User login
Smoking-cessation interest and success vary by race, ethnicity
Just over 55% of adult cigarette smokers made an attempt to quit in the past year, and 7.4% said that they recently quit, according to investigators from the Centers or Disease Control and Prevention.
Data from the 2015 National Health Interview Survey (NHIS) show that 68% of cigarette smokers were interested in quitting, with considerable variation seen according to race and ethnicity (MMWR. 2017;65[52]:1457-64).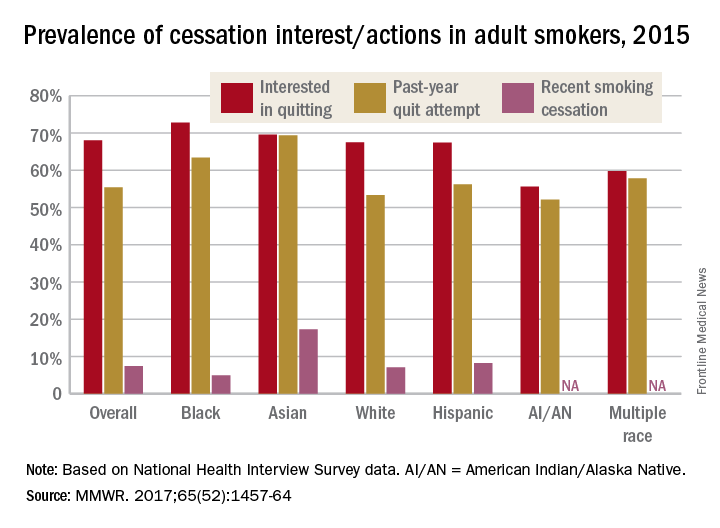
American Indian/Alaska Native smokers were the least likely to be interested in quitting (55.6%) and to have attempted to quit (52.1%), but the sample size was too small to report a reliable quit rate. The amount of survey participants of multiple races was also too small to report a reliable quit rate. Among that group, 59.8% were interested in quitting and 57.8% had attempted to quit in the past year, the NHIS data showed.
The sizes of surveyed populations for individual races and ethnicities were not reported, but the total sample size for the 2015 NHIS was 33,672.
Just over 55% of adult cigarette smokers made an attempt to quit in the past year, and 7.4% said that they recently quit, according to investigators from the Centers or Disease Control and Prevention.
Data from the 2015 National Health Interview Survey (NHIS) show that 68% of cigarette smokers were interested in quitting, with considerable variation seen according to race and ethnicity (MMWR. 2017;65[52]:1457-64).
American Indian/Alaska Native smokers were the least likely to be interested in quitting (55.6%) and to have attempted to quit (52.1%), but the sample size was too small to report a reliable quit rate. The amount of survey participants of multiple races was also too small to report a reliable quit rate. Among that group, 59.8% were interested in quitting and 57.8% had attempted to quit in the past year, the NHIS data showed.
The sizes of surveyed populations for individual races and ethnicities were not reported, but the total sample size for the 2015 NHIS was 33,672.
Just over 55% of adult cigarette smokers made an attempt to quit in the past year, and 7.4% said that they recently quit, according to investigators from the Centers or Disease Control and Prevention.
Data from the 2015 National Health Interview Survey (NHIS) show that 68% of cigarette smokers were interested in quitting, with considerable variation seen according to race and ethnicity (MMWR. 2017;65[52]:1457-64).
American Indian/Alaska Native smokers were the least likely to be interested in quitting (55.6%) and to have attempted to quit (52.1%), but the sample size was too small to report a reliable quit rate. The amount of survey participants of multiple races was also too small to report a reliable quit rate. Among that group, 59.8% were interested in quitting and 57.8% had attempted to quit in the past year, the NHIS data showed.
The sizes of surveyed populations for individual races and ethnicities were not reported, but the total sample size for the 2015 NHIS was 33,672.
Most cigarette smokers attempt to quit without evidence-based techniques
More than half of cigarette smokers have received advice to quit from a health care professional, but less than a third used medication or counseling in their cessation attempt, according to investigators from the Centers for Disease Control and Prevention.
In 2015, just over 57% of adult smokers said that a health care professional had advised them to quit in the past year. Of those who tried to quit, 29% used medication such as nicotine patches or gum, varenicline, or bupropion; 7% used counseling (including a stop-smoking clinic, class, or support group and a telephone help line); and 31% used counseling and/or medication, the investigators reported (MMWR 2017;65[52]:1457-64).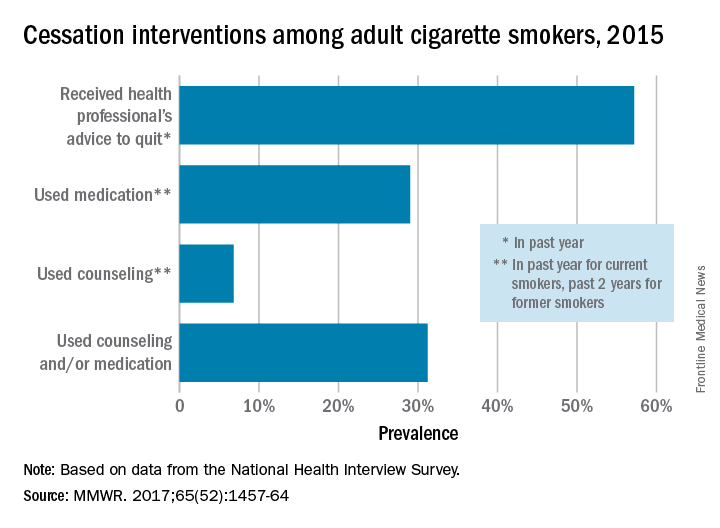
With the overall cessation rate at less than 10%, “it is critical for health care providers to consistently identify smokers, advise them to quit, and offer evidence-based cessation treatments, and for insurers to cover and promote the use of these treatments and remove barriers to accessing them,” the investigators wrote.
