User login
MDedge conference coverage features onsite reporting of the latest study results and expert perspectives from leading researchers.
Newly approved myasthenia gravis drug shows sustained benefits in early responders
PHOENIX – , a new analyses show.
“The information [in the studies] is valuable in making clinical decisions in managing myasthenia gravis, which is a chronic autoimmune condition that requires long-term use of immunosuppressives,” said Xinli Du, MD, PhD, an assistant professor in the department of neurology at Virginia Commonwealth University in Richmond, who was not involved in the research.
“Compared with conventional immunosuppressants, which take 3-9 months to know if the patient will respond, this is definitely a game-changer,” she said.
The research was presented at the 2023 annual meeting of the American Association of Neuromuscular & Electrodiagnostic Medicine (AANEM).
FDA approval
Approved by the U.S. Food and Drug Administration in October, the targeted peptide inhibitor of complement component 5 represents the only once-daily self-administered subcutaneous injection for adult patients with acetylcholine receptor autoantibody–positive (AChR+) generalized myasthenia gravis.
The multicenter, phase 3, placebo-controlled RAISE trial demonstrated that zilucoplan was associated with significant improvement in myasthenia gravis–specific outcomes in adult patients with mild to severe AChR+ generalized myasthenia gravis.
Of note, approximately 40% of patients in the zilucoplan phase 2 and 3 clinical trials have a significant response as early as the first week of treatment. For the current post hoc analysis, first author Miriam Freimer, MD, and colleagues took a closer look at the longer-term outcomes in these patients in the ongoing RAISE-XT open-label extension study.
In these two double-blind studies, patients were randomly assigned to receive either daily subcutaneous injections of 0.3 mg/kg zilucoplan or placebo.
Among 93 patients receiving zilucoplan in the two studies, 40 (43%) were identified as early responders based on having at least a 3-point reduction from baseline on the Myasthenia Gravis Activities of Daily Living scale (MG-ADL) within 1 week of treatment, and 31 (33%) qualified based on having at least 5-point reductions in Quantitative Myasthenia Gravis (QMG), at week 1.
Of these early responders, more than 80% meeting the MG-ADL and 85% meeting QMG criteria continued to show a treatment response at each assessment through week 60 in the open-label RAISE-XT trial.
Furthermore, week 1 responders maintained their response for 88.1% of their total treatment time in the MG-ADL group and 88.8% of their total treatment time on treatment in the QMG group, representing a median zilucoplan treatment duration of 450 days.
Of note, the week 1 early responders had no significant differences, compared with the study’s overall population. Participants had a mean age of 49.6 years versus 52.9 years in the overall population. Approximately 40% of patients in both studies were men, and 60%-64% were disease class III as assessed by the Myasthenia Gravis Foundation of America criteria.
“It is very exciting to see such a high response of rapid responders. This means that some patients may be able to avoid steroids or be able to taper them faster than with other accepted treatments for myasthenia gravis,” said Dr. Freimer, director of the division of neuromuscular disorders and the codirector of the Myasthenia Gravis Clinic at the Ohio State University in Columbus.
Impact on fatigue
In a separate post hoc analysis of patients who entered RAISE-XT, the researchers evaluated long-term effects of zilucoplan on fatigue.
Improvements in fatigue were already apparent at the end of the RAISE trial, with the least squares mean (LSM) change from baseline in fatigue assessed after 12 weeks with the Neuro-QOL T-score being –6.26 for zilucoplan (n = 86) compared with an increase of 2.65 for placebo (n = 88; LSM difference, –3.61; nominal P = .0060).
Patients who received placebo in the RAISE trial were able to switch to zilucoplan in the RAISE-XT open-label trail, and among those who did, fatigue, as measured in the T-scores, improved significantly within 1 week of switching.
Fatigue further improved out to week 16 in terms of T-scores for the placebo-switch as well as zilucoplan groups, and the improvements in the scores were sustained through week 60 (mean change from RAISE baseline, –10.71 [n = 42] and –9.15 [n = 42], respectively).
“Fatigue is a challenge for patients with [generalized myasthenia gravis]. Zilucoplan significantly and clinically meaningfully improved myasthenic fatigue versus placebo during RAISE,” the investigators report.
“Further improvements were observed during RAISE-XT and were sustained up to 60 weeks of treatment,” they added.
Favorable safety profile
Zilucoplan continued to show a favorable safety profile in the RAISE-XT trial and was well tolerated in the long term.
The most common adverse reactions (10% or more) in patients with generalized myasthenia gravis were injection-site reactions, upper respiratory tract infection, and diarrhea.
Though additional therapies have also recently entered the market for generalized myasthenia gravis, they are either intravenous or subcutaneous infusions requiring a health care professional to administer them.
“This self-administered medication allows patients to be more independent and can even travel since it is not dependent on an infusion center,” Dr. Freimer noted.
However, similar to the other complement inhibitors, zilucoplan carries a risk for infection, particularly meningitis, Dr. Du noted. “Complement meningitis vaccination protocol and close monitoring for signs of infection are required,” she said.
However, she added, “the rapid action benefits shown as early as week 1 are impressive, and potentially open the door to use as adjunctive therapy in myasthenia gravis crisis.”
“I think zilucoplan definitely adds more excitement to the already active field of myasthenia gravis management,” she said.
The study was funded by UCB Pharma. Dr. Freimer has reports she has served as a paid consultant for argenx, UCB Pharma, and Alexion Pharmaceuticals and reports research support from the National Institutes of Health, UCB Pharma, Jansen Pharmaceuticals, Alnylam, Avidity, and Fulcrum. Dr. Du reports no disclosures.
A version of this article appeared on Medscape.com.
PHOENIX – , a new analyses show.
“The information [in the studies] is valuable in making clinical decisions in managing myasthenia gravis, which is a chronic autoimmune condition that requires long-term use of immunosuppressives,” said Xinli Du, MD, PhD, an assistant professor in the department of neurology at Virginia Commonwealth University in Richmond, who was not involved in the research.
“Compared with conventional immunosuppressants, which take 3-9 months to know if the patient will respond, this is definitely a game-changer,” she said.
The research was presented at the 2023 annual meeting of the American Association of Neuromuscular & Electrodiagnostic Medicine (AANEM).
FDA approval
Approved by the U.S. Food and Drug Administration in October, the targeted peptide inhibitor of complement component 5 represents the only once-daily self-administered subcutaneous injection for adult patients with acetylcholine receptor autoantibody–positive (AChR+) generalized myasthenia gravis.
The multicenter, phase 3, placebo-controlled RAISE trial demonstrated that zilucoplan was associated with significant improvement in myasthenia gravis–specific outcomes in adult patients with mild to severe AChR+ generalized myasthenia gravis.
Of note, approximately 40% of patients in the zilucoplan phase 2 and 3 clinical trials have a significant response as early as the first week of treatment. For the current post hoc analysis, first author Miriam Freimer, MD, and colleagues took a closer look at the longer-term outcomes in these patients in the ongoing RAISE-XT open-label extension study.
In these two double-blind studies, patients were randomly assigned to receive either daily subcutaneous injections of 0.3 mg/kg zilucoplan or placebo.
Among 93 patients receiving zilucoplan in the two studies, 40 (43%) were identified as early responders based on having at least a 3-point reduction from baseline on the Myasthenia Gravis Activities of Daily Living scale (MG-ADL) within 1 week of treatment, and 31 (33%) qualified based on having at least 5-point reductions in Quantitative Myasthenia Gravis (QMG), at week 1.
Of these early responders, more than 80% meeting the MG-ADL and 85% meeting QMG criteria continued to show a treatment response at each assessment through week 60 in the open-label RAISE-XT trial.
Furthermore, week 1 responders maintained their response for 88.1% of their total treatment time in the MG-ADL group and 88.8% of their total treatment time on treatment in the QMG group, representing a median zilucoplan treatment duration of 450 days.
Of note, the week 1 early responders had no significant differences, compared with the study’s overall population. Participants had a mean age of 49.6 years versus 52.9 years in the overall population. Approximately 40% of patients in both studies were men, and 60%-64% were disease class III as assessed by the Myasthenia Gravis Foundation of America criteria.
“It is very exciting to see such a high response of rapid responders. This means that some patients may be able to avoid steroids or be able to taper them faster than with other accepted treatments for myasthenia gravis,” said Dr. Freimer, director of the division of neuromuscular disorders and the codirector of the Myasthenia Gravis Clinic at the Ohio State University in Columbus.
Impact on fatigue
In a separate post hoc analysis of patients who entered RAISE-XT, the researchers evaluated long-term effects of zilucoplan on fatigue.
Improvements in fatigue were already apparent at the end of the RAISE trial, with the least squares mean (LSM) change from baseline in fatigue assessed after 12 weeks with the Neuro-QOL T-score being –6.26 for zilucoplan (n = 86) compared with an increase of 2.65 for placebo (n = 88; LSM difference, –3.61; nominal P = .0060).
Patients who received placebo in the RAISE trial were able to switch to zilucoplan in the RAISE-XT open-label trail, and among those who did, fatigue, as measured in the T-scores, improved significantly within 1 week of switching.
Fatigue further improved out to week 16 in terms of T-scores for the placebo-switch as well as zilucoplan groups, and the improvements in the scores were sustained through week 60 (mean change from RAISE baseline, –10.71 [n = 42] and –9.15 [n = 42], respectively).
“Fatigue is a challenge for patients with [generalized myasthenia gravis]. Zilucoplan significantly and clinically meaningfully improved myasthenic fatigue versus placebo during RAISE,” the investigators report.
“Further improvements were observed during RAISE-XT and were sustained up to 60 weeks of treatment,” they added.
Favorable safety profile
Zilucoplan continued to show a favorable safety profile in the RAISE-XT trial and was well tolerated in the long term.
The most common adverse reactions (10% or more) in patients with generalized myasthenia gravis were injection-site reactions, upper respiratory tract infection, and diarrhea.
Though additional therapies have also recently entered the market for generalized myasthenia gravis, they are either intravenous or subcutaneous infusions requiring a health care professional to administer them.
“This self-administered medication allows patients to be more independent and can even travel since it is not dependent on an infusion center,” Dr. Freimer noted.
However, similar to the other complement inhibitors, zilucoplan carries a risk for infection, particularly meningitis, Dr. Du noted. “Complement meningitis vaccination protocol and close monitoring for signs of infection are required,” she said.
However, she added, “the rapid action benefits shown as early as week 1 are impressive, and potentially open the door to use as adjunctive therapy in myasthenia gravis crisis.”
“I think zilucoplan definitely adds more excitement to the already active field of myasthenia gravis management,” she said.
The study was funded by UCB Pharma. Dr. Freimer has reports she has served as a paid consultant for argenx, UCB Pharma, and Alexion Pharmaceuticals and reports research support from the National Institutes of Health, UCB Pharma, Jansen Pharmaceuticals, Alnylam, Avidity, and Fulcrum. Dr. Du reports no disclosures.
A version of this article appeared on Medscape.com.
PHOENIX – , a new analyses show.
“The information [in the studies] is valuable in making clinical decisions in managing myasthenia gravis, which is a chronic autoimmune condition that requires long-term use of immunosuppressives,” said Xinli Du, MD, PhD, an assistant professor in the department of neurology at Virginia Commonwealth University in Richmond, who was not involved in the research.
“Compared with conventional immunosuppressants, which take 3-9 months to know if the patient will respond, this is definitely a game-changer,” she said.
The research was presented at the 2023 annual meeting of the American Association of Neuromuscular & Electrodiagnostic Medicine (AANEM).
FDA approval
Approved by the U.S. Food and Drug Administration in October, the targeted peptide inhibitor of complement component 5 represents the only once-daily self-administered subcutaneous injection for adult patients with acetylcholine receptor autoantibody–positive (AChR+) generalized myasthenia gravis.
The multicenter, phase 3, placebo-controlled RAISE trial demonstrated that zilucoplan was associated with significant improvement in myasthenia gravis–specific outcomes in adult patients with mild to severe AChR+ generalized myasthenia gravis.
Of note, approximately 40% of patients in the zilucoplan phase 2 and 3 clinical trials have a significant response as early as the first week of treatment. For the current post hoc analysis, first author Miriam Freimer, MD, and colleagues took a closer look at the longer-term outcomes in these patients in the ongoing RAISE-XT open-label extension study.
In these two double-blind studies, patients were randomly assigned to receive either daily subcutaneous injections of 0.3 mg/kg zilucoplan or placebo.
Among 93 patients receiving zilucoplan in the two studies, 40 (43%) were identified as early responders based on having at least a 3-point reduction from baseline on the Myasthenia Gravis Activities of Daily Living scale (MG-ADL) within 1 week of treatment, and 31 (33%) qualified based on having at least 5-point reductions in Quantitative Myasthenia Gravis (QMG), at week 1.
Of these early responders, more than 80% meeting the MG-ADL and 85% meeting QMG criteria continued to show a treatment response at each assessment through week 60 in the open-label RAISE-XT trial.
Furthermore, week 1 responders maintained their response for 88.1% of their total treatment time in the MG-ADL group and 88.8% of their total treatment time on treatment in the QMG group, representing a median zilucoplan treatment duration of 450 days.
Of note, the week 1 early responders had no significant differences, compared with the study’s overall population. Participants had a mean age of 49.6 years versus 52.9 years in the overall population. Approximately 40% of patients in both studies were men, and 60%-64% were disease class III as assessed by the Myasthenia Gravis Foundation of America criteria.
“It is very exciting to see such a high response of rapid responders. This means that some patients may be able to avoid steroids or be able to taper them faster than with other accepted treatments for myasthenia gravis,” said Dr. Freimer, director of the division of neuromuscular disorders and the codirector of the Myasthenia Gravis Clinic at the Ohio State University in Columbus.
Impact on fatigue
In a separate post hoc analysis of patients who entered RAISE-XT, the researchers evaluated long-term effects of zilucoplan on fatigue.
Improvements in fatigue were already apparent at the end of the RAISE trial, with the least squares mean (LSM) change from baseline in fatigue assessed after 12 weeks with the Neuro-QOL T-score being –6.26 for zilucoplan (n = 86) compared with an increase of 2.65 for placebo (n = 88; LSM difference, –3.61; nominal P = .0060).
Patients who received placebo in the RAISE trial were able to switch to zilucoplan in the RAISE-XT open-label trail, and among those who did, fatigue, as measured in the T-scores, improved significantly within 1 week of switching.
Fatigue further improved out to week 16 in terms of T-scores for the placebo-switch as well as zilucoplan groups, and the improvements in the scores were sustained through week 60 (mean change from RAISE baseline, –10.71 [n = 42] and –9.15 [n = 42], respectively).
“Fatigue is a challenge for patients with [generalized myasthenia gravis]. Zilucoplan significantly and clinically meaningfully improved myasthenic fatigue versus placebo during RAISE,” the investigators report.
“Further improvements were observed during RAISE-XT and were sustained up to 60 weeks of treatment,” they added.
Favorable safety profile
Zilucoplan continued to show a favorable safety profile in the RAISE-XT trial and was well tolerated in the long term.
The most common adverse reactions (10% or more) in patients with generalized myasthenia gravis were injection-site reactions, upper respiratory tract infection, and diarrhea.
Though additional therapies have also recently entered the market for generalized myasthenia gravis, they are either intravenous or subcutaneous infusions requiring a health care professional to administer them.
“This self-administered medication allows patients to be more independent and can even travel since it is not dependent on an infusion center,” Dr. Freimer noted.
However, similar to the other complement inhibitors, zilucoplan carries a risk for infection, particularly meningitis, Dr. Du noted. “Complement meningitis vaccination protocol and close monitoring for signs of infection are required,” she said.
However, she added, “the rapid action benefits shown as early as week 1 are impressive, and potentially open the door to use as adjunctive therapy in myasthenia gravis crisis.”
“I think zilucoplan definitely adds more excitement to the already active field of myasthenia gravis management,” she said.
The study was funded by UCB Pharma. Dr. Freimer has reports she has served as a paid consultant for argenx, UCB Pharma, and Alexion Pharmaceuticals and reports research support from the National Institutes of Health, UCB Pharma, Jansen Pharmaceuticals, Alnylam, Avidity, and Fulcrum. Dr. Du reports no disclosures.
A version of this article appeared on Medscape.com.
At AANEM 2023
AI tool perfect in study of inflammatory diseases
Artificial intelligence can distinguish overlapping inflammatory conditions with total accuracy, according to a new study presented at the annual meeting of the American College of Rheumatology.
Texas pediatricians faced a conundrum during the pandemic. Endemic typhus, a flea-borne tropical infection common to the region, is nearly indistinguishable from multisystem inflammatory syndrome in children (MIS-C), a rare condition set in motion by SARS-CoV-2 infection. Children with either ailment had seemingly identical symptoms: fever, rash, gastrointestinal issues, and in need of swift treatment. A diagnosis of endemic typhus can take 4-6 days to confirm.
Tiphanie Vogel, MD, PhD, a pediatric rheumatologist at Texas Children’s Hospital, Houston, and colleagues sought to create a tool to hasten diagnosis and, ideally, treatment. To do so, they incorporated machine learning and clinical factors available within the first 6 hours of the onset of symptoms.
The team analyzed 49 demographic, clinical, and laboratory measures from the medical records of 133 children with MIS-C and 87 with endemic typhus. Using deep learning, they narrowed the model to 30 essential features that became the backbone of AI-MET, a two-phase clinical-decision support system.
Phase 1 uses 17 clinical factors and can be performed on paper. If a patient’s score in phase 1 is not determinative, clinicians proceed to phase 2, which uses an additional 13 weighted factors and machine learning.
In testing, the two-part tool classified each of the 220 test patients perfectly. And it diagnosed a second group of 111 patients with MIS-C with 99% (110/111) accuracy.
Of note, “that first step classifies [a patient] correctly half of the time,” Dr. Vogel said, so the second, AI phase of the tool was necessary for only half of cases. Dr. Vogel said that’s a good sign; it means that the tool is useful in settings where AI may not always be feasible, like in a busy ED.
Melissa Mizesko, MD, a pediatric rheumatologist at Driscoll Children’s Hospital in Corpus Christi, Tex., said that the new tool could help clinicians streamline care. When cases of MIS-C peaked in Texas, clinicians often would start sick children on doxycycline and treat for MIS-C at the same time, then wait to see whether the antibiotic brought the fever down.
“This [new tool] is helpful if you live in a part of the country that has typhus,” said Jane Burns, MD, director of the Kawasaki Disease Research Center at the University of California, San Diego, who helped develop a similar AI-based tool to distinguish MIS-C from Kawasaki disease. But she encouraged the researchers to expand their testing to include other conditions. Although the AI model Dr. Vogel’s group developed can pinpoint MIS-C or endemic typhus, what if a child has neither condition? “It’s not often you’re dealing with a diagnosis between just two specific diseases,” Dr. Burns said.
Dr. Vogel is also interested in making AI-MET more efficient. “This go-round we prioritized perfect accuracy,” she said. But 30 clinical factors, with 17 of them recorded and calculated by hand, is a lot. “Could we still get this to be very accurate, maybe not perfect, with less inputs?”
In addition to refining AI-MET, which Texas Children’s eventually hopes to make available to other institutions, Dr. Vogel and associates are also considering other use cases for AI. Lupus is one option. “Maybe with machine learning we could identify clues at diagnosis that would help recommend targeted treatment,” she said
Dr. Vogel disclosed potential conflicts of interest with Moderna, Novartis, Pfizer, and SOBI. Dr. Burns and Dr. Mizesko disclosed no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
Artificial intelligence can distinguish overlapping inflammatory conditions with total accuracy, according to a new study presented at the annual meeting of the American College of Rheumatology.
Texas pediatricians faced a conundrum during the pandemic. Endemic typhus, a flea-borne tropical infection common to the region, is nearly indistinguishable from multisystem inflammatory syndrome in children (MIS-C), a rare condition set in motion by SARS-CoV-2 infection. Children with either ailment had seemingly identical symptoms: fever, rash, gastrointestinal issues, and in need of swift treatment. A diagnosis of endemic typhus can take 4-6 days to confirm.
Tiphanie Vogel, MD, PhD, a pediatric rheumatologist at Texas Children’s Hospital, Houston, and colleagues sought to create a tool to hasten diagnosis and, ideally, treatment. To do so, they incorporated machine learning and clinical factors available within the first 6 hours of the onset of symptoms.
The team analyzed 49 demographic, clinical, and laboratory measures from the medical records of 133 children with MIS-C and 87 with endemic typhus. Using deep learning, they narrowed the model to 30 essential features that became the backbone of AI-MET, a two-phase clinical-decision support system.
Phase 1 uses 17 clinical factors and can be performed on paper. If a patient’s score in phase 1 is not determinative, clinicians proceed to phase 2, which uses an additional 13 weighted factors and machine learning.
In testing, the two-part tool classified each of the 220 test patients perfectly. And it diagnosed a second group of 111 patients with MIS-C with 99% (110/111) accuracy.
Of note, “that first step classifies [a patient] correctly half of the time,” Dr. Vogel said, so the second, AI phase of the tool was necessary for only half of cases. Dr. Vogel said that’s a good sign; it means that the tool is useful in settings where AI may not always be feasible, like in a busy ED.
