User login
Boston VA Medical Forum: HIV-Positive Veteran With Progressive Visual Changes
►Lakshmana Swamy, MD, chief medical resident, VA Boston Healthcare System (VABHS) and Boston Medical Center. Dr. Serrao, when you hear about vision changes in a patient with HIV, what differential diagnosis is generated? What epidemiologic or historical factors can help distinguish these entities?
►Richard Serrao, MD, Infectious Disease Service, VABHS and assistant professor of medicine, Boston University School of Medicine. The differential diagnoses for vision changes in a patient with HIV is based on the overall immunosuppression of the patient: the lower the patient’s CD4 count, the higher the number of etiologies.1 The portions of the visual pathway as well as the pattern of vision loss are useful in narrowing the differential. For example, monocular visual disturbances with dermatomal vesicles within the ophthalmic division of the trigeminal nerve strongly implicates varicella zoster retinitis or keratitis; abducens nerve palsy could suggest granulomatous basilar meningitis from cryptococcosis. Likewise, ongoing fevers in an advanced AIDS patient with concomitant colitis, hepatitis, and pneumonitis is strongly suspicious for cytomegalovirus (CMV) retinitis with wide dissemination.
Geographic epidemiologic factors can suggest pathogens more prevalent to certain regions of the world, such as histoplasma chorioretinitis in a resident of the central and eastern U.S. or tuberculosis in a returning traveler. Likewise, a cat owner or one who consumes steak tartare increases the likelihood for toxoplasma retinochoroiditis, or syphilis in men who have sex with men (MSM) in the U.S. given that the majority of new cases occur in this patient population. Other clues one should consider include the presence of splinter hemorrhages in the extremities in an intravenous drug user, raising the possibility of embolic endophthalmitis from bacterial or fungal endocarditis. A variety of other diagnoses can certainly occur as a result of drug treatment (uveitis from rifampin, for example), immune reconstitution from HAART, infections with other HIV-associated pathogens, such as Pneumocystis jiroveci, and many non-HIV-related ocular diseases.
►Dr. Swamy. Dr. Butler, what concerns do you have when you hear about an HIV-infected patient with vision loss from the ophthalmology perspective?
►Nicholas Butler, MD, Ophthalmology Service, Uveitis and Ocular Immunology, VABHS and assistant professor of ophthalmology, Harvard Medical School. Of course, patients with HIV suffer from common causes of vision loss—cataract, glaucoma, diabetes, macular degeneration, for instance—just like those without HIV infection. If there is no significant immunodeficiency, then the patient’s HIV status would be less relevant, and these more common causes of vision loss should be pursued. My first task would be to determine the patient’s most recent CD4 T-cell count.
Assuming an HIV-positive individual is experiencing visual symptoms related to his/her underlying HIV infection (especially in the setting of CD4 counts < 200 cells/mm3), ocular opportunistic infections (OOI) come to mind first. Despite a reduction in incidence of 75% to 80% in the HAART-era, CMV retinitis remains the most common OOI in patients with AIDS and carries the greatest risk of ocular morbidity.2 In fact, based on enrollment data for the Longitudinal Study of the Ocular Complications of AIDS (LSOCA), the prevalence of CMV retinitis among patients with AIDS is more than 20-fold higher than all other ocular complications of AIDS (OOIs and ocular neoplastic disease), including Kaposi sarcoma, lymphoma, herpes zoster ophthalmicus, ocular syphilis, ocular toxoplasma, necrotizing herpetic retinitis, cryptococcal choroiditis, and pneumocystis choroiditis.3 Beyond ocular opportunistic infections, the most common retinal finding in HIV-positive people is HIV retinopathy, nonspecific microvascular findings in the retina affecting nearly 70% of those with advanced HIV disease. Fortunately, HIV retinopathy is generally asymptomatic.4
►Dr. Swamy. Thank you for those explanations. Based on Dr. Serrao’s differential, it is worth noting that this patient is MSM. He was evaluated in urgent care with the initial examination showing a temperature of 98.0° F, pulse 83 beats per minute, and blood pressure 110/70 mm Hg. The eye exam showed no injection with normal extraocular movements. Initial laboratory data were notable for a CD4 count of 730 cells/mm3 with fewer than 20 HIV viral copies/mL. Cytomegalovirus immunoglobulin G (IgG) was positive, and immunoglobulin M (IgM) was negative. A Lyme antibody was positive with negative IgM and IgG by Western blot. Additional tests can be seen in Tables 1 and 2. The patient has good immunologic and virologic control. How does this change your thinking about the case?
►Dr. Serrao. His CD4 count is well above 350, increasing the likelihood of a relatively uncomplicated course and treatment. Cytomegalovirus antibodies reflect prior infection. As CMV generally does not manifest with disease of any variety (including CMV retinitis) at this high CD4 count, one can presume he does not have CMV retinitis as a cause for his visual changes. CMV retinitis occurs mainly when substantial CD4 depletion has occurred (typically less than 50 cells/mm3). A positive Lyme antibody screen, not specific to Lyme, can be falsely positive in other treponema diseases (eg, Treponema pallidum, the etiologic organism of syphilis) as evidenced by negative confirmatory Western blot IgG and IgM. Antineutrophil cystoplasmic antibodies, lysozyme, angiotensin-converting enzyme, rapid plasma reagin (RPR), herpes simplex virus, toxoplasma are generally included in the workup for the differential of uveitis, retinitis, choroiditis, etc.
►Dr. Swamy. Based on the visual changes, the patient was referred for urgent ophthalmologic evaluation. Dr. Butler, when should a generalist consider urgent ophthalmology referral?
►Dr. Butler. In general, all patients with acute (and significant) vision loss should be referred immediately to an ophthalmologist. The challenge for the general practitioner is determining the true extent of the reported vision loss. If possible, some assessment of visual acuity should be obtained, testing each eye independently and with the correct glasses correction (ie, the patient’s distance glasses if the test object is 12 feet or more from the patient or their reading glasses if the test object is held inside arm’s length). If the general practitioner does not have access to an eye chart or near card, any assessment of vision with an appropriate description will be useful (eg, the patient can quickly count fingers at 15 feet in the unaffected eye, but the eye with reported vision loss cannot reliably count fingers outside of 2 feet). Additional ocular symptoms associated with the vision loss, such as pain, redness, photophobia, new flashes or floaters, increase the urgency of the referral. The threshold for referral for any ocular complaint is lower compared with that of the general population for those with evidence of immunodeficiency, such as for this patient with HIV. Any CD4 count < 200 cells/mm3 should raise the practitioner’s concern for an ocular opportunistic infection, with the greatest concern with CD4 counts < 50 cells/mm3.
►Dr. Swamy. The patient underwent further testing in the ophthalmology clinic. Dr. Butler, can you please interpret the funduscopic exam?
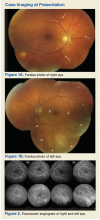
►Dr. Butler. Both eyes demonstrate findings (microaneurysms and small dot-blot hemorrhages) consistent with moderate nonproliferative diabetic retinopathy (Figure 1A, white arrows). HIV-associated retinopathy could produce similar findings, but it is not generally seen with CD4 counts > 200 cells/mm3. Additionally, in the left eye, there is a diffuse patch of retinal whitening (retinitis) associated with the inferotemporal vascular arcades (Figure 1B, white arrows). The entire area involved is poorly circumscribed and the whitening is subtle in areas. Overlying some areas of deeper, ground-glass whitening there are scattered, punctate white spots (Figure 1B, green arrows). Wickremasinghe and colleagues described this pattern of retinitis and suggested that it had a high positive-predictive value in the diagnosis of ocular syphilis.5
►Dr. Swamy. The patient then underwent fluorescein angiography and optical coherence tomography (OCT). Dr. Butler, what did the fluorescein angiography show?
►Dr. Butler. The fluorescein angiogram in both eyes revealed leakage of dye consistent with diabetic retinopathy, with the right eye (OD) worse than the left (OS). Additionally, the areas of active retinitis in the left eye displayed gradual staining with leopard-spot changes, along with late leakage of fluorescein dye, indicating vasculopathy in the infected area (Figure 2, arrows). The patient also underwent OCT in the left eye (images not displayed) demonstrating vitreous cells (vitritis), patches of inner retinal thickening with hyperreflectivity, and hyperreflective nodules at the level of the retinal pigment epithelium with overlying photoreceptor disruption. These OCT findings are fairly stereotypic for syphilitic chorioretinitis.6
►Dr. Swamy. Based on the ophthalmic findings, a diagnosis of ocular syphilis was made. Dr. Serrao, what should internists consider as they evaluate and manage a patient with ocular syphilis?
►Dr. Serrao. Although isolated ocular involvement from syphilis is possible, the majority of patients (up to 85%) with HIV can present with concomitant central nervous system infection and about 30% present with symptomatic neurosyphilis (a typical late manifestation of this disease) that reflects the aggressiveness, accelerated course and propensity for wide dissemination of syphilis in this patient population.7
The presence of concomitant cutaneous rashes should prompt universal precautions, because transmission can occur via skin to skin contact. Clinicians should watch for the Jarisch-Herxheimer reaction during treatment, a syndrome of fever, myalgias, and headache, which results from circulating cytokines produced because of rapidly dying spirochetes that could mimic a penicillin drug reaction, yet is treated supportively.
As syphilis is sexually acquired, clinicians should test for coexistent sexually transmitted infections, vaccinate for those that are preventable (eg, hepatitis B), notify sexual partners via assistance from local departments of public health, and assess for coexistent drug use and offer counseling in order to optimize risk reduction. Special attention should be paid to virologic control of HIV since some studies have shown an increase in the propensity for breakthrough HIV viremia while on effective ART.9 This should warrant counseling for ongoing optimal ART adherence and close monitoring in the follow-up visits with a provider specialized in the treatment of syphilis and HIV.
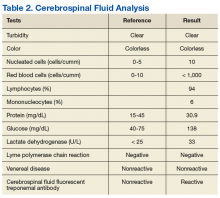
►Dr. Swamy. A lumbar puncture is performed with the results listed in Table 2. Dr. Serrao, is the CSF consistent with neurosyphilis? What would you do next?
►Dr. Serrao. The lumbar puncture is inflammatory with a lymphocytic predominance, consistent with active ocular/neurosyphilis. The CSF Venereal Disease Research Laboratory test is specific but not sensitive so a negative value does not rule out the presence of central nervous system infection.10 The CSF fluorescent treponemal antibody (CSF FTA-ABS) is sensitive but not specific. In this case, the ocular findings, positive serum RPR, CSF lymphocytic predominance, and CSF FTA ABS strongly supports the diagnosis of ocular/early neurosyphilis in a patient with HIV infection in whom early aggressive treatment is warranted to prevent rapid progression/potential loss of vision.11
►Dr. Swamy. Dr. Butler, how does syphilis behave in the eye as compared to other infectious or inflammatory diseases? Do visual symptoms respond well to treatment?
►Dr. Butler. As opposed to the dramatic reduction in rates and severity of CMV retinitis, HAART has had a negligible effect on ocular syphilis in the setting of HIV coinfection; in fact, rates of syphilis, including ocular syphilis, are currently surging world-wide, and HIV coinfection portends a worse prognosis.12 This is especially true among gay men. More so, there appears to be no correlation between CD4 count and incidence of developing ocular syphilis, as opposed to CMV retinitis, which occurs far more frequently in those with CD4 counts < 50 cells/mm3. In keeping with its epithet as one of the “Great Imitators,” syphilis can affect virtually every tissue of the eye—conjunctiva, sclera, cornea, iris, lens, vitreous, retina, choroid, optic nerve—unlike other OOI, such as CMV or toxoplasma, which generally hone to the retina. Nonetheless, various findings and patterns on clinical exam and ancillary testing, such as the more recently described punctate inner retinitis (as seen in our patient) and the more classic acute syphilitic posterior placoid chorioretinitis, carry high specificity for ocular syphilis.13
Patients with ocular syphilis should be treated according to neurosyphilis treatment protocols. In general, these patients respond very well to treatment with resolution of the ocular findings and recovery of complete, or nearly so, visual function, as long as an excessive delay between diagnosis and proper treatment does not occur.14
►Dr. Swamy. Following this testing, the patient completed 14 days of IV penicillin with resolution of symptoms. He had no further vision complaints. He was started on Triumeq (abacavir, dolutegravir, and lamivudine) with good adherence to therapy. Dr. Serrao, in 2016 the CDC released a clinical advisory about ocular syphilis. Can you tell us about why this is an important diagnosis to be aware of today?
►Dr. Serrao. As with any disease, the epidemiologic characteristics of an infection like syphilis allow the clinician to more carefully entertain such a diagnosis in any one individual by improving the index of suspicion for a particular disease. Awareness of an increase in ocular syphilis in HIV positive MSM allows for a more timely assessment and subsequent treatment with the goal of preventing loss of vision.15
1. Cunningham ET Jr, Margolis TP. Ocular manifestations of HIV infection. N Engl J Med. 1998;339(4):236-244.
2. Holtzer CD, Jacobson MA, Hadley WK, et al. Decline in the rate of specific opportunistic infections at San Francisco General Hospital, 1994-1997. AIDS. 1998;12(14):1931-1933.
3. Gangaputra S, Drye L, Vaidya V, Thorne JE, Jabs DA, Lyon AT. Non-cytomegalovirus ocular opportunistic infections in patients with acquired immunodeficiency syndrome. Am J Ophthalmol. 2013;155(2):206-212.e205.
4. Jabs DA, Van Natta ML, Holbrook JT, et al. Longitudinal study of the ocular complications of AIDS: 1. Ocular diagnoses at enrollment. Ophthalmology. 2007;114(4):780-786.
5. Wickremasinghe S, Ling C, Stawell R, Yeoh J, Hall A, Zamir E. Syphilitic punctate inner retinitis in immunocompetent gay men. Ophthalmology. 2009;116(6):1195-1200.
6. Burkholder BM, Leung TG, Ostheimer TA, Butler NJ, Thorne JE, Dunn JP. Spectral domain optical coherence tomography findings in acute syphilitic posterior placoid chorioretinitis. J Ophthalmic Inflamm Infect. 2014;4(1):2.
7. Musher DM, Hamill RJ, Baughn RE. Effect of human immunodeficiency virus (HIV) infection on the course of syphilis and on the response to treatment. Ann Intern Med. 1990;113(11):872-881.
8. Lukehart SA, Hook EW 3rd, Baker-Zander SA, Collier AC, Critchlow CW, Handsfield HH. Invasion of the central nervous system by Treponema pallidum: implications for diagnosis and treatment. Ann Intern Med. 1988;109(11):855-862.
9. Golden MR, Marra CM, Holmes KK. Update on syphilis: resurgence of an old problem. JAMA. 2003;290(11):1510-1514.
10. Marra CM, Tantalo LC, Maxwell CL, Ho EL, Sahi SK, Jones T. The rapid plasma reagin test cannot replace the venereal disease research laboratory test for neurosyphilis diagnosis. Sex Transm Dis. 2012;39(6):453-457.
11. Harding AS, Ghanem KG. The performance of cerebrospinal fluid treponemal-specific antibody tests in neurosyphilis: a systematic review. Sex Transm Dis. 2012;39(4):291-297.
12. Butler NJ, Thorne JE. Current status of HIV infection and ocular disease. Curr Opin Ophthalmol. 2012;23(6):517-522.
13. Gass JD, Braunstein RA, Chenoweth RG. Acute syphilitic posterior placoid chorioretinitis. Ophthalmology. 1990;97(10):1288-1297.
14. Davis JL. Ocular syphilis. Curr Opin Ophthalmol. 2014;25(6):513-518.
15. Clinical Advisory: Ocular Syphilis in the United States. https://www.cdc.gov/std/syphilis/clinicaladvisoryos2015.htm. Accessed September 11, 2017.
►Lakshmana Swamy, MD, chief medical resident, VA Boston Healthcare System (VABHS) and Boston Medical Center. Dr. Serrao, when you hear about vision changes in a patient with HIV, what differential diagnosis is generated? What epidemiologic or historical factors can help distinguish these entities?
►Richard Serrao, MD, Infectious Disease Service, VABHS and assistant professor of medicine, Boston University School of Medicine. The differential diagnoses for vision changes in a patient with HIV is based on the overall immunosuppression of the patient: the lower the patient’s CD4 count, the higher the number of etiologies.1 The portions of the visual pathway as well as the pattern of vision loss are useful in narrowing the differential. For example, monocular visual disturbances with dermatomal vesicles within the ophthalmic division of the trigeminal nerve strongly implicates varicella zoster retinitis or keratitis; abducens nerve palsy could suggest granulomatous basilar meningitis from cryptococcosis. Likewise, ongoing fevers in an advanced AIDS patient with concomitant colitis, hepatitis, and pneumonitis is strongly suspicious for cytomegalovirus (CMV) retinitis with wide dissemination.
Geographic epidemiologic factors can suggest pathogens more prevalent to certain regions of the world, such as histoplasma chorioretinitis in a resident of the central and eastern U.S. or tuberculosis in a returning traveler. Likewise, a cat owner or one who consumes steak tartare increases the likelihood for toxoplasma retinochoroiditis, or syphilis in men who have sex with men (MSM) in the U.S. given that the majority of new cases occur in this patient population. Other clues one should consider include the presence of splinter hemorrhages in the extremities in an intravenous drug user, raising the possibility of embolic endophthalmitis from bacterial or fungal endocarditis. A variety of other diagnoses can certainly occur as a result of drug treatment (uveitis from rifampin, for example), immune reconstitution from HAART, infections with other HIV-associated pathogens, such as Pneumocystis jiroveci, and many non-HIV-related ocular diseases.
►Dr. Swamy. Dr. Butler, what concerns do you have when you hear about an HIV-infected patient with vision loss from the ophthalmology perspective?
►Nicholas Butler, MD, Ophthalmology Service, Uveitis and Ocular Immunology, VABHS and assistant professor of ophthalmology, Harvard Medical School. Of course, patients with HIV suffer from common causes of vision loss—cataract, glaucoma, diabetes, macular degeneration, for instance—just like those without HIV infection. If there is no significant immunodeficiency, then the patient’s HIV status would be less relevant, and these more common causes of vision loss should be pursued. My first task would be to determine the patient’s most recent CD4 T-cell count.
