
- Doxycycline and isotretinoin have been widely used for treatment of inflammatory and nodulocystic acne. Although outstanding results can be achieved, paradoxical worsening of acne while starting these medications has been described. In patients with severe acne (ie, acne conglobata), initiation of doxycycline and especially isotretinoin at regular dosages as the sole treatment can impose devastating risks on the patient. These patients are best treated with a combination of low-dose isotretinoin (at the beginning) with a moderate dose of steroids, which should be gradually tapered while the isotretinoin dose is increased to 0.5 to 1 mg/kg once daily.
Sexual harassment
Question: A medical assistant alleged that Dr. Y sexually harassed her by sending anonymous gifts and messages such as, “you’re gorgeous,” and “I love your figure.” It was a repeat of Dr. Y’s previous behavior pattern directed at a different worker, who had lodged a complaint with the human resources department. The medical assistant now files a sexual harassment action under Title VII of the federal Civil Rights Act of 1964 against the health care institution, alleging a hostile work environment.
Which of the following is false?
A. Sexual harassment is a form of sexual misconduct regulated by state medical boards.
B. Mere words, without physical action, may suffice to be deemed sexual harassment.
C. A hostile environment arises when offensive conduct is so severe and pervasive as to amount to job discrimination.
D. Sexual harassment is a civil rights violation unique to the workplace.
E. Liability may attach to the supervisor, institution, or the harasser.
Answer: D. This hypothetical is modified from an actual Connecticut case that was recently decided in favor of the plaintiff.1 In that case, which involved a dentist, the federal Second Circuit unanimously rejected the University of Connecticut Health Center’s appeal against a jury’s verdict holding it responsible for its employee’s sexual harassment of a coworker, who was awarded $125,000. It ruled that the health center should have known of its employee’s harassing behavior.
Sexual harassment, a current hot topic, is pervasive, affecting a diversity of individuals in the fields of media, sports, politics, judiciary, education, entertainment, and others. The medical profession is no exception, and studies indicate that sexual harassment affects patients and physicians alike, occurring in hospitals, private offices, and academic centers.
In a large questionnaire study involving 4,501 female physicians, the authors found a prevalence rate of 47.7%. Harassment was more common while in medical school or during internship, residency, or fellowship than in practice.2 Patients may be the harassers. In 599 of the 1,064 licensed female family physicians in Ontario, more than 75% reported sexual harassment by patients at some time during their careers, either in their own offices by their own patients, or in settings such as emergency departments and clinics, where unknown patients presented an even higher risk.3
When physicians sexually harass fellow workers such as nurses, they distract their victims from providing attentive and competent care. In a review of the subject, researchers cited a study of 188 critical care nurses in hospitals, where nearly half (46%) reported experiencing sexual harassment that included “offensive sexual remarks, unwanted physical contact, unwanted nonverbal attention, requests for unwanted dates, sexual propositions, and physical assault.”4 To this list must now be added misconduct via the use of social media. In the study, physicians (82%), coworkers (20%), and immediate supervisors (7%) accounted for most of the incidents.
Neglecting to look seriously into complaints or to monitor and remedy the situation may create a hostile environment and trigger liability.
An example is the recent well-publicized case of Olympics team physician Dr. Larry Nassar, who was also a faculty member at Michigan State University. Olympic gold medalist McKayla Maroney named both the university and the U.S. Olympic Committee as codefendants in a lawsuit alleging that the institutions failed to properly investigate the team doctor’s criminal sexual conduct.
In Anania v. Daubenspeck Chiropractic, two employees of a chiropractor alleged that his patients sexually harassed them, but he did not remedy the situation.5 The trial court initially dismissed their lawsuit, holding that Ohio law did not recognize a cause of action for sexual harassment by a nonemployee patient, and that liability for sexual harassment can only exist in the context of respondeat superior (employer-employee) liability.
However, the court of appeals held that so long as the chiropractor knew or should have known of the harassment, and failed to take corrective action, he could be liable for allowing a hostile environment to exist.
Negligent supervision is another favorite plaintiff’s cause of action. In Doe v. Borromeo, a patient sought to hold the hospital liable for sexual assault by a physician during a medical exam.6 The lower state court had summarily dismissed the case, which was based on vicarious liability, but the state court of appeals reversed, finding the patient’s complaint against the hospital included a negligent supervision claim.
The appeals court reasoned that this was distinguishable from one based upon vicarious liability, so long as the supervising entity had a duty to protect the victim – and such a duty can only be established if the supervising entity knew or should have known of the existence of the harasser’s propensities, if any, to commit criminal and tortious acts.
Sexual harassment is a form of sex discrimination under Title VII of the Civil Rights Act of 1964, which is enforced by the Equal Employment Opportunity Commission. The commission’s website explains the law in clear and simple language:
“It is unlawful to harass a person (an applicant or employee) because of that person’s sex. Harassment can include ‘sexual harassment’ or unwelcome sexual advances, requests for sexual favors, and other verbal or physical harassment of a sexual nature. Harassment does not have to be of a sexual nature, however, and can include offensive remarks about a person’s sex.
“For example, it is illegal to harass a woman by making offensive comments about women in general. Both victim and the harasser can be either a woman or a man, and the victim and harasser can be the same sex.
“Although the law doesn’t prohibit simple teasing, offhand comments, or isolated incidents that are not very serious, harassment is illegal when it is so frequent or severe that it creates a hostile or offensive work environment or when it results in an adverse employment decision (such as the victim being fired or demoted). The harasser can be the victim’s supervisor, a supervisor in another area, a coworker, or someone who is not an employee of the employer, such as a client or customer.”7
For sexual harassment to occur, the aggrieved party must either show a “hostile environment” or “quid pro quo” situation.
In a hostile environment case, the harassment is serious and persistent, creating unacceptable and offensive work conditions. The plaintiff has to show that the employer knew or should have known of the situation but failed to remedy it.
The “quid pro quo” type of case requires a showing that a person in authority conditioned some aspect of the employee’s employment, such as promotion or retention, upon a sexual favor or relationship.
The U.S. Supreme Court has both clarified and muddied the law’s position on these two previously distinct types of sexual harassment.
In the landmark case of Burlington Industries v. Ellerth, the plaintiff, who was a salesperson, alleged that a supervisor made advances to her and threatened to deny her certain job benefits if she did not cooperate.8 The threats were never carried out, and she was in fact promoted; but her lawsuit alleged that the harassment caused her resignation and amounted to a “constructive” discharge.
Likewise, in Faragher v. City of Boca Raton, the plaintiff, employed as a lifeguard, alleged that her work environment was riddled with crude remarks and obscenities.9 One of the two supervisors reportedly once said to Faragher, “Date me or clean toilets for a year.” Another lifeguard had previously lodged similar complaints. The plaintiff ultimately resigned and brought suit.
The U.S. Supreme Court characterized both of these as “hostile environment” rather than “quid pro quo” cases, because the plaintiffs did not suffer any direct adverse job action. In its decisions, the court defined the scope of liability and affirmative defenses, holding that employers can be subject to vicarious liability when supervisors create actionable hostile work environments.
In other cases, the Supreme Court has ruled for the use of “the reasonable person in the plaintiff’s position” standard in judging the severity of sexual harassment. The court has also held that the genders of the harasser and the harassed employee are not material in determining whether sexual harassment has occurred.
A physician can be accused of harassing an employee, a nurse, an assistant, a fellow worker, a third party, or a patient. Focusing on misconduct within the doctor-patient relationship, the Federation of State Medical Boards adopted in May 2006 a policy entitled “Addressing Sexual Boundaries: Guidelines for State Medical Boards.”10
Although it did not use the term sexual harassment, the policy emphasized that physician sexual misconduct may include behavior that is verbal or physical, and may include expressions of thoughts and feelings or gestures that are sexual. It used the term “sexual impropriety” to denote behavior, gestures, or expressions that are seductive, sexually suggestive, disrespectful of patient privacy, or sexually demeaning to a patient. Together with “sexual violation,” a term the FSMB used when referring to physical sexual contact, they form the basis for disciplinary action by a state medical board.
Caveat: When performing a physical exam, physicians should always use good judgment and sensitivity, relying on the presence of a medical assistant to ensure patient comfort and to alleviate possible embarrassment or anxiety.
Under the federal EEOC rules, the employer rather than the harasser is the defendant. But there are other legal recourses, including tort and criminal actions, that directly target the harasser. Successful plaintiffs may be awarded lost wages, as well as damages for emotional distress, medical expenses, and punitive damages. They may also recover attorney fees.
In one case, a psychiatric nurse was awarded $1.2 million (later reduced to $850,000); in another, a nurse successfully sued a physician’s medical practice and received $150,000 in damages.4 And in an unusual case, a plaintiff was awarded only $1 in damages, but her counsel was paid $41,598 in fees.11 For the practicing doctor, medical board sanction, notoriety, and loss of professional standing and privileges constitute additional costs.
The medical profession is as susceptible as any other – perhaps more so – to allegations of sexual harassment. The magic words for actionable sexual harassment are severe, pervasive, and unwelcome. Although laws in the workplace generally do not prohibit simple teasing, offhand comments, or minor isolated incidents, the line separating these behaviors from bona fide sexual harassment is thin.
Erring on the side of strict and sober professional propriety seems prudent, given the current climate of zero tolerance.
References
1. MacCluskey v. University of Connecticut Health Center, United States Court of Appeals, Second Circuit, No. 17-0807-cv, Dec. 19, 2017.
2. Arch Intern Med. 1998 Feb 23;158(4):352-8.
3. N Engl J Med. 1993 Dec 23;329(26):1936-9.
4. J Nurs Care Qual. 2004 Jul-Sep;19(3):234-41.
5. Anania v. Daubenspeck Chiropractic, 718 N.E. 2d 480 (Ohio 1998).
6. Doe v. Borromeo, Nos. 305162, 305163 (Mich. Ct. App. Sept. 20, 2012).
7. Available at https://www.eeoc.gov/laws/types/sexual_harassment.cfm.
8. Burlington Industries, Inc. v. Ellerth, 524 US 742 (1998).
9. Faragher v. City of Boca Raton, 524 U.S. 775 (1998).
10. Federation of State Medical Boards, “Addressing Sexual Boundaries: Guidelines for State Medical Boards.”
11. J Healthc Risk Manag. 1999 Summer;19(3):14-25.
Question: A medical assistant alleged that Dr. Y sexually harassed her by sending anonymous gifts and messages such as, “you’re gorgeous,” and “I love your figure.” It was a repeat of Dr. Y’s previous behavior pattern directed at a different worker, who had lodged a complaint with the human resources department. The medical assistant now files a sexual harassment action under Title VII of the federal Civil Rights Act of 1964 against the health care institution, alleging a hostile work environment.
Which of the following is false?
A. Sexual harassment is a form of sexual misconduct regulated by state medical boards.
B. Mere words, without physical action, may suffice to be deemed sexual harassment.
C. A hostile environment arises when offensive conduct is so severe and pervasive as to amount to job discrimination.
D. Sexual harassment is a civil rights violation unique to the workplace.
E. Liability may attach to the supervisor, institution, or the harasser.
Answer: D. This hypothetical is modified from an actual Connecticut case that was recently decided in favor of the plaintiff.1 In that case, which involved a dentist, the federal Second Circuit unanimously rejected the University of Connecticut Health Center’s appeal against a jury’s verdict holding it responsible for its employee’s sexual harassment of a coworker, who was awarded $125,000. It ruled that the health center should have known of its employee’s harassing behavior.
Sexual harassment, a current hot topic, is pervasive, affecting a diversity of individuals in the fields of media, sports, politics, judiciary, education, entertainment, and others. The medical profession is no exception, and studies indicate that sexual harassment affects patients and physicians alike, occurring in hospitals, private offices, and academic centers.
In a large questionnaire study involving 4,501 female physicians, the authors found a prevalence rate of 47.7%. Harassment was more common while in medical school or during internship, residency, or fellowship than in practice.2 Patients may be the harassers. In 599 of the 1,064 licensed female family physicians in Ontario, more than 75% reported sexual harassment by patients at some time during their careers, either in their own offices by their own patients, or in settings such as emergency departments and clinics, where unknown patients presented an even higher risk.3
When physicians sexually harass fellow workers such as nurses, they distract their victims from providing attentive and competent care. In a review of the subject, researchers cited a study of 188 critical care nurses in hospitals, where nearly half (46%) reported experiencing sexual harassment that included “offensive sexual remarks, unwanted physical contact, unwanted nonverbal attention, requests for unwanted dates, sexual propositions, and physical assault.”4 To this list must now be added misconduct via the use of social media. In the study, physicians (82%), coworkers (20%), and immediate supervisors (7%) accounted for most of the incidents.
Neglecting to look seriously into complaints or to monitor and remedy the situation may create a hostile environment and trigger liability.
An example is the recent well-publicized case of Olympics team physician Dr. Larry Nassar, who was also a faculty member at Michigan State University. Olympic gold medalist McKayla Maroney named both the university and the U.S. Olympic Committee as codefendants in a lawsuit alleging that the institutions failed to properly investigate the team doctor’s criminal sexual conduct.
In Anania v. Daubenspeck Chiropractic, two employees of a chiropractor alleged that his patients sexually harassed them, but he did not remedy the situation.5 The trial court initially dismissed their lawsuit, holding that Ohio law did not recognize a cause of action for sexual harassment by a nonemployee patient, and that liability for sexual harassment can only exist in the context of respondeat superior (employer-employee) liability.
However, the court of appeals held that so long as the chiropractor knew or should have known of the harassment, and failed to take corrective action, he could be liable for allowing a hostile environment to exist.
Negligent supervision is another favorite plaintiff’s cause of action. In Doe v. Borromeo, a patient sought to hold the hospital liable for sexual assault by a physician during a medical exam.6 The lower state court had summarily dismissed the case, which was based on vicarious liability, but the state court of appeals reversed, finding the patient’s complaint against the hospital included a negligent supervision claim.
The appeals court reasoned that this was distinguishable from one based upon vicarious liability, so long as the supervising entity had a duty to protect the victim – and such a duty can only be established if the supervising entity knew or should have known of the existence of the harasser’s propensities, if any, to commit criminal and tortious acts.
Sexual harassment is a form of sex discrimination under Title VII of the Civil Rights Act of 1964, which is enforced by the Equal Employment Opportunity Commission. The commission’s website explains the law in clear and simple language:
“It is unlawful to harass a person (an applicant or employee) because of that person’s sex. Harassment can include ‘sexual harassment’ or unwelcome sexual advances, requests for sexual favors, and other verbal or physical harassment of a sexual nature. Harassment does not have to be of a sexual nature, however, and can include offensive remarks about a person’s sex.
“For example, it is illegal to harass a woman by making offensive comments about women in general. Both victim and the harasser can be either a woman or a man, and the victim and harasser can be the same sex.
“Although the law doesn’t prohibit simple teasing, offhand comments, or isolated incidents that are not very serious, harassment is illegal when it is so frequent or severe that it creates a hostile or offensive work environment or when it results in an adverse employment decision (such as the victim being fired or demoted). The harasser can be the victim’s supervisor, a supervisor in another area, a coworker, or someone who is not an employee of the employer, such as a client or customer.”7
For sexual harassment to occur, the aggrieved party must either show a “hostile environment” or “quid pro quo” situation.