Melissa Mizesko, MD, a pediatric rheumatologist at Driscoll Children’s Hospital in Corpus Christi, Tex., said that the new tool could help clinicians streamline care. When cases of MIS-C peaked in Texas, clinicians often would start sick children on doxycycline and treat for MIS-C at the same time, then wait to see whether the antibiotic brought the fever down.
“This [new tool] is helpful if you live in a part of the country that has typhus,” said Jane Burns, MD, director of the Kawasaki Disease Research Center at the University of California, San Diego, who helped develop a similar AI-based tool to distinguish MIS-C from Kawasaki disease. But she encouraged the researchers to expand their testing to include other conditions. Although the AI model Dr. Vogel’s group developed can pinpoint MIS-C or endemic typhus, what if a child has neither condition? “It’s not often you’re dealing with a diagnosis between just two specific diseases,” Dr. Burns said.
Dr. Vogel is also interested in making AI-MET more efficient. “This go-round we prioritized perfect accuracy,” she said. But 30 clinical factors, with 17 of them recorded and calculated by hand, is a lot. “Could we still get this to be very accurate, maybe not perfect, with less inputs?”
In addition to refining AI-MET, which Texas Children’s eventually hopes to make available to other institutions, Dr. Vogel and associates are also considering other use cases for AI. Lupus is one option. “Maybe with machine learning we could identify clues at diagnosis that would help recommend targeted treatment,” she said
Dr. Vogel disclosed potential conflicts of interest with Moderna, Novartis, Pfizer, and SOBI. Dr. Burns and Dr. Mizesko disclosed no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
Artificial intelligence can distinguish overlapping inflammatory conditions with total accuracy, according to a new study presented at the annual meeting of the American College of Rheumatology.
Texas pediatricians faced a conundrum during the pandemic. Endemic typhus, a flea-borne tropical infection common to the region, is nearly indistinguishable from multisystem inflammatory syndrome in children (MIS-C), a rare condition set in motion by SARS-CoV-2 infection. Children with either ailment had seemingly identical symptoms: fever, rash, gastrointestinal issues, and in need of swift treatment. A diagnosis of endemic typhus can take 4-6 days to confirm.
Tiphanie Vogel, MD, PhD, a pediatric rheumatologist at Texas Children’s Hospital, Houston, and colleagues sought to create a tool to hasten diagnosis and, ideally, treatment. To do so, they incorporated machine learning and clinical factors available within the first 6 hours of the onset of symptoms.
The team analyzed 49 demographic, clinical, and laboratory measures from the medical records of 133 children with MIS-C and 87 with endemic typhus. Using deep learning, they narrowed the model to 30 essential features that became the backbone of AI-MET, a two-phase clinical-decision support system.
Phase 1 uses 17 clinical factors and can be performed on paper. If a patient’s score in phase 1 is not determinative, clinicians proceed to phase 2, which uses an additional 13 weighted factors and machine learning.
In testing, the two-part tool classified each of the 220 test patients perfectly. And it diagnosed a second group of 111 patients with MIS-C with 99% (110/111) accuracy.
Of note, “that first step classifies [a patient] correctly half of the time,” Dr. Vogel said, so the second, AI phase of the tool was necessary for only half of cases. Dr. Vogel said that’s a good sign; it means that the tool is useful in settings where AI may not always be feasible, like in a busy ED.
Melissa Mizesko, MD, a pediatric rheumatologist at Driscoll Children’s Hospital in Corpus Christi, Tex., said that the new tool could help clinicians streamline care. When cases of MIS-C peaked in Texas, clinicians often would start sick children on doxycycline and treat for MIS-C at the same time, then wait to see whether the antibiotic brought the fever down.
“This [new tool] is helpful if you live in a part of the country that has typhus,” said Jane Burns, MD, director of the Kawasaki Disease Research Center at the University of California, San Diego, who helped develop a similar AI-based tool to distinguish MIS-C from Kawasaki disease. But she encouraged the researchers to expand their testing to include other conditions. Although the AI model Dr. Vogel’s group developed can pinpoint MIS-C or endemic typhus, what if a child has neither condition? “It’s not often you’re dealing with a diagnosis between just two specific diseases,” Dr. Burns said.
Dr. Vogel is also interested in making AI-MET more efficient. “This go-round we prioritized perfect accuracy,” she said. But 30 clinical factors, with 17 of them recorded and calculated by hand, is a lot. “Could we still get this to be very accurate, maybe not perfect, with less inputs?”
In addition to refining AI-MET, which Texas Children’s eventually hopes to make available to other institutions, Dr. Vogel and associates are also considering other use cases for AI. Lupus is one option. “Maybe with machine learning we could identify clues at diagnosis that would help recommend targeted treatment,” she said
Dr. Vogel disclosed potential conflicts of interest with Moderna, Novartis, Pfizer, and SOBI. Dr. Burns and Dr. Mizesko disclosed no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
FROM ACR 2023
TNF inhibitors may be OK for treating RA-associated interstitial lung disease
SAN DIEGO – Patients with rheumatoid arthritis-associated interstitial lung disease (RA-ILD) who start a tumor necrosis factor inhibitor (TNFi) appear to have rates of survival and respiratory-related hospitalization similar to those initiating a non-TNFi biologic disease-modifying antirheumatic drug (bDMARD) or Janus kinase inhibitor (JAKi), results from a large pharmacoepidemiologic study show.
“These results challenge some of the findings in prior literature that perhaps TNFi should be avoided in RA-ILD,” lead study investigator Bryant R. England, MD, PhD, said in an interview. The findings were presented during a plenary session at the American College of Rheumatology annual meeting.
Dr. England, associate professor of rheumatology and immunology at the University of Nebraska Medical Center, Omaha, said that while RA-ILD carries a poor prognosis, a paucity of evidence exists on the effectiveness and safety of disease-modifying therapies in this population.
It’s a pleasant surprise “to see that the investigators were unable to demonstrate a significant difference in the risk of respiratory hospitalization or death between people with RA-ILD initiating non-TNFi/JAKi versus TNFi. Here is a unique situation where a so called ‘negative’ study contributes important information. This study provides needed safety data, as they were unable to show that TNFi results in worsening of severe RA-ILD outcomes,” Sindhu R. Johnson, MD, PhD, professor of medicine at the University of Toronto, said when asked for comment on the study.
“While this study does not address the use of these medications for the treatment of RA-ILD, these data suggest that TNFi may remain a treatment option for articular disease in people with RA-ILD,” said Dr. Johnson, who was not involved with the study.
For the study, Dr. England and colleagues drew from Veterans Health Administration data between 2006 and 2018 to identify patients with RA-ILD initiating TNFi or non-TNFi biologic/JAKi for the first time. Those who received ILD-focused therapies such as mycophenolate and antifibrotics were excluded from the analysis.
The researchers used validated administrative algorithms requiring multiple RA and ILD diagnostic codes to identify RA-ILD and used 1:1 propensity score matching to compare TNFi and non-TNFi biologic/JAKi factors such as health care use, comorbidities, and several RA-ILD factors, such as pretreatment forced vital capacity, obtained from electronic health records and administrative data. The primary outcome was a composite of time to respiratory-related hospitalization or death using Cox regression models.
Dr. England reported findings from 237 TNFi initiators and 237 non-TNFi/JAKi initiators. Their mean age was 68 years and 92% were male. After matching, the mean standardized differences of variables in the propensity score model improved, but a few variables remained slightly imbalanced, such as two markers of inflammation, inhaled corticosteroid use, and body mass index. The most frequently prescribed TNFi drugs were adalimumab (51%) and etanercept (37%), and the most frequently prescribed non-TNFi/JAKi drugs were rituximab (53%) and abatacept (28%).
The researchers observed no significant difference in the primary outcome between non-TNFi/JAKi and TNFi initiators (adjusted hazard ratio [aHR], 1.22; 95% confidence interval [CI], 0.92-1.60). They also observed no significant differences in respiratory hospitalization, all-cause mortality, or respiratory-related death at 1 and 3 years. In sensitivity analyses with modified cohort eligibility requirements, no significant differences in outcomes were observed between non-TNFi/JAKi and TNFi initiators.
During his presentation at the meeting, Dr. England posed the question: Are TNFi drugs safe to be used in RA-ILD?
“The answer is: It’s complex,” he said. “Our findings don’t suggest that we should be systematically avoiding TNFis with every single person with RA-ILD. But that’s different than whether there are specific subpopulations of RA-ILD for which the choice of these therapies may differ. Unfortunately, we could not address that in this study. We also could not address whether TNFis have efficacy at stopping, slowing, or reversing progression of the ILD itself. This calls for us as a field to gather together and pursue clinical trials to try to generate robust evidence that can guide these important clinical decisions that we’re making with our patients.”
He acknowledged certain limitations of the analysis, including its observational design. “So, despite best efforts to minimize bias with pharmacoepidemiologic designs and approaches, there could still be confounding and selection,” he said. “Additionally, RA-ILD is a heterogeneous disease characterized by different patterns and trajectories. While we did account for several RA- and ILD-related factors, we could not account for all heterogeneity in RA-ILD.”
When asked for comment on the study, session moderator Janet Pope, MD, MPH, professor of medicine in the division of rheumatology at the University of Western Ontario, London, said that the study findings surprised her.
“Sometimes RA patients on TNFis were thought to have more new or worsening ILD vs. [those on] non-TNFi bDMARDs, but most [data were] from older studies where TNFis were used as initial bDMARD in sicker patients,” she told this news organization. “So, data were confounded previously. Even in this study, there may have been channeling bias as it was not a randomized controlled trial. We need a definitive randomized controlled trial to answer this question of what the most optimal therapy for RA-ILD is.”
Dr. England reports receiving consulting fees and research support from Boehringer Ingelheim, and several coauthors reported financial relationships from various pharmaceutical companies and medical publishers. Dr. Johnson reports no relevant financial relationships. Dr. Pope reports being a consultant for several pharmaceutical companies. She has received grant/research support from AbbVie/Abbott and Eli Lilly and is an adviser for Boehringer Ingelheim.
A version of this article appeared on Medscape.com.
SAN DIEGO – Patients with rheumatoid arthritis-associated interstitial lung disease (RA-ILD) who start a tumor necrosis factor inhibitor (TNFi) appear to have rates of survival and respiratory-related hospitalization similar to those initiating a non-TNFi biologic disease-modifying antirheumatic drug (bDMARD) or Janus kinase inhibitor (JAKi), results from a large pharmacoepidemiologic study show.
“These results challenge some of the findings in prior literature that perhaps TNFi should be avoided in RA-ILD,” lead study investigator Bryant R. England, MD, PhD, said in an interview. The findings were presented during a plenary session at the American College of Rheumatology annual meeting.
Dr. England, associate professor of rheumatology and immunology at the University of Nebraska Medical Center, Omaha, said that while RA-ILD carries a poor prognosis, a paucity of evidence exists on the effectiveness and safety of disease-modifying therapies in this population.
It’s a pleasant surprise “to see that the investigators were unable to demonstrate a significant difference in the risk of respiratory hospitalization or death between people with RA-ILD initiating non-TNFi/JAKi versus TNFi. Here is a unique situation where a so called ‘negative’ study contributes important information. This study provides needed safety data, as they were unable to show that TNFi results in worsening of severe RA-ILD outcomes,” Sindhu R. Johnson, MD, PhD, professor of medicine at the University of Toronto, said when asked for comment on the study.
“While this study does not address the use of these medications for the treatment of RA-ILD, these data suggest that TNFi may remain a treatment option for articular disease in people with RA-ILD,” said Dr. Johnson, who was not involved with the study.
For the study, Dr. England and colleagues drew from Veterans Health Administration data between 2006 and 2018 to identify patients with RA-ILD initiating TNFi or non-TNFi biologic/JAKi for the first time. Those who received ILD-focused therapies such as mycophenolate and antifibrotics were excluded from the analysis.
The researchers used validated administrative algorithms requiring multiple RA and ILD diagnostic codes to identify RA-ILD and used 1:1 propensity score matching to compare TNFi and non-TNFi biologic/JAKi factors such as health care use, comorbidities, and several RA-ILD factors, such as pretreatment forced vital capacity, obtained from electronic health records and administrative data. The primary outcome was a composite of time to respiratory-related hospitalization or death using Cox regression models.
Dr. England reported findings from 237 TNFi initiators and 237 non-TNFi/JAKi initiators. Their mean age was 68 years and 92% were male. After matching, the mean standardized differences of variables in the propensity score model improved, but a few variables remained slightly imbalanced, such as two markers of inflammation, inhaled corticosteroid use, and body mass index. The most frequently prescribed TNFi drugs were adalimumab (51%) and etanercept (37%), and the most frequently prescribed non-TNFi/JAKi drugs were rituximab (53%) and abatacept (28%).
The researchers observed no significant difference in the primary outcome between non-TNFi/JAKi and TNFi initiators (adjusted hazard ratio [aHR], 1.22; 95% confidence interval [CI], 0.92-1.60). They also observed no significant differences in respiratory hospitalization, all-cause mortality, or respiratory-related death at 1 and 3 years. In sensitivity analyses with modified cohort eligibility requirements, no significant differences in outcomes were observed between non-TNFi/JAKi and TNFi initiators.
During his presentation at the meeting, Dr. England posed the question: Are TNFi drugs safe to be used in RA-ILD?
“The answer is: It’s complex,” he said. “Our findings don’t suggest that we should be systematically avoiding TNFis with every single person with RA-ILD. But that’s different than whether there are specific subpopulations of RA-ILD for which the choice of these therapies may differ. Unfortunately, we could not address that in this study. We also could not address whether TNFis have efficacy at stopping, slowing, or reversing progression of the ILD itself. This calls for us as a field to gather together and pursue clinical trials to try to generate robust evidence that can guide these important clinical decisions that we’re making with our patients.”
He acknowledged certain limitations of the analysis, including its observational design. “So, despite best efforts to minimize bias with pharmacoepidemiologic designs and approaches, there could still be confounding and selection,” he said. “Additionally, RA-ILD is a heterogeneous disease characterized by different patterns and trajectories. While we did account for several RA- and ILD-related factors, we could not account for all heterogeneity in RA-ILD.”
When asked for comment on the study, session moderator Janet Pope, MD, MPH, professor of medicine in the division of rheumatology at the University of Western Ontario, London, said that the study findings surprised her.
“Sometimes RA patients on TNFis were thought to have more new or worsening ILD vs. [those on] non-TNFi bDMARDs, but most [data were] from older studies where TNFis were used as initial bDMARD in sicker patients,” she told this news organization. “So, data were confounded previously. Even in this study, there may have been channeling bias as it was not a randomized controlled trial. We need a definitive randomized controlled trial to answer this question of what the most optimal therapy for RA-ILD is.”
Dr. England reports receiving consulting fees and research support from Boehringer Ingelheim, and several coauthors reported financial relationships from various pharmaceutical companies and medical publishers. Dr. Johnson reports no relevant financial relationships. Dr. Pope reports being a consultant for several pharmaceutical companies. She has received grant/research support from AbbVie/Abbott and Eli Lilly and is an adviser for Boehringer Ingelheim.
A version of this article appeared on Medscape.com.
SAN DIEGO – Patients with rheumatoid arthritis-associated interstitial lung disease (RA-ILD) who start a tumor necrosis factor inhibitor (TNFi) appear to have rates of survival and respiratory-related hospitalization similar to those initiating a non-TNFi biologic disease-modifying antirheumatic drug (bDMARD) or Janus kinase inhibitor (JAKi), results from a large pharmacoepidemiologic study show.
“These results challenge some of the findings in prior literature that perhaps TNFi should be avoided in RA-ILD,” lead study investigator Bryant R. England, MD, PhD, said in an interview. The findings were presented during a plenary session at the American College of Rheumatology annual meeting.
Dr. England, associate professor of rheumatology and immunology at the University of Nebraska Medical Center, Omaha, said that while RA-ILD carries a poor prognosis, a paucity of evidence exists on the effectiveness and safety of disease-modifying therapies in this population.
It’s a pleasant surprise “to see that the investigators were unable to demonstrate a significant difference in the risk of respiratory hospitalization or death between people with RA-ILD initiating non-TNFi/JAKi versus TNFi. Here is a unique situation where a so called ‘negative’ study contributes important information. This study provides needed safety data, as they were unable to show that TNFi results in worsening of severe RA-ILD outcomes,” Sindhu R. Johnson, MD, PhD, professor of medicine at the University of Toronto, said when asked for comment on the study.
“While this study does not address the use of these medications for the treatment of RA-ILD, these data suggest that TNFi may remain a treatment option for articular disease in people with RA-ILD,” said Dr. Johnson, who was not involved with the study.
For the study, Dr. England and colleagues drew from Veterans Health Administration data between 2006 and 2018 to identify patients with RA-ILD initiating TNFi or non-TNFi biologic/JAKi for the first time. Those who received ILD-focused therapies such as mycophenolate and antifibrotics were excluded from the analysis.
The researchers used validated administrative algorithms requiring multiple RA and ILD diagnostic codes to identify RA-ILD and used 1:1 propensity score matching to compare TNFi and non-TNFi biologic/JAKi factors such as health care use, comorbidities, and several RA-ILD factors, such as pretreatment forced vital capacity, obtained from electronic health records and administrative data. The primary outcome was a composite of time to respiratory-related hospitalization or death using Cox regression models.
Dr. England reported findings from 237 TNFi initiators and 237 non-TNFi/JAKi initiators. Their mean age was 68 years and 92% were male. After matching, the mean standardized differences of variables in the propensity score model improved, but a few variables remained slightly imbalanced, such as two markers of inflammation, inhaled corticosteroid use, and body mass index. The most frequently prescribed TNFi drugs were adalimumab (51%) and etanercept (37%), and the most frequently prescribed non-TNFi/JAKi drugs were rituximab (53%) and abatacept (28%).
The researchers observed no significant difference in the primary outcome between non-TNFi/JAKi and TNFi initiators (adjusted hazard ratio [aHR], 1.22; 95% confidence interval [CI], 0.92-1.60). They also observed no significant differences in respiratory hospitalization, all-cause mortality, or respiratory-related death at 1 and 3 years. In sensitivity analyses with modified cohort eligibility requirements, no significant differences in outcomes were observed between non-TNFi/JAKi and TNFi initiators.
During his presentation at the meeting, Dr. England posed the question: Are TNFi drugs safe to be used in RA-ILD?
“The answer is: It’s complex,” he said. “Our findings don’t suggest that we should be systematically avoiding TNFis with every single person with RA-ILD. But that’s different than whether there are specific subpopulations of RA-ILD for which the choice of these therapies may differ. Unfortunately, we could not address that in this study. We also could not address whether TNFis have efficacy at stopping, slowing, or reversing progression of the ILD itself. This calls for us as a field to gather together and pursue clinical trials to try to generate robust evidence that can guide these important clinical decisions that we’re making with our patients.”
He acknowledged certain limitations of the analysis, including its observational design. “So, despite best efforts to minimize bias with pharmacoepidemiologic designs and approaches, there could still be confounding and selection,” he said. “Additionally, RA-ILD is a heterogeneous disease characterized by different patterns and trajectories. While we did account for several RA- and ILD-related factors, we could not account for all heterogeneity in RA-ILD.”
When asked for comment on the study, session moderator Janet Pope, MD, MPH, professor of medicine in the division of rheumatology at the University of Western Ontario, London, said that the study findings surprised her.
“Sometimes RA patients on TNFis were thought to have more new or worsening ILD vs. [those on] non-TNFi bDMARDs, but most [data were] from older studies where TNFis were used as initial bDMARD in sicker patients,” she told this news organization. “So, data were confounded previously. Even in this study, there may have been channeling bias as it was not a randomized controlled trial. We need a definitive randomized controlled trial to answer this question of what the most optimal therapy for RA-ILD is.”
Dr. England reports receiving consulting fees and research support from Boehringer Ingelheim, and several coauthors reported financial relationships from various pharmaceutical companies and medical publishers. Dr. Johnson reports no relevant financial relationships. Dr. Pope reports being a consultant for several pharmaceutical companies. She has received grant/research support from AbbVie/Abbott and Eli Lilly and is an adviser for Boehringer Ingelheim.
A version of this article appeared on Medscape.com.
AT ACR 2023
Vasculitis confers higher risk for adverse pregnancy outcomes
SAN DIEGO – Pregnancy in patients with vasculitis had a higher risk for preterm delivery and preeclampsia/eclampsia – especially those with small-vessel vasculitis – compared with the general obstetric population, in a large analysis of administrative claims data presented at the American College of Rheumatology annual meeting.
“We suspect that there is a relationship between the increased risk of these serious hypertensive disorders and preterm delivery, given the higher risk of medically indicated preterm delivery,” one the of the study authors, Audra Horomanski, MD, said in an interview prior to her presentation in a plenary session at the meeting.
Limited data exist on the risks of pregnancy in patients with systemic vasculitis, according to Dr. Horomanski, a rheumatologist who directs the Stanford Vasculitis Clinic at Stanford (Calif.) University. “The majority of what we do know comes from relatively small cohort studies,” she said. “This is the first U.S., nationwide database study looking at the risk of preterm delivery and other adverse pregnancy outcomes.”
