User login
Two more and counting: Suicide in medical trainees
Like everyone in the arc of social media impact, I was shocked and terribly saddened by the recent suicides of two New York women in medicine – a final-year medical student on May 1 and a second-year resident on May 5. As a specialist in physician health, a former training director, a long-standing member of our institution’s medical student admissions committee, and the ombudsman for our medical students, I am finding these tragedies harder and harder to reconcile. Something isn’t working. But before I get to that, what follows is a bulleted list of some events of the past couple of weeks that may give a context for my statements and have informed my two recommendations.
- May 3, 2018: I give an invited GI grand rounds on stress, burnout, depression, and suicide in physicians. The residents are quiet and say nothing. Faculty members seem only concerned about preventing and eradicating burnout – and not that interested in anything more severe.
- May 5: A psychiatry resident from Melbourne arrives to spend 10 days with me to do an elective in physician health. As in the United States, there is a significant suicide death rate in medical students and residents Down Under. In the afternoon, I present a paper at the annual meeting of the American Academy of Psychodynamic Psychiatry and Psychoanalysis on the use of psychotherapy in treatment-resistant suicidal depression in physicians. There is increasing hope that this essential modality of care will return to the contemporary psychiatrist’s toolbox.
- May 6: At the annual meeting of the American Psychiatric Association in New York, I’m the discussant for powerful heartfelt papers of five psychiatrists (mostly early career psychiatrists and one resident) that talked about living with a psychiatric illness. The audience is huge, and we hear narratives about internal stigma, self-disclosure, external stigma, shunning, bullying, acceptance, rejection, alienation, connection, and love by peers and family. The authenticity and valor of the speakers create an atmosphere of safety, which enables psychiatrists in attendance from all over the world to share their personal stories – some at the microphone, some privately.
- May 7: Again at the APA, I chair and facilitate a workshop on physician suicide. We hear from four speakers, all women, who have lost a loved one to suicide – a husband, a father, a brother, a son – all doctors. Two of the speakers are psychiatrists. The stories are gripping, detailed, and tender. Yes, the atmosphere is very sad, but there is not a pall. We learn how these doctors lived, not just how they died. They all loved medicine; they were creative; they cared deeply; they suffered silently; and with shame, they lost hope. Again, a big audience of psychiatrists, many of whom share their own stories, that they, too, had lost a physician son, wife, or mother to suicide. Some of their deceased family members fell through the cracks and did not receive the life-saving care they deserved; some, fearing assaults to their medical license, hospital privileges, or insurance, refused to see anyone. They died untreated.
- May 8: Still at the APA, a psychiatrist colleague and I collaborate on a clinical case conference. Each of us describes losing a physician patient to suicide. We walk the attendees through the clinical details of assessment, treatment, and the aftermath of their deaths. We talk openly and frankly about our feelings, grief, outreach to colleagues and the family, and our own personal journeys of learning, growth, and healing. The clinician audience members give constructive feedback, and some share their own stories of losing patients to suicide. Like the day before, some psychiatrists are grieving the loss of a physician son or sibling to suicide. As mental health professionals, they suffer from an additional layer of failure and guilt that a loved one died “under their watch.”
- May 8: I rush across the Javits Center to catch the discussant for a concurrent symposium on physician burnout and depression. She foregoes any prepared remarks to share her previous 48 hours with the audience. She is the training director of the program that lost the second-year resident on May 5. She did not learn of the death until 24 hours later. We are all on the edge of our seats as we listen to this grieving, courageous woman, a seasoned psychiatrist and educator, who has been blindsided by this tragedy. She has not slept. She called all of her residents and broke the news personally as best she could. Aided by “After A Suicide: A Toolkit for Residency/Fellowship Programs” (American Foundation for Suicide Prevention), she and her colleagues instituted a plan of action and worked with administration and faculty. Her strength and commitment to the well-being of her trainees is palpable and magnanimous. When the session ends, many of us stand in line to give her a hug. It is a stark reminder of how many lives are affected when someone you know or care about takes his/her own life – and how, in the house of medicine, medical students and residents really are part of an institutional family.
- May 10: I facilitate a meeting of our 12 second-year residents, many of whom knew of or had met the resident who died. Almost everyone speaks, shares their feelings, poses questions, and calls for answers and change. There is disbelief, sadness, confusion, some guilt, and lots of anger. Also a feeling of disillusionment or paradox about the field of psychiatry: “Of all branches of medicine, shouldn’t residents who are struggling with psychiatric issues feel safe, protected, cared for in psychiatry?” There is also a feeling of lip service being paid to personal treatment, as in quoted statements: “By all means, get treatment for your issues, but don’t let it encroach on your duty hours” or “It’s good you’re getting help, but do you still have to go weekly?”
In the immediate aftermath of suicide, feelings run high, as they should. But rather than wait it out – and fearing a return to “business as usual” – let me make only two suggestions:
2. In psychiatry, we need to redouble our efforts in fighting the stigma attached to psychiatric illness in trainees. It is unconscionable that medical students and residents are dying of treatable disorders (I’ve never heard of a doctor dying of cancer who didn’t go to an oncologist at least once), yet too many are not availing themselves of services we provide – even when they’re free of charge or covered by insurance. And are we certain that, when they knock on our doors, we are providing them with state-of-the-art care? Is it possible that unrecognized internal stigma and shame deep within us might make us hesitant to help our trainees in their hour of need? Or cut corners? Or not get a second opinion? Very few psychiatrists on faculty of our medical schools divulge their personal experiences of depression, posttraumatic stress disorders, substance use disorders, and more (with the exception of being in therapy during residency, which is normative and isn’t stigmatized). Coming out is leveling, humane, and respectful – and it shrinks the power differential in the teaching dyad. It might even save a life.
Dr. Myers is a professor of clinical psychiatry at State University of New York, Brooklyn, and the author of “Why Physicians Die by Suicide: Lessons Learned From Their Families and Others Who Cared.”
Like everyone in the arc of social media impact, I was shocked and terribly saddened by the recent suicides of two New York women in medicine – a final-year medical student on May 1 and a second-year resident on May 5. As a specialist in physician health, a former training director, a long-standing member of our institution’s medical student admissions committee, and the ombudsman for our medical students, I am finding these tragedies harder and harder to reconcile. Something isn’t working. But before I get to that, what follows is a bulleted list of some events of the past couple of weeks that may give a context for my statements and have informed my two recommendations.
- May 3, 2018: I give an invited GI grand rounds on stress, burnout, depression, and suicide in physicians. The residents are quiet and say nothing. Faculty members seem only concerned about preventing and eradicating burnout – and not that interested in anything more severe.
- May 5: A psychiatry resident from Melbourne arrives to spend 10 days with me to do an elective in physician health. As in the United States, there is a significant suicide death rate in medical students and residents Down Under. In the afternoon, I present a paper at the annual meeting of the American Academy of Psychodynamic Psychiatry and Psychoanalysis on the use of psychotherapy in treatment-resistant suicidal depression in physicians. There is increasing hope that this essential modality of care will return to the contemporary psychiatrist’s toolbox.
- May 6: At the annual meeting of the American Psychiatric Association in New York, I’m the discussant for powerful heartfelt papers of five psychiatrists (mostly early career psychiatrists and one resident) that talked about living with a psychiatric illness. The audience is huge, and we hear narratives about internal stigma, self-disclosure, external stigma, shunning, bullying, acceptance, rejection, alienation, connection, and love by peers and family. The authenticity and valor of the speakers create an atmosphere of safety, which enables psychiatrists in attendance from all over the world to share their personal stories – some at the microphone, some privately.
- May 7: Again at the APA, I chair and facilitate a workshop on physician suicide. We hear from four speakers, all women, who have lost a loved one to suicide – a husband, a father, a brother, a son – all doctors. Two of the speakers are psychiatrists. The stories are gripping, detailed, and tender. Yes, the atmosphere is very sad, but there is not a pall. We learn how these doctors lived, not just how they died. They all loved medicine; they were creative; they cared deeply; they suffered silently; and with shame, they lost hope. Again, a big audience of psychiatrists, many of whom share their own stories, that they, too, had lost a physician son, wife, or mother to suicide. Some of their deceased family members fell through the cracks and did not receive the life-saving care they deserved; some, fearing assaults to their medical license, hospital privileges, or insurance, refused to see anyone. They died untreated.
- May 8: Still at the APA, a psychiatrist colleague and I collaborate on a clinical case conference. Each of us describes losing a physician patient to suicide. We walk the attendees through the clinical details of assessment, treatment, and the aftermath of their deaths. We talk openly and frankly about our feelings, grief, outreach to colleagues and the family, and our own personal journeys of learning, growth, and healing. The clinician audience members give constructive feedback, and some share their own stories of losing patients to suicide. Like the day before, some psychiatrists are grieving the loss of a physician son or sibling to suicide. As mental health professionals, they suffer from an additional layer of failure and guilt that a loved one died “under their watch.”
- May 8: I rush across the Javits Center to catch the discussant for a concurrent symposium on physician burnout and depression. She foregoes any prepared remarks to share her previous 48 hours with the audience. She is the training director of the program that lost the second-year resident on May 5. She did not learn of the death until 24 hours later. We are all on the edge of our seats as we listen to this grieving, courageous woman, a seasoned psychiatrist and educator, who has been blindsided by this tragedy. She has not slept. She called all of her residents and broke the news personally as best she could. Aided by “After A Suicide: A Toolkit for Residency/Fellowship Programs” (American Foundation for Suicide Prevention), she and her colleagues instituted a plan of action and worked with administration and faculty. Her strength and commitment to the well-being of her trainees is palpable and magnanimous. When the session ends, many of us stand in line to give her a hug. It is a stark reminder of how many lives are affected when someone you know or care about takes his/her own life – and how, in the house of medicine, medical students and residents really are part of an institutional family.
- May 10: I facilitate a meeting of our 12 second-year residents, many of whom knew of or had met the resident who died. Almost everyone speaks, shares their feelings, poses questions, and calls for answers and change. There is disbelief, sadness, confusion, some guilt, and lots of anger. Also a feeling of disillusionment or paradox about the field of psychiatry: “Of all branches of medicine, shouldn’t residents who are struggling with psychiatric issues feel safe, protected, cared for in psychiatry?” There is also a feeling of lip service being paid to personal treatment, as in quoted statements: “By all means, get treatment for your issues, but don’t let it encroach on your duty hours” or “It’s good you’re getting help, but do you still have to go weekly?”
In the immediate aftermath of suicide, feelings run high, as they should. But rather than wait it out – and fearing a return to “business as usual” – let me make only two suggestions:
2. In psychiatry, we need to redouble our efforts in fighting the stigma attached to psychiatric illness in trainees. It is unconscionable that medical students and residents are dying of treatable disorders (I’ve never heard of a doctor dying of cancer who didn’t go to an oncologist at least once), yet too many are not availing themselves of services we provide – even when they’re free of charge or covered by insurance. And are we certain that, when they knock on our doors, we are providing them with state-of-the-art care? Is it possible that unrecognized internal stigma and shame deep within us might make us hesitant to help our trainees in their hour of need? Or cut corners? Or not get a second opinion? Very few psychiatrists on faculty of our medical schools divulge their personal experiences of depression, posttraumatic stress disorders, substance use disorders, and more (with the exception of being in therapy during residency, which is normative and isn’t stigmatized). Coming out is leveling, humane, and respectful – and it shrinks the power differential in the teaching dyad. It might even save a life.
Dr. Myers is a professor of clinical psychiatry at State University of New York, Brooklyn, and the author of “Why Physicians Die by Suicide: Lessons Learned From Their Families and Others Who Cared.”
Like everyone in the arc of social media impact, I was shocked and terribly saddened by the recent suicides of two New York women in medicine – a final-year medical student on May 1 and a second-year resident on May 5. As a specialist in physician health, a former training director, a long-standing member of our institution’s medical student admissions committee, and the ombudsman for our medical students, I am finding these tragedies harder and harder to reconcile. Something isn’t working. But before I get to that, what follows is a bulleted list of some events of the past couple of weeks that may give a context for my statements and have informed my two recommendations.
- May 3, 2018: I give an invited GI grand rounds on stress, burnout, depression, and suicide in physicians. The residents are quiet and say nothing. Faculty members seem only concerned about preventing and eradicating burnout – and not that interested in anything more severe.
- May 5: A psychiatry resident from Melbourne arrives to spend 10 days with me to do an elective in physician health. As in the United States, there is a significant suicide death rate in medical students and residents Down Under. In the afternoon, I present a paper at the annual meeting of the American Academy of Psychodynamic Psychiatry and Psychoanalysis on the use of psychotherapy in treatment-resistant suicidal depression in physicians. There is increasing hope that this essential modality of care will return to the contemporary psychiatrist’s toolbox.
- May 6: At the annual meeting of the American Psychiatric Association in New York, I’m the discussant for powerful heartfelt papers of five psychiatrists (mostly early career psychiatrists and one resident) that talked about living with a psychiatric illness. The audience is huge, and we hear narratives about internal stigma, self-disclosure, external stigma, shunning, bullying, acceptance, rejection, alienation, connection, and love by peers and family. The authenticity and valor of the speakers create an atmosphere of safety, which enables psychiatrists in attendance from all over the world to share their personal stories – some at the microphone, some privately.
- May 7: Again at the APA, I chair and facilitate a workshop on physician suicide. We hear from four speakers, all women, who have lost a loved one to suicide – a husband, a father, a brother, a son – all doctors. Two of the speakers are psychiatrists. The stories are gripping, detailed, and tender. Yes, the atmosphere is very sad, but there is not a pall. We learn how these doctors lived, not just how they died. They all loved medicine; they were creative; they cared deeply; they suffered silently; and with shame, they lost hope. Again, a big audience of psychiatrists, many of whom share their own stories, that they, too, had lost a physician son, wife, or mother to suicide. Some of their deceased family members fell through the cracks and did not receive the life-saving care they deserved; some, fearing assaults to their medical license, hospital privileges, or insurance, refused to see anyone. They died untreated.
- May 8: Still at the APA, a psychiatrist colleague and I collaborate on a clinical case conference. Each of us describes losing a physician patient to suicide. We walk the attendees through the clinical details of assessment, treatment, and the aftermath of their deaths. We talk openly and frankly about our feelings, grief, outreach to colleagues and the family, and our own personal journeys of learning, growth, and healing. The clinician audience members give constructive feedback, and some share their own stories of losing patients to suicide. Like the day before, some psychiatrists are grieving the loss of a physician son or sibling to suicide. As mental health professionals, they suffer from an additional layer of failure and guilt that a loved one died “under their watch.”
- May 8: I rush across the Javits Center to catch the discussant for a concurrent symposium on physician burnout and depression. She foregoes any prepared remarks to share her previous 48 hours with the audience. She is the training director of the program that lost the second-year resident on May 5. She did not learn of the death until 24 hours later. We are all on the edge of our seats as we listen to this grieving, courageous woman, a seasoned psychiatrist and educator, who has been blindsided by this tragedy. She has not slept. She called all of her residents and broke the news personally as best she could. Aided by “After A Suicide: A Toolkit for Residency/Fellowship Programs” (American Foundation for Suicide Prevention), she and her colleagues instituted a plan of action and worked with administration and faculty. Her strength and commitment to the well-being of her trainees is palpable and magnanimous. When the session ends, many of us stand in line to give her a hug. It is a stark reminder of how many lives are affected when someone you know or care about takes his/her own life – and how, in the house of medicine, medical students and residents really are part of an institutional family.
- May 10: I facilitate a meeting of our 12 second-year residents, many of whom knew of or had met the resident who died. Almost everyone speaks, shares their feelings, poses questions, and calls for answers and change. There is disbelief, sadness, confusion, some guilt, and lots of anger. Also a feeling of disillusionment or paradox about the field of psychiatry: “Of all branches of medicine, shouldn’t residents who are struggling with psychiatric issues feel safe, protected, cared for in psychiatry?” There is also a feeling of lip service being paid to personal treatment, as in quoted statements: “By all means, get treatment for your issues, but don’t let it encroach on your duty hours” or “It’s good you’re getting help, but do you still have to go weekly?”
In the immediate aftermath of suicide, feelings run high, as they should. But rather than wait it out – and fearing a return to “business as usual” – let me make only two suggestions:
2. In psychiatry, we need to redouble our efforts in fighting the stigma attached to psychiatric illness in trainees. It is unconscionable that medical students and residents are dying of treatable disorders (I’ve never heard of a doctor dying of cancer who didn’t go to an oncologist at least once), yet too many are not availing themselves of services we provide – even when they’re free of charge or covered by insurance. And are we certain that, when they knock on our doors, we are providing them with state-of-the-art care? Is it possible that unrecognized internal stigma and shame deep within us might make us hesitant to help our trainees in their hour of need? Or cut corners? Or not get a second opinion? Very few psychiatrists on faculty of our medical schools divulge their personal experiences of depression, posttraumatic stress disorders, substance use disorders, and more (with the exception of being in therapy during residency, which is normative and isn’t stigmatized). Coming out is leveling, humane, and respectful – and it shrinks the power differential in the teaching dyad. It might even save a life.
Dr. Myers is a professor of clinical psychiatry at State University of New York, Brooklyn, and the author of “Why Physicians Die by Suicide: Lessons Learned From Their Families and Others Who Cared.”
Diet
I’m about to embark on a controversial topic. Perhaps it’s safer to avoid, but I can’t put it off any longer. We need to talk about diet.
Discussing diet, like politics, religion, or salary, is best done just with oneself. Yet, I’m compelled to share what I’ve learned. First, I’m agnostic. I don’t believe you need to be vegan or paleo to be saved. I eat plant-based foods. I also eat things that eat plants. I’m sure you’d find a fine film of gluten in my kitchen. What I’ve learned is that for me, it doesn’t matter.
Specifically, I have little or nothing to eat from when I wake until dinner. As a busy dermatologist, that may seem draconian, but in fact it is easier than you might think. Patients are a constant all day, while hunger is fleeting. Got a craving at 10:15 a.m.? Easy. Walk in to see the next patient. Then repeat. Most days, this continues until 6:30 p.m. or so, when it’s time to head home. It’s not that hard, particularly when you don’t have anything in your office to eat except Dentyne Ice gum and green tea.
Now, this doesn’t always work. Why? Meetings. How do I manage fasting on those days? I don’t. If I know I have a lunch meeting scheduled, then I eat a healthy breakfast before I leave home, such as a protein smoothie or a bowl of hot oats with a dollop of Greek yogurt, sunflower seeds, walnuts, and berries. By eating a wholesome, well-balanced meal of fiber, carbs, lean protein, and good fats, I’m not starving before the meeting and am less likely to overeat. (That’s because I have also learned I’m not one of those enviable people who can simply say “no” to a crispy fish taco and guacamole if I’m hungry. I’m gonna eat it.) So, I avoid fasting and the inevitable frustration of breaking a fast on those days.
On days when I fast, I monitor how I feel. Fortunately, I have rarely felt hypoglycemic; except for that one Tuesday a couple of months ago. I had completed a long, hard early morning workout, and by mid-morning my hands were shaking and I felt nauseous. I quickly downed two RX bars and felt fine within minutes. Better for me, better for my patients.
Right now, intermittent fasting is working for me. Here’s my weekly plan:
I don’t fast on Fridays or weekends or when I travel. I eat out rarely. On weekends, my wife and I shop at the local farmers’ and fish markets to prepare ourselves for a week of healthy eating. And on Sundays, we continue our treasured family tradition of Sunday supper, which is basted with nostalgia and drizzled liberally with comfort. Often it requires long preparation, which is part of the appeal, and short attention is paid to its nutritional value. That’s not the point of Sunday dinner. A delicious dunk of fresh Italian bread in grassy-green olive oil or fresh pasta doused with homemade tomato basil sauce is the best possible meal I can have to prepare for a long, hard week ahead.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at dermnews@mdedge.com.
I’m about to embark on a controversial topic. Perhaps it’s safer to avoid, but I can’t put it off any longer. We need to talk about diet.
Discussing diet, like politics, religion, or salary, is best done just with oneself. Yet, I’m compelled to share what I’ve learned. First, I’m agnostic. I don’t believe you need to be vegan or paleo to be saved. I eat plant-based foods. I also eat things that eat plants. I’m sure you’d find a fine film of gluten in my kitchen. What I’ve learned is that for me, it doesn’t matter.
Specifically, I have little or nothing to eat from when I wake until dinner. As a busy dermatologist, that may seem draconian, but in fact it is easier than you might think. Patients are a constant all day, while hunger is fleeting. Got a craving at 10:15 a.m.? Easy. Walk in to see the next patient. Then repeat. Most days, this continues until 6:30 p.m. or so, when it’s time to head home. It’s not that hard, particularly when you don’t have anything in your office to eat except Dentyne Ice gum and green tea.
Now, this doesn’t always work. Why? Meetings. How do I manage fasting on those days? I don’t. If I know I have a lunch meeting scheduled, then I eat a healthy breakfast before I leave home, such as a protein smoothie or a bowl of hot oats with a dollop of Greek yogurt, sunflower seeds, walnuts, and berries. By eating a wholesome, well-balanced meal of fiber, carbs, lean protein, and good fats, I’m not starving before the meeting and am less likely to overeat. (That’s because I have also learned I’m not one of those enviable people who can simply say “no” to a crispy fish taco and guacamole if I’m hungry. I’m gonna eat it.) So, I avoid fasting and the inevitable frustration of breaking a fast on those days.
On days when I fast, I monitor how I feel. Fortunately, I have rarely felt hypoglycemic; except for that one Tuesday a couple of months ago. I had completed a long, hard early morning workout, and by mid-morning my hands were shaking and I felt nauseous. I quickly downed two RX bars and felt fine within minutes. Better for me, better for my patients.