More than half of cigarette smokers have received advice to quit from a health care professional, but less than a third used medication or counseling in their cessation attempt, according to investigators from the Centers for Disease Control and Prevention.
In 2015, just over 57% of adult smokers said that a health care professional had advised them to quit in the past year. Of those who tried to quit, 29% used medication such as nicotine patches or gum, varenicline, or bupropion; 7% used counseling (including a stop-smoking clinic, class, or support group and a telephone help line); and 31% used counseling and/or medication, the investigators reported (MMWR 2017;65[52]:1457-64).
With the overall cessation rate at less than 10%, “it is critical for health care providers to consistently identify smokers, advise them to quit, and offer evidence-based cessation treatments, and for insurers to cover and promote the use of these treatments and remove barriers to accessing them,” the investigators wrote.
More than half of cigarette smokers have received advice to quit from a health care professional, but less than a third used medication or counseling in their cessation attempt, according to investigators from the Centers for Disease Control and Prevention.
In 2015, just over 57% of adult smokers said that a health care professional had advised them to quit in the past year. Of those who tried to quit, 29% used medication such as nicotine patches or gum, varenicline, or bupropion; 7% used counseling (including a stop-smoking clinic, class, or support group and a telephone help line); and 31% used counseling and/or medication, the investigators reported (MMWR 2017;65[52]:1457-64).
With the overall cessation rate at less than 10%, “it is critical for health care providers to consistently identify smokers, advise them to quit, and offer evidence-based cessation treatments, and for insurers to cover and promote the use of these treatments and remove barriers to accessing them,” the investigators wrote.
FROM MMWR
Diabetes-related kidney failure down sharply in Native Americans
Kidney failure in Native Americans and Alaska Natives with diabetes has declined drastically over the last 20 years, according to new data released as part of this month’s Vital Signs report by the CDC.
“The 54 percent decline in kidney failure from diabetes followed implementation of public health and population approaches to diabetes as well as improvements in clinical care by the IHS [Indian Health Service],” said Mary L. Smith, principal deputy director of the Indian Health Service.
Of all U.S.-based populations, Native Americans are the most susceptible to diabetes and are about twice as likely as white Americans to develop diabetes. Furthermore, 69% of kidney failure deaths in Native Americans are the result of diabetes (MMWR. 2017 Jan 10. doi: 10.15585/mmwr.mm6601e1).
Since 1996, however, kidney failure has dropped more among Native Americans than any other ethnic group in the country. The 54% drop represents a decrease from 57.3 diabetes-related end-stage renal disease cases per 100,000 population in 1996 to 26.5 per 100,000 population in 2013 among U.S. adults.
“This decline is especially remarkable given the well-documented health and socioeconomic disparities in the [Native American and Alaska Natives] population, including poverty, limited health care resources, and disproportionate burden of many health problems,” wrote the authors of the Vital Signs report.
According to the report, blood sugar control among Native American populations has improved by 10%, kidney testing in diabetic Native Americans aged 65 years or older is 50% greater than Medicare diabetes patients of the same age, and the average blood pressure of Native Americans with both diabetes and hypertension was 133/76 in 2015.
“We believe these strategies can be effective in any population,” Ms. Smith stated, a sentiment that was also shared by Tom Frieden, MD, director of the CDC.
“Strong coordinated clinical care and education, community outreach and environmental changes can make a dramatic difference in reducing complications from diabetes for all Americans,” Dr. Frieden said in a statement.
Not only does diabetes persist as a significant burden on the U.S. health care system, but kidney failure in particular can be costly. Figures released by the CDC indicate that average medical costs associated with kidney failure in 2013 were as high as $82,000 per patient, with Medicare spending nearly $14 billion for kidney failure treatments in the same year.
“The findings in this report are consistent with other studies among [Native Americans and Alaska Natives] nationwide and among Pima Indians in the Southwest, which concluded that improvements in blood pressure, blood glucose, and the use of ACE inhibitors and [angiotensin II receptor blockers] played a significant role in the decline of [diabetes-related end-stage renal disease] in these populations,” the report concludes.
To ensure that kidney failure decreases continue in Native Americans, the U.S. government will continue funding diabetes screening and prevention efforts in applicable communities, assist community health care facilities to provide care for diabetes, and will establish a nationwide system for tracking chronic kidney disease. The CDC also advocates using population approaches and coordinated care to treat diabetes, advising health care professionals to “integrate kidney disease prevention and education into routine diabetes care.”
“The Indian Health Service has made tremendous progress by applying population health and team-based approaches to diabetes and kidney care,” Dr. Frieden stated.
Kidney failure in Native Americans and Alaska Natives with diabetes has declined drastically over the last 20 years, according to new data released as part of this month’s Vital Signs report by the CDC.
“The 54 percent decline in kidney failure from diabetes followed implementation of public health and population approaches to diabetes as well as improvements in clinical care by the IHS [Indian Health Service],” said Mary L. Smith, principal deputy director of the Indian Health Service.