Drawing on administrative claims data from private health insurance providers, Dr. Horomanski and her colleagues identified all pregnancies regardless of outcome for patients with and without vasculitis from 2007 to 2021. They defined vasculitis as ≥ 2 ICD-coded outpatient visits or ≥ 1 ICD-coded inpatient visit occurring before the estimated last menstrual period (LMP), and they further categorized vasculitis by vessel size: large, medium, small, and variable, based on Chapel Hill Consensus Conference criteria. For a referent population, they included patients without vasculitis or other rheumatic disease, defined as no ICD-coded outpatient or inpatient visits for vasculitis, systemic lupus erythematosus, rheumatoid arthritis, systemic sclerosis, or juvenile idiopathic arthritis before LMP or during pregnancy. Next, the researchers described pregnancy outcomes in patients with vasculitis compared with the referent population, and explored pregnancy characteristics and complications in patients with vasculitis stratified by parity (nulliparous vs. multiparous).
Dr. Horomanski reported results from 665 pregnancies in 527 patients with vasculitis and 4,209,034 pregnancies in 2,932,379 patients from the referent population. Patients with vasculitis had higher rates of spontaneous abortion (21% vs. 19%), elective termination (6% vs. 5%), ectopic and molar pregnancy (4% vs. 3%), and preterm delivery (13% vs. 6%). Approximately 12% of pregnancies among patients with vasculitis were complicated by preeclampsia. Multiparous pregnancies had a slightly higher frequency of preterm delivery than did nulliparous pregnancies (14% vs. 13%) and were more often comorbid with gestational diabetes (11% vs. 6%) and prepregnancy hypertension (23% vs. 13%). Patients with small-vessel vasculitis had higher frequencies of spontaneous abortion, preterm delivery, and comorbidities among vasculitis subtypes.
“I was surprised that vasculitis patients were less likely to be diagnosed with gestational hypertension compared to the general population, but more likely to be diagnosed with preeclampsia/eclampsia,” Dr. Horomanski added. “It raises questions about whether vasculitis patients are more likely to be diagnosed with more serious hypertensive disorders of pregnancy due to their underlying systemic disease or due to the perceptions of the treating clinicians.”
She acknowledged certain limitations of the study, including the fact that it lacked information on race and ethnicity and was limited to privately insured individuals. This “suggests that we are likely missing patients with disabilities and those who are uninsured, both groups that may be at higher risk for adverse pregnancy outcomes,” she said. “We also have no information on disease activity or flare events which may contribute to these outcomes, particularly medically indicated preterm delivery. There is also a risk of misclassification due to the use of claims data and ICD coding. This misclassification may impact vasculitis diagnoses, parity, and early pregnancy losses.”
Despite the limitations, she said that the work “highlights the value of large database analysis as a complement to prior cohort studies to further clarify this complex picture. Overall, this information is valuable for the counseling of vasculitis patients considering pregnancy and for creating a plan to monitor for pregnancy complications.”
Lindsay S. Lally, MD, a rheumatologist with Hospital for Special Surgery in New York, who was asked to comment on the study, characterized the findings as “important in how many women with vasculitis and vasculitis pregnancies were identified. These data are a start at heightening our awareness about potential complications these women may experience during pregnancy. This study should help inform our family planning conversations with our vasculitis patients. Discussing potential reproductive risks, which are likely mediated by the disease itself, as well as the treatments that we prescribe, is important to help our vasculitis patients make informed decisions.”
Dr. Lally noted that an ongoing project through the Vasculitis Clinical Research Consortium includes a prospective registry of pregnant women with vasculitis, which asks pregnant patients to enter information throughout their pregnancy. “These studies will ultimately help optimize care of our vasculitis patients during pregnancy, ensuring the best outcomes for mother and baby,” she said.
Dr. Horomanski disclosed that she has received research support from Principia, BeiGene, Gilead, and the National Institutes of Health. Dr. Lally reported having no relevant financial relationships.
A version of this article appeared on Medscape.com.
SAN DIEGO – Pregnancy in patients with vasculitis had a higher risk for preterm delivery and preeclampsia/eclampsia – especially those with small-vessel vasculitis – compared with the general obstetric population, in a large analysis of administrative claims data presented at the American College of Rheumatology annual meeting.
“We suspect that there is a relationship between the increased risk of these serious hypertensive disorders and preterm delivery, given the higher risk of medically indicated preterm delivery,” one the of the study authors, Audra Horomanski, MD, said in an interview prior to her presentation in a plenary session at the meeting.
Limited data exist on the risks of pregnancy in patients with systemic vasculitis, according to Dr. Horomanski, a rheumatologist who directs the Stanford Vasculitis Clinic at Stanford (Calif.) University. “The majority of what we do know comes from relatively small cohort studies,” she said. “This is the first U.S., nationwide database study looking at the risk of preterm delivery and other adverse pregnancy outcomes.”
Drawing on administrative claims data from private health insurance providers, Dr. Horomanski and her colleagues identified all pregnancies regardless of outcome for patients with and without vasculitis from 2007 to 2021. They defined vasculitis as ≥ 2 ICD-coded outpatient visits or ≥ 1 ICD-coded inpatient visit occurring before the estimated last menstrual period (LMP), and they further categorized vasculitis by vessel size: large, medium, small, and variable, based on Chapel Hill Consensus Conference criteria. For a referent population, they included patients without vasculitis or other rheumatic disease, defined as no ICD-coded outpatient or inpatient visits for vasculitis, systemic lupus erythematosus, rheumatoid arthritis, systemic sclerosis, or juvenile idiopathic arthritis before LMP or during pregnancy. Next, the researchers described pregnancy outcomes in patients with vasculitis compared with the referent population, and explored pregnancy characteristics and complications in patients with vasculitis stratified by parity (nulliparous vs. multiparous).
Dr. Horomanski reported results from 665 pregnancies in 527 patients with vasculitis and 4,209,034 pregnancies in 2,932,379 patients from the referent population. Patients with vasculitis had higher rates of spontaneous abortion (21% vs. 19%), elective termination (6% vs. 5%), ectopic and molar pregnancy (4% vs. 3%), and preterm delivery (13% vs. 6%). Approximately 12% of pregnancies among patients with vasculitis were complicated by preeclampsia. Multiparous pregnancies had a slightly higher frequency of preterm delivery than did nulliparous pregnancies (14% vs. 13%) and were more often comorbid with gestational diabetes (11% vs. 6%) and prepregnancy hypertension (23% vs. 13%). Patients with small-vessel vasculitis had higher frequencies of spontaneous abortion, preterm delivery, and comorbidities among vasculitis subtypes.
“I was surprised that vasculitis patients were less likely to be diagnosed with gestational hypertension compared to the general population, but more likely to be diagnosed with preeclampsia/eclampsia,” Dr. Horomanski added. “It raises questions about whether vasculitis patients are more likely to be diagnosed with more serious hypertensive disorders of pregnancy due to their underlying systemic disease or due to the perceptions of the treating clinicians.”
She acknowledged certain limitations of the study, including the fact that it lacked information on race and ethnicity and was limited to privately insured individuals. This “suggests that we are likely missing patients with disabilities and those who are uninsured, both groups that may be at higher risk for adverse pregnancy outcomes,” she said. “We also have no information on disease activity or flare events which may contribute to these outcomes, particularly medically indicated preterm delivery. There is also a risk of misclassification due to the use of claims data and ICD coding. This misclassification may impact vasculitis diagnoses, parity, and early pregnancy losses.”
Despite the limitations, she said that the work “highlights the value of large database analysis as a complement to prior cohort studies to further clarify this complex picture. Overall, this information is valuable for the counseling of vasculitis patients considering pregnancy and for creating a plan to monitor for pregnancy complications.”
Lindsay S. Lally, MD, a rheumatologist with Hospital for Special Surgery in New York, who was asked to comment on the study, characterized the findings as “important in how many women with vasculitis and vasculitis pregnancies were identified. These data are a start at heightening our awareness about potential complications these women may experience during pregnancy. This study should help inform our family planning conversations with our vasculitis patients. Discussing potential reproductive risks, which are likely mediated by the disease itself, as well as the treatments that we prescribe, is important to help our vasculitis patients make informed decisions.”
Dr. Lally noted that an ongoing project through the Vasculitis Clinical Research Consortium includes a prospective registry of pregnant women with vasculitis, which asks pregnant patients to enter information throughout their pregnancy. “These studies will ultimately help optimize care of our vasculitis patients during pregnancy, ensuring the best outcomes for mother and baby,” she said.
Dr. Horomanski disclosed that she has received research support from Principia, BeiGene, Gilead, and the National Institutes of Health. Dr. Lally reported having no relevant financial relationships.
A version of this article appeared on Medscape.com.
SAN DIEGO – Pregnancy in patients with vasculitis had a higher risk for preterm delivery and preeclampsia/eclampsia – especially those with small-vessel vasculitis – compared with the general obstetric population, in a large analysis of administrative claims data presented at the American College of Rheumatology annual meeting.
“We suspect that there is a relationship between the increased risk of these serious hypertensive disorders and preterm delivery, given the higher risk of medically indicated preterm delivery,” one the of the study authors, Audra Horomanski, MD, said in an interview prior to her presentation in a plenary session at the meeting.
Limited data exist on the risks of pregnancy in patients with systemic vasculitis, according to Dr. Horomanski, a rheumatologist who directs the Stanford Vasculitis Clinic at Stanford (Calif.) University. “The majority of what we do know comes from relatively small cohort studies,” she said. “This is the first U.S., nationwide database study looking at the risk of preterm delivery and other adverse pregnancy outcomes.”
Drawing on administrative claims data from private health insurance providers, Dr. Horomanski and her colleagues identified all pregnancies regardless of outcome for patients with and without vasculitis from 2007 to 2021. They defined vasculitis as ≥ 2 ICD-coded outpatient visits or ≥ 1 ICD-coded inpatient visit occurring before the estimated last menstrual period (LMP), and they further categorized vasculitis by vessel size: large, medium, small, and variable, based on Chapel Hill Consensus Conference criteria. For a referent population, they included patients without vasculitis or other rheumatic disease, defined as no ICD-coded outpatient or inpatient visits for vasculitis, systemic lupus erythematosus, rheumatoid arthritis, systemic sclerosis, or juvenile idiopathic arthritis before LMP or during pregnancy. Next, the researchers described pregnancy outcomes in patients with vasculitis compared with the referent population, and explored pregnancy characteristics and complications in patients with vasculitis stratified by parity (nulliparous vs. multiparous).
Dr. Horomanski reported results from 665 pregnancies in 527 patients with vasculitis and 4,209,034 pregnancies in 2,932,379 patients from the referent population. Patients with vasculitis had higher rates of spontaneous abortion (21% vs. 19%), elective termination (6% vs. 5%), ectopic and molar pregnancy (4% vs. 3%), and preterm delivery (13% vs. 6%). Approximately 12% of pregnancies among patients with vasculitis were complicated by preeclampsia. Multiparous pregnancies had a slightly higher frequency of preterm delivery than did nulliparous pregnancies (14% vs. 13%) and were more often comorbid with gestational diabetes (11% vs. 6%) and prepregnancy hypertension (23% vs. 13%). Patients with small-vessel vasculitis had higher frequencies of spontaneous abortion, preterm delivery, and comorbidities among vasculitis subtypes.
“I was surprised that vasculitis patients were less likely to be diagnosed with gestational hypertension compared to the general population, but more likely to be diagnosed with preeclampsia/eclampsia,” Dr. Horomanski added. “It raises questions about whether vasculitis patients are more likely to be diagnosed with more serious hypertensive disorders of pregnancy due to their underlying systemic disease or due to the perceptions of the treating clinicians.”
She acknowledged certain limitations of the study, including the fact that it lacked information on race and ethnicity and was limited to privately insured individuals. This “suggests that we are likely missing patients with disabilities and those who are uninsured, both groups that may be at higher risk for adverse pregnancy outcomes,” she said. “We also have no information on disease activity or flare events which may contribute to these outcomes, particularly medically indicated preterm delivery. There is also a risk of misclassification due to the use of claims data and ICD coding. This misclassification may impact vasculitis diagnoses, parity, and early pregnancy losses.”
Despite the limitations, she said that the work “highlights the value of large database analysis as a complement to prior cohort studies to further clarify this complex picture. Overall, this information is valuable for the counseling of vasculitis patients considering pregnancy and for creating a plan to monitor for pregnancy complications.”
Lindsay S. Lally, MD, a rheumatologist with Hospital for Special Surgery in New York, who was asked to comment on the study, characterized the findings as “important in how many women with vasculitis and vasculitis pregnancies were identified. These data are a start at heightening our awareness about potential complications these women may experience during pregnancy. This study should help inform our family planning conversations with our vasculitis patients. Discussing potential reproductive risks, which are likely mediated by the disease itself, as well as the treatments that we prescribe, is important to help our vasculitis patients make informed decisions.”
Dr. Lally noted that an ongoing project through the Vasculitis Clinical Research Consortium includes a prospective registry of pregnant women with vasculitis, which asks pregnant patients to enter information throughout their pregnancy. “These studies will ultimately help optimize care of our vasculitis patients during pregnancy, ensuring the best outcomes for mother and baby,” she said.
Dr. Horomanski disclosed that she has received research support from Principia, BeiGene, Gilead, and the National Institutes of Health. Dr. Lally reported having no relevant financial relationships.
A version of this article appeared on Medscape.com.
AT ACR 2023
Better postpartum BP control with self-monitoring: POP-HT
, new research suggests.
In a randomized trial of 220 women with preeclampsia or gestational hypertension, those who took daily postpartum BP readings and received clinician-guided advice for titrating antihypertensives had a 5 mm Hg–lower average diastolic BP at 9 months, compared with those receiving usual care.
Jamie Kitt, DPhil, from the University of Oxford (England) presented these findings from the Physicians Optimized Postpartum Hypertension Treatment (POP-HT, NCT04273854) clinical trial at the American Heart Association scientific sessions. The study was simultaneously published online in JAMA, and a cardiac imaging substudy was published online in Circulation.
“This trial identifies a potential need for a paradigm shift in the way women affected by hypertensive pregnancy are managed postnatally,” Dr. Kitt said. “If a 5–mm Hg improvement in BP is maintained longer term, it can result in about a 20% reduction in lifetime cardiovascular risk.”
The imaging substudy suggests that short-term postnatal optimization of BP control following hypertensive pregnancy through self-monitoring and physician-guided antihypertensive titration is linked with better cardiac remodeling changes seen by cardiovascular magnetic resonance and echocardiography.
POP-HT “proves for the first time that the first few weeks after delivery are a critical time that can determine the long-term cardiovascular health of the mother,” senior author Paul Leeson, PhD, also from the University of Oxford, who presented the findings in a press briefing, said in an interview.
“Interventions during this period can have long-term beneficial impacts on cardiovascular health,” he said. “These findings rewrite the textbook on our understanding of how and why hypertensive pregnancies associate with later cardiovascular disease in the mother.”
Next, Dr. Leeson said, “We need to work out the best ways to implement these interventions “at scale. Then we can ensure all women who have hypertensive pregnancies can get access to the long-term cardiovascular benefits we have demonstrated are possible through improving postpartum cardiac care,” he said, adding that “this is entirely achievable using current available technologies.”
Hypertension in pregnancy
About 1 in 10 pregnant women develop hypertension in pregnancy (preeclampsia or gestational hypertension), and 1 in 3 such women go on to develop chronic hypertension within 10 years, “when they are usually still in their 30s or 40s,” Dr. Leeson said.
During pregnancy, the heart remodels to cope with pregnancy, and it undergoes more severe changes if BP is high. Then during the 6 weeks after giving birth, this remodeling rapidly reverses.
Higher blood pressure in young adulthood is associated with a twofold higher risk of subsequent myocardial infarction and stroke. And abnormal cardiac remodeling postpartum is also linked with higher cardiovascular risk.
Self-monitoring blood pressure during the postpartum period may be a “critical window” for intervention.
Previously, the research group performed a pilot study, the Self-Management of Postnatal Antihypertensive Treatment (SNAP-HT) trial and the SNAP-extension trial, which compared a BP self-monitoring intervention with usual care in 91 women with gestational hypertension or preeclampsia requiring postnatal antihypertensive treatment.
Diastolic BP, which drives cardiovascular risk in younger populations, was 4.5–mm Hg lower at 6 months postpartum and 7–mm Hg lower at 4 years post partum in patients randomly assigned to BP self-management vs. usual care – even after they were no longer taking antihypertensives.
Building on these findings, the POP-HT trial enrolled 220 pregnant women seen at Oxford University Hospitals in the United Kingdom who were age 18 years or older, had either gestational hypertension or preeclampsia, and still required antihypertensives when they were being discharged from hospital after giving birth.
Following a baseline visit at day 1-6 after delivery, while in the postnatal ward, the patients were randomly assigned 1:1 to the intervention group (112 women) or usual-care group (108 women).
They had an average age of 32.6 years; 40% had gestational hypertension, and 60% had preeclampsia.
Women in the usual-care group typically received a BP review at 7-10 days after hospital discharge with a community midwife, and another at 6-8 weeks with their general practitioner.
The women in the intervention group were given and taught to use a Bluetooth-enabled OMRON Evolv BP monitor (Omron Healthcare Europe) while on the postnatal ward, and they installed a smartphone app on their mobile phones that transmitted self-monitored BP readings to a National Health Service-hosted, web-based platform.
They were instructed to take daily BP measurements (twice daily if out of target range). Dose titration of antihypertensives after hospital discharge was guided remotely by research clinicians, according to a guideline-based algorithm.
Patients in both groups had four study visits when their BP was measured: visit 1 (baseline) between days 1 and 6 post partum; visit 2 at week 1; visit 3 at week 6; and visit 4 between months 6 and 9 post partum.
Similar antihypertensive classes were prescribed in each group (enalapril 57%, nifedipine 27%, and labetalol 30% for intervention vs. enalapril 43%, nifedipine 30%, and labetalol 27% for control).
At 6 weeks, approximately 30% of participants in each group were still taking medication; this dropped to approximately 12% by visit 4.
The primary outcome – the mean 24-hour diastolic BP at visit 4 (roughly 9 months post partum), adjusted for baseline postnatal diastolic blood pressure – was 5.8–mm Hg lower in the intervention group than in the control group (71.2 mm Hg vs. 76.6 mm Hg; P < .001).
Secondary outcomes – between-group differences in systolic BP at 9 months, BP-related postnatal admission, and cardiac remodeling assessed by cardiac magnetic resonance – were all better in the intervention group.
The mean 24-hour average systolic BP at 9 months post partum, adjusted for baseline postnatal systolic BP was 6.5–mm Hg lower in the intervention group than in the control group (114.0 mm Hg vs. 120.3 mm Hg; P < .001).
There was an absolute risk reduction of 20% and a relative risk reduction of 73.5% in postnatal readmission. The number needed to treat to avoid one postnatal readmission was five, which “has potential for big cost savings,” said Dr. Leeson.
Blood pressure post partum can be improved with self-monitoring and physician-guided medication adjustment, Dr. Leeson summarized. The blood pressure remains low for at least 9 months, even when medication is stopped, and the intervention leads to beneficial cardiac remodeling.
U.S. pilot study
Non-Hispanic Black adults have a high hypertension and cardiovascular disease burden, and a related small U.S. study showed benefits of BP self-monitoring in a population comprising mainly Black women, Keith Ferdinand, MD, discussant of the POP-HT trial in the press briefing, said in an interview.
Dr. Ferdinand, from Tulane University, New Orleans, Louisiana, was lead author of the Text My Hypertension BP Meds NOLA pilot study that was published in February in the American Heart Journal Plus: Cardiology Research and Practice.
The study showed that text-messaging and social support increased hypertension medication adherence.
They enrolled 36 individuals, of whom 32 (89%) were non-Hispanic Black, and 23 (64%) were women. The participants received validated Bluetooth-enabled BP-monitoring devices that were synced to smartphones via a secured cloud-based application. The participants could send and receive messages to health care practitioners.
This intervention significantly improved medication adherence and systolic BP without modifying pharmacotherapy.
‘Need to be passionate about monitoring BP’
“The take-home messages from these exciting findings is that physicians and women who have had high BP during pregnancy need to be passionate about monitoring and controlling their blood pressure and not ignore it,” Anastasia Mihailidou, PhD, Royal North Shore Hospital, Sydney, the assigned discussant in the late-breaking trial session, said in an interview.
“It also resulted in fewer postpartum hospital readmissions for high blood pressure and benefit at 9 months in the structure and function of the heart and blood vessels of the women,” she said.
“While we need to see further studies in ethnically diverse women to see that they are reproducible, there are simple measures that clinicians can implement, and women can ask to have their BP monitored more frequently than the current practice. In the U.K. it is 5-10 days after delivery and then at 6-8 weeks after giving birth when changes in heart structure have already started,” Dr. Mihailidou noted.
“The procedure will need to be modified if there are no telemedicine facilities, but that should not stop having close monitoring of BP and treating it adequately. Monitoring requires an accurate BP monitor. There also has to be monitoring BP for the children.”
The trial was funded by a BHF Clinical Research Training Fellowship to Dr. Kitt, with additional support from the NIHR Oxford Biomedical Research Centre and Oxford BHF Centre for Research Excellence.
A version of this article first appeared on Medscape.com.
, new research suggests.
In a randomized trial of 220 women with preeclampsia or gestational hypertension, those who took daily postpartum BP readings and received clinician-guided advice for titrating antihypertensives had a 5 mm Hg–lower average diastolic BP at 9 months, compared with those receiving usual care.
Jamie Kitt, DPhil, from the University of Oxford (England) presented these findings from the Physicians Optimized Postpartum Hypertension Treatment (POP-HT, NCT04273854) clinical trial at the American Heart Association scientific sessions. The study was simultaneously published online in JAMA, and a cardiac imaging substudy was published online in Circulation.