Right now, intermittent fasting is working for me. Here’s my weekly plan:
I don’t fast on Fridays or weekends or when I travel. I eat out rarely. On weekends, my wife and I shop at the local farmers’ and fish markets to prepare ourselves for a week of healthy eating. And on Sundays, we continue our treasured family tradition of Sunday supper, which is basted with nostalgia and drizzled liberally with comfort. Often it requires long preparation, which is part of the appeal, and short attention is paid to its nutritional value. That’s not the point of Sunday dinner. A delicious dunk of fresh Italian bread in grassy-green olive oil or fresh pasta doused with homemade tomato basil sauce is the best possible meal I can have to prepare for a long, hard week ahead.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at dermnews@mdedge.com.
I’m about to embark on a controversial topic. Perhaps it’s safer to avoid, but I can’t put it off any longer. We need to talk about diet.
Discussing diet, like politics, religion, or salary, is best done just with oneself. Yet, I’m compelled to share what I’ve learned. First, I’m agnostic. I don’t believe you need to be vegan or paleo to be saved. I eat plant-based foods. I also eat things that eat plants. I’m sure you’d find a fine film of gluten in my kitchen. What I’ve learned is that for me, it doesn’t matter.
Specifically, I have little or nothing to eat from when I wake until dinner. As a busy dermatologist, that may seem draconian, but in fact it is easier than you might think. Patients are a constant all day, while hunger is fleeting. Got a craving at 10:15 a.m.? Easy. Walk in to see the next patient. Then repeat. Most days, this continues until 6:30 p.m. or so, when it’s time to head home. It’s not that hard, particularly when you don’t have anything in your office to eat except Dentyne Ice gum and green tea.
Now, this doesn’t always work. Why? Meetings. How do I manage fasting on those days? I don’t. If I know I have a lunch meeting scheduled, then I eat a healthy breakfast before I leave home, such as a protein smoothie or a bowl of hot oats with a dollop of Greek yogurt, sunflower seeds, walnuts, and berries. By eating a wholesome, well-balanced meal of fiber, carbs, lean protein, and good fats, I’m not starving before the meeting and am less likely to overeat. (That’s because I have also learned I’m not one of those enviable people who can simply say “no” to a crispy fish taco and guacamole if I’m hungry. I’m gonna eat it.) So, I avoid fasting and the inevitable frustration of breaking a fast on those days.
On days when I fast, I monitor how I feel. Fortunately, I have rarely felt hypoglycemic; except for that one Tuesday a couple of months ago. I had completed a long, hard early morning workout, and by mid-morning my hands were shaking and I felt nauseous. I quickly downed two RX bars and felt fine within minutes. Better for me, better for my patients.
Right now, intermittent fasting is working for me. Here’s my weekly plan:
I don’t fast on Fridays or weekends or when I travel. I eat out rarely. On weekends, my wife and I shop at the local farmers’ and fish markets to prepare ourselves for a week of healthy eating. And on Sundays, we continue our treasured family tradition of Sunday supper, which is basted with nostalgia and drizzled liberally with comfort. Often it requires long preparation, which is part of the appeal, and short attention is paid to its nutritional value. That’s not the point of Sunday dinner. A delicious dunk of fresh Italian bread in grassy-green olive oil or fresh pasta doused with homemade tomato basil sauce is the best possible meal I can have to prepare for a long, hard week ahead.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at dermnews@mdedge.com.
Tardive dyskinesia is theme of awards competition for early career psychiatrists
Important advances in neuroscience and clinical psychiatry have been achieved in recent years, but there are significant gaps in knowledge and much that we don’t understand about the brain and behavior. Further advances depend on cultivating and supporting a new generation of dedicated basic science and clinical investigators. While there is a compelling need to attract, recruit, and encourage talented individuals to pursue scholarly interests, competing life and career demands often prove daunting.
The theme of the competition this year concerning tardive dyskinesia is timely and consistent with the mission of NMSIS to promote knowledge on neurologic side effects of antipsychotic drugs. Tardive dyskinesia can have a negative impact on the social, psychological, and physical well-being of patients; it remains a legacy of past treatment with antipsychotics; it is an increasing concern among an ever widening population of patients receiving even newer antipsychotics; and there are now two Food and Drug Administration–approved treatments for the disorder. Early career psychiatrists may have had limited instruction on tardive dyskinesia, which has not received prominent attention in curricular programs in recent years. Thus, in addition to supporting scholarly work and research experience, the 2018 Promising Scholars Award Program aims to promote knowledge and skills in managing patients with tardive dyskinesia.
Specific learning objectives are:
- Participants will learn the steps necessary to prepare a scientific manuscript for publication.
- Participants will review comments by expert referees and learn to incorporate and respond to the peer review process.
- Participants will review the evidence related to the diagnosis and treatment of tardive dyskinesia.
- Participants will be introduced to the spectrum of educational and networking opportunities at the Institute for Psychiatric Services conference.
In the past, this program was very popular and gained national recognition among psychiatric trainees. Numerous submitted papers were accepted for publication in peer-reviewed journals after the competition was completed.
Instructions for manuscript preparation are:
- First author must be a student, resident, or fellow.
- Papers should address specific issues related to the theme of tardive dyskinesia and be no longer than 15 double-spaced typed pages in length (excluding references and illustrations).
- Literature reviews, case reports, or studies that are original and newly developed or recently published are acceptable.
- Reviews and feedback will be provided by a panel of academic psychiatrists.
- Papers will be judged on relevance to tardive dyskinesia, originality, scholarship, scientific rigor, valid methodology, clinical significance, and organization.
To participate, papers and curriculum vitae of the first author must be submitted by July 1, 2018, to Dianne Daugherty by email at dianne@mhaus.org. Winners will be announced by Aug. 10, 2018. For additional information, write to dianne@mhaus.org or visit www.mhaus.org/nmsis/about-us/what-is-nmsis.
Dr. Caroff, professor of psychiatry, Corporal Michael J. Crescenz VA Medical Center and at the University of Pennsylvania, both in Philadelphia, is director of the NMSIS. He served as consultant to Neurocrine Biosciences and Teva Pharmaceutical Industries, and receives research grant funding from Neurocrine Biosciences.
Important advances in neuroscience and clinical psychiatry have been achieved in recent years, but there are significant gaps in knowledge and much that we don’t understand about the brain and behavior. Further advances depend on cultivating and supporting a new generation of dedicated basic science and clinical investigators. While there is a compelling need to attract, recruit, and encourage talented individuals to pursue scholarly interests, competing life and career demands often prove daunting.
The theme of the competition this year concerning tardive dyskinesia is timely and consistent with the mission of NMSIS to promote knowledge on neurologic side effects of antipsychotic drugs. Tardive dyskinesia can have a negative impact on the social, psychological, and physical well-being of patients; it remains a legacy of past treatment with antipsychotics; it is an increasing concern among an ever widening population of patients receiving even newer antipsychotics; and there are now two Food and Drug Administration–approved treatments for the disorder. Early career psychiatrists may have had limited instruction on tardive dyskinesia, which has not received prominent attention in curricular programs in recent years. Thus, in addition to supporting scholarly work and research experience, the 2018 Promising Scholars Award Program aims to promote knowledge and skills in managing patients with tardive dyskinesia.
Specific learning objectives are:
- Participants will learn the steps necessary to prepare a scientific manuscript for publication.
- Participants will review comments by expert referees and learn to incorporate and respond to the peer review process.
- Participants will review the evidence related to the diagnosis and treatment of tardive dyskinesia.
- Participants will be introduced to the spectrum of educational and networking opportunities at the Institute for Psychiatric Services conference.
In the past, this program was very popular and gained national recognition among psychiatric trainees. Numerous submitted papers were accepted for publication in peer-reviewed journals after the competition was completed.
Instructions for manuscript preparation are:
- First author must be a student, resident, or fellow.
- Papers should address specific issues related to the theme of tardive dyskinesia and be no longer than 15 double-spaced typed pages in length (excluding references and illustrations).
- Literature reviews, case reports, or studies that are original and newly developed or recently published are acceptable.
- Reviews and feedback will be provided by a panel of academic psychiatrists.
- Papers will be judged on relevance to tardive dyskinesia, originality, scholarship, scientific rigor, valid methodology, clinical significance, and organization.
To participate, papers and curriculum vitae of the first author must be submitted by July 1, 2018, to Dianne Daugherty by email at dianne@mhaus.org. Winners will be announced by Aug. 10, 2018. For additional information, write to dianne@mhaus.org or visit www.mhaus.org/nmsis/about-us/what-is-nmsis.
Dr. Caroff, professor of psychiatry, Corporal Michael J. Crescenz VA Medical Center and at the University of Pennsylvania, both in Philadelphia, is director of the NMSIS. He served as consultant to Neurocrine Biosciences and Teva Pharmaceutical Industries, and receives research grant funding from Neurocrine Biosciences.
Important advances in neuroscience and clinical psychiatry have been achieved in recent years, but there are significant gaps in knowledge and much that we don’t understand about the brain and behavior. Further advances depend on cultivating and supporting a new generation of dedicated basic science and clinical investigators. While there is a compelling need to attract, recruit, and encourage talented individuals to pursue scholarly interests, competing life and career demands often prove daunting.
The theme of the competition this year concerning tardive dyskinesia is timely and consistent with the mission of NMSIS to promote knowledge on neurologic side effects of antipsychotic drugs. Tardive dyskinesia can have a negative impact on the social, psychological, and physical well-being of patients; it remains a legacy of past treatment with antipsychotics; it is an increasing concern among an ever widening population of patients receiving even newer antipsychotics; and there are now two Food and Drug Administration–approved treatments for the disorder. Early career psychiatrists may have had limited instruction on tardive dyskinesia, which has not received prominent attention in curricular programs in recent years. Thus, in addition to supporting scholarly work and research experience, the 2018 Promising Scholars Award Program aims to promote knowledge and skills in managing patients with tardive dyskinesia.
Specific learning objectives are:
- Participants will learn the steps necessary to prepare a scientific manuscript for publication.
- Participants will review comments by expert referees and learn to incorporate and respond to the peer review process.
- Participants will review the evidence related to the diagnosis and treatment of tardive dyskinesia.
- Participants will be introduced to the spectrum of educational and networking opportunities at the Institute for Psychiatric Services conference.
In the past, this program was very popular and gained national recognition among psychiatric trainees. Numerous submitted papers were accepted for publication in peer-reviewed journals after the competition was completed.
Instructions for manuscript preparation are:
- First author must be a student, resident, or fellow.
- Papers should address specific issues related to the theme of tardive dyskinesia and be no longer than 15 double-spaced typed pages in length (excluding references and illustrations).
- Literature reviews, case reports, or studies that are original and newly developed or recently published are acceptable.
- Reviews and feedback will be provided by a panel of academic psychiatrists.
- Papers will be judged on relevance to tardive dyskinesia, originality, scholarship, scientific rigor, valid methodology, clinical significance, and organization.
To participate, papers and curriculum vitae of the first author must be submitted by July 1, 2018, to Dianne Daugherty by email at dianne@mhaus.org. Winners will be announced by Aug. 10, 2018. For additional information, write to dianne@mhaus.org or visit www.mhaus.org/nmsis/about-us/what-is-nmsis.
Dr. Caroff, professor of psychiatry, Corporal Michael J. Crescenz VA Medical Center and at the University of Pennsylvania, both in Philadelphia, is director of the NMSIS. He served as consultant to Neurocrine Biosciences and Teva Pharmaceutical Industries, and receives research grant funding from Neurocrine Biosciences.
Perianal Extramammary Paget Disease Treated With Topical Imiquimod and Oral Cimetidine
Case Report
A 56-year-old woman with well-controlled hypertension, hyperlipidemia, and gastroesophageal reflux disease initially presented with itching and a rash in the perianal region of 1 year’s duration. She had been treated intermittently by her primary care physician over the past year for presumed hemorrhoids and a perianal fungal infection without improvement. Physical examination at the time of intitial presentation revealed a single, well-demarcated, scaly, pink plaque on the perianal area on the right buttock extending toward the anal canal (Figure 1).
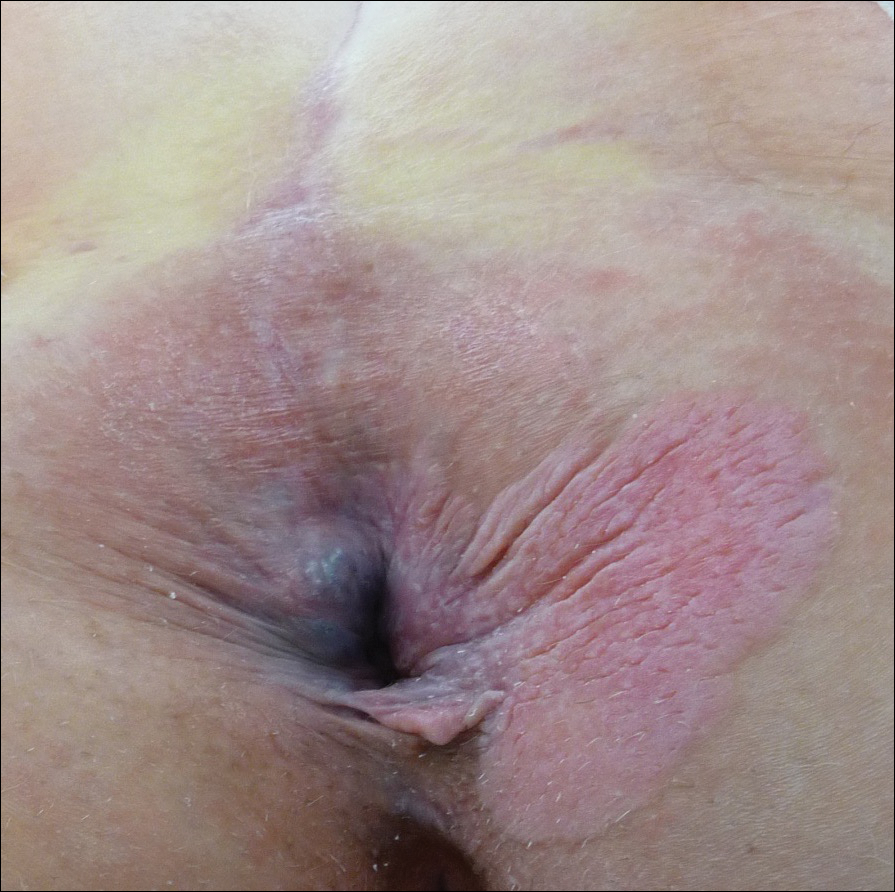
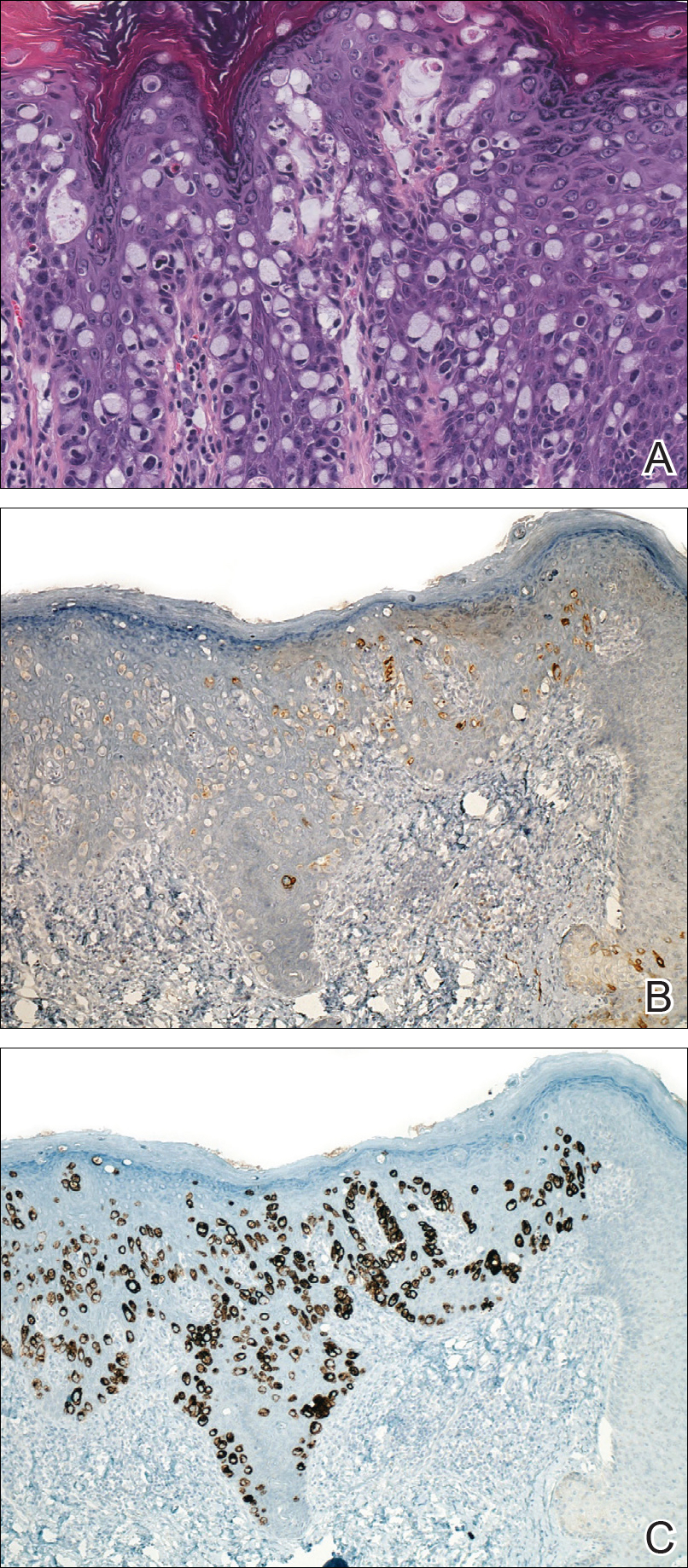
Four years later, the patient returned with new symptoms of bleeding when wiping the perianal region, pruritus, and fecal urgency of 3 to 4 months’ duration. Physical examination revealed scaly patches on the anus that were suspicious for recurrence of EMPD. Biopsies from the anal margin and anal canal confirmed recurrent EMPD involving the anal canal. Repeat evaluation for internal malignancy was negative.
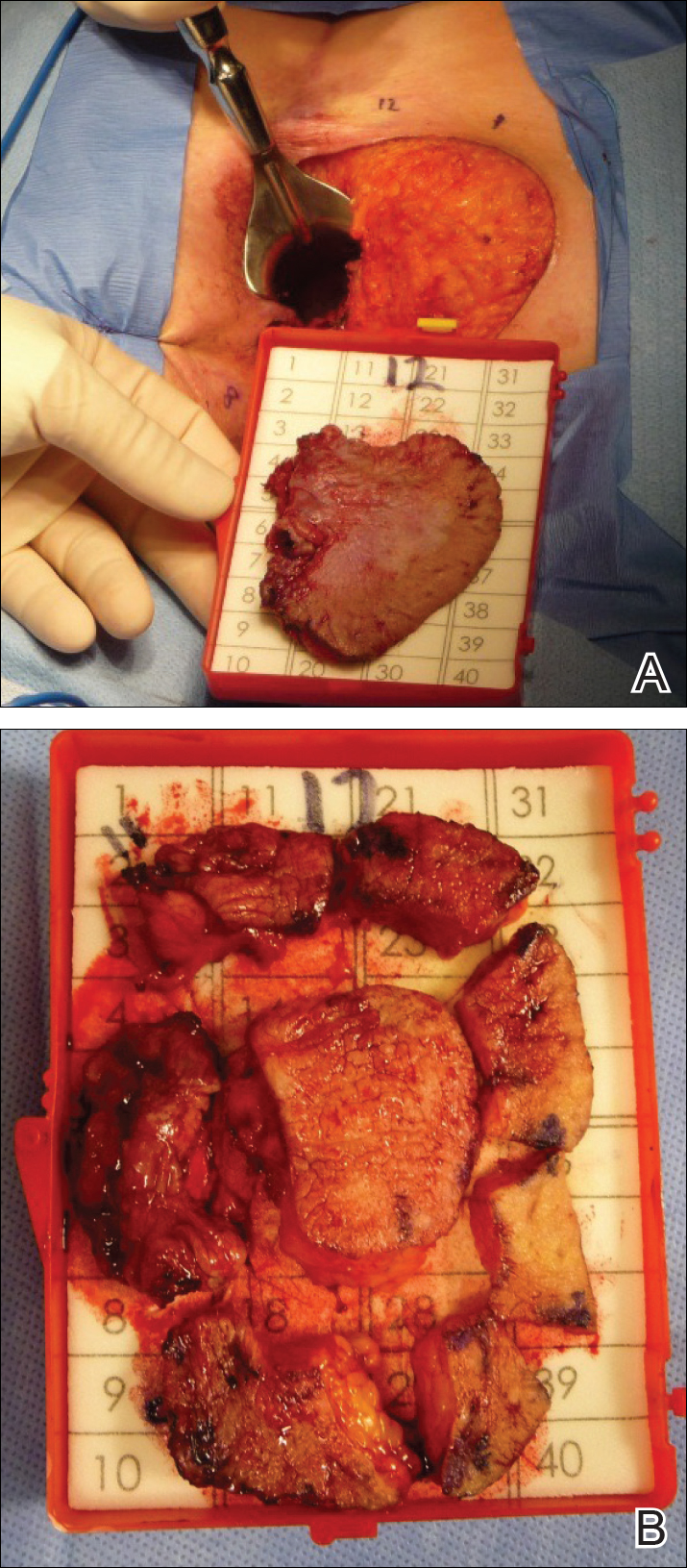
Given the involvement of the anal canal, repeat wide local excision would have required anal resection and would therefore have been functionally impairing. The patient refused further surgical intervention as well as radiotherapy. Rather, a novel 16-week immunomodulatory regimen involving imiquimod cream 5% cream and low-dose oral cimetidine was started. To address the anal involvement, the patient was instructed to lubricate glycerin suppositories with the imiquimod cream and insert intra-anally once weekly. Dosing was adjusted based on the patient’s inflammatory response and tolerability, as she did initially report some flulike symptoms with the first few weeks of treatment. For most of the 16-week course, she applied 250 mg of imiquimod cream 5% to the perianal area 3 times weekly and 250 mg into the anal canal once weekly. Oral cimetidine initially was dosed at 800 mg twice daily as tolerated, but due to stomach irritation, the patient self-reduced her intake to 800 mg 3 times weekly.