Of all U.S.-based populations, Native Americans are the most susceptible to diabetes and are about twice as likely as white Americans to develop diabetes. Furthermore, 69% of kidney failure deaths in Native Americans are the result of diabetes (MMWR. 2017 Jan 10. doi: 10.15585/mmwr.mm6601e1).
Since 1996, however, kidney failure has dropped more among Native Americans than any other ethnic group in the country. The 54% drop represents a decrease from 57.3 diabetes-related end-stage renal disease cases per 100,000 population in 1996 to 26.5 per 100,000 population in 2013 among U.S. adults.
“This decline is especially remarkable given the well-documented health and socioeconomic disparities in the [Native American and Alaska Natives] population, including poverty, limited health care resources, and disproportionate burden of many health problems,” wrote the authors of the Vital Signs report.
According to the report, blood sugar control among Native American populations has improved by 10%, kidney testing in diabetic Native Americans aged 65 years or older is 50% greater than Medicare diabetes patients of the same age, and the average blood pressure of Native Americans with both diabetes and hypertension was 133/76 in 2015.
“We believe these strategies can be effective in any population,” Ms. Smith stated, a sentiment that was also shared by Tom Frieden, MD, director of the CDC.
“Strong coordinated clinical care and education, community outreach and environmental changes can make a dramatic difference in reducing complications from diabetes for all Americans,” Dr. Frieden said in a statement.
Not only does diabetes persist as a significant burden on the U.S. health care system, but kidney failure in particular can be costly. Figures released by the CDC indicate that average medical costs associated with kidney failure in 2013 were as high as $82,000 per patient, with Medicare spending nearly $14 billion for kidney failure treatments in the same year.
“The findings in this report are consistent with other studies among [Native Americans and Alaska Natives] nationwide and among Pima Indians in the Southwest, which concluded that improvements in blood pressure, blood glucose, and the use of ACE inhibitors and [angiotensin II receptor blockers] played a significant role in the decline of [diabetes-related end-stage renal disease] in these populations,” the report concludes.
To ensure that kidney failure decreases continue in Native Americans, the U.S. government will continue funding diabetes screening and prevention efforts in applicable communities, assist community health care facilities to provide care for diabetes, and will establish a nationwide system for tracking chronic kidney disease. The CDC also advocates using population approaches and coordinated care to treat diabetes, advising health care professionals to “integrate kidney disease prevention and education into routine diabetes care.”
“The Indian Health Service has made tremendous progress by applying population health and team-based approaches to diabetes and kidney care,” Dr. Frieden stated.
Kidney failure in Native Americans and Alaska Natives with diabetes has declined drastically over the last 20 years, according to new data released as part of this month’s Vital Signs report by the CDC.
“The 54 percent decline in kidney failure from diabetes followed implementation of public health and population approaches to diabetes as well as improvements in clinical care by the IHS [Indian Health Service],” said Mary L. Smith, principal deputy director of the Indian Health Service.
Of all U.S.-based populations, Native Americans are the most susceptible to diabetes and are about twice as likely as white Americans to develop diabetes. Furthermore, 69% of kidney failure deaths in Native Americans are the result of diabetes (MMWR. 2017 Jan 10. doi: 10.15585/mmwr.mm6601e1).
Since 1996, however, kidney failure has dropped more among Native Americans than any other ethnic group in the country. The 54% drop represents a decrease from 57.3 diabetes-related end-stage renal disease cases per 100,000 population in 1996 to 26.5 per 100,000 population in 2013 among U.S. adults.
“This decline is especially remarkable given the well-documented health and socioeconomic disparities in the [Native American and Alaska Natives] population, including poverty, limited health care resources, and disproportionate burden of many health problems,” wrote the authors of the Vital Signs report.
According to the report, blood sugar control among Native American populations has improved by 10%, kidney testing in diabetic Native Americans aged 65 years or older is 50% greater than Medicare diabetes patients of the same age, and the average blood pressure of Native Americans with both diabetes and hypertension was 133/76 in 2015.
“We believe these strategies can be effective in any population,” Ms. Smith stated, a sentiment that was also shared by Tom Frieden, MD, director of the CDC.
“Strong coordinated clinical care and education, community outreach and environmental changes can make a dramatic difference in reducing complications from diabetes for all Americans,” Dr. Frieden said in a statement.
Not only does diabetes persist as a significant burden on the U.S. health care system, but kidney failure in particular can be costly. Figures released by the CDC indicate that average medical costs associated with kidney failure in 2013 were as high as $82,000 per patient, with Medicare spending nearly $14 billion for kidney failure treatments in the same year.
“The findings in this report are consistent with other studies among [Native Americans and Alaska Natives] nationwide and among Pima Indians in the Southwest, which concluded that improvements in blood pressure, blood glucose, and the use of ACE inhibitors and [angiotensin II receptor blockers] played a significant role in the decline of [diabetes-related end-stage renal disease] in these populations,” the report concludes.
To ensure that kidney failure decreases continue in Native Americans, the U.S. government will continue funding diabetes screening and prevention efforts in applicable communities, assist community health care facilities to provide care for diabetes, and will establish a nationwide system for tracking chronic kidney disease. The CDC also advocates using population approaches and coordinated care to treat diabetes, advising health care professionals to “integrate kidney disease prevention and education into routine diabetes care.”