“This trial identifies a potential need for a paradigm shift in the way women affected by hypertensive pregnancy are managed postnatally,” Dr. Kitt said. “If a 5–mm Hg improvement in BP is maintained longer term, it can result in about a 20% reduction in lifetime cardiovascular risk.”
The imaging substudy suggests that short-term postnatal optimization of BP control following hypertensive pregnancy through self-monitoring and physician-guided antihypertensive titration is linked with better cardiac remodeling changes seen by cardiovascular magnetic resonance and echocardiography.
POP-HT “proves for the first time that the first few weeks after delivery are a critical time that can determine the long-term cardiovascular health of the mother,” senior author Paul Leeson, PhD, also from the University of Oxford, who presented the findings in a press briefing, said in an interview.
“Interventions during this period can have long-term beneficial impacts on cardiovascular health,” he said. “These findings rewrite the textbook on our understanding of how and why hypertensive pregnancies associate with later cardiovascular disease in the mother.”
Next, Dr. Leeson said, “We need to work out the best ways to implement these interventions “at scale. Then we can ensure all women who have hypertensive pregnancies can get access to the long-term cardiovascular benefits we have demonstrated are possible through improving postpartum cardiac care,” he said, adding that “this is entirely achievable using current available technologies.”
Hypertension in pregnancy
About 1 in 10 pregnant women develop hypertension in pregnancy (preeclampsia or gestational hypertension), and 1 in 3 such women go on to develop chronic hypertension within 10 years, “when they are usually still in their 30s or 40s,” Dr. Leeson said.
During pregnancy, the heart remodels to cope with pregnancy, and it undergoes more severe changes if BP is high. Then during the 6 weeks after giving birth, this remodeling rapidly reverses.
Higher blood pressure in young adulthood is associated with a twofold higher risk of subsequent myocardial infarction and stroke. And abnormal cardiac remodeling postpartum is also linked with higher cardiovascular risk.
Self-monitoring blood pressure during the postpartum period may be a “critical window” for intervention.
Previously, the research group performed a pilot study, the Self-Management of Postnatal Antihypertensive Treatment (SNAP-HT) trial and the SNAP-extension trial, which compared a BP self-monitoring intervention with usual care in 91 women with gestational hypertension or preeclampsia requiring postnatal antihypertensive treatment.
Diastolic BP, which drives cardiovascular risk in younger populations, was 4.5–mm Hg lower at 6 months postpartum and 7–mm Hg lower at 4 years post partum in patients randomly assigned to BP self-management vs. usual care – even after they were no longer taking antihypertensives.
Building on these findings, the POP-HT trial enrolled 220 pregnant women seen at Oxford University Hospitals in the United Kingdom who were age 18 years or older, had either gestational hypertension or preeclampsia, and still required antihypertensives when they were being discharged from hospital after giving birth.
Following a baseline visit at day 1-6 after delivery, while in the postnatal ward, the patients were randomly assigned 1:1 to the intervention group (112 women) or usual-care group (108 women).
They had an average age of 32.6 years; 40% had gestational hypertension, and 60% had preeclampsia.
Women in the usual-care group typically received a BP review at 7-10 days after hospital discharge with a community midwife, and another at 6-8 weeks with their general practitioner.
The women in the intervention group were given and taught to use a Bluetooth-enabled OMRON Evolv BP monitor (Omron Healthcare Europe) while on the postnatal ward, and they installed a smartphone app on their mobile phones that transmitted self-monitored BP readings to a National Health Service-hosted, web-based platform.
They were instructed to take daily BP measurements (twice daily if out of target range). Dose titration of antihypertensives after hospital discharge was guided remotely by research clinicians, according to a guideline-based algorithm.
Patients in both groups had four study visits when their BP was measured: visit 1 (baseline) between days 1 and 6 post partum; visit 2 at week 1; visit 3 at week 6; and visit 4 between months 6 and 9 post partum.
Similar antihypertensive classes were prescribed in each group (enalapril 57%, nifedipine 27%, and labetalol 30% for intervention vs. enalapril 43%, nifedipine 30%, and labetalol 27% for control).
At 6 weeks, approximately 30% of participants in each group were still taking medication; this dropped to approximately 12% by visit 4.
The primary outcome – the mean 24-hour diastolic BP at visit 4 (roughly 9 months post partum), adjusted for baseline postnatal diastolic blood pressure – was 5.8–mm Hg lower in the intervention group than in the control group (71.2 mm Hg vs. 76.6 mm Hg; P < .001).
Secondary outcomes – between-group differences in systolic BP at 9 months, BP-related postnatal admission, and cardiac remodeling assessed by cardiac magnetic resonance – were all better in the intervention group.
The mean 24-hour average systolic BP at 9 months post partum, adjusted for baseline postnatal systolic BP was 6.5–mm Hg lower in the intervention group than in the control group (114.0 mm Hg vs. 120.3 mm Hg; P < .001).
There was an absolute risk reduction of 20% and a relative risk reduction of 73.5% in postnatal readmission. The number needed to treat to avoid one postnatal readmission was five, which “has potential for big cost savings,” said Dr. Leeson.
Blood pressure post partum can be improved with self-monitoring and physician-guided medication adjustment, Dr. Leeson summarized. The blood pressure remains low for at least 9 months, even when medication is stopped, and the intervention leads to beneficial cardiac remodeling.
U.S. pilot study
Non-Hispanic Black adults have a high hypertension and cardiovascular disease burden, and a related small U.S. study showed benefits of BP self-monitoring in a population comprising mainly Black women, Keith Ferdinand, MD, discussant of the POP-HT trial in the press briefing, said in an interview.
Dr. Ferdinand, from Tulane University, New Orleans, Louisiana, was lead author of the Text My Hypertension BP Meds NOLA pilot study that was published in February in the American Heart Journal Plus: Cardiology Research and Practice.
The study showed that text-messaging and social support increased hypertension medication adherence.
They enrolled 36 individuals, of whom 32 (89%) were non-Hispanic Black, and 23 (64%) were women. The participants received validated Bluetooth-enabled BP-monitoring devices that were synced to smartphones via a secured cloud-based application. The participants could send and receive messages to health care practitioners.
This intervention significantly improved medication adherence and systolic BP without modifying pharmacotherapy.
‘Need to be passionate about monitoring BP’
“The take-home messages from these exciting findings is that physicians and women who have had high BP during pregnancy need to be passionate about monitoring and controlling their blood pressure and not ignore it,” Anastasia Mihailidou, PhD, Royal North Shore Hospital, Sydney, the assigned discussant in the late-breaking trial session, said in an interview.
“It also resulted in fewer postpartum hospital readmissions for high blood pressure and benefit at 9 months in the structure and function of the heart and blood vessels of the women,” she said.
“While we need to see further studies in ethnically diverse women to see that they are reproducible, there are simple measures that clinicians can implement, and women can ask to have their BP monitored more frequently than the current practice. In the U.K. it is 5-10 days after delivery and then at 6-8 weeks after giving birth when changes in heart structure have already started,” Dr. Mihailidou noted.
“The procedure will need to be modified if there are no telemedicine facilities, but that should not stop having close monitoring of BP and treating it adequately. Monitoring requires an accurate BP monitor. There also has to be monitoring BP for the children.”
The trial was funded by a BHF Clinical Research Training Fellowship to Dr. Kitt, with additional support from the NIHR Oxford Biomedical Research Centre and Oxford BHF Centre for Research Excellence.
A version of this article first appeared on Medscape.com.
, new research suggests.
In a randomized trial of 220 women with preeclampsia or gestational hypertension, those who took daily postpartum BP readings and received clinician-guided advice for titrating antihypertensives had a 5 mm Hg–lower average diastolic BP at 9 months, compared with those receiving usual care.
Jamie Kitt, DPhil, from the University of Oxford (England) presented these findings from the Physicians Optimized Postpartum Hypertension Treatment (POP-HT, NCT04273854) clinical trial at the American Heart Association scientific sessions. The study was simultaneously published online in JAMA, and a cardiac imaging substudy was published online in Circulation.
“This trial identifies a potential need for a paradigm shift in the way women affected by hypertensive pregnancy are managed postnatally,” Dr. Kitt said. “If a 5–mm Hg improvement in BP is maintained longer term, it can result in about a 20% reduction in lifetime cardiovascular risk.”
The imaging substudy suggests that short-term postnatal optimization of BP control following hypertensive pregnancy through self-monitoring and physician-guided antihypertensive titration is linked with better cardiac remodeling changes seen by cardiovascular magnetic resonance and echocardiography.
POP-HT “proves for the first time that the first few weeks after delivery are a critical time that can determine the long-term cardiovascular health of the mother,” senior author Paul Leeson, PhD, also from the University of Oxford, who presented the findings in a press briefing, said in an interview.
“Interventions during this period can have long-term beneficial impacts on cardiovascular health,” he said. “These findings rewrite the textbook on our understanding of how and why hypertensive pregnancies associate with later cardiovascular disease in the mother.”
Next, Dr. Leeson said, “We need to work out the best ways to implement these interventions “at scale. Then we can ensure all women who have hypertensive pregnancies can get access to the long-term cardiovascular benefits we have demonstrated are possible through improving postpartum cardiac care,” he said, adding that “this is entirely achievable using current available technologies.”
Hypertension in pregnancy
About 1 in 10 pregnant women develop hypertension in pregnancy (preeclampsia or gestational hypertension), and 1 in 3 such women go on to develop chronic hypertension within 10 years, “when they are usually still in their 30s or 40s,” Dr. Leeson said.
During pregnancy, the heart remodels to cope with pregnancy, and it undergoes more severe changes if BP is high. Then during the 6 weeks after giving birth, this remodeling rapidly reverses.
Higher blood pressure in young adulthood is associated with a twofold higher risk of subsequent myocardial infarction and stroke. And abnormal cardiac remodeling postpartum is also linked with higher cardiovascular risk.
Self-monitoring blood pressure during the postpartum period may be a “critical window” for intervention.
Previously, the research group performed a pilot study, the Self-Management of Postnatal Antihypertensive Treatment (SNAP-HT) trial and the SNAP-extension trial, which compared a BP self-monitoring intervention with usual care in 91 women with gestational hypertension or preeclampsia requiring postnatal antihypertensive treatment.
Diastolic BP, which drives cardiovascular risk in younger populations, was 4.5–mm Hg lower at 6 months postpartum and 7–mm Hg lower at 4 years post partum in patients randomly assigned to BP self-management vs. usual care – even after they were no longer taking antihypertensives.
Building on these findings, the POP-HT trial enrolled 220 pregnant women seen at Oxford University Hospitals in the United Kingdom who were age 18 years or older, had either gestational hypertension or preeclampsia, and still required antihypertensives when they were being discharged from hospital after giving birth.
Following a baseline visit at day 1-6 after delivery, while in the postnatal ward, the patients were randomly assigned 1:1 to the intervention group (112 women) or usual-care group (108 women).
They had an average age of 32.6 years; 40% had gestational hypertension, and 60% had preeclampsia.
Women in the usual-care group typically received a BP review at 7-10 days after hospital discharge with a community midwife, and another at 6-8 weeks with their general practitioner.
The women in the intervention group were given and taught to use a Bluetooth-enabled OMRON Evolv BP monitor (Omron Healthcare Europe) while on the postnatal ward, and they installed a smartphone app on their mobile phones that transmitted self-monitored BP readings to a National Health Service-hosted, web-based platform.
They were instructed to take daily BP measurements (twice daily if out of target range). Dose titration of antihypertensives after hospital discharge was guided remotely by research clinicians, according to a guideline-based algorithm.
Patients in both groups had four study visits when their BP was measured: visit 1 (baseline) between days 1 and 6 post partum; visit 2 at week 1; visit 3 at week 6; and visit 4 between months 6 and 9 post partum.
Similar antihypertensive classes were prescribed in each group (enalapril 57%, nifedipine 27%, and labetalol 30% for intervention vs. enalapril 43%, nifedipine 30%, and labetalol 27% for control).
At 6 weeks, approximately 30% of participants in each group were still taking medication; this dropped to approximately 12% by visit 4.
The primary outcome – the mean 24-hour diastolic BP at visit 4 (roughly 9 months post partum), adjusted for baseline postnatal diastolic blood pressure – was 5.8–mm Hg lower in the intervention group than in the control group (71.2 mm Hg vs. 76.6 mm Hg; P < .001).
Secondary outcomes – between-group differences in systolic BP at 9 months, BP-related postnatal admission, and cardiac remodeling assessed by cardiac magnetic resonance – were all better in the intervention group.
The mean 24-hour average systolic BP at 9 months post partum, adjusted for baseline postnatal systolic BP was 6.5–mm Hg lower in the intervention group than in the control group (114.0 mm Hg vs. 120.3 mm Hg; P < .001).
There was an absolute risk reduction of 20% and a relative risk reduction of 73.5% in postnatal readmission. The number needed to treat to avoid one postnatal readmission was five, which “has potential for big cost savings,” said Dr. Leeson.
Blood pressure post partum can be improved with self-monitoring and physician-guided medication adjustment, Dr. Leeson summarized. The blood pressure remains low for at least 9 months, even when medication is stopped, and the intervention leads to beneficial cardiac remodeling.
U.S. pilot study
Non-Hispanic Black adults have a high hypertension and cardiovascular disease burden, and a related small U.S. study showed benefits of BP self-monitoring in a population comprising mainly Black women, Keith Ferdinand, MD, discussant of the POP-HT trial in the press briefing, said in an interview.
Dr. Ferdinand, from Tulane University, New Orleans, Louisiana, was lead author of the Text My Hypertension BP Meds NOLA pilot study that was published in February in the American Heart Journal Plus: Cardiology Research and Practice.
The study showed that text-messaging and social support increased hypertension medication adherence.
They enrolled 36 individuals, of whom 32 (89%) were non-Hispanic Black, and 23 (64%) were women. The participants received validated Bluetooth-enabled BP-monitoring devices that were synced to smartphones via a secured cloud-based application. The participants could send and receive messages to health care practitioners.
This intervention significantly improved medication adherence and systolic BP without modifying pharmacotherapy.
‘Need to be passionate about monitoring BP’
“The take-home messages from these exciting findings is that physicians and women who have had high BP during pregnancy need to be passionate about monitoring and controlling their blood pressure and not ignore it,” Anastasia Mihailidou, PhD, Royal North Shore Hospital, Sydney, the assigned discussant in the late-breaking trial session, said in an interview.
“It also resulted in fewer postpartum hospital readmissions for high blood pressure and benefit at 9 months in the structure and function of the heart and blood vessels of the women,” she said.
“While we need to see further studies in ethnically diverse women to see that they are reproducible, there are simple measures that clinicians can implement, and women can ask to have their BP monitored more frequently than the current practice. In the U.K. it is 5-10 days after delivery and then at 6-8 weeks after giving birth when changes in heart structure have already started,” Dr. Mihailidou noted.
“The procedure will need to be modified if there are no telemedicine facilities, but that should not stop having close monitoring of BP and treating it adequately. Monitoring requires an accurate BP monitor. There also has to be monitoring BP for the children.”
The trial was funded by a BHF Clinical Research Training Fellowship to Dr. Kitt, with additional support from the NIHR Oxford Biomedical Research Centre and Oxford BHF Centre for Research Excellence.
A version of this article first appeared on Medscape.com.
FROM AHA 2023
Apixaban cuts stroke but ups bleeding in subclinical AFib: ARTESIA
in the ARTESIA study.
The results appear to contrast somewhat with the recently reported NOAH-AFNET 6 trial, which failed to show a reduction in stroke with the anticoagulant edoxaban versus placebo in a similar patient group, but that trial was stopped early and so was underpowered.
However, the lead investigators of both trials say the studies actually show consistent results – both found a lower rate of stroke than expected in this population, but the confidence intervals for stroke reduction with anticoagulation overlap, suggesting there is likely some effect, albeit less than that in clinical AFib.
The big question is whether the reduction in stroke with anticoagulation outweighs the increase in major bleeding.
A new meta-analysis of the two trials showed that “oral anticoagulation with edoxaban or apixaban reduces the risk of ischemic stroke by approximately one-third and increases major bleeding by roughly double.”
In absolute numbers, there were three fewer ischemic strokes per 1,000 patient-years with anticoagulation in the two trials combined, at the cost of seven more major bleeds.
The lead investigators of the two trials have somewhat different opinions on how these findings may translate into clinical practice.
Jeff Healey, MD, Population Health Research Institute, McMaster University, Hamilton, Ont., lead investigator of the ARTESIA trial, believes that the risks and benefits need to be assessed in individual patients, but there should be some patient groups that will benefit from anticoagulation treatment.
“In patients with pacemakers or implantable loop recorders with continuous monitoring, subclinical AF[ib] is detected in about one third of patients, so this is extremely common,” he said in an interview. “The question is whether this is just a normal feature of getting older or is this like AF[ib] that we see in the clinic which increases stroke risk, and I think we can conclude from ARTESIA that this subclinical AF[ib] is associated with an increased risk of stroke, although that is lower than the risk with clinical AF[ib], and that it can be reduced by anticoagulation.”
Until recently it hasn’t been possible to quantify the risk associated with subclinical AFib, he noted. “But now we have a rich dataset to use to see if we can tease out some specifics on this. Future analyses of this dataset will help define patients where the benefits outweigh the risks of bleeding. For now, I think we can look at the data in a qualitative way and consider the totality of risk factors in each patient – their bleeding risk, stroke risk, how much AF[ib] they have, and make a decision as to whether to give anticoagulation or not.”
But Paulus Kirchhof, MD, University Heart and Vascular Center Hamburg (Germany), lead investigator of the NOAH-AFNET 6 trial said: “Both trials showed the stroke rate is low in these patients – about 1% per year – and that anticoagulation can reduce it a bit further at the expense of increasing major bleeding. I don’t believe the AF[ib] episodes picked up on these devices constitute a sufficient stroke risk to warrant anticoagulation, given the bleeding risk.”
Dr. Kirchhof suggests an alternate approach of performing further traditional AFib monitoring on these patients.
“I think going forward in my practice, when we come across this device-detected AF[ib], we will do further investigations with an established method for detecting AF[ib] involving surface ECG monitoring – maybe a 3-day or 7-day Holter. If that shows AF[ib], then we will be on firm ground to start anticoagulation. If that doesn’t show AF[ib], we will probably not use anticoagulation.”
The ARTESIA trial and the meta-analysis of the two trials were both presented at the annual scientific sessions of the American Heart Association. Both studies were also simultaneously published online – ARTESIA in the New England Journal of Medicine and the meta-analysis in Circulation.
ARTESIA
For the ARTESIA study, 4012 patients with device-detected AFib and other clinical risk factors for stroke were randomly assigned to treatment with apixaban (5 mg twice daily) or aspirin (81 mg daily).
After a mean follow-up of 3.5 years, the primary endpoint – stroke or systemic embolism – occurred in 55 patients in the apixaban group (0.78% per patient-year), compared with 86 patients in the aspirin group (1.24% per patient-year), giving a hazard ratio of 0.63 (95% confidence interval, 0.45-0.88; P = .007).
“The risk of stroke or systemic embolism was lower by 37% with apixaban than with aspirin, and the risk of disabling or fatal stroke was lower by 49%,” Dr. Healey reported.
In the “on-treatment” population, the rate of major bleeding was 1.71% per patient-year in the apixaban group and 0.94% per patient-year in the aspirin group (HR, 1.80; 95% CI, 1.26-2.57; P = .001).
Fatal bleeding occurred in five patients in the apixaban group and eight patients in the aspirin group. Symptomatic intracranial hemorrhage occurred in 12 patients with apixaban and 15 patients with aspirin.
One of the main findings of the trial is the lower-than-expected risk of ischemic stroke in this population – about 1% per year in the aspirin group, which was reduced to 0.64% per year in the apixaban group.
The authors noted that “simply counting strokes as compared with bleeding events might suggest a neutral overall effect. With apixaban as compared with aspirin, 31 fewer cases of stroke or systemic embolism were seen in the intention-to-treat analysis, as compared with 39 more major bleeding events in the on-treatment analysis.”
However, they pointed out that strokes involve permanent loss of brain tissue, whereas major bleeding is usually reversible, with most patients having complete recovery, which was the case in this study.
“Thus, on the basis of the considerably greater severity of the stroke events prevented than the bleeding events caused, we believe that these findings favor consideration of the use of oral anticoagulation for patients with risk factors for stroke in whom subclinical atrial fibrillation develops,” they concluded.
First well-powered trial addressing this question
Discussing the ARTESIA trial at an AHA press conference, Christine Albert, MD, Cedars-Sinai Medical Center, Los Angeles, said: “I want to emphasize how important this trial is.”
She explained that current guidelines do not recommend any treatment for patients with device-detected AFib that is not shown on ECG, even though it is known this confers some excess risk of stroke.
“ARTESIA is the first well-powered, long-term trial looking at this question,” she said. “It found a clear reduction in the risk of stroke/systemic embolism with apixaban vs aspirin, but there was also a significant amount of bleeding – about an 80% increase. The question is whether the benefit on stroke is worth it given the bleeding risk.”
Dr. Albert highlighted the low absolute risk of stroke in this study population of around 1.2%, pointing out that even with the 37% relative reduction with anticoagulation, stroke is only reduced in absolute terms by 0.4%.