In a hostile environment case, the harassment is serious and persistent, creating unacceptable and offensive work conditions. The plaintiff has to show that the employer knew or should have known of the situation but failed to remedy it.
The “quid pro quo” type of case requires a showing that a person in authority conditioned some aspect of the employee’s employment, such as promotion or retention, upon a sexual favor or relationship.
The U.S. Supreme Court has both clarified and muddied the law’s position on these two previously distinct types of sexual harassment.
In the landmark case of Burlington Industries v. Ellerth, the plaintiff, who was a salesperson, alleged that a supervisor made advances to her and threatened to deny her certain job benefits if she did not cooperate.8 The threats were never carried out, and she was in fact promoted; but her lawsuit alleged that the harassment caused her resignation and amounted to a “constructive” discharge.
Likewise, in Faragher v. City of Boca Raton, the plaintiff, employed as a lifeguard, alleged that her work environment was riddled with crude remarks and obscenities.9 One of the two supervisors reportedly once said to Faragher, “Date me or clean toilets for a year.” Another lifeguard had previously lodged similar complaints. The plaintiff ultimately resigned and brought suit.
The U.S. Supreme Court characterized both of these as “hostile environment” rather than “quid pro quo” cases, because the plaintiffs did not suffer any direct adverse job action. In its decisions, the court defined the scope of liability and affirmative defenses, holding that employers can be subject to vicarious liability when supervisors create actionable hostile work environments.
In other cases, the Supreme Court has ruled for the use of “the reasonable person in the plaintiff’s position” standard in judging the severity of sexual harassment. The court has also held that the genders of the harasser and the harassed employee are not material in determining whether sexual harassment has occurred.
A physician can be accused of harassing an employee, a nurse, an assistant, a fellow worker, a third party, or a patient. Focusing on misconduct within the doctor-patient relationship, the Federation of State Medical Boards adopted in May 2006 a policy entitled “Addressing Sexual Boundaries: Guidelines for State Medical Boards.”10
Although it did not use the term sexual harassment, the policy emphasized that physician sexual misconduct may include behavior that is verbal or physical, and may include expressions of thoughts and feelings or gestures that are sexual. It used the term “sexual impropriety” to denote behavior, gestures, or expressions that are seductive, sexually suggestive, disrespectful of patient privacy, or sexually demeaning to a patient. Together with “sexual violation,” a term the FSMB used when referring to physical sexual contact, they form the basis for disciplinary action by a state medical board.
Caveat: When performing a physical exam, physicians should always use good judgment and sensitivity, relying on the presence of a medical assistant to ensure patient comfort and to alleviate possible embarrassment or anxiety.
Under the federal EEOC rules, the employer rather than the harasser is the defendant. But there are other legal recourses, including tort and criminal actions, that directly target the harasser. Successful plaintiffs may be awarded lost wages, as well as damages for emotional distress, medical expenses, and punitive damages. They may also recover attorney fees.
In one case, a psychiatric nurse was awarded $1.2 million (later reduced to $850,000); in another, a nurse successfully sued a physician’s medical practice and received $150,000 in damages.4 And in an unusual case, a plaintiff was awarded only $1 in damages, but her counsel was paid $41,598 in fees.11 For the practicing doctor, medical board sanction, notoriety, and loss of professional standing and privileges constitute additional costs.
The medical profession is as susceptible as any other – perhaps more so – to allegations of sexual harassment. The magic words for actionable sexual harassment are severe, pervasive, and unwelcome. Although laws in the workplace generally do not prohibit simple teasing, offhand comments, or minor isolated incidents, the line separating these behaviors from bona fide sexual harassment is thin.
Erring on the side of strict and sober professional propriety seems prudent, given the current climate of zero tolerance.
References
1. MacCluskey v. University of Connecticut Health Center, United States Court of Appeals, Second Circuit, No. 17-0807-cv, Dec. 19, 2017.
2. Arch Intern Med. 1998 Feb 23;158(4):352-8.
3. N Engl J Med. 1993 Dec 23;329(26):1936-9.
4. J Nurs Care Qual. 2004 Jul-Sep;19(3):234-41.
5. Anania v. Daubenspeck Chiropractic, 718 N.E. 2d 480 (Ohio 1998).
6. Doe v. Borromeo, Nos. 305162, 305163 (Mich. Ct. App. Sept. 20, 2012).
7. Available at https://www.eeoc.gov/laws/types/sexual_harassment.cfm.
8. Burlington Industries, Inc. v. Ellerth, 524 US 742 (1998).
9. Faragher v. City of Boca Raton, 524 U.S. 775 (1998).
10. Federation of State Medical Boards, “Addressing Sexual Boundaries: Guidelines for State Medical Boards.”
11. J Healthc Risk Manag. 1999 Summer;19(3):14-25.
Question: A medical assistant alleged that Dr. Y sexually harassed her by sending anonymous gifts and messages such as, “you’re gorgeous,” and “I love your figure.” It was a repeat of Dr. Y’s previous behavior pattern directed at a different worker, who had lodged a complaint with the human resources department. The medical assistant now files a sexual harassment action under Title VII of the federal Civil Rights Act of 1964 against the health care institution, alleging a hostile work environment.
Which of the following is false?
A. Sexual harassment is a form of sexual misconduct regulated by state medical boards.
B. Mere words, without physical action, may suffice to be deemed sexual harassment.
C. A hostile environment arises when offensive conduct is so severe and pervasive as to amount to job discrimination.
D. Sexual harassment is a civil rights violation unique to the workplace.
E. Liability may attach to the supervisor, institution, or the harasser.
Answer: D. This hypothetical is modified from an actual Connecticut case that was recently decided in favor of the plaintiff.1 In that case, which involved a dentist, the federal Second Circuit unanimously rejected the University of Connecticut Health Center’s appeal against a jury’s verdict holding it responsible for its employee’s sexual harassment of a coworker, who was awarded $125,000. It ruled that the health center should have known of its employee’s harassing behavior.
Sexual harassment, a current hot topic, is pervasive, affecting a diversity of individuals in the fields of media, sports, politics, judiciary, education, entertainment, and others. The medical profession is no exception, and studies indicate that sexual harassment affects patients and physicians alike, occurring in hospitals, private offices, and academic centers.
In a large questionnaire study involving 4,501 female physicians, the authors found a prevalence rate of 47.7%. Harassment was more common while in medical school or during internship, residency, or fellowship than in practice.2 Patients may be the harassers. In 599 of the 1,064 licensed female family physicians in Ontario, more than 75% reported sexual harassment by patients at some time during their careers, either in their own offices by their own patients, or in settings such as emergency departments and clinics, where unknown patients presented an even higher risk.3
When physicians sexually harass fellow workers such as nurses, they distract their victims from providing attentive and competent care. In a review of the subject, researchers cited a study of 188 critical care nurses in hospitals, where nearly half (46%) reported experiencing sexual harassment that included “offensive sexual remarks, unwanted physical contact, unwanted nonverbal attention, requests for unwanted dates, sexual propositions, and physical assault.”4 To this list must now be added misconduct via the use of social media. In the study, physicians (82%), coworkers (20%), and immediate supervisors (7%) accounted for most of the incidents.
Neglecting to look seriously into complaints or to monitor and remedy the situation may create a hostile environment and trigger liability.
An example is the recent well-publicized case of Olympics team physician Dr. Larry Nassar, who was also a faculty member at Michigan State University. Olympic gold medalist McKayla Maroney named both the university and the U.S. Olympic Committee as codefendants in a lawsuit alleging that the institutions failed to properly investigate the team doctor’s criminal sexual conduct.
In Anania v. Daubenspeck Chiropractic, two employees of a chiropractor alleged that his patients sexually harassed them, but he did not remedy the situation.5 The trial court initially dismissed their lawsuit, holding that Ohio law did not recognize a cause of action for sexual harassment by a nonemployee patient, and that liability for sexual harassment can only exist in the context of respondeat superior (employer-employee) liability.
However, the court of appeals held that so long as the chiropractor knew or should have known of the harassment, and failed to take corrective action, he could be liable for allowing a hostile environment to exist.
Negligent supervision is another favorite plaintiff’s cause of action. In Doe v. Borromeo, a patient sought to hold the hospital liable for sexual assault by a physician during a medical exam.6 The lower state court had summarily dismissed the case, which was based on vicarious liability, but the state court of appeals reversed, finding the patient’s complaint against the hospital included a negligent supervision claim.
The appeals court reasoned that this was distinguishable from one based upon vicarious liability, so long as the supervising entity had a duty to protect the victim – and such a duty can only be established if the supervising entity knew or should have known of the existence of the harasser’s propensities, if any, to commit criminal and tortious acts.
Sexual harassment is a form of sex discrimination under Title VII of the Civil Rights Act of 1964, which is enforced by the Equal Employment Opportunity Commission. The commission’s website explains the law in clear and simple language:
“It is unlawful to harass a person (an applicant or employee) because of that person’s sex. Harassment can include ‘sexual harassment’ or unwelcome sexual advances, requests for sexual favors, and other verbal or physical harassment of a sexual nature. Harassment does not have to be of a sexual nature, however, and can include offensive remarks about a person’s sex.
“For example, it is illegal to harass a woman by making offensive comments about women in general. Both victim and the harasser can be either a woman or a man, and the victim and harasser can be the same sex.
“Although the law doesn’t prohibit simple teasing, offhand comments, or isolated incidents that are not very serious, harassment is illegal when it is so frequent or severe that it creates a hostile or offensive work environment or when it results in an adverse employment decision (such as the victim being fired or demoted). The harasser can be the victim’s supervisor, a supervisor in another area, a coworker, or someone who is not an employee of the employer, such as a client or customer.”7
For sexual harassment to occur, the aggrieved party must either show a “hostile environment” or “quid pro quo” situation.
In a hostile environment case, the harassment is serious and persistent, creating unacceptable and offensive work conditions. The plaintiff has to show that the employer knew or should have known of the situation but failed to remedy it.
The “quid pro quo” type of case requires a showing that a person in authority conditioned some aspect of the employee’s employment, such as promotion or retention, upon a sexual favor or relationship.
The U.S. Supreme Court has both clarified and muddied the law’s position on these two previously distinct types of sexual harassment.
In the landmark case of Burlington Industries v. Ellerth, the plaintiff, who was a salesperson, alleged that a supervisor made advances to her and threatened to deny her certain job benefits if she did not cooperate.8 The threats were never carried out, and she was in fact promoted; but her lawsuit alleged that the harassment caused her resignation and amounted to a “constructive” discharge.
Likewise, in Faragher v. City of Boca Raton, the plaintiff, employed as a lifeguard, alleged that her work environment was riddled with crude remarks and obscenities.9 One of the two supervisors reportedly once said to Faragher, “Date me or clean toilets for a year.” Another lifeguard had previously lodged similar complaints. The plaintiff ultimately resigned and brought suit.
The U.S. Supreme Court characterized both of these as “hostile environment” rather than “quid pro quo” cases, because the plaintiffs did not suffer any direct adverse job action. In its decisions, the court defined the scope of liability and affirmative defenses, holding that employers can be subject to vicarious liability when supervisors create actionable hostile work environments.
In other cases, the Supreme Court has ruled for the use of “the reasonable person in the plaintiff’s position” standard in judging the severity of sexual harassment. The court has also held that the genders of the harasser and the harassed employee are not material in determining whether sexual harassment has occurred.
A physician can be accused of harassing an employee, a nurse, an assistant, a fellow worker, a third party, or a patient. Focusing on misconduct within the doctor-patient relationship, the Federation of State Medical Boards adopted in May 2006 a policy entitled “Addressing Sexual Boundaries: Guidelines for State Medical Boards.”10
Although it did not use the term sexual harassment, the policy emphasized that physician sexual misconduct may include behavior that is verbal or physical, and may include expressions of thoughts and feelings or gestures that are sexual. It used the term “sexual impropriety” to denote behavior, gestures, or expressions that are seductive, sexually suggestive, disrespectful of patient privacy, or sexually demeaning to a patient. Together with “sexual violation,” a term the FSMB used when referring to physical sexual contact, they form the basis for disciplinary action by a state medical board.
Caveat: When performing a physical exam, physicians should always use good judgment and sensitivity, relying on the presence of a medical assistant to ensure patient comfort and to alleviate possible embarrassment or anxiety.
Under the federal EEOC rules, the employer rather than the harasser is the defendant. But there are other legal recourses, including tort and criminal actions, that directly target the harasser. Successful plaintiffs may be awarded lost wages, as well as damages for emotional distress, medical expenses, and punitive damages. They may also recover attorney fees.
In one case, a psychiatric nurse was awarded $1.2 million (later reduced to $850,000); in another, a nurse successfully sued a physician’s medical practice and received $150,000 in damages.4 And in an unusual case, a plaintiff was awarded only $1 in damages, but her counsel was paid $41,598 in fees.11 For the practicing doctor, medical board sanction, notoriety, and loss of professional standing and privileges constitute additional costs.
The medical profession is as susceptible as any other – perhaps more so – to allegations of sexual harassment. The magic words for actionable sexual harassment are severe, pervasive, and unwelcome. Although laws in the workplace generally do not prohibit simple teasing, offhand comments, or minor isolated incidents, the line separating these behaviors from bona fide sexual harassment is thin.
Erring on the side of strict and sober professional propriety seems prudent, given the current climate of zero tolerance.
References
1. MacCluskey v. University of Connecticut Health Center, United States Court of Appeals, Second Circuit, No. 17-0807-cv, Dec. 19, 2017.
2. Arch Intern Med. 1998 Feb 23;158(4):352-8.
3. N Engl J Med. 1993 Dec 23;329(26):1936-9.
4. J Nurs Care Qual. 2004 Jul-Sep;19(3):234-41.
5. Anania v. Daubenspeck Chiropractic, 718 N.E. 2d 480 (Ohio 1998).
6. Doe v. Borromeo, Nos. 305162, 305163 (Mich. Ct. App. Sept. 20, 2012).
7. Available at https://www.eeoc.gov/laws/types/sexual_harassment.cfm.
8. Burlington Industries, Inc. v. Ellerth, 524 US 742 (1998).
9. Faragher v. City of Boca Raton, 524 U.S. 775 (1998).
10. Federation of State Medical Boards, “Addressing Sexual Boundaries: Guidelines for State Medical Boards.”
11. J Healthc Risk Manag. 1999 Summer;19(3):14-25.
Career Choices: State hospital psychiatry
Editor’s note: Career Choices is a new feature of Residents’ Voices. It features a psychiatry resident/fellow interviewing a psychiatrist about why he (she) has chosen a specific career path. The goal is to inform trainees about the various psychiatric career options, and to give them a feel for the pros and cons of the various paths. Future installments will feature interviews with psychiatrists who have focused their careers on consultation-liaison psychiatry, academic psychiatry, rural psychiatry, and other career paths.
In this first Career Choices, Cornel Stanciu, MD, talked with Samantha Gnanasegaram, MD, a state hospital psychiatrist at New Hampshire Hospital, where she treats severe and chronic mental illness and testifies in various court proceedings.
Dr. Stanciu: What made you choose to become a state hospital psychiatrist?