“The Indian Health Service has made tremendous progress by applying population health and team-based approaches to diabetes and kidney care,” Dr. Frieden stated.
High levels of flu activity in 10 U.S. states
The 2016-2017 flu season shifted into high gear at the end of calendar year 2016, as four states were reported to be at the highest level of flu activity and six others were close behind, according to the Centers for Disease Control and Prevention.
For the week ending Dec. 31, 2016, Georgia, New Jersey, Oklahoma, and Oregon were at level 10 on the CDC’s 1-10 scale of influenza-like illness (ILI). Others in the “high” range were New York at level 9 and Alabama, Louisiana, Missouri, South Carolina, and Utah at level 8. Puerto Rico was also at level 8, after being at level 10 for the previous few weeks. An additional 10 states were in the “moderate” range (6-7), the CDC reported.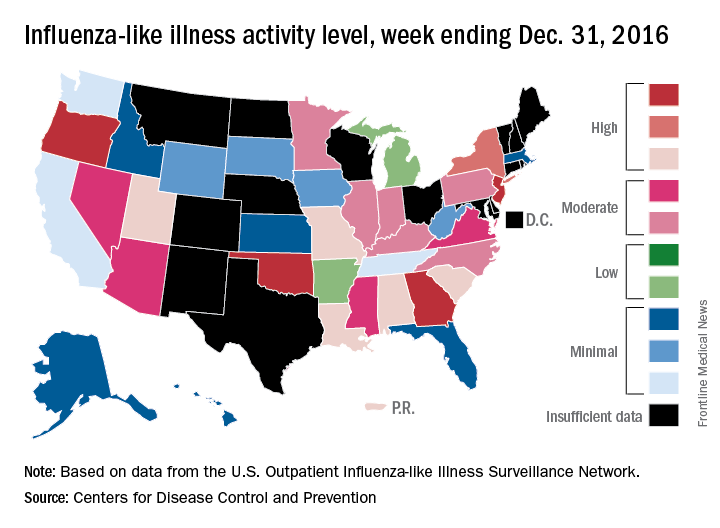
The CDC has not reported any flu-related pediatric deaths yet this season. Pediatric death totals for each of the last 3 years were 111 for 2013-2014, 148 for 2014-2015, and 89 for 2015-2016, the CDC said.
The 2016-2017 flu season shifted into high gear at the end of calendar year 2016, as four states were reported to be at the highest level of flu activity and six others were close behind, according to the Centers for Disease Control and Prevention.
For the week ending Dec. 31, 2016, Georgia, New Jersey, Oklahoma, and Oregon were at level 10 on the CDC’s 1-10 scale of influenza-like illness (ILI). Others in the “high” range were New York at level 9 and Alabama, Louisiana, Missouri, South Carolina, and Utah at level 8. Puerto Rico was also at level 8, after being at level 10 for the previous few weeks. An additional 10 states were in the “moderate” range (6-7), the CDC reported.
The CDC has not reported any flu-related pediatric deaths yet this season. Pediatric death totals for each of the last 3 years were 111 for 2013-2014, 148 for 2014-2015, and 89 for 2015-2016, the CDC said.
The 2016-2017 flu season shifted into high gear at the end of calendar year 2016, as four states were reported to be at the highest level of flu activity and six others were close behind, according to the Centers for Disease Control and Prevention.
For the week ending Dec. 31, 2016, Georgia, New Jersey, Oklahoma, and Oregon were at level 10 on the CDC’s 1-10 scale of influenza-like illness (ILI). Others in the “high” range were New York at level 9 and Alabama, Louisiana, Missouri, South Carolina, and Utah at level 8. Puerto Rico was also at level 8, after being at level 10 for the previous few weeks. An additional 10 states were in the “moderate” range (6-7), the CDC reported.
The CDC has not reported any flu-related pediatric deaths yet this season. Pediatric death totals for each of the last 3 years were 111 for 2013-2014, 148 for 2014-2015, and 89 for 2015-2016, the CDC said.
United States about to top 40,000 Zika cases
The number of pregnant women with laboratory evidence of Zika infection jumped up a bit at the end of 2016, and the United States approached 40,000 Zika cases among all Americans at the beginning of the new year, according to reports from the Centers for Disease Control and Prevention.
There were 187 additional pregnant women with Zika virus infection reported in the 2 weeks ending Dec. 27, compared with the 136 new reports of infected women in each of the two previous comparable periods (Dec. 1-13 and Nov. 18-30). Most of the 187 new cases were reported in the U.S. territories, while 46 were reported in the 50 states and the District of Columbia. There have been delays in reporting, the CDC noted, so these cannot be considered real-time estimates.
For the 2 weeks ending Dec. 27, there were reports of two more infants born with Zika-related birth defects, bringing the total to 36 for the states/D.C. The CDC is no longer reporting adverse pregnancy outcomes for the territories because Puerto Rico is not using the same inclusion criteria. The number of pregnancy losses remains at five in the states/D.C., where it has been since August. Aggregated data from the U.S. Zika Pregnancy Registry show that there have been 875 completed pregnancies with or without birth defects, the CDC said.
Zika-related birth defects reported by the CDC could include microcephaly, calcium deposits in the brain indicating possible brain damage, excess fluid in the brain cavities and surrounding the brain, absent or poorly formed brain structures, abnormal eye development, or other problems resulting from brain damage that affect nerves, muscles, and bones. The pregnancy losses encompass any miscarriage, stillbirth, and termination with evidence of birth defects.