Assuming an HIV-positive individual is experiencing visual symptoms related to his/her underlying HIV infection (especially in the setting of CD4 counts < 200 cells/mm3), ocular opportunistic infections (OOI) come to mind first. Despite a reduction in incidence of 75% to 80% in the HAART-era, CMV retinitis remains the most common OOI in patients with AIDS and carries the greatest risk of ocular morbidity.2 In fact, based on enrollment data for the Longitudinal Study of the Ocular Complications of AIDS (LSOCA), the prevalence of CMV retinitis among patients with AIDS is more than 20-fold higher than all other ocular complications of AIDS (OOIs and ocular neoplastic disease), including Kaposi sarcoma, lymphoma, herpes zoster ophthalmicus, ocular syphilis, ocular toxoplasma, necrotizing herpetic retinitis, cryptococcal choroiditis, and pneumocystis choroiditis.3 Beyond ocular opportunistic infections, the most common retinal finding in HIV-positive people is HIV retinopathy, nonspecific microvascular findings in the retina affecting nearly 70% of those with advanced HIV disease. Fortunately, HIV retinopathy is generally asymptomatic.4
►Dr. Swamy. Thank you for those explanations. Based on Dr. Serrao’s differential, it is worth noting that this patient is MSM. He was evaluated in urgent care with the initial examination showing a temperature of 98.0° F, pulse 83 beats per minute, and blood pressure 110/70 mm Hg. The eye exam showed no injection with normal extraocular movements. Initial laboratory data were notable for a CD4 count of 730 cells/mm3 with fewer than 20 HIV viral copies/mL. Cytomegalovirus immunoglobulin G (IgG) was positive, and immunoglobulin M (IgM) was negative. A Lyme antibody was positive with negative IgM and IgG by Western blot. Additional tests can be seen in Tables 1 and 2. The patient has good immunologic and virologic control. How does this change your thinking about the case?
►Dr. Serrao. His CD4 count is well above 350, increasing the likelihood of a relatively uncomplicated course and treatment. Cytomegalovirus antibodies reflect prior infection. As CMV generally does not manifest with disease of any variety (including CMV retinitis) at this high CD4 count, one can presume he does not have CMV retinitis as a cause for his visual changes. CMV retinitis occurs mainly when substantial CD4 depletion has occurred (typically less than 50 cells/mm3). A positive Lyme antibody screen, not specific to Lyme, can be falsely positive in other treponema diseases (eg, Treponema pallidum, the etiologic organism of syphilis) as evidenced by negative confirmatory Western blot IgG and IgM. Antineutrophil cystoplasmic antibodies, lysozyme, angiotensin-converting enzyme, rapid plasma reagin (RPR), herpes simplex virus, toxoplasma are generally included in the workup for the differential of uveitis, retinitis, choroiditis, etc.
►Dr. Swamy. Based on the visual changes, the patient was referred for urgent ophthalmologic evaluation. Dr. Butler, when should a generalist consider urgent ophthalmology referral?
►Dr. Butler. In general, all patients with acute (and significant) vision loss should be referred immediately to an ophthalmologist. The challenge for the general practitioner is determining the true extent of the reported vision loss. If possible, some assessment of visual acuity should be obtained, testing each eye independently and with the correct glasses correction (ie, the patient’s distance glasses if the test object is 12 feet or more from the patient or their reading glasses if the test object is held inside arm’s length). If the general practitioner does not have access to an eye chart or near card, any assessment of vision with an appropriate description will be useful (eg, the patient can quickly count fingers at 15 feet in the unaffected eye, but the eye with reported vision loss cannot reliably count fingers outside of 2 feet). Additional ocular symptoms associated with the vision loss, such as pain, redness, photophobia, new flashes or floaters, increase the urgency of the referral. The threshold for referral for any ocular complaint is lower compared with that of the general population for those with evidence of immunodeficiency, such as for this patient with HIV. Any CD4 count < 200 cells/mm3 should raise the practitioner’s concern for an ocular opportunistic infection, with the greatest concern with CD4 counts < 50 cells/mm3.
►Dr. Swamy. The patient underwent further testing in the ophthalmology clinic. Dr. Butler, can you please interpret the funduscopic exam?
►Dr. Butler. Both eyes demonstrate findings (microaneurysms and small dot-blot hemorrhages) consistent with moderate nonproliferative diabetic retinopathy (Figure 1A, white arrows). HIV-associated retinopathy could produce similar findings, but it is not generally seen with CD4 counts > 200 cells/mm3. Additionally, in the left eye, there is a diffuse patch of retinal whitening (retinitis) associated with the inferotemporal vascular arcades (Figure 1B, white arrows). The entire area involved is poorly circumscribed and the whitening is subtle in areas. Overlying some areas of deeper, ground-glass whitening there are scattered, punctate white spots (Figure 1B, green arrows). Wickremasinghe and colleagues described this pattern of retinitis and suggested that it had a high positive-predictive value in the diagnosis of ocular syphilis.5
►Dr. Swamy. The patient then underwent fluorescein angiography and optical coherence tomography (OCT). Dr. Butler, what did the fluorescein angiography show?
►Dr. Butler. The fluorescein angiogram in both eyes revealed leakage of dye consistent with diabetic retinopathy, with the right eye (OD) worse than the left (OS). Additionally, the areas of active retinitis in the left eye displayed gradual staining with leopard-spot changes, along with late leakage of fluorescein dye, indicating vasculopathy in the infected area (Figure 2, arrows). The patient also underwent OCT in the left eye (images not displayed) demonstrating vitreous cells (vitritis), patches of inner retinal thickening with hyperreflectivity, and hyperreflective nodules at the level of the retinal pigment epithelium with overlying photoreceptor disruption. These OCT findings are fairly stereotypic for syphilitic chorioretinitis.6
►Dr. Swamy. Based on the ophthalmic findings, a diagnosis of ocular syphilis was made. Dr. Serrao, what should internists consider as they evaluate and manage a patient with ocular syphilis?
►Dr. Serrao. Although isolated ocular involvement from syphilis is possible, the majority of patients (up to 85%) with HIV can present with concomitant central nervous system infection and about 30% present with symptomatic neurosyphilis (a typical late manifestation of this disease) that reflects the aggressiveness, accelerated course and propensity for wide dissemination of syphilis in this patient population.7
The presence of concomitant cutaneous rashes should prompt universal precautions, because transmission can occur via skin to skin contact. Clinicians should watch for the Jarisch-Herxheimer reaction during treatment, a syndrome of fever, myalgias, and headache, which results from circulating cytokines produced because of rapidly dying spirochetes that could mimic a penicillin drug reaction, yet is treated supportively.
As syphilis is sexually acquired, clinicians should test for coexistent sexually transmitted infections, vaccinate for those that are preventable (eg, hepatitis B), notify sexual partners via assistance from local departments of public health, and assess for coexistent drug use and offer counseling in order to optimize risk reduction. Special attention should be paid to virologic control of HIV since some studies have shown an increase in the propensity for breakthrough HIV viremia while on effective ART.9 This should warrant counseling for ongoing optimal ART adherence and close monitoring in the follow-up visits with a provider specialized in the treatment of syphilis and HIV.
►Dr. Swamy. A lumbar puncture is performed with the results listed in Table 2. Dr. Serrao, is the CSF consistent with neurosyphilis? What would you do next?
►Dr. Serrao. The lumbar puncture is inflammatory with a lymphocytic predominance, consistent with active ocular/neurosyphilis. The CSF Venereal Disease Research Laboratory test is specific but not sensitive so a negative value does not rule out the presence of central nervous system infection.10 The CSF fluorescent treponemal antibody (CSF FTA-ABS) is sensitive but not specific. In this case, the ocular findings, positive serum RPR, CSF lymphocytic predominance, and CSF FTA ABS strongly supports the diagnosis of ocular/early neurosyphilis in a patient with HIV infection in whom early aggressive treatment is warranted to prevent rapid progression/potential loss of vision.11
►Dr. Swamy. Dr. Butler, how does syphilis behave in the eye as compared to other infectious or inflammatory diseases? Do visual symptoms respond well to treatment?
►Dr. Butler. As opposed to the dramatic reduction in rates and severity of CMV retinitis, HAART has had a negligible effect on ocular syphilis in the setting of HIV coinfection; in fact, rates of syphilis, including ocular syphilis, are currently surging world-wide, and HIV coinfection portends a worse prognosis.12 This is especially true among gay men. More so, there appears to be no correlation between CD4 count and incidence of developing ocular syphilis, as opposed to CMV retinitis, which occurs far more frequently in those with CD4 counts < 50 cells/mm3. In keeping with its epithet as one of the “Great Imitators,” syphilis can affect virtually every tissue of the eye—conjunctiva, sclera, cornea, iris, lens, vitreous, retina, choroid, optic nerve—unlike other OOI, such as CMV or toxoplasma, which generally hone to the retina. Nonetheless, various findings and patterns on clinical exam and ancillary testing, such as the more recently described punctate inner retinitis (as seen in our patient) and the more classic acute syphilitic posterior placoid chorioretinitis, carry high specificity for ocular syphilis.13
Patients with ocular syphilis should be treated according to neurosyphilis treatment protocols. In general, these patients respond very well to treatment with resolution of the ocular findings and recovery of complete, or nearly so, visual function, as long as an excessive delay between diagnosis and proper treatment does not occur.14
►Dr. Swamy. Following this testing, the patient completed 14 days of IV penicillin with resolution of symptoms. He had no further vision complaints. He was started on Triumeq (abacavir, dolutegravir, and lamivudine) with good adherence to therapy. Dr. Serrao, in 2016 the CDC released a clinical advisory about ocular syphilis. Can you tell us about why this is an important diagnosis to be aware of today?
►Dr. Serrao. As with any disease, the epidemiologic characteristics of an infection like syphilis allow the clinician to more carefully entertain such a diagnosis in any one individual by improving the index of suspicion for a particular disease. Awareness of an increase in ocular syphilis in HIV positive MSM allows for a more timely assessment and subsequent treatment with the goal of preventing loss of vision.15
►Lakshmana Swamy, MD, chief medical resident, VA Boston Healthcare System (VABHS) and Boston Medical Center. Dr. Serrao, when you hear about vision changes in a patient with HIV, what differential diagnosis is generated? What epidemiologic or historical factors can help distinguish these entities?
►Richard Serrao, MD, Infectious Disease Service, VABHS and assistant professor of medicine, Boston University School of Medicine. The differential diagnoses for vision changes in a patient with HIV is based on the overall immunosuppression of the patient: the lower the patient’s CD4 count, the higher the number of etiologies.1 The portions of the visual pathway as well as the pattern of vision loss are useful in narrowing the differential. For example, monocular visual disturbances with dermatomal vesicles within the ophthalmic division of the trigeminal nerve strongly implicates varicella zoster retinitis or keratitis; abducens nerve palsy could suggest granulomatous basilar meningitis from cryptococcosis. Likewise, ongoing fevers in an advanced AIDS patient with concomitant colitis, hepatitis, and pneumonitis is strongly suspicious for cytomegalovirus (CMV) retinitis with wide dissemination.
Geographic epidemiologic factors can suggest pathogens more prevalent to certain regions of the world, such as histoplasma chorioretinitis in a resident of the central and eastern U.S. or tuberculosis in a returning traveler. Likewise, a cat owner or one who consumes steak tartare increases the likelihood for toxoplasma retinochoroiditis, or syphilis in men who have sex with men (MSM) in the U.S. given that the majority of new cases occur in this patient population. Other clues one should consider include the presence of splinter hemorrhages in the extremities in an intravenous drug user, raising the possibility of embolic endophthalmitis from bacterial or fungal endocarditis. A variety of other diagnoses can certainly occur as a result of drug treatment (uveitis from rifampin, for example), immune reconstitution from HAART, infections with other HIV-associated pathogens, such as Pneumocystis jiroveci, and many non-HIV-related ocular diseases.
►Dr. Swamy. Dr. Butler, what concerns do you have when you hear about an HIV-infected patient with vision loss from the ophthalmology perspective?
►Nicholas Butler, MD, Ophthalmology Service, Uveitis and Ocular Immunology, VABHS and assistant professor of ophthalmology, Harvard Medical School. Of course, patients with HIV suffer from common causes of vision loss—cataract, glaucoma, diabetes, macular degeneration, for instance—just like those without HIV infection. If there is no significant immunodeficiency, then the patient’s HIV status would be less relevant, and these more common causes of vision loss should be pursued. My first task would be to determine the patient’s most recent CD4 T-cell count.
Assuming an HIV-positive individual is experiencing visual symptoms related to his/her underlying HIV infection (especially in the setting of CD4 counts < 200 cells/mm3), ocular opportunistic infections (OOI) come to mind first. Despite a reduction in incidence of 75% to 80% in the HAART-era, CMV retinitis remains the most common OOI in patients with AIDS and carries the greatest risk of ocular morbidity.2 In fact, based on enrollment data for the Longitudinal Study of the Ocular Complications of AIDS (LSOCA), the prevalence of CMV retinitis among patients with AIDS is more than 20-fold higher than all other ocular complications of AIDS (OOIs and ocular neoplastic disease), including Kaposi sarcoma, lymphoma, herpes zoster ophthalmicus, ocular syphilis, ocular toxoplasma, necrotizing herpetic retinitis, cryptococcal choroiditis, and pneumocystis choroiditis.3 Beyond ocular opportunistic infections, the most common retinal finding in HIV-positive people is HIV retinopathy, nonspecific microvascular findings in the retina affecting nearly 70% of those with advanced HIV disease. Fortunately, HIV retinopathy is generally asymptomatic.4
►Dr. Swamy. Thank you for those explanations. Based on Dr. Serrao’s differential, it is worth noting that this patient is MSM. He was evaluated in urgent care with the initial examination showing a temperature of 98.0° F, pulse 83 beats per minute, and blood pressure 110/70 mm Hg. The eye exam showed no injection with normal extraocular movements. Initial laboratory data were notable for a CD4 count of 730 cells/mm3 with fewer than 20 HIV viral copies/mL. Cytomegalovirus immunoglobulin G (IgG) was positive, and immunoglobulin M (IgM) was negative. A Lyme antibody was positive with negative IgM and IgG by Western blot. Additional tests can be seen in Tables 1 and 2. The patient has good immunologic and virologic control. How does this change your thinking about the case?
►Dr. Serrao. His CD4 count is well above 350, increasing the likelihood of a relatively uncomplicated course and treatment. Cytomegalovirus antibodies reflect prior infection. As CMV generally does not manifest with disease of any variety (including CMV retinitis) at this high CD4 count, one can presume he does not have CMV retinitis as a cause for his visual changes. CMV retinitis occurs mainly when substantial CD4 depletion has occurred (typically less than 50 cells/mm3). A positive Lyme antibody screen, not specific to Lyme, can be falsely positive in other treponema diseases (eg, Treponema pallidum, the etiologic organism of syphilis) as evidenced by negative confirmatory Western blot IgG and IgM. Antineutrophil cystoplasmic antibodies, lysozyme, angiotensin-converting enzyme, rapid plasma reagin (RPR), herpes simplex virus, toxoplasma are generally included in the workup for the differential of uveitis, retinitis, choroiditis, etc.
►Dr. Swamy. Based on the visual changes, the patient was referred for urgent ophthalmologic evaluation. Dr. Butler, when should a generalist consider urgent ophthalmology referral?
►Dr. Butler. In general, all patients with acute (and significant) vision loss should be referred immediately to an ophthalmologist. The challenge for the general practitioner is determining the true extent of the reported vision loss. If possible, some assessment of visual acuity should be obtained, testing each eye independently and with the correct glasses correction (ie, the patient’s distance glasses if the test object is 12 feet or more from the patient or their reading glasses if the test object is held inside arm’s length). If the general practitioner does not have access to an eye chart or near card, any assessment of vision with an appropriate description will be useful (eg, the patient can quickly count fingers at 15 feet in the unaffected eye, but the eye with reported vision loss cannot reliably count fingers outside of 2 feet). Additional ocular symptoms associated with the vision loss, such as pain, redness, photophobia, new flashes or floaters, increase the urgency of the referral. The threshold for referral for any ocular complaint is lower compared with that of the general population for those with evidence of immunodeficiency, such as for this patient with HIV. Any CD4 count < 200 cells/mm3 should raise the practitioner’s concern for an ocular opportunistic infection, with the greatest concern with CD4 counts < 50 cells/mm3.
►Dr. Swamy. The patient underwent further testing in the ophthalmology clinic. Dr. Butler, can you please interpret the funduscopic exam?
►Dr. Butler. Both eyes demonstrate findings (microaneurysms and small dot-blot hemorrhages) consistent with moderate nonproliferative diabetic retinopathy (Figure 1A, white arrows). HIV-associated retinopathy could produce similar findings, but it is not generally seen with CD4 counts > 200 cells/mm3. Additionally, in the left eye, there is a diffuse patch of retinal whitening (retinitis) associated with the inferotemporal vascular arcades (Figure 1B, white arrows). The entire area involved is poorly circumscribed and the whitening is subtle in areas. Overlying some areas of deeper, ground-glass whitening there are scattered, punctate white spots (Figure 1B, green arrows). Wickremasinghe and colleagues described this pattern of retinitis and suggested that it had a high positive-predictive value in the diagnosis of ocular syphilis.5
►Dr. Swamy. The patient then underwent fluorescein angiography and optical coherence tomography (OCT). Dr. Butler, what did the fluorescein angiography show?
►Dr. Butler. The fluorescein angiogram in both eyes revealed leakage of dye consistent with diabetic retinopathy, with the right eye (OD) worse than the left (OS). Additionally, the areas of active retinitis in the left eye displayed gradual staining with leopard-spot changes, along with late leakage of fluorescein dye, indicating vasculopathy in the infected area (Figure 2, arrows). The patient also underwent OCT in the left eye (images not displayed) demonstrating vitreous cells (vitritis), patches of inner retinal thickening with hyperreflectivity, and hyperreflective nodules at the level of the retinal pigment epithelium with overlying photoreceptor disruption. These OCT findings are fairly stereotypic for syphilitic chorioretinitis.6
►Dr. Swamy. Based on the ophthalmic findings, a diagnosis of ocular syphilis was made. Dr. Serrao, what should internists consider as they evaluate and manage a patient with ocular syphilis?
►Dr. Serrao. Although isolated ocular involvement from syphilis is possible, the majority of patients (up to 85%) with HIV can present with concomitant central nervous system infection and about 30% present with symptomatic neurosyphilis (a typical late manifestation of this disease) that reflects the aggressiveness, accelerated course and propensity for wide dissemination of syphilis in this patient population.7
The presence of concomitant cutaneous rashes should prompt universal precautions, because transmission can occur via skin to skin contact. Clinicians should watch for the Jarisch-Herxheimer reaction during treatment, a syndrome of fever, myalgias, and headache, which results from circulating cytokines produced because of rapidly dying spirochetes that could mimic a penicillin drug reaction, yet is treated supportively.
As syphilis is sexually acquired, clinicians should test for coexistent sexually transmitted infections, vaccinate for those that are preventable (eg, hepatitis B), notify sexual partners via assistance from local departments of public health, and assess for coexistent drug use and offer counseling in order to optimize risk reduction. Special attention should be paid to virologic control of HIV since some studies have shown an increase in the propensity for breakthrough HIV viremia while on effective ART.9 This should warrant counseling for ongoing optimal ART adherence and close monitoring in the follow-up visits with a provider specialized in the treatment of syphilis and HIV.
