User login
Latuda receives FDA approval for adolescent schizophrenia treatment
Lurasidone HCl, marketed as Latuda, has received approval from the Food and Drug Administration for the treatment of schizophrenia in adolescents aged 13-17 years old, according to a press release from Sunovion Pharmaceuticals.
FDA approval of Latuda is based on results of a 6-week study in which adolescents with schizophrenia received either 40 mg of lurasidone per day, 80 mg per day, or a placebo. Patients who received lurasidone HCI showed statistical and clinical improvements in schizophrenia symptoms, compared with the placebo group. The drug was well tolerated with no significant adverse events reported.
“The impact on development and poor prognosis frequently associated with schizophrenia that begins in adolescence underscores the need for treatment that is both well tolerated and effective,” Robert L. Findling, MD, study investigator, and director of child and adolescent psychiatry at Johns Hopkins University, Baltimore, said in the press release. “The availability of Latuda provides health care providers with an important new option for helping adolescents with this illness that is chronic and severely disabling,” said Dr. Findling, also vice president of psychiatric services and research at the Kennedy Krieger Institute, Baltimore.
Find the full press release on the Sunovion Pharmaceuticals website.
Lurasidone HCl, marketed as Latuda, has received approval from the Food and Drug Administration for the treatment of schizophrenia in adolescents aged 13-17 years old, according to a press release from Sunovion Pharmaceuticals.
FDA approval of Latuda is based on results of a 6-week study in which adolescents with schizophrenia received either 40 mg of lurasidone per day, 80 mg per day, or a placebo. Patients who received lurasidone HCI showed statistical and clinical improvements in schizophrenia symptoms, compared with the placebo group. The drug was well tolerated with no significant adverse events reported.
“The impact on development and poor prognosis frequently associated with schizophrenia that begins in adolescence underscores the need for treatment that is both well tolerated and effective,” Robert L. Findling, MD, study investigator, and director of child and adolescent psychiatry at Johns Hopkins University, Baltimore, said in the press release. “The availability of Latuda provides health care providers with an important new option for helping adolescents with this illness that is chronic and severely disabling,” said Dr. Findling, also vice president of psychiatric services and research at the Kennedy Krieger Institute, Baltimore.
Find the full press release on the Sunovion Pharmaceuticals website.
Lurasidone HCl, marketed as Latuda, has received approval from the Food and Drug Administration for the treatment of schizophrenia in adolescents aged 13-17 years old, according to a press release from Sunovion Pharmaceuticals.
FDA approval of Latuda is based on results of a 6-week study in which adolescents with schizophrenia received either 40 mg of lurasidone per day, 80 mg per day, or a placebo. Patients who received lurasidone HCI showed statistical and clinical improvements in schizophrenia symptoms, compared with the placebo group. The drug was well tolerated with no significant adverse events reported.
“The impact on development and poor prognosis frequently associated with schizophrenia that begins in adolescence underscores the need for treatment that is both well tolerated and effective,” Robert L. Findling, MD, study investigator, and director of child and adolescent psychiatry at Johns Hopkins University, Baltimore, said in the press release. “The availability of Latuda provides health care providers with an important new option for helping adolescents with this illness that is chronic and severely disabling,” said Dr. Findling, also vice president of psychiatric services and research at the Kennedy Krieger Institute, Baltimore.
Find the full press release on the Sunovion Pharmaceuticals website.
Increase brings flu activity back to seasonal high
for the week ending Jan. 21, compared with three states the week before, according to the Centers for Disease Control and Prevention.
Alabama, Kansas, New Jersey, Oklahoma, and South Carolina were at level 10 on the CDC’s 1-10 scale of ILI activity, with Oklahoma reaching that level for the third consecutive week. Georgia (level 9) and Louisiana, Minnesota, Missouri, and Tennessee (level 8) were also in the “high” range, the CDC reported.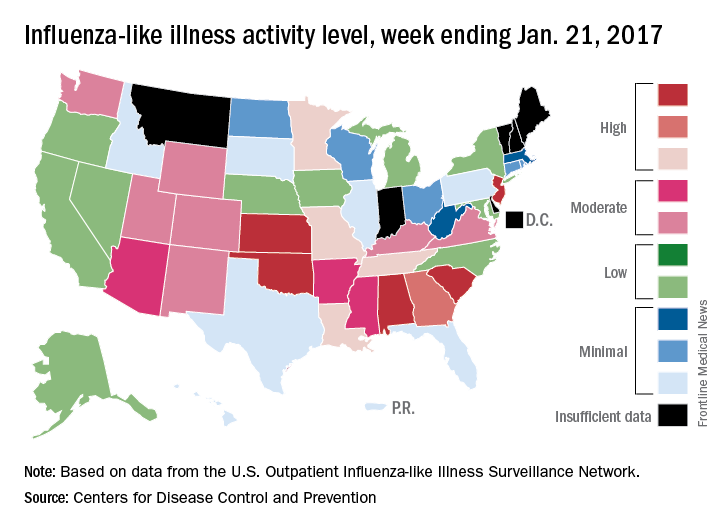
Three flu-related pediatric deaths were reported for the week, although two occurred during the week ending Jan. 14. The two earlier deaths were associated with an influenza A (H3) virus, and the more recent death was associated with an influenza B virus. For the 2016-2017 season so far, there have been a total of eight pediatric deaths, the CDC said.
for the week ending Jan. 21, compared with three states the week before, according to the Centers for Disease Control and Prevention.
Alabama, Kansas, New Jersey, Oklahoma, and South Carolina were at level 10 on the CDC’s 1-10 scale of ILI activity, with Oklahoma reaching that level for the third consecutive week. Georgia (level 9) and Louisiana, Minnesota, Missouri, and Tennessee (level 8) were also in the “high” range, the CDC reported.
Three flu-related pediatric deaths were reported for the week, although two occurred during the week ending Jan. 14. The two earlier deaths were associated with an influenza A (H3) virus, and the more recent death was associated with an influenza B virus. For the 2016-2017 season so far, there have been a total of eight pediatric deaths, the CDC said.
for the week ending Jan. 21, compared with three states the week before, according to the Centers for Disease Control and Prevention.