“We are going to have to take this back to committees and guidelines and look at the balance between the benefit on stroke and the increase in bleeding,” she concluded.
Noting that observational studies have shown that the duration of AFib impacts the risk of stroke, Dr. Albert suggested that patients with longer-duration AFib may benefit from anticoagulation to a greater extent; and given that the bleeding seen in ARTESIA was mainly GI bleeding, it might be possible to screen out patients at high risk of GI bleeding.
She also pointed out that a lot of patients discontinued anticoagulation treatment in both ARTESIA and NOAH-AFNET 6, showing that this is not an easy strategy for elderly patients.
In an editorial accompanying publication of the ARTESIA trial, Emma Svennberg, MD, Karolinska Institute, Stockholm, also concluded that, “going forward, we must balance the increased bleeding risks with the risk for disabling strokes,” and that “future substudies and meta-analyses may provide further insights regarding treatment benefits in specific subgroups.”
NOAH-AFNET 6: New subgroup analysis
The previously reported NOAH-AFNET 6 study randomly assigned 2,538 patients with subclinical AFib and additional risk factors for stroke to anticoagulation with edoxaban or placebo. The trial was stopped early, so it was underpowered – but it found no difference between groups in the incidence of the composite endpoint of stroke, systemic embolism, or death from cardiovascular causes or in the incidence of stroke, although there was higher risk of major bleeding.
Again, there was a low rate of stroke in this trial with just 49 strokes in total in the whole study. The NOAH-AFNET-6 investigators concluded that these patients should not receive anticoagulation because the risk of bleeding outweighed any potential benefits.
A new subanalysis of the 259 patients who had durations of subclinical AFib of 24 hours or longer in the NOAH-AFNET 6 trial was presented at the AHA meeting, and simultaneously published online in the European Heart Journal.
This showed that the rate of stroke also appeared low in patients with these long durations of subclinical AFib, and that there was no interaction between the duration of subclinical AFib and the efficacy and safety of oral anticoagulation.
But with such a low number of events in the study as a whole and in the long duration subclinical AFib subgroup (in which there were just two strokes in each treatment group), this analysis was unlikely to show a difference, Dr. Kirchhof commented.
The subgroup analysis did, however, show that patients experiencing subclinical AFib durations of 24 hours or more were more likely to develop clinical AFib over time than those with shorter durations, suggesting the need for regular ECGs in these patients.
Dr. Kirchhof said better methods are needed to detect patients with subclinical AFib at high risk of stroke. “I don’t think our clinical stroke risk factor scores such as CHA2DS2-VASc are sufficient to detect high-risk patients. Patients in both NOAH-AFNET 6 and ARTESIA had a median CHA2DS2-VASc score of 4, but they had a stroke rate of just 1% per year,” he noted.
The meta-analysis of the two trials showed that the results from both are consistent, with an overall reduction in ischemic stroke with oral anticoagulation (relative risk, 0.68). Oral anticoagulation also reduced a composite of cardiovascular death, all-cause stroke, peripheral arterial embolism, myocardial infarction, or pulmonary embolism (RR, 0.85).
There was no significant difference in cardiovascular death (RR, 0.95) or all-cause mortality (RR, 1.08), but anticoagulation significantly increased major bleeding (RR, 1.62).
Aspirin use complicates results
Dr. Healey said further analyses of the ARTESIA data will try to tease out the effect of concomitant aspirin use in the trial.
He explained that patients in this trial were allowed to take a single antiplatelet agent on top of study therapy.
“It is difficult to work out the exact use of antiplatelet therapy as it changed throughout the study,” he said. “About two-thirds were taking antiplatelet agents at the time of enrollment into the trial, but this decreased throughout the study. Many clinicians stopped open-label antiplatelet therapy during the trial when new evidence came out to suggest that there was no added benefit of adding aspirin on top of anticoagulants.
“We need to look carefully as to what impact that may have had,” Dr. Healey added. “We know from other studies that adding an antiplatelet on top of an anticoagulant doesn’t do much to thromboembolic events, but it approximately doubles the risk of major bleeding.”
In contrast, the NOAH-AFNET trial did not allow aspirin use in the anticoagulation group and aspirin was taken by around half the patients in the placebo group who had an indication for its use.
The authors of the meta-analysis pointed out that the omission of aspirin in nearly half of the control patients in NOAH-AFNET 6 and the early termination of the trial may have led to a slightly higher estimate for excess major bleeding with anticoagulation.
The ARTESIA study was supported by the Canadian Institutes for Health Research, the Bristol Myers Squibb-Pfizer Alliance, the Heart and Stroke Foundation of Canada, the Canadian Stroke Prevention Intervention Network, Hamilton Health Sciences, the Advancing Clinical Trials Network and the Population Health Research Institute. Dr. Healey reported research grants and speaking fees from BMS/Pfizer Alliance, Servier, Novartis, Boston Scientific, Medtronic; and acts as a consultant to Bayer, Servier and Boston Scientific. The NOAH-AFNET 6 trial was an investigator-initiated trial funded by the German Center for Cardiovascular Research and Daiichi Sankyo Europe. Dr. Kirchhof reported research support from several drug and device companies active in AFib. He is also listed as an inventor on two patents held by the University of Hamburg on AFib therapy and AFib markers.
A version of this article first appeared on Medscape.com.
in the ARTESIA study.
The results appear to contrast somewhat with the recently reported NOAH-AFNET 6 trial, which failed to show a reduction in stroke with the anticoagulant edoxaban versus placebo in a similar patient group, but that trial was stopped early and so was underpowered.
However, the lead investigators of both trials say the studies actually show consistent results – both found a lower rate of stroke than expected in this population, but the confidence intervals for stroke reduction with anticoagulation overlap, suggesting there is likely some effect, albeit less than that in clinical AFib.
The big question is whether the reduction in stroke with anticoagulation outweighs the increase in major bleeding.
A new meta-analysis of the two trials showed that “oral anticoagulation with edoxaban or apixaban reduces the risk of ischemic stroke by approximately one-third and increases major bleeding by roughly double.”
In absolute numbers, there were three fewer ischemic strokes per 1,000 patient-years with anticoagulation in the two trials combined, at the cost of seven more major bleeds.
The lead investigators of the two trials have somewhat different opinions on how these findings may translate into clinical practice.
Jeff Healey, MD, Population Health Research Institute, McMaster University, Hamilton, Ont., lead investigator of the ARTESIA trial, believes that the risks and benefits need to be assessed in individual patients, but there should be some patient groups that will benefit from anticoagulation treatment.
“In patients with pacemakers or implantable loop recorders with continuous monitoring, subclinical AF[ib] is detected in about one third of patients, so this is extremely common,” he said in an interview. “The question is whether this is just a normal feature of getting older or is this like AF[ib] that we see in the clinic which increases stroke risk, and I think we can conclude from ARTESIA that this subclinical AF[ib] is associated with an increased risk of stroke, although that is lower than the risk with clinical AF[ib], and that it can be reduced by anticoagulation.”
Until recently it hasn’t been possible to quantify the risk associated with subclinical AFib, he noted. “But now we have a rich dataset to use to see if we can tease out some specifics on this. Future analyses of this dataset will help define patients where the benefits outweigh the risks of bleeding. For now, I think we can look at the data in a qualitative way and consider the totality of risk factors in each patient – their bleeding risk, stroke risk, how much AF[ib] they have, and make a decision as to whether to give anticoagulation or not.”
But Paulus Kirchhof, MD, University Heart and Vascular Center Hamburg (Germany), lead investigator of the NOAH-AFNET 6 trial said: “Both trials showed the stroke rate is low in these patients – about 1% per year – and that anticoagulation can reduce it a bit further at the expense of increasing major bleeding. I don’t believe the AF[ib] episodes picked up on these devices constitute a sufficient stroke risk to warrant anticoagulation, given the bleeding risk.”
Dr. Kirchhof suggests an alternate approach of performing further traditional AFib monitoring on these patients.
“I think going forward in my practice, when we come across this device-detected AF[ib], we will do further investigations with an established method for detecting AF[ib] involving surface ECG monitoring – maybe a 3-day or 7-day Holter. If that shows AF[ib], then we will be on firm ground to start anticoagulation. If that doesn’t show AF[ib], we will probably not use anticoagulation.”
The ARTESIA trial and the meta-analysis of the two trials were both presented at the annual scientific sessions of the American Heart Association. Both studies were also simultaneously published online – ARTESIA in the New England Journal of Medicine and the meta-analysis in Circulation.
ARTESIA
For the ARTESIA study, 4012 patients with device-detected AFib and other clinical risk factors for stroke were randomly assigned to treatment with apixaban (5 mg twice daily) or aspirin (81 mg daily).
After a mean follow-up of 3.5 years, the primary endpoint – stroke or systemic embolism – occurred in 55 patients in the apixaban group (0.78% per patient-year), compared with 86 patients in the aspirin group (1.24% per patient-year), giving a hazard ratio of 0.63 (95% confidence interval, 0.45-0.88; P = .007).
“The risk of stroke or systemic embolism was lower by 37% with apixaban than with aspirin, and the risk of disabling or fatal stroke was lower by 49%,” Dr. Healey reported.
In the “on-treatment” population, the rate of major bleeding was 1.71% per patient-year in the apixaban group and 0.94% per patient-year in the aspirin group (HR, 1.80; 95% CI, 1.26-2.57; P = .001).
Fatal bleeding occurred in five patients in the apixaban group and eight patients in the aspirin group. Symptomatic intracranial hemorrhage occurred in 12 patients with apixaban and 15 patients with aspirin.
One of the main findings of the trial is the lower-than-expected risk of ischemic stroke in this population – about 1% per year in the aspirin group, which was reduced to 0.64% per year in the apixaban group.
The authors noted that “simply counting strokes as compared with bleeding events might suggest a neutral overall effect. With apixaban as compared with aspirin, 31 fewer cases of stroke or systemic embolism were seen in the intention-to-treat analysis, as compared with 39 more major bleeding events in the on-treatment analysis.”
However, they pointed out that strokes involve permanent loss of brain tissue, whereas major bleeding is usually reversible, with most patients having complete recovery, which was the case in this study.
“Thus, on the basis of the considerably greater severity of the stroke events prevented than the bleeding events caused, we believe that these findings favor consideration of the use of oral anticoagulation for patients with risk factors for stroke in whom subclinical atrial fibrillation develops,” they concluded.
First well-powered trial addressing this question
Discussing the ARTESIA trial at an AHA press conference, Christine Albert, MD, Cedars-Sinai Medical Center, Los Angeles, said: “I want to emphasize how important this trial is.”
She explained that current guidelines do not recommend any treatment for patients with device-detected AFib that is not shown on ECG, even though it is known this confers some excess risk of stroke.
“ARTESIA is the first well-powered, long-term trial looking at this question,” she said. “It found a clear reduction in the risk of stroke/systemic embolism with apixaban vs aspirin, but there was also a significant amount of bleeding – about an 80% increase. The question is whether the benefit on stroke is worth it given the bleeding risk.”
Dr. Albert highlighted the low absolute risk of stroke in this study population of around 1.2%, pointing out that even with the 37% relative reduction with anticoagulation, stroke is only reduced in absolute terms by 0.4%.
“We are going to have to take this back to committees and guidelines and look at the balance between the benefit on stroke and the increase in bleeding,” she concluded.
Noting that observational studies have shown that the duration of AFib impacts the risk of stroke, Dr. Albert suggested that patients with longer-duration AFib may benefit from anticoagulation to a greater extent; and given that the bleeding seen in ARTESIA was mainly GI bleeding, it might be possible to screen out patients at high risk of GI bleeding.
She also pointed out that a lot of patients discontinued anticoagulation treatment in both ARTESIA and NOAH-AFNET 6, showing that this is not an easy strategy for elderly patients.
In an editorial accompanying publication of the ARTESIA trial, Emma Svennberg, MD, Karolinska Institute, Stockholm, also concluded that, “going forward, we must balance the increased bleeding risks with the risk for disabling strokes,” and that “future substudies and meta-analyses may provide further insights regarding treatment benefits in specific subgroups.”
NOAH-AFNET 6: New subgroup analysis
The previously reported NOAH-AFNET 6 study randomly assigned 2,538 patients with subclinical AFib and additional risk factors for stroke to anticoagulation with edoxaban or placebo. The trial was stopped early, so it was underpowered – but it found no difference between groups in the incidence of the composite endpoint of stroke, systemic embolism, or death from cardiovascular causes or in the incidence of stroke, although there was higher risk of major bleeding.
Again, there was a low rate of stroke in this trial with just 49 strokes in total in the whole study. The NOAH-AFNET-6 investigators concluded that these patients should not receive anticoagulation because the risk of bleeding outweighed any potential benefits.
A new subanalysis of the 259 patients who had durations of subclinical AFib of 24 hours or longer in the NOAH-AFNET 6 trial was presented at the AHA meeting, and simultaneously published online in the European Heart Journal.
This showed that the rate of stroke also appeared low in patients with these long durations of subclinical AFib, and that there was no interaction between the duration of subclinical AFib and the efficacy and safety of oral anticoagulation.
But with such a low number of events in the study as a whole and in the long duration subclinical AFib subgroup (in which there were just two strokes in each treatment group), this analysis was unlikely to show a difference, Dr. Kirchhof commented.
The subgroup analysis did, however, show that patients experiencing subclinical AFib durations of 24 hours or more were more likely to develop clinical AFib over time than those with shorter durations, suggesting the need for regular ECGs in these patients.
Dr. Kirchhof said better methods are needed to detect patients with subclinical AFib at high risk of stroke. “I don’t think our clinical stroke risk factor scores such as CHA2DS2-VASc are sufficient to detect high-risk patients. Patients in both NOAH-AFNET 6 and ARTESIA had a median CHA2DS2-VASc score of 4, but they had a stroke rate of just 1% per year,” he noted.
The meta-analysis of the two trials showed that the results from both are consistent, with an overall reduction in ischemic stroke with oral anticoagulation (relative risk, 0.68). Oral anticoagulation also reduced a composite of cardiovascular death, all-cause stroke, peripheral arterial embolism, myocardial infarction, or pulmonary embolism (RR, 0.85).
There was no significant difference in cardiovascular death (RR, 0.95) or all-cause mortality (RR, 1.08), but anticoagulation significantly increased major bleeding (RR, 1.62).
Aspirin use complicates results
Dr. Healey said further analyses of the ARTESIA data will try to tease out the effect of concomitant aspirin use in the trial.
He explained that patients in this trial were allowed to take a single antiplatelet agent on top of study therapy.
“It is difficult to work out the exact use of antiplatelet therapy as it changed throughout the study,” he said. “About two-thirds were taking antiplatelet agents at the time of enrollment into the trial, but this decreased throughout the study. Many clinicians stopped open-label antiplatelet therapy during the trial when new evidence came out to suggest that there was no added benefit of adding aspirin on top of anticoagulants.
“We need to look carefully as to what impact that may have had,” Dr. Healey added. “We know from other studies that adding an antiplatelet on top of an anticoagulant doesn’t do much to thromboembolic events, but it approximately doubles the risk of major bleeding.”
In contrast, the NOAH-AFNET trial did not allow aspirin use in the anticoagulation group and aspirin was taken by around half the patients in the placebo group who had an indication for its use.
The authors of the meta-analysis pointed out that the omission of aspirin in nearly half of the control patients in NOAH-AFNET 6 and the early termination of the trial may have led to a slightly higher estimate for excess major bleeding with anticoagulation.
The ARTESIA study was supported by the Canadian Institutes for Health Research, the Bristol Myers Squibb-Pfizer Alliance, the Heart and Stroke Foundation of Canada, the Canadian Stroke Prevention Intervention Network, Hamilton Health Sciences, the Advancing Clinical Trials Network and the Population Health Research Institute. Dr. Healey reported research grants and speaking fees from BMS/Pfizer Alliance, Servier, Novartis, Boston Scientific, Medtronic; and acts as a consultant to Bayer, Servier and Boston Scientific. The NOAH-AFNET 6 trial was an investigator-initiated trial funded by the German Center for Cardiovascular Research and Daiichi Sankyo Europe. Dr. Kirchhof reported research support from several drug and device companies active in AFib. He is also listed as an inventor on two patents held by the University of Hamburg on AFib therapy and AFib markers.
A version of this article first appeared on Medscape.com.
in the ARTESIA study.
The results appear to contrast somewhat with the recently reported NOAH-AFNET 6 trial, which failed to show a reduction in stroke with the anticoagulant edoxaban versus placebo in a similar patient group, but that trial was stopped early and so was underpowered.
However, the lead investigators of both trials say the studies actually show consistent results – both found a lower rate of stroke than expected in this population, but the confidence intervals for stroke reduction with anticoagulation overlap, suggesting there is likely some effect, albeit less than that in clinical AFib.
The big question is whether the reduction in stroke with anticoagulation outweighs the increase in major bleeding.
A new meta-analysis of the two trials showed that “oral anticoagulation with edoxaban or apixaban reduces the risk of ischemic stroke by approximately one-third and increases major bleeding by roughly double.”
In absolute numbers, there were three fewer ischemic strokes per 1,000 patient-years with anticoagulation in the two trials combined, at the cost of seven more major bleeds.
The lead investigators of the two trials have somewhat different opinions on how these findings may translate into clinical practice.
Jeff Healey, MD, Population Health Research Institute, McMaster University, Hamilton, Ont., lead investigator of the ARTESIA trial, believes that the risks and benefits need to be assessed in individual patients, but there should be some patient groups that will benefit from anticoagulation treatment.
“In patients with pacemakers or implantable loop recorders with continuous monitoring, subclinical AF[ib] is detected in about one third of patients, so this is extremely common,” he said in an interview. “The question is whether this is just a normal feature of getting older or is this like AF[ib] that we see in the clinic which increases stroke risk, and I think we can conclude from ARTESIA that this subclinical AF[ib] is associated with an increased risk of stroke, although that is lower than the risk with clinical AF[ib], and that it can be reduced by anticoagulation.”
Until recently it hasn’t been possible to quantify the risk associated with subclinical AFib, he noted. “But now we have a rich dataset to use to see if we can tease out some specifics on this. Future analyses of this dataset will help define patients where the benefits outweigh the risks of bleeding. For now, I think we can look at the data in a qualitative way and consider the totality of risk factors in each patient – their bleeding risk, stroke risk, how much AF[ib] they have, and make a decision as to whether to give anticoagulation or not.”
But Paulus Kirchhof, MD, University Heart and Vascular Center Hamburg (Germany), lead investigator of the NOAH-AFNET 6 trial said: “Both trials showed the stroke rate is low in these patients – about 1% per year – and that anticoagulation can reduce it a bit further at the expense of increasing major bleeding. I don’t believe the AF[ib] episodes picked up on these devices constitute a sufficient stroke risk to warrant anticoagulation, given the bleeding risk.”
Dr. Kirchhof suggests an alternate approach of performing further traditional AFib monitoring on these patients.
“I think going forward in my practice, when we come across this device-detected AF[ib], we will do further investigations with an established method for detecting AF[ib] involving surface ECG monitoring – maybe a 3-day or 7-day Holter. If that shows AF[ib], then we will be on firm ground to start anticoagulation. If that doesn’t show AF[ib], we will probably not use anticoagulation.”
The ARTESIA trial and the meta-analysis of the two trials were both presented at the annual scientific sessions of the American Heart Association. Both studies were also simultaneously published online – ARTESIA in the New England Journal of Medicine and the meta-analysis in Circulation.
ARTESIA
For the ARTESIA study, 4012 patients with device-detected AFib and other clinical risk factors for stroke were randomly assigned to treatment with apixaban (5 mg twice daily) or aspirin (81 mg daily).
After a mean follow-up of 3.5 years, the primary endpoint – stroke or systemic embolism – occurred in 55 patients in the apixaban group (0.78% per patient-year), compared with 86 patients in the aspirin group (1.24% per patient-year), giving a hazard ratio of 0.63 (95% confidence interval, 0.45-0.88; P = .007).
“The risk of stroke or systemic embolism was lower by 37% with apixaban than with aspirin, and the risk of disabling or fatal stroke was lower by 49%,” Dr. Healey reported.
In the “on-treatment” population, the rate of major bleeding was 1.71% per patient-year in the apixaban group and 0.94% per patient-year in the aspirin group (HR, 1.80; 95% CI, 1.26-2.57; P = .001).
Fatal bleeding occurred in five patients in the apixaban group and eight patients in the aspirin group. Symptomatic intracranial hemorrhage occurred in 12 patients with apixaban and 15 patients with aspirin.
One of the main findings of the trial is the lower-than-expected risk of ischemic stroke in this population – about 1% per year in the aspirin group, which was reduced to 0.64% per year in the apixaban group.
The authors noted that “simply counting strokes as compared with bleeding events might suggest a neutral overall effect. With apixaban as compared with aspirin, 31 fewer cases of stroke or systemic embolism were seen in the intention-to-treat analysis, as compared with 39 more major bleeding events in the on-treatment analysis.”
However, they pointed out that strokes involve permanent loss of brain tissue, whereas major bleeding is usually reversible, with most patients having complete recovery, which was the case in this study.
“Thus, on the basis of the considerably greater severity of the stroke events prevented than the bleeding events caused, we believe that these findings favor consideration of the use of oral anticoagulation for patients with risk factors for stroke in whom subclinical atrial fibrillation develops,” they concluded.