►Dr. Swamy. A lumbar puncture is performed with the results listed in Table 2. Dr. Serrao, is the CSF consistent with neurosyphilis? What would you do next?
►Dr. Serrao. The lumbar puncture is inflammatory with a lymphocytic predominance, consistent with active ocular/neurosyphilis. The CSF Venereal Disease Research Laboratory test is specific but not sensitive so a negative value does not rule out the presence of central nervous system infection.10 The CSF fluorescent treponemal antibody (CSF FTA-ABS) is sensitive but not specific. In this case, the ocular findings, positive serum RPR, CSF lymphocytic predominance, and CSF FTA ABS strongly supports the diagnosis of ocular/early neurosyphilis in a patient with HIV infection in whom early aggressive treatment is warranted to prevent rapid progression/potential loss of vision.11
►Dr. Swamy. Dr. Butler, how does syphilis behave in the eye as compared to other infectious or inflammatory diseases? Do visual symptoms respond well to treatment?
►Dr. Butler. As opposed to the dramatic reduction in rates and severity of CMV retinitis, HAART has had a negligible effect on ocular syphilis in the setting of HIV coinfection; in fact, rates of syphilis, including ocular syphilis, are currently surging world-wide, and HIV coinfection portends a worse prognosis.12 This is especially true among gay men. More so, there appears to be no correlation between CD4 count and incidence of developing ocular syphilis, as opposed to CMV retinitis, which occurs far more frequently in those with CD4 counts < 50 cells/mm3. In keeping with its epithet as one of the “Great Imitators,” syphilis can affect virtually every tissue of the eye—conjunctiva, sclera, cornea, iris, lens, vitreous, retina, choroid, optic nerve—unlike other OOI, such as CMV or toxoplasma, which generally hone to the retina. Nonetheless, various findings and patterns on clinical exam and ancillary testing, such as the more recently described punctate inner retinitis (as seen in our patient) and the more classic acute syphilitic posterior placoid chorioretinitis, carry high specificity for ocular syphilis.13
Patients with ocular syphilis should be treated according to neurosyphilis treatment protocols. In general, these patients respond very well to treatment with resolution of the ocular findings and recovery of complete, or nearly so, visual function, as long as an excessive delay between diagnosis and proper treatment does not occur.14
►Dr. Swamy. Following this testing, the patient completed 14 days of IV penicillin with resolution of symptoms. He had no further vision complaints. He was started on Triumeq (abacavir, dolutegravir, and lamivudine) with good adherence to therapy. Dr. Serrao, in 2016 the CDC released a clinical advisory about ocular syphilis. Can you tell us about why this is an important diagnosis to be aware of today?
►Dr. Serrao. As with any disease, the epidemiologic characteristics of an infection like syphilis allow the clinician to more carefully entertain such a diagnosis in any one individual by improving the index of suspicion for a particular disease. Awareness of an increase in ocular syphilis in HIV positive MSM allows for a more timely assessment and subsequent treatment with the goal of preventing loss of vision.15
1. Cunningham ET Jr, Margolis TP. Ocular manifestations of HIV infection. N Engl J Med. 1998;339(4):236-244.
2. Holtzer CD, Jacobson MA, Hadley WK, et al. Decline in the rate of specific opportunistic infections at San Francisco General Hospital, 1994-1997. AIDS. 1998;12(14):1931-1933.
3. Gangaputra S, Drye L, Vaidya V, Thorne JE, Jabs DA, Lyon AT. Non-cytomegalovirus ocular opportunistic infections in patients with acquired immunodeficiency syndrome. Am J Ophthalmol. 2013;155(2):206-212.e205.
4. Jabs DA, Van Natta ML, Holbrook JT, et al. Longitudinal study of the ocular complications of AIDS: 1. Ocular diagnoses at enrollment. Ophthalmology. 2007;114(4):780-786.
5. Wickremasinghe S, Ling C, Stawell R, Yeoh J, Hall A, Zamir E. Syphilitic punctate inner retinitis in immunocompetent gay men. Ophthalmology. 2009;116(6):1195-1200.
6. Burkholder BM, Leung TG, Ostheimer TA, Butler NJ, Thorne JE, Dunn JP. Spectral domain optical coherence tomography findings in acute syphilitic posterior placoid chorioretinitis. J Ophthalmic Inflamm Infect. 2014;4(1):2.
7. Musher DM, Hamill RJ, Baughn RE. Effect of human immunodeficiency virus (HIV) infection on the course of syphilis and on the response to treatment. Ann Intern Med. 1990;113(11):872-881.
8. Lukehart SA, Hook EW 3rd, Baker-Zander SA, Collier AC, Critchlow CW, Handsfield HH. Invasion of the central nervous system by Treponema pallidum: implications for diagnosis and treatment. Ann Intern Med. 1988;109(11):855-862.
9. Golden MR, Marra CM, Holmes KK. Update on syphilis: resurgence of an old problem. JAMA. 2003;290(11):1510-1514.
10. Marra CM, Tantalo LC, Maxwell CL, Ho EL, Sahi SK, Jones T. The rapid plasma reagin test cannot replace the venereal disease research laboratory test for neurosyphilis diagnosis. Sex Transm Dis. 2012;39(6):453-457.
11. Harding AS, Ghanem KG. The performance of cerebrospinal fluid treponemal-specific antibody tests in neurosyphilis: a systematic review. Sex Transm Dis. 2012;39(4):291-297.
12. Butler NJ, Thorne JE. Current status of HIV infection and ocular disease. Curr Opin Ophthalmol. 2012;23(6):517-522.
13. Gass JD, Braunstein RA, Chenoweth RG. Acute syphilitic posterior placoid chorioretinitis. Ophthalmology. 1990;97(10):1288-1297.
14. Davis JL. Ocular syphilis. Curr Opin Ophthalmol. 2014;25(6):513-518.
15. Clinical Advisory: Ocular Syphilis in the United States. https://www.cdc.gov/std/syphilis/clinicaladvisoryos2015.htm. Accessed September 11, 2017.
1. Cunningham ET Jr, Margolis TP. Ocular manifestations of HIV infection. N Engl J Med. 1998;339(4):236-244.
2. Holtzer CD, Jacobson MA, Hadley WK, et al. Decline in the rate of specific opportunistic infections at San Francisco General Hospital, 1994-1997. AIDS. 1998;12(14):1931-1933.
3. Gangaputra S, Drye L, Vaidya V, Thorne JE, Jabs DA, Lyon AT. Non-cytomegalovirus ocular opportunistic infections in patients with acquired immunodeficiency syndrome. Am J Ophthalmol. 2013;155(2):206-212.e205.
4. Jabs DA, Van Natta ML, Holbrook JT, et al. Longitudinal study of the ocular complications of AIDS: 1. Ocular diagnoses at enrollment. Ophthalmology. 2007;114(4):780-786.
5. Wickremasinghe S, Ling C, Stawell R, Yeoh J, Hall A, Zamir E. Syphilitic punctate inner retinitis in immunocompetent gay men. Ophthalmology. 2009;116(6):1195-1200.
6. Burkholder BM, Leung TG, Ostheimer TA, Butler NJ, Thorne JE, Dunn JP. Spectral domain optical coherence tomography findings in acute syphilitic posterior placoid chorioretinitis. J Ophthalmic Inflamm Infect. 2014;4(1):2.
7. Musher DM, Hamill RJ, Baughn RE. Effect of human immunodeficiency virus (HIV) infection on the course of syphilis and on the response to treatment. Ann Intern Med. 1990;113(11):872-881.
8. Lukehart SA, Hook EW 3rd, Baker-Zander SA, Collier AC, Critchlow CW, Handsfield HH. Invasion of the central nervous system by Treponema pallidum: implications for diagnosis and treatment. Ann Intern Med. 1988;109(11):855-862.
9. Golden MR, Marra CM, Holmes KK. Update on syphilis: resurgence of an old problem. JAMA. 2003;290(11):1510-1514.
10. Marra CM, Tantalo LC, Maxwell CL, Ho EL, Sahi SK, Jones T. The rapid plasma reagin test cannot replace the venereal disease research laboratory test for neurosyphilis diagnosis. Sex Transm Dis. 2012;39(6):453-457.
11. Harding AS, Ghanem KG. The performance of cerebrospinal fluid treponemal-specific antibody tests in neurosyphilis: a systematic review. Sex Transm Dis. 2012;39(4):291-297.
12. Butler NJ, Thorne JE. Current status of HIV infection and ocular disease. Curr Opin Ophthalmol. 2012;23(6):517-522.
13. Gass JD, Braunstein RA, Chenoweth RG. Acute syphilitic posterior placoid chorioretinitis. Ophthalmology. 1990;97(10):1288-1297.
14. Davis JL. Ocular syphilis. Curr Opin Ophthalmol. 2014;25(6):513-518.
15. Clinical Advisory: Ocular Syphilis in the United States. https://www.cdc.gov/std/syphilis/clinicaladvisoryos2015.htm. Accessed September 11, 2017.
The art of psychopharmacology: Avoiding medication changes and slowing down
As physicians, we are cognizant of the importance of patient-centered care, active listening, empathy, and patience—the so-called “hidden curriculum of medicine.”1 However, our attempts to centralize these concepts may be overshadowed by the deeply rooted drive to treat and fix. At times, we are simply treating uncertainty, whether it be diagnostic uncertainty or the uncertainty arising from clinical responses and outcomes that are far from binary. Definitive actions, such as adding medications or altering dosages, may appear to both patients and physicians to be a step closer to a “cure.” However, watchful waiting, re-evaluation, and accepting uncertainty are the true skills of effective care.
Be savvy about psychopharmacology
Psychotropics can take weeks to months to reach their full potential, and have varying responses and adverse effects. Beware of changing regimens prematurely, and keep in mind basic, yet crucial, pharmacokinetic concepts (eg, 4 to 5 half-lives to reach steady state, variations in metabolism). Receptor binding and dosing heuristics are notably common in psychiatry. Although such concepts are important to grasp, there is no one-size-fits-all rule. The brain simply does not possess the heart’s machine-like, linear functioning. Therefore, targeting individual parts (ie, receptors) will not equate to fixing the whole organ systematically or predictably.
Is the patient truly treatment-resistant?
Even the best treatment regimen has no clinical benefit if the patient cannot afford the prescription or does not take the medication. If cost is an impediment, switch from brand name drugs to generic formulations or to older medications in the same class. Before declaring the patient “treatment-resistant” and making medication changes, assess for compliance. This may require assistance from collateral informants. Ask family members to count the number of pills remaining in the bottle, and call the pharmacy to find out the last refill dates. If the patient exhibits a partial response to what should be a therapeutic dose, consider obtaining drug plasma levels to rule out rapid metabolism before deeming the medication trial a failure.2
Medications as liabilities
Overreliance on medications can result in the medications becoming liabilities. The polypharmacy problem is not unique to psychiatry.3 However, psychiatric patients may be more likely to inadvertently use medications in a maladaptive manner and disrupt the fundamental goals of long-term care. Avoid making medication adjustments in response to a patient’s life stressors and normative situational reactions. Doing so is a disservice to patients, because we are robbing them of chances to develop necessary coping skills and defenses. This can be overtly damaging in certain patient populations, such as those with borderline personality disorder, who may use medication adjustments as a crutch during crises.4
Treat the patient, not yourself
We physicians mean well in prescribing evidence-based treatments; however, if the symptoms or adverse effects are not bothersome or cause functional impairment, we risk losing sight of the patient’s goals in treatment and imposing our own instead. Displacing the treatment focus can alienate the patient, harm the therapeutic alliance, and result in “pill fatigue.” For example, we may be tempted to treat antipsychotic-induced tardive dyskinesia, even if the patient is not concerned about abnormal movements. Although we see this adverse effect
Change does not happen overnight
Picking a treatment option out of a lineup of choices, à la UWorld questions, does not always translate into patients agreeing with the suggested treatment, let alone the idea of receiving treatment at all. Motivational interviewing is our chance to shine in such situations and the reason why we are physicians, rather than answer-picking bots. Patients cannot change if they are not ready. However, we should be ready to roll with resistance while looking for signs of readiness to change. We must accept that it may take a week, a month, a year, or even longer for patients to align with our plan of action. The only futile decision is deeming our efforts as futile while discounting the benefits of incremental care.
1. Hafferty FW, Gaufberg EH, O’Donnell JF. The role of the hidden curriculum in “on doctoring” courses. AMA J Ethics. 2015;17(2):130-139.
2. Horvitz-Lennon M, Mattke S, Predmore Z, et al. The role of antipsychotic plasma levels in the treatment of schizophrenia. Am J Psychiatry. 2017;174(5):421-426.
3. Kantor ED, Rehm CD, Haas JS, et al. Trends in prescription drug use among adults in the United States from 1999-2012. JAMA. 2015;314(17):1818-1831.
4. Gunderson JG. The emergence of a generalist model to meet public health needs for patients with borderline personality disorder. Am J Psychiatry. 2016;173(5):452-458.
5. Kikkert MJ, Schene AH, Koeter MW, et al. Medication adherence in schizophrenia: exploring patients’, carers’ and professionals’ views. Schizophr Bull. 2005;32(4):786-794.
As physicians, we are cognizant of the importance of patient-centered care, active listening, empathy, and patience—the so-called “hidden curriculum of medicine.”1 However, our attempts to centralize these concepts may be overshadowed by the deeply rooted drive to treat and fix. At times, we are simply treating uncertainty, whether it be diagnostic uncertainty or the uncertainty arising from clinical responses and outcomes that are far from binary. Definitive actions, such as adding medications or altering dosages, may appear to both patients and physicians to be a step closer to a “cure.” However, watchful waiting, re-evaluation, and accepting uncertainty are the true skills of effective care.
Be savvy about psychopharmacology
Psychotropics can take weeks to months to reach their full potential, and have varying responses and adverse effects. Beware of changing regimens prematurely, and keep in mind basic, yet crucial, pharmacokinetic concepts (eg, 4 to 5 half-lives to reach steady state, variations in metabolism). Receptor binding and dosing heuristics are notably common in psychiatry. Although such concepts are important to grasp, there is no one-size-fits-all rule. The brain simply does not possess the heart’s machine-like, linear functioning. Therefore, targeting individual parts (ie, receptors) will not equate to fixing the whole organ systematically or predictably.
Is the patient truly treatment-resistant?
Even the best treatment regimen has no clinical benefit if the patient cannot afford the prescription or does not take the medication. If cost is an impediment, switch from brand name drugs to generic formulations or to older medications in the same class. Before declaring the patient “treatment-resistant” and making medication changes, assess for compliance. This may require assistance from collateral informants. Ask family members to count the number of pills remaining in the bottle, and call the pharmacy to find out the last refill dates. If the patient exhibits a partial response to what should be a therapeutic dose, consider obtaining drug plasma levels to rule out rapid metabolism before deeming the medication trial a failure.2
Medications as liabilities
Overreliance on medications can result in the medications becoming liabilities. The polypharmacy problem is not unique to psychiatry.3 However, psychiatric patients may be more likely to inadvertently use medications in a maladaptive manner and disrupt the fundamental goals of long-term care. Avoid making medication adjustments in response to a patient’s life stressors and normative situational reactions. Doing so is a disservice to patients, because we are robbing them of chances to develop necessary coping skills and defenses. This can be overtly damaging in certain patient populations, such as those with borderline personality disorder, who may use medication adjustments as a crutch during crises.4
Treat the patient, not yourself
We physicians mean well in prescribing evidence-based treatments; however, if the symptoms or adverse effects are not bothersome or cause functional impairment, we risk losing sight of the patient’s goals in treatment and imposing our own instead. Displacing the treatment focus can alienate the patient, harm the therapeutic alliance, and result in “pill fatigue.” For example, we may be tempted to treat antipsychotic-induced tardive dyskinesia, even if the patient is not concerned about abnormal movements. Although we see this adverse effect
Change does not happen overnight
Picking a treatment option out of a lineup of choices, à la UWorld questions, does not always translate into patients agreeing with the suggested treatment, let alone the idea of receiving treatment at all. Motivational interviewing is our chance to shine in such situations and the reason why we are physicians, rather than answer-picking bots. Patients cannot change if they are not ready. However, we should be ready to roll with resistance while looking for signs of readiness to change. We must accept that it may take a week, a month, a year, or even longer for patients to align with our plan of action. The only futile decision is deeming our efforts as futile while discounting the benefits of incremental care.
As physicians, we are cognizant of the importance of patient-centered care, active listening, empathy, and patience—the so-called “hidden curriculum of medicine.”1 However, our attempts to centralize these concepts may be overshadowed by the deeply rooted drive to treat and fix. At times, we are simply treating uncertainty, whether it be diagnostic uncertainty or the uncertainty arising from clinical responses and outcomes that are far from binary. Definitive actions, such as adding medications or altering dosages, may appear to both patients and physicians to be a step closer to a “cure.” However, watchful waiting, re-evaluation, and accepting uncertainty are the true skills of effective care.
Be savvy about psychopharmacology
Psychotropics can take weeks to months to reach their full potential, and have varying responses and adverse effects. Beware of changing regimens prematurely, and keep in mind basic, yet crucial, pharmacokinetic concepts (eg, 4 to 5 half-lives to reach steady state, variations in metabolism). Receptor binding and dosing heuristics are notably common in psychiatry. Although such concepts are important to grasp, there is no one-size-fits-all rule. The brain simply does not possess the heart’s machine-like, linear functioning. Therefore, targeting individual parts (ie, receptors) will not equate to fixing the whole organ systematically or predictably.
Is the patient truly treatment-resistant?
Even the best treatment regimen has no clinical benefit if the patient cannot afford the prescription or does not take the medication. If cost is an impediment, switch from brand name drugs to generic formulations or to older medications in the same class. Before declaring the patient “treatment-resistant” and making medication changes, assess for compliance. This may require assistance from collateral informants. Ask family members to count the number of pills remaining in the bottle, and call the pharmacy to find out the last refill dates. If the patient exhibits a partial response to what should be a therapeutic dose, consider obtaining drug plasma levels to rule out rapid metabolism before deeming the medication trial a failure.2
Medications as liabilities
Overreliance on medications can result in the medications becoming liabilities. The polypharmacy problem is not unique to psychiatry.3 However, psychiatric patients may be more likely to inadvertently use medications in a maladaptive manner and disrupt the fundamental goals of long-term care. Avoid making medication adjustments in response to a patient’s life stressors and normative situational reactions. Doing so is a disservice to patients, because we are robbing them of chances to develop necessary coping skills and defenses. This can be overtly damaging in certain patient populations, such as those with borderline personality disorder, who may use medication adjustments as a crutch during crises.4
Treat the patient, not yourself
We physicians mean well in prescribing evidence-based treatments; however, if the symptoms or adverse effects are not bothersome or cause functional impairment, we risk losing sight of the patient’s goals in treatment and imposing our own instead. Displacing the treatment focus can alienate the patient, harm the therapeutic alliance, and result in “pill fatigue.” For example, we may be tempted to treat antipsychotic-induced tardive dyskinesia, even if the patient is not concerned about abnormal movements. Although we see this adverse effect
Change does not happen overnight
Picking a treatment option out of a lineup of choices, à la UWorld questions, does not always translate into patients agreeing with the suggested treatment, let alone the idea of receiving treatment at all. Motivational interviewing is our chance to shine in such situations and the reason why we are physicians, rather than answer-picking bots. Patients cannot change if they are not ready. However, we should be ready to roll with resistance while looking for signs of readiness to change. We must accept that it may take a week, a month, a year, or even longer for patients to align with our plan of action. The only futile decision is deeming our efforts as futile while discounting the benefits of incremental care.