Alabama, Kansas, New Jersey, Oklahoma, and South Carolina were at level 10 on the CDC’s 1-10 scale of ILI activity, with Oklahoma reaching that level for the third consecutive week. Georgia (level 9) and Louisiana, Minnesota, Missouri, and Tennessee (level 8) were also in the “high” range, the CDC reported.
Three flu-related pediatric deaths were reported for the week, although two occurred during the week ending Jan. 14. The two earlier deaths were associated with an influenza A (H3) virus, and the more recent death was associated with an influenza B virus. For the 2016-2017 season so far, there have been a total of eight pediatric deaths, the CDC said.
Second dose rates of meningococcal vaccines low among HIV-positive men who have sex with men
ATLANTA – HIV-positive men who have sex with men should be getting vaccinated against invasive meningococcal disease twice, but an alarming majority are only getting vaccinated once, according to a new study presented at a conference on STD prevention sponsored the Centers for Disease Control and Prevention.
“This analysis underscores the need for active patient recall in order to maximize return for second dose among HIV-infected [men who have sex with men], although [that] may be resource-intensive,” said Kelly Jamison, MPH, of New York City’s department of health and mental hygiene.
Ms. Jamison and her coinvestigators examined medical record data of HIV-infected men who have sex with men who visited New York City STD clinics between Oct. 5, 2012 and Dec. 31, 2014, looking for individuals who received their first meningococcal vaccinations during that time period. The primary endpoint was to find the rate at which individuals who received the first vaccination came back within 1 year (Dec. 31, 2015) to receive a second vaccination.
The study was prompted by the invasive meningococcal disease (IMD) outbreak that New York City experienced from 2010-2013, in which 22 cases were identified in men who have sex with men, of which 55% involved men who were HIV-infected. All IMD cases involved serotype C, with a case fatality rate that was three times what public health officials anticipated at the time.
Because of this, the city launched a meningitis vaccination campaign. Vaccination was recommended for all men who have sex with men who were residents of New York City and had high-risk sexual exposure after Sept. 1, 2012. In early October, STD clinics around the city began offering free MCV4 vaccines. By late November, the recommendations were updated to include men who have sex with men who lived in specific parts of Brooklyn and had experienced high-risk sexual exposure after Sept. 1. In March 2013, the recommendations were further updated to state that all HIV-infected men who have sex with men and all such men with high-risk sexual exposure should be vaccinated. In August 2013, after the outbreak was over, the recommendations were updated one last time to state that they were recommendations for “ongoing vaccination.”
“A single dose of MCV4 is not sufficient for HIV-infected persons, so a second dose is recommended to occur 8 weeks after the first dose, and in order to increase two-dose coverage among HIV-infected MSM, STD clinicians provided the date for person to return for their second dose on a vaccine card given to patients at time of their first dose,” Ms. Jamison explained.
In total, 1,212 individuals were included in study. Over the course of the study period, only 322 (26.6%) returned within 1 year for a second vaccination. In terms of individual years, 2012 experienced the highest rate of second vaccination returns, at 38.6% (P less than .001). Of the 322 who received the second vaccination, 144 (44.7%) came to the STD clinic specifically for the second dose, 69 (21.4%) asked for the second vaccination along with other STD services, and 109 (33.9%) were “opportunistically vaccinated while presenting for other services.”
Older men who have sex with men were more likely to return for their second vaccination, as only 63 (18%) of those who did were under the age of 30. Those aged between 30 and 39 years numbered 80 (23%), those between 40 and 49 years numbered 102 (33%), and those aged 50 years or older numbered 77 (40%), meaning that older men were two to three times more likely to get that second dose (P less than .001).
“We did see suboptimal return for second doses, but this may be an underestimate, [because] we were unable to capture second doses received at non-STD clinic providers,” Ms. Jamison noted.
Ms. Jamison did not report any financial disclosures for this study.
ATLANTA – HIV-positive men who have sex with men should be getting vaccinated against invasive meningococcal disease twice, but an alarming majority are only getting vaccinated once, according to a new study presented at a conference on STD prevention sponsored the Centers for Disease Control and Prevention.
“This analysis underscores the need for active patient recall in order to maximize return for second dose among HIV-infected [men who have sex with men], although [that] may be resource-intensive,” said Kelly Jamison, MPH, of New York City’s department of health and mental hygiene.
Ms. Jamison and her coinvestigators examined medical record data of HIV-infected men who have sex with men who visited New York City STD clinics between Oct. 5, 2012 and Dec. 31, 2014, looking for individuals who received their first meningococcal vaccinations during that time period. The primary endpoint was to find the rate at which individuals who received the first vaccination came back within 1 year (Dec. 31, 2015) to receive a second vaccination.
The study was prompted by the invasive meningococcal disease (IMD) outbreak that New York City experienced from 2010-2013, in which 22 cases were identified in men who have sex with men, of which 55% involved men who were HIV-infected. All IMD cases involved serotype C, with a case fatality rate that was three times what public health officials anticipated at the time.
Because of this, the city launched a meningitis vaccination campaign. Vaccination was recommended for all men who have sex with men who were residents of New York City and had high-risk sexual exposure after Sept. 1, 2012. In early October, STD clinics around the city began offering free MCV4 vaccines. By late November, the recommendations were updated to include men who have sex with men who lived in specific parts of Brooklyn and had experienced high-risk sexual exposure after Sept. 1. In March 2013, the recommendations were further updated to state that all HIV-infected men who have sex with men and all such men with high-risk sexual exposure should be vaccinated. In August 2013, after the outbreak was over, the recommendations were updated one last time to state that they were recommendations for “ongoing vaccination.”
“A single dose of MCV4 is not sufficient for HIV-infected persons, so a second dose is recommended to occur 8 weeks after the first dose, and in order to increase two-dose coverage among HIV-infected MSM, STD clinicians provided the date for person to return for their second dose on a vaccine card given to patients at time of their first dose,” Ms. Jamison explained.