To determine treatment response, scouting biopsies of the anal margin and anal canal were obtained 4 weeks after treatment cessation and demonstrated no evidence of residual disease. The patient resumed topical imiquimod applied once weekly into the anal canal and around the anus for a planned prolonged course of at least 1 year. To reduce the risk of recurrence, the patient continued taking oral cimetidine 800 mg 3 times weekly. Recommended follow-up included annual anoscopy or colonoscopy, serum carcinoembryonic antigen evaluation, and regular clinical monitoring by the dermatology and colorectal surgery teams.
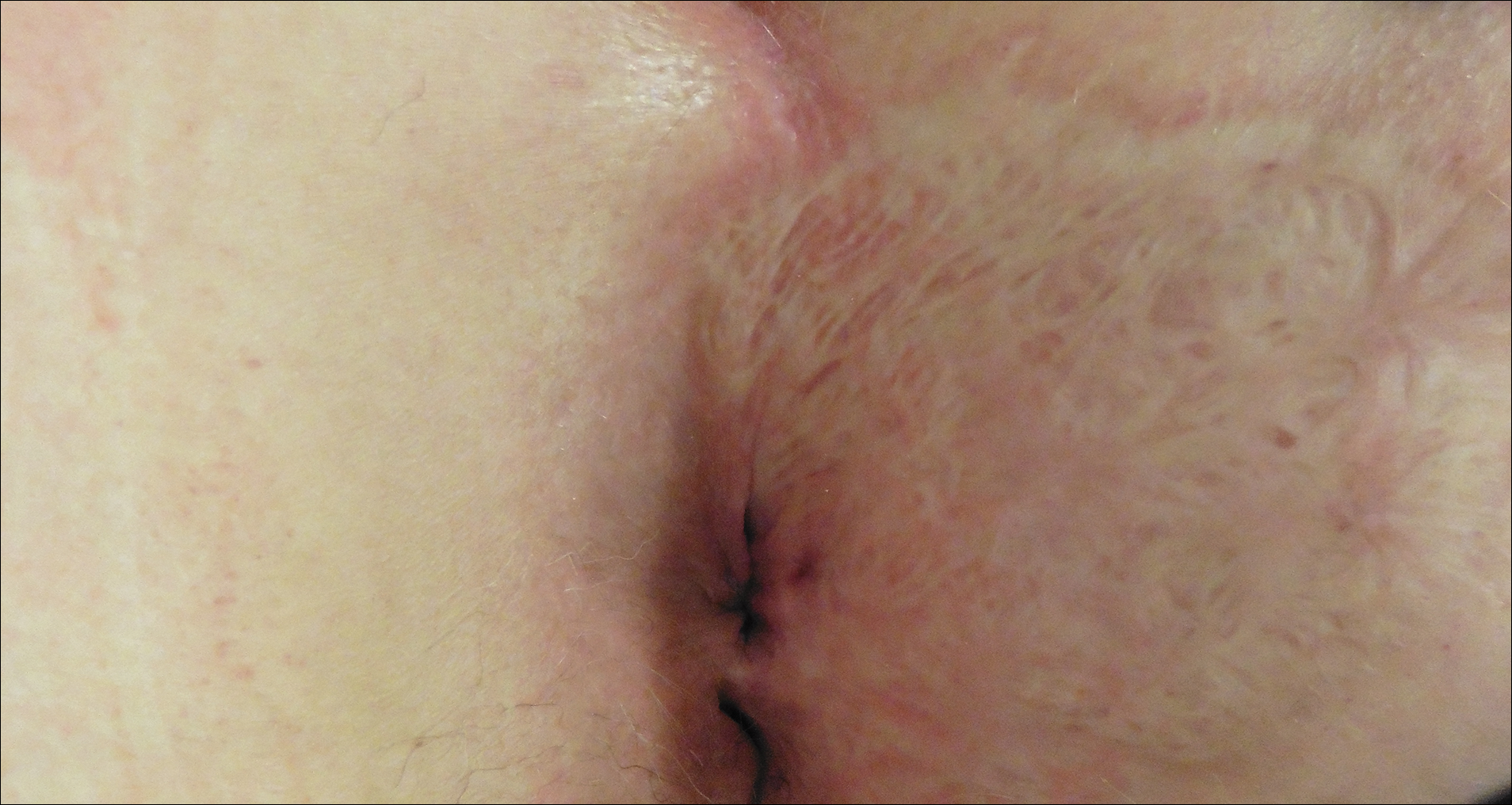
Six months after completing the combination therapy, she was seen by the dermatology department and remained clinically free of disease (Figure 4). Anoscopy examination by the colorectal surgery department 4 months later showed no clinical evidence of malignancy.
Comment
Extramammary Paget disease is a rare intraepithelial adenocarcinoma with a predilection for white females and an average age of onset of 50 to 80 years.1-3 The vulva, perianal region, scrotum, penis, and perineum are the most commonly affected sites.1-3 Clinically, EMPD presents as a chronic, well-demarcated, scaly, and often expanding plaque. The incidence of EMPD is unknown, as there are only a few hundred cases reported in the literature.2
Extramammary Paget disease can occur primarily, arising in the epidermis at the sweat-gland level or from primitive epidermal basal cells, or secondarily due to pagetoid spread of malignant cells from an adjacent or contiguous underlying adnexal adenocarcinoma or visceral malignancy.2 While primary EMPD is not associated with an underlying adenocarcinoma, it may become invasive, infiltrate the dermis, or metastasize via the lymphatics.2 Secondary EMPD is associated with underlying malignancy most often originating in the gastrointestinal or genitourinary tracts.1,2
Currently, treatment of primary EMPD typically is surgical with wide local excision or Mohs micrographic surgery.1,2 However, margins often are positive, and the local recurrence rate is high (ie, 33%–66%).2,3 There are a variety of other therapies that have been reported in the literature, including radiation, topical chemotherapeutics (eg, imiquimod, 5-fluorouracil, bleomycin), photodynamic therapy, and CO2 laser ablation.1,3 To our knowledge, there are no randomized controlled trials that compare surgery with other treatment options for EMPD.
Despite recurrence of EMPD with involvement of the anal canal, our patient refused further surgical intervention, as it would have required anal resection and radiotherapy due to the potentially negative impact on sphincter function. While investigating minimally invasive treatment options, we found several citations in the literature highlighting positive response with imiquimod cream 5% in patients with vulvar and periscrotal EMPD.4,5 A large, systematic review that analyzed 63 cases of vulvar EMPD—nearly half of which were recurrences of a prior malignancy—reported a response rate of 52% to 80% following treatment with imiquimod.5 Almost 70% of patients achieved complete clearance while applying imiquimod 3 to 4 times weekly for a median of 4 months; however, little has been written about the effectiveness of topical imiquimod in EMPD. Knight et al6 reported the case of a 40-year-old woman with perianal EMPD who was treated with imiquimod 3 times weekly for 16 weeks. At the end of treatment, the patient was completely clear of disease both clinically and histologically on random biopsies of the perianal skin; however, the EMPD later recurred with lymph node metastasis 18 months after stopping treatment.6
Given the growing evidence demonstrating disease control of EMPD with topical imiquimod, we elected to utilize this agent in combination with oral cimetidine in our patient. Cimetidine, an H2 receptor antagonist, has been shown to have antineoplastic properties in a broad range of preclinical and clinical studies for a number of different malignancies.7 Four distinct mechanisms of action have been shown. Cimetidine, which blocks the histamine pathway, has been shown to have a direct antiproliferative action on cancer cells.7 Histamine has been associated with increased regulatory T-cell activity, decreased antigen-presenting activity of dendritic cells, reduced natural killer cell activity, and increased myeloid-derived suppressor cell activity, which create an immunosuppressive tumor microenvironment in the setting of cancer. By blocking histamine and thus reversing this immunosuppressive environment, cimetidine demonstrates immunomodulatory effects.7 Cimetidine also has demonstrated an inhibitory effect on cancer cell adhesion to endothelial cells, which is noted to be independent of histamine-blocking activity.7 Finally, an antiangiogenic action is attributed to blocking of the upregulation of vascular endothelial growth factor that is normally induced by histamine.7
Cimetidine’s antineoplastic properties, specifically in the setting of colorectal cancer,8 were particularly compelling given our patient’s EMPD involvement of the anal canal. The most impressive clinical trial data showed a dramatically increased survival rate for colorectal cancer patients treated with oral cimetidine (800 mg once daily) and oral 5-fluorouracil (200 mg once daily) for 1 year following curative resection. The cimetidine-treated group had a 10-year survival rate of 84.6% versus 49.8% for the 5-fluorouracil–only group.8
Conclusion
We present this case of recurrent perianal and anal EMPD treated successfully with imiquimod cream 5% and oral cimetidine to highlight a potential alternative treatment regimen for poor surgical candidates with EMPD.
- Bolognia JL, Jorizzo JL, Schaffer JV. Dermatology. 3rd ed. London, England: Elsevier Saunders; 2012.
- Lam C, Funaro D. Extramammary Paget’s disease: summary of current knowledge. Dermatol Clin. 2010;28:807-826.
- Vergati M, Filingeri V, Palmieri G, et al. Perianal Paget’s disease: a case report and literature review. Anticancer Res. 2012;32:4461-4465.
- Liau MM, Yang SS, Tan KB, et al. Topical imiquimod in the treatment of extramammary Paget’s disease: a 10 year retrospective analysis in an Asian tertiary centre. Dermatol Ther. 2016;29:459-462.
- Machida H, Moeini A, Roman LD, et al. Effects of imiquimod on vulvar Paget’s disease: a systematic review of literature. Gynecol Oncol. 2015;139:165-171.
- Knight SR, Proby C, Ziyaie D, et al. Extramammary Paget disease of the perianal region: the potential role of imiquimod in achieving disease control. J Surg Case Rep. 2016;8:1-3.
- Pantziarka P, Bouche G, Meheus L, et al. Repurposing drugs in oncology (ReDO)—cimetidine as an anti-cancer agent. Ecancermedicalscience. 2014;8:485.
- Matsumoto S, Imaeda Y, Umemoto S, et al. Cimetidine increases survival of colorectal cancer patients with high levels of sialyl Lewis-X and sialyl Lewis-A epitope expression on tumour cells. Br J Cancer. 2002;86:161-167.
Case Report
A 56-year-old woman with well-controlled hypertension, hyperlipidemia, and gastroesophageal reflux disease initially presented with itching and a rash in the perianal region of 1 year’s duration. She had been treated intermittently by her primary care physician over the past year for presumed hemorrhoids and a perianal fungal infection without improvement. Physical examination at the time of intitial presentation revealed a single, well-demarcated, scaly, pink plaque on the perianal area on the right buttock extending toward the anal canal (Figure 1).



Four years later, the patient returned with new symptoms of bleeding when wiping the perianal region, pruritus, and fecal urgency of 3 to 4 months’ duration. Physical examination revealed scaly patches on the anus that were suspicious for recurrence of EMPD. Biopsies from the anal margin and anal canal confirmed recurrent EMPD involving the anal canal. Repeat evaluation for internal malignancy was negative.
Given the involvement of the anal canal, repeat wide local excision would have required anal resection and would therefore have been functionally impairing. The patient refused further surgical intervention as well as radiotherapy. Rather, a novel 16-week immunomodulatory regimen involving imiquimod cream 5% cream and low-dose oral cimetidine was started. To address the anal involvement, the patient was instructed to lubricate glycerin suppositories with the imiquimod cream and insert intra-anally once weekly. Dosing was adjusted based on the patient’s inflammatory response and tolerability, as she did initially report some flulike symptoms with the first few weeks of treatment. For most of the 16-week course, she applied 250 mg of imiquimod cream 5% to the perianal area 3 times weekly and 250 mg into the anal canal once weekly. Oral cimetidine initially was dosed at 800 mg twice daily as tolerated, but due to stomach irritation, the patient self-reduced her intake to 800 mg 3 times weekly.
To determine treatment response, scouting biopsies of the anal margin and anal canal were obtained 4 weeks after treatment cessation and demonstrated no evidence of residual disease. The patient resumed topical imiquimod applied once weekly into the anal canal and around the anus for a planned prolonged course of at least 1 year. To reduce the risk of recurrence, the patient continued taking oral cimetidine 800 mg 3 times weekly. Recommended follow-up included annual anoscopy or colonoscopy, serum carcinoembryonic antigen evaluation, and regular clinical monitoring by the dermatology and colorectal surgery teams.
Six months after completing the combination therapy, she was seen by the dermatology department and remained clinically free of disease (Figure 4). Anoscopy examination by the colorectal surgery department 4 months later showed no clinical evidence of malignancy.

Comment
Extramammary Paget disease is a rare intraepithelial adenocarcinoma with a predilection for white females and an average age of onset of 50 to 80 years.1-3 The vulva, perianal region, scrotum, penis, and perineum are the most commonly affected sites.1-3 Clinically, EMPD presents as a chronic, well-demarcated, scaly, and often expanding plaque. The incidence of EMPD is unknown, as there are only a few hundred cases reported in the literature.2
Extramammary Paget disease can occur primarily, arising in the epidermis at the sweat-gland level or from primitive epidermal basal cells, or secondarily due to pagetoid spread of malignant cells from an adjacent or contiguous underlying adnexal adenocarcinoma or visceral malignancy.2 While primary EMPD is not associated with an underlying adenocarcinoma, it may become invasive, infiltrate the dermis, or metastasize via the lymphatics.2 Secondary EMPD is associated with underlying malignancy most often originating in the gastrointestinal or genitourinary tracts.1,2
Currently, treatment of primary EMPD typically is surgical with wide local excision or Mohs micrographic surgery.1,2 However, margins often are positive, and the local recurrence rate is high (ie, 33%–66%).2,3 There are a variety of other therapies that have been reported in the literature, including radiation, topical chemotherapeutics (eg, imiquimod, 5-fluorouracil, bleomycin), photodynamic therapy, and CO2 laser ablation.1,3 To our knowledge, there are no randomized controlled trials that compare surgery with other treatment options for EMPD.
Despite recurrence of EMPD with involvement of the anal canal, our patient refused further surgical intervention, as it would have required anal resection and radiotherapy due to the potentially negative impact on sphincter function. While investigating minimally invasive treatment options, we found several citations in the literature highlighting positive response with imiquimod cream 5% in patients with vulvar and periscrotal EMPD.4,5 A large, systematic review that analyzed 63 cases of vulvar EMPD—nearly half of which were recurrences of a prior malignancy—reported a response rate of 52% to 80% following treatment with imiquimod.5 Almost 70% of patients achieved complete clearance while applying imiquimod 3 to 4 times weekly for a median of 4 months; however, little has been written about the effectiveness of topical imiquimod in EMPD. Knight et al6 reported the case of a 40-year-old woman with perianal EMPD who was treated with imiquimod 3 times weekly for 16 weeks. At the end of treatment, the patient was completely clear of disease both clinically and histologically on random biopsies of the perianal skin; however, the EMPD later recurred with lymph node metastasis 18 months after stopping treatment.6
Given the growing evidence demonstrating disease control of EMPD with topical imiquimod, we elected to utilize this agent in combination with oral cimetidine in our patient. Cimetidine, an H2 receptor antagonist, has been shown to have antineoplastic properties in a broad range of preclinical and clinical studies for a number of different malignancies.7 Four distinct mechanisms of action have been shown. Cimetidine, which blocks the histamine pathway, has been shown to have a direct antiproliferative action on cancer cells.7 Histamine has been associated with increased regulatory T-cell activity, decreased antigen-presenting activity of dendritic cells, reduced natural killer cell activity, and increased myeloid-derived suppressor cell activity, which create an immunosuppressive tumor microenvironment in the setting of cancer. By blocking histamine and thus reversing this immunosuppressive environment, cimetidine demonstrates immunomodulatory effects.7 Cimetidine also has demonstrated an inhibitory effect on cancer cell adhesion to endothelial cells, which is noted to be independent of histamine-blocking activity.7 Finally, an antiangiogenic action is attributed to blocking of the upregulation of vascular endothelial growth factor that is normally induced by histamine.7
Cimetidine’s antineoplastic properties, specifically in the setting of colorectal cancer,8 were particularly compelling given our patient’s EMPD involvement of the anal canal. The most impressive clinical trial data showed a dramatically increased survival rate for colorectal cancer patients treated with oral cimetidine (800 mg once daily) and oral 5-fluorouracil (200 mg once daily) for 1 year following curative resection. The cimetidine-treated group had a 10-year survival rate of 84.6% versus 49.8% for the 5-fluorouracil–only group.8
Conclusion
We present this case of recurrent perianal and anal EMPD treated successfully with imiquimod cream 5% and oral cimetidine to highlight a potential alternative treatment regimen for poor surgical candidates with EMPD.
Case Report
A 56-year-old woman with well-controlled hypertension, hyperlipidemia, and gastroesophageal reflux disease initially presented with itching and a rash in the perianal region of 1 year’s duration. She had been treated intermittently by her primary care physician over the past year for presumed hemorrhoids and a perianal fungal infection without improvement. Physical examination at the time of intitial presentation revealed a single, well-demarcated, scaly, pink plaque on the perianal area on the right buttock extending toward the anal canal (Figure 1).



Four years later, the patient returned with new symptoms of bleeding when wiping the perianal region, pruritus, and fecal urgency of 3 to 4 months’ duration. Physical examination revealed scaly patches on the anus that were suspicious for recurrence of EMPD. Biopsies from the anal margin and anal canal confirmed recurrent EMPD involving the anal canal. Repeat evaluation for internal malignancy was negative.
Given the involvement of the anal canal, repeat wide local excision would have required anal resection and would therefore have been functionally impairing. The patient refused further surgical intervention as well as radiotherapy. Rather, a novel 16-week immunomodulatory regimen involving imiquimod cream 5% cream and low-dose oral cimetidine was started. To address the anal involvement, the patient was instructed to lubricate glycerin suppositories with the imiquimod cream and insert intra-anally once weekly. Dosing was adjusted based on the patient’s inflammatory response and tolerability, as she did initially report some flulike symptoms with the first few weeks of treatment. For most of the 16-week course, she applied 250 mg of imiquimod cream 5% to the perianal area 3 times weekly and 250 mg into the anal canal once weekly. Oral cimetidine initially was dosed at 800 mg twice daily as tolerated, but due to stomach irritation, the patient self-reduced her intake to 800 mg 3 times weekly.
To determine treatment response, scouting biopsies of the anal margin and anal canal were obtained 4 weeks after treatment cessation and demonstrated no evidence of residual disease. The patient resumed topical imiquimod applied once weekly into the anal canal and around the anus for a planned prolonged course of at least 1 year. To reduce the risk of recurrence, the patient continued taking oral cimetidine 800 mg 3 times weekly. Recommended follow-up included annual anoscopy or colonoscopy, serum carcinoembryonic antigen evaluation, and regular clinical monitoring by the dermatology and colorectal surgery teams.
Six months after completing the combination therapy, she was seen by the dermatology department and remained clinically free of disease (Figure 4). Anoscopy examination by the colorectal surgery department 4 months later showed no clinical evidence of malignancy.

Comment
Extramammary Paget disease is a rare intraepithelial adenocarcinoma with a predilection for white females and an average age of onset of 50 to 80 years.1-3 The vulva, perianal region, scrotum, penis, and perineum are the most commonly affected sites.1-3 Clinically, EMPD presents as a chronic, well-demarcated, scaly, and often expanding plaque. The incidence of EMPD is unknown, as there are only a few hundred cases reported in the literature.2
Extramammary Paget disease can occur primarily, arising in the epidermis at the sweat-gland level or from primitive epidermal basal cells, or secondarily due to pagetoid spread of malignant cells from an adjacent or contiguous underlying adnexal adenocarcinoma or visceral malignancy.2 While primary EMPD is not associated with an underlying adenocarcinoma, it may become invasive, infiltrate the dermis, or metastasize via the lymphatics.2 Secondary EMPD is associated with underlying malignancy most often originating in the gastrointestinal or genitourinary tracts.1,2
Currently, treatment of primary EMPD typically is surgical with wide local excision or Mohs micrographic surgery.1,2 However, margins often are positive, and the local recurrence rate is high (ie, 33%–66%).2,3 There are a variety of other therapies that have been reported in the literature, including radiation, topical chemotherapeutics (eg, imiquimod, 5-fluorouracil, bleomycin), photodynamic therapy, and CO2 laser ablation.1,3 To our knowledge, there are no randomized controlled trials that compare surgery with other treatment options for EMPD.