1. Hafferty FW, Gaufberg EH, O’Donnell JF. The role of the hidden curriculum in “on doctoring” courses. AMA J Ethics. 2015;17(2):130-139.
2. Horvitz-Lennon M, Mattke S, Predmore Z, et al. The role of antipsychotic plasma levels in the treatment of schizophrenia. Am J Psychiatry. 2017;174(5):421-426.
3. Kantor ED, Rehm CD, Haas JS, et al. Trends in prescription drug use among adults in the United States from 1999-2012. JAMA. 2015;314(17):1818-1831.
4. Gunderson JG. The emergence of a generalist model to meet public health needs for patients with borderline personality disorder. Am J Psychiatry. 2016;173(5):452-458.
5. Kikkert MJ, Schene AH, Koeter MW, et al. Medication adherence in schizophrenia: exploring patients’, carers’ and professionals’ views. Schizophr Bull. 2005;32(4):786-794.
1. Hafferty FW, Gaufberg EH, O’Donnell JF. The role of the hidden curriculum in “on doctoring” courses. AMA J Ethics. 2015;17(2):130-139.
2. Horvitz-Lennon M, Mattke S, Predmore Z, et al. The role of antipsychotic plasma levels in the treatment of schizophrenia. Am J Psychiatry. 2017;174(5):421-426.
3. Kantor ED, Rehm CD, Haas JS, et al. Trends in prescription drug use among adults in the United States from 1999-2012. JAMA. 2015;314(17):1818-1831.
4. Gunderson JG. The emergence of a generalist model to meet public health needs for patients with borderline personality disorder. Am J Psychiatry. 2016;173(5):452-458.
5. Kikkert MJ, Schene AH, Koeter MW, et al. Medication adherence in schizophrenia: exploring patients’, carers’ and professionals’ views. Schizophr Bull. 2005;32(4):786-794.
Vesiculobullous and Pustular Diseases in Newborns
Vesiculobullous eruptions in neonates can readily generate anxiety from parents/guardians and pediatricians over both infectious and noninfectious causes. The role of the dermatology resident is critical to help diminish fear over common vesicular presentations or to escalate care in rarer situations if a more obscure or ominous diagnosis is clouding the patient’s clinical presentation and well-being. This article summarizes both common and uncommon vesiculobullous neonatal diseases to augment precise and efficient diagnoses in this vulnerable patient population.
Steps for Evaluating a Vesiculopustular Eruption
Receiving a consultation for a newborn with widespread vesicles can be a daunting scenario for a dermatology resident. Fear of missing an ominous diagnosis or aggressively treating a newborn for an erroneous infection when the diagnosis is actually a benign presentation can lead to an anxiety-provoking situation. Additionally, performing a procedure on a newborn can cause personal uneasiness. Dr. Lawrence A. Schachner, an eminent pediatric dermatologist at the University of Miami Miller School of Medicine (Miami, Florida), recently lectured on 5 key steps (Table 1) for the evaluation of a vesiculobullous eruption in the newborn to maximize the accuracy of diagnosis and patient care.1

First, draw out the fluid from the vesicle to send for bacterial and viral culture as well as Gram stain. Second, snip the roof of the vesicle to perform potassium hydroxide examination for yeast or fungi and frozen pathology when indicated. Third, use the base of the vesicle to obtain cells for a Tzanck smear to identify the predominant cell infiltrate, such as multinucleated giant cells in herpes simplex virus or eosinophils in erythema toxicum neonatorum (ETN). Fourth, a mineral oil preparation can be performed on several lesions, especially if a burrow is observed, to rule out bullous scabies in the appropriate clinical presentation. Lastly, a perilesional or lesional punch biopsy can be performed if the above steps have not yet clinched the diagnosis.2 By utilizing these steps, the resident efficiently utilizes 1 lesion to narrow down a formidable differential list of bullous disorders in the newborn.
Specific Diagnoses
A number of common diagnoses can present during the newborn period and can usually be readily diagnosed by clinical manifestations alone; a summary of these eruptions is provided in Table 2. Erythema toxicum neonatorum is the most common pustular eruption in neonates and presents in up to 50% of full-term infants at days 1 to 2 of life. Inflammatory pustules surrounded by characteristic blotchy erythema are displayed on the face, trunk, arms, and legs, usually sparing the palms and soles.3 Erythema toxicum neonatorum typically is a clinical diagnosis; however, it can be confirmed by demonstrating the predominance of eosinophils on Tzanck smear.
Transient neonatal pustular melanosis (TNPM) also presents in full-term infants; usually favors darkly pigmented neonates; and exhibits either pustules with a collarette of scale that lack surrounding erythema or with residual brown macules on the face, genitals, and acral surfaces. Postinflammatory pigmentary alteration on lesion clearance is another clue to diagnosis. Similarly, it is a clinical diagnosis but can be confirmed with a Tzanck smear demonstrating neutrophils as the major cell infiltrate.
In a prospective 1-year multicenter study performed by Reginatto et al,4 2831 neonates born in southern Brazil underwent a skin examination by a dermatologist within 72 hours of birth to characterize the prevalence and demographics of ETN and TNPM. They found a 21.3% (602 cases) prevalence of ETN compared to a 3.4% (97 cases) prevalence of TNPM, but they noted that most patients were white, and thus the diagnosis of TNPM likely is less prevalent in this group, as it favors darkly pigmented individuals. Additional predisposing factors associated with ETN were male gender, an Apgar score of 8 to 10 at 1 minute, non–neonatal intensive care unit (NICU) patients, and lack of gestational risk factors. The TNPM population was much smaller, though the authors were able to conclude that the disease also was correlated with healthy, non-NICU patients. The authors hypothesized that there may be a role of immune system maturity in the pathogenesis of ETN and thus dermatology residents should be aware of the setting of their consultation.4 A NICU consultation for ETN should raise suspicion, as ETN and TNPM favor healthy infants who likely are not residing in the NICU; we are reminded of the target populations for these disease processes.
Additional common causes of vesicular eruptions in neonates can likewise be diagnosed chiefly with clinical inspection. Miliaria presents with tiny superficial crystalline vesicles on the neck and back of newborns due to elevated temperature and resultant obstruction of the eccrine sweat ducts. Reassurance can be provided, as spontaneous resolution occurs with cooling and limitation of occlusive clothing and swaddling.2
Infants at a few weeks of life may present with a noncomedonal pustular eruption on the cheeks, forehead, and scalp commonly known as neonatal acne or neonatal cephalic pustulosis. The driving factor is thought to be an abnormal response to Malassezia and can be treated with ketoconazole cream or expectant management.2
Cutaneous candidiasis is the most common infectious cause of vesicles in the neonate and can present in 2 fashions. Neonatal candidiasis is common, presenting a week after birth and manifesting as oral thrush and red plaques with satellite pustules in the diaper area. Congenital candidiasis is due to infection in utero, presents prior to 1 week of life, exhibits diffuse erythroderma, and requires timely parenteral antifungals.5 Newborns and preterm infants are at higher risk for systemic disease, while full-term infants may experience a mild course of skin-limited lesions.
It is imperative to rule out other infectious etiologies in ill-appearing neonates with vesicles such as herpes simplex virus, bacterial infections, syphilis, and vertically transmitted TORCH (toxoplasmosis, other infections rubella, cytomegalovirus infection, and herpes simplex) diagnoses.6 Herpes simplex virus classically presents with grouped vesicles on an erythematous base; however, such characteristic lesions may be subtle in the newborn. The site of skin involvement usually is the area that first comes into contact with maternal lesions, such as the face for a newborn delivered in a cephalic presentation.2 It is critical to be cognizant of this diagnosis, as a delay in antiviral therapy can result in neurologic consequences due to disseminated disease.
If the clinical picture of vesiculobullous disease in the newborn is not as clear, less common causes must be considered. Infantile acropustulosis presents with recurring crops of pustules on the hands and feet at several months of age. The most common differential diagnosis is scabies; therefore, a mineral oil preparation should be performed to rule out this common mimicker. Potent topical corticosteroids are first-line therapy, and episodes generally resolve with time.
Another mimicker of pustules in neonates includes deficiency of IL-1ra, a rare entity described in 2009.7 Deficiency of IL-1ra is an autoinflammatory syndrome of skin and bone due to unopposed action of IL-1 with life-threatening inflammation; infants present with pustules, lytic bone lesions, elevated erythrocyte sedimentation rate and C-reactive protein, and failure to thrive.8 The characteristic mutation was discovered when the infants dramatically responded to therapy with anakinra, an IL-1ra.
Eosinophilic pustular folliculitis is an additional pustular dermatosis that manifests with lesions predominately in the head and neck area, and unlike the adult population, it usually is self-resolving and not associated with other comorbidities in newborns.2
Incontinentia pigmenti is an X-linked dominant syndrome due to a genetic mutation in NEMO, nuclear factor κβ essential modulator, which protects against apoptosis.3 Incontinentia pigmenti presents in newborn girls shortly after birth with vesicles in a blaschkoid distribution before evolving through 4 unique stages of vesicular lesions, verrucous lesions, hyperpigmentation, and ultimately resolves with residual hypopigmentation in the affected area.
Lastly, neonatal Behçet disease can present with vesicles in the mouth and genital region due to transfer of maternal antibodies. It is self-limiting in nature and would be readily diagnosed with a known maternal history, though judicious screening for infections may be needed in specific settings.2
Conclusion
In summary, a vast array of benign and worrisome dermatoses present in the neonatal period. A thorough history and physical examination, including the temporality of the lesions, the health status of the newborn, and the maternal history, can help delineate the diagnosis. The 5-step method presented can further elucidate the underlying mechanism and reduce an overwhelming differential diagnosis list by reviewing each finding yielded from each step. Dermatology residents should feel comfortable addressing this unique patient population to ameliorate unclear cutaneous diagnoses for pediatricians.
Acknowledgment
A special thank you to Lawrence A. Schachner, MD (Miami, Florida), for his help providing resources and guidance for this topic.
- Schachner L. Vesiculopustular dermatosis in neonates and infants. Lecture presented at: University of Miami Department of Dermatology & Cutaneous Surgery Grand Rounds; August 23, 2017; Miami, Florida.
- Eichenfield LF, Lee PW, Larraide M, et al. Neonatal skin and skin disorders. In: Schachner LA, Hansen RC, eds. Pediatric Dermatology. 4th ed. Philadelphia, PA: Elsevier Mosby; 2011:299-373.
- Goddard DS, Gilliam AE, Frieden IJ. Vesiculobullous and erosive diseases in the newborn. In: Bolognia JL, Jorizzo JL, Schaffer JV, eds. Dermatology. 3rd ed. Philadelphia, PA: Elsevier Saunders; 2012:523-537.
- Reginatto FP, Muller FM, Peruzzo J, et al. Epidemiology and predisposing factors for erythema toxicum neonatorum and transient neonatal pustular melanosis: a multicenter study [published online May 25, 2017]. Pediatr Dermatol. 2017;34:422-426.
- Aruna C, Seetharam K. Congenital candidiasis. Indian Dermatol Online J. 2014;5(suppl 1):S44-S47.
- O’Connor NR, McLaughlin MR, Ham P. Newborn skin: part I. common rashes. Am Fam Physician. 2008;77:47-52.
- Reddy S, Jia S, Geoffrey R, et al. An autoinflammatory disease due to homozygous deletion of the IL1RN locus. N Engl J Med. 2009;360:2438-2444.
- Minkis K, Aksentijevich I, Goldbach-Mansky R, et al. Interleukin 1 receptor antagonist deficiency presenting as infantile pustulosis mimicking infantile pustular psoriasis. Arch Dermatol. 2012;148:747-752.
Vesiculobullous eruptions in neonates can readily generate anxiety from parents/guardians and pediatricians over both infectious and noninfectious causes. The role of the dermatology resident is critical to help diminish fear over common vesicular presentations or to escalate care in rarer situations if a more obscure or ominous diagnosis is clouding the patient’s clinical presentation and well-being. This article summarizes both common and uncommon vesiculobullous neonatal diseases to augment precise and efficient diagnoses in this vulnerable patient population.
Steps for Evaluating a Vesiculopustular Eruption
Receiving a consultation for a newborn with widespread vesicles can be a daunting scenario for a dermatology resident. Fear of missing an ominous diagnosis or aggressively treating a newborn for an erroneous infection when the diagnosis is actually a benign presentation can lead to an anxiety-provoking situation. Additionally, performing a procedure on a newborn can cause personal uneasiness. Dr. Lawrence A. Schachner, an eminent pediatric dermatologist at the University of Miami Miller School of Medicine (Miami, Florida), recently lectured on 5 key steps (Table 1) for the evaluation of a vesiculobullous eruption in the newborn to maximize the accuracy of diagnosis and patient care.1

First, draw out the fluid from the vesicle to send for bacterial and viral culture as well as Gram stain. Second, snip the roof of the vesicle to perform potassium hydroxide examination for yeast or fungi and frozen pathology when indicated. Third, use the base of the vesicle to obtain cells for a Tzanck smear to identify the predominant cell infiltrate, such as multinucleated giant cells in herpes simplex virus or eosinophils in erythema toxicum neonatorum (ETN). Fourth, a mineral oil preparation can be performed on several lesions, especially if a burrow is observed, to rule out bullous scabies in the appropriate clinical presentation. Lastly, a perilesional or lesional punch biopsy can be performed if the above steps have not yet clinched the diagnosis.2 By utilizing these steps, the resident efficiently utilizes 1 lesion to narrow down a formidable differential list of bullous disorders in the newborn.
Specific Diagnoses
A number of common diagnoses can present during the newborn period and can usually be readily diagnosed by clinical manifestations alone; a summary of these eruptions is provided in Table 2. Erythema toxicum neonatorum is the most common pustular eruption in neonates and presents in up to 50% of full-term infants at days 1 to 2 of life. Inflammatory pustules surrounded by characteristic blotchy erythema are displayed on the face, trunk, arms, and legs, usually sparing the palms and soles.3 Erythema toxicum neonatorum typically is a clinical diagnosis; however, it can be confirmed by demonstrating the predominance of eosinophils on Tzanck smear.

Transient neonatal pustular melanosis (TNPM) also presents in full-term infants; usually favors darkly pigmented neonates; and exhibits either pustules with a collarette of scale that lack surrounding erythema or with residual brown macules on the face, genitals, and acral surfaces. Postinflammatory pigmentary alteration on lesion clearance is another clue to diagnosis. Similarly, it is a clinical diagnosis but can be confirmed with a Tzanck smear demonstrating neutrophils as the major cell infiltrate.
In a prospective 1-year multicenter study performed by Reginatto et al,4 2831 neonates born in southern Brazil underwent a skin examination by a dermatologist within 72 hours of birth to characterize the prevalence and demographics of ETN and TNPM. They found a 21.3% (602 cases) prevalence of ETN compared to a 3.4% (97 cases) prevalence of TNPM, but they noted that most patients were white, and thus the diagnosis of TNPM likely is less prevalent in this group, as it favors darkly pigmented individuals. Additional predisposing factors associated with ETN were male gender, an Apgar score of 8 to 10 at 1 minute, non–neonatal intensive care unit (NICU) patients, and lack of gestational risk factors. The TNPM population was much smaller, though the authors were able to conclude that the disease also was correlated with healthy, non-NICU patients. The authors hypothesized that there may be a role of immune system maturity in the pathogenesis of ETN and thus dermatology residents should be aware of the setting of their consultation.4 A NICU consultation for ETN should raise suspicion, as ETN and TNPM favor healthy infants who likely are not residing in the NICU; we are reminded of the target populations for these disease processes.
Additional common causes of vesicular eruptions in neonates can likewise be diagnosed chiefly with clinical inspection. Miliaria presents with tiny superficial crystalline vesicles on the neck and back of newborns due to elevated temperature and resultant obstruction of the eccrine sweat ducts. Reassurance can be provided, as spontaneous resolution occurs with cooling and limitation of occlusive clothing and swaddling.2
Infants at a few weeks of life may present with a noncomedonal pustular eruption on the cheeks, forehead, and scalp commonly known as neonatal acne or neonatal cephalic pustulosis. The driving factor is thought to be an abnormal response to Malassezia and can be treated with ketoconazole cream or expectant management.2
Cutaneous candidiasis is the most common infectious cause of vesicles in the neonate and can present in 2 fashions. Neonatal candidiasis is common, presenting a week after birth and manifesting as oral thrush and red plaques with satellite pustules in the diaper area. Congenital candidiasis is due to infection in utero, presents prior to 1 week of life, exhibits diffuse erythroderma, and requires timely parenteral antifungals.5 Newborns and preterm infants are at higher risk for systemic disease, while full-term infants may experience a mild course of skin-limited lesions.
It is imperative to rule out other infectious etiologies in ill-appearing neonates with vesicles such as herpes simplex virus, bacterial infections, syphilis, and vertically transmitted TORCH (toxoplasmosis, other infections rubella, cytomegalovirus infection, and herpes simplex) diagnoses.6 Herpes simplex virus classically presents with grouped vesicles on an erythematous base; however, such characteristic lesions may be subtle in the newborn. The site of skin involvement usually is the area that first comes into contact with maternal lesions, such as the face for a newborn delivered in a cephalic presentation.2 It is critical to be cognizant of this diagnosis, as a delay in antiviral therapy can result in neurologic consequences due to disseminated disease.
If the clinical picture of vesiculobullous disease in the newborn is not as clear, less common causes must be considered. Infantile acropustulosis presents with recurring crops of pustules on the hands and feet at several months of age. The most common differential diagnosis is scabies; therefore, a mineral oil preparation should be performed to rule out this common mimicker. Potent topical corticosteroids are first-line therapy, and episodes generally resolve with time.
Another mimicker of pustules in neonates includes deficiency of IL-1ra, a rare entity described in 2009.7 Deficiency of IL-1ra is an autoinflammatory syndrome of skin and bone due to unopposed action of IL-1 with life-threatening inflammation; infants present with pustules, lytic bone lesions, elevated erythrocyte sedimentation rate and C-reactive protein, and failure to thrive.8 The characteristic mutation was discovered when the infants dramatically responded to therapy with anakinra, an IL-1ra.