In total, 1,212 individuals were included in study. Over the course of the study period, only 322 (26.6%) returned within 1 year for a second vaccination. In terms of individual years, 2012 experienced the highest rate of second vaccination returns, at 38.6% (P less than .001). Of the 322 who received the second vaccination, 144 (44.7%) came to the STD clinic specifically for the second dose, 69 (21.4%) asked for the second vaccination along with other STD services, and 109 (33.9%) were “opportunistically vaccinated while presenting for other services.”
Older men who have sex with men were more likely to return for their second vaccination, as only 63 (18%) of those who did were under the age of 30. Those aged between 30 and 39 years numbered 80 (23%), those between 40 and 49 years numbered 102 (33%), and those aged 50 years or older numbered 77 (40%), meaning that older men were two to three times more likely to get that second dose (P less than .001).
“We did see suboptimal return for second doses, but this may be an underestimate, [because] we were unable to capture second doses received at non-STD clinic providers,” Ms. Jamison noted.
Ms. Jamison did not report any financial disclosures for this study.
ATLANTA – HIV-positive men who have sex with men should be getting vaccinated against invasive meningococcal disease twice, but an alarming majority are only getting vaccinated once, according to a new study presented at a conference on STD prevention sponsored the Centers for Disease Control and Prevention.
“This analysis underscores the need for active patient recall in order to maximize return for second dose among HIV-infected [men who have sex with men], although [that] may be resource-intensive,” said Kelly Jamison, MPH, of New York City’s department of health and mental hygiene.
Ms. Jamison and her coinvestigators examined medical record data of HIV-infected men who have sex with men who visited New York City STD clinics between Oct. 5, 2012 and Dec. 31, 2014, looking for individuals who received their first meningococcal vaccinations during that time period. The primary endpoint was to find the rate at which individuals who received the first vaccination came back within 1 year (Dec. 31, 2015) to receive a second vaccination.
The study was prompted by the invasive meningococcal disease (IMD) outbreak that New York City experienced from 2010-2013, in which 22 cases were identified in men who have sex with men, of which 55% involved men who were HIV-infected. All IMD cases involved serotype C, with a case fatality rate that was three times what public health officials anticipated at the time.
Because of this, the city launched a meningitis vaccination campaign. Vaccination was recommended for all men who have sex with men who were residents of New York City and had high-risk sexual exposure after Sept. 1, 2012. In early October, STD clinics around the city began offering free MCV4 vaccines. By late November, the recommendations were updated to include men who have sex with men who lived in specific parts of Brooklyn and had experienced high-risk sexual exposure after Sept. 1. In March 2013, the recommendations were further updated to state that all HIV-infected men who have sex with men and all such men with high-risk sexual exposure should be vaccinated. In August 2013, after the outbreak was over, the recommendations were updated one last time to state that they were recommendations for “ongoing vaccination.”
“A single dose of MCV4 is not sufficient for HIV-infected persons, so a second dose is recommended to occur 8 weeks after the first dose, and in order to increase two-dose coverage among HIV-infected MSM, STD clinicians provided the date for person to return for their second dose on a vaccine card given to patients at time of their first dose,” Ms. Jamison explained.
In total, 1,212 individuals were included in study. Over the course of the study period, only 322 (26.6%) returned within 1 year for a second vaccination. In terms of individual years, 2012 experienced the highest rate of second vaccination returns, at 38.6% (P less than .001). Of the 322 who received the second vaccination, 144 (44.7%) came to the STD clinic specifically for the second dose, 69 (21.4%) asked for the second vaccination along with other STD services, and 109 (33.9%) were “opportunistically vaccinated while presenting for other services.”
Older men who have sex with men were more likely to return for their second vaccination, as only 63 (18%) of those who did were under the age of 30. Those aged between 30 and 39 years numbered 80 (23%), those between 40 and 49 years numbered 102 (33%), and those aged 50 years or older numbered 77 (40%), meaning that older men were two to three times more likely to get that second dose (P less than .001).
“We did see suboptimal return for second doses, but this may be an underestimate, [because] we were unable to capture second doses received at non-STD clinic providers,” Ms. Jamison noted.
Ms. Jamison did not report any financial disclosures for this study.
AT THE 2016 STD PREVENTION CONFERENCE
Key clinical point:
Major finding: Only 26.6% (322 of 1,212) of men who have sex with men received a second dose within a year of receiving their first, with older men who have sex with men 2-3 times more likely to get the second dose than younger men who have sex with men.
Data source: Retrospective analysis of 1,212 men who have sex with men who visited New York City STD clinics from 2012-2015.
Disclosures: Ms. Jamison did not report any financial disclosures.
West Nile virus accounted for 95% of domestic arboviral disease in 2015
West Nile virus was the most common cause of domestically acquired arboviral disease in the United States in 2015, according to a report from the Centers for Disease Control and Prevention.
A total of 2,282 cases of arboviral disease were reported to the CDC in 2015. Of those, 2,175 cases were caused by the West Nile virus. Of the patients with WNV, 1,616 were hospitalized because of the disease, and 146 died. Neuroinvasive WNV, which occurred in 1,455 cases, accounted for 1,382 of 1,616 WNV hospitalizations and 142 of 146 deaths.
Of the 107 non-WNV arbovirus cases reported to the CDC, 55 were La Crosse virus, 23 were St. Louis encephalitis, 11 were Jamestown Canyon virus, 7 were Powassan virus, and 6 were eastern equine encephalitis. In addition to La Crosse and Jamestown Canyon, 4 cases of additional California serogroup viruses were reported, as was 1 case of Cache Valley virus.
“Health care providers should consider arboviral infections in the differential diagnosis of cases of aseptic meningitis and encephalitis, obtain appropriate specimens for laboratory testing, and promptly report cases to public health authorities. Because human vaccines against domestic arboviruses are not available, prevention depends on community and household efforts to reduce vector populations, personal protective measures to decrease exposure to mosquitoes and ticks, and screening of blood donors,” the CDC investigators concluded.