Despite recurrence of EMPD with involvement of the anal canal, our patient refused further surgical intervention, as it would have required anal resection and radiotherapy due to the potentially negative impact on sphincter function. While investigating minimally invasive treatment options, we found several citations in the literature highlighting positive response with imiquimod cream 5% in patients with vulvar and periscrotal EMPD.4,5 A large, systematic review that analyzed 63 cases of vulvar EMPD—nearly half of which were recurrences of a prior malignancy—reported a response rate of 52% to 80% following treatment with imiquimod.5 Almost 70% of patients achieved complete clearance while applying imiquimod 3 to 4 times weekly for a median of 4 months; however, little has been written about the effectiveness of topical imiquimod in EMPD. Knight et al6 reported the case of a 40-year-old woman with perianal EMPD who was treated with imiquimod 3 times weekly for 16 weeks. At the end of treatment, the patient was completely clear of disease both clinically and histologically on random biopsies of the perianal skin; however, the EMPD later recurred with lymph node metastasis 18 months after stopping treatment.6
Given the growing evidence demonstrating disease control of EMPD with topical imiquimod, we elected to utilize this agent in combination with oral cimetidine in our patient. Cimetidine, an H2 receptor antagonist, has been shown to have antineoplastic properties in a broad range of preclinical and clinical studies for a number of different malignancies.7 Four distinct mechanisms of action have been shown. Cimetidine, which blocks the histamine pathway, has been shown to have a direct antiproliferative action on cancer cells.7 Histamine has been associated with increased regulatory T-cell activity, decreased antigen-presenting activity of dendritic cells, reduced natural killer cell activity, and increased myeloid-derived suppressor cell activity, which create an immunosuppressive tumor microenvironment in the setting of cancer. By blocking histamine and thus reversing this immunosuppressive environment, cimetidine demonstrates immunomodulatory effects.7 Cimetidine also has demonstrated an inhibitory effect on cancer cell adhesion to endothelial cells, which is noted to be independent of histamine-blocking activity.7 Finally, an antiangiogenic action is attributed to blocking of the upregulation of vascular endothelial growth factor that is normally induced by histamine.7
Cimetidine’s antineoplastic properties, specifically in the setting of colorectal cancer,8 were particularly compelling given our patient’s EMPD involvement of the anal canal. The most impressive clinical trial data showed a dramatically increased survival rate for colorectal cancer patients treated with oral cimetidine (800 mg once daily) and oral 5-fluorouracil (200 mg once daily) for 1 year following curative resection. The cimetidine-treated group had a 10-year survival rate of 84.6% versus 49.8% for the 5-fluorouracil–only group.8
Conclusion
We present this case of recurrent perianal and anal EMPD treated successfully with imiquimod cream 5% and oral cimetidine to highlight a potential alternative treatment regimen for poor surgical candidates with EMPD.
- Bolognia JL, Jorizzo JL, Schaffer JV. Dermatology. 3rd ed. London, England: Elsevier Saunders; 2012.
- Lam C, Funaro D. Extramammary Paget’s disease: summary of current knowledge. Dermatol Clin. 2010;28:807-826.
- Vergati M, Filingeri V, Palmieri G, et al. Perianal Paget’s disease: a case report and literature review. Anticancer Res. 2012;32:4461-4465.
- Liau MM, Yang SS, Tan KB, et al. Topical imiquimod in the treatment of extramammary Paget’s disease: a 10 year retrospective analysis in an Asian tertiary centre. Dermatol Ther. 2016;29:459-462.
- Machida H, Moeini A, Roman LD, et al. Effects of imiquimod on vulvar Paget’s disease: a systematic review of literature. Gynecol Oncol. 2015;139:165-171.
- Knight SR, Proby C, Ziyaie D, et al. Extramammary Paget disease of the perianal region: the potential role of imiquimod in achieving disease control. J Surg Case Rep. 2016;8:1-3.
- Pantziarka P, Bouche G, Meheus L, et al. Repurposing drugs in oncology (ReDO)—cimetidine as an anti-cancer agent. Ecancermedicalscience. 2014;8:485.
- Matsumoto S, Imaeda Y, Umemoto S, et al. Cimetidine increases survival of colorectal cancer patients with high levels of sialyl Lewis-X and sialyl Lewis-A epitope expression on tumour cells. Br J Cancer. 2002;86:161-167.
- Bolognia JL, Jorizzo JL, Schaffer JV. Dermatology. 3rd ed. London, England: Elsevier Saunders; 2012.
- Lam C, Funaro D. Extramammary Paget’s disease: summary of current knowledge. Dermatol Clin. 2010;28:807-826.
- Vergati M, Filingeri V, Palmieri G, et al. Perianal Paget’s disease: a case report and literature review. Anticancer Res. 2012;32:4461-4465.
- Liau MM, Yang SS, Tan KB, et al. Topical imiquimod in the treatment of extramammary Paget’s disease: a 10 year retrospective analysis in an Asian tertiary centre. Dermatol Ther. 2016;29:459-462.
- Machida H, Moeini A, Roman LD, et al. Effects of imiquimod on vulvar Paget’s disease: a systematic review of literature. Gynecol Oncol. 2015;139:165-171.
- Knight SR, Proby C, Ziyaie D, et al. Extramammary Paget disease of the perianal region: the potential role of imiquimod in achieving disease control. J Surg Case Rep. 2016;8:1-3.
- Pantziarka P, Bouche G, Meheus L, et al. Repurposing drugs in oncology (ReDO)—cimetidine as an anti-cancer agent. Ecancermedicalscience. 2014;8:485.
- Matsumoto S, Imaeda Y, Umemoto S, et al. Cimetidine increases survival of colorectal cancer patients with high levels of sialyl Lewis-X and sialyl Lewis-A epitope expression on tumour cells. Br J Cancer. 2002;86:161-167.
Resident Pearls
- Topical imiquimod cream 5% and oral cimetidine can be a potential alternative treatment regimen for poor surgical candidates with perianal extramammary Paget disease (EMPD).
- Its antineoplastic and immunomodulatory properties may suggest a role for oral cimetidine as an adjuvant therapy in the treatment of perianal EMPD.
Career Choices: Directorship/leadership
Editor’s note: Career Choices features a psychiatry resident/fellow interviewing a psychiatrist about why he or she has chosen a specific career path. The goal is to inform trainees about the various psychiatric career options, and to give them a feel for the pros and cons of the various paths.
In this Career Choices, Cornel Stanciu, MD, talked with Thomas Penders, MS, MD. For most of his career, Dr. Penders has practiced in directorship roles. He currently serves as the leader of an addiction consultation service at the Walter B. Jones Center in Greenville, North Carolina, as well as working at the state level with federally qualified health centers to develop collaborative care models.
Dr. Stanciu: What led you to decide to pursue a director role?
Dr. Penders: Early in my career, I was offered opportunities to provide leadership for an organization in its efforts to assure quality and availability of appropriate medical and psychiatric care.
Dr. Stanciu: How has the director role evolved over the years?
Dr. Penders: Thirty years ago, when I got started, hospital administrations depended heavily on medical directors to provide advice on new service initiates. Medical directors were frequently provided with support by health care organizations when recommendations were made based on patient and community need as perceived by medical staff providers. There has been a dramatic shift in the relationship and role of medical directorship, particularly over the past decade. Budgetary constraints have influenced planning and operational decisions to the extent that these decisions are much more likely to be made based on financial analyses rather than on clinical needs identified by physicians. As a result, medical directors are encouraged to be mindful of the effect of their suggestions on the bottom line of the organization. This has resulted in a very significant shift away from programs that are needed but not funded, and toward programs that are revenue-positive or at least neutral.
Medical directors who do not conform in this way are unlikely to be part of the administration for very long in the present environment.
Continue to: What training qualifications are required or desirable to assume a medical leadership role (post residency fellowship, MBA, etc.)?
Dr. Stanciu: What training qualifications are required or desirable to assume a medical leadership role (post-residency fellowship, MBA, etc.)?
Dr. Penders: In addition to a foundation in evidence-based practices and knowledge of regulatory requirements, general leadership skills are probably the most important qualities for medical leadership. Hospitals are complex organizations with confusing reporting relationships. Negotiation skills and communication skills are critical to success. Because most modern health care organizations are well staffed with administrative personnel trained in business and finance, I would not suggest that an MBA is necessary or even important to a medical director’s success. Having said that, there are an increasing number of physicians assuming the role of chief executive officer in complex health care systems. In this case, MBA training will likely be advantageous.
I would suggest that the focus of training that occurs in MPH programs would provide more relevant tools for those in positions of medical leadership. Skills such as biostatistics and epidemiology provide those in such positions with the perspective required to understand the effectiveness of health care systems, and to relate to changes that might be beneficial to the populations they serve. A firm foundation in information systems and data analysis is becoming increasingly important as the payment system moves toward one that is value-based. Increasingly, health care systems decisions will be guided by the analysis of aggregated information gathered from electronic medical records.
Dr. Stanciu: What personal qualities makes a psychiatric physician well-suited for the role of a medical director?
Dr. Penders: Medical directors will confront a variety of difficult situations with colleagues, administrative staff, patients, and family members. A calm demeanor with an ability to reflect rather than react is important. As I previously mentioned, an ability to communicate, including strength as a listener, is another personality trait valued in this position.
Continue to: What are some of the challenges you face on a daily basis?
Dr. Stanciu: What are some of the challenges you face on a daily basis?
Dr. Penders: There are challenges in multiple areas. First and foremost, medical leadership is responsible for maintaining and improving the quality of patient care and experience. One can expect frequent conflicts to arise when providers vary from established standards or disagree with established policies.
Additionally, there appears to be an increasing lack of a distinct line between administrative and patient care decisions. It is often a challenge to manage the conflicting incentives involved when cost containment and quality care are seen to diverge.
Dr. Stanciu: What are the metrics that measure success by a medical administrator?
Dr. Penders: Some would say that the financial status of the organization is an important metric. Measures such as length of stay, patient satisfaction, and numbers of clinically relevant adverse events are how the success of medical leadership is assessed.
I would argue that patient outcomes as measured by standard clinical tools are the true measure of the success of the efforts of medical providers led by a medical director. Increasingly, measures of population health will likely be used to measure the overall success of health care organizations.
Continue to: How do you keep up-to-date on the latest rules and regulations to ensure facility compliance?
Dr. Stanciu: How do you keep up-to-date on the latest rules and regulations to ensure facility compliance?
Dr. Penders: Medical directors attend many professional meetings, both within their organizations and outside, which assures that information is provided on regulatory initiatives from government bodies and organizations such as the Joint Commission.
Hospital risk managers and attorneys also play a part in keeping everyone honest when it comes to changes in laws governing our work.
Dr. Stanciu: How is it working in a supervisory capacity with other physicians and the growing number of mid-level providers and their expanding scope of practice?
Dr. Penders: There is a variety of opinions today about the relationship between physicians and mid-level providers. Fairly recently, nurse practitioners and physician assistants were known as “extenders.” We don’t hear that term as much anymore, as these providers are becoming increasingly independent in their practice roles.
The supervisory challenge varies with each situation. Most hospital organizations have medical staff rules and regulations that define the relationships within hospitals. Efforts in outpatient care are often less well defined, and supervisory relationships can be tailored to the specific effort involved.
Continue to: Is there a stipend or additional compensation for administrative duties?
Dr. Stanciu: Is there a stipend or additional compensation for administrative duties?
Dr. Penders: Always. There is considerable time and effort needed on a flexibly “as needed” basis that serves as a justification for administrative compensation.
Dr. Stanciu: Any major differences when working in an independent facility vs a large corporation?
Dr. Penders: As health care organizations become larger and more complex, the role of medical directorships in the larger systems are generally defined by policies that can be restrictive. Small organizations may have less formal rules and allow some flexibility for the role of medical leadership in general.
Dr. Stanciu: What preparation do you suggest for trainees and early career psychiatrists who are contemplating such a role?
Dr. Penders: Become involved in quality and organizational initiatives whenever they are available. Generally, organizations will invite and value the input trainees can provide to these efforts. Functioning as a chief resident is real-world experience that can be invaluable.
Editor’s note: Career Choices features a psychiatry resident/fellow interviewing a psychiatrist about why he or she has chosen a specific career path. The goal is to inform trainees about the various psychiatric career options, and to give them a feel for the pros and cons of the various paths.
In this Career Choices, Cornel Stanciu, MD, talked with Thomas Penders, MS, MD. For most of his career, Dr. Penders has practiced in directorship roles. He currently serves as the leader of an addiction consultation service at the Walter B. Jones Center in Greenville, North Carolina, as well as working at the state level with federally qualified health centers to develop collaborative care models.
Dr. Stanciu: What led you to decide to pursue a director role?
Dr. Penders: Early in my career, I was offered opportunities to provide leadership for an organization in its efforts to assure quality and availability of appropriate medical and psychiatric care.
Dr. Stanciu: How has the director role evolved over the years?
Dr. Penders: Thirty years ago, when I got started, hospital administrations depended heavily on medical directors to provide advice on new service initiates. Medical directors were frequently provided with support by health care organizations when recommendations were made based on patient and community need as perceived by medical staff providers. There has been a dramatic shift in the relationship and role of medical directorship, particularly over the past decade. Budgetary constraints have influenced planning and operational decisions to the extent that these decisions are much more likely to be made based on financial analyses rather than on clinical needs identified by physicians. As a result, medical directors are encouraged to be mindful of the effect of their suggestions on the bottom line of the organization. This has resulted in a very significant shift away from programs that are needed but not funded, and toward programs that are revenue-positive or at least neutral.
Medical directors who do not conform in this way are unlikely to be part of the administration for very long in the present environment.
Continue to: What training qualifications are required or desirable to assume a medical leadership role (post residency fellowship, MBA, etc.)?
Dr. Stanciu: What training qualifications are required or desirable to assume a medical leadership role (post-residency fellowship, MBA, etc.)?
Dr. Penders: In addition to a foundation in evidence-based practices and knowledge of regulatory requirements, general leadership skills are probably the most important qualities for medical leadership. Hospitals are complex organizations with confusing reporting relationships. Negotiation skills and communication skills are critical to success. Because most modern health care organizations are well staffed with administrative personnel trained in business and finance, I would not suggest that an MBA is necessary or even important to a medical director’s success. Having said that, there are an increasing number of physicians assuming the role of chief executive officer in complex health care systems. In this case, MBA training will likely be advantageous.
I would suggest that the focus of training that occurs in MPH programs would provide more relevant tools for those in positions of medical leadership. Skills such as biostatistics and epidemiology provide those in such positions with the perspective required to understand the effectiveness of health care systems, and to relate to changes that might be beneficial to the populations they serve. A firm foundation in information systems and data analysis is becoming increasingly important as the payment system moves toward one that is value-based. Increasingly, health care systems decisions will be guided by the analysis of aggregated information gathered from electronic medical records.
Dr. Stanciu: What personal qualities makes a psychiatric physician well-suited for the role of a medical director?
Dr. Penders: Medical directors will confront a variety of difficult situations with colleagues, administrative staff, patients, and family members. A calm demeanor with an ability to reflect rather than react is important. As I previously mentioned, an ability to communicate, including strength as a listener, is another personality trait valued in this position.
Continue to: What are some of the challenges you face on a daily basis?
Dr. Stanciu: What are some of the challenges you face on a daily basis?
Dr. Penders: There are challenges in multiple areas. First and foremost, medical leadership is responsible for maintaining and improving the quality of patient care and experience. One can expect frequent conflicts to arise when providers vary from established standards or disagree with established policies.
Additionally, there appears to be an increasing lack of a distinct line between administrative and patient care decisions. It is often a challenge to manage the conflicting incentives involved when cost containment and quality care are seen to diverge.
Dr. Stanciu: What are the metrics that measure success by a medical administrator?
Dr. Penders: Some would say that the financial status of the organization is an important metric. Measures such as length of stay, patient satisfaction, and numbers of clinically relevant adverse events are how the success of medical leadership is assessed.
I would argue that patient outcomes as measured by standard clinical tools are the true measure of the success of the efforts of medical providers led by a medical director. Increasingly, measures of population health will likely be used to measure the overall success of health care organizations.
Continue to: How do you keep up-to-date on the latest rules and regulations to ensure facility compliance?
Dr. Stanciu: How do you keep up-to-date on the latest rules and regulations to ensure facility compliance?
Dr. Penders: Medical directors attend many professional meetings, both within their organizations and outside, which assures that information is provided on regulatory initiatives from government bodies and organizations such as the Joint Commission.
Hospital risk managers and attorneys also play a part in keeping everyone honest when it comes to changes in laws governing our work.
Dr. Stanciu: How is it working in a supervisory capacity with other physicians and the growing number of mid-level providers and their expanding scope of practice?
Dr. Penders: There is a variety of opinions today about the relationship between physicians and mid-level providers. Fairly recently, nurse practitioners and physician assistants were known as “extenders.” We don’t hear that term as much anymore, as these providers are becoming increasingly independent in their practice roles.
The supervisory challenge varies with each situation. Most hospital organizations have medical staff rules and regulations that define the relationships within hospitals. Efforts in outpatient care are often less well defined, and supervisory relationships can be tailored to the specific effort involved.
Continue to: Is there a stipend or additional compensation for administrative duties?
Dr. Stanciu: Is there a stipend or additional compensation for administrative duties?
Dr. Penders: Always. There is considerable time and effort needed on a flexibly “as needed” basis that serves as a justification for administrative compensation.
Dr. Stanciu: Any major differences when working in an independent facility vs a large corporation?
Dr. Penders: As health care organizations become larger and more complex, the role of medical directorships in the larger systems are generally defined by policies that can be restrictive. Small organizations may have less formal rules and allow some flexibility for the role of medical leadership in general.
Dr. Stanciu: What preparation do you suggest for trainees and early career psychiatrists who are contemplating such a role?
Dr. Penders: Become involved in quality and organizational initiatives whenever they are available. Generally, organizations will invite and value the input trainees can provide to these efforts. Functioning as a chief resident is real-world experience that can be invaluable.
Editor’s note: Career Choices features a psychiatry resident/fellow interviewing a psychiatrist about why he or she has chosen a specific career path. The goal is to inform trainees about the various psychiatric career options, and to give them a feel for the pros and cons of the various paths.
In this Career Choices, Cornel Stanciu, MD, talked with Thomas Penders, MS, MD. For most of his career, Dr. Penders has practiced in directorship roles. He currently serves as the leader of an addiction consultation service at the Walter B. Jones Center in Greenville, North Carolina, as well as working at the state level with federally qualified health centers to develop collaborative care models.
Dr. Stanciu: What led you to decide to pursue a director role?
Dr. Penders: Early in my career, I was offered opportunities to provide leadership for an organization in its efforts to assure quality and availability of appropriate medical and psychiatric care.
Dr. Stanciu: How has the director role evolved over the years?
Dr. Penders: Thirty years ago, when I got started, hospital administrations depended heavily on medical directors to provide advice on new service initiates. Medical directors were frequently provided with support by health care organizations when recommendations were made based on patient and community need as perceived by medical staff providers. There has been a dramatic shift in the relationship and role of medical directorship, particularly over the past decade. Budgetary constraints have influenced planning and operational decisions to the extent that these decisions are much more likely to be made based on financial analyses rather than on clinical needs identified by physicians. As a result, medical directors are encouraged to be mindful of the effect of their suggestions on the bottom line of the organization. This has resulted in a very significant shift away from programs that are needed but not funded, and toward programs that are revenue-positive or at least neutral.
Medical directors who do not conform in this way are unlikely to be part of the administration for very long in the present environment.
Continue to: What training qualifications are required or desirable to assume a medical leadership role (post residency fellowship, MBA, etc.)?
Dr. Stanciu: What training qualifications are required or desirable to assume a medical leadership role (post-residency fellowship, MBA, etc.)?
Dr. Penders: In addition to a foundation in evidence-based practices and knowledge of regulatory requirements, general leadership skills are probably the most important qualities for medical leadership. Hospitals are complex organizations with confusing reporting relationships. Negotiation skills and communication skills are critical to success. Because most modern health care organizations are well staffed with administrative personnel trained in business and finance, I would not suggest that an MBA is necessary or even important to a medical director’s success. Having said that, there are an increasing number of physicians assuming the role of chief executive officer in complex health care systems. In this case, MBA training will likely be advantageous.
I would suggest that the focus of training that occurs in MPH programs would provide more relevant tools for those in positions of medical leadership. Skills such as biostatistics and epidemiology provide those in such positions with the perspective required to understand the effectiveness of health care systems, and to relate to changes that might be beneficial to the populations they serve. A firm foundation in information systems and data analysis is becoming increasingly important as the payment system moves toward one that is value-based. Increasingly, health care systems decisions will be guided by the analysis of aggregated information gathered from electronic medical records.
Dr. Stanciu: What personal qualities makes a psychiatric physician well-suited for the role of a medical director?
Dr. Penders: Medical directors will confront a variety of difficult situations with colleagues, administrative staff, patients, and family members. A calm demeanor with an ability to reflect rather than react is important. As I previously mentioned, an ability to communicate, including strength as a listener, is another personality trait valued in this position.
Continue to: What are some of the challenges you face on a daily basis?
Dr. Stanciu: What are some of the challenges you face on a daily basis?
Dr. Penders: There are challenges in multiple areas. First and foremost, medical leadership is responsible for maintaining and improving the quality of patient care and experience. One can expect frequent conflicts to arise when providers vary from established standards or disagree with established policies.
Additionally, there appears to be an increasing lack of a distinct line between administrative and patient care decisions. It is often a challenge to manage the conflicting incentives involved when cost containment and quality care are seen to diverge.
Dr. Stanciu: What are the metrics that measure success by a medical administrator?
Dr. Penders: Some would say that the financial status of the organization is an important metric. Measures such as length of stay, patient satisfaction, and numbers of clinically relevant adverse events are how the success of medical leadership is assessed.
I would argue that patient outcomes as measured by standard clinical tools are the true measure of the success of the efforts of medical providers led by a medical director. Increasingly, measures of population health will likely be used to measure the overall success of health care organizations.
Continue to: How do you keep up-to-date on the latest rules and regulations to ensure facility compliance?
Dr. Stanciu: How do you keep up-to-date on the latest rules and regulations to ensure facility compliance?
Dr. Penders: Medical directors attend many professional meetings, both within their organizations and outside, which assures that information is provided on regulatory initiatives from government bodies and organizations such as the Joint Commission.