Eosinophilic pustular folliculitis is an additional pustular dermatosis that manifests with lesions predominately in the head and neck area, and unlike the adult population, it usually is self-resolving and not associated with other comorbidities in newborns.2
Incontinentia pigmenti is an X-linked dominant syndrome due to a genetic mutation in NEMO, nuclear factor κβ essential modulator, which protects against apoptosis.3 Incontinentia pigmenti presents in newborn girls shortly after birth with vesicles in a blaschkoid distribution before evolving through 4 unique stages of vesicular lesions, verrucous lesions, hyperpigmentation, and ultimately resolves with residual hypopigmentation in the affected area.
Lastly, neonatal Behçet disease can present with vesicles in the mouth and genital region due to transfer of maternal antibodies. It is self-limiting in nature and would be readily diagnosed with a known maternal history, though judicious screening for infections may be needed in specific settings.2
Conclusion
In summary, a vast array of benign and worrisome dermatoses present in the neonatal period. A thorough history and physical examination, including the temporality of the lesions, the health status of the newborn, and the maternal history, can help delineate the diagnosis. The 5-step method presented can further elucidate the underlying mechanism and reduce an overwhelming differential diagnosis list by reviewing each finding yielded from each step. Dermatology residents should feel comfortable addressing this unique patient population to ameliorate unclear cutaneous diagnoses for pediatricians.
Acknowledgment
A special thank you to Lawrence A. Schachner, MD (Miami, Florida), for his help providing resources and guidance for this topic.
Vesiculobullous eruptions in neonates can readily generate anxiety from parents/guardians and pediatricians over both infectious and noninfectious causes. The role of the dermatology resident is critical to help diminish fear over common vesicular presentations or to escalate care in rarer situations if a more obscure or ominous diagnosis is clouding the patient’s clinical presentation and well-being. This article summarizes both common and uncommon vesiculobullous neonatal diseases to augment precise and efficient diagnoses in this vulnerable patient population.
Steps for Evaluating a Vesiculopustular Eruption
Receiving a consultation for a newborn with widespread vesicles can be a daunting scenario for a dermatology resident. Fear of missing an ominous diagnosis or aggressively treating a newborn for an erroneous infection when the diagnosis is actually a benign presentation can lead to an anxiety-provoking situation. Additionally, performing a procedure on a newborn can cause personal uneasiness. Dr. Lawrence A. Schachner, an eminent pediatric dermatologist at the University of Miami Miller School of Medicine (Miami, Florida), recently lectured on 5 key steps (Table 1) for the evaluation of a vesiculobullous eruption in the newborn to maximize the accuracy of diagnosis and patient care.1

First, draw out the fluid from the vesicle to send for bacterial and viral culture as well as Gram stain. Second, snip the roof of the vesicle to perform potassium hydroxide examination for yeast or fungi and frozen pathology when indicated. Third, use the base of the vesicle to obtain cells for a Tzanck smear to identify the predominant cell infiltrate, such as multinucleated giant cells in herpes simplex virus or eosinophils in erythema toxicum neonatorum (ETN). Fourth, a mineral oil preparation can be performed on several lesions, especially if a burrow is observed, to rule out bullous scabies in the appropriate clinical presentation. Lastly, a perilesional or lesional punch biopsy can be performed if the above steps have not yet clinched the diagnosis.2 By utilizing these steps, the resident efficiently utilizes 1 lesion to narrow down a formidable differential list of bullous disorders in the newborn.
Specific Diagnoses
A number of common diagnoses can present during the newborn period and can usually be readily diagnosed by clinical manifestations alone; a summary of these eruptions is provided in Table 2. Erythema toxicum neonatorum is the most common pustular eruption in neonates and presents in up to 50% of full-term infants at days 1 to 2 of life. Inflammatory pustules surrounded by characteristic blotchy erythema are displayed on the face, trunk, arms, and legs, usually sparing the palms and soles.3 Erythema toxicum neonatorum typically is a clinical diagnosis; however, it can be confirmed by demonstrating the predominance of eosinophils on Tzanck smear.

Transient neonatal pustular melanosis (TNPM) also presents in full-term infants; usually favors darkly pigmented neonates; and exhibits either pustules with a collarette of scale that lack surrounding erythema or with residual brown macules on the face, genitals, and acral surfaces. Postinflammatory pigmentary alteration on lesion clearance is another clue to diagnosis. Similarly, it is a clinical diagnosis but can be confirmed with a Tzanck smear demonstrating neutrophils as the major cell infiltrate.
In a prospective 1-year multicenter study performed by Reginatto et al,4 2831 neonates born in southern Brazil underwent a skin examination by a dermatologist within 72 hours of birth to characterize the prevalence and demographics of ETN and TNPM. They found a 21.3% (602 cases) prevalence of ETN compared to a 3.4% (97 cases) prevalence of TNPM, but they noted that most patients were white, and thus the diagnosis of TNPM likely is less prevalent in this group, as it favors darkly pigmented individuals. Additional predisposing factors associated with ETN were male gender, an Apgar score of 8 to 10 at 1 minute, non–neonatal intensive care unit (NICU) patients, and lack of gestational risk factors. The TNPM population was much smaller, though the authors were able to conclude that the disease also was correlated with healthy, non-NICU patients. The authors hypothesized that there may be a role of immune system maturity in the pathogenesis of ETN and thus dermatology residents should be aware of the setting of their consultation.4 A NICU consultation for ETN should raise suspicion, as ETN and TNPM favor healthy infants who likely are not residing in the NICU; we are reminded of the target populations for these disease processes.
Additional common causes of vesicular eruptions in neonates can likewise be diagnosed chiefly with clinical inspection. Miliaria presents with tiny superficial crystalline vesicles on the neck and back of newborns due to elevated temperature and resultant obstruction of the eccrine sweat ducts. Reassurance can be provided, as spontaneous resolution occurs with cooling and limitation of occlusive clothing and swaddling.2
Infants at a few weeks of life may present with a noncomedonal pustular eruption on the cheeks, forehead, and scalp commonly known as neonatal acne or neonatal cephalic pustulosis. The driving factor is thought to be an abnormal response to Malassezia and can be treated with ketoconazole cream or expectant management.2
Cutaneous candidiasis is the most common infectious cause of vesicles in the neonate and can present in 2 fashions. Neonatal candidiasis is common, presenting a week after birth and manifesting as oral thrush and red plaques with satellite pustules in the diaper area. Congenital candidiasis is due to infection in utero, presents prior to 1 week of life, exhibits diffuse erythroderma, and requires timely parenteral antifungals.5 Newborns and preterm infants are at higher risk for systemic disease, while full-term infants may experience a mild course of skin-limited lesions.
It is imperative to rule out other infectious etiologies in ill-appearing neonates with vesicles such as herpes simplex virus, bacterial infections, syphilis, and vertically transmitted TORCH (toxoplasmosis, other infections rubella, cytomegalovirus infection, and herpes simplex) diagnoses.6 Herpes simplex virus classically presents with grouped vesicles on an erythematous base; however, such characteristic lesions may be subtle in the newborn. The site of skin involvement usually is the area that first comes into contact with maternal lesions, such as the face for a newborn delivered in a cephalic presentation.2 It is critical to be cognizant of this diagnosis, as a delay in antiviral therapy can result in neurologic consequences due to disseminated disease.
If the clinical picture of vesiculobullous disease in the newborn is not as clear, less common causes must be considered. Infantile acropustulosis presents with recurring crops of pustules on the hands and feet at several months of age. The most common differential diagnosis is scabies; therefore, a mineral oil preparation should be performed to rule out this common mimicker. Potent topical corticosteroids are first-line therapy, and episodes generally resolve with time.
Another mimicker of pustules in neonates includes deficiency of IL-1ra, a rare entity described in 2009.7 Deficiency of IL-1ra is an autoinflammatory syndrome of skin and bone due to unopposed action of IL-1 with life-threatening inflammation; infants present with pustules, lytic bone lesions, elevated erythrocyte sedimentation rate and C-reactive protein, and failure to thrive.8 The characteristic mutation was discovered when the infants dramatically responded to therapy with anakinra, an IL-1ra.
Eosinophilic pustular folliculitis is an additional pustular dermatosis that manifests with lesions predominately in the head and neck area, and unlike the adult population, it usually is self-resolving and not associated with other comorbidities in newborns.2
Incontinentia pigmenti is an X-linked dominant syndrome due to a genetic mutation in NEMO, nuclear factor κβ essential modulator, which protects against apoptosis.3 Incontinentia pigmenti presents in newborn girls shortly after birth with vesicles in a blaschkoid distribution before evolving through 4 unique stages of vesicular lesions, verrucous lesions, hyperpigmentation, and ultimately resolves with residual hypopigmentation in the affected area.
Lastly, neonatal Behçet disease can present with vesicles in the mouth and genital region due to transfer of maternal antibodies. It is self-limiting in nature and would be readily diagnosed with a known maternal history, though judicious screening for infections may be needed in specific settings.2
Conclusion
In summary, a vast array of benign and worrisome dermatoses present in the neonatal period. A thorough history and physical examination, including the temporality of the lesions, the health status of the newborn, and the maternal history, can help delineate the diagnosis. The 5-step method presented can further elucidate the underlying mechanism and reduce an overwhelming differential diagnosis list by reviewing each finding yielded from each step. Dermatology residents should feel comfortable addressing this unique patient population to ameliorate unclear cutaneous diagnoses for pediatricians.
Acknowledgment
A special thank you to Lawrence A. Schachner, MD (Miami, Florida), for his help providing resources and guidance for this topic.
- Schachner L. Vesiculopustular dermatosis in neonates and infants. Lecture presented at: University of Miami Department of Dermatology & Cutaneous Surgery Grand Rounds; August 23, 2017; Miami, Florida.
- Eichenfield LF, Lee PW, Larraide M, et al. Neonatal skin and skin disorders. In: Schachner LA, Hansen RC, eds. Pediatric Dermatology. 4th ed. Philadelphia, PA: Elsevier Mosby; 2011:299-373.
- Goddard DS, Gilliam AE, Frieden IJ. Vesiculobullous and erosive diseases in the newborn. In: Bolognia JL, Jorizzo JL, Schaffer JV, eds. Dermatology. 3rd ed. Philadelphia, PA: Elsevier Saunders; 2012:523-537.
- Reginatto FP, Muller FM, Peruzzo J, et al. Epidemiology and predisposing factors for erythema toxicum neonatorum and transient neonatal pustular melanosis: a multicenter study [published online May 25, 2017]. Pediatr Dermatol. 2017;34:422-426.
- Aruna C, Seetharam K. Congenital candidiasis. Indian Dermatol Online J. 2014;5(suppl 1):S44-S47.
- O’Connor NR, McLaughlin MR, Ham P. Newborn skin: part I. common rashes. Am Fam Physician. 2008;77:47-52.
- Reddy S, Jia S, Geoffrey R, et al. An autoinflammatory disease due to homozygous deletion of the IL1RN locus. N Engl J Med. 2009;360:2438-2444.
- Minkis K, Aksentijevich I, Goldbach-Mansky R, et al. Interleukin 1 receptor antagonist deficiency presenting as infantile pustulosis mimicking infantile pustular psoriasis. Arch Dermatol. 2012;148:747-752.
- Schachner L. Vesiculopustular dermatosis in neonates and infants. Lecture presented at: University of Miami Department of Dermatology & Cutaneous Surgery Grand Rounds; August 23, 2017; Miami, Florida.
- Eichenfield LF, Lee PW, Larraide M, et al. Neonatal skin and skin disorders. In: Schachner LA, Hansen RC, eds. Pediatric Dermatology. 4th ed. Philadelphia, PA: Elsevier Mosby; 2011:299-373.
- Goddard DS, Gilliam AE, Frieden IJ. Vesiculobullous and erosive diseases in the newborn. In: Bolognia JL, Jorizzo JL, Schaffer JV, eds. Dermatology. 3rd ed. Philadelphia, PA: Elsevier Saunders; 2012:523-537.
- Reginatto FP, Muller FM, Peruzzo J, et al. Epidemiology and predisposing factors for erythema toxicum neonatorum and transient neonatal pustular melanosis: a multicenter study [published online May 25, 2017]. Pediatr Dermatol. 2017;34:422-426.
- Aruna C, Seetharam K. Congenital candidiasis. Indian Dermatol Online J. 2014;5(suppl 1):S44-S47.
- O’Connor NR, McLaughlin MR, Ham P. Newborn skin: part I. common rashes. Am Fam Physician. 2008;77:47-52.
- Reddy S, Jia S, Geoffrey R, et al. An autoinflammatory disease due to homozygous deletion of the IL1RN locus. N Engl J Med. 2009;360:2438-2444.
- Minkis K, Aksentijevich I, Goldbach-Mansky R, et al. Interleukin 1 receptor antagonist deficiency presenting as infantile pustulosis mimicking infantile pustular psoriasis. Arch Dermatol. 2012;148:747-752.
The Exam of the Future: What Should Residents Expect?
Complications of Cosmetic Eye Whitening
First introduced in 2008 as a surgical treatment of chronic conjunctival injection, cosmetic eye whitening became popularized in South Kore
The procedure involves performing a localized conjunctivectomy with or without removal of the Tenon capsule.4 Brimonidine tartrate is given for vascular constriction. When conjunctivectomy is performed in the right eye, the medial conjunctiva is incised from the 2-o’clock to 5-o’clock positions and the lateral conjunctiva is incised from the 10-o’clock to 7-o’clock positions. After the conjunctiva and Tenon capsule are excised, hemostasis is achieved with electrocauterization. Postoperative management may consist of topical mitomycin C (MMC) 0.02% 4 times daily for 2 to 5 days along with topical steroids. The addition of bevacizumab 1.25 mg/mL also has been described.5
In this report, we provide a comprehensive review of the complications of cosmetic eye whitening based on a review of the literature. Clinicians in both aesthetic practice and ophthalmology should be aware of the potential complications to accurately educate their patients about the possible risks and benefits of this procedure.
Methods
A review of PubMed articles indexed for MEDLINE (January 2009 to July 2017) using the search terms cosmetic eye whitening, cosmetic wide conjunctivectomy, I-Brite, and chronic hyperemic conjuctiva was conducted to evaluate the number of reports of complications from cosmetic eye whitening. A total of 10 articles were included in the study based on a review of abstracts. Non–English-language abstracts were not reviewed.
Results
Based on a review of 10 articles commenting on the complications of cosmetic eye whitening, a total of 2400 patients had undergone a cosmetic conjunctivectomy with various postoperative complications and recurrences (Table).4-13 The most commonly recurring complications based on the reported frequencies in the articles included chronic conjunctival epithelial defects, scleral thinning, calcific plaques, dry eye syndrome, diplopia (sometimes requiring strabismus surgery), and elevated intraocular pressure.
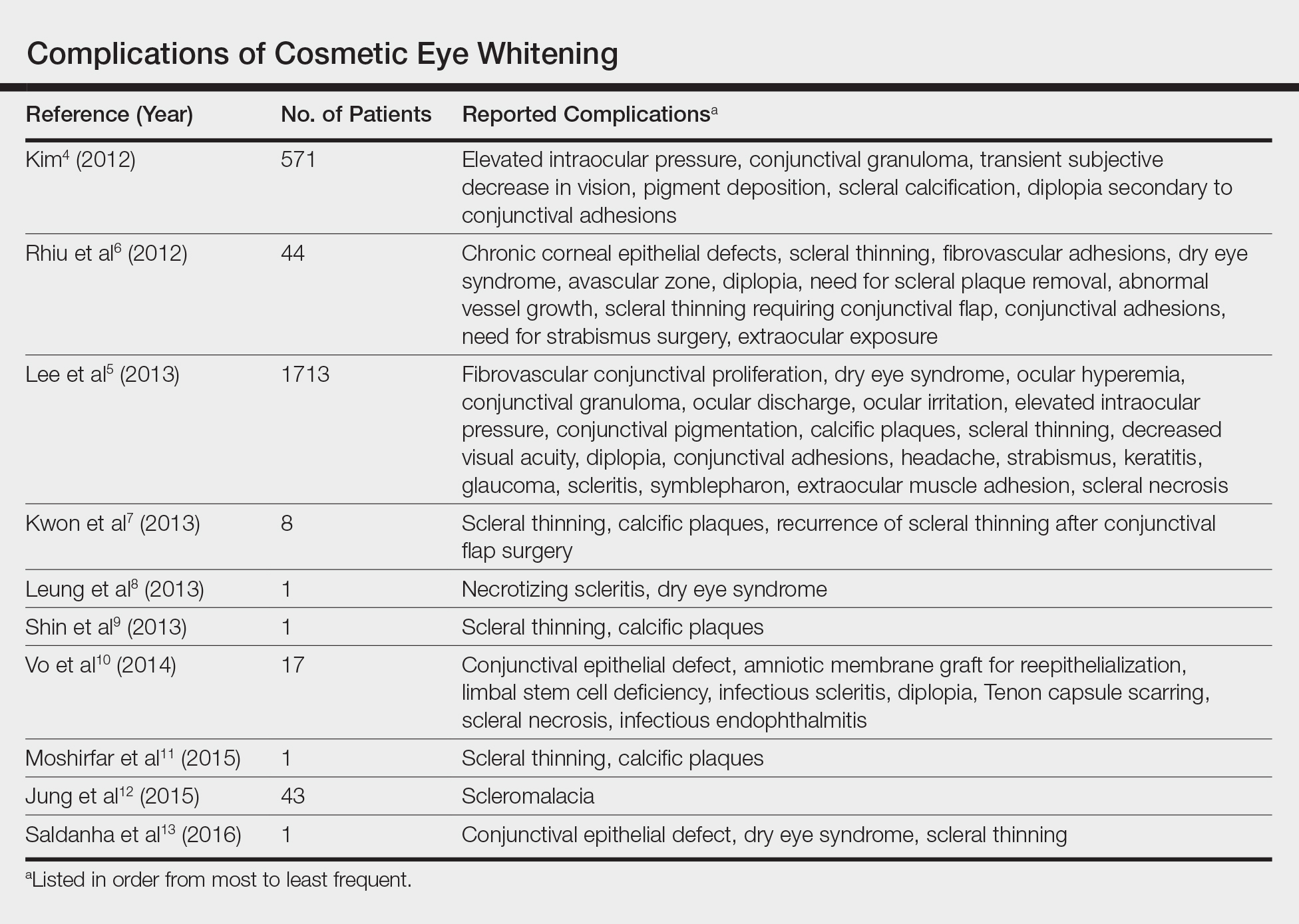
Kim4 was the first to report this surgical technique for irreversible hyperemic conjunctiva (N=1815). The reported success rate in South Korea was overwhelmingly high at 94.6%. In a mean (SD) follow-up time of 12.9 (7.8) months (range, 2–27 months), less than 20% of patients required surgical revision. During this time, the most common postoperative complications included elevation in intraocular pressure (17.2%), conjunctival granuloma (8.4%), transient vision decrease (7.5%), pigment deposition (5.3%), scleral calcifications (3.9%), and diplopia secondary to conjunctival adhesions (1.6%). No permanent defects were reported, and complications improved with surgical and medical management.4
Contrary to the findings of Kim,4 a large number of complications were seen; thus, on March 4, 2011, the Korean Ministry of Health & Welfare issued a declaration to discontinue the procedure under Article 49 of the Medical Service Act. Medical records from the single clinic in Korea from November 2007 to May 2010 were reviewed.5 One of the largest reviews of cosmetic eye whitening complications reviewed 1713 patients who underwent conjunctivectomy plus topical MMC with or without bevacizumab injection. Pterygium and chronic conjunctival hyperemia were the most common diagnoses that prompted patients to undergo treatment. Over an average follow-up period of 10.9 months, the overall complication rate was 82.9%, with severe complications being fibrovascular conjunctival proliferation (43.8%), recurrent hyperemic conjunctiva (28.1%), intraocular pressure (13.1%), scleral thinning with calcified plaques (6.2%), scleral thinning (4.4%), and diplopia (3.6%). A total of 56.9% of patients reported being satisfied with the cosmetic outcome of the surgery.5
In some of the smaller case series and case reports we reviewed, more vision-threatening complications have been described. Infectious endophthalmitis, infectious scleritis, and necrotizing scleritis have all been reported as complications of cosmetic eye whitening.8,10
Comment
The pathophysiology of the complications of cosmetic eye whitening stem from the disruption of the normal conjunctiva, destruction of the vascularization to the sclera, and loss of limbal stem cells. Mitomycin C is a topical antimetabolite antibiotic agent that inhibits DNA synthesis. This relatively safe and inexpensive product has decreased the recurrence rate in pterygium surgery as early as 1963.14,15 Complications of MMC in pterygium surgery include infectious scleritis, necrotizing scleritis, calcium formation, and even scleromalacia, occurring at incidence rates as low as 1.4%.16 These risks are balanced against the medical necessity of using MMC. Given the elective nature of cosmetic eye whitening, these complications in a cosmetic setting may not be justified.