The number of pregnant women with laboratory evidence of Zika infection jumped up a bit at the end of 2016, and the United States approached 40,000 Zika cases among all Americans at the beginning of the new year, according to reports from the Centers for Disease Control and Prevention.
There were 187 additional pregnant women with Zika virus infection reported in the 2 weeks ending Dec. 27, compared with the 136 new reports of infected women in each of the two previous comparable periods (Dec. 1-13 and Nov. 18-30). Most of the 187 new cases were reported in the U.S. territories, while 46 were reported in the 50 states and the District of Columbia. There have been delays in reporting, the CDC noted, so these cannot be considered real-time estimates.
For the 2 weeks ending Dec. 27, there were reports of two more infants born with Zika-related birth defects, bringing the total to 36 for the states/D.C. The CDC is no longer reporting adverse pregnancy outcomes for the territories because Puerto Rico is not using the same inclusion criteria. The number of pregnancy losses remains at five in the states/D.C., where it has been since August. Aggregated data from the U.S. Zika Pregnancy Registry show that there have been 875 completed pregnancies with or without birth defects, the CDC said.
Zika-related birth defects reported by the CDC could include microcephaly, calcium deposits in the brain indicating possible brain damage, excess fluid in the brain cavities and surrounding the brain, absent or poorly formed brain structures, abnormal eye development, or other problems resulting from brain damage that affect nerves, muscles, and bones. The pregnancy losses encompass any miscarriage, stillbirth, and termination with evidence of birth defects.
The number of pregnant women with laboratory evidence of Zika infection jumped up a bit at the end of 2016, and the United States approached 40,000 Zika cases among all Americans at the beginning of the new year, according to reports from the Centers for Disease Control and Prevention.
There were 187 additional pregnant women with Zika virus infection reported in the 2 weeks ending Dec. 27, compared with the 136 new reports of infected women in each of the two previous comparable periods (Dec. 1-13 and Nov. 18-30). Most of the 187 new cases were reported in the U.S. territories, while 46 were reported in the 50 states and the District of Columbia. There have been delays in reporting, the CDC noted, so these cannot be considered real-time estimates.
For the 2 weeks ending Dec. 27, there were reports of two more infants born with Zika-related birth defects, bringing the total to 36 for the states/D.C. The CDC is no longer reporting adverse pregnancy outcomes for the territories because Puerto Rico is not using the same inclusion criteria. The number of pregnancy losses remains at five in the states/D.C., where it has been since August. Aggregated data from the U.S. Zika Pregnancy Registry show that there have been 875 completed pregnancies with or without birth defects, the CDC said.
Zika-related birth defects reported by the CDC could include microcephaly, calcium deposits in the brain indicating possible brain damage, excess fluid in the brain cavities and surrounding the brain, absent or poorly formed brain structures, abnormal eye development, or other problems resulting from brain damage that affect nerves, muscles, and bones. The pregnancy losses encompass any miscarriage, stillbirth, and termination with evidence of birth defects.
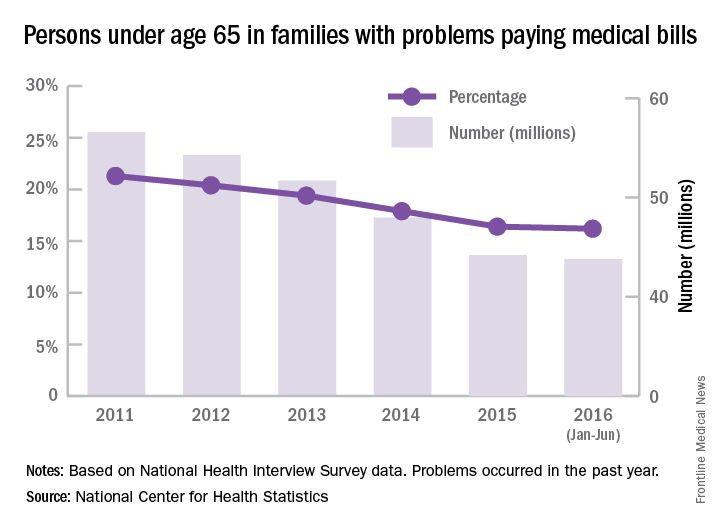
Fewer people having problems with medical bills
The number of people under age 65 years who were in families having trouble paying medical bills dropped by more than 22% from 2011 to 2016, according to the National Center for Health Statistics.
For the first 6 months of 2016, there were 43.8 million people, or 16.2% of the population under age 65 years, who were in families that had problems paying medical bills in the past year, which was down from 56.5 million (21.3 % of the population) in 2011, the NCHS reported.
The drop was consistent across race/ethnicity lines, but not the start and endpoints. The percentage of non-Hispanic blacks in families having trouble paying their medical bills dropped from 27.3% in 2011 to 23% in 2016, although there was actually a small increase from 2015 to 2016. Hispanics dropped from 24.3% in 2011 to 17.4% in 2016, non-Hispanic whites dropped from 19.8% to 15.1%, and non-Hispanic Asians went from 11% to 6%, according to data collected from 579,379 people for the National Health Interview Survey.
The number of people under age 65 years who were in families having trouble paying medical bills dropped by more than 22% from 2011 to 2016, according to the National Center for Health Statistics.