Find the full report in the MMWR (doi: 10.15585/mmwr.mm6602a3).
West Nile virus was the most common cause of domestically acquired arboviral disease in the United States in 2015, according to a report from the Centers for Disease Control and Prevention.
A total of 2,282 cases of arboviral disease were reported to the CDC in 2015. Of those, 2,175 cases were caused by the West Nile virus. Of the patients with WNV, 1,616 were hospitalized because of the disease, and 146 died. Neuroinvasive WNV, which occurred in 1,455 cases, accounted for 1,382 of 1,616 WNV hospitalizations and 142 of 146 deaths.
Of the 107 non-WNV arbovirus cases reported to the CDC, 55 were La Crosse virus, 23 were St. Louis encephalitis, 11 were Jamestown Canyon virus, 7 were Powassan virus, and 6 were eastern equine encephalitis. In addition to La Crosse and Jamestown Canyon, 4 cases of additional California serogroup viruses were reported, as was 1 case of Cache Valley virus.
“Health care providers should consider arboviral infections in the differential diagnosis of cases of aseptic meningitis and encephalitis, obtain appropriate specimens for laboratory testing, and promptly report cases to public health authorities. Because human vaccines against domestic arboviruses are not available, prevention depends on community and household efforts to reduce vector populations, personal protective measures to decrease exposure to mosquitoes and ticks, and screening of blood donors,” the CDC investigators concluded.
Find the full report in the MMWR (doi: 10.15585/mmwr.mm6602a3).
West Nile virus was the most common cause of domestically acquired arboviral disease in the United States in 2015, according to a report from the Centers for Disease Control and Prevention.
A total of 2,282 cases of arboviral disease were reported to the CDC in 2015. Of those, 2,175 cases were caused by the West Nile virus. Of the patients with WNV, 1,616 were hospitalized because of the disease, and 146 died. Neuroinvasive WNV, which occurred in 1,455 cases, accounted for 1,382 of 1,616 WNV hospitalizations and 142 of 146 deaths.
Of the 107 non-WNV arbovirus cases reported to the CDC, 55 were La Crosse virus, 23 were St. Louis encephalitis, 11 were Jamestown Canyon virus, 7 were Powassan virus, and 6 were eastern equine encephalitis. In addition to La Crosse and Jamestown Canyon, 4 cases of additional California serogroup viruses were reported, as was 1 case of Cache Valley virus.
“Health care providers should consider arboviral infections in the differential diagnosis of cases of aseptic meningitis and encephalitis, obtain appropriate specimens for laboratory testing, and promptly report cases to public health authorities. Because human vaccines against domestic arboviruses are not available, prevention depends on community and household efforts to reduce vector populations, personal protective measures to decrease exposure to mosquitoes and ticks, and screening of blood donors,” the CDC investigators concluded.
Find the full report in the MMWR (doi: 10.15585/mmwr.mm6602a3).
FROM MORBIDITY AND MORTALITY REPORT
FDA approves Trulance for chronic idiopathic constipation
Trulance, a once-daily oral medication for chronic idiopathic constipation, has been approved by the Food and Drug Administration for adult patients as of Jan. 19.
Manufactured by Synergy Pharmaceuticals, Trulance (plecanatide) stimulates intestinal fluid secretion in the upper GI tract. Its efficacy and safety were determined to be sufficient in two 12-week placebo-controlled trials (NCT01982240 and NCT02122471). Participants taking Trulance were more likely than were those taking placebo to have improved bowel function and stool.
The FDA’s statement noted that “an estimated 42 million people are affected by constipation. Chronic idiopathic constipation is a diagnosis given to those who experience persistent constipation and for whom there is no structural or biochemical explanation.”
Trulance, a once-daily oral medication for chronic idiopathic constipation, has been approved by the Food and Drug Administration for adult patients as of Jan. 19.
Manufactured by Synergy Pharmaceuticals, Trulance (plecanatide) stimulates intestinal fluid secretion in the upper GI tract. Its efficacy and safety were determined to be sufficient in two 12-week placebo-controlled trials (NCT01982240 and NCT02122471). Participants taking Trulance were more likely than were those taking placebo to have improved bowel function and stool.
The FDA’s statement noted that “an estimated 42 million people are affected by constipation. Chronic idiopathic constipation is a diagnosis given to those who experience persistent constipation and for whom there is no structural or biochemical explanation.”
Trulance, a once-daily oral medication for chronic idiopathic constipation, has been approved by the Food and Drug Administration for adult patients as of Jan. 19.
Manufactured by Synergy Pharmaceuticals, Trulance (plecanatide) stimulates intestinal fluid secretion in the upper GI tract. Its efficacy and safety were determined to be sufficient in two 12-week placebo-controlled trials (NCT01982240 and NCT02122471). Participants taking Trulance were more likely than were those taking placebo to have improved bowel function and stool.
The FDA’s statement noted that “an estimated 42 million people are affected by constipation. Chronic idiopathic constipation is a diagnosis given to those who experience persistent constipation and for whom there is no structural or biochemical explanation.”
FDA approves topical oxymetazoline for rosacea
A topical cream containing the vasoconstrictor oxymetazoline has been approved by the Food and Drug Administration to treat symptoms of rosacea, its manufacturer announced.
Oxymetazoline hydrochloride cream 1%, which will be marketed as Rhofade by Allergan, is indicated for the treatment of “persistent facial erythema associated with rosacea in adults.” While nasal sprays containing a lower concentration of oxymetazoline HCl, an alpha1A-adrenoceptor agonist, have been used off label for a decade, this is the first time this ingredient has been harnessed to formulate an approved rosacea treatment.
Safety results from three pooled trials showed 2% of patients in the active treatment arms (489 people) had treatment-site dermatitis, and 1% had worsening of rosacea symptoms, pruritus, or pain. The vehicle cream groups (483 people) experienced similar rates of pruritus but negligible rates of other adverse effects, according to the prescribing information.