Hospital risk managers and attorneys also play a part in keeping everyone honest when it comes to changes in laws governing our work.
Dr. Stanciu: How is it working in a supervisory capacity with other physicians and the growing number of mid-level providers and their expanding scope of practice?
Dr. Penders: There is a variety of opinions today about the relationship between physicians and mid-level providers. Fairly recently, nurse practitioners and physician assistants were known as “extenders.” We don’t hear that term as much anymore, as these providers are becoming increasingly independent in their practice roles.
The supervisory challenge varies with each situation. Most hospital organizations have medical staff rules and regulations that define the relationships within hospitals. Efforts in outpatient care are often less well defined, and supervisory relationships can be tailored to the specific effort involved.
Continue to: Is there a stipend or additional compensation for administrative duties?
Dr. Stanciu: Is there a stipend or additional compensation for administrative duties?
Dr. Penders: Always. There is considerable time and effort needed on a flexibly “as needed” basis that serves as a justification for administrative compensation.
Dr. Stanciu: Any major differences when working in an independent facility vs a large corporation?
Dr. Penders: As health care organizations become larger and more complex, the role of medical directorships in the larger systems are generally defined by policies that can be restrictive. Small organizations may have less formal rules and allow some flexibility for the role of medical leadership in general.
Dr. Stanciu: What preparation do you suggest for trainees and early career psychiatrists who are contemplating such a role?
Dr. Penders: Become involved in quality and organizational initiatives whenever they are available. Generally, organizations will invite and value the input trainees can provide to these efforts. Functioning as a chief resident is real-world experience that can be invaluable.
‘Robotripping’: What residents need to know
Dextromethorphan (DXM) is commonly found in over-the-counter (OTC) cold and cough preparations. When used at the therapeutic doses DXM has cough-suppressant properties through its action on the medulla. However, OTC preparations containing DXM are being increasingly used recreationally for the drug’s psychoactive effects, a practice referred to as “robotripping.” Such use can result in a toxidrome of delirium with agitation, paranoia, and hallucinations.1 Residents need to be able to recognize the signs of DXM abuse and manage its potentially serious complications.
How DXM works
DXM has a wide therapeutic window. A typical therapeutic dose for cough is up to 120 mg/d. The most common adverse effects are mild (fever, diaphoresis, dizziness, nausea). At higher dosages, it acts as a nonselective serotonin reuptake inhibitor, a sigma-1 receptor agonist, and an N-methyl-
Adverse effects include hallucinations, disorientation, mania, and aggression with delusions of supernatural abilities and insensitivity to pain; these effects are similar to those produced by phencyclidine (PCP).2-4 Physiologically, diaphoresis, hyperthermia, and tachycardia are often observed.3,5 These presentations carry a significant risk of mortality, and appropriate recognition and management is needed.
4 Phases of intoxication
DXM users have described 4 progressive behavioral phases that vary with dosage.3,6,7 First, at 1.5 to 2.5 mg/kg, users report stimulating effects with perceptual alterations similar to those produced by 3,4-methylenedioxymethamphetamine (“ecstasy”). The second phase, reached at 2.5 to 7.5 mg/kg, is similar to alcohol and marijuana intoxication but includes more pronounced dysfunction in motor, cognitive, and perceptual skills, and perhaps visual hallucinations.3,6,7 The third phase, noted at 7.5 to 15 mg/kg, resembles ketamine intoxication, with strong dissociation and hallucinations.3,6,7 At greater doses, out-of-body, trance-like experiences may occur. Delirious misperceptions often lead to violent behavior and limited perception of pain. Users may experience a long course of any of these phases, with presentations lasting for up to 1 to 2 weeks after discontinuing use.8
Management is mainly supportive
Early recognition of DXM use is essential for treatment. Unfortunately, without collateral reports, this can be challenging because specialized toxicology screens are needed to detect DXM. Basic screens sometimes show a false positive for PCP. Take an inventory of all substances in the patient’s possession, either by examining the patient’s belongings or by obtaining collateral information from the patient’s family or friends.
Supportive care should be implemented, with a primary goal of controlling agitation. Short-acting benzodiazepines are helpful. Low-dose, short-term antipsychotics have shown benefit when hallucinations and paranoia are prominent.3 Decreasing stimulation and avoiding physical restraints while attempting to control aggression and psychosis with these medications is recommended. Using physical restraints on an individual who is in a state of agitated delirium can lead to severe injuries, cardiac and respiratory arrest, and death.9-11
Patients typically experience rapid and complete remission of symptoms after discontinuing DXM use. However, evidence suggests DXM users can develop tolerance as well as psychological and physiological dependence. DXM withdrawal can be quite protracted and may include anxiety, dysphoria, insomnia, and suicidality.
1. Stanciu CN, Penders TM, Rouse EM. Recreational use of dextromethorphan,“Robotripping”-A brief review. Am J Addict. 2016;25(5):374-377.
2. Martinak B, Bolis RA, Black JR, et al. Dextromethorphan in cough syrup: The poor man’s psychosis. Psychopharmacol Bull. 2017;47(4):59-63.
3. Logan BK, Yeakel JK, Goldfogel G, et al. Dextromethorphan abuse leading to assault, suicide, or homicide. J Forensic Sci. 2012;57(5):1388-1394.
4. Dextromethorphan (Street names: DXM, CCC, Triple C, Skittles, Robo, Poor Man’s PCP). Drug Enforcement Administration. Office of Diversion Control. Drug & Chemical Evaluation Section. https://www.deadiversion.usdoj.gov/drug_chem_info/dextro_m.pdf. Published March 2014. Accessed April 22, 2018.
5. Reissig CJ, Carter LP, Johnson MW, et al. High doses of dextromethorphan, an NMDA antagonist, produce effects similar to classic hallucinogens. Psychopharmacology (Berl). 2012;223(1):1-15.
6. Boyer EW. Dextromethorphan abuse. Pediatr Emerg Care. 2004;20(12):858-863.
7. Drug Fact Sheet: Dextromethorphan (DXM). Drug Enforcement Administration. https://www.dea.gov/druginfo/drug_data_sheets/Detromethorphan.pdf. Accessed April 22, 2018.
8. Jacob R, Nicholapillai JN. Dextromethorphan induced bipolar disorder. Int Clin Psychopharmacol. 2012;28:e37-e38.
9. Hick JL, Smith SW, Lynch MT. Metabolic acidosis in restraint-associated cardiac arrest: a case series. Acad Emerg Med. 1999;6(3):239-243.
10. Mohr WK, Petti TA, Mohr BD. Adverse effects associated with physical restraint. Can J Psychiatry. 2003;48(5):330-337.
11. Otahbachi M, Cevik C, Bagdure S, et al. Excited delirium, restraints, and unexpected death: a review of pathogenesis. Am J Forensic Med Pathol. 2010;31(2):107-112.
Dextromethorphan (DXM) is commonly found in over-the-counter (OTC) cold and cough preparations. When used at the therapeutic doses DXM has cough-suppressant properties through its action on the medulla. However, OTC preparations containing DXM are being increasingly used recreationally for the drug’s psychoactive effects, a practice referred to as “robotripping.” Such use can result in a toxidrome of delirium with agitation, paranoia, and hallucinations.1 Residents need to be able to recognize the signs of DXM abuse and manage its potentially serious complications.
How DXM works
DXM has a wide therapeutic window. A typical therapeutic dose for cough is up to 120 mg/d. The most common adverse effects are mild (fever, diaphoresis, dizziness, nausea). At higher dosages, it acts as a nonselective serotonin reuptake inhibitor, a sigma-1 receptor agonist, and an N-methyl-
Adverse effects include hallucinations, disorientation, mania, and aggression with delusions of supernatural abilities and insensitivity to pain; these effects are similar to those produced by phencyclidine (PCP).2-4 Physiologically, diaphoresis, hyperthermia, and tachycardia are often observed.3,5 These presentations carry a significant risk of mortality, and appropriate recognition and management is needed.
4 Phases of intoxication
DXM users have described 4 progressive behavioral phases that vary with dosage.3,6,7 First, at 1.5 to 2.5 mg/kg, users report stimulating effects with perceptual alterations similar to those produced by 3,4-methylenedioxymethamphetamine (“ecstasy”). The second phase, reached at 2.5 to 7.5 mg/kg, is similar to alcohol and marijuana intoxication but includes more pronounced dysfunction in motor, cognitive, and perceptual skills, and perhaps visual hallucinations.3,6,7 The third phase, noted at 7.5 to 15 mg/kg, resembles ketamine intoxication, with strong dissociation and hallucinations.3,6,7 At greater doses, out-of-body, trance-like experiences may occur. Delirious misperceptions often lead to violent behavior and limited perception of pain. Users may experience a long course of any of these phases, with presentations lasting for up to 1 to 2 weeks after discontinuing use.8
Management is mainly supportive
Early recognition of DXM use is essential for treatment. Unfortunately, without collateral reports, this can be challenging because specialized toxicology screens are needed to detect DXM. Basic screens sometimes show a false positive for PCP. Take an inventory of all substances in the patient’s possession, either by examining the patient’s belongings or by obtaining collateral information from the patient’s family or friends.
Supportive care should be implemented, with a primary goal of controlling agitation. Short-acting benzodiazepines are helpful. Low-dose, short-term antipsychotics have shown benefit when hallucinations and paranoia are prominent.3 Decreasing stimulation and avoiding physical restraints while attempting to control aggression and psychosis with these medications is recommended. Using physical restraints on an individual who is in a state of agitated delirium can lead to severe injuries, cardiac and respiratory arrest, and death.9-11
Patients typically experience rapid and complete remission of symptoms after discontinuing DXM use. However, evidence suggests DXM users can develop tolerance as well as psychological and physiological dependence. DXM withdrawal can be quite protracted and may include anxiety, dysphoria, insomnia, and suicidality.
Dextromethorphan (DXM) is commonly found in over-the-counter (OTC) cold and cough preparations. When used at the therapeutic doses DXM has cough-suppressant properties through its action on the medulla. However, OTC preparations containing DXM are being increasingly used recreationally for the drug’s psychoactive effects, a practice referred to as “robotripping.” Such use can result in a toxidrome of delirium with agitation, paranoia, and hallucinations.1 Residents need to be able to recognize the signs of DXM abuse and manage its potentially serious complications.
How DXM works
DXM has a wide therapeutic window. A typical therapeutic dose for cough is up to 120 mg/d. The most common adverse effects are mild (fever, diaphoresis, dizziness, nausea). At higher dosages, it acts as a nonselective serotonin reuptake inhibitor, a sigma-1 receptor agonist, and an N-methyl-
Adverse effects include hallucinations, disorientation, mania, and aggression with delusions of supernatural abilities and insensitivity to pain; these effects are similar to those produced by phencyclidine (PCP).2-4 Physiologically, diaphoresis, hyperthermia, and tachycardia are often observed.3,5 These presentations carry a significant risk of mortality, and appropriate recognition and management is needed.
4 Phases of intoxication
DXM users have described 4 progressive behavioral phases that vary with dosage.3,6,7 First, at 1.5 to 2.5 mg/kg, users report stimulating effects with perceptual alterations similar to those produced by 3,4-methylenedioxymethamphetamine (“ecstasy”). The second phase, reached at 2.5 to 7.5 mg/kg, is similar to alcohol and marijuana intoxication but includes more pronounced dysfunction in motor, cognitive, and perceptual skills, and perhaps visual hallucinations.3,6,7 The third phase, noted at 7.5 to 15 mg/kg, resembles ketamine intoxication, with strong dissociation and hallucinations.3,6,7 At greater doses, out-of-body, trance-like experiences may occur. Delirious misperceptions often lead to violent behavior and limited perception of pain. Users may experience a long course of any of these phases, with presentations lasting for up to 1 to 2 weeks after discontinuing use.8
Management is mainly supportive
Early recognition of DXM use is essential for treatment. Unfortunately, without collateral reports, this can be challenging because specialized toxicology screens are needed to detect DXM. Basic screens sometimes show a false positive for PCP. Take an inventory of all substances in the patient’s possession, either by examining the patient’s belongings or by obtaining collateral information from the patient’s family or friends.
Supportive care should be implemented, with a primary goal of controlling agitation. Short-acting benzodiazepines are helpful. Low-dose, short-term antipsychotics have shown benefit when hallucinations and paranoia are prominent.3 Decreasing stimulation and avoiding physical restraints while attempting to control aggression and psychosis with these medications is recommended. Using physical restraints on an individual who is in a state of agitated delirium can lead to severe injuries, cardiac and respiratory arrest, and death.9-11
Patients typically experience rapid and complete remission of symptoms after discontinuing DXM use. However, evidence suggests DXM users can develop tolerance as well as psychological and physiological dependence. DXM withdrawal can be quite protracted and may include anxiety, dysphoria, insomnia, and suicidality.
1. Stanciu CN, Penders TM, Rouse EM. Recreational use of dextromethorphan,“Robotripping”-A brief review. Am J Addict. 2016;25(5):374-377.
2. Martinak B, Bolis RA, Black JR, et al. Dextromethorphan in cough syrup: The poor man’s psychosis. Psychopharmacol Bull. 2017;47(4):59-63.
3. Logan BK, Yeakel JK, Goldfogel G, et al. Dextromethorphan abuse leading to assault, suicide, or homicide. J Forensic Sci. 2012;57(5):1388-1394.
4. Dextromethorphan (Street names: DXM, CCC, Triple C, Skittles, Robo, Poor Man’s PCP). Drug Enforcement Administration. Office of Diversion Control. Drug & Chemical Evaluation Section. https://www.deadiversion.usdoj.gov/drug_chem_info/dextro_m.pdf. Published March 2014. Accessed April 22, 2018.
5. Reissig CJ, Carter LP, Johnson MW, et al. High doses of dextromethorphan, an NMDA antagonist, produce effects similar to classic hallucinogens. Psychopharmacology (Berl). 2012;223(1):1-15.
6. Boyer EW. Dextromethorphan abuse. Pediatr Emerg Care. 2004;20(12):858-863.
7. Drug Fact Sheet: Dextromethorphan (DXM). Drug Enforcement Administration. https://www.dea.gov/druginfo/drug_data_sheets/Detromethorphan.pdf. Accessed April 22, 2018.
8. Jacob R, Nicholapillai JN. Dextromethorphan induced bipolar disorder. Int Clin Psychopharmacol. 2012;28:e37-e38.
9. Hick JL, Smith SW, Lynch MT. Metabolic acidosis in restraint-associated cardiac arrest: a case series. Acad Emerg Med. 1999;6(3):239-243.
10. Mohr WK, Petti TA, Mohr BD. Adverse effects associated with physical restraint. Can J Psychiatry. 2003;48(5):330-337.
11. Otahbachi M, Cevik C, Bagdure S, et al. Excited delirium, restraints, and unexpected death: a review of pathogenesis. Am J Forensic Med Pathol. 2010;31(2):107-112.
1. Stanciu CN, Penders TM, Rouse EM. Recreational use of dextromethorphan,“Robotripping”-A brief review. Am J Addict. 2016;25(5):374-377.
2. Martinak B, Bolis RA, Black JR, et al. Dextromethorphan in cough syrup: The poor man’s psychosis. Psychopharmacol Bull. 2017;47(4):59-63.
3. Logan BK, Yeakel JK, Goldfogel G, et al. Dextromethorphan abuse leading to assault, suicide, or homicide. J Forensic Sci. 2012;57(5):1388-1394.
4. Dextromethorphan (Street names: DXM, CCC, Triple C, Skittles, Robo, Poor Man’s PCP). Drug Enforcement Administration. Office of Diversion Control. Drug & Chemical Evaluation Section. https://www.deadiversion.usdoj.gov/drug_chem_info/dextro_m.pdf. Published March 2014. Accessed April 22, 2018.
5. Reissig CJ, Carter LP, Johnson MW, et al. High doses of dextromethorphan, an NMDA antagonist, produce effects similar to classic hallucinogens. Psychopharmacology (Berl). 2012;223(1):1-15.
6. Boyer EW. Dextromethorphan abuse. Pediatr Emerg Care. 2004;20(12):858-863.
7. Drug Fact Sheet: Dextromethorphan (DXM). Drug Enforcement Administration. https://www.dea.gov/druginfo/drug_data_sheets/Detromethorphan.pdf. Accessed April 22, 2018.
8. Jacob R, Nicholapillai JN. Dextromethorphan induced bipolar disorder. Int Clin Psychopharmacol. 2012;28:e37-e38.
9. Hick JL, Smith SW, Lynch MT. Metabolic acidosis in restraint-associated cardiac arrest: a case series. Acad Emerg Med. 1999;6(3):239-243.
10. Mohr WK, Petti TA, Mohr BD. Adverse effects associated with physical restraint. Can J Psychiatry. 2003;48(5):330-337.
11. Otahbachi M, Cevik C, Bagdure S, et al. Excited delirium, restraints, and unexpected death: a review of pathogenesis. Am J Forensic Med Pathol. 2010;31(2):107-112.
Penile Squamous Cell Carcinoma With Urethral Extension Treated With Mohs Micrographic Surgery
Penile squamous cell carcinoma (SCC) with considerable urethral extension is uncommon and difficult to manage. It often is resistant to less invasive and nonsurgical treatments and frequently results in partial or total penectomy, which can lead to cosmetic disfigurement, functional issues, and psychological distress. We report a case of penile SCC in situ with considerable urethral extension with a focus of cells suspicious for moderately well-differentiated and invasive SCC that was treated with
Mohs micrographic surgery with distal urethrectomy and reconstruction is a valuable treatment technique for cases of SCC involving the glans penis and distal urethra. It offers equivalent or better overall cure rates compared to more radical interventions. Additionally, preservation of the penis with MMS spares patients from considerable physical and psychosocial morbidity. Our case, along with growing body of literature,1-4 calls on dermatologists and urologists to consider MMS as a treatment for penile SCC with or without urethral involvement.
Case Report
A 61-year-old man presented to the dermatology department with a pruritic lesion on the penis that had been present for 6 years. Shave biopsy demonstrated SCC in situ with a focus of cells suspicious for moderately well-differentiated and invasive SCC. Physical examination revealed an ill-defined, 2.2×1.9-cm, pink, eroded plaque involving the tip of the penis and surrounding the external urinary meatus (Figure 1). There was no palpable inguinal lymphadenopathy.
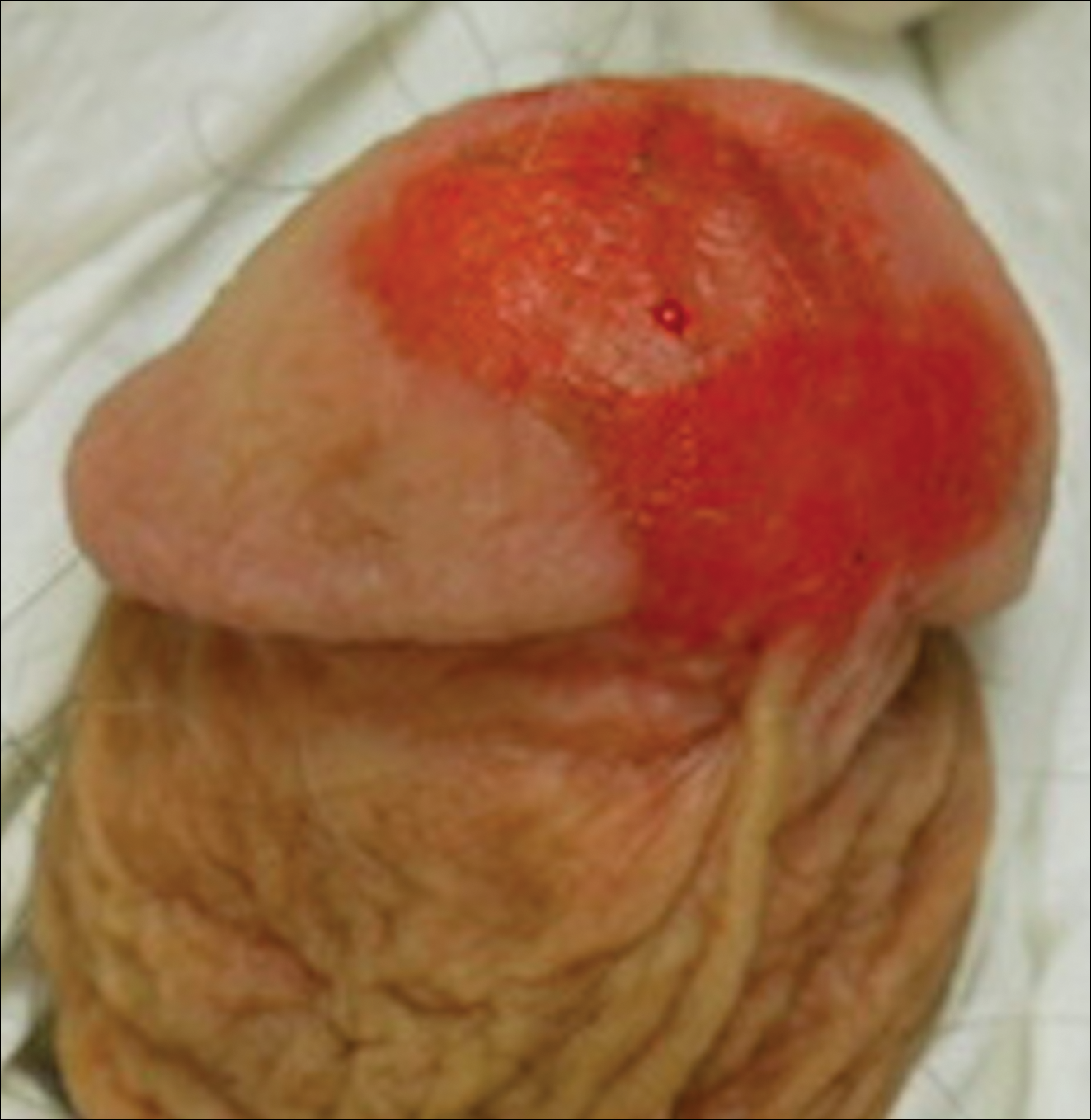
Distal penectomy and lymph node biopsy was recommended following evaluation by the urologic oncology department, but the patient declined these interventions and presented to our dermatology department (A.H.) for a second opinion. The tumor, including the invasive perineural portion, was removed using MMS several weeks after initially presenting to urologic oncology. Ventral meatotomy allowed access to the SCC in situ portion extending proximally up the pendulous urethra (Figure 2). Clear margins were obtained after the eighth stage of MMS, which required removal of 4 to 5 cm of the distal urethra (Figure 3). Reconstruction of the wound required urethral advancement, urethrostomy, and meatoplasty. A positive outcome was achieved with preservation of the length and shape of the penis as well as the cosmetic appearance of the glans penis (Figure 4). The patient was satisfied with the outcome. At 49 months’ follow-up, no evidence of local recurrence or disease progression was noted, and the distal urethrostomy remained intact and functional.