Dr. Gnanasegaram: When I started thinking about career options after residency, I knew I wanted to start my career in a facility where I could be challenged, remain up-to-date with the most current evidence-based literature, and have the support and mentorship of seasoned psychiatrists in the field. The opportunity to work under the auspices of a great academic institution with the “bread and butter” of psychiatry reminds me every day why I chose the field in the first place. The often chronic and sometimes refractory cases I encounter daily are extremely thought-provoking, and they motivate me to think and pursue more complex management options. [This setting] also enables me to work closely as [part of] an interdisciplinary team with nursing, social work, and recreational and occupational therapy in ensuring these individuals get the best care and aftercare plans.
We often forget that psychosis often takes weeks to respond [to treatment]. Unfortunately, often in private hospitals, the longer stays that are necessary for patient care are not always possible, leading to premature psychotropic changes and discharge. In this setting, I am able to practice medicine based on what is best for the patient from an evidence-based standpoint. Additionally, being in the state system also allows me to learn first-hand and work closely with the legal system in this state and to testify in various settings to ensure my patients get the best possible care.
Dr. Stanciu: How did your career path prepare you to become a state hospital psychiatrist?
Dr. Gnanasegaram: During my residency, I had exposure to the affiliated state psychiatric hospital and spent some time on various units, each geared toward different patient populations. I also became very familiar with a wide range of psychotropics, ranging from first-line to second- and third-tier medications, as well as off-label. The ECT exposure as well as Crisis Prevention Institute training in how to deal with violent and aggressive individuals certainly added extra layers to my proficiency.
Dr. Stanciu: How would you describe a physician who is well-suited for such a setting?
Dr. Gnanasegaram: This setting is great for someone who likes to be challenged and stay current with literature. Furthermore, this is a great setting for those who are comfortable with the use of medications such as [clozapine] and long-acting injectables, and procedures such as ECT. Additionally, an ideal candidate is someone who understands the chronicity and complexity of mental illness, and has the patience to follow the course and does not rush to make drastic changes or panics at the first sign of a patient taking a step back.
A good candidate also should be comfortable with medical comorbidities, because severe mental illness often leads to poor self-care, diabetes, hypertension, etc., and should be able to work effectively in a team setting and interact with other specialties. State hospital physicians need to be cognizant of outpatient resources available to prevent decompensation in the community and not only focus on acute stabilization. Additionally, this is a great setting for those who enjoy working in an interdisciplinary team and learning from the expertise of different members of a treatment team.
Dr. Stanciu: What challenges and surprises did you encounter when you first began to practice in this setting?
Dr. Gnanasegaram: When I started, the biggest challenge was learning about the differences in practice and legislature in a different state, because all states vary in their involuntary commitment laws, process, and ability to institute forced medications. Learning this as well as how they apply to my practice occurred quicker than I anticipated. As I started practicing, I became more proficient in being able to incorporate the resources I have available.
Dr. Stanciu: What are the disadvantages compared with other branches of psychiatry?
Dr. Gnanasegaram: This is a subjective question. Some physicians may desire a rapid turnaround of patients, which is not always the case in state psychiatric hospitals. Even at discharge, some patients may have low-functioning baselines, requiring guardianship and/or placement in a more supervised setting to ensure they receive the care they need. It is also important to realize these are primarily not voluntary patients, but rather patients committed here involuntarily for treatment due to impaired insight and judgment. At times, the acuity can be high, but the potential for violence is mitigated through comprehensive risk assessments, staff training, and prevention strategies to help ensure patient and staff safety.
Dr. Stanciu: What advice do you have for early career psychiatrists and trainees who are contemplating a state hospital career?
Dr. Gnanasegaram: I would recommend seeking exposure to working in a state psychiatric hospital early in your training so you can see the daily routine and protocol. It would help to obtain mentorship from a state hospital psychiatrist in the state where you intend to work. Ask as many questions as needed and seek their insight into the challenges and benefits of working there. During training, it’s important to familiarize yourself with managing difficult and refractory cases, and don’t shy away from challenging patients. The next step would be to apply for a position of interest to interview and learn more about the facility and the staff that you will be working with.
Dr. Stanciu: How important is the academic affiliation?
Dr. Gnanasegaram: Very important. Especially during the early phase of your career, it is important to have at your fingertips senior mentors and to be involved in the conferences and CME activities offered. This ensures good quality measures in patient care. The academic affiliation helps keep you up-to-date with advancements and maintains an atmosphere that fosters ongoing learning and the best possible care for your patients. Working with trainees at various levels, such as medical students, residents, and fellows, allows you to maintain an evidence-based practice approach as well as share your knowledge and experience with those in training. Being in this academic setting, you also have the opportunity for involvement in research activities and publications.
Editor’s note: Career Choices is a new feature of Residents’ Voices. It features a psychiatry resident/fellow interviewing a psychiatrist about why he (she) has chosen a specific career path. The goal is to inform trainees about the various psychiatric career options, and to give them a feel for the pros and cons of the various paths. Future installments will feature interviews with psychiatrists who have focused their careers on consultation-liaison psychiatry, academic psychiatry, rural psychiatry, and other career paths.
In this first Career Choices, Cornel Stanciu, MD, talked with Samantha Gnanasegaram, MD, a state hospital psychiatrist at New Hampshire Hospital, where she treats severe and chronic mental illness and testifies in various court proceedings.
Dr. Stanciu: What made you choose to become a state hospital psychiatrist?
Dr. Gnanasegaram: When I started thinking about career options after residency, I knew I wanted to start my career in a facility where I could be challenged, remain up-to-date with the most current evidence-based literature, and have the support and mentorship of seasoned psychiatrists in the field. The opportunity to work under the auspices of a great academic institution with the “bread and butter” of psychiatry reminds me every day why I chose the field in the first place. The often chronic and sometimes refractory cases I encounter daily are extremely thought-provoking, and they motivate me to think and pursue more complex management options. [This setting] also enables me to work closely as [part of] an interdisciplinary team with nursing, social work, and recreational and occupational therapy in ensuring these individuals get the best care and aftercare plans.
We often forget that psychosis often takes weeks to respond [to treatment]. Unfortunately, often in private hospitals, the longer stays that are necessary for patient care are not always possible, leading to premature psychotropic changes and discharge. In this setting, I am able to practice medicine based on what is best for the patient from an evidence-based standpoint. Additionally, being in the state system also allows me to learn first-hand and work closely with the legal system in this state and to testify in various settings to ensure my patients get the best possible care.
Dr. Stanciu: How did your career path prepare you to become a state hospital psychiatrist?
Dr. Gnanasegaram: During my residency, I had exposure to the affiliated state psychiatric hospital and spent some time on various units, each geared toward different patient populations. I also became very familiar with a wide range of psychotropics, ranging from first-line to second- and third-tier medications, as well as off-label. The ECT exposure as well as Crisis Prevention Institute training in how to deal with violent and aggressive individuals certainly added extra layers to my proficiency.
Dr. Stanciu: How would you describe a physician who is well-suited for such a setting?
Dr. Gnanasegaram: This setting is great for someone who likes to be challenged and stay current with literature. Furthermore, this is a great setting for those who are comfortable with the use of medications such as [clozapine] and long-acting injectables, and procedures such as ECT. Additionally, an ideal candidate is someone who understands the chronicity and complexity of mental illness, and has the patience to follow the course and does not rush to make drastic changes or panics at the first sign of a patient taking a step back.
A good candidate also should be comfortable with medical comorbidities, because severe mental illness often leads to poor self-care, diabetes, hypertension, etc., and should be able to work effectively in a team setting and interact with other specialties. State hospital physicians need to be cognizant of outpatient resources available to prevent decompensation in the community and not only focus on acute stabilization. Additionally, this is a great setting for those who enjoy working in an interdisciplinary team and learning from the expertise of different members of a treatment team.
Dr. Stanciu: What challenges and surprises did you encounter when you first began to practice in this setting?
Dr. Gnanasegaram: When I started, the biggest challenge was learning about the differences in practice and legislature in a different state, because all states vary in their involuntary commitment laws, process, and ability to institute forced medications. Learning this as well as how they apply to my practice occurred quicker than I anticipated. As I started practicing, I became more proficient in being able to incorporate the resources I have available.
Dr. Stanciu: What are the disadvantages compared with other branches of psychiatry?
Dr. Gnanasegaram: This is a subjective question. Some physicians may desire a rapid turnaround of patients, which is not always the case in state psychiatric hospitals. Even at discharge, some patients may have low-functioning baselines, requiring guardianship and/or placement in a more supervised setting to ensure they receive the care they need. It is also important to realize these are primarily not voluntary patients, but rather patients committed here involuntarily for treatment due to impaired insight and judgment. At times, the acuity can be high, but the potential for violence is mitigated through comprehensive risk assessments, staff training, and prevention strategies to help ensure patient and staff safety.
Dr. Stanciu: What advice do you have for early career psychiatrists and trainees who are contemplating a state hospital career?
Dr. Gnanasegaram: I would recommend seeking exposure to working in a state psychiatric hospital early in your training so you can see the daily routine and protocol. It would help to obtain mentorship from a state hospital psychiatrist in the state where you intend to work. Ask as many questions as needed and seek their insight into the challenges and benefits of working there. During training, it’s important to familiarize yourself with managing difficult and refractory cases, and don’t shy away from challenging patients. The next step would be to apply for a position of interest to interview and learn more about the facility and the staff that you will be working with.
Dr. Stanciu: How important is the academic affiliation?
Dr. Gnanasegaram: Very important. Especially during the early phase of your career, it is important to have at your fingertips senior mentors and to be involved in the conferences and CME activities offered. This ensures good quality measures in patient care. The academic affiliation helps keep you up-to-date with advancements and maintains an atmosphere that fosters ongoing learning and the best possible care for your patients. Working with trainees at various levels, such as medical students, residents, and fellows, allows you to maintain an evidence-based practice approach as well as share your knowledge and experience with those in training. Being in this academic setting, you also have the opportunity for involvement in research activities and publications.
Editor’s note: Career Choices is a new feature of Residents’ Voices. It features a psychiatry resident/fellow interviewing a psychiatrist about why he (she) has chosen a specific career path. The goal is to inform trainees about the various psychiatric career options, and to give them a feel for the pros and cons of the various paths. Future installments will feature interviews with psychiatrists who have focused their careers on consultation-liaison psychiatry, academic psychiatry, rural psychiatry, and other career paths.
In this first Career Choices, Cornel Stanciu, MD, talked with Samantha Gnanasegaram, MD, a state hospital psychiatrist at New Hampshire Hospital, where she treats severe and chronic mental illness and testifies in various court proceedings.
Dr. Stanciu: What made you choose to become a state hospital psychiatrist?
Dr. Gnanasegaram: When I started thinking about career options after residency, I knew I wanted to start my career in a facility where I could be challenged, remain up-to-date with the most current evidence-based literature, and have the support and mentorship of seasoned psychiatrists in the field. The opportunity to work under the auspices of a great academic institution with the “bread and butter” of psychiatry reminds me every day why I chose the field in the first place. The often chronic and sometimes refractory cases I encounter daily are extremely thought-provoking, and they motivate me to think and pursue more complex management options. [This setting] also enables me to work closely as [part of] an interdisciplinary team with nursing, social work, and recreational and occupational therapy in ensuring these individuals get the best care and aftercare plans.
We often forget that psychosis often takes weeks to respond [to treatment]. Unfortunately, often in private hospitals, the longer stays that are necessary for patient care are not always possible, leading to premature psychotropic changes and discharge. In this setting, I am able to practice medicine based on what is best for the patient from an evidence-based standpoint. Additionally, being in the state system also allows me to learn first-hand and work closely with the legal system in this state and to testify in various settings to ensure my patients get the best possible care.
Dr. Stanciu: How did your career path prepare you to become a state hospital psychiatrist?
Dr. Gnanasegaram: During my residency, I had exposure to the affiliated state psychiatric hospital and spent some time on various units, each geared toward different patient populations. I also became very familiar with a wide range of psychotropics, ranging from first-line to second- and third-tier medications, as well as off-label. The ECT exposure as well as Crisis Prevention Institute training in how to deal with violent and aggressive individuals certainly added extra layers to my proficiency.
Dr. Stanciu: How would you describe a physician who is well-suited for such a setting?
Dr. Gnanasegaram: This setting is great for someone who likes to be challenged and stay current with literature. Furthermore, this is a great setting for those who are comfortable with the use of medications such as [clozapine] and long-acting injectables, and procedures such as ECT. Additionally, an ideal candidate is someone who understands the chronicity and complexity of mental illness, and has the patience to follow the course and does not rush to make drastic changes or panics at the first sign of a patient taking a step back.
A good candidate also should be comfortable with medical comorbidities, because severe mental illness often leads to poor self-care, diabetes, hypertension, etc., and should be able to work effectively in a team setting and interact with other specialties. State hospital physicians need to be cognizant of outpatient resources available to prevent decompensation in the community and not only focus on acute stabilization. Additionally, this is a great setting for those who enjoy working in an interdisciplinary team and learning from the expertise of different members of a treatment team.
Dr. Stanciu: What challenges and surprises did you encounter when you first began to practice in this setting?
Dr. Gnanasegaram: When I started, the biggest challenge was learning about the differences in practice and legislature in a different state, because all states vary in their involuntary commitment laws, process, and ability to institute forced medications. Learning this as well as how they apply to my practice occurred quicker than I anticipated. As I started practicing, I became more proficient in being able to incorporate the resources I have available.
Dr. Stanciu: What are the disadvantages compared with other branches of psychiatry?
Dr. Gnanasegaram: This is a subjective question. Some physicians may desire a rapid turnaround of patients, which is not always the case in state psychiatric hospitals. Even at discharge, some patients may have low-functioning baselines, requiring guardianship and/or placement in a more supervised setting to ensure they receive the care they need. It is also important to realize these are primarily not voluntary patients, but rather patients committed here involuntarily for treatment due to impaired insight and judgment. At times, the acuity can be high, but the potential for violence is mitigated through comprehensive risk assessments, staff training, and prevention strategies to help ensure patient and staff safety.
Dr. Stanciu: What advice do you have for early career psychiatrists and trainees who are contemplating a state hospital career?
Dr. Gnanasegaram: I would recommend seeking exposure to working in a state psychiatric hospital early in your training so you can see the daily routine and protocol. It would help to obtain mentorship from a state hospital psychiatrist in the state where you intend to work. Ask as many questions as needed and seek their insight into the challenges and benefits of working there. During training, it’s important to familiarize yourself with managing difficult and refractory cases, and don’t shy away from challenging patients. The next step would be to apply for a position of interest to interview and learn more about the facility and the staff that you will be working with.
Dr. Stanciu: How important is the academic affiliation?
Dr. Gnanasegaram: Very important. Especially during the early phase of your career, it is important to have at your fingertips senior mentors and to be involved in the conferences and CME activities offered. This ensures good quality measures in patient care. The academic affiliation helps keep you up-to-date with advancements and maintains an atmosphere that fosters ongoing learning and the best possible care for your patients. Working with trainees at various levels, such as medical students, residents, and fellows, allows you to maintain an evidence-based practice approach as well as share your knowledge and experience with those in training. Being in this academic setting, you also have the opportunity for involvement in research activities and publications.