First well-powered trial addressing this question
Discussing the ARTESIA trial at an AHA press conference, Christine Albert, MD, Cedars-Sinai Medical Center, Los Angeles, said: “I want to emphasize how important this trial is.”
She explained that current guidelines do not recommend any treatment for patients with device-detected AFib that is not shown on ECG, even though it is known this confers some excess risk of stroke.
“ARTESIA is the first well-powered, long-term trial looking at this question,” she said. “It found a clear reduction in the risk of stroke/systemic embolism with apixaban vs aspirin, but there was also a significant amount of bleeding – about an 80% increase. The question is whether the benefit on stroke is worth it given the bleeding risk.”
Dr. Albert highlighted the low absolute risk of stroke in this study population of around 1.2%, pointing out that even with the 37% relative reduction with anticoagulation, stroke is only reduced in absolute terms by 0.4%.
“We are going to have to take this back to committees and guidelines and look at the balance between the benefit on stroke and the increase in bleeding,” she concluded.
Noting that observational studies have shown that the duration of AFib impacts the risk of stroke, Dr. Albert suggested that patients with longer-duration AFib may benefit from anticoagulation to a greater extent; and given that the bleeding seen in ARTESIA was mainly GI bleeding, it might be possible to screen out patients at high risk of GI bleeding.
She also pointed out that a lot of patients discontinued anticoagulation treatment in both ARTESIA and NOAH-AFNET 6, showing that this is not an easy strategy for elderly patients.
In an editorial accompanying publication of the ARTESIA trial, Emma Svennberg, MD, Karolinska Institute, Stockholm, also concluded that, “going forward, we must balance the increased bleeding risks with the risk for disabling strokes,” and that “future substudies and meta-analyses may provide further insights regarding treatment benefits in specific subgroups.”
NOAH-AFNET 6: New subgroup analysis
The previously reported NOAH-AFNET 6 study randomly assigned 2,538 patients with subclinical AFib and additional risk factors for stroke to anticoagulation with edoxaban or placebo. The trial was stopped early, so it was underpowered – but it found no difference between groups in the incidence of the composite endpoint of stroke, systemic embolism, or death from cardiovascular causes or in the incidence of stroke, although there was higher risk of major bleeding.
Again, there was a low rate of stroke in this trial with just 49 strokes in total in the whole study. The NOAH-AFNET-6 investigators concluded that these patients should not receive anticoagulation because the risk of bleeding outweighed any potential benefits.
A new subanalysis of the 259 patients who had durations of subclinical AFib of 24 hours or longer in the NOAH-AFNET 6 trial was presented at the AHA meeting, and simultaneously published online in the European Heart Journal.
This showed that the rate of stroke also appeared low in patients with these long durations of subclinical AFib, and that there was no interaction between the duration of subclinical AFib and the efficacy and safety of oral anticoagulation.
But with such a low number of events in the study as a whole and in the long duration subclinical AFib subgroup (in which there were just two strokes in each treatment group), this analysis was unlikely to show a difference, Dr. Kirchhof commented.
The subgroup analysis did, however, show that patients experiencing subclinical AFib durations of 24 hours or more were more likely to develop clinical AFib over time than those with shorter durations, suggesting the need for regular ECGs in these patients.
Dr. Kirchhof said better methods are needed to detect patients with subclinical AFib at high risk of stroke. “I don’t think our clinical stroke risk factor scores such as CHA2DS2-VASc are sufficient to detect high-risk patients. Patients in both NOAH-AFNET 6 and ARTESIA had a median CHA2DS2-VASc score of 4, but they had a stroke rate of just 1% per year,” he noted.
The meta-analysis of the two trials showed that the results from both are consistent, with an overall reduction in ischemic stroke with oral anticoagulation (relative risk, 0.68). Oral anticoagulation also reduced a composite of cardiovascular death, all-cause stroke, peripheral arterial embolism, myocardial infarction, or pulmonary embolism (RR, 0.85).
There was no significant difference in cardiovascular death (RR, 0.95) or all-cause mortality (RR, 1.08), but anticoagulation significantly increased major bleeding (RR, 1.62).
Aspirin use complicates results
Dr. Healey said further analyses of the ARTESIA data will try to tease out the effect of concomitant aspirin use in the trial.
He explained that patients in this trial were allowed to take a single antiplatelet agent on top of study therapy.
“It is difficult to work out the exact use of antiplatelet therapy as it changed throughout the study,” he said. “About two-thirds were taking antiplatelet agents at the time of enrollment into the trial, but this decreased throughout the study. Many clinicians stopped open-label antiplatelet therapy during the trial when new evidence came out to suggest that there was no added benefit of adding aspirin on top of anticoagulants.
“We need to look carefully as to what impact that may have had,” Dr. Healey added. “We know from other studies that adding an antiplatelet on top of an anticoagulant doesn’t do much to thromboembolic events, but it approximately doubles the risk of major bleeding.”
In contrast, the NOAH-AFNET trial did not allow aspirin use in the anticoagulation group and aspirin was taken by around half the patients in the placebo group who had an indication for its use.
The authors of the meta-analysis pointed out that the omission of aspirin in nearly half of the control patients in NOAH-AFNET 6 and the early termination of the trial may have led to a slightly higher estimate for excess major bleeding with anticoagulation.
The ARTESIA study was supported by the Canadian Institutes for Health Research, the Bristol Myers Squibb-Pfizer Alliance, the Heart and Stroke Foundation of Canada, the Canadian Stroke Prevention Intervention Network, Hamilton Health Sciences, the Advancing Clinical Trials Network and the Population Health Research Institute. Dr. Healey reported research grants and speaking fees from BMS/Pfizer Alliance, Servier, Novartis, Boston Scientific, Medtronic; and acts as a consultant to Bayer, Servier and Boston Scientific. The NOAH-AFNET 6 trial was an investigator-initiated trial funded by the German Center for Cardiovascular Research and Daiichi Sankyo Europe. Dr. Kirchhof reported research support from several drug and device companies active in AFib. He is also listed as an inventor on two patents held by the University of Hamburg on AFib therapy and AFib markers.
A version of this article first appeared on Medscape.com.
FROM AHA 2023
Chemotherapy-induced peripheral neuropathy tied to compromised executive function
PHOENIX – , a new finding that may increase the risk for compromised mobility and fall risk.
“Among older cancer survivors treated with chemotherapy, the presence of CIPN was independently associated with reduced executive function,” said study investigator Brendan L. McNeish, MD, of the department of physical medicine and rehabilitation, University of Pittsburgh.
“Importantly, given the emerging relationship of executive function with mobility in this population, stakeholders and clinicians are called to acknowledge that chemotherapy-related mobility declines in CIPN survivors are likely due to both neuromuscular and executive dysfunction,” he said.
The findings were presented at the 2023 annual meeting of the American Association for Neuromuscular and Electrodiagnostic Medicine (AANEM).
Research gap
Characterized by numbness, tingling, pain, and motor impairment, CIPN affects up to 50% of all patients with cancer that is treated with taxane-, platinum-, or vinca alkaloid–based chemotherapy. The condition is among the leading dose-limiting toxicities, potentially increasing mortality risk.
Though the effects of chemotherapy on cognitive function are well-established, less is known about a potential relationship between this side effect and CIPN, Dr. McNeish said.
“Chemotherapy can be neurotoxic, but few studies have linked neurotoxicity to the central nervous system and peripheral nervous system,” Dr. McNeish said.
To compare cognitive outcomes in patients treated with chemotherapy who did and did not develop CIPN, the investigators conducted a cross-sectional study that included 50 chemotherapy-treated cancer survivors at a single time point post chemotherapy. The mean age of participants was 65.6 years, and 90% were women.
Twenty-two (44%) patients had CIPN on the basis of patient-reported distal paresthesias or numbness that started when chemotherapy was initiated and was present at the time of study enrollment.
Patients with CIPN had a greater decline in executive function, compared with those without the condition, as measured by the Trail Making Test Part B (TMT-B; CIPN-positive, 84.9 sec vs. 59.1 sec, respectively; P = .01) and the Stroop Color and Word Test (SCWT; CIPN-positive, 178.1 sec vs. CIPN-negative, 152.7 sec; P = .04), as well as lower rapid reaction accuracy (CIPN-positive, 60.3% vs. CIPN-negative, 70.6%; P = .01).
The association between CIPN and decreased executive function remained after multivariate adjusting for age, gender, depression, and benzodiazepine use for TMT-B (beta, 18.7; P = .046) and rapid reaction accuracy (beta, -.088; P = .018) but not SCWT (beta, 9.52; P = .233).
Clinical guidance
A recent study by the same investigators showed a link between executive function and balance in cancer survivors (mean age, 65.6 years; 88% women) treated with chemotherapy.
Another study of 116 patients treated with chemotherapy, including 32 who developed CIPN, showed that those with CIPN were nearly three times more likely to report a fall or near fall than were those without CIPN symptoms. In addition, those with CIPN symptoms were also more likely to have received medical care for falls.
Based on the current findings, the research suggests that “current clinical approaches to caring for this growing population [of cancer patients] should not assume that the well-known increased fall risk is solely related to CIPN.”
Dr. McNeish speculated that two potential hypotheses could explain the association between CIPN and reduced executive function in older cancer survivors.
“First, CIPN is associated with other conditions such as depression and anxiety, which are associated with reduced executive function,” he said.
“The second is that cancer-related cognitive dysfunction and CIPN share pathogenic mechanisms of neuronal injury, inflammation, and advanced aging, and thus some patients are vulnerable to both central (cancer-related cognitive function) and peripheral neurotoxicity.”
Either way, Dr. McNeish noted that “all interventions should measure both CIPN and executive function, as one could confound the other.”
Need for increased awareness
Commenting on the study, Ting Bao, MD, co-director of the Leonard P. Zakim Center for Integrative Therapies & Healthy Living at the Dana-Farber Cancer Institute, Boston, said that the findings underscore that “there is a need for increased awareness of the diverse manifestations of chemotherapy-induced neuropathy.”
These include the fact that “neurotoxic chemotherapy impacts both the peripheral and central nervous systems, affecting balance through distinct mechanisms.”
Although treatments routinely recommended for CIPN include duloxetine, tricyclic antidepressants, or gabapentin as well as topical agents such as lidocaine, evidence also shows benefits of nonpharmacologic approaches including exercise, acupuncture, and yoga. Dr. Bao’s own research has suggested that those benefits can extend improved balance and reduced fall risk.
Dr. Bao and colleagues recently conducted a randomized study that included 41 patients with CIPN to receive either yoga or usual care.
“The findings revealed that after eight biweekly sessions of yoga, there was a notable improvement in the far-reach test, which is a predictor of fall risk,” she said.
To validate these findings, the researchers are currently conducting a larger randomized controlled trial, she said.
In the meantime, “further research into the mechanisms and effective treatments for chemotherapy-induced neurotoxicity is essential,” added Dr. Bao.
Dr. McNeish and Dr. Bao report no relevant disclosures.
A version of this article first appeared on Medscape.com.
PHOENIX – , a new finding that may increase the risk for compromised mobility and fall risk.
“Among older cancer survivors treated with chemotherapy, the presence of CIPN was independently associated with reduced executive function,” said study investigator Brendan L. McNeish, MD, of the department of physical medicine and rehabilitation, University of Pittsburgh.
“Importantly, given the emerging relationship of executive function with mobility in this population, stakeholders and clinicians are called to acknowledge that chemotherapy-related mobility declines in CIPN survivors are likely due to both neuromuscular and executive dysfunction,” he said.
The findings were presented at the 2023 annual meeting of the American Association for Neuromuscular and Electrodiagnostic Medicine (AANEM).
Research gap
Characterized by numbness, tingling, pain, and motor impairment, CIPN affects up to 50% of all patients with cancer that is treated with taxane-, platinum-, or vinca alkaloid–based chemotherapy. The condition is among the leading dose-limiting toxicities, potentially increasing mortality risk.
Though the effects of chemotherapy on cognitive function are well-established, less is known about a potential relationship between this side effect and CIPN, Dr. McNeish said.
“Chemotherapy can be neurotoxic, but few studies have linked neurotoxicity to the central nervous system and peripheral nervous system,” Dr. McNeish said.
To compare cognitive outcomes in patients treated with chemotherapy who did and did not develop CIPN, the investigators conducted a cross-sectional study that included 50 chemotherapy-treated cancer survivors at a single time point post chemotherapy. The mean age of participants was 65.6 years, and 90% were women.
Twenty-two (44%) patients had CIPN on the basis of patient-reported distal paresthesias or numbness that started when chemotherapy was initiated and was present at the time of study enrollment.
Patients with CIPN had a greater decline in executive function, compared with those without the condition, as measured by the Trail Making Test Part B (TMT-B; CIPN-positive, 84.9 sec vs. 59.1 sec, respectively; P = .01) and the Stroop Color and Word Test (SCWT; CIPN-positive, 178.1 sec vs. CIPN-negative, 152.7 sec; P = .04), as well as lower rapid reaction accuracy (CIPN-positive, 60.3% vs. CIPN-negative, 70.6%; P = .01).
The association between CIPN and decreased executive function remained after multivariate adjusting for age, gender, depression, and benzodiazepine use for TMT-B (beta, 18.7; P = .046) and rapid reaction accuracy (beta, -.088; P = .018) but not SCWT (beta, 9.52; P = .233).
Clinical guidance
A recent study by the same investigators showed a link between executive function and balance in cancer survivors (mean age, 65.6 years; 88% women) treated with chemotherapy.
Another study of 116 patients treated with chemotherapy, including 32 who developed CIPN, showed that those with CIPN were nearly three times more likely to report a fall or near fall than were those without CIPN symptoms. In addition, those with CIPN symptoms were also more likely to have received medical care for falls.
Based on the current findings, the research suggests that “current clinical approaches to caring for this growing population [of cancer patients] should not assume that the well-known increased fall risk is solely related to CIPN.”
Dr. McNeish speculated that two potential hypotheses could explain the association between CIPN and reduced executive function in older cancer survivors.
“First, CIPN is associated with other conditions such as depression and anxiety, which are associated with reduced executive function,” he said.
“The second is that cancer-related cognitive dysfunction and CIPN share pathogenic mechanisms of neuronal injury, inflammation, and advanced aging, and thus some patients are vulnerable to both central (cancer-related cognitive function) and peripheral neurotoxicity.”
Either way, Dr. McNeish noted that “all interventions should measure both CIPN and executive function, as one could confound the other.”
Need for increased awareness
Commenting on the study, Ting Bao, MD, co-director of the Leonard P. Zakim Center for Integrative Therapies & Healthy Living at the Dana-Farber Cancer Institute, Boston, said that the findings underscore that “there is a need for increased awareness of the diverse manifestations of chemotherapy-induced neuropathy.”
These include the fact that “neurotoxic chemotherapy impacts both the peripheral and central nervous systems, affecting balance through distinct mechanisms.”
Although treatments routinely recommended for CIPN include duloxetine, tricyclic antidepressants, or gabapentin as well as topical agents such as lidocaine, evidence also shows benefits of nonpharmacologic approaches including exercise, acupuncture, and yoga. Dr. Bao’s own research has suggested that those benefits can extend improved balance and reduced fall risk.
Dr. Bao and colleagues recently conducted a randomized study that included 41 patients with CIPN to receive either yoga or usual care.
“The findings revealed that after eight biweekly sessions of yoga, there was a notable improvement in the far-reach test, which is a predictor of fall risk,” she said.
To validate these findings, the researchers are currently conducting a larger randomized controlled trial, she said.
In the meantime, “further research into the mechanisms and effective treatments for chemotherapy-induced neurotoxicity is essential,” added Dr. Bao.
Dr. McNeish and Dr. Bao report no relevant disclosures.
A version of this article first appeared on Medscape.com.
PHOENIX – , a new finding that may increase the risk for compromised mobility and fall risk.
“Among older cancer survivors treated with chemotherapy, the presence of CIPN was independently associated with reduced executive function,” said study investigator Brendan L. McNeish, MD, of the department of physical medicine and rehabilitation, University of Pittsburgh.
“Importantly, given the emerging relationship of executive function with mobility in this population, stakeholders and clinicians are called to acknowledge that chemotherapy-related mobility declines in CIPN survivors are likely due to both neuromuscular and executive dysfunction,” he said.
The findings were presented at the 2023 annual meeting of the American Association for Neuromuscular and Electrodiagnostic Medicine (AANEM).
Research gap
Characterized by numbness, tingling, pain, and motor impairment, CIPN affects up to 50% of all patients with cancer that is treated with taxane-, platinum-, or vinca alkaloid–based chemotherapy. The condition is among the leading dose-limiting toxicities, potentially increasing mortality risk.
Though the effects of chemotherapy on cognitive function are well-established, less is known about a potential relationship between this side effect and CIPN, Dr. McNeish said.
“Chemotherapy can be neurotoxic, but few studies have linked neurotoxicity to the central nervous system and peripheral nervous system,” Dr. McNeish said.
To compare cognitive outcomes in patients treated with chemotherapy who did and did not develop CIPN, the investigators conducted a cross-sectional study that included 50 chemotherapy-treated cancer survivors at a single time point post chemotherapy. The mean age of participants was 65.6 years, and 90% were women.
Twenty-two (44%) patients had CIPN on the basis of patient-reported distal paresthesias or numbness that started when chemotherapy was initiated and was present at the time of study enrollment.
Patients with CIPN had a greater decline in executive function, compared with those without the condition, as measured by the Trail Making Test Part B (TMT-B; CIPN-positive, 84.9 sec vs. 59.1 sec, respectively; P = .01) and the Stroop Color and Word Test (SCWT; CIPN-positive, 178.1 sec vs. CIPN-negative, 152.7 sec; P = .04), as well as lower rapid reaction accuracy (CIPN-positive, 60.3% vs. CIPN-negative, 70.6%; P = .01).
The association between CIPN and decreased executive function remained after multivariate adjusting for age, gender, depression, and benzodiazepine use for TMT-B (beta, 18.7; P = .046) and rapid reaction accuracy (beta, -.088; P = .018) but not SCWT (beta, 9.52; P = .233).
Clinical guidance
A recent study by the same investigators showed a link between executive function and balance in cancer survivors (mean age, 65.6 years; 88% women) treated with chemotherapy.
Another study of 116 patients treated with chemotherapy, including 32 who developed CIPN, showed that those with CIPN were nearly three times more likely to report a fall or near fall than were those without CIPN symptoms. In addition, those with CIPN symptoms were also more likely to have received medical care for falls.
Based on the current findings, the research suggests that “current clinical approaches to caring for this growing population [of cancer patients] should not assume that the well-known increased fall risk is solely related to CIPN.”
Dr. McNeish speculated that two potential hypotheses could explain the association between CIPN and reduced executive function in older cancer survivors.
“First, CIPN is associated with other conditions such as depression and anxiety, which are associated with reduced executive function,” he said.
“The second is that cancer-related cognitive dysfunction and CIPN share pathogenic mechanisms of neuronal injury, inflammation, and advanced aging, and thus some patients are vulnerable to both central (cancer-related cognitive function) and peripheral neurotoxicity.”
Either way, Dr. McNeish noted that “all interventions should measure both CIPN and executive function, as one could confound the other.”
Need for increased awareness
Commenting on the study, Ting Bao, MD, co-director of the Leonard P. Zakim Center for Integrative Therapies & Healthy Living at the Dana-Farber Cancer Institute, Boston, said that the findings underscore that “there is a need for increased awareness of the diverse manifestations of chemotherapy-induced neuropathy.”
These include the fact that “neurotoxic chemotherapy impacts both the peripheral and central nervous systems, affecting balance through distinct mechanisms.”
Although treatments routinely recommended for CIPN include duloxetine, tricyclic antidepressants, or gabapentin as well as topical agents such as lidocaine, evidence also shows benefits of nonpharmacologic approaches including exercise, acupuncture, and yoga. Dr. Bao’s own research has suggested that those benefits can extend improved balance and reduced fall risk.
Dr. Bao and colleagues recently conducted a randomized study that included 41 patients with CIPN to receive either yoga or usual care.
“The findings revealed that after eight biweekly sessions of yoga, there was a notable improvement in the far-reach test, which is a predictor of fall risk,” she said.
To validate these findings, the researchers are currently conducting a larger randomized controlled trial, she said.
In the meantime, “further research into the mechanisms and effective treatments for chemotherapy-induced neurotoxicity is essential,” added Dr. Bao.
Dr. McNeish and Dr. Bao report no relevant disclosures.
A version of this article first appeared on Medscape.com.
AT AANEM 2023
Impressive bleeding profile with factor XI inhibitor in AFib: AZALEA
; the risk of stroke was moderate to high.
The trial was stopped earlier this year because of an “overwhelming” reduction in bleeding with abelacimab in comparison to rivaroxaban. Abelacimab is a monoclonal antibody given by subcutaneous injection once a month.
“Details of the bleeding results have now shown that the 150-mg dose of abelacimab, which is the dose being carried forward to phase 3 trials, was associated with a 67% reduction in major or clinically relevant nonmajor bleeding, the primary endpoint of the study.”
In addition, major bleeding was reduced by 74%, and major gastrointestinal bleeding was reduced by 93%.
“We are seeing really profound reductions in bleeding with this agent vs. a NOAC [novel oral anticoagulant],” lead AZALEA investigator Christian Ruff, MD, professor of medicine at Brigham and Women’s Hospital, Boston, said in an interview.