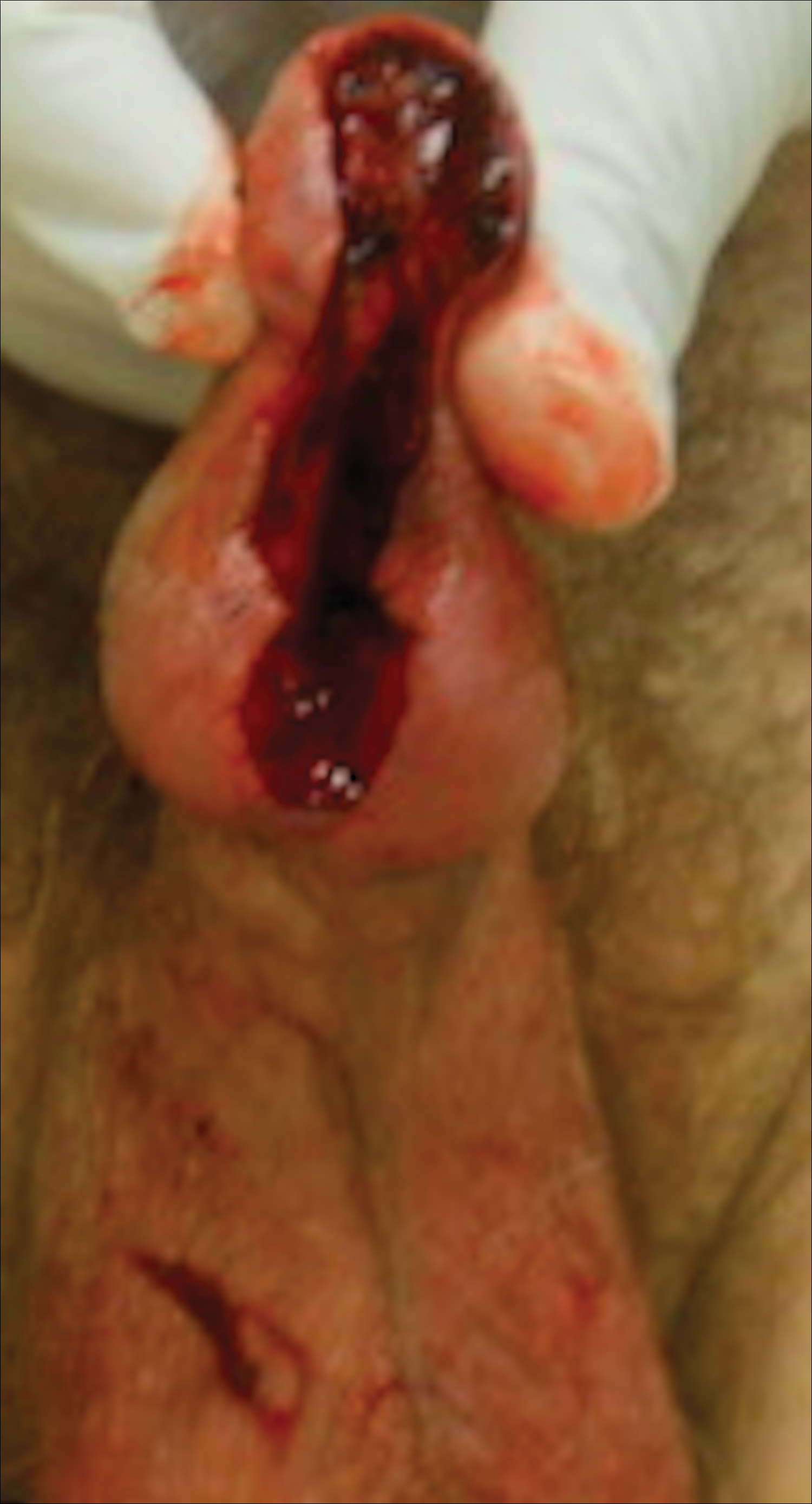


Comment
Penile SCC is a rare malignancy that represents between 0.4% and 0.6% of all malignant tumors in the United States and occurs most commonly in men aged 50 to 70 years.4 The incidence is higher in developing countries, approaching 10% of malignancies in men. It occurs most commonly on the glans penis, prepuce, and coronal sulcus, and has multiple possible appearances, including erythematous and indurated, warty and exophytic, or flat and ulcerated lesions.5 Some reports indicate that more than 40% of penile SCCs are attributable to human papilloma virus,6 while lack of circumcision, chronic inflammation, poor hygiene, balanitis xerotica obliterans, penile trauma, human immunodeficiency virus, UVA treatment of penile psoriasis, and tobacco use are known risk factors.5
Invasive penile SCC generally is treated with penectomy (partial or total), radiation therapy, or MMS; SCC in situ can be treated with topical chemotherapy, laser therapy, and wide local excision (2-cm margins) including circumcision, complete glansectomy, or MMS.5 Squamous cell carcinoma in situ with urethral involvement treated with nonsurgical therapies is associated with higher recurrence rates, ultimately necessitating more aggressive treatments, most commonly partial penectomy.7 The high local recurrence rate of SCC in situ with urethral involvement treated with nonsurgical therapies reflects the fact that determining the presence of urethral extension is difficult and, if present, is inherently inaccessible to these local therapies because the urethra is not an outward-facing tissue surface; MMS represents one possible solution to these issues.
Across all treatment modalities, the most prognostic factor of cancer-specific survival in patients with penile SCC is pelvic lymph node involvement. Some reports cite 5-year survival rates as low as 0% in the setting of pelvic lymph node involvement,5 whereas others had cited rates of 29% to 40%4; 5-year survival rates of higher than 85% have been reported in node-negative patients.4 Recurrence rates vary widely by treatment modality, ranging from less than 10% with partial penectomy and long-term follow-up8 and up to 50% within 2 years with penile-preserving approaches (eg, topical chemotherapy, laser therapy, radiotherapy).5 Multiple case series of penile cancer (the most common of which was SCC/SCC in situ) treated with MMS report comparable and at times superior survival and recurrence data (Table).1-4 Slightly higher recurrences of penile SCC treated with MMS compared to penectomy have been reported, along with considerably higher recurrence rates compared to nonpenile cutaneous SCC treated with MMS (reported to be less than 3%).4 The elastic and expansile nature of penile tissue may lead to distortion from swelling/local anesthesia when taking individual Mohs layers. Additionally, as a large percentage of penile SCCs are attributable to human papillomavirus, difficulty in detecting human papilloma virus–infected cells (which may have oncogenic potential) with the naked eye or histologically with typical staining techniques may help explain the higher recurrence rate of penile SCC treated with MMS compared to penectomy. Despite the higher recurrence rates, survival is comparable or higher in cases treated with MMS (Table).
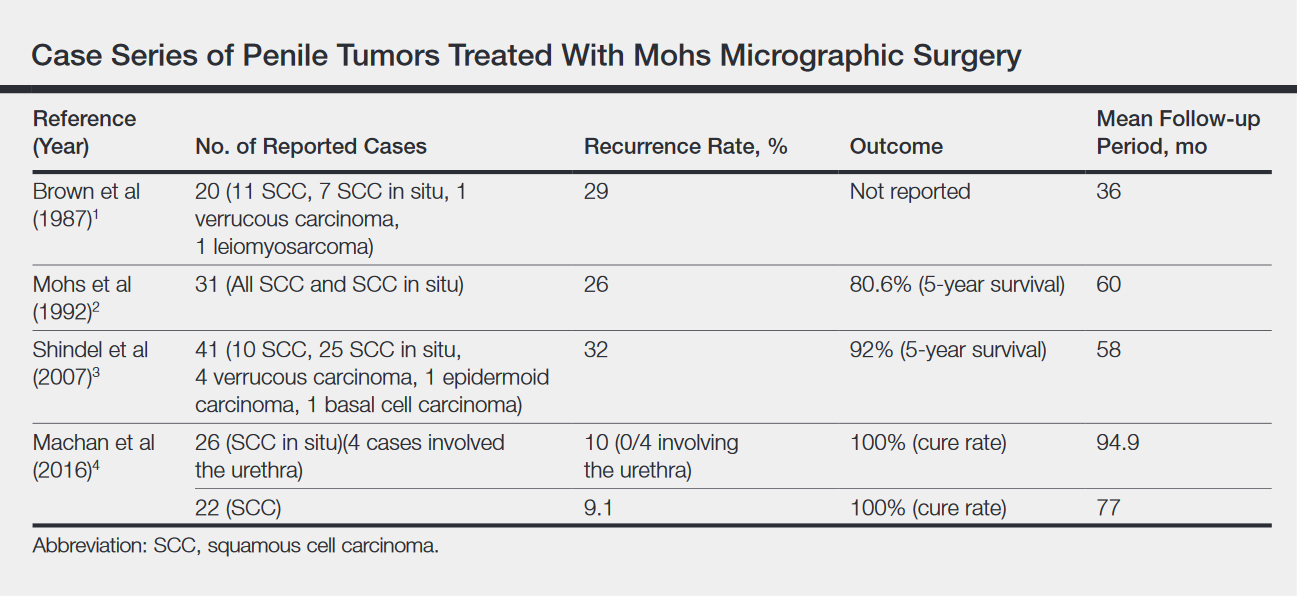
Partial penectomy also has a negative impact on health-related quality of life. Kieffer et al9 compared the impact of penile-sparing surgery (PSS)(including MMS) versus partial or total penectomy on sexual function and health-related quality of life in 90 patients with penile cancer. Although the association between the extent of surgery (partial penectomy/total penectomy/PSS) surgery type and extent and most outcome measures was not statistically significant, partial penectomy was associated with significantly more problems with orgasm (P=.031), concerns about appearance (P=.008), interference in daily life (P=.032), and urinary function (P<.0001) when compared to patients treated with PSS.9 Although this study included only laser/local excision with or without circumcision or glans penis amputation with or without reconstruction as PSSs and did not explicitly include MMS, MMS is clearly a tissue-sparing technique and the study results are generaliz
Conclusion
Penile SCC with considerable urethral extension is uncommon, difficult to manage, and often is resistant to less invasive and nonsurgical treatments. As a result, partial or total penectomy is sometimes necessary. Such cases benefit from MMS with distal urethrectomy and reconstruction because MMS provides equivalent or better overall cure rates compared to more radical interventions.1-4 Importantly, preservation of the penis with MMS can spare patients considerable physical and psychosocial morbidity. Partial penectomy is associated with more health-related quality-of-life problems with orgasm, concerns about appearance, interference in daily life, and urinary function compared to PSSs such as MMS.9 This case, and a growing body of literature, are a call to dermatologists and urologists to consider MMS as a treatment for penile SCC, even with involvement of the urethra.
- Brown MD, Zachary CB, Grekin RC, et al. Penile tumors: their management by Mohs micrographic surgery. J Dermatol Surg Oncol. 1987;13:1163-1167.
- Mohs FE, Snow SN, Larson PO. Mohs micrographic surgery for penile tumors. Urol Clin North Am. 1992;19:291-304.
- Shindel AW, Mann MW, Lev RY, et al. Mohs micrographic surgery for penile cancer: management and long-term followup. J Urol. 2007;178:1980-1985.
- Machan M, Brodland D, Zitelli J. Penile squamous cell carcinoma: penis-preserving treatment with Mohs micrographic surgery. Dermatol Surg. 2016;42:936-944.
- Spiess PE, Horenblas S, Pagliaro LC, et al. Current concepts in penile cancer. J Natl Compr Canc Netw. 2013;11:617-624.
- Hernandez BY, Barnholtz-Sloan J, German RR, et al. Burden of invasive squamous cell carcinoma of the penis in the United States, 1998-2003. Cancer. 2008;113(10 suppl):2883-2891.
- Nash PA, Bihrle R, Gleason PE, et al. Mohs micrographic surgery and distal urethrectomy with immediate urethral reconstruction for glanular carcinoma in situ with significant urethral extension. Urology. 1996;47:108-110.
- Djordjevic ML, Palminteri E, Martins F. Male genital reconstruction for the penile cancer survivor. Curr Opin Urol. 2014;24:427-433.
- Kieffer JM, Djajadiningrat RS, van Muilekom EA, et al. Quality of life for patients treated for penile cancer. J Urol. 2014;192:1105-1110.
Penile squamous cell carcinoma (SCC) with considerable urethral extension is uncommon and difficult to manage. It often is resistant to less invasive and nonsurgical treatments and frequently results in partial or total penectomy, which can lead to cosmetic disfigurement, functional issues, and psychological distress. We report a case of penile SCC in situ with considerable urethral extension with a focus of cells suspicious for moderately well-differentiated and invasive SCC that was treated with
Mohs micrographic surgery with distal urethrectomy and reconstruction is a valuable treatment technique for cases of SCC involving the glans penis and distal urethra. It offers equivalent or better overall cure rates compared to more radical interventions. Additionally, preservation of the penis with MMS spares patients from considerable physical and psychosocial morbidity. Our case, along with growing body of literature,1-4 calls on dermatologists and urologists to consider MMS as a treatment for penile SCC with or without urethral involvement.
Case Report
A 61-year-old man presented to the dermatology department with a pruritic lesion on the penis that had been present for 6 years. Shave biopsy demonstrated SCC in situ with a focus of cells suspicious for moderately well-differentiated and invasive SCC. Physical examination revealed an ill-defined, 2.2×1.9-cm, pink, eroded plaque involving the tip of the penis and surrounding the external urinary meatus (Figure 1). There was no palpable inguinal lymphadenopathy.

Distal penectomy and lymph node biopsy was recommended following evaluation by the urologic oncology department, but the patient declined these interventions and presented to our dermatology department (A.H.) for a second opinion. The tumor, including the invasive perineural portion, was removed using MMS several weeks after initially presenting to urologic oncology. Ventral meatotomy allowed access to the SCC in situ portion extending proximally up the pendulous urethra (Figure 2). Clear margins were obtained after the eighth stage of MMS, which required removal of 4 to 5 cm of the distal urethra (Figure 3). Reconstruction of the wound required urethral advancement, urethrostomy, and meatoplasty. A positive outcome was achieved with preservation of the length and shape of the penis as well as the cosmetic appearance of the glans penis (Figure 4). The patient was satisfied with the outcome. At 49 months’ follow-up, no evidence of local recurrence or disease progression was noted, and the distal urethrostomy remained intact and functional.



Comment
Penile SCC is a rare malignancy that represents between 0.4% and 0.6% of all malignant tumors in the United States and occurs most commonly in men aged 50 to 70 years.4 The incidence is higher in developing countries, approaching 10% of malignancies in men. It occurs most commonly on the glans penis, prepuce, and coronal sulcus, and has multiple possible appearances, including erythematous and indurated, warty and exophytic, or flat and ulcerated lesions.5 Some reports indicate that more than 40% of penile SCCs are attributable to human papilloma virus,6 while lack of circumcision, chronic inflammation, poor hygiene, balanitis xerotica obliterans, penile trauma, human immunodeficiency virus, UVA treatment of penile psoriasis, and tobacco use are known risk factors.5
Invasive penile SCC generally is treated with penectomy (partial or total), radiation therapy, or MMS; SCC in situ can be treated with topical chemotherapy, laser therapy, and wide local excision (2-cm margins) including circumcision, complete glansectomy, or MMS.5 Squamous cell carcinoma in situ with urethral involvement treated with nonsurgical therapies is associated with higher recurrence rates, ultimately necessitating more aggressive treatments, most commonly partial penectomy.7 The high local recurrence rate of SCC in situ with urethral involvement treated with nonsurgical therapies reflects the fact that determining the presence of urethral extension is difficult and, if present, is inherently inaccessible to these local therapies because the urethra is not an outward-facing tissue surface; MMS represents one possible solution to these issues.
Across all treatment modalities, the most prognostic factor of cancer-specific survival in patients with penile SCC is pelvic lymph node involvement. Some reports cite 5-year survival rates as low as 0% in the setting of pelvic lymph node involvement,5 whereas others had cited rates of 29% to 40%4; 5-year survival rates of higher than 85% have been reported in node-negative patients.4 Recurrence rates vary widely by treatment modality, ranging from less than 10% with partial penectomy and long-term follow-up8 and up to 50% within 2 years with penile-preserving approaches (eg, topical chemotherapy, laser therapy, radiotherapy).5 Multiple case series of penile cancer (the most common of which was SCC/SCC in situ) treated with MMS report comparable and at times superior survival and recurrence data (Table).1-4 Slightly higher recurrences of penile SCC treated with MMS compared to penectomy have been reported, along with considerably higher recurrence rates compared to nonpenile cutaneous SCC treated with MMS (reported to be less than 3%).4 The elastic and expansile nature of penile tissue may lead to distortion from swelling/local anesthesia when taking individual Mohs layers. Additionally, as a large percentage of penile SCCs are attributable to human papillomavirus, difficulty in detecting human papilloma virus–infected cells (which may have oncogenic potential) with the naked eye or histologically with typical staining techniques may help explain the higher recurrence rate of penile SCC treated with MMS compared to penectomy. Despite the higher recurrence rates, survival is comparable or higher in cases treated with MMS (Table).

Partial penectomy also has a negative impact on health-related quality of life. Kieffer et al9 compared the impact of penile-sparing surgery (PSS)(including MMS) versus partial or total penectomy on sexual function and health-related quality of life in 90 patients with penile cancer. Although the association between the extent of surgery (partial penectomy/total penectomy/PSS) surgery type and extent and most outcome measures was not statistically significant, partial penectomy was associated with significantly more problems with orgasm (P=.031), concerns about appearance (P=.008), interference in daily life (P=.032), and urinary function (P<.0001) when compared to patients treated with PSS.9 Although this study included only laser/local excision with or without circumcision or glans penis amputation with or without reconstruction as PSSs and did not explicitly include MMS, MMS is clearly a tissue-sparing technique and the study results are generaliz
Conclusion
Penile SCC with considerable urethral extension is uncommon, difficult to manage, and often is resistant to less invasive and nonsurgical treatments. As a result, partial or total penectomy is sometimes necessary. Such cases benefit from MMS with distal urethrectomy and reconstruction because MMS provides equivalent or better overall cure rates compared to more radical interventions.1-4 Importantly, preservation of the penis with MMS can spare patients considerable physical and psychosocial morbidity. Partial penectomy is associated with more health-related quality-of-life problems with orgasm, concerns about appearance, interference in daily life, and urinary function compared to PSSs such as MMS.9 This case, and a growing body of literature, are a call to dermatologists and urologists to consider MMS as a treatment for penile SCC, even with involvement of the urethra.
Penile squamous cell carcinoma (SCC) with considerable urethral extension is uncommon and difficult to manage. It often is resistant to less invasive and nonsurgical treatments and frequently results in partial or total penectomy, which can lead to cosmetic disfigurement, functional issues, and psychological distress. We report a case of penile SCC in situ with considerable urethral extension with a focus of cells suspicious for moderately well-differentiated and invasive SCC that was treated with
Mohs micrographic surgery with distal urethrectomy and reconstruction is a valuable treatment technique for cases of SCC involving the glans penis and distal urethra. It offers equivalent or better overall cure rates compared to more radical interventions. Additionally, preservation of the penis with MMS spares patients from considerable physical and psychosocial morbidity. Our case, along with growing body of literature,1-4 calls on dermatologists and urologists to consider MMS as a treatment for penile SCC with or without urethral involvement.
Case Report
A 61-year-old man presented to the dermatology department with a pruritic lesion on the penis that had been present for 6 years. Shave biopsy demonstrated SCC in situ with a focus of cells suspicious for moderately well-differentiated and invasive SCC. Physical examination revealed an ill-defined, 2.2×1.9-cm, pink, eroded plaque involving the tip of the penis and surrounding the external urinary meatus (Figure 1). There was no palpable inguinal lymphadenopathy.

Distal penectomy and lymph node biopsy was recommended following evaluation by the urologic oncology department, but the patient declined these interventions and presented to our dermatology department (A.H.) for a second opinion. The tumor, including the invasive perineural portion, was removed using MMS several weeks after initially presenting to urologic oncology. Ventral meatotomy allowed access to the SCC in situ portion extending proximally up the pendulous urethra (Figure 2). Clear margins were obtained after the eighth stage of MMS, which required removal of 4 to 5 cm of the distal urethra (Figure 3). Reconstruction of the wound required urethral advancement, urethrostomy, and meatoplasty. A positive outcome was achieved with preservation of the length and shape of the penis as well as the cosmetic appearance of the glans penis (Figure 4). The patient was satisfied with the outcome. At 49 months’ follow-up, no evidence of local recurrence or disease progression was noted, and the distal urethrostomy remained intact and functional.