Pediatric Leg Ulcers: Going Out on a Limb for the Diagnosis
Compared to the adult population with a prevalence of lower extremity ulcers reaching approximately 1% to 2%, pediatric leg ulcers are much less common and require dermatologists to think outside the box for differential diagnoses.1 Although the most common types of lower extremity ulcers in the adult population include venous leg ulcers, arterial ulcers, and diabetic foot ulcers, the etiology for pediatric ulcers is vastly different, and thus these statistics cannot be extrapolated to this younger group. Additionally, scant research has been conducted to construct a systemic algorithm for helping these patients. In 1998, Dangoisse and Song2 concluded that juvenile leg ulcers secondary to causes other than trauma are uncommon, with the infectious origin fairly frequent; however, they stated further workup should be pursued to investigate for underlying vascular, metabolic, hematologic, and immunologic disorders. They also added that an infectious etiology must be ruled out with foremost priority, and a subsequent biopsy could assist in the ultimate diagnosis.2
To further investigate pediatric leg ulcers and their unique causes, a PubMed search of articles indexed for MEDLINE published from 1995 to present was performed using the term pediatric leg ulcers. The search yielded approximately 100 relevant articles. The search generated more than 47 different causes of leg ulcers and produced unusual etiologies such as trophic ulcers of Lesch-Nyhan syndrome, ulcers secondary to disabling pansclerotic morphea of childhood, dracunculiasis, and dengue hemorrhagic fever, among others.3-6 The articles were further divided into 4 categories to better characterize the causes—hematologic, infectious, genodermatoses, and autoimmune—which are reviewed here.
Hematologic Causes
Hematologic causes predominated in this juvenile arena, with sickle cell disease specifically comprising the vast majority of causes of pediatric leg ulcers.7,8 Sickle cell disease is a chronic disease with anemia and sickling crises contributing to a myriad of health problems. In a 13-year study following 44 patients with sickle cell disease, Silva et al8 found that leg ulcers affected approximately 5% of pediatric patients; however, the authors noted that this statistic may underestimate the accurate prevalence, as the ulcers typically affect older children and their study population was a younger distribution. The lesions manifest as painful, well-demarcated ulcers with surrounding hyperpigmentation mimicking venous ulcers.9 The ulcers may be readily diagnosed if the history is known, and it is critical to maximize care of these lesions, as they may heal at least 10 times slower than venous leg ulcers and frequently recur, with the vast majority recurring in less than 1 year. Furthermore, the presence of leg ulcers in sickle cell disease may be associated with increased hemolysis and pulmonary hypertension, demonstrating the severity of disease in these patients.10 Local wound care is the mainstay of therapy including compression, leg elevation, and adjuvant wound dressings. Systemic therapies such as hydroxyurea, zinc supplementation, pentoxifylline, and transfusion therapy may be pursued in refractory cases, though an ideal systemic regimen is still under exploration.9,10 Other major hematologic abnormalities leading to leg ulcers included additional causes of anemia, such as thalassemia and hereditary spherocytosis. These patients additionally were treated with local wound care to maximize healing.11,12
Infectious Causes
Infectious causes of pediatric ulcers were much more varied with a myriad of etiologies such as ulcers from ecthyma gangrenosum caused by Pseudomonas aeruginosa to leishmaniasis and tularemia. The most commonly reported infection causing leg ulcers in the pediatric literature was Mycobacterium ulcerans, which led to the characteristic Buruli ulcer; however, this infection is likely grossly overrepresented, as more common etiologies are underreported; the geographic location for a Buruli ulcer also is important, as cases are rare in the United States.13,14 A Buruli ulcer presents as a well-defined, painless, chronic skin ulceration and most commonly affects children.15 Exposure to stagnant water in tropical climates is thought to play a role in the pathogenesis of this slow-growing, acid-fast bacillus. The bacteria produces a potent cytotoxin called mycolactone, which then induces tissue necrosis and ulceration, leading to the clinical manifestations of disease.15 The ulcers may heal spontaneously; however, up to 15% can be associated with osteomyelitis; treatment includes surgical excision and prolonged antibiotics.14 Given the numerous additional causes of pediatric leg ulcers harboring infections, it is critical to be cognizant of the travel history and immune status of the patient. The infectious cause of leg ulcers likely predominates, making a biopsy with culture necessary in any nonhealing wound in this population prior to pursuing further workup.
Genodermatoses
A number of genodermatoses also contribute to persistent wounds in the pediatric population; specifically, genodermatoses that predispose to neuropathies and decreased pain sensation, which affect the child’s ability to detect sensation in the lower extremities, can result in inadvertent trauma leading to refractory wounds. For example, hereditary, sensory, and autonomic neuropathies are rare disorders causing progressive distal sensory loss, leading to ulcerations, osteomyelitis, arthritis, and even amputation.16 Hereditary, sensory, and autonomic neuropathies are further categorized into several different types; however, the unifying theme of diminished sensation is the culprit for troublesome wounds. Therapeutic endeavors to maximize preventative care with orthotics are vital in allaying recurrent wounds in these patients. Another uncommon hereditary disorder that promotes poor wound healing is caused by an inborn error of collagen synthesis. Prolidase deficiency is an autosomal-recessive condition resulting in characteristic facies, recurrent infections, and recalcitrant leg ulcerations due to impaired collagen formation.17 More than 50% of affected patients experience leg ulcers comprised of irregular borders with prominent granulation tissue. Treatment is aimed at restoring collagen synthesis and optimizing wound healing with the use of topical proline, glycine, and even growth hormone to promote repair.18 Additional genodermatoses predisposing to leg ulcerations include Lesch-Nyhan syndrome due to self-mutilating behaviors and epidermolysis bullosa due to impaired barrier and a decreased ability to repair cutaneous defects.
Autoimmune Causes
Although a much smaller category, ulcers due to autoimmune etiologies were reported in the literature. Fibrosing disorders including morphea and scleroderma can cause extensive disease in severe cases. Disabling pansclerotic morphea of childhood can cause sclerosis that extends into muscle, fascia, and even bone, resulting in contractures and ulcerations.4 The initial areas of involvement are the arms and legs, followed by spread to the trunk and head and neck area.4 Immunosuppressant therapy is needed to halt disease progression. Pediatric cases of systemic lupus erythematosus also have been associated with digital ulcers. One case was thought to be due to vasculitis,19 and another resulted from peripheral gangrene in association with Raynaud phenomenon.20 Albeit rare, it is important to consider autoimmune connective tissue diseases when faced with recurrent wounds and to search for additional symptoms that might yield the underlying diagnosis.
Conclusion
Pediatric leg ulcers are a relatively uncommon phenomenon; however, the etiologies are vastly different than adult leg ulcers and require careful contemplation surrounding the cardinal etiology. The main categories of disease in pediatric leg ulcers after trauma include hematologic abnormalities, infection, genodermatoses, and autoimmune diseases. The evaluation requires obtaining a thorough history and physical examination, including pertinent family histories for associated inheritable disorders. If the clinical picture remains elusive and the ulceration fails conservative management, a biopsy with tissue culture may be necessary to rule out an infectious etiology.
- Morton LM, Phillips TJ. Wound healing and treating wounds: differential diagnosis and evaluation of chronic wounds. J Am Acad Dermatol. 2016;74:589-605.
- Dangoisse C, Song M. Particular aspects of ulcers in children [in French]. Rev Med Brux. 1998;18:241-244.
- Kesiktas E, Gencel E, Acarturk S. Lesch-Nyhan syndrome: reconstruction of a calcaneal defect with a sural flap. Scand J Plast Reconstr Surg Hand Surg. 2006;40:117-119.
- Kura MM, Jindal SR. Disabling pansclerotic morphea of childhood with extracutaneous manifestations. Indian J Dermatol. 2013;58:159.
- Spring M, Spearman P. Dracunculiasis: report of an imported case in the United States. Clin Infect Dis. 1997;25:749-750.
- Vitug MR, Dayrit JF, Oblepias MS, et al. Group A streptococcal septic vasculitis in a child with dengue hemorrhagic fever. Int J Dermatol. 2006;45:1458-1461.
- Adegoke SA, Adeodu OO, Adekile AD. Sickle cell disease clinical phenotypes in children from South-Western Nigeria. Niger J Clin Pract. 2015;18:95-101.
- Silva IV, Reis AF, Palaré MJ, et al. Sickle cell disease in children: chronic complications and search of predictive factors for adverse outcomes. Eur J Haematol. 2015;94:157-161.
- Trent JT, Kirsner RS. Leg ulcers in sickle cell disease. Adv Skin Wound Care. 2004;17:410-416.
- Delaney KM, Axelrod KC, Buscetta A, et al. Leg ulcers in sickle cell disease: current patterns and practices. Hemoglobin. 2013;37:325-332.
- Matta B, Abbas O, Maakaron J, et al. Leg ulcers in patients with B-thalassemia intermedia: a single centre’s experience. J Eur Acad Dermatol Venereol. 2014;28:1245-1250.
- Giraldi S, Abbage KT, Marinoni LP, et al. Leg ulcer in hereditary spherocytosis. Pediatr Dermatol. 2003;20:427-428.
- Journeau P, Fitoussi F, Jehanno P, et al. Buruli’s ulcer: three cases diagnosed and treated in France. J Pediatr Orthop B. 2003;12: 229-232.
- Raghunathan PL, Whitney EA, Asamoa K, et al. Risk factors for Buruli ulcer disease (Mycobacterium ulcerans infection): results from a case-control study in Ghana. Clin Infect Dis. 2005;40:1445-1453.
- Buruli ulcer (Mycobacterium ulcerans infection). World Health Organization website. http://www.who.int/mediacentre/factsheets/fs199/en/. Updated February 2017. Accessed December 19, 2017.
- 16. Rao AG. Painless ulcers and fissures of toes: hereditary sensory neuropathy, not leprosy. Indian J Dermatol. 2016;61:121.
- Adışen E, Erduran FB, Ezqü FS, et al. A rare cause of lower extremity ulcers: prolidase deficiency. Int J Low Extrem Wounds. 2016;15:86-91.
- Trent JT, Kirsner RS. Leg ulcers secondary to prolidase deficiency. Adv Skin Wound Care. 2004;17:468-472.
- Olivieri AN, Mellos A, Duilio C, et al. Refractory vasculitis ulcer of the toe in adolescent suffering from systemic lupus erythematosus treated successfully with hyperbaric oxygen therapy. Ital J Pediatr. 2010;36:72.
- Ziaee V, Yeganeh MH, Moradinejad MH. Peripheral gangrene: a rare presentation of systemic lupus erythematosus in a child. Am J Case Rep. 2013;14:337-340.
Compared to the adult population with a prevalence of lower extremity ulcers reaching approximately 1% to 2%, pediatric leg ulcers are much less common and require dermatologists to think outside the box for differential diagnoses.1 Although the most common types of lower extremity ulcers in the adult population include venous leg ulcers, arterial ulcers, and diabetic foot ulcers, the etiology for pediatric ulcers is vastly different, and thus these statistics cannot be extrapolated to this younger group. Additionally, scant research has been conducted to construct a systemic algorithm for helping these patients. In 1998, Dangoisse and Song2 concluded that juvenile leg ulcers secondary to causes other than trauma are uncommon, with the infectious origin fairly frequent; however, they stated further workup should be pursued to investigate for underlying vascular, metabolic, hematologic, and immunologic disorders. They also added that an infectious etiology must be ruled out with foremost priority, and a subsequent biopsy could assist in the ultimate diagnosis.2
To further investigate pediatric leg ulcers and their unique causes, a PubMed search of articles indexed for MEDLINE published from 1995 to present was performed using the term pediatric leg ulcers. The search yielded approximately 100 relevant articles. The search generated more than 47 different causes of leg ulcers and produced unusual etiologies such as trophic ulcers of Lesch-Nyhan syndrome, ulcers secondary to disabling pansclerotic morphea of childhood, dracunculiasis, and dengue hemorrhagic fever, among others.3-6 The articles were further divided into 4 categories to better characterize the causes—hematologic, infectious, genodermatoses, and autoimmune—which are reviewed here.
Hematologic Causes
Hematologic causes predominated in this juvenile arena, with sickle cell disease specifically comprising the vast majority of causes of pediatric leg ulcers.7,8 Sickle cell disease is a chronic disease with anemia and sickling crises contributing to a myriad of health problems. In a 13-year study following 44 patients with sickle cell disease, Silva et al8 found that leg ulcers affected approximately 5% of pediatric patients; however, the authors noted that this statistic may underestimate the accurate prevalence, as the ulcers typically affect older children and their study population was a younger distribution. The lesions manifest as painful, well-demarcated ulcers with surrounding hyperpigmentation mimicking venous ulcers.9 The ulcers may be readily diagnosed if the history is known, and it is critical to maximize care of these lesions, as they may heal at least 10 times slower than venous leg ulcers and frequently recur, with the vast majority recurring in less than 1 year. Furthermore, the presence of leg ulcers in sickle cell disease may be associated with increased hemolysis and pulmonary hypertension, demonstrating the severity of disease in these patients.10 Local wound care is the mainstay of therapy including compression, leg elevation, and adjuvant wound dressings. Systemic therapies such as hydroxyurea, zinc supplementation, pentoxifylline, and transfusion therapy may be pursued in refractory cases, though an ideal systemic regimen is still under exploration.9,10 Other major hematologic abnormalities leading to leg ulcers included additional causes of anemia, such as thalassemia and hereditary spherocytosis. These patients additionally were treated with local wound care to maximize healing.11,12
Infectious Causes
Infectious causes of pediatric ulcers were much more varied with a myriad of etiologies such as ulcers from ecthyma gangrenosum caused by Pseudomonas aeruginosa to leishmaniasis and tularemia. The most commonly reported infection causing leg ulcers in the pediatric literature was Mycobacterium ulcerans, which led to the characteristic Buruli ulcer; however, this infection is likely grossly overrepresented, as more common etiologies are underreported; the geographic location for a Buruli ulcer also is important, as cases are rare in the United States.13,14 A Buruli ulcer presents as a well-defined, painless, chronic skin ulceration and most commonly affects children.15 Exposure to stagnant water in tropical climates is thought to play a role in the pathogenesis of this slow-growing, acid-fast bacillus. The bacteria produces a potent cytotoxin called mycolactone, which then induces tissue necrosis and ulceration, leading to the clinical manifestations of disease.15 The ulcers may heal spontaneously; however, up to 15% can be associated with osteomyelitis; treatment includes surgical excision and prolonged antibiotics.14 Given the numerous additional causes of pediatric leg ulcers harboring infections, it is critical to be cognizant of the travel history and immune status of the patient. The infectious cause of leg ulcers likely predominates, making a biopsy with culture necessary in any nonhealing wound in this population prior to pursuing further workup.