For the first 6 months of 2016, there were 43.8 million people, or 16.2% of the population under age 65 years, who were in families that had problems paying medical bills in the past year, which was down from 56.5 million (21.3 % of the population) in 2011, the NCHS reported.
The drop was consistent across race/ethnicity lines, but not the start and endpoints. The percentage of non-Hispanic blacks in families having trouble paying their medical bills dropped from 27.3% in 2011 to 23% in 2016, although there was actually a small increase from 2015 to 2016. Hispanics dropped from 24.3% in 2011 to 17.4% in 2016, non-Hispanic whites dropped from 19.8% to 15.1%, and non-Hispanic Asians went from 11% to 6%, according to data collected from 579,379 people for the National Health Interview Survey.
The number of people under age 65 years who were in families having trouble paying medical bills dropped by more than 22% from 2011 to 2016, according to the National Center for Health Statistics.
For the first 6 months of 2016, there were 43.8 million people, or 16.2% of the population under age 65 years, who were in families that had problems paying medical bills in the past year, which was down from 56.5 million (21.3 % of the population) in 2011, the NCHS reported.
The drop was consistent across race/ethnicity lines, but not the start and endpoints. The percentage of non-Hispanic blacks in families having trouble paying their medical bills dropped from 27.3% in 2011 to 23% in 2016, although there was actually a small increase from 2015 to 2016. Hispanics dropped from 24.3% in 2011 to 17.4% in 2016, non-Hispanic whites dropped from 19.8% to 15.1%, and non-Hispanic Asians went from 11% to 6%, according to data collected from 579,379 people for the National Health Interview Survey.
First drug for spinal muscular atrophy approved
The antisense oligonucleotide drug nusinersen is the first therapy approved by the U.S. Food and Drug Administration to treat children and adults with spinal muscular atrophy.
The drug was developed by Ionis Pharmaceuticals and will be marketed by Biogen under the brand name Spinraza. The antisense oligonucleotide drug is administered via an intrathecal injection and promotes transcription of the full-length survival motor neuron (SMN) protein from the SMN2 gene.
The efficacy of nusinersen was tested during a randomized clinical trial in 121 patients with infantile-onset spinal muscular atrophy who were diagnosed before the age of 6 months or were younger than 7 months at the time of their first dose. Patients were randomized 2:1 to receive an injection of nusinersen into the fluid surrounding the spinal cord or undergo a mock procedure without drug injection (a skin prick).
A total of 82 of 121 patients who were randomized were eligible for an interim analysis of the results that was requested by the FDA. The results showed 40% of patients treated with nusinersen achieved improvement in motor milestones as defined in the study, whereas none of the control patients did. The side effects during the clinical trials among patients on nusinersen included upper respiratory infection, lower respiratory infection, and constipation. Warnings and precautions include low blood platelet count and renal toxicity. Neurotoxicity was observed in animal studies.
Read the full announcement from the agency here.
The antisense oligonucleotide drug nusinersen is the first therapy approved by the U.S. Food and Drug Administration to treat children and adults with spinal muscular atrophy.
The drug was developed by Ionis Pharmaceuticals and will be marketed by Biogen under the brand name Spinraza. The antisense oligonucleotide drug is administered via an intrathecal injection and promotes transcription of the full-length survival motor neuron (SMN) protein from the SMN2 gene.
The efficacy of nusinersen was tested during a randomized clinical trial in 121 patients with infantile-onset spinal muscular atrophy who were diagnosed before the age of 6 months or were younger than 7 months at the time of their first dose. Patients were randomized 2:1 to receive an injection of nusinersen into the fluid surrounding the spinal cord or undergo a mock procedure without drug injection (a skin prick).
A total of 82 of 121 patients who were randomized were eligible for an interim analysis of the results that was requested by the FDA. The results showed 40% of patients treated with nusinersen achieved improvement in motor milestones as defined in the study, whereas none of the control patients did. The side effects during the clinical trials among patients on nusinersen included upper respiratory infection, lower respiratory infection, and constipation. Warnings and precautions include low blood platelet count and renal toxicity. Neurotoxicity was observed in animal studies.
Read the full announcement from the agency here.
The antisense oligonucleotide drug nusinersen is the first therapy approved by the U.S. Food and Drug Administration to treat children and adults with spinal muscular atrophy.
The drug was developed by Ionis Pharmaceuticals and will be marketed by Biogen under the brand name Spinraza. The antisense oligonucleotide drug is administered via an intrathecal injection and promotes transcription of the full-length survival motor neuron (SMN) protein from the SMN2 gene.
The efficacy of nusinersen was tested during a randomized clinical trial in 121 patients with infantile-onset spinal muscular atrophy who were diagnosed before the age of 6 months or were younger than 7 months at the time of their first dose. Patients were randomized 2:1 to receive an injection of nusinersen into the fluid surrounding the spinal cord or undergo a mock procedure without drug injection (a skin prick).
A total of 82 of 121 patients who were randomized were eligible for an interim analysis of the results that was requested by the FDA. The results showed 40% of patients treated with nusinersen achieved improvement in motor milestones as defined in the study, whereas none of the control patients did. The side effects during the clinical trials among patients on nusinersen included upper respiratory infection, lower respiratory infection, and constipation. Warnings and precautions include low blood platelet count and renal toxicity. Neurotoxicity was observed in animal studies.
Read the full announcement from the agency here.
FDA warns of false-positive results with Zika IgM test
, according to a safety alert issued by the Food and Drug Administration.