The debate of the use of this procedure continues to occur in ophthalmologic societies. Both the Korean Ministry of Health & Welfare and the American Society of Cataract Refractive Surgery do not condone the use of regional conjunctivectomy for cosmetic eye whitening.5,17 Evidence shows that complications from cosmetic conjunctivectomy can be devastating and unnecessary given its elective nature. Although some complications (eg, dry eye syndrome, pain, discomfort) may be considered mild, the number of potentially serious complications brings the usefulness of the procedure into question.
This review is a launchpad to inform the medical community of the potential downside to conjunctivectomy for cosmetic eye whitening with the hope that it can initiate meaningful risk-benefit discussions between providers and physicians.
- Kim BH. Cosmetic eye whitening. Poster presented at: American Society of Cataract and Refractive Surgery; April 4-9, 2008; Chicago, IL.
- Kim BH. Cosmetic eye whitening by regional conjunctivectomy. Poster presented at: European Society of Cataract & Refractive Surgeons; September 13-17, 2008; Berlin, Germany.
- Raiskup F, Solomon A, Landau D, et al. Mitomycin C for pterygium: long term evaluation. Br J Ophthalmol. 2004;88:1425-1428.
- Kim BH. Regional conjunctivectomy with postoperative mitomycin C to treat chronic hyperemic conjunctiva. Cornea. 2012;31:236-244.
- Lee S, Go J, Rhiu S, et al. Cosmetic regional conjunctivectomy with postoperative mitomycin C application with or without bevacizumab injection [published online April 6, 2013]. Am J Ophthalmol. 2013;156:616-622.
- Rhiu S, Shim J, Kim EK, et al. Complications of cosmetic wide conjunctivectomy combined with postsurgical mitomycin C application. Cornea. 2012;31:245-252.
- Kwon HJ, Nam SM, Lee SY, et al. Conjunctival flap surgery for calcified scleromalacia after cosmetic conjunctivectomy. Cornea. 2013;32:821-825.
- Leung TG, Dunn JP, Akpek EK, et al. Necrotizing scleritis as a complication of cosmetic eye whitening procedure. J Ophthalmic Inflamm Infect. 2013;3:39.
- Shin HY, Kim MS, Chung SK. The development of scleromalacia after regional conjunctivectomy with the postoperative application of mitomycin C as an adjuvant therapy. Korean J Ophthalmol. 2013;27:208-210.
- Vo RC, Stafeeva K, Aldave AJ, et al. Complications related to a cosmetic eye-whitening procedure. Am J Ophthalmol. 2014;158:967-973.
- Moshirfar M, McCaughey MV, Fenzl CR, et al. Delayed manifestation of bilateral scleral thinning after I-BRITE® procedure and review of literature for cosmetic eye-whitening procedures. Clin Ophthalmol. 2015;9:445-451.
- Jung JW, Kwon KY, Choi DL, et al. Long-term clinical outcomes of conjunctival flap surgery for calcified scleromalacia after periocular surgery. Cornea. 2015;34:308-312.
- Saldanha MJ, Yang PT, Chan CC. Scleral thinning after I-BRITE procedure treated with amniotic membrane graft. Can J Ophthalmol. 2016;51:e115-e116.
- Seiler T, Schnelle B, Wollensak J. Pterygium excision using 193-nm excimer laser smoothing and topical mitomycin C. Ger J Ophthalmol. 1992;1:429-431.
- Singh G, Wilson MR, Foster CS. Long-term follow-up study of mitomycin eye drops as adjunctive treatment of pterygia and its comparison with conjunctival autograft transplantation. Cornea. 1990;9:331-334.
- Lam DS, Wong AK, Fan DS, et al. Intraoperative mitomycin C to prevent recurrence of pterygium after excision: a 30-month follow-up study. Ophthalmology. 1998;105:901-904; discussion 904-905.
- ASCRS Cornea Clinical Committee. Clinical alert: eye-whitening procedure: regional conjunctivectomy with mitomycin-C application [press release]. Fairfax, VA: American Society of Cataract and Refractive Surgery. http://www.ascrs.org/node/1352. Accessed January 22, 2015.
First introduced in 2008 as a surgical treatment of chronic conjunctival injection, cosmetic eye whitening became popularized in South Kore
The procedure involves performing a localized conjunctivectomy with or without removal of the Tenon capsule.4 Brimonidine tartrate is given for vascular constriction. When conjunctivectomy is performed in the right eye, the medial conjunctiva is incised from the 2-o’clock to 5-o’clock positions and the lateral conjunctiva is incised from the 10-o’clock to 7-o’clock positions. After the conjunctiva and Tenon capsule are excised, hemostasis is achieved with electrocauterization. Postoperative management may consist of topical mitomycin C (MMC) 0.02% 4 times daily for 2 to 5 days along with topical steroids. The addition of bevacizumab 1.25 mg/mL also has been described.5
In this report, we provide a comprehensive review of the complications of cosmetic eye whitening based on a review of the literature. Clinicians in both aesthetic practice and ophthalmology should be aware of the potential complications to accurately educate their patients about the possible risks and benefits of this procedure.
Methods
A review of PubMed articles indexed for MEDLINE (January 2009 to July 2017) using the search terms cosmetic eye whitening, cosmetic wide conjunctivectomy, I-Brite, and chronic hyperemic conjuctiva was conducted to evaluate the number of reports of complications from cosmetic eye whitening. A total of 10 articles were included in the study based on a review of abstracts. Non–English-language abstracts were not reviewed.
Results
Based on a review of 10 articles commenting on the complications of cosmetic eye whitening, a total of 2400 patients had undergone a cosmetic conjunctivectomy with various postoperative complications and recurrences (Table).4-13 The most commonly recurring complications based on the reported frequencies in the articles included chronic conjunctival epithelial defects, scleral thinning, calcific plaques, dry eye syndrome, diplopia (sometimes requiring strabismus surgery), and elevated intraocular pressure.

Kim4 was the first to report this surgical technique for irreversible hyperemic conjunctiva (N=1815). The reported success rate in South Korea was overwhelmingly high at 94.6%. In a mean (SD) follow-up time of 12.9 (7.8) months (range, 2–27 months), less than 20% of patients required surgical revision. During this time, the most common postoperative complications included elevation in intraocular pressure (17.2%), conjunctival granuloma (8.4%), transient vision decrease (7.5%), pigment deposition (5.3%), scleral calcifications (3.9%), and diplopia secondary to conjunctival adhesions (1.6%). No permanent defects were reported, and complications improved with surgical and medical management.4
Contrary to the findings of Kim,4 a large number of complications were seen; thus, on March 4, 2011, the Korean Ministry of Health & Welfare issued a declaration to discontinue the procedure under Article 49 of the Medical Service Act. Medical records from the single clinic in Korea from November 2007 to May 2010 were reviewed.5 One of the largest reviews of cosmetic eye whitening complications reviewed 1713 patients who underwent conjunctivectomy plus topical MMC with or without bevacizumab injection. Pterygium and chronic conjunctival hyperemia were the most common diagnoses that prompted patients to undergo treatment. Over an average follow-up period of 10.9 months, the overall complication rate was 82.9%, with severe complications being fibrovascular conjunctival proliferation (43.8%), recurrent hyperemic conjunctiva (28.1%), intraocular pressure (13.1%), scleral thinning with calcified plaques (6.2%), scleral thinning (4.4%), and diplopia (3.6%). A total of 56.9% of patients reported being satisfied with the cosmetic outcome of the surgery.5
In some of the smaller case series and case reports we reviewed, more vision-threatening complications have been described. Infectious endophthalmitis, infectious scleritis, and necrotizing scleritis have all been reported as complications of cosmetic eye whitening.8,10
Comment
The pathophysiology of the complications of cosmetic eye whitening stem from the disruption of the normal conjunctiva, destruction of the vascularization to the sclera, and loss of limbal stem cells. Mitomycin C is a topical antimetabolite antibiotic agent that inhibits DNA synthesis. This relatively safe and inexpensive product has decreased the recurrence rate in pterygium surgery as early as 1963.14,15 Complications of MMC in pterygium surgery include infectious scleritis, necrotizing scleritis, calcium formation, and even scleromalacia, occurring at incidence rates as low as 1.4%.16 These risks are balanced against the medical necessity of using MMC. Given the elective nature of cosmetic eye whitening, these complications in a cosmetic setting may not be justified.
The debate of the use of this procedure continues to occur in ophthalmologic societies. Both the Korean Ministry of Health & Welfare and the American Society of Cataract Refractive Surgery do not condone the use of regional conjunctivectomy for cosmetic eye whitening.5,17 Evidence shows that complications from cosmetic conjunctivectomy can be devastating and unnecessary given its elective nature. Although some complications (eg, dry eye syndrome, pain, discomfort) may be considered mild, the number of potentially serious complications brings the usefulness of the procedure into question.
This review is a launchpad to inform the medical community of the potential downside to conjunctivectomy for cosmetic eye whitening with the hope that it can initiate meaningful risk-benefit discussions between providers and physicians.
First introduced in 2008 as a surgical treatment of chronic conjunctival injection, cosmetic eye whitening became popularized in South Kore
The procedure involves performing a localized conjunctivectomy with or without removal of the Tenon capsule.4 Brimonidine tartrate is given for vascular constriction. When conjunctivectomy is performed in the right eye, the medial conjunctiva is incised from the 2-o’clock to 5-o’clock positions and the lateral conjunctiva is incised from the 10-o’clock to 7-o’clock positions. After the conjunctiva and Tenon capsule are excised, hemostasis is achieved with electrocauterization. Postoperative management may consist of topical mitomycin C (MMC) 0.02% 4 times daily for 2 to 5 days along with topical steroids. The addition of bevacizumab 1.25 mg/mL also has been described.5
In this report, we provide a comprehensive review of the complications of cosmetic eye whitening based on a review of the literature. Clinicians in both aesthetic practice and ophthalmology should be aware of the potential complications to accurately educate their patients about the possible risks and benefits of this procedure.
Methods
A review of PubMed articles indexed for MEDLINE (January 2009 to July 2017) using the search terms cosmetic eye whitening, cosmetic wide conjunctivectomy, I-Brite, and chronic hyperemic conjuctiva was conducted to evaluate the number of reports of complications from cosmetic eye whitening. A total of 10 articles were included in the study based on a review of abstracts. Non–English-language abstracts were not reviewed.
Results
Based on a review of 10 articles commenting on the complications of cosmetic eye whitening, a total of 2400 patients had undergone a cosmetic conjunctivectomy with various postoperative complications and recurrences (Table).4-13 The most commonly recurring complications based on the reported frequencies in the articles included chronic conjunctival epithelial defects, scleral thinning, calcific plaques, dry eye syndrome, diplopia (sometimes requiring strabismus surgery), and elevated intraocular pressure.

Kim4 was the first to report this surgical technique for irreversible hyperemic conjunctiva (N=1815). The reported success rate in South Korea was overwhelmingly high at 94.6%. In a mean (SD) follow-up time of 12.9 (7.8) months (range, 2–27 months), less than 20% of patients required surgical revision. During this time, the most common postoperative complications included elevation in intraocular pressure (17.2%), conjunctival granuloma (8.4%), transient vision decrease (7.5%), pigment deposition (5.3%), scleral calcifications (3.9%), and diplopia secondary to conjunctival adhesions (1.6%). No permanent defects were reported, and complications improved with surgical and medical management.4
Contrary to the findings of Kim,4 a large number of complications were seen; thus, on March 4, 2011, the Korean Ministry of Health & Welfare issued a declaration to discontinue the procedure under Article 49 of the Medical Service Act. Medical records from the single clinic in Korea from November 2007 to May 2010 were reviewed.5 One of the largest reviews of cosmetic eye whitening complications reviewed 1713 patients who underwent conjunctivectomy plus topical MMC with or without bevacizumab injection. Pterygium and chronic conjunctival hyperemia were the most common diagnoses that prompted patients to undergo treatment. Over an average follow-up period of 10.9 months, the overall complication rate was 82.9%, with severe complications being fibrovascular conjunctival proliferation (43.8%), recurrent hyperemic conjunctiva (28.1%), intraocular pressure (13.1%), scleral thinning with calcified plaques (6.2%), scleral thinning (4.4%), and diplopia (3.6%). A total of 56.9% of patients reported being satisfied with the cosmetic outcome of the surgery.5
In some of the smaller case series and case reports we reviewed, more vision-threatening complications have been described. Infectious endophthalmitis, infectious scleritis, and necrotizing scleritis have all been reported as complications of cosmetic eye whitening.8,10
Comment
The pathophysiology of the complications of cosmetic eye whitening stem from the disruption of the normal conjunctiva, destruction of the vascularization to the sclera, and loss of limbal stem cells. Mitomycin C is a topical antimetabolite antibiotic agent that inhibits DNA synthesis. This relatively safe and inexpensive product has decreased the recurrence rate in pterygium surgery as early as 1963.14,15 Complications of MMC in pterygium surgery include infectious scleritis, necrotizing scleritis, calcium formation, and even scleromalacia, occurring at incidence rates as low as 1.4%.16 These risks are balanced against the medical necessity of using MMC. Given the elective nature of cosmetic eye whitening, these complications in a cosmetic setting may not be justified.
The debate of the use of this procedure continues to occur in ophthalmologic societies. Both the Korean Ministry of Health & Welfare and the American Society of Cataract Refractive Surgery do not condone the use of regional conjunctivectomy for cosmetic eye whitening.5,17 Evidence shows that complications from cosmetic conjunctivectomy can be devastating and unnecessary given its elective nature. Although some complications (eg, dry eye syndrome, pain, discomfort) may be considered mild, the number of potentially serious complications brings the usefulness of the procedure into question.
This review is a launchpad to inform the medical community of the potential downside to conjunctivectomy for cosmetic eye whitening with the hope that it can initiate meaningful risk-benefit discussions between providers and physicians.
- Kim BH. Cosmetic eye whitening. Poster presented at: American Society of Cataract and Refractive Surgery; April 4-9, 2008; Chicago, IL.
- Kim BH. Cosmetic eye whitening by regional conjunctivectomy. Poster presented at: European Society of Cataract & Refractive Surgeons; September 13-17, 2008; Berlin, Germany.
- Raiskup F, Solomon A, Landau D, et al. Mitomycin C for pterygium: long term evaluation. Br J Ophthalmol. 2004;88:1425-1428.
- Kim BH. Regional conjunctivectomy with postoperative mitomycin C to treat chronic hyperemic conjunctiva. Cornea. 2012;31:236-244.
- Lee S, Go J, Rhiu S, et al. Cosmetic regional conjunctivectomy with postoperative mitomycin C application with or without bevacizumab injection [published online April 6, 2013]. Am J Ophthalmol. 2013;156:616-622.
- Rhiu S, Shim J, Kim EK, et al. Complications of cosmetic wide conjunctivectomy combined with postsurgical mitomycin C application. Cornea. 2012;31:245-252.
- Kwon HJ, Nam SM, Lee SY, et al. Conjunctival flap surgery for calcified scleromalacia after cosmetic conjunctivectomy. Cornea. 2013;32:821-825.
- Leung TG, Dunn JP, Akpek EK, et al. Necrotizing scleritis as a complication of cosmetic eye whitening procedure. J Ophthalmic Inflamm Infect. 2013;3:39.
- Shin HY, Kim MS, Chung SK. The development of scleromalacia after regional conjunctivectomy with the postoperative application of mitomycin C as an adjuvant therapy. Korean J Ophthalmol. 2013;27:208-210.
- Vo RC, Stafeeva K, Aldave AJ, et al. Complications related to a cosmetic eye-whitening procedure. Am J Ophthalmol. 2014;158:967-973.
- Moshirfar M, McCaughey MV, Fenzl CR, et al. Delayed manifestation of bilateral scleral thinning after I-BRITE® procedure and review of literature for cosmetic eye-whitening procedures. Clin Ophthalmol. 2015;9:445-451.
- Jung JW, Kwon KY, Choi DL, et al. Long-term clinical outcomes of conjunctival flap surgery for calcified scleromalacia after periocular surgery. Cornea. 2015;34:308-312.
- Saldanha MJ, Yang PT, Chan CC. Scleral thinning after I-BRITE procedure treated with amniotic membrane graft. Can J Ophthalmol. 2016;51:e115-e116.
- Seiler T, Schnelle B, Wollensak J. Pterygium excision using 193-nm excimer laser smoothing and topical mitomycin C. Ger J Ophthalmol. 1992;1:429-431.
- Singh G, Wilson MR, Foster CS. Long-term follow-up study of mitomycin eye drops as adjunctive treatment of pterygia and its comparison with conjunctival autograft transplantation. Cornea. 1990;9:331-334.