Genodermatoses
A number of genodermatoses also contribute to persistent wounds in the pediatric population; specifically, genodermatoses that predispose to neuropathies and decreased pain sensation, which affect the child’s ability to detect sensation in the lower extremities, can result in inadvertent trauma leading to refractory wounds. For example, hereditary, sensory, and autonomic neuropathies are rare disorders causing progressive distal sensory loss, leading to ulcerations, osteomyelitis, arthritis, and even amputation.16 Hereditary, sensory, and autonomic neuropathies are further categorized into several different types; however, the unifying theme of diminished sensation is the culprit for troublesome wounds. Therapeutic endeavors to maximize preventative care with orthotics are vital in allaying recurrent wounds in these patients. Another uncommon hereditary disorder that promotes poor wound healing is caused by an inborn error of collagen synthesis. Prolidase deficiency is an autosomal-recessive condition resulting in characteristic facies, recurrent infections, and recalcitrant leg ulcerations due to impaired collagen formation.17 More than 50% of affected patients experience leg ulcers comprised of irregular borders with prominent granulation tissue. Treatment is aimed at restoring collagen synthesis and optimizing wound healing with the use of topical proline, glycine, and even growth hormone to promote repair.18 Additional genodermatoses predisposing to leg ulcerations include Lesch-Nyhan syndrome due to self-mutilating behaviors and epidermolysis bullosa due to impaired barrier and a decreased ability to repair cutaneous defects.
Autoimmune Causes
Although a much smaller category, ulcers due to autoimmune etiologies were reported in the literature. Fibrosing disorders including morphea and scleroderma can cause extensive disease in severe cases. Disabling pansclerotic morphea of childhood can cause sclerosis that extends into muscle, fascia, and even bone, resulting in contractures and ulcerations.4 The initial areas of involvement are the arms and legs, followed by spread to the trunk and head and neck area.4 Immunosuppressant therapy is needed to halt disease progression. Pediatric cases of systemic lupus erythematosus also have been associated with digital ulcers. One case was thought to be due to vasculitis,19 and another resulted from peripheral gangrene in association with Raynaud phenomenon.20 Albeit rare, it is important to consider autoimmune connective tissue diseases when faced with recurrent wounds and to search for additional symptoms that might yield the underlying diagnosis.
Conclusion
Pediatric leg ulcers are a relatively uncommon phenomenon; however, the etiologies are vastly different than adult leg ulcers and require careful contemplation surrounding the cardinal etiology. The main categories of disease in pediatric leg ulcers after trauma include hematologic abnormalities, infection, genodermatoses, and autoimmune diseases. The evaluation requires obtaining a thorough history and physical examination, including pertinent family histories for associated inheritable disorders. If the clinical picture remains elusive and the ulceration fails conservative management, a biopsy with tissue culture may be necessary to rule out an infectious etiology.
Compared to the adult population with a prevalence of lower extremity ulcers reaching approximately 1% to 2%, pediatric leg ulcers are much less common and require dermatologists to think outside the box for differential diagnoses.1 Although the most common types of lower extremity ulcers in the adult population include venous leg ulcers, arterial ulcers, and diabetic foot ulcers, the etiology for pediatric ulcers is vastly different, and thus these statistics cannot be extrapolated to this younger group. Additionally, scant research has been conducted to construct a systemic algorithm for helping these patients. In 1998, Dangoisse and Song2 concluded that juvenile leg ulcers secondary to causes other than trauma are uncommon, with the infectious origin fairly frequent; however, they stated further workup should be pursued to investigate for underlying vascular, metabolic, hematologic, and immunologic disorders. They also added that an infectious etiology must be ruled out with foremost priority, and a subsequent biopsy could assist in the ultimate diagnosis.2
To further investigate pediatric leg ulcers and their unique causes, a PubMed search of articles indexed for MEDLINE published from 1995 to present was performed using the term pediatric leg ulcers. The search yielded approximately 100 relevant articles. The search generated more than 47 different causes of leg ulcers and produced unusual etiologies such as trophic ulcers of Lesch-Nyhan syndrome, ulcers secondary to disabling pansclerotic morphea of childhood, dracunculiasis, and dengue hemorrhagic fever, among others.3-6 The articles were further divided into 4 categories to better characterize the causes—hematologic, infectious, genodermatoses, and autoimmune—which are reviewed here.
Hematologic Causes
Hematologic causes predominated in this juvenile arena, with sickle cell disease specifically comprising the vast majority of causes of pediatric leg ulcers.7,8 Sickle cell disease is a chronic disease with anemia and sickling crises contributing to a myriad of health problems. In a 13-year study following 44 patients with sickle cell disease, Silva et al8 found that leg ulcers affected approximately 5% of pediatric patients; however, the authors noted that this statistic may underestimate the accurate prevalence, as the ulcers typically affect older children and their study population was a younger distribution. The lesions manifest as painful, well-demarcated ulcers with surrounding hyperpigmentation mimicking venous ulcers.9 The ulcers may be readily diagnosed if the history is known, and it is critical to maximize care of these lesions, as they may heal at least 10 times slower than venous leg ulcers and frequently recur, with the vast majority recurring in less than 1 year. Furthermore, the presence of leg ulcers in sickle cell disease may be associated with increased hemolysis and pulmonary hypertension, demonstrating the severity of disease in these patients.10 Local wound care is the mainstay of therapy including compression, leg elevation, and adjuvant wound dressings. Systemic therapies such as hydroxyurea, zinc supplementation, pentoxifylline, and transfusion therapy may be pursued in refractory cases, though an ideal systemic regimen is still under exploration.9,10 Other major hematologic abnormalities leading to leg ulcers included additional causes of anemia, such as thalassemia and hereditary spherocytosis. These patients additionally were treated with local wound care to maximize healing.11,12
Infectious Causes
Infectious causes of pediatric ulcers were much more varied with a myriad of etiologies such as ulcers from ecthyma gangrenosum caused by Pseudomonas aeruginosa to leishmaniasis and tularemia. The most commonly reported infection causing leg ulcers in the pediatric literature was Mycobacterium ulcerans, which led to the characteristic Buruli ulcer; however, this infection is likely grossly overrepresented, as more common etiologies are underreported; the geographic location for a Buruli ulcer also is important, as cases are rare in the United States.13,14 A Buruli ulcer presents as a well-defined, painless, chronic skin ulceration and most commonly affects children.15 Exposure to stagnant water in tropical climates is thought to play a role in the pathogenesis of this slow-growing, acid-fast bacillus. The bacteria produces a potent cytotoxin called mycolactone, which then induces tissue necrosis and ulceration, leading to the clinical manifestations of disease.15 The ulcers may heal spontaneously; however, up to 15% can be associated with osteomyelitis; treatment includes surgical excision and prolonged antibiotics.14 Given the numerous additional causes of pediatric leg ulcers harboring infections, it is critical to be cognizant of the travel history and immune status of the patient. The infectious cause of leg ulcers likely predominates, making a biopsy with culture necessary in any nonhealing wound in this population prior to pursuing further workup.
Genodermatoses
A number of genodermatoses also contribute to persistent wounds in the pediatric population; specifically, genodermatoses that predispose to neuropathies and decreased pain sensation, which affect the child’s ability to detect sensation in the lower extremities, can result in inadvertent trauma leading to refractory wounds. For example, hereditary, sensory, and autonomic neuropathies are rare disorders causing progressive distal sensory loss, leading to ulcerations, osteomyelitis, arthritis, and even amputation.16 Hereditary, sensory, and autonomic neuropathies are further categorized into several different types; however, the unifying theme of diminished sensation is the culprit for troublesome wounds. Therapeutic endeavors to maximize preventative care with orthotics are vital in allaying recurrent wounds in these patients. Another uncommon hereditary disorder that promotes poor wound healing is caused by an inborn error of collagen synthesis. Prolidase deficiency is an autosomal-recessive condition resulting in characteristic facies, recurrent infections, and recalcitrant leg ulcerations due to impaired collagen formation.17 More than 50% of affected patients experience leg ulcers comprised of irregular borders with prominent granulation tissue. Treatment is aimed at restoring collagen synthesis and optimizing wound healing with the use of topical proline, glycine, and even growth hormone to promote repair.18 Additional genodermatoses predisposing to leg ulcerations include Lesch-Nyhan syndrome due to self-mutilating behaviors and epidermolysis bullosa due to impaired barrier and a decreased ability to repair cutaneous defects.
Autoimmune Causes
Although a much smaller category, ulcers due to autoimmune etiologies were reported in the literature. Fibrosing disorders including morphea and scleroderma can cause extensive disease in severe cases. Disabling pansclerotic morphea of childhood can cause sclerosis that extends into muscle, fascia, and even bone, resulting in contractures and ulcerations.4 The initial areas of involvement are the arms and legs, followed by spread to the trunk and head and neck area.4 Immunosuppressant therapy is needed to halt disease progression. Pediatric cases of systemic lupus erythematosus also have been associated with digital ulcers. One case was thought to be due to vasculitis,19 and another resulted from peripheral gangrene in association with Raynaud phenomenon.20 Albeit rare, it is important to consider autoimmune connective tissue diseases when faced with recurrent wounds and to search for additional symptoms that might yield the underlying diagnosis.
Conclusion
Pediatric leg ulcers are a relatively uncommon phenomenon; however, the etiologies are vastly different than adult leg ulcers and require careful contemplation surrounding the cardinal etiology. The main categories of disease in pediatric leg ulcers after trauma include hematologic abnormalities, infection, genodermatoses, and autoimmune diseases. The evaluation requires obtaining a thorough history and physical examination, including pertinent family histories for associated inheritable disorders. If the clinical picture remains elusive and the ulceration fails conservative management, a biopsy with tissue culture may be necessary to rule out an infectious etiology.
- Morton LM, Phillips TJ. Wound healing and treating wounds: differential diagnosis and evaluation of chronic wounds. J Am Acad Dermatol. 2016;74:589-605.
- Dangoisse C, Song M. Particular aspects of ulcers in children [in French]. Rev Med Brux. 1998;18:241-244.
- Kesiktas E, Gencel E, Acarturk S. Lesch-Nyhan syndrome: reconstruction of a calcaneal defect with a sural flap. Scand J Plast Reconstr Surg Hand Surg. 2006;40:117-119.
- Kura MM, Jindal SR. Disabling pansclerotic morphea of childhood with extracutaneous manifestations. Indian J Dermatol. 2013;58:159.
- Spring M, Spearman P. Dracunculiasis: report of an imported case in the United States. Clin Infect Dis. 1997;25:749-750.
- Vitug MR, Dayrit JF, Oblepias MS, et al. Group A streptococcal septic vasculitis in a child with dengue hemorrhagic fever. Int J Dermatol. 2006;45:1458-1461.
- Adegoke SA, Adeodu OO, Adekile AD. Sickle cell disease clinical phenotypes in children from South-Western Nigeria. Niger J Clin Pract. 2015;18:95-101.
- Silva IV, Reis AF, Palaré MJ, et al. Sickle cell disease in children: chronic complications and search of predictive factors for adverse outcomes. Eur J Haematol. 2015;94:157-161.
- Trent JT, Kirsner RS. Leg ulcers in sickle cell disease. Adv Skin Wound Care. 2004;17:410-416.
- Delaney KM, Axelrod KC, Buscetta A, et al. Leg ulcers in sickle cell disease: current patterns and practices. Hemoglobin. 2013;37:325-332.
- Matta B, Abbas O, Maakaron J, et al. Leg ulcers in patients with B-thalassemia intermedia: a single centre’s experience. J Eur Acad Dermatol Venereol. 2014;28:1245-1250.
- Giraldi S, Abbage KT, Marinoni LP, et al. Leg ulcer in hereditary spherocytosis. Pediatr Dermatol. 2003;20:427-428.
- Journeau P, Fitoussi F, Jehanno P, et al. Buruli’s ulcer: three cases diagnosed and treated in France. J Pediatr Orthop B. 2003;12: 229-232.
- Raghunathan PL, Whitney EA, Asamoa K, et al. Risk factors for Buruli ulcer disease (Mycobacterium ulcerans infection): results from a case-control study in Ghana. Clin Infect Dis. 2005;40:1445-1453.
- Buruli ulcer (Mycobacterium ulcerans infection). World Health Organization website. http://www.who.int/mediacentre/factsheets/fs199/en/. Updated February 2017. Accessed December 19, 2017.
- 16. Rao AG. Painless ulcers and fissures of toes: hereditary sensory neuropathy, not leprosy. Indian J Dermatol. 2016;61:121.
- Adışen E, Erduran FB, Ezqü FS, et al. A rare cause of lower extremity ulcers: prolidase deficiency. Int J Low Extrem Wounds. 2016;15:86-91.
- Trent JT, Kirsner RS. Leg ulcers secondary to prolidase deficiency. Adv Skin Wound Care. 2004;17:468-472.
- Olivieri AN, Mellos A, Duilio C, et al. Refractory vasculitis ulcer of the toe in adolescent suffering from systemic lupus erythematosus treated successfully with hyperbaric oxygen therapy. Ital J Pediatr. 2010;36:72.
- Ziaee V, Yeganeh MH, Moradinejad MH. Peripheral gangrene: a rare presentation of systemic lupus erythematosus in a child. Am J Case Rep. 2013;14:337-340.
- Morton LM, Phillips TJ. Wound healing and treating wounds: differential diagnosis and evaluation of chronic wounds. J Am Acad Dermatol. 2016;74:589-605.
- Dangoisse C, Song M. Particular aspects of ulcers in children [in French]. Rev Med Brux. 1998;18:241-244.
- Kesiktas E, Gencel E, Acarturk S. Lesch-Nyhan syndrome: reconstruction of a calcaneal defect with a sural flap. Scand J Plast Reconstr Surg Hand Surg. 2006;40:117-119.
- Kura MM, Jindal SR. Disabling pansclerotic morphea of childhood with extracutaneous manifestations. Indian J Dermatol. 2013;58:159.
- Spring M, Spearman P. Dracunculiasis: report of an imported case in the United States. Clin Infect Dis. 1997;25:749-750.
- Vitug MR, Dayrit JF, Oblepias MS, et al. Group A streptococcal septic vasculitis in a child with dengue hemorrhagic fever. Int J Dermatol. 2006;45:1458-1461.
- Adegoke SA, Adeodu OO, Adekile AD. Sickle cell disease clinical phenotypes in children from South-Western Nigeria. Niger J Clin Pract. 2015;18:95-101.
- Silva IV, Reis AF, Palaré MJ, et al. Sickle cell disease in children: chronic complications and search of predictive factors for adverse outcomes. Eur J Haematol. 2015;94:157-161.
- Trent JT, Kirsner RS. Leg ulcers in sickle cell disease. Adv Skin Wound Care. 2004;17:410-416.
- Delaney KM, Axelrod KC, Buscetta A, et al. Leg ulcers in sickle cell disease: current patterns and practices. Hemoglobin. 2013;37:325-332.
- Matta B, Abbas O, Maakaron J, et al. Leg ulcers in patients with B-thalassemia intermedia: a single centre’s experience. J Eur Acad Dermatol Venereol. 2014;28:1245-1250.
- Giraldi S, Abbage KT, Marinoni LP, et al. Leg ulcer in hereditary spherocytosis. Pediatr Dermatol. 2003;20:427-428.
- Journeau P, Fitoussi F, Jehanno P, et al. Buruli’s ulcer: three cases diagnosed and treated in France. J Pediatr Orthop B. 2003;12: 229-232.
- Raghunathan PL, Whitney EA, Asamoa K, et al. Risk factors for Buruli ulcer disease (Mycobacterium ulcerans infection): results from a case-control study in Ghana. Clin Infect Dis. 2005;40:1445-1453.