The ZIKV Detect IgM Capture ELISA test is the first commercially available Zika serological IgM test – it was approved by the FDA in August 2016 and is used by several commercial laboratories. The test reports only presumptive positive results and a sample has to be sent to the Centers for Disease Control and Prevention for confirmation. Final results can take up to a month to be delivered. In most instances, the preliminary test results have matched the confirmed sample results.
The FDA recommends that health care providers inform patients that presumptive positive results need to be confirmed and that they not rely on positive IgM test results as the sole basis of patient management. If a patient is pregnant, the FDA recommends contacting the laboratory to expedite the confirmation testing.
FDA officials are working with LabCorp and ZIKV Detect manufacturer InBios International to determine if the false-positive results are related to problems with the test or the commercial testing facility.
Find the full safety alert on the FDA website.
, according to a safety alert issued by the Food and Drug Administration.
The ZIKV Detect IgM Capture ELISA test is the first commercially available Zika serological IgM test – it was approved by the FDA in August 2016 and is used by several commercial laboratories. The test reports only presumptive positive results and a sample has to be sent to the Centers for Disease Control and Prevention for confirmation. Final results can take up to a month to be delivered. In most instances, the preliminary test results have matched the confirmed sample results.
The FDA recommends that health care providers inform patients that presumptive positive results need to be confirmed and that they not rely on positive IgM test results as the sole basis of patient management. If a patient is pregnant, the FDA recommends contacting the laboratory to expedite the confirmation testing.
FDA officials are working with LabCorp and ZIKV Detect manufacturer InBios International to determine if the false-positive results are related to problems with the test or the commercial testing facility.
Find the full safety alert on the FDA website.
, according to a safety alert issued by the Food and Drug Administration.
The ZIKV Detect IgM Capture ELISA test is the first commercially available Zika serological IgM test – it was approved by the FDA in August 2016 and is used by several commercial laboratories. The test reports only presumptive positive results and a sample has to be sent to the Centers for Disease Control and Prevention for confirmation. Final results can take up to a month to be delivered. In most instances, the preliminary test results have matched the confirmed sample results.
The FDA recommends that health care providers inform patients that presumptive positive results need to be confirmed and that they not rely on positive IgM test results as the sole basis of patient management. If a patient is pregnant, the FDA recommends contacting the laboratory to expedite the confirmation testing.
FDA officials are working with LabCorp and ZIKV Detect manufacturer InBios International to determine if the false-positive results are related to problems with the test or the commercial testing facility.
Find the full safety alert on the FDA website.
FDA expands indication for continuous glucose monitoring system
People with diabetes have come a step closer to a life without multiple daily finger sticks. The with diabetes, the Food and Drug Administration announced .
“Although this system still requires calibration with two daily fingersticks, it eliminates the need for any additional fingerstick blood glucose testing in order to make treatment decisions,” Alberto Gutierrez, Ph.D., director of the office of in vitro diagnostics and radiological health in the FDA’s Center for Devices and Radiological Health, said in the FDA statement.
The FDA based its decision on data from two clinical studies of 130 adults and children aged 2 years and older with diabetes. No serious adverse events were reported during a 7-day period when system readings were compared with blood glucose meter values and lab glucose measures.
The action comes just a few months after the agency approved the MiniMed 670G by Medtronic, a hybrid closed-loop system designed to automatically monitor glucose and deliver appropriate basal insulin doses in patients aged 14 years and older. Medtronic is currently evaluating the safety and efficacy of the device in children aged 7-13 years.
People with diabetes have come a step closer to a life without multiple daily finger sticks. The with diabetes, the Food and Drug Administration announced .
“Although this system still requires calibration with two daily fingersticks, it eliminates the need for any additional fingerstick blood glucose testing in order to make treatment decisions,” Alberto Gutierrez, Ph.D., director of the office of in vitro diagnostics and radiological health in the FDA’s Center for Devices and Radiological Health, said in the FDA statement.
The FDA based its decision on data from two clinical studies of 130 adults and children aged 2 years and older with diabetes. No serious adverse events were reported during a 7-day period when system readings were compared with blood glucose meter values and lab glucose measures.
The action comes just a few months after the agency approved the MiniMed 670G by Medtronic, a hybrid closed-loop system designed to automatically monitor glucose and deliver appropriate basal insulin doses in patients aged 14 years and older. Medtronic is currently evaluating the safety and efficacy of the device in children aged 7-13 years.
People with diabetes have come a step closer to a life without multiple daily finger sticks. The with diabetes, the Food and Drug Administration announced .
“Although this system still requires calibration with two daily fingersticks, it eliminates the need for any additional fingerstick blood glucose testing in order to make treatment decisions,” Alberto Gutierrez, Ph.D., director of the office of in vitro diagnostics and radiological health in the FDA’s Center for Devices and Radiological Health, said in the FDA statement.
The FDA based its decision on data from two clinical studies of 130 adults and children aged 2 years and older with diabetes. No serious adverse events were reported during a 7-day period when system readings were compared with blood glucose meter values and lab glucose measures.
The action comes just a few months after the agency approved the MiniMed 670G by Medtronic, a hybrid closed-loop system designed to automatically monitor glucose and deliver appropriate basal insulin doses in patients aged 14 years and older. Medtronic is currently evaluating the safety and efficacy of the device in children aged 7-13 years.
FDA eases mental health warnings in smoking cessation drugs’ labels
Labels on two smoking cessation treatments will offer less severe warnings for mental health risk potentials in people with no history of psychiatric disorders, the Food and Drug Administration has announced.