Comment
Penile SCC is a rare malignancy that represents between 0.4% and 0.6% of all malignant tumors in the United States and occurs most commonly in men aged 50 to 70 years.4 The incidence is higher in developing countries, approaching 10% of malignancies in men. It occurs most commonly on the glans penis, prepuce, and coronal sulcus, and has multiple possible appearances, including erythematous and indurated, warty and exophytic, or flat and ulcerated lesions.5 Some reports indicate that more than 40% of penile SCCs are attributable to human papilloma virus,6 while lack of circumcision, chronic inflammation, poor hygiene, balanitis xerotica obliterans, penile trauma, human immunodeficiency virus, UVA treatment of penile psoriasis, and tobacco use are known risk factors.5
Invasive penile SCC generally is treated with penectomy (partial or total), radiation therapy, or MMS; SCC in situ can be treated with topical chemotherapy, laser therapy, and wide local excision (2-cm margins) including circumcision, complete glansectomy, or MMS.5 Squamous cell carcinoma in situ with urethral involvement treated with nonsurgical therapies is associated with higher recurrence rates, ultimately necessitating more aggressive treatments, most commonly partial penectomy.7 The high local recurrence rate of SCC in situ with urethral involvement treated with nonsurgical therapies reflects the fact that determining the presence of urethral extension is difficult and, if present, is inherently inaccessible to these local therapies because the urethra is not an outward-facing tissue surface; MMS represents one possible solution to these issues.
Across all treatment modalities, the most prognostic factor of cancer-specific survival in patients with penile SCC is pelvic lymph node involvement. Some reports cite 5-year survival rates as low as 0% in the setting of pelvic lymph node involvement,5 whereas others had cited rates of 29% to 40%4; 5-year survival rates of higher than 85% have been reported in node-negative patients.4 Recurrence rates vary widely by treatment modality, ranging from less than 10% with partial penectomy and long-term follow-up8 and up to 50% within 2 years with penile-preserving approaches (eg, topical chemotherapy, laser therapy, radiotherapy).5 Multiple case series of penile cancer (the most common of which was SCC/SCC in situ) treated with MMS report comparable and at times superior survival and recurrence data (Table).1-4 Slightly higher recurrences of penile SCC treated with MMS compared to penectomy have been reported, along with considerably higher recurrence rates compared to nonpenile cutaneous SCC treated with MMS (reported to be less than 3%).4 The elastic and expansile nature of penile tissue may lead to distortion from swelling/local anesthesia when taking individual Mohs layers. Additionally, as a large percentage of penile SCCs are attributable to human papillomavirus, difficulty in detecting human papilloma virus–infected cells (which may have oncogenic potential) with the naked eye or histologically with typical staining techniques may help explain the higher recurrence rate of penile SCC treated with MMS compared to penectomy. Despite the higher recurrence rates, survival is comparable or higher in cases treated with MMS (Table).

Partial penectomy also has a negative impact on health-related quality of life. Kieffer et al9 compared the impact of penile-sparing surgery (PSS)(including MMS) versus partial or total penectomy on sexual function and health-related quality of life in 90 patients with penile cancer. Although the association between the extent of surgery (partial penectomy/total penectomy/PSS) surgery type and extent and most outcome measures was not statistically significant, partial penectomy was associated with significantly more problems with orgasm (P=.031), concerns about appearance (P=.008), interference in daily life (P=.032), and urinary function (P<.0001) when compared to patients treated with PSS.9 Although this study included only laser/local excision with or without circumcision or glans penis amputation with or without reconstruction as PSSs and did not explicitly include MMS, MMS is clearly a tissue-sparing technique and the study results are generaliz
Conclusion
Penile SCC with considerable urethral extension is uncommon, difficult to manage, and often is resistant to less invasive and nonsurgical treatments. As a result, partial or total penectomy is sometimes necessary. Such cases benefit from MMS with distal urethrectomy and reconstruction because MMS provides equivalent or better overall cure rates compared to more radical interventions.1-4 Importantly, preservation of the penis with MMS can spare patients considerable physical and psychosocial morbidity. Partial penectomy is associated with more health-related quality-of-life problems with orgasm, concerns about appearance, interference in daily life, and urinary function compared to PSSs such as MMS.9 This case, and a growing body of literature, are a call to dermatologists and urologists to consider MMS as a treatment for penile SCC, even with involvement of the urethra.
- Brown MD, Zachary CB, Grekin RC, et al. Penile tumors: their management by Mohs micrographic surgery. J Dermatol Surg Oncol. 1987;13:1163-1167.
- Mohs FE, Snow SN, Larson PO. Mohs micrographic surgery for penile tumors. Urol Clin North Am. 1992;19:291-304.
- Shindel AW, Mann MW, Lev RY, et al. Mohs micrographic surgery for penile cancer: management and long-term followup. J Urol. 2007;178:1980-1985.
- Machan M, Brodland D, Zitelli J. Penile squamous cell carcinoma: penis-preserving treatment with Mohs micrographic surgery. Dermatol Surg. 2016;42:936-944.
- Spiess PE, Horenblas S, Pagliaro LC, et al. Current concepts in penile cancer. J Natl Compr Canc Netw. 2013;11:617-624.
- Hernandez BY, Barnholtz-Sloan J, German RR, et al. Burden of invasive squamous cell carcinoma of the penis in the United States, 1998-2003. Cancer. 2008;113(10 suppl):2883-2891.
- Nash PA, Bihrle R, Gleason PE, et al. Mohs micrographic surgery and distal urethrectomy with immediate urethral reconstruction for glanular carcinoma in situ with significant urethral extension. Urology. 1996;47:108-110.
- Djordjevic ML, Palminteri E, Martins F. Male genital reconstruction for the penile cancer survivor. Curr Opin Urol. 2014;24:427-433.
- Kieffer JM, Djajadiningrat RS, van Muilekom EA, et al. Quality of life for patients treated for penile cancer. J Urol. 2014;192:1105-1110.
- Brown MD, Zachary CB, Grekin RC, et al. Penile tumors: their management by Mohs micrographic surgery. J Dermatol Surg Oncol. 1987;13:1163-1167.
- Mohs FE, Snow SN, Larson PO. Mohs micrographic surgery for penile tumors. Urol Clin North Am. 1992;19:291-304.
- Shindel AW, Mann MW, Lev RY, et al. Mohs micrographic surgery for penile cancer: management and long-term followup. J Urol. 2007;178:1980-1985.
- Machan M, Brodland D, Zitelli J. Penile squamous cell carcinoma: penis-preserving treatment with Mohs micrographic surgery. Dermatol Surg. 2016;42:936-944.
- Spiess PE, Horenblas S, Pagliaro LC, et al. Current concepts in penile cancer. J Natl Compr Canc Netw. 2013;11:617-624.
- Hernandez BY, Barnholtz-Sloan J, German RR, et al. Burden of invasive squamous cell carcinoma of the penis in the United States, 1998-2003. Cancer. 2008;113(10 suppl):2883-2891.
- Nash PA, Bihrle R, Gleason PE, et al. Mohs micrographic surgery and distal urethrectomy with immediate urethral reconstruction for glanular carcinoma in situ with significant urethral extension. Urology. 1996;47:108-110.
- Djordjevic ML, Palminteri E, Martins F. Male genital reconstruction for the penile cancer survivor. Curr Opin Urol. 2014;24:427-433.
- Kieffer JM, Djajadiningrat RS, van Muilekom EA, et al. Quality of life for patients treated for penile cancer. J Urol. 2014;192:1105-1110.
Resident Pearl
- Penile squamous cell carcinoma (SCC) often is treated with partial or total penectomy, especially when there is urethral extension. Mohs micrographic surgery for penile SCC results in equivalent or better overall cure rates and decreases morbidity.
Climate Change and Skin Disease
The term climate refers to the average weather conditions of a specific geographic location measured over several decades.1 While a certain degree of variation in the Earth’s climate is expected, a persistent warming or cooling trend is not. The factors driving the Earth’s warming remain difficult to prove.2 We know the Earth previously has undergone dramatic climate changes and that natural factors driving these changes are varied (eg, the relationship between the Earth and the Sun, volcanic eruptions, solar irradiance).1,3 These factors ideally change over protracted periods of time in a way that allows organisms to adapt to new environments.
Anthropogenic climate change refers to human-caused climate change. This is thought to be a major driving factor in the Earth’s recent warming trend, partly due to the rapidity of warming in recent years.3 According to climate scientists, the Earth’s temperature has risen 4°C to 7°C over the past 5000 years, but it has risen 0.7°C in just the past 100 years alone.4 Greenhouse gases such as carbon dioxide are emitted by various natural processes and human activities and play a central role in current warming because they trap solar heat and increase ambient temperature.3
In a recent edition of the commonly cited textbook Dermatology, Bolognia et al5 referenced climate change only once in a figure legend regarding the expansion of dengue fever in the Americas. However, climate change may have the potential to cause outright skin disease epidemics worldwide, and the Climate Change Committee of the International Society of Dermatology has called upon dermatologists across the globe to help raise awareness of this issue.6
Much of the literature regarding the effects of climate change on human health focuses on insect-borne diseases, but over the past decade other areas of impact also have been investigated, such as increases in airborne diseases, zoonoses, newly endemic saprophytic and dimorphic fungal infections, fecal-oral diseases, and severe allergic disease.7,8 It is postulated that climate change leads to region-specific increases in human disease because it creates newly favorable habitats for infectious agents, their vectors. and their reservoirs, allowing expansion of their ranges and access to immunologically naïve populations.9 Furthermore, extreme weather events such as heat waves, hurricanes, and flooding, which are expected to increase in frequency as a result of climate change, have all been linked to infectious disease outbreaks.10
Lyme Disease
In the past 20 years, Lyme disease incidence has tripled in the United States.11 It has been hypothesized that the increase may be occurring as a result of the expanding geographic distribution of the Ixodes tick and its mammalian hosts (eg, white-tailed deer) under the influence of climate change.12 Lyme disease is a multisystem disease affecting the skin, joints, heart, and nervous system. Its most characteristic manifestation is cutaneous in the form of erythema migrans. Dermatologists may be called upon to play an increasingly important role in early detection and treatment of this potentially chronic and debilitating condition.
Arboviruses
Arboviruses are transmitted by arthropods and are an important category of climate change–related diseases due to the expansion of the mosquito habitat worldwide. The vectorial capacity for the transmission of dengue fever has increased worldwide by 9.4% via Aedes aegypti and 11.1% via Aedes albopictus since 1950.13 Dengue fever, also known as breakbone fever, presents with intense joint pain, fevers, headaches, and a transient morbilliform rash that desquamates with defervescence and in some cases will incite hemorrhagic skin lesions.14 Dengue fever previously was considered to be a tropical disease but locally acquired cases have been reported in the United States, including Texas, Hawaii, and Florida.15,16
Reports of local transmission of chikungunya, another arbovirus transmitted by A albopictus and A aegypti mosquitoes in Florida, the US Virgin Islands, and Puerto Rico, began in 2014.17 A higher prevalence of these diseases within the United States also may be related to increased globalization, with US travelers returning from endemic regions with infections. Prior to 2014, transmission occurred in traditional endemic regions, primarily in Asia, Africa, or island nations in the Indian Ocean. Like dengue fever, chikungunya causes high fevers, cutaneous manifestations (eg, urticarial papules, morbilliform eruption, hypermelanosis, intertriginous lesions, lymphedema),17 and intense joint pain. Unlike dengue fever, however, joint involvement can be chronic, erosive, and debilitating.
Lastly, New World leishmaniasis, an arboviral disease characterized by mucocutaneous ulcers and transmitted by phlebotomine sand flies, has been acquired locally in Oklahoma and Texas when it was previously considered to be endemic to Mexico and Central and South America.14,18 The habitats of New World Leishmania species are expected to expand northward, with an ecological niche model predicting that they reach southern Canada by the year 2080 due to the expanding habitats of sand fly and rodent vectors.19
Fungal Infections
In the Pacific Northwest, there have been reports of newly endemic Cryptococcus gattii and Coccidioides immitis, both of which previously had been confined to the southwestern United States.8 Endemic ranges of these mycotic pathogens may be expanding for a variety of reasons, with climate change creating new regions conducive to the colonization of these species.8,20,21Coccidioides immitis is a soil-dwelling fungus that usually presents with primary pulmonary disease that can disseminate acutely or even months later. Prompt recognition of disseminated disease may allow life-saving therapy to be initiated. Cryptococcus gattii is a fungus with multiple niches, including oil, trees, and birds.20 This fungus also is acquired via inhalation, with dissemination occurring most commonly in immunosuppressed patients to the central nervous system, bone, and skin. Primary or secondary infection with both of these fungi may present with cutaneous manifestations presenting as polymorphous lesions, including umbilicated or ulcerated papules, indurated nodules, and acneiform pustules.
Final Thoughts
Awareness of the shifting habitats of microorganisms and vectors locally is important in order for clinicians to make correct diagnoses in a timely fashion. Regional or endemic diseases are presenting outside their traditional boundaries due to changing habitats of microbes and vectors and may be easily overlooked, resulting in a delayed diagnosis. Being prepared to diagnose diseases with increasing incidence secondary to climate change and discussing this with patients is an important physician obligation, but it is not the only one. We cannot effectively advocate for the health of patients and the community while ignoring the destruction of the environment. Our additional responsibility is straightforward—being advocates for good stewardship of the Earth’s resources now on both a personal and a policy level.22
- Climate Central. Global Weirdness: Severe Storms, Deadly Heat Waves, Relentless Drought, Rising Seas, and the Weather of the Future. New York, NY: Pantheon Books; 2012.
- Cook J, Nuccitelli D, Green SA, et al. Quantifying the consensus on anthropogenic global warming in the scientific literature. Environ Res Lett. 2013;8:024024.
- A blanket around the Earth. NASA Climate website. https://climate.nasa.gov/causes/. Accessed February 5, 2018.
- How is today’s warming different from the past? NASA Earth Observatory website. https://earthobservatory.nasa.gov/Features/GlobalWarming/page3.php. Accessed February 5, 2018.
- Mancini AJ, Shani-Adir A, Sidbury R. Other viral diseases. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Philadelphia, PA: Elsevier; 2017:1425-1446.
- Andersen LK, Davis MDP. A wake-up call to dermatologists—climate change affects the skin. Int J Dermatol. 2017;56:E198-E199.
- Liang L, Gong P. Climate change and human infectious diseases: a synthesis of research findings from global and spatio-temporal perspectives [published online March 23, 2017]. Environ Int. 2017;103:99-108.
- Lockhart SR, McCotter OZ, Chiller TM. Emerging fungal infections in the Pacific Northwest: the unrecognized burden and geographic range of Cryptococcus gattii and Coccidioides immitis. Microbiol Spectr. 2016;4. doi:10.1128/microbiolspec.EI10-0016-2016.
- Kilpatrick AM, Randolph SE. Drivers, dynamics, and control of emerging vector-borne zoonotic diseases. Lancet. 2012;380:1946-1955.
- McMichael AJ. Extreme weather events and infectious disease outbreaks. Virulence. 2015;6:543-547.
- Lyme disease graphs. CDC website. https://www.cdc.gov/lyme/stats/graphs.html. Updated November 1, 2017. Accessed April 12, 2018.
- Stone BL, Tourand Y, Brissette CA. Brave new worlds: the expanding universe of Lyme disease. Vector Borne Zoonotic Dis. 2017;17:619-629.
- Watts N, Amann M, Ayeb-Karlsson S, et al. The Lancet Countdown on health and climate change: from 25 years of inaction to a global transformation for public health [published online October 30, 2017]. Lancet. doi:10.1016/S0140-6736(17)32464-9.
- Nawas ZY, Tong Y, Kollipara R, et al. Emerging infectious diseases with cutaneous manifestations: viral and bacterial infections. J Am Acad Dermatol. 2016;75:1-16.
- Kaffenberger BH, Shetlar D, Norton SA, et al. The effect of climate change on skin disease in North America. J Am Acad Dermatol. 2017;76:140-147.
- Dengue. CDC website. https://www.cdc.gov/dengue/epidemiology/index.html. Updated June 9, 2014. Accessed April 3, 2018.
- Chikungunya virus in the United States. CDC website. https://www.cdc.gov/chikungunya/geo/united-states.html. Updated October 30, 2017. Accessed April 4, 2018.
- Clarke CF, Bradley KK, Wright JH, et al. Emergence of autochthonous cutaneous leishmaniasis in northeastern Texas and southeastern Oklahoma. Am J Trop Med Hyg. 2013;88:157-61.
- González C, Wang O, Strutz SE, et al. Climate change and risk of leishmaniasis in North America: predictions from ecological niche models of vector and reservoir species. PLoS Negl Trop Dis. 2010;4:E585.
- Chang CC, Chen SC. Colliding epidemics and the rise of cryptococcosis. J Fungi (Basel). 2015;2. doi: 10.3390/jof2010001.
- Marsden-Haug N, Goldoft M, Ralston C, et al. Coccidioidomycosis acquired in Washington state. Clin Infect Dis. 2013;56:847-850.
- Rosenbach M. Climate change & dermatology: what can you do? Paper presented at: American Academy of Dermatology Annual Meeting; March 3-7, 2017; Orlando, FL.
The term climate refers to the average weather conditions of a specific geographic location measured over several decades.1 While a certain degree of variation in the Earth’s climate is expected, a persistent warming or cooling trend is not. The factors driving the Earth’s warming remain difficult to prove.2 We know the Earth previously has undergone dramatic climate changes and that natural factors driving these changes are varied (eg, the relationship between the Earth and the Sun, volcanic eruptions, solar irradiance).1,3 These factors ideally change over protracted periods of time in a way that allows organisms to adapt to new environments.
Anthropogenic climate change refers to human-caused climate change. This is thought to be a major driving factor in the Earth’s recent warming trend, partly due to the rapidity of warming in recent years.3 According to climate scientists, the Earth’s temperature has risen 4°C to 7°C over the past 5000 years, but it has risen 0.7°C in just the past 100 years alone.4 Greenhouse gases such as carbon dioxide are emitted by various natural processes and human activities and play a central role in current warming because they trap solar heat and increase ambient temperature.3
In a recent edition of the commonly cited textbook Dermatology, Bolognia et al5 referenced climate change only once in a figure legend regarding the expansion of dengue fever in the Americas. However, climate change may have the potential to cause outright skin disease epidemics worldwide, and the Climate Change Committee of the International Society of Dermatology has called upon dermatologists across the globe to help raise awareness of this issue.6
Much of the literature regarding the effects of climate change on human health focuses on insect-borne diseases, but over the past decade other areas of impact also have been investigated, such as increases in airborne diseases, zoonoses, newly endemic saprophytic and dimorphic fungal infections, fecal-oral diseases, and severe allergic disease.7,8 It is postulated that climate change leads to region-specific increases in human disease because it creates newly favorable habitats for infectious agents, their vectors. and their reservoirs, allowing expansion of their ranges and access to immunologically naïve populations.9 Furthermore, extreme weather events such as heat waves, hurricanes, and flooding, which are expected to increase in frequency as a result of climate change, have all been linked to infectious disease outbreaks.10
Lyme Disease
In the past 20 years, Lyme disease incidence has tripled in the United States.11 It has been hypothesized that the increase may be occurring as a result of the expanding geographic distribution of the Ixodes tick and its mammalian hosts (eg, white-tailed deer) under the influence of climate change.12 Lyme disease is a multisystem disease affecting the skin, joints, heart, and nervous system. Its most characteristic manifestation is cutaneous in the form of erythema migrans. Dermatologists may be called upon to play an increasingly important role in early detection and treatment of this potentially chronic and debilitating condition.
Arboviruses
Arboviruses are transmitted by arthropods and are an important category of climate change–related diseases due to the expansion of the mosquito habitat worldwide. The vectorial capacity for the transmission of dengue fever has increased worldwide by 9.4% via Aedes aegypti and 11.1% via Aedes albopictus since 1950.13 Dengue fever, also known as breakbone fever, presents with intense joint pain, fevers, headaches, and a transient morbilliform rash that desquamates with defervescence and in some cases will incite hemorrhagic skin lesions.14 Dengue fever previously was considered to be a tropical disease but locally acquired cases have been reported in the United States, including Texas, Hawaii, and Florida.15,16
Reports of local transmission of chikungunya, another arbovirus transmitted by A albopictus and A aegypti mosquitoes in Florida, the US Virgin Islands, and Puerto Rico, began in 2014.17 A higher prevalence of these diseases within the United States also may be related to increased globalization, with US travelers returning from endemic regions with infections. Prior to 2014, transmission occurred in traditional endemic regions, primarily in Asia, Africa, or island nations in the Indian Ocean. Like dengue fever, chikungunya causes high fevers, cutaneous manifestations (eg, urticarial papules, morbilliform eruption, hypermelanosis, intertriginous lesions, lymphedema),17 and intense joint pain. Unlike dengue fever, however, joint involvement can be chronic, erosive, and debilitating.
Lastly, New World leishmaniasis, an arboviral disease characterized by mucocutaneous ulcers and transmitted by phlebotomine sand flies, has been acquired locally in Oklahoma and Texas when it was previously considered to be endemic to Mexico and Central and South America.14,18 The habitats of New World Leishmania species are expected to expand northward, with an ecological niche model predicting that they reach southern Canada by the year 2080 due to the expanding habitats of sand fly and rodent vectors.19
Fungal Infections
In the Pacific Northwest, there have been reports of newly endemic Cryptococcus gattii and Coccidioides immitis, both of which previously had been confined to the southwestern United States.8 Endemic ranges of these mycotic pathogens may be expanding for a variety of reasons, with climate change creating new regions conducive to the colonization of these species.8,20,21Coccidioides immitis is a soil-dwelling fungus that usually presents with primary pulmonary disease that can disseminate acutely or even months later. Prompt recognition of disseminated disease may allow life-saving therapy to be initiated. Cryptococcus gattii is a fungus with multiple niches, including oil, trees, and birds.20 This fungus also is acquired via inhalation, with dissemination occurring most commonly in immunosuppressed patients to the central nervous system, bone, and skin. Primary or secondary infection with both of these fungi may present with cutaneous manifestations presenting as polymorphous lesions, including umbilicated or ulcerated papules, indurated nodules, and acneiform pustules.