Brimonidine (Mirvaso) is another topical treatment approved by the FDA for treating rosacea, and its active ingredient is also an alpha-adrenergic agonist that works on the cutaneous microvasculature. However, there are differences in the two agents’ activity. Oxymetazoline acts on alpha1A receptors and brimonidine on alpha2 receptors. There have been reports of rebound erythema more severe than at baseline with brimonidine, and its manufacturer, Galderma, acknowledges the phenomenon in patient labeling.
When Allergan announced the FDA application for oxymetazoline in May 2016, it issued a press statement, describing oxymetazoline as a “sympathomimetic agonist that is selective for the alpha1A adrenoceptor or over other alpha1 adrenoceptors and nonselective for the alpha2 adrenoceptors.”In a 1-year open label trial of oxymetazoline (440 people), 3% of patients had worsening inflammatory lesions of rosacea, according to the prescribing information for oxymetazoline HCl 1%.
A topical cream containing the vasoconstrictor oxymetazoline has been approved by the Food and Drug Administration to treat symptoms of rosacea, its manufacturer announced.
Oxymetazoline hydrochloride cream 1%, which will be marketed as Rhofade by Allergan, is indicated for the treatment of “persistent facial erythema associated with rosacea in adults.” While nasal sprays containing a lower concentration of oxymetazoline HCl, an alpha1A-adrenoceptor agonist, have been used off label for a decade, this is the first time this ingredient has been harnessed to formulate an approved rosacea treatment.
Safety results from three pooled trials showed 2% of patients in the active treatment arms (489 people) had treatment-site dermatitis, and 1% had worsening of rosacea symptoms, pruritus, or pain. The vehicle cream groups (483 people) experienced similar rates of pruritus but negligible rates of other adverse effects, according to the prescribing information.
Brimonidine (Mirvaso) is another topical treatment approved by the FDA for treating rosacea, and its active ingredient is also an alpha-adrenergic agonist that works on the cutaneous microvasculature. However, there are differences in the two agents’ activity. Oxymetazoline acts on alpha1A receptors and brimonidine on alpha2 receptors. There have been reports of rebound erythema more severe than at baseline with brimonidine, and its manufacturer, Galderma, acknowledges the phenomenon in patient labeling.
When Allergan announced the FDA application for oxymetazoline in May 2016, it issued a press statement, describing oxymetazoline as a “sympathomimetic agonist that is selective for the alpha1A adrenoceptor or over other alpha1 adrenoceptors and nonselective for the alpha2 adrenoceptors.”In a 1-year open label trial of oxymetazoline (440 people), 3% of patients had worsening inflammatory lesions of rosacea, according to the prescribing information for oxymetazoline HCl 1%.
A topical cream containing the vasoconstrictor oxymetazoline has been approved by the Food and Drug Administration to treat symptoms of rosacea, its manufacturer announced.
Oxymetazoline hydrochloride cream 1%, which will be marketed as Rhofade by Allergan, is indicated for the treatment of “persistent facial erythema associated with rosacea in adults.” While nasal sprays containing a lower concentration of oxymetazoline HCl, an alpha1A-adrenoceptor agonist, have been used off label for a decade, this is the first time this ingredient has been harnessed to formulate an approved rosacea treatment.
Safety results from three pooled trials showed 2% of patients in the active treatment arms (489 people) had treatment-site dermatitis, and 1% had worsening of rosacea symptoms, pruritus, or pain. The vehicle cream groups (483 people) experienced similar rates of pruritus but negligible rates of other adverse effects, according to the prescribing information.
Brimonidine (Mirvaso) is another topical treatment approved by the FDA for treating rosacea, and its active ingredient is also an alpha-adrenergic agonist that works on the cutaneous microvasculature. However, there are differences in the two agents’ activity. Oxymetazoline acts on alpha1A receptors and brimonidine on alpha2 receptors. There have been reports of rebound erythema more severe than at baseline with brimonidine, and its manufacturer, Galderma, acknowledges the phenomenon in patient labeling.
When Allergan announced the FDA application for oxymetazoline in May 2016, it issued a press statement, describing oxymetazoline as a “sympathomimetic agonist that is selective for the alpha1A adrenoceptor or over other alpha1 adrenoceptors and nonselective for the alpha2 adrenoceptors.”In a 1-year open label trial of oxymetazoline (440 people), 3% of patients had worsening inflammatory lesions of rosacea, according to the prescribing information for oxymetazoline HCl 1%.
Flu activity up slightly, but still down from seasonal peak
After rising to a high point for the season in the last week of 2016, influenza activity dropped a bit in the first week of the new year but then rose again in the second week, according to the Centers for Disease Control and Prevention.
As measured by outpatient visits for influenza-like illness (ILI), activity slipped from 3.4% at the end of 2016 to 3.2% for the week ending Jan. 7 but then ticked up to 3.3% for the week ending Jan. 14, the CDC reported. The national baseline level of outpatient visits is 2.2% for ILI, which is defined as fever (temperature of 100° F or greater) and cough and/or sore throat.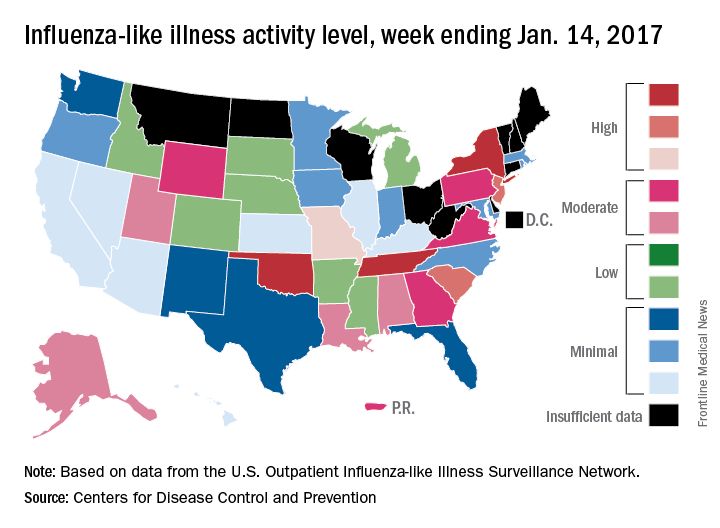
Two influenza-related pediatric deaths were reported for the week ending Jan. 14, although both occurred in earlier weeks: one during the week ending Dec. 10 and one during the week ending Jan. 7. So far for the 2016-2017 season, a total of five flu-related pediatric deaths have been reported, according to the CDC.