- Lam DS, Wong AK, Fan DS, et al. Intraoperative mitomycin C to prevent recurrence of pterygium after excision: a 30-month follow-up study. Ophthalmology. 1998;105:901-904; discussion 904-905.
- ASCRS Cornea Clinical Committee. Clinical alert: eye-whitening procedure: regional conjunctivectomy with mitomycin-C application [press release]. Fairfax, VA: American Society of Cataract and Refractive Surgery. http://www.ascrs.org/node/1352. Accessed January 22, 2015.
- Kim BH. Cosmetic eye whitening. Poster presented at: American Society of Cataract and Refractive Surgery; April 4-9, 2008; Chicago, IL.
- Kim BH. Cosmetic eye whitening by regional conjunctivectomy. Poster presented at: European Society of Cataract & Refractive Surgeons; September 13-17, 2008; Berlin, Germany.
- Raiskup F, Solomon A, Landau D, et al. Mitomycin C for pterygium: long term evaluation. Br J Ophthalmol. 2004;88:1425-1428.
- Kim BH. Regional conjunctivectomy with postoperative mitomycin C to treat chronic hyperemic conjunctiva. Cornea. 2012;31:236-244.
- Lee S, Go J, Rhiu S, et al. Cosmetic regional conjunctivectomy with postoperative mitomycin C application with or without bevacizumab injection [published online April 6, 2013]. Am J Ophthalmol. 2013;156:616-622.
- Rhiu S, Shim J, Kim EK, et al. Complications of cosmetic wide conjunctivectomy combined with postsurgical mitomycin C application. Cornea. 2012;31:245-252.
- Kwon HJ, Nam SM, Lee SY, et al. Conjunctival flap surgery for calcified scleromalacia after cosmetic conjunctivectomy. Cornea. 2013;32:821-825.
- Leung TG, Dunn JP, Akpek EK, et al. Necrotizing scleritis as a complication of cosmetic eye whitening procedure. J Ophthalmic Inflamm Infect. 2013;3:39.
- Shin HY, Kim MS, Chung SK. The development of scleromalacia after regional conjunctivectomy with the postoperative application of mitomycin C as an adjuvant therapy. Korean J Ophthalmol. 2013;27:208-210.
- Vo RC, Stafeeva K, Aldave AJ, et al. Complications related to a cosmetic eye-whitening procedure. Am J Ophthalmol. 2014;158:967-973.
- Moshirfar M, McCaughey MV, Fenzl CR, et al. Delayed manifestation of bilateral scleral thinning after I-BRITE® procedure and review of literature for cosmetic eye-whitening procedures. Clin Ophthalmol. 2015;9:445-451.
- Jung JW, Kwon KY, Choi DL, et al. Long-term clinical outcomes of conjunctival flap surgery for calcified scleromalacia after periocular surgery. Cornea. 2015;34:308-312.
- Saldanha MJ, Yang PT, Chan CC. Scleral thinning after I-BRITE procedure treated with amniotic membrane graft. Can J Ophthalmol. 2016;51:e115-e116.
- Seiler T, Schnelle B, Wollensak J. Pterygium excision using 193-nm excimer laser smoothing and topical mitomycin C. Ger J Ophthalmol. 1992;1:429-431.
- Singh G, Wilson MR, Foster CS. Long-term follow-up study of mitomycin eye drops as adjunctive treatment of pterygia and its comparison with conjunctival autograft transplantation. Cornea. 1990;9:331-334.
- Lam DS, Wong AK, Fan DS, et al. Intraoperative mitomycin C to prevent recurrence of pterygium after excision: a 30-month follow-up study. Ophthalmology. 1998;105:901-904; discussion 904-905.
- ASCRS Cornea Clinical Committee. Clinical alert: eye-whitening procedure: regional conjunctivectomy with mitomycin-C application [press release]. Fairfax, VA: American Society of Cataract and Refractive Surgery. http://www.ascrs.org/node/1352. Accessed January 22, 2015.
Resident Pearl
- Cosmetic eye whitening has severe and vision-threatening complications that should be aware to all cosmetic surgeons.
Top translator apps can help you communicate with patients who have limited English proficiency
As the population of patients with limited English proficiency increases throughout English-speaking countries, health care providers often need translator services. Medical translator smartphone applications (apps) are useful tools that can provide ad hoc translator services.
According to the US Census Bureau in 2015, more than 60 million individuals — about 19% of Americans — reported speaking a language other than English at home, and more than 25 million said that they speak English “less than very well.”1,2 The top 5 non-English languages spoken at home were Spanish, French, Chinese, Tagalog, and Vietnamese, encompassing 72% of non-English speakers.
In the health care sector, translator services are essential for providing accurate and culturally competent care. Current options for translator services include face-to-face interpreters, phone-based translator services, and translator apps on mobile devices. In settings where face-to-face interpreters or phone-based translator services are not available, translator apps may provide reasonable alternatives. My colleagues, Dr. Amrin Khander and Dr. Sara Farag, and I identified and evaluated medical translator apps that are available from the Apple iTunes and Google Play stores to aid clinicians in using such apps during clinical encounters.3
Three types of translator apps
Preset medical phrase translator apps require the user to search for or find a question or statement in order to facilitate a conversation. With these types of apps, a health care provider can choose fully conjugated sentences, which then can be played or read back to the patient in the chosen translated language. Within this group of apps, Canopy Speak and Universal Doctor Speaker are highly accessible, since both apps are available from the Apple iTunes and Google Play stores and both are free.
Medical dictionary apps require the user to search for a medical term in one language to receive a translation in another language. These apps are less useful, but they can help providers find and define specific terms in a given language.
General language translator apps require the user to enter a term, statement, or question in one language and then provide a translation in another language. Google Translate and Vocre Translate are examples.
The top recommended translator apps are listed in the TABLE alphabetically and are detailed with a shortened version of the APPLICATIONS scoring system, APPLI (app comprehensiveness, price, platform, literature use, and important special features).4 I hope the apps described here will help you enhance communication with your patients who have limited English proficiency.
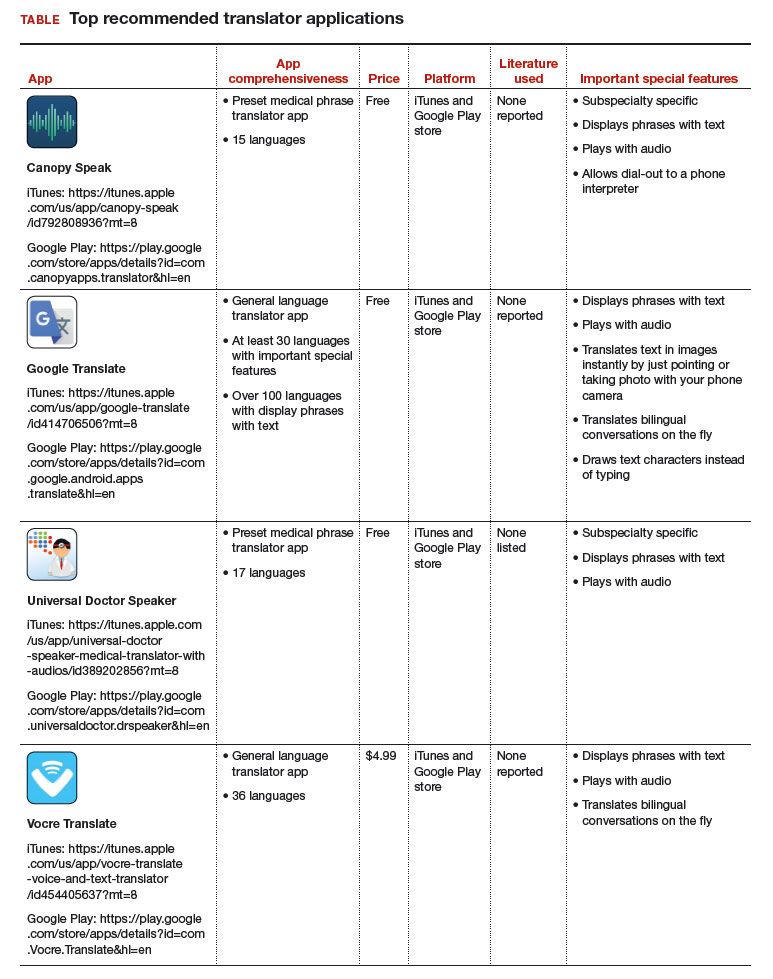
Share your thoughts! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
- United States Census Bureau. Detailed language spoken at home and ability to speak English for the population 5 years and over: 2009–2013. http://www.census.gov/data/tables/2013/demo/2009-2013-lang-tables.html. Published October 2015. Accessed August 31, 2017.
- United States Census Bureau. US population world clock. http://www.census.gov/popclock/?intcmp=home_pop. Accessed August 31, 2017.
- Khander A, Farag S, Chen KT. Identification and rating of medical translator mobile applications using the APPLICATIONS scoring system [abstract 321]. Obstet Gynecol. 2017;129(5 suppl):101S. doi:10.1097/01.AOG.0000514971.96123.20
- Chyjek K, Farag S, Chen KT. Rating pregnancy wheel applications using the APPLICATIONS scoring system. Obstet Gynecol. 2015;125(6):1478–1483.
As the population of patients with limited English proficiency increases throughout English-speaking countries, health care providers often need translator services. Medical translator smartphone applications (apps) are useful tools that can provide ad hoc translator services.
According to the US Census Bureau in 2015, more than 60 million individuals — about 19% of Americans — reported speaking a language other than English at home, and more than 25 million said that they speak English “less than very well.”1,2 The top 5 non-English languages spoken at home were Spanish, French, Chinese, Tagalog, and Vietnamese, encompassing 72% of non-English speakers.
In the health care sector, translator services are essential for providing accurate and culturally competent care. Current options for translator services include face-to-face interpreters, phone-based translator services, and translator apps on mobile devices. In settings where face-to-face interpreters or phone-based translator services are not available, translator apps may provide reasonable alternatives. My colleagues, Dr. Amrin Khander and Dr. Sara Farag, and I identified and evaluated medical translator apps that are available from the Apple iTunes and Google Play stores to aid clinicians in using such apps during clinical encounters.3
Three types of translator apps
Preset medical phrase translator apps require the user to search for or find a question or statement in order to facilitate a conversation. With these types of apps, a health care provider can choose fully conjugated sentences, which then can be played or read back to the patient in the chosen translated language. Within this group of apps, Canopy Speak and Universal Doctor Speaker are highly accessible, since both apps are available from the Apple iTunes and Google Play stores and both are free.
Medical dictionary apps require the user to search for a medical term in one language to receive a translation in another language. These apps are less useful, but they can help providers find and define specific terms in a given language.
General language translator apps require the user to enter a term, statement, or question in one language and then provide a translation in another language. Google Translate and Vocre Translate are examples.
The top recommended translator apps are listed in the TABLE alphabetically and are detailed with a shortened version of the APPLICATIONS scoring system, APPLI (app comprehensiveness, price, platform, literature use, and important special features).4 I hope the apps described here will help you enhance communication with your patients who have limited English proficiency.

Share your thoughts! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
As the population of patients with limited English proficiency increases throughout English-speaking countries, health care providers often need translator services. Medical translator smartphone applications (apps) are useful tools that can provide ad hoc translator services.
According to the US Census Bureau in 2015, more than 60 million individuals — about 19% of Americans — reported speaking a language other than English at home, and more than 25 million said that they speak English “less than very well.”1,2 The top 5 non-English languages spoken at home were Spanish, French, Chinese, Tagalog, and Vietnamese, encompassing 72% of non-English speakers.
In the health care sector, translator services are essential for providing accurate and culturally competent care. Current options for translator services include face-to-face interpreters, phone-based translator services, and translator apps on mobile devices. In settings where face-to-face interpreters or phone-based translator services are not available, translator apps may provide reasonable alternatives. My colleagues, Dr. Amrin Khander and Dr. Sara Farag, and I identified and evaluated medical translator apps that are available from the Apple iTunes and Google Play stores to aid clinicians in using such apps during clinical encounters.3
Three types of translator apps
Preset medical phrase translator apps require the user to search for or find a question or statement in order to facilitate a conversation. With these types of apps, a health care provider can choose fully conjugated sentences, which then can be played or read back to the patient in the chosen translated language. Within this group of apps, Canopy Speak and Universal Doctor Speaker are highly accessible, since both apps are available from the Apple iTunes and Google Play stores and both are free.
Medical dictionary apps require the user to search for a medical term in one language to receive a translation in another language. These apps are less useful, but they can help providers find and define specific terms in a given language.
General language translator apps require the user to enter a term, statement, or question in one language and then provide a translation in another language. Google Translate and Vocre Translate are examples.
The top recommended translator apps are listed in the TABLE alphabetically and are detailed with a shortened version of the APPLICATIONS scoring system, APPLI (app comprehensiveness, price, platform, literature use, and important special features).4 I hope the apps described here will help you enhance communication with your patients who have limited English proficiency.

Share your thoughts! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
- United States Census Bureau. Detailed language spoken at home and ability to speak English for the population 5 years and over: 2009–2013. http://www.census.gov/data/tables/2013/demo/2009-2013-lang-tables.html. Published October 2015. Accessed August 31, 2017.
- United States Census Bureau. US population world clock. http://www.census.gov/popclock/?intcmp=home_pop. Accessed August 31, 2017.
- Khander A, Farag S, Chen KT. Identification and rating of medical translator mobile applications using the APPLICATIONS scoring system [abstract 321]. Obstet Gynecol. 2017;129(5 suppl):101S. doi:10.1097/01.AOG.0000514971.96123.20
- Chyjek K, Farag S, Chen KT. Rating pregnancy wheel applications using the APPLICATIONS scoring system. Obstet Gynecol. 2015;125(6):1478–1483.
- United States Census Bureau. Detailed language spoken at home and ability to speak English for the population 5 years and over: 2009–2013. http://www.census.gov/data/tables/2013/demo/2009-2013-lang-tables.html. Published October 2015. Accessed August 31, 2017.
- United States Census Bureau. US population world clock. http://www.census.gov/popclock/?intcmp=home_pop. Accessed August 31, 2017.
- Khander A, Farag S, Chen KT. Identification and rating of medical translator mobile applications using the APPLICATIONS scoring system [abstract 321]. Obstet Gynecol. 2017;129(5 suppl):101S. doi:10.1097/01.AOG.0000514971.96123.20
- Chyjek K, Farag S, Chen KT. Rating pregnancy wheel applications using the APPLICATIONS scoring system. Obstet Gynecol. 2015;125(6):1478–1483.
Residents getting unprecedented attention from employers
Half of final-year residents surveyed this April had received 100 or more job solicitations during their training, according to physician-recruiting firm Merritt Hawkins.
Among the 935 survey responses the company received, 70% of physicians in their final year of residency had gotten more than 50 job offers: 20% received 51-100, and 50% received more than 100. The numbers were even higher for those in primary care: 76% had received at least 51 job solicitations, and 55% reported more than 100 offers, Merritt Hawkins said in its 2017 Survey of Final-Year Medical Residents.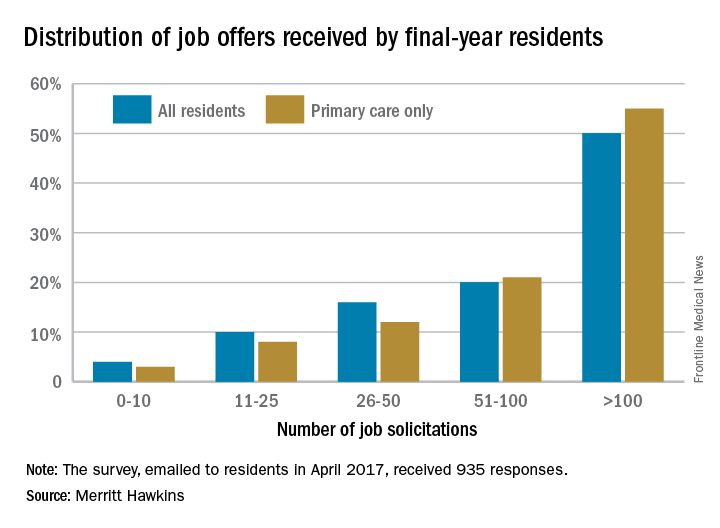
The 50% of all residents who received 100 or more job solicitations is the highest at that level since the survey began in 1991 (it is generally conducted every 2 or 3 years), with much lower figures seen as recently as 2008 – only 6% got that many offers – and 2006, when the number was 16%.
That primary care physicians are even more in demand shows their importance “as leaders of interdisciplinary clinical teams [and] managers of care and resources in emerging quality-driven delivery models such as ACOs [accountable care organizations],” the company said.
Demand for specialists is slightly lower, with 46% of residents in internal medicine, surgical, or diagnostic specialties getting more than 100 job offers and 64% getting more than 50. Current trends in mental health services, however, led Merritt Hawkins to single out psychiatry for special mention: 78% of final-year psychiatry residents had received more than 50 job solicitations – even more than primary care – although the number at the over-100 level, 48%, was lower, according to the report.
Half of final-year residents surveyed this April had received 100 or more job solicitations during their training, according to physician-recruiting firm Merritt Hawkins.
Among the 935 survey responses the company received, 70% of physicians in their final year of residency had gotten more than 50 job offers: 20% received 51-100, and 50% received more than 100. The numbers were even higher for those in primary care: 76% had received at least 51 job solicitations, and 55% reported more than 100 offers, Merritt Hawkins said in its 2017 Survey of Final-Year Medical Residents.
The 50% of all residents who received 100 or more job solicitations is the highest at that level since the survey began in 1991 (it is generally conducted every 2 or 3 years), with much lower figures seen as recently as 2008 – only 6% got that many offers – and 2006, when the number was 16%.
That primary care physicians are even more in demand shows their importance “as leaders of interdisciplinary clinical teams [and] managers of care and resources in emerging quality-driven delivery models such as ACOs [accountable care organizations],” the company said.
Demand for specialists is slightly lower, with 46% of residents in internal medicine, surgical, or diagnostic specialties getting more than 100 job offers and 64% getting more than 50. Current trends in mental health services, however, led Merritt Hawkins to single out psychiatry for special mention: 78% of final-year psychiatry residents had received more than 50 job solicitations – even more than primary care – although the number at the over-100 level, 48%, was lower, according to the report.
Half of final-year residents surveyed this April had received 100 or more job solicitations during their training, according to physician-recruiting firm Merritt Hawkins.
Among the 935 survey responses the company received, 70% of physicians in their final year of residency had gotten more than 50 job offers: 20% received 51-100, and 50% received more than 100. The numbers were even higher for those in primary care: 76% had received at least 51 job solicitations, and 55% reported more than 100 offers, Merritt Hawkins said in its 2017 Survey of Final-Year Medical Residents.