“Major bleeding – effectively the type of bleeding that results in hospitalization – is reduced by more than two-thirds, and major GI bleeding – which is the most common type of bleeding experienced by AF patients on anticoagulants – is almost eliminated. This gives us real hope that we have finally found an anticoagulant that is remarkably safe and will allow us to use anticoagulation in our most vulnerable patients,” he said.
Dr. Ruff presented the full results from the AZALEA trial at the American Heart Association scientific sessions.
He noted that AFib is one of the most common medical conditions in the world and that it confers an increased risk of stroke. Anticoagulants reduce this risk very effectively, and while the NOACS, such as apixaban and rivaroxaban, are safer than warfarin, significant bleeding still occurs, and “shockingly,” he said, between 30% and 60% of patients are not prescribed an anticoagulant or discontinue treatment because of bleeding concerns.
“Clearly, we need safer anticoagulants to protect these patients. Factor XI inhibitors, of which abelacimab is one, have emerged as the most promising agents, as they are thought to provide precision anticoagulation,” Dr. Ruff said.
He explained that factor XI appears to be involved in the formation of thrombus, which blocks arteries and causes strokes and myocardial infarction (thrombosis), but not in the healing process of blood vessels after injury (hemostasis). So, it is believed that inhibiting factor XI should reduce thrombotic events without causing excess bleeding.
AZALEA, which is the largest and longest trial of a factor XI inhibitor to date, enrolled 1,287 adults with AF who were at moderate to high risk of stroke.
They were randomly assigned to receive one of three treatments: oral rivaroxaban 20 mg daily; abelacimab 90 mg; or abelacimab 150 mg. Abelacimab was given monthly by injection.
Both doses of abelacimab inhibited factor XI almost completely; 97% inhibition was achieved with the 90-mg dose, and 99% inhibition was achieved with the 150-mg dose.
Results showed that after a median follow-up of 1.8 years, there was a clear reduction in all bleeding endpoints with both doses of abelacimab, compared with rivaroxaban.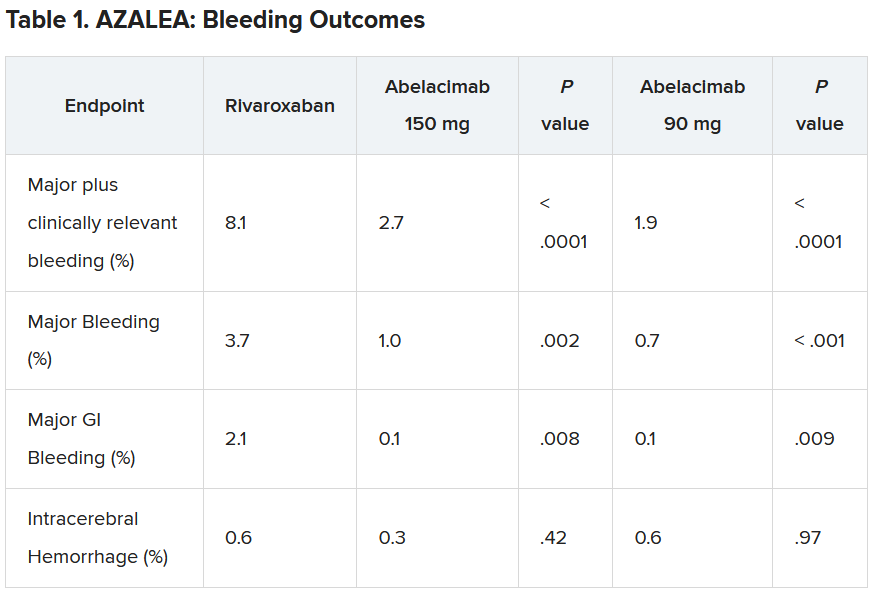
Dr. Ruff explained that the trial was powered to detect differences in bleeding, not stroke, but the investigators approached this in an exploratory way.
“As expected, the numbers were low, with just 25 strokes (23 ischemic strokes) across all three groups in the trial. So, because of this very low rate, we are really not able to compare how abelacimab compares with rivaroxaban in reducing stroke,” he commented.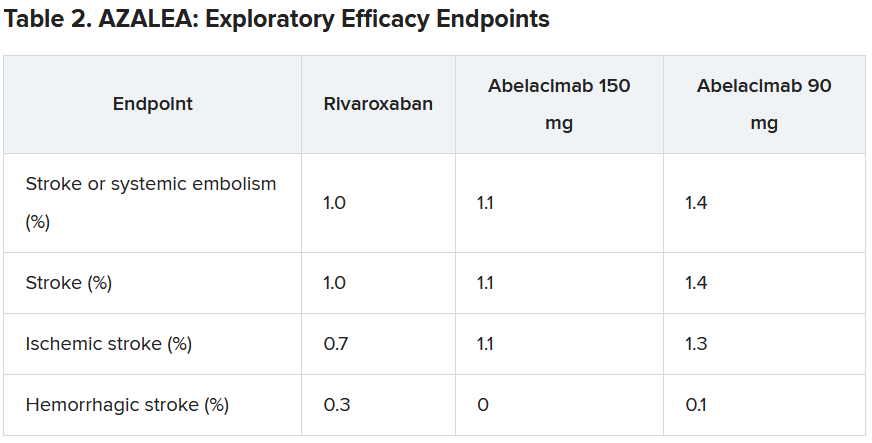
He did, however, suggest that the low stroke rate in the study was encouraging.
“If we look at the same population without anticoagulation, the stroke rate would be about 7% per year. And we see here in this trial that in all three arms, the stroke rate was just above 1% per year. I think this shows that all the patients in the trial were getting highly effective anticoagulation,” he said.
“But what this trial doesn’t answer – because the numbers are so low – is exactly how effective factor XI inhibition with abelacimab is, compared to NOACs in reducing stroke rates. That requires dedicated phase 3 trials.”
Dr. Ruff pointed out that there are some reassuring data from phase 2 trials in venous thromboembolism (VTE), in which the 150-mg dose of abelacimab was associated with an 80% reduction in VTE, compared with enoxaparin. “Historically in the development of anticoagulants, efficacy in VTE has translated into efficacy in stroke prevention, so that is very encouraging,” he commented.
“So, I think our results along with the VTE results are encouraging, but the precision regarding the relative efficacy compared to NOACs is still an open question that needs to be clarified in phase 3 trials,” he concluded.
Several phase 3 trials are now underway with abelacimab and two other small-molecule orally available factor XI inhibitors, milvexian (BMS/Janssen) and asundexian (Bayer).
The designated discussant of the AZALEA study at the AHA meeting, Manesh Patel. MD, Duke University, Durham, N.C., described the results as “an important step forward.”
“This trial, with the prior data in this field, show that factor XI inhibition as a target is biologically possible (studies showing > 95% inhibition), significantly less bleeding than NOACS. We await the phase 3 studies, but having significantly less bleeding and similar or less stroke would be a substantial step forward for the field,” he said.
John Alexander, MD, also from Duke University, said: “There were clinically important reductions in bleeding with both doses of abelacimab, compared with rivaroxaban. This is consistent to what we’ve seen with comparisons between other factor XI inhibitors and other factor Xa inhibitors.”
On the exploratory efficacy results, Dr. Alexander agreed with Dr. Ruff that it was not possible to get any idea of how abelacimab compared with rivaroxaban in reducing stroke. “The hazard ratio and confidence intervals comparing abelacimab and rivaroxaban include substantial lower rates, no difference, and substantially higher rates,” he noted.
“We need to wait for the results of phase 3 trials, with abelacimab and other factor XI inhibitors, to understand how well factor XI inhibition prevents stroke and systemic embolism in patients with atrial fibrillation,” Dr. Alexander added. “These trials are ongoing.”
Dr. Ruff concluded: “Assuming the data from ongoing phase 3 trials confirm the benefit of factor XI inhibitors for stroke prevention in people with AF, it will really be transformative for the field of cardiology.
“Our first mission in treating people with AF is to prevent stroke, and our ability to do this with a remarkably safe anticoagulant such as abelacimab would be an incredible advance,” he concluded.
Dr. Ruff receives research funding from Anthos for abelacimab trials, is on an AF executive committee for BMS/Janssen (milvexian), and has been on an advisory board for Bayer (asundexian). Dr. Patel has received grants from and acts as an advisor to Bayer and Janssen. Dr. Alexander receives research funding from Bayer.
A version of this article first appeared on Medscape.com.
; the risk of stroke was moderate to high.
The trial was stopped earlier this year because of an “overwhelming” reduction in bleeding with abelacimab in comparison to rivaroxaban. Abelacimab is a monoclonal antibody given by subcutaneous injection once a month.
“Details of the bleeding results have now shown that the 150-mg dose of abelacimab, which is the dose being carried forward to phase 3 trials, was associated with a 67% reduction in major or clinically relevant nonmajor bleeding, the primary endpoint of the study.”
In addition, major bleeding was reduced by 74%, and major gastrointestinal bleeding was reduced by 93%.
“We are seeing really profound reductions in bleeding with this agent vs. a NOAC [novel oral anticoagulant],” lead AZALEA investigator Christian Ruff, MD, professor of medicine at Brigham and Women’s Hospital, Boston, said in an interview.
“Major bleeding – effectively the type of bleeding that results in hospitalization – is reduced by more than two-thirds, and major GI bleeding – which is the most common type of bleeding experienced by AF patients on anticoagulants – is almost eliminated. This gives us real hope that we have finally found an anticoagulant that is remarkably safe and will allow us to use anticoagulation in our most vulnerable patients,” he said.
Dr. Ruff presented the full results from the AZALEA trial at the American Heart Association scientific sessions.
He noted that AFib is one of the most common medical conditions in the world and that it confers an increased risk of stroke. Anticoagulants reduce this risk very effectively, and while the NOACS, such as apixaban and rivaroxaban, are safer than warfarin, significant bleeding still occurs, and “shockingly,” he said, between 30% and 60% of patients are not prescribed an anticoagulant or discontinue treatment because of bleeding concerns.
“Clearly, we need safer anticoagulants to protect these patients. Factor XI inhibitors, of which abelacimab is one, have emerged as the most promising agents, as they are thought to provide precision anticoagulation,” Dr. Ruff said.
He explained that factor XI appears to be involved in the formation of thrombus, which blocks arteries and causes strokes and myocardial infarction (thrombosis), but not in the healing process of blood vessels after injury (hemostasis). So, it is believed that inhibiting factor XI should reduce thrombotic events without causing excess bleeding.
AZALEA, which is the largest and longest trial of a factor XI inhibitor to date, enrolled 1,287 adults with AF who were at moderate to high risk of stroke.
They were randomly assigned to receive one of three treatments: oral rivaroxaban 20 mg daily; abelacimab 90 mg; or abelacimab 150 mg. Abelacimab was given monthly by injection.
Both doses of abelacimab inhibited factor XI almost completely; 97% inhibition was achieved with the 90-mg dose, and 99% inhibition was achieved with the 150-mg dose.
Results showed that after a median follow-up of 1.8 years, there was a clear reduction in all bleeding endpoints with both doses of abelacimab, compared with rivaroxaban.
Dr. Ruff explained that the trial was powered to detect differences in bleeding, not stroke, but the investigators approached this in an exploratory way.
“As expected, the numbers were low, with just 25 strokes (23 ischemic strokes) across all three groups in the trial. So, because of this very low rate, we are really not able to compare how abelacimab compares with rivaroxaban in reducing stroke,” he commented.
He did, however, suggest that the low stroke rate in the study was encouraging.
“If we look at the same population without anticoagulation, the stroke rate would be about 7% per year. And we see here in this trial that in all three arms, the stroke rate was just above 1% per year. I think this shows that all the patients in the trial were getting highly effective anticoagulation,” he said.
“But what this trial doesn’t answer – because the numbers are so low – is exactly how effective factor XI inhibition with abelacimab is, compared to NOACs in reducing stroke rates. That requires dedicated phase 3 trials.”
Dr. Ruff pointed out that there are some reassuring data from phase 2 trials in venous thromboembolism (VTE), in which the 150-mg dose of abelacimab was associated with an 80% reduction in VTE, compared with enoxaparin. “Historically in the development of anticoagulants, efficacy in VTE has translated into efficacy in stroke prevention, so that is very encouraging,” he commented.
“So, I think our results along with the VTE results are encouraging, but the precision regarding the relative efficacy compared to NOACs is still an open question that needs to be clarified in phase 3 trials,” he concluded.
Several phase 3 trials are now underway with abelacimab and two other small-molecule orally available factor XI inhibitors, milvexian (BMS/Janssen) and asundexian (Bayer).
The designated discussant of the AZALEA study at the AHA meeting, Manesh Patel. MD, Duke University, Durham, N.C., described the results as “an important step forward.”
“This trial, with the prior data in this field, show that factor XI inhibition as a target is biologically possible (studies showing > 95% inhibition), significantly less bleeding than NOACS. We await the phase 3 studies, but having significantly less bleeding and similar or less stroke would be a substantial step forward for the field,” he said.
John Alexander, MD, also from Duke University, said: “There were clinically important reductions in bleeding with both doses of abelacimab, compared with rivaroxaban. This is consistent to what we’ve seen with comparisons between other factor XI inhibitors and other factor Xa inhibitors.”
On the exploratory efficacy results, Dr. Alexander agreed with Dr. Ruff that it was not possible to get any idea of how abelacimab compared with rivaroxaban in reducing stroke. “The hazard ratio and confidence intervals comparing abelacimab and rivaroxaban include substantial lower rates, no difference, and substantially higher rates,” he noted.
“We need to wait for the results of phase 3 trials, with abelacimab and other factor XI inhibitors, to understand how well factor XI inhibition prevents stroke and systemic embolism in patients with atrial fibrillation,” Dr. Alexander added. “These trials are ongoing.”
Dr. Ruff concluded: “Assuming the data from ongoing phase 3 trials confirm the benefit of factor XI inhibitors for stroke prevention in people with AF, it will really be transformative for the field of cardiology.
“Our first mission in treating people with AF is to prevent stroke, and our ability to do this with a remarkably safe anticoagulant such as abelacimab would be an incredible advance,” he concluded.
Dr. Ruff receives research funding from Anthos for abelacimab trials, is on an AF executive committee for BMS/Janssen (milvexian), and has been on an advisory board for Bayer (asundexian). Dr. Patel has received grants from and acts as an advisor to Bayer and Janssen. Dr. Alexander receives research funding from Bayer.
A version of this article first appeared on Medscape.com.
; the risk of stroke was moderate to high.
The trial was stopped earlier this year because of an “overwhelming” reduction in bleeding with abelacimab in comparison to rivaroxaban. Abelacimab is a monoclonal antibody given by subcutaneous injection once a month.
“Details of the bleeding results have now shown that the 150-mg dose of abelacimab, which is the dose being carried forward to phase 3 trials, was associated with a 67% reduction in major or clinically relevant nonmajor bleeding, the primary endpoint of the study.”
In addition, major bleeding was reduced by 74%, and major gastrointestinal bleeding was reduced by 93%.
“We are seeing really profound reductions in bleeding with this agent vs. a NOAC [novel oral anticoagulant],” lead AZALEA investigator Christian Ruff, MD, professor of medicine at Brigham and Women’s Hospital, Boston, said in an interview.
“Major bleeding – effectively the type of bleeding that results in hospitalization – is reduced by more than two-thirds, and major GI bleeding – which is the most common type of bleeding experienced by AF patients on anticoagulants – is almost eliminated. This gives us real hope that we have finally found an anticoagulant that is remarkably safe and will allow us to use anticoagulation in our most vulnerable patients,” he said.
Dr. Ruff presented the full results from the AZALEA trial at the American Heart Association scientific sessions.
He noted that AFib is one of the most common medical conditions in the world and that it confers an increased risk of stroke. Anticoagulants reduce this risk very effectively, and while the NOACS, such as apixaban and rivaroxaban, are safer than warfarin, significant bleeding still occurs, and “shockingly,” he said, between 30% and 60% of patients are not prescribed an anticoagulant or discontinue treatment because of bleeding concerns.
“Clearly, we need safer anticoagulants to protect these patients. Factor XI inhibitors, of which abelacimab is one, have emerged as the most promising agents, as they are thought to provide precision anticoagulation,” Dr. Ruff said.
He explained that factor XI appears to be involved in the formation of thrombus, which blocks arteries and causes strokes and myocardial infarction (thrombosis), but not in the healing process of blood vessels after injury (hemostasis). So, it is believed that inhibiting factor XI should reduce thrombotic events without causing excess bleeding.
AZALEA, which is the largest and longest trial of a factor XI inhibitor to date, enrolled 1,287 adults with AF who were at moderate to high risk of stroke.
They were randomly assigned to receive one of three treatments: oral rivaroxaban 20 mg daily; abelacimab 90 mg; or abelacimab 150 mg. Abelacimab was given monthly by injection.
Both doses of abelacimab inhibited factor XI almost completely; 97% inhibition was achieved with the 90-mg dose, and 99% inhibition was achieved with the 150-mg dose.
Results showed that after a median follow-up of 1.8 years, there was a clear reduction in all bleeding endpoints with both doses of abelacimab, compared with rivaroxaban.
Dr. Ruff explained that the trial was powered to detect differences in bleeding, not stroke, but the investigators approached this in an exploratory way.
“As expected, the numbers were low, with just 25 strokes (23 ischemic strokes) across all three groups in the trial. So, because of this very low rate, we are really not able to compare how abelacimab compares with rivaroxaban in reducing stroke,” he commented.
He did, however, suggest that the low stroke rate in the study was encouraging.
“If we look at the same population without anticoagulation, the stroke rate would be about 7% per year. And we see here in this trial that in all three arms, the stroke rate was just above 1% per year. I think this shows that all the patients in the trial were getting highly effective anticoagulation,” he said.
“But what this trial doesn’t answer – because the numbers are so low – is exactly how effective factor XI inhibition with abelacimab is, compared to NOACs in reducing stroke rates. That requires dedicated phase 3 trials.”
Dr. Ruff pointed out that there are some reassuring data from phase 2 trials in venous thromboembolism (VTE), in which the 150-mg dose of abelacimab was associated with an 80% reduction in VTE, compared with enoxaparin. “Historically in the development of anticoagulants, efficacy in VTE has translated into efficacy in stroke prevention, so that is very encouraging,” he commented.
“So, I think our results along with the VTE results are encouraging, but the precision regarding the relative efficacy compared to NOACs is still an open question that needs to be clarified in phase 3 trials,” he concluded.
Several phase 3 trials are now underway with abelacimab and two other small-molecule orally available factor XI inhibitors, milvexian (BMS/Janssen) and asundexian (Bayer).
The designated discussant of the AZALEA study at the AHA meeting, Manesh Patel. MD, Duke University, Durham, N.C., described the results as “an important step forward.”
“This trial, with the prior data in this field, show that factor XI inhibition as a target is biologically possible (studies showing > 95% inhibition), significantly less bleeding than NOACS. We await the phase 3 studies, but having significantly less bleeding and similar or less stroke would be a substantial step forward for the field,” he said.
John Alexander, MD, also from Duke University, said: “There were clinically important reductions in bleeding with both doses of abelacimab, compared with rivaroxaban. This is consistent to what we’ve seen with comparisons between other factor XI inhibitors and other factor Xa inhibitors.”
On the exploratory efficacy results, Dr. Alexander agreed with Dr. Ruff that it was not possible to get any idea of how abelacimab compared with rivaroxaban in reducing stroke. “The hazard ratio and confidence intervals comparing abelacimab and rivaroxaban include substantial lower rates, no difference, and substantially higher rates,” he noted.
“We need to wait for the results of phase 3 trials, with abelacimab and other factor XI inhibitors, to understand how well factor XI inhibition prevents stroke and systemic embolism in patients with atrial fibrillation,” Dr. Alexander added. “These trials are ongoing.”
Dr. Ruff concluded: “Assuming the data from ongoing phase 3 trials confirm the benefit of factor XI inhibitors for stroke prevention in people with AF, it will really be transformative for the field of cardiology.
“Our first mission in treating people with AF is to prevent stroke, and our ability to do this with a remarkably safe anticoagulant such as abelacimab would be an incredible advance,” he concluded.
Dr. Ruff receives research funding from Anthos for abelacimab trials, is on an AF executive committee for BMS/Janssen (milvexian), and has been on an advisory board for Bayer (asundexian). Dr. Patel has received grants from and acts as an advisor to Bayer and Janssen. Dr. Alexander receives research funding from Bayer.
A version of this article first appeared on Medscape.com.
FROM AHA 2023
Apremilast beats placebo in early PsA affecting few joints
SAN DIEGO – Patients with early oligoarticular psoriatic arthritis (PsA) who took apremilast (Otezla) had more than double the response rate of placebo-treated patients by 16 weeks in a double-blind and randomized phase 4 study.
Oligoarticular PsA can significantly affect quality of life even though few joints are affected, and there’s a lack of relevant clinical data to guide treatment, said rheumatologist Philip J. Mease, MD, of the University of Washington and Swedish Medical Center, Seattle, who reported the results in a presentation at the annual meeting of the American College of Rheumatology.
The findings of the study, called FOREMOST, support the use of the drug in mild PsA, Alexis Ogdie, MD, director of the Penn Psoriatic Arthritis Clinic and the Penn Center for Clinical Epidemiology and Biostatistics at the University of Pennsylvania, Philadelphia, said in an interview. Dr. Ogdie, who was not involved with the research, noted that rheumatologists commonly prescribe apremilast for mild PsA, although previous research has focused on severe PsA cases.