After rising to a high point for the season in the last week of 2016, influenza activity dropped a bit in the first week of the new year but then rose again in the second week, according to the Centers for Disease Control and Prevention.
As measured by outpatient visits for influenza-like illness (ILI), activity slipped from 3.4% at the end of 2016 to 3.2% for the week ending Jan. 7 but then ticked up to 3.3% for the week ending Jan. 14, the CDC reported. The national baseline level of outpatient visits is 2.2% for ILI, which is defined as fever (temperature of 100° F or greater) and cough and/or sore throat.
Two influenza-related pediatric deaths were reported for the week ending Jan. 14, although both occurred in earlier weeks: one during the week ending Dec. 10 and one during the week ending Jan. 7. So far for the 2016-2017 season, a total of five flu-related pediatric deaths have been reported, according to the CDC.
After rising to a high point for the season in the last week of 2016, influenza activity dropped a bit in the first week of the new year but then rose again in the second week, according to the Centers for Disease Control and Prevention.
As measured by outpatient visits for influenza-like illness (ILI), activity slipped from 3.4% at the end of 2016 to 3.2% for the week ending Jan. 7 but then ticked up to 3.3% for the week ending Jan. 14, the CDC reported. The national baseline level of outpatient visits is 2.2% for ILI, which is defined as fever (temperature of 100° F or greater) and cough and/or sore throat.
Two influenza-related pediatric deaths were reported for the week ending Jan. 14, although both occurred in earlier weeks: one during the week ending Dec. 10 and one during the week ending Jan. 7. So far for the 2016-2017 season, a total of five flu-related pediatric deaths have been reported, according to the CDC.
Zika virus slowdown continues
Zika activity is slowing as winter progresses, with less than a hundred new cases of pregnant women with laboratory evidence of infection reported over the 2 weeks ending Jan. 10, according to data from the Centers for Disease Control and Prevention.
The most recent CDC data show that, for the first time since early August, the majority of new cases of Zika infection among pregnant women were reported in the 50 states and the District of Columbia, compared with the U.S. territories. There were a total of 98 new cases, with 55 reported in the states and 43 in the U.S. territories.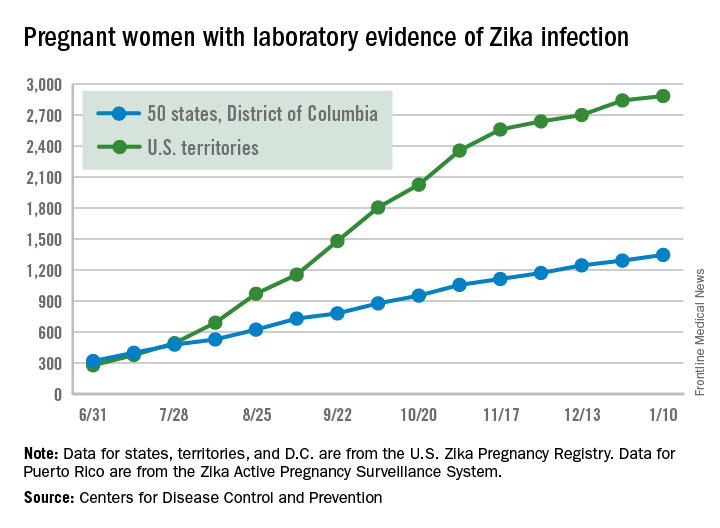
The total number of Zika-infected pregnant women in the United States is now 4,232 for 2016-2017. There have been 2,885 cases in the territories and 1,347 cases reported in the states/D.C. Among the cases in the states/D.C., 940 pregnancies have been completed, with Zika-related birth defects seen in 37 live-born infants and five pregnancy losses, the CDC said. The CDC is no longer reporting adverse pregnancy outcomes for the territories because Puerto Rico is not using the same inclusion criteria.
Zika cases among all Americans are still being reported weekly by the CDC, and the increase there has slowed as well: Total cases were up by 146 for the week ending Jan. 18, compared with 294 and 205 for each of the previous 2 weeks, according to CDC reports.
Zika-related birth defects reported by the CDC could include microcephaly, calcium deposits in the brain indicating possible brain damage, excess fluid in the brain cavities and surrounding the brain, absent or poorly formed brain structures, abnormal eye development, or other problems resulting from brain damage that affect nerves, muscles, and bones. The pregnancy losses encompass any miscarriage, stillbirth, and termination with evidence of birth defects.
Zika activity is slowing as winter progresses, with less than a hundred new cases of pregnant women with laboratory evidence of infection reported over the 2 weeks ending Jan. 10, according to data from the Centers for Disease Control and Prevention.
The most recent CDC data show that, for the first time since early August, the majority of new cases of Zika infection among pregnant women were reported in the 50 states and the District of Columbia, compared with the U.S. territories. There were a total of 98 new cases, with 55 reported in the states and 43 in the U.S. territories.
The total number of Zika-infected pregnant women in the United States is now 4,232 for 2016-2017. There have been 2,885 cases in the territories and 1,347 cases reported in the states/D.C. Among the cases in the states/D.C., 940 pregnancies have been completed, with Zika-related birth defects seen in 37 live-born infants and five pregnancy losses, the CDC said. The CDC is no longer reporting adverse pregnancy outcomes for the territories because Puerto Rico is not using the same inclusion criteria.
Zika cases among all Americans are still being reported weekly by the CDC, and the increase there has slowed as well: Total cases were up by 146 for the week ending Jan. 18, compared with 294 and 205 for each of the previous 2 weeks, according to CDC reports.