Final Thoughts
Awareness of the shifting habitats of microorganisms and vectors locally is important in order for clinicians to make correct diagnoses in a timely fashion. Regional or endemic diseases are presenting outside their traditional boundaries due to changing habitats of microbes and vectors and may be easily overlooked, resulting in a delayed diagnosis. Being prepared to diagnose diseases with increasing incidence secondary to climate change and discussing this with patients is an important physician obligation, but it is not the only one. We cannot effectively advocate for the health of patients and the community while ignoring the destruction of the environment. Our additional responsibility is straightforward—being advocates for good stewardship of the Earth’s resources now on both a personal and a policy level.22
The term climate refers to the average weather conditions of a specific geographic location measured over several decades.1 While a certain degree of variation in the Earth’s climate is expected, a persistent warming or cooling trend is not. The factors driving the Earth’s warming remain difficult to prove.2 We know the Earth previously has undergone dramatic climate changes and that natural factors driving these changes are varied (eg, the relationship between the Earth and the Sun, volcanic eruptions, solar irradiance).1,3 These factors ideally change over protracted periods of time in a way that allows organisms to adapt to new environments.
Anthropogenic climate change refers to human-caused climate change. This is thought to be a major driving factor in the Earth’s recent warming trend, partly due to the rapidity of warming in recent years.3 According to climate scientists, the Earth’s temperature has risen 4°C to 7°C over the past 5000 years, but it has risen 0.7°C in just the past 100 years alone.4 Greenhouse gases such as carbon dioxide are emitted by various natural processes and human activities and play a central role in current warming because they trap solar heat and increase ambient temperature.3
In a recent edition of the commonly cited textbook Dermatology, Bolognia et al5 referenced climate change only once in a figure legend regarding the expansion of dengue fever in the Americas. However, climate change may have the potential to cause outright skin disease epidemics worldwide, and the Climate Change Committee of the International Society of Dermatology has called upon dermatologists across the globe to help raise awareness of this issue.6
Much of the literature regarding the effects of climate change on human health focuses on insect-borne diseases, but over the past decade other areas of impact also have been investigated, such as increases in airborne diseases, zoonoses, newly endemic saprophytic and dimorphic fungal infections, fecal-oral diseases, and severe allergic disease.7,8 It is postulated that climate change leads to region-specific increases in human disease because it creates newly favorable habitats for infectious agents, their vectors. and their reservoirs, allowing expansion of their ranges and access to immunologically naïve populations.9 Furthermore, extreme weather events such as heat waves, hurricanes, and flooding, which are expected to increase in frequency as a result of climate change, have all been linked to infectious disease outbreaks.10
Lyme Disease
In the past 20 years, Lyme disease incidence has tripled in the United States.11 It has been hypothesized that the increase may be occurring as a result of the expanding geographic distribution of the Ixodes tick and its mammalian hosts (eg, white-tailed deer) under the influence of climate change.12 Lyme disease is a multisystem disease affecting the skin, joints, heart, and nervous system. Its most characteristic manifestation is cutaneous in the form of erythema migrans. Dermatologists may be called upon to play an increasingly important role in early detection and treatment of this potentially chronic and debilitating condition.
Arboviruses
Arboviruses are transmitted by arthropods and are an important category of climate change–related diseases due to the expansion of the mosquito habitat worldwide. The vectorial capacity for the transmission of dengue fever has increased worldwide by 9.4% via Aedes aegypti and 11.1% via Aedes albopictus since 1950.13 Dengue fever, also known as breakbone fever, presents with intense joint pain, fevers, headaches, and a transient morbilliform rash that desquamates with defervescence and in some cases will incite hemorrhagic skin lesions.14 Dengue fever previously was considered to be a tropical disease but locally acquired cases have been reported in the United States, including Texas, Hawaii, and Florida.15,16
Reports of local transmission of chikungunya, another arbovirus transmitted by A albopictus and A aegypti mosquitoes in Florida, the US Virgin Islands, and Puerto Rico, began in 2014.17 A higher prevalence of these diseases within the United States also may be related to increased globalization, with US travelers returning from endemic regions with infections. Prior to 2014, transmission occurred in traditional endemic regions, primarily in Asia, Africa, or island nations in the Indian Ocean. Like dengue fever, chikungunya causes high fevers, cutaneous manifestations (eg, urticarial papules, morbilliform eruption, hypermelanosis, intertriginous lesions, lymphedema),17 and intense joint pain. Unlike dengue fever, however, joint involvement can be chronic, erosive, and debilitating.
Lastly, New World leishmaniasis, an arboviral disease characterized by mucocutaneous ulcers and transmitted by phlebotomine sand flies, has been acquired locally in Oklahoma and Texas when it was previously considered to be endemic to Mexico and Central and South America.14,18 The habitats of New World Leishmania species are expected to expand northward, with an ecological niche model predicting that they reach southern Canada by the year 2080 due to the expanding habitats of sand fly and rodent vectors.19
Fungal Infections
In the Pacific Northwest, there have been reports of newly endemic Cryptococcus gattii and Coccidioides immitis, both of which previously had been confined to the southwestern United States.8 Endemic ranges of these mycotic pathogens may be expanding for a variety of reasons, with climate change creating new regions conducive to the colonization of these species.8,20,21Coccidioides immitis is a soil-dwelling fungus that usually presents with primary pulmonary disease that can disseminate acutely or even months later. Prompt recognition of disseminated disease may allow life-saving therapy to be initiated. Cryptococcus gattii is a fungus with multiple niches, including oil, trees, and birds.20 This fungus also is acquired via inhalation, with dissemination occurring most commonly in immunosuppressed patients to the central nervous system, bone, and skin. Primary or secondary infection with both of these fungi may present with cutaneous manifestations presenting as polymorphous lesions, including umbilicated or ulcerated papules, indurated nodules, and acneiform pustules.
Final Thoughts
Awareness of the shifting habitats of microorganisms and vectors locally is important in order for clinicians to make correct diagnoses in a timely fashion. Regional or endemic diseases are presenting outside their traditional boundaries due to changing habitats of microbes and vectors and may be easily overlooked, resulting in a delayed diagnosis. Being prepared to diagnose diseases with increasing incidence secondary to climate change and discussing this with patients is an important physician obligation, but it is not the only one. We cannot effectively advocate for the health of patients and the community while ignoring the destruction of the environment. Our additional responsibility is straightforward—being advocates for good stewardship of the Earth’s resources now on both a personal and a policy level.22
- Climate Central. Global Weirdness: Severe Storms, Deadly Heat Waves, Relentless Drought, Rising Seas, and the Weather of the Future. New York, NY: Pantheon Books; 2012.
- Cook J, Nuccitelli D, Green SA, et al. Quantifying the consensus on anthropogenic global warming in the scientific literature. Environ Res Lett. 2013;8:024024.
- A blanket around the Earth. NASA Climate website. https://climate.nasa.gov/causes/. Accessed February 5, 2018.
- How is today’s warming different from the past? NASA Earth Observatory website. https://earthobservatory.nasa.gov/Features/GlobalWarming/page3.php. Accessed February 5, 2018.
- Mancini AJ, Shani-Adir A, Sidbury R. Other viral diseases. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Philadelphia, PA: Elsevier; 2017:1425-1446.
- Andersen LK, Davis MDP. A wake-up call to dermatologists—climate change affects the skin. Int J Dermatol. 2017;56:E198-E199.
- Liang L, Gong P. Climate change and human infectious diseases: a synthesis of research findings from global and spatio-temporal perspectives [published online March 23, 2017]. Environ Int. 2017;103:99-108.
- Lockhart SR, McCotter OZ, Chiller TM. Emerging fungal infections in the Pacific Northwest: the unrecognized burden and geographic range of Cryptococcus gattii and Coccidioides immitis. Microbiol Spectr. 2016;4. doi:10.1128/microbiolspec.EI10-0016-2016.
- Kilpatrick AM, Randolph SE. Drivers, dynamics, and control of emerging vector-borne zoonotic diseases. Lancet. 2012;380:1946-1955.
- McMichael AJ. Extreme weather events and infectious disease outbreaks. Virulence. 2015;6:543-547.
- Lyme disease graphs. CDC website. https://www.cdc.gov/lyme/stats/graphs.html. Updated November 1, 2017. Accessed April 12, 2018.
- Stone BL, Tourand Y, Brissette CA. Brave new worlds: the expanding universe of Lyme disease. Vector Borne Zoonotic Dis. 2017;17:619-629.
- Watts N, Amann M, Ayeb-Karlsson S, et al. The Lancet Countdown on health and climate change: from 25 years of inaction to a global transformation for public health [published online October 30, 2017]. Lancet. doi:10.1016/S0140-6736(17)32464-9.
- Nawas ZY, Tong Y, Kollipara R, et al. Emerging infectious diseases with cutaneous manifestations: viral and bacterial infections. J Am Acad Dermatol. 2016;75:1-16.
- Kaffenberger BH, Shetlar D, Norton SA, et al. The effect of climate change on skin disease in North America. J Am Acad Dermatol. 2017;76:140-147.
- Dengue. CDC website. https://www.cdc.gov/dengue/epidemiology/index.html. Updated June 9, 2014. Accessed April 3, 2018.
- Chikungunya virus in the United States. CDC website. https://www.cdc.gov/chikungunya/geo/united-states.html. Updated October 30, 2017. Accessed April 4, 2018.
- Clarke CF, Bradley KK, Wright JH, et al. Emergence of autochthonous cutaneous leishmaniasis in northeastern Texas and southeastern Oklahoma. Am J Trop Med Hyg. 2013;88:157-61.
- González C, Wang O, Strutz SE, et al. Climate change and risk of leishmaniasis in North America: predictions from ecological niche models of vector and reservoir species. PLoS Negl Trop Dis. 2010;4:E585.
- Chang CC, Chen SC. Colliding epidemics and the rise of cryptococcosis. J Fungi (Basel). 2015;2. doi: 10.3390/jof2010001.
- Marsden-Haug N, Goldoft M, Ralston C, et al. Coccidioidomycosis acquired in Washington state. Clin Infect Dis. 2013;56:847-850.
- Rosenbach M. Climate change & dermatology: what can you do? Paper presented at: American Academy of Dermatology Annual Meeting; March 3-7, 2017; Orlando, FL.
- Climate Central. Global Weirdness: Severe Storms, Deadly Heat Waves, Relentless Drought, Rising Seas, and the Weather of the Future. New York, NY: Pantheon Books; 2012.
- Cook J, Nuccitelli D, Green SA, et al. Quantifying the consensus on anthropogenic global warming in the scientific literature. Environ Res Lett. 2013;8:024024.
- A blanket around the Earth. NASA Climate website. https://climate.nasa.gov/causes/. Accessed February 5, 2018.
- How is today’s warming different from the past? NASA Earth Observatory website. https://earthobservatory.nasa.gov/Features/GlobalWarming/page3.php. Accessed February 5, 2018.
- Mancini AJ, Shani-Adir A, Sidbury R. Other viral diseases. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Philadelphia, PA: Elsevier; 2017:1425-1446.
- Andersen LK, Davis MDP. A wake-up call to dermatologists—climate change affects the skin. Int J Dermatol. 2017;56:E198-E199.
- Liang L, Gong P. Climate change and human infectious diseases: a synthesis of research findings from global and spatio-temporal perspectives [published online March 23, 2017]. Environ Int. 2017;103:99-108.
- Lockhart SR, McCotter OZ, Chiller TM. Emerging fungal infections in the Pacific Northwest: the unrecognized burden and geographic range of Cryptococcus gattii and Coccidioides immitis. Microbiol Spectr. 2016;4. doi:10.1128/microbiolspec.EI10-0016-2016.
- Kilpatrick AM, Randolph SE. Drivers, dynamics, and control of emerging vector-borne zoonotic diseases. Lancet. 2012;380:1946-1955.
- McMichael AJ. Extreme weather events and infectious disease outbreaks. Virulence. 2015;6:543-547.
- Lyme disease graphs. CDC website. https://www.cdc.gov/lyme/stats/graphs.html. Updated November 1, 2017. Accessed April 12, 2018.
- Stone BL, Tourand Y, Brissette CA. Brave new worlds: the expanding universe of Lyme disease. Vector Borne Zoonotic Dis. 2017;17:619-629.
- Watts N, Amann M, Ayeb-Karlsson S, et al. The Lancet Countdown on health and climate change: from 25 years of inaction to a global transformation for public health [published online October 30, 2017]. Lancet. doi:10.1016/S0140-6736(17)32464-9.
- Nawas ZY, Tong Y, Kollipara R, et al. Emerging infectious diseases with cutaneous manifestations: viral and bacterial infections. J Am Acad Dermatol. 2016;75:1-16.
- Kaffenberger BH, Shetlar D, Norton SA, et al. The effect of climate change on skin disease in North America. J Am Acad Dermatol. 2017;76:140-147.
- Dengue. CDC website. https://www.cdc.gov/dengue/epidemiology/index.html. Updated June 9, 2014. Accessed April 3, 2018.
- Chikungunya virus in the United States. CDC website. https://www.cdc.gov/chikungunya/geo/united-states.html. Updated October 30, 2017. Accessed April 4, 2018.
- Clarke CF, Bradley KK, Wright JH, et al. Emergence of autochthonous cutaneous leishmaniasis in northeastern Texas and southeastern Oklahoma. Am J Trop Med Hyg. 2013;88:157-61.
- González C, Wang O, Strutz SE, et al. Climate change and risk of leishmaniasis in North America: predictions from ecological niche models of vector and reservoir species. PLoS Negl Trop Dis. 2010;4:E585.
- Chang CC, Chen SC. Colliding epidemics and the rise of cryptococcosis. J Fungi (Basel). 2015;2. doi: 10.3390/jof2010001.
- Marsden-Haug N, Goldoft M, Ralston C, et al. Coccidioidomycosis acquired in Washington state. Clin Infect Dis. 2013;56:847-850.
- Rosenbach M. Climate change & dermatology: what can you do? Paper presented at: American Academy of Dermatology Annual Meeting; March 3-7, 2017; Orlando, FL.
Two good apps for management of cervical cancer screening results
Mobile applications are useful for clinical decision making. An example is in the area of cervical cancer screening. The incidence of cervical cancer and mortality from the disease in the United States has decreased with the implementation of cervical cancer screening programs.1 However, being up to date on the guidelines can be challenging. In 2001, the revised Bethesda system terminology for reporting cervical cytology results became available. In response, the American Society for Colposcopy and Cervical Pathology (ASCCP) developed comprehensive, evidence-based consensus guidelines to assist health care providers in managing abnormal screening results. In 2006, the guidelines were revised, and in 2012, revised again.2
In a search in the Apple iTunes and Google Play stores for apps useful for gynecologic oncology providers, Dr. Sara Farag, colleagues, and I identified and evaluated highly 2 cervical cancer screening apps: ASCCP Mobile and Pap Reader.3 These apps can aid any health care provider who performs Pap smear screening and who manages screening results.
ASCCP Mobile includes follow-up guidelines regarding colposcopy results as well as guidelines for posthysterectomy and pregnant women. The app also has a clinical decision support system (an active knowledge system that uses 2 or more items of patient data to generate case-specific advice).
Pap Reader includes guidelines for postmenopausal and pregnant women and also has a clinical decision support system. Unlike ASCCP Mobile, Pap Reader is free.
The recommended cervical cancer screening apps are listed in the TABLE alphabetically and are detailed with a shortened version of the APPLICATIONS scoring system, APPLI (app comprehensiveness, price, platform, literature use, and important special features).4 I hope these apps will assist you in your management of patients who undergo Pap smear screening.
Share your thoughts! Send your Letter to the Editor to rbarbieri@mdedge.com. Please include your name and the city and state in which you practice.
- American College of Obstetricians and Gynecologists. Practice bulletin summary no. 168: cervical cancer screening and prevention. Obstet Gynecol. 2016;128(4):923-925.
- Massad LS, Einstein MH, Huh WK, et al; 2012 ASCCP Consensus Guidelines Conference. 2012 updated consensus guidelines for the management of abnormal cervical cancer screening tests and cancer precursors. Obstet Gynecol. 2013;121(4):829-846.
- Farag S, Fields J, Pereira E, Chyjek K, Chen KT. Identification and rating of gynecologic oncology applications using the APPLICATIONS Scoring System. Telemed J E Health. 2016;22(12):1001-1007.
- Chyjek K, Farag S, Chen KT. Rating pregnancy wheel applications using the APPLICATIONS scoring system. Obstet Gynecol. 2015;125(6):1478-1483.
Mobile applications are useful for clinical decision making. An example is in the area of cervical cancer screening. The incidence of cervical cancer and mortality from the disease in the United States has decreased with the implementation of cervical cancer screening programs.1 However, being up to date on the guidelines can be challenging. In 2001, the revised Bethesda system terminology for reporting cervical cytology results became available. In response, the American Society for Colposcopy and Cervical Pathology (ASCCP) developed comprehensive, evidence-based consensus guidelines to assist health care providers in managing abnormal screening results. In 2006, the guidelines were revised, and in 2012, revised again.2
In a search in the Apple iTunes and Google Play stores for apps useful for gynecologic oncology providers, Dr. Sara Farag, colleagues, and I identified and evaluated highly 2 cervical cancer screening apps: ASCCP Mobile and Pap Reader.3 These apps can aid any health care provider who performs Pap smear screening and who manages screening results.
ASCCP Mobile includes follow-up guidelines regarding colposcopy results as well as guidelines for posthysterectomy and pregnant women. The app also has a clinical decision support system (an active knowledge system that uses 2 or more items of patient data to generate case-specific advice).
Pap Reader includes guidelines for postmenopausal and pregnant women and also has a clinical decision support system. Unlike ASCCP Mobile, Pap Reader is free.
The recommended cervical cancer screening apps are listed in the TABLE alphabetically and are detailed with a shortened version of the APPLICATIONS scoring system, APPLI (app comprehensiveness, price, platform, literature use, and important special features).4 I hope these apps will assist you in your management of patients who undergo Pap smear screening.

Share your thoughts! Send your Letter to the Editor to rbarbieri@mdedge.com. Please include your name and the city and state in which you practice.
Mobile applications are useful for clinical decision making. An example is in the area of cervical cancer screening. The incidence of cervical cancer and mortality from the disease in the United States has decreased with the implementation of cervical cancer screening programs.1 However, being up to date on the guidelines can be challenging. In 2001, the revised Bethesda system terminology for reporting cervical cytology results became available. In response, the American Society for Colposcopy and Cervical Pathology (ASCCP) developed comprehensive, evidence-based consensus guidelines to assist health care providers in managing abnormal screening results. In 2006, the guidelines were revised, and in 2012, revised again.2
In a search in the Apple iTunes and Google Play stores for apps useful for gynecologic oncology providers, Dr. Sara Farag, colleagues, and I identified and evaluated highly 2 cervical cancer screening apps: ASCCP Mobile and Pap Reader.3 These apps can aid any health care provider who performs Pap smear screening and who manages screening results.
ASCCP Mobile includes follow-up guidelines regarding colposcopy results as well as guidelines for posthysterectomy and pregnant women. The app also has a clinical decision support system (an active knowledge system that uses 2 or more items of patient data to generate case-specific advice).
Pap Reader includes guidelines for postmenopausal and pregnant women and also has a clinical decision support system. Unlike ASCCP Mobile, Pap Reader is free.
The recommended cervical cancer screening apps are listed in the TABLE alphabetically and are detailed with a shortened version of the APPLICATIONS scoring system, APPLI (app comprehensiveness, price, platform, literature use, and important special features).4 I hope these apps will assist you in your management of patients who undergo Pap smear screening.

Share your thoughts! Send your Letter to the Editor to rbarbieri@mdedge.com. Please include your name and the city and state in which you practice.
- American College of Obstetricians and Gynecologists. Practice bulletin summary no. 168: cervical cancer screening and prevention. Obstet Gynecol. 2016;128(4):923-925.
- Massad LS, Einstein MH, Huh WK, et al; 2012 ASCCP Consensus Guidelines Conference. 2012 updated consensus guidelines for the management of abnormal cervical cancer screening tests and cancer precursors. Obstet Gynecol. 2013;121(4):829-846.
- Farag S, Fields J, Pereira E, Chyjek K, Chen KT. Identification and rating of gynecologic oncology applications using the APPLICATIONS Scoring System. Telemed J E Health. 2016;22(12):1001-1007.
- Chyjek K, Farag S, Chen KT. Rating pregnancy wheel applications using the APPLICATIONS scoring system. Obstet Gynecol. 2015;125(6):1478-1483.
- American College of Obstetricians and Gynecologists. Practice bulletin summary no. 168: cervical cancer screening and prevention. Obstet Gynecol. 2016;128(4):923-925.
- Massad LS, Einstein MH, Huh WK, et al; 2012 ASCCP Consensus Guidelines Conference. 2012 updated consensus guidelines for the management of abnormal cervical cancer screening tests and cancer precursors. Obstet Gynecol. 2013;121(4):829-846.
- Farag S, Fields J, Pereira E, Chyjek K, Chen KT. Identification and rating of gynecologic oncology applications using the APPLICATIONS Scoring System. Telemed J E Health. 2016;22(12):1001-1007.
- Chyjek K, Farag S, Chen KT. Rating pregnancy wheel applications using the APPLICATIONS scoring system. Obstet Gynecol. 2015;125(6):1478-1483.