- Buruli ulcer (Mycobacterium ulcerans infection). World Health Organization website. http://www.who.int/mediacentre/factsheets/fs199/en/. Updated February 2017. Accessed December 19, 2017.
- 16. Rao AG. Painless ulcers and fissures of toes: hereditary sensory neuropathy, not leprosy. Indian J Dermatol. 2016;61:121.
- Adışen E, Erduran FB, Ezqü FS, et al. A rare cause of lower extremity ulcers: prolidase deficiency. Int J Low Extrem Wounds. 2016;15:86-91.
- Trent JT, Kirsner RS. Leg ulcers secondary to prolidase deficiency. Adv Skin Wound Care. 2004;17:468-472.
- Olivieri AN, Mellos A, Duilio C, et al. Refractory vasculitis ulcer of the toe in adolescent suffering from systemic lupus erythematosus treated successfully with hyperbaric oxygen therapy. Ital J Pediatr. 2010;36:72.
- Ziaee V, Yeganeh MH, Moradinejad MH. Peripheral gangrene: a rare presentation of systemic lupus erythematosus in a child. Am J Case Rep. 2013;14:337-340.
Call for Applications
Dr. Henry Nasrallah, Editor-in-Chief of Current Psychiatry, and Chair of the Department of Psychiatry and Behavioral Neuroscience at Saint Louis University, invites PGY-4 residents who will complete their training in June 2018 to consider applying for a full-time faculty position, which is a mix of student mental health at the university and supervision of the resident's outpatient clinic.
Those interested can email Dr. Nasrallah directly at henry.nasrallah@health.slu.edu and include a CV.
Dr. Henry Nasrallah, Editor-in-Chief of Current Psychiatry, and Chair of the Department of Psychiatry and Behavioral Neuroscience at Saint Louis University, invites PGY-4 residents who will complete their training in June 2018 to consider applying for a full-time faculty position, which is a mix of student mental health at the university and supervision of the resident's outpatient clinic.
Those interested can email Dr. Nasrallah directly at henry.nasrallah@health.slu.edu and include a CV.
Dr. Henry Nasrallah, Editor-in-Chief of Current Psychiatry, and Chair of the Department of Psychiatry and Behavioral Neuroscience at Saint Louis University, invites PGY-4 residents who will complete their training in June 2018 to consider applying for a full-time faculty position, which is a mix of student mental health at the university and supervision of the resident's outpatient clinic.
Those interested can email Dr. Nasrallah directly at henry.nasrallah@health.slu.edu and include a CV.
Nearly all rheumatology slots filled on Specialty Match Day
Once again, rheumatology filled almost every slot offered in the annual Specialty Match.
The specialty had 221 open positions to be filled, with only 3 left open when the 2017 results were announced on Dec. 6.
The number of available positions was up from the 217 slots in 2016, with 7 positions unfilled last year. The number of applicants naming rheumatology as the preferred choice was virtually unchanged year over year, with 313 this year vs. 312 last year.
Of the 313 applicants who identified rheumatology as the preferred choice, 217 were matched to rheumatology, 3 were matched to a different specialty, and 93 did not match.
“Rheumatology did very, very well this year, even better than last year, although it was comparable,” Anne Bass, MD, rheumatology fellowship program director at the Hospital for Special Surgery, New York, said in an interview.
Dr. Bass, who also serves as chair of the American College of Rheumatology Committee on Rheumatology and Training and Workforce Issues, said she expects the remaining open slots to be filled.
“Usually what happens is something called the scramble, which is after the match is over, [the National Resident Matching Program] has a clearinghouse for fellows who haven’t matched and programs that still have slots,” she said in an interview. “It connects them together. I would guess the positions will either be filled or that they are positions that are really targeted, say, for research applicants and there wasn’t anybody appropriate for that position.”
“You do want some excess because not all applicants are necessarily qualified, so you don’t want to have just the right amount of spots if you want to have the best people filling them,” she said, noting that this year, like last, there was around 100 applicants who listed rheumatology as their first choice that did not get assigned as fellowship, about double the number from a few years ago. “At the same time, with the anticipated workforce shortage, what this is telling us is if we created more fellowship slots, we have lots and lots of applicants to fill those slots.”
Dr. Bass said funding issues were the primary stumbling block to training more fellows. She also said more needs to be done to address geographic disparities, as these programs tend to be at major academic institutions in dense urban areas and many fellows practice at sights where they are trained, magnifying shortages in rural areas.
The similarities between last year’s rheumatology fellowship match results and this year’s are in line with what is happening in other specialties.
“Overall, the percentages of programs and positions filled were almost identical to last year,” Mona Singer, president and CEO of the National Resident Matching Program, said. “The same fellowships were competitive this year when compared to last year.”
Specialties filling more than 90% of positions included cardiovascular disease, endocrinology, gastroenterology, hematology/oncology, pulmonary/critical care, and rheumatology.
Ms. Singer noted that infectious diseases and nephrology “have implemented the ‘all-in policy,’ which requires any program participating in the match to place all positions. This was the first year infectious diseases used the policy, and there was a modest improvement in the number of positions filled. Nephrology was unchanged.”
Overall, 4,831 fellowships were available and 4,242 filled. The 87.8% of positions filled is a slight uptick over the 87.5% of positions filled in 2016. A total of 1,249 applicants did not match.
Once again, rheumatology filled almost every slot offered in the annual Specialty Match.
The specialty had 221 open positions to be filled, with only 3 left open when the 2017 results were announced on Dec. 6.
The number of available positions was up from the 217 slots in 2016, with 7 positions unfilled last year. The number of applicants naming rheumatology as the preferred choice was virtually unchanged year over year, with 313 this year vs. 312 last year.
Of the 313 applicants who identified rheumatology as the preferred choice, 217 were matched to rheumatology, 3 were matched to a different specialty, and 93 did not match.
“Rheumatology did very, very well this year, even better than last year, although it was comparable,” Anne Bass, MD, rheumatology fellowship program director at the Hospital for Special Surgery, New York, said in an interview.
Dr. Bass, who also serves as chair of the American College of Rheumatology Committee on Rheumatology and Training and Workforce Issues, said she expects the remaining open slots to be filled.
“Usually what happens is something called the scramble, which is after the match is over, [the National Resident Matching Program] has a clearinghouse for fellows who haven’t matched and programs that still have slots,” she said in an interview. “It connects them together. I would guess the positions will either be filled or that they are positions that are really targeted, say, for research applicants and there wasn’t anybody appropriate for that position.”
“You do want some excess because not all applicants are necessarily qualified, so you don’t want to have just the right amount of spots if you want to have the best people filling them,” she said, noting that this year, like last, there was around 100 applicants who listed rheumatology as their first choice that did not get assigned as fellowship, about double the number from a few years ago. “At the same time, with the anticipated workforce shortage, what this is telling us is if we created more fellowship slots, we have lots and lots of applicants to fill those slots.”
Dr. Bass said funding issues were the primary stumbling block to training more fellows. She also said more needs to be done to address geographic disparities, as these programs tend to be at major academic institutions in dense urban areas and many fellows practice at sights where they are trained, magnifying shortages in rural areas.
The similarities between last year’s rheumatology fellowship match results and this year’s are in line with what is happening in other specialties.
“Overall, the percentages of programs and positions filled were almost identical to last year,” Mona Singer, president and CEO of the National Resident Matching Program, said. “The same fellowships were competitive this year when compared to last year.”
Specialties filling more than 90% of positions included cardiovascular disease, endocrinology, gastroenterology, hematology/oncology, pulmonary/critical care, and rheumatology.
Ms. Singer noted that infectious diseases and nephrology “have implemented the ‘all-in policy,’ which requires any program participating in the match to place all positions. This was the first year infectious diseases used the policy, and there was a modest improvement in the number of positions filled. Nephrology was unchanged.”
Overall, 4,831 fellowships were available and 4,242 filled. The 87.8% of positions filled is a slight uptick over the 87.5% of positions filled in 2016. A total of 1,249 applicants did not match.
Once again, rheumatology filled almost every slot offered in the annual Specialty Match.
The specialty had 221 open positions to be filled, with only 3 left open when the 2017 results were announced on Dec. 6.
The number of available positions was up from the 217 slots in 2016, with 7 positions unfilled last year. The number of applicants naming rheumatology as the preferred choice was virtually unchanged year over year, with 313 this year vs. 312 last year.
Of the 313 applicants who identified rheumatology as the preferred choice, 217 were matched to rheumatology, 3 were matched to a different specialty, and 93 did not match.
“Rheumatology did very, very well this year, even better than last year, although it was comparable,” Anne Bass, MD, rheumatology fellowship program director at the Hospital for Special Surgery, New York, said in an interview.
Dr. Bass, who also serves as chair of the American College of Rheumatology Committee on Rheumatology and Training and Workforce Issues, said she expects the remaining open slots to be filled.
“Usually what happens is something called the scramble, which is after the match is over, [the National Resident Matching Program] has a clearinghouse for fellows who haven’t matched and programs that still have slots,” she said in an interview. “It connects them together. I would guess the positions will either be filled or that they are positions that are really targeted, say, for research applicants and there wasn’t anybody appropriate for that position.”
“You do want some excess because not all applicants are necessarily qualified, so you don’t want to have just the right amount of spots if you want to have the best people filling them,” she said, noting that this year, like last, there was around 100 applicants who listed rheumatology as their first choice that did not get assigned as fellowship, about double the number from a few years ago. “At the same time, with the anticipated workforce shortage, what this is telling us is if we created more fellowship slots, we have lots and lots of applicants to fill those slots.”
Dr. Bass said funding issues were the primary stumbling block to training more fellows. She also said more needs to be done to address geographic disparities, as these programs tend to be at major academic institutions in dense urban areas and many fellows practice at sights where they are trained, magnifying shortages in rural areas.
The similarities between last year’s rheumatology fellowship match results and this year’s are in line with what is happening in other specialties.
“Overall, the percentages of programs and positions filled were almost identical to last year,” Mona Singer, president and CEO of the National Resident Matching Program, said. “The same fellowships were competitive this year when compared to last year.”
Specialties filling more than 90% of positions included cardiovascular disease, endocrinology, gastroenterology, hematology/oncology, pulmonary/critical care, and rheumatology.
Ms. Singer noted that infectious diseases and nephrology “have implemented the ‘all-in policy,’ which requires any program participating in the match to place all positions. This was the first year infectious diseases used the policy, and there was a modest improvement in the number of positions filled. Nephrology was unchanged.”
Overall, 4,831 fellowships were available and 4,242 filled. The 87.8% of positions filled is a slight uptick over the 87.5% of positions filled in 2016. A total of 1,249 applicants did not match.
Cellular Versus Acellular Grafts for Diabetic Foot Ulcers: Altering the Protocol to Improve Recruitment to a Comparative Efficacy Trial
Chronic diabetic foot ulcers (DFUs) remain a serious therapeutic challenge worldwide.1-2 Patients with DFUs are at higher risk for infections, which may lead to limb loss.1-5 In fact, 1 in 6 patients with DFUs will undergo an amputation.6 The long-term consequences of DFUs are numerous and can severely affect patients’ quality of life, including loss of productivity.7 The current standard of care for DFUs consists of debridement of the necrotic tissue, application of a moist dressing, and use of an off-loading device that protects the wound from pressure or trauma related to ambulation and other acts of daily living.4-6,8 Unfortunately, studies have shown that the best standard of care (SOC) only heals 30% of DFUs after 20 weeks of therapy.9 With the estimated cost per episode of care approaching $40,000, DFUs remain a costly and important problem.10
The altered extracellular matrix (ECM) in DFUs has been a target for the development of new therapeutic devices that provide a new matrix that is either devoid of cells or can be enriched with fibroblasts.8,11 These bioengineered skin substitutes stimulate the growth of new vessels and generate cytokines essential for tissue repair. In 2013, Lev-Tov et al12 published this study protocol (Dermagraft Oasis Longitudinal Comparative Efficacy [DOLCE] trial) to compare the effectiveness of 2 advanced wound care devices, specifically to evaluate the clinical efficacy of a cellular matrix versus an acellular matrix, which we have amended. The cellular matrix used in the study is a dermal substitute composed of viable newborn foreskin fibroblasts seeded onto a bioabsorbable polyglactin mesh on which fibroblasts generate an ECM.13,14 It is supplied frozen and requires specific thawing steps prior to application. The recommended regimen for treatment of DFUs for this cellular matrix is 8 weekly applications.13,14 In 2016, the cost of the product was reported as $1411 per 5.0×7.5-cm sheet.15 The acellular matrix product used in the study is a bioabsorbable ECM that is derived from porcine small intestinal submucosa.16,17 It is stored at room temperature and has a long shelf life, with a current price of $112.6 for a 3.0×3.5-cm single-layer fenestrated sheet ($1126.60 per box of 10 sheets). The industry-supported randomized controlled trials for each of these devices have reported a 20% added benefit in the rate of wound closure at week 12 compared to SOC.14,17
This article provides the interim report of the trial (registered at www.clinicaltrials.gov with the identifier NCT01450943) described in the published protocol and initiated in 2011,12 focusing on elements that required modification during the trial’s duration.
Methods
Study Protocol
The clinical trial was approved by the Veterans’ Affairs Institutional Research and Development Committee and their institutional review board. This study was funded by the Veteran’s Administration Merit Award (#10554640), which was awarded to 2 of the investigators (S.E.D. and R.R.I.). Eligible veterans were recruited from all 7 sites of the VA Northern California Healthcare System. This trial is a randomized, single-blinded, 3-armed, controlled clinical equivalence trial comparing the effectiveness of an SOC treatment, cellular ECM, and acellular ECM.
Study Products
The SOC dressing applied in the clinical trial included a sterile antimicrobial gel, a nonadherent dressing, and gauze.12 The SOC dressing also was used as a secondary dressing for the active treatment arms. Bacitracin antibiotic ointment was used as an alternative for patients with allergy to iodine.12
Randomization
The inclusion and exclusion criteria were previously outlined.12 After a 2-week screening phase to exclude rapid healers, patients were randomized into a treatment arm and entered the active phase for 12 weeks.
Primary and Secondary Outcomes
The primary outcome was complete wound closure by week 12.12 Complete healing was defined as full reepithelialization with no drainage or dressing requirement. The secondary outcomes included healing at 28 weeks, rate of healing, ulcer recurrence at week 20, association of wound healing with ulcer characteristics or patients’ characteristic, incidence of adverse events, and cost-effectiveness of each treatment compared to the SOC arm.12
Statistical Analysis
To detect a 25% difference in the incidence of ulcer closure between the 2 study groups and the SOC group, the estimation of the sample size was based on 80% power with a significance level of 0.05. Specifically, it was expected that 50% of the cellular and acellular matrix groups and 25% of the SOC group would reach complete wound closure. The protocol indicated that 57 participants would be enrolled in each arm (total of 171 participants). Lev-Tov et al12 discussed the statistical analysis in more detail.