Zika-related birth defects reported by the CDC could include microcephaly, calcium deposits in the brain indicating possible brain damage, excess fluid in the brain cavities and surrounding the brain, absent or poorly formed brain structures, abnormal eye development, or other problems resulting from brain damage that affect nerves, muscles, and bones. The pregnancy losses encompass any miscarriage, stillbirth, and termination with evidence of birth defects.
Zika activity is slowing as winter progresses, with less than a hundred new cases of pregnant women with laboratory evidence of infection reported over the 2 weeks ending Jan. 10, according to data from the Centers for Disease Control and Prevention.
The most recent CDC data show that, for the first time since early August, the majority of new cases of Zika infection among pregnant women were reported in the 50 states and the District of Columbia, compared with the U.S. territories. There were a total of 98 new cases, with 55 reported in the states and 43 in the U.S. territories.
The total number of Zika-infected pregnant women in the United States is now 4,232 for 2016-2017. There have been 2,885 cases in the territories and 1,347 cases reported in the states/D.C. Among the cases in the states/D.C., 940 pregnancies have been completed, with Zika-related birth defects seen in 37 live-born infants and five pregnancy losses, the CDC said. The CDC is no longer reporting adverse pregnancy outcomes for the territories because Puerto Rico is not using the same inclusion criteria.
Zika cases among all Americans are still being reported weekly by the CDC, and the increase there has slowed as well: Total cases were up by 146 for the week ending Jan. 18, compared with 294 and 205 for each of the previous 2 weeks, according to CDC reports.
Zika-related birth defects reported by the CDC could include microcephaly, calcium deposits in the brain indicating possible brain damage, excess fluid in the brain cavities and surrounding the brain, absent or poorly formed brain structures, abnormal eye development, or other problems resulting from brain damage that affect nerves, muscles, and bones. The pregnancy losses encompass any miscarriage, stillbirth, and termination with evidence of birth defects.
FDA approves ibrutinib for refractory MZL
The Food and Drug Administration has approved ibrutinib for the treatment of patients with relapsed or refractory marginal zone lymphoma (MZL), the drug’s manufacturers report.
The approval marks the fifth indication for ibrutinib (Imbruvica) in just over 4 years, and ibrutinib is the first agent specifically approved for relapsed/refractory MZL, according to press releases issued by Janssen Biotech and Pharmacyclics, the two manufacturers that jointly developed and marketed the Bruton tyrosine kinase inhibitor.
After receiving various fast-track, breakthrough therapy, priority review, and accelerated approval designations from the FDA, ibrutinib was previously approved to treat mantle cell lymphoma; refractory chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL); CLL/SLL with 17p deletion; and Waldenstrom’s macroglobulinemia, another rare form of non-Hodgkin lymphoma. The MCL and MZL approvals are based on overall response rates, and full approval is likely to require additional confirmatory data.
The new indication is based on data from a phase II, open-label, single-arm manufacturer-sponsored study that showed a 46% overall response rate (95% confidence interval, 33.4-59.1) in a cohort of 63 MZL patients who had failed one or more prior therapies. Of these, 3.2% had a complete response and 42.9% had a partial response. The median duration of response was not reached (NR) (range, 16.7 months–NR), with median follow-up of 19.4 months. The median time to initial response was 4.5 months (2.3-16.4 months).
All three MZL subtypes were represented in the cohort, and ibrutinib appeared to be effective across subtypes. Thrombocytopenia, fatigue, anemia, diarrhea, bruising, and musculoskeletal pain were commonly reported adverse events.
hematologynews@frontlinemedcom.com
On Twitter @HematologyNews1
The Food and Drug Administration has approved ibrutinib for the treatment of patients with relapsed or refractory marginal zone lymphoma (MZL), the drug’s manufacturers report.
The approval marks the fifth indication for ibrutinib (Imbruvica) in just over 4 years, and ibrutinib is the first agent specifically approved for relapsed/refractory MZL, according to press releases issued by Janssen Biotech and Pharmacyclics, the two manufacturers that jointly developed and marketed the Bruton tyrosine kinase inhibitor.
After receiving various fast-track, breakthrough therapy, priority review, and accelerated approval designations from the FDA, ibrutinib was previously approved to treat mantle cell lymphoma; refractory chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL); CLL/SLL with 17p deletion; and Waldenstrom’s macroglobulinemia, another rare form of non-Hodgkin lymphoma. The MCL and MZL approvals are based on overall response rates, and full approval is likely to require additional confirmatory data.
The new indication is based on data from a phase II, open-label, single-arm manufacturer-sponsored study that showed a 46% overall response rate (95% confidence interval, 33.4-59.1) in a cohort of 63 MZL patients who had failed one or more prior therapies. Of these, 3.2% had a complete response and 42.9% had a partial response. The median duration of response was not reached (NR) (range, 16.7 months–NR), with median follow-up of 19.4 months. The median time to initial response was 4.5 months (2.3-16.4 months).
All three MZL subtypes were represented in the cohort, and ibrutinib appeared to be effective across subtypes. Thrombocytopenia, fatigue, anemia, diarrhea, bruising, and musculoskeletal pain were commonly reported adverse events.
hematologynews@frontlinemedcom.com
On Twitter @HematologyNews1
The Food and Drug Administration has approved ibrutinib for the treatment of patients with relapsed or refractory marginal zone lymphoma (MZL), the drug’s manufacturers report.
The approval marks the fifth indication for ibrutinib (Imbruvica) in just over 4 years, and ibrutinib is the first agent specifically approved for relapsed/refractory MZL, according to press releases issued by Janssen Biotech and Pharmacyclics, the two manufacturers that jointly developed and marketed the Bruton tyrosine kinase inhibitor.
After receiving various fast-track, breakthrough therapy, priority review, and accelerated approval designations from the FDA, ibrutinib was previously approved to treat mantle cell lymphoma; refractory chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL); CLL/SLL with 17p deletion; and Waldenstrom’s macroglobulinemia, another rare form of non-Hodgkin lymphoma. The MCL and MZL approvals are based on overall response rates, and full approval is likely to require additional confirmatory data.