The 50% of all residents who received 100 or more job solicitations is the highest at that level since the survey began in 1991 (it is generally conducted every 2 or 3 years), with much lower figures seen as recently as 2008 – only 6% got that many offers – and 2006, when the number was 16%.
That primary care physicians are even more in demand shows their importance “as leaders of interdisciplinary clinical teams [and] managers of care and resources in emerging quality-driven delivery models such as ACOs [accountable care organizations],” the company said.
Demand for specialists is slightly lower, with 46% of residents in internal medicine, surgical, or diagnostic specialties getting more than 100 job offers and 64% getting more than 50. Current trends in mental health services, however, led Merritt Hawkins to single out psychiatry for special mention: 78% of final-year psychiatry residents had received more than 50 job solicitations – even more than primary care – although the number at the over-100 level, 48%, was lower, according to the report.
Videodermoscopy as a Novel Tool for Dermatologic Education
Dermoscopy, or the noninvasive in vivo examination of the epidermis and superficial dermis using magnification, facilitates the diagnosis of pigmented and nonpigmented skin lesions.1 Despite the benefit of dermoscopy in making early and accurate diagnoses of potentially life-limiting skin cancers, only 48% of dermatologists in the United States use dermoscopy in their practices.2 The most commonly cited reason for not using dermoscopy is lack of training.
Although the use of dermoscopy is associated with younger age and more recent graduation from residency compared to nonusers, dermatology resident physicians continue to receive limited training in dermoscopy.2 In a survey of 139 dermatology chief residents, 48% were not satisfied with the dermoscopy training that they had received during residency. Residents who received bedside instruction in dermoscopy reported greater satisfaction with their dermoscopy training compared to those who did not receive bedside instruction.3 This article provides a brief comparison of standard dermoscopy versus videodermoscopy for the instruction of trainees on common dermatologic diagnoses.
Bedside Dermoscopy
Standard optical dermatoscopes used for patient care and educational purposes typically incorporate 10-fold magnification and permit examination by a single viewer through a lens. With standard dermatoscopes, bedside dermoscopy instruction consists of the independent sequential viewing of skin lesions by instructors and trainees. Trainees must independently search for dermoscopic features noted by the instructor, which may be difficult for novice users. Simultaneous viewing of lesions would allow instructors to clearly indicate in real time pertinent dermoscopic features to their trainees.
Videodermatoscopes facilitate the simultaneous examination of cutaneous lesions by projecting the dermoscopic image onto a digital screen. Furthermore, these devices can incorporate magnifications of up to 200-fold or greater. In recent years, research pertaining to videodermoscopy has focused on the high magnification capabilities of these devices, specifically dermoscopic features that are visualized at magnifications greater than 10-fold, including the light brown nests of basal cell carcinomas that are seen at 50- to 70-fold magnification, twisted red capillary loops seen in active scalp psoriasis at 50-fold magnification, and longitudinal white indentations seen on nail plates affected by onychomycosis at 20-fold magnification.4-6 The potential value of videodermoscopy in medical education lies not only in the high magnification potential, which may make subtle dermoscopic findings more apparent to novice dermoscopists, but also in the ability to facilitate simultaneous dermoscopic examinations by instructors and trainees.
Educational Applications for Videodermoscopy
To illustrate the educational potential of videodermoscopy, images taken with a standard dermatoscope at 10-fold magnification are presented with videodermoscopic images taken at magnifications ranging from 60- to 185-fold (Figures 1–3). These examples demonstrate the potential for videodermoscopy to facilitate the visualization of subtle dermoscopic features by novice dermoscopists, relating to both the enhanced magnification potential and the potential for simultaneous rather than sequential examination.
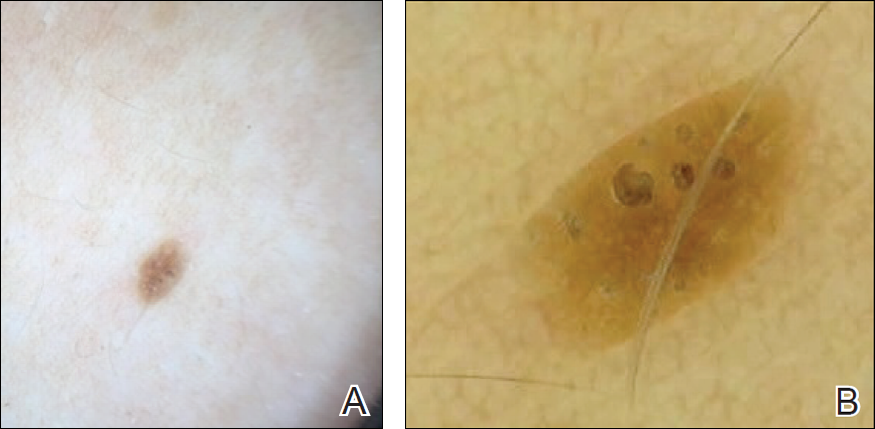
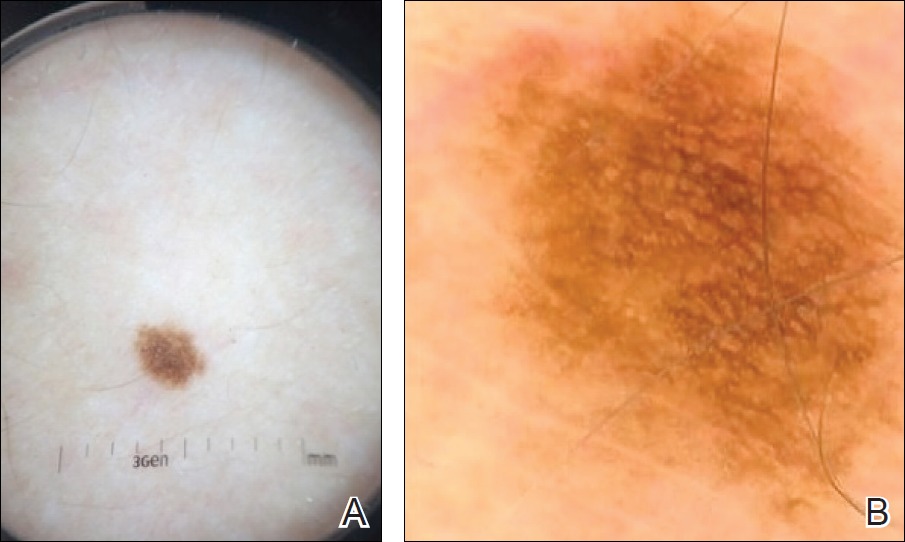

Final Thoughts
High-magnification videodermoscopy may be a useful tool to further dermoscopic education. Videodermatoscopes vary in functionality and cost but are available at price points comparable to those of standard optical dermatoscopes. Owners of standard dermatoscopes can approximate some of the benefits of a digital videodermatoscope by using the standard dermatoscope in conjunction with a camera, including those integrated into mobile phones and tablets. By attaching the standard dermatoscope to a camera with a digital display, the digital zoom of the camera can be used to magnify the standard dermoscopic image, enhancing the ability of novice dermoscopists to visualize subtle findings. By presenting this magnified image on a digital display, dermoscopy instructors and trainees would be able to simultaneously view dermoscopic images of lesions, sometimes with magnifications comparable to videodermatoscopes.
In the setting of a dermatology residency program, videodermoscopy can be incorporated into bedside teaching with experienced dermoscopists and for the live presentation of dermoscopic features at departmental grand rounds. By facilitating the simultaneous, high-magnification and live viewing of skin lesions by dermoscopy instructors and trainees, digital videodermoscopy has the potential to address an area of weakness in dermatologic training.
- Vestergaard ME, Macaskill P, Holt PE, et al. Dermoscopy compared with naked eye examination for the diagnosis of primary melanoma: a meta-analysis of studies performed in a clinical setting. Br J Dermatol. 2008;159:669-676.
- Engasser HC, Warshaw EM. Dermatoscopy use by US dermatologists: a cross-sectional survey [published online July 8, 2010]. J Am Acad Dermatol. 2010;63:412-419, 419.e1-419.e2.
- Wu TP, Newlove T, Smith L, et al. The importance of dedicated dermoscopy training during residency: a survey of US dermatology chief residents. J Am Acad Dermatol. 2013;68:1000-1005.
- Seidenari S, Bellucci C, Bassoli S, et al. High magnification digital dermoscopy of basal cell carcinoma: a single-centre study on 400 cases. Acta Derm Venereol. 2014;94:677-682.
- Ross EK, Vincenzi C, Tosti A. Videodermoscopy in the evaluation of hair and scalp disorders. J Am Acad Dermatol. 2006;55:799-806.
- Piraccini BM, Balestri R, Starace M, et al. Nail digital dermoscopy (onychoscopy) in the diagnosis of onychomycosis. J Eur Acad Dermatol Venereol. 2013;27:509-513.
Dermoscopy, or the noninvasive in vivo examination of the epidermis and superficial dermis using magnification, facilitates the diagnosis of pigmented and nonpigmented skin lesions.1 Despite the benefit of dermoscopy in making early and accurate diagnoses of potentially life-limiting skin cancers, only 48% of dermatologists in the United States use dermoscopy in their practices.2 The most commonly cited reason for not using dermoscopy is lack of training.
Although the use of dermoscopy is associated with younger age and more recent graduation from residency compared to nonusers, dermatology resident physicians continue to receive limited training in dermoscopy.2 In a survey of 139 dermatology chief residents, 48% were not satisfied with the dermoscopy training that they had received during residency. Residents who received bedside instruction in dermoscopy reported greater satisfaction with their dermoscopy training compared to those who did not receive bedside instruction.3 This article provides a brief comparison of standard dermoscopy versus videodermoscopy for the instruction of trainees on common dermatologic diagnoses.
Bedside Dermoscopy
Standard optical dermatoscopes used for patient care and educational purposes typically incorporate 10-fold magnification and permit examination by a single viewer through a lens. With standard dermatoscopes, bedside dermoscopy instruction consists of the independent sequential viewing of skin lesions by instructors and trainees. Trainees must independently search for dermoscopic features noted by the instructor, which may be difficult for novice users. Simultaneous viewing of lesions would allow instructors to clearly indicate in real time pertinent dermoscopic features to their trainees.
Videodermatoscopes facilitate the simultaneous examination of cutaneous lesions by projecting the dermoscopic image onto a digital screen. Furthermore, these devices can incorporate magnifications of up to 200-fold or greater. In recent years, research pertaining to videodermoscopy has focused on the high magnification capabilities of these devices, specifically dermoscopic features that are visualized at magnifications greater than 10-fold, including the light brown nests of basal cell carcinomas that are seen at 50- to 70-fold magnification, twisted red capillary loops seen in active scalp psoriasis at 50-fold magnification, and longitudinal white indentations seen on nail plates affected by onychomycosis at 20-fold magnification.4-6 The potential value of videodermoscopy in medical education lies not only in the high magnification potential, which may make subtle dermoscopic findings more apparent to novice dermoscopists, but also in the ability to facilitate simultaneous dermoscopic examinations by instructors and trainees.
Educational Applications for Videodermoscopy
To illustrate the educational potential of videodermoscopy, images taken with a standard dermatoscope at 10-fold magnification are presented with videodermoscopic images taken at magnifications ranging from 60- to 185-fold (Figures 1–3). These examples demonstrate the potential for videodermoscopy to facilitate the visualization of subtle dermoscopic features by novice dermoscopists, relating to both the enhanced magnification potential and the potential for simultaneous rather than sequential examination.



Final Thoughts
High-magnification videodermoscopy may be a useful tool to further dermoscopic education. Videodermatoscopes vary in functionality and cost but are available at price points comparable to those of standard optical dermatoscopes. Owners of standard dermatoscopes can approximate some of the benefits of a digital videodermatoscope by using the standard dermatoscope in conjunction with a camera, including those integrated into mobile phones and tablets. By attaching the standard dermatoscope to a camera with a digital display, the digital zoom of the camera can be used to magnify the standard dermoscopic image, enhancing the ability of novice dermoscopists to visualize subtle findings. By presenting this magnified image on a digital display, dermoscopy instructors and trainees would be able to simultaneously view dermoscopic images of lesions, sometimes with magnifications comparable to videodermatoscopes.
In the setting of a dermatology residency program, videodermoscopy can be incorporated into bedside teaching with experienced dermoscopists and for the live presentation of dermoscopic features at departmental grand rounds. By facilitating the simultaneous, high-magnification and live viewing of skin lesions by dermoscopy instructors and trainees, digital videodermoscopy has the potential to address an area of weakness in dermatologic training.
Dermoscopy, or the noninvasive in vivo examination of the epidermis and superficial dermis using magnification, facilitates the diagnosis of pigmented and nonpigmented skin lesions.1 Despite the benefit of dermoscopy in making early and accurate diagnoses of potentially life-limiting skin cancers, only 48% of dermatologists in the United States use dermoscopy in their practices.2 The most commonly cited reason for not using dermoscopy is lack of training.
Although the use of dermoscopy is associated with younger age and more recent graduation from residency compared to nonusers, dermatology resident physicians continue to receive limited training in dermoscopy.2 In a survey of 139 dermatology chief residents, 48% were not satisfied with the dermoscopy training that they had received during residency. Residents who received bedside instruction in dermoscopy reported greater satisfaction with their dermoscopy training compared to those who did not receive bedside instruction.3 This article provides a brief comparison of standard dermoscopy versus videodermoscopy for the instruction of trainees on common dermatologic diagnoses.
Bedside Dermoscopy
Standard optical dermatoscopes used for patient care and educational purposes typically incorporate 10-fold magnification and permit examination by a single viewer through a lens. With standard dermatoscopes, bedside dermoscopy instruction consists of the independent sequential viewing of skin lesions by instructors and trainees. Trainees must independently search for dermoscopic features noted by the instructor, which may be difficult for novice users. Simultaneous viewing of lesions would allow instructors to clearly indicate in real time pertinent dermoscopic features to their trainees.
Videodermatoscopes facilitate the simultaneous examination of cutaneous lesions by projecting the dermoscopic image onto a digital screen. Furthermore, these devices can incorporate magnifications of up to 200-fold or greater. In recent years, research pertaining to videodermoscopy has focused on the high magnification capabilities of these devices, specifically dermoscopic features that are visualized at magnifications greater than 10-fold, including the light brown nests of basal cell carcinomas that are seen at 50- to 70-fold magnification, twisted red capillary loops seen in active scalp psoriasis at 50-fold magnification, and longitudinal white indentations seen on nail plates affected by onychomycosis at 20-fold magnification.4-6 The potential value of videodermoscopy in medical education lies not only in the high magnification potential, which may make subtle dermoscopic findings more apparent to novice dermoscopists, but also in the ability to facilitate simultaneous dermoscopic examinations by instructors and trainees.
Educational Applications for Videodermoscopy
To illustrate the educational potential of videodermoscopy, images taken with a standard dermatoscope at 10-fold magnification are presented with videodermoscopic images taken at magnifications ranging from 60- to 185-fold (Figures 1–3). These examples demonstrate the potential for videodermoscopy to facilitate the visualization of subtle dermoscopic features by novice dermoscopists, relating to both the enhanced magnification potential and the potential for simultaneous rather than sequential examination.



Final Thoughts
High-magnification videodermoscopy may be a useful tool to further dermoscopic education. Videodermatoscopes vary in functionality and cost but are available at price points comparable to those of standard optical dermatoscopes. Owners of standard dermatoscopes can approximate some of the benefits of a digital videodermatoscope by using the standard dermatoscope in conjunction with a camera, including those integrated into mobile phones and tablets. By attaching the standard dermatoscope to a camera with a digital display, the digital zoom of the camera can be used to magnify the standard dermoscopic image, enhancing the ability of novice dermoscopists to visualize subtle findings. By presenting this magnified image on a digital display, dermoscopy instructors and trainees would be able to simultaneously view dermoscopic images of lesions, sometimes with magnifications comparable to videodermatoscopes.
In the setting of a dermatology residency program, videodermoscopy can be incorporated into bedside teaching with experienced dermoscopists and for the live presentation of dermoscopic features at departmental grand rounds. By facilitating the simultaneous, high-magnification and live viewing of skin lesions by dermoscopy instructors and trainees, digital videodermoscopy has the potential to address an area of weakness in dermatologic training.
- Vestergaard ME, Macaskill P, Holt PE, et al. Dermoscopy compared with naked eye examination for the diagnosis of primary melanoma: a meta-analysis of studies performed in a clinical setting. Br J Dermatol. 2008;159:669-676.
- Engasser HC, Warshaw EM. Dermatoscopy use by US dermatologists: a cross-sectional survey [published online July 8, 2010]. J Am Acad Dermatol. 2010;63:412-419, 419.e1-419.e2.
- Wu TP, Newlove T, Smith L, et al. The importance of dedicated dermoscopy training during residency: a survey of US dermatology chief residents. J Am Acad Dermatol. 2013;68:1000-1005.
- Seidenari S, Bellucci C, Bassoli S, et al. High magnification digital dermoscopy of basal cell carcinoma: a single-centre study on 400 cases. Acta Derm Venereol. 2014;94:677-682.
- Ross EK, Vincenzi C, Tosti A. Videodermoscopy in the evaluation of hair and scalp disorders. J Am Acad Dermatol. 2006;55:799-806.
- Piraccini BM, Balestri R, Starace M, et al. Nail digital dermoscopy (onychoscopy) in the diagnosis of onychomycosis. J Eur Acad Dermatol Venereol. 2013;27:509-513.
- Vestergaard ME, Macaskill P, Holt PE, et al. Dermoscopy compared with naked eye examination for the diagnosis of primary melanoma: a meta-analysis of studies performed in a clinical setting. Br J Dermatol. 2008;159:669-676.
- Engasser HC, Warshaw EM. Dermatoscopy use by US dermatologists: a cross-sectional survey [published online July 8, 2010]. J Am Acad Dermatol. 2010;63:412-419, 419.e1-419.e2.
- Wu TP, Newlove T, Smith L, et al. The importance of dedicated dermoscopy training during residency: a survey of US dermatology chief residents. J Am Acad Dermatol. 2013;68:1000-1005.
- Seidenari S, Bellucci C, Bassoli S, et al. High magnification digital dermoscopy of basal cell carcinoma: a single-centre study on 400 cases. Acta Derm Venereol. 2014;94:677-682.
- Ross EK, Vincenzi C, Tosti A. Videodermoscopy in the evaluation of hair and scalp disorders. J Am Acad Dermatol. 2006;55:799-806.
- Piraccini BM, Balestri R, Starace M, et al. Nail digital dermoscopy (onychoscopy) in the diagnosis of onychomycosis. J Eur Acad Dermatol Venereol. 2013;27:509-513.
Resident Pearl
- Bedside dermoscopy training can be enhanced through the use of videodermoscopy, which permits simultaneous, high-magnification viewing.