Varenicline (Chantix) will no longer include a boxed warning for serious mental health side effects. The label for bupropion (Zyban) will still include a boxed warning, but language describing the potential for serious psychiatric adverse events will no longer appear within it. Updates will also be made to both labels to describe side effects on mood, behavior, or thinking.
In addition, varenicline’s label will reflect trial data showing its superior efficacy, compared with oral bupropion or nicotine patch. Although a patient medication guide will still be included with each prescription, the risk evaluation and mitigation strategy that prompted the guide will no longer be in place.
Earlier this year, two FDA advisory committees voted in favor of updating varenicline’s label, based on data from a randomized, controlled trial of more than 8,000 smokers, half of whom had a history of psychiatric disorders.
The trial showed no clinically significant difference in risk of adverse events across the smoking cessation treatments varenicline, bupropion, nicotine patch, or placebo study arms, although the risk was higher in the psychiatric cohorts in each.
Overall, 2% of those without a history of mental illness experienced neuropsychiatric adverse events, compared with between 5% and 7% of those with such a history.
The trial was cosponsored by Pfizer, maker of Chantix, and GlaxoSmithKline, maker of Zyban.
The FDA approved varenicline for smoking cessation in 2006 and approved bupropion, which also is indicated to treat depression and seasonal affective disorder, in 1997. After numerous postmarketing reports of increased incidents of psychiatric disorders occurring in smokers who used either drug, the agency added the boxed warning to each in 2009.
FDA officials advised clinicians to guard against changes in mental health status in smokers using these therapies. However, “the results of the trial confirm that the benefits of stopping smoking outweigh the risks of these medicines,” they noted.
On Twitter @whitneymcknight
Labels on two smoking cessation treatments will offer less severe warnings for mental health risk potentials in people with no history of psychiatric disorders, the Food and Drug Administration has announced.
Varenicline (Chantix) will no longer include a boxed warning for serious mental health side effects. The label for bupropion (Zyban) will still include a boxed warning, but language describing the potential for serious psychiatric adverse events will no longer appear within it. Updates will also be made to both labels to describe side effects on mood, behavior, or thinking.
In addition, varenicline’s label will reflect trial data showing its superior efficacy, compared with oral bupropion or nicotine patch. Although a patient medication guide will still be included with each prescription, the risk evaluation and mitigation strategy that prompted the guide will no longer be in place.
Earlier this year, two FDA advisory committees voted in favor of updating varenicline’s label, based on data from a randomized, controlled trial of more than 8,000 smokers, half of whom had a history of psychiatric disorders.
The trial showed no clinically significant difference in risk of adverse events across the smoking cessation treatments varenicline, bupropion, nicotine patch, or placebo study arms, although the risk was higher in the psychiatric cohorts in each.
Overall, 2% of those without a history of mental illness experienced neuropsychiatric adverse events, compared with between 5% and 7% of those with such a history.
The trial was cosponsored by Pfizer, maker of Chantix, and GlaxoSmithKline, maker of Zyban.
The FDA approved varenicline for smoking cessation in 2006 and approved bupropion, which also is indicated to treat depression and seasonal affective disorder, in 1997. After numerous postmarketing reports of increased incidents of psychiatric disorders occurring in smokers who used either drug, the agency added the boxed warning to each in 2009.
FDA officials advised clinicians to guard against changes in mental health status in smokers using these therapies. However, “the results of the trial confirm that the benefits of stopping smoking outweigh the risks of these medicines,” they noted.
On Twitter @whitneymcknight
Labels on two smoking cessation treatments will offer less severe warnings for mental health risk potentials in people with no history of psychiatric disorders, the Food and Drug Administration has announced.
Varenicline (Chantix) will no longer include a boxed warning for serious mental health side effects. The label for bupropion (Zyban) will still include a boxed warning, but language describing the potential for serious psychiatric adverse events will no longer appear within it. Updates will also be made to both labels to describe side effects on mood, behavior, or thinking.
In addition, varenicline’s label will reflect trial data showing its superior efficacy, compared with oral bupropion or nicotine patch. Although a patient medication guide will still be included with each prescription, the risk evaluation and mitigation strategy that prompted the guide will no longer be in place.
Earlier this year, two FDA advisory committees voted in favor of updating varenicline’s label, based on data from a randomized, controlled trial of more than 8,000 smokers, half of whom had a history of psychiatric disorders.
The trial showed no clinically significant difference in risk of adverse events across the smoking cessation treatments varenicline, bupropion, nicotine patch, or placebo study arms, although the risk was higher in the psychiatric cohorts in each.
Overall, 2% of those without a history of mental illness experienced neuropsychiatric adverse events, compared with between 5% and 7% of those with such a history.
The trial was cosponsored by Pfizer, maker of Chantix, and GlaxoSmithKline, maker of Zyban.
The FDA approved varenicline for smoking cessation in 2006 and approved bupropion, which also is indicated to treat depression and seasonal affective disorder, in 1997. After numerous postmarketing reports of increased incidents of psychiatric disorders occurring in smokers who used either drug, the agency added the boxed warning to each in 2009.
FDA officials advised clinicians to guard against changes in mental health status in smokers using these therapies. However, “the results of the trial confirm that the benefits of stopping smoking outweigh the risks of these medicines,” they noted.
On Twitter @whitneymcknight