The new indication is based on data from a phase II, open-label, single-arm manufacturer-sponsored study that showed a 46% overall response rate (95% confidence interval, 33.4-59.1) in a cohort of 63 MZL patients who had failed one or more prior therapies. Of these, 3.2% had a complete response and 42.9% had a partial response. The median duration of response was not reached (NR) (range, 16.7 months–NR), with median follow-up of 19.4 months. The median time to initial response was 4.5 months (2.3-16.4 months).
All three MZL subtypes were represented in the cohort, and ibrutinib appeared to be effective across subtypes. Thrombocytopenia, fatigue, anemia, diarrhea, bruising, and musculoskeletal pain were commonly reported adverse events.
hematologynews@frontlinemedcom.com
On Twitter @HematologyNews1
FDA, EPA clarify which fish pregnant women and young children should eat
The Food and Drug Administration and the Environmental Protection Agency have issued updated guidance about fish consumption for pregnant women and young children, clarifying which types of fish are recommended and what types of fish to avoid.
In guidance issued Jan. 18, the agencies sort 62 types of fish into three categories based on mercury level: best choices, good choices, and fish to avoid. They recommend that women who are pregnant, women who may become pregnant, breastfeeding mothers, and young children eat two to three servings of fish in the “best choices” category per week. Women and young children are advised to eat one serving per week of fish in the “good choices” category, according to the announcement. Fish in the “best choices” category make up nearly 90% of fish eaten in the United States, according to the FDA.
“Fish are an important source of protein and other nutrients for young children and women who are or may become pregnant or are breastfeeding,” Stephen Ostroff, MD, FDA’s deputy commissioner for Foods and Veterinary Medicine, said in a statement. “This advice clearly shows the great diversity of fish in the U.S. market that they can consume safely. This new, clear and concrete advice is an excellent tool for making safe and healthy choices when buying fish.”
The updated advice cautions pregnant women and others to avoid seven types of fish that generally have higher mercury levels. This includes tilefish from the Gulf of Mexico, shark; swordfish; orange roughy, bigeye tuna; marlin, and king mackerel. Meanwhile, recommended choices lower in mercury include such fish as shrimp, pollock, salmon, canned light tuna, tilapia, catfish, and cod.
Consumers are urged to check local advisories for fish caught recreationally and gauge their fish consumption based on any local and state advisories for those waters. If no information on fishing advisories is available, the FDA recommends eating just one fish meal a week from local waters and to avoid other fish that week.
agallegos@frontlinemedcom.com
On Twitter @legal_med
The Food and Drug Administration and the Environmental Protection Agency have issued updated guidance about fish consumption for pregnant women and young children, clarifying which types of fish are recommended and what types of fish to avoid.
In guidance issued Jan. 18, the agencies sort 62 types of fish into three categories based on mercury level: best choices, good choices, and fish to avoid. They recommend that women who are pregnant, women who may become pregnant, breastfeeding mothers, and young children eat two to three servings of fish in the “best choices” category per week. Women and young children are advised to eat one serving per week of fish in the “good choices” category, according to the announcement. Fish in the “best choices” category make up nearly 90% of fish eaten in the United States, according to the FDA.
“Fish are an important source of protein and other nutrients for young children and women who are or may become pregnant or are breastfeeding,” Stephen Ostroff, MD, FDA’s deputy commissioner for Foods and Veterinary Medicine, said in a statement. “This advice clearly shows the great diversity of fish in the U.S. market that they can consume safely. This new, clear and concrete advice is an excellent tool for making safe and healthy choices when buying fish.”
The updated advice cautions pregnant women and others to avoid seven types of fish that generally have higher mercury levels. This includes tilefish from the Gulf of Mexico, shark; swordfish; orange roughy, bigeye tuna; marlin, and king mackerel. Meanwhile, recommended choices lower in mercury include such fish as shrimp, pollock, salmon, canned light tuna, tilapia, catfish, and cod.
Consumers are urged to check local advisories for fish caught recreationally and gauge their fish consumption based on any local and state advisories for those waters. If no information on fishing advisories is available, the FDA recommends eating just one fish meal a week from local waters and to avoid other fish that week.
agallegos@frontlinemedcom.com
On Twitter @legal_med
The Food and Drug Administration and the Environmental Protection Agency have issued updated guidance about fish consumption for pregnant women and young children, clarifying which types of fish are recommended and what types of fish to avoid.
In guidance issued Jan. 18, the agencies sort 62 types of fish into three categories based on mercury level: best choices, good choices, and fish to avoid. They recommend that women who are pregnant, women who may become pregnant, breastfeeding mothers, and young children eat two to three servings of fish in the “best choices” category per week. Women and young children are advised to eat one serving per week of fish in the “good choices” category, according to the announcement. Fish in the “best choices” category make up nearly 90% of fish eaten in the United States, according to the FDA.
“Fish are an important source of protein and other nutrients for young children and women who are or may become pregnant or are breastfeeding,” Stephen Ostroff, MD, FDA’s deputy commissioner for Foods and Veterinary Medicine, said in a statement. “This advice clearly shows the great diversity of fish in the U.S. market that they can consume safely. This new, clear and concrete advice is an excellent tool for making safe and healthy choices when buying fish.”
The updated advice cautions pregnant women and others to avoid seven types of fish that generally have higher mercury levels. This includes tilefish from the Gulf of Mexico, shark; swordfish; orange roughy, bigeye tuna; marlin, and king mackerel. Meanwhile, recommended choices lower in mercury include such fish as shrimp, pollock, salmon, canned light tuna, tilapia, catfish, and cod.
Consumers are urged to check local advisories for fish caught recreationally and gauge their fish consumption based on any local and state advisories for those waters. If no information on fishing advisories is available, the FDA recommends eating just one fish meal a week from local waters and to avoid other fish that week.
agallegos@frontlinemedcom.com
On Twitter @legal_med