Results
Study Protocol Amendments
Given the number of diabetic patients in the US veteran population, we anticipated that there would be enough participants meeting the inclusion and exclusion criteria; however, because of the difficulty with recruitment, the initial study criteria were modified. The study was initially designed to incorporate DFUs with a minimum size of 1.0 cm2.12
Another limiting criterion was the percentage of total hemoglobin level for hemoglobin A1C (HbA1C). The study was originally established to include participants with an HbA1C level of 10% of total hemoglobin or below.12 Unfortunately, the majority of the potential participants had values substantially higher, and thus could not be enrolled in the trial, requiring another amendment to the study protocol in 2014, which was approved to include patients with an HbA1C level less than 12% of total hemoglobin. This change contributed considerably to the noted increase in enrollment rates in 2015, which almost doubled relative to enrollment under the original exclusion criteria (Figure).
The study has screened more than 600 patients. Among them, 137 were assessed for eligibility; 71 were excluded for various reasons, including screen failure (eg, decrease in wound size by >40% during the 2-week screening phase), loss to follow-up, and adverse events. Sixty-six participants reached the primary outcome at week 12, while 55 participants completed the study (19 in the SOC group; 18 in the cellular matrix group; 18 in the acellular matrix group).
We have stopped enrolling patients from all sites and the community, as we have reached our target enrollment.
Comment
One of the challenges of clinical trials is the recruitment of an adequate number of participants within an appropriate time frame, which is explained by Lasagna’s Law,18 a well-described phenomenon whereby the investigator overestimates the number of potential participants available to meet the inclusion criteria. This so-called funnel-effect was partly encountered in our selection process. A review of the veteran population with DFUs seemed to be more than adequate to fulfill the sample size; however, some important participant-related factors also played a substantial role.
In addition, the Veterans’ Affairs network centralizes health information, making it readily available to all providers participating in their care. As a result, patients with diabetes mellitus typically are seen by a primary care physician along with an endocrinologist, a diabetic nurse, and/or a dietician. Despite the collaboration with an interdisciplinary team, the glycemic control of the participants remains an issue along with other psychosocial factors that are deterrents in patient compliance. As a result, patients with poorly controlled diabetes and an HbA1C level above 10% (and less than 12%) of total hemoglobin who were initially excluded from the study were reincluded after modifying the inclusion criteria. Some patients were interested in joining the study, but physical limitations (eg, impaired mobility) prompted their decision not to join the trial, even though they met all the inclusion criteria.
As far as research-related factors that could affect participation, it is notable that most of the patients were retired; thus, the interventions did not cause additional burden of taking time off from work or loss of productivity. Although randomization could be a deterrent in many clinical trials, the majority of patients were willing to participate without demanding to be assigned to a particular treatment group.
There are many factors that are intertwined and can lead to enrollment and/or attrition rates. It was critical for our team to make some adjustment without compromising the controlled nature of a randomized trial.
Acknowledgment
The authors wish to acknowledge Huong Le, DPM, MPH, who was the coauthor of the study protocol.
- Sen CK, Gordillo GM, Roy S, et al. Human skin wounds: a major and snowballing threat to public health and the economy. Wound Repair Regen. 2009;17:763-771.
- Gurtner GC, Werner S, Barrandon Y, et al. Wound repair and regeneration. Nature. 2008;453:314-321.
- Falanga V. Wound healing and its impairment in the diabetic foot. Lancet. 2005;366:1736-1743.
- Boulton AJ. The diabetic foot: grand overview, epidemiology and pathogenesis. Diabetes Metab Res Rev. 2008;24(suppl 1):S3-S6.
- Singh N, Armstrong DG, Lipsky BA. Preventing foot ulcers in patients with diabetes. JAMA. 2005;293:217-228.
- Vuorisalo S, Venermo M, Lepäntalo M. Treatment of diabetic foot ulcers. J Cardiovasc Surg (Torino). 2009;50:275-291.
- Meijer JW, Trip J, Jaegers SM, et al. Quality of life in patients with diabetic foot ulcers. Disabil Rehabil. 2001;23:336-340.
- Santema TB, Poyck PP, Ubbink DT. Skin grafting and tissue replacement for treating foot ulcers in people with diabetes. Cochrane Database Syst Rev. 2016;2:CD011255.
- Margolis DJ, Kantor J, Berlin JA. Healing of diabetic neuropathic foot ulcers receiving standard treatment. a meta-analysis. Diabetes Care. 1999;22:692-695.
- Cavanagh P, Attinger C, Abbas Z, et al. Cost of treating diabetic foot ulcers in five different countries. Diabetes Metab Res Rev. 2012;2(suppl 1):107-111.
- Panuncialman J, Falanga V. The science of wound bed preparation. Surg Clin North Am. 2009;89:611-626.
- Lev-Tov H, Li CS, Dahle S, et al. Cellular versus acellular matrix devices in treatment of diabetic foot ulcers: study protocol for a comparative efficacy randomized controlled trial. Trials. 2013;14:8.
- Gentzkow GD, Iwasaki SD, Hershon KS, et al. Use of dermagraft, a cultured human dermis, to treat diabetic foot ulcers. Diabetes Care. 1996;19:350-354.
- Marston WA, Hanft J, Norwood P, et al; Dermagraft Diabetic Foot Ulcer Study Group. The efficacy and safety of Dermagraft in improving the healing of chronic diabetic foot ulcers: results of a prospective randomized trial. Diabetes Care. 2003;26:1701-1705.
- 2016 Dermagraft® Medicare Product and Related Procedure Payment. http://www.dermagraft.com/wp-content/uploads/sites/1/Dermagraft_Hotsheet%202016%20Q1%20HOSPITAL_FINAL.pdf. Accessed November 23, 2017.
- Oasis® Wound Matrix. http://www.oasiswoundmatrix.com/aboutowm. Accessed November 23, 2017.
- Niezgoda JA, Van Gils CC, Frykberg RG, et al. Randomized clinical trial comparing OASIS Wound Matrix to Regranex Gel for diabetic ulcers. Adv Skin Wound Care. 2005;18(5, pt 1):258-266.
- Torgerson JS, Arlinger K, Käppi M, et al. Principles for enhanced recruitment of subjects in a large clinical trial. the XENDOS (XENical in the prevention of Diabetes in Obese Subjects) study experience. Controlled Clin Trials. 2001;22:515-525.
Chronic diabetic foot ulcers (DFUs) remain a serious therapeutic challenge worldwide.1-2 Patients with DFUs are at higher risk for infections, which may lead to limb loss.1-5 In fact, 1 in 6 patients with DFUs will undergo an amputation.6 The long-term consequences of DFUs are numerous and can severely affect patients’ quality of life, including loss of productivity.7 The current standard of care for DFUs consists of debridement of the necrotic tissue, application of a moist dressing, and use of an off-loading device that protects the wound from pressure or trauma related to ambulation and other acts of daily living.4-6,8 Unfortunately, studies have shown that the best standard of care (SOC) only heals 30% of DFUs after 20 weeks of therapy.9 With the estimated cost per episode of care approaching $40,000, DFUs remain a costly and important problem.10
The altered extracellular matrix (ECM) in DFUs has been a target for the development of new therapeutic devices that provide a new matrix that is either devoid of cells or can be enriched with fibroblasts.8,11 These bioengineered skin substitutes stimulate the growth of new vessels and generate cytokines essential for tissue repair. In 2013, Lev-Tov et al12 published this study protocol (Dermagraft Oasis Longitudinal Comparative Efficacy [DOLCE] trial) to compare the effectiveness of 2 advanced wound care devices, specifically to evaluate the clinical efficacy of a cellular matrix versus an acellular matrix, which we have amended. The cellular matrix used in the study is a dermal substitute composed of viable newborn foreskin fibroblasts seeded onto a bioabsorbable polyglactin mesh on which fibroblasts generate an ECM.13,14 It is supplied frozen and requires specific thawing steps prior to application. The recommended regimen for treatment of DFUs for this cellular matrix is 8 weekly applications.13,14 In 2016, the cost of the product was reported as $1411 per 5.0×7.5-cm sheet.15 The acellular matrix product used in the study is a bioabsorbable ECM that is derived from porcine small intestinal submucosa.16,17 It is stored at room temperature and has a long shelf life, with a current price of $112.6 for a 3.0×3.5-cm single-layer fenestrated sheet ($1126.60 per box of 10 sheets). The industry-supported randomized controlled trials for each of these devices have reported a 20% added benefit in the rate of wound closure at week 12 compared to SOC.14,17
This article provides the interim report of the trial (registered at www.clinicaltrials.gov with the identifier NCT01450943) described in the published protocol and initiated in 2011,12 focusing on elements that required modification during the trial’s duration.
Methods
Study Protocol
The clinical trial was approved by the Veterans’ Affairs Institutional Research and Development Committee and their institutional review board. This study was funded by the Veteran’s Administration Merit Award (#10554640), which was awarded to 2 of the investigators (S.E.D. and R.R.I.). Eligible veterans were recruited from all 7 sites of the VA Northern California Healthcare System. This trial is a randomized, single-blinded, 3-armed, controlled clinical equivalence trial comparing the effectiveness of an SOC treatment, cellular ECM, and acellular ECM.
Study Products
The SOC dressing applied in the clinical trial included a sterile antimicrobial gel, a nonadherent dressing, and gauze.12 The SOC dressing also was used as a secondary dressing for the active treatment arms. Bacitracin antibiotic ointment was used as an alternative for patients with allergy to iodine.12
Randomization
The inclusion and exclusion criteria were previously outlined.12 After a 2-week screening phase to exclude rapid healers, patients were randomized into a treatment arm and entered the active phase for 12 weeks.
Primary and Secondary Outcomes
The primary outcome was complete wound closure by week 12.12 Complete healing was defined as full reepithelialization with no drainage or dressing requirement. The secondary outcomes included healing at 28 weeks, rate of healing, ulcer recurrence at week 20, association of wound healing with ulcer characteristics or patients’ characteristic, incidence of adverse events, and cost-effectiveness of each treatment compared to the SOC arm.12
Statistical Analysis
To detect a 25% difference in the incidence of ulcer closure between the 2 study groups and the SOC group, the estimation of the sample size was based on 80% power with a significance level of 0.05. Specifically, it was expected that 50% of the cellular and acellular matrix groups and 25% of the SOC group would reach complete wound closure. The protocol indicated that 57 participants would be enrolled in each arm (total of 171 participants). Lev-Tov et al12 discussed the statistical analysis in more detail.
Results
Study Protocol Amendments
Given the number of diabetic patients in the US veteran population, we anticipated that there would be enough participants meeting the inclusion and exclusion criteria; however, because of the difficulty with recruitment, the initial study criteria were modified. The study was initially designed to incorporate DFUs with a minimum size of 1.0 cm2.12
Another limiting criterion was the percentage of total hemoglobin level for hemoglobin A1C (HbA1C). The study was originally established to include participants with an HbA1C level of 10% of total hemoglobin or below.12 Unfortunately, the majority of the potential participants had values substantially higher, and thus could not be enrolled in the trial, requiring another amendment to the study protocol in 2014, which was approved to include patients with an HbA1C level less than 12% of total hemoglobin. This change contributed considerably to the noted increase in enrollment rates in 2015, which almost doubled relative to enrollment under the original exclusion criteria (Figure).

The study has screened more than 600 patients. Among them, 137 were assessed for eligibility; 71 were excluded for various reasons, including screen failure (eg, decrease in wound size by >40% during the 2-week screening phase), loss to follow-up, and adverse events. Sixty-six participants reached the primary outcome at week 12, while 55 participants completed the study (19 in the SOC group; 18 in the cellular matrix group; 18 in the acellular matrix group).
We have stopped enrolling patients from all sites and the community, as we have reached our target enrollment.
Comment
One of the challenges of clinical trials is the recruitment of an adequate number of participants within an appropriate time frame, which is explained by Lasagna’s Law,18 a well-described phenomenon whereby the investigator overestimates the number of potential participants available to meet the inclusion criteria. This so-called funnel-effect was partly encountered in our selection process. A review of the veteran population with DFUs seemed to be more than adequate to fulfill the sample size; however, some important participant-related factors also played a substantial role.
In addition, the Veterans’ Affairs network centralizes health information, making it readily available to all providers participating in their care. As a result, patients with diabetes mellitus typically are seen by a primary care physician along with an endocrinologist, a diabetic nurse, and/or a dietician. Despite the collaboration with an interdisciplinary team, the glycemic control of the participants remains an issue along with other psychosocial factors that are deterrents in patient compliance. As a result, patients with poorly controlled diabetes and an HbA1C level above 10% (and less than 12%) of total hemoglobin who were initially excluded from the study were reincluded after modifying the inclusion criteria. Some patients were interested in joining the study, but physical limitations (eg, impaired mobility) prompted their decision not to join the trial, even though they met all the inclusion criteria.
As far as research-related factors that could affect participation, it is notable that most of the patients were retired; thus, the interventions did not cause additional burden of taking time off from work or loss of productivity. Although randomization could be a deterrent in many clinical trials, the majority of patients were willing to participate without demanding to be assigned to a particular treatment group.
There are many factors that are intertwined and can lead to enrollment and/or attrition rates. It was critical for our team to make some adjustment without compromising the controlled nature of a randomized trial.
Acknowledgment
The authors wish to acknowledge Huong Le, DPM, MPH, who was the coauthor of the study protocol.
Chronic diabetic foot ulcers (DFUs) remain a serious therapeutic challenge worldwide.1-2 Patients with DFUs are at higher risk for infections, which may lead to limb loss.1-5 In fact, 1 in 6 patients with DFUs will undergo an amputation.6 The long-term consequences of DFUs are numerous and can severely affect patients’ quality of life, including loss of productivity.7 The current standard of care for DFUs consists of debridement of the necrotic tissue, application of a moist dressing, and use of an off-loading device that protects the wound from pressure or trauma related to ambulation and other acts of daily living.4-6,8 Unfortunately, studies have shown that the best standard of care (SOC) only heals 30% of DFUs after 20 weeks of therapy.9 With the estimated cost per episode of care approaching $40,000, DFUs remain a costly and important problem.10
The altered extracellular matrix (ECM) in DFUs has been a target for the development of new therapeutic devices that provide a new matrix that is either devoid of cells or can be enriched with fibroblasts.8,11 These bioengineered skin substitutes stimulate the growth of new vessels and generate cytokines essential for tissue repair. In 2013, Lev-Tov et al12 published this study protocol (Dermagraft Oasis Longitudinal Comparative Efficacy [DOLCE] trial) to compare the effectiveness of 2 advanced wound care devices, specifically to evaluate the clinical efficacy of a cellular matrix versus an acellular matrix, which we have amended. The cellular matrix used in the study is a dermal substitute composed of viable newborn foreskin fibroblasts seeded onto a bioabsorbable polyglactin mesh on which fibroblasts generate an ECM.13,14 It is supplied frozen and requires specific thawing steps prior to application. The recommended regimen for treatment of DFUs for this cellular matrix is 8 weekly applications.13,14 In 2016, the cost of the product was reported as $1411 per 5.0×7.5-cm sheet.15 The acellular matrix product used in the study is a bioabsorbable ECM that is derived from porcine small intestinal submucosa.16,17 It is stored at room temperature and has a long shelf life, with a current price of $112.6 for a 3.0×3.5-cm single-layer fenestrated sheet ($1126.60 per box of 10 sheets). The industry-supported randomized controlled trials for each of these devices have reported a 20% added benefit in the rate of wound closure at week 12 compared to SOC.14,17
This article provides the interim report of the trial (registered at www.clinicaltrials.gov with the identifier NCT01450943) described in the published protocol and initiated in 2011,12 focusing on elements that required modification during the trial’s duration.