By 16 weeks, 33.9% of 203 who received apremilast and 16% of 105 who received placebo (difference, 18.5%; 95% confidence interval, 8.9-28.1; P = .0008) met the trial’s primary outcome, a modified version of minimal disease activity score (MDA-Joints), which required attainment of 1 or fewer swollen and/or tender joints plus three of five additional criteria (psoriasis body surface area of 3% or less, a patient pain visual analog scale assessment of 15 mm or less out of 0-100 mm, a patient global assessment of 20 mm or less out of 0-100 mm, a Health Assessment Questionnaire-Disability Index score of 0.5 or less, and a Leeds Enthesitis Index score of 1 or less). The primary analysis was conducted only in joints affected at baseline.
The researchers recruited patients with 2-4 swollen and/or tender joints out of a total of 66-68 joints assessed; most patients (87%) randomized in the study had 4 or fewer active joints at baseline. The patients had a mean age of 50.9. The mean duration of PsA was 9.9 months, and 39.9% of patients were taking a conventional disease-modifying antirheumatic drug.
In a clinically important outcome, the percentage who had a patient-reported pain response improvement defined as “significant” reached 31.4% with placebo, compared with 48.8% for apremilast (difference, 17.7%; 95% CI, 6.0-29.4; P = .0044), and the percentage who reached a patient-reported pain response defined as “major” totaled 19.1% for placebo vs. 41.3% for apremilast (difference, 22.3%; 95% CI, 11.7-32.9; P = .002).
In an exploratory analysis of all joints, the percentages meeting MDA-Joints criteria for response were 7.9% with placebo and 21.3% with apremilast (difference, 13.6%; 95% CI, 5.9-21.4; P = .0028. Focusing on this exploratory analysis, Dr. Ogdie noted that examination of all joints is “more consistent” with the understanding of disease activity than only looking at the initial joints that had disease activity.
A post-hoc analysis among subjects with 2-4 affected joints found rates similar to the primary endpoint analysis: MDA-Joints response rates were reached by 34.4% of those who took apremilast and by 17.2% of those who took placebo.
When asked about the relatively low response rate for apremilast, Dr. Ogdie said the drug is “a really mild medication, which is why it belongs in the mild disease population. That’s balanced by the fact that it has a pretty good safety profile,” especially compared with the alternative of methotrexate, she said.
Almost all patients can tolerate apremilast, she said, although they may experience nausea or diarrhea. (The study found that adverse events were as expected for apremilast, and the drug was well tolerated.) Blood labs aren’t necessary, she added, as they are in patients taking methotrexate.
As for cost, apremilast is a highly expensive drug, especially when compared to methotrexate, which costs pennies per tablet at some pharmacies. Amgen, the manufacturer of apremilast, lists the price as $4,600 a month. Still, insurers generally cover apremilast, Dr. Ogdie said.
The study was sponsored by Amgen. Dr. Mease reported financial relationships with many pharmaceutical companies, including Amgen. Many other coauthors reported financial relationships with Amgen and other pharmaceutical companies or were employees of Amgen. Dr. Ogdie reported having multiple consulting relationships with pharmaceutical companies, including Amgen, and receiving grant funding from multiple companies as well as the National Institute of Arthritis and Musculoskeletal and Skin Diseases, National Psoriasis Foundation, Rheumatology Research Foundation, and Forward Databank.
SAN DIEGO – Patients with early oligoarticular psoriatic arthritis (PsA) who took apremilast (Otezla) had more than double the response rate of placebo-treated patients by 16 weeks in a double-blind and randomized phase 4 study.
Oligoarticular PsA can significantly affect quality of life even though few joints are affected, and there’s a lack of relevant clinical data to guide treatment, said rheumatologist Philip J. Mease, MD, of the University of Washington and Swedish Medical Center, Seattle, who reported the results in a presentation at the annual meeting of the American College of Rheumatology.
The findings of the study, called FOREMOST, support the use of the drug in mild PsA, Alexis Ogdie, MD, director of the Penn Psoriatic Arthritis Clinic and the Penn Center for Clinical Epidemiology and Biostatistics at the University of Pennsylvania, Philadelphia, said in an interview. Dr. Ogdie, who was not involved with the research, noted that rheumatologists commonly prescribe apremilast for mild PsA, although previous research has focused on severe PsA cases.
By 16 weeks, 33.9% of 203 who received apremilast and 16% of 105 who received placebo (difference, 18.5%; 95% confidence interval, 8.9-28.1; P = .0008) met the trial’s primary outcome, a modified version of minimal disease activity score (MDA-Joints), which required attainment of 1 or fewer swollen and/or tender joints plus three of five additional criteria (psoriasis body surface area of 3% or less, a patient pain visual analog scale assessment of 15 mm or less out of 0-100 mm, a patient global assessment of 20 mm or less out of 0-100 mm, a Health Assessment Questionnaire-Disability Index score of 0.5 or less, and a Leeds Enthesitis Index score of 1 or less). The primary analysis was conducted only in joints affected at baseline.
The researchers recruited patients with 2-4 swollen and/or tender joints out of a total of 66-68 joints assessed; most patients (87%) randomized in the study had 4 or fewer active joints at baseline. The patients had a mean age of 50.9. The mean duration of PsA was 9.9 months, and 39.9% of patients were taking a conventional disease-modifying antirheumatic drug.
In a clinically important outcome, the percentage who had a patient-reported pain response improvement defined as “significant” reached 31.4% with placebo, compared with 48.8% for apremilast (difference, 17.7%; 95% CI, 6.0-29.4; P = .0044), and the percentage who reached a patient-reported pain response defined as “major” totaled 19.1% for placebo vs. 41.3% for apremilast (difference, 22.3%; 95% CI, 11.7-32.9; P = .002).
In an exploratory analysis of all joints, the percentages meeting MDA-Joints criteria for response were 7.9% with placebo and 21.3% with apremilast (difference, 13.6%; 95% CI, 5.9-21.4; P = .0028. Focusing on this exploratory analysis, Dr. Ogdie noted that examination of all joints is “more consistent” with the understanding of disease activity than only looking at the initial joints that had disease activity.
A post-hoc analysis among subjects with 2-4 affected joints found rates similar to the primary endpoint analysis: MDA-Joints response rates were reached by 34.4% of those who took apremilast and by 17.2% of those who took placebo.
When asked about the relatively low response rate for apremilast, Dr. Ogdie said the drug is “a really mild medication, which is why it belongs in the mild disease population. That’s balanced by the fact that it has a pretty good safety profile,” especially compared with the alternative of methotrexate, she said.
Almost all patients can tolerate apremilast, she said, although they may experience nausea or diarrhea. (The study found that adverse events were as expected for apremilast, and the drug was well tolerated.) Blood labs aren’t necessary, she added, as they are in patients taking methotrexate.
As for cost, apremilast is a highly expensive drug, especially when compared to methotrexate, which costs pennies per tablet at some pharmacies. Amgen, the manufacturer of apremilast, lists the price as $4,600 a month. Still, insurers generally cover apremilast, Dr. Ogdie said.
The study was sponsored by Amgen. Dr. Mease reported financial relationships with many pharmaceutical companies, including Amgen. Many other coauthors reported financial relationships with Amgen and other pharmaceutical companies or were employees of Amgen. Dr. Ogdie reported having multiple consulting relationships with pharmaceutical companies, including Amgen, and receiving grant funding from multiple companies as well as the National Institute of Arthritis and Musculoskeletal and Skin Diseases, National Psoriasis Foundation, Rheumatology Research Foundation, and Forward Databank.
SAN DIEGO – Patients with early oligoarticular psoriatic arthritis (PsA) who took apremilast (Otezla) had more than double the response rate of placebo-treated patients by 16 weeks in a double-blind and randomized phase 4 study.
Oligoarticular PsA can significantly affect quality of life even though few joints are affected, and there’s a lack of relevant clinical data to guide treatment, said rheumatologist Philip J. Mease, MD, of the University of Washington and Swedish Medical Center, Seattle, who reported the results in a presentation at the annual meeting of the American College of Rheumatology.
The findings of the study, called FOREMOST, support the use of the drug in mild PsA, Alexis Ogdie, MD, director of the Penn Psoriatic Arthritis Clinic and the Penn Center for Clinical Epidemiology and Biostatistics at the University of Pennsylvania, Philadelphia, said in an interview. Dr. Ogdie, who was not involved with the research, noted that rheumatologists commonly prescribe apremilast for mild PsA, although previous research has focused on severe PsA cases.
By 16 weeks, 33.9% of 203 who received apremilast and 16% of 105 who received placebo (difference, 18.5%; 95% confidence interval, 8.9-28.1; P = .0008) met the trial’s primary outcome, a modified version of minimal disease activity score (MDA-Joints), which required attainment of 1 or fewer swollen and/or tender joints plus three of five additional criteria (psoriasis body surface area of 3% or less, a patient pain visual analog scale assessment of 15 mm or less out of 0-100 mm, a patient global assessment of 20 mm or less out of 0-100 mm, a Health Assessment Questionnaire-Disability Index score of 0.5 or less, and a Leeds Enthesitis Index score of 1 or less). The primary analysis was conducted only in joints affected at baseline.
The researchers recruited patients with 2-4 swollen and/or tender joints out of a total of 66-68 joints assessed; most patients (87%) randomized in the study had 4 or fewer active joints at baseline. The patients had a mean age of 50.9. The mean duration of PsA was 9.9 months, and 39.9% of patients were taking a conventional disease-modifying antirheumatic drug.
In a clinically important outcome, the percentage who had a patient-reported pain response improvement defined as “significant” reached 31.4% with placebo, compared with 48.8% for apremilast (difference, 17.7%; 95% CI, 6.0-29.4; P = .0044), and the percentage who reached a patient-reported pain response defined as “major” totaled 19.1% for placebo vs. 41.3% for apremilast (difference, 22.3%; 95% CI, 11.7-32.9; P = .002).
In an exploratory analysis of all joints, the percentages meeting MDA-Joints criteria for response were 7.9% with placebo and 21.3% with apremilast (difference, 13.6%; 95% CI, 5.9-21.4; P = .0028. Focusing on this exploratory analysis, Dr. Ogdie noted that examination of all joints is “more consistent” with the understanding of disease activity than only looking at the initial joints that had disease activity.
A post-hoc analysis among subjects with 2-4 affected joints found rates similar to the primary endpoint analysis: MDA-Joints response rates were reached by 34.4% of those who took apremilast and by 17.2% of those who took placebo.
When asked about the relatively low response rate for apremilast, Dr. Ogdie said the drug is “a really mild medication, which is why it belongs in the mild disease population. That’s balanced by the fact that it has a pretty good safety profile,” especially compared with the alternative of methotrexate, she said.
Almost all patients can tolerate apremilast, she said, although they may experience nausea or diarrhea. (The study found that adverse events were as expected for apremilast, and the drug was well tolerated.) Blood labs aren’t necessary, she added, as they are in patients taking methotrexate.
As for cost, apremilast is a highly expensive drug, especially when compared to methotrexate, which costs pennies per tablet at some pharmacies. Amgen, the manufacturer of apremilast, lists the price as $4,600 a month. Still, insurers generally cover apremilast, Dr. Ogdie said.
The study was sponsored by Amgen. Dr. Mease reported financial relationships with many pharmaceutical companies, including Amgen. Many other coauthors reported financial relationships with Amgen and other pharmaceutical companies or were employees of Amgen. Dr. Ogdie reported having multiple consulting relationships with pharmaceutical companies, including Amgen, and receiving grant funding from multiple companies as well as the National Institute of Arthritis and Musculoskeletal and Skin Diseases, National Psoriasis Foundation, Rheumatology Research Foundation, and Forward Databank.
AT ACR 2023
FDA’s Project Optimus aims to transform early cancer research
SAN DIEGO –
The goal is “to better identify and characterize optimized doses” in early stages of research and move away from the default of the traditional maximum tolerated dose strategy, hematologist-oncologist Marc R. Theoret, MD, deputy director of the FDA’s Oncology Center of Excellence, said in a presentation at the 2023 Society for Immunotherapy of Cancer annual meeting.
Earlier this year, the FDA released a draft guidance regarding the changes it hopes to see. The agency supported randomized, parallel dose-response trials when feasible, and “strong rationale for choice of dosage should be provided before initiating a registration trial(s) to support a subsequent indication and usage.”
The goal of controlling toxicity is “very highly important” in hematology research since blood cancer drugs can cause significant adverse effects in areas such as the lungs and heart, said Cecilia Yeung, MD, who led the SITC session about Project Optimus. Dr. Yeung is a clinical pathologist who works on investigational trials at Fred Hutchinson Cancer Research Center in Seattle.
In an interview, Dr. Yeung, who has a subspecialty in hematopathology, explained why the foundations of cancer research are changing and what hematologist-oncologists can expect to see on the horizon.
Q: Project Optimus aims to move beyond the traditional dose-escalation approach to the development of cancer drugs. How does that strategy work?
Dr. Yeung: Prior to Project Optimus, they’d use a 3+3 strategy in phase 1 trials: They’d give a dose to three fairly healthy patients, then they’d go up by escalating doses in more patients. They’d keep going up until two-thirds of patients at a specific dose suffered from bad side effects, then they’d back off to the last dose.
Q: This approach, which aims to identify the “maximum tolerated dose,” seemed to work well over decades of research into chemotherapy drugs. But worries arose as targeted therapies appeared in oncology areas such as blood cancer. Why did things change?
Dr. Yeung: With 3+3, you could tell pretty quickly how toxic chemotherapy was. But in targeted therapy, we were finding that these studies are not representative of actual toxicity. You’re not treating these patients for a very long time in phase 1, while patients on targeted therapy may be on these drugs for years. Concerns actually started with the first targeted drugs to treat leukemias and lymphomas. They were shown to have unexpected toxicity. A 2016 study found that drug developers had to reduce the original phase 1 dose in 45% of phase 3 trials [of small molecule and monoclonal antibody targeted agents] approved by the FDA over 12 years because of toxicity.
Q: What is FDA’s goal for Project Optimus?
Dr. Yeung: They want to have a second piece, to balance that maximum tolerated dose with a safe and tolerable dose for most people.
Q: What kind of resistance is the FDA getting from drug companies?
Dr. Yeung: The FDA makes a good argument that the system wasn’t working. But drug companies say this will drive up the cost of clinical trials and won’t allow them to treat patients with the maximal doses they could give them. I see arguments from both sides. There has to be a balance between the two.
Q: How will all this affect drug development?
Dr. Yeung: Drugs may become more expensive because much more testing will happen during clinical trials.
Q: Could this reduce the number of investigational drugs?
Dr. Yeung: Hopefully not, but this is huge endeavor for smaller companies that are strapped for funding.
Q: What do you think the future holds?
Dr. Yeung: Ultimately, this is a good thing because if everything works out, we’ll have fewer toxic side effects. But we’re going to have to go through a period of growing pains.
SAN DIEGO –
The goal is “to better identify and characterize optimized doses” in early stages of research and move away from the default of the traditional maximum tolerated dose strategy, hematologist-oncologist Marc R. Theoret, MD, deputy director of the FDA’s Oncology Center of Excellence, said in a presentation at the 2023 Society for Immunotherapy of Cancer annual meeting.
Earlier this year, the FDA released a draft guidance regarding the changes it hopes to see. The agency supported randomized, parallel dose-response trials when feasible, and “strong rationale for choice of dosage should be provided before initiating a registration trial(s) to support a subsequent indication and usage.”
The goal of controlling toxicity is “very highly important” in hematology research since blood cancer drugs can cause significant adverse effects in areas such as the lungs and heart, said Cecilia Yeung, MD, who led the SITC session about Project Optimus. Dr. Yeung is a clinical pathologist who works on investigational trials at Fred Hutchinson Cancer Research Center in Seattle.
In an interview, Dr. Yeung, who has a subspecialty in hematopathology, explained why the foundations of cancer research are changing and what hematologist-oncologists can expect to see on the horizon.
Q: Project Optimus aims to move beyond the traditional dose-escalation approach to the development of cancer drugs. How does that strategy work?
Dr. Yeung: Prior to Project Optimus, they’d use a 3+3 strategy in phase 1 trials: They’d give a dose to three fairly healthy patients, then they’d go up by escalating doses in more patients. They’d keep going up until two-thirds of patients at a specific dose suffered from bad side effects, then they’d back off to the last dose.
Q: This approach, which aims to identify the “maximum tolerated dose,” seemed to work well over decades of research into chemotherapy drugs. But worries arose as targeted therapies appeared in oncology areas such as blood cancer. Why did things change?
Dr. Yeung: With 3+3, you could tell pretty quickly how toxic chemotherapy was. But in targeted therapy, we were finding that these studies are not representative of actual toxicity. You’re not treating these patients for a very long time in phase 1, while patients on targeted therapy may be on these drugs for years. Concerns actually started with the first targeted drugs to treat leukemias and lymphomas. They were shown to have unexpected toxicity. A 2016 study found that drug developers had to reduce the original phase 1 dose in 45% of phase 3 trials [of small molecule and monoclonal antibody targeted agents] approved by the FDA over 12 years because of toxicity.
Q: What is FDA’s goal for Project Optimus?
Dr. Yeung: They want to have a second piece, to balance that maximum tolerated dose with a safe and tolerable dose for most people.
Q: What kind of resistance is the FDA getting from drug companies?
Dr. Yeung: The FDA makes a good argument that the system wasn’t working. But drug companies say this will drive up the cost of clinical trials and won’t allow them to treat patients with the maximal doses they could give them. I see arguments from both sides. There has to be a balance between the two.
Q: How will all this affect drug development?
Dr. Yeung: Drugs may become more expensive because much more testing will happen during clinical trials.
Q: Could this reduce the number of investigational drugs?
Dr. Yeung: Hopefully not, but this is huge endeavor for smaller companies that are strapped for funding.
Q: What do you think the future holds?
Dr. Yeung: Ultimately, this is a good thing because if everything works out, we’ll have fewer toxic side effects. But we’re going to have to go through a period of growing pains.
SAN DIEGO –
The goal is “to better identify and characterize optimized doses” in early stages of research and move away from the default of the traditional maximum tolerated dose strategy, hematologist-oncologist Marc R. Theoret, MD, deputy director of the FDA’s Oncology Center of Excellence, said in a presentation at the 2023 Society for Immunotherapy of Cancer annual meeting.
Earlier this year, the FDA released a draft guidance regarding the changes it hopes to see. The agency supported randomized, parallel dose-response trials when feasible, and “strong rationale for choice of dosage should be provided before initiating a registration trial(s) to support a subsequent indication and usage.”
The goal of controlling toxicity is “very highly important” in hematology research since blood cancer drugs can cause significant adverse effects in areas such as the lungs and heart, said Cecilia Yeung, MD, who led the SITC session about Project Optimus. Dr. Yeung is a clinical pathologist who works on investigational trials at Fred Hutchinson Cancer Research Center in Seattle.
In an interview, Dr. Yeung, who has a subspecialty in hematopathology, explained why the foundations of cancer research are changing and what hematologist-oncologists can expect to see on the horizon.
Q: Project Optimus aims to move beyond the traditional dose-escalation approach to the development of cancer drugs. How does that strategy work?
Dr. Yeung: Prior to Project Optimus, they’d use a 3+3 strategy in phase 1 trials: They’d give a dose to three fairly healthy patients, then they’d go up by escalating doses in more patients. They’d keep going up until two-thirds of patients at a specific dose suffered from bad side effects, then they’d back off to the last dose.
Q: This approach, which aims to identify the “maximum tolerated dose,” seemed to work well over decades of research into chemotherapy drugs. But worries arose as targeted therapies appeared in oncology areas such as blood cancer. Why did things change?
Dr. Yeung: With 3+3, you could tell pretty quickly how toxic chemotherapy was. But in targeted therapy, we were finding that these studies are not representative of actual toxicity. You’re not treating these patients for a very long time in phase 1, while patients on targeted therapy may be on these drugs for years. Concerns actually started with the first targeted drugs to treat leukemias and lymphomas. They were shown to have unexpected toxicity. A 2016 study found that drug developers had to reduce the original phase 1 dose in 45% of phase 3 trials [of small molecule and monoclonal antibody targeted agents] approved by the FDA over 12 years because of toxicity.
Q: What is FDA’s goal for Project Optimus?
Dr. Yeung: They want to have a second piece, to balance that maximum tolerated dose with a safe and tolerable dose for most people.
Q: What kind of resistance is the FDA getting from drug companies?
Dr. Yeung: The FDA makes a good argument that the system wasn’t working. But drug companies say this will drive up the cost of clinical trials and won’t allow them to treat patients with the maximal doses they could give them. I see arguments from both sides. There has to be a balance between the two.
Q: How will all this affect drug development?
Dr. Yeung: Drugs may become more expensive because much more testing will happen during clinical trials.
Q: Could this reduce the number of investigational drugs?
Dr. Yeung: Hopefully not, but this is huge endeavor for smaller companies that are strapped for funding.
Q: What do you think the future holds?
Dr. Yeung: Ultimately, this is a good thing because if everything works out, we’ll have fewer toxic side effects. But we’re going to have to go through a period of growing pains.
AT SITC 2023