Methods
Study Protocol
The clinical trial was approved by the Veterans’ Affairs Institutional Research and Development Committee and their institutional review board. This study was funded by the Veteran’s Administration Merit Award (#10554640), which was awarded to 2 of the investigators (S.E.D. and R.R.I.). Eligible veterans were recruited from all 7 sites of the VA Northern California Healthcare System. This trial is a randomized, single-blinded, 3-armed, controlled clinical equivalence trial comparing the effectiveness of an SOC treatment, cellular ECM, and acellular ECM.
Study Products
The SOC dressing applied in the clinical trial included a sterile antimicrobial gel, a nonadherent dressing, and gauze.12 The SOC dressing also was used as a secondary dressing for the active treatment arms. Bacitracin antibiotic ointment was used as an alternative for patients with allergy to iodine.12
Randomization
The inclusion and exclusion criteria were previously outlined.12 After a 2-week screening phase to exclude rapid healers, patients were randomized into a treatment arm and entered the active phase for 12 weeks.
Primary and Secondary Outcomes
The primary outcome was complete wound closure by week 12.12 Complete healing was defined as full reepithelialization with no drainage or dressing requirement. The secondary outcomes included healing at 28 weeks, rate of healing, ulcer recurrence at week 20, association of wound healing with ulcer characteristics or patients’ characteristic, incidence of adverse events, and cost-effectiveness of each treatment compared to the SOC arm.12
Statistical Analysis
To detect a 25% difference in the incidence of ulcer closure between the 2 study groups and the SOC group, the estimation of the sample size was based on 80% power with a significance level of 0.05. Specifically, it was expected that 50% of the cellular and acellular matrix groups and 25% of the SOC group would reach complete wound closure. The protocol indicated that 57 participants would be enrolled in each arm (total of 171 participants). Lev-Tov et al12 discussed the statistical analysis in more detail.
Results
Study Protocol Amendments
Given the number of diabetic patients in the US veteran population, we anticipated that there would be enough participants meeting the inclusion and exclusion criteria; however, because of the difficulty with recruitment, the initial study criteria were modified. The study was initially designed to incorporate DFUs with a minimum size of 1.0 cm2.12
Another limiting criterion was the percentage of total hemoglobin level for hemoglobin A1C (HbA1C). The study was originally established to include participants with an HbA1C level of 10% of total hemoglobin or below.12 Unfortunately, the majority of the potential participants had values substantially higher, and thus could not be enrolled in the trial, requiring another amendment to the study protocol in 2014, which was approved to include patients with an HbA1C level less than 12% of total hemoglobin. This change contributed considerably to the noted increase in enrollment rates in 2015, which almost doubled relative to enrollment under the original exclusion criteria (Figure).

The study has screened more than 600 patients. Among them, 137 were assessed for eligibility; 71 were excluded for various reasons, including screen failure (eg, decrease in wound size by >40% during the 2-week screening phase), loss to follow-up, and adverse events. Sixty-six participants reached the primary outcome at week 12, while 55 participants completed the study (19 in the SOC group; 18 in the cellular matrix group; 18 in the acellular matrix group).
We have stopped enrolling patients from all sites and the community, as we have reached our target enrollment.
Comment
One of the challenges of clinical trials is the recruitment of an adequate number of participants within an appropriate time frame, which is explained by Lasagna’s Law,18 a well-described phenomenon whereby the investigator overestimates the number of potential participants available to meet the inclusion criteria. This so-called funnel-effect was partly encountered in our selection process. A review of the veteran population with DFUs seemed to be more than adequate to fulfill the sample size; however, some important participant-related factors also played a substantial role.
In addition, the Veterans’ Affairs network centralizes health information, making it readily available to all providers participating in their care. As a result, patients with diabetes mellitus typically are seen by a primary care physician along with an endocrinologist, a diabetic nurse, and/or a dietician. Despite the collaboration with an interdisciplinary team, the glycemic control of the participants remains an issue along with other psychosocial factors that are deterrents in patient compliance. As a result, patients with poorly controlled diabetes and an HbA1C level above 10% (and less than 12%) of total hemoglobin who were initially excluded from the study were reincluded after modifying the inclusion criteria. Some patients were interested in joining the study, but physical limitations (eg, impaired mobility) prompted their decision not to join the trial, even though they met all the inclusion criteria.
As far as research-related factors that could affect participation, it is notable that most of the patients were retired; thus, the interventions did not cause additional burden of taking time off from work or loss of productivity. Although randomization could be a deterrent in many clinical trials, the majority of patients were willing to participate without demanding to be assigned to a particular treatment group.
There are many factors that are intertwined and can lead to enrollment and/or attrition rates. It was critical for our team to make some adjustment without compromising the controlled nature of a randomized trial.
Acknowledgment
The authors wish to acknowledge Huong Le, DPM, MPH, who was the coauthor of the study protocol.
- Sen CK, Gordillo GM, Roy S, et al. Human skin wounds: a major and snowballing threat to public health and the economy. Wound Repair Regen. 2009;17:763-771.
- Gurtner GC, Werner S, Barrandon Y, et al. Wound repair and regeneration. Nature. 2008;453:314-321.
- Falanga V. Wound healing and its impairment in the diabetic foot. Lancet. 2005;366:1736-1743.
- Boulton AJ. The diabetic foot: grand overview, epidemiology and pathogenesis. Diabetes Metab Res Rev. 2008;24(suppl 1):S3-S6.
- Singh N, Armstrong DG, Lipsky BA. Preventing foot ulcers in patients with diabetes. JAMA. 2005;293:217-228.
- Vuorisalo S, Venermo M, Lepäntalo M. Treatment of diabetic foot ulcers. J Cardiovasc Surg (Torino). 2009;50:275-291.
- Meijer JW, Trip J, Jaegers SM, et al. Quality of life in patients with diabetic foot ulcers. Disabil Rehabil. 2001;23:336-340.
- Santema TB, Poyck PP, Ubbink DT. Skin grafting and tissue replacement for treating foot ulcers in people with diabetes. Cochrane Database Syst Rev. 2016;2:CD011255.
- Margolis DJ, Kantor J, Berlin JA. Healing of diabetic neuropathic foot ulcers receiving standard treatment. a meta-analysis. Diabetes Care. 1999;22:692-695.
- Cavanagh P, Attinger C, Abbas Z, et al. Cost of treating diabetic foot ulcers in five different countries. Diabetes Metab Res Rev. 2012;2(suppl 1):107-111.
- Panuncialman J, Falanga V. The science of wound bed preparation. Surg Clin North Am. 2009;89:611-626.
- Lev-Tov H, Li CS, Dahle S, et al. Cellular versus acellular matrix devices in treatment of diabetic foot ulcers: study protocol for a comparative efficacy randomized controlled trial. Trials. 2013;14:8.
- Gentzkow GD, Iwasaki SD, Hershon KS, et al. Use of dermagraft, a cultured human dermis, to treat diabetic foot ulcers. Diabetes Care. 1996;19:350-354.
- Marston WA, Hanft J, Norwood P, et al; Dermagraft Diabetic Foot Ulcer Study Group. The efficacy and safety of Dermagraft in improving the healing of chronic diabetic foot ulcers: results of a prospective randomized trial. Diabetes Care. 2003;26:1701-1705.
- 2016 Dermagraft® Medicare Product and Related Procedure Payment. http://www.dermagraft.com/wp-content/uploads/sites/1/Dermagraft_Hotsheet%202016%20Q1%20HOSPITAL_FINAL.pdf. Accessed November 23, 2017.
- Oasis® Wound Matrix. http://www.oasiswoundmatrix.com/aboutowm. Accessed November 23, 2017.
- Niezgoda JA, Van Gils CC, Frykberg RG, et al. Randomized clinical trial comparing OASIS Wound Matrix to Regranex Gel for diabetic ulcers. Adv Skin Wound Care. 2005;18(5, pt 1):258-266.
- Torgerson JS, Arlinger K, Käppi M, et al. Principles for enhanced recruitment of subjects in a large clinical trial. the XENDOS (XENical in the prevention of Diabetes in Obese Subjects) study experience. Controlled Clin Trials. 2001;22:515-525.
- Sen CK, Gordillo GM, Roy S, et al. Human skin wounds: a major and snowballing threat to public health and the economy. Wound Repair Regen. 2009;17:763-771.
- Gurtner GC, Werner S, Barrandon Y, et al. Wound repair and regeneration. Nature. 2008;453:314-321.
- Falanga V. Wound healing and its impairment in the diabetic foot. Lancet. 2005;366:1736-1743.
- Boulton AJ. The diabetic foot: grand overview, epidemiology and pathogenesis. Diabetes Metab Res Rev. 2008;24(suppl 1):S3-S6.
- Singh N, Armstrong DG, Lipsky BA. Preventing foot ulcers in patients with diabetes. JAMA. 2005;293:217-228.
- Vuorisalo S, Venermo M, Lepäntalo M. Treatment of diabetic foot ulcers. J Cardiovasc Surg (Torino). 2009;50:275-291.
- Meijer JW, Trip J, Jaegers SM, et al. Quality of life in patients with diabetic foot ulcers. Disabil Rehabil. 2001;23:336-340.
- Santema TB, Poyck PP, Ubbink DT. Skin grafting and tissue replacement for treating foot ulcers in people with diabetes. Cochrane Database Syst Rev. 2016;2:CD011255.
- Margolis DJ, Kantor J, Berlin JA. Healing of diabetic neuropathic foot ulcers receiving standard treatment. a meta-analysis. Diabetes Care. 1999;22:692-695.
- Cavanagh P, Attinger C, Abbas Z, et al. Cost of treating diabetic foot ulcers in five different countries. Diabetes Metab Res Rev. 2012;2(suppl 1):107-111.
- Panuncialman J, Falanga V. The science of wound bed preparation. Surg Clin North Am. 2009;89:611-626.
- Lev-Tov H, Li CS, Dahle S, et al. Cellular versus acellular matrix devices in treatment of diabetic foot ulcers: study protocol for a comparative efficacy randomized controlled trial. Trials. 2013;14:8.
- Gentzkow GD, Iwasaki SD, Hershon KS, et al. Use of dermagraft, a cultured human dermis, to treat diabetic foot ulcers. Diabetes Care. 1996;19:350-354.
- Marston WA, Hanft J, Norwood P, et al; Dermagraft Diabetic Foot Ulcer Study Group. The efficacy and safety of Dermagraft in improving the healing of chronic diabetic foot ulcers: results of a prospective randomized trial. Diabetes Care. 2003;26:1701-1705.
- 2016 Dermagraft® Medicare Product and Related Procedure Payment. http://www.dermagraft.com/wp-content/uploads/sites/1/Dermagraft_Hotsheet%202016%20Q1%20HOSPITAL_FINAL.pdf. Accessed November 23, 2017.
- Oasis® Wound Matrix. http://www.oasiswoundmatrix.com/aboutowm. Accessed November 23, 2017.
- Niezgoda JA, Van Gils CC, Frykberg RG, et al. Randomized clinical trial comparing OASIS Wound Matrix to Regranex Gel for diabetic ulcers. Adv Skin Wound Care. 2005;18(5, pt 1):258-266.
- Torgerson JS, Arlinger K, Käppi M, et al. Principles for enhanced recruitment of subjects in a large clinical trial. the XENDOS (XENical in the prevention of Diabetes in Obese Subjects) study experience. Controlled Clin Trials. 2001;22:515-525.
Resident Pearl
- Deciding on the appropriate wound care regimen for diabetic foot ulcers is difficult given the vast amount of wound products on the market. This head-to-head clinical trial compared the use of an expensive cellular matrix and an inexpensive acellular matrix relative to the standard of care. We hope that this study will help to guide therapy based on cost-effectiveness of wound adjuncts without compromising patient care.
The ‘Virtual Radiology Resident’—Coming to a Computer Near You
Researchers around the world may be able to teach computers how to better detect and diagnose disease, thanks to > 100,000 chest x-ray images and corresponding data recently released by the NIH Clinical Center.
Reading and diagnosing chest x-rays requires careful observation, as well as knowledge of anatomy, physiology, and pathology. When that is combined with the need to consider all common thoracic diseases, it becomes hard to automate a consistent technique for reading images, the NIH says. With the free dataset, the hope is that academic and research institution staff will be able to teach their computers to read and process enormous amounts of scans, to confirm radiologists’ results, and potentially identify anything that may have been overlooked.
The NIH says in addition to being a “virtual radiology resident,” advanced computer technology has other potential benefits: For instance, it could identify slow changes occurring over the course of multiple chest x-rays that might otherwise be overlooked. The technology also would be useful in poor countries that lack radiologists. And in the future, the “resident” might be taught to read more complex images, such as CT and MRI.
The dataset, compiled from scans from > 30,000 patients, including many with advanced lung disease, was scrubbed of private information before release. The images are available via Box at https://nihcc.app.box.com/v/ChestXray-NIHCC.
Researchers around the world may be able to teach computers how to better detect and diagnose disease, thanks to > 100,000 chest x-ray images and corresponding data recently released by the NIH Clinical Center.
Reading and diagnosing chest x-rays requires careful observation, as well as knowledge of anatomy, physiology, and pathology. When that is combined with the need to consider all common thoracic diseases, it becomes hard to automate a consistent technique for reading images, the NIH says. With the free dataset, the hope is that academic and research institution staff will be able to teach their computers to read and process enormous amounts of scans, to confirm radiologists’ results, and potentially identify anything that may have been overlooked.
The NIH says in addition to being a “virtual radiology resident,” advanced computer technology has other potential benefits: For instance, it could identify slow changes occurring over the course of multiple chest x-rays that might otherwise be overlooked. The technology also would be useful in poor countries that lack radiologists. And in the future, the “resident” might be taught to read more complex images, such as CT and MRI.
The dataset, compiled from scans from > 30,000 patients, including many with advanced lung disease, was scrubbed of private information before release. The images are available via Box at https://nihcc.app.box.com/v/ChestXray-NIHCC.
Researchers around the world may be able to teach computers how to better detect and diagnose disease, thanks to > 100,000 chest x-ray images and corresponding data recently released by the NIH Clinical Center.
Reading and diagnosing chest x-rays requires careful observation, as well as knowledge of anatomy, physiology, and pathology. When that is combined with the need to consider all common thoracic diseases, it becomes hard to automate a consistent technique for reading images, the NIH says. With the free dataset, the hope is that academic and research institution staff will be able to teach their computers to read and process enormous amounts of scans, to confirm radiologists’ results, and potentially identify anything that may have been overlooked.
The NIH says in addition to being a “virtual radiology resident,” advanced computer technology has other potential benefits: For instance, it could identify slow changes occurring over the course of multiple chest x-rays that might otherwise be overlooked. The technology also would be useful in poor countries that lack radiologists. And in the future, the “resident” might be taught to read more complex images, such as CT and MRI.
The dataset, compiled from scans from > 30,000 patients, including many with advanced lung disease, was scrubbed of private information before release. The images are available via Box at https://nihcc.app.box.com/v/ChestXray-NIHCC.